User login
Higher serum omega-3 tied to better outcome after STEMI
Regular consumption of foods rich in omega-3 fatty acids was associated with improved prognosis after ST-segment myocardial infarction (STEMI) in a new observational study.
The prospective study, which involved 944 patients with STEMI who underwent primary percutaneous coronary intervention (PCI), showed that plasma levels of fatty acids at the time of the STEMI were inversely associated with both incident major adverse cardiovascular events (MACE) and cardiovascular readmissions (adjusted hazard ratio, 0.76 and 0.74 for 1-SD increase; for both, P < .05).
No association was seen for the endpoint of all-cause mortality.
“What we showed is that your consumption of fish and other sources of omega-3 fatty acids before the heart attack impacts your prognosis after the heart attack. It’s a novel approach because it’s not primary prevention or secondary prevention,” said Aleix Sala-Vila, PharmD, PhD, from the Institut Hospital del Mar d’Investigacions Mèdiques (IMIM) in Barcelona, Spain.
Sala-Vila, co–senior author Antoni Bayés-Genís, MD, PhD, Heart Universitari Germans Trias I Pujol, Barcelona, and first author Iolanda Lázaro, PhD, also from IMIM, reported their findings online Oct. 26 in the Journal of the American College of Cardiology.
It has been established that dietary omega-3 eicosapentaenoic acid (EPA) has cardioprotective properties, but observational studies and randomized trials of EPA intake have yielded disparate findings.
This study avoided the usual traps of nutritional epidemiology research – self-reported food diaries and intake questionnaires. For this study, the researchers measured tissue levels of EPA and alpha-linolenic acid (ALA) by measuring serum phosphatidylcholine (PC) levels, which reflect dietary intake during the previous 3 or 4 weeks.
This technique, said Sala-Vila, not only provides a more reliable measure of fatty acid intake over time but also avoids measurement errors related to fatty acid content variation.
For example, “The EPA content of a piece of fish eaten in January could be very different from one eaten in June,” explained Sala-Vila.
That said, he acknowledged that this technique, which uses gas chromatography, does not at present have a clear clinical application. “It’s quite difficult just to convert levels of serum-PC EPA into consumption of fatty fish. We feel that the best advice at this point is that given by the American Heart Association to eat two servings of fatty fish a week.”
EPA and ALA: Partners in prevention?
In addition to the findings regarding EPA, the researchers also found that serum-PC ALA was inversely related to all-cause mortality after STEMI (HR, 0.65 for 1-SD increase; P < .05).
A trend was seen for an association between ALA and lower risk for incident MACE (P = .093).
ALA is readily available from inexpensive plant sources (eg, chia seeds, flax seeds, walnuts, soy beans) and has been associated with lower all-cause mortality in high-risk individuals.
This omega-3 fatty acid is often given short shrift in the fatty acid world because of the seven-step enzymatic process needed to convert it into more beneficial forms.
“We know that the conversion of ALA to EPA or DHA [docohexaenoic acid] is marginal, but we decided to include it in the study because we feel that this fatty acid is becoming more important because there are some issues with fish consumption – people are concerned about pollutants and sustainability, and some just don’t like it,” explained Sala-Vila.
“We were shocked to see that the marine-derived and vegetable-derived fatty acids don’t appear to compete, but rather they act synergistically,” said Sala-Villa. The researchers suggested that marine and vegetable omega-3 fatty acids may act as “partners in prevention.”
“We are not metabolically adapted to converting ALA to EPA, but despite this, there is a large body of evidence showing that one way to increase the status of EPA and DHA in our membranes is by eating these sources of fatty acids,” said Sala-Vila.
For almost 20 years, Sala-Vila has been studying how the consumption of foods rich in omega-3 affects disease. Two of his current projects involve studying levels of ALA in red blood cell membranes as a risk factor for ischemic stroke and omega-3 status in individuals with cognitive impairment who are at high risk for Alzheimer’s disease.
Applicable to all patients with atherosclerosis
In comments to theheart.org | Medscape Cardiology, Deepak Bhatt, MD, called the study “terrific,” adding that the effort is “as good as it gets” for observational nutrition research.
“I think one has to view these findings in the larger universe of what is really a revolution in omega-3 fatty acid research,” said Bhatt.
This universe, he said, includes a wealth of observational research showing the benefits of omega-3s, two outcome trials – JELIS and REDUCE-IT – that showed the benefits of EPA supplementation, and two imaging studies – EVAPORATE and CHERRY – that showed favorable effects of EPA on the vasculature.
REDUCE-IT, for which Bhatt served as principal investigator, showed that treatment with icosapent ethyl (Vascepa), a high-dose purified form of EPA, led to a 25% relative risk reduction in MACE in an at-risk Western population.
The results, said Bhatt, who co-wrote an editorial that accompanies the current Sala-Vila article, “likely apply to all patients with atherosclerosis or who are at high risk for it” and supports the practice of counseling patients to increase their intake of food rich in omega-3 fatty acids.
The field may be due for a shake-up, he noted. At next month’s American Heart Association meeting, the results of another trial of another prescription-grade EPA/DHA supplement will be presented, and they are expected to be negative.
AstraZeneca announced in January 2020 the early closure of the STRENGTH trial of Epanova after an interim analysis showed a low likelihood of their product demonstrating benefit in the enrolled population.
Epanova is a fish-oil derived mixture of free fatty acids, primarily EPA and DHA. It is approved in the United States and is indicated as an adjunct to diet to reduce triglyceride levels in adults with severe (≥500 mg/dL) hypertriglyceridemia. This indication is not affected by the data from the STRENGTH trial, according to a company press release.
Sala-Vila has received grants and support from the California Walnut Commission, including a grant to support part of this study. Bayés-Genís and Bhatt have relationships with a number of companies.
This article first appeared on Medscape.com.
Regular consumption of foods rich in omega-3 fatty acids was associated with improved prognosis after ST-segment myocardial infarction (STEMI) in a new observational study.
The prospective study, which involved 944 patients with STEMI who underwent primary percutaneous coronary intervention (PCI), showed that plasma levels of fatty acids at the time of the STEMI were inversely associated with both incident major adverse cardiovascular events (MACE) and cardiovascular readmissions (adjusted hazard ratio, 0.76 and 0.74 for 1-SD increase; for both, P < .05).
No association was seen for the endpoint of all-cause mortality.
“What we showed is that your consumption of fish and other sources of omega-3 fatty acids before the heart attack impacts your prognosis after the heart attack. It’s a novel approach because it’s not primary prevention or secondary prevention,” said Aleix Sala-Vila, PharmD, PhD, from the Institut Hospital del Mar d’Investigacions Mèdiques (IMIM) in Barcelona, Spain.
Sala-Vila, co–senior author Antoni Bayés-Genís, MD, PhD, Heart Universitari Germans Trias I Pujol, Barcelona, and first author Iolanda Lázaro, PhD, also from IMIM, reported their findings online Oct. 26 in the Journal of the American College of Cardiology.
It has been established that dietary omega-3 eicosapentaenoic acid (EPA) has cardioprotective properties, but observational studies and randomized trials of EPA intake have yielded disparate findings.
This study avoided the usual traps of nutritional epidemiology research – self-reported food diaries and intake questionnaires. For this study, the researchers measured tissue levels of EPA and alpha-linolenic acid (ALA) by measuring serum phosphatidylcholine (PC) levels, which reflect dietary intake during the previous 3 or 4 weeks.
This technique, said Sala-Vila, not only provides a more reliable measure of fatty acid intake over time but also avoids measurement errors related to fatty acid content variation.
For example, “The EPA content of a piece of fish eaten in January could be very different from one eaten in June,” explained Sala-Vila.
That said, he acknowledged that this technique, which uses gas chromatography, does not at present have a clear clinical application. “It’s quite difficult just to convert levels of serum-PC EPA into consumption of fatty fish. We feel that the best advice at this point is that given by the American Heart Association to eat two servings of fatty fish a week.”
EPA and ALA: Partners in prevention?
In addition to the findings regarding EPA, the researchers also found that serum-PC ALA was inversely related to all-cause mortality after STEMI (HR, 0.65 for 1-SD increase; P < .05).
A trend was seen for an association between ALA and lower risk for incident MACE (P = .093).
ALA is readily available from inexpensive plant sources (eg, chia seeds, flax seeds, walnuts, soy beans) and has been associated with lower all-cause mortality in high-risk individuals.
This omega-3 fatty acid is often given short shrift in the fatty acid world because of the seven-step enzymatic process needed to convert it into more beneficial forms.
“We know that the conversion of ALA to EPA or DHA [docohexaenoic acid] is marginal, but we decided to include it in the study because we feel that this fatty acid is becoming more important because there are some issues with fish consumption – people are concerned about pollutants and sustainability, and some just don’t like it,” explained Sala-Vila.
“We were shocked to see that the marine-derived and vegetable-derived fatty acids don’t appear to compete, but rather they act synergistically,” said Sala-Villa. The researchers suggested that marine and vegetable omega-3 fatty acids may act as “partners in prevention.”
“We are not metabolically adapted to converting ALA to EPA, but despite this, there is a large body of evidence showing that one way to increase the status of EPA and DHA in our membranes is by eating these sources of fatty acids,” said Sala-Vila.
For almost 20 years, Sala-Vila has been studying how the consumption of foods rich in omega-3 affects disease. Two of his current projects involve studying levels of ALA in red blood cell membranes as a risk factor for ischemic stroke and omega-3 status in individuals with cognitive impairment who are at high risk for Alzheimer’s disease.
Applicable to all patients with atherosclerosis
In comments to theheart.org | Medscape Cardiology, Deepak Bhatt, MD, called the study “terrific,” adding that the effort is “as good as it gets” for observational nutrition research.
“I think one has to view these findings in the larger universe of what is really a revolution in omega-3 fatty acid research,” said Bhatt.
This universe, he said, includes a wealth of observational research showing the benefits of omega-3s, two outcome trials – JELIS and REDUCE-IT – that showed the benefits of EPA supplementation, and two imaging studies – EVAPORATE and CHERRY – that showed favorable effects of EPA on the vasculature.
REDUCE-IT, for which Bhatt served as principal investigator, showed that treatment with icosapent ethyl (Vascepa), a high-dose purified form of EPA, led to a 25% relative risk reduction in MACE in an at-risk Western population.
The results, said Bhatt, who co-wrote an editorial that accompanies the current Sala-Vila article, “likely apply to all patients with atherosclerosis or who are at high risk for it” and supports the practice of counseling patients to increase their intake of food rich in omega-3 fatty acids.
The field may be due for a shake-up, he noted. At next month’s American Heart Association meeting, the results of another trial of another prescription-grade EPA/DHA supplement will be presented, and they are expected to be negative.
AstraZeneca announced in January 2020 the early closure of the STRENGTH trial of Epanova after an interim analysis showed a low likelihood of their product demonstrating benefit in the enrolled population.
Epanova is a fish-oil derived mixture of free fatty acids, primarily EPA and DHA. It is approved in the United States and is indicated as an adjunct to diet to reduce triglyceride levels in adults with severe (≥500 mg/dL) hypertriglyceridemia. This indication is not affected by the data from the STRENGTH trial, according to a company press release.
Sala-Vila has received grants and support from the California Walnut Commission, including a grant to support part of this study. Bayés-Genís and Bhatt have relationships with a number of companies.
This article first appeared on Medscape.com.
Regular consumption of foods rich in omega-3 fatty acids was associated with improved prognosis after ST-segment myocardial infarction (STEMI) in a new observational study.
The prospective study, which involved 944 patients with STEMI who underwent primary percutaneous coronary intervention (PCI), showed that plasma levels of fatty acids at the time of the STEMI were inversely associated with both incident major adverse cardiovascular events (MACE) and cardiovascular readmissions (adjusted hazard ratio, 0.76 and 0.74 for 1-SD increase; for both, P < .05).
No association was seen for the endpoint of all-cause mortality.
“What we showed is that your consumption of fish and other sources of omega-3 fatty acids before the heart attack impacts your prognosis after the heart attack. It’s a novel approach because it’s not primary prevention or secondary prevention,” said Aleix Sala-Vila, PharmD, PhD, from the Institut Hospital del Mar d’Investigacions Mèdiques (IMIM) in Barcelona, Spain.
Sala-Vila, co–senior author Antoni Bayés-Genís, MD, PhD, Heart Universitari Germans Trias I Pujol, Barcelona, and first author Iolanda Lázaro, PhD, also from IMIM, reported their findings online Oct. 26 in the Journal of the American College of Cardiology.
It has been established that dietary omega-3 eicosapentaenoic acid (EPA) has cardioprotective properties, but observational studies and randomized trials of EPA intake have yielded disparate findings.
This study avoided the usual traps of nutritional epidemiology research – self-reported food diaries and intake questionnaires. For this study, the researchers measured tissue levels of EPA and alpha-linolenic acid (ALA) by measuring serum phosphatidylcholine (PC) levels, which reflect dietary intake during the previous 3 or 4 weeks.
This technique, said Sala-Vila, not only provides a more reliable measure of fatty acid intake over time but also avoids measurement errors related to fatty acid content variation.
For example, “The EPA content of a piece of fish eaten in January could be very different from one eaten in June,” explained Sala-Vila.
That said, he acknowledged that this technique, which uses gas chromatography, does not at present have a clear clinical application. “It’s quite difficult just to convert levels of serum-PC EPA into consumption of fatty fish. We feel that the best advice at this point is that given by the American Heart Association to eat two servings of fatty fish a week.”
EPA and ALA: Partners in prevention?
In addition to the findings regarding EPA, the researchers also found that serum-PC ALA was inversely related to all-cause mortality after STEMI (HR, 0.65 for 1-SD increase; P < .05).
A trend was seen for an association between ALA and lower risk for incident MACE (P = .093).
ALA is readily available from inexpensive plant sources (eg, chia seeds, flax seeds, walnuts, soy beans) and has been associated with lower all-cause mortality in high-risk individuals.
This omega-3 fatty acid is often given short shrift in the fatty acid world because of the seven-step enzymatic process needed to convert it into more beneficial forms.
“We know that the conversion of ALA to EPA or DHA [docohexaenoic acid] is marginal, but we decided to include it in the study because we feel that this fatty acid is becoming more important because there are some issues with fish consumption – people are concerned about pollutants and sustainability, and some just don’t like it,” explained Sala-Vila.
“We were shocked to see that the marine-derived and vegetable-derived fatty acids don’t appear to compete, but rather they act synergistically,” said Sala-Villa. The researchers suggested that marine and vegetable omega-3 fatty acids may act as “partners in prevention.”
“We are not metabolically adapted to converting ALA to EPA, but despite this, there is a large body of evidence showing that one way to increase the status of EPA and DHA in our membranes is by eating these sources of fatty acids,” said Sala-Vila.
For almost 20 years, Sala-Vila has been studying how the consumption of foods rich in omega-3 affects disease. Two of his current projects involve studying levels of ALA in red blood cell membranes as a risk factor for ischemic stroke and omega-3 status in individuals with cognitive impairment who are at high risk for Alzheimer’s disease.
Applicable to all patients with atherosclerosis
In comments to theheart.org | Medscape Cardiology, Deepak Bhatt, MD, called the study “terrific,” adding that the effort is “as good as it gets” for observational nutrition research.
“I think one has to view these findings in the larger universe of what is really a revolution in omega-3 fatty acid research,” said Bhatt.
This universe, he said, includes a wealth of observational research showing the benefits of omega-3s, two outcome trials – JELIS and REDUCE-IT – that showed the benefits of EPA supplementation, and two imaging studies – EVAPORATE and CHERRY – that showed favorable effects of EPA on the vasculature.
REDUCE-IT, for which Bhatt served as principal investigator, showed that treatment with icosapent ethyl (Vascepa), a high-dose purified form of EPA, led to a 25% relative risk reduction in MACE in an at-risk Western population.
The results, said Bhatt, who co-wrote an editorial that accompanies the current Sala-Vila article, “likely apply to all patients with atherosclerosis or who are at high risk for it” and supports the practice of counseling patients to increase their intake of food rich in omega-3 fatty acids.
The field may be due for a shake-up, he noted. At next month’s American Heart Association meeting, the results of another trial of another prescription-grade EPA/DHA supplement will be presented, and they are expected to be negative.
AstraZeneca announced in January 2020 the early closure of the STRENGTH trial of Epanova after an interim analysis showed a low likelihood of their product demonstrating benefit in the enrolled population.
Epanova is a fish-oil derived mixture of free fatty acids, primarily EPA and DHA. It is approved in the United States and is indicated as an adjunct to diet to reduce triglyceride levels in adults with severe (≥500 mg/dL) hypertriglyceridemia. This indication is not affected by the data from the STRENGTH trial, according to a company press release.
Sala-Vila has received grants and support from the California Walnut Commission, including a grant to support part of this study. Bayés-Genís and Bhatt have relationships with a number of companies.
This article first appeared on Medscape.com.
Valvular disease and COVID-19 are a deadly mix; don’t delay intervention
Danny Dvir, MD, has a message for physicians who have patients with severe valvular heart disease who are deferring valve replacement or repair until after the COVID-19 pandemic: Urge them not to wait.
Data from the Multicenter International Valve Disease Registry vividly demonstrate that clinical outcomes are poor in patients with uncorrected valve disease who become hospitalized with COVID-19. Indeed, the mortality rate within 30 days after hospital admission in 136 such patients enrolled in the registry from centers in Europe, North America, and Israel was 42%, Dr. Dvir reported at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
“That’s dramatically higher than for an age-matched population infected with COVID-19 without valvular heart disease, which is 10%-15%,” he noted at the meeting sponsored by the Cardiovascular Research Foundation.
The bright spot was that, in the small subgroup of 15 registry participants who underwent transcatheter or, much less frequently, surgical treatment of their failing valve while COVID-19 infected, 30-day mortality was far lower. In fact, it was comparable with the background rate in hospitalized COVID-19 patients without valve disease, according to Dr. Dvir, an interventional cardiologist at Shaare Zedek Medical Center, Hebrew University, Jerusalem.
He personally did several of the transcatheter aortic valve replacements.
“It’s doable. I truly believe that when you get a severe aortic stenosis patient who’s infected with the coronavirus, they get very unstable, but we can treat them. We can treat them even during the infection,” Dr. Dvir said.
The majority of patients in the registry had severe aortic stenosis. In the 42 such patients aged 80 years or more who didn’t undergo transcatheter aortic valve replacement (TAVR) or surgical valve replacement, 30-day mortality was 60%. In contrast, only one of the six patients in this advanced-age category who underwent valve replacement while infected died. Similarly, 30-day mortality was 24% among those younger than age 80 who valve remained untreated, but it dropped to 11% in those who received a prosthetic valve.
“We try our best to protect our patients through social distancing, but we have a treatment that can potentially reduce their mortality risk if they get infected later on. So I say to my patients: ‘Don’t wait at home. Do not wait! If you get infected when you have severe aortic stenosis, the clinical outcome is bad.’ But it seems reasonable that if they get infected when they’ve already been treated for their aortic stenosis or mitral regurgitation, they will do better.”
Dr. Dvir noted that, although the case numbers in the registry series were small and subject to potential bias, the data suggest this treatment approach may be lifesaving.
Session comoderator Timothy D. Henry, MD, commented that this registry study contains a great take-home point: “This is really consistent with what see in a lot of the other areas of COVID, that what we know to be best clinical care, we should do it, with or without the COVID.”
He asked Dr. Dvir about any special measures he takes while doing TAVR in this extreme setting. In the United States, for example, interventionalists are increasingly using transesophageal echocardiography to guide their procedures using conscious sedation, without intubation, noted Dr. Henry, medical director of the Carl and Edyth Lindner Center for Research at the Christ Hospital, Cincinnati.
“We try to minimize the procedure time; that’s one of the important things,” Dr. Dvir replied. “And you need to be protected during the procedure in a very cautious and meticulous way. You need many fans in the room because you sweat a lot.”
Discussant Renu Virmani, MD, president of the CVPath Institute in Gaithersburg, Md., commented: “The main thing I get from this presentation is the need for patients to be educated that if you’ve got valve disease, you’re better off getting it treated before you’ve got COVID. Obviously, try to prevent getting COVID – that’s the best thing you can do – but you can’t always control that.”
Discussant Mamas Mamas, MD, professor of cardiology at Keele University, Staffordshire, England, said deferred treatment of severe valvular heart disease during the pandemic has created a looming public health crisis in the United Kingdom.
“We’ve analyzed the U.K. management of aortic stenosis, and what we’ve found is that during the COVID pandemic there have been 2,500 fewer cases of aortic stenosis that have been treated. We’ve got 2,500 patients on the waiting list, and we’ve got to work out how we’re going to treat them. We estimate with simulations that about 300 of them are going to die before we can get them treated for their aortic stenosis,” according to Dr. Mamas.
Dr. Henry commented that deferral of valve procedures is “really challenging” for a couple of reasons: Not only are patients scared to come into the hospital because they fear getting COVID, but they don’t want to be hospitalized during the pandemic because their family can’t visit them there.
“These patients are mostly over 80 years old. No one wants to come in the hospital when the family won’t be around, especially when you’re 90 years old,” the interventional cardiologist said.
Dr. Dvir reported serving as a consultant to Medtronic, Edwards Lifesciences, Abbott, and Jena.
Danny Dvir, MD, has a message for physicians who have patients with severe valvular heart disease who are deferring valve replacement or repair until after the COVID-19 pandemic: Urge them not to wait.
Data from the Multicenter International Valve Disease Registry vividly demonstrate that clinical outcomes are poor in patients with uncorrected valve disease who become hospitalized with COVID-19. Indeed, the mortality rate within 30 days after hospital admission in 136 such patients enrolled in the registry from centers in Europe, North America, and Israel was 42%, Dr. Dvir reported at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
“That’s dramatically higher than for an age-matched population infected with COVID-19 without valvular heart disease, which is 10%-15%,” he noted at the meeting sponsored by the Cardiovascular Research Foundation.
The bright spot was that, in the small subgroup of 15 registry participants who underwent transcatheter or, much less frequently, surgical treatment of their failing valve while COVID-19 infected, 30-day mortality was far lower. In fact, it was comparable with the background rate in hospitalized COVID-19 patients without valve disease, according to Dr. Dvir, an interventional cardiologist at Shaare Zedek Medical Center, Hebrew University, Jerusalem.
He personally did several of the transcatheter aortic valve replacements.
“It’s doable. I truly believe that when you get a severe aortic stenosis patient who’s infected with the coronavirus, they get very unstable, but we can treat them. We can treat them even during the infection,” Dr. Dvir said.
The majority of patients in the registry had severe aortic stenosis. In the 42 such patients aged 80 years or more who didn’t undergo transcatheter aortic valve replacement (TAVR) or surgical valve replacement, 30-day mortality was 60%. In contrast, only one of the six patients in this advanced-age category who underwent valve replacement while infected died. Similarly, 30-day mortality was 24% among those younger than age 80 who valve remained untreated, but it dropped to 11% in those who received a prosthetic valve.
“We try our best to protect our patients through social distancing, but we have a treatment that can potentially reduce their mortality risk if they get infected later on. So I say to my patients: ‘Don’t wait at home. Do not wait! If you get infected when you have severe aortic stenosis, the clinical outcome is bad.’ But it seems reasonable that if they get infected when they’ve already been treated for their aortic stenosis or mitral regurgitation, they will do better.”
Dr. Dvir noted that, although the case numbers in the registry series were small and subject to potential bias, the data suggest this treatment approach may be lifesaving.
Session comoderator Timothy D. Henry, MD, commented that this registry study contains a great take-home point: “This is really consistent with what see in a lot of the other areas of COVID, that what we know to be best clinical care, we should do it, with or without the COVID.”
He asked Dr. Dvir about any special measures he takes while doing TAVR in this extreme setting. In the United States, for example, interventionalists are increasingly using transesophageal echocardiography to guide their procedures using conscious sedation, without intubation, noted Dr. Henry, medical director of the Carl and Edyth Lindner Center for Research at the Christ Hospital, Cincinnati.
“We try to minimize the procedure time; that’s one of the important things,” Dr. Dvir replied. “And you need to be protected during the procedure in a very cautious and meticulous way. You need many fans in the room because you sweat a lot.”
Discussant Renu Virmani, MD, president of the CVPath Institute in Gaithersburg, Md., commented: “The main thing I get from this presentation is the need for patients to be educated that if you’ve got valve disease, you’re better off getting it treated before you’ve got COVID. Obviously, try to prevent getting COVID – that’s the best thing you can do – but you can’t always control that.”
Discussant Mamas Mamas, MD, professor of cardiology at Keele University, Staffordshire, England, said deferred treatment of severe valvular heart disease during the pandemic has created a looming public health crisis in the United Kingdom.
“We’ve analyzed the U.K. management of aortic stenosis, and what we’ve found is that during the COVID pandemic there have been 2,500 fewer cases of aortic stenosis that have been treated. We’ve got 2,500 patients on the waiting list, and we’ve got to work out how we’re going to treat them. We estimate with simulations that about 300 of them are going to die before we can get them treated for their aortic stenosis,” according to Dr. Mamas.
Dr. Henry commented that deferral of valve procedures is “really challenging” for a couple of reasons: Not only are patients scared to come into the hospital because they fear getting COVID, but they don’t want to be hospitalized during the pandemic because their family can’t visit them there.
“These patients are mostly over 80 years old. No one wants to come in the hospital when the family won’t be around, especially when you’re 90 years old,” the interventional cardiologist said.
Dr. Dvir reported serving as a consultant to Medtronic, Edwards Lifesciences, Abbott, and Jena.
Danny Dvir, MD, has a message for physicians who have patients with severe valvular heart disease who are deferring valve replacement or repair until after the COVID-19 pandemic: Urge them not to wait.
Data from the Multicenter International Valve Disease Registry vividly demonstrate that clinical outcomes are poor in patients with uncorrected valve disease who become hospitalized with COVID-19. Indeed, the mortality rate within 30 days after hospital admission in 136 such patients enrolled in the registry from centers in Europe, North America, and Israel was 42%, Dr. Dvir reported at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
“That’s dramatically higher than for an age-matched population infected with COVID-19 without valvular heart disease, which is 10%-15%,” he noted at the meeting sponsored by the Cardiovascular Research Foundation.
The bright spot was that, in the small subgroup of 15 registry participants who underwent transcatheter or, much less frequently, surgical treatment of their failing valve while COVID-19 infected, 30-day mortality was far lower. In fact, it was comparable with the background rate in hospitalized COVID-19 patients without valve disease, according to Dr. Dvir, an interventional cardiologist at Shaare Zedek Medical Center, Hebrew University, Jerusalem.
He personally did several of the transcatheter aortic valve replacements.
“It’s doable. I truly believe that when you get a severe aortic stenosis patient who’s infected with the coronavirus, they get very unstable, but we can treat them. We can treat them even during the infection,” Dr. Dvir said.
The majority of patients in the registry had severe aortic stenosis. In the 42 such patients aged 80 years or more who didn’t undergo transcatheter aortic valve replacement (TAVR) or surgical valve replacement, 30-day mortality was 60%. In contrast, only one of the six patients in this advanced-age category who underwent valve replacement while infected died. Similarly, 30-day mortality was 24% among those younger than age 80 who valve remained untreated, but it dropped to 11% in those who received a prosthetic valve.
“We try our best to protect our patients through social distancing, but we have a treatment that can potentially reduce their mortality risk if they get infected later on. So I say to my patients: ‘Don’t wait at home. Do not wait! If you get infected when you have severe aortic stenosis, the clinical outcome is bad.’ But it seems reasonable that if they get infected when they’ve already been treated for their aortic stenosis or mitral regurgitation, they will do better.”
Dr. Dvir noted that, although the case numbers in the registry series were small and subject to potential bias, the data suggest this treatment approach may be lifesaving.
Session comoderator Timothy D. Henry, MD, commented that this registry study contains a great take-home point: “This is really consistent with what see in a lot of the other areas of COVID, that what we know to be best clinical care, we should do it, with or without the COVID.”
He asked Dr. Dvir about any special measures he takes while doing TAVR in this extreme setting. In the United States, for example, interventionalists are increasingly using transesophageal echocardiography to guide their procedures using conscious sedation, without intubation, noted Dr. Henry, medical director of the Carl and Edyth Lindner Center for Research at the Christ Hospital, Cincinnati.
“We try to minimize the procedure time; that’s one of the important things,” Dr. Dvir replied. “And you need to be protected during the procedure in a very cautious and meticulous way. You need many fans in the room because you sweat a lot.”
Discussant Renu Virmani, MD, president of the CVPath Institute in Gaithersburg, Md., commented: “The main thing I get from this presentation is the need for patients to be educated that if you’ve got valve disease, you’re better off getting it treated before you’ve got COVID. Obviously, try to prevent getting COVID – that’s the best thing you can do – but you can’t always control that.”
Discussant Mamas Mamas, MD, professor of cardiology at Keele University, Staffordshire, England, said deferred treatment of severe valvular heart disease during the pandemic has created a looming public health crisis in the United Kingdom.
“We’ve analyzed the U.K. management of aortic stenosis, and what we’ve found is that during the COVID pandemic there have been 2,500 fewer cases of aortic stenosis that have been treated. We’ve got 2,500 patients on the waiting list, and we’ve got to work out how we’re going to treat them. We estimate with simulations that about 300 of them are going to die before we can get them treated for their aortic stenosis,” according to Dr. Mamas.
Dr. Henry commented that deferral of valve procedures is “really challenging” for a couple of reasons: Not only are patients scared to come into the hospital because they fear getting COVID, but they don’t want to be hospitalized during the pandemic because their family can’t visit them there.
“These patients are mostly over 80 years old. No one wants to come in the hospital when the family won’t be around, especially when you’re 90 years old,” the interventional cardiologist said.
Dr. Dvir reported serving as a consultant to Medtronic, Edwards Lifesciences, Abbott, and Jena.
FROM TCT 2020
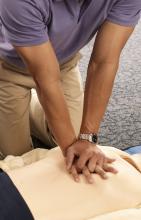
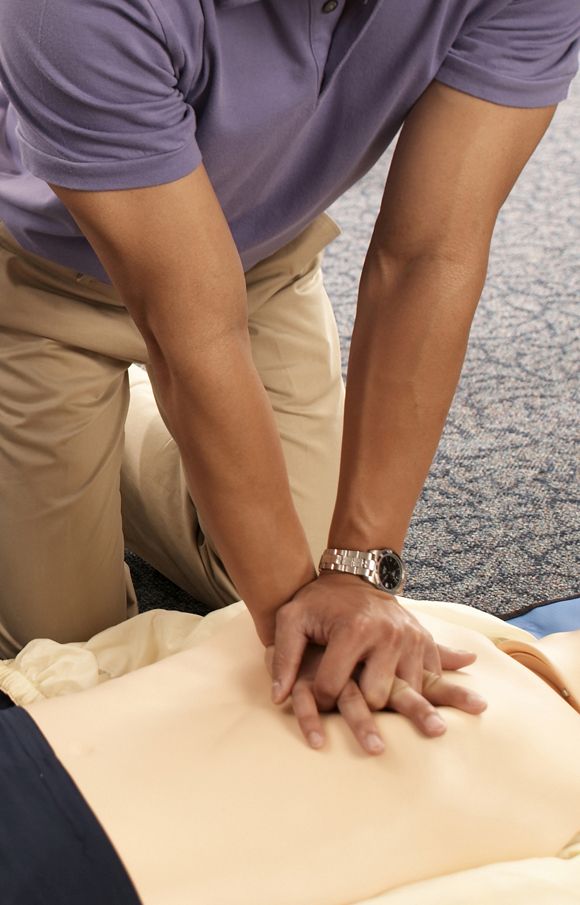
AHA adds recovery, emotional support to CPR guidelines
Highlights of new updated guidelines for cardiopulmonary resuscitation and emergency cardiovascular care from the American Heart Association include management of opioid-related emergencies; discussion of health disparities; and a new emphasis on physical, social, and emotional recovery after resuscitation.
The AHA is also exploring digital territory to improve CPR outcomes. The guidelines encourage use of mobile phone technology to summon trained laypeople to individuals requiring CPR, and an adaptive learning suite will be available online for personalized CPR instruction, with lessons catered to individual needs and knowledge levels.
These novel approaches reflect an ongoing effort by the AHA to ensure that the guidelines evolve rapidly with science and technology, reported Raina Merchant, MD, chair of the AHA Emergency Cardiovascular Care Committee and associate professor of emergency medicine at the University of Pennsylvania, Philadelphia, and colleagues. In 2015, the committee shifted from 5-year updates to a continuous online review process, citing a need for more immediate implementation of practice-altering data, they wrote in Circulation.
And new approaches do appear to save lives, at least in a hospital setting.
Since 2004, in-hospital cardiac arrest outcomes have been improving, but similar gains have yet to be realized for out-of-hospital cardiac arrest.
“Much of the variation in survival rates is thought to be due to the strength of the Chain of Survival, the [five] critical actions that must occur in rapid succession to maximize the chance of survival from cardiac arrest,” the committee wrote.
Update adds sixth link to Chains of Survival: Recovery
“Recovery expectations and survivorship plans that address treatment, surveillance, and rehabilitation need to be provided to cardiac arrest survivors and their caregivers at hospital discharge to address the sequelae of cardiac arrest and optimize transitions of care to independent physical, social, emotional, and role function,” the committee wrote.
Dr. Merchant and colleagues identified three “critically important” recommendations for both cardiac arrest survivors and caregivers during the recovery process: structured psychological assessment; multimodal rehabilitation assessment and treatment; and comprehensive, multidisciplinary discharge planning.
The recovery process is now part of all four Chains of Survival, which are specific to in-hospital and out-of-hospital arrest for adults and children.
New advice on opioid overdoses and bystander training
Among instances of out-of-hospital cardiac arrest, the committee noted that opioid overdoses are “sharply on the rise,” leading to new, scenario-specific recommendations. Among them, the committee encouraged lay rescuers and trained responders to activate emergency response systems immediately while awaiting improvements with naloxone and other interventions. They also suggested that, for individuals in known or suspected cardiac arrest, high-quality CPR, including compressions and ventilation, should be prioritized over naloxone administration.
In a broader discussion, the committee identified disparities in CPR training, which could explain lower rates of bystander CPR and poorer outcomes among certain demographics, such as black and Hispanic populations, as well as those with lower socioeconomic status.
“Targeting training efforts should consider barriers such as language, financial considerations, and poor access to information,” the committee wrote.
While low bystander CPR in these areas may be improved through mobile phone technology that alerts trained laypeople to individuals in need, the committee noted that this approach may be impacted by cultural and geographic factors. To date, use of mobile devices to improve bystander intervention rates has been demonstrated through “uniformly positive data,” but never in North America.
According to the guidelines, bystander intervention rates may also be improved through video-based learning, which is as effective as in-person, instructor-led training.
This led the AHA to create an online adaptive learning platform, which the organization describes as a “digital resuscitation portfolio” that connects programs and courses such as the Resuscitation Quality Improvement program and the HeartCode blended learning course.
“It will cover all of the guideline changes,” said Monica Sales, communications manager at the AHA. “It’s really groundbreaking because it’s the first time that we’re able to kind of close that gap between new science and new products.”
The online content also addresses CPR considerations for COVID-19, which were first addressed by interim CPR guidance published by the AHA in April.
According to Alexis Topjian, MD, coauthor of the present guidelines and pediatric critical care medicine physician at Children’s Hospital of Philadelphia, CPR awareness is more important now than ever.
“The major message [of the guidelines] is that high-quality CPR saves lives,” she said. “So push hard, and push fast. You have the power in your hands to make a difference, more so than ever during this pandemic.”
Concerning coronavirus precautions, Dr. Topjian noted that roughly 70% of out-of-hospital CPR events involve people who know each other, so most bystanders have already been exposed to the person in need, thereby reducing the concern of infection.
When asked about performing CPR on strangers, Dr. Topjian remained encouraging, though she noted that decision making may be informed by local coronavirus rates.
“It’s always a personal choice,” she said.
More for clinicians
For clinicians, Dr. Topjian highlighted several recommendations, including use of epinephrine as soon as possible during CPR, preferential use of a cuffed endotracheal tube, continuous EEG monitoring during and after cardiac arrest, and rapid intervention for clinical seizures and of nonconvulsive status epilepticus.
From a pediatric perspective, Dr. Topjian pointed out a change in breathing rate for infants and children who are receiving CPR or rescue breathing with a pulse, from 12-20 breaths/min to 20-30 breaths/min. While not a new recommendation, Dr. Topjian also pointed out the lifesaving benefit of early defibrillation among pediatric patients.
The guidelines were funded by the American Heart Association. The investigators disclosed additional relationships with BTG Pharmaceuticals, Zoll Foundation, the National Institutes of Health, and others.
SOURCE: American Heart Association. Circulation. 2020 Oct 20. Suppl 2.
Highlights of new updated guidelines for cardiopulmonary resuscitation and emergency cardiovascular care from the American Heart Association include management of opioid-related emergencies; discussion of health disparities; and a new emphasis on physical, social, and emotional recovery after resuscitation.
The AHA is also exploring digital territory to improve CPR outcomes. The guidelines encourage use of mobile phone technology to summon trained laypeople to individuals requiring CPR, and an adaptive learning suite will be available online for personalized CPR instruction, with lessons catered to individual needs and knowledge levels.
These novel approaches reflect an ongoing effort by the AHA to ensure that the guidelines evolve rapidly with science and technology, reported Raina Merchant, MD, chair of the AHA Emergency Cardiovascular Care Committee and associate professor of emergency medicine at the University of Pennsylvania, Philadelphia, and colleagues. In 2015, the committee shifted from 5-year updates to a continuous online review process, citing a need for more immediate implementation of practice-altering data, they wrote in Circulation.
And new approaches do appear to save lives, at least in a hospital setting.
Since 2004, in-hospital cardiac arrest outcomes have been improving, but similar gains have yet to be realized for out-of-hospital cardiac arrest.
“Much of the variation in survival rates is thought to be due to the strength of the Chain of Survival, the [five] critical actions that must occur in rapid succession to maximize the chance of survival from cardiac arrest,” the committee wrote.
Update adds sixth link to Chains of Survival: Recovery
“Recovery expectations and survivorship plans that address treatment, surveillance, and rehabilitation need to be provided to cardiac arrest survivors and their caregivers at hospital discharge to address the sequelae of cardiac arrest and optimize transitions of care to independent physical, social, emotional, and role function,” the committee wrote.
Dr. Merchant and colleagues identified three “critically important” recommendations for both cardiac arrest survivors and caregivers during the recovery process: structured psychological assessment; multimodal rehabilitation assessment and treatment; and comprehensive, multidisciplinary discharge planning.
The recovery process is now part of all four Chains of Survival, which are specific to in-hospital and out-of-hospital arrest for adults and children.
New advice on opioid overdoses and bystander training
Among instances of out-of-hospital cardiac arrest, the committee noted that opioid overdoses are “sharply on the rise,” leading to new, scenario-specific recommendations. Among them, the committee encouraged lay rescuers and trained responders to activate emergency response systems immediately while awaiting improvements with naloxone and other interventions. They also suggested that, for individuals in known or suspected cardiac arrest, high-quality CPR, including compressions and ventilation, should be prioritized over naloxone administration.
In a broader discussion, the committee identified disparities in CPR training, which could explain lower rates of bystander CPR and poorer outcomes among certain demographics, such as black and Hispanic populations, as well as those with lower socioeconomic status.
“Targeting training efforts should consider barriers such as language, financial considerations, and poor access to information,” the committee wrote.
While low bystander CPR in these areas may be improved through mobile phone technology that alerts trained laypeople to individuals in need, the committee noted that this approach may be impacted by cultural and geographic factors. To date, use of mobile devices to improve bystander intervention rates has been demonstrated through “uniformly positive data,” but never in North America.
According to the guidelines, bystander intervention rates may also be improved through video-based learning, which is as effective as in-person, instructor-led training.
This led the AHA to create an online adaptive learning platform, which the organization describes as a “digital resuscitation portfolio” that connects programs and courses such as the Resuscitation Quality Improvement program and the HeartCode blended learning course.
“It will cover all of the guideline changes,” said Monica Sales, communications manager at the AHA. “It’s really groundbreaking because it’s the first time that we’re able to kind of close that gap between new science and new products.”
The online content also addresses CPR considerations for COVID-19, which were first addressed by interim CPR guidance published by the AHA in April.
According to Alexis Topjian, MD, coauthor of the present guidelines and pediatric critical care medicine physician at Children’s Hospital of Philadelphia, CPR awareness is more important now than ever.
“The major message [of the guidelines] is that high-quality CPR saves lives,” she said. “So push hard, and push fast. You have the power in your hands to make a difference, more so than ever during this pandemic.”
Concerning coronavirus precautions, Dr. Topjian noted that roughly 70% of out-of-hospital CPR events involve people who know each other, so most bystanders have already been exposed to the person in need, thereby reducing the concern of infection.
When asked about performing CPR on strangers, Dr. Topjian remained encouraging, though she noted that decision making may be informed by local coronavirus rates.
“It’s always a personal choice,” she said.
More for clinicians
For clinicians, Dr. Topjian highlighted several recommendations, including use of epinephrine as soon as possible during CPR, preferential use of a cuffed endotracheal tube, continuous EEG monitoring during and after cardiac arrest, and rapid intervention for clinical seizures and of nonconvulsive status epilepticus.
From a pediatric perspective, Dr. Topjian pointed out a change in breathing rate for infants and children who are receiving CPR or rescue breathing with a pulse, from 12-20 breaths/min to 20-30 breaths/min. While not a new recommendation, Dr. Topjian also pointed out the lifesaving benefit of early defibrillation among pediatric patients.
The guidelines were funded by the American Heart Association. The investigators disclosed additional relationships with BTG Pharmaceuticals, Zoll Foundation, the National Institutes of Health, and others.
SOURCE: American Heart Association. Circulation. 2020 Oct 20. Suppl 2.
Highlights of new updated guidelines for cardiopulmonary resuscitation and emergency cardiovascular care from the American Heart Association include management of opioid-related emergencies; discussion of health disparities; and a new emphasis on physical, social, and emotional recovery after resuscitation.
The AHA is also exploring digital territory to improve CPR outcomes. The guidelines encourage use of mobile phone technology to summon trained laypeople to individuals requiring CPR, and an adaptive learning suite will be available online for personalized CPR instruction, with lessons catered to individual needs and knowledge levels.
These novel approaches reflect an ongoing effort by the AHA to ensure that the guidelines evolve rapidly with science and technology, reported Raina Merchant, MD, chair of the AHA Emergency Cardiovascular Care Committee and associate professor of emergency medicine at the University of Pennsylvania, Philadelphia, and colleagues. In 2015, the committee shifted from 5-year updates to a continuous online review process, citing a need for more immediate implementation of practice-altering data, they wrote in Circulation.
And new approaches do appear to save lives, at least in a hospital setting.
Since 2004, in-hospital cardiac arrest outcomes have been improving, but similar gains have yet to be realized for out-of-hospital cardiac arrest.
“Much of the variation in survival rates is thought to be due to the strength of the Chain of Survival, the [five] critical actions that must occur in rapid succession to maximize the chance of survival from cardiac arrest,” the committee wrote.
Update adds sixth link to Chains of Survival: Recovery
“Recovery expectations and survivorship plans that address treatment, surveillance, and rehabilitation need to be provided to cardiac arrest survivors and their caregivers at hospital discharge to address the sequelae of cardiac arrest and optimize transitions of care to independent physical, social, emotional, and role function,” the committee wrote.
Dr. Merchant and colleagues identified three “critically important” recommendations for both cardiac arrest survivors and caregivers during the recovery process: structured psychological assessment; multimodal rehabilitation assessment and treatment; and comprehensive, multidisciplinary discharge planning.
The recovery process is now part of all four Chains of Survival, which are specific to in-hospital and out-of-hospital arrest for adults and children.
New advice on opioid overdoses and bystander training
Among instances of out-of-hospital cardiac arrest, the committee noted that opioid overdoses are “sharply on the rise,” leading to new, scenario-specific recommendations. Among them, the committee encouraged lay rescuers and trained responders to activate emergency response systems immediately while awaiting improvements with naloxone and other interventions. They also suggested that, for individuals in known or suspected cardiac arrest, high-quality CPR, including compressions and ventilation, should be prioritized over naloxone administration.
In a broader discussion, the committee identified disparities in CPR training, which could explain lower rates of bystander CPR and poorer outcomes among certain demographics, such as black and Hispanic populations, as well as those with lower socioeconomic status.
“Targeting training efforts should consider barriers such as language, financial considerations, and poor access to information,” the committee wrote.
While low bystander CPR in these areas may be improved through mobile phone technology that alerts trained laypeople to individuals in need, the committee noted that this approach may be impacted by cultural and geographic factors. To date, use of mobile devices to improve bystander intervention rates has been demonstrated through “uniformly positive data,” but never in North America.
According to the guidelines, bystander intervention rates may also be improved through video-based learning, which is as effective as in-person, instructor-led training.
This led the AHA to create an online adaptive learning platform, which the organization describes as a “digital resuscitation portfolio” that connects programs and courses such as the Resuscitation Quality Improvement program and the HeartCode blended learning course.
“It will cover all of the guideline changes,” said Monica Sales, communications manager at the AHA. “It’s really groundbreaking because it’s the first time that we’re able to kind of close that gap between new science and new products.”
The online content also addresses CPR considerations for COVID-19, which were first addressed by interim CPR guidance published by the AHA in April.
According to Alexis Topjian, MD, coauthor of the present guidelines and pediatric critical care medicine physician at Children’s Hospital of Philadelphia, CPR awareness is more important now than ever.
“The major message [of the guidelines] is that high-quality CPR saves lives,” she said. “So push hard, and push fast. You have the power in your hands to make a difference, more so than ever during this pandemic.”
Concerning coronavirus precautions, Dr. Topjian noted that roughly 70% of out-of-hospital CPR events involve people who know each other, so most bystanders have already been exposed to the person in need, thereby reducing the concern of infection.
When asked about performing CPR on strangers, Dr. Topjian remained encouraging, though she noted that decision making may be informed by local coronavirus rates.
“It’s always a personal choice,” she said.
More for clinicians
For clinicians, Dr. Topjian highlighted several recommendations, including use of epinephrine as soon as possible during CPR, preferential use of a cuffed endotracheal tube, continuous EEG monitoring during and after cardiac arrest, and rapid intervention for clinical seizures and of nonconvulsive status epilepticus.
From a pediatric perspective, Dr. Topjian pointed out a change in breathing rate for infants and children who are receiving CPR or rescue breathing with a pulse, from 12-20 breaths/min to 20-30 breaths/min. While not a new recommendation, Dr. Topjian also pointed out the lifesaving benefit of early defibrillation among pediatric patients.
The guidelines were funded by the American Heart Association. The investigators disclosed additional relationships with BTG Pharmaceuticals, Zoll Foundation, the National Institutes of Health, and others.
SOURCE: American Heart Association. Circulation. 2020 Oct 20. Suppl 2.
FROM CIRCULATION
Artificially sweetened drinks add to CVD risk
Sugary and artificially sweetened drinks are each associated with an increased risk of developing cardiovascular disease, according to results from a large prospective cohort study.
However, the design of that study fails to take into account other sources of dietary sugar, according to one expert.
In a research letter published online Oct. 26 in the Journal of the American College of Cardiology, Eloi Chazelas, a PhD candidate at Sorbonne Paris Nord University in Paris, and colleagues, shared results from nearly 105,000 subjects (79% women, mean age 43 at baseline, median follow up 6.6 years) enrolled in the NutriNet-Santé cohort study.
In this observational study, which began recruiting in 2009, dietary patterns are self-reported by subjects, while health outcomes are validated by investigators.
Mr. Chazelas and his colleagues identified 1,379 first incident cases of stroke, transient ischemic attack, myocardial infarction, acute coronary syndrome, and angioplasty in the cohort during 2009-2019. Cases that occurred during the first 3 years’ follow up were excluded from the analysis, to avoid potential reverse causality bias.
After adjustment for a wide range of dietary, demographic and health confounders, the investigators found that high consumers of sugary drinks or artificially sweetened drinks saw 20% and 32% higher risk of such events, respectively, compared with people who reported drinking neither beverage type (hazard ratio: 1.20; 95% confidence interval 1.04-1.40, P for trend < .0009 and HR: 1.32; 95% CI, 1.00-1.73, P for trend < .03).
Sugary drinks were defined as containing 5% or more of sugars, including natural fruit juices. The high consumers in the study had a median intake of 185 mL per day of sugary drinks, or 176 mL per day for artificially sweetened drinks. Natural noncaloric sweeteners such as Stevia were included in the artificially sweetened group.
The findings, Mr. Chazelas and colleagues wrote in their analysis, add to evidence that artificially sweetened beverages “might not be a healthy substitute for sugary drinks.” While research has suggested that artificial sweeteners induce glucose intolerance by disturbing gut microbiota, they noted, more and bigger studies are needed to understand the mechanisms by which they might bear on cardiovascular disease risk.
Robert A. Vogel, MD, of the University of Colorado Denver, urged caution in interpreting the researchers’ results. In an interview, Dr. Vogel, a preventive cardiologist, said that it is “notoriously difficult” to evaluate what a food or food group does to the body outside of a carefully controlled trial. What little randomized trial evidence exists comparing the health effects of artificially sweetened and sugary drinks includes a 2012 trial in children that found diet drinks associated with reductions in body fat – if anything a positive indication for heart health.
With adults enrolled in an observational study, things are much more easily confounded, Dr. Vogel said. “So subjects self-report that they’re not consuming one thing – sugary or sweetened beverages. What else are they putting into their diet? Maybe they’re eating dessert and consuming sugar that way. Try as you will to unconfound, to do a multivariate correction for all these factors is just very difficult.”
In addition, Dr. Vogel noted, the investigators made no attempt to discern among the different sweeteners consumed. “Stevia, saccharine, Sucralose – it’s highly unlikely that each of these agents has the same effect on gut microbiota.”
In 2019, researchers led by Mr. Chazelas looked at cancer risk in high consumers of the sugary and artificially sweetened drinks in some 107,000 patients from the cohort, and reported that sugary drinks were significantly associated with the risk of overall cancer. They saw no similar association for artificially sweetened drinks.
The NutriNet-Santé study is funded by the French government, and the investigators disclosed no financial support from commercial entities. Dr. Vogel has received research support from Sanofi and speaking fees from Regeneron.
Sugary and artificially sweetened drinks are each associated with an increased risk of developing cardiovascular disease, according to results from a large prospective cohort study.
However, the design of that study fails to take into account other sources of dietary sugar, according to one expert.
In a research letter published online Oct. 26 in the Journal of the American College of Cardiology, Eloi Chazelas, a PhD candidate at Sorbonne Paris Nord University in Paris, and colleagues, shared results from nearly 105,000 subjects (79% women, mean age 43 at baseline, median follow up 6.6 years) enrolled in the NutriNet-Santé cohort study.
In this observational study, which began recruiting in 2009, dietary patterns are self-reported by subjects, while health outcomes are validated by investigators.
Mr. Chazelas and his colleagues identified 1,379 first incident cases of stroke, transient ischemic attack, myocardial infarction, acute coronary syndrome, and angioplasty in the cohort during 2009-2019. Cases that occurred during the first 3 years’ follow up were excluded from the analysis, to avoid potential reverse causality bias.
After adjustment for a wide range of dietary, demographic and health confounders, the investigators found that high consumers of sugary drinks or artificially sweetened drinks saw 20% and 32% higher risk of such events, respectively, compared with people who reported drinking neither beverage type (hazard ratio: 1.20; 95% confidence interval 1.04-1.40, P for trend < .0009 and HR: 1.32; 95% CI, 1.00-1.73, P for trend < .03).
Sugary drinks were defined as containing 5% or more of sugars, including natural fruit juices. The high consumers in the study had a median intake of 185 mL per day of sugary drinks, or 176 mL per day for artificially sweetened drinks. Natural noncaloric sweeteners such as Stevia were included in the artificially sweetened group.
The findings, Mr. Chazelas and colleagues wrote in their analysis, add to evidence that artificially sweetened beverages “might not be a healthy substitute for sugary drinks.” While research has suggested that artificial sweeteners induce glucose intolerance by disturbing gut microbiota, they noted, more and bigger studies are needed to understand the mechanisms by which they might bear on cardiovascular disease risk.
Robert A. Vogel, MD, of the University of Colorado Denver, urged caution in interpreting the researchers’ results. In an interview, Dr. Vogel, a preventive cardiologist, said that it is “notoriously difficult” to evaluate what a food or food group does to the body outside of a carefully controlled trial. What little randomized trial evidence exists comparing the health effects of artificially sweetened and sugary drinks includes a 2012 trial in children that found diet drinks associated with reductions in body fat – if anything a positive indication for heart health.
With adults enrolled in an observational study, things are much more easily confounded, Dr. Vogel said. “So subjects self-report that they’re not consuming one thing – sugary or sweetened beverages. What else are they putting into their diet? Maybe they’re eating dessert and consuming sugar that way. Try as you will to unconfound, to do a multivariate correction for all these factors is just very difficult.”
In addition, Dr. Vogel noted, the investigators made no attempt to discern among the different sweeteners consumed. “Stevia, saccharine, Sucralose – it’s highly unlikely that each of these agents has the same effect on gut microbiota.”
In 2019, researchers led by Mr. Chazelas looked at cancer risk in high consumers of the sugary and artificially sweetened drinks in some 107,000 patients from the cohort, and reported that sugary drinks were significantly associated with the risk of overall cancer. They saw no similar association for artificially sweetened drinks.
The NutriNet-Santé study is funded by the French government, and the investigators disclosed no financial support from commercial entities. Dr. Vogel has received research support from Sanofi and speaking fees from Regeneron.
Sugary and artificially sweetened drinks are each associated with an increased risk of developing cardiovascular disease, according to results from a large prospective cohort study.
However, the design of that study fails to take into account other sources of dietary sugar, according to one expert.
In a research letter published online Oct. 26 in the Journal of the American College of Cardiology, Eloi Chazelas, a PhD candidate at Sorbonne Paris Nord University in Paris, and colleagues, shared results from nearly 105,000 subjects (79% women, mean age 43 at baseline, median follow up 6.6 years) enrolled in the NutriNet-Santé cohort study.
In this observational study, which began recruiting in 2009, dietary patterns are self-reported by subjects, while health outcomes are validated by investigators.
Mr. Chazelas and his colleagues identified 1,379 first incident cases of stroke, transient ischemic attack, myocardial infarction, acute coronary syndrome, and angioplasty in the cohort during 2009-2019. Cases that occurred during the first 3 years’ follow up were excluded from the analysis, to avoid potential reverse causality bias.
After adjustment for a wide range of dietary, demographic and health confounders, the investigators found that high consumers of sugary drinks or artificially sweetened drinks saw 20% and 32% higher risk of such events, respectively, compared with people who reported drinking neither beverage type (hazard ratio: 1.20; 95% confidence interval 1.04-1.40, P for trend < .0009 and HR: 1.32; 95% CI, 1.00-1.73, P for trend < .03).
Sugary drinks were defined as containing 5% or more of sugars, including natural fruit juices. The high consumers in the study had a median intake of 185 mL per day of sugary drinks, or 176 mL per day for artificially sweetened drinks. Natural noncaloric sweeteners such as Stevia were included in the artificially sweetened group.
The findings, Mr. Chazelas and colleagues wrote in their analysis, add to evidence that artificially sweetened beverages “might not be a healthy substitute for sugary drinks.” While research has suggested that artificial sweeteners induce glucose intolerance by disturbing gut microbiota, they noted, more and bigger studies are needed to understand the mechanisms by which they might bear on cardiovascular disease risk.
Robert A. Vogel, MD, of the University of Colorado Denver, urged caution in interpreting the researchers’ results. In an interview, Dr. Vogel, a preventive cardiologist, said that it is “notoriously difficult” to evaluate what a food or food group does to the body outside of a carefully controlled trial. What little randomized trial evidence exists comparing the health effects of artificially sweetened and sugary drinks includes a 2012 trial in children that found diet drinks associated with reductions in body fat – if anything a positive indication for heart health.
With adults enrolled in an observational study, things are much more easily confounded, Dr. Vogel said. “So subjects self-report that they’re not consuming one thing – sugary or sweetened beverages. What else are they putting into their diet? Maybe they’re eating dessert and consuming sugar that way. Try as you will to unconfound, to do a multivariate correction for all these factors is just very difficult.”
In addition, Dr. Vogel noted, the investigators made no attempt to discern among the different sweeteners consumed. “Stevia, saccharine, Sucralose – it’s highly unlikely that each of these agents has the same effect on gut microbiota.”
In 2019, researchers led by Mr. Chazelas looked at cancer risk in high consumers of the sugary and artificially sweetened drinks in some 107,000 patients from the cohort, and reported that sugary drinks were significantly associated with the risk of overall cancer. They saw no similar association for artificially sweetened drinks.
The NutriNet-Santé study is funded by the French government, and the investigators disclosed no financial support from commercial entities. Dr. Vogel has received research support from Sanofi and speaking fees from Regeneron.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
When should students resume sports after a COVID-19 diagnosis?
Many student athletes who test positive for COVID-19 likely can have an uneventful return to their sports after they have rested for 2 weeks in quarantine, doctors suggest.
There are reasons for caution, however, especially when a patient has symptoms that indicate possible cardiac involvement. In these cases, patients should undergo cardiac testing before a physician clears them to return to play, according to guidance from professional associations. Reports of myocarditis in college athletes who tested positive for SARS-CoV-2 but were asymptomatic are among the reasons for concern. Myocarditis may increase the risk of sudden death during exercise.
“The thing that you need to keep in mind is that this is not just a respiratory illness,” David T. Bernhardt, MD, professor of pediatrics, orthopedics, and rehabilitation at the University of Wisconsin in Madison, said in a presentation at the annual meeting of the American Academy of Pediatrics, held virtually this year. High school and college athletes have had cardiac, neurologic, hematologic, and renal problems that “can complicate their recovery and their return to sport.”
Still, children who test positive for COVID-19 tend to have mild illness and often are asymptomatic. “It is more than likely going to be safe for the majority of the student athletes who are in the elementary and middle school age to return to sport,” said Dr. Bernhardt. Given that 18-year-old college freshmen have had cardiac complications, there may be reason for more caution with high school students.
Limited data
The AAP has released interim guidance on returning to sports and recommends that primary care physicians clear all patients with COVID-19 before they resume training. Physicians should screen for cardiac symptoms such as chest pain, shortness of breath, fatigue, palpitations, or syncope.
Those with severe illness should be restricted from exercise and participation for 3-6 months. Primary care physicians, preferably in consultation with pediatric cardiologists, should clear athletes who experience severe illness.
“Most of the recommendations come from the fact that we simply do not know what we do not know with COVID-19,” Susannah Briskin, MD, a coauthor of the interim guidance, said in an interview. “We have to be cautious in returning individuals to play and closely monitor them as we learn more about the disease process and its effect on kids.”
Patients with severe illness could include those who were hospitalized and experienced hypotension or arrhythmias, required intubation or extracorporeal membrane oxygenation (ECMO) support, had kidney or cardiac failure, or developed multisystem inflammatory syndrome in children (MIS-C), said Dr. Briskin, a specialist in pediatric sports medicine at Case Western Reserve University, Cleveland.
“The majority of COVID-19 cases will not present like this in kids. We have no idea how common myocarditis is in kids post infection. We do know that, if anyone has chest pain, shortness of breath, excessive fatigue, syncope [passing out], or arrhythmia [feeling of their heart skipping beats], they should undergo further evaluation for myocarditis,” Dr. Briskin said.
Patients who are asymptomatic or have mild symptoms should rest for 14 days after their positive test. After their infectious period has passed, a doctor should assess for any concerning cardiac symptoms. “Anyone with prolonged fever or moderate symptoms should see their pediatrician and have an EKG performed, at a minimum, prior to return to sports,” Dr. Briskin said. “Anyone with an abnormal EKG or concerning signs or symptoms should be referred on to pediatric cardiology for a further assessment.”
Most patients who Dr. Briskin has seen have been asymptomatic or mildly symptomatic. “They have done well with a gradual return to physical activity,” she said. “We recommend a gradual return so individuals can be monitored for any signs or symptoms concerning for myocarditis. The far majority of individuals likely have an uneventful return to play.”
Mitigating risk
COVID-19 adds elements of uncertainty and complexity to the usual process of mitigating risk in sports, Dr. Bernhardt noted in his lecture. “You are dealing with an infection that we do not know a lot about,” he said. “And we are trying to mitigate risk not only for the individual who may or may not have underlying health problems, but you are also trying to mitigate risk for anybody else involved with the sport, including athletic trainers and team physicians, coaches, spectators, custodial staff, people working at a snack shack, and all the other people that can be involved in a typical sporting type of atmosphere.”
When patients do return to play after an illness, they should gradually increase the training load to avoid injury. In addition, clinicians should screen for depression and anxiety using tools such as the Four-Item Patient Health Questionnaire (PHQ-4) when they see patients. “The pandemic has been quite stressful for everybody, including our high school student athletes,” Dr. Bernhardt said. “Giving everybody a PHQ-4 when they come into clinic right now probably makes sense in terms of the stress levels that all of us are experiencing.”
If a patient screens positive, take additional history and refer for more in-depth mental health evaluation and treatment if warranted. Sharing breathing and relaxation exercises, promoting healthy behaviors, and paying attention to unhealthy strategies also may help, Dr. Bernhardt suggested.
Ultimately, determining when an athlete with COVID-19 can be medically cleared to return to play may be a challenge. There are limited data on epidemiology and clinical presentations that could help identify cardiac injury related to the disease, Dr. Bernhardt said. Guidance from the American College of Cardiology provides a framework for evaluating athletes for return to play, and pediatric cardiologists have discussed how the guidance relates to a pediatric population. Cardiac assessments may include measures of biomarkers such as troponin, B-type natriuretic peptide, and sedimentation rate, along with electrocardiograms, echocardiograms, and cardiac MRI.
Beyond return-to-play decisions, encourage the use of cloth face coverings on the sidelines and away from the playing field, and stress proper quarantining, Dr. Briskin added. Too often, she hears about children not quarantining properly. “Individuals with a known exposure should be quarantined in their house – ideally in a separate room from everyone else. ... When they come out of their room, they should wash their hands well and wear a cloth face covering. They should not be eating with other people.”
Dr. Bernhardt had no relevant disclosures.
Many student athletes who test positive for COVID-19 likely can have an uneventful return to their sports after they have rested for 2 weeks in quarantine, doctors suggest.
There are reasons for caution, however, especially when a patient has symptoms that indicate possible cardiac involvement. In these cases, patients should undergo cardiac testing before a physician clears them to return to play, according to guidance from professional associations. Reports of myocarditis in college athletes who tested positive for SARS-CoV-2 but were asymptomatic are among the reasons for concern. Myocarditis may increase the risk of sudden death during exercise.
“The thing that you need to keep in mind is that this is not just a respiratory illness,” David T. Bernhardt, MD, professor of pediatrics, orthopedics, and rehabilitation at the University of Wisconsin in Madison, said in a presentation at the annual meeting of the American Academy of Pediatrics, held virtually this year. High school and college athletes have had cardiac, neurologic, hematologic, and renal problems that “can complicate their recovery and their return to sport.”
Still, children who test positive for COVID-19 tend to have mild illness and often are asymptomatic. “It is more than likely going to be safe for the majority of the student athletes who are in the elementary and middle school age to return to sport,” said Dr. Bernhardt. Given that 18-year-old college freshmen have had cardiac complications, there may be reason for more caution with high school students.
Limited data
The AAP has released interim guidance on returning to sports and recommends that primary care physicians clear all patients with COVID-19 before they resume training. Physicians should screen for cardiac symptoms such as chest pain, shortness of breath, fatigue, palpitations, or syncope.
Those with severe illness should be restricted from exercise and participation for 3-6 months. Primary care physicians, preferably in consultation with pediatric cardiologists, should clear athletes who experience severe illness.
“Most of the recommendations come from the fact that we simply do not know what we do not know with COVID-19,” Susannah Briskin, MD, a coauthor of the interim guidance, said in an interview. “We have to be cautious in returning individuals to play and closely monitor them as we learn more about the disease process and its effect on kids.”
Patients with severe illness could include those who were hospitalized and experienced hypotension or arrhythmias, required intubation or extracorporeal membrane oxygenation (ECMO) support, had kidney or cardiac failure, or developed multisystem inflammatory syndrome in children (MIS-C), said Dr. Briskin, a specialist in pediatric sports medicine at Case Western Reserve University, Cleveland.
“The majority of COVID-19 cases will not present like this in kids. We have no idea how common myocarditis is in kids post infection. We do know that, if anyone has chest pain, shortness of breath, excessive fatigue, syncope [passing out], or arrhythmia [feeling of their heart skipping beats], they should undergo further evaluation for myocarditis,” Dr. Briskin said.
Patients who are asymptomatic or have mild symptoms should rest for 14 days after their positive test. After their infectious period has passed, a doctor should assess for any concerning cardiac symptoms. “Anyone with prolonged fever or moderate symptoms should see their pediatrician and have an EKG performed, at a minimum, prior to return to sports,” Dr. Briskin said. “Anyone with an abnormal EKG or concerning signs or symptoms should be referred on to pediatric cardiology for a further assessment.”
Most patients who Dr. Briskin has seen have been asymptomatic or mildly symptomatic. “They have done well with a gradual return to physical activity,” she said. “We recommend a gradual return so individuals can be monitored for any signs or symptoms concerning for myocarditis. The far majority of individuals likely have an uneventful return to play.”
Mitigating risk
COVID-19 adds elements of uncertainty and complexity to the usual process of mitigating risk in sports, Dr. Bernhardt noted in his lecture. “You are dealing with an infection that we do not know a lot about,” he said. “And we are trying to mitigate risk not only for the individual who may or may not have underlying health problems, but you are also trying to mitigate risk for anybody else involved with the sport, including athletic trainers and team physicians, coaches, spectators, custodial staff, people working at a snack shack, and all the other people that can be involved in a typical sporting type of atmosphere.”
When patients do return to play after an illness, they should gradually increase the training load to avoid injury. In addition, clinicians should screen for depression and anxiety using tools such as the Four-Item Patient Health Questionnaire (PHQ-4) when they see patients. “The pandemic has been quite stressful for everybody, including our high school student athletes,” Dr. Bernhardt said. “Giving everybody a PHQ-4 when they come into clinic right now probably makes sense in terms of the stress levels that all of us are experiencing.”
If a patient screens positive, take additional history and refer for more in-depth mental health evaluation and treatment if warranted. Sharing breathing and relaxation exercises, promoting healthy behaviors, and paying attention to unhealthy strategies also may help, Dr. Bernhardt suggested.
Ultimately, determining when an athlete with COVID-19 can be medically cleared to return to play may be a challenge. There are limited data on epidemiology and clinical presentations that could help identify cardiac injury related to the disease, Dr. Bernhardt said. Guidance from the American College of Cardiology provides a framework for evaluating athletes for return to play, and pediatric cardiologists have discussed how the guidance relates to a pediatric population. Cardiac assessments may include measures of biomarkers such as troponin, B-type natriuretic peptide, and sedimentation rate, along with electrocardiograms, echocardiograms, and cardiac MRI.
Beyond return-to-play decisions, encourage the use of cloth face coverings on the sidelines and away from the playing field, and stress proper quarantining, Dr. Briskin added. Too often, she hears about children not quarantining properly. “Individuals with a known exposure should be quarantined in their house – ideally in a separate room from everyone else. ... When they come out of their room, they should wash their hands well and wear a cloth face covering. They should not be eating with other people.”
Dr. Bernhardt had no relevant disclosures.
Many student athletes who test positive for COVID-19 likely can have an uneventful return to their sports after they have rested for 2 weeks in quarantine, doctors suggest.
There are reasons for caution, however, especially when a patient has symptoms that indicate possible cardiac involvement. In these cases, patients should undergo cardiac testing before a physician clears them to return to play, according to guidance from professional associations. Reports of myocarditis in college athletes who tested positive for SARS-CoV-2 but were asymptomatic are among the reasons for concern. Myocarditis may increase the risk of sudden death during exercise.
“The thing that you need to keep in mind is that this is not just a respiratory illness,” David T. Bernhardt, MD, professor of pediatrics, orthopedics, and rehabilitation at the University of Wisconsin in Madison, said in a presentation at the annual meeting of the American Academy of Pediatrics, held virtually this year. High school and college athletes have had cardiac, neurologic, hematologic, and renal problems that “can complicate their recovery and their return to sport.”
Still, children who test positive for COVID-19 tend to have mild illness and often are asymptomatic. “It is more than likely going to be safe for the majority of the student athletes who are in the elementary and middle school age to return to sport,” said Dr. Bernhardt. Given that 18-year-old college freshmen have had cardiac complications, there may be reason for more caution with high school students.
Limited data
The AAP has released interim guidance on returning to sports and recommends that primary care physicians clear all patients with COVID-19 before they resume training. Physicians should screen for cardiac symptoms such as chest pain, shortness of breath, fatigue, palpitations, or syncope.
Those with severe illness should be restricted from exercise and participation for 3-6 months. Primary care physicians, preferably in consultation with pediatric cardiologists, should clear athletes who experience severe illness.
“Most of the recommendations come from the fact that we simply do not know what we do not know with COVID-19,” Susannah Briskin, MD, a coauthor of the interim guidance, said in an interview. “We have to be cautious in returning individuals to play and closely monitor them as we learn more about the disease process and its effect on kids.”
Patients with severe illness could include those who were hospitalized and experienced hypotension or arrhythmias, required intubation or extracorporeal membrane oxygenation (ECMO) support, had kidney or cardiac failure, or developed multisystem inflammatory syndrome in children (MIS-C), said Dr. Briskin, a specialist in pediatric sports medicine at Case Western Reserve University, Cleveland.
“The majority of COVID-19 cases will not present like this in kids. We have no idea how common myocarditis is in kids post infection. We do know that, if anyone has chest pain, shortness of breath, excessive fatigue, syncope [passing out], or arrhythmia [feeling of their heart skipping beats], they should undergo further evaluation for myocarditis,” Dr. Briskin said.
Patients who are asymptomatic or have mild symptoms should rest for 14 days after their positive test. After their infectious period has passed, a doctor should assess for any concerning cardiac symptoms. “Anyone with prolonged fever or moderate symptoms should see their pediatrician and have an EKG performed, at a minimum, prior to return to sports,” Dr. Briskin said. “Anyone with an abnormal EKG or concerning signs or symptoms should be referred on to pediatric cardiology for a further assessment.”
Most patients who Dr. Briskin has seen have been asymptomatic or mildly symptomatic. “They have done well with a gradual return to physical activity,” she said. “We recommend a gradual return so individuals can be monitored for any signs or symptoms concerning for myocarditis. The far majority of individuals likely have an uneventful return to play.”
Mitigating risk
COVID-19 adds elements of uncertainty and complexity to the usual process of mitigating risk in sports, Dr. Bernhardt noted in his lecture. “You are dealing with an infection that we do not know a lot about,” he said. “And we are trying to mitigate risk not only for the individual who may or may not have underlying health problems, but you are also trying to mitigate risk for anybody else involved with the sport, including athletic trainers and team physicians, coaches, spectators, custodial staff, people working at a snack shack, and all the other people that can be involved in a typical sporting type of atmosphere.”
When patients do return to play after an illness, they should gradually increase the training load to avoid injury. In addition, clinicians should screen for depression and anxiety using tools such as the Four-Item Patient Health Questionnaire (PHQ-4) when they see patients. “The pandemic has been quite stressful for everybody, including our high school student athletes,” Dr. Bernhardt said. “Giving everybody a PHQ-4 when they come into clinic right now probably makes sense in terms of the stress levels that all of us are experiencing.”
If a patient screens positive, take additional history and refer for more in-depth mental health evaluation and treatment if warranted. Sharing breathing and relaxation exercises, promoting healthy behaviors, and paying attention to unhealthy strategies also may help, Dr. Bernhardt suggested.
Ultimately, determining when an athlete with COVID-19 can be medically cleared to return to play may be a challenge. There are limited data on epidemiology and clinical presentations that could help identify cardiac injury related to the disease, Dr. Bernhardt said. Guidance from the American College of Cardiology provides a framework for evaluating athletes for return to play, and pediatric cardiologists have discussed how the guidance relates to a pediatric population. Cardiac assessments may include measures of biomarkers such as troponin, B-type natriuretic peptide, and sedimentation rate, along with electrocardiograms, echocardiograms, and cardiac MRI.
Beyond return-to-play decisions, encourage the use of cloth face coverings on the sidelines and away from the playing field, and stress proper quarantining, Dr. Briskin added. Too often, she hears about children not quarantining properly. “Individuals with a known exposure should be quarantined in their house – ideally in a separate room from everyone else. ... When they come out of their room, they should wash their hands well and wear a cloth face covering. They should not be eating with other people.”
Dr. Bernhardt had no relevant disclosures.
FROM AAP 2020
No mortality benefit after intensive glucose control once Hb A1c curves equalize
Background: A previous study reported that a median of 5.6 years of intensive versus standard glucose lowering in veterans with type 2 diabetes resulted in significantly reduced risk of major cardiovascular events after 10 years of combined intervention and observational follow-up.
Study design: Prospective cohort.
Setting: Veterans Affairs Healthcare System.
Synopsis: In the original trial, 1,791 veterans were randomly assigned to receive either intensive or standard glucose control therapy. After conclusion of that study, 1,655 participants were followed using central databases, and 1,391 also provided data via surveys and chart review. Initially the difference in the glycated hemoglobin (Hb A1c) curves between the two groups averaged 1.5%, but it declined to 0.2%-0.3% 3 years after the trial ended. The median Hb A1c then stabilized to 8% in both groups.
Over a period of 15 years of combined intervention and posttrial follow-up, the risks of major cardiovascular events or death were not lower in the intensive-therapy group (hazard ratio for composite outcome, 0.91; 95% confidence interval, 0.78-1.06; P = .23; HR for death, 1.02; 95% CI, 0.88-1.18). The risk of major cardiovascular disease outcomes was reduced during the approximately 10-year interval of separation of the Hb A1c curves (HR, 0.83; 95% CI, 0.70-0.99), but it did not persist after equalization of Hb A1c levels (HR, 1.26; 95% CI, 0.90-1.75). Limitations include the observational study design, the study population of mostly older men, and reliance on administrative data for outcomes.
Bottom line: More than 5 years of intensive glucose lowering, compared with standard therapy, did not show significantly lower risks of cardiovascular events or mortality once the glycated hemoglobin curves equalized during follow-up in years 11-15.
Citation: Reaven PD et al. Intensive glucose control in patients with type 2 diabetes – 15-year follow-up. New Engl J Med. 2019 Jun 6;380(23):2215-24.
Dr. Burke is a hospitalist at Vanderbilt University Medical Center, Nashville, Tenn.
Background: A previous study reported that a median of 5.6 years of intensive versus standard glucose lowering in veterans with type 2 diabetes resulted in significantly reduced risk of major cardiovascular events after 10 years of combined intervention and observational follow-up.
Study design: Prospective cohort.
Setting: Veterans Affairs Healthcare System.
Synopsis: In the original trial, 1,791 veterans were randomly assigned to receive either intensive or standard glucose control therapy. After conclusion of that study, 1,655 participants were followed using central databases, and 1,391 also provided data via surveys and chart review. Initially the difference in the glycated hemoglobin (Hb A1c) curves between the two groups averaged 1.5%, but it declined to 0.2%-0.3% 3 years after the trial ended. The median Hb A1c then stabilized to 8% in both groups.
Over a period of 15 years of combined intervention and posttrial follow-up, the risks of major cardiovascular events or death were not lower in the intensive-therapy group (hazard ratio for composite outcome, 0.91; 95% confidence interval, 0.78-1.06; P = .23; HR for death, 1.02; 95% CI, 0.88-1.18). The risk of major cardiovascular disease outcomes was reduced during the approximately 10-year interval of separation of the Hb A1c curves (HR, 0.83; 95% CI, 0.70-0.99), but it did not persist after equalization of Hb A1c levels (HR, 1.26; 95% CI, 0.90-1.75). Limitations include the observational study design, the study population of mostly older men, and reliance on administrative data for outcomes.
Bottom line: More than 5 years of intensive glucose lowering, compared with standard therapy, did not show significantly lower risks of cardiovascular events or mortality once the glycated hemoglobin curves equalized during follow-up in years 11-15.
Citation: Reaven PD et al. Intensive glucose control in patients with type 2 diabetes – 15-year follow-up. New Engl J Med. 2019 Jun 6;380(23):2215-24.
Dr. Burke is a hospitalist at Vanderbilt University Medical Center, Nashville, Tenn.
Background: A previous study reported that a median of 5.6 years of intensive versus standard glucose lowering in veterans with type 2 diabetes resulted in significantly reduced risk of major cardiovascular events after 10 years of combined intervention and observational follow-up.
Study design: Prospective cohort.
Setting: Veterans Affairs Healthcare System.
Synopsis: In the original trial, 1,791 veterans were randomly assigned to receive either intensive or standard glucose control therapy. After conclusion of that study, 1,655 participants were followed using central databases, and 1,391 also provided data via surveys and chart review. Initially the difference in the glycated hemoglobin (Hb A1c) curves between the two groups averaged 1.5%, but it declined to 0.2%-0.3% 3 years after the trial ended. The median Hb A1c then stabilized to 8% in both groups.
Over a period of 15 years of combined intervention and posttrial follow-up, the risks of major cardiovascular events or death were not lower in the intensive-therapy group (hazard ratio for composite outcome, 0.91; 95% confidence interval, 0.78-1.06; P = .23; HR for death, 1.02; 95% CI, 0.88-1.18). The risk of major cardiovascular disease outcomes was reduced during the approximately 10-year interval of separation of the Hb A1c curves (HR, 0.83; 95% CI, 0.70-0.99), but it did not persist after equalization of Hb A1c levels (HR, 1.26; 95% CI, 0.90-1.75). Limitations include the observational study design, the study population of mostly older men, and reliance on administrative data for outcomes.
Bottom line: More than 5 years of intensive glucose lowering, compared with standard therapy, did not show significantly lower risks of cardiovascular events or mortality once the glycated hemoglobin curves equalized during follow-up in years 11-15.
Citation: Reaven PD et al. Intensive glucose control in patients with type 2 diabetes – 15-year follow-up. New Engl J Med. 2019 Jun 6;380(23):2215-24.
Dr. Burke is a hospitalist at Vanderbilt University Medical Center, Nashville, Tenn.
Popularity of virtual conferences may mean a permanent shift
Fifteen days. That’s how much time the American College of Cardiology (ACC) had to convert its annual conference, scheduled for the end of March this year in Chicago, into a virtual meeting for the estimated 17,000 people who had planned to attend.
Because of the coronavirus pandemic, Illinois announced restrictions on the size of gatherings on March 13, causing the ACC to pivot to an online-only model.
“One big advantage was that we already had all of our content planned,” Janice Sibley, the ACC’s executive vice president of education, told Medscape Medical News. “We knew who the faculty would be for different sessions, and many of them had already planned their slides.”
But determining how to present those hundreds of presentations at an online conference, not to mention addressing the logistics related to registrations, tech platforms, exhibit hall sponsors, and other aspects of an annual meeting, would be no small task.
But according to a Medscape poll, many physicians think that, while the virtual experience is worthwhile and getting better, it’s never going to be the same as spending several days on site, immersed in the experience of an annual meeting.
As one respondent commented, “I miss the intellectual excitement, the electricity in the room, when there is a live presentation that announces a major breakthrough.”
Large medical societies have an advantage
As ACC rapidly prepared for its virtual conference, the society first refunded all registration and expo fees and worked with the vendor partners to resolve the cancellation of rental space, food and beverage services, and decorating. Then they organized a team of 15 people split into three groups. One group focused on the intellectual, scientific, and educational elements of the virtual conference. They chose 24 sessions to livestream and decided to prerecord the rest for on-demand access, limiting the number of presenters they needed to train for online presentation.
A second team focused on business and worked with industry partners on how to translate a large expo into digital offerings. They developed virtual pages, advertisements, promotions, and industry-sponsored education.
The third team’s focus, Ms. Sibley said, was most critical, and the hardest: addressing socio-emotional needs.
“That group was responsible for trying to create the buzz and excitement we would have had at the event,” she said, “pivoting that experience we would have had in a live event to a virtual environment. What we were worried about was, would anyone even come?”
But ACC built it, and they did indeed come. Within a half hour of the opening session, nearly 13,000 people logged on from around the world. “It worked beautifully,” Ms. Sibley said.
By the end of the 3-day event, approximately 34,000 unique visitors had logged in for live or prerecorded sessions. Although ACC worried at first about technical glitches and bandwidth needs, everything ran smoothly. By 90 days after the meeting, 63,000 unique users had logged in to access the conference content.
ACC was among the first organizations forced to switch from an in-person to all-online meeting, but dozens of other organizations have now done the same, discovering the benefits and drawbacks of a virtual environment while experimenting with different formats and offerings. Talks with a few large medical societies about the experience revealed several common themes, including the following:
- Finding new ways to attract and measure attendance.
- Ensuring the actual scientific content was as robust online as in person.
- Realizing the value of social media in enhancing the socio-emotional experience.
- Believing that virtual meetings will become a permanent fixture in a future of “hybrid” conferences.
New ways of attracting and measuring attendance
Previous ways to measure meeting attendance were straightforward: number of registrations and number of people physically walking into sessions. An online conference, however, offers dozens of ways to measure attendance. While the number of registrations remained one tool – and all the organizations interviewed reported record numbers of registrations – organizations also used other metrics to measure success, such as “participation,” “engagement,” and “viewing time.”
ACC defined “participation” as a unique user logging in, and it defined “engagement” as sticking around for a while, possibly using chat functions or discussing the content on social media. The American Society of Clinical Oncology (ASCO) annual conference in May, which attracted more than 44,000 registered attendees, also measured total content views – more than 2.5 million during the meeting – and monitored social media. More than 8,800 Twitter users posted more than 45,000 tweets with the #ASCO20 hashtag during the meeting, generating 750 million likes, shares, and comments. The European League Against Rheumatism (EULAR) annual congress registered a record 18,700 delegates – up from 14,500 in 2019 – but it also measured attendance by average viewing time and visits by congress day and by category.
Organizations shifted fee structures as well. While ACC refunded fees for its first online meeting, it has since developed tiers to match fees to anticipated value, such as charging more for livestreamed sessions that allow interactivity than for viewing recordings. ASCO offered a one-time fee waiver for members plus free registration to cancer survivors and caregivers, discounted registration for patient advocates, and reduced fees for other categories. But adjusting how to measure attendance and charge for events were the easy parts of transitioning to online.
Priority for having robust content
The biggest difficulty for most organizations was the short time they had to move online, with a host of challenges accompanying the switch, said the executive director of EULAR, Julia Rautenstrauch, DrMed. These included technical requirements, communication, training, finances, legal issues, compliance rules, and other logistics.
“The year 2020 will be remembered for being the year of unexpected transformation,” said a spokesperson from European Society for Medical Oncology (ESMO), who declined to be named. “The number of fundamental questions we had to ask ourselves is pages long. The solutions we have implemented so far have been successful, but we won’t rest on our laurels.”
ASCO had an advantage in the pivot, despite only 6 weeks to make the switch, because they already had a robust online platform to build on. “We weren’t starting from scratch, but we were sure changing the way we prepared,” ASCO CEO Clifford Hudis, MD, said.
All of the organizations made the breadth and quality of scientific and educational content a top priority, and those who have already hosted meetings this year report positive feedback.
“The rating of the scientific content was excellent, and the event did indeed fulfill the educational goals and expected learning outcomes for the vast majority of delegates,” EULAR’s Dr. Rautenstrauch said.
“Our goal, when we went into this, was that, in the future when somebody looks back at ASCO20, they should not be able to tell that it was a different year from any other in terms of the science,” Dr. Hudis said.
Missing out on networking and social interaction
Even when logistics run smoothly, virtual conferences must overcome two other challenges: the loss of in-person interactions and the potential for “Zoom burnout.”
“You do miss that human contact, the unsaid reactions in the room when you’re speaking or providing a controversial statement, even the facial expression or seeing people lean in or being distracted,” Ms. Sibley said.
Taher Modarressi, MD, an endocrinologist with Diabetes and Endocrine Associates of Hunterdon in Flemington, N.J., said all the digital conferences he has attended were missing those key social elements: “seeing old friends, sideline discussions that generate new ideas, and meeting new colleagues. However, this has been partly alleviated with the robust rise of social media and ‘MedTwitter,’ in particular, where these discussions and interactions continue.”
To attempt to meet that need for social interaction, societies came up with a variety of options. EULAR offered chatrooms, “Meet the Expert” sessions, and other virtual opportunities for live interaction. ASCO hosted discussion groups with subsets of participants, such as virtual meetings with oncology fellows, and it plans to offer networking sessions and “poster walks” during future meetings.
“The value of an in-person meeting is connecting with people, exchanging ideas over coffee, and making new contacts,” ASCO’s Dr. Hudis said. While virtual meetings lose many of those personal interactions, knowledge can also be shared with more people, he said.
The key to combating digital fatigue is focusing on opportunities for interactivity, ACC’s Ms. Sibley said. “When you are creating a virtual environment, it’s important that you offer choices.” Online learners tend to have shorter attention spans than in-person learners, so people need opportunities to flip between sessions, like flipping between TV channels. Different engagement options are also essential, such as chat functions on the video platforms, asking questions of presenters orally or in writing, and using the familiar hashtags for social media discussion.
“We set up all those different ways to interact, and you allow the user to choose,” Ms. Sibley said.
Some conferences, however, had less time or fewer resources to adjust to a virtual format and couldn’t make up for the lost social interaction. Andy Bowman, MD, a neonatologist in Lubbock, Tex., was supposed to attend the Neonatal & Pediatric Airborne Transport Conference sponsored by International Biomed in the spring, but it was canceled at the last minute. Several weeks later, the organizers released videos of scheduled speakers giving their talks, but it was less engaging and too easy to get distracted, Dr. Bowman said.
“There is a noticeable decrease in energy – you can’t look around to feed off other’s reactions when a speaker says something off the wall, or new, or contrary to expectations,” he said. He also especially missed the social interactions, such as “missing out on the chance encounters in the hallway or seeing the same face in back-to-back sessions and figuring out you have shared interest.” He was also sorry to miss the expo because neonatal transport requires a lot of specialty equipment, and he appreciates the chance to actually touch and see it in person.
Advantages of an online meeting
Despite the challenges, online meetings can overcome obstacles of in-person meetings, particularly for those in low- and middle-income countries, such as travel and registration costs, the hardships of being away from practice, and visa restrictions.
“You really have the potential to broaden your reach,” Ms. Sibley said, noting that people in 157 countries participated in ACC.20.
Another advantage is keeping the experience available to people after the livestreamed event.
“Virtual events have demonstrated the potential for a more democratic conference world, expanding the dissemination of information to a much wider community of stakeholders,” ESMO’s spokesperson said.
Not traveling can actually mean getting more out of the conference, said Atisha Patel Manhas, MD, a hematologist/oncologist in Dallas, who attended ASCO. “I have really enjoyed the access aspect – on the virtual platform there is so much more content available to you, and travel time doesn’t cut into conference time,” she said, though she also missed the interaction with colleagues.
Others found that virtual conferences provided more engagement than in-person conferences. Marwah Abdalla, MD, MPH, an assistant professor of medicine and director of education for the Cardiac Intensive Care Unit at Columbia University Medical Center, New York, felt that moderated Q&A sessions offered more interaction among participants. She attended and spoke on a panel during virtual SLEEP 2020, a joint meeting of the American Academy of Sleep Medicine (AASM) and the Sleep Research Society (SRS).
“Usually during in-person sessions, only a few questions are possible, and participants rarely have an opportunity to discuss the presentations within the session due to time limits,” Dr. Abdalla said. “Because the conference presentations can also be viewed asynchronously, participants have been able to comment on lectures and continue the discussion offline, either via social media or via email.” She acknowledged drawbacks of the virtual experience, such as an inability to socialize in person and participate in activities but appreciated the new opportunities to network and learn from international colleagues who would not have been able to attend in person.
Ritu Thamman, MD, assistant professor of medicine at the University of Pittsburgh School of Medicine, pointed out that many institutions have cut their travel budgets, and physicians would be unable to attend in-person conferences for financial or other reasons. She especially appreciated that the European Society of Cardiology had no registration fee for ESC 2020 and made their content free for all of September, which led to more than 100,000 participants.
“That meant anyone anywhere could learn,” she said. “It makes it much more diverse and more egalitarian. That feels like a good step in the right direction for all of us.”
Dr. Modarressi, who found ESC “exhilarating,” similarly noted the benefit of such an equitably accessible conference. “Decreasing barriers and improving access to top-line results and up-to-date information has always been a challenge to the global health community,” he said, noting that the map of attendance for the virtual meeting was “astonishing.”
Given these benefits, organizers said they expect a future of hybrid conferences: physical meetings for those able to attend in person and virtual ones for those who cannot.
“We also expect that the hybrid congress will cater to the needs of people on-site by allowing them additional access to more scientific content than by physical attendance alone,” Dr. Rautenstrauch said.
Everyone has been in reactive mode this year, Ms. Sibley said, but the future looks bright as they seek ways to overcome challenges such as socio-emotional needs and virtual expo spaces.
“We’ve been thrust into the virtual world much faster than we expected, but we’re finding it’s opening more opportunities than we had live,” Ms. Sibley said. “This has catapulted us, for better or worse, into a new way to deliver education and other types of information.
“I think, if we’re smart, we’ll continue to think of ways this can augment our live environment and not replace it.”
A version of this article originally appeared on Medscape.com.
Fifteen days. That’s how much time the American College of Cardiology (ACC) had to convert its annual conference, scheduled for the end of March this year in Chicago, into a virtual meeting for the estimated 17,000 people who had planned to attend.
Because of the coronavirus pandemic, Illinois announced restrictions on the size of gatherings on March 13, causing the ACC to pivot to an online-only model.
“One big advantage was that we already had all of our content planned,” Janice Sibley, the ACC’s executive vice president of education, told Medscape Medical News. “We knew who the faculty would be for different sessions, and many of them had already planned their slides.”
But determining how to present those hundreds of presentations at an online conference, not to mention addressing the logistics related to registrations, tech platforms, exhibit hall sponsors, and other aspects of an annual meeting, would be no small task.
But according to a Medscape poll, many physicians think that, while the virtual experience is worthwhile and getting better, it’s never going to be the same as spending several days on site, immersed in the experience of an annual meeting.
As one respondent commented, “I miss the intellectual excitement, the electricity in the room, when there is a live presentation that announces a major breakthrough.”
Large medical societies have an advantage
As ACC rapidly prepared for its virtual conference, the society first refunded all registration and expo fees and worked with the vendor partners to resolve the cancellation of rental space, food and beverage services, and decorating. Then they organized a team of 15 people split into three groups. One group focused on the intellectual, scientific, and educational elements of the virtual conference. They chose 24 sessions to livestream and decided to prerecord the rest for on-demand access, limiting the number of presenters they needed to train for online presentation.
A second team focused on business and worked with industry partners on how to translate a large expo into digital offerings. They developed virtual pages, advertisements, promotions, and industry-sponsored education.
The third team’s focus, Ms. Sibley said, was most critical, and the hardest: addressing socio-emotional needs.
“That group was responsible for trying to create the buzz and excitement we would have had at the event,” she said, “pivoting that experience we would have had in a live event to a virtual environment. What we were worried about was, would anyone even come?”
But ACC built it, and they did indeed come. Within a half hour of the opening session, nearly 13,000 people logged on from around the world. “It worked beautifully,” Ms. Sibley said.
By the end of the 3-day event, approximately 34,000 unique visitors had logged in for live or prerecorded sessions. Although ACC worried at first about technical glitches and bandwidth needs, everything ran smoothly. By 90 days after the meeting, 63,000 unique users had logged in to access the conference content.
ACC was among the first organizations forced to switch from an in-person to all-online meeting, but dozens of other organizations have now done the same, discovering the benefits and drawbacks of a virtual environment while experimenting with different formats and offerings. Talks with a few large medical societies about the experience revealed several common themes, including the following:
- Finding new ways to attract and measure attendance.
- Ensuring the actual scientific content was as robust online as in person.
- Realizing the value of social media in enhancing the socio-emotional experience.
- Believing that virtual meetings will become a permanent fixture in a future of “hybrid” conferences.
New ways of attracting and measuring attendance
Previous ways to measure meeting attendance were straightforward: number of registrations and number of people physically walking into sessions. An online conference, however, offers dozens of ways to measure attendance. While the number of registrations remained one tool – and all the organizations interviewed reported record numbers of registrations – organizations also used other metrics to measure success, such as “participation,” “engagement,” and “viewing time.”
ACC defined “participation” as a unique user logging in, and it defined “engagement” as sticking around for a while, possibly using chat functions or discussing the content on social media. The American Society of Clinical Oncology (ASCO) annual conference in May, which attracted more than 44,000 registered attendees, also measured total content views – more than 2.5 million during the meeting – and monitored social media. More than 8,800 Twitter users posted more than 45,000 tweets with the #ASCO20 hashtag during the meeting, generating 750 million likes, shares, and comments. The European League Against Rheumatism (EULAR) annual congress registered a record 18,700 delegates – up from 14,500 in 2019 – but it also measured attendance by average viewing time and visits by congress day and by category.
Organizations shifted fee structures as well. While ACC refunded fees for its first online meeting, it has since developed tiers to match fees to anticipated value, such as charging more for livestreamed sessions that allow interactivity than for viewing recordings. ASCO offered a one-time fee waiver for members plus free registration to cancer survivors and caregivers, discounted registration for patient advocates, and reduced fees for other categories. But adjusting how to measure attendance and charge for events were the easy parts of transitioning to online.
Priority for having robust content
The biggest difficulty for most organizations was the short time they had to move online, with a host of challenges accompanying the switch, said the executive director of EULAR, Julia Rautenstrauch, DrMed. These included technical requirements, communication, training, finances, legal issues, compliance rules, and other logistics.
“The year 2020 will be remembered for being the year of unexpected transformation,” said a spokesperson from European Society for Medical Oncology (ESMO), who declined to be named. “The number of fundamental questions we had to ask ourselves is pages long. The solutions we have implemented so far have been successful, but we won’t rest on our laurels.”
ASCO had an advantage in the pivot, despite only 6 weeks to make the switch, because they already had a robust online platform to build on. “We weren’t starting from scratch, but we were sure changing the way we prepared,” ASCO CEO Clifford Hudis, MD, said.
All of the organizations made the breadth and quality of scientific and educational content a top priority, and those who have already hosted meetings this year report positive feedback.
“The rating of the scientific content was excellent, and the event did indeed fulfill the educational goals and expected learning outcomes for the vast majority of delegates,” EULAR’s Dr. Rautenstrauch said.
“Our goal, when we went into this, was that, in the future when somebody looks back at ASCO20, they should not be able to tell that it was a different year from any other in terms of the science,” Dr. Hudis said.
Missing out on networking and social interaction
Even when logistics run smoothly, virtual conferences must overcome two other challenges: the loss of in-person interactions and the potential for “Zoom burnout.”
“You do miss that human contact, the unsaid reactions in the room when you’re speaking or providing a controversial statement, even the facial expression or seeing people lean in or being distracted,” Ms. Sibley said.
Taher Modarressi, MD, an endocrinologist with Diabetes and Endocrine Associates of Hunterdon in Flemington, N.J., said all the digital conferences he has attended were missing those key social elements: “seeing old friends, sideline discussions that generate new ideas, and meeting new colleagues. However, this has been partly alleviated with the robust rise of social media and ‘MedTwitter,’ in particular, where these discussions and interactions continue.”
To attempt to meet that need for social interaction, societies came up with a variety of options. EULAR offered chatrooms, “Meet the Expert” sessions, and other virtual opportunities for live interaction. ASCO hosted discussion groups with subsets of participants, such as virtual meetings with oncology fellows, and it plans to offer networking sessions and “poster walks” during future meetings.
“The value of an in-person meeting is connecting with people, exchanging ideas over coffee, and making new contacts,” ASCO’s Dr. Hudis said. While virtual meetings lose many of those personal interactions, knowledge can also be shared with more people, he said.
The key to combating digital fatigue is focusing on opportunities for interactivity, ACC’s Ms. Sibley said. “When you are creating a virtual environment, it’s important that you offer choices.” Online learners tend to have shorter attention spans than in-person learners, so people need opportunities to flip between sessions, like flipping between TV channels. Different engagement options are also essential, such as chat functions on the video platforms, asking questions of presenters orally or in writing, and using the familiar hashtags for social media discussion.
“We set up all those different ways to interact, and you allow the user to choose,” Ms. Sibley said.
Some conferences, however, had less time or fewer resources to adjust to a virtual format and couldn’t make up for the lost social interaction. Andy Bowman, MD, a neonatologist in Lubbock, Tex., was supposed to attend the Neonatal & Pediatric Airborne Transport Conference sponsored by International Biomed in the spring, but it was canceled at the last minute. Several weeks later, the organizers released videos of scheduled speakers giving their talks, but it was less engaging and too easy to get distracted, Dr. Bowman said.
“There is a noticeable decrease in energy – you can’t look around to feed off other’s reactions when a speaker says something off the wall, or new, or contrary to expectations,” he said. He also especially missed the social interactions, such as “missing out on the chance encounters in the hallway or seeing the same face in back-to-back sessions and figuring out you have shared interest.” He was also sorry to miss the expo because neonatal transport requires a lot of specialty equipment, and he appreciates the chance to actually touch and see it in person.
Advantages of an online meeting
Despite the challenges, online meetings can overcome obstacles of in-person meetings, particularly for those in low- and middle-income countries, such as travel and registration costs, the hardships of being away from practice, and visa restrictions.
“You really have the potential to broaden your reach,” Ms. Sibley said, noting that people in 157 countries participated in ACC.20.
Another advantage is keeping the experience available to people after the livestreamed event.
“Virtual events have demonstrated the potential for a more democratic conference world, expanding the dissemination of information to a much wider community of stakeholders,” ESMO’s spokesperson said.
Not traveling can actually mean getting more out of the conference, said Atisha Patel Manhas, MD, a hematologist/oncologist in Dallas, who attended ASCO. “I have really enjoyed the access aspect – on the virtual platform there is so much more content available to you, and travel time doesn’t cut into conference time,” she said, though she also missed the interaction with colleagues.
Others found that virtual conferences provided more engagement than in-person conferences. Marwah Abdalla, MD, MPH, an assistant professor of medicine and director of education for the Cardiac Intensive Care Unit at Columbia University Medical Center, New York, felt that moderated Q&A sessions offered more interaction among participants. She attended and spoke on a panel during virtual SLEEP 2020, a joint meeting of the American Academy of Sleep Medicine (AASM) and the Sleep Research Society (SRS).
“Usually during in-person sessions, only a few questions are possible, and participants rarely have an opportunity to discuss the presentations within the session due to time limits,” Dr. Abdalla said. “Because the conference presentations can also be viewed asynchronously, participants have been able to comment on lectures and continue the discussion offline, either via social media or via email.” She acknowledged drawbacks of the virtual experience, such as an inability to socialize in person and participate in activities but appreciated the new opportunities to network and learn from international colleagues who would not have been able to attend in person.
Ritu Thamman, MD, assistant professor of medicine at the University of Pittsburgh School of Medicine, pointed out that many institutions have cut their travel budgets, and physicians would be unable to attend in-person conferences for financial or other reasons. She especially appreciated that the European Society of Cardiology had no registration fee for ESC 2020 and made their content free for all of September, which led to more than 100,000 participants.
“That meant anyone anywhere could learn,” she said. “It makes it much more diverse and more egalitarian. That feels like a good step in the right direction for all of us.”
Dr. Modarressi, who found ESC “exhilarating,” similarly noted the benefit of such an equitably accessible conference. “Decreasing barriers and improving access to top-line results and up-to-date information has always been a challenge to the global health community,” he said, noting that the map of attendance for the virtual meeting was “astonishing.”
Given these benefits, organizers said they expect a future of hybrid conferences: physical meetings for those able to attend in person and virtual ones for those who cannot.
“We also expect that the hybrid congress will cater to the needs of people on-site by allowing them additional access to more scientific content than by physical attendance alone,” Dr. Rautenstrauch said.
Everyone has been in reactive mode this year, Ms. Sibley said, but the future looks bright as they seek ways to overcome challenges such as socio-emotional needs and virtual expo spaces.
“We’ve been thrust into the virtual world much faster than we expected, but we’re finding it’s opening more opportunities than we had live,” Ms. Sibley said. “This has catapulted us, for better or worse, into a new way to deliver education and other types of information.
“I think, if we’re smart, we’ll continue to think of ways this can augment our live environment and not replace it.”
A version of this article originally appeared on Medscape.com.
Fifteen days. That’s how much time the American College of Cardiology (ACC) had to convert its annual conference, scheduled for the end of March this year in Chicago, into a virtual meeting for the estimated 17,000 people who had planned to attend.
Because of the coronavirus pandemic, Illinois announced restrictions on the size of gatherings on March 13, causing the ACC to pivot to an online-only model.
“One big advantage was that we already had all of our content planned,” Janice Sibley, the ACC’s executive vice president of education, told Medscape Medical News. “We knew who the faculty would be for different sessions, and many of them had already planned their slides.”
But determining how to present those hundreds of presentations at an online conference, not to mention addressing the logistics related to registrations, tech platforms, exhibit hall sponsors, and other aspects of an annual meeting, would be no small task.
But according to a Medscape poll, many physicians think that, while the virtual experience is worthwhile and getting better, it’s never going to be the same as spending several days on site, immersed in the experience of an annual meeting.
As one respondent commented, “I miss the intellectual excitement, the electricity in the room, when there is a live presentation that announces a major breakthrough.”
Large medical societies have an advantage
As ACC rapidly prepared for its virtual conference, the society first refunded all registration and expo fees and worked with the vendor partners to resolve the cancellation of rental space, food and beverage services, and decorating. Then they organized a team of 15 people split into three groups. One group focused on the intellectual, scientific, and educational elements of the virtual conference. They chose 24 sessions to livestream and decided to prerecord the rest for on-demand access, limiting the number of presenters they needed to train for online presentation.
A second team focused on business and worked with industry partners on how to translate a large expo into digital offerings. They developed virtual pages, advertisements, promotions, and industry-sponsored education.
The third team’s focus, Ms. Sibley said, was most critical, and the hardest: addressing socio-emotional needs.
“That group was responsible for trying to create the buzz and excitement we would have had at the event,” she said, “pivoting that experience we would have had in a live event to a virtual environment. What we were worried about was, would anyone even come?”
But ACC built it, and they did indeed come. Within a half hour of the opening session, nearly 13,000 people logged on from around the world. “It worked beautifully,” Ms. Sibley said.
By the end of the 3-day event, approximately 34,000 unique visitors had logged in for live or prerecorded sessions. Although ACC worried at first about technical glitches and bandwidth needs, everything ran smoothly. By 90 days after the meeting, 63,000 unique users had logged in to access the conference content.
ACC was among the first organizations forced to switch from an in-person to all-online meeting, but dozens of other organizations have now done the same, discovering the benefits and drawbacks of a virtual environment while experimenting with different formats and offerings. Talks with a few large medical societies about the experience revealed several common themes, including the following:
- Finding new ways to attract and measure attendance.
- Ensuring the actual scientific content was as robust online as in person.
- Realizing the value of social media in enhancing the socio-emotional experience.
- Believing that virtual meetings will become a permanent fixture in a future of “hybrid” conferences.
New ways of attracting and measuring attendance
Previous ways to measure meeting attendance were straightforward: number of registrations and number of people physically walking into sessions. An online conference, however, offers dozens of ways to measure attendance. While the number of registrations remained one tool – and all the organizations interviewed reported record numbers of registrations – organizations also used other metrics to measure success, such as “participation,” “engagement,” and “viewing time.”
ACC defined “participation” as a unique user logging in, and it defined “engagement” as sticking around for a while, possibly using chat functions or discussing the content on social media. The American Society of Clinical Oncology (ASCO) annual conference in May, which attracted more than 44,000 registered attendees, also measured total content views – more than 2.5 million during the meeting – and monitored social media. More than 8,800 Twitter users posted more than 45,000 tweets with the #ASCO20 hashtag during the meeting, generating 750 million likes, shares, and comments. The European League Against Rheumatism (EULAR) annual congress registered a record 18,700 delegates – up from 14,500 in 2019 – but it also measured attendance by average viewing time and visits by congress day and by category.
Organizations shifted fee structures as well. While ACC refunded fees for its first online meeting, it has since developed tiers to match fees to anticipated value, such as charging more for livestreamed sessions that allow interactivity than for viewing recordings. ASCO offered a one-time fee waiver for members plus free registration to cancer survivors and caregivers, discounted registration for patient advocates, and reduced fees for other categories. But adjusting how to measure attendance and charge for events were the easy parts of transitioning to online.
Priority for having robust content
The biggest difficulty for most organizations was the short time they had to move online, with a host of challenges accompanying the switch, said the executive director of EULAR, Julia Rautenstrauch, DrMed. These included technical requirements, communication, training, finances, legal issues, compliance rules, and other logistics.
“The year 2020 will be remembered for being the year of unexpected transformation,” said a spokesperson from European Society for Medical Oncology (ESMO), who declined to be named. “The number of fundamental questions we had to ask ourselves is pages long. The solutions we have implemented so far have been successful, but we won’t rest on our laurels.”
ASCO had an advantage in the pivot, despite only 6 weeks to make the switch, because they already had a robust online platform to build on. “We weren’t starting from scratch, but we were sure changing the way we prepared,” ASCO CEO Clifford Hudis, MD, said.
All of the organizations made the breadth and quality of scientific and educational content a top priority, and those who have already hosted meetings this year report positive feedback.
“The rating of the scientific content was excellent, and the event did indeed fulfill the educational goals and expected learning outcomes for the vast majority of delegates,” EULAR’s Dr. Rautenstrauch said.
“Our goal, when we went into this, was that, in the future when somebody looks back at ASCO20, they should not be able to tell that it was a different year from any other in terms of the science,” Dr. Hudis said.
Missing out on networking and social interaction
Even when logistics run smoothly, virtual conferences must overcome two other challenges: the loss of in-person interactions and the potential for “Zoom burnout.”
“You do miss that human contact, the unsaid reactions in the room when you’re speaking or providing a controversial statement, even the facial expression or seeing people lean in or being distracted,” Ms. Sibley said.
Taher Modarressi, MD, an endocrinologist with Diabetes and Endocrine Associates of Hunterdon in Flemington, N.J., said all the digital conferences he has attended were missing those key social elements: “seeing old friends, sideline discussions that generate new ideas, and meeting new colleagues. However, this has been partly alleviated with the robust rise of social media and ‘MedTwitter,’ in particular, where these discussions and interactions continue.”
To attempt to meet that need for social interaction, societies came up with a variety of options. EULAR offered chatrooms, “Meet the Expert” sessions, and other virtual opportunities for live interaction. ASCO hosted discussion groups with subsets of participants, such as virtual meetings with oncology fellows, and it plans to offer networking sessions and “poster walks” during future meetings.
“The value of an in-person meeting is connecting with people, exchanging ideas over coffee, and making new contacts,” ASCO’s Dr. Hudis said. While virtual meetings lose many of those personal interactions, knowledge can also be shared with more people, he said.
The key to combating digital fatigue is focusing on opportunities for interactivity, ACC’s Ms. Sibley said. “When you are creating a virtual environment, it’s important that you offer choices.” Online learners tend to have shorter attention spans than in-person learners, so people need opportunities to flip between sessions, like flipping between TV channels. Different engagement options are also essential, such as chat functions on the video platforms, asking questions of presenters orally or in writing, and using the familiar hashtags for social media discussion.
“We set up all those different ways to interact, and you allow the user to choose,” Ms. Sibley said.
Some conferences, however, had less time or fewer resources to adjust to a virtual format and couldn’t make up for the lost social interaction. Andy Bowman, MD, a neonatologist in Lubbock, Tex., was supposed to attend the Neonatal & Pediatric Airborne Transport Conference sponsored by International Biomed in the spring, but it was canceled at the last minute. Several weeks later, the organizers released videos of scheduled speakers giving their talks, but it was less engaging and too easy to get distracted, Dr. Bowman said.
“There is a noticeable decrease in energy – you can’t look around to feed off other’s reactions when a speaker says something off the wall, or new, or contrary to expectations,” he said. He also especially missed the social interactions, such as “missing out on the chance encounters in the hallway or seeing the same face in back-to-back sessions and figuring out you have shared interest.” He was also sorry to miss the expo because neonatal transport requires a lot of specialty equipment, and he appreciates the chance to actually touch and see it in person.
Advantages of an online meeting
Despite the challenges, online meetings can overcome obstacles of in-person meetings, particularly for those in low- and middle-income countries, such as travel and registration costs, the hardships of being away from practice, and visa restrictions.
“You really have the potential to broaden your reach,” Ms. Sibley said, noting that people in 157 countries participated in ACC.20.
Another advantage is keeping the experience available to people after the livestreamed event.
“Virtual events have demonstrated the potential for a more democratic conference world, expanding the dissemination of information to a much wider community of stakeholders,” ESMO’s spokesperson said.
Not traveling can actually mean getting more out of the conference, said Atisha Patel Manhas, MD, a hematologist/oncologist in Dallas, who attended ASCO. “I have really enjoyed the access aspect – on the virtual platform there is so much more content available to you, and travel time doesn’t cut into conference time,” she said, though she also missed the interaction with colleagues.
Others found that virtual conferences provided more engagement than in-person conferences. Marwah Abdalla, MD, MPH, an assistant professor of medicine and director of education for the Cardiac Intensive Care Unit at Columbia University Medical Center, New York, felt that moderated Q&A sessions offered more interaction among participants. She attended and spoke on a panel during virtual SLEEP 2020, a joint meeting of the American Academy of Sleep Medicine (AASM) and the Sleep Research Society (SRS).
“Usually during in-person sessions, only a few questions are possible, and participants rarely have an opportunity to discuss the presentations within the session due to time limits,” Dr. Abdalla said. “Because the conference presentations can also be viewed asynchronously, participants have been able to comment on lectures and continue the discussion offline, either via social media or via email.” She acknowledged drawbacks of the virtual experience, such as an inability to socialize in person and participate in activities but appreciated the new opportunities to network and learn from international colleagues who would not have been able to attend in person.
Ritu Thamman, MD, assistant professor of medicine at the University of Pittsburgh School of Medicine, pointed out that many institutions have cut their travel budgets, and physicians would be unable to attend in-person conferences for financial or other reasons. She especially appreciated that the European Society of Cardiology had no registration fee for ESC 2020 and made their content free for all of September, which led to more than 100,000 participants.
“That meant anyone anywhere could learn,” she said. “It makes it much more diverse and more egalitarian. That feels like a good step in the right direction for all of us.”
Dr. Modarressi, who found ESC “exhilarating,” similarly noted the benefit of such an equitably accessible conference. “Decreasing barriers and improving access to top-line results and up-to-date information has always been a challenge to the global health community,” he said, noting that the map of attendance for the virtual meeting was “astonishing.”
Given these benefits, organizers said they expect a future of hybrid conferences: physical meetings for those able to attend in person and virtual ones for those who cannot.
“We also expect that the hybrid congress will cater to the needs of people on-site by allowing them additional access to more scientific content than by physical attendance alone,” Dr. Rautenstrauch said.
Everyone has been in reactive mode this year, Ms. Sibley said, but the future looks bright as they seek ways to overcome challenges such as socio-emotional needs and virtual expo spaces.
“We’ve been thrust into the virtual world much faster than we expected, but we’re finding it’s opening more opportunities than we had live,” Ms. Sibley said. “This has catapulted us, for better or worse, into a new way to deliver education and other types of information.
“I think, if we’re smart, we’ll continue to think of ways this can augment our live environment and not replace it.”
A version of this article originally appeared on Medscape.com.
Cardiogenic shock rate soars in COVID-positive ACS
COVID-19–positive patients undergoing an invasive strategy for acute coronary syndrome presented hours later than uninfected historical controls, had a far higher incidence of cardiogenic shock, and their in-hospital mortality rate was four- to fivefold greater, according to data from the Global Multicenter Prospective COVID–ACS Registry. These phenomena are probably interrelated, according to Anthony Gershlick, MBBS, who presented the registry results at the Transcatheter Cardiovascular Therapeutics virtual annual meeting.
“We know that increasing ischemic time leads to bigger infarcts. And we know that bigger infarcts lead to cardiogenic shock, with its known higher mortality,” said Dr. Gershlick, professor of interventional cardiology at the University of Leicester (England).
“These data suggest that patients may have presented late, likely due to COVID concerns, and they had worse outcomes. If these data are borne out, future public information strategies need to be reassuring, proactive, simple, and more effective because we think patients stayed away,” the cardiologist added. “There are important public information messages to be taken from these data about getting patients to come to hospital during such pandemics.”
He presented prospectively collected registry data on 144 patients with confirmed ST-elevation MI (STEMI) and 122 with non-ST–elevation MI (NSTEMI), all COVID-19 positive on presentation at 85 hospitals in the United Kingdom, Europe, and North America during March through August of 2020. Since the initial message to the public early in the pandemic in many places was to try to avoid the hospital, the investigators selected for their no-COVID comparison group the data on more than 22,000 STEMI and NSTEMI patients included in two British national databases covering 2018-2019.
The COVID-positive STEMI patients were significantly younger, had more comorbidities, and had a higher mean heart rate and lower systolic blood pressure at admission than the non-COVID STEMI control group. Their median time from symptom onset to admission was 339 minutes, compared with 178 minutes in controls. Their door-to-balloon time averaged 83 minutes, versus 37 minutes in the era before the pandemic.
“I suspect that’s got something to do with the donning and doffing of personal protective equipment,” he said at the meeting sponsored by the Cardiovascular Research Foundation.
The in-hospital mortality rates were strikingly different: 27.1% in COVID-positive STEMI patients versus 5.7% in controls. Bleeding Academic Research Consortium type 3-5 bleeding was increased as well, by a margin of 2.8% to 0.3%. So was stroke, with a 2.1% in-hospital incidence in COVID-positive STEMI patients and a 0.1% rate in the comparator arm.
“But the biggest headline here for me was that the cardiogenic shock rate was 20.1% in the COVID-positive patients versus 8.7% in the non-COVID STEMI patients,” the cardiologist continued.
The same pattern held true among the COVID-positive NSTEMI patients: They were younger, sicker, and slower to present to the hospital than the non-COVID group. The in-hospital mortality rate was 6.6% in the COVID-positive NSTEMI patients, compared with 1.2% in the reference group. The COVID-positive patients had a 2.5% bleeding rate versus 0.1% in the controls. And the incidence of cardiogenic shock was 5%, compared with 1.4% in the controls from before the pandemic.
“Even though NSTEMI is traditionally regarded as lower risk, this is really quite dramatic. These are sick patients,” Dr. Gershlick observed.
Nearly two-thirds of in-hospital deaths in COVID-positive ACS patients were cardiovascular, and three-quarters of those cardiovascular deaths occurred in patients with cardiogenic shock. Thirty-two percent of deaths in COVID-positive ACS patients were of respiratory causes, and 4.9% were neurologic.
Notably, the ischemic time of patients with cardiogenic shock who died – that is, the time from symptom onset to balloon deployment – averaged 1,271 minutes, compared with 441 minutes in those who died without being in cardiogenic shock.
Session comoderator Sahil A. Parikh, MD, director of endovascular services at Columbia University Medical Center in New York, commented, “One of the striking things that is resonating with me is the high incidence of cardiogenic shock and the mortality. It’s akin to what we’ve seen in New York.”
Discussant Valentin Fuster, MD, PhD, said he doubts that the increased in-hospital mortality in the COVID–ACS registry is related to the prolonged time to presentation at the hospital. More likely, it’s related to the greater thrombotic burden various studies have shown accompanies COVID-positive ACS. It might even be caused by a direct effect of the virus on the myocardium, added Dr. Fuster, director of the Zena and Michael A. Wiener Cardiovascular Institute and professor of medicine at the Icahn School of Medicine at Mount Sinai in New York.
“I have to say I absolutely disagree,” responded Dr. Gershlick. “I think it’s important that we try to understand all the mechanisms, but we know that patients with COVID are anxious, and I think one of the messages from this registry is patients took longer to come to hospital, they were sicker, they had more cardiogenic shock, and they died. And I don’t think it’s anything more complicated than that.”
Another discussant, Mamas Mamas, MD, is involved with a 500-patient U.K. pandemic ACS registry nearing publication. The findings, he said, are similar to what Dr. Gershlick reported in terms of the high rate of presentation with cardiogenic shock and elevated in-hospital mortality. The COVID-positive ACS patients were also more likely to present with out-of-hospital cardiac arrest. But like Dr. Fuster, he is skeptical that their worse outcomes can be explained by a delay in seeking care.
“I don’t think the delay in presentation is really associated with the high mortality rate that we see. The delay in our U.K. registry is maybe half an hour for STEMIs and maybe 2-3 hours for NSTEMIs. And I don’t think that can produce a 30%-40% increase in mortality,” asserted Dr. Mamas, professor of cardiology at Keele University in Staffordshire, England.
Dr. Gershlick reported having no financial conflicts regarding his presentation.
COVID-19–positive patients undergoing an invasive strategy for acute coronary syndrome presented hours later than uninfected historical controls, had a far higher incidence of cardiogenic shock, and their in-hospital mortality rate was four- to fivefold greater, according to data from the Global Multicenter Prospective COVID–ACS Registry. These phenomena are probably interrelated, according to Anthony Gershlick, MBBS, who presented the registry results at the Transcatheter Cardiovascular Therapeutics virtual annual meeting.
“We know that increasing ischemic time leads to bigger infarcts. And we know that bigger infarcts lead to cardiogenic shock, with its known higher mortality,” said Dr. Gershlick, professor of interventional cardiology at the University of Leicester (England).
“These data suggest that patients may have presented late, likely due to COVID concerns, and they had worse outcomes. If these data are borne out, future public information strategies need to be reassuring, proactive, simple, and more effective because we think patients stayed away,” the cardiologist added. “There are important public information messages to be taken from these data about getting patients to come to hospital during such pandemics.”
He presented prospectively collected registry data on 144 patients with confirmed ST-elevation MI (STEMI) and 122 with non-ST–elevation MI (NSTEMI), all COVID-19 positive on presentation at 85 hospitals in the United Kingdom, Europe, and North America during March through August of 2020. Since the initial message to the public early in the pandemic in many places was to try to avoid the hospital, the investigators selected for their no-COVID comparison group the data on more than 22,000 STEMI and NSTEMI patients included in two British national databases covering 2018-2019.
The COVID-positive STEMI patients were significantly younger, had more comorbidities, and had a higher mean heart rate and lower systolic blood pressure at admission than the non-COVID STEMI control group. Their median time from symptom onset to admission was 339 minutes, compared with 178 minutes in controls. Their door-to-balloon time averaged 83 minutes, versus 37 minutes in the era before the pandemic.
“I suspect that’s got something to do with the donning and doffing of personal protective equipment,” he said at the meeting sponsored by the Cardiovascular Research Foundation.
The in-hospital mortality rates were strikingly different: 27.1% in COVID-positive STEMI patients versus 5.7% in controls. Bleeding Academic Research Consortium type 3-5 bleeding was increased as well, by a margin of 2.8% to 0.3%. So was stroke, with a 2.1% in-hospital incidence in COVID-positive STEMI patients and a 0.1% rate in the comparator arm.
“But the biggest headline here for me was that the cardiogenic shock rate was 20.1% in the COVID-positive patients versus 8.7% in the non-COVID STEMI patients,” the cardiologist continued.
The same pattern held true among the COVID-positive NSTEMI patients: They were younger, sicker, and slower to present to the hospital than the non-COVID group. The in-hospital mortality rate was 6.6% in the COVID-positive NSTEMI patients, compared with 1.2% in the reference group. The COVID-positive patients had a 2.5% bleeding rate versus 0.1% in the controls. And the incidence of cardiogenic shock was 5%, compared with 1.4% in the controls from before the pandemic.
“Even though NSTEMI is traditionally regarded as lower risk, this is really quite dramatic. These are sick patients,” Dr. Gershlick observed.
Nearly two-thirds of in-hospital deaths in COVID-positive ACS patients were cardiovascular, and three-quarters of those cardiovascular deaths occurred in patients with cardiogenic shock. Thirty-two percent of deaths in COVID-positive ACS patients were of respiratory causes, and 4.9% were neurologic.
Notably, the ischemic time of patients with cardiogenic shock who died – that is, the time from symptom onset to balloon deployment – averaged 1,271 minutes, compared with 441 minutes in those who died without being in cardiogenic shock.
Session comoderator Sahil A. Parikh, MD, director of endovascular services at Columbia University Medical Center in New York, commented, “One of the striking things that is resonating with me is the high incidence of cardiogenic shock and the mortality. It’s akin to what we’ve seen in New York.”
Discussant Valentin Fuster, MD, PhD, said he doubts that the increased in-hospital mortality in the COVID–ACS registry is related to the prolonged time to presentation at the hospital. More likely, it’s related to the greater thrombotic burden various studies have shown accompanies COVID-positive ACS. It might even be caused by a direct effect of the virus on the myocardium, added Dr. Fuster, director of the Zena and Michael A. Wiener Cardiovascular Institute and professor of medicine at the Icahn School of Medicine at Mount Sinai in New York.
“I have to say I absolutely disagree,” responded Dr. Gershlick. “I think it’s important that we try to understand all the mechanisms, but we know that patients with COVID are anxious, and I think one of the messages from this registry is patients took longer to come to hospital, they were sicker, they had more cardiogenic shock, and they died. And I don’t think it’s anything more complicated than that.”
Another discussant, Mamas Mamas, MD, is involved with a 500-patient U.K. pandemic ACS registry nearing publication. The findings, he said, are similar to what Dr. Gershlick reported in terms of the high rate of presentation with cardiogenic shock and elevated in-hospital mortality. The COVID-positive ACS patients were also more likely to present with out-of-hospital cardiac arrest. But like Dr. Fuster, he is skeptical that their worse outcomes can be explained by a delay in seeking care.
“I don’t think the delay in presentation is really associated with the high mortality rate that we see. The delay in our U.K. registry is maybe half an hour for STEMIs and maybe 2-3 hours for NSTEMIs. And I don’t think that can produce a 30%-40% increase in mortality,” asserted Dr. Mamas, professor of cardiology at Keele University in Staffordshire, England.
Dr. Gershlick reported having no financial conflicts regarding his presentation.
COVID-19–positive patients undergoing an invasive strategy for acute coronary syndrome presented hours later than uninfected historical controls, had a far higher incidence of cardiogenic shock, and their in-hospital mortality rate was four- to fivefold greater, according to data from the Global Multicenter Prospective COVID–ACS Registry. These phenomena are probably interrelated, according to Anthony Gershlick, MBBS, who presented the registry results at the Transcatheter Cardiovascular Therapeutics virtual annual meeting.
“We know that increasing ischemic time leads to bigger infarcts. And we know that bigger infarcts lead to cardiogenic shock, with its known higher mortality,” said Dr. Gershlick, professor of interventional cardiology at the University of Leicester (England).
“These data suggest that patients may have presented late, likely due to COVID concerns, and they had worse outcomes. If these data are borne out, future public information strategies need to be reassuring, proactive, simple, and more effective because we think patients stayed away,” the cardiologist added. “There are important public information messages to be taken from these data about getting patients to come to hospital during such pandemics.”
He presented prospectively collected registry data on 144 patients with confirmed ST-elevation MI (STEMI) and 122 with non-ST–elevation MI (NSTEMI), all COVID-19 positive on presentation at 85 hospitals in the United Kingdom, Europe, and North America during March through August of 2020. Since the initial message to the public early in the pandemic in many places was to try to avoid the hospital, the investigators selected for their no-COVID comparison group the data on more than 22,000 STEMI and NSTEMI patients included in two British national databases covering 2018-2019.
The COVID-positive STEMI patients were significantly younger, had more comorbidities, and had a higher mean heart rate and lower systolic blood pressure at admission than the non-COVID STEMI control group. Their median time from symptom onset to admission was 339 minutes, compared with 178 minutes in controls. Their door-to-balloon time averaged 83 minutes, versus 37 minutes in the era before the pandemic.
“I suspect that’s got something to do with the donning and doffing of personal protective equipment,” he said at the meeting sponsored by the Cardiovascular Research Foundation.
The in-hospital mortality rates were strikingly different: 27.1% in COVID-positive STEMI patients versus 5.7% in controls. Bleeding Academic Research Consortium type 3-5 bleeding was increased as well, by a margin of 2.8% to 0.3%. So was stroke, with a 2.1% in-hospital incidence in COVID-positive STEMI patients and a 0.1% rate in the comparator arm.
“But the biggest headline here for me was that the cardiogenic shock rate was 20.1% in the COVID-positive patients versus 8.7% in the non-COVID STEMI patients,” the cardiologist continued.
The same pattern held true among the COVID-positive NSTEMI patients: They were younger, sicker, and slower to present to the hospital than the non-COVID group. The in-hospital mortality rate was 6.6% in the COVID-positive NSTEMI patients, compared with 1.2% in the reference group. The COVID-positive patients had a 2.5% bleeding rate versus 0.1% in the controls. And the incidence of cardiogenic shock was 5%, compared with 1.4% in the controls from before the pandemic.
“Even though NSTEMI is traditionally regarded as lower risk, this is really quite dramatic. These are sick patients,” Dr. Gershlick observed.
Nearly two-thirds of in-hospital deaths in COVID-positive ACS patients were cardiovascular, and three-quarters of those cardiovascular deaths occurred in patients with cardiogenic shock. Thirty-two percent of deaths in COVID-positive ACS patients were of respiratory causes, and 4.9% were neurologic.
Notably, the ischemic time of patients with cardiogenic shock who died – that is, the time from symptom onset to balloon deployment – averaged 1,271 minutes, compared with 441 minutes in those who died without being in cardiogenic shock.
Session comoderator Sahil A. Parikh, MD, director of endovascular services at Columbia University Medical Center in New York, commented, “One of the striking things that is resonating with me is the high incidence of cardiogenic shock and the mortality. It’s akin to what we’ve seen in New York.”
Discussant Valentin Fuster, MD, PhD, said he doubts that the increased in-hospital mortality in the COVID–ACS registry is related to the prolonged time to presentation at the hospital. More likely, it’s related to the greater thrombotic burden various studies have shown accompanies COVID-positive ACS. It might even be caused by a direct effect of the virus on the myocardium, added Dr. Fuster, director of the Zena and Michael A. Wiener Cardiovascular Institute and professor of medicine at the Icahn School of Medicine at Mount Sinai in New York.
“I have to say I absolutely disagree,” responded Dr. Gershlick. “I think it’s important that we try to understand all the mechanisms, but we know that patients with COVID are anxious, and I think one of the messages from this registry is patients took longer to come to hospital, they were sicker, they had more cardiogenic shock, and they died. And I don’t think it’s anything more complicated than that.”
Another discussant, Mamas Mamas, MD, is involved with a 500-patient U.K. pandemic ACS registry nearing publication. The findings, he said, are similar to what Dr. Gershlick reported in terms of the high rate of presentation with cardiogenic shock and elevated in-hospital mortality. The COVID-positive ACS patients were also more likely to present with out-of-hospital cardiac arrest. But like Dr. Fuster, he is skeptical that their worse outcomes can be explained by a delay in seeking care.
“I don’t think the delay in presentation is really associated with the high mortality rate that we see. The delay in our U.K. registry is maybe half an hour for STEMIs and maybe 2-3 hours for NSTEMIs. And I don’t think that can produce a 30%-40% increase in mortality,” asserted Dr. Mamas, professor of cardiology at Keele University in Staffordshire, England.
Dr. Gershlick reported having no financial conflicts regarding his presentation.
FROM TCT 2020
Experts tout immediate quadruple therapy for HFrEF patients
Gregg C. Fonarow, MD, recommended.
Less than 2 months before Dr. Fonarow made that striking statement during the virtual annual meeting of the Heart Failure Society of America, investigators first reported results from the EMPEROR-Reduced trial at the European Society of Cardiology’s virtual annual meeting, showing that the sodium-glucose transporter 2 (SGLT2) inhibitor empagliflozin (Jardiance) successfully cut events in patients with heart failure with reduced ejection fraction (HFrEF). That report, a year after results from a similar trial (DAPA-HF) showed the same outcome using a different drug from the same class, dapagliflozin (Farxiga), cemented the SGLT2 inhibitor drug class as the fourth pillar for treating HFrEF, joining the angiotensin receptor neprilysin inhibitor (ARNI) class (sacubitril valsartan), beta-blockers (like carvedilol), and mineralocorticoid receptor antagonists (like spironolactone).
This rejiggering of the consensus expert approach for treating HFrEF left cardiologists wondering what sequence to use when starting this quadruple therapy. Within weeks, the answer from heart failure opinion leaders was clear:
“Start all four pillars simultaneously. Most patients can tolerate, and will benefit from, a simultaneous start,” declared Dr. Fonarow, professor and chief of cardiology at the University of California, Los Angeles.
His rationale? Patients get benefits from each of these drug classes “surprisingly early,” with improved outcomes in clinical trials appearing within a few weeks, compared with patients in control arms. The consequence is that any delay in starting treatment denies patients time with improved health status, function, and survival.
Study results documented that the four foundational drug classes can produce rapid improvements in health status, left ventricular size and shape, and make clinically meaningful cuts in both first and recurrent hospitalizations for heart failure and in mortality, Dr. Fonarow said. After 30 days on quadruple treatment, a patient’s relative risk for death drops by more than three-quarters, compared with patients not on these medications.
The benefits from each of the four classes involve distinct physiologic pathways and hence are not diminished by concurrent treatment. And immediate initiation avoids the risk of clinical inertia and a negligence to prescribe one or more of the four important drug classes. Introducing the four classes in a sequential manner could mean spending as long as a year to get all four on board and up-titrated to optimal therapeutic levels, he noted.
“Overcome inertia by prescribing [all four drug classes] at the time of diagnosis,” Dr. Fonarow admonished his audience.
The challenge of prescribing inertia
The risk for inertia in prescribing heart failure medications is real. Data collected in the CHAMP-HF (Change the Management of Patients with Heart Failure) registry from more than 3,500 HFrEF patients managed at any of 150 U.S. primary care and cardiology practices starting in late 2015 and continuing through 2017 showed that, among patients eligible for treatment with renin-angiotensin system (RAS) inhibition (with either ARNI or a single RAS inhibiting drug), a beta-blocker, and a mineralocorticoid receptor antagonist (MRA), 22% received all three drug classes. A scant 1% were on target dosages of all three drug classes, noted Stephen J. Greene, MD, in a separate talk at the meeting when he cited his published findings.
The sole formulation currently in the ARNI class, sacubitril/valsartan (Entresto) has in recent years been the poster child for prescribing inertia in HFrEF patients after coming onto the U.S. market for routine use in 2015. A review run by Dr. Greene of more than 9,000 HFrEF patients who were at least 65 years old and discharged from a hospital participating in the Get With the Guidelines–Heart Failure registry during October 2015–September 2017 showed that 8% of eligible patients actually received a sacubitril/valsartan prescription. Separate assessment of outpatients with HFrEF from the same era showed 13% uptake, said D. Greene, a cardiologist at Duke University, Durham, N.C.
Substantial gaps in prescribing evidence-based treatments to HFrEF patients have existed for the past couple of decades, said Dr. Greene. “Even a blockbuster drug like sacubitril/valsartan has been slow to implement.”
Quadruple therapy adds an average of 6 years of life
One of the most strongest arguments favoring the start-four-at-once approach was detailed in what’s quickly become a widely cited analysis published in July 2020 by a team of researchers led by Muthiah Vaduganathan, MD. Using data from three key pivotal trials they estimated that timely treatment with all four drug classes would on average produce an extra 6 years of overall survival in a 55-year old HFrEF patient, and an added 8 years free from cardiovascular death or first hospitalization for heart failure, compared with less comprehensive treatment. The analysis also showed a significant 3-year average boost in overall survival among HFrEF patients who were 80 years old when using quadruple therapy compared with the “conventional medical therapy” used on control patients in the three trials examined.
Dr. Greene called these findings “remarkable.”
“Four drugs use five mechanistic pathways to produce 6 added years of survival,” summed up Dr. Vaduganathan during a separate talk at the virtual meeting.
In addition to this substantial potential for a meaningful impact on patents’ lives, he cited other factors that add to the case for early prescription of the pharmaceutical gauntlet: avoiding missed treatment opportunities that occur with slower, step-wise drug introduction; simplifying, streamlining, and standardizing the care pathway, which helps avoid care inequities and disrupts the potential for inertia; magnifying benefit when comprehensive treatment starts sooner; and providing additive benefits without drug-drug interactions.
“Upfront treatment at the time of [HFrEF] diagnosis or hospitalization is an approach that disrupts treatment inertia,” emphasized Dr. Vaduganathan, a cardiologist at Brigham and Women’s Hospital in Boston.
New approaches needed to encourage quick uptake
“Efficacy alone has not been enough for efficient uptake in U.S. practice” of sacubitril/valsartan, other RAS inhibitors, beta-blockers, and MRAs, noted Dr. Greene.
He was more optimistic about prospects for relatively quick uptake of early SGLT2 inhibitor treatment as part of routine HFrEF management given all the positives that this new HFrEF treatment offers, including some “unique features” among HFrEF drugs. These include the simplicity of the regimen, which involves a single dosage for everyone that’s taken once daily; minimal blood pressure effects and no adverse renal effects while also producing substantial renal protection; and two SGLT2 inhibitors with proven HFrEF benefit (dapagliflozin and empagliflozin), which bodes well for an eventual price drop.
The SGLT2 inhibitors stack up as an “ideal” HFrEF treatment, concluded Dr. Greene, which should facilitate quick uptake. As far as getting clinicians to also add early on the other three members of the core four treatment classes in routine treatment, he conceded that “innovative and evidence-based approaches to improving real-world uptake of guideline-directed medical therapy are urgently needed.”
EMPEROR-Reduced was funded by Boehringer Ingelheim and Lilly, the companies that market empagliflozin (Jardiance). CHAMP-HF was funded by Novartis, the company that markets sacubitril/valsartan (Entresto). Dr. Fonarow has been a consultant or adviser to Novartis, as well as to Abbott, Amgen, AstraZeneca, Bayer, CHF Solutions, Edwards, Janssen, Medtronic, and Merck. Dr. Greene has received research funding from Novartis, has been a consultant to Amgen and Merck, an adviser to Amgen and Cytokinetics, and has received research funding from Amgen, AstraZeneca, Bristol-Myers Squibb, and Merck. Dr. Vaduganathan has had financial relationships with Boehringer Ingelheim and Novartis, as well as with Amgen, AstraZeneca, Baxter Healthcare, Bayer, Cytokinetics, and Relypsa.
Gregg C. Fonarow, MD, recommended.
Less than 2 months before Dr. Fonarow made that striking statement during the virtual annual meeting of the Heart Failure Society of America, investigators first reported results from the EMPEROR-Reduced trial at the European Society of Cardiology’s virtual annual meeting, showing that the sodium-glucose transporter 2 (SGLT2) inhibitor empagliflozin (Jardiance) successfully cut events in patients with heart failure with reduced ejection fraction (HFrEF). That report, a year after results from a similar trial (DAPA-HF) showed the same outcome using a different drug from the same class, dapagliflozin (Farxiga), cemented the SGLT2 inhibitor drug class as the fourth pillar for treating HFrEF, joining the angiotensin receptor neprilysin inhibitor (ARNI) class (sacubitril valsartan), beta-blockers (like carvedilol), and mineralocorticoid receptor antagonists (like spironolactone).
This rejiggering of the consensus expert approach for treating HFrEF left cardiologists wondering what sequence to use when starting this quadruple therapy. Within weeks, the answer from heart failure opinion leaders was clear:
“Start all four pillars simultaneously. Most patients can tolerate, and will benefit from, a simultaneous start,” declared Dr. Fonarow, professor and chief of cardiology at the University of California, Los Angeles.
His rationale? Patients get benefits from each of these drug classes “surprisingly early,” with improved outcomes in clinical trials appearing within a few weeks, compared with patients in control arms. The consequence is that any delay in starting treatment denies patients time with improved health status, function, and survival.
Study results documented that the four foundational drug classes can produce rapid improvements in health status, left ventricular size and shape, and make clinically meaningful cuts in both first and recurrent hospitalizations for heart failure and in mortality, Dr. Fonarow said. After 30 days on quadruple treatment, a patient’s relative risk for death drops by more than three-quarters, compared with patients not on these medications.
The benefits from each of the four classes involve distinct physiologic pathways and hence are not diminished by concurrent treatment. And immediate initiation avoids the risk of clinical inertia and a negligence to prescribe one or more of the four important drug classes. Introducing the four classes in a sequential manner could mean spending as long as a year to get all four on board and up-titrated to optimal therapeutic levels, he noted.
“Overcome inertia by prescribing [all four drug classes] at the time of diagnosis,” Dr. Fonarow admonished his audience.
The challenge of prescribing inertia
The risk for inertia in prescribing heart failure medications is real. Data collected in the CHAMP-HF (Change the Management of Patients with Heart Failure) registry from more than 3,500 HFrEF patients managed at any of 150 U.S. primary care and cardiology practices starting in late 2015 and continuing through 2017 showed that, among patients eligible for treatment with renin-angiotensin system (RAS) inhibition (with either ARNI or a single RAS inhibiting drug), a beta-blocker, and a mineralocorticoid receptor antagonist (MRA), 22% received all three drug classes. A scant 1% were on target dosages of all three drug classes, noted Stephen J. Greene, MD, in a separate talk at the meeting when he cited his published findings.
The sole formulation currently in the ARNI class, sacubitril/valsartan (Entresto) has in recent years been the poster child for prescribing inertia in HFrEF patients after coming onto the U.S. market for routine use in 2015. A review run by Dr. Greene of more than 9,000 HFrEF patients who were at least 65 years old and discharged from a hospital participating in the Get With the Guidelines–Heart Failure registry during October 2015–September 2017 showed that 8% of eligible patients actually received a sacubitril/valsartan prescription. Separate assessment of outpatients with HFrEF from the same era showed 13% uptake, said D. Greene, a cardiologist at Duke University, Durham, N.C.
Substantial gaps in prescribing evidence-based treatments to HFrEF patients have existed for the past couple of decades, said Dr. Greene. “Even a blockbuster drug like sacubitril/valsartan has been slow to implement.”
Quadruple therapy adds an average of 6 years of life
One of the most strongest arguments favoring the start-four-at-once approach was detailed in what’s quickly become a widely cited analysis published in July 2020 by a team of researchers led by Muthiah Vaduganathan, MD. Using data from three key pivotal trials they estimated that timely treatment with all four drug classes would on average produce an extra 6 years of overall survival in a 55-year old HFrEF patient, and an added 8 years free from cardiovascular death or first hospitalization for heart failure, compared with less comprehensive treatment. The analysis also showed a significant 3-year average boost in overall survival among HFrEF patients who were 80 years old when using quadruple therapy compared with the “conventional medical therapy” used on control patients in the three trials examined.
Dr. Greene called these findings “remarkable.”
“Four drugs use five mechanistic pathways to produce 6 added years of survival,” summed up Dr. Vaduganathan during a separate talk at the virtual meeting.
In addition to this substantial potential for a meaningful impact on patents’ lives, he cited other factors that add to the case for early prescription of the pharmaceutical gauntlet: avoiding missed treatment opportunities that occur with slower, step-wise drug introduction; simplifying, streamlining, and standardizing the care pathway, which helps avoid care inequities and disrupts the potential for inertia; magnifying benefit when comprehensive treatment starts sooner; and providing additive benefits without drug-drug interactions.
“Upfront treatment at the time of [HFrEF] diagnosis or hospitalization is an approach that disrupts treatment inertia,” emphasized Dr. Vaduganathan, a cardiologist at Brigham and Women’s Hospital in Boston.
New approaches needed to encourage quick uptake
“Efficacy alone has not been enough for efficient uptake in U.S. practice” of sacubitril/valsartan, other RAS inhibitors, beta-blockers, and MRAs, noted Dr. Greene.
He was more optimistic about prospects for relatively quick uptake of early SGLT2 inhibitor treatment as part of routine HFrEF management given all the positives that this new HFrEF treatment offers, including some “unique features” among HFrEF drugs. These include the simplicity of the regimen, which involves a single dosage for everyone that’s taken once daily; minimal blood pressure effects and no adverse renal effects while also producing substantial renal protection; and two SGLT2 inhibitors with proven HFrEF benefit (dapagliflozin and empagliflozin), which bodes well for an eventual price drop.
The SGLT2 inhibitors stack up as an “ideal” HFrEF treatment, concluded Dr. Greene, which should facilitate quick uptake. As far as getting clinicians to also add early on the other three members of the core four treatment classes in routine treatment, he conceded that “innovative and evidence-based approaches to improving real-world uptake of guideline-directed medical therapy are urgently needed.”
EMPEROR-Reduced was funded by Boehringer Ingelheim and Lilly, the companies that market empagliflozin (Jardiance). CHAMP-HF was funded by Novartis, the company that markets sacubitril/valsartan (Entresto). Dr. Fonarow has been a consultant or adviser to Novartis, as well as to Abbott, Amgen, AstraZeneca, Bayer, CHF Solutions, Edwards, Janssen, Medtronic, and Merck. Dr. Greene has received research funding from Novartis, has been a consultant to Amgen and Merck, an adviser to Amgen and Cytokinetics, and has received research funding from Amgen, AstraZeneca, Bristol-Myers Squibb, and Merck. Dr. Vaduganathan has had financial relationships with Boehringer Ingelheim and Novartis, as well as with Amgen, AstraZeneca, Baxter Healthcare, Bayer, Cytokinetics, and Relypsa.
Gregg C. Fonarow, MD, recommended.
Less than 2 months before Dr. Fonarow made that striking statement during the virtual annual meeting of the Heart Failure Society of America, investigators first reported results from the EMPEROR-Reduced trial at the European Society of Cardiology’s virtual annual meeting, showing that the sodium-glucose transporter 2 (SGLT2) inhibitor empagliflozin (Jardiance) successfully cut events in patients with heart failure with reduced ejection fraction (HFrEF). That report, a year after results from a similar trial (DAPA-HF) showed the same outcome using a different drug from the same class, dapagliflozin (Farxiga), cemented the SGLT2 inhibitor drug class as the fourth pillar for treating HFrEF, joining the angiotensin receptor neprilysin inhibitor (ARNI) class (sacubitril valsartan), beta-blockers (like carvedilol), and mineralocorticoid receptor antagonists (like spironolactone).
This rejiggering of the consensus expert approach for treating HFrEF left cardiologists wondering what sequence to use when starting this quadruple therapy. Within weeks, the answer from heart failure opinion leaders was clear:
“Start all four pillars simultaneously. Most patients can tolerate, and will benefit from, a simultaneous start,” declared Dr. Fonarow, professor and chief of cardiology at the University of California, Los Angeles.
His rationale? Patients get benefits from each of these drug classes “surprisingly early,” with improved outcomes in clinical trials appearing within a few weeks, compared with patients in control arms. The consequence is that any delay in starting treatment denies patients time with improved health status, function, and survival.
Study results documented that the four foundational drug classes can produce rapid improvements in health status, left ventricular size and shape, and make clinically meaningful cuts in both first and recurrent hospitalizations for heart failure and in mortality, Dr. Fonarow said. After 30 days on quadruple treatment, a patient’s relative risk for death drops by more than three-quarters, compared with patients not on these medications.
The benefits from each of the four classes involve distinct physiologic pathways and hence are not diminished by concurrent treatment. And immediate initiation avoids the risk of clinical inertia and a negligence to prescribe one or more of the four important drug classes. Introducing the four classes in a sequential manner could mean spending as long as a year to get all four on board and up-titrated to optimal therapeutic levels, he noted.
“Overcome inertia by prescribing [all four drug classes] at the time of diagnosis,” Dr. Fonarow admonished his audience.
The challenge of prescribing inertia
The risk for inertia in prescribing heart failure medications is real. Data collected in the CHAMP-HF (Change the Management of Patients with Heart Failure) registry from more than 3,500 HFrEF patients managed at any of 150 U.S. primary care and cardiology practices starting in late 2015 and continuing through 2017 showed that, among patients eligible for treatment with renin-angiotensin system (RAS) inhibition (with either ARNI or a single RAS inhibiting drug), a beta-blocker, and a mineralocorticoid receptor antagonist (MRA), 22% received all three drug classes. A scant 1% were on target dosages of all three drug classes, noted Stephen J. Greene, MD, in a separate talk at the meeting when he cited his published findings.
The sole formulation currently in the ARNI class, sacubitril/valsartan (Entresto) has in recent years been the poster child for prescribing inertia in HFrEF patients after coming onto the U.S. market for routine use in 2015. A review run by Dr. Greene of more than 9,000 HFrEF patients who were at least 65 years old and discharged from a hospital participating in the Get With the Guidelines–Heart Failure registry during October 2015–September 2017 showed that 8% of eligible patients actually received a sacubitril/valsartan prescription. Separate assessment of outpatients with HFrEF from the same era showed 13% uptake, said D. Greene, a cardiologist at Duke University, Durham, N.C.
Substantial gaps in prescribing evidence-based treatments to HFrEF patients have existed for the past couple of decades, said Dr. Greene. “Even a blockbuster drug like sacubitril/valsartan has been slow to implement.”
Quadruple therapy adds an average of 6 years of life
One of the most strongest arguments favoring the start-four-at-once approach was detailed in what’s quickly become a widely cited analysis published in July 2020 by a team of researchers led by Muthiah Vaduganathan, MD. Using data from three key pivotal trials they estimated that timely treatment with all four drug classes would on average produce an extra 6 years of overall survival in a 55-year old HFrEF patient, and an added 8 years free from cardiovascular death or first hospitalization for heart failure, compared with less comprehensive treatment. The analysis also showed a significant 3-year average boost in overall survival among HFrEF patients who were 80 years old when using quadruple therapy compared with the “conventional medical therapy” used on control patients in the three trials examined.
Dr. Greene called these findings “remarkable.”
“Four drugs use five mechanistic pathways to produce 6 added years of survival,” summed up Dr. Vaduganathan during a separate talk at the virtual meeting.
In addition to this substantial potential for a meaningful impact on patents’ lives, he cited other factors that add to the case for early prescription of the pharmaceutical gauntlet: avoiding missed treatment opportunities that occur with slower, step-wise drug introduction; simplifying, streamlining, and standardizing the care pathway, which helps avoid care inequities and disrupts the potential for inertia; magnifying benefit when comprehensive treatment starts sooner; and providing additive benefits without drug-drug interactions.
“Upfront treatment at the time of [HFrEF] diagnosis or hospitalization is an approach that disrupts treatment inertia,” emphasized Dr. Vaduganathan, a cardiologist at Brigham and Women’s Hospital in Boston.
New approaches needed to encourage quick uptake
“Efficacy alone has not been enough for efficient uptake in U.S. practice” of sacubitril/valsartan, other RAS inhibitors, beta-blockers, and MRAs, noted Dr. Greene.
He was more optimistic about prospects for relatively quick uptake of early SGLT2 inhibitor treatment as part of routine HFrEF management given all the positives that this new HFrEF treatment offers, including some “unique features” among HFrEF drugs. These include the simplicity of the regimen, which involves a single dosage for everyone that’s taken once daily; minimal blood pressure effects and no adverse renal effects while also producing substantial renal protection; and two SGLT2 inhibitors with proven HFrEF benefit (dapagliflozin and empagliflozin), which bodes well for an eventual price drop.
The SGLT2 inhibitors stack up as an “ideal” HFrEF treatment, concluded Dr. Greene, which should facilitate quick uptake. As far as getting clinicians to also add early on the other three members of the core four treatment classes in routine treatment, he conceded that “innovative and evidence-based approaches to improving real-world uptake of guideline-directed medical therapy are urgently needed.”
EMPEROR-Reduced was funded by Boehringer Ingelheim and Lilly, the companies that market empagliflozin (Jardiance). CHAMP-HF was funded by Novartis, the company that markets sacubitril/valsartan (Entresto). Dr. Fonarow has been a consultant or adviser to Novartis, as well as to Abbott, Amgen, AstraZeneca, Bayer, CHF Solutions, Edwards, Janssen, Medtronic, and Merck. Dr. Greene has received research funding from Novartis, has been a consultant to Amgen and Merck, an adviser to Amgen and Cytokinetics, and has received research funding from Amgen, AstraZeneca, Bristol-Myers Squibb, and Merck. Dr. Vaduganathan has had financial relationships with Boehringer Ingelheim and Novartis, as well as with Amgen, AstraZeneca, Baxter Healthcare, Bayer, Cytokinetics, and Relypsa.
FROM HFSA 2020
‘Modest’ benefit for post-MI T2D glucose monitoring
Following a heart attack, there appears to be a “modest” benefit of using flash glucose monitoring over fingerstick testing to monitor blood glucose levels in patients with type 2 diabetes being treated with insulin or a sulfonylurea, according to investigators of the LIBERATES trial.
The results showed a nonsignificant increase in the time that subjects’ blood glucose was spent in the target range of 3.9-10.00 mmol/L (70-180 mg/dL) 3 months after experiencing an acute coronary syndrome (ACS).
At best, flash monitoring using Abbott’s Freestyle Libre system was associated with an increase in time spent in range (TIR) of 17-28 or 48 minutes per day over self-monitoring of blood glucose (SMBG), depending on the type of statistical analysis used. There was no difference in glycated hemoglobin A1c levels between the two groups, but there was a trend for less time spent in hypoglycemia in the flash monitoring arm.
Viewers underwhelmed
“My overall impression is that the effects were less pronounced than anticipated,” Kare Birkeland, MD, PhD, a specialist in internal medicine and endocrinology at Oslo University Hospital, Rikshospitalet, Norway, observed after the findings were presented at the virtual annual meeting of the European Association for the Study of Diabetes.
Others who had watched the live session seemed similarly underwhelmed by the findings, with one viewer questioning the value of devoting an hour-and-a-half session to the phase 2 trial.
However, the session chair Simon Heller, BA, MB, BChir, DM, professor of clinical diabetes at the University of Sheffield, and trial coinvestigator, defended the detailed look at the trial’s findings, noting that it was worthwhile to present the data from the trial as it “really helps explain why we do phase 2 and phase 3 trials.”
Strong rationale for monitoring post-MI
There is a strong rationale for ensuring that blood glucose is well controlled in type 2 diabetes patients who have experienced a myocardial infarction, observed Robert Storey, BSc, BM, DM, professor of cardiology at the University of Sheffield. One way to do that potentially is through improved glucose monitoring.
“There’s clearly a close link between diabetes and the risk of MI: Both high and low HbA1c are associated with adverse outcome, and high and low glucose levels following MI are also associated with adverse outcome,” he observed, noting also that hypoglycemia was not given enough attention in post-ACS patients.
“The hypothesis of the LIBERATES study was that a modern glycemic monitoring strategy can optimize blood glucose levels in type 2 diabetes patients following MI with the potential to reduce mortality and morbidity and improve quality of life,” Dr. Storey said. “The main research question of LIBERATES says, ‘Do new approaches in glucose monitoring increase the time in range and reduce hypoglycemia?’ ”
Pragmatic trial design
LIBERATES was a prospective, multicenter, parallel group, randomized controlled trial, explained the study’s statistician Deborah Stocken, PhD, professor of clinical trials research at the University of Leeds. There was “limited ability to blind the interventions,” so it was an open-label design.
“The patient population in LIBERATES was kept as inclusive and as pragmatic as possible to ensure that the results at the end of the trial are generalizable,” said Dr. Stocken. Patients with type 2 diabetes were recruited within 5 days of hospital admission for ACS, which could include both ST- and non-ST elevation MI. In all, 141 of a calculated 150 patients that would be needed were recruited and randomized to the flash monitoring (69) or SMBG (72) arm.
Dr. Stocken noted that early in the recruitment phase, the trials oversight committee recommended that Bayesian methodology should be used as the most robust analytical approach.
“Essentially, a Bayesian approach would avoid a hypothesis test, and instead would provide a probability of there being a treatment benefit for continuous monitoring. And if this probability was high enough, this would warrant further research in the phase 3 setting,” Dr. Stocken said.
What else was shown?
“We had a number of prespecified secondary endpoints, which to me are equally important,” said Ramzi Ajjan, MD, MMed.Sci, PhD, associate professor and consultant in diabetes and endocrinology at Leeds University and Leeds Teaching Hospitals Trust.
Among these was the TIR at days 16-30, which showed a 90-minute increase per day in favor of flash monitoring over SMBG. This “seems to be driven by those who are an insulin,” Dr. Ajjan said, adding that “you get almost a 3-hour increase in time in range in people who are on insulin at baseline, and you don’t see that in people who are on sulfonylurea.”
Conversely, sulfonylurea treatment seemed to drive the reduction in the time spent in hypoglycemia defined as 3.9 mmol/L (70 g/dL) at 3 months. For the whole group, there was a 1.3-hour reduction in hypoglycemia per day with flash monitoring versus SMBG, which increased to 2 hours for those on sulfonylureas.
There also was a “pattern of reduction” in time spent in hypoglycemia defined as less than 3.0 mmol/L (54 g/dL) both early on and becoming more pronounced with time.
“Flash glucose monitoring is associated with higher treatment satisfaction score, compared with SMBG,” Dr. Ajjan said.
Although A1c dropped in both groups to a similar extent, he noted that the reduction seen in the flash monitoring group was associated with a decrease in hypoglycemia.
There was a huge amount of data collected during the trial and there are many more analyses that could be done, Dr. Ajjan said. The outcome of those may determine whether a phase 3 trial is likely, assuming sponsorship can be secured.
The LIBERATES Trial was funded by grants from the UK National Institute for Health Research and Abbott Diabetes Care. None of the investigators were additionally compensated for their work within the trial. Dr. Stocken had no disclosures in relation to this trial. Dr. Ajjan has received research funding and other financial support from Abbott, Bayer, Eli Lilly, Johnson & Johnson, and Novo Nordisk.
SOURCE: Ajjan R et al. EASD 2020. S11 – The LIBERATES Trial.
Following a heart attack, there appears to be a “modest” benefit of using flash glucose monitoring over fingerstick testing to monitor blood glucose levels in patients with type 2 diabetes being treated with insulin or a sulfonylurea, according to investigators of the LIBERATES trial.
The results showed a nonsignificant increase in the time that subjects’ blood glucose was spent in the target range of 3.9-10.00 mmol/L (70-180 mg/dL) 3 months after experiencing an acute coronary syndrome (ACS).
At best, flash monitoring using Abbott’s Freestyle Libre system was associated with an increase in time spent in range (TIR) of 17-28 or 48 minutes per day over self-monitoring of blood glucose (SMBG), depending on the type of statistical analysis used. There was no difference in glycated hemoglobin A1c levels between the two groups, but there was a trend for less time spent in hypoglycemia in the flash monitoring arm.
Viewers underwhelmed
“My overall impression is that the effects were less pronounced than anticipated,” Kare Birkeland, MD, PhD, a specialist in internal medicine and endocrinology at Oslo University Hospital, Rikshospitalet, Norway, observed after the findings were presented at the virtual annual meeting of the European Association for the Study of Diabetes.
Others who had watched the live session seemed similarly underwhelmed by the findings, with one viewer questioning the value of devoting an hour-and-a-half session to the phase 2 trial.
However, the session chair Simon Heller, BA, MB, BChir, DM, professor of clinical diabetes at the University of Sheffield, and trial coinvestigator, defended the detailed look at the trial’s findings, noting that it was worthwhile to present the data from the trial as it “really helps explain why we do phase 2 and phase 3 trials.”
Strong rationale for monitoring post-MI
There is a strong rationale for ensuring that blood glucose is well controlled in type 2 diabetes patients who have experienced a myocardial infarction, observed Robert Storey, BSc, BM, DM, professor of cardiology at the University of Sheffield. One way to do that potentially is through improved glucose monitoring.
“There’s clearly a close link between diabetes and the risk of MI: Both high and low HbA1c are associated with adverse outcome, and high and low glucose levels following MI are also associated with adverse outcome,” he observed, noting also that hypoglycemia was not given enough attention in post-ACS patients.
“The hypothesis of the LIBERATES study was that a modern glycemic monitoring strategy can optimize blood glucose levels in type 2 diabetes patients following MI with the potential to reduce mortality and morbidity and improve quality of life,” Dr. Storey said. “The main research question of LIBERATES says, ‘Do new approaches in glucose monitoring increase the time in range and reduce hypoglycemia?’ ”
Pragmatic trial design
LIBERATES was a prospective, multicenter, parallel group, randomized controlled trial, explained the study’s statistician Deborah Stocken, PhD, professor of clinical trials research at the University of Leeds. There was “limited ability to blind the interventions,” so it was an open-label design.
“The patient population in LIBERATES was kept as inclusive and as pragmatic as possible to ensure that the results at the end of the trial are generalizable,” said Dr. Stocken. Patients with type 2 diabetes were recruited within 5 days of hospital admission for ACS, which could include both ST- and non-ST elevation MI. In all, 141 of a calculated 150 patients that would be needed were recruited and randomized to the flash monitoring (69) or SMBG (72) arm.
Dr. Stocken noted that early in the recruitment phase, the trials oversight committee recommended that Bayesian methodology should be used as the most robust analytical approach.
“Essentially, a Bayesian approach would avoid a hypothesis test, and instead would provide a probability of there being a treatment benefit for continuous monitoring. And if this probability was high enough, this would warrant further research in the phase 3 setting,” Dr. Stocken said.
What else was shown?
“We had a number of prespecified secondary endpoints, which to me are equally important,” said Ramzi Ajjan, MD, MMed.Sci, PhD, associate professor and consultant in diabetes and endocrinology at Leeds University and Leeds Teaching Hospitals Trust.
Among these was the TIR at days 16-30, which showed a 90-minute increase per day in favor of flash monitoring over SMBG. This “seems to be driven by those who are an insulin,” Dr. Ajjan said, adding that “you get almost a 3-hour increase in time in range in people who are on insulin at baseline, and you don’t see that in people who are on sulfonylurea.”
Conversely, sulfonylurea treatment seemed to drive the reduction in the time spent in hypoglycemia defined as 3.9 mmol/L (70 g/dL) at 3 months. For the whole group, there was a 1.3-hour reduction in hypoglycemia per day with flash monitoring versus SMBG, which increased to 2 hours for those on sulfonylureas.
There also was a “pattern of reduction” in time spent in hypoglycemia defined as less than 3.0 mmol/L (54 g/dL) both early on and becoming more pronounced with time.
“Flash glucose monitoring is associated with higher treatment satisfaction score, compared with SMBG,” Dr. Ajjan said.
Although A1c dropped in both groups to a similar extent, he noted that the reduction seen in the flash monitoring group was associated with a decrease in hypoglycemia.
There was a huge amount of data collected during the trial and there are many more analyses that could be done, Dr. Ajjan said. The outcome of those may determine whether a phase 3 trial is likely, assuming sponsorship can be secured.
The LIBERATES Trial was funded by grants from the UK National Institute for Health Research and Abbott Diabetes Care. None of the investigators were additionally compensated for their work within the trial. Dr. Stocken had no disclosures in relation to this trial. Dr. Ajjan has received research funding and other financial support from Abbott, Bayer, Eli Lilly, Johnson & Johnson, and Novo Nordisk.
SOURCE: Ajjan R et al. EASD 2020. S11 – The LIBERATES Trial.
Following a heart attack, there appears to be a “modest” benefit of using flash glucose monitoring over fingerstick testing to monitor blood glucose levels in patients with type 2 diabetes being treated with insulin or a sulfonylurea, according to investigators of the LIBERATES trial.
The results showed a nonsignificant increase in the time that subjects’ blood glucose was spent in the target range of 3.9-10.00 mmol/L (70-180 mg/dL) 3 months after experiencing an acute coronary syndrome (ACS).
At best, flash monitoring using Abbott’s Freestyle Libre system was associated with an increase in time spent in range (TIR) of 17-28 or 48 minutes per day over self-monitoring of blood glucose (SMBG), depending on the type of statistical analysis used. There was no difference in glycated hemoglobin A1c levels between the two groups, but there was a trend for less time spent in hypoglycemia in the flash monitoring arm.
Viewers underwhelmed
“My overall impression is that the effects were less pronounced than anticipated,” Kare Birkeland, MD, PhD, a specialist in internal medicine and endocrinology at Oslo University Hospital, Rikshospitalet, Norway, observed after the findings were presented at the virtual annual meeting of the European Association for the Study of Diabetes.
Others who had watched the live session seemed similarly underwhelmed by the findings, with one viewer questioning the value of devoting an hour-and-a-half session to the phase 2 trial.
However, the session chair Simon Heller, BA, MB, BChir, DM, professor of clinical diabetes at the University of Sheffield, and trial coinvestigator, defended the detailed look at the trial’s findings, noting that it was worthwhile to present the data from the trial as it “really helps explain why we do phase 2 and phase 3 trials.”
Strong rationale for monitoring post-MI
There is a strong rationale for ensuring that blood glucose is well controlled in type 2 diabetes patients who have experienced a myocardial infarction, observed Robert Storey, BSc, BM, DM, professor of cardiology at the University of Sheffield. One way to do that potentially is through improved glucose monitoring.
“There’s clearly a close link between diabetes and the risk of MI: Both high and low HbA1c are associated with adverse outcome, and high and low glucose levels following MI are also associated with adverse outcome,” he observed, noting also that hypoglycemia was not given enough attention in post-ACS patients.
“The hypothesis of the LIBERATES study was that a modern glycemic monitoring strategy can optimize blood glucose levels in type 2 diabetes patients following MI with the potential to reduce mortality and morbidity and improve quality of life,” Dr. Storey said. “The main research question of LIBERATES says, ‘Do new approaches in glucose monitoring increase the time in range and reduce hypoglycemia?’ ”
Pragmatic trial design
LIBERATES was a prospective, multicenter, parallel group, randomized controlled trial, explained the study’s statistician Deborah Stocken, PhD, professor of clinical trials research at the University of Leeds. There was “limited ability to blind the interventions,” so it was an open-label design.
“The patient population in LIBERATES was kept as inclusive and as pragmatic as possible to ensure that the results at the end of the trial are generalizable,” said Dr. Stocken. Patients with type 2 diabetes were recruited within 5 days of hospital admission for ACS, which could include both ST- and non-ST elevation MI. In all, 141 of a calculated 150 patients that would be needed were recruited and randomized to the flash monitoring (69) or SMBG (72) arm.
Dr. Stocken noted that early in the recruitment phase, the trials oversight committee recommended that Bayesian methodology should be used as the most robust analytical approach.
“Essentially, a Bayesian approach would avoid a hypothesis test, and instead would provide a probability of there being a treatment benefit for continuous monitoring. And if this probability was high enough, this would warrant further research in the phase 3 setting,” Dr. Stocken said.
What else was shown?
“We had a number of prespecified secondary endpoints, which to me are equally important,” said Ramzi Ajjan, MD, MMed.Sci, PhD, associate professor and consultant in diabetes and endocrinology at Leeds University and Leeds Teaching Hospitals Trust.
Among these was the TIR at days 16-30, which showed a 90-minute increase per day in favor of flash monitoring over SMBG. This “seems to be driven by those who are an insulin,” Dr. Ajjan said, adding that “you get almost a 3-hour increase in time in range in people who are on insulin at baseline, and you don’t see that in people who are on sulfonylurea.”
Conversely, sulfonylurea treatment seemed to drive the reduction in the time spent in hypoglycemia defined as 3.9 mmol/L (70 g/dL) at 3 months. For the whole group, there was a 1.3-hour reduction in hypoglycemia per day with flash monitoring versus SMBG, which increased to 2 hours for those on sulfonylureas.
There also was a “pattern of reduction” in time spent in hypoglycemia defined as less than 3.0 mmol/L (54 g/dL) both early on and becoming more pronounced with time.
“Flash glucose monitoring is associated with higher treatment satisfaction score, compared with SMBG,” Dr. Ajjan said.
Although A1c dropped in both groups to a similar extent, he noted that the reduction seen in the flash monitoring group was associated with a decrease in hypoglycemia.
There was a huge amount of data collected during the trial and there are many more analyses that could be done, Dr. Ajjan said. The outcome of those may determine whether a phase 3 trial is likely, assuming sponsorship can be secured.
The LIBERATES Trial was funded by grants from the UK National Institute for Health Research and Abbott Diabetes Care. None of the investigators were additionally compensated for their work within the trial. Dr. Stocken had no disclosures in relation to this trial. Dr. Ajjan has received research funding and other financial support from Abbott, Bayer, Eli Lilly, Johnson & Johnson, and Novo Nordisk.
SOURCE: Ajjan R et al. EASD 2020. S11 – The LIBERATES Trial.
FROM EASD 2020