User login
Diagnosing and managing diabetes and depression
SAN DIEGO – Nearly 350 years ago, British physician Thomas Willis wrote that diabetes seemed often to occur in patients who were experiencing “significant life stress, sadness, or long sorrow.” That, according to Ellen D. Mandel, DMH, MPA, MS, PA-C, RDN, CDE, a clinical professor at Pace University in New York City, was an important insight into the link between mind and body in patients with diabetes.
“As clinicians, we should be worried about mental illness in our patients with diabetes,” Dr. Mandel, a physician assistant educator, said during a presentation at the Metabolic & Endocrine Disease Summit by Global Academy for Medical Education.
In particular, she said, – and vice versa.
Dr. Mandel pointed to findings suggesting that 11% of patients with diabetes show signs of clinical depression, which is higher than in the general population, with many more believed to have subclinical depression (Diabetes Care. 2015;38[4]:551-60).
Anxiety can be a key factor in trying to understand how diabetes might contribute to depression. “Diabetes is a very stressful condition ... [and patients] may be fatigued and exhausted.” On top of that, they have to make nutrition changes, or at least pay attention to their diet and overall care, all of which can have a cumulatively negative impact on patient well-being.
Conversely, depression can contribute to diabetes. “They kind of go hand in hand,” she said, pointing to depression’s ability to disrupt appetite, diminish energy, and boost levels of cortisol.
Among the findings that provide evidence of a link between diabetes and depression are those from a study in which investigators estimated that for every 1-point increase in depression symptoms, the risk of diabetes will go up by as much as 5% (Clin Diabetes Endocrinol. 2018 Jan 4. doi: 10.1186/s40842-017-0052-1). Moreover, a 2013 review linked the combination of diabetes and depression to an adjusted 1.5-fold increase in risk of all-cause death (PLoS One. 2013 Mar 5. doi: 10.1371/journal.pone.0057058).
Dr. Mandel offered these tips about diagnosing depression in patients with diabetes and helping them feel comfortable:
- Put yourself in the patient’s shoes. “One of the biggest barriers to referring patients to diabetic education is that they don’t want to have to admit to a group that they have diabetes. They keep it to themselves, to their own detriment. In addition, there’s a lot of worry about insurance.” Patients with diabetes often have self-esteem issues and financial or insurance challenges, all of which need to be factored in when working with them, Dr. Mandel said.
- Ask questions and use screening tools. Two simple questions are helpful in starting a conversation and gathering useful information: Over the past 2 weeks, have you often been bothered by [having] little interest or pleasure in doing things? What about being bothered by feeling down, depressed, or hopeless? If the patient answers “yes” to either of these questions, it will be a positive screen, and two “no” answers will be a negative screen. With the “yes” responses, one should follow-up with a screening tool – typically, the one approved by your institution. Dr. Mandel also highlighted the Patient Health Questionnaire depression scale (PHQ-9), which is available online, or the brief, two-item Diabetes Distress Scale (DDS2) questionnaire.
- Keep your own language in mind. “The way you communicate with your patients can elevate their feeling about themselves or destroy how they feel about themselves,” Dr. Mandel said. “We’re trying to stop calling people with diabetes ‘diabetics.’ People don’t want to be labeled like that. Don’t blame yourself if you use this language, but work to make the changes,” Dr. Mandel suggested.
- Watch out for other forms of bias. Beware of unconsciously stereotyping your patients. “It affects how people relate to you, how they adhere to your suggestions, and how much they’ll trust [and confide in] you, which can have clinical implications,” Dr. Mandel said.
Global Academy and this news organization are owned by the same parent company. Dr. Mandel has no disclosures.
SAN DIEGO – Nearly 350 years ago, British physician Thomas Willis wrote that diabetes seemed often to occur in patients who were experiencing “significant life stress, sadness, or long sorrow.” That, according to Ellen D. Mandel, DMH, MPA, MS, PA-C, RDN, CDE, a clinical professor at Pace University in New York City, was an important insight into the link between mind and body in patients with diabetes.
“As clinicians, we should be worried about mental illness in our patients with diabetes,” Dr. Mandel, a physician assistant educator, said during a presentation at the Metabolic & Endocrine Disease Summit by Global Academy for Medical Education.
In particular, she said, – and vice versa.
Dr. Mandel pointed to findings suggesting that 11% of patients with diabetes show signs of clinical depression, which is higher than in the general population, with many more believed to have subclinical depression (Diabetes Care. 2015;38[4]:551-60).
Anxiety can be a key factor in trying to understand how diabetes might contribute to depression. “Diabetes is a very stressful condition ... [and patients] may be fatigued and exhausted.” On top of that, they have to make nutrition changes, or at least pay attention to their diet and overall care, all of which can have a cumulatively negative impact on patient well-being.
Conversely, depression can contribute to diabetes. “They kind of go hand in hand,” she said, pointing to depression’s ability to disrupt appetite, diminish energy, and boost levels of cortisol.
Among the findings that provide evidence of a link between diabetes and depression are those from a study in which investigators estimated that for every 1-point increase in depression symptoms, the risk of diabetes will go up by as much as 5% (Clin Diabetes Endocrinol. 2018 Jan 4. doi: 10.1186/s40842-017-0052-1). Moreover, a 2013 review linked the combination of diabetes and depression to an adjusted 1.5-fold increase in risk of all-cause death (PLoS One. 2013 Mar 5. doi: 10.1371/journal.pone.0057058).
Dr. Mandel offered these tips about diagnosing depression in patients with diabetes and helping them feel comfortable:
- Put yourself in the patient’s shoes. “One of the biggest barriers to referring patients to diabetic education is that they don’t want to have to admit to a group that they have diabetes. They keep it to themselves, to their own detriment. In addition, there’s a lot of worry about insurance.” Patients with diabetes often have self-esteem issues and financial or insurance challenges, all of which need to be factored in when working with them, Dr. Mandel said.
- Ask questions and use screening tools. Two simple questions are helpful in starting a conversation and gathering useful information: Over the past 2 weeks, have you often been bothered by [having] little interest or pleasure in doing things? What about being bothered by feeling down, depressed, or hopeless? If the patient answers “yes” to either of these questions, it will be a positive screen, and two “no” answers will be a negative screen. With the “yes” responses, one should follow-up with a screening tool – typically, the one approved by your institution. Dr. Mandel also highlighted the Patient Health Questionnaire depression scale (PHQ-9), which is available online, or the brief, two-item Diabetes Distress Scale (DDS2) questionnaire.
- Keep your own language in mind. “The way you communicate with your patients can elevate their feeling about themselves or destroy how they feel about themselves,” Dr. Mandel said. “We’re trying to stop calling people with diabetes ‘diabetics.’ People don’t want to be labeled like that. Don’t blame yourself if you use this language, but work to make the changes,” Dr. Mandel suggested.
- Watch out for other forms of bias. Beware of unconsciously stereotyping your patients. “It affects how people relate to you, how they adhere to your suggestions, and how much they’ll trust [and confide in] you, which can have clinical implications,” Dr. Mandel said.
Global Academy and this news organization are owned by the same parent company. Dr. Mandel has no disclosures.
SAN DIEGO – Nearly 350 years ago, British physician Thomas Willis wrote that diabetes seemed often to occur in patients who were experiencing “significant life stress, sadness, or long sorrow.” That, according to Ellen D. Mandel, DMH, MPA, MS, PA-C, RDN, CDE, a clinical professor at Pace University in New York City, was an important insight into the link between mind and body in patients with diabetes.
“As clinicians, we should be worried about mental illness in our patients with diabetes,” Dr. Mandel, a physician assistant educator, said during a presentation at the Metabolic & Endocrine Disease Summit by Global Academy for Medical Education.
In particular, she said, – and vice versa.
Dr. Mandel pointed to findings suggesting that 11% of patients with diabetes show signs of clinical depression, which is higher than in the general population, with many more believed to have subclinical depression (Diabetes Care. 2015;38[4]:551-60).
Anxiety can be a key factor in trying to understand how diabetes might contribute to depression. “Diabetes is a very stressful condition ... [and patients] may be fatigued and exhausted.” On top of that, they have to make nutrition changes, or at least pay attention to their diet and overall care, all of which can have a cumulatively negative impact on patient well-being.
Conversely, depression can contribute to diabetes. “They kind of go hand in hand,” she said, pointing to depression’s ability to disrupt appetite, diminish energy, and boost levels of cortisol.
Among the findings that provide evidence of a link between diabetes and depression are those from a study in which investigators estimated that for every 1-point increase in depression symptoms, the risk of diabetes will go up by as much as 5% (Clin Diabetes Endocrinol. 2018 Jan 4. doi: 10.1186/s40842-017-0052-1). Moreover, a 2013 review linked the combination of diabetes and depression to an adjusted 1.5-fold increase in risk of all-cause death (PLoS One. 2013 Mar 5. doi: 10.1371/journal.pone.0057058).
Dr. Mandel offered these tips about diagnosing depression in patients with diabetes and helping them feel comfortable:
- Put yourself in the patient’s shoes. “One of the biggest barriers to referring patients to diabetic education is that they don’t want to have to admit to a group that they have diabetes. They keep it to themselves, to their own detriment. In addition, there’s a lot of worry about insurance.” Patients with diabetes often have self-esteem issues and financial or insurance challenges, all of which need to be factored in when working with them, Dr. Mandel said.
- Ask questions and use screening tools. Two simple questions are helpful in starting a conversation and gathering useful information: Over the past 2 weeks, have you often been bothered by [having] little interest or pleasure in doing things? What about being bothered by feeling down, depressed, or hopeless? If the patient answers “yes” to either of these questions, it will be a positive screen, and two “no” answers will be a negative screen. With the “yes” responses, one should follow-up with a screening tool – typically, the one approved by your institution. Dr. Mandel also highlighted the Patient Health Questionnaire depression scale (PHQ-9), which is available online, or the brief, two-item Diabetes Distress Scale (DDS2) questionnaire.
- Keep your own language in mind. “The way you communicate with your patients can elevate their feeling about themselves or destroy how they feel about themselves,” Dr. Mandel said. “We’re trying to stop calling people with diabetes ‘diabetics.’ People don’t want to be labeled like that. Don’t blame yourself if you use this language, but work to make the changes,” Dr. Mandel suggested.
- Watch out for other forms of bias. Beware of unconsciously stereotyping your patients. “It affects how people relate to you, how they adhere to your suggestions, and how much they’ll trust [and confide in] you, which can have clinical implications,” Dr. Mandel said.
Global Academy and this news organization are owned by the same parent company. Dr. Mandel has no disclosures.
EXPERT ANALYSIS FROM MEDS 2019
Oral semaglutide monotherapy delivers HbA1c improvements in type 2 diabetes
Oral semaglutide monotherapy was superior to placebo for improving glycated hemoglobin (HbA1c) levels at all doses tested in adults with type 2 diabetes who had been previously insufficiently managed with diet and exercise, according to findings from a global, randomized trial.
The drug also showed dose-dependent weight loss, with a statistically significant effect on body weight, compared with placebo, at higher doses.
To date, the glucagon-like peptide–1 receptor agonist has been available as weekly subcutaneous shots for patients with type 2 diabetes, and in that form they have been shown to be effective in reducing HbA1c, inducing weight loss, and lowering the risk of cardiovascular events in patients with cardiovascular disease or those who are at high risk for it, wrote Vanita R. Aroda, MD, of Brigham and Women’s Hospital, Boston, and colleagues. The report is in Diabetes Care.
The novel oral semaglutide tablet is designed to enhance medication absorption, and the pharmacokinetics and dosage were established in phase 2 studies, they noted.
In the phase 3 Peptide Innovation for Early Diabetes Treatment 1 (PIONEER 1) study, Dr. Aroda and colleagues randomized 703 adults with type 2 diabetes to receive either 3 mg, 7 mg, or 14 mg of oral semaglutide daily, or placebo. The average age of the patients was 55 years, about half were women, and the average baseline HbA1c was 8.0% (64 mmol/mol). The primary endpoint was change in HbA1c level from baseline to week 26, and the secondary endpoint was change in body weight over the same period.
After 26 weeks of once-daily treatment, patients in semaglutide group showed significant reductions in HbA1c from baseline with all three doses: –0.6% (3 mg), –0.9% (7 mg), and –1.1% (14 mg), with P less than .001 for all, based on an intention-to-treat analysis. Similar results occurred using an on-treatment analysis, with differences of –0.7%, –1.2%, and –1.4%, respectively, for the three doses.
In addition, patients in all dose groups achieved the secondary endpoint of reduction in body weight, compared with placebo, from baseline to 26 weeks based on both types of analyses. “Significantly more patients achieved body weight loss of at least 5% with oral semaglutide at 7 mg and 14 mg, compared with placebo,” Dr. Aroda and colleagues wrote (intention-to-treat: –0.1 for 3 mg daily [P = .87], –0.9 for 7 mg [P = .09], –2.3 for 14 mg [P less than .001]; and on-treatment: –0.2 for 3 mg [P = .71], –1.0 for 7 mg [P = .01], –2.6 for 14 mg [P less than .001]).
The overall incidence of adverse events and serious adverse events was similar in the treatment and placebo groups, with the most frequent being nausea and diarrhea. No deaths occurred among patients on the medication.
The findings were limited by several factors, including a patient population that had a relatively short duration of diabetes (mean, 3.5 years) and that the oral semaglutide was used as first-line monotherapy, without first using metformin, the researchers noted. However, oral semaglutide “achieved clinically meaningful and superior glucose lowering,” compared with placebo, at all three doses, they wrote.
“Ongoing additional studies in the PIONEER program will further define the effect when used in combination with other glucose-lowering therapies and in other populations of interest, such as those with high cardiovascular risk or renal impairment,” they emphasized
Novo Nordisk funded the study. The lead author disclosed relationships with Novo Nordisk, and several coauthors disclosed relationships with or employment by the company.
SOURCE: Aroda VR et al. Diabetes Care. 2019 Jul. doi: 10.2337/dc19-0749.
Oral semaglutide monotherapy was superior to placebo for improving glycated hemoglobin (HbA1c) levels at all doses tested in adults with type 2 diabetes who had been previously insufficiently managed with diet and exercise, according to findings from a global, randomized trial.
The drug also showed dose-dependent weight loss, with a statistically significant effect on body weight, compared with placebo, at higher doses.
To date, the glucagon-like peptide–1 receptor agonist has been available as weekly subcutaneous shots for patients with type 2 diabetes, and in that form they have been shown to be effective in reducing HbA1c, inducing weight loss, and lowering the risk of cardiovascular events in patients with cardiovascular disease or those who are at high risk for it, wrote Vanita R. Aroda, MD, of Brigham and Women’s Hospital, Boston, and colleagues. The report is in Diabetes Care.
The novel oral semaglutide tablet is designed to enhance medication absorption, and the pharmacokinetics and dosage were established in phase 2 studies, they noted.
In the phase 3 Peptide Innovation for Early Diabetes Treatment 1 (PIONEER 1) study, Dr. Aroda and colleagues randomized 703 adults with type 2 diabetes to receive either 3 mg, 7 mg, or 14 mg of oral semaglutide daily, or placebo. The average age of the patients was 55 years, about half were women, and the average baseline HbA1c was 8.0% (64 mmol/mol). The primary endpoint was change in HbA1c level from baseline to week 26, and the secondary endpoint was change in body weight over the same period.
After 26 weeks of once-daily treatment, patients in semaglutide group showed significant reductions in HbA1c from baseline with all three doses: –0.6% (3 mg), –0.9% (7 mg), and –1.1% (14 mg), with P less than .001 for all, based on an intention-to-treat analysis. Similar results occurred using an on-treatment analysis, with differences of –0.7%, –1.2%, and –1.4%, respectively, for the three doses.
In addition, patients in all dose groups achieved the secondary endpoint of reduction in body weight, compared with placebo, from baseline to 26 weeks based on both types of analyses. “Significantly more patients achieved body weight loss of at least 5% with oral semaglutide at 7 mg and 14 mg, compared with placebo,” Dr. Aroda and colleagues wrote (intention-to-treat: –0.1 for 3 mg daily [P = .87], –0.9 for 7 mg [P = .09], –2.3 for 14 mg [P less than .001]; and on-treatment: –0.2 for 3 mg [P = .71], –1.0 for 7 mg [P = .01], –2.6 for 14 mg [P less than .001]).
The overall incidence of adverse events and serious adverse events was similar in the treatment and placebo groups, with the most frequent being nausea and diarrhea. No deaths occurred among patients on the medication.
The findings were limited by several factors, including a patient population that had a relatively short duration of diabetes (mean, 3.5 years) and that the oral semaglutide was used as first-line monotherapy, without first using metformin, the researchers noted. However, oral semaglutide “achieved clinically meaningful and superior glucose lowering,” compared with placebo, at all three doses, they wrote.
“Ongoing additional studies in the PIONEER program will further define the effect when used in combination with other glucose-lowering therapies and in other populations of interest, such as those with high cardiovascular risk or renal impairment,” they emphasized
Novo Nordisk funded the study. The lead author disclosed relationships with Novo Nordisk, and several coauthors disclosed relationships with or employment by the company.
SOURCE: Aroda VR et al. Diabetes Care. 2019 Jul. doi: 10.2337/dc19-0749.
Oral semaglutide monotherapy was superior to placebo for improving glycated hemoglobin (HbA1c) levels at all doses tested in adults with type 2 diabetes who had been previously insufficiently managed with diet and exercise, according to findings from a global, randomized trial.
The drug also showed dose-dependent weight loss, with a statistically significant effect on body weight, compared with placebo, at higher doses.
To date, the glucagon-like peptide–1 receptor agonist has been available as weekly subcutaneous shots for patients with type 2 diabetes, and in that form they have been shown to be effective in reducing HbA1c, inducing weight loss, and lowering the risk of cardiovascular events in patients with cardiovascular disease or those who are at high risk for it, wrote Vanita R. Aroda, MD, of Brigham and Women’s Hospital, Boston, and colleagues. The report is in Diabetes Care.
The novel oral semaglutide tablet is designed to enhance medication absorption, and the pharmacokinetics and dosage were established in phase 2 studies, they noted.
In the phase 3 Peptide Innovation for Early Diabetes Treatment 1 (PIONEER 1) study, Dr. Aroda and colleagues randomized 703 adults with type 2 diabetes to receive either 3 mg, 7 mg, or 14 mg of oral semaglutide daily, or placebo. The average age of the patients was 55 years, about half were women, and the average baseline HbA1c was 8.0% (64 mmol/mol). The primary endpoint was change in HbA1c level from baseline to week 26, and the secondary endpoint was change in body weight over the same period.
After 26 weeks of once-daily treatment, patients in semaglutide group showed significant reductions in HbA1c from baseline with all three doses: –0.6% (3 mg), –0.9% (7 mg), and –1.1% (14 mg), with P less than .001 for all, based on an intention-to-treat analysis. Similar results occurred using an on-treatment analysis, with differences of –0.7%, –1.2%, and –1.4%, respectively, for the three doses.
In addition, patients in all dose groups achieved the secondary endpoint of reduction in body weight, compared with placebo, from baseline to 26 weeks based on both types of analyses. “Significantly more patients achieved body weight loss of at least 5% with oral semaglutide at 7 mg and 14 mg, compared with placebo,” Dr. Aroda and colleagues wrote (intention-to-treat: –0.1 for 3 mg daily [P = .87], –0.9 for 7 mg [P = .09], –2.3 for 14 mg [P less than .001]; and on-treatment: –0.2 for 3 mg [P = .71], –1.0 for 7 mg [P = .01], –2.6 for 14 mg [P less than .001]).
The overall incidence of adverse events and serious adverse events was similar in the treatment and placebo groups, with the most frequent being nausea and diarrhea. No deaths occurred among patients on the medication.
The findings were limited by several factors, including a patient population that had a relatively short duration of diabetes (mean, 3.5 years) and that the oral semaglutide was used as first-line monotherapy, without first using metformin, the researchers noted. However, oral semaglutide “achieved clinically meaningful and superior glucose lowering,” compared with placebo, at all three doses, they wrote.
“Ongoing additional studies in the PIONEER program will further define the effect when used in combination with other glucose-lowering therapies and in other populations of interest, such as those with high cardiovascular risk or renal impairment,” they emphasized
Novo Nordisk funded the study. The lead author disclosed relationships with Novo Nordisk, and several coauthors disclosed relationships with or employment by the company.
SOURCE: Aroda VR et al. Diabetes Care. 2019 Jul. doi: 10.2337/dc19-0749.
FROM DIABETES CARE
Mediterranean diet tied to improved cognition in type 2 diabetes
People with type 2 diabetes whose diet followed a “Mediterranean” pattern – high in vegetables, legumes, fish, and unsaturated fats – saw global cognitive improvements over a 2-year period, compared with individuals with different eating patterns, even if the latter incorporated healthy dietary features. In addition, effective glycemic control seemed to have a role in sustaining the benefits associated with the Mediterranean-type diet.
Adults without type 2 diabetes, meanwhile, did not see the cognitive improvements associated with a Mediterranean diet, suggesting that the pathways linking diet to cognition may be different for individuals with and without diabetes, according to Josiemer Mattei, PhD, of the Harvard T.H. Chan School of Public Health in Boston and colleagues.
The investigators used data from the Boston Puerto Rican Health Study, a longitudinal cohort of about 1,499 adults aged 45-75 years who lived in Boston and identified as Puerto Rican, for their research, which was published in Diabetes Care.
At baseline, participants were administered a questionnaire to capture their eating patterns. Four diet-quality scores – Mediterranean Diet Score, Healthy Eating Index, Alternate Healthy Eating Index, and DASH (Dietary Approaches to Stop Hypertension) were analyzed. The participants were also screened for diabetes, and nearly 40% of them were found to have type 2 diabetes at baseline (74% uncontrolled). They underwent a battery of cognitive tests, including the Mini-Mental State Exam and tests for verbal fluency, executive function, word recognition, and figure copying. The study endpoints included 2-year change in global cognitive function as well as executive and memory function. At 2 years, data was available for 913 participants.
Among participants with type 2 diabetes, greater adherence to a Mediterranean-style diet was significantly associated with a higher positive change at the 2-year follow-up in global cognitive function score (0.027 [SD, 0.011]; P = .016), the Mini-Mental State Exam, and other individual tests. The association was significant for those who were under glycemic control at baseline and who remained stable or improved over 2 years, but not for those with poor or worsening glycemic control.
“The Mediterranean diet explained as much or more of the variability in predicting changes in cognitive function in our study as did age, especially for participants with type 2 diabetes under glycemic control. ... This dietary pattern may provide more cognitive benefits [in this patient group] than other modifiable and nonmodifiable factors,” the authors wrote in their analysis. They stressed that a Mediterranean dietary pattern can be realized through foods and dishes that are already standard in many Puerto Rican households.
In participants who did not have diabetes, improvement in memory function measures was seen in association with a Mediterranean diet, but also with adherence to other eating patterns that are deemed healthy. That suggests that for this subgroup, any evidence-based healthy diet – not just the Mediterranean diet – may have some benefits for memory function.
“Dietary recommendations for cognitive health may need to be tailored for individuals with versus without type 2 diabetes,” the authors concluded.
Dr. Mattei and colleagues acknowledged as a limitation of their study its observational design.
The study received funding from the National Heart, Lung, and Blood Institute; the National Institute on Aging; and Harvard University. The authors reported no financial conflicts of interest.
SOURCE: Mattei et al. Diabetes Care. 2019;42(8):1372-9.
People with type 2 diabetes whose diet followed a “Mediterranean” pattern – high in vegetables, legumes, fish, and unsaturated fats – saw global cognitive improvements over a 2-year period, compared with individuals with different eating patterns, even if the latter incorporated healthy dietary features. In addition, effective glycemic control seemed to have a role in sustaining the benefits associated with the Mediterranean-type diet.
Adults without type 2 diabetes, meanwhile, did not see the cognitive improvements associated with a Mediterranean diet, suggesting that the pathways linking diet to cognition may be different for individuals with and without diabetes, according to Josiemer Mattei, PhD, of the Harvard T.H. Chan School of Public Health in Boston and colleagues.
The investigators used data from the Boston Puerto Rican Health Study, a longitudinal cohort of about 1,499 adults aged 45-75 years who lived in Boston and identified as Puerto Rican, for their research, which was published in Diabetes Care.
At baseline, participants were administered a questionnaire to capture their eating patterns. Four diet-quality scores – Mediterranean Diet Score, Healthy Eating Index, Alternate Healthy Eating Index, and DASH (Dietary Approaches to Stop Hypertension) were analyzed. The participants were also screened for diabetes, and nearly 40% of them were found to have type 2 diabetes at baseline (74% uncontrolled). They underwent a battery of cognitive tests, including the Mini-Mental State Exam and tests for verbal fluency, executive function, word recognition, and figure copying. The study endpoints included 2-year change in global cognitive function as well as executive and memory function. At 2 years, data was available for 913 participants.
Among participants with type 2 diabetes, greater adherence to a Mediterranean-style diet was significantly associated with a higher positive change at the 2-year follow-up in global cognitive function score (0.027 [SD, 0.011]; P = .016), the Mini-Mental State Exam, and other individual tests. The association was significant for those who were under glycemic control at baseline and who remained stable or improved over 2 years, but not for those with poor or worsening glycemic control.
“The Mediterranean diet explained as much or more of the variability in predicting changes in cognitive function in our study as did age, especially for participants with type 2 diabetes under glycemic control. ... This dietary pattern may provide more cognitive benefits [in this patient group] than other modifiable and nonmodifiable factors,” the authors wrote in their analysis. They stressed that a Mediterranean dietary pattern can be realized through foods and dishes that are already standard in many Puerto Rican households.
In participants who did not have diabetes, improvement in memory function measures was seen in association with a Mediterranean diet, but also with adherence to other eating patterns that are deemed healthy. That suggests that for this subgroup, any evidence-based healthy diet – not just the Mediterranean diet – may have some benefits for memory function.
“Dietary recommendations for cognitive health may need to be tailored for individuals with versus without type 2 diabetes,” the authors concluded.
Dr. Mattei and colleagues acknowledged as a limitation of their study its observational design.
The study received funding from the National Heart, Lung, and Blood Institute; the National Institute on Aging; and Harvard University. The authors reported no financial conflicts of interest.
SOURCE: Mattei et al. Diabetes Care. 2019;42(8):1372-9.
People with type 2 diabetes whose diet followed a “Mediterranean” pattern – high in vegetables, legumes, fish, and unsaturated fats – saw global cognitive improvements over a 2-year period, compared with individuals with different eating patterns, even if the latter incorporated healthy dietary features. In addition, effective glycemic control seemed to have a role in sustaining the benefits associated with the Mediterranean-type diet.
Adults without type 2 diabetes, meanwhile, did not see the cognitive improvements associated with a Mediterranean diet, suggesting that the pathways linking diet to cognition may be different for individuals with and without diabetes, according to Josiemer Mattei, PhD, of the Harvard T.H. Chan School of Public Health in Boston and colleagues.
The investigators used data from the Boston Puerto Rican Health Study, a longitudinal cohort of about 1,499 adults aged 45-75 years who lived in Boston and identified as Puerto Rican, for their research, which was published in Diabetes Care.
At baseline, participants were administered a questionnaire to capture their eating patterns. Four diet-quality scores – Mediterranean Diet Score, Healthy Eating Index, Alternate Healthy Eating Index, and DASH (Dietary Approaches to Stop Hypertension) were analyzed. The participants were also screened for diabetes, and nearly 40% of them were found to have type 2 diabetes at baseline (74% uncontrolled). They underwent a battery of cognitive tests, including the Mini-Mental State Exam and tests for verbal fluency, executive function, word recognition, and figure copying. The study endpoints included 2-year change in global cognitive function as well as executive and memory function. At 2 years, data was available for 913 participants.
Among participants with type 2 diabetes, greater adherence to a Mediterranean-style diet was significantly associated with a higher positive change at the 2-year follow-up in global cognitive function score (0.027 [SD, 0.011]; P = .016), the Mini-Mental State Exam, and other individual tests. The association was significant for those who were under glycemic control at baseline and who remained stable or improved over 2 years, but not for those with poor or worsening glycemic control.
“The Mediterranean diet explained as much or more of the variability in predicting changes in cognitive function in our study as did age, especially for participants with type 2 diabetes under glycemic control. ... This dietary pattern may provide more cognitive benefits [in this patient group] than other modifiable and nonmodifiable factors,” the authors wrote in their analysis. They stressed that a Mediterranean dietary pattern can be realized through foods and dishes that are already standard in many Puerto Rican households.
In participants who did not have diabetes, improvement in memory function measures was seen in association with a Mediterranean diet, but also with adherence to other eating patterns that are deemed healthy. That suggests that for this subgroup, any evidence-based healthy diet – not just the Mediterranean diet – may have some benefits for memory function.
“Dietary recommendations for cognitive health may need to be tailored for individuals with versus without type 2 diabetes,” the authors concluded.
Dr. Mattei and colleagues acknowledged as a limitation of their study its observational design.
The study received funding from the National Heart, Lung, and Blood Institute; the National Institute on Aging; and Harvard University. The authors reported no financial conflicts of interest.
SOURCE: Mattei et al. Diabetes Care. 2019;42(8):1372-9.
FROM DIABETES CARE
Perils of ‘type’-casting: When adult-onset diabetes isn’t what you think it is
SAN DIEGO – We all know about type 1 and type 2 diabetes. But when an adult patient comes in with symptoms suggestive of diabetes, it is never a good idea to assume it’s either one or the other. In fact, said physician assistant Ji Hyun “CJ” Chun, PA-C, MPAS, BC-ADM, there are plenty of other possibilities from cancer, to monogenetic diabetes, to a condition informally known as type 1.5.
“In most cases, it will be type 1 or type 2, but don’t default everything,” Mr. Chun said at the Metabolic & Endocrine Disease Summit by Global Academy for Medical Education.
He offered the following advice on the diagnosis of adult-onset diabetes:
- Don’t forget the 5% ... and the other 5%. In adults, an estimated 90% of cases of diabetes are type 2, but 5% are type 1 and another 5% are secondary to other conditions, said Mr. Chun, who is based at OptumCare Medical Group, Laguna Niguel, Calif., and has served as president of the American Society of Endocrine PAs. In the past, age seemed to be an important tool for diagnosis, because younger patients typically had type 1 diabetes and older patients typically had type 2, he said. But age alone is no longer useful for diagnosis. Cases of type 2 diabetes are much more common in children these days because of the prevalence of obesity in that population, and an estimated 60% of cases of type 1 disease are diagnosed after the age of 20. In fact, patients may develop type 1 into their 30s, 40s, or 50s, he said, depending on the severity of their autoimmunity.
- Keep ‘type 1.5’ in mind. Latent autoimmune diabetes in adults (LADA), also known as type 1.5, is a slowly developing subtype of type 1 diabetes, Mr. Chun said. There is reason to suspect LADA in lean patients, those younger than 50, and those with personal or family histories of autoimmunity, Mr. Chun said.
- Consider monogenetic diabetes. Many conditions can cause secondary diabetes, among them, monogenetic diabetes, which is caused by a single genetic mutation, whereas type 1 and type 2 diabetes are caused by multiple mutations. Monogenetic diabetes causes an estimated 1%-2% of diabetes cases, said Mr. Chun. Research findings have suggested that it is most likely to be misdiagnosed in younger adults, and that patients may go many years without receiving a correct diagnosis. Maturity-onset diabetes of the young (MODY) is a kind of monogenetic diabetes and typically occurs before the age of 25. There are many subtypes, of which one – MODY 2 – requires no treatment at all. In those patients, said Mr. Chun, “you do nothing. You leave them alone.” It is important to keep in mind that the genetic testing for MODY is expensive, Mr Chun cautioned. Some labs charge between $5,000 and $7,000 for panels, so “look for labs that perform cheaper tests,” he advised, adding that he has found a lab that charges just $250.
- Watch out for cancer. There is a long list of other possible causes of secondary diabetes, including Cushing’s syndrome, hyperthyroidism, hemochromatosis (iron overload), and pancreatic cancer. Mr. Chun said he has lost three patients to pancreatic cancer, while two other patients did well. “Keep in mind that these cases aren’t that common, but they’re there.” He suggested that pancreatic cancer should be considered in patients with rapid onset or worsening of diabetes without known cause, abnormal weight loss, abnormal liver/biliary studies, and jaundice.
Global Academy and this news organization are owned by the same parent company. Mr. Chun disclosed that he is on the AstraZeneca speakers bureau and the Sanofi advisory board.
SAN DIEGO – We all know about type 1 and type 2 diabetes. But when an adult patient comes in with symptoms suggestive of diabetes, it is never a good idea to assume it’s either one or the other. In fact, said physician assistant Ji Hyun “CJ” Chun, PA-C, MPAS, BC-ADM, there are plenty of other possibilities from cancer, to monogenetic diabetes, to a condition informally known as type 1.5.
“In most cases, it will be type 1 or type 2, but don’t default everything,” Mr. Chun said at the Metabolic & Endocrine Disease Summit by Global Academy for Medical Education.
He offered the following advice on the diagnosis of adult-onset diabetes:
- Don’t forget the 5% ... and the other 5%. In adults, an estimated 90% of cases of diabetes are type 2, but 5% are type 1 and another 5% are secondary to other conditions, said Mr. Chun, who is based at OptumCare Medical Group, Laguna Niguel, Calif., and has served as president of the American Society of Endocrine PAs. In the past, age seemed to be an important tool for diagnosis, because younger patients typically had type 1 diabetes and older patients typically had type 2, he said. But age alone is no longer useful for diagnosis. Cases of type 2 diabetes are much more common in children these days because of the prevalence of obesity in that population, and an estimated 60% of cases of type 1 disease are diagnosed after the age of 20. In fact, patients may develop type 1 into their 30s, 40s, or 50s, he said, depending on the severity of their autoimmunity.
- Keep ‘type 1.5’ in mind. Latent autoimmune diabetes in adults (LADA), also known as type 1.5, is a slowly developing subtype of type 1 diabetes, Mr. Chun said. There is reason to suspect LADA in lean patients, those younger than 50, and those with personal or family histories of autoimmunity, Mr. Chun said.
- Consider monogenetic diabetes. Many conditions can cause secondary diabetes, among them, monogenetic diabetes, which is caused by a single genetic mutation, whereas type 1 and type 2 diabetes are caused by multiple mutations. Monogenetic diabetes causes an estimated 1%-2% of diabetes cases, said Mr. Chun. Research findings have suggested that it is most likely to be misdiagnosed in younger adults, and that patients may go many years without receiving a correct diagnosis. Maturity-onset diabetes of the young (MODY) is a kind of monogenetic diabetes and typically occurs before the age of 25. There are many subtypes, of which one – MODY 2 – requires no treatment at all. In those patients, said Mr. Chun, “you do nothing. You leave them alone.” It is important to keep in mind that the genetic testing for MODY is expensive, Mr Chun cautioned. Some labs charge between $5,000 and $7,000 for panels, so “look for labs that perform cheaper tests,” he advised, adding that he has found a lab that charges just $250.
- Watch out for cancer. There is a long list of other possible causes of secondary diabetes, including Cushing’s syndrome, hyperthyroidism, hemochromatosis (iron overload), and pancreatic cancer. Mr. Chun said he has lost three patients to pancreatic cancer, while two other patients did well. “Keep in mind that these cases aren’t that common, but they’re there.” He suggested that pancreatic cancer should be considered in patients with rapid onset or worsening of diabetes without known cause, abnormal weight loss, abnormal liver/biliary studies, and jaundice.
Global Academy and this news organization are owned by the same parent company. Mr. Chun disclosed that he is on the AstraZeneca speakers bureau and the Sanofi advisory board.
SAN DIEGO – We all know about type 1 and type 2 diabetes. But when an adult patient comes in with symptoms suggestive of diabetes, it is never a good idea to assume it’s either one or the other. In fact, said physician assistant Ji Hyun “CJ” Chun, PA-C, MPAS, BC-ADM, there are plenty of other possibilities from cancer, to monogenetic diabetes, to a condition informally known as type 1.5.
“In most cases, it will be type 1 or type 2, but don’t default everything,” Mr. Chun said at the Metabolic & Endocrine Disease Summit by Global Academy for Medical Education.
He offered the following advice on the diagnosis of adult-onset diabetes:
- Don’t forget the 5% ... and the other 5%. In adults, an estimated 90% of cases of diabetes are type 2, but 5% are type 1 and another 5% are secondary to other conditions, said Mr. Chun, who is based at OptumCare Medical Group, Laguna Niguel, Calif., and has served as president of the American Society of Endocrine PAs. In the past, age seemed to be an important tool for diagnosis, because younger patients typically had type 1 diabetes and older patients typically had type 2, he said. But age alone is no longer useful for diagnosis. Cases of type 2 diabetes are much more common in children these days because of the prevalence of obesity in that population, and an estimated 60% of cases of type 1 disease are diagnosed after the age of 20. In fact, patients may develop type 1 into their 30s, 40s, or 50s, he said, depending on the severity of their autoimmunity.
- Keep ‘type 1.5’ in mind. Latent autoimmune diabetes in adults (LADA), also known as type 1.5, is a slowly developing subtype of type 1 diabetes, Mr. Chun said. There is reason to suspect LADA in lean patients, those younger than 50, and those with personal or family histories of autoimmunity, Mr. Chun said.
- Consider monogenetic diabetes. Many conditions can cause secondary diabetes, among them, monogenetic diabetes, which is caused by a single genetic mutation, whereas type 1 and type 2 diabetes are caused by multiple mutations. Monogenetic diabetes causes an estimated 1%-2% of diabetes cases, said Mr. Chun. Research findings have suggested that it is most likely to be misdiagnosed in younger adults, and that patients may go many years without receiving a correct diagnosis. Maturity-onset diabetes of the young (MODY) is a kind of monogenetic diabetes and typically occurs before the age of 25. There are many subtypes, of which one – MODY 2 – requires no treatment at all. In those patients, said Mr. Chun, “you do nothing. You leave them alone.” It is important to keep in mind that the genetic testing for MODY is expensive, Mr Chun cautioned. Some labs charge between $5,000 and $7,000 for panels, so “look for labs that perform cheaper tests,” he advised, adding that he has found a lab that charges just $250.
- Watch out for cancer. There is a long list of other possible causes of secondary diabetes, including Cushing’s syndrome, hyperthyroidism, hemochromatosis (iron overload), and pancreatic cancer. Mr. Chun said he has lost three patients to pancreatic cancer, while two other patients did well. “Keep in mind that these cases aren’t that common, but they’re there.” He suggested that pancreatic cancer should be considered in patients with rapid onset or worsening of diabetes without known cause, abnormal weight loss, abnormal liver/biliary studies, and jaundice.
Global Academy and this news organization are owned by the same parent company. Mr. Chun disclosed that he is on the AstraZeneca speakers bureau and the Sanofi advisory board.
REPORTING FROM MEDS 2019
Urine albumin testing is crucial for patients at risk for CKD, but drop the 24-hour urine test
SAN DIEGO – Whatever you do, don’t order a 24-hour urine test. Do encourage “pork holidays.” And choose between an ACE inhibitor and an ARB – don’t give them both to a single patient, according to Kim Zuber, PA-C, MS, a nephrology physician assistant from St. Petersburg, Fla., in a presentation about kidney disease, hypertension, and diabetes at the Metabolic & Endocrine Disease Summit, sponsored by Global Academy for Medical Education.
Ms. Zuber, who is the executive director of the American Academy of Nephrology PAs and the outreach chair of the National Kidney Foundation, outlined some approaches for the diagnosis, management, and treatment of chronic kidney disease (CKD) with comorbid hypertension and diabetes.
- Use the right urine test. Ms. Zuber said, although it’s often not performed. In fact, research suggests that most Medicare patients with diabetes, hypertension, or both do not have this test, she said. Order a urine albumin-to-creatinine ratio (UACR) test at least once a year in at-risk patients, she recommended, and more frequently if they show signs of abnormal values. But be aware, she said, some labs might refer to the test as microalbuminuria instead of UACR, and be prepared to calculate the UACR yourself if your institution provides only albumin and creatinine levels. Also watch out for mix-ups regarding UACR measurements. Nephrotic-range proteinuria starts at 3 g/dL or 3,000 mg/dL, she said, and residents often confuse those two sets of units. “Many have gotten in trouble with that,” she said.
- Don’t go near a 24-hour urine test. Thinking about ordering a 24-hour urine test that requires a patients to collect all their urine for a day? Think again. “We’ve been telling you almost 20 years not to do this,” Ms. Zuber said. These tests “are unreliable, and they don’t work.”
- Don’t focus on tight blood pressure control. Studies provide little insight into the ideal blood pressure readings for patients with diabetes and CKD, according to Ms. Zuber, but some findings suggest that tight control can be harmful to the kidneys. She urges her patients to treat hypertension in part by embracing lifestyle change. “I tell them that if you improve your lifestyle, you can give up one of your drugs. When they average 15 drugs a day, that becomes popular.” Physical activity, the DASH diet, salt restriction, moderate alcohol consumption, weight loss, stress reduction, and smoking cessation can all lower blood pressure, she added.
- Talk up the “pork holiday.” For patients with hypertension, “sodium restriction is huge,” Ms. Zuber said, especially among black patients. She urges her patients to take “pork holidays”, that is, eat pork only four times a year, on holidays such as the Fourth of July. She also urges them to prepare food in ways that begin with B, as in bake, boil, and barbecue. “You’ll notice that ‘fry’ doesn’t start with a B.”
- Try an ACE inhibitor or an ARB, but not both. In patients with hypertension plus diabetes and/or CKD, Ms. Zuber suggests using an angiotensin-converting enzyme inhibitor or angiotensin receptor blockers, but not both. One or the other can improve albuminuria, she said, but together they can boost risk of CKD, hyperkalemia, and hypotension.
Consider factors such as formularies and personal experience when trying to decide which drug to use, she said. If a patient still has hypertension, consider a diuretic and then move to a calcium channel or beta blocker. However, she cautioned, although beta blockers, they can cause erectile dysfunction.
Global Academy for Medical Education and this news organization are owned by the same parent company. Ms. Zuber reported no disclosures.
SAN DIEGO – Whatever you do, don’t order a 24-hour urine test. Do encourage “pork holidays.” And choose between an ACE inhibitor and an ARB – don’t give them both to a single patient, according to Kim Zuber, PA-C, MS, a nephrology physician assistant from St. Petersburg, Fla., in a presentation about kidney disease, hypertension, and diabetes at the Metabolic & Endocrine Disease Summit, sponsored by Global Academy for Medical Education.
Ms. Zuber, who is the executive director of the American Academy of Nephrology PAs and the outreach chair of the National Kidney Foundation, outlined some approaches for the diagnosis, management, and treatment of chronic kidney disease (CKD) with comorbid hypertension and diabetes.
- Use the right urine test. Ms. Zuber said, although it’s often not performed. In fact, research suggests that most Medicare patients with diabetes, hypertension, or both do not have this test, she said. Order a urine albumin-to-creatinine ratio (UACR) test at least once a year in at-risk patients, she recommended, and more frequently if they show signs of abnormal values. But be aware, she said, some labs might refer to the test as microalbuminuria instead of UACR, and be prepared to calculate the UACR yourself if your institution provides only albumin and creatinine levels. Also watch out for mix-ups regarding UACR measurements. Nephrotic-range proteinuria starts at 3 g/dL or 3,000 mg/dL, she said, and residents often confuse those two sets of units. “Many have gotten in trouble with that,” she said.
- Don’t go near a 24-hour urine test. Thinking about ordering a 24-hour urine test that requires a patients to collect all their urine for a day? Think again. “We’ve been telling you almost 20 years not to do this,” Ms. Zuber said. These tests “are unreliable, and they don’t work.”
- Don’t focus on tight blood pressure control. Studies provide little insight into the ideal blood pressure readings for patients with diabetes and CKD, according to Ms. Zuber, but some findings suggest that tight control can be harmful to the kidneys. She urges her patients to treat hypertension in part by embracing lifestyle change. “I tell them that if you improve your lifestyle, you can give up one of your drugs. When they average 15 drugs a day, that becomes popular.” Physical activity, the DASH diet, salt restriction, moderate alcohol consumption, weight loss, stress reduction, and smoking cessation can all lower blood pressure, she added.
- Talk up the “pork holiday.” For patients with hypertension, “sodium restriction is huge,” Ms. Zuber said, especially among black patients. She urges her patients to take “pork holidays”, that is, eat pork only four times a year, on holidays such as the Fourth of July. She also urges them to prepare food in ways that begin with B, as in bake, boil, and barbecue. “You’ll notice that ‘fry’ doesn’t start with a B.”
- Try an ACE inhibitor or an ARB, but not both. In patients with hypertension plus diabetes and/or CKD, Ms. Zuber suggests using an angiotensin-converting enzyme inhibitor or angiotensin receptor blockers, but not both. One or the other can improve albuminuria, she said, but together they can boost risk of CKD, hyperkalemia, and hypotension.
Consider factors such as formularies and personal experience when trying to decide which drug to use, she said. If a patient still has hypertension, consider a diuretic and then move to a calcium channel or beta blocker. However, she cautioned, although beta blockers, they can cause erectile dysfunction.
Global Academy for Medical Education and this news organization are owned by the same parent company. Ms. Zuber reported no disclosures.
SAN DIEGO – Whatever you do, don’t order a 24-hour urine test. Do encourage “pork holidays.” And choose between an ACE inhibitor and an ARB – don’t give them both to a single patient, according to Kim Zuber, PA-C, MS, a nephrology physician assistant from St. Petersburg, Fla., in a presentation about kidney disease, hypertension, and diabetes at the Metabolic & Endocrine Disease Summit, sponsored by Global Academy for Medical Education.
Ms. Zuber, who is the executive director of the American Academy of Nephrology PAs and the outreach chair of the National Kidney Foundation, outlined some approaches for the diagnosis, management, and treatment of chronic kidney disease (CKD) with comorbid hypertension and diabetes.
- Use the right urine test. Ms. Zuber said, although it’s often not performed. In fact, research suggests that most Medicare patients with diabetes, hypertension, or both do not have this test, she said. Order a urine albumin-to-creatinine ratio (UACR) test at least once a year in at-risk patients, she recommended, and more frequently if they show signs of abnormal values. But be aware, she said, some labs might refer to the test as microalbuminuria instead of UACR, and be prepared to calculate the UACR yourself if your institution provides only albumin and creatinine levels. Also watch out for mix-ups regarding UACR measurements. Nephrotic-range proteinuria starts at 3 g/dL or 3,000 mg/dL, she said, and residents often confuse those two sets of units. “Many have gotten in trouble with that,” she said.
- Don’t go near a 24-hour urine test. Thinking about ordering a 24-hour urine test that requires a patients to collect all their urine for a day? Think again. “We’ve been telling you almost 20 years not to do this,” Ms. Zuber said. These tests “are unreliable, and they don’t work.”
- Don’t focus on tight blood pressure control. Studies provide little insight into the ideal blood pressure readings for patients with diabetes and CKD, according to Ms. Zuber, but some findings suggest that tight control can be harmful to the kidneys. She urges her patients to treat hypertension in part by embracing lifestyle change. “I tell them that if you improve your lifestyle, you can give up one of your drugs. When they average 15 drugs a day, that becomes popular.” Physical activity, the DASH diet, salt restriction, moderate alcohol consumption, weight loss, stress reduction, and smoking cessation can all lower blood pressure, she added.
- Talk up the “pork holiday.” For patients with hypertension, “sodium restriction is huge,” Ms. Zuber said, especially among black patients. She urges her patients to take “pork holidays”, that is, eat pork only four times a year, on holidays such as the Fourth of July. She also urges them to prepare food in ways that begin with B, as in bake, boil, and barbecue. “You’ll notice that ‘fry’ doesn’t start with a B.”
- Try an ACE inhibitor or an ARB, but not both. In patients with hypertension plus diabetes and/or CKD, Ms. Zuber suggests using an angiotensin-converting enzyme inhibitor or angiotensin receptor blockers, but not both. One or the other can improve albuminuria, she said, but together they can boost risk of CKD, hyperkalemia, and hypotension.
Consider factors such as formularies and personal experience when trying to decide which drug to use, she said. If a patient still has hypertension, consider a diuretic and then move to a calcium channel or beta blocker. However, she cautioned, although beta blockers, they can cause erectile dysfunction.
Global Academy for Medical Education and this news organization are owned by the same parent company. Ms. Zuber reported no disclosures.
EXPERT ANALYSIS FROM MEDS 2019
Endoscopic duodenal mucosal resection found effective for some patients with T2D
Among patients with suboptimally controlled type 2 diabetes who use oral glucose-lowering medication, endoscopic duodenal mucosal resection (DMR) can be implemented safely and effectively, results from a multicenter, international, phase 2 study demonstrated.
“DMR elicited a substantial improvement in parameters of glycemia as well as a decrease in liver transaminase levels at 24 weeks, which was sustained at 12 months post procedure,” researchers led by Annieke C.G. van Baar, MD, wrote in a study published online in Gut. “These findings were also associated with an improvement in patients’ diabetes treatment satisfaction.”
For the study, Dr. van Baar, of the department of gastroenterology and hepatology at Amsterdam University Medical Center, and colleagues at seven clinical sites enrolled 46 patients with type 2 diabetes who were on stable glucose-lowering medication to undergo DMR. The procedure “involves circumferential hydrothermal ablation of the duodenal mucosa resulting in subsequent regeneration of the mucosa,” they wrote. “Before ablation, the mucosa is lifted with saline to protect the outer layers of the duodenum.” DMR was performed under either general anesthesia or deep sedation with propofol by a single endoscopist at each site with extensive experience in therapeutic upper GI endoscopy and guidewire management.
The mean age of the study participants was 55 years and 63% were male. Of the 46 patients, 37 (80%) underwent complete DMR and results were reported for 36 of them. A total of 24 patients had at least one adverse event related to DMR (52%), mostly GI symptoms such as diarrhea, abdominal pain, nausea, and oropharyngeal pain. Of these, 81% were mild. One serious adverse event was considered to be related to the procedure. “This concerned a patient with general malaise, mild fever, and increased C-reactive protein level on the first day after DMR,” the researchers wrote. “The mild fever resolved within 24 hours and [C-reactive protein] level normalized within 3 days.” No unanticipated adverse events were reported.
During follow-up measures taken 24 weeks after their DMR, hemoglobin A1c fell by a mean of 10 mmol/mol (P less than .001), fasting plasma glucose by 1.7 mmol/L (P less than .001), and the Homeostatic Model Assessment of Insulin Resistance improved significantly (P less than .001). In addition, the procedure conferred a moderate reduction in weight (a mean loss of 2.5 kg) and a decrease in hepatic transaminase levels. The effects were sustained at 12 months.
“While the majority of patients showed a durable glycemic response over 12 months, a minority exhibited less benefit from DMR and required additional glucose-lowering medication at 24 weeks,” the researchers wrote. “Of note, approximately two-thirds of the patients who required addition of antidiabetic medication in the latter phase of study had undergone insulin secretagogue medication withdrawal at screening. For future study, it may not be necessary to discontinue these medications before DMR, and this will allow an even more precise measure of DMR effect.”
Dr. van Baar and colleagues acknowledged certain limitations of the phase 2 study, including its open-label, uncontrolled design. “The results of this multicenter study need to be confirmed in a proper controlled study. Nevertheless, this study forms the requisite solid foundation for further research, and controlled studies are currently underway.”
The study was funded by Fractyl Laboratories. Dr. van Baar reported having no financial disclosures. Four of the study authors reported having financial relationships with numerous pharmaceutical and device companies.
SOURCE: van Baar ACG et al. Gut. 2019 Jul 22. doi: 10.1136/gutjnl-2019-318349.
Among patients with suboptimally controlled type 2 diabetes who use oral glucose-lowering medication, endoscopic duodenal mucosal resection (DMR) can be implemented safely and effectively, results from a multicenter, international, phase 2 study demonstrated.
“DMR elicited a substantial improvement in parameters of glycemia as well as a decrease in liver transaminase levels at 24 weeks, which was sustained at 12 months post procedure,” researchers led by Annieke C.G. van Baar, MD, wrote in a study published online in Gut. “These findings were also associated with an improvement in patients’ diabetes treatment satisfaction.”
For the study, Dr. van Baar, of the department of gastroenterology and hepatology at Amsterdam University Medical Center, and colleagues at seven clinical sites enrolled 46 patients with type 2 diabetes who were on stable glucose-lowering medication to undergo DMR. The procedure “involves circumferential hydrothermal ablation of the duodenal mucosa resulting in subsequent regeneration of the mucosa,” they wrote. “Before ablation, the mucosa is lifted with saline to protect the outer layers of the duodenum.” DMR was performed under either general anesthesia or deep sedation with propofol by a single endoscopist at each site with extensive experience in therapeutic upper GI endoscopy and guidewire management.
The mean age of the study participants was 55 years and 63% were male. Of the 46 patients, 37 (80%) underwent complete DMR and results were reported for 36 of them. A total of 24 patients had at least one adverse event related to DMR (52%), mostly GI symptoms such as diarrhea, abdominal pain, nausea, and oropharyngeal pain. Of these, 81% were mild. One serious adverse event was considered to be related to the procedure. “This concerned a patient with general malaise, mild fever, and increased C-reactive protein level on the first day after DMR,” the researchers wrote. “The mild fever resolved within 24 hours and [C-reactive protein] level normalized within 3 days.” No unanticipated adverse events were reported.
During follow-up measures taken 24 weeks after their DMR, hemoglobin A1c fell by a mean of 10 mmol/mol (P less than .001), fasting plasma glucose by 1.7 mmol/L (P less than .001), and the Homeostatic Model Assessment of Insulin Resistance improved significantly (P less than .001). In addition, the procedure conferred a moderate reduction in weight (a mean loss of 2.5 kg) and a decrease in hepatic transaminase levels. The effects were sustained at 12 months.
“While the majority of patients showed a durable glycemic response over 12 months, a minority exhibited less benefit from DMR and required additional glucose-lowering medication at 24 weeks,” the researchers wrote. “Of note, approximately two-thirds of the patients who required addition of antidiabetic medication in the latter phase of study had undergone insulin secretagogue medication withdrawal at screening. For future study, it may not be necessary to discontinue these medications before DMR, and this will allow an even more precise measure of DMR effect.”
Dr. van Baar and colleagues acknowledged certain limitations of the phase 2 study, including its open-label, uncontrolled design. “The results of this multicenter study need to be confirmed in a proper controlled study. Nevertheless, this study forms the requisite solid foundation for further research, and controlled studies are currently underway.”
The study was funded by Fractyl Laboratories. Dr. van Baar reported having no financial disclosures. Four of the study authors reported having financial relationships with numerous pharmaceutical and device companies.
SOURCE: van Baar ACG et al. Gut. 2019 Jul 22. doi: 10.1136/gutjnl-2019-318349.
Among patients with suboptimally controlled type 2 diabetes who use oral glucose-lowering medication, endoscopic duodenal mucosal resection (DMR) can be implemented safely and effectively, results from a multicenter, international, phase 2 study demonstrated.
“DMR elicited a substantial improvement in parameters of glycemia as well as a decrease in liver transaminase levels at 24 weeks, which was sustained at 12 months post procedure,” researchers led by Annieke C.G. van Baar, MD, wrote in a study published online in Gut. “These findings were also associated with an improvement in patients’ diabetes treatment satisfaction.”
For the study, Dr. van Baar, of the department of gastroenterology and hepatology at Amsterdam University Medical Center, and colleagues at seven clinical sites enrolled 46 patients with type 2 diabetes who were on stable glucose-lowering medication to undergo DMR. The procedure “involves circumferential hydrothermal ablation of the duodenal mucosa resulting in subsequent regeneration of the mucosa,” they wrote. “Before ablation, the mucosa is lifted with saline to protect the outer layers of the duodenum.” DMR was performed under either general anesthesia or deep sedation with propofol by a single endoscopist at each site with extensive experience in therapeutic upper GI endoscopy and guidewire management.
The mean age of the study participants was 55 years and 63% were male. Of the 46 patients, 37 (80%) underwent complete DMR and results were reported for 36 of them. A total of 24 patients had at least one adverse event related to DMR (52%), mostly GI symptoms such as diarrhea, abdominal pain, nausea, and oropharyngeal pain. Of these, 81% were mild. One serious adverse event was considered to be related to the procedure. “This concerned a patient with general malaise, mild fever, and increased C-reactive protein level on the first day after DMR,” the researchers wrote. “The mild fever resolved within 24 hours and [C-reactive protein] level normalized within 3 days.” No unanticipated adverse events were reported.
During follow-up measures taken 24 weeks after their DMR, hemoglobin A1c fell by a mean of 10 mmol/mol (P less than .001), fasting plasma glucose by 1.7 mmol/L (P less than .001), and the Homeostatic Model Assessment of Insulin Resistance improved significantly (P less than .001). In addition, the procedure conferred a moderate reduction in weight (a mean loss of 2.5 kg) and a decrease in hepatic transaminase levels. The effects were sustained at 12 months.
“While the majority of patients showed a durable glycemic response over 12 months, a minority exhibited less benefit from DMR and required additional glucose-lowering medication at 24 weeks,” the researchers wrote. “Of note, approximately two-thirds of the patients who required addition of antidiabetic medication in the latter phase of study had undergone insulin secretagogue medication withdrawal at screening. For future study, it may not be necessary to discontinue these medications before DMR, and this will allow an even more precise measure of DMR effect.”
Dr. van Baar and colleagues acknowledged certain limitations of the phase 2 study, including its open-label, uncontrolled design. “The results of this multicenter study need to be confirmed in a proper controlled study. Nevertheless, this study forms the requisite solid foundation for further research, and controlled studies are currently underway.”
The study was funded by Fractyl Laboratories. Dr. van Baar reported having no financial disclosures. Four of the study authors reported having financial relationships with numerous pharmaceutical and device companies.
SOURCE: van Baar ACG et al. Gut. 2019 Jul 22. doi: 10.1136/gutjnl-2019-318349.
FROM GUT
Gastroparesis
To the Editor: We read with great pleasure the article by Sharayah et al about acute gastroparesis in a patient with diabetic ketoacidosis.1 However, in the case description, the authors reached a diagnosis of gastroparesis secondary to diabetic ketoacidosis without aptly ruling out some of its most common causes such as hypokalemia and other electrolyte imbalances seen in diabetic patients (in the setting of recurrent vomiting).
The authors also did not include the patient’s duration of diabetes or hemoglobin A1c level, both of which are linked with gastroparesis in diabetic patients.2 Pertinent biochemical information that can help readers formulate a rational approach and journey to making a diagnosis appears elusive in their article.
- Sharayah AM, Hajjaj N, Osman R, Livornese D. Gastroparesis in a patient with diabetic ketoacidosis. Cleve Clin J Med 2019; 86(4):238–239. doi:10.3949/ccjm.86a.18116
- Bharucha AE, Kudva Y, Basu A, et al. Relationship between glycemic control and gastric emptying in poorly controlled type 2 diabetes. Clin Gastroenterol Hepatol 2015; 13(3):466–476.e461. doi:10.1016/j.cgh.2014.06.034
To the Editor: We read with great pleasure the article by Sharayah et al about acute gastroparesis in a patient with diabetic ketoacidosis.1 However, in the case description, the authors reached a diagnosis of gastroparesis secondary to diabetic ketoacidosis without aptly ruling out some of its most common causes such as hypokalemia and other electrolyte imbalances seen in diabetic patients (in the setting of recurrent vomiting).
The authors also did not include the patient’s duration of diabetes or hemoglobin A1c level, both of which are linked with gastroparesis in diabetic patients.2 Pertinent biochemical information that can help readers formulate a rational approach and journey to making a diagnosis appears elusive in their article.
To the Editor: We read with great pleasure the article by Sharayah et al about acute gastroparesis in a patient with diabetic ketoacidosis.1 However, in the case description, the authors reached a diagnosis of gastroparesis secondary to diabetic ketoacidosis without aptly ruling out some of its most common causes such as hypokalemia and other electrolyte imbalances seen in diabetic patients (in the setting of recurrent vomiting).
The authors also did not include the patient’s duration of diabetes or hemoglobin A1c level, both of which are linked with gastroparesis in diabetic patients.2 Pertinent biochemical information that can help readers formulate a rational approach and journey to making a diagnosis appears elusive in their article.
- Sharayah AM, Hajjaj N, Osman R, Livornese D. Gastroparesis in a patient with diabetic ketoacidosis. Cleve Clin J Med 2019; 86(4):238–239. doi:10.3949/ccjm.86a.18116
- Bharucha AE, Kudva Y, Basu A, et al. Relationship between glycemic control and gastric emptying in poorly controlled type 2 diabetes. Clin Gastroenterol Hepatol 2015; 13(3):466–476.e461. doi:10.1016/j.cgh.2014.06.034
- Sharayah AM, Hajjaj N, Osman R, Livornese D. Gastroparesis in a patient with diabetic ketoacidosis. Cleve Clin J Med 2019; 86(4):238–239. doi:10.3949/ccjm.86a.18116
- Bharucha AE, Kudva Y, Basu A, et al. Relationship between glycemic control and gastric emptying in poorly controlled type 2 diabetes. Clin Gastroenterol Hepatol 2015; 13(3):466–476.e461. doi:10.1016/j.cgh.2014.06.034
In reply: Gastroparesis
In Reply: We thank the readers for their letter. Our patient’s laboratory values at the time of presentation were as follows:
- Corrected sodium 142 mmol/L
- Potassium 5.5 mmol/L
- Phosphorus 6.6 mmol/L.
The rest of the electrolyte levels were within normal limits.
These reported electrolyte levels were unlikely to cause such gastroparesis. The patient’s hemoglobin A1c was 8.7% at the time of presentation, with no previous values available. However, since abdominal computed tomography done 1 year before this presentation did not show stomach dilation and the patient was asymptomatic, his gastroparesis was presumed to be acute.
In Reply: We thank the readers for their letter. Our patient’s laboratory values at the time of presentation were as follows:
- Corrected sodium 142 mmol/L
- Potassium 5.5 mmol/L
- Phosphorus 6.6 mmol/L.
The rest of the electrolyte levels were within normal limits.
These reported electrolyte levels were unlikely to cause such gastroparesis. The patient’s hemoglobin A1c was 8.7% at the time of presentation, with no previous values available. However, since abdominal computed tomography done 1 year before this presentation did not show stomach dilation and the patient was asymptomatic, his gastroparesis was presumed to be acute.
In Reply: We thank the readers for their letter. Our patient’s laboratory values at the time of presentation were as follows:
- Corrected sodium 142 mmol/L
- Potassium 5.5 mmol/L
- Phosphorus 6.6 mmol/L.
The rest of the electrolyte levels were within normal limits.
These reported electrolyte levels were unlikely to cause such gastroparesis. The patient’s hemoglobin A1c was 8.7% at the time of presentation, with no previous values available. However, since abdominal computed tomography done 1 year before this presentation did not show stomach dilation and the patient was asymptomatic, his gastroparesis was presumed to be acute.
What we know—and don’t—about non-nutritive sweeteners
An estimated 93.3 million Americans (roughly 40% of the US population) were obese in 2015-2016, and most of them had at least 1 chronic disease.1 As a result, patient education focused on lifestyle modification, including healthy nutrition and physical activity, has become an integral part of our everyday practice.
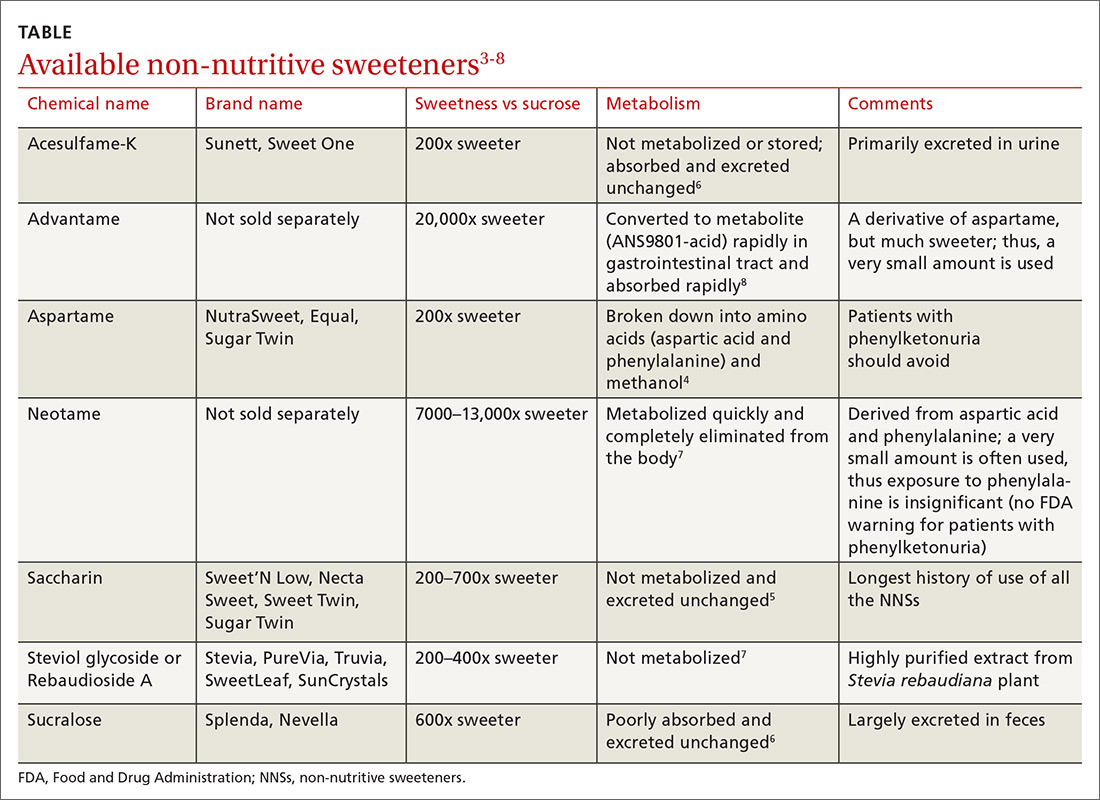
At the same time, the most recent dietary guidelines recommend that added sugar make up < 10% of daily calories.2 In the United States, low-calorie food and beverages containing non-nutritive sweeteners (NNSs; TABLE3-8) have become a popular means of keeping the sweetness in our diet without the health ramifications associated with sugar. These NNSs (aka, artificial sweeteners, high-intensity sweeteners, and non-caloric sweeteners) are ubiquitous in soft drinks, processed grains (including breads, cereals, and granola bars), and dairy products (including yogurts, flavored milk, and ice cream). As examples, NNSs are present in 42% of flavored waters, 33% of yogurts, and all diet beverages.9,10 They can even be found in medications, multivitamins, toothpaste, and mouthwash.
Business is booming
Global NNS consumption has been growing more than 5% per year, meaning that by 2020, NNSs are expected to be a $2.2 billion industry.11 One study using data from the National Health and Nutrition Examination Survey (NHANES) found that the use of NNSs in the United States increased from 21.1% in 2003 to 24.9% in 2009-2010 among adults and increased from 7.8% to 18.9% over the same time period among children.12
The main increase in the consumption of NNSs across all age groups has been via the consumption of beverages. Approximately 11% of healthy weight, 19% of overweight, and 22% of obese adults consume diet beverages.13,14 Consumption of diet beverages or NNSs increases with age12 and is especially common among women with higher levels of education and income.15
However, concerns remain about the safety of these agents and their effect on weight, appetite, and the body’s glycemic response. This article reviews the available research and current recommendations regarding the use of NNSs.
WHAT EFFECT DO NNS s HAVE ON WEIGHT?
The data on NNSs and weight are inconsistent. One randomized controlled trial(RCT) compared weight loss over the course of 1 year (12-week weight loss phase; 9-month weight maintenance phase) when 303 participants consumed either water or drinks sweetened with NNSs.16 Weight loss was significantly greater in the NNS drink group when compared with the water group.16
Observational studies have revealed similar findings.17,18 Data from NHANES revealed that US adults (n = 14,098) during 2 nonconsecutive 24-hour dietary recall periods demonstrated lower total energy (calorie) intake if they consumed NNSs vs no NNSs.19 Another study using 2011-2016 NHANES data on adolescents (n = 7026) found no difference in energy intake between those who consumed beverages containing NNSs vs those who consumed beverages containing sugar.20
Continue to: Other lines of investigation...
Other lines of investigation, including animal studies, have shown that long-term use of NNSs is associated with numerous metabolic derangements including weight gain.21 The negative effects of NNSs appear to be the greatest in males and those who are obese and have high-calorie diets.21
A 2017 meta-analysis concluded that evidence from RCTs does not support a benefit of NNSs on weight management, and that routine consumption of NNSs may be associated with increased body mass index (BMI) and cardiometabolic risk.22 Another systematic review and meta-analysis found that there was a higher pooled risk for obesity among those who drank beverages containing NNSs vs those who drank sugar-containing beverages.23
Based on the most current literature, we conclude that NNSs are not beneficial for weight loss. While there is concern about weight gain through psychological effects (stimulation of sweetness receptors without satiety), further well-designed research is needed to explore whether this concern has merit.
WHAT IS THE EFFECT OF NNSs ON APPETITE?
There appears to be no effect. While original studies seemed to indicate there was an effect, later studies leaned to the contrary.
The notion that NNSs might enhance appetite and food intake was advanced in the 1980s by John Blundell and his research team.24 The hypothesis was that since NNSs uncouple sweet taste and calories, they do not exert the normal post-ingestive inhibitory influence that real sugar does. This, in turn, disrupts appetite control mechanisms.25-27
Continue to: However, subsequent research studies...
However, subsequent research studies found no relationship between the use of NNSs and appetite.28-30 Mattes and colleagues hypothesized that such a difference in findings could result from the fact that earlier studies focused on isolating NNSs from other energy-yielding products, which emphasized an association with heightened hunger.29 Subsequent studies showed that when NNSs were incorporated into energy-yielding products, there was no association between NNSs and increased hunger or appetite.
DO NNSs INCREASE THE RISK FOR TYPE 2 DIABETES MELLITUS?
The data are mixed. One study of women participating in the Nurses’ Health Study II showed that those who consumed caffeinated, artificially-sweetened beverages had a 35% higher risk of developing type 2 diabetes mellitus (T2DM); however, this risk was no longer significant after adjusting for BMI and energy intake.31
The Health Professionals Follow-Up Trial studied more than 40,000 men for more than 20 years and found that NNS consumption increased the risk of developing T2DM by 40%.32 However, this finding lost statistical significance after adjusting for BMI.32
These results make it difficult to determine whether there is any association between NNSs and T2DM; rather NNS-containing beverages are likely consumed more often by those who have higher BMIs and by those trying to lose weight.
A 2017 randomized crossover study involving 10 healthy men looked at the effects of a variety of caloric and non-caloric sweeteners on 24-hour glucose profiles and found no differences.33 Another study, a randomized, double-blind, crossover trial involving 60 non-obese adults without diabetes who did not consume NNSs, randomized the participants one-to-one to drink either 2 cans per day of either a beverage containing aspartame and acesulfame K or an unsweetened, no-calorie beverage for 12 weeks.34
Continue to: After a 4-week washout period...
After a 4-week washout period, the participants then switched to the opposite beverage for 12 weeks. The study concluded that consumption of 2 cans of a beverage containing aspartame and acesulfame K over 12 weeks had no significant effect on insulin sensitivity or secretion in nondiabetic adults.34
Similar results were obtained from a study involving 100 non-obese adults.35 The researchers found that aspartame ingested at 2 different doses (350 or 1050 mg/d) in beverages over 12 weeks had no effect on a 240-minute oral glucose tolerance test, blood pressure, appetite, or body weight.35
A 2016 systematic review critically evaluated the effect of NNSs on both glucose absorption and appetite.36 The review included 14 observational prospective trials, 28 RCTs, and 2 meta-analyses. The sweeteners studied included aspartame, sucralose, saccharin, acesulfame K, and stevia.36 The studies were focused largely on single-exposure outcomes (20 trials), but a minority of the studies (8 trials) looked at longer exposures from 1 to 18 weeks. Only some of the studies controlled for critical variables, such as BMI. In the end, there was no consistent pattern of increased or decreased risk for insulin resistance or diabetes.36
Two meta-analyses tried to determine if an association exists between consumption of beverages containing NNSs and the development of T2DM.37,38 The first meta-analysis with 4 studies showed a slight, but significant, relative risk (RR) of 1.13 (95% confidence interval [CI], 1.02-1.25) for those who consumed beverages containing NNSs.37 In the second meta-analysis (10 studies), NNS consumption had an RR of 1.48 (95% CI, 1.35-1.62), but the risk was lower (and no longer significant) after adjusting for BMI.38 A study of 98 Hispanic adolescents who were overweight or obese found that chronic users (n = 9) of NNSs had higher HbA1c levels 1 year later than did controls (n = 75) and people who initiated use of NNSs between the baseline and 1-year visit (n = 14).39
The American Diabetes Association (ADA) and American Heart Association joint position statement on NNSs, first published in 2012, says that NNSs can be utilized to reduce caloric and carbohydrate consumption for overall diabetes control and to obtain a healthy body weight.40 These principles were reaffirmed in the ADA Standards of Care in 2019.41
Continue to: The 2015 US Scientific Reports on Dietary Guidelines...
The 2015 US Scientific Reports on Dietary Guidelines provided a consensus statement saying, “Future experimental studies should examine the relationship between artificially sweetened soft drinks and biomarkers of insulin resistance and other diabetes markers.”42
DO NNSs HAVE ANY ADVERSE HEALTH EFFECTS?
Maybe. Many individuals avoid NNSs due to fear of developing cancer. While rat studies have previously shown a dose-dependent increased risk of developing cancer, epidemiologic studies in humans have not confirmed an association.43 The National Cancer Institute reports that carcinogenicity studies of NNSs have not shown an association with cancer in humans.44
A prospective study—the Nurses’ Health Study, which followed over 88,000 women for 24 years—found that consumption of > 2 diet sodas per day was associated with an increased risk for coronary heart disease (CHD) and chronic kidney disease (CKD) compared with consumption of < 1 diet soda per month.45 However, other prospective studies have shown that these specific negative health effects may not be present when controlling for weight.45,46
While the prospective studies found some associations between medical conditions (eg, CHD and CKD) and NNS consumption, the literature is limited to intake from beverages and does not include NNS-containing foods. More studies are needed to determine the relationship between NNSs and potential adverse health events, since the current literature is observational and cannot predict causation.
A 2019 study explored the associations between long-term consumption of sugar-sweetened beverages and artificially sweetened beverages (ASBs) and the risk of mortality in the United States.47 This study included 37,716 men from the Health Professionals Follow-up Study and 80,647 women from the Nurses’ Health Study. Subjects who had the highest consumption of ASBs had higher risks for total and cardiovascular disease mortality.47 Cohort-specific analyses showed that an association between ASB consumption and mortality was observed in the participants from the Nurses’ Health Study but not in those from the Health Professionals Follow-up Study, warranting further investigation.47 Cancer mortality and ASB consumption were not shown to have an association in this study.
Continue to: WHY ARE THE DATA INCONCLUSIVE?
WHY ARE THE DATA INCONCLUSIVE?
Nutritional studies are hard to complete accurately outside of the laboratory setting. Also, the science of NNSs is new and evolving.
With regard to obesity and NNSs, it is possible that findings have been due to reverse causation. People who are overweight or obese are more likely to consume low-calorie foods and beverages; they are also at greater risk for developing diseases, such as T2DM.48,49
HOW SAFE ARE NNSs?
They appear to be safe, but more data are needed. Each of the 7 FDA-approved NNSs has passed extensive laboratory, animal, and human testing, and appears to cause no harm in the human body when consumed.49 But clearly the data are incomplete. As we continue to gain a greater understanding of the metabolism of NNSs, we may need to revisit the issue of safety.
ARE THERE ANY NNSs THAT SOME PEOPLE SHOULD AVOID?
Yes. People with phenylketonuria, who have difficulty metabolizing phenylalanine (a component of aspartame), should avoid consumption of aspartame.50
In addition, NNSs have been found to be present in breast milk.51 While the significance of this finding is yet to be determined, we warn against the use of NNSs by women who are breastfeeding.51
WHAT EFFECT—IF ANY—DO NNSs HAVE ON GUT MICROBIOTA?
We don’t know. Disruptions in the gut microbiome have been linked to numerous metabolic abnormalities, including obesity, insulin resistance, and diabetes, as well as cardiovascular disorders.52,53 Diet is a main determinant of balance in the gut microbiota.54 The gut microbiota are centrally involved in energy harvest, and studies have suggested that low gut bacterial diversity is associated with increased adiposity, insulin resistance, and low-grade inflammation.55-60 Whether NNSs have a relationship with abnormal changes in gut microbiota requires further study.
CORRESPONDENCE
Clipper F. Young, PharmD, MPH, CDE, BC-ADM, BCGP, Touro University California, College of Osteopathic Medicine, 1310 Club Drive, Vallejo, CA 94592; [email protected].
1. Adult obesity facts. Centers for Disease Control and Prevention Web site. https://www.cdc.gov/obesity/data/adult.html. Reviewed August 13, 2018. Accessed July 15, 2019.
2. Dietary guidelines for Americans 2015-2020: answers to your questions. USDA ChooseMyPlate.gov Web site. https://www.choosemyplate.gov/2015-2020-dietary-guidelines-answers-your-questions. Accessed July 15, 2019.
3. Additional information about high-intensity sweeteners permitted for use in food in the United States. US Food and Drug Administration Web site. https://www.fda.gov/food/food-additives-petitions/additional-information-about-high-intensity-sweeteners-permitted-use-food-united-states. Published February 8, 2018. Accessed July 15, 2019.
4. Magnuson B, for the Aspartame Expert Work Group. Nutritive and non-nutritive sweeteners. NNNS: aspartame, methanol and formaldehyde relationships (2011). https://www.foodsweeteners.com/wp-content/uploads/2015/08/Aspartame-Methanol-and-Formaldehyde-Relationships.pdf. Accessed July 15, 2019.
5. Jo JH, Kim S, Jeon TW, et al. Investigation of the regulatory effects of saccharin on cytochrome P450s in male ICR mice. Toxicol Res. 2017;33:25-30.
6. Shwide-Slavin C, Swift C, Ross T. Nonnutritive sweetener: where are we today? Diabetes Spectrum. 2012;25:104-110.
7. Chattopadhyay S, Raychaudhuri U, Chakraborty R. Artificial sweeteners – a review. J Food Sci Technol. 2014;51:611-621.
8. EFSA Panel on Food Additives and Nutrient Sources added to Food. Scientific opinion on the safety of advantame for the proposed uses as a food additive. EFSA Journal. 2013;11:3301.
9. Fitch C, Keim KS; Academy of Nutrition and Dietetics. Position of the Academy of Nutrition and Dietetics: use of nutritive and nonnutritive sweeteners. J Acad Nutr Diet. 2012;112:739-758.
10. Ng SW, Slining MM, Popkin BM. Use of caloric and non-caloric sweeteners in US consumer packaged foods, 2005-2009. J Acad Nutr Diet. 2012;112:1828-1834.
11. Sylvetsky AC, Rother KI. Trends in the consumption of low-calorie sweeteners. Physiol Behav. 2016;164(Pt B):446-450.
12. Piernas C, Ng SW, Popkin B. Trends in purchases and intake of foods and beverages containing caloric and low-calorie sweeteners over the last decade in the United States. Pediatr Obes. 2013;8:294-306.
13. Sylvetsky AC, Welsh JA, Brown RJ, et al. Low-calorie sweetener consumption is increasing in the United States. Am J Clin Nutr. 2012;96:640-646.
14. Bleich SN, Wolfson JA, Vine S, et al. Diet-beverage consumption and caloric intake among US adults, overall and by body weight. Am J Public Health. 2014;104:e72-e78.
15. Drewnowski A, Rehm CD. Socio-demographic correlates and trends in low-calorie sweetener use among adults in the United States from 1999 to 2008. Eur J Clin Nutr. 2015;69:1035-1041.
16. Peters JC, Wyatt HR, Foster GD, et al. The effects of water and non-nutritive sweetened beverages on weight loss during a 12-week weight loss treatment program. Obesity 2014;22:1415-1421.
17. Peters JC, Wyatt HR, Foster GD, et al. The effects of water and non-nutritive sweetened beverages on weight loss during a 12-week weight loss treatment program. Obesity (Silver Spring). 2014;22:1415-1421.
18. Bellisle F, Drewnowski A. Intense sweeteners, energy intake and the control of body weight. Eur J Clin Nutr. 2007;61:691-700.
19. Malek AM, Hunt KJ, DellaValle DM, et al. Reported consumption of low-calorie sweetener in foods, beverages, and food and beverage additions by US adults: NHANES 2007-2012. Curr Dev Nutr. 2018;2:nzy054.
20. Sylvetsky AC, Figueroa J, Zimmerman T, et al. Consumption of low-calorie sweetened beverages is associated with higher total energy and sugar intake among children, NHANES 2011-2016. Pediatr Obes. 2019;2:e12535.
21. Fowler SPG. Low-calorie sweetener use and energy balance: results from experimental studies in animals, and large-scale prospective studies in humans. Physiol Behav. 2016;164(Pt B):517-523.
22. Azad MB, Abou-Setta AM, Chauhan BF, et al. Nonnutritive sweeteners and cardiometabolic health: a systematic review and meta-analysis of randomized controlled trials and prospective cohort studies. CMAJ. 2017;189: E929-E939.
23. Ruanpeng D, Thongprayoon C, Cheungpasitporn W, et al. Sugar and artificially-sweetened beverages linked to obesity: a systematic review and meta-analysis. QJM. 2017;110:513-520.
24. Blundell JE, Rogers PJ, Hill AJ. Uncoupling sweetness and calories: methodological aspects of laboratory studies on appetite control. Appetite. 1988;11(Suppl 1):54-61.
25. Bellisle F. Intense sweeteners, appetite for the sweet taste, and relationship to weight management. Curr Obes Rep. 2015;4:106-110.
26. Bryant CE, Wasse LK, Astbury N, et al. Non-nutritive sweeteners: no class effect on the glycaemic or appetite responses to ingested glucose. Eur J Clin Nutr. 2014;68:629-631.
27. Canty DJ, Chan MM. Effects of consumption of caloric vs noncaloric sweet drinks on indices of hunger and food consumption in normal adults. Am J Clin Nutr. 1991;53:1159-1164.
28. Meyer-Gerspach AC, Wolnerhanssen B, Beglinger C. Functional roles of low calorie sweeteners on gut function. Physiol Behav. 2016;164(Pt B):479-481.
29. Mattes RD, Popkin BM. Nonnutritive sweetener consumption in humans: effects on appetite and food intake and their putative mechanisms. Am J Clin Nutr. 2009;89:1-14.
30. Bhupathiraju SN, Pan A, Malik VS, et al. Caffeinated and caffeine-free beverages and risk of type 2 diabetes. Am J Clin Nutr. 2013;97:155-166.
31. Schulze MB, Manson JE, Ludwig DS, et al. Sugar-sweetened beverages, weight gain, and incidence of type 2 diabetes in young and middle-aged women. JAMA. 2004;292:927-934.
32. de Koning L, Malik VS, Rimm EB, et al. Sugar-sweetened and artificially sweetened beverage consumption and the risk of type 2 diabetes in men. Am J Clin Nutr. 2011;93:1321-1327.
33. Tey SL, Salleh NB, Henry CJ, et al. Effect of non-nutritive (artificial vs natural) sweeteners on 24-hour glucose profile. Eur J Clin Nutr. 2017;71:1129-1132.
34. Bonnet F, Tavenard A, Esvan M, et al. Consumption of a carbonated beverage with high-intensity sweeteners has no effect on insulin sensitivity and secretion in nondiabetic adults. J Nutr. 2018;148:1293-1299.
35. Higgins KA, Considine RV, Mattes RD. Aspartame consumption for 12 weeks does not affect glycemia, appetite, or body weight of healthy, lean adults in a randomized controlled trial. J Nutr. 2018;148:650-657.
36. Romo-Romo A, Aguilar-Salinas CA, Brito-Cordova GX, et al. Effects of the non-nutritive sweeteners on glucose metabolism and appetite regulating hormones: systematic review of observational prospective studies and clinical trials. PloS One. 2016;11:e0161264.
37. Greenwood DC, Threspleton DE, Evans CE, et al. Association between sugar-sweetened and artificially sweetened soft drinks and type 2 diabetes: systematic review and dose-response meta-analysis of prospective studies. Br J Nutr. 2014;112:725-734.
38. Imamura F, O’Conner L, Ye M, et al. Consumption of sugar sweetened beverages, artificially sweetened beverages, and fruit juice and incidence of type 2 diabetes: systematic review, meta-analysis, and estimation of population attributable fraction. BMJ. 2015;351:h3576.
39. Davis JN, Asigbee FM, Markowitz AK, et al. Consumption of artificial sweetened beverages associated with adiposity and increasing HbA1c in Hispanic youth. Clin Obes. 2018;8:236-243.
40. Gardner C, Wylie-Rosett J, Gidding SS, et al. Nonnutritive sweeteners: current use and health perspectives. a scientific statement from the American Heart Association and the American Diabetes Association. Diabetes Care. 2012;35:1798-1808.
41. American Diabetes Association. Standards of Medical Care in Diabetes—2019. Diabetes Care. 2019;42(Suppl 1):S1-S183.
42. Dietary Guidelines Advisory Committee. Scientific Report of the 2015 Dietary Guidelines Advisory Committee: advisory report to the Secretary of Health and Human Services and the Secretary of Agriculture. Office of Disease Prevention and Health Promotion Web site. https://health.gov/dietaryguidelines/2015-scientific-report/.Published February 2015. Accessed July 15, 2019.
43. Aune D. Soft drinks, aspartame, and the risk of cancer and cardiovascular disease. Am J Clin Nutr. 2012;96:1249-1251.
44. Artificial sweeteners and cancer. National Cancer Institute Web site. https://www.cancer.gov/about-cancer/causes-prevention/risk/diet/artificial-sweeteners-fact-sheet. Reviewed August 10, 2016. Accessed July 15, 2019.
45. Fung TT, Malik V, Rexrode KM, et al. Sweetened beverage consumption and risk of coronary heart disease in women. Am J Clin Nutr. 2009;89:1037-1042.
46. Lin J, Curhan GC. Associations of sugar and artificially sweetened soda with albuminuria and kidney function decline in women. Clin J Am Soc Nephrol. 2011;6:160-166.
47. Malik VS, Li Y, Pan A, et al. Long-term consumption of sugar-sweetened and artificially sweetened beverages and risk of mortality in US adults. Circulation. 2019;139:2113-2125.
48. Gardener H, Rundek T, Markert M, et al. Diet soft drink consumption is associated with an increased risk of vascular events in the Northern Manhattan Study. J Gen Inten Med. 2012;27:1120-1126.
49. Fitch C, Keim KS. Position of the Academy of Nutrition and Dietetics: use of nutritive and nonnutritive sweeteners. J Acad Nutr Diet. 2012;112:739-758.
50. US Food and Drug Administration. Additional information about high-intensity sweeteners permitted for use in food in the United States. https://www.fda.gov/food/ingredientspackaginglabeling/foodadditivesingredients/ucm397725.htm#Aspartame. Accessed May 26, 2019.
51. Sylvetsky AC, Gardner AL, Bauman V, et al. Nonnutritive sweeteners in breast milk. J Toxicol Environ Health. 2015;78:1029-1032.
52. Rajani C, Jia W. Disruptions in gut microbial-host co-metabolism and the development of metabolic disorders. Clin Sci (Lond). 2018;132:791-811.
53. Kho ZY, Lal SK. The human gut microbiome—a potential controller of wellness and disease. Front Microbiol. 2018;9:1835.
54. Nettleton JE, Reimer RA, Shearer J. Reshaping the gut microbiota: impact of low calorie sweeteners and the link to insulin resistance. Physiol Behav. 2016;164(Pt B):488-493.
55. Turnbaugh PJ, Hamady M, Yatsunenko T, et al. A core gut microbiome in obese and lean twins. Nature. 2009;457:480-484.
56. Cotillard A, Kennedy SP, Kong LC, et al. Dietary intervention impact on gut microbial gene richness. Nature. 2013;500:585-588.
57. Le Chatelier E, Nielsen T, Qin J, et al. Richness of human gut microbiome correlates with metabolic markers. Nature. 2013;500:541-546.
58. Abou-Donia MB, El-Masry EM, Abdel-Rahman AA, et al. Splenda alters gut microflora and increases intestinal p-glycoprotein and cytochrome P-450 in male rats. J Toxicol Environ Health A. 2008;71:1415-1429.
59. Anderson RL. Effect of saccharin ingestion on stool composition in relation to caecal enlargement and increased stool hydration. Food Chem Toxicol. 1983;21:255-257.
60. Suez J, Korem T, Zeevi D, et al. Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature. 2014;514:181-186.
An estimated 93.3 million Americans (roughly 40% of the US population) were obese in 2015-2016, and most of them had at least 1 chronic disease.1 As a result, patient education focused on lifestyle modification, including healthy nutrition and physical activity, has become an integral part of our everyday practice.
At the same time, the most recent dietary guidelines recommend that added sugar make up < 10% of daily calories.2 In the United States, low-calorie food and beverages containing non-nutritive sweeteners (NNSs; TABLE3-8) have become a popular means of keeping the sweetness in our diet without the health ramifications associated with sugar. These NNSs (aka, artificial sweeteners, high-intensity sweeteners, and non-caloric sweeteners) are ubiquitous in soft drinks, processed grains (including breads, cereals, and granola bars), and dairy products (including yogurts, flavored milk, and ice cream). As examples, NNSs are present in 42% of flavored waters, 33% of yogurts, and all diet beverages.9,10 They can even be found in medications, multivitamins, toothpaste, and mouthwash.

Business is booming
Global NNS consumption has been growing more than 5% per year, meaning that by 2020, NNSs are expected to be a $2.2 billion industry.11 One study using data from the National Health and Nutrition Examination Survey (NHANES) found that the use of NNSs in the United States increased from 21.1% in 2003 to 24.9% in 2009-2010 among adults and increased from 7.8% to 18.9% over the same time period among children.12
The main increase in the consumption of NNSs across all age groups has been via the consumption of beverages. Approximately 11% of healthy weight, 19% of overweight, and 22% of obese adults consume diet beverages.13,14 Consumption of diet beverages or NNSs increases with age12 and is especially common among women with higher levels of education and income.15
However, concerns remain about the safety of these agents and their effect on weight, appetite, and the body’s glycemic response. This article reviews the available research and current recommendations regarding the use of NNSs.
WHAT EFFECT DO NNS s HAVE ON WEIGHT?
The data on NNSs and weight are inconsistent. One randomized controlled trial(RCT) compared weight loss over the course of 1 year (12-week weight loss phase; 9-month weight maintenance phase) when 303 participants consumed either water or drinks sweetened with NNSs.16 Weight loss was significantly greater in the NNS drink group when compared with the water group.16
Observational studies have revealed similar findings.17,18 Data from NHANES revealed that US adults (n = 14,098) during 2 nonconsecutive 24-hour dietary recall periods demonstrated lower total energy (calorie) intake if they consumed NNSs vs no NNSs.19 Another study using 2011-2016 NHANES data on adolescents (n = 7026) found no difference in energy intake between those who consumed beverages containing NNSs vs those who consumed beverages containing sugar.20
Continue to: Other lines of investigation...
Other lines of investigation, including animal studies, have shown that long-term use of NNSs is associated with numerous metabolic derangements including weight gain.21 The negative effects of NNSs appear to be the greatest in males and those who are obese and have high-calorie diets.21
A 2017 meta-analysis concluded that evidence from RCTs does not support a benefit of NNSs on weight management, and that routine consumption of NNSs may be associated with increased body mass index (BMI) and cardiometabolic risk.22 Another systematic review and meta-analysis found that there was a higher pooled risk for obesity among those who drank beverages containing NNSs vs those who drank sugar-containing beverages.23
Based on the most current literature, we conclude that NNSs are not beneficial for weight loss. While there is concern about weight gain through psychological effects (stimulation of sweetness receptors without satiety), further well-designed research is needed to explore whether this concern has merit.
WHAT IS THE EFFECT OF NNSs ON APPETITE?
There appears to be no effect. While original studies seemed to indicate there was an effect, later studies leaned to the contrary.
The notion that NNSs might enhance appetite and food intake was advanced in the 1980s by John Blundell and his research team.24 The hypothesis was that since NNSs uncouple sweet taste and calories, they do not exert the normal post-ingestive inhibitory influence that real sugar does. This, in turn, disrupts appetite control mechanisms.25-27
Continue to: However, subsequent research studies...
However, subsequent research studies found no relationship between the use of NNSs and appetite.28-30 Mattes and colleagues hypothesized that such a difference in findings could result from the fact that earlier studies focused on isolating NNSs from other energy-yielding products, which emphasized an association with heightened hunger.29 Subsequent studies showed that when NNSs were incorporated into energy-yielding products, there was no association between NNSs and increased hunger or appetite.
DO NNSs INCREASE THE RISK FOR TYPE 2 DIABETES MELLITUS?
The data are mixed. One study of women participating in the Nurses’ Health Study II showed that those who consumed caffeinated, artificially-sweetened beverages had a 35% higher risk of developing type 2 diabetes mellitus (T2DM); however, this risk was no longer significant after adjusting for BMI and energy intake.31
The Health Professionals Follow-Up Trial studied more than 40,000 men for more than 20 years and found that NNS consumption increased the risk of developing T2DM by 40%.32 However, this finding lost statistical significance after adjusting for BMI.32
These results make it difficult to determine whether there is any association between NNSs and T2DM; rather NNS-containing beverages are likely consumed more often by those who have higher BMIs and by those trying to lose weight.
A 2017 randomized crossover study involving 10 healthy men looked at the effects of a variety of caloric and non-caloric sweeteners on 24-hour glucose profiles and found no differences.33 Another study, a randomized, double-blind, crossover trial involving 60 non-obese adults without diabetes who did not consume NNSs, randomized the participants one-to-one to drink either 2 cans per day of either a beverage containing aspartame and acesulfame K or an unsweetened, no-calorie beverage for 12 weeks.34
Continue to: After a 4-week washout period...
After a 4-week washout period, the participants then switched to the opposite beverage for 12 weeks. The study concluded that consumption of 2 cans of a beverage containing aspartame and acesulfame K over 12 weeks had no significant effect on insulin sensitivity or secretion in nondiabetic adults.34
Similar results were obtained from a study involving 100 non-obese adults.35 The researchers found that aspartame ingested at 2 different doses (350 or 1050 mg/d) in beverages over 12 weeks had no effect on a 240-minute oral glucose tolerance test, blood pressure, appetite, or body weight.35
A 2016 systematic review critically evaluated the effect of NNSs on both glucose absorption and appetite.36 The review included 14 observational prospective trials, 28 RCTs, and 2 meta-analyses. The sweeteners studied included aspartame, sucralose, saccharin, acesulfame K, and stevia.36 The studies were focused largely on single-exposure outcomes (20 trials), but a minority of the studies (8 trials) looked at longer exposures from 1 to 18 weeks. Only some of the studies controlled for critical variables, such as BMI. In the end, there was no consistent pattern of increased or decreased risk for insulin resistance or diabetes.36
Two meta-analyses tried to determine if an association exists between consumption of beverages containing NNSs and the development of T2DM.37,38 The first meta-analysis with 4 studies showed a slight, but significant, relative risk (RR) of 1.13 (95% confidence interval [CI], 1.02-1.25) for those who consumed beverages containing NNSs.37 In the second meta-analysis (10 studies), NNS consumption had an RR of 1.48 (95% CI, 1.35-1.62), but the risk was lower (and no longer significant) after adjusting for BMI.38 A study of 98 Hispanic adolescents who were overweight or obese found that chronic users (n = 9) of NNSs had higher HbA1c levels 1 year later than did controls (n = 75) and people who initiated use of NNSs between the baseline and 1-year visit (n = 14).39
The American Diabetes Association (ADA) and American Heart Association joint position statement on NNSs, first published in 2012, says that NNSs can be utilized to reduce caloric and carbohydrate consumption for overall diabetes control and to obtain a healthy body weight.40 These principles were reaffirmed in the ADA Standards of Care in 2019.41
Continue to: The 2015 US Scientific Reports on Dietary Guidelines...
The 2015 US Scientific Reports on Dietary Guidelines provided a consensus statement saying, “Future experimental studies should examine the relationship between artificially sweetened soft drinks and biomarkers of insulin resistance and other diabetes markers.”42
DO NNSs HAVE ANY ADVERSE HEALTH EFFECTS?
Maybe. Many individuals avoid NNSs due to fear of developing cancer. While rat studies have previously shown a dose-dependent increased risk of developing cancer, epidemiologic studies in humans have not confirmed an association.43 The National Cancer Institute reports that carcinogenicity studies of NNSs have not shown an association with cancer in humans.44
A prospective study—the Nurses’ Health Study, which followed over 88,000 women for 24 years—found that consumption of > 2 diet sodas per day was associated with an increased risk for coronary heart disease (CHD) and chronic kidney disease (CKD) compared with consumption of < 1 diet soda per month.45 However, other prospective studies have shown that these specific negative health effects may not be present when controlling for weight.45,46
While the prospective studies found some associations between medical conditions (eg, CHD and CKD) and NNS consumption, the literature is limited to intake from beverages and does not include NNS-containing foods. More studies are needed to determine the relationship between NNSs and potential adverse health events, since the current literature is observational and cannot predict causation.
A 2019 study explored the associations between long-term consumption of sugar-sweetened beverages and artificially sweetened beverages (ASBs) and the risk of mortality in the United States.47 This study included 37,716 men from the Health Professionals Follow-up Study and 80,647 women from the Nurses’ Health Study. Subjects who had the highest consumption of ASBs had higher risks for total and cardiovascular disease mortality.47 Cohort-specific analyses showed that an association between ASB consumption and mortality was observed in the participants from the Nurses’ Health Study but not in those from the Health Professionals Follow-up Study, warranting further investigation.47 Cancer mortality and ASB consumption were not shown to have an association in this study.
Continue to: WHY ARE THE DATA INCONCLUSIVE?
WHY ARE THE DATA INCONCLUSIVE?
Nutritional studies are hard to complete accurately outside of the laboratory setting. Also, the science of NNSs is new and evolving.
With regard to obesity and NNSs, it is possible that findings have been due to reverse causation. People who are overweight or obese are more likely to consume low-calorie foods and beverages; they are also at greater risk for developing diseases, such as T2DM.48,49
HOW SAFE ARE NNSs?
They appear to be safe, but more data are needed. Each of the 7 FDA-approved NNSs has passed extensive laboratory, animal, and human testing, and appears to cause no harm in the human body when consumed.49 But clearly the data are incomplete. As we continue to gain a greater understanding of the metabolism of NNSs, we may need to revisit the issue of safety.
ARE THERE ANY NNSs THAT SOME PEOPLE SHOULD AVOID?
Yes. People with phenylketonuria, who have difficulty metabolizing phenylalanine (a component of aspartame), should avoid consumption of aspartame.50
In addition, NNSs have been found to be present in breast milk.51 While the significance of this finding is yet to be determined, we warn against the use of NNSs by women who are breastfeeding.51
WHAT EFFECT—IF ANY—DO NNSs HAVE ON GUT MICROBIOTA?
We don’t know. Disruptions in the gut microbiome have been linked to numerous metabolic abnormalities, including obesity, insulin resistance, and diabetes, as well as cardiovascular disorders.52,53 Diet is a main determinant of balance in the gut microbiota.54 The gut microbiota are centrally involved in energy harvest, and studies have suggested that low gut bacterial diversity is associated with increased adiposity, insulin resistance, and low-grade inflammation.55-60 Whether NNSs have a relationship with abnormal changes in gut microbiota requires further study.
CORRESPONDENCE
Clipper F. Young, PharmD, MPH, CDE, BC-ADM, BCGP, Touro University California, College of Osteopathic Medicine, 1310 Club Drive, Vallejo, CA 94592; [email protected].
An estimated 93.3 million Americans (roughly 40% of the US population) were obese in 2015-2016, and most of them had at least 1 chronic disease.1 As a result, patient education focused on lifestyle modification, including healthy nutrition and physical activity, has become an integral part of our everyday practice.
At the same time, the most recent dietary guidelines recommend that added sugar make up < 10% of daily calories.2 In the United States, low-calorie food and beverages containing non-nutritive sweeteners (NNSs; TABLE3-8) have become a popular means of keeping the sweetness in our diet without the health ramifications associated with sugar. These NNSs (aka, artificial sweeteners, high-intensity sweeteners, and non-caloric sweeteners) are ubiquitous in soft drinks, processed grains (including breads, cereals, and granola bars), and dairy products (including yogurts, flavored milk, and ice cream). As examples, NNSs are present in 42% of flavored waters, 33% of yogurts, and all diet beverages.9,10 They can even be found in medications, multivitamins, toothpaste, and mouthwash.

Business is booming
Global NNS consumption has been growing more than 5% per year, meaning that by 2020, NNSs are expected to be a $2.2 billion industry.11 One study using data from the National Health and Nutrition Examination Survey (NHANES) found that the use of NNSs in the United States increased from 21.1% in 2003 to 24.9% in 2009-2010 among adults and increased from 7.8% to 18.9% over the same time period among children.12
The main increase in the consumption of NNSs across all age groups has been via the consumption of beverages. Approximately 11% of healthy weight, 19% of overweight, and 22% of obese adults consume diet beverages.13,14 Consumption of diet beverages or NNSs increases with age12 and is especially common among women with higher levels of education and income.15
However, concerns remain about the safety of these agents and their effect on weight, appetite, and the body’s glycemic response. This article reviews the available research and current recommendations regarding the use of NNSs.
WHAT EFFECT DO NNS s HAVE ON WEIGHT?
The data on NNSs and weight are inconsistent. One randomized controlled trial(RCT) compared weight loss over the course of 1 year (12-week weight loss phase; 9-month weight maintenance phase) when 303 participants consumed either water or drinks sweetened with NNSs.16 Weight loss was significantly greater in the NNS drink group when compared with the water group.16
Observational studies have revealed similar findings.17,18 Data from NHANES revealed that US adults (n = 14,098) during 2 nonconsecutive 24-hour dietary recall periods demonstrated lower total energy (calorie) intake if they consumed NNSs vs no NNSs.19 Another study using 2011-2016 NHANES data on adolescents (n = 7026) found no difference in energy intake between those who consumed beverages containing NNSs vs those who consumed beverages containing sugar.20
Continue to: Other lines of investigation...
Other lines of investigation, including animal studies, have shown that long-term use of NNSs is associated with numerous metabolic derangements including weight gain.21 The negative effects of NNSs appear to be the greatest in males and those who are obese and have high-calorie diets.21
A 2017 meta-analysis concluded that evidence from RCTs does not support a benefit of NNSs on weight management, and that routine consumption of NNSs may be associated with increased body mass index (BMI) and cardiometabolic risk.22 Another systematic review and meta-analysis found that there was a higher pooled risk for obesity among those who drank beverages containing NNSs vs those who drank sugar-containing beverages.23
Based on the most current literature, we conclude that NNSs are not beneficial for weight loss. While there is concern about weight gain through psychological effects (stimulation of sweetness receptors without satiety), further well-designed research is needed to explore whether this concern has merit.
WHAT IS THE EFFECT OF NNSs ON APPETITE?
There appears to be no effect. While original studies seemed to indicate there was an effect, later studies leaned to the contrary.
The notion that NNSs might enhance appetite and food intake was advanced in the 1980s by John Blundell and his research team.24 The hypothesis was that since NNSs uncouple sweet taste and calories, they do not exert the normal post-ingestive inhibitory influence that real sugar does. This, in turn, disrupts appetite control mechanisms.25-27
Continue to: However, subsequent research studies...
However, subsequent research studies found no relationship between the use of NNSs and appetite.28-30 Mattes and colleagues hypothesized that such a difference in findings could result from the fact that earlier studies focused on isolating NNSs from other energy-yielding products, which emphasized an association with heightened hunger.29 Subsequent studies showed that when NNSs were incorporated into energy-yielding products, there was no association between NNSs and increased hunger or appetite.
DO NNSs INCREASE THE RISK FOR TYPE 2 DIABETES MELLITUS?
The data are mixed. One study of women participating in the Nurses’ Health Study II showed that those who consumed caffeinated, artificially-sweetened beverages had a 35% higher risk of developing type 2 diabetes mellitus (T2DM); however, this risk was no longer significant after adjusting for BMI and energy intake.31
The Health Professionals Follow-Up Trial studied more than 40,000 men for more than 20 years and found that NNS consumption increased the risk of developing T2DM by 40%.32 However, this finding lost statistical significance after adjusting for BMI.32
These results make it difficult to determine whether there is any association between NNSs and T2DM; rather NNS-containing beverages are likely consumed more often by those who have higher BMIs and by those trying to lose weight.
A 2017 randomized crossover study involving 10 healthy men looked at the effects of a variety of caloric and non-caloric sweeteners on 24-hour glucose profiles and found no differences.33 Another study, a randomized, double-blind, crossover trial involving 60 non-obese adults without diabetes who did not consume NNSs, randomized the participants one-to-one to drink either 2 cans per day of either a beverage containing aspartame and acesulfame K or an unsweetened, no-calorie beverage for 12 weeks.34
Continue to: After a 4-week washout period...
After a 4-week washout period, the participants then switched to the opposite beverage for 12 weeks. The study concluded that consumption of 2 cans of a beverage containing aspartame and acesulfame K over 12 weeks had no significant effect on insulin sensitivity or secretion in nondiabetic adults.34
Similar results were obtained from a study involving 100 non-obese adults.35 The researchers found that aspartame ingested at 2 different doses (350 or 1050 mg/d) in beverages over 12 weeks had no effect on a 240-minute oral glucose tolerance test, blood pressure, appetite, or body weight.35
A 2016 systematic review critically evaluated the effect of NNSs on both glucose absorption and appetite.36 The review included 14 observational prospective trials, 28 RCTs, and 2 meta-analyses. The sweeteners studied included aspartame, sucralose, saccharin, acesulfame K, and stevia.36 The studies were focused largely on single-exposure outcomes (20 trials), but a minority of the studies (8 trials) looked at longer exposures from 1 to 18 weeks. Only some of the studies controlled for critical variables, such as BMI. In the end, there was no consistent pattern of increased or decreased risk for insulin resistance or diabetes.36
Two meta-analyses tried to determine if an association exists between consumption of beverages containing NNSs and the development of T2DM.37,38 The first meta-analysis with 4 studies showed a slight, but significant, relative risk (RR) of 1.13 (95% confidence interval [CI], 1.02-1.25) for those who consumed beverages containing NNSs.37 In the second meta-analysis (10 studies), NNS consumption had an RR of 1.48 (95% CI, 1.35-1.62), but the risk was lower (and no longer significant) after adjusting for BMI.38 A study of 98 Hispanic adolescents who were overweight or obese found that chronic users (n = 9) of NNSs had higher HbA1c levels 1 year later than did controls (n = 75) and people who initiated use of NNSs between the baseline and 1-year visit (n = 14).39
The American Diabetes Association (ADA) and American Heart Association joint position statement on NNSs, first published in 2012, says that NNSs can be utilized to reduce caloric and carbohydrate consumption for overall diabetes control and to obtain a healthy body weight.40 These principles were reaffirmed in the ADA Standards of Care in 2019.41
Continue to: The 2015 US Scientific Reports on Dietary Guidelines...
The 2015 US Scientific Reports on Dietary Guidelines provided a consensus statement saying, “Future experimental studies should examine the relationship between artificially sweetened soft drinks and biomarkers of insulin resistance and other diabetes markers.”42
DO NNSs HAVE ANY ADVERSE HEALTH EFFECTS?
Maybe. Many individuals avoid NNSs due to fear of developing cancer. While rat studies have previously shown a dose-dependent increased risk of developing cancer, epidemiologic studies in humans have not confirmed an association.43 The National Cancer Institute reports that carcinogenicity studies of NNSs have not shown an association with cancer in humans.44
A prospective study—the Nurses’ Health Study, which followed over 88,000 women for 24 years—found that consumption of > 2 diet sodas per day was associated with an increased risk for coronary heart disease (CHD) and chronic kidney disease (CKD) compared with consumption of < 1 diet soda per month.45 However, other prospective studies have shown that these specific negative health effects may not be present when controlling for weight.45,46
While the prospective studies found some associations between medical conditions (eg, CHD and CKD) and NNS consumption, the literature is limited to intake from beverages and does not include NNS-containing foods. More studies are needed to determine the relationship between NNSs and potential adverse health events, since the current literature is observational and cannot predict causation.
A 2019 study explored the associations between long-term consumption of sugar-sweetened beverages and artificially sweetened beverages (ASBs) and the risk of mortality in the United States.47 This study included 37,716 men from the Health Professionals Follow-up Study and 80,647 women from the Nurses’ Health Study. Subjects who had the highest consumption of ASBs had higher risks for total and cardiovascular disease mortality.47 Cohort-specific analyses showed that an association between ASB consumption and mortality was observed in the participants from the Nurses’ Health Study but not in those from the Health Professionals Follow-up Study, warranting further investigation.47 Cancer mortality and ASB consumption were not shown to have an association in this study.
Continue to: WHY ARE THE DATA INCONCLUSIVE?
WHY ARE THE DATA INCONCLUSIVE?
Nutritional studies are hard to complete accurately outside of the laboratory setting. Also, the science of NNSs is new and evolving.
With regard to obesity and NNSs, it is possible that findings have been due to reverse causation. People who are overweight or obese are more likely to consume low-calorie foods and beverages; they are also at greater risk for developing diseases, such as T2DM.48,49
HOW SAFE ARE NNSs?
They appear to be safe, but more data are needed. Each of the 7 FDA-approved NNSs has passed extensive laboratory, animal, and human testing, and appears to cause no harm in the human body when consumed.49 But clearly the data are incomplete. As we continue to gain a greater understanding of the metabolism of NNSs, we may need to revisit the issue of safety.
ARE THERE ANY NNSs THAT SOME PEOPLE SHOULD AVOID?
Yes. People with phenylketonuria, who have difficulty metabolizing phenylalanine (a component of aspartame), should avoid consumption of aspartame.50
In addition, NNSs have been found to be present in breast milk.51 While the significance of this finding is yet to be determined, we warn against the use of NNSs by women who are breastfeeding.51
WHAT EFFECT—IF ANY—DO NNSs HAVE ON GUT MICROBIOTA?
We don’t know. Disruptions in the gut microbiome have been linked to numerous metabolic abnormalities, including obesity, insulin resistance, and diabetes, as well as cardiovascular disorders.52,53 Diet is a main determinant of balance in the gut microbiota.54 The gut microbiota are centrally involved in energy harvest, and studies have suggested that low gut bacterial diversity is associated with increased adiposity, insulin resistance, and low-grade inflammation.55-60 Whether NNSs have a relationship with abnormal changes in gut microbiota requires further study.
CORRESPONDENCE
Clipper F. Young, PharmD, MPH, CDE, BC-ADM, BCGP, Touro University California, College of Osteopathic Medicine, 1310 Club Drive, Vallejo, CA 94592; [email protected].
1. Adult obesity facts. Centers for Disease Control and Prevention Web site. https://www.cdc.gov/obesity/data/adult.html. Reviewed August 13, 2018. Accessed July 15, 2019.
2. Dietary guidelines for Americans 2015-2020: answers to your questions. USDA ChooseMyPlate.gov Web site. https://www.choosemyplate.gov/2015-2020-dietary-guidelines-answers-your-questions. Accessed July 15, 2019.
3. Additional information about high-intensity sweeteners permitted for use in food in the United States. US Food and Drug Administration Web site. https://www.fda.gov/food/food-additives-petitions/additional-information-about-high-intensity-sweeteners-permitted-use-food-united-states. Published February 8, 2018. Accessed July 15, 2019.
4. Magnuson B, for the Aspartame Expert Work Group. Nutritive and non-nutritive sweeteners. NNNS: aspartame, methanol and formaldehyde relationships (2011). https://www.foodsweeteners.com/wp-content/uploads/2015/08/Aspartame-Methanol-and-Formaldehyde-Relationships.pdf. Accessed July 15, 2019.
5. Jo JH, Kim S, Jeon TW, et al. Investigation of the regulatory effects of saccharin on cytochrome P450s in male ICR mice. Toxicol Res. 2017;33:25-30.
6. Shwide-Slavin C, Swift C, Ross T. Nonnutritive sweetener: where are we today? Diabetes Spectrum. 2012;25:104-110.
7. Chattopadhyay S, Raychaudhuri U, Chakraborty R. Artificial sweeteners – a review. J Food Sci Technol. 2014;51:611-621.
8. EFSA Panel on Food Additives and Nutrient Sources added to Food. Scientific opinion on the safety of advantame for the proposed uses as a food additive. EFSA Journal. 2013;11:3301.
9. Fitch C, Keim KS; Academy of Nutrition and Dietetics. Position of the Academy of Nutrition and Dietetics: use of nutritive and nonnutritive sweeteners. J Acad Nutr Diet. 2012;112:739-758.
10. Ng SW, Slining MM, Popkin BM. Use of caloric and non-caloric sweeteners in US consumer packaged foods, 2005-2009. J Acad Nutr Diet. 2012;112:1828-1834.
11. Sylvetsky AC, Rother KI. Trends in the consumption of low-calorie sweeteners. Physiol Behav. 2016;164(Pt B):446-450.
12. Piernas C, Ng SW, Popkin B. Trends in purchases and intake of foods and beverages containing caloric and low-calorie sweeteners over the last decade in the United States. Pediatr Obes. 2013;8:294-306.
13. Sylvetsky AC, Welsh JA, Brown RJ, et al. Low-calorie sweetener consumption is increasing in the United States. Am J Clin Nutr. 2012;96:640-646.
14. Bleich SN, Wolfson JA, Vine S, et al. Diet-beverage consumption and caloric intake among US adults, overall and by body weight. Am J Public Health. 2014;104:e72-e78.
15. Drewnowski A, Rehm CD. Socio-demographic correlates and trends in low-calorie sweetener use among adults in the United States from 1999 to 2008. Eur J Clin Nutr. 2015;69:1035-1041.
16. Peters JC, Wyatt HR, Foster GD, et al. The effects of water and non-nutritive sweetened beverages on weight loss during a 12-week weight loss treatment program. Obesity 2014;22:1415-1421.
17. Peters JC, Wyatt HR, Foster GD, et al. The effects of water and non-nutritive sweetened beverages on weight loss during a 12-week weight loss treatment program. Obesity (Silver Spring). 2014;22:1415-1421.
18. Bellisle F, Drewnowski A. Intense sweeteners, energy intake and the control of body weight. Eur J Clin Nutr. 2007;61:691-700.
19. Malek AM, Hunt KJ, DellaValle DM, et al. Reported consumption of low-calorie sweetener in foods, beverages, and food and beverage additions by US adults: NHANES 2007-2012. Curr Dev Nutr. 2018;2:nzy054.
20. Sylvetsky AC, Figueroa J, Zimmerman T, et al. Consumption of low-calorie sweetened beverages is associated with higher total energy and sugar intake among children, NHANES 2011-2016. Pediatr Obes. 2019;2:e12535.
21. Fowler SPG. Low-calorie sweetener use and energy balance: results from experimental studies in animals, and large-scale prospective studies in humans. Physiol Behav. 2016;164(Pt B):517-523.
22. Azad MB, Abou-Setta AM, Chauhan BF, et al. Nonnutritive sweeteners and cardiometabolic health: a systematic review and meta-analysis of randomized controlled trials and prospective cohort studies. CMAJ. 2017;189: E929-E939.
23. Ruanpeng D, Thongprayoon C, Cheungpasitporn W, et al. Sugar and artificially-sweetened beverages linked to obesity: a systematic review and meta-analysis. QJM. 2017;110:513-520.
24. Blundell JE, Rogers PJ, Hill AJ. Uncoupling sweetness and calories: methodological aspects of laboratory studies on appetite control. Appetite. 1988;11(Suppl 1):54-61.
25. Bellisle F. Intense sweeteners, appetite for the sweet taste, and relationship to weight management. Curr Obes Rep. 2015;4:106-110.
26. Bryant CE, Wasse LK, Astbury N, et al. Non-nutritive sweeteners: no class effect on the glycaemic or appetite responses to ingested glucose. Eur J Clin Nutr. 2014;68:629-631.
27. Canty DJ, Chan MM. Effects of consumption of caloric vs noncaloric sweet drinks on indices of hunger and food consumption in normal adults. Am J Clin Nutr. 1991;53:1159-1164.
28. Meyer-Gerspach AC, Wolnerhanssen B, Beglinger C. Functional roles of low calorie sweeteners on gut function. Physiol Behav. 2016;164(Pt B):479-481.
29. Mattes RD, Popkin BM. Nonnutritive sweetener consumption in humans: effects on appetite and food intake and their putative mechanisms. Am J Clin Nutr. 2009;89:1-14.
30. Bhupathiraju SN, Pan A, Malik VS, et al. Caffeinated and caffeine-free beverages and risk of type 2 diabetes. Am J Clin Nutr. 2013;97:155-166.
31. Schulze MB, Manson JE, Ludwig DS, et al. Sugar-sweetened beverages, weight gain, and incidence of type 2 diabetes in young and middle-aged women. JAMA. 2004;292:927-934.
32. de Koning L, Malik VS, Rimm EB, et al. Sugar-sweetened and artificially sweetened beverage consumption and the risk of type 2 diabetes in men. Am J Clin Nutr. 2011;93:1321-1327.
33. Tey SL, Salleh NB, Henry CJ, et al. Effect of non-nutritive (artificial vs natural) sweeteners on 24-hour glucose profile. Eur J Clin Nutr. 2017;71:1129-1132.
34. Bonnet F, Tavenard A, Esvan M, et al. Consumption of a carbonated beverage with high-intensity sweeteners has no effect on insulin sensitivity and secretion in nondiabetic adults. J Nutr. 2018;148:1293-1299.
35. Higgins KA, Considine RV, Mattes RD. Aspartame consumption for 12 weeks does not affect glycemia, appetite, or body weight of healthy, lean adults in a randomized controlled trial. J Nutr. 2018;148:650-657.
36. Romo-Romo A, Aguilar-Salinas CA, Brito-Cordova GX, et al. Effects of the non-nutritive sweeteners on glucose metabolism and appetite regulating hormones: systematic review of observational prospective studies and clinical trials. PloS One. 2016;11:e0161264.
37. Greenwood DC, Threspleton DE, Evans CE, et al. Association between sugar-sweetened and artificially sweetened soft drinks and type 2 diabetes: systematic review and dose-response meta-analysis of prospective studies. Br J Nutr. 2014;112:725-734.
38. Imamura F, O’Conner L, Ye M, et al. Consumption of sugar sweetened beverages, artificially sweetened beverages, and fruit juice and incidence of type 2 diabetes: systematic review, meta-analysis, and estimation of population attributable fraction. BMJ. 2015;351:h3576.
39. Davis JN, Asigbee FM, Markowitz AK, et al. Consumption of artificial sweetened beverages associated with adiposity and increasing HbA1c in Hispanic youth. Clin Obes. 2018;8:236-243.
40. Gardner C, Wylie-Rosett J, Gidding SS, et al. Nonnutritive sweeteners: current use and health perspectives. a scientific statement from the American Heart Association and the American Diabetes Association. Diabetes Care. 2012;35:1798-1808.
41. American Diabetes Association. Standards of Medical Care in Diabetes—2019. Diabetes Care. 2019;42(Suppl 1):S1-S183.
42. Dietary Guidelines Advisory Committee. Scientific Report of the 2015 Dietary Guidelines Advisory Committee: advisory report to the Secretary of Health and Human Services and the Secretary of Agriculture. Office of Disease Prevention and Health Promotion Web site. https://health.gov/dietaryguidelines/2015-scientific-report/.Published February 2015. Accessed July 15, 2019.
43. Aune D. Soft drinks, aspartame, and the risk of cancer and cardiovascular disease. Am J Clin Nutr. 2012;96:1249-1251.
44. Artificial sweeteners and cancer. National Cancer Institute Web site. https://www.cancer.gov/about-cancer/causes-prevention/risk/diet/artificial-sweeteners-fact-sheet. Reviewed August 10, 2016. Accessed July 15, 2019.
45. Fung TT, Malik V, Rexrode KM, et al. Sweetened beverage consumption and risk of coronary heart disease in women. Am J Clin Nutr. 2009;89:1037-1042.
46. Lin J, Curhan GC. Associations of sugar and artificially sweetened soda with albuminuria and kidney function decline in women. Clin J Am Soc Nephrol. 2011;6:160-166.
47. Malik VS, Li Y, Pan A, et al. Long-term consumption of sugar-sweetened and artificially sweetened beverages and risk of mortality in US adults. Circulation. 2019;139:2113-2125.
48. Gardener H, Rundek T, Markert M, et al. Diet soft drink consumption is associated with an increased risk of vascular events in the Northern Manhattan Study. J Gen Inten Med. 2012;27:1120-1126.
49. Fitch C, Keim KS. Position of the Academy of Nutrition and Dietetics: use of nutritive and nonnutritive sweeteners. J Acad Nutr Diet. 2012;112:739-758.
50. US Food and Drug Administration. Additional information about high-intensity sweeteners permitted for use in food in the United States. https://www.fda.gov/food/ingredientspackaginglabeling/foodadditivesingredients/ucm397725.htm#Aspartame. Accessed May 26, 2019.
51. Sylvetsky AC, Gardner AL, Bauman V, et al. Nonnutritive sweeteners in breast milk. J Toxicol Environ Health. 2015;78:1029-1032.
52. Rajani C, Jia W. Disruptions in gut microbial-host co-metabolism and the development of metabolic disorders. Clin Sci (Lond). 2018;132:791-811.
53. Kho ZY, Lal SK. The human gut microbiome—a potential controller of wellness and disease. Front Microbiol. 2018;9:1835.
54. Nettleton JE, Reimer RA, Shearer J. Reshaping the gut microbiota: impact of low calorie sweeteners and the link to insulin resistance. Physiol Behav. 2016;164(Pt B):488-493.
55. Turnbaugh PJ, Hamady M, Yatsunenko T, et al. A core gut microbiome in obese and lean twins. Nature. 2009;457:480-484.
56. Cotillard A, Kennedy SP, Kong LC, et al. Dietary intervention impact on gut microbial gene richness. Nature. 2013;500:585-588.
57. Le Chatelier E, Nielsen T, Qin J, et al. Richness of human gut microbiome correlates with metabolic markers. Nature. 2013;500:541-546.
58. Abou-Donia MB, El-Masry EM, Abdel-Rahman AA, et al. Splenda alters gut microflora and increases intestinal p-glycoprotein and cytochrome P-450 in male rats. J Toxicol Environ Health A. 2008;71:1415-1429.
59. Anderson RL. Effect of saccharin ingestion on stool composition in relation to caecal enlargement and increased stool hydration. Food Chem Toxicol. 1983;21:255-257.
60. Suez J, Korem T, Zeevi D, et al. Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature. 2014;514:181-186.
1. Adult obesity facts. Centers for Disease Control and Prevention Web site. https://www.cdc.gov/obesity/data/adult.html. Reviewed August 13, 2018. Accessed July 15, 2019.
2. Dietary guidelines for Americans 2015-2020: answers to your questions. USDA ChooseMyPlate.gov Web site. https://www.choosemyplate.gov/2015-2020-dietary-guidelines-answers-your-questions. Accessed July 15, 2019.
3. Additional information about high-intensity sweeteners permitted for use in food in the United States. US Food and Drug Administration Web site. https://www.fda.gov/food/food-additives-petitions/additional-information-about-high-intensity-sweeteners-permitted-use-food-united-states. Published February 8, 2018. Accessed July 15, 2019.
4. Magnuson B, for the Aspartame Expert Work Group. Nutritive and non-nutritive sweeteners. NNNS: aspartame, methanol and formaldehyde relationships (2011). https://www.foodsweeteners.com/wp-content/uploads/2015/08/Aspartame-Methanol-and-Formaldehyde-Relationships.pdf. Accessed July 15, 2019.
5. Jo JH, Kim S, Jeon TW, et al. Investigation of the regulatory effects of saccharin on cytochrome P450s in male ICR mice. Toxicol Res. 2017;33:25-30.
6. Shwide-Slavin C, Swift C, Ross T. Nonnutritive sweetener: where are we today? Diabetes Spectrum. 2012;25:104-110.
7. Chattopadhyay S, Raychaudhuri U, Chakraborty R. Artificial sweeteners – a review. J Food Sci Technol. 2014;51:611-621.
8. EFSA Panel on Food Additives and Nutrient Sources added to Food. Scientific opinion on the safety of advantame for the proposed uses as a food additive. EFSA Journal. 2013;11:3301.
9. Fitch C, Keim KS; Academy of Nutrition and Dietetics. Position of the Academy of Nutrition and Dietetics: use of nutritive and nonnutritive sweeteners. J Acad Nutr Diet. 2012;112:739-758.
10. Ng SW, Slining MM, Popkin BM. Use of caloric and non-caloric sweeteners in US consumer packaged foods, 2005-2009. J Acad Nutr Diet. 2012;112:1828-1834.
11. Sylvetsky AC, Rother KI. Trends in the consumption of low-calorie sweeteners. Physiol Behav. 2016;164(Pt B):446-450.
12. Piernas C, Ng SW, Popkin B. Trends in purchases and intake of foods and beverages containing caloric and low-calorie sweeteners over the last decade in the United States. Pediatr Obes. 2013;8:294-306.
13. Sylvetsky AC, Welsh JA, Brown RJ, et al. Low-calorie sweetener consumption is increasing in the United States. Am J Clin Nutr. 2012;96:640-646.
14. Bleich SN, Wolfson JA, Vine S, et al. Diet-beverage consumption and caloric intake among US adults, overall and by body weight. Am J Public Health. 2014;104:e72-e78.
15. Drewnowski A, Rehm CD. Socio-demographic correlates and trends in low-calorie sweetener use among adults in the United States from 1999 to 2008. Eur J Clin Nutr. 2015;69:1035-1041.
16. Peters JC, Wyatt HR, Foster GD, et al. The effects of water and non-nutritive sweetened beverages on weight loss during a 12-week weight loss treatment program. Obesity 2014;22:1415-1421.
17. Peters JC, Wyatt HR, Foster GD, et al. The effects of water and non-nutritive sweetened beverages on weight loss during a 12-week weight loss treatment program. Obesity (Silver Spring). 2014;22:1415-1421.
18. Bellisle F, Drewnowski A. Intense sweeteners, energy intake and the control of body weight. Eur J Clin Nutr. 2007;61:691-700.
19. Malek AM, Hunt KJ, DellaValle DM, et al. Reported consumption of low-calorie sweetener in foods, beverages, and food and beverage additions by US adults: NHANES 2007-2012. Curr Dev Nutr. 2018;2:nzy054.
20. Sylvetsky AC, Figueroa J, Zimmerman T, et al. Consumption of low-calorie sweetened beverages is associated with higher total energy and sugar intake among children, NHANES 2011-2016. Pediatr Obes. 2019;2:e12535.
21. Fowler SPG. Low-calorie sweetener use and energy balance: results from experimental studies in animals, and large-scale prospective studies in humans. Physiol Behav. 2016;164(Pt B):517-523.
22. Azad MB, Abou-Setta AM, Chauhan BF, et al. Nonnutritive sweeteners and cardiometabolic health: a systematic review and meta-analysis of randomized controlled trials and prospective cohort studies. CMAJ. 2017;189: E929-E939.
23. Ruanpeng D, Thongprayoon C, Cheungpasitporn W, et al. Sugar and artificially-sweetened beverages linked to obesity: a systematic review and meta-analysis. QJM. 2017;110:513-520.
24. Blundell JE, Rogers PJ, Hill AJ. Uncoupling sweetness and calories: methodological aspects of laboratory studies on appetite control. Appetite. 1988;11(Suppl 1):54-61.
25. Bellisle F. Intense sweeteners, appetite for the sweet taste, and relationship to weight management. Curr Obes Rep. 2015;4:106-110.
26. Bryant CE, Wasse LK, Astbury N, et al. Non-nutritive sweeteners: no class effect on the glycaemic or appetite responses to ingested glucose. Eur J Clin Nutr. 2014;68:629-631.
27. Canty DJ, Chan MM. Effects of consumption of caloric vs noncaloric sweet drinks on indices of hunger and food consumption in normal adults. Am J Clin Nutr. 1991;53:1159-1164.
28. Meyer-Gerspach AC, Wolnerhanssen B, Beglinger C. Functional roles of low calorie sweeteners on gut function. Physiol Behav. 2016;164(Pt B):479-481.
29. Mattes RD, Popkin BM. Nonnutritive sweetener consumption in humans: effects on appetite and food intake and their putative mechanisms. Am J Clin Nutr. 2009;89:1-14.
30. Bhupathiraju SN, Pan A, Malik VS, et al. Caffeinated and caffeine-free beverages and risk of type 2 diabetes. Am J Clin Nutr. 2013;97:155-166.
31. Schulze MB, Manson JE, Ludwig DS, et al. Sugar-sweetened beverages, weight gain, and incidence of type 2 diabetes in young and middle-aged women. JAMA. 2004;292:927-934.
32. de Koning L, Malik VS, Rimm EB, et al. Sugar-sweetened and artificially sweetened beverage consumption and the risk of type 2 diabetes in men. Am J Clin Nutr. 2011;93:1321-1327.
33. Tey SL, Salleh NB, Henry CJ, et al. Effect of non-nutritive (artificial vs natural) sweeteners on 24-hour glucose profile. Eur J Clin Nutr. 2017;71:1129-1132.
34. Bonnet F, Tavenard A, Esvan M, et al. Consumption of a carbonated beverage with high-intensity sweeteners has no effect on insulin sensitivity and secretion in nondiabetic adults. J Nutr. 2018;148:1293-1299.
35. Higgins KA, Considine RV, Mattes RD. Aspartame consumption for 12 weeks does not affect glycemia, appetite, or body weight of healthy, lean adults in a randomized controlled trial. J Nutr. 2018;148:650-657.
36. Romo-Romo A, Aguilar-Salinas CA, Brito-Cordova GX, et al. Effects of the non-nutritive sweeteners on glucose metabolism and appetite regulating hormones: systematic review of observational prospective studies and clinical trials. PloS One. 2016;11:e0161264.
37. Greenwood DC, Threspleton DE, Evans CE, et al. Association between sugar-sweetened and artificially sweetened soft drinks and type 2 diabetes: systematic review and dose-response meta-analysis of prospective studies. Br J Nutr. 2014;112:725-734.
38. Imamura F, O’Conner L, Ye M, et al. Consumption of sugar sweetened beverages, artificially sweetened beverages, and fruit juice and incidence of type 2 diabetes: systematic review, meta-analysis, and estimation of population attributable fraction. BMJ. 2015;351:h3576.
39. Davis JN, Asigbee FM, Markowitz AK, et al. Consumption of artificial sweetened beverages associated with adiposity and increasing HbA1c in Hispanic youth. Clin Obes. 2018;8:236-243.
40. Gardner C, Wylie-Rosett J, Gidding SS, et al. Nonnutritive sweeteners: current use and health perspectives. a scientific statement from the American Heart Association and the American Diabetes Association. Diabetes Care. 2012;35:1798-1808.
41. American Diabetes Association. Standards of Medical Care in Diabetes—2019. Diabetes Care. 2019;42(Suppl 1):S1-S183.
42. Dietary Guidelines Advisory Committee. Scientific Report of the 2015 Dietary Guidelines Advisory Committee: advisory report to the Secretary of Health and Human Services and the Secretary of Agriculture. Office of Disease Prevention and Health Promotion Web site. https://health.gov/dietaryguidelines/2015-scientific-report/.Published February 2015. Accessed July 15, 2019.
43. Aune D. Soft drinks, aspartame, and the risk of cancer and cardiovascular disease. Am J Clin Nutr. 2012;96:1249-1251.
44. Artificial sweeteners and cancer. National Cancer Institute Web site. https://www.cancer.gov/about-cancer/causes-prevention/risk/diet/artificial-sweeteners-fact-sheet. Reviewed August 10, 2016. Accessed July 15, 2019.
45. Fung TT, Malik V, Rexrode KM, et al. Sweetened beverage consumption and risk of coronary heart disease in women. Am J Clin Nutr. 2009;89:1037-1042.
46. Lin J, Curhan GC. Associations of sugar and artificially sweetened soda with albuminuria and kidney function decline in women. Clin J Am Soc Nephrol. 2011;6:160-166.
47. Malik VS, Li Y, Pan A, et al. Long-term consumption of sugar-sweetened and artificially sweetened beverages and risk of mortality in US adults. Circulation. 2019;139:2113-2125.
48. Gardener H, Rundek T, Markert M, et al. Diet soft drink consumption is associated with an increased risk of vascular events in the Northern Manhattan Study. J Gen Inten Med. 2012;27:1120-1126.
49. Fitch C, Keim KS. Position of the Academy of Nutrition and Dietetics: use of nutritive and nonnutritive sweeteners. J Acad Nutr Diet. 2012;112:739-758.
50. US Food and Drug Administration. Additional information about high-intensity sweeteners permitted for use in food in the United States. https://www.fda.gov/food/ingredientspackaginglabeling/foodadditivesingredients/ucm397725.htm#Aspartame. Accessed May 26, 2019.
51. Sylvetsky AC, Gardner AL, Bauman V, et al. Nonnutritive sweeteners in breast milk. J Toxicol Environ Health. 2015;78:1029-1032.
52. Rajani C, Jia W. Disruptions in gut microbial-host co-metabolism and the development of metabolic disorders. Clin Sci (Lond). 2018;132:791-811.
53. Kho ZY, Lal SK. The human gut microbiome—a potential controller of wellness and disease. Front Microbiol. 2018;9:1835.
54. Nettleton JE, Reimer RA, Shearer J. Reshaping the gut microbiota: impact of low calorie sweeteners and the link to insulin resistance. Physiol Behav. 2016;164(Pt B):488-493.
55. Turnbaugh PJ, Hamady M, Yatsunenko T, et al. A core gut microbiome in obese and lean twins. Nature. 2009;457:480-484.
56. Cotillard A, Kennedy SP, Kong LC, et al. Dietary intervention impact on gut microbial gene richness. Nature. 2013;500:585-588.
57. Le Chatelier E, Nielsen T, Qin J, et al. Richness of human gut microbiome correlates with metabolic markers. Nature. 2013;500:541-546.
58. Abou-Donia MB, El-Masry EM, Abdel-Rahman AA, et al. Splenda alters gut microflora and increases intestinal p-glycoprotein and cytochrome P-450 in male rats. J Toxicol Environ Health A. 2008;71:1415-1429.
59. Anderson RL. Effect of saccharin ingestion on stool composition in relation to caecal enlargement and increased stool hydration. Food Chem Toxicol. 1983;21:255-257.
60. Suez J, Korem T, Zeevi D, et al. Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature. 2014;514:181-186.
PRACTICE RECOMMENDATIONS
› Advise patients who are trying to lose weight that non-nutritive sweeteners (NNSs) are not beneficial for weight loss. A
› Reassure patients that NNSs do not appear to cause, or increase the risk of, developing type 2 diabetes mellitus. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
GLP-1 agonists, SGLT2 inhibitors offer more options in diabetes management
ORLANDO – The big news in diabetes management this year is “happy cardiologists and nephrologists.”
According to Christine Kessler, MN, ANP-C, CNS, BC-ADM, FAANP, founder of Metabolic Medicine Associates in King George, Va., these specialists are happy because the American College of Cardiology and the American Diabetes Association both recently updated their respective societies’ guidelines to include evidence that treating patients with type 2 diabetes with glucagonlike peptide-1 (GLP-1) agonists, sodium-glucose cotransporter 2 (SGLT2) inhibitors, or metformin can lower risk of cardiovascular disease and chronic kidney disease.
“Finally, the ACC is aligned with the ADA,” Ms. Kessler said in her presentation. “This is amazing, and it’s good news.”
Recent innovations in diabetes management technology, such as continuous glucose monitors, are also helping to make diabetes management easier. “If you’re not using some of this technology in your primary care practice, it’s coming to you, and it’s amazing the data it can provide to us,” said Ms. Kessler at the Cardiovascular & Respiratory Summit by Global Academy for Medical Education.
In endocrinology, diabetes is thought of in terms of macrovascular and microvascular disease, she said. Macrovascular disease is cardiovascular disease and stroke, while microvascular disease is nephropathy, neuropathy, and retinopathy. Diabetes is a cardiovascular risk factor and puts patients at higher risk for cardiovascular death, all-cause mortality, and hospitalization because of myocardial infarction or stroke, compared with patients who do not have type 2 diabetes. There is also a higher risk of kidney disease, nerve damage, blindness, nonalcoholic fatty liver disease, depression, complications during pregnancy, periodontal disease, and erectile dysfunction, said Ms. Kessler, who also is a nurse practitioner and researcher.
However, including getting enough sleep, dietary interventions that target weight loss and blood glucose control, and increasing physical activity that has cardiopulmonary benefits. Clinicians should also treat underlying conditions that contribute to increased cardiovascular risk, such as obesity, dyslipidemia, hypertension, and nonalcoholic fatty liver disease.
Addressing insulin resistance and hyperglycemia are also important, but patients must avoid hypoglycemia. “Any patient with diabetes, we don’t want to drive them there because that’s a cardiac risk,” said Ms. Kessler. The endothelial microvascular and macrovascular damage is believed to be caused by glycemic swings, she added.
For pharmacologic therapy, patients with type 2 diabetes should stay on metformin if they are already on the drug, and it can even be used in cases where patients have reduced kidney function, with a glomerular filtration rate (GFR) between 30 and 60 mL/min per 1.73 m2, with a lower dose used between 30 and 45 mL/min per 1.73 m2. To treat patients with atherosclerotic cardiovascular disease, recent evidence has shown GLP-1 agonists are beneficial and can also promote appetite satiety, prandial support, and reduce a patient’s weight, but the drug is expensive, and about 15% of patients will not see therapeutic benefit while on the medication, said Ms. Kessler. Clinicians should also watch for increased risk of pancreatitis while patients use GLP-1 agonists, and it should not be prescribed in patients with a history of thyroid medullary cancer or multiple endocrine neoplasia type 2 (MEN2).
SGLT2 inhibitors can benefit type 1 diabetes and type 2 diabetes patients with heart failure and diabetic kidney disease, but should be the second or third choice in therapy. The dosage of SGLT2 inhibitors should be cut in half when used with insulin and sulfonylurea, and the drug can also increase LDL cholesterol.
Ms. Kessler noted that while GLP-1 agonists and SGLT2 inhibitors prevent or reduce cardiovascular risk, they are not currently approved to treat cardiovascular disease.
Ms. Kessler reports being an advisor and speaker for Novo Nordisk on the subject of obesity. Global Academy for Medical Education and this news organization are owned by the same parent company.
ORLANDO – The big news in diabetes management this year is “happy cardiologists and nephrologists.”
According to Christine Kessler, MN, ANP-C, CNS, BC-ADM, FAANP, founder of Metabolic Medicine Associates in King George, Va., these specialists are happy because the American College of Cardiology and the American Diabetes Association both recently updated their respective societies’ guidelines to include evidence that treating patients with type 2 diabetes with glucagonlike peptide-1 (GLP-1) agonists, sodium-glucose cotransporter 2 (SGLT2) inhibitors, or metformin can lower risk of cardiovascular disease and chronic kidney disease.
“Finally, the ACC is aligned with the ADA,” Ms. Kessler said in her presentation. “This is amazing, and it’s good news.”
Recent innovations in diabetes management technology, such as continuous glucose monitors, are also helping to make diabetes management easier. “If you’re not using some of this technology in your primary care practice, it’s coming to you, and it’s amazing the data it can provide to us,” said Ms. Kessler at the Cardiovascular & Respiratory Summit by Global Academy for Medical Education.
In endocrinology, diabetes is thought of in terms of macrovascular and microvascular disease, she said. Macrovascular disease is cardiovascular disease and stroke, while microvascular disease is nephropathy, neuropathy, and retinopathy. Diabetes is a cardiovascular risk factor and puts patients at higher risk for cardiovascular death, all-cause mortality, and hospitalization because of myocardial infarction or stroke, compared with patients who do not have type 2 diabetes. There is also a higher risk of kidney disease, nerve damage, blindness, nonalcoholic fatty liver disease, depression, complications during pregnancy, periodontal disease, and erectile dysfunction, said Ms. Kessler, who also is a nurse practitioner and researcher.
However, including getting enough sleep, dietary interventions that target weight loss and blood glucose control, and increasing physical activity that has cardiopulmonary benefits. Clinicians should also treat underlying conditions that contribute to increased cardiovascular risk, such as obesity, dyslipidemia, hypertension, and nonalcoholic fatty liver disease.
Addressing insulin resistance and hyperglycemia are also important, but patients must avoid hypoglycemia. “Any patient with diabetes, we don’t want to drive them there because that’s a cardiac risk,” said Ms. Kessler. The endothelial microvascular and macrovascular damage is believed to be caused by glycemic swings, she added.
For pharmacologic therapy, patients with type 2 diabetes should stay on metformin if they are already on the drug, and it can even be used in cases where patients have reduced kidney function, with a glomerular filtration rate (GFR) between 30 and 60 mL/min per 1.73 m2, with a lower dose used between 30 and 45 mL/min per 1.73 m2. To treat patients with atherosclerotic cardiovascular disease, recent evidence has shown GLP-1 agonists are beneficial and can also promote appetite satiety, prandial support, and reduce a patient’s weight, but the drug is expensive, and about 15% of patients will not see therapeutic benefit while on the medication, said Ms. Kessler. Clinicians should also watch for increased risk of pancreatitis while patients use GLP-1 agonists, and it should not be prescribed in patients with a history of thyroid medullary cancer or multiple endocrine neoplasia type 2 (MEN2).
SGLT2 inhibitors can benefit type 1 diabetes and type 2 diabetes patients with heart failure and diabetic kidney disease, but should be the second or third choice in therapy. The dosage of SGLT2 inhibitors should be cut in half when used with insulin and sulfonylurea, and the drug can also increase LDL cholesterol.
Ms. Kessler noted that while GLP-1 agonists and SGLT2 inhibitors prevent or reduce cardiovascular risk, they are not currently approved to treat cardiovascular disease.
Ms. Kessler reports being an advisor and speaker for Novo Nordisk on the subject of obesity. Global Academy for Medical Education and this news organization are owned by the same parent company.
ORLANDO – The big news in diabetes management this year is “happy cardiologists and nephrologists.”
According to Christine Kessler, MN, ANP-C, CNS, BC-ADM, FAANP, founder of Metabolic Medicine Associates in King George, Va., these specialists are happy because the American College of Cardiology and the American Diabetes Association both recently updated their respective societies’ guidelines to include evidence that treating patients with type 2 diabetes with glucagonlike peptide-1 (GLP-1) agonists, sodium-glucose cotransporter 2 (SGLT2) inhibitors, or metformin can lower risk of cardiovascular disease and chronic kidney disease.
“Finally, the ACC is aligned with the ADA,” Ms. Kessler said in her presentation. “This is amazing, and it’s good news.”
Recent innovations in diabetes management technology, such as continuous glucose monitors, are also helping to make diabetes management easier. “If you’re not using some of this technology in your primary care practice, it’s coming to you, and it’s amazing the data it can provide to us,” said Ms. Kessler at the Cardiovascular & Respiratory Summit by Global Academy for Medical Education.
In endocrinology, diabetes is thought of in terms of macrovascular and microvascular disease, she said. Macrovascular disease is cardiovascular disease and stroke, while microvascular disease is nephropathy, neuropathy, and retinopathy. Diabetes is a cardiovascular risk factor and puts patients at higher risk for cardiovascular death, all-cause mortality, and hospitalization because of myocardial infarction or stroke, compared with patients who do not have type 2 diabetes. There is also a higher risk of kidney disease, nerve damage, blindness, nonalcoholic fatty liver disease, depression, complications during pregnancy, periodontal disease, and erectile dysfunction, said Ms. Kessler, who also is a nurse practitioner and researcher.
However, including getting enough sleep, dietary interventions that target weight loss and blood glucose control, and increasing physical activity that has cardiopulmonary benefits. Clinicians should also treat underlying conditions that contribute to increased cardiovascular risk, such as obesity, dyslipidemia, hypertension, and nonalcoholic fatty liver disease.
Addressing insulin resistance and hyperglycemia are also important, but patients must avoid hypoglycemia. “Any patient with diabetes, we don’t want to drive them there because that’s a cardiac risk,” said Ms. Kessler. The endothelial microvascular and macrovascular damage is believed to be caused by glycemic swings, she added.
For pharmacologic therapy, patients with type 2 diabetes should stay on metformin if they are already on the drug, and it can even be used in cases where patients have reduced kidney function, with a glomerular filtration rate (GFR) between 30 and 60 mL/min per 1.73 m2, with a lower dose used between 30 and 45 mL/min per 1.73 m2. To treat patients with atherosclerotic cardiovascular disease, recent evidence has shown GLP-1 agonists are beneficial and can also promote appetite satiety, prandial support, and reduce a patient’s weight, but the drug is expensive, and about 15% of patients will not see therapeutic benefit while on the medication, said Ms. Kessler. Clinicians should also watch for increased risk of pancreatitis while patients use GLP-1 agonists, and it should not be prescribed in patients with a history of thyroid medullary cancer or multiple endocrine neoplasia type 2 (MEN2).
SGLT2 inhibitors can benefit type 1 diabetes and type 2 diabetes patients with heart failure and diabetic kidney disease, but should be the second or third choice in therapy. The dosage of SGLT2 inhibitors should be cut in half when used with insulin and sulfonylurea, and the drug can also increase LDL cholesterol.
Ms. Kessler noted that while GLP-1 agonists and SGLT2 inhibitors prevent or reduce cardiovascular risk, they are not currently approved to treat cardiovascular disease.
Ms. Kessler reports being an advisor and speaker for Novo Nordisk on the subject of obesity. Global Academy for Medical Education and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM CARPS 2019