User login
Post-COVID fatigue, exercise intolerance signal ME/CFS
A new study provides yet more evidence that a significant subset of people who experience persistent fatigue and exercise intolerance following COVID-19 will meet diagnostic criteria for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS).
Data from the prospective observational study of 42 patients with “post-COVID-19 syndrome (PCS),” including persistent fatigue and exercise intolerance, suggest that a large proportion will meet strict diagnostic criteria for ME/CFS, including the hallmark postexertional malaise (PEM). Still others may experience similar disability but lack duration and/or severity requirements for the diagnosis.
Moreover, disease severity and symptom burden were found similar in those with ME/CFS following COVID-19 and in a group of 19 age- and sex-matched individuals with ME/CFS that wasn’t associated with COVID-19.
“The major finding is that ME/CFS is indeed part of the spectrum of the post-COVID syndrome and very similar to the ME/CFS we know after other infectious triggers,” senior author Carmen Scheibenbogen, MD, acting director of the Institute for Medical Immunology at the Charité University Medicine Campus Virchow-Klinikum, Berlin, told this news organization.
Importantly, from a clinical standpoint, both diminished hand-grip strength (HGS) and orthostatic intolerance were common across all patient groups, as were several laboratory values, Claudia Kedor, MD, and colleagues at Charité report in the paper, published online in Nature Communications.
Of the 42 with PCS, including persistent fatigue and exercise intolerance lasting at least 6 months, 19 met the rigorous Canadian Consensus Criteria (CCC) for ME/CFS, established in 2003, which require PEM, along with sleep dysfunction, significant persistent fatigue, pain, and several other symptoms from neurological/cognitive, autonomic, neuroendocrine, and immune categories that persist for at least 6 months.
Of the 23 who did not meet the CCC criteria, 18 still experienced PEM but for less than the required 14 hours set by the authors based on recent data. The original CCC had suggested 24 hours as the PEM duration. Eight subjects met all the Canadian criteria except for the neurological/cognitive symptoms. None of the 42 had evidence of severe depression.
The previously widely used 1994 “Fukuda” criteria for ME/CFS are no longer recommended because they don’t require PEM, which is now considered a key symptom. The more recent 2015 Institute (now Academy) of Medicine criteria don’t define the length of PEM, the authors note in the paper.
Dr. Scheibenbogen said, “Post-COVID has a spectrum of syndromes and conditions. We see that a subset of patients have similar symptoms of ME/CFS but don’t fulfill the CCC, although they may meet less stringent criteria. We think this is of relevance for both diagnostic markers and development of therapy, because there may be different pathomechanisms between the subsets of post-COVID patients.”
She pointed to other studies from her group suggesting that inflammation is present early in post-COVID (not yet published), while in the subset that goes on to ME/CFS, autoantibodies or endothelial dysfunction play a more important role. «At the moment, it’s quite complex, and I don’t think in the end we will have just one pathomechanism. So I think we’ll need to develop various treatment strategies.”
Asked to comment on the new data, Anthony L. Komaroff, MD, professor of medicine at Harvard Medical School, senior physician at Brigham and Women’s Hospital, both in Boston, and editor in chief of the Harvard Health Letter, told this news organization, “This paper adds to the evidence that an illness with symptoms that meet criteria for ME/CFS can follow COVID-19 in nearly half of those patients who have lingering symptoms. This can occur even in people who initially have only mild symptoms from COVID-19, although it is more likely to happen in the people who are sickest when they first get COVID-19. And those who meet criteria for ME/CFS were seriously impaired in their ability to function, [both] at work and at home.”
But, Dr. Komaroff also cautioned, “the study does not help in determining what fraction of all people who are infected with SARS-CoV-2 go on to develop a condition like ME/CFS, nor how long that condition will last. It is crucial that we get answers to these questions, as the impact on the economy, the health care system, and the disability system could be substantial.”
He pointed to a recent report from the Brookings Institution (2022 Aug 24. “New data shows long Covid is keeping as many as 4 million people out of work” Katie Bach) “finding that “long COVID may be a major contributor to the shortage of job applicants plaguing many businesses.”
Biomarkers include hand-grip strength, orthostatic intolerance, lab measures
Hand-grip strength, as assessed by 10 repeat grips at maximum force and repeated after 60 minutes, were lower for all those meeting ME/CFS criteria, compared with the healthy controls. Hand-grip strength parameters were also positively correlated with laboratory hemoglobin measures in both PCS groups who did and didn’t meet the Canadian ME/CFS criteria.
A total of three patients with PCS who didn’t meet ME/CFS criteria and seven with PCS who met ME/CFS criteria had sitting blood pressures of greater than 140 mm Hg systolic and/or greater than 90 mm Hg diastolic. Five patients with PCS – four who met ME/CFS criteria and one who didn’t – fulfilled criteria for postural orthostatic tachycardia syndrome. Orthostatic hypotension was diagnosed in a total of seven with PCS, including one who did not meet ME/CFS criteria and the rest who did.
Among significant laboratory findings, mannose-binding lectin deficiency, which is associated with increased infection susceptibility and found in only about 6% of historical controls, was found more frequently in both of the PCS cohorts (17% of those with ME/CFS and 23% of those without) than it has been in the past among those with ME/CFS, compared with historical controls (15%).
There was only slight elevation in C-reactive protein, the most commonly measured marker of inflammation. However, another marker indicating inflammation within the last 3-4 months, interleukin 8 assessed in erythrocytes, was above normal in 37% with PCS and ME/CFS and in 48% with PCS who did not meet the ME/CFS criteria.
Elevated antinuclear antibodies, anti–thyroid peroxidase antibodies, vitamin D deficiencies, and folic acid deficiencies were all seen in small numbers of the PCS patients. Angiotensin-converting enzyme 1 levels were below the normal range in 31% of all patients.
“We must anticipate that this pandemic has the potential to dramatically increase the number of ME/CFS patients,” Dr. Kedor and colleagues write. “At the same time, it offers the unique chance to identify ME/CFS patients in a very early stage of disease and apply interventions such as pacing and coping early with a better therapeutic prognosis. Further, it is an unprecedented opportunity to understand the underlying pathomechanism and characterize targets for specific treatment approaches.”
Dr. Scheibenbogen and Dr. Komaroff reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study provides yet more evidence that a significant subset of people who experience persistent fatigue and exercise intolerance following COVID-19 will meet diagnostic criteria for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS).
Data from the prospective observational study of 42 patients with “post-COVID-19 syndrome (PCS),” including persistent fatigue and exercise intolerance, suggest that a large proportion will meet strict diagnostic criteria for ME/CFS, including the hallmark postexertional malaise (PEM). Still others may experience similar disability but lack duration and/or severity requirements for the diagnosis.
Moreover, disease severity and symptom burden were found similar in those with ME/CFS following COVID-19 and in a group of 19 age- and sex-matched individuals with ME/CFS that wasn’t associated with COVID-19.
“The major finding is that ME/CFS is indeed part of the spectrum of the post-COVID syndrome and very similar to the ME/CFS we know after other infectious triggers,” senior author Carmen Scheibenbogen, MD, acting director of the Institute for Medical Immunology at the Charité University Medicine Campus Virchow-Klinikum, Berlin, told this news organization.
Importantly, from a clinical standpoint, both diminished hand-grip strength (HGS) and orthostatic intolerance were common across all patient groups, as were several laboratory values, Claudia Kedor, MD, and colleagues at Charité report in the paper, published online in Nature Communications.
Of the 42 with PCS, including persistent fatigue and exercise intolerance lasting at least 6 months, 19 met the rigorous Canadian Consensus Criteria (CCC) for ME/CFS, established in 2003, which require PEM, along with sleep dysfunction, significant persistent fatigue, pain, and several other symptoms from neurological/cognitive, autonomic, neuroendocrine, and immune categories that persist for at least 6 months.
Of the 23 who did not meet the CCC criteria, 18 still experienced PEM but for less than the required 14 hours set by the authors based on recent data. The original CCC had suggested 24 hours as the PEM duration. Eight subjects met all the Canadian criteria except for the neurological/cognitive symptoms. None of the 42 had evidence of severe depression.
The previously widely used 1994 “Fukuda” criteria for ME/CFS are no longer recommended because they don’t require PEM, which is now considered a key symptom. The more recent 2015 Institute (now Academy) of Medicine criteria don’t define the length of PEM, the authors note in the paper.
Dr. Scheibenbogen said, “Post-COVID has a spectrum of syndromes and conditions. We see that a subset of patients have similar symptoms of ME/CFS but don’t fulfill the CCC, although they may meet less stringent criteria. We think this is of relevance for both diagnostic markers and development of therapy, because there may be different pathomechanisms between the subsets of post-COVID patients.”
She pointed to other studies from her group suggesting that inflammation is present early in post-COVID (not yet published), while in the subset that goes on to ME/CFS, autoantibodies or endothelial dysfunction play a more important role. «At the moment, it’s quite complex, and I don’t think in the end we will have just one pathomechanism. So I think we’ll need to develop various treatment strategies.”
Asked to comment on the new data, Anthony L. Komaroff, MD, professor of medicine at Harvard Medical School, senior physician at Brigham and Women’s Hospital, both in Boston, and editor in chief of the Harvard Health Letter, told this news organization, “This paper adds to the evidence that an illness with symptoms that meet criteria for ME/CFS can follow COVID-19 in nearly half of those patients who have lingering symptoms. This can occur even in people who initially have only mild symptoms from COVID-19, although it is more likely to happen in the people who are sickest when they first get COVID-19. And those who meet criteria for ME/CFS were seriously impaired in their ability to function, [both] at work and at home.”
But, Dr. Komaroff also cautioned, “the study does not help in determining what fraction of all people who are infected with SARS-CoV-2 go on to develop a condition like ME/CFS, nor how long that condition will last. It is crucial that we get answers to these questions, as the impact on the economy, the health care system, and the disability system could be substantial.”
He pointed to a recent report from the Brookings Institution (2022 Aug 24. “New data shows long Covid is keeping as many as 4 million people out of work” Katie Bach) “finding that “long COVID may be a major contributor to the shortage of job applicants plaguing many businesses.”
Biomarkers include hand-grip strength, orthostatic intolerance, lab measures
Hand-grip strength, as assessed by 10 repeat grips at maximum force and repeated after 60 minutes, were lower for all those meeting ME/CFS criteria, compared with the healthy controls. Hand-grip strength parameters were also positively correlated with laboratory hemoglobin measures in both PCS groups who did and didn’t meet the Canadian ME/CFS criteria.
A total of three patients with PCS who didn’t meet ME/CFS criteria and seven with PCS who met ME/CFS criteria had sitting blood pressures of greater than 140 mm Hg systolic and/or greater than 90 mm Hg diastolic. Five patients with PCS – four who met ME/CFS criteria and one who didn’t – fulfilled criteria for postural orthostatic tachycardia syndrome. Orthostatic hypotension was diagnosed in a total of seven with PCS, including one who did not meet ME/CFS criteria and the rest who did.
Among significant laboratory findings, mannose-binding lectin deficiency, which is associated with increased infection susceptibility and found in only about 6% of historical controls, was found more frequently in both of the PCS cohorts (17% of those with ME/CFS and 23% of those without) than it has been in the past among those with ME/CFS, compared with historical controls (15%).
There was only slight elevation in C-reactive protein, the most commonly measured marker of inflammation. However, another marker indicating inflammation within the last 3-4 months, interleukin 8 assessed in erythrocytes, was above normal in 37% with PCS and ME/CFS and in 48% with PCS who did not meet the ME/CFS criteria.
Elevated antinuclear antibodies, anti–thyroid peroxidase antibodies, vitamin D deficiencies, and folic acid deficiencies were all seen in small numbers of the PCS patients. Angiotensin-converting enzyme 1 levels were below the normal range in 31% of all patients.
“We must anticipate that this pandemic has the potential to dramatically increase the number of ME/CFS patients,” Dr. Kedor and colleagues write. “At the same time, it offers the unique chance to identify ME/CFS patients in a very early stage of disease and apply interventions such as pacing and coping early with a better therapeutic prognosis. Further, it is an unprecedented opportunity to understand the underlying pathomechanism and characterize targets for specific treatment approaches.”
Dr. Scheibenbogen and Dr. Komaroff reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study provides yet more evidence that a significant subset of people who experience persistent fatigue and exercise intolerance following COVID-19 will meet diagnostic criteria for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS).
Data from the prospective observational study of 42 patients with “post-COVID-19 syndrome (PCS),” including persistent fatigue and exercise intolerance, suggest that a large proportion will meet strict diagnostic criteria for ME/CFS, including the hallmark postexertional malaise (PEM). Still others may experience similar disability but lack duration and/or severity requirements for the diagnosis.
Moreover, disease severity and symptom burden were found similar in those with ME/CFS following COVID-19 and in a group of 19 age- and sex-matched individuals with ME/CFS that wasn’t associated with COVID-19.
“The major finding is that ME/CFS is indeed part of the spectrum of the post-COVID syndrome and very similar to the ME/CFS we know after other infectious triggers,” senior author Carmen Scheibenbogen, MD, acting director of the Institute for Medical Immunology at the Charité University Medicine Campus Virchow-Klinikum, Berlin, told this news organization.
Importantly, from a clinical standpoint, both diminished hand-grip strength (HGS) and orthostatic intolerance were common across all patient groups, as were several laboratory values, Claudia Kedor, MD, and colleagues at Charité report in the paper, published online in Nature Communications.
Of the 42 with PCS, including persistent fatigue and exercise intolerance lasting at least 6 months, 19 met the rigorous Canadian Consensus Criteria (CCC) for ME/CFS, established in 2003, which require PEM, along with sleep dysfunction, significant persistent fatigue, pain, and several other symptoms from neurological/cognitive, autonomic, neuroendocrine, and immune categories that persist for at least 6 months.
Of the 23 who did not meet the CCC criteria, 18 still experienced PEM but for less than the required 14 hours set by the authors based on recent data. The original CCC had suggested 24 hours as the PEM duration. Eight subjects met all the Canadian criteria except for the neurological/cognitive symptoms. None of the 42 had evidence of severe depression.
The previously widely used 1994 “Fukuda” criteria for ME/CFS are no longer recommended because they don’t require PEM, which is now considered a key symptom. The more recent 2015 Institute (now Academy) of Medicine criteria don’t define the length of PEM, the authors note in the paper.
Dr. Scheibenbogen said, “Post-COVID has a spectrum of syndromes and conditions. We see that a subset of patients have similar symptoms of ME/CFS but don’t fulfill the CCC, although they may meet less stringent criteria. We think this is of relevance for both diagnostic markers and development of therapy, because there may be different pathomechanisms between the subsets of post-COVID patients.”
She pointed to other studies from her group suggesting that inflammation is present early in post-COVID (not yet published), while in the subset that goes on to ME/CFS, autoantibodies or endothelial dysfunction play a more important role. «At the moment, it’s quite complex, and I don’t think in the end we will have just one pathomechanism. So I think we’ll need to develop various treatment strategies.”
Asked to comment on the new data, Anthony L. Komaroff, MD, professor of medicine at Harvard Medical School, senior physician at Brigham and Women’s Hospital, both in Boston, and editor in chief of the Harvard Health Letter, told this news organization, “This paper adds to the evidence that an illness with symptoms that meet criteria for ME/CFS can follow COVID-19 in nearly half of those patients who have lingering symptoms. This can occur even in people who initially have only mild symptoms from COVID-19, although it is more likely to happen in the people who are sickest when they first get COVID-19. And those who meet criteria for ME/CFS were seriously impaired in their ability to function, [both] at work and at home.”
But, Dr. Komaroff also cautioned, “the study does not help in determining what fraction of all people who are infected with SARS-CoV-2 go on to develop a condition like ME/CFS, nor how long that condition will last. It is crucial that we get answers to these questions, as the impact on the economy, the health care system, and the disability system could be substantial.”
He pointed to a recent report from the Brookings Institution (2022 Aug 24. “New data shows long Covid is keeping as many as 4 million people out of work” Katie Bach) “finding that “long COVID may be a major contributor to the shortage of job applicants plaguing many businesses.”
Biomarkers include hand-grip strength, orthostatic intolerance, lab measures
Hand-grip strength, as assessed by 10 repeat grips at maximum force and repeated after 60 minutes, were lower for all those meeting ME/CFS criteria, compared with the healthy controls. Hand-grip strength parameters were also positively correlated with laboratory hemoglobin measures in both PCS groups who did and didn’t meet the Canadian ME/CFS criteria.
A total of three patients with PCS who didn’t meet ME/CFS criteria and seven with PCS who met ME/CFS criteria had sitting blood pressures of greater than 140 mm Hg systolic and/or greater than 90 mm Hg diastolic. Five patients with PCS – four who met ME/CFS criteria and one who didn’t – fulfilled criteria for postural orthostatic tachycardia syndrome. Orthostatic hypotension was diagnosed in a total of seven with PCS, including one who did not meet ME/CFS criteria and the rest who did.
Among significant laboratory findings, mannose-binding lectin deficiency, which is associated with increased infection susceptibility and found in only about 6% of historical controls, was found more frequently in both of the PCS cohorts (17% of those with ME/CFS and 23% of those without) than it has been in the past among those with ME/CFS, compared with historical controls (15%).
There was only slight elevation in C-reactive protein, the most commonly measured marker of inflammation. However, another marker indicating inflammation within the last 3-4 months, interleukin 8 assessed in erythrocytes, was above normal in 37% with PCS and ME/CFS and in 48% with PCS who did not meet the ME/CFS criteria.
Elevated antinuclear antibodies, anti–thyroid peroxidase antibodies, vitamin D deficiencies, and folic acid deficiencies were all seen in small numbers of the PCS patients. Angiotensin-converting enzyme 1 levels were below the normal range in 31% of all patients.
“We must anticipate that this pandemic has the potential to dramatically increase the number of ME/CFS patients,” Dr. Kedor and colleagues write. “At the same time, it offers the unique chance to identify ME/CFS patients in a very early stage of disease and apply interventions such as pacing and coping early with a better therapeutic prognosis. Further, it is an unprecedented opportunity to understand the underlying pathomechanism and characterize targets for specific treatment approaches.”
Dr. Scheibenbogen and Dr. Komaroff reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NATURE COMMUNICATIONS
Prior psychological distress tied to ‘long-COVID’ conditions
In an analysis of almost 55,000 adult participants in three ongoing studies, having depression, anxiety, worry, perceived stress, or loneliness early in the pandemic, before SARS-CoV-2 infection, was associated with a 50% increased risk for developing long COVID. These types of psychological distress were also associated with a 15% to 51% greater risk for impairment in daily life among individuals with long COVID.
Psychological distress was even more strongly associated with developing long COVID than were physical health risk factors, and the increased risk was not explained by health behaviors such as smoking or physical comorbidities, researchers note.
“Our findings suggest the need to consider psychological health in addition to physical health as risk factors of long COVID-19,” lead author Siwen Wang, MD, postdoctoral fellow, department of nutrition, Harvard T. H. Chan School of Public Health, Boston, said in an interview.
“We need to increase public awareness of the importance of mental health and focus on getting mental health care for people who need it, increasing the supply of mental health clinicians and improving access to care,” she said.
The findings were published online in JAMA Psychiatry.
‘Poorly understood’
Postacute sequelae of SARS-CoV-2 (“long COVID”), which are “signs and symptoms consistent with COVID-19 that extend beyond 4 weeks from onset of infection” constitute “an emerging health issue,” the investigators write.
Dr. Wang noted that it has been estimated that 8-23 million Americans have developed long COVID. However, “despite the high prevalence and daily life impairment associated with long COVID, it is still poorly understood, and few risk factors have been established,” she said.
Although psychological distress may be implicated in long COVID, only three previous studies investigated psychological factors as potential contributors, the researchers note. Also, no study has investigated the potential role of other common manifestations of distress that have increased during the pandemic, such as loneliness and perceived stress, they add.
To investigate these issues, the researchers turned to three large ongoing longitudinal studies: the Nurses’ Health Study II (NSHII), the Nurses’ Health study 3 (NHS3), and the Growing Up Today Study (GUTS).
They analyzed data on 54,960 total participants (96.6% women; mean age, 57.5 years). Of the full group, 38% were active health care workers.
Participants completed an online COVID-19 questionnaire from April 2020 to Sept. 1, 2020 (baseline), and monthly surveys thereafter. Beginning in August 2020, surveys were administered quarterly. The end of follow-up was in November 2021.
The COVID questionnaires included questions about positive SARS-CoV-2 test results, COVID symptoms and hospitalization since March 1, 2020, and the presence of long-term COVID symptoms, such as fatigue, respiratory problems, persistent cough, muscle/joint/chest pain, smell/taste problems, confusion/disorientation/brain fog, depression/anxiety/changes in mood, headache, and memory problems.
Participants who reported these post-COVID conditions were asked about the frequency of symptoms and the degree of impairment in daily life.
Inflammation, immune dysregulation implicated?
The Patient Health Questionnaire–4 (PHQ-4) was used to assess for anxiety and depressive symptoms in the past 2 weeks. It consists of a two-item depression measure (PHQ-2) and a two-item Generalized Anxiety Disorder Scale (GAD-2).
Non–health care providers completed two additional assessments of psychological distress: the four-item Perceived Stress Scale and the three-item UCLA Loneliness Scale.
The researchers included demographic factors, weight, smoking status, marital status, and medical conditions, including diabetes, hypertension, hypercholesterolemia, asthma, and cancer, and socioeconomic factors as covariates.
For each participant, the investigators calculated the number of types of distress experienced at a high level, including probable depression, probable anxiety, worry about COVID-19, being in the top quartile of perceived stress, and loneliness.
During the 19 months of follow-up (1-47 weeks after baseline), 6% of respondents reported a positive result on a SARS-CoV-2 antibody, antigen, or polymerase chain reaction test.
Of these, 43.9% reported long-COVID conditions, with most reporting that symptoms lasted 2 months or longer; 55.8% reported at least occasional daily life impairment.
The most common post-COVID conditions were fatigue (reported by 56%), loss of smell or taste problems (44.6%), shortness of breath (25.5%), confusion/disorientation/ brain fog (24.5%), and memory issues (21.8%).
Among patients who had been infected, there was a considerably higher rate of preinfection psychological distress after adjusting for sociodemographic factors, health behaviors, and comorbidities. Each type of distress was associated with post-COVID conditions.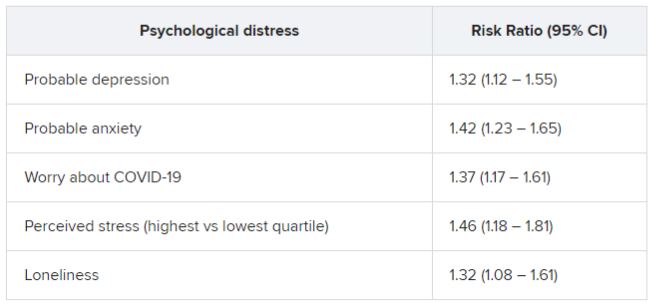
In addition, participants who had experienced at least two types of distress prior to infection were at nearly 50% increased risk for post–COVID conditions (risk ratio, 1.49; 95% confidence interval, 1.23-1.80).
Among those with post-COVID conditions, all types of distress were associated with increased risk for daily life impairment (RR range, 1.15-1.51).
Senior author Andrea Roberts, PhD, senior research scientist at the Harvard T. H. Chan School of Public Health, Boston, noted that the investigators did not examine biological mechanisms potentially underlying the association they found.
However, “based on prior research, it may be that inflammation and immune dysregulation related to psychological distress play a role in the association of distress with long COVID, but we can’t be sure,” Dr. Roberts said.
Contributes to the field
Commenting for this article, Yapeng Su, PhD, a postdoctoral researcher at the Fred Hutchinson Cancer Research Center in Seattle, called the study “great work contributing to the long-COVID research field and revealing important connections” with psychological stress prior to infection.
Dr. Su, who was not involved with the study, was previously at the Institute for Systems Biology, also in Seattle, and has written about long COVID.
He noted that the “biological mechanism of such intriguing linkage is definitely the important next step, which will likely require deep phenotyping of biological specimens from these patients longitudinally.”
Dr. Wang pointed to past research suggesting that some patients with mental illness “sometimes develop autoantibodies that have also been associated with increased risk of long COVID.” In addition, depression “affects the brain in ways that may explain certain cognitive symptoms in long COVID,” she added.
More studies are now needed to understand how psychological distress increases the risk for long COVID, said Dr. Wang.
The research was supported by grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Institutes of Health, the Dean’s Fund for Scientific Advancement Acceleration Award from the Harvard T. H. Chan School of Public Health, the Massachusetts Consortium on Pathogen Readiness Evergrande COVID-19 Response Fund Award, and the Veterans Affairs Health Services Research and Development Service funds. Dr. Wang and Dr. Roberts have reported no relevant financial relationships. The other investigators’ disclosures are listed in the original article. Dr. Su reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In an analysis of almost 55,000 adult participants in three ongoing studies, having depression, anxiety, worry, perceived stress, or loneliness early in the pandemic, before SARS-CoV-2 infection, was associated with a 50% increased risk for developing long COVID. These types of psychological distress were also associated with a 15% to 51% greater risk for impairment in daily life among individuals with long COVID.
Psychological distress was even more strongly associated with developing long COVID than were physical health risk factors, and the increased risk was not explained by health behaviors such as smoking or physical comorbidities, researchers note.
“Our findings suggest the need to consider psychological health in addition to physical health as risk factors of long COVID-19,” lead author Siwen Wang, MD, postdoctoral fellow, department of nutrition, Harvard T. H. Chan School of Public Health, Boston, said in an interview.
“We need to increase public awareness of the importance of mental health and focus on getting mental health care for people who need it, increasing the supply of mental health clinicians and improving access to care,” she said.
The findings were published online in JAMA Psychiatry.
‘Poorly understood’
Postacute sequelae of SARS-CoV-2 (“long COVID”), which are “signs and symptoms consistent with COVID-19 that extend beyond 4 weeks from onset of infection” constitute “an emerging health issue,” the investigators write.
Dr. Wang noted that it has been estimated that 8-23 million Americans have developed long COVID. However, “despite the high prevalence and daily life impairment associated with long COVID, it is still poorly understood, and few risk factors have been established,” she said.
Although psychological distress may be implicated in long COVID, only three previous studies investigated psychological factors as potential contributors, the researchers note. Also, no study has investigated the potential role of other common manifestations of distress that have increased during the pandemic, such as loneliness and perceived stress, they add.
To investigate these issues, the researchers turned to three large ongoing longitudinal studies: the Nurses’ Health Study II (NSHII), the Nurses’ Health study 3 (NHS3), and the Growing Up Today Study (GUTS).
They analyzed data on 54,960 total participants (96.6% women; mean age, 57.5 years). Of the full group, 38% were active health care workers.
Participants completed an online COVID-19 questionnaire from April 2020 to Sept. 1, 2020 (baseline), and monthly surveys thereafter. Beginning in August 2020, surveys were administered quarterly. The end of follow-up was in November 2021.
The COVID questionnaires included questions about positive SARS-CoV-2 test results, COVID symptoms and hospitalization since March 1, 2020, and the presence of long-term COVID symptoms, such as fatigue, respiratory problems, persistent cough, muscle/joint/chest pain, smell/taste problems, confusion/disorientation/brain fog, depression/anxiety/changes in mood, headache, and memory problems.
Participants who reported these post-COVID conditions were asked about the frequency of symptoms and the degree of impairment in daily life.
Inflammation, immune dysregulation implicated?
The Patient Health Questionnaire–4 (PHQ-4) was used to assess for anxiety and depressive symptoms in the past 2 weeks. It consists of a two-item depression measure (PHQ-2) and a two-item Generalized Anxiety Disorder Scale (GAD-2).
Non–health care providers completed two additional assessments of psychological distress: the four-item Perceived Stress Scale and the three-item UCLA Loneliness Scale.
The researchers included demographic factors, weight, smoking status, marital status, and medical conditions, including diabetes, hypertension, hypercholesterolemia, asthma, and cancer, and socioeconomic factors as covariates.
For each participant, the investigators calculated the number of types of distress experienced at a high level, including probable depression, probable anxiety, worry about COVID-19, being in the top quartile of perceived stress, and loneliness.
During the 19 months of follow-up (1-47 weeks after baseline), 6% of respondents reported a positive result on a SARS-CoV-2 antibody, antigen, or polymerase chain reaction test.
Of these, 43.9% reported long-COVID conditions, with most reporting that symptoms lasted 2 months or longer; 55.8% reported at least occasional daily life impairment.
The most common post-COVID conditions were fatigue (reported by 56%), loss of smell or taste problems (44.6%), shortness of breath (25.5%), confusion/disorientation/ brain fog (24.5%), and memory issues (21.8%).
Among patients who had been infected, there was a considerably higher rate of preinfection psychological distress after adjusting for sociodemographic factors, health behaviors, and comorbidities. Each type of distress was associated with post-COVID conditions.
In addition, participants who had experienced at least two types of distress prior to infection were at nearly 50% increased risk for post–COVID conditions (risk ratio, 1.49; 95% confidence interval, 1.23-1.80).
Among those with post-COVID conditions, all types of distress were associated with increased risk for daily life impairment (RR range, 1.15-1.51).
Senior author Andrea Roberts, PhD, senior research scientist at the Harvard T. H. Chan School of Public Health, Boston, noted that the investigators did not examine biological mechanisms potentially underlying the association they found.
However, “based on prior research, it may be that inflammation and immune dysregulation related to psychological distress play a role in the association of distress with long COVID, but we can’t be sure,” Dr. Roberts said.
Contributes to the field
Commenting for this article, Yapeng Su, PhD, a postdoctoral researcher at the Fred Hutchinson Cancer Research Center in Seattle, called the study “great work contributing to the long-COVID research field and revealing important connections” with psychological stress prior to infection.
Dr. Su, who was not involved with the study, was previously at the Institute for Systems Biology, also in Seattle, and has written about long COVID.
He noted that the “biological mechanism of such intriguing linkage is definitely the important next step, which will likely require deep phenotyping of biological specimens from these patients longitudinally.”
Dr. Wang pointed to past research suggesting that some patients with mental illness “sometimes develop autoantibodies that have also been associated with increased risk of long COVID.” In addition, depression “affects the brain in ways that may explain certain cognitive symptoms in long COVID,” she added.
More studies are now needed to understand how psychological distress increases the risk for long COVID, said Dr. Wang.
The research was supported by grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Institutes of Health, the Dean’s Fund for Scientific Advancement Acceleration Award from the Harvard T. H. Chan School of Public Health, the Massachusetts Consortium on Pathogen Readiness Evergrande COVID-19 Response Fund Award, and the Veterans Affairs Health Services Research and Development Service funds. Dr. Wang and Dr. Roberts have reported no relevant financial relationships. The other investigators’ disclosures are listed in the original article. Dr. Su reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In an analysis of almost 55,000 adult participants in three ongoing studies, having depression, anxiety, worry, perceived stress, or loneliness early in the pandemic, before SARS-CoV-2 infection, was associated with a 50% increased risk for developing long COVID. These types of psychological distress were also associated with a 15% to 51% greater risk for impairment in daily life among individuals with long COVID.
Psychological distress was even more strongly associated with developing long COVID than were physical health risk factors, and the increased risk was not explained by health behaviors such as smoking or physical comorbidities, researchers note.
“Our findings suggest the need to consider psychological health in addition to physical health as risk factors of long COVID-19,” lead author Siwen Wang, MD, postdoctoral fellow, department of nutrition, Harvard T. H. Chan School of Public Health, Boston, said in an interview.
“We need to increase public awareness of the importance of mental health and focus on getting mental health care for people who need it, increasing the supply of mental health clinicians and improving access to care,” she said.
The findings were published online in JAMA Psychiatry.
‘Poorly understood’
Postacute sequelae of SARS-CoV-2 (“long COVID”), which are “signs and symptoms consistent with COVID-19 that extend beyond 4 weeks from onset of infection” constitute “an emerging health issue,” the investigators write.
Dr. Wang noted that it has been estimated that 8-23 million Americans have developed long COVID. However, “despite the high prevalence and daily life impairment associated with long COVID, it is still poorly understood, and few risk factors have been established,” she said.
Although psychological distress may be implicated in long COVID, only three previous studies investigated psychological factors as potential contributors, the researchers note. Also, no study has investigated the potential role of other common manifestations of distress that have increased during the pandemic, such as loneliness and perceived stress, they add.
To investigate these issues, the researchers turned to three large ongoing longitudinal studies: the Nurses’ Health Study II (NSHII), the Nurses’ Health study 3 (NHS3), and the Growing Up Today Study (GUTS).
They analyzed data on 54,960 total participants (96.6% women; mean age, 57.5 years). Of the full group, 38% were active health care workers.
Participants completed an online COVID-19 questionnaire from April 2020 to Sept. 1, 2020 (baseline), and monthly surveys thereafter. Beginning in August 2020, surveys were administered quarterly. The end of follow-up was in November 2021.
The COVID questionnaires included questions about positive SARS-CoV-2 test results, COVID symptoms and hospitalization since March 1, 2020, and the presence of long-term COVID symptoms, such as fatigue, respiratory problems, persistent cough, muscle/joint/chest pain, smell/taste problems, confusion/disorientation/brain fog, depression/anxiety/changes in mood, headache, and memory problems.
Participants who reported these post-COVID conditions were asked about the frequency of symptoms and the degree of impairment in daily life.
Inflammation, immune dysregulation implicated?
The Patient Health Questionnaire–4 (PHQ-4) was used to assess for anxiety and depressive symptoms in the past 2 weeks. It consists of a two-item depression measure (PHQ-2) and a two-item Generalized Anxiety Disorder Scale (GAD-2).
Non–health care providers completed two additional assessments of psychological distress: the four-item Perceived Stress Scale and the three-item UCLA Loneliness Scale.
The researchers included demographic factors, weight, smoking status, marital status, and medical conditions, including diabetes, hypertension, hypercholesterolemia, asthma, and cancer, and socioeconomic factors as covariates.
For each participant, the investigators calculated the number of types of distress experienced at a high level, including probable depression, probable anxiety, worry about COVID-19, being in the top quartile of perceived stress, and loneliness.
During the 19 months of follow-up (1-47 weeks after baseline), 6% of respondents reported a positive result on a SARS-CoV-2 antibody, antigen, or polymerase chain reaction test.
Of these, 43.9% reported long-COVID conditions, with most reporting that symptoms lasted 2 months or longer; 55.8% reported at least occasional daily life impairment.
The most common post-COVID conditions were fatigue (reported by 56%), loss of smell or taste problems (44.6%), shortness of breath (25.5%), confusion/disorientation/ brain fog (24.5%), and memory issues (21.8%).
Among patients who had been infected, there was a considerably higher rate of preinfection psychological distress after adjusting for sociodemographic factors, health behaviors, and comorbidities. Each type of distress was associated with post-COVID conditions.
In addition, participants who had experienced at least two types of distress prior to infection were at nearly 50% increased risk for post–COVID conditions (risk ratio, 1.49; 95% confidence interval, 1.23-1.80).
Among those with post-COVID conditions, all types of distress were associated with increased risk for daily life impairment (RR range, 1.15-1.51).
Senior author Andrea Roberts, PhD, senior research scientist at the Harvard T. H. Chan School of Public Health, Boston, noted that the investigators did not examine biological mechanisms potentially underlying the association they found.
However, “based on prior research, it may be that inflammation and immune dysregulation related to psychological distress play a role in the association of distress with long COVID, but we can’t be sure,” Dr. Roberts said.
Contributes to the field
Commenting for this article, Yapeng Su, PhD, a postdoctoral researcher at the Fred Hutchinson Cancer Research Center in Seattle, called the study “great work contributing to the long-COVID research field and revealing important connections” with psychological stress prior to infection.
Dr. Su, who was not involved with the study, was previously at the Institute for Systems Biology, also in Seattle, and has written about long COVID.
He noted that the “biological mechanism of such intriguing linkage is definitely the important next step, which will likely require deep phenotyping of biological specimens from these patients longitudinally.”
Dr. Wang pointed to past research suggesting that some patients with mental illness “sometimes develop autoantibodies that have also been associated with increased risk of long COVID.” In addition, depression “affects the brain in ways that may explain certain cognitive symptoms in long COVID,” she added.
More studies are now needed to understand how psychological distress increases the risk for long COVID, said Dr. Wang.
The research was supported by grants from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Institutes of Health, the Dean’s Fund for Scientific Advancement Acceleration Award from the Harvard T. H. Chan School of Public Health, the Massachusetts Consortium on Pathogen Readiness Evergrande COVID-19 Response Fund Award, and the Veterans Affairs Health Services Research and Development Service funds. Dr. Wang and Dr. Roberts have reported no relevant financial relationships. The other investigators’ disclosures are listed in the original article. Dr. Su reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA PSYCHIATRY
How do you live with COVID? One doctor’s personal experience
Early in 2020, Anne Peters, MD, caught COVID-19. The author of Medscape’s “Peters on Diabetes” column was sick in March 2020 before state-mandated lockdowns, and well before there were any vaccines.
She remembers sitting in a small exam room with two patients who had flown to her Los Angeles office from New York. The elderly couple had hearing difficulties, so Dr. Peters sat close to them, putting on a continuous glucose monitor. “At that time, we didn’t think of COVID-19 as being in L.A.,” Dr. Peters recalled, “so I think we were not terribly consistent at mask-wearing due to the need to educate.”
“Several days later, I got COVID, but I didn’t know I had COVID per se. I felt crappy, had a terrible sore throat, lost my sense of taste and smell [which was not yet described as a COVID symptom], was completely exhausted, but had no fever or cough, which were the only criteria for getting COVID tested at the time. I didn’t know I had been exposed until 2 weeks later, when the patient’s assistant returned the sensor warning us to ‘be careful’ with it because the patient and his wife were recovering from COVID.”
That early battle with COVID-19 was just the beginning of what would become a 2-year struggle, including familial loss amid her own health problems and concerns about the under-resourced patients she cares for. Here, she shares her journey through the pandemic with this news organization.
Question: Thanks for talking to us. Let’s discuss your journey over these past 2.5 years.
Answer: Everybody has their own COVID story because we all went through this together. Some of us have worse COVID stories, and some of us have better ones, but all have been impacted.
I’m not a sick person. I’m a very healthy person but COVID made me so unwell for 2 years. The brain fog and fatigue were nothing compared to the autonomic neuropathy that affected my heart. It was really limiting for me. And I still don’t know the long-term implications, looking 20-30 years from now.
Q: When you initially had COVID, what were your symptoms? What was the impact?
A: I had all the symptoms of COVID, except for a cough and fever. I lost my sense of taste and smell. I had a horrible headache, a sore throat, and I was exhausted. I couldn’t get tested because I didn’t have the right symptoms.
Despite being sick, I never stopped working but just switched to telemedicine. I also took my regular monthly trip to our cabin in Montana. I unknowingly flew on a plane with COVID. I wore a well-fitted N95 mask, so I don’t think I gave anybody COVID. I didn’t give COVID to my partner, Eric, which is hard to believe as – at 77 – he’s older than me. He has diabetes, heart disease, and every other high-risk characteristic. If he’d gotten COVID back then, it would have been terrible, as there were no treatments, but luckily he didn’t get it.
Q: When were you officially diagnosed?
A: Two or 3 months after I thought I might have had COVID, I checked my antibodies, which tested strongly positive for a prior COVID infection. That was when I knew all the symptoms I’d had were due to the disease.
Q: Not only were you dealing with your own illness, but also that of those close to you. Can you talk about that?
A: In April 2020, my mother who was in her 90s and otherwise healthy except for dementia, got COVID. She could have gotten it from me. I visited often but wore a mask. She had all the horrible pulmonary symptoms. In her advance directive, she didn’t want to be hospitalized so I kept her in her home. She died from COVID in her own bed. It was fairly brutal, but at least I kept her where she felt comforted.
My 91-year-old dad was living in a different residential facility. Throughout COVID he had become very depressed because his social patterns had changed. Prior to COVID, they all ate together, but during the pandemic they were unable to. He missed his social connections, disliked being isolated in his room, hated everyone in masks.
He was a bit demented, but not so much that he couldn’t communicate with me or remember where his grandson was going to law school. I wasn’t allowed inside the facility, which was hard on him. I hadn’t told him his wife died because the hospice social workers advised me that I shouldn’t give him news that he couldn’t process readily until I could spend time with him. Unfortunately, that time never came. In December 2020, he got COVID. One of the people in that facility had gone to the hospital, came back, and tested negative, but actually had COVID and gave it to my dad. The guy who gave it to my dad didn’t die but my dad was terribly ill. He died 2 weeks short of getting his vaccine. He was coherent enough to have a conversation. I asked him: ‘Do you want to go to the hospital?’ And he said: ‘No, because it would be too scary,’ since he couldn’t be with me. I put him on hospice and held his hand as he died from pulmonary COVID, which was awful. I couldn’t give him enough morphine or valium to ease his breathing. But his last words to me were “I love you,” and at the very end he seemed peaceful, which was a blessing.
I got an autopsy, because he wanted one. Nothing else was wrong with him other than COVID. It destroyed his lungs. The rest of him was fine – no heart disease, cancer, or anything else. He died of COVID-19, the same as my mother.
That same week, my aunt, my only surviving older relative, who was in Des Moines, Iowa, died of COVID-19. All three family members died before the vaccine came out.
It was hard to lose my parents. I’m the only surviving child because my sister died in her 20s. It’s not been an easy pandemic. But what pandemic is easy? I just happened to have lost more people than most. Ironically, my grandfather was one of the legionnaires at the Bellevue-Stratford Hotel in Philadelphia in 1976 and died of Legionnaire’s disease before we knew what was causing the outbreak.
Q: Were you still struggling with COVID?
A: COVID impacted my whole body. I lost a lot of weight. I didn’t want to eat, and my gastrointestinal system was not happy. It took a while for my sense of taste and smell to come back. Nothing tasted good. I’m not a foodie; I don’t really care about food. We could get takeout or whatever, but none of it appealed to me. I’m not so sure it was a taste thing, I just didn’t feel like eating.
I didn’t realize I had “brain fog” per se, because I felt stressed and overwhelmed by the pandemic and my patients’ concerns. But one day, about 3 months after I had developed COVID, I woke up without the fog. Which made me aware that I hadn’t been feeling right up until that point.
The worst symptoms, however, were cardiac. I noticed also immediately that my heart rate went up very quickly with minimal exertion. My pulse has always been in the 55-60 bpm range, and suddenly just walking across a room made it go up to over 140 bpm. If I did any aerobic activity, it went up over 160 and would be associated with dyspnea and chest pain. I believed these were all post-COVID symptoms and felt validated when reports of others having similar issues were published in the literature.
Q: Did you continue seeing patients?
A: Yes, of course. Patients never needed their doctors more. In East L.A., where patients don’t have easy access to telemedicine, I kept going into clinic throughout the pandemic. In the more affluent Westside of Los Angeles, we switched to telemedicine, which was quite effective for most. However, because diabetes was associated with an increased risk of hospitalization and death from COVID, my patients were understandably afraid. I’ve never been busier, but (like all health care providers), I became more of a COVID provider than a diabetologist.
Q: Do you feel your battle with COVID impacted your work?
A: It didn’t affect me at work. If I was sitting still, I was fine. Sitting at home at a desk, I didn’t notice any symptoms. But as a habitual stair-user, I would be gasping for breath in the stairwell because I couldn’t go up the stairs to my office as I once could.
I think you empathize more with people who had COVID (when you’ve had it yourself). There was such a huge patient burden. And I think that’s been the thing that’s affected health care providers the most – no matter what specialty we’re in – that nobody has answers.
Q: What happened after you had your vaccine?
A: The vaccine itself was fine. I didn’t have any reaction to the first two doses. But the first booster made my cardiac issues worse.
By this point, my cardiac problems stopped me from exercising. I even went to the ER with chest pain once because I was having palpitations and chest pressure caused by simply taking my morning shower. Fortunately, I wasn’t having an MI, but I certainly wasn’t “normal.”
My measure of my fitness is the cross-country skiing trail I use in Montana. I know exactly how far I can ski. Usually I can do the loop in 35 minutes. After COVID, I lasted 10 minutes. I would be tachycardic, short of breath with chest pain radiating down my left arm. I would rest and try to keep going. But with each rest period, I only got worse. I would be laying in the snow and strangers would ask if I needed help.
Q: What helped you?
A: I’ve read a lot about long COVID and have tried to learn from the experts. Of course, I never went to a doctor directly, although I did ask colleagues for advice. What I learned was to never push myself. I forced myself to create an exercise schedule where I only exercised three times a week with rest days in between. When exercising, the second my heart rate went above 140 bpm, I stopped until I could get it back down. I would push against this new limit, even though my limit was low.
Additionally, I worked on my breathing patterns and did meditative breathing for 10 minutes twice daily using a commercially available app.
Although progress was slow, I did improve, and by June 2022, I seemed back to normal. I was not as fit as I was prior to COVID and needed to improve, but the tachycardic response to exercise and cardiac symptoms were gone. I felt like my normal self. Normal enough to go on a spot packing trip in the Sierras in August. (Horses carried us and a mule carried the gear over the 12,000-foot pass into the mountains, and then left my friend and me high in the Sierras for a week.) We were camped above 10,000 feet and every day hiked up to another high mountain lake where we fly-fished for trout that we ate for dinner. The hikes were a challenge, but not abnormally so. Not as they would have been while I had long COVID.
Q: What is the current atmosphere in your clinic?
A: COVID is much milder now in my vaccinated patients, but I feel most health care providers are exhausted. Many of my staff left when COVID hit because they didn’t want to keep working. It made practicing medicine exhausting. There’s been a shortage of nurses, a shortage of everything. We’ve been required to do a whole lot more than we ever did before. It’s much harder to be a doctor. This pandemic is the first time I’ve ever thought of quitting. Granted, I lost my whole family, or at least the older generation, but it’s just been almost overwhelming.
On the plus side, almost every one of my patients has been vaccinated, because early on, people would ask: “Do you trust this vaccine?” I would reply: “I saw my parents die from COVID when they weren’t vaccinated, so you’re getting vaccinated. This is real and the vaccines help.” It made me very good at convincing people to get vaccines because I knew what it was like to see someone dying from COVID up close.
Q: What advice do you have for those struggling with the COVID pandemic?
A: People need to decide what their own risk is for getting sick and how many times they want to get COVID. At this point, I want people to go out, but safely. In the beginning, when my patients said, “can I go visit my granddaughter?” I said, “no,” but that was before we had the vaccine. Now I feel it is safe to go out using common sense. I still have my patients wear masks on planes. I still have patients try to eat outside as much as possible. And I tell people to take the precautions that make sense, but I tell them to go out and do things because life is short.
I had a patient in his 70s who has many risk factors like heart disease and diabetes. His granddaughter’s Bat Mitzvah in Florida was coming up. He asked: “Can I go?” I told him “Yes,” but to be safe – to wear an N95 mask on the plane and at the event, and stay in his own hotel room, rather than with the whole family. I said, “You need to do this.” Earlier in the pandemic, I saw people who literally died from loneliness and isolation.
He and his wife flew there. He sent me a picture of himself with his granddaughter. When he returned, he showed me a handwritten note from her that said, “I love you so much. Everyone else canceled, which made me cry. You’re the only one who came. You have no idea how much this meant to me.”
He’s back in L.A., and he didn’t get COVID. He said, “It was the best thing I’ve done in years.” That’s what I need to help people with, navigating this world with COVID and assessing risks and benefits. As with all of medicine, my advice is individualized. My advice changes based on the major circulating variant and the rates of the virus in the population, as well as the risk factors of the individual.
Q: What are you doing now?
A: I’m trying to avoid getting COVID again, or another booster. I could get pre-exposure monoclonal antibodies but am waiting to do anything further until I see what happens over the fall and winter. I still wear a mask inside but now do a mix of in-person and telemedicine visits. I still try to go to outdoor restaurants, which is easy in California. But I’m flying to see my son in New York and plan to go to Europe this fall for a meeting. I also go to my cabin in Montana every month to get my “dose” of the wilderness. Overall, I travel for conferences and speaking engagements much less because I have learned the joy of staying home.
Thinking back on my life as a doctor, my career began as an intern at Stanford rotating through Ward 5B, the AIDS unit at San Francisco General Hospital, and will likely end with COVID. In spite of all our medical advances, my generation of physicians, much as many generations before us, has a front-row seat to the vulnerability of humans to infectious diseases and how far we still need to go to protect our patients from communicable illness.
A version of this article first appeared on Medscape.com.
Anne L. Peters, MD, is a professor of medicine at the University of Southern California, Los Angeles, and director of the USC clinical diabetes programs. She has published more than 200 articles, reviews, and abstracts; three books on diabetes; and has been an investigator for more than 40 research studies. She has spoken internationally at over 400 programs and serves on many committees of several professional organizations.
Early in 2020, Anne Peters, MD, caught COVID-19. The author of Medscape’s “Peters on Diabetes” column was sick in March 2020 before state-mandated lockdowns, and well before there were any vaccines.
She remembers sitting in a small exam room with two patients who had flown to her Los Angeles office from New York. The elderly couple had hearing difficulties, so Dr. Peters sat close to them, putting on a continuous glucose monitor. “At that time, we didn’t think of COVID-19 as being in L.A.,” Dr. Peters recalled, “so I think we were not terribly consistent at mask-wearing due to the need to educate.”
“Several days later, I got COVID, but I didn’t know I had COVID per se. I felt crappy, had a terrible sore throat, lost my sense of taste and smell [which was not yet described as a COVID symptom], was completely exhausted, but had no fever or cough, which were the only criteria for getting COVID tested at the time. I didn’t know I had been exposed until 2 weeks later, when the patient’s assistant returned the sensor warning us to ‘be careful’ with it because the patient and his wife were recovering from COVID.”
That early battle with COVID-19 was just the beginning of what would become a 2-year struggle, including familial loss amid her own health problems and concerns about the under-resourced patients she cares for. Here, she shares her journey through the pandemic with this news organization.
Question: Thanks for talking to us. Let’s discuss your journey over these past 2.5 years.
Answer: Everybody has their own COVID story because we all went through this together. Some of us have worse COVID stories, and some of us have better ones, but all have been impacted.
I’m not a sick person. I’m a very healthy person but COVID made me so unwell for 2 years. The brain fog and fatigue were nothing compared to the autonomic neuropathy that affected my heart. It was really limiting for me. And I still don’t know the long-term implications, looking 20-30 years from now.
Q: When you initially had COVID, what were your symptoms? What was the impact?
A: I had all the symptoms of COVID, except for a cough and fever. I lost my sense of taste and smell. I had a horrible headache, a sore throat, and I was exhausted. I couldn’t get tested because I didn’t have the right symptoms.
Despite being sick, I never stopped working but just switched to telemedicine. I also took my regular monthly trip to our cabin in Montana. I unknowingly flew on a plane with COVID. I wore a well-fitted N95 mask, so I don’t think I gave anybody COVID. I didn’t give COVID to my partner, Eric, which is hard to believe as – at 77 – he’s older than me. He has diabetes, heart disease, and every other high-risk characteristic. If he’d gotten COVID back then, it would have been terrible, as there were no treatments, but luckily he didn’t get it.
Q: When were you officially diagnosed?
A: Two or 3 months after I thought I might have had COVID, I checked my antibodies, which tested strongly positive for a prior COVID infection. That was when I knew all the symptoms I’d had were due to the disease.
Q: Not only were you dealing with your own illness, but also that of those close to you. Can you talk about that?
A: In April 2020, my mother who was in her 90s and otherwise healthy except for dementia, got COVID. She could have gotten it from me. I visited often but wore a mask. She had all the horrible pulmonary symptoms. In her advance directive, she didn’t want to be hospitalized so I kept her in her home. She died from COVID in her own bed. It was fairly brutal, but at least I kept her where she felt comforted.
My 91-year-old dad was living in a different residential facility. Throughout COVID he had become very depressed because his social patterns had changed. Prior to COVID, they all ate together, but during the pandemic they were unable to. He missed his social connections, disliked being isolated in his room, hated everyone in masks.
He was a bit demented, but not so much that he couldn’t communicate with me or remember where his grandson was going to law school. I wasn’t allowed inside the facility, which was hard on him. I hadn’t told him his wife died because the hospice social workers advised me that I shouldn’t give him news that he couldn’t process readily until I could spend time with him. Unfortunately, that time never came. In December 2020, he got COVID. One of the people in that facility had gone to the hospital, came back, and tested negative, but actually had COVID and gave it to my dad. The guy who gave it to my dad didn’t die but my dad was terribly ill. He died 2 weeks short of getting his vaccine. He was coherent enough to have a conversation. I asked him: ‘Do you want to go to the hospital?’ And he said: ‘No, because it would be too scary,’ since he couldn’t be with me. I put him on hospice and held his hand as he died from pulmonary COVID, which was awful. I couldn’t give him enough morphine or valium to ease his breathing. But his last words to me were “I love you,” and at the very end he seemed peaceful, which was a blessing.
I got an autopsy, because he wanted one. Nothing else was wrong with him other than COVID. It destroyed his lungs. The rest of him was fine – no heart disease, cancer, or anything else. He died of COVID-19, the same as my mother.
That same week, my aunt, my only surviving older relative, who was in Des Moines, Iowa, died of COVID-19. All three family members died before the vaccine came out.
It was hard to lose my parents. I’m the only surviving child because my sister died in her 20s. It’s not been an easy pandemic. But what pandemic is easy? I just happened to have lost more people than most. Ironically, my grandfather was one of the legionnaires at the Bellevue-Stratford Hotel in Philadelphia in 1976 and died of Legionnaire’s disease before we knew what was causing the outbreak.
Q: Were you still struggling with COVID?
A: COVID impacted my whole body. I lost a lot of weight. I didn’t want to eat, and my gastrointestinal system was not happy. It took a while for my sense of taste and smell to come back. Nothing tasted good. I’m not a foodie; I don’t really care about food. We could get takeout or whatever, but none of it appealed to me. I’m not so sure it was a taste thing, I just didn’t feel like eating.
I didn’t realize I had “brain fog” per se, because I felt stressed and overwhelmed by the pandemic and my patients’ concerns. But one day, about 3 months after I had developed COVID, I woke up without the fog. Which made me aware that I hadn’t been feeling right up until that point.
The worst symptoms, however, were cardiac. I noticed also immediately that my heart rate went up very quickly with minimal exertion. My pulse has always been in the 55-60 bpm range, and suddenly just walking across a room made it go up to over 140 bpm. If I did any aerobic activity, it went up over 160 and would be associated with dyspnea and chest pain. I believed these were all post-COVID symptoms and felt validated when reports of others having similar issues were published in the literature.
Q: Did you continue seeing patients?
A: Yes, of course. Patients never needed their doctors more. In East L.A., where patients don’t have easy access to telemedicine, I kept going into clinic throughout the pandemic. In the more affluent Westside of Los Angeles, we switched to telemedicine, which was quite effective for most. However, because diabetes was associated with an increased risk of hospitalization and death from COVID, my patients were understandably afraid. I’ve never been busier, but (like all health care providers), I became more of a COVID provider than a diabetologist.
Q: Do you feel your battle with COVID impacted your work?
A: It didn’t affect me at work. If I was sitting still, I was fine. Sitting at home at a desk, I didn’t notice any symptoms. But as a habitual stair-user, I would be gasping for breath in the stairwell because I couldn’t go up the stairs to my office as I once could.
I think you empathize more with people who had COVID (when you’ve had it yourself). There was such a huge patient burden. And I think that’s been the thing that’s affected health care providers the most – no matter what specialty we’re in – that nobody has answers.
Q: What happened after you had your vaccine?
A: The vaccine itself was fine. I didn’t have any reaction to the first two doses. But the first booster made my cardiac issues worse.
By this point, my cardiac problems stopped me from exercising. I even went to the ER with chest pain once because I was having palpitations and chest pressure caused by simply taking my morning shower. Fortunately, I wasn’t having an MI, but I certainly wasn’t “normal.”
My measure of my fitness is the cross-country skiing trail I use in Montana. I know exactly how far I can ski. Usually I can do the loop in 35 minutes. After COVID, I lasted 10 minutes. I would be tachycardic, short of breath with chest pain radiating down my left arm. I would rest and try to keep going. But with each rest period, I only got worse. I would be laying in the snow and strangers would ask if I needed help.
Q: What helped you?
A: I’ve read a lot about long COVID and have tried to learn from the experts. Of course, I never went to a doctor directly, although I did ask colleagues for advice. What I learned was to never push myself. I forced myself to create an exercise schedule where I only exercised three times a week with rest days in between. When exercising, the second my heart rate went above 140 bpm, I stopped until I could get it back down. I would push against this new limit, even though my limit was low.
Additionally, I worked on my breathing patterns and did meditative breathing for 10 minutes twice daily using a commercially available app.
Although progress was slow, I did improve, and by June 2022, I seemed back to normal. I was not as fit as I was prior to COVID and needed to improve, but the tachycardic response to exercise and cardiac symptoms were gone. I felt like my normal self. Normal enough to go on a spot packing trip in the Sierras in August. (Horses carried us and a mule carried the gear over the 12,000-foot pass into the mountains, and then left my friend and me high in the Sierras for a week.) We were camped above 10,000 feet and every day hiked up to another high mountain lake where we fly-fished for trout that we ate for dinner. The hikes were a challenge, but not abnormally so. Not as they would have been while I had long COVID.
Q: What is the current atmosphere in your clinic?
A: COVID is much milder now in my vaccinated patients, but I feel most health care providers are exhausted. Many of my staff left when COVID hit because they didn’t want to keep working. It made practicing medicine exhausting. There’s been a shortage of nurses, a shortage of everything. We’ve been required to do a whole lot more than we ever did before. It’s much harder to be a doctor. This pandemic is the first time I’ve ever thought of quitting. Granted, I lost my whole family, or at least the older generation, but it’s just been almost overwhelming.
On the plus side, almost every one of my patients has been vaccinated, because early on, people would ask: “Do you trust this vaccine?” I would reply: “I saw my parents die from COVID when they weren’t vaccinated, so you’re getting vaccinated. This is real and the vaccines help.” It made me very good at convincing people to get vaccines because I knew what it was like to see someone dying from COVID up close.
Q: What advice do you have for those struggling with the COVID pandemic?
A: People need to decide what their own risk is for getting sick and how many times they want to get COVID. At this point, I want people to go out, but safely. In the beginning, when my patients said, “can I go visit my granddaughter?” I said, “no,” but that was before we had the vaccine. Now I feel it is safe to go out using common sense. I still have my patients wear masks on planes. I still have patients try to eat outside as much as possible. And I tell people to take the precautions that make sense, but I tell them to go out and do things because life is short.
I had a patient in his 70s who has many risk factors like heart disease and diabetes. His granddaughter’s Bat Mitzvah in Florida was coming up. He asked: “Can I go?” I told him “Yes,” but to be safe – to wear an N95 mask on the plane and at the event, and stay in his own hotel room, rather than with the whole family. I said, “You need to do this.” Earlier in the pandemic, I saw people who literally died from loneliness and isolation.
He and his wife flew there. He sent me a picture of himself with his granddaughter. When he returned, he showed me a handwritten note from her that said, “I love you so much. Everyone else canceled, which made me cry. You’re the only one who came. You have no idea how much this meant to me.”
He’s back in L.A., and he didn’t get COVID. He said, “It was the best thing I’ve done in years.” That’s what I need to help people with, navigating this world with COVID and assessing risks and benefits. As with all of medicine, my advice is individualized. My advice changes based on the major circulating variant and the rates of the virus in the population, as well as the risk factors of the individual.
Q: What are you doing now?
A: I’m trying to avoid getting COVID again, or another booster. I could get pre-exposure monoclonal antibodies but am waiting to do anything further until I see what happens over the fall and winter. I still wear a mask inside but now do a mix of in-person and telemedicine visits. I still try to go to outdoor restaurants, which is easy in California. But I’m flying to see my son in New York and plan to go to Europe this fall for a meeting. I also go to my cabin in Montana every month to get my “dose” of the wilderness. Overall, I travel for conferences and speaking engagements much less because I have learned the joy of staying home.
Thinking back on my life as a doctor, my career began as an intern at Stanford rotating through Ward 5B, the AIDS unit at San Francisco General Hospital, and will likely end with COVID. In spite of all our medical advances, my generation of physicians, much as many generations before us, has a front-row seat to the vulnerability of humans to infectious diseases and how far we still need to go to protect our patients from communicable illness.
A version of this article first appeared on Medscape.com.
Anne L. Peters, MD, is a professor of medicine at the University of Southern California, Los Angeles, and director of the USC clinical diabetes programs. She has published more than 200 articles, reviews, and abstracts; three books on diabetes; and has been an investigator for more than 40 research studies. She has spoken internationally at over 400 programs and serves on many committees of several professional organizations.
Early in 2020, Anne Peters, MD, caught COVID-19. The author of Medscape’s “Peters on Diabetes” column was sick in March 2020 before state-mandated lockdowns, and well before there were any vaccines.
She remembers sitting in a small exam room with two patients who had flown to her Los Angeles office from New York. The elderly couple had hearing difficulties, so Dr. Peters sat close to them, putting on a continuous glucose monitor. “At that time, we didn’t think of COVID-19 as being in L.A.,” Dr. Peters recalled, “so I think we were not terribly consistent at mask-wearing due to the need to educate.”
“Several days later, I got COVID, but I didn’t know I had COVID per se. I felt crappy, had a terrible sore throat, lost my sense of taste and smell [which was not yet described as a COVID symptom], was completely exhausted, but had no fever or cough, which were the only criteria for getting COVID tested at the time. I didn’t know I had been exposed until 2 weeks later, when the patient’s assistant returned the sensor warning us to ‘be careful’ with it because the patient and his wife were recovering from COVID.”
That early battle with COVID-19 was just the beginning of what would become a 2-year struggle, including familial loss amid her own health problems and concerns about the under-resourced patients she cares for. Here, she shares her journey through the pandemic with this news organization.
Question: Thanks for talking to us. Let’s discuss your journey over these past 2.5 years.
Answer: Everybody has their own COVID story because we all went through this together. Some of us have worse COVID stories, and some of us have better ones, but all have been impacted.
I’m not a sick person. I’m a very healthy person but COVID made me so unwell for 2 years. The brain fog and fatigue were nothing compared to the autonomic neuropathy that affected my heart. It was really limiting for me. And I still don’t know the long-term implications, looking 20-30 years from now.
Q: When you initially had COVID, what were your symptoms? What was the impact?
A: I had all the symptoms of COVID, except for a cough and fever. I lost my sense of taste and smell. I had a horrible headache, a sore throat, and I was exhausted. I couldn’t get tested because I didn’t have the right symptoms.
Despite being sick, I never stopped working but just switched to telemedicine. I also took my regular monthly trip to our cabin in Montana. I unknowingly flew on a plane with COVID. I wore a well-fitted N95 mask, so I don’t think I gave anybody COVID. I didn’t give COVID to my partner, Eric, which is hard to believe as – at 77 – he’s older than me. He has diabetes, heart disease, and every other high-risk characteristic. If he’d gotten COVID back then, it would have been terrible, as there were no treatments, but luckily he didn’t get it.
Q: When were you officially diagnosed?
A: Two or 3 months after I thought I might have had COVID, I checked my antibodies, which tested strongly positive for a prior COVID infection. That was when I knew all the symptoms I’d had were due to the disease.
Q: Not only were you dealing with your own illness, but also that of those close to you. Can you talk about that?
A: In April 2020, my mother who was in her 90s and otherwise healthy except for dementia, got COVID. She could have gotten it from me. I visited often but wore a mask. She had all the horrible pulmonary symptoms. In her advance directive, she didn’t want to be hospitalized so I kept her in her home. She died from COVID in her own bed. It was fairly brutal, but at least I kept her where she felt comforted.
My 91-year-old dad was living in a different residential facility. Throughout COVID he had become very depressed because his social patterns had changed. Prior to COVID, they all ate together, but during the pandemic they were unable to. He missed his social connections, disliked being isolated in his room, hated everyone in masks.
He was a bit demented, but not so much that he couldn’t communicate with me or remember where his grandson was going to law school. I wasn’t allowed inside the facility, which was hard on him. I hadn’t told him his wife died because the hospice social workers advised me that I shouldn’t give him news that he couldn’t process readily until I could spend time with him. Unfortunately, that time never came. In December 2020, he got COVID. One of the people in that facility had gone to the hospital, came back, and tested negative, but actually had COVID and gave it to my dad. The guy who gave it to my dad didn’t die but my dad was terribly ill. He died 2 weeks short of getting his vaccine. He was coherent enough to have a conversation. I asked him: ‘Do you want to go to the hospital?’ And he said: ‘No, because it would be too scary,’ since he couldn’t be with me. I put him on hospice and held his hand as he died from pulmonary COVID, which was awful. I couldn’t give him enough morphine or valium to ease his breathing. But his last words to me were “I love you,” and at the very end he seemed peaceful, which was a blessing.
I got an autopsy, because he wanted one. Nothing else was wrong with him other than COVID. It destroyed his lungs. The rest of him was fine – no heart disease, cancer, or anything else. He died of COVID-19, the same as my mother.
That same week, my aunt, my only surviving older relative, who was in Des Moines, Iowa, died of COVID-19. All three family members died before the vaccine came out.
It was hard to lose my parents. I’m the only surviving child because my sister died in her 20s. It’s not been an easy pandemic. But what pandemic is easy? I just happened to have lost more people than most. Ironically, my grandfather was one of the legionnaires at the Bellevue-Stratford Hotel in Philadelphia in 1976 and died of Legionnaire’s disease before we knew what was causing the outbreak.
Q: Were you still struggling with COVID?
A: COVID impacted my whole body. I lost a lot of weight. I didn’t want to eat, and my gastrointestinal system was not happy. It took a while for my sense of taste and smell to come back. Nothing tasted good. I’m not a foodie; I don’t really care about food. We could get takeout or whatever, but none of it appealed to me. I’m not so sure it was a taste thing, I just didn’t feel like eating.
I didn’t realize I had “brain fog” per se, because I felt stressed and overwhelmed by the pandemic and my patients’ concerns. But one day, about 3 months after I had developed COVID, I woke up without the fog. Which made me aware that I hadn’t been feeling right up until that point.
The worst symptoms, however, were cardiac. I noticed also immediately that my heart rate went up very quickly with minimal exertion. My pulse has always been in the 55-60 bpm range, and suddenly just walking across a room made it go up to over 140 bpm. If I did any aerobic activity, it went up over 160 and would be associated with dyspnea and chest pain. I believed these were all post-COVID symptoms and felt validated when reports of others having similar issues were published in the literature.
Q: Did you continue seeing patients?
A: Yes, of course. Patients never needed their doctors more. In East L.A., where patients don’t have easy access to telemedicine, I kept going into clinic throughout the pandemic. In the more affluent Westside of Los Angeles, we switched to telemedicine, which was quite effective for most. However, because diabetes was associated with an increased risk of hospitalization and death from COVID, my patients were understandably afraid. I’ve never been busier, but (like all health care providers), I became more of a COVID provider than a diabetologist.
Q: Do you feel your battle with COVID impacted your work?
A: It didn’t affect me at work. If I was sitting still, I was fine. Sitting at home at a desk, I didn’t notice any symptoms. But as a habitual stair-user, I would be gasping for breath in the stairwell because I couldn’t go up the stairs to my office as I once could.
I think you empathize more with people who had COVID (when you’ve had it yourself). There was such a huge patient burden. And I think that’s been the thing that’s affected health care providers the most – no matter what specialty we’re in – that nobody has answers.
Q: What happened after you had your vaccine?
A: The vaccine itself was fine. I didn’t have any reaction to the first two doses. But the first booster made my cardiac issues worse.
By this point, my cardiac problems stopped me from exercising. I even went to the ER with chest pain once because I was having palpitations and chest pressure caused by simply taking my morning shower. Fortunately, I wasn’t having an MI, but I certainly wasn’t “normal.”
My measure of my fitness is the cross-country skiing trail I use in Montana. I know exactly how far I can ski. Usually I can do the loop in 35 minutes. After COVID, I lasted 10 minutes. I would be tachycardic, short of breath with chest pain radiating down my left arm. I would rest and try to keep going. But with each rest period, I only got worse. I would be laying in the snow and strangers would ask if I needed help.
Q: What helped you?
A: I’ve read a lot about long COVID and have tried to learn from the experts. Of course, I never went to a doctor directly, although I did ask colleagues for advice. What I learned was to never push myself. I forced myself to create an exercise schedule where I only exercised three times a week with rest days in between. When exercising, the second my heart rate went above 140 bpm, I stopped until I could get it back down. I would push against this new limit, even though my limit was low.
Additionally, I worked on my breathing patterns and did meditative breathing for 10 minutes twice daily using a commercially available app.
Although progress was slow, I did improve, and by June 2022, I seemed back to normal. I was not as fit as I was prior to COVID and needed to improve, but the tachycardic response to exercise and cardiac symptoms were gone. I felt like my normal self. Normal enough to go on a spot packing trip in the Sierras in August. (Horses carried us and a mule carried the gear over the 12,000-foot pass into the mountains, and then left my friend and me high in the Sierras for a week.) We were camped above 10,000 feet and every day hiked up to another high mountain lake where we fly-fished for trout that we ate for dinner. The hikes were a challenge, but not abnormally so. Not as they would have been while I had long COVID.
Q: What is the current atmosphere in your clinic?
A: COVID is much milder now in my vaccinated patients, but I feel most health care providers are exhausted. Many of my staff left when COVID hit because they didn’t want to keep working. It made practicing medicine exhausting. There’s been a shortage of nurses, a shortage of everything. We’ve been required to do a whole lot more than we ever did before. It’s much harder to be a doctor. This pandemic is the first time I’ve ever thought of quitting. Granted, I lost my whole family, or at least the older generation, but it’s just been almost overwhelming.
On the plus side, almost every one of my patients has been vaccinated, because early on, people would ask: “Do you trust this vaccine?” I would reply: “I saw my parents die from COVID when they weren’t vaccinated, so you’re getting vaccinated. This is real and the vaccines help.” It made me very good at convincing people to get vaccines because I knew what it was like to see someone dying from COVID up close.
Q: What advice do you have for those struggling with the COVID pandemic?
A: People need to decide what their own risk is for getting sick and how many times they want to get COVID. At this point, I want people to go out, but safely. In the beginning, when my patients said, “can I go visit my granddaughter?” I said, “no,” but that was before we had the vaccine. Now I feel it is safe to go out using common sense. I still have my patients wear masks on planes. I still have patients try to eat outside as much as possible. And I tell people to take the precautions that make sense, but I tell them to go out and do things because life is short.
I had a patient in his 70s who has many risk factors like heart disease and diabetes. His granddaughter’s Bat Mitzvah in Florida was coming up. He asked: “Can I go?” I told him “Yes,” but to be safe – to wear an N95 mask on the plane and at the event, and stay in his own hotel room, rather than with the whole family. I said, “You need to do this.” Earlier in the pandemic, I saw people who literally died from loneliness and isolation.
He and his wife flew there. He sent me a picture of himself with his granddaughter. When he returned, he showed me a handwritten note from her that said, “I love you so much. Everyone else canceled, which made me cry. You’re the only one who came. You have no idea how much this meant to me.”
He’s back in L.A., and he didn’t get COVID. He said, “It was the best thing I’ve done in years.” That’s what I need to help people with, navigating this world with COVID and assessing risks and benefits. As with all of medicine, my advice is individualized. My advice changes based on the major circulating variant and the rates of the virus in the population, as well as the risk factors of the individual.
Q: What are you doing now?
A: I’m trying to avoid getting COVID again, or another booster. I could get pre-exposure monoclonal antibodies but am waiting to do anything further until I see what happens over the fall and winter. I still wear a mask inside but now do a mix of in-person and telemedicine visits. I still try to go to outdoor restaurants, which is easy in California. But I’m flying to see my son in New York and plan to go to Europe this fall for a meeting. I also go to my cabin in Montana every month to get my “dose” of the wilderness. Overall, I travel for conferences and speaking engagements much less because I have learned the joy of staying home.
Thinking back on my life as a doctor, my career began as an intern at Stanford rotating through Ward 5B, the AIDS unit at San Francisco General Hospital, and will likely end with COVID. In spite of all our medical advances, my generation of physicians, much as many generations before us, has a front-row seat to the vulnerability of humans to infectious diseases and how far we still need to go to protect our patients from communicable illness.
A version of this article first appeared on Medscape.com.
Anne L. Peters, MD, is a professor of medicine at the University of Southern California, Los Angeles, and director of the USC clinical diabetes programs. She has published more than 200 articles, reviews, and abstracts; three books on diabetes; and has been an investigator for more than 40 research studies. She has spoken internationally at over 400 programs and serves on many committees of several professional organizations.
Long COVID mimics other postviral conditions
When Jaime Seltzer first heard about a new virus that was spreading globally early in 2020, she was on full alert. As an advocate for the post-viral condition known as myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), she worried about a new wave of people having long-term disabilities.
“The hair on my arms stood on end,” said Ms. Seltzer, director of scientific and medical outreach at the advocacy group MEAction and a consultant researcher at Stanford University.
Ms. Seltzer, who has had ME/CFS herself, said she wondered.
Sure enough, later in 2020, reports began emerging about people with extreme fatigue, postexertion crashes, brain fog, unrefreshing sleep, and dizziness when standing up months after a bout with the then-new viral illness. Those same symptoms had been designated as “core criteria” of ME/CFS by the National Academy of Medicine in a 2015 report.
Now, advocates like Ms. Seltzer are hoping the research and medical communities will give ME/CFS and other postviral illnesses the same attention they have increasingly focused on long COVID.
The emergence of long COVID was no surprise to researchers who study ME/CFS, because the same set of symptoms has arisen after many other viruses.
“This for all the world looks like ME/CFS. We think they are frighteningly similar, if not identical,” said David M. Systrom, MD, a pulmonary and critical care medicine specialist at Brigham and Women’s Hospital in Boston, who studies people with both diagnoses.
The actual numbers are hard to determine, since many people who meet ME/CFS criteria aren’t formally diagnosed. But a combined analysis of data from several studies published in March found that about one in three people had fatigue and about one in five reported having a hard time with thinking and memory 12 or more weeks after they had COVID-19.
According to some estimates, about half of people with long COVID will meet the criteria for ME/CFS, whether they’re given that specific diagnosis or not.
Other conditions that often exist with ME/CFS are also being seen in people with long COVID, including postural orthostatic tachycardia syndrome, which causes people to feel dizzy when they stand, along with other symptoms; other problems with the autonomic nervous system, which controls body systems such as heart rate, blood pressure, and digestion, known together as dysautonomia; and a condition related to allergies called mast cell activation disorder.
Post–acute infection syndromes have been linked to a long list of viruses, including Ebola, the 2003-2004 SARS virus, and Epstein-Barr – the virus most commonly associated with ME/CFS.
The problem in clinical medicine is that once an infection has cleared, the teaching has been that the person should no longer feel sick, said Nancy G. Klimas, MD, director of the Institute for Neuro-Immune Medicine at Nova Southeastern University in Miami. “I was taught that there has to be an antigen [such as a viral protein] in the system to drive the immune system to make it create sickness, and the immune system should shut off when it’s done,” she said.
Thus, if virus is gone and other routine lab tests come up negative, doctors often deem the person’s reported symptoms to be psychological, which can upset patients, Anthony Komaroff, MD, of Brigham and Women’s Hospital in Boston, wrote in July 2021.
Only recently have doctors started to appreciate the idea that the immune system may be overreacting long term, Dr. Klimas said.
Now, long COVID appears to be speeding up that recognition. Dr. Systrom said he has “absolutely” seen a change in attitude among fellow doctors who had been skeptical of ME/CFS as a “real” illness because there’s no test for it.
“I’m very keenly aware of a large group of health care professionals who really had not bought into the concept of ME/CFS as a real disease who have had an epiphany of sorts with long COVID and now, in a backwards way, have applied that same thinking to their very same patients with ME/CFS,” he said.
Science showing ‘frighteningly similar’ symptoms
Dr. Systrom has spent several years researching how ME/CFS patients cannot tolerate exercise and now is doing similar studies in people with long COVID. “Several months into the pandemic, we began receiving reports of patients who had survived COVID and maybe even had a relatively mild disease ... and as the summer of 2020 moved into the fall, it became apparent that there was a subset of patients who for all the world appeared to meet ME/CFS clinical criteria,” he said.
Using bicycle exercise tests on long COVID patients with catheters placed in their veins, Dr. Systrom and associates have shown a lack of exercise capacity that isn’t caused by heart or lung disease but instead is related to abnormal nerves and blood vessels, just as they’d shown previously in ME/CFS patient.
Avindra Nath, MD, senior investigator and clinical director of intramural research at the National Institute of Neurological Disorders and Stroke, Bethesda, Md., was doing a deep-dive scientific study on ME/CFS when the COVID-19 pandemic hit. Since then, he›s begun another study using the same protocol and sophisticated laboratory measurement to evaluate people with long COVID.
“As terrible as [long COVID] is, it’s kind of a blessing in disguise for ME/CFS because there’s just so much overlap between the two and they could very well be in many ways one in the same thing. The problem with studying ME/CFS is oftentimes you didn’t know what the trigger was. You see patients many years later, then try to backtrack and find out what happened,” said Dr. Nath, a neuroimmunologist.
With long COVID, on the other hand, “we know when they got infected and when their symptoms actually started, so it becomes much more uniform. ... It gives us an opportunity to maybe solve certain things in a much more well-defined population and try to find answers.”
Advocacy groups want to see more.
In February 2021, Solve M.E. launched the Long COVID Alliance, made up of several organizations, companies, and people with a goal to influence policy and speed up research into a range of postviral illnesses.
Solve M.E. has also pushed for inclusion of language regarding ME/CFS and related conditions into congressional bills addressing long COVID, including those that call for funding of research and clinical care.
“On the political front, we’ve really capitalized on a moment in time in which we have the spotlight,” said Emily Taylor, vice president of advocacy and engagement for Solve M.E.
“One of the hardest parts about ME/CFS is how to show that it’s real when it’s invisible. Most people agree that COVID is real and therefore if somebody gets ME/CFS after COVID, it’s real,” she said.
The advocacy groups are now pushing for non-COVID postinfection illnesses to be included in efforts aimed at helping people with long COVID, with mixed results. For example, the RECOVER Initiative, established in February 2021 with $1.5 billion in funding from Congress to the National Institutes of Health, is specifically for studying long COVID and does not fund research into other postinfection illnesses, although representatives from the ME/CFS community are advisers.
Language addressing ME/CFS and other postinfectious chronic illnesses has been included in several long COVID bills now pending in Congress, including the Care for Long COVID Act in the Senate and its companion COVID-19 Long Haulers Act in the House. “Our goal is to push for passage of a long COVID bill by the end of the year,” Ms. Taylor said.
A version of this article first appeared on WebMD.com.
When Jaime Seltzer first heard about a new virus that was spreading globally early in 2020, she was on full alert. As an advocate for the post-viral condition known as myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), she worried about a new wave of people having long-term disabilities.
“The hair on my arms stood on end,” said Ms. Seltzer, director of scientific and medical outreach at the advocacy group MEAction and a consultant researcher at Stanford University.
Ms. Seltzer, who has had ME/CFS herself, said she wondered.
Sure enough, later in 2020, reports began emerging about people with extreme fatigue, postexertion crashes, brain fog, unrefreshing sleep, and dizziness when standing up months after a bout with the then-new viral illness. Those same symptoms had been designated as “core criteria” of ME/CFS by the National Academy of Medicine in a 2015 report.
Now, advocates like Ms. Seltzer are hoping the research and medical communities will give ME/CFS and other postviral illnesses the same attention they have increasingly focused on long COVID.
The emergence of long COVID was no surprise to researchers who study ME/CFS, because the same set of symptoms has arisen after many other viruses.
“This for all the world looks like ME/CFS. We think they are frighteningly similar, if not identical,” said David M. Systrom, MD, a pulmonary and critical care medicine specialist at Brigham and Women’s Hospital in Boston, who studies people with both diagnoses.
The actual numbers are hard to determine, since many people who meet ME/CFS criteria aren’t formally diagnosed. But a combined analysis of data from several studies published in March found that about one in three people had fatigue and about one in five reported having a hard time with thinking and memory 12 or more weeks after they had COVID-19.
According to some estimates, about half of people with long COVID will meet the criteria for ME/CFS, whether they’re given that specific diagnosis or not.
Other conditions that often exist with ME/CFS are also being seen in people with long COVID, including postural orthostatic tachycardia syndrome, which causes people to feel dizzy when they stand, along with other symptoms; other problems with the autonomic nervous system, which controls body systems such as heart rate, blood pressure, and digestion, known together as dysautonomia; and a condition related to allergies called mast cell activation disorder.
Post–acute infection syndromes have been linked to a long list of viruses, including Ebola, the 2003-2004 SARS virus, and Epstein-Barr – the virus most commonly associated with ME/CFS.
The problem in clinical medicine is that once an infection has cleared, the teaching has been that the person should no longer feel sick, said Nancy G. Klimas, MD, director of the Institute for Neuro-Immune Medicine at Nova Southeastern University in Miami. “I was taught that there has to be an antigen [such as a viral protein] in the system to drive the immune system to make it create sickness, and the immune system should shut off when it’s done,” she said.
Thus, if virus is gone and other routine lab tests come up negative, doctors often deem the person’s reported symptoms to be psychological, which can upset patients, Anthony Komaroff, MD, of Brigham and Women’s Hospital in Boston, wrote in July 2021.
Only recently have doctors started to appreciate the idea that the immune system may be overreacting long term, Dr. Klimas said.
Now, long COVID appears to be speeding up that recognition. Dr. Systrom said he has “absolutely” seen a change in attitude among fellow doctors who had been skeptical of ME/CFS as a “real” illness because there’s no test for it.
“I’m very keenly aware of a large group of health care professionals who really had not bought into the concept of ME/CFS as a real disease who have had an epiphany of sorts with long COVID and now, in a backwards way, have applied that same thinking to their very same patients with ME/CFS,” he said.
Science showing ‘frighteningly similar’ symptoms
Dr. Systrom has spent several years researching how ME/CFS patients cannot tolerate exercise and now is doing similar studies in people with long COVID. “Several months into the pandemic, we began receiving reports of patients who had survived COVID and maybe even had a relatively mild disease ... and as the summer of 2020 moved into the fall, it became apparent that there was a subset of patients who for all the world appeared to meet ME/CFS clinical criteria,” he said.
Using bicycle exercise tests on long COVID patients with catheters placed in their veins, Dr. Systrom and associates have shown a lack of exercise capacity that isn’t caused by heart or lung disease but instead is related to abnormal nerves and blood vessels, just as they’d shown previously in ME/CFS patient.
Avindra Nath, MD, senior investigator and clinical director of intramural research at the National Institute of Neurological Disorders and Stroke, Bethesda, Md., was doing a deep-dive scientific study on ME/CFS when the COVID-19 pandemic hit. Since then, he›s begun another study using the same protocol and sophisticated laboratory measurement to evaluate people with long COVID.
“As terrible as [long COVID] is, it’s kind of a blessing in disguise for ME/CFS because there’s just so much overlap between the two and they could very well be in many ways one in the same thing. The problem with studying ME/CFS is oftentimes you didn’t know what the trigger was. You see patients many years later, then try to backtrack and find out what happened,” said Dr. Nath, a neuroimmunologist.
With long COVID, on the other hand, “we know when they got infected and when their symptoms actually started, so it becomes much more uniform. ... It gives us an opportunity to maybe solve certain things in a much more well-defined population and try to find answers.”
Advocacy groups want to see more.
In February 2021, Solve M.E. launched the Long COVID Alliance, made up of several organizations, companies, and people with a goal to influence policy and speed up research into a range of postviral illnesses.
Solve M.E. has also pushed for inclusion of language regarding ME/CFS and related conditions into congressional bills addressing long COVID, including those that call for funding of research and clinical care.
“On the political front, we’ve really capitalized on a moment in time in which we have the spotlight,” said Emily Taylor, vice president of advocacy and engagement for Solve M.E.
“One of the hardest parts about ME/CFS is how to show that it’s real when it’s invisible. Most people agree that COVID is real and therefore if somebody gets ME/CFS after COVID, it’s real,” she said.
The advocacy groups are now pushing for non-COVID postinfection illnesses to be included in efforts aimed at helping people with long COVID, with mixed results. For example, the RECOVER Initiative, established in February 2021 with $1.5 billion in funding from Congress to the National Institutes of Health, is specifically for studying long COVID and does not fund research into other postinfection illnesses, although representatives from the ME/CFS community are advisers.
Language addressing ME/CFS and other postinfectious chronic illnesses has been included in several long COVID bills now pending in Congress, including the Care for Long COVID Act in the Senate and its companion COVID-19 Long Haulers Act in the House. “Our goal is to push for passage of a long COVID bill by the end of the year,” Ms. Taylor said.
A version of this article first appeared on WebMD.com.
When Jaime Seltzer first heard about a new virus that was spreading globally early in 2020, she was on full alert. As an advocate for the post-viral condition known as myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), she worried about a new wave of people having long-term disabilities.
“The hair on my arms stood on end,” said Ms. Seltzer, director of scientific and medical outreach at the advocacy group MEAction and a consultant researcher at Stanford University.
Ms. Seltzer, who has had ME/CFS herself, said she wondered.
Sure enough, later in 2020, reports began emerging about people with extreme fatigue, postexertion crashes, brain fog, unrefreshing sleep, and dizziness when standing up months after a bout with the then-new viral illness. Those same symptoms had been designated as “core criteria” of ME/CFS by the National Academy of Medicine in a 2015 report.
Now, advocates like Ms. Seltzer are hoping the research and medical communities will give ME/CFS and other postviral illnesses the same attention they have increasingly focused on long COVID.
The emergence of long COVID was no surprise to researchers who study ME/CFS, because the same set of symptoms has arisen after many other viruses.
“This for all the world looks like ME/CFS. We think they are frighteningly similar, if not identical,” said David M. Systrom, MD, a pulmonary and critical care medicine specialist at Brigham and Women’s Hospital in Boston, who studies people with both diagnoses.
The actual numbers are hard to determine, since many people who meet ME/CFS criteria aren’t formally diagnosed. But a combined analysis of data from several studies published in March found that about one in three people had fatigue and about one in five reported having a hard time with thinking and memory 12 or more weeks after they had COVID-19.
According to some estimates, about half of people with long COVID will meet the criteria for ME/CFS, whether they’re given that specific diagnosis or not.
Other conditions that often exist with ME/CFS are also being seen in people with long COVID, including postural orthostatic tachycardia syndrome, which causes people to feel dizzy when they stand, along with other symptoms; other problems with the autonomic nervous system, which controls body systems such as heart rate, blood pressure, and digestion, known together as dysautonomia; and a condition related to allergies called mast cell activation disorder.
Post–acute infection syndromes have been linked to a long list of viruses, including Ebola, the 2003-2004 SARS virus, and Epstein-Barr – the virus most commonly associated with ME/CFS.
The problem in clinical medicine is that once an infection has cleared, the teaching has been that the person should no longer feel sick, said Nancy G. Klimas, MD, director of the Institute for Neuro-Immune Medicine at Nova Southeastern University in Miami. “I was taught that there has to be an antigen [such as a viral protein] in the system to drive the immune system to make it create sickness, and the immune system should shut off when it’s done,” she said.
Thus, if virus is gone and other routine lab tests come up negative, doctors often deem the person’s reported symptoms to be psychological, which can upset patients, Anthony Komaroff, MD, of Brigham and Women’s Hospital in Boston, wrote in July 2021.
Only recently have doctors started to appreciate the idea that the immune system may be overreacting long term, Dr. Klimas said.
Now, long COVID appears to be speeding up that recognition. Dr. Systrom said he has “absolutely” seen a change in attitude among fellow doctors who had been skeptical of ME/CFS as a “real” illness because there’s no test for it.
“I’m very keenly aware of a large group of health care professionals who really had not bought into the concept of ME/CFS as a real disease who have had an epiphany of sorts with long COVID and now, in a backwards way, have applied that same thinking to their very same patients with ME/CFS,” he said.
Science showing ‘frighteningly similar’ symptoms
Dr. Systrom has spent several years researching how ME/CFS patients cannot tolerate exercise and now is doing similar studies in people with long COVID. “Several months into the pandemic, we began receiving reports of patients who had survived COVID and maybe even had a relatively mild disease ... and as the summer of 2020 moved into the fall, it became apparent that there was a subset of patients who for all the world appeared to meet ME/CFS clinical criteria,” he said.
Using bicycle exercise tests on long COVID patients with catheters placed in their veins, Dr. Systrom and associates have shown a lack of exercise capacity that isn’t caused by heart or lung disease but instead is related to abnormal nerves and blood vessels, just as they’d shown previously in ME/CFS patient.
Avindra Nath, MD, senior investigator and clinical director of intramural research at the National Institute of Neurological Disorders and Stroke, Bethesda, Md., was doing a deep-dive scientific study on ME/CFS when the COVID-19 pandemic hit. Since then, he›s begun another study using the same protocol and sophisticated laboratory measurement to evaluate people with long COVID.
“As terrible as [long COVID] is, it’s kind of a blessing in disguise for ME/CFS because there’s just so much overlap between the two and they could very well be in many ways one in the same thing. The problem with studying ME/CFS is oftentimes you didn’t know what the trigger was. You see patients many years later, then try to backtrack and find out what happened,” said Dr. Nath, a neuroimmunologist.
With long COVID, on the other hand, “we know when they got infected and when their symptoms actually started, so it becomes much more uniform. ... It gives us an opportunity to maybe solve certain things in a much more well-defined population and try to find answers.”
Advocacy groups want to see more.
In February 2021, Solve M.E. launched the Long COVID Alliance, made up of several organizations, companies, and people with a goal to influence policy and speed up research into a range of postviral illnesses.
Solve M.E. has also pushed for inclusion of language regarding ME/CFS and related conditions into congressional bills addressing long COVID, including those that call for funding of research and clinical care.
“On the political front, we’ve really capitalized on a moment in time in which we have the spotlight,” said Emily Taylor, vice president of advocacy and engagement for Solve M.E.
“One of the hardest parts about ME/CFS is how to show that it’s real when it’s invisible. Most people agree that COVID is real and therefore if somebody gets ME/CFS after COVID, it’s real,” she said.
The advocacy groups are now pushing for non-COVID postinfection illnesses to be included in efforts aimed at helping people with long COVID, with mixed results. For example, the RECOVER Initiative, established in February 2021 with $1.5 billion in funding from Congress to the National Institutes of Health, is specifically for studying long COVID and does not fund research into other postinfection illnesses, although representatives from the ME/CFS community are advisers.
Language addressing ME/CFS and other postinfectious chronic illnesses has been included in several long COVID bills now pending in Congress, including the Care for Long COVID Act in the Senate and its companion COVID-19 Long Haulers Act in the House. “Our goal is to push for passage of a long COVID bill by the end of the year,” Ms. Taylor said.
A version of this article first appeared on WebMD.com.
Is it COVID or long COVID? Your organs may know
There’s little doubt long COVID is real. The federal government recognizes long COVID as a condition and said in two reports issued in August that one in five adult COVID-19 survivors have a health condition related to their illness.
COVID-19 can damage multiple organs in the body. Sometimes this damage leads to long COVID; sometimes other reasons are at play. Doctors are beginning to sort it out.
“COVID itself can actually cause prolonged illness, and we don’t really call that long COVID,” said Nisha Viswanathan, MD, a doctor at UCLA Health in Los Angeles. But if symptoms extend beyond 12 weeks, that puts patients in the realm of long COVID.
Symptoms can range from mild to severe and can keep people from resuming their normal lives and jobs. Sometimes they last for months, according to the U.S. Department of Health & Human Services.
Multiorgan damage
Lung scarring and other lung problems are common after COVID, said Leora Horwitz, MD, an internal medicine specialist at New York University. Even after a mild case, people can have breathing issues for months, a team at Johns Hopkins Medicine, Baltimore, said in an online briefing. One study published in the journal Radiology found damage in people a full year after a COVID-19 diagnosis.
Some people have persistent heart, kidney, liver, and nervous system problems after COVID-19. A study published in 2020 in JAMA Cardiology found 60% of people who had COVID-19 had ongoing signs of heart inflammation. Nearly a third of people hospitalized for COVID-19 get kidney damage that can become chronic, and some end up needing dialysis or a transplant, said C. John Sperati, MD, a kidney specialist at Johns Hopkins Medicine.
This might be, in part, because SARS-CoV-2, the virus that causes COVID-19, directly infects the cells in many organs.
Nicole Bhave, MD, a cardiologist at University of Michigan Health, Ann Arbor is concerned that COVID-19 appears to increase the risk of heart problems in some people.
“Some of the uptick may just be recognition bias, in that people with symptoms are seeking care,” she said. “But there’s definitely a biological basis by which COVID could tip people over into a new diagnosis of heart failure.”
Inflammation
Inflammation is probably a key part of the long-term effects of COVID-19.
Some people have a serious immune reaction to COVID-19 called a cytokine storm, said Nitra Aggarwal Gilotra, MD, a cardiologist at Johns Hopkins Medicine. This release of inflammation-causing molecules called cytokines is meant to attack the invading virus. But it can be so severe that it wreaks havoc on healthy tissues and organs and causes lasting damage – if patients even survive it.
In some people, inflammation can affect the heart, causing myocarditis. Myocarditis symptoms include chest pain, breathlessness, and heart palpitations. Though rare, it can be serious and can raise the risk of other heart problems, including heart failure, down the line.
Long COVID may also trigger an autoimmune condition, said Eline Luning Prak, MD, PhD, a pathologist at the Hospital of the University of Pennsylvania, Philadelphia. Long COVID can share many hallmark symptoms with autoimmune diseases, including fatigue, widespread pain, memory problems, and mood disorders.
Blood clots
Studies have shown the overcharged inflammatory response to COVID-19 can cause blood clots. This sometimes overwhelming clotting was an early hallmark of COVID-19 infection, and when clots restrict blood flow in the brain, lungs, kidneys, or limbs, they can cause long-term damage. Some can be deadly. Researchers in Sweden found patients were at risk of deep vein thrombosis – a blood clot usually in the leg – up to 3 months after infection and at higher risk of a blood clot in the lung, called pulmonary embolism, for as long as 3 months.
Viral reservoirs
The virus itself may also linger in a patient’s body, causing continued symptoms and, potentially, new flare-ups. Zoe Swank, PhD, of Harvard Medical School, Boston, and colleagues reported in a preprint study that they found pieces of the SARS-CoV-2 virus in the blood of most patients with long COVID symptoms they tested – some as long as a year after infection. The study has not yet been peer reviewed.
Another team found evidence of the virus in stool up to 7 months later, which suggests the virus hides out in the gut. Other early studies have found bits of viral RNA in the appendix, breast tissue, heart, eyes, and brain.
Diabetes
Diabetes is a risk factor for getting severe COVID-19, and multiple studies have shown people can get diabetes both while battling infection and afterward. One study of veterans, published in The Lancet Diabetes and Endocrinology, found COVID-19 survivors were about 40% more likely to get diabetes over the next year.
There are a few ways this might happen. Insulin-producing cells in the pancreas have SARS-CoV-2 receptors – a type of molecular doorway the coronavirus can attach to. Damage to these cells could make the body less able to produce insulin, which in turn can lead to diabetes. The virus could also disrupt the balance in the body or cause inflammation that leads to insulin resistance, which can develop into diabetes, Ziad Al-Aly, MD, of the Veterans Affairs St. Louis Health Care System, and colleagues wrote.
Nervous system issues
People who get COVID-19 are also more vulnerable to postural orthostatic tachycardia syndrome (POTS). This affects what’s known as the autonomic nervous system, which regulates blood circulation, and includes those things that happen in your body without your having to think about them, like breathing, heartbeat, and digestion. POTS can cause common long COVID neurologic symptoms, including headaches, fatigue, brain fog, insomnia, and problems thinking and concentrating. “This was a known condition prior to COVID, but it was incredibly rare,” said Dr. Viswanathan. “After COVID, I’ve seen it with increasing frequency.”
Long-term outlook
Lasting issues after COVID-19 are much more likely after a moderate or severe infection. Still, plenty of people are battling them even after a mild illness. “As for why, that’s the billion-dollar question,” said Dr. Horwitz. “It’s well known that viral infections can cause long-term dysregulation. Why that is, we really just don’t know.”
Whether it’s virus hiding out in the body, long-term organ damage, or an autoimmune reaction likely differs from person to person. “I’m believing, increasingly, that it’s a combination of all of these, just based on how different patients are responding to different medications,” said Dr. Viswanathan. “One patient will respond to something beautifully, and another patient won’t at all.”
But it’s clear a significant number of people are facing long-term health struggles because of COVID-19, which has infected at least 580 million people globally and 92 million – likely many more – in the United States, according to Johns Hopkins University.
Even a small increased risk of conditions like heart disease or diabetes translates to a huge number of people, Dr. Horwitz said. “If even 1% of people getting COVID have long-term symptoms, that’s a major public health crisis, because that’s 1% of pretty much everybody in the country.”
A version of this article first appeared on WebMD.com.
There’s little doubt long COVID is real. The federal government recognizes long COVID as a condition and said in two reports issued in August that one in five adult COVID-19 survivors have a health condition related to their illness.
COVID-19 can damage multiple organs in the body. Sometimes this damage leads to long COVID; sometimes other reasons are at play. Doctors are beginning to sort it out.
“COVID itself can actually cause prolonged illness, and we don’t really call that long COVID,” said Nisha Viswanathan, MD, a doctor at UCLA Health in Los Angeles. But if symptoms extend beyond 12 weeks, that puts patients in the realm of long COVID.
Symptoms can range from mild to severe and can keep people from resuming their normal lives and jobs. Sometimes they last for months, according to the U.S. Department of Health & Human Services.
Multiorgan damage
Lung scarring and other lung problems are common after COVID, said Leora Horwitz, MD, an internal medicine specialist at New York University. Even after a mild case, people can have breathing issues for months, a team at Johns Hopkins Medicine, Baltimore, said in an online briefing. One study published in the journal Radiology found damage in people a full year after a COVID-19 diagnosis.
Some people have persistent heart, kidney, liver, and nervous system problems after COVID-19. A study published in 2020 in JAMA Cardiology found 60% of people who had COVID-19 had ongoing signs of heart inflammation. Nearly a third of people hospitalized for COVID-19 get kidney damage that can become chronic, and some end up needing dialysis or a transplant, said C. John Sperati, MD, a kidney specialist at Johns Hopkins Medicine.
This might be, in part, because SARS-CoV-2, the virus that causes COVID-19, directly infects the cells in many organs.
Nicole Bhave, MD, a cardiologist at University of Michigan Health, Ann Arbor is concerned that COVID-19 appears to increase the risk of heart problems in some people.
“Some of the uptick may just be recognition bias, in that people with symptoms are seeking care,” she said. “But there’s definitely a biological basis by which COVID could tip people over into a new diagnosis of heart failure.”
Inflammation
Inflammation is probably a key part of the long-term effects of COVID-19.
Some people have a serious immune reaction to COVID-19 called a cytokine storm, said Nitra Aggarwal Gilotra, MD, a cardiologist at Johns Hopkins Medicine. This release of inflammation-causing molecules called cytokines is meant to attack the invading virus. But it can be so severe that it wreaks havoc on healthy tissues and organs and causes lasting damage – if patients even survive it.
In some people, inflammation can affect the heart, causing myocarditis. Myocarditis symptoms include chest pain, breathlessness, and heart palpitations. Though rare, it can be serious and can raise the risk of other heart problems, including heart failure, down the line.
Long COVID may also trigger an autoimmune condition, said Eline Luning Prak, MD, PhD, a pathologist at the Hospital of the University of Pennsylvania, Philadelphia. Long COVID can share many hallmark symptoms with autoimmune diseases, including fatigue, widespread pain, memory problems, and mood disorders.
Blood clots
Studies have shown the overcharged inflammatory response to COVID-19 can cause blood clots. This sometimes overwhelming clotting was an early hallmark of COVID-19 infection, and when clots restrict blood flow in the brain, lungs, kidneys, or limbs, they can cause long-term damage. Some can be deadly. Researchers in Sweden found patients were at risk of deep vein thrombosis – a blood clot usually in the leg – up to 3 months after infection and at higher risk of a blood clot in the lung, called pulmonary embolism, for as long as 3 months.
Viral reservoirs
The virus itself may also linger in a patient’s body, causing continued symptoms and, potentially, new flare-ups. Zoe Swank, PhD, of Harvard Medical School, Boston, and colleagues reported in a preprint study that they found pieces of the SARS-CoV-2 virus in the blood of most patients with long COVID symptoms they tested – some as long as a year after infection. The study has not yet been peer reviewed.
Another team found evidence of the virus in stool up to 7 months later, which suggests the virus hides out in the gut. Other early studies have found bits of viral RNA in the appendix, breast tissue, heart, eyes, and brain.
Diabetes
Diabetes is a risk factor for getting severe COVID-19, and multiple studies have shown people can get diabetes both while battling infection and afterward. One study of veterans, published in The Lancet Diabetes and Endocrinology, found COVID-19 survivors were about 40% more likely to get diabetes over the next year.
There are a few ways this might happen. Insulin-producing cells in the pancreas have SARS-CoV-2 receptors – a type of molecular doorway the coronavirus can attach to. Damage to these cells could make the body less able to produce insulin, which in turn can lead to diabetes. The virus could also disrupt the balance in the body or cause inflammation that leads to insulin resistance, which can develop into diabetes, Ziad Al-Aly, MD, of the Veterans Affairs St. Louis Health Care System, and colleagues wrote.
Nervous system issues
People who get COVID-19 are also more vulnerable to postural orthostatic tachycardia syndrome (POTS). This affects what’s known as the autonomic nervous system, which regulates blood circulation, and includes those things that happen in your body without your having to think about them, like breathing, heartbeat, and digestion. POTS can cause common long COVID neurologic symptoms, including headaches, fatigue, brain fog, insomnia, and problems thinking and concentrating. “This was a known condition prior to COVID, but it was incredibly rare,” said Dr. Viswanathan. “After COVID, I’ve seen it with increasing frequency.”
Long-term outlook
Lasting issues after COVID-19 are much more likely after a moderate or severe infection. Still, plenty of people are battling them even after a mild illness. “As for why, that’s the billion-dollar question,” said Dr. Horwitz. “It’s well known that viral infections can cause long-term dysregulation. Why that is, we really just don’t know.”
Whether it’s virus hiding out in the body, long-term organ damage, or an autoimmune reaction likely differs from person to person. “I’m believing, increasingly, that it’s a combination of all of these, just based on how different patients are responding to different medications,” said Dr. Viswanathan. “One patient will respond to something beautifully, and another patient won’t at all.”
But it’s clear a significant number of people are facing long-term health struggles because of COVID-19, which has infected at least 580 million people globally and 92 million – likely many more – in the United States, according to Johns Hopkins University.
Even a small increased risk of conditions like heart disease or diabetes translates to a huge number of people, Dr. Horwitz said. “If even 1% of people getting COVID have long-term symptoms, that’s a major public health crisis, because that’s 1% of pretty much everybody in the country.”
A version of this article first appeared on WebMD.com.
There’s little doubt long COVID is real. The federal government recognizes long COVID as a condition and said in two reports issued in August that one in five adult COVID-19 survivors have a health condition related to their illness.
COVID-19 can damage multiple organs in the body. Sometimes this damage leads to long COVID; sometimes other reasons are at play. Doctors are beginning to sort it out.
“COVID itself can actually cause prolonged illness, and we don’t really call that long COVID,” said Nisha Viswanathan, MD, a doctor at UCLA Health in Los Angeles. But if symptoms extend beyond 12 weeks, that puts patients in the realm of long COVID.
Symptoms can range from mild to severe and can keep people from resuming their normal lives and jobs. Sometimes they last for months, according to the U.S. Department of Health & Human Services.
Multiorgan damage
Lung scarring and other lung problems are common after COVID, said Leora Horwitz, MD, an internal medicine specialist at New York University. Even after a mild case, people can have breathing issues for months, a team at Johns Hopkins Medicine, Baltimore, said in an online briefing. One study published in the journal Radiology found damage in people a full year after a COVID-19 diagnosis.
Some people have persistent heart, kidney, liver, and nervous system problems after COVID-19. A study published in 2020 in JAMA Cardiology found 60% of people who had COVID-19 had ongoing signs of heart inflammation. Nearly a third of people hospitalized for COVID-19 get kidney damage that can become chronic, and some end up needing dialysis or a transplant, said C. John Sperati, MD, a kidney specialist at Johns Hopkins Medicine.
This might be, in part, because SARS-CoV-2, the virus that causes COVID-19, directly infects the cells in many organs.
Nicole Bhave, MD, a cardiologist at University of Michigan Health, Ann Arbor is concerned that COVID-19 appears to increase the risk of heart problems in some people.
“Some of the uptick may just be recognition bias, in that people with symptoms are seeking care,” she said. “But there’s definitely a biological basis by which COVID could tip people over into a new diagnosis of heart failure.”
Inflammation
Inflammation is probably a key part of the long-term effects of COVID-19.
Some people have a serious immune reaction to COVID-19 called a cytokine storm, said Nitra Aggarwal Gilotra, MD, a cardiologist at Johns Hopkins Medicine. This release of inflammation-causing molecules called cytokines is meant to attack the invading virus. But it can be so severe that it wreaks havoc on healthy tissues and organs and causes lasting damage – if patients even survive it.
In some people, inflammation can affect the heart, causing myocarditis. Myocarditis symptoms include chest pain, breathlessness, and heart palpitations. Though rare, it can be serious and can raise the risk of other heart problems, including heart failure, down the line.
Long COVID may also trigger an autoimmune condition, said Eline Luning Prak, MD, PhD, a pathologist at the Hospital of the University of Pennsylvania, Philadelphia. Long COVID can share many hallmark symptoms with autoimmune diseases, including fatigue, widespread pain, memory problems, and mood disorders.
Blood clots
Studies have shown the overcharged inflammatory response to COVID-19 can cause blood clots. This sometimes overwhelming clotting was an early hallmark of COVID-19 infection, and when clots restrict blood flow in the brain, lungs, kidneys, or limbs, they can cause long-term damage. Some can be deadly. Researchers in Sweden found patients were at risk of deep vein thrombosis – a blood clot usually in the leg – up to 3 months after infection and at higher risk of a blood clot in the lung, called pulmonary embolism, for as long as 3 months.
Viral reservoirs
The virus itself may also linger in a patient’s body, causing continued symptoms and, potentially, new flare-ups. Zoe Swank, PhD, of Harvard Medical School, Boston, and colleagues reported in a preprint study that they found pieces of the SARS-CoV-2 virus in the blood of most patients with long COVID symptoms they tested – some as long as a year after infection. The study has not yet been peer reviewed.
Another team found evidence of the virus in stool up to 7 months later, which suggests the virus hides out in the gut. Other early studies have found bits of viral RNA in the appendix, breast tissue, heart, eyes, and brain.
Diabetes
Diabetes is a risk factor for getting severe COVID-19, and multiple studies have shown people can get diabetes both while battling infection and afterward. One study of veterans, published in The Lancet Diabetes and Endocrinology, found COVID-19 survivors were about 40% more likely to get diabetes over the next year.
There are a few ways this might happen. Insulin-producing cells in the pancreas have SARS-CoV-2 receptors – a type of molecular doorway the coronavirus can attach to. Damage to these cells could make the body less able to produce insulin, which in turn can lead to diabetes. The virus could also disrupt the balance in the body or cause inflammation that leads to insulin resistance, which can develop into diabetes, Ziad Al-Aly, MD, of the Veterans Affairs St. Louis Health Care System, and colleagues wrote.
Nervous system issues
People who get COVID-19 are also more vulnerable to postural orthostatic tachycardia syndrome (POTS). This affects what’s known as the autonomic nervous system, which regulates blood circulation, and includes those things that happen in your body without your having to think about them, like breathing, heartbeat, and digestion. POTS can cause common long COVID neurologic symptoms, including headaches, fatigue, brain fog, insomnia, and problems thinking and concentrating. “This was a known condition prior to COVID, but it was incredibly rare,” said Dr. Viswanathan. “After COVID, I’ve seen it with increasing frequency.”
Long-term outlook
Lasting issues after COVID-19 are much more likely after a moderate or severe infection. Still, plenty of people are battling them even after a mild illness. “As for why, that’s the billion-dollar question,” said Dr. Horwitz. “It’s well known that viral infections can cause long-term dysregulation. Why that is, we really just don’t know.”
Whether it’s virus hiding out in the body, long-term organ damage, or an autoimmune reaction likely differs from person to person. “I’m believing, increasingly, that it’s a combination of all of these, just based on how different patients are responding to different medications,” said Dr. Viswanathan. “One patient will respond to something beautifully, and another patient won’t at all.”
But it’s clear a significant number of people are facing long-term health struggles because of COVID-19, which has infected at least 580 million people globally and 92 million – likely many more – in the United States, according to Johns Hopkins University.
Even a small increased risk of conditions like heart disease or diabetes translates to a huge number of people, Dr. Horwitz said. “If even 1% of people getting COVID have long-term symptoms, that’s a major public health crisis, because that’s 1% of pretty much everybody in the country.”
A version of this article first appeared on WebMD.com.
COVID-19 may trigger irritable bowel syndrome
Gastrointestinal symptoms are common with long COVID, also known as post-acute COVID-19 syndrome, according to Walter Chan, MD, MPH, and Madhusudan Grover, MBBS.
Dr. Chan, an assistant professor at Harvard Medical School, Boston, and Dr. Grover, an associate professor of medicine and physiology at Mayo Clinic, Rochester, Minn., conducted a review of the literature on COVID-19’s long-term gastrointestinal effects. Their review was published in Clinical Gastroenterology and Hepatology.
Estimates of the prevalence of gastrointestinal symptoms with COVID-19 have ranged as high as 60%, Dr. Chan and Dr. Grover report, and the symptoms may be present in patients with long COVID, a syndrome that continues 4 weeks or longer.
In one survey of 749 COVID-19 survivors, 29% reported at least one new chronic gastrointestinal symptom. The most common were heartburn, constipation, diarrhea, and abdominal pain. Of those with abdominal pain, 39% had symptoms that met Rome IV criteria for irritable bowel syndrome.
People who have gastrointestinal symptoms after their initial SARS-CoV-2 infection are more likely to have them with long COVID. Psychiatric diagnoses, hospitalization, and the loss of smell and taste are predictors of gastrointestinal symptoms.
Infectious gastroenteritis can increase the risk for disorders of gut-brain interaction, especially postinfection IBS, Dr. Chan and Dr. Grover write.
COVID-19 likely causes gastrointestinal symptoms through multiple mechanisms. It may suppress angiotensin-converting enzyme 2, which protects intestinal cells. It can alter the microbiome. It can cause or worsen weight gain and diabetes. It may disrupt the immune system and trigger an autoimmune reaction. It can cause depression and anxiety, and it can alter dietary habits.
No specific treatments for gastrointestinal symptoms associated with long COVID have emerged, so clinicians should make use of established therapies for disorders of gut-brain interaction, Dr. Chan and Dr. Grover recommend.
Beyond adequate sleep and exercise, these may include high-fiber, low FODMAP (fermentable oligosaccharides, disaccharides, monosaccharides, and polyols), gluten-free, low-carbohydrate, or elimination diets.
For diarrhea, they list loperamide, ondansetron, alosetron, eluxadoline, antispasmodics, rifaximin, and bile acid sequestrants.
For constipation, they mention fiber supplements, polyethylene glycol, linaclotide, plecanatide, lubiprostone, tenapanor, tegaserod, and prucalopride.
For modulating intestinal permeability, they recommend glutamine.
Neuromodulation may be achieved with tricyclic antidepressants, selective serotonin reuptake inhibitors, serotonin norepinephrine reuptake inhibitors, azaperones, and delta ligands, they write.
For psychological therapy, they recommend cognitive-behavioral therapy and gut-directed hypnotherapy.
A handful of studies have suggested benefits from Lactiplantibacillus plantarum and Pediococcus acidilactici as probiotic therapies. Additionally, one study showed positive results with a high-fiber formula, perhaps by nourishing short-chain fatty acid-producing bacteria, Dr. Chan and Dr. Grover write.
Dr. Chan reported financial relationships with Ironwood, Takeda, and Phathom Pharmaceuticals. Dr. Grover reported financial relationships with Takeda, Donga, Alexza Pharmaceuticals, and Alfasigma.
A version of this article first appeared on Medscape.com.
Gastrointestinal symptoms are common with long COVID, also known as post-acute COVID-19 syndrome, according to Walter Chan, MD, MPH, and Madhusudan Grover, MBBS.
Dr. Chan, an assistant professor at Harvard Medical School, Boston, and Dr. Grover, an associate professor of medicine and physiology at Mayo Clinic, Rochester, Minn., conducted a review of the literature on COVID-19’s long-term gastrointestinal effects. Their review was published in Clinical Gastroenterology and Hepatology.
Estimates of the prevalence of gastrointestinal symptoms with COVID-19 have ranged as high as 60%, Dr. Chan and Dr. Grover report, and the symptoms may be present in patients with long COVID, a syndrome that continues 4 weeks or longer.
In one survey of 749 COVID-19 survivors, 29% reported at least one new chronic gastrointestinal symptom. The most common were heartburn, constipation, diarrhea, and abdominal pain. Of those with abdominal pain, 39% had symptoms that met Rome IV criteria for irritable bowel syndrome.
People who have gastrointestinal symptoms after their initial SARS-CoV-2 infection are more likely to have them with long COVID. Psychiatric diagnoses, hospitalization, and the loss of smell and taste are predictors of gastrointestinal symptoms.
Infectious gastroenteritis can increase the risk for disorders of gut-brain interaction, especially postinfection IBS, Dr. Chan and Dr. Grover write.
COVID-19 likely causes gastrointestinal symptoms through multiple mechanisms. It may suppress angiotensin-converting enzyme 2, which protects intestinal cells. It can alter the microbiome. It can cause or worsen weight gain and diabetes. It may disrupt the immune system and trigger an autoimmune reaction. It can cause depression and anxiety, and it can alter dietary habits.
No specific treatments for gastrointestinal symptoms associated with long COVID have emerged, so clinicians should make use of established therapies for disorders of gut-brain interaction, Dr. Chan and Dr. Grover recommend.
Beyond adequate sleep and exercise, these may include high-fiber, low FODMAP (fermentable oligosaccharides, disaccharides, monosaccharides, and polyols), gluten-free, low-carbohydrate, or elimination diets.
For diarrhea, they list loperamide, ondansetron, alosetron, eluxadoline, antispasmodics, rifaximin, and bile acid sequestrants.
For constipation, they mention fiber supplements, polyethylene glycol, linaclotide, plecanatide, lubiprostone, tenapanor, tegaserod, and prucalopride.
For modulating intestinal permeability, they recommend glutamine.
Neuromodulation may be achieved with tricyclic antidepressants, selective serotonin reuptake inhibitors, serotonin norepinephrine reuptake inhibitors, azaperones, and delta ligands, they write.
For psychological therapy, they recommend cognitive-behavioral therapy and gut-directed hypnotherapy.
A handful of studies have suggested benefits from Lactiplantibacillus plantarum and Pediococcus acidilactici as probiotic therapies. Additionally, one study showed positive results with a high-fiber formula, perhaps by nourishing short-chain fatty acid-producing bacteria, Dr. Chan and Dr. Grover write.
Dr. Chan reported financial relationships with Ironwood, Takeda, and Phathom Pharmaceuticals. Dr. Grover reported financial relationships with Takeda, Donga, Alexza Pharmaceuticals, and Alfasigma.
A version of this article first appeared on Medscape.com.
Gastrointestinal symptoms are common with long COVID, also known as post-acute COVID-19 syndrome, according to Walter Chan, MD, MPH, and Madhusudan Grover, MBBS.
Dr. Chan, an assistant professor at Harvard Medical School, Boston, and Dr. Grover, an associate professor of medicine and physiology at Mayo Clinic, Rochester, Minn., conducted a review of the literature on COVID-19’s long-term gastrointestinal effects. Their review was published in Clinical Gastroenterology and Hepatology.
Estimates of the prevalence of gastrointestinal symptoms with COVID-19 have ranged as high as 60%, Dr. Chan and Dr. Grover report, and the symptoms may be present in patients with long COVID, a syndrome that continues 4 weeks or longer.
In one survey of 749 COVID-19 survivors, 29% reported at least one new chronic gastrointestinal symptom. The most common were heartburn, constipation, diarrhea, and abdominal pain. Of those with abdominal pain, 39% had symptoms that met Rome IV criteria for irritable bowel syndrome.
People who have gastrointestinal symptoms after their initial SARS-CoV-2 infection are more likely to have them with long COVID. Psychiatric diagnoses, hospitalization, and the loss of smell and taste are predictors of gastrointestinal symptoms.
Infectious gastroenteritis can increase the risk for disorders of gut-brain interaction, especially postinfection IBS, Dr. Chan and Dr. Grover write.
COVID-19 likely causes gastrointestinal symptoms through multiple mechanisms. It may suppress angiotensin-converting enzyme 2, which protects intestinal cells. It can alter the microbiome. It can cause or worsen weight gain and diabetes. It may disrupt the immune system and trigger an autoimmune reaction. It can cause depression and anxiety, and it can alter dietary habits.
No specific treatments for gastrointestinal symptoms associated with long COVID have emerged, so clinicians should make use of established therapies for disorders of gut-brain interaction, Dr. Chan and Dr. Grover recommend.
Beyond adequate sleep and exercise, these may include high-fiber, low FODMAP (fermentable oligosaccharides, disaccharides, monosaccharides, and polyols), gluten-free, low-carbohydrate, or elimination diets.
For diarrhea, they list loperamide, ondansetron, alosetron, eluxadoline, antispasmodics, rifaximin, and bile acid sequestrants.
For constipation, they mention fiber supplements, polyethylene glycol, linaclotide, plecanatide, lubiprostone, tenapanor, tegaserod, and prucalopride.
For modulating intestinal permeability, they recommend glutamine.
Neuromodulation may be achieved with tricyclic antidepressants, selective serotonin reuptake inhibitors, serotonin norepinephrine reuptake inhibitors, azaperones, and delta ligands, they write.
For psychological therapy, they recommend cognitive-behavioral therapy and gut-directed hypnotherapy.
A handful of studies have suggested benefits from Lactiplantibacillus plantarum and Pediococcus acidilactici as probiotic therapies. Additionally, one study showed positive results with a high-fiber formula, perhaps by nourishing short-chain fatty acid-producing bacteria, Dr. Chan and Dr. Grover write.
Dr. Chan reported financial relationships with Ironwood, Takeda, and Phathom Pharmaceuticals. Dr. Grover reported financial relationships with Takeda, Donga, Alexza Pharmaceuticals, and Alfasigma.
A version of this article first appeared on Medscape.com.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Long COVID case study: persistent hormone deficiencies
A case study of a 65-year-old man in Japan with long COVID describes how he recovered from certain impaired hormone deficiencies that persisted for more than a year.
Days after the patient recovered from respiratory failure and came off a ventilator, he had a sudden drop in blood pressure, which responded to hydrocortisone.
The patient was found to have low levels of growth hormone and adrenocorticotropic hormone (ACTH), hypopituitarism, that persisted for more than a year. He also had low levels of testosterone that remained low at 15 months (the study end).
“An important finding in the present case is the eventual recovery from hypopituitarism over time but not from hypogonadism,” the researchers write in their study published in Endocrine Journal.
, which was confirmed using an insulin tolerance test, Kai Yoshimura, Kakogawa Medical Center, Japan, and colleagues report.
The findings show that “pituitary insufficiency should be considered in patients with prolonged symptoms of COVID-19,” they report, since it can be treated with hormone supplements that markedly improve symptoms and quality of life.
“It might be worthwhile to screen for endocrine dysfunction in patients with such persistent symptoms after their recovery from the acute disease,” the researchers conclude.
Case study timeline
The patient in this study was healthy without obesity, previous endocrine disease, or steroid use. He was admitted to hospital because he had dyspnea and fever for 8 days and a reverse transcription-polymerase chain reaction (RT-PCR) test that was positive for COVID-19.
He received ciclesonide 200 mcg/day for 2 days. Then he was put on a ventilator and the drug was discontinued and “favipiravir, ritonavir, and lopinavir, a standard regimen during the early phase of the COVID-19 pandemic, were initiated;” the researchers explain.
On day 25 of his hospital stay the patient had recovered from respiratory failure and was extubated.
On day 31, he had a negative PCR test for COVID-19.
On day 36, the patient’s blood pressure suddenly dropped from 120/80 mmHg to 80/50 mmHg. His plasma ACTH and serum cortisol levels were low, suggesting secondary adrenal insufficiency. The low blood pressure responded to hydrocortisone 100 mg, which was gradually tapered.
At day 96, the patient was discharged from hospital with a dose of 15 mg/day hydrocortisone.
At 3 months after discharge, an insulin tolerance test revealed that the patient’s ACTH and cortisol responses were blunted, suggestive of adrenal insufficiency. The patient also had moderate growth hormone deficiency and symptoms of hypogonadism.
At 6 months after discharge, the patient started testosterone therapy because his dysspermatism had worsened.
At 12 months after discharge, a repeat insulin tolerance test showed that both ACTH and cortisol responses were low but improved. The patient was no longer deficient in growth hormone.
At 15 months after discharge, early morning levels of ACTH and cortisol were now in the normal range. The patient discontinued testosterone treatment, but the symptoms returned, so he resumed it.
Long COVID symptoms, possible biological mechanism
The present case shows how certain COVID-19–associated conditions develop after the onset of, or the recovery from, respiratory disorders, the authors note.
Symptoms of long COVID-19 include fatigue, weakness, hair loss, diarrhea, arthralgia, and depression, and these symptoms are associated with pituitary insufficiency, especially secondary adrenocortical insufficiency.
In addition, an estimated 25% of sexually active men who recover from COVID have semen disorders such as azoospermia and oligospermia.
The underlying mechanism by which COVID-19 might trigger pituitary insufficiency is unknown, but other viral infections such as influenza-A and herpes simplex are also associated with transient hypopituitarism. An exaggerated immune response triggered by SARS-CoV-2 may explain the dysfunction of multiple endocrine organs, the researchers write.
The researchers have declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
A case study of a 65-year-old man in Japan with long COVID describes how he recovered from certain impaired hormone deficiencies that persisted for more than a year.
Days after the patient recovered from respiratory failure and came off a ventilator, he had a sudden drop in blood pressure, which responded to hydrocortisone.
The patient was found to have low levels of growth hormone and adrenocorticotropic hormone (ACTH), hypopituitarism, that persisted for more than a year. He also had low levels of testosterone that remained low at 15 months (the study end).
“An important finding in the present case is the eventual recovery from hypopituitarism over time but not from hypogonadism,” the researchers write in their study published in Endocrine Journal.
, which was confirmed using an insulin tolerance test, Kai Yoshimura, Kakogawa Medical Center, Japan, and colleagues report.
The findings show that “pituitary insufficiency should be considered in patients with prolonged symptoms of COVID-19,” they report, since it can be treated with hormone supplements that markedly improve symptoms and quality of life.
“It might be worthwhile to screen for endocrine dysfunction in patients with such persistent symptoms after their recovery from the acute disease,” the researchers conclude.
Case study timeline
The patient in this study was healthy without obesity, previous endocrine disease, or steroid use. He was admitted to hospital because he had dyspnea and fever for 8 days and a reverse transcription-polymerase chain reaction (RT-PCR) test that was positive for COVID-19.
He received ciclesonide 200 mcg/day for 2 days. Then he was put on a ventilator and the drug was discontinued and “favipiravir, ritonavir, and lopinavir, a standard regimen during the early phase of the COVID-19 pandemic, were initiated;” the researchers explain.
On day 25 of his hospital stay the patient had recovered from respiratory failure and was extubated.
On day 31, he had a negative PCR test for COVID-19.
On day 36, the patient’s blood pressure suddenly dropped from 120/80 mmHg to 80/50 mmHg. His plasma ACTH and serum cortisol levels were low, suggesting secondary adrenal insufficiency. The low blood pressure responded to hydrocortisone 100 mg, which was gradually tapered.
At day 96, the patient was discharged from hospital with a dose of 15 mg/day hydrocortisone.
At 3 months after discharge, an insulin tolerance test revealed that the patient’s ACTH and cortisol responses were blunted, suggestive of adrenal insufficiency. The patient also had moderate growth hormone deficiency and symptoms of hypogonadism.
At 6 months after discharge, the patient started testosterone therapy because his dysspermatism had worsened.
At 12 months after discharge, a repeat insulin tolerance test showed that both ACTH and cortisol responses were low but improved. The patient was no longer deficient in growth hormone.
At 15 months after discharge, early morning levels of ACTH and cortisol were now in the normal range. The patient discontinued testosterone treatment, but the symptoms returned, so he resumed it.
Long COVID symptoms, possible biological mechanism
The present case shows how certain COVID-19–associated conditions develop after the onset of, or the recovery from, respiratory disorders, the authors note.
Symptoms of long COVID-19 include fatigue, weakness, hair loss, diarrhea, arthralgia, and depression, and these symptoms are associated with pituitary insufficiency, especially secondary adrenocortical insufficiency.
In addition, an estimated 25% of sexually active men who recover from COVID have semen disorders such as azoospermia and oligospermia.
The underlying mechanism by which COVID-19 might trigger pituitary insufficiency is unknown, but other viral infections such as influenza-A and herpes simplex are also associated with transient hypopituitarism. An exaggerated immune response triggered by SARS-CoV-2 may explain the dysfunction of multiple endocrine organs, the researchers write.
The researchers have declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
A case study of a 65-year-old man in Japan with long COVID describes how he recovered from certain impaired hormone deficiencies that persisted for more than a year.
Days after the patient recovered from respiratory failure and came off a ventilator, he had a sudden drop in blood pressure, which responded to hydrocortisone.
The patient was found to have low levels of growth hormone and adrenocorticotropic hormone (ACTH), hypopituitarism, that persisted for more than a year. He also had low levels of testosterone that remained low at 15 months (the study end).
“An important finding in the present case is the eventual recovery from hypopituitarism over time but not from hypogonadism,” the researchers write in their study published in Endocrine Journal.
, which was confirmed using an insulin tolerance test, Kai Yoshimura, Kakogawa Medical Center, Japan, and colleagues report.
The findings show that “pituitary insufficiency should be considered in patients with prolonged symptoms of COVID-19,” they report, since it can be treated with hormone supplements that markedly improve symptoms and quality of life.
“It might be worthwhile to screen for endocrine dysfunction in patients with such persistent symptoms after their recovery from the acute disease,” the researchers conclude.
Case study timeline
The patient in this study was healthy without obesity, previous endocrine disease, or steroid use. He was admitted to hospital because he had dyspnea and fever for 8 days and a reverse transcription-polymerase chain reaction (RT-PCR) test that was positive for COVID-19.
He received ciclesonide 200 mcg/day for 2 days. Then he was put on a ventilator and the drug was discontinued and “favipiravir, ritonavir, and lopinavir, a standard regimen during the early phase of the COVID-19 pandemic, were initiated;” the researchers explain.
On day 25 of his hospital stay the patient had recovered from respiratory failure and was extubated.
On day 31, he had a negative PCR test for COVID-19.
On day 36, the patient’s blood pressure suddenly dropped from 120/80 mmHg to 80/50 mmHg. His plasma ACTH and serum cortisol levels were low, suggesting secondary adrenal insufficiency. The low blood pressure responded to hydrocortisone 100 mg, which was gradually tapered.
At day 96, the patient was discharged from hospital with a dose of 15 mg/day hydrocortisone.
At 3 months after discharge, an insulin tolerance test revealed that the patient’s ACTH and cortisol responses were blunted, suggestive of adrenal insufficiency. The patient also had moderate growth hormone deficiency and symptoms of hypogonadism.
At 6 months after discharge, the patient started testosterone therapy because his dysspermatism had worsened.
At 12 months after discharge, a repeat insulin tolerance test showed that both ACTH and cortisol responses were low but improved. The patient was no longer deficient in growth hormone.
At 15 months after discharge, early morning levels of ACTH and cortisol were now in the normal range. The patient discontinued testosterone treatment, but the symptoms returned, so he resumed it.
Long COVID symptoms, possible biological mechanism
The present case shows how certain COVID-19–associated conditions develop after the onset of, or the recovery from, respiratory disorders, the authors note.
Symptoms of long COVID-19 include fatigue, weakness, hair loss, diarrhea, arthralgia, and depression, and these symptoms are associated with pituitary insufficiency, especially secondary adrenocortical insufficiency.
In addition, an estimated 25% of sexually active men who recover from COVID have semen disorders such as azoospermia and oligospermia.
The underlying mechanism by which COVID-19 might trigger pituitary insufficiency is unknown, but other viral infections such as influenza-A and herpes simplex are also associated with transient hypopituitarism. An exaggerated immune response triggered by SARS-CoV-2 may explain the dysfunction of multiple endocrine organs, the researchers write.
The researchers have declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
Diagnosing children with long COVID can be tricky: Experts
When Spencer Siedlecki got COVID-19 in March 2021, he was sick for weeks with extreme fatigue, fevers, a sore throat, bad headaches, nausea, and eventually, pneumonia.
That was scary enough for the then-13-year-old and his parents, who live in Ohio. More than a year later, Spencer still had many of the symptoms and, more alarming, the once-healthy teen had postural orthostatic tachycardia syndrome, a condition that has caused dizziness, a racing heart when he stands, and fainting. Spencer missed most of the last few months of eighth grade because of long COVID.
“He gets sick very easily,” said his mother, Melissa Siedlecki, who works in technology sales. “The common cold that he would shake off in a few days takes weeks for him to feel better.”
The transformation from regular teen life to someone with a chronic illness “sucked,” said Spencer, who will turn 15 in August. “I felt like I was never going to get better.” Fortunately, after some therapy at a specialized clinic, Spencer is back to playing baseball and golf.
Spencer’s journey to better health was difficult; his regular pediatrician told the family at first that there were no treatments to help him – a reaction that is not uncommon. “I still get a lot of parents who heard of me through the grapevine,” said Amy Edwards, MD, director of the pediatric COVID clinic at University Hospitals Rainbow Babies & Children’s and an assistant professor of pediatrics at Case Western Reserve University, both in Cleveland. “The pediatricians either are unsure of what is wrong, or worse, tell children ‘there is nothing wrong with you. Stop faking it.’ ” Dr. Edwards treated Spencer after his mother found the clinic through an internet search.
Alexandra Yonts, MD, a pediatric infectious diseases doctor and director of the post-COVID program clinic at Children’s National Medical Center in Washington, has seen this too. she said.
But those who do get attention tend to be White and affluent, something Dr. Yonts said “doesn’t jibe with the epidemiologic data of who COVID has affected the most.” Black, Latino, and American Indian and Alaska Native children are more likely to be infected with COVID than White children, and have higher rates of hospitalization and death than White children.
It’s not clear whether these children have a particular risk factor, or if they are just the ones who have the resources to get to the clinics. But Dr. Yonts and Dr. Edwards believe many children are not getting the help they need. High-performing kids are coming in “because they are the ones whose symptoms are most obvious,” said Dr. Edwards. “I think there are kids out there who are getting missed because they’re already struggling because of socioeconomic reasons.”
Spencer is one of 14 million children who have tested positive for SARS-CoV-2 since the start of the pandemic. Many pediatricians are still grappling with how to address cases like Spencer’s. The American Academy of Pediatrics has issued only brief guidance on long COVID in children, in part because there have been so few studies to use as a basis for guidance.
The federal government is aiming to change that with a newly launched National Research Action Plan on Long COVID that includes speeding up research on how the condition affects children and youths, including their ability to learn and thrive.
A CDC study found children with COVID were significantly more likely to have smell and taste disturbances, circulatory system problems, fatigue and malaise, and pain. Those who had been infected had higher rates of acute blockage of a lung artery, myocarditis and weakening of the heart, kidney failure, and type 1 diabetes.
Difficult to diagnose
Even with increased media attention and more published studies on pediatric long COVID, it’s still hard for a busy primary care doctor “to sort through what could just be a cold or what could be a series of colds and trying to look at the bigger picture of what’s been going on in a 1- to 3-month period with a kid,” Dr. Yonts said.
Most children with potential or definite long COVID are still being seen by individual pediatricians, not in a specialized clinic with easy access to an army of specialists. It’s not clear how many of those pediatric clinics exist. Survivor Corps, an advocacy group for people with long COVID, has posted a map of locations providing care, but few are specialized or focus on pediatric long COVID.
Long COVID is different from multisystem inflammatory syndrome in children (MIS-C), which occurs within a month or so of infection, triggers high fevers and severe symptoms in the gut, and often results in hospitalization. MIS-C “is not subtle,” said Dr. Edwards.
The long COVID clinic doctors said most of their patients were not very sick at first. “Anecdotally, of the 83 kids that we’ve seen, most have had mild, very mild, or even asymptomatic infections initially,” and then went on to have long COVID, said Dr. Yonts.
“We see it even in children who have very mild disease or even are asymptomatic,” agreed Allison Eckard, MD, director of pediatric infectious diseases at the Medical University of South Carolina, Charleston.
Fatigue, mood problems
Dr. Yonts said 90% of her patients have fatigue, and many also have severe symptoms in their gut. Those and other long COVID symptoms will be looked at more closely in a 3-year study the Children’s National Medical Center is doing along with the National Institute of Allergy and Infectious Diseases.
There are no treatments for long COVID itself.
“Management is probably more the correct term for what we do in our clinic at this point,” said Dr. Yonts. That means dealing with fatigue and managing headache and digestive symptoms with medications or coping strategies. Guidelines from the American Academy of Physical Medicine and Rehabilitation help inform how to help kids safely resume exercise.
At the Children’s National Medical Center clinic, children will typically meet with a team of specialists including infectious diseases doctors on the same day, said Dr. Yonts. Psychologists help children with coping skills. Dr. Yonts is careful not to imply that long COVID is a psychological illness. Parents “will just shut down, because for so long, they’ve been told this is all a mental thing.”
In about a third of children, symptoms get better on their own, and most kids get better over time. But many still struggle. “We don’t talk about cure, because we don’t know what cure looks like,” said Dr. Edwards.
Vaccination may be best protection
Vaccination seems to help reduce the risk of long COVID, perhaps by as much as half. But parents have been slow to vaccinate children, especially the very young. The AAP reported that, as of Aug. 3, just 5% of children under age 5, 37% of those ages 5-11, and 69% of 12- to 17-year-olds have received at least one dose of a COVID-19 vaccine.
“We have tried to really push vaccine as one of the ways to help prevent some of these long COVID syndromes,” said Dr. Eckard. But that advice is not always welcome. Dr. Eckard told the story of a mother who refused to have her autistic son vaccinated, even as she tearfully pleaded for help with his long COVID symptoms, which had also worsened his autism. The woman told Dr. Eckard: “Nothing you can say will convince me to get him vaccinated.” She thought a vaccine could make his symptoms even worse.
The best prevention is to avoid being infected in the first place.
“The more times you get COVID, the more you increase your risk of getting long COVID,” said Dr. Yonts. “The more times you roll the dice, eventually your number could come up.
A version of this article first appeared on WebMD.com.
When Spencer Siedlecki got COVID-19 in March 2021, he was sick for weeks with extreme fatigue, fevers, a sore throat, bad headaches, nausea, and eventually, pneumonia.
That was scary enough for the then-13-year-old and his parents, who live in Ohio. More than a year later, Spencer still had many of the symptoms and, more alarming, the once-healthy teen had postural orthostatic tachycardia syndrome, a condition that has caused dizziness, a racing heart when he stands, and fainting. Spencer missed most of the last few months of eighth grade because of long COVID.
“He gets sick very easily,” said his mother, Melissa Siedlecki, who works in technology sales. “The common cold that he would shake off in a few days takes weeks for him to feel better.”
The transformation from regular teen life to someone with a chronic illness “sucked,” said Spencer, who will turn 15 in August. “I felt like I was never going to get better.” Fortunately, after some therapy at a specialized clinic, Spencer is back to playing baseball and golf.
Spencer’s journey to better health was difficult; his regular pediatrician told the family at first that there were no treatments to help him – a reaction that is not uncommon. “I still get a lot of parents who heard of me through the grapevine,” said Amy Edwards, MD, director of the pediatric COVID clinic at University Hospitals Rainbow Babies & Children’s and an assistant professor of pediatrics at Case Western Reserve University, both in Cleveland. “The pediatricians either are unsure of what is wrong, or worse, tell children ‘there is nothing wrong with you. Stop faking it.’ ” Dr. Edwards treated Spencer after his mother found the clinic through an internet search.
Alexandra Yonts, MD, a pediatric infectious diseases doctor and director of the post-COVID program clinic at Children’s National Medical Center in Washington, has seen this too. she said.
But those who do get attention tend to be White and affluent, something Dr. Yonts said “doesn’t jibe with the epidemiologic data of who COVID has affected the most.” Black, Latino, and American Indian and Alaska Native children are more likely to be infected with COVID than White children, and have higher rates of hospitalization and death than White children.
It’s not clear whether these children have a particular risk factor, or if they are just the ones who have the resources to get to the clinics. But Dr. Yonts and Dr. Edwards believe many children are not getting the help they need. High-performing kids are coming in “because they are the ones whose symptoms are most obvious,” said Dr. Edwards. “I think there are kids out there who are getting missed because they’re already struggling because of socioeconomic reasons.”
Spencer is one of 14 million children who have tested positive for SARS-CoV-2 since the start of the pandemic. Many pediatricians are still grappling with how to address cases like Spencer’s. The American Academy of Pediatrics has issued only brief guidance on long COVID in children, in part because there have been so few studies to use as a basis for guidance.
The federal government is aiming to change that with a newly launched National Research Action Plan on Long COVID that includes speeding up research on how the condition affects children and youths, including their ability to learn and thrive.
A CDC study found children with COVID were significantly more likely to have smell and taste disturbances, circulatory system problems, fatigue and malaise, and pain. Those who had been infected had higher rates of acute blockage of a lung artery, myocarditis and weakening of the heart, kidney failure, and type 1 diabetes.
Difficult to diagnose
Even with increased media attention and more published studies on pediatric long COVID, it’s still hard for a busy primary care doctor “to sort through what could just be a cold or what could be a series of colds and trying to look at the bigger picture of what’s been going on in a 1- to 3-month period with a kid,” Dr. Yonts said.
Most children with potential or definite long COVID are still being seen by individual pediatricians, not in a specialized clinic with easy access to an army of specialists. It’s not clear how many of those pediatric clinics exist. Survivor Corps, an advocacy group for people with long COVID, has posted a map of locations providing care, but few are specialized or focus on pediatric long COVID.
Long COVID is different from multisystem inflammatory syndrome in children (MIS-C), which occurs within a month or so of infection, triggers high fevers and severe symptoms in the gut, and often results in hospitalization. MIS-C “is not subtle,” said Dr. Edwards.
The long COVID clinic doctors said most of their patients were not very sick at first. “Anecdotally, of the 83 kids that we’ve seen, most have had mild, very mild, or even asymptomatic infections initially,” and then went on to have long COVID, said Dr. Yonts.
“We see it even in children who have very mild disease or even are asymptomatic,” agreed Allison Eckard, MD, director of pediatric infectious diseases at the Medical University of South Carolina, Charleston.
Fatigue, mood problems
Dr. Yonts said 90% of her patients have fatigue, and many also have severe symptoms in their gut. Those and other long COVID symptoms will be looked at more closely in a 3-year study the Children’s National Medical Center is doing along with the National Institute of Allergy and Infectious Diseases.
There are no treatments for long COVID itself.
“Management is probably more the correct term for what we do in our clinic at this point,” said Dr. Yonts. That means dealing with fatigue and managing headache and digestive symptoms with medications or coping strategies. Guidelines from the American Academy of Physical Medicine and Rehabilitation help inform how to help kids safely resume exercise.
At the Children’s National Medical Center clinic, children will typically meet with a team of specialists including infectious diseases doctors on the same day, said Dr. Yonts. Psychologists help children with coping skills. Dr. Yonts is careful not to imply that long COVID is a psychological illness. Parents “will just shut down, because for so long, they’ve been told this is all a mental thing.”
In about a third of children, symptoms get better on their own, and most kids get better over time. But many still struggle. “We don’t talk about cure, because we don’t know what cure looks like,” said Dr. Edwards.
Vaccination may be best protection
Vaccination seems to help reduce the risk of long COVID, perhaps by as much as half. But parents have been slow to vaccinate children, especially the very young. The AAP reported that, as of Aug. 3, just 5% of children under age 5, 37% of those ages 5-11, and 69% of 12- to 17-year-olds have received at least one dose of a COVID-19 vaccine.
“We have tried to really push vaccine as one of the ways to help prevent some of these long COVID syndromes,” said Dr. Eckard. But that advice is not always welcome. Dr. Eckard told the story of a mother who refused to have her autistic son vaccinated, even as she tearfully pleaded for help with his long COVID symptoms, which had also worsened his autism. The woman told Dr. Eckard: “Nothing you can say will convince me to get him vaccinated.” She thought a vaccine could make his symptoms even worse.
The best prevention is to avoid being infected in the first place.
“The more times you get COVID, the more you increase your risk of getting long COVID,” said Dr. Yonts. “The more times you roll the dice, eventually your number could come up.
A version of this article first appeared on WebMD.com.
When Spencer Siedlecki got COVID-19 in March 2021, he was sick for weeks with extreme fatigue, fevers, a sore throat, bad headaches, nausea, and eventually, pneumonia.
That was scary enough for the then-13-year-old and his parents, who live in Ohio. More than a year later, Spencer still had many of the symptoms and, more alarming, the once-healthy teen had postural orthostatic tachycardia syndrome, a condition that has caused dizziness, a racing heart when he stands, and fainting. Spencer missed most of the last few months of eighth grade because of long COVID.
“He gets sick very easily,” said his mother, Melissa Siedlecki, who works in technology sales. “The common cold that he would shake off in a few days takes weeks for him to feel better.”
The transformation from regular teen life to someone with a chronic illness “sucked,” said Spencer, who will turn 15 in August. “I felt like I was never going to get better.” Fortunately, after some therapy at a specialized clinic, Spencer is back to playing baseball and golf.
Spencer’s journey to better health was difficult; his regular pediatrician told the family at first that there were no treatments to help him – a reaction that is not uncommon. “I still get a lot of parents who heard of me through the grapevine,” said Amy Edwards, MD, director of the pediatric COVID clinic at University Hospitals Rainbow Babies & Children’s and an assistant professor of pediatrics at Case Western Reserve University, both in Cleveland. “The pediatricians either are unsure of what is wrong, or worse, tell children ‘there is nothing wrong with you. Stop faking it.’ ” Dr. Edwards treated Spencer after his mother found the clinic through an internet search.
Alexandra Yonts, MD, a pediatric infectious diseases doctor and director of the post-COVID program clinic at Children’s National Medical Center in Washington, has seen this too. she said.
But those who do get attention tend to be White and affluent, something Dr. Yonts said “doesn’t jibe with the epidemiologic data of who COVID has affected the most.” Black, Latino, and American Indian and Alaska Native children are more likely to be infected with COVID than White children, and have higher rates of hospitalization and death than White children.
It’s not clear whether these children have a particular risk factor, or if they are just the ones who have the resources to get to the clinics. But Dr. Yonts and Dr. Edwards believe many children are not getting the help they need. High-performing kids are coming in “because they are the ones whose symptoms are most obvious,” said Dr. Edwards. “I think there are kids out there who are getting missed because they’re already struggling because of socioeconomic reasons.”
Spencer is one of 14 million children who have tested positive for SARS-CoV-2 since the start of the pandemic. Many pediatricians are still grappling with how to address cases like Spencer’s. The American Academy of Pediatrics has issued only brief guidance on long COVID in children, in part because there have been so few studies to use as a basis for guidance.
The federal government is aiming to change that with a newly launched National Research Action Plan on Long COVID that includes speeding up research on how the condition affects children and youths, including their ability to learn and thrive.
A CDC study found children with COVID were significantly more likely to have smell and taste disturbances, circulatory system problems, fatigue and malaise, and pain. Those who had been infected had higher rates of acute blockage of a lung artery, myocarditis and weakening of the heart, kidney failure, and type 1 diabetes.
Difficult to diagnose
Even with increased media attention and more published studies on pediatric long COVID, it’s still hard for a busy primary care doctor “to sort through what could just be a cold or what could be a series of colds and trying to look at the bigger picture of what’s been going on in a 1- to 3-month period with a kid,” Dr. Yonts said.
Most children with potential or definite long COVID are still being seen by individual pediatricians, not in a specialized clinic with easy access to an army of specialists. It’s not clear how many of those pediatric clinics exist. Survivor Corps, an advocacy group for people with long COVID, has posted a map of locations providing care, but few are specialized or focus on pediatric long COVID.
Long COVID is different from multisystem inflammatory syndrome in children (MIS-C), which occurs within a month or so of infection, triggers high fevers and severe symptoms in the gut, and often results in hospitalization. MIS-C “is not subtle,” said Dr. Edwards.
The long COVID clinic doctors said most of their patients were not very sick at first. “Anecdotally, of the 83 kids that we’ve seen, most have had mild, very mild, or even asymptomatic infections initially,” and then went on to have long COVID, said Dr. Yonts.
“We see it even in children who have very mild disease or even are asymptomatic,” agreed Allison Eckard, MD, director of pediatric infectious diseases at the Medical University of South Carolina, Charleston.
Fatigue, mood problems
Dr. Yonts said 90% of her patients have fatigue, and many also have severe symptoms in their gut. Those and other long COVID symptoms will be looked at more closely in a 3-year study the Children’s National Medical Center is doing along with the National Institute of Allergy and Infectious Diseases.
There are no treatments for long COVID itself.
“Management is probably more the correct term for what we do in our clinic at this point,” said Dr. Yonts. That means dealing with fatigue and managing headache and digestive symptoms with medications or coping strategies. Guidelines from the American Academy of Physical Medicine and Rehabilitation help inform how to help kids safely resume exercise.
At the Children’s National Medical Center clinic, children will typically meet with a team of specialists including infectious diseases doctors on the same day, said Dr. Yonts. Psychologists help children with coping skills. Dr. Yonts is careful not to imply that long COVID is a psychological illness. Parents “will just shut down, because for so long, they’ve been told this is all a mental thing.”
In about a third of children, symptoms get better on their own, and most kids get better over time. But many still struggle. “We don’t talk about cure, because we don’t know what cure looks like,” said Dr. Edwards.
Vaccination may be best protection
Vaccination seems to help reduce the risk of long COVID, perhaps by as much as half. But parents have been slow to vaccinate children, especially the very young. The AAP reported that, as of Aug. 3, just 5% of children under age 5, 37% of those ages 5-11, and 69% of 12- to 17-year-olds have received at least one dose of a COVID-19 vaccine.
“We have tried to really push vaccine as one of the ways to help prevent some of these long COVID syndromes,” said Dr. Eckard. But that advice is not always welcome. Dr. Eckard told the story of a mother who refused to have her autistic son vaccinated, even as she tearfully pleaded for help with his long COVID symptoms, which had also worsened his autism. The woman told Dr. Eckard: “Nothing you can say will convince me to get him vaccinated.” She thought a vaccine could make his symptoms even worse.
The best prevention is to avoid being infected in the first place.
“The more times you get COVID, the more you increase your risk of getting long COVID,” said Dr. Yonts. “The more times you roll the dice, eventually your number could come up.
A version of this article first appeared on WebMD.com.
Sexual dysfunction, hair loss linked with long COVID
according to findings of a large study.
Anuradhaa Subramanian, PhD, with the Institute of Applied Health Research at the University of Birmingham (England), led the research published online in Nature Medicine.
The team analyzed 486,149 electronic health records from adult patients with confirmed COVID in the United Kingdom, compared with 1.9 million people with no history of COVID, from January 2020 to April 2021. Researchers matched both groups closely in terms of demographic, social, and clinical traits.
New symptoms
The team identified 62 symptoms, including the well-known indicators of long COVID, such as fatigue, loss of sense of smell, shortness of breath, and brain fog, but also hair loss, sexual dysfunction, chest pain, fever, loss of control of bowel movements, and limb swelling.
“These differences in symptoms reported between the infected and uninfected groups remained even after we accounted for age, sex, ethnic group, socioeconomic status, body mass index, smoking status, the presence of more than 80 health conditions, and past reporting of the same symptom,” Dr. Subramanian and coresearcher Shamil Haroon, PhD, wrote in a summary of their research in The Conversation.
They pointed out that only 20 of the symptoms they found are included in the World Health Organization’s clinical case definition for long COVID.
They also found that people more likely to have persistent symptoms 3 months after COVID infection were also more likely to be young, female, smokers, to belong to certain minority ethnic groups, and to have lower socioeconomic status. They were also more likely to be obese and have a wide range of health conditions.
Dr. Haroon, an associate clinical professor at the University of Birmingham, said that one reason it appeared that younger people were more likely to get symptoms of long COVID may be that older adults with COVID were more likely to be hospitalized and weren’t included in this study.
“Since we only considered nonhospitalized adults, the older adults we included in our study may have been relatively healthier and thus had a lower symptom burden,” he said.
Dr. Subramania noted that older patients were more likely to report lasting COVID-related symptoms in the study, but when researchers accounted for a wide range of other conditions that patients had before infection (which generally more commonly happen in older adults), they found younger age as a risk factor for long-term COVID-related symptoms.
In the study period, most patients were unvaccinated, and results came before the widespread Delta and Omicron variants.
More than half (56.6%) of the patients infected with the virus that causes COVID had been diagnosed in 2020, and 43.4% in 2021. Less than 5% (4.5%) of the patients infected with the virus and 4.7% of the patients with no recorded evidence of a COVID infection had received at least a single dose of a COVID vaccine before the study started.
Eric Topol, MD, founder and director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape, said more studies need to be done to see whether results would be different with vaccination status and evolving variants.
But he noted that this study has several strengths: “The hair loss, libido loss, and ejaculation difficulty are all new symptoms,” and the study – large and carefully controlled – shows these issues were among those more likely to occur.
A loss of sense of smell – which is not a new observation – was still the most likely risk shown in the study, followed by hair loss, sneezing, ejaculation difficulty, and reduced sex drive; followed by shortness of breath, fatigue, chest pain associated with breathing difficulties, hoarseness, and fever.
Three main clusters of symptoms
Given the wide range of symptoms, long COVID likely represents a group of conditions, the authors wrote.
They found three main clusters. The largest, with roughly 80% of people with long COVID in the study, faced a broad spectrum of symptoms, ranging from fatigue to headache and pain. The second-largest group, (15%) mostly had symptoms having to do with mental health and thinking skills, including depression, anxiety, brain fog, and insomnia. The smallest group (5%) had mainly respiratory symptoms such as shortness of breath, coughing, and wheezing.
Putting symptoms in clusters will be important to start understanding what leads to long COVID, said Farha Ikramuddin, MD, a rehabilitation specialist at the University of Minnesota, Minneapolis.
She added that, while the symptoms listed in this paper are new in published research, she has certainly been seeing them over time in her long COVID clinic. (The researchers also used only coded health care data, so they were limited in what symptoms they could discover, she notes.)
Dr. Ikramuddin said a strength of the paper is its large size, but she also cautioned that it’s difficult to determine whether members of the comparison group truly had no COVID infection when the information is taken from their medical records. Often, people test at home or assume they have COVID and don’t test; therefore the information wouldn’t be recorded.
Evaluating nonhospitalized patients is also important, she said, as much of the research on long COVID has come from hospitalized patients, so little has been known about the symptoms of those with milder infections.
“Patients who have been hospitalized and have long COVID look very different from the patients who were not hospitalized,” Dr. Ikramuddin said.
One clear message from the paper, she said, is that listening and asking extensive questions about symptoms are important with patients who have had COVID.
“Counseling has also become very important for our patients in the pandemic,” she said.
It will also be important to do studies on returning to work for patients with long COVID to see how many are able to return and at what capacity, Dr. Ikramuddin said.
A version of this article first appeared on WebMD.com.
according to findings of a large study.
Anuradhaa Subramanian, PhD, with the Institute of Applied Health Research at the University of Birmingham (England), led the research published online in Nature Medicine.
The team analyzed 486,149 electronic health records from adult patients with confirmed COVID in the United Kingdom, compared with 1.9 million people with no history of COVID, from January 2020 to April 2021. Researchers matched both groups closely in terms of demographic, social, and clinical traits.
New symptoms
The team identified 62 symptoms, including the well-known indicators of long COVID, such as fatigue, loss of sense of smell, shortness of breath, and brain fog, but also hair loss, sexual dysfunction, chest pain, fever, loss of control of bowel movements, and limb swelling.
“These differences in symptoms reported between the infected and uninfected groups remained even after we accounted for age, sex, ethnic group, socioeconomic status, body mass index, smoking status, the presence of more than 80 health conditions, and past reporting of the same symptom,” Dr. Subramanian and coresearcher Shamil Haroon, PhD, wrote in a summary of their research in The Conversation.
They pointed out that only 20 of the symptoms they found are included in the World Health Organization’s clinical case definition for long COVID.
They also found that people more likely to have persistent symptoms 3 months after COVID infection were also more likely to be young, female, smokers, to belong to certain minority ethnic groups, and to have lower socioeconomic status. They were also more likely to be obese and have a wide range of health conditions.
Dr. Haroon, an associate clinical professor at the University of Birmingham, said that one reason it appeared that younger people were more likely to get symptoms of long COVID may be that older adults with COVID were more likely to be hospitalized and weren’t included in this study.
“Since we only considered nonhospitalized adults, the older adults we included in our study may have been relatively healthier and thus had a lower symptom burden,” he said.
Dr. Subramania noted that older patients were more likely to report lasting COVID-related symptoms in the study, but when researchers accounted for a wide range of other conditions that patients had before infection (which generally more commonly happen in older adults), they found younger age as a risk factor for long-term COVID-related symptoms.
In the study period, most patients were unvaccinated, and results came before the widespread Delta and Omicron variants.
More than half (56.6%) of the patients infected with the virus that causes COVID had been diagnosed in 2020, and 43.4% in 2021. Less than 5% (4.5%) of the patients infected with the virus and 4.7% of the patients with no recorded evidence of a COVID infection had received at least a single dose of a COVID vaccine before the study started.
Eric Topol, MD, founder and director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape, said more studies need to be done to see whether results would be different with vaccination status and evolving variants.
But he noted that this study has several strengths: “The hair loss, libido loss, and ejaculation difficulty are all new symptoms,” and the study – large and carefully controlled – shows these issues were among those more likely to occur.
A loss of sense of smell – which is not a new observation – was still the most likely risk shown in the study, followed by hair loss, sneezing, ejaculation difficulty, and reduced sex drive; followed by shortness of breath, fatigue, chest pain associated with breathing difficulties, hoarseness, and fever.
Three main clusters of symptoms
Given the wide range of symptoms, long COVID likely represents a group of conditions, the authors wrote.
They found three main clusters. The largest, with roughly 80% of people with long COVID in the study, faced a broad spectrum of symptoms, ranging from fatigue to headache and pain. The second-largest group, (15%) mostly had symptoms having to do with mental health and thinking skills, including depression, anxiety, brain fog, and insomnia. The smallest group (5%) had mainly respiratory symptoms such as shortness of breath, coughing, and wheezing.
Putting symptoms in clusters will be important to start understanding what leads to long COVID, said Farha Ikramuddin, MD, a rehabilitation specialist at the University of Minnesota, Minneapolis.
She added that, while the symptoms listed in this paper are new in published research, she has certainly been seeing them over time in her long COVID clinic. (The researchers also used only coded health care data, so they were limited in what symptoms they could discover, she notes.)
Dr. Ikramuddin said a strength of the paper is its large size, but she also cautioned that it’s difficult to determine whether members of the comparison group truly had no COVID infection when the information is taken from their medical records. Often, people test at home or assume they have COVID and don’t test; therefore the information wouldn’t be recorded.
Evaluating nonhospitalized patients is also important, she said, as much of the research on long COVID has come from hospitalized patients, so little has been known about the symptoms of those with milder infections.
“Patients who have been hospitalized and have long COVID look very different from the patients who were not hospitalized,” Dr. Ikramuddin said.
One clear message from the paper, she said, is that listening and asking extensive questions about symptoms are important with patients who have had COVID.
“Counseling has also become very important for our patients in the pandemic,” she said.
It will also be important to do studies on returning to work for patients with long COVID to see how many are able to return and at what capacity, Dr. Ikramuddin said.
A version of this article first appeared on WebMD.com.
according to findings of a large study.
Anuradhaa Subramanian, PhD, with the Institute of Applied Health Research at the University of Birmingham (England), led the research published online in Nature Medicine.
The team analyzed 486,149 electronic health records from adult patients with confirmed COVID in the United Kingdom, compared with 1.9 million people with no history of COVID, from January 2020 to April 2021. Researchers matched both groups closely in terms of demographic, social, and clinical traits.
New symptoms
The team identified 62 symptoms, including the well-known indicators of long COVID, such as fatigue, loss of sense of smell, shortness of breath, and brain fog, but also hair loss, sexual dysfunction, chest pain, fever, loss of control of bowel movements, and limb swelling.
“These differences in symptoms reported between the infected and uninfected groups remained even after we accounted for age, sex, ethnic group, socioeconomic status, body mass index, smoking status, the presence of more than 80 health conditions, and past reporting of the same symptom,” Dr. Subramanian and coresearcher Shamil Haroon, PhD, wrote in a summary of their research in The Conversation.
They pointed out that only 20 of the symptoms they found are included in the World Health Organization’s clinical case definition for long COVID.
They also found that people more likely to have persistent symptoms 3 months after COVID infection were also more likely to be young, female, smokers, to belong to certain minority ethnic groups, and to have lower socioeconomic status. They were also more likely to be obese and have a wide range of health conditions.
Dr. Haroon, an associate clinical professor at the University of Birmingham, said that one reason it appeared that younger people were more likely to get symptoms of long COVID may be that older adults with COVID were more likely to be hospitalized and weren’t included in this study.
“Since we only considered nonhospitalized adults, the older adults we included in our study may have been relatively healthier and thus had a lower symptom burden,” he said.
Dr. Subramania noted that older patients were more likely to report lasting COVID-related symptoms in the study, but when researchers accounted for a wide range of other conditions that patients had before infection (which generally more commonly happen in older adults), they found younger age as a risk factor for long-term COVID-related symptoms.
In the study period, most patients were unvaccinated, and results came before the widespread Delta and Omicron variants.
More than half (56.6%) of the patients infected with the virus that causes COVID had been diagnosed in 2020, and 43.4% in 2021. Less than 5% (4.5%) of the patients infected with the virus and 4.7% of the patients with no recorded evidence of a COVID infection had received at least a single dose of a COVID vaccine before the study started.
Eric Topol, MD, founder and director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape, said more studies need to be done to see whether results would be different with vaccination status and evolving variants.
But he noted that this study has several strengths: “The hair loss, libido loss, and ejaculation difficulty are all new symptoms,” and the study – large and carefully controlled – shows these issues were among those more likely to occur.
A loss of sense of smell – which is not a new observation – was still the most likely risk shown in the study, followed by hair loss, sneezing, ejaculation difficulty, and reduced sex drive; followed by shortness of breath, fatigue, chest pain associated with breathing difficulties, hoarseness, and fever.
Three main clusters of symptoms
Given the wide range of symptoms, long COVID likely represents a group of conditions, the authors wrote.
They found three main clusters. The largest, with roughly 80% of people with long COVID in the study, faced a broad spectrum of symptoms, ranging from fatigue to headache and pain. The second-largest group, (15%) mostly had symptoms having to do with mental health and thinking skills, including depression, anxiety, brain fog, and insomnia. The smallest group (5%) had mainly respiratory symptoms such as shortness of breath, coughing, and wheezing.
Putting symptoms in clusters will be important to start understanding what leads to long COVID, said Farha Ikramuddin, MD, a rehabilitation specialist at the University of Minnesota, Minneapolis.
She added that, while the symptoms listed in this paper are new in published research, she has certainly been seeing them over time in her long COVID clinic. (The researchers also used only coded health care data, so they were limited in what symptoms they could discover, she notes.)
Dr. Ikramuddin said a strength of the paper is its large size, but she also cautioned that it’s difficult to determine whether members of the comparison group truly had no COVID infection when the information is taken from their medical records. Often, people test at home or assume they have COVID and don’t test; therefore the information wouldn’t be recorded.
Evaluating nonhospitalized patients is also important, she said, as much of the research on long COVID has come from hospitalized patients, so little has been known about the symptoms of those with milder infections.
“Patients who have been hospitalized and have long COVID look very different from the patients who were not hospitalized,” Dr. Ikramuddin said.
One clear message from the paper, she said, is that listening and asking extensive questions about symptoms are important with patients who have had COVID.
“Counseling has also become very important for our patients in the pandemic,” she said.
It will also be important to do studies on returning to work for patients with long COVID to see how many are able to return and at what capacity, Dr. Ikramuddin said.
A version of this article first appeared on WebMD.com.
FROM NATURE MEDICINE
Long COVID’s grip will likely tighten as infections continue
COVID-19 is far from done in the United States, with more than 111,000 new cases being recorded a day in the second week of August, according to Johns Hopkins University, and 625 deaths being reported every day. , a condition that already has affected between 7.7 million and 23 million Americans, according to U.S. government estimates.
“It is evident that long COVID is real, that it already impacts a substantial number of people, and that this number may continue to grow as new infections occur,” the U.S. Department of Health and Human Services (HHS) said in a research action plan released Aug. 4.
“We are heading towards a big problem on our hands,” says Ziyad Al-Aly, MD, chief of research and development at the Veterans Affairs Hospital in St. Louis. “It’s like if we are falling in a plane, hurtling towards the ground. It doesn’t matter at what speed we are falling; what matters is that we are all falling, and falling fast. It’s a real problem. We needed to bring attention to this, yesterday,” he said.
Bryan Lau, PhD, professor of epidemiology at Johns Hopkins Bloomberg School of Public Health, Baltimore, and co-lead of a long COVID study there, says whether it’s 5% of the 92 million officially recorded U.S. COVID-19 cases, or 30% – on the higher end of estimates – that means anywhere between 4.5 million and 27 million Americans will have the effects of long COVID.
Other experts put the estimates even higher.
“If we conservatively assume 100 million working-age adults have been infected, that implies 10 to 33 million may have long COVID,” Alice Burns, PhD, associate director for the Kaiser Family Foundation’s Program on Medicaid and the Uninsured, wrote in an analysis.
And even the Centers for Disease Control and Prevention says only a fraction of cases have been recorded.
That, in turn, means tens of millions of people who struggle to work, to get to school, and to take care of their families – and who will be making demands on an already stressed U.S. health care system.
The HHS said in its Aug. 4 report that long COVID could keep 1 million people a day out of work, with a loss of $50 billion in annual pay.
Dr. Lau said health workers and policymakers are woefully unprepared.
“If you have a family unit, and the mom or dad can’t work, or has trouble taking their child to activities, where does the question of support come into play? Where is there potential for food issues, or housing issues?” he asked. “I see the potential for the burden to be extremely large in that capacity.”
Dr. Lau said he has yet to see any strong estimates of how many cases of long COVID might develop. Because a person has to get COVID-19 to ultimately get long COVID, the two are linked. In other words, as COVID-19 cases rise, so will cases of long COVID, and vice versa.
Evidence from the Kaiser Family Foundation analysis suggests a significant impact on employment: Surveys showed more than half of adults with long COVID who worked before becoming infected are either out of work or working fewer hours. Conditions associated with long COVID – such as fatigue, malaise, or problems concentrating – limit people’s ability to work, even if they have jobs that allow for accommodations.
Two surveys of people with long COVID who had worked before becoming infected showed that between 22% and 27% of them were out of work after getting long COVID. In comparison, among all working-age adults in 2019, only 7% were out of work. Given the sheer number of working-age adults with long COVID, the effects on employment may be profound and are likely to involve more people over time. One study estimates that long COVID already accounts for 15% of unfilled jobs.
The most severe symptoms of long COVID include brain fog and heart complications, known to persist for weeks for months after a COVID-19 infection.
A study from the University of Norway published in Open Forum Infectious Diseases found 53% of people tested had at least one symptom of thinking problems 13 months after infection with COVID-19. According to the HHS’ latest report on long COVID, people with thinking problems, heart conditions, mobility issues, and other symptoms are going to need a considerable amount of care. Many will need lengthy periods of rehabilitation.
Dr. Al-Aly worries that long COVID has already severely affected the labor force and the job market, all while burdening the country’s health care system.
“While there are variations in how individuals respond and cope with long COVID, the unifying thread is that with the level of disability it causes, more people will be struggling to keep up with the demands of the workforce and more people will be out on disability than ever before,” he said.
Studies from Johns Hopkins and the University of Washington estimate that 5%-30% of people could get long COVID in the future. Projections beyond that are hazy.
“So far, all the studies we have done on long COVID have been reactionary. Much of the activism around long COVID has been patient led. We are seeing more and more people with lasting symptoms. We need our research to catch up,” Dr. Lau said.
Theo Vos, MD, PhD, professor of health sciences at University of Washington, Seattle, said the main reasons for the huge range of predictions are the variety of methods used, as well as differences in sample size. Also, much long COVID data is self-reported, making it difficult for epidemiologists to track.
“With self-reported data, you can’t plug people into a machine and say this is what they have or this is what they don’t have. At the population level, the only thing you can do is ask questions. There is no systematic way to define long COVID,” he said.
Dr. Vos’s most recent study, which is being peer-reviewed and revised, found that most people with long COVID have symptoms similar to those seen in other autoimmune diseases. But sometimes the immune system can overreact, causing the more severe symptoms, such as brain fog and heart problems, associated with long COVID.
One reason that researchers struggle to come up with numbers, said Dr. Al-Aly, is the rapid rise of new variants. These variants appear to sometimes cause less severe disease than previous ones, but it’s not clear whether that means different risks for long COVID.
“There’s a wide diversity in severity. Someone can have long COVID and be fully functional, while others are not functional at all. We still have a long way to go before we figure out why,” Dr. Lau said.
A version of this article first appeared on WebMD.com.
COVID-19 is far from done in the United States, with more than 111,000 new cases being recorded a day in the second week of August, according to Johns Hopkins University, and 625 deaths being reported every day. , a condition that already has affected between 7.7 million and 23 million Americans, according to U.S. government estimates.
“It is evident that long COVID is real, that it already impacts a substantial number of people, and that this number may continue to grow as new infections occur,” the U.S. Department of Health and Human Services (HHS) said in a research action plan released Aug. 4.
“We are heading towards a big problem on our hands,” says Ziyad Al-Aly, MD, chief of research and development at the Veterans Affairs Hospital in St. Louis. “It’s like if we are falling in a plane, hurtling towards the ground. It doesn’t matter at what speed we are falling; what matters is that we are all falling, and falling fast. It’s a real problem. We needed to bring attention to this, yesterday,” he said.
Bryan Lau, PhD, professor of epidemiology at Johns Hopkins Bloomberg School of Public Health, Baltimore, and co-lead of a long COVID study there, says whether it’s 5% of the 92 million officially recorded U.S. COVID-19 cases, or 30% – on the higher end of estimates – that means anywhere between 4.5 million and 27 million Americans will have the effects of long COVID.
Other experts put the estimates even higher.
“If we conservatively assume 100 million working-age adults have been infected, that implies 10 to 33 million may have long COVID,” Alice Burns, PhD, associate director for the Kaiser Family Foundation’s Program on Medicaid and the Uninsured, wrote in an analysis.
And even the Centers for Disease Control and Prevention says only a fraction of cases have been recorded.
That, in turn, means tens of millions of people who struggle to work, to get to school, and to take care of their families – and who will be making demands on an already stressed U.S. health care system.
The HHS said in its Aug. 4 report that long COVID could keep 1 million people a day out of work, with a loss of $50 billion in annual pay.
Dr. Lau said health workers and policymakers are woefully unprepared.
“If you have a family unit, and the mom or dad can’t work, or has trouble taking their child to activities, where does the question of support come into play? Where is there potential for food issues, or housing issues?” he asked. “I see the potential for the burden to be extremely large in that capacity.”
Dr. Lau said he has yet to see any strong estimates of how many cases of long COVID might develop. Because a person has to get COVID-19 to ultimately get long COVID, the two are linked. In other words, as COVID-19 cases rise, so will cases of long COVID, and vice versa.
Evidence from the Kaiser Family Foundation analysis suggests a significant impact on employment: Surveys showed more than half of adults with long COVID who worked before becoming infected are either out of work or working fewer hours. Conditions associated with long COVID – such as fatigue, malaise, or problems concentrating – limit people’s ability to work, even if they have jobs that allow for accommodations.
Two surveys of people with long COVID who had worked before becoming infected showed that between 22% and 27% of them were out of work after getting long COVID. In comparison, among all working-age adults in 2019, only 7% were out of work. Given the sheer number of working-age adults with long COVID, the effects on employment may be profound and are likely to involve more people over time. One study estimates that long COVID already accounts for 15% of unfilled jobs.
The most severe symptoms of long COVID include brain fog and heart complications, known to persist for weeks for months after a COVID-19 infection.
A study from the University of Norway published in Open Forum Infectious Diseases found 53% of people tested had at least one symptom of thinking problems 13 months after infection with COVID-19. According to the HHS’ latest report on long COVID, people with thinking problems, heart conditions, mobility issues, and other symptoms are going to need a considerable amount of care. Many will need lengthy periods of rehabilitation.
Dr. Al-Aly worries that long COVID has already severely affected the labor force and the job market, all while burdening the country’s health care system.
“While there are variations in how individuals respond and cope with long COVID, the unifying thread is that with the level of disability it causes, more people will be struggling to keep up with the demands of the workforce and more people will be out on disability than ever before,” he said.
Studies from Johns Hopkins and the University of Washington estimate that 5%-30% of people could get long COVID in the future. Projections beyond that are hazy.
“So far, all the studies we have done on long COVID have been reactionary. Much of the activism around long COVID has been patient led. We are seeing more and more people with lasting symptoms. We need our research to catch up,” Dr. Lau said.
Theo Vos, MD, PhD, professor of health sciences at University of Washington, Seattle, said the main reasons for the huge range of predictions are the variety of methods used, as well as differences in sample size. Also, much long COVID data is self-reported, making it difficult for epidemiologists to track.
“With self-reported data, you can’t plug people into a machine and say this is what they have or this is what they don’t have. At the population level, the only thing you can do is ask questions. There is no systematic way to define long COVID,” he said.
Dr. Vos’s most recent study, which is being peer-reviewed and revised, found that most people with long COVID have symptoms similar to those seen in other autoimmune diseases. But sometimes the immune system can overreact, causing the more severe symptoms, such as brain fog and heart problems, associated with long COVID.
One reason that researchers struggle to come up with numbers, said Dr. Al-Aly, is the rapid rise of new variants. These variants appear to sometimes cause less severe disease than previous ones, but it’s not clear whether that means different risks for long COVID.
“There’s a wide diversity in severity. Someone can have long COVID and be fully functional, while others are not functional at all. We still have a long way to go before we figure out why,” Dr. Lau said.
A version of this article first appeared on WebMD.com.
COVID-19 is far from done in the United States, with more than 111,000 new cases being recorded a day in the second week of August, according to Johns Hopkins University, and 625 deaths being reported every day. , a condition that already has affected between 7.7 million and 23 million Americans, according to U.S. government estimates.
“It is evident that long COVID is real, that it already impacts a substantial number of people, and that this number may continue to grow as new infections occur,” the U.S. Department of Health and Human Services (HHS) said in a research action plan released Aug. 4.
“We are heading towards a big problem on our hands,” says Ziyad Al-Aly, MD, chief of research and development at the Veterans Affairs Hospital in St. Louis. “It’s like if we are falling in a plane, hurtling towards the ground. It doesn’t matter at what speed we are falling; what matters is that we are all falling, and falling fast. It’s a real problem. We needed to bring attention to this, yesterday,” he said.
Bryan Lau, PhD, professor of epidemiology at Johns Hopkins Bloomberg School of Public Health, Baltimore, and co-lead of a long COVID study there, says whether it’s 5% of the 92 million officially recorded U.S. COVID-19 cases, or 30% – on the higher end of estimates – that means anywhere between 4.5 million and 27 million Americans will have the effects of long COVID.
Other experts put the estimates even higher.
“If we conservatively assume 100 million working-age adults have been infected, that implies 10 to 33 million may have long COVID,” Alice Burns, PhD, associate director for the Kaiser Family Foundation’s Program on Medicaid and the Uninsured, wrote in an analysis.
And even the Centers for Disease Control and Prevention says only a fraction of cases have been recorded.
That, in turn, means tens of millions of people who struggle to work, to get to school, and to take care of their families – and who will be making demands on an already stressed U.S. health care system.
The HHS said in its Aug. 4 report that long COVID could keep 1 million people a day out of work, with a loss of $50 billion in annual pay.
Dr. Lau said health workers and policymakers are woefully unprepared.
“If you have a family unit, and the mom or dad can’t work, or has trouble taking their child to activities, where does the question of support come into play? Where is there potential for food issues, or housing issues?” he asked. “I see the potential for the burden to be extremely large in that capacity.”
Dr. Lau said he has yet to see any strong estimates of how many cases of long COVID might develop. Because a person has to get COVID-19 to ultimately get long COVID, the two are linked. In other words, as COVID-19 cases rise, so will cases of long COVID, and vice versa.
Evidence from the Kaiser Family Foundation analysis suggests a significant impact on employment: Surveys showed more than half of adults with long COVID who worked before becoming infected are either out of work or working fewer hours. Conditions associated with long COVID – such as fatigue, malaise, or problems concentrating – limit people’s ability to work, even if they have jobs that allow for accommodations.
Two surveys of people with long COVID who had worked before becoming infected showed that between 22% and 27% of them were out of work after getting long COVID. In comparison, among all working-age adults in 2019, only 7% were out of work. Given the sheer number of working-age adults with long COVID, the effects on employment may be profound and are likely to involve more people over time. One study estimates that long COVID already accounts for 15% of unfilled jobs.
The most severe symptoms of long COVID include brain fog and heart complications, known to persist for weeks for months after a COVID-19 infection.
A study from the University of Norway published in Open Forum Infectious Diseases found 53% of people tested had at least one symptom of thinking problems 13 months after infection with COVID-19. According to the HHS’ latest report on long COVID, people with thinking problems, heart conditions, mobility issues, and other symptoms are going to need a considerable amount of care. Many will need lengthy periods of rehabilitation.
Dr. Al-Aly worries that long COVID has already severely affected the labor force and the job market, all while burdening the country’s health care system.
“While there are variations in how individuals respond and cope with long COVID, the unifying thread is that with the level of disability it causes, more people will be struggling to keep up with the demands of the workforce and more people will be out on disability than ever before,” he said.
Studies from Johns Hopkins and the University of Washington estimate that 5%-30% of people could get long COVID in the future. Projections beyond that are hazy.
“So far, all the studies we have done on long COVID have been reactionary. Much of the activism around long COVID has been patient led. We are seeing more and more people with lasting symptoms. We need our research to catch up,” Dr. Lau said.
Theo Vos, MD, PhD, professor of health sciences at University of Washington, Seattle, said the main reasons for the huge range of predictions are the variety of methods used, as well as differences in sample size. Also, much long COVID data is self-reported, making it difficult for epidemiologists to track.
“With self-reported data, you can’t plug people into a machine and say this is what they have or this is what they don’t have. At the population level, the only thing you can do is ask questions. There is no systematic way to define long COVID,” he said.
Dr. Vos’s most recent study, which is being peer-reviewed and revised, found that most people with long COVID have symptoms similar to those seen in other autoimmune diseases. But sometimes the immune system can overreact, causing the more severe symptoms, such as brain fog and heart problems, associated with long COVID.
One reason that researchers struggle to come up with numbers, said Dr. Al-Aly, is the rapid rise of new variants. These variants appear to sometimes cause less severe disease than previous ones, but it’s not clear whether that means different risks for long COVID.
“There’s a wide diversity in severity. Someone can have long COVID and be fully functional, while others are not functional at all. We still have a long way to go before we figure out why,” Dr. Lau said.
A version of this article first appeared on WebMD.com.




