User login
FDA approves Zephyr endobronchial valve to treat severe emphysema
The valve is the first minimally invasive device approved in the United States for treating such patients, according to Pulmonx, the device manufacturer.
The FDA previously granted the novel device expedited review, as patients who did not respond to drug treatment had only limited alternative options, including lung volume reduction and lung transplant, Tina Kiang, PhD, of the FDA’s Center for Devices and Radiological Health, said in a press release. “This novel device is a less invasive treatment that expands the options available to patients,” said Dr. Kiang, acting director of the center’s Division of Anesthesiology, General Hospital, Respiratory, Infection Control, and Dental Devices.
The approval is based on a multicenter study of 190 patients with severe emphysema. A total of 128 received Zephyr valves and medical management, while 62 received medical management only. The primary measure was the number of patients who achieved at least a 15% improvement in their pulmonary function score: At 1 year, 47.7% of the Zephyr valve patients had achieved such improvement versus 16.8% of the control group, according to the FDA.
Adverse events included death, pneumothorax, pneumonia, worsening of emphysema, coughing up blood, shortness of breath, and chest pain. The valve is contraindicated in patients with active lung infections; those allergic to nitinol, nickel, titanium, or silicone; and active smokers.
Read more about this approval in the full FDA press announcement.
The valve is the first minimally invasive device approved in the United States for treating such patients, according to Pulmonx, the device manufacturer.
The FDA previously granted the novel device expedited review, as patients who did not respond to drug treatment had only limited alternative options, including lung volume reduction and lung transplant, Tina Kiang, PhD, of the FDA’s Center for Devices and Radiological Health, said in a press release. “This novel device is a less invasive treatment that expands the options available to patients,” said Dr. Kiang, acting director of the center’s Division of Anesthesiology, General Hospital, Respiratory, Infection Control, and Dental Devices.
The approval is based on a multicenter study of 190 patients with severe emphysema. A total of 128 received Zephyr valves and medical management, while 62 received medical management only. The primary measure was the number of patients who achieved at least a 15% improvement in their pulmonary function score: At 1 year, 47.7% of the Zephyr valve patients had achieved such improvement versus 16.8% of the control group, according to the FDA.
Adverse events included death, pneumothorax, pneumonia, worsening of emphysema, coughing up blood, shortness of breath, and chest pain. The valve is contraindicated in patients with active lung infections; those allergic to nitinol, nickel, titanium, or silicone; and active smokers.
Read more about this approval in the full FDA press announcement.
The valve is the first minimally invasive device approved in the United States for treating such patients, according to Pulmonx, the device manufacturer.
The FDA previously granted the novel device expedited review, as patients who did not respond to drug treatment had only limited alternative options, including lung volume reduction and lung transplant, Tina Kiang, PhD, of the FDA’s Center for Devices and Radiological Health, said in a press release. “This novel device is a less invasive treatment that expands the options available to patients,” said Dr. Kiang, acting director of the center’s Division of Anesthesiology, General Hospital, Respiratory, Infection Control, and Dental Devices.
The approval is based on a multicenter study of 190 patients with severe emphysema. A total of 128 received Zephyr valves and medical management, while 62 received medical management only. The primary measure was the number of patients who achieved at least a 15% improvement in their pulmonary function score: At 1 year, 47.7% of the Zephyr valve patients had achieved such improvement versus 16.8% of the control group, according to the FDA.
Adverse events included death, pneumothorax, pneumonia, worsening of emphysema, coughing up blood, shortness of breath, and chest pain. The valve is contraindicated in patients with active lung infections; those allergic to nitinol, nickel, titanium, or silicone; and active smokers.
Read more about this approval in the full FDA press announcement.
Customized airway stents show promise in feasibility trial
SAN DIEGO – for whom conventional stents were not suitable or failed, results from a small study demonstrated.
“Anatomically complex airway stenosis remains a challenging situation,” lead study author Nicolas Guibert, MD, said at an international conference of the American Thoracic Society. “Conventional devices are either not suited or may result in a significant complication rate, including poor clinical tolerance, migration, or granulation tissue reaction due to lack of congruence.”
Dr. Guibert reported results from eight patients. Of these, three had posttransplant complex airway stenoses involving the bronchus intermedius. Each improved after placement of the customized stents. For example, one patient with vanishing bronchus intermedius syndrome experienced improvements in NYHA dyspnea score from 3 to 1, the VQ11 score from 22 to 11/55, and forced expiratory volume in 1 second (FEV1) from 70% to 107%. The stent was removed after 3 months. Meanwhile, a patient with localized malacia and stenosis of the right main bronchus experienced improvements in NYHA dyspnea score from 3 to 1, VQ11 score from 27 to 15/55, and FEV1 from 70% to 102%. That person’s stent is still in place with no complications. Another patient with localized malacia and stenosis of the bronchus intermedius experienced improvements in FEV1 from 84% to 100%. That person’s device was removed after 3 months, with no residual stenosis.
A fourth patient underwent stent placement for localized malacia (cartilage ring rupture). That person experienced improvements in NYHA dyspnea score from 3 to 1, VQ11 from 23 to 15/55, and FEV1 from 66% to 92%, and peak flow from 49% to 82%. The device is still in place with no complications. A fifth patient received stent placement for extensive tracheobronchomalacia, but it had imperfect congruence and was removed after 3 months because it caused intense cough.
One patient with post-tracheotomy stenosis experienced improvements in NYHA dyspnea score from 3 to 0, VQ11 from 29 to 12/55, and peak flow from 45% to 81%. That person’s device is still in place, Dr. Guibert said. Two other patients treated for post-tracheotomy experienced stent migration (conventional stents also migrated in these two cases), despite good bronchoscopic congruence after placement.
“Tracheal diseases result in suboptimal congruence, probably due to higher respiratory variation,” Dr. Guibert said. “These devices need to be studied in less selected populations and the technology has to be improved.” He reported having no financial disclosures.
SOURCE: Guibert N et al. ATS 2018, Abstract 4433.
SAN DIEGO – for whom conventional stents were not suitable or failed, results from a small study demonstrated.
“Anatomically complex airway stenosis remains a challenging situation,” lead study author Nicolas Guibert, MD, said at an international conference of the American Thoracic Society. “Conventional devices are either not suited or may result in a significant complication rate, including poor clinical tolerance, migration, or granulation tissue reaction due to lack of congruence.”
Dr. Guibert reported results from eight patients. Of these, three had posttransplant complex airway stenoses involving the bronchus intermedius. Each improved after placement of the customized stents. For example, one patient with vanishing bronchus intermedius syndrome experienced improvements in NYHA dyspnea score from 3 to 1, the VQ11 score from 22 to 11/55, and forced expiratory volume in 1 second (FEV1) from 70% to 107%. The stent was removed after 3 months. Meanwhile, a patient with localized malacia and stenosis of the right main bronchus experienced improvements in NYHA dyspnea score from 3 to 1, VQ11 score from 27 to 15/55, and FEV1 from 70% to 102%. That person’s stent is still in place with no complications. Another patient with localized malacia and stenosis of the bronchus intermedius experienced improvements in FEV1 from 84% to 100%. That person’s device was removed after 3 months, with no residual stenosis.
A fourth patient underwent stent placement for localized malacia (cartilage ring rupture). That person experienced improvements in NYHA dyspnea score from 3 to 1, VQ11 from 23 to 15/55, and FEV1 from 66% to 92%, and peak flow from 49% to 82%. The device is still in place with no complications. A fifth patient received stent placement for extensive tracheobronchomalacia, but it had imperfect congruence and was removed after 3 months because it caused intense cough.
One patient with post-tracheotomy stenosis experienced improvements in NYHA dyspnea score from 3 to 0, VQ11 from 29 to 12/55, and peak flow from 45% to 81%. That person’s device is still in place, Dr. Guibert said. Two other patients treated for post-tracheotomy experienced stent migration (conventional stents also migrated in these two cases), despite good bronchoscopic congruence after placement.
“Tracheal diseases result in suboptimal congruence, probably due to higher respiratory variation,” Dr. Guibert said. “These devices need to be studied in less selected populations and the technology has to be improved.” He reported having no financial disclosures.
SOURCE: Guibert N et al. ATS 2018, Abstract 4433.
SAN DIEGO – for whom conventional stents were not suitable or failed, results from a small study demonstrated.
“Anatomically complex airway stenosis remains a challenging situation,” lead study author Nicolas Guibert, MD, said at an international conference of the American Thoracic Society. “Conventional devices are either not suited or may result in a significant complication rate, including poor clinical tolerance, migration, or granulation tissue reaction due to lack of congruence.”
Dr. Guibert reported results from eight patients. Of these, three had posttransplant complex airway stenoses involving the bronchus intermedius. Each improved after placement of the customized stents. For example, one patient with vanishing bronchus intermedius syndrome experienced improvements in NYHA dyspnea score from 3 to 1, the VQ11 score from 22 to 11/55, and forced expiratory volume in 1 second (FEV1) from 70% to 107%. The stent was removed after 3 months. Meanwhile, a patient with localized malacia and stenosis of the right main bronchus experienced improvements in NYHA dyspnea score from 3 to 1, VQ11 score from 27 to 15/55, and FEV1 from 70% to 102%. That person’s stent is still in place with no complications. Another patient with localized malacia and stenosis of the bronchus intermedius experienced improvements in FEV1 from 84% to 100%. That person’s device was removed after 3 months, with no residual stenosis.
A fourth patient underwent stent placement for localized malacia (cartilage ring rupture). That person experienced improvements in NYHA dyspnea score from 3 to 1, VQ11 from 23 to 15/55, and FEV1 from 66% to 92%, and peak flow from 49% to 82%. The device is still in place with no complications. A fifth patient received stent placement for extensive tracheobronchomalacia, but it had imperfect congruence and was removed after 3 months because it caused intense cough.
One patient with post-tracheotomy stenosis experienced improvements in NYHA dyspnea score from 3 to 0, VQ11 from 29 to 12/55, and peak flow from 45% to 81%. That person’s device is still in place, Dr. Guibert said. Two other patients treated for post-tracheotomy experienced stent migration (conventional stents also migrated in these two cases), despite good bronchoscopic congruence after placement.
“Tracheal diseases result in suboptimal congruence, probably due to higher respiratory variation,” Dr. Guibert said. “These devices need to be studied in less selected populations and the technology has to be improved.” He reported having no financial disclosures.
SOURCE: Guibert N et al. ATS 2018, Abstract 4433.
REPORTING FROM ATS 2018
Key clinical point: Customized, 3-D airway stents have the potential for improving tolerance and decreasing the complication rate.
Major finding: Congruence and outcomes tended to be better in stenoses involving the bronchial level (three of three, no complications).
Study details: A feasibility study of eight patients with nonmalignant, anatomically complex, and symptomatic stenosis for which conventional stents were not suitable.
Disclosures: Dr. Guibert reported having no financial disclosures.
Source: Guibert N et al. ATS 2018, Abstract 4433.
Single Botox treatment cuts AF for 3 years
BOSTON – A single set of four injections with botulinum toxin into neuron-containing cardiac fat pads of patients during open-chest cardiac artery bypass surgery led to a long-term cut in the cumulative incidence of atrial tachyarrhythmias during 3-year follow-up in a pilot, sham-controlled study with 60 patients at two Russian centers.
“Because the favorable reduction of atrial fibrillation [AF] outlasted the anticipated botulinum toxin effects on autonomic nervous system activity, this may represent a form of autonomic reverse remodeling” triggered by just one injection of the paralyzing toxin at each of four intracardiac fat pads, Alexander B. Romanov, MD, said at the annual scientific sessions of the Heart Rhythm Society. Botulinum toxin (BT) blocks neuronal release of acetylcholine, thereby interfering with cholinergic neurotransmission and producing hypothesized neurologic remodeling, explained Dr. Romanov, a researcher at the Meshalkin National Medical Research Center in Novosibirsk, Russia.
The 3-year results also showed statistically significant differences or trends favoring BT injections for several other clinical outcomes. Two deaths and two strokes occurred, all among the control patients. Two patients required a total of three hospitalizations during follow-up in the BT-treated group, compared with 10 patients hospitalized a total of 21 times in the control arm. Clinicians prescribed antiarrhythmic drugs to six of the BT-treated patients and to 15 of the controls.
All patients received an implanted heart rhythm monitor during their bypass surgery, and the researchers measured AF burden – the percentage of time during which AF occurred. After 12 months, 24 months, and 36 months, the AF burden averaged 0.2%, 1.6%, and 1.2%, respectively, in the BT-treated patients and 1.9%, 9.5%, and 6.9% in the sham-control patients.

“We don’t know why this works, but it’s a fascinating new approach that is worthy of further study,” commented Kalyanam Shivkumar, MD, professor and director of the Cardiac Arrhythmia Center at the University of California, Los Angeles, and designated discussant for the report.
“This is an extremely exciting study, but it remains inconclusive because how it works is not fully understood,” commented Andrew D. Krahn, MD, professor and chief of cardiology at the University of British Columbia in Vancouver.
BOSTON – A single set of four injections with botulinum toxin into neuron-containing cardiac fat pads of patients during open-chest cardiac artery bypass surgery led to a long-term cut in the cumulative incidence of atrial tachyarrhythmias during 3-year follow-up in a pilot, sham-controlled study with 60 patients at two Russian centers.
“Because the favorable reduction of atrial fibrillation [AF] outlasted the anticipated botulinum toxin effects on autonomic nervous system activity, this may represent a form of autonomic reverse remodeling” triggered by just one injection of the paralyzing toxin at each of four intracardiac fat pads, Alexander B. Romanov, MD, said at the annual scientific sessions of the Heart Rhythm Society. Botulinum toxin (BT) blocks neuronal release of acetylcholine, thereby interfering with cholinergic neurotransmission and producing hypothesized neurologic remodeling, explained Dr. Romanov, a researcher at the Meshalkin National Medical Research Center in Novosibirsk, Russia.
The 3-year results also showed statistically significant differences or trends favoring BT injections for several other clinical outcomes. Two deaths and two strokes occurred, all among the control patients. Two patients required a total of three hospitalizations during follow-up in the BT-treated group, compared with 10 patients hospitalized a total of 21 times in the control arm. Clinicians prescribed antiarrhythmic drugs to six of the BT-treated patients and to 15 of the controls.
All patients received an implanted heart rhythm monitor during their bypass surgery, and the researchers measured AF burden – the percentage of time during which AF occurred. After 12 months, 24 months, and 36 months, the AF burden averaged 0.2%, 1.6%, and 1.2%, respectively, in the BT-treated patients and 1.9%, 9.5%, and 6.9% in the sham-control patients.

“We don’t know why this works, but it’s a fascinating new approach that is worthy of further study,” commented Kalyanam Shivkumar, MD, professor and director of the Cardiac Arrhythmia Center at the University of California, Los Angeles, and designated discussant for the report.
“This is an extremely exciting study, but it remains inconclusive because how it works is not fully understood,” commented Andrew D. Krahn, MD, professor and chief of cardiology at the University of British Columbia in Vancouver.
BOSTON – A single set of four injections with botulinum toxin into neuron-containing cardiac fat pads of patients during open-chest cardiac artery bypass surgery led to a long-term cut in the cumulative incidence of atrial tachyarrhythmias during 3-year follow-up in a pilot, sham-controlled study with 60 patients at two Russian centers.
“Because the favorable reduction of atrial fibrillation [AF] outlasted the anticipated botulinum toxin effects on autonomic nervous system activity, this may represent a form of autonomic reverse remodeling” triggered by just one injection of the paralyzing toxin at each of four intracardiac fat pads, Alexander B. Romanov, MD, said at the annual scientific sessions of the Heart Rhythm Society. Botulinum toxin (BT) blocks neuronal release of acetylcholine, thereby interfering with cholinergic neurotransmission and producing hypothesized neurologic remodeling, explained Dr. Romanov, a researcher at the Meshalkin National Medical Research Center in Novosibirsk, Russia.
The 3-year results also showed statistically significant differences or trends favoring BT injections for several other clinical outcomes. Two deaths and two strokes occurred, all among the control patients. Two patients required a total of three hospitalizations during follow-up in the BT-treated group, compared with 10 patients hospitalized a total of 21 times in the control arm. Clinicians prescribed antiarrhythmic drugs to six of the BT-treated patients and to 15 of the controls.
All patients received an implanted heart rhythm monitor during their bypass surgery, and the researchers measured AF burden – the percentage of time during which AF occurred. After 12 months, 24 months, and 36 months, the AF burden averaged 0.2%, 1.6%, and 1.2%, respectively, in the BT-treated patients and 1.9%, 9.5%, and 6.9% in the sham-control patients.

“We don’t know why this works, but it’s a fascinating new approach that is worthy of further study,” commented Kalyanam Shivkumar, MD, professor and director of the Cardiac Arrhythmia Center at the University of California, Los Angeles, and designated discussant for the report.
“This is an extremely exciting study, but it remains inconclusive because how it works is not fully understood,” commented Andrew D. Krahn, MD, professor and chief of cardiology at the University of British Columbia in Vancouver.
REPORTING FROM HEART RHYTHM 2018
Key clinical point:
Major finding: During 3-year follow-up, atrial tachyarrhythmias occurred in 23% of botulinum toxin-treated patients and in 50% of sham controls.
Study details: Randomized, sham-controlled study with 60 patients at two Russian centers.
Disclosures: The study received no commercial funding. Dr. Romanov, Dr. Shivkumar, and Dr. Krahn had no relevant disclosures.
Source: Romanov A et al. Heart Rhythm 2018, Abstract B-LBCT02-01.
Malignant pleural mesothelioma guidelines often are ignored
SAN DIEGO – National guidelines for the treatment of malignant pleural mesothelioma often are not followed, a new study showed, with fewer than one-third of patients receiving cancer-directed surgery.
Another 32% received no treatment, although that didn’t seem to have an impact on median months of survival.
Still, “there can be a wide variation in median survival time, depending on clinical factors and tumor characteristics,” said study coauthor Harmik Soukiasian, MD, of Cedars-Sinai Medical Center, Los Angeles. “Given the variation in prognosis, it is quite astonishing that over 30% of MPM patients are not receiving any form of treatment. As clinicians armed with these data, we need to investigate why that is.”
Dr. Soukiasian presented the study findings at the annual meeting of the American Association for Thoracic Surgery.
MPM, a rare cancer, is mainly linked to asbestos exposure. “MPM is almost always a fatal disease, and the prognosis can only be modestly influenced by oncological treatments,” according to the authors of guidelines released in 2013. “The diagnostic process can be complex, with highly specialized advice frequently required to arrive at a definite diagnosis. Treatment varies from therapeutic nihilism to radical combined-modality treatment approaches” (J Thorac Dis. 2013 Dec;5[6]:E254-E307).
Surgical resection is a controversial treatment for MPM, Dr. Soukiasian said. It is “based on the principle of macroscopic resection of solid tumor with adjuvant therapy to treat micrometastatic disease,” he explained. “Cancer-directed surgery for MPM is usually reserved for localized epithelial type histology and is associated with a 5-year survival rate of 15%.”
For the new study, the investigators tracked 3,834 patients in the National Cancer Database (2004-2014) diagnosed with MPM clinical stages I-III. Most had epithelioid MPM (69%), with sarcomatoid (17%) and mixed subtype (15%) making up the rest. They examined whether patient treatment complied with the National Comprehensive Cancer Network (NCCN) guidelines, which recommend surgery in resectable epithelioid MPM.
“Our study revealed significant lack of compliance with NCCN guidelines, as well as many disparities in the management of MPM,” Dr. Soukiasian said. “For the overall cohort, 32.3% of patients did not receive any treatment, 18.1% had surgery plus chemotherapy, 38.6% chemotherapy alone, and only 7% received trimodality therapy. In patients with epithelial histology, surgery was significantly underutilized, with only 30% of patients receiving cancer-directed surgery.”
In addition, he said, “our study reveals several disparities that affect compliance with NCCN guidelines. Treatment disparities were observed in women, octogenarians, the uninsured, the Medicaid-insured, and in patients with comorbidities. Guideline adherence was significantly increased in academic and high-volume hospitals with an associated increase in survival.”
But the study also found that median survival estimates were similar regardless of treatment: 10 months for no treatment, 15 months for chemotherapy only, 17 months for surgery only, and 22 months for surgery plus chemotherapy.
During the AATS presentation, an audience member asked about how performance status – a measure of a person’s ability to perform everyday activities – affects the eligibility for surgery.
“It’s quite common for low performance status to exclude someone from surgery,” the audience member said. “Some of these patients are very sick.”
Dr. Soukiasian acknowledged that performance status was not included in the data. The study was focused on the gap between guidelines and real-world practice, and generated questions of why and about the potential opportunity for improved treatment of these patients.
How do patient choices, cost, and quality of life factor in? “These are very important questions and concerns,” Dr. Soukiasian said. “Although our research does not provide data or conclusions on quality of life or cost, these topics will be important to address in follow-up studies to elucidate possible barriers in the treatment of MPM and the initiation of future educational opportunities for our patients.”
No disclosures and no study funding were reported.
SOURCE: Espinoza-Mercado F et al. General Thoracic Surgery Simultaneous Scientific Session. Abstract 18.
SAN DIEGO – National guidelines for the treatment of malignant pleural mesothelioma often are not followed, a new study showed, with fewer than one-third of patients receiving cancer-directed surgery.
Another 32% received no treatment, although that didn’t seem to have an impact on median months of survival.
Still, “there can be a wide variation in median survival time, depending on clinical factors and tumor characteristics,” said study coauthor Harmik Soukiasian, MD, of Cedars-Sinai Medical Center, Los Angeles. “Given the variation in prognosis, it is quite astonishing that over 30% of MPM patients are not receiving any form of treatment. As clinicians armed with these data, we need to investigate why that is.”
Dr. Soukiasian presented the study findings at the annual meeting of the American Association for Thoracic Surgery.
MPM, a rare cancer, is mainly linked to asbestos exposure. “MPM is almost always a fatal disease, and the prognosis can only be modestly influenced by oncological treatments,” according to the authors of guidelines released in 2013. “The diagnostic process can be complex, with highly specialized advice frequently required to arrive at a definite diagnosis. Treatment varies from therapeutic nihilism to radical combined-modality treatment approaches” (J Thorac Dis. 2013 Dec;5[6]:E254-E307).
Surgical resection is a controversial treatment for MPM, Dr. Soukiasian said. It is “based on the principle of macroscopic resection of solid tumor with adjuvant therapy to treat micrometastatic disease,” he explained. “Cancer-directed surgery for MPM is usually reserved for localized epithelial type histology and is associated with a 5-year survival rate of 15%.”
For the new study, the investigators tracked 3,834 patients in the National Cancer Database (2004-2014) diagnosed with MPM clinical stages I-III. Most had epithelioid MPM (69%), with sarcomatoid (17%) and mixed subtype (15%) making up the rest. They examined whether patient treatment complied with the National Comprehensive Cancer Network (NCCN) guidelines, which recommend surgery in resectable epithelioid MPM.
“Our study revealed significant lack of compliance with NCCN guidelines, as well as many disparities in the management of MPM,” Dr. Soukiasian said. “For the overall cohort, 32.3% of patients did not receive any treatment, 18.1% had surgery plus chemotherapy, 38.6% chemotherapy alone, and only 7% received trimodality therapy. In patients with epithelial histology, surgery was significantly underutilized, with only 30% of patients receiving cancer-directed surgery.”
In addition, he said, “our study reveals several disparities that affect compliance with NCCN guidelines. Treatment disparities were observed in women, octogenarians, the uninsured, the Medicaid-insured, and in patients with comorbidities. Guideline adherence was significantly increased in academic and high-volume hospitals with an associated increase in survival.”
But the study also found that median survival estimates were similar regardless of treatment: 10 months for no treatment, 15 months for chemotherapy only, 17 months for surgery only, and 22 months for surgery plus chemotherapy.
During the AATS presentation, an audience member asked about how performance status – a measure of a person’s ability to perform everyday activities – affects the eligibility for surgery.
“It’s quite common for low performance status to exclude someone from surgery,” the audience member said. “Some of these patients are very sick.”
Dr. Soukiasian acknowledged that performance status was not included in the data. The study was focused on the gap between guidelines and real-world practice, and generated questions of why and about the potential opportunity for improved treatment of these patients.
How do patient choices, cost, and quality of life factor in? “These are very important questions and concerns,” Dr. Soukiasian said. “Although our research does not provide data or conclusions on quality of life or cost, these topics will be important to address in follow-up studies to elucidate possible barriers in the treatment of MPM and the initiation of future educational opportunities for our patients.”
No disclosures and no study funding were reported.
SOURCE: Espinoza-Mercado F et al. General Thoracic Surgery Simultaneous Scientific Session. Abstract 18.
SAN DIEGO – National guidelines for the treatment of malignant pleural mesothelioma often are not followed, a new study showed, with fewer than one-third of patients receiving cancer-directed surgery.
Another 32% received no treatment, although that didn’t seem to have an impact on median months of survival.
Still, “there can be a wide variation in median survival time, depending on clinical factors and tumor characteristics,” said study coauthor Harmik Soukiasian, MD, of Cedars-Sinai Medical Center, Los Angeles. “Given the variation in prognosis, it is quite astonishing that over 30% of MPM patients are not receiving any form of treatment. As clinicians armed with these data, we need to investigate why that is.”
Dr. Soukiasian presented the study findings at the annual meeting of the American Association for Thoracic Surgery.
MPM, a rare cancer, is mainly linked to asbestos exposure. “MPM is almost always a fatal disease, and the prognosis can only be modestly influenced by oncological treatments,” according to the authors of guidelines released in 2013. “The diagnostic process can be complex, with highly specialized advice frequently required to arrive at a definite diagnosis. Treatment varies from therapeutic nihilism to radical combined-modality treatment approaches” (J Thorac Dis. 2013 Dec;5[6]:E254-E307).
Surgical resection is a controversial treatment for MPM, Dr. Soukiasian said. It is “based on the principle of macroscopic resection of solid tumor with adjuvant therapy to treat micrometastatic disease,” he explained. “Cancer-directed surgery for MPM is usually reserved for localized epithelial type histology and is associated with a 5-year survival rate of 15%.”
For the new study, the investigators tracked 3,834 patients in the National Cancer Database (2004-2014) diagnosed with MPM clinical stages I-III. Most had epithelioid MPM (69%), with sarcomatoid (17%) and mixed subtype (15%) making up the rest. They examined whether patient treatment complied with the National Comprehensive Cancer Network (NCCN) guidelines, which recommend surgery in resectable epithelioid MPM.
“Our study revealed significant lack of compliance with NCCN guidelines, as well as many disparities in the management of MPM,” Dr. Soukiasian said. “For the overall cohort, 32.3% of patients did not receive any treatment, 18.1% had surgery plus chemotherapy, 38.6% chemotherapy alone, and only 7% received trimodality therapy. In patients with epithelial histology, surgery was significantly underutilized, with only 30% of patients receiving cancer-directed surgery.”
In addition, he said, “our study reveals several disparities that affect compliance with NCCN guidelines. Treatment disparities were observed in women, octogenarians, the uninsured, the Medicaid-insured, and in patients with comorbidities. Guideline adherence was significantly increased in academic and high-volume hospitals with an associated increase in survival.”
But the study also found that median survival estimates were similar regardless of treatment: 10 months for no treatment, 15 months for chemotherapy only, 17 months for surgery only, and 22 months for surgery plus chemotherapy.
During the AATS presentation, an audience member asked about how performance status – a measure of a person’s ability to perform everyday activities – affects the eligibility for surgery.
“It’s quite common for low performance status to exclude someone from surgery,” the audience member said. “Some of these patients are very sick.”
Dr. Soukiasian acknowledged that performance status was not included in the data. The study was focused on the gap between guidelines and real-world practice, and generated questions of why and about the potential opportunity for improved treatment of these patients.
How do patient choices, cost, and quality of life factor in? “These are very important questions and concerns,” Dr. Soukiasian said. “Although our research does not provide data or conclusions on quality of life or cost, these topics will be important to address in follow-up studies to elucidate possible barriers in the treatment of MPM and the initiation of future educational opportunities for our patients.”
No disclosures and no study funding were reported.
SOURCE: Espinoza-Mercado F et al. General Thoracic Surgery Simultaneous Scientific Session. Abstract 18.
REPORTING FROM THE AATS ANNUAL MEETING
Key clinical point:
Major finding: Guidelines recommend surgery in epithelioid MPM, but only 30% of patients received it.
Study details: Analysis of 3,834 patients diagnosed with MPM clinical stages I-III during 2004-2014.
Disclosures: No disclosures and no study funding were reported.
Source: Espinoza-Mercado F et al. General Thoracic Surgery Simultaneous Scientific Session. Abstract 18.
MOMENTUM 3 HeartMate 3 LVAD ‘practice changing’
ORLANDO – of follow-up in patients with advanced heart failure in the large multicenter MOMENTUM 3 trial, Mandeep R. Mehra, MD, reported at the annual meeting of the American College of Cardiology.
HeartMate 3 recipients had a 90% lower risk of undergoing reoperation to replace or remove their device because of malfunction, and a stroke rate half that in the HeartMate II group.
“This was the lowest rate of stroke ever seen in any LVAD trial,” according to Dr. Mehra, medical director of the Brigham and Women’s Hospital Heart and Vascular Center, Boston, and professor of medicine at Harvard Medical School.
“We believe this is a practice-changing result in the field, and that the real implication of our findings is to reassure those who refer or treat patients with advanced heart failure that it is perhaps going to be ignorant not to refer patients for consideration for destination therapy,” he said at a press conference highlighting the MOMENTUM 3 results, also presented in a late-breaking clinical trials session.
The HeartMate 3 is a miniaturized centrifugal-flow device that fits entirely within the chest, whereas the HeartMate II requires creation of a pocket in the abdomen. The HeartMate 3 was designed to prevent pump thrombosis – a common limiting problem with the HeartMate II and other LVADs – by employing three innovations: use of wide blood-flow passages to reduce shear stress and minimize disruption of red blood cells as they pass through the pump; reliance on magnetic levitation technology to create a frictionless pump with no mechanical bearings, which are subject to wear and tear; and incorporation of an artificial fixed pulse that speeds up and slows every 2 seconds in order to minimize blood stasis, which promotes thrombosis, the cardiologist explained in a video interview.
MOMENTUM 3 is the largest-ever randomized trial of LVAD therapy, involving 1,028 advanced heart failure patients at 69 U.S. centers. The study population is a mix of bridge-to-transplant patients and others who weren’t eligible for heart transplantation and are using their device as lifelong destination therapy. In an earlier report on the first 294 patients to reach 6 months of follow-up post implantation, Dr. Mehra and his coinvestigators showed that the HeartMate 3 group had a significantly lower incidence of the composite endpoint of disabling stroke or reoperation to replace or remove the device (N Engl J Med. 2017 Feb 2;376[5]:440-50).
At ACC 2018, he presented the prespecified 2-year analysis of results in the first 366 patients to reach that benchmark. The rate of survival free of disabling stroke or reoperation for device malfunction was 79.5% in the HeartMate 3 group and 60.2% with the HeartMate II, for a highly significant 54% reduction in the risk of bad outcome. Reoperation for device malfunction occurred in 1.6% of HeartMate 3 patients versus 17% of those with a HeartMate II, for a 92% reduction in risk. Two-year survival was 82.8% in the HeartMate 3 group and 76.2% in HeartMate II recipients.
The overall stroke rate was 10% with the HeartMate 3, compared with 19% with the older, axial-flow LVAD. The incidence of disabling stroke was 3% in the HeartMate 3 group and similar at 2% with the HeartMate II; however, nondisabling stroke occurred in only 3% of HeartMate 3 recipients, compared with 14% of patients with the HeartMate II.
“There has always been this notion that, ‘There are so many complications with this device, so let’s suffer with the disease rather than suffer with the pump.’ Now we’re showing that you don’t suffer with the pump as with the earlier-generation devices. I think this is going to open the gates for more referrals and more opportunities for destination therapy in patients who are deemed ineligible for transplant,” Dr. Mehra predicted.
Discussant James L. Janzuzzi Jr., called the MOMENTUM 3 results “a very-much-needed step forward.”
“Perhaps the most dramatic observation in this study is the dramatic reduction in thrombosis events requiring reoperation. In essence, this problem was entirely prevented by the use of this magnetically levitated centrifugal-flow device. Reoperation for thrombosis accounted for two-thirds of the reoperations in the HeartMate II group and the rate was zero in the HeartMate 3 population. Essentially, with this technology we’ve addressed a very important unmet need by reducing the onset of pump thrombosis, which is the precursor to either pump dysfunction or embolic stroke,” commented Dr. Januzzi, professor of medicine at Harvard Medical School, Boston.
Given the 83% survival rate at 2 years in the HeartMate 3 group in the MOMENTUM 3 trial, the on-average 50% survival at 10 years for heart transplant recipients, and the perpetual enormous shortage of donor organs, it’s time to consider a randomized trial of an advanced LVAD such as the HeartMate 3 versus heart transplantation, with quality-of-life outcomes front and center, he asserted.
Dr. Mehra was all for the idea. He noted that within the community of physicians and surgeons who provide care for advanced heart failure patients there is a growing move to replace problematic axial-flow LVADs requiring reoperation with a HeartMate 3 upgrade.
The MOMENTUM 3 trial is funded by Abbott. Dr. Mehra reported receiving research funds from and serving as a consultant to the company.
Simultaneous with his presentation at ACC 2018, the 2-year results of MOMENTUM 3 were published online at NEJM.org (doi: 10.1056/NEJMoa1800866).
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SOURCE: Mehra M et al. ACC 18.
ORLANDO – of follow-up in patients with advanced heart failure in the large multicenter MOMENTUM 3 trial, Mandeep R. Mehra, MD, reported at the annual meeting of the American College of Cardiology.
HeartMate 3 recipients had a 90% lower risk of undergoing reoperation to replace or remove their device because of malfunction, and a stroke rate half that in the HeartMate II group.
“This was the lowest rate of stroke ever seen in any LVAD trial,” according to Dr. Mehra, medical director of the Brigham and Women’s Hospital Heart and Vascular Center, Boston, and professor of medicine at Harvard Medical School.
“We believe this is a practice-changing result in the field, and that the real implication of our findings is to reassure those who refer or treat patients with advanced heart failure that it is perhaps going to be ignorant not to refer patients for consideration for destination therapy,” he said at a press conference highlighting the MOMENTUM 3 results, also presented in a late-breaking clinical trials session.
The HeartMate 3 is a miniaturized centrifugal-flow device that fits entirely within the chest, whereas the HeartMate II requires creation of a pocket in the abdomen. The HeartMate 3 was designed to prevent pump thrombosis – a common limiting problem with the HeartMate II and other LVADs – by employing three innovations: use of wide blood-flow passages to reduce shear stress and minimize disruption of red blood cells as they pass through the pump; reliance on magnetic levitation technology to create a frictionless pump with no mechanical bearings, which are subject to wear and tear; and incorporation of an artificial fixed pulse that speeds up and slows every 2 seconds in order to minimize blood stasis, which promotes thrombosis, the cardiologist explained in a video interview.
MOMENTUM 3 is the largest-ever randomized trial of LVAD therapy, involving 1,028 advanced heart failure patients at 69 U.S. centers. The study population is a mix of bridge-to-transplant patients and others who weren’t eligible for heart transplantation and are using their device as lifelong destination therapy. In an earlier report on the first 294 patients to reach 6 months of follow-up post implantation, Dr. Mehra and his coinvestigators showed that the HeartMate 3 group had a significantly lower incidence of the composite endpoint of disabling stroke or reoperation to replace or remove the device (N Engl J Med. 2017 Feb 2;376[5]:440-50).
At ACC 2018, he presented the prespecified 2-year analysis of results in the first 366 patients to reach that benchmark. The rate of survival free of disabling stroke or reoperation for device malfunction was 79.5% in the HeartMate 3 group and 60.2% with the HeartMate II, for a highly significant 54% reduction in the risk of bad outcome. Reoperation for device malfunction occurred in 1.6% of HeartMate 3 patients versus 17% of those with a HeartMate II, for a 92% reduction in risk. Two-year survival was 82.8% in the HeartMate 3 group and 76.2% in HeartMate II recipients.
The overall stroke rate was 10% with the HeartMate 3, compared with 19% with the older, axial-flow LVAD. The incidence of disabling stroke was 3% in the HeartMate 3 group and similar at 2% with the HeartMate II; however, nondisabling stroke occurred in only 3% of HeartMate 3 recipients, compared with 14% of patients with the HeartMate II.
“There has always been this notion that, ‘There are so many complications with this device, so let’s suffer with the disease rather than suffer with the pump.’ Now we’re showing that you don’t suffer with the pump as with the earlier-generation devices. I think this is going to open the gates for more referrals and more opportunities for destination therapy in patients who are deemed ineligible for transplant,” Dr. Mehra predicted.
Discussant James L. Janzuzzi Jr., called the MOMENTUM 3 results “a very-much-needed step forward.”
“Perhaps the most dramatic observation in this study is the dramatic reduction in thrombosis events requiring reoperation. In essence, this problem was entirely prevented by the use of this magnetically levitated centrifugal-flow device. Reoperation for thrombosis accounted for two-thirds of the reoperations in the HeartMate II group and the rate was zero in the HeartMate 3 population. Essentially, with this technology we’ve addressed a very important unmet need by reducing the onset of pump thrombosis, which is the precursor to either pump dysfunction or embolic stroke,” commented Dr. Januzzi, professor of medicine at Harvard Medical School, Boston.
Given the 83% survival rate at 2 years in the HeartMate 3 group in the MOMENTUM 3 trial, the on-average 50% survival at 10 years for heart transplant recipients, and the perpetual enormous shortage of donor organs, it’s time to consider a randomized trial of an advanced LVAD such as the HeartMate 3 versus heart transplantation, with quality-of-life outcomes front and center, he asserted.
Dr. Mehra was all for the idea. He noted that within the community of physicians and surgeons who provide care for advanced heart failure patients there is a growing move to replace problematic axial-flow LVADs requiring reoperation with a HeartMate 3 upgrade.
The MOMENTUM 3 trial is funded by Abbott. Dr. Mehra reported receiving research funds from and serving as a consultant to the company.
Simultaneous with his presentation at ACC 2018, the 2-year results of MOMENTUM 3 were published online at NEJM.org (doi: 10.1056/NEJMoa1800866).
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SOURCE: Mehra M et al. ACC 18.
ORLANDO – of follow-up in patients with advanced heart failure in the large multicenter MOMENTUM 3 trial, Mandeep R. Mehra, MD, reported at the annual meeting of the American College of Cardiology.
HeartMate 3 recipients had a 90% lower risk of undergoing reoperation to replace or remove their device because of malfunction, and a stroke rate half that in the HeartMate II group.
“This was the lowest rate of stroke ever seen in any LVAD trial,” according to Dr. Mehra, medical director of the Brigham and Women’s Hospital Heart and Vascular Center, Boston, and professor of medicine at Harvard Medical School.
“We believe this is a practice-changing result in the field, and that the real implication of our findings is to reassure those who refer or treat patients with advanced heart failure that it is perhaps going to be ignorant not to refer patients for consideration for destination therapy,” he said at a press conference highlighting the MOMENTUM 3 results, also presented in a late-breaking clinical trials session.
The HeartMate 3 is a miniaturized centrifugal-flow device that fits entirely within the chest, whereas the HeartMate II requires creation of a pocket in the abdomen. The HeartMate 3 was designed to prevent pump thrombosis – a common limiting problem with the HeartMate II and other LVADs – by employing three innovations: use of wide blood-flow passages to reduce shear stress and minimize disruption of red blood cells as they pass through the pump; reliance on magnetic levitation technology to create a frictionless pump with no mechanical bearings, which are subject to wear and tear; and incorporation of an artificial fixed pulse that speeds up and slows every 2 seconds in order to minimize blood stasis, which promotes thrombosis, the cardiologist explained in a video interview.
MOMENTUM 3 is the largest-ever randomized trial of LVAD therapy, involving 1,028 advanced heart failure patients at 69 U.S. centers. The study population is a mix of bridge-to-transplant patients and others who weren’t eligible for heart transplantation and are using their device as lifelong destination therapy. In an earlier report on the first 294 patients to reach 6 months of follow-up post implantation, Dr. Mehra and his coinvestigators showed that the HeartMate 3 group had a significantly lower incidence of the composite endpoint of disabling stroke or reoperation to replace or remove the device (N Engl J Med. 2017 Feb 2;376[5]:440-50).
At ACC 2018, he presented the prespecified 2-year analysis of results in the first 366 patients to reach that benchmark. The rate of survival free of disabling stroke or reoperation for device malfunction was 79.5% in the HeartMate 3 group and 60.2% with the HeartMate II, for a highly significant 54% reduction in the risk of bad outcome. Reoperation for device malfunction occurred in 1.6% of HeartMate 3 patients versus 17% of those with a HeartMate II, for a 92% reduction in risk. Two-year survival was 82.8% in the HeartMate 3 group and 76.2% in HeartMate II recipients.
The overall stroke rate was 10% with the HeartMate 3, compared with 19% with the older, axial-flow LVAD. The incidence of disabling stroke was 3% in the HeartMate 3 group and similar at 2% with the HeartMate II; however, nondisabling stroke occurred in only 3% of HeartMate 3 recipients, compared with 14% of patients with the HeartMate II.
“There has always been this notion that, ‘There are so many complications with this device, so let’s suffer with the disease rather than suffer with the pump.’ Now we’re showing that you don’t suffer with the pump as with the earlier-generation devices. I think this is going to open the gates for more referrals and more opportunities for destination therapy in patients who are deemed ineligible for transplant,” Dr. Mehra predicted.
Discussant James L. Janzuzzi Jr., called the MOMENTUM 3 results “a very-much-needed step forward.”
“Perhaps the most dramatic observation in this study is the dramatic reduction in thrombosis events requiring reoperation. In essence, this problem was entirely prevented by the use of this magnetically levitated centrifugal-flow device. Reoperation for thrombosis accounted for two-thirds of the reoperations in the HeartMate II group and the rate was zero in the HeartMate 3 population. Essentially, with this technology we’ve addressed a very important unmet need by reducing the onset of pump thrombosis, which is the precursor to either pump dysfunction or embolic stroke,” commented Dr. Januzzi, professor of medicine at Harvard Medical School, Boston.
Given the 83% survival rate at 2 years in the HeartMate 3 group in the MOMENTUM 3 trial, the on-average 50% survival at 10 years for heart transplant recipients, and the perpetual enormous shortage of donor organs, it’s time to consider a randomized trial of an advanced LVAD such as the HeartMate 3 versus heart transplantation, with quality-of-life outcomes front and center, he asserted.
Dr. Mehra was all for the idea. He noted that within the community of physicians and surgeons who provide care for advanced heart failure patients there is a growing move to replace problematic axial-flow LVADs requiring reoperation with a HeartMate 3 upgrade.
The MOMENTUM 3 trial is funded by Abbott. Dr. Mehra reported receiving research funds from and serving as a consultant to the company.
Simultaneous with his presentation at ACC 2018, the 2-year results of MOMENTUM 3 were published online at NEJM.org (doi: 10.1056/NEJMoa1800866).
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SOURCE: Mehra M et al. ACC 18.
REPORTING FROM ACC 18
Key clinical point: Landmark trial paves way to much wider use of LVADs as destination therapy.
Major finding: The 2-year rate of survival free of disabling stroke or reoperation to replace or remove a malfunctioning LVAD was 79.5% with the novel HeartMate 3 LVAD, compared with 60.2% with the commonly used HeartMate II axial-flow device.
Study details: This was a randomized, unblinded, multicenter study of more than 1,000 patients with advanced heart failure.
Disclosures: The MOMENTUM 3 trial was funded by Abbott. The presenter reported receiving research grants from and serving as a consultant to the company.
Source: Mehra MR et al. ACC 18.
Reconstruction may reduce chest-related distress in transmasculine youth
, according to study results.
This study is one of the first to document the ongoing impact of chest dysphoria in transgender youths, defined as individuals assigned female at birth who have a masculine gender identity.
“Given these findings, professional guidelines and clinical practice should consider patients for chest surgery based on individual need rather than chronologic age,” said Johanna Olson-Kennedy, MD, of the division of adolescent medicine at Children’s Hospital Los Angeles, and her coauthors.
National guidelines on transgender health care are unclear as to whether minors should be referred for chest surgery because of a lack of data documenting effects of chest surgery in individuals younger than 18 years of age, Dr. Olson-Kennedy and her colleagues wrote in the study, published in JAMA Pediatrics.
To evaluate the discomfort and subsequent consequences of chest dysphoria, the researchers developed a 10-minute, 21-item survey based on Dr. Olson-Kennedy’s 11 years of experience providing care for transgender youth. It was reviewed by a small number of transmasculine youth and adults to determine whether the questions contained the elements of chest dysphoria effectively, used appropriate language, and was otherwise acceptable. From the survey, the researchers derived a chest dysphoria composite score of 0-51, with higher scores indicating increased distress.
Some of the items on the chest dysphoria survey included avoiding exercise, not seeking medical care, and not swimming because of “my chest,” and that taking a shower is difficult as is dating and physical intimacy.
The study included surveys completed by nonsurgical (n = 68) and postsurgical (n = 68) cohorts of transmasculine individuals aged 13-25 years.
The chest dysphoria composite score was significantly higher for minors and young adults who had not undergone chest reconstruction, compared with those who had undergone the procedure (29.6 vs. 3.3; P less than .001), the investigators reported.
Among transmasculine youths who had not undergone surgery, 94% perceived the procedure as very important, Dr. Olson-Kennedy and her coauthors noted.
Moreover, chest dysphoria increased by 0.33 points for every month that passed between a youth starting testosterone therapy and undergoing surgery, results of a linear regression analysis showed.
Most survey respondents reported that they were currently taking testosterone: 87% of the nonsurgical group and 97% of the postsurgical group.
Of the individuals who had undergone chest reconstruction, one (less than 1%) reported regretting the procedure “sometimes,” said Dr. Olson-Kennedy and her coauthors.
The mean age of survey participants was 17 (13-23 years) and 19 years (14-25 years) in the nonsurgical and postsurgical cohorts, respectively.Based on the results, Dr. Olson-Kennedy and her associates called for changes to clinical practice and to insurance plans, which sometimes require 12 months of continuous testosterone therapy prior to chest surgery. “Individualized, patient-centered care plans should be considered the standard of care for all transgender adolescents, and referrals should be made accordingly.”
Dr. Olson-Kennedy and her coauthors reported no conflicts of interest related to the study, which was funded by the Eunice Kennedy Shriver National Institute for Child Health and Human Development.
SOURCE: Olson-Kennedy J et al. JAMA Pediatrics. 2018 Mar 5. doi: 10.1001/jamapediatrics.2017.5440.
, according to study results.
This study is one of the first to document the ongoing impact of chest dysphoria in transgender youths, defined as individuals assigned female at birth who have a masculine gender identity.
“Given these findings, professional guidelines and clinical practice should consider patients for chest surgery based on individual need rather than chronologic age,” said Johanna Olson-Kennedy, MD, of the division of adolescent medicine at Children’s Hospital Los Angeles, and her coauthors.
National guidelines on transgender health care are unclear as to whether minors should be referred for chest surgery because of a lack of data documenting effects of chest surgery in individuals younger than 18 years of age, Dr. Olson-Kennedy and her colleagues wrote in the study, published in JAMA Pediatrics.
To evaluate the discomfort and subsequent consequences of chest dysphoria, the researchers developed a 10-minute, 21-item survey based on Dr. Olson-Kennedy’s 11 years of experience providing care for transgender youth. It was reviewed by a small number of transmasculine youth and adults to determine whether the questions contained the elements of chest dysphoria effectively, used appropriate language, and was otherwise acceptable. From the survey, the researchers derived a chest dysphoria composite score of 0-51, with higher scores indicating increased distress.
Some of the items on the chest dysphoria survey included avoiding exercise, not seeking medical care, and not swimming because of “my chest,” and that taking a shower is difficult as is dating and physical intimacy.
The study included surveys completed by nonsurgical (n = 68) and postsurgical (n = 68) cohorts of transmasculine individuals aged 13-25 years.
The chest dysphoria composite score was significantly higher for minors and young adults who had not undergone chest reconstruction, compared with those who had undergone the procedure (29.6 vs. 3.3; P less than .001), the investigators reported.
Among transmasculine youths who had not undergone surgery, 94% perceived the procedure as very important, Dr. Olson-Kennedy and her coauthors noted.
Moreover, chest dysphoria increased by 0.33 points for every month that passed between a youth starting testosterone therapy and undergoing surgery, results of a linear regression analysis showed.
Most survey respondents reported that they were currently taking testosterone: 87% of the nonsurgical group and 97% of the postsurgical group.
Of the individuals who had undergone chest reconstruction, one (less than 1%) reported regretting the procedure “sometimes,” said Dr. Olson-Kennedy and her coauthors.
The mean age of survey participants was 17 (13-23 years) and 19 years (14-25 years) in the nonsurgical and postsurgical cohorts, respectively.Based on the results, Dr. Olson-Kennedy and her associates called for changes to clinical practice and to insurance plans, which sometimes require 12 months of continuous testosterone therapy prior to chest surgery. “Individualized, patient-centered care plans should be considered the standard of care for all transgender adolescents, and referrals should be made accordingly.”
Dr. Olson-Kennedy and her coauthors reported no conflicts of interest related to the study, which was funded by the Eunice Kennedy Shriver National Institute for Child Health and Human Development.
SOURCE: Olson-Kennedy J et al. JAMA Pediatrics. 2018 Mar 5. doi: 10.1001/jamapediatrics.2017.5440.
, according to study results.
This study is one of the first to document the ongoing impact of chest dysphoria in transgender youths, defined as individuals assigned female at birth who have a masculine gender identity.
“Given these findings, professional guidelines and clinical practice should consider patients for chest surgery based on individual need rather than chronologic age,” said Johanna Olson-Kennedy, MD, of the division of adolescent medicine at Children’s Hospital Los Angeles, and her coauthors.
National guidelines on transgender health care are unclear as to whether minors should be referred for chest surgery because of a lack of data documenting effects of chest surgery in individuals younger than 18 years of age, Dr. Olson-Kennedy and her colleagues wrote in the study, published in JAMA Pediatrics.
To evaluate the discomfort and subsequent consequences of chest dysphoria, the researchers developed a 10-minute, 21-item survey based on Dr. Olson-Kennedy’s 11 years of experience providing care for transgender youth. It was reviewed by a small number of transmasculine youth and adults to determine whether the questions contained the elements of chest dysphoria effectively, used appropriate language, and was otherwise acceptable. From the survey, the researchers derived a chest dysphoria composite score of 0-51, with higher scores indicating increased distress.
Some of the items on the chest dysphoria survey included avoiding exercise, not seeking medical care, and not swimming because of “my chest,” and that taking a shower is difficult as is dating and physical intimacy.
The study included surveys completed by nonsurgical (n = 68) and postsurgical (n = 68) cohorts of transmasculine individuals aged 13-25 years.
The chest dysphoria composite score was significantly higher for minors and young adults who had not undergone chest reconstruction, compared with those who had undergone the procedure (29.6 vs. 3.3; P less than .001), the investigators reported.
Among transmasculine youths who had not undergone surgery, 94% perceived the procedure as very important, Dr. Olson-Kennedy and her coauthors noted.
Moreover, chest dysphoria increased by 0.33 points for every month that passed between a youth starting testosterone therapy and undergoing surgery, results of a linear regression analysis showed.
Most survey respondents reported that they were currently taking testosterone: 87% of the nonsurgical group and 97% of the postsurgical group.
Of the individuals who had undergone chest reconstruction, one (less than 1%) reported regretting the procedure “sometimes,” said Dr. Olson-Kennedy and her coauthors.
The mean age of survey participants was 17 (13-23 years) and 19 years (14-25 years) in the nonsurgical and postsurgical cohorts, respectively.Based on the results, Dr. Olson-Kennedy and her associates called for changes to clinical practice and to insurance plans, which sometimes require 12 months of continuous testosterone therapy prior to chest surgery. “Individualized, patient-centered care plans should be considered the standard of care for all transgender adolescents, and referrals should be made accordingly.”
Dr. Olson-Kennedy and her coauthors reported no conflicts of interest related to the study, which was funded by the Eunice Kennedy Shriver National Institute for Child Health and Human Development.
SOURCE: Olson-Kennedy J et al. JAMA Pediatrics. 2018 Mar 5. doi: 10.1001/jamapediatrics.2017.5440.
FROM JAMA PEDIATRICS
Key clinical point: Chest surgery for transmasculine youths should be considered based on individual needs, rather than chronologic age.
Major finding: Chest dysphoria composite score was higher for minors and young adults who had not undergone chest reconstruction, compared with those who had undergone the procedure (P less than .001).
Study details: Comparison of surveys completed by nonsurgical (n = 68) and postsurgical (n = 68) cohorts of individuals 13-25years old who were assigned female at birth but identified as masculine.
Disclosures: The Eunice Kennedy Shriver National Institute for Child Health and Human Development funded the study. The authors reported no conflicts of interest.
Source: Olson-Kennedy J et al. JAMA Pediatrics. 2018 Mar 5. doi: 10.1001/jamapediatrics.2017.5440.
Preoperative exercise lowers postoperative lung resection complications
with a systematic review suggesting it reduces postoperative complications and duration of hospital stay.
The review and meta-analysis, published in the February British Journal of Sports Medicine, looked at the impact of preoperative exercise in patients undergoing surgery for a range of cancers.
Their review of 13 interventional trials, involving 806 patients and six tumor types, found the postoperative benefits of exercise were evident only in patients undergoing lung resection.
Data from five randomized controlled trials and one quasirandomized trial in lung cancer patients showed a significant 48% reduction in postoperative complications, and a significant mean reduction of 2.86 days in hospital stay among patients undergoing lung resection, compared with controls.
“Postoperative complication is a major concern for patients undergoing oncological surgery,” wrote Dr. Daniel Steffens, from the Surgical Outcomes Research Centre at the Royal Prince Alfred Hospital, Sydney, and his coauthors. They suggested the benefits for patients undergoing lung resection were significant enough that exercise before surgery should be considered as standard preoperative care.
“Such findings may also [have impacts] on health care costs and on patients’ quality of life, and consequently, have important implications for patients, health care professionals and policy makers.”
The exercise regimens in the lung cancer studies mostly involved aerobic exercise, such as walking, and breathing exercises to train respiratory muscles, as well as use of an exercise bicycle. The exercises were undertaken in the 1-2 weeks before surgery, with a frequency ranging from three times a week to three times a day.
The authors noted that trials involving a higher frequency of exercise showed a larger effect size, which suggested there was a dose-response relationship.
There was little evidence of benefit in other tumor types. Two studies examined the benefits of preoperative pelvic floor muscle exercises in men undergoing radical prostatectomy and found significant benefits in quality of life, assessed using the International Continence Society Male Short form. However, the authors pointed out that the quality of evidence was very low.
One study investigated the effects of preoperative mouth-opening exercise training in patients undergoing surgery for oral cancer and found enhanced postoperative quality of life in these patients, but the researchers did not report estimates.
For patients undergoing surgery for colon cancer, colorectal liver metastases, and esophageal cancer, there was no benefit of exercise either in postoperative complications or duration of hospital stay. In all these studies, the authors rated the quality of evidence as “very low.”
“Despite the evidence suggesting that exercise improves physical and mental health in patients with cancer, there are only a limited number of trials investigating the effect of preoperative exercise on patients’ quality of life,” the authors wrote. “Therefore, the effect of preoperative exercise on quality of life at short-term and long-term postoperation should be explored in future trials.”
No conflicts of interest were declared.
SOURCE: Steffens D et al. Br J Sports Med. 2018 Feb 1. doi: 10.1136/bjsports-2017-098032
with a systematic review suggesting it reduces postoperative complications and duration of hospital stay.
The review and meta-analysis, published in the February British Journal of Sports Medicine, looked at the impact of preoperative exercise in patients undergoing surgery for a range of cancers.
Their review of 13 interventional trials, involving 806 patients and six tumor types, found the postoperative benefits of exercise were evident only in patients undergoing lung resection.
Data from five randomized controlled trials and one quasirandomized trial in lung cancer patients showed a significant 48% reduction in postoperative complications, and a significant mean reduction of 2.86 days in hospital stay among patients undergoing lung resection, compared with controls.
“Postoperative complication is a major concern for patients undergoing oncological surgery,” wrote Dr. Daniel Steffens, from the Surgical Outcomes Research Centre at the Royal Prince Alfred Hospital, Sydney, and his coauthors. They suggested the benefits for patients undergoing lung resection were significant enough that exercise before surgery should be considered as standard preoperative care.
“Such findings may also [have impacts] on health care costs and on patients’ quality of life, and consequently, have important implications for patients, health care professionals and policy makers.”
The exercise regimens in the lung cancer studies mostly involved aerobic exercise, such as walking, and breathing exercises to train respiratory muscles, as well as use of an exercise bicycle. The exercises were undertaken in the 1-2 weeks before surgery, with a frequency ranging from three times a week to three times a day.
The authors noted that trials involving a higher frequency of exercise showed a larger effect size, which suggested there was a dose-response relationship.
There was little evidence of benefit in other tumor types. Two studies examined the benefits of preoperative pelvic floor muscle exercises in men undergoing radical prostatectomy and found significant benefits in quality of life, assessed using the International Continence Society Male Short form. However, the authors pointed out that the quality of evidence was very low.
One study investigated the effects of preoperative mouth-opening exercise training in patients undergoing surgery for oral cancer and found enhanced postoperative quality of life in these patients, but the researchers did not report estimates.
For patients undergoing surgery for colon cancer, colorectal liver metastases, and esophageal cancer, there was no benefit of exercise either in postoperative complications or duration of hospital stay. In all these studies, the authors rated the quality of evidence as “very low.”
“Despite the evidence suggesting that exercise improves physical and mental health in patients with cancer, there are only a limited number of trials investigating the effect of preoperative exercise on patients’ quality of life,” the authors wrote. “Therefore, the effect of preoperative exercise on quality of life at short-term and long-term postoperation should be explored in future trials.”
No conflicts of interest were declared.
SOURCE: Steffens D et al. Br J Sports Med. 2018 Feb 1. doi: 10.1136/bjsports-2017-098032
with a systematic review suggesting it reduces postoperative complications and duration of hospital stay.
The review and meta-analysis, published in the February British Journal of Sports Medicine, looked at the impact of preoperative exercise in patients undergoing surgery for a range of cancers.
Their review of 13 interventional trials, involving 806 patients and six tumor types, found the postoperative benefits of exercise were evident only in patients undergoing lung resection.
Data from five randomized controlled trials and one quasirandomized trial in lung cancer patients showed a significant 48% reduction in postoperative complications, and a significant mean reduction of 2.86 days in hospital stay among patients undergoing lung resection, compared with controls.
“Postoperative complication is a major concern for patients undergoing oncological surgery,” wrote Dr. Daniel Steffens, from the Surgical Outcomes Research Centre at the Royal Prince Alfred Hospital, Sydney, and his coauthors. They suggested the benefits for patients undergoing lung resection were significant enough that exercise before surgery should be considered as standard preoperative care.
“Such findings may also [have impacts] on health care costs and on patients’ quality of life, and consequently, have important implications for patients, health care professionals and policy makers.”
The exercise regimens in the lung cancer studies mostly involved aerobic exercise, such as walking, and breathing exercises to train respiratory muscles, as well as use of an exercise bicycle. The exercises were undertaken in the 1-2 weeks before surgery, with a frequency ranging from three times a week to three times a day.
The authors noted that trials involving a higher frequency of exercise showed a larger effect size, which suggested there was a dose-response relationship.
There was little evidence of benefit in other tumor types. Two studies examined the benefits of preoperative pelvic floor muscle exercises in men undergoing radical prostatectomy and found significant benefits in quality of life, assessed using the International Continence Society Male Short form. However, the authors pointed out that the quality of evidence was very low.
One study investigated the effects of preoperative mouth-opening exercise training in patients undergoing surgery for oral cancer and found enhanced postoperative quality of life in these patients, but the researchers did not report estimates.
For patients undergoing surgery for colon cancer, colorectal liver metastases, and esophageal cancer, there was no benefit of exercise either in postoperative complications or duration of hospital stay. In all these studies, the authors rated the quality of evidence as “very low.”
“Despite the evidence suggesting that exercise improves physical and mental health in patients with cancer, there are only a limited number of trials investigating the effect of preoperative exercise on patients’ quality of life,” the authors wrote. “Therefore, the effect of preoperative exercise on quality of life at short-term and long-term postoperation should be explored in future trials.”
No conflicts of interest were declared.
SOURCE: Steffens D et al. Br J Sports Med. 2018 Feb 1. doi: 10.1136/bjsports-2017-098032
FROM THE BRITISH JOURNAL OF SPORTS MEDICINE
Key clinical point: Exercising before oncologic surgery appears to lower the risk of postoperative complications and reduce hospital stay for lung cancer patients.
Major finding: Patients who participated in preoperative exercise before lung cancer surgery had a 48% reduction in postoperative complications, compared with controls.
Data source: Systematic review and meta-analysis of 13 interventional trials involving 806 patients.
Disclosures: No conflicts of interest were declared.
Source: Steffens D et al. Br J Sports Med. 2018, Feb 1. doi: 10.1136/bjsports-2017-098032
Transcatheter aortic valve-in-ring for mitral disease a winner
DENVER – Transseptal mitral valve implantation of an off-the-shelf, commercially available TAVR valve in high-surgical-risk patients with a failing surgically implanted mitral ring prosthesis has become a reasonable treatment strategy in light of the interim findings of the ground-breaking MITRAL trial, Mayra E. Guerrero, MD, said at the Transcatheter Cardiovascular Therapeutics annual educational meeting.
Her presentation of the preliminary results of the MITRAL (Mitral Implantation of Transcatheter Valves) trial showed this valve-in-ring (ViR) treatment strategy using the Sapien 3 valve was associated with low 30-day morbidity and mortality rates and impressive symptomatic improvement.
In contrast, another arm of the MITRAL trial showed that placement of the Sapien 3 TAVR valve in high-surgical-risk patients with severe mitral stenosis due to mitral annular calcification (MAC) of their native valve is a treatment strategy that’s not yet ready for prime time, she added at the meeting, which was sponsored by the Cardiovascular Research Foundation.
“Transcatheter mitral valve replacement in MAC is a challenging procedure associated with complications,” Dr. Guerrero observed. “It may become a reasonable alternative for high-surgical-risk patients with favorable anatomy, but techniques require further refinement.”
The ViR arm of the observational multicenter prospective MITRAL trial included 30 patients with extremely high surgical risk and either severe mitral stenosis as defined by a mitral valve area of 1.5 cm2 or less or moderate mitral stenosis plus severe mitral regurgitation. The most common type of failing ring was the Edwards Physio, in nine patients. Access for transcatheter mitral valve replacement (TMVR) was transseptal in 100% of patients.
The technical success rate at exit from the catheterization lab was 70%. The procedural success rate at 30 days was 62%.
Six patients required a second valve. This was mainly because of malpositioning of the first valve with resultant mitral regurgitation; however, this problem became a nonissue as operator experience grew. All six affected patients were alive at 30 days, and four of the six were New York Heart Association (NYHA) functional class I or II.
In-hospital and 30-day mortality rates were low. There was a single cardiovascular death and one noncardiac death in hospital, with no additional deaths through 30 days. No cases of stroke, acute MI, or valve embolization or thrombosis occurred. The mean mitral valve area at 30 days was 2.1 cm2, although three patients still had a mitral valve area of less 1.5 cm2. Three patients experienced acute renal failure requiring hemodialysis. Seventy-five percent of patients had no or trace mitral regurgitation by echocardiography; the rest had mild regurgitation.
Although at baseline more than 60% of the patients were New York Heart Association class III, 10% were class IV, and the rest were class II, at 30 days more than 30% were New York Heart Association class I, 40% were class II, and the rest were class III.
The 30-day all-cause mortality rate of 6.8% in the MITRAL study is roughly half that reported for ViR patients in the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry. Dr. Guerrero attributed this to refined procedural techniques and improved patient selection through the use of CT imaging and echocardiography.
Heart valve design changes, such as a longer inner skirt, might further improve the technical success rate for ViR, according to Dr. Guerrero, an interventional cardiologist at NorthShore University Health System in Evanston, Ill.
Picking the right ring
Given that studies show one-third of recipients of a surgical mitral ring or surgical mitral valve will require a repeat intervention within 10 years, she made a plea to surgeons: “If we are going to be treating patients with valve-in-ring TMVR, that means when surgeons do a repair they should pick a ring that is amenable to a ViR procedure. So don’t use flexible incomplete bands or very rigid rings because those are really difficult to treat later on. We should pick a ring thinking of the future. That ring is going to fail at some point, and when it fails it’s going to make our lives much easier if we’d picked the right ring.”
MAC TMVR needs more work
In the MAC arm of the MITRAL trial, 96 patients were screened so the researchers could find 30 candidates for TMVR. The 61 rejections were for high risk of left ventricular outflow tract obstruction (LVOTO), embolization, or both.
Fourteen patients underwent transseptal TMVR, and one with anatomy unsuitable for a transseptal procedure had a transapical approach. The other 15 patients had a transatrial surgical approach, which allows resection of the anterior leaflet to reduce the risk of LVOTO and placement of sutures to reduce the embolization risk. However, this came at the cost of increased mortality risk: Three of the five in-hospital deaths were in the transatrial TMVR group.
The technical success rate at exit from the cath lab in the MAC patients was 73%, with a 30-day procedural success rate of 46% and a 19% 30-day mortality. Three patients developed severe LVOTO with hemodynamic compromise.
One transseptal and one transapical TMVR were complicated by LVOTO, both treated by bailout alcohol septal ablation. This led Dr. Guerrero and her coinvestigators to the concept of preemptive alcohol septal ablation, which they used in seven patients deemed at high risk for LVOTO an average of 6 weeks prior to transseptal TMVR as a successful risk reduction strategy.
Survival climbing with operator experience
“In the early days of the TMVR MAC registry, the 30-day mortality rate was 37%. It came down to 22% in the middle third of the registry, then about 18% in the final third. Now we’ve got it down in MITRAL to 16.7%, but when you separate the rate in the transseptal versus the transatrial patients, it’s 13% versus 20%. The difference is not statistically significant, but it’s promising, and I think we are making great progress,” Dr. Guerrero said.
Safety and efficacy endpoints in MITRAL will be reported again at 1 year of follow-up.
The MITRAL trial was partially supported by Edwards Lifesciences. Dr. Guerrero reported receiving a research grant from that company and serving as a consultant to Tendyne Holdings/Abbott and on a speakers bureau for Abiomed.
SOURCE: Guerrero M. No abstract.
DENVER – Transseptal mitral valve implantation of an off-the-shelf, commercially available TAVR valve in high-surgical-risk patients with a failing surgically implanted mitral ring prosthesis has become a reasonable treatment strategy in light of the interim findings of the ground-breaking MITRAL trial, Mayra E. Guerrero, MD, said at the Transcatheter Cardiovascular Therapeutics annual educational meeting.
Her presentation of the preliminary results of the MITRAL (Mitral Implantation of Transcatheter Valves) trial showed this valve-in-ring (ViR) treatment strategy using the Sapien 3 valve was associated with low 30-day morbidity and mortality rates and impressive symptomatic improvement.
In contrast, another arm of the MITRAL trial showed that placement of the Sapien 3 TAVR valve in high-surgical-risk patients with severe mitral stenosis due to mitral annular calcification (MAC) of their native valve is a treatment strategy that’s not yet ready for prime time, she added at the meeting, which was sponsored by the Cardiovascular Research Foundation.
“Transcatheter mitral valve replacement in MAC is a challenging procedure associated with complications,” Dr. Guerrero observed. “It may become a reasonable alternative for high-surgical-risk patients with favorable anatomy, but techniques require further refinement.”
The ViR arm of the observational multicenter prospective MITRAL trial included 30 patients with extremely high surgical risk and either severe mitral stenosis as defined by a mitral valve area of 1.5 cm2 or less or moderate mitral stenosis plus severe mitral regurgitation. The most common type of failing ring was the Edwards Physio, in nine patients. Access for transcatheter mitral valve replacement (TMVR) was transseptal in 100% of patients.
The technical success rate at exit from the catheterization lab was 70%. The procedural success rate at 30 days was 62%.
Six patients required a second valve. This was mainly because of malpositioning of the first valve with resultant mitral regurgitation; however, this problem became a nonissue as operator experience grew. All six affected patients were alive at 30 days, and four of the six were New York Heart Association (NYHA) functional class I or II.
In-hospital and 30-day mortality rates were low. There was a single cardiovascular death and one noncardiac death in hospital, with no additional deaths through 30 days. No cases of stroke, acute MI, or valve embolization or thrombosis occurred. The mean mitral valve area at 30 days was 2.1 cm2, although three patients still had a mitral valve area of less 1.5 cm2. Three patients experienced acute renal failure requiring hemodialysis. Seventy-five percent of patients had no or trace mitral regurgitation by echocardiography; the rest had mild regurgitation.
Although at baseline more than 60% of the patients were New York Heart Association class III, 10% were class IV, and the rest were class II, at 30 days more than 30% were New York Heart Association class I, 40% were class II, and the rest were class III.
The 30-day all-cause mortality rate of 6.8% in the MITRAL study is roughly half that reported for ViR patients in the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry. Dr. Guerrero attributed this to refined procedural techniques and improved patient selection through the use of CT imaging and echocardiography.
Heart valve design changes, such as a longer inner skirt, might further improve the technical success rate for ViR, according to Dr. Guerrero, an interventional cardiologist at NorthShore University Health System in Evanston, Ill.
Picking the right ring
Given that studies show one-third of recipients of a surgical mitral ring or surgical mitral valve will require a repeat intervention within 10 years, she made a plea to surgeons: “If we are going to be treating patients with valve-in-ring TMVR, that means when surgeons do a repair they should pick a ring that is amenable to a ViR procedure. So don’t use flexible incomplete bands or very rigid rings because those are really difficult to treat later on. We should pick a ring thinking of the future. That ring is going to fail at some point, and when it fails it’s going to make our lives much easier if we’d picked the right ring.”
MAC TMVR needs more work
In the MAC arm of the MITRAL trial, 96 patients were screened so the researchers could find 30 candidates for TMVR. The 61 rejections were for high risk of left ventricular outflow tract obstruction (LVOTO), embolization, or both.
Fourteen patients underwent transseptal TMVR, and one with anatomy unsuitable for a transseptal procedure had a transapical approach. The other 15 patients had a transatrial surgical approach, which allows resection of the anterior leaflet to reduce the risk of LVOTO and placement of sutures to reduce the embolization risk. However, this came at the cost of increased mortality risk: Three of the five in-hospital deaths were in the transatrial TMVR group.
The technical success rate at exit from the cath lab in the MAC patients was 73%, with a 30-day procedural success rate of 46% and a 19% 30-day mortality. Three patients developed severe LVOTO with hemodynamic compromise.
One transseptal and one transapical TMVR were complicated by LVOTO, both treated by bailout alcohol septal ablation. This led Dr. Guerrero and her coinvestigators to the concept of preemptive alcohol septal ablation, which they used in seven patients deemed at high risk for LVOTO an average of 6 weeks prior to transseptal TMVR as a successful risk reduction strategy.
Survival climbing with operator experience
“In the early days of the TMVR MAC registry, the 30-day mortality rate was 37%. It came down to 22% in the middle third of the registry, then about 18% in the final third. Now we’ve got it down in MITRAL to 16.7%, but when you separate the rate in the transseptal versus the transatrial patients, it’s 13% versus 20%. The difference is not statistically significant, but it’s promising, and I think we are making great progress,” Dr. Guerrero said.
Safety and efficacy endpoints in MITRAL will be reported again at 1 year of follow-up.
The MITRAL trial was partially supported by Edwards Lifesciences. Dr. Guerrero reported receiving a research grant from that company and serving as a consultant to Tendyne Holdings/Abbott and on a speakers bureau for Abiomed.
SOURCE: Guerrero M. No abstract.
DENVER – Transseptal mitral valve implantation of an off-the-shelf, commercially available TAVR valve in high-surgical-risk patients with a failing surgically implanted mitral ring prosthesis has become a reasonable treatment strategy in light of the interim findings of the ground-breaking MITRAL trial, Mayra E. Guerrero, MD, said at the Transcatheter Cardiovascular Therapeutics annual educational meeting.
Her presentation of the preliminary results of the MITRAL (Mitral Implantation of Transcatheter Valves) trial showed this valve-in-ring (ViR) treatment strategy using the Sapien 3 valve was associated with low 30-day morbidity and mortality rates and impressive symptomatic improvement.
In contrast, another arm of the MITRAL trial showed that placement of the Sapien 3 TAVR valve in high-surgical-risk patients with severe mitral stenosis due to mitral annular calcification (MAC) of their native valve is a treatment strategy that’s not yet ready for prime time, she added at the meeting, which was sponsored by the Cardiovascular Research Foundation.
“Transcatheter mitral valve replacement in MAC is a challenging procedure associated with complications,” Dr. Guerrero observed. “It may become a reasonable alternative for high-surgical-risk patients with favorable anatomy, but techniques require further refinement.”
The ViR arm of the observational multicenter prospective MITRAL trial included 30 patients with extremely high surgical risk and either severe mitral stenosis as defined by a mitral valve area of 1.5 cm2 or less or moderate mitral stenosis plus severe mitral regurgitation. The most common type of failing ring was the Edwards Physio, in nine patients. Access for transcatheter mitral valve replacement (TMVR) was transseptal in 100% of patients.
The technical success rate at exit from the catheterization lab was 70%. The procedural success rate at 30 days was 62%.
Six patients required a second valve. This was mainly because of malpositioning of the first valve with resultant mitral regurgitation; however, this problem became a nonissue as operator experience grew. All six affected patients were alive at 30 days, and four of the six were New York Heart Association (NYHA) functional class I or II.
In-hospital and 30-day mortality rates were low. There was a single cardiovascular death and one noncardiac death in hospital, with no additional deaths through 30 days. No cases of stroke, acute MI, or valve embolization or thrombosis occurred. The mean mitral valve area at 30 days was 2.1 cm2, although three patients still had a mitral valve area of less 1.5 cm2. Three patients experienced acute renal failure requiring hemodialysis. Seventy-five percent of patients had no or trace mitral regurgitation by echocardiography; the rest had mild regurgitation.
Although at baseline more than 60% of the patients were New York Heart Association class III, 10% were class IV, and the rest were class II, at 30 days more than 30% were New York Heart Association class I, 40% were class II, and the rest were class III.
The 30-day all-cause mortality rate of 6.8% in the MITRAL study is roughly half that reported for ViR patients in the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry. Dr. Guerrero attributed this to refined procedural techniques and improved patient selection through the use of CT imaging and echocardiography.
Heart valve design changes, such as a longer inner skirt, might further improve the technical success rate for ViR, according to Dr. Guerrero, an interventional cardiologist at NorthShore University Health System in Evanston, Ill.
Picking the right ring
Given that studies show one-third of recipients of a surgical mitral ring or surgical mitral valve will require a repeat intervention within 10 years, she made a plea to surgeons: “If we are going to be treating patients with valve-in-ring TMVR, that means when surgeons do a repair they should pick a ring that is amenable to a ViR procedure. So don’t use flexible incomplete bands or very rigid rings because those are really difficult to treat later on. We should pick a ring thinking of the future. That ring is going to fail at some point, and when it fails it’s going to make our lives much easier if we’d picked the right ring.”
MAC TMVR needs more work
In the MAC arm of the MITRAL trial, 96 patients were screened so the researchers could find 30 candidates for TMVR. The 61 rejections were for high risk of left ventricular outflow tract obstruction (LVOTO), embolization, or both.
Fourteen patients underwent transseptal TMVR, and one with anatomy unsuitable for a transseptal procedure had a transapical approach. The other 15 patients had a transatrial surgical approach, which allows resection of the anterior leaflet to reduce the risk of LVOTO and placement of sutures to reduce the embolization risk. However, this came at the cost of increased mortality risk: Three of the five in-hospital deaths were in the transatrial TMVR group.
The technical success rate at exit from the cath lab in the MAC patients was 73%, with a 30-day procedural success rate of 46% and a 19% 30-day mortality. Three patients developed severe LVOTO with hemodynamic compromise.
One transseptal and one transapical TMVR were complicated by LVOTO, both treated by bailout alcohol septal ablation. This led Dr. Guerrero and her coinvestigators to the concept of preemptive alcohol septal ablation, which they used in seven patients deemed at high risk for LVOTO an average of 6 weeks prior to transseptal TMVR as a successful risk reduction strategy.
Survival climbing with operator experience
“In the early days of the TMVR MAC registry, the 30-day mortality rate was 37%. It came down to 22% in the middle third of the registry, then about 18% in the final third. Now we’ve got it down in MITRAL to 16.7%, but when you separate the rate in the transseptal versus the transatrial patients, it’s 13% versus 20%. The difference is not statistically significant, but it’s promising, and I think we are making great progress,” Dr. Guerrero said.
Safety and efficacy endpoints in MITRAL will be reported again at 1 year of follow-up.
The MITRAL trial was partially supported by Edwards Lifesciences. Dr. Guerrero reported receiving a research grant from that company and serving as a consultant to Tendyne Holdings/Abbott and on a speakers bureau for Abiomed.
SOURCE: Guerrero M. No abstract.
REPORTING FROM TCT 2017
Key clinical point: .
Major finding: Thirty-day all-cause mortality following a transcatheter valve-in-ring procedure in unacceptably high surgical-risk patients with severe mitral valve disease due to a failing annuloplasty ring was 6.8%.
Study details: This prospective observational study included 60 patients who underwent transcatheter mitral valve replacement for severe mitral valve disease, half due to a failed annuloplasty ring and half secondary to mitral annular calcification.
Disclosures: The MITRAL trial was partially supported by Edwards Lifesciences. The study presenter reported receiving a research grant from the company.
Source: Guerrero M. No abstract.
New frontier in TAVR is bicuspid disease
DENVER – Thirty-day transcatheter aortic valve replacement (TAVR) outcomes in real-world clinical practice using the Evolut R self-expanding valve were as good in patients treated for bicuspid disease as for tricuspid disease, according to a retrospective analysis of the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy (STS/ACC TVT) national registry.
“I’ve always been insecure about whether we have the right technology to be able to treat bicuspid disease. This registry data is reassuring to me that we might. I think it may be time to do a prospective registry for low-surgical-risk patients with bicuspid disease and see if we can emulate these kinds of results,” said Dr. Popma, the director of interventional cardiology at Beth Israel Deaconess Medical Center and a professor of medicine at Harvard Medical School, both in Boston.
“I think that the one limitation to recruitment in our low-risk TAVR trial is patients with bicuspid disease. Probably 25%-30% of low-risk patients are bicuspid, so we can’t include them right now in our low-risk trial,” he added at the meeting sponsored by the Cardiovascular Research Foundation.
Even though TAVR for patients with bicuspid disease is off-label, operators do perform the procedure. All of these cases are captured in the STS/ACC TVT registry. Dr. Popma reported on 6,717 patients who underwent TAVR with placement of the Evolut R valve at 305 U.S. centers during 2014-2016. The purpose of this retrospective study was to compare 30-day outcomes in the 191 TAVR patients with native valve bicuspid disease with the outcomes in the 6,526 with tricuspid disease.
The two groups were evenly matched in terms of key baseline characteristics, including aortic valve mean gradient, severity of aortic, mitral, and tricuspid regurgitation, and comorbid conditions – with the exception of coronary artery disease, which was present in 48% of the bicuspid group versus 65% of those with tricuspid disease. Also, the bicuspid disease group was younger by an average of nearly 9 years, and their mean baseline left ventricular ejection fraction of 52.5% was lower than the LVEF of 55.5% seen in the tricuspid group.
Procedure time averaged 126 minutes in the bicuspid group and 116 in the tricuspid group. Femoral access was utilized in 87% of the bicuspid patients and in 92% of tricuspid patients. The device was implanted successfully in 97% of the bicuspid group and in 99% of the tricuspid group. More than one valve was required in 3.7% of the bicuspid disease group, a rate similar to that in the tricuspid group. Total hospital length of stay was roughly 6 days in both groups.
Rates of symptomatic improvement at 30 days were closely similar in the two groups. Preprocedurally, two-thirds of patients in both groups had a New York Heart Association class III; at 30 days, however, that was true for a mere 2.4% of the bicuspid patients and 10.3% of the tricuspid patients. By day 30, 52% of the bicuspid group and 48% of the tricuspid group were NYHA class I.
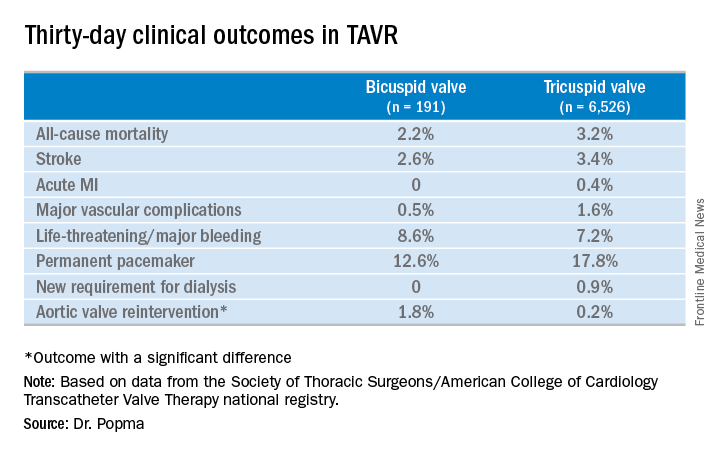
No or only trace aortic regurgitation was present at 30 days in 62% of the bicuspid group and in 61% of the tricuspid group, while mild aortic regurgitation was noted in 31% and 33%, respectively.
Thirty-day mean aortic valve gradient improved to a similar extent in the two groups: from a baseline of 47.2 mm Hg to 9.4 mm Hg in the bicuspid group and from 42.9 mm Hg to 7.5 mm Hg in the tricuspid group.
Dr. Popma noted that an earlier analysis he carried out comparing outcomes of TAVR using the earlier-generation CoreValve in bicuspid versus tricuspid disease showed suboptimal rates of paravalvular regurgitation and an increased need for multiple valves in the bicuspid group.
“The lesson is ‘Thank God we’ve got new technology!’ because the new technology has made a big difference for us,” the cardiologist observed. “We think that the advancement in the technique and the advancement in the valves is going to give us fairly comparable outcomes with Evolut in bicuspid and tricuspid patients.”
Discussant Hasan Jilaihawi, MD, a codirector of transcatheter valve therapy at New York University, pronounced the short-term outcomes in patients with bicuspid aortic valve disease “better than I would have expected,” adding that he, too, thinks it’s time for a prospective registry study of the Evolut valve in such patients.
Dr. Popma’s study was supported by Medtronic. He reported having received research grants from Medtronic and other medical device companies.
SOURCE: Popma JJ. TCT 2017.
DENVER – Thirty-day transcatheter aortic valve replacement (TAVR) outcomes in real-world clinical practice using the Evolut R self-expanding valve were as good in patients treated for bicuspid disease as for tricuspid disease, according to a retrospective analysis of the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy (STS/ACC TVT) national registry.
“I’ve always been insecure about whether we have the right technology to be able to treat bicuspid disease. This registry data is reassuring to me that we might. I think it may be time to do a prospective registry for low-surgical-risk patients with bicuspid disease and see if we can emulate these kinds of results,” said Dr. Popma, the director of interventional cardiology at Beth Israel Deaconess Medical Center and a professor of medicine at Harvard Medical School, both in Boston.
“I think that the one limitation to recruitment in our low-risk TAVR trial is patients with bicuspid disease. Probably 25%-30% of low-risk patients are bicuspid, so we can’t include them right now in our low-risk trial,” he added at the meeting sponsored by the Cardiovascular Research Foundation.
Even though TAVR for patients with bicuspid disease is off-label, operators do perform the procedure. All of these cases are captured in the STS/ACC TVT registry. Dr. Popma reported on 6,717 patients who underwent TAVR with placement of the Evolut R valve at 305 U.S. centers during 2014-2016. The purpose of this retrospective study was to compare 30-day outcomes in the 191 TAVR patients with native valve bicuspid disease with the outcomes in the 6,526 with tricuspid disease.
The two groups were evenly matched in terms of key baseline characteristics, including aortic valve mean gradient, severity of aortic, mitral, and tricuspid regurgitation, and comorbid conditions – with the exception of coronary artery disease, which was present in 48% of the bicuspid group versus 65% of those with tricuspid disease. Also, the bicuspid disease group was younger by an average of nearly 9 years, and their mean baseline left ventricular ejection fraction of 52.5% was lower than the LVEF of 55.5% seen in the tricuspid group.
Procedure time averaged 126 minutes in the bicuspid group and 116 in the tricuspid group. Femoral access was utilized in 87% of the bicuspid patients and in 92% of tricuspid patients. The device was implanted successfully in 97% of the bicuspid group and in 99% of the tricuspid group. More than one valve was required in 3.7% of the bicuspid disease group, a rate similar to that in the tricuspid group. Total hospital length of stay was roughly 6 days in both groups.
Rates of symptomatic improvement at 30 days were closely similar in the two groups. Preprocedurally, two-thirds of patients in both groups had a New York Heart Association class III; at 30 days, however, that was true for a mere 2.4% of the bicuspid patients and 10.3% of the tricuspid patients. By day 30, 52% of the bicuspid group and 48% of the tricuspid group were NYHA class I.

No or only trace aortic regurgitation was present at 30 days in 62% of the bicuspid group and in 61% of the tricuspid group, while mild aortic regurgitation was noted in 31% and 33%, respectively.
Thirty-day mean aortic valve gradient improved to a similar extent in the two groups: from a baseline of 47.2 mm Hg to 9.4 mm Hg in the bicuspid group and from 42.9 mm Hg to 7.5 mm Hg in the tricuspid group.
Dr. Popma noted that an earlier analysis he carried out comparing outcomes of TAVR using the earlier-generation CoreValve in bicuspid versus tricuspid disease showed suboptimal rates of paravalvular regurgitation and an increased need for multiple valves in the bicuspid group.
“The lesson is ‘Thank God we’ve got new technology!’ because the new technology has made a big difference for us,” the cardiologist observed. “We think that the advancement in the technique and the advancement in the valves is going to give us fairly comparable outcomes with Evolut in bicuspid and tricuspid patients.”
Discussant Hasan Jilaihawi, MD, a codirector of transcatheter valve therapy at New York University, pronounced the short-term outcomes in patients with bicuspid aortic valve disease “better than I would have expected,” adding that he, too, thinks it’s time for a prospective registry study of the Evolut valve in such patients.
Dr. Popma’s study was supported by Medtronic. He reported having received research grants from Medtronic and other medical device companies.
SOURCE: Popma JJ. TCT 2017.
DENVER – Thirty-day transcatheter aortic valve replacement (TAVR) outcomes in real-world clinical practice using the Evolut R self-expanding valve were as good in patients treated for bicuspid disease as for tricuspid disease, according to a retrospective analysis of the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy (STS/ACC TVT) national registry.
“I’ve always been insecure about whether we have the right technology to be able to treat bicuspid disease. This registry data is reassuring to me that we might. I think it may be time to do a prospective registry for low-surgical-risk patients with bicuspid disease and see if we can emulate these kinds of results,” said Dr. Popma, the director of interventional cardiology at Beth Israel Deaconess Medical Center and a professor of medicine at Harvard Medical School, both in Boston.
“I think that the one limitation to recruitment in our low-risk TAVR trial is patients with bicuspid disease. Probably 25%-30% of low-risk patients are bicuspid, so we can’t include them right now in our low-risk trial,” he added at the meeting sponsored by the Cardiovascular Research Foundation.
Even though TAVR for patients with bicuspid disease is off-label, operators do perform the procedure. All of these cases are captured in the STS/ACC TVT registry. Dr. Popma reported on 6,717 patients who underwent TAVR with placement of the Evolut R valve at 305 U.S. centers during 2014-2016. The purpose of this retrospective study was to compare 30-day outcomes in the 191 TAVR patients with native valve bicuspid disease with the outcomes in the 6,526 with tricuspid disease.
The two groups were evenly matched in terms of key baseline characteristics, including aortic valve mean gradient, severity of aortic, mitral, and tricuspid regurgitation, and comorbid conditions – with the exception of coronary artery disease, which was present in 48% of the bicuspid group versus 65% of those with tricuspid disease. Also, the bicuspid disease group was younger by an average of nearly 9 years, and their mean baseline left ventricular ejection fraction of 52.5% was lower than the LVEF of 55.5% seen in the tricuspid group.
Procedure time averaged 126 minutes in the bicuspid group and 116 in the tricuspid group. Femoral access was utilized in 87% of the bicuspid patients and in 92% of tricuspid patients. The device was implanted successfully in 97% of the bicuspid group and in 99% of the tricuspid group. More than one valve was required in 3.7% of the bicuspid disease group, a rate similar to that in the tricuspid group. Total hospital length of stay was roughly 6 days in both groups.
Rates of symptomatic improvement at 30 days were closely similar in the two groups. Preprocedurally, two-thirds of patients in both groups had a New York Heart Association class III; at 30 days, however, that was true for a mere 2.4% of the bicuspid patients and 10.3% of the tricuspid patients. By day 30, 52% of the bicuspid group and 48% of the tricuspid group were NYHA class I.

No or only trace aortic regurgitation was present at 30 days in 62% of the bicuspid group and in 61% of the tricuspid group, while mild aortic regurgitation was noted in 31% and 33%, respectively.
Thirty-day mean aortic valve gradient improved to a similar extent in the two groups: from a baseline of 47.2 mm Hg to 9.4 mm Hg in the bicuspid group and from 42.9 mm Hg to 7.5 mm Hg in the tricuspid group.
Dr. Popma noted that an earlier analysis he carried out comparing outcomes of TAVR using the earlier-generation CoreValve in bicuspid versus tricuspid disease showed suboptimal rates of paravalvular regurgitation and an increased need for multiple valves in the bicuspid group.
“The lesson is ‘Thank God we’ve got new technology!’ because the new technology has made a big difference for us,” the cardiologist observed. “We think that the advancement in the technique and the advancement in the valves is going to give us fairly comparable outcomes with Evolut in bicuspid and tricuspid patients.”
Discussant Hasan Jilaihawi, MD, a codirector of transcatheter valve therapy at New York University, pronounced the short-term outcomes in patients with bicuspid aortic valve disease “better than I would have expected,” adding that he, too, thinks it’s time for a prospective registry study of the Evolut valve in such patients.
Dr. Popma’s study was supported by Medtronic. He reported having received research grants from Medtronic and other medical device companies.
SOURCE: Popma JJ. TCT 2017.
REPORTING FROM TCT 2017
Key clinical point:
Major finding: Thirty-day clinical outcomes and symptomatic improvement were reassuringly similar both in TAVR patients who received the Evolut R valve for tricuspid disease and off-label for bicuspid disease.
Study details: This was a retrospective U.S. national registry study comparing 30-day outcomes in 191 TAVR patients with native valve bicuspid disease and 6,526 with tricuspid disease, all of whom underwent TAVR with placement of the Evolut R valve.
Disclosures: The study presenter reported having received research grants from Medtronic, the study sponsor, as well as other medical device companies.
Source: Popma JJ. TCT 2017.
VAMPIRE 3: Embolic filter for primary PCI reduces MACE
DENVER – Distal embolic protection using a filter device during primary PCI for acute MI significantly reduced the incidence of the no-reflow phenomenon and serious adverse events in the randomized VAMPIRE 3 trial.
The reason VAMPIRE 3 (Vacuum Aspiration Thrombus Removal) was a positive trial when other studies of distal embolic protection during coronary intervention have failed was that VAMPIRE 3 targeted a high-risk, high-benefit population: the subset of acute MI patients with attenuated, high-risk coronary plaque identified by intravascular ultrasound, Kiyoshi Hibi, MD, explained at the Transcatheter Cardiovascular Therapeutics annual educational meeting.
Also, the filter device used in the trial was easily deployed and problem free, which doesn’t appear to be true of distal protection devices used in some earlier trials.
Discussants agreed with his conclusion, but predicted distal protection during primary PCI for acute coronary syndrome is unlikely to catch on with U.S. interventional cardiologists anytime soon. That’s because, for the most part, they don’t perform advanced imaging in the setting of primary PCI for ACS.
VAMPIRE 3 was a multicenter Japanese study involving 200 patients undergoing primary PCI for acute MI who displayed intravascular ultrasound evidence of attenuated plaque at least 5 mm in length as defined by images showing backward signal attenuation of 180 degrees or more behind a noncalcified plaque. Participants were randomized to placement of the Filtrap distal embolic protection device manufactured by Nipro of Tokyo or to no filter during their procedure, which in all cases entailed the use of a thrombus aspiration catheter.
The primary endpoint was the incidence of the no-reflow phenomenon, an adverse event previously shown by German investigators to be associated with a 66% increase in 5-year mortality (J Am Coll Cardiol. 2010 May 25;55[21]:2383-9). In VAMPIRE 3, the no-reflow phenomenon occurred in 26.5% of patients randomized to distal protection and 41.7% of those managed conventionally. The secondary endpoint – the corrected TIMI frame count – was 23.0 in the distal protection group, significantly better than the 30.5 in control patients, the cardiologist reported at the meeting, which was sponsored by the Cardiovascular Research Foundation.
The rate of in-hospital adverse events – a composite of death, cardiac arrest, cardiogenic shock, and ischemic stroke adjudicated by an independent committee – was 1% in the distal protection group and significantly higher at 8.3% in conventionally managed controls. All five cases of cardiac arrest/cardiogenic shock occurred in patients who didn’t receive the prophylactic filter, he continued.
In a previous study, Dr. Hibi and his coworkers showed that the incidence of the no-reflow phenomenon in a series of 179 acute MI patients was 18%. More importantly, the incidence was 71% among patients with attenuated plaque of 5 mm or more, compared with 10%-11% among those with less or no attenuated plaque (JACC Cardiovasc Interv. 2010 May;3[5]:540-9).
“I am convinced by these results,” discussant Jonathan M. Hill, MD, said about VAMPIRE 3, “but the concern is the dependency on imaging. All of these cases were entirely dependent on an imaging diagnosis.
“Certainly in the acute setting, I think Japanese practice is the extreme, with greater than 80% penetration for intravascular imaging, compared to less than 10% in the United States. So to implement your protocol would be difficult unless there was acceptance of the requirement for intravascular imaging,” added Dr. Hill, an interventional cardiologist at King’s College London.
The prevailing philosophy among American cardiologists is that time is heart muscle. The priority is to open the blocked coronary vessel as quickly as possible.
“We don’t use that much advanced imaging,“ agreed David J. Cohen, MD, director of cardiovascular research at Saint Luke’s Mid America Heart Institute in Kansas City, Mo.
“It seems like the difference between this trial and previous ones that have not used advanced imaging was the use of intravascular imaging to identify the optimal group of patients that might benefit from this. So I think until that practice changes in the United States, I suspect this is going to get very limited use. I don’t see these devices ramping up quickly for coronary applications,” he predicted.
VAMPIRE 3 was sponsored by Yokohama City University Medical Center and Teikyo University. Dr. Hibi reported serving as a consultant to Nipro and Boston Scientific.
SOURCE: Hibi K. TCT 2017
DENVER – Distal embolic protection using a filter device during primary PCI for acute MI significantly reduced the incidence of the no-reflow phenomenon and serious adverse events in the randomized VAMPIRE 3 trial.
The reason VAMPIRE 3 (Vacuum Aspiration Thrombus Removal) was a positive trial when other studies of distal embolic protection during coronary intervention have failed was that VAMPIRE 3 targeted a high-risk, high-benefit population: the subset of acute MI patients with attenuated, high-risk coronary plaque identified by intravascular ultrasound, Kiyoshi Hibi, MD, explained at the Transcatheter Cardiovascular Therapeutics annual educational meeting.
Also, the filter device used in the trial was easily deployed and problem free, which doesn’t appear to be true of distal protection devices used in some earlier trials.
Discussants agreed with his conclusion, but predicted distal protection during primary PCI for acute coronary syndrome is unlikely to catch on with U.S. interventional cardiologists anytime soon. That’s because, for the most part, they don’t perform advanced imaging in the setting of primary PCI for ACS.
VAMPIRE 3 was a multicenter Japanese study involving 200 patients undergoing primary PCI for acute MI who displayed intravascular ultrasound evidence of attenuated plaque at least 5 mm in length as defined by images showing backward signal attenuation of 180 degrees or more behind a noncalcified plaque. Participants were randomized to placement of the Filtrap distal embolic protection device manufactured by Nipro of Tokyo or to no filter during their procedure, which in all cases entailed the use of a thrombus aspiration catheter.
The primary endpoint was the incidence of the no-reflow phenomenon, an adverse event previously shown by German investigators to be associated with a 66% increase in 5-year mortality (J Am Coll Cardiol. 2010 May 25;55[21]:2383-9). In VAMPIRE 3, the no-reflow phenomenon occurred in 26.5% of patients randomized to distal protection and 41.7% of those managed conventionally. The secondary endpoint – the corrected TIMI frame count – was 23.0 in the distal protection group, significantly better than the 30.5 in control patients, the cardiologist reported at the meeting, which was sponsored by the Cardiovascular Research Foundation.
The rate of in-hospital adverse events – a composite of death, cardiac arrest, cardiogenic shock, and ischemic stroke adjudicated by an independent committee – was 1% in the distal protection group and significantly higher at 8.3% in conventionally managed controls. All five cases of cardiac arrest/cardiogenic shock occurred in patients who didn’t receive the prophylactic filter, he continued.
In a previous study, Dr. Hibi and his coworkers showed that the incidence of the no-reflow phenomenon in a series of 179 acute MI patients was 18%. More importantly, the incidence was 71% among patients with attenuated plaque of 5 mm or more, compared with 10%-11% among those with less or no attenuated plaque (JACC Cardiovasc Interv. 2010 May;3[5]:540-9).
“I am convinced by these results,” discussant Jonathan M. Hill, MD, said about VAMPIRE 3, “but the concern is the dependency on imaging. All of these cases were entirely dependent on an imaging diagnosis.
“Certainly in the acute setting, I think Japanese practice is the extreme, with greater than 80% penetration for intravascular imaging, compared to less than 10% in the United States. So to implement your protocol would be difficult unless there was acceptance of the requirement for intravascular imaging,” added Dr. Hill, an interventional cardiologist at King’s College London.
The prevailing philosophy among American cardiologists is that time is heart muscle. The priority is to open the blocked coronary vessel as quickly as possible.
“We don’t use that much advanced imaging,“ agreed David J. Cohen, MD, director of cardiovascular research at Saint Luke’s Mid America Heart Institute in Kansas City, Mo.
“It seems like the difference between this trial and previous ones that have not used advanced imaging was the use of intravascular imaging to identify the optimal group of patients that might benefit from this. So I think until that practice changes in the United States, I suspect this is going to get very limited use. I don’t see these devices ramping up quickly for coronary applications,” he predicted.
VAMPIRE 3 was sponsored by Yokohama City University Medical Center and Teikyo University. Dr. Hibi reported serving as a consultant to Nipro and Boston Scientific.
SOURCE: Hibi K. TCT 2017
DENVER – Distal embolic protection using a filter device during primary PCI for acute MI significantly reduced the incidence of the no-reflow phenomenon and serious adverse events in the randomized VAMPIRE 3 trial.
The reason VAMPIRE 3 (Vacuum Aspiration Thrombus Removal) was a positive trial when other studies of distal embolic protection during coronary intervention have failed was that VAMPIRE 3 targeted a high-risk, high-benefit population: the subset of acute MI patients with attenuated, high-risk coronary plaque identified by intravascular ultrasound, Kiyoshi Hibi, MD, explained at the Transcatheter Cardiovascular Therapeutics annual educational meeting.
Also, the filter device used in the trial was easily deployed and problem free, which doesn’t appear to be true of distal protection devices used in some earlier trials.
Discussants agreed with his conclusion, but predicted distal protection during primary PCI for acute coronary syndrome is unlikely to catch on with U.S. interventional cardiologists anytime soon. That’s because, for the most part, they don’t perform advanced imaging in the setting of primary PCI for ACS.
VAMPIRE 3 was a multicenter Japanese study involving 200 patients undergoing primary PCI for acute MI who displayed intravascular ultrasound evidence of attenuated plaque at least 5 mm in length as defined by images showing backward signal attenuation of 180 degrees or more behind a noncalcified plaque. Participants were randomized to placement of the Filtrap distal embolic protection device manufactured by Nipro of Tokyo or to no filter during their procedure, which in all cases entailed the use of a thrombus aspiration catheter.
The primary endpoint was the incidence of the no-reflow phenomenon, an adverse event previously shown by German investigators to be associated with a 66% increase in 5-year mortality (J Am Coll Cardiol. 2010 May 25;55[21]:2383-9). In VAMPIRE 3, the no-reflow phenomenon occurred in 26.5% of patients randomized to distal protection and 41.7% of those managed conventionally. The secondary endpoint – the corrected TIMI frame count – was 23.0 in the distal protection group, significantly better than the 30.5 in control patients, the cardiologist reported at the meeting, which was sponsored by the Cardiovascular Research Foundation.
The rate of in-hospital adverse events – a composite of death, cardiac arrest, cardiogenic shock, and ischemic stroke adjudicated by an independent committee – was 1% in the distal protection group and significantly higher at 8.3% in conventionally managed controls. All five cases of cardiac arrest/cardiogenic shock occurred in patients who didn’t receive the prophylactic filter, he continued.
In a previous study, Dr. Hibi and his coworkers showed that the incidence of the no-reflow phenomenon in a series of 179 acute MI patients was 18%. More importantly, the incidence was 71% among patients with attenuated plaque of 5 mm or more, compared with 10%-11% among those with less or no attenuated plaque (JACC Cardiovasc Interv. 2010 May;3[5]:540-9).
“I am convinced by these results,” discussant Jonathan M. Hill, MD, said about VAMPIRE 3, “but the concern is the dependency on imaging. All of these cases were entirely dependent on an imaging diagnosis.
“Certainly in the acute setting, I think Japanese practice is the extreme, with greater than 80% penetration for intravascular imaging, compared to less than 10% in the United States. So to implement your protocol would be difficult unless there was acceptance of the requirement for intravascular imaging,” added Dr. Hill, an interventional cardiologist at King’s College London.
The prevailing philosophy among American cardiologists is that time is heart muscle. The priority is to open the blocked coronary vessel as quickly as possible.
“We don’t use that much advanced imaging,“ agreed David J. Cohen, MD, director of cardiovascular research at Saint Luke’s Mid America Heart Institute in Kansas City, Mo.
“It seems like the difference between this trial and previous ones that have not used advanced imaging was the use of intravascular imaging to identify the optimal group of patients that might benefit from this. So I think until that practice changes in the United States, I suspect this is going to get very limited use. I don’t see these devices ramping up quickly for coronary applications,” he predicted.
VAMPIRE 3 was sponsored by Yokohama City University Medical Center and Teikyo University. Dr. Hibi reported serving as a consultant to Nipro and Boston Scientific.
SOURCE: Hibi K. TCT 2017
REPORTING FROM TCT 2017
Key clinical point:
Major finding: The no-reflow phenomenon occurred in 26.5% of patients randomized to distal protection and 41.7% of those managed conventionally.
Study details: This randomized multicenter trial included 200 acute MI patients.
Disclosures: VAMPIRE 3 was sponsored by Yokohama City University Medical Center and Teikyo University. The presenter reported serving as a consultant to Nipro and Boston Scientific.
















