User login
MitraClip effective for post-MI acute mitral regurgitation with cardiogenic shock
Percutaneous mitral valve repair with the MitraClip appears to be a safe, effective, and life-saving new treatment for severe acute mitral regurgitation (MR) secondary to MI in surgical noncandidates, even when accompanied by cardiogenic shock, according to data from the international IREMMI registry.
“Cardiogenic shock, when adequately supported, does not seem to influence short- and mid-term outcomes, so the development of cardiogenic shock should not preclude percutaneous mitral valve repair in this scenario,” Rodrigo Estevez-Loureiro, MD, PhD, said in presenting the IREMMI (International Registry of MitraClip in Acute Myocardial Infarction) findings reported at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
Commentators hailed the prospective IREMMI data as potentially practice changing in light of the dire prognosis of such patients when surgery is deemed unacceptably high risk because medical management, the traditionally the only alternative, has a 30-day mortality of up to 50%.
Severe acute MR occurs in an estimated 3% of acute MIs, and in roughly 10% of patients who present with acute MI complicated by cardiogenic shock (CS). The impact of intervening with the MitraClip in an effort to correct the acute MR arising from MI with CS has previously been addressed only in sparse case reports. The new IREMMI study is easily the largest dataset to date detailing clinical and echocardiographic outcomes, Dr. Estevez-Loureiro of Alvaro Cunqueiro Hospital in Vigo, Spain, said at the meeting, sponsored by the Cardiovascular Research Foundation.
He reported on 93 consecutive patients who underwent MitraClip implantation for acute MR arising in the setting of MI, including 50 patients in CS at the time of the procedure. All 93 patients had been turned down by their surgical team because of extreme surgical risk. Three-quarters of the MIs showed ST-segment elevation. Only six patients had a papillary muscle rupture; in the rest, the mechanism of acute MR involved left ventricular global remodeling associated with mitral valve leaflet tethering. Percutaneous valve repair was performed at 18 expert valvular heart centers in the United States, Canada, Israel, and five European countries.
Procedural success
Time from MI to MitraClip implantation averaged 24 days in the CS patients and 33 days in the comparator arm without CS.
“These patients had been turned down for surgery, so the attending physicians generally followed a strategy of trying to cool them down with mechanical circulatory support and vasopressors. MitraClip wasn’t an option at the beginning, but after two or three failed weanings from all the possible therapies, then MitraClip becomes an option. This is one of the reasons why the time lapse between MI and the clip is so large,” the cardiologist explained.
Procedural success rates were similar in the two groups: 90% in those with CS and 93% in those without. However, average procedure time was significantly longer in the CS patients: 143 minutes versus 83 minutes in the patients without CS.
At baseline, 86% of the CS group had grade 4+ MR, similar to the 79% rate in the non-CS patients. Postprocedurally, 60% of the CS group were MR grade 0/1 and 34% were grade 2, comparable to the rates of 65% and 23% in the non-CS group.
At 3 months’ follow-up, 83.4% of the CS group had MR grade 2 or less, again not significantly different from the 90.5% rate in non-CS patients. Systolic pulmonary artery pressure was also similar: 39.6 mm Hg in the CS patients, 44 mm Hg in those without. While everyone was New York Heart Association functional class IV preprocedurally, 79.5% of the CS group were NYHA class I or II at 3 months, not significantly different from the 86.5% prevalence in the comparator arm.
Longer-term clinical outcomes
At a median follow-up of 7 months, the composite primary clinical outcome composed of all-cause mortality or heart failure rehospitalization did not differ between the two groups: a 28% rate in the CS group and 25.6% in non-CS patients. All-cause mortality occurred in 16% with CS and 9.3% without, again not a significant difference.
In a Cox regression analysis, neither surgical risk score, patient age, left ventricular geometry, nor CS was independently associated with the primary composite endpoint. Indeed, the only independent predictor of freedom from mortality or heart failure readmission at follow-up was procedural success, which is very much a function of the experience of the heart team, Dr. Estevez-Loureiro continued.
Michael A. Borger, MD, PhD, who comoderated the late-breaking clinical science session, was wowed by the IREMMI results.
“The mortality rates, I can tell you, compared to traditional surgical series of acute MR in the face of ACS [acute cardiogenic shock] are very, very respectable,” commented Dr. Borger, director of the cardiac surgery clinic at the Leipzig (Ger.) University Heart Center.
“Extremely impressive,” agreed discussant Vinayak N. Bapat, MD, a cardiothoracic surgeon and valve scientist at the Minneapolis Heart Institute Foundation. He posed a practical question: “Should we take from this presentation that patients should be stabilized with something like ECMO [extracorporeal membrane oxygenation] or Impella [left ventricular assist device], then transferred to an expert center for the procedure?”
“I think that the stabilization is essential in the patients with cardiogenic shock,” Dr. Estevez-Loureiro replied. “Unlike with surgery, it’s very difficult to establish a MitraClip procedure in a couple of hours in the middle of the night. You have to stabilize them and then treat for shock with ECMO, Impella, or both. I think they should be transferred to a center than can deliver the best treatment. In centers with less experience, patients can be put on mechanical support and transferred to an expert valve center, not only for MitraClip implantation, but for discussion of all the treatment possibilities, including surgery.”
At a press conference in which Dr. Estevez-Loureiro presented highlights of the IREMMI study, discussant Dee Dee Wang, MD, said the international coinvestigators “need to be applauded” for this study.
“Having these outcomes is incredible,” declared Dr. Wang, a structural heart disease specialist at the Henry Ford Health System, Detroit.
While this is an observational study, it’s a high-quality dataset with excellent methodology. And conducting a randomized trial in patients with such high surgical risk scores – the CS group had an average EuroSCORE II of 21 – would be extremely difficult, according to the cardiologist.
Dr. Estevez-Loureiro reported receiving research grants from Abbott and serving as a consultant to that company as well as Boston Scientific.
SOURCE: Estevez-Loureiro, R. TCT 2020, LBCS session IV.
Percutaneous mitral valve repair with the MitraClip appears to be a safe, effective, and life-saving new treatment for severe acute mitral regurgitation (MR) secondary to MI in surgical noncandidates, even when accompanied by cardiogenic shock, according to data from the international IREMMI registry.
“Cardiogenic shock, when adequately supported, does not seem to influence short- and mid-term outcomes, so the development of cardiogenic shock should not preclude percutaneous mitral valve repair in this scenario,” Rodrigo Estevez-Loureiro, MD, PhD, said in presenting the IREMMI (International Registry of MitraClip in Acute Myocardial Infarction) findings reported at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
Commentators hailed the prospective IREMMI data as potentially practice changing in light of the dire prognosis of such patients when surgery is deemed unacceptably high risk because medical management, the traditionally the only alternative, has a 30-day mortality of up to 50%.
Severe acute MR occurs in an estimated 3% of acute MIs, and in roughly 10% of patients who present with acute MI complicated by cardiogenic shock (CS). The impact of intervening with the MitraClip in an effort to correct the acute MR arising from MI with CS has previously been addressed only in sparse case reports. The new IREMMI study is easily the largest dataset to date detailing clinical and echocardiographic outcomes, Dr. Estevez-Loureiro of Alvaro Cunqueiro Hospital in Vigo, Spain, said at the meeting, sponsored by the Cardiovascular Research Foundation.
He reported on 93 consecutive patients who underwent MitraClip implantation for acute MR arising in the setting of MI, including 50 patients in CS at the time of the procedure. All 93 patients had been turned down by their surgical team because of extreme surgical risk. Three-quarters of the MIs showed ST-segment elevation. Only six patients had a papillary muscle rupture; in the rest, the mechanism of acute MR involved left ventricular global remodeling associated with mitral valve leaflet tethering. Percutaneous valve repair was performed at 18 expert valvular heart centers in the United States, Canada, Israel, and five European countries.
Procedural success
Time from MI to MitraClip implantation averaged 24 days in the CS patients and 33 days in the comparator arm without CS.
“These patients had been turned down for surgery, so the attending physicians generally followed a strategy of trying to cool them down with mechanical circulatory support and vasopressors. MitraClip wasn’t an option at the beginning, but after two or three failed weanings from all the possible therapies, then MitraClip becomes an option. This is one of the reasons why the time lapse between MI and the clip is so large,” the cardiologist explained.
Procedural success rates were similar in the two groups: 90% in those with CS and 93% in those without. However, average procedure time was significantly longer in the CS patients: 143 minutes versus 83 minutes in the patients without CS.
At baseline, 86% of the CS group had grade 4+ MR, similar to the 79% rate in the non-CS patients. Postprocedurally, 60% of the CS group were MR grade 0/1 and 34% were grade 2, comparable to the rates of 65% and 23% in the non-CS group.
At 3 months’ follow-up, 83.4% of the CS group had MR grade 2 or less, again not significantly different from the 90.5% rate in non-CS patients. Systolic pulmonary artery pressure was also similar: 39.6 mm Hg in the CS patients, 44 mm Hg in those without. While everyone was New York Heart Association functional class IV preprocedurally, 79.5% of the CS group were NYHA class I or II at 3 months, not significantly different from the 86.5% prevalence in the comparator arm.
Longer-term clinical outcomes
At a median follow-up of 7 months, the composite primary clinical outcome composed of all-cause mortality or heart failure rehospitalization did not differ between the two groups: a 28% rate in the CS group and 25.6% in non-CS patients. All-cause mortality occurred in 16% with CS and 9.3% without, again not a significant difference.
In a Cox regression analysis, neither surgical risk score, patient age, left ventricular geometry, nor CS was independently associated with the primary composite endpoint. Indeed, the only independent predictor of freedom from mortality or heart failure readmission at follow-up was procedural success, which is very much a function of the experience of the heart team, Dr. Estevez-Loureiro continued.
Michael A. Borger, MD, PhD, who comoderated the late-breaking clinical science session, was wowed by the IREMMI results.
“The mortality rates, I can tell you, compared to traditional surgical series of acute MR in the face of ACS [acute cardiogenic shock] are very, very respectable,” commented Dr. Borger, director of the cardiac surgery clinic at the Leipzig (Ger.) University Heart Center.
“Extremely impressive,” agreed discussant Vinayak N. Bapat, MD, a cardiothoracic surgeon and valve scientist at the Minneapolis Heart Institute Foundation. He posed a practical question: “Should we take from this presentation that patients should be stabilized with something like ECMO [extracorporeal membrane oxygenation] or Impella [left ventricular assist device], then transferred to an expert center for the procedure?”
“I think that the stabilization is essential in the patients with cardiogenic shock,” Dr. Estevez-Loureiro replied. “Unlike with surgery, it’s very difficult to establish a MitraClip procedure in a couple of hours in the middle of the night. You have to stabilize them and then treat for shock with ECMO, Impella, or both. I think they should be transferred to a center than can deliver the best treatment. In centers with less experience, patients can be put on mechanical support and transferred to an expert valve center, not only for MitraClip implantation, but for discussion of all the treatment possibilities, including surgery.”
At a press conference in which Dr. Estevez-Loureiro presented highlights of the IREMMI study, discussant Dee Dee Wang, MD, said the international coinvestigators “need to be applauded” for this study.
“Having these outcomes is incredible,” declared Dr. Wang, a structural heart disease specialist at the Henry Ford Health System, Detroit.
While this is an observational study, it’s a high-quality dataset with excellent methodology. And conducting a randomized trial in patients with such high surgical risk scores – the CS group had an average EuroSCORE II of 21 – would be extremely difficult, according to the cardiologist.
Dr. Estevez-Loureiro reported receiving research grants from Abbott and serving as a consultant to that company as well as Boston Scientific.
SOURCE: Estevez-Loureiro, R. TCT 2020, LBCS session IV.
Percutaneous mitral valve repair with the MitraClip appears to be a safe, effective, and life-saving new treatment for severe acute mitral regurgitation (MR) secondary to MI in surgical noncandidates, even when accompanied by cardiogenic shock, according to data from the international IREMMI registry.
“Cardiogenic shock, when adequately supported, does not seem to influence short- and mid-term outcomes, so the development of cardiogenic shock should not preclude percutaneous mitral valve repair in this scenario,” Rodrigo Estevez-Loureiro, MD, PhD, said in presenting the IREMMI (International Registry of MitraClip in Acute Myocardial Infarction) findings reported at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
Commentators hailed the prospective IREMMI data as potentially practice changing in light of the dire prognosis of such patients when surgery is deemed unacceptably high risk because medical management, the traditionally the only alternative, has a 30-day mortality of up to 50%.
Severe acute MR occurs in an estimated 3% of acute MIs, and in roughly 10% of patients who present with acute MI complicated by cardiogenic shock (CS). The impact of intervening with the MitraClip in an effort to correct the acute MR arising from MI with CS has previously been addressed only in sparse case reports. The new IREMMI study is easily the largest dataset to date detailing clinical and echocardiographic outcomes, Dr. Estevez-Loureiro of Alvaro Cunqueiro Hospital in Vigo, Spain, said at the meeting, sponsored by the Cardiovascular Research Foundation.
He reported on 93 consecutive patients who underwent MitraClip implantation for acute MR arising in the setting of MI, including 50 patients in CS at the time of the procedure. All 93 patients had been turned down by their surgical team because of extreme surgical risk. Three-quarters of the MIs showed ST-segment elevation. Only six patients had a papillary muscle rupture; in the rest, the mechanism of acute MR involved left ventricular global remodeling associated with mitral valve leaflet tethering. Percutaneous valve repair was performed at 18 expert valvular heart centers in the United States, Canada, Israel, and five European countries.
Procedural success
Time from MI to MitraClip implantation averaged 24 days in the CS patients and 33 days in the comparator arm without CS.
“These patients had been turned down for surgery, so the attending physicians generally followed a strategy of trying to cool them down with mechanical circulatory support and vasopressors. MitraClip wasn’t an option at the beginning, but after two or three failed weanings from all the possible therapies, then MitraClip becomes an option. This is one of the reasons why the time lapse between MI and the clip is so large,” the cardiologist explained.
Procedural success rates were similar in the two groups: 90% in those with CS and 93% in those without. However, average procedure time was significantly longer in the CS patients: 143 minutes versus 83 minutes in the patients without CS.
At baseline, 86% of the CS group had grade 4+ MR, similar to the 79% rate in the non-CS patients. Postprocedurally, 60% of the CS group were MR grade 0/1 and 34% were grade 2, comparable to the rates of 65% and 23% in the non-CS group.
At 3 months’ follow-up, 83.4% of the CS group had MR grade 2 or less, again not significantly different from the 90.5% rate in non-CS patients. Systolic pulmonary artery pressure was also similar: 39.6 mm Hg in the CS patients, 44 mm Hg in those without. While everyone was New York Heart Association functional class IV preprocedurally, 79.5% of the CS group were NYHA class I or II at 3 months, not significantly different from the 86.5% prevalence in the comparator arm.
Longer-term clinical outcomes
At a median follow-up of 7 months, the composite primary clinical outcome composed of all-cause mortality or heart failure rehospitalization did not differ between the two groups: a 28% rate in the CS group and 25.6% in non-CS patients. All-cause mortality occurred in 16% with CS and 9.3% without, again not a significant difference.
In a Cox regression analysis, neither surgical risk score, patient age, left ventricular geometry, nor CS was independently associated with the primary composite endpoint. Indeed, the only independent predictor of freedom from mortality or heart failure readmission at follow-up was procedural success, which is very much a function of the experience of the heart team, Dr. Estevez-Loureiro continued.
Michael A. Borger, MD, PhD, who comoderated the late-breaking clinical science session, was wowed by the IREMMI results.
“The mortality rates, I can tell you, compared to traditional surgical series of acute MR in the face of ACS [acute cardiogenic shock] are very, very respectable,” commented Dr. Borger, director of the cardiac surgery clinic at the Leipzig (Ger.) University Heart Center.
“Extremely impressive,” agreed discussant Vinayak N. Bapat, MD, a cardiothoracic surgeon and valve scientist at the Minneapolis Heart Institute Foundation. He posed a practical question: “Should we take from this presentation that patients should be stabilized with something like ECMO [extracorporeal membrane oxygenation] or Impella [left ventricular assist device], then transferred to an expert center for the procedure?”
“I think that the stabilization is essential in the patients with cardiogenic shock,” Dr. Estevez-Loureiro replied. “Unlike with surgery, it’s very difficult to establish a MitraClip procedure in a couple of hours in the middle of the night. You have to stabilize them and then treat for shock with ECMO, Impella, or both. I think they should be transferred to a center than can deliver the best treatment. In centers with less experience, patients can be put on mechanical support and transferred to an expert valve center, not only for MitraClip implantation, but for discussion of all the treatment possibilities, including surgery.”
At a press conference in which Dr. Estevez-Loureiro presented highlights of the IREMMI study, discussant Dee Dee Wang, MD, said the international coinvestigators “need to be applauded” for this study.
“Having these outcomes is incredible,” declared Dr. Wang, a structural heart disease specialist at the Henry Ford Health System, Detroit.
While this is an observational study, it’s a high-quality dataset with excellent methodology. And conducting a randomized trial in patients with such high surgical risk scores – the CS group had an average EuroSCORE II of 21 – would be extremely difficult, according to the cardiologist.
Dr. Estevez-Loureiro reported receiving research grants from Abbott and serving as a consultant to that company as well as Boston Scientific.
SOURCE: Estevez-Loureiro, R. TCT 2020, LBCS session IV.
FROM TCT 2020
Intravascular lithotripsy hailed as ‘game changer’ for coronary calcification
aimed at gaining U.S. regulatory approval.
The technology is basically the same as in extracorporeal lithotripsy, used for the treatment of kidney stones for more than 30 years: namely, transmission of pulsed acoustic pressure waves in order to fracture calcium. For interventional cardiology purposes, however, the transmitter is located within a balloon angioplasty catheter, Dean J. Kereiakes, MD, explained in presenting the study results at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
In Disrupt CAD III, intravascular lithotripsy far exceeded the procedural success and 30-day freedom from major adverse cardiovascular event (MACE) performance targets set in conjunction with the Food and Drug Administration. In so doing, the intravascular lithotripsy device developed by Shockwave Medical successfully addressed one of the banes of contemporary interventional cardiology: heavily calcified coronary lesions.
Currently available technologies targeting such lesions, including noncompliant high-pressure balloons, intravascular lasers, cutting balloons, and orbital and rotational atherectomy, often yield suboptimal results, noted Dr. Kereiakes, medical director of the Christ Hospital Heart and Cardiovascular Center in Cincinnati.
Severe vascular calcifications are becoming more common, due in part to an aging population and the growing prevalence of hypertension, diabetes, and renal insufficiency. Severely calcified coronary lesions complicate percutaneous coronary intervention. They’re associated with increased risks of dissection, perforation, and periprocedural MI. Moreover, heavily calcified lesions impede stent delivery and expansion – and stent underexpansion is the leading predictor of restenosis and stent thrombosis, he observed at the meeting, sponsored by the Cardiovascular Research Foundation. Disrupt CAD III was a prospective single-arm study of 384 patients at 47 sites in the United States and several European countries. All participants had de novo coronary calcifications graded as severe by core laboratory assessment, with a mean calcified length of 47.9 mm by quantitative coronary angiography and a mean calcium angle and thickness of 292.5 degrees and 0.96 mm by optical coherence tomography.
“It’s staggering, the level of calcification these patients had. It’s jaw dropping,” Dr. Kereiakes observed.
Intravascular lithotripsy was used to prepare these severely calcified lesions for stenting. The intervention entailed transmission of acoustic waves circumferentially and transmurally at 1 pulse per second through tissue at an effective pressure of about 50 atm. Patients received an average of 69 pulses.
This was not a randomized trial; there was no sham-treated control arm. Instead, the comparator group selected under regulatory guidance was comprised of patients who had received orbital atherectomy for severe coronary calcifications in the earlier, similarly designed ORBIT II trial, which led to FDA marketing approval of that technology.
Key outcomes
The procedural success rate, defined as successful stent delivery with less than a 50% residual stenosis and no in-hospital MACE, was 92.4% in Disrupt CAD III, compared to 83.4% for orbital atherectomy in ORBIT II. The primary safety endpoint of freedom from cardiac death, MI, or target vessel revascularization at 30 days was achieved in 92.2% of patients in the intravascular lithotripsy trial, versus 84.4% in ORBIT II.
The 30-day MACE rate of 7.8% in Disrupt CAD III was primarily driven by periprocedural MIs, which occurred in 6.8% of participants. Only one-third of the MIs were clinically relevant by the Society for Coronary Angiography and Intervention definition. There were two cardiac deaths and three cases of stent thrombosis, all of which were associated with known predictors of the complication. There was 1 case each of dissection, abrupt closure, and perforation, but no instances of slow flow or no reflow at the procedure’s end. Transient lithotripsy-induced left ventricular capture occurred in 41% of patients, but they were benign events with no lasting consequences.
The device was able to cross and deliver acoustic pressure wave therapy to 98.2% of lesions. The mean diameter stenosis preprocedure was 65.1%, dropping to 37.2% post lithotripsy, with a final in-stent residual stenosis diameter of 11.9%, with a 1.7-mm acute gain. The average stent expansion at the site of maximum calcification was 102%, with a minimum stent area of 6.5 mm2.
Optical coherence imaging revealed that 67% of treated lesions had circumferential and transmural fractures of both deep and superficial calcium post lithotripsy. Yet outcomes were the same regardless of whether fractures were evident on imaging.
At 30-day follow-up, 72.9% of patients had no angina, up from just 12.6% of participants pre-PCI. Follow-up will continue for 2 years.
Outcomes were similar for the first case done at each participating center and all cases thereafter.
“The ease of use was remarkable,” Dr. Kereiakes recalled. “The learning curve is virtually nonexistent.”
The reaction
At a press conference where Dr. Kereiakes presented the Disrupt CAD III results, discussant Allen Jeremias, MD, said he found the results compelling.
“The success rate is high, I think it’s relatively easy to use, as demonstrated, and I think the results are spectacular,” said Dr. Jeremias, director of interventional cardiology research and associate director of the cardiac catheterization laboratory at St. Francis Hospital in Roslyn, N.Y.
Cardiologists “really don’t do a good job most of the time” with severely calcified coronary lesions, added Dr. Jeremias, who wasn’t involved in the trial.
“A lot of times these patients have inadequate stent outcomes when we do intravascular imaging. So to do something to try to basically crack the calcium and expand the stent is, I think, critically important in these patients, and this is an amazing technology that accomplishes that,” the cardiologist said.
Juan F. Granada, MD, of Columbia University, New York, who moderated the press conference, said, “Some of the debulking techniques used for calcified stenoses actually require a lot of training, knowledge, experience, and hospital infrastructure.
I really think having a technology that is easy to use and familiar to all interventional cardiologists, such as a balloon, could potentially be a disruptive change in our field.”
“It’s an absolute game changer,” agreed Dr. Jeremias.
Dr. Kereiakes reported serving as a consultant to a handful of medical device companies, including Shockwave Medical, which sponsored Disrupt CAD III.
SOURCE: Kereiakes DJ. TCT 2020. Late Breaking Clinical Science session 2.
aimed at gaining U.S. regulatory approval.
The technology is basically the same as in extracorporeal lithotripsy, used for the treatment of kidney stones for more than 30 years: namely, transmission of pulsed acoustic pressure waves in order to fracture calcium. For interventional cardiology purposes, however, the transmitter is located within a balloon angioplasty catheter, Dean J. Kereiakes, MD, explained in presenting the study results at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
In Disrupt CAD III, intravascular lithotripsy far exceeded the procedural success and 30-day freedom from major adverse cardiovascular event (MACE) performance targets set in conjunction with the Food and Drug Administration. In so doing, the intravascular lithotripsy device developed by Shockwave Medical successfully addressed one of the banes of contemporary interventional cardiology: heavily calcified coronary lesions.
Currently available technologies targeting such lesions, including noncompliant high-pressure balloons, intravascular lasers, cutting balloons, and orbital and rotational atherectomy, often yield suboptimal results, noted Dr. Kereiakes, medical director of the Christ Hospital Heart and Cardiovascular Center in Cincinnati.
Severe vascular calcifications are becoming more common, due in part to an aging population and the growing prevalence of hypertension, diabetes, and renal insufficiency. Severely calcified coronary lesions complicate percutaneous coronary intervention. They’re associated with increased risks of dissection, perforation, and periprocedural MI. Moreover, heavily calcified lesions impede stent delivery and expansion – and stent underexpansion is the leading predictor of restenosis and stent thrombosis, he observed at the meeting, sponsored by the Cardiovascular Research Foundation. Disrupt CAD III was a prospective single-arm study of 384 patients at 47 sites in the United States and several European countries. All participants had de novo coronary calcifications graded as severe by core laboratory assessment, with a mean calcified length of 47.9 mm by quantitative coronary angiography and a mean calcium angle and thickness of 292.5 degrees and 0.96 mm by optical coherence tomography.
“It’s staggering, the level of calcification these patients had. It’s jaw dropping,” Dr. Kereiakes observed.
Intravascular lithotripsy was used to prepare these severely calcified lesions for stenting. The intervention entailed transmission of acoustic waves circumferentially and transmurally at 1 pulse per second through tissue at an effective pressure of about 50 atm. Patients received an average of 69 pulses.
This was not a randomized trial; there was no sham-treated control arm. Instead, the comparator group selected under regulatory guidance was comprised of patients who had received orbital atherectomy for severe coronary calcifications in the earlier, similarly designed ORBIT II trial, which led to FDA marketing approval of that technology.
Key outcomes
The procedural success rate, defined as successful stent delivery with less than a 50% residual stenosis and no in-hospital MACE, was 92.4% in Disrupt CAD III, compared to 83.4% for orbital atherectomy in ORBIT II. The primary safety endpoint of freedom from cardiac death, MI, or target vessel revascularization at 30 days was achieved in 92.2% of patients in the intravascular lithotripsy trial, versus 84.4% in ORBIT II.
The 30-day MACE rate of 7.8% in Disrupt CAD III was primarily driven by periprocedural MIs, which occurred in 6.8% of participants. Only one-third of the MIs were clinically relevant by the Society for Coronary Angiography and Intervention definition. There were two cardiac deaths and three cases of stent thrombosis, all of which were associated with known predictors of the complication. There was 1 case each of dissection, abrupt closure, and perforation, but no instances of slow flow or no reflow at the procedure’s end. Transient lithotripsy-induced left ventricular capture occurred in 41% of patients, but they were benign events with no lasting consequences.
The device was able to cross and deliver acoustic pressure wave therapy to 98.2% of lesions. The mean diameter stenosis preprocedure was 65.1%, dropping to 37.2% post lithotripsy, with a final in-stent residual stenosis diameter of 11.9%, with a 1.7-mm acute gain. The average stent expansion at the site of maximum calcification was 102%, with a minimum stent area of 6.5 mm2.
Optical coherence imaging revealed that 67% of treated lesions had circumferential and transmural fractures of both deep and superficial calcium post lithotripsy. Yet outcomes were the same regardless of whether fractures were evident on imaging.
At 30-day follow-up, 72.9% of patients had no angina, up from just 12.6% of participants pre-PCI. Follow-up will continue for 2 years.
Outcomes were similar for the first case done at each participating center and all cases thereafter.
“The ease of use was remarkable,” Dr. Kereiakes recalled. “The learning curve is virtually nonexistent.”
The reaction
At a press conference where Dr. Kereiakes presented the Disrupt CAD III results, discussant Allen Jeremias, MD, said he found the results compelling.
“The success rate is high, I think it’s relatively easy to use, as demonstrated, and I think the results are spectacular,” said Dr. Jeremias, director of interventional cardiology research and associate director of the cardiac catheterization laboratory at St. Francis Hospital in Roslyn, N.Y.
Cardiologists “really don’t do a good job most of the time” with severely calcified coronary lesions, added Dr. Jeremias, who wasn’t involved in the trial.
“A lot of times these patients have inadequate stent outcomes when we do intravascular imaging. So to do something to try to basically crack the calcium and expand the stent is, I think, critically important in these patients, and this is an amazing technology that accomplishes that,” the cardiologist said.
Juan F. Granada, MD, of Columbia University, New York, who moderated the press conference, said, “Some of the debulking techniques used for calcified stenoses actually require a lot of training, knowledge, experience, and hospital infrastructure.
I really think having a technology that is easy to use and familiar to all interventional cardiologists, such as a balloon, could potentially be a disruptive change in our field.”
“It’s an absolute game changer,” agreed Dr. Jeremias.
Dr. Kereiakes reported serving as a consultant to a handful of medical device companies, including Shockwave Medical, which sponsored Disrupt CAD III.
SOURCE: Kereiakes DJ. TCT 2020. Late Breaking Clinical Science session 2.
aimed at gaining U.S. regulatory approval.
The technology is basically the same as in extracorporeal lithotripsy, used for the treatment of kidney stones for more than 30 years: namely, transmission of pulsed acoustic pressure waves in order to fracture calcium. For interventional cardiology purposes, however, the transmitter is located within a balloon angioplasty catheter, Dean J. Kereiakes, MD, explained in presenting the study results at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
In Disrupt CAD III, intravascular lithotripsy far exceeded the procedural success and 30-day freedom from major adverse cardiovascular event (MACE) performance targets set in conjunction with the Food and Drug Administration. In so doing, the intravascular lithotripsy device developed by Shockwave Medical successfully addressed one of the banes of contemporary interventional cardiology: heavily calcified coronary lesions.
Currently available technologies targeting such lesions, including noncompliant high-pressure balloons, intravascular lasers, cutting balloons, and orbital and rotational atherectomy, often yield suboptimal results, noted Dr. Kereiakes, medical director of the Christ Hospital Heart and Cardiovascular Center in Cincinnati.
Severe vascular calcifications are becoming more common, due in part to an aging population and the growing prevalence of hypertension, diabetes, and renal insufficiency. Severely calcified coronary lesions complicate percutaneous coronary intervention. They’re associated with increased risks of dissection, perforation, and periprocedural MI. Moreover, heavily calcified lesions impede stent delivery and expansion – and stent underexpansion is the leading predictor of restenosis and stent thrombosis, he observed at the meeting, sponsored by the Cardiovascular Research Foundation. Disrupt CAD III was a prospective single-arm study of 384 patients at 47 sites in the United States and several European countries. All participants had de novo coronary calcifications graded as severe by core laboratory assessment, with a mean calcified length of 47.9 mm by quantitative coronary angiography and a mean calcium angle and thickness of 292.5 degrees and 0.96 mm by optical coherence tomography.
“It’s staggering, the level of calcification these patients had. It’s jaw dropping,” Dr. Kereiakes observed.
Intravascular lithotripsy was used to prepare these severely calcified lesions for stenting. The intervention entailed transmission of acoustic waves circumferentially and transmurally at 1 pulse per second through tissue at an effective pressure of about 50 atm. Patients received an average of 69 pulses.
This was not a randomized trial; there was no sham-treated control arm. Instead, the comparator group selected under regulatory guidance was comprised of patients who had received orbital atherectomy for severe coronary calcifications in the earlier, similarly designed ORBIT II trial, which led to FDA marketing approval of that technology.
Key outcomes
The procedural success rate, defined as successful stent delivery with less than a 50% residual stenosis and no in-hospital MACE, was 92.4% in Disrupt CAD III, compared to 83.4% for orbital atherectomy in ORBIT II. The primary safety endpoint of freedom from cardiac death, MI, or target vessel revascularization at 30 days was achieved in 92.2% of patients in the intravascular lithotripsy trial, versus 84.4% in ORBIT II.
The 30-day MACE rate of 7.8% in Disrupt CAD III was primarily driven by periprocedural MIs, which occurred in 6.8% of participants. Only one-third of the MIs were clinically relevant by the Society for Coronary Angiography and Intervention definition. There were two cardiac deaths and three cases of stent thrombosis, all of which were associated with known predictors of the complication. There was 1 case each of dissection, abrupt closure, and perforation, but no instances of slow flow or no reflow at the procedure’s end. Transient lithotripsy-induced left ventricular capture occurred in 41% of patients, but they were benign events with no lasting consequences.
The device was able to cross and deliver acoustic pressure wave therapy to 98.2% of lesions. The mean diameter stenosis preprocedure was 65.1%, dropping to 37.2% post lithotripsy, with a final in-stent residual stenosis diameter of 11.9%, with a 1.7-mm acute gain. The average stent expansion at the site of maximum calcification was 102%, with a minimum stent area of 6.5 mm2.
Optical coherence imaging revealed that 67% of treated lesions had circumferential and transmural fractures of both deep and superficial calcium post lithotripsy. Yet outcomes were the same regardless of whether fractures were evident on imaging.
At 30-day follow-up, 72.9% of patients had no angina, up from just 12.6% of participants pre-PCI. Follow-up will continue for 2 years.
Outcomes were similar for the first case done at each participating center and all cases thereafter.
“The ease of use was remarkable,” Dr. Kereiakes recalled. “The learning curve is virtually nonexistent.”
The reaction
At a press conference where Dr. Kereiakes presented the Disrupt CAD III results, discussant Allen Jeremias, MD, said he found the results compelling.
“The success rate is high, I think it’s relatively easy to use, as demonstrated, and I think the results are spectacular,” said Dr. Jeremias, director of interventional cardiology research and associate director of the cardiac catheterization laboratory at St. Francis Hospital in Roslyn, N.Y.
Cardiologists “really don’t do a good job most of the time” with severely calcified coronary lesions, added Dr. Jeremias, who wasn’t involved in the trial.
“A lot of times these patients have inadequate stent outcomes when we do intravascular imaging. So to do something to try to basically crack the calcium and expand the stent is, I think, critically important in these patients, and this is an amazing technology that accomplishes that,” the cardiologist said.
Juan F. Granada, MD, of Columbia University, New York, who moderated the press conference, said, “Some of the debulking techniques used for calcified stenoses actually require a lot of training, knowledge, experience, and hospital infrastructure.
I really think having a technology that is easy to use and familiar to all interventional cardiologists, such as a balloon, could potentially be a disruptive change in our field.”
“It’s an absolute game changer,” agreed Dr. Jeremias.
Dr. Kereiakes reported serving as a consultant to a handful of medical device companies, including Shockwave Medical, which sponsored Disrupt CAD III.
SOURCE: Kereiakes DJ. TCT 2020. Late Breaking Clinical Science session 2.
FROM TCT 2020
Key clinical point: Intravascular lithotripsy was safe and effective for treatment of severely calcified coronary stenoses in a pivotal trial.
Major finding: The 30-day rate of freedom from major adverse cardiovascular events was 92.2%, well above the prespecified performance goal of 84.4%.
Study details: Disrupt CAD III study is a multicenter, single-arm, prospective study of intravascular lithotripsy in 384 patients with severe coronary calcification.
Disclosures: The presenter reported serving as a consultant to Shockwave Medical Inc., the study sponsor, as well as several other medical device companies.
Source: Kereiakes DJ. TCT 2020. Late Breaking Clinical Science session 2.
NACMI: Clear benefit with PCI in STEMI COVID-19 patients
Patients with COVID-19 who present with ST-segment elevation MI (STEMI) represent a unique, high-risk population with greater risks for in-hospital death and stroke, according to initial results from the North American COVID-19 ST-Segment Elevation Myocardial Infarction Registry (NACMI).
Although COVID-19–confirmed patients were less likely to undergo angiography than patients under investigation (PUI) for COVID-19 or historical STEMI activation controls, 71% underwent primary percutaneous coronary intervention (PCI).
“Primary PCI is preferable and feasible in COVID-19–positive patients, with door-to-balloon times similar to PUI or COVID-negative patients, and that supports the updated COVID-specific STEMI guidelines,” study cochair Timothy D. Henry, MD, said in a late-breaking clinical science session at TCT 2020, the Transcatheter Cardiovascular Therapeutics virtual annual meeting.
The multisociety COVID-specific guidelines were initially issued in April, endorsing PCI as the standard of care and allowing for consideration of fibrinolysis-based therapy at non-PCI capable hospitals.
Five previous publications on a total of 174 COVID-19 patients with ST-elevation have shown there are more frequent in-hospital STEMI presentations, more cases without a clear culprit lesion, more thrombotic lesions and microthrombi, and higher mortality, ranging from 12% to 72%. Still, there has been considerable controversy over exactly what to do when COVID-19 patients with ST elevation reach the cath lab, he said at the meeting sponsored by the Cardiovascular Research Foundation.
NACMI represents the largest experience with ST-elevation patients and is a unique collaboration between the Society for Cardiovascular Angiography and Interventions, Canadian Association of Interventional Cardiology, American College of Cardiology, and Midwest STEMI Consortium, noted Dr. Henry, who is medical director of the Lindner Center for Research and Education at the Christ Hospital, Cincinnati.
The registry enrolled any COVID-19–positive patient or person under investigation older than 18 years with ST-segment elevation or new-onset left bundle branch block on electrocardiogram with a clinical correlate of myocardial ischemia such as chest pain, dyspnea, cardiac arrest, shock, or mechanical ventilation. There were no exclusion criteria.
Data from 171 patients with confirmed COVID-19 and 423 PUI from 64 sites were then propensity-matched to a control population from the Midwest STEMI Consortium, a prospective, multicenter registry of consecutive STEMI patients.
The three groups were similar in sex and age but there was a striking difference in race, with 27% of African American and 24% of Hispanic patients COVID-confirmed, compared with 11% and 6% in the PUI group and 4% and 1% in the control group. Likewise, there was a significant increase in diabetes (44% vs. 33% vs. 20%), which has been reported previously with influenza.
COVID-19–positive patients, as compared with PUI and controls, were significantly more likely to present with cardiogenic shock before PCI (20% vs. 14% vs. 5%), but not cardiac arrest (12% vs. 17% vs. 11%), and to have lower left ventricular ejection fractions (45% vs. 45% vs. 50%).
They also presented with more atypical symptoms than PUI patients, particularly infiltrates on chest x-ray (49% vs. 17%) and dyspnea (58% vs. 38%). Data were not available for these outcomes among historic controls.
Importantly, 21% of the COVID-19 patients did not undergo angiography, compared with 5% of PUI patients and 0% of controls (P < .001), “which is much higher than we would expect or have suspected,” Dr. Henry said. Thrombolytic use was very uncommon in those undergoing angiography, likely as a result of the guidelines.
Very surprisingly, there were no differences in door-to-balloon times between the COVID-positive, PUI, and control groups despite the ongoing pandemic (80 min vs. 78 min vs. 86 min).
But there was clear worsening in in-hospital mortality in COVID-19–positive patients (32% vs. 12% and 6%; P < .001), as well as in-hospital stroke (3.4% vs. 2% vs. 0.6%) that reached statistical significance only when compared with historical controls (P = .039). Total length of stay was twice as long in COVID-confirmed patients as in both PUI and controls (6 days vs. 3 days; P < .001).
Following the formal presentation, invited discussant Philippe Gabriel Steg, MD, Imperial College London, said the researchers have provided a great service in reporting the data so quickly but noted that an ongoing French registry of events before, during, and after the first COVID-19 wave has not seen an increased death rate.
“Can you tease out whether the increased death rate is related to cardiovascular deaths or to COVID-related pneumonias, shocks, ARDSs [acute respiratory distress syndromes], and so on and so forth? Because our impression – and that’s what we’ve published in Lancet Public Health – is that the cardiovascular morality rate doesn’t seem that affected by COVID.”
Dr. Henry replied that these are early data but “I will tell you that patients who did get PCI had a mortality rate that was only around 12% or 13%, and the patients who did not undergo angiography or were treated with medical therapy had higher mortality. Now, of course, that’s selected and we need to do a much better matching and look at that, but that’s our goal and we will have that information,” he said.
During a press briefing on the study, discussant Renu Virmani, MD, president and founder of CVPath Institute, noted that, in their analysis of 40 autopsy cases from Bergamot, Italy, small intramyocardial microthrombi were seen in nine patients, whereas epicardial microthrombi were seen in only three or four.
“Some of the cases are being taken as being related to coronary disease but may be more thrombotic than anything else,” she said. “I think there’s a combination, and that’s why the outcomes are so poor. You didn’t show us TIMI flow but that’s something to think about: Was TIMI flow different in the patients who died because you have very high mortality? I think we need to get to the bottom of what is the underlying cause of that thrombosis.”
Future topics of interest include ethnic and regional/country differences; time-to-treatment including chest pain onset-to-arrival; transfer, in-hospital, and no-culprit patients; changes over time during the pandemic; and eventually 1-year outcomes, Dr. Henry said.
Press briefing moderator Ajay Kirtane, MD, director of the cardiac catheterization labs at NewYork-Presbyterian/Columbia University Irving, New York, remarked that “a lot of times people will pooh-pooh observational data, but this is exactly the type of data that we need to try to be able to gather information about what our practices are, how they fit. And I think many of us around the world will see these data, and it will echo their own experience.”
The study was funded by the Society for Cardiovascular Angiography and Interventions and the Canadian Association of Interventional Cardiology. Dr. Henry has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Patients with COVID-19 who present with ST-segment elevation MI (STEMI) represent a unique, high-risk population with greater risks for in-hospital death and stroke, according to initial results from the North American COVID-19 ST-Segment Elevation Myocardial Infarction Registry (NACMI).
Although COVID-19–confirmed patients were less likely to undergo angiography than patients under investigation (PUI) for COVID-19 or historical STEMI activation controls, 71% underwent primary percutaneous coronary intervention (PCI).
“Primary PCI is preferable and feasible in COVID-19–positive patients, with door-to-balloon times similar to PUI or COVID-negative patients, and that supports the updated COVID-specific STEMI guidelines,” study cochair Timothy D. Henry, MD, said in a late-breaking clinical science session at TCT 2020, the Transcatheter Cardiovascular Therapeutics virtual annual meeting.
The multisociety COVID-specific guidelines were initially issued in April, endorsing PCI as the standard of care and allowing for consideration of fibrinolysis-based therapy at non-PCI capable hospitals.
Five previous publications on a total of 174 COVID-19 patients with ST-elevation have shown there are more frequent in-hospital STEMI presentations, more cases without a clear culprit lesion, more thrombotic lesions and microthrombi, and higher mortality, ranging from 12% to 72%. Still, there has been considerable controversy over exactly what to do when COVID-19 patients with ST elevation reach the cath lab, he said at the meeting sponsored by the Cardiovascular Research Foundation.
NACMI represents the largest experience with ST-elevation patients and is a unique collaboration between the Society for Cardiovascular Angiography and Interventions, Canadian Association of Interventional Cardiology, American College of Cardiology, and Midwest STEMI Consortium, noted Dr. Henry, who is medical director of the Lindner Center for Research and Education at the Christ Hospital, Cincinnati.
The registry enrolled any COVID-19–positive patient or person under investigation older than 18 years with ST-segment elevation or new-onset left bundle branch block on electrocardiogram with a clinical correlate of myocardial ischemia such as chest pain, dyspnea, cardiac arrest, shock, or mechanical ventilation. There were no exclusion criteria.
Data from 171 patients with confirmed COVID-19 and 423 PUI from 64 sites were then propensity-matched to a control population from the Midwest STEMI Consortium, a prospective, multicenter registry of consecutive STEMI patients.
The three groups were similar in sex and age but there was a striking difference in race, with 27% of African American and 24% of Hispanic patients COVID-confirmed, compared with 11% and 6% in the PUI group and 4% and 1% in the control group. Likewise, there was a significant increase in diabetes (44% vs. 33% vs. 20%), which has been reported previously with influenza.
COVID-19–positive patients, as compared with PUI and controls, were significantly more likely to present with cardiogenic shock before PCI (20% vs. 14% vs. 5%), but not cardiac arrest (12% vs. 17% vs. 11%), and to have lower left ventricular ejection fractions (45% vs. 45% vs. 50%).
They also presented with more atypical symptoms than PUI patients, particularly infiltrates on chest x-ray (49% vs. 17%) and dyspnea (58% vs. 38%). Data were not available for these outcomes among historic controls.
Importantly, 21% of the COVID-19 patients did not undergo angiography, compared with 5% of PUI patients and 0% of controls (P < .001), “which is much higher than we would expect or have suspected,” Dr. Henry said. Thrombolytic use was very uncommon in those undergoing angiography, likely as a result of the guidelines.
Very surprisingly, there were no differences in door-to-balloon times between the COVID-positive, PUI, and control groups despite the ongoing pandemic (80 min vs. 78 min vs. 86 min).
But there was clear worsening in in-hospital mortality in COVID-19–positive patients (32% vs. 12% and 6%; P < .001), as well as in-hospital stroke (3.4% vs. 2% vs. 0.6%) that reached statistical significance only when compared with historical controls (P = .039). Total length of stay was twice as long in COVID-confirmed patients as in both PUI and controls (6 days vs. 3 days; P < .001).
Following the formal presentation, invited discussant Philippe Gabriel Steg, MD, Imperial College London, said the researchers have provided a great service in reporting the data so quickly but noted that an ongoing French registry of events before, during, and after the first COVID-19 wave has not seen an increased death rate.
“Can you tease out whether the increased death rate is related to cardiovascular deaths or to COVID-related pneumonias, shocks, ARDSs [acute respiratory distress syndromes], and so on and so forth? Because our impression – and that’s what we’ve published in Lancet Public Health – is that the cardiovascular morality rate doesn’t seem that affected by COVID.”
Dr. Henry replied that these are early data but “I will tell you that patients who did get PCI had a mortality rate that was only around 12% or 13%, and the patients who did not undergo angiography or were treated with medical therapy had higher mortality. Now, of course, that’s selected and we need to do a much better matching and look at that, but that’s our goal and we will have that information,” he said.
During a press briefing on the study, discussant Renu Virmani, MD, president and founder of CVPath Institute, noted that, in their analysis of 40 autopsy cases from Bergamot, Italy, small intramyocardial microthrombi were seen in nine patients, whereas epicardial microthrombi were seen in only three or four.
“Some of the cases are being taken as being related to coronary disease but may be more thrombotic than anything else,” she said. “I think there’s a combination, and that’s why the outcomes are so poor. You didn’t show us TIMI flow but that’s something to think about: Was TIMI flow different in the patients who died because you have very high mortality? I think we need to get to the bottom of what is the underlying cause of that thrombosis.”
Future topics of interest include ethnic and regional/country differences; time-to-treatment including chest pain onset-to-arrival; transfer, in-hospital, and no-culprit patients; changes over time during the pandemic; and eventually 1-year outcomes, Dr. Henry said.
Press briefing moderator Ajay Kirtane, MD, director of the cardiac catheterization labs at NewYork-Presbyterian/Columbia University Irving, New York, remarked that “a lot of times people will pooh-pooh observational data, but this is exactly the type of data that we need to try to be able to gather information about what our practices are, how they fit. And I think many of us around the world will see these data, and it will echo their own experience.”
The study was funded by the Society for Cardiovascular Angiography and Interventions and the Canadian Association of Interventional Cardiology. Dr. Henry has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Patients with COVID-19 who present with ST-segment elevation MI (STEMI) represent a unique, high-risk population with greater risks for in-hospital death and stroke, according to initial results from the North American COVID-19 ST-Segment Elevation Myocardial Infarction Registry (NACMI).
Although COVID-19–confirmed patients were less likely to undergo angiography than patients under investigation (PUI) for COVID-19 or historical STEMI activation controls, 71% underwent primary percutaneous coronary intervention (PCI).
“Primary PCI is preferable and feasible in COVID-19–positive patients, with door-to-balloon times similar to PUI or COVID-negative patients, and that supports the updated COVID-specific STEMI guidelines,” study cochair Timothy D. Henry, MD, said in a late-breaking clinical science session at TCT 2020, the Transcatheter Cardiovascular Therapeutics virtual annual meeting.
The multisociety COVID-specific guidelines were initially issued in April, endorsing PCI as the standard of care and allowing for consideration of fibrinolysis-based therapy at non-PCI capable hospitals.
Five previous publications on a total of 174 COVID-19 patients with ST-elevation have shown there are more frequent in-hospital STEMI presentations, more cases without a clear culprit lesion, more thrombotic lesions and microthrombi, and higher mortality, ranging from 12% to 72%. Still, there has been considerable controversy over exactly what to do when COVID-19 patients with ST elevation reach the cath lab, he said at the meeting sponsored by the Cardiovascular Research Foundation.
NACMI represents the largest experience with ST-elevation patients and is a unique collaboration between the Society for Cardiovascular Angiography and Interventions, Canadian Association of Interventional Cardiology, American College of Cardiology, and Midwest STEMI Consortium, noted Dr. Henry, who is medical director of the Lindner Center for Research and Education at the Christ Hospital, Cincinnati.
The registry enrolled any COVID-19–positive patient or person under investigation older than 18 years with ST-segment elevation or new-onset left bundle branch block on electrocardiogram with a clinical correlate of myocardial ischemia such as chest pain, dyspnea, cardiac arrest, shock, or mechanical ventilation. There were no exclusion criteria.
Data from 171 patients with confirmed COVID-19 and 423 PUI from 64 sites were then propensity-matched to a control population from the Midwest STEMI Consortium, a prospective, multicenter registry of consecutive STEMI patients.
The three groups were similar in sex and age but there was a striking difference in race, with 27% of African American and 24% of Hispanic patients COVID-confirmed, compared with 11% and 6% in the PUI group and 4% and 1% in the control group. Likewise, there was a significant increase in diabetes (44% vs. 33% vs. 20%), which has been reported previously with influenza.
COVID-19–positive patients, as compared with PUI and controls, were significantly more likely to present with cardiogenic shock before PCI (20% vs. 14% vs. 5%), but not cardiac arrest (12% vs. 17% vs. 11%), and to have lower left ventricular ejection fractions (45% vs. 45% vs. 50%).
They also presented with more atypical symptoms than PUI patients, particularly infiltrates on chest x-ray (49% vs. 17%) and dyspnea (58% vs. 38%). Data were not available for these outcomes among historic controls.
Importantly, 21% of the COVID-19 patients did not undergo angiography, compared with 5% of PUI patients and 0% of controls (P < .001), “which is much higher than we would expect or have suspected,” Dr. Henry said. Thrombolytic use was very uncommon in those undergoing angiography, likely as a result of the guidelines.
Very surprisingly, there were no differences in door-to-balloon times between the COVID-positive, PUI, and control groups despite the ongoing pandemic (80 min vs. 78 min vs. 86 min).
But there was clear worsening in in-hospital mortality in COVID-19–positive patients (32% vs. 12% and 6%; P < .001), as well as in-hospital stroke (3.4% vs. 2% vs. 0.6%) that reached statistical significance only when compared with historical controls (P = .039). Total length of stay was twice as long in COVID-confirmed patients as in both PUI and controls (6 days vs. 3 days; P < .001).
Following the formal presentation, invited discussant Philippe Gabriel Steg, MD, Imperial College London, said the researchers have provided a great service in reporting the data so quickly but noted that an ongoing French registry of events before, during, and after the first COVID-19 wave has not seen an increased death rate.
“Can you tease out whether the increased death rate is related to cardiovascular deaths or to COVID-related pneumonias, shocks, ARDSs [acute respiratory distress syndromes], and so on and so forth? Because our impression – and that’s what we’ve published in Lancet Public Health – is that the cardiovascular morality rate doesn’t seem that affected by COVID.”
Dr. Henry replied that these are early data but “I will tell you that patients who did get PCI had a mortality rate that was only around 12% or 13%, and the patients who did not undergo angiography or were treated with medical therapy had higher mortality. Now, of course, that’s selected and we need to do a much better matching and look at that, but that’s our goal and we will have that information,” he said.
During a press briefing on the study, discussant Renu Virmani, MD, president and founder of CVPath Institute, noted that, in their analysis of 40 autopsy cases from Bergamot, Italy, small intramyocardial microthrombi were seen in nine patients, whereas epicardial microthrombi were seen in only three or four.
“Some of the cases are being taken as being related to coronary disease but may be more thrombotic than anything else,” she said. “I think there’s a combination, and that’s why the outcomes are so poor. You didn’t show us TIMI flow but that’s something to think about: Was TIMI flow different in the patients who died because you have very high mortality? I think we need to get to the bottom of what is the underlying cause of that thrombosis.”
Future topics of interest include ethnic and regional/country differences; time-to-treatment including chest pain onset-to-arrival; transfer, in-hospital, and no-culprit patients; changes over time during the pandemic; and eventually 1-year outcomes, Dr. Henry said.
Press briefing moderator Ajay Kirtane, MD, director of the cardiac catheterization labs at NewYork-Presbyterian/Columbia University Irving, New York, remarked that “a lot of times people will pooh-pooh observational data, but this is exactly the type of data that we need to try to be able to gather information about what our practices are, how they fit. And I think many of us around the world will see these data, and it will echo their own experience.”
The study was funded by the Society for Cardiovascular Angiography and Interventions and the Canadian Association of Interventional Cardiology. Dr. Henry has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Entresto halves renal events in preserved EF heart failure patients
Patients with heart failure with preserved ejection fraction (HFpEF) who received sacubitril/valsartan in the PARAGON-HF trial had significant protection against progression of renal dysfunction in a prespecified secondary analysis.
The 2,419 patients with HFpEF who received sacubitril/valsartan (Entresto) had half the rate of the primary adverse renal outcome, compared with the 2,403 patients randomized to valsartan alone in the comparator group, a significant difference, according to the results published online Sept. 29 in Circulation by Finnian R. McCausland, MBBCh, and colleagues.
In absolute terms, sacubitril/valsartan treatment, an angiotensin-receptor/neprilysin inhibitor (ARNI), cut the incidence of the combined renal endpoint – renal death, end-stage renal disease, or at least a 50% drop in estimated glomerular filtration rate (eGFR) – from 2.7% in the control group to 1.4% in the sacubitril/valsartan group during a median follow-up of 35 months.
The absolute difference of 1.3% equated to a number needed to treat of 51 to prevent one of these events.
Also notable was that renal protection from sacubitril/valsartan was equally robust across the range of baseline kidney function.
‘An important therapeutic option’
The efficacy “across the spectrum of baseline renal function” indicates treatment with sacubitril/valsartan is “an important therapeutic option to slow renal-function decline in patients with heart failure,” wrote Dr. McCausland, a nephrologist at Brigham and Women’s Hospital in Boston, and colleagues.
The authors’ conclusion is striking because currently no drug class has produced clear evidence for efficacy in HFpEF.
On the other hand, the PARAGON-HF trial that provided the data for this new analysis was statistically neutral for its primary endpoint – a reduction in the combined rate of cardiovascular death and hospitalizations for heart failure – with a P value of .06 and 95% confidence interval of 0.75-1.01.
“Because this difference [in the primary endpoint incidence between the two study group] did not meet the predetermined level of statistical significance, subsequent analyses were considered to be exploratory,” noted the authors of the primary analysis of PARAGON-HF, as reported by Medscape Medical News.
Despite this limitation in interpreting secondary outcomes from the trial, the new report of a significant renal benefit “opens the potential to provide evidence-based treatment for patients with HFpEF,” commented Sheldon W. Tobe, MD, and Stephanie Poon, MD, in an editorial accompanying the latest analysis.
“At the very least, these results are certainly intriguing and suggest that there may be important patient subgroups with HFpEF who might benefit from using sacubitril/valsartan,” they emphasized.
First large trial to show renal improvement in HFpEF
The editorialists’ enthusiasm for the implications of the new findings relate in part to the fact that “PARAGON-HF is the first large trial to demonstrate improvement in renal parameters in HFpEF,” they noted.
“The finding that the composite renal outcome did not differ according to baseline eGFR is significant and suggests that the beneficial effect on renal function was indirect, possibly linked to improved cardiac function,” say Dr. Tobe, a nephrologist, and Dr. Poon, a cardiologist, both at Sunnybrook Health Sciences Centre in Toronto.
PARAGON-HF enrolled 4,822 HFpEF patients at 848 centers in 43 countries, and the efficacy analysis included 4,796 patients.
The composite renal outcome was mainly driven by the incidence of a 50% or greater drop from baseline in eGFR, which occurred in 27 patients (1.1%) in the sacubitril/valsartan group and 60 patients (2.5%) who received valsartan alone.
The annual average drop in eGFR during the study was 2.0 mL/min per 1.73m2 in the sacubitril/valsartan group and 2.7 mL/min per 1.73m2 in the control group.
Although the heart failure community was disappointed that sacubitril/valsartan failed to show a significant benefit for the study’s primary outcome in HFpEF, the combination has become a mainstay of treatment for patients with HFpEF based on its performance in the PARADIGM-HF trial.
And despite the unqualified support sacubitril/valsartan now receives in guidelines and its label as a foundational treatment for HFpEF, the formulation has had a hard time gaining traction in U.S. practice, often because of barriers placed by third-party payers.
PARAGON-HF was sponsored by Novartis, which markets sacubitril/valsartan (Entresto). Dr. McCausland has reported no relevant financial relationships. Dr. Tobe has reported participating on a steering committee for Bayer Fidelio/Figaro studies and being a speaker on behalf of Pfizer and Servier. Dr. Poon has reported being an adviser to Novartis, Boehringer Ingelheim, and Servier.
A version of this article originally appeared on Medscape.com.
Patients with heart failure with preserved ejection fraction (HFpEF) who received sacubitril/valsartan in the PARAGON-HF trial had significant protection against progression of renal dysfunction in a prespecified secondary analysis.
The 2,419 patients with HFpEF who received sacubitril/valsartan (Entresto) had half the rate of the primary adverse renal outcome, compared with the 2,403 patients randomized to valsartan alone in the comparator group, a significant difference, according to the results published online Sept. 29 in Circulation by Finnian R. McCausland, MBBCh, and colleagues.
In absolute terms, sacubitril/valsartan treatment, an angiotensin-receptor/neprilysin inhibitor (ARNI), cut the incidence of the combined renal endpoint – renal death, end-stage renal disease, or at least a 50% drop in estimated glomerular filtration rate (eGFR) – from 2.7% in the control group to 1.4% in the sacubitril/valsartan group during a median follow-up of 35 months.
The absolute difference of 1.3% equated to a number needed to treat of 51 to prevent one of these events.
Also notable was that renal protection from sacubitril/valsartan was equally robust across the range of baseline kidney function.
‘An important therapeutic option’
The efficacy “across the spectrum of baseline renal function” indicates treatment with sacubitril/valsartan is “an important therapeutic option to slow renal-function decline in patients with heart failure,” wrote Dr. McCausland, a nephrologist at Brigham and Women’s Hospital in Boston, and colleagues.
The authors’ conclusion is striking because currently no drug class has produced clear evidence for efficacy in HFpEF.
On the other hand, the PARAGON-HF trial that provided the data for this new analysis was statistically neutral for its primary endpoint – a reduction in the combined rate of cardiovascular death and hospitalizations for heart failure – with a P value of .06 and 95% confidence interval of 0.75-1.01.
“Because this difference [in the primary endpoint incidence between the two study group] did not meet the predetermined level of statistical significance, subsequent analyses were considered to be exploratory,” noted the authors of the primary analysis of PARAGON-HF, as reported by Medscape Medical News.
Despite this limitation in interpreting secondary outcomes from the trial, the new report of a significant renal benefit “opens the potential to provide evidence-based treatment for patients with HFpEF,” commented Sheldon W. Tobe, MD, and Stephanie Poon, MD, in an editorial accompanying the latest analysis.
“At the very least, these results are certainly intriguing and suggest that there may be important patient subgroups with HFpEF who might benefit from using sacubitril/valsartan,” they emphasized.
First large trial to show renal improvement in HFpEF
The editorialists’ enthusiasm for the implications of the new findings relate in part to the fact that “PARAGON-HF is the first large trial to demonstrate improvement in renal parameters in HFpEF,” they noted.
“The finding that the composite renal outcome did not differ according to baseline eGFR is significant and suggests that the beneficial effect on renal function was indirect, possibly linked to improved cardiac function,” say Dr. Tobe, a nephrologist, and Dr. Poon, a cardiologist, both at Sunnybrook Health Sciences Centre in Toronto.
PARAGON-HF enrolled 4,822 HFpEF patients at 848 centers in 43 countries, and the efficacy analysis included 4,796 patients.
The composite renal outcome was mainly driven by the incidence of a 50% or greater drop from baseline in eGFR, which occurred in 27 patients (1.1%) in the sacubitril/valsartan group and 60 patients (2.5%) who received valsartan alone.
The annual average drop in eGFR during the study was 2.0 mL/min per 1.73m2 in the sacubitril/valsartan group and 2.7 mL/min per 1.73m2 in the control group.
Although the heart failure community was disappointed that sacubitril/valsartan failed to show a significant benefit for the study’s primary outcome in HFpEF, the combination has become a mainstay of treatment for patients with HFpEF based on its performance in the PARADIGM-HF trial.
And despite the unqualified support sacubitril/valsartan now receives in guidelines and its label as a foundational treatment for HFpEF, the formulation has had a hard time gaining traction in U.S. practice, often because of barriers placed by third-party payers.
PARAGON-HF was sponsored by Novartis, which markets sacubitril/valsartan (Entresto). Dr. McCausland has reported no relevant financial relationships. Dr. Tobe has reported participating on a steering committee for Bayer Fidelio/Figaro studies and being a speaker on behalf of Pfizer and Servier. Dr. Poon has reported being an adviser to Novartis, Boehringer Ingelheim, and Servier.
A version of this article originally appeared on Medscape.com.
Patients with heart failure with preserved ejection fraction (HFpEF) who received sacubitril/valsartan in the PARAGON-HF trial had significant protection against progression of renal dysfunction in a prespecified secondary analysis.
The 2,419 patients with HFpEF who received sacubitril/valsartan (Entresto) had half the rate of the primary adverse renal outcome, compared with the 2,403 patients randomized to valsartan alone in the comparator group, a significant difference, according to the results published online Sept. 29 in Circulation by Finnian R. McCausland, MBBCh, and colleagues.
In absolute terms, sacubitril/valsartan treatment, an angiotensin-receptor/neprilysin inhibitor (ARNI), cut the incidence of the combined renal endpoint – renal death, end-stage renal disease, or at least a 50% drop in estimated glomerular filtration rate (eGFR) – from 2.7% in the control group to 1.4% in the sacubitril/valsartan group during a median follow-up of 35 months.
The absolute difference of 1.3% equated to a number needed to treat of 51 to prevent one of these events.
Also notable was that renal protection from sacubitril/valsartan was equally robust across the range of baseline kidney function.
‘An important therapeutic option’
The efficacy “across the spectrum of baseline renal function” indicates treatment with sacubitril/valsartan is “an important therapeutic option to slow renal-function decline in patients with heart failure,” wrote Dr. McCausland, a nephrologist at Brigham and Women’s Hospital in Boston, and colleagues.
The authors’ conclusion is striking because currently no drug class has produced clear evidence for efficacy in HFpEF.
On the other hand, the PARAGON-HF trial that provided the data for this new analysis was statistically neutral for its primary endpoint – a reduction in the combined rate of cardiovascular death and hospitalizations for heart failure – with a P value of .06 and 95% confidence interval of 0.75-1.01.
“Because this difference [in the primary endpoint incidence between the two study group] did not meet the predetermined level of statistical significance, subsequent analyses were considered to be exploratory,” noted the authors of the primary analysis of PARAGON-HF, as reported by Medscape Medical News.
Despite this limitation in interpreting secondary outcomes from the trial, the new report of a significant renal benefit “opens the potential to provide evidence-based treatment for patients with HFpEF,” commented Sheldon W. Tobe, MD, and Stephanie Poon, MD, in an editorial accompanying the latest analysis.
“At the very least, these results are certainly intriguing and suggest that there may be important patient subgroups with HFpEF who might benefit from using sacubitril/valsartan,” they emphasized.
First large trial to show renal improvement in HFpEF
The editorialists’ enthusiasm for the implications of the new findings relate in part to the fact that “PARAGON-HF is the first large trial to demonstrate improvement in renal parameters in HFpEF,” they noted.
“The finding that the composite renal outcome did not differ according to baseline eGFR is significant and suggests that the beneficial effect on renal function was indirect, possibly linked to improved cardiac function,” say Dr. Tobe, a nephrologist, and Dr. Poon, a cardiologist, both at Sunnybrook Health Sciences Centre in Toronto.
PARAGON-HF enrolled 4,822 HFpEF patients at 848 centers in 43 countries, and the efficacy analysis included 4,796 patients.
The composite renal outcome was mainly driven by the incidence of a 50% or greater drop from baseline in eGFR, which occurred in 27 patients (1.1%) in the sacubitril/valsartan group and 60 patients (2.5%) who received valsartan alone.
The annual average drop in eGFR during the study was 2.0 mL/min per 1.73m2 in the sacubitril/valsartan group and 2.7 mL/min per 1.73m2 in the control group.
Although the heart failure community was disappointed that sacubitril/valsartan failed to show a significant benefit for the study’s primary outcome in HFpEF, the combination has become a mainstay of treatment for patients with HFpEF based on its performance in the PARADIGM-HF trial.
And despite the unqualified support sacubitril/valsartan now receives in guidelines and its label as a foundational treatment for HFpEF, the formulation has had a hard time gaining traction in U.S. practice, often because of barriers placed by third-party payers.
PARAGON-HF was sponsored by Novartis, which markets sacubitril/valsartan (Entresto). Dr. McCausland has reported no relevant financial relationships. Dr. Tobe has reported participating on a steering committee for Bayer Fidelio/Figaro studies and being a speaker on behalf of Pfizer and Servier. Dr. Poon has reported being an adviser to Novartis, Boehringer Ingelheim, and Servier.
A version of this article originally appeared on Medscape.com.
Ticagrelor monotherapy beats DAPT in STEMI
, a major randomized trial.
“This is the first report assessing the feasibility of ticagrelor monotherapy after short-term DAPT for STEMI patients with drug-eluting stents,” Byeong-Keuk Kim, MD, PhD, noted at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
The positive results were consistent with the earlier TWILIGHT study (Ticagrelor with Aspirin or Alone in High-Risk Patients after Coronary Intervention), which also showed clinical benefit at 1 year for 3 months of DAPT followed by ticagrelor (Brilinta) monotherapy, albeit only in PCI patients without an acute coronary syndrome (ACS) or with non-STEMI ACS (N Engl J Med. 2019 Nov 21;381[21]:2032-42).
TICO-STEMI was a prespecified substudy involving the 1,103 STEMI patients included in the previously reported parent 38-center South Korean TICO (Ticagrelor With or Without Aspirin in Acute Coronary Syndrome After PCI) study of 3,056 ACS patients treated with a second-generation ultrathin biodegradable polymer-coated sirolimus-eluting stent (JAMA. 2020 Jun 16;323[23]:2407-16).
The primary outcome in TICO-STEMI was the 12-month composite rate of net adverse clinical events, composed of major bleeding, all-cause mortality, acute MI, stroke, stent thrombosis, or target vessel revascularization. In an intention-to-treat analysis, the rate was 5.0% in the 12-month DAPT group and 3.7% with ticagrelor monotherapy after 3 months of DAPT, for a 27% relative risk reduction which didn’t achieve statistical significance. However, in an as-treated analysis, the between-group difference in the primary endpoint was stronger: a 5.2% incidence with 12 months of DAPT and 2.3% with ticagrelor monotherapy, for a relative risk reduction of 56%, which was statistically significant.
Major bleeding, one of two key secondary endpoints, was a different story: The incidence within 12 months by intention-to-treat was 2.9% with 12 months of DAPT compared to 0.9% with ticagrelor monotherapy, for a statistically significant 68% relative risk reduction in favor of ticagrelor monotherapy. In contrast, there was no between-group difference in the other secondary endpoint composed of major adverse cardio- and cerebrovascular events: 2.7% with ticagrelor monotherapy, 2.5% with 12 months of DAPT.
In the subgroup of TICO-STEMI patients at high bleeding risk, ticagrelor monotherapy was associated with a 12-month major bleeding rate of 1.8%, compared to 6.3% with a full year of DAPT. Conversely, in patients who underwent complex PCI, ticagrelor monotherapy was associated with a 4.9% rate of major adverse cardio- and cerebrovascular events through 1 year, numerically greater than but not statistically significantly different from the 2.7% rate with 12 months of DAPT.
Dr. Kim noted that the study had several limitations: It was open label, had no placebo control, and was underpowered to draw definite conclusions regarding the merits of dropping aspirin and continuing ticagrelor after 3 months in STEMI patients.
“Our findings should be interpreted with caution and call for confirmatory randomized trials,” he stressed.
Session comoderator Roxana Mehran, MD, said she “wholeheartedly” agrees with that assessment.
“We really do need a future trial, and we’re working to design TWILIGHT-STEMI,” a large randomized follow-up to the TWILIGHT trial, which she directed.
“I often imagine that we can have an even shorter duration of aspirin and ticagrelor and go to monotherapy sooner than 3 months in this very, very important subgroup,” added Dr. Mehran, professor of medicine, professor of population science and policy, and director of interventional cardiovascular research and clinical trials at the Icahn School of Medicine at Mount Sinai, New York.
Discussant Marco Valgimigli, MD, PhD, of University Hospital in Bern, Switzerland, calculated that the number-needed-to-treat with ticagrelor monotherapy rather than 12 months of DAPT in order to prevent one additional major bleeding event in TICO-STEMI participants at high bleeding risk was 22, as compared to an NNT of 77 in those without high bleeding risk.
“From a clinical standpoint, this strategy seems particularly appealing in high bleeding risk patients,” the cardiologist concluded at the at the meeting, which was sponsored by the Cardiovascular Research Foundation.
He added, however, that the TICO-STEMI data with respect to complex PCI “are not really reassuring and are probably worth another investigation.”
Dr. Kim reported having no financial conflicts regarding the TICO-STEMI trial, funded by Biotronik.
, a major randomized trial.
“This is the first report assessing the feasibility of ticagrelor monotherapy after short-term DAPT for STEMI patients with drug-eluting stents,” Byeong-Keuk Kim, MD, PhD, noted at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
The positive results were consistent with the earlier TWILIGHT study (Ticagrelor with Aspirin or Alone in High-Risk Patients after Coronary Intervention), which also showed clinical benefit at 1 year for 3 months of DAPT followed by ticagrelor (Brilinta) monotherapy, albeit only in PCI patients without an acute coronary syndrome (ACS) or with non-STEMI ACS (N Engl J Med. 2019 Nov 21;381[21]:2032-42).
TICO-STEMI was a prespecified substudy involving the 1,103 STEMI patients included in the previously reported parent 38-center South Korean TICO (Ticagrelor With or Without Aspirin in Acute Coronary Syndrome After PCI) study of 3,056 ACS patients treated with a second-generation ultrathin biodegradable polymer-coated sirolimus-eluting stent (JAMA. 2020 Jun 16;323[23]:2407-16).
The primary outcome in TICO-STEMI was the 12-month composite rate of net adverse clinical events, composed of major bleeding, all-cause mortality, acute MI, stroke, stent thrombosis, or target vessel revascularization. In an intention-to-treat analysis, the rate was 5.0% in the 12-month DAPT group and 3.7% with ticagrelor monotherapy after 3 months of DAPT, for a 27% relative risk reduction which didn’t achieve statistical significance. However, in an as-treated analysis, the between-group difference in the primary endpoint was stronger: a 5.2% incidence with 12 months of DAPT and 2.3% with ticagrelor monotherapy, for a relative risk reduction of 56%, which was statistically significant.
Major bleeding, one of two key secondary endpoints, was a different story: The incidence within 12 months by intention-to-treat was 2.9% with 12 months of DAPT compared to 0.9% with ticagrelor monotherapy, for a statistically significant 68% relative risk reduction in favor of ticagrelor monotherapy. In contrast, there was no between-group difference in the other secondary endpoint composed of major adverse cardio- and cerebrovascular events: 2.7% with ticagrelor monotherapy, 2.5% with 12 months of DAPT.
In the subgroup of TICO-STEMI patients at high bleeding risk, ticagrelor monotherapy was associated with a 12-month major bleeding rate of 1.8%, compared to 6.3% with a full year of DAPT. Conversely, in patients who underwent complex PCI, ticagrelor monotherapy was associated with a 4.9% rate of major adverse cardio- and cerebrovascular events through 1 year, numerically greater than but not statistically significantly different from the 2.7% rate with 12 months of DAPT.
Dr. Kim noted that the study had several limitations: It was open label, had no placebo control, and was underpowered to draw definite conclusions regarding the merits of dropping aspirin and continuing ticagrelor after 3 months in STEMI patients.
“Our findings should be interpreted with caution and call for confirmatory randomized trials,” he stressed.
Session comoderator Roxana Mehran, MD, said she “wholeheartedly” agrees with that assessment.
“We really do need a future trial, and we’re working to design TWILIGHT-STEMI,” a large randomized follow-up to the TWILIGHT trial, which she directed.
“I often imagine that we can have an even shorter duration of aspirin and ticagrelor and go to monotherapy sooner than 3 months in this very, very important subgroup,” added Dr. Mehran, professor of medicine, professor of population science and policy, and director of interventional cardiovascular research and clinical trials at the Icahn School of Medicine at Mount Sinai, New York.
Discussant Marco Valgimigli, MD, PhD, of University Hospital in Bern, Switzerland, calculated that the number-needed-to-treat with ticagrelor monotherapy rather than 12 months of DAPT in order to prevent one additional major bleeding event in TICO-STEMI participants at high bleeding risk was 22, as compared to an NNT of 77 in those without high bleeding risk.
“From a clinical standpoint, this strategy seems particularly appealing in high bleeding risk patients,” the cardiologist concluded at the at the meeting, which was sponsored by the Cardiovascular Research Foundation.
He added, however, that the TICO-STEMI data with respect to complex PCI “are not really reassuring and are probably worth another investigation.”
Dr. Kim reported having no financial conflicts regarding the TICO-STEMI trial, funded by Biotronik.
, a major randomized trial.
“This is the first report assessing the feasibility of ticagrelor monotherapy after short-term DAPT for STEMI patients with drug-eluting stents,” Byeong-Keuk Kim, MD, PhD, noted at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
The positive results were consistent with the earlier TWILIGHT study (Ticagrelor with Aspirin or Alone in High-Risk Patients after Coronary Intervention), which also showed clinical benefit at 1 year for 3 months of DAPT followed by ticagrelor (Brilinta) monotherapy, albeit only in PCI patients without an acute coronary syndrome (ACS) or with non-STEMI ACS (N Engl J Med. 2019 Nov 21;381[21]:2032-42).
TICO-STEMI was a prespecified substudy involving the 1,103 STEMI patients included in the previously reported parent 38-center South Korean TICO (Ticagrelor With or Without Aspirin in Acute Coronary Syndrome After PCI) study of 3,056 ACS patients treated with a second-generation ultrathin biodegradable polymer-coated sirolimus-eluting stent (JAMA. 2020 Jun 16;323[23]:2407-16).
The primary outcome in TICO-STEMI was the 12-month composite rate of net adverse clinical events, composed of major bleeding, all-cause mortality, acute MI, stroke, stent thrombosis, or target vessel revascularization. In an intention-to-treat analysis, the rate was 5.0% in the 12-month DAPT group and 3.7% with ticagrelor monotherapy after 3 months of DAPT, for a 27% relative risk reduction which didn’t achieve statistical significance. However, in an as-treated analysis, the between-group difference in the primary endpoint was stronger: a 5.2% incidence with 12 months of DAPT and 2.3% with ticagrelor monotherapy, for a relative risk reduction of 56%, which was statistically significant.
Major bleeding, one of two key secondary endpoints, was a different story: The incidence within 12 months by intention-to-treat was 2.9% with 12 months of DAPT compared to 0.9% with ticagrelor monotherapy, for a statistically significant 68% relative risk reduction in favor of ticagrelor monotherapy. In contrast, there was no between-group difference in the other secondary endpoint composed of major adverse cardio- and cerebrovascular events: 2.7% with ticagrelor monotherapy, 2.5% with 12 months of DAPT.
In the subgroup of TICO-STEMI patients at high bleeding risk, ticagrelor monotherapy was associated with a 12-month major bleeding rate of 1.8%, compared to 6.3% with a full year of DAPT. Conversely, in patients who underwent complex PCI, ticagrelor monotherapy was associated with a 4.9% rate of major adverse cardio- and cerebrovascular events through 1 year, numerically greater than but not statistically significantly different from the 2.7% rate with 12 months of DAPT.
Dr. Kim noted that the study had several limitations: It was open label, had no placebo control, and was underpowered to draw definite conclusions regarding the merits of dropping aspirin and continuing ticagrelor after 3 months in STEMI patients.
“Our findings should be interpreted with caution and call for confirmatory randomized trials,” he stressed.
Session comoderator Roxana Mehran, MD, said she “wholeheartedly” agrees with that assessment.
“We really do need a future trial, and we’re working to design TWILIGHT-STEMI,” a large randomized follow-up to the TWILIGHT trial, which she directed.
“I often imagine that we can have an even shorter duration of aspirin and ticagrelor and go to monotherapy sooner than 3 months in this very, very important subgroup,” added Dr. Mehran, professor of medicine, professor of population science and policy, and director of interventional cardiovascular research and clinical trials at the Icahn School of Medicine at Mount Sinai, New York.
Discussant Marco Valgimigli, MD, PhD, of University Hospital in Bern, Switzerland, calculated that the number-needed-to-treat with ticagrelor monotherapy rather than 12 months of DAPT in order to prevent one additional major bleeding event in TICO-STEMI participants at high bleeding risk was 22, as compared to an NNT of 77 in those without high bleeding risk.
“From a clinical standpoint, this strategy seems particularly appealing in high bleeding risk patients,” the cardiologist concluded at the at the meeting, which was sponsored by the Cardiovascular Research Foundation.
He added, however, that the TICO-STEMI data with respect to complex PCI “are not really reassuring and are probably worth another investigation.”
Dr. Kim reported having no financial conflicts regarding the TICO-STEMI trial, funded by Biotronik.
AT TCT 2020
Choose wisely
Four years ago, just prior to the 2016 presidential election, I mentioned the Choosing Wisely campaign in my JFP editorial.1 I said that family physicians should do their part in controlling health care costs by carefully selecting tests and treatments that are known to be effective and avoiding those that are not. This remains as true now as it was then.
The Choosing Wisely campaign was sparked by a family physician, Dr. Howard Brody, in the context of national health care reform. In a 2010 New England Journal of Medicine editorial, he challenged physicians to do their part in controlling health care costs by not ordering tests and treatments that have no value for patients.2 At that time, it was estimated that a third of tests and treatments ordered by US physicians were of marginal or no value.3
Dr. Brody’s editorial caught the attention of the National Physicians Alliance and eventually many other physician organizations. In 2012, the American Board of Internal Medicine Foundation launched the Choosing Wisely initiative; today, the campaign Web site, choosingwisely.org, has a wealth of information and practice recommendations from 78 medical specialty organizations, including the American Academy of Family Physicians (AAFP).
In this month’s issue of JFP, Dr. Kate Rowland has summarized 10 of the most important Choosing Wisely recommendations that apply to family physicians and other primary care clinicians. Here are 5 more recommendations from the Choosing Wisely list of tests and treatments to avoid ordering for your patients:
- Don’t perform pelvic exams on asymptomatic nonpregnant women, unless necessary for guideline-appropriate screening for cervical cancer.
- Don’t routinely screen for prostate cancer using a prostate-specific antigen (PSA) test or digital rectal exam. For men who want PSA screening, it should be performed only after engaging in shared decision-making.
- Don’t order annual electrocardiograms or any other cardiac screening for low-risk patients without symptoms.
- Don’t routinely prescribe antibiotics for otitis media in children ages 2 to 12 years with nonsevere symptoms when observation is reasonable.
- Don’t use dual-energy x-ray absorptiometry screening for osteoporosis in women younger than 65 or men younger than 70 with no risk factors.
In total, AAFP lists 18 recommendations (2 additional recommendations have been withdrawn, based on updated evidence) on the Choosing Wisely Web site. I encourage you to review them to see if you should change any of your current patient recommendations.
1. Hickner J. Count on this no matter who wins the election. J Fam Pract. 2016;65:664.
2. Brody H. Medicine’s ethical responsibility for health care reform—the Top Five list. N Engl J Med. 2010;362:283-285.
3. Fisher ES, Bynum JP, Skinner JS. Slowing the growth of health care costs—lessons from regional variation. N Engl J Med. 2009;360:849-852.
Four years ago, just prior to the 2016 presidential election, I mentioned the Choosing Wisely campaign in my JFP editorial.1 I said that family physicians should do their part in controlling health care costs by carefully selecting tests and treatments that are known to be effective and avoiding those that are not. This remains as true now as it was then.
The Choosing Wisely campaign was sparked by a family physician, Dr. Howard Brody, in the context of national health care reform. In a 2010 New England Journal of Medicine editorial, he challenged physicians to do their part in controlling health care costs by not ordering tests and treatments that have no value for patients.2 At that time, it was estimated that a third of tests and treatments ordered by US physicians were of marginal or no value.3
Dr. Brody’s editorial caught the attention of the National Physicians Alliance and eventually many other physician organizations. In 2012, the American Board of Internal Medicine Foundation launched the Choosing Wisely initiative; today, the campaign Web site, choosingwisely.org, has a wealth of information and practice recommendations from 78 medical specialty organizations, including the American Academy of Family Physicians (AAFP).
In this month’s issue of JFP, Dr. Kate Rowland has summarized 10 of the most important Choosing Wisely recommendations that apply to family physicians and other primary care clinicians. Here are 5 more recommendations from the Choosing Wisely list of tests and treatments to avoid ordering for your patients:
- Don’t perform pelvic exams on asymptomatic nonpregnant women, unless necessary for guideline-appropriate screening for cervical cancer.
- Don’t routinely screen for prostate cancer using a prostate-specific antigen (PSA) test or digital rectal exam. For men who want PSA screening, it should be performed only after engaging in shared decision-making.
- Don’t order annual electrocardiograms or any other cardiac screening for low-risk patients without symptoms.
- Don’t routinely prescribe antibiotics for otitis media in children ages 2 to 12 years with nonsevere symptoms when observation is reasonable.
- Don’t use dual-energy x-ray absorptiometry screening for osteoporosis in women younger than 65 or men younger than 70 with no risk factors.
In total, AAFP lists 18 recommendations (2 additional recommendations have been withdrawn, based on updated evidence) on the Choosing Wisely Web site. I encourage you to review them to see if you should change any of your current patient recommendations.
Four years ago, just prior to the 2016 presidential election, I mentioned the Choosing Wisely campaign in my JFP editorial.1 I said that family physicians should do their part in controlling health care costs by carefully selecting tests and treatments that are known to be effective and avoiding those that are not. This remains as true now as it was then.
The Choosing Wisely campaign was sparked by a family physician, Dr. Howard Brody, in the context of national health care reform. In a 2010 New England Journal of Medicine editorial, he challenged physicians to do their part in controlling health care costs by not ordering tests and treatments that have no value for patients.2 At that time, it was estimated that a third of tests and treatments ordered by US physicians were of marginal or no value.3
Dr. Brody’s editorial caught the attention of the National Physicians Alliance and eventually many other physician organizations. In 2012, the American Board of Internal Medicine Foundation launched the Choosing Wisely initiative; today, the campaign Web site, choosingwisely.org, has a wealth of information and practice recommendations from 78 medical specialty organizations, including the American Academy of Family Physicians (AAFP).
In this month’s issue of JFP, Dr. Kate Rowland has summarized 10 of the most important Choosing Wisely recommendations that apply to family physicians and other primary care clinicians. Here are 5 more recommendations from the Choosing Wisely list of tests and treatments to avoid ordering for your patients:
- Don’t perform pelvic exams on asymptomatic nonpregnant women, unless necessary for guideline-appropriate screening for cervical cancer.
- Don’t routinely screen for prostate cancer using a prostate-specific antigen (PSA) test or digital rectal exam. For men who want PSA screening, it should be performed only after engaging in shared decision-making.
- Don’t order annual electrocardiograms or any other cardiac screening for low-risk patients without symptoms.
- Don’t routinely prescribe antibiotics for otitis media in children ages 2 to 12 years with nonsevere symptoms when observation is reasonable.
- Don’t use dual-energy x-ray absorptiometry screening for osteoporosis in women younger than 65 or men younger than 70 with no risk factors.
In total, AAFP lists 18 recommendations (2 additional recommendations have been withdrawn, based on updated evidence) on the Choosing Wisely Web site. I encourage you to review them to see if you should change any of your current patient recommendations.
1. Hickner J. Count on this no matter who wins the election. J Fam Pract. 2016;65:664.
2. Brody H. Medicine’s ethical responsibility for health care reform—the Top Five list. N Engl J Med. 2010;362:283-285.
3. Fisher ES, Bynum JP, Skinner JS. Slowing the growth of health care costs—lessons from regional variation. N Engl J Med. 2009;360:849-852.
1. Hickner J. Count on this no matter who wins the election. J Fam Pract. 2016;65:664.
2. Brody H. Medicine’s ethical responsibility for health care reform—the Top Five list. N Engl J Med. 2010;362:283-285.
3. Fisher ES, Bynum JP, Skinner JS. Slowing the growth of health care costs—lessons from regional variation. N Engl J Med. 2009;360:849-852.
Female cardiac advantage essentially lost after MI
Women are known to lag 5-10 years behind men in experiencing coronary heart disease (CHD), but new research suggests the gap narrows substantially following a myocardial infarction.
“Women lose a considerable portion, but not all, of their coronary and survival advantage – i.e., the lower event rates – after suffering a MI,” study author Sanne Peters, PhD, George Institute for Global Health, Imperial College London, said in an interview.
Previous studies of sex differences in event rates after a coronary event have produced mixed results and were primarily focused on mortality following MI. Importantly, the studies also lacked a control group without a history of CHD and, thus, were unable to provide a reference point for the disparity in event rates, she explained.
Using the MarketScan and Medicare databases, however, Dr. Peters and colleagues matched 339,890 U.S. adults hospitalized for an MI between January 2015 and December 2016 with 1,359,560 U.S. adults without a history of CHD.
Over a median 1.3 years follow-up, there were 12,518 MIs in the non-CHD group and 27,115 recurrent MIs in the MI group.
The age-standardized rate of MI per 1,000 person-years was 4.0 in women and 6.1 in men without a history of CHD, compared with 57.6 in women and 62.7 in men with a prior MI.
After multivariate adjustment, the women-to-men hazard ratio for MI was 0.64 (95% confidence interval, 0.62-0.67) in the non-CHD group and 0.94 (95% CI, 0.92-0.96) in the prior MI group, the authors reported Oct. 5 in the Journal of the American College of Cardiology
Additional results show the multivariate adjusted women-to-men hazard ratios for three other cardiovascular outcomes follow a similar pattern in the non-CHD and prior MI groups:
- CHD events: 0.53 (95% CI, 0.51-0.54) and 0.87 (95% CI, 0.85-0.89).
- Heart failure hospitalization: 0.93 (95% CI, 0.90-0.96) and 1.02 (95% CI, 1.00-1.04).
- All-cause mortality: 0.72 (95% CI, 0.71-0.73) and 0.90 (95% CI, 0.89-0.92).
“By including a control group of individuals without CHD, we demonstrated that the magnitude of the sex difference in cardiac event rates and survival is considerably smaller among those with prior MI than among those without a history of CHD,” Dr. Peters said.
Of note, the sex differences were consistent across age and race/ethnicity groups for all events, except for heart failure hospitalizations, where the adjusted hazard ratio for women vs. men age 80 years or older was 0.95 for those without a history of CHD (95% CI, 0.91-0.98) and 0.99 (95% CI, 0.96-1.02) for participants with a previous MI.
Dr. Peters said it’s not clear why the female advantage is attenuated post-MI but that one explanation is that women are less likely than men to receive guideline-recommended treatments and dosages or to adhere to prescribed therapies after MI hospitalization, which could put them at a higher risk of subsequent events and worse outcomes than men.
“Sex differences in pathophysiology of CHD and its complications may also explain, to some extent, why the rates of recurrent events are considerably more similar between the sexes than incident event rates,” she said. Compared with men, women have a higher incidence of MI with nonobstructive coronary artery disease and of heart failure with preserved ejection fraction, and evidence-based treatment options are more limited for both conditions.
“After people read this, I think the important thing to recognize is we need to push– as much as we can, with what meds we have, and what data we have – secondary prevention in these women,” Laxmi Mehta, MD, director of preventive cardiology and women’s cardiovascular health at Ohio State University, Columbus, said in an interview.
The lack of a female advantage post-MI should also elicit a “really meaningful conversation with our patients on shared decision-making of why they need to be on medications, remembering on our part to prescribe the medications, remembering to prescribe cardiac rehab, and also reminding our community we do need more data and need to investigate this further,” she said.
In an accompanying editorial, Nanette Wenger, MD, of Emory University, Atlanta, also points out that nonobstructive coronary disease is more common in women and, “yet, guideline-based therapies are those validated for obstructive coronary disease in a predominantly male population but, nonetheless, are applied for nonobstructive coronary disease.”
She advocates for aggressive evaluation and treatment for women with chest pain symptoms as well as early identification of women at risk for CHD, specifically those with metabolic syndrome, preeclampsia, hypertensive disorders of pregnancy, chronic inflammatory conditions, and high-risk race/ethnicity.
“Next, when coronary angiography is undertaken, particularly in younger women, an assiduous search for spontaneous coronary artery dissection and its appropriate management, as well as prompt and evidence-based interventions and medical therapies for an acute coronary event [are indicated],” Dr. Wenger wrote. “However, basic to improving outcomes for women is the elucidation of the optimal noninvasive techniques to identify microvascular disease, which could then enable delineation of appropriate preventive and therapeutic approaches.”
Dr. Peters is supported by a U.K. Medical Research Council Skills Development Fellowship. Dr. Mehta and Dr. Wenger disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Women are known to lag 5-10 years behind men in experiencing coronary heart disease (CHD), but new research suggests the gap narrows substantially following a myocardial infarction.
“Women lose a considerable portion, but not all, of their coronary and survival advantage – i.e., the lower event rates – after suffering a MI,” study author Sanne Peters, PhD, George Institute for Global Health, Imperial College London, said in an interview.
Previous studies of sex differences in event rates after a coronary event have produced mixed results and were primarily focused on mortality following MI. Importantly, the studies also lacked a control group without a history of CHD and, thus, were unable to provide a reference point for the disparity in event rates, she explained.
Using the MarketScan and Medicare databases, however, Dr. Peters and colleagues matched 339,890 U.S. adults hospitalized for an MI between January 2015 and December 2016 with 1,359,560 U.S. adults without a history of CHD.
Over a median 1.3 years follow-up, there were 12,518 MIs in the non-CHD group and 27,115 recurrent MIs in the MI group.
The age-standardized rate of MI per 1,000 person-years was 4.0 in women and 6.1 in men without a history of CHD, compared with 57.6 in women and 62.7 in men with a prior MI.
After multivariate adjustment, the women-to-men hazard ratio for MI was 0.64 (95% confidence interval, 0.62-0.67) in the non-CHD group and 0.94 (95% CI, 0.92-0.96) in the prior MI group, the authors reported Oct. 5 in the Journal of the American College of Cardiology
Additional results show the multivariate adjusted women-to-men hazard ratios for three other cardiovascular outcomes follow a similar pattern in the non-CHD and prior MI groups:
- CHD events: 0.53 (95% CI, 0.51-0.54) and 0.87 (95% CI, 0.85-0.89).
- Heart failure hospitalization: 0.93 (95% CI, 0.90-0.96) and 1.02 (95% CI, 1.00-1.04).
- All-cause mortality: 0.72 (95% CI, 0.71-0.73) and 0.90 (95% CI, 0.89-0.92).
“By including a control group of individuals without CHD, we demonstrated that the magnitude of the sex difference in cardiac event rates and survival is considerably smaller among those with prior MI than among those without a history of CHD,” Dr. Peters said.
Of note, the sex differences were consistent across age and race/ethnicity groups for all events, except for heart failure hospitalizations, where the adjusted hazard ratio for women vs. men age 80 years or older was 0.95 for those without a history of CHD (95% CI, 0.91-0.98) and 0.99 (95% CI, 0.96-1.02) for participants with a previous MI.
Dr. Peters said it’s not clear why the female advantage is attenuated post-MI but that one explanation is that women are less likely than men to receive guideline-recommended treatments and dosages or to adhere to prescribed therapies after MI hospitalization, which could put them at a higher risk of subsequent events and worse outcomes than men.
“Sex differences in pathophysiology of CHD and its complications may also explain, to some extent, why the rates of recurrent events are considerably more similar between the sexes than incident event rates,” she said. Compared with men, women have a higher incidence of MI with nonobstructive coronary artery disease and of heart failure with preserved ejection fraction, and evidence-based treatment options are more limited for both conditions.
“After people read this, I think the important thing to recognize is we need to push– as much as we can, with what meds we have, and what data we have – secondary prevention in these women,” Laxmi Mehta, MD, director of preventive cardiology and women’s cardiovascular health at Ohio State University, Columbus, said in an interview.
The lack of a female advantage post-MI should also elicit a “really meaningful conversation with our patients on shared decision-making of why they need to be on medications, remembering on our part to prescribe the medications, remembering to prescribe cardiac rehab, and also reminding our community we do need more data and need to investigate this further,” she said.
In an accompanying editorial, Nanette Wenger, MD, of Emory University, Atlanta, also points out that nonobstructive coronary disease is more common in women and, “yet, guideline-based therapies are those validated for obstructive coronary disease in a predominantly male population but, nonetheless, are applied for nonobstructive coronary disease.”
She advocates for aggressive evaluation and treatment for women with chest pain symptoms as well as early identification of women at risk for CHD, specifically those with metabolic syndrome, preeclampsia, hypertensive disorders of pregnancy, chronic inflammatory conditions, and high-risk race/ethnicity.
“Next, when coronary angiography is undertaken, particularly in younger women, an assiduous search for spontaneous coronary artery dissection and its appropriate management, as well as prompt and evidence-based interventions and medical therapies for an acute coronary event [are indicated],” Dr. Wenger wrote. “However, basic to improving outcomes for women is the elucidation of the optimal noninvasive techniques to identify microvascular disease, which could then enable delineation of appropriate preventive and therapeutic approaches.”
Dr. Peters is supported by a U.K. Medical Research Council Skills Development Fellowship. Dr. Mehta and Dr. Wenger disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Women are known to lag 5-10 years behind men in experiencing coronary heart disease (CHD), but new research suggests the gap narrows substantially following a myocardial infarction.
“Women lose a considerable portion, but not all, of their coronary and survival advantage – i.e., the lower event rates – after suffering a MI,” study author Sanne Peters, PhD, George Institute for Global Health, Imperial College London, said in an interview.
Previous studies of sex differences in event rates after a coronary event have produced mixed results and were primarily focused on mortality following MI. Importantly, the studies also lacked a control group without a history of CHD and, thus, were unable to provide a reference point for the disparity in event rates, she explained.
Using the MarketScan and Medicare databases, however, Dr. Peters and colleagues matched 339,890 U.S. adults hospitalized for an MI between January 2015 and December 2016 with 1,359,560 U.S. adults without a history of CHD.
Over a median 1.3 years follow-up, there were 12,518 MIs in the non-CHD group and 27,115 recurrent MIs in the MI group.
The age-standardized rate of MI per 1,000 person-years was 4.0 in women and 6.1 in men without a history of CHD, compared with 57.6 in women and 62.7 in men with a prior MI.
After multivariate adjustment, the women-to-men hazard ratio for MI was 0.64 (95% confidence interval, 0.62-0.67) in the non-CHD group and 0.94 (95% CI, 0.92-0.96) in the prior MI group, the authors reported Oct. 5 in the Journal of the American College of Cardiology
Additional results show the multivariate adjusted women-to-men hazard ratios for three other cardiovascular outcomes follow a similar pattern in the non-CHD and prior MI groups:
- CHD events: 0.53 (95% CI, 0.51-0.54) and 0.87 (95% CI, 0.85-0.89).
- Heart failure hospitalization: 0.93 (95% CI, 0.90-0.96) and 1.02 (95% CI, 1.00-1.04).
- All-cause mortality: 0.72 (95% CI, 0.71-0.73) and 0.90 (95% CI, 0.89-0.92).
“By including a control group of individuals without CHD, we demonstrated that the magnitude of the sex difference in cardiac event rates and survival is considerably smaller among those with prior MI than among those without a history of CHD,” Dr. Peters said.
Of note, the sex differences were consistent across age and race/ethnicity groups for all events, except for heart failure hospitalizations, where the adjusted hazard ratio for women vs. men age 80 years or older was 0.95 for those without a history of CHD (95% CI, 0.91-0.98) and 0.99 (95% CI, 0.96-1.02) for participants with a previous MI.
Dr. Peters said it’s not clear why the female advantage is attenuated post-MI but that one explanation is that women are less likely than men to receive guideline-recommended treatments and dosages or to adhere to prescribed therapies after MI hospitalization, which could put them at a higher risk of subsequent events and worse outcomes than men.
“Sex differences in pathophysiology of CHD and its complications may also explain, to some extent, why the rates of recurrent events are considerably more similar between the sexes than incident event rates,” she said. Compared with men, women have a higher incidence of MI with nonobstructive coronary artery disease and of heart failure with preserved ejection fraction, and evidence-based treatment options are more limited for both conditions.
“After people read this, I think the important thing to recognize is we need to push– as much as we can, with what meds we have, and what data we have – secondary prevention in these women,” Laxmi Mehta, MD, director of preventive cardiology and women’s cardiovascular health at Ohio State University, Columbus, said in an interview.
The lack of a female advantage post-MI should also elicit a “really meaningful conversation with our patients on shared decision-making of why they need to be on medications, remembering on our part to prescribe the medications, remembering to prescribe cardiac rehab, and also reminding our community we do need more data and need to investigate this further,” she said.
In an accompanying editorial, Nanette Wenger, MD, of Emory University, Atlanta, also points out that nonobstructive coronary disease is more common in women and, “yet, guideline-based therapies are those validated for obstructive coronary disease in a predominantly male population but, nonetheless, are applied for nonobstructive coronary disease.”
She advocates for aggressive evaluation and treatment for women with chest pain symptoms as well as early identification of women at risk for CHD, specifically those with metabolic syndrome, preeclampsia, hypertensive disorders of pregnancy, chronic inflammatory conditions, and high-risk race/ethnicity.
“Next, when coronary angiography is undertaken, particularly in younger women, an assiduous search for spontaneous coronary artery dissection and its appropriate management, as well as prompt and evidence-based interventions and medical therapies for an acute coronary event [are indicated],” Dr. Wenger wrote. “However, basic to improving outcomes for women is the elucidation of the optimal noninvasive techniques to identify microvascular disease, which could then enable delineation of appropriate preventive and therapeutic approaches.”
Dr. Peters is supported by a U.K. Medical Research Council Skills Development Fellowship. Dr. Mehta and Dr. Wenger disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
T2D treatments create tension between glycemic and cardiovascular goals
It was no surprise that updated guidelines recently published by the European Society of Cardiology for managing cardiovascular disease in patients with diabetes highlighted optimized treatment from a cardiovascular disease perspective, while a nearly concurrent update from two major diabetes societies saw the same issue from a more glycemic point of view.
This difference led to divergent approaches to managing hyperglycemia in patients with type 2 diabetes (T2D). The two diabetes societies that wrote one set of recommendations, the American Diabetes Association and the European Association for the Study of Diabetes, put metformin at the pinnacle of their drug hierarchy. Patients with T2D and established atherosclerotic cardiovascular disease (CVD), chronic kidney disease, or heart failure should all receive metformin first unless contraindicated or not tolerated, their updated consensus report said.
Once metformin is on board, a clinician can then add a second diabetes agent from among the two drug classes recently proven to also reduce cardiovascular and renal events, either the SGLT2 (sodium-glucose transporter 2) inhibitors, or GLP-1 (glucagonlike peptide–1) receptor agonists, they advised.
Cardiovascular disease focus represents a ‘major paradigm shift’
In contrast, the ESC guidelines called for upfront, systematic assessment of CVD risk in patients with T2D before treatment starts, and for patients in high- or very high–risk strata, the guidelines recommended starting the patient first on an SGLT2 inhibitor or a GLP-1 receptor agonist, and only adding metformin in patients who need additional glycemic control.
The guidelines also recommended starting treatment-naive patients with moderate CVD risk on metformin. For patients already on metformin, the new ESC guidelines called for adding an agent from at least one of these two drug classes with proven CVD benefits for those at high or very high CVD risk. The guidelines also note that the CVD benefits of the two newer drug classes differ and hence require further individualization depending on the risks faced by each patient, such as the risk for heart failure hospitalizations.
It’s an approach “driven by data from the cardiovascular outcome trials,” that showed several drugs from both the SGLT2 inhibitor and GLP-1 receptor agonist classes have substantial benefit for preventing cardiovascular events, renal events, hospitalizations for heart failure, and in some studies all-cause mortality, said Francesco Cosentino, MD, during a discussion of the guideline differences at the virtual annual meeting of the European Association for the Study of Diabetes.
The ESC approach also represents “a major paradigm shift,” a “change from a glucose-centric approach to an approach driven by cardiovascular disease events,” summed up Dr. Cosentino, professor of cardiology at the Karolinska Institute in Stockholm and chair of the task force that wrote the ESC’s 2019 updated guidelines. The ESC approach advocates initiating drugs for treating patients with T2D “based on cardiovascular disease risk classification,” he highlighted. Results from some SGLT2 inhibitor cardiovascular outcome trials showed that the CVD benefit was similar regardless of whether or not patients also received metformin.
ADA, EASD call for ‘a different emphasis’
“There is a different emphasis” in the statement issued by the diabetologists of the ADA and EASD, admitted Peter J. Grant, MD, a professor of diabetes and endocrinology at the University of Leeds (England) and cochair of the ESC guidelines task force. Dr. Grant represented the EASD on the task force, and the Association collaborated with the ESC in producing its guidelines.
“The ADA and EASD recommendations “look primarily at glucose control, with cardiovascular disease management as secondary.” In contrast, the ESC guidelines “are primarily cardiovascular disease risk guidelines, with a glucose interest,” Dr. Grant declared.
Despite his involvement in writing the ESC guidelines, Dr. Grant tilted toward the ADA/EASD statement as more globally relevant.
“There is much more to vasculopathy in diabetes than just macrovascular disease. Many patients with type 2 diabetes without macrovascular complications have microvascular disease,” including the potential for retinopathy, nephropathy, and neuropathy, he said. These complications can also have a strong impact on psychological well being and treatment satisfaction.
“It’s important that we’re not glucocentric any more, but it’s equally important that we treat glucose because it has such a benefit for microvascular disease.” Dr. Grant also cited metformin’s long history of safety and good tolerance, clinician comfort prescribing it, and its low price. Heavier reliance on SGLT2 inhibitors and GLP-1 receptor agonists will be expensive for the short term while the cost of these drugs remains high, which places a higher burden on “knowing we’re doing it right,” said Dr. Grant.
Dr. Cosentino pointed out that the higher cost of the drugs in the two classes shown to exert important cardiovascular and renal effects needs to be considered in a cost-effectiveness context, not just by cost alone.
‘Clinical inertia’ could be a danger
Dr. Cosentino played down a major disagreement between the two guidelines, suggesting that “focusing on the differences leads to clinical inertia” by the practicing community when they are unsure how to reconcile the two positions.
Dr. Grant agreed that adding a second drug to metformin right away made sense in at least selected patients. “Look at each patient and decide whether they need glycemic control. If so, and if they also have cardiovascular disease, use both drugs,” metformin, plus one agent from one of the two newer classes.
Something both experts agreed on is that it’s time to generally steer clear of sulfonylurea drugs. “We have evidence for harmful effects from sulfonylureas,” Dr. Cosentino said.
“I’d dump sulfonylureas,” was Dr. Grant’s assessment, but he added that they still have a role for patients who need additional glycemic control but can’t afford the newer drugs.
Dr. Cosentino has had financial relationships with Abbott, AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, Merck, Mundipharma, Novo Nordisk, and Pfizer, Dr. Grant has lectured on behalf of AstraZeneca, GlaxoSmithKline, Merck, Novo Nordisk, the Medicines Company, and Takeda, and he has been an adviser to Amgen, AstraZeneca, Novartis, Novo Nordisk, and Synexus.
It was no surprise that updated guidelines recently published by the European Society of Cardiology for managing cardiovascular disease in patients with diabetes highlighted optimized treatment from a cardiovascular disease perspective, while a nearly concurrent update from two major diabetes societies saw the same issue from a more glycemic point of view.
This difference led to divergent approaches to managing hyperglycemia in patients with type 2 diabetes (T2D). The two diabetes societies that wrote one set of recommendations, the American Diabetes Association and the European Association for the Study of Diabetes, put metformin at the pinnacle of their drug hierarchy. Patients with T2D and established atherosclerotic cardiovascular disease (CVD), chronic kidney disease, or heart failure should all receive metformin first unless contraindicated or not tolerated, their updated consensus report said.
Once metformin is on board, a clinician can then add a second diabetes agent from among the two drug classes recently proven to also reduce cardiovascular and renal events, either the SGLT2 (sodium-glucose transporter 2) inhibitors, or GLP-1 (glucagonlike peptide–1) receptor agonists, they advised.
Cardiovascular disease focus represents a ‘major paradigm shift’
In contrast, the ESC guidelines called for upfront, systematic assessment of CVD risk in patients with T2D before treatment starts, and for patients in high- or very high–risk strata, the guidelines recommended starting the patient first on an SGLT2 inhibitor or a GLP-1 receptor agonist, and only adding metformin in patients who need additional glycemic control.
The guidelines also recommended starting treatment-naive patients with moderate CVD risk on metformin. For patients already on metformin, the new ESC guidelines called for adding an agent from at least one of these two drug classes with proven CVD benefits for those at high or very high CVD risk. The guidelines also note that the CVD benefits of the two newer drug classes differ and hence require further individualization depending on the risks faced by each patient, such as the risk for heart failure hospitalizations.
It’s an approach “driven by data from the cardiovascular outcome trials,” that showed several drugs from both the SGLT2 inhibitor and GLP-1 receptor agonist classes have substantial benefit for preventing cardiovascular events, renal events, hospitalizations for heart failure, and in some studies all-cause mortality, said Francesco Cosentino, MD, during a discussion of the guideline differences at the virtual annual meeting of the European Association for the Study of Diabetes.
The ESC approach also represents “a major paradigm shift,” a “change from a glucose-centric approach to an approach driven by cardiovascular disease events,” summed up Dr. Cosentino, professor of cardiology at the Karolinska Institute in Stockholm and chair of the task force that wrote the ESC’s 2019 updated guidelines. The ESC approach advocates initiating drugs for treating patients with T2D “based on cardiovascular disease risk classification,” he highlighted. Results from some SGLT2 inhibitor cardiovascular outcome trials showed that the CVD benefit was similar regardless of whether or not patients also received metformin.
ADA, EASD call for ‘a different emphasis’
“There is a different emphasis” in the statement issued by the diabetologists of the ADA and EASD, admitted Peter J. Grant, MD, a professor of diabetes and endocrinology at the University of Leeds (England) and cochair of the ESC guidelines task force. Dr. Grant represented the EASD on the task force, and the Association collaborated with the ESC in producing its guidelines.
“The ADA and EASD recommendations “look primarily at glucose control, with cardiovascular disease management as secondary.” In contrast, the ESC guidelines “are primarily cardiovascular disease risk guidelines, with a glucose interest,” Dr. Grant declared.
Despite his involvement in writing the ESC guidelines, Dr. Grant tilted toward the ADA/EASD statement as more globally relevant.
“There is much more to vasculopathy in diabetes than just macrovascular disease. Many patients with type 2 diabetes without macrovascular complications have microvascular disease,” including the potential for retinopathy, nephropathy, and neuropathy, he said. These complications can also have a strong impact on psychological well being and treatment satisfaction.
“It’s important that we’re not glucocentric any more, but it’s equally important that we treat glucose because it has such a benefit for microvascular disease.” Dr. Grant also cited metformin’s long history of safety and good tolerance, clinician comfort prescribing it, and its low price. Heavier reliance on SGLT2 inhibitors and GLP-1 receptor agonists will be expensive for the short term while the cost of these drugs remains high, which places a higher burden on “knowing we’re doing it right,” said Dr. Grant.
Dr. Cosentino pointed out that the higher cost of the drugs in the two classes shown to exert important cardiovascular and renal effects needs to be considered in a cost-effectiveness context, not just by cost alone.
‘Clinical inertia’ could be a danger
Dr. Cosentino played down a major disagreement between the two guidelines, suggesting that “focusing on the differences leads to clinical inertia” by the practicing community when they are unsure how to reconcile the two positions.
Dr. Grant agreed that adding a second drug to metformin right away made sense in at least selected patients. “Look at each patient and decide whether they need glycemic control. If so, and if they also have cardiovascular disease, use both drugs,” metformin, plus one agent from one of the two newer classes.
Something both experts agreed on is that it’s time to generally steer clear of sulfonylurea drugs. “We have evidence for harmful effects from sulfonylureas,” Dr. Cosentino said.
“I’d dump sulfonylureas,” was Dr. Grant’s assessment, but he added that they still have a role for patients who need additional glycemic control but can’t afford the newer drugs.
Dr. Cosentino has had financial relationships with Abbott, AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, Merck, Mundipharma, Novo Nordisk, and Pfizer, Dr. Grant has lectured on behalf of AstraZeneca, GlaxoSmithKline, Merck, Novo Nordisk, the Medicines Company, and Takeda, and he has been an adviser to Amgen, AstraZeneca, Novartis, Novo Nordisk, and Synexus.
It was no surprise that updated guidelines recently published by the European Society of Cardiology for managing cardiovascular disease in patients with diabetes highlighted optimized treatment from a cardiovascular disease perspective, while a nearly concurrent update from two major diabetes societies saw the same issue from a more glycemic point of view.
This difference led to divergent approaches to managing hyperglycemia in patients with type 2 diabetes (T2D). The two diabetes societies that wrote one set of recommendations, the American Diabetes Association and the European Association for the Study of Diabetes, put metformin at the pinnacle of their drug hierarchy. Patients with T2D and established atherosclerotic cardiovascular disease (CVD), chronic kidney disease, or heart failure should all receive metformin first unless contraindicated or not tolerated, their updated consensus report said.
Once metformin is on board, a clinician can then add a second diabetes agent from among the two drug classes recently proven to also reduce cardiovascular and renal events, either the SGLT2 (sodium-glucose transporter 2) inhibitors, or GLP-1 (glucagonlike peptide–1) receptor agonists, they advised.
Cardiovascular disease focus represents a ‘major paradigm shift’
In contrast, the ESC guidelines called for upfront, systematic assessment of CVD risk in patients with T2D before treatment starts, and for patients in high- or very high–risk strata, the guidelines recommended starting the patient first on an SGLT2 inhibitor or a GLP-1 receptor agonist, and only adding metformin in patients who need additional glycemic control.
The guidelines also recommended starting treatment-naive patients with moderate CVD risk on metformin. For patients already on metformin, the new ESC guidelines called for adding an agent from at least one of these two drug classes with proven CVD benefits for those at high or very high CVD risk. The guidelines also note that the CVD benefits of the two newer drug classes differ and hence require further individualization depending on the risks faced by each patient, such as the risk for heart failure hospitalizations.
It’s an approach “driven by data from the cardiovascular outcome trials,” that showed several drugs from both the SGLT2 inhibitor and GLP-1 receptor agonist classes have substantial benefit for preventing cardiovascular events, renal events, hospitalizations for heart failure, and in some studies all-cause mortality, said Francesco Cosentino, MD, during a discussion of the guideline differences at the virtual annual meeting of the European Association for the Study of Diabetes.
The ESC approach also represents “a major paradigm shift,” a “change from a glucose-centric approach to an approach driven by cardiovascular disease events,” summed up Dr. Cosentino, professor of cardiology at the Karolinska Institute in Stockholm and chair of the task force that wrote the ESC’s 2019 updated guidelines. The ESC approach advocates initiating drugs for treating patients with T2D “based on cardiovascular disease risk classification,” he highlighted. Results from some SGLT2 inhibitor cardiovascular outcome trials showed that the CVD benefit was similar regardless of whether or not patients also received metformin.
ADA, EASD call for ‘a different emphasis’
“There is a different emphasis” in the statement issued by the diabetologists of the ADA and EASD, admitted Peter J. Grant, MD, a professor of diabetes and endocrinology at the University of Leeds (England) and cochair of the ESC guidelines task force. Dr. Grant represented the EASD on the task force, and the Association collaborated with the ESC in producing its guidelines.
“The ADA and EASD recommendations “look primarily at glucose control, with cardiovascular disease management as secondary.” In contrast, the ESC guidelines “are primarily cardiovascular disease risk guidelines, with a glucose interest,” Dr. Grant declared.
Despite his involvement in writing the ESC guidelines, Dr. Grant tilted toward the ADA/EASD statement as more globally relevant.
“There is much more to vasculopathy in diabetes than just macrovascular disease. Many patients with type 2 diabetes without macrovascular complications have microvascular disease,” including the potential for retinopathy, nephropathy, and neuropathy, he said. These complications can also have a strong impact on psychological well being and treatment satisfaction.
“It’s important that we’re not glucocentric any more, but it’s equally important that we treat glucose because it has such a benefit for microvascular disease.” Dr. Grant also cited metformin’s long history of safety and good tolerance, clinician comfort prescribing it, and its low price. Heavier reliance on SGLT2 inhibitors and GLP-1 receptor agonists will be expensive for the short term while the cost of these drugs remains high, which places a higher burden on “knowing we’re doing it right,” said Dr. Grant.
Dr. Cosentino pointed out that the higher cost of the drugs in the two classes shown to exert important cardiovascular and renal effects needs to be considered in a cost-effectiveness context, not just by cost alone.
‘Clinical inertia’ could be a danger
Dr. Cosentino played down a major disagreement between the two guidelines, suggesting that “focusing on the differences leads to clinical inertia” by the practicing community when they are unsure how to reconcile the two positions.
Dr. Grant agreed that adding a second drug to metformin right away made sense in at least selected patients. “Look at each patient and decide whether they need glycemic control. If so, and if they also have cardiovascular disease, use both drugs,” metformin, plus one agent from one of the two newer classes.
Something both experts agreed on is that it’s time to generally steer clear of sulfonylurea drugs. “We have evidence for harmful effects from sulfonylureas,” Dr. Cosentino said.
“I’d dump sulfonylureas,” was Dr. Grant’s assessment, but he added that they still have a role for patients who need additional glycemic control but can’t afford the newer drugs.
Dr. Cosentino has had financial relationships with Abbott, AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, Merck, Mundipharma, Novo Nordisk, and Pfizer, Dr. Grant has lectured on behalf of AstraZeneca, GlaxoSmithKline, Merck, Novo Nordisk, the Medicines Company, and Takeda, and he has been an adviser to Amgen, AstraZeneca, Novartis, Novo Nordisk, and Synexus.
FROM EASD 2020
Primary prevention of VTE spans a spectrum
Venous thromboembolism (VTE) is a common and dangerous disease, affecting 0.1%-0.2% of the population annually—a rate that might be underreported.1 VTE is a collective term for venous blood clots, including (1) deep vein thrombosis (DVT) of peripheral veins and (2) pulmonary embolism, which occurs after a clot travels through the heart and becomes lodged in the pulmonary vasculature. Two-thirds of VTE cases present clinically as DVT2; most mortality from VTE disease is caused by the 20% of cases of pulmonary embolism that present as sudden death.1
VTE is comparable to myocardial infarction (MI) in incidence and severity. In 2008, 208 of every 100,000 people had an MI, with a 30-day mortality of 16/100,0003; VTE disease has an annual incidence of 161 of every 100,000 people and a 28-day mortality of 18/100,000.4 Although the incidence and severity of MI are steadily decreasing, the rate of VTE appears constant.3,5 The high mortality of VTE suggests that primary prevention, which we discuss in this article, is valuable (see “Key points: Primary prevention of venous thromboembolism”).
SIDEBAR
Key points: Primary prevention of venous thromboembolism
- Primary prevention of venous thromboembolism (VTE), a disease with mortality similar to myocardial infarction, should be an important consideration in at-risk patients.
- Although statins reduce the risk of VTE, their use is justified only if they are also required for prevention of cardiovascular disease.
- The risk of travel-related VTE can be reduced by wearing compression stockings.
- The choice of particular methods of contraception and of hormone replacement therapy can reduce VTE risk.
- Because of the risk of bleeding, using anticoagulants for primary prevention of VTE is justified only in certain circumstances.
- Pregnancy is the only condition in which there is a guideline indication for thrombophilia testing, because test results in this setting can change recommendations for preventing VTE.
- Using a risk-stratification model is key to determining risk in both medically and surgically hospitalized patients. Trauma and major orthopedic surgery always place the patient at high risk of VTE.
Risk factors
Virchow’s triad of venous stasis, vascular injury, and hypercoagulability describes predisposing factors for VTE.6 Although venous valves promote blood flow, they produce isolated low-flow areas adjacent to valves that become concentrated and locally hypoxic, increasing the risk of clotting.7 The great majority of DVTs (≥ 96%) occur in the lower extremity,8 starting in the calf; there, 75% of cases resolve spontaneously before they extend into the deep veins of the proximal leg.7 One-half of DVTs that do move into the proximal leg eventually embolize.7
Major risk factors for VTE comprise inherited conditions, medical history, medical therapeutics, and behaviors (TABLE 1).9-11 Unlike the preventive management of coronary artery disease (CAD), there is no simple, generalized prevention algorithm to address VTE risk factors.
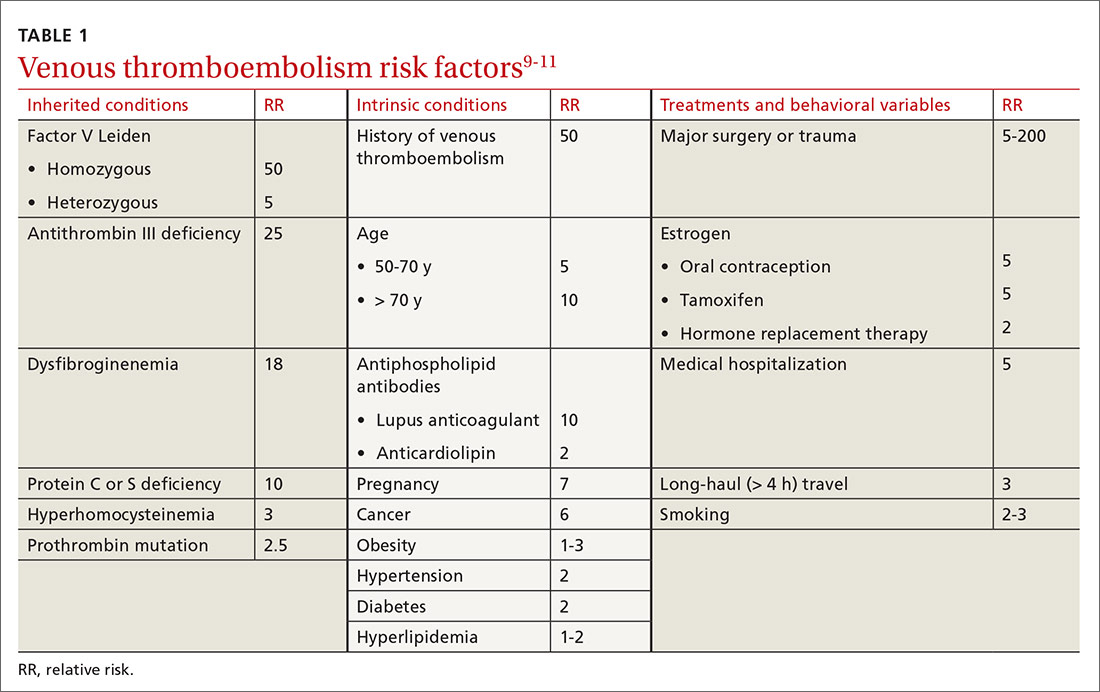
Risk factors for VTE and CAD overlap. Risk factors for atherosclerosis—obesity, diabetes, smoking, hypertension, hyperlipidemia—also increase the risk of VTE (TABLE 1).9-11 The association between risk factors for VTE and atherosclerosis is demonstrated by a doubling of the risk of MI and stroke in the year following VTE.11 Lifestyle changes are expected to reduce the risk of VTE, as they do for acute CAD, but studies are lacking to confirm this connection. There is no prospective evidence showing that weight loss or control of diabetes or hypertension reduces the risk of VTE.12 Smoking cessation does appear to reduce risk: Former smokers have the same VTE risk as never-smokers.13
Thrombophilia testing: Not generally useful
Inherited and acquired thrombophilic conditions define a group of disorders in which the risk of VTE is increased. Although thrombophilia testing was once considered for primary and secondary prevention of VTE, such testing is rarely used now because proof of benefit is lacking: A large case–control study showed that thrombophilia testing did not predict recurrence after a first VTE.14 Guidelines of the American College of Chest Physicians (ACCP) do not address thrombophilia, and the American Society of Hematology recommends against thrombophilia testing after a provoked VTE.15,16
Primary prophylaxis of patients with a family history of VTE and inherited thrombophilia is controversial. Patients with both a family history of VTE and demonstrated thrombophilia do have double the average incidence of VTE, but this increased risk does not offset the significant bleeding risk associated with anticoagulation.17 Recommendations for thrombophilia testing are limited to certain situations in pregnancy, discussed in a bit.16,18,19
Continue to: Primary prevention of VTE in the clinic
Primary prevention of VTE in the clinic
There is no single, overarching preventive strategy for VTE in an ambulatory patient (although statins, discussed in a moment, offer some benefit, broadly). There are, however, distinct behavioral characteristics and medical circumstances for which opportunities exist to reduce VTE risk—for example, when a person engages in long-distance travel, receives hormonal therapy, is pregnant, or has cancer. In each scenario, recognizing and mitigating risk are important.
Statins offer a (slight) benefit
There is evidence that statins reduce the risk of VTE—slightly20-23:
- A large randomized, controlled trial showed that rosuvastatin, 20 mg/d, reduced the rate of VTE, compared to placebo; however, the 2-year number needed to treat (NNT) was 349.20 The VTE benefit is minimal, however, compared to primary prevention of cardiovascular disease with statins (5-year NNT = 56).21 The sole significant adverse event associated with statins was new-onset type 2 diabetes (5-year number needed to harm = 235).21
- A subsequent meta-analysis confirmed a small reduction in VTE risk with statins.22 In its 2012 guidelines, ACCP declined to issue a recommendation on the use of statins for VTE prevention.23 When considering statins for primary cardiovascular disease prevention, take the additional VTE prevention into account.
Simple strategies can help prevent travel-related VTE
Travel is a common inciting factor for VTE. A systematic review showed that VTE risk triples after travel of ≥ 4 hours, increasing by 20% with each additional 2 hours.24 Most VTE occurs in travelers who have other VTE risk factors.25 Based on case–control studies,23 guidelines recommend these preventive measures:
- frequent calf exercises
- sitting in an aisle seat during air travel
- keeping hydrated.
A Cochrane review showed that graded compression stockings reduce asymptomatic DVT in travelers by a factor of 10, in high- and low-risk patients.26
VTE risk varies with type of hormonal contraception
Most contraceptives increase VTE risk (TABLE 227,28). Risk with combined oral contraceptives varies with the amount of estrogen and progesterone. To reduce VTE risk with oral contraceptives, patients can use an agent that contains a lower dose of estrogen or one in which levonorgestrel replaces other progesterones.27
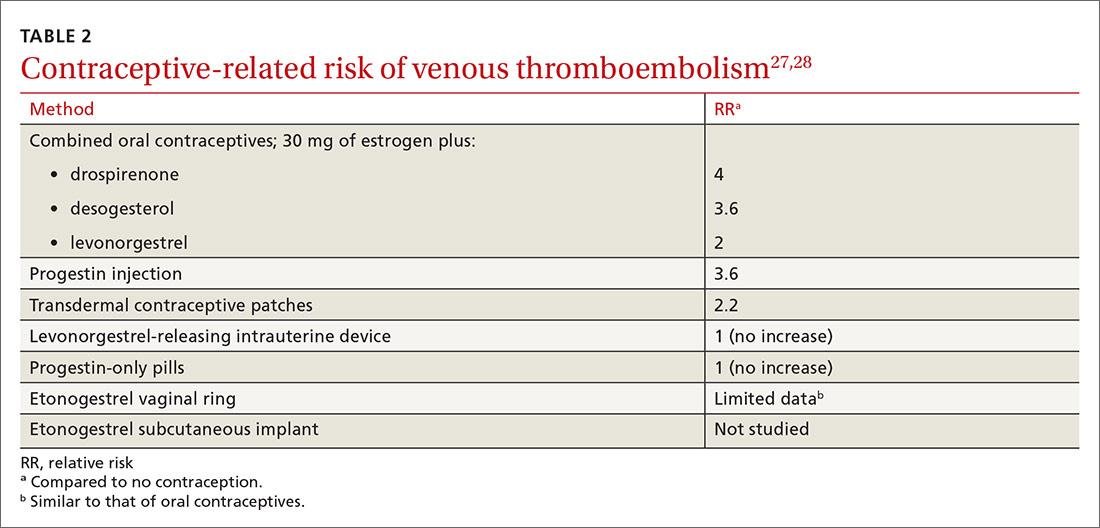
Continue to: Studies suggest that the levonorgestrel-releasing...
Studies suggest that the levonorgestrel-releasing intrauterine device and progestin-only pills are not associated with an increase in VTE risk.27 Although the quality of evidence varies, most nonoral hormonal contraceptives have been determined to carry a risk of VTE that is similar to that of combined oral contraceptives.28
In hormone replacement, avoid pills to lower risk
Hormone replacement therapy (HRT) for postmenopausal women increases VTE risk when administered in oral form, with combined estrogen and progestin HRT doubling the risk and estrogen-only formulations having a lower risk.29 VTE risk is highest in the first 6 months of HRT, declining to that of a non-HRT user within 5 years.29 Neither transdermal HRT nor estrogen creams increase the risk of VTE, according to a systematic review.30 The estradiol-containing vaginal ring also does not confer increased risk.29
Pregnancy, thrombophilia, and VTE prevention
VTE affects as many as 0.2% of pregnancies but causes 9% of pregnancy-related deaths.18 The severity of VTE in pregnancy led the American College of Obstetricians and Gynecologists (ACOG) to recommend primary VTE prophylaxis in patients with certain thrombophilias.18 Thrombophilia testing is recommended in patients with proven high-risk thrombophilia in a first-degree relative.18 ACOG recognizes 5 thrombophilias considered to carry a high risk of VTE in pregnancy18:
- homozygous Factor V Leiden
- homozygous prothrombin G20210A mutation
- antithrombin deficiency
- heterozygous Factor V Leiden and prothrombin G20210A mutation
- antiphospholipid antibody syndrome.
ACOG recommends limiting thrombophilia testing to (1) any specific thrombophilia carried by a relative and (2) possibly, the antiphospholipid antibodies anticardiolipin and lupus anticoagulant.18,19 Antiphospholipid testing is recommended when there is a history of stillbirth, 3 early pregnancy losses, or delivery earlier than 34 weeks secondary to preeclampsia.19
Primary VTE prophylaxis is recommended for pregnant patients with a high-risk thrombophilia; low-molecular-weight heparin (LMWH) is safe and its effects are predictable.18 Because postpartum risk of VTE is higher than antepartum risk, postpartum prophylaxis is also recommended with lower-risk thrombophilias18; a vitamin K antagonist or LMWH can be used.18 ACCP and ACOG recommendations for VTE prophylaxis in pregnancy differ slightly (TABLE 316,18,19).
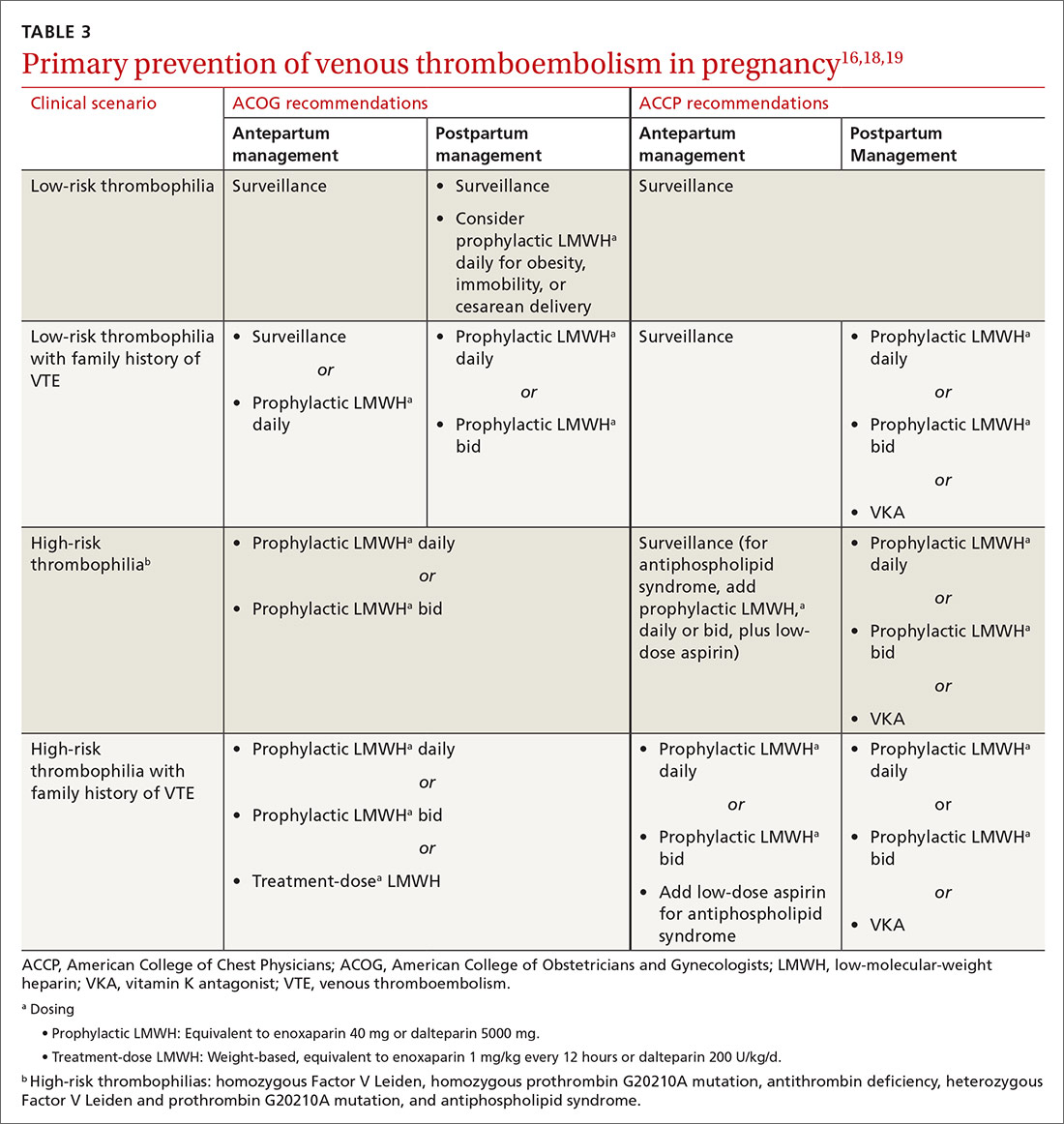
Continue to: Cancer increases risks of VTE and bleeding
Cancer increases risks of VTE and bleeding
Cancer increases VTE risk > 6-fold31; metastases, chemotherapy, and radiotherapy further increase risk. Cancer also greatly increases the risk of bleeding: Cancer patients with VTE have an annual major bleeding rate ≥ 20%.32 Guidelines do not recommend primary VTE prophylaxis for cancer, although American Society of Clinical Oncology guidelines discuss consideration of prophylaxis for select, high-risk patients,33,34 including those with multiple myeloma, metastatic gastrointestinal cancer, or metastatic brain cancer.31,34 Recent evidence (discussed in a moment) supports the use of apixaban for primary VTE prevention during chemotherapy for high-risk cancer.
The Khorana Risk Score (TABLE 435,36) for VTE was developed and validated for use in patients with solid cancer35: A score of 2 conveys nearly a 10% risk of VTE over 6 months.36 A recent study of 550 cancer patients with a Khorana score of ≥ 2—the first evidence of risk-guided primary VTE prevention in cancer—showed that primary prophylaxis with 2.5 mg of apixaban, bid, reduced the risk of VTE (NNT = 17); however, the number needed to harm (for major bleeding) was 59.37 Mortality was not changed with apixaban treatment
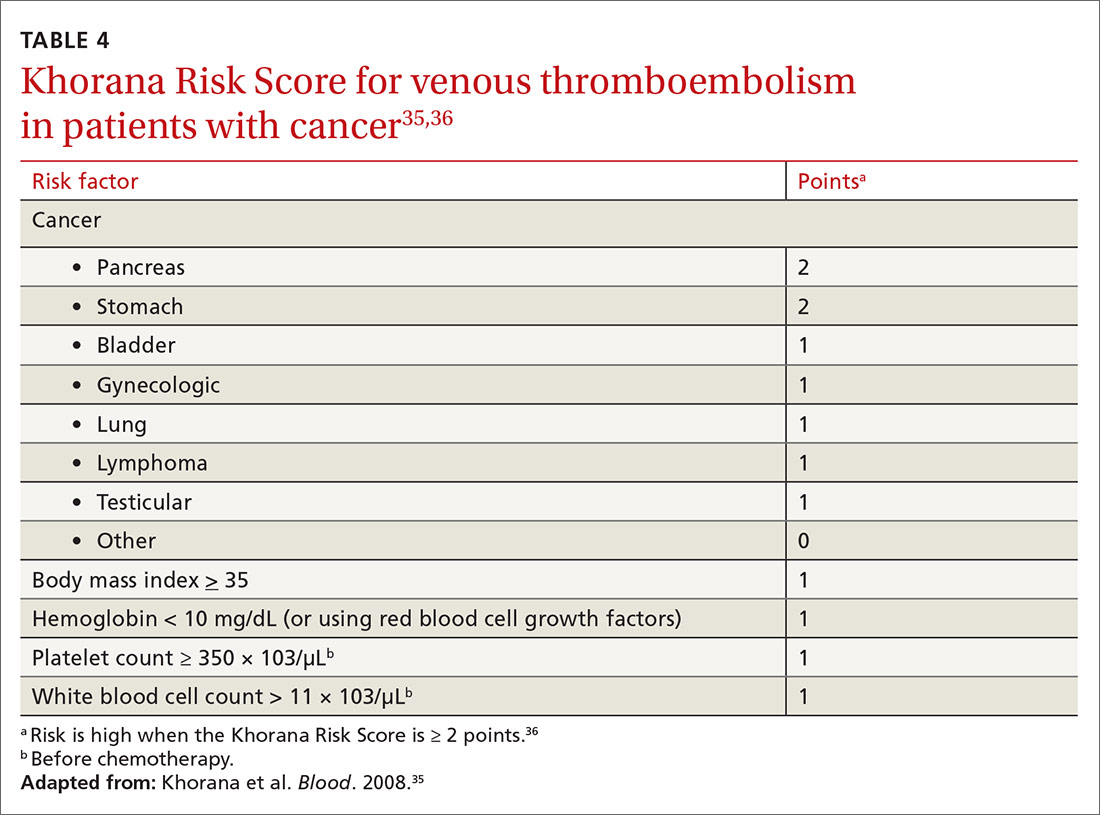
Primary VTE prevention in med-surg hospitalizations
The risk of VTE increases significantly during hospitalization, although not enough to justify universal prophylaxis. Recommended prevention strategies for different classes of hospitalized patients are summarized below.
In medically hospitalized patients, risk is stratified with a risk-assessment model. Medically hospitalized patients have, on average, a VTE risk of 1.2%23; 12 risk-assessment models designed to stratify risk were recently compared.38 Two models, the Caprini Score (TABLE 5)39 and the IMPROVE VTE Risk Calculator,40 were best able to identify low-risk patients (negative predictive value, > 99%).38 American Society of Hematology guidelines recommend IMPROVE VTE or the Padua Prediction Score for risk stratification.41 While the Caprini score only designates 11% of eventual VTE cases as low risk, both the IMPROVE VTE and Padua scores miss more than 35% of eventual VTE.38
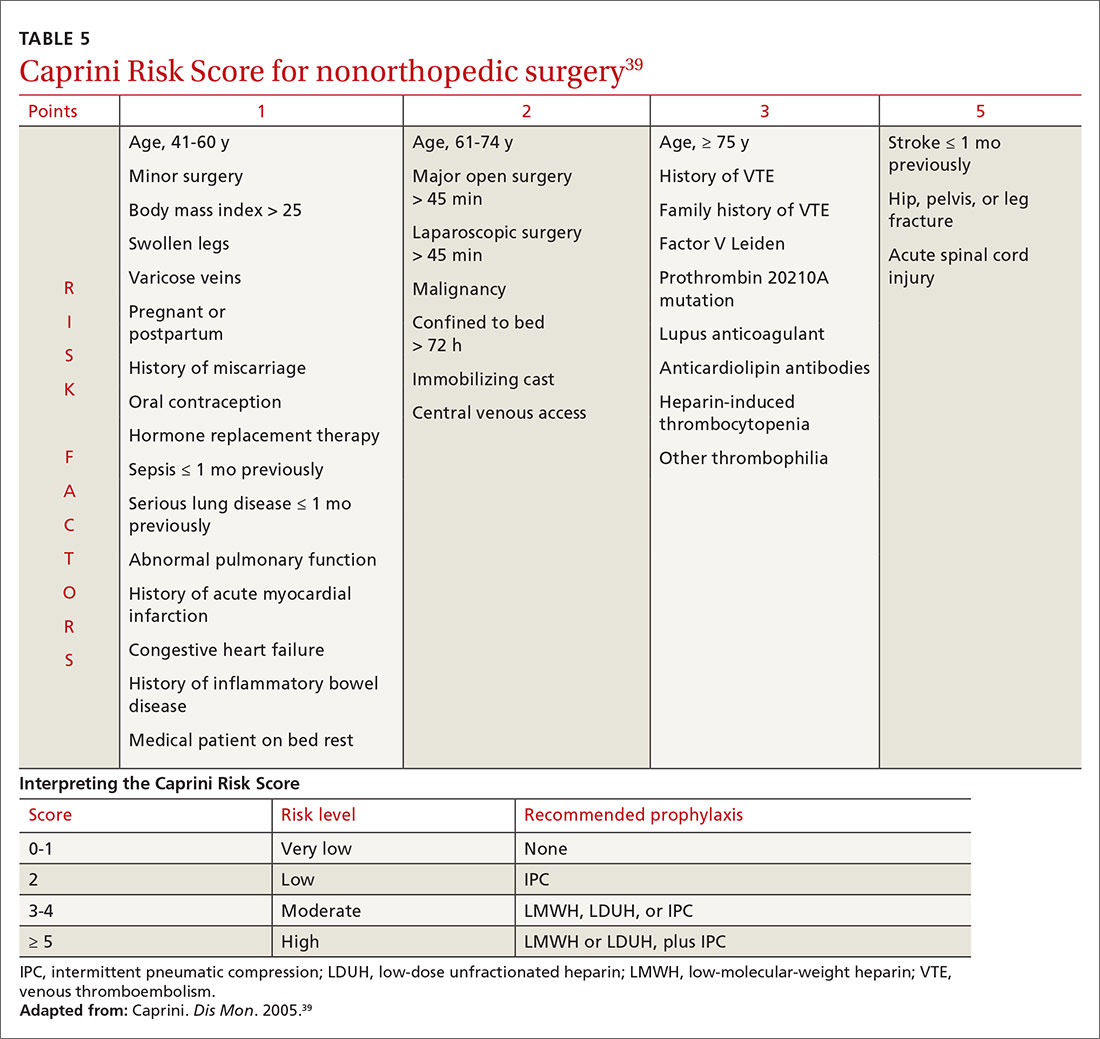
Because LMWH prophylaxis has been shown to reduce VTE by 40% without increasing the risk of major bleeding, using Caprini should prevent 2 VTEs for every 1000 patients, without an increase in major bleeding and with 13 additional minor bleeding events.42
Continue to: Critically ill patients
Critically ill patients are assumed to be at high risk of VTE and do not require stratification.23 For high-risk patients, prophylaxis with LMWH, low-dose unfractionated heparin (LDUH), or fondaparinux is recommended for the duration of admission.23 For patients at high risk of both VTE and bleeding, mechanical prophylaxis with intermittent pneumatic compression (IPC) is recommended instead of LMWH, LDUH, or fondaparinux.23
Surgery, like trauma (see next page), increases the risk of VTE and has been well studied. Prophylaxis after orthopedic surgery differs from that of other types of surgery.
In orthopedic surgery, risk depends on the procedure. For major orthopedic surgery, including total hip or knee arthroplasty and hip fracture surgery, VTE prophylaxis is recommended for 35 days postsurgically.43 LMWH is the preferred agent, although many other means have been shown to be beneficial.44 A recent systematic review demonstrated that aspirin is not inferior to other medications after hip or knee arthroplasty.45 No mechanical or pharmacotherapeutic prophylaxis is generally recommended after nonmajor orthopedic surgery.43
Nonorthopedic surgery is stratified by risk factors, using Caprini44 (TABLE 539). For medium-risk patients (Caprini score, 3-4) LDUH, LMWH, or IPC is recommended; for high-risk patients (Caprini score, ≥ 5) preventive treatment should combine pharmacotherapeutic and mechanical prophylaxis.46 A recent meta-analysis, comprising 14,776 patients, showed that surgical patients with a Caprini score ≥ 7 had a reduced incidence of VTE when given chemoprophylaxis, whereas patients whose score is < 7 do not benefit from chemoprophylaxis.43 When bleeding risk is high, IPC is recommended as sole therapy.43 Prophylaxis is not recommended when risk (determined by the Caprini score) is low.46
Post-hospitalization. Risk of VTE can persist for as long as 90 days after hospitalization; this finding has led to evaluation of the benefit of prolonged chemoprophylaxis.23 Extended-duration LMWH prophylaxis decreases the incidence of VTE, but at the cost of increased risk of major bleeding.47 Based on this evidence, guidelines recommend against prolonged-duration anticoagulation.23 A 2016 trial showed that 35 days of the direct-acting anticoagulant betrixaban reduced the risk of symptomatic VTE events, compared to 10 days of LMWH (NNT = 167), without increased risk of bleeding.48 This is a limited benefit, however, that is unlikely to change guideline recommendations.
Continue to: Trauma
Trauma: VTE risk increases with severity
Trauma increases the risk of VTE considerably. A national study showed that 1.5% of admitted trauma patients experienced VTE during hospitalization and that 1.2% were readmitted for VTE within 1 year.49 As many as 32% of trauma patients admitted to the intensive care unit experience VTE despite appropriate prophylaxis.50 A Cochrane Review51 found that:
- prophylaxis significantly reduces DVT risk
- pharmacotherapeutic prophylaxis is more effective than mechanical prophylaxis
- LMWH is more effective than LDUH.
Guidelines recommend that major trauma patients receive prophylaxis with LMWH, LDUH, or IPC.46
CORRESPONDENCE
Michael J. Arnold, MD, CDR, MC, USN; Uniformed Services University of the Health Sciences, 4301 Jones Bridge Road, Jacksonville, FL 32214; [email protected].
1. Beckman MG, Hooper WC, Critchley SE, et al. Venous thromboembolism: a public health concern. Am J Prev Med. 2010. 38(4 suppl):S495-S501.
2. Tagalakis V, Patenaude V, Kahn SR, et al. Incidence of and mortality from venous thromboembolism in a real-world population: the Q-VTE Study Cohort. Am J Med. 2013;126:832.e13-e21.
3. Yeh RW, Sidney S, Chandra M, et al. Population trends in the incidence and outcomes of acute myocardial infarction. N Engl J Med. 2010. 362:2155-2165.
4. Cushman M, Tsai AW, White RH, et al. Deep vein thrombosis and pulmonary embolism in two cohorts: the longitudinal investigation of thromboembolism etiology. Am J Med. 2004;117:19-25.
5. Goldhaber SZ. Venous thromboembolism: epidemiology and magnitude of the problem. Best Pract Res Clin Haematol. 2012;25:235-242.
6. Stone J, Hangge P, Albadawi H, et al. Deep vein thrombosis: pathogenesis, diagnosis, and medical management. Cardiovasc Diagn Ther. 2017;7(suppl 3):S276-S284.
7. Olaf M, Cooney R. Deep venous thrombosis. Emerg Med Clin North Am. 2017;35:743-770.
8. Sajid MS, Ahmed N, Desai M, et al. Upper limb deep vein thrombosis: a literature review to streamline the protocol for management. Acta Haematol. 2007;118:10-18.
9. Bates SM, Ginsberg JS. Clinical practice. Treatment of deep-vein thrombosis. N Engl J Med. 2004;351:268-277.
10. Chandra D, Parisini E, Mozaffarian D. Meta-analysis: travel and risk for venous thromboembolism. Ann Intern Med. 2009;151:180-190.
11. Goldhaber SZ. Risk factors for venous thromboembolism. J Am Col Cardiol. 2010;56:1-7.
12. Yang G, De Staercke C, Hooper WC. The effects of obesity on venous thromboembolism: a review. Open J Prev Med. 2012;2:499-509.
13. Severinsen MT, Kristensen SR, Johnsen SP, et al. Smoking and venous thromboembolism: a Danish follow-up study. J Thromb Haemost. 2009;7:1297-1303.
14. Coppens M, Reijnders JH, Middeldorp S, et al. Testing for inherited thrombophilia does not reduce the recurrence of venous thrombosis. J Thromb Haemost. 2008;6:1474-1477.
15. Choosing Wisely. American Society of Hematology. Ten things physicians and patients should question. www.choosingwisely.org/societies/american-society-of-hematology/. Accessed September 28, 2020.
16. Bates SM, Greer IA, Middeldorp S, et al. VTE, thrombophilia, antithrombotic therapy, and pregnancy: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e691S-e736S.
17. Vossen CY, Conard J, Fontcuberta J, et al. Risk of a first venous thrombotic event in carriers of a familial thrombophilic defect. The European Prospective Cohort on Thrombophilia (EPCOT). J Thromb Haemost. 2005;3:459-464.
18. Practice Bulletin No. 197: Inherited thrombophilias in pregnancy. Obstet Gynecol. 2018;132:e18-e34.
19. Committee on Practice Bulletins—Obstetrics, American College of Obstetricians and Gynecologists. Practice Bulletin No. 132: Antiphospholipid syndrome. Obstet Gynecol. 2012;120:1514-1521.
20. Glynn RJ, Danielson E, Fonseca FAH, et al. A randomized trial of rosuvastatin in the prevention of venous thromboembolism. N Engl J Med. 2009;360:1851-1861.
21. Taylor F, Huffman MD, Macedo AF, et al. Statins for the primary prevention of cardiovascular disease. Cochrane Database Syst Rev. 2013(1):CD004816.
22. Squizzato A, Galli M, Romualdi E, et al. Statins, fibrates, and venous thromboembolism: a meta-analysis. Eur Heart J. 2010;31:1248-1256.
23. Kahn SR, Lim W, Dunn AS, et al. Prevention of VTE in nonsurgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e195S-e226S.
24. Kelman CW, Kortt MA, Becker NG, et al. Deep vein thrombosis and air travel: record linkage study. BMJ. 2003;327:1072.
25. Johnston RV, Hudson MF; . Travelers’ thrombosis. Aviat Space Environ Med. 2014;85:191-194.
26. Clarke MJ, Broderick C, Hopewell S, et al. Compression stockings for preventing deep vein thrombosis in airline passengers. Cochrane Database Syst Rev. 2016;9:CD004002.
27. van Hylckama Vlieg A, Middledorp S. Hormone therapies and venous thromboembolism: where are we now? J Thromb Haemost. 2011;9:257-266.
28. Tepper NK, Dragoman MV, Gaffield ME, et al. Nonoral combined hormonal contraceptives and thromboembolism: a systematic review. Contraception. 2017;95:130-139.
29. Lekovic D, Miljic P, Dmitrovic A, et al. How do you decide on hormone replacement therapy in women with risk of venous thromboembolism? Blood Rev. 2017;31:151-157.
30. Rovinski D, Ramos RB, Fighera TM, et al. Risk of venous thromboembolism events in postmenopausal women using oral versus non-oral hormone therapy: a systematic review and meta-analysis. Thromb Res. 2018;168:83-95.
31. Horsted F, West J, Grainge MJ. Risk of venous thromboembolism in patients with cancer: a systematic review and meta-analysis. PLoS Med. 2012;9:e1001275.
32. Kamphuisen PW, Beyer-Westendorf J. Bleeding complications during anticoagulant treatment in patients with cancer. Thromb Res. 2014;133(suppl 2):S49-S55.
33. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST Guideline and Expert Panel Report. Chest. 2016;149:315-352.
34. Lyman GH, Khorana AA, Kuderer NM, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: American Society of Clinical Oncology Clinical Practice Guideline update. J Clin Oncol. 2013;31:2189-2204.
35. Khorana AA, Kuderer NM, Culakova E, et al. Development and validation of a predictive model for chemotherapy-associated thrombosis. Blood. 2008;111:4902-4907.
36. Ay C, Dunkler D, Marosi C, et al. Prediction of venous thromboembolism in cancer patients. Blood. 2010;116:5377-5382.
37. Carrier M, Abou-Nassar K, Mallick R, et al; AVERT Investigators. Apixaban to prevent venous thromboembolism in patients with cancer. N Engl J Med. 2019;380:711-719.
38. Cobben MRR, Nemeth B, Lijfering WM, et al. Validation of risk assessment models for venous thrombosis in hospitalized medical patients. Res Pract Thromb Haemost. 2019;3:217-225.
39. Caprini JA. Thrombosis risk assessment as a guide to quality patient care. Dis Mon. 2005;51:70-78.
40. Spyropoulos AC, Anderson FA Jr, FitzGerald G, et al; IMPROVE Investigators. Predictive and associative models to identify hospitalized medical patients at risk for VTE. Chest. 2011;140:706-714.
41. Kanaan AO, Silva MA, Donovan JL, et al. Meta-analysis of venous thromboembolism prophylaxis in medically Ill patients. Clin Ther. 2007;29:2395-2405.
42. HJ, Cushman M, Burnett AE, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: prophylaxis for hospitalized and nonhospitalized medical patients. Blood Adv. 2018;2:3198-3225.
43. Falck-Ytter Y, Francis CW, Johanson NA, et al. Prevention of VTE in orthopedic surgery patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e278S-e325S.
44. Pannucci CJ, Swistun L, MacDonald JK, et al. Individualized venous thromboembolism risk stratification using the 2005 Caprini Score to identify the benefits and harms of chemoprophylaxis in surgical patients: a meta-analysis. Ann Surg. 2017;265:1094-1103.
45. Matharu GS, Kunutsor SK, Judge A, et al. Clinical effectiveness and safety of aspirin for venous thromboembolism prophylaxis after total hip and knee replacement: a systematic review and meta-analysis of randomized clinical trials. JAMA Intern Med. 2020;180:376-384.
46. Gould MK, Garcia DA, Wren SM, et al. Prevention of VTE in nonorthopedic surgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e227S-e277S.
47. Hull RD, Schellong SM, Tapson VF, et al. Extended-duration venous thromboembolism prophylaxis in acutely ill medical patients with recent reduced mobility: a randomized trial. Ann Intern Med. 2010;153:8-18.
48. Cohen AT, Harrington RA, Goldhaber SZ, et al. Extended thromboprophylaxis with betrixaban in acutely ill medical patients. N Engl J Med. 2016;375:534-544.
49. Rattan R, Parreco J, Eidelson SA, et al. Hidden burden of venous thromboembolism after trauma: a national analysis. J Trauma Acute Care Surg. 2018;85:899-906.
50. Yumoto T, Naito H, Yamakawa Y, et al. Venous thromboembolism in major trauma patients: a single-center retrospective cohort study of the epidemiology and utility of D-dimer for screening. Acute Med Surg. 2017;4:394-400.
51. Barrera LM, Perel P, Ker K, et al. Thromboprophylaxis for trauma patients. Cochrane Database Syst Rev. 2013(3):CD008303.
Venous thromboembolism (VTE) is a common and dangerous disease, affecting 0.1%-0.2% of the population annually—a rate that might be underreported.1 VTE is a collective term for venous blood clots, including (1) deep vein thrombosis (DVT) of peripheral veins and (2) pulmonary embolism, which occurs after a clot travels through the heart and becomes lodged in the pulmonary vasculature. Two-thirds of VTE cases present clinically as DVT2; most mortality from VTE disease is caused by the 20% of cases of pulmonary embolism that present as sudden death.1
VTE is comparable to myocardial infarction (MI) in incidence and severity. In 2008, 208 of every 100,000 people had an MI, with a 30-day mortality of 16/100,0003; VTE disease has an annual incidence of 161 of every 100,000 people and a 28-day mortality of 18/100,000.4 Although the incidence and severity of MI are steadily decreasing, the rate of VTE appears constant.3,5 The high mortality of VTE suggests that primary prevention, which we discuss in this article, is valuable (see “Key points: Primary prevention of venous thromboembolism”).
SIDEBAR
Key points: Primary prevention of venous thromboembolism
- Primary prevention of venous thromboembolism (VTE), a disease with mortality similar to myocardial infarction, should be an important consideration in at-risk patients.
- Although statins reduce the risk of VTE, their use is justified only if they are also required for prevention of cardiovascular disease.
- The risk of travel-related VTE can be reduced by wearing compression stockings.
- The choice of particular methods of contraception and of hormone replacement therapy can reduce VTE risk.
- Because of the risk of bleeding, using anticoagulants for primary prevention of VTE is justified only in certain circumstances.
- Pregnancy is the only condition in which there is a guideline indication for thrombophilia testing, because test results in this setting can change recommendations for preventing VTE.
- Using a risk-stratification model is key to determining risk in both medically and surgically hospitalized patients. Trauma and major orthopedic surgery always place the patient at high risk of VTE.
Risk factors
Virchow’s triad of venous stasis, vascular injury, and hypercoagulability describes predisposing factors for VTE.6 Although venous valves promote blood flow, they produce isolated low-flow areas adjacent to valves that become concentrated and locally hypoxic, increasing the risk of clotting.7 The great majority of DVTs (≥ 96%) occur in the lower extremity,8 starting in the calf; there, 75% of cases resolve spontaneously before they extend into the deep veins of the proximal leg.7 One-half of DVTs that do move into the proximal leg eventually embolize.7
Major risk factors for VTE comprise inherited conditions, medical history, medical therapeutics, and behaviors (TABLE 1).9-11 Unlike the preventive management of coronary artery disease (CAD), there is no simple, generalized prevention algorithm to address VTE risk factors.

Risk factors for VTE and CAD overlap. Risk factors for atherosclerosis—obesity, diabetes, smoking, hypertension, hyperlipidemia—also increase the risk of VTE (TABLE 1).9-11 The association between risk factors for VTE and atherosclerosis is demonstrated by a doubling of the risk of MI and stroke in the year following VTE.11 Lifestyle changes are expected to reduce the risk of VTE, as they do for acute CAD, but studies are lacking to confirm this connection. There is no prospective evidence showing that weight loss or control of diabetes or hypertension reduces the risk of VTE.12 Smoking cessation does appear to reduce risk: Former smokers have the same VTE risk as never-smokers.13
Thrombophilia testing: Not generally useful
Inherited and acquired thrombophilic conditions define a group of disorders in which the risk of VTE is increased. Although thrombophilia testing was once considered for primary and secondary prevention of VTE, such testing is rarely used now because proof of benefit is lacking: A large case–control study showed that thrombophilia testing did not predict recurrence after a first VTE.14 Guidelines of the American College of Chest Physicians (ACCP) do not address thrombophilia, and the American Society of Hematology recommends against thrombophilia testing after a provoked VTE.15,16
Primary prophylaxis of patients with a family history of VTE and inherited thrombophilia is controversial. Patients with both a family history of VTE and demonstrated thrombophilia do have double the average incidence of VTE, but this increased risk does not offset the significant bleeding risk associated with anticoagulation.17 Recommendations for thrombophilia testing are limited to certain situations in pregnancy, discussed in a bit.16,18,19
Continue to: Primary prevention of VTE in the clinic
Primary prevention of VTE in the clinic
There is no single, overarching preventive strategy for VTE in an ambulatory patient (although statins, discussed in a moment, offer some benefit, broadly). There are, however, distinct behavioral characteristics and medical circumstances for which opportunities exist to reduce VTE risk—for example, when a person engages in long-distance travel, receives hormonal therapy, is pregnant, or has cancer. In each scenario, recognizing and mitigating risk are important.
Statins offer a (slight) benefit
There is evidence that statins reduce the risk of VTE—slightly20-23:
- A large randomized, controlled trial showed that rosuvastatin, 20 mg/d, reduced the rate of VTE, compared to placebo; however, the 2-year number needed to treat (NNT) was 349.20 The VTE benefit is minimal, however, compared to primary prevention of cardiovascular disease with statins (5-year NNT = 56).21 The sole significant adverse event associated with statins was new-onset type 2 diabetes (5-year number needed to harm = 235).21
- A subsequent meta-analysis confirmed a small reduction in VTE risk with statins.22 In its 2012 guidelines, ACCP declined to issue a recommendation on the use of statins for VTE prevention.23 When considering statins for primary cardiovascular disease prevention, take the additional VTE prevention into account.
Simple strategies can help prevent travel-related VTE
Travel is a common inciting factor for VTE. A systematic review showed that VTE risk triples after travel of ≥ 4 hours, increasing by 20% with each additional 2 hours.24 Most VTE occurs in travelers who have other VTE risk factors.25 Based on case–control studies,23 guidelines recommend these preventive measures:
- frequent calf exercises
- sitting in an aisle seat during air travel
- keeping hydrated.
A Cochrane review showed that graded compression stockings reduce asymptomatic DVT in travelers by a factor of 10, in high- and low-risk patients.26
VTE risk varies with type of hormonal contraception
Most contraceptives increase VTE risk (TABLE 227,28). Risk with combined oral contraceptives varies with the amount of estrogen and progesterone. To reduce VTE risk with oral contraceptives, patients can use an agent that contains a lower dose of estrogen or one in which levonorgestrel replaces other progesterones.27

Continue to: Studies suggest that the levonorgestrel-releasing...
Studies suggest that the levonorgestrel-releasing intrauterine device and progestin-only pills are not associated with an increase in VTE risk.27 Although the quality of evidence varies, most nonoral hormonal contraceptives have been determined to carry a risk of VTE that is similar to that of combined oral contraceptives.28
In hormone replacement, avoid pills to lower risk
Hormone replacement therapy (HRT) for postmenopausal women increases VTE risk when administered in oral form, with combined estrogen and progestin HRT doubling the risk and estrogen-only formulations having a lower risk.29 VTE risk is highest in the first 6 months of HRT, declining to that of a non-HRT user within 5 years.29 Neither transdermal HRT nor estrogen creams increase the risk of VTE, according to a systematic review.30 The estradiol-containing vaginal ring also does not confer increased risk.29
Pregnancy, thrombophilia, and VTE prevention
VTE affects as many as 0.2% of pregnancies but causes 9% of pregnancy-related deaths.18 The severity of VTE in pregnancy led the American College of Obstetricians and Gynecologists (ACOG) to recommend primary VTE prophylaxis in patients with certain thrombophilias.18 Thrombophilia testing is recommended in patients with proven high-risk thrombophilia in a first-degree relative.18 ACOG recognizes 5 thrombophilias considered to carry a high risk of VTE in pregnancy18:
- homozygous Factor V Leiden
- homozygous prothrombin G20210A mutation
- antithrombin deficiency
- heterozygous Factor V Leiden and prothrombin G20210A mutation
- antiphospholipid antibody syndrome.
ACOG recommends limiting thrombophilia testing to (1) any specific thrombophilia carried by a relative and (2) possibly, the antiphospholipid antibodies anticardiolipin and lupus anticoagulant.18,19 Antiphospholipid testing is recommended when there is a history of stillbirth, 3 early pregnancy losses, or delivery earlier than 34 weeks secondary to preeclampsia.19
Primary VTE prophylaxis is recommended for pregnant patients with a high-risk thrombophilia; low-molecular-weight heparin (LMWH) is safe and its effects are predictable.18 Because postpartum risk of VTE is higher than antepartum risk, postpartum prophylaxis is also recommended with lower-risk thrombophilias18; a vitamin K antagonist or LMWH can be used.18 ACCP and ACOG recommendations for VTE prophylaxis in pregnancy differ slightly (TABLE 316,18,19).

Continue to: Cancer increases risks of VTE and bleeding
Cancer increases risks of VTE and bleeding
Cancer increases VTE risk > 6-fold31; metastases, chemotherapy, and radiotherapy further increase risk. Cancer also greatly increases the risk of bleeding: Cancer patients with VTE have an annual major bleeding rate ≥ 20%.32 Guidelines do not recommend primary VTE prophylaxis for cancer, although American Society of Clinical Oncology guidelines discuss consideration of prophylaxis for select, high-risk patients,33,34 including those with multiple myeloma, metastatic gastrointestinal cancer, or metastatic brain cancer.31,34 Recent evidence (discussed in a moment) supports the use of apixaban for primary VTE prevention during chemotherapy for high-risk cancer.
The Khorana Risk Score (TABLE 435,36) for VTE was developed and validated for use in patients with solid cancer35: A score of 2 conveys nearly a 10% risk of VTE over 6 months.36 A recent study of 550 cancer patients with a Khorana score of ≥ 2—the first evidence of risk-guided primary VTE prevention in cancer—showed that primary prophylaxis with 2.5 mg of apixaban, bid, reduced the risk of VTE (NNT = 17); however, the number needed to harm (for major bleeding) was 59.37 Mortality was not changed with apixaban treatment

Primary VTE prevention in med-surg hospitalizations
The risk of VTE increases significantly during hospitalization, although not enough to justify universal prophylaxis. Recommended prevention strategies for different classes of hospitalized patients are summarized below.
In medically hospitalized patients, risk is stratified with a risk-assessment model. Medically hospitalized patients have, on average, a VTE risk of 1.2%23; 12 risk-assessment models designed to stratify risk were recently compared.38 Two models, the Caprini Score (TABLE 5)39 and the IMPROVE VTE Risk Calculator,40 were best able to identify low-risk patients (negative predictive value, > 99%).38 American Society of Hematology guidelines recommend IMPROVE VTE or the Padua Prediction Score for risk stratification.41 While the Caprini score only designates 11% of eventual VTE cases as low risk, both the IMPROVE VTE and Padua scores miss more than 35% of eventual VTE.38

Because LMWH prophylaxis has been shown to reduce VTE by 40% without increasing the risk of major bleeding, using Caprini should prevent 2 VTEs for every 1000 patients, without an increase in major bleeding and with 13 additional minor bleeding events.42
Continue to: Critically ill patients
Critically ill patients are assumed to be at high risk of VTE and do not require stratification.23 For high-risk patients, prophylaxis with LMWH, low-dose unfractionated heparin (LDUH), or fondaparinux is recommended for the duration of admission.23 For patients at high risk of both VTE and bleeding, mechanical prophylaxis with intermittent pneumatic compression (IPC) is recommended instead of LMWH, LDUH, or fondaparinux.23
Surgery, like trauma (see next page), increases the risk of VTE and has been well studied. Prophylaxis after orthopedic surgery differs from that of other types of surgery.
In orthopedic surgery, risk depends on the procedure. For major orthopedic surgery, including total hip or knee arthroplasty and hip fracture surgery, VTE prophylaxis is recommended for 35 days postsurgically.43 LMWH is the preferred agent, although many other means have been shown to be beneficial.44 A recent systematic review demonstrated that aspirin is not inferior to other medications after hip or knee arthroplasty.45 No mechanical or pharmacotherapeutic prophylaxis is generally recommended after nonmajor orthopedic surgery.43
Nonorthopedic surgery is stratified by risk factors, using Caprini44 (TABLE 539). For medium-risk patients (Caprini score, 3-4) LDUH, LMWH, or IPC is recommended; for high-risk patients (Caprini score, ≥ 5) preventive treatment should combine pharmacotherapeutic and mechanical prophylaxis.46 A recent meta-analysis, comprising 14,776 patients, showed that surgical patients with a Caprini score ≥ 7 had a reduced incidence of VTE when given chemoprophylaxis, whereas patients whose score is < 7 do not benefit from chemoprophylaxis.43 When bleeding risk is high, IPC is recommended as sole therapy.43 Prophylaxis is not recommended when risk (determined by the Caprini score) is low.46
Post-hospitalization. Risk of VTE can persist for as long as 90 days after hospitalization; this finding has led to evaluation of the benefit of prolonged chemoprophylaxis.23 Extended-duration LMWH prophylaxis decreases the incidence of VTE, but at the cost of increased risk of major bleeding.47 Based on this evidence, guidelines recommend against prolonged-duration anticoagulation.23 A 2016 trial showed that 35 days of the direct-acting anticoagulant betrixaban reduced the risk of symptomatic VTE events, compared to 10 days of LMWH (NNT = 167), without increased risk of bleeding.48 This is a limited benefit, however, that is unlikely to change guideline recommendations.
Continue to: Trauma
Trauma: VTE risk increases with severity
Trauma increases the risk of VTE considerably. A national study showed that 1.5% of admitted trauma patients experienced VTE during hospitalization and that 1.2% were readmitted for VTE within 1 year.49 As many as 32% of trauma patients admitted to the intensive care unit experience VTE despite appropriate prophylaxis.50 A Cochrane Review51 found that:
- prophylaxis significantly reduces DVT risk
- pharmacotherapeutic prophylaxis is more effective than mechanical prophylaxis
- LMWH is more effective than LDUH.
Guidelines recommend that major trauma patients receive prophylaxis with LMWH, LDUH, or IPC.46
CORRESPONDENCE
Michael J. Arnold, MD, CDR, MC, USN; Uniformed Services University of the Health Sciences, 4301 Jones Bridge Road, Jacksonville, FL 32214; [email protected].
Venous thromboembolism (VTE) is a common and dangerous disease, affecting 0.1%-0.2% of the population annually—a rate that might be underreported.1 VTE is a collective term for venous blood clots, including (1) deep vein thrombosis (DVT) of peripheral veins and (2) pulmonary embolism, which occurs after a clot travels through the heart and becomes lodged in the pulmonary vasculature. Two-thirds of VTE cases present clinically as DVT2; most mortality from VTE disease is caused by the 20% of cases of pulmonary embolism that present as sudden death.1
VTE is comparable to myocardial infarction (MI) in incidence and severity. In 2008, 208 of every 100,000 people had an MI, with a 30-day mortality of 16/100,0003; VTE disease has an annual incidence of 161 of every 100,000 people and a 28-day mortality of 18/100,000.4 Although the incidence and severity of MI are steadily decreasing, the rate of VTE appears constant.3,5 The high mortality of VTE suggests that primary prevention, which we discuss in this article, is valuable (see “Key points: Primary prevention of venous thromboembolism”).
SIDEBAR
Key points: Primary prevention of venous thromboembolism
- Primary prevention of venous thromboembolism (VTE), a disease with mortality similar to myocardial infarction, should be an important consideration in at-risk patients.
- Although statins reduce the risk of VTE, their use is justified only if they are also required for prevention of cardiovascular disease.
- The risk of travel-related VTE can be reduced by wearing compression stockings.
- The choice of particular methods of contraception and of hormone replacement therapy can reduce VTE risk.
- Because of the risk of bleeding, using anticoagulants for primary prevention of VTE is justified only in certain circumstances.
- Pregnancy is the only condition in which there is a guideline indication for thrombophilia testing, because test results in this setting can change recommendations for preventing VTE.
- Using a risk-stratification model is key to determining risk in both medically and surgically hospitalized patients. Trauma and major orthopedic surgery always place the patient at high risk of VTE.
Risk factors
Virchow’s triad of venous stasis, vascular injury, and hypercoagulability describes predisposing factors for VTE.6 Although venous valves promote blood flow, they produce isolated low-flow areas adjacent to valves that become concentrated and locally hypoxic, increasing the risk of clotting.7 The great majority of DVTs (≥ 96%) occur in the lower extremity,8 starting in the calf; there, 75% of cases resolve spontaneously before they extend into the deep veins of the proximal leg.7 One-half of DVTs that do move into the proximal leg eventually embolize.7
Major risk factors for VTE comprise inherited conditions, medical history, medical therapeutics, and behaviors (TABLE 1).9-11 Unlike the preventive management of coronary artery disease (CAD), there is no simple, generalized prevention algorithm to address VTE risk factors.

Risk factors for VTE and CAD overlap. Risk factors for atherosclerosis—obesity, diabetes, smoking, hypertension, hyperlipidemia—also increase the risk of VTE (TABLE 1).9-11 The association between risk factors for VTE and atherosclerosis is demonstrated by a doubling of the risk of MI and stroke in the year following VTE.11 Lifestyle changes are expected to reduce the risk of VTE, as they do for acute CAD, but studies are lacking to confirm this connection. There is no prospective evidence showing that weight loss or control of diabetes or hypertension reduces the risk of VTE.12 Smoking cessation does appear to reduce risk: Former smokers have the same VTE risk as never-smokers.13
Thrombophilia testing: Not generally useful
Inherited and acquired thrombophilic conditions define a group of disorders in which the risk of VTE is increased. Although thrombophilia testing was once considered for primary and secondary prevention of VTE, such testing is rarely used now because proof of benefit is lacking: A large case–control study showed that thrombophilia testing did not predict recurrence after a first VTE.14 Guidelines of the American College of Chest Physicians (ACCP) do not address thrombophilia, and the American Society of Hematology recommends against thrombophilia testing after a provoked VTE.15,16
Primary prophylaxis of patients with a family history of VTE and inherited thrombophilia is controversial. Patients with both a family history of VTE and demonstrated thrombophilia do have double the average incidence of VTE, but this increased risk does not offset the significant bleeding risk associated with anticoagulation.17 Recommendations for thrombophilia testing are limited to certain situations in pregnancy, discussed in a bit.16,18,19
Continue to: Primary prevention of VTE in the clinic
Primary prevention of VTE in the clinic
There is no single, overarching preventive strategy for VTE in an ambulatory patient (although statins, discussed in a moment, offer some benefit, broadly). There are, however, distinct behavioral characteristics and medical circumstances for which opportunities exist to reduce VTE risk—for example, when a person engages in long-distance travel, receives hormonal therapy, is pregnant, or has cancer. In each scenario, recognizing and mitigating risk are important.
Statins offer a (slight) benefit
There is evidence that statins reduce the risk of VTE—slightly20-23:
- A large randomized, controlled trial showed that rosuvastatin, 20 mg/d, reduced the rate of VTE, compared to placebo; however, the 2-year number needed to treat (NNT) was 349.20 The VTE benefit is minimal, however, compared to primary prevention of cardiovascular disease with statins (5-year NNT = 56).21 The sole significant adverse event associated with statins was new-onset type 2 diabetes (5-year number needed to harm = 235).21
- A subsequent meta-analysis confirmed a small reduction in VTE risk with statins.22 In its 2012 guidelines, ACCP declined to issue a recommendation on the use of statins for VTE prevention.23 When considering statins for primary cardiovascular disease prevention, take the additional VTE prevention into account.
Simple strategies can help prevent travel-related VTE
Travel is a common inciting factor for VTE. A systematic review showed that VTE risk triples after travel of ≥ 4 hours, increasing by 20% with each additional 2 hours.24 Most VTE occurs in travelers who have other VTE risk factors.25 Based on case–control studies,23 guidelines recommend these preventive measures:
- frequent calf exercises
- sitting in an aisle seat during air travel
- keeping hydrated.
A Cochrane review showed that graded compression stockings reduce asymptomatic DVT in travelers by a factor of 10, in high- and low-risk patients.26
VTE risk varies with type of hormonal contraception
Most contraceptives increase VTE risk (TABLE 227,28). Risk with combined oral contraceptives varies with the amount of estrogen and progesterone. To reduce VTE risk with oral contraceptives, patients can use an agent that contains a lower dose of estrogen or one in which levonorgestrel replaces other progesterones.27

Continue to: Studies suggest that the levonorgestrel-releasing...
Studies suggest that the levonorgestrel-releasing intrauterine device and progestin-only pills are not associated with an increase in VTE risk.27 Although the quality of evidence varies, most nonoral hormonal contraceptives have been determined to carry a risk of VTE that is similar to that of combined oral contraceptives.28
In hormone replacement, avoid pills to lower risk
Hormone replacement therapy (HRT) for postmenopausal women increases VTE risk when administered in oral form, with combined estrogen and progestin HRT doubling the risk and estrogen-only formulations having a lower risk.29 VTE risk is highest in the first 6 months of HRT, declining to that of a non-HRT user within 5 years.29 Neither transdermal HRT nor estrogen creams increase the risk of VTE, according to a systematic review.30 The estradiol-containing vaginal ring also does not confer increased risk.29
Pregnancy, thrombophilia, and VTE prevention
VTE affects as many as 0.2% of pregnancies but causes 9% of pregnancy-related deaths.18 The severity of VTE in pregnancy led the American College of Obstetricians and Gynecologists (ACOG) to recommend primary VTE prophylaxis in patients with certain thrombophilias.18 Thrombophilia testing is recommended in patients with proven high-risk thrombophilia in a first-degree relative.18 ACOG recognizes 5 thrombophilias considered to carry a high risk of VTE in pregnancy18:
- homozygous Factor V Leiden
- homozygous prothrombin G20210A mutation
- antithrombin deficiency
- heterozygous Factor V Leiden and prothrombin G20210A mutation
- antiphospholipid antibody syndrome.
ACOG recommends limiting thrombophilia testing to (1) any specific thrombophilia carried by a relative and (2) possibly, the antiphospholipid antibodies anticardiolipin and lupus anticoagulant.18,19 Antiphospholipid testing is recommended when there is a history of stillbirth, 3 early pregnancy losses, or delivery earlier than 34 weeks secondary to preeclampsia.19
Primary VTE prophylaxis is recommended for pregnant patients with a high-risk thrombophilia; low-molecular-weight heparin (LMWH) is safe and its effects are predictable.18 Because postpartum risk of VTE is higher than antepartum risk, postpartum prophylaxis is also recommended with lower-risk thrombophilias18; a vitamin K antagonist or LMWH can be used.18 ACCP and ACOG recommendations for VTE prophylaxis in pregnancy differ slightly (TABLE 316,18,19).

Continue to: Cancer increases risks of VTE and bleeding
Cancer increases risks of VTE and bleeding
Cancer increases VTE risk > 6-fold31; metastases, chemotherapy, and radiotherapy further increase risk. Cancer also greatly increases the risk of bleeding: Cancer patients with VTE have an annual major bleeding rate ≥ 20%.32 Guidelines do not recommend primary VTE prophylaxis for cancer, although American Society of Clinical Oncology guidelines discuss consideration of prophylaxis for select, high-risk patients,33,34 including those with multiple myeloma, metastatic gastrointestinal cancer, or metastatic brain cancer.31,34 Recent evidence (discussed in a moment) supports the use of apixaban for primary VTE prevention during chemotherapy for high-risk cancer.
The Khorana Risk Score (TABLE 435,36) for VTE was developed and validated for use in patients with solid cancer35: A score of 2 conveys nearly a 10% risk of VTE over 6 months.36 A recent study of 550 cancer patients with a Khorana score of ≥ 2—the first evidence of risk-guided primary VTE prevention in cancer—showed that primary prophylaxis with 2.5 mg of apixaban, bid, reduced the risk of VTE (NNT = 17); however, the number needed to harm (for major bleeding) was 59.37 Mortality was not changed with apixaban treatment

Primary VTE prevention in med-surg hospitalizations
The risk of VTE increases significantly during hospitalization, although not enough to justify universal prophylaxis. Recommended prevention strategies for different classes of hospitalized patients are summarized below.
In medically hospitalized patients, risk is stratified with a risk-assessment model. Medically hospitalized patients have, on average, a VTE risk of 1.2%23; 12 risk-assessment models designed to stratify risk were recently compared.38 Two models, the Caprini Score (TABLE 5)39 and the IMPROVE VTE Risk Calculator,40 were best able to identify low-risk patients (negative predictive value, > 99%).38 American Society of Hematology guidelines recommend IMPROVE VTE or the Padua Prediction Score for risk stratification.41 While the Caprini score only designates 11% of eventual VTE cases as low risk, both the IMPROVE VTE and Padua scores miss more than 35% of eventual VTE.38

Because LMWH prophylaxis has been shown to reduce VTE by 40% without increasing the risk of major bleeding, using Caprini should prevent 2 VTEs for every 1000 patients, without an increase in major bleeding and with 13 additional minor bleeding events.42
Continue to: Critically ill patients
Critically ill patients are assumed to be at high risk of VTE and do not require stratification.23 For high-risk patients, prophylaxis with LMWH, low-dose unfractionated heparin (LDUH), or fondaparinux is recommended for the duration of admission.23 For patients at high risk of both VTE and bleeding, mechanical prophylaxis with intermittent pneumatic compression (IPC) is recommended instead of LMWH, LDUH, or fondaparinux.23
Surgery, like trauma (see next page), increases the risk of VTE and has been well studied. Prophylaxis after orthopedic surgery differs from that of other types of surgery.
In orthopedic surgery, risk depends on the procedure. For major orthopedic surgery, including total hip or knee arthroplasty and hip fracture surgery, VTE prophylaxis is recommended for 35 days postsurgically.43 LMWH is the preferred agent, although many other means have been shown to be beneficial.44 A recent systematic review demonstrated that aspirin is not inferior to other medications after hip or knee arthroplasty.45 No mechanical or pharmacotherapeutic prophylaxis is generally recommended after nonmajor orthopedic surgery.43
Nonorthopedic surgery is stratified by risk factors, using Caprini44 (TABLE 539). For medium-risk patients (Caprini score, 3-4) LDUH, LMWH, or IPC is recommended; for high-risk patients (Caprini score, ≥ 5) preventive treatment should combine pharmacotherapeutic and mechanical prophylaxis.46 A recent meta-analysis, comprising 14,776 patients, showed that surgical patients with a Caprini score ≥ 7 had a reduced incidence of VTE when given chemoprophylaxis, whereas patients whose score is < 7 do not benefit from chemoprophylaxis.43 When bleeding risk is high, IPC is recommended as sole therapy.43 Prophylaxis is not recommended when risk (determined by the Caprini score) is low.46
Post-hospitalization. Risk of VTE can persist for as long as 90 days after hospitalization; this finding has led to evaluation of the benefit of prolonged chemoprophylaxis.23 Extended-duration LMWH prophylaxis decreases the incidence of VTE, but at the cost of increased risk of major bleeding.47 Based on this evidence, guidelines recommend against prolonged-duration anticoagulation.23 A 2016 trial showed that 35 days of the direct-acting anticoagulant betrixaban reduced the risk of symptomatic VTE events, compared to 10 days of LMWH (NNT = 167), without increased risk of bleeding.48 This is a limited benefit, however, that is unlikely to change guideline recommendations.
Continue to: Trauma
Trauma: VTE risk increases with severity
Trauma increases the risk of VTE considerably. A national study showed that 1.5% of admitted trauma patients experienced VTE during hospitalization and that 1.2% were readmitted for VTE within 1 year.49 As many as 32% of trauma patients admitted to the intensive care unit experience VTE despite appropriate prophylaxis.50 A Cochrane Review51 found that:
- prophylaxis significantly reduces DVT risk
- pharmacotherapeutic prophylaxis is more effective than mechanical prophylaxis
- LMWH is more effective than LDUH.
Guidelines recommend that major trauma patients receive prophylaxis with LMWH, LDUH, or IPC.46
CORRESPONDENCE
Michael J. Arnold, MD, CDR, MC, USN; Uniformed Services University of the Health Sciences, 4301 Jones Bridge Road, Jacksonville, FL 32214; [email protected].
1. Beckman MG, Hooper WC, Critchley SE, et al. Venous thromboembolism: a public health concern. Am J Prev Med. 2010. 38(4 suppl):S495-S501.
2. Tagalakis V, Patenaude V, Kahn SR, et al. Incidence of and mortality from venous thromboembolism in a real-world population: the Q-VTE Study Cohort. Am J Med. 2013;126:832.e13-e21.
3. Yeh RW, Sidney S, Chandra M, et al. Population trends in the incidence and outcomes of acute myocardial infarction. N Engl J Med. 2010. 362:2155-2165.
4. Cushman M, Tsai AW, White RH, et al. Deep vein thrombosis and pulmonary embolism in two cohorts: the longitudinal investigation of thromboembolism etiology. Am J Med. 2004;117:19-25.
5. Goldhaber SZ. Venous thromboembolism: epidemiology and magnitude of the problem. Best Pract Res Clin Haematol. 2012;25:235-242.
6. Stone J, Hangge P, Albadawi H, et al. Deep vein thrombosis: pathogenesis, diagnosis, and medical management. Cardiovasc Diagn Ther. 2017;7(suppl 3):S276-S284.
7. Olaf M, Cooney R. Deep venous thrombosis. Emerg Med Clin North Am. 2017;35:743-770.
8. Sajid MS, Ahmed N, Desai M, et al. Upper limb deep vein thrombosis: a literature review to streamline the protocol for management. Acta Haematol. 2007;118:10-18.
9. Bates SM, Ginsberg JS. Clinical practice. Treatment of deep-vein thrombosis. N Engl J Med. 2004;351:268-277.
10. Chandra D, Parisini E, Mozaffarian D. Meta-analysis: travel and risk for venous thromboembolism. Ann Intern Med. 2009;151:180-190.
11. Goldhaber SZ. Risk factors for venous thromboembolism. J Am Col Cardiol. 2010;56:1-7.
12. Yang G, De Staercke C, Hooper WC. The effects of obesity on venous thromboembolism: a review. Open J Prev Med. 2012;2:499-509.
13. Severinsen MT, Kristensen SR, Johnsen SP, et al. Smoking and venous thromboembolism: a Danish follow-up study. J Thromb Haemost. 2009;7:1297-1303.
14. Coppens M, Reijnders JH, Middeldorp S, et al. Testing for inherited thrombophilia does not reduce the recurrence of venous thrombosis. J Thromb Haemost. 2008;6:1474-1477.
15. Choosing Wisely. American Society of Hematology. Ten things physicians and patients should question. www.choosingwisely.org/societies/american-society-of-hematology/. Accessed September 28, 2020.
16. Bates SM, Greer IA, Middeldorp S, et al. VTE, thrombophilia, antithrombotic therapy, and pregnancy: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e691S-e736S.
17. Vossen CY, Conard J, Fontcuberta J, et al. Risk of a first venous thrombotic event in carriers of a familial thrombophilic defect. The European Prospective Cohort on Thrombophilia (EPCOT). J Thromb Haemost. 2005;3:459-464.
18. Practice Bulletin No. 197: Inherited thrombophilias in pregnancy. Obstet Gynecol. 2018;132:e18-e34.
19. Committee on Practice Bulletins—Obstetrics, American College of Obstetricians and Gynecologists. Practice Bulletin No. 132: Antiphospholipid syndrome. Obstet Gynecol. 2012;120:1514-1521.
20. Glynn RJ, Danielson E, Fonseca FAH, et al. A randomized trial of rosuvastatin in the prevention of venous thromboembolism. N Engl J Med. 2009;360:1851-1861.
21. Taylor F, Huffman MD, Macedo AF, et al. Statins for the primary prevention of cardiovascular disease. Cochrane Database Syst Rev. 2013(1):CD004816.
22. Squizzato A, Galli M, Romualdi E, et al. Statins, fibrates, and venous thromboembolism: a meta-analysis. Eur Heart J. 2010;31:1248-1256.
23. Kahn SR, Lim W, Dunn AS, et al. Prevention of VTE in nonsurgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e195S-e226S.
24. Kelman CW, Kortt MA, Becker NG, et al. Deep vein thrombosis and air travel: record linkage study. BMJ. 2003;327:1072.
25. Johnston RV, Hudson MF; . Travelers’ thrombosis. Aviat Space Environ Med. 2014;85:191-194.
26. Clarke MJ, Broderick C, Hopewell S, et al. Compression stockings for preventing deep vein thrombosis in airline passengers. Cochrane Database Syst Rev. 2016;9:CD004002.
27. van Hylckama Vlieg A, Middledorp S. Hormone therapies and venous thromboembolism: where are we now? J Thromb Haemost. 2011;9:257-266.
28. Tepper NK, Dragoman MV, Gaffield ME, et al. Nonoral combined hormonal contraceptives and thromboembolism: a systematic review. Contraception. 2017;95:130-139.
29. Lekovic D, Miljic P, Dmitrovic A, et al. How do you decide on hormone replacement therapy in women with risk of venous thromboembolism? Blood Rev. 2017;31:151-157.
30. Rovinski D, Ramos RB, Fighera TM, et al. Risk of venous thromboembolism events in postmenopausal women using oral versus non-oral hormone therapy: a systematic review and meta-analysis. Thromb Res. 2018;168:83-95.
31. Horsted F, West J, Grainge MJ. Risk of venous thromboembolism in patients with cancer: a systematic review and meta-analysis. PLoS Med. 2012;9:e1001275.
32. Kamphuisen PW, Beyer-Westendorf J. Bleeding complications during anticoagulant treatment in patients with cancer. Thromb Res. 2014;133(suppl 2):S49-S55.
33. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST Guideline and Expert Panel Report. Chest. 2016;149:315-352.
34. Lyman GH, Khorana AA, Kuderer NM, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: American Society of Clinical Oncology Clinical Practice Guideline update. J Clin Oncol. 2013;31:2189-2204.
35. Khorana AA, Kuderer NM, Culakova E, et al. Development and validation of a predictive model for chemotherapy-associated thrombosis. Blood. 2008;111:4902-4907.
36. Ay C, Dunkler D, Marosi C, et al. Prediction of venous thromboembolism in cancer patients. Blood. 2010;116:5377-5382.
37. Carrier M, Abou-Nassar K, Mallick R, et al; AVERT Investigators. Apixaban to prevent venous thromboembolism in patients with cancer. N Engl J Med. 2019;380:711-719.
38. Cobben MRR, Nemeth B, Lijfering WM, et al. Validation of risk assessment models for venous thrombosis in hospitalized medical patients. Res Pract Thromb Haemost. 2019;3:217-225.
39. Caprini JA. Thrombosis risk assessment as a guide to quality patient care. Dis Mon. 2005;51:70-78.
40. Spyropoulos AC, Anderson FA Jr, FitzGerald G, et al; IMPROVE Investigators. Predictive and associative models to identify hospitalized medical patients at risk for VTE. Chest. 2011;140:706-714.
41. Kanaan AO, Silva MA, Donovan JL, et al. Meta-analysis of venous thromboembolism prophylaxis in medically Ill patients. Clin Ther. 2007;29:2395-2405.
42. HJ, Cushman M, Burnett AE, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: prophylaxis for hospitalized and nonhospitalized medical patients. Blood Adv. 2018;2:3198-3225.
43. Falck-Ytter Y, Francis CW, Johanson NA, et al. Prevention of VTE in orthopedic surgery patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e278S-e325S.
44. Pannucci CJ, Swistun L, MacDonald JK, et al. Individualized venous thromboembolism risk stratification using the 2005 Caprini Score to identify the benefits and harms of chemoprophylaxis in surgical patients: a meta-analysis. Ann Surg. 2017;265:1094-1103.
45. Matharu GS, Kunutsor SK, Judge A, et al. Clinical effectiveness and safety of aspirin for venous thromboembolism prophylaxis after total hip and knee replacement: a systematic review and meta-analysis of randomized clinical trials. JAMA Intern Med. 2020;180:376-384.
46. Gould MK, Garcia DA, Wren SM, et al. Prevention of VTE in nonorthopedic surgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e227S-e277S.
47. Hull RD, Schellong SM, Tapson VF, et al. Extended-duration venous thromboembolism prophylaxis in acutely ill medical patients with recent reduced mobility: a randomized trial. Ann Intern Med. 2010;153:8-18.
48. Cohen AT, Harrington RA, Goldhaber SZ, et al. Extended thromboprophylaxis with betrixaban in acutely ill medical patients. N Engl J Med. 2016;375:534-544.
49. Rattan R, Parreco J, Eidelson SA, et al. Hidden burden of venous thromboembolism after trauma: a national analysis. J Trauma Acute Care Surg. 2018;85:899-906.
50. Yumoto T, Naito H, Yamakawa Y, et al. Venous thromboembolism in major trauma patients: a single-center retrospective cohort study of the epidemiology and utility of D-dimer for screening. Acute Med Surg. 2017;4:394-400.
51. Barrera LM, Perel P, Ker K, et al. Thromboprophylaxis for trauma patients. Cochrane Database Syst Rev. 2013(3):CD008303.
1. Beckman MG, Hooper WC, Critchley SE, et al. Venous thromboembolism: a public health concern. Am J Prev Med. 2010. 38(4 suppl):S495-S501.
2. Tagalakis V, Patenaude V, Kahn SR, et al. Incidence of and mortality from venous thromboembolism in a real-world population: the Q-VTE Study Cohort. Am J Med. 2013;126:832.e13-e21.
3. Yeh RW, Sidney S, Chandra M, et al. Population trends in the incidence and outcomes of acute myocardial infarction. N Engl J Med. 2010. 362:2155-2165.
4. Cushman M, Tsai AW, White RH, et al. Deep vein thrombosis and pulmonary embolism in two cohorts: the longitudinal investigation of thromboembolism etiology. Am J Med. 2004;117:19-25.
5. Goldhaber SZ. Venous thromboembolism: epidemiology and magnitude of the problem. Best Pract Res Clin Haematol. 2012;25:235-242.
6. Stone J, Hangge P, Albadawi H, et al. Deep vein thrombosis: pathogenesis, diagnosis, and medical management. Cardiovasc Diagn Ther. 2017;7(suppl 3):S276-S284.
7. Olaf M, Cooney R. Deep venous thrombosis. Emerg Med Clin North Am. 2017;35:743-770.
8. Sajid MS, Ahmed N, Desai M, et al. Upper limb deep vein thrombosis: a literature review to streamline the protocol for management. Acta Haematol. 2007;118:10-18.
9. Bates SM, Ginsberg JS. Clinical practice. Treatment of deep-vein thrombosis. N Engl J Med. 2004;351:268-277.
10. Chandra D, Parisini E, Mozaffarian D. Meta-analysis: travel and risk for venous thromboembolism. Ann Intern Med. 2009;151:180-190.
11. Goldhaber SZ. Risk factors for venous thromboembolism. J Am Col Cardiol. 2010;56:1-7.
12. Yang G, De Staercke C, Hooper WC. The effects of obesity on venous thromboembolism: a review. Open J Prev Med. 2012;2:499-509.
13. Severinsen MT, Kristensen SR, Johnsen SP, et al. Smoking and venous thromboembolism: a Danish follow-up study. J Thromb Haemost. 2009;7:1297-1303.
14. Coppens M, Reijnders JH, Middeldorp S, et al. Testing for inherited thrombophilia does not reduce the recurrence of venous thrombosis. J Thromb Haemost. 2008;6:1474-1477.
15. Choosing Wisely. American Society of Hematology. Ten things physicians and patients should question. www.choosingwisely.org/societies/american-society-of-hematology/. Accessed September 28, 2020.
16. Bates SM, Greer IA, Middeldorp S, et al. VTE, thrombophilia, antithrombotic therapy, and pregnancy: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e691S-e736S.
17. Vossen CY, Conard J, Fontcuberta J, et al. Risk of a first venous thrombotic event in carriers of a familial thrombophilic defect. The European Prospective Cohort on Thrombophilia (EPCOT). J Thromb Haemost. 2005;3:459-464.
18. Practice Bulletin No. 197: Inherited thrombophilias in pregnancy. Obstet Gynecol. 2018;132:e18-e34.
19. Committee on Practice Bulletins—Obstetrics, American College of Obstetricians and Gynecologists. Practice Bulletin No. 132: Antiphospholipid syndrome. Obstet Gynecol. 2012;120:1514-1521.
20. Glynn RJ, Danielson E, Fonseca FAH, et al. A randomized trial of rosuvastatin in the prevention of venous thromboembolism. N Engl J Med. 2009;360:1851-1861.
21. Taylor F, Huffman MD, Macedo AF, et al. Statins for the primary prevention of cardiovascular disease. Cochrane Database Syst Rev. 2013(1):CD004816.
22. Squizzato A, Galli M, Romualdi E, et al. Statins, fibrates, and venous thromboembolism: a meta-analysis. Eur Heart J. 2010;31:1248-1256.
23. Kahn SR, Lim W, Dunn AS, et al. Prevention of VTE in nonsurgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e195S-e226S.
24. Kelman CW, Kortt MA, Becker NG, et al. Deep vein thrombosis and air travel: record linkage study. BMJ. 2003;327:1072.
25. Johnston RV, Hudson MF; . Travelers’ thrombosis. Aviat Space Environ Med. 2014;85:191-194.
26. Clarke MJ, Broderick C, Hopewell S, et al. Compression stockings for preventing deep vein thrombosis in airline passengers. Cochrane Database Syst Rev. 2016;9:CD004002.
27. van Hylckama Vlieg A, Middledorp S. Hormone therapies and venous thromboembolism: where are we now? J Thromb Haemost. 2011;9:257-266.
28. Tepper NK, Dragoman MV, Gaffield ME, et al. Nonoral combined hormonal contraceptives and thromboembolism: a systematic review. Contraception. 2017;95:130-139.
29. Lekovic D, Miljic P, Dmitrovic A, et al. How do you decide on hormone replacement therapy in women with risk of venous thromboembolism? Blood Rev. 2017;31:151-157.
30. Rovinski D, Ramos RB, Fighera TM, et al. Risk of venous thromboembolism events in postmenopausal women using oral versus non-oral hormone therapy: a systematic review and meta-analysis. Thromb Res. 2018;168:83-95.
31. Horsted F, West J, Grainge MJ. Risk of venous thromboembolism in patients with cancer: a systematic review and meta-analysis. PLoS Med. 2012;9:e1001275.
32. Kamphuisen PW, Beyer-Westendorf J. Bleeding complications during anticoagulant treatment in patients with cancer. Thromb Res. 2014;133(suppl 2):S49-S55.
33. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST Guideline and Expert Panel Report. Chest. 2016;149:315-352.
34. Lyman GH, Khorana AA, Kuderer NM, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: American Society of Clinical Oncology Clinical Practice Guideline update. J Clin Oncol. 2013;31:2189-2204.
35. Khorana AA, Kuderer NM, Culakova E, et al. Development and validation of a predictive model for chemotherapy-associated thrombosis. Blood. 2008;111:4902-4907.
36. Ay C, Dunkler D, Marosi C, et al. Prediction of venous thromboembolism in cancer patients. Blood. 2010;116:5377-5382.
37. Carrier M, Abou-Nassar K, Mallick R, et al; AVERT Investigators. Apixaban to prevent venous thromboembolism in patients with cancer. N Engl J Med. 2019;380:711-719.
38. Cobben MRR, Nemeth B, Lijfering WM, et al. Validation of risk assessment models for venous thrombosis in hospitalized medical patients. Res Pract Thromb Haemost. 2019;3:217-225.
39. Caprini JA. Thrombosis risk assessment as a guide to quality patient care. Dis Mon. 2005;51:70-78.
40. Spyropoulos AC, Anderson FA Jr, FitzGerald G, et al; IMPROVE Investigators. Predictive and associative models to identify hospitalized medical patients at risk for VTE. Chest. 2011;140:706-714.
41. Kanaan AO, Silva MA, Donovan JL, et al. Meta-analysis of venous thromboembolism prophylaxis in medically Ill patients. Clin Ther. 2007;29:2395-2405.
42. HJ, Cushman M, Burnett AE, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: prophylaxis for hospitalized and nonhospitalized medical patients. Blood Adv. 2018;2:3198-3225.
43. Falck-Ytter Y, Francis CW, Johanson NA, et al. Prevention of VTE in orthopedic surgery patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e278S-e325S.
44. Pannucci CJ, Swistun L, MacDonald JK, et al. Individualized venous thromboembolism risk stratification using the 2005 Caprini Score to identify the benefits and harms of chemoprophylaxis in surgical patients: a meta-analysis. Ann Surg. 2017;265:1094-1103.
45. Matharu GS, Kunutsor SK, Judge A, et al. Clinical effectiveness and safety of aspirin for venous thromboembolism prophylaxis after total hip and knee replacement: a systematic review and meta-analysis of randomized clinical trials. JAMA Intern Med. 2020;180:376-384.
46. Gould MK, Garcia DA, Wren SM, et al. Prevention of VTE in nonorthopedic surgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 suppl):e227S-e277S.
47. Hull RD, Schellong SM, Tapson VF, et al. Extended-duration venous thromboembolism prophylaxis in acutely ill medical patients with recent reduced mobility: a randomized trial. Ann Intern Med. 2010;153:8-18.
48. Cohen AT, Harrington RA, Goldhaber SZ, et al. Extended thromboprophylaxis with betrixaban in acutely ill medical patients. N Engl J Med. 2016;375:534-544.
49. Rattan R, Parreco J, Eidelson SA, et al. Hidden burden of venous thromboembolism after trauma: a national analysis. J Trauma Acute Care Surg. 2018;85:899-906.
50. Yumoto T, Naito H, Yamakawa Y, et al. Venous thromboembolism in major trauma patients: a single-center retrospective cohort study of the epidemiology and utility of D-dimer for screening. Acute Med Surg. 2017;4:394-400.
51. Barrera LM, Perel P, Ker K, et al. Thromboprophylaxis for trauma patients. Cochrane Database Syst Rev. 2013(3):CD008303.
PRACTICE RECOMMENDATIONS
› Consider the mild reduction in the risk of venous thromboembolism (VTE) provided by statins when contemplating their use for cardiovascular disease prevention. B
› Avoid testing for thrombophilia to determine the risk of VTE, except in pregnant patients who meet criteria for antiphospholipid syndrome or have a family history of VTE. B
› Recommend an intrauterine device or progestin-only pill for contraception if the patient’s risk of VTE is high. B
› Stratify hospitalized medical and nonorthopedic surgical patients by risk score to determine the need for VTE prophylaxis. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Adrenal vein sampling looms as choke point for aldosteronism assessment of hypertensives
At a time when new evidence strongly suggests that roughly a fifth of patents with hypertension have primary aldosteronism as the cause, other recent findings suggest that many of these possibly tens of millions of patients with aldosterone-driven high blood pressure may as a consequence need an expensive and not-widely-available diagnostic test – adrenal vein sampling – to determine whether they are candidates for a definitive surgical cure to their aldosteronism.
Some endocrinologists worry the worldwide infrastructure for running adrenal vein sampling (AVS) isn’t close to being in place to deliver on this looming need for patients with primary aldosteronism (PA), especially given the burgeoning numbers now being cited for PA prevalence.
“The system could be overwhelmed,” warned Robert M. Carey, MD, a cardiovascular endocrinologist and professor of medicine at the University of Virginia in Charlottesville. “Right now, adrenal vein sampling [AVS] is the gold standard,” for distinguishing unilateral and bilateral excess aldosterone secretion, “but not every radiologist can do AVS. Until we find a surrogate biomarker that can distinguish unilateral and bilateral PA” many patients will need AVS, Dr. Carey said in an interview.
“AVS is important for accurate lateralization of aldosterone excess in patients, but it may not be feasible for all patients with PA to undergo AVS. If the prevalence of PA truly is on the order of 15% [of all patients with hypertension] then health systems would be stretched to offer all of them AVS, which is technically challenging and requires dedicated training and is therefore limited to expert centers,” commented Jun Yang, MBBS, a cardiovascular endocrinologist at the Hudson Institute of Medical Research and a hypertension researcher at Monash University, both in Melbourne. “At Monash, our interventional radiologists have increased their [AVS] success rate from 40% to more than 90% during the past 10 years, and our waiting list for patients scheduled for AVS is now 3-4 months long,” Dr. Yang said in an interview.
Finding a unilateral adrenal nodule as the cause of PA means that surgical removal is an option, a step that often fully resolves the PA and normalizes blood pressure. Patients with a bilateral source of the aldosterone are not candidates for surgical cure and must be managed with medical treatment, usually a mineralocorticoid receptor antagonist such as spironolactone that can neutralize or at least reduce the impact of hyperaldosteronism.
AVS finds unilateral adenomas when imaging can’t
The evidence that raised concerns about the reliability of imaging as an easier and noninvasive means to identify hypertensive patients with PA and a unilateral adrenal nodule that makes them candidates for surgical removal to resolve their PA and hypertension came out in May 2020 in a review of 174 PA patients who underwent AVS at a single center in Calgary, Alta., during 2006-2018.
The review included 366 patients with PA referred to the University of Calgary for assessment, of whom 179 had no adrenal nodule visible with either CT or MRI imaging, with 174 of these patients also undergoing successful AVS. The procedure revealed 70 patients (40%) had unilateral aldosterone secretion (Can J Cardiol. 2020 May 16. doi: 10.1016/j.cjca.2020.05.013).
In an editorial about this report that appeared a few weeks later, Ross D. Feldman, MD, a hypertension-management researcher and professor of medicine at the University of Manitoba in Winnipeg, Man., said the finding was “amazing,” and “confirms that lateralization of aldosterone secretion in a patient with PA but without an identifiable mass on that side is not a zebra,” but instead a presentation that “occurs in almost half of patients with PA and no discernible adenoma on the side that lateralizes.” (Can J. Cardiol. 2020 Jul 3. doi: 10.1016/j.cjca.2020.06.022).
Although this was just one center’s experience, the authors are not alone in making this finding, although prior reports seem to have been largely forgotten or ignored until now.
“The discordance between AVS and adrenal imaging has been documented by numerous groups, and in our own experience [in Melbourne] around 40% of patients with unilateral aldosterone excess do not have a distinct unilateral adenoma on CT,” said Dr. Yang.
“Here’s the problem,” summed up Dr. Feldman in an interview. “Nearly half of patients with hyperaldosteronism don’t localize based on a CT or MRI, so you have to do AVS, but AVS is not generally available; it’s only at tertiary centers; and you have to do a lot of them,” to do them well. “It’s a half-day procedure, and you have to hit the correct adrenal vein.”
AVS for millions?
Compounding the challenge is the other bit of bombshell news recently dropped on the endocrinology and hypertension communities: PA may be much more prevalent that previously suspected, occurring in roughly 20% of patients with hypertension, according to study results that also came out in 2020 (Ann Int Med. 2020 Jul 7;173[1]:10-20).
The upshot, according to Dr. Feldman and others, is that researchers will need to find reliable criteria besides imaging for identifying PA patients with an increased likelihood of having a lateralized source for their excess aldosterone production. That’s “the only hope,” said Dr. Feldman, “so we won’t have to do AVS on 20 million Americans.”
Unfortunately, the path toward a successful screen to winnow down candidates for AVS has been long and not especially fruitful, with efforts dating back at least 50 years, and with one of the most recent efforts at stratifying PA patients by certain laboratory measures getting dismissed as producing a benefit that “might not be substantial,” wrote Michael Stowasser, MBBS, in a published commentary (J Hypertension. 2020 Jul;38[7]:1259-61).
In contrast to Dr. Feldman, Dr. Stowasser was more optimistic about the prospects for avoiding an immediate crisis in AVS assessment of PA patients, mostly because so few patients with PA are now identified by clinicians. Given the poor record clinicians have historically rung up diagnosing PA, “it would seem unlikely that we are going to be flooded with AVS requests any time soon,” he wrote. There is also reason to hope that increased demand for AVS will help broaden availability, and innovative testing methods promise to speed up the procedure, said Dr. Stowasser, a professor of medicine at the University of Queensland in Brisbane, Australia and director of the Endocrine Hypertension Research Centre at Greenslopes and Princess Alexandra Hospitals in Brisbane, in an interview.
But regardless of whether AVS testing becomes more available or streamlined, recent events suggest there will be little way to avoid eventually having to run millions of these diagnostic procedures.
Patients with PA “who decide they will not want surgery do not need AVS. For all other patients with PA, you need AVS. The medical system will just have to respond,” Dr. Carey concluded.
Dr. Carey, Dr. Yang, Dr. Feldman, and Dr. Stowasser had no relevant disclosures.
At a time when new evidence strongly suggests that roughly a fifth of patents with hypertension have primary aldosteronism as the cause, other recent findings suggest that many of these possibly tens of millions of patients with aldosterone-driven high blood pressure may as a consequence need an expensive and not-widely-available diagnostic test – adrenal vein sampling – to determine whether they are candidates for a definitive surgical cure to their aldosteronism.
Some endocrinologists worry the worldwide infrastructure for running adrenal vein sampling (AVS) isn’t close to being in place to deliver on this looming need for patients with primary aldosteronism (PA), especially given the burgeoning numbers now being cited for PA prevalence.
“The system could be overwhelmed,” warned Robert M. Carey, MD, a cardiovascular endocrinologist and professor of medicine at the University of Virginia in Charlottesville. “Right now, adrenal vein sampling [AVS] is the gold standard,” for distinguishing unilateral and bilateral excess aldosterone secretion, “but not every radiologist can do AVS. Until we find a surrogate biomarker that can distinguish unilateral and bilateral PA” many patients will need AVS, Dr. Carey said in an interview.
“AVS is important for accurate lateralization of aldosterone excess in patients, but it may not be feasible for all patients with PA to undergo AVS. If the prevalence of PA truly is on the order of 15% [of all patients with hypertension] then health systems would be stretched to offer all of them AVS, which is technically challenging and requires dedicated training and is therefore limited to expert centers,” commented Jun Yang, MBBS, a cardiovascular endocrinologist at the Hudson Institute of Medical Research and a hypertension researcher at Monash University, both in Melbourne. “At Monash, our interventional radiologists have increased their [AVS] success rate from 40% to more than 90% during the past 10 years, and our waiting list for patients scheduled for AVS is now 3-4 months long,” Dr. Yang said in an interview.
Finding a unilateral adrenal nodule as the cause of PA means that surgical removal is an option, a step that often fully resolves the PA and normalizes blood pressure. Patients with a bilateral source of the aldosterone are not candidates for surgical cure and must be managed with medical treatment, usually a mineralocorticoid receptor antagonist such as spironolactone that can neutralize or at least reduce the impact of hyperaldosteronism.
AVS finds unilateral adenomas when imaging can’t
The evidence that raised concerns about the reliability of imaging as an easier and noninvasive means to identify hypertensive patients with PA and a unilateral adrenal nodule that makes them candidates for surgical removal to resolve their PA and hypertension came out in May 2020 in a review of 174 PA patients who underwent AVS at a single center in Calgary, Alta., during 2006-2018.
The review included 366 patients with PA referred to the University of Calgary for assessment, of whom 179 had no adrenal nodule visible with either CT or MRI imaging, with 174 of these patients also undergoing successful AVS. The procedure revealed 70 patients (40%) had unilateral aldosterone secretion (Can J Cardiol. 2020 May 16. doi: 10.1016/j.cjca.2020.05.013).
In an editorial about this report that appeared a few weeks later, Ross D. Feldman, MD, a hypertension-management researcher and professor of medicine at the University of Manitoba in Winnipeg, Man., said the finding was “amazing,” and “confirms that lateralization of aldosterone secretion in a patient with PA but without an identifiable mass on that side is not a zebra,” but instead a presentation that “occurs in almost half of patients with PA and no discernible adenoma on the side that lateralizes.” (Can J. Cardiol. 2020 Jul 3. doi: 10.1016/j.cjca.2020.06.022).
Although this was just one center’s experience, the authors are not alone in making this finding, although prior reports seem to have been largely forgotten or ignored until now.
“The discordance between AVS and adrenal imaging has been documented by numerous groups, and in our own experience [in Melbourne] around 40% of patients with unilateral aldosterone excess do not have a distinct unilateral adenoma on CT,” said Dr. Yang.
“Here’s the problem,” summed up Dr. Feldman in an interview. “Nearly half of patients with hyperaldosteronism don’t localize based on a CT or MRI, so you have to do AVS, but AVS is not generally available; it’s only at tertiary centers; and you have to do a lot of them,” to do them well. “It’s a half-day procedure, and you have to hit the correct adrenal vein.”
AVS for millions?
Compounding the challenge is the other bit of bombshell news recently dropped on the endocrinology and hypertension communities: PA may be much more prevalent that previously suspected, occurring in roughly 20% of patients with hypertension, according to study results that also came out in 2020 (Ann Int Med. 2020 Jul 7;173[1]:10-20).
The upshot, according to Dr. Feldman and others, is that researchers will need to find reliable criteria besides imaging for identifying PA patients with an increased likelihood of having a lateralized source for their excess aldosterone production. That’s “the only hope,” said Dr. Feldman, “so we won’t have to do AVS on 20 million Americans.”
Unfortunately, the path toward a successful screen to winnow down candidates for AVS has been long and not especially fruitful, with efforts dating back at least 50 years, and with one of the most recent efforts at stratifying PA patients by certain laboratory measures getting dismissed as producing a benefit that “might not be substantial,” wrote Michael Stowasser, MBBS, in a published commentary (J Hypertension. 2020 Jul;38[7]:1259-61).
In contrast to Dr. Feldman, Dr. Stowasser was more optimistic about the prospects for avoiding an immediate crisis in AVS assessment of PA patients, mostly because so few patients with PA are now identified by clinicians. Given the poor record clinicians have historically rung up diagnosing PA, “it would seem unlikely that we are going to be flooded with AVS requests any time soon,” he wrote. There is also reason to hope that increased demand for AVS will help broaden availability, and innovative testing methods promise to speed up the procedure, said Dr. Stowasser, a professor of medicine at the University of Queensland in Brisbane, Australia and director of the Endocrine Hypertension Research Centre at Greenslopes and Princess Alexandra Hospitals in Brisbane, in an interview.
But regardless of whether AVS testing becomes more available or streamlined, recent events suggest there will be little way to avoid eventually having to run millions of these diagnostic procedures.
Patients with PA “who decide they will not want surgery do not need AVS. For all other patients with PA, you need AVS. The medical system will just have to respond,” Dr. Carey concluded.
Dr. Carey, Dr. Yang, Dr. Feldman, and Dr. Stowasser had no relevant disclosures.
At a time when new evidence strongly suggests that roughly a fifth of patents with hypertension have primary aldosteronism as the cause, other recent findings suggest that many of these possibly tens of millions of patients with aldosterone-driven high blood pressure may as a consequence need an expensive and not-widely-available diagnostic test – adrenal vein sampling – to determine whether they are candidates for a definitive surgical cure to their aldosteronism.
Some endocrinologists worry the worldwide infrastructure for running adrenal vein sampling (AVS) isn’t close to being in place to deliver on this looming need for patients with primary aldosteronism (PA), especially given the burgeoning numbers now being cited for PA prevalence.
“The system could be overwhelmed,” warned Robert M. Carey, MD, a cardiovascular endocrinologist and professor of medicine at the University of Virginia in Charlottesville. “Right now, adrenal vein sampling [AVS] is the gold standard,” for distinguishing unilateral and bilateral excess aldosterone secretion, “but not every radiologist can do AVS. Until we find a surrogate biomarker that can distinguish unilateral and bilateral PA” many patients will need AVS, Dr. Carey said in an interview.
“AVS is important for accurate lateralization of aldosterone excess in patients, but it may not be feasible for all patients with PA to undergo AVS. If the prevalence of PA truly is on the order of 15% [of all patients with hypertension] then health systems would be stretched to offer all of them AVS, which is technically challenging and requires dedicated training and is therefore limited to expert centers,” commented Jun Yang, MBBS, a cardiovascular endocrinologist at the Hudson Institute of Medical Research and a hypertension researcher at Monash University, both in Melbourne. “At Monash, our interventional radiologists have increased their [AVS] success rate from 40% to more than 90% during the past 10 years, and our waiting list for patients scheduled for AVS is now 3-4 months long,” Dr. Yang said in an interview.
Finding a unilateral adrenal nodule as the cause of PA means that surgical removal is an option, a step that often fully resolves the PA and normalizes blood pressure. Patients with a bilateral source of the aldosterone are not candidates for surgical cure and must be managed with medical treatment, usually a mineralocorticoid receptor antagonist such as spironolactone that can neutralize or at least reduce the impact of hyperaldosteronism.
AVS finds unilateral adenomas when imaging can’t
The evidence that raised concerns about the reliability of imaging as an easier and noninvasive means to identify hypertensive patients with PA and a unilateral adrenal nodule that makes them candidates for surgical removal to resolve their PA and hypertension came out in May 2020 in a review of 174 PA patients who underwent AVS at a single center in Calgary, Alta., during 2006-2018.
The review included 366 patients with PA referred to the University of Calgary for assessment, of whom 179 had no adrenal nodule visible with either CT or MRI imaging, with 174 of these patients also undergoing successful AVS. The procedure revealed 70 patients (40%) had unilateral aldosterone secretion (Can J Cardiol. 2020 May 16. doi: 10.1016/j.cjca.2020.05.013).
In an editorial about this report that appeared a few weeks later, Ross D. Feldman, MD, a hypertension-management researcher and professor of medicine at the University of Manitoba in Winnipeg, Man., said the finding was “amazing,” and “confirms that lateralization of aldosterone secretion in a patient with PA but without an identifiable mass on that side is not a zebra,” but instead a presentation that “occurs in almost half of patients with PA and no discernible adenoma on the side that lateralizes.” (Can J. Cardiol. 2020 Jul 3. doi: 10.1016/j.cjca.2020.06.022).
Although this was just one center’s experience, the authors are not alone in making this finding, although prior reports seem to have been largely forgotten or ignored until now.
“The discordance between AVS and adrenal imaging has been documented by numerous groups, and in our own experience [in Melbourne] around 40% of patients with unilateral aldosterone excess do not have a distinct unilateral adenoma on CT,” said Dr. Yang.
“Here’s the problem,” summed up Dr. Feldman in an interview. “Nearly half of patients with hyperaldosteronism don’t localize based on a CT or MRI, so you have to do AVS, but AVS is not generally available; it’s only at tertiary centers; and you have to do a lot of them,” to do them well. “It’s a half-day procedure, and you have to hit the correct adrenal vein.”
AVS for millions?
Compounding the challenge is the other bit of bombshell news recently dropped on the endocrinology and hypertension communities: PA may be much more prevalent that previously suspected, occurring in roughly 20% of patients with hypertension, according to study results that also came out in 2020 (Ann Int Med. 2020 Jul 7;173[1]:10-20).
The upshot, according to Dr. Feldman and others, is that researchers will need to find reliable criteria besides imaging for identifying PA patients with an increased likelihood of having a lateralized source for their excess aldosterone production. That’s “the only hope,” said Dr. Feldman, “so we won’t have to do AVS on 20 million Americans.”
Unfortunately, the path toward a successful screen to winnow down candidates for AVS has been long and not especially fruitful, with efforts dating back at least 50 years, and with one of the most recent efforts at stratifying PA patients by certain laboratory measures getting dismissed as producing a benefit that “might not be substantial,” wrote Michael Stowasser, MBBS, in a published commentary (J Hypertension. 2020 Jul;38[7]:1259-61).
In contrast to Dr. Feldman, Dr. Stowasser was more optimistic about the prospects for avoiding an immediate crisis in AVS assessment of PA patients, mostly because so few patients with PA are now identified by clinicians. Given the poor record clinicians have historically rung up diagnosing PA, “it would seem unlikely that we are going to be flooded with AVS requests any time soon,” he wrote. There is also reason to hope that increased demand for AVS will help broaden availability, and innovative testing methods promise to speed up the procedure, said Dr. Stowasser, a professor of medicine at the University of Queensland in Brisbane, Australia and director of the Endocrine Hypertension Research Centre at Greenslopes and Princess Alexandra Hospitals in Brisbane, in an interview.
But regardless of whether AVS testing becomes more available or streamlined, recent events suggest there will be little way to avoid eventually having to run millions of these diagnostic procedures.
Patients with PA “who decide they will not want surgery do not need AVS. For all other patients with PA, you need AVS. The medical system will just have to respond,” Dr. Carey concluded.
Dr. Carey, Dr. Yang, Dr. Feldman, and Dr. Stowasser had no relevant disclosures.