User login
Passport to personality: Is it the patient who's 'difficult'?
You know that you’ve been at a valuable meeting when you can use on Monday morning the "take-home messages" you gathered over the weekend.
The annual conference of the American Psychosocial Oncology Society was just such a meeting, filled with valuable data and perspective on distress screening tools, survivorship guidance, and ways to integrate psychosocial care comfortably into the evolving medical model of cancer care.
But the lessons I put into play before I unpacked my briefcase were gleaned in a dynamic workshop led by Dr. John D. Wynn of the Swedish Cancer Institute in Seattle.
A psychiatrist, Dr. Wynn has long been regarded as an expert on the uneasy intersection of cancer and Axis II personality traits and disorders, having written the chapter on this topic in Dr. Jimmie Holland’s classic textbook Psycho-Oncology (New York: Oxford University Press, 2010).
Sorting through Axis II diagnoses, he frames Clusters A, B, and C as "weird," "wild," and "worried." Along the way, he challenged the accepted dogma that personality disorders are easily defined, enduring, and inflexible when research calls into question all of these notions.
To be sure, there are individuals whose ways of dealing with others and the world fall far outside the reassuring central band of "average." But labels – formal or informal –may be overused and unhelpful, he argued.
For example, "uncooperative" and "difficult" aren’t assigned codes in the DSM-IV or ICD-9, but they’re quasi-diagnoses used with great frequency in rounds, case conferences, and at the nursing station.
Dr. Wynn drove home the point that such terms are relative. Members of the treatment team are likely viewed as "difficult" by the very same patients who elicit the label from us. Consider the context, he counseled.
"Medical care encourages and reinforces dependency, passivity, and attention seeking," he explained.
While most people in society are able to adapt to the highly structured culture of Western medicine, grinning and bearing it in their new costumes (cotton gowns) and subservient patient roles, others may simply not possess that flexibility, particularly in the context of extreme fear, embarrassment, or mistrust.
Dr. Wynn argues that the people who are often diagnosed with personality disorders really have "adaptive failure" – a term he wishes would have made it into DSM-5. They have both a poor sense of self and a poor capacity for interpersonal functioning.
And this is a painful combination when it comes to coping with a life-threatening diagnosis in a strange new world, where amicability, conformity, and allegiance to the treatment plan are highly prized.
So how does all this play out in the clinic?
Dr. Wynn’s advice is to use our own reactions to such patients as diagnostic cues, tapping into our professionalism to guide our responses.
See each patient as unique and uniquely challenged by the circumstances of his or her disease, and respond based on what you see and feel, he advised.
Take extra time to listen to patients who fail to follow through with treatment, appear needy or overly-entitled, and even those who may "frighten or repel us."
Do fear, shame, or confusion lie at the heart of such behavior?
Could respect, collaboration, and limit setting put in check the most troubling interactions with such patients?
With Dr. Wynn’s conceptualization fresh in my mind, I was inspired to step back and put into context my own reactions to patients I find difficult.
I found myself more patient, more understanding, and more curious about the underpinnings of behaviors that play havoc with the treatment schedule and have the potential to bring out the worst in us all.
What particularly helps for me is to remember (and remind colleagues) that our shared enemy is cancer, not the patient. By seeing the patient as our ally in that fight, we are motivated to compassionately demystify our traditions and find creative bridges to connection with all types of people who need our care.
Dr. Freed is a psychologist in Santa Barbara, Calif., and a medical journalist.
You know that you’ve been at a valuable meeting when you can use on Monday morning the "take-home messages" you gathered over the weekend.
The annual conference of the American Psychosocial Oncology Society was just such a meeting, filled with valuable data and perspective on distress screening tools, survivorship guidance, and ways to integrate psychosocial care comfortably into the evolving medical model of cancer care.
But the lessons I put into play before I unpacked my briefcase were gleaned in a dynamic workshop led by Dr. John D. Wynn of the Swedish Cancer Institute in Seattle.
A psychiatrist, Dr. Wynn has long been regarded as an expert on the uneasy intersection of cancer and Axis II personality traits and disorders, having written the chapter on this topic in Dr. Jimmie Holland’s classic textbook Psycho-Oncology (New York: Oxford University Press, 2010).
Sorting through Axis II diagnoses, he frames Clusters A, B, and C as "weird," "wild," and "worried." Along the way, he challenged the accepted dogma that personality disorders are easily defined, enduring, and inflexible when research calls into question all of these notions.
To be sure, there are individuals whose ways of dealing with others and the world fall far outside the reassuring central band of "average." But labels – formal or informal –may be overused and unhelpful, he argued.
For example, "uncooperative" and "difficult" aren’t assigned codes in the DSM-IV or ICD-9, but they’re quasi-diagnoses used with great frequency in rounds, case conferences, and at the nursing station.
Dr. Wynn drove home the point that such terms are relative. Members of the treatment team are likely viewed as "difficult" by the very same patients who elicit the label from us. Consider the context, he counseled.
"Medical care encourages and reinforces dependency, passivity, and attention seeking," he explained.
While most people in society are able to adapt to the highly structured culture of Western medicine, grinning and bearing it in their new costumes (cotton gowns) and subservient patient roles, others may simply not possess that flexibility, particularly in the context of extreme fear, embarrassment, or mistrust.
Dr. Wynn argues that the people who are often diagnosed with personality disorders really have "adaptive failure" – a term he wishes would have made it into DSM-5. They have both a poor sense of self and a poor capacity for interpersonal functioning.
And this is a painful combination when it comes to coping with a life-threatening diagnosis in a strange new world, where amicability, conformity, and allegiance to the treatment plan are highly prized.
So how does all this play out in the clinic?
Dr. Wynn’s advice is to use our own reactions to such patients as diagnostic cues, tapping into our professionalism to guide our responses.
See each patient as unique and uniquely challenged by the circumstances of his or her disease, and respond based on what you see and feel, he advised.
Take extra time to listen to patients who fail to follow through with treatment, appear needy or overly-entitled, and even those who may "frighten or repel us."
Do fear, shame, or confusion lie at the heart of such behavior?
Could respect, collaboration, and limit setting put in check the most troubling interactions with such patients?
With Dr. Wynn’s conceptualization fresh in my mind, I was inspired to step back and put into context my own reactions to patients I find difficult.
I found myself more patient, more understanding, and more curious about the underpinnings of behaviors that play havoc with the treatment schedule and have the potential to bring out the worst in us all.
What particularly helps for me is to remember (and remind colleagues) that our shared enemy is cancer, not the patient. By seeing the patient as our ally in that fight, we are motivated to compassionately demystify our traditions and find creative bridges to connection with all types of people who need our care.
Dr. Freed is a psychologist in Santa Barbara, Calif., and a medical journalist.
You know that you’ve been at a valuable meeting when you can use on Monday morning the "take-home messages" you gathered over the weekend.
The annual conference of the American Psychosocial Oncology Society was just such a meeting, filled with valuable data and perspective on distress screening tools, survivorship guidance, and ways to integrate psychosocial care comfortably into the evolving medical model of cancer care.
But the lessons I put into play before I unpacked my briefcase were gleaned in a dynamic workshop led by Dr. John D. Wynn of the Swedish Cancer Institute in Seattle.
A psychiatrist, Dr. Wynn has long been regarded as an expert on the uneasy intersection of cancer and Axis II personality traits and disorders, having written the chapter on this topic in Dr. Jimmie Holland’s classic textbook Psycho-Oncology (New York: Oxford University Press, 2010).
Sorting through Axis II diagnoses, he frames Clusters A, B, and C as "weird," "wild," and "worried." Along the way, he challenged the accepted dogma that personality disorders are easily defined, enduring, and inflexible when research calls into question all of these notions.
To be sure, there are individuals whose ways of dealing with others and the world fall far outside the reassuring central band of "average." But labels – formal or informal –may be overused and unhelpful, he argued.
For example, "uncooperative" and "difficult" aren’t assigned codes in the DSM-IV or ICD-9, but they’re quasi-diagnoses used with great frequency in rounds, case conferences, and at the nursing station.
Dr. Wynn drove home the point that such terms are relative. Members of the treatment team are likely viewed as "difficult" by the very same patients who elicit the label from us. Consider the context, he counseled.
"Medical care encourages and reinforces dependency, passivity, and attention seeking," he explained.
While most people in society are able to adapt to the highly structured culture of Western medicine, grinning and bearing it in their new costumes (cotton gowns) and subservient patient roles, others may simply not possess that flexibility, particularly in the context of extreme fear, embarrassment, or mistrust.
Dr. Wynn argues that the people who are often diagnosed with personality disorders really have "adaptive failure" – a term he wishes would have made it into DSM-5. They have both a poor sense of self and a poor capacity for interpersonal functioning.
And this is a painful combination when it comes to coping with a life-threatening diagnosis in a strange new world, where amicability, conformity, and allegiance to the treatment plan are highly prized.
So how does all this play out in the clinic?
Dr. Wynn’s advice is to use our own reactions to such patients as diagnostic cues, tapping into our professionalism to guide our responses.
See each patient as unique and uniquely challenged by the circumstances of his or her disease, and respond based on what you see and feel, he advised.
Take extra time to listen to patients who fail to follow through with treatment, appear needy or overly-entitled, and even those who may "frighten or repel us."
Do fear, shame, or confusion lie at the heart of such behavior?
Could respect, collaboration, and limit setting put in check the most troubling interactions with such patients?
With Dr. Wynn’s conceptualization fresh in my mind, I was inspired to step back and put into context my own reactions to patients I find difficult.
I found myself more patient, more understanding, and more curious about the underpinnings of behaviors that play havoc with the treatment schedule and have the potential to bring out the worst in us all.
What particularly helps for me is to remember (and remind colleagues) that our shared enemy is cancer, not the patient. By seeing the patient as our ally in that fight, we are motivated to compassionately demystify our traditions and find creative bridges to connection with all types of people who need our care.
Dr. Freed is a psychologist in Santa Barbara, Calif., and a medical journalist.
End-of-life hypoactive delirium responds to antipsychotics
LAS VEGAS – Hypoactive delirium is common in terminally ill patients; just as disconcerting as hyperactive, agitated delirium; and just as likely to respond to antipsychotics, according to Dr. William Breitbart, chief of the psychiatry service at Memorial Sloan-Kettering Cancer Center, New York.
Outwardly, patients with hypoactive delirium appear less distressed than do patients with hyperactive delirium, yet they report just as much distress and about half of them report delusions and hallucinations.
About 80% of terminal cancer patients become delirious in their final weeks of life, and about half of them experience hypoactive delirium in association with opioid analgesia, metabolic derangements, and organ failure.
Addressing hypoactive delirium requires diplomacy, especially in patients with hallucinations, he said. Dying patients often see dead relatives – parents, for instance, who come to accompany them to the afterlife. "For me to barrel into the room and say those are psychotic symptoms or describe these experiences as absurd would be insensitive; they are profoundly meaningful experiences to patients and families," Dr. Breitbart said at the annual psychopharmacology update held by the Nevada Psychiatric Association. Instead, he acknowledges the significance of the visions, but notes that they can rapidly change to very frightening visions. He then discusses the use of medications to avert this possible progression.
Antipsychotics help to reduce agitation, psychotic symptoms, and more subtle problems such as disorientation in these patients, Dr. Breitbart said at the conference. When the underlying metabolic problems can be addressed in cancer patients, the drugs can be used for a few weeks and this sort of short-term use is not associated with increased mortality, he said. The ultimate goal of treatment is an "upright patient who is awake, alert, and conversant with family and staff."
In the patient with imminent terminal illness, however, sometimes "the best you can achieve (with antipsychotics) is a sedated patient," he said.
Low-dose haloperidol is generally preferred; atypical antipsychotics have not proven any more effective or safer. Close monitoring for side effects – particularly prolonged QTc intervals and extrapyramidal symptoms – is essential (J. Clin. Oncol. 2012;30:1206-14).
Haloperidol and chlorpromazine work equally well in both types of delirium. Dr. Breitbart said he’ll sometimes add chlorpromazine when agitation fails to respond to haloperidol alone.
Aripiprazole may work a bit better in hypoactive patients; olanzapine may be preferred in hyperactive patients. Risperidone is another option.
"We tend not to use benzodiazepines because they worsen delirium, but if your goal is to sedate a patient, then benzodiazepines are helpful. We often use haloperidol in combination with a drug like lorazepam to help get an agitated patient under better control," he said.
Dr. Breitbart said he has no relevant financial relationships with commercial interests.
LAS VEGAS – Hypoactive delirium is common in terminally ill patients; just as disconcerting as hyperactive, agitated delirium; and just as likely to respond to antipsychotics, according to Dr. William Breitbart, chief of the psychiatry service at Memorial Sloan-Kettering Cancer Center, New York.
Outwardly, patients with hypoactive delirium appear less distressed than do patients with hyperactive delirium, yet they report just as much distress and about half of them report delusions and hallucinations.
About 80% of terminal cancer patients become delirious in their final weeks of life, and about half of them experience hypoactive delirium in association with opioid analgesia, metabolic derangements, and organ failure.
Addressing hypoactive delirium requires diplomacy, especially in patients with hallucinations, he said. Dying patients often see dead relatives – parents, for instance, who come to accompany them to the afterlife. "For me to barrel into the room and say those are psychotic symptoms or describe these experiences as absurd would be insensitive; they are profoundly meaningful experiences to patients and families," Dr. Breitbart said at the annual psychopharmacology update held by the Nevada Psychiatric Association. Instead, he acknowledges the significance of the visions, but notes that they can rapidly change to very frightening visions. He then discusses the use of medications to avert this possible progression.
Antipsychotics help to reduce agitation, psychotic symptoms, and more subtle problems such as disorientation in these patients, Dr. Breitbart said at the conference. When the underlying metabolic problems can be addressed in cancer patients, the drugs can be used for a few weeks and this sort of short-term use is not associated with increased mortality, he said. The ultimate goal of treatment is an "upright patient who is awake, alert, and conversant with family and staff."
In the patient with imminent terminal illness, however, sometimes "the best you can achieve (with antipsychotics) is a sedated patient," he said.
Low-dose haloperidol is generally preferred; atypical antipsychotics have not proven any more effective or safer. Close monitoring for side effects – particularly prolonged QTc intervals and extrapyramidal symptoms – is essential (J. Clin. Oncol. 2012;30:1206-14).
Haloperidol and chlorpromazine work equally well in both types of delirium. Dr. Breitbart said he’ll sometimes add chlorpromazine when agitation fails to respond to haloperidol alone.
Aripiprazole may work a bit better in hypoactive patients; olanzapine may be preferred in hyperactive patients. Risperidone is another option.
"We tend not to use benzodiazepines because they worsen delirium, but if your goal is to sedate a patient, then benzodiazepines are helpful. We often use haloperidol in combination with a drug like lorazepam to help get an agitated patient under better control," he said.
Dr. Breitbart said he has no relevant financial relationships with commercial interests.
LAS VEGAS – Hypoactive delirium is common in terminally ill patients; just as disconcerting as hyperactive, agitated delirium; and just as likely to respond to antipsychotics, according to Dr. William Breitbart, chief of the psychiatry service at Memorial Sloan-Kettering Cancer Center, New York.
Outwardly, patients with hypoactive delirium appear less distressed than do patients with hyperactive delirium, yet they report just as much distress and about half of them report delusions and hallucinations.
About 80% of terminal cancer patients become delirious in their final weeks of life, and about half of them experience hypoactive delirium in association with opioid analgesia, metabolic derangements, and organ failure.
Addressing hypoactive delirium requires diplomacy, especially in patients with hallucinations, he said. Dying patients often see dead relatives – parents, for instance, who come to accompany them to the afterlife. "For me to barrel into the room and say those are psychotic symptoms or describe these experiences as absurd would be insensitive; they are profoundly meaningful experiences to patients and families," Dr. Breitbart said at the annual psychopharmacology update held by the Nevada Psychiatric Association. Instead, he acknowledges the significance of the visions, but notes that they can rapidly change to very frightening visions. He then discusses the use of medications to avert this possible progression.
Antipsychotics help to reduce agitation, psychotic symptoms, and more subtle problems such as disorientation in these patients, Dr. Breitbart said at the conference. When the underlying metabolic problems can be addressed in cancer patients, the drugs can be used for a few weeks and this sort of short-term use is not associated with increased mortality, he said. The ultimate goal of treatment is an "upright patient who is awake, alert, and conversant with family and staff."
In the patient with imminent terminal illness, however, sometimes "the best you can achieve (with antipsychotics) is a sedated patient," he said.
Low-dose haloperidol is generally preferred; atypical antipsychotics have not proven any more effective or safer. Close monitoring for side effects – particularly prolonged QTc intervals and extrapyramidal symptoms – is essential (J. Clin. Oncol. 2012;30:1206-14).
Haloperidol and chlorpromazine work equally well in both types of delirium. Dr. Breitbart said he’ll sometimes add chlorpromazine when agitation fails to respond to haloperidol alone.
Aripiprazole may work a bit better in hypoactive patients; olanzapine may be preferred in hyperactive patients. Risperidone is another option.
"We tend not to use benzodiazepines because they worsen delirium, but if your goal is to sedate a patient, then benzodiazepines are helpful. We often use haloperidol in combination with a drug like lorazepam to help get an agitated patient under better control," he said.
Dr. Breitbart said he has no relevant financial relationships with commercial interests.
AT NPA ANNUAL PSYCHOPHARMACOLOGY CONFERENCE
Harness side effects when selecting psychotropics for cancer patients
LAS VEGAS – When it comes to treating depression in end-stage cancer patients, the side effects of psychotropics can prove almost as helpful as their primary indications.
"We tend to select drugs based on the side effects we want to utilize," said Dr. William S. Breitbart, chief of the Memorial Sloan-Kettering Cancer Center Psychiatry Service in New York.
For instance, sedating, constipating medicines such as tricyclics would be work well for a depressed patient with insomnia, diarrhea, and mouth sores. However, it would make sense to avoid those side effects in a patient with constipation or one who sleeps too much because of opioid pain management, he said.
Tricyclics also have some pain-fighting ability, and antidepressants in general help with dyspnea and can, along with neuroleptics, counter steroid side effects.
Drugs such as venlafaxine, mirtazapine, citalopram, and escitalopram are favored for terminally ill cancer patients, when possible, because they have little effect on the cytochrome P450 system, and so are unlikely to interfere with the blood levels of chemotherapeutic agents such as tamoxifen, Dr. Breitbart said at the Nevada Psychiatric Association’s Annual Psychopharmacology Update conference.
In addition, excitement in psychiatry about ketamine as an antidepressant has also been extended to patients in palliative care settings. However, Dr. Breitbart said, so far, there has not been enough controlled trials to support the use of ketamine with these terminal patients.
The side effects of other psychotropics are useful, too. Mirtazapine can help patients gain weight; olanzapine can do that, has analgesic properties, and helps with nausea and vomiting. Fitful sleep can be calmed by various agents with sedating properties.
Pain, metabolic abnormalities, anemia, adrenal insufficiency, hypogonadism, and chemotherapy agents themselves are among the factors that can contribute to depression in cancer patients. Patients with pancreatic cancer have higher rates of depression than do those with other intra-abdominal or retroperitoneal cancers, as well, perhaps because they have high levels of endogenous proinflammatory cytokines, which have been linked to depression.
About 60% of patients will respond to antidepressant therapy; individual and group psychotherapy, music and relaxation therapy, and social support help, too.
Psychostimulants help improve mood, cognition, and concentration, as well. "We occasionally use combinations of psychostimulants with [selective serotonin reuptake inhibitors] to jump-start the effect of the antidepressant agent." They also potentiate the analgesic effect of opioids and counteract their sedation, "so you can achieve analgesia at higher [opioid] doses" instead of limiting them because of sedation, Dr. Breitbart said.
Rather then suppressing appetite, psychostimulants give cancer patients a better sense of well-being and improve their appetite, which may help them gain a few pounds.
It’s a challenge to identify depression in advanced cancer, because fatigue, weight loss, and other effects of cancer and its treatment mimic depression. One solution is to increase the threshold for depression diagnoses, requiring a couple more associated symptoms instead of the usual four. Also, just the simple question, "Have you been depressed most of the time for the past 2 weeks" is a great screening tool for advanced cancer patients, he said.
Dr. Breitbart said he has no relevant commercial interests.
LAS VEGAS – When it comes to treating depression in end-stage cancer patients, the side effects of psychotropics can prove almost as helpful as their primary indications.
"We tend to select drugs based on the side effects we want to utilize," said Dr. William S. Breitbart, chief of the Memorial Sloan-Kettering Cancer Center Psychiatry Service in New York.
For instance, sedating, constipating medicines such as tricyclics would be work well for a depressed patient with insomnia, diarrhea, and mouth sores. However, it would make sense to avoid those side effects in a patient with constipation or one who sleeps too much because of opioid pain management, he said.
Tricyclics also have some pain-fighting ability, and antidepressants in general help with dyspnea and can, along with neuroleptics, counter steroid side effects.
Drugs such as venlafaxine, mirtazapine, citalopram, and escitalopram are favored for terminally ill cancer patients, when possible, because they have little effect on the cytochrome P450 system, and so are unlikely to interfere with the blood levels of chemotherapeutic agents such as tamoxifen, Dr. Breitbart said at the Nevada Psychiatric Association’s Annual Psychopharmacology Update conference.
In addition, excitement in psychiatry about ketamine as an antidepressant has also been extended to patients in palliative care settings. However, Dr. Breitbart said, so far, there has not been enough controlled trials to support the use of ketamine with these terminal patients.
The side effects of other psychotropics are useful, too. Mirtazapine can help patients gain weight; olanzapine can do that, has analgesic properties, and helps with nausea and vomiting. Fitful sleep can be calmed by various agents with sedating properties.
Pain, metabolic abnormalities, anemia, adrenal insufficiency, hypogonadism, and chemotherapy agents themselves are among the factors that can contribute to depression in cancer patients. Patients with pancreatic cancer have higher rates of depression than do those with other intra-abdominal or retroperitoneal cancers, as well, perhaps because they have high levels of endogenous proinflammatory cytokines, which have been linked to depression.
About 60% of patients will respond to antidepressant therapy; individual and group psychotherapy, music and relaxation therapy, and social support help, too.
Psychostimulants help improve mood, cognition, and concentration, as well. "We occasionally use combinations of psychostimulants with [selective serotonin reuptake inhibitors] to jump-start the effect of the antidepressant agent." They also potentiate the analgesic effect of opioids and counteract their sedation, "so you can achieve analgesia at higher [opioid] doses" instead of limiting them because of sedation, Dr. Breitbart said.
Rather then suppressing appetite, psychostimulants give cancer patients a better sense of well-being and improve their appetite, which may help them gain a few pounds.
It’s a challenge to identify depression in advanced cancer, because fatigue, weight loss, and other effects of cancer and its treatment mimic depression. One solution is to increase the threshold for depression diagnoses, requiring a couple more associated symptoms instead of the usual four. Also, just the simple question, "Have you been depressed most of the time for the past 2 weeks" is a great screening tool for advanced cancer patients, he said.
Dr. Breitbart said he has no relevant commercial interests.
LAS VEGAS – When it comes to treating depression in end-stage cancer patients, the side effects of psychotropics can prove almost as helpful as their primary indications.
"We tend to select drugs based on the side effects we want to utilize," said Dr. William S. Breitbart, chief of the Memorial Sloan-Kettering Cancer Center Psychiatry Service in New York.
For instance, sedating, constipating medicines such as tricyclics would be work well for a depressed patient with insomnia, diarrhea, and mouth sores. However, it would make sense to avoid those side effects in a patient with constipation or one who sleeps too much because of opioid pain management, he said.
Tricyclics also have some pain-fighting ability, and antidepressants in general help with dyspnea and can, along with neuroleptics, counter steroid side effects.
Drugs such as venlafaxine, mirtazapine, citalopram, and escitalopram are favored for terminally ill cancer patients, when possible, because they have little effect on the cytochrome P450 system, and so are unlikely to interfere with the blood levels of chemotherapeutic agents such as tamoxifen, Dr. Breitbart said at the Nevada Psychiatric Association’s Annual Psychopharmacology Update conference.
In addition, excitement in psychiatry about ketamine as an antidepressant has also been extended to patients in palliative care settings. However, Dr. Breitbart said, so far, there has not been enough controlled trials to support the use of ketamine with these terminal patients.
The side effects of other psychotropics are useful, too. Mirtazapine can help patients gain weight; olanzapine can do that, has analgesic properties, and helps with nausea and vomiting. Fitful sleep can be calmed by various agents with sedating properties.
Pain, metabolic abnormalities, anemia, adrenal insufficiency, hypogonadism, and chemotherapy agents themselves are among the factors that can contribute to depression in cancer patients. Patients with pancreatic cancer have higher rates of depression than do those with other intra-abdominal or retroperitoneal cancers, as well, perhaps because they have high levels of endogenous proinflammatory cytokines, which have been linked to depression.
About 60% of patients will respond to antidepressant therapy; individual and group psychotherapy, music and relaxation therapy, and social support help, too.
Psychostimulants help improve mood, cognition, and concentration, as well. "We occasionally use combinations of psychostimulants with [selective serotonin reuptake inhibitors] to jump-start the effect of the antidepressant agent." They also potentiate the analgesic effect of opioids and counteract their sedation, "so you can achieve analgesia at higher [opioid] doses" instead of limiting them because of sedation, Dr. Breitbart said.
Rather then suppressing appetite, psychostimulants give cancer patients a better sense of well-being and improve their appetite, which may help them gain a few pounds.
It’s a challenge to identify depression in advanced cancer, because fatigue, weight loss, and other effects of cancer and its treatment mimic depression. One solution is to increase the threshold for depression diagnoses, requiring a couple more associated symptoms instead of the usual four. Also, just the simple question, "Have you been depressed most of the time for the past 2 weeks" is a great screening tool for advanced cancer patients, he said.
Dr. Breitbart said he has no relevant commercial interests.
FROM THE NPA ANNUAL PSYCHO-PHARMACOLOGY CONFERENCE
Frailty in older adults: Implications for end-of-life care
As people get older, they have more things wrong with them. And the more things they have wrong with them, the more likely they are to die. But everyone accumulates deficits at a different rate, and not all people of the same age have the same short-term risk of dying. This variable susceptibility to death and other adverse outcomes in older people of the same age is called frailty.1
Frailty poses special challenges to how we organize and deliver health care. These challenges are sometimes seen most starkly when people are most frail, especially as they approach the end of life.
In this paper, we will review how frailty is conceptualized and defined, consider how frailty affects the care of people at the end of their lives, and suggest practices that can make end-of-life care better for frail older adults.
DEFINING FRAILTY
As with all complex systems, when frail people become acutely unwell their highest-order functions fail first. Thus, cognitive impairment, functional decline, impaired mobility, and social withdrawal are hallmark presentations of the further accumulation of deficits in vulnerable seniors.
Delirium and falls are important clues that a person’s resilience is becoming compromised and that the person is at risk of further insults in a downward spiral or acceleration of things going wrong.1,2 Frailty is associated with poor health outcomes, from disability to institutionalization and death.3
This idea of frailty as vulnerability arising from dysregulation of multiple physiologic systems is reasonably non-controversial. Even so, there are competing views on how to systematically quantify those who are at an increased risk of adverse sequelae.
Quantifying frailty is particularly important if it can tell us if a patient is at high risk of further decline and death. As frailty advances, it is appropriate to shift the focus of care to palliation, with the goal of optimizing quality of life and easing symptoms.4 Identifying someone as frail can aid decision-making in the setting of critical illness, where the system commonly defaults to an “always do everything” mode without considering the ramifications of such an approach. Furthermore, without a routine means of measuring frailty, it is often left to critical care units or rapid-response medical teams to initiate a discussion about whether an aggressive course of care is appropriate or desired.5,6
Frailty as a syndrome
Fried et al7 defined frailty as a syndrome arising from the “physiologic triad” of sarcopenia and immune and neuroendocrine dysregulation. Patients are considered frail if they have three or more of the following five criteria:
- Reduced activity
- Slowing of mobility
- Weight loss
- Diminished handgrip strength
- Exhaustion.
Someone who has only one or two of these items is said to be “pre-frail”; someone with none is said to be “robust.”
The frailty index
An alternative viewpoint is that frailty is a state arising from the accumulation of deficits, which can be counted in a frailty index.
The frailty index is based on the concept that frailty is a consequence of interacting physical, psychological, and social factors. As deficits accumulate, people become increasingly vulnerable to adverse outcomes.
The frailty index is calculated as the number of deficits the patient has, divided by the number of deficits considered. For example, in a frailty index based on a comprehensive geriatric assessment, an individual with impairments in 4 of 10 domains and with 10 of 24 possible comorbidities would have 14 of 34 possible deficits, for a frailty index of 0.41.8
A criticism of the frailty index is that it includes functional dependence as a deficit. The criticism stems from the view that frailty should be seen as occurring prior to disability. According to this view, including dependence in instrumental and basic activities of daily living as a deficit confuses disability with frailty.
Proponents of the frailty index counter that frailty is not “all or none” and needs to be graded. The frailty index can distinguish between people with and without disability by means of the number of deficits that they have, which is most important. For example, a person disabled by a paraplegic injury would have a lower frailty index score and therefore would be considered less frail than a person with advanced cancer affecting multiple body systems. (This is assuming the person who has suffered the injury resulting in paraplegia doesn’t have a concomitant condition such as renal failure or heart disease. In the absence of other health insults, such patients are less at risk of further morbidity or death than the patient with advanced cancer until they get another health insult or insults added to their frailty.)
In any case, functional capacity is fundamental in medical decision-making and when estimating prognoses. An example is the use of the Eastern Cooperative Oncology Group’s functional status measure.9,10
Sum of physical and psychological stressors
Consensus is growing for the concept that frail people are made more vulnerable by the combination of both physical and psychological stressors. This is particularly important to bear in mind for patients who may appear physically robust but whose total health burden makes them vulnerable to further insults.
For example, think of a relatively young overweight patient with hypertension, diabetes, dyslipidemia, and ischemic white matter changes (which can manifest as low mood and even mild vascular cognitive impairment). In such a patient, an acute illness could result in cognitive and functional decline that can be permanent.
Balance of assets and deficits
About 20 years ago, we used the metaphor of a balance beam to describe how frailty comes about in older adults. In this view, there is an interplay of physiological and functional health determinants. Assets such as health, resources, and caregivers are balanced against deficits such as illnesses, dependency on others, and support burden.8
For the most part, later concepts of frailty have focused on the individual, with social factors construed separately as social vulnerability.11
Tools for assessing frailty in people who are not yet disabled
Several tools exist to clinically assess frailty in people who are not yet disabled.
The FRAIL scale.12 The Geriatric Advisory Panel of the International Academy of Nutrition and Aging formulated a scale for measuring frailty as a “pre-disability state.” The FRAIL scale consists of five easily remembered items:
- Fatigue
- Resistance (inability to climb one flight of stairs)
- Ambulation (inability to walk one block)
- Illnesses (more than five)
- Loss of weight (> 5%).
Like the “reduced activity” criterion of the frailty syndrome mentioned above (in practical terms, described as the inability to do heavy household chores),13 the FRAIL scale seems to blur the distinction between disability (here, the inability to climb stairs or to walk a block) and “pre-disability,” to an uncertain end. It also seems to blend the notion of a state and a syndrome; these points will need to be clarified in due course.
The Tilburg Frailty Indicator14 was constructed around the multidimensional viewpoint of frailty, beyond disease or disability state, to identify frail community-dwelling older individuals. The first part of this two-part questionnaire consists of 10 questions on frailty determinants and medical comorbidities, while the second part contains physical, psychological, and social variables strongly associated with frailty, as well as information about disability in walking and balance. Interestingly, although it includes both social and physical factors, it does not include cognition.
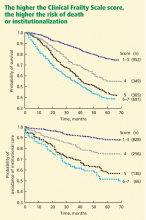
The Clinical Frailty Scale was developed as a practical approach to assess frailty using physical and functional indicators of health and illness burden. The descriptors for this 7-point scale guide clinicians in quantifying the degree of frailty present. It ranges from 1 (very fit) to 7 (severely frail).7 The higher the score, the higher the risks of death or institutionalization. Even mild frailty is associated with a 50% 5-year mortality rate in community-dwelling older adults (Figure 1).8
The Edmonton Frail Scale,15 like the Clinical Frailty Scale, was developed to be practical and usable at the bedside. It is based on the following domains: cognition, general health status, functional independence, social support, medication use, nutrition, mood, continence, and functional performance.
In a community-based sample, the Edmonton Frail Scale compared favorably with the clinical assessment of geriatric specialists who completed a comprehensive evaluation (Pearson’s correlation coefficient 0.64, P < .001).15
FRAILTY AS A PROGNOSTIC INDICATOR
Using frailty scales to aid in prognostication can be useful to clinicians. Survival prognostication is inherently challenging in individuals with multiple comorbidities and variable trajectories of decline, but it remains a vital clinical skill for all clinicians. Framing these difficult discussions in the context of degree of frailty provides a unifying concept, beyond a single-system construct, for care providers, patients, and their loved ones.
Patients nearing the end of their lives need this kind of clarity and support. Regardless of their diagnoses, patients typically want to know when they are at high risk of dying, as do their families and caregivers. People in general look for such information so that they can align medical decision-making congruently with predicted prognosis.16,17 They also use it to plan for the final chapter of their life and their death.
The frailty index is strongly correlated with risk of death
The frailty index is strongly correlated with the risk of death, with a correlation coefficient greater than 0.95. As such, an individual’s frailty index score is considered an estimate of biologic age, which has greater correlation with associated morbidity and death than does chronological age.18,19 In the general population, more than 99% of people have a frailty index value of less than 0.7. As people approach this value, the chance of survival is greatly diminished; indeed, one report suggested that of those who have a frailty index value of more than 0.5 (based on a comprehensive geriatric assessment), 100% are dead by about 20 months later.20,21
In short, there is a limit to which deficits can be added before the system fails. In this sense, the frailty index is akin to the concept of physiologic reserve. Reserve is finite, and as a system loses redundancy it can no longer survive new stresses.
What does this information mean for individual patients?
Even so, prognostication for individual patients remains probabilistic. Any patient has a chance to improve, stabilize, worsen, or die. However, a patient can reach an upper limit of frailty. At that point, instead of accumulating another deficit, death is much more likely. Similarly, although improvement can happen, the chance of improvement is low, and the improvement is typically modest.
Framing survival possibilities in terms of the number of things that people have wrong with them and the chance of death or of change (and the extent of change) makes sense to physicians, patients, and families. Being able to do so offers a much greater opportunity for realistic discussions of the likely outcomes of medical care than the foreseeable scenario of a junior doctor asking a senior citizen, “If your heart stops, do you want us to save your life?”
Understanding prognosis in the face of not just disease but also frailty can also help us focus not on disease but on health consequences of illness. Can the person think? Walk? Care for herself or himself? Interact with others? These questions need to be considered when end-of-life decisions are being discussed.22
Since making predictions about survival is most challenging when multiple comorbidities are present, using the concept of accumulating deficits to better define the slope of decline can be very helpful when discussing “the road ahead” with patients and their families. Visually mapping out the slope of decline and how it is accelerating as conditions progress and deficits accumulate can aid in medical decision-making. Looking individually at the deficits themselves and associated markers of progression can also help with prognostic discussions.
For example, a patient with chronic obstructive pulmonary disease could very well be unaware of the progression and ultimately terminal prognosis of this disease. The slope of clinical decline can be initially shallow, with saw-tooth fluctuations from acute exacerbations that seemingly “resolve to baseline” when antibiotic and steroid courses are completed. Talking with these patients and their families about heralding markers, such as more hospitalizations and cognitive decline with acute exacerbations, can clarify the steepening slope of decline and the way comorbidities interact.
FRAILTY AND END-OF-LIFE CARE
Frailty is progressive, and as it worsens, integrating a palliative approach is appropriate, with a focus on optimizing quality of life and relieving symptoms.4 This principle holds true regardless of the care setting, from acute care hospitals to hospice facilities and long-term care residences.
The principles of end-of-life care are applicable to frail individuals with progressive conditions from the time of diagnosis throughout the course of decline. As the population ages, more people suffer and die from progressive chronic conditions such as cerebrovascular disease, respiratory disease, and dementia.23 An interdisciplinary team approach can ensure all components of palliation are effectively delivered, such as easing symptoms, providing psychosocial and spiritual support, and improving quality of life.24
Pain management
Pain is widely underassessed and undertreated in older patients. Its management at the end of life is particularly challenging if the patient’s language is compromised, as in dementia.23,25,26
A recent cross-sectional analysis of self-reported pain in a longitudinal study of community-dwelling older adults showed an independent association between moderate or higher pain and frailty. The authors propose that persistent pain goes beyond physical discomfort in that it may contribute to homeostenosis (progressive diminishment of homeostatic reserve) and directly worsen frailty.27
Examine the medication list
In palliative care, medical interventions focus on optimizing quality of life.
This especially includes reexamining long medication lists that increase the chance of adverse drug effects.28 Many patients are on disease-modifying medications that may or may not help control symptoms—and might well exacerbate them. For example, betablockers for ischemic heart disease and angiotensin-converting enzyme inhibitors for diabetic nephropathy both can cause hypotension-induced lassitude or even falls due to orthostasis.
A sensible approach is to keep the drugs that may still contribute to quality of life, while discontinuing other drugs that may be causing side effects or that are unlikely to provide meaningful benefit in terms of prevention in patients who have limited life expectancy.29 Discontinuing ineffective, poorly tolerated, and duplicated medications also makes it easier to introduce new medications to manage symptoms—there will be fewer drug interactions, and fewer pills to take, an increasingly important issue in the setting of gastrointestinal symptoms such as dysphagia and gastroparesis or compliance issues as frequently encountered when cognitive impairment is present.29,30
In managing symptoms, start low and go slow—but get there
In managing symptoms in frail elderly patients we use the same classes of medications as in younger patients. The trick is to use appropriate doses.
The concept of “start low and go slow” is key, but so is “get there”—ie, reach the therapeutic goal. The principal drugs for symptom control, such as opioids for pain and dyspnea and anxiolytics for anxiety and restlessness, are associated with a higher rate of and more severe adverse effects in frail older adults.
Even so, most frail older adults appear to be undertreated in this regard, particularly if they are cognitively impaired.23 This fact, coupled with the reality that behavioral symptoms associated with advanced dementia can represent unmet care needs including undertreated pain, highlights the critical need to control symptoms optimally in frail seniors.
This is particularly relevant for those who can no longer verbally articulate their symptoms. Nonverbal pain scales and vigilant assessment of behavioral signs of pain are paramount skills for clinicians providing palliative care to patients with cognitive impairment. Caregivers and loved ones should be included in the assessment process.26,31
Adjunctive therapies for pain control
Maximizing adjunctive therapies can optimize pain control in this drug-sensitive population. Heat and cold packs to affected areas, acupuncture, massage therapy, and structured exercise regimens are some options that can improve quality of life. Cognitive behavioral therapy may offer coping strategies, provided the patient can participate in this process from a cognitive perspective.26,27 Topical preparations are often well tolerated and may include medicinal ingredients that are helpful without systemic effects, such as anti-inflammatory drugs or analgesics.
Optimal use of nonopioid drugs may help reduce the need for narcotics, particularly in the presence of musculoskeletal pain. An example is acetaminophen in regular doses—we would recommend no more than 3 g per day. Acetaminophen is preferred for older adults rather than nonsteroidal anti-inflammatory drugs, given the potential gastric and cardiovascular side effects of the latter medications.
Antidepressants and anticonvulsants such as gabapentin can also be considered as adjuncts for pain control, particularly in the setting of neuropathic pain, with careful monitoring for tolerance.25
When opioids are used, vigilance for constipation is essential.
Establishing goals of care
Goals of care need to be established incrementally along the course of clinical decline and as early as possible so that palliative support can be promptly implemented as symptoms worsen.32
End-of-life care can still include treatments with curative intent, depending on the overall prognosis and the state of the underlying terminal illness. On the other hand, frail older adults who are subjected to invasive treatments that are unlikely to provide cure, such as Whipple surgery, need special intervention postoperatively if they are not to suffer complications such as persisting delirium and functional decline.33
In this regard, geriatric palliative care is frequently about not “crossing a threshold.” Patients may still be receiving active management and be hospitalized for acute exacerbations of progressing chronic conditions, such as chronic obstructive pulmonary disease and heart failure, while palliative principles are introduced and increasingly become the focus of care.
To align goals of care with frailty burden, it is crucial to quantify frailty and to review the patient’s comorbidities. Particularly when dementia is present, lack of communication between the patient’s doctors or between the doctor and the family about disease burden can lead to inappropriately aggressive care.
Many family members and even clinicians do not recognize that advanced dementia is terminal.34,35 In light of this, a palliative approach to care may not even be considered as an appropriate plan when hallmark complications associated with progressing cognitive decline occur, such as aspiration pneumonia or dehydration. Education about dementia and other conditions with progressive organ failure should be done as soon as possible after diagnosis and readdressed at intervals throughout the patient’s clinical decline.
Earlier discussions also ensure that patients themselves can be involved in decision-making more often before cognitive impairment advances to the point where proxy discussions take over.16
BETTER PALLIATIVE CARE FOR ALL
Palliative care, developed initially to provide holistic and timely symptom-based care for patients with noncurable cancer, should also be available and offered to patients with nonmalignant, life-limiting diseases.23,36,37 Meeting this standard of geriatric care is not easy, given the burden of frailty in this population. Needed are multimodal palliative efforts across the spectrum of settings, from the home to the hospital and nursing home.23
To do this, we need to embrace the complexity posed by each person’s presentation and view care through the frailty lens. This will give us a common language in which to engage in a conversation with the same goal in mind: optimizing quality of life.
Furthermore, quantifying frailty can help minimize interventions that are futile or burdensome, that are not expected to ease symptoms, and that can worsen cognition and function. At the end of a patient’s life, we do not want to add to his or her frailty burden but rather minimize the morbidity associated with it.
The concept of frailty assessment is therefore essential for the timely delivery of holistic palliative care in geriatric patients who have progressive and ultimately terminal conditions.
- Rockwood K, Mitnitski A. Frailty defined by deficit accumulation and geriatric medicine defined by frailty. Clin Geriatr Med 2011; 27:17–26.
- Michel JP, Bonin-Guillame S, Gold G, Herrmann F. Cognition and frailty: possible interrelations. In:Carey JR, Robine JM, Michel JP, Christen Y, editors. Longevity and Frailty. Berlin Heidelberg: Springer; 2005:119–124. Research and Perspectives in Longevity.
- Espinoza S, Walston JD. Frailty in older adults: insights and interventions. Cleve Clin J Med 2005; 72:1105–1112.
- Boockvar KS, Meier DE. Palliative care for frail older adults: “there are things I can’t do anymore that I wish I could . . . “. JAMA 2006; 296:2245–2253.
- Abellan van Kan G, Rolland Y, Houles M, Gillette-Guyonnet S, Soto M, Vellas B. The assessment of frailty in older adults. Clin Geriatr Med 2010; 26:275–286.
- McDermid RC, Bagshaw SM. ICU and critical care outreach for the elderly. Best Pract Res Clin Anaesthesiol 2011; 25:439–449.
- Fried LP, Tangen CM, Walston J, et al; Cardiovascular Health Study Collaborative Research Group. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci 2001; 56:M146–M156.
- Rockwood K, Fox RA, Stolee P, Robertson D, Beattie BL. Frailty in elderly people: an evolving concept. CMAJ 1994; 150:489–495.
- Puts MT, Monette J, Girre V, et al. Are frailty markers useful for predicting treatment toxicity and mortality in older newly diagnosed cancer patients? Results from a prospective pilot study. Crit Rev Oncol Hematol 2011; 78:138–149.
- Oken MM, Creech RH, Tormey DC, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 1982; 5:649–655.
- Andrew MK, Mitnitski AB, Rockwood K. Social vulnerability, frailty and mortality in elderly people. PLoS One 2008; 3:e2232.
- Abellan van Kan G, Rolland YM, Morley JE, Vellas B. Frailty: toward a clinical definition. J Am Med Dir Assoc 2008; 9:71–72.
- Eckel SP, Bandeen-Roche K, Chaves PH, Fried LP, Louis TA. Surrogate screening models for the low physical activity criterion of frailty. Aging Clin Exp Res 2011; 23:209–216.
- Gobbens RJ, van Assen MA, Luijkx KG, Wijnen-Sponselee MT, Schols JM. The Tilburg Frailty Indicator: psychometric properties. J Am Med Dir Assoc 2010; 11:344–355.
- Rolfson DB, Majumdar SR, Tsuyuki RT, Tahir A, Rockwood K. Validity and reliability of the Edmonton Frail Scale. Age Ageing 2006; 35:526–529.
- Mallery LH, Moorhouse P. Respecting frailty. J Med Ethics 2011; 37:126–128.
- McDermid RC, Stelfox HT, Bagshaw SM. Frailty in the critically ill: a novel concept. Crit Care 2011; 15:301.
- Kulminski AM, Ukraintseva SV, Kulminskaya IV, Arbeev KG, Land K, Yashin AI. Cumulative deficits better characterize susceptibility to death in elderly people than phenotypic frailty: lessons from the Cardiovascular Health Study. J Am Geriatr Soc 2008; 56:898–903.
- Rockwood K, Mitnitski A. Frailty in relation to the accumulation of deficits. J Gerontol A Biol Sci Med Sci 2007; 62:722–727.
- Rockwood K, Mitnitski A. Limits to deficit accumulation in elderly people. Mech Ageing Dev 2006; 127:494–496.
- Rockwood K, Rockwood MR, Mitnitski A. Physiological redundancy in older adults in relation to the change with age in the slope of a frailty index. J Am Geriatr Soc 2010; 58:318–323.
- Kulminski A, Yashin A, Ukraintseva S, et al. Accumulation of health disorders as a systemic measure of aging: findings from the NLTCS data. Mech Ageing Dev 2006; 127:840–848.
- Davies E, Higginson IJ; WHO Europe. Better Palliative Care for Older People. Milan, Italy: Tipolitografia Trabella Sr; 2004.
- Raudonis BM, Daniel K. Frailty: an indication for palliative care. Geriatr Nurs 2010; 31:379–384.
- American Geriatrics Society Panel on Pharmacological Management of Persistent Pain in Older Persons. Pharmacological management of persistent pain in older persons. J Am Geriatr Soc 2009; 57:1331–1436.
- AGS Panel on Persistent Pain in Older Persons. The management of persistent pain in older persons. J Am Geriatr Soc 2002; 50(suppl 6):S205–S224.
- Shega JW, Dale W, Andrew M, Paice J, Rockwood K, Weiner DK. Persistent pain and frailty: a case for homeostenosis. J Am Geriatr Soc 2012; 60:113–117.
- Hanlon JT, Perera S, Sevick MA, Rodriguez KL, Jaffe EJ. Pain and its treatment in older nursing home hospice/palliative care residents. J Am Med Dir Assoc 2010; 11:579–583.
- Currow DC, Stevenson JP, Abernethy AP, Plummer J, Shelby-James TM. Prescribing in palliative care as death approaches. J Am Geriatr Soc 2007; 55:590–595.
- Holmes HM, Hayley DC, Alexander GC, Sachs GA. Reconsidering medication appropriateness for patients late in life. Arch Intern Med 2006; 166:605–609.
- Kapo J, Morrison LJ, Liao S. Palliative care for the older adult. J Palliat Med 2007; 10:185–209.
- Gaertner J, Wolf J, Frechen S, et al. Recommending early integration of palliative care—does it work? Support Care Cancer 2012; 20:507–513.
- Chen CC, Lin MT, Tien YW, Yen CJ, Huang GH, Inouye SK. Modified hospital elder life program: effects on abdominal surgery patients. J Am Coll Surg 2011; 213:245–252.
- Mitchell SL, Teno JM, Kiely DK, et al. The clinical course of advanced dementia. N Engl J Med 2009; 361:1529–1538.
- Arcand M, Monette J, Monette M, et al. Educating nursing home staff about the progression of dementia and the comfort care option: impact on family satisfaction with end-of-life care. J Am Med Dir Assoc 2009; 10:50–55.
- Mitchell SL, Kiely DK, Hamel MB. Dying with advanced dementia in the nursing home. Arch Intern Med 2004; 164:321–326.
- Birch D, Draper J. A critical literature review exploring the challenges of delivering effective palliative care to older people with dementia. J Clin Nurs 2008; 17:1144–1163.
As people get older, they have more things wrong with them. And the more things they have wrong with them, the more likely they are to die. But everyone accumulates deficits at a different rate, and not all people of the same age have the same short-term risk of dying. This variable susceptibility to death and other adverse outcomes in older people of the same age is called frailty.1
Frailty poses special challenges to how we organize and deliver health care. These challenges are sometimes seen most starkly when people are most frail, especially as they approach the end of life.
In this paper, we will review how frailty is conceptualized and defined, consider how frailty affects the care of people at the end of their lives, and suggest practices that can make end-of-life care better for frail older adults.
DEFINING FRAILTY
As with all complex systems, when frail people become acutely unwell their highest-order functions fail first. Thus, cognitive impairment, functional decline, impaired mobility, and social withdrawal are hallmark presentations of the further accumulation of deficits in vulnerable seniors.
Delirium and falls are important clues that a person’s resilience is becoming compromised and that the person is at risk of further insults in a downward spiral or acceleration of things going wrong.1,2 Frailty is associated with poor health outcomes, from disability to institutionalization and death.3
This idea of frailty as vulnerability arising from dysregulation of multiple physiologic systems is reasonably non-controversial. Even so, there are competing views on how to systematically quantify those who are at an increased risk of adverse sequelae.
Quantifying frailty is particularly important if it can tell us if a patient is at high risk of further decline and death. As frailty advances, it is appropriate to shift the focus of care to palliation, with the goal of optimizing quality of life and easing symptoms.4 Identifying someone as frail can aid decision-making in the setting of critical illness, where the system commonly defaults to an “always do everything” mode without considering the ramifications of such an approach. Furthermore, without a routine means of measuring frailty, it is often left to critical care units or rapid-response medical teams to initiate a discussion about whether an aggressive course of care is appropriate or desired.5,6
Frailty as a syndrome
Fried et al7 defined frailty as a syndrome arising from the “physiologic triad” of sarcopenia and immune and neuroendocrine dysregulation. Patients are considered frail if they have three or more of the following five criteria:
- Reduced activity
- Slowing of mobility
- Weight loss
- Diminished handgrip strength
- Exhaustion.
Someone who has only one or two of these items is said to be “pre-frail”; someone with none is said to be “robust.”
The frailty index
An alternative viewpoint is that frailty is a state arising from the accumulation of deficits, which can be counted in a frailty index.
The frailty index is based on the concept that frailty is a consequence of interacting physical, psychological, and social factors. As deficits accumulate, people become increasingly vulnerable to adverse outcomes.
The frailty index is calculated as the number of deficits the patient has, divided by the number of deficits considered. For example, in a frailty index based on a comprehensive geriatric assessment, an individual with impairments in 4 of 10 domains and with 10 of 24 possible comorbidities would have 14 of 34 possible deficits, for a frailty index of 0.41.8
A criticism of the frailty index is that it includes functional dependence as a deficit. The criticism stems from the view that frailty should be seen as occurring prior to disability. According to this view, including dependence in instrumental and basic activities of daily living as a deficit confuses disability with frailty.
Proponents of the frailty index counter that frailty is not “all or none” and needs to be graded. The frailty index can distinguish between people with and without disability by means of the number of deficits that they have, which is most important. For example, a person disabled by a paraplegic injury would have a lower frailty index score and therefore would be considered less frail than a person with advanced cancer affecting multiple body systems. (This is assuming the person who has suffered the injury resulting in paraplegia doesn’t have a concomitant condition such as renal failure or heart disease. In the absence of other health insults, such patients are less at risk of further morbidity or death than the patient with advanced cancer until they get another health insult or insults added to their frailty.)
In any case, functional capacity is fundamental in medical decision-making and when estimating prognoses. An example is the use of the Eastern Cooperative Oncology Group’s functional status measure.9,10
Sum of physical and psychological stressors
Consensus is growing for the concept that frail people are made more vulnerable by the combination of both physical and psychological stressors. This is particularly important to bear in mind for patients who may appear physically robust but whose total health burden makes them vulnerable to further insults.
For example, think of a relatively young overweight patient with hypertension, diabetes, dyslipidemia, and ischemic white matter changes (which can manifest as low mood and even mild vascular cognitive impairment). In such a patient, an acute illness could result in cognitive and functional decline that can be permanent.
Balance of assets and deficits
About 20 years ago, we used the metaphor of a balance beam to describe how frailty comes about in older adults. In this view, there is an interplay of physiological and functional health determinants. Assets such as health, resources, and caregivers are balanced against deficits such as illnesses, dependency on others, and support burden.8
For the most part, later concepts of frailty have focused on the individual, with social factors construed separately as social vulnerability.11
Tools for assessing frailty in people who are not yet disabled
Several tools exist to clinically assess frailty in people who are not yet disabled.
The FRAIL scale.12 The Geriatric Advisory Panel of the International Academy of Nutrition and Aging formulated a scale for measuring frailty as a “pre-disability state.” The FRAIL scale consists of five easily remembered items:
- Fatigue
- Resistance (inability to climb one flight of stairs)
- Ambulation (inability to walk one block)
- Illnesses (more than five)
- Loss of weight (> 5%).
Like the “reduced activity” criterion of the frailty syndrome mentioned above (in practical terms, described as the inability to do heavy household chores),13 the FRAIL scale seems to blur the distinction between disability (here, the inability to climb stairs or to walk a block) and “pre-disability,” to an uncertain end. It also seems to blend the notion of a state and a syndrome; these points will need to be clarified in due course.
The Tilburg Frailty Indicator14 was constructed around the multidimensional viewpoint of frailty, beyond disease or disability state, to identify frail community-dwelling older individuals. The first part of this two-part questionnaire consists of 10 questions on frailty determinants and medical comorbidities, while the second part contains physical, psychological, and social variables strongly associated with frailty, as well as information about disability in walking and balance. Interestingly, although it includes both social and physical factors, it does not include cognition.
The Clinical Frailty Scale was developed as a practical approach to assess frailty using physical and functional indicators of health and illness burden. The descriptors for this 7-point scale guide clinicians in quantifying the degree of frailty present. It ranges from 1 (very fit) to 7 (severely frail).7 The higher the score, the higher the risks of death or institutionalization. Even mild frailty is associated with a 50% 5-year mortality rate in community-dwelling older adults (Figure 1).8
The Edmonton Frail Scale,15 like the Clinical Frailty Scale, was developed to be practical and usable at the bedside. It is based on the following domains: cognition, general health status, functional independence, social support, medication use, nutrition, mood, continence, and functional performance.
In a community-based sample, the Edmonton Frail Scale compared favorably with the clinical assessment of geriatric specialists who completed a comprehensive evaluation (Pearson’s correlation coefficient 0.64, P < .001).15
FRAILTY AS A PROGNOSTIC INDICATOR
Using frailty scales to aid in prognostication can be useful to clinicians. Survival prognostication is inherently challenging in individuals with multiple comorbidities and variable trajectories of decline, but it remains a vital clinical skill for all clinicians. Framing these difficult discussions in the context of degree of frailty provides a unifying concept, beyond a single-system construct, for care providers, patients, and their loved ones.
Patients nearing the end of their lives need this kind of clarity and support. Regardless of their diagnoses, patients typically want to know when they are at high risk of dying, as do their families and caregivers. People in general look for such information so that they can align medical decision-making congruently with predicted prognosis.16,17 They also use it to plan for the final chapter of their life and their death.
The frailty index is strongly correlated with risk of death
The frailty index is strongly correlated with the risk of death, with a correlation coefficient greater than 0.95. As such, an individual’s frailty index score is considered an estimate of biologic age, which has greater correlation with associated morbidity and death than does chronological age.18,19 In the general population, more than 99% of people have a frailty index value of less than 0.7. As people approach this value, the chance of survival is greatly diminished; indeed, one report suggested that of those who have a frailty index value of more than 0.5 (based on a comprehensive geriatric assessment), 100% are dead by about 20 months later.20,21
In short, there is a limit to which deficits can be added before the system fails. In this sense, the frailty index is akin to the concept of physiologic reserve. Reserve is finite, and as a system loses redundancy it can no longer survive new stresses.
What does this information mean for individual patients?
Even so, prognostication for individual patients remains probabilistic. Any patient has a chance to improve, stabilize, worsen, or die. However, a patient can reach an upper limit of frailty. At that point, instead of accumulating another deficit, death is much more likely. Similarly, although improvement can happen, the chance of improvement is low, and the improvement is typically modest.
Framing survival possibilities in terms of the number of things that people have wrong with them and the chance of death or of change (and the extent of change) makes sense to physicians, patients, and families. Being able to do so offers a much greater opportunity for realistic discussions of the likely outcomes of medical care than the foreseeable scenario of a junior doctor asking a senior citizen, “If your heart stops, do you want us to save your life?”
Understanding prognosis in the face of not just disease but also frailty can also help us focus not on disease but on health consequences of illness. Can the person think? Walk? Care for herself or himself? Interact with others? These questions need to be considered when end-of-life decisions are being discussed.22
Since making predictions about survival is most challenging when multiple comorbidities are present, using the concept of accumulating deficits to better define the slope of decline can be very helpful when discussing “the road ahead” with patients and their families. Visually mapping out the slope of decline and how it is accelerating as conditions progress and deficits accumulate can aid in medical decision-making. Looking individually at the deficits themselves and associated markers of progression can also help with prognostic discussions.
For example, a patient with chronic obstructive pulmonary disease could very well be unaware of the progression and ultimately terminal prognosis of this disease. The slope of clinical decline can be initially shallow, with saw-tooth fluctuations from acute exacerbations that seemingly “resolve to baseline” when antibiotic and steroid courses are completed. Talking with these patients and their families about heralding markers, such as more hospitalizations and cognitive decline with acute exacerbations, can clarify the steepening slope of decline and the way comorbidities interact.
FRAILTY AND END-OF-LIFE CARE
Frailty is progressive, and as it worsens, integrating a palliative approach is appropriate, with a focus on optimizing quality of life and relieving symptoms.4 This principle holds true regardless of the care setting, from acute care hospitals to hospice facilities and long-term care residences.
The principles of end-of-life care are applicable to frail individuals with progressive conditions from the time of diagnosis throughout the course of decline. As the population ages, more people suffer and die from progressive chronic conditions such as cerebrovascular disease, respiratory disease, and dementia.23 An interdisciplinary team approach can ensure all components of palliation are effectively delivered, such as easing symptoms, providing psychosocial and spiritual support, and improving quality of life.24
Pain management
Pain is widely underassessed and undertreated in older patients. Its management at the end of life is particularly challenging if the patient’s language is compromised, as in dementia.23,25,26
A recent cross-sectional analysis of self-reported pain in a longitudinal study of community-dwelling older adults showed an independent association between moderate or higher pain and frailty. The authors propose that persistent pain goes beyond physical discomfort in that it may contribute to homeostenosis (progressive diminishment of homeostatic reserve) and directly worsen frailty.27
Examine the medication list
In palliative care, medical interventions focus on optimizing quality of life.
This especially includes reexamining long medication lists that increase the chance of adverse drug effects.28 Many patients are on disease-modifying medications that may or may not help control symptoms—and might well exacerbate them. For example, betablockers for ischemic heart disease and angiotensin-converting enzyme inhibitors for diabetic nephropathy both can cause hypotension-induced lassitude or even falls due to orthostasis.
A sensible approach is to keep the drugs that may still contribute to quality of life, while discontinuing other drugs that may be causing side effects or that are unlikely to provide meaningful benefit in terms of prevention in patients who have limited life expectancy.29 Discontinuing ineffective, poorly tolerated, and duplicated medications also makes it easier to introduce new medications to manage symptoms—there will be fewer drug interactions, and fewer pills to take, an increasingly important issue in the setting of gastrointestinal symptoms such as dysphagia and gastroparesis or compliance issues as frequently encountered when cognitive impairment is present.29,30
In managing symptoms, start low and go slow—but get there
In managing symptoms in frail elderly patients we use the same classes of medications as in younger patients. The trick is to use appropriate doses.
The concept of “start low and go slow” is key, but so is “get there”—ie, reach the therapeutic goal. The principal drugs for symptom control, such as opioids for pain and dyspnea and anxiolytics for anxiety and restlessness, are associated with a higher rate of and more severe adverse effects in frail older adults.
Even so, most frail older adults appear to be undertreated in this regard, particularly if they are cognitively impaired.23 This fact, coupled with the reality that behavioral symptoms associated with advanced dementia can represent unmet care needs including undertreated pain, highlights the critical need to control symptoms optimally in frail seniors.
This is particularly relevant for those who can no longer verbally articulate their symptoms. Nonverbal pain scales and vigilant assessment of behavioral signs of pain are paramount skills for clinicians providing palliative care to patients with cognitive impairment. Caregivers and loved ones should be included in the assessment process.26,31
Adjunctive therapies for pain control
Maximizing adjunctive therapies can optimize pain control in this drug-sensitive population. Heat and cold packs to affected areas, acupuncture, massage therapy, and structured exercise regimens are some options that can improve quality of life. Cognitive behavioral therapy may offer coping strategies, provided the patient can participate in this process from a cognitive perspective.26,27 Topical preparations are often well tolerated and may include medicinal ingredients that are helpful without systemic effects, such as anti-inflammatory drugs or analgesics.
Optimal use of nonopioid drugs may help reduce the need for narcotics, particularly in the presence of musculoskeletal pain. An example is acetaminophen in regular doses—we would recommend no more than 3 g per day. Acetaminophen is preferred for older adults rather than nonsteroidal anti-inflammatory drugs, given the potential gastric and cardiovascular side effects of the latter medications.
Antidepressants and anticonvulsants such as gabapentin can also be considered as adjuncts for pain control, particularly in the setting of neuropathic pain, with careful monitoring for tolerance.25
When opioids are used, vigilance for constipation is essential.
Establishing goals of care
Goals of care need to be established incrementally along the course of clinical decline and as early as possible so that palliative support can be promptly implemented as symptoms worsen.32
End-of-life care can still include treatments with curative intent, depending on the overall prognosis and the state of the underlying terminal illness. On the other hand, frail older adults who are subjected to invasive treatments that are unlikely to provide cure, such as Whipple surgery, need special intervention postoperatively if they are not to suffer complications such as persisting delirium and functional decline.33
In this regard, geriatric palliative care is frequently about not “crossing a threshold.” Patients may still be receiving active management and be hospitalized for acute exacerbations of progressing chronic conditions, such as chronic obstructive pulmonary disease and heart failure, while palliative principles are introduced and increasingly become the focus of care.
To align goals of care with frailty burden, it is crucial to quantify frailty and to review the patient’s comorbidities. Particularly when dementia is present, lack of communication between the patient’s doctors or between the doctor and the family about disease burden can lead to inappropriately aggressive care.
Many family members and even clinicians do not recognize that advanced dementia is terminal.34,35 In light of this, a palliative approach to care may not even be considered as an appropriate plan when hallmark complications associated with progressing cognitive decline occur, such as aspiration pneumonia or dehydration. Education about dementia and other conditions with progressive organ failure should be done as soon as possible after diagnosis and readdressed at intervals throughout the patient’s clinical decline.
Earlier discussions also ensure that patients themselves can be involved in decision-making more often before cognitive impairment advances to the point where proxy discussions take over.16
BETTER PALLIATIVE CARE FOR ALL
Palliative care, developed initially to provide holistic and timely symptom-based care for patients with noncurable cancer, should also be available and offered to patients with nonmalignant, life-limiting diseases.23,36,37 Meeting this standard of geriatric care is not easy, given the burden of frailty in this population. Needed are multimodal palliative efforts across the spectrum of settings, from the home to the hospital and nursing home.23
To do this, we need to embrace the complexity posed by each person’s presentation and view care through the frailty lens. This will give us a common language in which to engage in a conversation with the same goal in mind: optimizing quality of life.
Furthermore, quantifying frailty can help minimize interventions that are futile or burdensome, that are not expected to ease symptoms, and that can worsen cognition and function. At the end of a patient’s life, we do not want to add to his or her frailty burden but rather minimize the morbidity associated with it.
The concept of frailty assessment is therefore essential for the timely delivery of holistic palliative care in geriatric patients who have progressive and ultimately terminal conditions.
As people get older, they have more things wrong with them. And the more things they have wrong with them, the more likely they are to die. But everyone accumulates deficits at a different rate, and not all people of the same age have the same short-term risk of dying. This variable susceptibility to death and other adverse outcomes in older people of the same age is called frailty.1
Frailty poses special challenges to how we organize and deliver health care. These challenges are sometimes seen most starkly when people are most frail, especially as they approach the end of life.
In this paper, we will review how frailty is conceptualized and defined, consider how frailty affects the care of people at the end of their lives, and suggest practices that can make end-of-life care better for frail older adults.
DEFINING FRAILTY
As with all complex systems, when frail people become acutely unwell their highest-order functions fail first. Thus, cognitive impairment, functional decline, impaired mobility, and social withdrawal are hallmark presentations of the further accumulation of deficits in vulnerable seniors.
Delirium and falls are important clues that a person’s resilience is becoming compromised and that the person is at risk of further insults in a downward spiral or acceleration of things going wrong.1,2 Frailty is associated with poor health outcomes, from disability to institutionalization and death.3
This idea of frailty as vulnerability arising from dysregulation of multiple physiologic systems is reasonably non-controversial. Even so, there are competing views on how to systematically quantify those who are at an increased risk of adverse sequelae.
Quantifying frailty is particularly important if it can tell us if a patient is at high risk of further decline and death. As frailty advances, it is appropriate to shift the focus of care to palliation, with the goal of optimizing quality of life and easing symptoms.4 Identifying someone as frail can aid decision-making in the setting of critical illness, where the system commonly defaults to an “always do everything” mode without considering the ramifications of such an approach. Furthermore, without a routine means of measuring frailty, it is often left to critical care units or rapid-response medical teams to initiate a discussion about whether an aggressive course of care is appropriate or desired.5,6
Frailty as a syndrome
Fried et al7 defined frailty as a syndrome arising from the “physiologic triad” of sarcopenia and immune and neuroendocrine dysregulation. Patients are considered frail if they have three or more of the following five criteria:
- Reduced activity
- Slowing of mobility
- Weight loss
- Diminished handgrip strength
- Exhaustion.
Someone who has only one or two of these items is said to be “pre-frail”; someone with none is said to be “robust.”
The frailty index
An alternative viewpoint is that frailty is a state arising from the accumulation of deficits, which can be counted in a frailty index.
The frailty index is based on the concept that frailty is a consequence of interacting physical, psychological, and social factors. As deficits accumulate, people become increasingly vulnerable to adverse outcomes.
The frailty index is calculated as the number of deficits the patient has, divided by the number of deficits considered. For example, in a frailty index based on a comprehensive geriatric assessment, an individual with impairments in 4 of 10 domains and with 10 of 24 possible comorbidities would have 14 of 34 possible deficits, for a frailty index of 0.41.8
A criticism of the frailty index is that it includes functional dependence as a deficit. The criticism stems from the view that frailty should be seen as occurring prior to disability. According to this view, including dependence in instrumental and basic activities of daily living as a deficit confuses disability with frailty.
Proponents of the frailty index counter that frailty is not “all or none” and needs to be graded. The frailty index can distinguish between people with and without disability by means of the number of deficits that they have, which is most important. For example, a person disabled by a paraplegic injury would have a lower frailty index score and therefore would be considered less frail than a person with advanced cancer affecting multiple body systems. (This is assuming the person who has suffered the injury resulting in paraplegia doesn’t have a concomitant condition such as renal failure or heart disease. In the absence of other health insults, such patients are less at risk of further morbidity or death than the patient with advanced cancer until they get another health insult or insults added to their frailty.)
In any case, functional capacity is fundamental in medical decision-making and when estimating prognoses. An example is the use of the Eastern Cooperative Oncology Group’s functional status measure.9,10
Sum of physical and psychological stressors
Consensus is growing for the concept that frail people are made more vulnerable by the combination of both physical and psychological stressors. This is particularly important to bear in mind for patients who may appear physically robust but whose total health burden makes them vulnerable to further insults.
For example, think of a relatively young overweight patient with hypertension, diabetes, dyslipidemia, and ischemic white matter changes (which can manifest as low mood and even mild vascular cognitive impairment). In such a patient, an acute illness could result in cognitive and functional decline that can be permanent.
Balance of assets and deficits
About 20 years ago, we used the metaphor of a balance beam to describe how frailty comes about in older adults. In this view, there is an interplay of physiological and functional health determinants. Assets such as health, resources, and caregivers are balanced against deficits such as illnesses, dependency on others, and support burden.8
For the most part, later concepts of frailty have focused on the individual, with social factors construed separately as social vulnerability.11
Tools for assessing frailty in people who are not yet disabled
Several tools exist to clinically assess frailty in people who are not yet disabled.
The FRAIL scale.12 The Geriatric Advisory Panel of the International Academy of Nutrition and Aging formulated a scale for measuring frailty as a “pre-disability state.” The FRAIL scale consists of five easily remembered items:
- Fatigue
- Resistance (inability to climb one flight of stairs)
- Ambulation (inability to walk one block)
- Illnesses (more than five)
- Loss of weight (> 5%).
Like the “reduced activity” criterion of the frailty syndrome mentioned above (in practical terms, described as the inability to do heavy household chores),13 the FRAIL scale seems to blur the distinction between disability (here, the inability to climb stairs or to walk a block) and “pre-disability,” to an uncertain end. It also seems to blend the notion of a state and a syndrome; these points will need to be clarified in due course.
The Tilburg Frailty Indicator14 was constructed around the multidimensional viewpoint of frailty, beyond disease or disability state, to identify frail community-dwelling older individuals. The first part of this two-part questionnaire consists of 10 questions on frailty determinants and medical comorbidities, while the second part contains physical, psychological, and social variables strongly associated with frailty, as well as information about disability in walking and balance. Interestingly, although it includes both social and physical factors, it does not include cognition.
The Clinical Frailty Scale was developed as a practical approach to assess frailty using physical and functional indicators of health and illness burden. The descriptors for this 7-point scale guide clinicians in quantifying the degree of frailty present. It ranges from 1 (very fit) to 7 (severely frail).7 The higher the score, the higher the risks of death or institutionalization. Even mild frailty is associated with a 50% 5-year mortality rate in community-dwelling older adults (Figure 1).8
The Edmonton Frail Scale,15 like the Clinical Frailty Scale, was developed to be practical and usable at the bedside. It is based on the following domains: cognition, general health status, functional independence, social support, medication use, nutrition, mood, continence, and functional performance.
In a community-based sample, the Edmonton Frail Scale compared favorably with the clinical assessment of geriatric specialists who completed a comprehensive evaluation (Pearson’s correlation coefficient 0.64, P < .001).15
FRAILTY AS A PROGNOSTIC INDICATOR
Using frailty scales to aid in prognostication can be useful to clinicians. Survival prognostication is inherently challenging in individuals with multiple comorbidities and variable trajectories of decline, but it remains a vital clinical skill for all clinicians. Framing these difficult discussions in the context of degree of frailty provides a unifying concept, beyond a single-system construct, for care providers, patients, and their loved ones.
Patients nearing the end of their lives need this kind of clarity and support. Regardless of their diagnoses, patients typically want to know when they are at high risk of dying, as do their families and caregivers. People in general look for such information so that they can align medical decision-making congruently with predicted prognosis.16,17 They also use it to plan for the final chapter of their life and their death.
The frailty index is strongly correlated with risk of death
The frailty index is strongly correlated with the risk of death, with a correlation coefficient greater than 0.95. As such, an individual’s frailty index score is considered an estimate of biologic age, which has greater correlation with associated morbidity and death than does chronological age.18,19 In the general population, more than 99% of people have a frailty index value of less than 0.7. As people approach this value, the chance of survival is greatly diminished; indeed, one report suggested that of those who have a frailty index value of more than 0.5 (based on a comprehensive geriatric assessment), 100% are dead by about 20 months later.20,21
In short, there is a limit to which deficits can be added before the system fails. In this sense, the frailty index is akin to the concept of physiologic reserve. Reserve is finite, and as a system loses redundancy it can no longer survive new stresses.
What does this information mean for individual patients?
Even so, prognostication for individual patients remains probabilistic. Any patient has a chance to improve, stabilize, worsen, or die. However, a patient can reach an upper limit of frailty. At that point, instead of accumulating another deficit, death is much more likely. Similarly, although improvement can happen, the chance of improvement is low, and the improvement is typically modest.
Framing survival possibilities in terms of the number of things that people have wrong with them and the chance of death or of change (and the extent of change) makes sense to physicians, patients, and families. Being able to do so offers a much greater opportunity for realistic discussions of the likely outcomes of medical care than the foreseeable scenario of a junior doctor asking a senior citizen, “If your heart stops, do you want us to save your life?”
Understanding prognosis in the face of not just disease but also frailty can also help us focus not on disease but on health consequences of illness. Can the person think? Walk? Care for herself or himself? Interact with others? These questions need to be considered when end-of-life decisions are being discussed.22
Since making predictions about survival is most challenging when multiple comorbidities are present, using the concept of accumulating deficits to better define the slope of decline can be very helpful when discussing “the road ahead” with patients and their families. Visually mapping out the slope of decline and how it is accelerating as conditions progress and deficits accumulate can aid in medical decision-making. Looking individually at the deficits themselves and associated markers of progression can also help with prognostic discussions.
For example, a patient with chronic obstructive pulmonary disease could very well be unaware of the progression and ultimately terminal prognosis of this disease. The slope of clinical decline can be initially shallow, with saw-tooth fluctuations from acute exacerbations that seemingly “resolve to baseline” when antibiotic and steroid courses are completed. Talking with these patients and their families about heralding markers, such as more hospitalizations and cognitive decline with acute exacerbations, can clarify the steepening slope of decline and the way comorbidities interact.
FRAILTY AND END-OF-LIFE CARE
Frailty is progressive, and as it worsens, integrating a palliative approach is appropriate, with a focus on optimizing quality of life and relieving symptoms.4 This principle holds true regardless of the care setting, from acute care hospitals to hospice facilities and long-term care residences.
The principles of end-of-life care are applicable to frail individuals with progressive conditions from the time of diagnosis throughout the course of decline. As the population ages, more people suffer and die from progressive chronic conditions such as cerebrovascular disease, respiratory disease, and dementia.23 An interdisciplinary team approach can ensure all components of palliation are effectively delivered, such as easing symptoms, providing psychosocial and spiritual support, and improving quality of life.24
Pain management
Pain is widely underassessed and undertreated in older patients. Its management at the end of life is particularly challenging if the patient’s language is compromised, as in dementia.23,25,26
A recent cross-sectional analysis of self-reported pain in a longitudinal study of community-dwelling older adults showed an independent association between moderate or higher pain and frailty. The authors propose that persistent pain goes beyond physical discomfort in that it may contribute to homeostenosis (progressive diminishment of homeostatic reserve) and directly worsen frailty.27
Examine the medication list
In palliative care, medical interventions focus on optimizing quality of life.
This especially includes reexamining long medication lists that increase the chance of adverse drug effects.28 Many patients are on disease-modifying medications that may or may not help control symptoms—and might well exacerbate them. For example, betablockers for ischemic heart disease and angiotensin-converting enzyme inhibitors for diabetic nephropathy both can cause hypotension-induced lassitude or even falls due to orthostasis.
A sensible approach is to keep the drugs that may still contribute to quality of life, while discontinuing other drugs that may be causing side effects or that are unlikely to provide meaningful benefit in terms of prevention in patients who have limited life expectancy.29 Discontinuing ineffective, poorly tolerated, and duplicated medications also makes it easier to introduce new medications to manage symptoms—there will be fewer drug interactions, and fewer pills to take, an increasingly important issue in the setting of gastrointestinal symptoms such as dysphagia and gastroparesis or compliance issues as frequently encountered when cognitive impairment is present.29,30
In managing symptoms, start low and go slow—but get there
In managing symptoms in frail elderly patients we use the same classes of medications as in younger patients. The trick is to use appropriate doses.
The concept of “start low and go slow” is key, but so is “get there”—ie, reach the therapeutic goal. The principal drugs for symptom control, such as opioids for pain and dyspnea and anxiolytics for anxiety and restlessness, are associated with a higher rate of and more severe adverse effects in frail older adults.
Even so, most frail older adults appear to be undertreated in this regard, particularly if they are cognitively impaired.23 This fact, coupled with the reality that behavioral symptoms associated with advanced dementia can represent unmet care needs including undertreated pain, highlights the critical need to control symptoms optimally in frail seniors.
This is particularly relevant for those who can no longer verbally articulate their symptoms. Nonverbal pain scales and vigilant assessment of behavioral signs of pain are paramount skills for clinicians providing palliative care to patients with cognitive impairment. Caregivers and loved ones should be included in the assessment process.26,31
Adjunctive therapies for pain control
Maximizing adjunctive therapies can optimize pain control in this drug-sensitive population. Heat and cold packs to affected areas, acupuncture, massage therapy, and structured exercise regimens are some options that can improve quality of life. Cognitive behavioral therapy may offer coping strategies, provided the patient can participate in this process from a cognitive perspective.26,27 Topical preparations are often well tolerated and may include medicinal ingredients that are helpful without systemic effects, such as anti-inflammatory drugs or analgesics.
Optimal use of nonopioid drugs may help reduce the need for narcotics, particularly in the presence of musculoskeletal pain. An example is acetaminophen in regular doses—we would recommend no more than 3 g per day. Acetaminophen is preferred for older adults rather than nonsteroidal anti-inflammatory drugs, given the potential gastric and cardiovascular side effects of the latter medications.
Antidepressants and anticonvulsants such as gabapentin can also be considered as adjuncts for pain control, particularly in the setting of neuropathic pain, with careful monitoring for tolerance.25
When opioids are used, vigilance for constipation is essential.
Establishing goals of care
Goals of care need to be established incrementally along the course of clinical decline and as early as possible so that palliative support can be promptly implemented as symptoms worsen.32
End-of-life care can still include treatments with curative intent, depending on the overall prognosis and the state of the underlying terminal illness. On the other hand, frail older adults who are subjected to invasive treatments that are unlikely to provide cure, such as Whipple surgery, need special intervention postoperatively if they are not to suffer complications such as persisting delirium and functional decline.33
In this regard, geriatric palliative care is frequently about not “crossing a threshold.” Patients may still be receiving active management and be hospitalized for acute exacerbations of progressing chronic conditions, such as chronic obstructive pulmonary disease and heart failure, while palliative principles are introduced and increasingly become the focus of care.
To align goals of care with frailty burden, it is crucial to quantify frailty and to review the patient’s comorbidities. Particularly when dementia is present, lack of communication between the patient’s doctors or between the doctor and the family about disease burden can lead to inappropriately aggressive care.
Many family members and even clinicians do not recognize that advanced dementia is terminal.34,35 In light of this, a palliative approach to care may not even be considered as an appropriate plan when hallmark complications associated with progressing cognitive decline occur, such as aspiration pneumonia or dehydration. Education about dementia and other conditions with progressive organ failure should be done as soon as possible after diagnosis and readdressed at intervals throughout the patient’s clinical decline.
Earlier discussions also ensure that patients themselves can be involved in decision-making more often before cognitive impairment advances to the point where proxy discussions take over.16
BETTER PALLIATIVE CARE FOR ALL
Palliative care, developed initially to provide holistic and timely symptom-based care for patients with noncurable cancer, should also be available and offered to patients with nonmalignant, life-limiting diseases.23,36,37 Meeting this standard of geriatric care is not easy, given the burden of frailty in this population. Needed are multimodal palliative efforts across the spectrum of settings, from the home to the hospital and nursing home.23
To do this, we need to embrace the complexity posed by each person’s presentation and view care through the frailty lens. This will give us a common language in which to engage in a conversation with the same goal in mind: optimizing quality of life.
Furthermore, quantifying frailty can help minimize interventions that are futile or burdensome, that are not expected to ease symptoms, and that can worsen cognition and function. At the end of a patient’s life, we do not want to add to his or her frailty burden but rather minimize the morbidity associated with it.
The concept of frailty assessment is therefore essential for the timely delivery of holistic palliative care in geriatric patients who have progressive and ultimately terminal conditions.
- Rockwood K, Mitnitski A. Frailty defined by deficit accumulation and geriatric medicine defined by frailty. Clin Geriatr Med 2011; 27:17–26.
- Michel JP, Bonin-Guillame S, Gold G, Herrmann F. Cognition and frailty: possible interrelations. In:Carey JR, Robine JM, Michel JP, Christen Y, editors. Longevity and Frailty. Berlin Heidelberg: Springer; 2005:119–124. Research and Perspectives in Longevity.
- Espinoza S, Walston JD. Frailty in older adults: insights and interventions. Cleve Clin J Med 2005; 72:1105–1112.
- Boockvar KS, Meier DE. Palliative care for frail older adults: “there are things I can’t do anymore that I wish I could . . . “. JAMA 2006; 296:2245–2253.
- Abellan van Kan G, Rolland Y, Houles M, Gillette-Guyonnet S, Soto M, Vellas B. The assessment of frailty in older adults. Clin Geriatr Med 2010; 26:275–286.
- McDermid RC, Bagshaw SM. ICU and critical care outreach for the elderly. Best Pract Res Clin Anaesthesiol 2011; 25:439–449.
- Fried LP, Tangen CM, Walston J, et al; Cardiovascular Health Study Collaborative Research Group. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci 2001; 56:M146–M156.
- Rockwood K, Fox RA, Stolee P, Robertson D, Beattie BL. Frailty in elderly people: an evolving concept. CMAJ 1994; 150:489–495.
- Puts MT, Monette J, Girre V, et al. Are frailty markers useful for predicting treatment toxicity and mortality in older newly diagnosed cancer patients? Results from a prospective pilot study. Crit Rev Oncol Hematol 2011; 78:138–149.
- Oken MM, Creech RH, Tormey DC, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 1982; 5:649–655.
- Andrew MK, Mitnitski AB, Rockwood K. Social vulnerability, frailty and mortality in elderly people. PLoS One 2008; 3:e2232.
- Abellan van Kan G, Rolland YM, Morley JE, Vellas B. Frailty: toward a clinical definition. J Am Med Dir Assoc 2008; 9:71–72.
- Eckel SP, Bandeen-Roche K, Chaves PH, Fried LP, Louis TA. Surrogate screening models for the low physical activity criterion of frailty. Aging Clin Exp Res 2011; 23:209–216.
- Gobbens RJ, van Assen MA, Luijkx KG, Wijnen-Sponselee MT, Schols JM. The Tilburg Frailty Indicator: psychometric properties. J Am Med Dir Assoc 2010; 11:344–355.
- Rolfson DB, Majumdar SR, Tsuyuki RT, Tahir A, Rockwood K. Validity and reliability of the Edmonton Frail Scale. Age Ageing 2006; 35:526–529.
- Mallery LH, Moorhouse P. Respecting frailty. J Med Ethics 2011; 37:126–128.
- McDermid RC, Stelfox HT, Bagshaw SM. Frailty in the critically ill: a novel concept. Crit Care 2011; 15:301.
- Kulminski AM, Ukraintseva SV, Kulminskaya IV, Arbeev KG, Land K, Yashin AI. Cumulative deficits better characterize susceptibility to death in elderly people than phenotypic frailty: lessons from the Cardiovascular Health Study. J Am Geriatr Soc 2008; 56:898–903.
- Rockwood K, Mitnitski A. Frailty in relation to the accumulation of deficits. J Gerontol A Biol Sci Med Sci 2007; 62:722–727.
- Rockwood K, Mitnitski A. Limits to deficit accumulation in elderly people. Mech Ageing Dev 2006; 127:494–496.
- Rockwood K, Rockwood MR, Mitnitski A. Physiological redundancy in older adults in relation to the change with age in the slope of a frailty index. J Am Geriatr Soc 2010; 58:318–323.
- Kulminski A, Yashin A, Ukraintseva S, et al. Accumulation of health disorders as a systemic measure of aging: findings from the NLTCS data. Mech Ageing Dev 2006; 127:840–848.
- Davies E, Higginson IJ; WHO Europe. Better Palliative Care for Older People. Milan, Italy: Tipolitografia Trabella Sr; 2004.
- Raudonis BM, Daniel K. Frailty: an indication for palliative care. Geriatr Nurs 2010; 31:379–384.
- American Geriatrics Society Panel on Pharmacological Management of Persistent Pain in Older Persons. Pharmacological management of persistent pain in older persons. J Am Geriatr Soc 2009; 57:1331–1436.
- AGS Panel on Persistent Pain in Older Persons. The management of persistent pain in older persons. J Am Geriatr Soc 2002; 50(suppl 6):S205–S224.
- Shega JW, Dale W, Andrew M, Paice J, Rockwood K, Weiner DK. Persistent pain and frailty: a case for homeostenosis. J Am Geriatr Soc 2012; 60:113–117.
- Hanlon JT, Perera S, Sevick MA, Rodriguez KL, Jaffe EJ. Pain and its treatment in older nursing home hospice/palliative care residents. J Am Med Dir Assoc 2010; 11:579–583.
- Currow DC, Stevenson JP, Abernethy AP, Plummer J, Shelby-James TM. Prescribing in palliative care as death approaches. J Am Geriatr Soc 2007; 55:590–595.
- Holmes HM, Hayley DC, Alexander GC, Sachs GA. Reconsidering medication appropriateness for patients late in life. Arch Intern Med 2006; 166:605–609.
- Kapo J, Morrison LJ, Liao S. Palliative care for the older adult. J Palliat Med 2007; 10:185–209.
- Gaertner J, Wolf J, Frechen S, et al. Recommending early integration of palliative care—does it work? Support Care Cancer 2012; 20:507–513.
- Chen CC, Lin MT, Tien YW, Yen CJ, Huang GH, Inouye SK. Modified hospital elder life program: effects on abdominal surgery patients. J Am Coll Surg 2011; 213:245–252.
- Mitchell SL, Teno JM, Kiely DK, et al. The clinical course of advanced dementia. N Engl J Med 2009; 361:1529–1538.
- Arcand M, Monette J, Monette M, et al. Educating nursing home staff about the progression of dementia and the comfort care option: impact on family satisfaction with end-of-life care. J Am Med Dir Assoc 2009; 10:50–55.
- Mitchell SL, Kiely DK, Hamel MB. Dying with advanced dementia in the nursing home. Arch Intern Med 2004; 164:321–326.
- Birch D, Draper J. A critical literature review exploring the challenges of delivering effective palliative care to older people with dementia. J Clin Nurs 2008; 17:1144–1163.
- Rockwood K, Mitnitski A. Frailty defined by deficit accumulation and geriatric medicine defined by frailty. Clin Geriatr Med 2011; 27:17–26.
- Michel JP, Bonin-Guillame S, Gold G, Herrmann F. Cognition and frailty: possible interrelations. In:Carey JR, Robine JM, Michel JP, Christen Y, editors. Longevity and Frailty. Berlin Heidelberg: Springer; 2005:119–124. Research and Perspectives in Longevity.
- Espinoza S, Walston JD. Frailty in older adults: insights and interventions. Cleve Clin J Med 2005; 72:1105–1112.
- Boockvar KS, Meier DE. Palliative care for frail older adults: “there are things I can’t do anymore that I wish I could . . . “. JAMA 2006; 296:2245–2253.
- Abellan van Kan G, Rolland Y, Houles M, Gillette-Guyonnet S, Soto M, Vellas B. The assessment of frailty in older adults. Clin Geriatr Med 2010; 26:275–286.
- McDermid RC, Bagshaw SM. ICU and critical care outreach for the elderly. Best Pract Res Clin Anaesthesiol 2011; 25:439–449.
- Fried LP, Tangen CM, Walston J, et al; Cardiovascular Health Study Collaborative Research Group. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci 2001; 56:M146–M156.
- Rockwood K, Fox RA, Stolee P, Robertson D, Beattie BL. Frailty in elderly people: an evolving concept. CMAJ 1994; 150:489–495.
- Puts MT, Monette J, Girre V, et al. Are frailty markers useful for predicting treatment toxicity and mortality in older newly diagnosed cancer patients? Results from a prospective pilot study. Crit Rev Oncol Hematol 2011; 78:138–149.
- Oken MM, Creech RH, Tormey DC, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 1982; 5:649–655.
- Andrew MK, Mitnitski AB, Rockwood K. Social vulnerability, frailty and mortality in elderly people. PLoS One 2008; 3:e2232.
- Abellan van Kan G, Rolland YM, Morley JE, Vellas B. Frailty: toward a clinical definition. J Am Med Dir Assoc 2008; 9:71–72.
- Eckel SP, Bandeen-Roche K, Chaves PH, Fried LP, Louis TA. Surrogate screening models for the low physical activity criterion of frailty. Aging Clin Exp Res 2011; 23:209–216.
- Gobbens RJ, van Assen MA, Luijkx KG, Wijnen-Sponselee MT, Schols JM. The Tilburg Frailty Indicator: psychometric properties. J Am Med Dir Assoc 2010; 11:344–355.
- Rolfson DB, Majumdar SR, Tsuyuki RT, Tahir A, Rockwood K. Validity and reliability of the Edmonton Frail Scale. Age Ageing 2006; 35:526–529.
- Mallery LH, Moorhouse P. Respecting frailty. J Med Ethics 2011; 37:126–128.
- McDermid RC, Stelfox HT, Bagshaw SM. Frailty in the critically ill: a novel concept. Crit Care 2011; 15:301.
- Kulminski AM, Ukraintseva SV, Kulminskaya IV, Arbeev KG, Land K, Yashin AI. Cumulative deficits better characterize susceptibility to death in elderly people than phenotypic frailty: lessons from the Cardiovascular Health Study. J Am Geriatr Soc 2008; 56:898–903.
- Rockwood K, Mitnitski A. Frailty in relation to the accumulation of deficits. J Gerontol A Biol Sci Med Sci 2007; 62:722–727.
- Rockwood K, Mitnitski A. Limits to deficit accumulation in elderly people. Mech Ageing Dev 2006; 127:494–496.
- Rockwood K, Rockwood MR, Mitnitski A. Physiological redundancy in older adults in relation to the change with age in the slope of a frailty index. J Am Geriatr Soc 2010; 58:318–323.
- Kulminski A, Yashin A, Ukraintseva S, et al. Accumulation of health disorders as a systemic measure of aging: findings from the NLTCS data. Mech Ageing Dev 2006; 127:840–848.
- Davies E, Higginson IJ; WHO Europe. Better Palliative Care for Older People. Milan, Italy: Tipolitografia Trabella Sr; 2004.
- Raudonis BM, Daniel K. Frailty: an indication for palliative care. Geriatr Nurs 2010; 31:379–384.
- American Geriatrics Society Panel on Pharmacological Management of Persistent Pain in Older Persons. Pharmacological management of persistent pain in older persons. J Am Geriatr Soc 2009; 57:1331–1436.
- AGS Panel on Persistent Pain in Older Persons. The management of persistent pain in older persons. J Am Geriatr Soc 2002; 50(suppl 6):S205–S224.
- Shega JW, Dale W, Andrew M, Paice J, Rockwood K, Weiner DK. Persistent pain and frailty: a case for homeostenosis. J Am Geriatr Soc 2012; 60:113–117.
- Hanlon JT, Perera S, Sevick MA, Rodriguez KL, Jaffe EJ. Pain and its treatment in older nursing home hospice/palliative care residents. J Am Med Dir Assoc 2010; 11:579–583.
- Currow DC, Stevenson JP, Abernethy AP, Plummer J, Shelby-James TM. Prescribing in palliative care as death approaches. J Am Geriatr Soc 2007; 55:590–595.
- Holmes HM, Hayley DC, Alexander GC, Sachs GA. Reconsidering medication appropriateness for patients late in life. Arch Intern Med 2006; 166:605–609.
- Kapo J, Morrison LJ, Liao S. Palliative care for the older adult. J Palliat Med 2007; 10:185–209.
- Gaertner J, Wolf J, Frechen S, et al. Recommending early integration of palliative care—does it work? Support Care Cancer 2012; 20:507–513.
- Chen CC, Lin MT, Tien YW, Yen CJ, Huang GH, Inouye SK. Modified hospital elder life program: effects on abdominal surgery patients. J Am Coll Surg 2011; 213:245–252.
- Mitchell SL, Teno JM, Kiely DK, et al. The clinical course of advanced dementia. N Engl J Med 2009; 361:1529–1538.
- Arcand M, Monette J, Monette M, et al. Educating nursing home staff about the progression of dementia and the comfort care option: impact on family satisfaction with end-of-life care. J Am Med Dir Assoc 2009; 10:50–55.
- Mitchell SL, Kiely DK, Hamel MB. Dying with advanced dementia in the nursing home. Arch Intern Med 2004; 164:321–326.
- Birch D, Draper J. A critical literature review exploring the challenges of delivering effective palliative care to older people with dementia. J Clin Nurs 2008; 17:1144–1163.
KEY POINTS
- Frail older adults are more susceptible to delirium, functional decline, impaired mobility, falls, social withdrawal, and death.
- Evaluating the health care needs of people who are frail requires assessment of their cognition, function, mobility, balance, and social circumstances, in addition to understanding their medical problems.
- When people are so frail that they cannot withstand interventions that can cause significant injury, such as surgery or chemotherapy, then appropriate end-of-life care should focus on maintaining their highest-order functions.
- End-of-life care can include curative treatments of some episodes if they threaten cognition, mobility, or function or cause pain and suffering, even in the context of an overall palliative care plan.
Online network offers 24/7 patient support
When a cancer patient from Menomonie, Wis., reached out to the American Cancer Society’s WhatNext online network seeking treatment reassurance, a ready-made support group jumped into action.
"Anyone familiar with 5-FU [5-fluorouracil], cetuximab (Erbitux), or carboplatin? Has anyone ever worn a pump?" asked Packerbacker. "New nodule revealed metastatic cancer. ... I had cisplatin in the past and tolerated it with minor side effects. I’m still nervous, though. Wish me luck?"
An optimistic response came from a user in New Jersey: "I received 5-FU via a pump. ... Not fun, more of a pain in the butt. It turned out to not be a big deal at all," wrote IKickedIt.
"Good luck!" wrote FreeBird in Tampa, Fla. "Dad had carboplatin and Taxol [paclitaxel] for his previous lung cancer. He did really well with it!"
Patients and family members of patients described how to wear the pump, how to shower with the pump (by using plastic wrap and a shower head from a home improvement store), and how to disguise the pump (in a "cute backpack," suggested one user). Another user wrote, "Just kept the pump on my desk."
The new network boasts more than 10,000 registered users who pose questions and offer suggestions on topics both weighty and mundane.
"For those of you who had both options available, how did you decide between a lumpectomy and a mastectomy?" asked a mom from Cincinnati.
"Best advice for a caregiver who gets a bad cold?" queried Iibcarolek from California. "First of all, take care of yourself," advised Fizztig, who recommended frequent hand washing, using hand sanitizer, protecting sneezes, and getting enough rest.
By negotiating the well-organized website, users will find categories where they can share their experience and journey. Categories include "Oh, No," "Celebration," "Decision Point," and "Procedure or Surgery."
The website is organized by diagnosis. There is also a Pinboard where users can search, browse, or post inspirational photos and videos (similar to Pinterest).
One photo on the Pinboard is of a young girl wearing a scarf on her head with the following note from bcncnabb: "People stare. You all know this. You’ve all felt it in restaurants, at the grocery store, or any other place where people don’t get it. ... But wow, what people don’t notice is the fight, the spirit, and the grace that it takes to live with cancer."
Dr. Len Lichtenfeld, deputy chief medical officer of the American Cancer Society (ACS), endorsed the new online tool as an "exciting" use of technology to extend the reach of both the ACS and the oncology community. "We know that people who connect with each other in times like this feel more empowered, less anxious, and can approach this crucial time in their lives feeling supported," he noted.
The website’s exchange of practical tips reminded me of a caregiver’s lament about the difficulties of managing her husband’s percutaneous endoscopic gastrostomy (PEG) tube, which even the visiting in-home nurses didn’t really understand. In the exhausting days following his discharge after surgery for esophageal cancer, she taught herself how to triple flush the tube to prevent middle-of-the-night clogs.
Today, I would refer her to WhatNext, where I’m confident she would receive (and offer) pep talks and helpful advice.
In some cases, connections formed on WhatNext have led to offline friendships. The ACS highlighted a bond formed between MGM48 and MichaelV, both of whom have prostate cancer. "We compare notes and discuss life with cancer," said MGM48. "I’m happy to have a live sounding board sometimes.
"Let’s face it, no one outside really completely gets it," he said.
Dr. Freed is a clinical psychologist in Santa Barbara, Calif., and a medical journalist.
When a cancer patient from Menomonie, Wis., reached out to the American Cancer Society’s WhatNext online network seeking treatment reassurance, a ready-made support group jumped into action.
"Anyone familiar with 5-FU [5-fluorouracil], cetuximab (Erbitux), or carboplatin? Has anyone ever worn a pump?" asked Packerbacker. "New nodule revealed metastatic cancer. ... I had cisplatin in the past and tolerated it with minor side effects. I’m still nervous, though. Wish me luck?"
An optimistic response came from a user in New Jersey: "I received 5-FU via a pump. ... Not fun, more of a pain in the butt. It turned out to not be a big deal at all," wrote IKickedIt.
"Good luck!" wrote FreeBird in Tampa, Fla. "Dad had carboplatin and Taxol [paclitaxel] for his previous lung cancer. He did really well with it!"
Patients and family members of patients described how to wear the pump, how to shower with the pump (by using plastic wrap and a shower head from a home improvement store), and how to disguise the pump (in a "cute backpack," suggested one user). Another user wrote, "Just kept the pump on my desk."
The new network boasts more than 10,000 registered users who pose questions and offer suggestions on topics both weighty and mundane.
"For those of you who had both options available, how did you decide between a lumpectomy and a mastectomy?" asked a mom from Cincinnati.
"Best advice for a caregiver who gets a bad cold?" queried Iibcarolek from California. "First of all, take care of yourself," advised Fizztig, who recommended frequent hand washing, using hand sanitizer, protecting sneezes, and getting enough rest.
By negotiating the well-organized website, users will find categories where they can share their experience and journey. Categories include "Oh, No," "Celebration," "Decision Point," and "Procedure or Surgery."
The website is organized by diagnosis. There is also a Pinboard where users can search, browse, or post inspirational photos and videos (similar to Pinterest).
One photo on the Pinboard is of a young girl wearing a scarf on her head with the following note from bcncnabb: "People stare. You all know this. You’ve all felt it in restaurants, at the grocery store, or any other place where people don’t get it. ... But wow, what people don’t notice is the fight, the spirit, and the grace that it takes to live with cancer."
Dr. Len Lichtenfeld, deputy chief medical officer of the American Cancer Society (ACS), endorsed the new online tool as an "exciting" use of technology to extend the reach of both the ACS and the oncology community. "We know that people who connect with each other in times like this feel more empowered, less anxious, and can approach this crucial time in their lives feeling supported," he noted.
The website’s exchange of practical tips reminded me of a caregiver’s lament about the difficulties of managing her husband’s percutaneous endoscopic gastrostomy (PEG) tube, which even the visiting in-home nurses didn’t really understand. In the exhausting days following his discharge after surgery for esophageal cancer, she taught herself how to triple flush the tube to prevent middle-of-the-night clogs.
Today, I would refer her to WhatNext, where I’m confident she would receive (and offer) pep talks and helpful advice.
In some cases, connections formed on WhatNext have led to offline friendships. The ACS highlighted a bond formed between MGM48 and MichaelV, both of whom have prostate cancer. "We compare notes and discuss life with cancer," said MGM48. "I’m happy to have a live sounding board sometimes.
"Let’s face it, no one outside really completely gets it," he said.
Dr. Freed is a clinical psychologist in Santa Barbara, Calif., and a medical journalist.
When a cancer patient from Menomonie, Wis., reached out to the American Cancer Society’s WhatNext online network seeking treatment reassurance, a ready-made support group jumped into action.
"Anyone familiar with 5-FU [5-fluorouracil], cetuximab (Erbitux), or carboplatin? Has anyone ever worn a pump?" asked Packerbacker. "New nodule revealed metastatic cancer. ... I had cisplatin in the past and tolerated it with minor side effects. I’m still nervous, though. Wish me luck?"
An optimistic response came from a user in New Jersey: "I received 5-FU via a pump. ... Not fun, more of a pain in the butt. It turned out to not be a big deal at all," wrote IKickedIt.
"Good luck!" wrote FreeBird in Tampa, Fla. "Dad had carboplatin and Taxol [paclitaxel] for his previous lung cancer. He did really well with it!"
Patients and family members of patients described how to wear the pump, how to shower with the pump (by using plastic wrap and a shower head from a home improvement store), and how to disguise the pump (in a "cute backpack," suggested one user). Another user wrote, "Just kept the pump on my desk."
The new network boasts more than 10,000 registered users who pose questions and offer suggestions on topics both weighty and mundane.
"For those of you who had both options available, how did you decide between a lumpectomy and a mastectomy?" asked a mom from Cincinnati.
"Best advice for a caregiver who gets a bad cold?" queried Iibcarolek from California. "First of all, take care of yourself," advised Fizztig, who recommended frequent hand washing, using hand sanitizer, protecting sneezes, and getting enough rest.
By negotiating the well-organized website, users will find categories where they can share their experience and journey. Categories include "Oh, No," "Celebration," "Decision Point," and "Procedure or Surgery."
The website is organized by diagnosis. There is also a Pinboard where users can search, browse, or post inspirational photos and videos (similar to Pinterest).
One photo on the Pinboard is of a young girl wearing a scarf on her head with the following note from bcncnabb: "People stare. You all know this. You’ve all felt it in restaurants, at the grocery store, or any other place where people don’t get it. ... But wow, what people don’t notice is the fight, the spirit, and the grace that it takes to live with cancer."
Dr. Len Lichtenfeld, deputy chief medical officer of the American Cancer Society (ACS), endorsed the new online tool as an "exciting" use of technology to extend the reach of both the ACS and the oncology community. "We know that people who connect with each other in times like this feel more empowered, less anxious, and can approach this crucial time in their lives feeling supported," he noted.
The website’s exchange of practical tips reminded me of a caregiver’s lament about the difficulties of managing her husband’s percutaneous endoscopic gastrostomy (PEG) tube, which even the visiting in-home nurses didn’t really understand. In the exhausting days following his discharge after surgery for esophageal cancer, she taught herself how to triple flush the tube to prevent middle-of-the-night clogs.
Today, I would refer her to WhatNext, where I’m confident she would receive (and offer) pep talks and helpful advice.
In some cases, connections formed on WhatNext have led to offline friendships. The ACS highlighted a bond formed between MGM48 and MichaelV, both of whom have prostate cancer. "We compare notes and discuss life with cancer," said MGM48. "I’m happy to have a live sounding board sometimes.
"Let’s face it, no one outside really completely gets it," he said.
Dr. Freed is a clinical psychologist in Santa Barbara, Calif., and a medical journalist.
Multidisciplinary Palliative-Care Consults Help Reduce Hospital Readmissions
Research on seriously ill, hospitalized, Medicare-age patients finds that those who received inpatient consultations from a multidisciplinary, palliative-care team (including a physician, nurse, and social worker) had lower 30-day hospital readmission rates.1 Ten percent of discharged patients who received the palliative-care consult were readmitted within 30 days at an urban HMO medical center in Los Angeles County during the same period, even though they were sicker than the overall discharged population.
Receipt of hospice care or home-based palliative-care services following discharge was also associated with significantly lower rates of readmissions, suggesting opportunities for systemic cost savings from earlier access to longitudinal, or ongoing, palliative-care services, says Susan Enguidanos, MPH, PhD, assistant professor of gerontology at the University of Southern California in Los Angeles. Patients discharged from the hospital without any follow-up care in the home had higher odds of readmission.
“Hospitals and medical centers should seriously consider an inpatient palliative care consultation team for many reasons, mostly arising from findings from other studies that have demonstrated improved quality of life, pain and symptom management, satisfaction with medical care, and other promising outcomes,” Dr. Enguidanos says. “Our study suggests that longitudinal palliative care is also associated with the lower readmission rate.”
References
- Enguidanos S, Vesper E, Lorenz K. 30-day readmissions among seriously ill older adults. J Palliat Med. 2012;15(12):1356-1361.
- The Advisory Board Company. Mastering the cardiovascular care continuum: strategies for bridging divides among providers and across time. The Advisory Board Company website. Available at: http://www.advisory.com/Research/Cardiovascular-Roundtable/Studies/2012/Mastering-the-Cardiovascular-Care-Continuum. Accessed Jan. 8, 2013.
- Misky G, Carlson T, Klem P, et al. Development and implementation of a clinical care pathway for acute VTE reduces hospital utilization and cost at an urban tertiary care center [abstract]. J Hosp Med. 2012;7 Suppl 2:S66-S67.
- Versel N. Health IT holds key to better care integration. Information Week website. Available at: http://www.informationweek.com/healthcare/interoperability/health-it-holds-key-to-better-care-integ/240012443. Accessed Jan. 8, 2013.
- Office of Inspector General. Early Assessment Finds That CMS Faces Obstacles in Overseeing the Medicare EHR Incentive Program. Office of Inspector General website. Available at: https://oig.hhs.gov/oei/reports/oei-05-11-00250.asp. Accessed Jan. 8, 2013.
Research on seriously ill, hospitalized, Medicare-age patients finds that those who received inpatient consultations from a multidisciplinary, palliative-care team (including a physician, nurse, and social worker) had lower 30-day hospital readmission rates.1 Ten percent of discharged patients who received the palliative-care consult were readmitted within 30 days at an urban HMO medical center in Los Angeles County during the same period, even though they were sicker than the overall discharged population.
Receipt of hospice care or home-based palliative-care services following discharge was also associated with significantly lower rates of readmissions, suggesting opportunities for systemic cost savings from earlier access to longitudinal, or ongoing, palliative-care services, says Susan Enguidanos, MPH, PhD, assistant professor of gerontology at the University of Southern California in Los Angeles. Patients discharged from the hospital without any follow-up care in the home had higher odds of readmission.
“Hospitals and medical centers should seriously consider an inpatient palliative care consultation team for many reasons, mostly arising from findings from other studies that have demonstrated improved quality of life, pain and symptom management, satisfaction with medical care, and other promising outcomes,” Dr. Enguidanos says. “Our study suggests that longitudinal palliative care is also associated with the lower readmission rate.”
References
- Enguidanos S, Vesper E, Lorenz K. 30-day readmissions among seriously ill older adults. J Palliat Med. 2012;15(12):1356-1361.
- The Advisory Board Company. Mastering the cardiovascular care continuum: strategies for bridging divides among providers and across time. The Advisory Board Company website. Available at: http://www.advisory.com/Research/Cardiovascular-Roundtable/Studies/2012/Mastering-the-Cardiovascular-Care-Continuum. Accessed Jan. 8, 2013.
- Misky G, Carlson T, Klem P, et al. Development and implementation of a clinical care pathway for acute VTE reduces hospital utilization and cost at an urban tertiary care center [abstract]. J Hosp Med. 2012;7 Suppl 2:S66-S67.
- Versel N. Health IT holds key to better care integration. Information Week website. Available at: http://www.informationweek.com/healthcare/interoperability/health-it-holds-key-to-better-care-integ/240012443. Accessed Jan. 8, 2013.
- Office of Inspector General. Early Assessment Finds That CMS Faces Obstacles in Overseeing the Medicare EHR Incentive Program. Office of Inspector General website. Available at: https://oig.hhs.gov/oei/reports/oei-05-11-00250.asp. Accessed Jan. 8, 2013.
Research on seriously ill, hospitalized, Medicare-age patients finds that those who received inpatient consultations from a multidisciplinary, palliative-care team (including a physician, nurse, and social worker) had lower 30-day hospital readmission rates.1 Ten percent of discharged patients who received the palliative-care consult were readmitted within 30 days at an urban HMO medical center in Los Angeles County during the same period, even though they were sicker than the overall discharged population.
Receipt of hospice care or home-based palliative-care services following discharge was also associated with significantly lower rates of readmissions, suggesting opportunities for systemic cost savings from earlier access to longitudinal, or ongoing, palliative-care services, says Susan Enguidanos, MPH, PhD, assistant professor of gerontology at the University of Southern California in Los Angeles. Patients discharged from the hospital without any follow-up care in the home had higher odds of readmission.
“Hospitals and medical centers should seriously consider an inpatient palliative care consultation team for many reasons, mostly arising from findings from other studies that have demonstrated improved quality of life, pain and symptom management, satisfaction with medical care, and other promising outcomes,” Dr. Enguidanos says. “Our study suggests that longitudinal palliative care is also associated with the lower readmission rate.”
References
- Enguidanos S, Vesper E, Lorenz K. 30-day readmissions among seriously ill older adults. J Palliat Med. 2012;15(12):1356-1361.
- The Advisory Board Company. Mastering the cardiovascular care continuum: strategies for bridging divides among providers and across time. The Advisory Board Company website. Available at: http://www.advisory.com/Research/Cardiovascular-Roundtable/Studies/2012/Mastering-the-Cardiovascular-Care-Continuum. Accessed Jan. 8, 2013.
- Misky G, Carlson T, Klem P, et al. Development and implementation of a clinical care pathway for acute VTE reduces hospital utilization and cost at an urban tertiary care center [abstract]. J Hosp Med. 2012;7 Suppl 2:S66-S67.
- Versel N. Health IT holds key to better care integration. Information Week website. Available at: http://www.informationweek.com/healthcare/interoperability/health-it-holds-key-to-better-care-integ/240012443. Accessed Jan. 8, 2013.
- Office of Inspector General. Early Assessment Finds That CMS Faces Obstacles in Overseeing the Medicare EHR Incentive Program. Office of Inspector General website. Available at: https://oig.hhs.gov/oei/reports/oei-05-11-00250.asp. Accessed Jan. 8, 2013.
Who Decides What Comes Next?
The past year has been quite a roller coaster ride for many of my friends and their families. Several were caught in the throes of Hurricane Sandy and spent days without power and water; many lost their homes and possessions. Others dealt with the loss of a parent or sibling (unrelated to the storm). We, as a nation, faced the unimaginable tragedy of the violent death of children at the hands of a gunman.
While we singularly or collectively grieved these losses, many of us reflected on the reality that our time here on earth is limited. And that started me thinking about death and dying and how we deal (or not) with that juncture in our lives. When it comes to dying, we are often ill prepared for this part of life’s journey. When we are young, we have the misconception (wonderful though it may be) that we are invincible and immortal. We sail through our days seemingly without a care in the world, believing that we will live forever. But, alas, at some point we face the reality of life, when we witness or experience the death of a loved one. And unfortunately, many are left with the responsibility to deal with end-of-life decisions, without ever having had the discussion of what the person wanted in regard to this process. Even as I write this, I am counseling a neighbor whose 78-year-old husband—suddenly and seriously ill—is now in a drug-induced coma. He has a chronic illness, but they never had the conversation.
There is a lot to consider—and tackling the question is difficult, because much of what needs to be identified or clarified is a sensitive subject. Many are not comfortable asking for or sharing information considered very private. But it is important that our wishes are known, so that in the event we cannot act on our own behalf, our preferences for care and treatment are respected. Moreover, we as heath care providers must embrace that Primum non nocere(First do no harm) might include not initiating treatments in an attempt to prolong life when the outcome is inevitable.
Health care providers have spent their careers dedicated to preventing or treating illnesses and eliminating suffering. Advances in technology have allowed us to prolong life. However, many of these advances do not improve the quality of life; rather, they often only delay the process of dying. It is important that we understand which and how much of these technologies our loved ones (and we ourselves) want utilized, or discontinued, when our disease or age has reached the point when it is time to realize our end is near. But the time to discuss and plan for that phase of our life must occur long before the crisis hits.
End-of-life planning is not a new concept. Since the 1980s, the Agency for Healthcare Research and Quality (AHRQ) has both funded and supported dozens of research projects on the topic of end-of-life care. The research funded by AHRQ indicates that many people have not participated in advance planning, although they are interested in and willing to discuss end-of-life care.1 Worse yet, in many instances, researchers found that care at the end of life sometimes appears to be inconsistent with the patient’s preference to forgo life-sustaining treatment, and patients may receive care they do not want.2
In 1997, the NIH designated the National Institute of Nursing Research (NINR) as the lead institute for end-of-life research to advance the science of palliative and end-of-life care and to lead the way in addressing some of the most critical challenges in clinical care.3 This is further evidence that end-of-life care is an important topic to be discussed, and we, the health care providers, need to initiate the conversation.
The conversation is not reserved for the terminally ill; we must also discuss plans with those who have a chronic illness and even with those who are in “perfect health.” These plans, also known as advance directives, can allay many of the anxieties that patients and families have regarding what to do in the event the patient cannot speak for him- or herself, and instill confidence that the wishes of the patient will be honored.
Advance directives are not reserved for the old and infirm. All of us must plan for the unexpected—the sudden life—threatening illness or injury or freak accident—that leaves us unable to communicate what treatments we want or do not and when we want treatments discontinued. Initiating the discussion is not easy for some. For others, when we see or hear about a tragedy that leaves someone paralyzed or in a coma, we are quick to voice what we would want in a similar situation. That is the perfect lead—in to the discussion regarding our wishes. A structured process for an end-of-life discussion has been outlined by AHRQ researchers.1 Theirs is a simple five-step plan:
1. Initiate a guided discussion.
2. Introduce the subject of advance care planning and offer information.
3. Prepare and complete advance care planning documents.
4. Review the patient’s preferences on a regular basis and update documentation.
5. Apply the patient’s desires to actual circumstances.1
(A detailed discussion of these steps can be found at www.ahrq.gov/RESEARCH/endliferia/endria.pdf.)
Now is the time to assist friends, family members, and your patients in discussing and documenting their plans. Take the time to document your own advance directives. Let your family and your health care provider know what you want, and have it put in your health record. Review it annually, in the event that there have been changes in your family or in technology. Do not miss the opportunity to let your family know what you want.
Do you have a plan in place? Share your thoughts by emailing [email protected].
References
1. Kass-Bartelmes BL, Hughes R, Rutherford MK. Advance care planning: preferences for care at the end of life. Rockville, MD: Agency for Healthcare Research and Quality; 2003. Research in Action Issue #12. AHRQ Pub No. 03-0018.
2. Teno JM, Licks S, Lynn J, et al. Do advance directives provide instructions that direct care? J Am Geriatr Soc. 1997;45(4):508-512.
3. NIH National Institute of Nursing Research. Spotlight on end-of-life research. www.ninr .nih.gov/researchandfunding/spotlight-on-end-of-life-research. Accessed January 22, 2013.
The past year has been quite a roller coaster ride for many of my friends and their families. Several were caught in the throes of Hurricane Sandy and spent days without power and water; many lost their homes and possessions. Others dealt with the loss of a parent or sibling (unrelated to the storm). We, as a nation, faced the unimaginable tragedy of the violent death of children at the hands of a gunman.
While we singularly or collectively grieved these losses, many of us reflected on the reality that our time here on earth is limited. And that started me thinking about death and dying and how we deal (or not) with that juncture in our lives. When it comes to dying, we are often ill prepared for this part of life’s journey. When we are young, we have the misconception (wonderful though it may be) that we are invincible and immortal. We sail through our days seemingly without a care in the world, believing that we will live forever. But, alas, at some point we face the reality of life, when we witness or experience the death of a loved one. And unfortunately, many are left with the responsibility to deal with end-of-life decisions, without ever having had the discussion of what the person wanted in regard to this process. Even as I write this, I am counseling a neighbor whose 78-year-old husband—suddenly and seriously ill—is now in a drug-induced coma. He has a chronic illness, but they never had the conversation.
There is a lot to consider—and tackling the question is difficult, because much of what needs to be identified or clarified is a sensitive subject. Many are not comfortable asking for or sharing information considered very private. But it is important that our wishes are known, so that in the event we cannot act on our own behalf, our preferences for care and treatment are respected. Moreover, we as heath care providers must embrace that Primum non nocere(First do no harm) might include not initiating treatments in an attempt to prolong life when the outcome is inevitable.
Health care providers have spent their careers dedicated to preventing or treating illnesses and eliminating suffering. Advances in technology have allowed us to prolong life. However, many of these advances do not improve the quality of life; rather, they often only delay the process of dying. It is important that we understand which and how much of these technologies our loved ones (and we ourselves) want utilized, or discontinued, when our disease or age has reached the point when it is time to realize our end is near. But the time to discuss and plan for that phase of our life must occur long before the crisis hits.
End-of-life planning is not a new concept. Since the 1980s, the Agency for Healthcare Research and Quality (AHRQ) has both funded and supported dozens of research projects on the topic of end-of-life care. The research funded by AHRQ indicates that many people have not participated in advance planning, although they are interested in and willing to discuss end-of-life care.1 Worse yet, in many instances, researchers found that care at the end of life sometimes appears to be inconsistent with the patient’s preference to forgo life-sustaining treatment, and patients may receive care they do not want.2
In 1997, the NIH designated the National Institute of Nursing Research (NINR) as the lead institute for end-of-life research to advance the science of palliative and end-of-life care and to lead the way in addressing some of the most critical challenges in clinical care.3 This is further evidence that end-of-life care is an important topic to be discussed, and we, the health care providers, need to initiate the conversation.
The conversation is not reserved for the terminally ill; we must also discuss plans with those who have a chronic illness and even with those who are in “perfect health.” These plans, also known as advance directives, can allay many of the anxieties that patients and families have regarding what to do in the event the patient cannot speak for him- or herself, and instill confidence that the wishes of the patient will be honored.
Advance directives are not reserved for the old and infirm. All of us must plan for the unexpected—the sudden life—threatening illness or injury or freak accident—that leaves us unable to communicate what treatments we want or do not and when we want treatments discontinued. Initiating the discussion is not easy for some. For others, when we see or hear about a tragedy that leaves someone paralyzed or in a coma, we are quick to voice what we would want in a similar situation. That is the perfect lead—in to the discussion regarding our wishes. A structured process for an end-of-life discussion has been outlined by AHRQ researchers.1 Theirs is a simple five-step plan:
1. Initiate a guided discussion.
2. Introduce the subject of advance care planning and offer information.
3. Prepare and complete advance care planning documents.
4. Review the patient’s preferences on a regular basis and update documentation.
5. Apply the patient’s desires to actual circumstances.1
(A detailed discussion of these steps can be found at www.ahrq.gov/RESEARCH/endliferia/endria.pdf.)
Now is the time to assist friends, family members, and your patients in discussing and documenting their plans. Take the time to document your own advance directives. Let your family and your health care provider know what you want, and have it put in your health record. Review it annually, in the event that there have been changes in your family or in technology. Do not miss the opportunity to let your family know what you want.
Do you have a plan in place? Share your thoughts by emailing [email protected].
References
1. Kass-Bartelmes BL, Hughes R, Rutherford MK. Advance care planning: preferences for care at the end of life. Rockville, MD: Agency for Healthcare Research and Quality; 2003. Research in Action Issue #12. AHRQ Pub No. 03-0018.
2. Teno JM, Licks S, Lynn J, et al. Do advance directives provide instructions that direct care? J Am Geriatr Soc. 1997;45(4):508-512.
3. NIH National Institute of Nursing Research. Spotlight on end-of-life research. www.ninr .nih.gov/researchandfunding/spotlight-on-end-of-life-research. Accessed January 22, 2013.
The past year has been quite a roller coaster ride for many of my friends and their families. Several were caught in the throes of Hurricane Sandy and spent days without power and water; many lost their homes and possessions. Others dealt with the loss of a parent or sibling (unrelated to the storm). We, as a nation, faced the unimaginable tragedy of the violent death of children at the hands of a gunman.
While we singularly or collectively grieved these losses, many of us reflected on the reality that our time here on earth is limited. And that started me thinking about death and dying and how we deal (or not) with that juncture in our lives. When it comes to dying, we are often ill prepared for this part of life’s journey. When we are young, we have the misconception (wonderful though it may be) that we are invincible and immortal. We sail through our days seemingly without a care in the world, believing that we will live forever. But, alas, at some point we face the reality of life, when we witness or experience the death of a loved one. And unfortunately, many are left with the responsibility to deal with end-of-life decisions, without ever having had the discussion of what the person wanted in regard to this process. Even as I write this, I am counseling a neighbor whose 78-year-old husband—suddenly and seriously ill—is now in a drug-induced coma. He has a chronic illness, but they never had the conversation.
There is a lot to consider—and tackling the question is difficult, because much of what needs to be identified or clarified is a sensitive subject. Many are not comfortable asking for or sharing information considered very private. But it is important that our wishes are known, so that in the event we cannot act on our own behalf, our preferences for care and treatment are respected. Moreover, we as heath care providers must embrace that Primum non nocere(First do no harm) might include not initiating treatments in an attempt to prolong life when the outcome is inevitable.
Health care providers have spent their careers dedicated to preventing or treating illnesses and eliminating suffering. Advances in technology have allowed us to prolong life. However, many of these advances do not improve the quality of life; rather, they often only delay the process of dying. It is important that we understand which and how much of these technologies our loved ones (and we ourselves) want utilized, or discontinued, when our disease or age has reached the point when it is time to realize our end is near. But the time to discuss and plan for that phase of our life must occur long before the crisis hits.
End-of-life planning is not a new concept. Since the 1980s, the Agency for Healthcare Research and Quality (AHRQ) has both funded and supported dozens of research projects on the topic of end-of-life care. The research funded by AHRQ indicates that many people have not participated in advance planning, although they are interested in and willing to discuss end-of-life care.1 Worse yet, in many instances, researchers found that care at the end of life sometimes appears to be inconsistent with the patient’s preference to forgo life-sustaining treatment, and patients may receive care they do not want.2
In 1997, the NIH designated the National Institute of Nursing Research (NINR) as the lead institute for end-of-life research to advance the science of palliative and end-of-life care and to lead the way in addressing some of the most critical challenges in clinical care.3 This is further evidence that end-of-life care is an important topic to be discussed, and we, the health care providers, need to initiate the conversation.
The conversation is not reserved for the terminally ill; we must also discuss plans with those who have a chronic illness and even with those who are in “perfect health.” These plans, also known as advance directives, can allay many of the anxieties that patients and families have regarding what to do in the event the patient cannot speak for him- or herself, and instill confidence that the wishes of the patient will be honored.
Advance directives are not reserved for the old and infirm. All of us must plan for the unexpected—the sudden life—threatening illness or injury or freak accident—that leaves us unable to communicate what treatments we want or do not and when we want treatments discontinued. Initiating the discussion is not easy for some. For others, when we see or hear about a tragedy that leaves someone paralyzed or in a coma, we are quick to voice what we would want in a similar situation. That is the perfect lead—in to the discussion regarding our wishes. A structured process for an end-of-life discussion has been outlined by AHRQ researchers.1 Theirs is a simple five-step plan:
1. Initiate a guided discussion.
2. Introduce the subject of advance care planning and offer information.
3. Prepare and complete advance care planning documents.
4. Review the patient’s preferences on a regular basis and update documentation.
5. Apply the patient’s desires to actual circumstances.1
(A detailed discussion of these steps can be found at www.ahrq.gov/RESEARCH/endliferia/endria.pdf.)
Now is the time to assist friends, family members, and your patients in discussing and documenting their plans. Take the time to document your own advance directives. Let your family and your health care provider know what you want, and have it put in your health record. Review it annually, in the event that there have been changes in your family or in technology. Do not miss the opportunity to let your family know what you want.
Do you have a plan in place? Share your thoughts by emailing [email protected].
References
1. Kass-Bartelmes BL, Hughes R, Rutherford MK. Advance care planning: preferences for care at the end of life. Rockville, MD: Agency for Healthcare Research and Quality; 2003. Research in Action Issue #12. AHRQ Pub No. 03-0018.
2. Teno JM, Licks S, Lynn J, et al. Do advance directives provide instructions that direct care? J Am Geriatr Soc. 1997;45(4):508-512.
3. NIH National Institute of Nursing Research. Spotlight on end-of-life research. www.ninr .nih.gov/researchandfunding/spotlight-on-end-of-life-research. Accessed January 22, 2013.
No heroic measures
The page came around 2 a.m. It had been a very busy night, with a dozen or so surgical consults from an overflowing emergency department in a large urban hospital. Most of the patients referred to the surgical service were immigrants with a limited knowledge of English. Most were elderly. And most were alone.
I was 2 months out of medical school, on call without a senior resident and with staff backup at home. I struggled to remain awake while catering to the seemingly endless pages from the ward and the emergency department. It was a rite of passage that every intern has had to endure.
The patient was an 85-year-old Cantonese-speaking woman transferred to the emergency department by ambulance from her nursing home. The accompanying note, mostly illegible, suggested that she had been complaining of abdominal pain and distention for the past 2 days and had been initially managed with laxatives.
That afternoon, however, her level of consciousness began to deteriorate and she became febrile and hypotensive, so she was transferred to our hospital. She had a history of atrial fibrillation, hypertension, and advanced Alzheimer’s dementia. She also had an accompanying "Do Not Resuscitate/No Life Support" form.
A CT scan of the abdomen arranged by the emergency department physician identified a clot in the superior mesenteric artery and extensive ischemic changes in most of her small bowel. She had a high white blood cell count, and her serum lactate was alarmingly elevated, despite several boluses of intravenous crystalloid solution. She was also becoming hemodynamically unstable.
I immediately called the staff surgeon to advocate for an emergency laparotomy. Given the patient’s multiple comorbidities and grim prognosis, we decided not to operate. The staff surgeon’s instructions were as abrupt as they were clear: "Turf her to medicine; she’s palliative."
The medical senior resident, however, would have none of it. Equally overwhelmed with consults and not in a particularly good mood, he refused to even see the patient. If she was expected to die overnight because of a surgical issue, he argued, then the surgery team was just as equipped as he to take care of her. The emergency department physician agreed, and so the patient was admitted to the surgical service for her few remaining hours of life.
What followed was agonizing for the entire surgical team. The nurses on the ward were asking some very reasonable questions that I could not answer: Do we keep titrating the patient’s oxygen to keep up with her desaturation? What do we give her for pain control? How do we manage her shortness of breath? Should we continue giving her intravenous fluids? Can you help us find her family members? Should we keep the bladder catheter in place?
I strained to remember what we had learned in our few palliative care lectures back in medical school. Intravenous fluids were OK, but oxygen was not. Or was it the other way around? Google, the ever-faithful guardian of the panicking intern, was unhelpful. I could not find a reputable online resource to help guide the management of this patient and, given the other sick patients who needed attention, I could not sit down to perform a thorough online literature search. The hospital’s switchboard was equally disappointing – there were no palliative care physicians on call, and the palliative team only accepted pages after 8 a.m., an appalling 5 hours away.
Eventually, I decided to support the patient with an oxygen mask and to continue her intravenous fluid resuscitation. I gave her plenty of narcotics and antiemetics to soothe the discomfort. We were unable to reach any family members despite several attempts. She died just after 5 a.m., without any visible signs of discomfort. It was the first death certificate I had ever signed.
Reflecting on this experience, I was upset at my inability to quickly and adequately care for this patient’s end-of-life needs. I was also angry that I had not prepared myself appropriately for that night on call, and I would have done several things differently had I had the chance.
General surgeons take immense pride in being internists who can operate. We enjoy debating antibiotic, diuretics, and anticoagulation choices with the specialists consulting on our patients, but we often simply "switch off" whenever the patient is labeled "palliative" or "end of life" – relying entirely on the palliative care team for help. And yet, we’re the ones who admit, operate on, and develop therapeutic relationships with those patients and their families. It just doesn’t seem right, but there are several reasons for this dilemma.
First, the fast pace and busy nature of any surgical service means that trainees and staff are under a lot of pressure to round as quickly and efficiently as possible and get to the operating room early in the morning. Given current trainee work-hour limitations and mandated hand-over, today’s junior staff feel this pressure all the more acutely. There simply isn’t enough time or opportunity for continuity of care. Surgical teams can’t engage palliative patients and their families in the way that dedicated palliative care teams can, and so surgical teams are often more comfortable delegating this responsibility entirely to their palliative care colleagues.
Second, there is an emotional burden to caring for palliative patients and their families. Palliation and end-of-life care imply that surgery is neither possible nor advisable any longer. It can be difficult for the surgeon to make that mental segue and assume the dual role of the psychological and medical care giver. Just as surgeons prefer that cardiologists manage their patients’ arrhythmias and nephrologists their patients’ kidney failures, they may prefer that palliative care specialists manage the emotional and medical needs of the patients on whom they can no longer operate.
Furthermore, surgical trainees have limited exposure to palliative care in the clinical setting. As the field of surgery becomes progressively more complex, palliative care training has not been at the forefront of the educational agenda. While it is taught in medical schools and in the surgical residency curriculum, there are few formalized core rotations that offer the surgical resident an insight into the clinical applications of palliative care, except for those who are particularly interested and seek such learning opportunities out in the form of an elective rotation.
The development of dedicated and multidisciplinary palliative care teams that include staff, nurse practitioners, and palliative fellows has further limited surgical resident exposure to the field. Simply put, the current medical system encourages surgical trainees to sign off on the surgical palliative patient.
This, however, isn’t good enough. Palliative care of the surgical patient needs to be a pillar of surgical training. Trainees need to be as comfortable with managing the symptoms of the dying surgical patient as they are administering fluid boluses and ordering narcotics for postoperative pain, especially late at night when there is no palliative care support.
The American College of Surgeons has responded to this challenge by developing numerous educational initiatives through its Palliative Workgroup. It has published a comprehensive surgical palliative care self-study guide specifically for residents (Surgical Palliative Care: A Residents Guide, available for free download).
There are also several regularly updated online palliative care references available either through subscription websites such as UpToDate or for free, such as the Medical College of Wisconsin’s End of Life/Palliative Education Resource Center, which offers "Fast Facts" on the management of the dying patient.
Palliative care is widely accepted today as an important clinical discipline that provides vital support to a growing proportion of the patient population. Surgical training must keep pace with this evolving reality because, at the end of the day, to be without agony and discomfort is a standard of care that every dying patient expects – not just a heroic measure that we can simply ignore.
Dr. Kayssi is a PGY IV in the general surgery program at the University of Toronto.
Dr. Easson is an ACS Fellow and assistant professor in the department of surgery and the Institute of Health, Policy, Management and Evaluation at the University of Toronto.
The page came around 2 a.m. It had been a very busy night, with a dozen or so surgical consults from an overflowing emergency department in a large urban hospital. Most of the patients referred to the surgical service were immigrants with a limited knowledge of English. Most were elderly. And most were alone.
I was 2 months out of medical school, on call without a senior resident and with staff backup at home. I struggled to remain awake while catering to the seemingly endless pages from the ward and the emergency department. It was a rite of passage that every intern has had to endure.
The patient was an 85-year-old Cantonese-speaking woman transferred to the emergency department by ambulance from her nursing home. The accompanying note, mostly illegible, suggested that she had been complaining of abdominal pain and distention for the past 2 days and had been initially managed with laxatives.
That afternoon, however, her level of consciousness began to deteriorate and she became febrile and hypotensive, so she was transferred to our hospital. She had a history of atrial fibrillation, hypertension, and advanced Alzheimer’s dementia. She also had an accompanying "Do Not Resuscitate/No Life Support" form.
A CT scan of the abdomen arranged by the emergency department physician identified a clot in the superior mesenteric artery and extensive ischemic changes in most of her small bowel. She had a high white blood cell count, and her serum lactate was alarmingly elevated, despite several boluses of intravenous crystalloid solution. She was also becoming hemodynamically unstable.
I immediately called the staff surgeon to advocate for an emergency laparotomy. Given the patient’s multiple comorbidities and grim prognosis, we decided not to operate. The staff surgeon’s instructions were as abrupt as they were clear: "Turf her to medicine; she’s palliative."
The medical senior resident, however, would have none of it. Equally overwhelmed with consults and not in a particularly good mood, he refused to even see the patient. If she was expected to die overnight because of a surgical issue, he argued, then the surgery team was just as equipped as he to take care of her. The emergency department physician agreed, and so the patient was admitted to the surgical service for her few remaining hours of life.
What followed was agonizing for the entire surgical team. The nurses on the ward were asking some very reasonable questions that I could not answer: Do we keep titrating the patient’s oxygen to keep up with her desaturation? What do we give her for pain control? How do we manage her shortness of breath? Should we continue giving her intravenous fluids? Can you help us find her family members? Should we keep the bladder catheter in place?
I strained to remember what we had learned in our few palliative care lectures back in medical school. Intravenous fluids were OK, but oxygen was not. Or was it the other way around? Google, the ever-faithful guardian of the panicking intern, was unhelpful. I could not find a reputable online resource to help guide the management of this patient and, given the other sick patients who needed attention, I could not sit down to perform a thorough online literature search. The hospital’s switchboard was equally disappointing – there were no palliative care physicians on call, and the palliative team only accepted pages after 8 a.m., an appalling 5 hours away.
Eventually, I decided to support the patient with an oxygen mask and to continue her intravenous fluid resuscitation. I gave her plenty of narcotics and antiemetics to soothe the discomfort. We were unable to reach any family members despite several attempts. She died just after 5 a.m., without any visible signs of discomfort. It was the first death certificate I had ever signed.
Reflecting on this experience, I was upset at my inability to quickly and adequately care for this patient’s end-of-life needs. I was also angry that I had not prepared myself appropriately for that night on call, and I would have done several things differently had I had the chance.
General surgeons take immense pride in being internists who can operate. We enjoy debating antibiotic, diuretics, and anticoagulation choices with the specialists consulting on our patients, but we often simply "switch off" whenever the patient is labeled "palliative" or "end of life" – relying entirely on the palliative care team for help. And yet, we’re the ones who admit, operate on, and develop therapeutic relationships with those patients and their families. It just doesn’t seem right, but there are several reasons for this dilemma.
First, the fast pace and busy nature of any surgical service means that trainees and staff are under a lot of pressure to round as quickly and efficiently as possible and get to the operating room early in the morning. Given current trainee work-hour limitations and mandated hand-over, today’s junior staff feel this pressure all the more acutely. There simply isn’t enough time or opportunity for continuity of care. Surgical teams can’t engage palliative patients and their families in the way that dedicated palliative care teams can, and so surgical teams are often more comfortable delegating this responsibility entirely to their palliative care colleagues.
Second, there is an emotional burden to caring for palliative patients and their families. Palliation and end-of-life care imply that surgery is neither possible nor advisable any longer. It can be difficult for the surgeon to make that mental segue and assume the dual role of the psychological and medical care giver. Just as surgeons prefer that cardiologists manage their patients’ arrhythmias and nephrologists their patients’ kidney failures, they may prefer that palliative care specialists manage the emotional and medical needs of the patients on whom they can no longer operate.
Furthermore, surgical trainees have limited exposure to palliative care in the clinical setting. As the field of surgery becomes progressively more complex, palliative care training has not been at the forefront of the educational agenda. While it is taught in medical schools and in the surgical residency curriculum, there are few formalized core rotations that offer the surgical resident an insight into the clinical applications of palliative care, except for those who are particularly interested and seek such learning opportunities out in the form of an elective rotation.
The development of dedicated and multidisciplinary palliative care teams that include staff, nurse practitioners, and palliative fellows has further limited surgical resident exposure to the field. Simply put, the current medical system encourages surgical trainees to sign off on the surgical palliative patient.
This, however, isn’t good enough. Palliative care of the surgical patient needs to be a pillar of surgical training. Trainees need to be as comfortable with managing the symptoms of the dying surgical patient as they are administering fluid boluses and ordering narcotics for postoperative pain, especially late at night when there is no palliative care support.
The American College of Surgeons has responded to this challenge by developing numerous educational initiatives through its Palliative Workgroup. It has published a comprehensive surgical palliative care self-study guide specifically for residents (Surgical Palliative Care: A Residents Guide, available for free download).
There are also several regularly updated online palliative care references available either through subscription websites such as UpToDate or for free, such as the Medical College of Wisconsin’s End of Life/Palliative Education Resource Center, which offers "Fast Facts" on the management of the dying patient.
Palliative care is widely accepted today as an important clinical discipline that provides vital support to a growing proportion of the patient population. Surgical training must keep pace with this evolving reality because, at the end of the day, to be without agony and discomfort is a standard of care that every dying patient expects – not just a heroic measure that we can simply ignore.
Dr. Kayssi is a PGY IV in the general surgery program at the University of Toronto.
Dr. Easson is an ACS Fellow and assistant professor in the department of surgery and the Institute of Health, Policy, Management and Evaluation at the University of Toronto.
The page came around 2 a.m. It had been a very busy night, with a dozen or so surgical consults from an overflowing emergency department in a large urban hospital. Most of the patients referred to the surgical service were immigrants with a limited knowledge of English. Most were elderly. And most were alone.
I was 2 months out of medical school, on call without a senior resident and with staff backup at home. I struggled to remain awake while catering to the seemingly endless pages from the ward and the emergency department. It was a rite of passage that every intern has had to endure.
The patient was an 85-year-old Cantonese-speaking woman transferred to the emergency department by ambulance from her nursing home. The accompanying note, mostly illegible, suggested that she had been complaining of abdominal pain and distention for the past 2 days and had been initially managed with laxatives.
That afternoon, however, her level of consciousness began to deteriorate and she became febrile and hypotensive, so she was transferred to our hospital. She had a history of atrial fibrillation, hypertension, and advanced Alzheimer’s dementia. She also had an accompanying "Do Not Resuscitate/No Life Support" form.
A CT scan of the abdomen arranged by the emergency department physician identified a clot in the superior mesenteric artery and extensive ischemic changes in most of her small bowel. She had a high white blood cell count, and her serum lactate was alarmingly elevated, despite several boluses of intravenous crystalloid solution. She was also becoming hemodynamically unstable.
I immediately called the staff surgeon to advocate for an emergency laparotomy. Given the patient’s multiple comorbidities and grim prognosis, we decided not to operate. The staff surgeon’s instructions were as abrupt as they were clear: "Turf her to medicine; she’s palliative."
The medical senior resident, however, would have none of it. Equally overwhelmed with consults and not in a particularly good mood, he refused to even see the patient. If she was expected to die overnight because of a surgical issue, he argued, then the surgery team was just as equipped as he to take care of her. The emergency department physician agreed, and so the patient was admitted to the surgical service for her few remaining hours of life.
What followed was agonizing for the entire surgical team. The nurses on the ward were asking some very reasonable questions that I could not answer: Do we keep titrating the patient’s oxygen to keep up with her desaturation? What do we give her for pain control? How do we manage her shortness of breath? Should we continue giving her intravenous fluids? Can you help us find her family members? Should we keep the bladder catheter in place?
I strained to remember what we had learned in our few palliative care lectures back in medical school. Intravenous fluids were OK, but oxygen was not. Or was it the other way around? Google, the ever-faithful guardian of the panicking intern, was unhelpful. I could not find a reputable online resource to help guide the management of this patient and, given the other sick patients who needed attention, I could not sit down to perform a thorough online literature search. The hospital’s switchboard was equally disappointing – there were no palliative care physicians on call, and the palliative team only accepted pages after 8 a.m., an appalling 5 hours away.
Eventually, I decided to support the patient with an oxygen mask and to continue her intravenous fluid resuscitation. I gave her plenty of narcotics and antiemetics to soothe the discomfort. We were unable to reach any family members despite several attempts. She died just after 5 a.m., without any visible signs of discomfort. It was the first death certificate I had ever signed.
Reflecting on this experience, I was upset at my inability to quickly and adequately care for this patient’s end-of-life needs. I was also angry that I had not prepared myself appropriately for that night on call, and I would have done several things differently had I had the chance.
General surgeons take immense pride in being internists who can operate. We enjoy debating antibiotic, diuretics, and anticoagulation choices with the specialists consulting on our patients, but we often simply "switch off" whenever the patient is labeled "palliative" or "end of life" – relying entirely on the palliative care team for help. And yet, we’re the ones who admit, operate on, and develop therapeutic relationships with those patients and their families. It just doesn’t seem right, but there are several reasons for this dilemma.
First, the fast pace and busy nature of any surgical service means that trainees and staff are under a lot of pressure to round as quickly and efficiently as possible and get to the operating room early in the morning. Given current trainee work-hour limitations and mandated hand-over, today’s junior staff feel this pressure all the more acutely. There simply isn’t enough time or opportunity for continuity of care. Surgical teams can’t engage palliative patients and their families in the way that dedicated palliative care teams can, and so surgical teams are often more comfortable delegating this responsibility entirely to their palliative care colleagues.
Second, there is an emotional burden to caring for palliative patients and their families. Palliation and end-of-life care imply that surgery is neither possible nor advisable any longer. It can be difficult for the surgeon to make that mental segue and assume the dual role of the psychological and medical care giver. Just as surgeons prefer that cardiologists manage their patients’ arrhythmias and nephrologists their patients’ kidney failures, they may prefer that palliative care specialists manage the emotional and medical needs of the patients on whom they can no longer operate.
Furthermore, surgical trainees have limited exposure to palliative care in the clinical setting. As the field of surgery becomes progressively more complex, palliative care training has not been at the forefront of the educational agenda. While it is taught in medical schools and in the surgical residency curriculum, there are few formalized core rotations that offer the surgical resident an insight into the clinical applications of palliative care, except for those who are particularly interested and seek such learning opportunities out in the form of an elective rotation.
The development of dedicated and multidisciplinary palliative care teams that include staff, nurse practitioners, and palliative fellows has further limited surgical resident exposure to the field. Simply put, the current medical system encourages surgical trainees to sign off on the surgical palliative patient.
This, however, isn’t good enough. Palliative care of the surgical patient needs to be a pillar of surgical training. Trainees need to be as comfortable with managing the symptoms of the dying surgical patient as they are administering fluid boluses and ordering narcotics for postoperative pain, especially late at night when there is no palliative care support.
The American College of Surgeons has responded to this challenge by developing numerous educational initiatives through its Palliative Workgroup. It has published a comprehensive surgical palliative care self-study guide specifically for residents (Surgical Palliative Care: A Residents Guide, available for free download).
There are also several regularly updated online palliative care references available either through subscription websites such as UpToDate or for free, such as the Medical College of Wisconsin’s End of Life/Palliative Education Resource Center, which offers "Fast Facts" on the management of the dying patient.
Palliative care is widely accepted today as an important clinical discipline that provides vital support to a growing proportion of the patient population. Surgical training must keep pace with this evolving reality because, at the end of the day, to be without agony and discomfort is a standard of care that every dying patient expects – not just a heroic measure that we can simply ignore.
Dr. Kayssi is a PGY IV in the general surgery program at the University of Toronto.
Dr. Easson is an ACS Fellow and assistant professor in the department of surgery and the Institute of Health, Policy, Management and Evaluation at the University of Toronto.
End-of-life care gains increasing prominence
From Hollywood to Washington, care at the end of life increasingly is part of the national conversation.
As more than 70 million baby boomers (Americans born between 1946 and 1964) approach the end of their life spans, the cultural, clinical and socioeconomic impacts of end-of-life care have become high-profile topics. The French film "Amour," which depicts the challenges faced by an octogenarian couple after the wife has a series of strokes, has received an Academy Award Best Picture nomination. The Institute of Medicine will convene a panel of experts on Feb. 22-24 in Washington to examine the state of end-of-life care. The goal of the IOM Committee on Transforming End of Life Care is to produce a consensus report by 2014 to address end-of-life care. "Coordinated, expert, compassionate care for people dying from chronic diseases continues to challenge the American health care system," according to the IOM’s online announcement of the meeting.
In addition, the Coalition to Transform Advanced Care is hosting a National Summit on Advanced Illness Care on Jan. 29-30 in Washington. The Coalition includes a wide range of constituencies including the IOM, the American Heart Association, the American Academy of Hospice and Palliative Medicine, and the American Geriatrics Society, as well as insurers, health systems, patient advocacy groups, and others.
As policy issues are discussed and begin to take shape, outcomes research focused on end-of-life care will increasingly gain importance. In the area of cancer care, here are some of the highlights of such research presented at a Quality Care Symposium sponsored by the American Society of Clinical Oncology in December 2012 in San Diego.
Palliative care teams: A palliative care team formed by Dr. Allen R. Chen and his associates at Johns Hopkins’s comprehensive cancer center helped change patient and family decisions regarding end-of-life care during a 4-year period and decreased use of intensive or invasive procedures in the last 6 months of life.
The team offered inpatient and clinic consultations and didactic sessions to physicians to improve how they discuss end-of-life issues with patients, plus support for family meetings to discuss and document the goals of care, said Dr. Chen, associate professor of oncology and pediatrics at Johns Hopkins University, Baltimore.
Of the 525 oncology patients who died in the cancer center while hospitalized from 2008 through 2011, the proportion who chose to have do-not-resuscitate (DNR) orders, withdrew ICU support, or chose comfort care instead of more aggressive care increased from 81% to 95% over the course of the 4 years, a statistically significant difference, he reported.
The rate of ICU care during patients’ final hospitalizations did not change significantly, but the proportion of patients who were put on mechanical ventilation for more than 14 days decreased significantly from 10% to 5%. These earlier withdrawals of care did not increase the risk of death, Dr. Chen said – the rate of survival to discharge from the oncology ICU did not change significantly.
The investigators now are looking for ways to minimize ICU visits at the end of life, he said. The presence of advanced cancer, being on a cardiac monitor, or needing supplemental oxygen foreshadowed critical illness in patients in the study. "We want to facilitate the difficult discussion" about end-of-life care choices "before critical care is needed," he said. So far, their efforts haven’t resulted in decreased use of ICU services at the end of life.
Minimizing surgical risks: One of the first comprehensive analyses of surgical outcomes in patients with disseminated cancer found that the risk of death increases greatly with emergency surgery.
John Pesavento, a medical student at Creighton University, Omaha, Neb., and his associates analyzed preoperative and postoperative data from the American College of Surgeon’s National Surgical Quality Improvement Program database on move than 147,000 patients who underwent surgical procedures between 2005 and 2008.
Eight of the 10 most common procedures resulted in significantly higher 30-day mortality rates in the cancer patients as compared with the same surgeries in patients who did not have disseminated cancer.
Comparing patients with and without disseminated cancer, 30-day mortality rates were 21% and 15%, respectively, after exploration of the abdomen, 15% and 9% after removal of the small bowel, 11% and 6% after colon removal, 11% and 5% after colostomy, 10% and 5% after partial removal of the colon, 10% and 0.5% after cholecystectomy, 1.8% and 0.9% after repair of a bowel opening, and 1% and 0.1% after mastectomy, he reported. The most common operations in patients with disseminated cancer were partial removal of the colon (in 11%), partial removal of the liver (9%), partial removal of the intestine (5%), and abdominal exploration (5%), he reported in a poster presentation.
When done as emergency procedures, however, 7 of the top 10 surgeries led to significantly higher 30-day mortality rates for disseminated cancer patients. Death rates in patients with disseminated cancer were 44% for emergency surgery and 12% for nonemergent surgery to explore the abdomen; the respective rates were 33% and 6% after bowel-to-bowel fusion, 31% and 4% after removal of the colon, 28% and 10% after removal of the small intestine, 23% and 5% after cholecystectomy, 22% and 7% after partial removal of the colon, and 19% and 9% after colostomy.
The findings should help physicians counsel their patients with disseminated cancer to help them decide whether it’s worth undergoing surgery – especially emergency surgery – given the higher risks of death, Mr. Pesavento said.
Physicians’ attitudes and education: In a separate study, 16 urologists and four primary care physicians who were undergoing surgical training in urology were interviewed about their attitudes regarding end-of-life care for men dying of prostate cancer. They viewed current end-of-life care as shoddily organized and poorly integrated, and said that ideal outcomes should be defined by patients’ own values and preferences, Dr. Jonathan Bergman and his associates reported in a poster presentation.
The physicians said that, ideally, a multidisciplinary team would care for dying patients, but respondents varied in the degree to which they saw themselves participating, reported Dr. Bergman of the University of California, Los Angeles.
The findings suggest that physician education about end-of-life care needs improvement, and that clinicians should be guided to deliver end-of-life care that is patient-centered and congruent with patients’ values, he said.
Dying in the hospital: Among 2,621 patients with solid tumors, those who had contact with a specialist palliative care team more than a month before their deaths were less likely to die in the hospital (16%) than were patients who had later or no contact with the team (20%), J. Brian Cassel, Ph.D., reported in a poster presentation.
Dr. Cassel and his associates analyzed claims data on the last 6 months of life for 3,128 adults with cancer who had at least one contact with the Virginia Commonwealth University cancer center in Richmond between January 2009 and July 2011. The data set included patients who had solid tumors, underwent bone marrow transfer, or had other hematologic malignancies.
The findings provided a snapshot of end-of-life care at the university, where 32% of the cancer patients were admitted to the hospital within their last 30 days of life, 19% had at least one readmission during their last 6 months of life, and 15% died in the hospital. Chemotherapy was given to 11% in their last month of life, and to 7% in their last 2 weeks of life, said Dr. Cassel of the university.
The specialist palliative care team made contact with 28% of patients a median of 6-10 days before the death of patients with bone marrow transfer or other nonhematologic malignancies and a median of 25 days before the death of patients with solid tumors.
Chemotherapy at the end of life: Patients with hematologic malignancies were significantly more likely to get chemotherapy in the last 30 days of life (38%) compared with patients with solid tumors (8%), Dr. Alma Rodriguez and her associates reported in a poster presentation.
The investigators reviewed data on 7,399 patients who received care for a solid tumor or hematological malignancy at the University of Texas M.D. Anderson Cancer Center, Houston, and died between December 2010 and May 2012. Overall, 14% of patients received chemotherapy within the last 30 days of life.
Of the 1,262 patients who died in the hospital, 44% received chemotherapy within the last 30 days of life, compared with 7% of the 6,137 patients who died in other locations, reported Dr. Rodriguez, professor of medicine at the cancer center.
Chemotherapy within the last 30 days of life was 3 times more likely in patients with metastatic solid tumors, 14 times more likely in patients with nonrelapsed hematologic malignancy, and 36 times more likely in patients with historical or current relapse of hematologic malignancy.
Patients 65 years or older were 38% less likely than were younger patients to get chemotherapy within the last 30 days of life. Patients with one or more comorbidity (most frequently heart failure and coronary artery diseases) were 28% less likely to get chemotherapy within the last 30 days of life as compared with patients without comorbidities.
"As oncologists, we must communicate clearly with our patients about realistic goals of treatment and the likelihood of life-threatening complications of chemotherapy," Dr. Rodriguez said in the poster.
Outpatient palliative care: Dr. Kavitha Ramchandran and her associates at Stanford (Calif.) University studied their system’s electronic medical records to look at the use of palliative care in outpatient clinics from January through September 2012. The number of palliative care contacts with outpatient clinic patients increased from 10 in January to 45 in September. The number of physicians referring outpatients to palliative care increased from 6 in January to 21 in September, she said in a poster presentation.
More than 65% of patients who were referred for palliative care were younger than 65 years, said Dr. Ramchandran of the university. Most patients had only one visit with palliative care (41%) or two visits (23%).
The findings suggest that outpatient palliative care is a growing specialty, she suggested. The data are not mature enough to assess any impact on the quality of end-of-life care or on the efficiency of care, she added.
Dr. Chen and Mr. Pesavento reported having no financial disclosures. Dr. Ramchandran’s study was funded in part by the Stanford Corporate Partners Fund and by various Stanford departments. No financial disclosures were available for the other presenters.
Regarding Dr. Chen’s study, this study does find that we can provide support to patients and their families with appropriately staffed and trained medical teams that specialize in palliative care to really decrease futile, invasive, and expensive care at the end of life. This is broad implications to our patients and their families as they talk about prognosis with their physicians, and also to the physicians taking care of these patients, who may have not had a longitudinal relationship with these patients. Immediate feedback to the caring physicians can decrease some of these high resource utilization maneuvers that don’t provide improvement in quality of life.
Dr. Jyoti D. Patel is a member of ASCO’s Cancer Communications Committee and a thoracic oncologist at Northwestern University, Chicago. She gave these remarks in an online press conference organized by ASCO.
Regarding Dr. Chen’s study, this study does find that we can provide support to patients and their families with appropriately staffed and trained medical teams that specialize in palliative care to really decrease futile, invasive, and expensive care at the end of life. This is broad implications to our patients and their families as they talk about prognosis with their physicians, and also to the physicians taking care of these patients, who may have not had a longitudinal relationship with these patients. Immediate feedback to the caring physicians can decrease some of these high resource utilization maneuvers that don’t provide improvement in quality of life.
Dr. Jyoti D. Patel is a member of ASCO’s Cancer Communications Committee and a thoracic oncologist at Northwestern University, Chicago. She gave these remarks in an online press conference organized by ASCO.
Regarding Dr. Chen’s study, this study does find that we can provide support to patients and their families with appropriately staffed and trained medical teams that specialize in palliative care to really decrease futile, invasive, and expensive care at the end of life. This is broad implications to our patients and their families as they talk about prognosis with their physicians, and also to the physicians taking care of these patients, who may have not had a longitudinal relationship with these patients. Immediate feedback to the caring physicians can decrease some of these high resource utilization maneuvers that don’t provide improvement in quality of life.
Dr. Jyoti D. Patel is a member of ASCO’s Cancer Communications Committee and a thoracic oncologist at Northwestern University, Chicago. She gave these remarks in an online press conference organized by ASCO.
From Hollywood to Washington, care at the end of life increasingly is part of the national conversation.
As more than 70 million baby boomers (Americans born between 1946 and 1964) approach the end of their life spans, the cultural, clinical and socioeconomic impacts of end-of-life care have become high-profile topics. The French film "Amour," which depicts the challenges faced by an octogenarian couple after the wife has a series of strokes, has received an Academy Award Best Picture nomination. The Institute of Medicine will convene a panel of experts on Feb. 22-24 in Washington to examine the state of end-of-life care. The goal of the IOM Committee on Transforming End of Life Care is to produce a consensus report by 2014 to address end-of-life care. "Coordinated, expert, compassionate care for people dying from chronic diseases continues to challenge the American health care system," according to the IOM’s online announcement of the meeting.
In addition, the Coalition to Transform Advanced Care is hosting a National Summit on Advanced Illness Care on Jan. 29-30 in Washington. The Coalition includes a wide range of constituencies including the IOM, the American Heart Association, the American Academy of Hospice and Palliative Medicine, and the American Geriatrics Society, as well as insurers, health systems, patient advocacy groups, and others.
As policy issues are discussed and begin to take shape, outcomes research focused on end-of-life care will increasingly gain importance. In the area of cancer care, here are some of the highlights of such research presented at a Quality Care Symposium sponsored by the American Society of Clinical Oncology in December 2012 in San Diego.
Palliative care teams: A palliative care team formed by Dr. Allen R. Chen and his associates at Johns Hopkins’s comprehensive cancer center helped change patient and family decisions regarding end-of-life care during a 4-year period and decreased use of intensive or invasive procedures in the last 6 months of life.
The team offered inpatient and clinic consultations and didactic sessions to physicians to improve how they discuss end-of-life issues with patients, plus support for family meetings to discuss and document the goals of care, said Dr. Chen, associate professor of oncology and pediatrics at Johns Hopkins University, Baltimore.
Of the 525 oncology patients who died in the cancer center while hospitalized from 2008 through 2011, the proportion who chose to have do-not-resuscitate (DNR) orders, withdrew ICU support, or chose comfort care instead of more aggressive care increased from 81% to 95% over the course of the 4 years, a statistically significant difference, he reported.
The rate of ICU care during patients’ final hospitalizations did not change significantly, but the proportion of patients who were put on mechanical ventilation for more than 14 days decreased significantly from 10% to 5%. These earlier withdrawals of care did not increase the risk of death, Dr. Chen said – the rate of survival to discharge from the oncology ICU did not change significantly.
The investigators now are looking for ways to minimize ICU visits at the end of life, he said. The presence of advanced cancer, being on a cardiac monitor, or needing supplemental oxygen foreshadowed critical illness in patients in the study. "We want to facilitate the difficult discussion" about end-of-life care choices "before critical care is needed," he said. So far, their efforts haven’t resulted in decreased use of ICU services at the end of life.
Minimizing surgical risks: One of the first comprehensive analyses of surgical outcomes in patients with disseminated cancer found that the risk of death increases greatly with emergency surgery.
John Pesavento, a medical student at Creighton University, Omaha, Neb., and his associates analyzed preoperative and postoperative data from the American College of Surgeon’s National Surgical Quality Improvement Program database on move than 147,000 patients who underwent surgical procedures between 2005 and 2008.
Eight of the 10 most common procedures resulted in significantly higher 30-day mortality rates in the cancer patients as compared with the same surgeries in patients who did not have disseminated cancer.
Comparing patients with and without disseminated cancer, 30-day mortality rates were 21% and 15%, respectively, after exploration of the abdomen, 15% and 9% after removal of the small bowel, 11% and 6% after colon removal, 11% and 5% after colostomy, 10% and 5% after partial removal of the colon, 10% and 0.5% after cholecystectomy, 1.8% and 0.9% after repair of a bowel opening, and 1% and 0.1% after mastectomy, he reported. The most common operations in patients with disseminated cancer were partial removal of the colon (in 11%), partial removal of the liver (9%), partial removal of the intestine (5%), and abdominal exploration (5%), he reported in a poster presentation.
When done as emergency procedures, however, 7 of the top 10 surgeries led to significantly higher 30-day mortality rates for disseminated cancer patients. Death rates in patients with disseminated cancer were 44% for emergency surgery and 12% for nonemergent surgery to explore the abdomen; the respective rates were 33% and 6% after bowel-to-bowel fusion, 31% and 4% after removal of the colon, 28% and 10% after removal of the small intestine, 23% and 5% after cholecystectomy, 22% and 7% after partial removal of the colon, and 19% and 9% after colostomy.
The findings should help physicians counsel their patients with disseminated cancer to help them decide whether it’s worth undergoing surgery – especially emergency surgery – given the higher risks of death, Mr. Pesavento said.
Physicians’ attitudes and education: In a separate study, 16 urologists and four primary care physicians who were undergoing surgical training in urology were interviewed about their attitudes regarding end-of-life care for men dying of prostate cancer. They viewed current end-of-life care as shoddily organized and poorly integrated, and said that ideal outcomes should be defined by patients’ own values and preferences, Dr. Jonathan Bergman and his associates reported in a poster presentation.
The physicians said that, ideally, a multidisciplinary team would care for dying patients, but respondents varied in the degree to which they saw themselves participating, reported Dr. Bergman of the University of California, Los Angeles.
The findings suggest that physician education about end-of-life care needs improvement, and that clinicians should be guided to deliver end-of-life care that is patient-centered and congruent with patients’ values, he said.
Dying in the hospital: Among 2,621 patients with solid tumors, those who had contact with a specialist palliative care team more than a month before their deaths were less likely to die in the hospital (16%) than were patients who had later or no contact with the team (20%), J. Brian Cassel, Ph.D., reported in a poster presentation.
Dr. Cassel and his associates analyzed claims data on the last 6 months of life for 3,128 adults with cancer who had at least one contact with the Virginia Commonwealth University cancer center in Richmond between January 2009 and July 2011. The data set included patients who had solid tumors, underwent bone marrow transfer, or had other hematologic malignancies.
The findings provided a snapshot of end-of-life care at the university, where 32% of the cancer patients were admitted to the hospital within their last 30 days of life, 19% had at least one readmission during their last 6 months of life, and 15% died in the hospital. Chemotherapy was given to 11% in their last month of life, and to 7% in their last 2 weeks of life, said Dr. Cassel of the university.
The specialist palliative care team made contact with 28% of patients a median of 6-10 days before the death of patients with bone marrow transfer or other nonhematologic malignancies and a median of 25 days before the death of patients with solid tumors.
Chemotherapy at the end of life: Patients with hematologic malignancies were significantly more likely to get chemotherapy in the last 30 days of life (38%) compared with patients with solid tumors (8%), Dr. Alma Rodriguez and her associates reported in a poster presentation.
The investigators reviewed data on 7,399 patients who received care for a solid tumor or hematological malignancy at the University of Texas M.D. Anderson Cancer Center, Houston, and died between December 2010 and May 2012. Overall, 14% of patients received chemotherapy within the last 30 days of life.
Of the 1,262 patients who died in the hospital, 44% received chemotherapy within the last 30 days of life, compared with 7% of the 6,137 patients who died in other locations, reported Dr. Rodriguez, professor of medicine at the cancer center.
Chemotherapy within the last 30 days of life was 3 times more likely in patients with metastatic solid tumors, 14 times more likely in patients with nonrelapsed hematologic malignancy, and 36 times more likely in patients with historical or current relapse of hematologic malignancy.
Patients 65 years or older were 38% less likely than were younger patients to get chemotherapy within the last 30 days of life. Patients with one or more comorbidity (most frequently heart failure and coronary artery diseases) were 28% less likely to get chemotherapy within the last 30 days of life as compared with patients without comorbidities.
"As oncologists, we must communicate clearly with our patients about realistic goals of treatment and the likelihood of life-threatening complications of chemotherapy," Dr. Rodriguez said in the poster.
Outpatient palliative care: Dr. Kavitha Ramchandran and her associates at Stanford (Calif.) University studied their system’s electronic medical records to look at the use of palliative care in outpatient clinics from January through September 2012. The number of palliative care contacts with outpatient clinic patients increased from 10 in January to 45 in September. The number of physicians referring outpatients to palliative care increased from 6 in January to 21 in September, she said in a poster presentation.
More than 65% of patients who were referred for palliative care were younger than 65 years, said Dr. Ramchandran of the university. Most patients had only one visit with palliative care (41%) or two visits (23%).
The findings suggest that outpatient palliative care is a growing specialty, she suggested. The data are not mature enough to assess any impact on the quality of end-of-life care or on the efficiency of care, she added.
Dr. Chen and Mr. Pesavento reported having no financial disclosures. Dr. Ramchandran’s study was funded in part by the Stanford Corporate Partners Fund and by various Stanford departments. No financial disclosures were available for the other presenters.
From Hollywood to Washington, care at the end of life increasingly is part of the national conversation.
As more than 70 million baby boomers (Americans born between 1946 and 1964) approach the end of their life spans, the cultural, clinical and socioeconomic impacts of end-of-life care have become high-profile topics. The French film "Amour," which depicts the challenges faced by an octogenarian couple after the wife has a series of strokes, has received an Academy Award Best Picture nomination. The Institute of Medicine will convene a panel of experts on Feb. 22-24 in Washington to examine the state of end-of-life care. The goal of the IOM Committee on Transforming End of Life Care is to produce a consensus report by 2014 to address end-of-life care. "Coordinated, expert, compassionate care for people dying from chronic diseases continues to challenge the American health care system," according to the IOM’s online announcement of the meeting.
In addition, the Coalition to Transform Advanced Care is hosting a National Summit on Advanced Illness Care on Jan. 29-30 in Washington. The Coalition includes a wide range of constituencies including the IOM, the American Heart Association, the American Academy of Hospice and Palliative Medicine, and the American Geriatrics Society, as well as insurers, health systems, patient advocacy groups, and others.
As policy issues are discussed and begin to take shape, outcomes research focused on end-of-life care will increasingly gain importance. In the area of cancer care, here are some of the highlights of such research presented at a Quality Care Symposium sponsored by the American Society of Clinical Oncology in December 2012 in San Diego.
Palliative care teams: A palliative care team formed by Dr. Allen R. Chen and his associates at Johns Hopkins’s comprehensive cancer center helped change patient and family decisions regarding end-of-life care during a 4-year period and decreased use of intensive or invasive procedures in the last 6 months of life.
The team offered inpatient and clinic consultations and didactic sessions to physicians to improve how they discuss end-of-life issues with patients, plus support for family meetings to discuss and document the goals of care, said Dr. Chen, associate professor of oncology and pediatrics at Johns Hopkins University, Baltimore.
Of the 525 oncology patients who died in the cancer center while hospitalized from 2008 through 2011, the proportion who chose to have do-not-resuscitate (DNR) orders, withdrew ICU support, or chose comfort care instead of more aggressive care increased from 81% to 95% over the course of the 4 years, a statistically significant difference, he reported.
The rate of ICU care during patients’ final hospitalizations did not change significantly, but the proportion of patients who were put on mechanical ventilation for more than 14 days decreased significantly from 10% to 5%. These earlier withdrawals of care did not increase the risk of death, Dr. Chen said – the rate of survival to discharge from the oncology ICU did not change significantly.
The investigators now are looking for ways to minimize ICU visits at the end of life, he said. The presence of advanced cancer, being on a cardiac monitor, or needing supplemental oxygen foreshadowed critical illness in patients in the study. "We want to facilitate the difficult discussion" about end-of-life care choices "before critical care is needed," he said. So far, their efforts haven’t resulted in decreased use of ICU services at the end of life.
Minimizing surgical risks: One of the first comprehensive analyses of surgical outcomes in patients with disseminated cancer found that the risk of death increases greatly with emergency surgery.
John Pesavento, a medical student at Creighton University, Omaha, Neb., and his associates analyzed preoperative and postoperative data from the American College of Surgeon’s National Surgical Quality Improvement Program database on move than 147,000 patients who underwent surgical procedures between 2005 and 2008.
Eight of the 10 most common procedures resulted in significantly higher 30-day mortality rates in the cancer patients as compared with the same surgeries in patients who did not have disseminated cancer.
Comparing patients with and without disseminated cancer, 30-day mortality rates were 21% and 15%, respectively, after exploration of the abdomen, 15% and 9% after removal of the small bowel, 11% and 6% after colon removal, 11% and 5% after colostomy, 10% and 5% after partial removal of the colon, 10% and 0.5% after cholecystectomy, 1.8% and 0.9% after repair of a bowel opening, and 1% and 0.1% after mastectomy, he reported. The most common operations in patients with disseminated cancer were partial removal of the colon (in 11%), partial removal of the liver (9%), partial removal of the intestine (5%), and abdominal exploration (5%), he reported in a poster presentation.
When done as emergency procedures, however, 7 of the top 10 surgeries led to significantly higher 30-day mortality rates for disseminated cancer patients. Death rates in patients with disseminated cancer were 44% for emergency surgery and 12% for nonemergent surgery to explore the abdomen; the respective rates were 33% and 6% after bowel-to-bowel fusion, 31% and 4% after removal of the colon, 28% and 10% after removal of the small intestine, 23% and 5% after cholecystectomy, 22% and 7% after partial removal of the colon, and 19% and 9% after colostomy.
The findings should help physicians counsel their patients with disseminated cancer to help them decide whether it’s worth undergoing surgery – especially emergency surgery – given the higher risks of death, Mr. Pesavento said.
Physicians’ attitudes and education: In a separate study, 16 urologists and four primary care physicians who were undergoing surgical training in urology were interviewed about their attitudes regarding end-of-life care for men dying of prostate cancer. They viewed current end-of-life care as shoddily organized and poorly integrated, and said that ideal outcomes should be defined by patients’ own values and preferences, Dr. Jonathan Bergman and his associates reported in a poster presentation.
The physicians said that, ideally, a multidisciplinary team would care for dying patients, but respondents varied in the degree to which they saw themselves participating, reported Dr. Bergman of the University of California, Los Angeles.
The findings suggest that physician education about end-of-life care needs improvement, and that clinicians should be guided to deliver end-of-life care that is patient-centered and congruent with patients’ values, he said.
Dying in the hospital: Among 2,621 patients with solid tumors, those who had contact with a specialist palliative care team more than a month before their deaths were less likely to die in the hospital (16%) than were patients who had later or no contact with the team (20%), J. Brian Cassel, Ph.D., reported in a poster presentation.
Dr. Cassel and his associates analyzed claims data on the last 6 months of life for 3,128 adults with cancer who had at least one contact with the Virginia Commonwealth University cancer center in Richmond between January 2009 and July 2011. The data set included patients who had solid tumors, underwent bone marrow transfer, or had other hematologic malignancies.
The findings provided a snapshot of end-of-life care at the university, where 32% of the cancer patients were admitted to the hospital within their last 30 days of life, 19% had at least one readmission during their last 6 months of life, and 15% died in the hospital. Chemotherapy was given to 11% in their last month of life, and to 7% in their last 2 weeks of life, said Dr. Cassel of the university.
The specialist palliative care team made contact with 28% of patients a median of 6-10 days before the death of patients with bone marrow transfer or other nonhematologic malignancies and a median of 25 days before the death of patients with solid tumors.
Chemotherapy at the end of life: Patients with hematologic malignancies were significantly more likely to get chemotherapy in the last 30 days of life (38%) compared with patients with solid tumors (8%), Dr. Alma Rodriguez and her associates reported in a poster presentation.
The investigators reviewed data on 7,399 patients who received care for a solid tumor or hematological malignancy at the University of Texas M.D. Anderson Cancer Center, Houston, and died between December 2010 and May 2012. Overall, 14% of patients received chemotherapy within the last 30 days of life.
Of the 1,262 patients who died in the hospital, 44% received chemotherapy within the last 30 days of life, compared with 7% of the 6,137 patients who died in other locations, reported Dr. Rodriguez, professor of medicine at the cancer center.
Chemotherapy within the last 30 days of life was 3 times more likely in patients with metastatic solid tumors, 14 times more likely in patients with nonrelapsed hematologic malignancy, and 36 times more likely in patients with historical or current relapse of hematologic malignancy.
Patients 65 years or older were 38% less likely than were younger patients to get chemotherapy within the last 30 days of life. Patients with one or more comorbidity (most frequently heart failure and coronary artery diseases) were 28% less likely to get chemotherapy within the last 30 days of life as compared with patients without comorbidities.
"As oncologists, we must communicate clearly with our patients about realistic goals of treatment and the likelihood of life-threatening complications of chemotherapy," Dr. Rodriguez said in the poster.
Outpatient palliative care: Dr. Kavitha Ramchandran and her associates at Stanford (Calif.) University studied their system’s electronic medical records to look at the use of palliative care in outpatient clinics from January through September 2012. The number of palliative care contacts with outpatient clinic patients increased from 10 in January to 45 in September. The number of physicians referring outpatients to palliative care increased from 6 in January to 21 in September, she said in a poster presentation.
More than 65% of patients who were referred for palliative care were younger than 65 years, said Dr. Ramchandran of the university. Most patients had only one visit with palliative care (41%) or two visits (23%).
The findings suggest that outpatient palliative care is a growing specialty, she suggested. The data are not mature enough to assess any impact on the quality of end-of-life care or on the efficiency of care, she added.
Dr. Chen and Mr. Pesavento reported having no financial disclosures. Dr. Ramchandran’s study was funded in part by the Stanford Corporate Partners Fund and by various Stanford departments. No financial disclosures were available for the other presenters.
Optimizing Home Health Care: Enhanced Value and Improved Outcomes
Supplement Editor:
William Zafirau, MD
Supplement Co-Editors:
Steven H. Landers, MD, MPH, and Cindy Vunovich, RN, BSN, MSM
Contents
Introduction—Medicine’s future: Helping patients stay healthy at home
Steven H. Landers, MD, MPH
Care transitions and advanced home care models
Improving patient outcomes with better care transitions: The role for home health
Michael O. Fleming, MD, and Tara Trahan Haney
Improving outcomes and lowering costs by applying advanced models of in-home care
Peter A. Boling, MD; Rashmi V. Chandekar, MD; Beth Hungate, MS, ANP-BC; Martha Purvis, MSN; Rachel Selby-Penczak, MD; and Linda J. Abbey, MD
Home care for knee replacement and heart failure
In-home care following total knee replacement
Mark I. Froimson, MD, MBA
Home-based care for heart failure: Cleveland Clinic's "Heart Care at Home" transitional care program
Eiran Z. Gorodeski, MD, MPH; Sandra Chlad, NP; and Seth Vilensky, MBA
Technology innovations and palliative care
The case for "connected health" at home
Steven H. Landers, MD, MPH
Innovative models of home-based palliative care
Margherita C. Labson, RN, MSHSA, CPHQ, CCM; Michele M. Sacco, MS; David E. Weissman, MD; Betsy Gornet, FACHE; and Brad Stuart, MD
Cleveland Clinic Journal of Medicine interview
Accountable care and patient-centered medical homes: Implications for office-based practice
An interview with David L. Longworth, MD
Supplement Editor:
William Zafirau, MD
Supplement Co-Editors:
Steven H. Landers, MD, MPH, and Cindy Vunovich, RN, BSN, MSM
Contents
Introduction—Medicine’s future: Helping patients stay healthy at home
Steven H. Landers, MD, MPH
Care transitions and advanced home care models
Improving patient outcomes with better care transitions: The role for home health
Michael O. Fleming, MD, and Tara Trahan Haney
Improving outcomes and lowering costs by applying advanced models of in-home care
Peter A. Boling, MD; Rashmi V. Chandekar, MD; Beth Hungate, MS, ANP-BC; Martha Purvis, MSN; Rachel Selby-Penczak, MD; and Linda J. Abbey, MD
Home care for knee replacement and heart failure
In-home care following total knee replacement
Mark I. Froimson, MD, MBA
Home-based care for heart failure: Cleveland Clinic's "Heart Care at Home" transitional care program
Eiran Z. Gorodeski, MD, MPH; Sandra Chlad, NP; and Seth Vilensky, MBA
Technology innovations and palliative care
The case for "connected health" at home
Steven H. Landers, MD, MPH
Innovative models of home-based palliative care
Margherita C. Labson, RN, MSHSA, CPHQ, CCM; Michele M. Sacco, MS; David E. Weissman, MD; Betsy Gornet, FACHE; and Brad Stuart, MD
Cleveland Clinic Journal of Medicine interview
Accountable care and patient-centered medical homes: Implications for office-based practice
An interview with David L. Longworth, MD
Supplement Editor:
William Zafirau, MD
Supplement Co-Editors:
Steven H. Landers, MD, MPH, and Cindy Vunovich, RN, BSN, MSM
Contents
Introduction—Medicine’s future: Helping patients stay healthy at home
Steven H. Landers, MD, MPH
Care transitions and advanced home care models
Improving patient outcomes with better care transitions: The role for home health
Michael O. Fleming, MD, and Tara Trahan Haney
Improving outcomes and lowering costs by applying advanced models of in-home care
Peter A. Boling, MD; Rashmi V. Chandekar, MD; Beth Hungate, MS, ANP-BC; Martha Purvis, MSN; Rachel Selby-Penczak, MD; and Linda J. Abbey, MD
Home care for knee replacement and heart failure
In-home care following total knee replacement
Mark I. Froimson, MD, MBA
Home-based care for heart failure: Cleveland Clinic's "Heart Care at Home" transitional care program
Eiran Z. Gorodeski, MD, MPH; Sandra Chlad, NP; and Seth Vilensky, MBA
Technology innovations and palliative care
The case for "connected health" at home
Steven H. Landers, MD, MPH
Innovative models of home-based palliative care
Margherita C. Labson, RN, MSHSA, CPHQ, CCM; Michele M. Sacco, MS; David E. Weissman, MD; Betsy Gornet, FACHE; and Brad Stuart, MD
Cleveland Clinic Journal of Medicine interview
Accountable care and patient-centered medical homes: Implications for office-based practice
An interview with David L. Longworth, MD