User login
Tricyclics may raise fracture risk in type 2 diabetes
VANCOUVER – , independent of any prevalent neuropathy, according to findings from an analysis of a large, randomized clinical trial.
Although the findings are suggestive, they don’t definitively pin blame on TCAs, said Rachel Elam, MD, who presented the study at the annual meeting of the American Society for Bone and Mineral Research. “I think that there’s not enough information to conclude that tricyclic antidepressants directly lead to fractures, but I think it opens the door [to] something we should look into more. Is it being mediated by a better predictor, or is it the medication itself? I think it’s more hypothesis generating,” said Dr. Elam, an assistant professor of medicine in the division of rheumatology at the Medical College of Georgia, Augusta.
Patients with type 2 diabetes are known to be at increased risk of fracture, but prediction tools tend to underestimate this risk, Dr. Elam said. “Type 2 diabetes–specific clinical risk factors may be helpful for finding out fracture risk in this population,” Dr. Elam said during her talk.
Glycemic control is one candidate risk factor because advanced glycation end products are linked to reduced bone strength. Other factors include antidiabetic medication use, neuropathy, and microvascular disease, which has been linked to increased cortical porosity.
The study examined a somewhat younger population than previous surveys, having drawn from the Look AHEAD-C clinical trial, which examined the effects of an intensive lifestyle intervention on type 2 diabetes. Look AHEAD-C included 4,697 participants aged 45-75 from 16 U.S. clinical sites. Participants had a body mass index of 25.0 kg/m2 or higher and hemoglobin A1c levels of 11% or below.
Dr. Elam cited the database’s inclusion of factors like A1c levels, renal parameters, and diabetic neuropathy. “It gave us a really good population to look at those risk factors” in a large group of people with type 2 diabetes, she said.
Over a median follow-up of 16.6 years, there were 649 participants with incident first clinical fracture(s). Statistically significant factors predicting fracture risk included TCA use (hazard ratio, 2.24; 95% confidence interval, 1.14-4.43), female gender (HR, 2.20; 95% CI, 1.83-2.66), insulin use (HR, 1.26; 95% CI, 1.02-1.57), increases in A1c level (per 1% increase: HR, 1.12; 95% CI, 1.04-1.20), age (HR, 1.02; 95% CI, 1.01-1.04), other or mixed race/ethnicity (HR, 0.68; 95% CI, 0.52-0.87), Hispanic White race/ethnicity (HR, 0.60; 95% CI, 0.39-0.91), non-Hispanic Black race/ethnicity (HR, 0.35; 95% CI, 0.26-0.47), and estrogen use (HR, 0.65; 95% CI, 0.44-0.98).
During the Q&A session following the presentation, Elsa Strotmeyer, PhD, commented that TCAs have been linked to central nervous system pathways in falls in other populations. “It’s a very nice study. It’s important to look at the diabetes complications related to the fracture risk, but I thought that they should have emphasized some more of the diabetes complications being related to fracture rather than these tricyclic antidepressants, because that is not a unique factor to that population,” said Dr. Strotmeyer, who is an associate professor of epidemiology at the University of Pittsburgh.
Instead, she noted a different strength of the study. “The study population is important because they’re a relatively young population with type 2 diabetes, compared to many studies [that] have been published in older populations. Showing similar things that we found in older populations was the unique piece and the important piece of this study,” Dr. Strotmeyer said.
Ultimately, the model wasn’t sufficient to be used as a fall risk predictor, but it should inform future work, according to Dr. Elam. “I think it does lay some new groundwork that when we’re looking forward, it may [help in building] other models to better predict fracture risk in type 2 diabetes. Things that would be important to include [in future models] would be medication use, such as tricyclic antidepressants,” and to make sure we include glycemic control, A1c, and insulin medication.
The study was independently funded. Dr. Elam and Dr. Strotmeyer report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VANCOUVER – , independent of any prevalent neuropathy, according to findings from an analysis of a large, randomized clinical trial.
Although the findings are suggestive, they don’t definitively pin blame on TCAs, said Rachel Elam, MD, who presented the study at the annual meeting of the American Society for Bone and Mineral Research. “I think that there’s not enough information to conclude that tricyclic antidepressants directly lead to fractures, but I think it opens the door [to] something we should look into more. Is it being mediated by a better predictor, or is it the medication itself? I think it’s more hypothesis generating,” said Dr. Elam, an assistant professor of medicine in the division of rheumatology at the Medical College of Georgia, Augusta.
Patients with type 2 diabetes are known to be at increased risk of fracture, but prediction tools tend to underestimate this risk, Dr. Elam said. “Type 2 diabetes–specific clinical risk factors may be helpful for finding out fracture risk in this population,” Dr. Elam said during her talk.
Glycemic control is one candidate risk factor because advanced glycation end products are linked to reduced bone strength. Other factors include antidiabetic medication use, neuropathy, and microvascular disease, which has been linked to increased cortical porosity.
The study examined a somewhat younger population than previous surveys, having drawn from the Look AHEAD-C clinical trial, which examined the effects of an intensive lifestyle intervention on type 2 diabetes. Look AHEAD-C included 4,697 participants aged 45-75 from 16 U.S. clinical sites. Participants had a body mass index of 25.0 kg/m2 or higher and hemoglobin A1c levels of 11% or below.
Dr. Elam cited the database’s inclusion of factors like A1c levels, renal parameters, and diabetic neuropathy. “It gave us a really good population to look at those risk factors” in a large group of people with type 2 diabetes, she said.
Over a median follow-up of 16.6 years, there were 649 participants with incident first clinical fracture(s). Statistically significant factors predicting fracture risk included TCA use (hazard ratio, 2.24; 95% confidence interval, 1.14-4.43), female gender (HR, 2.20; 95% CI, 1.83-2.66), insulin use (HR, 1.26; 95% CI, 1.02-1.57), increases in A1c level (per 1% increase: HR, 1.12; 95% CI, 1.04-1.20), age (HR, 1.02; 95% CI, 1.01-1.04), other or mixed race/ethnicity (HR, 0.68; 95% CI, 0.52-0.87), Hispanic White race/ethnicity (HR, 0.60; 95% CI, 0.39-0.91), non-Hispanic Black race/ethnicity (HR, 0.35; 95% CI, 0.26-0.47), and estrogen use (HR, 0.65; 95% CI, 0.44-0.98).
During the Q&A session following the presentation, Elsa Strotmeyer, PhD, commented that TCAs have been linked to central nervous system pathways in falls in other populations. “It’s a very nice study. It’s important to look at the diabetes complications related to the fracture risk, but I thought that they should have emphasized some more of the diabetes complications being related to fracture rather than these tricyclic antidepressants, because that is not a unique factor to that population,” said Dr. Strotmeyer, who is an associate professor of epidemiology at the University of Pittsburgh.
Instead, she noted a different strength of the study. “The study population is important because they’re a relatively young population with type 2 diabetes, compared to many studies [that] have been published in older populations. Showing similar things that we found in older populations was the unique piece and the important piece of this study,” Dr. Strotmeyer said.
Ultimately, the model wasn’t sufficient to be used as a fall risk predictor, but it should inform future work, according to Dr. Elam. “I think it does lay some new groundwork that when we’re looking forward, it may [help in building] other models to better predict fracture risk in type 2 diabetes. Things that would be important to include [in future models] would be medication use, such as tricyclic antidepressants,” and to make sure we include glycemic control, A1c, and insulin medication.
The study was independently funded. Dr. Elam and Dr. Strotmeyer report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VANCOUVER – , independent of any prevalent neuropathy, according to findings from an analysis of a large, randomized clinical trial.
Although the findings are suggestive, they don’t definitively pin blame on TCAs, said Rachel Elam, MD, who presented the study at the annual meeting of the American Society for Bone and Mineral Research. “I think that there’s not enough information to conclude that tricyclic antidepressants directly lead to fractures, but I think it opens the door [to] something we should look into more. Is it being mediated by a better predictor, or is it the medication itself? I think it’s more hypothesis generating,” said Dr. Elam, an assistant professor of medicine in the division of rheumatology at the Medical College of Georgia, Augusta.
Patients with type 2 diabetes are known to be at increased risk of fracture, but prediction tools tend to underestimate this risk, Dr. Elam said. “Type 2 diabetes–specific clinical risk factors may be helpful for finding out fracture risk in this population,” Dr. Elam said during her talk.
Glycemic control is one candidate risk factor because advanced glycation end products are linked to reduced bone strength. Other factors include antidiabetic medication use, neuropathy, and microvascular disease, which has been linked to increased cortical porosity.
The study examined a somewhat younger population than previous surveys, having drawn from the Look AHEAD-C clinical trial, which examined the effects of an intensive lifestyle intervention on type 2 diabetes. Look AHEAD-C included 4,697 participants aged 45-75 from 16 U.S. clinical sites. Participants had a body mass index of 25.0 kg/m2 or higher and hemoglobin A1c levels of 11% or below.
Dr. Elam cited the database’s inclusion of factors like A1c levels, renal parameters, and diabetic neuropathy. “It gave us a really good population to look at those risk factors” in a large group of people with type 2 diabetes, she said.
Over a median follow-up of 16.6 years, there were 649 participants with incident first clinical fracture(s). Statistically significant factors predicting fracture risk included TCA use (hazard ratio, 2.24; 95% confidence interval, 1.14-4.43), female gender (HR, 2.20; 95% CI, 1.83-2.66), insulin use (HR, 1.26; 95% CI, 1.02-1.57), increases in A1c level (per 1% increase: HR, 1.12; 95% CI, 1.04-1.20), age (HR, 1.02; 95% CI, 1.01-1.04), other or mixed race/ethnicity (HR, 0.68; 95% CI, 0.52-0.87), Hispanic White race/ethnicity (HR, 0.60; 95% CI, 0.39-0.91), non-Hispanic Black race/ethnicity (HR, 0.35; 95% CI, 0.26-0.47), and estrogen use (HR, 0.65; 95% CI, 0.44-0.98).
During the Q&A session following the presentation, Elsa Strotmeyer, PhD, commented that TCAs have been linked to central nervous system pathways in falls in other populations. “It’s a very nice study. It’s important to look at the diabetes complications related to the fracture risk, but I thought that they should have emphasized some more of the diabetes complications being related to fracture rather than these tricyclic antidepressants, because that is not a unique factor to that population,” said Dr. Strotmeyer, who is an associate professor of epidemiology at the University of Pittsburgh.
Instead, she noted a different strength of the study. “The study population is important because they’re a relatively young population with type 2 diabetes, compared to many studies [that] have been published in older populations. Showing similar things that we found in older populations was the unique piece and the important piece of this study,” Dr. Strotmeyer said.
Ultimately, the model wasn’t sufficient to be used as a fall risk predictor, but it should inform future work, according to Dr. Elam. “I think it does lay some new groundwork that when we’re looking forward, it may [help in building] other models to better predict fracture risk in type 2 diabetes. Things that would be important to include [in future models] would be medication use, such as tricyclic antidepressants,” and to make sure we include glycemic control, A1c, and insulin medication.
The study was independently funded. Dr. Elam and Dr. Strotmeyer report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ASBMR 2023
Treatment order evidence comes to light for premenopausal idiopathic osteoporosis: What to do after denosumab
VANCOUVER – With treatment with a bisphosphonate following sequential use of teriparatide (Forteo) and denosumab (Prolia) for premenopausal women with idiopathic osteoporosis, bone mineral density (BMD) was maintained over the first year following denosumab cessation, according to results from a small, nonrandomized extension of a phase 2 study.
Bisphosphonates are recommended for patients after they have completed a course of denosumab because cessation of the bone resorption blocker is known to increase bone turnover markers, decrease BMD, and raise the risk of vertebral fractures. Although there is evidence to support this treatment sequence for postmenopausal women, there was no evidence regarding premenopausal women with idiopathic osteoporosis, said Adi Cohen, MD, who presented the results of the study at the annual meeting of the American Society for Bone and Mineral Research.
In the extension study, neither length of treatment with denosumab nor transition to menopause affected BMD results. Weekly doses of alendronate (ALN) better suppressed C-terminal telopeptide (CTX) than did zoledronic acid (ZOL) and led to better maintenance of BMD than did a single dose of ZOL. The researchers suggested that single-dose ZOL may not prevent bone loss for an entire year.
It is too early to call the results practice changing, said Dr. Cohen, professor of medicine and endocrinology at Columbia University Irving Medical Center, New York, but she noted, “It’s important just to provide information about how sequences of osteoporosis medications might be used in a rare but certainly understudied group of premenopausal women with osteoporosis who need treatment, and these data hopefully will help make some treatment decisions.”
In the early 2000s, researchers initially believed that premenopausal women with low BMD had experienced some kind of temporary event and that they would likely improve on their own over time. “I think we now recognize that whatever it is that causes this is an ongoing issue and that this is a problem they’re going to have to deal with for the rest of their lives. This is something that they have to stay on top of,” said coauthor Elizabeth Shane, MD, who is a professor of medicine at CUIMC.
However, there are no practice guidelines for the management of osteoporosis in premenopausal women, according to Dr. Shane. She noted that there is controversy as to whether to treat women with low bone density who do not have a history of fractures. “I think that there’s pretty much agreement that anybody who has a lot of fractures has an early-onset form of osteoporosis. The controversy is what to do about the person who just has a low bone density and hasn’t yet fractured and what is the utility of trying to treat them at that point and perhaps prevent a fracture. I don’t think we have enough data to address that,” Dr. Shane said.
Still, the research has provided some clarity in her own practice. “I think if somebody would come to my office who had very low bone density, I would probably treat them. If they have fractures, I would definitely treat them. I think that our work has provided a framework for people to approach that,” she said.
The study was an extension of a sequential treatment approach that began with 2 years of teriparatide (20 mcg daily) followed by an extension study of 2–3 years of treatment with denosumab (60 mg every 6 months). Seven months after the last dose of denosumab, patients underwent 1 year of treatment with ALN (70 mg weekly; n = 18) or a single dose of ZOL (5 mg IV; n = 6), according to patient choice.
The original phase 2 study started with 41 women. At 24 months, teriparatide treatment led to BMD increases of 13% in the lumbar spine (LS), 5% in the total hip (TH), and 5% in the femoral neck (FN). There was a 2% decline in BMD in the forearm (distal radius [DR]). A group of 32 of the women participated in an extension study and took denosumab for 12 months. Of those patients, 29 continued to take it for another 12 months. At 12 months, BMD increased 5% in the LS, 3% in the TH, 3% in the FN, and 1% in the DR (P < .05 for all). At 24 months, BMD rose by 22%, 10%, and 10% at the first three of those locations. BMD in the DR remained stable, compared with the baseline after taking teriparatide.
The bisphosphonate phase of the extension study included 24 women (mean age, 43 years). The mean body mass index of the patients was 23.0 kg/m2. The patients had experienced a mean of 3.0 fractures in adulthood, and 38% of patients had a history of vertebral fracture.
Over 12 months of follow-up, the researchers found no statistically significant difference in BMD in the LS, TH, or FN, compared with bisphosphonate extension baseline. There was also no statistically significant change in serum CTX. There was evidence that, among patients with higher rates of bone turnover, there were higher rates of LS and FN bone loss during bisphosphonate treatment.
Among patients taking ZOL, at 12 months there was a statistically significant rise in CTX levels, but not among patients taking ALN. There were no new vertebral fractures among any participants during the bisphosphonate extension period.
The results represent critical data for an understudied population, according to Yumie Rhee, MD, PhD, who was comoderator of the session in which the study was presented. “They are showing that by using a bisphosphonate [patients] have this just slight decrease, but within error, so it’s maintaining the BMD, at least. I think it’s very important. It will be fascinating to see next year’s follow-up,” said Dr. Rhee, a professor of endocrinology at Yonsei University College of Medicine in Seoul, South Korea. “The problem with premenopausal osteoporosis is that we don’t have good evidence. Even though this study is very small, we’re just following that data, all of us.”
Comoderator Maria Zanchetta, MD, a professor of osteology at the Institute of Diagnostics and Metabolic Research, Universidad del Salvador, Buenos Aires, agreed. “We know what to do when we stop denosumab in postmenopausal women. We didn’t have any work about what to do when we stopped in premenopausal women. You can think that probably it’s going to be the same, but this is the first time you have the evidence that if you give bisphosphonate, you will maintain BMD.”
Limitations to the study include its small size and the lack of a placebo-treated control group. In addition, the bisphosphonate extension was not randomized.
The studies were funded by the U.S. Food and Drug Administration and Amgen. Dr. Cohen and Dr. Shane received research funding from Amgen. Dr. Rhee and Dr. Zanchetta have disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
VANCOUVER – With treatment with a bisphosphonate following sequential use of teriparatide (Forteo) and denosumab (Prolia) for premenopausal women with idiopathic osteoporosis, bone mineral density (BMD) was maintained over the first year following denosumab cessation, according to results from a small, nonrandomized extension of a phase 2 study.
Bisphosphonates are recommended for patients after they have completed a course of denosumab because cessation of the bone resorption blocker is known to increase bone turnover markers, decrease BMD, and raise the risk of vertebral fractures. Although there is evidence to support this treatment sequence for postmenopausal women, there was no evidence regarding premenopausal women with idiopathic osteoporosis, said Adi Cohen, MD, who presented the results of the study at the annual meeting of the American Society for Bone and Mineral Research.
In the extension study, neither length of treatment with denosumab nor transition to menopause affected BMD results. Weekly doses of alendronate (ALN) better suppressed C-terminal telopeptide (CTX) than did zoledronic acid (ZOL) and led to better maintenance of BMD than did a single dose of ZOL. The researchers suggested that single-dose ZOL may not prevent bone loss for an entire year.
It is too early to call the results practice changing, said Dr. Cohen, professor of medicine and endocrinology at Columbia University Irving Medical Center, New York, but she noted, “It’s important just to provide information about how sequences of osteoporosis medications might be used in a rare but certainly understudied group of premenopausal women with osteoporosis who need treatment, and these data hopefully will help make some treatment decisions.”
In the early 2000s, researchers initially believed that premenopausal women with low BMD had experienced some kind of temporary event and that they would likely improve on their own over time. “I think we now recognize that whatever it is that causes this is an ongoing issue and that this is a problem they’re going to have to deal with for the rest of their lives. This is something that they have to stay on top of,” said coauthor Elizabeth Shane, MD, who is a professor of medicine at CUIMC.
However, there are no practice guidelines for the management of osteoporosis in premenopausal women, according to Dr. Shane. She noted that there is controversy as to whether to treat women with low bone density who do not have a history of fractures. “I think that there’s pretty much agreement that anybody who has a lot of fractures has an early-onset form of osteoporosis. The controversy is what to do about the person who just has a low bone density and hasn’t yet fractured and what is the utility of trying to treat them at that point and perhaps prevent a fracture. I don’t think we have enough data to address that,” Dr. Shane said.
Still, the research has provided some clarity in her own practice. “I think if somebody would come to my office who had very low bone density, I would probably treat them. If they have fractures, I would definitely treat them. I think that our work has provided a framework for people to approach that,” she said.
The study was an extension of a sequential treatment approach that began with 2 years of teriparatide (20 mcg daily) followed by an extension study of 2–3 years of treatment with denosumab (60 mg every 6 months). Seven months after the last dose of denosumab, patients underwent 1 year of treatment with ALN (70 mg weekly; n = 18) or a single dose of ZOL (5 mg IV; n = 6), according to patient choice.
The original phase 2 study started with 41 women. At 24 months, teriparatide treatment led to BMD increases of 13% in the lumbar spine (LS), 5% in the total hip (TH), and 5% in the femoral neck (FN). There was a 2% decline in BMD in the forearm (distal radius [DR]). A group of 32 of the women participated in an extension study and took denosumab for 12 months. Of those patients, 29 continued to take it for another 12 months. At 12 months, BMD increased 5% in the LS, 3% in the TH, 3% in the FN, and 1% in the DR (P < .05 for all). At 24 months, BMD rose by 22%, 10%, and 10% at the first three of those locations. BMD in the DR remained stable, compared with the baseline after taking teriparatide.
The bisphosphonate phase of the extension study included 24 women (mean age, 43 years). The mean body mass index of the patients was 23.0 kg/m2. The patients had experienced a mean of 3.0 fractures in adulthood, and 38% of patients had a history of vertebral fracture.
Over 12 months of follow-up, the researchers found no statistically significant difference in BMD in the LS, TH, or FN, compared with bisphosphonate extension baseline. There was also no statistically significant change in serum CTX. There was evidence that, among patients with higher rates of bone turnover, there were higher rates of LS and FN bone loss during bisphosphonate treatment.
Among patients taking ZOL, at 12 months there was a statistically significant rise in CTX levels, but not among patients taking ALN. There were no new vertebral fractures among any participants during the bisphosphonate extension period.
The results represent critical data for an understudied population, according to Yumie Rhee, MD, PhD, who was comoderator of the session in which the study was presented. “They are showing that by using a bisphosphonate [patients] have this just slight decrease, but within error, so it’s maintaining the BMD, at least. I think it’s very important. It will be fascinating to see next year’s follow-up,” said Dr. Rhee, a professor of endocrinology at Yonsei University College of Medicine in Seoul, South Korea. “The problem with premenopausal osteoporosis is that we don’t have good evidence. Even though this study is very small, we’re just following that data, all of us.”
Comoderator Maria Zanchetta, MD, a professor of osteology at the Institute of Diagnostics and Metabolic Research, Universidad del Salvador, Buenos Aires, agreed. “We know what to do when we stop denosumab in postmenopausal women. We didn’t have any work about what to do when we stopped in premenopausal women. You can think that probably it’s going to be the same, but this is the first time you have the evidence that if you give bisphosphonate, you will maintain BMD.”
Limitations to the study include its small size and the lack of a placebo-treated control group. In addition, the bisphosphonate extension was not randomized.
The studies were funded by the U.S. Food and Drug Administration and Amgen. Dr. Cohen and Dr. Shane received research funding from Amgen. Dr. Rhee and Dr. Zanchetta have disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
VANCOUVER – With treatment with a bisphosphonate following sequential use of teriparatide (Forteo) and denosumab (Prolia) for premenopausal women with idiopathic osteoporosis, bone mineral density (BMD) was maintained over the first year following denosumab cessation, according to results from a small, nonrandomized extension of a phase 2 study.
Bisphosphonates are recommended for patients after they have completed a course of denosumab because cessation of the bone resorption blocker is known to increase bone turnover markers, decrease BMD, and raise the risk of vertebral fractures. Although there is evidence to support this treatment sequence for postmenopausal women, there was no evidence regarding premenopausal women with idiopathic osteoporosis, said Adi Cohen, MD, who presented the results of the study at the annual meeting of the American Society for Bone and Mineral Research.
In the extension study, neither length of treatment with denosumab nor transition to menopause affected BMD results. Weekly doses of alendronate (ALN) better suppressed C-terminal telopeptide (CTX) than did zoledronic acid (ZOL) and led to better maintenance of BMD than did a single dose of ZOL. The researchers suggested that single-dose ZOL may not prevent bone loss for an entire year.
It is too early to call the results practice changing, said Dr. Cohen, professor of medicine and endocrinology at Columbia University Irving Medical Center, New York, but she noted, “It’s important just to provide information about how sequences of osteoporosis medications might be used in a rare but certainly understudied group of premenopausal women with osteoporosis who need treatment, and these data hopefully will help make some treatment decisions.”
In the early 2000s, researchers initially believed that premenopausal women with low BMD had experienced some kind of temporary event and that they would likely improve on their own over time. “I think we now recognize that whatever it is that causes this is an ongoing issue and that this is a problem they’re going to have to deal with for the rest of their lives. This is something that they have to stay on top of,” said coauthor Elizabeth Shane, MD, who is a professor of medicine at CUIMC.
However, there are no practice guidelines for the management of osteoporosis in premenopausal women, according to Dr. Shane. She noted that there is controversy as to whether to treat women with low bone density who do not have a history of fractures. “I think that there’s pretty much agreement that anybody who has a lot of fractures has an early-onset form of osteoporosis. The controversy is what to do about the person who just has a low bone density and hasn’t yet fractured and what is the utility of trying to treat them at that point and perhaps prevent a fracture. I don’t think we have enough data to address that,” Dr. Shane said.
Still, the research has provided some clarity in her own practice. “I think if somebody would come to my office who had very low bone density, I would probably treat them. If they have fractures, I would definitely treat them. I think that our work has provided a framework for people to approach that,” she said.
The study was an extension of a sequential treatment approach that began with 2 years of teriparatide (20 mcg daily) followed by an extension study of 2–3 years of treatment with denosumab (60 mg every 6 months). Seven months after the last dose of denosumab, patients underwent 1 year of treatment with ALN (70 mg weekly; n = 18) or a single dose of ZOL (5 mg IV; n = 6), according to patient choice.
The original phase 2 study started with 41 women. At 24 months, teriparatide treatment led to BMD increases of 13% in the lumbar spine (LS), 5% in the total hip (TH), and 5% in the femoral neck (FN). There was a 2% decline in BMD in the forearm (distal radius [DR]). A group of 32 of the women participated in an extension study and took denosumab for 12 months. Of those patients, 29 continued to take it for another 12 months. At 12 months, BMD increased 5% in the LS, 3% in the TH, 3% in the FN, and 1% in the DR (P < .05 for all). At 24 months, BMD rose by 22%, 10%, and 10% at the first three of those locations. BMD in the DR remained stable, compared with the baseline after taking teriparatide.
The bisphosphonate phase of the extension study included 24 women (mean age, 43 years). The mean body mass index of the patients was 23.0 kg/m2. The patients had experienced a mean of 3.0 fractures in adulthood, and 38% of patients had a history of vertebral fracture.
Over 12 months of follow-up, the researchers found no statistically significant difference in BMD in the LS, TH, or FN, compared with bisphosphonate extension baseline. There was also no statistically significant change in serum CTX. There was evidence that, among patients with higher rates of bone turnover, there were higher rates of LS and FN bone loss during bisphosphonate treatment.
Among patients taking ZOL, at 12 months there was a statistically significant rise in CTX levels, but not among patients taking ALN. There were no new vertebral fractures among any participants during the bisphosphonate extension period.
The results represent critical data for an understudied population, according to Yumie Rhee, MD, PhD, who was comoderator of the session in which the study was presented. “They are showing that by using a bisphosphonate [patients] have this just slight decrease, but within error, so it’s maintaining the BMD, at least. I think it’s very important. It will be fascinating to see next year’s follow-up,” said Dr. Rhee, a professor of endocrinology at Yonsei University College of Medicine in Seoul, South Korea. “The problem with premenopausal osteoporosis is that we don’t have good evidence. Even though this study is very small, we’re just following that data, all of us.”
Comoderator Maria Zanchetta, MD, a professor of osteology at the Institute of Diagnostics and Metabolic Research, Universidad del Salvador, Buenos Aires, agreed. “We know what to do when we stop denosumab in postmenopausal women. We didn’t have any work about what to do when we stopped in premenopausal women. You can think that probably it’s going to be the same, but this is the first time you have the evidence that if you give bisphosphonate, you will maintain BMD.”
Limitations to the study include its small size and the lack of a placebo-treated control group. In addition, the bisphosphonate extension was not randomized.
The studies were funded by the U.S. Food and Drug Administration and Amgen. Dr. Cohen and Dr. Shane received research funding from Amgen. Dr. Rhee and Dr. Zanchetta have disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
AT ASBMR 2023
A focus on women with diabetes and their offspring
In 2021, diabetes and related complications was the 8th leading cause of death in the United States.1 As of 2022, more than 11% of the U.S. population had diabetes and 38% of the adult U.S. population had prediabetes.2 Diabetes is the most expensive chronic condition in the United States, where $1 of every $4 in health care costs is spent on care.3
Where this is most concerning is diabetes in pregnancy. While childbirth rates in the United States have decreased since the 2007 high of 4.32 million births4 to 3.66 million in 2021,5 the incidence of diabetes in pregnancy – both pregestational and gestational – has increased. The rate of pregestational diabetes in 2021 was 10.9 per 1,000 births, a 27% increase from 2016 (8.6 per 1,000).6 The percentage of those giving birth who also were diagnosed with gestational diabetes mellitus (GDM) was 8.3% in 2021, up from 6.0% in 2016.7
Adverse outcomes for an infant born to a mother with diabetes include a higher risk of obesity and diabetes as adults, potentially leading to a forward-feeding cycle.
We and our colleagues established the Diabetes in Pregnancy Study Group of North America in 1997 because we had witnessed too frequently the devastating diabetes-induced pregnancy complications in our patients. The mission we set forth was to provide a forum for dialogue among maternal-fetal medicine subspecialists. The three main goals we set forth to support this mission were to provide a catalyst for research, contribute to the creation and refinement of medical policies, and influence professional practices in diabetes in pregnancy.8
In the last quarter century, DPSG-NA, through its annual and biennial meetings, has brought together several hundred practitioners that include physicians, nurses, statisticians, researchers, nutritionists, and allied health professionals, among others. As a group, it has improved the detection and management of diabetes in pregnant women and their offspring through knowledge sharing and influencing policies on GDM screening, diagnosis, management, and treatment. Our members have shown that preconceptional counseling for women with diabetes can significantly reduce congenital malformation and perinatal mortality compared with those women with pregestational diabetes who receive no counseling.9,10
We have addressed a wide variety of topics including the paucity of data in determining the timing of delivery for women with diabetes and the Institute of Medicine/National Academy of Medicine recommendations of gestational weight gain and risks of not adhering to them. We have learned about new scientific discoveries that reveal underlying mechanisms to diabetes-related birth defects and potential therapeutic targets; and we have discussed the health literacy requirements, ethics, and opportunities for lifestyle intervention.11-16
But we need to do more.
Two risk factors are at play: Women continue to choose to have babies at later ages and their pregnancies continue to be complicated by the rising incidence of obesity (see Figure 1 and Figure 2). 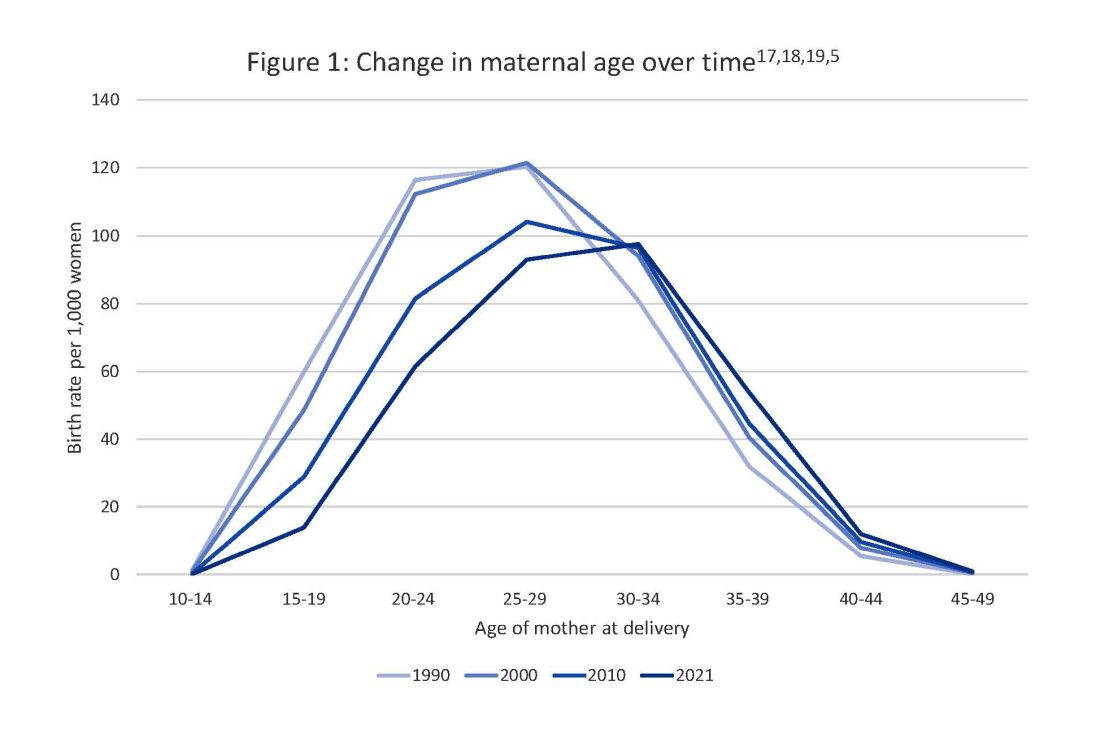
The global obesity epidemic has become a significant concern for all aspects of health and particularly for diabetes in pregnancy. 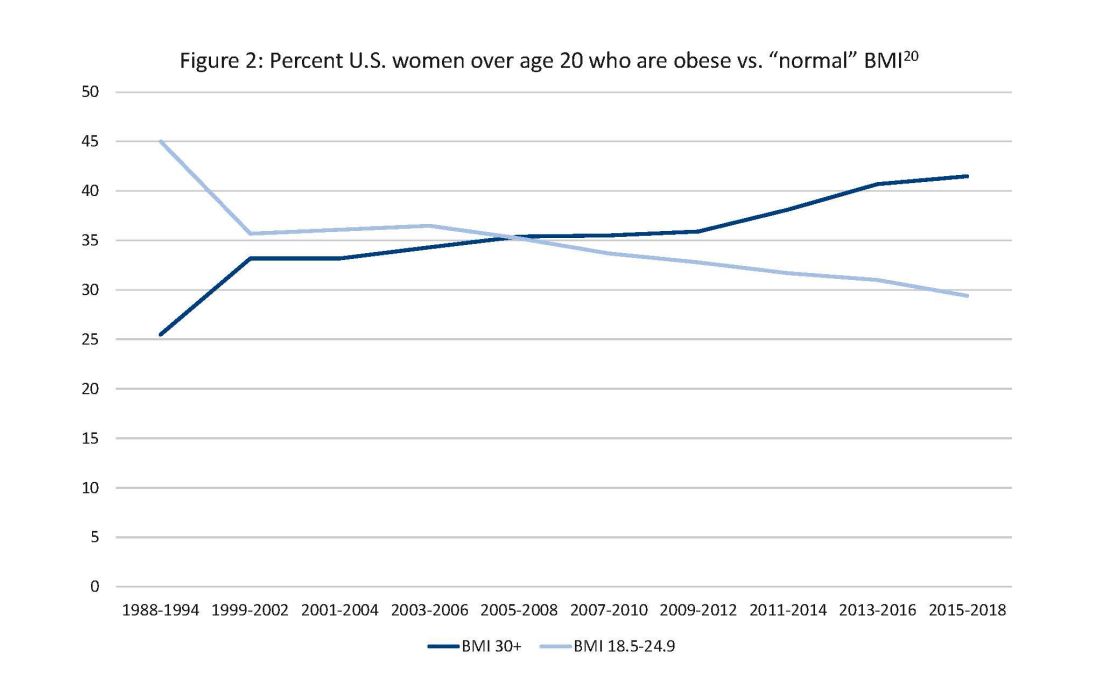
In 1990, 24.9% of women in the United States were obese; in 2010, 35.8%; and now more than 41%. Some experts project that by 2030 more than 80% of women in the United States will be overweight or obese.21
If we are to stop this cycle of diabetes begets more diabetes, now more than ever we need to come together and accelerate the research and education around the diabetes in pregnancy. Join us at this year’s DPSG-NA meeting Oct. 26-28 to take part in the knowledge sharing, discussions, and planning. More information can be found online at https://events.dpsg-na.com/home.
Dr. Miodovnik is adjunct professor of obstetrics, gynecology, and reproductive sciences at University of Maryland School of Medicine. Dr. Reece is professor of obstetrics, gynecology, and reproductive sciences and senior scientist at the Center for Birth Defects Research at University of Maryland School of Medicine.
References
1. Xu J et al. Mortality in the United States, 2021. NCHS Data Brief. 2022 Dec;(456):1-8. PMID: 36598387.
2. Centers for Disease Control and Prevention, diabetes data and statistics.
3. American Diabetes Association. The Cost of Diabetes.
4. Martin JA et al. Births: Final data for 2007. Natl Vital Stat Rep. 2010 Aug 9;58(24):1-85. PMID: 21254725.
5. Osterman MJK et al. Births: Final data for 2021. Natl Vital Stat Rep. 2023 Jan;72(1):1-53. PMID: 36723449.
6. Gregory ECW and Ely DM. Trends and characteristics in prepregnancy diabetes: United States, 2016-2021. Natl Vital Stat Rep. 2023 May;72(6):1-13. PMID: 37256333.
7. QuickStats: Percentage of mothers with gestational diabetes, by maternal age – National Vital Statistics System, United States, 2016 and 2021. MMWR Morb Mortal Wkly Rep. 2023 Jan 6;72(1):16. doi: 10.15585/mmwr.mm7201a4.
8. Langer O et al. The Diabetes in Pregnancy Study Group of North America – Introduction and summary statement. Prenat Neonat Med. 1998;3(6):514-6.
9. Willhoite MB et al. The impact of preconception counseling on pregnancy outcomes. The experience of the Maine Diabetes in Pregnancy Program. Diabetes Care. 1993 Feb;16(2):450-5. doi: 10.2337/diacare.16.2.450.
10. McElvy SS et al. A focused preconceptional and early pregnancy program in women with type 1 diabetes reduces perinatal mortality and malformation rates to general population levels. J Matern Fetal Med. 2000 Jan-Feb;9(1):14-20. doi: 10.1002/(SICI)1520-6661(200001/02)9:1<14::AID-MFM5>3.0.CO;2-K.
11. Rosen JA et al. The history and contributions of the Diabetes in Pregnancy Study Group of North America (1997-2015). Am J Perinatol. 2016 Nov;33(13):1223-6. doi: 10.1055/s-0036-1585082.
12. Driggers RW and Baschat A. The 12th meeting of the Diabetes in Pregnancy Study Group of North America (DPSG-NA): Introduction and overview. J Matern Fetal Neonatal Med. 2012 Jan;25(1):3-4. doi: 10.3109/14767058.2012.626917.
13. Langer O et al. The proceedings of the Diabetes in Pregnancy Study Group of North America 2009 conference. J Matern Fetal Neonatal Med. 2010 Mar;23(3):196-8. doi: 10.3109/14767050903550634.
14. Reece EA et al. A consensus report of the Diabetes in Pregnancy Study Group of North America Conference, Little Rock, Ark., May 2002. J Matern Fetal Neonatal Med. 2002 Dec;12(6):362-4. doi: 10.1080/jmf.12.6.362.364.
15. Reece EA and Maulik D. A consensus conference of the Diabetes in Pregnancy Study Group of North America. J Matern Fetal Neonatal Med. 2002 Dec;12(6):361. doi: 10.1080/jmf.12.6.361.361.
16. Gabbe SG. Summation of the second meeting of the Diabetes in Pregnancy Study Group of North America (DPSG-NA). J Matern Fetal Med. 2000 Jan-Feb;9(1):3-9.
17. Vital Statistics of the United States 1990: Volume I – Natality.
18. Martin JA et al. Births: final data for 2000. Natl Vital Stat Rep. 2002 Feb 12;50(5):1-101. PMID: 11876093.
19. Martin JA et al. Births: final data for 2010. Natl Vital Stat Rep. 2012 Aug 28;61(1):1-72. PMID: 24974589.
20. CDC Website. Normal weight, overweight, and obesity among adults aged 20 and over, by selected characteristics: United States.
21. Wang Y et al. Has the prevalence of overweight, obesity, and central obesity levelled off in the United States? Trends, patterns, disparities, and future projections for the obesity epidemic. Int J Epidemiol. 2020 Jun 1;49(3):810-23. doi: 10.1093/ije/dyz273.
In 2021, diabetes and related complications was the 8th leading cause of death in the United States.1 As of 2022, more than 11% of the U.S. population had diabetes and 38% of the adult U.S. population had prediabetes.2 Diabetes is the most expensive chronic condition in the United States, where $1 of every $4 in health care costs is spent on care.3
Where this is most concerning is diabetes in pregnancy. While childbirth rates in the United States have decreased since the 2007 high of 4.32 million births4 to 3.66 million in 2021,5 the incidence of diabetes in pregnancy – both pregestational and gestational – has increased. The rate of pregestational diabetes in 2021 was 10.9 per 1,000 births, a 27% increase from 2016 (8.6 per 1,000).6 The percentage of those giving birth who also were diagnosed with gestational diabetes mellitus (GDM) was 8.3% in 2021, up from 6.0% in 2016.7
Adverse outcomes for an infant born to a mother with diabetes include a higher risk of obesity and diabetes as adults, potentially leading to a forward-feeding cycle.
We and our colleagues established the Diabetes in Pregnancy Study Group of North America in 1997 because we had witnessed too frequently the devastating diabetes-induced pregnancy complications in our patients. The mission we set forth was to provide a forum for dialogue among maternal-fetal medicine subspecialists. The three main goals we set forth to support this mission were to provide a catalyst for research, contribute to the creation and refinement of medical policies, and influence professional practices in diabetes in pregnancy.8
In the last quarter century, DPSG-NA, through its annual and biennial meetings, has brought together several hundred practitioners that include physicians, nurses, statisticians, researchers, nutritionists, and allied health professionals, among others. As a group, it has improved the detection and management of diabetes in pregnant women and their offspring through knowledge sharing and influencing policies on GDM screening, diagnosis, management, and treatment. Our members have shown that preconceptional counseling for women with diabetes can significantly reduce congenital malformation and perinatal mortality compared with those women with pregestational diabetes who receive no counseling.9,10
We have addressed a wide variety of topics including the paucity of data in determining the timing of delivery for women with diabetes and the Institute of Medicine/National Academy of Medicine recommendations of gestational weight gain and risks of not adhering to them. We have learned about new scientific discoveries that reveal underlying mechanisms to diabetes-related birth defects and potential therapeutic targets; and we have discussed the health literacy requirements, ethics, and opportunities for lifestyle intervention.11-16
But we need to do more.
Two risk factors are at play: Women continue to choose to have babies at later ages and their pregnancies continue to be complicated by the rising incidence of obesity (see Figure 1 and Figure 2). 
The global obesity epidemic has become a significant concern for all aspects of health and particularly for diabetes in pregnancy. 
In 1990, 24.9% of women in the United States were obese; in 2010, 35.8%; and now more than 41%. Some experts project that by 2030 more than 80% of women in the United States will be overweight or obese.21
If we are to stop this cycle of diabetes begets more diabetes, now more than ever we need to come together and accelerate the research and education around the diabetes in pregnancy. Join us at this year’s DPSG-NA meeting Oct. 26-28 to take part in the knowledge sharing, discussions, and planning. More information can be found online at https://events.dpsg-na.com/home.
Dr. Miodovnik is adjunct professor of obstetrics, gynecology, and reproductive sciences at University of Maryland School of Medicine. Dr. Reece is professor of obstetrics, gynecology, and reproductive sciences and senior scientist at the Center for Birth Defects Research at University of Maryland School of Medicine.
References
1. Xu J et al. Mortality in the United States, 2021. NCHS Data Brief. 2022 Dec;(456):1-8. PMID: 36598387.
2. Centers for Disease Control and Prevention, diabetes data and statistics.
3. American Diabetes Association. The Cost of Diabetes.
4. Martin JA et al. Births: Final data for 2007. Natl Vital Stat Rep. 2010 Aug 9;58(24):1-85. PMID: 21254725.
5. Osterman MJK et al. Births: Final data for 2021. Natl Vital Stat Rep. 2023 Jan;72(1):1-53. PMID: 36723449.
6. Gregory ECW and Ely DM. Trends and characteristics in prepregnancy diabetes: United States, 2016-2021. Natl Vital Stat Rep. 2023 May;72(6):1-13. PMID: 37256333.
7. QuickStats: Percentage of mothers with gestational diabetes, by maternal age – National Vital Statistics System, United States, 2016 and 2021. MMWR Morb Mortal Wkly Rep. 2023 Jan 6;72(1):16. doi: 10.15585/mmwr.mm7201a4.
8. Langer O et al. The Diabetes in Pregnancy Study Group of North America – Introduction and summary statement. Prenat Neonat Med. 1998;3(6):514-6.
9. Willhoite MB et al. The impact of preconception counseling on pregnancy outcomes. The experience of the Maine Diabetes in Pregnancy Program. Diabetes Care. 1993 Feb;16(2):450-5. doi: 10.2337/diacare.16.2.450.
10. McElvy SS et al. A focused preconceptional and early pregnancy program in women with type 1 diabetes reduces perinatal mortality and malformation rates to general population levels. J Matern Fetal Med. 2000 Jan-Feb;9(1):14-20. doi: 10.1002/(SICI)1520-6661(200001/02)9:1<14::AID-MFM5>3.0.CO;2-K.
11. Rosen JA et al. The history and contributions of the Diabetes in Pregnancy Study Group of North America (1997-2015). Am J Perinatol. 2016 Nov;33(13):1223-6. doi: 10.1055/s-0036-1585082.
12. Driggers RW and Baschat A. The 12th meeting of the Diabetes in Pregnancy Study Group of North America (DPSG-NA): Introduction and overview. J Matern Fetal Neonatal Med. 2012 Jan;25(1):3-4. doi: 10.3109/14767058.2012.626917.
13. Langer O et al. The proceedings of the Diabetes in Pregnancy Study Group of North America 2009 conference. J Matern Fetal Neonatal Med. 2010 Mar;23(3):196-8. doi: 10.3109/14767050903550634.
14. Reece EA et al. A consensus report of the Diabetes in Pregnancy Study Group of North America Conference, Little Rock, Ark., May 2002. J Matern Fetal Neonatal Med. 2002 Dec;12(6):362-4. doi: 10.1080/jmf.12.6.362.364.
15. Reece EA and Maulik D. A consensus conference of the Diabetes in Pregnancy Study Group of North America. J Matern Fetal Neonatal Med. 2002 Dec;12(6):361. doi: 10.1080/jmf.12.6.361.361.
16. Gabbe SG. Summation of the second meeting of the Diabetes in Pregnancy Study Group of North America (DPSG-NA). J Matern Fetal Med. 2000 Jan-Feb;9(1):3-9.
17. Vital Statistics of the United States 1990: Volume I – Natality.
18. Martin JA et al. Births: final data for 2000. Natl Vital Stat Rep. 2002 Feb 12;50(5):1-101. PMID: 11876093.
19. Martin JA et al. Births: final data for 2010. Natl Vital Stat Rep. 2012 Aug 28;61(1):1-72. PMID: 24974589.
20. CDC Website. Normal weight, overweight, and obesity among adults aged 20 and over, by selected characteristics: United States.
21. Wang Y et al. Has the prevalence of overweight, obesity, and central obesity levelled off in the United States? Trends, patterns, disparities, and future projections for the obesity epidemic. Int J Epidemiol. 2020 Jun 1;49(3):810-23. doi: 10.1093/ije/dyz273.
In 2021, diabetes and related complications was the 8th leading cause of death in the United States.1 As of 2022, more than 11% of the U.S. population had diabetes and 38% of the adult U.S. population had prediabetes.2 Diabetes is the most expensive chronic condition in the United States, where $1 of every $4 in health care costs is spent on care.3
Where this is most concerning is diabetes in pregnancy. While childbirth rates in the United States have decreased since the 2007 high of 4.32 million births4 to 3.66 million in 2021,5 the incidence of diabetes in pregnancy – both pregestational and gestational – has increased. The rate of pregestational diabetes in 2021 was 10.9 per 1,000 births, a 27% increase from 2016 (8.6 per 1,000).6 The percentage of those giving birth who also were diagnosed with gestational diabetes mellitus (GDM) was 8.3% in 2021, up from 6.0% in 2016.7
Adverse outcomes for an infant born to a mother with diabetes include a higher risk of obesity and diabetes as adults, potentially leading to a forward-feeding cycle.
We and our colleagues established the Diabetes in Pregnancy Study Group of North America in 1997 because we had witnessed too frequently the devastating diabetes-induced pregnancy complications in our patients. The mission we set forth was to provide a forum for dialogue among maternal-fetal medicine subspecialists. The three main goals we set forth to support this mission were to provide a catalyst for research, contribute to the creation and refinement of medical policies, and influence professional practices in diabetes in pregnancy.8
In the last quarter century, DPSG-NA, through its annual and biennial meetings, has brought together several hundred practitioners that include physicians, nurses, statisticians, researchers, nutritionists, and allied health professionals, among others. As a group, it has improved the detection and management of diabetes in pregnant women and their offspring through knowledge sharing and influencing policies on GDM screening, diagnosis, management, and treatment. Our members have shown that preconceptional counseling for women with diabetes can significantly reduce congenital malformation and perinatal mortality compared with those women with pregestational diabetes who receive no counseling.9,10
We have addressed a wide variety of topics including the paucity of data in determining the timing of delivery for women with diabetes and the Institute of Medicine/National Academy of Medicine recommendations of gestational weight gain and risks of not adhering to them. We have learned about new scientific discoveries that reveal underlying mechanisms to diabetes-related birth defects and potential therapeutic targets; and we have discussed the health literacy requirements, ethics, and opportunities for lifestyle intervention.11-16
But we need to do more.
Two risk factors are at play: Women continue to choose to have babies at later ages and their pregnancies continue to be complicated by the rising incidence of obesity (see Figure 1 and Figure 2). 
The global obesity epidemic has become a significant concern for all aspects of health and particularly for diabetes in pregnancy. 
In 1990, 24.9% of women in the United States were obese; in 2010, 35.8%; and now more than 41%. Some experts project that by 2030 more than 80% of women in the United States will be overweight or obese.21
If we are to stop this cycle of diabetes begets more diabetes, now more than ever we need to come together and accelerate the research and education around the diabetes in pregnancy. Join us at this year’s DPSG-NA meeting Oct. 26-28 to take part in the knowledge sharing, discussions, and planning. More information can be found online at https://events.dpsg-na.com/home.
Dr. Miodovnik is adjunct professor of obstetrics, gynecology, and reproductive sciences at University of Maryland School of Medicine. Dr. Reece is professor of obstetrics, gynecology, and reproductive sciences and senior scientist at the Center for Birth Defects Research at University of Maryland School of Medicine.
References
1. Xu J et al. Mortality in the United States, 2021. NCHS Data Brief. 2022 Dec;(456):1-8. PMID: 36598387.
2. Centers for Disease Control and Prevention, diabetes data and statistics.
3. American Diabetes Association. The Cost of Diabetes.
4. Martin JA et al. Births: Final data for 2007. Natl Vital Stat Rep. 2010 Aug 9;58(24):1-85. PMID: 21254725.
5. Osterman MJK et al. Births: Final data for 2021. Natl Vital Stat Rep. 2023 Jan;72(1):1-53. PMID: 36723449.
6. Gregory ECW and Ely DM. Trends and characteristics in prepregnancy diabetes: United States, 2016-2021. Natl Vital Stat Rep. 2023 May;72(6):1-13. PMID: 37256333.
7. QuickStats: Percentage of mothers with gestational diabetes, by maternal age – National Vital Statistics System, United States, 2016 and 2021. MMWR Morb Mortal Wkly Rep. 2023 Jan 6;72(1):16. doi: 10.15585/mmwr.mm7201a4.
8. Langer O et al. The Diabetes in Pregnancy Study Group of North America – Introduction and summary statement. Prenat Neonat Med. 1998;3(6):514-6.
9. Willhoite MB et al. The impact of preconception counseling on pregnancy outcomes. The experience of the Maine Diabetes in Pregnancy Program. Diabetes Care. 1993 Feb;16(2):450-5. doi: 10.2337/diacare.16.2.450.
10. McElvy SS et al. A focused preconceptional and early pregnancy program in women with type 1 diabetes reduces perinatal mortality and malformation rates to general population levels. J Matern Fetal Med. 2000 Jan-Feb;9(1):14-20. doi: 10.1002/(SICI)1520-6661(200001/02)9:1<14::AID-MFM5>3.0.CO;2-K.
11. Rosen JA et al. The history and contributions of the Diabetes in Pregnancy Study Group of North America (1997-2015). Am J Perinatol. 2016 Nov;33(13):1223-6. doi: 10.1055/s-0036-1585082.
12. Driggers RW and Baschat A. The 12th meeting of the Diabetes in Pregnancy Study Group of North America (DPSG-NA): Introduction and overview. J Matern Fetal Neonatal Med. 2012 Jan;25(1):3-4. doi: 10.3109/14767058.2012.626917.
13. Langer O et al. The proceedings of the Diabetes in Pregnancy Study Group of North America 2009 conference. J Matern Fetal Neonatal Med. 2010 Mar;23(3):196-8. doi: 10.3109/14767050903550634.
14. Reece EA et al. A consensus report of the Diabetes in Pregnancy Study Group of North America Conference, Little Rock, Ark., May 2002. J Matern Fetal Neonatal Med. 2002 Dec;12(6):362-4. doi: 10.1080/jmf.12.6.362.364.
15. Reece EA and Maulik D. A consensus conference of the Diabetes in Pregnancy Study Group of North America. J Matern Fetal Neonatal Med. 2002 Dec;12(6):361. doi: 10.1080/jmf.12.6.361.361.
16. Gabbe SG. Summation of the second meeting of the Diabetes in Pregnancy Study Group of North America (DPSG-NA). J Matern Fetal Med. 2000 Jan-Feb;9(1):3-9.
17. Vital Statistics of the United States 1990: Volume I – Natality.
18. Martin JA et al. Births: final data for 2000. Natl Vital Stat Rep. 2002 Feb 12;50(5):1-101. PMID: 11876093.
19. Martin JA et al. Births: final data for 2010. Natl Vital Stat Rep. 2012 Aug 28;61(1):1-72. PMID: 24974589.
20. CDC Website. Normal weight, overweight, and obesity among adults aged 20 and over, by selected characteristics: United States.
21. Wang Y et al. Has the prevalence of overweight, obesity, and central obesity levelled off in the United States? Trends, patterns, disparities, and future projections for the obesity epidemic. Int J Epidemiol. 2020 Jun 1;49(3):810-23. doi: 10.1093/ije/dyz273.
Taking a new obesity drug and birth control pills? Be careful
For women who are obese, daily life is wrought with landmines. Whether it’s the challenges of air travel because plane seats are too small, the need to shield themselves from the world’s discriminating eyes, or the great lengths many will go to achieve better health and the promise of longevity, navigating life as an obese person requires a thick skin.
So, it’s no wonder so many are willing to pay more than $1,000 a month out of pocket to get their hands on drugs like semaglutide (Ozempic and Wegovy) or tirzepatide (Mounjaro). The benefits of these drugs, which are part of a new class called glucagonlike peptide–1 (GLP-1) receptor agonists, include significant and rapid weight loss, blood sugar control, and improved life quality; they are unprecedented in a setting where surgery has long been considered the most effective long-term option.
On the flip side, the desire for rapid weight loss and better blood sugar control also comes with an unexpected cost. , making an unintended pregnancy more likely.
Neel Shah, MD, an endocrinologist and associate professor at the University of Texas Health Science Center at Houston, said he has had several patients become pregnant without intending to.
“It was when Mounjaro came out on the market when we started using it,” he said of the drug the Food and Drug Administration approved for type 2 diabetes in 2022. “It [the warning] was in the product insert, but clinically speaking, I don’t know if it was at the top of providers’ minds when they were prescribing Mounjaro.”
When asked if he believed that we were going to be seeing a significant increase in so-called Mounjaro babies, Dr. Shah was sure in his response.
“Absolutely. We will because the sheer volume [of patients] will increase,” he said.
It’s all in the gut
One of the ways that drugs like Mounjaro work is by delaying the time that it takes for food to move from the stomach to the small intestine. Although data are still evolving, it is believed that this process – delayed gastric emptying – may affect the absorption of birth control pills.
Dr. Shah said another theory is that vomiting, which is a common side effect of these types of drugs, also affects the pills’ ability to prevent pregnancy.
And “there’s a prolonged period of ramping up the dose because of the GI side effects,” said Pinar Kodaman, MD, PhD, a reproductive endocrinologist and assistant professor of gynecology at Yale University in New Haven, Conn.
“Initially, at the lowest dose, there may not be a lot of potential effect on absorption and gastric emptying. But as the dose goes up, it becomes more common, and it can cause diarrhea, which is another condition that can affect the absorption of any medication,” she said.
Unanticipated outcomes, extra prevention
Roughly 42% of women in the United States are obese, 40% of whom are between the ages of 20 and 39. Although these new drugs can improve fertility outcomes for women who are obese (especially those with polycystic ovary syndrome, or PCOS), only one – Mounjaro – currently carries a warning about birth control pill effectiveness on its label. Unfortunately, it appears that some doctors are unaware or not counseling patients about this risk, and the data are unclear about whether other drugs in this class, like Ozempic and Wegovy, have the same risks.
“To date, it hasn’t been a typical thing that we counsel about,” said Dr. Kodaman. “It’s all fairly new, but when we have patients on birth control pills, we do review other medications that they are on because some can affect efficacy, and it’s something to keep in mind.”
It’s also unclear if other forms of birth control – for example, birth control patches that deliver through the skin – might carry similar pregnancy risks. Dr. Shah said some of his patients who became pregnant without intending to were using these patches. This raises even more questions, since they deliver drugs through the skin directly into the bloodstream and not through the GI system.
What can women do to help ensure that they don’t become pregnant while using these drugs?
“I really think that if patients want to protect themselves from an unplanned pregnancy, that as soon as they start the GLP receptor agonists, it wouldn’t be a bad idea to use condoms, because the onset of action is pretty quick,” said Dr. Kodaman, noting also that “at the lowest dose there may not be a lot of potential effect on gastric emptying. But as the dose goes up, it becomes much more common or can cause diarrhea.”
Dr. Shah said that in his practice he’s “been telling patients to add barrier contraception” 4 weeks before they start their first dose “and at any dose adjustment.”
Zoobia Chaudhry, an obesity medicine doctor and assistant professor of medicine at Johns Hopkins University in Baltimore, recommends that “patients just make sure that the injection and medication that they take are at least 1 hour apart.”
“Most of the time, patients do take birth control before bedtime, so if the two are spaced, it should be OK,” she said.
Another option is for women to speak to their doctors about other contraceptive options like IUDs or implantable rods, where gastric absorption is not going to be an issue.
“There’s very little research on this class of drugs,” said Emily Goodstein, a 40-year-old small-business owner in Washington, who recently switched from Ozempic to Mounjaro. “Being a person who lives in a larger body is such a horrifying experience because of the way that the world discriminates against you.”
She appreciates the feeling of being proactive that these new drugs grant. It has “opened up a bunch of opportunities for me to be seen as a full individual by the medical establishment,” she said. “I was willing to take the risk, knowing that I would be on these drugs for the rest of my life.”
In addition to being what Dr. Goodstein refers to as a guinea pig, she said she made sure that her primary care doctor was aware that she was not trying or planning to become pregnant again. (She has a 3-year-old child.) Still, her doctor mentioned only the most common side effects linked to these drugs, like nausea, vomiting, and diarrhea, and did not mention the risk of pregnancy.
“Folks are really not talking about the reproductive implications,” she said, referring to members of a Facebook group on these drugs that she belongs to.
Like patients themselves, many doctors are just beginning to get their arms around these agents. “Awareness, education, provider involvement, and having a multidisciplinary team could help patients achieve the goals that they set out for themselves,” said Dr. Shah.
Clear conversations are key.
A version of this article first appeared on WebMD.com.
For women who are obese, daily life is wrought with landmines. Whether it’s the challenges of air travel because plane seats are too small, the need to shield themselves from the world’s discriminating eyes, or the great lengths many will go to achieve better health and the promise of longevity, navigating life as an obese person requires a thick skin.
So, it’s no wonder so many are willing to pay more than $1,000 a month out of pocket to get their hands on drugs like semaglutide (Ozempic and Wegovy) or tirzepatide (Mounjaro). The benefits of these drugs, which are part of a new class called glucagonlike peptide–1 (GLP-1) receptor agonists, include significant and rapid weight loss, blood sugar control, and improved life quality; they are unprecedented in a setting where surgery has long been considered the most effective long-term option.
On the flip side, the desire for rapid weight loss and better blood sugar control also comes with an unexpected cost. , making an unintended pregnancy more likely.
Neel Shah, MD, an endocrinologist and associate professor at the University of Texas Health Science Center at Houston, said he has had several patients become pregnant without intending to.
“It was when Mounjaro came out on the market when we started using it,” he said of the drug the Food and Drug Administration approved for type 2 diabetes in 2022. “It [the warning] was in the product insert, but clinically speaking, I don’t know if it was at the top of providers’ minds when they were prescribing Mounjaro.”
When asked if he believed that we were going to be seeing a significant increase in so-called Mounjaro babies, Dr. Shah was sure in his response.
“Absolutely. We will because the sheer volume [of patients] will increase,” he said.
It’s all in the gut
One of the ways that drugs like Mounjaro work is by delaying the time that it takes for food to move from the stomach to the small intestine. Although data are still evolving, it is believed that this process – delayed gastric emptying – may affect the absorption of birth control pills.
Dr. Shah said another theory is that vomiting, which is a common side effect of these types of drugs, also affects the pills’ ability to prevent pregnancy.
And “there’s a prolonged period of ramping up the dose because of the GI side effects,” said Pinar Kodaman, MD, PhD, a reproductive endocrinologist and assistant professor of gynecology at Yale University in New Haven, Conn.
“Initially, at the lowest dose, there may not be a lot of potential effect on absorption and gastric emptying. But as the dose goes up, it becomes more common, and it can cause diarrhea, which is another condition that can affect the absorption of any medication,” she said.
Unanticipated outcomes, extra prevention
Roughly 42% of women in the United States are obese, 40% of whom are between the ages of 20 and 39. Although these new drugs can improve fertility outcomes for women who are obese (especially those with polycystic ovary syndrome, or PCOS), only one – Mounjaro – currently carries a warning about birth control pill effectiveness on its label. Unfortunately, it appears that some doctors are unaware or not counseling patients about this risk, and the data are unclear about whether other drugs in this class, like Ozempic and Wegovy, have the same risks.
“To date, it hasn’t been a typical thing that we counsel about,” said Dr. Kodaman. “It’s all fairly new, but when we have patients on birth control pills, we do review other medications that they are on because some can affect efficacy, and it’s something to keep in mind.”
It’s also unclear if other forms of birth control – for example, birth control patches that deliver through the skin – might carry similar pregnancy risks. Dr. Shah said some of his patients who became pregnant without intending to were using these patches. This raises even more questions, since they deliver drugs through the skin directly into the bloodstream and not through the GI system.
What can women do to help ensure that they don’t become pregnant while using these drugs?
“I really think that if patients want to protect themselves from an unplanned pregnancy, that as soon as they start the GLP receptor agonists, it wouldn’t be a bad idea to use condoms, because the onset of action is pretty quick,” said Dr. Kodaman, noting also that “at the lowest dose there may not be a lot of potential effect on gastric emptying. But as the dose goes up, it becomes much more common or can cause diarrhea.”
Dr. Shah said that in his practice he’s “been telling patients to add barrier contraception” 4 weeks before they start their first dose “and at any dose adjustment.”
Zoobia Chaudhry, an obesity medicine doctor and assistant professor of medicine at Johns Hopkins University in Baltimore, recommends that “patients just make sure that the injection and medication that they take are at least 1 hour apart.”
“Most of the time, patients do take birth control before bedtime, so if the two are spaced, it should be OK,” she said.
Another option is for women to speak to their doctors about other contraceptive options like IUDs or implantable rods, where gastric absorption is not going to be an issue.
“There’s very little research on this class of drugs,” said Emily Goodstein, a 40-year-old small-business owner in Washington, who recently switched from Ozempic to Mounjaro. “Being a person who lives in a larger body is such a horrifying experience because of the way that the world discriminates against you.”
She appreciates the feeling of being proactive that these new drugs grant. It has “opened up a bunch of opportunities for me to be seen as a full individual by the medical establishment,” she said. “I was willing to take the risk, knowing that I would be on these drugs for the rest of my life.”
In addition to being what Dr. Goodstein refers to as a guinea pig, she said she made sure that her primary care doctor was aware that she was not trying or planning to become pregnant again. (She has a 3-year-old child.) Still, her doctor mentioned only the most common side effects linked to these drugs, like nausea, vomiting, and diarrhea, and did not mention the risk of pregnancy.
“Folks are really not talking about the reproductive implications,” she said, referring to members of a Facebook group on these drugs that she belongs to.
Like patients themselves, many doctors are just beginning to get their arms around these agents. “Awareness, education, provider involvement, and having a multidisciplinary team could help patients achieve the goals that they set out for themselves,” said Dr. Shah.
Clear conversations are key.
A version of this article first appeared on WebMD.com.
For women who are obese, daily life is wrought with landmines. Whether it’s the challenges of air travel because plane seats are too small, the need to shield themselves from the world’s discriminating eyes, or the great lengths many will go to achieve better health and the promise of longevity, navigating life as an obese person requires a thick skin.
So, it’s no wonder so many are willing to pay more than $1,000 a month out of pocket to get their hands on drugs like semaglutide (Ozempic and Wegovy) or tirzepatide (Mounjaro). The benefits of these drugs, which are part of a new class called glucagonlike peptide–1 (GLP-1) receptor agonists, include significant and rapid weight loss, blood sugar control, and improved life quality; they are unprecedented in a setting where surgery has long been considered the most effective long-term option.
On the flip side, the desire for rapid weight loss and better blood sugar control also comes with an unexpected cost. , making an unintended pregnancy more likely.
Neel Shah, MD, an endocrinologist and associate professor at the University of Texas Health Science Center at Houston, said he has had several patients become pregnant without intending to.
“It was when Mounjaro came out on the market when we started using it,” he said of the drug the Food and Drug Administration approved for type 2 diabetes in 2022. “It [the warning] was in the product insert, but clinically speaking, I don’t know if it was at the top of providers’ minds when they were prescribing Mounjaro.”
When asked if he believed that we were going to be seeing a significant increase in so-called Mounjaro babies, Dr. Shah was sure in his response.
“Absolutely. We will because the sheer volume [of patients] will increase,” he said.
It’s all in the gut
One of the ways that drugs like Mounjaro work is by delaying the time that it takes for food to move from the stomach to the small intestine. Although data are still evolving, it is believed that this process – delayed gastric emptying – may affect the absorption of birth control pills.
Dr. Shah said another theory is that vomiting, which is a common side effect of these types of drugs, also affects the pills’ ability to prevent pregnancy.
And “there’s a prolonged period of ramping up the dose because of the GI side effects,” said Pinar Kodaman, MD, PhD, a reproductive endocrinologist and assistant professor of gynecology at Yale University in New Haven, Conn.
“Initially, at the lowest dose, there may not be a lot of potential effect on absorption and gastric emptying. But as the dose goes up, it becomes more common, and it can cause diarrhea, which is another condition that can affect the absorption of any medication,” she said.
Unanticipated outcomes, extra prevention
Roughly 42% of women in the United States are obese, 40% of whom are between the ages of 20 and 39. Although these new drugs can improve fertility outcomes for women who are obese (especially those with polycystic ovary syndrome, or PCOS), only one – Mounjaro – currently carries a warning about birth control pill effectiveness on its label. Unfortunately, it appears that some doctors are unaware or not counseling patients about this risk, and the data are unclear about whether other drugs in this class, like Ozempic and Wegovy, have the same risks.
“To date, it hasn’t been a typical thing that we counsel about,” said Dr. Kodaman. “It’s all fairly new, but when we have patients on birth control pills, we do review other medications that they are on because some can affect efficacy, and it’s something to keep in mind.”
It’s also unclear if other forms of birth control – for example, birth control patches that deliver through the skin – might carry similar pregnancy risks. Dr. Shah said some of his patients who became pregnant without intending to were using these patches. This raises even more questions, since they deliver drugs through the skin directly into the bloodstream and not through the GI system.
What can women do to help ensure that they don’t become pregnant while using these drugs?
“I really think that if patients want to protect themselves from an unplanned pregnancy, that as soon as they start the GLP receptor agonists, it wouldn’t be a bad idea to use condoms, because the onset of action is pretty quick,” said Dr. Kodaman, noting also that “at the lowest dose there may not be a lot of potential effect on gastric emptying. But as the dose goes up, it becomes much more common or can cause diarrhea.”
Dr. Shah said that in his practice he’s “been telling patients to add barrier contraception” 4 weeks before they start their first dose “and at any dose adjustment.”
Zoobia Chaudhry, an obesity medicine doctor and assistant professor of medicine at Johns Hopkins University in Baltimore, recommends that “patients just make sure that the injection and medication that they take are at least 1 hour apart.”
“Most of the time, patients do take birth control before bedtime, so if the two are spaced, it should be OK,” she said.
Another option is for women to speak to their doctors about other contraceptive options like IUDs or implantable rods, where gastric absorption is not going to be an issue.
“There’s very little research on this class of drugs,” said Emily Goodstein, a 40-year-old small-business owner in Washington, who recently switched from Ozempic to Mounjaro. “Being a person who lives in a larger body is such a horrifying experience because of the way that the world discriminates against you.”
She appreciates the feeling of being proactive that these new drugs grant. It has “opened up a bunch of opportunities for me to be seen as a full individual by the medical establishment,” she said. “I was willing to take the risk, knowing that I would be on these drugs for the rest of my life.”
In addition to being what Dr. Goodstein refers to as a guinea pig, she said she made sure that her primary care doctor was aware that she was not trying or planning to become pregnant again. (She has a 3-year-old child.) Still, her doctor mentioned only the most common side effects linked to these drugs, like nausea, vomiting, and diarrhea, and did not mention the risk of pregnancy.
“Folks are really not talking about the reproductive implications,” she said, referring to members of a Facebook group on these drugs that she belongs to.
Like patients themselves, many doctors are just beginning to get their arms around these agents. “Awareness, education, provider involvement, and having a multidisciplinary team could help patients achieve the goals that they set out for themselves,” said Dr. Shah.
Clear conversations are key.
A version of this article first appeared on WebMD.com.
Scientists find the ‘on’ switch for energy-burning brown fat
A process your body uses to stay warm in cool weather could one day lead to new therapies for obesity.
Scientists have, for the first time, mapped the precise nerve pathways that activate brown fat, or brown adipose tissue (BAT), a specialized fat that generates heat. Low temperatures kick brown fat into gear, helping the body keep its temperature and burning calories in the process.
“It has long been speculated that activating this type of fat may be useful in treating obesity and related metabolic conditions,” said Preethi Srikanthan, MD, an endocrinologist and professor of medicine who oversaw the research at the UCLA School of Medicine. “The challenge has been finding a way of selectively stimulating [it].”
Brown fat is different from the fat typically linked to obesity: the kind that accumulates around the belly, hips, and thighs. That’s white fat. White fat stores energy; brown fat burns it. That’s because brown fat cells have more mitochondria, a part of the cell that generates energy.
After dissecting the necks of eight human cadavers, Dr. Srikanthan and her team traced the sympathetic nerve branches in the fat pad above the collarbone – where the largest depot of brown fat in adults is stored. They stained the nerves, took samples, and viewed them under a microscope.
They found that nerves from brown fat traveled to the third and fourth cranial nerves of the brain, bundles of nerve fibers that control blinking and some eye movements.
In a previous case study, damage to these nerves appeared to block a chemical tracer from reaching brown fat. , Dr. Srikanthan said.
A possible mechanism for Ozempic?
Brown fat has already been linked to at least one breakthrough in obesity treatment. Some evidence suggests that popular medications like semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro) may affect brown fat activity. These belong to a class of drugs known as glucagon-like peptide-1 (GLP-1) receptor agonists. They work by mimicking the hormone GLP-1, which is released in the gut and brain in response to eating glucose (sugary foods or drinks).
“GLP-1 agonists have been shown to increase [brown fat] activity in rodents and humans, but likely indirectly, via activation of specific regions in the brain,” explained Varman Samuel, MD, PhD, an associate professor of medicine at the Yale School of Medicine, New Haven, Conn., and chief of endocrinology for the VA Connecticut Healthcare System.
The scientific literature is divided on this, but there is enough evidence to support further inquiry, Dr. Srikanthan said. Her team has begun a study to examine that link.
Opening the door to future obesity treatments
But their discovery means other new treatments could be on the horizon.
Previous research had shown that the sympathetic nervous system drives brown fat activity. But now that the UCLA scientists have revealed the exact nerves connecting brown fat to the sympathetic nervous system, we could find ways to stimulate those pathways to activate brown fat – without stimulating the many organs (such as the heart and stomach) also connected to this vast network of nerves, Dr. Srikanthan said.
Methods for doing that could include medication, electrical stimulation, or heat therapy, according to the study.
Still, there is reason to temper expectations. “[Brown fat] depots, while highly metabolically active, are quite small,” Dr. Samuel said. “So, the overall contribution to whole-body energy balance in humans will likely be small.”
On the other hand, that prediction doesn’t account for what we don’t know.
“We’re learning more about how tissues communicate with each other, beyond the release of hormones or metabolites,” Dr. Samuel said. Activating brown fat could trigger “signals that help coordinate whole-body energy metabolism.”
A version of this article first appeared on WebMD.com.
A process your body uses to stay warm in cool weather could one day lead to new therapies for obesity.
Scientists have, for the first time, mapped the precise nerve pathways that activate brown fat, or brown adipose tissue (BAT), a specialized fat that generates heat. Low temperatures kick brown fat into gear, helping the body keep its temperature and burning calories in the process.
“It has long been speculated that activating this type of fat may be useful in treating obesity and related metabolic conditions,” said Preethi Srikanthan, MD, an endocrinologist and professor of medicine who oversaw the research at the UCLA School of Medicine. “The challenge has been finding a way of selectively stimulating [it].”
Brown fat is different from the fat typically linked to obesity: the kind that accumulates around the belly, hips, and thighs. That’s white fat. White fat stores energy; brown fat burns it. That’s because brown fat cells have more mitochondria, a part of the cell that generates energy.
After dissecting the necks of eight human cadavers, Dr. Srikanthan and her team traced the sympathetic nerve branches in the fat pad above the collarbone – where the largest depot of brown fat in adults is stored. They stained the nerves, took samples, and viewed them under a microscope.
They found that nerves from brown fat traveled to the third and fourth cranial nerves of the brain, bundles of nerve fibers that control blinking and some eye movements.
In a previous case study, damage to these nerves appeared to block a chemical tracer from reaching brown fat. , Dr. Srikanthan said.
A possible mechanism for Ozempic?
Brown fat has already been linked to at least one breakthrough in obesity treatment. Some evidence suggests that popular medications like semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro) may affect brown fat activity. These belong to a class of drugs known as glucagon-like peptide-1 (GLP-1) receptor agonists. They work by mimicking the hormone GLP-1, which is released in the gut and brain in response to eating glucose (sugary foods or drinks).
“GLP-1 agonists have been shown to increase [brown fat] activity in rodents and humans, but likely indirectly, via activation of specific regions in the brain,” explained Varman Samuel, MD, PhD, an associate professor of medicine at the Yale School of Medicine, New Haven, Conn., and chief of endocrinology for the VA Connecticut Healthcare System.
The scientific literature is divided on this, but there is enough evidence to support further inquiry, Dr. Srikanthan said. Her team has begun a study to examine that link.
Opening the door to future obesity treatments
But their discovery means other new treatments could be on the horizon.
Previous research had shown that the sympathetic nervous system drives brown fat activity. But now that the UCLA scientists have revealed the exact nerves connecting brown fat to the sympathetic nervous system, we could find ways to stimulate those pathways to activate brown fat – without stimulating the many organs (such as the heart and stomach) also connected to this vast network of nerves, Dr. Srikanthan said.
Methods for doing that could include medication, electrical stimulation, or heat therapy, according to the study.
Still, there is reason to temper expectations. “[Brown fat] depots, while highly metabolically active, are quite small,” Dr. Samuel said. “So, the overall contribution to whole-body energy balance in humans will likely be small.”
On the other hand, that prediction doesn’t account for what we don’t know.
“We’re learning more about how tissues communicate with each other, beyond the release of hormones or metabolites,” Dr. Samuel said. Activating brown fat could trigger “signals that help coordinate whole-body energy metabolism.”
A version of this article first appeared on WebMD.com.
A process your body uses to stay warm in cool weather could one day lead to new therapies for obesity.
Scientists have, for the first time, mapped the precise nerve pathways that activate brown fat, or brown adipose tissue (BAT), a specialized fat that generates heat. Low temperatures kick brown fat into gear, helping the body keep its temperature and burning calories in the process.
“It has long been speculated that activating this type of fat may be useful in treating obesity and related metabolic conditions,” said Preethi Srikanthan, MD, an endocrinologist and professor of medicine who oversaw the research at the UCLA School of Medicine. “The challenge has been finding a way of selectively stimulating [it].”
Brown fat is different from the fat typically linked to obesity: the kind that accumulates around the belly, hips, and thighs. That’s white fat. White fat stores energy; brown fat burns it. That’s because brown fat cells have more mitochondria, a part of the cell that generates energy.
After dissecting the necks of eight human cadavers, Dr. Srikanthan and her team traced the sympathetic nerve branches in the fat pad above the collarbone – where the largest depot of brown fat in adults is stored. They stained the nerves, took samples, and viewed them under a microscope.
They found that nerves from brown fat traveled to the third and fourth cranial nerves of the brain, bundles of nerve fibers that control blinking and some eye movements.
In a previous case study, damage to these nerves appeared to block a chemical tracer from reaching brown fat. , Dr. Srikanthan said.
A possible mechanism for Ozempic?
Brown fat has already been linked to at least one breakthrough in obesity treatment. Some evidence suggests that popular medications like semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro) may affect brown fat activity. These belong to a class of drugs known as glucagon-like peptide-1 (GLP-1) receptor agonists. They work by mimicking the hormone GLP-1, which is released in the gut and brain in response to eating glucose (sugary foods or drinks).
“GLP-1 agonists have been shown to increase [brown fat] activity in rodents and humans, but likely indirectly, via activation of specific regions in the brain,” explained Varman Samuel, MD, PhD, an associate professor of medicine at the Yale School of Medicine, New Haven, Conn., and chief of endocrinology for the VA Connecticut Healthcare System.
The scientific literature is divided on this, but there is enough evidence to support further inquiry, Dr. Srikanthan said. Her team has begun a study to examine that link.
Opening the door to future obesity treatments
But their discovery means other new treatments could be on the horizon.
Previous research had shown that the sympathetic nervous system drives brown fat activity. But now that the UCLA scientists have revealed the exact nerves connecting brown fat to the sympathetic nervous system, we could find ways to stimulate those pathways to activate brown fat – without stimulating the many organs (such as the heart and stomach) also connected to this vast network of nerves, Dr. Srikanthan said.
Methods for doing that could include medication, electrical stimulation, or heat therapy, according to the study.
Still, there is reason to temper expectations. “[Brown fat] depots, while highly metabolically active, are quite small,” Dr. Samuel said. “So, the overall contribution to whole-body energy balance in humans will likely be small.”
On the other hand, that prediction doesn’t account for what we don’t know.
“We’re learning more about how tissues communicate with each other, beyond the release of hormones or metabolites,” Dr. Samuel said. Activating brown fat could trigger “signals that help coordinate whole-body energy metabolism.”
A version of this article first appeared on WebMD.com.
FROM PLOS ONE
Fractures beget fractures at any age
VANCOUVER – The occurrence of a fracture predicts future fracture risk, but the increase in risk is the same no matter what the age of the patient, according to a new population-based study drawn from the Manitoba BMD Registry.
The work expands previous studies that focused mostly on fracture risk prediction after a first fracture among individuals aged 45-50 and older. Other limitations of prior studies include large age categories (such as “premenopausal”), reliance on self-reporting, and small sample sizes.
As a result, some guidelines recommend considering fracture history only for patients older than a certain age when assessing for future risk, such as with the Fracture Risk Assessment Tool (FRAX). The new study suggests a potential need to reconsider that stance.
“The [percentage] of increased risk from having had prevalent fractures in the past, no matter what your age, is about the same. I think that it’s really paradigm shifting because [when] most of us think [of] young people who fracture, we’re not thinking of osteoporosis or future fracture risk. We’re not saying, ‘Oh, I had a fracture when I was 25. When I’m 70, I should be thinking about osteoporosis.’ So, I think this study is quite eye-opening that way,” Carrie Ye, MD, who presented the study at the annual meeting of the American Society for Bone and Mineral Research, said in an interview.
Participants of younger age who are referred for dual-energy x-ray absorptiometry (DXA) likely represent a population at increased risk of osteoporosis, according to Dr. Ye. “Maybe they have Crohn’s disease or maybe they’re on a bunch of steroids, and so a clinician has flagged them,” said Dr. Ye, who is an assistant professor and rheumatologist at the University of Alberta, Edmonton.
The researchers limited the analysis to nontraumatic fractures, but session moderator Nicholas Harvey, MD, PhD, wondered if a similar finding would occur with traumatic fractures. In an interview, he noted that researchers led by William Leslie, MD, at the University of Manitoba, Winnipeg, found that prior traumatic fracture also predicted future low bone-mineral density (BMD) and osteoporotic fracture. “I think that would have been one interesting question,” said Dr. Harvey, director of the Medical Research Council Lifecourse Epidemiology Centre at the University of Southampton, England.
Dr. Ye’s study included 88,696 individuals who underwent a first DXA scan between 1996 and 2018, which researchers then linked to provincial administrative health data collected between 1979 and 2018. The mean age at first DXA was 64.6 years, and 90.3% were women. Their mean body mass index was 27.4 kg/m2. Current smokers made up 10.1% of the cohort, 5.5% had a history of prolonged glucocorticoid use, 3.1% had rheumatoid arthritis, and among 14.9% of patients, there was a secondary cause of osteoporosis. Over a median 25.1 years of observation prior to DXA, clinical fracture occurred in 23.8% of participants.
The mean age of the patients at the time of their first prior fracture was 57.7 years. Over a mean 9.0 years of follow-up, 14.6% of participants experienced a fracture of any kind, 14.0% had osteoporotic fractures, 10.6% had a major osteoporotic fracture (nonankle), and 3.5% had a hip fracture. Among persons aged 20-29 years to 80 years or older, the adjusted hazard ratios for future fractures were similar, ranging from 1.51 to 2.12 (P for trend = .120).
The results were similar when age groups were analyzed with regard to all fractures, osteoporotic fractures, major osteoporotic fractures, or hip fractures.
Going forward, Dr. Ye hopes to expand the research into childhood fractures. “They can break their bones pretty easily, especially as they’re going through growth spurts and things like that,” she said.
Asked what her advice to physicians would be, Dr. Ye responded: “Don’t ignore prior fractures, even if they occurred at an early age. I think if someone’s had a fracture, they bought themselves a fracture risk assessment, and that doesn’t mean necessarily a DXA scan. It means you go through their other risk factors: What medications are they on? Do they have a family history? Are they super low BMI? Look at other reasons why you should be worried about their bones, and if you should be worried about their bones, certainly [measure their] BMD and see what’s going on.”
Dr. Ye and Dr. Harvey have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VANCOUVER – The occurrence of a fracture predicts future fracture risk, but the increase in risk is the same no matter what the age of the patient, according to a new population-based study drawn from the Manitoba BMD Registry.
The work expands previous studies that focused mostly on fracture risk prediction after a first fracture among individuals aged 45-50 and older. Other limitations of prior studies include large age categories (such as “premenopausal”), reliance on self-reporting, and small sample sizes.
As a result, some guidelines recommend considering fracture history only for patients older than a certain age when assessing for future risk, such as with the Fracture Risk Assessment Tool (FRAX). The new study suggests a potential need to reconsider that stance.
“The [percentage] of increased risk from having had prevalent fractures in the past, no matter what your age, is about the same. I think that it’s really paradigm shifting because [when] most of us think [of] young people who fracture, we’re not thinking of osteoporosis or future fracture risk. We’re not saying, ‘Oh, I had a fracture when I was 25. When I’m 70, I should be thinking about osteoporosis.’ So, I think this study is quite eye-opening that way,” Carrie Ye, MD, who presented the study at the annual meeting of the American Society for Bone and Mineral Research, said in an interview.
Participants of younger age who are referred for dual-energy x-ray absorptiometry (DXA) likely represent a population at increased risk of osteoporosis, according to Dr. Ye. “Maybe they have Crohn’s disease or maybe they’re on a bunch of steroids, and so a clinician has flagged them,” said Dr. Ye, who is an assistant professor and rheumatologist at the University of Alberta, Edmonton.
The researchers limited the analysis to nontraumatic fractures, but session moderator Nicholas Harvey, MD, PhD, wondered if a similar finding would occur with traumatic fractures. In an interview, he noted that researchers led by William Leslie, MD, at the University of Manitoba, Winnipeg, found that prior traumatic fracture also predicted future low bone-mineral density (BMD) and osteoporotic fracture. “I think that would have been one interesting question,” said Dr. Harvey, director of the Medical Research Council Lifecourse Epidemiology Centre at the University of Southampton, England.
Dr. Ye’s study included 88,696 individuals who underwent a first DXA scan between 1996 and 2018, which researchers then linked to provincial administrative health data collected between 1979 and 2018. The mean age at first DXA was 64.6 years, and 90.3% were women. Their mean body mass index was 27.4 kg/m2. Current smokers made up 10.1% of the cohort, 5.5% had a history of prolonged glucocorticoid use, 3.1% had rheumatoid arthritis, and among 14.9% of patients, there was a secondary cause of osteoporosis. Over a median 25.1 years of observation prior to DXA, clinical fracture occurred in 23.8% of participants.
The mean age of the patients at the time of their first prior fracture was 57.7 years. Over a mean 9.0 years of follow-up, 14.6% of participants experienced a fracture of any kind, 14.0% had osteoporotic fractures, 10.6% had a major osteoporotic fracture (nonankle), and 3.5% had a hip fracture. Among persons aged 20-29 years to 80 years or older, the adjusted hazard ratios for future fractures were similar, ranging from 1.51 to 2.12 (P for trend = .120).
The results were similar when age groups were analyzed with regard to all fractures, osteoporotic fractures, major osteoporotic fractures, or hip fractures.
Going forward, Dr. Ye hopes to expand the research into childhood fractures. “They can break their bones pretty easily, especially as they’re going through growth spurts and things like that,” she said.
Asked what her advice to physicians would be, Dr. Ye responded: “Don’t ignore prior fractures, even if they occurred at an early age. I think if someone’s had a fracture, they bought themselves a fracture risk assessment, and that doesn’t mean necessarily a DXA scan. It means you go through their other risk factors: What medications are they on? Do they have a family history? Are they super low BMI? Look at other reasons why you should be worried about their bones, and if you should be worried about their bones, certainly [measure their] BMD and see what’s going on.”
Dr. Ye and Dr. Harvey have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VANCOUVER – The occurrence of a fracture predicts future fracture risk, but the increase in risk is the same no matter what the age of the patient, according to a new population-based study drawn from the Manitoba BMD Registry.
The work expands previous studies that focused mostly on fracture risk prediction after a first fracture among individuals aged 45-50 and older. Other limitations of prior studies include large age categories (such as “premenopausal”), reliance on self-reporting, and small sample sizes.
As a result, some guidelines recommend considering fracture history only for patients older than a certain age when assessing for future risk, such as with the Fracture Risk Assessment Tool (FRAX). The new study suggests a potential need to reconsider that stance.
“The [percentage] of increased risk from having had prevalent fractures in the past, no matter what your age, is about the same. I think that it’s really paradigm shifting because [when] most of us think [of] young people who fracture, we’re not thinking of osteoporosis or future fracture risk. We’re not saying, ‘Oh, I had a fracture when I was 25. When I’m 70, I should be thinking about osteoporosis.’ So, I think this study is quite eye-opening that way,” Carrie Ye, MD, who presented the study at the annual meeting of the American Society for Bone and Mineral Research, said in an interview.
Participants of younger age who are referred for dual-energy x-ray absorptiometry (DXA) likely represent a population at increased risk of osteoporosis, according to Dr. Ye. “Maybe they have Crohn’s disease or maybe they’re on a bunch of steroids, and so a clinician has flagged them,” said Dr. Ye, who is an assistant professor and rheumatologist at the University of Alberta, Edmonton.
The researchers limited the analysis to nontraumatic fractures, but session moderator Nicholas Harvey, MD, PhD, wondered if a similar finding would occur with traumatic fractures. In an interview, he noted that researchers led by William Leslie, MD, at the University of Manitoba, Winnipeg, found that prior traumatic fracture also predicted future low bone-mineral density (BMD) and osteoporotic fracture. “I think that would have been one interesting question,” said Dr. Harvey, director of the Medical Research Council Lifecourse Epidemiology Centre at the University of Southampton, England.
Dr. Ye’s study included 88,696 individuals who underwent a first DXA scan between 1996 and 2018, which researchers then linked to provincial administrative health data collected between 1979 and 2018. The mean age at first DXA was 64.6 years, and 90.3% were women. Their mean body mass index was 27.4 kg/m2. Current smokers made up 10.1% of the cohort, 5.5% had a history of prolonged glucocorticoid use, 3.1% had rheumatoid arthritis, and among 14.9% of patients, there was a secondary cause of osteoporosis. Over a median 25.1 years of observation prior to DXA, clinical fracture occurred in 23.8% of participants.
The mean age of the patients at the time of their first prior fracture was 57.7 years. Over a mean 9.0 years of follow-up, 14.6% of participants experienced a fracture of any kind, 14.0% had osteoporotic fractures, 10.6% had a major osteoporotic fracture (nonankle), and 3.5% had a hip fracture. Among persons aged 20-29 years to 80 years or older, the adjusted hazard ratios for future fractures were similar, ranging from 1.51 to 2.12 (P for trend = .120).
The results were similar when age groups were analyzed with regard to all fractures, osteoporotic fractures, major osteoporotic fractures, or hip fractures.
Going forward, Dr. Ye hopes to expand the research into childhood fractures. “They can break their bones pretty easily, especially as they’re going through growth spurts and things like that,” she said.
Asked what her advice to physicians would be, Dr. Ye responded: “Don’t ignore prior fractures, even if they occurred at an early age. I think if someone’s had a fracture, they bought themselves a fracture risk assessment, and that doesn’t mean necessarily a DXA scan. It means you go through their other risk factors: What medications are they on? Do they have a family history? Are they super low BMI? Look at other reasons why you should be worried about their bones, and if you should be worried about their bones, certainly [measure their] BMD and see what’s going on.”
Dr. Ye and Dr. Harvey have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ASBMR 2023
Greater fracture risk reduction seen with denosumab vs. zoledronic acid in postmenopausal women
VANCOUVER –
A previous head-to-head comparison showed that denosumab increased bone mineral density at key skeletal sites compared with zoledronic acid, but only a single, small observational study has examined fracture risk, and it found no difference.
The new study, presented at the annual meeting of the American Society for Bone and Mineral Research, used a relatively new method of real-world comparative effectiveness analysis called negative control outcome (NCO) to analyze Medicare fee-for-service data.
NCO analysis takes extra pains to remove bias through data that might be linked to potential confounders but could not reasonably be attributed to a drug. For example, people who have greater contact with the health care system may be more likely to get one drug or another. The researchers used the frequency of receiving a flu or pneumonia vaccine as a proxy for this. If the two comparison groups had a significant difference in a proxy, it suggested a hidden bias and forced the researchers to abandon those groupings. Another example used car accidents as a proxy for cognitive impairment.
“If you find meaningful differences between the two groups, and you can say there’s no way a bone drug could account for these differences, then we shouldn’t do this analysis because these groups just aren’t comparable. They probably differ by that confounding factor we couldn’t measure,” said Jeffrey Curtis, MD, who presented the study. He is a professor of medicine in the division of clinical immunology and rheumatology at the University of Alabama at Birmingham.
The study strongly suggests superiority for denosumab. “There was a significant difference in multiple different groupings of fractures – beginning at year 2, extending to year 3 and even out to year 5 – that showed that there is a significant reduction in fracture risk if you get treated with denosumab [that was greater] than if you get treated with zoledronic acid,” Dr. Curtis said.
The researchers weighed 118 covariates and ultimately identified a population of 90,805 women taking denosumab and 37,328 taking zoledronic acid that was equally balanced in all patient characteristics. The mean age was about 75 years in the denosumab group and 74 in the zoledronic acid group.
The researchers found a 34% lower risk for hip fracture in the denosumab group by 5 years (relative risk, 0.66; 95% confidence interval, 0.43-0.90).
Similar patterns in fracture risk reduction were observed at 5 years for nonvertebral fracture (RR, 0.67; 95% CI, 0.52-0.82), nonhip nonvertebral fracture (RR, 0.69; 95% CI, 0.50-0.88), and major osteoporotic fracture (RR, 0.74; 95% CI, 0.59-0.89).
During the Q&A session after the talk, one audience member commented that the study was limited because the researchers only followed patients who received zoledronic acid for 60 days, which could have missed potential long-term benefits of the drug, especially since bisphosphonates have a lengthy skeletal retention time. Dr. Curtis acknowledged the point but said, “Usually, that’s not something we do, but these are different enough mechanisms of action that it may be warranted at least as a sensitivity analysis,” he said.
The study and its methodology were impressive, according to Yumie Rhee, MD, who comoderated the session where the study was presented. “I think they did a really good job by doing the negative control analysis. We’re not going to have a head-to-head clinical trial, so we don’t know the real fracture reduction differences [between denosumab and zoledronic acid]. [The NCO analysis] is more than the propensity matching score that we do usually,” said Dr. Rhee, who is a professor of endocrinology at Yonsei University College of Medicine in Seoul, South Korea.
In particular, the study showed a significantly greater reduction in hip fractures with denosumab. “Even in the RCTs, it was really hard to see the reduction in hip fracture, so I think this is showing much stronger data for denosumab. Especially in patients who have more [general fracture] risk and patients with higher hip fracture risk, I would go with denosumab,” Dr. Rhee said.
Her comoderator, Maria Zanchetta, MD, agreed. “It can have clinical implication, because we think denosumab is better than [zoledronic acid] for higher-risk patients, but we didn’t have the evidence. So at least we have a new [study] to look at, and I think it’s very important for our practice,” said Dr. Zanchetta, who is a professor of osteology at the Institute of Diagnostics and Metabolic Research, Universidad del Salvador, Buenos Aires.
The study was funded by Amgen, which markets denosumab. Dr. Curtis has consulted for Amgen. Dr. Rhee and Dr. Zanchetta report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VANCOUVER –
A previous head-to-head comparison showed that denosumab increased bone mineral density at key skeletal sites compared with zoledronic acid, but only a single, small observational study has examined fracture risk, and it found no difference.
The new study, presented at the annual meeting of the American Society for Bone and Mineral Research, used a relatively new method of real-world comparative effectiveness analysis called negative control outcome (NCO) to analyze Medicare fee-for-service data.
NCO analysis takes extra pains to remove bias through data that might be linked to potential confounders but could not reasonably be attributed to a drug. For example, people who have greater contact with the health care system may be more likely to get one drug or another. The researchers used the frequency of receiving a flu or pneumonia vaccine as a proxy for this. If the two comparison groups had a significant difference in a proxy, it suggested a hidden bias and forced the researchers to abandon those groupings. Another example used car accidents as a proxy for cognitive impairment.
“If you find meaningful differences between the two groups, and you can say there’s no way a bone drug could account for these differences, then we shouldn’t do this analysis because these groups just aren’t comparable. They probably differ by that confounding factor we couldn’t measure,” said Jeffrey Curtis, MD, who presented the study. He is a professor of medicine in the division of clinical immunology and rheumatology at the University of Alabama at Birmingham.
The study strongly suggests superiority for denosumab. “There was a significant difference in multiple different groupings of fractures – beginning at year 2, extending to year 3 and even out to year 5 – that showed that there is a significant reduction in fracture risk if you get treated with denosumab [that was greater] than if you get treated with zoledronic acid,” Dr. Curtis said.
The researchers weighed 118 covariates and ultimately identified a population of 90,805 women taking denosumab and 37,328 taking zoledronic acid that was equally balanced in all patient characteristics. The mean age was about 75 years in the denosumab group and 74 in the zoledronic acid group.
The researchers found a 34% lower risk for hip fracture in the denosumab group by 5 years (relative risk, 0.66; 95% confidence interval, 0.43-0.90).
Similar patterns in fracture risk reduction were observed at 5 years for nonvertebral fracture (RR, 0.67; 95% CI, 0.52-0.82), nonhip nonvertebral fracture (RR, 0.69; 95% CI, 0.50-0.88), and major osteoporotic fracture (RR, 0.74; 95% CI, 0.59-0.89).
During the Q&A session after the talk, one audience member commented that the study was limited because the researchers only followed patients who received zoledronic acid for 60 days, which could have missed potential long-term benefits of the drug, especially since bisphosphonates have a lengthy skeletal retention time. Dr. Curtis acknowledged the point but said, “Usually, that’s not something we do, but these are different enough mechanisms of action that it may be warranted at least as a sensitivity analysis,” he said.
The study and its methodology were impressive, according to Yumie Rhee, MD, who comoderated the session where the study was presented. “I think they did a really good job by doing the negative control analysis. We’re not going to have a head-to-head clinical trial, so we don’t know the real fracture reduction differences [between denosumab and zoledronic acid]. [The NCO analysis] is more than the propensity matching score that we do usually,” said Dr. Rhee, who is a professor of endocrinology at Yonsei University College of Medicine in Seoul, South Korea.
In particular, the study showed a significantly greater reduction in hip fractures with denosumab. “Even in the RCTs, it was really hard to see the reduction in hip fracture, so I think this is showing much stronger data for denosumab. Especially in patients who have more [general fracture] risk and patients with higher hip fracture risk, I would go with denosumab,” Dr. Rhee said.
Her comoderator, Maria Zanchetta, MD, agreed. “It can have clinical implication, because we think denosumab is better than [zoledronic acid] for higher-risk patients, but we didn’t have the evidence. So at least we have a new [study] to look at, and I think it’s very important for our practice,” said Dr. Zanchetta, who is a professor of osteology at the Institute of Diagnostics and Metabolic Research, Universidad del Salvador, Buenos Aires.
The study was funded by Amgen, which markets denosumab. Dr. Curtis has consulted for Amgen. Dr. Rhee and Dr. Zanchetta report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
VANCOUVER –
A previous head-to-head comparison showed that denosumab increased bone mineral density at key skeletal sites compared with zoledronic acid, but only a single, small observational study has examined fracture risk, and it found no difference.
The new study, presented at the annual meeting of the American Society for Bone and Mineral Research, used a relatively new method of real-world comparative effectiveness analysis called negative control outcome (NCO) to analyze Medicare fee-for-service data.
NCO analysis takes extra pains to remove bias through data that might be linked to potential confounders but could not reasonably be attributed to a drug. For example, people who have greater contact with the health care system may be more likely to get one drug or another. The researchers used the frequency of receiving a flu or pneumonia vaccine as a proxy for this. If the two comparison groups had a significant difference in a proxy, it suggested a hidden bias and forced the researchers to abandon those groupings. Another example used car accidents as a proxy for cognitive impairment.
“If you find meaningful differences between the two groups, and you can say there’s no way a bone drug could account for these differences, then we shouldn’t do this analysis because these groups just aren’t comparable. They probably differ by that confounding factor we couldn’t measure,” said Jeffrey Curtis, MD, who presented the study. He is a professor of medicine in the division of clinical immunology and rheumatology at the University of Alabama at Birmingham.
The study strongly suggests superiority for denosumab. “There was a significant difference in multiple different groupings of fractures – beginning at year 2, extending to year 3 and even out to year 5 – that showed that there is a significant reduction in fracture risk if you get treated with denosumab [that was greater] than if you get treated with zoledronic acid,” Dr. Curtis said.
The researchers weighed 118 covariates and ultimately identified a population of 90,805 women taking denosumab and 37,328 taking zoledronic acid that was equally balanced in all patient characteristics. The mean age was about 75 years in the denosumab group and 74 in the zoledronic acid group.
The researchers found a 34% lower risk for hip fracture in the denosumab group by 5 years (relative risk, 0.66; 95% confidence interval, 0.43-0.90).
Similar patterns in fracture risk reduction were observed at 5 years for nonvertebral fracture (RR, 0.67; 95% CI, 0.52-0.82), nonhip nonvertebral fracture (RR, 0.69; 95% CI, 0.50-0.88), and major osteoporotic fracture (RR, 0.74; 95% CI, 0.59-0.89).
During the Q&A session after the talk, one audience member commented that the study was limited because the researchers only followed patients who received zoledronic acid for 60 days, which could have missed potential long-term benefits of the drug, especially since bisphosphonates have a lengthy skeletal retention time. Dr. Curtis acknowledged the point but said, “Usually, that’s not something we do, but these are different enough mechanisms of action that it may be warranted at least as a sensitivity analysis,” he said.
The study and its methodology were impressive, according to Yumie Rhee, MD, who comoderated the session where the study was presented. “I think they did a really good job by doing the negative control analysis. We’re not going to have a head-to-head clinical trial, so we don’t know the real fracture reduction differences [between denosumab and zoledronic acid]. [The NCO analysis] is more than the propensity matching score that we do usually,” said Dr. Rhee, who is a professor of endocrinology at Yonsei University College of Medicine in Seoul, South Korea.
In particular, the study showed a significantly greater reduction in hip fractures with denosumab. “Even in the RCTs, it was really hard to see the reduction in hip fracture, so I think this is showing much stronger data for denosumab. Especially in patients who have more [general fracture] risk and patients with higher hip fracture risk, I would go with denosumab,” Dr. Rhee said.
Her comoderator, Maria Zanchetta, MD, agreed. “It can have clinical implication, because we think denosumab is better than [zoledronic acid] for higher-risk patients, but we didn’t have the evidence. So at least we have a new [study] to look at, and I think it’s very important for our practice,” said Dr. Zanchetta, who is a professor of osteology at the Institute of Diagnostics and Metabolic Research, Universidad del Salvador, Buenos Aires.
The study was funded by Amgen, which markets denosumab. Dr. Curtis has consulted for Amgen. Dr. Rhee and Dr. Zanchetta report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ASBMR 2023
Metabolic effects of estetrol are promising in postmenopausal women
PHILADELPHIA – presented at the annual meeting of the Menopause Society (formerly The North American Menopause Society).
Participants taking estetrol experienced a decrease in hemoglobin A1c, fasting plasma glucose, total cholesterol, LDL and lipoprotein as well as an increase in HDL cholesterol, according to the findings presented by Wolf Utian, MD, PhD, DSC, a professor emeritus of reproductive biology at Case Western Reserve University, Cleveland, and medical director emeritus of the Menopause Society.
A separate poster at the conference from the same trial also reported significant improvements from estetrol in quality of life, including that related to vasomotor symptoms, and several psychosocial and sexual functioning areas.
E4 is already available as combination oral contraception and is now being considered for treating vasomotor symptoms, explained Chrisandra Shufelt, MD, professor and chair of general internal of medicine and associate director of the Women’s Health Research Center at Mayo Clinic Florida, who was not involved in the study.
Background on estetrol
E4 is a human fetal liver estrogen produced during pregnancy that’s synthesized from plants for pharmaceutical use, including as the oral contraceptive drospirenone, Dr. Utian told attendees. It’s classified as a native estrogen with selective tissue activity (NEST), he said.
“E4 is a completely different native estrogen with oral administration mimicking the benefits of transdermals and hence safe and effective,” Dr. Utian said in an interview. “It would be a significant new addition to the pharmaceutical armamentarium.”
Two phase 3 trials presented by Dr. Utian at the same conference last year found estetrol reduced the frequency and severity of moderate to severe vasomotor symptoms, and a previous phase 2 trial finding vasomotor and genitourinary symptom benefits suggested it had potential benefits for lipids, carbohydrate metabolism, and bone turnover.
“In summary, E4 at a daily dose of 15 mg exhibited estrogenic effects in the vagina, leading to improved vaginal health and reduced signs of atrophy, emerging as a promising treatment option not only for vasomotor symptoms but also for other significant menopausal symptoms,” Dr. Utian said. “E4 could offer comprehensive relief for women experiencing a range of menopause-related discomforts.”
Dr. Utian also referenced a 2017 trial in which estetrol positively impacted lipid profiles, “lowering low-density lipoprotein cholesterol, increasing high-density lipoprotein cholesterol, and showing minimal influence on triglycerides,” he said. “Importantly, estetrol was associated with a significant decrease in osteocalcin levels in the higher dose groups, suggesting a potential preventive effect on bone loss,” he added. A recent review of the overall evidence on estetrol suggests its use is “promising,” Dr. Utian noted.
Current trial
His current randomized controlled phase 3 trial included postmenopausal women ages 40-65 from 151 sites in 14 countries in Europe, Latin America, and North America, and Russia. Among the 640 participants in the trial, 213 women randomly received 15 mg of estetrol, 213 women received 20 mg of estetrol, and 214 women received a placebo every day for 3 months. All women without hysterectomies also received 200 mg of progesterone once daily for two weeks after completing the estetrol treatment to protect the endometrium.
Researchers took blood samples from the participants at baseline and week 12 to assess total cholesterol, LDL, HDL, the total cholesterol/HDL ratio, triglycerides, lipoprotein A, fasting plasma glucose, insulin, and A1c.
Compared with women in the placebo group, women in both the 15 mg and 20 mg groups saw a statistically significant decrease in lipoprotein A and in the ratio of total cholesterol to HDL, and a statistically significant increase in HDL. Only the women in the 15 mg group saw a statistically significant decrease in LDL and increase in triglycerides; an increase in triglycerides in the 20 mg group did not reach statistical significance.
Statistically significant decreases in fasting plasma glucose and A1c also occurred in both treatment groups, but a decrease in insulin levels and in the homeostasis model-assessment-estimated insulin resistance (HOMA-IR) seen in both treatment arms did not reach significance.
“While the mean changes after 12 weeks from baseline overall were small changes to the cholesterol and blood sugar profiles, they are clinically meaningful because it suggests that E4 does not have any adverse effects to these measures,” Dr. Shufelt said in an interview. “An advantage is that this gives us another hormone option for vasomotor symptoms since it is a native estrogen with selective tissue.”
It’s too early, however, to determine whether estetrol offers benefits in terms of its safety profile, compared with currently available therapies, Dr. Shufelt said.
”These findings of E4 are similar to how oral estradiol changes lipids, which finds an increase in high-density lipoprotein cholesterol, and decreases plasma concentrations of total and low-density lipoprotein cholesterol. an increase in HDL-C and triglycerides and decrease in LDL-C,” she said.
Poster findings also promising
For the findings reported in the poster, researchers assessed quality of life and the clinical meaningfulness of vasomotor symptoms’ reduction at baseline and 12 weeks using the Menopause-Specific Quality of Life (MENQOL) questionnaire and the Clinical Global Impression questionnaire, respectively. They also assessed women’s self-reported genitourinary symptoms, including vaginal dryness, pain during urination, vaginal pain and bleeding related to sex, and vaginal or vulvar irritation or itching. Most of these findings primarily confirmed previous positive effects from E4 in other trials.
Women in both the 15 mg and 20 mg estetrol groups reported a statistically significant improvement at 12 weeks, compared with placebo, in their total MENQOL score and in the vasomotor, psychosocial, and sexual functioning domain scores (P < .05). Those in the 20 mg group also had a statistically significant improvement in their physical domain score (P < .05).
Although numerical improvements in genitourinary symptoms occurred at 12 weeks across all three groups, the only statistically significant difference from baseline occurred in patients taking 15 mg of estetrol, who experienced a decrease in vaginal dryness and vaginal pain during sex (P = .0142 and P = .003, respectively).
The Clinical Global Impression questionnaire asked women at 4 and 12 weeks to rate on a seven-item Likert scale their response to this question: “Rate the total improvement, whether or not in your judgment it is due entirely to drug treatment. Compared to your condition at admission to the study, how much has it changed?” Responses of “very much improved” and “much improved” counted as a clinically meaningful difference.
Compared with 27.9% of patients in the placebo group, 52.9% of patients in the 15 mg group and 59.8% of patients in the 20 mg group rated the weekly frequency of moderate to severe vasomotor symptoms as “much improved” or “very much improved” at 4 weeks (P < .0001). At 12 weeks, those numbers rose to 47% in the placebo group, 73.3% in the 15 mg group and 77.8% in the 20 mg group (P < .0001).
The trial’s primary limitation at this point is having only a 12-week follow-up, Dr. Shufelt said, though a few other questions remain.
“Because the two phase 3 RCTs included hysterectomized and nonhysterectomized women, it was unclear how many women in the study had E4 alone versus E4 with progesterone, as that might play a role in both cholesterol and carbohydrate metabolism,” Dr. Shufelt said. “While baseline data was not presented, it would also be important to know baseline values for the women and confirm that none were on lipid-lowering medications.”
The research was funded by Estetra SRL, an affiliate of Mithra Pharmaceuticals. Dr. Utian is a member of the Mithra and Elektra Scientific Advisory Boards. Dr. Shufelt has no disclosures.
PHILADELPHIA – presented at the annual meeting of the Menopause Society (formerly The North American Menopause Society).
Participants taking estetrol experienced a decrease in hemoglobin A1c, fasting plasma glucose, total cholesterol, LDL and lipoprotein as well as an increase in HDL cholesterol, according to the findings presented by Wolf Utian, MD, PhD, DSC, a professor emeritus of reproductive biology at Case Western Reserve University, Cleveland, and medical director emeritus of the Menopause Society.
A separate poster at the conference from the same trial also reported significant improvements from estetrol in quality of life, including that related to vasomotor symptoms, and several psychosocial and sexual functioning areas.
E4 is already available as combination oral contraception and is now being considered for treating vasomotor symptoms, explained Chrisandra Shufelt, MD, professor and chair of general internal of medicine and associate director of the Women’s Health Research Center at Mayo Clinic Florida, who was not involved in the study.
Background on estetrol
E4 is a human fetal liver estrogen produced during pregnancy that’s synthesized from plants for pharmaceutical use, including as the oral contraceptive drospirenone, Dr. Utian told attendees. It’s classified as a native estrogen with selective tissue activity (NEST), he said.
“E4 is a completely different native estrogen with oral administration mimicking the benefits of transdermals and hence safe and effective,” Dr. Utian said in an interview. “It would be a significant new addition to the pharmaceutical armamentarium.”
Two phase 3 trials presented by Dr. Utian at the same conference last year found estetrol reduced the frequency and severity of moderate to severe vasomotor symptoms, and a previous phase 2 trial finding vasomotor and genitourinary symptom benefits suggested it had potential benefits for lipids, carbohydrate metabolism, and bone turnover.
“In summary, E4 at a daily dose of 15 mg exhibited estrogenic effects in the vagina, leading to improved vaginal health and reduced signs of atrophy, emerging as a promising treatment option not only for vasomotor symptoms but also for other significant menopausal symptoms,” Dr. Utian said. “E4 could offer comprehensive relief for women experiencing a range of menopause-related discomforts.”
Dr. Utian also referenced a 2017 trial in which estetrol positively impacted lipid profiles, “lowering low-density lipoprotein cholesterol, increasing high-density lipoprotein cholesterol, and showing minimal influence on triglycerides,” he said. “Importantly, estetrol was associated with a significant decrease in osteocalcin levels in the higher dose groups, suggesting a potential preventive effect on bone loss,” he added. A recent review of the overall evidence on estetrol suggests its use is “promising,” Dr. Utian noted.
Current trial
His current randomized controlled phase 3 trial included postmenopausal women ages 40-65 from 151 sites in 14 countries in Europe, Latin America, and North America, and Russia. Among the 640 participants in the trial, 213 women randomly received 15 mg of estetrol, 213 women received 20 mg of estetrol, and 214 women received a placebo every day for 3 months. All women without hysterectomies also received 200 mg of progesterone once daily for two weeks after completing the estetrol treatment to protect the endometrium.
Researchers took blood samples from the participants at baseline and week 12 to assess total cholesterol, LDL, HDL, the total cholesterol/HDL ratio, triglycerides, lipoprotein A, fasting plasma glucose, insulin, and A1c.
Compared with women in the placebo group, women in both the 15 mg and 20 mg groups saw a statistically significant decrease in lipoprotein A and in the ratio of total cholesterol to HDL, and a statistically significant increase in HDL. Only the women in the 15 mg group saw a statistically significant decrease in LDL and increase in triglycerides; an increase in triglycerides in the 20 mg group did not reach statistical significance.
Statistically significant decreases in fasting plasma glucose and A1c also occurred in both treatment groups, but a decrease in insulin levels and in the homeostasis model-assessment-estimated insulin resistance (HOMA-IR) seen in both treatment arms did not reach significance.
“While the mean changes after 12 weeks from baseline overall were small changes to the cholesterol and blood sugar profiles, they are clinically meaningful because it suggests that E4 does not have any adverse effects to these measures,” Dr. Shufelt said in an interview. “An advantage is that this gives us another hormone option for vasomotor symptoms since it is a native estrogen with selective tissue.”
It’s too early, however, to determine whether estetrol offers benefits in terms of its safety profile, compared with currently available therapies, Dr. Shufelt said.
”These findings of E4 are similar to how oral estradiol changes lipids, which finds an increase in high-density lipoprotein cholesterol, and decreases plasma concentrations of total and low-density lipoprotein cholesterol. an increase in HDL-C and triglycerides and decrease in LDL-C,” she said.
Poster findings also promising
For the findings reported in the poster, researchers assessed quality of life and the clinical meaningfulness of vasomotor symptoms’ reduction at baseline and 12 weeks using the Menopause-Specific Quality of Life (MENQOL) questionnaire and the Clinical Global Impression questionnaire, respectively. They also assessed women’s self-reported genitourinary symptoms, including vaginal dryness, pain during urination, vaginal pain and bleeding related to sex, and vaginal or vulvar irritation or itching. Most of these findings primarily confirmed previous positive effects from E4 in other trials.
Women in both the 15 mg and 20 mg estetrol groups reported a statistically significant improvement at 12 weeks, compared with placebo, in their total MENQOL score and in the vasomotor, psychosocial, and sexual functioning domain scores (P < .05). Those in the 20 mg group also had a statistically significant improvement in their physical domain score (P < .05).
Although numerical improvements in genitourinary symptoms occurred at 12 weeks across all three groups, the only statistically significant difference from baseline occurred in patients taking 15 mg of estetrol, who experienced a decrease in vaginal dryness and vaginal pain during sex (P = .0142 and P = .003, respectively).
The Clinical Global Impression questionnaire asked women at 4 and 12 weeks to rate on a seven-item Likert scale their response to this question: “Rate the total improvement, whether or not in your judgment it is due entirely to drug treatment. Compared to your condition at admission to the study, how much has it changed?” Responses of “very much improved” and “much improved” counted as a clinically meaningful difference.
Compared with 27.9% of patients in the placebo group, 52.9% of patients in the 15 mg group and 59.8% of patients in the 20 mg group rated the weekly frequency of moderate to severe vasomotor symptoms as “much improved” or “very much improved” at 4 weeks (P < .0001). At 12 weeks, those numbers rose to 47% in the placebo group, 73.3% in the 15 mg group and 77.8% in the 20 mg group (P < .0001).
The trial’s primary limitation at this point is having only a 12-week follow-up, Dr. Shufelt said, though a few other questions remain.
“Because the two phase 3 RCTs included hysterectomized and nonhysterectomized women, it was unclear how many women in the study had E4 alone versus E4 with progesterone, as that might play a role in both cholesterol and carbohydrate metabolism,” Dr. Shufelt said. “While baseline data was not presented, it would also be important to know baseline values for the women and confirm that none were on lipid-lowering medications.”
The research was funded by Estetra SRL, an affiliate of Mithra Pharmaceuticals. Dr. Utian is a member of the Mithra and Elektra Scientific Advisory Boards. Dr. Shufelt has no disclosures.
PHILADELPHIA – presented at the annual meeting of the Menopause Society (formerly The North American Menopause Society).
Participants taking estetrol experienced a decrease in hemoglobin A1c, fasting plasma glucose, total cholesterol, LDL and lipoprotein as well as an increase in HDL cholesterol, according to the findings presented by Wolf Utian, MD, PhD, DSC, a professor emeritus of reproductive biology at Case Western Reserve University, Cleveland, and medical director emeritus of the Menopause Society.
A separate poster at the conference from the same trial also reported significant improvements from estetrol in quality of life, including that related to vasomotor symptoms, and several psychosocial and sexual functioning areas.
E4 is already available as combination oral contraception and is now being considered for treating vasomotor symptoms, explained Chrisandra Shufelt, MD, professor and chair of general internal of medicine and associate director of the Women’s Health Research Center at Mayo Clinic Florida, who was not involved in the study.
Background on estetrol
E4 is a human fetal liver estrogen produced during pregnancy that’s synthesized from plants for pharmaceutical use, including as the oral contraceptive drospirenone, Dr. Utian told attendees. It’s classified as a native estrogen with selective tissue activity (NEST), he said.
“E4 is a completely different native estrogen with oral administration mimicking the benefits of transdermals and hence safe and effective,” Dr. Utian said in an interview. “It would be a significant new addition to the pharmaceutical armamentarium.”
Two phase 3 trials presented by Dr. Utian at the same conference last year found estetrol reduced the frequency and severity of moderate to severe vasomotor symptoms, and a previous phase 2 trial finding vasomotor and genitourinary symptom benefits suggested it had potential benefits for lipids, carbohydrate metabolism, and bone turnover.
“In summary, E4 at a daily dose of 15 mg exhibited estrogenic effects in the vagina, leading to improved vaginal health and reduced signs of atrophy, emerging as a promising treatment option not only for vasomotor symptoms but also for other significant menopausal symptoms,” Dr. Utian said. “E4 could offer comprehensive relief for women experiencing a range of menopause-related discomforts.”
Dr. Utian also referenced a 2017 trial in which estetrol positively impacted lipid profiles, “lowering low-density lipoprotein cholesterol, increasing high-density lipoprotein cholesterol, and showing minimal influence on triglycerides,” he said. “Importantly, estetrol was associated with a significant decrease in osteocalcin levels in the higher dose groups, suggesting a potential preventive effect on bone loss,” he added. A recent review of the overall evidence on estetrol suggests its use is “promising,” Dr. Utian noted.
Current trial
His current randomized controlled phase 3 trial included postmenopausal women ages 40-65 from 151 sites in 14 countries in Europe, Latin America, and North America, and Russia. Among the 640 participants in the trial, 213 women randomly received 15 mg of estetrol, 213 women received 20 mg of estetrol, and 214 women received a placebo every day for 3 months. All women without hysterectomies also received 200 mg of progesterone once daily for two weeks after completing the estetrol treatment to protect the endometrium.
Researchers took blood samples from the participants at baseline and week 12 to assess total cholesterol, LDL, HDL, the total cholesterol/HDL ratio, triglycerides, lipoprotein A, fasting plasma glucose, insulin, and A1c.
Compared with women in the placebo group, women in both the 15 mg and 20 mg groups saw a statistically significant decrease in lipoprotein A and in the ratio of total cholesterol to HDL, and a statistically significant increase in HDL. Only the women in the 15 mg group saw a statistically significant decrease in LDL and increase in triglycerides; an increase in triglycerides in the 20 mg group did not reach statistical significance.
Statistically significant decreases in fasting plasma glucose and A1c also occurred in both treatment groups, but a decrease in insulin levels and in the homeostasis model-assessment-estimated insulin resistance (HOMA-IR) seen in both treatment arms did not reach significance.
“While the mean changes after 12 weeks from baseline overall were small changes to the cholesterol and blood sugar profiles, they are clinically meaningful because it suggests that E4 does not have any adverse effects to these measures,” Dr. Shufelt said in an interview. “An advantage is that this gives us another hormone option for vasomotor symptoms since it is a native estrogen with selective tissue.”
It’s too early, however, to determine whether estetrol offers benefits in terms of its safety profile, compared with currently available therapies, Dr. Shufelt said.
”These findings of E4 are similar to how oral estradiol changes lipids, which finds an increase in high-density lipoprotein cholesterol, and decreases plasma concentrations of total and low-density lipoprotein cholesterol. an increase in HDL-C and triglycerides and decrease in LDL-C,” she said.
Poster findings also promising
For the findings reported in the poster, researchers assessed quality of life and the clinical meaningfulness of vasomotor symptoms’ reduction at baseline and 12 weeks using the Menopause-Specific Quality of Life (MENQOL) questionnaire and the Clinical Global Impression questionnaire, respectively. They also assessed women’s self-reported genitourinary symptoms, including vaginal dryness, pain during urination, vaginal pain and bleeding related to sex, and vaginal or vulvar irritation or itching. Most of these findings primarily confirmed previous positive effects from E4 in other trials.
Women in both the 15 mg and 20 mg estetrol groups reported a statistically significant improvement at 12 weeks, compared with placebo, in their total MENQOL score and in the vasomotor, psychosocial, and sexual functioning domain scores (P < .05). Those in the 20 mg group also had a statistically significant improvement in their physical domain score (P < .05).
Although numerical improvements in genitourinary symptoms occurred at 12 weeks across all three groups, the only statistically significant difference from baseline occurred in patients taking 15 mg of estetrol, who experienced a decrease in vaginal dryness and vaginal pain during sex (P = .0142 and P = .003, respectively).
The Clinical Global Impression questionnaire asked women at 4 and 12 weeks to rate on a seven-item Likert scale their response to this question: “Rate the total improvement, whether or not in your judgment it is due entirely to drug treatment. Compared to your condition at admission to the study, how much has it changed?” Responses of “very much improved” and “much improved” counted as a clinically meaningful difference.
Compared with 27.9% of patients in the placebo group, 52.9% of patients in the 15 mg group and 59.8% of patients in the 20 mg group rated the weekly frequency of moderate to severe vasomotor symptoms as “much improved” or “very much improved” at 4 weeks (P < .0001). At 12 weeks, those numbers rose to 47% in the placebo group, 73.3% in the 15 mg group and 77.8% in the 20 mg group (P < .0001).
The trial’s primary limitation at this point is having only a 12-week follow-up, Dr. Shufelt said, though a few other questions remain.
“Because the two phase 3 RCTs included hysterectomized and nonhysterectomized women, it was unclear how many women in the study had E4 alone versus E4 with progesterone, as that might play a role in both cholesterol and carbohydrate metabolism,” Dr. Shufelt said. “While baseline data was not presented, it would also be important to know baseline values for the women and confirm that none were on lipid-lowering medications.”
The research was funded by Estetra SRL, an affiliate of Mithra Pharmaceuticals. Dr. Utian is a member of the Mithra and Elektra Scientific Advisory Boards. Dr. Shufelt has no disclosures.
AT NAMS 2023
Bone degradation measure can sway osteoporosis diagnosis
Assessing a key aspect of bone architecture, for which clinicians can now be reimbursed under Medicare, can significantly improve the ability to predict a patient’s risk for bone fracture.
Although bone mineral density (BMD) is traditionally used to identify patients with osteoporosis or low bone mass, some physicians have begun incorporating the trabecular bone score (TBS) into their exams.
At the Cleveland Clinic Center for Specialized Women’s Health, factoring in the TBS changed the diagnosis for 16% of 432 patients, according to Holly Thacker, MD, the center’s director.
“Importantly, 11% got worse diagnoses, and I use that in terms of prioritizing treatment,” Dr. Thacker said in an interview. The ability to determine how degraded the bone microarchitecture is through a software system “is a huge advance.”
Dr. Thacker described her center’s experience with the technology at the annual meeting of the Menopause Society (formerly The North American Menopause Society).
While BMD captures the amount of minerals like calcium in the skeleton, TBS assesses the underlying microarchitecture by looking at the distribution of shades of gray on dual-energy x-ray absorptiometry (DXA) scans.
Based on the TBS, patients’ bones are classified as normal, partially degraded, or degraded. Among the 432 patients who received a TBS analysis in 2022, 3% shifted from a normal diagnosis to osteopenia, 8% worsened from osteopenia to osteoporosis, 4% went from osteopenia to normal, and 1.6% downgraded from osteoporosis to osteopenia, Dr. Thacker reported.
The new test may also provide some reassurance for female patients who have thinner bones, which may raise alarms based on BMD. TBS, however, may show that the structure of the bone looks normal.
“When you know that the microarchitecture is normal, you’re a lot less concerned that they actually have a bone disease of osteoporosis,” Dr. Thacker said.
Conversely, unexpectedly degraded bone raises questions.
“That makes you go back and say [to the patient]: ‘Have you been on steroids? Were you malnourished? Is there some other metabolic problem? Have you had some calcium disorder?’ ” Dr. Thacker said.
Dr. Thacker leverages the TBS to help patients obtain effective therapy, typically an anabolic agent followed by antiresorptive medication.
“When I see a patient who not only has osteoporosis on bone density but has completely degraded bone architecture, it’s a lot easier for me to make the argument to the insurance company that this patient is at grave risk for a low trauma fracture and bad outcome without the best treatment,” Dr. Thacker said.
10-year-old tech, recently covered
The Food and Drug Administration approved TBS software in 2012, but Medicare only recently started paying for it.
Medimaps Group, a company that markets imaging software to calculate TBS, announced in 2022 that reimbursement from the Centers for Medicare & Medicaid Services was available, at $41.53 on the Physician Fee Schedule and $82.61 on the Hospital Outpatient Prospective Payment Schedule.
“Reimbursement through CMS is an important endorsement of the clinical value of TBS for clinicians and their patients,” Didier Hans, PhD, MBA, the CEO of Medimaps, said in a statement at the time. He noted that more than 600,000 TBS procedures were being performed in the United States each year.
Nevertheless, the initial investment in purchasing the software may be a barrier for health systems.
“We are the first and only site in our health system to offer TBS, as this is an extra expense and not uniformly reimbursed by insurers,” Dr. Thacker reported at the meeting.
Potential drawbacks
The TBS software used in Dr. Thacker’s study has been validated only in Asian and White patients between certain ages and weights, meaning the system is not designed to be used for other populations. Other researchers have highlighted a need for trabecular bone scoring to be validated more broadly. The authors of a recent analysis, however, suggest that TBS can be used the same way no matter a patient’s race.
TBS “is going to be most helpful in those with osteopenia who are right near the threshold for treatment,” said Marcella Donovan Walker, MD, MS, in a presentation on bone quality at the meeting.
Many studies have shown that TBS “provides additive information to bone density,” said Dr. Walker, a professor of medicine in the division of endocrinology at Columbia University, New York. For example, a large study of women in Manitoba found that, regardless of whether their bone density was normal, osteopenic, or osteoporotic, those with a low TBS had a much higher risk for fracture.
‘Opportunistic screening’ with CT?
TBS relies on the same DXA scans that are used to calculate bone mineral density, so obtaining the score does not add time or radiation to the scanning process, Dr. Thacker said.
But many patients who should receive DXA scans do not, which adds to the promise of “opportunistic screening” for osteoporosis, Dr. Walker said. With this approach, physicians would analyze a CT scan that a patient received for another purpose, such as to investigate abdominal pain or chest pain.
“In these images is information about the bone,” Dr. Walker said.
Researchers have used high-resolution peripheral quantitative CT to perform finite element analysis, where a computer program simulates compression of the bone to create a measure of bone stiffness and determine the load required for a break.
One study found that including those elements predicted fractures better than bone mineral density or the Fracture Risk Assessment Tool alone, Dr. Walker noted.
Other aspects of bone quality include how many cracks are in the bone, the amount of adipose in the marrow space, and the rate at which bone is broken down and rebuilt. But Dr. Walker suggested that the longstanding focus on bone mineral density in clinical practice makes sense.
“By far, bone mass is the most important bone quality,” Dr. Walker said.
Dr. Thacker is the executive director of the nonprofit Speaking of Women’s Health. Dr. Walker reported receiving funding from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and Amgen.
A version of this article first appeared on Medscape.com.
Assessing a key aspect of bone architecture, for which clinicians can now be reimbursed under Medicare, can significantly improve the ability to predict a patient’s risk for bone fracture.
Although bone mineral density (BMD) is traditionally used to identify patients with osteoporosis or low bone mass, some physicians have begun incorporating the trabecular bone score (TBS) into their exams.
At the Cleveland Clinic Center for Specialized Women’s Health, factoring in the TBS changed the diagnosis for 16% of 432 patients, according to Holly Thacker, MD, the center’s director.
“Importantly, 11% got worse diagnoses, and I use that in terms of prioritizing treatment,” Dr. Thacker said in an interview. The ability to determine how degraded the bone microarchitecture is through a software system “is a huge advance.”
Dr. Thacker described her center’s experience with the technology at the annual meeting of the Menopause Society (formerly The North American Menopause Society).
While BMD captures the amount of minerals like calcium in the skeleton, TBS assesses the underlying microarchitecture by looking at the distribution of shades of gray on dual-energy x-ray absorptiometry (DXA) scans.
Based on the TBS, patients’ bones are classified as normal, partially degraded, or degraded. Among the 432 patients who received a TBS analysis in 2022, 3% shifted from a normal diagnosis to osteopenia, 8% worsened from osteopenia to osteoporosis, 4% went from osteopenia to normal, and 1.6% downgraded from osteoporosis to osteopenia, Dr. Thacker reported.
The new test may also provide some reassurance for female patients who have thinner bones, which may raise alarms based on BMD. TBS, however, may show that the structure of the bone looks normal.
“When you know that the microarchitecture is normal, you’re a lot less concerned that they actually have a bone disease of osteoporosis,” Dr. Thacker said.
Conversely, unexpectedly degraded bone raises questions.
“That makes you go back and say [to the patient]: ‘Have you been on steroids? Were you malnourished? Is there some other metabolic problem? Have you had some calcium disorder?’ ” Dr. Thacker said.
Dr. Thacker leverages the TBS to help patients obtain effective therapy, typically an anabolic agent followed by antiresorptive medication.
“When I see a patient who not only has osteoporosis on bone density but has completely degraded bone architecture, it’s a lot easier for me to make the argument to the insurance company that this patient is at grave risk for a low trauma fracture and bad outcome without the best treatment,” Dr. Thacker said.
10-year-old tech, recently covered
The Food and Drug Administration approved TBS software in 2012, but Medicare only recently started paying for it.
Medimaps Group, a company that markets imaging software to calculate TBS, announced in 2022 that reimbursement from the Centers for Medicare & Medicaid Services was available, at $41.53 on the Physician Fee Schedule and $82.61 on the Hospital Outpatient Prospective Payment Schedule.
“Reimbursement through CMS is an important endorsement of the clinical value of TBS for clinicians and their patients,” Didier Hans, PhD, MBA, the CEO of Medimaps, said in a statement at the time. He noted that more than 600,000 TBS procedures were being performed in the United States each year.
Nevertheless, the initial investment in purchasing the software may be a barrier for health systems.
“We are the first and only site in our health system to offer TBS, as this is an extra expense and not uniformly reimbursed by insurers,” Dr. Thacker reported at the meeting.
Potential drawbacks
The TBS software used in Dr. Thacker’s study has been validated only in Asian and White patients between certain ages and weights, meaning the system is not designed to be used for other populations. Other researchers have highlighted a need for trabecular bone scoring to be validated more broadly. The authors of a recent analysis, however, suggest that TBS can be used the same way no matter a patient’s race.
TBS “is going to be most helpful in those with osteopenia who are right near the threshold for treatment,” said Marcella Donovan Walker, MD, MS, in a presentation on bone quality at the meeting.
Many studies have shown that TBS “provides additive information to bone density,” said Dr. Walker, a professor of medicine in the division of endocrinology at Columbia University, New York. For example, a large study of women in Manitoba found that, regardless of whether their bone density was normal, osteopenic, or osteoporotic, those with a low TBS had a much higher risk for fracture.
‘Opportunistic screening’ with CT?
TBS relies on the same DXA scans that are used to calculate bone mineral density, so obtaining the score does not add time or radiation to the scanning process, Dr. Thacker said.
But many patients who should receive DXA scans do not, which adds to the promise of “opportunistic screening” for osteoporosis, Dr. Walker said. With this approach, physicians would analyze a CT scan that a patient received for another purpose, such as to investigate abdominal pain or chest pain.
“In these images is information about the bone,” Dr. Walker said.
Researchers have used high-resolution peripheral quantitative CT to perform finite element analysis, where a computer program simulates compression of the bone to create a measure of bone stiffness and determine the load required for a break.
One study found that including those elements predicted fractures better than bone mineral density or the Fracture Risk Assessment Tool alone, Dr. Walker noted.
Other aspects of bone quality include how many cracks are in the bone, the amount of adipose in the marrow space, and the rate at which bone is broken down and rebuilt. But Dr. Walker suggested that the longstanding focus on bone mineral density in clinical practice makes sense.
“By far, bone mass is the most important bone quality,” Dr. Walker said.
Dr. Thacker is the executive director of the nonprofit Speaking of Women’s Health. Dr. Walker reported receiving funding from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and Amgen.
A version of this article first appeared on Medscape.com.
Assessing a key aspect of bone architecture, for which clinicians can now be reimbursed under Medicare, can significantly improve the ability to predict a patient’s risk for bone fracture.
Although bone mineral density (BMD) is traditionally used to identify patients with osteoporosis or low bone mass, some physicians have begun incorporating the trabecular bone score (TBS) into their exams.
At the Cleveland Clinic Center for Specialized Women’s Health, factoring in the TBS changed the diagnosis for 16% of 432 patients, according to Holly Thacker, MD, the center’s director.
“Importantly, 11% got worse diagnoses, and I use that in terms of prioritizing treatment,” Dr. Thacker said in an interview. The ability to determine how degraded the bone microarchitecture is through a software system “is a huge advance.”
Dr. Thacker described her center’s experience with the technology at the annual meeting of the Menopause Society (formerly The North American Menopause Society).
While BMD captures the amount of minerals like calcium in the skeleton, TBS assesses the underlying microarchitecture by looking at the distribution of shades of gray on dual-energy x-ray absorptiometry (DXA) scans.
Based on the TBS, patients’ bones are classified as normal, partially degraded, or degraded. Among the 432 patients who received a TBS analysis in 2022, 3% shifted from a normal diagnosis to osteopenia, 8% worsened from osteopenia to osteoporosis, 4% went from osteopenia to normal, and 1.6% downgraded from osteoporosis to osteopenia, Dr. Thacker reported.
The new test may also provide some reassurance for female patients who have thinner bones, which may raise alarms based on BMD. TBS, however, may show that the structure of the bone looks normal.
“When you know that the microarchitecture is normal, you’re a lot less concerned that they actually have a bone disease of osteoporosis,” Dr. Thacker said.
Conversely, unexpectedly degraded bone raises questions.
“That makes you go back and say [to the patient]: ‘Have you been on steroids? Were you malnourished? Is there some other metabolic problem? Have you had some calcium disorder?’ ” Dr. Thacker said.
Dr. Thacker leverages the TBS to help patients obtain effective therapy, typically an anabolic agent followed by antiresorptive medication.
“When I see a patient who not only has osteoporosis on bone density but has completely degraded bone architecture, it’s a lot easier for me to make the argument to the insurance company that this patient is at grave risk for a low trauma fracture and bad outcome without the best treatment,” Dr. Thacker said.
10-year-old tech, recently covered
The Food and Drug Administration approved TBS software in 2012, but Medicare only recently started paying for it.
Medimaps Group, a company that markets imaging software to calculate TBS, announced in 2022 that reimbursement from the Centers for Medicare & Medicaid Services was available, at $41.53 on the Physician Fee Schedule and $82.61 on the Hospital Outpatient Prospective Payment Schedule.
“Reimbursement through CMS is an important endorsement of the clinical value of TBS for clinicians and their patients,” Didier Hans, PhD, MBA, the CEO of Medimaps, said in a statement at the time. He noted that more than 600,000 TBS procedures were being performed in the United States each year.
Nevertheless, the initial investment in purchasing the software may be a barrier for health systems.
“We are the first and only site in our health system to offer TBS, as this is an extra expense and not uniformly reimbursed by insurers,” Dr. Thacker reported at the meeting.
Potential drawbacks
The TBS software used in Dr. Thacker’s study has been validated only in Asian and White patients between certain ages and weights, meaning the system is not designed to be used for other populations. Other researchers have highlighted a need for trabecular bone scoring to be validated more broadly. The authors of a recent analysis, however, suggest that TBS can be used the same way no matter a patient’s race.
TBS “is going to be most helpful in those with osteopenia who are right near the threshold for treatment,” said Marcella Donovan Walker, MD, MS, in a presentation on bone quality at the meeting.
Many studies have shown that TBS “provides additive information to bone density,” said Dr. Walker, a professor of medicine in the division of endocrinology at Columbia University, New York. For example, a large study of women in Manitoba found that, regardless of whether their bone density was normal, osteopenic, or osteoporotic, those with a low TBS had a much higher risk for fracture.
‘Opportunistic screening’ with CT?
TBS relies on the same DXA scans that are used to calculate bone mineral density, so obtaining the score does not add time or radiation to the scanning process, Dr. Thacker said.
But many patients who should receive DXA scans do not, which adds to the promise of “opportunistic screening” for osteoporosis, Dr. Walker said. With this approach, physicians would analyze a CT scan that a patient received for another purpose, such as to investigate abdominal pain or chest pain.
“In these images is information about the bone,” Dr. Walker said.
Researchers have used high-resolution peripheral quantitative CT to perform finite element analysis, where a computer program simulates compression of the bone to create a measure of bone stiffness and determine the load required for a break.
One study found that including those elements predicted fractures better than bone mineral density or the Fracture Risk Assessment Tool alone, Dr. Walker noted.
Other aspects of bone quality include how many cracks are in the bone, the amount of adipose in the marrow space, and the rate at which bone is broken down and rebuilt. But Dr. Walker suggested that the longstanding focus on bone mineral density in clinical practice makes sense.
“By far, bone mass is the most important bone quality,” Dr. Walker said.
Dr. Thacker is the executive director of the nonprofit Speaking of Women’s Health. Dr. Walker reported receiving funding from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and Amgen.
A version of this article first appeared on Medscape.com.
FROM NAMS 2023
Nonsurgical option for more large thyroid nodule patients?
WASHINGTON – , compared with only those that were.
While more research is needed, “the risk of false negative FNA results for large nodules may not be as high as reported in previous studies if you include patients who do not have indication for surgery, such as compressive symptoms, suspicious ultrasound features, etc.,” senior author Tracy Tylee, MD, an associate professor of endocrinology at the University of Washington, Seattle, said in an interview.
The implication is that nonsurgical options such as radiofrequency ablation may be appropriate for more patients than realized, she added.
“Clinicians should consider following these patients more conservatively, either with a second FNA to confirm [the] nodule is benign or with ultrasound follow-up for 5 years with intervention only if [there are] significant changes on imaging,” she said.
The findings were presented at the annual meeting of the American Thyroid Association.
Concerns about nodules over 4 cm having high false negative rates
Management of large thyroid nodules over 4 cm that are classified as Bethesda II, indicative of being benign, is complicated by concerns of false negatives in such cases. While the false negative rate for thyroid nodules in general is approximately 3%, the rate for large nodules over 4 cm has been reported as high as 35%.
Importantly, however, most studies evaluating the issue only involve patients who have received thyroid surgery, whereas most benign nodules are not referred for surgery.
“This may overestimate the false negative FNA biopsy risk for this group,” first author Melbin Thomas, MD, also of the University of Washington, said in her talk.
To better assess the false negative rate in the broader context of large nodules that did and did not undergo surgery, Dr. Thomas and her colleagues conducted a retrospective chart review of all patients undergoing FNA biopsy at her center between 2008 and 2014 for thyroid nodules larger than 4 cm and initially classified as Bethesda II, or benign.
With a follow-up of up to 10 years, nodules were considered accurately benign if they showed benign pathology on surgical resection, if they remained benign based on repeat FNA biopsy with Bethesda II results, or if there were no changes on imaging characteristics on ultrasound after at least 2 years.
Overall, 47 nodules over 4 cm and Bethesda II cytology were included, with an average follow-up of 5 years (range 2.2-9.7 years).
Of the nodules, 23 were treated with surgery, two of which were determined to have been malignant (8.7%) and, hence, false negatives. Nine of the nodules had repeat FNA, with none found to be malignant, and 15 received repeat ultrasound, also with no malignancies.
Overall, the false negative rate including all patients was 4.3%.
“False negative FNA biopsy results were not markedly elevated if nodules greater than 4 cm are evaluated, but rates were considerably higher if limited to surgical patients,” Dr. Thomas said.
Clinicians may be compelled to perform more aggressive surgery on large but benign thyroid nodules for a number of reasons, Dr. Tylee noted.
“A concern is that we may discontinue follow-up on these larger nodules and fail to diagnose a cancer early on, before there has been extrathyroidal extension or lymph node metastases,” she said.
In such cases, patients could wind up presenting at a higher stage of disease and require more intensive therapy.
However, with a low false negative rate overall, “all of this can increase the long-term health care costs and anxiety for patients, so having a better understanding of the true benign rate for large nodules is important,” she concluded.
Commenting on the research, Rodis D. Paparodis, MD, chief of Endocrinology, Diabetes, and Metabolism Clinics, in Patras, Greece, said the findings underscore that, as a surgical procedure, “thyroidectomy should be used cautiously, only when the benefit outweighs the risk.”
In his own previous multicenter study, Dr. Paparodis conducted a review of nearly 2,500 thyroidectomies that were performed based on size or longterm slow growth despite preoperative benign FNA findings. The results showed that only 1.9% of patients had any form of thyroid cancer in the nodule that had led to surgery; however, multiple other significant cancers were often present in other locations in the gland.
“Therefore, we suggest that careful sonographic evaluation of all thyroid nodules is warranted prior to deciding and planning the extent of surgical management for multinodular goiter,” he told this news organization.
“In addition, FNA of all suspicious nodules is required as well, to avoid unnecessary surprises in surgical pathology.”
Dr. Tylee, Dr. Thomas, and Dr. Paparodis report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
WASHINGTON – , compared with only those that were.
While more research is needed, “the risk of false negative FNA results for large nodules may not be as high as reported in previous studies if you include patients who do not have indication for surgery, such as compressive symptoms, suspicious ultrasound features, etc.,” senior author Tracy Tylee, MD, an associate professor of endocrinology at the University of Washington, Seattle, said in an interview.
The implication is that nonsurgical options such as radiofrequency ablation may be appropriate for more patients than realized, she added.
“Clinicians should consider following these patients more conservatively, either with a second FNA to confirm [the] nodule is benign or with ultrasound follow-up for 5 years with intervention only if [there are] significant changes on imaging,” she said.
The findings were presented at the annual meeting of the American Thyroid Association.
Concerns about nodules over 4 cm having high false negative rates
Management of large thyroid nodules over 4 cm that are classified as Bethesda II, indicative of being benign, is complicated by concerns of false negatives in such cases. While the false negative rate for thyroid nodules in general is approximately 3%, the rate for large nodules over 4 cm has been reported as high as 35%.
Importantly, however, most studies evaluating the issue only involve patients who have received thyroid surgery, whereas most benign nodules are not referred for surgery.
“This may overestimate the false negative FNA biopsy risk for this group,” first author Melbin Thomas, MD, also of the University of Washington, said in her talk.
To better assess the false negative rate in the broader context of large nodules that did and did not undergo surgery, Dr. Thomas and her colleagues conducted a retrospective chart review of all patients undergoing FNA biopsy at her center between 2008 and 2014 for thyroid nodules larger than 4 cm and initially classified as Bethesda II, or benign.
With a follow-up of up to 10 years, nodules were considered accurately benign if they showed benign pathology on surgical resection, if they remained benign based on repeat FNA biopsy with Bethesda II results, or if there were no changes on imaging characteristics on ultrasound after at least 2 years.
Overall, 47 nodules over 4 cm and Bethesda II cytology were included, with an average follow-up of 5 years (range 2.2-9.7 years).
Of the nodules, 23 were treated with surgery, two of which were determined to have been malignant (8.7%) and, hence, false negatives. Nine of the nodules had repeat FNA, with none found to be malignant, and 15 received repeat ultrasound, also with no malignancies.
Overall, the false negative rate including all patients was 4.3%.
“False negative FNA biopsy results were not markedly elevated if nodules greater than 4 cm are evaluated, but rates were considerably higher if limited to surgical patients,” Dr. Thomas said.
Clinicians may be compelled to perform more aggressive surgery on large but benign thyroid nodules for a number of reasons, Dr. Tylee noted.
“A concern is that we may discontinue follow-up on these larger nodules and fail to diagnose a cancer early on, before there has been extrathyroidal extension or lymph node metastases,” she said.
In such cases, patients could wind up presenting at a higher stage of disease and require more intensive therapy.
However, with a low false negative rate overall, “all of this can increase the long-term health care costs and anxiety for patients, so having a better understanding of the true benign rate for large nodules is important,” she concluded.
Commenting on the research, Rodis D. Paparodis, MD, chief of Endocrinology, Diabetes, and Metabolism Clinics, in Patras, Greece, said the findings underscore that, as a surgical procedure, “thyroidectomy should be used cautiously, only when the benefit outweighs the risk.”
In his own previous multicenter study, Dr. Paparodis conducted a review of nearly 2,500 thyroidectomies that were performed based on size or longterm slow growth despite preoperative benign FNA findings. The results showed that only 1.9% of patients had any form of thyroid cancer in the nodule that had led to surgery; however, multiple other significant cancers were often present in other locations in the gland.
“Therefore, we suggest that careful sonographic evaluation of all thyroid nodules is warranted prior to deciding and planning the extent of surgical management for multinodular goiter,” he told this news organization.
“In addition, FNA of all suspicious nodules is required as well, to avoid unnecessary surprises in surgical pathology.”
Dr. Tylee, Dr. Thomas, and Dr. Paparodis report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
WASHINGTON – , compared with only those that were.
While more research is needed, “the risk of false negative FNA results for large nodules may not be as high as reported in previous studies if you include patients who do not have indication for surgery, such as compressive symptoms, suspicious ultrasound features, etc.,” senior author Tracy Tylee, MD, an associate professor of endocrinology at the University of Washington, Seattle, said in an interview.
The implication is that nonsurgical options such as radiofrequency ablation may be appropriate for more patients than realized, she added.
“Clinicians should consider following these patients more conservatively, either with a second FNA to confirm [the] nodule is benign or with ultrasound follow-up for 5 years with intervention only if [there are] significant changes on imaging,” she said.
The findings were presented at the annual meeting of the American Thyroid Association.
Concerns about nodules over 4 cm having high false negative rates
Management of large thyroid nodules over 4 cm that are classified as Bethesda II, indicative of being benign, is complicated by concerns of false negatives in such cases. While the false negative rate for thyroid nodules in general is approximately 3%, the rate for large nodules over 4 cm has been reported as high as 35%.
Importantly, however, most studies evaluating the issue only involve patients who have received thyroid surgery, whereas most benign nodules are not referred for surgery.
“This may overestimate the false negative FNA biopsy risk for this group,” first author Melbin Thomas, MD, also of the University of Washington, said in her talk.
To better assess the false negative rate in the broader context of large nodules that did and did not undergo surgery, Dr. Thomas and her colleagues conducted a retrospective chart review of all patients undergoing FNA biopsy at her center between 2008 and 2014 for thyroid nodules larger than 4 cm and initially classified as Bethesda II, or benign.
With a follow-up of up to 10 years, nodules were considered accurately benign if they showed benign pathology on surgical resection, if they remained benign based on repeat FNA biopsy with Bethesda II results, or if there were no changes on imaging characteristics on ultrasound after at least 2 years.
Overall, 47 nodules over 4 cm and Bethesda II cytology were included, with an average follow-up of 5 years (range 2.2-9.7 years).
Of the nodules, 23 were treated with surgery, two of which were determined to have been malignant (8.7%) and, hence, false negatives. Nine of the nodules had repeat FNA, with none found to be malignant, and 15 received repeat ultrasound, also with no malignancies.
Overall, the false negative rate including all patients was 4.3%.
“False negative FNA biopsy results were not markedly elevated if nodules greater than 4 cm are evaluated, but rates were considerably higher if limited to surgical patients,” Dr. Thomas said.
Clinicians may be compelled to perform more aggressive surgery on large but benign thyroid nodules for a number of reasons, Dr. Tylee noted.
“A concern is that we may discontinue follow-up on these larger nodules and fail to diagnose a cancer early on, before there has been extrathyroidal extension or lymph node metastases,” she said.
In such cases, patients could wind up presenting at a higher stage of disease and require more intensive therapy.
However, with a low false negative rate overall, “all of this can increase the long-term health care costs and anxiety for patients, so having a better understanding of the true benign rate for large nodules is important,” she concluded.
Commenting on the research, Rodis D. Paparodis, MD, chief of Endocrinology, Diabetes, and Metabolism Clinics, in Patras, Greece, said the findings underscore that, as a surgical procedure, “thyroidectomy should be used cautiously, only when the benefit outweighs the risk.”
In his own previous multicenter study, Dr. Paparodis conducted a review of nearly 2,500 thyroidectomies that were performed based on size or longterm slow growth despite preoperative benign FNA findings. The results showed that only 1.9% of patients had any form of thyroid cancer in the nodule that had led to surgery; however, multiple other significant cancers were often present in other locations in the gland.
“Therefore, we suggest that careful sonographic evaluation of all thyroid nodules is warranted prior to deciding and planning the extent of surgical management for multinodular goiter,” he told this news organization.
“In addition, FNA of all suspicious nodules is required as well, to avoid unnecessary surprises in surgical pathology.”
Dr. Tylee, Dr. Thomas, and Dr. Paparodis report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ATA 2023




