User login
Nonadherent Diabetes Patients: An Unexpected Group
“Time-specific” dosing of insulin can be an obstacle to adherence for patients with complicated, busy lives. More than half of patients with type 2 diabetes do not achieve their target HbA1c of 7% after insulin is added to their treatment regimen. Researchers from CAPHRI School for Public Health and Primary Care, and CARIM Institute in The Netherlands, who surveyed 1,483 adults with diabetes suggest that it may be time to rethink both prescribing and patient education, in part because of who fell into the nonadherent group.
The researchers conducted a web-based self-report survey. Of the respondents, 58% used bolus insulin before meals, 24% after meals, and 18% before, during, or after meals. The researchers excluded the “mixed” cohort, including 1,218 in the analysis.
Half the respondents in the postmeal cohort reported experiencing minor hypoglycemic events at least once a week compared with 35% of the premeal cohort. Similarly, more in the postmeal group had had major hypoglycemic events (38% vs 26%). The postmeal respondents also were more likely to have HbA1c ≥ 9% (40% vs 29%). And they were less likely to report always testing their blood glucose before injecting insulin (36% vs 54%).
Perhaps contrary to some expectations, the respondents who injected insulin postmeal were younger, had shorter duration of diabetes, had the highest level of college or university education, were more likely to be employed, and more frequently participated in diabetes education programs (including one-on-one programs).
The researchers say those data suggest that factors other than lack of diabetes education, education, or low socioeconomic status should be considered in explaining the nonadherence. They add that some research has shown that education programs have an “inconsistent relationship with patient adherence.” They suggest that such programs might be improved by placing greater emphasis on the importance of dosing insulin before meals.
Of the nearly 20% of patients who use insulin treatment, > 90% receive bolus insulin. The researchers note that respondents preferred a form of bolus insulin they can administer before, after, or during meals as they see fit. The respondents who injected postmeal were more likely than the premeal respondents to prefer this formulation.
“Time-specific” dosing of insulin can be an obstacle to adherence for patients with complicated, busy lives. More than half of patients with type 2 diabetes do not achieve their target HbA1c of 7% after insulin is added to their treatment regimen. Researchers from CAPHRI School for Public Health and Primary Care, and CARIM Institute in The Netherlands, who surveyed 1,483 adults with diabetes suggest that it may be time to rethink both prescribing and patient education, in part because of who fell into the nonadherent group.
The researchers conducted a web-based self-report survey. Of the respondents, 58% used bolus insulin before meals, 24% after meals, and 18% before, during, or after meals. The researchers excluded the “mixed” cohort, including 1,218 in the analysis.
Half the respondents in the postmeal cohort reported experiencing minor hypoglycemic events at least once a week compared with 35% of the premeal cohort. Similarly, more in the postmeal group had had major hypoglycemic events (38% vs 26%). The postmeal respondents also were more likely to have HbA1c ≥ 9% (40% vs 29%). And they were less likely to report always testing their blood glucose before injecting insulin (36% vs 54%).
Perhaps contrary to some expectations, the respondents who injected insulin postmeal were younger, had shorter duration of diabetes, had the highest level of college or university education, were more likely to be employed, and more frequently participated in diabetes education programs (including one-on-one programs).
The researchers say those data suggest that factors other than lack of diabetes education, education, or low socioeconomic status should be considered in explaining the nonadherence. They add that some research has shown that education programs have an “inconsistent relationship with patient adherence.” They suggest that such programs might be improved by placing greater emphasis on the importance of dosing insulin before meals.
Of the nearly 20% of patients who use insulin treatment, > 90% receive bolus insulin. The researchers note that respondents preferred a form of bolus insulin they can administer before, after, or during meals as they see fit. The respondents who injected postmeal were more likely than the premeal respondents to prefer this formulation.
“Time-specific” dosing of insulin can be an obstacle to adherence for patients with complicated, busy lives. More than half of patients with type 2 diabetes do not achieve their target HbA1c of 7% after insulin is added to their treatment regimen. Researchers from CAPHRI School for Public Health and Primary Care, and CARIM Institute in The Netherlands, who surveyed 1,483 adults with diabetes suggest that it may be time to rethink both prescribing and patient education, in part because of who fell into the nonadherent group.
The researchers conducted a web-based self-report survey. Of the respondents, 58% used bolus insulin before meals, 24% after meals, and 18% before, during, or after meals. The researchers excluded the “mixed” cohort, including 1,218 in the analysis.
Half the respondents in the postmeal cohort reported experiencing minor hypoglycemic events at least once a week compared with 35% of the premeal cohort. Similarly, more in the postmeal group had had major hypoglycemic events (38% vs 26%). The postmeal respondents also were more likely to have HbA1c ≥ 9% (40% vs 29%). And they were less likely to report always testing their blood glucose before injecting insulin (36% vs 54%).
Perhaps contrary to some expectations, the respondents who injected insulin postmeal were younger, had shorter duration of diabetes, had the highest level of college or university education, were more likely to be employed, and more frequently participated in diabetes education programs (including one-on-one programs).
The researchers say those data suggest that factors other than lack of diabetes education, education, or low socioeconomic status should be considered in explaining the nonadherence. They add that some research has shown that education programs have an “inconsistent relationship with patient adherence.” They suggest that such programs might be improved by placing greater emphasis on the importance of dosing insulin before meals.
Of the nearly 20% of patients who use insulin treatment, > 90% receive bolus insulin. The researchers note that respondents preferred a form of bolus insulin they can administer before, after, or during meals as they see fit. The respondents who injected postmeal were more likely than the premeal respondents to prefer this formulation.
Sudden-onset rash on the trunk and limbs • morbid obesity • family history of diabetes mellitus • Dx?
THE CASE
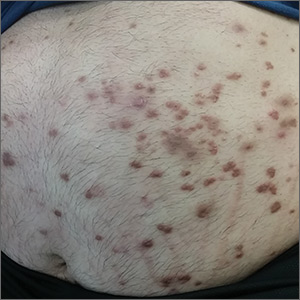
A 37-year-old man presented with a sudden-onset, nonpruritic, nonpainful, papular rash of 1 month’s duration on his trunk and both arms and legs. Two weeks prior to the current presentation, he consulted a general practitioner, who treated the rash with a course of unknown oral antibiotics; the patient showed no improvement. He recalled that on a few occasions, he used his fingers to express a creamy discharge from some of the lesions. This temporarily reduced the size of those papules.
His medical history was unremarkable except for morbid obesity. He did not drink alcohol regularly and was not taking any medications prior to the onset of the rash. He had no family history of hyperlipidemia, but his mother had a history of diabetes mellitus.
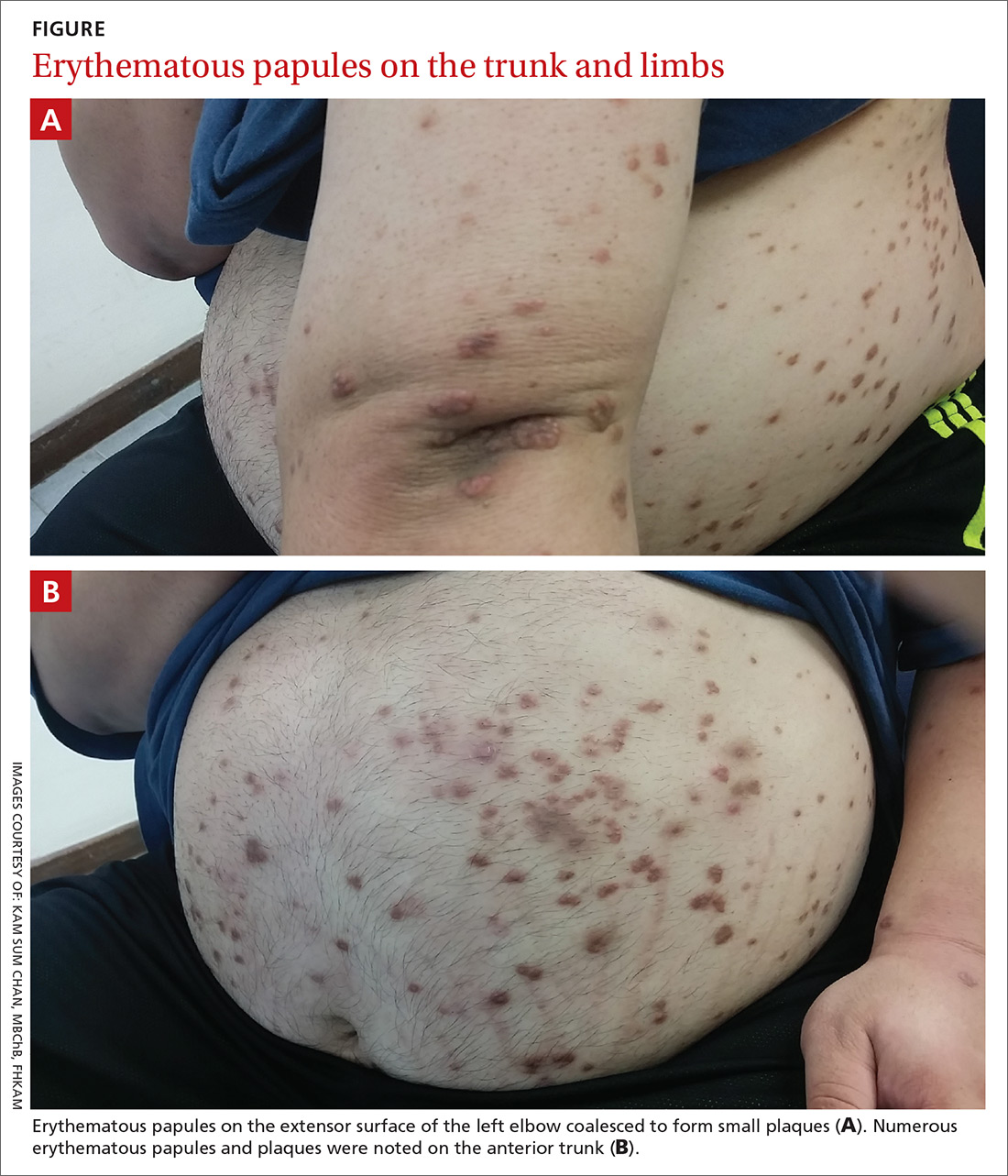
Physical examination showed numerous discrete erythematous papules with a creamy center on his trunk and his arms and legs. The lesions were more numerous on the extensor surfaces of the arms and legs. Some of the papules coalesced to form small plaques (FIGURE). There was no scaling, and the lesions were firm in texture. The patient’s face was spared, and there was no mucosal involvement. The patient was otherwise systemically well.
THE DIAGNOSIS
Based on the morphology, distribution, and abrupt onset of the diffuse nonpruritic papules in this morbidly obese (but otherwise systemically well) middle-aged man, a clinical diagnosis of eruptive xanthoma was suspected. Subsequent blood testing revealed an elevated serum triglyceride level of 47.8 mmol/L (reference range, <1.7 mmol/L), elevated serum total cholesterol of 7.1 mmol/L (reference range, <6.2 mmol/L), and low serum high-density lipoprotein cholesterol of 0.7 mmol/L (reference range, >1 mmol/L in men). He also had an elevated fasting serum glucose level of 12.9 mmol/L (reference range, 3.9–5.6 mmol/L) and an elevated hemoglobin A1c (glycated hemoglobin) level of 10.9%.
Subsequent thyroid, liver, and renal function tests were normal, but the patient had heavy proteinuria, with an elevated urine albumin-to-creatinine ratio of 355.6 mg/mmol (reference range, ≤2.5 mg/mmol). The patient was referred to a dermatologist, who confirmed the clinical diagnosis without the need for a skin biopsy.
DISCUSSION

Eruptive xanthoma is characterized by an abrupt onset of crops of multiple yellowish to brownish papules that can coalesce into small plaques. The lesions can be generalized, but tend to be more densely distributed on the extensor surfaces of the arms and legs, buttocks, and thighs.5 Eruptive xanthoma often is associated with hypertriglyceridemia, which can be primary—as a result of a genetic defect caused by familial hypertriglyceridemia—or secondary, associated with poorly controlled diabetes mellitus, morbid obesity, excessive alcohol consumption, nephrotic syndrome, hypothyroidism, primary biliary cholangitis, and drugs like estrogen replacement therapies, corticosteroids, and isotretinoin.6 Pruritus and tenderness may or may not be present, and the Köbner phenomenon may occur.7
Continue to: The differential diagnosis
The differential diagnosis for eruptive xanthoma includes xanthoma disseminatum, non–Langerhans cell histiocytoses (eg, generalized eruptive histiocytosis), and cutaneous mastocytosis.1
Xanthoma disseminatum is an extremely rare, but benign, disorder of non–Langerhans cell origin. The average age of onset is older than 40 years. The rash consists of multiple red-yellow papules and nodules that most commonly present in flexural areas. Forty percent to 60% of patients have mucosal involvement, and rarely the central nervous system is involved.8
Generalized eruptive histiocytosis is another rare non–Langerhans cell histiocytosis that occurs mainly in adults and is characterized by widespread, symmetric, red-brown papules on the trunk, arms, and legs, and rarely the mucous membranes.9
Cutaneous mastocytosis, especially xanthelasmoid mastocytosis, consists of multiple pruritic, yellowish, papular or nodular lesions that may mimic eruptive xanthoma. It occurs mainly in children and rarely in adults.10
Confirming the diagnosis, initiating treatment
The diagnosis of eruptive xanthoma can be confirmed by skin biopsy if other differential diagnoses cannot be ruled out or the lesions do not resolve with treatment. Skin biopsy will reveal lipid-laden macrophages (known as foam cells) deposited in the dermis.7
Continue to: Treatment of eruptive xanthoma
Treatment of eruptive xanthoma involves management of the underlying causes of the condition. In most cases, dietary control, intensive triglyceride-lowering therapies, and treatment of other secondary causes of hypertriglyceridemia result in complete resolution of the lesions within several weeks.5
Our patient’s outcome
Our patient’s sudden-onset rash alerted us to the presence of type 2 diabetes mellitus, hypertriglyceridemia, and heavy proteinuria, which he was not aware of previously. We counselled him about stringent low-sugar, low-lipid diet control and exercise, and we started him on metformin and gemfibrozil. He was referred to an internal medicine specialist for further assessment and management of his severe hypertriglyceridemia and heavy proteinuria.
The rash started to wane 1 month after the patient started the metformin and gemfibrozil, and his drug regimen was changed to combination therapy with metformin/glimepiride and fenofibrate/simvastatin 6 weeks later when he was seen in the medical specialty clinic. Fundus photography performed 1 month after starting oral antidiabetic therapy showed no diabetic retinopathy or lipemia retinalis.
After 3 months of treatment, his serum triglycerides and hemoglobin A1c levels dropped to 3.8 mmol/L and 8.7%, respectively. The rash also resolved considerably, with only residual papules on the abdomen. This rapid clinical response to treatment of the underlying hypertriglyceridemia and diabetes further supported the clinical diagnosis of eruptive xanthoma.
THE TAKEAWAY
Eruptive xanthoma is relatively rare, but it is important for family physicians to recognize this clinical presentation as a potential indicator of severe hypertriglyceridemia. Recognizing hypertriglyceridemia early is important, as it can be associated with an increased risk for acute pancreatitis. Moreover, eruptive xanthoma might be the sole presenting symptom of underlying diabetes mellitus or familial hyperlipidemia, both of which can lead to a significant increase in cardiovascular risk if uncontrolled.
CORRESPONDENCE
Chan Kam Sum, MBChB, FRACGP, Tseung Kwan O Jockey Club General Out-patient Clinic, 99 Po Lam Road North, G/F, Tseung Kwan O, Kowloon, Hong Kong; [email protected]
1. Tang WK. Eruptive xanthoma. [case reports]. Hong Kong Dermatol Venereol Bull. 2001;9:172-175.
2. Frew J, Murrell D, Haber R. Fifty shades of yellow: a review of the xanthodermatoses. Int J Dermatol. 2015;54:1109-1123.
3. Zak A, Zeman M, Slaby A, et al. Xanthomas: clinical and pathophysiological relations. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2014;158:181-188.
4. Sandhu S, Al-Sarraf A, Taraboanta C, et al. Incidence of pancreatitis, secondary causes, and treatment of patients referred to specialty lipid clinic with severe hypertriglyceridemia: a retrospective cohort study. Lipids Health Dis. 2011;10:157.
5. Holsinger JM, Campbell SM, Witman P. Multiple erythematous-yellow, dome-shaped papules. Am Fam Physician. 2010;82:517.
6. Loeckermann S, Braun-Falco M. Eruptive xanthomas in association with metabolic syndrome. Clin Exp Dermatol. 2010;35:565-566.
7. Merola JF, Mengden SJ, Soldano A, et al. Eruptive xanthomas. Dermatol Online J. 2008;14:10.
8. Park M, Boone B, Devas S. Xanthoma disseminatum: case report and mini-review of the literature. Acta Dermatovenerol Croat. 2014;22:150-154.
9. Attia A, Seleit I, El Badawy N, et al. Photoletter to the editor: generalized eruptive histiocytoma. J Dermatol Case Rep. 2011;5:53-55.
10. Nabavi NS, Nejad MH, Feli S, et al. Adult onset of xanthelasmoid mastocytosis: report of a rare entity. Indian J Dermatol. 2016;61:468.
THE CASE
A 37-year-old man presented with a sudden-onset, nonpruritic, nonpainful, papular rash of 1 month’s duration on his trunk and both arms and legs. Two weeks prior to the current presentation, he consulted a general practitioner, who treated the rash with a course of unknown oral antibiotics; the patient showed no improvement. He recalled that on a few occasions, he used his fingers to express a creamy discharge from some of the lesions. This temporarily reduced the size of those papules.
His medical history was unremarkable except for morbid obesity. He did not drink alcohol regularly and was not taking any medications prior to the onset of the rash. He had no family history of hyperlipidemia, but his mother had a history of diabetes mellitus.
Physical examination showed numerous discrete erythematous papules with a creamy center on his trunk and his arms and legs. The lesions were more numerous on the extensor surfaces of the arms and legs. Some of the papules coalesced to form small plaques (FIGURE). There was no scaling, and the lesions were firm in texture. The patient’s face was spared, and there was no mucosal involvement. The patient was otherwise systemically well.

THE DIAGNOSIS
Based on the morphology, distribution, and abrupt onset of the diffuse nonpruritic papules in this morbidly obese (but otherwise systemically well) middle-aged man, a clinical diagnosis of eruptive xanthoma was suspected. Subsequent blood testing revealed an elevated serum triglyceride level of 47.8 mmol/L (reference range, <1.7 mmol/L), elevated serum total cholesterol of 7.1 mmol/L (reference range, <6.2 mmol/L), and low serum high-density lipoprotein cholesterol of 0.7 mmol/L (reference range, >1 mmol/L in men). He also had an elevated fasting serum glucose level of 12.9 mmol/L (reference range, 3.9–5.6 mmol/L) and an elevated hemoglobin A1c (glycated hemoglobin) level of 10.9%.
Subsequent thyroid, liver, and renal function tests were normal, but the patient had heavy proteinuria, with an elevated urine albumin-to-creatinine ratio of 355.6 mg/mmol (reference range, ≤2.5 mg/mmol). The patient was referred to a dermatologist, who confirmed the clinical diagnosis without the need for a skin biopsy.
DISCUSSION

Eruptive xanthoma is characterized by an abrupt onset of crops of multiple yellowish to brownish papules that can coalesce into small plaques. The lesions can be generalized, but tend to be more densely distributed on the extensor surfaces of the arms and legs, buttocks, and thighs.5 Eruptive xanthoma often is associated with hypertriglyceridemia, which can be primary—as a result of a genetic defect caused by familial hypertriglyceridemia—or secondary, associated with poorly controlled diabetes mellitus, morbid obesity, excessive alcohol consumption, nephrotic syndrome, hypothyroidism, primary biliary cholangitis, and drugs like estrogen replacement therapies, corticosteroids, and isotretinoin.6 Pruritus and tenderness may or may not be present, and the Köbner phenomenon may occur.7
Continue to: The differential diagnosis
The differential diagnosis for eruptive xanthoma includes xanthoma disseminatum, non–Langerhans cell histiocytoses (eg, generalized eruptive histiocytosis), and cutaneous mastocytosis.1
Xanthoma disseminatum is an extremely rare, but benign, disorder of non–Langerhans cell origin. The average age of onset is older than 40 years. The rash consists of multiple red-yellow papules and nodules that most commonly present in flexural areas. Forty percent to 60% of patients have mucosal involvement, and rarely the central nervous system is involved.8
Generalized eruptive histiocytosis is another rare non–Langerhans cell histiocytosis that occurs mainly in adults and is characterized by widespread, symmetric, red-brown papules on the trunk, arms, and legs, and rarely the mucous membranes.9
Cutaneous mastocytosis, especially xanthelasmoid mastocytosis, consists of multiple pruritic, yellowish, papular or nodular lesions that may mimic eruptive xanthoma. It occurs mainly in children and rarely in adults.10
Confirming the diagnosis, initiating treatment
The diagnosis of eruptive xanthoma can be confirmed by skin biopsy if other differential diagnoses cannot be ruled out or the lesions do not resolve with treatment. Skin biopsy will reveal lipid-laden macrophages (known as foam cells) deposited in the dermis.7
Continue to: Treatment of eruptive xanthoma
Treatment of eruptive xanthoma involves management of the underlying causes of the condition. In most cases, dietary control, intensive triglyceride-lowering therapies, and treatment of other secondary causes of hypertriglyceridemia result in complete resolution of the lesions within several weeks.5
Our patient’s outcome
Our patient’s sudden-onset rash alerted us to the presence of type 2 diabetes mellitus, hypertriglyceridemia, and heavy proteinuria, which he was not aware of previously. We counselled him about stringent low-sugar, low-lipid diet control and exercise, and we started him on metformin and gemfibrozil. He was referred to an internal medicine specialist for further assessment and management of his severe hypertriglyceridemia and heavy proteinuria.
The rash started to wane 1 month after the patient started the metformin and gemfibrozil, and his drug regimen was changed to combination therapy with metformin/glimepiride and fenofibrate/simvastatin 6 weeks later when he was seen in the medical specialty clinic. Fundus photography performed 1 month after starting oral antidiabetic therapy showed no diabetic retinopathy or lipemia retinalis.
After 3 months of treatment, his serum triglycerides and hemoglobin A1c levels dropped to 3.8 mmol/L and 8.7%, respectively. The rash also resolved considerably, with only residual papules on the abdomen. This rapid clinical response to treatment of the underlying hypertriglyceridemia and diabetes further supported the clinical diagnosis of eruptive xanthoma.
THE TAKEAWAY
Eruptive xanthoma is relatively rare, but it is important for family physicians to recognize this clinical presentation as a potential indicator of severe hypertriglyceridemia. Recognizing hypertriglyceridemia early is important, as it can be associated with an increased risk for acute pancreatitis. Moreover, eruptive xanthoma might be the sole presenting symptom of underlying diabetes mellitus or familial hyperlipidemia, both of which can lead to a significant increase in cardiovascular risk if uncontrolled.
CORRESPONDENCE
Chan Kam Sum, MBChB, FRACGP, Tseung Kwan O Jockey Club General Out-patient Clinic, 99 Po Lam Road North, G/F, Tseung Kwan O, Kowloon, Hong Kong; [email protected]
THE CASE
A 37-year-old man presented with a sudden-onset, nonpruritic, nonpainful, papular rash of 1 month’s duration on his trunk and both arms and legs. Two weeks prior to the current presentation, he consulted a general practitioner, who treated the rash with a course of unknown oral antibiotics; the patient showed no improvement. He recalled that on a few occasions, he used his fingers to express a creamy discharge from some of the lesions. This temporarily reduced the size of those papules.
His medical history was unremarkable except for morbid obesity. He did not drink alcohol regularly and was not taking any medications prior to the onset of the rash. He had no family history of hyperlipidemia, but his mother had a history of diabetes mellitus.
Physical examination showed numerous discrete erythematous papules with a creamy center on his trunk and his arms and legs. The lesions were more numerous on the extensor surfaces of the arms and legs. Some of the papules coalesced to form small plaques (FIGURE). There was no scaling, and the lesions were firm in texture. The patient’s face was spared, and there was no mucosal involvement. The patient was otherwise systemically well.

THE DIAGNOSIS
Based on the morphology, distribution, and abrupt onset of the diffuse nonpruritic papules in this morbidly obese (but otherwise systemically well) middle-aged man, a clinical diagnosis of eruptive xanthoma was suspected. Subsequent blood testing revealed an elevated serum triglyceride level of 47.8 mmol/L (reference range, <1.7 mmol/L), elevated serum total cholesterol of 7.1 mmol/L (reference range, <6.2 mmol/L), and low serum high-density lipoprotein cholesterol of 0.7 mmol/L (reference range, >1 mmol/L in men). He also had an elevated fasting serum glucose level of 12.9 mmol/L (reference range, 3.9–5.6 mmol/L) and an elevated hemoglobin A1c (glycated hemoglobin) level of 10.9%.
Subsequent thyroid, liver, and renal function tests were normal, but the patient had heavy proteinuria, with an elevated urine albumin-to-creatinine ratio of 355.6 mg/mmol (reference range, ≤2.5 mg/mmol). The patient was referred to a dermatologist, who confirmed the clinical diagnosis without the need for a skin biopsy.
DISCUSSION

Eruptive xanthoma is characterized by an abrupt onset of crops of multiple yellowish to brownish papules that can coalesce into small plaques. The lesions can be generalized, but tend to be more densely distributed on the extensor surfaces of the arms and legs, buttocks, and thighs.5 Eruptive xanthoma often is associated with hypertriglyceridemia, which can be primary—as a result of a genetic defect caused by familial hypertriglyceridemia—or secondary, associated with poorly controlled diabetes mellitus, morbid obesity, excessive alcohol consumption, nephrotic syndrome, hypothyroidism, primary biliary cholangitis, and drugs like estrogen replacement therapies, corticosteroids, and isotretinoin.6 Pruritus and tenderness may or may not be present, and the Köbner phenomenon may occur.7
Continue to: The differential diagnosis
The differential diagnosis for eruptive xanthoma includes xanthoma disseminatum, non–Langerhans cell histiocytoses (eg, generalized eruptive histiocytosis), and cutaneous mastocytosis.1
Xanthoma disseminatum is an extremely rare, but benign, disorder of non–Langerhans cell origin. The average age of onset is older than 40 years. The rash consists of multiple red-yellow papules and nodules that most commonly present in flexural areas. Forty percent to 60% of patients have mucosal involvement, and rarely the central nervous system is involved.8
Generalized eruptive histiocytosis is another rare non–Langerhans cell histiocytosis that occurs mainly in adults and is characterized by widespread, symmetric, red-brown papules on the trunk, arms, and legs, and rarely the mucous membranes.9
Cutaneous mastocytosis, especially xanthelasmoid mastocytosis, consists of multiple pruritic, yellowish, papular or nodular lesions that may mimic eruptive xanthoma. It occurs mainly in children and rarely in adults.10
Confirming the diagnosis, initiating treatment
The diagnosis of eruptive xanthoma can be confirmed by skin biopsy if other differential diagnoses cannot be ruled out or the lesions do not resolve with treatment. Skin biopsy will reveal lipid-laden macrophages (known as foam cells) deposited in the dermis.7
Continue to: Treatment of eruptive xanthoma
Treatment of eruptive xanthoma involves management of the underlying causes of the condition. In most cases, dietary control, intensive triglyceride-lowering therapies, and treatment of other secondary causes of hypertriglyceridemia result in complete resolution of the lesions within several weeks.5
Our patient’s outcome
Our patient’s sudden-onset rash alerted us to the presence of type 2 diabetes mellitus, hypertriglyceridemia, and heavy proteinuria, which he was not aware of previously. We counselled him about stringent low-sugar, low-lipid diet control and exercise, and we started him on metformin and gemfibrozil. He was referred to an internal medicine specialist for further assessment and management of his severe hypertriglyceridemia and heavy proteinuria.
The rash started to wane 1 month after the patient started the metformin and gemfibrozil, and his drug regimen was changed to combination therapy with metformin/glimepiride and fenofibrate/simvastatin 6 weeks later when he was seen in the medical specialty clinic. Fundus photography performed 1 month after starting oral antidiabetic therapy showed no diabetic retinopathy or lipemia retinalis.
After 3 months of treatment, his serum triglycerides and hemoglobin A1c levels dropped to 3.8 mmol/L and 8.7%, respectively. The rash also resolved considerably, with only residual papules on the abdomen. This rapid clinical response to treatment of the underlying hypertriglyceridemia and diabetes further supported the clinical diagnosis of eruptive xanthoma.
THE TAKEAWAY
Eruptive xanthoma is relatively rare, but it is important for family physicians to recognize this clinical presentation as a potential indicator of severe hypertriglyceridemia. Recognizing hypertriglyceridemia early is important, as it can be associated with an increased risk for acute pancreatitis. Moreover, eruptive xanthoma might be the sole presenting symptom of underlying diabetes mellitus or familial hyperlipidemia, both of which can lead to a significant increase in cardiovascular risk if uncontrolled.
CORRESPONDENCE
Chan Kam Sum, MBChB, FRACGP, Tseung Kwan O Jockey Club General Out-patient Clinic, 99 Po Lam Road North, G/F, Tseung Kwan O, Kowloon, Hong Kong; [email protected]
1. Tang WK. Eruptive xanthoma. [case reports]. Hong Kong Dermatol Venereol Bull. 2001;9:172-175.
2. Frew J, Murrell D, Haber R. Fifty shades of yellow: a review of the xanthodermatoses. Int J Dermatol. 2015;54:1109-1123.
3. Zak A, Zeman M, Slaby A, et al. Xanthomas: clinical and pathophysiological relations. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2014;158:181-188.
4. Sandhu S, Al-Sarraf A, Taraboanta C, et al. Incidence of pancreatitis, secondary causes, and treatment of patients referred to specialty lipid clinic with severe hypertriglyceridemia: a retrospective cohort study. Lipids Health Dis. 2011;10:157.
5. Holsinger JM, Campbell SM, Witman P. Multiple erythematous-yellow, dome-shaped papules. Am Fam Physician. 2010;82:517.
6. Loeckermann S, Braun-Falco M. Eruptive xanthomas in association with metabolic syndrome. Clin Exp Dermatol. 2010;35:565-566.
7. Merola JF, Mengden SJ, Soldano A, et al. Eruptive xanthomas. Dermatol Online J. 2008;14:10.
8. Park M, Boone B, Devas S. Xanthoma disseminatum: case report and mini-review of the literature. Acta Dermatovenerol Croat. 2014;22:150-154.
9. Attia A, Seleit I, El Badawy N, et al. Photoletter to the editor: generalized eruptive histiocytoma. J Dermatol Case Rep. 2011;5:53-55.
10. Nabavi NS, Nejad MH, Feli S, et al. Adult onset of xanthelasmoid mastocytosis: report of a rare entity. Indian J Dermatol. 2016;61:468.
1. Tang WK. Eruptive xanthoma. [case reports]. Hong Kong Dermatol Venereol Bull. 2001;9:172-175.
2. Frew J, Murrell D, Haber R. Fifty shades of yellow: a review of the xanthodermatoses. Int J Dermatol. 2015;54:1109-1123.
3. Zak A, Zeman M, Slaby A, et al. Xanthomas: clinical and pathophysiological relations. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2014;158:181-188.
4. Sandhu S, Al-Sarraf A, Taraboanta C, et al. Incidence of pancreatitis, secondary causes, and treatment of patients referred to specialty lipid clinic with severe hypertriglyceridemia: a retrospective cohort study. Lipids Health Dis. 2011;10:157.
5. Holsinger JM, Campbell SM, Witman P. Multiple erythematous-yellow, dome-shaped papules. Am Fam Physician. 2010;82:517.
6. Loeckermann S, Braun-Falco M. Eruptive xanthomas in association with metabolic syndrome. Clin Exp Dermatol. 2010;35:565-566.
7. Merola JF, Mengden SJ, Soldano A, et al. Eruptive xanthomas. Dermatol Online J. 2008;14:10.
8. Park M, Boone B, Devas S. Xanthoma disseminatum: case report and mini-review of the literature. Acta Dermatovenerol Croat. 2014;22:150-154.
9. Attia A, Seleit I, El Badawy N, et al. Photoletter to the editor: generalized eruptive histiocytoma. J Dermatol Case Rep. 2011;5:53-55.
10. Nabavi NS, Nejad MH, Feli S, et al. Adult onset of xanthelasmoid mastocytosis: report of a rare entity. Indian J Dermatol. 2016;61:468.
Bariatric surgery + medical therapy: Effective Tx for T2DM?
ILLUSTRATIVE CASE
A 46-year-old woman presents with a body mass index (BMI) of 28 kg/m2, a 4-year history of type 2 diabetes mellitus (T2DM), and a glycated hemoglobin (HgbA1c) of 9.8%. The patient is currently being treated with intensive medical therapy (IMT), including metformin 2000 mg/d, sitagliptin 100 mg/d, and insulin glargine 12 units/d, with minimal change in HgbA1c. Should you recommend bariatric surgery as an option for the treatment of diabetes?
One in 11 Americans has diabetes and at least 95% of those have type 2.2,3 The treatment of T2DM is generally multimodal in order to target the various mechanisms that cause hyperglycemia. Treatment strategies may include lifestyle modifications, decreasing insulin resistance, increasing secretion of insulin, insulin replacement, and targeting incretin-hormonal pathways.
The American Diabetes Association (ADA) currently recommends diet, exercise, and behavioral modifications as first-line therapy for the management of diabetes,2 but these by themselves are often inadequate. In addition to various pharmacotherapeutic strategies for other populations with T2DM (see the PURL, “How do these 3 diabetes agents compare in reducing mortality?”), the ADA recommends bariatric surgery for the treatment of patients with T2DM, a BMI ≥35 kg/m2, and uncontrolled hyperglycemia.2,4 However, this recommendation from the ADA supporting bariatric surgery is based only on short-term studies.
For example, one single-center nonblinded randomized controlled trial (RCT) involving 60 patients with a BMI ≥35 kg/m2 found reductions in HgbA1C levels from the average baseline of 8.65±1.45% to 7.7±0.6% in the IMT group and to 6.4±1.4% in the gastric-bypass group at 2 years.5 In another study, a randomized double-blind trial involving 60 moderately obese patients (BMI, 25-35 kg/m2), gastric bypass had better outcomes than sleeve gastrectomy, with 93% of patients in the gastric bypass group achieving remission of T2DM vs 47% of patients in the sleeve gastrectomy group (P=.02) over a 12-month period.6
The current study sought to examine the long-term outcomes of IMT alone vs bariatric surgery with IMT for the treatment of T2DM in patients who are overweight or obese.1
STUDY SUMMARY
5-year follow-up shows surgery + intensive medical therapy works
This study by Schauer et al was a 5-year follow-up of a nonblinded, single-center RCT comparing IMT alone to IMT with Roux-en-Y gastric bypass or sleeve gastrectomy in 150 patients with T2DM.1 Patients were included if they were 20 to 60 years of age, had a BMI of 27 to 43 kg/m2, and had an HgbA1C >7%. Patients with previous bariatric surgery, complex abdominal surgery, or uncontrolled medical or psychiatric disorders were excluded.
Each patient was randomly placed in a 1:1:1 fashion into 3 groups: IMT only, IMT and gastric bypass, or IMT and sleeve gastrectomy. All patients underwent IMT as defined by the ADA. The primary outcome was the number of patients with an HgbA1c ≤6%. Secondary outcomes included weight loss, glucose control, lipid levels, blood pressure, medication use, renal function, adverse effects, ophthalmologic outcomes, and quality of life.
Continue to: Of the 150 patients...
Of the 150 patients, 1 died during the follow-up period leaving 149; 134 completed the 5-year follow-up; 8 patients in the IMT group and 1 patient in the sleeve gastrectomy group never initiated assigned treatment; an additional 6 patients were lost to follow-up. One patient from the IMT group and 1 patient from the sleeve gastrectomy group crossed over to the gastric bypass group.
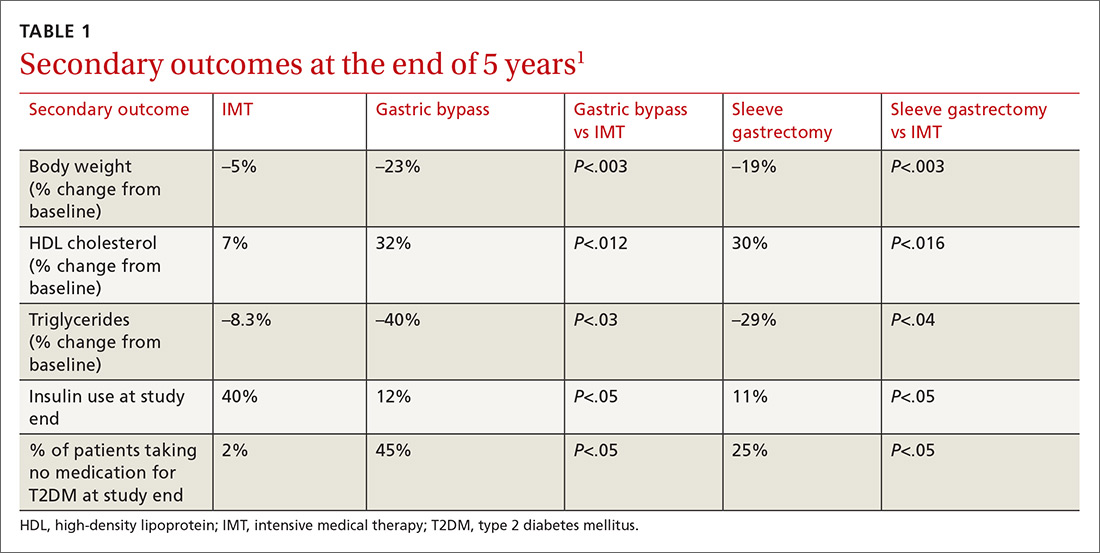
Results. More patients in the bariatric surgery and sleeve gastrectomy groups achieved an HgbA1c of ≤6% compared with the IMT group (14 of 49 gastric bypass patients vs 2 of 38 IMT patients; P=.01; 11 of 47 sleeve gastrectomy patients vs 2 of 38 IMT patients; P=.03). Compared with those in the IMT group, the patients in the bariatric surgery and sleeve gastrectomy groups showed greater reductions from baseline in body weight and triglyceride levels, and greater increases from baseline in high-density lipoprotein (HDL) cholesterol levels; they also required less diabetic medication for glycemic control (see TABLE 11). However, when data were imputed for the intention-to-treat analysis, P-values were P=0.08 for gastric bypass and P=0.17 for sleeve gastrectomy compared with the IMT group for lowering HgbA1c.
WHAT’S NEW?
Adding surgery has big benefits with minimal adverse effects
Prior studies that evaluated the effect of gastric bypass surgery on diabetes were observational or had a shorter follow-up duration. This study demonstrates bariatric surgery plus IMT has long-term benefits with minimal adverse events compared with IMT alone.1,5 Additionally, this study supports recommendations for bariatric surgery as treatment for T2DM for patients with a BMI ≥27 kg/m2, which is below the starting BMI (35 kg/m2) recommended by the ADA.1,4
CAVEATS
Surgery is not without risks
The risk for surgical complications, such as gastrointestinal bleeding, severe hypoglycemia requiring intervention, and ketoacidosis, in this patient population is significant.1 Complications can include gastrointestinal leak, stroke, and infection.1 Additionally, long-term complications from bariatric surgery are emerging and include choledocholithiasis, intestinal obstruction, and esophageal pathology.7 Extensive patient counseling regarding the possible complications is necessary to ensure that patients make an informed decision regarding surgery.
This study utilized surrogate markers (A1c, lipid levels, and body weight) as disease-oriented outcome measures. Patient-oriented outcomes, such as morbidity and mortality, were not explored in this study.
Continue to: Due to the small sample size of the study...
Due to the small sample size of the study, it is unclear if the outcomes of the 2 surgery groups were significantly different. Patients who received gastric bypass surgery had more weight loss and used less diabetes medication at the end of follow-up compared with the patients who received sleeve gastrectomy. More information is needed to determine which gastric surgery is preferable for the treatment of T2DM while minimizing adverse effects. However, both of the procedures had outcomes superior to that with IMT, and selection of a particular type of surgery should be a joint decision between the patient and provider.
CHALLENGES TO IMPLEMENTATION
Access and cost may be barriers
The major barriers to implementation are access to, and the cost of, bariatric surgery.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. Schauer PR, Bhatt DL, Kirwan JP, et al; STAMPEDE Investigators. Bariatric surgery versus intensive medical therapy for diabetes—5-year outcomes. N Engl J Med. 2017;376:641-651.
2. American Diabetes Asssociation. Obesity management for the treatment of type 2 diabetes: standards of medical care in diabetes—2019. Diabetes Care. 2019;42 (suppl 1):S81-S89.
3. Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2017. Atlanta, GA: Centers for Disease Control and Prevention, US Department of Health and Human Services; 2017. www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed March 1, 2019.
4. Rubino F, Nathan DM, Eckel RH, et al. Metabolic surgery in the treatment algorithm for type 2 diabetes: a joint statement by international diabetes organizations. Diabetes Care. 2016;39:861-877.
5. Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med. 2012;366:1577-1585.
6. Lee WJ, Chong K, Ser KH, et al. Gastric bypass vs sleeve gastrectomy for type 2 diabetes mellitus: a randomized controlled trial. Arch Surg. 2011;146:143-148.
7. Schulman AR, Thompson CC. Complications of bariatric surgery: what you can expect to see in your GI practice. Am J Gastroenterol. 2017;112:1640-1655.
ILLUSTRATIVE CASE
A 46-year-old woman presents with a body mass index (BMI) of 28 kg/m2, a 4-year history of type 2 diabetes mellitus (T2DM), and a glycated hemoglobin (HgbA1c) of 9.8%. The patient is currently being treated with intensive medical therapy (IMT), including metformin 2000 mg/d, sitagliptin 100 mg/d, and insulin glargine 12 units/d, with minimal change in HgbA1c. Should you recommend bariatric surgery as an option for the treatment of diabetes?
One in 11 Americans has diabetes and at least 95% of those have type 2.2,3 The treatment of T2DM is generally multimodal in order to target the various mechanisms that cause hyperglycemia. Treatment strategies may include lifestyle modifications, decreasing insulin resistance, increasing secretion of insulin, insulin replacement, and targeting incretin-hormonal pathways.
The American Diabetes Association (ADA) currently recommends diet, exercise, and behavioral modifications as first-line therapy for the management of diabetes,2 but these by themselves are often inadequate. In addition to various pharmacotherapeutic strategies for other populations with T2DM (see the PURL, “How do these 3 diabetes agents compare in reducing mortality?”), the ADA recommends bariatric surgery for the treatment of patients with T2DM, a BMI ≥35 kg/m2, and uncontrolled hyperglycemia.2,4 However, this recommendation from the ADA supporting bariatric surgery is based only on short-term studies.
For example, one single-center nonblinded randomized controlled trial (RCT) involving 60 patients with a BMI ≥35 kg/m2 found reductions in HgbA1C levels from the average baseline of 8.65±1.45% to 7.7±0.6% in the IMT group and to 6.4±1.4% in the gastric-bypass group at 2 years.5 In another study, a randomized double-blind trial involving 60 moderately obese patients (BMI, 25-35 kg/m2), gastric bypass had better outcomes than sleeve gastrectomy, with 93% of patients in the gastric bypass group achieving remission of T2DM vs 47% of patients in the sleeve gastrectomy group (P=.02) over a 12-month period.6
The current study sought to examine the long-term outcomes of IMT alone vs bariatric surgery with IMT for the treatment of T2DM in patients who are overweight or obese.1
STUDY SUMMARY
5-year follow-up shows surgery + intensive medical therapy works
This study by Schauer et al was a 5-year follow-up of a nonblinded, single-center RCT comparing IMT alone to IMT with Roux-en-Y gastric bypass or sleeve gastrectomy in 150 patients with T2DM.1 Patients were included if they were 20 to 60 years of age, had a BMI of 27 to 43 kg/m2, and had an HgbA1C >7%. Patients with previous bariatric surgery, complex abdominal surgery, or uncontrolled medical or psychiatric disorders were excluded.
Each patient was randomly placed in a 1:1:1 fashion into 3 groups: IMT only, IMT and gastric bypass, or IMT and sleeve gastrectomy. All patients underwent IMT as defined by the ADA. The primary outcome was the number of patients with an HgbA1c ≤6%. Secondary outcomes included weight loss, glucose control, lipid levels, blood pressure, medication use, renal function, adverse effects, ophthalmologic outcomes, and quality of life.
Continue to: Of the 150 patients...
Of the 150 patients, 1 died during the follow-up period leaving 149; 134 completed the 5-year follow-up; 8 patients in the IMT group and 1 patient in the sleeve gastrectomy group never initiated assigned treatment; an additional 6 patients were lost to follow-up. One patient from the IMT group and 1 patient from the sleeve gastrectomy group crossed over to the gastric bypass group.
Results. More patients in the bariatric surgery and sleeve gastrectomy groups achieved an HgbA1c of ≤6% compared with the IMT group (14 of 49 gastric bypass patients vs 2 of 38 IMT patients; P=.01; 11 of 47 sleeve gastrectomy patients vs 2 of 38 IMT patients; P=.03). Compared with those in the IMT group, the patients in the bariatric surgery and sleeve gastrectomy groups showed greater reductions from baseline in body weight and triglyceride levels, and greater increases from baseline in high-density lipoprotein (HDL) cholesterol levels; they also required less diabetic medication for glycemic control (see TABLE 11). However, when data were imputed for the intention-to-treat analysis, P-values were P=0.08 for gastric bypass and P=0.17 for sleeve gastrectomy compared with the IMT group for lowering HgbA1c.

WHAT’S NEW?
Adding surgery has big benefits with minimal adverse effects
Prior studies that evaluated the effect of gastric bypass surgery on diabetes were observational or had a shorter follow-up duration. This study demonstrates bariatric surgery plus IMT has long-term benefits with minimal adverse events compared with IMT alone.1,5 Additionally, this study supports recommendations for bariatric surgery as treatment for T2DM for patients with a BMI ≥27 kg/m2, which is below the starting BMI (35 kg/m2) recommended by the ADA.1,4
CAVEATS
Surgery is not without risks
The risk for surgical complications, such as gastrointestinal bleeding, severe hypoglycemia requiring intervention, and ketoacidosis, in this patient population is significant.1 Complications can include gastrointestinal leak, stroke, and infection.1 Additionally, long-term complications from bariatric surgery are emerging and include choledocholithiasis, intestinal obstruction, and esophageal pathology.7 Extensive patient counseling regarding the possible complications is necessary to ensure that patients make an informed decision regarding surgery.
This study utilized surrogate markers (A1c, lipid levels, and body weight) as disease-oriented outcome measures. Patient-oriented outcomes, such as morbidity and mortality, were not explored in this study.
Continue to: Due to the small sample size of the study...
Due to the small sample size of the study, it is unclear if the outcomes of the 2 surgery groups were significantly different. Patients who received gastric bypass surgery had more weight loss and used less diabetes medication at the end of follow-up compared with the patients who received sleeve gastrectomy. More information is needed to determine which gastric surgery is preferable for the treatment of T2DM while minimizing adverse effects. However, both of the procedures had outcomes superior to that with IMT, and selection of a particular type of surgery should be a joint decision between the patient and provider.
CHALLENGES TO IMPLEMENTATION
Access and cost may be barriers
The major barriers to implementation are access to, and the cost of, bariatric surgery.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
A 46-year-old woman presents with a body mass index (BMI) of 28 kg/m2, a 4-year history of type 2 diabetes mellitus (T2DM), and a glycated hemoglobin (HgbA1c) of 9.8%. The patient is currently being treated with intensive medical therapy (IMT), including metformin 2000 mg/d, sitagliptin 100 mg/d, and insulin glargine 12 units/d, with minimal change in HgbA1c. Should you recommend bariatric surgery as an option for the treatment of diabetes?
One in 11 Americans has diabetes and at least 95% of those have type 2.2,3 The treatment of T2DM is generally multimodal in order to target the various mechanisms that cause hyperglycemia. Treatment strategies may include lifestyle modifications, decreasing insulin resistance, increasing secretion of insulin, insulin replacement, and targeting incretin-hormonal pathways.
The American Diabetes Association (ADA) currently recommends diet, exercise, and behavioral modifications as first-line therapy for the management of diabetes,2 but these by themselves are often inadequate. In addition to various pharmacotherapeutic strategies for other populations with T2DM (see the PURL, “How do these 3 diabetes agents compare in reducing mortality?”), the ADA recommends bariatric surgery for the treatment of patients with T2DM, a BMI ≥35 kg/m2, and uncontrolled hyperglycemia.2,4 However, this recommendation from the ADA supporting bariatric surgery is based only on short-term studies.
For example, one single-center nonblinded randomized controlled trial (RCT) involving 60 patients with a BMI ≥35 kg/m2 found reductions in HgbA1C levels from the average baseline of 8.65±1.45% to 7.7±0.6% in the IMT group and to 6.4±1.4% in the gastric-bypass group at 2 years.5 In another study, a randomized double-blind trial involving 60 moderately obese patients (BMI, 25-35 kg/m2), gastric bypass had better outcomes than sleeve gastrectomy, with 93% of patients in the gastric bypass group achieving remission of T2DM vs 47% of patients in the sleeve gastrectomy group (P=.02) over a 12-month period.6
The current study sought to examine the long-term outcomes of IMT alone vs bariatric surgery with IMT for the treatment of T2DM in patients who are overweight or obese.1
STUDY SUMMARY
5-year follow-up shows surgery + intensive medical therapy works
This study by Schauer et al was a 5-year follow-up of a nonblinded, single-center RCT comparing IMT alone to IMT with Roux-en-Y gastric bypass or sleeve gastrectomy in 150 patients with T2DM.1 Patients were included if they were 20 to 60 years of age, had a BMI of 27 to 43 kg/m2, and had an HgbA1C >7%. Patients with previous bariatric surgery, complex abdominal surgery, or uncontrolled medical or psychiatric disorders were excluded.
Each patient was randomly placed in a 1:1:1 fashion into 3 groups: IMT only, IMT and gastric bypass, or IMT and sleeve gastrectomy. All patients underwent IMT as defined by the ADA. The primary outcome was the number of patients with an HgbA1c ≤6%. Secondary outcomes included weight loss, glucose control, lipid levels, blood pressure, medication use, renal function, adverse effects, ophthalmologic outcomes, and quality of life.
Continue to: Of the 150 patients...
Of the 150 patients, 1 died during the follow-up period leaving 149; 134 completed the 5-year follow-up; 8 patients in the IMT group and 1 patient in the sleeve gastrectomy group never initiated assigned treatment; an additional 6 patients were lost to follow-up. One patient from the IMT group and 1 patient from the sleeve gastrectomy group crossed over to the gastric bypass group.
Results. More patients in the bariatric surgery and sleeve gastrectomy groups achieved an HgbA1c of ≤6% compared with the IMT group (14 of 49 gastric bypass patients vs 2 of 38 IMT patients; P=.01; 11 of 47 sleeve gastrectomy patients vs 2 of 38 IMT patients; P=.03). Compared with those in the IMT group, the patients in the bariatric surgery and sleeve gastrectomy groups showed greater reductions from baseline in body weight and triglyceride levels, and greater increases from baseline in high-density lipoprotein (HDL) cholesterol levels; they also required less diabetic medication for glycemic control (see TABLE 11). However, when data were imputed for the intention-to-treat analysis, P-values were P=0.08 for gastric bypass and P=0.17 for sleeve gastrectomy compared with the IMT group for lowering HgbA1c.

WHAT’S NEW?
Adding surgery has big benefits with minimal adverse effects
Prior studies that evaluated the effect of gastric bypass surgery on diabetes were observational or had a shorter follow-up duration. This study demonstrates bariatric surgery plus IMT has long-term benefits with minimal adverse events compared with IMT alone.1,5 Additionally, this study supports recommendations for bariatric surgery as treatment for T2DM for patients with a BMI ≥27 kg/m2, which is below the starting BMI (35 kg/m2) recommended by the ADA.1,4
CAVEATS
Surgery is not without risks
The risk for surgical complications, such as gastrointestinal bleeding, severe hypoglycemia requiring intervention, and ketoacidosis, in this patient population is significant.1 Complications can include gastrointestinal leak, stroke, and infection.1 Additionally, long-term complications from bariatric surgery are emerging and include choledocholithiasis, intestinal obstruction, and esophageal pathology.7 Extensive patient counseling regarding the possible complications is necessary to ensure that patients make an informed decision regarding surgery.
This study utilized surrogate markers (A1c, lipid levels, and body weight) as disease-oriented outcome measures. Patient-oriented outcomes, such as morbidity and mortality, were not explored in this study.
Continue to: Due to the small sample size of the study...
Due to the small sample size of the study, it is unclear if the outcomes of the 2 surgery groups were significantly different. Patients who received gastric bypass surgery had more weight loss and used less diabetes medication at the end of follow-up compared with the patients who received sleeve gastrectomy. More information is needed to determine which gastric surgery is preferable for the treatment of T2DM while minimizing adverse effects. However, both of the procedures had outcomes superior to that with IMT, and selection of a particular type of surgery should be a joint decision between the patient and provider.
CHALLENGES TO IMPLEMENTATION
Access and cost may be barriers
The major barriers to implementation are access to, and the cost of, bariatric surgery.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. Schauer PR, Bhatt DL, Kirwan JP, et al; STAMPEDE Investigators. Bariatric surgery versus intensive medical therapy for diabetes—5-year outcomes. N Engl J Med. 2017;376:641-651.
2. American Diabetes Asssociation. Obesity management for the treatment of type 2 diabetes: standards of medical care in diabetes—2019. Diabetes Care. 2019;42 (suppl 1):S81-S89.
3. Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2017. Atlanta, GA: Centers for Disease Control and Prevention, US Department of Health and Human Services; 2017. www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed March 1, 2019.
4. Rubino F, Nathan DM, Eckel RH, et al. Metabolic surgery in the treatment algorithm for type 2 diabetes: a joint statement by international diabetes organizations. Diabetes Care. 2016;39:861-877.
5. Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med. 2012;366:1577-1585.
6. Lee WJ, Chong K, Ser KH, et al. Gastric bypass vs sleeve gastrectomy for type 2 diabetes mellitus: a randomized controlled trial. Arch Surg. 2011;146:143-148.
7. Schulman AR, Thompson CC. Complications of bariatric surgery: what you can expect to see in your GI practice. Am J Gastroenterol. 2017;112:1640-1655.
1. Schauer PR, Bhatt DL, Kirwan JP, et al; STAMPEDE Investigators. Bariatric surgery versus intensive medical therapy for diabetes—5-year outcomes. N Engl J Med. 2017;376:641-651.
2. American Diabetes Asssociation. Obesity management for the treatment of type 2 diabetes: standards of medical care in diabetes—2019. Diabetes Care. 2019;42 (suppl 1):S81-S89.
3. Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2017. Atlanta, GA: Centers for Disease Control and Prevention, US Department of Health and Human Services; 2017. www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed March 1, 2019.
4. Rubino F, Nathan DM, Eckel RH, et al. Metabolic surgery in the treatment algorithm for type 2 diabetes: a joint statement by international diabetes organizations. Diabetes Care. 2016;39:861-877.
5. Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med. 2012;366:1577-1585.
6. Lee WJ, Chong K, Ser KH, et al. Gastric bypass vs sleeve gastrectomy for type 2 diabetes mellitus: a randomized controlled trial. Arch Surg. 2011;146:143-148.
7. Schulman AR, Thompson CC. Complications of bariatric surgery: what you can expect to see in your GI practice. Am J Gastroenterol. 2017;112:1640-1655.
PRACTICE CHANGER
Consider bariatric surgery with medical therapy as a treatment option for adults with uncontrolled type 2 diabetes and a body mass index ≥27 kg/m2.1
STRENGTH OF RECOMMENDATION
B: Based on a nonblinded, single-center, randomized controlled trial.
Schauer PR, Bhatt DL, Kirwan JP, et al; STAMPEDE Investigators. Bariatric surgery versus intensive medical therapy for diabetes—5-year outcomes. N Engl J Med. 2017;376:641-651.
How do these 3 diabetes agents compare in reducing mortality?
ILLUSTRATIVE CASE
A 64-year-old man with taype 2 diabetes mellitus (T2DM) presents for a follow-up visit. His point-of-care hemoglobin A1c is 9.5%, and he is currently taking only metformin 1000 mg bid. You are considering adding an SGLT-2 inhibitor, a GLP-1 agonist, or a dipeptidyl peptidase 4 (DPP-4) inhibitor to his treatment regimen. Which do you choose to better control his diabetes and reduce his all-cause and cardiovascular (CV) mortality risk?
Over the past several years, the number of patients with T2DM has continued to climb. In the United States, approximately 30 million people, or 1 of every 11, now struggles to reduce their blood sugar.2 As prevalence of the disease has increased, so has the number of medications available that are aimed at lowering blood sugar and improving diabetes control.2 In particular, the introduction of SGLT-2 inhibitors, GLP-1 agonists, and DPP-4 inhibitors over the past several years has produced an area of some clinical ambiguity, due to the lack of randomized controlled trials (RCTs) comparing their efficacy.
The “American Diabetes Association Standards of Medical Care in Diabetes” points specifically to the potential roles of the SGLT-2 inhibitors empagliflozin and canagliflozin, and the GLP-1 agonist liraglutide, as agents that should be added to metformin and lifestyle modification in patients with established atherosclerotic CV disease. They cite data indicating that these drugs reduce major adverse CV events and CV mortality in this population.3 Deciding among these 3 medications, however, is left to providers and patients. For dual therapy in patients with T2DM without CV disease who remain hyperglycemic despite metformin and lifestyle modifications, SGLT-2 inhibitors, GLP-1 agonists, and DPP-4 inhibitors are recommended equally, with the choice among them to be determined by “consideration of drug-specific effects and patient factors.”3
The National Institute for Health and Care Excellence (NICE) guidelines on T2DM management list both SGLT-2 inhibitors and DPP-4 inhibitors among the potential options for intensifying therapy after metformin.4 The American Association of Clinical Endocrinologists and the American College of Endocrinology guidelines do include a hierarchical recommendation to try a GLP-1 agonist first, followed by an SGLT-2 inhibitor, followed by a DPP-4 inhibitor, after metformin and lifestyle modifications—although the difference in strength of recommendations for these classes is noted to be small.5
STUDY SUMMARY
SGLT-2s, GLP-1s are associated with better mortality outcomes than DPP-4s
Zheng and colleagues performed a network meta-analysis of 236 RCTs involving 176,310 patients to compare the clinical efficacy of SGLT-2 inhibitors, GLP-1 agonists, and DPP-4 inhibitors to reduce all-cause mortality and CV endpoints in patients with T2DM. The authors analyzed English-language RCTs that followed patients with T2DM for at least 12 weeks and compared SGLT-2 inhibitors, GLP-1 agonists, and DPP-4 inhibitors to one another, to placebo, or to no treatment.
A majority of the patients in both the intervention and control groups were taking additional diabetes medications, such as metformin, prior to enrollment and during the trials. About half of the patients analyzed were enrolled in trials that specifically evaluated patients at elevated CV risk, which is notable because patients with higher CV risk ultimately derived the most benefit from the treatments studied.
The primary outcome was all-cause mortality. Secondary outcomes were CV mortality, heart failure (HF) events, myocardial infarction (MI), unstable angina, and stroke, as well as the safety outcomes of hypoglycemia and adverse events (any events, serious events, and those leading to study withdrawal).
Continue to: Results
Results. Compared with the patients in the control groups (placebo or no treatment), patients in both the SGLT-2 inhibitor and GLP-1 agonist groups had decreased all-cause mortality (SGLT-2 inhibitor group, hazard ratio [HR]=0.80; 95% credible interval [CrI], 0.71-0.89; absolute risk difference [RD]= –1%; number needed to treat [NNT]=100; GLP-1 agonist group, HR=0.88; 95% CrI, 0.81-0.94; absolute RD= -0.6%; NNT=167). Patients in the DPP-4 inhibitor group did not have a difference in mortality compared with the control groups (HR=1.02; 95% CrI, 0.94-1.11; absolute RD=0.1%). Both the SGLT-2 inhibitor (HR=0.78; 95% CrI, 0.68-0.90; absolute RD= –0.9%; NNT=111) and GLP-1 agonist (HR=0.86; 95% CrI, 0.77-0.96; absolute RD= –0.5%; NNT=200) groups had reduced all-cause mortality when compared with the DPP-4 inhibitor group.
CV endpoints. Similarly, the SGLT-2 inhibitor (HR=0.79; 95% Crl, 0.69-0.91; absolute RD= –0.8%; NNT=125) and GLP-1 agonist (HR=0.85; Crl, 95% 0.77-0.94; absolute RD= –0.5%; NNT=200) groups had a reduction in CV mortality compared with the control groups, while those in the DPP-4 inhibitor group experienced no effect. Additionally, those taking SGLT-2 inhibitors had lower rates of HF events (HR=0.62; 95% CrI, 0.54-0.72; absolute RD= –1.1%; NNT=91) and MIs (HR=0.86; 95% CrI, 0.77–0.97; absolute RD= –0.6%; NNT=167) than those in the control groups. They also had lower rates of HF than those taking GLP-1 agonists (HR=0.67; 95% CrI, 0.57 to 0.80; absolute RD= –0.9; NNT=111) or DPP-4 inhibitors (HR=0.55; 95% CrI, 0.46-0.67; absolute RD= –1.1%; NNT=91). Neither the GLP-1 agonist groups nor the DPP-4 inhibitor groups saw lower rates of HF or MI than the control groups.
Adverse effects. DPP-4 inhibitors, GLP-1 agonists, and SGLT-2 inhibitors were all associated with a small increased risk for hypoglycemia compared with the control groups, but there were no significant differences between drug classes. All agents resulted in an increased risk for adverse events leading to trial withdrawal compared with the control groups (GPL-1 agonists, HR=2; 95% CrI, 1.70-2.37; absolute RD=4.7%; number needed to harm [NNH]=21; SGLT-2 inhibitors, HR=1.8; 95% CrI, 1.44-2.25; absolute RD=5.8%; NNH=17; and DPP-4 inhibitors, HR=1.93; 95% CrI, 1.59-2.35; absolute RD=3.1%; NNH=32).
When compared with the control groups, the SGLT-2 inhibitor group was associated with an increased risk for genital infection (relative risk [RR]=4.19; 95% confidence interval [CI], 3.45-5.09; absolute RD=6%; NNH=16), but not of urinary tract infection or lower limb amputation, although the authors noted high heterogeneity among studies with regard to the limb amputation outcome. DPP-4 inhibitors were associated with an increased risk for acute pancreatitis (RR=1.58; 95% CI, 1.04-2.39; absolute RD=0.1%; NNH=1000) compared with control groups.
WHAT’S NEW
SGLT-2s: Lower mortality, fewer heart failure events
This meta-analysis concludes that when compared with placebo or no treatment, the use of SGLT-2 inhibitors or GLP-1 agonists is associated with lower all-cause mortality and lower CV mortality than is the use of DPP-4 inhibitors. Additionally, SGLT-2 inhibitors are associated with lower rates of HF events than GLP-1 agonists or DPP-4 inhibitors.
Continue to: CAVEATS
CAVEATS
A lack of head-to-head RCTs
This study was a network meta-analysis that included many trials, the majority of which compared SGLT-1 inhibitors, GLP-1 agonists, and DPP-4 inhibitors with controls rather than to one another. Thus, the findings are not derived from a robust base of head-to-head RCTs involving the 3 classes of medication.
However, there was relatively low heterogeneity among the studies included (I2=12), which lends strength to the meta-analysis.6 Patients with the highest baseline CV risk likely gleaned the greatest benefits from these treatments and may have driven much of the observed mortality reduction. This may limit the generalizability of the results to people with low CV risk. The comparative effectiveness and risk for adverse effects among individual medications within each class is unknown because the analysis was completed by drug class in order to adequately power the study to detect treatment effects.
CHALLENGES TO IMPLEMENTATION
Cost, adverse effects, and formulation may represent challenges
The cost of SGLT-2 inhibitors and GLP-1 agonists may present challenges to patients wishing to use these options. Additionally, the increased risk for genital infections with SGLT-2 inhibitors, and of overall adverse effects (many of which were gastrointestinal) with GLP-1 agonists, must be considered. Lastly, the injectable formulation of GLP-1 agonists may present a barrier to patients’ ability and willingness to effectively administer these agents.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. Zheng S, Roddick A, Aghar-Jaffar R, et al. Association between use of sodium-glucose cotransporter 2 inhibitors, glucagon-like peptide 1 agonists, and dipeptidyl peptidase 4 inhibitors with all-cause mortality in patients with type 2 diabetes: a systematic review and meta-analysis. JAMA. 2018;319:1580-1591.
2. Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2017. Atlanta, GA: Centers for Disease Control and Prevention, US Department of Health and Human Services; 2017.
3. American Diabetes Association. Standards of medical care in diabetes–2019. Diabetes Care. 2019;42(suppl 1):S1-S193.
4. National Institute for Health and Care Excellence. Type 2 diabetes in adults: management. www.nice.org.uk/guidance/ng28. Published December 2015. Updated May 2017. Accessed March 1, 2019.
5. Garber A, Abrahamson M, Barzilay J, et al. Consensus Statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the Comprehensive Type 2 Diabetes Management Algorithm—2018 Executive Summary. Endocr Pract. 2018;24:91-120.
6. Salanti G, Del Giovane C, Chaimani A, et al. Evaluating the quality of evidence from a network meta-analysis. PLoS ONE. 2014;9:1-14.
ILLUSTRATIVE CASE
A 64-year-old man with taype 2 diabetes mellitus (T2DM) presents for a follow-up visit. His point-of-care hemoglobin A1c is 9.5%, and he is currently taking only metformin 1000 mg bid. You are considering adding an SGLT-2 inhibitor, a GLP-1 agonist, or a dipeptidyl peptidase 4 (DPP-4) inhibitor to his treatment regimen. Which do you choose to better control his diabetes and reduce his all-cause and cardiovascular (CV) mortality risk?
Over the past several years, the number of patients with T2DM has continued to climb. In the United States, approximately 30 million people, or 1 of every 11, now struggles to reduce their blood sugar.2 As prevalence of the disease has increased, so has the number of medications available that are aimed at lowering blood sugar and improving diabetes control.2 In particular, the introduction of SGLT-2 inhibitors, GLP-1 agonists, and DPP-4 inhibitors over the past several years has produced an area of some clinical ambiguity, due to the lack of randomized controlled trials (RCTs) comparing their efficacy.
The “American Diabetes Association Standards of Medical Care in Diabetes” points specifically to the potential roles of the SGLT-2 inhibitors empagliflozin and canagliflozin, and the GLP-1 agonist liraglutide, as agents that should be added to metformin and lifestyle modification in patients with established atherosclerotic CV disease. They cite data indicating that these drugs reduce major adverse CV events and CV mortality in this population.3 Deciding among these 3 medications, however, is left to providers and patients. For dual therapy in patients with T2DM without CV disease who remain hyperglycemic despite metformin and lifestyle modifications, SGLT-2 inhibitors, GLP-1 agonists, and DPP-4 inhibitors are recommended equally, with the choice among them to be determined by “consideration of drug-specific effects and patient factors.”3
The National Institute for Health and Care Excellence (NICE) guidelines on T2DM management list both SGLT-2 inhibitors and DPP-4 inhibitors among the potential options for intensifying therapy after metformin.4 The American Association of Clinical Endocrinologists and the American College of Endocrinology guidelines do include a hierarchical recommendation to try a GLP-1 agonist first, followed by an SGLT-2 inhibitor, followed by a DPP-4 inhibitor, after metformin and lifestyle modifications—although the difference in strength of recommendations for these classes is noted to be small.5
STUDY SUMMARY
SGLT-2s, GLP-1s are associated with better mortality outcomes than DPP-4s
Zheng and colleagues performed a network meta-analysis of 236 RCTs involving 176,310 patients to compare the clinical efficacy of SGLT-2 inhibitors, GLP-1 agonists, and DPP-4 inhibitors to reduce all-cause mortality and CV endpoints in patients with T2DM. The authors analyzed English-language RCTs that followed patients with T2DM for at least 12 weeks and compared SGLT-2 inhibitors, GLP-1 agonists, and DPP-4 inhibitors to one another, to placebo, or to no treatment.
A majority of the patients in both the intervention and control groups were taking additional diabetes medications, such as metformin, prior to enrollment and during the trials. About half of the patients analyzed were enrolled in trials that specifically evaluated patients at elevated CV risk, which is notable because patients with higher CV risk ultimately derived the most benefit from the treatments studied.
The primary outcome was all-cause mortality. Secondary outcomes were CV mortality, heart failure (HF) events, myocardial infarction (MI), unstable angina, and stroke, as well as the safety outcomes of hypoglycemia and adverse events (any events, serious events, and those leading to study withdrawal).
Continue to: Results
Results. Compared with the patients in the control groups (placebo or no treatment), patients in both the SGLT-2 inhibitor and GLP-1 agonist groups had decreased all-cause mortality (SGLT-2 inhibitor group, hazard ratio [HR]=0.80; 95% credible interval [CrI], 0.71-0.89; absolute risk difference [RD]= –1%; number needed to treat [NNT]=100; GLP-1 agonist group, HR=0.88; 95% CrI, 0.81-0.94; absolute RD= -0.6%; NNT=167). Patients in the DPP-4 inhibitor group did not have a difference in mortality compared with the control groups (HR=1.02; 95% CrI, 0.94-1.11; absolute RD=0.1%). Both the SGLT-2 inhibitor (HR=0.78; 95% CrI, 0.68-0.90; absolute RD= –0.9%; NNT=111) and GLP-1 agonist (HR=0.86; 95% CrI, 0.77-0.96; absolute RD= –0.5%; NNT=200) groups had reduced all-cause mortality when compared with the DPP-4 inhibitor group.
CV endpoints. Similarly, the SGLT-2 inhibitor (HR=0.79; 95% Crl, 0.69-0.91; absolute RD= –0.8%; NNT=125) and GLP-1 agonist (HR=0.85; Crl, 95% 0.77-0.94; absolute RD= –0.5%; NNT=200) groups had a reduction in CV mortality compared with the control groups, while those in the DPP-4 inhibitor group experienced no effect. Additionally, those taking SGLT-2 inhibitors had lower rates of HF events (HR=0.62; 95% CrI, 0.54-0.72; absolute RD= –1.1%; NNT=91) and MIs (HR=0.86; 95% CrI, 0.77–0.97; absolute RD= –0.6%; NNT=167) than those in the control groups. They also had lower rates of HF than those taking GLP-1 agonists (HR=0.67; 95% CrI, 0.57 to 0.80; absolute RD= –0.9; NNT=111) or DPP-4 inhibitors (HR=0.55; 95% CrI, 0.46-0.67; absolute RD= –1.1%; NNT=91). Neither the GLP-1 agonist groups nor the DPP-4 inhibitor groups saw lower rates of HF or MI than the control groups.
Adverse effects. DPP-4 inhibitors, GLP-1 agonists, and SGLT-2 inhibitors were all associated with a small increased risk for hypoglycemia compared with the control groups, but there were no significant differences between drug classes. All agents resulted in an increased risk for adverse events leading to trial withdrawal compared with the control groups (GPL-1 agonists, HR=2; 95% CrI, 1.70-2.37; absolute RD=4.7%; number needed to harm [NNH]=21; SGLT-2 inhibitors, HR=1.8; 95% CrI, 1.44-2.25; absolute RD=5.8%; NNH=17; and DPP-4 inhibitors, HR=1.93; 95% CrI, 1.59-2.35; absolute RD=3.1%; NNH=32).
When compared with the control groups, the SGLT-2 inhibitor group was associated with an increased risk for genital infection (relative risk [RR]=4.19; 95% confidence interval [CI], 3.45-5.09; absolute RD=6%; NNH=16), but not of urinary tract infection or lower limb amputation, although the authors noted high heterogeneity among studies with regard to the limb amputation outcome. DPP-4 inhibitors were associated with an increased risk for acute pancreatitis (RR=1.58; 95% CI, 1.04-2.39; absolute RD=0.1%; NNH=1000) compared with control groups.
WHAT’S NEW
SGLT-2s: Lower mortality, fewer heart failure events
This meta-analysis concludes that when compared with placebo or no treatment, the use of SGLT-2 inhibitors or GLP-1 agonists is associated with lower all-cause mortality and lower CV mortality than is the use of DPP-4 inhibitors. Additionally, SGLT-2 inhibitors are associated with lower rates of HF events than GLP-1 agonists or DPP-4 inhibitors.
Continue to: CAVEATS
CAVEATS
A lack of head-to-head RCTs
This study was a network meta-analysis that included many trials, the majority of which compared SGLT-1 inhibitors, GLP-1 agonists, and DPP-4 inhibitors with controls rather than to one another. Thus, the findings are not derived from a robust base of head-to-head RCTs involving the 3 classes of medication.
However, there was relatively low heterogeneity among the studies included (I2=12), which lends strength to the meta-analysis.6 Patients with the highest baseline CV risk likely gleaned the greatest benefits from these treatments and may have driven much of the observed mortality reduction. This may limit the generalizability of the results to people with low CV risk. The comparative effectiveness and risk for adverse effects among individual medications within each class is unknown because the analysis was completed by drug class in order to adequately power the study to detect treatment effects.
CHALLENGES TO IMPLEMENTATION
Cost, adverse effects, and formulation may represent challenges
The cost of SGLT-2 inhibitors and GLP-1 agonists may present challenges to patients wishing to use these options. Additionally, the increased risk for genital infections with SGLT-2 inhibitors, and of overall adverse effects (many of which were gastrointestinal) with GLP-1 agonists, must be considered. Lastly, the injectable formulation of GLP-1 agonists may present a barrier to patients’ ability and willingness to effectively administer these agents.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
A 64-year-old man with taype 2 diabetes mellitus (T2DM) presents for a follow-up visit. His point-of-care hemoglobin A1c is 9.5%, and he is currently taking only metformin 1000 mg bid. You are considering adding an SGLT-2 inhibitor, a GLP-1 agonist, or a dipeptidyl peptidase 4 (DPP-4) inhibitor to his treatment regimen. Which do you choose to better control his diabetes and reduce his all-cause and cardiovascular (CV) mortality risk?
Over the past several years, the number of patients with T2DM has continued to climb. In the United States, approximately 30 million people, or 1 of every 11, now struggles to reduce their blood sugar.2 As prevalence of the disease has increased, so has the number of medications available that are aimed at lowering blood sugar and improving diabetes control.2 In particular, the introduction of SGLT-2 inhibitors, GLP-1 agonists, and DPP-4 inhibitors over the past several years has produced an area of some clinical ambiguity, due to the lack of randomized controlled trials (RCTs) comparing their efficacy.
The “American Diabetes Association Standards of Medical Care in Diabetes” points specifically to the potential roles of the SGLT-2 inhibitors empagliflozin and canagliflozin, and the GLP-1 agonist liraglutide, as agents that should be added to metformin and lifestyle modification in patients with established atherosclerotic CV disease. They cite data indicating that these drugs reduce major adverse CV events and CV mortality in this population.3 Deciding among these 3 medications, however, is left to providers and patients. For dual therapy in patients with T2DM without CV disease who remain hyperglycemic despite metformin and lifestyle modifications, SGLT-2 inhibitors, GLP-1 agonists, and DPP-4 inhibitors are recommended equally, with the choice among them to be determined by “consideration of drug-specific effects and patient factors.”3
The National Institute for Health and Care Excellence (NICE) guidelines on T2DM management list both SGLT-2 inhibitors and DPP-4 inhibitors among the potential options for intensifying therapy after metformin.4 The American Association of Clinical Endocrinologists and the American College of Endocrinology guidelines do include a hierarchical recommendation to try a GLP-1 agonist first, followed by an SGLT-2 inhibitor, followed by a DPP-4 inhibitor, after metformin and lifestyle modifications—although the difference in strength of recommendations for these classes is noted to be small.5
STUDY SUMMARY
SGLT-2s, GLP-1s are associated with better mortality outcomes than DPP-4s
Zheng and colleagues performed a network meta-analysis of 236 RCTs involving 176,310 patients to compare the clinical efficacy of SGLT-2 inhibitors, GLP-1 agonists, and DPP-4 inhibitors to reduce all-cause mortality and CV endpoints in patients with T2DM. The authors analyzed English-language RCTs that followed patients with T2DM for at least 12 weeks and compared SGLT-2 inhibitors, GLP-1 agonists, and DPP-4 inhibitors to one another, to placebo, or to no treatment.
A majority of the patients in both the intervention and control groups were taking additional diabetes medications, such as metformin, prior to enrollment and during the trials. About half of the patients analyzed were enrolled in trials that specifically evaluated patients at elevated CV risk, which is notable because patients with higher CV risk ultimately derived the most benefit from the treatments studied.
The primary outcome was all-cause mortality. Secondary outcomes were CV mortality, heart failure (HF) events, myocardial infarction (MI), unstable angina, and stroke, as well as the safety outcomes of hypoglycemia and adverse events (any events, serious events, and those leading to study withdrawal).
Continue to: Results
Results. Compared with the patients in the control groups (placebo or no treatment), patients in both the SGLT-2 inhibitor and GLP-1 agonist groups had decreased all-cause mortality (SGLT-2 inhibitor group, hazard ratio [HR]=0.80; 95% credible interval [CrI], 0.71-0.89; absolute risk difference [RD]= –1%; number needed to treat [NNT]=100; GLP-1 agonist group, HR=0.88; 95% CrI, 0.81-0.94; absolute RD= -0.6%; NNT=167). Patients in the DPP-4 inhibitor group did not have a difference in mortality compared with the control groups (HR=1.02; 95% CrI, 0.94-1.11; absolute RD=0.1%). Both the SGLT-2 inhibitor (HR=0.78; 95% CrI, 0.68-0.90; absolute RD= –0.9%; NNT=111) and GLP-1 agonist (HR=0.86; 95% CrI, 0.77-0.96; absolute RD= –0.5%; NNT=200) groups had reduced all-cause mortality when compared with the DPP-4 inhibitor group.
CV endpoints. Similarly, the SGLT-2 inhibitor (HR=0.79; 95% Crl, 0.69-0.91; absolute RD= –0.8%; NNT=125) and GLP-1 agonist (HR=0.85; Crl, 95% 0.77-0.94; absolute RD= –0.5%; NNT=200) groups had a reduction in CV mortality compared with the control groups, while those in the DPP-4 inhibitor group experienced no effect. Additionally, those taking SGLT-2 inhibitors had lower rates of HF events (HR=0.62; 95% CrI, 0.54-0.72; absolute RD= –1.1%; NNT=91) and MIs (HR=0.86; 95% CrI, 0.77–0.97; absolute RD= –0.6%; NNT=167) than those in the control groups. They also had lower rates of HF than those taking GLP-1 agonists (HR=0.67; 95% CrI, 0.57 to 0.80; absolute RD= –0.9; NNT=111) or DPP-4 inhibitors (HR=0.55; 95% CrI, 0.46-0.67; absolute RD= –1.1%; NNT=91). Neither the GLP-1 agonist groups nor the DPP-4 inhibitor groups saw lower rates of HF or MI than the control groups.
Adverse effects. DPP-4 inhibitors, GLP-1 agonists, and SGLT-2 inhibitors were all associated with a small increased risk for hypoglycemia compared with the control groups, but there were no significant differences between drug classes. All agents resulted in an increased risk for adverse events leading to trial withdrawal compared with the control groups (GPL-1 agonists, HR=2; 95% CrI, 1.70-2.37; absolute RD=4.7%; number needed to harm [NNH]=21; SGLT-2 inhibitors, HR=1.8; 95% CrI, 1.44-2.25; absolute RD=5.8%; NNH=17; and DPP-4 inhibitors, HR=1.93; 95% CrI, 1.59-2.35; absolute RD=3.1%; NNH=32).
When compared with the control groups, the SGLT-2 inhibitor group was associated with an increased risk for genital infection (relative risk [RR]=4.19; 95% confidence interval [CI], 3.45-5.09; absolute RD=6%; NNH=16), but not of urinary tract infection or lower limb amputation, although the authors noted high heterogeneity among studies with regard to the limb amputation outcome. DPP-4 inhibitors were associated with an increased risk for acute pancreatitis (RR=1.58; 95% CI, 1.04-2.39; absolute RD=0.1%; NNH=1000) compared with control groups.
WHAT’S NEW
SGLT-2s: Lower mortality, fewer heart failure events
This meta-analysis concludes that when compared with placebo or no treatment, the use of SGLT-2 inhibitors or GLP-1 agonists is associated with lower all-cause mortality and lower CV mortality than is the use of DPP-4 inhibitors. Additionally, SGLT-2 inhibitors are associated with lower rates of HF events than GLP-1 agonists or DPP-4 inhibitors.
Continue to: CAVEATS
CAVEATS
A lack of head-to-head RCTs
This study was a network meta-analysis that included many trials, the majority of which compared SGLT-1 inhibitors, GLP-1 agonists, and DPP-4 inhibitors with controls rather than to one another. Thus, the findings are not derived from a robust base of head-to-head RCTs involving the 3 classes of medication.
However, there was relatively low heterogeneity among the studies included (I2=12), which lends strength to the meta-analysis.6 Patients with the highest baseline CV risk likely gleaned the greatest benefits from these treatments and may have driven much of the observed mortality reduction. This may limit the generalizability of the results to people with low CV risk. The comparative effectiveness and risk for adverse effects among individual medications within each class is unknown because the analysis was completed by drug class in order to adequately power the study to detect treatment effects.
CHALLENGES TO IMPLEMENTATION
Cost, adverse effects, and formulation may represent challenges
The cost of SGLT-2 inhibitors and GLP-1 agonists may present challenges to patients wishing to use these options. Additionally, the increased risk for genital infections with SGLT-2 inhibitors, and of overall adverse effects (many of which were gastrointestinal) with GLP-1 agonists, must be considered. Lastly, the injectable formulation of GLP-1 agonists may present a barrier to patients’ ability and willingness to effectively administer these agents.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. Zheng S, Roddick A, Aghar-Jaffar R, et al. Association between use of sodium-glucose cotransporter 2 inhibitors, glucagon-like peptide 1 agonists, and dipeptidyl peptidase 4 inhibitors with all-cause mortality in patients with type 2 diabetes: a systematic review and meta-analysis. JAMA. 2018;319:1580-1591.
2. Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2017. Atlanta, GA: Centers for Disease Control and Prevention, US Department of Health and Human Services; 2017.
3. American Diabetes Association. Standards of medical care in diabetes–2019. Diabetes Care. 2019;42(suppl 1):S1-S193.
4. National Institute for Health and Care Excellence. Type 2 diabetes in adults: management. www.nice.org.uk/guidance/ng28. Published December 2015. Updated May 2017. Accessed March 1, 2019.
5. Garber A, Abrahamson M, Barzilay J, et al. Consensus Statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the Comprehensive Type 2 Diabetes Management Algorithm—2018 Executive Summary. Endocr Pract. 2018;24:91-120.
6. Salanti G, Del Giovane C, Chaimani A, et al. Evaluating the quality of evidence from a network meta-analysis. PLoS ONE. 2014;9:1-14.
1. Zheng S, Roddick A, Aghar-Jaffar R, et al. Association between use of sodium-glucose cotransporter 2 inhibitors, glucagon-like peptide 1 agonists, and dipeptidyl peptidase 4 inhibitors with all-cause mortality in patients with type 2 diabetes: a systematic review and meta-analysis. JAMA. 2018;319:1580-1591.
2. Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2017. Atlanta, GA: Centers for Disease Control and Prevention, US Department of Health and Human Services; 2017.
3. American Diabetes Association. Standards of medical care in diabetes–2019. Diabetes Care. 2019;42(suppl 1):S1-S193.
4. National Institute for Health and Care Excellence. Type 2 diabetes in adults: management. www.nice.org.uk/guidance/ng28. Published December 2015. Updated May 2017. Accessed March 1, 2019.
5. Garber A, Abrahamson M, Barzilay J, et al. Consensus Statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the Comprehensive Type 2 Diabetes Management Algorithm—2018 Executive Summary. Endocr Pract. 2018;24:91-120.
6. Salanti G, Del Giovane C, Chaimani A, et al. Evaluating the quality of evidence from a network meta-analysis. PLoS ONE. 2014;9:1-14.
PRACTICE CHANGER
Consider adding a sodium-glucose cotransporter 2 (SGLT-2) inhibitor or a glucagon-like peptide 1 (GLP-1) agonist to the treatment regimen of patients with poorly controlled type 2 diabetes—especially those with higher CV risk. Doing so can reduce all-cause and cardiovascular (CV) mortality 1
STRENGTH OF RECOMMENDATION
B: Based on a network meta-analysis of 236 randomized controlled trials.
Zheng S, Roddick A, Aghar-Jaffar R, et al. Association between use of sodium-glucose cotransporter 2 inhibitors, glucagon-like peptide 1 agonists, and dipeptidyl peptidase 4 inhibitors with all-cause mortality in patients with type 2 diabetes: a systematic review and meta-analysis. JAMA. 2018;319:1580-1591.
More sleep can help youth manage type 1 diabetes
, according to a study of sleep duration and quality in young diabetes patients.
“This study adds to the growing body of literature that supports the cascading effects of sleep on multiple aspects of diabetes-related outcomes,” wrote lead author Sara S. Frye, PhD, of the University of Arizona, Tucson, and her coauthors, adding that the results “highlight the importance of assessing sleep in this population that appears to be at high risk for insufficient sleep duration.” The study was published in Sleep Medicine.
Dr. Frye and her colleagues recruited 111 children between the ages of 10 and 16 with type 1 diabetes mellitus to participate in their Glucose Regulation and Neurobehavioral Effects of Sleep (GRANES) study. The participants wore wrist actigraphs for an average of 5.5 nights to objectively measure sleep, including duration, quality, timing, and consistency. They completed self-reported sleep diaries each morning of the study. Glycemic control and diabetes management were assessed via hemoglobin A1c (HbA1c) levels and self-monitoring of blood glucose (SMBG) frequency, which were obtained via medical records. The participants and their parents also completed the Diabetes Management Scale.
Based on actigraphy data, the average total sleep time was 7.45 hours (standard deviation, 0.74), below the recommended duration of 9 hours for youths in this age group. All but one participant was recorded as sleeping less than the recommended amount. Average HbA1c of 9.11% (SD, 1.95) indicated poor diabetic control, and the average SMBG frequency was 4.90 (SD, 2.71) with a range of 1-14 checks per day. Per mediation analysis, for every additional hour of sleep, HbA1c was reduced by 0.33% and SMBG frequency went up by 0.88. In addition, SMBG frequency was related to HbA1c, supporting previous findings that “self-management behaviors play a critical role in maintaining diabetes control.”
The coauthors acknowledged the limitations of their study, including actigraphy data being logged over a 1-week period instead of the recommended 2 weeks. They also relied on medical records to determine HbA1c and SMBG rather than collecting that information along with the actigraphy data. However, they did note that HbA1c measures glucose levels over a 3-month period, which would have covered their participation in the study.
The study was supported by American Diabetes Association and cosponsored by the Order of the Amaranth Diabetes Foundation. The authors reported no conflicts of interest.
SOURCE: Frye SS et al. Sleep Med. 2019 Feb 16. doi: 10.1016/j.sleep.2019.01.043.
, according to a study of sleep duration and quality in young diabetes patients.
“This study adds to the growing body of literature that supports the cascading effects of sleep on multiple aspects of diabetes-related outcomes,” wrote lead author Sara S. Frye, PhD, of the University of Arizona, Tucson, and her coauthors, adding that the results “highlight the importance of assessing sleep in this population that appears to be at high risk for insufficient sleep duration.” The study was published in Sleep Medicine.
Dr. Frye and her colleagues recruited 111 children between the ages of 10 and 16 with type 1 diabetes mellitus to participate in their Glucose Regulation and Neurobehavioral Effects of Sleep (GRANES) study. The participants wore wrist actigraphs for an average of 5.5 nights to objectively measure sleep, including duration, quality, timing, and consistency. They completed self-reported sleep diaries each morning of the study. Glycemic control and diabetes management were assessed via hemoglobin A1c (HbA1c) levels and self-monitoring of blood glucose (SMBG) frequency, which were obtained via medical records. The participants and their parents also completed the Diabetes Management Scale.
Based on actigraphy data, the average total sleep time was 7.45 hours (standard deviation, 0.74), below the recommended duration of 9 hours for youths in this age group. All but one participant was recorded as sleeping less than the recommended amount. Average HbA1c of 9.11% (SD, 1.95) indicated poor diabetic control, and the average SMBG frequency was 4.90 (SD, 2.71) with a range of 1-14 checks per day. Per mediation analysis, for every additional hour of sleep, HbA1c was reduced by 0.33% and SMBG frequency went up by 0.88. In addition, SMBG frequency was related to HbA1c, supporting previous findings that “self-management behaviors play a critical role in maintaining diabetes control.”
The coauthors acknowledged the limitations of their study, including actigraphy data being logged over a 1-week period instead of the recommended 2 weeks. They also relied on medical records to determine HbA1c and SMBG rather than collecting that information along with the actigraphy data. However, they did note that HbA1c measures glucose levels over a 3-month period, which would have covered their participation in the study.
The study was supported by American Diabetes Association and cosponsored by the Order of the Amaranth Diabetes Foundation. The authors reported no conflicts of interest.
SOURCE: Frye SS et al. Sleep Med. 2019 Feb 16. doi: 10.1016/j.sleep.2019.01.043.
, according to a study of sleep duration and quality in young diabetes patients.
“This study adds to the growing body of literature that supports the cascading effects of sleep on multiple aspects of diabetes-related outcomes,” wrote lead author Sara S. Frye, PhD, of the University of Arizona, Tucson, and her coauthors, adding that the results “highlight the importance of assessing sleep in this population that appears to be at high risk for insufficient sleep duration.” The study was published in Sleep Medicine.
Dr. Frye and her colleagues recruited 111 children between the ages of 10 and 16 with type 1 diabetes mellitus to participate in their Glucose Regulation and Neurobehavioral Effects of Sleep (GRANES) study. The participants wore wrist actigraphs for an average of 5.5 nights to objectively measure sleep, including duration, quality, timing, and consistency. They completed self-reported sleep diaries each morning of the study. Glycemic control and diabetes management were assessed via hemoglobin A1c (HbA1c) levels and self-monitoring of blood glucose (SMBG) frequency, which were obtained via medical records. The participants and their parents also completed the Diabetes Management Scale.
Based on actigraphy data, the average total sleep time was 7.45 hours (standard deviation, 0.74), below the recommended duration of 9 hours for youths in this age group. All but one participant was recorded as sleeping less than the recommended amount. Average HbA1c of 9.11% (SD, 1.95) indicated poor diabetic control, and the average SMBG frequency was 4.90 (SD, 2.71) with a range of 1-14 checks per day. Per mediation analysis, for every additional hour of sleep, HbA1c was reduced by 0.33% and SMBG frequency went up by 0.88. In addition, SMBG frequency was related to HbA1c, supporting previous findings that “self-management behaviors play a critical role in maintaining diabetes control.”
The coauthors acknowledged the limitations of their study, including actigraphy data being logged over a 1-week period instead of the recommended 2 weeks. They also relied on medical records to determine HbA1c and SMBG rather than collecting that information along with the actigraphy data. However, they did note that HbA1c measures glucose levels over a 3-month period, which would have covered their participation in the study.
The study was supported by American Diabetes Association and cosponsored by the Order of the Amaranth Diabetes Foundation. The authors reported no conflicts of interest.
SOURCE: Frye SS et al. Sleep Med. 2019 Feb 16. doi: 10.1016/j.sleep.2019.01.043.
FROM SLEEP MEDICINE
HCV treatment with DAA regimens linked to reduced diabetes risk
SEATTLE – Treatment of hepatitis C virus (HCV) with new direct-acting antiviral (DAA) regimens is associated with improved glucose control and reduced incidence of type 2 diabetes when compared to treatment with pegylated interferon/ribavirin (PEG/RBV ) and untreated controls, according to a new analysis of the Electronically Retrieved Cohort of HCV Infected Veterans.
“Previously, people who had diabetes were considered slightly more difficult to treat because their virologic response was a little lower, but now this is not the case, and we have the added benefit of reducing the incidence of diabetes,” said Adeel Butt, MD, professor of medicine and health care policy and research at Weill Cornell Medicine, New York and Qatar, in an interview. Dr. Butt presented the study at the Conference on Retroviruses & Opportunistic Infections.
The incidence of diabetes dropped in the overall treated cohort, compared with untreated patients, but this benefit was driven by the effect of DAAs, as there was no significant difference between PEG/RBV–treated patients and controls. “It’s another reason to argue with people who make it difficult to treat. Our biggest barriers to treating everyone with hepatitis C has to do with reimbursement and the capacity of the health care system, and this is another reason that we need to overcome those barriers. It’s an important insight that provides one more reason to try to continue to eradicate hepatitis C in our population,” said Robert Schooley, MD, professor of medicine at the University of California, San Diego, in an interview.
Patients may also need some reassurance, given concerns that have arisen over the potential for older regimens to cause diabetes. Dr. Butt cited an example of a patient who has an acute myocardial infarction, has a high body mass, and wants to know if DAAs will help or hurt them. “We see [such patients] frequently. This is pretty reassuring not only that DAAs don’t increase risk, but they actually decrease the risk of diabetes as opposed to older treatments. There is a growing body of evidence that non–liver [related conditions] significantly improve with treatment,” he said.
The results could also help prioritize patients for treatment. “It may be important to the patients who are at elevated risk of developing diabetes. They may need to be monitored more closely and offered treatment earlier, perhaps, but that requires more study,” said Dr. Butt.
The researchers excluded patients with HIV or hepatitis B virus, and those who had prevalent diabetes. The cohort included 26,043 treated patients and 26,043 propensity score–matched untreated control patients. Treated patients underwent at least 8 weeks of DAA or 24 weeks of PEG/RBV. Demographically, 54% of patients were white, 29% were black, 3% were Hispanic, and 96% of the patients were male. About one-third had a body mass index of 30 or above.
The incidence of diabetes was 20.6 per 1,000 person-years of follow-up among untreated patients, compared with 15.5 among treated patients (P less than .0001). The incidence was 19.8 in patients treated with PEG/RBV (P =.39) and 9.9 in those treated with DAAs (P less than. 001; hazard ratio, 0.48; P less than .0001). The incidence of diabetes in those with a sustained viral response (SVR) was 13.3 per 1,000 person-years, compared with 19.2 in patients with no SVR (P less than .0001). The incidence of diabetes was lower in treated patients regardless of baseline FIB-4 (Fibrosis-4, a liver fibrosis score) levels.
The study was funded by Gilead. Dr. Butt has had research grants from Gilead and Dr. Schooley is on Gilead’s scientific advisory board.
SOURCE: A Butt et al. CROI 2019. Abstract 88.
SEATTLE – Treatment of hepatitis C virus (HCV) with new direct-acting antiviral (DAA) regimens is associated with improved glucose control and reduced incidence of type 2 diabetes when compared to treatment with pegylated interferon/ribavirin (PEG/RBV ) and untreated controls, according to a new analysis of the Electronically Retrieved Cohort of HCV Infected Veterans.
“Previously, people who had diabetes were considered slightly more difficult to treat because their virologic response was a little lower, but now this is not the case, and we have the added benefit of reducing the incidence of diabetes,” said Adeel Butt, MD, professor of medicine and health care policy and research at Weill Cornell Medicine, New York and Qatar, in an interview. Dr. Butt presented the study at the Conference on Retroviruses & Opportunistic Infections.
The incidence of diabetes dropped in the overall treated cohort, compared with untreated patients, but this benefit was driven by the effect of DAAs, as there was no significant difference between PEG/RBV–treated patients and controls. “It’s another reason to argue with people who make it difficult to treat. Our biggest barriers to treating everyone with hepatitis C has to do with reimbursement and the capacity of the health care system, and this is another reason that we need to overcome those barriers. It’s an important insight that provides one more reason to try to continue to eradicate hepatitis C in our population,” said Robert Schooley, MD, professor of medicine at the University of California, San Diego, in an interview.
Patients may also need some reassurance, given concerns that have arisen over the potential for older regimens to cause diabetes. Dr. Butt cited an example of a patient who has an acute myocardial infarction, has a high body mass, and wants to know if DAAs will help or hurt them. “We see [such patients] frequently. This is pretty reassuring not only that DAAs don’t increase risk, but they actually decrease the risk of diabetes as opposed to older treatments. There is a growing body of evidence that non–liver [related conditions] significantly improve with treatment,” he said.
The results could also help prioritize patients for treatment. “It may be important to the patients who are at elevated risk of developing diabetes. They may need to be monitored more closely and offered treatment earlier, perhaps, but that requires more study,” said Dr. Butt.
The researchers excluded patients with HIV or hepatitis B virus, and those who had prevalent diabetes. The cohort included 26,043 treated patients and 26,043 propensity score–matched untreated control patients. Treated patients underwent at least 8 weeks of DAA or 24 weeks of PEG/RBV. Demographically, 54% of patients were white, 29% were black, 3% were Hispanic, and 96% of the patients were male. About one-third had a body mass index of 30 or above.
The incidence of diabetes was 20.6 per 1,000 person-years of follow-up among untreated patients, compared with 15.5 among treated patients (P less than .0001). The incidence was 19.8 in patients treated with PEG/RBV (P =.39) and 9.9 in those treated with DAAs (P less than. 001; hazard ratio, 0.48; P less than .0001). The incidence of diabetes in those with a sustained viral response (SVR) was 13.3 per 1,000 person-years, compared with 19.2 in patients with no SVR (P less than .0001). The incidence of diabetes was lower in treated patients regardless of baseline FIB-4 (Fibrosis-4, a liver fibrosis score) levels.
The study was funded by Gilead. Dr. Butt has had research grants from Gilead and Dr. Schooley is on Gilead’s scientific advisory board.
SOURCE: A Butt et al. CROI 2019. Abstract 88.
SEATTLE – Treatment of hepatitis C virus (HCV) with new direct-acting antiviral (DAA) regimens is associated with improved glucose control and reduced incidence of type 2 diabetes when compared to treatment with pegylated interferon/ribavirin (PEG/RBV ) and untreated controls, according to a new analysis of the Electronically Retrieved Cohort of HCV Infected Veterans.
“Previously, people who had diabetes were considered slightly more difficult to treat because their virologic response was a little lower, but now this is not the case, and we have the added benefit of reducing the incidence of diabetes,” said Adeel Butt, MD, professor of medicine and health care policy and research at Weill Cornell Medicine, New York and Qatar, in an interview. Dr. Butt presented the study at the Conference on Retroviruses & Opportunistic Infections.
The incidence of diabetes dropped in the overall treated cohort, compared with untreated patients, but this benefit was driven by the effect of DAAs, as there was no significant difference between PEG/RBV–treated patients and controls. “It’s another reason to argue with people who make it difficult to treat. Our biggest barriers to treating everyone with hepatitis C has to do with reimbursement and the capacity of the health care system, and this is another reason that we need to overcome those barriers. It’s an important insight that provides one more reason to try to continue to eradicate hepatitis C in our population,” said Robert Schooley, MD, professor of medicine at the University of California, San Diego, in an interview.
Patients may also need some reassurance, given concerns that have arisen over the potential for older regimens to cause diabetes. Dr. Butt cited an example of a patient who has an acute myocardial infarction, has a high body mass, and wants to know if DAAs will help or hurt them. “We see [such patients] frequently. This is pretty reassuring not only that DAAs don’t increase risk, but they actually decrease the risk of diabetes as opposed to older treatments. There is a growing body of evidence that non–liver [related conditions] significantly improve with treatment,” he said.
The results could also help prioritize patients for treatment. “It may be important to the patients who are at elevated risk of developing diabetes. They may need to be monitored more closely and offered treatment earlier, perhaps, but that requires more study,” said Dr. Butt.
The researchers excluded patients with HIV or hepatitis B virus, and those who had prevalent diabetes. The cohort included 26,043 treated patients and 26,043 propensity score–matched untreated control patients. Treated patients underwent at least 8 weeks of DAA or 24 weeks of PEG/RBV. Demographically, 54% of patients were white, 29% were black, 3% were Hispanic, and 96% of the patients were male. About one-third had a body mass index of 30 or above.
The incidence of diabetes was 20.6 per 1,000 person-years of follow-up among untreated patients, compared with 15.5 among treated patients (P less than .0001). The incidence was 19.8 in patients treated with PEG/RBV (P =.39) and 9.9 in those treated with DAAs (P less than. 001; hazard ratio, 0.48; P less than .0001). The incidence of diabetes in those with a sustained viral response (SVR) was 13.3 per 1,000 person-years, compared with 19.2 in patients with no SVR (P less than .0001). The incidence of diabetes was lower in treated patients regardless of baseline FIB-4 (Fibrosis-4, a liver fibrosis score) levels.
The study was funded by Gilead. Dr. Butt has had research grants from Gilead and Dr. Schooley is on Gilead’s scientific advisory board.
SOURCE: A Butt et al. CROI 2019. Abstract 88.
REPORTING FROM CROI 2019
Umbilical cord allograft may boost diabetic foot ulcer healing
Dehydrated human umbilical cord allograft may have benefit over alginate wound dressings as a treatment for chronic, nonhealing diabetic foot ulcers (DFU), findings from an industry-funded, randomized controlled study suggest.
The findings “provide additional evidence of the safety and efficacy of dehydrated placental tissues,” wrote William Tettelbach, MD, and his colleagues. Their report is in International Wound Journal.
The burden of diabetic foot disease in the United States is immense. A 2014 study estimated that treatment of DFUs alone cost public and private insurers as much as $13 billion per year (Diabetes Care. 2014;37(3):651-8).
MiMedx, which funded the new study, has developed a product called EpiCord to protect the DFU wound site. The product’s website describes it as a “unique, thick membrane derived from umbilical cord” that’s “minimally manipulated, dehydrated, [and] non-viable” (www.mimedx.com/epicord). The study authors noted that “immunogenicity of placental tissue lends credence to its use as an allograft material for difficult-to-heal wounds.”
For the new study, which was conducted from 2016 to 2018 and led by Dr. Tettelbach, an infectious disease specialist who is now an employee of MiMedx, the researchers enlisted 155 adult patients with stubborn DFUs at 11 centers in the United States.
All the ulcers had 30% or less wound area reduction after 14 days of standard care. The majority of patients (81%) were male; 63% were obese, 43% were smokers, and 17% had a prior amputation.
The patients were randomly assigned to receive a weekly application of EpiCord (n = 101) or treatment with an alginate wound dressing (n = 54) in addition to standard care. The percentage of patients whose wounds healed completely by 12 weeks later was higher in the study group than in those who were treated with alginate dressings (70% vs. 48%, respectively; P = .0089), per an intent-to-treat analysis.
The researchers also focused purely on patients who had received adequate debridement (107/155 ulcers, 69%). Of those ulcers, 64/67 (96%), in the study group healed completely at 12 weeks, compared with 26/40 (65%) of the alginate group (P less than .0001.)
The researchers did not notice any adverse effects related to either dressing.
According to the study, the findings regarding EpiCord are comparable with a sister study of a similar product by the same company that was tested in diabetic lower-extremity ulcers. That study, of a product called EpiFix, was published in the same issue of the journal (Int Wound J. 2019 Feb;16[1]:19-29).
“A thicker and more durable allograft such as EpiCord may be a good choice for implantation into deeper wounds and in situations where suturing the allograft in place is desired,” the authors wrote of the EpiCord study.
MiMedx provided research funding to all of the authors.
SOURCE: Tettelbach W et al. Int Wound J. 2019;16(1):122-130. doi: 10.1111/iwj.12976.
Dehydrated human umbilical cord allograft may have benefit over alginate wound dressings as a treatment for chronic, nonhealing diabetic foot ulcers (DFU), findings from an industry-funded, randomized controlled study suggest.
The findings “provide additional evidence of the safety and efficacy of dehydrated placental tissues,” wrote William Tettelbach, MD, and his colleagues. Their report is in International Wound Journal.
The burden of diabetic foot disease in the United States is immense. A 2014 study estimated that treatment of DFUs alone cost public and private insurers as much as $13 billion per year (Diabetes Care. 2014;37(3):651-8).
MiMedx, which funded the new study, has developed a product called EpiCord to protect the DFU wound site. The product’s website describes it as a “unique, thick membrane derived from umbilical cord” that’s “minimally manipulated, dehydrated, [and] non-viable” (www.mimedx.com/epicord). The study authors noted that “immunogenicity of placental tissue lends credence to its use as an allograft material for difficult-to-heal wounds.”
For the new study, which was conducted from 2016 to 2018 and led by Dr. Tettelbach, an infectious disease specialist who is now an employee of MiMedx, the researchers enlisted 155 adult patients with stubborn DFUs at 11 centers in the United States.
All the ulcers had 30% or less wound area reduction after 14 days of standard care. The majority of patients (81%) were male; 63% were obese, 43% were smokers, and 17% had a prior amputation.
The patients were randomly assigned to receive a weekly application of EpiCord (n = 101) or treatment with an alginate wound dressing (n = 54) in addition to standard care. The percentage of patients whose wounds healed completely by 12 weeks later was higher in the study group than in those who were treated with alginate dressings (70% vs. 48%, respectively; P = .0089), per an intent-to-treat analysis.
The researchers also focused purely on patients who had received adequate debridement (107/155 ulcers, 69%). Of those ulcers, 64/67 (96%), in the study group healed completely at 12 weeks, compared with 26/40 (65%) of the alginate group (P less than .0001.)
The researchers did not notice any adverse effects related to either dressing.
According to the study, the findings regarding EpiCord are comparable with a sister study of a similar product by the same company that was tested in diabetic lower-extremity ulcers. That study, of a product called EpiFix, was published in the same issue of the journal (Int Wound J. 2019 Feb;16[1]:19-29).
“A thicker and more durable allograft such as EpiCord may be a good choice for implantation into deeper wounds and in situations where suturing the allograft in place is desired,” the authors wrote of the EpiCord study.
MiMedx provided research funding to all of the authors.
SOURCE: Tettelbach W et al. Int Wound J. 2019;16(1):122-130. doi: 10.1111/iwj.12976.
Dehydrated human umbilical cord allograft may have benefit over alginate wound dressings as a treatment for chronic, nonhealing diabetic foot ulcers (DFU), findings from an industry-funded, randomized controlled study suggest.
The findings “provide additional evidence of the safety and efficacy of dehydrated placental tissues,” wrote William Tettelbach, MD, and his colleagues. Their report is in International Wound Journal.
The burden of diabetic foot disease in the United States is immense. A 2014 study estimated that treatment of DFUs alone cost public and private insurers as much as $13 billion per year (Diabetes Care. 2014;37(3):651-8).
MiMedx, which funded the new study, has developed a product called EpiCord to protect the DFU wound site. The product’s website describes it as a “unique, thick membrane derived from umbilical cord” that’s “minimally manipulated, dehydrated, [and] non-viable” (www.mimedx.com/epicord). The study authors noted that “immunogenicity of placental tissue lends credence to its use as an allograft material for difficult-to-heal wounds.”
For the new study, which was conducted from 2016 to 2018 and led by Dr. Tettelbach, an infectious disease specialist who is now an employee of MiMedx, the researchers enlisted 155 adult patients with stubborn DFUs at 11 centers in the United States.
All the ulcers had 30% or less wound area reduction after 14 days of standard care. The majority of patients (81%) were male; 63% were obese, 43% were smokers, and 17% had a prior amputation.
The patients were randomly assigned to receive a weekly application of EpiCord (n = 101) or treatment with an alginate wound dressing (n = 54) in addition to standard care. The percentage of patients whose wounds healed completely by 12 weeks later was higher in the study group than in those who were treated with alginate dressings (70% vs. 48%, respectively; P = .0089), per an intent-to-treat analysis.
The researchers also focused purely on patients who had received adequate debridement (107/155 ulcers, 69%). Of those ulcers, 64/67 (96%), in the study group healed completely at 12 weeks, compared with 26/40 (65%) of the alginate group (P less than .0001.)
The researchers did not notice any adverse effects related to either dressing.
According to the study, the findings regarding EpiCord are comparable with a sister study of a similar product by the same company that was tested in diabetic lower-extremity ulcers. That study, of a product called EpiFix, was published in the same issue of the journal (Int Wound J. 2019 Feb;16[1]:19-29).
“A thicker and more durable allograft such as EpiCord may be a good choice for implantation into deeper wounds and in situations where suturing the allograft in place is desired,” the authors wrote of the EpiCord study.
MiMedx provided research funding to all of the authors.
SOURCE: Tettelbach W et al. Int Wound J. 2019;16(1):122-130. doi: 10.1111/iwj.12976.
FROM INTERNATIONAL WOUND JOURNAL
Behavioral intervention improves physical activity in patients with diabetes
A behavioral intervention that involves regular counseling sessions could help patients with type 2 diabetes increase their levels of physical activity and decrease their amount of sedentary time, according to findings from a prospective, randomized trial of 300 physically inactive patients with type 2 diabetes.
“The primary strength of this study is the application of an intervention targeting both physical activity and sedentary time across all settings (e.g., leisure, transportation, household, and occupation), based on theoretical grounds and using several behavior-change techniques,” wrote Stefano Balducci, MD, of Sapienza University in Rome and his colleagues. The findings were published in JAMA.
Half the participants were randomized to an intervention that involved one individual theoretical counseling session with a diabetologist and eight biweekly theoretical and practical counseling sessions with an exercise specialist each year for 3 years. The other half received standard care in the form of recommendations from their general physician about increasing physical activity and decreasing sedentary time. Both groups also received the same general treatment regimen according to guidelines.
The findings showed significant increases in volume of physical activity, light-intensity physical activity, and moderate to vigorous physical activity in the intervention group during the first 4 months of the trial. Those increases also were greater than the increases seen in the usual care group. Patients in the intervention group also showed greater decreases in sedentary time, compared with those in the control group during the same time.
After 4 months, the increases in physical activity in the intervention group plateaued but remained stable until 2 years. After that, the levels of activity declined but still remained significantly higher than at baseline. The level of sedentary time also increased after 2 years but was still lower than at baseline.
By the end of the study, the intervention group accumulated 13.8 metabolic equivalent hours/week of physical activity volume, compared with 10.5 hours in the control group; 18.9 minutes/day of moderate to vigorous intensity physical activity, compared with 12.5 minutes in the control group; and 4.6 hours/day of light-intensity physical activity, compared with 3.8 hours in the control group. In regard to sedentary time, the intervention group accumulated 10.9 hours/day, compared with 11.7 hours in the control group. All differences were statistically significant.
“The present findings support the need for interventions targeting all domains of behavior to obtain substantial lifestyle changes, not limited to moderate- to vigorous-intensity physical activity, which has little effect on sedentary time,” Dr. Balducci and his coauthors wrote. “This concept is consistent with a 2018 report showing that physical activity, sedentary time, and cardiorespiratory fitness are all important for cardiometabolic health.”
For the secondary outcomes of cardiorespiratory fitness and lower-body strength, the authors saw significant improvements in the intervention group, whereas the control group showed a worsening in those outcomes. The intervention group also showed significant improvements in fasting plasma glucose level, systolic blood pressure, total coronary heart disease 10-year risk score, and fatal coronary heart disease 10-year risk score. They also had significantly greater improvements than did the control group in total stroke risk score, hemoglobin A1c, fasting plasma glucose levels, and coronary heart disease risk.
In all, there were 41 adverse events in the intervention group, compared with 59 in the control group, outside of the sessions. During the sessions, participants in the intervention group experienced mild hypoglycemia (8 episodes), tachycardia/arrhythmia (3), and musculoskeletal injury or discomfort (19).
One of the limitations highlighted by the authors was that the benefits of their strategy could vary in other cohorts because of differences in climatic, socioeconomic, or cultural settings.
The study was supported by the Metabolic Fitness Association. Three authors declared grants and personal fees from pharmaceutical companies, and one author was an employee of Technogym. No other conflicts of interest were declared.
SOURCE: Balducci S et al. JAMA. 2019;321:880-90.
A behavioral intervention that involves regular counseling sessions could help patients with type 2 diabetes increase their levels of physical activity and decrease their amount of sedentary time, according to findings from a prospective, randomized trial of 300 physically inactive patients with type 2 diabetes.
“The primary strength of this study is the application of an intervention targeting both physical activity and sedentary time across all settings (e.g., leisure, transportation, household, and occupation), based on theoretical grounds and using several behavior-change techniques,” wrote Stefano Balducci, MD, of Sapienza University in Rome and his colleagues. The findings were published in JAMA.
Half the participants were randomized to an intervention that involved one individual theoretical counseling session with a diabetologist and eight biweekly theoretical and practical counseling sessions with an exercise specialist each year for 3 years. The other half received standard care in the form of recommendations from their general physician about increasing physical activity and decreasing sedentary time. Both groups also received the same general treatment regimen according to guidelines.
The findings showed significant increases in volume of physical activity, light-intensity physical activity, and moderate to vigorous physical activity in the intervention group during the first 4 months of the trial. Those increases also were greater than the increases seen in the usual care group. Patients in the intervention group also showed greater decreases in sedentary time, compared with those in the control group during the same time.
After 4 months, the increases in physical activity in the intervention group plateaued but remained stable until 2 years. After that, the levels of activity declined but still remained significantly higher than at baseline. The level of sedentary time also increased after 2 years but was still lower than at baseline.
By the end of the study, the intervention group accumulated 13.8 metabolic equivalent hours/week of physical activity volume, compared with 10.5 hours in the control group; 18.9 minutes/day of moderate to vigorous intensity physical activity, compared with 12.5 minutes in the control group; and 4.6 hours/day of light-intensity physical activity, compared with 3.8 hours in the control group. In regard to sedentary time, the intervention group accumulated 10.9 hours/day, compared with 11.7 hours in the control group. All differences were statistically significant.
“The present findings support the need for interventions targeting all domains of behavior to obtain substantial lifestyle changes, not limited to moderate- to vigorous-intensity physical activity, which has little effect on sedentary time,” Dr. Balducci and his coauthors wrote. “This concept is consistent with a 2018 report showing that physical activity, sedentary time, and cardiorespiratory fitness are all important for cardiometabolic health.”
For the secondary outcomes of cardiorespiratory fitness and lower-body strength, the authors saw significant improvements in the intervention group, whereas the control group showed a worsening in those outcomes. The intervention group also showed significant improvements in fasting plasma glucose level, systolic blood pressure, total coronary heart disease 10-year risk score, and fatal coronary heart disease 10-year risk score. They also had significantly greater improvements than did the control group in total stroke risk score, hemoglobin A1c, fasting plasma glucose levels, and coronary heart disease risk.
In all, there were 41 adverse events in the intervention group, compared with 59 in the control group, outside of the sessions. During the sessions, participants in the intervention group experienced mild hypoglycemia (8 episodes), tachycardia/arrhythmia (3), and musculoskeletal injury or discomfort (19).
One of the limitations highlighted by the authors was that the benefits of their strategy could vary in other cohorts because of differences in climatic, socioeconomic, or cultural settings.
The study was supported by the Metabolic Fitness Association. Three authors declared grants and personal fees from pharmaceutical companies, and one author was an employee of Technogym. No other conflicts of interest were declared.
SOURCE: Balducci S et al. JAMA. 2019;321:880-90.
A behavioral intervention that involves regular counseling sessions could help patients with type 2 diabetes increase their levels of physical activity and decrease their amount of sedentary time, according to findings from a prospective, randomized trial of 300 physically inactive patients with type 2 diabetes.
“The primary strength of this study is the application of an intervention targeting both physical activity and sedentary time across all settings (e.g., leisure, transportation, household, and occupation), based on theoretical grounds and using several behavior-change techniques,” wrote Stefano Balducci, MD, of Sapienza University in Rome and his colleagues. The findings were published in JAMA.
Half the participants were randomized to an intervention that involved one individual theoretical counseling session with a diabetologist and eight biweekly theoretical and practical counseling sessions with an exercise specialist each year for 3 years. The other half received standard care in the form of recommendations from their general physician about increasing physical activity and decreasing sedentary time. Both groups also received the same general treatment regimen according to guidelines.
The findings showed significant increases in volume of physical activity, light-intensity physical activity, and moderate to vigorous physical activity in the intervention group during the first 4 months of the trial. Those increases also were greater than the increases seen in the usual care group. Patients in the intervention group also showed greater decreases in sedentary time, compared with those in the control group during the same time.
After 4 months, the increases in physical activity in the intervention group plateaued but remained stable until 2 years. After that, the levels of activity declined but still remained significantly higher than at baseline. The level of sedentary time also increased after 2 years but was still lower than at baseline.
By the end of the study, the intervention group accumulated 13.8 metabolic equivalent hours/week of physical activity volume, compared with 10.5 hours in the control group; 18.9 minutes/day of moderate to vigorous intensity physical activity, compared with 12.5 minutes in the control group; and 4.6 hours/day of light-intensity physical activity, compared with 3.8 hours in the control group. In regard to sedentary time, the intervention group accumulated 10.9 hours/day, compared with 11.7 hours in the control group. All differences were statistically significant.
“The present findings support the need for interventions targeting all domains of behavior to obtain substantial lifestyle changes, not limited to moderate- to vigorous-intensity physical activity, which has little effect on sedentary time,” Dr. Balducci and his coauthors wrote. “This concept is consistent with a 2018 report showing that physical activity, sedentary time, and cardiorespiratory fitness are all important for cardiometabolic health.”
For the secondary outcomes of cardiorespiratory fitness and lower-body strength, the authors saw significant improvements in the intervention group, whereas the control group showed a worsening in those outcomes. The intervention group also showed significant improvements in fasting plasma glucose level, systolic blood pressure, total coronary heart disease 10-year risk score, and fatal coronary heart disease 10-year risk score. They also had significantly greater improvements than did the control group in total stroke risk score, hemoglobin A1c, fasting plasma glucose levels, and coronary heart disease risk.
In all, there were 41 adverse events in the intervention group, compared with 59 in the control group, outside of the sessions. During the sessions, participants in the intervention group experienced mild hypoglycemia (8 episodes), tachycardia/arrhythmia (3), and musculoskeletal injury or discomfort (19).
One of the limitations highlighted by the authors was that the benefits of their strategy could vary in other cohorts because of differences in climatic, socioeconomic, or cultural settings.
The study was supported by the Metabolic Fitness Association. Three authors declared grants and personal fees from pharmaceutical companies, and one author was an employee of Technogym. No other conflicts of interest were declared.
SOURCE: Balducci S et al. JAMA. 2019;321:880-90.
FROM JAMA
Dermatologists name isobornyl acrylate contact allergen of the year
WASHINGTON – The American Contact Dermatitis Society has selected isobornyl acrylate the contact allergen of the year. It is an acrylic monomer used as an adhesive.

Among other applications, isobornyl acrylate is often used in medical devices. The selection was made based in part on multiple case reports of diabetes patients developing contact allergies to their diabetes devices, such as insulin pumps, explained Golara Honari, MD, of Stanford (Calif.) University, who presented the selection at the ACDS annual meeting.
The significance of this allergen is that testing through routine panels does not identify it, so clinician awareness is especially important, Dr. Honari noted in a video interview at the meeting.
Most of the reported contact allergen cases have been in patients with diabetes, but clinicians should think about other possible sources, such as acrylic nails, she said. As for treatment, clinicians and patients can consider alternative diabetes devices without isobornyl acrylate, she said.
In the future, close collaboration between clinicians and the medical device industry to develop appropriate labeling can help increase awareness of the potential for allergic reactions, she added.
Dr. Honari had no relevant financial conflicts to disclose.
WASHINGTON – The American Contact Dermatitis Society has selected isobornyl acrylate the contact allergen of the year. It is an acrylic monomer used as an adhesive.

Among other applications, isobornyl acrylate is often used in medical devices. The selection was made based in part on multiple case reports of diabetes patients developing contact allergies to their diabetes devices, such as insulin pumps, explained Golara Honari, MD, of Stanford (Calif.) University, who presented the selection at the ACDS annual meeting.
The significance of this allergen is that testing through routine panels does not identify it, so clinician awareness is especially important, Dr. Honari noted in a video interview at the meeting.
Most of the reported contact allergen cases have been in patients with diabetes, but clinicians should think about other possible sources, such as acrylic nails, she said. As for treatment, clinicians and patients can consider alternative diabetes devices without isobornyl acrylate, she said.
In the future, close collaboration between clinicians and the medical device industry to develop appropriate labeling can help increase awareness of the potential for allergic reactions, she added.
Dr. Honari had no relevant financial conflicts to disclose.
WASHINGTON – The American Contact Dermatitis Society has selected isobornyl acrylate the contact allergen of the year. It is an acrylic monomer used as an adhesive.

Among other applications, isobornyl acrylate is often used in medical devices. The selection was made based in part on multiple case reports of diabetes patients developing contact allergies to their diabetes devices, such as insulin pumps, explained Golara Honari, MD, of Stanford (Calif.) University, who presented the selection at the ACDS annual meeting.
The significance of this allergen is that testing through routine panels does not identify it, so clinician awareness is especially important, Dr. Honari noted in a video interview at the meeting.
Most of the reported contact allergen cases have been in patients with diabetes, but clinicians should think about other possible sources, such as acrylic nails, she said. As for treatment, clinicians and patients can consider alternative diabetes devices without isobornyl acrylate, she said.
In the future, close collaboration between clinicians and the medical device industry to develop appropriate labeling can help increase awareness of the potential for allergic reactions, she added.
Dr. Honari had no relevant financial conflicts to disclose.
AT ACDS 2019
Click for Credit: Endometriosis surgery benefits; diabetes & aging; more
Here are 5 articles from the March issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Endometriosis surgery: Women can expect years-long benefits
To take the posttest, go to: https://bit.ly/2Ez8mdu
Expires January 3, 2019
2. Cerebral small vessel disease progression linked to MCI in hypertensive patients
To take the posttest, go to: https://bit.ly/2ExDV7o
Expires January 4, 2019
3. Adult atopic dermatitis is fraught with dermatologic comorbidities
To take the posttest, go to: https://bit.ly/2Vl7E9a
Expires January 11, 2019
4. Antidepressants tied to greater hip fracture incidence in older adults
To take the posttest, go to: https://bit.ly/2GRfMeH
Expires January 4, 2019
5. Researchers exploring ways to mitigate aging’s impact on diabetes
To take the posttest, go to: https://bit.ly/2tFxF7v
Expires January 8, 2019
Here are 5 articles from the March issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Endometriosis surgery: Women can expect years-long benefits
To take the posttest, go to: https://bit.ly/2Ez8mdu
Expires January 3, 2019
2. Cerebral small vessel disease progression linked to MCI in hypertensive patients
To take the posttest, go to: https://bit.ly/2ExDV7o
Expires January 4, 2019
3. Adult atopic dermatitis is fraught with dermatologic comorbidities
To take the posttest, go to: https://bit.ly/2Vl7E9a
Expires January 11, 2019
4. Antidepressants tied to greater hip fracture incidence in older adults
To take the posttest, go to: https://bit.ly/2GRfMeH
Expires January 4, 2019
5. Researchers exploring ways to mitigate aging’s impact on diabetes
To take the posttest, go to: https://bit.ly/2tFxF7v
Expires January 8, 2019
Here are 5 articles from the March issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Endometriosis surgery: Women can expect years-long benefits
To take the posttest, go to: https://bit.ly/2Ez8mdu
Expires January 3, 2019
2. Cerebral small vessel disease progression linked to MCI in hypertensive patients
To take the posttest, go to: https://bit.ly/2ExDV7o
Expires January 4, 2019
3. Adult atopic dermatitis is fraught with dermatologic comorbidities
To take the posttest, go to: https://bit.ly/2Vl7E9a
Expires January 11, 2019
4. Antidepressants tied to greater hip fracture incidence in older adults
To take the posttest, go to: https://bit.ly/2GRfMeH
Expires January 4, 2019
5. Researchers exploring ways to mitigate aging’s impact on diabetes
To take the posttest, go to: https://bit.ly/2tFxF7v
Expires January 8, 2019