User login
Postop surveillance sufficient for stage I testicular cancer
Surveillance is sufficient for most men with stage I seminoma after successful orchiectomy, according to the findings of the largest study ever performed to address the issue.
The 1,822 men followed only with surveillance in Denmark had an excellent disease-specific survival of 99.5%, Dr. Mette Saskø Mortensen said at a press briefing highlighting research to be presented at the upcoming American Society of Clinical Oncology annual meeting. Only 10 men died of testicular cancer or treatment-related causes during a median follow-up of 15.4 years.
This finding means that for every 1,000 men followed by a surveillance program, only 4 will die within 10 years, said incoming ASCO president Dr. Clifford Hudis, chief of breast cancer medicine service at Memorial Sloan-Kettering Cancer Center in New York.
He described the study as one of several recent reminders that sometimes "less is more" in patient care and noted that opting for surveillance spares patients from the harmful side effects of chemotherapy and radiation without diminishing their chances for long-term survival.
Seminoma is a relatively rare cancer, but it is the most common solid tumor among young men. Initial treatment is typically orchiectomy, but no standard postoperative management strategy has been established. The current results will likely accelerate the trend toward surveillance in the United States, where roughly 50% of men undergo either radiotherapy or chemotherapy with carboplatin after surgery.
Overall, 355 (19.5%) of the 1,822 men experienced a relapse during surveillance, said Dr. Mortensen, a PhD student in the oncology department at Copenhagen University Hospital.
The median time to relapse was 13.7 months, with the majority of patients (72.4%) relapsing within the first 2 years. Another 20.3% relapsed within years 2-5 and 7.3% after 5 years.
"With only 19.5% of the patients relapsing, the surveillance strategy spares the remaining 80% of patients from unnecessary treatment after orchiectomy," Dr. Mortensen said. "Surveillance is a safe strategy for stage I seminoma patients."
Surveillance has been the main follow-up strategy for stage I seminoma in Denmark since 1984, and consists of 5 years of scheduled clinical visits, computed tomography scans/chest x-rays, and blood measurements of tumor markers. Men in the analysis were diagnosed from 1984 to 2008, and their data were collected up to December 2012 from patient files and linked national registries.
As observed in other smaller studies, the risk for relapse was increased with elevated human chorionic gonadotropin levels of more than 200 IU/L, vascular invasion, and tumors larger than 4 cm, Dr. Mortensen said.
The study was supported in part by the Danish Cancer Society, Danish Research Foundation and the Preben and Anna Simonsen Foundation. Dr. Mortensen reported having no financial disclosures.
Surveillance is sufficient for most men with stage I seminoma after successful orchiectomy, according to the findings of the largest study ever performed to address the issue.
The 1,822 men followed only with surveillance in Denmark had an excellent disease-specific survival of 99.5%, Dr. Mette Saskø Mortensen said at a press briefing highlighting research to be presented at the upcoming American Society of Clinical Oncology annual meeting. Only 10 men died of testicular cancer or treatment-related causes during a median follow-up of 15.4 years.
This finding means that for every 1,000 men followed by a surveillance program, only 4 will die within 10 years, said incoming ASCO president Dr. Clifford Hudis, chief of breast cancer medicine service at Memorial Sloan-Kettering Cancer Center in New York.
He described the study as one of several recent reminders that sometimes "less is more" in patient care and noted that opting for surveillance spares patients from the harmful side effects of chemotherapy and radiation without diminishing their chances for long-term survival.
Seminoma is a relatively rare cancer, but it is the most common solid tumor among young men. Initial treatment is typically orchiectomy, but no standard postoperative management strategy has been established. The current results will likely accelerate the trend toward surveillance in the United States, where roughly 50% of men undergo either radiotherapy or chemotherapy with carboplatin after surgery.
Overall, 355 (19.5%) of the 1,822 men experienced a relapse during surveillance, said Dr. Mortensen, a PhD student in the oncology department at Copenhagen University Hospital.
The median time to relapse was 13.7 months, with the majority of patients (72.4%) relapsing within the first 2 years. Another 20.3% relapsed within years 2-5 and 7.3% after 5 years.
"With only 19.5% of the patients relapsing, the surveillance strategy spares the remaining 80% of patients from unnecessary treatment after orchiectomy," Dr. Mortensen said. "Surveillance is a safe strategy for stage I seminoma patients."
Surveillance has been the main follow-up strategy for stage I seminoma in Denmark since 1984, and consists of 5 years of scheduled clinical visits, computed tomography scans/chest x-rays, and blood measurements of tumor markers. Men in the analysis were diagnosed from 1984 to 2008, and their data were collected up to December 2012 from patient files and linked national registries.
As observed in other smaller studies, the risk for relapse was increased with elevated human chorionic gonadotropin levels of more than 200 IU/L, vascular invasion, and tumors larger than 4 cm, Dr. Mortensen said.
The study was supported in part by the Danish Cancer Society, Danish Research Foundation and the Preben and Anna Simonsen Foundation. Dr. Mortensen reported having no financial disclosures.
Surveillance is sufficient for most men with stage I seminoma after successful orchiectomy, according to the findings of the largest study ever performed to address the issue.
The 1,822 men followed only with surveillance in Denmark had an excellent disease-specific survival of 99.5%, Dr. Mette Saskø Mortensen said at a press briefing highlighting research to be presented at the upcoming American Society of Clinical Oncology annual meeting. Only 10 men died of testicular cancer or treatment-related causes during a median follow-up of 15.4 years.
This finding means that for every 1,000 men followed by a surveillance program, only 4 will die within 10 years, said incoming ASCO president Dr. Clifford Hudis, chief of breast cancer medicine service at Memorial Sloan-Kettering Cancer Center in New York.
He described the study as one of several recent reminders that sometimes "less is more" in patient care and noted that opting for surveillance spares patients from the harmful side effects of chemotherapy and radiation without diminishing their chances for long-term survival.
Seminoma is a relatively rare cancer, but it is the most common solid tumor among young men. Initial treatment is typically orchiectomy, but no standard postoperative management strategy has been established. The current results will likely accelerate the trend toward surveillance in the United States, where roughly 50% of men undergo either radiotherapy or chemotherapy with carboplatin after surgery.
Overall, 355 (19.5%) of the 1,822 men experienced a relapse during surveillance, said Dr. Mortensen, a PhD student in the oncology department at Copenhagen University Hospital.
The median time to relapse was 13.7 months, with the majority of patients (72.4%) relapsing within the first 2 years. Another 20.3% relapsed within years 2-5 and 7.3% after 5 years.
"With only 19.5% of the patients relapsing, the surveillance strategy spares the remaining 80% of patients from unnecessary treatment after orchiectomy," Dr. Mortensen said. "Surveillance is a safe strategy for stage I seminoma patients."
Surveillance has been the main follow-up strategy for stage I seminoma in Denmark since 1984, and consists of 5 years of scheduled clinical visits, computed tomography scans/chest x-rays, and blood measurements of tumor markers. Men in the analysis were diagnosed from 1984 to 2008, and their data were collected up to December 2012 from patient files and linked national registries.
As observed in other smaller studies, the risk for relapse was increased with elevated human chorionic gonadotropin levels of more than 200 IU/L, vascular invasion, and tumors larger than 4 cm, Dr. Mortensen said.
The study was supported in part by the Danish Cancer Society, Danish Research Foundation and the Preben and Anna Simonsen Foundation. Dr. Mortensen reported having no financial disclosures.
AT THE ASCO 2013 PRESSCAST
Major finding: The 10-year cancer-specific survival was 99.6%.
Data source: Retrospective, nationwide cohort study of surveillance in 1,822 men with stage I seminoma.
Disclosures: The study was supported in part by the Danish Cancer Society, Danish Research Foundation, and the Preben and Anna Simonsen Foundation. Dr. Mortensen reported having no financial disclosures.
FDA approves oral nimodipine solution for enteral use only
An oral liquid formulation of nimodipine has been approved by the Food and Drug Administration to help reduce serious and fatal medication errors that have occurred when the drug is extracted for intravenous administration from the only previously approved formulation, a liquid-filled gel capsule, the agency announced on May 14.
The nimodipine oral solution was approved on May 10 for improving neurologic outcomes in adults who have had a subarachnoid hemorrhage. The liquid-filled gel capsule formulation was approved in 1988.
The new oral formulation, which will be marketed as Nymalize by Arbor Pharmaceuticals, can be administered orally or via a nasogastric or gastric tube, "and there is no need for a needle to be used, which is what caused past medication errors," Dr. Russell Katz, director of the division of neurology products in the FDA’s Center for Drug Evaluation and Research, said in an FDA statement. "Having an oral version of this product may help reduce the medication errors we’ve seen from erroneous intravenous administration of the contents of oral capsules," he added.
The FDA has received reports of serious adverse events, including fatalities, associated with intravenous administration of the liquid contents of nimodipine capsules, including deaths, cardiac arrest, severe drops in blood pressure, and other cardiac complications. In 2006, a boxed warning was added to the prescribing information of nimodipine, a dihydropyridine calcium channel blocker, cautioning against intravenous use of the drug. And, in August 2010, the FDA issued a drug safety notice alerting health care professionals that nimodipine capsules should be administered only by mouth or through a feeding tube, and that it should "never" be administered intravenously.
The approved indication for liquid nimodipine is for the "improvement of neurological outcome by reducing the incidence and severity of ischemic deficits in adult patients with subarachnoid hemorrhage from ruptured intracranial berry aneurysms regardless of their post-ictus neurological condition (i.e., Hunt and Hess Grades I-V)."
The new formulation’s approval was based on clinical studies of nimodipine oral capsules in patients with subarachnoid hemorrhage. The prescribing information says that the bioavailability of nimodipine oral solution is comparable with that of nimodipine oral capsules, that hypotension is the most common adverse event in trials, and that blood pressure should be carefully monitored during treatment.
The prescribing information is available here.
Serious adverse events associated with Nymalize should be reported to the FDA’s MedWatch program or at 800-332-1088.
An oral liquid formulation of nimodipine has been approved by the Food and Drug Administration to help reduce serious and fatal medication errors that have occurred when the drug is extracted for intravenous administration from the only previously approved formulation, a liquid-filled gel capsule, the agency announced on May 14.
The nimodipine oral solution was approved on May 10 for improving neurologic outcomes in adults who have had a subarachnoid hemorrhage. The liquid-filled gel capsule formulation was approved in 1988.
The new oral formulation, which will be marketed as Nymalize by Arbor Pharmaceuticals, can be administered orally or via a nasogastric or gastric tube, "and there is no need for a needle to be used, which is what caused past medication errors," Dr. Russell Katz, director of the division of neurology products in the FDA’s Center for Drug Evaluation and Research, said in an FDA statement. "Having an oral version of this product may help reduce the medication errors we’ve seen from erroneous intravenous administration of the contents of oral capsules," he added.
The FDA has received reports of serious adverse events, including fatalities, associated with intravenous administration of the liquid contents of nimodipine capsules, including deaths, cardiac arrest, severe drops in blood pressure, and other cardiac complications. In 2006, a boxed warning was added to the prescribing information of nimodipine, a dihydropyridine calcium channel blocker, cautioning against intravenous use of the drug. And, in August 2010, the FDA issued a drug safety notice alerting health care professionals that nimodipine capsules should be administered only by mouth or through a feeding tube, and that it should "never" be administered intravenously.
The approved indication for liquid nimodipine is for the "improvement of neurological outcome by reducing the incidence and severity of ischemic deficits in adult patients with subarachnoid hemorrhage from ruptured intracranial berry aneurysms regardless of their post-ictus neurological condition (i.e., Hunt and Hess Grades I-V)."
The new formulation’s approval was based on clinical studies of nimodipine oral capsules in patients with subarachnoid hemorrhage. The prescribing information says that the bioavailability of nimodipine oral solution is comparable with that of nimodipine oral capsules, that hypotension is the most common adverse event in trials, and that blood pressure should be carefully monitored during treatment.
The prescribing information is available here.
Serious adverse events associated with Nymalize should be reported to the FDA’s MedWatch program or at 800-332-1088.
An oral liquid formulation of nimodipine has been approved by the Food and Drug Administration to help reduce serious and fatal medication errors that have occurred when the drug is extracted for intravenous administration from the only previously approved formulation, a liquid-filled gel capsule, the agency announced on May 14.
The nimodipine oral solution was approved on May 10 for improving neurologic outcomes in adults who have had a subarachnoid hemorrhage. The liquid-filled gel capsule formulation was approved in 1988.
The new oral formulation, which will be marketed as Nymalize by Arbor Pharmaceuticals, can be administered orally or via a nasogastric or gastric tube, "and there is no need for a needle to be used, which is what caused past medication errors," Dr. Russell Katz, director of the division of neurology products in the FDA’s Center for Drug Evaluation and Research, said in an FDA statement. "Having an oral version of this product may help reduce the medication errors we’ve seen from erroneous intravenous administration of the contents of oral capsules," he added.
The FDA has received reports of serious adverse events, including fatalities, associated with intravenous administration of the liquid contents of nimodipine capsules, including deaths, cardiac arrest, severe drops in blood pressure, and other cardiac complications. In 2006, a boxed warning was added to the prescribing information of nimodipine, a dihydropyridine calcium channel blocker, cautioning against intravenous use of the drug. And, in August 2010, the FDA issued a drug safety notice alerting health care professionals that nimodipine capsules should be administered only by mouth or through a feeding tube, and that it should "never" be administered intravenously.
The approved indication for liquid nimodipine is for the "improvement of neurological outcome by reducing the incidence and severity of ischemic deficits in adult patients with subarachnoid hemorrhage from ruptured intracranial berry aneurysms regardless of their post-ictus neurological condition (i.e., Hunt and Hess Grades I-V)."
The new formulation’s approval was based on clinical studies of nimodipine oral capsules in patients with subarachnoid hemorrhage. The prescribing information says that the bioavailability of nimodipine oral solution is comparable with that of nimodipine oral capsules, that hypotension is the most common adverse event in trials, and that blood pressure should be carefully monitored during treatment.
The prescribing information is available here.
Serious adverse events associated with Nymalize should be reported to the FDA’s MedWatch program or at 800-332-1088.
Endovascular AAA repair superior for kidney disease patients
INDIANAPOLIS – Contrary to conventional wisdom, endovascular aneurysm repair (EVAR) provides outcomes superior to those achieved with open surgical repair of abdominal aortic aneurysm in patients with chronic renal insufficiency, a large study indicates.
"EVAR should be the first-line therapy in the patient with chronic renal insufficiency when the patient has the appropriate anatomy. However, in patients with severe renal impairment, a higher threshold should be applied for repair because the risks of both open repair and EVAR are significantly higher," Dr. Bao-Ngoc H. Nguyen declared at the annual meeting of the American Surgical Association.
"Chronic renal failure is quite prevalent in patients with abdominal aortic aneurysm: up to 30%. It is quite worrisome because any further decline in renal function in these patients could push them toward dialysis. More than that, postoperative renal failure is a predictor for early and late mortality," noted Dr. Nguyen of George Washington University, Washington.
She presented a retrospective study in patients with abdominal aortic aneurysm and chronic kidney disease. The aim, she explained, was to answer a key question: "Which one of these two treatment modalities is the lesser of two evils?"
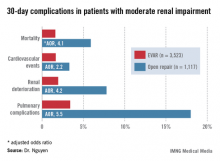
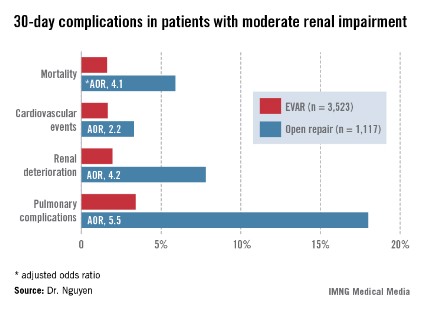
For answers, Dr. Nguyen and coinvestigators turned to the American College of Surgeons National Quality Improvement Program (NSQIP) database for 2005-2010. They identified 3,523 patients with moderate chronic renal insufficiency, defined as an estimated glomerular filtration rate (eGFR) of 30-60 mL/minute, who underwent EVAR for abdominal aortic aneurysm and 1,117 treated via open surgical repair. Another 363 EVAR patients had severe chronic renal insufficiency, with an eGFR of less than 30 mL/minute, as did 139 patients who underwent open repair. Vascular surgeons performed all procedures in this study.
Patients with moderate renal insufficiency who underwent EVAR had markedly lower 30-day rates of mortality, pulmonary complications, cardiovascular events, and postoperative renal dysfunction, including acute kidney injury, than did those who had open surgical repair. One or more adverse events occurred in 6% of the EVAR group, compared with 24.1% of open repair patients. In a multivariate analysis controlled for preoperative differences in the patient groups, those undergoing open repair had an adjusted 4.1-fold greater risk of mortality as well as a 2.2-fold increased risk of cardiovascular events, a 4.2-fold increased risk of renal deterioration including a 5.2-fold greater risk of dialysis, and additional hazards.
In contrast, among the much smaller population of patients with baseline severe chronic renal insufficiency, there was no significant difference between the two treatment groups in terms of 30-day mortality, postoperative renal deterioration, or cardiovascular complications, although pulmonary complications were an adjusted fivefold more likely in the open surgery than among EVAR patients. Of note, rates of all adverse outcomes were markedly higher in both groups than in those with moderate chronic renal insufficiency, such that one or more adverse events occurred in 16.9% of EVAR patients and 42.5% of the open repair patients with severe chronic renal insufficiency.
Discussant Dr. Michael Watkins commented that this study has one glaring shortcoming resulting from a limitation of the NSQIP database.
While NSQIP contains only validated data entered by unbiased, well-trained professionals and NSQIP is "far superior" to the various administrative databases commonly used in evaluating outcomes, it doesn’t include key details about patients’ presenting anatomy, observed Dr. Watkins, director of the vascular research laboratory at Massachusetts General Hospital, Boston.
"Was the anatomy really similar in the two groups, or were patients who underwent open repair not candidates for EVAR?" he asked.
Dr. Nguyen conceded that this constitutes a major study limitation, adding that she agrees with Dr. Watkins that anatomy should be the first and foremost factor considered in deciding upon the surgical approach in abdominal aortic aneurysm repair.
She reported having no financial conflicts.
INDIANAPOLIS – Contrary to conventional wisdom, endovascular aneurysm repair (EVAR) provides outcomes superior to those achieved with open surgical repair of abdominal aortic aneurysm in patients with chronic renal insufficiency, a large study indicates.
"EVAR should be the first-line therapy in the patient with chronic renal insufficiency when the patient has the appropriate anatomy. However, in patients with severe renal impairment, a higher threshold should be applied for repair because the risks of both open repair and EVAR are significantly higher," Dr. Bao-Ngoc H. Nguyen declared at the annual meeting of the American Surgical Association.
"Chronic renal failure is quite prevalent in patients with abdominal aortic aneurysm: up to 30%. It is quite worrisome because any further decline in renal function in these patients could push them toward dialysis. More than that, postoperative renal failure is a predictor for early and late mortality," noted Dr. Nguyen of George Washington University, Washington.
She presented a retrospective study in patients with abdominal aortic aneurysm and chronic kidney disease. The aim, she explained, was to answer a key question: "Which one of these two treatment modalities is the lesser of two evils?"
For answers, Dr. Nguyen and coinvestigators turned to the American College of Surgeons National Quality Improvement Program (NSQIP) database for 2005-2010. They identified 3,523 patients with moderate chronic renal insufficiency, defined as an estimated glomerular filtration rate (eGFR) of 30-60 mL/minute, who underwent EVAR for abdominal aortic aneurysm and 1,117 treated via open surgical repair. Another 363 EVAR patients had severe chronic renal insufficiency, with an eGFR of less than 30 mL/minute, as did 139 patients who underwent open repair. Vascular surgeons performed all procedures in this study.
Patients with moderate renal insufficiency who underwent EVAR had markedly lower 30-day rates of mortality, pulmonary complications, cardiovascular events, and postoperative renal dysfunction, including acute kidney injury, than did those who had open surgical repair. One or more adverse events occurred in 6% of the EVAR group, compared with 24.1% of open repair patients. In a multivariate analysis controlled for preoperative differences in the patient groups, those undergoing open repair had an adjusted 4.1-fold greater risk of mortality as well as a 2.2-fold increased risk of cardiovascular events, a 4.2-fold increased risk of renal deterioration including a 5.2-fold greater risk of dialysis, and additional hazards.
In contrast, among the much smaller population of patients with baseline severe chronic renal insufficiency, there was no significant difference between the two treatment groups in terms of 30-day mortality, postoperative renal deterioration, or cardiovascular complications, although pulmonary complications were an adjusted fivefold more likely in the open surgery than among EVAR patients. Of note, rates of all adverse outcomes were markedly higher in both groups than in those with moderate chronic renal insufficiency, such that one or more adverse events occurred in 16.9% of EVAR patients and 42.5% of the open repair patients with severe chronic renal insufficiency.
Discussant Dr. Michael Watkins commented that this study has one glaring shortcoming resulting from a limitation of the NSQIP database.
While NSQIP contains only validated data entered by unbiased, well-trained professionals and NSQIP is "far superior" to the various administrative databases commonly used in evaluating outcomes, it doesn’t include key details about patients’ presenting anatomy, observed Dr. Watkins, director of the vascular research laboratory at Massachusetts General Hospital, Boston.
"Was the anatomy really similar in the two groups, or were patients who underwent open repair not candidates for EVAR?" he asked.
Dr. Nguyen conceded that this constitutes a major study limitation, adding that she agrees with Dr. Watkins that anatomy should be the first and foremost factor considered in deciding upon the surgical approach in abdominal aortic aneurysm repair.
She reported having no financial conflicts.
INDIANAPOLIS – Contrary to conventional wisdom, endovascular aneurysm repair (EVAR) provides outcomes superior to those achieved with open surgical repair of abdominal aortic aneurysm in patients with chronic renal insufficiency, a large study indicates.
"EVAR should be the first-line therapy in the patient with chronic renal insufficiency when the patient has the appropriate anatomy. However, in patients with severe renal impairment, a higher threshold should be applied for repair because the risks of both open repair and EVAR are significantly higher," Dr. Bao-Ngoc H. Nguyen declared at the annual meeting of the American Surgical Association.
"Chronic renal failure is quite prevalent in patients with abdominal aortic aneurysm: up to 30%. It is quite worrisome because any further decline in renal function in these patients could push them toward dialysis. More than that, postoperative renal failure is a predictor for early and late mortality," noted Dr. Nguyen of George Washington University, Washington.
She presented a retrospective study in patients with abdominal aortic aneurysm and chronic kidney disease. The aim, she explained, was to answer a key question: "Which one of these two treatment modalities is the lesser of two evils?"
For answers, Dr. Nguyen and coinvestigators turned to the American College of Surgeons National Quality Improvement Program (NSQIP) database for 2005-2010. They identified 3,523 patients with moderate chronic renal insufficiency, defined as an estimated glomerular filtration rate (eGFR) of 30-60 mL/minute, who underwent EVAR for abdominal aortic aneurysm and 1,117 treated via open surgical repair. Another 363 EVAR patients had severe chronic renal insufficiency, with an eGFR of less than 30 mL/minute, as did 139 patients who underwent open repair. Vascular surgeons performed all procedures in this study.
Patients with moderate renal insufficiency who underwent EVAR had markedly lower 30-day rates of mortality, pulmonary complications, cardiovascular events, and postoperative renal dysfunction, including acute kidney injury, than did those who had open surgical repair. One or more adverse events occurred in 6% of the EVAR group, compared with 24.1% of open repair patients. In a multivariate analysis controlled for preoperative differences in the patient groups, those undergoing open repair had an adjusted 4.1-fold greater risk of mortality as well as a 2.2-fold increased risk of cardiovascular events, a 4.2-fold increased risk of renal deterioration including a 5.2-fold greater risk of dialysis, and additional hazards.
In contrast, among the much smaller population of patients with baseline severe chronic renal insufficiency, there was no significant difference between the two treatment groups in terms of 30-day mortality, postoperative renal deterioration, or cardiovascular complications, although pulmonary complications were an adjusted fivefold more likely in the open surgery than among EVAR patients. Of note, rates of all adverse outcomes were markedly higher in both groups than in those with moderate chronic renal insufficiency, such that one or more adverse events occurred in 16.9% of EVAR patients and 42.5% of the open repair patients with severe chronic renal insufficiency.
Discussant Dr. Michael Watkins commented that this study has one glaring shortcoming resulting from a limitation of the NSQIP database.
While NSQIP contains only validated data entered by unbiased, well-trained professionals and NSQIP is "far superior" to the various administrative databases commonly used in evaluating outcomes, it doesn’t include key details about patients’ presenting anatomy, observed Dr. Watkins, director of the vascular research laboratory at Massachusetts General Hospital, Boston.
"Was the anatomy really similar in the two groups, or were patients who underwent open repair not candidates for EVAR?" he asked.
Dr. Nguyen conceded that this constitutes a major study limitation, adding that she agrees with Dr. Watkins that anatomy should be the first and foremost factor considered in deciding upon the surgical approach in abdominal aortic aneurysm repair.
She reported having no financial conflicts.
AT THE ASA ANNUAL MEETING
Major Finding: Patients with moderate chronic renal insufficiency who underwent open surgical repair of abdominal aortic aneurysm had a 4.2-fold greater risk of postoperative renal deterioration than did those who had an endovascular aneurysm repair.
Data Source: A retrospective study of a large national surgical database.
Disclosures: The presenter reported having no conflicts of interest.
IOM: Cut daily sodium, but not below 2,300 mg
Americans should lower sodium consumption to optimize their health – but not below levels of 2,300 mg per day, according to a new report from the Institute of Medicine.
People aged 51 years and older, African-Americans, and those with hypertension, diabetes, or chronic kidney disease can follow the same advice, according to authors of the report, released during a webinar May 14.
That recommendation is a departure from the current Dietary Guidelines for Americans from the U.S. Department of Health and Human Services, which recommends that these higher-risk groups limit sodium intake to 1,500 mg daily. The HHS guidelines recommend that most other people aged 14-50 years limit sodium intake to 2,300 mg per day.
On average, Americans consume 3,400 mg or more of sodium a day (equivalent to 1.5 teaspoons of salt), despite efforts over the past few decades to reduce sodium consumption, according to the IOM report, which was sponsored by the Centers for Disease Control and Prevention.
"We found no consistent evidence to support an association between sodium intake and either a beneficial or adverse effect on most direct health outcomes," said Dr. Brian L. Strom, George S. Pepper Professor of Public Health and Preventive Medicine at the University of Pennsylvania, Philadelphia, who chaired the committee that released the report.
The committee did not suggest an optimal target range for sodium consumption.
Dr. Strom and his colleagues reviewed medical studies from 2003 to 2012 that evaluated the direct impact of sodium intake on outcomes such as heart disease and death. Although the studies varied widely in the quality of their methodology and data collection, the report’s authors came to the following conclusions:
• There was a positive relationship between higher levels of sodium and risk of heart disease, consistent with previous research looking at sodium’s effects on blood pressure.
• There were insufficient data to determine if lowering sodium intake below 2,300 mg/day conferred any increase or decrease in the risk of heart disease, stroke, or death.
• Sodium intake of 1,840 mg/day or less may increase the risk of adverse health effects such as cardiovascular events or death among people with mid- to late-stage heart failure receiving aggressive treatment.
• Health outcomes studies provide little evidence about the effects of low sodium intake among those with diabetes, kidney disease, and heart disease, suggesting that people affected by these conditions could follow recommendations for the general public.
But the recommendations prompted calls for caution by some health experts.
The IOM report included "weak studies with numerous problems" and did not include evidence on the effects of sodium reduction on blood pressure, "a key determinant of health and the largest determinant of preventable mortality worldwide," Dr. Lawrence Appel, director of the Welch Center for Prevention, Epidemiology, and Clinical Research at Johns Hopkins University, Baltimore, said in an interview.
Dr. Appel said he still supports the recommendations of the Dietary Guidelines, which he helped create. "Middle- and older-aged adults, African Americans, and patients with hypertension, diabetes, and chronic kidney disease are those at greatest risk for blood pressure–related heart disease and stroke," he said, and they stand to benefit from keeping sodium levels to 1,500 mg/day or less.
Americans should lower sodium consumption to optimize their health – but not below levels of 2,300 mg per day, according to a new report from the Institute of Medicine.
People aged 51 years and older, African-Americans, and those with hypertension, diabetes, or chronic kidney disease can follow the same advice, according to authors of the report, released during a webinar May 14.
That recommendation is a departure from the current Dietary Guidelines for Americans from the U.S. Department of Health and Human Services, which recommends that these higher-risk groups limit sodium intake to 1,500 mg daily. The HHS guidelines recommend that most other people aged 14-50 years limit sodium intake to 2,300 mg per day.
On average, Americans consume 3,400 mg or more of sodium a day (equivalent to 1.5 teaspoons of salt), despite efforts over the past few decades to reduce sodium consumption, according to the IOM report, which was sponsored by the Centers for Disease Control and Prevention.
"We found no consistent evidence to support an association between sodium intake and either a beneficial or adverse effect on most direct health outcomes," said Dr. Brian L. Strom, George S. Pepper Professor of Public Health and Preventive Medicine at the University of Pennsylvania, Philadelphia, who chaired the committee that released the report.
The committee did not suggest an optimal target range for sodium consumption.
Dr. Strom and his colleagues reviewed medical studies from 2003 to 2012 that evaluated the direct impact of sodium intake on outcomes such as heart disease and death. Although the studies varied widely in the quality of their methodology and data collection, the report’s authors came to the following conclusions:
• There was a positive relationship between higher levels of sodium and risk of heart disease, consistent with previous research looking at sodium’s effects on blood pressure.
• There were insufficient data to determine if lowering sodium intake below 2,300 mg/day conferred any increase or decrease in the risk of heart disease, stroke, or death.
• Sodium intake of 1,840 mg/day or less may increase the risk of adverse health effects such as cardiovascular events or death among people with mid- to late-stage heart failure receiving aggressive treatment.
• Health outcomes studies provide little evidence about the effects of low sodium intake among those with diabetes, kidney disease, and heart disease, suggesting that people affected by these conditions could follow recommendations for the general public.
But the recommendations prompted calls for caution by some health experts.
The IOM report included "weak studies with numerous problems" and did not include evidence on the effects of sodium reduction on blood pressure, "a key determinant of health and the largest determinant of preventable mortality worldwide," Dr. Lawrence Appel, director of the Welch Center for Prevention, Epidemiology, and Clinical Research at Johns Hopkins University, Baltimore, said in an interview.
Dr. Appel said he still supports the recommendations of the Dietary Guidelines, which he helped create. "Middle- and older-aged adults, African Americans, and patients with hypertension, diabetes, and chronic kidney disease are those at greatest risk for blood pressure–related heart disease and stroke," he said, and they stand to benefit from keeping sodium levels to 1,500 mg/day or less.
Americans should lower sodium consumption to optimize their health – but not below levels of 2,300 mg per day, according to a new report from the Institute of Medicine.
People aged 51 years and older, African-Americans, and those with hypertension, diabetes, or chronic kidney disease can follow the same advice, according to authors of the report, released during a webinar May 14.
That recommendation is a departure from the current Dietary Guidelines for Americans from the U.S. Department of Health and Human Services, which recommends that these higher-risk groups limit sodium intake to 1,500 mg daily. The HHS guidelines recommend that most other people aged 14-50 years limit sodium intake to 2,300 mg per day.
On average, Americans consume 3,400 mg or more of sodium a day (equivalent to 1.5 teaspoons of salt), despite efforts over the past few decades to reduce sodium consumption, according to the IOM report, which was sponsored by the Centers for Disease Control and Prevention.
"We found no consistent evidence to support an association between sodium intake and either a beneficial or adverse effect on most direct health outcomes," said Dr. Brian L. Strom, George S. Pepper Professor of Public Health and Preventive Medicine at the University of Pennsylvania, Philadelphia, who chaired the committee that released the report.
The committee did not suggest an optimal target range for sodium consumption.
Dr. Strom and his colleagues reviewed medical studies from 2003 to 2012 that evaluated the direct impact of sodium intake on outcomes such as heart disease and death. Although the studies varied widely in the quality of their methodology and data collection, the report’s authors came to the following conclusions:
• There was a positive relationship between higher levels of sodium and risk of heart disease, consistent with previous research looking at sodium’s effects on blood pressure.
• There were insufficient data to determine if lowering sodium intake below 2,300 mg/day conferred any increase or decrease in the risk of heart disease, stroke, or death.
• Sodium intake of 1,840 mg/day or less may increase the risk of adverse health effects such as cardiovascular events or death among people with mid- to late-stage heart failure receiving aggressive treatment.
• Health outcomes studies provide little evidence about the effects of low sodium intake among those with diabetes, kidney disease, and heart disease, suggesting that people affected by these conditions could follow recommendations for the general public.
But the recommendations prompted calls for caution by some health experts.
The IOM report included "weak studies with numerous problems" and did not include evidence on the effects of sodium reduction on blood pressure, "a key determinant of health and the largest determinant of preventable mortality worldwide," Dr. Lawrence Appel, director of the Welch Center for Prevention, Epidemiology, and Clinical Research at Johns Hopkins University, Baltimore, said in an interview.
Dr. Appel said he still supports the recommendations of the Dietary Guidelines, which he helped create. "Middle- and older-aged adults, African Americans, and patients with hypertension, diabetes, and chronic kidney disease are those at greatest risk for blood pressure–related heart disease and stroke," he said, and they stand to benefit from keeping sodium levels to 1,500 mg/day or less.
Erectile dysfunction: 75% with diagnosis go untreated
SAN DIEGO – Of men given an ICD-9 diagnosis of erectile dysfunction, 25% receive any treatment for the condition.
Treatment frequency was higher in men who had low levels of testosterone (51% treated) and lower in those who had prostate cancer (15% treated). Treatment frequency did not vary significantly with other associated comorbidities, Dr. Brian T. Helfand said in a press briefing at the annual meeting of the American Urological Association.
Dr. Helfand of the department of urology at NorthShore University Health System in Evanston, Ill., and his associates used an IMS data set to identify 6,228,509 men over the age of 30 years who received an ICD-9 diagnosis of erectile dysfunction (ED) during a 12-month period that ended in July 2011. IMS is a large insurance claims data set that encompasses more than 80% of prescription data in the United States.
Men were classified as treated if they filled a prescription for a phosphodiesterase type-5 (PDE5) inhibitor, injection or urethral prostaglandins, or androgen replacement therapy. They were classified as untreated if they received an ED diagnosis but did not fill a prescription in the study period. The researchers monitored the therapies by prescription frequency, age, comorbidities, and physician specialty.
Among the 25% of men who filled prescriptions, PDE5 was the most commonly prescribed medication (75%), followed by androgen replacement therapy (31%). Fewer than 2% of patients used any prostaglandin therapy. "The men who were in the oldest age groups were the least likely to fill a prescription," Dr. Helfand said.
The greatest proportion of prescriptions overall were ordered by primary care physicians (28%), followed by endocrinologists (27%), and urologists (21%). The remaining 24% were ordered by various other clinicians.
Dr. Helfand said limitations of the study include the exclusion of Medicare data plus lack of information about the severity of ED, efficacy of treatments, and adherence to long-term therapy.
Dr. Ajay Nangia, who is an associate professor of urology at the University of Kansas Medical Center and the moderator of the press briefing, noted that ED is a medical disease that is often a portent of other disorders. Recognizing that men with ED go untreated may mean that they’re also possibly underinvestigated for associated condition such as diabetes, lipid disorders, and risk factors such as smoking.
Dr. Helfand said that he had no relevant financial conflicts to disclose. One of the study authors, Dr. Kevin McVary, disclosed consultant, advisory, or other roles with several companies, including Allergan, Lilly, NxThera, Watson, NeoTract, and GSK.
SAN DIEGO – Of men given an ICD-9 diagnosis of erectile dysfunction, 25% receive any treatment for the condition.
Treatment frequency was higher in men who had low levels of testosterone (51% treated) and lower in those who had prostate cancer (15% treated). Treatment frequency did not vary significantly with other associated comorbidities, Dr. Brian T. Helfand said in a press briefing at the annual meeting of the American Urological Association.
Dr. Helfand of the department of urology at NorthShore University Health System in Evanston, Ill., and his associates used an IMS data set to identify 6,228,509 men over the age of 30 years who received an ICD-9 diagnosis of erectile dysfunction (ED) during a 12-month period that ended in July 2011. IMS is a large insurance claims data set that encompasses more than 80% of prescription data in the United States.
Men were classified as treated if they filled a prescription for a phosphodiesterase type-5 (PDE5) inhibitor, injection or urethral prostaglandins, or androgen replacement therapy. They were classified as untreated if they received an ED diagnosis but did not fill a prescription in the study period. The researchers monitored the therapies by prescription frequency, age, comorbidities, and physician specialty.
Among the 25% of men who filled prescriptions, PDE5 was the most commonly prescribed medication (75%), followed by androgen replacement therapy (31%). Fewer than 2% of patients used any prostaglandin therapy. "The men who were in the oldest age groups were the least likely to fill a prescription," Dr. Helfand said.
The greatest proportion of prescriptions overall were ordered by primary care physicians (28%), followed by endocrinologists (27%), and urologists (21%). The remaining 24% were ordered by various other clinicians.
Dr. Helfand said limitations of the study include the exclusion of Medicare data plus lack of information about the severity of ED, efficacy of treatments, and adherence to long-term therapy.
Dr. Ajay Nangia, who is an associate professor of urology at the University of Kansas Medical Center and the moderator of the press briefing, noted that ED is a medical disease that is often a portent of other disorders. Recognizing that men with ED go untreated may mean that they’re also possibly underinvestigated for associated condition such as diabetes, lipid disorders, and risk factors such as smoking.
Dr. Helfand said that he had no relevant financial conflicts to disclose. One of the study authors, Dr. Kevin McVary, disclosed consultant, advisory, or other roles with several companies, including Allergan, Lilly, NxThera, Watson, NeoTract, and GSK.
SAN DIEGO – Of men given an ICD-9 diagnosis of erectile dysfunction, 25% receive any treatment for the condition.
Treatment frequency was higher in men who had low levels of testosterone (51% treated) and lower in those who had prostate cancer (15% treated). Treatment frequency did not vary significantly with other associated comorbidities, Dr. Brian T. Helfand said in a press briefing at the annual meeting of the American Urological Association.
Dr. Helfand of the department of urology at NorthShore University Health System in Evanston, Ill., and his associates used an IMS data set to identify 6,228,509 men over the age of 30 years who received an ICD-9 diagnosis of erectile dysfunction (ED) during a 12-month period that ended in July 2011. IMS is a large insurance claims data set that encompasses more than 80% of prescription data in the United States.
Men were classified as treated if they filled a prescription for a phosphodiesterase type-5 (PDE5) inhibitor, injection or urethral prostaglandins, or androgen replacement therapy. They were classified as untreated if they received an ED diagnosis but did not fill a prescription in the study period. The researchers monitored the therapies by prescription frequency, age, comorbidities, and physician specialty.
Among the 25% of men who filled prescriptions, PDE5 was the most commonly prescribed medication (75%), followed by androgen replacement therapy (31%). Fewer than 2% of patients used any prostaglandin therapy. "The men who were in the oldest age groups were the least likely to fill a prescription," Dr. Helfand said.
The greatest proportion of prescriptions overall were ordered by primary care physicians (28%), followed by endocrinologists (27%), and urologists (21%). The remaining 24% were ordered by various other clinicians.
Dr. Helfand said limitations of the study include the exclusion of Medicare data plus lack of information about the severity of ED, efficacy of treatments, and adherence to long-term therapy.
Dr. Ajay Nangia, who is an associate professor of urology at the University of Kansas Medical Center and the moderator of the press briefing, noted that ED is a medical disease that is often a portent of other disorders. Recognizing that men with ED go untreated may mean that they’re also possibly underinvestigated for associated condition such as diabetes, lipid disorders, and risk factors such as smoking.
Dr. Helfand said that he had no relevant financial conflicts to disclose. One of the study authors, Dr. Kevin McVary, disclosed consultant, advisory, or other roles with several companies, including Allergan, Lilly, NxThera, Watson, NeoTract, and GSK.
AT THE AUA ANNUAL MEETING
Major finding: Among men over the age of 30 who received a diagnosis of erectile dysfunction, 25% were treated.
Data source: A study of 6,228,509 men from the IMS Health data set who received an ICD-9 diagnosis of erectile dysfunction.
Disclosures: Dr. Helfand said that he had no relevant financial conflicts to disclose. One of the study authors, Dr. Kevin McVary, disclosed consultant, advisory, or other roles with several companies, including Allergan, Lilly, NxThera, Watson, NeoTract, and GSK. He also has served as an investigator for the National Institute of Diabetes and Digestive and Kidney Diseases.
Forecast warns of urologist shortage
SAN DIEGO – The number of urologists practicing in the United States is expected to decrease by 29% between 2009 and 2025, according to a new analysis.
"It’s one thing if the demand for urologists is going up and the supply is stable, but to have the demand go up and the supply almost falling off of a cliff is worrisome," Dr. Raj S. Pruthi said in an interview at the annual meeting of the American Urological Association. "The people who will be hardest hit by this are ones who already struggle with access: those who live in rural communities."
Dr. Pruthi and his colleagues used stock and flow models, starting with the supply of urologists in 2009. They added new entrants from the graduate medical education (GME) pipeline and subtracted attrition from training and from the workforce due to retirement or breaks from practice. The forecast model estimates a 29% head count reduction and a 25% decrease in the full-time equivalent (FTE) supply of urologists between 2009 and 2025. The projected decrease is more than four times greater than the Health Resources and Services Administration’s Physician Supply Model, which estimated a 7% decrease over the same time period.
Dr. Pruthi warned that none of the proposed changes to GME (recommendations from the Council of Graduate Medical Education’s 16th report or a recent proposal to Congress) will increase GME enough to offset the projected decline in head count. "GME funding has been capped since 1996," he said. "We’re setting forth a recipe for a very big problem that we’re going to have for future generations in terms of who’s going to take care of" a rapidly aging population.
As the Affordable Health Care Act takes shape, "one thing that’s not been considered adequately is physician supply," added Dr. Pruthi, chief of urologic surgery at the University of North Carolina at Chapel Hill. "Are there enough of us to help care for the population? That needs to be part of the calculus. We need to do efficient, appropriate care. We need to cut health care costs, but we have to remember our physician supply."
The shrinking number of urologists could affect mortality rates, as research has demonstrated an association between a higher density of urologists in a defined area and lower mortality from prostate, bladder, and kidney cancer. "As supply contracts, rural areas are likely to have an even greater loss of urologic surgeons since these areas have a higher percentage of surgeons closer to retirement age than urban areas," the researchers noted in their abstract. "The result may decrease access to screening, medical and surgical treatment for urologic conditions."
Dr. Pruthi acknowledged that the ability to predict physician demand is an imprecise science. However, "there is indirect data to suggest that our demand isn’t going to go away. It’s only going to go up with that rising incidence and with the rising number of aging baby boomers. Second, we don’t know the appropriate ratio of supply and demand. If you have limited supply, you have limited access. Is our culture accepting of that? Some of these limitations to access may have health consequences."
He and his associates plan to conduct more detailed work on the projection model, including the impact of increasing numbers of women entering the urology field. "About 5% of urologists are female, but in current residency matching the numbers are about 25% female," he said. "What impact will that have? We don’t know yet."
Dr. Pruthi said that he had no relevant financial conflicts to disclose.
SAN DIEGO – The number of urologists practicing in the United States is expected to decrease by 29% between 2009 and 2025, according to a new analysis.
"It’s one thing if the demand for urologists is going up and the supply is stable, but to have the demand go up and the supply almost falling off of a cliff is worrisome," Dr. Raj S. Pruthi said in an interview at the annual meeting of the American Urological Association. "The people who will be hardest hit by this are ones who already struggle with access: those who live in rural communities."
Dr. Pruthi and his colleagues used stock and flow models, starting with the supply of urologists in 2009. They added new entrants from the graduate medical education (GME) pipeline and subtracted attrition from training and from the workforce due to retirement or breaks from practice. The forecast model estimates a 29% head count reduction and a 25% decrease in the full-time equivalent (FTE) supply of urologists between 2009 and 2025. The projected decrease is more than four times greater than the Health Resources and Services Administration’s Physician Supply Model, which estimated a 7% decrease over the same time period.
Dr. Pruthi warned that none of the proposed changes to GME (recommendations from the Council of Graduate Medical Education’s 16th report or a recent proposal to Congress) will increase GME enough to offset the projected decline in head count. "GME funding has been capped since 1996," he said. "We’re setting forth a recipe for a very big problem that we’re going to have for future generations in terms of who’s going to take care of" a rapidly aging population.
As the Affordable Health Care Act takes shape, "one thing that’s not been considered adequately is physician supply," added Dr. Pruthi, chief of urologic surgery at the University of North Carolina at Chapel Hill. "Are there enough of us to help care for the population? That needs to be part of the calculus. We need to do efficient, appropriate care. We need to cut health care costs, but we have to remember our physician supply."
The shrinking number of urologists could affect mortality rates, as research has demonstrated an association between a higher density of urologists in a defined area and lower mortality from prostate, bladder, and kidney cancer. "As supply contracts, rural areas are likely to have an even greater loss of urologic surgeons since these areas have a higher percentage of surgeons closer to retirement age than urban areas," the researchers noted in their abstract. "The result may decrease access to screening, medical and surgical treatment for urologic conditions."
Dr. Pruthi acknowledged that the ability to predict physician demand is an imprecise science. However, "there is indirect data to suggest that our demand isn’t going to go away. It’s only going to go up with that rising incidence and with the rising number of aging baby boomers. Second, we don’t know the appropriate ratio of supply and demand. If you have limited supply, you have limited access. Is our culture accepting of that? Some of these limitations to access may have health consequences."
He and his associates plan to conduct more detailed work on the projection model, including the impact of increasing numbers of women entering the urology field. "About 5% of urologists are female, but in current residency matching the numbers are about 25% female," he said. "What impact will that have? We don’t know yet."
Dr. Pruthi said that he had no relevant financial conflicts to disclose.
SAN DIEGO – The number of urologists practicing in the United States is expected to decrease by 29% between 2009 and 2025, according to a new analysis.
"It’s one thing if the demand for urologists is going up and the supply is stable, but to have the demand go up and the supply almost falling off of a cliff is worrisome," Dr. Raj S. Pruthi said in an interview at the annual meeting of the American Urological Association. "The people who will be hardest hit by this are ones who already struggle with access: those who live in rural communities."
Dr. Pruthi and his colleagues used stock and flow models, starting with the supply of urologists in 2009. They added new entrants from the graduate medical education (GME) pipeline and subtracted attrition from training and from the workforce due to retirement or breaks from practice. The forecast model estimates a 29% head count reduction and a 25% decrease in the full-time equivalent (FTE) supply of urologists between 2009 and 2025. The projected decrease is more than four times greater than the Health Resources and Services Administration’s Physician Supply Model, which estimated a 7% decrease over the same time period.
Dr. Pruthi warned that none of the proposed changes to GME (recommendations from the Council of Graduate Medical Education’s 16th report or a recent proposal to Congress) will increase GME enough to offset the projected decline in head count. "GME funding has been capped since 1996," he said. "We’re setting forth a recipe for a very big problem that we’re going to have for future generations in terms of who’s going to take care of" a rapidly aging population.
As the Affordable Health Care Act takes shape, "one thing that’s not been considered adequately is physician supply," added Dr. Pruthi, chief of urologic surgery at the University of North Carolina at Chapel Hill. "Are there enough of us to help care for the population? That needs to be part of the calculus. We need to do efficient, appropriate care. We need to cut health care costs, but we have to remember our physician supply."
The shrinking number of urologists could affect mortality rates, as research has demonstrated an association between a higher density of urologists in a defined area and lower mortality from prostate, bladder, and kidney cancer. "As supply contracts, rural areas are likely to have an even greater loss of urologic surgeons since these areas have a higher percentage of surgeons closer to retirement age than urban areas," the researchers noted in their abstract. "The result may decrease access to screening, medical and surgical treatment for urologic conditions."
Dr. Pruthi acknowledged that the ability to predict physician demand is an imprecise science. However, "there is indirect data to suggest that our demand isn’t going to go away. It’s only going to go up with that rising incidence and with the rising number of aging baby boomers. Second, we don’t know the appropriate ratio of supply and demand. If you have limited supply, you have limited access. Is our culture accepting of that? Some of these limitations to access may have health consequences."
He and his associates plan to conduct more detailed work on the projection model, including the impact of increasing numbers of women entering the urology field. "About 5% of urologists are female, but in current residency matching the numbers are about 25% female," he said. "What impact will that have? We don’t know yet."
Dr. Pruthi said that he had no relevant financial conflicts to disclose.
AT THE AUA ANNUAL MEETING
Major finding: The forecast model estimates a 29% head count reduction and a 25% decrease in the full-time equivalent (FTE) supply of urologists between 2009 and 2025.
Data source: An analysis that used stock and flow models, starting with the supply of urologists in 2009.
Disclosures: Dr. Pruthi said that he had no relevant disclosures.
Study IDs risks for bladder test failure following sling procedure
CHARLESTON, S.C. – Most women who undergo an isolated midurethral sling procedure pass active bladder testing on the first attempt, but the likelihood of being discharged with an indwelling catheter increases as bladder capacity increases, maximum flow rate decreases, or detrusor overactivity occurs, findings from a retrospective cross-sectional study have shown.
Of 112 patients who underwent an isolated midurethral sling procedure at a single site, 90 (80.4%) passed active bladder testing (ABT) on the first attempt prior to discharge home, and 22 (19.6%) failed, Dr. Meadow M. Good reported at the annual meeting of the Society of Gynecologic Surgeons.
Bladder testing involved filling the bladder with sterile fluid to 300 mL or to patient discomfort, whichever came first. Passing the test required that two-thirds of the instilled volume was voided.
Average bladder capacity among those who failed ABT was 415 cc, compared with 381 cc in those who passed; average maximum flow rate in those who failed ABT was 15.6 cc/second, compared with 21.6 cc/second in those who passed; and detrusor overactivity was observed in 41% of those who failed ABT, compared with 20% of those who passed, said Dr. Good of the University of Texas Southwestern Medical Center, Dallas.
Furthermore, the capacity-to-infused volume ratio was 1:5 for those who failed ABT and 1:3 for those who passed. Most (81.8%) of those who failed had bladder capacity greater than the infused amount, but no significant difference in the capacity-to-infused volume ratios was seen in the group who passed ABT.
On multivariate analysis, every 50-cc increase in bladder capacity significantly increased the odds of failing ABT (odds ratio, 1.25); every unit increase in the maximum flow rate significantly decreased the odds of ABT failure (OR, 0.91); and the presence of detrusor overactivity was associated with a fivefold increase in the odds of failure (OR, 5.0).
Age, race, body mass index, maximum urethral closure pressure, and maximum detrusor pressure during pressure-flow studies were not found to be associated with ABT outcomes.
Patients included in this study were all those who underwent an isolated midurethral sling procedure at the medical center between January 2011 and August 2012, excluding those who had intraoperative complications requiring discharge with a Foley catheter.
The findings are important because the rates of postoperative urinary retention following midurethral sling procedures are highly variable, and the identification of factors associated with ABT failure could help improve outcomes.
"Patients with postoperative retention are generally discharged home with an indwelling catheter or intermittent self-catheterization; catheter-associated bacteria lead to increased urinary tract infections and health care costs, and to decreased quality of life," Dr. Good said, adding that reducing the rates of postoperative catheter use should be a priority.
"While further research is needed, filling patients to cystometric capacity may improve the success of postoperative bladder testing," she said.
Dr. Good reported having no relevant financial disclosures.
CHARLESTON, S.C. – Most women who undergo an isolated midurethral sling procedure pass active bladder testing on the first attempt, but the likelihood of being discharged with an indwelling catheter increases as bladder capacity increases, maximum flow rate decreases, or detrusor overactivity occurs, findings from a retrospective cross-sectional study have shown.
Of 112 patients who underwent an isolated midurethral sling procedure at a single site, 90 (80.4%) passed active bladder testing (ABT) on the first attempt prior to discharge home, and 22 (19.6%) failed, Dr. Meadow M. Good reported at the annual meeting of the Society of Gynecologic Surgeons.
Bladder testing involved filling the bladder with sterile fluid to 300 mL or to patient discomfort, whichever came first. Passing the test required that two-thirds of the instilled volume was voided.
Average bladder capacity among those who failed ABT was 415 cc, compared with 381 cc in those who passed; average maximum flow rate in those who failed ABT was 15.6 cc/second, compared with 21.6 cc/second in those who passed; and detrusor overactivity was observed in 41% of those who failed ABT, compared with 20% of those who passed, said Dr. Good of the University of Texas Southwestern Medical Center, Dallas.
Furthermore, the capacity-to-infused volume ratio was 1:5 for those who failed ABT and 1:3 for those who passed. Most (81.8%) of those who failed had bladder capacity greater than the infused amount, but no significant difference in the capacity-to-infused volume ratios was seen in the group who passed ABT.
On multivariate analysis, every 50-cc increase in bladder capacity significantly increased the odds of failing ABT (odds ratio, 1.25); every unit increase in the maximum flow rate significantly decreased the odds of ABT failure (OR, 0.91); and the presence of detrusor overactivity was associated with a fivefold increase in the odds of failure (OR, 5.0).
Age, race, body mass index, maximum urethral closure pressure, and maximum detrusor pressure during pressure-flow studies were not found to be associated with ABT outcomes.
Patients included in this study were all those who underwent an isolated midurethral sling procedure at the medical center between January 2011 and August 2012, excluding those who had intraoperative complications requiring discharge with a Foley catheter.
The findings are important because the rates of postoperative urinary retention following midurethral sling procedures are highly variable, and the identification of factors associated with ABT failure could help improve outcomes.
"Patients with postoperative retention are generally discharged home with an indwelling catheter or intermittent self-catheterization; catheter-associated bacteria lead to increased urinary tract infections and health care costs, and to decreased quality of life," Dr. Good said, adding that reducing the rates of postoperative catheter use should be a priority.
"While further research is needed, filling patients to cystometric capacity may improve the success of postoperative bladder testing," she said.
Dr. Good reported having no relevant financial disclosures.
CHARLESTON, S.C. – Most women who undergo an isolated midurethral sling procedure pass active bladder testing on the first attempt, but the likelihood of being discharged with an indwelling catheter increases as bladder capacity increases, maximum flow rate decreases, or detrusor overactivity occurs, findings from a retrospective cross-sectional study have shown.
Of 112 patients who underwent an isolated midurethral sling procedure at a single site, 90 (80.4%) passed active bladder testing (ABT) on the first attempt prior to discharge home, and 22 (19.6%) failed, Dr. Meadow M. Good reported at the annual meeting of the Society of Gynecologic Surgeons.
Bladder testing involved filling the bladder with sterile fluid to 300 mL or to patient discomfort, whichever came first. Passing the test required that two-thirds of the instilled volume was voided.
Average bladder capacity among those who failed ABT was 415 cc, compared with 381 cc in those who passed; average maximum flow rate in those who failed ABT was 15.6 cc/second, compared with 21.6 cc/second in those who passed; and detrusor overactivity was observed in 41% of those who failed ABT, compared with 20% of those who passed, said Dr. Good of the University of Texas Southwestern Medical Center, Dallas.
Furthermore, the capacity-to-infused volume ratio was 1:5 for those who failed ABT and 1:3 for those who passed. Most (81.8%) of those who failed had bladder capacity greater than the infused amount, but no significant difference in the capacity-to-infused volume ratios was seen in the group who passed ABT.
On multivariate analysis, every 50-cc increase in bladder capacity significantly increased the odds of failing ABT (odds ratio, 1.25); every unit increase in the maximum flow rate significantly decreased the odds of ABT failure (OR, 0.91); and the presence of detrusor overactivity was associated with a fivefold increase in the odds of failure (OR, 5.0).
Age, race, body mass index, maximum urethral closure pressure, and maximum detrusor pressure during pressure-flow studies were not found to be associated with ABT outcomes.
Patients included in this study were all those who underwent an isolated midurethral sling procedure at the medical center between January 2011 and August 2012, excluding those who had intraoperative complications requiring discharge with a Foley catheter.
The findings are important because the rates of postoperative urinary retention following midurethral sling procedures are highly variable, and the identification of factors associated with ABT failure could help improve outcomes.
"Patients with postoperative retention are generally discharged home with an indwelling catheter or intermittent self-catheterization; catheter-associated bacteria lead to increased urinary tract infections and health care costs, and to decreased quality of life," Dr. Good said, adding that reducing the rates of postoperative catheter use should be a priority.
"While further research is needed, filling patients to cystometric capacity may improve the success of postoperative bladder testing," she said.
Dr. Good reported having no relevant financial disclosures.
AT THE SGS ANNUAL MEETING
Major finding: Every 50-cc increase in bladder capacity significantly increased the odds of failing active bladder testing (OR, 1.25); every unit increase in the maximum flow rate significantly decreased the odds of ABT failure (OR, 0.91); and the presence of detrusor overactivity was associated with a fivefold increase in the odds of failure (OR, 5.0).
Data source: A single-site, retrospective cross-sectional study involving 112 patients.
Disclosures: Dr. Good reported having no relevant financial disclosures.
High selenium exposure may lower incidence of advanced prostate cancer
Increased exposure to selenium was associated with a significantly reduced risk of advanced prostate cancer, according to the results of a large, prospective cohort study of men in the Netherlands.
The results of the study, conducted in men with low to moderate selenium levels, "suggest that selenium may prevent advanced, clinically relevant, prostate cancer," noted Milan Geybels, M.Sc., at the annual meeting of the American Association for Cancer Research. And since there is little evidence on risk factors that can modify prostate cancer risk, "any compound that would prevent the incidence of advanced, clinically relevant prostate cancer would have a substantial impact on public health," Mr. Geybels said in an interview.
The men were enrolled in the Netherlands Cohort Study, a study of diet and cancer in 58,279 men between the ages of 55 and 69 years at entry. Over 17 years, 898 men were diagnosed with advanced prostate cancer and were compared with 1,203 men also in the study. Using the concentration of selenium in the men’s toenails, an indication of long-term exposure, the investigators determined that the risk of advanced prostate cancer was significantly reduced as selenium levels increased. The risk for advanced prostate cancer was more than 60% lower in men with the highest levels of toenail selenium, when compared with those with the lowest levels, and the association was "more pronounced" among the men diagnosed later in the follow-up period, noted Mr. Geybels, a doctoral candidate in cancer epidemiology at Maastricht University, the Netherlands.
Previous prospective studies on the possible link between selenium and prostate cancer risk have produced conflicting results. Many of the studies included men with moderate to high selenium levels. Also, they did not exclusively study advanced prostate cancer, and "almost exclusively" used blood levels of selenium, which reflect recent exposure, he noted.
SELECT (Selenium and Vitamin E Cancer Prevention Trial) found no relationship between selenium supplementation and prostate cancer risk. However, "a possible explanation for the null finding is that selenium intake among SELECT participants was adequate ... [so], further selenium supplementation had no effect on prostate cancer incidence," he said.
The intake of selenium varies worldwide as a result of variations in soil content and the related variability in the selenium content in foods; in the Netherlands, low selenium is common.
The results need to be replicated in more prospective studies, and if confirmed, "A prevention trial of selenium and advanced prostate cancer in a low selenium population may be justified," said Mr. Geybels. Whether levels higher than those detected in the men in the study are associated with a further reduction in risk is unknown.
He and his coauthors had no disclosures to report.
Increased exposure to selenium was associated with a significantly reduced risk of advanced prostate cancer, according to the results of a large, prospective cohort study of men in the Netherlands.
The results of the study, conducted in men with low to moderate selenium levels, "suggest that selenium may prevent advanced, clinically relevant, prostate cancer," noted Milan Geybels, M.Sc., at the annual meeting of the American Association for Cancer Research. And since there is little evidence on risk factors that can modify prostate cancer risk, "any compound that would prevent the incidence of advanced, clinically relevant prostate cancer would have a substantial impact on public health," Mr. Geybels said in an interview.
The men were enrolled in the Netherlands Cohort Study, a study of diet and cancer in 58,279 men between the ages of 55 and 69 years at entry. Over 17 years, 898 men were diagnosed with advanced prostate cancer and were compared with 1,203 men also in the study. Using the concentration of selenium in the men’s toenails, an indication of long-term exposure, the investigators determined that the risk of advanced prostate cancer was significantly reduced as selenium levels increased. The risk for advanced prostate cancer was more than 60% lower in men with the highest levels of toenail selenium, when compared with those with the lowest levels, and the association was "more pronounced" among the men diagnosed later in the follow-up period, noted Mr. Geybels, a doctoral candidate in cancer epidemiology at Maastricht University, the Netherlands.
Previous prospective studies on the possible link between selenium and prostate cancer risk have produced conflicting results. Many of the studies included men with moderate to high selenium levels. Also, they did not exclusively study advanced prostate cancer, and "almost exclusively" used blood levels of selenium, which reflect recent exposure, he noted.
SELECT (Selenium and Vitamin E Cancer Prevention Trial) found no relationship between selenium supplementation and prostate cancer risk. However, "a possible explanation for the null finding is that selenium intake among SELECT participants was adequate ... [so], further selenium supplementation had no effect on prostate cancer incidence," he said.
The intake of selenium varies worldwide as a result of variations in soil content and the related variability in the selenium content in foods; in the Netherlands, low selenium is common.
The results need to be replicated in more prospective studies, and if confirmed, "A prevention trial of selenium and advanced prostate cancer in a low selenium population may be justified," said Mr. Geybels. Whether levels higher than those detected in the men in the study are associated with a further reduction in risk is unknown.
He and his coauthors had no disclosures to report.
Increased exposure to selenium was associated with a significantly reduced risk of advanced prostate cancer, according to the results of a large, prospective cohort study of men in the Netherlands.
The results of the study, conducted in men with low to moderate selenium levels, "suggest that selenium may prevent advanced, clinically relevant, prostate cancer," noted Milan Geybels, M.Sc., at the annual meeting of the American Association for Cancer Research. And since there is little evidence on risk factors that can modify prostate cancer risk, "any compound that would prevent the incidence of advanced, clinically relevant prostate cancer would have a substantial impact on public health," Mr. Geybels said in an interview.
The men were enrolled in the Netherlands Cohort Study, a study of diet and cancer in 58,279 men between the ages of 55 and 69 years at entry. Over 17 years, 898 men were diagnosed with advanced prostate cancer and were compared with 1,203 men also in the study. Using the concentration of selenium in the men’s toenails, an indication of long-term exposure, the investigators determined that the risk of advanced prostate cancer was significantly reduced as selenium levels increased. The risk for advanced prostate cancer was more than 60% lower in men with the highest levels of toenail selenium, when compared with those with the lowest levels, and the association was "more pronounced" among the men diagnosed later in the follow-up period, noted Mr. Geybels, a doctoral candidate in cancer epidemiology at Maastricht University, the Netherlands.
Previous prospective studies on the possible link between selenium and prostate cancer risk have produced conflicting results. Many of the studies included men with moderate to high selenium levels. Also, they did not exclusively study advanced prostate cancer, and "almost exclusively" used blood levels of selenium, which reflect recent exposure, he noted.
SELECT (Selenium and Vitamin E Cancer Prevention Trial) found no relationship between selenium supplementation and prostate cancer risk. However, "a possible explanation for the null finding is that selenium intake among SELECT participants was adequate ... [so], further selenium supplementation had no effect on prostate cancer incidence," he said.
The intake of selenium varies worldwide as a result of variations in soil content and the related variability in the selenium content in foods; in the Netherlands, low selenium is common.
The results need to be replicated in more prospective studies, and if confirmed, "A prevention trial of selenium and advanced prostate cancer in a low selenium population may be justified," said Mr. Geybels. Whether levels higher than those detected in the men in the study are associated with a further reduction in risk is unknown.
He and his coauthors had no disclosures to report.
FROM THE ANNUAL AACR MEETING
Major finding: The risk for advanced prostate cancer was more than 60% lower in men with the highest levels of toenail selenium, when compared with those with the lowest levels.
Data source: Men in the Netherlands Cohort Study diagnosed with advanced prostate cancer (898) were compared with 1,203 men also in the study.
Disclosures: Mr. Geybels and his colleagues had no disclosures to report.
Detecting and controlling diabetic nephropathy: What do we know?
Diabetes is on the rise, and so is diabetic nephropathy. In view of this epidemic, physicians should consider strategies to detect and control kidney disease in their diabetic patients.
This article will focus on kidney disease in adult-onset type 2 diabetes. Although it has different pathogenetic mechanisms than type 1 diabetes, the clinical course of the two conditions is very similar in terms of the prevalence of proteinuria after diagnosis, the progression to renal failure after the onset of proteinuria, and treatment options.1
DIABETES AND DIABETIC KIDNEY DISEASE ARE ON THE RISE
The incidence of diabetes increases with age, and with the aging of the baby boomers, its prevalence is growing dramatically. The 2005– 2008 National Health and Nutrition Examination Survey estimated the prevalence as 3.7% in adults age 20 to 44, 13.7% at age 45 to 64, and 26.9% in people age 65 and older. The obesity epidemic is also contributing to the increase in diabetes in all age groups.
Diabetic kidney disease has increased in the United States from about 4 million cases 20 years ago to about 7 million in 2005–2008.2 Diabetes is the major cause of end-stage renal disease in the developed world, accounting for 40% to 50% of cases. Other major causes are hypertension (27%) and glomerulonephritis (13%).3
Physicians in nearly every field of medicine now care for patients with diabetic nephropathy. The classic presentation—a patient who has impaired vision, fluid retention with edema, and hypertension—is commonly seen in dialysis units and ophthalmology and cardiovascular clinics.
CLINICAL PROGRESSION
Early in the course of diabetic nephropathy, blood pressure is normal and microalbuminuria is not evident, but many patients have a high glomerular filtration rate (GFR), indicating temporarily “enhanced” renal function or hyperfiltration. The next stage is characterized by microalbuminuria, correlating with glomerular mesangial expansion: the GFR falls back into the normal range and blood pressure starts to increase. Finally, macroalbuminuria occurs, accompanied by rising blood pressure and a declining GFR, correlating with the histologic appearance of glomerulosclerosis and Kimmelstiel-Wilson nodules.4
Hypertension develops in 5% of patients by 10 years after type 1 diabetes is diagnosed, 33% by 20 years, and 70% by 40 years. In contrast, 40% of patients with type 2 diabetes have high blood pressure at diagnosis.
Unfortunately, in most cases, this progression is a one-way street, so it is critical to intervene to try to slow the progression early in the course of the disease process.
SCREENING FOR DIABETIC NEPHROPATHY
Nephropathy screening guidelines for patients with diabetes are provided in Table 1.5
Blood pressure should be monitored at each office visit (Table 1). The goal for adults with diabetes should be to reduce blood pressure to 130/80 mm Hg. Reduction beyond this level may be associated with an increased mortality rate.6 Very high blood pressure (> 180 mm Hg systolic) should be lowered slowly. Lowering blood pressure delays the progression from microalbuminuria (30–299 mg/day or 20–199 μg/min) to macroalbuminuria (> 300 mg/day or > 200 μg/min) and slows the progression to renal failure.
Urinary albumin. Proteinuria takes 5 to 10 years to develop after the onset of diabetes. Because it is possible for patients with type 2 diabetes to have had the disease for some time before being diagnosed, urinary albumin screening should be performed at diagnosis and annually thereafter. Patients with type 1 are usually diagnosed with diabetes at or near onset of disease; therefore, annual screening for urinary albumin can begin 5 years after diagnosis.5
Proteinuria can be measured in different ways (Table 2). The basic screening test for clinical proteinuria is the urine dipstick, which is very sensitive to albumin and relatively insensitive to other proteins. “Trace-positive” results are common in healthy people, so proteinuria is not confirmed unless a patient has repeatedly positive results.
Microalbuminuria is important to measure, especially if it helps determine therapy. It is not detectable by the urinary dipstick, but can be measured in the following ways:
- Measurement of the albumin-creatinine ratio in a random spot collection
- 24-hour collection (creatinine should simultaneously be measured and creatinine clearance calculated)
- Timed collection (4 hours or overnight).
The first method is preferred, and any positive test result must be confirmed by repeat analyses of urinary albumin before a patient is diagnosed with microalbuminuria.
Occasionally a patient presenting with proteinuria but normal blood sugar and hemoglobin A1c will have a biopsy that reveals morphologic changes of classic diabetic nephropathy. Most such patients have a history of hyperglycemia, indicating that they actually have been diabetic.
Proteinuria—the best marker of disease progression
Proteinuria is the strongest predictor of renal outcomes. The Reduction in End Points in Noninsulin-Dependent Diabetes Mellitus With the Angiotensin II Antagonist Losartan (RENAAL) study was a randomized, placebo-controlled trial in more than 1,500 patients with type 2 diabetes to test the effects of losartan on renal outcome. Those with high albuminuria (> 3.0 g albumin/g creatinine) at baseline were five times more likely to reach a renal end point and were eight times more likely to have progression to end-stage renal disease than patients with low albuminuria (< 1.5 g/g).7 The degree of albuminuria after 6 months of treatment showed similar predictive trends, indicating that monitoring and treating proteinuria are extremely important goals.
STRATEGY 1 TO LIMIT RENAL INJURY: REDUCE BLOOD PRESSURE
Blood pressure control improves renal and cardiovascular function.
As early as 1983, Parving et al,8 in a study of only 10 insulin-dependent diabetic patients, showed strong evidence that early aggressive antihypertensive treatment improved the course of diabetic nephropathy. During the mean pretreatment period of 29 months, the GFR decreased significantly and the urinary albumin excretion rate and arterial blood pressure rose significantly. During the mean 39-month period of antihypertensive treatment with metoprolol, hydralazine, and furosemide or a thiazide, mean arterial blood pressure fell from 144/97 to 128/84 mm Hg and urinary albumin excretion from 977 to 433 μg/ min. The rate of decline in GFR slowed from 0.91 mL/min/month before treatment to 0.39 mL/min/month during treatment.
The Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation (ADVANCE) trial9 enrolled more than 11,000 patients internationally with type 2 diabetes at high risk for cardiovascular events. In addition to standard therapy, blood pressure was intensively controlled in one group with a combination of the angiotensin-converting enzyme (ACE) inhibitor perindopril and the diuretic indapamide. The intensive-therapy group achieved blood pressures less than 140/80 mm Hg and had a mean reduction of systolic blood pressure of 5.6 mm Hg and diastolic blood pressure of 2.2 mm Hg vs controls. Despite these apparently modest reductions, the intensively controlled group had a significant 9% reduction of the primary outcome of combined macrovascular events (cardiovascular death, myocardial infarction, and stroke) and microvascular events (new or worsening nephropathy, or retinopathy).10
A meta-analysis of studies of patients with type 2 diabetes found reduced nephropathy with systolic blood pressure control to less than 130 mm Hg.11
The United Kingdom Prospective Diabetes Study (UKPDS) is a series of studies of diabetes. The original study in 1998 enrolled 5,102 patients with newly diagnosed type 2 diabetes.12 The more than 1,000 patients with hypertension were randomized to either tight blood pressure control or regular care. The intensive treatment group had a mean blood pressure reduction of 9 mm Hg systolic and 3 mm Hg diastolic, along with major reductions in all diabetes end points, diabetes deaths, microvascular disease, and stroke over a median follow-up of 8.4 years.
Continuous blood pressure control is critical
Tight blood pressure control must be maintained to have continued benefit. During the 10 years following the UKPDS, no attempts were made to maintain the previously assigned therapies. A follow-up study13 of 884 UKPDS patients found that blood pressures were the same again between the two groups 2 years after the trial was stopped, and no beneficial legacy effect from previous blood pressure control was evident on end points.
Control below 120 mm Hg systolic not needed
Blood pressure control slows kidney disease and prevents major macrovascular disease, but there is no evidence that lowering systolic blood pressure below 120 mm Hg provides additional benefit. In the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial,14 more than 10,000 patients with type 2 diabetes and existing cardiovascular disease or additional cardiovascular risk factors were randomized to a goal of systolic blood pressure less than 120 mm Hg or less than 140 mm Hg (actual mean systolic pressures were 119 vs 134 mm Hg, respectively). Over nearly 5 years, there was no difference in cardiovascular events or deaths between the two groups.15
Since 1997, six international organizations have revised their recommended blood pressure goals in diabetes mellitus and renal diseases. Randomized clinical trials and observational studies have demonstrated the importance of blood pressure control to the level of 125/75 to 140/80 mm Hg. The National Kidney Foundation, the American Diabetes Association, and the Canadian Hypertension Society have developed consensus guidelines for blood pressure control to less than 130/80 mm Hg.16–21 Table 3 summarizes blood pressure goals for patients with diabetes.
STRATEGY 2: CONTROL BLOOD SUGAR
Recommendations for blood sugar goals are more controversial.
The Diabetes Control and Complications Trial22 provided early evidence that tight blood sugar control slows the development of microalbuminuria and macroalbuminuria. The study randomized more than 1,400 patients with type 1 diabetes to either standard therapy (1 or 2 daily insulin injections) or intensive therapy (an external insulin pump or 3 or more insulin injections guided by frequent blood glucose monitoring) to keep blood glucose levels close to normal. About half the patients had mild retinopathy at baseline and the others had no retinopathy. After 6.5 years, intensive therapy was found to significantly delay the onset and slow the progression of diabetic retinopathy and nephropathy.
The Kumamoto Study23 randomized 110 patients with type 2 diabetes and either no retinopathy (primary prevention cohort) or simple retinopathy (secondary prevention cohort) to receive either multiple insulin injections or conventional insulin therapy over 8 years. Intensive therapy led to lower rates of retinopathy (7.7% vs 32% in primary prevention and 19% vs 44% in secondary prevention) and progressive nephropathy (7% vs 28% in primary prevention at 6 years and 11% vs 32% in secondary prevention).
In addition to studying the effects of blood pressure control, the UKPDS also studied the effects of intensive blood glucose control.24,25 Nearly 4,000 patients with newly diagnosed type 2 diabetes were randomized to intensive treatment with a sulfonylurea or insulin, or to conventional treatment with diet. Over 10 years, the mean hemoglobin A1c was reduced to 7.0% in the intensive group and 7.9% in the conventional group. The risk of any diabetes-related end point was 12% lower in the intensive group, 10% lower for diabetes-related death, and 6% lower for all-cause mortality. There was also a 25% reduction in microvascular disease (retinopathy and nephropathy). However, the intensive group had more hypoglycemic episodes than the conventional group and a tendency to some increase in macrovascular events. A legacy effect was evident: patients who had intensive treatment had less microvascular disease progression years after stopping therapy.
Tight glycemic control reduces nephropathy, but does it increase cardiovascular risk?
Earlier trials provided strong evidence that blood glucose control prevents or slows retinopathy and nephropathy. The critical question is, “At what expense?” Although diabetes is the most common cause of kidney failure in the United States, most people with diabetes do not die of kidney failure, but of cardiovascular disease. Two recent large trials had different results regarding glycemic control below hemoglobin A1c of 7.0% and macrovascular risk, creating a controversy about what recommendations are best.
The ADVANCE trial, enrolling 11,140 patients with type 2 diabetes, was largely conducted in Australia and used the sulfonylurea glipizide for glycemic control. Compared with the group that received standard therapy (n=5,569), the intensive-treatment group (n=5,571) achieved mean hemoglobin A1c levels of 6.5% compared with 7.3% in the standard group, and had less nephropathy, less microalbuminuria, less doubling of creatinine, and a lower rate of end-stage renal disease (4% vs 5% in the standard therapy group). No difference between the two groups was found in retinopathy. Rates of all-cause mortality did not differ between the groups.9
The ACCORD trial had more than 10,000 subjects with type 2 diabetes and took place mostly in the United States. Using mainly rosiglitazone for intensive therapy, the intensive group achieved hemoglobin A1c levels of 6.4% vs 7.5% in the standard-therapy group. The trial was stopped early, at 3.7 years, because of a higher risk of death and cardiovascular events in the group with intensive glycemic control. However, the intensive-therapy group did have a significant decrease in microvascular renal outcomes and a reduction in the progression of retinopathy.14,26
In summary, tighter glycemic control improves microvascular complications—both retinopathy and nephropathy—in patients with type 2 diabetes. The benefit of intensive therapy on macrovascular complications (stroke, myocardial infarction) in long-standing diabetes has not been convincingly demonstrated in randomized trials. The UKPDS suggested that maintaining a hemoglobin A1c of 7% in patients newly diagnosed with type 2 diabetes confers long-term cardiovascular benefits. The target hemoglobin A1c for type 2 diabetes should be tailored to the patient: 7% is a reasonable goal for most patients, but the goal should be higher for the elderly and frail. Reducing the risk of cardiovascular death is still best done by controlling blood pressure, reducing lipids, quitting smoking, and losing weight.
STRATEGY 3: INHIBIT THE RENIN-ANGIOTENSIN-ALDOSTERONE AXIS
Components of the renin-angiotensin-aldo-sterone system are present not only in the circulation but also in many tissues, including the heart, brain, kidney, blood vessels, and adrenal glands. The role of renin-angiotensin-aldosterone system blockers in treating and preventing diabetic nephropathy has become controversial in recent years with findings from new studies.
The renin-angiotensin-aldosterone system is important in the development or maintenance of high blood pressure and the resultant damage to the brain, heart, and kidney. Drug development has focused on inhibiting steps in the biochemical pathway. ACE inhibitors block the formation of angiotensin II—the most biologically potent angiotensin peptide—and are among the most commonly used drugs to treat hypertension and concomitant conditions, such as renal insufficiency, proteinuria, and heart failure. Angiotensin receptor blockers (ARBs) interact with the angiotensin AT1 receptor and block most of its actions. They are approved by the US Food and Drug Administration (FDA) for the treatment of hypertension, and they help prevent left ventricular hypertrophy and mesangial sclerosis. Large studies have shown that ACE inhibitors and ARBs offer similar cardiovascular benefit.
The glomerulus has the only capillary bed with a blood supply that drains into an efferent arteriole instead of a venule, providing high resistance to aid filtration. Efferent arterioles are rich in AT1 receptors. In the presence of angiotensin II they constrict, increasing pressure in the glomerulus, which can lead to proteinuria and glomerulosclerosis. ACE inhibitors and ARBs relax the efferent arteriole, allowing increased blood flow through the glomerulus. This reduction in intraglomerular pressure is associated with less proteinuria and less glomerulosclerosis.
Diabetes promotes renal disease in many ways. Glucose and advanced glycation end products can lead to increased blood flow and increased pressure in the glomerulus. Through a variety of pathways, hyperglycemia, acting on angiotensin II, leads to NF-kapa beta production, profibrotic cytokines, increased matrix, and eventual fibrosis. ACE inhibitors and ARBs counteract many of these.
ACE inhibitors and ARBs slow nephropathy progression beyond blood pressure control
Several major clinical trials27–32 examined the effects of either ACE inhibitors or ARBs in slowing the progression of diabetic nephropathy and have had consistently positive results.
The Collaborative Study Group30 was a 3-year randomized trial in 419 patients with type 1 diabetes, using the ACE inhibitor captopril vs placebo. Captopril was associated with less decline in kidney function and a 50% reduction in the risk of the combined end points of death, dialysis, and transplantation that was independent of the small difference in blood pressures between the two groups.
The Irbesartan Diabetic Nephropathy Trial (IDNT)31 studied the effect of the ARB irbesartan vs the calcium channel blocker amlodipine vs placebo over 2.6 years in 1,715 patients with type 2 diabetes. Irbesartan was found to be significantly more effective in protecting against the progression of nephropathy, independent of reduction in blood pressure.
The RENAAL trial,32 published in 2001, was a 3-year, randomized, double-blind study comparing the ARB losartan at increasing dosages with placebo (both taken in addition to conventional antihypertensive treatment) in 1,513 patients with type 2 diabetes and nephropathy. The blood pressure goal was 140/90 mm Hg in both groups, but the losartan group had a lower rate of doubling of serum creatinine, end-stage renal disease, and combined end-stage renal disease or death.
‘Aldosterone escape’ motivates the search for new therapies
An important reason for developing more ways to block the renin-angiotensin-aldosterone system is because of “aldosterone escape,” the phenomenon of angiotensin II or aldosterone returning to pretreatment levels despite continued ACE inhibition.
Biollaz et al,33 in a 1982 study of 19 patients with hypertension, showed that despite reducing blood pressure and keeping the blood level of ACE very low with twice-daily enalapril 20 mg, blood and urine levels of angiotensin II steadily rose back to baseline levels within a few months.
A growing body of evidence suggests that despite effective inhibition of angiotensin II activity, non-ACE synthetic pathways still permit angiotensin II generation via serine proteases such as chymase, cathepsin G, and tissue plasminogen activator.
Thus, efforts have been made to block the renin-angiotensin system in other places. In addition to ACE inhibitors and ARBs, two aldosterone receptor antagonists are available, spironolactone and eplerenone, both used to treat heart failure. A direct renin inhibitor, aliskiren, is also available.
Combination therapy—less proteinuria, but…
A number of studies have shown that combination treatment with agents having different targets in the renin-angiotensin-aldosterone system leads to larger reductions in albuminuria than does single-agent therapy.
Mogensen et al34 studied the effect of the ACE inhibitor lisinopril (20 mg per day) plus the ARB candesartan (16 mg per day) in subjects with microalbuminuria, hypertension, and type 2 diabetes. Combined treatment was more effective in reducing proteinuria.
Epstein et al35 studied the effects of the ACE inhibitor enalapril (20 mg/day) combined with either of two doses of the selective aldosterone receptor antagonist eplerenone (50 or 100 mg/day) or placebo. Both eplerenone dosages, when added to the enalapril treatment, significantly reduced albuminuria from baseline as early as week 4 (P < .001), but placebo treatment added to the enalapril did not result in any significant decrease in urinary albumin excretion. Systolic blood pressure decreased significantly in all treatment groups and by about the same amount.
The Aliskiren Combined With Losartan in Type 2 Diabetes and Nephropathy (AVOID) trial36 randomized more than 600 patients with type 2 diabetes and nephropathy to aliskiren (a renin inhibitor) or placebo added to the ARB losartan. Again, combination treatment was more renoprotective, independent of blood pressure lowering.
Worse outcomes with combination therapy?
More recent studies have indicated that although combination therapy reduces proteinuria to a greater extent than monotherapy, overall it worsens major renal and cardiovascular outcomes. The multicenter Ongoing Telmisartan Alone and in Combination With Ramipril Global Endpoint Trial (ONTARGET)37 randomized more than 25,000 patients age 55 and older with established atherosclerotic vascular disease or with diabetes and end-organ damage to receive either the ARB telmisartan 80 mg daily, the ACE inhibitor ramipril 10 mg daily, or both. Mean follow-up was 56 months. The combination-treatment group had higher rates of death and renal disease than the single-therapy groups (which did not differ from one another).
Why the combination therapy had poorer outcomes is under debate. Patients may get sudden drops in blood pressure that are not detected with only periodic monitoring. Renal failure was mostly acute rather than chronic, and the estimated GFR declined more in the combined therapy group than in the single-therapy groups.
The Aliskiren Trial in Type 2 Diabetes Using Cardiovascular and Renal Disease Endpoints (ALTITUDE) was designed to test the effect of the direct renin inhibitor aliskiren or placebo, both arms combined with either an ACE inhibitor or an ARB in patients with type 2 diabetes at high risk for cardiovascular and renal events. The trial was terminated early because of more strokes and deaths in the combination therapy arms. The results led the FDA to issue black box warnings against using aliskiren with these other classes of agents, and all studies testing similar combinations have been stopped. (In one study that was stopped and has not yet been published, 100 patients with proteinuria were treated with either aliskiren, the ARB losartan, or both, to evaluate the effects of aldosterone escape. Results showed no differences: about one-third of each group had this phenomenon.)
My personal recommendation is as follows: for younger patients with proteinuria, at lower risk for cardiovascular events and with disease due not to diabetes but to immunoglobulin A nephropathy or another proteinuric kidney disease, treat with both an ACE inhibitor and ARB. But the combination should not be used for patients at high risk of cardiovascular disease, which includes almost all patients with diabetes.
If more aggressive renin-angiotensin system blockade is needed against diabetic nephropathy, adding a diuretic increases the impact of blocking the renin-angiotensin-aldosterone system on both proteinuria and progression of renal disease. The aldosterone blocker spironolactone 25 mg can be added if potassium levels are carefully monitored.
ACE inhibitor plus calcium channel blocker is safer than ACE inhibitor plus diuretic
The Avoiding Cardiovascular Events Through Combination Therapy in Patients Living With Systolic Hypertension (ACCOMPLISH) trial38 randomized more than 11,000 high-risk patients with hypertension to receive an ACE inhibitor (benazepril) plus either a calcium channel blocker (amlodipine) or thiazide diuretic (hydrochlorothiazide). Blood pressures were identical between the two groups, but the trial was terminated early, at 36 months, because of a higher risk of the combined end point of cardiovascular death, myocardial infarction, stroke, and other major cardiac events in the ACE inhibitor-thiazide group.
Although some experts believe this study is definitive and indicates that high blood pressure should never be treated with an ACE inhibitor-thiazide combination, I believe that caution is needed in interpreting these findings. This regimen should be avoided in older patients with diabetes at high risk for cardiovascular disease, but otherwise, getting blood pressure under control is critical, and this combination can be used if it works and the patient is tolerating it well.
In summary, the choice of blood pressure-lowering medications is based on reducing cardiovascular events and slowing the progression of kidney disease. Either an ACE inhibitor or an ARB is the first choice for patients with diabetes, hypertension, and any degree of proteinuria. Many experts recommend beginning one of these agents even if proteinuria is not present. However, the combination of an ACE inhibitor and ARB should not be used in diabetic patients, especially if they have cardiovascular disease, until further data clarify the results of the ONTARGET and ALTITUDE trials.
STRATEGY 4: METABOLIC MANIPULATION WITH NOVEL AGENTS
Several new agents have recently been studied for the treatment of diabetic nephropathy, including aminoguanidine, which reduces levels of advanced glycation end-products, and sulodexide, which blocks basement membrane permeability. Neither agent has been shown to be safe and effective in diabetic nephropathy. The newest agent is bardoxolone methyl. It induces the Keap1–Nrf2 pathway, which up-regulates cytoprotective factors, suppressing inflammatory and other cytokines that are major mediators of progression of chronic kidney disease.39
Pergola et al,40 in a phase 2, double-blind trial, randomized 227 adults with diabetic kidney disease and a low estimated GFR (20–45 mL/min/1.73 m2) to receive placebo or bardoxolone 25, 75, or 150 mg daily. Drug treatment was associated with improvement in the estimated GFR, a finding that persisted throughout the 52 weeks of treatment. Surprisingly, proteinuria did not decrease with drug treatment.
As of this writing, a large multicenter controlled randomized trial has been halted because of concerns by the data safety monitoring board, which found increased rates of death and fluid retention with the drug. A number of recent trials have shown a beneficial effect of sodium bicarbonate therapy in patients with late-stage chronic kidney disease. They have shown slowing of the progression of GFR decline in a number of renal diseases, including diabetes.
- Ritz E, Orth SR. Nephropathy in patients with type 2 diabetes mellitus. N Engl J Med 1999; 341:1127–1133.
- de Boer IH, Rue TC, Hall YN, Heagerty PJ, Weiss NS, Himmelfarb J. Temporal trends in the prevalence of diabetic kidney disease in the United States. JAMA 2011; 305:2532–2539.
- United States Renal Data System (USRDS) 2000 Annual Data Report. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases – Division of Kidney, Urologic and Hematologic Diseases. USRDS Coordinating Center operated by the Minneapolis Medical Research Foundation. www.usrds.org
- Macisaac RJ, Jerums G. Diabetic kidney disease with and without albuminuria. Curr Opin Nephrol Hypertens 2011; 20:246–257.
- Molitch ME, DeFronzo RA, Franz MJ, et al; American Diabetes Association. Nephropathy in diabetes. Diabetes Care 2004; 27(suppl 1):S79–S83.
- Vamos EP, Harris M, Millett C, et al. Association of systolic and diastolic blood pressure and all cause mortality in people with newly diagnosed type 2 diabetes: retrospective cohort study. BMJ 2012; 345:e5567.
- de Zeeuw D, Remuzzi G, Parving HH, et al. Proteinuria, a target for renoprotection in patients with type 2 diabetic nephropathy: lessons from RENAAL. Kidney Int 2004; 65:2309–2320.
- Parving HH, Andersen AR, Smidt UM, Svendsen PA. Early aggressive antihypertensive treatment reduces rate of decline in kidney function in diabetic nephropathy. Lancet 1983; 1:1175–1179.
- ADVANCE Collaborative Group; Patel A, MacMahon S, Chalmers J, et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008; 358:2560–2572.
- ADVANCE Collaborative Group; Patel A, MacMahon S, Chalmers J, et al. Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): a randomised controlled trial. Lancet 2007; 370:829–840.
- Bangalore S, Kumar S, Lobach I, Messerli FH. Blood pressure targets in subjects with type 2 diabetes mellitus/impaired fasting glucose: observations from traditional and bayesian random-effects meta-analyses of randomized trials. Circulation 2011; 123:2799–2810.
- UK Prospective Diabetes Study Group. Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. BMJ 1998; 317:703–713.
- Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 2008; 359:1577–1589.
- Action to Control Cardiovascular Risk in Diabetes Study Group; Gerstein HC, Miller ME, Byington RP, et al. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008; 358:2545–2559.
- ACCORD Study Group; Cushman WC, Evans GW, Byington RP, et al. Effects of intensive blood-pressure control in type 2 diabetes mellitus. N Engl J Med 2010; 362:1575–1585.
- American Diabetes Association. Standards of medical care in diabetes—2012. Diabetes Care 2012; 35(suppl 1):S11–S63. (Erratum in: Diabetes Care 2012; 35:660.)
- Bakris GL, Williams M, Dworkin L, et al. Preserving renal function in adults with hypertension and diabetes: a consensus approach. National Kidney Foundation Hypertension and Diabetes Executive Committees Working Group. Am J Kidney Dis 2000; 36:646–661.
- Ramsay L, Williams B, Johnston G, et al. Guidelines for management of hypertension: report of the third working party of the British Hypertension Society. J Hum Hypertens 1999; 13:569–592.
- Feldman RD, Campbell N, Larochelle P, et al. 1999 Canadian recommendations for the management of hypertension. Task Force for the Development of the 1999 Canadian Recommendations for the Management of Hypertension. CMAJ 1999; 161(suppl):12:S1–S17.
- Chalmers J, MacMahon S, Mancia G, et al. 1999 World Health Organization-International Society of Hypertension Guidelines for the management of hypertension. Guidelines Sub-committee of the World Health Organization. Clin Exp Hypertens 1999; 21:1009–1060.
- The seventh report of the Joint National Committee on Prevention, Detection Evaluation, and Treatment of High Blood Pressure. Hypertension 2003; 42:1206–1252.
- The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med 1993; 329:977–986.
- Shichiri M, Kishikawa H, Ohkubo Y, Wake N. Long-term results of the Kumamoto Study on optimal diabetes control in type 2 diabetic patients. Diabetes Care 2000; 23(suppl 2):B21–B29.
- Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998; 352:837–853. Erratum in: Lancet 1999; 354:602.
- Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. UK Prospective Diabetes Study Group. BMJ 1998; 317:703–713. Erratum in: BMJ 1999; 318:29.
- Ismail-Beigi F, Craven T, Banerji MA, et al; ACCORD trial group. Effect of intensive treatment of hyperglycaemia on microvascular outcomes in type 2 diabetes: an analysis of the ACCORD randomised trial. Lancet 2010; 376:419–430. Erratum in: Lancet 2010; 376:1466.
- Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Heart Outcomes Prevention Evaluation Study Investigators. Lancet 2000: 355:253–259. Erratum in: Lancet2000; 356:860.
- Parving HH, Lehnert H, Bröchner-Mortensen J, et al; Irbesartan in Patients with Type 2 Diabetes and Microalbuminuria Study Group. The effect of irbesartan on the development of diabetic nephropathy in patients with type 2 diabetes. N Engl J Med 2001; 345:870–878.
- Viberti G, Wheeldon NM; MicroAlbuminuria Reduction With VALsartan (MARVAL) Study Investigators. Microalbuminuria reduction with valsartan in patients with type 2 diabetes mellitus: a blood pressure-independent effect. Circulation 2002; 106:672–678.
- Lewis EJ, Hunsicker LG, Bain R P, Rohde RD. The effect of angiotensin-converting-enzyme inhibition on diabetic nephropathy. The Collaborative Study Group. N Engl J Med 1993; 329:1456–1462.
- Lewis EJ, Hunsicker LG, Clarke WR, et al; Collaborative Study Group. Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med 2001; 345:851–860.
- Brenner BM, Cooper ME, de Zeeuw D, et al; RENAAL Study Investigators. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med 2001; 345:861–869.
- Biollaz J, Brunner HR, Gavras I, Waeber B, Gavras H. Antihypertensive therapy with MK 421: angiotensin II--renin relationships to evaluate efficacy of converting enzyme blockade. J Cardiovasc Pharmacol 1982; 4:966–972.
- Mogensen CE, Neldam S, Tikkanen I, et al. Randomised controlled trial of dual blockade of renin-angiotensin system in patients with hypertension, microalbuminuria, and non-insulin dependent diabetes: the candesartan and lisinopril microalbuminuria (CALM) study. BMJ 2000; 321:1440–1444.
- Epstein M, Williams GH, Weinberger M, et al. Selective aldosterone blockade with eplerenone reduces albuminuria in patients with type 2 diabetes. Clin J Am Soc Nephrol 2006; 1:940–951.
- Parving HH, Persson F, Lewis JB, Lewis EJ, Hollenberg NK; AVOID Study Investigators. Aliskiren combined with losartan in type 2 diabetes and nephropathy. N Engl J Med 2008; 358:2433–2446.
- Mann JF, Schmieder RE, McQueen M, et al; ONTARGET investigators. Renal outcomes with telmisartan, ramipril, or both, in people at high vascular risk (the ONTARGET study): a multicentre, randomised, double-blind, controlled trial. Lancet 2008; 372:547–553.
- Jamerson K, Weber MA, Bakris GL, et al; ACCOMPLISH Trial Investigators. Benazepril plus amlodipine or hydrochlorothiazide for hypertension in high-risk patients. N Engl J Med 2008; 359:2417–2428.
- Kim HJ, Vaziri ND. Contribution of impaired Nrf2-Keap1 pathway to oxidative stress and inflammation in chronic renal failure. Am J Physiol Renal Physiol 2010; 298:F662–F671.
- Pergola PE, Raskin P, Toto RD, et al; BEAM Study Investigators Bardoxolone methyl and kidney function in CKD with type 2 diabetes. N Engl J Med 2011; 365:327–336.
Diabetes is on the rise, and so is diabetic nephropathy. In view of this epidemic, physicians should consider strategies to detect and control kidney disease in their diabetic patients.
This article will focus on kidney disease in adult-onset type 2 diabetes. Although it has different pathogenetic mechanisms than type 1 diabetes, the clinical course of the two conditions is very similar in terms of the prevalence of proteinuria after diagnosis, the progression to renal failure after the onset of proteinuria, and treatment options.1
DIABETES AND DIABETIC KIDNEY DISEASE ARE ON THE RISE
The incidence of diabetes increases with age, and with the aging of the baby boomers, its prevalence is growing dramatically. The 2005– 2008 National Health and Nutrition Examination Survey estimated the prevalence as 3.7% in adults age 20 to 44, 13.7% at age 45 to 64, and 26.9% in people age 65 and older. The obesity epidemic is also contributing to the increase in diabetes in all age groups.
Diabetic kidney disease has increased in the United States from about 4 million cases 20 years ago to about 7 million in 2005–2008.2 Diabetes is the major cause of end-stage renal disease in the developed world, accounting for 40% to 50% of cases. Other major causes are hypertension (27%) and glomerulonephritis (13%).3
Physicians in nearly every field of medicine now care for patients with diabetic nephropathy. The classic presentation—a patient who has impaired vision, fluid retention with edema, and hypertension—is commonly seen in dialysis units and ophthalmology and cardiovascular clinics.
CLINICAL PROGRESSION
Early in the course of diabetic nephropathy, blood pressure is normal and microalbuminuria is not evident, but many patients have a high glomerular filtration rate (GFR), indicating temporarily “enhanced” renal function or hyperfiltration. The next stage is characterized by microalbuminuria, correlating with glomerular mesangial expansion: the GFR falls back into the normal range and blood pressure starts to increase. Finally, macroalbuminuria occurs, accompanied by rising blood pressure and a declining GFR, correlating with the histologic appearance of glomerulosclerosis and Kimmelstiel-Wilson nodules.4
Hypertension develops in 5% of patients by 10 years after type 1 diabetes is diagnosed, 33% by 20 years, and 70% by 40 years. In contrast, 40% of patients with type 2 diabetes have high blood pressure at diagnosis.
Unfortunately, in most cases, this progression is a one-way street, so it is critical to intervene to try to slow the progression early in the course of the disease process.
SCREENING FOR DIABETIC NEPHROPATHY
Nephropathy screening guidelines for patients with diabetes are provided in Table 1.5
Blood pressure should be monitored at each office visit (Table 1). The goal for adults with diabetes should be to reduce blood pressure to 130/80 mm Hg. Reduction beyond this level may be associated with an increased mortality rate.6 Very high blood pressure (> 180 mm Hg systolic) should be lowered slowly. Lowering blood pressure delays the progression from microalbuminuria (30–299 mg/day or 20–199 μg/min) to macroalbuminuria (> 300 mg/day or > 200 μg/min) and slows the progression to renal failure.
Urinary albumin. Proteinuria takes 5 to 10 years to develop after the onset of diabetes. Because it is possible for patients with type 2 diabetes to have had the disease for some time before being diagnosed, urinary albumin screening should be performed at diagnosis and annually thereafter. Patients with type 1 are usually diagnosed with diabetes at or near onset of disease; therefore, annual screening for urinary albumin can begin 5 years after diagnosis.5
Proteinuria can be measured in different ways (Table 2). The basic screening test for clinical proteinuria is the urine dipstick, which is very sensitive to albumin and relatively insensitive to other proteins. “Trace-positive” results are common in healthy people, so proteinuria is not confirmed unless a patient has repeatedly positive results.
Microalbuminuria is important to measure, especially if it helps determine therapy. It is not detectable by the urinary dipstick, but can be measured in the following ways:
- Measurement of the albumin-creatinine ratio in a random spot collection
- 24-hour collection (creatinine should simultaneously be measured and creatinine clearance calculated)
- Timed collection (4 hours or overnight).
The first method is preferred, and any positive test result must be confirmed by repeat analyses of urinary albumin before a patient is diagnosed with microalbuminuria.
Occasionally a patient presenting with proteinuria but normal blood sugar and hemoglobin A1c will have a biopsy that reveals morphologic changes of classic diabetic nephropathy. Most such patients have a history of hyperglycemia, indicating that they actually have been diabetic.
Proteinuria—the best marker of disease progression
Proteinuria is the strongest predictor of renal outcomes. The Reduction in End Points in Noninsulin-Dependent Diabetes Mellitus With the Angiotensin II Antagonist Losartan (RENAAL) study was a randomized, placebo-controlled trial in more than 1,500 patients with type 2 diabetes to test the effects of losartan on renal outcome. Those with high albuminuria (> 3.0 g albumin/g creatinine) at baseline were five times more likely to reach a renal end point and were eight times more likely to have progression to end-stage renal disease than patients with low albuminuria (< 1.5 g/g).7 The degree of albuminuria after 6 months of treatment showed similar predictive trends, indicating that monitoring and treating proteinuria are extremely important goals.
STRATEGY 1 TO LIMIT RENAL INJURY: REDUCE BLOOD PRESSURE
Blood pressure control improves renal and cardiovascular function.
As early as 1983, Parving et al,8 in a study of only 10 insulin-dependent diabetic patients, showed strong evidence that early aggressive antihypertensive treatment improved the course of diabetic nephropathy. During the mean pretreatment period of 29 months, the GFR decreased significantly and the urinary albumin excretion rate and arterial blood pressure rose significantly. During the mean 39-month period of antihypertensive treatment with metoprolol, hydralazine, and furosemide or a thiazide, mean arterial blood pressure fell from 144/97 to 128/84 mm Hg and urinary albumin excretion from 977 to 433 μg/ min. The rate of decline in GFR slowed from 0.91 mL/min/month before treatment to 0.39 mL/min/month during treatment.
The Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation (ADVANCE) trial9 enrolled more than 11,000 patients internationally with type 2 diabetes at high risk for cardiovascular events. In addition to standard therapy, blood pressure was intensively controlled in one group with a combination of the angiotensin-converting enzyme (ACE) inhibitor perindopril and the diuretic indapamide. The intensive-therapy group achieved blood pressures less than 140/80 mm Hg and had a mean reduction of systolic blood pressure of 5.6 mm Hg and diastolic blood pressure of 2.2 mm Hg vs controls. Despite these apparently modest reductions, the intensively controlled group had a significant 9% reduction of the primary outcome of combined macrovascular events (cardiovascular death, myocardial infarction, and stroke) and microvascular events (new or worsening nephropathy, or retinopathy).10
A meta-analysis of studies of patients with type 2 diabetes found reduced nephropathy with systolic blood pressure control to less than 130 mm Hg.11
The United Kingdom Prospective Diabetes Study (UKPDS) is a series of studies of diabetes. The original study in 1998 enrolled 5,102 patients with newly diagnosed type 2 diabetes.12 The more than 1,000 patients with hypertension were randomized to either tight blood pressure control or regular care. The intensive treatment group had a mean blood pressure reduction of 9 mm Hg systolic and 3 mm Hg diastolic, along with major reductions in all diabetes end points, diabetes deaths, microvascular disease, and stroke over a median follow-up of 8.4 years.
Continuous blood pressure control is critical
Tight blood pressure control must be maintained to have continued benefit. During the 10 years following the UKPDS, no attempts were made to maintain the previously assigned therapies. A follow-up study13 of 884 UKPDS patients found that blood pressures were the same again between the two groups 2 years after the trial was stopped, and no beneficial legacy effect from previous blood pressure control was evident on end points.
Control below 120 mm Hg systolic not needed
Blood pressure control slows kidney disease and prevents major macrovascular disease, but there is no evidence that lowering systolic blood pressure below 120 mm Hg provides additional benefit. In the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial,14 more than 10,000 patients with type 2 diabetes and existing cardiovascular disease or additional cardiovascular risk factors were randomized to a goal of systolic blood pressure less than 120 mm Hg or less than 140 mm Hg (actual mean systolic pressures were 119 vs 134 mm Hg, respectively). Over nearly 5 years, there was no difference in cardiovascular events or deaths between the two groups.15
Since 1997, six international organizations have revised their recommended blood pressure goals in diabetes mellitus and renal diseases. Randomized clinical trials and observational studies have demonstrated the importance of blood pressure control to the level of 125/75 to 140/80 mm Hg. The National Kidney Foundation, the American Diabetes Association, and the Canadian Hypertension Society have developed consensus guidelines for blood pressure control to less than 130/80 mm Hg.16–21 Table 3 summarizes blood pressure goals for patients with diabetes.
STRATEGY 2: CONTROL BLOOD SUGAR
Recommendations for blood sugar goals are more controversial.
The Diabetes Control and Complications Trial22 provided early evidence that tight blood sugar control slows the development of microalbuminuria and macroalbuminuria. The study randomized more than 1,400 patients with type 1 diabetes to either standard therapy (1 or 2 daily insulin injections) or intensive therapy (an external insulin pump or 3 or more insulin injections guided by frequent blood glucose monitoring) to keep blood glucose levels close to normal. About half the patients had mild retinopathy at baseline and the others had no retinopathy. After 6.5 years, intensive therapy was found to significantly delay the onset and slow the progression of diabetic retinopathy and nephropathy.
The Kumamoto Study23 randomized 110 patients with type 2 diabetes and either no retinopathy (primary prevention cohort) or simple retinopathy (secondary prevention cohort) to receive either multiple insulin injections or conventional insulin therapy over 8 years. Intensive therapy led to lower rates of retinopathy (7.7% vs 32% in primary prevention and 19% vs 44% in secondary prevention) and progressive nephropathy (7% vs 28% in primary prevention at 6 years and 11% vs 32% in secondary prevention).
In addition to studying the effects of blood pressure control, the UKPDS also studied the effects of intensive blood glucose control.24,25 Nearly 4,000 patients with newly diagnosed type 2 diabetes were randomized to intensive treatment with a sulfonylurea or insulin, or to conventional treatment with diet. Over 10 years, the mean hemoglobin A1c was reduced to 7.0% in the intensive group and 7.9% in the conventional group. The risk of any diabetes-related end point was 12% lower in the intensive group, 10% lower for diabetes-related death, and 6% lower for all-cause mortality. There was also a 25% reduction in microvascular disease (retinopathy and nephropathy). However, the intensive group had more hypoglycemic episodes than the conventional group and a tendency to some increase in macrovascular events. A legacy effect was evident: patients who had intensive treatment had less microvascular disease progression years after stopping therapy.
Tight glycemic control reduces nephropathy, but does it increase cardiovascular risk?
Earlier trials provided strong evidence that blood glucose control prevents or slows retinopathy and nephropathy. The critical question is, “At what expense?” Although diabetes is the most common cause of kidney failure in the United States, most people with diabetes do not die of kidney failure, but of cardiovascular disease. Two recent large trials had different results regarding glycemic control below hemoglobin A1c of 7.0% and macrovascular risk, creating a controversy about what recommendations are best.
The ADVANCE trial, enrolling 11,140 patients with type 2 diabetes, was largely conducted in Australia and used the sulfonylurea glipizide for glycemic control. Compared with the group that received standard therapy (n=5,569), the intensive-treatment group (n=5,571) achieved mean hemoglobin A1c levels of 6.5% compared with 7.3% in the standard group, and had less nephropathy, less microalbuminuria, less doubling of creatinine, and a lower rate of end-stage renal disease (4% vs 5% in the standard therapy group). No difference between the two groups was found in retinopathy. Rates of all-cause mortality did not differ between the groups.9
The ACCORD trial had more than 10,000 subjects with type 2 diabetes and took place mostly in the United States. Using mainly rosiglitazone for intensive therapy, the intensive group achieved hemoglobin A1c levels of 6.4% vs 7.5% in the standard-therapy group. The trial was stopped early, at 3.7 years, because of a higher risk of death and cardiovascular events in the group with intensive glycemic control. However, the intensive-therapy group did have a significant decrease in microvascular renal outcomes and a reduction in the progression of retinopathy.14,26
In summary, tighter glycemic control improves microvascular complications—both retinopathy and nephropathy—in patients with type 2 diabetes. The benefit of intensive therapy on macrovascular complications (stroke, myocardial infarction) in long-standing diabetes has not been convincingly demonstrated in randomized trials. The UKPDS suggested that maintaining a hemoglobin A1c of 7% in patients newly diagnosed with type 2 diabetes confers long-term cardiovascular benefits. The target hemoglobin A1c for type 2 diabetes should be tailored to the patient: 7% is a reasonable goal for most patients, but the goal should be higher for the elderly and frail. Reducing the risk of cardiovascular death is still best done by controlling blood pressure, reducing lipids, quitting smoking, and losing weight.
STRATEGY 3: INHIBIT THE RENIN-ANGIOTENSIN-ALDOSTERONE AXIS
Components of the renin-angiotensin-aldo-sterone system are present not only in the circulation but also in many tissues, including the heart, brain, kidney, blood vessels, and adrenal glands. The role of renin-angiotensin-aldosterone system blockers in treating and preventing diabetic nephropathy has become controversial in recent years with findings from new studies.
The renin-angiotensin-aldosterone system is important in the development or maintenance of high blood pressure and the resultant damage to the brain, heart, and kidney. Drug development has focused on inhibiting steps in the biochemical pathway. ACE inhibitors block the formation of angiotensin II—the most biologically potent angiotensin peptide—and are among the most commonly used drugs to treat hypertension and concomitant conditions, such as renal insufficiency, proteinuria, and heart failure. Angiotensin receptor blockers (ARBs) interact with the angiotensin AT1 receptor and block most of its actions. They are approved by the US Food and Drug Administration (FDA) for the treatment of hypertension, and they help prevent left ventricular hypertrophy and mesangial sclerosis. Large studies have shown that ACE inhibitors and ARBs offer similar cardiovascular benefit.
The glomerulus has the only capillary bed with a blood supply that drains into an efferent arteriole instead of a venule, providing high resistance to aid filtration. Efferent arterioles are rich in AT1 receptors. In the presence of angiotensin II they constrict, increasing pressure in the glomerulus, which can lead to proteinuria and glomerulosclerosis. ACE inhibitors and ARBs relax the efferent arteriole, allowing increased blood flow through the glomerulus. This reduction in intraglomerular pressure is associated with less proteinuria and less glomerulosclerosis.
Diabetes promotes renal disease in many ways. Glucose and advanced glycation end products can lead to increased blood flow and increased pressure in the glomerulus. Through a variety of pathways, hyperglycemia, acting on angiotensin II, leads to NF-kapa beta production, profibrotic cytokines, increased matrix, and eventual fibrosis. ACE inhibitors and ARBs counteract many of these.
ACE inhibitors and ARBs slow nephropathy progression beyond blood pressure control
Several major clinical trials27–32 examined the effects of either ACE inhibitors or ARBs in slowing the progression of diabetic nephropathy and have had consistently positive results.
The Collaborative Study Group30 was a 3-year randomized trial in 419 patients with type 1 diabetes, using the ACE inhibitor captopril vs placebo. Captopril was associated with less decline in kidney function and a 50% reduction in the risk of the combined end points of death, dialysis, and transplantation that was independent of the small difference in blood pressures between the two groups.
The Irbesartan Diabetic Nephropathy Trial (IDNT)31 studied the effect of the ARB irbesartan vs the calcium channel blocker amlodipine vs placebo over 2.6 years in 1,715 patients with type 2 diabetes. Irbesartan was found to be significantly more effective in protecting against the progression of nephropathy, independent of reduction in blood pressure.
The RENAAL trial,32 published in 2001, was a 3-year, randomized, double-blind study comparing the ARB losartan at increasing dosages with placebo (both taken in addition to conventional antihypertensive treatment) in 1,513 patients with type 2 diabetes and nephropathy. The blood pressure goal was 140/90 mm Hg in both groups, but the losartan group had a lower rate of doubling of serum creatinine, end-stage renal disease, and combined end-stage renal disease or death.
‘Aldosterone escape’ motivates the search for new therapies
An important reason for developing more ways to block the renin-angiotensin-aldosterone system is because of “aldosterone escape,” the phenomenon of angiotensin II or aldosterone returning to pretreatment levels despite continued ACE inhibition.
Biollaz et al,33 in a 1982 study of 19 patients with hypertension, showed that despite reducing blood pressure and keeping the blood level of ACE very low with twice-daily enalapril 20 mg, blood and urine levels of angiotensin II steadily rose back to baseline levels within a few months.
A growing body of evidence suggests that despite effective inhibition of angiotensin II activity, non-ACE synthetic pathways still permit angiotensin II generation via serine proteases such as chymase, cathepsin G, and tissue plasminogen activator.
Thus, efforts have been made to block the renin-angiotensin system in other places. In addition to ACE inhibitors and ARBs, two aldosterone receptor antagonists are available, spironolactone and eplerenone, both used to treat heart failure. A direct renin inhibitor, aliskiren, is also available.
Combination therapy—less proteinuria, but…
A number of studies have shown that combination treatment with agents having different targets in the renin-angiotensin-aldosterone system leads to larger reductions in albuminuria than does single-agent therapy.
Mogensen et al34 studied the effect of the ACE inhibitor lisinopril (20 mg per day) plus the ARB candesartan (16 mg per day) in subjects with microalbuminuria, hypertension, and type 2 diabetes. Combined treatment was more effective in reducing proteinuria.
Epstein et al35 studied the effects of the ACE inhibitor enalapril (20 mg/day) combined with either of two doses of the selective aldosterone receptor antagonist eplerenone (50 or 100 mg/day) or placebo. Both eplerenone dosages, when added to the enalapril treatment, significantly reduced albuminuria from baseline as early as week 4 (P < .001), but placebo treatment added to the enalapril did not result in any significant decrease in urinary albumin excretion. Systolic blood pressure decreased significantly in all treatment groups and by about the same amount.
The Aliskiren Combined With Losartan in Type 2 Diabetes and Nephropathy (AVOID) trial36 randomized more than 600 patients with type 2 diabetes and nephropathy to aliskiren (a renin inhibitor) or placebo added to the ARB losartan. Again, combination treatment was more renoprotective, independent of blood pressure lowering.
Worse outcomes with combination therapy?
More recent studies have indicated that although combination therapy reduces proteinuria to a greater extent than monotherapy, overall it worsens major renal and cardiovascular outcomes. The multicenter Ongoing Telmisartan Alone and in Combination With Ramipril Global Endpoint Trial (ONTARGET)37 randomized more than 25,000 patients age 55 and older with established atherosclerotic vascular disease or with diabetes and end-organ damage to receive either the ARB telmisartan 80 mg daily, the ACE inhibitor ramipril 10 mg daily, or both. Mean follow-up was 56 months. The combination-treatment group had higher rates of death and renal disease than the single-therapy groups (which did not differ from one another).
Why the combination therapy had poorer outcomes is under debate. Patients may get sudden drops in blood pressure that are not detected with only periodic monitoring. Renal failure was mostly acute rather than chronic, and the estimated GFR declined more in the combined therapy group than in the single-therapy groups.
The Aliskiren Trial in Type 2 Diabetes Using Cardiovascular and Renal Disease Endpoints (ALTITUDE) was designed to test the effect of the direct renin inhibitor aliskiren or placebo, both arms combined with either an ACE inhibitor or an ARB in patients with type 2 diabetes at high risk for cardiovascular and renal events. The trial was terminated early because of more strokes and deaths in the combination therapy arms. The results led the FDA to issue black box warnings against using aliskiren with these other classes of agents, and all studies testing similar combinations have been stopped. (In one study that was stopped and has not yet been published, 100 patients with proteinuria were treated with either aliskiren, the ARB losartan, or both, to evaluate the effects of aldosterone escape. Results showed no differences: about one-third of each group had this phenomenon.)
My personal recommendation is as follows: for younger patients with proteinuria, at lower risk for cardiovascular events and with disease due not to diabetes but to immunoglobulin A nephropathy or another proteinuric kidney disease, treat with both an ACE inhibitor and ARB. But the combination should not be used for patients at high risk of cardiovascular disease, which includes almost all patients with diabetes.
If more aggressive renin-angiotensin system blockade is needed against diabetic nephropathy, adding a diuretic increases the impact of blocking the renin-angiotensin-aldosterone system on both proteinuria and progression of renal disease. The aldosterone blocker spironolactone 25 mg can be added if potassium levels are carefully monitored.
ACE inhibitor plus calcium channel blocker is safer than ACE inhibitor plus diuretic
The Avoiding Cardiovascular Events Through Combination Therapy in Patients Living With Systolic Hypertension (ACCOMPLISH) trial38 randomized more than 11,000 high-risk patients with hypertension to receive an ACE inhibitor (benazepril) plus either a calcium channel blocker (amlodipine) or thiazide diuretic (hydrochlorothiazide). Blood pressures were identical between the two groups, but the trial was terminated early, at 36 months, because of a higher risk of the combined end point of cardiovascular death, myocardial infarction, stroke, and other major cardiac events in the ACE inhibitor-thiazide group.
Although some experts believe this study is definitive and indicates that high blood pressure should never be treated with an ACE inhibitor-thiazide combination, I believe that caution is needed in interpreting these findings. This regimen should be avoided in older patients with diabetes at high risk for cardiovascular disease, but otherwise, getting blood pressure under control is critical, and this combination can be used if it works and the patient is tolerating it well.
In summary, the choice of blood pressure-lowering medications is based on reducing cardiovascular events and slowing the progression of kidney disease. Either an ACE inhibitor or an ARB is the first choice for patients with diabetes, hypertension, and any degree of proteinuria. Many experts recommend beginning one of these agents even if proteinuria is not present. However, the combination of an ACE inhibitor and ARB should not be used in diabetic patients, especially if they have cardiovascular disease, until further data clarify the results of the ONTARGET and ALTITUDE trials.
STRATEGY 4: METABOLIC MANIPULATION WITH NOVEL AGENTS
Several new agents have recently been studied for the treatment of diabetic nephropathy, including aminoguanidine, which reduces levels of advanced glycation end-products, and sulodexide, which blocks basement membrane permeability. Neither agent has been shown to be safe and effective in diabetic nephropathy. The newest agent is bardoxolone methyl. It induces the Keap1–Nrf2 pathway, which up-regulates cytoprotective factors, suppressing inflammatory and other cytokines that are major mediators of progression of chronic kidney disease.39
Pergola et al,40 in a phase 2, double-blind trial, randomized 227 adults with diabetic kidney disease and a low estimated GFR (20–45 mL/min/1.73 m2) to receive placebo or bardoxolone 25, 75, or 150 mg daily. Drug treatment was associated with improvement in the estimated GFR, a finding that persisted throughout the 52 weeks of treatment. Surprisingly, proteinuria did not decrease with drug treatment.
As of this writing, a large multicenter controlled randomized trial has been halted because of concerns by the data safety monitoring board, which found increased rates of death and fluid retention with the drug. A number of recent trials have shown a beneficial effect of sodium bicarbonate therapy in patients with late-stage chronic kidney disease. They have shown slowing of the progression of GFR decline in a number of renal diseases, including diabetes.
Diabetes is on the rise, and so is diabetic nephropathy. In view of this epidemic, physicians should consider strategies to detect and control kidney disease in their diabetic patients.
This article will focus on kidney disease in adult-onset type 2 diabetes. Although it has different pathogenetic mechanisms than type 1 diabetes, the clinical course of the two conditions is very similar in terms of the prevalence of proteinuria after diagnosis, the progression to renal failure after the onset of proteinuria, and treatment options.1
DIABETES AND DIABETIC KIDNEY DISEASE ARE ON THE RISE
The incidence of diabetes increases with age, and with the aging of the baby boomers, its prevalence is growing dramatically. The 2005– 2008 National Health and Nutrition Examination Survey estimated the prevalence as 3.7% in adults age 20 to 44, 13.7% at age 45 to 64, and 26.9% in people age 65 and older. The obesity epidemic is also contributing to the increase in diabetes in all age groups.
Diabetic kidney disease has increased in the United States from about 4 million cases 20 years ago to about 7 million in 2005–2008.2 Diabetes is the major cause of end-stage renal disease in the developed world, accounting for 40% to 50% of cases. Other major causes are hypertension (27%) and glomerulonephritis (13%).3
Physicians in nearly every field of medicine now care for patients with diabetic nephropathy. The classic presentation—a patient who has impaired vision, fluid retention with edema, and hypertension—is commonly seen in dialysis units and ophthalmology and cardiovascular clinics.
CLINICAL PROGRESSION
Early in the course of diabetic nephropathy, blood pressure is normal and microalbuminuria is not evident, but many patients have a high glomerular filtration rate (GFR), indicating temporarily “enhanced” renal function or hyperfiltration. The next stage is characterized by microalbuminuria, correlating with glomerular mesangial expansion: the GFR falls back into the normal range and blood pressure starts to increase. Finally, macroalbuminuria occurs, accompanied by rising blood pressure and a declining GFR, correlating with the histologic appearance of glomerulosclerosis and Kimmelstiel-Wilson nodules.4
Hypertension develops in 5% of patients by 10 years after type 1 diabetes is diagnosed, 33% by 20 years, and 70% by 40 years. In contrast, 40% of patients with type 2 diabetes have high blood pressure at diagnosis.
Unfortunately, in most cases, this progression is a one-way street, so it is critical to intervene to try to slow the progression early in the course of the disease process.
SCREENING FOR DIABETIC NEPHROPATHY
Nephropathy screening guidelines for patients with diabetes are provided in Table 1.5
Blood pressure should be monitored at each office visit (Table 1). The goal for adults with diabetes should be to reduce blood pressure to 130/80 mm Hg. Reduction beyond this level may be associated with an increased mortality rate.6 Very high blood pressure (> 180 mm Hg systolic) should be lowered slowly. Lowering blood pressure delays the progression from microalbuminuria (30–299 mg/day or 20–199 μg/min) to macroalbuminuria (> 300 mg/day or > 200 μg/min) and slows the progression to renal failure.
Urinary albumin. Proteinuria takes 5 to 10 years to develop after the onset of diabetes. Because it is possible for patients with type 2 diabetes to have had the disease for some time before being diagnosed, urinary albumin screening should be performed at diagnosis and annually thereafter. Patients with type 1 are usually diagnosed with diabetes at or near onset of disease; therefore, annual screening for urinary albumin can begin 5 years after diagnosis.5
Proteinuria can be measured in different ways (Table 2). The basic screening test for clinical proteinuria is the urine dipstick, which is very sensitive to albumin and relatively insensitive to other proteins. “Trace-positive” results are common in healthy people, so proteinuria is not confirmed unless a patient has repeatedly positive results.
Microalbuminuria is important to measure, especially if it helps determine therapy. It is not detectable by the urinary dipstick, but can be measured in the following ways:
- Measurement of the albumin-creatinine ratio in a random spot collection
- 24-hour collection (creatinine should simultaneously be measured and creatinine clearance calculated)
- Timed collection (4 hours or overnight).
The first method is preferred, and any positive test result must be confirmed by repeat analyses of urinary albumin before a patient is diagnosed with microalbuminuria.
Occasionally a patient presenting with proteinuria but normal blood sugar and hemoglobin A1c will have a biopsy that reveals morphologic changes of classic diabetic nephropathy. Most such patients have a history of hyperglycemia, indicating that they actually have been diabetic.
Proteinuria—the best marker of disease progression
Proteinuria is the strongest predictor of renal outcomes. The Reduction in End Points in Noninsulin-Dependent Diabetes Mellitus With the Angiotensin II Antagonist Losartan (RENAAL) study was a randomized, placebo-controlled trial in more than 1,500 patients with type 2 diabetes to test the effects of losartan on renal outcome. Those with high albuminuria (> 3.0 g albumin/g creatinine) at baseline were five times more likely to reach a renal end point and were eight times more likely to have progression to end-stage renal disease than patients with low albuminuria (< 1.5 g/g).7 The degree of albuminuria after 6 months of treatment showed similar predictive trends, indicating that monitoring and treating proteinuria are extremely important goals.
STRATEGY 1 TO LIMIT RENAL INJURY: REDUCE BLOOD PRESSURE
Blood pressure control improves renal and cardiovascular function.
As early as 1983, Parving et al,8 in a study of only 10 insulin-dependent diabetic patients, showed strong evidence that early aggressive antihypertensive treatment improved the course of diabetic nephropathy. During the mean pretreatment period of 29 months, the GFR decreased significantly and the urinary albumin excretion rate and arterial blood pressure rose significantly. During the mean 39-month period of antihypertensive treatment with metoprolol, hydralazine, and furosemide or a thiazide, mean arterial blood pressure fell from 144/97 to 128/84 mm Hg and urinary albumin excretion from 977 to 433 μg/ min. The rate of decline in GFR slowed from 0.91 mL/min/month before treatment to 0.39 mL/min/month during treatment.
The Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation (ADVANCE) trial9 enrolled more than 11,000 patients internationally with type 2 diabetes at high risk for cardiovascular events. In addition to standard therapy, blood pressure was intensively controlled in one group with a combination of the angiotensin-converting enzyme (ACE) inhibitor perindopril and the diuretic indapamide. The intensive-therapy group achieved blood pressures less than 140/80 mm Hg and had a mean reduction of systolic blood pressure of 5.6 mm Hg and diastolic blood pressure of 2.2 mm Hg vs controls. Despite these apparently modest reductions, the intensively controlled group had a significant 9% reduction of the primary outcome of combined macrovascular events (cardiovascular death, myocardial infarction, and stroke) and microvascular events (new or worsening nephropathy, or retinopathy).10
A meta-analysis of studies of patients with type 2 diabetes found reduced nephropathy with systolic blood pressure control to less than 130 mm Hg.11
The United Kingdom Prospective Diabetes Study (UKPDS) is a series of studies of diabetes. The original study in 1998 enrolled 5,102 patients with newly diagnosed type 2 diabetes.12 The more than 1,000 patients with hypertension were randomized to either tight blood pressure control or regular care. The intensive treatment group had a mean blood pressure reduction of 9 mm Hg systolic and 3 mm Hg diastolic, along with major reductions in all diabetes end points, diabetes deaths, microvascular disease, and stroke over a median follow-up of 8.4 years.
Continuous blood pressure control is critical
Tight blood pressure control must be maintained to have continued benefit. During the 10 years following the UKPDS, no attempts were made to maintain the previously assigned therapies. A follow-up study13 of 884 UKPDS patients found that blood pressures were the same again between the two groups 2 years after the trial was stopped, and no beneficial legacy effect from previous blood pressure control was evident on end points.
Control below 120 mm Hg systolic not needed
Blood pressure control slows kidney disease and prevents major macrovascular disease, but there is no evidence that lowering systolic blood pressure below 120 mm Hg provides additional benefit. In the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial,14 more than 10,000 patients with type 2 diabetes and existing cardiovascular disease or additional cardiovascular risk factors were randomized to a goal of systolic blood pressure less than 120 mm Hg or less than 140 mm Hg (actual mean systolic pressures were 119 vs 134 mm Hg, respectively). Over nearly 5 years, there was no difference in cardiovascular events or deaths between the two groups.15
Since 1997, six international organizations have revised their recommended blood pressure goals in diabetes mellitus and renal diseases. Randomized clinical trials and observational studies have demonstrated the importance of blood pressure control to the level of 125/75 to 140/80 mm Hg. The National Kidney Foundation, the American Diabetes Association, and the Canadian Hypertension Society have developed consensus guidelines for blood pressure control to less than 130/80 mm Hg.16–21 Table 3 summarizes blood pressure goals for patients with diabetes.
STRATEGY 2: CONTROL BLOOD SUGAR
Recommendations for blood sugar goals are more controversial.
The Diabetes Control and Complications Trial22 provided early evidence that tight blood sugar control slows the development of microalbuminuria and macroalbuminuria. The study randomized more than 1,400 patients with type 1 diabetes to either standard therapy (1 or 2 daily insulin injections) or intensive therapy (an external insulin pump or 3 or more insulin injections guided by frequent blood glucose monitoring) to keep blood glucose levels close to normal. About half the patients had mild retinopathy at baseline and the others had no retinopathy. After 6.5 years, intensive therapy was found to significantly delay the onset and slow the progression of diabetic retinopathy and nephropathy.
The Kumamoto Study23 randomized 110 patients with type 2 diabetes and either no retinopathy (primary prevention cohort) or simple retinopathy (secondary prevention cohort) to receive either multiple insulin injections or conventional insulin therapy over 8 years. Intensive therapy led to lower rates of retinopathy (7.7% vs 32% in primary prevention and 19% vs 44% in secondary prevention) and progressive nephropathy (7% vs 28% in primary prevention at 6 years and 11% vs 32% in secondary prevention).
In addition to studying the effects of blood pressure control, the UKPDS also studied the effects of intensive blood glucose control.24,25 Nearly 4,000 patients with newly diagnosed type 2 diabetes were randomized to intensive treatment with a sulfonylurea or insulin, or to conventional treatment with diet. Over 10 years, the mean hemoglobin A1c was reduced to 7.0% in the intensive group and 7.9% in the conventional group. The risk of any diabetes-related end point was 12% lower in the intensive group, 10% lower for diabetes-related death, and 6% lower for all-cause mortality. There was also a 25% reduction in microvascular disease (retinopathy and nephropathy). However, the intensive group had more hypoglycemic episodes than the conventional group and a tendency to some increase in macrovascular events. A legacy effect was evident: patients who had intensive treatment had less microvascular disease progression years after stopping therapy.
Tight glycemic control reduces nephropathy, but does it increase cardiovascular risk?
Earlier trials provided strong evidence that blood glucose control prevents or slows retinopathy and nephropathy. The critical question is, “At what expense?” Although diabetes is the most common cause of kidney failure in the United States, most people with diabetes do not die of kidney failure, but of cardiovascular disease. Two recent large trials had different results regarding glycemic control below hemoglobin A1c of 7.0% and macrovascular risk, creating a controversy about what recommendations are best.
The ADVANCE trial, enrolling 11,140 patients with type 2 diabetes, was largely conducted in Australia and used the sulfonylurea glipizide for glycemic control. Compared with the group that received standard therapy (n=5,569), the intensive-treatment group (n=5,571) achieved mean hemoglobin A1c levels of 6.5% compared with 7.3% in the standard group, and had less nephropathy, less microalbuminuria, less doubling of creatinine, and a lower rate of end-stage renal disease (4% vs 5% in the standard therapy group). No difference between the two groups was found in retinopathy. Rates of all-cause mortality did not differ between the groups.9
The ACCORD trial had more than 10,000 subjects with type 2 diabetes and took place mostly in the United States. Using mainly rosiglitazone for intensive therapy, the intensive group achieved hemoglobin A1c levels of 6.4% vs 7.5% in the standard-therapy group. The trial was stopped early, at 3.7 years, because of a higher risk of death and cardiovascular events in the group with intensive glycemic control. However, the intensive-therapy group did have a significant decrease in microvascular renal outcomes and a reduction in the progression of retinopathy.14,26
In summary, tighter glycemic control improves microvascular complications—both retinopathy and nephropathy—in patients with type 2 diabetes. The benefit of intensive therapy on macrovascular complications (stroke, myocardial infarction) in long-standing diabetes has not been convincingly demonstrated in randomized trials. The UKPDS suggested that maintaining a hemoglobin A1c of 7% in patients newly diagnosed with type 2 diabetes confers long-term cardiovascular benefits. The target hemoglobin A1c for type 2 diabetes should be tailored to the patient: 7% is a reasonable goal for most patients, but the goal should be higher for the elderly and frail. Reducing the risk of cardiovascular death is still best done by controlling blood pressure, reducing lipids, quitting smoking, and losing weight.
STRATEGY 3: INHIBIT THE RENIN-ANGIOTENSIN-ALDOSTERONE AXIS
Components of the renin-angiotensin-aldo-sterone system are present not only in the circulation but also in many tissues, including the heart, brain, kidney, blood vessels, and adrenal glands. The role of renin-angiotensin-aldosterone system blockers in treating and preventing diabetic nephropathy has become controversial in recent years with findings from new studies.
The renin-angiotensin-aldosterone system is important in the development or maintenance of high blood pressure and the resultant damage to the brain, heart, and kidney. Drug development has focused on inhibiting steps in the biochemical pathway. ACE inhibitors block the formation of angiotensin II—the most biologically potent angiotensin peptide—and are among the most commonly used drugs to treat hypertension and concomitant conditions, such as renal insufficiency, proteinuria, and heart failure. Angiotensin receptor blockers (ARBs) interact with the angiotensin AT1 receptor and block most of its actions. They are approved by the US Food and Drug Administration (FDA) for the treatment of hypertension, and they help prevent left ventricular hypertrophy and mesangial sclerosis. Large studies have shown that ACE inhibitors and ARBs offer similar cardiovascular benefit.
The glomerulus has the only capillary bed with a blood supply that drains into an efferent arteriole instead of a venule, providing high resistance to aid filtration. Efferent arterioles are rich in AT1 receptors. In the presence of angiotensin II they constrict, increasing pressure in the glomerulus, which can lead to proteinuria and glomerulosclerosis. ACE inhibitors and ARBs relax the efferent arteriole, allowing increased blood flow through the glomerulus. This reduction in intraglomerular pressure is associated with less proteinuria and less glomerulosclerosis.
Diabetes promotes renal disease in many ways. Glucose and advanced glycation end products can lead to increased blood flow and increased pressure in the glomerulus. Through a variety of pathways, hyperglycemia, acting on angiotensin II, leads to NF-kapa beta production, profibrotic cytokines, increased matrix, and eventual fibrosis. ACE inhibitors and ARBs counteract many of these.
ACE inhibitors and ARBs slow nephropathy progression beyond blood pressure control
Several major clinical trials27–32 examined the effects of either ACE inhibitors or ARBs in slowing the progression of diabetic nephropathy and have had consistently positive results.
The Collaborative Study Group30 was a 3-year randomized trial in 419 patients with type 1 diabetes, using the ACE inhibitor captopril vs placebo. Captopril was associated with less decline in kidney function and a 50% reduction in the risk of the combined end points of death, dialysis, and transplantation that was independent of the small difference in blood pressures between the two groups.
The Irbesartan Diabetic Nephropathy Trial (IDNT)31 studied the effect of the ARB irbesartan vs the calcium channel blocker amlodipine vs placebo over 2.6 years in 1,715 patients with type 2 diabetes. Irbesartan was found to be significantly more effective in protecting against the progression of nephropathy, independent of reduction in blood pressure.
The RENAAL trial,32 published in 2001, was a 3-year, randomized, double-blind study comparing the ARB losartan at increasing dosages with placebo (both taken in addition to conventional antihypertensive treatment) in 1,513 patients with type 2 diabetes and nephropathy. The blood pressure goal was 140/90 mm Hg in both groups, but the losartan group had a lower rate of doubling of serum creatinine, end-stage renal disease, and combined end-stage renal disease or death.
‘Aldosterone escape’ motivates the search for new therapies
An important reason for developing more ways to block the renin-angiotensin-aldosterone system is because of “aldosterone escape,” the phenomenon of angiotensin II or aldosterone returning to pretreatment levels despite continued ACE inhibition.
Biollaz et al,33 in a 1982 study of 19 patients with hypertension, showed that despite reducing blood pressure and keeping the blood level of ACE very low with twice-daily enalapril 20 mg, blood and urine levels of angiotensin II steadily rose back to baseline levels within a few months.
A growing body of evidence suggests that despite effective inhibition of angiotensin II activity, non-ACE synthetic pathways still permit angiotensin II generation via serine proteases such as chymase, cathepsin G, and tissue plasminogen activator.
Thus, efforts have been made to block the renin-angiotensin system in other places. In addition to ACE inhibitors and ARBs, two aldosterone receptor antagonists are available, spironolactone and eplerenone, both used to treat heart failure. A direct renin inhibitor, aliskiren, is also available.
Combination therapy—less proteinuria, but…
A number of studies have shown that combination treatment with agents having different targets in the renin-angiotensin-aldosterone system leads to larger reductions in albuminuria than does single-agent therapy.
Mogensen et al34 studied the effect of the ACE inhibitor lisinopril (20 mg per day) plus the ARB candesartan (16 mg per day) in subjects with microalbuminuria, hypertension, and type 2 diabetes. Combined treatment was more effective in reducing proteinuria.
Epstein et al35 studied the effects of the ACE inhibitor enalapril (20 mg/day) combined with either of two doses of the selective aldosterone receptor antagonist eplerenone (50 or 100 mg/day) or placebo. Both eplerenone dosages, when added to the enalapril treatment, significantly reduced albuminuria from baseline as early as week 4 (P < .001), but placebo treatment added to the enalapril did not result in any significant decrease in urinary albumin excretion. Systolic blood pressure decreased significantly in all treatment groups and by about the same amount.
The Aliskiren Combined With Losartan in Type 2 Diabetes and Nephropathy (AVOID) trial36 randomized more than 600 patients with type 2 diabetes and nephropathy to aliskiren (a renin inhibitor) or placebo added to the ARB losartan. Again, combination treatment was more renoprotective, independent of blood pressure lowering.
Worse outcomes with combination therapy?
More recent studies have indicated that although combination therapy reduces proteinuria to a greater extent than monotherapy, overall it worsens major renal and cardiovascular outcomes. The multicenter Ongoing Telmisartan Alone and in Combination With Ramipril Global Endpoint Trial (ONTARGET)37 randomized more than 25,000 patients age 55 and older with established atherosclerotic vascular disease or with diabetes and end-organ damage to receive either the ARB telmisartan 80 mg daily, the ACE inhibitor ramipril 10 mg daily, or both. Mean follow-up was 56 months. The combination-treatment group had higher rates of death and renal disease than the single-therapy groups (which did not differ from one another).
Why the combination therapy had poorer outcomes is under debate. Patients may get sudden drops in blood pressure that are not detected with only periodic monitoring. Renal failure was mostly acute rather than chronic, and the estimated GFR declined more in the combined therapy group than in the single-therapy groups.
The Aliskiren Trial in Type 2 Diabetes Using Cardiovascular and Renal Disease Endpoints (ALTITUDE) was designed to test the effect of the direct renin inhibitor aliskiren or placebo, both arms combined with either an ACE inhibitor or an ARB in patients with type 2 diabetes at high risk for cardiovascular and renal events. The trial was terminated early because of more strokes and deaths in the combination therapy arms. The results led the FDA to issue black box warnings against using aliskiren with these other classes of agents, and all studies testing similar combinations have been stopped. (In one study that was stopped and has not yet been published, 100 patients with proteinuria were treated with either aliskiren, the ARB losartan, or both, to evaluate the effects of aldosterone escape. Results showed no differences: about one-third of each group had this phenomenon.)
My personal recommendation is as follows: for younger patients with proteinuria, at lower risk for cardiovascular events and with disease due not to diabetes but to immunoglobulin A nephropathy or another proteinuric kidney disease, treat with both an ACE inhibitor and ARB. But the combination should not be used for patients at high risk of cardiovascular disease, which includes almost all patients with diabetes.
If more aggressive renin-angiotensin system blockade is needed against diabetic nephropathy, adding a diuretic increases the impact of blocking the renin-angiotensin-aldosterone system on both proteinuria and progression of renal disease. The aldosterone blocker spironolactone 25 mg can be added if potassium levels are carefully monitored.
ACE inhibitor plus calcium channel blocker is safer than ACE inhibitor plus diuretic
The Avoiding Cardiovascular Events Through Combination Therapy in Patients Living With Systolic Hypertension (ACCOMPLISH) trial38 randomized more than 11,000 high-risk patients with hypertension to receive an ACE inhibitor (benazepril) plus either a calcium channel blocker (amlodipine) or thiazide diuretic (hydrochlorothiazide). Blood pressures were identical between the two groups, but the trial was terminated early, at 36 months, because of a higher risk of the combined end point of cardiovascular death, myocardial infarction, stroke, and other major cardiac events in the ACE inhibitor-thiazide group.
Although some experts believe this study is definitive and indicates that high blood pressure should never be treated with an ACE inhibitor-thiazide combination, I believe that caution is needed in interpreting these findings. This regimen should be avoided in older patients with diabetes at high risk for cardiovascular disease, but otherwise, getting blood pressure under control is critical, and this combination can be used if it works and the patient is tolerating it well.
In summary, the choice of blood pressure-lowering medications is based on reducing cardiovascular events and slowing the progression of kidney disease. Either an ACE inhibitor or an ARB is the first choice for patients with diabetes, hypertension, and any degree of proteinuria. Many experts recommend beginning one of these agents even if proteinuria is not present. However, the combination of an ACE inhibitor and ARB should not be used in diabetic patients, especially if they have cardiovascular disease, until further data clarify the results of the ONTARGET and ALTITUDE trials.
STRATEGY 4: METABOLIC MANIPULATION WITH NOVEL AGENTS
Several new agents have recently been studied for the treatment of diabetic nephropathy, including aminoguanidine, which reduces levels of advanced glycation end-products, and sulodexide, which blocks basement membrane permeability. Neither agent has been shown to be safe and effective in diabetic nephropathy. The newest agent is bardoxolone methyl. It induces the Keap1–Nrf2 pathway, which up-regulates cytoprotective factors, suppressing inflammatory and other cytokines that are major mediators of progression of chronic kidney disease.39
Pergola et al,40 in a phase 2, double-blind trial, randomized 227 adults with diabetic kidney disease and a low estimated GFR (20–45 mL/min/1.73 m2) to receive placebo or bardoxolone 25, 75, or 150 mg daily. Drug treatment was associated with improvement in the estimated GFR, a finding that persisted throughout the 52 weeks of treatment. Surprisingly, proteinuria did not decrease with drug treatment.
As of this writing, a large multicenter controlled randomized trial has been halted because of concerns by the data safety monitoring board, which found increased rates of death and fluid retention with the drug. A number of recent trials have shown a beneficial effect of sodium bicarbonate therapy in patients with late-stage chronic kidney disease. They have shown slowing of the progression of GFR decline in a number of renal diseases, including diabetes.
- Ritz E, Orth SR. Nephropathy in patients with type 2 diabetes mellitus. N Engl J Med 1999; 341:1127–1133.
- de Boer IH, Rue TC, Hall YN, Heagerty PJ, Weiss NS, Himmelfarb J. Temporal trends in the prevalence of diabetic kidney disease in the United States. JAMA 2011; 305:2532–2539.
- United States Renal Data System (USRDS) 2000 Annual Data Report. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases – Division of Kidney, Urologic and Hematologic Diseases. USRDS Coordinating Center operated by the Minneapolis Medical Research Foundation. www.usrds.org
- Macisaac RJ, Jerums G. Diabetic kidney disease with and without albuminuria. Curr Opin Nephrol Hypertens 2011; 20:246–257.
- Molitch ME, DeFronzo RA, Franz MJ, et al; American Diabetes Association. Nephropathy in diabetes. Diabetes Care 2004; 27(suppl 1):S79–S83.
- Vamos EP, Harris M, Millett C, et al. Association of systolic and diastolic blood pressure and all cause mortality in people with newly diagnosed type 2 diabetes: retrospective cohort study. BMJ 2012; 345:e5567.
- de Zeeuw D, Remuzzi G, Parving HH, et al. Proteinuria, a target for renoprotection in patients with type 2 diabetic nephropathy: lessons from RENAAL. Kidney Int 2004; 65:2309–2320.
- Parving HH, Andersen AR, Smidt UM, Svendsen PA. Early aggressive antihypertensive treatment reduces rate of decline in kidney function in diabetic nephropathy. Lancet 1983; 1:1175–1179.
- ADVANCE Collaborative Group; Patel A, MacMahon S, Chalmers J, et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008; 358:2560–2572.
- ADVANCE Collaborative Group; Patel A, MacMahon S, Chalmers J, et al. Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): a randomised controlled trial. Lancet 2007; 370:829–840.
- Bangalore S, Kumar S, Lobach I, Messerli FH. Blood pressure targets in subjects with type 2 diabetes mellitus/impaired fasting glucose: observations from traditional and bayesian random-effects meta-analyses of randomized trials. Circulation 2011; 123:2799–2810.
- UK Prospective Diabetes Study Group. Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. BMJ 1998; 317:703–713.
- Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 2008; 359:1577–1589.
- Action to Control Cardiovascular Risk in Diabetes Study Group; Gerstein HC, Miller ME, Byington RP, et al. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008; 358:2545–2559.
- ACCORD Study Group; Cushman WC, Evans GW, Byington RP, et al. Effects of intensive blood-pressure control in type 2 diabetes mellitus. N Engl J Med 2010; 362:1575–1585.
- American Diabetes Association. Standards of medical care in diabetes—2012. Diabetes Care 2012; 35(suppl 1):S11–S63. (Erratum in: Diabetes Care 2012; 35:660.)
- Bakris GL, Williams M, Dworkin L, et al. Preserving renal function in adults with hypertension and diabetes: a consensus approach. National Kidney Foundation Hypertension and Diabetes Executive Committees Working Group. Am J Kidney Dis 2000; 36:646–661.
- Ramsay L, Williams B, Johnston G, et al. Guidelines for management of hypertension: report of the third working party of the British Hypertension Society. J Hum Hypertens 1999; 13:569–592.
- Feldman RD, Campbell N, Larochelle P, et al. 1999 Canadian recommendations for the management of hypertension. Task Force for the Development of the 1999 Canadian Recommendations for the Management of Hypertension. CMAJ 1999; 161(suppl):12:S1–S17.
- Chalmers J, MacMahon S, Mancia G, et al. 1999 World Health Organization-International Society of Hypertension Guidelines for the management of hypertension. Guidelines Sub-committee of the World Health Organization. Clin Exp Hypertens 1999; 21:1009–1060.
- The seventh report of the Joint National Committee on Prevention, Detection Evaluation, and Treatment of High Blood Pressure. Hypertension 2003; 42:1206–1252.
- The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med 1993; 329:977–986.
- Shichiri M, Kishikawa H, Ohkubo Y, Wake N. Long-term results of the Kumamoto Study on optimal diabetes control in type 2 diabetic patients. Diabetes Care 2000; 23(suppl 2):B21–B29.
- Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998; 352:837–853. Erratum in: Lancet 1999; 354:602.
- Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. UK Prospective Diabetes Study Group. BMJ 1998; 317:703–713. Erratum in: BMJ 1999; 318:29.
- Ismail-Beigi F, Craven T, Banerji MA, et al; ACCORD trial group. Effect of intensive treatment of hyperglycaemia on microvascular outcomes in type 2 diabetes: an analysis of the ACCORD randomised trial. Lancet 2010; 376:419–430. Erratum in: Lancet 2010; 376:1466.
- Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Heart Outcomes Prevention Evaluation Study Investigators. Lancet 2000: 355:253–259. Erratum in: Lancet2000; 356:860.
- Parving HH, Lehnert H, Bröchner-Mortensen J, et al; Irbesartan in Patients with Type 2 Diabetes and Microalbuminuria Study Group. The effect of irbesartan on the development of diabetic nephropathy in patients with type 2 diabetes. N Engl J Med 2001; 345:870–878.
- Viberti G, Wheeldon NM; MicroAlbuminuria Reduction With VALsartan (MARVAL) Study Investigators. Microalbuminuria reduction with valsartan in patients with type 2 diabetes mellitus: a blood pressure-independent effect. Circulation 2002; 106:672–678.
- Lewis EJ, Hunsicker LG, Bain R P, Rohde RD. The effect of angiotensin-converting-enzyme inhibition on diabetic nephropathy. The Collaborative Study Group. N Engl J Med 1993; 329:1456–1462.
- Lewis EJ, Hunsicker LG, Clarke WR, et al; Collaborative Study Group. Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med 2001; 345:851–860.
- Brenner BM, Cooper ME, de Zeeuw D, et al; RENAAL Study Investigators. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med 2001; 345:861–869.
- Biollaz J, Brunner HR, Gavras I, Waeber B, Gavras H. Antihypertensive therapy with MK 421: angiotensin II--renin relationships to evaluate efficacy of converting enzyme blockade. J Cardiovasc Pharmacol 1982; 4:966–972.
- Mogensen CE, Neldam S, Tikkanen I, et al. Randomised controlled trial of dual blockade of renin-angiotensin system in patients with hypertension, microalbuminuria, and non-insulin dependent diabetes: the candesartan and lisinopril microalbuminuria (CALM) study. BMJ 2000; 321:1440–1444.
- Epstein M, Williams GH, Weinberger M, et al. Selective aldosterone blockade with eplerenone reduces albuminuria in patients with type 2 diabetes. Clin J Am Soc Nephrol 2006; 1:940–951.
- Parving HH, Persson F, Lewis JB, Lewis EJ, Hollenberg NK; AVOID Study Investigators. Aliskiren combined with losartan in type 2 diabetes and nephropathy. N Engl J Med 2008; 358:2433–2446.
- Mann JF, Schmieder RE, McQueen M, et al; ONTARGET investigators. Renal outcomes with telmisartan, ramipril, or both, in people at high vascular risk (the ONTARGET study): a multicentre, randomised, double-blind, controlled trial. Lancet 2008; 372:547–553.
- Jamerson K, Weber MA, Bakris GL, et al; ACCOMPLISH Trial Investigators. Benazepril plus amlodipine or hydrochlorothiazide for hypertension in high-risk patients. N Engl J Med 2008; 359:2417–2428.
- Kim HJ, Vaziri ND. Contribution of impaired Nrf2-Keap1 pathway to oxidative stress and inflammation in chronic renal failure. Am J Physiol Renal Physiol 2010; 298:F662–F671.
- Pergola PE, Raskin P, Toto RD, et al; BEAM Study Investigators Bardoxolone methyl and kidney function in CKD with type 2 diabetes. N Engl J Med 2011; 365:327–336.
- Ritz E, Orth SR. Nephropathy in patients with type 2 diabetes mellitus. N Engl J Med 1999; 341:1127–1133.
- de Boer IH, Rue TC, Hall YN, Heagerty PJ, Weiss NS, Himmelfarb J. Temporal trends in the prevalence of diabetic kidney disease in the United States. JAMA 2011; 305:2532–2539.
- United States Renal Data System (USRDS) 2000 Annual Data Report. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases – Division of Kidney, Urologic and Hematologic Diseases. USRDS Coordinating Center operated by the Minneapolis Medical Research Foundation. www.usrds.org
- Macisaac RJ, Jerums G. Diabetic kidney disease with and without albuminuria. Curr Opin Nephrol Hypertens 2011; 20:246–257.
- Molitch ME, DeFronzo RA, Franz MJ, et al; American Diabetes Association. Nephropathy in diabetes. Diabetes Care 2004; 27(suppl 1):S79–S83.
- Vamos EP, Harris M, Millett C, et al. Association of systolic and diastolic blood pressure and all cause mortality in people with newly diagnosed type 2 diabetes: retrospective cohort study. BMJ 2012; 345:e5567.
- de Zeeuw D, Remuzzi G, Parving HH, et al. Proteinuria, a target for renoprotection in patients with type 2 diabetic nephropathy: lessons from RENAAL. Kidney Int 2004; 65:2309–2320.
- Parving HH, Andersen AR, Smidt UM, Svendsen PA. Early aggressive antihypertensive treatment reduces rate of decline in kidney function in diabetic nephropathy. Lancet 1983; 1:1175–1179.
- ADVANCE Collaborative Group; Patel A, MacMahon S, Chalmers J, et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008; 358:2560–2572.
- ADVANCE Collaborative Group; Patel A, MacMahon S, Chalmers J, et al. Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): a randomised controlled trial. Lancet 2007; 370:829–840.
- Bangalore S, Kumar S, Lobach I, Messerli FH. Blood pressure targets in subjects with type 2 diabetes mellitus/impaired fasting glucose: observations from traditional and bayesian random-effects meta-analyses of randomized trials. Circulation 2011; 123:2799–2810.
- UK Prospective Diabetes Study Group. Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. BMJ 1998; 317:703–713.
- Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 2008; 359:1577–1589.
- Action to Control Cardiovascular Risk in Diabetes Study Group; Gerstein HC, Miller ME, Byington RP, et al. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008; 358:2545–2559.
- ACCORD Study Group; Cushman WC, Evans GW, Byington RP, et al. Effects of intensive blood-pressure control in type 2 diabetes mellitus. N Engl J Med 2010; 362:1575–1585.
- American Diabetes Association. Standards of medical care in diabetes—2012. Diabetes Care 2012; 35(suppl 1):S11–S63. (Erratum in: Diabetes Care 2012; 35:660.)
- Bakris GL, Williams M, Dworkin L, et al. Preserving renal function in adults with hypertension and diabetes: a consensus approach. National Kidney Foundation Hypertension and Diabetes Executive Committees Working Group. Am J Kidney Dis 2000; 36:646–661.
- Ramsay L, Williams B, Johnston G, et al. Guidelines for management of hypertension: report of the third working party of the British Hypertension Society. J Hum Hypertens 1999; 13:569–592.
- Feldman RD, Campbell N, Larochelle P, et al. 1999 Canadian recommendations for the management of hypertension. Task Force for the Development of the 1999 Canadian Recommendations for the Management of Hypertension. CMAJ 1999; 161(suppl):12:S1–S17.
- Chalmers J, MacMahon S, Mancia G, et al. 1999 World Health Organization-International Society of Hypertension Guidelines for the management of hypertension. Guidelines Sub-committee of the World Health Organization. Clin Exp Hypertens 1999; 21:1009–1060.
- The seventh report of the Joint National Committee on Prevention, Detection Evaluation, and Treatment of High Blood Pressure. Hypertension 2003; 42:1206–1252.
- The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med 1993; 329:977–986.
- Shichiri M, Kishikawa H, Ohkubo Y, Wake N. Long-term results of the Kumamoto Study on optimal diabetes control in type 2 diabetic patients. Diabetes Care 2000; 23(suppl 2):B21–B29.
- Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998; 352:837–853. Erratum in: Lancet 1999; 354:602.
- Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. UK Prospective Diabetes Study Group. BMJ 1998; 317:703–713. Erratum in: BMJ 1999; 318:29.
- Ismail-Beigi F, Craven T, Banerji MA, et al; ACCORD trial group. Effect of intensive treatment of hyperglycaemia on microvascular outcomes in type 2 diabetes: an analysis of the ACCORD randomised trial. Lancet 2010; 376:419–430. Erratum in: Lancet 2010; 376:1466.
- Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Heart Outcomes Prevention Evaluation Study Investigators. Lancet 2000: 355:253–259. Erratum in: Lancet2000; 356:860.
- Parving HH, Lehnert H, Bröchner-Mortensen J, et al; Irbesartan in Patients with Type 2 Diabetes and Microalbuminuria Study Group. The effect of irbesartan on the development of diabetic nephropathy in patients with type 2 diabetes. N Engl J Med 2001; 345:870–878.
- Viberti G, Wheeldon NM; MicroAlbuminuria Reduction With VALsartan (MARVAL) Study Investigators. Microalbuminuria reduction with valsartan in patients with type 2 diabetes mellitus: a blood pressure-independent effect. Circulation 2002; 106:672–678.
- Lewis EJ, Hunsicker LG, Bain R P, Rohde RD. The effect of angiotensin-converting-enzyme inhibition on diabetic nephropathy. The Collaborative Study Group. N Engl J Med 1993; 329:1456–1462.
- Lewis EJ, Hunsicker LG, Clarke WR, et al; Collaborative Study Group. Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med 2001; 345:851–860.
- Brenner BM, Cooper ME, de Zeeuw D, et al; RENAAL Study Investigators. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med 2001; 345:861–869.
- Biollaz J, Brunner HR, Gavras I, Waeber B, Gavras H. Antihypertensive therapy with MK 421: angiotensin II--renin relationships to evaluate efficacy of converting enzyme blockade. J Cardiovasc Pharmacol 1982; 4:966–972.
- Mogensen CE, Neldam S, Tikkanen I, et al. Randomised controlled trial of dual blockade of renin-angiotensin system in patients with hypertension, microalbuminuria, and non-insulin dependent diabetes: the candesartan and lisinopril microalbuminuria (CALM) study. BMJ 2000; 321:1440–1444.
- Epstein M, Williams GH, Weinberger M, et al. Selective aldosterone blockade with eplerenone reduces albuminuria in patients with type 2 diabetes. Clin J Am Soc Nephrol 2006; 1:940–951.
- Parving HH, Persson F, Lewis JB, Lewis EJ, Hollenberg NK; AVOID Study Investigators. Aliskiren combined with losartan in type 2 diabetes and nephropathy. N Engl J Med 2008; 358:2433–2446.
- Mann JF, Schmieder RE, McQueen M, et al; ONTARGET investigators. Renal outcomes with telmisartan, ramipril, or both, in people at high vascular risk (the ONTARGET study): a multicentre, randomised, double-blind, controlled trial. Lancet 2008; 372:547–553.
- Jamerson K, Weber MA, Bakris GL, et al; ACCOMPLISH Trial Investigators. Benazepril plus amlodipine or hydrochlorothiazide for hypertension in high-risk patients. N Engl J Med 2008; 359:2417–2428.
- Kim HJ, Vaziri ND. Contribution of impaired Nrf2-Keap1 pathway to oxidative stress and inflammation in chronic renal failure. Am J Physiol Renal Physiol 2010; 298:F662–F671.
- Pergola PE, Raskin P, Toto RD, et al; BEAM Study Investigators Bardoxolone methyl and kidney function in CKD with type 2 diabetes. N Engl J Med 2011; 365:327–336.
KEY POINTS
- The progression from no proteinuria to microalbuminuria to clinical proteinuria parallels glomerular changes of thickening of the basement membrane, mesangial expansion, and the development of Kimmelstiel-Wilson nodules and sclerosis.
- Blood pressure control to 130/80 mm Hg slows microvascular and macrovascular disease, but the goal should not be lower in older patients with diabetes.
- Glycemic control slows microvascular disease: the goal for most patients for hemoglobin A1c is 7.0%. Tighter control may increase cardiovascular risk.
- Either an angiotensin-converting enzyme inhibitor or an angiotensin receptor blocker is the first-line treatment for diabetic nephropathy; combining the two is no longer recommended.
- If more aggressive treatment is needed, a diuretic or spironolactone (with potassium monitoring) can be added.
- The role of sodium bicarbonate and new agents such as blockers of transcription factors is still emerging.