User login
Dermatologic Manifestations of Musicians: A Case Report and Review of Skin Conditions in Musicians
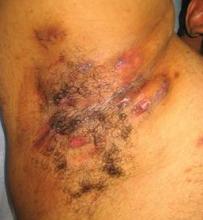
AAD: Adalimumab Improves Life Quality for Hidradenitis Suppurativa Patients
NEW ORLEANS – Adalimumab demonstrated significant efficacy for moderate to severe hidradenitis suppurativa in an interim 16-week analysis of a 52-week placebo-controlled randomized trial.
"Overall I was really pleased with seeing these results in terms of moving the science forward and managing these patients. ...This is a disease that I think has really been under-appreciated and under-managed and under-researched," Dr. Alexa B. Kimball said in presenting the interim phase II study findings at the annual meeting of the American Academy of Dermatology.
Anti–tumor necrosis factor (TNF) therapy is of considerable research interest because of the disease's prominent inflammatory component and the shortcomings of current therapies, explained Dr. Kimball of Harvard Medical School, Boston.
The clinical trial involved 154 patients with moderate to severe hidradenitis suppurativa who were randomized to 16 weeks of double-blind adalimumab (Humira) at 40 mg per week, 40 mg every other week, or to placebo. After 16 weeks, everyone was placed on 36 weeks of open-label adalimumab at 40 mg every other week, the standard psoriasis dosing. The loading dose was 80 mg at week 0 in the every-other-week group and 160 mg at week 0 and 80 mg at week 2 in the weekly therapy arm.
Because no practical instrument existed for grading the severity of hidradenitis suppurativa and scoring changes over time, Dr. Kimball and her coinvestigators created one: the Hidradenitis Suppurativa Physician Global Assessment (HS-PGA).
At baseline, two-thirds of participants had moderate disease as defined by the HS-PGA. Another 20% had very severe disease, with more than five abscesses or draining fistulas. The rest had severe disease.
At 16 weeks, 49% of patients on weekly adalimumab had improved to clear, mild, or minimal disease status. This was a significantly better result than the 21% rate in patients on alternate-week adalimumab or the 24% on placebo.
The primary study end point required achieving an HS-PGA score of clear, minimal, or mild plus at least a two-grade improvement. However, the 20% of subjects with baseline severe disease could only reach the primary end point by improving at least three grades to mild disease, and this proved a high bar for the severely affected subgroup, who often have scarring and tracts that are irreversible. Nonetheless, 18% of patients on weekly adalimumab achieved the primary end point, a significantly higher proportion than the 10% on every-other-week anti-TNF therapy or the 4% on placebo, Dr. Kimball noted.
Skin pain was a prominent feature among study participants. Their mean baseline pain score was roughly 55 points on a 0-100 visual analog scale. At week 16, however, 48% of patients in the weekly adalimumab arm had a 30% or greater reduction in pain score, compared with baseline, which is widely accepted in pain studies as a clinically meaningful improvement. This was a significantly higher proportion than the 36% rate among patients on alternate-week adalimumab or the 27% rate among controls.
Treatment-emergent side effects were what dermatologists have come to expect with anti-TNF therapy for psoriasis. Serious adverse events occurred in two patients in the placebo arm, three on alternate-week adalimumab, and four on weekly therapy with the biologic agent.
Dr. Kimball said she suspects that minor skin infections, which were fairly common in the first 16 weeks of the study, will become less of an issue with longer-term adalimumab therapy, as inflammation diminishes and the substantial baseline microbiologic load decreases. In the future, she added, it might be useful to prescribe antibiotics during the first 12-16 weeks of adalimumab therapy while waiting for the TNF inhibitor to help patients gain good control of the disease.
She noted that the hidradenitis suppurativa population in this study differed in several key respects from the psoriasis patients for whom dermatologists are accustomed to prescribing anti-TNF biologics. Seventy percent of participants in the hidradenitis suppurativa study were women, and the mean body weight across the three study arms was 95-100 kg. In contrast, typically two-thirds of participants in studies of anti-TNF therapy for psoriasis are men, with a mean weight of 85-90 kg.
Effective new therapy for hidradenitis suppurativa is sorely needed because affected patients often end up undergoing surgery, which entails removing large areas of skin and a 4- to 6-month recovery. "While surgery does result in significant improvements in quality of life overall, it’s a really long haul and incredibly disfiguring," the dermatologist observed.
Dr. Kimball declared that she serves as an investigator for and consultant to Abbott Laboratories, which is sponsoring the phase II trial, and in similar capacities with other pharmaceutical companies.
NEW ORLEANS – Adalimumab demonstrated significant efficacy for moderate to severe hidradenitis suppurativa in an interim 16-week analysis of a 52-week placebo-controlled randomized trial.
"Overall I was really pleased with seeing these results in terms of moving the science forward and managing these patients. ...This is a disease that I think has really been under-appreciated and under-managed and under-researched," Dr. Alexa B. Kimball said in presenting the interim phase II study findings at the annual meeting of the American Academy of Dermatology.
Anti–tumor necrosis factor (TNF) therapy is of considerable research interest because of the disease's prominent inflammatory component and the shortcomings of current therapies, explained Dr. Kimball of Harvard Medical School, Boston.
The clinical trial involved 154 patients with moderate to severe hidradenitis suppurativa who were randomized to 16 weeks of double-blind adalimumab (Humira) at 40 mg per week, 40 mg every other week, or to placebo. After 16 weeks, everyone was placed on 36 weeks of open-label adalimumab at 40 mg every other week, the standard psoriasis dosing. The loading dose was 80 mg at week 0 in the every-other-week group and 160 mg at week 0 and 80 mg at week 2 in the weekly therapy arm.
Because no practical instrument existed for grading the severity of hidradenitis suppurativa and scoring changes over time, Dr. Kimball and her coinvestigators created one: the Hidradenitis Suppurativa Physician Global Assessment (HS-PGA).
At baseline, two-thirds of participants had moderate disease as defined by the HS-PGA. Another 20% had very severe disease, with more than five abscesses or draining fistulas. The rest had severe disease.
At 16 weeks, 49% of patients on weekly adalimumab had improved to clear, mild, or minimal disease status. This was a significantly better result than the 21% rate in patients on alternate-week adalimumab or the 24% on placebo.
The primary study end point required achieving an HS-PGA score of clear, minimal, or mild plus at least a two-grade improvement. However, the 20% of subjects with baseline severe disease could only reach the primary end point by improving at least three grades to mild disease, and this proved a high bar for the severely affected subgroup, who often have scarring and tracts that are irreversible. Nonetheless, 18% of patients on weekly adalimumab achieved the primary end point, a significantly higher proportion than the 10% on every-other-week anti-TNF therapy or the 4% on placebo, Dr. Kimball noted.
Skin pain was a prominent feature among study participants. Their mean baseline pain score was roughly 55 points on a 0-100 visual analog scale. At week 16, however, 48% of patients in the weekly adalimumab arm had a 30% or greater reduction in pain score, compared with baseline, which is widely accepted in pain studies as a clinically meaningful improvement. This was a significantly higher proportion than the 36% rate among patients on alternate-week adalimumab or the 27% rate among controls.
Treatment-emergent side effects were what dermatologists have come to expect with anti-TNF therapy for psoriasis. Serious adverse events occurred in two patients in the placebo arm, three on alternate-week adalimumab, and four on weekly therapy with the biologic agent.
Dr. Kimball said she suspects that minor skin infections, which were fairly common in the first 16 weeks of the study, will become less of an issue with longer-term adalimumab therapy, as inflammation diminishes and the substantial baseline microbiologic load decreases. In the future, she added, it might be useful to prescribe antibiotics during the first 12-16 weeks of adalimumab therapy while waiting for the TNF inhibitor to help patients gain good control of the disease.
She noted that the hidradenitis suppurativa population in this study differed in several key respects from the psoriasis patients for whom dermatologists are accustomed to prescribing anti-TNF biologics. Seventy percent of participants in the hidradenitis suppurativa study were women, and the mean body weight across the three study arms was 95-100 kg. In contrast, typically two-thirds of participants in studies of anti-TNF therapy for psoriasis are men, with a mean weight of 85-90 kg.
Effective new therapy for hidradenitis suppurativa is sorely needed because affected patients often end up undergoing surgery, which entails removing large areas of skin and a 4- to 6-month recovery. "While surgery does result in significant improvements in quality of life overall, it’s a really long haul and incredibly disfiguring," the dermatologist observed.
Dr. Kimball declared that she serves as an investigator for and consultant to Abbott Laboratories, which is sponsoring the phase II trial, and in similar capacities with other pharmaceutical companies.
NEW ORLEANS – Adalimumab demonstrated significant efficacy for moderate to severe hidradenitis suppurativa in an interim 16-week analysis of a 52-week placebo-controlled randomized trial.
"Overall I was really pleased with seeing these results in terms of moving the science forward and managing these patients. ...This is a disease that I think has really been under-appreciated and under-managed and under-researched," Dr. Alexa B. Kimball said in presenting the interim phase II study findings at the annual meeting of the American Academy of Dermatology.
Anti–tumor necrosis factor (TNF) therapy is of considerable research interest because of the disease's prominent inflammatory component and the shortcomings of current therapies, explained Dr. Kimball of Harvard Medical School, Boston.
The clinical trial involved 154 patients with moderate to severe hidradenitis suppurativa who were randomized to 16 weeks of double-blind adalimumab (Humira) at 40 mg per week, 40 mg every other week, or to placebo. After 16 weeks, everyone was placed on 36 weeks of open-label adalimumab at 40 mg every other week, the standard psoriasis dosing. The loading dose was 80 mg at week 0 in the every-other-week group and 160 mg at week 0 and 80 mg at week 2 in the weekly therapy arm.
Because no practical instrument existed for grading the severity of hidradenitis suppurativa and scoring changes over time, Dr. Kimball and her coinvestigators created one: the Hidradenitis Suppurativa Physician Global Assessment (HS-PGA).
At baseline, two-thirds of participants had moderate disease as defined by the HS-PGA. Another 20% had very severe disease, with more than five abscesses or draining fistulas. The rest had severe disease.
At 16 weeks, 49% of patients on weekly adalimumab had improved to clear, mild, or minimal disease status. This was a significantly better result than the 21% rate in patients on alternate-week adalimumab or the 24% on placebo.
The primary study end point required achieving an HS-PGA score of clear, minimal, or mild plus at least a two-grade improvement. However, the 20% of subjects with baseline severe disease could only reach the primary end point by improving at least three grades to mild disease, and this proved a high bar for the severely affected subgroup, who often have scarring and tracts that are irreversible. Nonetheless, 18% of patients on weekly adalimumab achieved the primary end point, a significantly higher proportion than the 10% on every-other-week anti-TNF therapy or the 4% on placebo, Dr. Kimball noted.
Skin pain was a prominent feature among study participants. Their mean baseline pain score was roughly 55 points on a 0-100 visual analog scale. At week 16, however, 48% of patients in the weekly adalimumab arm had a 30% or greater reduction in pain score, compared with baseline, which is widely accepted in pain studies as a clinically meaningful improvement. This was a significantly higher proportion than the 36% rate among patients on alternate-week adalimumab or the 27% rate among controls.
Treatment-emergent side effects were what dermatologists have come to expect with anti-TNF therapy for psoriasis. Serious adverse events occurred in two patients in the placebo arm, three on alternate-week adalimumab, and four on weekly therapy with the biologic agent.
Dr. Kimball said she suspects that minor skin infections, which were fairly common in the first 16 weeks of the study, will become less of an issue with longer-term adalimumab therapy, as inflammation diminishes and the substantial baseline microbiologic load decreases. In the future, she added, it might be useful to prescribe antibiotics during the first 12-16 weeks of adalimumab therapy while waiting for the TNF inhibitor to help patients gain good control of the disease.
She noted that the hidradenitis suppurativa population in this study differed in several key respects from the psoriasis patients for whom dermatologists are accustomed to prescribing anti-TNF biologics. Seventy percent of participants in the hidradenitis suppurativa study were women, and the mean body weight across the three study arms was 95-100 kg. In contrast, typically two-thirds of participants in studies of anti-TNF therapy for psoriasis are men, with a mean weight of 85-90 kg.
Effective new therapy for hidradenitis suppurativa is sorely needed because affected patients often end up undergoing surgery, which entails removing large areas of skin and a 4- to 6-month recovery. "While surgery does result in significant improvements in quality of life overall, it’s a really long haul and incredibly disfiguring," the dermatologist observed.
Dr. Kimball declared that she serves as an investigator for and consultant to Abbott Laboratories, which is sponsoring the phase II trial, and in similar capacities with other pharmaceutical companies.
FROM THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF DERMATOLOGY
Major Finding: At 16 weeks, 49% of patients on weekly adalimumab had improved to clear, mild, or minimal disease status. This was a significantly better result than the 21% rate in patients on alternate-week adalimumab or the 24% on placebo.
Data Source: A clinical trial involving 154 patients with moderate to severe hidradenitis suppurativa who were randomized to 16 weeks of double-blind adalimumab (Humira) at 40 mg per week, 40 mg every other week, or to placebo. After 16 weeks, everyone was placed on 36 weeks of open-label adalimumab at 40 mg every other week.
Disclosures: Dr. Kimball declared that she serves as an investigator for and consultant to Abbott Laboratories, which is sponsoring the Phase II trial, and in similar capacities with other pharmaceutical companies.
AAD: Lasers, Light Therapy Hold Promise for Onychomycosis
NEW ORLEANS - Lasers and photodynamic therapy for the treatment of fungal toenails are beginning to generate substantial buzz among patients and podiatrists, but key questions regarding these novel proposed device therapies remain to be answered before they can truly be said to be the future of onychomycosis therapy.
For laser therapy, these questions include "Does it actually work?" and if so, by what mechanism? Dr. Boni E. Elewski said at the annual meeting of the American Academy of Dermatology.
Interest in laser therapy for fungal nails took off when one device, the PinPointe FootLaser, received Food and Drug Administration clearance for onychomycosis last October. Of note, however, the FDA didn't clear the device as a curative therapy, but rather "for the temporary increase of clear nail in patients with onychomycosis." This hasn't stopped some podiatrists from offering treatment with the PinPointe or other neodymium:YAG 1,064-nm lasers at a price tag of up to $1,000 per toe, a marketing ploy that implies definitive therapy and prompted Dr. Elewski to take a closer look.
Her in vitro studies in the mycology lab have left her convinced that lasers don't eradicate fungi through heat killing; the required nail temperatures would be intolerably painful. Moreover, direct lasering of fungi on agar plates and dilute broth had absolutely no impact on fungal growth. But these negative studies don't rule out other potential mechanisms of action, including a possible immunologic effect or laser-induced denaturization of enzymes that fungi need to digest skin cells, noted Dr. Elewski, professor of dermatology at the University of Alabama at Birmingham.
She is now conducting a clinical study in which patients with onychomycosis are being treated with an Nd:YAG 1,064-nm laser – not the PinPointe FootLaser – with a 5-mm spot size, a frequency of 2 Hz, and an energy density of 16 J/cm2. Patients get a total of five treatments, each consisting of more than 300 pulses administered over the nail during a couple of minutes in a predetermined pattern. Anecdotally, in individual patients she has observed instances of fungi evacuating laser-treated nails, and the nails becoming culture negative over a period of several months. The study, however, remains ongoing.
"The jury is still out. I can't say yet whether laser therapy works," Dr. Elewski commented.
Unlike laser therapy for onychomycosis, photodynamic therapy (PDT) is backed by a published rigorously conducted study. And the mechanism of action is understood: In vitro, Trichophyton rubrum absorbs 5-aminolevulinic acid and can be photo killed.
But onychomycosis is not an FDA-approved indication for PDT. Moreover, the results of the published study cited by Dr. Elewski – a 43% cure rate 12 months after PDT and 37% at 18 months of follow-up – are comparable to but not better than success rates attained in the major clinical trials of terbinafine. Plus, the PDT sessions must be preceded by a lengthy, labor-intensive chemical nail avulsion. Nonetheless, PDT may provide an alternative option for onychomycosis when terbinafine and other oral agents are contraindicated, she continued.
The PDT study involved 30 patients with onychomycosis resulting from Trichophyton rubrum who were treated at Aristotle University of Thessaloniki (Greece). Following 10 consecutive nights in which 20% urea ointment was applied under occlusion to the nail plate, dermatologists removed the nail with forceps and applied 20% 5-aminolevulinic acid for 3 hours before treatment with red light at 570-670 nm, a light density of 40 J/cm2, and a fluence of 40 mW/cm2. Patients got three treatment sessions, each 2 weeks apart.
The Greek investigators demanded a rigorous, FDA-style definition of cure: complete absence of clinical signs of fungal infection, or less than 10% of the nail being affected by subungual hyperkeratosis along with mycologic cure. Thirteen of 30 (43%) patients fulfilled this definition at 12 months, as did 11 (37%) at 18 months. No fungal resistance was seen (Acta Derm. Venereol. 2010;90:216-7).
Dr. Elewski said that she receives research support from Cutera.
NEW ORLEANS - Lasers and photodynamic therapy for the treatment of fungal toenails are beginning to generate substantial buzz among patients and podiatrists, but key questions regarding these novel proposed device therapies remain to be answered before they can truly be said to be the future of onychomycosis therapy.
For laser therapy, these questions include "Does it actually work?" and if so, by what mechanism? Dr. Boni E. Elewski said at the annual meeting of the American Academy of Dermatology.
Interest in laser therapy for fungal nails took off when one device, the PinPointe FootLaser, received Food and Drug Administration clearance for onychomycosis last October. Of note, however, the FDA didn't clear the device as a curative therapy, but rather "for the temporary increase of clear nail in patients with onychomycosis." This hasn't stopped some podiatrists from offering treatment with the PinPointe or other neodymium:YAG 1,064-nm lasers at a price tag of up to $1,000 per toe, a marketing ploy that implies definitive therapy and prompted Dr. Elewski to take a closer look.
Her in vitro studies in the mycology lab have left her convinced that lasers don't eradicate fungi through heat killing; the required nail temperatures would be intolerably painful. Moreover, direct lasering of fungi on agar plates and dilute broth had absolutely no impact on fungal growth. But these negative studies don't rule out other potential mechanisms of action, including a possible immunologic effect or laser-induced denaturization of enzymes that fungi need to digest skin cells, noted Dr. Elewski, professor of dermatology at the University of Alabama at Birmingham.
She is now conducting a clinical study in which patients with onychomycosis are being treated with an Nd:YAG 1,064-nm laser – not the PinPointe FootLaser – with a 5-mm spot size, a frequency of 2 Hz, and an energy density of 16 J/cm2. Patients get a total of five treatments, each consisting of more than 300 pulses administered over the nail during a couple of minutes in a predetermined pattern. Anecdotally, in individual patients she has observed instances of fungi evacuating laser-treated nails, and the nails becoming culture negative over a period of several months. The study, however, remains ongoing.
"The jury is still out. I can't say yet whether laser therapy works," Dr. Elewski commented.
Unlike laser therapy for onychomycosis, photodynamic therapy (PDT) is backed by a published rigorously conducted study. And the mechanism of action is understood: In vitro, Trichophyton rubrum absorbs 5-aminolevulinic acid and can be photo killed.
But onychomycosis is not an FDA-approved indication for PDT. Moreover, the results of the published study cited by Dr. Elewski – a 43% cure rate 12 months after PDT and 37% at 18 months of follow-up – are comparable to but not better than success rates attained in the major clinical trials of terbinafine. Plus, the PDT sessions must be preceded by a lengthy, labor-intensive chemical nail avulsion. Nonetheless, PDT may provide an alternative option for onychomycosis when terbinafine and other oral agents are contraindicated, she continued.
The PDT study involved 30 patients with onychomycosis resulting from Trichophyton rubrum who were treated at Aristotle University of Thessaloniki (Greece). Following 10 consecutive nights in which 20% urea ointment was applied under occlusion to the nail plate, dermatologists removed the nail with forceps and applied 20% 5-aminolevulinic acid for 3 hours before treatment with red light at 570-670 nm, a light density of 40 J/cm2, and a fluence of 40 mW/cm2. Patients got three treatment sessions, each 2 weeks apart.
The Greek investigators demanded a rigorous, FDA-style definition of cure: complete absence of clinical signs of fungal infection, or less than 10% of the nail being affected by subungual hyperkeratosis along with mycologic cure. Thirteen of 30 (43%) patients fulfilled this definition at 12 months, as did 11 (37%) at 18 months. No fungal resistance was seen (Acta Derm. Venereol. 2010;90:216-7).
Dr. Elewski said that she receives research support from Cutera.
NEW ORLEANS - Lasers and photodynamic therapy for the treatment of fungal toenails are beginning to generate substantial buzz among patients and podiatrists, but key questions regarding these novel proposed device therapies remain to be answered before they can truly be said to be the future of onychomycosis therapy.
For laser therapy, these questions include "Does it actually work?" and if so, by what mechanism? Dr. Boni E. Elewski said at the annual meeting of the American Academy of Dermatology.
Interest in laser therapy for fungal nails took off when one device, the PinPointe FootLaser, received Food and Drug Administration clearance for onychomycosis last October. Of note, however, the FDA didn't clear the device as a curative therapy, but rather "for the temporary increase of clear nail in patients with onychomycosis." This hasn't stopped some podiatrists from offering treatment with the PinPointe or other neodymium:YAG 1,064-nm lasers at a price tag of up to $1,000 per toe, a marketing ploy that implies definitive therapy and prompted Dr. Elewski to take a closer look.
Her in vitro studies in the mycology lab have left her convinced that lasers don't eradicate fungi through heat killing; the required nail temperatures would be intolerably painful. Moreover, direct lasering of fungi on agar plates and dilute broth had absolutely no impact on fungal growth. But these negative studies don't rule out other potential mechanisms of action, including a possible immunologic effect or laser-induced denaturization of enzymes that fungi need to digest skin cells, noted Dr. Elewski, professor of dermatology at the University of Alabama at Birmingham.
She is now conducting a clinical study in which patients with onychomycosis are being treated with an Nd:YAG 1,064-nm laser – not the PinPointe FootLaser – with a 5-mm spot size, a frequency of 2 Hz, and an energy density of 16 J/cm2. Patients get a total of five treatments, each consisting of more than 300 pulses administered over the nail during a couple of minutes in a predetermined pattern. Anecdotally, in individual patients she has observed instances of fungi evacuating laser-treated nails, and the nails becoming culture negative over a period of several months. The study, however, remains ongoing.
"The jury is still out. I can't say yet whether laser therapy works," Dr. Elewski commented.
Unlike laser therapy for onychomycosis, photodynamic therapy (PDT) is backed by a published rigorously conducted study. And the mechanism of action is understood: In vitro, Trichophyton rubrum absorbs 5-aminolevulinic acid and can be photo killed.
But onychomycosis is not an FDA-approved indication for PDT. Moreover, the results of the published study cited by Dr. Elewski – a 43% cure rate 12 months after PDT and 37% at 18 months of follow-up – are comparable to but not better than success rates attained in the major clinical trials of terbinafine. Plus, the PDT sessions must be preceded by a lengthy, labor-intensive chemical nail avulsion. Nonetheless, PDT may provide an alternative option for onychomycosis when terbinafine and other oral agents are contraindicated, she continued.
The PDT study involved 30 patients with onychomycosis resulting from Trichophyton rubrum who were treated at Aristotle University of Thessaloniki (Greece). Following 10 consecutive nights in which 20% urea ointment was applied under occlusion to the nail plate, dermatologists removed the nail with forceps and applied 20% 5-aminolevulinic acid for 3 hours before treatment with red light at 570-670 nm, a light density of 40 J/cm2, and a fluence of 40 mW/cm2. Patients got three treatment sessions, each 2 weeks apart.
The Greek investigators demanded a rigorous, FDA-style definition of cure: complete absence of clinical signs of fungal infection, or less than 10% of the nail being affected by subungual hyperkeratosis along with mycologic cure. Thirteen of 30 (43%) patients fulfilled this definition at 12 months, as did 11 (37%) at 18 months. No fungal resistance was seen (Acta Derm. Venereol. 2010;90:216-7).
Dr. Elewski said that she receives research support from Cutera.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF DERMATOLOGY
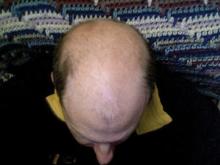
Early Balding Found to Double Prostate Cancer Risk
Men with prostate cancer are twice as likely to have had male pattern baldness starting at age 20, according to results of a study that found no increased risk among men who began balding in their 30s or 40s.
The findings, published Feb. 16 in Annals of Oncology (doi:10.1093/annonc/mdq695), suggest that men with early baldness may benefit from routine prostate cancer screening or preventive measures that could include the systematic use of 5-alpha reductase inhibitors, the researchers wrote.
For their research, Dr. Michael Yassa, who was a radiation oncology fellow at the Georges Pompidou European Hospital in Paris at the time of the study, and his associates studied 388 men with a diagnosis of prostate cancer, recruited from radiation oncology clinics in three French institutions. The study also included 281 matched controls with no history of cancer or hormonal pathologies, but with family histories similar to those of the cases. The mean age of the subjects was 67.2, and the controls, 66.4.
All study participants were asked to report any personal history of prostate cancer and their fathers' histories of the same, and to describe their balding pattern at ages 20, 30, and 40 along with their fathers', using a set of four images adapted from the Hamilton-Norwood scale of male pattern baldness. Case subjects' age at diagnosis, stage of disease at diagnosis, treatment, and other information were recorded.
The men with prostate cancer were twice as likely to have had male pattern baldness at age 20 (odds ratio [OR] 2.01). "This trend was lost at ages 30 or 40," the researchers wrote. No specific pattern of hair loss appeared to be a predictive factor for the development of prostate cancer.
Any balding present at age 20 was associated with an increased incidence of prostate cancer later in life. Cancer patients with early balding did not develop cancers younger – those with any pattern of balding by age 20 and 40 had a mean age of diagnosis of 64.4 and 64.5 years, respectively, compared with 64.3 years for patients with no balding by age 40. The researchers also found no associations between early balding and more aggressive types of tumors.
Dr. Yassa, now with the University of Montreal, and colleagues cited a number of earlier studies with conflicting evidence on the links between baldness and cancer. One Duke University study (Cancer Epidemiol. Biomarkers Prev. 2000;9:325-8) showed that men who developed vertex baldness by age 30 had nearly a twofold increase in risk of developing prostate cancer, but a more recent population-based study (Cancer Epidemiol. [doi:10.1016/j.canep.2010.02.003]) showed baldness at age 30 to be associated with 29% relative risk reduction for prostate cancer.
The investigators in the current study speculated that androgens might be implicated in any link between early balding and cancer. "Finasteride blocks the conversion of testosterone to dihydrotestosterone, the active metabolite of testosterone, slowing the progression of androgenic alopecia and decreasing the incidence of prostate cancer," they wrote.
They also acknowledged that their own study was limited by its small size and a case-control design involving self-reporting, that could allow for recall and selective recall bias, and a lack of controlling for factors including African heritage and dietary differences.
Neither Dr. Hassa nor his coauthors declared conflicts of interest.
Men with prostate cancer are twice as likely to have had male pattern baldness starting at age 20, according to results of a study that found no increased risk among men who began balding in their 30s or 40s.
The findings, published Feb. 16 in Annals of Oncology (doi:10.1093/annonc/mdq695), suggest that men with early baldness may benefit from routine prostate cancer screening or preventive measures that could include the systematic use of 5-alpha reductase inhibitors, the researchers wrote.
For their research, Dr. Michael Yassa, who was a radiation oncology fellow at the Georges Pompidou European Hospital in Paris at the time of the study, and his associates studied 388 men with a diagnosis of prostate cancer, recruited from radiation oncology clinics in three French institutions. The study also included 281 matched controls with no history of cancer or hormonal pathologies, but with family histories similar to those of the cases. The mean age of the subjects was 67.2, and the controls, 66.4.
All study participants were asked to report any personal history of prostate cancer and their fathers' histories of the same, and to describe their balding pattern at ages 20, 30, and 40 along with their fathers', using a set of four images adapted from the Hamilton-Norwood scale of male pattern baldness. Case subjects' age at diagnosis, stage of disease at diagnosis, treatment, and other information were recorded.
The men with prostate cancer were twice as likely to have had male pattern baldness at age 20 (odds ratio [OR] 2.01). "This trend was lost at ages 30 or 40," the researchers wrote. No specific pattern of hair loss appeared to be a predictive factor for the development of prostate cancer.
Any balding present at age 20 was associated with an increased incidence of prostate cancer later in life. Cancer patients with early balding did not develop cancers younger – those with any pattern of balding by age 20 and 40 had a mean age of diagnosis of 64.4 and 64.5 years, respectively, compared with 64.3 years for patients with no balding by age 40. The researchers also found no associations between early balding and more aggressive types of tumors.
Dr. Yassa, now with the University of Montreal, and colleagues cited a number of earlier studies with conflicting evidence on the links between baldness and cancer. One Duke University study (Cancer Epidemiol. Biomarkers Prev. 2000;9:325-8) showed that men who developed vertex baldness by age 30 had nearly a twofold increase in risk of developing prostate cancer, but a more recent population-based study (Cancer Epidemiol. [doi:10.1016/j.canep.2010.02.003]) showed baldness at age 30 to be associated with 29% relative risk reduction for prostate cancer.
The investigators in the current study speculated that androgens might be implicated in any link between early balding and cancer. "Finasteride blocks the conversion of testosterone to dihydrotestosterone, the active metabolite of testosterone, slowing the progression of androgenic alopecia and decreasing the incidence of prostate cancer," they wrote.
They also acknowledged that their own study was limited by its small size and a case-control design involving self-reporting, that could allow for recall and selective recall bias, and a lack of controlling for factors including African heritage and dietary differences.
Neither Dr. Hassa nor his coauthors declared conflicts of interest.
Men with prostate cancer are twice as likely to have had male pattern baldness starting at age 20, according to results of a study that found no increased risk among men who began balding in their 30s or 40s.
The findings, published Feb. 16 in Annals of Oncology (doi:10.1093/annonc/mdq695), suggest that men with early baldness may benefit from routine prostate cancer screening or preventive measures that could include the systematic use of 5-alpha reductase inhibitors, the researchers wrote.
For their research, Dr. Michael Yassa, who was a radiation oncology fellow at the Georges Pompidou European Hospital in Paris at the time of the study, and his associates studied 388 men with a diagnosis of prostate cancer, recruited from radiation oncology clinics in three French institutions. The study also included 281 matched controls with no history of cancer or hormonal pathologies, but with family histories similar to those of the cases. The mean age of the subjects was 67.2, and the controls, 66.4.
All study participants were asked to report any personal history of prostate cancer and their fathers' histories of the same, and to describe their balding pattern at ages 20, 30, and 40 along with their fathers', using a set of four images adapted from the Hamilton-Norwood scale of male pattern baldness. Case subjects' age at diagnosis, stage of disease at diagnosis, treatment, and other information were recorded.
The men with prostate cancer were twice as likely to have had male pattern baldness at age 20 (odds ratio [OR] 2.01). "This trend was lost at ages 30 or 40," the researchers wrote. No specific pattern of hair loss appeared to be a predictive factor for the development of prostate cancer.
Any balding present at age 20 was associated with an increased incidence of prostate cancer later in life. Cancer patients with early balding did not develop cancers younger – those with any pattern of balding by age 20 and 40 had a mean age of diagnosis of 64.4 and 64.5 years, respectively, compared with 64.3 years for patients with no balding by age 40. The researchers also found no associations between early balding and more aggressive types of tumors.
Dr. Yassa, now with the University of Montreal, and colleagues cited a number of earlier studies with conflicting evidence on the links between baldness and cancer. One Duke University study (Cancer Epidemiol. Biomarkers Prev. 2000;9:325-8) showed that men who developed vertex baldness by age 30 had nearly a twofold increase in risk of developing prostate cancer, but a more recent population-based study (Cancer Epidemiol. [doi:10.1016/j.canep.2010.02.003]) showed baldness at age 30 to be associated with 29% relative risk reduction for prostate cancer.
The investigators in the current study speculated that androgens might be implicated in any link between early balding and cancer. "Finasteride blocks the conversion of testosterone to dihydrotestosterone, the active metabolite of testosterone, slowing the progression of androgenic alopecia and decreasing the incidence of prostate cancer," they wrote.
They also acknowledged that their own study was limited by its small size and a case-control design involving self-reporting, that could allow for recall and selective recall bias, and a lack of controlling for factors including African heritage and dietary differences.
Neither Dr. Hassa nor his coauthors declared conflicts of interest.
FROM ANNALS OF ONCOLOGY
Major Finding: The men with prostate cancer were twice as likely to have had male pattern baldness at age 20 (OR 2.01).
Data Source: Men with a diagnosis of prostate cancer (n = 388), recruited from radiation
oncology clinics in three French institutions.
Disclosures: Neither Dr. Hassa nor his coauthors declared conflicts of interest.
AAD: Tinea Capitis Rates Falling Sharply In Northern California
NEW ORLEANS - The annual incidence of tinea capitis dropped dramatically during 1998-2007, at least in Northern California.
The incidence among children enrolled in Kaiser Permanente of Northern California was 1.3% in 1998 and again in 1999 before it began a steady decline, culminating in an incidence of 0.3% in 2007, Dr. Paradi Mirmirani reported at the annual meeting of the American Academy of Dermatology.
Her retrospective, population-based study included all Kaiser members younger than 15 years, with an average of 672,373 children per year. Some 70% of all cases of tinea capitis were diagnosed after age 5 years, noted Dr. Mirmirani of the Permanente Medical Group in Vallejo, Calif.
As in numerous other studies, the highest rates of tinea capitis in the Kaiser study occurred in black children. They were also the group with the sharpest decline during the 10-year study period. The incidence of tinea capitis in black children was 450 cases per 10,000 in 1998, plunging to about 200 per 10,000 by 2007.
The incidence declined significantly in all other ethnic groups as well, but those declines started from far lower baseline rates of 75-130 cases per 10,000 in 1998.
"That's quite a dramatic decrease. Maybe we're doing something right," commented session chair Dr. Richard L. Gallo, professor of medicine and pediatrics and chief of the division of dermatology at the University of California, San Diego.
Dr. Mirmirani said that girls had a significantly lower incidence than did boys (1.1% vs. 1.6%). Trichophyton tonsurans remained the predominant causative organism, as has been the case across the United States for decades. T. tonsurans accounted for 89% of all positive cultures at Kaiser in 1998, and 92% in 2007, Dr. Mirmirani continued.
The incidence of tinea capitis in Northern California showed no clear correlation with population density. Although the highest rate was seen in San Francisco County (the most population-dense county), the second-highest rate was in Solano County, which ranked only seventh in terms of population density.
Rates of coexisting atopic diseases in kids with tinea capitis were similar to those reported in the general population. In all, 16% of Kaiser patients with tinea capitis had a coexisting diagnosis of atopic dermatitis, 20% had asthma, and 15% had allergic rhinitis.
The big question, Dr. Mirmirani noted, is why the incidence of tinea capitis in 2007 was less than one-quarter the rate 10 years earlier. There are several possible explanations. For example, the number of new prescriptions for fluconazole and terbinafine rose significantly in the Kaiser system during the study years, while prescriptions for griseofulvin correspondingly declined. It's possible that the increased use of newer antifungal agents contributed to the fall in tinea capitis during 1998-2007.
It's also possible that since the late 1990s, dermatologists have done a better job of educating pediatricians and family physicians (the first-line physicians in dealing with dermatophyte infections) regarding recognition and treatment of cases of tinea capitis and carriers, she added.
Dr. Mirmirani observed that although T. tonsurans is the predominant pathogen today, it wasn't always so. In the 1940s and 1950s, it was Microsporum audouinii, an easier-to-diagnose organism that causes a more inflammatory infection and is visible by Wood's lamp.
"It seems like there's a constant tug of war between host and fungus, a bit of a cat-and-mouse game that causes the causative organism to evolve," the dermatologist said.
Her study was funded by the Kaiser Permanente division of research. Dr. Mirmirani declared having no relevant financial relationships with industry.
NEW ORLEANS - The annual incidence of tinea capitis dropped dramatically during 1998-2007, at least in Northern California.
The incidence among children enrolled in Kaiser Permanente of Northern California was 1.3% in 1998 and again in 1999 before it began a steady decline, culminating in an incidence of 0.3% in 2007, Dr. Paradi Mirmirani reported at the annual meeting of the American Academy of Dermatology.
Her retrospective, population-based study included all Kaiser members younger than 15 years, with an average of 672,373 children per year. Some 70% of all cases of tinea capitis were diagnosed after age 5 years, noted Dr. Mirmirani of the Permanente Medical Group in Vallejo, Calif.
As in numerous other studies, the highest rates of tinea capitis in the Kaiser study occurred in black children. They were also the group with the sharpest decline during the 10-year study period. The incidence of tinea capitis in black children was 450 cases per 10,000 in 1998, plunging to about 200 per 10,000 by 2007.
The incidence declined significantly in all other ethnic groups as well, but those declines started from far lower baseline rates of 75-130 cases per 10,000 in 1998.
"That's quite a dramatic decrease. Maybe we're doing something right," commented session chair Dr. Richard L. Gallo, professor of medicine and pediatrics and chief of the division of dermatology at the University of California, San Diego.
Dr. Mirmirani said that girls had a significantly lower incidence than did boys (1.1% vs. 1.6%). Trichophyton tonsurans remained the predominant causative organism, as has been the case across the United States for decades. T. tonsurans accounted for 89% of all positive cultures at Kaiser in 1998, and 92% in 2007, Dr. Mirmirani continued.
The incidence of tinea capitis in Northern California showed no clear correlation with population density. Although the highest rate was seen in San Francisco County (the most population-dense county), the second-highest rate was in Solano County, which ranked only seventh in terms of population density.
Rates of coexisting atopic diseases in kids with tinea capitis were similar to those reported in the general population. In all, 16% of Kaiser patients with tinea capitis had a coexisting diagnosis of atopic dermatitis, 20% had asthma, and 15% had allergic rhinitis.
The big question, Dr. Mirmirani noted, is why the incidence of tinea capitis in 2007 was less than one-quarter the rate 10 years earlier. There are several possible explanations. For example, the number of new prescriptions for fluconazole and terbinafine rose significantly in the Kaiser system during the study years, while prescriptions for griseofulvin correspondingly declined. It's possible that the increased use of newer antifungal agents contributed to the fall in tinea capitis during 1998-2007.
It's also possible that since the late 1990s, dermatologists have done a better job of educating pediatricians and family physicians (the first-line physicians in dealing with dermatophyte infections) regarding recognition and treatment of cases of tinea capitis and carriers, she added.
Dr. Mirmirani observed that although T. tonsurans is the predominant pathogen today, it wasn't always so. In the 1940s and 1950s, it was Microsporum audouinii, an easier-to-diagnose organism that causes a more inflammatory infection and is visible by Wood's lamp.
"It seems like there's a constant tug of war between host and fungus, a bit of a cat-and-mouse game that causes the causative organism to evolve," the dermatologist said.
Her study was funded by the Kaiser Permanente division of research. Dr. Mirmirani declared having no relevant financial relationships with industry.
NEW ORLEANS - The annual incidence of tinea capitis dropped dramatically during 1998-2007, at least in Northern California.
The incidence among children enrolled in Kaiser Permanente of Northern California was 1.3% in 1998 and again in 1999 before it began a steady decline, culminating in an incidence of 0.3% in 2007, Dr. Paradi Mirmirani reported at the annual meeting of the American Academy of Dermatology.
Her retrospective, population-based study included all Kaiser members younger than 15 years, with an average of 672,373 children per year. Some 70% of all cases of tinea capitis were diagnosed after age 5 years, noted Dr. Mirmirani of the Permanente Medical Group in Vallejo, Calif.
As in numerous other studies, the highest rates of tinea capitis in the Kaiser study occurred in black children. They were also the group with the sharpest decline during the 10-year study period. The incidence of tinea capitis in black children was 450 cases per 10,000 in 1998, plunging to about 200 per 10,000 by 2007.
The incidence declined significantly in all other ethnic groups as well, but those declines started from far lower baseline rates of 75-130 cases per 10,000 in 1998.
"That's quite a dramatic decrease. Maybe we're doing something right," commented session chair Dr. Richard L. Gallo, professor of medicine and pediatrics and chief of the division of dermatology at the University of California, San Diego.
Dr. Mirmirani said that girls had a significantly lower incidence than did boys (1.1% vs. 1.6%). Trichophyton tonsurans remained the predominant causative organism, as has been the case across the United States for decades. T. tonsurans accounted for 89% of all positive cultures at Kaiser in 1998, and 92% in 2007, Dr. Mirmirani continued.
The incidence of tinea capitis in Northern California showed no clear correlation with population density. Although the highest rate was seen in San Francisco County (the most population-dense county), the second-highest rate was in Solano County, which ranked only seventh in terms of population density.
Rates of coexisting atopic diseases in kids with tinea capitis were similar to those reported in the general population. In all, 16% of Kaiser patients with tinea capitis had a coexisting diagnosis of atopic dermatitis, 20% had asthma, and 15% had allergic rhinitis.
The big question, Dr. Mirmirani noted, is why the incidence of tinea capitis in 2007 was less than one-quarter the rate 10 years earlier. There are several possible explanations. For example, the number of new prescriptions for fluconazole and terbinafine rose significantly in the Kaiser system during the study years, while prescriptions for griseofulvin correspondingly declined. It's possible that the increased use of newer antifungal agents contributed to the fall in tinea capitis during 1998-2007.
It's also possible that since the late 1990s, dermatologists have done a better job of educating pediatricians and family physicians (the first-line physicians in dealing with dermatophyte infections) regarding recognition and treatment of cases of tinea capitis and carriers, she added.
Dr. Mirmirani observed that although T. tonsurans is the predominant pathogen today, it wasn't always so. In the 1940s and 1950s, it was Microsporum audouinii, an easier-to-diagnose organism that causes a more inflammatory infection and is visible by Wood's lamp.
"It seems like there's a constant tug of war between host and fungus, a bit of a cat-and-mouse game that causes the causative organism to evolve," the dermatologist said.
Her study was funded by the Kaiser Permanente division of research. Dr. Mirmirani declared having no relevant financial relationships with industry.
FROM THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF DERMATOLOGY
Major Finding: The incidence of tinea capitis among children enrolled in Kaiser Permanente of Northern California was 1.3% in 1998 and again in 1999 before it began a steady decline, culminating in an incidence of 0.3% in 2007.
Data Source: A retrospective, population-based study of 1998-2007 including all Kaiser members in Northern California under age 15 years, with an average of 672,373 children per year.
Disclosures: Dr. Mirmirani's study was funded by the Kaiser Permanente division of research. She declared having no relevant financial relationships with industry.
AAD: Topical 5-FU Well Tolerated for Pediatric Warts
NEW ORLEANS - Any dermatologist who's not regularly using topical 5% 5-fluorouracil cream for treatment of warts in children is missing out on a therapy that's singularly safe, well tolerated, and effective.
That's the considered opinion of Dr. Bari B. Cunningham, who not only uses this therapy on a daily basis in her Encinitas, Calif., pediatric dermatology practice, but was also senior author of an open-label study demonstrating its benefits.
"I would strongly urge you to consider this therapy. This is a pearl that will make a major impact on the pediatric patients you see every day," she promised at the annual meeting of the American Academy of Dermatology.
From her standpoint, the most important thing about topical 5-FU cream for warts in kids is its safety, since this therapy does after all involve off-label use of a potent drug in little children. Reassuringly, in her 39-patient study there were no detectable blood levels of 5-FU during or after 6 weeks of therapy.
From the perspective of the patient and family, however, the big appeal is the therapy's ease and tolerability.
"When you're a dermatologist dealing with kids you really need to look for alternatives to the painful therapies that we currently use. If you have child with extensive warts you really can't be considering painful treatment options such as liquid nitrogen or intralesional bleomycin. That's just not going to cut it, and that family is never going to come back to you again. It’s not humane to expect a child to sit through painful treatments for something that benign," explained Dr. Cunningham.
The 39 children in her study all had at least two hand warts to which topical 5-FU cream was applied once or twice daily under occlusion for 6 weeks. Eighty-eight percent of patients were significantly improved after 6 weeks, and 41% had complete resolution of at least one wart. The treatment response didn't differ between once- and twice-daily therapy. Tolerability and patient satisfaction were excellent. At 6 months of follow-up, 87% of complete responders had no wart recurrences (Pediatr. Dermatol. 2009;26:279-85).
Since publication of her study, Dr. Cunningham has modified how she uses topical 5-FU cream for pediatric warts. The drug is applied at night under duct tape occlusion, alternating with salicylic acid under duct tape occlusion every second night because she believes the two agents are complementary. The duct tape is removed each morning and the wart is left uncovered and untreated during the day.
"I only do this for a month at a time because that's really all I ever need. If this medicine is going to work – and it usually does – it works fast," she said.
The ideal warts for this form of therapy are single and rapidly growing. "The ones that are doubling in size every couple of weeks like they've got a mind of their own, those are the perfect ones to treat with topical 5-FU because 5-FU is going to impair cell division and really shut that wart down," Dr. Cunningham continued.
She offered a few cautionary notes: Don't use this therapy on periungual warts; it will result in serious onycholysis, and the nail will fall off. Avoid treating warts on the face. In order to prevent systemic absorption, don’t use topical 5-FU cream in orally fixated thumb suckers. And make sure to keep the medication out of reach of the family dog. Dogs have a paradoxical toxic reaction to topical 5-FU. There are dozens of reports of dog fatalities in the veterinary literature, Dr. Cunningham said.
She declared having no relevant financial interests.
NEW ORLEANS - Any dermatologist who's not regularly using topical 5% 5-fluorouracil cream for treatment of warts in children is missing out on a therapy that's singularly safe, well tolerated, and effective.
That's the considered opinion of Dr. Bari B. Cunningham, who not only uses this therapy on a daily basis in her Encinitas, Calif., pediatric dermatology practice, but was also senior author of an open-label study demonstrating its benefits.
"I would strongly urge you to consider this therapy. This is a pearl that will make a major impact on the pediatric patients you see every day," she promised at the annual meeting of the American Academy of Dermatology.
From her standpoint, the most important thing about topical 5-FU cream for warts in kids is its safety, since this therapy does after all involve off-label use of a potent drug in little children. Reassuringly, in her 39-patient study there were no detectable blood levels of 5-FU during or after 6 weeks of therapy.
From the perspective of the patient and family, however, the big appeal is the therapy's ease and tolerability.
"When you're a dermatologist dealing with kids you really need to look for alternatives to the painful therapies that we currently use. If you have child with extensive warts you really can't be considering painful treatment options such as liquid nitrogen or intralesional bleomycin. That's just not going to cut it, and that family is never going to come back to you again. It’s not humane to expect a child to sit through painful treatments for something that benign," explained Dr. Cunningham.
The 39 children in her study all had at least two hand warts to which topical 5-FU cream was applied once or twice daily under occlusion for 6 weeks. Eighty-eight percent of patients were significantly improved after 6 weeks, and 41% had complete resolution of at least one wart. The treatment response didn't differ between once- and twice-daily therapy. Tolerability and patient satisfaction were excellent. At 6 months of follow-up, 87% of complete responders had no wart recurrences (Pediatr. Dermatol. 2009;26:279-85).
Since publication of her study, Dr. Cunningham has modified how she uses topical 5-FU cream for pediatric warts. The drug is applied at night under duct tape occlusion, alternating with salicylic acid under duct tape occlusion every second night because she believes the two agents are complementary. The duct tape is removed each morning and the wart is left uncovered and untreated during the day.
"I only do this for a month at a time because that's really all I ever need. If this medicine is going to work – and it usually does – it works fast," she said.
The ideal warts for this form of therapy are single and rapidly growing. "The ones that are doubling in size every couple of weeks like they've got a mind of their own, those are the perfect ones to treat with topical 5-FU because 5-FU is going to impair cell division and really shut that wart down," Dr. Cunningham continued.
She offered a few cautionary notes: Don't use this therapy on periungual warts; it will result in serious onycholysis, and the nail will fall off. Avoid treating warts on the face. In order to prevent systemic absorption, don’t use topical 5-FU cream in orally fixated thumb suckers. And make sure to keep the medication out of reach of the family dog. Dogs have a paradoxical toxic reaction to topical 5-FU. There are dozens of reports of dog fatalities in the veterinary literature, Dr. Cunningham said.
She declared having no relevant financial interests.
NEW ORLEANS - Any dermatologist who's not regularly using topical 5% 5-fluorouracil cream for treatment of warts in children is missing out on a therapy that's singularly safe, well tolerated, and effective.
That's the considered opinion of Dr. Bari B. Cunningham, who not only uses this therapy on a daily basis in her Encinitas, Calif., pediatric dermatology practice, but was also senior author of an open-label study demonstrating its benefits.
"I would strongly urge you to consider this therapy. This is a pearl that will make a major impact on the pediatric patients you see every day," she promised at the annual meeting of the American Academy of Dermatology.
From her standpoint, the most important thing about topical 5-FU cream for warts in kids is its safety, since this therapy does after all involve off-label use of a potent drug in little children. Reassuringly, in her 39-patient study there were no detectable blood levels of 5-FU during or after 6 weeks of therapy.
From the perspective of the patient and family, however, the big appeal is the therapy's ease and tolerability.
"When you're a dermatologist dealing with kids you really need to look for alternatives to the painful therapies that we currently use. If you have child with extensive warts you really can't be considering painful treatment options such as liquid nitrogen or intralesional bleomycin. That's just not going to cut it, and that family is never going to come back to you again. It’s not humane to expect a child to sit through painful treatments for something that benign," explained Dr. Cunningham.
The 39 children in her study all had at least two hand warts to which topical 5-FU cream was applied once or twice daily under occlusion for 6 weeks. Eighty-eight percent of patients were significantly improved after 6 weeks, and 41% had complete resolution of at least one wart. The treatment response didn't differ between once- and twice-daily therapy. Tolerability and patient satisfaction were excellent. At 6 months of follow-up, 87% of complete responders had no wart recurrences (Pediatr. Dermatol. 2009;26:279-85).
Since publication of her study, Dr. Cunningham has modified how she uses topical 5-FU cream for pediatric warts. The drug is applied at night under duct tape occlusion, alternating with salicylic acid under duct tape occlusion every second night because she believes the two agents are complementary. The duct tape is removed each morning and the wart is left uncovered and untreated during the day.
"I only do this for a month at a time because that's really all I ever need. If this medicine is going to work – and it usually does – it works fast," she said.
The ideal warts for this form of therapy are single and rapidly growing. "The ones that are doubling in size every couple of weeks like they've got a mind of their own, those are the perfect ones to treat with topical 5-FU because 5-FU is going to impair cell division and really shut that wart down," Dr. Cunningham continued.
She offered a few cautionary notes: Don't use this therapy on periungual warts; it will result in serious onycholysis, and the nail will fall off. Avoid treating warts on the face. In order to prevent systemic absorption, don’t use topical 5-FU cream in orally fixated thumb suckers. And make sure to keep the medication out of reach of the family dog. Dogs have a paradoxical toxic reaction to topical 5-FU. There are dozens of reports of dog fatalities in the veterinary literature, Dr. Cunningham said.
She declared having no relevant financial interests.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF DERMATOLOGY
All Hair Is Not the Same
Efficacy and Safety Study of Tazarotene Cream 0.1% for the Treatment of Brittle Nail Syndrome
Intraocular Choristoma, Anterior Staphyloma With Ipsilateral Nevus Sebaceus, and Congenital Giant Hairy Nevus: A Case Report
FDA Approves New Head Lice Treatment
The Food and Drug Administration has approved a new topical treatment for head lice in children and adults.
Natroba Topical Suspension (spinosad 0.9%), proved more effective than Nix (permethrin 1%) when compared directly in clinical trials. In two trials involving a total of 1,038 children and adults, after one or two applications of spinosad, 85% and 87% of patients were lice free, compared with 45% and 43% of patients receiving permethrin (Pediatrics 2009;124:e389-95).
Another advantage of spinosad, according to its labeling, is that it doesn’t require combing to be effective. A single, 10-minute application of spinosad is followed by a warm-water rinse. If desired, a fine-toothed comb can be used to remove dead lice and nits from the hair. A second application is permissible if the patient continues to harbor live lice 7 days later.
Investigators noted no serious adverse events in the trials, and only a small number of mild to moderate adverse events. The most common were application site erythema (seen in 6.8% of the patients given spinosad), ocular hyperemia (in 3.3% of patients), and application site irritation (in 1.5% of patients).
Spinosad works by causing neuronal excitation in insects. After a period of hyperexcitation, lice become paralyzed and die.
The FDA approval covered the use of spinosad in adults and children aged 4 years and older. The agency said that it is important not to use spinosad in infants younger than age 6 months. The product contains benzyl alcohol, which can cause serious reactions and even death in infants.
The clinical trials reported in Pediatrics were sponsored by ParaPRO, the manufacturer of Natroba. Two of the coauthors received research funding from ParaPRO, and two others served as consultants to the company.
The Food and Drug Administration has approved a new topical treatment for head lice in children and adults.
Natroba Topical Suspension (spinosad 0.9%), proved more effective than Nix (permethrin 1%) when compared directly in clinical trials. In two trials involving a total of 1,038 children and adults, after one or two applications of spinosad, 85% and 87% of patients were lice free, compared with 45% and 43% of patients receiving permethrin (Pediatrics 2009;124:e389-95).
Another advantage of spinosad, according to its labeling, is that it doesn’t require combing to be effective. A single, 10-minute application of spinosad is followed by a warm-water rinse. If desired, a fine-toothed comb can be used to remove dead lice and nits from the hair. A second application is permissible if the patient continues to harbor live lice 7 days later.
Investigators noted no serious adverse events in the trials, and only a small number of mild to moderate adverse events. The most common were application site erythema (seen in 6.8% of the patients given spinosad), ocular hyperemia (in 3.3% of patients), and application site irritation (in 1.5% of patients).
Spinosad works by causing neuronal excitation in insects. After a period of hyperexcitation, lice become paralyzed and die.
The FDA approval covered the use of spinosad in adults and children aged 4 years and older. The agency said that it is important not to use spinosad in infants younger than age 6 months. The product contains benzyl alcohol, which can cause serious reactions and even death in infants.
The clinical trials reported in Pediatrics were sponsored by ParaPRO, the manufacturer of Natroba. Two of the coauthors received research funding from ParaPRO, and two others served as consultants to the company.
The Food and Drug Administration has approved a new topical treatment for head lice in children and adults.
Natroba Topical Suspension (spinosad 0.9%), proved more effective than Nix (permethrin 1%) when compared directly in clinical trials. In two trials involving a total of 1,038 children and adults, after one or two applications of spinosad, 85% and 87% of patients were lice free, compared with 45% and 43% of patients receiving permethrin (Pediatrics 2009;124:e389-95).
Another advantage of spinosad, according to its labeling, is that it doesn’t require combing to be effective. A single, 10-minute application of spinosad is followed by a warm-water rinse. If desired, a fine-toothed comb can be used to remove dead lice and nits from the hair. A second application is permissible if the patient continues to harbor live lice 7 days later.
Investigators noted no serious adverse events in the trials, and only a small number of mild to moderate adverse events. The most common were application site erythema (seen in 6.8% of the patients given spinosad), ocular hyperemia (in 3.3% of patients), and application site irritation (in 1.5% of patients).
Spinosad works by causing neuronal excitation in insects. After a period of hyperexcitation, lice become paralyzed and die.
The FDA approval covered the use of spinosad in adults and children aged 4 years and older. The agency said that it is important not to use spinosad in infants younger than age 6 months. The product contains benzyl alcohol, which can cause serious reactions and even death in infants.
The clinical trials reported in Pediatrics were sponsored by ParaPRO, the manufacturer of Natroba. Two of the coauthors received research funding from ParaPRO, and two others served as consultants to the company.
FROM THE FOOD AND DRUG ADMINISTRATION