User login
Neuro-politics: Will you vote with your cortex or limbic system?
It’s election season again. Every 4 years, October becomes the purgatory month of politics. But this year, it’s even more complicated, being juxtaposed against a chaotic mosaic of a viral pandemic, economic travails, social upheaval, and exceptionally toxic political hyperpartisanship.
The widespread expectation is that citizens will vote for their party’s candidates, but there is now a body of evidence suggesting that our brains may be pre-wired to be liberal or conservative.
Enter neuro-politics. This discipline is younger than neuro-economics, neuro-law, neuro-ethics, neuro-marketing, neuro-art, neuro-culture, or neuro-esthetics. Neuro-politics focuses on the intersection of politics with neuroscience.1 However, there are many antecedents to neuro-politics reflected in the writings of Plato, Aristotle, Niccolò Machiavelli, John Locke, Baruch Spinoza, Henri Bergson, William James, and others.
Neuro-politics attempts to generate data to answer a variety of questions about political behavior, such as:
- Is political orientation associated with differences in certain brain regions?
- Are there reliable neural biomarkers of political orientation?
- Is political orientation modifiable, and if so, why are some individuals ferociously entrenched to one political dogma while others are able to untether themselves and adopt another political doctrine?
- What are the brain characteristics of “swing voters” who may align themselves with different parties in different election cycles?
- Is there a “religification” of politics among the ardent fanatics who regard the tenets of their political beliefs as “articles of faith?”
- Is the brain modified by certain attributes (such as educational level, age, sex, marital status, race, ethnicity, and religious affiliation) that translate to political decision-making?
- Can neuro-politics explain the sprouting of psychiatric symptoms such as obsessions, anxiety, irritability, anger, hatred, and conspiracy theories?
- Is political extremism driven by cortical structures, limbic structures, or both?
Politics and the brain
Here is a brief review of some studies that examined the relationship of political orientation or voting behavior with brain structure and function:
1. Roger Sperry, the 1981 Nobel Laureate (for his studies on split-brain patients) reported that in patients who underwent callosotomy, both cerebral hemispheres gave the same ratings of politicians when their photos were shown to each hemisphere separately.2
2. A functional magnetic resonance imaging (fMRI) study found that the faces of candidates activated participants’ ventromedial and anterior prefrontal cortices. Amygdala activation was associated with the intensity of the emotion.3
Continue to: A skin conductance...
3. A skin conductance study reported that politically liberal individuals had low reactivity to sudden noises and threatening stimuli, while conservative counterparts demonstrated high physiological reactions to noises and stimuli.4
4. Images of a losing candidate elicited greater activation on fMRI in the insula and ventral anterior cingulate compared to no activation by exposure to an image of the winning candidate.5
5. Another fMRI study found that “individualism” was associated with activation of the medial prefrontal cortex and temporo-parietal junction when participants listened to a set of political statements. On the other hand, “conservatism” activated the dorsolateral prefrontal cortex, while “radicalism” activated the ventral striatum and posterior cingulate.6
6. An EEG activity study of healthy individuals revealed desynchronization in the alpha band related to the politicians who lost simulated elections and were judged as “less trustworthy” when the participant watched their faces.7
7. A structural MRI study of young adults reported that liberalism was associated with increased gray matter volume in the anterior cingulate, while conservatism was associated with increased volume of the right amygdala. The authors replicated their findings and concluded there is a possible link between brain structure and psychological mechanisms that mediate political attitudes.8
Continue to: To examine the effect of...
8. To examine the effect of a “first impression” based on the physical appearance of candidates, researchers compared individuals with damage to the lateral orbitofrontal cortex (OFC) with a group that had frontal damage that spared the lateral OFC and another group of matched healthy volunteers. They used a simulated elections paradigm in which participants voted based solely on photographs of the candidates’ faces. Only the group with OFC damage was influenced by attractiveness, while those with an intact frontal lobe or non-OFC frontal damage relied on other data, such as competence.9 These researchers concluded that an intact OFC is necessary for political decision-making.
9. A study using cognitive tasks reported that liberals are more adept at dealing with novel information than conservatives.10
What part of your brain will you use?
Regardless of the data generated by the neuro-politics studies, the bottom line is: What part of your brain do you use when you cast your vote for an issue, a representative, a senator, or a president? Is it a purely intellectual decision (ie, cortical), or is it driven by visceral emotions (ie, limbic)? Do you believe that every single item in your party’s platform is right and virtuous, while every item in the other party’s platform is wrong and evil? Can you think of any redeeming feature of the candidate you hate or the party you despise?
One attribute that we psychiatrists possess by virtue of our training and clinical work is that we are able to transcend dichotomies and to perceive nuances and shades of gray about controversial issues. So I hope we employ the circuits of our brain where wisdom putatively resides11 and which may develop further (via neuroplasticity) with the conduct of psychotherapy.12 Those brain circuits include:
- prefrontal cortex (for emotional regulation, decision-making, and value relativism)
- lateral prefrontal cortex (to facilitate calculated, reason-based decision-making)
- medial prefrontal cortex (for emotional valence and pro-social attitudes and behaviors).
However, being human, it is quite likely that our amygdala may “seep through” and color our judgment and decisions. But let us try to cast a vote that is not only good for the country but also good for our patients, many of whom may not even be able to vote. Election season is a time to make a positive difference in our patients’ lives, not just ours. Let’s hope our brains exploit this unique opportunity.
1. Schreiber D. Neuropolitics: twenty years later. Politics Life Sci. 2017;36(2):114-131.
2. Sperry RW, Zaidel E, Zaidel D. Self recognition and social awareness in the deconnected minor hemisphere. Neuropsychologia. 1979;17(2):153-166.
3. Knutson KM, Wood JN, Spampinato MV, et al. Politics on the brain: an FMRI investigation. Soc Neurosci. 2006;1(1):25-40.
4. Oxley DR, Smith KB, Alford JR, et al. Political attitudes vary with physiological traits. Science. 2008;321(5896):1667-1670.
5. Spezio ML, Rangel A, Alvarez RM, et al. A neural basis for the effect of candidate appearance on election outcomes. Soc Cogn Affect Neurosci. 2008;3(4):344-352.
6. Zamboni G, Gozzi M, Krueger F, et al. Individualism, conservatism, and radicalism as criteria for processing political beliefs: a parametric fMRI study. Soc Neurosci. 2009;4(5):367-383.
7. Vecchiato G, Toppi J, Cincotti F, et al. Neuropolitics: EEG spectral maps related to a political vote based on the first impression of the candidate’s face. Conf Proc IEEE Eng Med Biol Soc. 2010;2010:2902-2905.
8. Kanai R, Feilden T, Firth C, et al. Political orientations are correlated with brain structure in young adults. Curr Biol. 2011;21(8):677-680.
9. Xia C, Stolle D, Gidengil E, et al. Lateral orbitofrontal cortex links social impressions to political choices. J Neurosci. 2015;35(22):8507-8514.
10. Bernabel RT, Oliveira A. Conservatism and liberalism predict performance in two nonideological cognitive tasks. Politics Life Sci. 2017;36(2):49-59.
11. Meeks TW, Jeste DV. Neurobiology of wisdom: a literature overview. Arch Gen Psychiatry. 2009;66(4):355-365.
12. Nasrallah HA. Does psychiatric practice make us wiser? Current Psychiatry. 2009;8(10):12,14.
It’s election season again. Every 4 years, October becomes the purgatory month of politics. But this year, it’s even more complicated, being juxtaposed against a chaotic mosaic of a viral pandemic, economic travails, social upheaval, and exceptionally toxic political hyperpartisanship.
The widespread expectation is that citizens will vote for their party’s candidates, but there is now a body of evidence suggesting that our brains may be pre-wired to be liberal or conservative.
Enter neuro-politics. This discipline is younger than neuro-economics, neuro-law, neuro-ethics, neuro-marketing, neuro-art, neuro-culture, or neuro-esthetics. Neuro-politics focuses on the intersection of politics with neuroscience.1 However, there are many antecedents to neuro-politics reflected in the writings of Plato, Aristotle, Niccolò Machiavelli, John Locke, Baruch Spinoza, Henri Bergson, William James, and others.
Neuro-politics attempts to generate data to answer a variety of questions about political behavior, such as:
- Is political orientation associated with differences in certain brain regions?
- Are there reliable neural biomarkers of political orientation?
- Is political orientation modifiable, and if so, why are some individuals ferociously entrenched to one political dogma while others are able to untether themselves and adopt another political doctrine?
- What are the brain characteristics of “swing voters” who may align themselves with different parties in different election cycles?
- Is there a “religification” of politics among the ardent fanatics who regard the tenets of their political beliefs as “articles of faith?”
- Is the brain modified by certain attributes (such as educational level, age, sex, marital status, race, ethnicity, and religious affiliation) that translate to political decision-making?
- Can neuro-politics explain the sprouting of psychiatric symptoms such as obsessions, anxiety, irritability, anger, hatred, and conspiracy theories?
- Is political extremism driven by cortical structures, limbic structures, or both?
Politics and the brain
Here is a brief review of some studies that examined the relationship of political orientation or voting behavior with brain structure and function:
1. Roger Sperry, the 1981 Nobel Laureate (for his studies on split-brain patients) reported that in patients who underwent callosotomy, both cerebral hemispheres gave the same ratings of politicians when their photos were shown to each hemisphere separately.2
2. A functional magnetic resonance imaging (fMRI) study found that the faces of candidates activated participants’ ventromedial and anterior prefrontal cortices. Amygdala activation was associated with the intensity of the emotion.3
Continue to: A skin conductance...
3. A skin conductance study reported that politically liberal individuals had low reactivity to sudden noises and threatening stimuli, while conservative counterparts demonstrated high physiological reactions to noises and stimuli.4
4. Images of a losing candidate elicited greater activation on fMRI in the insula and ventral anterior cingulate compared to no activation by exposure to an image of the winning candidate.5
5. Another fMRI study found that “individualism” was associated with activation of the medial prefrontal cortex and temporo-parietal junction when participants listened to a set of political statements. On the other hand, “conservatism” activated the dorsolateral prefrontal cortex, while “radicalism” activated the ventral striatum and posterior cingulate.6
6. An EEG activity study of healthy individuals revealed desynchronization in the alpha band related to the politicians who lost simulated elections and were judged as “less trustworthy” when the participant watched their faces.7
7. A structural MRI study of young adults reported that liberalism was associated with increased gray matter volume in the anterior cingulate, while conservatism was associated with increased volume of the right amygdala. The authors replicated their findings and concluded there is a possible link between brain structure and psychological mechanisms that mediate political attitudes.8
Continue to: To examine the effect of...
8. To examine the effect of a “first impression” based on the physical appearance of candidates, researchers compared individuals with damage to the lateral orbitofrontal cortex (OFC) with a group that had frontal damage that spared the lateral OFC and another group of matched healthy volunteers. They used a simulated elections paradigm in which participants voted based solely on photographs of the candidates’ faces. Only the group with OFC damage was influenced by attractiveness, while those with an intact frontal lobe or non-OFC frontal damage relied on other data, such as competence.9 These researchers concluded that an intact OFC is necessary for political decision-making.
9. A study using cognitive tasks reported that liberals are more adept at dealing with novel information than conservatives.10
What part of your brain will you use?
Regardless of the data generated by the neuro-politics studies, the bottom line is: What part of your brain do you use when you cast your vote for an issue, a representative, a senator, or a president? Is it a purely intellectual decision (ie, cortical), or is it driven by visceral emotions (ie, limbic)? Do you believe that every single item in your party’s platform is right and virtuous, while every item in the other party’s platform is wrong and evil? Can you think of any redeeming feature of the candidate you hate or the party you despise?
One attribute that we psychiatrists possess by virtue of our training and clinical work is that we are able to transcend dichotomies and to perceive nuances and shades of gray about controversial issues. So I hope we employ the circuits of our brain where wisdom putatively resides11 and which may develop further (via neuroplasticity) with the conduct of psychotherapy.12 Those brain circuits include:
- prefrontal cortex (for emotional regulation, decision-making, and value relativism)
- lateral prefrontal cortex (to facilitate calculated, reason-based decision-making)
- medial prefrontal cortex (for emotional valence and pro-social attitudes and behaviors).
However, being human, it is quite likely that our amygdala may “seep through” and color our judgment and decisions. But let us try to cast a vote that is not only good for the country but also good for our patients, many of whom may not even be able to vote. Election season is a time to make a positive difference in our patients’ lives, not just ours. Let’s hope our brains exploit this unique opportunity.
It’s election season again. Every 4 years, October becomes the purgatory month of politics. But this year, it’s even more complicated, being juxtaposed against a chaotic mosaic of a viral pandemic, economic travails, social upheaval, and exceptionally toxic political hyperpartisanship.
The widespread expectation is that citizens will vote for their party’s candidates, but there is now a body of evidence suggesting that our brains may be pre-wired to be liberal or conservative.
Enter neuro-politics. This discipline is younger than neuro-economics, neuro-law, neuro-ethics, neuro-marketing, neuro-art, neuro-culture, or neuro-esthetics. Neuro-politics focuses on the intersection of politics with neuroscience.1 However, there are many antecedents to neuro-politics reflected in the writings of Plato, Aristotle, Niccolò Machiavelli, John Locke, Baruch Spinoza, Henri Bergson, William James, and others.
Neuro-politics attempts to generate data to answer a variety of questions about political behavior, such as:
- Is political orientation associated with differences in certain brain regions?
- Are there reliable neural biomarkers of political orientation?
- Is political orientation modifiable, and if so, why are some individuals ferociously entrenched to one political dogma while others are able to untether themselves and adopt another political doctrine?
- What are the brain characteristics of “swing voters” who may align themselves with different parties in different election cycles?
- Is there a “religification” of politics among the ardent fanatics who regard the tenets of their political beliefs as “articles of faith?”
- Is the brain modified by certain attributes (such as educational level, age, sex, marital status, race, ethnicity, and religious affiliation) that translate to political decision-making?
- Can neuro-politics explain the sprouting of psychiatric symptoms such as obsessions, anxiety, irritability, anger, hatred, and conspiracy theories?
- Is political extremism driven by cortical structures, limbic structures, or both?
Politics and the brain
Here is a brief review of some studies that examined the relationship of political orientation or voting behavior with brain structure and function:
1. Roger Sperry, the 1981 Nobel Laureate (for his studies on split-brain patients) reported that in patients who underwent callosotomy, both cerebral hemispheres gave the same ratings of politicians when their photos were shown to each hemisphere separately.2
2. A functional magnetic resonance imaging (fMRI) study found that the faces of candidates activated participants’ ventromedial and anterior prefrontal cortices. Amygdala activation was associated with the intensity of the emotion.3
Continue to: A skin conductance...
3. A skin conductance study reported that politically liberal individuals had low reactivity to sudden noises and threatening stimuli, while conservative counterparts demonstrated high physiological reactions to noises and stimuli.4
4. Images of a losing candidate elicited greater activation on fMRI in the insula and ventral anterior cingulate compared to no activation by exposure to an image of the winning candidate.5
5. Another fMRI study found that “individualism” was associated with activation of the medial prefrontal cortex and temporo-parietal junction when participants listened to a set of political statements. On the other hand, “conservatism” activated the dorsolateral prefrontal cortex, while “radicalism” activated the ventral striatum and posterior cingulate.6
6. An EEG activity study of healthy individuals revealed desynchronization in the alpha band related to the politicians who lost simulated elections and were judged as “less trustworthy” when the participant watched their faces.7
7. A structural MRI study of young adults reported that liberalism was associated with increased gray matter volume in the anterior cingulate, while conservatism was associated with increased volume of the right amygdala. The authors replicated their findings and concluded there is a possible link between brain structure and psychological mechanisms that mediate political attitudes.8
Continue to: To examine the effect of...
8. To examine the effect of a “first impression” based on the physical appearance of candidates, researchers compared individuals with damage to the lateral orbitofrontal cortex (OFC) with a group that had frontal damage that spared the lateral OFC and another group of matched healthy volunteers. They used a simulated elections paradigm in which participants voted based solely on photographs of the candidates’ faces. Only the group with OFC damage was influenced by attractiveness, while those with an intact frontal lobe or non-OFC frontal damage relied on other data, such as competence.9 These researchers concluded that an intact OFC is necessary for political decision-making.
9. A study using cognitive tasks reported that liberals are more adept at dealing with novel information than conservatives.10
What part of your brain will you use?
Regardless of the data generated by the neuro-politics studies, the bottom line is: What part of your brain do you use when you cast your vote for an issue, a representative, a senator, or a president? Is it a purely intellectual decision (ie, cortical), or is it driven by visceral emotions (ie, limbic)? Do you believe that every single item in your party’s platform is right and virtuous, while every item in the other party’s platform is wrong and evil? Can you think of any redeeming feature of the candidate you hate or the party you despise?
One attribute that we psychiatrists possess by virtue of our training and clinical work is that we are able to transcend dichotomies and to perceive nuances and shades of gray about controversial issues. So I hope we employ the circuits of our brain where wisdom putatively resides11 and which may develop further (via neuroplasticity) with the conduct of psychotherapy.12 Those brain circuits include:
- prefrontal cortex (for emotional regulation, decision-making, and value relativism)
- lateral prefrontal cortex (to facilitate calculated, reason-based decision-making)
- medial prefrontal cortex (for emotional valence and pro-social attitudes and behaviors).
However, being human, it is quite likely that our amygdala may “seep through” and color our judgment and decisions. But let us try to cast a vote that is not only good for the country but also good for our patients, many of whom may not even be able to vote. Election season is a time to make a positive difference in our patients’ lives, not just ours. Let’s hope our brains exploit this unique opportunity.
1. Schreiber D. Neuropolitics: twenty years later. Politics Life Sci. 2017;36(2):114-131.
2. Sperry RW, Zaidel E, Zaidel D. Self recognition and social awareness in the deconnected minor hemisphere. Neuropsychologia. 1979;17(2):153-166.
3. Knutson KM, Wood JN, Spampinato MV, et al. Politics on the brain: an FMRI investigation. Soc Neurosci. 2006;1(1):25-40.
4. Oxley DR, Smith KB, Alford JR, et al. Political attitudes vary with physiological traits. Science. 2008;321(5896):1667-1670.
5. Spezio ML, Rangel A, Alvarez RM, et al. A neural basis for the effect of candidate appearance on election outcomes. Soc Cogn Affect Neurosci. 2008;3(4):344-352.
6. Zamboni G, Gozzi M, Krueger F, et al. Individualism, conservatism, and radicalism as criteria for processing political beliefs: a parametric fMRI study. Soc Neurosci. 2009;4(5):367-383.
7. Vecchiato G, Toppi J, Cincotti F, et al. Neuropolitics: EEG spectral maps related to a political vote based on the first impression of the candidate’s face. Conf Proc IEEE Eng Med Biol Soc. 2010;2010:2902-2905.
8. Kanai R, Feilden T, Firth C, et al. Political orientations are correlated with brain structure in young adults. Curr Biol. 2011;21(8):677-680.
9. Xia C, Stolle D, Gidengil E, et al. Lateral orbitofrontal cortex links social impressions to political choices. J Neurosci. 2015;35(22):8507-8514.
10. Bernabel RT, Oliveira A. Conservatism and liberalism predict performance in two nonideological cognitive tasks. Politics Life Sci. 2017;36(2):49-59.
11. Meeks TW, Jeste DV. Neurobiology of wisdom: a literature overview. Arch Gen Psychiatry. 2009;66(4):355-365.
12. Nasrallah HA. Does psychiatric practice make us wiser? Current Psychiatry. 2009;8(10):12,14.
1. Schreiber D. Neuropolitics: twenty years later. Politics Life Sci. 2017;36(2):114-131.
2. Sperry RW, Zaidel E, Zaidel D. Self recognition and social awareness in the deconnected minor hemisphere. Neuropsychologia. 1979;17(2):153-166.
3. Knutson KM, Wood JN, Spampinato MV, et al. Politics on the brain: an FMRI investigation. Soc Neurosci. 2006;1(1):25-40.
4. Oxley DR, Smith KB, Alford JR, et al. Political attitudes vary with physiological traits. Science. 2008;321(5896):1667-1670.
5. Spezio ML, Rangel A, Alvarez RM, et al. A neural basis for the effect of candidate appearance on election outcomes. Soc Cogn Affect Neurosci. 2008;3(4):344-352.
6. Zamboni G, Gozzi M, Krueger F, et al. Individualism, conservatism, and radicalism as criteria for processing political beliefs: a parametric fMRI study. Soc Neurosci. 2009;4(5):367-383.
7. Vecchiato G, Toppi J, Cincotti F, et al. Neuropolitics: EEG spectral maps related to a political vote based on the first impression of the candidate’s face. Conf Proc IEEE Eng Med Biol Soc. 2010;2010:2902-2905.
8. Kanai R, Feilden T, Firth C, et al. Political orientations are correlated with brain structure in young adults. Curr Biol. 2011;21(8):677-680.
9. Xia C, Stolle D, Gidengil E, et al. Lateral orbitofrontal cortex links social impressions to political choices. J Neurosci. 2015;35(22):8507-8514.
10. Bernabel RT, Oliveira A. Conservatism and liberalism predict performance in two nonideological cognitive tasks. Politics Life Sci. 2017;36(2):49-59.
11. Meeks TW, Jeste DV. Neurobiology of wisdom: a literature overview. Arch Gen Psychiatry. 2009;66(4):355-365.
12. Nasrallah HA. Does psychiatric practice make us wiser? Current Psychiatry. 2009;8(10):12,14.
OTC ‘brain boosters’ may pose serious risks, experts say
, new research shows.
“Americans spend more than $600 million on over-the-counter smart pills every year, but we know very little about what is actually in these products,” said Pieter A. Cohen, MD, of the department of medicine at Harvard Medical School, Boston.
“Finding new combinations of drugs [that have] never been tested in humans in over-the-counter brain-boosting supplements is alarming,” said Dr. Cohen.
The study was published online Sept. 23 in Neurology Clinical Practice, a journal of the American Academy of Neurology.
Buyer beware
In a search of the National Institutes of Health Dietary Supplement Label Database and the Natural Medicines Database, Dr. Cohen and colleagues identified 10 supplements labeled as containing omberacetam, aniracetam, phenylpiracetam, or oxiracetam – four analogues of piracetam that are not approved for human use in the United States. Piracetam is also not approved in the United States.
In these 10 products, five unapproved drugs were discovered – omberacetam and aniracetam along with three others (phenibut, vinpocetine and picamilon).
By consuming the recommended serving size of these products, consumers could be exposed to pharmaceutical-level dosages of drugs including a maximum of 40.6 mg omberacetam (typical pharmacologic dose 10 mg), 502 mg of aniracetam (typical pharmacologic dose 200-750 mg), 15.4 mg of phenibut (typical dose 250-500 mg), 4.3 mg of vinpocetine (typical dose 5-40 mg), and 90.1 mg of picamilon (typical dose 50-200 mg), the study team reported.
Several drugs detected in these “smart” pills were not declared on the label, and several declared drugs were not detected in the products. For those products with drug quantities provided on the labels, three-quarters of declared quantities were inaccurate.
Consumers who use these cognitive enhancers could be exposed to amounts of these unapproved drugs that are fourfold greater than pharmaceutical dosages and combinations never tested in humans, the study team says. One product combined three different unapproved drugs and another product contained four different drugs.
“We have previously shown that these products may contain individual foreign drugs, but in our new study we found complex combinations of foreign drugs, up to four different drugs in a single product,” Dr. Cohen said.
The presence of these unapproved drugs in supplements, including at supratherapeutic dosages, suggests “serious risks to consumers and weaknesses in the regulatory framework under which supplements are permitted to be introduced in the U.S.,” Dr. Cohen and colleagues wrote.
“We should counsel our patients to avoid over-the-counter ‘smart pills’ until we can be assured as to the safety and efficacy of these products,” said Dr. Cohen.
Concerning findings
Glen R. Finney, MD, director of the Geisinger Memory and Cognition Program at the Neuroscience Institute, Geisinger Health System, Wilkes-Barre, Penn., said in an interview that two findings are very concerning: the lack of listed ingredients and especially the presence of unlisted drugs at active levels. “What if a person has a sensitivity or allergy to one of the unlisted drugs? This is a safety issue and a consumer issue,” Dr. Finney said.
Despite being widely promoted on television, “over-the-counter supplements are not regulated, so there is no guarantee that they contain what they claim, and there is very little evidence that they help memory and thinking even when they do have the ingredients they claim in the supplement,” said Dr. Finney,
“The best way to stay safe and help memory and thinking is to speak with your health providers about proven treatments that have good safety regulation, so you know what you’re getting, and what you’re getting from it,” Dr. Finney advised.
The study had no targeted funding. Dr. Cohen has collaborated in research with NSF International, received compensation from UptoDate, and received research support from Consumers Union and PEW Charitable Trusts. Dr. Finney has no relevant disclosures.
A version of this article originally appeared on Medscape.com.
, new research shows.
“Americans spend more than $600 million on over-the-counter smart pills every year, but we know very little about what is actually in these products,” said Pieter A. Cohen, MD, of the department of medicine at Harvard Medical School, Boston.
“Finding new combinations of drugs [that have] never been tested in humans in over-the-counter brain-boosting supplements is alarming,” said Dr. Cohen.
The study was published online Sept. 23 in Neurology Clinical Practice, a journal of the American Academy of Neurology.
Buyer beware
In a search of the National Institutes of Health Dietary Supplement Label Database and the Natural Medicines Database, Dr. Cohen and colleagues identified 10 supplements labeled as containing omberacetam, aniracetam, phenylpiracetam, or oxiracetam – four analogues of piracetam that are not approved for human use in the United States. Piracetam is also not approved in the United States.
In these 10 products, five unapproved drugs were discovered – omberacetam and aniracetam along with three others (phenibut, vinpocetine and picamilon).
By consuming the recommended serving size of these products, consumers could be exposed to pharmaceutical-level dosages of drugs including a maximum of 40.6 mg omberacetam (typical pharmacologic dose 10 mg), 502 mg of aniracetam (typical pharmacologic dose 200-750 mg), 15.4 mg of phenibut (typical dose 250-500 mg), 4.3 mg of vinpocetine (typical dose 5-40 mg), and 90.1 mg of picamilon (typical dose 50-200 mg), the study team reported.
Several drugs detected in these “smart” pills were not declared on the label, and several declared drugs were not detected in the products. For those products with drug quantities provided on the labels, three-quarters of declared quantities were inaccurate.
Consumers who use these cognitive enhancers could be exposed to amounts of these unapproved drugs that are fourfold greater than pharmaceutical dosages and combinations never tested in humans, the study team says. One product combined three different unapproved drugs and another product contained four different drugs.
“We have previously shown that these products may contain individual foreign drugs, but in our new study we found complex combinations of foreign drugs, up to four different drugs in a single product,” Dr. Cohen said.
The presence of these unapproved drugs in supplements, including at supratherapeutic dosages, suggests “serious risks to consumers and weaknesses in the regulatory framework under which supplements are permitted to be introduced in the U.S.,” Dr. Cohen and colleagues wrote.
“We should counsel our patients to avoid over-the-counter ‘smart pills’ until we can be assured as to the safety and efficacy of these products,” said Dr. Cohen.
Concerning findings
Glen R. Finney, MD, director of the Geisinger Memory and Cognition Program at the Neuroscience Institute, Geisinger Health System, Wilkes-Barre, Penn., said in an interview that two findings are very concerning: the lack of listed ingredients and especially the presence of unlisted drugs at active levels. “What if a person has a sensitivity or allergy to one of the unlisted drugs? This is a safety issue and a consumer issue,” Dr. Finney said.
Despite being widely promoted on television, “over-the-counter supplements are not regulated, so there is no guarantee that they contain what they claim, and there is very little evidence that they help memory and thinking even when they do have the ingredients they claim in the supplement,” said Dr. Finney,
“The best way to stay safe and help memory and thinking is to speak with your health providers about proven treatments that have good safety regulation, so you know what you’re getting, and what you’re getting from it,” Dr. Finney advised.
The study had no targeted funding. Dr. Cohen has collaborated in research with NSF International, received compensation from UptoDate, and received research support from Consumers Union and PEW Charitable Trusts. Dr. Finney has no relevant disclosures.
A version of this article originally appeared on Medscape.com.
, new research shows.
“Americans spend more than $600 million on over-the-counter smart pills every year, but we know very little about what is actually in these products,” said Pieter A. Cohen, MD, of the department of medicine at Harvard Medical School, Boston.
“Finding new combinations of drugs [that have] never been tested in humans in over-the-counter brain-boosting supplements is alarming,” said Dr. Cohen.
The study was published online Sept. 23 in Neurology Clinical Practice, a journal of the American Academy of Neurology.
Buyer beware
In a search of the National Institutes of Health Dietary Supplement Label Database and the Natural Medicines Database, Dr. Cohen and colleagues identified 10 supplements labeled as containing omberacetam, aniracetam, phenylpiracetam, or oxiracetam – four analogues of piracetam that are not approved for human use in the United States. Piracetam is also not approved in the United States.
In these 10 products, five unapproved drugs were discovered – omberacetam and aniracetam along with three others (phenibut, vinpocetine and picamilon).
By consuming the recommended serving size of these products, consumers could be exposed to pharmaceutical-level dosages of drugs including a maximum of 40.6 mg omberacetam (typical pharmacologic dose 10 mg), 502 mg of aniracetam (typical pharmacologic dose 200-750 mg), 15.4 mg of phenibut (typical dose 250-500 mg), 4.3 mg of vinpocetine (typical dose 5-40 mg), and 90.1 mg of picamilon (typical dose 50-200 mg), the study team reported.
Several drugs detected in these “smart” pills were not declared on the label, and several declared drugs were not detected in the products. For those products with drug quantities provided on the labels, three-quarters of declared quantities were inaccurate.
Consumers who use these cognitive enhancers could be exposed to amounts of these unapproved drugs that are fourfold greater than pharmaceutical dosages and combinations never tested in humans, the study team says. One product combined three different unapproved drugs and another product contained four different drugs.
“We have previously shown that these products may contain individual foreign drugs, but in our new study we found complex combinations of foreign drugs, up to four different drugs in a single product,” Dr. Cohen said.
The presence of these unapproved drugs in supplements, including at supratherapeutic dosages, suggests “serious risks to consumers and weaknesses in the regulatory framework under which supplements are permitted to be introduced in the U.S.,” Dr. Cohen and colleagues wrote.
“We should counsel our patients to avoid over-the-counter ‘smart pills’ until we can be assured as to the safety and efficacy of these products,” said Dr. Cohen.
Concerning findings
Glen R. Finney, MD, director of the Geisinger Memory and Cognition Program at the Neuroscience Institute, Geisinger Health System, Wilkes-Barre, Penn., said in an interview that two findings are very concerning: the lack of listed ingredients and especially the presence of unlisted drugs at active levels. “What if a person has a sensitivity or allergy to one of the unlisted drugs? This is a safety issue and a consumer issue,” Dr. Finney said.
Despite being widely promoted on television, “over-the-counter supplements are not regulated, so there is no guarantee that they contain what they claim, and there is very little evidence that they help memory and thinking even when they do have the ingredients they claim in the supplement,” said Dr. Finney,
“The best way to stay safe and help memory and thinking is to speak with your health providers about proven treatments that have good safety regulation, so you know what you’re getting, and what you’re getting from it,” Dr. Finney advised.
The study had no targeted funding. Dr. Cohen has collaborated in research with NSF International, received compensation from UptoDate, and received research support from Consumers Union and PEW Charitable Trusts. Dr. Finney has no relevant disclosures.
A version of this article originally appeared on Medscape.com.
FROM NEUROLOGY CLINICAL PRACTICE
Vascular dementia risk particularly high in type 2 diabetes
Persons with type 2 diabetes may be at heightened risk for developing vascular dementia than other types of dementia, a team of international researchers has found.
Compared with a nondiabetic control population, those with type 2 diabetes had a statistically significant 35% increased chance of having vascular dementia in a large observational study.
By comparison, the risk for nonvascular dementia was increased by a “more modest” 8%, said the researchers from the University of Glasgow and the University of Gothenburg (Sweden), while the risk for Alzheimer’s dementia appeared to be reduced by 8%.
The link between type 2 diabetes and dementia is not new, observed Carlos Celis-Morales, PhD, who presented the study’s findings at the virtual annual meeting of the European Association for the Study of Diabetes. With people living longer thanks to improved preventative strategies and treatments, there is a risk for developing other chronic conditions, such as dementia.
“A third of all dementia cases may be attributable to modifiable risk factors, among them type 2 diabetes, which accounts for 3.2% of all dementia cases,” said Dr. Celis-Morales, a research fellow at the University of Glasgow’s Institute of Cardiovascular and Medical Sciences.
“Although we know that diabetes is linked to dementia, what we don’t know really well is how much of this association between diabetes and dementia outcomes are explained by modifiable and nonmodifiable risk factors,” Dr. Celis-Morales added.
“Diabetes and dementia share certain risk factors,” commented coinvestigator Naveed Sattar, MD, in a press release issued by the EASD. These include obesity, smoking, and lack of physical activity and might explain part of the association between the two conditions.
Dr. Sattar said that the heightened vascular dementia risk found in the study was “in itself an argument for preventive measures such as healthier lifestyle,” adding that “the importance of prevention is underscored by the fact that, for the majority of dementia diseases, there is no good treatment.”
Using data from the Swedish National Diabetes Register, the research team set out to determine the extent to which type 2 diabetes was associated with dementia and the incidence of different subtypes of dementia. They also looked to see if there were any associations with blood glucose control and what risk factors may be involved.
In total, data on 378,299 individuals with type 2 diabetes were compared with data on 1,886,022 similarly aged (average, 64 years) and gender-matched controls from the general population.
After a mean 7 years of follow-up, 10,143 people with and 46,479 people without type 2 diabetes developed dementia. Nonvascular dementia was the most common type of dementia recorded, followed by Alzheimer’s disease and then vascular dementia.
“Within type 2 diabetes individuals, poor glycemic [control] increased the risk of dementia especially for vascular dementia and nonvascular dementia. However, these associations were not as evident for Alzheimer’s disease,” Dr. Celis-Morales reported.
Comparing those with hemoglobin bA1c of less than 52 mmol/mol (7%) with those whose A1c was above 87 mmol/mol (10.1%), there was 93% increase in the risk for vascular dementia, a 67% increase in the risk for nonvascular dementia, and a 34% higher risk for Alzheimer’s disease–associated dementia.
“We have focused on high levels of HbA1c, but what happens if you have really low limits? It’s something we’re working on right now,” Dr. Celis-Morales said.
Importantly, cardiovascular-related risk factors – some of which, like systolic blood pressure and body weight, were potentially modifiable – accounted for more than 40% of the risk for dementia in type 2 diabetes. This suggests that a large percentage of the dementia risk could perhaps be addressed by identifying high-risk individuals and tailoring interventions accordingly.
“These are observational findings, so we need to be careful before we translate to any sort of recommendation,” Dr. Celis-Morales said.
The study was financed by the Swedish state under the agreement between the government and the county councils, the ALF agreement, as well as grant from the Novo Nordisk Foundation and the Swedish Association of Local Authorities and Regions. Dr. Celis-Morales and Dr. Sattar had no conflicts of interest.
SOURCE: Celis-Morales C et al. EASD 2020, Oral presentation 06.
Persons with type 2 diabetes may be at heightened risk for developing vascular dementia than other types of dementia, a team of international researchers has found.
Compared with a nondiabetic control population, those with type 2 diabetes had a statistically significant 35% increased chance of having vascular dementia in a large observational study.
By comparison, the risk for nonvascular dementia was increased by a “more modest” 8%, said the researchers from the University of Glasgow and the University of Gothenburg (Sweden), while the risk for Alzheimer’s dementia appeared to be reduced by 8%.
The link between type 2 diabetes and dementia is not new, observed Carlos Celis-Morales, PhD, who presented the study’s findings at the virtual annual meeting of the European Association for the Study of Diabetes. With people living longer thanks to improved preventative strategies and treatments, there is a risk for developing other chronic conditions, such as dementia.
“A third of all dementia cases may be attributable to modifiable risk factors, among them type 2 diabetes, which accounts for 3.2% of all dementia cases,” said Dr. Celis-Morales, a research fellow at the University of Glasgow’s Institute of Cardiovascular and Medical Sciences.
“Although we know that diabetes is linked to dementia, what we don’t know really well is how much of this association between diabetes and dementia outcomes are explained by modifiable and nonmodifiable risk factors,” Dr. Celis-Morales added.
“Diabetes and dementia share certain risk factors,” commented coinvestigator Naveed Sattar, MD, in a press release issued by the EASD. These include obesity, smoking, and lack of physical activity and might explain part of the association between the two conditions.
Dr. Sattar said that the heightened vascular dementia risk found in the study was “in itself an argument for preventive measures such as healthier lifestyle,” adding that “the importance of prevention is underscored by the fact that, for the majority of dementia diseases, there is no good treatment.”
Using data from the Swedish National Diabetes Register, the research team set out to determine the extent to which type 2 diabetes was associated with dementia and the incidence of different subtypes of dementia. They also looked to see if there were any associations with blood glucose control and what risk factors may be involved.
In total, data on 378,299 individuals with type 2 diabetes were compared with data on 1,886,022 similarly aged (average, 64 years) and gender-matched controls from the general population.
After a mean 7 years of follow-up, 10,143 people with and 46,479 people without type 2 diabetes developed dementia. Nonvascular dementia was the most common type of dementia recorded, followed by Alzheimer’s disease and then vascular dementia.
“Within type 2 diabetes individuals, poor glycemic [control] increased the risk of dementia especially for vascular dementia and nonvascular dementia. However, these associations were not as evident for Alzheimer’s disease,” Dr. Celis-Morales reported.
Comparing those with hemoglobin bA1c of less than 52 mmol/mol (7%) with those whose A1c was above 87 mmol/mol (10.1%), there was 93% increase in the risk for vascular dementia, a 67% increase in the risk for nonvascular dementia, and a 34% higher risk for Alzheimer’s disease–associated dementia.
“We have focused on high levels of HbA1c, but what happens if you have really low limits? It’s something we’re working on right now,” Dr. Celis-Morales said.
Importantly, cardiovascular-related risk factors – some of which, like systolic blood pressure and body weight, were potentially modifiable – accounted for more than 40% of the risk for dementia in type 2 diabetes. This suggests that a large percentage of the dementia risk could perhaps be addressed by identifying high-risk individuals and tailoring interventions accordingly.
“These are observational findings, so we need to be careful before we translate to any sort of recommendation,” Dr. Celis-Morales said.
The study was financed by the Swedish state under the agreement between the government and the county councils, the ALF agreement, as well as grant from the Novo Nordisk Foundation and the Swedish Association of Local Authorities and Regions. Dr. Celis-Morales and Dr. Sattar had no conflicts of interest.
SOURCE: Celis-Morales C et al. EASD 2020, Oral presentation 06.
Persons with type 2 diabetes may be at heightened risk for developing vascular dementia than other types of dementia, a team of international researchers has found.
Compared with a nondiabetic control population, those with type 2 diabetes had a statistically significant 35% increased chance of having vascular dementia in a large observational study.
By comparison, the risk for nonvascular dementia was increased by a “more modest” 8%, said the researchers from the University of Glasgow and the University of Gothenburg (Sweden), while the risk for Alzheimer’s dementia appeared to be reduced by 8%.
The link between type 2 diabetes and dementia is not new, observed Carlos Celis-Morales, PhD, who presented the study’s findings at the virtual annual meeting of the European Association for the Study of Diabetes. With people living longer thanks to improved preventative strategies and treatments, there is a risk for developing other chronic conditions, such as dementia.
“A third of all dementia cases may be attributable to modifiable risk factors, among them type 2 diabetes, which accounts for 3.2% of all dementia cases,” said Dr. Celis-Morales, a research fellow at the University of Glasgow’s Institute of Cardiovascular and Medical Sciences.
“Although we know that diabetes is linked to dementia, what we don’t know really well is how much of this association between diabetes and dementia outcomes are explained by modifiable and nonmodifiable risk factors,” Dr. Celis-Morales added.
“Diabetes and dementia share certain risk factors,” commented coinvestigator Naveed Sattar, MD, in a press release issued by the EASD. These include obesity, smoking, and lack of physical activity and might explain part of the association between the two conditions.
Dr. Sattar said that the heightened vascular dementia risk found in the study was “in itself an argument for preventive measures such as healthier lifestyle,” adding that “the importance of prevention is underscored by the fact that, for the majority of dementia diseases, there is no good treatment.”
Using data from the Swedish National Diabetes Register, the research team set out to determine the extent to which type 2 diabetes was associated with dementia and the incidence of different subtypes of dementia. They also looked to see if there were any associations with blood glucose control and what risk factors may be involved.
In total, data on 378,299 individuals with type 2 diabetes were compared with data on 1,886,022 similarly aged (average, 64 years) and gender-matched controls from the general population.
After a mean 7 years of follow-up, 10,143 people with and 46,479 people without type 2 diabetes developed dementia. Nonvascular dementia was the most common type of dementia recorded, followed by Alzheimer’s disease and then vascular dementia.
“Within type 2 diabetes individuals, poor glycemic [control] increased the risk of dementia especially for vascular dementia and nonvascular dementia. However, these associations were not as evident for Alzheimer’s disease,” Dr. Celis-Morales reported.
Comparing those with hemoglobin bA1c of less than 52 mmol/mol (7%) with those whose A1c was above 87 mmol/mol (10.1%), there was 93% increase in the risk for vascular dementia, a 67% increase in the risk for nonvascular dementia, and a 34% higher risk for Alzheimer’s disease–associated dementia.
“We have focused on high levels of HbA1c, but what happens if you have really low limits? It’s something we’re working on right now,” Dr. Celis-Morales said.
Importantly, cardiovascular-related risk factors – some of which, like systolic blood pressure and body weight, were potentially modifiable – accounted for more than 40% of the risk for dementia in type 2 diabetes. This suggests that a large percentage of the dementia risk could perhaps be addressed by identifying high-risk individuals and tailoring interventions accordingly.
“These are observational findings, so we need to be careful before we translate to any sort of recommendation,” Dr. Celis-Morales said.
The study was financed by the Swedish state under the agreement between the government and the county councils, the ALF agreement, as well as grant from the Novo Nordisk Foundation and the Swedish Association of Local Authorities and Regions. Dr. Celis-Morales and Dr. Sattar had no conflicts of interest.
SOURCE: Celis-Morales C et al. EASD 2020, Oral presentation 06.
FROM EASD 2020
COVID-19 Screening and Testing Among Patients With Neurologic Dysfunction: The Neuro-COVID-19 Time-out Process and Checklist
From the University of Mississippi Medical Center, Department of Neurology, Division of Neuroscience Intensive Care, Jackson, MS.
Abstract
Objective: To test a coronavirus disease 2019 (COVID-19) screening tool to identify patients who qualify for testing among patients with neurologic dysfunction who are unable to answer the usual screening questions, which could help to prevent unprotected exposure of patients and health care workers to COVID-19.
Methods: The Neuro-COVID-19 Time-out Process and Checklist (NCOT-PC) was implemented at our institution for 1 week as a quality improvement project to improve the pathway for COVID-19 screening and testing among patients with neurologic dysfunction.
Results: A total of 14 new patients were admitted into the neuroscience intensive care unit (NSICU) service during the pilot period. The NCOT-PC was utilized on 9 (64%) patients with neurologic dysfunction; 7 of these patients were found to have a likelihood of requiring testing based on the NCOT-PC and were subsequently screened for COVID-19 testing by contacting the institution’s COVID-19 testing hotline (Med-Com). All these patients were subsequently transitioned into person-under-investigation status based on the determination from Med-Com. The NSICU staff involved were able to utilize NCOT-PC without issues. The NCOT-PC was immediately adopted into the NSICU process.
Conclusion: Use of the NCOT-PC tool was found to be feasible and improved the screening methodology of patients with neurologic dysfunction.
Keywords: coronavirus; health care planning; quality improvement; patient safety; medical decision-making; neuroscience intensive care unit.
The coronavirus disease 2019 (COVID-19) pandemic has altered various standard emergent care pathways. Current recommendations regarding COVID-19 screening for testing involve asking patients about their symptoms, including fever, cough, chest pain, and dyspnea.1 This standard screening method poses a problem when caring for patients with neurologic dysfunction. COVID-19 patients may pre-sent with conditions that affect their ability to answer questions, such as stroke, encephalitis, neuromuscular disorders, or headache, and that may preclude the use of standard screening for testing.2 Patients with acute neurologic dysfunction who cannot undergo standard screening may leave the emergency department (ED) and transition into the neuroscience intensive care unit (NSICU) or any intensive care unit (ICU) without a reliable COVID-19 screening test.
The Protected Code Stroke pathway offers protection in the emergent setting for patients with stroke when their COVID-19 status is unknown.3 A similar process has been applied at our institution for emergent management of patients with cerebrovascular disease (stroke, intracerebral hemorrhage, and subarachnoid hemorrhage). However, the process from the ED after designating “difficult to screen” patients as persons under investigation (PUI) is unclear. The Centers for Disease Control and Prevention (CDC) has delineated the priorities for testing, with not all declared PUIs requiring testing.4 This poses a great challenge, because patients designated as PUIs require the same management as a COVID-19-positive patient, with negative-pressure isolation rooms as well as use of protective personal equipment (PPE), which may not be readily available. It was also recognized that, because the ED staff can be overwhelmed by COVID-19 patients, there may not be enough time to perform detailed screening of patients with neurologic dysfunction and that “reverse masking” may not be done consistently for nonintubated patients. This may place patients and health care workers at risk of unprotected exposure.
Recognizing these challenges, we created a Neuro-COVID-19 Time-out Process and Checklist (NCOT-PC) as a quality improvement project. The aim of this project was to improve and standardize the current process of identifying patients with neurologic dysfunction who require COVID-19 testing to decrease the risk of unprotected exposure of patients and health care workers.
Methods
Patients and Definitions
This quality improvement project was undertaken at the University of Mississippi Medical Center NSICU. Because this was a quality improvement project, an Institutional Review Board exemption was granted.
The NCOT-PC was utilized in consecutive patients with neurologic dysfunction admitted to the NSICU during a period of 1 week. “Neurologic dysfunction” encompasses any neurologic illness affecting the mental status and/or level of alertness, subsequently precluding the ability to reliably screen the patient utilizing standard COVID-19 screening. “Med-Com” at our institution is the equivalent of the national COVID-19 testing hotline, where our institution’s infectious diseases experts screen calls for testing and determine whether testing is warranted. “Unprotected exposure” means exposure to COVID-19 without adequate and appropriate PPE.
Quality Improvement Process
As more PUIs were being admitted to the institution, we used the Plan-Do-Study-Act method for process improvements in the NSICU.5 NSICU stakeholders, including attendings, the nurse manager, and nurse practitioners (NPs), developed an algorithm to facilitate the coordination of the NSICU staff in screening patients to identify those with a high likelihood of needing COVID-19 testing upon arrival in the NSICU (Figure 1). Once the NCOT-PC was finalized, NSICU stakeholders were educated regarding the use of this screening tool.
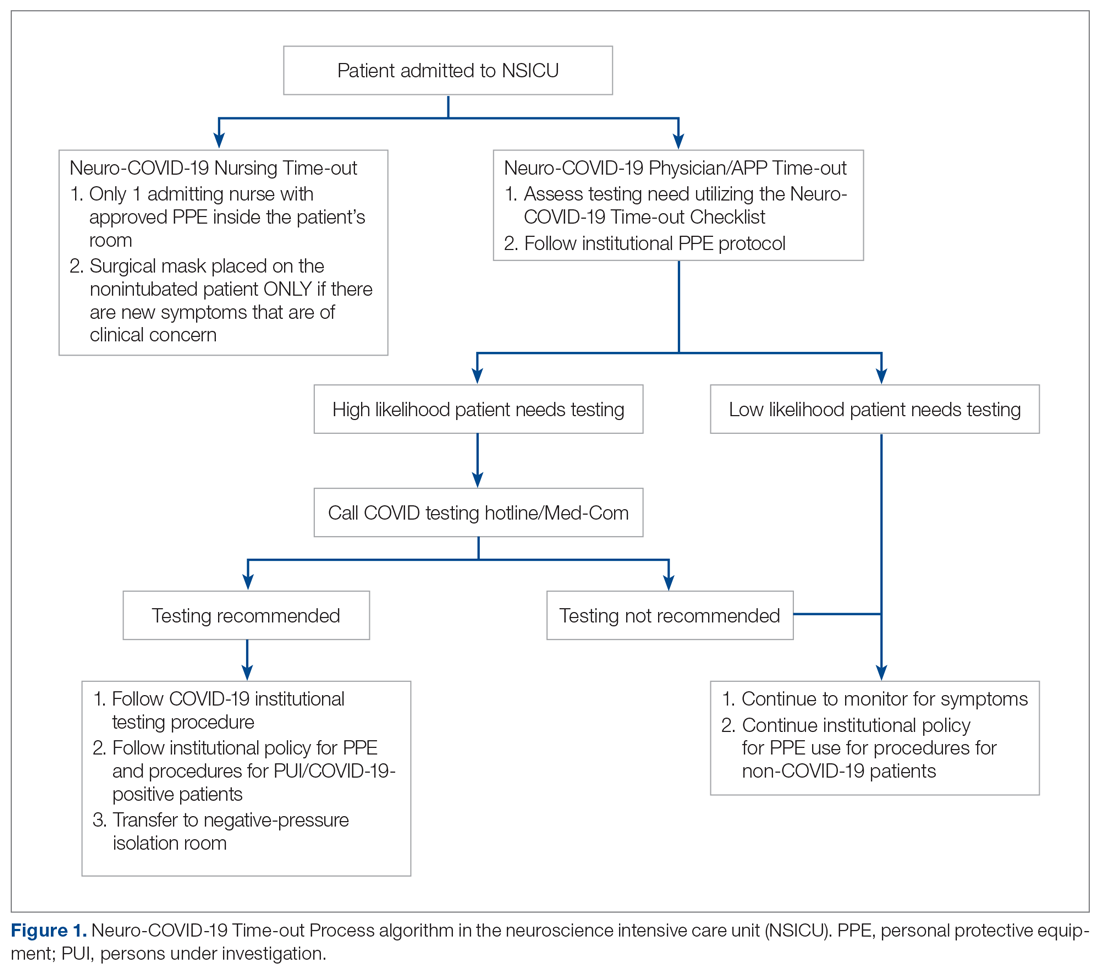
The checklist clinicians review when screening patients is shown in Figure 2. The risk factors comprising the checklist include patient history and clinical and radiographic characteristics that have been shown to be relevant for identifying patients with COVID-19.6,7 The imaging criteria utilize imaging that is part of the standard of care for NSICU patients. For example, computed tomography angiogram of the head and neck performed as part of the acute stroke protocol captures the upper part of the chest. These images are utilized for their incidental findings, such as apical ground-glass opacities and tree-in-bud formation. The risk factors applicable to the patient determine whether the clinician will call Med-Com for testing approval. Institutional COVID-19 processes were then followed accordingly.8 The decision from Med-Com was considered final, and no deviation from institutional policies was allowed.
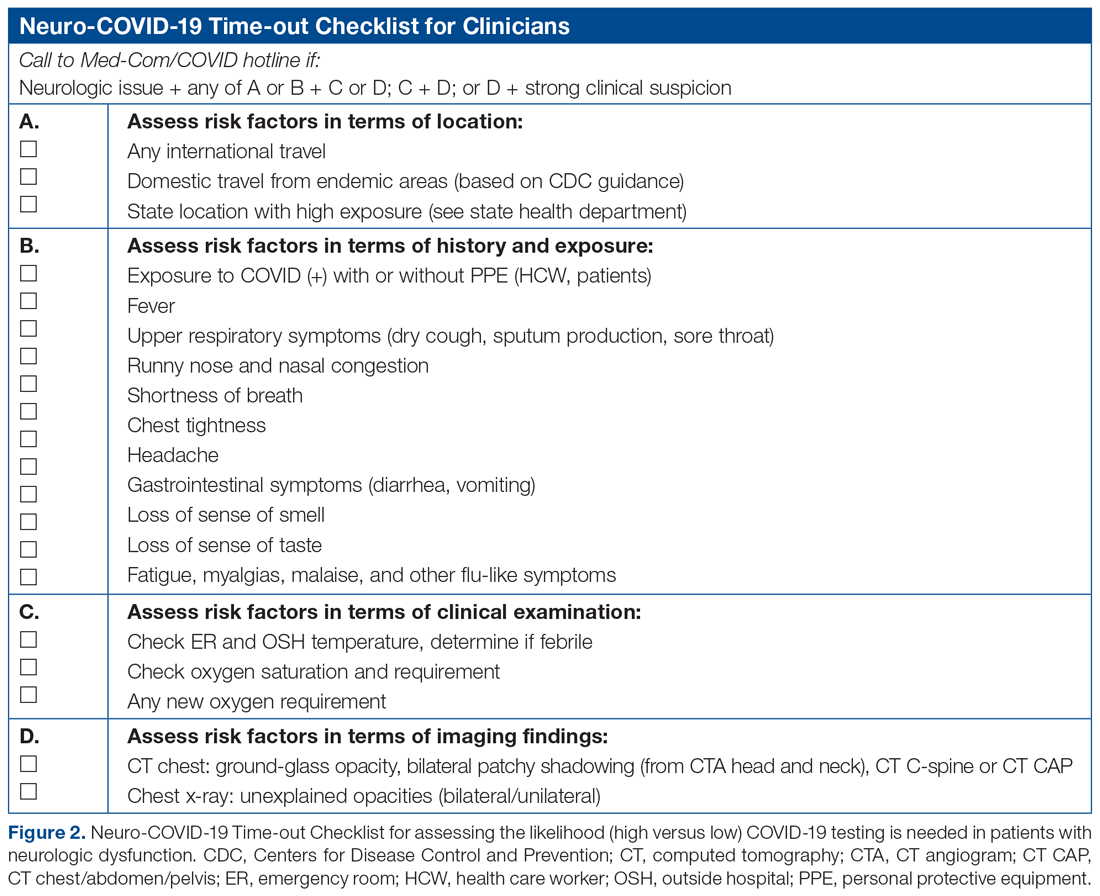
NCOT-PC was utilized for consecutive days for 1 week before re-evaluation of its feasibility and adaptability.
Data Collection and Analysis
Consecutive patients with neurologic dysfunction admitted into the NSICU were assigned nonlinkable patient numbers. No identifiers were collected for the purpose of this project. The primary diagnosis for admission, the neurologic dysfunction that precluded standard screening, and checklist components that the patient fulfilled were collected.
To assess the tool’s feasibility, feedback regarding the ease of use of the NCOT-PC was gathered from the nurses, NPs, charge nurses, fellows, and other attendings. To assess the utility of the NCOT-PC in identifying patients who will be approved for COVID-19 testing, we calculated the proportion of patients who were deemed to have a high likelihood of testing and the proportion of patients who were approved for testing. Descriptive statistics were used, as applicable for the project, to summarize the utility of the NCOT-PC.
Results
We found that the NCOT-PC can be easily used by clinicians. The NSICU staff did not communicate any implementation issues, and since the NCOT-PC was implemented, no problems have been identified.
During the pilot period of the NCOT-PC, 14 new patients were admitted to the NSICU service. Nine (64%) of these had neurologic dysfunction, and the NCOT-PC was used to determine whether Med-Com should be called based on the patients’ likelihood (high vs low) of needing a COVID-19 test. Of those patients with neurologic dysfunction, 7 (78%) were deemed to have a high likelihood of needing a COVID-19 test based on the NCOT-PC. Med-Com was contacted regarding these patients, and all were deemed to require the COVID-19 test by Med-Com and were transitioned into PUI status per institutional policy (Table).
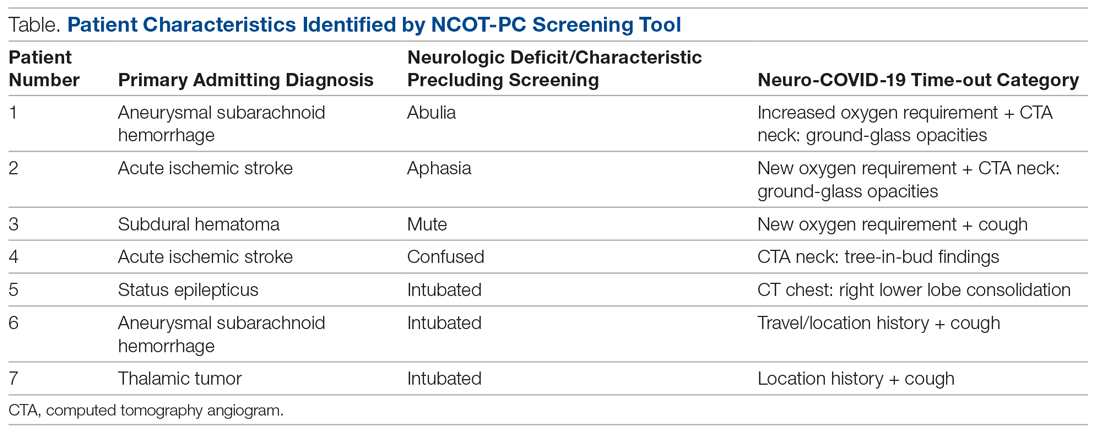
Discussion
The NCOT-PC project improved and standardized the process of identifying and screening patients with neurologic dysfunction for COVID-19 testing. The screening tool is feasible to use, and it decreased inadvertent unprotected exposure of patients and health care workers.
The NCOT-PC was easy to administer. Educating the staff regarding the new process took only a few minutes and involved a meeting with the nurse manager, NPs, fellows, residents, and attendings. We found that this process works well in tandem with the standard institutional processes in place in terms of Protected Code Stroke pathway, PUI isolation, PPE use, and Med-Com screening for COVID-19 testing. Med-Com was called only if the patient fulfilled the checklist criteria. In addition, no extra cost was attributed to implementing the NCOT-PC, since we utilized imaging that was already done as part of the standard of care for patients with neurologic dysfunction.
The standardization of the process of screening for COVID-19 testing among patients with neurologic dysfunction improved patient selection. Before the NCOT-PC, there was no consistency in terms of who should get tested and the reason for testing patients with neurologic dysfunction. Patients can pass through the ED and arrive in the NSICU with an unclear screening status, which may cause inadvertent patient and health care worker exposure to COVID-19. With the NCOT-PC, we have avoided instances of inadvertent staff or patient exposure in the NSICU.
The NCOT-PC was adopted into the NSICU process after the first week it was piloted. Beyond the NSICU, the application of the NCOT-PC can be extended to any patient presentation that precludes standard screening, such as ED and interhospital transfers for stroke codes, trauma codes, code blue, or myocardial infarction codes. In our department, as we started the process of PCS for stroke codes, we included NCOT-PC for stroke patients with neurologic dysfunction.
The results of our initiative are largely limited by the decision-making process of Med-Com when patients are called in for testing. At the time of our project, there were no specific criteria used for patients with altered mental status, except for the standard screening methods, and it was through clinician-to-clinician discussion that testing decisions were made. Another limitation is the short period of time that the NCOT-PC was applied before adoption.
In summary, the NCOT-PC tool improved the screening process for COVID-19 testing in patients with neurologic dysfunction admitted to the NSICU. It was feasible and prevented unprotected staff and patient exposure to COVID-19. The NCOT-PC functionality was compatible with institutional COVID-19 policies in place, which contributed to its overall sustainability.
The Standards for Quality Improvement Reporting Excellence (SQUIRE 2.0) were utilized in preparing this manuscript.9
Acknowledgment: The authors thank the University of Mississippi Medical Center NSICU staff for their input with implementation of the NCOT-PC.
Corresponding author: Prashant A. Natteru, MD, University of Mississippi Medical Center, Department of Neurology, 2500 North State St., Jackson, MS 39216; [email protected].
Financial disclosures: None.
1. Coronavirus disease 2019 (COVID-19) Symptoms. www.cdc.gov/coronavirus/2019-ncov/symptoms-testing/symptoms.html. Accessed April 9, 2020.
2. Mao L, Jin H, Wang M, et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020;77:1-9.
3. Khosravani H, Rajendram P, Notario L, et al. Protected code stroke: hyperacute stroke management during the coronavirus disease 2019. (COVID-19) pandemic. Stroke. 2020;51:1891-1895.
4. Coronavirus disease 2019 (COVID-19) evaluation and testing. www.cdc.gov/coronavirus/2019-nCoV/hcp/clinical-criteria.html. Accessed April 9, 2020.
5. Plan-Do-Study-Act Worksheet. Institute for Healthcare Improvement website. www.ihi.org/resources/Pages/Tools/PlanDoStudyActWorksheet.aspx. Accessed March 31,2020.
6. Li YC, Bai WZ, Hashikawa T. The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J Med Virol. 2020;10.1002/jmv.25728.
7. Rodriguez-Morales AJ, Cardona-Ospina JA, Gutiérrez-Ocampo E, et al. Clinical, laboratory and imaging features of COVID-19: A systematic review and meta-analysis. Travel Med Infect Dis. 2020;101623.
8. UMMC’s COVID-19 Clinical Processes. www.umc.edu/CoronaVirus/Mississippi-Health-Care-Professionals/Clinical-Resources/Clinical-Resources.html. Accessed April 9, 2020.
9. SQUIRE 2.0 (Standards for QUality Improvement Reporting Excellence): Revised Publication Guidelines from a Detailed Consensus Process. The EQUATOR Network. www.equator-network.org/reporting-guidelines/squire/. Accessed May 12, 2020.
From the University of Mississippi Medical Center, Department of Neurology, Division of Neuroscience Intensive Care, Jackson, MS.
Abstract
Objective: To test a coronavirus disease 2019 (COVID-19) screening tool to identify patients who qualify for testing among patients with neurologic dysfunction who are unable to answer the usual screening questions, which could help to prevent unprotected exposure of patients and health care workers to COVID-19.
Methods: The Neuro-COVID-19 Time-out Process and Checklist (NCOT-PC) was implemented at our institution for 1 week as a quality improvement project to improve the pathway for COVID-19 screening and testing among patients with neurologic dysfunction.
Results: A total of 14 new patients were admitted into the neuroscience intensive care unit (NSICU) service during the pilot period. The NCOT-PC was utilized on 9 (64%) patients with neurologic dysfunction; 7 of these patients were found to have a likelihood of requiring testing based on the NCOT-PC and were subsequently screened for COVID-19 testing by contacting the institution’s COVID-19 testing hotline (Med-Com). All these patients were subsequently transitioned into person-under-investigation status based on the determination from Med-Com. The NSICU staff involved were able to utilize NCOT-PC without issues. The NCOT-PC was immediately adopted into the NSICU process.
Conclusion: Use of the NCOT-PC tool was found to be feasible and improved the screening methodology of patients with neurologic dysfunction.
Keywords: coronavirus; health care planning; quality improvement; patient safety; medical decision-making; neuroscience intensive care unit.
The coronavirus disease 2019 (COVID-19) pandemic has altered various standard emergent care pathways. Current recommendations regarding COVID-19 screening for testing involve asking patients about their symptoms, including fever, cough, chest pain, and dyspnea.1 This standard screening method poses a problem when caring for patients with neurologic dysfunction. COVID-19 patients may pre-sent with conditions that affect their ability to answer questions, such as stroke, encephalitis, neuromuscular disorders, or headache, and that may preclude the use of standard screening for testing.2 Patients with acute neurologic dysfunction who cannot undergo standard screening may leave the emergency department (ED) and transition into the neuroscience intensive care unit (NSICU) or any intensive care unit (ICU) without a reliable COVID-19 screening test.
The Protected Code Stroke pathway offers protection in the emergent setting for patients with stroke when their COVID-19 status is unknown.3 A similar process has been applied at our institution for emergent management of patients with cerebrovascular disease (stroke, intracerebral hemorrhage, and subarachnoid hemorrhage). However, the process from the ED after designating “difficult to screen” patients as persons under investigation (PUI) is unclear. The Centers for Disease Control and Prevention (CDC) has delineated the priorities for testing, with not all declared PUIs requiring testing.4 This poses a great challenge, because patients designated as PUIs require the same management as a COVID-19-positive patient, with negative-pressure isolation rooms as well as use of protective personal equipment (PPE), which may not be readily available. It was also recognized that, because the ED staff can be overwhelmed by COVID-19 patients, there may not be enough time to perform detailed screening of patients with neurologic dysfunction and that “reverse masking” may not be done consistently for nonintubated patients. This may place patients and health care workers at risk of unprotected exposure.
Recognizing these challenges, we created a Neuro-COVID-19 Time-out Process and Checklist (NCOT-PC) as a quality improvement project. The aim of this project was to improve and standardize the current process of identifying patients with neurologic dysfunction who require COVID-19 testing to decrease the risk of unprotected exposure of patients and health care workers.
Methods
Patients and Definitions
This quality improvement project was undertaken at the University of Mississippi Medical Center NSICU. Because this was a quality improvement project, an Institutional Review Board exemption was granted.
The NCOT-PC was utilized in consecutive patients with neurologic dysfunction admitted to the NSICU during a period of 1 week. “Neurologic dysfunction” encompasses any neurologic illness affecting the mental status and/or level of alertness, subsequently precluding the ability to reliably screen the patient utilizing standard COVID-19 screening. “Med-Com” at our institution is the equivalent of the national COVID-19 testing hotline, where our institution’s infectious diseases experts screen calls for testing and determine whether testing is warranted. “Unprotected exposure” means exposure to COVID-19 without adequate and appropriate PPE.
Quality Improvement Process
As more PUIs were being admitted to the institution, we used the Plan-Do-Study-Act method for process improvements in the NSICU.5 NSICU stakeholders, including attendings, the nurse manager, and nurse practitioners (NPs), developed an algorithm to facilitate the coordination of the NSICU staff in screening patients to identify those with a high likelihood of needing COVID-19 testing upon arrival in the NSICU (Figure 1). Once the NCOT-PC was finalized, NSICU stakeholders were educated regarding the use of this screening tool.

The checklist clinicians review when screening patients is shown in Figure 2. The risk factors comprising the checklist include patient history and clinical and radiographic characteristics that have been shown to be relevant for identifying patients with COVID-19.6,7 The imaging criteria utilize imaging that is part of the standard of care for NSICU patients. For example, computed tomography angiogram of the head and neck performed as part of the acute stroke protocol captures the upper part of the chest. These images are utilized for their incidental findings, such as apical ground-glass opacities and tree-in-bud formation. The risk factors applicable to the patient determine whether the clinician will call Med-Com for testing approval. Institutional COVID-19 processes were then followed accordingly.8 The decision from Med-Com was considered final, and no deviation from institutional policies was allowed.

NCOT-PC was utilized for consecutive days for 1 week before re-evaluation of its feasibility and adaptability.
Data Collection and Analysis
Consecutive patients with neurologic dysfunction admitted into the NSICU were assigned nonlinkable patient numbers. No identifiers were collected for the purpose of this project. The primary diagnosis for admission, the neurologic dysfunction that precluded standard screening, and checklist components that the patient fulfilled were collected.
To assess the tool’s feasibility, feedback regarding the ease of use of the NCOT-PC was gathered from the nurses, NPs, charge nurses, fellows, and other attendings. To assess the utility of the NCOT-PC in identifying patients who will be approved for COVID-19 testing, we calculated the proportion of patients who were deemed to have a high likelihood of testing and the proportion of patients who were approved for testing. Descriptive statistics were used, as applicable for the project, to summarize the utility of the NCOT-PC.
Results
We found that the NCOT-PC can be easily used by clinicians. The NSICU staff did not communicate any implementation issues, and since the NCOT-PC was implemented, no problems have been identified.
During the pilot period of the NCOT-PC, 14 new patients were admitted to the NSICU service. Nine (64%) of these had neurologic dysfunction, and the NCOT-PC was used to determine whether Med-Com should be called based on the patients’ likelihood (high vs low) of needing a COVID-19 test. Of those patients with neurologic dysfunction, 7 (78%) were deemed to have a high likelihood of needing a COVID-19 test based on the NCOT-PC. Med-Com was contacted regarding these patients, and all were deemed to require the COVID-19 test by Med-Com and were transitioned into PUI status per institutional policy (Table).

Discussion
The NCOT-PC project improved and standardized the process of identifying and screening patients with neurologic dysfunction for COVID-19 testing. The screening tool is feasible to use, and it decreased inadvertent unprotected exposure of patients and health care workers.
The NCOT-PC was easy to administer. Educating the staff regarding the new process took only a few minutes and involved a meeting with the nurse manager, NPs, fellows, residents, and attendings. We found that this process works well in tandem with the standard institutional processes in place in terms of Protected Code Stroke pathway, PUI isolation, PPE use, and Med-Com screening for COVID-19 testing. Med-Com was called only if the patient fulfilled the checklist criteria. In addition, no extra cost was attributed to implementing the NCOT-PC, since we utilized imaging that was already done as part of the standard of care for patients with neurologic dysfunction.
The standardization of the process of screening for COVID-19 testing among patients with neurologic dysfunction improved patient selection. Before the NCOT-PC, there was no consistency in terms of who should get tested and the reason for testing patients with neurologic dysfunction. Patients can pass through the ED and arrive in the NSICU with an unclear screening status, which may cause inadvertent patient and health care worker exposure to COVID-19. With the NCOT-PC, we have avoided instances of inadvertent staff or patient exposure in the NSICU.
The NCOT-PC was adopted into the NSICU process after the first week it was piloted. Beyond the NSICU, the application of the NCOT-PC can be extended to any patient presentation that precludes standard screening, such as ED and interhospital transfers for stroke codes, trauma codes, code blue, or myocardial infarction codes. In our department, as we started the process of PCS for stroke codes, we included NCOT-PC for stroke patients with neurologic dysfunction.
The results of our initiative are largely limited by the decision-making process of Med-Com when patients are called in for testing. At the time of our project, there were no specific criteria used for patients with altered mental status, except for the standard screening methods, and it was through clinician-to-clinician discussion that testing decisions were made. Another limitation is the short period of time that the NCOT-PC was applied before adoption.
In summary, the NCOT-PC tool improved the screening process for COVID-19 testing in patients with neurologic dysfunction admitted to the NSICU. It was feasible and prevented unprotected staff and patient exposure to COVID-19. The NCOT-PC functionality was compatible with institutional COVID-19 policies in place, which contributed to its overall sustainability.
The Standards for Quality Improvement Reporting Excellence (SQUIRE 2.0) were utilized in preparing this manuscript.9
Acknowledgment: The authors thank the University of Mississippi Medical Center NSICU staff for their input with implementation of the NCOT-PC.
Corresponding author: Prashant A. Natteru, MD, University of Mississippi Medical Center, Department of Neurology, 2500 North State St., Jackson, MS 39216; [email protected].
Financial disclosures: None.
From the University of Mississippi Medical Center, Department of Neurology, Division of Neuroscience Intensive Care, Jackson, MS.
Abstract
Objective: To test a coronavirus disease 2019 (COVID-19) screening tool to identify patients who qualify for testing among patients with neurologic dysfunction who are unable to answer the usual screening questions, which could help to prevent unprotected exposure of patients and health care workers to COVID-19.
Methods: The Neuro-COVID-19 Time-out Process and Checklist (NCOT-PC) was implemented at our institution for 1 week as a quality improvement project to improve the pathway for COVID-19 screening and testing among patients with neurologic dysfunction.
Results: A total of 14 new patients were admitted into the neuroscience intensive care unit (NSICU) service during the pilot period. The NCOT-PC was utilized on 9 (64%) patients with neurologic dysfunction; 7 of these patients were found to have a likelihood of requiring testing based on the NCOT-PC and were subsequently screened for COVID-19 testing by contacting the institution’s COVID-19 testing hotline (Med-Com). All these patients were subsequently transitioned into person-under-investigation status based on the determination from Med-Com. The NSICU staff involved were able to utilize NCOT-PC without issues. The NCOT-PC was immediately adopted into the NSICU process.
Conclusion: Use of the NCOT-PC tool was found to be feasible and improved the screening methodology of patients with neurologic dysfunction.
Keywords: coronavirus; health care planning; quality improvement; patient safety; medical decision-making; neuroscience intensive care unit.
The coronavirus disease 2019 (COVID-19) pandemic has altered various standard emergent care pathways. Current recommendations regarding COVID-19 screening for testing involve asking patients about their symptoms, including fever, cough, chest pain, and dyspnea.1 This standard screening method poses a problem when caring for patients with neurologic dysfunction. COVID-19 patients may pre-sent with conditions that affect their ability to answer questions, such as stroke, encephalitis, neuromuscular disorders, or headache, and that may preclude the use of standard screening for testing.2 Patients with acute neurologic dysfunction who cannot undergo standard screening may leave the emergency department (ED) and transition into the neuroscience intensive care unit (NSICU) or any intensive care unit (ICU) without a reliable COVID-19 screening test.
The Protected Code Stroke pathway offers protection in the emergent setting for patients with stroke when their COVID-19 status is unknown.3 A similar process has been applied at our institution for emergent management of patients with cerebrovascular disease (stroke, intracerebral hemorrhage, and subarachnoid hemorrhage). However, the process from the ED after designating “difficult to screen” patients as persons under investigation (PUI) is unclear. The Centers for Disease Control and Prevention (CDC) has delineated the priorities for testing, with not all declared PUIs requiring testing.4 This poses a great challenge, because patients designated as PUIs require the same management as a COVID-19-positive patient, with negative-pressure isolation rooms as well as use of protective personal equipment (PPE), which may not be readily available. It was also recognized that, because the ED staff can be overwhelmed by COVID-19 patients, there may not be enough time to perform detailed screening of patients with neurologic dysfunction and that “reverse masking” may not be done consistently for nonintubated patients. This may place patients and health care workers at risk of unprotected exposure.
Recognizing these challenges, we created a Neuro-COVID-19 Time-out Process and Checklist (NCOT-PC) as a quality improvement project. The aim of this project was to improve and standardize the current process of identifying patients with neurologic dysfunction who require COVID-19 testing to decrease the risk of unprotected exposure of patients and health care workers.
Methods
Patients and Definitions
This quality improvement project was undertaken at the University of Mississippi Medical Center NSICU. Because this was a quality improvement project, an Institutional Review Board exemption was granted.
The NCOT-PC was utilized in consecutive patients with neurologic dysfunction admitted to the NSICU during a period of 1 week. “Neurologic dysfunction” encompasses any neurologic illness affecting the mental status and/or level of alertness, subsequently precluding the ability to reliably screen the patient utilizing standard COVID-19 screening. “Med-Com” at our institution is the equivalent of the national COVID-19 testing hotline, where our institution’s infectious diseases experts screen calls for testing and determine whether testing is warranted. “Unprotected exposure” means exposure to COVID-19 without adequate and appropriate PPE.
Quality Improvement Process
As more PUIs were being admitted to the institution, we used the Plan-Do-Study-Act method for process improvements in the NSICU.5 NSICU stakeholders, including attendings, the nurse manager, and nurse practitioners (NPs), developed an algorithm to facilitate the coordination of the NSICU staff in screening patients to identify those with a high likelihood of needing COVID-19 testing upon arrival in the NSICU (Figure 1). Once the NCOT-PC was finalized, NSICU stakeholders were educated regarding the use of this screening tool.

The checklist clinicians review when screening patients is shown in Figure 2. The risk factors comprising the checklist include patient history and clinical and radiographic characteristics that have been shown to be relevant for identifying patients with COVID-19.6,7 The imaging criteria utilize imaging that is part of the standard of care for NSICU patients. For example, computed tomography angiogram of the head and neck performed as part of the acute stroke protocol captures the upper part of the chest. These images are utilized for their incidental findings, such as apical ground-glass opacities and tree-in-bud formation. The risk factors applicable to the patient determine whether the clinician will call Med-Com for testing approval. Institutional COVID-19 processes were then followed accordingly.8 The decision from Med-Com was considered final, and no deviation from institutional policies was allowed.

NCOT-PC was utilized for consecutive days for 1 week before re-evaluation of its feasibility and adaptability.
Data Collection and Analysis
Consecutive patients with neurologic dysfunction admitted into the NSICU were assigned nonlinkable patient numbers. No identifiers were collected for the purpose of this project. The primary diagnosis for admission, the neurologic dysfunction that precluded standard screening, and checklist components that the patient fulfilled were collected.
To assess the tool’s feasibility, feedback regarding the ease of use of the NCOT-PC was gathered from the nurses, NPs, charge nurses, fellows, and other attendings. To assess the utility of the NCOT-PC in identifying patients who will be approved for COVID-19 testing, we calculated the proportion of patients who were deemed to have a high likelihood of testing and the proportion of patients who were approved for testing. Descriptive statistics were used, as applicable for the project, to summarize the utility of the NCOT-PC.
Results
We found that the NCOT-PC can be easily used by clinicians. The NSICU staff did not communicate any implementation issues, and since the NCOT-PC was implemented, no problems have been identified.
During the pilot period of the NCOT-PC, 14 new patients were admitted to the NSICU service. Nine (64%) of these had neurologic dysfunction, and the NCOT-PC was used to determine whether Med-Com should be called based on the patients’ likelihood (high vs low) of needing a COVID-19 test. Of those patients with neurologic dysfunction, 7 (78%) were deemed to have a high likelihood of needing a COVID-19 test based on the NCOT-PC. Med-Com was contacted regarding these patients, and all were deemed to require the COVID-19 test by Med-Com and were transitioned into PUI status per institutional policy (Table).

Discussion
The NCOT-PC project improved and standardized the process of identifying and screening patients with neurologic dysfunction for COVID-19 testing. The screening tool is feasible to use, and it decreased inadvertent unprotected exposure of patients and health care workers.
The NCOT-PC was easy to administer. Educating the staff regarding the new process took only a few minutes and involved a meeting with the nurse manager, NPs, fellows, residents, and attendings. We found that this process works well in tandem with the standard institutional processes in place in terms of Protected Code Stroke pathway, PUI isolation, PPE use, and Med-Com screening for COVID-19 testing. Med-Com was called only if the patient fulfilled the checklist criteria. In addition, no extra cost was attributed to implementing the NCOT-PC, since we utilized imaging that was already done as part of the standard of care for patients with neurologic dysfunction.
The standardization of the process of screening for COVID-19 testing among patients with neurologic dysfunction improved patient selection. Before the NCOT-PC, there was no consistency in terms of who should get tested and the reason for testing patients with neurologic dysfunction. Patients can pass through the ED and arrive in the NSICU with an unclear screening status, which may cause inadvertent patient and health care worker exposure to COVID-19. With the NCOT-PC, we have avoided instances of inadvertent staff or patient exposure in the NSICU.
The NCOT-PC was adopted into the NSICU process after the first week it was piloted. Beyond the NSICU, the application of the NCOT-PC can be extended to any patient presentation that precludes standard screening, such as ED and interhospital transfers for stroke codes, trauma codes, code blue, or myocardial infarction codes. In our department, as we started the process of PCS for stroke codes, we included NCOT-PC for stroke patients with neurologic dysfunction.
The results of our initiative are largely limited by the decision-making process of Med-Com when patients are called in for testing. At the time of our project, there were no specific criteria used for patients with altered mental status, except for the standard screening methods, and it was through clinician-to-clinician discussion that testing decisions were made. Another limitation is the short period of time that the NCOT-PC was applied before adoption.
In summary, the NCOT-PC tool improved the screening process for COVID-19 testing in patients with neurologic dysfunction admitted to the NSICU. It was feasible and prevented unprotected staff and patient exposure to COVID-19. The NCOT-PC functionality was compatible with institutional COVID-19 policies in place, which contributed to its overall sustainability.
The Standards for Quality Improvement Reporting Excellence (SQUIRE 2.0) were utilized in preparing this manuscript.9
Acknowledgment: The authors thank the University of Mississippi Medical Center NSICU staff for their input with implementation of the NCOT-PC.
Corresponding author: Prashant A. Natteru, MD, University of Mississippi Medical Center, Department of Neurology, 2500 North State St., Jackson, MS 39216; [email protected].
Financial disclosures: None.
1. Coronavirus disease 2019 (COVID-19) Symptoms. www.cdc.gov/coronavirus/2019-ncov/symptoms-testing/symptoms.html. Accessed April 9, 2020.
2. Mao L, Jin H, Wang M, et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020;77:1-9.
3. Khosravani H, Rajendram P, Notario L, et al. Protected code stroke: hyperacute stroke management during the coronavirus disease 2019. (COVID-19) pandemic. Stroke. 2020;51:1891-1895.
4. Coronavirus disease 2019 (COVID-19) evaluation and testing. www.cdc.gov/coronavirus/2019-nCoV/hcp/clinical-criteria.html. Accessed April 9, 2020.
5. Plan-Do-Study-Act Worksheet. Institute for Healthcare Improvement website. www.ihi.org/resources/Pages/Tools/PlanDoStudyActWorksheet.aspx. Accessed March 31,2020.
6. Li YC, Bai WZ, Hashikawa T. The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J Med Virol. 2020;10.1002/jmv.25728.
7. Rodriguez-Morales AJ, Cardona-Ospina JA, Gutiérrez-Ocampo E, et al. Clinical, laboratory and imaging features of COVID-19: A systematic review and meta-analysis. Travel Med Infect Dis. 2020;101623.
8. UMMC’s COVID-19 Clinical Processes. www.umc.edu/CoronaVirus/Mississippi-Health-Care-Professionals/Clinical-Resources/Clinical-Resources.html. Accessed April 9, 2020.
9. SQUIRE 2.0 (Standards for QUality Improvement Reporting Excellence): Revised Publication Guidelines from a Detailed Consensus Process. The EQUATOR Network. www.equator-network.org/reporting-guidelines/squire/. Accessed May 12, 2020.
1. Coronavirus disease 2019 (COVID-19) Symptoms. www.cdc.gov/coronavirus/2019-ncov/symptoms-testing/symptoms.html. Accessed April 9, 2020.
2. Mao L, Jin H, Wang M, et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020;77:1-9.
3. Khosravani H, Rajendram P, Notario L, et al. Protected code stroke: hyperacute stroke management during the coronavirus disease 2019. (COVID-19) pandemic. Stroke. 2020;51:1891-1895.
4. Coronavirus disease 2019 (COVID-19) evaluation and testing. www.cdc.gov/coronavirus/2019-nCoV/hcp/clinical-criteria.html. Accessed April 9, 2020.
5. Plan-Do-Study-Act Worksheet. Institute for Healthcare Improvement website. www.ihi.org/resources/Pages/Tools/PlanDoStudyActWorksheet.aspx. Accessed March 31,2020.
6. Li YC, Bai WZ, Hashikawa T. The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J Med Virol. 2020;10.1002/jmv.25728.
7. Rodriguez-Morales AJ, Cardona-Ospina JA, Gutiérrez-Ocampo E, et al. Clinical, laboratory and imaging features of COVID-19: A systematic review and meta-analysis. Travel Med Infect Dis. 2020;101623.
8. UMMC’s COVID-19 Clinical Processes. www.umc.edu/CoronaVirus/Mississippi-Health-Care-Professionals/Clinical-Resources/Clinical-Resources.html. Accessed April 9, 2020.
9. SQUIRE 2.0 (Standards for QUality Improvement Reporting Excellence): Revised Publication Guidelines from a Detailed Consensus Process. The EQUATOR Network. www.equator-network.org/reporting-guidelines/squire/. Accessed May 12, 2020.
Study validates OSA phenotypes in Latinos
Three previously described clinical phenotypes of obstructive sleep apnea (OSA) have been validated in a large and diverse Hispanic/Latino community-based population for the first time, according to findings presented at the virtual annual meeting of the Associated Professional Sleep Societies.
The three OSA symptom profiles present in this population – labeled “minimally symptomatic,” “disturbed sleep,” and “daytime sleepiness” – are consistent with recent findings from the Sleep Apnea Global Interdisciplinary Consortium, which were published in Sleep, but there are notable differences in the prevalence of these clusters, with the minimally symptomatic cluster much more prevalent than in prior research, reported Kevin Gonzalez, of the University of California, San Diego.
“Other biopsychosocial factors may be contributing to OSA phenotypes among Hispanics and Latinos,” Mr. Gonzalez said in his presentation. Prior research to characterize the heterogeneity of sleep apnea has not included a diverse Latino population, he emphasized.
The adults studied were aged 18-74 years and participants in the multisite Hispanic Community Health Study/Study of Latinos (HCHS/SOL), a comprehensive study of Hispanic/Latino health and disease in the United States. Their respiratory events were measured overnight in HCHS/SOL sleep reading centers with an ARES Unicorder 5.2, B-Alert. Sleep patterns and risk factors were assessed using the Sleep Heart Health Study Sleep Habits Questionnaire and the Epworth Sleepiness Scale.
Participants meeting the criteria for moderate to severe OSA (with an Apnea Hypopnea Index of 15 or above) were included in the analysis (n = 1,623). Their average age was 52.4 ± 13.9 years, and 34.1% were female.
To identify phenotype clusters, investigators performed a latent class analysis using 15 common OSA symptoms and a survey weighted to adjust for selection bias. The three clusters offering the “best” fit for the data aligned with the previously reported phenotypes and identified daytime sleepiness in 15.3%, disturbed sleep (insomnia-like symptoms) in 37.7%, and minimally symptomatic (a low symptom profile) in 46.9%.
These phenotypes were reported in the European Respiratory Journal in 2014 in a cluster analysis of data from a sleep apnea cohort in Iceland and later replicated in the analysis of data from the Sleep Apnea Global Interdisciplinary Consortium published in Sleep in 2018. The consortium study also added two additional phenotypes, labeled “upper airway symptoms dominant” and “sleepiness dominant.”
The prevalence of a “minimally symptomatic group” in the new analysis of the Hispanics/Latinos in the United States is much higher than reported in these prior studies, at least partly, the investigators believed, because the “prior studies were clinical samples, and the people who were minimally symptomatic didn’t get to the sleep centers,” Mr. Gonzalez said in an interview after the meeting.
Patients with a phenotype of daytime sleepiness – the most common phenotype in prior research – constituted only a minority in the Hispanic/Latino population, he said.
Alberto Ramos, MD, of the University of Miami and the principal investigator, said in an interview that the research team is currently analyzing “if and how these different [phenotypic] clusters could affect the incidence of comorbidities” recorded in the HCHS/SOL study, such as hypertension, diabetes, cardiovascular disease, and cognitive decline.
For now, he said, the findings suggest that OSA may be especially underrecognized in Hispanics and Latinos and that there is more research to be done to better identify and stratify patients with varying symptomatology for more personalized treatment and for clinical trial selection. “Maybe we should expand our criteria ... broaden our [recognition] of the presentation of sleep apnea and the symptoms associated with it, not only in Hispanics but maybe in the general population,” Dr. Ramos said.
In commenting on the study, Krishna M. Sundar, MD, FCCP, director of the Sleep-Wake Center at the University of Utah, Salt Lake City, said that insomnia and daytime sleepiness are “key associations with obstructive sleep apnea and may predict different outcomes with untreated OSA.” Such heterogeneity is “only beginning to be appreciated,” he said. “The expression of OSA with these symptoms points to how OSA impacts quality of life” and how symptomatology in addition to Apnea Hypopnea Index “may be an important determinant of treatment benefit and compliance.”
The investigators reported no relevant disclosures. Dr. Sundar said that he is cofounder of Hypnoscure, software for population management of sleep apnea, but with no monies received.
Three previously described clinical phenotypes of obstructive sleep apnea (OSA) have been validated in a large and diverse Hispanic/Latino community-based population for the first time, according to findings presented at the virtual annual meeting of the Associated Professional Sleep Societies.
The three OSA symptom profiles present in this population – labeled “minimally symptomatic,” “disturbed sleep,” and “daytime sleepiness” – are consistent with recent findings from the Sleep Apnea Global Interdisciplinary Consortium, which were published in Sleep, but there are notable differences in the prevalence of these clusters, with the minimally symptomatic cluster much more prevalent than in prior research, reported Kevin Gonzalez, of the University of California, San Diego.
“Other biopsychosocial factors may be contributing to OSA phenotypes among Hispanics and Latinos,” Mr. Gonzalez said in his presentation. Prior research to characterize the heterogeneity of sleep apnea has not included a diverse Latino population, he emphasized.
The adults studied were aged 18-74 years and participants in the multisite Hispanic Community Health Study/Study of Latinos (HCHS/SOL), a comprehensive study of Hispanic/Latino health and disease in the United States. Their respiratory events were measured overnight in HCHS/SOL sleep reading centers with an ARES Unicorder 5.2, B-Alert. Sleep patterns and risk factors were assessed using the Sleep Heart Health Study Sleep Habits Questionnaire and the Epworth Sleepiness Scale.
Participants meeting the criteria for moderate to severe OSA (with an Apnea Hypopnea Index of 15 or above) were included in the analysis (n = 1,623). Their average age was 52.4 ± 13.9 years, and 34.1% were female.
To identify phenotype clusters, investigators performed a latent class analysis using 15 common OSA symptoms and a survey weighted to adjust for selection bias. The three clusters offering the “best” fit for the data aligned with the previously reported phenotypes and identified daytime sleepiness in 15.3%, disturbed sleep (insomnia-like symptoms) in 37.7%, and minimally symptomatic (a low symptom profile) in 46.9%.
These phenotypes were reported in the European Respiratory Journal in 2014 in a cluster analysis of data from a sleep apnea cohort in Iceland and later replicated in the analysis of data from the Sleep Apnea Global Interdisciplinary Consortium published in Sleep in 2018. The consortium study also added two additional phenotypes, labeled “upper airway symptoms dominant” and “sleepiness dominant.”
The prevalence of a “minimally symptomatic group” in the new analysis of the Hispanics/Latinos in the United States is much higher than reported in these prior studies, at least partly, the investigators believed, because the “prior studies were clinical samples, and the people who were minimally symptomatic didn’t get to the sleep centers,” Mr. Gonzalez said in an interview after the meeting.
Patients with a phenotype of daytime sleepiness – the most common phenotype in prior research – constituted only a minority in the Hispanic/Latino population, he said.
Alberto Ramos, MD, of the University of Miami and the principal investigator, said in an interview that the research team is currently analyzing “if and how these different [phenotypic] clusters could affect the incidence of comorbidities” recorded in the HCHS/SOL study, such as hypertension, diabetes, cardiovascular disease, and cognitive decline.
For now, he said, the findings suggest that OSA may be especially underrecognized in Hispanics and Latinos and that there is more research to be done to better identify and stratify patients with varying symptomatology for more personalized treatment and for clinical trial selection. “Maybe we should expand our criteria ... broaden our [recognition] of the presentation of sleep apnea and the symptoms associated with it, not only in Hispanics but maybe in the general population,” Dr. Ramos said.
In commenting on the study, Krishna M. Sundar, MD, FCCP, director of the Sleep-Wake Center at the University of Utah, Salt Lake City, said that insomnia and daytime sleepiness are “key associations with obstructive sleep apnea and may predict different outcomes with untreated OSA.” Such heterogeneity is “only beginning to be appreciated,” he said. “The expression of OSA with these symptoms points to how OSA impacts quality of life” and how symptomatology in addition to Apnea Hypopnea Index “may be an important determinant of treatment benefit and compliance.”
The investigators reported no relevant disclosures. Dr. Sundar said that he is cofounder of Hypnoscure, software for population management of sleep apnea, but with no monies received.
Three previously described clinical phenotypes of obstructive sleep apnea (OSA) have been validated in a large and diverse Hispanic/Latino community-based population for the first time, according to findings presented at the virtual annual meeting of the Associated Professional Sleep Societies.
The three OSA symptom profiles present in this population – labeled “minimally symptomatic,” “disturbed sleep,” and “daytime sleepiness” – are consistent with recent findings from the Sleep Apnea Global Interdisciplinary Consortium, which were published in Sleep, but there are notable differences in the prevalence of these clusters, with the minimally symptomatic cluster much more prevalent than in prior research, reported Kevin Gonzalez, of the University of California, San Diego.
“Other biopsychosocial factors may be contributing to OSA phenotypes among Hispanics and Latinos,” Mr. Gonzalez said in his presentation. Prior research to characterize the heterogeneity of sleep apnea has not included a diverse Latino population, he emphasized.
The adults studied were aged 18-74 years and participants in the multisite Hispanic Community Health Study/Study of Latinos (HCHS/SOL), a comprehensive study of Hispanic/Latino health and disease in the United States. Their respiratory events were measured overnight in HCHS/SOL sleep reading centers with an ARES Unicorder 5.2, B-Alert. Sleep patterns and risk factors were assessed using the Sleep Heart Health Study Sleep Habits Questionnaire and the Epworth Sleepiness Scale.
Participants meeting the criteria for moderate to severe OSA (with an Apnea Hypopnea Index of 15 or above) were included in the analysis (n = 1,623). Their average age was 52.4 ± 13.9 years, and 34.1% were female.
To identify phenotype clusters, investigators performed a latent class analysis using 15 common OSA symptoms and a survey weighted to adjust for selection bias. The three clusters offering the “best” fit for the data aligned with the previously reported phenotypes and identified daytime sleepiness in 15.3%, disturbed sleep (insomnia-like symptoms) in 37.7%, and minimally symptomatic (a low symptom profile) in 46.9%.
These phenotypes were reported in the European Respiratory Journal in 2014 in a cluster analysis of data from a sleep apnea cohort in Iceland and later replicated in the analysis of data from the Sleep Apnea Global Interdisciplinary Consortium published in Sleep in 2018. The consortium study also added two additional phenotypes, labeled “upper airway symptoms dominant” and “sleepiness dominant.”
The prevalence of a “minimally symptomatic group” in the new analysis of the Hispanics/Latinos in the United States is much higher than reported in these prior studies, at least partly, the investigators believed, because the “prior studies were clinical samples, and the people who were minimally symptomatic didn’t get to the sleep centers,” Mr. Gonzalez said in an interview after the meeting.
Patients with a phenotype of daytime sleepiness – the most common phenotype in prior research – constituted only a minority in the Hispanic/Latino population, he said.
Alberto Ramos, MD, of the University of Miami and the principal investigator, said in an interview that the research team is currently analyzing “if and how these different [phenotypic] clusters could affect the incidence of comorbidities” recorded in the HCHS/SOL study, such as hypertension, diabetes, cardiovascular disease, and cognitive decline.
For now, he said, the findings suggest that OSA may be especially underrecognized in Hispanics and Latinos and that there is more research to be done to better identify and stratify patients with varying symptomatology for more personalized treatment and for clinical trial selection. “Maybe we should expand our criteria ... broaden our [recognition] of the presentation of sleep apnea and the symptoms associated with it, not only in Hispanics but maybe in the general population,” Dr. Ramos said.
In commenting on the study, Krishna M. Sundar, MD, FCCP, director of the Sleep-Wake Center at the University of Utah, Salt Lake City, said that insomnia and daytime sleepiness are “key associations with obstructive sleep apnea and may predict different outcomes with untreated OSA.” Such heterogeneity is “only beginning to be appreciated,” he said. “The expression of OSA with these symptoms points to how OSA impacts quality of life” and how symptomatology in addition to Apnea Hypopnea Index “may be an important determinant of treatment benefit and compliance.”
The investigators reported no relevant disclosures. Dr. Sundar said that he is cofounder of Hypnoscure, software for population management of sleep apnea, but with no monies received.
REPORTING FROM SLEEP 2020
Insomnia may have a role in generation of stressful life events
Insomnia disorder appears to play a causal role in the development of new stressful life events, especially “dependent” events for which individuals are at least partly responsible, said the investigators of an ongoing longitudinal study of people who have experienced involuntary job loss.
The “stress-generation hypothesis” has been applied for several decades in the context of depression. It posits that depressed individuals generate more stressful life events – events that create family conflict or disrupt careers, for instance – than individuals who are not depressed.
The new analysis of individuals with involuntary job loss suggests that the same can be said of insomnia. “Insomnia disorder is associated with fatigue, daytime sleepiness, impaired concentration, and difficulties in emotional regulation,” Iva Skobic, MSPH, MA, a PhD student at the University of Arizona, Tucson, said at the virtual annual meeting of the Associated Professional Sleep Societies.
“These may lead to impaired decision-making, interpersonal conflicts, difficulty meeting deadlines and keeping commitments, and other sources [of stressful life events],” she said. “This extension of the stress-generation hypothesis has important implications for harm reduction interventions for insomnia disorder.”
Investigators conducted a cross-lagged panel analysis using baseline and 3-month follow-up data from 137 individuals who completed a standardized, textual life event measure called the Life Events and Difficulties Schedule after having lost their jobs involuntarily. Participants were interviewed and their events were rated for severity by a consensus panel using operationalized criteria. The analysis employed linear regression controlling for covariates (age, gender, and race) and logistic regression that controlled for insomnia at baseline. Insomnia disorder was defined as meeting ICSD-2/3 criteria using the Duke Structured Interview for Sleep Disorders.
The findings: Insomnia disorder at baseline predicted the number of stressful life events (either dependent or interpersonal) generated within 3 months (beta, 0.70; standard error, 0.31; Tscore, 2.27; P = .03). Conversely, the number of stressful events at baseline did not predict insomnia (odds ratio, 0.97; 95% confidence interval, 0.73-1.29). There also was a trend toward increased generation of dependent events specifically among those with insomnia disorder.
Participants were a mean age of 42 years, and all had been in their previous place of employment for at least 6 months. Nearly 60% met the diagnostic threshold for insomnia at baseline. They were part of a larger ongoing study examining the linkages between job loss and sleep disturbances, obesity, and mental health – the Assessing Daily Activity Patterns through Occupational Transitions (ADAPT) study, supported by the National Heart, Lung, and Blood Institute.
This analysis on insomnia was completed before the COVID-19 pandemic began, but it and other analyses soon to be reported are highly relevant to the economic climate, said Patricia Haynes, PhD, principal investigator of ADAPT and a coauthor of the insomnia study, in an interview after the meeting.
Insomnia is a frequent comorbidity of depression and shares many of its symptoms, from increased fatigue to emotional dysregulation and an increased risk of maladaptive coping strategies. “Interestingly, the literature on the stress-generation hypothesis posits that these very symptoms are on the casual pathway between depression and stressful life events,” said Ms. Skobic at the meeting.
In commenting on the study, Krishna M. Sundar, MD, medical director of the Sleep-Wake Center at the University of Utah, Salt Lake City, noted that the analysis did not include any measure of the severity of insomnia. Still, he said, “finding an association [with stress generation] at [just] 3 months with the presence of insomnia disorder is quite interesting.”
There were higher rates of insomnia in the sample than depression, Dr. Haynes said, but the analysis did not control for depression or take it into account.
“We know [from prior research] that stress clearly leads to insomnia. The big [takeaway] here is that insomnia can also lead to more stress,” she said. “It’s important to think of it as a reciprocal relationship. If we can potentially treat insomnia, we may be able to stop that cycle of other stressful events that affect both [the individuals] and others as well.”
Ms. Skobic had no disclosures.
Insomnia disorder appears to play a causal role in the development of new stressful life events, especially “dependent” events for which individuals are at least partly responsible, said the investigators of an ongoing longitudinal study of people who have experienced involuntary job loss.
The “stress-generation hypothesis” has been applied for several decades in the context of depression. It posits that depressed individuals generate more stressful life events – events that create family conflict or disrupt careers, for instance – than individuals who are not depressed.
The new analysis of individuals with involuntary job loss suggests that the same can be said of insomnia. “Insomnia disorder is associated with fatigue, daytime sleepiness, impaired concentration, and difficulties in emotional regulation,” Iva Skobic, MSPH, MA, a PhD student at the University of Arizona, Tucson, said at the virtual annual meeting of the Associated Professional Sleep Societies.
“These may lead to impaired decision-making, interpersonal conflicts, difficulty meeting deadlines and keeping commitments, and other sources [of stressful life events],” she said. “This extension of the stress-generation hypothesis has important implications for harm reduction interventions for insomnia disorder.”
Investigators conducted a cross-lagged panel analysis using baseline and 3-month follow-up data from 137 individuals who completed a standardized, textual life event measure called the Life Events and Difficulties Schedule after having lost their jobs involuntarily. Participants were interviewed and their events were rated for severity by a consensus panel using operationalized criteria. The analysis employed linear regression controlling for covariates (age, gender, and race) and logistic regression that controlled for insomnia at baseline. Insomnia disorder was defined as meeting ICSD-2/3 criteria using the Duke Structured Interview for Sleep Disorders.
The findings: Insomnia disorder at baseline predicted the number of stressful life events (either dependent or interpersonal) generated within 3 months (beta, 0.70; standard error, 0.31; Tscore, 2.27; P = .03). Conversely, the number of stressful events at baseline did not predict insomnia (odds ratio, 0.97; 95% confidence interval, 0.73-1.29). There also was a trend toward increased generation of dependent events specifically among those with insomnia disorder.
Participants were a mean age of 42 years, and all had been in their previous place of employment for at least 6 months. Nearly 60% met the diagnostic threshold for insomnia at baseline. They were part of a larger ongoing study examining the linkages between job loss and sleep disturbances, obesity, and mental health – the Assessing Daily Activity Patterns through Occupational Transitions (ADAPT) study, supported by the National Heart, Lung, and Blood Institute.
This analysis on insomnia was completed before the COVID-19 pandemic began, but it and other analyses soon to be reported are highly relevant to the economic climate, said Patricia Haynes, PhD, principal investigator of ADAPT and a coauthor of the insomnia study, in an interview after the meeting.
Insomnia is a frequent comorbidity of depression and shares many of its symptoms, from increased fatigue to emotional dysregulation and an increased risk of maladaptive coping strategies. “Interestingly, the literature on the stress-generation hypothesis posits that these very symptoms are on the casual pathway between depression and stressful life events,” said Ms. Skobic at the meeting.
In commenting on the study, Krishna M. Sundar, MD, medical director of the Sleep-Wake Center at the University of Utah, Salt Lake City, noted that the analysis did not include any measure of the severity of insomnia. Still, he said, “finding an association [with stress generation] at [just] 3 months with the presence of insomnia disorder is quite interesting.”
There were higher rates of insomnia in the sample than depression, Dr. Haynes said, but the analysis did not control for depression or take it into account.
“We know [from prior research] that stress clearly leads to insomnia. The big [takeaway] here is that insomnia can also lead to more stress,” she said. “It’s important to think of it as a reciprocal relationship. If we can potentially treat insomnia, we may be able to stop that cycle of other stressful events that affect both [the individuals] and others as well.”
Ms. Skobic had no disclosures.
Insomnia disorder appears to play a causal role in the development of new stressful life events, especially “dependent” events for which individuals are at least partly responsible, said the investigators of an ongoing longitudinal study of people who have experienced involuntary job loss.
The “stress-generation hypothesis” has been applied for several decades in the context of depression. It posits that depressed individuals generate more stressful life events – events that create family conflict or disrupt careers, for instance – than individuals who are not depressed.
The new analysis of individuals with involuntary job loss suggests that the same can be said of insomnia. “Insomnia disorder is associated with fatigue, daytime sleepiness, impaired concentration, and difficulties in emotional regulation,” Iva Skobic, MSPH, MA, a PhD student at the University of Arizona, Tucson, said at the virtual annual meeting of the Associated Professional Sleep Societies.
“These may lead to impaired decision-making, interpersonal conflicts, difficulty meeting deadlines and keeping commitments, and other sources [of stressful life events],” she said. “This extension of the stress-generation hypothesis has important implications for harm reduction interventions for insomnia disorder.”
Investigators conducted a cross-lagged panel analysis using baseline and 3-month follow-up data from 137 individuals who completed a standardized, textual life event measure called the Life Events and Difficulties Schedule after having lost their jobs involuntarily. Participants were interviewed and their events were rated for severity by a consensus panel using operationalized criteria. The analysis employed linear regression controlling for covariates (age, gender, and race) and logistic regression that controlled for insomnia at baseline. Insomnia disorder was defined as meeting ICSD-2/3 criteria using the Duke Structured Interview for Sleep Disorders.
The findings: Insomnia disorder at baseline predicted the number of stressful life events (either dependent or interpersonal) generated within 3 months (beta, 0.70; standard error, 0.31; Tscore, 2.27; P = .03). Conversely, the number of stressful events at baseline did not predict insomnia (odds ratio, 0.97; 95% confidence interval, 0.73-1.29). There also was a trend toward increased generation of dependent events specifically among those with insomnia disorder.
Participants were a mean age of 42 years, and all had been in their previous place of employment for at least 6 months. Nearly 60% met the diagnostic threshold for insomnia at baseline. They were part of a larger ongoing study examining the linkages between job loss and sleep disturbances, obesity, and mental health – the Assessing Daily Activity Patterns through Occupational Transitions (ADAPT) study, supported by the National Heart, Lung, and Blood Institute.
This analysis on insomnia was completed before the COVID-19 pandemic began, but it and other analyses soon to be reported are highly relevant to the economic climate, said Patricia Haynes, PhD, principal investigator of ADAPT and a coauthor of the insomnia study, in an interview after the meeting.
Insomnia is a frequent comorbidity of depression and shares many of its symptoms, from increased fatigue to emotional dysregulation and an increased risk of maladaptive coping strategies. “Interestingly, the literature on the stress-generation hypothesis posits that these very symptoms are on the casual pathway between depression and stressful life events,” said Ms. Skobic at the meeting.
In commenting on the study, Krishna M. Sundar, MD, medical director of the Sleep-Wake Center at the University of Utah, Salt Lake City, noted that the analysis did not include any measure of the severity of insomnia. Still, he said, “finding an association [with stress generation] at [just] 3 months with the presence of insomnia disorder is quite interesting.”
There were higher rates of insomnia in the sample than depression, Dr. Haynes said, but the analysis did not control for depression or take it into account.
“We know [from prior research] that stress clearly leads to insomnia. The big [takeaway] here is that insomnia can also lead to more stress,” she said. “It’s important to think of it as a reciprocal relationship. If we can potentially treat insomnia, we may be able to stop that cycle of other stressful events that affect both [the individuals] and others as well.”
Ms. Skobic had no disclosures.
FROM SLEEP 2020
The earlier the better for colchicine post-MI: COLCOT
The earlier the anti-inflammatory drug colchicine is initiated after a myocardial infarction (MI) the greater the benefit, a new COLCOT analysis suggests.
The parent trial was conducted in patients with a recent MI because of the intense inflammation present at that time, and added colchicine 0.5 mg daily to standard care within 30 days following MI.
As previously reported, colchicine significantly reduced the risk of the primary end point – a composite of cardiovascular (CV) death, resuscitated cardiac arrest, MI, stroke, or urgent hospitalization for angina requiring revascularization – by 23% compared with placebo.
This new analysis shows the risk was reduced by 48% in patients receiving colchicine within 3 days of an MI (4.3% vs. 8.3%; adjusted hazard ratio, 0.52; 95% confidence interval, 0.32-0.84, P = .007).
Risk of a secondary efficacy end point – CV death, resuscitated cardiac arrest, MI, or stroke – was reduced by 45% over an average follow up of 22.7 months (3.3% vs 6.1%; adjusted HR, 0.55; 95% CI, 0.32-0.95, P = .031).
“We believe that our results support an early, in-hospital initiation of adjunctive colchicine for post-MI prevention,” Nadia Bouabdallaoui, MD, Montreal Heart Institute, Quebec, Canada, said during an online session devoted to colchicine at the European Society of Cardiology Congress 2020.
Session moderator Massimo Imazio, MD, professor of cardiology at the University of Turin, Italy, said the improved outcomes suggest that earlier treatment is better – a finding that parallels his own experience using colchicine in patients with pericarditis.
“This substudy is very important because this is probably also the year in cardiovascular applications [that] early use of the drug could improve outcomes,” he said.
Positive data have been accumulating for colchicine from COLCOT, LoDoCo, and, most recently, the LoDoCo2 trial, even as another anti-inflammatory drug, methotrexate, flamed out as secondary prevention in the CIRT trial.
The new COLCOT substudy included 4,661 of the 4,745 original patients and examined treatment initiation using three strata: within 0-3 days (n = 1,193), 4-7 days (n = 720), and 8-30 days (n = 2,748). Patients who received treatment within 3 days were slightly younger, more likely to be smokers, and to have a shorter time from MI to randomization (2.1 days vs 5.1 days vs. 20.8 days, respectively).
In the subset receiving treatment within 3 days, those assigned to colchicine had the same number of cardiac deaths as those given placebo (2 vs. 2) but fewer resuscitated cardiac arrests (1 vs. 3), MIs (17 vs. 29), strokes (1 vs. 5), and urgent hospitalizations for angina requiring revascularization (6 vs. 17).
“A larger trial might have allowed for a better assessment of individual endpoints and subgroups,” observed Bouabdallaoui.
Although there is growing support for colchicine, experts caution that the drug many not be for everyone. In COLCOT, 1 in 10 patients were unable to tolerate the drug, largely because of gastrointestinal (GI) issues.
Pharmacogenomics substudy
A second COLCOT substudy aimed to identify genetic markers predictive of colchicine response and to gain insights into the mechanisms behind this response. It included 767 patients treated with colchicine and another 755 treated with placebo – or about one-third the patients in the original trial.
A genome-wide association study did not find a significant association for the primary CV endpoint, although a prespecified subgroup analysis in men identified an interesting region on chromosome 9 (variant: rs10811106), which just missed reaching genomewide significance, said Marie-Pierre Dubé, PhD, director of the Université de Montréal Beaulieu-Saucier Pharmacogenomics Centre at the Montreal Heart Institute.
In addition, the genomewide analysis found two significant regions for GI events: one on chromosome 6 (variant: rs6916345) and one on chromosome 10 (variant: rs74795203).
For each of the identified regions, the researchers then tested the effect of the allele in the placebo group and the interaction between the genetic variant and treatment with colchicine. For the chromosome 9 region in males, there was no effect in the placebo group and a significant interaction in the colchicine group.
For the significant GI event findings, there was a small effect for the chromosome 6 region in the placebo group and a very significant interaction with colchicine, Dubé said. Similarly, there was no effect for the chromosome 10 region in the placebo group and a significant interaction with colchicine.
Additional analyses in stratified patient populations showed that males with the protective allele (CC) for the chromosome 9 region represented 83% of the population. The primary CV endpoint occurred in 3.2% of these men treated with colchicine and 6.3% treated with placebo (HR, 0.46; 95% CI, 0.24 - 0.86).
For the gastrointestinal events, 25% of patients carried the risk allele (AA) for the chromosome 6 region and 36.9% of these had GI events when treated with colchicine versus 18.6% when treated with placebo (HR, 2.42; 95% CI, 1.57-3.72).
Similarly, 13% of individuals carried one or two copies of the risk allele (AG+GG) for the chromosome 10 region and the risk of GI events in these was nearly four times higher with colchicine (47.1% vs. 18.9%; HR, 3.98; 95% CI 2.24-7.07).
Functional genomic analyses of the identified regions were also performed and showed that the chromosome 9 locus overlaps with the SAXO1 gene, a stabilizer of axonemal microtubules 1.
“The leading variant at this locus (rs10811106 C allele) correlated with the expression of the HAUS6 gene, which is involved in microtubule generation from existing microtubules, and may interact with the effect of colchicine, which is known to inhibit microtubule formation,” observed Dubé.
Also, the chromosome 6 locus associated with gastrointestinal events was colocalizing with the Crohn’s disease locus, adding further support for this region.
“The results support potential personalized approaches to inflammation reduction for cardiovascular prevention,” Dubé said.
This is a post hoc subgroup analysis, however, and replication is necessary, ideally in prospective randomized trials, she noted.
The substudy is important because it provides further insights into the link between colchicine and microtubule polymerization, affecting the activation of the inflammasome, session moderator Imazio said.
“Second, it is important because pharmacogenomics can help us to better understand the optimal responder to colchicine and colchicine resistance,” he said. “So it can be useful for personalized medicine, leading to the proper use of the drug for the proper patient.”
COLCOT was supported by the government of Quebec, the Canadian Institutes of Health Research, and philanthropic foundations. Bouabdallaoui has disclosed no relevant financial relationships. Dubé reported grants from the government of Quebec; personal fees from DalCor and GlaxoSmithKline; research support from AstraZeneca, Pfizer, Servier, Sanofi; and minor equity interest in DalCor. Dubé is also coauthor of patents on pharmacogenomics-guided CETP inhibition, and pharmacogenomics markers of response to colchicine.
This article first appeared on Medscape.com.
The earlier the anti-inflammatory drug colchicine is initiated after a myocardial infarction (MI) the greater the benefit, a new COLCOT analysis suggests.
The parent trial was conducted in patients with a recent MI because of the intense inflammation present at that time, and added colchicine 0.5 mg daily to standard care within 30 days following MI.
As previously reported, colchicine significantly reduced the risk of the primary end point – a composite of cardiovascular (CV) death, resuscitated cardiac arrest, MI, stroke, or urgent hospitalization for angina requiring revascularization – by 23% compared with placebo.
This new analysis shows the risk was reduced by 48% in patients receiving colchicine within 3 days of an MI (4.3% vs. 8.3%; adjusted hazard ratio, 0.52; 95% confidence interval, 0.32-0.84, P = .007).
Risk of a secondary efficacy end point – CV death, resuscitated cardiac arrest, MI, or stroke – was reduced by 45% over an average follow up of 22.7 months (3.3% vs 6.1%; adjusted HR, 0.55; 95% CI, 0.32-0.95, P = .031).
“We believe that our results support an early, in-hospital initiation of adjunctive colchicine for post-MI prevention,” Nadia Bouabdallaoui, MD, Montreal Heart Institute, Quebec, Canada, said during an online session devoted to colchicine at the European Society of Cardiology Congress 2020.
Session moderator Massimo Imazio, MD, professor of cardiology at the University of Turin, Italy, said the improved outcomes suggest that earlier treatment is better – a finding that parallels his own experience using colchicine in patients with pericarditis.
“This substudy is very important because this is probably also the year in cardiovascular applications [that] early use of the drug could improve outcomes,” he said.
Positive data have been accumulating for colchicine from COLCOT, LoDoCo, and, most recently, the LoDoCo2 trial, even as another anti-inflammatory drug, methotrexate, flamed out as secondary prevention in the CIRT trial.
The new COLCOT substudy included 4,661 of the 4,745 original patients and examined treatment initiation using three strata: within 0-3 days (n = 1,193), 4-7 days (n = 720), and 8-30 days (n = 2,748). Patients who received treatment within 3 days were slightly younger, more likely to be smokers, and to have a shorter time from MI to randomization (2.1 days vs 5.1 days vs. 20.8 days, respectively).
In the subset receiving treatment within 3 days, those assigned to colchicine had the same number of cardiac deaths as those given placebo (2 vs. 2) but fewer resuscitated cardiac arrests (1 vs. 3), MIs (17 vs. 29), strokes (1 vs. 5), and urgent hospitalizations for angina requiring revascularization (6 vs. 17).
“A larger trial might have allowed for a better assessment of individual endpoints and subgroups,” observed Bouabdallaoui.
Although there is growing support for colchicine, experts caution that the drug many not be for everyone. In COLCOT, 1 in 10 patients were unable to tolerate the drug, largely because of gastrointestinal (GI) issues.
Pharmacogenomics substudy
A second COLCOT substudy aimed to identify genetic markers predictive of colchicine response and to gain insights into the mechanisms behind this response. It included 767 patients treated with colchicine and another 755 treated with placebo – or about one-third the patients in the original trial.
A genome-wide association study did not find a significant association for the primary CV endpoint, although a prespecified subgroup analysis in men identified an interesting region on chromosome 9 (variant: rs10811106), which just missed reaching genomewide significance, said Marie-Pierre Dubé, PhD, director of the Université de Montréal Beaulieu-Saucier Pharmacogenomics Centre at the Montreal Heart Institute.
In addition, the genomewide analysis found two significant regions for GI events: one on chromosome 6 (variant: rs6916345) and one on chromosome 10 (variant: rs74795203).
For each of the identified regions, the researchers then tested the effect of the allele in the placebo group and the interaction between the genetic variant and treatment with colchicine. For the chromosome 9 region in males, there was no effect in the placebo group and a significant interaction in the colchicine group.
For the significant GI event findings, there was a small effect for the chromosome 6 region in the placebo group and a very significant interaction with colchicine, Dubé said. Similarly, there was no effect for the chromosome 10 region in the placebo group and a significant interaction with colchicine.
Additional analyses in stratified patient populations showed that males with the protective allele (CC) for the chromosome 9 region represented 83% of the population. The primary CV endpoint occurred in 3.2% of these men treated with colchicine and 6.3% treated with placebo (HR, 0.46; 95% CI, 0.24 - 0.86).
For the gastrointestinal events, 25% of patients carried the risk allele (AA) for the chromosome 6 region and 36.9% of these had GI events when treated with colchicine versus 18.6% when treated with placebo (HR, 2.42; 95% CI, 1.57-3.72).
Similarly, 13% of individuals carried one or two copies of the risk allele (AG+GG) for the chromosome 10 region and the risk of GI events in these was nearly four times higher with colchicine (47.1% vs. 18.9%; HR, 3.98; 95% CI 2.24-7.07).
Functional genomic analyses of the identified regions were also performed and showed that the chromosome 9 locus overlaps with the SAXO1 gene, a stabilizer of axonemal microtubules 1.
“The leading variant at this locus (rs10811106 C allele) correlated with the expression of the HAUS6 gene, which is involved in microtubule generation from existing microtubules, and may interact with the effect of colchicine, which is known to inhibit microtubule formation,” observed Dubé.
Also, the chromosome 6 locus associated with gastrointestinal events was colocalizing with the Crohn’s disease locus, adding further support for this region.
“The results support potential personalized approaches to inflammation reduction for cardiovascular prevention,” Dubé said.
This is a post hoc subgroup analysis, however, and replication is necessary, ideally in prospective randomized trials, she noted.
The substudy is important because it provides further insights into the link between colchicine and microtubule polymerization, affecting the activation of the inflammasome, session moderator Imazio said.
“Second, it is important because pharmacogenomics can help us to better understand the optimal responder to colchicine and colchicine resistance,” he said. “So it can be useful for personalized medicine, leading to the proper use of the drug for the proper patient.”
COLCOT was supported by the government of Quebec, the Canadian Institutes of Health Research, and philanthropic foundations. Bouabdallaoui has disclosed no relevant financial relationships. Dubé reported grants from the government of Quebec; personal fees from DalCor and GlaxoSmithKline; research support from AstraZeneca, Pfizer, Servier, Sanofi; and minor equity interest in DalCor. Dubé is also coauthor of patents on pharmacogenomics-guided CETP inhibition, and pharmacogenomics markers of response to colchicine.
This article first appeared on Medscape.com.
The earlier the anti-inflammatory drug colchicine is initiated after a myocardial infarction (MI) the greater the benefit, a new COLCOT analysis suggests.
The parent trial was conducted in patients with a recent MI because of the intense inflammation present at that time, and added colchicine 0.5 mg daily to standard care within 30 days following MI.
As previously reported, colchicine significantly reduced the risk of the primary end point – a composite of cardiovascular (CV) death, resuscitated cardiac arrest, MI, stroke, or urgent hospitalization for angina requiring revascularization – by 23% compared with placebo.
This new analysis shows the risk was reduced by 48% in patients receiving colchicine within 3 days of an MI (4.3% vs. 8.3%; adjusted hazard ratio, 0.52; 95% confidence interval, 0.32-0.84, P = .007).
Risk of a secondary efficacy end point – CV death, resuscitated cardiac arrest, MI, or stroke – was reduced by 45% over an average follow up of 22.7 months (3.3% vs 6.1%; adjusted HR, 0.55; 95% CI, 0.32-0.95, P = .031).
“We believe that our results support an early, in-hospital initiation of adjunctive colchicine for post-MI prevention,” Nadia Bouabdallaoui, MD, Montreal Heart Institute, Quebec, Canada, said during an online session devoted to colchicine at the European Society of Cardiology Congress 2020.
Session moderator Massimo Imazio, MD, professor of cardiology at the University of Turin, Italy, said the improved outcomes suggest that earlier treatment is better – a finding that parallels his own experience using colchicine in patients with pericarditis.
“This substudy is very important because this is probably also the year in cardiovascular applications [that] early use of the drug could improve outcomes,” he said.
Positive data have been accumulating for colchicine from COLCOT, LoDoCo, and, most recently, the LoDoCo2 trial, even as another anti-inflammatory drug, methotrexate, flamed out as secondary prevention in the CIRT trial.
The new COLCOT substudy included 4,661 of the 4,745 original patients and examined treatment initiation using three strata: within 0-3 days (n = 1,193), 4-7 days (n = 720), and 8-30 days (n = 2,748). Patients who received treatment within 3 days were slightly younger, more likely to be smokers, and to have a shorter time from MI to randomization (2.1 days vs 5.1 days vs. 20.8 days, respectively).
In the subset receiving treatment within 3 days, those assigned to colchicine had the same number of cardiac deaths as those given placebo (2 vs. 2) but fewer resuscitated cardiac arrests (1 vs. 3), MIs (17 vs. 29), strokes (1 vs. 5), and urgent hospitalizations for angina requiring revascularization (6 vs. 17).
“A larger trial might have allowed for a better assessment of individual endpoints and subgroups,” observed Bouabdallaoui.
Although there is growing support for colchicine, experts caution that the drug many not be for everyone. In COLCOT, 1 in 10 patients were unable to tolerate the drug, largely because of gastrointestinal (GI) issues.
Pharmacogenomics substudy
A second COLCOT substudy aimed to identify genetic markers predictive of colchicine response and to gain insights into the mechanisms behind this response. It included 767 patients treated with colchicine and another 755 treated with placebo – or about one-third the patients in the original trial.
A genome-wide association study did not find a significant association for the primary CV endpoint, although a prespecified subgroup analysis in men identified an interesting region on chromosome 9 (variant: rs10811106), which just missed reaching genomewide significance, said Marie-Pierre Dubé, PhD, director of the Université de Montréal Beaulieu-Saucier Pharmacogenomics Centre at the Montreal Heart Institute.
In addition, the genomewide analysis found two significant regions for GI events: one on chromosome 6 (variant: rs6916345) and one on chromosome 10 (variant: rs74795203).
For each of the identified regions, the researchers then tested the effect of the allele in the placebo group and the interaction between the genetic variant and treatment with colchicine. For the chromosome 9 region in males, there was no effect in the placebo group and a significant interaction in the colchicine group.
For the significant GI event findings, there was a small effect for the chromosome 6 region in the placebo group and a very significant interaction with colchicine, Dubé said. Similarly, there was no effect for the chromosome 10 region in the placebo group and a significant interaction with colchicine.
Additional analyses in stratified patient populations showed that males with the protective allele (CC) for the chromosome 9 region represented 83% of the population. The primary CV endpoint occurred in 3.2% of these men treated with colchicine and 6.3% treated with placebo (HR, 0.46; 95% CI, 0.24 - 0.86).
For the gastrointestinal events, 25% of patients carried the risk allele (AA) for the chromosome 6 region and 36.9% of these had GI events when treated with colchicine versus 18.6% when treated with placebo (HR, 2.42; 95% CI, 1.57-3.72).
Similarly, 13% of individuals carried one or two copies of the risk allele (AG+GG) for the chromosome 10 region and the risk of GI events in these was nearly four times higher with colchicine (47.1% vs. 18.9%; HR, 3.98; 95% CI 2.24-7.07).
Functional genomic analyses of the identified regions were also performed and showed that the chromosome 9 locus overlaps with the SAXO1 gene, a stabilizer of axonemal microtubules 1.
“The leading variant at this locus (rs10811106 C allele) correlated with the expression of the HAUS6 gene, which is involved in microtubule generation from existing microtubules, and may interact with the effect of colchicine, which is known to inhibit microtubule formation,” observed Dubé.
Also, the chromosome 6 locus associated with gastrointestinal events was colocalizing with the Crohn’s disease locus, adding further support for this region.
“The results support potential personalized approaches to inflammation reduction for cardiovascular prevention,” Dubé said.
This is a post hoc subgroup analysis, however, and replication is necessary, ideally in prospective randomized trials, she noted.
The substudy is important because it provides further insights into the link between colchicine and microtubule polymerization, affecting the activation of the inflammasome, session moderator Imazio said.
“Second, it is important because pharmacogenomics can help us to better understand the optimal responder to colchicine and colchicine resistance,” he said. “So it can be useful for personalized medicine, leading to the proper use of the drug for the proper patient.”
COLCOT was supported by the government of Quebec, the Canadian Institutes of Health Research, and philanthropic foundations. Bouabdallaoui has disclosed no relevant financial relationships. Dubé reported grants from the government of Quebec; personal fees from DalCor and GlaxoSmithKline; research support from AstraZeneca, Pfizer, Servier, Sanofi; and minor equity interest in DalCor. Dubé is also coauthor of patents on pharmacogenomics-guided CETP inhibition, and pharmacogenomics markers of response to colchicine.
This article first appeared on Medscape.com.
App for MS aims to capture elusive signals of progression
At the Joint European Committee for Treatment and Research in Multiple Sclerosis–Americas Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS–ACTRIMS) 2020, this year known as MSVirtual2020, researchers at the University Hospital and University of Basel in Switzerland, presented data on their dreaMS app. The investigators are validating the app in a nonblinded cohort of 30 people with MS in the early to middle stages of progression and 30 controls without MS.
The application comprises a series of active tests measuring movement, fine motor skills, cognition, and vision, as well as questionnaires to assess quality of life, walking ability, and fatigue in people with Expanded Disability Status Scale (EDSS) scores of 6.5 or lower. A wrist device, used concurrently with the app, passively monitors subjects’ step count, heart rate, and different measures of activity.
If validated, such smartphone-based “digital biomarkers” will provide clinicians and investigators with a steadier flow of information for assessing MS disease progression and informing clinical decision-making. In June, Ludwig Kappos, MD, the app study’s senior researcher, co-authored an analysis of randomized trial data that argued for discarding the standard categories of relapsing and progressive MS in favor of seeing the disease as a continuum, in which progression can and does occur in the absence of relapses.
The digital biomarker work builds on that more unified view of the disease, Dr. Kappos said in an interview.
Outside of disease exacerbations or relapses, “progression can be very difficult to capture, especially in the first stage of the disease because of compensation in the central nervous system,” he said. “Our ability to see these very slight changes during a neurological examination is limited even if we do it very thoroughly. But by having these more frequent assessments we may be able to.”
Smartphone-gleaned biomarkers may have implications for prognosis and for choice of therapy, Dr. Kappos added. “We expect that these digital biomarkers will be even more sensitive and to be able to recognize before severe deficits are evident who is a candidate for a more intensive treatment and who is not.”
At the MSVirtual2020 congress, Dr. Kappos’s colleagues at the university Johannes Lorscheider, MD, and Yvonne Naegelin, MD, presented their feasibility and acceptance study currently underway in 60 volunteers. One of the concerns the investigators have had was whether engaged users would remain with the app. “We have designed the tests as little challenges to help keep people interested—we want to make these tests as appealing as possible,” Dr. Kappos said.
In this study, the reliability of each test is determined by intra-class correlation and median coefficient of variation. Preliminary reliability testing with healthy controls showed intra-class correlation coefficients of greater than 60% for the digital biomarkers and greater than 80% for at least one in every domain.
Once the best tests are selected and the app is fine-tuned, the group intends to embark on larger studies of the digital biomarkers. The next, planned for 2021, will recruit approximately 400 patients from the Swiss MS cohort, whose 1,000-some MS participants are followed with standardized examination and imaging protocols across healthcare centers.
“This is a very well characterized group of patients who are followed continuously with state-of-the-art neurological examinations, high-end MRI, and blood biomarkers,” Dr. Kappos said. “We want to see if we can add value by using digital biomarkers.”
The dreaMS app project is an independent investigator-initiated venture in cooperation with a technological partner. The study was supported by the Swiss Innovation Agency. The University Hospital Basel has received research funding for clinical trials from a number of pharmaceutical manufacturers.
SOURCE: Lorscheider J, et al. MSVirtual2020. Abstract P0069.
At the Joint European Committee for Treatment and Research in Multiple Sclerosis–Americas Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS–ACTRIMS) 2020, this year known as MSVirtual2020, researchers at the University Hospital and University of Basel in Switzerland, presented data on their dreaMS app. The investigators are validating the app in a nonblinded cohort of 30 people with MS in the early to middle stages of progression and 30 controls without MS.
The application comprises a series of active tests measuring movement, fine motor skills, cognition, and vision, as well as questionnaires to assess quality of life, walking ability, and fatigue in people with Expanded Disability Status Scale (EDSS) scores of 6.5 or lower. A wrist device, used concurrently with the app, passively monitors subjects’ step count, heart rate, and different measures of activity.
If validated, such smartphone-based “digital biomarkers” will provide clinicians and investigators with a steadier flow of information for assessing MS disease progression and informing clinical decision-making. In June, Ludwig Kappos, MD, the app study’s senior researcher, co-authored an analysis of randomized trial data that argued for discarding the standard categories of relapsing and progressive MS in favor of seeing the disease as a continuum, in which progression can and does occur in the absence of relapses.
The digital biomarker work builds on that more unified view of the disease, Dr. Kappos said in an interview.
Outside of disease exacerbations or relapses, “progression can be very difficult to capture, especially in the first stage of the disease because of compensation in the central nervous system,” he said. “Our ability to see these very slight changes during a neurological examination is limited even if we do it very thoroughly. But by having these more frequent assessments we may be able to.”
Smartphone-gleaned biomarkers may have implications for prognosis and for choice of therapy, Dr. Kappos added. “We expect that these digital biomarkers will be even more sensitive and to be able to recognize before severe deficits are evident who is a candidate for a more intensive treatment and who is not.”
At the MSVirtual2020 congress, Dr. Kappos’s colleagues at the university Johannes Lorscheider, MD, and Yvonne Naegelin, MD, presented their feasibility and acceptance study currently underway in 60 volunteers. One of the concerns the investigators have had was whether engaged users would remain with the app. “We have designed the tests as little challenges to help keep people interested—we want to make these tests as appealing as possible,” Dr. Kappos said.
In this study, the reliability of each test is determined by intra-class correlation and median coefficient of variation. Preliminary reliability testing with healthy controls showed intra-class correlation coefficients of greater than 60% for the digital biomarkers and greater than 80% for at least one in every domain.
Once the best tests are selected and the app is fine-tuned, the group intends to embark on larger studies of the digital biomarkers. The next, planned for 2021, will recruit approximately 400 patients from the Swiss MS cohort, whose 1,000-some MS participants are followed with standardized examination and imaging protocols across healthcare centers.
“This is a very well characterized group of patients who are followed continuously with state-of-the-art neurological examinations, high-end MRI, and blood biomarkers,” Dr. Kappos said. “We want to see if we can add value by using digital biomarkers.”
The dreaMS app project is an independent investigator-initiated venture in cooperation with a technological partner. The study was supported by the Swiss Innovation Agency. The University Hospital Basel has received research funding for clinical trials from a number of pharmaceutical manufacturers.
SOURCE: Lorscheider J, et al. MSVirtual2020. Abstract P0069.
At the Joint European Committee for Treatment and Research in Multiple Sclerosis–Americas Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS–ACTRIMS) 2020, this year known as MSVirtual2020, researchers at the University Hospital and University of Basel in Switzerland, presented data on their dreaMS app. The investigators are validating the app in a nonblinded cohort of 30 people with MS in the early to middle stages of progression and 30 controls without MS.
The application comprises a series of active tests measuring movement, fine motor skills, cognition, and vision, as well as questionnaires to assess quality of life, walking ability, and fatigue in people with Expanded Disability Status Scale (EDSS) scores of 6.5 or lower. A wrist device, used concurrently with the app, passively monitors subjects’ step count, heart rate, and different measures of activity.
If validated, such smartphone-based “digital biomarkers” will provide clinicians and investigators with a steadier flow of information for assessing MS disease progression and informing clinical decision-making. In June, Ludwig Kappos, MD, the app study’s senior researcher, co-authored an analysis of randomized trial data that argued for discarding the standard categories of relapsing and progressive MS in favor of seeing the disease as a continuum, in which progression can and does occur in the absence of relapses.
The digital biomarker work builds on that more unified view of the disease, Dr. Kappos said in an interview.
Outside of disease exacerbations or relapses, “progression can be very difficult to capture, especially in the first stage of the disease because of compensation in the central nervous system,” he said. “Our ability to see these very slight changes during a neurological examination is limited even if we do it very thoroughly. But by having these more frequent assessments we may be able to.”
Smartphone-gleaned biomarkers may have implications for prognosis and for choice of therapy, Dr. Kappos added. “We expect that these digital biomarkers will be even more sensitive and to be able to recognize before severe deficits are evident who is a candidate for a more intensive treatment and who is not.”
At the MSVirtual2020 congress, Dr. Kappos’s colleagues at the university Johannes Lorscheider, MD, and Yvonne Naegelin, MD, presented their feasibility and acceptance study currently underway in 60 volunteers. One of the concerns the investigators have had was whether engaged users would remain with the app. “We have designed the tests as little challenges to help keep people interested—we want to make these tests as appealing as possible,” Dr. Kappos said.
In this study, the reliability of each test is determined by intra-class correlation and median coefficient of variation. Preliminary reliability testing with healthy controls showed intra-class correlation coefficients of greater than 60% for the digital biomarkers and greater than 80% for at least one in every domain.
Once the best tests are selected and the app is fine-tuned, the group intends to embark on larger studies of the digital biomarkers. The next, planned for 2021, will recruit approximately 400 patients from the Swiss MS cohort, whose 1,000-some MS participants are followed with standardized examination and imaging protocols across healthcare centers.
“This is a very well characterized group of patients who are followed continuously with state-of-the-art neurological examinations, high-end MRI, and blood biomarkers,” Dr. Kappos said. “We want to see if we can add value by using digital biomarkers.”
The dreaMS app project is an independent investigator-initiated venture in cooperation with a technological partner. The study was supported by the Swiss Innovation Agency. The University Hospital Basel has received research funding for clinical trials from a number of pharmaceutical manufacturers.
SOURCE: Lorscheider J, et al. MSVirtual2020. Abstract P0069.
FROM MSVirtual2020
Satralizumab reduces risk of severe NMOSD relapse
(NMOSD), according to investigators. The drug also was associated with a lower likelihood of using acute relapse therapy.
These results were presented at the Joint European Committee for Treatment and Research in Multiple Sclerosis–Americas Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS–ACTRIMS) 2020, this year known as MSVirtual2020.
NMOSD is characterized by acute relapses that are unpredictable and lead to the accumulation of disability. “Patients with NMOSD often recover poorly from relapses, therefore, the primary goal for disease management is to reduce attack frequency,” said Ingo Kleiter, MD, medical director of Marianne-Strauß-Klinik in Berg, Germany. “In the two phase 3 trials SAkuraSky and SAkuraStar, the IL-6 receptor inhibitor satralizumab was found to significantly reduce the risk of relapses versus placebo.” Satralizumab is a humanized, monoclonal, recycling antibody that targets the interleukin-6 receptor.
Dr. Kleiter and colleagues examined pooled data from the two phase 3 trials of satralizumab to determine the treatment’s effect on relapse severity in patients with NMOSD. Participants in those trials received placebo or 120 mg of satralizumab at weeks 0, 2, 4, and every 4 weeks thereafter.
For their research, the investigators analyzed data from the pooled intention-to-treat population in the double-blind periods of both studies. To evaluate the severity of protocol-defined relapses, they compared patients’ Expanded Disability Status Scale (EDSS) scores at the time of relapse with their scores before the relapse (i.e., their scores at the last scheduled study visit). Using the visual Functional Systems Score (FSS), Dr. Kleiter and colleagues performed a similar analysis on optic neuritis relapses. They categorized a protocol-defined relapse as severe if it entailed a change of two or more points on the EDSS or visual FSS. The investigators conducted Kaplan-Meier analyses to evaluate the time to first severe protocol-defined relapse. They also compared the number of patients receiving acute therapy for any relapse between treatment groups.
Safety profile confirmed
Dr. Kleiter and colleagues included 178 patients in their analyses. A total of 27 of 104 patients (26%) who received satralizumab had a protocol-defined relapse, compared with 34 of 74 patients (46%) who received placebo. The number and proportion of severe protocol-defined relapses were lower in the satralizumab group (5 of 27 events [19%]), compared with the placebo group (12 of 34 events [35%]). In addition, the number and proportion of severe protocol-defined optic neuritis relapses were lower in patients receiving satralizumab (2 of 8 events [25%]), compared with those receiving placebo (5 of 13 events [39%]). Compared with placebo, satralizumab was associated with a 79% reduction in the risk of severe protocol-defined relapse (hazard ratio, 0.21).
A lower proportion of patients receiving satralizumab was prescribed acute relapse therapy (38%), compared with patients receiving placebo (58%). The odds ratio of receiving a prescription of acute relapse therapy was 0.46 among patients receiving satralizumab.
The activity of IL-6 may cause neurologic damage in patients with NMOSD through astrocytic damage, disruption of the blood–brain barrier, and T cell polarization. “It is proposed that through inhibiting IL-6 across these multiple mechanisms, satralizumab reduces the risk and severity of NMOSD attacks,” Dr. Kleiter said.
To date, the rates of infection and serious infection for patients treated with satralizumab in the combined double-blind and open-label extension periods have been consistent with those for patients treated with satralizumab in the double-blind portion. These rates have not increased over time. Satralizumab is administered as a subcutaneous injection every 4 weeks, and treatment can be self-administered at the discretion of the managing physician. “These data provide reassurance to physicians about the overall profile of satralizumab, with respect to efficacy and safety in the longer term,” said Dr. Kleiter.
Does satralizumab differ from other new agents?
The main strength of the study is that sufficient numbers of relapses were available for analysis in the active and control groups, said Achim Berthele, MD, associate professor of neurology at the Technical University of Munich. This allowed the researchers to examine whether satralizumab led to a better outcome after each relapse, which it did. “A weakness is how the severity of relapses was quantified,” said Dr. Berthele. “The EDSS as a measure is not linear, and its functional systems are not clinically equivalent. However, the whole NMOSD community is struggling with this problem.”
The study’s implications for neurologists’ clinical practice are unclear, however. “Although the results presented are encouraging, the data are still too small to say with certainty that satralizumab does indeed improve the outcome of relapses,” said Dr. Berthele. “It is also an open question whether satralizumab differs in this respect from the other new immunotherapeutic agents.”
Investigators must collect further data on the outcome of relapses that occur during treatment with modern immunomodulatory therapy, Dr. Berthele added. Future research could examine whether the new anti-inflammatory immunotherapeutic agents also are suitable drugs for relapse therapy. Another salient question is whether clinical vigilance or relapse therapy in NMOSD has improved in general. “This is what Kleiter and colleagues show as well: The number of severe relapses under placebo was much lower than expected,” said Dr. Berthele.
Chugai/Roche funded the study. Dr. Kleiter has received compensation for consulting, speaking, or serving on advisory boards for Alexion, Biogen, Celgene, Merck, and Roche. Dr. Berthele was not involved in any of the satralizumab trials, but is an investigator and coauthor of the PREVENT trial of eculizumab.
SOURCE: Kleiter I, et al. MSVirtual2020. Abstract FC01.03.
(NMOSD), according to investigators. The drug also was associated with a lower likelihood of using acute relapse therapy.
These results were presented at the Joint European Committee for Treatment and Research in Multiple Sclerosis–Americas Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS–ACTRIMS) 2020, this year known as MSVirtual2020.
NMOSD is characterized by acute relapses that are unpredictable and lead to the accumulation of disability. “Patients with NMOSD often recover poorly from relapses, therefore, the primary goal for disease management is to reduce attack frequency,” said Ingo Kleiter, MD, medical director of Marianne-Strauß-Klinik in Berg, Germany. “In the two phase 3 trials SAkuraSky and SAkuraStar, the IL-6 receptor inhibitor satralizumab was found to significantly reduce the risk of relapses versus placebo.” Satralizumab is a humanized, monoclonal, recycling antibody that targets the interleukin-6 receptor.
Dr. Kleiter and colleagues examined pooled data from the two phase 3 trials of satralizumab to determine the treatment’s effect on relapse severity in patients with NMOSD. Participants in those trials received placebo or 120 mg of satralizumab at weeks 0, 2, 4, and every 4 weeks thereafter.
For their research, the investigators analyzed data from the pooled intention-to-treat population in the double-blind periods of both studies. To evaluate the severity of protocol-defined relapses, they compared patients’ Expanded Disability Status Scale (EDSS) scores at the time of relapse with their scores before the relapse (i.e., their scores at the last scheduled study visit). Using the visual Functional Systems Score (FSS), Dr. Kleiter and colleagues performed a similar analysis on optic neuritis relapses. They categorized a protocol-defined relapse as severe if it entailed a change of two or more points on the EDSS or visual FSS. The investigators conducted Kaplan-Meier analyses to evaluate the time to first severe protocol-defined relapse. They also compared the number of patients receiving acute therapy for any relapse between treatment groups.
Safety profile confirmed
Dr. Kleiter and colleagues included 178 patients in their analyses. A total of 27 of 104 patients (26%) who received satralizumab had a protocol-defined relapse, compared with 34 of 74 patients (46%) who received placebo. The number and proportion of severe protocol-defined relapses were lower in the satralizumab group (5 of 27 events [19%]), compared with the placebo group (12 of 34 events [35%]). In addition, the number and proportion of severe protocol-defined optic neuritis relapses were lower in patients receiving satralizumab (2 of 8 events [25%]), compared with those receiving placebo (5 of 13 events [39%]). Compared with placebo, satralizumab was associated with a 79% reduction in the risk of severe protocol-defined relapse (hazard ratio, 0.21).
A lower proportion of patients receiving satralizumab was prescribed acute relapse therapy (38%), compared with patients receiving placebo (58%). The odds ratio of receiving a prescription of acute relapse therapy was 0.46 among patients receiving satralizumab.
The activity of IL-6 may cause neurologic damage in patients with NMOSD through astrocytic damage, disruption of the blood–brain barrier, and T cell polarization. “It is proposed that through inhibiting IL-6 across these multiple mechanisms, satralizumab reduces the risk and severity of NMOSD attacks,” Dr. Kleiter said.
To date, the rates of infection and serious infection for patients treated with satralizumab in the combined double-blind and open-label extension periods have been consistent with those for patients treated with satralizumab in the double-blind portion. These rates have not increased over time. Satralizumab is administered as a subcutaneous injection every 4 weeks, and treatment can be self-administered at the discretion of the managing physician. “These data provide reassurance to physicians about the overall profile of satralizumab, with respect to efficacy and safety in the longer term,” said Dr. Kleiter.
Does satralizumab differ from other new agents?
The main strength of the study is that sufficient numbers of relapses were available for analysis in the active and control groups, said Achim Berthele, MD, associate professor of neurology at the Technical University of Munich. This allowed the researchers to examine whether satralizumab led to a better outcome after each relapse, which it did. “A weakness is how the severity of relapses was quantified,” said Dr. Berthele. “The EDSS as a measure is not linear, and its functional systems are not clinically equivalent. However, the whole NMOSD community is struggling with this problem.”
The study’s implications for neurologists’ clinical practice are unclear, however. “Although the results presented are encouraging, the data are still too small to say with certainty that satralizumab does indeed improve the outcome of relapses,” said Dr. Berthele. “It is also an open question whether satralizumab differs in this respect from the other new immunotherapeutic agents.”
Investigators must collect further data on the outcome of relapses that occur during treatment with modern immunomodulatory therapy, Dr. Berthele added. Future research could examine whether the new anti-inflammatory immunotherapeutic agents also are suitable drugs for relapse therapy. Another salient question is whether clinical vigilance or relapse therapy in NMOSD has improved in general. “This is what Kleiter and colleagues show as well: The number of severe relapses under placebo was much lower than expected,” said Dr. Berthele.
Chugai/Roche funded the study. Dr. Kleiter has received compensation for consulting, speaking, or serving on advisory boards for Alexion, Biogen, Celgene, Merck, and Roche. Dr. Berthele was not involved in any of the satralizumab trials, but is an investigator and coauthor of the PREVENT trial of eculizumab.
SOURCE: Kleiter I, et al. MSVirtual2020. Abstract FC01.03.
(NMOSD), according to investigators. The drug also was associated with a lower likelihood of using acute relapse therapy.
These results were presented at the Joint European Committee for Treatment and Research in Multiple Sclerosis–Americas Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS–ACTRIMS) 2020, this year known as MSVirtual2020.
NMOSD is characterized by acute relapses that are unpredictable and lead to the accumulation of disability. “Patients with NMOSD often recover poorly from relapses, therefore, the primary goal for disease management is to reduce attack frequency,” said Ingo Kleiter, MD, medical director of Marianne-Strauß-Klinik in Berg, Germany. “In the two phase 3 trials SAkuraSky and SAkuraStar, the IL-6 receptor inhibitor satralizumab was found to significantly reduce the risk of relapses versus placebo.” Satralizumab is a humanized, monoclonal, recycling antibody that targets the interleukin-6 receptor.
Dr. Kleiter and colleagues examined pooled data from the two phase 3 trials of satralizumab to determine the treatment’s effect on relapse severity in patients with NMOSD. Participants in those trials received placebo or 120 mg of satralizumab at weeks 0, 2, 4, and every 4 weeks thereafter.
For their research, the investigators analyzed data from the pooled intention-to-treat population in the double-blind periods of both studies. To evaluate the severity of protocol-defined relapses, they compared patients’ Expanded Disability Status Scale (EDSS) scores at the time of relapse with their scores before the relapse (i.e., their scores at the last scheduled study visit). Using the visual Functional Systems Score (FSS), Dr. Kleiter and colleagues performed a similar analysis on optic neuritis relapses. They categorized a protocol-defined relapse as severe if it entailed a change of two or more points on the EDSS or visual FSS. The investigators conducted Kaplan-Meier analyses to evaluate the time to first severe protocol-defined relapse. They also compared the number of patients receiving acute therapy for any relapse between treatment groups.
Safety profile confirmed
Dr. Kleiter and colleagues included 178 patients in their analyses. A total of 27 of 104 patients (26%) who received satralizumab had a protocol-defined relapse, compared with 34 of 74 patients (46%) who received placebo. The number and proportion of severe protocol-defined relapses were lower in the satralizumab group (5 of 27 events [19%]), compared with the placebo group (12 of 34 events [35%]). In addition, the number and proportion of severe protocol-defined optic neuritis relapses were lower in patients receiving satralizumab (2 of 8 events [25%]), compared with those receiving placebo (5 of 13 events [39%]). Compared with placebo, satralizumab was associated with a 79% reduction in the risk of severe protocol-defined relapse (hazard ratio, 0.21).
A lower proportion of patients receiving satralizumab was prescribed acute relapse therapy (38%), compared with patients receiving placebo (58%). The odds ratio of receiving a prescription of acute relapse therapy was 0.46 among patients receiving satralizumab.
The activity of IL-6 may cause neurologic damage in patients with NMOSD through astrocytic damage, disruption of the blood–brain barrier, and T cell polarization. “It is proposed that through inhibiting IL-6 across these multiple mechanisms, satralizumab reduces the risk and severity of NMOSD attacks,” Dr. Kleiter said.
To date, the rates of infection and serious infection for patients treated with satralizumab in the combined double-blind and open-label extension periods have been consistent with those for patients treated with satralizumab in the double-blind portion. These rates have not increased over time. Satralizumab is administered as a subcutaneous injection every 4 weeks, and treatment can be self-administered at the discretion of the managing physician. “These data provide reassurance to physicians about the overall profile of satralizumab, with respect to efficacy and safety in the longer term,” said Dr. Kleiter.
Does satralizumab differ from other new agents?
The main strength of the study is that sufficient numbers of relapses were available for analysis in the active and control groups, said Achim Berthele, MD, associate professor of neurology at the Technical University of Munich. This allowed the researchers to examine whether satralizumab led to a better outcome after each relapse, which it did. “A weakness is how the severity of relapses was quantified,” said Dr. Berthele. “The EDSS as a measure is not linear, and its functional systems are not clinically equivalent. However, the whole NMOSD community is struggling with this problem.”
The study’s implications for neurologists’ clinical practice are unclear, however. “Although the results presented are encouraging, the data are still too small to say with certainty that satralizumab does indeed improve the outcome of relapses,” said Dr. Berthele. “It is also an open question whether satralizumab differs in this respect from the other new immunotherapeutic agents.”
Investigators must collect further data on the outcome of relapses that occur during treatment with modern immunomodulatory therapy, Dr. Berthele added. Future research could examine whether the new anti-inflammatory immunotherapeutic agents also are suitable drugs for relapse therapy. Another salient question is whether clinical vigilance or relapse therapy in NMOSD has improved in general. “This is what Kleiter and colleagues show as well: The number of severe relapses under placebo was much lower than expected,” said Dr. Berthele.
Chugai/Roche funded the study. Dr. Kleiter has received compensation for consulting, speaking, or serving on advisory boards for Alexion, Biogen, Celgene, Merck, and Roche. Dr. Berthele was not involved in any of the satralizumab trials, but is an investigator and coauthor of the PREVENT trial of eculizumab.
SOURCE: Kleiter I, et al. MSVirtual2020. Abstract FC01.03.
FROM MSVirtual2020
Lessons for patients with MS and COVID-19
Two important lessons about managing patients with multiple sclerosis (MS) and COVID-19 have emerged from a hospital clinic in Madrid that managed COVID-infected patients with MS through the peak of the pandemic: Combined polymeric chain reaction and serology testing helped avoid disease reactivation in asymptomatic carriers during the pandemic peak, although after the peak PCR alone proved just as effective; and
Virginia Meca-Lallana, MD, a neurologist and coordinator of the demyelinating diseases unit at the Hospital of the University of the Princess in Madrid, and colleagues presented their findings in two posters at the Joint European Committee for Treatment and Research in Multiple Sclerosis-Americas Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS-ACTRIMS) 2020, this year known as MSVirtual2020.
“MS treatments don’t seem to make the prognosis of COVID-19 worse, but it is very important to evaluate other risk factors,” Dr. Meca-Lallana said in an interview. “MS treatments prevent the patients’ disability, and it is very important not to stop them if it isn’t necessary.”
The results arose from a multidisciplinary safety protocol involving neurology, microbiology, and preventive medicine that the University of Princess physicians developed to keep MS stable in patients diagnosed with SARS-CoV-2.
The researchers obtained 152 PCR nasopharyngeal swabs and 140 serology tests in 90 patients with MS over 3 months before starting a variety of MS treatments: Natalizumab (96 tests), ocrelizumab (36), rituximab (3), methylprednisolone (7), cladribine (4), and dimethyl fumarate (3). The protocol identified 7 asymptomatic carriers—7.8% of the total population—5 of whom had positive immunoglobulin M and G serology. The study also confirmed 5 patients with positive IgM+IgG serology post-infection, but no COVID-19 reactivations were detected after implementation of the protocol.
“The safety protocol reached its objective of avoiding disease reactivation and clinical activation in asymptomatic carriers,” Dr. Meca-Lallana said.
The second poster she presented reported on the real-world experience with SARS-CoV-2 in the MS unit at her hospital. The observational, prospective study included 41 cases, 38 of which were relapsing-remitting MS and the remainder progressive MS. The patients had MS for an average of 9 years.
“We need more patients to draw more robust conclusions, but in our patients, MS treatments seem safe in this situation,” Dr. Meca-Lallana said. “We did not discontinue treatments, and after our first results, we only delayed treatments in patients with any additional comorbidity or when coming to the hospital was not safe.”
A total of 39 patients were taking disease-modifying therapies (DMTs): 46.3% with oral agents, 39% with monoclonal antibodies, and 10% with injectable agents; 27 patients were previously treated with other DMTs. The median Expanded Disability Status Scale (EDSS) was 2.5, and 11 patients had clinical activity the previous year. Eighteen cases were confirmed by PCR or serology, or both, and 23 were diagnosed clinically.
Among the patients with MS and COVID-19, 17% were admitted to the hospital. Six patients had pneumonia, but none required admission to the intensive care unit, and no deaths occurred. Three patients had other comorbidities. Admitted patients tended to be older and had higher EDSS scores, although the difference was not statistically significant. MS worsened in 7 patients, and 10 patients stopped or paused DMTs because of the infection.
“Multiple sclerosis is a weakening illness,” Dr. Meca-Lallana said. “MS treatments do not seem to make the prognosis of COVID-19 worse, but it is very important to evaluate other risk factors.”
The SARS-CoV-2 infection does not seem to result in a more aggressive form of the disease in MS patients, and selective immunosuppression may improve their outcomes, she noted.
“MS treatments avoid the patient’s disability,” the investigator added, “and it is very important not to stop them if it isn’t necessary.”
Dr. Meca-Lallana had no relevant financial disclosures.
Two important lessons about managing patients with multiple sclerosis (MS) and COVID-19 have emerged from a hospital clinic in Madrid that managed COVID-infected patients with MS through the peak of the pandemic: Combined polymeric chain reaction and serology testing helped avoid disease reactivation in asymptomatic carriers during the pandemic peak, although after the peak PCR alone proved just as effective; and
Virginia Meca-Lallana, MD, a neurologist and coordinator of the demyelinating diseases unit at the Hospital of the University of the Princess in Madrid, and colleagues presented their findings in two posters at the Joint European Committee for Treatment and Research in Multiple Sclerosis-Americas Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS-ACTRIMS) 2020, this year known as MSVirtual2020.
“MS treatments don’t seem to make the prognosis of COVID-19 worse, but it is very important to evaluate other risk factors,” Dr. Meca-Lallana said in an interview. “MS treatments prevent the patients’ disability, and it is very important not to stop them if it isn’t necessary.”
The results arose from a multidisciplinary safety protocol involving neurology, microbiology, and preventive medicine that the University of Princess physicians developed to keep MS stable in patients diagnosed with SARS-CoV-2.
The researchers obtained 152 PCR nasopharyngeal swabs and 140 serology tests in 90 patients with MS over 3 months before starting a variety of MS treatments: Natalizumab (96 tests), ocrelizumab (36), rituximab (3), methylprednisolone (7), cladribine (4), and dimethyl fumarate (3). The protocol identified 7 asymptomatic carriers—7.8% of the total population—5 of whom had positive immunoglobulin M and G serology. The study also confirmed 5 patients with positive IgM+IgG serology post-infection, but no COVID-19 reactivations were detected after implementation of the protocol.
“The safety protocol reached its objective of avoiding disease reactivation and clinical activation in asymptomatic carriers,” Dr. Meca-Lallana said.
The second poster she presented reported on the real-world experience with SARS-CoV-2 in the MS unit at her hospital. The observational, prospective study included 41 cases, 38 of which were relapsing-remitting MS and the remainder progressive MS. The patients had MS for an average of 9 years.
“We need more patients to draw more robust conclusions, but in our patients, MS treatments seem safe in this situation,” Dr. Meca-Lallana said. “We did not discontinue treatments, and after our first results, we only delayed treatments in patients with any additional comorbidity or when coming to the hospital was not safe.”
A total of 39 patients were taking disease-modifying therapies (DMTs): 46.3% with oral agents, 39% with monoclonal antibodies, and 10% with injectable agents; 27 patients were previously treated with other DMTs. The median Expanded Disability Status Scale (EDSS) was 2.5, and 11 patients had clinical activity the previous year. Eighteen cases were confirmed by PCR or serology, or both, and 23 were diagnosed clinically.
Among the patients with MS and COVID-19, 17% were admitted to the hospital. Six patients had pneumonia, but none required admission to the intensive care unit, and no deaths occurred. Three patients had other comorbidities. Admitted patients tended to be older and had higher EDSS scores, although the difference was not statistically significant. MS worsened in 7 patients, and 10 patients stopped or paused DMTs because of the infection.
“Multiple sclerosis is a weakening illness,” Dr. Meca-Lallana said. “MS treatments do not seem to make the prognosis of COVID-19 worse, but it is very important to evaluate other risk factors.”
The SARS-CoV-2 infection does not seem to result in a more aggressive form of the disease in MS patients, and selective immunosuppression may improve their outcomes, she noted.
“MS treatments avoid the patient’s disability,” the investigator added, “and it is very important not to stop them if it isn’t necessary.”
Dr. Meca-Lallana had no relevant financial disclosures.
Two important lessons about managing patients with multiple sclerosis (MS) and COVID-19 have emerged from a hospital clinic in Madrid that managed COVID-infected patients with MS through the peak of the pandemic: Combined polymeric chain reaction and serology testing helped avoid disease reactivation in asymptomatic carriers during the pandemic peak, although after the peak PCR alone proved just as effective; and
Virginia Meca-Lallana, MD, a neurologist and coordinator of the demyelinating diseases unit at the Hospital of the University of the Princess in Madrid, and colleagues presented their findings in two posters at the Joint European Committee for Treatment and Research in Multiple Sclerosis-Americas Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS-ACTRIMS) 2020, this year known as MSVirtual2020.
“MS treatments don’t seem to make the prognosis of COVID-19 worse, but it is very important to evaluate other risk factors,” Dr. Meca-Lallana said in an interview. “MS treatments prevent the patients’ disability, and it is very important not to stop them if it isn’t necessary.”
The results arose from a multidisciplinary safety protocol involving neurology, microbiology, and preventive medicine that the University of Princess physicians developed to keep MS stable in patients diagnosed with SARS-CoV-2.
The researchers obtained 152 PCR nasopharyngeal swabs and 140 serology tests in 90 patients with MS over 3 months before starting a variety of MS treatments: Natalizumab (96 tests), ocrelizumab (36), rituximab (3), methylprednisolone (7), cladribine (4), and dimethyl fumarate (3). The protocol identified 7 asymptomatic carriers—7.8% of the total population—5 of whom had positive immunoglobulin M and G serology. The study also confirmed 5 patients with positive IgM+IgG serology post-infection, but no COVID-19 reactivations were detected after implementation of the protocol.
“The safety protocol reached its objective of avoiding disease reactivation and clinical activation in asymptomatic carriers,” Dr. Meca-Lallana said.
The second poster she presented reported on the real-world experience with SARS-CoV-2 in the MS unit at her hospital. The observational, prospective study included 41 cases, 38 of which were relapsing-remitting MS and the remainder progressive MS. The patients had MS for an average of 9 years.
“We need more patients to draw more robust conclusions, but in our patients, MS treatments seem safe in this situation,” Dr. Meca-Lallana said. “We did not discontinue treatments, and after our first results, we only delayed treatments in patients with any additional comorbidity or when coming to the hospital was not safe.”
A total of 39 patients were taking disease-modifying therapies (DMTs): 46.3% with oral agents, 39% with monoclonal antibodies, and 10% with injectable agents; 27 patients were previously treated with other DMTs. The median Expanded Disability Status Scale (EDSS) was 2.5, and 11 patients had clinical activity the previous year. Eighteen cases were confirmed by PCR or serology, or both, and 23 were diagnosed clinically.
Among the patients with MS and COVID-19, 17% were admitted to the hospital. Six patients had pneumonia, but none required admission to the intensive care unit, and no deaths occurred. Three patients had other comorbidities. Admitted patients tended to be older and had higher EDSS scores, although the difference was not statistically significant. MS worsened in 7 patients, and 10 patients stopped or paused DMTs because of the infection.
“Multiple sclerosis is a weakening illness,” Dr. Meca-Lallana said. “MS treatments do not seem to make the prognosis of COVID-19 worse, but it is very important to evaluate other risk factors.”
The SARS-CoV-2 infection does not seem to result in a more aggressive form of the disease in MS patients, and selective immunosuppression may improve their outcomes, she noted.
“MS treatments avoid the patient’s disability,” the investigator added, “and it is very important not to stop them if it isn’t necessary.”
Dr. Meca-Lallana had no relevant financial disclosures.
FROM MSVirtual2020








