User login
FDA approves label extension for dapagliflozin
The Food and Drug Administration has approved a label extension for Farxiga (dapagliflozin) and Xigduo XR (extended-release dapagliflozin and metformin HCl) for use in patients with type 2 diabetes and moderate renal impairment, lowering the estimated glomerular filtration rate (eGFR) threshold to 45 mL/min per 1.73 m2 from the current60 mL/min per 1.73 m2.
The update is based on results from DERIVE, a phase 3 study in patients with inadequately controlled diabetes and an eGFR of 45-59 mL/min per 1.73 m2 who received either dapagliflozin 10 mg or placebo during a 24-week period. After that time, patients who received dapagliflozin had significant reductions in glycosylated hemoglobin, compared with placebo. The safety profile was similar to that in other studies with dapagliflozin.
The most common adverse events associated with Farxiga are female genital mycotic infections, nasopharyngitis, and urinary tract infections. For Xigduo XR, the most common adverse events are female genital mycotic infection, nasopharyngitis, urinary tract infection, diarrhea, and headache.
“The DERIVE study, which further confirmed the well-established efficacy and safety profile for Farxiga and Xigduo XR, has resulted in important label changes for patients with type 2 diabetes that enable a broader population with impaired renal function to potentially benefit from these important treatment options,” Jim McDermott, PhD, vice president, U.S. medical affairs, diabetes, at AstraZeneca, said in the press release.
Find the full press release on the AstraZeneca website.
The Food and Drug Administration has approved a label extension for Farxiga (dapagliflozin) and Xigduo XR (extended-release dapagliflozin and metformin HCl) for use in patients with type 2 diabetes and moderate renal impairment, lowering the estimated glomerular filtration rate (eGFR) threshold to 45 mL/min per 1.73 m2 from the current60 mL/min per 1.73 m2.
The update is based on results from DERIVE, a phase 3 study in patients with inadequately controlled diabetes and an eGFR of 45-59 mL/min per 1.73 m2 who received either dapagliflozin 10 mg or placebo during a 24-week period. After that time, patients who received dapagliflozin had significant reductions in glycosylated hemoglobin, compared with placebo. The safety profile was similar to that in other studies with dapagliflozin.
The most common adverse events associated with Farxiga are female genital mycotic infections, nasopharyngitis, and urinary tract infections. For Xigduo XR, the most common adverse events are female genital mycotic infection, nasopharyngitis, urinary tract infection, diarrhea, and headache.
“The DERIVE study, which further confirmed the well-established efficacy and safety profile for Farxiga and Xigduo XR, has resulted in important label changes for patients with type 2 diabetes that enable a broader population with impaired renal function to potentially benefit from these important treatment options,” Jim McDermott, PhD, vice president, U.S. medical affairs, diabetes, at AstraZeneca, said in the press release.
Find the full press release on the AstraZeneca website.
The Food and Drug Administration has approved a label extension for Farxiga (dapagliflozin) and Xigduo XR (extended-release dapagliflozin and metformin HCl) for use in patients with type 2 diabetes and moderate renal impairment, lowering the estimated glomerular filtration rate (eGFR) threshold to 45 mL/min per 1.73 m2 from the current60 mL/min per 1.73 m2.
The update is based on results from DERIVE, a phase 3 study in patients with inadequately controlled diabetes and an eGFR of 45-59 mL/min per 1.73 m2 who received either dapagliflozin 10 mg or placebo during a 24-week period. After that time, patients who received dapagliflozin had significant reductions in glycosylated hemoglobin, compared with placebo. The safety profile was similar to that in other studies with dapagliflozin.
The most common adverse events associated with Farxiga are female genital mycotic infections, nasopharyngitis, and urinary tract infections. For Xigduo XR, the most common adverse events are female genital mycotic infection, nasopharyngitis, urinary tract infection, diarrhea, and headache.
“The DERIVE study, which further confirmed the well-established efficacy and safety profile for Farxiga and Xigduo XR, has resulted in important label changes for patients with type 2 diabetes that enable a broader population with impaired renal function to potentially benefit from these important treatment options,” Jim McDermott, PhD, vice president, U.S. medical affairs, diabetes, at AstraZeneca, said in the press release.
Find the full press release on the AstraZeneca website.
ICU admissions raise chronic condition risk
SAN DIEGO – The research showed rising likelihood of conditions such as depression, diabetes, and heart disease.
By merging two existing databases, the researchers were able to capture a more comprehensive picture of post-ICU patients. “We were able to include almost the entire country,” Ilse van Beusekom, a PhD candidate in health sciences at the University of Amsterdam and data manager at the National Intensive Care Evaluation (NICE) foundation, said in an interview.
Ms. van Beusekom presented the study at the Critical Care Congress sponsored by the Society of Critical Care Medicine. The study was simultaneously published in Critical Care Medicine.
The work compared 56,760 ICU survivors from 81 facilities across the Netherlands to 75,232 age-, sex-, and socioeconomic status–matched controls. The mean age was 65 years and 60% of the population was male. “The types of chronic conditions are the same, only the prevalences are different,” said Ms. van Beusekom.
The researchers compared chronic conditions in the year before ICU admission and the year after, based on data pulled from the NICE national quality database, which includes data describing the first 24 hours of ICU admission, and the Vektis insurance claims database, which includes information on medical treatment. Before ICU admission, 45% of the ICU population was free of chronic conditions, as were 62% of controls. One chronic condition was present in 36% of ICU patients, versus 29% of controls, and two or more conditions were present in 19% versus 9% of controls.
The ICU population was more likely to have high cholesterol (16% vs. 14%), heart disease (14% vs. 6%), chronic obstructive pulmonary disease (8% vs. 3%), type II diabetes (8% vs. 6%), type I diabetes (6% vs. 3%), and depression (6% vs. 4%).
The ICU population also was at greater risk of developing one or more new chronic conditions during the year following their stay. The risk was three- to fourfold higher throughout age ranges.
The study suggests the need for greater follow-up after an ICU admission in order to help patients cope with lingering problems. Ms. van Beusekom noted that there are follow-up programs in the Netherlands for several patient groups, but none for ICU survivors. One possibility would be to have the patient return to the ICU 3 months or so after release to discuss their diagnosis, treatment, and any lingering concerns. “A lot of people don’t know that their complaints are linked with the ICU visit,” said Ms. van Beusekom.
Timothy G. Buchman, MD, professor of surgery at Emory University, Atlanta, who moderated the session, wondered why the ICU seems to be an inflection point for developing new chronic conditions. Could it simply be because patients are sicker to begin with and have reached an inflection point of their illness, or could the treatments in ICU be contributing to or exposing those conditions? Ms. van Beusekom believed it was likely a combination of factors, and she referred to data she had not presented showing that even control patients who had been to the hospital (though not the ICU) during the study period were at lower risk of new chronic conditions than ICU patients.
Ms. van Beusekom’s group plans to investigate ICU-related variables that might be associated with risk of chronic conditions.
The study was not funded. Ms. van Beusekom had no relevant disclosures.
SOURCE: van Beusekom I et al. CCC48, Abstract Crit Care Med. 2019;47:324-30.
SAN DIEGO – The research showed rising likelihood of conditions such as depression, diabetes, and heart disease.
By merging two existing databases, the researchers were able to capture a more comprehensive picture of post-ICU patients. “We were able to include almost the entire country,” Ilse van Beusekom, a PhD candidate in health sciences at the University of Amsterdam and data manager at the National Intensive Care Evaluation (NICE) foundation, said in an interview.
Ms. van Beusekom presented the study at the Critical Care Congress sponsored by the Society of Critical Care Medicine. The study was simultaneously published in Critical Care Medicine.
The work compared 56,760 ICU survivors from 81 facilities across the Netherlands to 75,232 age-, sex-, and socioeconomic status–matched controls. The mean age was 65 years and 60% of the population was male. “The types of chronic conditions are the same, only the prevalences are different,” said Ms. van Beusekom.
The researchers compared chronic conditions in the year before ICU admission and the year after, based on data pulled from the NICE national quality database, which includes data describing the first 24 hours of ICU admission, and the Vektis insurance claims database, which includes information on medical treatment. Before ICU admission, 45% of the ICU population was free of chronic conditions, as were 62% of controls. One chronic condition was present in 36% of ICU patients, versus 29% of controls, and two or more conditions were present in 19% versus 9% of controls.
The ICU population was more likely to have high cholesterol (16% vs. 14%), heart disease (14% vs. 6%), chronic obstructive pulmonary disease (8% vs. 3%), type II diabetes (8% vs. 6%), type I diabetes (6% vs. 3%), and depression (6% vs. 4%).
The ICU population also was at greater risk of developing one or more new chronic conditions during the year following their stay. The risk was three- to fourfold higher throughout age ranges.
The study suggests the need for greater follow-up after an ICU admission in order to help patients cope with lingering problems. Ms. van Beusekom noted that there are follow-up programs in the Netherlands for several patient groups, but none for ICU survivors. One possibility would be to have the patient return to the ICU 3 months or so after release to discuss their diagnosis, treatment, and any lingering concerns. “A lot of people don’t know that their complaints are linked with the ICU visit,” said Ms. van Beusekom.
Timothy G. Buchman, MD, professor of surgery at Emory University, Atlanta, who moderated the session, wondered why the ICU seems to be an inflection point for developing new chronic conditions. Could it simply be because patients are sicker to begin with and have reached an inflection point of their illness, or could the treatments in ICU be contributing to or exposing those conditions? Ms. van Beusekom believed it was likely a combination of factors, and she referred to data she had not presented showing that even control patients who had been to the hospital (though not the ICU) during the study period were at lower risk of new chronic conditions than ICU patients.
Ms. van Beusekom’s group plans to investigate ICU-related variables that might be associated with risk of chronic conditions.
The study was not funded. Ms. van Beusekom had no relevant disclosures.
SOURCE: van Beusekom I et al. CCC48, Abstract Crit Care Med. 2019;47:324-30.
SAN DIEGO – The research showed rising likelihood of conditions such as depression, diabetes, and heart disease.
By merging two existing databases, the researchers were able to capture a more comprehensive picture of post-ICU patients. “We were able to include almost the entire country,” Ilse van Beusekom, a PhD candidate in health sciences at the University of Amsterdam and data manager at the National Intensive Care Evaluation (NICE) foundation, said in an interview.
Ms. van Beusekom presented the study at the Critical Care Congress sponsored by the Society of Critical Care Medicine. The study was simultaneously published in Critical Care Medicine.
The work compared 56,760 ICU survivors from 81 facilities across the Netherlands to 75,232 age-, sex-, and socioeconomic status–matched controls. The mean age was 65 years and 60% of the population was male. “The types of chronic conditions are the same, only the prevalences are different,” said Ms. van Beusekom.
The researchers compared chronic conditions in the year before ICU admission and the year after, based on data pulled from the NICE national quality database, which includes data describing the first 24 hours of ICU admission, and the Vektis insurance claims database, which includes information on medical treatment. Before ICU admission, 45% of the ICU population was free of chronic conditions, as were 62% of controls. One chronic condition was present in 36% of ICU patients, versus 29% of controls, and two or more conditions were present in 19% versus 9% of controls.
The ICU population was more likely to have high cholesterol (16% vs. 14%), heart disease (14% vs. 6%), chronic obstructive pulmonary disease (8% vs. 3%), type II diabetes (8% vs. 6%), type I diabetes (6% vs. 3%), and depression (6% vs. 4%).
The ICU population also was at greater risk of developing one or more new chronic conditions during the year following their stay. The risk was three- to fourfold higher throughout age ranges.
The study suggests the need for greater follow-up after an ICU admission in order to help patients cope with lingering problems. Ms. van Beusekom noted that there are follow-up programs in the Netherlands for several patient groups, but none for ICU survivors. One possibility would be to have the patient return to the ICU 3 months or so after release to discuss their diagnosis, treatment, and any lingering concerns. “A lot of people don’t know that their complaints are linked with the ICU visit,” said Ms. van Beusekom.
Timothy G. Buchman, MD, professor of surgery at Emory University, Atlanta, who moderated the session, wondered why the ICU seems to be an inflection point for developing new chronic conditions. Could it simply be because patients are sicker to begin with and have reached an inflection point of their illness, or could the treatments in ICU be contributing to or exposing those conditions? Ms. van Beusekom believed it was likely a combination of factors, and she referred to data she had not presented showing that even control patients who had been to the hospital (though not the ICU) during the study period were at lower risk of new chronic conditions than ICU patients.
Ms. van Beusekom’s group plans to investigate ICU-related variables that might be associated with risk of chronic conditions.
The study was not funded. Ms. van Beusekom had no relevant disclosures.
SOURCE: van Beusekom I et al. CCC48, Abstract Crit Care Med. 2019;47:324-30.
REPORTING FROM CCC48
Brilinta reduces MACE in THEMIS trial
The maker of ticagrelor has released top-line results from the phase 3 trial known as THEMIS.
AstraZeneca announced that THEMIS met its primary endpoint of reduction of major adverse cardiovascular events (MACE) among patients with coronary artery disease (CAD) and type 2 diabetes with no history of heart attack or stroke. (MACE is a composite of cardiovascular death, heart attack, and stroke.)
The trial (NCT01991795) explored these risks by comparing ticagrelor (Brilinta) plus aspirin versus aspirin alone. The oral, reversibly binding, direct-acting P2Y12 receptor antagonist is currently indicated for reducing MACE among patients with acute coronary syndrome or a history of MI. According to the press release, the multinational, randomized, double-blind trial is exploring the use of ticagrelor/aspirin among patients with CAD and type 2 diabetes because of this population’s high risk and the lack of treatment options.
Gabriel Steg, MD, THEMIS cochair and professor at Université Paris–Diderot, said in the release that “patients who have both stable coronary artery disease and diabetes are a sizable group which remains at particularly high risk of major adverse cardiac events. The optimal long-term antiplatelet therapy in that group is not fully established.” He added that the full results from the THEMIS trial will be presented later this year.
Ticagrelor comes with risks of significant and sometimes fatal bleeding; as such it is contraindicated for patients with pathological bleeding risk or history of intracranial hemorrhage. Its use is also discouraged among patients with severe hepatic impairment and in patients who are breastfeeding. Although usually self-limiting when related to ticagrelor use, dyspnea was reported in about 14% of patients taking the drug. Dyspnea and bleeding were among the most common adverse reactions seen with ticagrelor.
The maker of ticagrelor has released top-line results from the phase 3 trial known as THEMIS.
AstraZeneca announced that THEMIS met its primary endpoint of reduction of major adverse cardiovascular events (MACE) among patients with coronary artery disease (CAD) and type 2 diabetes with no history of heart attack or stroke. (MACE is a composite of cardiovascular death, heart attack, and stroke.)
The trial (NCT01991795) explored these risks by comparing ticagrelor (Brilinta) plus aspirin versus aspirin alone. The oral, reversibly binding, direct-acting P2Y12 receptor antagonist is currently indicated for reducing MACE among patients with acute coronary syndrome or a history of MI. According to the press release, the multinational, randomized, double-blind trial is exploring the use of ticagrelor/aspirin among patients with CAD and type 2 diabetes because of this population’s high risk and the lack of treatment options.
Gabriel Steg, MD, THEMIS cochair and professor at Université Paris–Diderot, said in the release that “patients who have both stable coronary artery disease and diabetes are a sizable group which remains at particularly high risk of major adverse cardiac events. The optimal long-term antiplatelet therapy in that group is not fully established.” He added that the full results from the THEMIS trial will be presented later this year.
Ticagrelor comes with risks of significant and sometimes fatal bleeding; as such it is contraindicated for patients with pathological bleeding risk or history of intracranial hemorrhage. Its use is also discouraged among patients with severe hepatic impairment and in patients who are breastfeeding. Although usually self-limiting when related to ticagrelor use, dyspnea was reported in about 14% of patients taking the drug. Dyspnea and bleeding were among the most common adverse reactions seen with ticagrelor.
The maker of ticagrelor has released top-line results from the phase 3 trial known as THEMIS.
AstraZeneca announced that THEMIS met its primary endpoint of reduction of major adverse cardiovascular events (MACE) among patients with coronary artery disease (CAD) and type 2 diabetes with no history of heart attack or stroke. (MACE is a composite of cardiovascular death, heart attack, and stroke.)
The trial (NCT01991795) explored these risks by comparing ticagrelor (Brilinta) plus aspirin versus aspirin alone. The oral, reversibly binding, direct-acting P2Y12 receptor antagonist is currently indicated for reducing MACE among patients with acute coronary syndrome or a history of MI. According to the press release, the multinational, randomized, double-blind trial is exploring the use of ticagrelor/aspirin among patients with CAD and type 2 diabetes because of this population’s high risk and the lack of treatment options.
Gabriel Steg, MD, THEMIS cochair and professor at Université Paris–Diderot, said in the release that “patients who have both stable coronary artery disease and diabetes are a sizable group which remains at particularly high risk of major adverse cardiac events. The optimal long-term antiplatelet therapy in that group is not fully established.” He added that the full results from the THEMIS trial will be presented later this year.
Ticagrelor comes with risks of significant and sometimes fatal bleeding; as such it is contraindicated for patients with pathological bleeding risk or history of intracranial hemorrhage. Its use is also discouraged among patients with severe hepatic impairment and in patients who are breastfeeding. Although usually self-limiting when related to ticagrelor use, dyspnea was reported in about 14% of patients taking the drug. Dyspnea and bleeding were among the most common adverse reactions seen with ticagrelor.
Paclitaxel drug-coated balloons appear safe for PAD treatment
Mortality after the use of drug-coated balloons (DCB) for femoropopliteal peripheral arterial disease (PAD) was not correlated with paclitaxel exposure, according to the results of a meta-analysis of 5-year outcomes, according to a report published online.
“Paclitaxel DCBs are safe and effective to treat the symptoms of [Rutherford classification categories] 2-4 femoropopliteal PAD,” according to Peter A. Schneider, MD, of Hawaii Permanente Medical Group, Kaiser Foundation Hospital, Honolulu, and his coauthors.
Their study analyzed data pooled from five clinical trials NCT01175850, NCT01566461, NCT01947478, NCT02118532, and NCT01609296, comprising 1,980 patients from a variety of ethnic populations with Rutherford classification 2-4 disease.
Among these patients, 1,837 received DCB and 143 received uncoated percutaneous transluminal angioplasty (PTA). The mean age of the overall cohort was 68.5 years; 68.4% of patients were men. Baseline characteristics were similar between groups. However, patients treated with a DCB were more likely to have critical limb ischemia, compared with PTA. DCB subjects were less likely to have hyperlipidemia, coronary artery disease, and diabetes mellitus than were those treated with uncoated PTA. In addition, PTA patients who died were more likely to be active smokers than were DCB patients that died.
There was no statistically significant difference in all-cause mortality between DCB and PTA through 5 years (9.3% vs 11.2%, respectively, P = .399).
A Kaplan-Meier survival analysis stratified paclitaxel dosage into three groups: low-dose, mid-dose, and upper-dose groups. Mean dosages for the three groups were 5,019, 10,008, and 19,978 mcg, respectively. The analysis showed no significant difference in mortality between groups, “demonstrating no direct impact of levels of nominal paclitaxel dose exposure at the index procedure and survival status in the DCB patients through 5 years (P = .700),” according to the authors.
Limitations of the study reported by the authors include the fact that pooling data from distinct trials has shortcomings. Some of data included had not yet undergone peer review, and PTA patients were included in only two randomized trials in a 2:1 ratio.
“The small numbers of PTA control patients (less than 10%) may not be representative of PTA patients in general and limits the strength of this analysis of mortality.” In addition, only patients with Rutherford classification 2-4 were included in these studies.
“Results from this independent patient-level meta-analysis show no difference in mortality between DCB and PTA at 5 years and no correlation between varying levels of paclitaxel exposure and mortality. ... Data transparency and additional analyses are needed to better understand how other factors influence long-term outcomes in this complex patient population,” the researchers concluded.
The study was funded by Medtronic, which provided the data for independent analysis to the Baim Institute for Clinical Research. Dr. Schneider is a member of the advisory board for Medtronic, Abbott, and Boston Scientific and is a consultant for Medtronic and other device companies. Coauthors had consulting, advisory board, or honoraria relationships with Medtronic and other device companies.
SOURCE: Schneider PA et al. JACC 2019 Jan 25. doi: 10.1016/j.jacc.2019.01.013.
Mortality after the use of drug-coated balloons (DCB) for femoropopliteal peripheral arterial disease (PAD) was not correlated with paclitaxel exposure, according to the results of a meta-analysis of 5-year outcomes, according to a report published online.
“Paclitaxel DCBs are safe and effective to treat the symptoms of [Rutherford classification categories] 2-4 femoropopliteal PAD,” according to Peter A. Schneider, MD, of Hawaii Permanente Medical Group, Kaiser Foundation Hospital, Honolulu, and his coauthors.
Their study analyzed data pooled from five clinical trials NCT01175850, NCT01566461, NCT01947478, NCT02118532, and NCT01609296, comprising 1,980 patients from a variety of ethnic populations with Rutherford classification 2-4 disease.
Among these patients, 1,837 received DCB and 143 received uncoated percutaneous transluminal angioplasty (PTA). The mean age of the overall cohort was 68.5 years; 68.4% of patients were men. Baseline characteristics were similar between groups. However, patients treated with a DCB were more likely to have critical limb ischemia, compared with PTA. DCB subjects were less likely to have hyperlipidemia, coronary artery disease, and diabetes mellitus than were those treated with uncoated PTA. In addition, PTA patients who died were more likely to be active smokers than were DCB patients that died.
There was no statistically significant difference in all-cause mortality between DCB and PTA through 5 years (9.3% vs 11.2%, respectively, P = .399).
A Kaplan-Meier survival analysis stratified paclitaxel dosage into three groups: low-dose, mid-dose, and upper-dose groups. Mean dosages for the three groups were 5,019, 10,008, and 19,978 mcg, respectively. The analysis showed no significant difference in mortality between groups, “demonstrating no direct impact of levels of nominal paclitaxel dose exposure at the index procedure and survival status in the DCB patients through 5 years (P = .700),” according to the authors.
Limitations of the study reported by the authors include the fact that pooling data from distinct trials has shortcomings. Some of data included had not yet undergone peer review, and PTA patients were included in only two randomized trials in a 2:1 ratio.
“The small numbers of PTA control patients (less than 10%) may not be representative of PTA patients in general and limits the strength of this analysis of mortality.” In addition, only patients with Rutherford classification 2-4 were included in these studies.
“Results from this independent patient-level meta-analysis show no difference in mortality between DCB and PTA at 5 years and no correlation between varying levels of paclitaxel exposure and mortality. ... Data transparency and additional analyses are needed to better understand how other factors influence long-term outcomes in this complex patient population,” the researchers concluded.
The study was funded by Medtronic, which provided the data for independent analysis to the Baim Institute for Clinical Research. Dr. Schneider is a member of the advisory board for Medtronic, Abbott, and Boston Scientific and is a consultant for Medtronic and other device companies. Coauthors had consulting, advisory board, or honoraria relationships with Medtronic and other device companies.
SOURCE: Schneider PA et al. JACC 2019 Jan 25. doi: 10.1016/j.jacc.2019.01.013.
Mortality after the use of drug-coated balloons (DCB) for femoropopliteal peripheral arterial disease (PAD) was not correlated with paclitaxel exposure, according to the results of a meta-analysis of 5-year outcomes, according to a report published online.
“Paclitaxel DCBs are safe and effective to treat the symptoms of [Rutherford classification categories] 2-4 femoropopliteal PAD,” according to Peter A. Schneider, MD, of Hawaii Permanente Medical Group, Kaiser Foundation Hospital, Honolulu, and his coauthors.
Their study analyzed data pooled from five clinical trials NCT01175850, NCT01566461, NCT01947478, NCT02118532, and NCT01609296, comprising 1,980 patients from a variety of ethnic populations with Rutherford classification 2-4 disease.
Among these patients, 1,837 received DCB and 143 received uncoated percutaneous transluminal angioplasty (PTA). The mean age of the overall cohort was 68.5 years; 68.4% of patients were men. Baseline characteristics were similar between groups. However, patients treated with a DCB were more likely to have critical limb ischemia, compared with PTA. DCB subjects were less likely to have hyperlipidemia, coronary artery disease, and diabetes mellitus than were those treated with uncoated PTA. In addition, PTA patients who died were more likely to be active smokers than were DCB patients that died.
There was no statistically significant difference in all-cause mortality between DCB and PTA through 5 years (9.3% vs 11.2%, respectively, P = .399).
A Kaplan-Meier survival analysis stratified paclitaxel dosage into three groups: low-dose, mid-dose, and upper-dose groups. Mean dosages for the three groups were 5,019, 10,008, and 19,978 mcg, respectively. The analysis showed no significant difference in mortality between groups, “demonstrating no direct impact of levels of nominal paclitaxel dose exposure at the index procedure and survival status in the DCB patients through 5 years (P = .700),” according to the authors.
Limitations of the study reported by the authors include the fact that pooling data from distinct trials has shortcomings. Some of data included had not yet undergone peer review, and PTA patients were included in only two randomized trials in a 2:1 ratio.
“The small numbers of PTA control patients (less than 10%) may not be representative of PTA patients in general and limits the strength of this analysis of mortality.” In addition, only patients with Rutherford classification 2-4 were included in these studies.
“Results from this independent patient-level meta-analysis show no difference in mortality between DCB and PTA at 5 years and no correlation between varying levels of paclitaxel exposure and mortality. ... Data transparency and additional analyses are needed to better understand how other factors influence long-term outcomes in this complex patient population,” the researchers concluded.
The study was funded by Medtronic, which provided the data for independent analysis to the Baim Institute for Clinical Research. Dr. Schneider is a member of the advisory board for Medtronic, Abbott, and Boston Scientific and is a consultant for Medtronic and other device companies. Coauthors had consulting, advisory board, or honoraria relationships with Medtronic and other device companies.
SOURCE: Schneider PA et al. JACC 2019 Jan 25. doi: 10.1016/j.jacc.2019.01.013.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Diabetes and the Commercial Motor Vehicle Driver
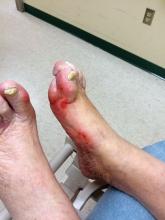
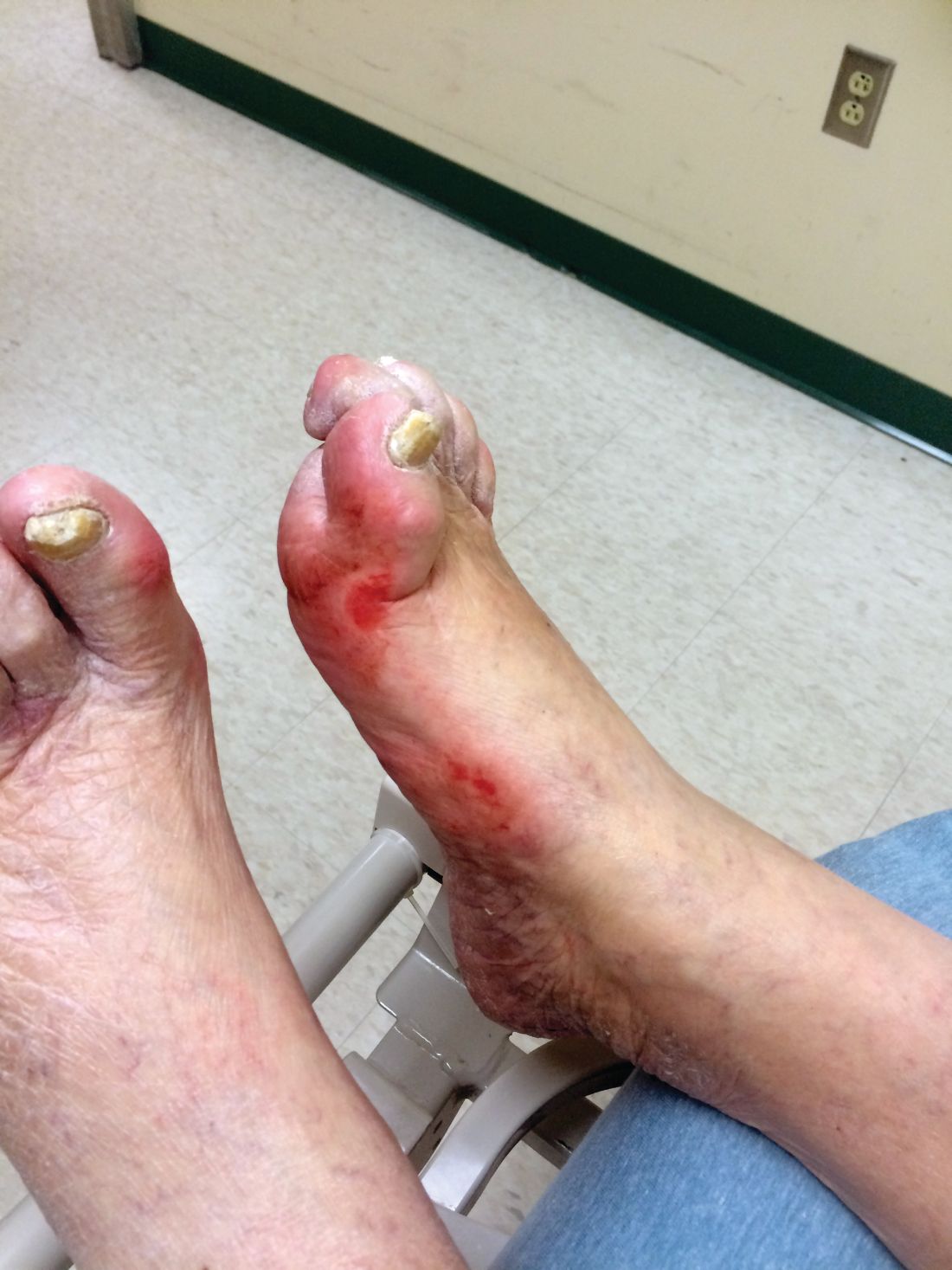
A 60-year-old man is sent by his new employer to your urgent care for a pre-employment Department of Transportation (DOT) physical to obtain clearance to drive a commercial motor vehicle. His medical history is significant for hypertension, for which he takes lisinopril. Otherwise, he is healthy, with normal vital signs. His physical exam is unremarkable, but the urine sample is notably positive for glucose. A fingerstick glucose test yields a measurement of 212 m
Commercial motor vehicle (CMV) drivers are mandated by the Federal Motor Carrier Safety Administration (FMCSA) to receive a DOT physical examination by a licensed medical examiner. To qualify to perform the exam, physician assistants, advanced practice nurses, physicians, and chiropractors must complete an educational program and pass a written certification examination.1 Subsequently, the examiners are placed on a national registry—the National Registry of Certified Medical Examiners—with the mission to improve highway safety by determining whether a CMV driver’s health meets standards and guidelines set by the FMCSA.2
Under current guidelines, a DOT physical exam for a healthy CMV driver is considered valid for a maximum of 24 months. However, some diseases and medications require frequent follow-up, which can shorten the length of time a driver can be medically cleared to operate a CMV. Furthermore, certain conditions can disqualify the driver from meeting the necessary standards required for medical certification.
This case presentation offers the opportunity to review the requirements for evaluation and certification of a CMV driver with new-onset hyperglycemia and, ultimately, diabetes. In the United States, types 1 and 2 diabetes are estimated to affect 30.3 million people.3 About 33% of CMV drivers have been diagnosed with diabetes, which is significant since research has demonstrated an increased risk for crashes in individuals with diabetes, due to potential incapacitation from hypoglycemia.4-6
Thus, for practitioners and medical examiners, it is prudent to screen and manage diabetes in CMV drivers. In fact, over the past 15 years, federal regulations have stipulated that any driver with diabetes requiring insulin for control was disqualified from this type of work.7 This standard was developed in response to the increased risk for hypoglycemic reactions with the use of insulin. However, in September 2018, the FMCSA revised this regulation, permitting individuals with a stable insulin regimen and properly controlled diabetes to be qualified to operate a CMV. As a result, for drivers requiring insulin, the treating clinician must complete a standardized form within 45 days of the DOT exam, documenting management of the patient’s diabetes.8 For drivers with diabetes who do not require insulin, determinations are made on a case-by-case basis, with discernment of the driver’s ability to manage the disease and concurrently meet other standards for qualification.
HEALTH HISTORY AND EXAMINATION
Each CMV driver completes a standard medical history form that asks about specific medications, surgeries, or medical conditions, including diabetes or blood glucose problems. Subsequently, the driver and, ultimately, the medical examiner must expand upon and discuss every “yes” response to this questionnaire.
Regarding diabetes, the examiner should determine whether the disease is controlled by diet, pills, and/or insulin, with clarification of the doses, frequency, and prescriber. In addition, the examiner should review and document glucose control, blood glucose monitoring, history of hypoglycemic episodes, and episodes of fainting, dizziness, or loss of consciousness.7
Continue to: The physical exam should focus on...
The physical exam should focus on identifying signs of complications from diabetes, such as retinopathy, nephropathy, or peripheral neuropathy. At each certification visit, the examiner should assess the patient’s height and weight, BMI, vision, hearing, blood pressure, and heart rate, and perform urinalysis to screen for proteinuria or glycosuria. A fingerstick test to obtain a random blood glucose reading is often performed in a driver with glycosuria.
Likewise, the A1C level should be documented in every patient with new-onset or known diabetes, with the recommendation from the FMCSA that a level >10% is an indicator of poor glucose control.7 It is important to note that an A1C level up to 10% is not the glycemic target recognized by the American Diabetes Association and the American Association of Clinical Endocrinologists. The FMCSA is focused more on hypoglycemic concerns than on providing management guidelines.
DETERMINING CERTIFICATION
Currently, the recertification time recommended for CMV drivers with diabetes and documented glucose control is 1 year. This is based on the assumption that the driver is under medical care with a treatment plan and that he/she is not currently experiencing any complications from the disease. Furthermore, insulin secretagogues (eg, sulfonylureas) can be used for glucose control as long as adverse effects (eg, hypoglycemia) do not interfere with safe driving. However, the FMCSA does not recommend certifying any driver who
- In the past 12 months has experienced a hypoglycemic reaction resulting in seizure; loss of consciousness; need of assistance from another person; or period of impaired cognitive function that occurred without warning.
- In the past 5 years has had recurring (≥ 2) disqualifying hypoglycemic reactions.
- Has received a formal diagnosis of peripheral neuropathy, loss of position, or pedal sensation.
- Has resting tachycardia or orthostatic hypotension.
- Has severe diabetic nephropathy requiring dialysis.
- Has severe nonproliferative or proliferative retinopathy.8
In drivers with new-onset hyperglycemia, it is appropriate for the medical examiner to refer the driver to his/her primary care provider for further testing (eg, A1C), determination of treatment, a copy of the diabetes medical standard for driving, and written opinion of the driver’s medical fitness for duty. Subsequently, the medical examiner can utilize this information from the primary care provider to determine certification for the driver. While there are no specific guidelines on the waiting period for certification, the driver should demonstrate glucose control with treatment that is adequate, effective, safe, and stable.7
Overall, while living with diabetes can be challenging, patients who demonstrate control of the disease can maintain their occupation as a CMV driver. The role of the medical examiner is to evaluate the driver’s risk to safely operate a CMV—in particular, considering the possibilities of a severe hypoglycemic episode or target organ dysfunction—whereas the clinician treating the driver’s diabetes is focused on minimizing the complications associated with hyperglycemia.
Continue to: As a reminder...
As a reminder, due to the progressive nature of the disease, recertification is recommended annually for drivers.7 Nevertheless, it is reassuring that the DOT has implemented safeguards designed to keep our citizens safe while travelling the highways and byways of the United States.
Given the patient’s elevated glucose, more information is needed to safely provide clearance for driving a CMV. The patient would be disqualified until he could provide documentation of glucose control. Therefore, this patient would benefit from a referral to his primary care provider to obtain a list of medications used to manage his disease, documentation of an A1C level <10% and no evidence of complications from diabetes, and a written opinion from the primary care provider indicating the driver is medically fit for duty. Accordingly, the primary care provider can ensure the patient demonstrates compliance in managing diabetes and can safely operate a CMV.
1. Federal Motor Carrier Safety Administration. DOT Medical Exam and Commercial Motor Vehicle Certification. www.fmcsa.dot.gov/medical/driver-medical-requirements/dot-medical-exam-and-commercial-motor-vehicle-certification. Accessed February 22, 2019.
2. Federal Motor Carrier Safety Administration. National Registry of Certified Medical Examiners. www.fmcsa.dot.gov/medical/driver-medical-requirements/national-registry-certified-medical-examiners. Accessed February 22, 2019.
3. CDC. National Diabetes Statistics Report, 2017: estimates of diabetes and its burden in the United States. www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed February 22, 2019.
4. Abu Dabrh AM, Firwana B, Cowl CT, et al. Health assessment of commercial drivers: a meta-narrative systematic review. BMJ Open. 2014;4:e003434.
5. Laberge-Nadeau C, Dionne G, Maag U, et al. Medical conditions and the severity of commercial motor vehicle drivers’ road accidents. Accid Anal Prev. 1996;28:43-51.
6. Redelmeier DA, Kenshole AB, Ray JG. Motor vehicle crashes in diabetic patients with tight glycemic control: a population-based case control analysis. PLoS Med. 2009;6:e1000192.
7. Federal Motor Carrier Safety Administration. Medical Examiner Handbook. www.fmcsa.dot.gov/sites/fmcsa.dot.gov/files/docs/mission/advisory-committees/mrb/83401/fmcsamedicalexaminerhandbook.pdf. Accessed February 22, 2019.
8. Federal Motor Carrier Safety Administration. Qualifications of Drivers; Diabetes Standard. Federal Register. September 19, 2018. www.federalregister.gov/documents/2018/09/19/2018-20161/qualifications-of-drivers-diabetes-standard. Accessed February 25, 2019.
A 60-year-old man is sent by his new employer to your urgent care for a pre-employment Department of Transportation (DOT) physical to obtain clearance to drive a commercial motor vehicle. His medical history is significant for hypertension, for which he takes lisinopril. Otherwise, he is healthy, with normal vital signs. His physical exam is unremarkable, but the urine sample is notably positive for glucose. A fingerstick glucose test yields a measurement of 212 m
Commercial motor vehicle (CMV) drivers are mandated by the Federal Motor Carrier Safety Administration (FMCSA) to receive a DOT physical examination by a licensed medical examiner. To qualify to perform the exam, physician assistants, advanced practice nurses, physicians, and chiropractors must complete an educational program and pass a written certification examination.1 Subsequently, the examiners are placed on a national registry—the National Registry of Certified Medical Examiners—with the mission to improve highway safety by determining whether a CMV driver’s health meets standards and guidelines set by the FMCSA.2
Under current guidelines, a DOT physical exam for a healthy CMV driver is considered valid for a maximum of 24 months. However, some diseases and medications require frequent follow-up, which can shorten the length of time a driver can be medically cleared to operate a CMV. Furthermore, certain conditions can disqualify the driver from meeting the necessary standards required for medical certification.
This case presentation offers the opportunity to review the requirements for evaluation and certification of a CMV driver with new-onset hyperglycemia and, ultimately, diabetes. In the United States, types 1 and 2 diabetes are estimated to affect 30.3 million people.3 About 33% of CMV drivers have been diagnosed with diabetes, which is significant since research has demonstrated an increased risk for crashes in individuals with diabetes, due to potential incapacitation from hypoglycemia.4-6
Thus, for practitioners and medical examiners, it is prudent to screen and manage diabetes in CMV drivers. In fact, over the past 15 years, federal regulations have stipulated that any driver with diabetes requiring insulin for control was disqualified from this type of work.7 This standard was developed in response to the increased risk for hypoglycemic reactions with the use of insulin. However, in September 2018, the FMCSA revised this regulation, permitting individuals with a stable insulin regimen and properly controlled diabetes to be qualified to operate a CMV. As a result, for drivers requiring insulin, the treating clinician must complete a standardized form within 45 days of the DOT exam, documenting management of the patient’s diabetes.8 For drivers with diabetes who do not require insulin, determinations are made on a case-by-case basis, with discernment of the driver’s ability to manage the disease and concurrently meet other standards for qualification.
HEALTH HISTORY AND EXAMINATION
Each CMV driver completes a standard medical history form that asks about specific medications, surgeries, or medical conditions, including diabetes or blood glucose problems. Subsequently, the driver and, ultimately, the medical examiner must expand upon and discuss every “yes” response to this questionnaire.
Regarding diabetes, the examiner should determine whether the disease is controlled by diet, pills, and/or insulin, with clarification of the doses, frequency, and prescriber. In addition, the examiner should review and document glucose control, blood glucose monitoring, history of hypoglycemic episodes, and episodes of fainting, dizziness, or loss of consciousness.7
Continue to: The physical exam should focus on...
The physical exam should focus on identifying signs of complications from diabetes, such as retinopathy, nephropathy, or peripheral neuropathy. At each certification visit, the examiner should assess the patient’s height and weight, BMI, vision, hearing, blood pressure, and heart rate, and perform urinalysis to screen for proteinuria or glycosuria. A fingerstick test to obtain a random blood glucose reading is often performed in a driver with glycosuria.
Likewise, the A1C level should be documented in every patient with new-onset or known diabetes, with the recommendation from the FMCSA that a level >10% is an indicator of poor glucose control.7 It is important to note that an A1C level up to 10% is not the glycemic target recognized by the American Diabetes Association and the American Association of Clinical Endocrinologists. The FMCSA is focused more on hypoglycemic concerns than on providing management guidelines.
DETERMINING CERTIFICATION
Currently, the recertification time recommended for CMV drivers with diabetes and documented glucose control is 1 year. This is based on the assumption that the driver is under medical care with a treatment plan and that he/she is not currently experiencing any complications from the disease. Furthermore, insulin secretagogues (eg, sulfonylureas) can be used for glucose control as long as adverse effects (eg, hypoglycemia) do not interfere with safe driving. However, the FMCSA does not recommend certifying any driver who
- In the past 12 months has experienced a hypoglycemic reaction resulting in seizure; loss of consciousness; need of assistance from another person; or period of impaired cognitive function that occurred without warning.
- In the past 5 years has had recurring (≥ 2) disqualifying hypoglycemic reactions.
- Has received a formal diagnosis of peripheral neuropathy, loss of position, or pedal sensation.
- Has resting tachycardia or orthostatic hypotension.
- Has severe diabetic nephropathy requiring dialysis.
- Has severe nonproliferative or proliferative retinopathy.8
In drivers with new-onset hyperglycemia, it is appropriate for the medical examiner to refer the driver to his/her primary care provider for further testing (eg, A1C), determination of treatment, a copy of the diabetes medical standard for driving, and written opinion of the driver’s medical fitness for duty. Subsequently, the medical examiner can utilize this information from the primary care provider to determine certification for the driver. While there are no specific guidelines on the waiting period for certification, the driver should demonstrate glucose control with treatment that is adequate, effective, safe, and stable.7
Overall, while living with diabetes can be challenging, patients who demonstrate control of the disease can maintain their occupation as a CMV driver. The role of the medical examiner is to evaluate the driver’s risk to safely operate a CMV—in particular, considering the possibilities of a severe hypoglycemic episode or target organ dysfunction—whereas the clinician treating the driver’s diabetes is focused on minimizing the complications associated with hyperglycemia.
Continue to: As a reminder...
As a reminder, due to the progressive nature of the disease, recertification is recommended annually for drivers.7 Nevertheless, it is reassuring that the DOT has implemented safeguards designed to keep our citizens safe while travelling the highways and byways of the United States.
Given the patient’s elevated glucose, more information is needed to safely provide clearance for driving a CMV. The patient would be disqualified until he could provide documentation of glucose control. Therefore, this patient would benefit from a referral to his primary care provider to obtain a list of medications used to manage his disease, documentation of an A1C level <10% and no evidence of complications from diabetes, and a written opinion from the primary care provider indicating the driver is medically fit for duty. Accordingly, the primary care provider can ensure the patient demonstrates compliance in managing diabetes and can safely operate a CMV.
A 60-year-old man is sent by his new employer to your urgent care for a pre-employment Department of Transportation (DOT) physical to obtain clearance to drive a commercial motor vehicle. His medical history is significant for hypertension, for which he takes lisinopril. Otherwise, he is healthy, with normal vital signs. His physical exam is unremarkable, but the urine sample is notably positive for glucose. A fingerstick glucose test yields a measurement of 212 m
Commercial motor vehicle (CMV) drivers are mandated by the Federal Motor Carrier Safety Administration (FMCSA) to receive a DOT physical examination by a licensed medical examiner. To qualify to perform the exam, physician assistants, advanced practice nurses, physicians, and chiropractors must complete an educational program and pass a written certification examination.1 Subsequently, the examiners are placed on a national registry—the National Registry of Certified Medical Examiners—with the mission to improve highway safety by determining whether a CMV driver’s health meets standards and guidelines set by the FMCSA.2
Under current guidelines, a DOT physical exam for a healthy CMV driver is considered valid for a maximum of 24 months. However, some diseases and medications require frequent follow-up, which can shorten the length of time a driver can be medically cleared to operate a CMV. Furthermore, certain conditions can disqualify the driver from meeting the necessary standards required for medical certification.
This case presentation offers the opportunity to review the requirements for evaluation and certification of a CMV driver with new-onset hyperglycemia and, ultimately, diabetes. In the United States, types 1 and 2 diabetes are estimated to affect 30.3 million people.3 About 33% of CMV drivers have been diagnosed with diabetes, which is significant since research has demonstrated an increased risk for crashes in individuals with diabetes, due to potential incapacitation from hypoglycemia.4-6
Thus, for practitioners and medical examiners, it is prudent to screen and manage diabetes in CMV drivers. In fact, over the past 15 years, federal regulations have stipulated that any driver with diabetes requiring insulin for control was disqualified from this type of work.7 This standard was developed in response to the increased risk for hypoglycemic reactions with the use of insulin. However, in September 2018, the FMCSA revised this regulation, permitting individuals with a stable insulin regimen and properly controlled diabetes to be qualified to operate a CMV. As a result, for drivers requiring insulin, the treating clinician must complete a standardized form within 45 days of the DOT exam, documenting management of the patient’s diabetes.8 For drivers with diabetes who do not require insulin, determinations are made on a case-by-case basis, with discernment of the driver’s ability to manage the disease and concurrently meet other standards for qualification.
HEALTH HISTORY AND EXAMINATION
Each CMV driver completes a standard medical history form that asks about specific medications, surgeries, or medical conditions, including diabetes or blood glucose problems. Subsequently, the driver and, ultimately, the medical examiner must expand upon and discuss every “yes” response to this questionnaire.
Regarding diabetes, the examiner should determine whether the disease is controlled by diet, pills, and/or insulin, with clarification of the doses, frequency, and prescriber. In addition, the examiner should review and document glucose control, blood glucose monitoring, history of hypoglycemic episodes, and episodes of fainting, dizziness, or loss of consciousness.7
Continue to: The physical exam should focus on...
The physical exam should focus on identifying signs of complications from diabetes, such as retinopathy, nephropathy, or peripheral neuropathy. At each certification visit, the examiner should assess the patient’s height and weight, BMI, vision, hearing, blood pressure, and heart rate, and perform urinalysis to screen for proteinuria or glycosuria. A fingerstick test to obtain a random blood glucose reading is often performed in a driver with glycosuria.
Likewise, the A1C level should be documented in every patient with new-onset or known diabetes, with the recommendation from the FMCSA that a level >10% is an indicator of poor glucose control.7 It is important to note that an A1C level up to 10% is not the glycemic target recognized by the American Diabetes Association and the American Association of Clinical Endocrinologists. The FMCSA is focused more on hypoglycemic concerns than on providing management guidelines.
DETERMINING CERTIFICATION
Currently, the recertification time recommended for CMV drivers with diabetes and documented glucose control is 1 year. This is based on the assumption that the driver is under medical care with a treatment plan and that he/she is not currently experiencing any complications from the disease. Furthermore, insulin secretagogues (eg, sulfonylureas) can be used for glucose control as long as adverse effects (eg, hypoglycemia) do not interfere with safe driving. However, the FMCSA does not recommend certifying any driver who
- In the past 12 months has experienced a hypoglycemic reaction resulting in seizure; loss of consciousness; need of assistance from another person; or period of impaired cognitive function that occurred without warning.
- In the past 5 years has had recurring (≥ 2) disqualifying hypoglycemic reactions.
- Has received a formal diagnosis of peripheral neuropathy, loss of position, or pedal sensation.
- Has resting tachycardia or orthostatic hypotension.
- Has severe diabetic nephropathy requiring dialysis.
- Has severe nonproliferative or proliferative retinopathy.8
In drivers with new-onset hyperglycemia, it is appropriate for the medical examiner to refer the driver to his/her primary care provider for further testing (eg, A1C), determination of treatment, a copy of the diabetes medical standard for driving, and written opinion of the driver’s medical fitness for duty. Subsequently, the medical examiner can utilize this information from the primary care provider to determine certification for the driver. While there are no specific guidelines on the waiting period for certification, the driver should demonstrate glucose control with treatment that is adequate, effective, safe, and stable.7
Overall, while living with diabetes can be challenging, patients who demonstrate control of the disease can maintain their occupation as a CMV driver. The role of the medical examiner is to evaluate the driver’s risk to safely operate a CMV—in particular, considering the possibilities of a severe hypoglycemic episode or target organ dysfunction—whereas the clinician treating the driver’s diabetes is focused on minimizing the complications associated with hyperglycemia.
Continue to: As a reminder...
As a reminder, due to the progressive nature of the disease, recertification is recommended annually for drivers.7 Nevertheless, it is reassuring that the DOT has implemented safeguards designed to keep our citizens safe while travelling the highways and byways of the United States.
Given the patient’s elevated glucose, more information is needed to safely provide clearance for driving a CMV. The patient would be disqualified until he could provide documentation of glucose control. Therefore, this patient would benefit from a referral to his primary care provider to obtain a list of medications used to manage his disease, documentation of an A1C level <10% and no evidence of complications from diabetes, and a written opinion from the primary care provider indicating the driver is medically fit for duty. Accordingly, the primary care provider can ensure the patient demonstrates compliance in managing diabetes and can safely operate a CMV.
1. Federal Motor Carrier Safety Administration. DOT Medical Exam and Commercial Motor Vehicle Certification. www.fmcsa.dot.gov/medical/driver-medical-requirements/dot-medical-exam-and-commercial-motor-vehicle-certification. Accessed February 22, 2019.
2. Federal Motor Carrier Safety Administration. National Registry of Certified Medical Examiners. www.fmcsa.dot.gov/medical/driver-medical-requirements/national-registry-certified-medical-examiners. Accessed February 22, 2019.
3. CDC. National Diabetes Statistics Report, 2017: estimates of diabetes and its burden in the United States. www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed February 22, 2019.
4. Abu Dabrh AM, Firwana B, Cowl CT, et al. Health assessment of commercial drivers: a meta-narrative systematic review. BMJ Open. 2014;4:e003434.
5. Laberge-Nadeau C, Dionne G, Maag U, et al. Medical conditions and the severity of commercial motor vehicle drivers’ road accidents. Accid Anal Prev. 1996;28:43-51.
6. Redelmeier DA, Kenshole AB, Ray JG. Motor vehicle crashes in diabetic patients with tight glycemic control: a population-based case control analysis. PLoS Med. 2009;6:e1000192.
7. Federal Motor Carrier Safety Administration. Medical Examiner Handbook. www.fmcsa.dot.gov/sites/fmcsa.dot.gov/files/docs/mission/advisory-committees/mrb/83401/fmcsamedicalexaminerhandbook.pdf. Accessed February 22, 2019.
8. Federal Motor Carrier Safety Administration. Qualifications of Drivers; Diabetes Standard. Federal Register. September 19, 2018. www.federalregister.gov/documents/2018/09/19/2018-20161/qualifications-of-drivers-diabetes-standard. Accessed February 25, 2019.
1. Federal Motor Carrier Safety Administration. DOT Medical Exam and Commercial Motor Vehicle Certification. www.fmcsa.dot.gov/medical/driver-medical-requirements/dot-medical-exam-and-commercial-motor-vehicle-certification. Accessed February 22, 2019.
2. Federal Motor Carrier Safety Administration. National Registry of Certified Medical Examiners. www.fmcsa.dot.gov/medical/driver-medical-requirements/national-registry-certified-medical-examiners. Accessed February 22, 2019.
3. CDC. National Diabetes Statistics Report, 2017: estimates of diabetes and its burden in the United States. www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed February 22, 2019.
4. Abu Dabrh AM, Firwana B, Cowl CT, et al. Health assessment of commercial drivers: a meta-narrative systematic review. BMJ Open. 2014;4:e003434.
5. Laberge-Nadeau C, Dionne G, Maag U, et al. Medical conditions and the severity of commercial motor vehicle drivers’ road accidents. Accid Anal Prev. 1996;28:43-51.
6. Redelmeier DA, Kenshole AB, Ray JG. Motor vehicle crashes in diabetic patients with tight glycemic control: a population-based case control analysis. PLoS Med. 2009;6:e1000192.
7. Federal Motor Carrier Safety Administration. Medical Examiner Handbook. www.fmcsa.dot.gov/sites/fmcsa.dot.gov/files/docs/mission/advisory-committees/mrb/83401/fmcsamedicalexaminerhandbook.pdf. Accessed February 22, 2019.
8. Federal Motor Carrier Safety Administration. Qualifications of Drivers; Diabetes Standard. Federal Register. September 19, 2018. www.federalregister.gov/documents/2018/09/19/2018-20161/qualifications-of-drivers-diabetes-standard. Accessed February 25, 2019.
Insulin-treated diabetes in pregnancy carries preterm risk
Women with insulin-treated diabetes are at significantly greater risk of preterm birth and of delivering babies who are large for gestational age (LGA), regardless of prepregnancy body weight, new findings suggest.
Researchers examined the role of maternal diabetes and weight on pregnancy outcomes in the population-based cohort study. The study comprised 649,043 live births in Finland between Jan. 1, 2004, and Dec. 31, 2014, including 4,000 in women with insulin-treated diabetes, 3,740 in women with type 2 diabetes, and 98,568 women with gestational diabetes.
Prepregnancy body mass index was normal for nearly 60% of mothers, while 4% were underweight, 21% were overweight, 8% were moderately obese, and 4% were severely obese.
Overall, the researchers found that women with insulin-treated diabetes had a 43-fold higher odds of having an LGA infant, compared with the reference group of women of normal BMI without diabetes (adjusted odds ratio [aOR], 43.80; 95% confidence interval, 40.88-46.93). And there was an 11-fold greater odds of having a preterm birth in this group (aOR, 11.17; 95% CI, 10.46-11.93).
The findings were published in JAMA Pediatrics.
“Smaller, but clearly statistically significant, increased LGA risks were found also for mothers with type 2 diabetes and gestational diabetes not treated with insulin, especially in combination with prepregnancy overweight or obesity that were stronger for type 2 diabetes than gestational diabetes,” wrote Linghua Kong, MSc, of the department of molecular medicine and surgery at Karolinska Institutet, and coauthors.
The aOR for LGA among women with type 2 diabetes was 9.57 (95% CI, 8.65-10.58), compared with the reference group. And for women with maternal gestational diabetes, the aOR for LGA was 3.80 (95% CI, 3.66-3.96).
Looking at the risk for preterm birth, the researchers found that the aOR among women with type 2 diabetes was 2.12 (95% CI, 1.90-2.36), while there was no association between gestational diabetes and preterm birth.
The researchers also reported that for women with gestational diabetes or no diabetes, the odds of preterm birth increased slightly as maternal prepregnancy BMI increased.
“Maternal glucose metabolism during pregnancy differs from that in the non-pregnant state; insulin resistance is increased, directing fat as the mother’s energy source to ensure adequate carbohydrate supply for the growing fetus,” the researchers wrote. “This increase in insulin resistance is mediated by a number of factors, such as increased levels of progesterone, estrogen, and human placental lactogen.”
The authors noted that their data did not include information on congenital anomalies, maternal complications such as preeclampsia, and grade of diabetes control during pregnancy. In addition, the data on maternal BMI was derived from a single time point.
“These findings may have implications for counseling and managing pregnancies to prevent adverse birth outcomes,” they wrote.
The study and some authors were supported by the THL National Institute for Health and Welfare, the Swedish Research Council, Stockholm County Council, the China Scholarship Council, and the Swedish Brain Foundation.
SOURCE: Kong L et al. JAMA Pediatr. 2019 Feb 25. doi: 10.1001/jamapediatrics.2018.5541.
Women with insulin-treated diabetes are at significantly greater risk of preterm birth and of delivering babies who are large for gestational age (LGA), regardless of prepregnancy body weight, new findings suggest.
Researchers examined the role of maternal diabetes and weight on pregnancy outcomes in the population-based cohort study. The study comprised 649,043 live births in Finland between Jan. 1, 2004, and Dec. 31, 2014, including 4,000 in women with insulin-treated diabetes, 3,740 in women with type 2 diabetes, and 98,568 women with gestational diabetes.
Prepregnancy body mass index was normal for nearly 60% of mothers, while 4% were underweight, 21% were overweight, 8% were moderately obese, and 4% were severely obese.
Overall, the researchers found that women with insulin-treated diabetes had a 43-fold higher odds of having an LGA infant, compared with the reference group of women of normal BMI without diabetes (adjusted odds ratio [aOR], 43.80; 95% confidence interval, 40.88-46.93). And there was an 11-fold greater odds of having a preterm birth in this group (aOR, 11.17; 95% CI, 10.46-11.93).
The findings were published in JAMA Pediatrics.
“Smaller, but clearly statistically significant, increased LGA risks were found also for mothers with type 2 diabetes and gestational diabetes not treated with insulin, especially in combination with prepregnancy overweight or obesity that were stronger for type 2 diabetes than gestational diabetes,” wrote Linghua Kong, MSc, of the department of molecular medicine and surgery at Karolinska Institutet, and coauthors.
The aOR for LGA among women with type 2 diabetes was 9.57 (95% CI, 8.65-10.58), compared with the reference group. And for women with maternal gestational diabetes, the aOR for LGA was 3.80 (95% CI, 3.66-3.96).
Looking at the risk for preterm birth, the researchers found that the aOR among women with type 2 diabetes was 2.12 (95% CI, 1.90-2.36), while there was no association between gestational diabetes and preterm birth.
The researchers also reported that for women with gestational diabetes or no diabetes, the odds of preterm birth increased slightly as maternal prepregnancy BMI increased.
“Maternal glucose metabolism during pregnancy differs from that in the non-pregnant state; insulin resistance is increased, directing fat as the mother’s energy source to ensure adequate carbohydrate supply for the growing fetus,” the researchers wrote. “This increase in insulin resistance is mediated by a number of factors, such as increased levels of progesterone, estrogen, and human placental lactogen.”
The authors noted that their data did not include information on congenital anomalies, maternal complications such as preeclampsia, and grade of diabetes control during pregnancy. In addition, the data on maternal BMI was derived from a single time point.
“These findings may have implications for counseling and managing pregnancies to prevent adverse birth outcomes,” they wrote.
The study and some authors were supported by the THL National Institute for Health and Welfare, the Swedish Research Council, Stockholm County Council, the China Scholarship Council, and the Swedish Brain Foundation.
SOURCE: Kong L et al. JAMA Pediatr. 2019 Feb 25. doi: 10.1001/jamapediatrics.2018.5541.
Women with insulin-treated diabetes are at significantly greater risk of preterm birth and of delivering babies who are large for gestational age (LGA), regardless of prepregnancy body weight, new findings suggest.
Researchers examined the role of maternal diabetes and weight on pregnancy outcomes in the population-based cohort study. The study comprised 649,043 live births in Finland between Jan. 1, 2004, and Dec. 31, 2014, including 4,000 in women with insulin-treated diabetes, 3,740 in women with type 2 diabetes, and 98,568 women with gestational diabetes.
Prepregnancy body mass index was normal for nearly 60% of mothers, while 4% were underweight, 21% were overweight, 8% were moderately obese, and 4% were severely obese.
Overall, the researchers found that women with insulin-treated diabetes had a 43-fold higher odds of having an LGA infant, compared with the reference group of women of normal BMI without diabetes (adjusted odds ratio [aOR], 43.80; 95% confidence interval, 40.88-46.93). And there was an 11-fold greater odds of having a preterm birth in this group (aOR, 11.17; 95% CI, 10.46-11.93).
The findings were published in JAMA Pediatrics.
“Smaller, but clearly statistically significant, increased LGA risks were found also for mothers with type 2 diabetes and gestational diabetes not treated with insulin, especially in combination with prepregnancy overweight or obesity that were stronger for type 2 diabetes than gestational diabetes,” wrote Linghua Kong, MSc, of the department of molecular medicine and surgery at Karolinska Institutet, and coauthors.
The aOR for LGA among women with type 2 diabetes was 9.57 (95% CI, 8.65-10.58), compared with the reference group. And for women with maternal gestational diabetes, the aOR for LGA was 3.80 (95% CI, 3.66-3.96).
Looking at the risk for preterm birth, the researchers found that the aOR among women with type 2 diabetes was 2.12 (95% CI, 1.90-2.36), while there was no association between gestational diabetes and preterm birth.
The researchers also reported that for women with gestational diabetes or no diabetes, the odds of preterm birth increased slightly as maternal prepregnancy BMI increased.
“Maternal glucose metabolism during pregnancy differs from that in the non-pregnant state; insulin resistance is increased, directing fat as the mother’s energy source to ensure adequate carbohydrate supply for the growing fetus,” the researchers wrote. “This increase in insulin resistance is mediated by a number of factors, such as increased levels of progesterone, estrogen, and human placental lactogen.”
The authors noted that their data did not include information on congenital anomalies, maternal complications such as preeclampsia, and grade of diabetes control during pregnancy. In addition, the data on maternal BMI was derived from a single time point.
“These findings may have implications for counseling and managing pregnancies to prevent adverse birth outcomes,” they wrote.
The study and some authors were supported by the THL National Institute for Health and Welfare, the Swedish Research Council, Stockholm County Council, the China Scholarship Council, and the Swedish Brain Foundation.
SOURCE: Kong L et al. JAMA Pediatr. 2019 Feb 25. doi: 10.1001/jamapediatrics.2018.5541.
FROM JAMA PEDIATRICS
Key clinical point:
Major finding: Pregnant women with insulin-treated diabetes have a 43-fold higher odds of having a child who is large for gestational age and 11-fold high risk for preterm birth.
Study details: A population-based cohort study of 649,043 live births in Finland between 2004 and 2014.
Disclosures: The study and some authors were supported by the THL National Institute for Health and Welfare, the Swedish Research Council, Stockholm County Council, the China Scholarship Council, and the Swedish Brain Foundation.
Source: Kong L et al. JAMA Pediatr. 2019 Feb 25. doi: 10.1001/jamapediatrics.2018.5541.
AHA: Consider obesity as CVD risk factor in children
The American Heart Association has included obesity and severe obesity in its updated scientific statement outlining risk factors and considerations for cardiovascular risk reduction in high-risk pediatric patients.
The scientific statement is an update to a 2006 American Heart Association (AHA) statement, adding details about obesity as an at-risk condition and severe obesity as a moderate-risk condition. Other additions include classifying type 2 diabetes as a high-risk condition and expanding on new risk factors for cardiovascular disease (CVD) among patients who received treatment for childhood cancer.
The AHA said the statement is aimed at pediatric cardiologists, primary care physicians, and subspecialists who care for at-risk pediatric patients, as well as providers who will care for these patients as they transition to adult life.
Obesity
In the AHA scientific statement, Sarah de Ferranti, MD, MPH, of Boston Children’s Hospital, chair of the writing group, and her colleagues, highlighted a 2016 study that identified a twofold to threefold higher risk of CVD-related mortality among patients who were overweight or obese, compared with patients of normal weight (Diabetes Care. 2016 Nov;39[11]:1996-2003).
Patients with obesity and severe obesity are at increased risk of aortic or coronary fatty streaks, dyslipidemia, high blood pressure, hyperglycemia, and insulin resistance, as well as inflammatory and oxidative stress, the AHA writing group noted.
They estimated that approximately 6% of U.S. children aged 2-19 years old are considered severely obese.
After identifying patients with obesity, the writing group said, a “multimodal and graduated approach to treatment” for these patients is generally warranted, with a focus on dietary and lifestyle changes, and use of pharmacotherapy and bariatric surgery if indicated.
However, the authors said therapeutic life change modification “is limited in severe obesity because of small effect size and difficulty with sustainability,” while use of pharmacotherapy for treatment of pediatric obesity remains understudied and medications such as orlistat and metformin offer only modest weight loss.
Bariatric surgery, “the only treatment for severe pediatric obesity consistently associated with clinically meaningful and durable weight loss,” is not consistently offered to patients under 12 years old, they added.
Diabetes
The AHA statement also addresses risks from type 1 (T1D) and type 2 diabetes (T2D). Children with T1D and T2D are at increased risk for dyslipidemia, hypertension, microalbuminuria, and obesity. Annual screening for these patients is indicated, and cardiovascular risk factor reduction can be achieved by managing hyperglycemia, controlling weight gain as a result of medication, and implementing therapeutic lifestyle changes, when possible.
Childhood cancer
As survival rates from childhood cancer have improved, there is a need to address the increased risk of cardiovascular-related mortality (estimated at 8-10 times higher than the general population) as well as cancer relapse, according to the writing group.
Among patients recruited to the Childhood Cancer Survivor Study, there was a 9-fold increase in cerebrovascular accident, 10-fold increased risk of coronary artery disease, and 15-fold increase in heart failure for childhood cancer survivors, compared with their siblings who were cancer free.
Cancer treatments such as radiation exposure are linked to increased rates of myocardial infarction, heart failure, valvular abnormalities, and pericardial disease at a twofold to sixfold higher rate when administered at a greater than 1,500 centigray dose, compared to cancer survivors who did not receive radiation, the authors wrote.
Anthracycline treatment is associated with a dose-dependent increase in the risk of dilated cardiomyopathy, while hematopoietic stem cell transplantation may increase the risk of CVD-related mortality from heart failure, cerebrovascular accident, cardiomyopathy, coronary artery disease, and rhythm disorders.
In treating childhood cancer survivors for CVD risk factors, “a low threshold should be used when considering the initiation of pharmacological agents because of the high risk of these youth,” and standard pharmacotherapies can be used, the authors said. “Treatment of cardiovascular risk factors should consider the cancer therapies the patient has received previously.”
In the AHA statement, Dr. de Ferranti and her colleagues also outlined epidemiology, screening, and treatment data for other cardiovascular risk factors such as familial hypercholesterolemia, Lipoprotein(a), hypertension, chronic kidney disease, congenital heart disease, Kawasaki disease, and heart transplantation.
Some members of the writing group reported research grants from Amgen, Sanofi, the Wisconsin Partnership Program, and the National Institutes of Health. One author reported unpaid consultancies with Novo Nordisk, Orexigen, and Vivus.
SOURCE: de Ferranti SD et al. Circulation. 2019 Feb 25. doi: 10.1161/CIR.0000000000000618.
The American Heart Association has included obesity and severe obesity in its updated scientific statement outlining risk factors and considerations for cardiovascular risk reduction in high-risk pediatric patients.
The scientific statement is an update to a 2006 American Heart Association (AHA) statement, adding details about obesity as an at-risk condition and severe obesity as a moderate-risk condition. Other additions include classifying type 2 diabetes as a high-risk condition and expanding on new risk factors for cardiovascular disease (CVD) among patients who received treatment for childhood cancer.
The AHA said the statement is aimed at pediatric cardiologists, primary care physicians, and subspecialists who care for at-risk pediatric patients, as well as providers who will care for these patients as they transition to adult life.
Obesity
In the AHA scientific statement, Sarah de Ferranti, MD, MPH, of Boston Children’s Hospital, chair of the writing group, and her colleagues, highlighted a 2016 study that identified a twofold to threefold higher risk of CVD-related mortality among patients who were overweight or obese, compared with patients of normal weight (Diabetes Care. 2016 Nov;39[11]:1996-2003).
Patients with obesity and severe obesity are at increased risk of aortic or coronary fatty streaks, dyslipidemia, high blood pressure, hyperglycemia, and insulin resistance, as well as inflammatory and oxidative stress, the AHA writing group noted.
They estimated that approximately 6% of U.S. children aged 2-19 years old are considered severely obese.
After identifying patients with obesity, the writing group said, a “multimodal and graduated approach to treatment” for these patients is generally warranted, with a focus on dietary and lifestyle changes, and use of pharmacotherapy and bariatric surgery if indicated.
However, the authors said therapeutic life change modification “is limited in severe obesity because of small effect size and difficulty with sustainability,” while use of pharmacotherapy for treatment of pediatric obesity remains understudied and medications such as orlistat and metformin offer only modest weight loss.
Bariatric surgery, “the only treatment for severe pediatric obesity consistently associated with clinically meaningful and durable weight loss,” is not consistently offered to patients under 12 years old, they added.
Diabetes
The AHA statement also addresses risks from type 1 (T1D) and type 2 diabetes (T2D). Children with T1D and T2D are at increased risk for dyslipidemia, hypertension, microalbuminuria, and obesity. Annual screening for these patients is indicated, and cardiovascular risk factor reduction can be achieved by managing hyperglycemia, controlling weight gain as a result of medication, and implementing therapeutic lifestyle changes, when possible.
Childhood cancer
As survival rates from childhood cancer have improved, there is a need to address the increased risk of cardiovascular-related mortality (estimated at 8-10 times higher than the general population) as well as cancer relapse, according to the writing group.
Among patients recruited to the Childhood Cancer Survivor Study, there was a 9-fold increase in cerebrovascular accident, 10-fold increased risk of coronary artery disease, and 15-fold increase in heart failure for childhood cancer survivors, compared with their siblings who were cancer free.
Cancer treatments such as radiation exposure are linked to increased rates of myocardial infarction, heart failure, valvular abnormalities, and pericardial disease at a twofold to sixfold higher rate when administered at a greater than 1,500 centigray dose, compared to cancer survivors who did not receive radiation, the authors wrote.
Anthracycline treatment is associated with a dose-dependent increase in the risk of dilated cardiomyopathy, while hematopoietic stem cell transplantation may increase the risk of CVD-related mortality from heart failure, cerebrovascular accident, cardiomyopathy, coronary artery disease, and rhythm disorders.
In treating childhood cancer survivors for CVD risk factors, “a low threshold should be used when considering the initiation of pharmacological agents because of the high risk of these youth,” and standard pharmacotherapies can be used, the authors said. “Treatment of cardiovascular risk factors should consider the cancer therapies the patient has received previously.”
In the AHA statement, Dr. de Ferranti and her colleagues also outlined epidemiology, screening, and treatment data for other cardiovascular risk factors such as familial hypercholesterolemia, Lipoprotein(a), hypertension, chronic kidney disease, congenital heart disease, Kawasaki disease, and heart transplantation.
Some members of the writing group reported research grants from Amgen, Sanofi, the Wisconsin Partnership Program, and the National Institutes of Health. One author reported unpaid consultancies with Novo Nordisk, Orexigen, and Vivus.
SOURCE: de Ferranti SD et al. Circulation. 2019 Feb 25. doi: 10.1161/CIR.0000000000000618.
The American Heart Association has included obesity and severe obesity in its updated scientific statement outlining risk factors and considerations for cardiovascular risk reduction in high-risk pediatric patients.
The scientific statement is an update to a 2006 American Heart Association (AHA) statement, adding details about obesity as an at-risk condition and severe obesity as a moderate-risk condition. Other additions include classifying type 2 diabetes as a high-risk condition and expanding on new risk factors for cardiovascular disease (CVD) among patients who received treatment for childhood cancer.
The AHA said the statement is aimed at pediatric cardiologists, primary care physicians, and subspecialists who care for at-risk pediatric patients, as well as providers who will care for these patients as they transition to adult life.
Obesity
In the AHA scientific statement, Sarah de Ferranti, MD, MPH, of Boston Children’s Hospital, chair of the writing group, and her colleagues, highlighted a 2016 study that identified a twofold to threefold higher risk of CVD-related mortality among patients who were overweight or obese, compared with patients of normal weight (Diabetes Care. 2016 Nov;39[11]:1996-2003).
Patients with obesity and severe obesity are at increased risk of aortic or coronary fatty streaks, dyslipidemia, high blood pressure, hyperglycemia, and insulin resistance, as well as inflammatory and oxidative stress, the AHA writing group noted.
They estimated that approximately 6% of U.S. children aged 2-19 years old are considered severely obese.
After identifying patients with obesity, the writing group said, a “multimodal and graduated approach to treatment” for these patients is generally warranted, with a focus on dietary and lifestyle changes, and use of pharmacotherapy and bariatric surgery if indicated.
However, the authors said therapeutic life change modification “is limited in severe obesity because of small effect size and difficulty with sustainability,” while use of pharmacotherapy for treatment of pediatric obesity remains understudied and medications such as orlistat and metformin offer only modest weight loss.
Bariatric surgery, “the only treatment for severe pediatric obesity consistently associated with clinically meaningful and durable weight loss,” is not consistently offered to patients under 12 years old, they added.
Diabetes
The AHA statement also addresses risks from type 1 (T1D) and type 2 diabetes (T2D). Children with T1D and T2D are at increased risk for dyslipidemia, hypertension, microalbuminuria, and obesity. Annual screening for these patients is indicated, and cardiovascular risk factor reduction can be achieved by managing hyperglycemia, controlling weight gain as a result of medication, and implementing therapeutic lifestyle changes, when possible.
Childhood cancer
As survival rates from childhood cancer have improved, there is a need to address the increased risk of cardiovascular-related mortality (estimated at 8-10 times higher than the general population) as well as cancer relapse, according to the writing group.
Among patients recruited to the Childhood Cancer Survivor Study, there was a 9-fold increase in cerebrovascular accident, 10-fold increased risk of coronary artery disease, and 15-fold increase in heart failure for childhood cancer survivors, compared with their siblings who were cancer free.
Cancer treatments such as radiation exposure are linked to increased rates of myocardial infarction, heart failure, valvular abnormalities, and pericardial disease at a twofold to sixfold higher rate when administered at a greater than 1,500 centigray dose, compared to cancer survivors who did not receive radiation, the authors wrote.
Anthracycline treatment is associated with a dose-dependent increase in the risk of dilated cardiomyopathy, while hematopoietic stem cell transplantation may increase the risk of CVD-related mortality from heart failure, cerebrovascular accident, cardiomyopathy, coronary artery disease, and rhythm disorders.
In treating childhood cancer survivors for CVD risk factors, “a low threshold should be used when considering the initiation of pharmacological agents because of the high risk of these youth,” and standard pharmacotherapies can be used, the authors said. “Treatment of cardiovascular risk factors should consider the cancer therapies the patient has received previously.”
In the AHA statement, Dr. de Ferranti and her colleagues also outlined epidemiology, screening, and treatment data for other cardiovascular risk factors such as familial hypercholesterolemia, Lipoprotein(a), hypertension, chronic kidney disease, congenital heart disease, Kawasaki disease, and heart transplantation.
Some members of the writing group reported research grants from Amgen, Sanofi, the Wisconsin Partnership Program, and the National Institutes of Health. One author reported unpaid consultancies with Novo Nordisk, Orexigen, and Vivus.
SOURCE: de Ferranti SD et al. Circulation. 2019 Feb 25. doi: 10.1161/CIR.0000000000000618.
FROM CIRCULATION
FDA clears first-of-its-kind phone app for insulin management
The Food and Drug Administration has granted 501(k) clearance to a phone app for managing insulin in patients with type 2 diabetes.
The app will enhance Hygieia’s d-Nav Insulin Guidance Service, which uses Cloud-based technology and a small group of health care professionals to support physicians and help patients with diabetes achieve better glycemic control by providing personalized insulin adjustments. The system has been shown, in a 90-day clinical study, to reduce both costs and levels of glycosylated hemoglobin. Patients can use the app to enter glucose-event data and receive a recommended insulin dose.
“Insulin therapy is critical to the health of more than 8 million people in the United States, but it is often ineffective, largely because it requires a continual, personalized adjustment that is not practical for physicians and not manageable for patients. The d-Nav service, including the user-friendly phone app, makes insulin therapy more effective, less time intensive, and less costly for everyone involved,” Eran Bashan, CEO of Hygieia, said in a press release.
This is the first insulin-mangement app that can titrate individualized doses for all insulin regimens and the first that can connect to any glucose meter that shares information with the cloud. It is available for both Android and iOS.
Find the full press release on the Hygieia website.
The Food and Drug Administration has granted 501(k) clearance to a phone app for managing insulin in patients with type 2 diabetes.
The app will enhance Hygieia’s d-Nav Insulin Guidance Service, which uses Cloud-based technology and a small group of health care professionals to support physicians and help patients with diabetes achieve better glycemic control by providing personalized insulin adjustments. The system has been shown, in a 90-day clinical study, to reduce both costs and levels of glycosylated hemoglobin. Patients can use the app to enter glucose-event data and receive a recommended insulin dose.
“Insulin therapy is critical to the health of more than 8 million people in the United States, but it is often ineffective, largely because it requires a continual, personalized adjustment that is not practical for physicians and not manageable for patients. The d-Nav service, including the user-friendly phone app, makes insulin therapy more effective, less time intensive, and less costly for everyone involved,” Eran Bashan, CEO of Hygieia, said in a press release.
This is the first insulin-mangement app that can titrate individualized doses for all insulin regimens and the first that can connect to any glucose meter that shares information with the cloud. It is available for both Android and iOS.
Find the full press release on the Hygieia website.
The Food and Drug Administration has granted 501(k) clearance to a phone app for managing insulin in patients with type 2 diabetes.
The app will enhance Hygieia’s d-Nav Insulin Guidance Service, which uses Cloud-based technology and a small group of health care professionals to support physicians and help patients with diabetes achieve better glycemic control by providing personalized insulin adjustments. The system has been shown, in a 90-day clinical study, to reduce both costs and levels of glycosylated hemoglobin. Patients can use the app to enter glucose-event data and receive a recommended insulin dose.
“Insulin therapy is critical to the health of more than 8 million people in the United States, but it is often ineffective, largely because it requires a continual, personalized adjustment that is not practical for physicians and not manageable for patients. The d-Nav service, including the user-friendly phone app, makes insulin therapy more effective, less time intensive, and less costly for everyone involved,” Eran Bashan, CEO of Hygieia, said in a press release.
This is the first insulin-mangement app that can titrate individualized doses for all insulin regimens and the first that can connect to any glucose meter that shares information with the cloud. It is available for both Android and iOS.
Find the full press release on the Hygieia website.
Diabetic kidney disease, retinopathy associated with PAD in patients with foot ulcers
Patients with diabetic foot ulcers have a high incidence of associated chronic vascular disease, including kidney disease, retinopathy, and peripheral artery disease (PAD). In addition, according to a study reported by Magdy H. Megallaa, MD, and colleagues at Alexandria (Egypt) University.
Their cross-sectional study, published in Diabetes & Metabolic Syndrome: Clinical Research and Reviews, comprised 180 type 2 diabetic patients (aged 30-70 years) with diabetic foot ulcers (DFUs) who attended an outpatient clinic between July and December 2017.
The prevalence of diabetic kidney disease and diabetic retinopathy was 86.1% and 90.0%, respectively, with 86.7% of patients having neuropathic DFUs, 11.1% having ischemic DFUs, and 2.2% having neuroischemic DFUs. The prevalence of peripheral neuropathy and PAD was 82% and 20%, respectively.
Using albuminuria as a measure of diabetic kidney disease, the researchers found that 86.1% of the patients had albuminuria and that there was a statistically significant association between albuminuria and the patient’s vibration reception threshold (VPT), a measure of diabetic neuropathy (P less than .001), and the ankle brachial index (ABI), a measure of PAD (P less than .031).
In addition, there was a statistically significant association between diabetic retinopathy and VPT (P less than .008) and between diabetic retinopathy and ABI (P less than .001).
“Albuminuria, diabetic retinopathy and peripheral neuropathy are very common among those patients and strongly associated with risk factors of diabetic foot ulceration,” the researchers concluded.
They reported that they had no conflicts.
SOURCE: Megallaa MH et al. Diabetes Metab Syndr. 2019 Mar-Apr;13(2):1287-92.
Patients with diabetic foot ulcers have a high incidence of associated chronic vascular disease, including kidney disease, retinopathy, and peripheral artery disease (PAD). In addition, according to a study reported by Magdy H. Megallaa, MD, and colleagues at Alexandria (Egypt) University.
Their cross-sectional study, published in Diabetes & Metabolic Syndrome: Clinical Research and Reviews, comprised 180 type 2 diabetic patients (aged 30-70 years) with diabetic foot ulcers (DFUs) who attended an outpatient clinic between July and December 2017.
The prevalence of diabetic kidney disease and diabetic retinopathy was 86.1% and 90.0%, respectively, with 86.7% of patients having neuropathic DFUs, 11.1% having ischemic DFUs, and 2.2% having neuroischemic DFUs. The prevalence of peripheral neuropathy and PAD was 82% and 20%, respectively.
Using albuminuria as a measure of diabetic kidney disease, the researchers found that 86.1% of the patients had albuminuria and that there was a statistically significant association between albuminuria and the patient’s vibration reception threshold (VPT), a measure of diabetic neuropathy (P less than .001), and the ankle brachial index (ABI), a measure of PAD (P less than .031).
In addition, there was a statistically significant association between diabetic retinopathy and VPT (P less than .008) and between diabetic retinopathy and ABI (P less than .001).
“Albuminuria, diabetic retinopathy and peripheral neuropathy are very common among those patients and strongly associated with risk factors of diabetic foot ulceration,” the researchers concluded.
They reported that they had no conflicts.
SOURCE: Megallaa MH et al. Diabetes Metab Syndr. 2019 Mar-Apr;13(2):1287-92.
Patients with diabetic foot ulcers have a high incidence of associated chronic vascular disease, including kidney disease, retinopathy, and peripheral artery disease (PAD). In addition, according to a study reported by Magdy H. Megallaa, MD, and colleagues at Alexandria (Egypt) University.
Their cross-sectional study, published in Diabetes & Metabolic Syndrome: Clinical Research and Reviews, comprised 180 type 2 diabetic patients (aged 30-70 years) with diabetic foot ulcers (DFUs) who attended an outpatient clinic between July and December 2017.
The prevalence of diabetic kidney disease and diabetic retinopathy was 86.1% and 90.0%, respectively, with 86.7% of patients having neuropathic DFUs, 11.1% having ischemic DFUs, and 2.2% having neuroischemic DFUs. The prevalence of peripheral neuropathy and PAD was 82% and 20%, respectively.
Using albuminuria as a measure of diabetic kidney disease, the researchers found that 86.1% of the patients had albuminuria and that there was a statistically significant association between albuminuria and the patient’s vibration reception threshold (VPT), a measure of diabetic neuropathy (P less than .001), and the ankle brachial index (ABI), a measure of PAD (P less than .031).
In addition, there was a statistically significant association between diabetic retinopathy and VPT (P less than .008) and between diabetic retinopathy and ABI (P less than .001).
“Albuminuria, diabetic retinopathy and peripheral neuropathy are very common among those patients and strongly associated with risk factors of diabetic foot ulceration,” the researchers concluded.
They reported that they had no conflicts.
SOURCE: Megallaa MH et al. Diabetes Metab Syndr. 2019 Mar-Apr;13(2):1287-92.
FROM DIABETES & METABOLIC SYNDROME: CLINICAL RESEARCH & REVIEWS
AACE/ACE algorithm provides practical clinical guidance on managing diabetes
Leading endocrinology societies have copublished an algorithm offering updated, specific clinical guidance on lifestyle therapy, management of hypertension and dyslipidemia, and glucose control in patients with type 2 diabetes.
This update from the American Association of Clinical Endocrinologists and the American College of Endocrinology, published in Endocrine Practice, also highlights obesity and prediabetes as underlying risk factors for development of diabetes.
The algorithm, based on new and “comprehensive clinical data” on type 2 diabetes management, is designed as a supplement 2015 AACE/ACE clinical practice guidelines, according to Alan J. Garber, MD, PhD, chair of the Diabetes Management Algorithm Task Force.
“It’s intended to provide clinicians with a practical guide that prompts them to look for factors or influences in the patient’s lifestyle or health that may be a factor in identifying the best treatment approach or medication,” he said in a statement.
Lifestyle medication is critical for all patients with diabetes, according to Dr. Garber and the algorithm coauthors, who recommended a “primarily plant-based meal plan” that limits intake of saturated fatty acids and avoids trans fats. They said overweight patients should restrict caloric intake with a goal of reducing body weight by up to 10%.
Physical activity should include at least 150 minutes per week of activities such as brisk walking or weight training, they said, adding that patients should be advised to sleep 7 hours per night, on average.
Weight loss medications might be needed along with lifestyle modification for patients with body mass index (BMI) over 27 kg/m2 with complications, and for all patients with BMI over 30 regardless of whether they have complications, according to the AACE/ACE committee members who drafted the report.
Bariatric surgery might be considered in patients with BMI over 35 and comorbidities, particularly if patients fail to achieve weight loss goals using other means, they added.
The primary goal of prediabetes management is weight loss, wrote the authors. While there are no Food and Drug Administration–approved agents for prediabetes management, they said, antihyperglycemic agents such as metformin and acarbose have been shown to reduce risk of diabetes by 25%-30% in patients with prediabetes.
While pressure control needs to be individualized, but a goal of less than 130/80 mm Hg is warranted for most patients with diabetes, according to the authors, who note that most patients will require medication to reach their goal.
“Because angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers can slow progression of nephropathy and retinopathy, they are preferred for patients with type 2 diabetes,” said Dr. Garber and his coauthors in the executive summary accompanying the algorithm.
Early and intensive management of dyslipidemia is important to reduce the significant risk of atherosclerotic cardiovascular disease in patients with diabetes, according to the authors, who say diabetes patients should be classified as high risk, very high risk, or extreme risk. They recommended LDL cholesterol targets of less than 100 mg/dL for high-risk patients, less than 70 mg/dL for very-high-risk patients, and less than 55 mg/dL for the extreme-risk group.
Statins should be considered first-line treatment for lowering cholesterol, unless contraindicated, with other lipid-modifying agents added as needed to reach lipid targets.
Inhibitors of proprotein convertase subtilisin/kexin type 9 serine protease (PCSK9) address a “large unmet need” for more aggressive lipid lowering in patients with clinical atherosclerotic disease and diabetes, the authors noted.
Added to maximal statin therapy, PCSK9 inhibitors reduce LDL cholesterol by about 50% while also raising HDL cholesterol and having positive effects on other lipids, according to the authors.
Pharmacotherapy for type 2 diabetes requires a “nuanced approach” that takes into account factors such as age, comorbidities, and risk of hypoglycemia, the authors wrote, noting that the AACE supports a hemoglobin A1c target of 6.5% or less for most patients.
The algorithm for glycemic control lists glucose-lowering agents in order of recommended usage. For example, in patients with an entry HbA1c less than 7.5%, the strongest recommendations for were monotherapy with metformin, followed by GLP1 receptor agonists and sodium-glucose cotransporter 2 (SGLT2) inhibitors.
If insulin becomes necessary, the recommended approach is to add a single daily dose of basal insulin, and if a basal insulin regimen fails to control glucose, it may help to add a GLP1 receptor agonist or dipeptidyl peptidase 4 (DPP4) inhibitor, according to the algorithm.
Avoiding hypoglycemia is important, and one possible “safety measure” to prevent that is using a continuous glucose monitoring device that provides real-time glucose data. “Significant advances have been made in accuracy and availability of CGM devices,” the authors wrote.
Current expert consensus is that clinical CGM devices should be considered if patients have not achieved their glycemic target after 3 months or if they need a treatment that puts them at risk for hypoglycemia, according to Dr. Garber and his colleagues.
Dr. Garber reported that he had no financial relationships relevant to the consensus statement and algorithm. Coauthors of the report provided disclosures related to Novo Nordisk, Eli Lilly, Janssen Pharmaceuticals, Abbott, Sanofi-Aventis, and other pharmaceutical companies.
SOURCE: Garber AJ et al. Endocr Pract. 2019 Jan;25(1):91-120.
Leading endocrinology societies have copublished an algorithm offering updated, specific clinical guidance on lifestyle therapy, management of hypertension and dyslipidemia, and glucose control in patients with type 2 diabetes.
This update from the American Association of Clinical Endocrinologists and the American College of Endocrinology, published in Endocrine Practice, also highlights obesity and prediabetes as underlying risk factors for development of diabetes.
The algorithm, based on new and “comprehensive clinical data” on type 2 diabetes management, is designed as a supplement 2015 AACE/ACE clinical practice guidelines, according to Alan J. Garber, MD, PhD, chair of the Diabetes Management Algorithm Task Force.
“It’s intended to provide clinicians with a practical guide that prompts them to look for factors or influences in the patient’s lifestyle or health that may be a factor in identifying the best treatment approach or medication,” he said in a statement.
Lifestyle medication is critical for all patients with diabetes, according to Dr. Garber and the algorithm coauthors, who recommended a “primarily plant-based meal plan” that limits intake of saturated fatty acids and avoids trans fats. They said overweight patients should restrict caloric intake with a goal of reducing body weight by up to 10%.
Physical activity should include at least 150 minutes per week of activities such as brisk walking or weight training, they said, adding that patients should be advised to sleep 7 hours per night, on average.
Weight loss medications might be needed along with lifestyle modification for patients with body mass index (BMI) over 27 kg/m2 with complications, and for all patients with BMI over 30 regardless of whether they have complications, according to the AACE/ACE committee members who drafted the report.
Bariatric surgery might be considered in patients with BMI over 35 and comorbidities, particularly if patients fail to achieve weight loss goals using other means, they added.
The primary goal of prediabetes management is weight loss, wrote the authors. While there are no Food and Drug Administration–approved agents for prediabetes management, they said, antihyperglycemic agents such as metformin and acarbose have been shown to reduce risk of diabetes by 25%-30% in patients with prediabetes.
While pressure control needs to be individualized, but a goal of less than 130/80 mm Hg is warranted for most patients with diabetes, according to the authors, who note that most patients will require medication to reach their goal.
“Because angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers can slow progression of nephropathy and retinopathy, they are preferred for patients with type 2 diabetes,” said Dr. Garber and his coauthors in the executive summary accompanying the algorithm.
Early and intensive management of dyslipidemia is important to reduce the significant risk of atherosclerotic cardiovascular disease in patients with diabetes, according to the authors, who say diabetes patients should be classified as high risk, very high risk, or extreme risk. They recommended LDL cholesterol targets of less than 100 mg/dL for high-risk patients, less than 70 mg/dL for very-high-risk patients, and less than 55 mg/dL for the extreme-risk group.
Statins should be considered first-line treatment for lowering cholesterol, unless contraindicated, with other lipid-modifying agents added as needed to reach lipid targets.
Inhibitors of proprotein convertase subtilisin/kexin type 9 serine protease (PCSK9) address a “large unmet need” for more aggressive lipid lowering in patients with clinical atherosclerotic disease and diabetes, the authors noted.
Added to maximal statin therapy, PCSK9 inhibitors reduce LDL cholesterol by about 50% while also raising HDL cholesterol and having positive effects on other lipids, according to the authors.
Pharmacotherapy for type 2 diabetes requires a “nuanced approach” that takes into account factors such as age, comorbidities, and risk of hypoglycemia, the authors wrote, noting that the AACE supports a hemoglobin A1c target of 6.5% or less for most patients.
The algorithm for glycemic control lists glucose-lowering agents in order of recommended usage. For example, in patients with an entry HbA1c less than 7.5%, the strongest recommendations for were monotherapy with metformin, followed by GLP1 receptor agonists and sodium-glucose cotransporter 2 (SGLT2) inhibitors.
If insulin becomes necessary, the recommended approach is to add a single daily dose of basal insulin, and if a basal insulin regimen fails to control glucose, it may help to add a GLP1 receptor agonist or dipeptidyl peptidase 4 (DPP4) inhibitor, according to the algorithm.
Avoiding hypoglycemia is important, and one possible “safety measure” to prevent that is using a continuous glucose monitoring device that provides real-time glucose data. “Significant advances have been made in accuracy and availability of CGM devices,” the authors wrote.
Current expert consensus is that clinical CGM devices should be considered if patients have not achieved their glycemic target after 3 months or if they need a treatment that puts them at risk for hypoglycemia, according to Dr. Garber and his colleagues.
Dr. Garber reported that he had no financial relationships relevant to the consensus statement and algorithm. Coauthors of the report provided disclosures related to Novo Nordisk, Eli Lilly, Janssen Pharmaceuticals, Abbott, Sanofi-Aventis, and other pharmaceutical companies.
SOURCE: Garber AJ et al. Endocr Pract. 2019 Jan;25(1):91-120.
Leading endocrinology societies have copublished an algorithm offering updated, specific clinical guidance on lifestyle therapy, management of hypertension and dyslipidemia, and glucose control in patients with type 2 diabetes.
This update from the American Association of Clinical Endocrinologists and the American College of Endocrinology, published in Endocrine Practice, also highlights obesity and prediabetes as underlying risk factors for development of diabetes.
The algorithm, based on new and “comprehensive clinical data” on type 2 diabetes management, is designed as a supplement 2015 AACE/ACE clinical practice guidelines, according to Alan J. Garber, MD, PhD, chair of the Diabetes Management Algorithm Task Force.
“It’s intended to provide clinicians with a practical guide that prompts them to look for factors or influences in the patient’s lifestyle or health that may be a factor in identifying the best treatment approach or medication,” he said in a statement.
Lifestyle medication is critical for all patients with diabetes, according to Dr. Garber and the algorithm coauthors, who recommended a “primarily plant-based meal plan” that limits intake of saturated fatty acids and avoids trans fats. They said overweight patients should restrict caloric intake with a goal of reducing body weight by up to 10%.
Physical activity should include at least 150 minutes per week of activities such as brisk walking or weight training, they said, adding that patients should be advised to sleep 7 hours per night, on average.
Weight loss medications might be needed along with lifestyle modification for patients with body mass index (BMI) over 27 kg/m2 with complications, and for all patients with BMI over 30 regardless of whether they have complications, according to the AACE/ACE committee members who drafted the report.
Bariatric surgery might be considered in patients with BMI over 35 and comorbidities, particularly if patients fail to achieve weight loss goals using other means, they added.
The primary goal of prediabetes management is weight loss, wrote the authors. While there are no Food and Drug Administration–approved agents for prediabetes management, they said, antihyperglycemic agents such as metformin and acarbose have been shown to reduce risk of diabetes by 25%-30% in patients with prediabetes.
While pressure control needs to be individualized, but a goal of less than 130/80 mm Hg is warranted for most patients with diabetes, according to the authors, who note that most patients will require medication to reach their goal.
“Because angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers can slow progression of nephropathy and retinopathy, they are preferred for patients with type 2 diabetes,” said Dr. Garber and his coauthors in the executive summary accompanying the algorithm.
Early and intensive management of dyslipidemia is important to reduce the significant risk of atherosclerotic cardiovascular disease in patients with diabetes, according to the authors, who say diabetes patients should be classified as high risk, very high risk, or extreme risk. They recommended LDL cholesterol targets of less than 100 mg/dL for high-risk patients, less than 70 mg/dL for very-high-risk patients, and less than 55 mg/dL for the extreme-risk group.
Statins should be considered first-line treatment for lowering cholesterol, unless contraindicated, with other lipid-modifying agents added as needed to reach lipid targets.
Inhibitors of proprotein convertase subtilisin/kexin type 9 serine protease (PCSK9) address a “large unmet need” for more aggressive lipid lowering in patients with clinical atherosclerotic disease and diabetes, the authors noted.
Added to maximal statin therapy, PCSK9 inhibitors reduce LDL cholesterol by about 50% while also raising HDL cholesterol and having positive effects on other lipids, according to the authors.
Pharmacotherapy for type 2 diabetes requires a “nuanced approach” that takes into account factors such as age, comorbidities, and risk of hypoglycemia, the authors wrote, noting that the AACE supports a hemoglobin A1c target of 6.5% or less for most patients.
The algorithm for glycemic control lists glucose-lowering agents in order of recommended usage. For example, in patients with an entry HbA1c less than 7.5%, the strongest recommendations for were monotherapy with metformin, followed by GLP1 receptor agonists and sodium-glucose cotransporter 2 (SGLT2) inhibitors.
If insulin becomes necessary, the recommended approach is to add a single daily dose of basal insulin, and if a basal insulin regimen fails to control glucose, it may help to add a GLP1 receptor agonist or dipeptidyl peptidase 4 (DPP4) inhibitor, according to the algorithm.
Avoiding hypoglycemia is important, and one possible “safety measure” to prevent that is using a continuous glucose monitoring device that provides real-time glucose data. “Significant advances have been made in accuracy and availability of CGM devices,” the authors wrote.
Current expert consensus is that clinical CGM devices should be considered if patients have not achieved their glycemic target after 3 months or if they need a treatment that puts them at risk for hypoglycemia, according to Dr. Garber and his colleagues.
Dr. Garber reported that he had no financial relationships relevant to the consensus statement and algorithm. Coauthors of the report provided disclosures related to Novo Nordisk, Eli Lilly, Janssen Pharmaceuticals, Abbott, Sanofi-Aventis, and other pharmaceutical companies.
SOURCE: Garber AJ et al. Endocr Pract. 2019 Jan;25(1):91-120.
FROM ENDOCRINE PRACTICE