User login
A Reader Asks About Wild Ginger
Q: A friend of my teenage daughter was telling her about a new, “natural” weight-loss medication that contains wild ginger. Besides the obvious (a teen should not be taking “drugs” to lose weight), I seem to remember a problem with wild ginger. Is this substance dangerous?
A source of the herbal drug aristolochic acid (AA), wild ginger is a plant in the birthwort family. Its common name may be explained by the fact that it smells and tastes somewhat like ginger. However, wild ginger is not related to the common herb, ginger, that is found in the grocery store.
After multiple incidents of acute kidney failure in Europe, China, India, and the Balkans were linked to AA, the FDA sent out warning letters in 2001.5 However, as the saying goes, what goes around, comes around. A new wave of “natural” weight-loss remedies containing AA has become available for sale to unsuspecting consumers over the Internet.
Wild ginger plants grow in temperate regions, with a kidney-shaped leaf—ironic, since ingesting this substance can induce kidney failure. In an article published in 2012, researchers reported that a Chinese company had replaced Stephania tetrandra with Aristolochia fangchi in their weight-loss formula, resulting in multiple incidents of kidney failure.6 In 2013, investigators from London and Germany showed that products containing AA were commonly available online.7
Symptoms of AA ingestion include acute kidney failure with normal blood pressure; a normochromic, normocytic anemia with a moderate amount of urine protein excretion (< 1.5 g/d); and urine sediment with a few red blood cells. The serum creatininehas been reported anywhere from 1.4 to 12.7 mg/dL on presentation,7 but due to AA-associated reductions in fluid and food intake (ie, its “weight-loss” component), this can quickly progress to kidney failure.
Discontinuing use of the herbal remedy does not appear to stop users’ progression to kidney failure, as damage to the interstitial cells is already done by the time of presentation.
AA use is also associated with an increased incidence of both kidney cancer and urinary cancer. Quite a high-risk “natural” herbal remedy!
Kim Zuber, PA-C, MSPS, DFAAPA,Department Co-editor
References
5. Boyle B. FDA warns consumers to discontinue use of botanical products that contain aristolochic acid (2001). www.hcvadvocate.org/news/NewsUpdates_pdf/2.1.1.2_News_Review_Archive_2001/aristocholic.pdf. Accessed April 5, 2013.
6. Asif M. A brief study of toxic effects of some medicinal herbs on kidney. Adv Biomed Res. 2012;1:44.
7. Gökmen MR, Cosyns JP, Arlt VM, et al. The epidemiology, diagnosis, and management of aristolochic acid nephropathy: a narrative review. Ann Intern Med. 2013;158:469-477.
Q: A friend of my teenage daughter was telling her about a new, “natural” weight-loss medication that contains wild ginger. Besides the obvious (a teen should not be taking “drugs” to lose weight), I seem to remember a problem with wild ginger. Is this substance dangerous?
A source of the herbal drug aristolochic acid (AA), wild ginger is a plant in the birthwort family. Its common name may be explained by the fact that it smells and tastes somewhat like ginger. However, wild ginger is not related to the common herb, ginger, that is found in the grocery store.
After multiple incidents of acute kidney failure in Europe, China, India, and the Balkans were linked to AA, the FDA sent out warning letters in 2001.5 However, as the saying goes, what goes around, comes around. A new wave of “natural” weight-loss remedies containing AA has become available for sale to unsuspecting consumers over the Internet.
Wild ginger plants grow in temperate regions, with a kidney-shaped leaf—ironic, since ingesting this substance can induce kidney failure. In an article published in 2012, researchers reported that a Chinese company had replaced Stephania tetrandra with Aristolochia fangchi in their weight-loss formula, resulting in multiple incidents of kidney failure.6 In 2013, investigators from London and Germany showed that products containing AA were commonly available online.7
Symptoms of AA ingestion include acute kidney failure with normal blood pressure; a normochromic, normocytic anemia with a moderate amount of urine protein excretion (< 1.5 g/d); and urine sediment with a few red blood cells. The serum creatininehas been reported anywhere from 1.4 to 12.7 mg/dL on presentation,7 but due to AA-associated reductions in fluid and food intake (ie, its “weight-loss” component), this can quickly progress to kidney failure.
Discontinuing use of the herbal remedy does not appear to stop users’ progression to kidney failure, as damage to the interstitial cells is already done by the time of presentation.
AA use is also associated with an increased incidence of both kidney cancer and urinary cancer. Quite a high-risk “natural” herbal remedy!
Kim Zuber, PA-C, MSPS, DFAAPA,Department Co-editor
References
5. Boyle B. FDA warns consumers to discontinue use of botanical products that contain aristolochic acid (2001). www.hcvadvocate.org/news/NewsUpdates_pdf/2.1.1.2_News_Review_Archive_2001/aristocholic.pdf. Accessed April 5, 2013.
6. Asif M. A brief study of toxic effects of some medicinal herbs on kidney. Adv Biomed Res. 2012;1:44.
7. Gökmen MR, Cosyns JP, Arlt VM, et al. The epidemiology, diagnosis, and management of aristolochic acid nephropathy: a narrative review. Ann Intern Med. 2013;158:469-477.
Q: A friend of my teenage daughter was telling her about a new, “natural” weight-loss medication that contains wild ginger. Besides the obvious (a teen should not be taking “drugs” to lose weight), I seem to remember a problem with wild ginger. Is this substance dangerous?
A source of the herbal drug aristolochic acid (AA), wild ginger is a plant in the birthwort family. Its common name may be explained by the fact that it smells and tastes somewhat like ginger. However, wild ginger is not related to the common herb, ginger, that is found in the grocery store.
After multiple incidents of acute kidney failure in Europe, China, India, and the Balkans were linked to AA, the FDA sent out warning letters in 2001.5 However, as the saying goes, what goes around, comes around. A new wave of “natural” weight-loss remedies containing AA has become available for sale to unsuspecting consumers over the Internet.
Wild ginger plants grow in temperate regions, with a kidney-shaped leaf—ironic, since ingesting this substance can induce kidney failure. In an article published in 2012, researchers reported that a Chinese company had replaced Stephania tetrandra with Aristolochia fangchi in their weight-loss formula, resulting in multiple incidents of kidney failure.6 In 2013, investigators from London and Germany showed that products containing AA were commonly available online.7
Symptoms of AA ingestion include acute kidney failure with normal blood pressure; a normochromic, normocytic anemia with a moderate amount of urine protein excretion (< 1.5 g/d); and urine sediment with a few red blood cells. The serum creatininehas been reported anywhere from 1.4 to 12.7 mg/dL on presentation,7 but due to AA-associated reductions in fluid and food intake (ie, its “weight-loss” component), this can quickly progress to kidney failure.
Discontinuing use of the herbal remedy does not appear to stop users’ progression to kidney failure, as damage to the interstitial cells is already done by the time of presentation.
AA use is also associated with an increased incidence of both kidney cancer and urinary cancer. Quite a high-risk “natural” herbal remedy!
Kim Zuber, PA-C, MSPS, DFAAPA,Department Co-editor
References
5. Boyle B. FDA warns consumers to discontinue use of botanical products that contain aristolochic acid (2001). www.hcvadvocate.org/news/NewsUpdates_pdf/2.1.1.2_News_Review_Archive_2001/aristocholic.pdf. Accessed April 5, 2013.
6. Asif M. A brief study of toxic effects of some medicinal herbs on kidney. Adv Biomed Res. 2012;1:44.
7. Gökmen MR, Cosyns JP, Arlt VM, et al. The epidemiology, diagnosis, and management of aristolochic acid nephropathy: a narrative review. Ann Intern Med. 2013;158:469-477.
Interstitial Cystitis: A Painful Syndrome
CE/CME No: CR-1307
PROGRAM OVERVIEW
Earn credit by reading this article and successfully completing the posttest. Successful completion is defined as a cumulative score of at least 70% correct.
EDUCATIONAL OBJECTIVES
•
Describe the pathophysiology
of interstitial cystitis/bladder
pain syndrome (IC/BPS), as
it is currently understood.
•
Discuss urogenital signs and
symptoms that should prompt suspicion for IC/BPS in a primary care patient.
•
Explain the clinical diagnosis of
IC/BPS and key considerations for referral.
•
Review medical management, nonoperative therapy, and surgical treatment of IC/BPS.
FACULTY
LaToya M. Haynes practices at the Carolinas Pain Institute and the Center for Clinical Research in Winston-Salem, North Carolina, and is a preceptor for PA students. Kelly Bilello is a PA at Genitourinary Surgical Consultants in Denver. Jade Breeback practices at Cone Health Primary Care in Kernersville, North Carolina. Jessica Cain is a PA in emergency medicine at the University of Cincinnati Medical Center. Jennifer Wenninger is a cardiothoracic and vascular surgery PA at Bellin Health Care Systems in Green Bay, Wisconsin. M. Jane McDaniel is an Instructor in the Department of Physician Assistant Studies at Wake Forest School of Medicine in Winston-Salem.
The authors have no significant financial relationships to disclose.
ACCREDITATION STATEMENT
Article begins on next page >>
Interstitial cystitis/bladder pain syndrome (IC/BPS) is a common, painful disease of the urinary bladder. Difficult to diagnose and frequently misdiagnosed as another common urologic disorder, IC/BPS challenges health care providers to identify it early and implement current treatment algorithms that may simplify management and improve quality of life for affected patients.
Interstitial cystitis (IC), or bladder pain syndrome (BPS), is a clinical condition characterized by bladder pain, urinary frequency and urgency, and increased nighttime urination (nocturia).1 More specifically, IC/BPS is defined as an unpleasant sensation in the bladder, abdomen, or pelvis (ie, pain, pressure, burning, and/or other discomfort) perceived to be originating in the urinary bladder. The condition is associated with lower urinary tract symptoms of more than six weeks’ duration, with no infection or other identifiable cause present.2
IC/BPS lacks a single known etiology; rather, it most likely results from multiple contributing factors that cascade into a painful and potentially debilitating syndrome. The condition was first described more than a century ago,3,4 but its complex nature and conflicting theories about its pathogenesis present both diagnostic and therapeutic challenges for health care professionals. Frequent misdiagnosis of IC/BPS as another common urologic disorder can make timely, appropriate treatment elusive.
Without a clearly described pathophysiology, IC/BPS has always been difficult to define using standardized diagnostic criteria and precise terminology. The definition of the condition was revised in 2002 and again in 2008, when the nomenclature bladder pain syndrome was introduced.1,5,6
Less than 10 years ago, US researchers described IC as a subgroup of BPS,7 while in Europe, BPS is used as the broader term, with IC still considered a well-defined subgroup that usually involves ulceration.6 The future may find IC, BPS, and painful bladder syndrome (PBS) used as interchangeable terms—or as unique diagnoses. A better understanding of the pathophysiology of IC/BPS/PBS would contribute not only to resolving issues of nomenclature, but also to establishing an accurate diagnosis earlier in the disease process and providing more efficient, effective treatment.
THE PROBLEM OF EPIDEMIOLOGY
Inconsistencies in the terminology, definitions, and diagnostic criteria of IC/BPS have made epidemiology difficult to establish.1 It has been suggested that IC/BPS is underdiagnosed in the United States and that its prevalence is much greater than generally reported.8
According to one study of IC in a managed care population, its prevalence in 2005 was 197 per 100,000 women and 41 per 100,000 men, with the female-to-male ratio estimated at 5:1.9 In 2011, researchers for the RAND Corporation published what they called the first population-based “symptom prevalence estimate” among US women older than 18, based on more than 100,000 screening interviews conducted by phone. According to their findings, between 3.3 and 7.9 million US women meet the stated criteria for IC/BPS (ie, between 3,113 and 7,453 women per 100,000).10 These conflicting data exemplify the range of epidemiologic conclusions that exist regarding this condition.
On the next page: Proposed pathophysiology >>
THE PROPOSED PATHOPHYSIOLOGY
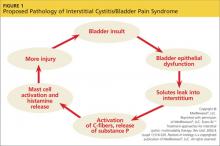
IC/BPS is thought to begin with an initial insult to the bladder that leads to dysfunction of the epithelial layer. This insult may be the result of a neurogenic inflammation, autoimmunity, subclinical or chronic infection, or bladder urothelial defects.1 Dysfunction in the epithelial layer includes altered bladder epithelial expression of human leukocyte antigen I and II; decreased expression of uroplakin (an antitoxic protein in the bladder), and a defective glycosaminoglycan mucus layer.4 This damage to the epithelial layer alters the permeability of the bladder, allowing potassium ions to enter the urothelium and depolarize motor and sensory nerves. This potassium leak then activates the mast cells, causing mastocytosis and the release of histamine.11 These processes disrupt the homeostasis of the urinary tract and allow the development of inflammation—a main cause of the pelvic pain associated with IC/BPS4,12,13 (see Figure 114).
Other factors that exacerbate the primary inflammation in the bladder are C-fibers and nerve growth factor (NGF). C-fibers are afferent fibers found in the peripheral nerves of the somatic sensory system that convey input signals from the periphery to the central nervous system.3 In patients with IC/BPS, initial inflammation activates C-fibers, which produce substance P, nociceptor, and other inflammatory mediators. These mediators exacerbate existing inflammation and further facilitate mast cell activation.3
NGF is a protein that is critical for the maintenance of sympathetic and sensory neurons; it is important not only in the urinary tract but in all organ systems. Increased levels of NGF, a prevalent finding in patients with IC/BPS, is an indicator of inflammation in the body. The precise mechanism that causes elevated NGF in patients with IC/BPS is not well understood, but its presence supports the theory that inflammation is a cause of pelvic pain in IC/BPS.12
The urinary urgency and frequency experienced by patients with IC/BPS is in part due to the role nitric oxide (NO) plays in bladder activity. Patients with IC have decreased levels of urinary NO (a reduction thought to be the result of a decrease in L-arginine) and urinary NO synthase.12,15,16 Ordinarily, NO synthase converts L-arginine to NO, which helps to control relaxation of the bladder smooth muscle, allowing more urine to be stored. In patients with IC/BPS, NO insufficiency leads to bladder overactivity.15
On the next page: Patient history and presentation >>
PATIENT HISTORY AND PRESENTATION
A detailed patient history is imperative in establishing the diagnosis of IC/BPS. Symptoms that should prompt the clinician to consider IC/BPS include:
• Pelvic or bladder pain relieved with voiding
• Dyspareunia
• Increased frequency of urination with no infection present
• Urinary urgency with pain, and
• Increased nocturia.17,18
Early IC presents variably, and pain, though a common symptom, is not always present.19 Chronic pain is defined by duration of at least six months, with the discomfort perceived as originating in the bladder.8 In addition to patients who experience pain, those who void several times during the night should also be considered for further evaluation.19
Many patients describe their symptoms in terms of flares and periods of remission. Some patients associate flares with stress, seasonal allergies, sexual activity, consumption of certain foods, and the premenstrual week.17,20 Patients with IC/BPS are commonly misdiagnosed with recurrent urinary tract infections; hence the need to standardize the criteria for diagnosis of IC/BPS.17
DIAGNOSIS
There are currently three available sets of diagnostic criteria for patients with IC/BPS. These are the National Institute for Diabetes and Diseases of the Kidney (NIDDK) definition (1990),21 the International Continence Society (ICS) definition of painful bladder disorders (2002),5 and the European Society for the Study of IC/BPS (ESSIC) definition (2008).6 In particular, the ESSIC criteria were formulated to help identify IC/BPS earlier in the disease course.
The 1990 NIDDK protocol, developed for research purposes,12 featured inclusion and exclusion criteria. Exclusion criteria included age younger than 18 years and presence of benign bladder tumors, radiation cystitis, tuberculosis cystitis, bacterial cystitis, vaginitis, symptomatic urethral diverticulum, uterine/cervical/vaginal cancers, and/or active herpes; urinary frequency of less than five episodes in 12 hours; and less than two episodes of nocturia per night.19
NIDDK inclusion criteria required two or more of the following: Hunner’s ulcer, pain on bladder filling, general pelvic pain, glomerulations on endoscopy, and decreased bladder compliance on cystometrogram.19
This protocol proved to be excessively restrictive for clinical use and was widely replaced by the ICS criteria in 2002. The ICS criteria5 allowed more varied patient presentations; the exclusions featured in the NIDDK guideline, it has been estimated, could have eliminated at least one-third of patients who would reasonably be considered to have IC/BPS.21
In contrast to the NIDDK criteria, the ICS criteria5 defined BPS as “the complaint of suprapubic pain related to bladder filling, accompanied by other symptoms such as increased daytime and nighttime urinary frequency in the absence of proven urinary tract infection or other obvious pathology.”5 Additionally, the ICS document restricts the diagnosis of IC to patients with painful bladder syndrome in addition to “typical cystoscopic and histologic features.”6
According to the ESSIC proposal on diagnostic criteria, classification, and nomenclature,6 diagnosing IC/BPS requires symptoms of chronic pain related to the urinary bladder accompanied by at least one other urinary symptom, such as daytime and nighttime frequency and exclusion of confusable diseases and cystoscopy with hydrodistention and biopsy, if indicated. The new statement does not include the required absence of UTI or other pathology identified in the previous (ICS) criteria; these, too, overlooked a portion of the population who would be considered to have IC/BPS. Therefore, the ESSIC classification provides the most comprehensive criteria for diagnosing IC/BPS and has been determined as best for diagnostic purposes in the early disease stages.12
On the next page: Applying the criteria and referral >>
Applying the Criteria
IC/BPS remains a diagnosis of exclusion.12 The most common disorders seen in the differential diagnosis for IC/BPS (ie, “confusable diseases”6) include bacterial cystitis, vaginitis, pelvic pain, vulvodynia, urinary tract infections, yeast infections, sexually transmitted infections, endometriosis, overactive bladder, and genitourinary malignancies.5,12
Biopsy or cystoscopy with short-duration, low-pressure hydrodistention can be performed on patients who present with persistent pelvic pain and urinary symptoms.2,12 Common cystoscopic findings in patients with IC/BPS include Hunner’s lesions, glomerulations, and inflammatory infiltrates on biopsy.12,21 Hunner’s lesions are described as “patches of red mucosa exhibiting small vessels radiating to a central pale scar.”21 These lesions may also be referred to as Hunner’s ulcers.12 Not always visible on cystoscopy, Hunner’s lesions may be seen only after hydrodistention of the bladder under anesthesia.
Cystoscopic findings can be misleading for providers, as not all stages of IC/BPS manifest in the same manner. No single laboratory finding will identify IC/BPS. The only way to diagnose this disease is to rule out all other diseases with similar presentations.18
When to Refer
Specific findings that may indicate the need for referral include severe pain, hematuria, chronic UTI, and pyuria. Generally, however, the decision to refer the patient with IC/BPS to a urologist or urogynecologist depends on the primary care provider’s comfort level. Some providers choose to refer as soon as identifying symptoms of IC/BPS have been confirmed, whereas others may wish to proceed with further evaluation and/or treatment before referring.2,22
Even if the provider decides to refer immediately after identifying symptoms, it is important to initiate some patient education: for example, explaining that the patient will likely require further tests, including cystoscopy and possibly urodynamic evaluation.2,23 Smokers and other patients at high risk for bladder cancer should be referred for cystoscopy.2
If the primary care provider chooses to proceed with evaluation and treatment before referring the patient, follow-up is typically recommended at one-month intervals for the first three months, then every three months thereafter.18 This allows the clinician to monitor a patient’s progress and address concerns that may develop. Symptoms may be slow to respond to treatment, so it is essential to encourage the patient to adhere to the prescribed regimen. If three to six months of first-line treatment yield no response, further consultation and evaluation are warranted. Overall, a multidisciplinary approach that includes the participation of a urologist, a gynecologist, or other appropriate specialist will help ensure optimal treatment and care.18
A good tool that is often used to gauge the patient’s progress is the O’Leary/Sant Voiding and Pain Indices23-25 (see Figure 224). Reviewing patient responses to this questionnaire, with its precise numerical system, at each follow-up appointment can be especially helpful.
On the next page: Treatment >>
TREATMENT
Management of IC/BPS can be challenging, because it is such a multifaceted disorder. Patient education beginning shortly after diagnosis is crucial, as treatment regimens may involve complex multimodal therapy over long periods of time, oftentimes with a very gradual response (see “For Your Patient”).
Lifestyle changes for patients with IC/BPS are considered an important component of treatment. Dietary changes—specifically, reducing intake of foods with high acidic content (citrus fruits, tomatoes), alcoholic beverages, spices, and potassium—have been found helpful.5 Reducing stress and anxiety, whenever possible, has also been noted to alleviate symptoms.24
Another nonpharmacologic option is physical therapy, including biofeedback and bladder retraining.12,13 Biofeedback is particularly useful in patients who experience pelvic pain attributed to spasms of the pelvic floor.12 Bladder retraining can be used to reduce urinary frequency through techniques that include scheduled voiding. Physical therapy strategies should be revisited regularly to maintain their therapeutic benefits.5,12
Oral Medications
The mainstay of pharmacologic treatment, and the one most thoroughly studied, is oral pentosan polysulfate (PPS), which belongs to the class of heparins or heparinoids.2,26 PPS is thought to attach to the mucosa of the bladder, reestablishing its glycosaminoglycan layer and restoring normal function of this permeable barrier.14 Overall, this drug is well tolerated and relieves the symptoms of pain, urgency, and frequency. Patients may start to experience improvement in symptoms after four weeks of treatment; however, it can take six months or longer to achieve the full benefit of this therapy.13,25
Other pharmacologic agents used in the treatment of IC/BPS include antihistamines, tricyclic antidepressants, and some antiepileptic medications. Some patients with IC/BPS experience symptoms attributable to bladder mastocytosis and mast cell activation, explaining the efficacy of antihistamines for these particular patients.27 Among the antihistamines, hydroxyzine, an H1-receptor antagonist, is a common pharmacologic option. Similarly, cetirizine can be used in patients for whom the sedating effects of hydroxyzine may prove hazardous.28
Antidepressants, especially tricyclic antidepressants (TCAs, eg, amitriptyline), can also provide some relief for patients, including alleviation of pain, possible antihistamine effects, and mild anticholinergic action, leading to decreased urinary urgency and frequency.2,26,29 Of note, the TCA imipramine should be avoided in patients with IC/BPS, as it has a sympathomimetic effect that can worsen symptoms of dysfunctional voiding in this patient population.14
Gabapentin, an antiepileptic, is used for improvement of severe, persistent pain. Alternatives to gabapentin include, but are not limited to, phenytoin, carbamazepine, and valproic acid.14 The effectiveness of these medications in the treatment of IC/BPS lend credence to the theory that, in addition to bladder mucosa dysfunction, symptoms are also mediated through an inflammatory neurogenic pathway.
Patients should be encouraged to continue use of PPS or other prescribed pharmacologic treatments even if there is no immediate relief of symptoms.25 According to a treatment algorithm from the American Urological Association,2 however, ineffective treatments should be stopped and diagnosis should be reconsidered if there is no improvement within a “clinically meaningful time frame.”
Additional Pharmacologic Options
Intravesical therapy is another mode of pharmacologic treatment.2,29 This treatment is usually reserved for IC/BPS flares and management of cases lacking the desired response to oral medications. Dimethyl sulfoxide (DMSO) is a commonly used intravesical agent. DMSO acts to provide pain relief and reduce inflammation, in addition to effecting histamine release from mast cells.27 Intravesical heparinoids essentially employ the same mechanism of action as oral PPS to maintain and enhance the bladder’s mucosal lining. This treatment is also commonly used in patients who need to discontinue use of oral PPS due to side effects.27,30
Treatment options for refractory IC/BPS include immunosuppression (and surgical therapy, below). Prednisone and cyclosporine have been shown to be effective immunosuppressive agents.23 Side effects make the use of these medications less desirable; also, symptoms have been shown to return in many patients after treatment is stopped.23
The FDA has recently approved the use of onabotulinum toxin A (Botox) injections into the bladder for treatment of urinary urgency and frequency that are not responsive to standard medical therapy. Since patients with IC often experience such symptoms with no relief from standard therapy, intratrigonal and periurethral injections of Botox are being administered for treatment of IC in some patients with moderate success.31-33 Although intradetrusor Botox use is recommended as a fifth-line treatment in the AUA guidelines,2 it is important to note that this agent is not FDA-approved specifically for IC, but rather for any refractory condition presenting with urinary frequency and urgency.
Surgical Therapy
Surgical intervention (a sixth-line treatment option, according to the AUA guidelines2) is rarely indicated except in cases of severe IC/BPS that have been refractory to all other treatment options and in which spontaneous remission of symptoms seems unlikely. Supravesical urinary diversion, usually through the creation of an ileal conduit, is the procedure of choice and is often performed in conjunction with a cystectomy. Unfortunately in some cases, pelvic pain has been noted to continue postcystectomy, a finding that also supports a neurogenic etiology for IC/BPS.14
On the Horizon
Although IC/PBS is difficult to treat, new data suggest that use of extended diagnostics, including molecular markers to detect the disease early and guide effective treatment, may greatly improve current therapeutic options.34
Prescribing selective anticholinergic and antihistamine pharmacotherapy based on the patient’s specific muscarinic and histamine receptor profile, respectively, may provide greater symptom relief.34 Maintaining the appropriate, individualized therapy could represent a significant advance in treatment for IC/BPS. However, further research on the topic is needed.
On the next page: Conclusion >>
CONCLUSION
IC/BPS is a complex multifactorial syndrome that may manifest with disabling pain. Further research on this disease is warranted to help facilitate an earlier, more consistent diagnosis and produce more effective treatment options. Early diagnosis of IC/BPS is essential for successful therapy. Once the diagnosis is made, a cautious regimen of different treatments, following the American Urological Association’s clinical practice guidelines for interstitial cystitis, should be implemented. Patients should also be encouraged to consider specific dietary changes and other lifestyle adjustments under a clinician’s supervision.
The authors wish to thank Carol Hildebrandt for her help in preparing this manuscript and Robert J. Evans, MD, Associate Professor of Urology, Wake Forest Baptist Medical Center, for his editorial expertise.
1. Dasgupta J, Tincello DG. Interstitial cystitis/bladder pain syndrome: an update. Maturitas. 2009;64:212-217.
2. Hanno PM, Burks DA, Clemens JQ, et al. Diagnosis and treatment of interstitial cystitis/bladder pain syndrome: American Urological Association (AUA) Guideline (2011). www.auanet.org/education/guidelines/ic-bladder-pain-syndrome.cfm. Accessed June 5, 2013.
3. Evans RJ; University of Tennessee Advanced Studies in Pharmacy. Pathophysiology and clinical presentation of interstitial cystitis (2005). www.utasip.com/files/articlefiles/pdf/XASIP_Issue_Mar_p8_14.pdf. Accessed June 5, 2013.
4. Sant GR. Etiology, pathogenesis, and diagnosis of interstitial cystitis. Rev Urol. 2002;(4 suppl 1):S9-S15.
5. Abrams P, Cardozo L, Fall M, et al. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Neurourol Urodyn. 2002;21:167-178.
6. van de Merwe JP, Nordling J, Bouchelouche P, et al. Diagnostic criteria, classification, and nomenclature for painful bladder syndrome/interstitial cystitis: an ESSIC proposal. Eur Urol. 2008;53:60-67.
7. Bogart LM, Berry SH, Clemens JQ. Symptoms of interstitial cystitis, painful bladder syndrome and similar diseases in women: a systematic review. J Urol. 2007;177:450-456.
8. Kusek JW, Nyberg LM. The epidemiology of interstitial cystitis: is it time to expand our definition? Urology. 2001;57(6 suppl 1):95-99.
9. Clemens JQ, Meenan RT, Rosetti MC, et al. Prevalence and incidence of interstitial cystitis in a managed care population. J Urol. 2005;173:98-102.
10. Berry SH, Elliott MN, Suttorp M, et al. Prevalence of symptoms of bladder pain syndrome/interstitial cystitis among adult females in the United States. J Urol. 2011;186:540-544.
11. Rosamilia A, Dwyer PL. Pathophysiology of interstitial cystitis. Curr Opin Obstet Gynecol. 2000;12:405-410.
12. Grover S, Srivastava A, Lee R, et al. Role of inflammation in bladder function and interstitial cystitis. Ther Adv Urol. 2011;3:19-33.
13. Rosenberg MT, Page S, Hazzard MA. Prevalence of interstitial cystitis in a primary care setting. Urology. 2007;69(4 suppl):S48-S52.
14. Evans RJ. Treatment approaches for interstitial cystitis: multimodality therapy. Rev Urol. 2002;(4 suppl 1):S16-S20.
15. Wesselmann U. Interstitial cystitis: a chronic visceral pain syndrome. Urology. 2001;57(6 suppl 1):102.
16. Hosseini A, Ehrén I, Wiklund NP. Nitric oxide as an objective marker for evaluation of treatment response in patients with classic interstitial cystitis. J Urol. 2004;172(6 pt 1):2261-2265.
17. Ho MH, Bhatia NN, Khorram O. Physiologic role of nitric oxide and nitric oxide synthase in female lower urinary tract. Curr Opin Obstet Gynecol. 2004;16:423-429.
18. Rosenberg MT, Newman DK, Page SA. Interstitial cystitis/painful bladder syndrome: symptom recognition is key to early identification, treatment. Cleve Clin J Med. 2007;74:854-862.
19. Driscoll A, Teichman JM. How do patients with interstitial cystitis present? J Urol. 2001;166:2118-2120.
20. Parsons CL. Interstitial cystitis: epidemiology and clinical presentation. Clin Obstet Gynecol. 2002;45:242-249.
21. Hanno PM, Landis JR, Matthews-Cook Y, et al. The diagnosis of interstitial cystitis revisited: lessons learned from the National Institutes of Health Interstitial Cystitis Database study. J Urol. 1999;161:553-557.
22. Whitmore KE, Theoharides TC. When to suspect interstitial cystitis. J Fam Pract. 2011;60:340-348.
23. Butrick CW, Howard FM, Sand PK. Diagnosis and treatment of interstitial cystitis/painful bladder syndrome: a review. J Womens Health (Larchmt). 2010;19:1185-1193.
24. O’Leary MP, Sant GR, Fowler FJ Jr, et al. The interstitial cystitis symptom index and problem index. Urology. 1997;49(5A suppl):58-63.
25. Nickel JC. Forensic dissection of a clinical trial: lessons learned in understanding and managing interstitial cystitis. Rev Urol. 2010;12:e78-e85.
26. Anger JT, Zabihi N, Clemens JQ, et al. Treatment choice, duration, and cost in patients with interstitial cystitis and painful bladder syndrome. Int Urogynecol J. 2011;22:395-400.
27. Nickel JC. Interstitial cystitis: characterization and management of an enigmatic urologic syndrome. Rev Urol. 2002;4:112-121.
28. Dell JR. Interstitial cystitis/painful bladder syndrome: appropriate diagnosis and management. J Womens Health (Larchmt). 2007;16:1181-1187.
29. National Kidney and Urologic Diseases Information Clearinghouse. Interstitial cystitis/painful bladder syndrome (2011). NIH Publication No. 11–3220. http://kidney.niddk.nih.gov/kudiseases/pubs/interstitialcystitis/IC_PBS_T_508.pdf. Accessed June 5, 2013.
30. Davis EL, El Khoudary SR, Talbott EO, et al. Safety and efficacy of the use of intravesical and oral pentosan polysulfate sodium for interstitial cystitis: a randomized double-blind clinical trial. J Urol. 2008;179:177-185.
31. Pinto R, Lopes T, Silva J, et al. Persistent therapeutic effect of repeated injections of onabotulinum toxin a in refractory bladder pain syndrome/interstitial cystitis. J Urol. 2013;189:548-553.
32. Pinto R, Lopoes T, Frias B, et al. Trigonal injection of botulinum toxin A in patients with refractory bladder pain syndrome/interstitial cystitis. Eur Urol. 2010;58:360-365.
33. Gottsch HP, Miller JL, Yang CC, Berger RE. A pilot study of botulinum toxin for interstitial cystitis/painful bladder syndrome. Neurourol Urodyn. 2011; 30:93-96.
34. Neuhaus J, Schwalenberg T, Horn LC, et al. New aspects in the differential diagnosis and therapy of bladder pain syndrome/interstitial cystitis. Adv Urol. 2011;2011:639479.
CE/CME No: CR-1307
PROGRAM OVERVIEW
Earn credit by reading this article and successfully completing the posttest. Successful completion is defined as a cumulative score of at least 70% correct.
EDUCATIONAL OBJECTIVES
•
Describe the pathophysiology
of interstitial cystitis/bladder
pain syndrome (IC/BPS), as
it is currently understood.
•
Discuss urogenital signs and
symptoms that should prompt suspicion for IC/BPS in a primary care patient.
•
Explain the clinical diagnosis of
IC/BPS and key considerations for referral.
•
Review medical management, nonoperative therapy, and surgical treatment of IC/BPS.
FACULTY
LaToya M. Haynes practices at the Carolinas Pain Institute and the Center for Clinical Research in Winston-Salem, North Carolina, and is a preceptor for PA students. Kelly Bilello is a PA at Genitourinary Surgical Consultants in Denver. Jade Breeback practices at Cone Health Primary Care in Kernersville, North Carolina. Jessica Cain is a PA in emergency medicine at the University of Cincinnati Medical Center. Jennifer Wenninger is a cardiothoracic and vascular surgery PA at Bellin Health Care Systems in Green Bay, Wisconsin. M. Jane McDaniel is an Instructor in the Department of Physician Assistant Studies at Wake Forest School of Medicine in Winston-Salem.
The authors have no significant financial relationships to disclose.
ACCREDITATION STATEMENT
Article begins on next page >>
Interstitial cystitis/bladder pain syndrome (IC/BPS) is a common, painful disease of the urinary bladder. Difficult to diagnose and frequently misdiagnosed as another common urologic disorder, IC/BPS challenges health care providers to identify it early and implement current treatment algorithms that may simplify management and improve quality of life for affected patients.
Interstitial cystitis (IC), or bladder pain syndrome (BPS), is a clinical condition characterized by bladder pain, urinary frequency and urgency, and increased nighttime urination (nocturia).1 More specifically, IC/BPS is defined as an unpleasant sensation in the bladder, abdomen, or pelvis (ie, pain, pressure, burning, and/or other discomfort) perceived to be originating in the urinary bladder. The condition is associated with lower urinary tract symptoms of more than six weeks’ duration, with no infection or other identifiable cause present.2
IC/BPS lacks a single known etiology; rather, it most likely results from multiple contributing factors that cascade into a painful and potentially debilitating syndrome. The condition was first described more than a century ago,3,4 but its complex nature and conflicting theories about its pathogenesis present both diagnostic and therapeutic challenges for health care professionals. Frequent misdiagnosis of IC/BPS as another common urologic disorder can make timely, appropriate treatment elusive.
Without a clearly described pathophysiology, IC/BPS has always been difficult to define using standardized diagnostic criteria and precise terminology. The definition of the condition was revised in 2002 and again in 2008, when the nomenclature bladder pain syndrome was introduced.1,5,6
Less than 10 years ago, US researchers described IC as a subgroup of BPS,7 while in Europe, BPS is used as the broader term, with IC still considered a well-defined subgroup that usually involves ulceration.6 The future may find IC, BPS, and painful bladder syndrome (PBS) used as interchangeable terms—or as unique diagnoses. A better understanding of the pathophysiology of IC/BPS/PBS would contribute not only to resolving issues of nomenclature, but also to establishing an accurate diagnosis earlier in the disease process and providing more efficient, effective treatment.
THE PROBLEM OF EPIDEMIOLOGY
Inconsistencies in the terminology, definitions, and diagnostic criteria of IC/BPS have made epidemiology difficult to establish.1 It has been suggested that IC/BPS is underdiagnosed in the United States and that its prevalence is much greater than generally reported.8
According to one study of IC in a managed care population, its prevalence in 2005 was 197 per 100,000 women and 41 per 100,000 men, with the female-to-male ratio estimated at 5:1.9 In 2011, researchers for the RAND Corporation published what they called the first population-based “symptom prevalence estimate” among US women older than 18, based on more than 100,000 screening interviews conducted by phone. According to their findings, between 3.3 and 7.9 million US women meet the stated criteria for IC/BPS (ie, between 3,113 and 7,453 women per 100,000).10 These conflicting data exemplify the range of epidemiologic conclusions that exist regarding this condition.
On the next page: Proposed pathophysiology >>
THE PROPOSED PATHOPHYSIOLOGY
IC/BPS is thought to begin with an initial insult to the bladder that leads to dysfunction of the epithelial layer. This insult may be the result of a neurogenic inflammation, autoimmunity, subclinical or chronic infection, or bladder urothelial defects.1 Dysfunction in the epithelial layer includes altered bladder epithelial expression of human leukocyte antigen I and II; decreased expression of uroplakin (an antitoxic protein in the bladder), and a defective glycosaminoglycan mucus layer.4 This damage to the epithelial layer alters the permeability of the bladder, allowing potassium ions to enter the urothelium and depolarize motor and sensory nerves. This potassium leak then activates the mast cells, causing mastocytosis and the release of histamine.11 These processes disrupt the homeostasis of the urinary tract and allow the development of inflammation—a main cause of the pelvic pain associated with IC/BPS4,12,13 (see Figure 114).
Other factors that exacerbate the primary inflammation in the bladder are C-fibers and nerve growth factor (NGF). C-fibers are afferent fibers found in the peripheral nerves of the somatic sensory system that convey input signals from the periphery to the central nervous system.3 In patients with IC/BPS, initial inflammation activates C-fibers, which produce substance P, nociceptor, and other inflammatory mediators. These mediators exacerbate existing inflammation and further facilitate mast cell activation.3
NGF is a protein that is critical for the maintenance of sympathetic and sensory neurons; it is important not only in the urinary tract but in all organ systems. Increased levels of NGF, a prevalent finding in patients with IC/BPS, is an indicator of inflammation in the body. The precise mechanism that causes elevated NGF in patients with IC/BPS is not well understood, but its presence supports the theory that inflammation is a cause of pelvic pain in IC/BPS.12
The urinary urgency and frequency experienced by patients with IC/BPS is in part due to the role nitric oxide (NO) plays in bladder activity. Patients with IC have decreased levels of urinary NO (a reduction thought to be the result of a decrease in L-arginine) and urinary NO synthase.12,15,16 Ordinarily, NO synthase converts L-arginine to NO, which helps to control relaxation of the bladder smooth muscle, allowing more urine to be stored. In patients with IC/BPS, NO insufficiency leads to bladder overactivity.15
On the next page: Patient history and presentation >>
PATIENT HISTORY AND PRESENTATION
A detailed patient history is imperative in establishing the diagnosis of IC/BPS. Symptoms that should prompt the clinician to consider IC/BPS include:
• Pelvic or bladder pain relieved with voiding
• Dyspareunia
• Increased frequency of urination with no infection present
• Urinary urgency with pain, and
• Increased nocturia.17,18
Early IC presents variably, and pain, though a common symptom, is not always present.19 Chronic pain is defined by duration of at least six months, with the discomfort perceived as originating in the bladder.8 In addition to patients who experience pain, those who void several times during the night should also be considered for further evaluation.19
Many patients describe their symptoms in terms of flares and periods of remission. Some patients associate flares with stress, seasonal allergies, sexual activity, consumption of certain foods, and the premenstrual week.17,20 Patients with IC/BPS are commonly misdiagnosed with recurrent urinary tract infections; hence the need to standardize the criteria for diagnosis of IC/BPS.17
DIAGNOSIS
There are currently three available sets of diagnostic criteria for patients with IC/BPS. These are the National Institute for Diabetes and Diseases of the Kidney (NIDDK) definition (1990),21 the International Continence Society (ICS) definition of painful bladder disorders (2002),5 and the European Society for the Study of IC/BPS (ESSIC) definition (2008).6 In particular, the ESSIC criteria were formulated to help identify IC/BPS earlier in the disease course.
The 1990 NIDDK protocol, developed for research purposes,12 featured inclusion and exclusion criteria. Exclusion criteria included age younger than 18 years and presence of benign bladder tumors, radiation cystitis, tuberculosis cystitis, bacterial cystitis, vaginitis, symptomatic urethral diverticulum, uterine/cervical/vaginal cancers, and/or active herpes; urinary frequency of less than five episodes in 12 hours; and less than two episodes of nocturia per night.19
NIDDK inclusion criteria required two or more of the following: Hunner’s ulcer, pain on bladder filling, general pelvic pain, glomerulations on endoscopy, and decreased bladder compliance on cystometrogram.19
This protocol proved to be excessively restrictive for clinical use and was widely replaced by the ICS criteria in 2002. The ICS criteria5 allowed more varied patient presentations; the exclusions featured in the NIDDK guideline, it has been estimated, could have eliminated at least one-third of patients who would reasonably be considered to have IC/BPS.21
In contrast to the NIDDK criteria, the ICS criteria5 defined BPS as “the complaint of suprapubic pain related to bladder filling, accompanied by other symptoms such as increased daytime and nighttime urinary frequency in the absence of proven urinary tract infection or other obvious pathology.”5 Additionally, the ICS document restricts the diagnosis of IC to patients with painful bladder syndrome in addition to “typical cystoscopic and histologic features.”6
According to the ESSIC proposal on diagnostic criteria, classification, and nomenclature,6 diagnosing IC/BPS requires symptoms of chronic pain related to the urinary bladder accompanied by at least one other urinary symptom, such as daytime and nighttime frequency and exclusion of confusable diseases and cystoscopy with hydrodistention and biopsy, if indicated. The new statement does not include the required absence of UTI or other pathology identified in the previous (ICS) criteria; these, too, overlooked a portion of the population who would be considered to have IC/BPS. Therefore, the ESSIC classification provides the most comprehensive criteria for diagnosing IC/BPS and has been determined as best for diagnostic purposes in the early disease stages.12
On the next page: Applying the criteria and referral >>
Applying the Criteria
IC/BPS remains a diagnosis of exclusion.12 The most common disorders seen in the differential diagnosis for IC/BPS (ie, “confusable diseases”6) include bacterial cystitis, vaginitis, pelvic pain, vulvodynia, urinary tract infections, yeast infections, sexually transmitted infections, endometriosis, overactive bladder, and genitourinary malignancies.5,12
Biopsy or cystoscopy with short-duration, low-pressure hydrodistention can be performed on patients who present with persistent pelvic pain and urinary symptoms.2,12 Common cystoscopic findings in patients with IC/BPS include Hunner’s lesions, glomerulations, and inflammatory infiltrates on biopsy.12,21 Hunner’s lesions are described as “patches of red mucosa exhibiting small vessels radiating to a central pale scar.”21 These lesions may also be referred to as Hunner’s ulcers.12 Not always visible on cystoscopy, Hunner’s lesions may be seen only after hydrodistention of the bladder under anesthesia.
Cystoscopic findings can be misleading for providers, as not all stages of IC/BPS manifest in the same manner. No single laboratory finding will identify IC/BPS. The only way to diagnose this disease is to rule out all other diseases with similar presentations.18
When to Refer
Specific findings that may indicate the need for referral include severe pain, hematuria, chronic UTI, and pyuria. Generally, however, the decision to refer the patient with IC/BPS to a urologist or urogynecologist depends on the primary care provider’s comfort level. Some providers choose to refer as soon as identifying symptoms of IC/BPS have been confirmed, whereas others may wish to proceed with further evaluation and/or treatment before referring.2,22
Even if the provider decides to refer immediately after identifying symptoms, it is important to initiate some patient education: for example, explaining that the patient will likely require further tests, including cystoscopy and possibly urodynamic evaluation.2,23 Smokers and other patients at high risk for bladder cancer should be referred for cystoscopy.2
If the primary care provider chooses to proceed with evaluation and treatment before referring the patient, follow-up is typically recommended at one-month intervals for the first three months, then every three months thereafter.18 This allows the clinician to monitor a patient’s progress and address concerns that may develop. Symptoms may be slow to respond to treatment, so it is essential to encourage the patient to adhere to the prescribed regimen. If three to six months of first-line treatment yield no response, further consultation and evaluation are warranted. Overall, a multidisciplinary approach that includes the participation of a urologist, a gynecologist, or other appropriate specialist will help ensure optimal treatment and care.18
A good tool that is often used to gauge the patient’s progress is the O’Leary/Sant Voiding and Pain Indices23-25 (see Figure 224). Reviewing patient responses to this questionnaire, with its precise numerical system, at each follow-up appointment can be especially helpful.
On the next page: Treatment >>
TREATMENT
Management of IC/BPS can be challenging, because it is such a multifaceted disorder. Patient education beginning shortly after diagnosis is crucial, as treatment regimens may involve complex multimodal therapy over long periods of time, oftentimes with a very gradual response (see “For Your Patient”).
Lifestyle changes for patients with IC/BPS are considered an important component of treatment. Dietary changes—specifically, reducing intake of foods with high acidic content (citrus fruits, tomatoes), alcoholic beverages, spices, and potassium—have been found helpful.5 Reducing stress and anxiety, whenever possible, has also been noted to alleviate symptoms.24
Another nonpharmacologic option is physical therapy, including biofeedback and bladder retraining.12,13 Biofeedback is particularly useful in patients who experience pelvic pain attributed to spasms of the pelvic floor.12 Bladder retraining can be used to reduce urinary frequency through techniques that include scheduled voiding. Physical therapy strategies should be revisited regularly to maintain their therapeutic benefits.5,12
Oral Medications
The mainstay of pharmacologic treatment, and the one most thoroughly studied, is oral pentosan polysulfate (PPS), which belongs to the class of heparins or heparinoids.2,26 PPS is thought to attach to the mucosa of the bladder, reestablishing its glycosaminoglycan layer and restoring normal function of this permeable barrier.14 Overall, this drug is well tolerated and relieves the symptoms of pain, urgency, and frequency. Patients may start to experience improvement in symptoms after four weeks of treatment; however, it can take six months or longer to achieve the full benefit of this therapy.13,25
Other pharmacologic agents used in the treatment of IC/BPS include antihistamines, tricyclic antidepressants, and some antiepileptic medications. Some patients with IC/BPS experience symptoms attributable to bladder mastocytosis and mast cell activation, explaining the efficacy of antihistamines for these particular patients.27 Among the antihistamines, hydroxyzine, an H1-receptor antagonist, is a common pharmacologic option. Similarly, cetirizine can be used in patients for whom the sedating effects of hydroxyzine may prove hazardous.28
Antidepressants, especially tricyclic antidepressants (TCAs, eg, amitriptyline), can also provide some relief for patients, including alleviation of pain, possible antihistamine effects, and mild anticholinergic action, leading to decreased urinary urgency and frequency.2,26,29 Of note, the TCA imipramine should be avoided in patients with IC/BPS, as it has a sympathomimetic effect that can worsen symptoms of dysfunctional voiding in this patient population.14
Gabapentin, an antiepileptic, is used for improvement of severe, persistent pain. Alternatives to gabapentin include, but are not limited to, phenytoin, carbamazepine, and valproic acid.14 The effectiveness of these medications in the treatment of IC/BPS lend credence to the theory that, in addition to bladder mucosa dysfunction, symptoms are also mediated through an inflammatory neurogenic pathway.
Patients should be encouraged to continue use of PPS or other prescribed pharmacologic treatments even if there is no immediate relief of symptoms.25 According to a treatment algorithm from the American Urological Association,2 however, ineffective treatments should be stopped and diagnosis should be reconsidered if there is no improvement within a “clinically meaningful time frame.”
Additional Pharmacologic Options
Intravesical therapy is another mode of pharmacologic treatment.2,29 This treatment is usually reserved for IC/BPS flares and management of cases lacking the desired response to oral medications. Dimethyl sulfoxide (DMSO) is a commonly used intravesical agent. DMSO acts to provide pain relief and reduce inflammation, in addition to effecting histamine release from mast cells.27 Intravesical heparinoids essentially employ the same mechanism of action as oral PPS to maintain and enhance the bladder’s mucosal lining. This treatment is also commonly used in patients who need to discontinue use of oral PPS due to side effects.27,30
Treatment options for refractory IC/BPS include immunosuppression (and surgical therapy, below). Prednisone and cyclosporine have been shown to be effective immunosuppressive agents.23 Side effects make the use of these medications less desirable; also, symptoms have been shown to return in many patients after treatment is stopped.23
The FDA has recently approved the use of onabotulinum toxin A (Botox) injections into the bladder for treatment of urinary urgency and frequency that are not responsive to standard medical therapy. Since patients with IC often experience such symptoms with no relief from standard therapy, intratrigonal and periurethral injections of Botox are being administered for treatment of IC in some patients with moderate success.31-33 Although intradetrusor Botox use is recommended as a fifth-line treatment in the AUA guidelines,2 it is important to note that this agent is not FDA-approved specifically for IC, but rather for any refractory condition presenting with urinary frequency and urgency.
Surgical Therapy
Surgical intervention (a sixth-line treatment option, according to the AUA guidelines2) is rarely indicated except in cases of severe IC/BPS that have been refractory to all other treatment options and in which spontaneous remission of symptoms seems unlikely. Supravesical urinary diversion, usually through the creation of an ileal conduit, is the procedure of choice and is often performed in conjunction with a cystectomy. Unfortunately in some cases, pelvic pain has been noted to continue postcystectomy, a finding that also supports a neurogenic etiology for IC/BPS.14
On the Horizon
Although IC/PBS is difficult to treat, new data suggest that use of extended diagnostics, including molecular markers to detect the disease early and guide effective treatment, may greatly improve current therapeutic options.34
Prescribing selective anticholinergic and antihistamine pharmacotherapy based on the patient’s specific muscarinic and histamine receptor profile, respectively, may provide greater symptom relief.34 Maintaining the appropriate, individualized therapy could represent a significant advance in treatment for IC/BPS. However, further research on the topic is needed.
On the next page: Conclusion >>
CONCLUSION
IC/BPS is a complex multifactorial syndrome that may manifest with disabling pain. Further research on this disease is warranted to help facilitate an earlier, more consistent diagnosis and produce more effective treatment options. Early diagnosis of IC/BPS is essential for successful therapy. Once the diagnosis is made, a cautious regimen of different treatments, following the American Urological Association’s clinical practice guidelines for interstitial cystitis, should be implemented. Patients should also be encouraged to consider specific dietary changes and other lifestyle adjustments under a clinician’s supervision.
The authors wish to thank Carol Hildebrandt for her help in preparing this manuscript and Robert J. Evans, MD, Associate Professor of Urology, Wake Forest Baptist Medical Center, for his editorial expertise.
CE/CME No: CR-1307
PROGRAM OVERVIEW
Earn credit by reading this article and successfully completing the posttest. Successful completion is defined as a cumulative score of at least 70% correct.
EDUCATIONAL OBJECTIVES
•
Describe the pathophysiology
of interstitial cystitis/bladder
pain syndrome (IC/BPS), as
it is currently understood.
•
Discuss urogenital signs and
symptoms that should prompt suspicion for IC/BPS in a primary care patient.
•
Explain the clinical diagnosis of
IC/BPS and key considerations for referral.
•
Review medical management, nonoperative therapy, and surgical treatment of IC/BPS.
FACULTY
LaToya M. Haynes practices at the Carolinas Pain Institute and the Center for Clinical Research in Winston-Salem, North Carolina, and is a preceptor for PA students. Kelly Bilello is a PA at Genitourinary Surgical Consultants in Denver. Jade Breeback practices at Cone Health Primary Care in Kernersville, North Carolina. Jessica Cain is a PA in emergency medicine at the University of Cincinnati Medical Center. Jennifer Wenninger is a cardiothoracic and vascular surgery PA at Bellin Health Care Systems in Green Bay, Wisconsin. M. Jane McDaniel is an Instructor in the Department of Physician Assistant Studies at Wake Forest School of Medicine in Winston-Salem.
The authors have no significant financial relationships to disclose.
ACCREDITATION STATEMENT
Article begins on next page >>
Interstitial cystitis/bladder pain syndrome (IC/BPS) is a common, painful disease of the urinary bladder. Difficult to diagnose and frequently misdiagnosed as another common urologic disorder, IC/BPS challenges health care providers to identify it early and implement current treatment algorithms that may simplify management and improve quality of life for affected patients.
Interstitial cystitis (IC), or bladder pain syndrome (BPS), is a clinical condition characterized by bladder pain, urinary frequency and urgency, and increased nighttime urination (nocturia).1 More specifically, IC/BPS is defined as an unpleasant sensation in the bladder, abdomen, or pelvis (ie, pain, pressure, burning, and/or other discomfort) perceived to be originating in the urinary bladder. The condition is associated with lower urinary tract symptoms of more than six weeks’ duration, with no infection or other identifiable cause present.2
IC/BPS lacks a single known etiology; rather, it most likely results from multiple contributing factors that cascade into a painful and potentially debilitating syndrome. The condition was first described more than a century ago,3,4 but its complex nature and conflicting theories about its pathogenesis present both diagnostic and therapeutic challenges for health care professionals. Frequent misdiagnosis of IC/BPS as another common urologic disorder can make timely, appropriate treatment elusive.
Without a clearly described pathophysiology, IC/BPS has always been difficult to define using standardized diagnostic criteria and precise terminology. The definition of the condition was revised in 2002 and again in 2008, when the nomenclature bladder pain syndrome was introduced.1,5,6
Less than 10 years ago, US researchers described IC as a subgroup of BPS,7 while in Europe, BPS is used as the broader term, with IC still considered a well-defined subgroup that usually involves ulceration.6 The future may find IC, BPS, and painful bladder syndrome (PBS) used as interchangeable terms—or as unique diagnoses. A better understanding of the pathophysiology of IC/BPS/PBS would contribute not only to resolving issues of nomenclature, but also to establishing an accurate diagnosis earlier in the disease process and providing more efficient, effective treatment.
THE PROBLEM OF EPIDEMIOLOGY
Inconsistencies in the terminology, definitions, and diagnostic criteria of IC/BPS have made epidemiology difficult to establish.1 It has been suggested that IC/BPS is underdiagnosed in the United States and that its prevalence is much greater than generally reported.8
According to one study of IC in a managed care population, its prevalence in 2005 was 197 per 100,000 women and 41 per 100,000 men, with the female-to-male ratio estimated at 5:1.9 In 2011, researchers for the RAND Corporation published what they called the first population-based “symptom prevalence estimate” among US women older than 18, based on more than 100,000 screening interviews conducted by phone. According to their findings, between 3.3 and 7.9 million US women meet the stated criteria for IC/BPS (ie, between 3,113 and 7,453 women per 100,000).10 These conflicting data exemplify the range of epidemiologic conclusions that exist regarding this condition.
On the next page: Proposed pathophysiology >>
THE PROPOSED PATHOPHYSIOLOGY
IC/BPS is thought to begin with an initial insult to the bladder that leads to dysfunction of the epithelial layer. This insult may be the result of a neurogenic inflammation, autoimmunity, subclinical or chronic infection, or bladder urothelial defects.1 Dysfunction in the epithelial layer includes altered bladder epithelial expression of human leukocyte antigen I and II; decreased expression of uroplakin (an antitoxic protein in the bladder), and a defective glycosaminoglycan mucus layer.4 This damage to the epithelial layer alters the permeability of the bladder, allowing potassium ions to enter the urothelium and depolarize motor and sensory nerves. This potassium leak then activates the mast cells, causing mastocytosis and the release of histamine.11 These processes disrupt the homeostasis of the urinary tract and allow the development of inflammation—a main cause of the pelvic pain associated with IC/BPS4,12,13 (see Figure 114).
Other factors that exacerbate the primary inflammation in the bladder are C-fibers and nerve growth factor (NGF). C-fibers are afferent fibers found in the peripheral nerves of the somatic sensory system that convey input signals from the periphery to the central nervous system.3 In patients with IC/BPS, initial inflammation activates C-fibers, which produce substance P, nociceptor, and other inflammatory mediators. These mediators exacerbate existing inflammation and further facilitate mast cell activation.3
NGF is a protein that is critical for the maintenance of sympathetic and sensory neurons; it is important not only in the urinary tract but in all organ systems. Increased levels of NGF, a prevalent finding in patients with IC/BPS, is an indicator of inflammation in the body. The precise mechanism that causes elevated NGF in patients with IC/BPS is not well understood, but its presence supports the theory that inflammation is a cause of pelvic pain in IC/BPS.12
The urinary urgency and frequency experienced by patients with IC/BPS is in part due to the role nitric oxide (NO) plays in bladder activity. Patients with IC have decreased levels of urinary NO (a reduction thought to be the result of a decrease in L-arginine) and urinary NO synthase.12,15,16 Ordinarily, NO synthase converts L-arginine to NO, which helps to control relaxation of the bladder smooth muscle, allowing more urine to be stored. In patients with IC/BPS, NO insufficiency leads to bladder overactivity.15
On the next page: Patient history and presentation >>
PATIENT HISTORY AND PRESENTATION
A detailed patient history is imperative in establishing the diagnosis of IC/BPS. Symptoms that should prompt the clinician to consider IC/BPS include:
• Pelvic or bladder pain relieved with voiding
• Dyspareunia
• Increased frequency of urination with no infection present
• Urinary urgency with pain, and
• Increased nocturia.17,18
Early IC presents variably, and pain, though a common symptom, is not always present.19 Chronic pain is defined by duration of at least six months, with the discomfort perceived as originating in the bladder.8 In addition to patients who experience pain, those who void several times during the night should also be considered for further evaluation.19
Many patients describe their symptoms in terms of flares and periods of remission. Some patients associate flares with stress, seasonal allergies, sexual activity, consumption of certain foods, and the premenstrual week.17,20 Patients with IC/BPS are commonly misdiagnosed with recurrent urinary tract infections; hence the need to standardize the criteria for diagnosis of IC/BPS.17
DIAGNOSIS
There are currently three available sets of diagnostic criteria for patients with IC/BPS. These are the National Institute for Diabetes and Diseases of the Kidney (NIDDK) definition (1990),21 the International Continence Society (ICS) definition of painful bladder disorders (2002),5 and the European Society for the Study of IC/BPS (ESSIC) definition (2008).6 In particular, the ESSIC criteria were formulated to help identify IC/BPS earlier in the disease course.
The 1990 NIDDK protocol, developed for research purposes,12 featured inclusion and exclusion criteria. Exclusion criteria included age younger than 18 years and presence of benign bladder tumors, radiation cystitis, tuberculosis cystitis, bacterial cystitis, vaginitis, symptomatic urethral diverticulum, uterine/cervical/vaginal cancers, and/or active herpes; urinary frequency of less than five episodes in 12 hours; and less than two episodes of nocturia per night.19
NIDDK inclusion criteria required two or more of the following: Hunner’s ulcer, pain on bladder filling, general pelvic pain, glomerulations on endoscopy, and decreased bladder compliance on cystometrogram.19
This protocol proved to be excessively restrictive for clinical use and was widely replaced by the ICS criteria in 2002. The ICS criteria5 allowed more varied patient presentations; the exclusions featured in the NIDDK guideline, it has been estimated, could have eliminated at least one-third of patients who would reasonably be considered to have IC/BPS.21
In contrast to the NIDDK criteria, the ICS criteria5 defined BPS as “the complaint of suprapubic pain related to bladder filling, accompanied by other symptoms such as increased daytime and nighttime urinary frequency in the absence of proven urinary tract infection or other obvious pathology.”5 Additionally, the ICS document restricts the diagnosis of IC to patients with painful bladder syndrome in addition to “typical cystoscopic and histologic features.”6
According to the ESSIC proposal on diagnostic criteria, classification, and nomenclature,6 diagnosing IC/BPS requires symptoms of chronic pain related to the urinary bladder accompanied by at least one other urinary symptom, such as daytime and nighttime frequency and exclusion of confusable diseases and cystoscopy with hydrodistention and biopsy, if indicated. The new statement does not include the required absence of UTI or other pathology identified in the previous (ICS) criteria; these, too, overlooked a portion of the population who would be considered to have IC/BPS. Therefore, the ESSIC classification provides the most comprehensive criteria for diagnosing IC/BPS and has been determined as best for diagnostic purposes in the early disease stages.12
On the next page: Applying the criteria and referral >>
Applying the Criteria
IC/BPS remains a diagnosis of exclusion.12 The most common disorders seen in the differential diagnosis for IC/BPS (ie, “confusable diseases”6) include bacterial cystitis, vaginitis, pelvic pain, vulvodynia, urinary tract infections, yeast infections, sexually transmitted infections, endometriosis, overactive bladder, and genitourinary malignancies.5,12
Biopsy or cystoscopy with short-duration, low-pressure hydrodistention can be performed on patients who present with persistent pelvic pain and urinary symptoms.2,12 Common cystoscopic findings in patients with IC/BPS include Hunner’s lesions, glomerulations, and inflammatory infiltrates on biopsy.12,21 Hunner’s lesions are described as “patches of red mucosa exhibiting small vessels radiating to a central pale scar.”21 These lesions may also be referred to as Hunner’s ulcers.12 Not always visible on cystoscopy, Hunner’s lesions may be seen only after hydrodistention of the bladder under anesthesia.
Cystoscopic findings can be misleading for providers, as not all stages of IC/BPS manifest in the same manner. No single laboratory finding will identify IC/BPS. The only way to diagnose this disease is to rule out all other diseases with similar presentations.18
When to Refer
Specific findings that may indicate the need for referral include severe pain, hematuria, chronic UTI, and pyuria. Generally, however, the decision to refer the patient with IC/BPS to a urologist or urogynecologist depends on the primary care provider’s comfort level. Some providers choose to refer as soon as identifying symptoms of IC/BPS have been confirmed, whereas others may wish to proceed with further evaluation and/or treatment before referring.2,22
Even if the provider decides to refer immediately after identifying symptoms, it is important to initiate some patient education: for example, explaining that the patient will likely require further tests, including cystoscopy and possibly urodynamic evaluation.2,23 Smokers and other patients at high risk for bladder cancer should be referred for cystoscopy.2
If the primary care provider chooses to proceed with evaluation and treatment before referring the patient, follow-up is typically recommended at one-month intervals for the first three months, then every three months thereafter.18 This allows the clinician to monitor a patient’s progress and address concerns that may develop. Symptoms may be slow to respond to treatment, so it is essential to encourage the patient to adhere to the prescribed regimen. If three to six months of first-line treatment yield no response, further consultation and evaluation are warranted. Overall, a multidisciplinary approach that includes the participation of a urologist, a gynecologist, or other appropriate specialist will help ensure optimal treatment and care.18
A good tool that is often used to gauge the patient’s progress is the O’Leary/Sant Voiding and Pain Indices23-25 (see Figure 224). Reviewing patient responses to this questionnaire, with its precise numerical system, at each follow-up appointment can be especially helpful.
On the next page: Treatment >>
TREATMENT
Management of IC/BPS can be challenging, because it is such a multifaceted disorder. Patient education beginning shortly after diagnosis is crucial, as treatment regimens may involve complex multimodal therapy over long periods of time, oftentimes with a very gradual response (see “For Your Patient”).
Lifestyle changes for patients with IC/BPS are considered an important component of treatment. Dietary changes—specifically, reducing intake of foods with high acidic content (citrus fruits, tomatoes), alcoholic beverages, spices, and potassium—have been found helpful.5 Reducing stress and anxiety, whenever possible, has also been noted to alleviate symptoms.24
Another nonpharmacologic option is physical therapy, including biofeedback and bladder retraining.12,13 Biofeedback is particularly useful in patients who experience pelvic pain attributed to spasms of the pelvic floor.12 Bladder retraining can be used to reduce urinary frequency through techniques that include scheduled voiding. Physical therapy strategies should be revisited regularly to maintain their therapeutic benefits.5,12
Oral Medications
The mainstay of pharmacologic treatment, and the one most thoroughly studied, is oral pentosan polysulfate (PPS), which belongs to the class of heparins or heparinoids.2,26 PPS is thought to attach to the mucosa of the bladder, reestablishing its glycosaminoglycan layer and restoring normal function of this permeable barrier.14 Overall, this drug is well tolerated and relieves the symptoms of pain, urgency, and frequency. Patients may start to experience improvement in symptoms after four weeks of treatment; however, it can take six months or longer to achieve the full benefit of this therapy.13,25
Other pharmacologic agents used in the treatment of IC/BPS include antihistamines, tricyclic antidepressants, and some antiepileptic medications. Some patients with IC/BPS experience symptoms attributable to bladder mastocytosis and mast cell activation, explaining the efficacy of antihistamines for these particular patients.27 Among the antihistamines, hydroxyzine, an H1-receptor antagonist, is a common pharmacologic option. Similarly, cetirizine can be used in patients for whom the sedating effects of hydroxyzine may prove hazardous.28
Antidepressants, especially tricyclic antidepressants (TCAs, eg, amitriptyline), can also provide some relief for patients, including alleviation of pain, possible antihistamine effects, and mild anticholinergic action, leading to decreased urinary urgency and frequency.2,26,29 Of note, the TCA imipramine should be avoided in patients with IC/BPS, as it has a sympathomimetic effect that can worsen symptoms of dysfunctional voiding in this patient population.14
Gabapentin, an antiepileptic, is used for improvement of severe, persistent pain. Alternatives to gabapentin include, but are not limited to, phenytoin, carbamazepine, and valproic acid.14 The effectiveness of these medications in the treatment of IC/BPS lend credence to the theory that, in addition to bladder mucosa dysfunction, symptoms are also mediated through an inflammatory neurogenic pathway.
Patients should be encouraged to continue use of PPS or other prescribed pharmacologic treatments even if there is no immediate relief of symptoms.25 According to a treatment algorithm from the American Urological Association,2 however, ineffective treatments should be stopped and diagnosis should be reconsidered if there is no improvement within a “clinically meaningful time frame.”
Additional Pharmacologic Options
Intravesical therapy is another mode of pharmacologic treatment.2,29 This treatment is usually reserved for IC/BPS flares and management of cases lacking the desired response to oral medications. Dimethyl sulfoxide (DMSO) is a commonly used intravesical agent. DMSO acts to provide pain relief and reduce inflammation, in addition to effecting histamine release from mast cells.27 Intravesical heparinoids essentially employ the same mechanism of action as oral PPS to maintain and enhance the bladder’s mucosal lining. This treatment is also commonly used in patients who need to discontinue use of oral PPS due to side effects.27,30
Treatment options for refractory IC/BPS include immunosuppression (and surgical therapy, below). Prednisone and cyclosporine have been shown to be effective immunosuppressive agents.23 Side effects make the use of these medications less desirable; also, symptoms have been shown to return in many patients after treatment is stopped.23
The FDA has recently approved the use of onabotulinum toxin A (Botox) injections into the bladder for treatment of urinary urgency and frequency that are not responsive to standard medical therapy. Since patients with IC often experience such symptoms with no relief from standard therapy, intratrigonal and periurethral injections of Botox are being administered for treatment of IC in some patients with moderate success.31-33 Although intradetrusor Botox use is recommended as a fifth-line treatment in the AUA guidelines,2 it is important to note that this agent is not FDA-approved specifically for IC, but rather for any refractory condition presenting with urinary frequency and urgency.
Surgical Therapy
Surgical intervention (a sixth-line treatment option, according to the AUA guidelines2) is rarely indicated except in cases of severe IC/BPS that have been refractory to all other treatment options and in which spontaneous remission of symptoms seems unlikely. Supravesical urinary diversion, usually through the creation of an ileal conduit, is the procedure of choice and is often performed in conjunction with a cystectomy. Unfortunately in some cases, pelvic pain has been noted to continue postcystectomy, a finding that also supports a neurogenic etiology for IC/BPS.14
On the Horizon
Although IC/PBS is difficult to treat, new data suggest that use of extended diagnostics, including molecular markers to detect the disease early and guide effective treatment, may greatly improve current therapeutic options.34
Prescribing selective anticholinergic and antihistamine pharmacotherapy based on the patient’s specific muscarinic and histamine receptor profile, respectively, may provide greater symptom relief.34 Maintaining the appropriate, individualized therapy could represent a significant advance in treatment for IC/BPS. However, further research on the topic is needed.
On the next page: Conclusion >>
CONCLUSION
IC/BPS is a complex multifactorial syndrome that may manifest with disabling pain. Further research on this disease is warranted to help facilitate an earlier, more consistent diagnosis and produce more effective treatment options. Early diagnosis of IC/BPS is essential for successful therapy. Once the diagnosis is made, a cautious regimen of different treatments, following the American Urological Association’s clinical practice guidelines for interstitial cystitis, should be implemented. Patients should also be encouraged to consider specific dietary changes and other lifestyle adjustments under a clinician’s supervision.
The authors wish to thank Carol Hildebrandt for her help in preparing this manuscript and Robert J. Evans, MD, Associate Professor of Urology, Wake Forest Baptist Medical Center, for his editorial expertise.
1. Dasgupta J, Tincello DG. Interstitial cystitis/bladder pain syndrome: an update. Maturitas. 2009;64:212-217.
2. Hanno PM, Burks DA, Clemens JQ, et al. Diagnosis and treatment of interstitial cystitis/bladder pain syndrome: American Urological Association (AUA) Guideline (2011). www.auanet.org/education/guidelines/ic-bladder-pain-syndrome.cfm. Accessed June 5, 2013.
3. Evans RJ; University of Tennessee Advanced Studies in Pharmacy. Pathophysiology and clinical presentation of interstitial cystitis (2005). www.utasip.com/files/articlefiles/pdf/XASIP_Issue_Mar_p8_14.pdf. Accessed June 5, 2013.
4. Sant GR. Etiology, pathogenesis, and diagnosis of interstitial cystitis. Rev Urol. 2002;(4 suppl 1):S9-S15.
5. Abrams P, Cardozo L, Fall M, et al. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Neurourol Urodyn. 2002;21:167-178.
6. van de Merwe JP, Nordling J, Bouchelouche P, et al. Diagnostic criteria, classification, and nomenclature for painful bladder syndrome/interstitial cystitis: an ESSIC proposal. Eur Urol. 2008;53:60-67.
7. Bogart LM, Berry SH, Clemens JQ. Symptoms of interstitial cystitis, painful bladder syndrome and similar diseases in women: a systematic review. J Urol. 2007;177:450-456.
8. Kusek JW, Nyberg LM. The epidemiology of interstitial cystitis: is it time to expand our definition? Urology. 2001;57(6 suppl 1):95-99.
9. Clemens JQ, Meenan RT, Rosetti MC, et al. Prevalence and incidence of interstitial cystitis in a managed care population. J Urol. 2005;173:98-102.
10. Berry SH, Elliott MN, Suttorp M, et al. Prevalence of symptoms of bladder pain syndrome/interstitial cystitis among adult females in the United States. J Urol. 2011;186:540-544.
11. Rosamilia A, Dwyer PL. Pathophysiology of interstitial cystitis. Curr Opin Obstet Gynecol. 2000;12:405-410.
12. Grover S, Srivastava A, Lee R, et al. Role of inflammation in bladder function and interstitial cystitis. Ther Adv Urol. 2011;3:19-33.
13. Rosenberg MT, Page S, Hazzard MA. Prevalence of interstitial cystitis in a primary care setting. Urology. 2007;69(4 suppl):S48-S52.
14. Evans RJ. Treatment approaches for interstitial cystitis: multimodality therapy. Rev Urol. 2002;(4 suppl 1):S16-S20.
15. Wesselmann U. Interstitial cystitis: a chronic visceral pain syndrome. Urology. 2001;57(6 suppl 1):102.
16. Hosseini A, Ehrén I, Wiklund NP. Nitric oxide as an objective marker for evaluation of treatment response in patients with classic interstitial cystitis. J Urol. 2004;172(6 pt 1):2261-2265.
17. Ho MH, Bhatia NN, Khorram O. Physiologic role of nitric oxide and nitric oxide synthase in female lower urinary tract. Curr Opin Obstet Gynecol. 2004;16:423-429.
18. Rosenberg MT, Newman DK, Page SA. Interstitial cystitis/painful bladder syndrome: symptom recognition is key to early identification, treatment. Cleve Clin J Med. 2007;74:854-862.
19. Driscoll A, Teichman JM. How do patients with interstitial cystitis present? J Urol. 2001;166:2118-2120.
20. Parsons CL. Interstitial cystitis: epidemiology and clinical presentation. Clin Obstet Gynecol. 2002;45:242-249.
21. Hanno PM, Landis JR, Matthews-Cook Y, et al. The diagnosis of interstitial cystitis revisited: lessons learned from the National Institutes of Health Interstitial Cystitis Database study. J Urol. 1999;161:553-557.
22. Whitmore KE, Theoharides TC. When to suspect interstitial cystitis. J Fam Pract. 2011;60:340-348.
23. Butrick CW, Howard FM, Sand PK. Diagnosis and treatment of interstitial cystitis/painful bladder syndrome: a review. J Womens Health (Larchmt). 2010;19:1185-1193.
24. O’Leary MP, Sant GR, Fowler FJ Jr, et al. The interstitial cystitis symptom index and problem index. Urology. 1997;49(5A suppl):58-63.
25. Nickel JC. Forensic dissection of a clinical trial: lessons learned in understanding and managing interstitial cystitis. Rev Urol. 2010;12:e78-e85.
26. Anger JT, Zabihi N, Clemens JQ, et al. Treatment choice, duration, and cost in patients with interstitial cystitis and painful bladder syndrome. Int Urogynecol J. 2011;22:395-400.
27. Nickel JC. Interstitial cystitis: characterization and management of an enigmatic urologic syndrome. Rev Urol. 2002;4:112-121.
28. Dell JR. Interstitial cystitis/painful bladder syndrome: appropriate diagnosis and management. J Womens Health (Larchmt). 2007;16:1181-1187.
29. National Kidney and Urologic Diseases Information Clearinghouse. Interstitial cystitis/painful bladder syndrome (2011). NIH Publication No. 11–3220. http://kidney.niddk.nih.gov/kudiseases/pubs/interstitialcystitis/IC_PBS_T_508.pdf. Accessed June 5, 2013.
30. Davis EL, El Khoudary SR, Talbott EO, et al. Safety and efficacy of the use of intravesical and oral pentosan polysulfate sodium for interstitial cystitis: a randomized double-blind clinical trial. J Urol. 2008;179:177-185.
31. Pinto R, Lopes T, Silva J, et al. Persistent therapeutic effect of repeated injections of onabotulinum toxin a in refractory bladder pain syndrome/interstitial cystitis. J Urol. 2013;189:548-553.
32. Pinto R, Lopoes T, Frias B, et al. Trigonal injection of botulinum toxin A in patients with refractory bladder pain syndrome/interstitial cystitis. Eur Urol. 2010;58:360-365.
33. Gottsch HP, Miller JL, Yang CC, Berger RE. A pilot study of botulinum toxin for interstitial cystitis/painful bladder syndrome. Neurourol Urodyn. 2011; 30:93-96.
34. Neuhaus J, Schwalenberg T, Horn LC, et al. New aspects in the differential diagnosis and therapy of bladder pain syndrome/interstitial cystitis. Adv Urol. 2011;2011:639479.
1. Dasgupta J, Tincello DG. Interstitial cystitis/bladder pain syndrome: an update. Maturitas. 2009;64:212-217.
2. Hanno PM, Burks DA, Clemens JQ, et al. Diagnosis and treatment of interstitial cystitis/bladder pain syndrome: American Urological Association (AUA) Guideline (2011). www.auanet.org/education/guidelines/ic-bladder-pain-syndrome.cfm. Accessed June 5, 2013.
3. Evans RJ; University of Tennessee Advanced Studies in Pharmacy. Pathophysiology and clinical presentation of interstitial cystitis (2005). www.utasip.com/files/articlefiles/pdf/XASIP_Issue_Mar_p8_14.pdf. Accessed June 5, 2013.
4. Sant GR. Etiology, pathogenesis, and diagnosis of interstitial cystitis. Rev Urol. 2002;(4 suppl 1):S9-S15.
5. Abrams P, Cardozo L, Fall M, et al. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Neurourol Urodyn. 2002;21:167-178.
6. van de Merwe JP, Nordling J, Bouchelouche P, et al. Diagnostic criteria, classification, and nomenclature for painful bladder syndrome/interstitial cystitis: an ESSIC proposal. Eur Urol. 2008;53:60-67.
7. Bogart LM, Berry SH, Clemens JQ. Symptoms of interstitial cystitis, painful bladder syndrome and similar diseases in women: a systematic review. J Urol. 2007;177:450-456.
8. Kusek JW, Nyberg LM. The epidemiology of interstitial cystitis: is it time to expand our definition? Urology. 2001;57(6 suppl 1):95-99.
9. Clemens JQ, Meenan RT, Rosetti MC, et al. Prevalence and incidence of interstitial cystitis in a managed care population. J Urol. 2005;173:98-102.
10. Berry SH, Elliott MN, Suttorp M, et al. Prevalence of symptoms of bladder pain syndrome/interstitial cystitis among adult females in the United States. J Urol. 2011;186:540-544.
11. Rosamilia A, Dwyer PL. Pathophysiology of interstitial cystitis. Curr Opin Obstet Gynecol. 2000;12:405-410.
12. Grover S, Srivastava A, Lee R, et al. Role of inflammation in bladder function and interstitial cystitis. Ther Adv Urol. 2011;3:19-33.
13. Rosenberg MT, Page S, Hazzard MA. Prevalence of interstitial cystitis in a primary care setting. Urology. 2007;69(4 suppl):S48-S52.
14. Evans RJ. Treatment approaches for interstitial cystitis: multimodality therapy. Rev Urol. 2002;(4 suppl 1):S16-S20.
15. Wesselmann U. Interstitial cystitis: a chronic visceral pain syndrome. Urology. 2001;57(6 suppl 1):102.
16. Hosseini A, Ehrén I, Wiklund NP. Nitric oxide as an objective marker for evaluation of treatment response in patients with classic interstitial cystitis. J Urol. 2004;172(6 pt 1):2261-2265.
17. Ho MH, Bhatia NN, Khorram O. Physiologic role of nitric oxide and nitric oxide synthase in female lower urinary tract. Curr Opin Obstet Gynecol. 2004;16:423-429.
18. Rosenberg MT, Newman DK, Page SA. Interstitial cystitis/painful bladder syndrome: symptom recognition is key to early identification, treatment. Cleve Clin J Med. 2007;74:854-862.
19. Driscoll A, Teichman JM. How do patients with interstitial cystitis present? J Urol. 2001;166:2118-2120.
20. Parsons CL. Interstitial cystitis: epidemiology and clinical presentation. Clin Obstet Gynecol. 2002;45:242-249.
21. Hanno PM, Landis JR, Matthews-Cook Y, et al. The diagnosis of interstitial cystitis revisited: lessons learned from the National Institutes of Health Interstitial Cystitis Database study. J Urol. 1999;161:553-557.
22. Whitmore KE, Theoharides TC. When to suspect interstitial cystitis. J Fam Pract. 2011;60:340-348.
23. Butrick CW, Howard FM, Sand PK. Diagnosis and treatment of interstitial cystitis/painful bladder syndrome: a review. J Womens Health (Larchmt). 2010;19:1185-1193.
24. O’Leary MP, Sant GR, Fowler FJ Jr, et al. The interstitial cystitis symptom index and problem index. Urology. 1997;49(5A suppl):58-63.
25. Nickel JC. Forensic dissection of a clinical trial: lessons learned in understanding and managing interstitial cystitis. Rev Urol. 2010;12:e78-e85.
26. Anger JT, Zabihi N, Clemens JQ, et al. Treatment choice, duration, and cost in patients with interstitial cystitis and painful bladder syndrome. Int Urogynecol J. 2011;22:395-400.
27. Nickel JC. Interstitial cystitis: characterization and management of an enigmatic urologic syndrome. Rev Urol. 2002;4:112-121.
28. Dell JR. Interstitial cystitis/painful bladder syndrome: appropriate diagnosis and management. J Womens Health (Larchmt). 2007;16:1181-1187.
29. National Kidney and Urologic Diseases Information Clearinghouse. Interstitial cystitis/painful bladder syndrome (2011). NIH Publication No. 11–3220. http://kidney.niddk.nih.gov/kudiseases/pubs/interstitialcystitis/IC_PBS_T_508.pdf. Accessed June 5, 2013.
30. Davis EL, El Khoudary SR, Talbott EO, et al. Safety and efficacy of the use of intravesical and oral pentosan polysulfate sodium for interstitial cystitis: a randomized double-blind clinical trial. J Urol. 2008;179:177-185.
31. Pinto R, Lopes T, Silva J, et al. Persistent therapeutic effect of repeated injections of onabotulinum toxin a in refractory bladder pain syndrome/interstitial cystitis. J Urol. 2013;189:548-553.
32. Pinto R, Lopoes T, Frias B, et al. Trigonal injection of botulinum toxin A in patients with refractory bladder pain syndrome/interstitial cystitis. Eur Urol. 2010;58:360-365.
33. Gottsch HP, Miller JL, Yang CC, Berger RE. A pilot study of botulinum toxin for interstitial cystitis/painful bladder syndrome. Neurourol Urodyn. 2011; 30:93-96.
34. Neuhaus J, Schwalenberg T, Horn LC, et al. New aspects in the differential diagnosis and therapy of bladder pain syndrome/interstitial cystitis. Adv Urol. 2011;2011:639479.
FDA: No starch solutions for critically ill, cardiopulmonary bypass patients
The Food and Drug Administration is warning that hydroxyethyl starch solutions should no longer be used in the treatment of critically ill adult patients –including patients with sepsis and those admitted to an intensive care unit – after completing an analysis of data indicating that treatment with these solutions increases the risk of death and renal injury in these groups.
In a statement, the agency also recommended that the use of hydroxyethyl starch (HES) solutions, used as plasma volume expanders, be avoided in patients who are having open heart surgery with cardiopulmonary bypass, because of an increased risk of excessive bleeding.
The FDA is making these recommendations after completing an analysis of data from randomized controlled trials, meta-analyses, and observational studies in thousands of critically ill patients, and from a meta-analysis of 18 randomized controlled trials of almost 1,000 patients undergoing open heart surgery with cardiopulmonary bypass. The risks and benefits of HES products were the focus of a meeting in September 2012 convened by the FDA.
"Based on the totality of the evidence, [the] FDA considers increased mortality and renal injury requiring RRT [renal replacement therapy] in critically ill adult patients, including patients with sepsis and those admitted to the ICU, and excess bleeding in patients undergoing open heart surgery in association with cardiopulmonary bypass, to be HES class effects," according to the statement. The agency also recommended that health care professionals avoid the use of these products in patients with preexisting renal dysfunction, discontinue their use at the first sign of renal injury, and monitor renal function for at least 90 days in all patients. In addition, HES products should be discontinued "at the first sign of coagulopathy."
The data on critically ill adults included three double-blind, multicenter, randomized controlled studies published in 2012, which compared HES with saline solution or Ringer’s acetate, in patients with severe sepsis (two studies), or patients in the ICU who had sepsis, had undergone elective surgery, and had an APACHE II score of at least 25. In these studies, which monitored patients for 90 days, HES was associated with increased mortality and/or renal injury requiring RRT, the FDA statements said. The results of meta-analyses and observational studies in similar populations lend additional support to these findings, the statement added.
But there was no evidence of an increased risk of renal injury associated with these products in a review of 59 randomized controlled studies of adult and pediatric surgical patients who received HES in the operating room and were followed for less than a week. This could have been due to less exposure to the product, a relatively short follow-up, being relatively healthy, or some unknown factor, the statement said.
In the meta-analysis of studies of patients undergoing open heart surgery with cardiopulmonary bypass, the "use of different HES products, irrespective of molecular weight or degree of molar substitution, was associated with increased bleeding," according to the statement. The meta-analysis was published in 2012 (J. Thoracic Cardiovasc. Surg. 2012:144;223-30).
A boxed warning about the risk in ICU and septic patients is being added to the labels of HES products, and the information about the excessive bleeding risk in open heart surgery patients is being added to the warnings and precautions section.
There are four FDA-approved HES products for treating and preventing hypovolemia; they are used when plasma volume expansion is needed: HESPAN (6% HES 450/0.7a in sodium chloride injection) manufactured by B. Braun Medical); Hetastarch (6%) in 0.9% sodium chloride injection, a generic equivalent of Hespan, manufactured by Teva Pharmaceuticals USA; Hextend (6% HES 450/0.7 in physiologic solution), manufactured by BioTime; and Voluven (6% HES 130/0.4 in normal saline), manufactured by Fresenius Kabi USA.
The advisory is available at www.fda.gov/BiologicsBloodVaccines/SafetyAvailability/ucm358271.htm. Adverse events associated with HES solutions should be reported to the FDA’s MedWatch program at 80-332-1088 or www.fda.gov/Safety/MedWatch/HowToReport/default.htm.
The Food and Drug Administration is warning that hydroxyethyl starch solutions should no longer be used in the treatment of critically ill adult patients –including patients with sepsis and those admitted to an intensive care unit – after completing an analysis of data indicating that treatment with these solutions increases the risk of death and renal injury in these groups.
In a statement, the agency also recommended that the use of hydroxyethyl starch (HES) solutions, used as plasma volume expanders, be avoided in patients who are having open heart surgery with cardiopulmonary bypass, because of an increased risk of excessive bleeding.
The FDA is making these recommendations after completing an analysis of data from randomized controlled trials, meta-analyses, and observational studies in thousands of critically ill patients, and from a meta-analysis of 18 randomized controlled trials of almost 1,000 patients undergoing open heart surgery with cardiopulmonary bypass. The risks and benefits of HES products were the focus of a meeting in September 2012 convened by the FDA.
"Based on the totality of the evidence, [the] FDA considers increased mortality and renal injury requiring RRT [renal replacement therapy] in critically ill adult patients, including patients with sepsis and those admitted to the ICU, and excess bleeding in patients undergoing open heart surgery in association with cardiopulmonary bypass, to be HES class effects," according to the statement. The agency also recommended that health care professionals avoid the use of these products in patients with preexisting renal dysfunction, discontinue their use at the first sign of renal injury, and monitor renal function for at least 90 days in all patients. In addition, HES products should be discontinued "at the first sign of coagulopathy."
The data on critically ill adults included three double-blind, multicenter, randomized controlled studies published in 2012, which compared HES with saline solution or Ringer’s acetate, in patients with severe sepsis (two studies), or patients in the ICU who had sepsis, had undergone elective surgery, and had an APACHE II score of at least 25. In these studies, which monitored patients for 90 days, HES was associated with increased mortality and/or renal injury requiring RRT, the FDA statements said. The results of meta-analyses and observational studies in similar populations lend additional support to these findings, the statement added.
But there was no evidence of an increased risk of renal injury associated with these products in a review of 59 randomized controlled studies of adult and pediatric surgical patients who received HES in the operating room and were followed for less than a week. This could have been due to less exposure to the product, a relatively short follow-up, being relatively healthy, or some unknown factor, the statement said.
In the meta-analysis of studies of patients undergoing open heart surgery with cardiopulmonary bypass, the "use of different HES products, irrespective of molecular weight or degree of molar substitution, was associated with increased bleeding," according to the statement. The meta-analysis was published in 2012 (J. Thoracic Cardiovasc. Surg. 2012:144;223-30).
A boxed warning about the risk in ICU and septic patients is being added to the labels of HES products, and the information about the excessive bleeding risk in open heart surgery patients is being added to the warnings and precautions section.
There are four FDA-approved HES products for treating and preventing hypovolemia; they are used when plasma volume expansion is needed: HESPAN (6% HES 450/0.7a in sodium chloride injection) manufactured by B. Braun Medical); Hetastarch (6%) in 0.9% sodium chloride injection, a generic equivalent of Hespan, manufactured by Teva Pharmaceuticals USA; Hextend (6% HES 450/0.7 in physiologic solution), manufactured by BioTime; and Voluven (6% HES 130/0.4 in normal saline), manufactured by Fresenius Kabi USA.
The advisory is available at www.fda.gov/BiologicsBloodVaccines/SafetyAvailability/ucm358271.htm. Adverse events associated with HES solutions should be reported to the FDA’s MedWatch program at 80-332-1088 or www.fda.gov/Safety/MedWatch/HowToReport/default.htm.
The Food and Drug Administration is warning that hydroxyethyl starch solutions should no longer be used in the treatment of critically ill adult patients –including patients with sepsis and those admitted to an intensive care unit – after completing an analysis of data indicating that treatment with these solutions increases the risk of death and renal injury in these groups.
In a statement, the agency also recommended that the use of hydroxyethyl starch (HES) solutions, used as plasma volume expanders, be avoided in patients who are having open heart surgery with cardiopulmonary bypass, because of an increased risk of excessive bleeding.
The FDA is making these recommendations after completing an analysis of data from randomized controlled trials, meta-analyses, and observational studies in thousands of critically ill patients, and from a meta-analysis of 18 randomized controlled trials of almost 1,000 patients undergoing open heart surgery with cardiopulmonary bypass. The risks and benefits of HES products were the focus of a meeting in September 2012 convened by the FDA.
"Based on the totality of the evidence, [the] FDA considers increased mortality and renal injury requiring RRT [renal replacement therapy] in critically ill adult patients, including patients with sepsis and those admitted to the ICU, and excess bleeding in patients undergoing open heart surgery in association with cardiopulmonary bypass, to be HES class effects," according to the statement. The agency also recommended that health care professionals avoid the use of these products in patients with preexisting renal dysfunction, discontinue their use at the first sign of renal injury, and monitor renal function for at least 90 days in all patients. In addition, HES products should be discontinued "at the first sign of coagulopathy."
The data on critically ill adults included three double-blind, multicenter, randomized controlled studies published in 2012, which compared HES with saline solution or Ringer’s acetate, in patients with severe sepsis (two studies), or patients in the ICU who had sepsis, had undergone elective surgery, and had an APACHE II score of at least 25. In these studies, which monitored patients for 90 days, HES was associated with increased mortality and/or renal injury requiring RRT, the FDA statements said. The results of meta-analyses and observational studies in similar populations lend additional support to these findings, the statement added.
But there was no evidence of an increased risk of renal injury associated with these products in a review of 59 randomized controlled studies of adult and pediatric surgical patients who received HES in the operating room and were followed for less than a week. This could have been due to less exposure to the product, a relatively short follow-up, being relatively healthy, or some unknown factor, the statement said.
In the meta-analysis of studies of patients undergoing open heart surgery with cardiopulmonary bypass, the "use of different HES products, irrespective of molecular weight or degree of molar substitution, was associated with increased bleeding," according to the statement. The meta-analysis was published in 2012 (J. Thoracic Cardiovasc. Surg. 2012:144;223-30).
A boxed warning about the risk in ICU and septic patients is being added to the labels of HES products, and the information about the excessive bleeding risk in open heart surgery patients is being added to the warnings and precautions section.
There are four FDA-approved HES products for treating and preventing hypovolemia; they are used when plasma volume expansion is needed: HESPAN (6% HES 450/0.7a in sodium chloride injection) manufactured by B. Braun Medical); Hetastarch (6%) in 0.9% sodium chloride injection, a generic equivalent of Hespan, manufactured by Teva Pharmaceuticals USA; Hextend (6% HES 450/0.7 in physiologic solution), manufactured by BioTime; and Voluven (6% HES 130/0.4 in normal saline), manufactured by Fresenius Kabi USA.
The advisory is available at www.fda.gov/BiologicsBloodVaccines/SafetyAvailability/ucm358271.htm. Adverse events associated with HES solutions should be reported to the FDA’s MedWatch program at 80-332-1088 or www.fda.gov/Safety/MedWatch/HowToReport/default.htm.
Risk stratifying asymptomatic microscopic hematuria
Asymptomatic microscopic hematuria is a clinical challenge fraught with uncertainty, risk, and expense. With an estimated hematuria prevalence of 9%-18% and a threshold of at least 3 red blood cells per high-power field (RBC/HPF) as a cutoff for evaluation, we are all dealing with this problem – a lot. CT urograms, urine cytologies, and cystoscopies commonly compose the evaluation algorithm for this relatively low-prevalence disease.
What we need is a reliable and predictable way to place patients into different risk categories.
Dr. Ronald K. Loo of the Southern California Permanente Medical Group, Los Angeles, and his colleagues tried to answer this need by evaluating the performance of the Hematuria Risk Index (Mayo Clin. Proc. 2013;88:129-38).
The investigators assembled a prospective cohort of patients who were in the Kaiser Permanente system and had been referred to a urologist to undergo a full evaluation for asymptomatic microscopic hematuria. They derived the risk index from a "test cohort" composed of 2,630 patients, among whom 2.1% had a cancer detected and 1.9% had a pathologically confirmed urinary tract cancer.
The Hematuria Risk Index they developed is scored as follows: 4 points for gross hematuria and/or age at least 50 years and 1 point for a history of smoking, male gender, and/or greater than 25 RBC/HPF on recent urinalysis. The range is from 0 to 11 points, with patients stratified as low risk (0-4 points), moderate risk (5-8 points), or high risk (9-11 points).
Applying this risk index to a validation cohort, cancer was detected in 10.7% of the high-risk patients, 2.5% of the moderate-risk patients, and 0% of the low-risk patients.
Importantly, Dr. Loo and his associates concluded that microscopic hematuria is an unreliable indicator of urothelial malignancy. They further concluded that the risk of identifying a urinary tract cancer in anyone younger than 50 years without a history of gross hematuria is close to zero. Non-neoplastic findings included urinary stones, prostatic bleeding, urinary tract infection, and glomerular disease
This is a fantastically helpful study. Now, getting this Hematuria Risk Index as an app on my smartphone will make my year.
Dr. Ebbert is professor of medicine and a primary care clinician at the Mayo Clinic in Rochester, Minn. He reported having no relevant financial conflicts. The opinions expressed are those of the author. Reply via e-mail at [email protected].
Asymptomatic microscopic hematuria is a clinical challenge fraught with uncertainty, risk, and expense. With an estimated hematuria prevalence of 9%-18% and a threshold of at least 3 red blood cells per high-power field (RBC/HPF) as a cutoff for evaluation, we are all dealing with this problem – a lot. CT urograms, urine cytologies, and cystoscopies commonly compose the evaluation algorithm for this relatively low-prevalence disease.
What we need is a reliable and predictable way to place patients into different risk categories.
Dr. Ronald K. Loo of the Southern California Permanente Medical Group, Los Angeles, and his colleagues tried to answer this need by evaluating the performance of the Hematuria Risk Index (Mayo Clin. Proc. 2013;88:129-38).
The investigators assembled a prospective cohort of patients who were in the Kaiser Permanente system and had been referred to a urologist to undergo a full evaluation for asymptomatic microscopic hematuria. They derived the risk index from a "test cohort" composed of 2,630 patients, among whom 2.1% had a cancer detected and 1.9% had a pathologically confirmed urinary tract cancer.
The Hematuria Risk Index they developed is scored as follows: 4 points for gross hematuria and/or age at least 50 years and 1 point for a history of smoking, male gender, and/or greater than 25 RBC/HPF on recent urinalysis. The range is from 0 to 11 points, with patients stratified as low risk (0-4 points), moderate risk (5-8 points), or high risk (9-11 points).
Applying this risk index to a validation cohort, cancer was detected in 10.7% of the high-risk patients, 2.5% of the moderate-risk patients, and 0% of the low-risk patients.
Importantly, Dr. Loo and his associates concluded that microscopic hematuria is an unreliable indicator of urothelial malignancy. They further concluded that the risk of identifying a urinary tract cancer in anyone younger than 50 years without a history of gross hematuria is close to zero. Non-neoplastic findings included urinary stones, prostatic bleeding, urinary tract infection, and glomerular disease
This is a fantastically helpful study. Now, getting this Hematuria Risk Index as an app on my smartphone will make my year.
Dr. Ebbert is professor of medicine and a primary care clinician at the Mayo Clinic in Rochester, Minn. He reported having no relevant financial conflicts. The opinions expressed are those of the author. Reply via e-mail at [email protected].
Asymptomatic microscopic hematuria is a clinical challenge fraught with uncertainty, risk, and expense. With an estimated hematuria prevalence of 9%-18% and a threshold of at least 3 red blood cells per high-power field (RBC/HPF) as a cutoff for evaluation, we are all dealing with this problem – a lot. CT urograms, urine cytologies, and cystoscopies commonly compose the evaluation algorithm for this relatively low-prevalence disease.
What we need is a reliable and predictable way to place patients into different risk categories.
Dr. Ronald K. Loo of the Southern California Permanente Medical Group, Los Angeles, and his colleagues tried to answer this need by evaluating the performance of the Hematuria Risk Index (Mayo Clin. Proc. 2013;88:129-38).
The investigators assembled a prospective cohort of patients who were in the Kaiser Permanente system and had been referred to a urologist to undergo a full evaluation for asymptomatic microscopic hematuria. They derived the risk index from a "test cohort" composed of 2,630 patients, among whom 2.1% had a cancer detected and 1.9% had a pathologically confirmed urinary tract cancer.
The Hematuria Risk Index they developed is scored as follows: 4 points for gross hematuria and/or age at least 50 years and 1 point for a history of smoking, male gender, and/or greater than 25 RBC/HPF on recent urinalysis. The range is from 0 to 11 points, with patients stratified as low risk (0-4 points), moderate risk (5-8 points), or high risk (9-11 points).
Applying this risk index to a validation cohort, cancer was detected in 10.7% of the high-risk patients, 2.5% of the moderate-risk patients, and 0% of the low-risk patients.
Importantly, Dr. Loo and his associates concluded that microscopic hematuria is an unreliable indicator of urothelial malignancy. They further concluded that the risk of identifying a urinary tract cancer in anyone younger than 50 years without a history of gross hematuria is close to zero. Non-neoplastic findings included urinary stones, prostatic bleeding, urinary tract infection, and glomerular disease
This is a fantastically helpful study. Now, getting this Hematuria Risk Index as an app on my smartphone will make my year.
Dr. Ebbert is professor of medicine and a primary care clinician at the Mayo Clinic in Rochester, Minn. He reported having no relevant financial conflicts. The opinions expressed are those of the author. Reply via e-mail at [email protected].
Vancomycin, properly dosed, is as safe on the kidneys as linezolid
LAS VEGAS – Vancomycin is as safe on the kidneys of critically ill patients as linezolid, so long as trough levels don’t exceed the recommended upper limit of 20 mcg/mL, according to researchers from the University of Virginia in Charlottesville.
"When correct dosing is performed, the use of alternative agents to treat Gram-positive infections – specifically to avoid [vancomycin] nephrotoxicity – is unnecessary. Vancomycin use in critically ill patients is no different than linezolid use regarding nephrotoxicity or new-onset need for hemodialysis," said lead investigator Stephen Davies, a resident in the department of surgery, said at the annual meeting of the Surgical Infection Society.
The kidney safety of vancomycin has been in doubt, with mixed results from prior studies. To shed light on the issue, Dr. Davies and his team compared renal outcomes in 298 critically ill patients treated with vancomycin for 571 Gram-positive infections with outcomes in 247 patients treated with linezolid for 475 Gram-positive infections.
Infection sites included the lungs, peritoneum, blood stream, and urinary tract, among others, and isolates included Staphylococcus aureus (77 methicillin-resistant S. aureus), Enterococcus faecium (67 vancomycin-resistant Enterococcus), E. faecalis, and Streptococcus species. Vancomycin patients were dosed at 15-20 mg/kg, with serial trough monitoring, and treated for a mean of 16.2 days, vs. 14.3 days for linezolid. Patients were on no other nephrotoxic agents.
In the end, "there were no [statistically significant] differences between the two groups regarding maximum creatinine during treatment, final creatinine after treatment, change in creatinine maximum and initial values, and final and initial creatinine values." There were also no differences "in new-onset hemodialysis or death," Dr. Davies said.
APACHE II (Acute Physiology and Chronic Health Evaluation II) scores above 30 and initial creatinine levels above 1.2 mg/dL both predicted the need for new-onset hemodialysis, but antibiotic choice did not.
Those same two variables also predicted an increase in creatinine of more than 1.0 mg/dl during treatment; vancomycin use did, as well (relative risk, vancomycin 0.49; 95% confidence interval: 0.25-0.94). Concerned, the team looked into the issue. "What we found was that a rise in creatinine was typically not encountered until trough levels greater than 20 mg/dL were reached. As long as you stay within the recommended doses, you should be safe," Dr. Davies said.
There were no statistically significant baseline differences between the two groups in renal function, hemodialysis, creatinine levels, or APACHE II scores.
Despite the results, Dr. Philip Barie, who helped moderate Dr. Davies’ talk, said in an interview that he’s still concerned about vancomycin kidney safety.
"We are having to use higher doses to treat tougher bugs, and whereas nephrotoxicity pretty much went away with vancomycin after they purified the drug [decades ago], now we are having to use higher doses more, and nephrotoxicity is beginning to creep back into the picture. You have to dose really carefully, and if you have an organism that is among the more resistant to vancomycin, the safest thing to do with vancomycin is to use linezolid," said Dr. Barie, a professor of surgery and public heath at Cornell University, New York.
Dr. Davies and Dr. Barie reported no relevant disclosures.
Multiple factors must be considered in determining the optimal antibiotic treatment of gram-positive infections including but not limited to clinical and microbiological efficacy, development of resistant microorganisms, adverse effects, and costs. Several recent meta-analyses have reported conflicting results regarding whether linezolid is superior to vancomycin in clinical and/or microbiological efficacy, but all have reported increased nephrotoxicity with vancomycin (PLoS ONE 2013;8:e58240; Int. J. Antimicrob. Agents 2013;41:426-33; Eur. J. Clin. Microbiol. Infect. Dis. 2013).
However, none of these performed metaregression analyses to determine whether there is an association between vancomycin troughs and risk of nephrotoxicity. While the study by Dr. Davies et al. suggests such a relationship, there is inadequate data to determine if the higher vancomycin troughs, which were associated with creatinine elevations above 1.0 mg/dL, were necessary to eradicate the Gram-positive infections. Furthermore, despite the adjustment for severity of illness, there may have been a selection bias in the initial choice of antibiotics. Lastly, treatment selection for Gram-positive infections should also take into account: the costs and resources necessary to monitor vancomycin troughs, and the feasibility of performing timely and appropriate dose adjustments based on troughs to achieve a therapeutic yet nontoxic level.
Lillian S. Kao, MD, FACS, of the department of surgery, University of Texas Health Science Center at Houston.
Multiple factors must be considered in determining the optimal antibiotic treatment of gram-positive infections including but not limited to clinical and microbiological efficacy, development of resistant microorganisms, adverse effects, and costs. Several recent meta-analyses have reported conflicting results regarding whether linezolid is superior to vancomycin in clinical and/or microbiological efficacy, but all have reported increased nephrotoxicity with vancomycin (PLoS ONE 2013;8:e58240; Int. J. Antimicrob. Agents 2013;41:426-33; Eur. J. Clin. Microbiol. Infect. Dis. 2013).
However, none of these performed metaregression analyses to determine whether there is an association between vancomycin troughs and risk of nephrotoxicity. While the study by Dr. Davies et al. suggests such a relationship, there is inadequate data to determine if the higher vancomycin troughs, which were associated with creatinine elevations above 1.0 mg/dL, were necessary to eradicate the Gram-positive infections. Furthermore, despite the adjustment for severity of illness, there may have been a selection bias in the initial choice of antibiotics. Lastly, treatment selection for Gram-positive infections should also take into account: the costs and resources necessary to monitor vancomycin troughs, and the feasibility of performing timely and appropriate dose adjustments based on troughs to achieve a therapeutic yet nontoxic level.
Lillian S. Kao, MD, FACS, of the department of surgery, University of Texas Health Science Center at Houston.
Multiple factors must be considered in determining the optimal antibiotic treatment of gram-positive infections including but not limited to clinical and microbiological efficacy, development of resistant microorganisms, adverse effects, and costs. Several recent meta-analyses have reported conflicting results regarding whether linezolid is superior to vancomycin in clinical and/or microbiological efficacy, but all have reported increased nephrotoxicity with vancomycin (PLoS ONE 2013;8:e58240; Int. J. Antimicrob. Agents 2013;41:426-33; Eur. J. Clin. Microbiol. Infect. Dis. 2013).
However, none of these performed metaregression analyses to determine whether there is an association between vancomycin troughs and risk of nephrotoxicity. While the study by Dr. Davies et al. suggests such a relationship, there is inadequate data to determine if the higher vancomycin troughs, which were associated with creatinine elevations above 1.0 mg/dL, were necessary to eradicate the Gram-positive infections. Furthermore, despite the adjustment for severity of illness, there may have been a selection bias in the initial choice of antibiotics. Lastly, treatment selection for Gram-positive infections should also take into account: the costs and resources necessary to monitor vancomycin troughs, and the feasibility of performing timely and appropriate dose adjustments based on troughs to achieve a therapeutic yet nontoxic level.
Lillian S. Kao, MD, FACS, of the department of surgery, University of Texas Health Science Center at Houston.
LAS VEGAS – Vancomycin is as safe on the kidneys of critically ill patients as linezolid, so long as trough levels don’t exceed the recommended upper limit of 20 mcg/mL, according to researchers from the University of Virginia in Charlottesville.
"When correct dosing is performed, the use of alternative agents to treat Gram-positive infections – specifically to avoid [vancomycin] nephrotoxicity – is unnecessary. Vancomycin use in critically ill patients is no different than linezolid use regarding nephrotoxicity or new-onset need for hemodialysis," said lead investigator Stephen Davies, a resident in the department of surgery, said at the annual meeting of the Surgical Infection Society.
The kidney safety of vancomycin has been in doubt, with mixed results from prior studies. To shed light on the issue, Dr. Davies and his team compared renal outcomes in 298 critically ill patients treated with vancomycin for 571 Gram-positive infections with outcomes in 247 patients treated with linezolid for 475 Gram-positive infections.
Infection sites included the lungs, peritoneum, blood stream, and urinary tract, among others, and isolates included Staphylococcus aureus (77 methicillin-resistant S. aureus), Enterococcus faecium (67 vancomycin-resistant Enterococcus), E. faecalis, and Streptococcus species. Vancomycin patients were dosed at 15-20 mg/kg, with serial trough monitoring, and treated for a mean of 16.2 days, vs. 14.3 days for linezolid. Patients were on no other nephrotoxic agents.
In the end, "there were no [statistically significant] differences between the two groups regarding maximum creatinine during treatment, final creatinine after treatment, change in creatinine maximum and initial values, and final and initial creatinine values." There were also no differences "in new-onset hemodialysis or death," Dr. Davies said.
APACHE II (Acute Physiology and Chronic Health Evaluation II) scores above 30 and initial creatinine levels above 1.2 mg/dL both predicted the need for new-onset hemodialysis, but antibiotic choice did not.
Those same two variables also predicted an increase in creatinine of more than 1.0 mg/dl during treatment; vancomycin use did, as well (relative risk, vancomycin 0.49; 95% confidence interval: 0.25-0.94). Concerned, the team looked into the issue. "What we found was that a rise in creatinine was typically not encountered until trough levels greater than 20 mg/dL were reached. As long as you stay within the recommended doses, you should be safe," Dr. Davies said.
There were no statistically significant baseline differences between the two groups in renal function, hemodialysis, creatinine levels, or APACHE II scores.
Despite the results, Dr. Philip Barie, who helped moderate Dr. Davies’ talk, said in an interview that he’s still concerned about vancomycin kidney safety.
"We are having to use higher doses to treat tougher bugs, and whereas nephrotoxicity pretty much went away with vancomycin after they purified the drug [decades ago], now we are having to use higher doses more, and nephrotoxicity is beginning to creep back into the picture. You have to dose really carefully, and if you have an organism that is among the more resistant to vancomycin, the safest thing to do with vancomycin is to use linezolid," said Dr. Barie, a professor of surgery and public heath at Cornell University, New York.
Dr. Davies and Dr. Barie reported no relevant disclosures.
LAS VEGAS – Vancomycin is as safe on the kidneys of critically ill patients as linezolid, so long as trough levels don’t exceed the recommended upper limit of 20 mcg/mL, according to researchers from the University of Virginia in Charlottesville.
"When correct dosing is performed, the use of alternative agents to treat Gram-positive infections – specifically to avoid [vancomycin] nephrotoxicity – is unnecessary. Vancomycin use in critically ill patients is no different than linezolid use regarding nephrotoxicity or new-onset need for hemodialysis," said lead investigator Stephen Davies, a resident in the department of surgery, said at the annual meeting of the Surgical Infection Society.
The kidney safety of vancomycin has been in doubt, with mixed results from prior studies. To shed light on the issue, Dr. Davies and his team compared renal outcomes in 298 critically ill patients treated with vancomycin for 571 Gram-positive infections with outcomes in 247 patients treated with linezolid for 475 Gram-positive infections.
Infection sites included the lungs, peritoneum, blood stream, and urinary tract, among others, and isolates included Staphylococcus aureus (77 methicillin-resistant S. aureus), Enterococcus faecium (67 vancomycin-resistant Enterococcus), E. faecalis, and Streptococcus species. Vancomycin patients were dosed at 15-20 mg/kg, with serial trough monitoring, and treated for a mean of 16.2 days, vs. 14.3 days for linezolid. Patients were on no other nephrotoxic agents.
In the end, "there were no [statistically significant] differences between the two groups regarding maximum creatinine during treatment, final creatinine after treatment, change in creatinine maximum and initial values, and final and initial creatinine values." There were also no differences "in new-onset hemodialysis or death," Dr. Davies said.
APACHE II (Acute Physiology and Chronic Health Evaluation II) scores above 30 and initial creatinine levels above 1.2 mg/dL both predicted the need for new-onset hemodialysis, but antibiotic choice did not.
Those same two variables also predicted an increase in creatinine of more than 1.0 mg/dl during treatment; vancomycin use did, as well (relative risk, vancomycin 0.49; 95% confidence interval: 0.25-0.94). Concerned, the team looked into the issue. "What we found was that a rise in creatinine was typically not encountered until trough levels greater than 20 mg/dL were reached. As long as you stay within the recommended doses, you should be safe," Dr. Davies said.
There were no statistically significant baseline differences between the two groups in renal function, hemodialysis, creatinine levels, or APACHE II scores.
Despite the results, Dr. Philip Barie, who helped moderate Dr. Davies’ talk, said in an interview that he’s still concerned about vancomycin kidney safety.
"We are having to use higher doses to treat tougher bugs, and whereas nephrotoxicity pretty much went away with vancomycin after they purified the drug [decades ago], now we are having to use higher doses more, and nephrotoxicity is beginning to creep back into the picture. You have to dose really carefully, and if you have an organism that is among the more resistant to vancomycin, the safest thing to do with vancomycin is to use linezolid," said Dr. Barie, a professor of surgery and public heath at Cornell University, New York.
Dr. Davies and Dr. Barie reported no relevant disclosures.
AT THE SIS ANNUAL MEETING
Major finding: Vancomycin is as safe on the kidneys of critically ill patients as linezolid, so long as trough levels don’t exceed the recommended upper limit of 20 mcg/mL.
Data source: Single-center, retrospective cohort study in 545 critically ill patients
Disclosures: The lead investigator said he has no financial conflicts.
Sleep-disordered breathing and resistant hypertension
To the Editor: We recently read the article by Dr. Emmanuel Bravo.1 In his comprehensive paper, he defined a road map for the workup of resistant hypertension. Resistant hypertension is a challenging problem in everyday practice, with multiple pitfalls at each step from diagnosis to treatment.
Although not mentioned in the paper, obstructive sleep apnea is strongly associated with hypertension, and its prevalence in patients with resistant hypertension can be as high as 83%.2 The upper airway resistance syndrome is another form of sleep-disordered breathing in which transient increases in upper airway resistance result in repetitive electroencephalographic arousals. Unlike obstructive sleep apnea, upper airway resistance syndrome is not associated with apnea or diminished airflow, although snoring and excessive daytime somnolence are common. Repeated arousals, desaturations, or both during sleep lead to recurrent sympathetic surges with resultant nocturnal hypertension. There are a number of reports in the literature of large blood-pressure reductions after continuous positive airway pressure treatment.3
In conclusion, sleep-disordered breathing syndromes should be sought vigorously in cases of resistant hypertension, and every effort should be taken for proper management.
- Bravo E. Resistant hypertension: diagnostic strategies and management. Cleve Clin J Med 2013; 80:91–96.
- Logan AG, Perlikowski SM, Mente A, et al. High prevalence of unrecognized sleep apnoea in drug-resistant hypertension. J Hypertens 2001; 19:2271–2277.
- Logan AG, Tkacova R, Perlikowski SM, et al. Refractory hypertension and sleep apnoea: effect of CPAP on blood pressure and baroreflex. Eur Respir J 2003; 21:241–247.
To the Editor: We recently read the article by Dr. Emmanuel Bravo.1 In his comprehensive paper, he defined a road map for the workup of resistant hypertension. Resistant hypertension is a challenging problem in everyday practice, with multiple pitfalls at each step from diagnosis to treatment.
Although not mentioned in the paper, obstructive sleep apnea is strongly associated with hypertension, and its prevalence in patients with resistant hypertension can be as high as 83%.2 The upper airway resistance syndrome is another form of sleep-disordered breathing in which transient increases in upper airway resistance result in repetitive electroencephalographic arousals. Unlike obstructive sleep apnea, upper airway resistance syndrome is not associated with apnea or diminished airflow, although snoring and excessive daytime somnolence are common. Repeated arousals, desaturations, or both during sleep lead to recurrent sympathetic surges with resultant nocturnal hypertension. There are a number of reports in the literature of large blood-pressure reductions after continuous positive airway pressure treatment.3
In conclusion, sleep-disordered breathing syndromes should be sought vigorously in cases of resistant hypertension, and every effort should be taken for proper management.
To the Editor: We recently read the article by Dr. Emmanuel Bravo.1 In his comprehensive paper, he defined a road map for the workup of resistant hypertension. Resistant hypertension is a challenging problem in everyday practice, with multiple pitfalls at each step from diagnosis to treatment.
Although not mentioned in the paper, obstructive sleep apnea is strongly associated with hypertension, and its prevalence in patients with resistant hypertension can be as high as 83%.2 The upper airway resistance syndrome is another form of sleep-disordered breathing in which transient increases in upper airway resistance result in repetitive electroencephalographic arousals. Unlike obstructive sleep apnea, upper airway resistance syndrome is not associated with apnea or diminished airflow, although snoring and excessive daytime somnolence are common. Repeated arousals, desaturations, or both during sleep lead to recurrent sympathetic surges with resultant nocturnal hypertension. There are a number of reports in the literature of large blood-pressure reductions after continuous positive airway pressure treatment.3
In conclusion, sleep-disordered breathing syndromes should be sought vigorously in cases of resistant hypertension, and every effort should be taken for proper management.
- Bravo E. Resistant hypertension: diagnostic strategies and management. Cleve Clin J Med 2013; 80:91–96.
- Logan AG, Perlikowski SM, Mente A, et al. High prevalence of unrecognized sleep apnoea in drug-resistant hypertension. J Hypertens 2001; 19:2271–2277.
- Logan AG, Tkacova R, Perlikowski SM, et al. Refractory hypertension and sleep apnoea: effect of CPAP on blood pressure and baroreflex. Eur Respir J 2003; 21:241–247.
- Bravo E. Resistant hypertension: diagnostic strategies and management. Cleve Clin J Med 2013; 80:91–96.
- Logan AG, Perlikowski SM, Mente A, et al. High prevalence of unrecognized sleep apnoea in drug-resistant hypertension. J Hypertens 2001; 19:2271–2277.
- Logan AG, Tkacova R, Perlikowski SM, et al. Refractory hypertension and sleep apnoea: effect of CPAP on blood pressure and baroreflex. Eur Respir J 2003; 21:241–247.
Passing stones
Nephrolithiasis occurs in 5%-12% of the population and frequently on Friday afternoons. Eighty percent of them are calcium oxalate, 20% are in the ureter on presentation, and all of the ones we see hurt ... a lot. We may have more of these with the launch of a new weight-loss medication containing topiramate ER. Topiramate is a carbonic anhydrase inhibitor associated with an increased risk for serum metabolic acidosis and kidney stones.
We have become more comfortable conservatively managing uninfected stones even if there is a degree of hydronephrosis. Stones that are less than 5 mm in size have an 85% chance of passing spontaneously, those that are 5-10 mm have a 50% chance, and those larger than 8 mm have a 20% chance. A systematic review demonstrated the efficacy of tamsulosin for facilitating expulsion of distal ureteral stones less than 10 mm in size (19% improvement) (Urol. Int. 2012;89:107-15). Tamsulosin antagonizes the alpha-1 adrenergic receptors that are present throughout the ureter but have a high concentration in the ureter’s distal third.
But sometimes the stones are stubborn. Is there anything else we can do?
Building on the theory that kidney stones in the ureter cause inflammation, investigators in India conducted a clinical trial investigating the safety and efficacy of alpha-1 adrenergic receptor antagonists combined with prednisolone for the expulsion of distal ureter stones (Korean J. Urol. 2013;54:311-5).
A total of 120 adults presenting with distal ureteral stones (below common iliac vessels as assessed by CT) between 5 mm and 10 mm in size were randomized to one of three groups: A) 0.4 mg tamsulosin plus 5 mg prednisolone; B) naftopidil (a selective alpha-1 adrenergic receptor antagonist not available in the United States) plus 5 mg prednisolone; and C) watchful waiting. Prednisolone was continued for a maximum of 1 week, and the alpha-1 adrenergic receptor antagonist was continued for a maximum of 4 weeks. Patients received intramuscular diclofenac as needed for pain.
The stone expulsion rate was 70%, 87.5%, and 32.5% in groups A, B, and C, respectively. Expulsion rates for groups A and B were significantly greater than group C but not significantly different from one another. In group A, the expulsion rate in the first week was 12.5% and 65% in the second week. No patients expelled stones in the third or fourth week. Use of analgesics was significantly lower in groups A and B. No serious adverse events were noted.
This study does not actually inform us if tamsulosin alone is better than tamsulosin plus steroids, but a previous study from 2006 suggests that this is the case (Eur. Urol. 2006;50:339-44). However, that 2006 study used a steroid equivalent dose five times the dose used in the current study. Recall that prednisolone is equivalent to prednisone, and 5 mg is not huge.
So, for patients with no contraindications for steroids, this might be a reasonable option.
Dr. Ebbert is professor of medicine and a primary care clinician at the Mayo Clinic in Rochester, Minn. He reported having no relevant financial conflicts. The opinions expressed are those of the author.
Nephrolithiasis occurs in 5%-12% of the population and frequently on Friday afternoons. Eighty percent of them are calcium oxalate, 20% are in the ureter on presentation, and all of the ones we see hurt ... a lot. We may have more of these with the launch of a new weight-loss medication containing topiramate ER. Topiramate is a carbonic anhydrase inhibitor associated with an increased risk for serum metabolic acidosis and kidney stones.
We have become more comfortable conservatively managing uninfected stones even if there is a degree of hydronephrosis. Stones that are less than 5 mm in size have an 85% chance of passing spontaneously, those that are 5-10 mm have a 50% chance, and those larger than 8 mm have a 20% chance. A systematic review demonstrated the efficacy of tamsulosin for facilitating expulsion of distal ureteral stones less than 10 mm in size (19% improvement) (Urol. Int. 2012;89:107-15). Tamsulosin antagonizes the alpha-1 adrenergic receptors that are present throughout the ureter but have a high concentration in the ureter’s distal third.
But sometimes the stones are stubborn. Is there anything else we can do?
Building on the theory that kidney stones in the ureter cause inflammation, investigators in India conducted a clinical trial investigating the safety and efficacy of alpha-1 adrenergic receptor antagonists combined with prednisolone for the expulsion of distal ureter stones (Korean J. Urol. 2013;54:311-5).
A total of 120 adults presenting with distal ureteral stones (below common iliac vessels as assessed by CT) between 5 mm and 10 mm in size were randomized to one of three groups: A) 0.4 mg tamsulosin plus 5 mg prednisolone; B) naftopidil (a selective alpha-1 adrenergic receptor antagonist not available in the United States) plus 5 mg prednisolone; and C) watchful waiting. Prednisolone was continued for a maximum of 1 week, and the alpha-1 adrenergic receptor antagonist was continued for a maximum of 4 weeks. Patients received intramuscular diclofenac as needed for pain.
The stone expulsion rate was 70%, 87.5%, and 32.5% in groups A, B, and C, respectively. Expulsion rates for groups A and B were significantly greater than group C but not significantly different from one another. In group A, the expulsion rate in the first week was 12.5% and 65% in the second week. No patients expelled stones in the third or fourth week. Use of analgesics was significantly lower in groups A and B. No serious adverse events were noted.
This study does not actually inform us if tamsulosin alone is better than tamsulosin plus steroids, but a previous study from 2006 suggests that this is the case (Eur. Urol. 2006;50:339-44). However, that 2006 study used a steroid equivalent dose five times the dose used in the current study. Recall that prednisolone is equivalent to prednisone, and 5 mg is not huge.
So, for patients with no contraindications for steroids, this might be a reasonable option.
Dr. Ebbert is professor of medicine and a primary care clinician at the Mayo Clinic in Rochester, Minn. He reported having no relevant financial conflicts. The opinions expressed are those of the author.
Nephrolithiasis occurs in 5%-12% of the population and frequently on Friday afternoons. Eighty percent of them are calcium oxalate, 20% are in the ureter on presentation, and all of the ones we see hurt ... a lot. We may have more of these with the launch of a new weight-loss medication containing topiramate ER. Topiramate is a carbonic anhydrase inhibitor associated with an increased risk for serum metabolic acidosis and kidney stones.
We have become more comfortable conservatively managing uninfected stones even if there is a degree of hydronephrosis. Stones that are less than 5 mm in size have an 85% chance of passing spontaneously, those that are 5-10 mm have a 50% chance, and those larger than 8 mm have a 20% chance. A systematic review demonstrated the efficacy of tamsulosin for facilitating expulsion of distal ureteral stones less than 10 mm in size (19% improvement) (Urol. Int. 2012;89:107-15). Tamsulosin antagonizes the alpha-1 adrenergic receptors that are present throughout the ureter but have a high concentration in the ureter’s distal third.
But sometimes the stones are stubborn. Is there anything else we can do?
Building on the theory that kidney stones in the ureter cause inflammation, investigators in India conducted a clinical trial investigating the safety and efficacy of alpha-1 adrenergic receptor antagonists combined with prednisolone for the expulsion of distal ureter stones (Korean J. Urol. 2013;54:311-5).
A total of 120 adults presenting with distal ureteral stones (below common iliac vessels as assessed by CT) between 5 mm and 10 mm in size were randomized to one of three groups: A) 0.4 mg tamsulosin plus 5 mg prednisolone; B) naftopidil (a selective alpha-1 adrenergic receptor antagonist not available in the United States) plus 5 mg prednisolone; and C) watchful waiting. Prednisolone was continued for a maximum of 1 week, and the alpha-1 adrenergic receptor antagonist was continued for a maximum of 4 weeks. Patients received intramuscular diclofenac as needed for pain.
The stone expulsion rate was 70%, 87.5%, and 32.5% in groups A, B, and C, respectively. Expulsion rates for groups A and B were significantly greater than group C but not significantly different from one another. In group A, the expulsion rate in the first week was 12.5% and 65% in the second week. No patients expelled stones in the third or fourth week. Use of analgesics was significantly lower in groups A and B. No serious adverse events were noted.
This study does not actually inform us if tamsulosin alone is better than tamsulosin plus steroids, but a previous study from 2006 suggests that this is the case (Eur. Urol. 2006;50:339-44). However, that 2006 study used a steroid equivalent dose five times the dose used in the current study. Recall that prednisolone is equivalent to prednisone, and 5 mg is not huge.
So, for patients with no contraindications for steroids, this might be a reasonable option.
Dr. Ebbert is professor of medicine and a primary care clinician at the Mayo Clinic in Rochester, Minn. He reported having no relevant financial conflicts. The opinions expressed are those of the author.
Renal dysfunction improved after AF ablation
DENVER – Catheter ablation of atrial fibrillation in patients with comorbid moderate chronic kidney disease may have a side benefit: improved renal function, according to Dr. Nazem W. Akoum.
Among 81 patients with stage 3 or 4 chronic kidney disease (CKD) who underwent successful AF ablation, the mean estimated glomerular filtration rate (eGFR) improved significantly, from 46.7 mL/min per 1.73 m2 at baseline to 53 mL/min per 1.73 m2 at follow-up 3 months post procedure, Dr. Akoum reported at the annual meeting of the Heart Rhythm Society.
In contrast, the eGFR in 382 patients with stage 1 or 2 CKD who underwent AF ablation didn’t change significantly, from a baseline mean of 103.6 mL/min per 1.73 m2, added Dr. Akoum, an electrophysiologist at the University of Utah, Salt Lake City.
Patients with AF and stage 5 or 6 CKD were excluded from participation in this study. Given the requirement for preablation quantification of the extent of atrial fibrosis via delayed-enhancement MRI with gadolinium contrast, there were safety concerns regarding exposing patients with advanced CKD to gadolinium.
Patients with stage 1 or 2 CKD averaged 15.8% left atrial fibrosis, while stage 3 or 4 patients averaged 19.1%.
The improvement in eGFR noted in the group with stage 3 or 4 CKD following restoration of sinus rhythm appeared to be independent of left ventricular systolic performance or medication effects. The postablation regularization of ventricular activation and the resultant improvement in endothelial function may have contributed to the welcome renal side benefit, Dr. Akoum said.
He reported having no relevant financial conflicts.
DENVER – Catheter ablation of atrial fibrillation in patients with comorbid moderate chronic kidney disease may have a side benefit: improved renal function, according to Dr. Nazem W. Akoum.
Among 81 patients with stage 3 or 4 chronic kidney disease (CKD) who underwent successful AF ablation, the mean estimated glomerular filtration rate (eGFR) improved significantly, from 46.7 mL/min per 1.73 m2 at baseline to 53 mL/min per 1.73 m2 at follow-up 3 months post procedure, Dr. Akoum reported at the annual meeting of the Heart Rhythm Society.
In contrast, the eGFR in 382 patients with stage 1 or 2 CKD who underwent AF ablation didn’t change significantly, from a baseline mean of 103.6 mL/min per 1.73 m2, added Dr. Akoum, an electrophysiologist at the University of Utah, Salt Lake City.
Patients with AF and stage 5 or 6 CKD were excluded from participation in this study. Given the requirement for preablation quantification of the extent of atrial fibrosis via delayed-enhancement MRI with gadolinium contrast, there were safety concerns regarding exposing patients with advanced CKD to gadolinium.
Patients with stage 1 or 2 CKD averaged 15.8% left atrial fibrosis, while stage 3 or 4 patients averaged 19.1%.
The improvement in eGFR noted in the group with stage 3 or 4 CKD following restoration of sinus rhythm appeared to be independent of left ventricular systolic performance or medication effects. The postablation regularization of ventricular activation and the resultant improvement in endothelial function may have contributed to the welcome renal side benefit, Dr. Akoum said.
He reported having no relevant financial conflicts.
DENVER – Catheter ablation of atrial fibrillation in patients with comorbid moderate chronic kidney disease may have a side benefit: improved renal function, according to Dr. Nazem W. Akoum.
Among 81 patients with stage 3 or 4 chronic kidney disease (CKD) who underwent successful AF ablation, the mean estimated glomerular filtration rate (eGFR) improved significantly, from 46.7 mL/min per 1.73 m2 at baseline to 53 mL/min per 1.73 m2 at follow-up 3 months post procedure, Dr. Akoum reported at the annual meeting of the Heart Rhythm Society.
In contrast, the eGFR in 382 patients with stage 1 or 2 CKD who underwent AF ablation didn’t change significantly, from a baseline mean of 103.6 mL/min per 1.73 m2, added Dr. Akoum, an electrophysiologist at the University of Utah, Salt Lake City.
Patients with AF and stage 5 or 6 CKD were excluded from participation in this study. Given the requirement for preablation quantification of the extent of atrial fibrosis via delayed-enhancement MRI with gadolinium contrast, there were safety concerns regarding exposing patients with advanced CKD to gadolinium.
Patients with stage 1 or 2 CKD averaged 15.8% left atrial fibrosis, while stage 3 or 4 patients averaged 19.1%.
The improvement in eGFR noted in the group with stage 3 or 4 CKD following restoration of sinus rhythm appeared to be independent of left ventricular systolic performance or medication effects. The postablation regularization of ventricular activation and the resultant improvement in endothelial function may have contributed to the welcome renal side benefit, Dr. Akoum said.
He reported having no relevant financial conflicts.
AT HEART RHYTHM 2013
Major finding: Patients with stage 3 or 4 chronic kidney disease who underwent catheter ablation of atrial fibrillation showed significant improvement in renal function 3 months post procedure, with their estimated glomerular filtration rate improving from a baseline mean of 46.7 mL/min per 1.73 m2 to 53 mL/min per 1.73 m2.
Data source: A prospective study of 463 patients with stage 1-4 chronic kidney disease undergoing AF ablation.
Disclosures: Dr. Nazem W. Akoum reported having no relevant financial conflicts.
Novel biomarker may measure prostate cancer aggressiveness
SAN DIEGO – Prostate-specific antigen peptidase activity is higher in patients with less-aggressive prostate cancer than in patients with advanced disease, results from a pilot study demonstrated.
If the finding is confirmed by larger studies, this marker "may improve identification of men who may be better candidates for active surveillance," Dr. William J. Catalona said at the annual meeting of the American Urological Association. "In our study, if you considered those patients, it may have delayed or prevented surgery in 22% of this study population."
The marker, an assay developed by Ohmx under a National Institutes of Health small-business grant, "is a completely different approach to PSA testing," said Dr. Catalona, professor of urology at Northwestern University, Chicago.
"Current prostate cancer detection techniques generally suffer from a limited ability to differentiate indolent from aggressive prostate cancers," he explained. "We’re looking for tests that would detect life-threatening prostate cancer. That’s the real challenge in front of us today."
PSA peptidase activity was measured in a blinded study of 100 randomly selected patients who were treated with radical retropubic prostatectomy. Of the 100 patients, 50 had aggressive disease (defined as cancer resulting in prostate cancer–specific death, lymph node or distant metastases, seminal vesicle invasion, or extracapsular tumor extension), and 50 had nonaggressive disease (defined as cancer with a Gleason score of 6 or lower, pathologic stage T2, and no evidence of clinical or biochemical tumor recurrence on follow-up of 2-5 years). At surgery, fluid from the excised gland was gently milked from the apical urethral stump into a 2-mL conical vial and was immediately frozen at –80° C.
Next, researchers used a fluorogenic peptide probe to measure the level of proteolytic enzyme activity of PSA (aPSA) in each sample.
"All PSA tests measure the amount of PSA," said Dr. Catalona, who developed the PSA as a screening test for prostate cancer. "What this test focuses on is the enzymatic activity of PSA in prostatic fluid."
The aPSA value was significantly higher in patients with nonaggressive disease, compared with their counterparts who had advanced disease (a mean of 865 mcg/mL vs. a mean of 518 mcg/mL), a difference Dr. Catalona described as "striking." This reciprocal relationship between the PSA peptidase activity among the two patient groups "may improve identification of men who may be better candidates for active surveillance."
On receiver operating characteristic analysis, aPSA and the normalized ratio of aPSA to serum total PSA had the highest discriminatory power for predicting the presence of aggressive prostate cancer. Dr. Catalona estimated that using aPSA as an aggressiveness biomarker could result in 22% of the patients diagnosed with nonaggressive prostate cancer being able to avoid or delay radical prostatectomy.
Dr. Catalona said he and his associates plan to expand studies of the biomarker to include samples collected retrospectively before surgery, during attentive digital rectal exam.
The study was supported in part by a grant from the National Institutes of Health. Dr. Catalona disclosed that he received grant and research support from Beckman Coulter, Ohmx, and deCODE Genetics.
SAN DIEGO – Prostate-specific antigen peptidase activity is higher in patients with less-aggressive prostate cancer than in patients with advanced disease, results from a pilot study demonstrated.
If the finding is confirmed by larger studies, this marker "may improve identification of men who may be better candidates for active surveillance," Dr. William J. Catalona said at the annual meeting of the American Urological Association. "In our study, if you considered those patients, it may have delayed or prevented surgery in 22% of this study population."
The marker, an assay developed by Ohmx under a National Institutes of Health small-business grant, "is a completely different approach to PSA testing," said Dr. Catalona, professor of urology at Northwestern University, Chicago.
"Current prostate cancer detection techniques generally suffer from a limited ability to differentiate indolent from aggressive prostate cancers," he explained. "We’re looking for tests that would detect life-threatening prostate cancer. That’s the real challenge in front of us today."
PSA peptidase activity was measured in a blinded study of 100 randomly selected patients who were treated with radical retropubic prostatectomy. Of the 100 patients, 50 had aggressive disease (defined as cancer resulting in prostate cancer–specific death, lymph node or distant metastases, seminal vesicle invasion, or extracapsular tumor extension), and 50 had nonaggressive disease (defined as cancer with a Gleason score of 6 or lower, pathologic stage T2, and no evidence of clinical or biochemical tumor recurrence on follow-up of 2-5 years). At surgery, fluid from the excised gland was gently milked from the apical urethral stump into a 2-mL conical vial and was immediately frozen at –80° C.
Next, researchers used a fluorogenic peptide probe to measure the level of proteolytic enzyme activity of PSA (aPSA) in each sample.
"All PSA tests measure the amount of PSA," said Dr. Catalona, who developed the PSA as a screening test for prostate cancer. "What this test focuses on is the enzymatic activity of PSA in prostatic fluid."
The aPSA value was significantly higher in patients with nonaggressive disease, compared with their counterparts who had advanced disease (a mean of 865 mcg/mL vs. a mean of 518 mcg/mL), a difference Dr. Catalona described as "striking." This reciprocal relationship between the PSA peptidase activity among the two patient groups "may improve identification of men who may be better candidates for active surveillance."
On receiver operating characteristic analysis, aPSA and the normalized ratio of aPSA to serum total PSA had the highest discriminatory power for predicting the presence of aggressive prostate cancer. Dr. Catalona estimated that using aPSA as an aggressiveness biomarker could result in 22% of the patients diagnosed with nonaggressive prostate cancer being able to avoid or delay radical prostatectomy.
Dr. Catalona said he and his associates plan to expand studies of the biomarker to include samples collected retrospectively before surgery, during attentive digital rectal exam.
The study was supported in part by a grant from the National Institutes of Health. Dr. Catalona disclosed that he received grant and research support from Beckman Coulter, Ohmx, and deCODE Genetics.
SAN DIEGO – Prostate-specific antigen peptidase activity is higher in patients with less-aggressive prostate cancer than in patients with advanced disease, results from a pilot study demonstrated.
If the finding is confirmed by larger studies, this marker "may improve identification of men who may be better candidates for active surveillance," Dr. William J. Catalona said at the annual meeting of the American Urological Association. "In our study, if you considered those patients, it may have delayed or prevented surgery in 22% of this study population."
The marker, an assay developed by Ohmx under a National Institutes of Health small-business grant, "is a completely different approach to PSA testing," said Dr. Catalona, professor of urology at Northwestern University, Chicago.
"Current prostate cancer detection techniques generally suffer from a limited ability to differentiate indolent from aggressive prostate cancers," he explained. "We’re looking for tests that would detect life-threatening prostate cancer. That’s the real challenge in front of us today."
PSA peptidase activity was measured in a blinded study of 100 randomly selected patients who were treated with radical retropubic prostatectomy. Of the 100 patients, 50 had aggressive disease (defined as cancer resulting in prostate cancer–specific death, lymph node or distant metastases, seminal vesicle invasion, or extracapsular tumor extension), and 50 had nonaggressive disease (defined as cancer with a Gleason score of 6 or lower, pathologic stage T2, and no evidence of clinical or biochemical tumor recurrence on follow-up of 2-5 years). At surgery, fluid from the excised gland was gently milked from the apical urethral stump into a 2-mL conical vial and was immediately frozen at –80° C.
Next, researchers used a fluorogenic peptide probe to measure the level of proteolytic enzyme activity of PSA (aPSA) in each sample.
"All PSA tests measure the amount of PSA," said Dr. Catalona, who developed the PSA as a screening test for prostate cancer. "What this test focuses on is the enzymatic activity of PSA in prostatic fluid."
The aPSA value was significantly higher in patients with nonaggressive disease, compared with their counterparts who had advanced disease (a mean of 865 mcg/mL vs. a mean of 518 mcg/mL), a difference Dr. Catalona described as "striking." This reciprocal relationship between the PSA peptidase activity among the two patient groups "may improve identification of men who may be better candidates for active surveillance."
On receiver operating characteristic analysis, aPSA and the normalized ratio of aPSA to serum total PSA had the highest discriminatory power for predicting the presence of aggressive prostate cancer. Dr. Catalona estimated that using aPSA as an aggressiveness biomarker could result in 22% of the patients diagnosed with nonaggressive prostate cancer being able to avoid or delay radical prostatectomy.
Dr. Catalona said he and his associates plan to expand studies of the biomarker to include samples collected retrospectively before surgery, during attentive digital rectal exam.
The study was supported in part by a grant from the National Institutes of Health. Dr. Catalona disclosed that he received grant and research support from Beckman Coulter, Ohmx, and deCODE Genetics.
AT THE AUA ANNUAL MEETING
Major finding: The level of proteolytic enzyme activity of PSA (aPSA) was significantly higher in patients with nonaggressive prostate cancer than in their counterparts who had advanced disease (a mean of 865 mcg/mL vs. a mean of 518 mcg/mL).
Data source: A blinded study of 100 randomly selected patients who were treated with radical retropubic prostatectomy and who underwent aPSA measurement via a fluorogenic peptide probe.
Disclosures: The study was supported in part by a grant from the National Institutes of Health. Dr. Catalona disclosed that he received grant and research support from Beckman Coulter, Ohmx, and deCODE Genetics.
Angiotensin blockade linked to kidney injury in orthopedic surgery
NATIONAL HARBOR, MD. – Angiotensin receptor blockade on the morning of a major orthopedic surgery was associated with a tripling in the risk of acute kidney injury – a link probably driven by the drugs’ association with perioperative hypotension, a retrospective analysis has found.
"Our study does support the idea of withholding beta-blockers on the morning of these major procedures," Dr. Eileen Hennrikus reported at the annual meeting of the Society of Hospital Medicine.
Extant literature has confirmed a link between beta-blockers and hypotension in connection with cardiovascular surgery, but not orthopedic surgery. Dr. Hennrikus, of the Milton S. Hershey Medical Center, Hershey, Penn., examined the link in patients undergoing elective spine fusions, total hip or total knee replacement – procedures typically associated with greater blood loss than cardiovascular surgeries.
She used retrospective data from 922 patients who had undergone any of the three surgeries during 2010. Induction hypotension was defined as a systolic blood pressure of 80 mm Hg for at least 10 minutes within a half hour of anesthesia induction. Intraoperative hypotension was defined as a systolic blood pressure of 80 mm Hg for at least 10 minutes during maintenance anesthesia. The measure of acute kidney injury was an increase in serum creative of at least 0.3 mg/dL, or a 50% increase over preoperative levels.
Of the entire cohort, 37% (343) patients received their prescribed angiotensin receptor blocker or angiotensin converting enzyme inhibitor on the morning of their surgery. The incidence of induction hypotension was significantly greater in those patients in patients who took the medication than in those who did not (12% vs. 6.7%; OR 1.93). Acute kidney injury was also significantly more common among those who had the medications, however (8% vs. 2%; OR 5.4). Intraoperative hypotension was not significantly different between the groups.
Dr. Hennrikus conducted a multivariate analysis to further tease out the relationship between the medications and acute kidney injury. The regression controlled for age, medical comorbidities (diabetes, coronary artery disease, hypertension, and congestive heart failure), body mass index, medications (diuretics, beta-blockers, calcium channel blockers, general anesthesia-induction agents, and vasopressors), and blood loss.
After adjustment for all of those factors, angiotensin blockade conferred a threefold increase in the risk of acute kidney injury (OR 2.97). Although in the primary analysis, intraoperative hypotension had not been significantly associated with the drugs, it more than doubled the risk of kidney injury the risk (OR 2.6) in the multivariate analysis.
The model also revealed something of a surprise, Dr. Hennrikus said: Body mass index was an independent risk factor for acute kidney injury. For every 5 kg/m2 increase in BMI, the risk increased by 29% (OR 1.29).
Acute kidney injury exerted its own influence over hospital length of stay and mortality. The length of stay was significantly longer in patients who had kidney injury (6. days vs. 3 days). Two-year mortality was also significantly higher (6% vs. 2%).
Neither Dr. Hennrikus nor her coinvestigators reported having any financial conflicts.
NATIONAL HARBOR, MD. – Angiotensin receptor blockade on the morning of a major orthopedic surgery was associated with a tripling in the risk of acute kidney injury – a link probably driven by the drugs’ association with perioperative hypotension, a retrospective analysis has found.
"Our study does support the idea of withholding beta-blockers on the morning of these major procedures," Dr. Eileen Hennrikus reported at the annual meeting of the Society of Hospital Medicine.
Extant literature has confirmed a link between beta-blockers and hypotension in connection with cardiovascular surgery, but not orthopedic surgery. Dr. Hennrikus, of the Milton S. Hershey Medical Center, Hershey, Penn., examined the link in patients undergoing elective spine fusions, total hip or total knee replacement – procedures typically associated with greater blood loss than cardiovascular surgeries.
She used retrospective data from 922 patients who had undergone any of the three surgeries during 2010. Induction hypotension was defined as a systolic blood pressure of 80 mm Hg for at least 10 minutes within a half hour of anesthesia induction. Intraoperative hypotension was defined as a systolic blood pressure of 80 mm Hg for at least 10 minutes during maintenance anesthesia. The measure of acute kidney injury was an increase in serum creative of at least 0.3 mg/dL, or a 50% increase over preoperative levels.
Of the entire cohort, 37% (343) patients received their prescribed angiotensin receptor blocker or angiotensin converting enzyme inhibitor on the morning of their surgery. The incidence of induction hypotension was significantly greater in those patients in patients who took the medication than in those who did not (12% vs. 6.7%; OR 1.93). Acute kidney injury was also significantly more common among those who had the medications, however (8% vs. 2%; OR 5.4). Intraoperative hypotension was not significantly different between the groups.
Dr. Hennrikus conducted a multivariate analysis to further tease out the relationship between the medications and acute kidney injury. The regression controlled for age, medical comorbidities (diabetes, coronary artery disease, hypertension, and congestive heart failure), body mass index, medications (diuretics, beta-blockers, calcium channel blockers, general anesthesia-induction agents, and vasopressors), and blood loss.
After adjustment for all of those factors, angiotensin blockade conferred a threefold increase in the risk of acute kidney injury (OR 2.97). Although in the primary analysis, intraoperative hypotension had not been significantly associated with the drugs, it more than doubled the risk of kidney injury the risk (OR 2.6) in the multivariate analysis.
The model also revealed something of a surprise, Dr. Hennrikus said: Body mass index was an independent risk factor for acute kidney injury. For every 5 kg/m2 increase in BMI, the risk increased by 29% (OR 1.29).
Acute kidney injury exerted its own influence over hospital length of stay and mortality. The length of stay was significantly longer in patients who had kidney injury (6. days vs. 3 days). Two-year mortality was also significantly higher (6% vs. 2%).
Neither Dr. Hennrikus nor her coinvestigators reported having any financial conflicts.
NATIONAL HARBOR, MD. – Angiotensin receptor blockade on the morning of a major orthopedic surgery was associated with a tripling in the risk of acute kidney injury – a link probably driven by the drugs’ association with perioperative hypotension, a retrospective analysis has found.
"Our study does support the idea of withholding beta-blockers on the morning of these major procedures," Dr. Eileen Hennrikus reported at the annual meeting of the Society of Hospital Medicine.
Extant literature has confirmed a link between beta-blockers and hypotension in connection with cardiovascular surgery, but not orthopedic surgery. Dr. Hennrikus, of the Milton S. Hershey Medical Center, Hershey, Penn., examined the link in patients undergoing elective spine fusions, total hip or total knee replacement – procedures typically associated with greater blood loss than cardiovascular surgeries.
She used retrospective data from 922 patients who had undergone any of the three surgeries during 2010. Induction hypotension was defined as a systolic blood pressure of 80 mm Hg for at least 10 minutes within a half hour of anesthesia induction. Intraoperative hypotension was defined as a systolic blood pressure of 80 mm Hg for at least 10 minutes during maintenance anesthesia. The measure of acute kidney injury was an increase in serum creative of at least 0.3 mg/dL, or a 50% increase over preoperative levels.
Of the entire cohort, 37% (343) patients received their prescribed angiotensin receptor blocker or angiotensin converting enzyme inhibitor on the morning of their surgery. The incidence of induction hypotension was significantly greater in those patients in patients who took the medication than in those who did not (12% vs. 6.7%; OR 1.93). Acute kidney injury was also significantly more common among those who had the medications, however (8% vs. 2%; OR 5.4). Intraoperative hypotension was not significantly different between the groups.
Dr. Hennrikus conducted a multivariate analysis to further tease out the relationship between the medications and acute kidney injury. The regression controlled for age, medical comorbidities (diabetes, coronary artery disease, hypertension, and congestive heart failure), body mass index, medications (diuretics, beta-blockers, calcium channel blockers, general anesthesia-induction agents, and vasopressors), and blood loss.
After adjustment for all of those factors, angiotensin blockade conferred a threefold increase in the risk of acute kidney injury (OR 2.97). Although in the primary analysis, intraoperative hypotension had not been significantly associated with the drugs, it more than doubled the risk of kidney injury the risk (OR 2.6) in the multivariate analysis.
The model also revealed something of a surprise, Dr. Hennrikus said: Body mass index was an independent risk factor for acute kidney injury. For every 5 kg/m2 increase in BMI, the risk increased by 29% (OR 1.29).
Acute kidney injury exerted its own influence over hospital length of stay and mortality. The length of stay was significantly longer in patients who had kidney injury (6. days vs. 3 days). Two-year mortality was also significantly higher (6% vs. 2%).
Neither Dr. Hennrikus nor her coinvestigators reported having any financial conflicts.
At HOSPITAL MEDICINE 13
Major finding: Angiotensin axis blockade on the morning of major orthopedic surgery tripled the risk of acute kidney injury.
Data source: A retrospective analysis of 922 surgical patients.
Disclosures: Dr. Hennrikus reported having no financial conflicts of interest.