User login
Long COVID & Chronic Fatigue: The Similarities are Uncanny
An estimated two million people in England and Scotland were experiencing symptoms of long COVID as of March 2024, according to the Office for National Statistics. Of these, 1.5 million said the condition was adversely affecting their day-to-day activities.
As more research emerges about long COVID, some experts are noticing that its trigger factors, symptoms, and causative mechanisms overlap with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS).
ME/CFS is characterized by severe fatigue that does not improve with rest, in addition to pain and cognitive problems. One in four patients are bed- or house-bound with severe forms of the condition, sometimes experiencing atypical seizures, and speech and swallowing difficulties.
Despite affecting around 250,000 people in the UK and around 2 million people in the European Union (EU), it is a relatively poorly funded disease research area. Increased research into long COVID is thus providing a much-needed boost to ME/CFS research.
“What we already know about the possible causation of ME/CFS is helping research into the causes of long COVID. At the same time, research into long COVID is opening up new avenues of research that may also be relevant to ME/CFS. It is becoming a two-way process,” Dr. Charles Shepherd, honorary medical adviser to the UK-based ME Association, told this news organization.
While funding remains an issue, promising research is currently underway in the UK to improve diagnosis, treatment, and understanding of the pathology of ME/CFS.
Viral Reactivation
Dr. David Newton is research director at ME Research UK. “Viral infection is commonly reported as a trigger for [ME/CFS, meaning that the disease] may be caused by reactivation of latent viruses, including human herpes viruses and enteroviruses,” he said.
Herpes viruses can lie dormant in their host’s immune system for long periods of time. They can be reactivated by factors including infections, stress, and a weakened immune system, and may cause temporary symptoms or persistent disease.
A 2021 pilot study found that people with ME/CFS have a higher concentration of human herpesvirus 6B (HHV-6B) DNA in their saliva, and that concentration correlates with symptom severity. HHV-6B is a common virus typically contracted during infancy and childhood.
A continuation of this research is now underway at Brunel University to improve understanding of HHV-6B’s role in the onset and progression of ME/CFS, and to support the development of diagnostic and prognostic markers, as well as therapeutics such as antiviral therapies.
Mitochondrial Dysfunction
Dr. Shepherd explained that there is now sound evidence demonstrating that biochemical abnormalities in ME/CFS affect how mitochondria produce energy after physical exertion. Research is thus underway to see if treating mitochondrial dysfunction improves ME/CFS symptoms.
A phase 2a placebo-controlled clinical trial from 2023 found that AXA1125, a drug that works by modulating energy metabolism, significantly improved symptoms of fatigue in patients with fatigue-dominant long COVID, although it did not improve mitochondrial respiration.
“[The findings suggest] that improving mitochondrial health may be one way to restore normal functioning among people with long COVID, and by extension CFS,” study author Betty Raman, associate professor of cardiovascular medicine at the University of Oxford, told this news organization. She noted, however, that plans for a phase III trial have stalled due to insufficient funding.
Meanwhile, researchers from the Quadram Institute in Norwich and the University of East Anglia are conducting a pilot study to see if red light therapy can relieve symptoms of ME/CFS. Red light can be absorbed by mitochondria and is used to boost energy production. The trial will monitor patients remotely from their homes and will assess cognitive function and physical activity levels.
Gut Dysbiosis
Many studies have found that people with ME/CFS have altered gut microbiota, which suggests that changes in gut bacteria may contribute to the condition. Researchers at the Quadram Institute will thus conduct a clinical trial called RESTORE-ME to see whether fecal microbiota transplants (FMT) can treat the condition.
Rik Haagmans is a research scientist and PhD candidate at the Quadram Institute. He told this news organization: “Our FMT studies, if effective, could provide a longer lasting or even permanent relief of ME/CFS, as restoring the gut microbial composition wouldn’t require continuous medication,” he said.
Biobank and Biomarkers
Europe’s first ME/CFS-specific biobank is in the UK and is called UKMEB. It now has more than 30,000 blood samples from patients with ME/CFS, multiple sclerosis, and healthy controls. Uniquely, it includes samples from people with ME/CFS who are house- and bed-bound. Caroline Kingdon, RN, MSc, a research fellow and biobank lead at the London School of Hygiene and Tropical Medicine, told this news organization that samples and data from the UKMEB have been provided to research groups all over the world and have contributed to widely cited literature.
One group making use of these samples is led by Fatima Labeed, PhD, senior lecturer in human biology at the University of Surrey. Dr. Labeed and her team are developing a diagnostic test for ME/CFS based on electrical properties in white blood cells.
“To date, studies of ME/CFS have focused on the biochemical behavior of cells: the amount and type of proteins that cells use. We have taken a different approach, studying the electrical properties,” she explained to this news organization.
Her research builds on initial observations from 2019 that found differences in the electrical impedance of white blood cells between people with ME/CFS and controls. While the biological implications remain unknown, the findings may represent a biomarker for the condition.
Using blood samples from the UKMEB, the researchers are now investigating this potential biomarker with improved techniques and a larger patient cohort, including those with mild/moderate and severe forms of ME/CFS. So far, they have received more than 100 blood samples and have analyzed the electrical properties of 42.
“Based on the results we have so far, we are very close to having a biomarker for diagnosis. Our results so far show a high degree of accuracy and are able to distinguish between ME/CFS and other diseases,” said Dr. Labeed.
Genetic Test
Another promising avenue for diagnostics comes from a research team at the University of Edinburgh led by Professor Chris Ponting at the university’s Institute of Genetics and Cancer. They are currently working on DecodeMe, a large genetic study of ME using data from more than 26,000 people.
“We are studying blood-based biomarkers that distinguish people with ME from population controls. We’ve found a large number — including some found previously in other studies — and are writing these results up for publication,” said Ponting. The results should be published in early 2025.
The Future
While research into ME/CFS has picked up pace in recent years, funding remains a key bottleneck.
“Over the last 10 years, only £8.05m has been spent on ME research,” Sonya Chowdhury, chief executive of UK charity Action for ME told this news organization. She believes this amount is not equitably comparable to research funding allocated to other diseases.
In 2022, the UK government announced its intention to develop a cross-government interim delivery plan on ME/CFS for England, however publication of the final plan has been delayed numerous times.
Dr. Shepherd agreed that increased funding is crucial for progress to be made. He said the biggest help to ME/CFS research would be to end the disparity in government research funding for the disease, and match what is given for many other disabling long-term conditions.
“It’s not fair to continue to rely on the charity sector to fund almost all of the biomedical research into ME/CFS here in the UK,” he said.
A version of this article appeared on Medscape.com.
An estimated two million people in England and Scotland were experiencing symptoms of long COVID as of March 2024, according to the Office for National Statistics. Of these, 1.5 million said the condition was adversely affecting their day-to-day activities.
As more research emerges about long COVID, some experts are noticing that its trigger factors, symptoms, and causative mechanisms overlap with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS).
ME/CFS is characterized by severe fatigue that does not improve with rest, in addition to pain and cognitive problems. One in four patients are bed- or house-bound with severe forms of the condition, sometimes experiencing atypical seizures, and speech and swallowing difficulties.
Despite affecting around 250,000 people in the UK and around 2 million people in the European Union (EU), it is a relatively poorly funded disease research area. Increased research into long COVID is thus providing a much-needed boost to ME/CFS research.
“What we already know about the possible causation of ME/CFS is helping research into the causes of long COVID. At the same time, research into long COVID is opening up new avenues of research that may also be relevant to ME/CFS. It is becoming a two-way process,” Dr. Charles Shepherd, honorary medical adviser to the UK-based ME Association, told this news organization.
While funding remains an issue, promising research is currently underway in the UK to improve diagnosis, treatment, and understanding of the pathology of ME/CFS.
Viral Reactivation
Dr. David Newton is research director at ME Research UK. “Viral infection is commonly reported as a trigger for [ME/CFS, meaning that the disease] may be caused by reactivation of latent viruses, including human herpes viruses and enteroviruses,” he said.
Herpes viruses can lie dormant in their host’s immune system for long periods of time. They can be reactivated by factors including infections, stress, and a weakened immune system, and may cause temporary symptoms or persistent disease.
A 2021 pilot study found that people with ME/CFS have a higher concentration of human herpesvirus 6B (HHV-6B) DNA in their saliva, and that concentration correlates with symptom severity. HHV-6B is a common virus typically contracted during infancy and childhood.
A continuation of this research is now underway at Brunel University to improve understanding of HHV-6B’s role in the onset and progression of ME/CFS, and to support the development of diagnostic and prognostic markers, as well as therapeutics such as antiviral therapies.
Mitochondrial Dysfunction
Dr. Shepherd explained that there is now sound evidence demonstrating that biochemical abnormalities in ME/CFS affect how mitochondria produce energy after physical exertion. Research is thus underway to see if treating mitochondrial dysfunction improves ME/CFS symptoms.
A phase 2a placebo-controlled clinical trial from 2023 found that AXA1125, a drug that works by modulating energy metabolism, significantly improved symptoms of fatigue in patients with fatigue-dominant long COVID, although it did not improve mitochondrial respiration.
“[The findings suggest] that improving mitochondrial health may be one way to restore normal functioning among people with long COVID, and by extension CFS,” study author Betty Raman, associate professor of cardiovascular medicine at the University of Oxford, told this news organization. She noted, however, that plans for a phase III trial have stalled due to insufficient funding.
Meanwhile, researchers from the Quadram Institute in Norwich and the University of East Anglia are conducting a pilot study to see if red light therapy can relieve symptoms of ME/CFS. Red light can be absorbed by mitochondria and is used to boost energy production. The trial will monitor patients remotely from their homes and will assess cognitive function and physical activity levels.
Gut Dysbiosis
Many studies have found that people with ME/CFS have altered gut microbiota, which suggests that changes in gut bacteria may contribute to the condition. Researchers at the Quadram Institute will thus conduct a clinical trial called RESTORE-ME to see whether fecal microbiota transplants (FMT) can treat the condition.
Rik Haagmans is a research scientist and PhD candidate at the Quadram Institute. He told this news organization: “Our FMT studies, if effective, could provide a longer lasting or even permanent relief of ME/CFS, as restoring the gut microbial composition wouldn’t require continuous medication,” he said.
Biobank and Biomarkers
Europe’s first ME/CFS-specific biobank is in the UK and is called UKMEB. It now has more than 30,000 blood samples from patients with ME/CFS, multiple sclerosis, and healthy controls. Uniquely, it includes samples from people with ME/CFS who are house- and bed-bound. Caroline Kingdon, RN, MSc, a research fellow and biobank lead at the London School of Hygiene and Tropical Medicine, told this news organization that samples and data from the UKMEB have been provided to research groups all over the world and have contributed to widely cited literature.
One group making use of these samples is led by Fatima Labeed, PhD, senior lecturer in human biology at the University of Surrey. Dr. Labeed and her team are developing a diagnostic test for ME/CFS based on electrical properties in white blood cells.
“To date, studies of ME/CFS have focused on the biochemical behavior of cells: the amount and type of proteins that cells use. We have taken a different approach, studying the electrical properties,” she explained to this news organization.
Her research builds on initial observations from 2019 that found differences in the electrical impedance of white blood cells between people with ME/CFS and controls. While the biological implications remain unknown, the findings may represent a biomarker for the condition.
Using blood samples from the UKMEB, the researchers are now investigating this potential biomarker with improved techniques and a larger patient cohort, including those with mild/moderate and severe forms of ME/CFS. So far, they have received more than 100 blood samples and have analyzed the electrical properties of 42.
“Based on the results we have so far, we are very close to having a biomarker for diagnosis. Our results so far show a high degree of accuracy and are able to distinguish between ME/CFS and other diseases,” said Dr. Labeed.
Genetic Test
Another promising avenue for diagnostics comes from a research team at the University of Edinburgh led by Professor Chris Ponting at the university’s Institute of Genetics and Cancer. They are currently working on DecodeMe, a large genetic study of ME using data from more than 26,000 people.
“We are studying blood-based biomarkers that distinguish people with ME from population controls. We’ve found a large number — including some found previously in other studies — and are writing these results up for publication,” said Ponting. The results should be published in early 2025.
The Future
While research into ME/CFS has picked up pace in recent years, funding remains a key bottleneck.
“Over the last 10 years, only £8.05m has been spent on ME research,” Sonya Chowdhury, chief executive of UK charity Action for ME told this news organization. She believes this amount is not equitably comparable to research funding allocated to other diseases.
In 2022, the UK government announced its intention to develop a cross-government interim delivery plan on ME/CFS for England, however publication of the final plan has been delayed numerous times.
Dr. Shepherd agreed that increased funding is crucial for progress to be made. He said the biggest help to ME/CFS research would be to end the disparity in government research funding for the disease, and match what is given for many other disabling long-term conditions.
“It’s not fair to continue to rely on the charity sector to fund almost all of the biomedical research into ME/CFS here in the UK,” he said.
A version of this article appeared on Medscape.com.
An estimated two million people in England and Scotland were experiencing symptoms of long COVID as of March 2024, according to the Office for National Statistics. Of these, 1.5 million said the condition was adversely affecting their day-to-day activities.
As more research emerges about long COVID, some experts are noticing that its trigger factors, symptoms, and causative mechanisms overlap with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS).
ME/CFS is characterized by severe fatigue that does not improve with rest, in addition to pain and cognitive problems. One in four patients are bed- or house-bound with severe forms of the condition, sometimes experiencing atypical seizures, and speech and swallowing difficulties.
Despite affecting around 250,000 people in the UK and around 2 million people in the European Union (EU), it is a relatively poorly funded disease research area. Increased research into long COVID is thus providing a much-needed boost to ME/CFS research.
“What we already know about the possible causation of ME/CFS is helping research into the causes of long COVID. At the same time, research into long COVID is opening up new avenues of research that may also be relevant to ME/CFS. It is becoming a two-way process,” Dr. Charles Shepherd, honorary medical adviser to the UK-based ME Association, told this news organization.
While funding remains an issue, promising research is currently underway in the UK to improve diagnosis, treatment, and understanding of the pathology of ME/CFS.
Viral Reactivation
Dr. David Newton is research director at ME Research UK. “Viral infection is commonly reported as a trigger for [ME/CFS, meaning that the disease] may be caused by reactivation of latent viruses, including human herpes viruses and enteroviruses,” he said.
Herpes viruses can lie dormant in their host’s immune system for long periods of time. They can be reactivated by factors including infections, stress, and a weakened immune system, and may cause temporary symptoms or persistent disease.
A 2021 pilot study found that people with ME/CFS have a higher concentration of human herpesvirus 6B (HHV-6B) DNA in their saliva, and that concentration correlates with symptom severity. HHV-6B is a common virus typically contracted during infancy and childhood.
A continuation of this research is now underway at Brunel University to improve understanding of HHV-6B’s role in the onset and progression of ME/CFS, and to support the development of diagnostic and prognostic markers, as well as therapeutics such as antiviral therapies.
Mitochondrial Dysfunction
Dr. Shepherd explained that there is now sound evidence demonstrating that biochemical abnormalities in ME/CFS affect how mitochondria produce energy after physical exertion. Research is thus underway to see if treating mitochondrial dysfunction improves ME/CFS symptoms.
A phase 2a placebo-controlled clinical trial from 2023 found that AXA1125, a drug that works by modulating energy metabolism, significantly improved symptoms of fatigue in patients with fatigue-dominant long COVID, although it did not improve mitochondrial respiration.
“[The findings suggest] that improving mitochondrial health may be one way to restore normal functioning among people with long COVID, and by extension CFS,” study author Betty Raman, associate professor of cardiovascular medicine at the University of Oxford, told this news organization. She noted, however, that plans for a phase III trial have stalled due to insufficient funding.
Meanwhile, researchers from the Quadram Institute in Norwich and the University of East Anglia are conducting a pilot study to see if red light therapy can relieve symptoms of ME/CFS. Red light can be absorbed by mitochondria and is used to boost energy production. The trial will monitor patients remotely from their homes and will assess cognitive function and physical activity levels.
Gut Dysbiosis
Many studies have found that people with ME/CFS have altered gut microbiota, which suggests that changes in gut bacteria may contribute to the condition. Researchers at the Quadram Institute will thus conduct a clinical trial called RESTORE-ME to see whether fecal microbiota transplants (FMT) can treat the condition.
Rik Haagmans is a research scientist and PhD candidate at the Quadram Institute. He told this news organization: “Our FMT studies, if effective, could provide a longer lasting or even permanent relief of ME/CFS, as restoring the gut microbial composition wouldn’t require continuous medication,” he said.
Biobank and Biomarkers
Europe’s first ME/CFS-specific biobank is in the UK and is called UKMEB. It now has more than 30,000 blood samples from patients with ME/CFS, multiple sclerosis, and healthy controls. Uniquely, it includes samples from people with ME/CFS who are house- and bed-bound. Caroline Kingdon, RN, MSc, a research fellow and biobank lead at the London School of Hygiene and Tropical Medicine, told this news organization that samples and data from the UKMEB have been provided to research groups all over the world and have contributed to widely cited literature.
One group making use of these samples is led by Fatima Labeed, PhD, senior lecturer in human biology at the University of Surrey. Dr. Labeed and her team are developing a diagnostic test for ME/CFS based on electrical properties in white blood cells.
“To date, studies of ME/CFS have focused on the biochemical behavior of cells: the amount and type of proteins that cells use. We have taken a different approach, studying the electrical properties,” she explained to this news organization.
Her research builds on initial observations from 2019 that found differences in the electrical impedance of white blood cells between people with ME/CFS and controls. While the biological implications remain unknown, the findings may represent a biomarker for the condition.
Using blood samples from the UKMEB, the researchers are now investigating this potential biomarker with improved techniques and a larger patient cohort, including those with mild/moderate and severe forms of ME/CFS. So far, they have received more than 100 blood samples and have analyzed the electrical properties of 42.
“Based on the results we have so far, we are very close to having a biomarker for diagnosis. Our results so far show a high degree of accuracy and are able to distinguish between ME/CFS and other diseases,” said Dr. Labeed.
Genetic Test
Another promising avenue for diagnostics comes from a research team at the University of Edinburgh led by Professor Chris Ponting at the university’s Institute of Genetics and Cancer. They are currently working on DecodeMe, a large genetic study of ME using data from more than 26,000 people.
“We are studying blood-based biomarkers that distinguish people with ME from population controls. We’ve found a large number — including some found previously in other studies — and are writing these results up for publication,” said Ponting. The results should be published in early 2025.
The Future
While research into ME/CFS has picked up pace in recent years, funding remains a key bottleneck.
“Over the last 10 years, only £8.05m has been spent on ME research,” Sonya Chowdhury, chief executive of UK charity Action for ME told this news organization. She believes this amount is not equitably comparable to research funding allocated to other diseases.
In 2022, the UK government announced its intention to develop a cross-government interim delivery plan on ME/CFS for England, however publication of the final plan has been delayed numerous times.
Dr. Shepherd agreed that increased funding is crucial for progress to be made. He said the biggest help to ME/CFS research would be to end the disparity in government research funding for the disease, and match what is given for many other disabling long-term conditions.
“It’s not fair to continue to rely on the charity sector to fund almost all of the biomedical research into ME/CFS here in the UK,” he said.
A version of this article appeared on Medscape.com.
Almost 10% of Infected Pregnant People Develop Long COVID
Almost 1 in 10 pregnant people infected with COVID-19 end up developing long COVID, according to a study published in Obstetrics & Gynecology.
Researchers at University of Utah Health looked at the medical records of more than 1500 people who got COVID-19 while pregnant and checked their self-reported symptoms at least 6 months after infection, according to a news release from the school.
The scientists discovered that 9.3% of those people reported long COVID symptoms, such as fatigue and issues in their gut.
To make sure those long COVID symptoms were not actually symptoms of pregnancy, the research team did a second analysis of people who reported symptoms more than 12 weeks after giving birth. The risk of long COVID was about the same as in the first analysis.
“It was surprising to me that the prevalence was that high,” Torri D. Metz, MD, vice chair for research of obstetrics and gynecology at the school and co-leader of the study, said in the release. “This is something that does continue to affect otherwise reasonably healthy and young populations.”
The school said this is the first study to look at long COVID risks in pregnant people. Previous research found other dangers for pregnant people who get COVID, such as a higher chance of hospitalization or death, or complications such as preterm birth.
In the general population, research shows that 10%-20% of people who get COVID develop long COVID.
Dr. Metz said healthcare providers need to remain alert about long COVID, including in pregnant people.
“We need to have this on our radar as we’re seeing patients. It’s something we really don’t want to miss. And we want to get people referred to appropriate specialists who treat long COVID,” she said.
A version of this article first appeared on WebMD.com.
Almost 1 in 10 pregnant people infected with COVID-19 end up developing long COVID, according to a study published in Obstetrics & Gynecology.
Researchers at University of Utah Health looked at the medical records of more than 1500 people who got COVID-19 while pregnant and checked their self-reported symptoms at least 6 months after infection, according to a news release from the school.
The scientists discovered that 9.3% of those people reported long COVID symptoms, such as fatigue and issues in their gut.
To make sure those long COVID symptoms were not actually symptoms of pregnancy, the research team did a second analysis of people who reported symptoms more than 12 weeks after giving birth. The risk of long COVID was about the same as in the first analysis.
“It was surprising to me that the prevalence was that high,” Torri D. Metz, MD, vice chair for research of obstetrics and gynecology at the school and co-leader of the study, said in the release. “This is something that does continue to affect otherwise reasonably healthy and young populations.”
The school said this is the first study to look at long COVID risks in pregnant people. Previous research found other dangers for pregnant people who get COVID, such as a higher chance of hospitalization or death, or complications such as preterm birth.
In the general population, research shows that 10%-20% of people who get COVID develop long COVID.
Dr. Metz said healthcare providers need to remain alert about long COVID, including in pregnant people.
“We need to have this on our radar as we’re seeing patients. It’s something we really don’t want to miss. And we want to get people referred to appropriate specialists who treat long COVID,” she said.
A version of this article first appeared on WebMD.com.
Almost 1 in 10 pregnant people infected with COVID-19 end up developing long COVID, according to a study published in Obstetrics & Gynecology.
Researchers at University of Utah Health looked at the medical records of more than 1500 people who got COVID-19 while pregnant and checked their self-reported symptoms at least 6 months after infection, according to a news release from the school.
The scientists discovered that 9.3% of those people reported long COVID symptoms, such as fatigue and issues in their gut.
To make sure those long COVID symptoms were not actually symptoms of pregnancy, the research team did a second analysis of people who reported symptoms more than 12 weeks after giving birth. The risk of long COVID was about the same as in the first analysis.
“It was surprising to me that the prevalence was that high,” Torri D. Metz, MD, vice chair for research of obstetrics and gynecology at the school and co-leader of the study, said in the release. “This is something that does continue to affect otherwise reasonably healthy and young populations.”
The school said this is the first study to look at long COVID risks in pregnant people. Previous research found other dangers for pregnant people who get COVID, such as a higher chance of hospitalization or death, or complications such as preterm birth.
In the general population, research shows that 10%-20% of people who get COVID develop long COVID.
Dr. Metz said healthcare providers need to remain alert about long COVID, including in pregnant people.
“We need to have this on our radar as we’re seeing patients. It’s something we really don’t want to miss. And we want to get people referred to appropriate specialists who treat long COVID,” she said.
A version of this article first appeared on WebMD.com.
FROM OBSTETRICS & GYNECOLOGY
Cold or Flu Virus May Trigger Relapse of Long COVID
researchers have found.
In some cases, they may be experiencing what researchers call viral interference, something also experienced by people with HIV and other infections associated with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS).
Clinical studies on the issue are limited, but patients, doctors, and researchers report many people who previously had long COVID have developed recurring symptoms after consequent viral infections.
Viral persistence — where bits of virus linger in the body — and viral reactivation remain two of the leading suspects for Yale researchers. Viral activation occurs when the immune system responds to an infection by triggering a dormant virus.
Anecdotally, these flare-ups occur more commonly in patients with long COVID with autonomic dysfunction — severe dizziness when standing up — and other symptoms of ME/CFS, said Alba Azola, MD, a Johns Hopkins Medicine rehabilitation specialist in Baltimore, Maryland, who works with patients with long COVID and other “fatiguing illnesses.”
At last count, about 18% of those surveyed by the Centers for Disease Control and Prevention said they had experienced long COVID. Nearly 60% of those surveyed said they had contracted COVID-19 at least once.
Dr. Azola said that very afternoon she had seen a patient with the flu and a recurrence of previous long COVID symptoms. Not much data exist about cases like this.
“I can’t say there is a specific study looking at this, but anecdotally, we see it all the time,” Dr. Azola said.
She has not seen completely different symptoms; more commonly, she sees a flare-up of previously existing symptoms.
David Putrino, PhD, is director of rehabilitation innovation for the Mount Sinai Health System in New York City. He treats and studies patients with long COVID and echoes what others have seen.
Patients can “recover (or feel recovered) from long COVID until the next immune challenge — another COVID infection, flu infection, pregnancy, food poisoning (all examples we have seen in the clinic) — and experience a significant flare-up of your initial COVID infection,” he said.
“Relapse” is a better term than reinfection, said Jeffrey Parsonnet, MD, an infectious diseases specialist and director of the Dartmouth Hitchcock Post-Acute COVID Syndrome Clinic, Lebanon, New Hampshire.
“We see patients who had COVID-19 followed by long COVID who then get better — either completely or mostly better. Then they’ve gotten COVID again, and this is followed by recurrence of long COVID symptoms,” he said.
“Every patient looks different in terms of what gets better and how quickly. And again, some patients are not better (or even minimally so) after a couple of years,” he said.
Patients Tell Their Stories
On the COVID-19 Long Haulers Support Facebook group, many of the 100,000 followers ask about viral reactivation. Delainne “Laney” Bond, RN, who has battled postinfection chronic illness herself, runs the Facebook group. From what she sees, “each time a person is infected or reinfected with SARS-CoV-2, they have a risk of developing long COVID or experiencing worse long COVID. Multiple infections can lead to progressive health complications.”
The posts on her site include many queries about reinfections. A post from December included nearly 80 comments with people describing the full range of symptoms. Some stories relayed how the reinfection symptoms were short lived. Some report returning to their baseline — not completely symptom free but improved.
Doctors and patients say long COVID comes and goes — relapsing-remitting — and shares many features with other complex multisystem chronic conditions, according to a new National Academy of Sciences report. Those include ME/CFS and the Epstein-Barr virus.
As far as how to treat, Dr. Putrino is one of the clinical researchers testing antivirals. One is Paxlovid; the others are drugs developed for the AIDS virus.
“A plausible mechanism for long COVID is persistence of the SARS-CoV-2 virus in tissue and/or the reactivation of latent pathogens,” according to an explanation of the research on the PolyBio Institute website, which is involved with the research.
In the meantime, “long COVID appears to be a chronic condition with few patients achieving full remission,” according to a new Academy of Sciences report. The report concludes that long COVID recovery can plateau at 6-12 months. They also note that 18%-22% of people who have long COVID symptoms at 5 months are still ill at 1 year.
A version of this article first appeared on Medscape.com.
researchers have found.
In some cases, they may be experiencing what researchers call viral interference, something also experienced by people with HIV and other infections associated with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS).
Clinical studies on the issue are limited, but patients, doctors, and researchers report many people who previously had long COVID have developed recurring symptoms after consequent viral infections.
Viral persistence — where bits of virus linger in the body — and viral reactivation remain two of the leading suspects for Yale researchers. Viral activation occurs when the immune system responds to an infection by triggering a dormant virus.
Anecdotally, these flare-ups occur more commonly in patients with long COVID with autonomic dysfunction — severe dizziness when standing up — and other symptoms of ME/CFS, said Alba Azola, MD, a Johns Hopkins Medicine rehabilitation specialist in Baltimore, Maryland, who works with patients with long COVID and other “fatiguing illnesses.”
At last count, about 18% of those surveyed by the Centers for Disease Control and Prevention said they had experienced long COVID. Nearly 60% of those surveyed said they had contracted COVID-19 at least once.
Dr. Azola said that very afternoon she had seen a patient with the flu and a recurrence of previous long COVID symptoms. Not much data exist about cases like this.
“I can’t say there is a specific study looking at this, but anecdotally, we see it all the time,” Dr. Azola said.
She has not seen completely different symptoms; more commonly, she sees a flare-up of previously existing symptoms.
David Putrino, PhD, is director of rehabilitation innovation for the Mount Sinai Health System in New York City. He treats and studies patients with long COVID and echoes what others have seen.
Patients can “recover (or feel recovered) from long COVID until the next immune challenge — another COVID infection, flu infection, pregnancy, food poisoning (all examples we have seen in the clinic) — and experience a significant flare-up of your initial COVID infection,” he said.
“Relapse” is a better term than reinfection, said Jeffrey Parsonnet, MD, an infectious diseases specialist and director of the Dartmouth Hitchcock Post-Acute COVID Syndrome Clinic, Lebanon, New Hampshire.
“We see patients who had COVID-19 followed by long COVID who then get better — either completely or mostly better. Then they’ve gotten COVID again, and this is followed by recurrence of long COVID symptoms,” he said.
“Every patient looks different in terms of what gets better and how quickly. And again, some patients are not better (or even minimally so) after a couple of years,” he said.
Patients Tell Their Stories
On the COVID-19 Long Haulers Support Facebook group, many of the 100,000 followers ask about viral reactivation. Delainne “Laney” Bond, RN, who has battled postinfection chronic illness herself, runs the Facebook group. From what she sees, “each time a person is infected or reinfected with SARS-CoV-2, they have a risk of developing long COVID or experiencing worse long COVID. Multiple infections can lead to progressive health complications.”
The posts on her site include many queries about reinfections. A post from December included nearly 80 comments with people describing the full range of symptoms. Some stories relayed how the reinfection symptoms were short lived. Some report returning to their baseline — not completely symptom free but improved.
Doctors and patients say long COVID comes and goes — relapsing-remitting — and shares many features with other complex multisystem chronic conditions, according to a new National Academy of Sciences report. Those include ME/CFS and the Epstein-Barr virus.
As far as how to treat, Dr. Putrino is one of the clinical researchers testing antivirals. One is Paxlovid; the others are drugs developed for the AIDS virus.
“A plausible mechanism for long COVID is persistence of the SARS-CoV-2 virus in tissue and/or the reactivation of latent pathogens,” according to an explanation of the research on the PolyBio Institute website, which is involved with the research.
In the meantime, “long COVID appears to be a chronic condition with few patients achieving full remission,” according to a new Academy of Sciences report. The report concludes that long COVID recovery can plateau at 6-12 months. They also note that 18%-22% of people who have long COVID symptoms at 5 months are still ill at 1 year.
A version of this article first appeared on Medscape.com.
researchers have found.
In some cases, they may be experiencing what researchers call viral interference, something also experienced by people with HIV and other infections associated with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS).
Clinical studies on the issue are limited, but patients, doctors, and researchers report many people who previously had long COVID have developed recurring symptoms after consequent viral infections.
Viral persistence — where bits of virus linger in the body — and viral reactivation remain two of the leading suspects for Yale researchers. Viral activation occurs when the immune system responds to an infection by triggering a dormant virus.
Anecdotally, these flare-ups occur more commonly in patients with long COVID with autonomic dysfunction — severe dizziness when standing up — and other symptoms of ME/CFS, said Alba Azola, MD, a Johns Hopkins Medicine rehabilitation specialist in Baltimore, Maryland, who works with patients with long COVID and other “fatiguing illnesses.”
At last count, about 18% of those surveyed by the Centers for Disease Control and Prevention said they had experienced long COVID. Nearly 60% of those surveyed said they had contracted COVID-19 at least once.
Dr. Azola said that very afternoon she had seen a patient with the flu and a recurrence of previous long COVID symptoms. Not much data exist about cases like this.
“I can’t say there is a specific study looking at this, but anecdotally, we see it all the time,” Dr. Azola said.
She has not seen completely different symptoms; more commonly, she sees a flare-up of previously existing symptoms.
David Putrino, PhD, is director of rehabilitation innovation for the Mount Sinai Health System in New York City. He treats and studies patients with long COVID and echoes what others have seen.
Patients can “recover (or feel recovered) from long COVID until the next immune challenge — another COVID infection, flu infection, pregnancy, food poisoning (all examples we have seen in the clinic) — and experience a significant flare-up of your initial COVID infection,” he said.
“Relapse” is a better term than reinfection, said Jeffrey Parsonnet, MD, an infectious diseases specialist and director of the Dartmouth Hitchcock Post-Acute COVID Syndrome Clinic, Lebanon, New Hampshire.
“We see patients who had COVID-19 followed by long COVID who then get better — either completely or mostly better. Then they’ve gotten COVID again, and this is followed by recurrence of long COVID symptoms,” he said.
“Every patient looks different in terms of what gets better and how quickly. And again, some patients are not better (or even minimally so) after a couple of years,” he said.
Patients Tell Their Stories
On the COVID-19 Long Haulers Support Facebook group, many of the 100,000 followers ask about viral reactivation. Delainne “Laney” Bond, RN, who has battled postinfection chronic illness herself, runs the Facebook group. From what she sees, “each time a person is infected or reinfected with SARS-CoV-2, they have a risk of developing long COVID or experiencing worse long COVID. Multiple infections can lead to progressive health complications.”
The posts on her site include many queries about reinfections. A post from December included nearly 80 comments with people describing the full range of symptoms. Some stories relayed how the reinfection symptoms were short lived. Some report returning to their baseline — not completely symptom free but improved.
Doctors and patients say long COVID comes and goes — relapsing-remitting — and shares many features with other complex multisystem chronic conditions, according to a new National Academy of Sciences report. Those include ME/CFS and the Epstein-Barr virus.
As far as how to treat, Dr. Putrino is one of the clinical researchers testing antivirals. One is Paxlovid; the others are drugs developed for the AIDS virus.
“A plausible mechanism for long COVID is persistence of the SARS-CoV-2 virus in tissue and/or the reactivation of latent pathogens,” according to an explanation of the research on the PolyBio Institute website, which is involved with the research.
In the meantime, “long COVID appears to be a chronic condition with few patients achieving full remission,” according to a new Academy of Sciences report. The report concludes that long COVID recovery can plateau at 6-12 months. They also note that 18%-22% of people who have long COVID symptoms at 5 months are still ill at 1 year.
A version of this article first appeared on Medscape.com.
Long COVID Can’t Be Solved Until We Decide What It Is
This transcript has been edited for clarity.
I want to help people suffering from long COVID as much as anyone. But we have a real problem. In brief, we are being too inclusive. The first thing you learn, when you start studying the epidemiology of diseases, is that you need a good case definition. And our case definition for long COVID sucks. Just last week, the National Academies of Sciences, Engineering, and Medicine (NASEM) issued a definition of long COVID with the aim of “improving consistency, documentation, and treatment.” Good news, right? Here’s the definition: “Long COVID is an infection-associated chronic condition that occurs after SARS-CoV-2 infection and is present for at least 3 months as a continuous, relapsing and remitting, or progressive disease state that affects one or more organ systems.”
This is not helpful. The symptoms can be in any organ system, can be continuous or relapsing and remitting. Basically, if you’ve had COVID — and essentially all of us have by now — and you have any symptom, even one that comes and goes, 3 months after that, it’s long COVID. They don’t even specify that it has to be a new symptom.
And I have sort of a case study in this problem today, based on a paper getting a lot of press suggesting that one out of every five people has long COVID.
We are talking about this study, “Epidemiologic Features of Recovery From SARS-CoV-2 Infection,” appearing in JAMA Network Open this week. While I think the idea is important, the study really highlights why it can be so hard to study long COVID.
As part of efforts to understand long COVID, the National Institutes of Health (NIH) leveraged 14 of its ongoing cohort studies. The NIH has multiple longitudinal cohort studies that follow various groups of people over time. You may have heard of the REGARDS study, for example, which focuses on cardiovascular risks to people living in the southern United States. Or the ARIC study, which followed adults in four communities across the United States for the development of heart disease. All 14 of the cohorts in this study are long-running projects with ongoing data collection. So, it was not a huge lift to add some questions to the yearly surveys and studies the participants were already getting.
To wit: “Do you think that you have had COVID-19?” and “Would you say that you are completely recovered now?” Those who said they weren’t fully recovered were asked how long it had been since their infection, and anyone who answered with a duration > 90 days was considered to have long COVID.
So, we have self-report of infection, self-report of duration of symptoms, and self-report of recovery. This is fine, of course; individuals’ perceptions of their own health are meaningful. But the vagaries inherent in those perceptions are going to muddy the waters as we attempt to discover the true nature of the long COVID syndrome.
But let’s look at some results. Out of 4708 individuals studied, 842 (17.9%) had not recovered by 90 days.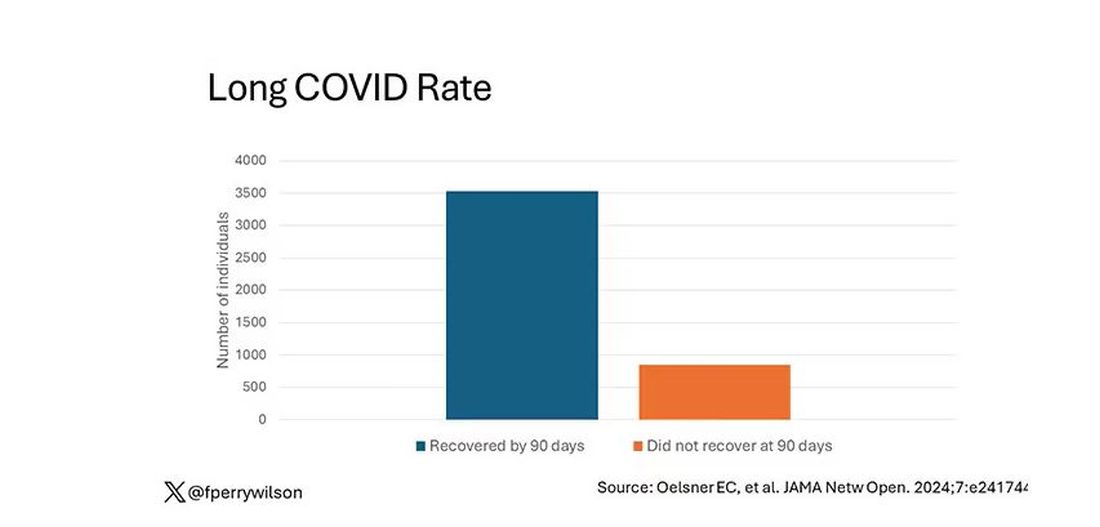
This study included not only people hospitalized with COVID, as some prior long COVID studies did, but people self-diagnosed, tested at home, etc. This estimate is as reflective of the broader US population as we can get.
And there are some interesting trends here.
Recovery time was longer in the first waves of COVID than in the Omicron wave.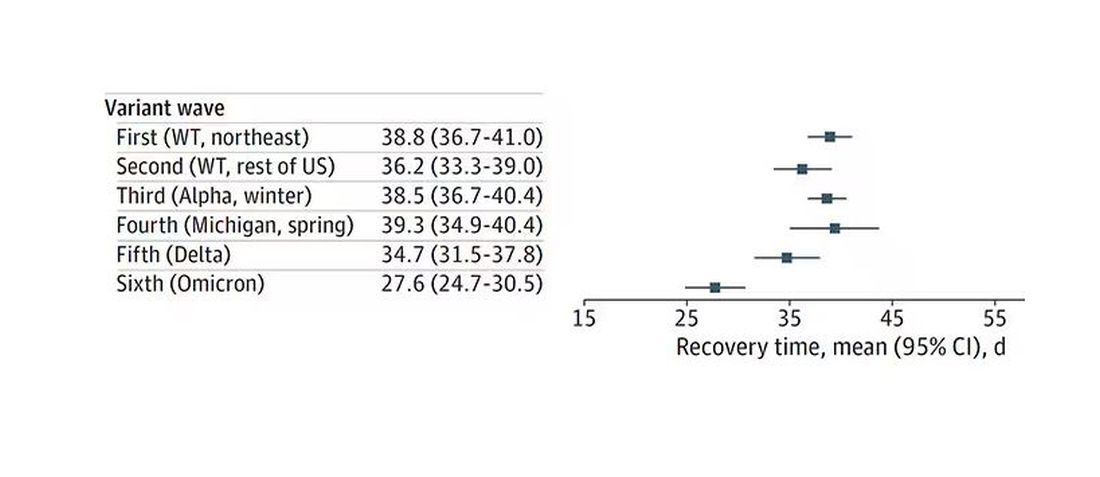
Recovery times were longer for smokers, those with diabetes, and those who were obese.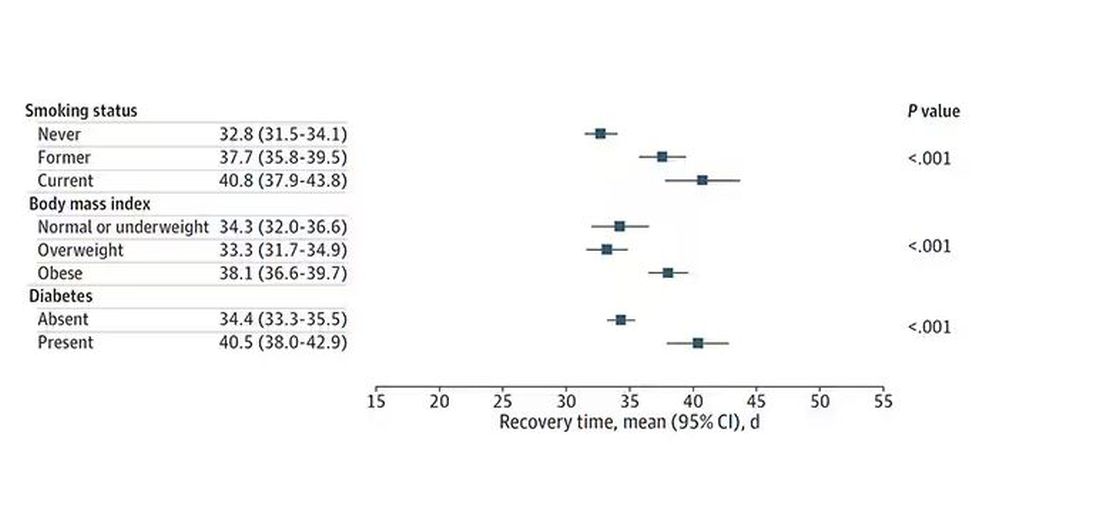
Recovery times were longer if the disease was more severe, in general. Though there is an unusual finding that women had longer recovery times despite their lower average severity of illness.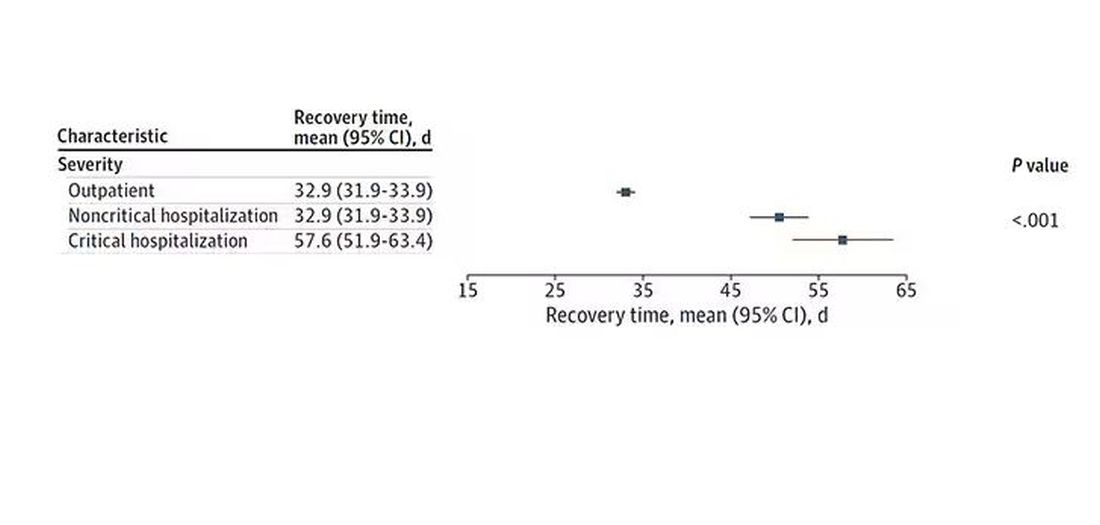
Vaccination was associated with shorter recovery times, as you can see here. 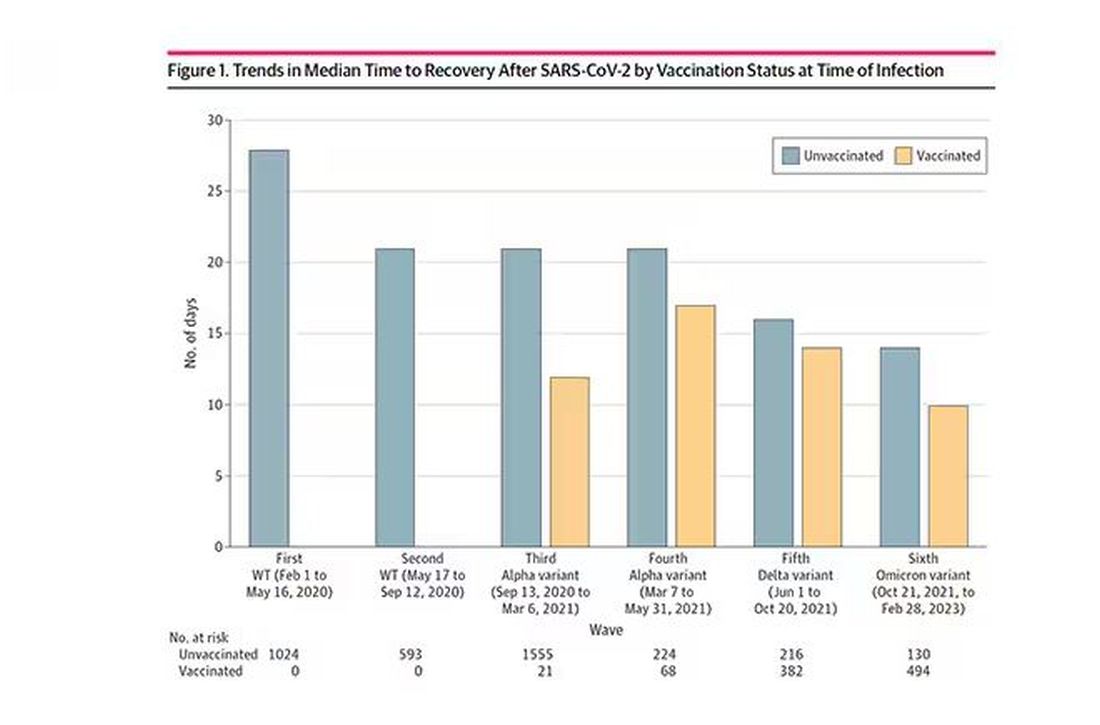
This is all quite interesting. It’s clear that people feel they are sick for a while after COVID. But we need to understand whether these symptoms are due to the lingering effects of a bad infection that knocks you down a peg, or to an ongoing syndrome — this thing we call long COVID — that has a physiologic basis and thus can be treated. And this study doesn’t help us much with that.
Not that this was the authors’ intention. This is a straight-up epidemiology study. But the problem is deeper than that. Let’s imagine that you want to really dig into this long COVID thing and get blood samples from people with it, ideally from controls with some other respiratory virus infection, and do all kinds of genetic and proteomic studies and stuff to really figure out what’s going on. Who do you enroll to be in the long COVID group? Do you enroll anyone who says they had COVID and still has some symptom more than 90 days after? You are going to find an awful lot of eligible people, and I guarantee that if there is a pathognomonic signature of long COVID, not all of them will have it.
And what about other respiratory viruses? This study in The Lancet Infectious Diseases compared long-term outcomes among hospitalized patients with COVID vs influenza. In general, the COVID outcomes are worse, but let’s not knock the concept of “long flu.” Across the board, roughly 50% of people report symptoms across any given organ system.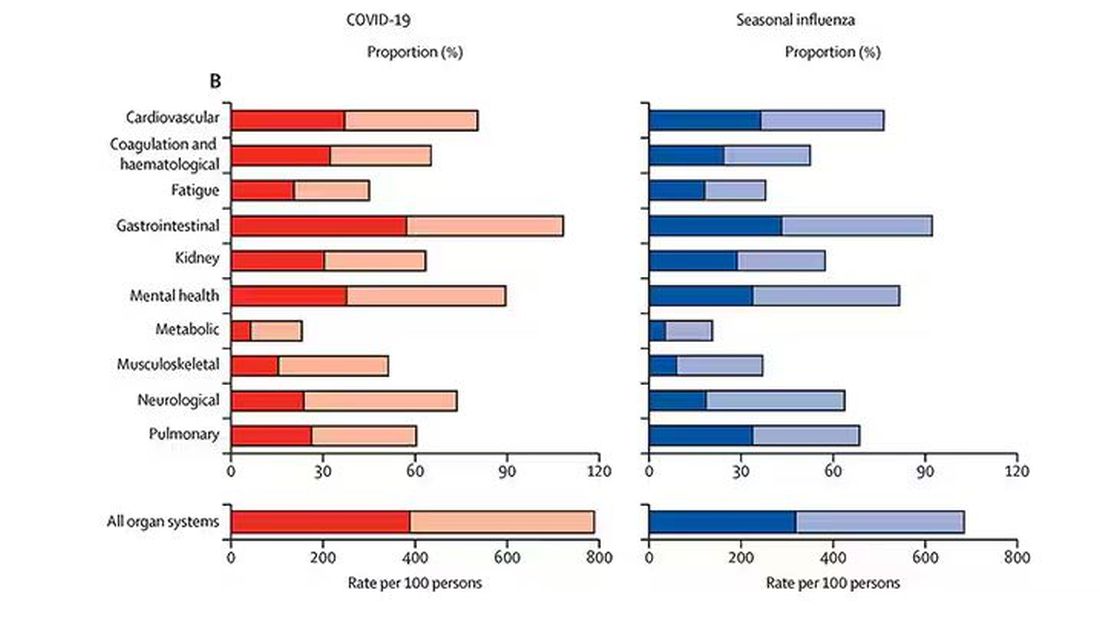
What this is all about is something called misclassification bias, a form of information bias that arises in a study where you label someone as diseased when they are not, or vice versa. If this happens at random, it’s bad; you’ve lost your ability to distinguish characteristics from the diseased and nondiseased population.
When it’s not random, it’s really bad. If we are more likely to misclassify women as having long COVID, for example, then it will appear that long COVID is more likely among women, or more likely among those with higher estrogen levels, or something. And that might simply be wrong.
I’m not saying that’s what happened here; this study does a really great job of what it set out to do, which was to describe the patterns of lingering symptoms after COVID. But we are not going to make progress toward understanding long COVID until we are less inclusive with our case definition. To paraphrase Syndrome from The Incredibles: If everyone has long COVID, then no one does.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I want to help people suffering from long COVID as much as anyone. But we have a real problem. In brief, we are being too inclusive. The first thing you learn, when you start studying the epidemiology of diseases, is that you need a good case definition. And our case definition for long COVID sucks. Just last week, the National Academies of Sciences, Engineering, and Medicine (NASEM) issued a definition of long COVID with the aim of “improving consistency, documentation, and treatment.” Good news, right? Here’s the definition: “Long COVID is an infection-associated chronic condition that occurs after SARS-CoV-2 infection and is present for at least 3 months as a continuous, relapsing and remitting, or progressive disease state that affects one or more organ systems.”
This is not helpful. The symptoms can be in any organ system, can be continuous or relapsing and remitting. Basically, if you’ve had COVID — and essentially all of us have by now — and you have any symptom, even one that comes and goes, 3 months after that, it’s long COVID. They don’t even specify that it has to be a new symptom.
And I have sort of a case study in this problem today, based on a paper getting a lot of press suggesting that one out of every five people has long COVID.
We are talking about this study, “Epidemiologic Features of Recovery From SARS-CoV-2 Infection,” appearing in JAMA Network Open this week. While I think the idea is important, the study really highlights why it can be so hard to study long COVID.
As part of efforts to understand long COVID, the National Institutes of Health (NIH) leveraged 14 of its ongoing cohort studies. The NIH has multiple longitudinal cohort studies that follow various groups of people over time. You may have heard of the REGARDS study, for example, which focuses on cardiovascular risks to people living in the southern United States. Or the ARIC study, which followed adults in four communities across the United States for the development of heart disease. All 14 of the cohorts in this study are long-running projects with ongoing data collection. So, it was not a huge lift to add some questions to the yearly surveys and studies the participants were already getting.
To wit: “Do you think that you have had COVID-19?” and “Would you say that you are completely recovered now?” Those who said they weren’t fully recovered were asked how long it had been since their infection, and anyone who answered with a duration > 90 days was considered to have long COVID.
So, we have self-report of infection, self-report of duration of symptoms, and self-report of recovery. This is fine, of course; individuals’ perceptions of their own health are meaningful. But the vagaries inherent in those perceptions are going to muddy the waters as we attempt to discover the true nature of the long COVID syndrome.
But let’s look at some results. Out of 4708 individuals studied, 842 (17.9%) had not recovered by 90 days.
This study included not only people hospitalized with COVID, as some prior long COVID studies did, but people self-diagnosed, tested at home, etc. This estimate is as reflective of the broader US population as we can get.
And there are some interesting trends here.
Recovery time was longer in the first waves of COVID than in the Omicron wave.
Recovery times were longer for smokers, those with diabetes, and those who were obese.
Recovery times were longer if the disease was more severe, in general. Though there is an unusual finding that women had longer recovery times despite their lower average severity of illness.
Vaccination was associated with shorter recovery times, as you can see here. 
This is all quite interesting. It’s clear that people feel they are sick for a while after COVID. But we need to understand whether these symptoms are due to the lingering effects of a bad infection that knocks you down a peg, or to an ongoing syndrome — this thing we call long COVID — that has a physiologic basis and thus can be treated. And this study doesn’t help us much with that.
Not that this was the authors’ intention. This is a straight-up epidemiology study. But the problem is deeper than that. Let’s imagine that you want to really dig into this long COVID thing and get blood samples from people with it, ideally from controls with some other respiratory virus infection, and do all kinds of genetic and proteomic studies and stuff to really figure out what’s going on. Who do you enroll to be in the long COVID group? Do you enroll anyone who says they had COVID and still has some symptom more than 90 days after? You are going to find an awful lot of eligible people, and I guarantee that if there is a pathognomonic signature of long COVID, not all of them will have it.
And what about other respiratory viruses? This study in The Lancet Infectious Diseases compared long-term outcomes among hospitalized patients with COVID vs influenza. In general, the COVID outcomes are worse, but let’s not knock the concept of “long flu.” Across the board, roughly 50% of people report symptoms across any given organ system.
What this is all about is something called misclassification bias, a form of information bias that arises in a study where you label someone as diseased when they are not, or vice versa. If this happens at random, it’s bad; you’ve lost your ability to distinguish characteristics from the diseased and nondiseased population.
When it’s not random, it’s really bad. If we are more likely to misclassify women as having long COVID, for example, then it will appear that long COVID is more likely among women, or more likely among those with higher estrogen levels, or something. And that might simply be wrong.
I’m not saying that’s what happened here; this study does a really great job of what it set out to do, which was to describe the patterns of lingering symptoms after COVID. But we are not going to make progress toward understanding long COVID until we are less inclusive with our case definition. To paraphrase Syndrome from The Incredibles: If everyone has long COVID, then no one does.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I want to help people suffering from long COVID as much as anyone. But we have a real problem. In brief, we are being too inclusive. The first thing you learn, when you start studying the epidemiology of diseases, is that you need a good case definition. And our case definition for long COVID sucks. Just last week, the National Academies of Sciences, Engineering, and Medicine (NASEM) issued a definition of long COVID with the aim of “improving consistency, documentation, and treatment.” Good news, right? Here’s the definition: “Long COVID is an infection-associated chronic condition that occurs after SARS-CoV-2 infection and is present for at least 3 months as a continuous, relapsing and remitting, or progressive disease state that affects one or more organ systems.”
This is not helpful. The symptoms can be in any organ system, can be continuous or relapsing and remitting. Basically, if you’ve had COVID — and essentially all of us have by now — and you have any symptom, even one that comes and goes, 3 months after that, it’s long COVID. They don’t even specify that it has to be a new symptom.
And I have sort of a case study in this problem today, based on a paper getting a lot of press suggesting that one out of every five people has long COVID.
We are talking about this study, “Epidemiologic Features of Recovery From SARS-CoV-2 Infection,” appearing in JAMA Network Open this week. While I think the idea is important, the study really highlights why it can be so hard to study long COVID.
As part of efforts to understand long COVID, the National Institutes of Health (NIH) leveraged 14 of its ongoing cohort studies. The NIH has multiple longitudinal cohort studies that follow various groups of people over time. You may have heard of the REGARDS study, for example, which focuses on cardiovascular risks to people living in the southern United States. Or the ARIC study, which followed adults in four communities across the United States for the development of heart disease. All 14 of the cohorts in this study are long-running projects with ongoing data collection. So, it was not a huge lift to add some questions to the yearly surveys and studies the participants were already getting.
To wit: “Do you think that you have had COVID-19?” and “Would you say that you are completely recovered now?” Those who said they weren’t fully recovered were asked how long it had been since their infection, and anyone who answered with a duration > 90 days was considered to have long COVID.
So, we have self-report of infection, self-report of duration of symptoms, and self-report of recovery. This is fine, of course; individuals’ perceptions of their own health are meaningful. But the vagaries inherent in those perceptions are going to muddy the waters as we attempt to discover the true nature of the long COVID syndrome.
But let’s look at some results. Out of 4708 individuals studied, 842 (17.9%) had not recovered by 90 days.
This study included not only people hospitalized with COVID, as some prior long COVID studies did, but people self-diagnosed, tested at home, etc. This estimate is as reflective of the broader US population as we can get.
And there are some interesting trends here.
Recovery time was longer in the first waves of COVID than in the Omicron wave.
Recovery times were longer for smokers, those with diabetes, and those who were obese.
Recovery times were longer if the disease was more severe, in general. Though there is an unusual finding that women had longer recovery times despite their lower average severity of illness.
Vaccination was associated with shorter recovery times, as you can see here. 
This is all quite interesting. It’s clear that people feel they are sick for a while after COVID. But we need to understand whether these symptoms are due to the lingering effects of a bad infection that knocks you down a peg, or to an ongoing syndrome — this thing we call long COVID — that has a physiologic basis and thus can be treated. And this study doesn’t help us much with that.
Not that this was the authors’ intention. This is a straight-up epidemiology study. But the problem is deeper than that. Let’s imagine that you want to really dig into this long COVID thing and get blood samples from people with it, ideally from controls with some other respiratory virus infection, and do all kinds of genetic and proteomic studies and stuff to really figure out what’s going on. Who do you enroll to be in the long COVID group? Do you enroll anyone who says they had COVID and still has some symptom more than 90 days after? You are going to find an awful lot of eligible people, and I guarantee that if there is a pathognomonic signature of long COVID, not all of them will have it.
And what about other respiratory viruses? This study in The Lancet Infectious Diseases compared long-term outcomes among hospitalized patients with COVID vs influenza. In general, the COVID outcomes are worse, but let’s not knock the concept of “long flu.” Across the board, roughly 50% of people report symptoms across any given organ system.
What this is all about is something called misclassification bias, a form of information bias that arises in a study where you label someone as diseased when they are not, or vice versa. If this happens at random, it’s bad; you’ve lost your ability to distinguish characteristics from the diseased and nondiseased population.
When it’s not random, it’s really bad. If we are more likely to misclassify women as having long COVID, for example, then it will appear that long COVID is more likely among women, or more likely among those with higher estrogen levels, or something. And that might simply be wrong.
I’m not saying that’s what happened here; this study does a really great job of what it set out to do, which was to describe the patterns of lingering symptoms after COVID. But we are not going to make progress toward understanding long COVID until we are less inclusive with our case definition. To paraphrase Syndrome from The Incredibles: If everyone has long COVID, then no one does.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
National Academies Issue New Broad Definition of Long COVID
A new broadly inclusive definition of long COVID from the National Academies of Sciences, Engineering, and Medicine (NASEM) has been developed with the aim of improving consistency, documentation, and treatment for both adults and children.
According to the 2024 NASEM definition of long COVID issued on June 11, 2024, “Long COVID is an infection-associated chronic condition that occurs after SARS-CoV-2 infection and is present for at least 3 months as a continuous, relapsing and remitting, or progressive disease state that affects one or more organ systems.”
People with long COVID may present with one or more of a long list of symptoms, such as shortness of breath, rapid heartbeat, extreme fatigue, post-exertional malaise, or sleep disturbance and with single or multiple diagnosable conditions, including interstitial lung disease, arrhythmias, postural orthostatic tachycardia syndrome (POTS), myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), diabetes, or autoimmune disorders. The condition can exacerbate preexisting health conditions or present as new ones.
The definition does not require laboratory confirmation or other proof of initial infection. Long COVID can follow SARS-CoV-2 infection of any severity, including asymptomatic infections, whether or not they were initially recognized.
Several working definitions and terms for long COVID had previously been proposed, including those from the World Health Organization (WHO) and the US Centers for Disease Control and Prevention, but no common definition or terminology had been established.
The new definition was developed at the request of the Administration for Strategic Preparedness and Response and the Office of the Assistant Secretary for Health (OASH). It was written by a multi-stakeholder panel convened by NASEM, which recommended that the new definition be universally adopted by the federal government, clinical societies and associations, public health practitioners, clinicians, payers, the drug industry, and others using the term long COVID.
Recent surveys suggest that approximately 7% of Americans have experienced or are experiencing long COVID. “It’s millions of people,” panel chair Harvey V. Fineberg, MD, president of the Gordon and Betty Moore Foundation, told this news organization.
The new definition “does not erase the problem of clinical judgment ... But we think this definition has the real advantage of elevating to the clinician’s mind the real likelihood in the current environment of prevalence of this virus that a presenting patient’s strange symptoms are both real and maybe related as an expression of long COVID,” Dr. Fineberg noted.
One way this new definition differs from previous ones such as WHO’s, he said, is “they talk about a diagnosis of exclusion. One of the important points in our definition is that other diagnosable conditions like ME/CFS or POTS can be part of the picture of long COVID. They are not alternative. They are, in fact, an expression of long COVID.”
Indeed, the NASEM report also introduces the term infection-associated chronic condition (IACC). This was important, Dr. Fineberg said, “because it’s the larger family of conditions of which long COVID is a part. It emphasizes a relatedness of long COVID to other conditions that can follow from a variety of infections. We also adopted the term ‘disease state’ to convey the seriousness and reality of this condition in the lives of patients.”
Comments on New Definition
In a statement provided to this news organization, Lucinda Bateman, MD, and Brayden Yellman, MD, co-medical directors of the Bateman-Horne Center in Salt Lake City, said that “describing long COVID as an IACC ... not only meets the NASEM goal of allowing clinicians, researchers, and public health officials to meaningfully identify and serve all persons who suffer illness or disability in the wake of a SARS-CoV-2 infection, but also draws direct comparison to other known IACC’s (such as ME/CFS, post-treatment Lyme, POTS) that have been plaguing many for decades.”
Dr. Fineberg noted another important aspect of the NASEM report: “Our definition includes an explicit statement on equity, explaining that long COVID can affect anyone, young and old, different races, different ages, different sexes, different genders, different orientations, different socioeconomic conditions ... This does not mean that every single person is at equal risk. There are risk factors, but the important point is the universal nature of this as a condition.”
Two clinical directors of long COVID programs who were contacted by this news organization praised the new definition. Zijian Chen, MD, director of Mount Sinai’s Center for Post-COVID Care, New York, said that it’s “very similar to the definition that we have used for our clinical practice since 2020. It is very important that the broad definition helps to be inclusive of all patients that may be affected. The inclusion of children as a consideration is important as well, since there is routinely less focus on children because they tend to have less disease frequency ... The creation of a unified definition helps both with clinical practice and research.”
Nisha Viswanathan, MD, director of the long COVID program at the University of California, Los Angeles, said: “I think they left it intentionally broad for the medical practitioner to not necessarily use the definition to rule out individuals, but to perhaps use more of a clinical gestalt to help rule in this diagnosis ... I think this definition is providing clarity to health care providers on what exactly would be falling under the long-COVID diagnosis header.”
Dr. Viswanathan also said that she anticipates this definition to help patients make their case in filing disability claims. “Because long COVID has not previously had a good fleshed-out definition, it was very easy for disability providers to reject claims for patients who continue to have symptoms ... I actually think this might help our patients ultimately in their attempt to be able to have the ability to care for themselves when they’re disabled enough to not be able to work.”
Written into the report is the expectation that the definition “will evolve as new evidence emerges and the understanding of long COVID matures.” The writing committee calls for reexamination in “no more than 3 years.” Factors that would prompt a reevaluation could include improved testing methods, discovery of medical factors and/or biomarkers that distinguish long COVID from other conditions, and new treatments.
Meanwhile, Dr. Fineberg told this news organization, “If this definition adds to the readiness, awareness, openness, and response to the patient with long COVID, it will have done its job.”
Dr. Fineberg, Dr. Bateman, Dr. Yellman, Dr. Viswanathan, and Dr. Chen have no relevant disclosures.
A version of this article appeared on Medscape.com.
A new broadly inclusive definition of long COVID from the National Academies of Sciences, Engineering, and Medicine (NASEM) has been developed with the aim of improving consistency, documentation, and treatment for both adults and children.
According to the 2024 NASEM definition of long COVID issued on June 11, 2024, “Long COVID is an infection-associated chronic condition that occurs after SARS-CoV-2 infection and is present for at least 3 months as a continuous, relapsing and remitting, or progressive disease state that affects one or more organ systems.”
People with long COVID may present with one or more of a long list of symptoms, such as shortness of breath, rapid heartbeat, extreme fatigue, post-exertional malaise, or sleep disturbance and with single or multiple diagnosable conditions, including interstitial lung disease, arrhythmias, postural orthostatic tachycardia syndrome (POTS), myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), diabetes, or autoimmune disorders. The condition can exacerbate preexisting health conditions or present as new ones.
The definition does not require laboratory confirmation or other proof of initial infection. Long COVID can follow SARS-CoV-2 infection of any severity, including asymptomatic infections, whether or not they were initially recognized.
Several working definitions and terms for long COVID had previously been proposed, including those from the World Health Organization (WHO) and the US Centers for Disease Control and Prevention, but no common definition or terminology had been established.
The new definition was developed at the request of the Administration for Strategic Preparedness and Response and the Office of the Assistant Secretary for Health (OASH). It was written by a multi-stakeholder panel convened by NASEM, which recommended that the new definition be universally adopted by the federal government, clinical societies and associations, public health practitioners, clinicians, payers, the drug industry, and others using the term long COVID.
Recent surveys suggest that approximately 7% of Americans have experienced or are experiencing long COVID. “It’s millions of people,” panel chair Harvey V. Fineberg, MD, president of the Gordon and Betty Moore Foundation, told this news organization.
The new definition “does not erase the problem of clinical judgment ... But we think this definition has the real advantage of elevating to the clinician’s mind the real likelihood in the current environment of prevalence of this virus that a presenting patient’s strange symptoms are both real and maybe related as an expression of long COVID,” Dr. Fineberg noted.
One way this new definition differs from previous ones such as WHO’s, he said, is “they talk about a diagnosis of exclusion. One of the important points in our definition is that other diagnosable conditions like ME/CFS or POTS can be part of the picture of long COVID. They are not alternative. They are, in fact, an expression of long COVID.”
Indeed, the NASEM report also introduces the term infection-associated chronic condition (IACC). This was important, Dr. Fineberg said, “because it’s the larger family of conditions of which long COVID is a part. It emphasizes a relatedness of long COVID to other conditions that can follow from a variety of infections. We also adopted the term ‘disease state’ to convey the seriousness and reality of this condition in the lives of patients.”
Comments on New Definition
In a statement provided to this news organization, Lucinda Bateman, MD, and Brayden Yellman, MD, co-medical directors of the Bateman-Horne Center in Salt Lake City, said that “describing long COVID as an IACC ... not only meets the NASEM goal of allowing clinicians, researchers, and public health officials to meaningfully identify and serve all persons who suffer illness or disability in the wake of a SARS-CoV-2 infection, but also draws direct comparison to other known IACC’s (such as ME/CFS, post-treatment Lyme, POTS) that have been plaguing many for decades.”
Dr. Fineberg noted another important aspect of the NASEM report: “Our definition includes an explicit statement on equity, explaining that long COVID can affect anyone, young and old, different races, different ages, different sexes, different genders, different orientations, different socioeconomic conditions ... This does not mean that every single person is at equal risk. There are risk factors, but the important point is the universal nature of this as a condition.”
Two clinical directors of long COVID programs who were contacted by this news organization praised the new definition. Zijian Chen, MD, director of Mount Sinai’s Center for Post-COVID Care, New York, said that it’s “very similar to the definition that we have used for our clinical practice since 2020. It is very important that the broad definition helps to be inclusive of all patients that may be affected. The inclusion of children as a consideration is important as well, since there is routinely less focus on children because they tend to have less disease frequency ... The creation of a unified definition helps both with clinical practice and research.”
Nisha Viswanathan, MD, director of the long COVID program at the University of California, Los Angeles, said: “I think they left it intentionally broad for the medical practitioner to not necessarily use the definition to rule out individuals, but to perhaps use more of a clinical gestalt to help rule in this diagnosis ... I think this definition is providing clarity to health care providers on what exactly would be falling under the long-COVID diagnosis header.”
Dr. Viswanathan also said that she anticipates this definition to help patients make their case in filing disability claims. “Because long COVID has not previously had a good fleshed-out definition, it was very easy for disability providers to reject claims for patients who continue to have symptoms ... I actually think this might help our patients ultimately in their attempt to be able to have the ability to care for themselves when they’re disabled enough to not be able to work.”
Written into the report is the expectation that the definition “will evolve as new evidence emerges and the understanding of long COVID matures.” The writing committee calls for reexamination in “no more than 3 years.” Factors that would prompt a reevaluation could include improved testing methods, discovery of medical factors and/or biomarkers that distinguish long COVID from other conditions, and new treatments.
Meanwhile, Dr. Fineberg told this news organization, “If this definition adds to the readiness, awareness, openness, and response to the patient with long COVID, it will have done its job.”
Dr. Fineberg, Dr. Bateman, Dr. Yellman, Dr. Viswanathan, and Dr. Chen have no relevant disclosures.
A version of this article appeared on Medscape.com.
A new broadly inclusive definition of long COVID from the National Academies of Sciences, Engineering, and Medicine (NASEM) has been developed with the aim of improving consistency, documentation, and treatment for both adults and children.
According to the 2024 NASEM definition of long COVID issued on June 11, 2024, “Long COVID is an infection-associated chronic condition that occurs after SARS-CoV-2 infection and is present for at least 3 months as a continuous, relapsing and remitting, or progressive disease state that affects one or more organ systems.”
People with long COVID may present with one or more of a long list of symptoms, such as shortness of breath, rapid heartbeat, extreme fatigue, post-exertional malaise, or sleep disturbance and with single or multiple diagnosable conditions, including interstitial lung disease, arrhythmias, postural orthostatic tachycardia syndrome (POTS), myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), diabetes, or autoimmune disorders. The condition can exacerbate preexisting health conditions or present as new ones.
The definition does not require laboratory confirmation or other proof of initial infection. Long COVID can follow SARS-CoV-2 infection of any severity, including asymptomatic infections, whether or not they were initially recognized.
Several working definitions and terms for long COVID had previously been proposed, including those from the World Health Organization (WHO) and the US Centers for Disease Control and Prevention, but no common definition or terminology had been established.
The new definition was developed at the request of the Administration for Strategic Preparedness and Response and the Office of the Assistant Secretary for Health (OASH). It was written by a multi-stakeholder panel convened by NASEM, which recommended that the new definition be universally adopted by the federal government, clinical societies and associations, public health practitioners, clinicians, payers, the drug industry, and others using the term long COVID.
Recent surveys suggest that approximately 7% of Americans have experienced or are experiencing long COVID. “It’s millions of people,” panel chair Harvey V. Fineberg, MD, president of the Gordon and Betty Moore Foundation, told this news organization.
The new definition “does not erase the problem of clinical judgment ... But we think this definition has the real advantage of elevating to the clinician’s mind the real likelihood in the current environment of prevalence of this virus that a presenting patient’s strange symptoms are both real and maybe related as an expression of long COVID,” Dr. Fineberg noted.
One way this new definition differs from previous ones such as WHO’s, he said, is “they talk about a diagnosis of exclusion. One of the important points in our definition is that other diagnosable conditions like ME/CFS or POTS can be part of the picture of long COVID. They are not alternative. They are, in fact, an expression of long COVID.”
Indeed, the NASEM report also introduces the term infection-associated chronic condition (IACC). This was important, Dr. Fineberg said, “because it’s the larger family of conditions of which long COVID is a part. It emphasizes a relatedness of long COVID to other conditions that can follow from a variety of infections. We also adopted the term ‘disease state’ to convey the seriousness and reality of this condition in the lives of patients.”
Comments on New Definition
In a statement provided to this news organization, Lucinda Bateman, MD, and Brayden Yellman, MD, co-medical directors of the Bateman-Horne Center in Salt Lake City, said that “describing long COVID as an IACC ... not only meets the NASEM goal of allowing clinicians, researchers, and public health officials to meaningfully identify and serve all persons who suffer illness or disability in the wake of a SARS-CoV-2 infection, but also draws direct comparison to other known IACC’s (such as ME/CFS, post-treatment Lyme, POTS) that have been plaguing many for decades.”
Dr. Fineberg noted another important aspect of the NASEM report: “Our definition includes an explicit statement on equity, explaining that long COVID can affect anyone, young and old, different races, different ages, different sexes, different genders, different orientations, different socioeconomic conditions ... This does not mean that every single person is at equal risk. There are risk factors, but the important point is the universal nature of this as a condition.”
Two clinical directors of long COVID programs who were contacted by this news organization praised the new definition. Zijian Chen, MD, director of Mount Sinai’s Center for Post-COVID Care, New York, said that it’s “very similar to the definition that we have used for our clinical practice since 2020. It is very important that the broad definition helps to be inclusive of all patients that may be affected. The inclusion of children as a consideration is important as well, since there is routinely less focus on children because they tend to have less disease frequency ... The creation of a unified definition helps both with clinical practice and research.”
Nisha Viswanathan, MD, director of the long COVID program at the University of California, Los Angeles, said: “I think they left it intentionally broad for the medical practitioner to not necessarily use the definition to rule out individuals, but to perhaps use more of a clinical gestalt to help rule in this diagnosis ... I think this definition is providing clarity to health care providers on what exactly would be falling under the long-COVID diagnosis header.”
Dr. Viswanathan also said that she anticipates this definition to help patients make their case in filing disability claims. “Because long COVID has not previously had a good fleshed-out definition, it was very easy for disability providers to reject claims for patients who continue to have symptoms ... I actually think this might help our patients ultimately in their attempt to be able to have the ability to care for themselves when they’re disabled enough to not be able to work.”
Written into the report is the expectation that the definition “will evolve as new evidence emerges and the understanding of long COVID matures.” The writing committee calls for reexamination in “no more than 3 years.” Factors that would prompt a reevaluation could include improved testing methods, discovery of medical factors and/or biomarkers that distinguish long COVID from other conditions, and new treatments.
Meanwhile, Dr. Fineberg told this news organization, “If this definition adds to the readiness, awareness, openness, and response to the patient with long COVID, it will have done its job.”
Dr. Fineberg, Dr. Bateman, Dr. Yellman, Dr. Viswanathan, and Dr. Chen have no relevant disclosures.
A version of this article appeared on Medscape.com.
MDs’ One-Word Summary of Long COVID Progress: ‘Frustration’
Stuart Malcolm, MD, a primary care physician who practices in Oregon and northern California, started seeing patients with long COVID early in the pandemic. Back then, he was frustrated by the obstacles and lack of standard diagnostic tests and treatments. Four years later, well, he still is.
“Something I learned the last few years is the logistics to get people care is really, really hard,” he said. “There’s a lot of frustration. It’s mostly frustration.”
For long COVID doctors and patients, there has been little to no progress addressing the challenges, leaving many discouraged. Researchers and clinicians now have a greater understanding of what health agencies formally call post-COVID condition, but the wide spectrum of symptoms, slow progress in launching pharmacologic clinical trials, and the research toward understanding the underlying causes mean standardized diagnostic tests and definitive treatments remain elusive.
“The frustration is that we aren’t able to help everyone with our current knowledge base. And I think the frustration lies not just with us physicians but also with patients because they’re at the point where if they tried everything, literally everything and haven’t gotten better,” said Zijian Chen, MD, director of the Mount Sinai Center for Post-COVID Care in New York City.
Wanted: More Funding, More Doctors, More Clinics
Between 10% and 20% of the estimated hundreds of millions of people infected worldwide with SARS-CoV-2 in the first 2 years went on to develop long-term symptoms. While many recover over time, doctors who have treated long COVID since 2020 said they see some patients still wrestling with the condition after 4 years.
The latest Centers for Disease Control and Prevention Household Pulse Survey, taken between March 5 and April 1, 2024, estimated that nearly 7% of the adult population — more than 18 million people — currently have long COVID. Data from other countries also suggest that millions have been living with long COVID for years now, and hundreds of thousands have seen their day-to-day activities significantly affected.
There is an urgent need for more funding, long COVID clinicians, multidisciplinary clinics, and education for non–long COVID physicians and specialists, doctors said. Instead, funding remains limited, clinics are closing, wait times are “horrendously long,” patients are left in limbo, and physicians are burning out.
“What’s changed in some ways is that there’s even less access to COVID rehab, which sounds crazy because there was very little to begin with,” said Alexandra Rendely, MD, a physical medicine and rehabilitation physician with the interdisciplinary Toronto Rehab, a part of the University Health Network of teaching hospitals in Toronto, Ontario, Canada.
“Patients are still being diagnosed every day, yet the resources available are becoming less and less.”
COVID-19 money earmarked during the pandemic was mostly limited to temporary emergency measures. As those funds dwindled, governments and institutions have decreased financial support. The Long COVID Moonshot campaign, organized by patients with long COVID, is pushing Congress to support $1 billion in annual research funding to close the financial chasm.
The Clinical Trial Conundrum
While long COVID clinics have come a long way in helping patients, gaps remain. Doctors may be unwilling to prescribe off-label treatments without proper clinical trials due to the potential risks and liabilities involved or due to the controversial or unconventional nature of the therapies, said Dr. Malcolm, who left his primary care practice more than 2 years ago to focus on long COVID.
In the absence of standard treatments, Dr. Malcolm and other doctors said they must take a trial-and-error approach in treating patients with long COVID that centers on addressing symptoms and not the underlying condition.
“There are actually a lot of treatments and a lot of them are not curative, but they can help people,” he said.
Dr. Malcolm, who is a medical director at Real Time Health Monitoring, a private clinic in the San Francisco Bay Area that specializes in long COVID and myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), said it was important for him to be with a clinical team that understood and was supportive of his treatment decisions and was able to offer clinical support for those treatments if needed.
For physicians looking for clinical data before prescribing certain medications, the wait may be long. More than $1.5 billion in US federal funding has been earmarked to study long COVID, but the National Institutes of Health (NIH) has faced criticism from patients and scientists alike for its slow progress and emphasis on observational studies instead of research that could unravel the biological roots of long COVID. Among the clinical trials announced by the NIH’s RECOVER initiative, only a handful involve studying pharmaceutical treatments.
A 2023 editorial published in The Lancet called out the “dismal state of clinical research relative to the substantial burden of [long COVID]” and said, “we are clearly lacking tested pharmacological interventions that treat the underlying pathophysiology.” At the time of publication, it noted that of the 386 long COVID trials listed on ClinicalTrials.gov, only 12 were actually testing pharmacologic interventions.
There are also diagnostic and insurance barriers. The specialized tests that can detect long COVID anomalies are neither commonly known by primary care practitioners nor easily requested at the local lab, can be expensive, and are typically not covered by insurance, Dr. Malcolm explained.
Patients with long COVID also have the added barrier of being unable to advocate as easily because of their energy limitations, doctors said. Patients may appear outwardly fine, but fatigue and brain fog are among the many problems that cannot be measured in appearances. The condition has upended lives, some losing jobs, even homes, and the mental toll is why there is a “not insignificant” suicide rate.
One Patient’s 4-Year Journey
Charlie McCone, 34, used to be a tennis player and an active musician. But he’s spent the past 4 years mostly housebound, grappling with the aftermath of a SARS-CoV-2 infection he contracted in March 2020. He went from biking daily to work 10 miles and back to having at most 2 hours of energy per day.
In the first year alone, Mr. McCone saw more than two dozen doctors and specialists. The conditions now associated with long COVID, like ME/CFS, mast cell activation syndrome (a condition in which a patient experiences episodes of allergic symptoms such as hives, swelling, low blood pressure, and difficulty breathing), or dysautonomia (conditions that affect the autonomic nervous system, which controls automatic processes in the body) were not on physicians’ radars.
Then in 2021, he became bedbound for more than half a year after a Delta variant reinfection. He developed neurologic symptoms, including incapacitating fatigue, post-exertional malaise (where symptoms worsened after minimal physical or mental activity), left-sided weakness, and cognitive impairment. He stopped working altogether. But the worst was the shortness of breath he felt 24/7, even at rest. A battery of lab tests revealed nothing abnormal. He tried numerous drugs and the classic respiratory treatments.
Mr. McCone eventually connected with Dr. Malcolm over X and developed what he describes as an effective patient-doctor collaboration. When studies came out suggesting microclots were a common issue with patients with long COVID and positive outcomes were reported from anticoagulant therapy, they knew it could be one of the answers.
“After 3 weeks on [the antiplatelet drug], I was like, oh my god, my lungs are finally opening up,” said Mr. McCone. He has taken the medication for more than a year and a half, and some days he doesn’t even think about his respiratory symptoms.
“That trial-and-error process is just really long and hard and costly,” said Dr. Malcolm.
Today, fatigue and cognitive stamina are Mr. McCone’s main challenges, and he is far from recovered.
“[I had a] very fulfilling, happy life and now, it’s hard to think about. I’ve come a long way with my mental health and all this, but I’ve lost 4 years,” Mr. McCone said. “The prospect of me being here when I’m 40 seems very real ... so it’s pretty devastating.”
Lessons Learned, Hope Amid Ongoing Research
Despite the daunting obstacle, doctors said the science has come a long way for a new disease. We now know long COVID is likely caused by a combination of triggers, including viral reservoir in the tissue, inflammation, autoimmunity, and microclots; severity of infection is not necessarily an accurate risk factor predictor — long COVID can strike even those who had a mild infection; upward of 200 symptoms have been identified; and we know more about potential biomarkers that could lead to better diagnostic tools.
Unlike many other diseases and conditions with standard treatment protocols, long COVID treatments are typically aimed at addressing individual symptoms.
“It is very detailed and individualized to the patient’s specific symptoms and to the patient’s specific needs,” Dr. Rendely said. Symptoms can also fluctuate, relapse, or wax and wane, for example, so what ails a patient at their first doctor’s appointment could be completely different at the next appointment 2 months later.
Doctors are still hopeful the RECOVER research, which includes trials that look at autonomic and cognitive dysfunctions, will pave the way for more effective long COVID therapies. In Canada, Dr. Rendely is also eying the RECLAIM trial that is currently testing the effectiveness of pentoxifylline, which helps blood flow, and ibudilast, an anti-inflammatory drug.
Doctors are also hopeful when they see patients who have made “tremendous gains” or even full recoveries through their clinics. “It’s a new diagnosis, so I always tell my patients to think of this as a journey because I’m learning along with you,” said Jai Marathe, MD, an infectious disease physician at Boston Medical Center and an assistant professor of infectious diseases at Boston University Chobanian & Avedisian School of Medicine.
“Now we have 4 years of experience, but at the same time, no two long COVID patients are alike.”
Long COVID has also changed the way physicians view healthcare and how they practice medicine.
“I am a completely different person than I used to be because of this illness, and I don’t even have it. That is how profoundly it has affected how I view the universe,” said Dr. Malcolm. “I’ve been doing this for 4 years, and I’m very hopeful. But I don’t think about this in terms of months anymore. I think about this in terms of years.”
A version of this article first appeared on Medscape.com.
Stuart Malcolm, MD, a primary care physician who practices in Oregon and northern California, started seeing patients with long COVID early in the pandemic. Back then, he was frustrated by the obstacles and lack of standard diagnostic tests and treatments. Four years later, well, he still is.
“Something I learned the last few years is the logistics to get people care is really, really hard,” he said. “There’s a lot of frustration. It’s mostly frustration.”
For long COVID doctors and patients, there has been little to no progress addressing the challenges, leaving many discouraged. Researchers and clinicians now have a greater understanding of what health agencies formally call post-COVID condition, but the wide spectrum of symptoms, slow progress in launching pharmacologic clinical trials, and the research toward understanding the underlying causes mean standardized diagnostic tests and definitive treatments remain elusive.
“The frustration is that we aren’t able to help everyone with our current knowledge base. And I think the frustration lies not just with us physicians but also with patients because they’re at the point where if they tried everything, literally everything and haven’t gotten better,” said Zijian Chen, MD, director of the Mount Sinai Center for Post-COVID Care in New York City.
Wanted: More Funding, More Doctors, More Clinics
Between 10% and 20% of the estimated hundreds of millions of people infected worldwide with SARS-CoV-2 in the first 2 years went on to develop long-term symptoms. While many recover over time, doctors who have treated long COVID since 2020 said they see some patients still wrestling with the condition after 4 years.
The latest Centers for Disease Control and Prevention Household Pulse Survey, taken between March 5 and April 1, 2024, estimated that nearly 7% of the adult population — more than 18 million people — currently have long COVID. Data from other countries also suggest that millions have been living with long COVID for years now, and hundreds of thousands have seen their day-to-day activities significantly affected.
There is an urgent need for more funding, long COVID clinicians, multidisciplinary clinics, and education for non–long COVID physicians and specialists, doctors said. Instead, funding remains limited, clinics are closing, wait times are “horrendously long,” patients are left in limbo, and physicians are burning out.
“What’s changed in some ways is that there’s even less access to COVID rehab, which sounds crazy because there was very little to begin with,” said Alexandra Rendely, MD, a physical medicine and rehabilitation physician with the interdisciplinary Toronto Rehab, a part of the University Health Network of teaching hospitals in Toronto, Ontario, Canada.
“Patients are still being diagnosed every day, yet the resources available are becoming less and less.”
COVID-19 money earmarked during the pandemic was mostly limited to temporary emergency measures. As those funds dwindled, governments and institutions have decreased financial support. The Long COVID Moonshot campaign, organized by patients with long COVID, is pushing Congress to support $1 billion in annual research funding to close the financial chasm.
The Clinical Trial Conundrum
While long COVID clinics have come a long way in helping patients, gaps remain. Doctors may be unwilling to prescribe off-label treatments without proper clinical trials due to the potential risks and liabilities involved or due to the controversial or unconventional nature of the therapies, said Dr. Malcolm, who left his primary care practice more than 2 years ago to focus on long COVID.
In the absence of standard treatments, Dr. Malcolm and other doctors said they must take a trial-and-error approach in treating patients with long COVID that centers on addressing symptoms and not the underlying condition.
“There are actually a lot of treatments and a lot of them are not curative, but they can help people,” he said.
Dr. Malcolm, who is a medical director at Real Time Health Monitoring, a private clinic in the San Francisco Bay Area that specializes in long COVID and myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), said it was important for him to be with a clinical team that understood and was supportive of his treatment decisions and was able to offer clinical support for those treatments if needed.
For physicians looking for clinical data before prescribing certain medications, the wait may be long. More than $1.5 billion in US federal funding has been earmarked to study long COVID, but the National Institutes of Health (NIH) has faced criticism from patients and scientists alike for its slow progress and emphasis on observational studies instead of research that could unravel the biological roots of long COVID. Among the clinical trials announced by the NIH’s RECOVER initiative, only a handful involve studying pharmaceutical treatments.
A 2023 editorial published in The Lancet called out the “dismal state of clinical research relative to the substantial burden of [long COVID]” and said, “we are clearly lacking tested pharmacological interventions that treat the underlying pathophysiology.” At the time of publication, it noted that of the 386 long COVID trials listed on ClinicalTrials.gov, only 12 were actually testing pharmacologic interventions.
There are also diagnostic and insurance barriers. The specialized tests that can detect long COVID anomalies are neither commonly known by primary care practitioners nor easily requested at the local lab, can be expensive, and are typically not covered by insurance, Dr. Malcolm explained.
Patients with long COVID also have the added barrier of being unable to advocate as easily because of their energy limitations, doctors said. Patients may appear outwardly fine, but fatigue and brain fog are among the many problems that cannot be measured in appearances. The condition has upended lives, some losing jobs, even homes, and the mental toll is why there is a “not insignificant” suicide rate.
One Patient’s 4-Year Journey
Charlie McCone, 34, used to be a tennis player and an active musician. But he’s spent the past 4 years mostly housebound, grappling with the aftermath of a SARS-CoV-2 infection he contracted in March 2020. He went from biking daily to work 10 miles and back to having at most 2 hours of energy per day.
In the first year alone, Mr. McCone saw more than two dozen doctors and specialists. The conditions now associated with long COVID, like ME/CFS, mast cell activation syndrome (a condition in which a patient experiences episodes of allergic symptoms such as hives, swelling, low blood pressure, and difficulty breathing), or dysautonomia (conditions that affect the autonomic nervous system, which controls automatic processes in the body) were not on physicians’ radars.
Then in 2021, he became bedbound for more than half a year after a Delta variant reinfection. He developed neurologic symptoms, including incapacitating fatigue, post-exertional malaise (where symptoms worsened after minimal physical or mental activity), left-sided weakness, and cognitive impairment. He stopped working altogether. But the worst was the shortness of breath he felt 24/7, even at rest. A battery of lab tests revealed nothing abnormal. He tried numerous drugs and the classic respiratory treatments.
Mr. McCone eventually connected with Dr. Malcolm over X and developed what he describes as an effective patient-doctor collaboration. When studies came out suggesting microclots were a common issue with patients with long COVID and positive outcomes were reported from anticoagulant therapy, they knew it could be one of the answers.
“After 3 weeks on [the antiplatelet drug], I was like, oh my god, my lungs are finally opening up,” said Mr. McCone. He has taken the medication for more than a year and a half, and some days he doesn’t even think about his respiratory symptoms.
“That trial-and-error process is just really long and hard and costly,” said Dr. Malcolm.
Today, fatigue and cognitive stamina are Mr. McCone’s main challenges, and he is far from recovered.
“[I had a] very fulfilling, happy life and now, it’s hard to think about. I’ve come a long way with my mental health and all this, but I’ve lost 4 years,” Mr. McCone said. “The prospect of me being here when I’m 40 seems very real ... so it’s pretty devastating.”
Lessons Learned, Hope Amid Ongoing Research
Despite the daunting obstacle, doctors said the science has come a long way for a new disease. We now know long COVID is likely caused by a combination of triggers, including viral reservoir in the tissue, inflammation, autoimmunity, and microclots; severity of infection is not necessarily an accurate risk factor predictor — long COVID can strike even those who had a mild infection; upward of 200 symptoms have been identified; and we know more about potential biomarkers that could lead to better diagnostic tools.
Unlike many other diseases and conditions with standard treatment protocols, long COVID treatments are typically aimed at addressing individual symptoms.
“It is very detailed and individualized to the patient’s specific symptoms and to the patient’s specific needs,” Dr. Rendely said. Symptoms can also fluctuate, relapse, or wax and wane, for example, so what ails a patient at their first doctor’s appointment could be completely different at the next appointment 2 months later.
Doctors are still hopeful the RECOVER research, which includes trials that look at autonomic and cognitive dysfunctions, will pave the way for more effective long COVID therapies. In Canada, Dr. Rendely is also eying the RECLAIM trial that is currently testing the effectiveness of pentoxifylline, which helps blood flow, and ibudilast, an anti-inflammatory drug.
Doctors are also hopeful when they see patients who have made “tremendous gains” or even full recoveries through their clinics. “It’s a new diagnosis, so I always tell my patients to think of this as a journey because I’m learning along with you,” said Jai Marathe, MD, an infectious disease physician at Boston Medical Center and an assistant professor of infectious diseases at Boston University Chobanian & Avedisian School of Medicine.
“Now we have 4 years of experience, but at the same time, no two long COVID patients are alike.”
Long COVID has also changed the way physicians view healthcare and how they practice medicine.
“I am a completely different person than I used to be because of this illness, and I don’t even have it. That is how profoundly it has affected how I view the universe,” said Dr. Malcolm. “I’ve been doing this for 4 years, and I’m very hopeful. But I don’t think about this in terms of months anymore. I think about this in terms of years.”
A version of this article first appeared on Medscape.com.
Stuart Malcolm, MD, a primary care physician who practices in Oregon and northern California, started seeing patients with long COVID early in the pandemic. Back then, he was frustrated by the obstacles and lack of standard diagnostic tests and treatments. Four years later, well, he still is.
“Something I learned the last few years is the logistics to get people care is really, really hard,” he said. “There’s a lot of frustration. It’s mostly frustration.”
For long COVID doctors and patients, there has been little to no progress addressing the challenges, leaving many discouraged. Researchers and clinicians now have a greater understanding of what health agencies formally call post-COVID condition, but the wide spectrum of symptoms, slow progress in launching pharmacologic clinical trials, and the research toward understanding the underlying causes mean standardized diagnostic tests and definitive treatments remain elusive.
“The frustration is that we aren’t able to help everyone with our current knowledge base. And I think the frustration lies not just with us physicians but also with patients because they’re at the point where if they tried everything, literally everything and haven’t gotten better,” said Zijian Chen, MD, director of the Mount Sinai Center for Post-COVID Care in New York City.
Wanted: More Funding, More Doctors, More Clinics
Between 10% and 20% of the estimated hundreds of millions of people infected worldwide with SARS-CoV-2 in the first 2 years went on to develop long-term symptoms. While many recover over time, doctors who have treated long COVID since 2020 said they see some patients still wrestling with the condition after 4 years.
The latest Centers for Disease Control and Prevention Household Pulse Survey, taken between March 5 and April 1, 2024, estimated that nearly 7% of the adult population — more than 18 million people — currently have long COVID. Data from other countries also suggest that millions have been living with long COVID for years now, and hundreds of thousands have seen their day-to-day activities significantly affected.
There is an urgent need for more funding, long COVID clinicians, multidisciplinary clinics, and education for non–long COVID physicians and specialists, doctors said. Instead, funding remains limited, clinics are closing, wait times are “horrendously long,” patients are left in limbo, and physicians are burning out.
“What’s changed in some ways is that there’s even less access to COVID rehab, which sounds crazy because there was very little to begin with,” said Alexandra Rendely, MD, a physical medicine and rehabilitation physician with the interdisciplinary Toronto Rehab, a part of the University Health Network of teaching hospitals in Toronto, Ontario, Canada.
“Patients are still being diagnosed every day, yet the resources available are becoming less and less.”
COVID-19 money earmarked during the pandemic was mostly limited to temporary emergency measures. As those funds dwindled, governments and institutions have decreased financial support. The Long COVID Moonshot campaign, organized by patients with long COVID, is pushing Congress to support $1 billion in annual research funding to close the financial chasm.
The Clinical Trial Conundrum
While long COVID clinics have come a long way in helping patients, gaps remain. Doctors may be unwilling to prescribe off-label treatments without proper clinical trials due to the potential risks and liabilities involved or due to the controversial or unconventional nature of the therapies, said Dr. Malcolm, who left his primary care practice more than 2 years ago to focus on long COVID.
In the absence of standard treatments, Dr. Malcolm and other doctors said they must take a trial-and-error approach in treating patients with long COVID that centers on addressing symptoms and not the underlying condition.
“There are actually a lot of treatments and a lot of them are not curative, but they can help people,” he said.
Dr. Malcolm, who is a medical director at Real Time Health Monitoring, a private clinic in the San Francisco Bay Area that specializes in long COVID and myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), said it was important for him to be with a clinical team that understood and was supportive of his treatment decisions and was able to offer clinical support for those treatments if needed.
For physicians looking for clinical data before prescribing certain medications, the wait may be long. More than $1.5 billion in US federal funding has been earmarked to study long COVID, but the National Institutes of Health (NIH) has faced criticism from patients and scientists alike for its slow progress and emphasis on observational studies instead of research that could unravel the biological roots of long COVID. Among the clinical trials announced by the NIH’s RECOVER initiative, only a handful involve studying pharmaceutical treatments.
A 2023 editorial published in The Lancet called out the “dismal state of clinical research relative to the substantial burden of [long COVID]” and said, “we are clearly lacking tested pharmacological interventions that treat the underlying pathophysiology.” At the time of publication, it noted that of the 386 long COVID trials listed on ClinicalTrials.gov, only 12 were actually testing pharmacologic interventions.
There are also diagnostic and insurance barriers. The specialized tests that can detect long COVID anomalies are neither commonly known by primary care practitioners nor easily requested at the local lab, can be expensive, and are typically not covered by insurance, Dr. Malcolm explained.
Patients with long COVID also have the added barrier of being unable to advocate as easily because of their energy limitations, doctors said. Patients may appear outwardly fine, but fatigue and brain fog are among the many problems that cannot be measured in appearances. The condition has upended lives, some losing jobs, even homes, and the mental toll is why there is a “not insignificant” suicide rate.
One Patient’s 4-Year Journey
Charlie McCone, 34, used to be a tennis player and an active musician. But he’s spent the past 4 years mostly housebound, grappling with the aftermath of a SARS-CoV-2 infection he contracted in March 2020. He went from biking daily to work 10 miles and back to having at most 2 hours of energy per day.
In the first year alone, Mr. McCone saw more than two dozen doctors and specialists. The conditions now associated with long COVID, like ME/CFS, mast cell activation syndrome (a condition in which a patient experiences episodes of allergic symptoms such as hives, swelling, low blood pressure, and difficulty breathing), or dysautonomia (conditions that affect the autonomic nervous system, which controls automatic processes in the body) were not on physicians’ radars.
Then in 2021, he became bedbound for more than half a year after a Delta variant reinfection. He developed neurologic symptoms, including incapacitating fatigue, post-exertional malaise (where symptoms worsened after minimal physical or mental activity), left-sided weakness, and cognitive impairment. He stopped working altogether. But the worst was the shortness of breath he felt 24/7, even at rest. A battery of lab tests revealed nothing abnormal. He tried numerous drugs and the classic respiratory treatments.
Mr. McCone eventually connected with Dr. Malcolm over X and developed what he describes as an effective patient-doctor collaboration. When studies came out suggesting microclots were a common issue with patients with long COVID and positive outcomes were reported from anticoagulant therapy, they knew it could be one of the answers.
“After 3 weeks on [the antiplatelet drug], I was like, oh my god, my lungs are finally opening up,” said Mr. McCone. He has taken the medication for more than a year and a half, and some days he doesn’t even think about his respiratory symptoms.
“That trial-and-error process is just really long and hard and costly,” said Dr. Malcolm.
Today, fatigue and cognitive stamina are Mr. McCone’s main challenges, and he is far from recovered.
“[I had a] very fulfilling, happy life and now, it’s hard to think about. I’ve come a long way with my mental health and all this, but I’ve lost 4 years,” Mr. McCone said. “The prospect of me being here when I’m 40 seems very real ... so it’s pretty devastating.”
Lessons Learned, Hope Amid Ongoing Research
Despite the daunting obstacle, doctors said the science has come a long way for a new disease. We now know long COVID is likely caused by a combination of triggers, including viral reservoir in the tissue, inflammation, autoimmunity, and microclots; severity of infection is not necessarily an accurate risk factor predictor — long COVID can strike even those who had a mild infection; upward of 200 symptoms have been identified; and we know more about potential biomarkers that could lead to better diagnostic tools.
Unlike many other diseases and conditions with standard treatment protocols, long COVID treatments are typically aimed at addressing individual symptoms.
“It is very detailed and individualized to the patient’s specific symptoms and to the patient’s specific needs,” Dr. Rendely said. Symptoms can also fluctuate, relapse, or wax and wane, for example, so what ails a patient at their first doctor’s appointment could be completely different at the next appointment 2 months later.
Doctors are still hopeful the RECOVER research, which includes trials that look at autonomic and cognitive dysfunctions, will pave the way for more effective long COVID therapies. In Canada, Dr. Rendely is also eying the RECLAIM trial that is currently testing the effectiveness of pentoxifylline, which helps blood flow, and ibudilast, an anti-inflammatory drug.
Doctors are also hopeful when they see patients who have made “tremendous gains” or even full recoveries through their clinics. “It’s a new diagnosis, so I always tell my patients to think of this as a journey because I’m learning along with you,” said Jai Marathe, MD, an infectious disease physician at Boston Medical Center and an assistant professor of infectious diseases at Boston University Chobanian & Avedisian School of Medicine.
“Now we have 4 years of experience, but at the same time, no two long COVID patients are alike.”
Long COVID has also changed the way physicians view healthcare and how they practice medicine.
“I am a completely different person than I used to be because of this illness, and I don’t even have it. That is how profoundly it has affected how I view the universe,” said Dr. Malcolm. “I’ve been doing this for 4 years, and I’m very hopeful. But I don’t think about this in terms of months anymore. I think about this in terms of years.”
A version of this article first appeared on Medscape.com.
The Push to Get More People Into Long COVID Studies
When Ezra Spier was diagnosed with long COVID in late 2022, his main symptom, postexertional malaise, caused fatigue so severe that it forced him to quit his job as a technology entrepreneur. Since then, it’s been a tough road for Spier, 37, who said he wouldn’t wish his hellish condition on anyone.
Last spring, he enrolled in a clinical trial of a new long COVID therapy at Stanford University, and he’s about to start another at the University of California, San Francisco.
For Spier, who lives in Oakland, California, being part of the clinical trials connected him with people dealing with similar health issues while also moving the needle toward better treatments for everyone. Yet many potential participants are unaware that these clinical trials exist. Clinical trial researchers also express frustration over the challenge of enrolling participants.
That’s why Spier created a new website to help match long COVID patients with clinical trials that can help.
“I wanted a way to make long COVID clinical trials more accessible to the general public,” he said. Spier’s website, aptly named Long Covid Studies, launched in March. The site already includes details from about 550 trials globally and, in the future, will include many more.
It’s Not the Number of Studies, It’s Navigating Them
In all, nearly 9300 long COVID trials are listed on ClinicalTrials.gov. But many patients find the site difficult to navigate, said David F. Putrino, PhD, who runs the long COVID clinic at Mount Sinai Health System in New York City. He said Spier’s website helps make trials easier for patients to manage in ways that remove the enrollment challenges.
“Ezra’s platform pulls data from ClinicalTrials.gov and puts it into a space that’s much easier for patients to manage,” said Dr. Putrino. The site only includes the most relevant information, such as the study location, eligibility, and purpose and how to sign up.
Another of Spier’s goals is to make the process easier for patients who are already marginalized and often excluded from the healthcare system. Long COVID disproportionately impacts people in minority ethnic groups and women, as well as those who are impoverished or live in rural areas.
According to the National Institutes of Health (NIH), 1 in 4 patients with severe long COVID-19 are Black or Hispanic whereas only 1 in 7 are White. Yet participation by White persons in clinical trials is much higher overall: 77% of participants are White, compared with only 14% for Black persons and 15% for Hispanic persons. Without more balanced representation, research becomes skewed and less accurate, said Grace McComsey, MD, who leads one of the 15 nationwide long COVID centers funded by the federal RECOVER (Researching COVID to Enhance Recovery) Initiative in Cleveland.
Websites that are easier for the layperson to access would allow for wider participation, said McComsey.
Too Many Barriers to Entry
A study published in the Journal of Applied Gerontology found that transportation plays an outsized role in influencing study participation, which may also lead to less diverse participation.
Decentralized trials — in which participants receive therapy at home — also make enrolling in clinical trials easier for marginalized patients and those too sick to make it to a research center, said Dr. Putrino. Research published recently in The American Journal of Medicine demonstrated that for many patients, remote studies are the future of COVID research. The study, focusing on the efficacy of Paxlovid, recruited patients living in the 48 contiguous US states. Participation was entirely remote.
“We need to have more consideration for bedbound and housebound patients in our research,” said Dr. Putrino. “Some people don’t have the ability to show up to a prestigious university to take part in an academic trial.”
Dr. Putrino and colleagues at Yale School of Medicine’s Yale COVID Recovery Study plan to release a paper in the near future on the methodology for running decentralized or remote studies that could provide guidance for researchers elsewhere.
Decentralized studies serve a larger audience, but they’re also more expensive and cost has plagued long COVID research from the start, said Michael Peluso, MD, an assistant research professor of infectious medicine at UCSF School of Medicine, University of California, San Francisco.
“You need to have a staff in place that’s trained to do home visits in order to conduct remote trials,” Dr. Peluso said, adding that his biggest challenge has been connecting patients to appropriate clinical trials.
Individual eligibility has been an ongoing issue. For example, Dr. Peluso’s current trials are testing monoclonal antibodies — antibodies produced by cloning unique white blood cells to target viral persistence, which is thought to be a cause of long COVID. Only patients who were infected with certain variants of acute COVID are eligible because of the antibodies needed to target SARS-CoV-2 spike proteins.
“This can lead to a lot of frustration among patients who might think they can participate, but aren’t eligible,” said Dr. Peluso.
Long Fight for Better Long COVID Research
For Spier, one of the hardest parts of his health issues and lack of energy is that they have sharply curtailed his social interactions with friends and colleagues.
He has channeled his energies into researching new treatments that could potentially improve his symptoms. That research is partly what drove him to create the Long Covid Studies website.
His goal is still to help others with long COVID find trials that can improve their symptoms as well. The more people who participate, the closer scientists will come to providing effective treatments for everyone, he said.
“For all my frustrations, we’re still at the forefront of science globally,” he said. “And if we have the level of funding the NIH is equipped to provide, we can show the world what’s possible with long COVID research.”
A version of this article first appeared on Medscape.com.
When Ezra Spier was diagnosed with long COVID in late 2022, his main symptom, postexertional malaise, caused fatigue so severe that it forced him to quit his job as a technology entrepreneur. Since then, it’s been a tough road for Spier, 37, who said he wouldn’t wish his hellish condition on anyone.
Last spring, he enrolled in a clinical trial of a new long COVID therapy at Stanford University, and he’s about to start another at the University of California, San Francisco.
For Spier, who lives in Oakland, California, being part of the clinical trials connected him with people dealing with similar health issues while also moving the needle toward better treatments for everyone. Yet many potential participants are unaware that these clinical trials exist. Clinical trial researchers also express frustration over the challenge of enrolling participants.
That’s why Spier created a new website to help match long COVID patients with clinical trials that can help.
“I wanted a way to make long COVID clinical trials more accessible to the general public,” he said. Spier’s website, aptly named Long Covid Studies, launched in March. The site already includes details from about 550 trials globally and, in the future, will include many more.
It’s Not the Number of Studies, It’s Navigating Them
In all, nearly 9300 long COVID trials are listed on ClinicalTrials.gov. But many patients find the site difficult to navigate, said David F. Putrino, PhD, who runs the long COVID clinic at Mount Sinai Health System in New York City. He said Spier’s website helps make trials easier for patients to manage in ways that remove the enrollment challenges.
“Ezra’s platform pulls data from ClinicalTrials.gov and puts it into a space that’s much easier for patients to manage,” said Dr. Putrino. The site only includes the most relevant information, such as the study location, eligibility, and purpose and how to sign up.
Another of Spier’s goals is to make the process easier for patients who are already marginalized and often excluded from the healthcare system. Long COVID disproportionately impacts people in minority ethnic groups and women, as well as those who are impoverished or live in rural areas.
According to the National Institutes of Health (NIH), 1 in 4 patients with severe long COVID-19 are Black or Hispanic whereas only 1 in 7 are White. Yet participation by White persons in clinical trials is much higher overall: 77% of participants are White, compared with only 14% for Black persons and 15% for Hispanic persons. Without more balanced representation, research becomes skewed and less accurate, said Grace McComsey, MD, who leads one of the 15 nationwide long COVID centers funded by the federal RECOVER (Researching COVID to Enhance Recovery) Initiative in Cleveland.
Websites that are easier for the layperson to access would allow for wider participation, said McComsey.
Too Many Barriers to Entry
A study published in the Journal of Applied Gerontology found that transportation plays an outsized role in influencing study participation, which may also lead to less diverse participation.
Decentralized trials — in which participants receive therapy at home — also make enrolling in clinical trials easier for marginalized patients and those too sick to make it to a research center, said Dr. Putrino. Research published recently in The American Journal of Medicine demonstrated that for many patients, remote studies are the future of COVID research. The study, focusing on the efficacy of Paxlovid, recruited patients living in the 48 contiguous US states. Participation was entirely remote.
“We need to have more consideration for bedbound and housebound patients in our research,” said Dr. Putrino. “Some people don’t have the ability to show up to a prestigious university to take part in an academic trial.”
Dr. Putrino and colleagues at Yale School of Medicine’s Yale COVID Recovery Study plan to release a paper in the near future on the methodology for running decentralized or remote studies that could provide guidance for researchers elsewhere.
Decentralized studies serve a larger audience, but they’re also more expensive and cost has plagued long COVID research from the start, said Michael Peluso, MD, an assistant research professor of infectious medicine at UCSF School of Medicine, University of California, San Francisco.
“You need to have a staff in place that’s trained to do home visits in order to conduct remote trials,” Dr. Peluso said, adding that his biggest challenge has been connecting patients to appropriate clinical trials.
Individual eligibility has been an ongoing issue. For example, Dr. Peluso’s current trials are testing monoclonal antibodies — antibodies produced by cloning unique white blood cells to target viral persistence, which is thought to be a cause of long COVID. Only patients who were infected with certain variants of acute COVID are eligible because of the antibodies needed to target SARS-CoV-2 spike proteins.
“This can lead to a lot of frustration among patients who might think they can participate, but aren’t eligible,” said Dr. Peluso.
Long Fight for Better Long COVID Research
For Spier, one of the hardest parts of his health issues and lack of energy is that they have sharply curtailed his social interactions with friends and colleagues.
He has channeled his energies into researching new treatments that could potentially improve his symptoms. That research is partly what drove him to create the Long Covid Studies website.
His goal is still to help others with long COVID find trials that can improve their symptoms as well. The more people who participate, the closer scientists will come to providing effective treatments for everyone, he said.
“For all my frustrations, we’re still at the forefront of science globally,” he said. “And if we have the level of funding the NIH is equipped to provide, we can show the world what’s possible with long COVID research.”
A version of this article first appeared on Medscape.com.
When Ezra Spier was diagnosed with long COVID in late 2022, his main symptom, postexertional malaise, caused fatigue so severe that it forced him to quit his job as a technology entrepreneur. Since then, it’s been a tough road for Spier, 37, who said he wouldn’t wish his hellish condition on anyone.
Last spring, he enrolled in a clinical trial of a new long COVID therapy at Stanford University, and he’s about to start another at the University of California, San Francisco.
For Spier, who lives in Oakland, California, being part of the clinical trials connected him with people dealing with similar health issues while also moving the needle toward better treatments for everyone. Yet many potential participants are unaware that these clinical trials exist. Clinical trial researchers also express frustration over the challenge of enrolling participants.
That’s why Spier created a new website to help match long COVID patients with clinical trials that can help.
“I wanted a way to make long COVID clinical trials more accessible to the general public,” he said. Spier’s website, aptly named Long Covid Studies, launched in March. The site already includes details from about 550 trials globally and, in the future, will include many more.
It’s Not the Number of Studies, It’s Navigating Them
In all, nearly 9300 long COVID trials are listed on ClinicalTrials.gov. But many patients find the site difficult to navigate, said David F. Putrino, PhD, who runs the long COVID clinic at Mount Sinai Health System in New York City. He said Spier’s website helps make trials easier for patients to manage in ways that remove the enrollment challenges.
“Ezra’s platform pulls data from ClinicalTrials.gov and puts it into a space that’s much easier for patients to manage,” said Dr. Putrino. The site only includes the most relevant information, such as the study location, eligibility, and purpose and how to sign up.
Another of Spier’s goals is to make the process easier for patients who are already marginalized and often excluded from the healthcare system. Long COVID disproportionately impacts people in minority ethnic groups and women, as well as those who are impoverished or live in rural areas.
According to the National Institutes of Health (NIH), 1 in 4 patients with severe long COVID-19 are Black or Hispanic whereas only 1 in 7 are White. Yet participation by White persons in clinical trials is much higher overall: 77% of participants are White, compared with only 14% for Black persons and 15% for Hispanic persons. Without more balanced representation, research becomes skewed and less accurate, said Grace McComsey, MD, who leads one of the 15 nationwide long COVID centers funded by the federal RECOVER (Researching COVID to Enhance Recovery) Initiative in Cleveland.
Websites that are easier for the layperson to access would allow for wider participation, said McComsey.
Too Many Barriers to Entry
A study published in the Journal of Applied Gerontology found that transportation plays an outsized role in influencing study participation, which may also lead to less diverse participation.
Decentralized trials — in which participants receive therapy at home — also make enrolling in clinical trials easier for marginalized patients and those too sick to make it to a research center, said Dr. Putrino. Research published recently in The American Journal of Medicine demonstrated that for many patients, remote studies are the future of COVID research. The study, focusing on the efficacy of Paxlovid, recruited patients living in the 48 contiguous US states. Participation was entirely remote.
“We need to have more consideration for bedbound and housebound patients in our research,” said Dr. Putrino. “Some people don’t have the ability to show up to a prestigious university to take part in an academic trial.”
Dr. Putrino and colleagues at Yale School of Medicine’s Yale COVID Recovery Study plan to release a paper in the near future on the methodology for running decentralized or remote studies that could provide guidance for researchers elsewhere.
Decentralized studies serve a larger audience, but they’re also more expensive and cost has plagued long COVID research from the start, said Michael Peluso, MD, an assistant research professor of infectious medicine at UCSF School of Medicine, University of California, San Francisco.
“You need to have a staff in place that’s trained to do home visits in order to conduct remote trials,” Dr. Peluso said, adding that his biggest challenge has been connecting patients to appropriate clinical trials.
Individual eligibility has been an ongoing issue. For example, Dr. Peluso’s current trials are testing monoclonal antibodies — antibodies produced by cloning unique white blood cells to target viral persistence, which is thought to be a cause of long COVID. Only patients who were infected with certain variants of acute COVID are eligible because of the antibodies needed to target SARS-CoV-2 spike proteins.
“This can lead to a lot of frustration among patients who might think they can participate, but aren’t eligible,” said Dr. Peluso.
Long Fight for Better Long COVID Research
For Spier, one of the hardest parts of his health issues and lack of energy is that they have sharply curtailed his social interactions with friends and colleagues.
He has channeled his energies into researching new treatments that could potentially improve his symptoms. That research is partly what drove him to create the Long Covid Studies website.
His goal is still to help others with long COVID find trials that can improve their symptoms as well. The more people who participate, the closer scientists will come to providing effective treatments for everyone, he said.
“For all my frustrations, we’re still at the forefront of science globally,” he said. “And if we have the level of funding the NIH is equipped to provide, we can show the world what’s possible with long COVID research.”
A version of this article first appeared on Medscape.com.
NIH to Begin Long COVID Trial Focused on Sleep, Exercise
The National institutes of Health will soon start a clinical trial in an attempt to find potential treatments for symptoms of long COVID, focusing on sleep disturbances, problems with exercise, and post-exertional malaise.
“When people can’t get reliable sleep, can’t exert themselves and feel sick following tasks that used to be simple, the physical and mental anguish can lead to feelings of utter helplessness,” Walter J. Koroshetz, MD, director of the NIH’s National Institute of Neurological Disorders and Stroke, said in a statement. “We urgently need to come up with answers to help those struggling with long COVID feel whole again.”
The new trials will be part of the NIH’s Researching COVID to Enhance Recovery initiative, known as RECOVER. Since beginning enrollment in July 2023 for four trials, RECOVER now features eight trials across the country looking at all parts of long COVID. RECOVER is part of a $1.15 billion nationwide program that Congress approved in 2020 for the NIH to research and test treatments for long COVID.
While focused on sleep disturbances, the trial will test two Food and Drug Administration–approved drugs currently used to treat people with hypersomnia. There will also be a trial to test if melatonin helps people with long COVID-related sleep problems. Light therapy will also be tested.
The trials that deal with problems people have had with exercise will focus on personalized cardiopulmonary rehabilitation, where patients experiment with exercise training, strength and flexibility training, education, and social support.
Another trial will look at structured pacing, which is designed to help people with exercise problems identify, control, and ease long COVID symptoms.
A version of this article appeared on WebMD.com.
The National institutes of Health will soon start a clinical trial in an attempt to find potential treatments for symptoms of long COVID, focusing on sleep disturbances, problems with exercise, and post-exertional malaise.
“When people can’t get reliable sleep, can’t exert themselves and feel sick following tasks that used to be simple, the physical and mental anguish can lead to feelings of utter helplessness,” Walter J. Koroshetz, MD, director of the NIH’s National Institute of Neurological Disorders and Stroke, said in a statement. “We urgently need to come up with answers to help those struggling with long COVID feel whole again.”
The new trials will be part of the NIH’s Researching COVID to Enhance Recovery initiative, known as RECOVER. Since beginning enrollment in July 2023 for four trials, RECOVER now features eight trials across the country looking at all parts of long COVID. RECOVER is part of a $1.15 billion nationwide program that Congress approved in 2020 for the NIH to research and test treatments for long COVID.
While focused on sleep disturbances, the trial will test two Food and Drug Administration–approved drugs currently used to treat people with hypersomnia. There will also be a trial to test if melatonin helps people with long COVID-related sleep problems. Light therapy will also be tested.
The trials that deal with problems people have had with exercise will focus on personalized cardiopulmonary rehabilitation, where patients experiment with exercise training, strength and flexibility training, education, and social support.
Another trial will look at structured pacing, which is designed to help people with exercise problems identify, control, and ease long COVID symptoms.
A version of this article appeared on WebMD.com.
The National institutes of Health will soon start a clinical trial in an attempt to find potential treatments for symptoms of long COVID, focusing on sleep disturbances, problems with exercise, and post-exertional malaise.
“When people can’t get reliable sleep, can’t exert themselves and feel sick following tasks that used to be simple, the physical and mental anguish can lead to feelings of utter helplessness,” Walter J. Koroshetz, MD, director of the NIH’s National Institute of Neurological Disorders and Stroke, said in a statement. “We urgently need to come up with answers to help those struggling with long COVID feel whole again.”
The new trials will be part of the NIH’s Researching COVID to Enhance Recovery initiative, known as RECOVER. Since beginning enrollment in July 2023 for four trials, RECOVER now features eight trials across the country looking at all parts of long COVID. RECOVER is part of a $1.15 billion nationwide program that Congress approved in 2020 for the NIH to research and test treatments for long COVID.
While focused on sleep disturbances, the trial will test two Food and Drug Administration–approved drugs currently used to treat people with hypersomnia. There will also be a trial to test if melatonin helps people with long COVID-related sleep problems. Light therapy will also be tested.
The trials that deal with problems people have had with exercise will focus on personalized cardiopulmonary rehabilitation, where patients experiment with exercise training, strength and flexibility training, education, and social support.
Another trial will look at structured pacing, which is designed to help people with exercise problems identify, control, and ease long COVID symptoms.
A version of this article appeared on WebMD.com.
4 Years In, a Sobering Look at Long COVID Progress
Four years ago in the spring of 2020, physicians and patients coined the term “long COVID” to describe a form of the viral infection from which recovery seemed impossible. (And the old nickname “long-haulers” seems so quaint now.)
What started as a pandemic that killed nearly 3 million people globally in 2020 alone would turn into a chronic disease causing a long list of symptoms — from extreme fatigue, to brain fog, tremors, nausea, headaches, rapid heartbeat, and more.
Today, 6.4% of Americans report symptoms of long COVID, and many have never recovered.
Still, we’ve come a long way, although there’s much we don’t understand about the condition. At the very least, physicians have a greater understanding that long COVID exists and can cause serious long-term symptoms.
While physicians may not have a blanket diagnostic tool that works for all patients with long COVID, they have refined existing tests for more accurate results, said Nisha Viswanathan, MD, director of the University of California Los Angeles Long COVID Program at UCLA Health.
Also, a range of new treatments, now undergoing clinical trials, have emerged that have proved effective in managing long COVID symptoms.
Catecholamine testing, for example, is now commonly used to diagnose long COVID, particularly in those who have dysautonomia, a condition caused by dysfunction of the autonomic nervous system and marked by dizziness, low blood pressure, nausea, and brain fog.
Very high levels of the neurotransmitter, for example, were shown to indicate long COVID in a January 2021 study published in the journal Clinical Medicine.
Certain biomarkers have also been shown indicative of the condition, including low serotonin levels. A study published this year in Cell found lower serotonin levels in patients with long COVID driven by low levels of circulating SARS-CoV-2, the virus that causes the condition.
Still, said Dr. Viswanathan, long COVID is a disease diagnosed by figuring out what a patient does not have — by ruling out other causes — rather than what they do. “It’s still a moving target,” she said, meaning that the disease is always changing based on the variant of acute COVID.
Promising Treatments Have Emerged
Dysautonomia, and especially the associated brain fog, fatigue, and dizziness, are now common conditions. As a result, physicians have gotten better at treating them. The vagus nerve is the main nerve of the parasympathetic nervous system that controls everything from digestion to mental health. A February 2022 pilot study suggested a link between vagus nerve dysfunction and some long COVID symptoms.
Vagus nerve stimulation is one form of treatment which involves using a device to stimulate the vagus nerve with electrical impulses. Dr. Viswanathan has been using the treatment in patients with fatigue, brain fog, anxiety, and depression — results, she contends, have been positive.
“This is something tangible that we can offer to patients,” she said.
Curative treatments for long COVID remain elusive, but doctors have many more tools for symptom management than before, said Ziyad Al-Aly, MD, a global expert on long COVID and chief of research and development at the Veterans Affairs St. Louis Health Care System.
For example, physicians are using beta-blockers to treat postural tachycardia syndrome (POTS), a symptom of long COVID that happens when the heart rate increases rapidly after someone stands up or lies down. Beta-blockers, such as the off-label medication ivabradine, have been used clinically to control heart rate, according to a March 2022 study published in the journal HeartRhythm Case Reports.
“It’s not a cure, but beta-blockers can help patients manage their symptoms,” said Dr. Al-Aly.
Additionally, some patients respond well to low-dose naltrexone for the treatment of extreme fatigue associated with long COVID. A January 2024 article in the journal Clinical Therapeutics found that fatigue symptoms improved in patients taking the medication.
Dr. Al-Aly said doctors treating patients with long COVID are getting better at pinpointing the phenotype or manifestation of the condition and diagnosing a treatment accordingly. Treating long COVID fatigue is not the same as treating POTS or symptoms of headache and joint pain.
It’s still all about the management of symptoms and doctors lack any US Food and Drug Administration–approved medications specifically for the condition.
Clinical Trials Exploring New Therapies
Still, a number of large clinical trials currently underway may change that, said David F. Putrino, PhD, who runs the long COVID clinic at Mount Sinai Health System in New York City.
Two clinical trials headed by Dr. Putrino’s lab are looking into repurposing two HIV antivirals to see whether they affect the levels of circulating SARS-CoV-2 virus in the body that may cause long COVID. The hope is that the antivirals Truvada and maraviroc can reduce the «reactivation of latent virus» that, said Dr. Putrino, causes lingering long COVID symptoms.
Ongoing trials are looking into the promise of SARS-CoV-2 monoclonal antibodies, produced from cells made by cloning a unique white blood cell, as a treatment option. The trials are investigating whether these antibodies may similarly target viral reservoirs that are causing persistence of symptoms in some patients.
Other trials are underway through the National Institutes of Health (NIH) RECOVER initiative in which more than 17,000 patients are enrolled, the largest study of its kind, said Grace McComsey, MD.
Dr. McComsey, who leads the study at University Hospitals Health System in Cleveland, said that after following patients for up to 4 years researchers have gathered “a massive repository of information” they hope will help scientists crack the code of this very complex disease.
She and other RECOVER researchers have recently published studies on a variety of findings, reporting in February, for example, that COVID infections may trigger other autoimmune diseases such as rheumatoid arthritis and type 2 diabetes. Another recent finding showed that people with HIV are at a higher risk for complications due to acute COVID-19.
Lack of Urgency Holds Back Progress
Still, others like Dr. Al-Aly and Dr. Putrino felt that the initiative isn’t moving fast enough. Dr. Al-Aly said that the NIH needs to “get its act together” and do more for long COVID. In the future, he said that we need to double down on our efforts to expand funding and increase urgency to better understand the mechanism of disease, risk factors, and treatments, as well as societal and economic implications.
“We did trials for COVID-19 vaccines at warp speed, but we’re doing trials for long COVID at a snail’s pace,” he said.
Dr. Al-Aly is concerned about the chronic nature of the disease and how it affects patients down the line. His large-scale study published last month in the journal Science looked specifically at chronic fatigue syndrome triggered by the infection and its long-term impact on patients.
He’s concerned about the practical implications for people who are weighted down with symptoms for multiple years.
“Being fatigued and ill for a few months is one thing, but being at home for 5 years is a totally different ballgame.”
A version of this article first appeared on Medscape.com.
Four years ago in the spring of 2020, physicians and patients coined the term “long COVID” to describe a form of the viral infection from which recovery seemed impossible. (And the old nickname “long-haulers” seems so quaint now.)
What started as a pandemic that killed nearly 3 million people globally in 2020 alone would turn into a chronic disease causing a long list of symptoms — from extreme fatigue, to brain fog, tremors, nausea, headaches, rapid heartbeat, and more.
Today, 6.4% of Americans report symptoms of long COVID, and many have never recovered.
Still, we’ve come a long way, although there’s much we don’t understand about the condition. At the very least, physicians have a greater understanding that long COVID exists and can cause serious long-term symptoms.
While physicians may not have a blanket diagnostic tool that works for all patients with long COVID, they have refined existing tests for more accurate results, said Nisha Viswanathan, MD, director of the University of California Los Angeles Long COVID Program at UCLA Health.
Also, a range of new treatments, now undergoing clinical trials, have emerged that have proved effective in managing long COVID symptoms.
Catecholamine testing, for example, is now commonly used to diagnose long COVID, particularly in those who have dysautonomia, a condition caused by dysfunction of the autonomic nervous system and marked by dizziness, low blood pressure, nausea, and brain fog.
Very high levels of the neurotransmitter, for example, were shown to indicate long COVID in a January 2021 study published in the journal Clinical Medicine.
Certain biomarkers have also been shown indicative of the condition, including low serotonin levels. A study published this year in Cell found lower serotonin levels in patients with long COVID driven by low levels of circulating SARS-CoV-2, the virus that causes the condition.
Still, said Dr. Viswanathan, long COVID is a disease diagnosed by figuring out what a patient does not have — by ruling out other causes — rather than what they do. “It’s still a moving target,” she said, meaning that the disease is always changing based on the variant of acute COVID.
Promising Treatments Have Emerged
Dysautonomia, and especially the associated brain fog, fatigue, and dizziness, are now common conditions. As a result, physicians have gotten better at treating them. The vagus nerve is the main nerve of the parasympathetic nervous system that controls everything from digestion to mental health. A February 2022 pilot study suggested a link between vagus nerve dysfunction and some long COVID symptoms.
Vagus nerve stimulation is one form of treatment which involves using a device to stimulate the vagus nerve with electrical impulses. Dr. Viswanathan has been using the treatment in patients with fatigue, brain fog, anxiety, and depression — results, she contends, have been positive.
“This is something tangible that we can offer to patients,” she said.
Curative treatments for long COVID remain elusive, but doctors have many more tools for symptom management than before, said Ziyad Al-Aly, MD, a global expert on long COVID and chief of research and development at the Veterans Affairs St. Louis Health Care System.
For example, physicians are using beta-blockers to treat postural tachycardia syndrome (POTS), a symptom of long COVID that happens when the heart rate increases rapidly after someone stands up or lies down. Beta-blockers, such as the off-label medication ivabradine, have been used clinically to control heart rate, according to a March 2022 study published in the journal HeartRhythm Case Reports.
“It’s not a cure, but beta-blockers can help patients manage their symptoms,” said Dr. Al-Aly.
Additionally, some patients respond well to low-dose naltrexone for the treatment of extreme fatigue associated with long COVID. A January 2024 article in the journal Clinical Therapeutics found that fatigue symptoms improved in patients taking the medication.
Dr. Al-Aly said doctors treating patients with long COVID are getting better at pinpointing the phenotype or manifestation of the condition and diagnosing a treatment accordingly. Treating long COVID fatigue is not the same as treating POTS or symptoms of headache and joint pain.
It’s still all about the management of symptoms and doctors lack any US Food and Drug Administration–approved medications specifically for the condition.
Clinical Trials Exploring New Therapies
Still, a number of large clinical trials currently underway may change that, said David F. Putrino, PhD, who runs the long COVID clinic at Mount Sinai Health System in New York City.
Two clinical trials headed by Dr. Putrino’s lab are looking into repurposing two HIV antivirals to see whether they affect the levels of circulating SARS-CoV-2 virus in the body that may cause long COVID. The hope is that the antivirals Truvada and maraviroc can reduce the «reactivation of latent virus» that, said Dr. Putrino, causes lingering long COVID symptoms.
Ongoing trials are looking into the promise of SARS-CoV-2 monoclonal antibodies, produced from cells made by cloning a unique white blood cell, as a treatment option. The trials are investigating whether these antibodies may similarly target viral reservoirs that are causing persistence of symptoms in some patients.
Other trials are underway through the National Institutes of Health (NIH) RECOVER initiative in which more than 17,000 patients are enrolled, the largest study of its kind, said Grace McComsey, MD.
Dr. McComsey, who leads the study at University Hospitals Health System in Cleveland, said that after following patients for up to 4 years researchers have gathered “a massive repository of information” they hope will help scientists crack the code of this very complex disease.
She and other RECOVER researchers have recently published studies on a variety of findings, reporting in February, for example, that COVID infections may trigger other autoimmune diseases such as rheumatoid arthritis and type 2 diabetes. Another recent finding showed that people with HIV are at a higher risk for complications due to acute COVID-19.
Lack of Urgency Holds Back Progress
Still, others like Dr. Al-Aly and Dr. Putrino felt that the initiative isn’t moving fast enough. Dr. Al-Aly said that the NIH needs to “get its act together” and do more for long COVID. In the future, he said that we need to double down on our efforts to expand funding and increase urgency to better understand the mechanism of disease, risk factors, and treatments, as well as societal and economic implications.
“We did trials for COVID-19 vaccines at warp speed, but we’re doing trials for long COVID at a snail’s pace,” he said.
Dr. Al-Aly is concerned about the chronic nature of the disease and how it affects patients down the line. His large-scale study published last month in the journal Science looked specifically at chronic fatigue syndrome triggered by the infection and its long-term impact on patients.
He’s concerned about the practical implications for people who are weighted down with symptoms for multiple years.
“Being fatigued and ill for a few months is one thing, but being at home for 5 years is a totally different ballgame.”
A version of this article first appeared on Medscape.com.
Four years ago in the spring of 2020, physicians and patients coined the term “long COVID” to describe a form of the viral infection from which recovery seemed impossible. (And the old nickname “long-haulers” seems so quaint now.)
What started as a pandemic that killed nearly 3 million people globally in 2020 alone would turn into a chronic disease causing a long list of symptoms — from extreme fatigue, to brain fog, tremors, nausea, headaches, rapid heartbeat, and more.
Today, 6.4% of Americans report symptoms of long COVID, and many have never recovered.
Still, we’ve come a long way, although there’s much we don’t understand about the condition. At the very least, physicians have a greater understanding that long COVID exists and can cause serious long-term symptoms.
While physicians may not have a blanket diagnostic tool that works for all patients with long COVID, they have refined existing tests for more accurate results, said Nisha Viswanathan, MD, director of the University of California Los Angeles Long COVID Program at UCLA Health.
Also, a range of new treatments, now undergoing clinical trials, have emerged that have proved effective in managing long COVID symptoms.
Catecholamine testing, for example, is now commonly used to diagnose long COVID, particularly in those who have dysautonomia, a condition caused by dysfunction of the autonomic nervous system and marked by dizziness, low blood pressure, nausea, and brain fog.
Very high levels of the neurotransmitter, for example, were shown to indicate long COVID in a January 2021 study published in the journal Clinical Medicine.
Certain biomarkers have also been shown indicative of the condition, including low serotonin levels. A study published this year in Cell found lower serotonin levels in patients with long COVID driven by low levels of circulating SARS-CoV-2, the virus that causes the condition.
Still, said Dr. Viswanathan, long COVID is a disease diagnosed by figuring out what a patient does not have — by ruling out other causes — rather than what they do. “It’s still a moving target,” she said, meaning that the disease is always changing based on the variant of acute COVID.
Promising Treatments Have Emerged
Dysautonomia, and especially the associated brain fog, fatigue, and dizziness, are now common conditions. As a result, physicians have gotten better at treating them. The vagus nerve is the main nerve of the parasympathetic nervous system that controls everything from digestion to mental health. A February 2022 pilot study suggested a link between vagus nerve dysfunction and some long COVID symptoms.
Vagus nerve stimulation is one form of treatment which involves using a device to stimulate the vagus nerve with electrical impulses. Dr. Viswanathan has been using the treatment in patients with fatigue, brain fog, anxiety, and depression — results, she contends, have been positive.
“This is something tangible that we can offer to patients,” she said.
Curative treatments for long COVID remain elusive, but doctors have many more tools for symptom management than before, said Ziyad Al-Aly, MD, a global expert on long COVID and chief of research and development at the Veterans Affairs St. Louis Health Care System.
For example, physicians are using beta-blockers to treat postural tachycardia syndrome (POTS), a symptom of long COVID that happens when the heart rate increases rapidly after someone stands up or lies down. Beta-blockers, such as the off-label medication ivabradine, have been used clinically to control heart rate, according to a March 2022 study published in the journal HeartRhythm Case Reports.
“It’s not a cure, but beta-blockers can help patients manage their symptoms,” said Dr. Al-Aly.
Additionally, some patients respond well to low-dose naltrexone for the treatment of extreme fatigue associated with long COVID. A January 2024 article in the journal Clinical Therapeutics found that fatigue symptoms improved in patients taking the medication.
Dr. Al-Aly said doctors treating patients with long COVID are getting better at pinpointing the phenotype or manifestation of the condition and diagnosing a treatment accordingly. Treating long COVID fatigue is not the same as treating POTS or symptoms of headache and joint pain.
It’s still all about the management of symptoms and doctors lack any US Food and Drug Administration–approved medications specifically for the condition.
Clinical Trials Exploring New Therapies
Still, a number of large clinical trials currently underway may change that, said David F. Putrino, PhD, who runs the long COVID clinic at Mount Sinai Health System in New York City.
Two clinical trials headed by Dr. Putrino’s lab are looking into repurposing two HIV antivirals to see whether they affect the levels of circulating SARS-CoV-2 virus in the body that may cause long COVID. The hope is that the antivirals Truvada and maraviroc can reduce the «reactivation of latent virus» that, said Dr. Putrino, causes lingering long COVID symptoms.
Ongoing trials are looking into the promise of SARS-CoV-2 monoclonal antibodies, produced from cells made by cloning a unique white blood cell, as a treatment option. The trials are investigating whether these antibodies may similarly target viral reservoirs that are causing persistence of symptoms in some patients.
Other trials are underway through the National Institutes of Health (NIH) RECOVER initiative in which more than 17,000 patients are enrolled, the largest study of its kind, said Grace McComsey, MD.
Dr. McComsey, who leads the study at University Hospitals Health System in Cleveland, said that after following patients for up to 4 years researchers have gathered “a massive repository of information” they hope will help scientists crack the code of this very complex disease.
She and other RECOVER researchers have recently published studies on a variety of findings, reporting in February, for example, that COVID infections may trigger other autoimmune diseases such as rheumatoid arthritis and type 2 diabetes. Another recent finding showed that people with HIV are at a higher risk for complications due to acute COVID-19.
Lack of Urgency Holds Back Progress
Still, others like Dr. Al-Aly and Dr. Putrino felt that the initiative isn’t moving fast enough. Dr. Al-Aly said that the NIH needs to “get its act together” and do more for long COVID. In the future, he said that we need to double down on our efforts to expand funding and increase urgency to better understand the mechanism of disease, risk factors, and treatments, as well as societal and economic implications.
“We did trials for COVID-19 vaccines at warp speed, but we’re doing trials for long COVID at a snail’s pace,” he said.
Dr. Al-Aly is concerned about the chronic nature of the disease and how it affects patients down the line. His large-scale study published last month in the journal Science looked specifically at chronic fatigue syndrome triggered by the infection and its long-term impact on patients.
He’s concerned about the practical implications for people who are weighted down with symptoms for multiple years.
“Being fatigued and ill for a few months is one thing, but being at home for 5 years is a totally different ballgame.”
A version of this article first appeared on Medscape.com.
For Some MDs, Long COVID Burnout Is a New Reality
Dhaval Desai, MD, was teaching his 4-year-old to ride a bike after another exhausting shift at the hospital during the summer after the first COVID-19 surge. He was putting on a happy face and forcing out a “Yay!” he did not feel. The pandemic had taken its toll, and he just wanted to lie down and be alone. Realizing that he was “scraping to find joy” was when he knew something was wrong.
“I was giving, giving, giving at work a lot, and I had little left to give at home,” said Dr. Desai, director of hospital medicine at Emory Saint Joseph’s Hospital and an assistant professor of medicine at Emory University in Atlanta, Georgia.
At work, he worried about his wife managing two kids — including a newborn — during the pandemic. At home, he stressed about work and the crush of patients with COVID the hospital was grappling to handle. He was exhausted, resentful, and angry, and it was jeopardizing what mattered most to him: His home life.
“It was all colliding…I realized, OK, I’m struggling,” he said.
Dr. Desai is one of thousands of physicians across the United States who have experienced burnout and depression, exacerbated by the pandemic. After 4 years, the impact is still being felt. Medscape’s 2024 annual report on this issue found that burnout and depression among doctors — while encouragingly better than the prior year — remain higher than before COVID. For doctors caring for patients with long COVID, those suffering from the debilitating aftereffects of an infection, the sense of helplessness when recovery is elusive can also weigh heavily.
Overall, more female physicians reported feeling burned out and depressed. Experts attributed this gap to issues including fewer women in supportive leadership and mentoring roles, compensation disparities, fewer career advancement opportunities, and more responsibilities caring for children and elders.
Multiple international studies and reports have highlighted the surge in burnout experienced by physicians and healthcare workers globally during the pandemic. Even before COVID, studies found the suicide rate among male and female US physicians was higher than the general population and higher than any other profession, including the military. The risk among female physicians, in particular, was 250%-400% higher.
“That’s really, on average, one a day, and that’s really unacceptable. No one should die by suicide, but a physician who knows the risks and knows that, should never do that,” said Dr. Desai about suicides overall among doctors.
The story of Lorna Breen had rattled Dr. Desai. Dr. Breen was a Manhattan physician who died by suicide in April 2020 after grappling with the city’s devastating first wave and then contracting COVID-19 herself. While Dr. Desai did not have thoughts of suicide, he was facing his own battles. Those experiences and the stigma around mental health prompted him to write his book, Burning Out on the Covid Front Lines: A Doctor’s Memoir of Fatherhood, Race and Perseverance in the Pandemic, with the hope that it can help others like him.
Mental Health Stigma
But despite the body of research and growing awareness toward addressing mental health among physicians, almost four in 10 doctors are wary of revealing their personal struggles.
More than half of those surveyed in the Medscape Medical News report said they had not consulted a mental health professional before and would not do so going forward either. The fear of tarnishing their reputation or even losing their license keeps doctors silent. Advocates and groups like the Dr. Lorna Breen Heroes’ Foundation are pushing for hospitals and healthcare systems to remove and rephrase invasive and stigmatizing language around mental health in licensure, credentialing, or insurance applications.
Burnout Triggers: Systemic Problems, Social Tensions
Burnout can make a person feel “depleted and used up” and is characterized by extreme tiredness, low energy, frustration about work, emotional distance or numbness, and difficulty with concentration, responsibilities, or creativity. It can make an individual feel helpless, alone, defeated, cynical, and without purpose and can also cause physical symptoms such as headaches, loss of appetite, insomnia, and body aches. Unaddressed, it can lead to depression, anxiety, and a variety of physical health issues.
“We can still be highly functional and not okay,” said Dr. Desai.
For doctors, burnout often builds over time from large and small systemic problems and inefficiencies, multiplied by a dozen or more patients each day: Not enough time for documentation, complicated paperwork, navigating byzantine health and insurance systems, and hitting roadblocks. The administrative work, combined with an enormous patient load, and staffing and resource shortages create barriers to care and cuts into the amount of time they can spend providing actual care.
These existing problems worsened as patients with COVID overwhelmed hospitals and clinics. At the same time, healthcare workers worried about caring for the sick, getting infected themselves, or having multiple staff falling ill at once. As each surge came and went, backlash, hostility, abuse, and even violence toward healthcare workers also increased. The discrimination some medical staff were subjected to compounded the burnout.
“When we’re not getting the support we need as physicians and healthcare workers, that adds to burnout, and I saw that in my colleagues,” said Dr. Desai.
Impact of Burnout
At the Mount Sinai Center for Post-COVID Care in New York City, doctors grapple with feelings of helplessness in caring for patients with long COVID who show little sign of recovery. That emotional toll can also be difficult, said director Zijian Chen, MD, who helped launch the clinic in May 2020.
“Sometimes you’re faced with patients who you’re trying to do everything for, but they’re not just not getting better,” said Dr. Chen. “It’s really frustrating because we want everybody to get better. So, there’s that lack of fulfillment there that can cause a sense of burnout.”
While the worst outcomes and death rates initially brought on by acute infections have lessened, long COVID clinics exemplify some of the ongoing challenges within healthcare. Many operate with insufficient financial and staffing resources despite wait-lists and a steady flow of new and returning patients. Even with the demand, a number of these clinics have shuttered, leaving patients without access to much-needed medical help.
“There are clinicians who are burning out. That is definitely something that I’ve seen,” said Monica Verduzco-Gutierrez, MD, a professor and chair of the Department of Rehabilitation Medicine at the University of Texas Health Science Center in San Antonio, Texas.
“[It] takes a lot of resources for a successful long COVID clinic. A lot of special funding may be drying up and couple that with clinicians burning out, then they’re going to shut their doors.”
And it’s not just long COVID clinics. Data have shown an overall exodus in healthcare, especially during the pandemic. One study found burnout was one of the “most impactful” predictors of a physician’s intention to leave the profession during the pandemic. The loss of talent and skills during a major health crisis can put the entire system under stress, with patients ultimately suffering from poorer care.
“Healthcare system fragility and the chaos is far worse than it was before. We are continuing to be forced to do more with less,” said Dr. Desai.
Alleviating Burnout
While it is difficult to assess whether burnout from the pandemic is transient, experts say this is an opportunity for health institutions to learn from these experiences and implement policies and actions that can help reduce the mental health strain on staff. One study found that changes made by organizations had a bigger positive impact on reducing burnout than individual changes.
Advocates say more support staff, more work flexibility, and higher compensation would significantly ease the burden that drives burnout and depression.
In addition, half the physicians surveyed in the Medscape Medical News report felt their employers were not acknowledging how pervasive burnout is at their workplace. Having a trusted peer or leader set an example by sharing his or her own challenging experiences and saying it›s time to address these struggles can be an enormously validating step forward, said Dr. Desai. Acknowledging his own difficulties was not only a huge weight off his shoulders but also helped surpris colleagues who sought him out for counsel.
“I’m not suggesting everybody get on medication,” he said. “But talking to a therapist, acknowledging there’s issues, restructuring your life to realize something’s off, and just knowing that you’re not alone? That’s huge.”
Dr. Desai said he still faces personal challenges but is in a much better place, doing well at work and at home. He talks to a therapist, is taking medication, and has developed better coping mechanisms. He is spending more time with his family, detaching for a few hours from work-related emails, learning to draw boundaries and say no, and trying to be more present and “intentional” in connecting with colleagues and patients.
“It’s okay to not be okay,” said Dr. Desai. “It’s okay to be vulnerable and acknowledge when we can’t do more.”
Are you in a crisis? Call or text 988 or text TALK to 741741. For immediate support for healthcare professionals, as well as resources for institutions and organizations, visit: afsp.org/suicide-prevention-for-healthcare-professionals/#facts-about-mental-health-and-suicide.
A version of this article appeared on Medscape.com.
Dhaval Desai, MD, was teaching his 4-year-old to ride a bike after another exhausting shift at the hospital during the summer after the first COVID-19 surge. He was putting on a happy face and forcing out a “Yay!” he did not feel. The pandemic had taken its toll, and he just wanted to lie down and be alone. Realizing that he was “scraping to find joy” was when he knew something was wrong.
“I was giving, giving, giving at work a lot, and I had little left to give at home,” said Dr. Desai, director of hospital medicine at Emory Saint Joseph’s Hospital and an assistant professor of medicine at Emory University in Atlanta, Georgia.
At work, he worried about his wife managing two kids — including a newborn — during the pandemic. At home, he stressed about work and the crush of patients with COVID the hospital was grappling to handle. He was exhausted, resentful, and angry, and it was jeopardizing what mattered most to him: His home life.
“It was all colliding…I realized, OK, I’m struggling,” he said.
Dr. Desai is one of thousands of physicians across the United States who have experienced burnout and depression, exacerbated by the pandemic. After 4 years, the impact is still being felt. Medscape’s 2024 annual report on this issue found that burnout and depression among doctors — while encouragingly better than the prior year — remain higher than before COVID. For doctors caring for patients with long COVID, those suffering from the debilitating aftereffects of an infection, the sense of helplessness when recovery is elusive can also weigh heavily.
Overall, more female physicians reported feeling burned out and depressed. Experts attributed this gap to issues including fewer women in supportive leadership and mentoring roles, compensation disparities, fewer career advancement opportunities, and more responsibilities caring for children and elders.
Multiple international studies and reports have highlighted the surge in burnout experienced by physicians and healthcare workers globally during the pandemic. Even before COVID, studies found the suicide rate among male and female US physicians was higher than the general population and higher than any other profession, including the military. The risk among female physicians, in particular, was 250%-400% higher.
“That’s really, on average, one a day, and that’s really unacceptable. No one should die by suicide, but a physician who knows the risks and knows that, should never do that,” said Dr. Desai about suicides overall among doctors.
The story of Lorna Breen had rattled Dr. Desai. Dr. Breen was a Manhattan physician who died by suicide in April 2020 after grappling with the city’s devastating first wave and then contracting COVID-19 herself. While Dr. Desai did not have thoughts of suicide, he was facing his own battles. Those experiences and the stigma around mental health prompted him to write his book, Burning Out on the Covid Front Lines: A Doctor’s Memoir of Fatherhood, Race and Perseverance in the Pandemic, with the hope that it can help others like him.
Mental Health Stigma
But despite the body of research and growing awareness toward addressing mental health among physicians, almost four in 10 doctors are wary of revealing their personal struggles.
More than half of those surveyed in the Medscape Medical News report said they had not consulted a mental health professional before and would not do so going forward either. The fear of tarnishing their reputation or even losing their license keeps doctors silent. Advocates and groups like the Dr. Lorna Breen Heroes’ Foundation are pushing for hospitals and healthcare systems to remove and rephrase invasive and stigmatizing language around mental health in licensure, credentialing, or insurance applications.
Burnout Triggers: Systemic Problems, Social Tensions
Burnout can make a person feel “depleted and used up” and is characterized by extreme tiredness, low energy, frustration about work, emotional distance or numbness, and difficulty with concentration, responsibilities, or creativity. It can make an individual feel helpless, alone, defeated, cynical, and without purpose and can also cause physical symptoms such as headaches, loss of appetite, insomnia, and body aches. Unaddressed, it can lead to depression, anxiety, and a variety of physical health issues.
“We can still be highly functional and not okay,” said Dr. Desai.
For doctors, burnout often builds over time from large and small systemic problems and inefficiencies, multiplied by a dozen or more patients each day: Not enough time for documentation, complicated paperwork, navigating byzantine health and insurance systems, and hitting roadblocks. The administrative work, combined with an enormous patient load, and staffing and resource shortages create barriers to care and cuts into the amount of time they can spend providing actual care.
These existing problems worsened as patients with COVID overwhelmed hospitals and clinics. At the same time, healthcare workers worried about caring for the sick, getting infected themselves, or having multiple staff falling ill at once. As each surge came and went, backlash, hostility, abuse, and even violence toward healthcare workers also increased. The discrimination some medical staff were subjected to compounded the burnout.
“When we’re not getting the support we need as physicians and healthcare workers, that adds to burnout, and I saw that in my colleagues,” said Dr. Desai.
Impact of Burnout
At the Mount Sinai Center for Post-COVID Care in New York City, doctors grapple with feelings of helplessness in caring for patients with long COVID who show little sign of recovery. That emotional toll can also be difficult, said director Zijian Chen, MD, who helped launch the clinic in May 2020.
“Sometimes you’re faced with patients who you’re trying to do everything for, but they’re not just not getting better,” said Dr. Chen. “It’s really frustrating because we want everybody to get better. So, there’s that lack of fulfillment there that can cause a sense of burnout.”
While the worst outcomes and death rates initially brought on by acute infections have lessened, long COVID clinics exemplify some of the ongoing challenges within healthcare. Many operate with insufficient financial and staffing resources despite wait-lists and a steady flow of new and returning patients. Even with the demand, a number of these clinics have shuttered, leaving patients without access to much-needed medical help.
“There are clinicians who are burning out. That is definitely something that I’ve seen,” said Monica Verduzco-Gutierrez, MD, a professor and chair of the Department of Rehabilitation Medicine at the University of Texas Health Science Center in San Antonio, Texas.
“[It] takes a lot of resources for a successful long COVID clinic. A lot of special funding may be drying up and couple that with clinicians burning out, then they’re going to shut their doors.”
And it’s not just long COVID clinics. Data have shown an overall exodus in healthcare, especially during the pandemic. One study found burnout was one of the “most impactful” predictors of a physician’s intention to leave the profession during the pandemic. The loss of talent and skills during a major health crisis can put the entire system under stress, with patients ultimately suffering from poorer care.
“Healthcare system fragility and the chaos is far worse than it was before. We are continuing to be forced to do more with less,” said Dr. Desai.
Alleviating Burnout
While it is difficult to assess whether burnout from the pandemic is transient, experts say this is an opportunity for health institutions to learn from these experiences and implement policies and actions that can help reduce the mental health strain on staff. One study found that changes made by organizations had a bigger positive impact on reducing burnout than individual changes.
Advocates say more support staff, more work flexibility, and higher compensation would significantly ease the burden that drives burnout and depression.
In addition, half the physicians surveyed in the Medscape Medical News report felt their employers were not acknowledging how pervasive burnout is at their workplace. Having a trusted peer or leader set an example by sharing his or her own challenging experiences and saying it›s time to address these struggles can be an enormously validating step forward, said Dr. Desai. Acknowledging his own difficulties was not only a huge weight off his shoulders but also helped surpris colleagues who sought him out for counsel.
“I’m not suggesting everybody get on medication,” he said. “But talking to a therapist, acknowledging there’s issues, restructuring your life to realize something’s off, and just knowing that you’re not alone? That’s huge.”
Dr. Desai said he still faces personal challenges but is in a much better place, doing well at work and at home. He talks to a therapist, is taking medication, and has developed better coping mechanisms. He is spending more time with his family, detaching for a few hours from work-related emails, learning to draw boundaries and say no, and trying to be more present and “intentional” in connecting with colleagues and patients.
“It’s okay to not be okay,” said Dr. Desai. “It’s okay to be vulnerable and acknowledge when we can’t do more.”
Are you in a crisis? Call or text 988 or text TALK to 741741. For immediate support for healthcare professionals, as well as resources for institutions and organizations, visit: afsp.org/suicide-prevention-for-healthcare-professionals/#facts-about-mental-health-and-suicide.
A version of this article appeared on Medscape.com.
Dhaval Desai, MD, was teaching his 4-year-old to ride a bike after another exhausting shift at the hospital during the summer after the first COVID-19 surge. He was putting on a happy face and forcing out a “Yay!” he did not feel. The pandemic had taken its toll, and he just wanted to lie down and be alone. Realizing that he was “scraping to find joy” was when he knew something was wrong.
“I was giving, giving, giving at work a lot, and I had little left to give at home,” said Dr. Desai, director of hospital medicine at Emory Saint Joseph’s Hospital and an assistant professor of medicine at Emory University in Atlanta, Georgia.
At work, he worried about his wife managing two kids — including a newborn — during the pandemic. At home, he stressed about work and the crush of patients with COVID the hospital was grappling to handle. He was exhausted, resentful, and angry, and it was jeopardizing what mattered most to him: His home life.
“It was all colliding…I realized, OK, I’m struggling,” he said.
Dr. Desai is one of thousands of physicians across the United States who have experienced burnout and depression, exacerbated by the pandemic. After 4 years, the impact is still being felt. Medscape’s 2024 annual report on this issue found that burnout and depression among doctors — while encouragingly better than the prior year — remain higher than before COVID. For doctors caring for patients with long COVID, those suffering from the debilitating aftereffects of an infection, the sense of helplessness when recovery is elusive can also weigh heavily.
Overall, more female physicians reported feeling burned out and depressed. Experts attributed this gap to issues including fewer women in supportive leadership and mentoring roles, compensation disparities, fewer career advancement opportunities, and more responsibilities caring for children and elders.
Multiple international studies and reports have highlighted the surge in burnout experienced by physicians and healthcare workers globally during the pandemic. Even before COVID, studies found the suicide rate among male and female US physicians was higher than the general population and higher than any other profession, including the military. The risk among female physicians, in particular, was 250%-400% higher.
“That’s really, on average, one a day, and that’s really unacceptable. No one should die by suicide, but a physician who knows the risks and knows that, should never do that,” said Dr. Desai about suicides overall among doctors.
The story of Lorna Breen had rattled Dr. Desai. Dr. Breen was a Manhattan physician who died by suicide in April 2020 after grappling with the city’s devastating first wave and then contracting COVID-19 herself. While Dr. Desai did not have thoughts of suicide, he was facing his own battles. Those experiences and the stigma around mental health prompted him to write his book, Burning Out on the Covid Front Lines: A Doctor’s Memoir of Fatherhood, Race and Perseverance in the Pandemic, with the hope that it can help others like him.
Mental Health Stigma
But despite the body of research and growing awareness toward addressing mental health among physicians, almost four in 10 doctors are wary of revealing their personal struggles.
More than half of those surveyed in the Medscape Medical News report said they had not consulted a mental health professional before and would not do so going forward either. The fear of tarnishing their reputation or even losing their license keeps doctors silent. Advocates and groups like the Dr. Lorna Breen Heroes’ Foundation are pushing for hospitals and healthcare systems to remove and rephrase invasive and stigmatizing language around mental health in licensure, credentialing, or insurance applications.
Burnout Triggers: Systemic Problems, Social Tensions
Burnout can make a person feel “depleted and used up” and is characterized by extreme tiredness, low energy, frustration about work, emotional distance or numbness, and difficulty with concentration, responsibilities, or creativity. It can make an individual feel helpless, alone, defeated, cynical, and without purpose and can also cause physical symptoms such as headaches, loss of appetite, insomnia, and body aches. Unaddressed, it can lead to depression, anxiety, and a variety of physical health issues.
“We can still be highly functional and not okay,” said Dr. Desai.
For doctors, burnout often builds over time from large and small systemic problems and inefficiencies, multiplied by a dozen or more patients each day: Not enough time for documentation, complicated paperwork, navigating byzantine health and insurance systems, and hitting roadblocks. The administrative work, combined with an enormous patient load, and staffing and resource shortages create barriers to care and cuts into the amount of time they can spend providing actual care.
These existing problems worsened as patients with COVID overwhelmed hospitals and clinics. At the same time, healthcare workers worried about caring for the sick, getting infected themselves, or having multiple staff falling ill at once. As each surge came and went, backlash, hostility, abuse, and even violence toward healthcare workers also increased. The discrimination some medical staff were subjected to compounded the burnout.
“When we’re not getting the support we need as physicians and healthcare workers, that adds to burnout, and I saw that in my colleagues,” said Dr. Desai.
Impact of Burnout
At the Mount Sinai Center for Post-COVID Care in New York City, doctors grapple with feelings of helplessness in caring for patients with long COVID who show little sign of recovery. That emotional toll can also be difficult, said director Zijian Chen, MD, who helped launch the clinic in May 2020.
“Sometimes you’re faced with patients who you’re trying to do everything for, but they’re not just not getting better,” said Dr. Chen. “It’s really frustrating because we want everybody to get better. So, there’s that lack of fulfillment there that can cause a sense of burnout.”
While the worst outcomes and death rates initially brought on by acute infections have lessened, long COVID clinics exemplify some of the ongoing challenges within healthcare. Many operate with insufficient financial and staffing resources despite wait-lists and a steady flow of new and returning patients. Even with the demand, a number of these clinics have shuttered, leaving patients without access to much-needed medical help.
“There are clinicians who are burning out. That is definitely something that I’ve seen,” said Monica Verduzco-Gutierrez, MD, a professor and chair of the Department of Rehabilitation Medicine at the University of Texas Health Science Center in San Antonio, Texas.
“[It] takes a lot of resources for a successful long COVID clinic. A lot of special funding may be drying up and couple that with clinicians burning out, then they’re going to shut their doors.”
And it’s not just long COVID clinics. Data have shown an overall exodus in healthcare, especially during the pandemic. One study found burnout was one of the “most impactful” predictors of a physician’s intention to leave the profession during the pandemic. The loss of talent and skills during a major health crisis can put the entire system under stress, with patients ultimately suffering from poorer care.
“Healthcare system fragility and the chaos is far worse than it was before. We are continuing to be forced to do more with less,” said Dr. Desai.
Alleviating Burnout
While it is difficult to assess whether burnout from the pandemic is transient, experts say this is an opportunity for health institutions to learn from these experiences and implement policies and actions that can help reduce the mental health strain on staff. One study found that changes made by organizations had a bigger positive impact on reducing burnout than individual changes.
Advocates say more support staff, more work flexibility, and higher compensation would significantly ease the burden that drives burnout and depression.
In addition, half the physicians surveyed in the Medscape Medical News report felt their employers were not acknowledging how pervasive burnout is at their workplace. Having a trusted peer or leader set an example by sharing his or her own challenging experiences and saying it›s time to address these struggles can be an enormously validating step forward, said Dr. Desai. Acknowledging his own difficulties was not only a huge weight off his shoulders but also helped surpris colleagues who sought him out for counsel.
“I’m not suggesting everybody get on medication,” he said. “But talking to a therapist, acknowledging there’s issues, restructuring your life to realize something’s off, and just knowing that you’re not alone? That’s huge.”
Dr. Desai said he still faces personal challenges but is in a much better place, doing well at work and at home. He talks to a therapist, is taking medication, and has developed better coping mechanisms. He is spending more time with his family, detaching for a few hours from work-related emails, learning to draw boundaries and say no, and trying to be more present and “intentional” in connecting with colleagues and patients.
“It’s okay to not be okay,” said Dr. Desai. “It’s okay to be vulnerable and acknowledge when we can’t do more.”
Are you in a crisis? Call or text 988 or text TALK to 741741. For immediate support for healthcare professionals, as well as resources for institutions and organizations, visit: afsp.org/suicide-prevention-for-healthcare-professionals/#facts-about-mental-health-and-suicide.
A version of this article appeared on Medscape.com.