User login
Endocrine Society advises on diabetes care for older adults
according to a new guideline on diabetes care for older adults issued by the Endocrine Society.
“The prevalence of diabetes in the United States is projected to increase dramatically during the next 3 decades as the population ages, the numbers of higher-risk minority groups increase, and people with diabetes live longer because of decreasing rates of cardiovascular deaths,” wrote Derek LeRoith, MD, of Icahn School of Medicine at Mount Sinai, New York, and his writing committee colleagues. They said their goal was to provide health care providers with guidance for the management of type 1 or type 2 diabetes in older patients, with a focus on simplifying medication regimens and management strategies to avoid “unnecessary and/or harmful adverse effects.”
The guideline, published in the Journal of Clinical Endocrinology & Metabolism, is based mainly on evidence from controlled trials in two systematic reviews that specifically focused on adults aged 65 years and older. The guideline addresses six areas of consideration for this patient population:
- Role of the endocrinologist and diabetes care specialist.
- Screening for diabetes and prediabetes, and diabetes prevention.
- Assessment of older patients with diabetes.
- Treatment of hyperglycemia.
- Treating complications of diabetes.
- Special settings and populations.
Partnerships and screening
The guideline recommends that primary care providers partner with an endocrinologist or diabetes specialist in the care of patients aged 65 and older with newly diagnosed diabetes, and that the specialist take primary responsibility for diabetes care of patients with type 1 diabetes or those who need more complex intervention to achieve treatment goals.
Screening for diabetes in adults aged 65 years and older using fasting plasma glucose and/or hemoglobin A1c should occur every 2 years, but that schedule should be adjusted based on shared decision making with the patient, the committee said. Providers are advised to assess the patient’s overall health and personal values before settling on treatment goals and strategies. The writing group also recommends periodic cognitive screening and that medication regimens be simplified as much as possible.
Tackling hyperglycemia
For treatment of hyperglycemia, the guideline recommends outpatient strategies to minimize hypoglycemia and periodic or continuous glucose monitoring. The strategies include lifestyle modifications as a first-line intervention for ambulatory patients, as well as nutritional assessment. A high-protein diet is recommended for older patients with frailty, but no restrictions on diet are advised for patients who cannot meet glycemic targets with lifestyle modification and who are at risk for malnutrition.
Metformin is the first-choice recommendation for patients with diabetes aged 65 and older who need medical management in addition to lifestyle modification, but it is not recommended for individuals with impaired kidney function or gastrointestinal intolerance, according to the guideline. Oral and injectable drugs and/or insulin are recommended if metformin and lifestyle changes are insufficient to meet glycemic targets, the writers noted.
Managing complications
Hypertension is among the diabetes-related complications that need to be managed in older adults, and the guideline recommends a target blood pressure of 140/90 mm Hg, but other targets – based on patient-provider shared decision making – may be considered for patients in high-risk groups.
The guideline calls for management of hyperlipidemia with statin therapy and “use of an annual lipid profile to achieve the recommended levels for reducing absolute cardiovascular disease events and all-cause mortality.” The committee does not specify low-density lipoprotein cholesterol targets because of insufficient evidence, but recommends alternative treatments, including ezetimibe or proprotein convertase subtilisin/kexin type 9 inhibitors, if statin therapy is not enough to help the patients meet goals. The writers also advocate fish oil and/or fenofibrate for patients with fasting triglycerides of more than 500 mg/dL.
To manage congestive heart failure in older patients with diabetes, the guideline recommends following standard clinical practice guidelines for the condition, and cautious use of oral hypoglycemic agents, including glinides, rosiglitazone, pioglitazone, and dipeptidyl peptidase–4 inhibitors. The writers noted that low-dose aspirin is recommended for patients with diabetes with a history of atherosclerotic cardiovascular disease.
The committee also recommends an annual comprehensive eye exam for patients with diabetes aged 65 years and older to identify retinal disease and suggests that actions, such as physical therapy and reduced use of sedatives, be taken to minimize the risk of falls in patients with neuropathy or problems with balance and gait.
Older patients with diabetes also should be screened annually for chronic kidney disease, and the dosage of diabetes medications should be adjusted to minimize side effects in patients with kidney problems.
Tailoring care to setting
Finally, the guideline addresses special settings and populations, including managing diabetes in hospitals or nursing homes, or in patients who are transitioning to homes or long-term care facilities. Recommendations in this category include simplifying medications for older adults with terminal illness or severe comorbidities, as well as setting glycemic targets as part of a hospital discharge plan.
“The most important aspect of successful transition is effective, detailed, and thorough bidirectional communication between the discharging and receiving teams of health care providers,” the writers emphasized.
The guideline is cosponsored by the European Society of Endocrinology, the Gerontological Society of America, and the Obesity Society. The chair of the committee had no relevant financial conflicts to disclose, and at least 50% of the committee members were free of relevant conflicts of interest.
SOURCE: LeRoith D et al. J Clin Endocrinol Metab. 2019;104:1520-74.
according to a new guideline on diabetes care for older adults issued by the Endocrine Society.
“The prevalence of diabetes in the United States is projected to increase dramatically during the next 3 decades as the population ages, the numbers of higher-risk minority groups increase, and people with diabetes live longer because of decreasing rates of cardiovascular deaths,” wrote Derek LeRoith, MD, of Icahn School of Medicine at Mount Sinai, New York, and his writing committee colleagues. They said their goal was to provide health care providers with guidance for the management of type 1 or type 2 diabetes in older patients, with a focus on simplifying medication regimens and management strategies to avoid “unnecessary and/or harmful adverse effects.”
The guideline, published in the Journal of Clinical Endocrinology & Metabolism, is based mainly on evidence from controlled trials in two systematic reviews that specifically focused on adults aged 65 years and older. The guideline addresses six areas of consideration for this patient population:
- Role of the endocrinologist and diabetes care specialist.
- Screening for diabetes and prediabetes, and diabetes prevention.
- Assessment of older patients with diabetes.
- Treatment of hyperglycemia.
- Treating complications of diabetes.
- Special settings and populations.
Partnerships and screening
The guideline recommends that primary care providers partner with an endocrinologist or diabetes specialist in the care of patients aged 65 and older with newly diagnosed diabetes, and that the specialist take primary responsibility for diabetes care of patients with type 1 diabetes or those who need more complex intervention to achieve treatment goals.
Screening for diabetes in adults aged 65 years and older using fasting plasma glucose and/or hemoglobin A1c should occur every 2 years, but that schedule should be adjusted based on shared decision making with the patient, the committee said. Providers are advised to assess the patient’s overall health and personal values before settling on treatment goals and strategies. The writing group also recommends periodic cognitive screening and that medication regimens be simplified as much as possible.
Tackling hyperglycemia
For treatment of hyperglycemia, the guideline recommends outpatient strategies to minimize hypoglycemia and periodic or continuous glucose monitoring. The strategies include lifestyle modifications as a first-line intervention for ambulatory patients, as well as nutritional assessment. A high-protein diet is recommended for older patients with frailty, but no restrictions on diet are advised for patients who cannot meet glycemic targets with lifestyle modification and who are at risk for malnutrition.
Metformin is the first-choice recommendation for patients with diabetes aged 65 and older who need medical management in addition to lifestyle modification, but it is not recommended for individuals with impaired kidney function or gastrointestinal intolerance, according to the guideline. Oral and injectable drugs and/or insulin are recommended if metformin and lifestyle changes are insufficient to meet glycemic targets, the writers noted.
Managing complications
Hypertension is among the diabetes-related complications that need to be managed in older adults, and the guideline recommends a target blood pressure of 140/90 mm Hg, but other targets – based on patient-provider shared decision making – may be considered for patients in high-risk groups.
The guideline calls for management of hyperlipidemia with statin therapy and “use of an annual lipid profile to achieve the recommended levels for reducing absolute cardiovascular disease events and all-cause mortality.” The committee does not specify low-density lipoprotein cholesterol targets because of insufficient evidence, but recommends alternative treatments, including ezetimibe or proprotein convertase subtilisin/kexin type 9 inhibitors, if statin therapy is not enough to help the patients meet goals. The writers also advocate fish oil and/or fenofibrate for patients with fasting triglycerides of more than 500 mg/dL.
To manage congestive heart failure in older patients with diabetes, the guideline recommends following standard clinical practice guidelines for the condition, and cautious use of oral hypoglycemic agents, including glinides, rosiglitazone, pioglitazone, and dipeptidyl peptidase–4 inhibitors. The writers noted that low-dose aspirin is recommended for patients with diabetes with a history of atherosclerotic cardiovascular disease.
The committee also recommends an annual comprehensive eye exam for patients with diabetes aged 65 years and older to identify retinal disease and suggests that actions, such as physical therapy and reduced use of sedatives, be taken to minimize the risk of falls in patients with neuropathy or problems with balance and gait.
Older patients with diabetes also should be screened annually for chronic kidney disease, and the dosage of diabetes medications should be adjusted to minimize side effects in patients with kidney problems.
Tailoring care to setting
Finally, the guideline addresses special settings and populations, including managing diabetes in hospitals or nursing homes, or in patients who are transitioning to homes or long-term care facilities. Recommendations in this category include simplifying medications for older adults with terminal illness or severe comorbidities, as well as setting glycemic targets as part of a hospital discharge plan.
“The most important aspect of successful transition is effective, detailed, and thorough bidirectional communication between the discharging and receiving teams of health care providers,” the writers emphasized.
The guideline is cosponsored by the European Society of Endocrinology, the Gerontological Society of America, and the Obesity Society. The chair of the committee had no relevant financial conflicts to disclose, and at least 50% of the committee members were free of relevant conflicts of interest.
SOURCE: LeRoith D et al. J Clin Endocrinol Metab. 2019;104:1520-74.
according to a new guideline on diabetes care for older adults issued by the Endocrine Society.
“The prevalence of diabetes in the United States is projected to increase dramatically during the next 3 decades as the population ages, the numbers of higher-risk minority groups increase, and people with diabetes live longer because of decreasing rates of cardiovascular deaths,” wrote Derek LeRoith, MD, of Icahn School of Medicine at Mount Sinai, New York, and his writing committee colleagues. They said their goal was to provide health care providers with guidance for the management of type 1 or type 2 diabetes in older patients, with a focus on simplifying medication regimens and management strategies to avoid “unnecessary and/or harmful adverse effects.”
The guideline, published in the Journal of Clinical Endocrinology & Metabolism, is based mainly on evidence from controlled trials in two systematic reviews that specifically focused on adults aged 65 years and older. The guideline addresses six areas of consideration for this patient population:
- Role of the endocrinologist and diabetes care specialist.
- Screening for diabetes and prediabetes, and diabetes prevention.
- Assessment of older patients with diabetes.
- Treatment of hyperglycemia.
- Treating complications of diabetes.
- Special settings and populations.
Partnerships and screening
The guideline recommends that primary care providers partner with an endocrinologist or diabetes specialist in the care of patients aged 65 and older with newly diagnosed diabetes, and that the specialist take primary responsibility for diabetes care of patients with type 1 diabetes or those who need more complex intervention to achieve treatment goals.
Screening for diabetes in adults aged 65 years and older using fasting plasma glucose and/or hemoglobin A1c should occur every 2 years, but that schedule should be adjusted based on shared decision making with the patient, the committee said. Providers are advised to assess the patient’s overall health and personal values before settling on treatment goals and strategies. The writing group also recommends periodic cognitive screening and that medication regimens be simplified as much as possible.
Tackling hyperglycemia
For treatment of hyperglycemia, the guideline recommends outpatient strategies to minimize hypoglycemia and periodic or continuous glucose monitoring. The strategies include lifestyle modifications as a first-line intervention for ambulatory patients, as well as nutritional assessment. A high-protein diet is recommended for older patients with frailty, but no restrictions on diet are advised for patients who cannot meet glycemic targets with lifestyle modification and who are at risk for malnutrition.
Metformin is the first-choice recommendation for patients with diabetes aged 65 and older who need medical management in addition to lifestyle modification, but it is not recommended for individuals with impaired kidney function or gastrointestinal intolerance, according to the guideline. Oral and injectable drugs and/or insulin are recommended if metformin and lifestyle changes are insufficient to meet glycemic targets, the writers noted.
Managing complications
Hypertension is among the diabetes-related complications that need to be managed in older adults, and the guideline recommends a target blood pressure of 140/90 mm Hg, but other targets – based on patient-provider shared decision making – may be considered for patients in high-risk groups.
The guideline calls for management of hyperlipidemia with statin therapy and “use of an annual lipid profile to achieve the recommended levels for reducing absolute cardiovascular disease events and all-cause mortality.” The committee does not specify low-density lipoprotein cholesterol targets because of insufficient evidence, but recommends alternative treatments, including ezetimibe or proprotein convertase subtilisin/kexin type 9 inhibitors, if statin therapy is not enough to help the patients meet goals. The writers also advocate fish oil and/or fenofibrate for patients with fasting triglycerides of more than 500 mg/dL.
To manage congestive heart failure in older patients with diabetes, the guideline recommends following standard clinical practice guidelines for the condition, and cautious use of oral hypoglycemic agents, including glinides, rosiglitazone, pioglitazone, and dipeptidyl peptidase–4 inhibitors. The writers noted that low-dose aspirin is recommended for patients with diabetes with a history of atherosclerotic cardiovascular disease.
The committee also recommends an annual comprehensive eye exam for patients with diabetes aged 65 years and older to identify retinal disease and suggests that actions, such as physical therapy and reduced use of sedatives, be taken to minimize the risk of falls in patients with neuropathy or problems with balance and gait.
Older patients with diabetes also should be screened annually for chronic kidney disease, and the dosage of diabetes medications should be adjusted to minimize side effects in patients with kidney problems.
Tailoring care to setting
Finally, the guideline addresses special settings and populations, including managing diabetes in hospitals or nursing homes, or in patients who are transitioning to homes or long-term care facilities. Recommendations in this category include simplifying medications for older adults with terminal illness or severe comorbidities, as well as setting glycemic targets as part of a hospital discharge plan.
“The most important aspect of successful transition is effective, detailed, and thorough bidirectional communication between the discharging and receiving teams of health care providers,” the writers emphasized.
The guideline is cosponsored by the European Society of Endocrinology, the Gerontological Society of America, and the Obesity Society. The chair of the committee had no relevant financial conflicts to disclose, and at least 50% of the committee members were free of relevant conflicts of interest.
SOURCE: LeRoith D et al. J Clin Endocrinol Metab. 2019;104:1520-74.
FROM THE JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM
No increase in UTI risk with SGLT-2 inhibitors
Sodium-glucose cotransporter 2 inhibitors do not appear to increase the risk of urinary tract infections, compared with other antidiabetic medications, new research has found.
In a paper published in Annals of Internal Medicine, researchers reported the outcomes of a population-based, propensity-matched cohort study in which the analyzed data from 235,730 individuals newly prescribed sodium-glucose cotransporter 2 (SGLT-2) inhibitors, dipeptidyl peptidase–4 (DPP-4) inhibitors, and glucagonlike peptide–1 receptor (GLP-1) agonists.
In the first cohort, comparing SGLT-2 inhibitors with DDP-4 inhibitors, there were no significant differences between the two groups in the incidence rate of severe urinary tract infections (UTIs) (adjusted hazard ratio, 0.98; 95% confidence interval, 0.68-1.41).
In the second cohort, which compared patients taking GLP-1 receptor agonists with those taking SGLT-2 inhibitors, researchers saw a slightly lower incidence of severe UTIs among individuals on SGLT-2 inhibitors (HR, 0.72; 95% CI, 0.53-0.99; P = .04).
The analysis also failed to find any evidence that SGLT-2 inhibitors were associated with an increase in the risk of hospitalization or outpatient treatment for a UTI.
Chintan V. Dave, PharmD – now at Rutgers University, New Brunswick, N.J. – and colleagues from Brigham and Women’s Hospital and Harvard Medical School, Boston, wrote that the findings have significant clinical implications.
“Patients who may be good candidates to receive SGLT-2 inhibitors for diabetes control but who have a history of recurrent UTIs may be precluded from being prescribed these agents; because UTIs are highly prevalent in patients with diabetes, this could exclude a substantial number of patients from receiving an entire class of medications that has been shown to decrease risk for major cardiovascular events and death,” they wrote.
The researchers stressed that “other factors beyond risk for UTI events should be considered in decisions about whether to prescribe SGLT-2 therapy for patients with diabetes.”
The authors did note that their study was subject to the usual limitations of observational studies, such as susceptibility to confounding, and was also limited to people with health insurance.
An accompanying editorial by Kristian B. Filion, PhD, and Oriana H. Yu, MD, of McGill University, Montreal, commented that the data provided reassuring, real-world evidence on the potential safety issue of UTI risk with SGLT-2 inhibitors.
However, they also stressed that the study excluded individuals at high risk or with a history of UTIs, and that these were subgroups who required further investigation.
The study was supported by the Harvard Medical School and the National Institute on Aging. Three authors reported support from private industry outside the submitted work.
SOURCE: Dave CV et al. Ann Intern Med. 2019 Jul 29. doi: 10.7326/M18-3136.
Sodium-glucose cotransporter 2 inhibitors do not appear to increase the risk of urinary tract infections, compared with other antidiabetic medications, new research has found.
In a paper published in Annals of Internal Medicine, researchers reported the outcomes of a population-based, propensity-matched cohort study in which the analyzed data from 235,730 individuals newly prescribed sodium-glucose cotransporter 2 (SGLT-2) inhibitors, dipeptidyl peptidase–4 (DPP-4) inhibitors, and glucagonlike peptide–1 receptor (GLP-1) agonists.
In the first cohort, comparing SGLT-2 inhibitors with DDP-4 inhibitors, there were no significant differences between the two groups in the incidence rate of severe urinary tract infections (UTIs) (adjusted hazard ratio, 0.98; 95% confidence interval, 0.68-1.41).
In the second cohort, which compared patients taking GLP-1 receptor agonists with those taking SGLT-2 inhibitors, researchers saw a slightly lower incidence of severe UTIs among individuals on SGLT-2 inhibitors (HR, 0.72; 95% CI, 0.53-0.99; P = .04).
The analysis also failed to find any evidence that SGLT-2 inhibitors were associated with an increase in the risk of hospitalization or outpatient treatment for a UTI.
Chintan V. Dave, PharmD – now at Rutgers University, New Brunswick, N.J. – and colleagues from Brigham and Women’s Hospital and Harvard Medical School, Boston, wrote that the findings have significant clinical implications.
“Patients who may be good candidates to receive SGLT-2 inhibitors for diabetes control but who have a history of recurrent UTIs may be precluded from being prescribed these agents; because UTIs are highly prevalent in patients with diabetes, this could exclude a substantial number of patients from receiving an entire class of medications that has been shown to decrease risk for major cardiovascular events and death,” they wrote.
The researchers stressed that “other factors beyond risk for UTI events should be considered in decisions about whether to prescribe SGLT-2 therapy for patients with diabetes.”
The authors did note that their study was subject to the usual limitations of observational studies, such as susceptibility to confounding, and was also limited to people with health insurance.
An accompanying editorial by Kristian B. Filion, PhD, and Oriana H. Yu, MD, of McGill University, Montreal, commented that the data provided reassuring, real-world evidence on the potential safety issue of UTI risk with SGLT-2 inhibitors.
However, they also stressed that the study excluded individuals at high risk or with a history of UTIs, and that these were subgroups who required further investigation.
The study was supported by the Harvard Medical School and the National Institute on Aging. Three authors reported support from private industry outside the submitted work.
SOURCE: Dave CV et al. Ann Intern Med. 2019 Jul 29. doi: 10.7326/M18-3136.
Sodium-glucose cotransporter 2 inhibitors do not appear to increase the risk of urinary tract infections, compared with other antidiabetic medications, new research has found.
In a paper published in Annals of Internal Medicine, researchers reported the outcomes of a population-based, propensity-matched cohort study in which the analyzed data from 235,730 individuals newly prescribed sodium-glucose cotransporter 2 (SGLT-2) inhibitors, dipeptidyl peptidase–4 (DPP-4) inhibitors, and glucagonlike peptide–1 receptor (GLP-1) agonists.
In the first cohort, comparing SGLT-2 inhibitors with DDP-4 inhibitors, there were no significant differences between the two groups in the incidence rate of severe urinary tract infections (UTIs) (adjusted hazard ratio, 0.98; 95% confidence interval, 0.68-1.41).
In the second cohort, which compared patients taking GLP-1 receptor agonists with those taking SGLT-2 inhibitors, researchers saw a slightly lower incidence of severe UTIs among individuals on SGLT-2 inhibitors (HR, 0.72; 95% CI, 0.53-0.99; P = .04).
The analysis also failed to find any evidence that SGLT-2 inhibitors were associated with an increase in the risk of hospitalization or outpatient treatment for a UTI.
Chintan V. Dave, PharmD – now at Rutgers University, New Brunswick, N.J. – and colleagues from Brigham and Women’s Hospital and Harvard Medical School, Boston, wrote that the findings have significant clinical implications.
“Patients who may be good candidates to receive SGLT-2 inhibitors for diabetes control but who have a history of recurrent UTIs may be precluded from being prescribed these agents; because UTIs are highly prevalent in patients with diabetes, this could exclude a substantial number of patients from receiving an entire class of medications that has been shown to decrease risk for major cardiovascular events and death,” they wrote.
The researchers stressed that “other factors beyond risk for UTI events should be considered in decisions about whether to prescribe SGLT-2 therapy for patients with diabetes.”
The authors did note that their study was subject to the usual limitations of observational studies, such as susceptibility to confounding, and was also limited to people with health insurance.
An accompanying editorial by Kristian B. Filion, PhD, and Oriana H. Yu, MD, of McGill University, Montreal, commented that the data provided reassuring, real-world evidence on the potential safety issue of UTI risk with SGLT-2 inhibitors.
However, they also stressed that the study excluded individuals at high risk or with a history of UTIs, and that these were subgroups who required further investigation.
The study was supported by the Harvard Medical School and the National Institute on Aging. Three authors reported support from private industry outside the submitted work.
SOURCE: Dave CV et al. Ann Intern Med. 2019 Jul 29. doi: 10.7326/M18-3136.
FROM ANNALS OF INTERNAL MEDICINE
Key clinical point: Sodium-glucose cotransporter 2 inhibitors are not associated with a greater risk of urinary tract infections, compared with dipeptidyl peptidase-4 inhibitors or glucagonlike peptide–1 receptor agonists.
Major finding: The incidence of severe urinary tract infections is similar among patients taking sodium-glucose cotransporter 2 inhibitors, dipeptidyl peptidase–4, inhibitors or glucagonlike peptide–1 receptor agonists.
Study details: A population-based, propensity-matched cohort study in 235,730 individuals with type 2 diabetes.
Disclosures: The study was supported by the Harvard Medical School and the National Institute on Aging. Three authors reported support from private industry outside the submitted work.
Source: Dave CV et al. Ann Intern Med. 2019 Jul 29. doi: 10.7326/M18-3136.
Polypharmacy in the Elderly
FDA approves Baqsimi nasal powder for emergency hypoglycemia treatment
in patients aged 4 years and older.
Injectable glucagon has been approved in the United States for several decades.
The safety and efficacy of the Baqsimi powder was assessed in two studies with adults with diabetes and one with pediatric patients. In all three studies, a single dose of Baqsimi was compared with a single dose of glucagon injection, and Baqsimi adequately raised blood sugar levels in response to insulin-induced hypoglycemia.
The most common adverse events associated with Baqsimi include nausea, vomiting, headache, upper respiratory tract irritation, watery eyes, redness of eyes, and itchiness. The safety profile is similar to that of injectable glucagon, with the addition of nasal- and eye-related symptoms because of the method of delivery.
“There are many products on the market for those who need insulin, but until now, people suffering from a severe hypoglycemic episode had to be treated with a glucagon injection that first had to be mixed in a several-step process. This new way to administer glucagon may simplify the process, which can be critical during an episode, especially since the patient may have lost consciousness or may be having a seizure. In those situations, we want the process to treat the suffering person to be as simple as possible,” Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research, said in the press release.
Find the full press release on the FDA website.
in patients aged 4 years and older.
Injectable glucagon has been approved in the United States for several decades.
The safety and efficacy of the Baqsimi powder was assessed in two studies with adults with diabetes and one with pediatric patients. In all three studies, a single dose of Baqsimi was compared with a single dose of glucagon injection, and Baqsimi adequately raised blood sugar levels in response to insulin-induced hypoglycemia.
The most common adverse events associated with Baqsimi include nausea, vomiting, headache, upper respiratory tract irritation, watery eyes, redness of eyes, and itchiness. The safety profile is similar to that of injectable glucagon, with the addition of nasal- and eye-related symptoms because of the method of delivery.
“There are many products on the market for those who need insulin, but until now, people suffering from a severe hypoglycemic episode had to be treated with a glucagon injection that first had to be mixed in a several-step process. This new way to administer glucagon may simplify the process, which can be critical during an episode, especially since the patient may have lost consciousness or may be having a seizure. In those situations, we want the process to treat the suffering person to be as simple as possible,” Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research, said in the press release.
Find the full press release on the FDA website.
in patients aged 4 years and older.
Injectable glucagon has been approved in the United States for several decades.
The safety and efficacy of the Baqsimi powder was assessed in two studies with adults with diabetes and one with pediatric patients. In all three studies, a single dose of Baqsimi was compared with a single dose of glucagon injection, and Baqsimi adequately raised blood sugar levels in response to insulin-induced hypoglycemia.
The most common adverse events associated with Baqsimi include nausea, vomiting, headache, upper respiratory tract irritation, watery eyes, redness of eyes, and itchiness. The safety profile is similar to that of injectable glucagon, with the addition of nasal- and eye-related symptoms because of the method of delivery.
“There are many products on the market for those who need insulin, but until now, people suffering from a severe hypoglycemic episode had to be treated with a glucagon injection that first had to be mixed in a several-step process. This new way to administer glucagon may simplify the process, which can be critical during an episode, especially since the patient may have lost consciousness or may be having a seizure. In those situations, we want the process to treat the suffering person to be as simple as possible,” Janet Woodcock, MD, director of the FDA’s Center for Drug Evaluation and Research, said in the press release.
Find the full press release on the FDA website.
Inadequate glycemic control in type 1 diabetes leads to increased fracture risk
A single percentage increase in the level of hemoglobin A1c (HbA1c) in patients with newly diagnosed type 1 diabetes is significantly associated with an increase in fracture risk, according to findings in a study published in Diabetic Medicine.
To determine the effect of glycemic control on fracture risk, Rasiah Thayakaran, PhD, of the University of Birmingham (England) and colleagues analyzed data from 5,368 patients with newly diagnosed type 1 diabetes in the United Kingdom. HbA1c measurements were collected until either fracture or the end of the study, and were then converted from percentages to mmol/mol. Patient age ranged between 1 and 60 years, and the mean age was 22 years.
During 37,830 person‐years of follow‐up, 525 fractures were observed, with an incidence rate of 14 per 1,000 person‐years. The rate among men was 15 per 1,000 person‐years, compared with 12 per 1,000 person‐years among women. There was a significant association between hemoglobin level and risk of fractures (adjusted hazard ratio, 1.007 mmol/mol; 95% confidence interval, 1.002-1.011 mmol/mol), representing an increase of 7% in risk for fracture for each percentage increase in hemoglobin level.
“When assessing an individual with newly diagnosed type 1 diabetes and high HbA1c, increased clinical awareness about the fracture risk may be incorporated in decision‐making regarding the clinical management and even in prompting early antiosteoporotic intervention,” Dr. Thayakaran and coauthors wrote.
The researchers acknowledged the study’s limitations, including a possibility of residual confounding because of their use of observational data. In addition, they could not confirm whether the increase in fracture risk should be attributed to bone fragility or to increased risk of falls. Finally, though they noted using a comprehensive list of codes to identify fractures, they could not verify “completeness of recording ... and therefore reported overall fracture incidence should be interpreted with caution.”
The study was not funded. The authors reported no conflicts of interest.
SOURCE: Thayakaran R et al. Diab Med. 2019 Mar 8. doi: 10.1111/dme.13945.
A single percentage increase in the level of hemoglobin A1c (HbA1c) in patients with newly diagnosed type 1 diabetes is significantly associated with an increase in fracture risk, according to findings in a study published in Diabetic Medicine.
To determine the effect of glycemic control on fracture risk, Rasiah Thayakaran, PhD, of the University of Birmingham (England) and colleagues analyzed data from 5,368 patients with newly diagnosed type 1 diabetes in the United Kingdom. HbA1c measurements were collected until either fracture or the end of the study, and were then converted from percentages to mmol/mol. Patient age ranged between 1 and 60 years, and the mean age was 22 years.
During 37,830 person‐years of follow‐up, 525 fractures were observed, with an incidence rate of 14 per 1,000 person‐years. The rate among men was 15 per 1,000 person‐years, compared with 12 per 1,000 person‐years among women. There was a significant association between hemoglobin level and risk of fractures (adjusted hazard ratio, 1.007 mmol/mol; 95% confidence interval, 1.002-1.011 mmol/mol), representing an increase of 7% in risk for fracture for each percentage increase in hemoglobin level.
“When assessing an individual with newly diagnosed type 1 diabetes and high HbA1c, increased clinical awareness about the fracture risk may be incorporated in decision‐making regarding the clinical management and even in prompting early antiosteoporotic intervention,” Dr. Thayakaran and coauthors wrote.
The researchers acknowledged the study’s limitations, including a possibility of residual confounding because of their use of observational data. In addition, they could not confirm whether the increase in fracture risk should be attributed to bone fragility or to increased risk of falls. Finally, though they noted using a comprehensive list of codes to identify fractures, they could not verify “completeness of recording ... and therefore reported overall fracture incidence should be interpreted with caution.”
The study was not funded. The authors reported no conflicts of interest.
SOURCE: Thayakaran R et al. Diab Med. 2019 Mar 8. doi: 10.1111/dme.13945.
A single percentage increase in the level of hemoglobin A1c (HbA1c) in patients with newly diagnosed type 1 diabetes is significantly associated with an increase in fracture risk, according to findings in a study published in Diabetic Medicine.
To determine the effect of glycemic control on fracture risk, Rasiah Thayakaran, PhD, of the University of Birmingham (England) and colleagues analyzed data from 5,368 patients with newly diagnosed type 1 diabetes in the United Kingdom. HbA1c measurements were collected until either fracture or the end of the study, and were then converted from percentages to mmol/mol. Patient age ranged between 1 and 60 years, and the mean age was 22 years.
During 37,830 person‐years of follow‐up, 525 fractures were observed, with an incidence rate of 14 per 1,000 person‐years. The rate among men was 15 per 1,000 person‐years, compared with 12 per 1,000 person‐years among women. There was a significant association between hemoglobin level and risk of fractures (adjusted hazard ratio, 1.007 mmol/mol; 95% confidence interval, 1.002-1.011 mmol/mol), representing an increase of 7% in risk for fracture for each percentage increase in hemoglobin level.
“When assessing an individual with newly diagnosed type 1 diabetes and high HbA1c, increased clinical awareness about the fracture risk may be incorporated in decision‐making regarding the clinical management and even in prompting early antiosteoporotic intervention,” Dr. Thayakaran and coauthors wrote.
The researchers acknowledged the study’s limitations, including a possibility of residual confounding because of their use of observational data. In addition, they could not confirm whether the increase in fracture risk should be attributed to bone fragility or to increased risk of falls. Finally, though they noted using a comprehensive list of codes to identify fractures, they could not verify “completeness of recording ... and therefore reported overall fracture incidence should be interpreted with caution.”
The study was not funded. The authors reported no conflicts of interest.
SOURCE: Thayakaran R et al. Diab Med. 2019 Mar 8. doi: 10.1111/dme.13945.
FROM DIABETIC MEDICINE
DECLARE-TIMI58 shows improved kidney function with dapagliflozin
SAN FRANCISCO – and it showed reductions in the relative risk of renal-specific and cardiorenal outcomes of 47% and 24%, respectively, over 4 years, according to an analysis of the DECLARE-TIMI58 trial presented at the annual scientific sessions of the American Diabetes Association.
The findings add to a message physicians are hearing more and more frequently – that SGLT2 inhibitors, which also include empagliflozin (Jardiance) and canagliflozin (Invokana), could be useful for the early prevention of chronic kidney disease in patients who have type 2 diabetes. The main difference between the DECLARE-TIMI58 and previous study data was that most of its population did not have chronic kidney disease.
The drug should be given “at a very early stage of disease – it can reverse the disease and its complications [and] change the outcomes of patients,” said investigator Itamar Raz, MD, of Hebrew University, Jerusalem, in presenting the findings, which were published simultaneously in the Lancet Diabetes & Endocrinology (2019 Jun 10. doi: 10.1016/S2213-8587[19]30180-9).
What’s new in this renal analysis of DECLARE-TIMI58 is that patients were relatively healthy – 60% of them were without established cardiovascular disease and had much better renal function at baseline, compared with patients in other studies. About half of the patients started out with an estimated glomerular filtration rate (eGFR) more than 90 mL/min per 1.73 m2, which means that they had normal kidney function, and most of the rest of the patients had normal to near-normal renal function or mild renal failure. Findings from previous trials with SGLT2 inhibitors that showed renal protection generally included patients with established cardiovascular disease who started out with greater kidney impairment.
Previous findings have also demonstrated cardioprotective effects with SGLT2 inhibitors in patients with type 2 diabetes. For instance, in earlier results from the DECLARE-TIMI58 trial, dapagliflozin reduced the frequency of cardiovascular death or hospitalization for heart failure, compared with placebo (4.9% vs. 5.8%, respectively; New Engl J Med. 2019;380:347-57), although it did not significantly reduce the frequency of stroke, heart attack, or all-cause death, unlike the results from glucagonlike peptide–1 receptor agonists in much sicker patients (Circulation. 2019;139[17]:2022-31).
Taken as a whole, Dr. Raz said that findings for SGLT2 inhibitors, which, like the glucagonlike peptide–1 receptor agonists, are indicated as second-line therapy in type 2 diabetes after metformin, suggest that they should be used sooner in type 2 disease, perhaps in patients with a hemoglobin A1c level as low at 6.5%.
The absolute benefits of dapagliflozin, compared with placebo, notwithstanding, there are safety concerns with SGLT2s, including genitourinary infections, acute kidney injury, Fournier gangrene, diabetic ketoacidosis, bone fractures, and leg amputations (the latter two only with canagliflozin), many of which have been subject to warnings from the Food and Drug Administration.
Safety outcomes for dapagliflozin, compared with placebo, were not tabulated in the new renal report, but the authors noted a previously reported decrease of 30% in risk for acute kidney injury and major hypoglycemia with dapagliflozin over placebo. There was 1 case of Fournier gangrene with the drug versus 5 with placebo; 27 cases of diabetic ketoacidosis versus 12, respectively; 123 amputations versus 113; and 76 genital infections versus 9.
Matthew Riddle, MD, of Oregon Health and Science University, Portland, said SGLT2 studies “have shown important short-term benefit, but we have no information on long-term safety. These drugs are not physiologic; they do something powerful, but it’s nothing you see in nature. We don’t really know what they do yet; the physiology is still being worked out.”
DECLARE-TIMI58 randomized 8,582 patients with type 2 diabetes to receive dapagliflozin 10 mg orally daily and 8,578 patients to receive placebo. The patients remained on routine diabetes and cardiovascular care. Inclusion criteria included either established atherosclerotic cardiovascular disease (41% of patients) or cardiovascular risk factors (almost 60%), and creatinine clearance of at least 60 mL/min. Median follow-up was 4.2 years.
Overall, 4.2% of patients receiving dapagliflozin and 5.3% of those receiving placebo, met a prespecified secondary cardiorenal composite outcome of end-stage renal disease; death from renal or cardiovascular causes; or a decline of at least 40% in eGFR to less than 60 mL/min per 1.73 m2 according to two tests at least 4 weeks apart (hazard ratio, 0.76; P less than .0001).
Similarly, 1.5% of dapagliflozin patients and 2.6% of those on placebo met a prespecified renal-specific composite of those factors minus death from cardiovascular causes (HR, 0.53; P less than .0001).
Among placebo-treated patients, 2.5% had a sustained decline in eGFR of at least 40% to less than 60 mL/min per 1.73 m2, compared with 1.4% in the dapagliflozin group (HR, 0.54; P less than .0001). There were 11 cases of end-stage renal disease or renal death with dapagliflozin (0.1%), compared with 27 (0.3%) with placebo (HR, 0.41; P = .012).
Among patients who entered the trial with significant renal impairment – an eGFR of less than 60 mL/min per 1.73 m2 – the difference in further renal decline with dapagliflozin was not statistically significant against placebo because of the small number of patients, but Dr. Raz said the drug should still be used earlier in type 2 disease.
Dapagliflozin patients fared worse on renal measurements at 6 months, but caught up by year 2, and surpassed placebo at years 3 and 4, the authors wrote in the latest report.
Just more than 60% of participants were men; patients were in their 60s, on average, and overweight. About 80% of the patients were white.
AstraZeneca, which makes dapagliflozin, was involved with study design, data collection, data analysis, interpretation, and writing of the report. Four authors were employees of the company, and all but two of the 17 others, including Dr. Raz, disclosed personal payments from the company and/or research funding. Dr. Riddle disclosed receiving research funding from AstraZeneca.
SAN FRANCISCO – and it showed reductions in the relative risk of renal-specific and cardiorenal outcomes of 47% and 24%, respectively, over 4 years, according to an analysis of the DECLARE-TIMI58 trial presented at the annual scientific sessions of the American Diabetes Association.
The findings add to a message physicians are hearing more and more frequently – that SGLT2 inhibitors, which also include empagliflozin (Jardiance) and canagliflozin (Invokana), could be useful for the early prevention of chronic kidney disease in patients who have type 2 diabetes. The main difference between the DECLARE-TIMI58 and previous study data was that most of its population did not have chronic kidney disease.
The drug should be given “at a very early stage of disease – it can reverse the disease and its complications [and] change the outcomes of patients,” said investigator Itamar Raz, MD, of Hebrew University, Jerusalem, in presenting the findings, which were published simultaneously in the Lancet Diabetes & Endocrinology (2019 Jun 10. doi: 10.1016/S2213-8587[19]30180-9).
What’s new in this renal analysis of DECLARE-TIMI58 is that patients were relatively healthy – 60% of them were without established cardiovascular disease and had much better renal function at baseline, compared with patients in other studies. About half of the patients started out with an estimated glomerular filtration rate (eGFR) more than 90 mL/min per 1.73 m2, which means that they had normal kidney function, and most of the rest of the patients had normal to near-normal renal function or mild renal failure. Findings from previous trials with SGLT2 inhibitors that showed renal protection generally included patients with established cardiovascular disease who started out with greater kidney impairment.
Previous findings have also demonstrated cardioprotective effects with SGLT2 inhibitors in patients with type 2 diabetes. For instance, in earlier results from the DECLARE-TIMI58 trial, dapagliflozin reduced the frequency of cardiovascular death or hospitalization for heart failure, compared with placebo (4.9% vs. 5.8%, respectively; New Engl J Med. 2019;380:347-57), although it did not significantly reduce the frequency of stroke, heart attack, or all-cause death, unlike the results from glucagonlike peptide–1 receptor agonists in much sicker patients (Circulation. 2019;139[17]:2022-31).
Taken as a whole, Dr. Raz said that findings for SGLT2 inhibitors, which, like the glucagonlike peptide–1 receptor agonists, are indicated as second-line therapy in type 2 diabetes after metformin, suggest that they should be used sooner in type 2 disease, perhaps in patients with a hemoglobin A1c level as low at 6.5%.
The absolute benefits of dapagliflozin, compared with placebo, notwithstanding, there are safety concerns with SGLT2s, including genitourinary infections, acute kidney injury, Fournier gangrene, diabetic ketoacidosis, bone fractures, and leg amputations (the latter two only with canagliflozin), many of which have been subject to warnings from the Food and Drug Administration.
Safety outcomes for dapagliflozin, compared with placebo, were not tabulated in the new renal report, but the authors noted a previously reported decrease of 30% in risk for acute kidney injury and major hypoglycemia with dapagliflozin over placebo. There was 1 case of Fournier gangrene with the drug versus 5 with placebo; 27 cases of diabetic ketoacidosis versus 12, respectively; 123 amputations versus 113; and 76 genital infections versus 9.
Matthew Riddle, MD, of Oregon Health and Science University, Portland, said SGLT2 studies “have shown important short-term benefit, but we have no information on long-term safety. These drugs are not physiologic; they do something powerful, but it’s nothing you see in nature. We don’t really know what they do yet; the physiology is still being worked out.”
DECLARE-TIMI58 randomized 8,582 patients with type 2 diabetes to receive dapagliflozin 10 mg orally daily and 8,578 patients to receive placebo. The patients remained on routine diabetes and cardiovascular care. Inclusion criteria included either established atherosclerotic cardiovascular disease (41% of patients) or cardiovascular risk factors (almost 60%), and creatinine clearance of at least 60 mL/min. Median follow-up was 4.2 years.
Overall, 4.2% of patients receiving dapagliflozin and 5.3% of those receiving placebo, met a prespecified secondary cardiorenal composite outcome of end-stage renal disease; death from renal or cardiovascular causes; or a decline of at least 40% in eGFR to less than 60 mL/min per 1.73 m2 according to two tests at least 4 weeks apart (hazard ratio, 0.76; P less than .0001).
Similarly, 1.5% of dapagliflozin patients and 2.6% of those on placebo met a prespecified renal-specific composite of those factors minus death from cardiovascular causes (HR, 0.53; P less than .0001).
Among placebo-treated patients, 2.5% had a sustained decline in eGFR of at least 40% to less than 60 mL/min per 1.73 m2, compared with 1.4% in the dapagliflozin group (HR, 0.54; P less than .0001). There were 11 cases of end-stage renal disease or renal death with dapagliflozin (0.1%), compared with 27 (0.3%) with placebo (HR, 0.41; P = .012).
Among patients who entered the trial with significant renal impairment – an eGFR of less than 60 mL/min per 1.73 m2 – the difference in further renal decline with dapagliflozin was not statistically significant against placebo because of the small number of patients, but Dr. Raz said the drug should still be used earlier in type 2 disease.
Dapagliflozin patients fared worse on renal measurements at 6 months, but caught up by year 2, and surpassed placebo at years 3 and 4, the authors wrote in the latest report.
Just more than 60% of participants were men; patients were in their 60s, on average, and overweight. About 80% of the patients were white.
AstraZeneca, which makes dapagliflozin, was involved with study design, data collection, data analysis, interpretation, and writing of the report. Four authors were employees of the company, and all but two of the 17 others, including Dr. Raz, disclosed personal payments from the company and/or research funding. Dr. Riddle disclosed receiving research funding from AstraZeneca.
SAN FRANCISCO – and it showed reductions in the relative risk of renal-specific and cardiorenal outcomes of 47% and 24%, respectively, over 4 years, according to an analysis of the DECLARE-TIMI58 trial presented at the annual scientific sessions of the American Diabetes Association.
The findings add to a message physicians are hearing more and more frequently – that SGLT2 inhibitors, which also include empagliflozin (Jardiance) and canagliflozin (Invokana), could be useful for the early prevention of chronic kidney disease in patients who have type 2 diabetes. The main difference between the DECLARE-TIMI58 and previous study data was that most of its population did not have chronic kidney disease.
The drug should be given “at a very early stage of disease – it can reverse the disease and its complications [and] change the outcomes of patients,” said investigator Itamar Raz, MD, of Hebrew University, Jerusalem, in presenting the findings, which were published simultaneously in the Lancet Diabetes & Endocrinology (2019 Jun 10. doi: 10.1016/S2213-8587[19]30180-9).
What’s new in this renal analysis of DECLARE-TIMI58 is that patients were relatively healthy – 60% of them were without established cardiovascular disease and had much better renal function at baseline, compared with patients in other studies. About half of the patients started out with an estimated glomerular filtration rate (eGFR) more than 90 mL/min per 1.73 m2, which means that they had normal kidney function, and most of the rest of the patients had normal to near-normal renal function or mild renal failure. Findings from previous trials with SGLT2 inhibitors that showed renal protection generally included patients with established cardiovascular disease who started out with greater kidney impairment.
Previous findings have also demonstrated cardioprotective effects with SGLT2 inhibitors in patients with type 2 diabetes. For instance, in earlier results from the DECLARE-TIMI58 trial, dapagliflozin reduced the frequency of cardiovascular death or hospitalization for heart failure, compared with placebo (4.9% vs. 5.8%, respectively; New Engl J Med. 2019;380:347-57), although it did not significantly reduce the frequency of stroke, heart attack, or all-cause death, unlike the results from glucagonlike peptide–1 receptor agonists in much sicker patients (Circulation. 2019;139[17]:2022-31).
Taken as a whole, Dr. Raz said that findings for SGLT2 inhibitors, which, like the glucagonlike peptide–1 receptor agonists, are indicated as second-line therapy in type 2 diabetes after metformin, suggest that they should be used sooner in type 2 disease, perhaps in patients with a hemoglobin A1c level as low at 6.5%.
The absolute benefits of dapagliflozin, compared with placebo, notwithstanding, there are safety concerns with SGLT2s, including genitourinary infections, acute kidney injury, Fournier gangrene, diabetic ketoacidosis, bone fractures, and leg amputations (the latter two only with canagliflozin), many of which have been subject to warnings from the Food and Drug Administration.
Safety outcomes for dapagliflozin, compared with placebo, were not tabulated in the new renal report, but the authors noted a previously reported decrease of 30% in risk for acute kidney injury and major hypoglycemia with dapagliflozin over placebo. There was 1 case of Fournier gangrene with the drug versus 5 with placebo; 27 cases of diabetic ketoacidosis versus 12, respectively; 123 amputations versus 113; and 76 genital infections versus 9.
Matthew Riddle, MD, of Oregon Health and Science University, Portland, said SGLT2 studies “have shown important short-term benefit, but we have no information on long-term safety. These drugs are not physiologic; they do something powerful, but it’s nothing you see in nature. We don’t really know what they do yet; the physiology is still being worked out.”
DECLARE-TIMI58 randomized 8,582 patients with type 2 diabetes to receive dapagliflozin 10 mg orally daily and 8,578 patients to receive placebo. The patients remained on routine diabetes and cardiovascular care. Inclusion criteria included either established atherosclerotic cardiovascular disease (41% of patients) or cardiovascular risk factors (almost 60%), and creatinine clearance of at least 60 mL/min. Median follow-up was 4.2 years.
Overall, 4.2% of patients receiving dapagliflozin and 5.3% of those receiving placebo, met a prespecified secondary cardiorenal composite outcome of end-stage renal disease; death from renal or cardiovascular causes; or a decline of at least 40% in eGFR to less than 60 mL/min per 1.73 m2 according to two tests at least 4 weeks apart (hazard ratio, 0.76; P less than .0001).
Similarly, 1.5% of dapagliflozin patients and 2.6% of those on placebo met a prespecified renal-specific composite of those factors minus death from cardiovascular causes (HR, 0.53; P less than .0001).
Among placebo-treated patients, 2.5% had a sustained decline in eGFR of at least 40% to less than 60 mL/min per 1.73 m2, compared with 1.4% in the dapagliflozin group (HR, 0.54; P less than .0001). There were 11 cases of end-stage renal disease or renal death with dapagliflozin (0.1%), compared with 27 (0.3%) with placebo (HR, 0.41; P = .012).
Among patients who entered the trial with significant renal impairment – an eGFR of less than 60 mL/min per 1.73 m2 – the difference in further renal decline with dapagliflozin was not statistically significant against placebo because of the small number of patients, but Dr. Raz said the drug should still be used earlier in type 2 disease.
Dapagliflozin patients fared worse on renal measurements at 6 months, but caught up by year 2, and surpassed placebo at years 3 and 4, the authors wrote in the latest report.
Just more than 60% of participants were men; patients were in their 60s, on average, and overweight. About 80% of the patients were white.
AstraZeneca, which makes dapagliflozin, was involved with study design, data collection, data analysis, interpretation, and writing of the report. Four authors were employees of the company, and all but two of the 17 others, including Dr. Raz, disclosed personal payments from the company and/or research funding. Dr. Riddle disclosed receiving research funding from AstraZeneca.
REPORTING FROM ADA 2019
Key clinical point: The sodium-glucose transporter 2 inhibitor dapagliflozin slowed progression of kidney disease in patients with type 2 diabetes.
Major finding: Overall, 4.2% of dapagliflozin and 5.3% of placebo patients met a prespecified secondary cardiorenal composite outcome of end-stage renal disease, death from renal or cardiovascular causes, or a decline of at least 40% in estimated glomerular filtration rate to less than 60 mL/min per 1.73m2 (HR, 0.76; P less than .0001).
Study details: Placebo-controlled trial in more than 17,000 patients with type 2 diabetes
Disclosures: AstraZeneca, the maker of dapagliflozin, funded and conducted the study. Four authors were employees of the company, and all but two of the remaining 17, including Dr. Raz, disclosed personal payments from the company and/or research funding. Dr. Riddle reported receiving research funding from AstraZeneca.
Source: Mosenzon O et al. Lancet Diabetes Endocrinol. 2019 Jun 10. doi: 10.1016/S2213-8587(19)30180-9.
Plant-based foods could keep type 2 diabetes at bay
according to a new systematic review and meta-analysis of nine observational studies.
The findings aren’t conclusive, but the study authors wrote in JAMA Internal Medicine that they suggest that “plant-based dietary patterns were associated with lower risk of type 2 diabetes, even after adjustment for [body mass index].”
According to 2015 figures provided by the American Diabetes Association, about 29 million people in the United States have type 2 diabetes. Overall, diabetes contributes to hundreds of thousands of deaths each year, which makes it the seventh-leading cause of death in the nation.
For the new analysis, researchers led by Frank Qian, MPH, of Harvard T.H. Chan School of Public Health included nine studies that examined diet and type 2 diabetes. In all, the studies included 307,099 participants, and there were 23,544 cases of incident type 2 diabetes. They were conducted in five countries, including the United States, and tracked participants for 2-28 years; the studies were all published within the last 11 years. The mean ages of participants ranged from 36-65 years.
The meta-analysis linked higher consumption of plant-based foods to a 23% reduced risk of type 2 diabetes, compared with lower consumption (relative risk, 0.77; 95% confidence interval, 0.71-0.84; P = .07 for heterogeneity). The risk dipped even further (down to 30%) when the researchers analyzed four studies that focused on healthy plant-based foods, such as fruits and vegetables, instead of foods such as refined grains, starches, and sugars (RR, 0.70; 95% CI, 0.62-0.79).
The researchers suggested that plant-based diets may lower the risk of diabetes type 2 by limiting weight gain. They also noted various limitations to their analysis, such as the reliance on self-reports and the observational nature of the studies.
Still, “in general populations that do not practice strict vegetarian or vegan diets, replacing animal products with healthful plant-based foods is likely to exert a significant reduction in the risk of diabetes,” the authors wrote.
The study was funded by the National Institutes of Health, and one author received support from the National Institute of Diabetes and Digestive and Kidney Diseases. The remaining authors reported no conflicts of interest withing the scope of this study.
SOURCE: Qian F et al. JAMA Internal Medicine. 2019 Jul 22. doi: 10.1001/jamainternmed.2019.2195.
according to a new systematic review and meta-analysis of nine observational studies.
The findings aren’t conclusive, but the study authors wrote in JAMA Internal Medicine that they suggest that “plant-based dietary patterns were associated with lower risk of type 2 diabetes, even after adjustment for [body mass index].”
According to 2015 figures provided by the American Diabetes Association, about 29 million people in the United States have type 2 diabetes. Overall, diabetes contributes to hundreds of thousands of deaths each year, which makes it the seventh-leading cause of death in the nation.
For the new analysis, researchers led by Frank Qian, MPH, of Harvard T.H. Chan School of Public Health included nine studies that examined diet and type 2 diabetes. In all, the studies included 307,099 participants, and there were 23,544 cases of incident type 2 diabetes. They were conducted in five countries, including the United States, and tracked participants for 2-28 years; the studies were all published within the last 11 years. The mean ages of participants ranged from 36-65 years.
The meta-analysis linked higher consumption of plant-based foods to a 23% reduced risk of type 2 diabetes, compared with lower consumption (relative risk, 0.77; 95% confidence interval, 0.71-0.84; P = .07 for heterogeneity). The risk dipped even further (down to 30%) when the researchers analyzed four studies that focused on healthy plant-based foods, such as fruits and vegetables, instead of foods such as refined grains, starches, and sugars (RR, 0.70; 95% CI, 0.62-0.79).
The researchers suggested that plant-based diets may lower the risk of diabetes type 2 by limiting weight gain. They also noted various limitations to their analysis, such as the reliance on self-reports and the observational nature of the studies.
Still, “in general populations that do not practice strict vegetarian or vegan diets, replacing animal products with healthful plant-based foods is likely to exert a significant reduction in the risk of diabetes,” the authors wrote.
The study was funded by the National Institutes of Health, and one author received support from the National Institute of Diabetes and Digestive and Kidney Diseases. The remaining authors reported no conflicts of interest withing the scope of this study.
SOURCE: Qian F et al. JAMA Internal Medicine. 2019 Jul 22. doi: 10.1001/jamainternmed.2019.2195.
according to a new systematic review and meta-analysis of nine observational studies.
The findings aren’t conclusive, but the study authors wrote in JAMA Internal Medicine that they suggest that “plant-based dietary patterns were associated with lower risk of type 2 diabetes, even after adjustment for [body mass index].”
According to 2015 figures provided by the American Diabetes Association, about 29 million people in the United States have type 2 diabetes. Overall, diabetes contributes to hundreds of thousands of deaths each year, which makes it the seventh-leading cause of death in the nation.
For the new analysis, researchers led by Frank Qian, MPH, of Harvard T.H. Chan School of Public Health included nine studies that examined diet and type 2 diabetes. In all, the studies included 307,099 participants, and there were 23,544 cases of incident type 2 diabetes. They were conducted in five countries, including the United States, and tracked participants for 2-28 years; the studies were all published within the last 11 years. The mean ages of participants ranged from 36-65 years.
The meta-analysis linked higher consumption of plant-based foods to a 23% reduced risk of type 2 diabetes, compared with lower consumption (relative risk, 0.77; 95% confidence interval, 0.71-0.84; P = .07 for heterogeneity). The risk dipped even further (down to 30%) when the researchers analyzed four studies that focused on healthy plant-based foods, such as fruits and vegetables, instead of foods such as refined grains, starches, and sugars (RR, 0.70; 95% CI, 0.62-0.79).
The researchers suggested that plant-based diets may lower the risk of diabetes type 2 by limiting weight gain. They also noted various limitations to their analysis, such as the reliance on self-reports and the observational nature of the studies.
Still, “in general populations that do not practice strict vegetarian or vegan diets, replacing animal products with healthful plant-based foods is likely to exert a significant reduction in the risk of diabetes,” the authors wrote.
The study was funded by the National Institutes of Health, and one author received support from the National Institute of Diabetes and Digestive and Kidney Diseases. The remaining authors reported no conflicts of interest withing the scope of this study.
SOURCE: Qian F et al. JAMA Internal Medicine. 2019 Jul 22. doi: 10.1001/jamainternmed.2019.2195.
FROM JAMA INTERNAL MEDICINE
FDA declines dapagliflozin indication as adjunct for type 1 diabetes
The Food and Drug Administration has rejected AstraZeneca’s supplemental New Drug Application for the sodium-glucose cotransporter 2 inhibitor dapagliflozin (Farxiga) as an adjunct treatment to insulin in adult patients with type 1 diabetes.
The company said in a press statement that the FDA had issued a complete response letter regarding the application. No reason was given for the decision, but the company said it would work with the agency to discuss the next steps.
The once-daily therapy has been approved as both a monotherapy and combination therapy, as an adjunct to diet and exercise, for improving glycemic control in adults with type 2 diabetes who cannot achieve control with insulin alone. It also has additional demonstrated benefits of weight loss and reduction in blood pressure.
On March 25, 2019, the drug received its first approval for treatment of patients with type 1 diabetes when the European Commission gave it the green light for use in patients with a body mass index of 27 kg/m2 or more when insulin alone does not provide adequate glycemic control. Japan followed a few days later with its approval of the sodium-glucose cotransporter 2 inhibitor, also for type 1 disease in adults.
The approvals for type 1 diabetes in the European Union and Japan were based on data from the phase 3 DEPICT (Dapagliflozin Evaluation in Patients With Inadequately Controlled Type 1 Diabetes) trial program (DEPICT-1 and DEPICT-2), which showed that 5 mg dapagliflozin, taken daily as an oral adjunct to insulin in patients with hard-to-control type 1 disease, reduced blood glucose levels from baseline (the primary endpoint). Secondary endpoints – reductions in weight and total daily insulin use – were also achieved.
Dapagliflozin’s safety profile in the trials in patients with type 1 diabetes was consistent with that established in patients with type 2 disease. However, there was a higher number of cases of diabetic ketoacidosis events in patients who received dapagliflozin. Diabetic ketoacidosis is a known complication for adults with type 1 diabetes and is more prevalent in patients with type 1 disease than in those with type 2.
The Food and Drug Administration has rejected AstraZeneca’s supplemental New Drug Application for the sodium-glucose cotransporter 2 inhibitor dapagliflozin (Farxiga) as an adjunct treatment to insulin in adult patients with type 1 diabetes.
The company said in a press statement that the FDA had issued a complete response letter regarding the application. No reason was given for the decision, but the company said it would work with the agency to discuss the next steps.
The once-daily therapy has been approved as both a monotherapy and combination therapy, as an adjunct to diet and exercise, for improving glycemic control in adults with type 2 diabetes who cannot achieve control with insulin alone. It also has additional demonstrated benefits of weight loss and reduction in blood pressure.
On March 25, 2019, the drug received its first approval for treatment of patients with type 1 diabetes when the European Commission gave it the green light for use in patients with a body mass index of 27 kg/m2 or more when insulin alone does not provide adequate glycemic control. Japan followed a few days later with its approval of the sodium-glucose cotransporter 2 inhibitor, also for type 1 disease in adults.
The approvals for type 1 diabetes in the European Union and Japan were based on data from the phase 3 DEPICT (Dapagliflozin Evaluation in Patients With Inadequately Controlled Type 1 Diabetes) trial program (DEPICT-1 and DEPICT-2), which showed that 5 mg dapagliflozin, taken daily as an oral adjunct to insulin in patients with hard-to-control type 1 disease, reduced blood glucose levels from baseline (the primary endpoint). Secondary endpoints – reductions in weight and total daily insulin use – were also achieved.
Dapagliflozin’s safety profile in the trials in patients with type 1 diabetes was consistent with that established in patients with type 2 disease. However, there was a higher number of cases of diabetic ketoacidosis events in patients who received dapagliflozin. Diabetic ketoacidosis is a known complication for adults with type 1 diabetes and is more prevalent in patients with type 1 disease than in those with type 2.
The Food and Drug Administration has rejected AstraZeneca’s supplemental New Drug Application for the sodium-glucose cotransporter 2 inhibitor dapagliflozin (Farxiga) as an adjunct treatment to insulin in adult patients with type 1 diabetes.
The company said in a press statement that the FDA had issued a complete response letter regarding the application. No reason was given for the decision, but the company said it would work with the agency to discuss the next steps.
The once-daily therapy has been approved as both a monotherapy and combination therapy, as an adjunct to diet and exercise, for improving glycemic control in adults with type 2 diabetes who cannot achieve control with insulin alone. It also has additional demonstrated benefits of weight loss and reduction in blood pressure.
On March 25, 2019, the drug received its first approval for treatment of patients with type 1 diabetes when the European Commission gave it the green light for use in patients with a body mass index of 27 kg/m2 or more when insulin alone does not provide adequate glycemic control. Japan followed a few days later with its approval of the sodium-glucose cotransporter 2 inhibitor, also for type 1 disease in adults.
The approvals for type 1 diabetes in the European Union and Japan were based on data from the phase 3 DEPICT (Dapagliflozin Evaluation in Patients With Inadequately Controlled Type 1 Diabetes) trial program (DEPICT-1 and DEPICT-2), which showed that 5 mg dapagliflozin, taken daily as an oral adjunct to insulin in patients with hard-to-control type 1 disease, reduced blood glucose levels from baseline (the primary endpoint). Secondary endpoints – reductions in weight and total daily insulin use – were also achieved.
Dapagliflozin’s safety profile in the trials in patients with type 1 diabetes was consistent with that established in patients with type 2 disease. However, there was a higher number of cases of diabetic ketoacidosis events in patients who received dapagliflozin. Diabetic ketoacidosis is a known complication for adults with type 1 diabetes and is more prevalent in patients with type 1 disease than in those with type 2.
CARMELINA confirms linagliptin’s renal, CV safety, but it’s still third-line for type 2 diabetes
SAN FRANCISCO – The dipeptidyl peptidase-4 inhibitor linagliptin (Tradjenta) is safe on the kidneys, the cardiovascular system, and in older people with type 2 diabetes, according to findings presented at the annual scientific sessions of the American Diabetes Association.
Investigators in the international Cardiovascular and Renal Microvascular Outcome Study with Linagliptin (CARMELINA) randomized 6,979 patients with type 2 diabetes who also had cardiovascular and/or kidney disease 1:1 to daily oral linagliptin 5 mg or placebo on top of standard of care, and they followed them for a median of 2.2 years. The mean age was 65.9 years, baseline hemoglobin A1c was 8.0%, and disease duration was about 15 years. Almost 63% of the patients were men, and about a quarter had a history of heart failure at baseline (JAMA. 2019;321[1]:69-79).
The study was unusual among other DPP-4 trials in that almost 60% of the patients were older than 65 years and 62.3% had impaired renal function with an estimated glomerular filtration rate (eGFR) of less than 60 ml/min per 1.73 m2.
There was no increased risk with linagliptin, compared with placebo, in the primary composite outcome of cardiovascular death, nonfatal stroke, or nonfatal myocardial infarction (12.4% vs. 12.1%, respectively; hazard ratio, 1.02; P = .74), and there was no difference between the individual components even when broken down by age (younger than 65, 65-75, or older than 75 years) or by renal function (eGFR 60 or more, 45 to less than 60, 30 to less than 45, or less than 30 ml/min per 1.73 m2), according to investigator Mark Cooper, MBBS, PhD, of the department of diabetes at Monash University, Melbourne, who presented the findings.
There was no increase in the number of hospitalizations for heart failure with linagliptin, compared with placebo (6% vs. 6.5%, respectively; HR, 0.90; P = .26) – a concern with some DPP-4 inhibitors – and no increase in hypoglycemia (just over a quarter in both groups), even when broken down by age and renal function.
A decrease in albuminuria with linagliptin held across all renal subgroups. It is not known if that was because of glucose lowering or some other effect, but Dr. Cooper said he believed there was “a modest renal protective effect, [although] not at the level one would expect to translate into hard renal outcomes.”
Robert Eckel, MD, a professor of medicine at the University of Colorado at Denver, Aurora, who moderated the session, said the results were reassuring. “Ultimately, linagliptin seems safe,” even in older people with reduced eGFR. “It does not improve cardiovascular outcomes, but based on many DPP-4 trials, we didn’t expect it to,” he said.
“I don’t think DPP-4s are going to fall into any different place in the [treatment] algorithm” based on these results, he added. The class is currently third-line after metformin or insulin, followed by sodium-glucose cotransporter 2 inhibitors or glucagonlike peptide–1 receptor agonists for cardiovascular protection.
“When we look at [cardiovascular outcomes], ultimately, the SGLT2 inhibitors and the GLP-1 receptor agonists win,” he said. In addition, the blood glucose effects of linagliptin are “pretty modest, so if lowering hemoglobin A1c is the focus, this drug would be lower down on the list.”
Overall, linagliptin “falls into a lesser class, but a safe class for certain circumstances,” said Dr. Eckel, who gave the example of a woman in her late 70s with moderate to severe kidney function, an HbA1c level of 7.9%, and no cardiovascular disease. Her HbA1c might get down to 7.6% or so with linagliptin, he said, “but I’m not sure we have absolute proof of the benefit” of such a modest decline.
Boehringer Ingelheim, the maker of linagliptin, funded the study. The presenter disclosed honoraria, speaking fees, and grants from the company. A number of the investigators were employees of the company.
SAN FRANCISCO – The dipeptidyl peptidase-4 inhibitor linagliptin (Tradjenta) is safe on the kidneys, the cardiovascular system, and in older people with type 2 diabetes, according to findings presented at the annual scientific sessions of the American Diabetes Association.
Investigators in the international Cardiovascular and Renal Microvascular Outcome Study with Linagliptin (CARMELINA) randomized 6,979 patients with type 2 diabetes who also had cardiovascular and/or kidney disease 1:1 to daily oral linagliptin 5 mg or placebo on top of standard of care, and they followed them for a median of 2.2 years. The mean age was 65.9 years, baseline hemoglobin A1c was 8.0%, and disease duration was about 15 years. Almost 63% of the patients were men, and about a quarter had a history of heart failure at baseline (JAMA. 2019;321[1]:69-79).
The study was unusual among other DPP-4 trials in that almost 60% of the patients were older than 65 years and 62.3% had impaired renal function with an estimated glomerular filtration rate (eGFR) of less than 60 ml/min per 1.73 m2.
There was no increased risk with linagliptin, compared with placebo, in the primary composite outcome of cardiovascular death, nonfatal stroke, or nonfatal myocardial infarction (12.4% vs. 12.1%, respectively; hazard ratio, 1.02; P = .74), and there was no difference between the individual components even when broken down by age (younger than 65, 65-75, or older than 75 years) or by renal function (eGFR 60 or more, 45 to less than 60, 30 to less than 45, or less than 30 ml/min per 1.73 m2), according to investigator Mark Cooper, MBBS, PhD, of the department of diabetes at Monash University, Melbourne, who presented the findings.
There was no increase in the number of hospitalizations for heart failure with linagliptin, compared with placebo (6% vs. 6.5%, respectively; HR, 0.90; P = .26) – a concern with some DPP-4 inhibitors – and no increase in hypoglycemia (just over a quarter in both groups), even when broken down by age and renal function.
A decrease in albuminuria with linagliptin held across all renal subgroups. It is not known if that was because of glucose lowering or some other effect, but Dr. Cooper said he believed there was “a modest renal protective effect, [although] not at the level one would expect to translate into hard renal outcomes.”
Robert Eckel, MD, a professor of medicine at the University of Colorado at Denver, Aurora, who moderated the session, said the results were reassuring. “Ultimately, linagliptin seems safe,” even in older people with reduced eGFR. “It does not improve cardiovascular outcomes, but based on many DPP-4 trials, we didn’t expect it to,” he said.
“I don’t think DPP-4s are going to fall into any different place in the [treatment] algorithm” based on these results, he added. The class is currently third-line after metformin or insulin, followed by sodium-glucose cotransporter 2 inhibitors or glucagonlike peptide–1 receptor agonists for cardiovascular protection.
“When we look at [cardiovascular outcomes], ultimately, the SGLT2 inhibitors and the GLP-1 receptor agonists win,” he said. In addition, the blood glucose effects of linagliptin are “pretty modest, so if lowering hemoglobin A1c is the focus, this drug would be lower down on the list.”
Overall, linagliptin “falls into a lesser class, but a safe class for certain circumstances,” said Dr. Eckel, who gave the example of a woman in her late 70s with moderate to severe kidney function, an HbA1c level of 7.9%, and no cardiovascular disease. Her HbA1c might get down to 7.6% or so with linagliptin, he said, “but I’m not sure we have absolute proof of the benefit” of such a modest decline.
Boehringer Ingelheim, the maker of linagliptin, funded the study. The presenter disclosed honoraria, speaking fees, and grants from the company. A number of the investigators were employees of the company.
SAN FRANCISCO – The dipeptidyl peptidase-4 inhibitor linagliptin (Tradjenta) is safe on the kidneys, the cardiovascular system, and in older people with type 2 diabetes, according to findings presented at the annual scientific sessions of the American Diabetes Association.
Investigators in the international Cardiovascular and Renal Microvascular Outcome Study with Linagliptin (CARMELINA) randomized 6,979 patients with type 2 diabetes who also had cardiovascular and/or kidney disease 1:1 to daily oral linagliptin 5 mg or placebo on top of standard of care, and they followed them for a median of 2.2 years. The mean age was 65.9 years, baseline hemoglobin A1c was 8.0%, and disease duration was about 15 years. Almost 63% of the patients were men, and about a quarter had a history of heart failure at baseline (JAMA. 2019;321[1]:69-79).
The study was unusual among other DPP-4 trials in that almost 60% of the patients were older than 65 years and 62.3% had impaired renal function with an estimated glomerular filtration rate (eGFR) of less than 60 ml/min per 1.73 m2.
There was no increased risk with linagliptin, compared with placebo, in the primary composite outcome of cardiovascular death, nonfatal stroke, or nonfatal myocardial infarction (12.4% vs. 12.1%, respectively; hazard ratio, 1.02; P = .74), and there was no difference between the individual components even when broken down by age (younger than 65, 65-75, or older than 75 years) or by renal function (eGFR 60 or more, 45 to less than 60, 30 to less than 45, or less than 30 ml/min per 1.73 m2), according to investigator Mark Cooper, MBBS, PhD, of the department of diabetes at Monash University, Melbourne, who presented the findings.
There was no increase in the number of hospitalizations for heart failure with linagliptin, compared with placebo (6% vs. 6.5%, respectively; HR, 0.90; P = .26) – a concern with some DPP-4 inhibitors – and no increase in hypoglycemia (just over a quarter in both groups), even when broken down by age and renal function.
A decrease in albuminuria with linagliptin held across all renal subgroups. It is not known if that was because of glucose lowering or some other effect, but Dr. Cooper said he believed there was “a modest renal protective effect, [although] not at the level one would expect to translate into hard renal outcomes.”
Robert Eckel, MD, a professor of medicine at the University of Colorado at Denver, Aurora, who moderated the session, said the results were reassuring. “Ultimately, linagliptin seems safe,” even in older people with reduced eGFR. “It does not improve cardiovascular outcomes, but based on many DPP-4 trials, we didn’t expect it to,” he said.
“I don’t think DPP-4s are going to fall into any different place in the [treatment] algorithm” based on these results, he added. The class is currently third-line after metformin or insulin, followed by sodium-glucose cotransporter 2 inhibitors or glucagonlike peptide–1 receptor agonists for cardiovascular protection.
“When we look at [cardiovascular outcomes], ultimately, the SGLT2 inhibitors and the GLP-1 receptor agonists win,” he said. In addition, the blood glucose effects of linagliptin are “pretty modest, so if lowering hemoglobin A1c is the focus, this drug would be lower down on the list.”
Overall, linagliptin “falls into a lesser class, but a safe class for certain circumstances,” said Dr. Eckel, who gave the example of a woman in her late 70s with moderate to severe kidney function, an HbA1c level of 7.9%, and no cardiovascular disease. Her HbA1c might get down to 7.6% or so with linagliptin, he said, “but I’m not sure we have absolute proof of the benefit” of such a modest decline.
Boehringer Ingelheim, the maker of linagliptin, funded the study. The presenter disclosed honoraria, speaking fees, and grants from the company. A number of the investigators were employees of the company.
REPORTING FROM ADA 2019
The costs and benefits of SGLT2 inhibitors & GLP-1 RAs
The options for treating type 2 diabetes without insulin have grown beyond metformin to include a long list of sodium-glucose cotransporter 2 (SGLT2) inhibitors and glucagonlike peptide–1 (GLP-1) receptor agonists that can be taken with or without metformin. These new drugs have cardiovascular and kidney benefits and help with weight loss, but they also carry risks and, according to some experts, their costs can be prohibitively expensive.
Given the medical community’s long-term experience with treating patients with metformin, and metformin’s lower cost, most of the physicians interviewed for this article advise using SGLT2 inhibitors and GLP-1 receptor agonists as second-line treatments. Others said that they would prefer to use the newer drugs as first-line therapies in select high-risk patients, but prior authorization hurdles created by insurance companies make that approach too burdensome.
“The economics of U.S. health care is stacked against many of our patients with diabetes in the current era,” Robert H. Hopkins Jr., MD, said in an interview.
Even when their insurance approves the drugs, patients still may not be able to afford the copay, explained Dr. Hopkins, professor of internal medicine and pediatrics and director of the division of general internal medicine at the University of Arkansas for Medical Sciences, Little Rock. “Sometimes patients can purchase drugs at a lower cost than the copay to purchase with the ‘drug coverage’ in their insurance plan – unfortunately, this is not the case with the newer diabetes medications we are discussing here.”
“SGLT2 inhibitors and GLP-1 agonists can cost several hundred dollars a month, and insurers often balk at paying for them. They’ll say, ‘Have you tried metformin?’ ” explained endocrinologist Victor Lawrence Roberts, MD, in a interview. “We have to work with insurance companies the best we can in a stepwise fashion.”
According to Dr. Roberts, 80% of his patients with diabetes struggle with the cost of medicine in general. “They’re either underinsured or not insured or their formulary is limited.
Douglas S. Paauw, MD, agreed in an interview that the newer drugs can be problematic on the insurance front.
“For some patients they aren’t affordable, especially for the uninsured if you can’t get them on an assistance program,” said Dr. Paauw, who is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the university.
Dr. Hopkins, who is on the Internal Medicine News board, noted that “unfortunately, the treatment of type 2 diabetes in patients who cannot achieve control with metformin, diet, weight control, and exercise is a story of the ‘haves’ and the ‘have nots.’ The ‘haves’ are those who have pharmacy benefits which make access to newer agents like SGLT2 inhibitors and GLP-1 agonists a possibility.”
“I have had very few of the ‘have nots’ who have been able to even consider these newer agents, which carry price tags of $600-$1,300 a month even with the availability of discounting coupons in the marketplace,” he added. “Most of these patients end up requiring a sulfonylurea or TZD [thiazolidinedione] as a second agent to achieve glycemic control. This makes it very difficult to achieve sufficient weight and metabolic control to avoid an eventual switch to insulin.”
Fatima Z. Syed, MD, an endocrine-trained general internist at DukeHealth in Durham, N.C., said she prescribes SGLT2 inhibitors and GLP-1 receptor agonists in combination with metformin. “I prescribe them frequently, but they are not first-line treatments,” she explained.
“Nothing replaces diet and exercise” as therapy for patients with type 2 diabetes, she added.
Neil S. Skolnik, MD, said that insurance companies were not preventing patients from using these drugs in his experience. He also provided an optimistic take on the accessibility of these drugs in the near future.
“Most insurance companies are now covering select SGLT2 inhibitors and GLP-1 receptor agonists for appropriate patients and those companies that currently do not will soon have to,” said Dr. Skolnik, who is a professor of family and community medicine at Jefferson Medical College, Philadelphia, and an associate director of the family medicine residency program at Abington (Pa.) Jefferson Health.
“The outcomes [associated with use of the new drugs] are robust, the benefits are large, and are well worth the cost,” he added.
The side effects
While others praised these drugs for their beneficial effects, they also noted that the side effects of these drugs are serious and must be discussed with patients.
GLP-1 receptor agonists are linked to gastrointestinal symptoms, especially nausea, while SGLT2 inhibitors have been linked to kidney failure, ketoacidosis, and more. The Food and Drug Administration warned in 2018 that the SGLT2 inhibitors can cause a rare serious infection known as Fournier’s gangrene – necrotizing fasciitis of the perineum.
“We have to tell our patients to let us know right away if they get pain or swelling in the genital area,” Dr. Paauw, who is on the Internal Medicine News board, noted. “The chance that an infection could explode quickly is higher in those who take these drugs.”
Amputation risks also are associated with taking the SGLT2 inhibitor canagliflozin (Invokana). The FDA requires the manufacturer of this drug to include a black-box warning about the risk of “lower-limb amputations, most frequently of the toe and midfoot,” but also the leg. In approval trials, the risk doubled versus placebo.
These amputation risks “put a damper on some of the enthusiasm on behalf of physicians and patients ... for taking this drug,” noted Dr. Roberts, who is a professor of internal medicine at the University of Central Florida, Orlando.
While a manufacturer-funded study released last year found no link to amputations, the results weren’t powerful enough to rule out a moderately increased risk.
“[If] you are at high risk for having an amputation, we really have to take this risk very seriously,” said John B. Buse, MD, chief of the division of endocrinology at the University of North Carolina at Chapel Hill, in a presentation about the study at the 2018 annual scientific sessions of the American Diabetes Association.
The benefits
Despite these risks of adverse events, most interviewed agreed that the many benefits observed in those taking SGLT2 inhibitors or GLP-1 receptor agonists make them worth prescribing, at least to those who are able to afford them.
Both SGLT2 inhibitors and GLP-1 receptor agonists appear to have significant cardiovascular benefits. A 2019 meta-analysis and systematic review found that both drugs reduced major adverse cardiac events by about 12% (Circulation. 2019 Apr 23;139[17]:2022-31).
“They don’t cause hypoglycemia, they lower blood pressure, they don’t cause weight gain, and they might promote weight loss,” noted Dr. Paauw.
SGLT2 inhibitors also have shown signs of kidney benefits. The CREDENCE trial linked canagliflozin to a lowering of kidney disorders versus placebo (N Engl J Med. 2019 Jun 13;380[24]:2295-306). “The relative risk of the renal-specific composite of end-stage kidney disease, a doubling of the creatinine level, or death from renal causes was lower by 34% (hazard ratio, 0.66; 95% confidence interval, 0.53-0.81; P less than .001), and the relative risk of end-stage kidney disease was lower by 32% (HR, 0.68; 95% CI, 0.54-0.86; P = .002),” the trial investigators wrote.
“They showed very nicely that the drug improved the kidney function of those patients and reduced the kidney deterioration,” said Yehuda Handelsman, MD, an endocrinologist in Tarzana, Calif., who chaired the 2011 and 2015 American Association of Clinical Endocrinologists’ Comprehensive Diabetes Guidelines. The study was especially impressive, he added, because it included patients with low kidney function.
SGLT2 inhibitors’ “diuretic mechanism explains why there is a substantial reduction in heart failure hospitalizations in patients who take these drugs,” said cardiologist Marc E. Goldschmidt, MD, director of the Heart Success Program at Atlantic Health System’s Morristown (N.J.) Medical Center, in an interview. “Both the EMPA-REG Outcome and the CREDENCE trials demonstrated substantial benefit of this class of medications by showing a lower risk of cardiovascular death as well as death from any cause and a lower risk of hospitalization for heart failure."
Overall, the SGLT2 trial data have been very consistent with a benefit for cardiovascular risk reduction, particularly in regard to heart failure hospitalizations and even in potentially preventing heart failure in diabetics,” he added.
Dr. Skolnik, a columnist for Family Practice News, cited SGLT2 inhibitors and GLP-1 receptor agonists’ ability to slow renal disease progression, promote weight loss, and prevent poor cardiac outcomes.“These drugs should be used, in addition to metformin, in all patients with diabetes and vascular disease. These proven outcomes are far better than we ever were able to achieve previously and the strength of the evidence at this point is very strong,” said Dr. Skolnik. “In addition to the benefits of decreasing the development of cardiovascular disease, serious heart failure, and slowing progression of renal disease, these two classes of medication have additional benefits. Both classes help patients lose weight, which is very different from what was found with either sulfonylureas or insulin, which cause patients to gain weight. Also both the SGLT2 inhibitors and the GLP-1 RAs [receptor agonists] have a low incidence of hypoglycemia. For all these reasons, these have become important medications for us to use in primary care.”
Other recent trials offer “very powerful data” about SGLT2 inhibitors, Dr. Roberts said. That’s good news, since “our approach needs to be toward cardiovascular protection and preservation as well as managing blood sugar.”An Israeli trial, whose results were released in May 2019 at the annual meeting of the American College of Cardiology, found that, compared with other glucose-lowering drugs, taking an SGLT2 inhibitor was associated with lower risks of heart failure hospitalization and all-cause mortality (HR, 0.54; 95% CI, 0.44-0.65; P less than .001). This trial also offered a new detail: The patients gained the benefit regardless of whether their baseline left ventricular ejection fraction was preserved or reduced (J Coll Cardiol. 2019 Mar;73[9]:suppl 1). The SGLT2 inhibitors used in this trial included dapagliflozin (Farxiga) and empagliflozin (Jardiance).
In another study released this year, a subanalysis of the DECLARE-TIMI 58 trial, researchers reported that the SGLT2 inhibitor dapagliflozin reduced risks of both major adverse cardiovascular events and heart failure hospitalization in the subset of patients with type 2 diabetes and prior myocardial infarction versus controls (Circulation. 2019 May 28;139[22]:2516-27). The absolute risk reduction for major adverse cardiovascular events was 1.9% (HR, 0.81; 95% CI, 0.65-1.00; P = .046), while it was 0.6% for heart failure hospitalization (HR, 0.85; 95% CI, 0.72-1.00; P = .055).
These and other studies “speak volumes about the efficacy of managing blood sugar and addressing our biggest nemesis, which is cardiovascular disease,” Dr. Roberts said. “It’s irrefutable. The data [are] very good.”
Dr. Paauw said an SGLT2 inhibitor or GLP-1 receptor agonist is best reserved for use in select patients with cardiovascular risks and type 2 diabetes that need management beyond metformin.
For example, they might fit a 70-year-old with persistent hypertension who’s already taking a couple of blood pressure medications. “If they have another cardiovascular risk factor, the cardiovascular protection piece will be a bigger deal,” he said. Also, “it will probably help lower their blood pressure so they can avoid taking another blood pressure medicine.”
Trials of both GLP-1 receptor agonists and SGLT2 inhibitors have shown benefits “in improving [major adverse cardiac events], with the SGLT2 class showing substantial benefit in improving both heart failure and renal outcomes as well,” noted Dr. Skolnik. “It is in this context that one must address the question of whether the price of the medications are worthwhile. With such substantial benefit, there is no question in my mind that – for patients who have underlying cardiovascular illness, which includes patients with existent coronary disease, history of stroke, transient ischemic attack, or peripheral vascular disease – it is far and away worth it to prescribe these classes of medications.”
Indeed, the American Diabetes Association and the European Association for the Study of Diabetes’ most recent guidelines now call for a GLP-1 receptor agonist – instead of insulin – to be the first injectable used to treat type 2 diabetes (Diabetes Care 2018 Dec; 41[12]:2669-701).
“For the relatively small number of my patients who have been able to access and use these medications for months or longer, more have tolerated the GLP-1 agonists than SGLT2 inhibitors primarily due to urinary issues,” noted Dr. Hopkins.
Dipeptidyl peptidase–4 inhibitors are another option in patients with type 2 diabetes, but research suggests they may not be a top option for patients with cardiovascular risk. A 2018 review noted that cardiovascular outcome trials for alogliptin (Nesina), saxagliptin (Onglyza), and sitagliptin (Januvia) showed noninferiority but failed to demonstrate any superiority, compared with placebo in patients with type 2 diabetes mellitus and high cardiovascular risk (Circ Res. 2018 May 11;122[10]:1439-59).
The combination therapies
Many of the newer drugs are available as combinations with other types of diabetes drugs. In some cases, physicians create their own form of combination therapy by separately prescribing two or more diabetes drugs. Earlier this year, a study suggested the benefits of this kind of add-on therapy: Diabetes outcomes improved in patients who took the GLP-1 receptor agonist semaglutide and an SGLT2 inhibitor (Lancet Diabetes Endocrinol. 2019 Mar 1. doi: 10.1016/S2213-8587[19]30066-X).
Dr. Roberts suggested caution, however, when prescribing combination therapies. “My recommendation is always to begin with the individual medications to see if the patient tolerates the drugs and then decide which component needs to be titrated. It’s hard to titrate a combination drug, and it doesn’t leave a lot of flexibility. You never know which drug is doing what.
Dr. Handelsman said some patients may need to take three medications such as metformin, an SGLT2 inhibitor, and a GLP-1 receptor agonist.
“I don’t recommend using the combinations if you’re not familiar with the drugs ... These are relatively new pharmaceuticals, and most of us are on a learning curve as to how they fit into the armamentarium. If a drug is tolerated with a good response, you can certainly consider going to the combination tablets,” he added.
There is at least one drug that combines these three classes: The newly FDA-approved Qternmet XR, which combines dapagliflozin (an SGLT2 inhibitor), saxagliptin (a GLP-1 receptor agonist), and metformin. As of mid-June 2019, it was not yet available in the United States. Its sister drug Qtern, which combines dapagliflozin and saxagliptin, costs more than $500 a month with a free coupon, according to goodrx.com. In contrast, metformin is extremely inexpensive, costing just a few dollars a month for a common starting dose.
What about adding insulin?
“Both [SGLT2 inhibitors and GLP-1 receptor agonists] work very well with insulin,” Dr. Handelsman said. “There is a nice additive effect on the reduction of [hemoglobin] A1c. The only caution is that, although neither SGLT2 inhibitors nor GLP-1 receptor agonists cause hypoglycemia, in combination with insulin they do increase the risk of hypoglycemia. You may have to adjust the dose of insulin.”
Dr. Hopkins warned that cost becomes an even bigger issue when you add insulin into the mix.
“When insulin comes into the discussion, we are again stuck with astronomical costs which many struggle to afford,” he explained.
Indeed, the price tag on these drugs seems to be the biggest problem physicians have with them.
“The challenges in managing patients with diabetes aren’t the risks associated with the drugs. It’s dealing with their insurers,” noted Dr. Roberts.
Dr. Hopkins, Dr. Paauw, Dr. Roberts, and Dr. Syed reported no disclosures. Dr. Buse is an investigator for Johnson and Johnson. Dr. Goldschmidt is paid to speak by Novartis. Dr. Handelsman reported research grants, consulting work, and speaker honoraria from Amgen, Gilead, Lilly, Merck, Novo Nordisk, and others. Dr Skolnik reported nonfinancial support from AstraZeneca, Boehringer Ingelheim, Sanofi, and GlaxoSmithKline and personal fees from AstraZeneca, Boehringer Ingelheim, and Eli Lilly. He also serves on the advisory boards of AstraZeneca, Boehringer Ingelheim, Teva Pharmaceutical, Eli Lilly, Sanofi, Janssen Pharmaceuticals, Intarcia, Mylan, and GlaxoSmithKline.
Dr. Paauw and Dr. Skolnik are columnists for Family Practice News and Internal Medicine News.
M. Alexander Otto contributed to this report.
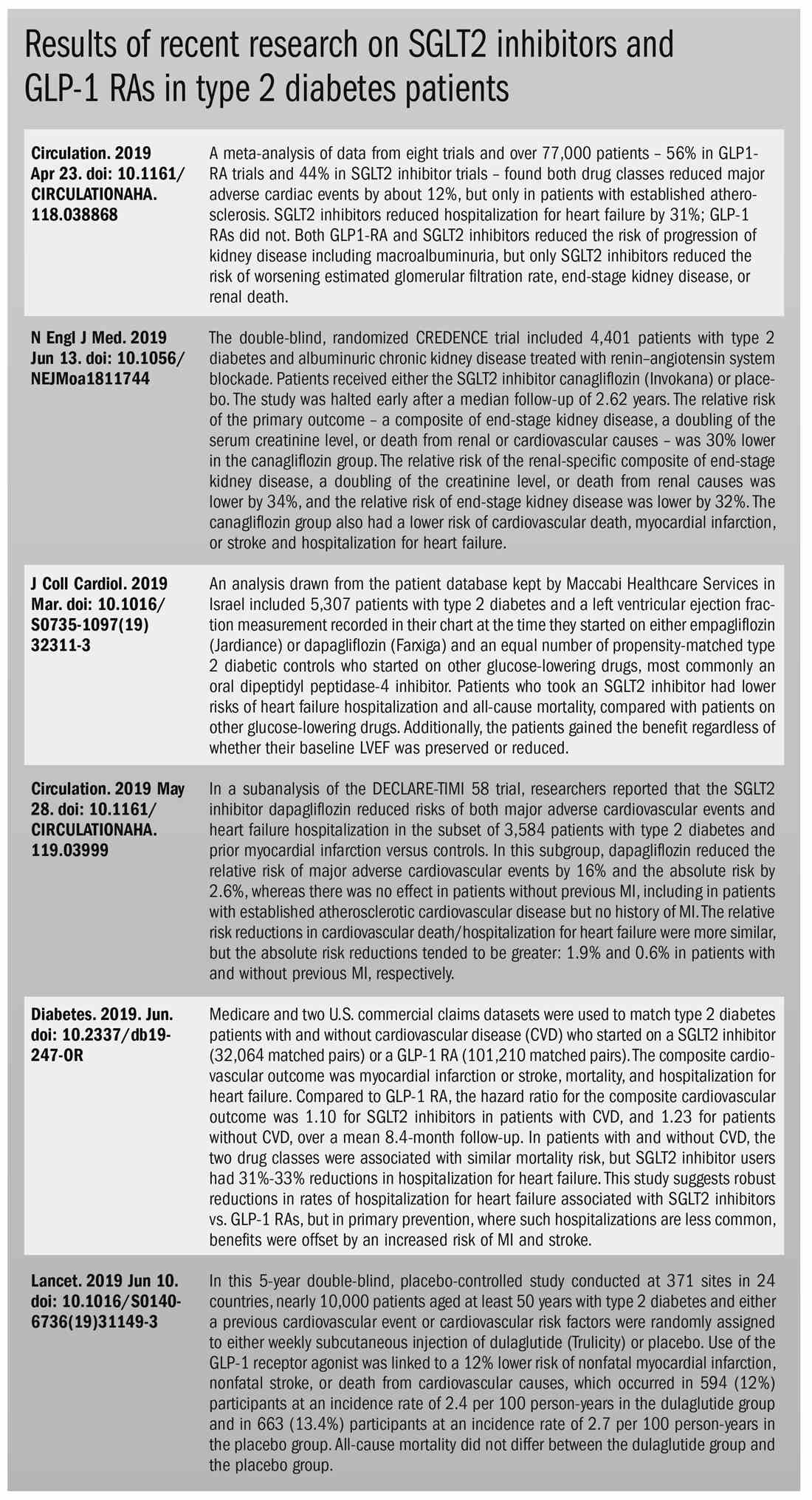
The options for treating type 2 diabetes without insulin have grown beyond metformin to include a long list of sodium-glucose cotransporter 2 (SGLT2) inhibitors and glucagonlike peptide–1 (GLP-1) receptor agonists that can be taken with or without metformin. These new drugs have cardiovascular and kidney benefits and help with weight loss, but they also carry risks and, according to some experts, their costs can be prohibitively expensive.
Given the medical community’s long-term experience with treating patients with metformin, and metformin’s lower cost, most of the physicians interviewed for this article advise using SGLT2 inhibitors and GLP-1 receptor agonists as second-line treatments. Others said that they would prefer to use the newer drugs as first-line therapies in select high-risk patients, but prior authorization hurdles created by insurance companies make that approach too burdensome.
“The economics of U.S. health care is stacked against many of our patients with diabetes in the current era,” Robert H. Hopkins Jr., MD, said in an interview.
Even when their insurance approves the drugs, patients still may not be able to afford the copay, explained Dr. Hopkins, professor of internal medicine and pediatrics and director of the division of general internal medicine at the University of Arkansas for Medical Sciences, Little Rock. “Sometimes patients can purchase drugs at a lower cost than the copay to purchase with the ‘drug coverage’ in their insurance plan – unfortunately, this is not the case with the newer diabetes medications we are discussing here.”
“SGLT2 inhibitors and GLP-1 agonists can cost several hundred dollars a month, and insurers often balk at paying for them. They’ll say, ‘Have you tried metformin?’ ” explained endocrinologist Victor Lawrence Roberts, MD, in a interview. “We have to work with insurance companies the best we can in a stepwise fashion.”
According to Dr. Roberts, 80% of his patients with diabetes struggle with the cost of medicine in general. “They’re either underinsured or not insured or their formulary is limited.
Douglas S. Paauw, MD, agreed in an interview that the newer drugs can be problematic on the insurance front.
“For some patients they aren’t affordable, especially for the uninsured if you can’t get them on an assistance program,” said Dr. Paauw, who is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the university.
Dr. Hopkins, who is on the Internal Medicine News board, noted that “unfortunately, the treatment of type 2 diabetes in patients who cannot achieve control with metformin, diet, weight control, and exercise is a story of the ‘haves’ and the ‘have nots.’ The ‘haves’ are those who have pharmacy benefits which make access to newer agents like SGLT2 inhibitors and GLP-1 agonists a possibility.”
“I have had very few of the ‘have nots’ who have been able to even consider these newer agents, which carry price tags of $600-$1,300 a month even with the availability of discounting coupons in the marketplace,” he added. “Most of these patients end up requiring a sulfonylurea or TZD [thiazolidinedione] as a second agent to achieve glycemic control. This makes it very difficult to achieve sufficient weight and metabolic control to avoid an eventual switch to insulin.”
Fatima Z. Syed, MD, an endocrine-trained general internist at DukeHealth in Durham, N.C., said she prescribes SGLT2 inhibitors and GLP-1 receptor agonists in combination with metformin. “I prescribe them frequently, but they are not first-line treatments,” she explained.
“Nothing replaces diet and exercise” as therapy for patients with type 2 diabetes, she added.
Neil S. Skolnik, MD, said that insurance companies were not preventing patients from using these drugs in his experience. He also provided an optimistic take on the accessibility of these drugs in the near future.
“Most insurance companies are now covering select SGLT2 inhibitors and GLP-1 receptor agonists for appropriate patients and those companies that currently do not will soon have to,” said Dr. Skolnik, who is a professor of family and community medicine at Jefferson Medical College, Philadelphia, and an associate director of the family medicine residency program at Abington (Pa.) Jefferson Health.
“The outcomes [associated with use of the new drugs] are robust, the benefits are large, and are well worth the cost,” he added.
The side effects
While others praised these drugs for their beneficial effects, they also noted that the side effects of these drugs are serious and must be discussed with patients.
GLP-1 receptor agonists are linked to gastrointestinal symptoms, especially nausea, while SGLT2 inhibitors have been linked to kidney failure, ketoacidosis, and more. The Food and Drug Administration warned in 2018 that the SGLT2 inhibitors can cause a rare serious infection known as Fournier’s gangrene – necrotizing fasciitis of the perineum.
“We have to tell our patients to let us know right away if they get pain or swelling in the genital area,” Dr. Paauw, who is on the Internal Medicine News board, noted. “The chance that an infection could explode quickly is higher in those who take these drugs.”
Amputation risks also are associated with taking the SGLT2 inhibitor canagliflozin (Invokana). The FDA requires the manufacturer of this drug to include a black-box warning about the risk of “lower-limb amputations, most frequently of the toe and midfoot,” but also the leg. In approval trials, the risk doubled versus placebo.
These amputation risks “put a damper on some of the enthusiasm on behalf of physicians and patients ... for taking this drug,” noted Dr. Roberts, who is a professor of internal medicine at the University of Central Florida, Orlando.
While a manufacturer-funded study released last year found no link to amputations, the results weren’t powerful enough to rule out a moderately increased risk.
“[If] you are at high risk for having an amputation, we really have to take this risk very seriously,” said John B. Buse, MD, chief of the division of endocrinology at the University of North Carolina at Chapel Hill, in a presentation about the study at the 2018 annual scientific sessions of the American Diabetes Association.
The benefits
Despite these risks of adverse events, most interviewed agreed that the many benefits observed in those taking SGLT2 inhibitors or GLP-1 receptor agonists make them worth prescribing, at least to those who are able to afford them.
Both SGLT2 inhibitors and GLP-1 receptor agonists appear to have significant cardiovascular benefits. A 2019 meta-analysis and systematic review found that both drugs reduced major adverse cardiac events by about 12% (Circulation. 2019 Apr 23;139[17]:2022-31).
“They don’t cause hypoglycemia, they lower blood pressure, they don’t cause weight gain, and they might promote weight loss,” noted Dr. Paauw.
SGLT2 inhibitors also have shown signs of kidney benefits. The CREDENCE trial linked canagliflozin to a lowering of kidney disorders versus placebo (N Engl J Med. 2019 Jun 13;380[24]:2295-306). “The relative risk of the renal-specific composite of end-stage kidney disease, a doubling of the creatinine level, or death from renal causes was lower by 34% (hazard ratio, 0.66; 95% confidence interval, 0.53-0.81; P less than .001), and the relative risk of end-stage kidney disease was lower by 32% (HR, 0.68; 95% CI, 0.54-0.86; P = .002),” the trial investigators wrote.
“They showed very nicely that the drug improved the kidney function of those patients and reduced the kidney deterioration,” said Yehuda Handelsman, MD, an endocrinologist in Tarzana, Calif., who chaired the 2011 and 2015 American Association of Clinical Endocrinologists’ Comprehensive Diabetes Guidelines. The study was especially impressive, he added, because it included patients with low kidney function.
SGLT2 inhibitors’ “diuretic mechanism explains why there is a substantial reduction in heart failure hospitalizations in patients who take these drugs,” said cardiologist Marc E. Goldschmidt, MD, director of the Heart Success Program at Atlantic Health System’s Morristown (N.J.) Medical Center, in an interview. “Both the EMPA-REG Outcome and the CREDENCE trials demonstrated substantial benefit of this class of medications by showing a lower risk of cardiovascular death as well as death from any cause and a lower risk of hospitalization for heart failure."
Overall, the SGLT2 trial data have been very consistent with a benefit for cardiovascular risk reduction, particularly in regard to heart failure hospitalizations and even in potentially preventing heart failure in diabetics,” he added.
Dr. Skolnik, a columnist for Family Practice News, cited SGLT2 inhibitors and GLP-1 receptor agonists’ ability to slow renal disease progression, promote weight loss, and prevent poor cardiac outcomes.“These drugs should be used, in addition to metformin, in all patients with diabetes and vascular disease. These proven outcomes are far better than we ever were able to achieve previously and the strength of the evidence at this point is very strong,” said Dr. Skolnik. “In addition to the benefits of decreasing the development of cardiovascular disease, serious heart failure, and slowing progression of renal disease, these two classes of medication have additional benefits. Both classes help patients lose weight, which is very different from what was found with either sulfonylureas or insulin, which cause patients to gain weight. Also both the SGLT2 inhibitors and the GLP-1 RAs [receptor agonists] have a low incidence of hypoglycemia. For all these reasons, these have become important medications for us to use in primary care.”
Other recent trials offer “very powerful data” about SGLT2 inhibitors, Dr. Roberts said. That’s good news, since “our approach needs to be toward cardiovascular protection and preservation as well as managing blood sugar.”An Israeli trial, whose results were released in May 2019 at the annual meeting of the American College of Cardiology, found that, compared with other glucose-lowering drugs, taking an SGLT2 inhibitor was associated with lower risks of heart failure hospitalization and all-cause mortality (HR, 0.54; 95% CI, 0.44-0.65; P less than .001). This trial also offered a new detail: The patients gained the benefit regardless of whether their baseline left ventricular ejection fraction was preserved or reduced (J Coll Cardiol. 2019 Mar;73[9]:suppl 1). The SGLT2 inhibitors used in this trial included dapagliflozin (Farxiga) and empagliflozin (Jardiance).
In another study released this year, a subanalysis of the DECLARE-TIMI 58 trial, researchers reported that the SGLT2 inhibitor dapagliflozin reduced risks of both major adverse cardiovascular events and heart failure hospitalization in the subset of patients with type 2 diabetes and prior myocardial infarction versus controls (Circulation. 2019 May 28;139[22]:2516-27). The absolute risk reduction for major adverse cardiovascular events was 1.9% (HR, 0.81; 95% CI, 0.65-1.00; P = .046), while it was 0.6% for heart failure hospitalization (HR, 0.85; 95% CI, 0.72-1.00; P = .055).
These and other studies “speak volumes about the efficacy of managing blood sugar and addressing our biggest nemesis, which is cardiovascular disease,” Dr. Roberts said. “It’s irrefutable. The data [are] very good.”
Dr. Paauw said an SGLT2 inhibitor or GLP-1 receptor agonist is best reserved for use in select patients with cardiovascular risks and type 2 diabetes that need management beyond metformin.
For example, they might fit a 70-year-old with persistent hypertension who’s already taking a couple of blood pressure medications. “If they have another cardiovascular risk factor, the cardiovascular protection piece will be a bigger deal,” he said. Also, “it will probably help lower their blood pressure so they can avoid taking another blood pressure medicine.”
Trials of both GLP-1 receptor agonists and SGLT2 inhibitors have shown benefits “in improving [major adverse cardiac events], with the SGLT2 class showing substantial benefit in improving both heart failure and renal outcomes as well,” noted Dr. Skolnik. “It is in this context that one must address the question of whether the price of the medications are worthwhile. With such substantial benefit, there is no question in my mind that – for patients who have underlying cardiovascular illness, which includes patients with existent coronary disease, history of stroke, transient ischemic attack, or peripheral vascular disease – it is far and away worth it to prescribe these classes of medications.”
Indeed, the American Diabetes Association and the European Association for the Study of Diabetes’ most recent guidelines now call for a GLP-1 receptor agonist – instead of insulin – to be the first injectable used to treat type 2 diabetes (Diabetes Care 2018 Dec; 41[12]:2669-701).
“For the relatively small number of my patients who have been able to access and use these medications for months or longer, more have tolerated the GLP-1 agonists than SGLT2 inhibitors primarily due to urinary issues,” noted Dr. Hopkins.
Dipeptidyl peptidase–4 inhibitors are another option in patients with type 2 diabetes, but research suggests they may not be a top option for patients with cardiovascular risk. A 2018 review noted that cardiovascular outcome trials for alogliptin (Nesina), saxagliptin (Onglyza), and sitagliptin (Januvia) showed noninferiority but failed to demonstrate any superiority, compared with placebo in patients with type 2 diabetes mellitus and high cardiovascular risk (Circ Res. 2018 May 11;122[10]:1439-59).
The combination therapies
Many of the newer drugs are available as combinations with other types of diabetes drugs. In some cases, physicians create their own form of combination therapy by separately prescribing two or more diabetes drugs. Earlier this year, a study suggested the benefits of this kind of add-on therapy: Diabetes outcomes improved in patients who took the GLP-1 receptor agonist semaglutide and an SGLT2 inhibitor (Lancet Diabetes Endocrinol. 2019 Mar 1. doi: 10.1016/S2213-8587[19]30066-X).
Dr. Roberts suggested caution, however, when prescribing combination therapies. “My recommendation is always to begin with the individual medications to see if the patient tolerates the drugs and then decide which component needs to be titrated. It’s hard to titrate a combination drug, and it doesn’t leave a lot of flexibility. You never know which drug is doing what.
Dr. Handelsman said some patients may need to take three medications such as metformin, an SGLT2 inhibitor, and a GLP-1 receptor agonist.
“I don’t recommend using the combinations if you’re not familiar with the drugs ... These are relatively new pharmaceuticals, and most of us are on a learning curve as to how they fit into the armamentarium. If a drug is tolerated with a good response, you can certainly consider going to the combination tablets,” he added.
There is at least one drug that combines these three classes: The newly FDA-approved Qternmet XR, which combines dapagliflozin (an SGLT2 inhibitor), saxagliptin (a GLP-1 receptor agonist), and metformin. As of mid-June 2019, it was not yet available in the United States. Its sister drug Qtern, which combines dapagliflozin and saxagliptin, costs more than $500 a month with a free coupon, according to goodrx.com. In contrast, metformin is extremely inexpensive, costing just a few dollars a month for a common starting dose.
What about adding insulin?
“Both [SGLT2 inhibitors and GLP-1 receptor agonists] work very well with insulin,” Dr. Handelsman said. “There is a nice additive effect on the reduction of [hemoglobin] A1c. The only caution is that, although neither SGLT2 inhibitors nor GLP-1 receptor agonists cause hypoglycemia, in combination with insulin they do increase the risk of hypoglycemia. You may have to adjust the dose of insulin.”
Dr. Hopkins warned that cost becomes an even bigger issue when you add insulin into the mix.
“When insulin comes into the discussion, we are again stuck with astronomical costs which many struggle to afford,” he explained.
Indeed, the price tag on these drugs seems to be the biggest problem physicians have with them.
“The challenges in managing patients with diabetes aren’t the risks associated with the drugs. It’s dealing with their insurers,” noted Dr. Roberts.
Dr. Hopkins, Dr. Paauw, Dr. Roberts, and Dr. Syed reported no disclosures. Dr. Buse is an investigator for Johnson and Johnson. Dr. Goldschmidt is paid to speak by Novartis. Dr. Handelsman reported research grants, consulting work, and speaker honoraria from Amgen, Gilead, Lilly, Merck, Novo Nordisk, and others. Dr Skolnik reported nonfinancial support from AstraZeneca, Boehringer Ingelheim, Sanofi, and GlaxoSmithKline and personal fees from AstraZeneca, Boehringer Ingelheim, and Eli Lilly. He also serves on the advisory boards of AstraZeneca, Boehringer Ingelheim, Teva Pharmaceutical, Eli Lilly, Sanofi, Janssen Pharmaceuticals, Intarcia, Mylan, and GlaxoSmithKline.
Dr. Paauw and Dr. Skolnik are columnists for Family Practice News and Internal Medicine News.
M. Alexander Otto contributed to this report.

The options for treating type 2 diabetes without insulin have grown beyond metformin to include a long list of sodium-glucose cotransporter 2 (SGLT2) inhibitors and glucagonlike peptide–1 (GLP-1) receptor agonists that can be taken with or without metformin. These new drugs have cardiovascular and kidney benefits and help with weight loss, but they also carry risks and, according to some experts, their costs can be prohibitively expensive.
Given the medical community’s long-term experience with treating patients with metformin, and metformin’s lower cost, most of the physicians interviewed for this article advise using SGLT2 inhibitors and GLP-1 receptor agonists as second-line treatments. Others said that they would prefer to use the newer drugs as first-line therapies in select high-risk patients, but prior authorization hurdles created by insurance companies make that approach too burdensome.
“The economics of U.S. health care is stacked against many of our patients with diabetes in the current era,” Robert H. Hopkins Jr., MD, said in an interview.
Even when their insurance approves the drugs, patients still may not be able to afford the copay, explained Dr. Hopkins, professor of internal medicine and pediatrics and director of the division of general internal medicine at the University of Arkansas for Medical Sciences, Little Rock. “Sometimes patients can purchase drugs at a lower cost than the copay to purchase with the ‘drug coverage’ in their insurance plan – unfortunately, this is not the case with the newer diabetes medications we are discussing here.”
“SGLT2 inhibitors and GLP-1 agonists can cost several hundred dollars a month, and insurers often balk at paying for them. They’ll say, ‘Have you tried metformin?’ ” explained endocrinologist Victor Lawrence Roberts, MD, in a interview. “We have to work with insurance companies the best we can in a stepwise fashion.”
According to Dr. Roberts, 80% of his patients with diabetes struggle with the cost of medicine in general. “They’re either underinsured or not insured or their formulary is limited.
Douglas S. Paauw, MD, agreed in an interview that the newer drugs can be problematic on the insurance front.
“For some patients they aren’t affordable, especially for the uninsured if you can’t get them on an assistance program,” said Dr. Paauw, who is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the university.
Dr. Hopkins, who is on the Internal Medicine News board, noted that “unfortunately, the treatment of type 2 diabetes in patients who cannot achieve control with metformin, diet, weight control, and exercise is a story of the ‘haves’ and the ‘have nots.’ The ‘haves’ are those who have pharmacy benefits which make access to newer agents like SGLT2 inhibitors and GLP-1 agonists a possibility.”
“I have had very few of the ‘have nots’ who have been able to even consider these newer agents, which carry price tags of $600-$1,300 a month even with the availability of discounting coupons in the marketplace,” he added. “Most of these patients end up requiring a sulfonylurea or TZD [thiazolidinedione] as a second agent to achieve glycemic control. This makes it very difficult to achieve sufficient weight and metabolic control to avoid an eventual switch to insulin.”
Fatima Z. Syed, MD, an endocrine-trained general internist at DukeHealth in Durham, N.C., said she prescribes SGLT2 inhibitors and GLP-1 receptor agonists in combination with metformin. “I prescribe them frequently, but they are not first-line treatments,” she explained.
“Nothing replaces diet and exercise” as therapy for patients with type 2 diabetes, she added.
Neil S. Skolnik, MD, said that insurance companies were not preventing patients from using these drugs in his experience. He also provided an optimistic take on the accessibility of these drugs in the near future.
“Most insurance companies are now covering select SGLT2 inhibitors and GLP-1 receptor agonists for appropriate patients and those companies that currently do not will soon have to,” said Dr. Skolnik, who is a professor of family and community medicine at Jefferson Medical College, Philadelphia, and an associate director of the family medicine residency program at Abington (Pa.) Jefferson Health.
“The outcomes [associated with use of the new drugs] are robust, the benefits are large, and are well worth the cost,” he added.
The side effects
While others praised these drugs for their beneficial effects, they also noted that the side effects of these drugs are serious and must be discussed with patients.
GLP-1 receptor agonists are linked to gastrointestinal symptoms, especially nausea, while SGLT2 inhibitors have been linked to kidney failure, ketoacidosis, and more. The Food and Drug Administration warned in 2018 that the SGLT2 inhibitors can cause a rare serious infection known as Fournier’s gangrene – necrotizing fasciitis of the perineum.
“We have to tell our patients to let us know right away if they get pain or swelling in the genital area,” Dr. Paauw, who is on the Internal Medicine News board, noted. “The chance that an infection could explode quickly is higher in those who take these drugs.”
Amputation risks also are associated with taking the SGLT2 inhibitor canagliflozin (Invokana). The FDA requires the manufacturer of this drug to include a black-box warning about the risk of “lower-limb amputations, most frequently of the toe and midfoot,” but also the leg. In approval trials, the risk doubled versus placebo.
These amputation risks “put a damper on some of the enthusiasm on behalf of physicians and patients ... for taking this drug,” noted Dr. Roberts, who is a professor of internal medicine at the University of Central Florida, Orlando.
While a manufacturer-funded study released last year found no link to amputations, the results weren’t powerful enough to rule out a moderately increased risk.
“[If] you are at high risk for having an amputation, we really have to take this risk very seriously,” said John B. Buse, MD, chief of the division of endocrinology at the University of North Carolina at Chapel Hill, in a presentation about the study at the 2018 annual scientific sessions of the American Diabetes Association.
The benefits
Despite these risks of adverse events, most interviewed agreed that the many benefits observed in those taking SGLT2 inhibitors or GLP-1 receptor agonists make them worth prescribing, at least to those who are able to afford them.
Both SGLT2 inhibitors and GLP-1 receptor agonists appear to have significant cardiovascular benefits. A 2019 meta-analysis and systematic review found that both drugs reduced major adverse cardiac events by about 12% (Circulation. 2019 Apr 23;139[17]:2022-31).
“They don’t cause hypoglycemia, they lower blood pressure, they don’t cause weight gain, and they might promote weight loss,” noted Dr. Paauw.
SGLT2 inhibitors also have shown signs of kidney benefits. The CREDENCE trial linked canagliflozin to a lowering of kidney disorders versus placebo (N Engl J Med. 2019 Jun 13;380[24]:2295-306). “The relative risk of the renal-specific composite of end-stage kidney disease, a doubling of the creatinine level, or death from renal causes was lower by 34% (hazard ratio, 0.66; 95% confidence interval, 0.53-0.81; P less than .001), and the relative risk of end-stage kidney disease was lower by 32% (HR, 0.68; 95% CI, 0.54-0.86; P = .002),” the trial investigators wrote.
“They showed very nicely that the drug improved the kidney function of those patients and reduced the kidney deterioration,” said Yehuda Handelsman, MD, an endocrinologist in Tarzana, Calif., who chaired the 2011 and 2015 American Association of Clinical Endocrinologists’ Comprehensive Diabetes Guidelines. The study was especially impressive, he added, because it included patients with low kidney function.
SGLT2 inhibitors’ “diuretic mechanism explains why there is a substantial reduction in heart failure hospitalizations in patients who take these drugs,” said cardiologist Marc E. Goldschmidt, MD, director of the Heart Success Program at Atlantic Health System’s Morristown (N.J.) Medical Center, in an interview. “Both the EMPA-REG Outcome and the CREDENCE trials demonstrated substantial benefit of this class of medications by showing a lower risk of cardiovascular death as well as death from any cause and a lower risk of hospitalization for heart failure."
Overall, the SGLT2 trial data have been very consistent with a benefit for cardiovascular risk reduction, particularly in regard to heart failure hospitalizations and even in potentially preventing heart failure in diabetics,” he added.
Dr. Skolnik, a columnist for Family Practice News, cited SGLT2 inhibitors and GLP-1 receptor agonists’ ability to slow renal disease progression, promote weight loss, and prevent poor cardiac outcomes.“These drugs should be used, in addition to metformin, in all patients with diabetes and vascular disease. These proven outcomes are far better than we ever were able to achieve previously and the strength of the evidence at this point is very strong,” said Dr. Skolnik. “In addition to the benefits of decreasing the development of cardiovascular disease, serious heart failure, and slowing progression of renal disease, these two classes of medication have additional benefits. Both classes help patients lose weight, which is very different from what was found with either sulfonylureas or insulin, which cause patients to gain weight. Also both the SGLT2 inhibitors and the GLP-1 RAs [receptor agonists] have a low incidence of hypoglycemia. For all these reasons, these have become important medications for us to use in primary care.”
Other recent trials offer “very powerful data” about SGLT2 inhibitors, Dr. Roberts said. That’s good news, since “our approach needs to be toward cardiovascular protection and preservation as well as managing blood sugar.”An Israeli trial, whose results were released in May 2019 at the annual meeting of the American College of Cardiology, found that, compared with other glucose-lowering drugs, taking an SGLT2 inhibitor was associated with lower risks of heart failure hospitalization and all-cause mortality (HR, 0.54; 95% CI, 0.44-0.65; P less than .001). This trial also offered a new detail: The patients gained the benefit regardless of whether their baseline left ventricular ejection fraction was preserved or reduced (J Coll Cardiol. 2019 Mar;73[9]:suppl 1). The SGLT2 inhibitors used in this trial included dapagliflozin (Farxiga) and empagliflozin (Jardiance).
In another study released this year, a subanalysis of the DECLARE-TIMI 58 trial, researchers reported that the SGLT2 inhibitor dapagliflozin reduced risks of both major adverse cardiovascular events and heart failure hospitalization in the subset of patients with type 2 diabetes and prior myocardial infarction versus controls (Circulation. 2019 May 28;139[22]:2516-27). The absolute risk reduction for major adverse cardiovascular events was 1.9% (HR, 0.81; 95% CI, 0.65-1.00; P = .046), while it was 0.6% for heart failure hospitalization (HR, 0.85; 95% CI, 0.72-1.00; P = .055).
These and other studies “speak volumes about the efficacy of managing blood sugar and addressing our biggest nemesis, which is cardiovascular disease,” Dr. Roberts said. “It’s irrefutable. The data [are] very good.”
Dr. Paauw said an SGLT2 inhibitor or GLP-1 receptor agonist is best reserved for use in select patients with cardiovascular risks and type 2 diabetes that need management beyond metformin.
For example, they might fit a 70-year-old with persistent hypertension who’s already taking a couple of blood pressure medications. “If they have another cardiovascular risk factor, the cardiovascular protection piece will be a bigger deal,” he said. Also, “it will probably help lower their blood pressure so they can avoid taking another blood pressure medicine.”
Trials of both GLP-1 receptor agonists and SGLT2 inhibitors have shown benefits “in improving [major adverse cardiac events], with the SGLT2 class showing substantial benefit in improving both heart failure and renal outcomes as well,” noted Dr. Skolnik. “It is in this context that one must address the question of whether the price of the medications are worthwhile. With such substantial benefit, there is no question in my mind that – for patients who have underlying cardiovascular illness, which includes patients with existent coronary disease, history of stroke, transient ischemic attack, or peripheral vascular disease – it is far and away worth it to prescribe these classes of medications.”
Indeed, the American Diabetes Association and the European Association for the Study of Diabetes’ most recent guidelines now call for a GLP-1 receptor agonist – instead of insulin – to be the first injectable used to treat type 2 diabetes (Diabetes Care 2018 Dec; 41[12]:2669-701).
“For the relatively small number of my patients who have been able to access and use these medications for months or longer, more have tolerated the GLP-1 agonists than SGLT2 inhibitors primarily due to urinary issues,” noted Dr. Hopkins.
Dipeptidyl peptidase–4 inhibitors are another option in patients with type 2 diabetes, but research suggests they may not be a top option for patients with cardiovascular risk. A 2018 review noted that cardiovascular outcome trials for alogliptin (Nesina), saxagliptin (Onglyza), and sitagliptin (Januvia) showed noninferiority but failed to demonstrate any superiority, compared with placebo in patients with type 2 diabetes mellitus and high cardiovascular risk (Circ Res. 2018 May 11;122[10]:1439-59).
The combination therapies
Many of the newer drugs are available as combinations with other types of diabetes drugs. In some cases, physicians create their own form of combination therapy by separately prescribing two or more diabetes drugs. Earlier this year, a study suggested the benefits of this kind of add-on therapy: Diabetes outcomes improved in patients who took the GLP-1 receptor agonist semaglutide and an SGLT2 inhibitor (Lancet Diabetes Endocrinol. 2019 Mar 1. doi: 10.1016/S2213-8587[19]30066-X).
Dr. Roberts suggested caution, however, when prescribing combination therapies. “My recommendation is always to begin with the individual medications to see if the patient tolerates the drugs and then decide which component needs to be titrated. It’s hard to titrate a combination drug, and it doesn’t leave a lot of flexibility. You never know which drug is doing what.
Dr. Handelsman said some patients may need to take three medications such as metformin, an SGLT2 inhibitor, and a GLP-1 receptor agonist.
“I don’t recommend using the combinations if you’re not familiar with the drugs ... These are relatively new pharmaceuticals, and most of us are on a learning curve as to how they fit into the armamentarium. If a drug is tolerated with a good response, you can certainly consider going to the combination tablets,” he added.
There is at least one drug that combines these three classes: The newly FDA-approved Qternmet XR, which combines dapagliflozin (an SGLT2 inhibitor), saxagliptin (a GLP-1 receptor agonist), and metformin. As of mid-June 2019, it was not yet available in the United States. Its sister drug Qtern, which combines dapagliflozin and saxagliptin, costs more than $500 a month with a free coupon, according to goodrx.com. In contrast, metformin is extremely inexpensive, costing just a few dollars a month for a common starting dose.
What about adding insulin?
“Both [SGLT2 inhibitors and GLP-1 receptor agonists] work very well with insulin,” Dr. Handelsman said. “There is a nice additive effect on the reduction of [hemoglobin] A1c. The only caution is that, although neither SGLT2 inhibitors nor GLP-1 receptor agonists cause hypoglycemia, in combination with insulin they do increase the risk of hypoglycemia. You may have to adjust the dose of insulin.”
Dr. Hopkins warned that cost becomes an even bigger issue when you add insulin into the mix.
“When insulin comes into the discussion, we are again stuck with astronomical costs which many struggle to afford,” he explained.
Indeed, the price tag on these drugs seems to be the biggest problem physicians have with them.
“The challenges in managing patients with diabetes aren’t the risks associated with the drugs. It’s dealing with their insurers,” noted Dr. Roberts.
Dr. Hopkins, Dr. Paauw, Dr. Roberts, and Dr. Syed reported no disclosures. Dr. Buse is an investigator for Johnson and Johnson. Dr. Goldschmidt is paid to speak by Novartis. Dr. Handelsman reported research grants, consulting work, and speaker honoraria from Amgen, Gilead, Lilly, Merck, Novo Nordisk, and others. Dr Skolnik reported nonfinancial support from AstraZeneca, Boehringer Ingelheim, Sanofi, and GlaxoSmithKline and personal fees from AstraZeneca, Boehringer Ingelheim, and Eli Lilly. He also serves on the advisory boards of AstraZeneca, Boehringer Ingelheim, Teva Pharmaceutical, Eli Lilly, Sanofi, Janssen Pharmaceuticals, Intarcia, Mylan, and GlaxoSmithKline.
Dr. Paauw and Dr. Skolnik are columnists for Family Practice News and Internal Medicine News.
M. Alexander Otto contributed to this report.

















