User login
Similar brain atrophy in obesity and Alzheimer’s disease
Comparisons of MRI scans for more than 1,000 participants indicate correlations between the two conditions, especially in areas of gray matter thinning, suggesting that managing excess weight might slow cognitive decline and lower the risk for AD, according to the researchers.
However, brain maps of obesity did not correlate with maps of amyloid or tau protein accumulation.
“The fact that obesity-related brain atrophy did not correlate with the distribution of amyloid and tau proteins in AD was not what we expected,” study author Filip Morys, PhD, a postdoctoral researcher at McGill University, Montreal, said in an interview. “But it might just show that the specific mechanisms underpinning obesity- and Alzheimer’s disease–related neurodegeneration are different. This remains to be confirmed.”
The study was published in the Journal of Alzheimer’s Disease.
Cortical Thinning
The current study was prompted by the team’s earlier study, which showed that obesity-related neurodegeneration patterns were visually similar to those of AD, said Dr. Morys. “It was known previously that obesity is a risk factor for AD, but we wanted to directly compare brain atrophy patterns in both, which is what we did in this new study.”
The researchers analyzed data from a pooled sample of more than 1,300 participants. From the ADNI database, the researchers selected participants with AD and age- and sex-matched cognitively healthy controls. From the UK Biobank, the researchers drew a sample of lean, overweight, and obese participants without neurologic disease.
To determine how the weight status of patients with AD affects the correspondence between AD and obesity maps, they categorized participants with AD and healthy controls from the ADNI database into lean, overweight, and obese subgroups.
Then, to investigate mechanisms that might drive the similarities between obesity-related brain atrophy and AD-related amyloid-beta accumulation, they looked for overlapping areas in PET brain maps between patients with these outcomes.
The investigations showed that obesity maps were highly correlated with AD maps, but not with amyloid-beta or tau protein maps. The researchers also found significant correlations between obesity and the lean individuals with AD.
Brain regions with the highest similarities between obesity and AD were located mainly in the left temporal and bilateral prefrontal cortices.
“Our research confirms that obesity-related gray matter atrophy resembles that of AD,” the authors concluded. “Excess weight management could lead to improved health outcomes, slow down cognitive decline in aging, and lower the risk for AD.”
Upcoming research “will focus on investigating how weight loss can affect the risk for AD, other dementias, and cognitive decline in general,” said Dr. Morys. “At this point, our study suggests that obesity prevention, weight loss, but also decreasing other metabolic risk factors related to obesity, such as type-2 diabetes or hypertension, might reduce the risk for AD and have beneficial effects on cognition.”
Lifestyle habits
Commenting on the findings, Claire Sexton, DPhil, vice president of scientific programs and outreach at the Alzheimer’s Association, cautioned that a single cross-sectional study isn’t conclusive. “Previous studies have illustrated that the relationship between obesity and dementia is complex. Growing evidence indicates that people can reduce their risk of cognitive decline by adopting key lifestyle habits, like regular exercise, a heart-healthy diet and staying socially and cognitively engaged.”
The Alzheimer’s Association is leading a 2-year clinical trial, U.S. Pointer, to study how targeting these risk factors in combination may reduce risk for cognitive decline in older adults.
The work was supported by a Foundation Scheme award from the Canadian Institutes of Health Research. Dr. Morys received a postdoctoral fellowship from Fonds de Recherche du Quebec – Santé. Data collection and sharing were funded by the Alzheimer’s Disease Neuroimaging Initiative, the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and multiple pharmaceutical companies and other private sector organizations. Dr. Morys and Dr. Sexton reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Comparisons of MRI scans for more than 1,000 participants indicate correlations between the two conditions, especially in areas of gray matter thinning, suggesting that managing excess weight might slow cognitive decline and lower the risk for AD, according to the researchers.
However, brain maps of obesity did not correlate with maps of amyloid or tau protein accumulation.
“The fact that obesity-related brain atrophy did not correlate with the distribution of amyloid and tau proteins in AD was not what we expected,” study author Filip Morys, PhD, a postdoctoral researcher at McGill University, Montreal, said in an interview. “But it might just show that the specific mechanisms underpinning obesity- and Alzheimer’s disease–related neurodegeneration are different. This remains to be confirmed.”
The study was published in the Journal of Alzheimer’s Disease.
Cortical Thinning
The current study was prompted by the team’s earlier study, which showed that obesity-related neurodegeneration patterns were visually similar to those of AD, said Dr. Morys. “It was known previously that obesity is a risk factor for AD, but we wanted to directly compare brain atrophy patterns in both, which is what we did in this new study.”
The researchers analyzed data from a pooled sample of more than 1,300 participants. From the ADNI database, the researchers selected participants with AD and age- and sex-matched cognitively healthy controls. From the UK Biobank, the researchers drew a sample of lean, overweight, and obese participants without neurologic disease.
To determine how the weight status of patients with AD affects the correspondence between AD and obesity maps, they categorized participants with AD and healthy controls from the ADNI database into lean, overweight, and obese subgroups.
Then, to investigate mechanisms that might drive the similarities between obesity-related brain atrophy and AD-related amyloid-beta accumulation, they looked for overlapping areas in PET brain maps between patients with these outcomes.
The investigations showed that obesity maps were highly correlated with AD maps, but not with amyloid-beta or tau protein maps. The researchers also found significant correlations between obesity and the lean individuals with AD.
Brain regions with the highest similarities between obesity and AD were located mainly in the left temporal and bilateral prefrontal cortices.
“Our research confirms that obesity-related gray matter atrophy resembles that of AD,” the authors concluded. “Excess weight management could lead to improved health outcomes, slow down cognitive decline in aging, and lower the risk for AD.”
Upcoming research “will focus on investigating how weight loss can affect the risk for AD, other dementias, and cognitive decline in general,” said Dr. Morys. “At this point, our study suggests that obesity prevention, weight loss, but also decreasing other metabolic risk factors related to obesity, such as type-2 diabetes or hypertension, might reduce the risk for AD and have beneficial effects on cognition.”
Lifestyle habits
Commenting on the findings, Claire Sexton, DPhil, vice president of scientific programs and outreach at the Alzheimer’s Association, cautioned that a single cross-sectional study isn’t conclusive. “Previous studies have illustrated that the relationship between obesity and dementia is complex. Growing evidence indicates that people can reduce their risk of cognitive decline by adopting key lifestyle habits, like regular exercise, a heart-healthy diet and staying socially and cognitively engaged.”
The Alzheimer’s Association is leading a 2-year clinical trial, U.S. Pointer, to study how targeting these risk factors in combination may reduce risk for cognitive decline in older adults.
The work was supported by a Foundation Scheme award from the Canadian Institutes of Health Research. Dr. Morys received a postdoctoral fellowship from Fonds de Recherche du Quebec – Santé. Data collection and sharing were funded by the Alzheimer’s Disease Neuroimaging Initiative, the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and multiple pharmaceutical companies and other private sector organizations. Dr. Morys and Dr. Sexton reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Comparisons of MRI scans for more than 1,000 participants indicate correlations between the two conditions, especially in areas of gray matter thinning, suggesting that managing excess weight might slow cognitive decline and lower the risk for AD, according to the researchers.
However, brain maps of obesity did not correlate with maps of amyloid or tau protein accumulation.
“The fact that obesity-related brain atrophy did not correlate with the distribution of amyloid and tau proteins in AD was not what we expected,” study author Filip Morys, PhD, a postdoctoral researcher at McGill University, Montreal, said in an interview. “But it might just show that the specific mechanisms underpinning obesity- and Alzheimer’s disease–related neurodegeneration are different. This remains to be confirmed.”
The study was published in the Journal of Alzheimer’s Disease.
Cortical Thinning
The current study was prompted by the team’s earlier study, which showed that obesity-related neurodegeneration patterns were visually similar to those of AD, said Dr. Morys. “It was known previously that obesity is a risk factor for AD, but we wanted to directly compare brain atrophy patterns in both, which is what we did in this new study.”
The researchers analyzed data from a pooled sample of more than 1,300 participants. From the ADNI database, the researchers selected participants with AD and age- and sex-matched cognitively healthy controls. From the UK Biobank, the researchers drew a sample of lean, overweight, and obese participants without neurologic disease.
To determine how the weight status of patients with AD affects the correspondence between AD and obesity maps, they categorized participants with AD and healthy controls from the ADNI database into lean, overweight, and obese subgroups.
Then, to investigate mechanisms that might drive the similarities between obesity-related brain atrophy and AD-related amyloid-beta accumulation, they looked for overlapping areas in PET brain maps between patients with these outcomes.
The investigations showed that obesity maps were highly correlated with AD maps, but not with amyloid-beta or tau protein maps. The researchers also found significant correlations between obesity and the lean individuals with AD.
Brain regions with the highest similarities between obesity and AD were located mainly in the left temporal and bilateral prefrontal cortices.
“Our research confirms that obesity-related gray matter atrophy resembles that of AD,” the authors concluded. “Excess weight management could lead to improved health outcomes, slow down cognitive decline in aging, and lower the risk for AD.”
Upcoming research “will focus on investigating how weight loss can affect the risk for AD, other dementias, and cognitive decline in general,” said Dr. Morys. “At this point, our study suggests that obesity prevention, weight loss, but also decreasing other metabolic risk factors related to obesity, such as type-2 diabetes or hypertension, might reduce the risk for AD and have beneficial effects on cognition.”
Lifestyle habits
Commenting on the findings, Claire Sexton, DPhil, vice president of scientific programs and outreach at the Alzheimer’s Association, cautioned that a single cross-sectional study isn’t conclusive. “Previous studies have illustrated that the relationship between obesity and dementia is complex. Growing evidence indicates that people can reduce their risk of cognitive decline by adopting key lifestyle habits, like regular exercise, a heart-healthy diet and staying socially and cognitively engaged.”
The Alzheimer’s Association is leading a 2-year clinical trial, U.S. Pointer, to study how targeting these risk factors in combination may reduce risk for cognitive decline in older adults.
The work was supported by a Foundation Scheme award from the Canadian Institutes of Health Research. Dr. Morys received a postdoctoral fellowship from Fonds de Recherche du Quebec – Santé. Data collection and sharing were funded by the Alzheimer’s Disease Neuroimaging Initiative, the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and multiple pharmaceutical companies and other private sector organizations. Dr. Morys and Dr. Sexton reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF ALZHEIMER’S DISEASE
Psychiatric illnesses share common brain network
Investigators used coordinate and lesion network mapping to assess whether there was a shared brain network common to multiple psychiatric disorders. In a meta-analysis of almost 200 studies encompassing more than 15,000 individuals, they found that atrophy coordinates across these six psychiatric conditions all mapped to a common brain network.
Moreover, lesion damage to this network in patients with penetrating head trauma correlated with the number of psychiatric illnesses that the patients were diagnosed with post trauma.
The findings have “bigger-picture potential implications,” lead author Joseph Taylor, MD, PhD, medical director of transcranial magnetic stimulation at Brigham and Women’s Hospital’s Center for Brain Circuit Therapeutics, Boston, told this news organization.
“In psychiatry, we talk about symptoms and define our disorders based on symptom checklists, which are fairly reliable but don’t have neurobiological underpinnings,” said Dr. Taylor, who is also an associate psychiatrist in Brigham’s department of psychiatry.
By contrast, “in neurology, we ask: ‘Where is the lesion?’ Studying brain networks could potentially help us diagnose and treat people with psychiatric illness more effectively, just as we treat neurological disorders,” he added.
The findings were published online in Nature Human Behavior.
Beyond symptom checklists
Dr. Taylor noted that, in the field of psychiatry, “we often study disorders in isolation,” such as generalized anxiety disorder and major depressive disorder.
“But what see clinically is that half of patients meet the criteria for more than one psychiatric disorder,” he said. “It can be difficult to diagnose and treat these patients, and there are worse treatment outcomes.”
There is also a “discrepancy” between how these disorders are studied (one at a time) and how patients are treated in clinic, Dr. Taylor noted. And there is increasing evidence that psychiatric disorders may share a common neurobiology.
This “highlights the possibility of potentially developing transdiagnostic treatments based on common neurobiology, not just symptom checklists,” Dr. Taylor said.
Prior work “has attempted to map abnormalities to common brain regions rather than to a common brain network,” the investigators wrote. Moreover, “prior studies have rarely tested specificity by comparing psychiatric disorders to other brain disorders.”
In the current study, the researchers used “morphometric brain lesion datasets coupled with a wiring diagram of the human brain to derive a convergent brain network for psychiatric illness.”
They analyzed four large published datasets. Dataset 1 was sourced from an activation likelihood estimation meta-analysis (ALE) of whole-brain voxel-based studies that compared patients with psychiatric disorders such as schizophrenia, BD, depression, addiction, OCD, and anxiety to healthy controls (n = 193 studies; 15,892 individuals in total).
Dataset 2 was drawn from published neuroimaging studies involving patients with Alzheimer’s disease (AD) and other neurodegenerative conditions (n = 72 studies). They reported coordinates regarding which patients with these disorders had more atrophy compared with control persons.
Dataset 3 was sourced from the Vietnam Head Injury study, which followed veterans with and those without penetrating head injuries (n = 194 veterans with injuries). Dataset 4 was sourced from published neurosurgical ablation coordinates for depression.
Shared neurobiology
Upon analyzing dataset 1, the researchers found decreased gray matter in the bilateral anterior insula, dorsal anterior cingulate cortex, dorsomedial prefrontal cortex, thalamus, amygdala, hippocampus, and parietal operculum – findings that are “consistent with prior work.”
However, fewer than 35% of the studies contributed to any single cluster; and no cluster was specific to psychiatric versus neurodegenerative coordinates (drawn from dataset 2).
On the other hand, coordinate network mapping yielded “more statistically robust” (P < .001) results, which were found in 85% of the studies. “Psychiatric atrophy coordinates were functionally connected to the same network of brain regions,” the researchers reported.
This network was defined by two types of connectivity, positive and negative.
“The topography of this transdiagnostic network was independent of the statistical threshold and specific to psychiatric (vs. neurodegenerative) disorders, with the strongest peak occurring in the posterior parietal cortex (Brodmann Area 7) near the intraparietal sulcus,” the investigators wrote.
When lesions from dataset 3 were overlaid onto the ALE map and the transdiagnostic network in order to evaluate whether damage to either map correlated with number of post-lesion psychiatric diagnosis, results showed no evidence of a correlation between psychiatric comorbidity and damage on the ALE map (Pearson r, 0.02; P = .766).
However, when the same approach was applied to the transdiagnostic network, a statistically significant correlation was found between psychiatric comorbidity and lesion damage (Pearson r, –0.21; P = .01). A multiple regression model showed that the transdiagnostic, but not the ALE, network “independently predicted the number of post-lesion psychiatric diagnoses” (P = .003 vs. P = .1), the investigators reported.
All four neurosurgical ablative targets for psychiatric disorders found on analysis of dataset 4 “intersected” and aligned with the transdiagnostic network.
“The study does not immediately impact clinical practice, but it would be helpful for practicing clinicians to know that psychiatric disorders commonly co-occur and might share common neurobiology and a convergent brain network,” Dr. Taylor said.
“Future work based on our findings could potentially influence clinical trials and clinical practice, especially in the area of brain stimulation,” he added.
‘Exciting new targets’
In a comment, Desmond Oathes, PhD, associate director, Center for Neuromodulation and Stress, University of Pennsylvania, Philadelphia, said the “next step in the science is to combine individual brain imaging, aka, ‘individualized connectomes,’ with these promising group maps to determine something meaningful at the individual patient level.”
Dr. Oathes, who is also a faculty clinician at the Center for the Treatment and Study of Anxiety and was not involved with the study, noted that an open question is whether the brain volume abnormalities/atrophy “can be changed with treatment and in what direction.”
A “strong take-home message from this paper is that brain volume measures from single coordinates are noisy as measures of psychiatric abnormality, whereas network effects seem to be especially sensitive for capturing these effects,” Dr. Oathes said.
The “abnormal networks across these disorders do not fit easily into well-known networks from healthy participants. However, they map well onto other databases relevant to psychiatric disorders and offer exciting new potential targets for prospective treatment studies,” he added.
The investigators received no specific funding for this work. Dr. Taylor reported no relevant financial relationships. Dr. Oathes reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Investigators used coordinate and lesion network mapping to assess whether there was a shared brain network common to multiple psychiatric disorders. In a meta-analysis of almost 200 studies encompassing more than 15,000 individuals, they found that atrophy coordinates across these six psychiatric conditions all mapped to a common brain network.
Moreover, lesion damage to this network in patients with penetrating head trauma correlated with the number of psychiatric illnesses that the patients were diagnosed with post trauma.
The findings have “bigger-picture potential implications,” lead author Joseph Taylor, MD, PhD, medical director of transcranial magnetic stimulation at Brigham and Women’s Hospital’s Center for Brain Circuit Therapeutics, Boston, told this news organization.
“In psychiatry, we talk about symptoms and define our disorders based on symptom checklists, which are fairly reliable but don’t have neurobiological underpinnings,” said Dr. Taylor, who is also an associate psychiatrist in Brigham’s department of psychiatry.
By contrast, “in neurology, we ask: ‘Where is the lesion?’ Studying brain networks could potentially help us diagnose and treat people with psychiatric illness more effectively, just as we treat neurological disorders,” he added.
The findings were published online in Nature Human Behavior.
Beyond symptom checklists
Dr. Taylor noted that, in the field of psychiatry, “we often study disorders in isolation,” such as generalized anxiety disorder and major depressive disorder.
“But what see clinically is that half of patients meet the criteria for more than one psychiatric disorder,” he said. “It can be difficult to diagnose and treat these patients, and there are worse treatment outcomes.”
There is also a “discrepancy” between how these disorders are studied (one at a time) and how patients are treated in clinic, Dr. Taylor noted. And there is increasing evidence that psychiatric disorders may share a common neurobiology.
This “highlights the possibility of potentially developing transdiagnostic treatments based on common neurobiology, not just symptom checklists,” Dr. Taylor said.
Prior work “has attempted to map abnormalities to common brain regions rather than to a common brain network,” the investigators wrote. Moreover, “prior studies have rarely tested specificity by comparing psychiatric disorders to other brain disorders.”
In the current study, the researchers used “morphometric brain lesion datasets coupled with a wiring diagram of the human brain to derive a convergent brain network for psychiatric illness.”
They analyzed four large published datasets. Dataset 1 was sourced from an activation likelihood estimation meta-analysis (ALE) of whole-brain voxel-based studies that compared patients with psychiatric disorders such as schizophrenia, BD, depression, addiction, OCD, and anxiety to healthy controls (n = 193 studies; 15,892 individuals in total).
Dataset 2 was drawn from published neuroimaging studies involving patients with Alzheimer’s disease (AD) and other neurodegenerative conditions (n = 72 studies). They reported coordinates regarding which patients with these disorders had more atrophy compared with control persons.
Dataset 3 was sourced from the Vietnam Head Injury study, which followed veterans with and those without penetrating head injuries (n = 194 veterans with injuries). Dataset 4 was sourced from published neurosurgical ablation coordinates for depression.
Shared neurobiology
Upon analyzing dataset 1, the researchers found decreased gray matter in the bilateral anterior insula, dorsal anterior cingulate cortex, dorsomedial prefrontal cortex, thalamus, amygdala, hippocampus, and parietal operculum – findings that are “consistent with prior work.”
However, fewer than 35% of the studies contributed to any single cluster; and no cluster was specific to psychiatric versus neurodegenerative coordinates (drawn from dataset 2).
On the other hand, coordinate network mapping yielded “more statistically robust” (P < .001) results, which were found in 85% of the studies. “Psychiatric atrophy coordinates were functionally connected to the same network of brain regions,” the researchers reported.
This network was defined by two types of connectivity, positive and negative.
“The topography of this transdiagnostic network was independent of the statistical threshold and specific to psychiatric (vs. neurodegenerative) disorders, with the strongest peak occurring in the posterior parietal cortex (Brodmann Area 7) near the intraparietal sulcus,” the investigators wrote.
When lesions from dataset 3 were overlaid onto the ALE map and the transdiagnostic network in order to evaluate whether damage to either map correlated with number of post-lesion psychiatric diagnosis, results showed no evidence of a correlation between psychiatric comorbidity and damage on the ALE map (Pearson r, 0.02; P = .766).
However, when the same approach was applied to the transdiagnostic network, a statistically significant correlation was found between psychiatric comorbidity and lesion damage (Pearson r, –0.21; P = .01). A multiple regression model showed that the transdiagnostic, but not the ALE, network “independently predicted the number of post-lesion psychiatric diagnoses” (P = .003 vs. P = .1), the investigators reported.
All four neurosurgical ablative targets for psychiatric disorders found on analysis of dataset 4 “intersected” and aligned with the transdiagnostic network.
“The study does not immediately impact clinical practice, but it would be helpful for practicing clinicians to know that psychiatric disorders commonly co-occur and might share common neurobiology and a convergent brain network,” Dr. Taylor said.
“Future work based on our findings could potentially influence clinical trials and clinical practice, especially in the area of brain stimulation,” he added.
‘Exciting new targets’
In a comment, Desmond Oathes, PhD, associate director, Center for Neuromodulation and Stress, University of Pennsylvania, Philadelphia, said the “next step in the science is to combine individual brain imaging, aka, ‘individualized connectomes,’ with these promising group maps to determine something meaningful at the individual patient level.”
Dr. Oathes, who is also a faculty clinician at the Center for the Treatment and Study of Anxiety and was not involved with the study, noted that an open question is whether the brain volume abnormalities/atrophy “can be changed with treatment and in what direction.”
A “strong take-home message from this paper is that brain volume measures from single coordinates are noisy as measures of psychiatric abnormality, whereas network effects seem to be especially sensitive for capturing these effects,” Dr. Oathes said.
The “abnormal networks across these disorders do not fit easily into well-known networks from healthy participants. However, they map well onto other databases relevant to psychiatric disorders and offer exciting new potential targets for prospective treatment studies,” he added.
The investigators received no specific funding for this work. Dr. Taylor reported no relevant financial relationships. Dr. Oathes reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Investigators used coordinate and lesion network mapping to assess whether there was a shared brain network common to multiple psychiatric disorders. In a meta-analysis of almost 200 studies encompassing more than 15,000 individuals, they found that atrophy coordinates across these six psychiatric conditions all mapped to a common brain network.
Moreover, lesion damage to this network in patients with penetrating head trauma correlated with the number of psychiatric illnesses that the patients were diagnosed with post trauma.
The findings have “bigger-picture potential implications,” lead author Joseph Taylor, MD, PhD, medical director of transcranial magnetic stimulation at Brigham and Women’s Hospital’s Center for Brain Circuit Therapeutics, Boston, told this news organization.
“In psychiatry, we talk about symptoms and define our disorders based on symptom checklists, which are fairly reliable but don’t have neurobiological underpinnings,” said Dr. Taylor, who is also an associate psychiatrist in Brigham’s department of psychiatry.
By contrast, “in neurology, we ask: ‘Where is the lesion?’ Studying brain networks could potentially help us diagnose and treat people with psychiatric illness more effectively, just as we treat neurological disorders,” he added.
The findings were published online in Nature Human Behavior.
Beyond symptom checklists
Dr. Taylor noted that, in the field of psychiatry, “we often study disorders in isolation,” such as generalized anxiety disorder and major depressive disorder.
“But what see clinically is that half of patients meet the criteria for more than one psychiatric disorder,” he said. “It can be difficult to diagnose and treat these patients, and there are worse treatment outcomes.”
There is also a “discrepancy” between how these disorders are studied (one at a time) and how patients are treated in clinic, Dr. Taylor noted. And there is increasing evidence that psychiatric disorders may share a common neurobiology.
This “highlights the possibility of potentially developing transdiagnostic treatments based on common neurobiology, not just symptom checklists,” Dr. Taylor said.
Prior work “has attempted to map abnormalities to common brain regions rather than to a common brain network,” the investigators wrote. Moreover, “prior studies have rarely tested specificity by comparing psychiatric disorders to other brain disorders.”
In the current study, the researchers used “morphometric brain lesion datasets coupled with a wiring diagram of the human brain to derive a convergent brain network for psychiatric illness.”
They analyzed four large published datasets. Dataset 1 was sourced from an activation likelihood estimation meta-analysis (ALE) of whole-brain voxel-based studies that compared patients with psychiatric disorders such as schizophrenia, BD, depression, addiction, OCD, and anxiety to healthy controls (n = 193 studies; 15,892 individuals in total).
Dataset 2 was drawn from published neuroimaging studies involving patients with Alzheimer’s disease (AD) and other neurodegenerative conditions (n = 72 studies). They reported coordinates regarding which patients with these disorders had more atrophy compared with control persons.
Dataset 3 was sourced from the Vietnam Head Injury study, which followed veterans with and those without penetrating head injuries (n = 194 veterans with injuries). Dataset 4 was sourced from published neurosurgical ablation coordinates for depression.
Shared neurobiology
Upon analyzing dataset 1, the researchers found decreased gray matter in the bilateral anterior insula, dorsal anterior cingulate cortex, dorsomedial prefrontal cortex, thalamus, amygdala, hippocampus, and parietal operculum – findings that are “consistent with prior work.”
However, fewer than 35% of the studies contributed to any single cluster; and no cluster was specific to psychiatric versus neurodegenerative coordinates (drawn from dataset 2).
On the other hand, coordinate network mapping yielded “more statistically robust” (P < .001) results, which were found in 85% of the studies. “Psychiatric atrophy coordinates were functionally connected to the same network of brain regions,” the researchers reported.
This network was defined by two types of connectivity, positive and negative.
“The topography of this transdiagnostic network was independent of the statistical threshold and specific to psychiatric (vs. neurodegenerative) disorders, with the strongest peak occurring in the posterior parietal cortex (Brodmann Area 7) near the intraparietal sulcus,” the investigators wrote.
When lesions from dataset 3 were overlaid onto the ALE map and the transdiagnostic network in order to evaluate whether damage to either map correlated with number of post-lesion psychiatric diagnosis, results showed no evidence of a correlation between psychiatric comorbidity and damage on the ALE map (Pearson r, 0.02; P = .766).
However, when the same approach was applied to the transdiagnostic network, a statistically significant correlation was found between psychiatric comorbidity and lesion damage (Pearson r, –0.21; P = .01). A multiple regression model showed that the transdiagnostic, but not the ALE, network “independently predicted the number of post-lesion psychiatric diagnoses” (P = .003 vs. P = .1), the investigators reported.
All four neurosurgical ablative targets for psychiatric disorders found on analysis of dataset 4 “intersected” and aligned with the transdiagnostic network.
“The study does not immediately impact clinical practice, but it would be helpful for practicing clinicians to know that psychiatric disorders commonly co-occur and might share common neurobiology and a convergent brain network,” Dr. Taylor said.
“Future work based on our findings could potentially influence clinical trials and clinical practice, especially in the area of brain stimulation,” he added.
‘Exciting new targets’
In a comment, Desmond Oathes, PhD, associate director, Center for Neuromodulation and Stress, University of Pennsylvania, Philadelphia, said the “next step in the science is to combine individual brain imaging, aka, ‘individualized connectomes,’ with these promising group maps to determine something meaningful at the individual patient level.”
Dr. Oathes, who is also a faculty clinician at the Center for the Treatment and Study of Anxiety and was not involved with the study, noted that an open question is whether the brain volume abnormalities/atrophy “can be changed with treatment and in what direction.”
A “strong take-home message from this paper is that brain volume measures from single coordinates are noisy as measures of psychiatric abnormality, whereas network effects seem to be especially sensitive for capturing these effects,” Dr. Oathes said.
The “abnormal networks across these disorders do not fit easily into well-known networks from healthy participants. However, they map well onto other databases relevant to psychiatric disorders and offer exciting new potential targets for prospective treatment studies,” he added.
The investigators received no specific funding for this work. Dr. Taylor reported no relevant financial relationships. Dr. Oathes reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NATURE HUMAN BEHAVIOR
Can a ‘smart’ skin patch detect early neurodegenerative diseases?
A new “smart patch” composed of microneedles that can detect proinflammatory markers via simulated skin interstitial fluid (ISF) may help diagnose neurodegenerative disorders such as Alzheimer’s disease and Parkinson’s disease very early on.
Originally developed to deliver medications and vaccines via the skin in a minimally invasive manner, the microneedle arrays were fitted with molecular sensors that, when placed on the skin, detect neuroinflammatory biomarkers such as interleukin-6 in as little as 6 minutes.
The literature suggests that these biomarkers of neurodegenerative disease are present years before patients become symptomatic, said study investigator Sanjiv Sharma, PhD.
“Neurodegenerative disorders such as Parkinson’s disease and Alzheimer’s disease are [characterized by] progressive loss in nerve cell and brain cells, which leads to memory problems and a loss of mental ability. That is why early diagnosis is key to preventing the loss of brain tissue in dementia, which can go undetected for years,” added Dr. Sharma, who is a lecturer in medical engineering at Swansea (Wales) University.
Dr. Sharma developed the patch with scientists at the Polytechnic of Porto (Portugal) School of Engineering in Portugal. In 2022, they designed, and are currently testing, a microneedle patch that will deliver the COVID vaccine.
The investigators describe their research on the patch’s ability to detect IL-6 in an article published in ACS Omega.
At-home diagnosis?
“The skin is the largest organ in the body – it contains more skin interstitial fluid than the total blood volume,” Dr. Sharma noted. “This fluid is an ultrafiltrate of blood and holds biomarkers that complement other biofluids, such as sweat, saliva, and urine. It can be sampled in a minimally invasive manner and used either for point-of-care testing or real-time using microneedle devices.”
Dr. Sharma and associates tested the microneedle patch in artificial ISF that contained the inflammatory cytokine IL-6. They found that the patch accurately detected IL-6 concentrations as low as 1 pg/mL in the fabricated ISF solution.
“In general, the transdermal sensor presented here showed simplicity in designing, short measuring time, high accuracy, and low detection limit. This approach seems a successful tool for the screening of inflammatory biomarkers in point of care testing wherein the skin acts as a window to the body,” the investigators reported.
Dr. Sharma noted that early detection of neurodegenerative diseases is crucial, as once symptoms appear, the disease may have already progressed significantly, and meaningful intervention is challenging.
The device has yet to be tested in humans, which is the next step, said Dr. Sharma.
“We will have to test the hypothesis through extensive preclinical and clinical studies to determine if bloodless, transdermal (skin) diagnostics can offer a cost-effective device that could allow testing in simpler settings such as a clinician’s practice or even home settings,” he noted.
Early days
Commenting on the research, David K. Simon, MD, PhD, professor of neurology at Harvard Medical School, Boston, said it is “a promising step regarding validation of a potentially beneficial method for rapidly and accurately measuring IL-6.”
However, he added, “many additional steps are needed to validate the method in actual human skin and to determine whether or not measuring these biomarkers in skin will be useful in studies of neurodegenerative diseases.”
He noted that one study limitation is that inflammatory cytokines such as IL-6 are highly nonspecific, and levels are elevated in various diseases associated with inflammation.
“It is highly unlikely that measuring IL-6 will be useful as a diagnostic tool. However, it does have potential as a biomarker for measuring the impact of treatments aimed at reducing inflammation. As the authors point out, it’s more likely that clinicians will require a panel of biomarkers rather than only measuring IL-6,” he said.
The study was funded by Fundação para a Ciência e Tecnologia. The investigators disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new “smart patch” composed of microneedles that can detect proinflammatory markers via simulated skin interstitial fluid (ISF) may help diagnose neurodegenerative disorders such as Alzheimer’s disease and Parkinson’s disease very early on.
Originally developed to deliver medications and vaccines via the skin in a minimally invasive manner, the microneedle arrays were fitted with molecular sensors that, when placed on the skin, detect neuroinflammatory biomarkers such as interleukin-6 in as little as 6 minutes.
The literature suggests that these biomarkers of neurodegenerative disease are present years before patients become symptomatic, said study investigator Sanjiv Sharma, PhD.
“Neurodegenerative disorders such as Parkinson’s disease and Alzheimer’s disease are [characterized by] progressive loss in nerve cell and brain cells, which leads to memory problems and a loss of mental ability. That is why early diagnosis is key to preventing the loss of brain tissue in dementia, which can go undetected for years,” added Dr. Sharma, who is a lecturer in medical engineering at Swansea (Wales) University.
Dr. Sharma developed the patch with scientists at the Polytechnic of Porto (Portugal) School of Engineering in Portugal. In 2022, they designed, and are currently testing, a microneedle patch that will deliver the COVID vaccine.
The investigators describe their research on the patch’s ability to detect IL-6 in an article published in ACS Omega.
At-home diagnosis?
“The skin is the largest organ in the body – it contains more skin interstitial fluid than the total blood volume,” Dr. Sharma noted. “This fluid is an ultrafiltrate of blood and holds biomarkers that complement other biofluids, such as sweat, saliva, and urine. It can be sampled in a minimally invasive manner and used either for point-of-care testing or real-time using microneedle devices.”
Dr. Sharma and associates tested the microneedle patch in artificial ISF that contained the inflammatory cytokine IL-6. They found that the patch accurately detected IL-6 concentrations as low as 1 pg/mL in the fabricated ISF solution.
“In general, the transdermal sensor presented here showed simplicity in designing, short measuring time, high accuracy, and low detection limit. This approach seems a successful tool for the screening of inflammatory biomarkers in point of care testing wherein the skin acts as a window to the body,” the investigators reported.
Dr. Sharma noted that early detection of neurodegenerative diseases is crucial, as once symptoms appear, the disease may have already progressed significantly, and meaningful intervention is challenging.
The device has yet to be tested in humans, which is the next step, said Dr. Sharma.
“We will have to test the hypothesis through extensive preclinical and clinical studies to determine if bloodless, transdermal (skin) diagnostics can offer a cost-effective device that could allow testing in simpler settings such as a clinician’s practice or even home settings,” he noted.
Early days
Commenting on the research, David K. Simon, MD, PhD, professor of neurology at Harvard Medical School, Boston, said it is “a promising step regarding validation of a potentially beneficial method for rapidly and accurately measuring IL-6.”
However, he added, “many additional steps are needed to validate the method in actual human skin and to determine whether or not measuring these biomarkers in skin will be useful in studies of neurodegenerative diseases.”
He noted that one study limitation is that inflammatory cytokines such as IL-6 are highly nonspecific, and levels are elevated in various diseases associated with inflammation.
“It is highly unlikely that measuring IL-6 will be useful as a diagnostic tool. However, it does have potential as a biomarker for measuring the impact of treatments aimed at reducing inflammation. As the authors point out, it’s more likely that clinicians will require a panel of biomarkers rather than only measuring IL-6,” he said.
The study was funded by Fundação para a Ciência e Tecnologia. The investigators disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new “smart patch” composed of microneedles that can detect proinflammatory markers via simulated skin interstitial fluid (ISF) may help diagnose neurodegenerative disorders such as Alzheimer’s disease and Parkinson’s disease very early on.
Originally developed to deliver medications and vaccines via the skin in a minimally invasive manner, the microneedle arrays were fitted with molecular sensors that, when placed on the skin, detect neuroinflammatory biomarkers such as interleukin-6 in as little as 6 minutes.
The literature suggests that these biomarkers of neurodegenerative disease are present years before patients become symptomatic, said study investigator Sanjiv Sharma, PhD.
“Neurodegenerative disorders such as Parkinson’s disease and Alzheimer’s disease are [characterized by] progressive loss in nerve cell and brain cells, which leads to memory problems and a loss of mental ability. That is why early diagnosis is key to preventing the loss of brain tissue in dementia, which can go undetected for years,” added Dr. Sharma, who is a lecturer in medical engineering at Swansea (Wales) University.
Dr. Sharma developed the patch with scientists at the Polytechnic of Porto (Portugal) School of Engineering in Portugal. In 2022, they designed, and are currently testing, a microneedle patch that will deliver the COVID vaccine.
The investigators describe their research on the patch’s ability to detect IL-6 in an article published in ACS Omega.
At-home diagnosis?
“The skin is the largest organ in the body – it contains more skin interstitial fluid than the total blood volume,” Dr. Sharma noted. “This fluid is an ultrafiltrate of blood and holds biomarkers that complement other biofluids, such as sweat, saliva, and urine. It can be sampled in a minimally invasive manner and used either for point-of-care testing or real-time using microneedle devices.”
Dr. Sharma and associates tested the microneedle patch in artificial ISF that contained the inflammatory cytokine IL-6. They found that the patch accurately detected IL-6 concentrations as low as 1 pg/mL in the fabricated ISF solution.
“In general, the transdermal sensor presented here showed simplicity in designing, short measuring time, high accuracy, and low detection limit. This approach seems a successful tool for the screening of inflammatory biomarkers in point of care testing wherein the skin acts as a window to the body,” the investigators reported.
Dr. Sharma noted that early detection of neurodegenerative diseases is crucial, as once symptoms appear, the disease may have already progressed significantly, and meaningful intervention is challenging.
The device has yet to be tested in humans, which is the next step, said Dr. Sharma.
“We will have to test the hypothesis through extensive preclinical and clinical studies to determine if bloodless, transdermal (skin) diagnostics can offer a cost-effective device that could allow testing in simpler settings such as a clinician’s practice or even home settings,” he noted.
Early days
Commenting on the research, David K. Simon, MD, PhD, professor of neurology at Harvard Medical School, Boston, said it is “a promising step regarding validation of a potentially beneficial method for rapidly and accurately measuring IL-6.”
However, he added, “many additional steps are needed to validate the method in actual human skin and to determine whether or not measuring these biomarkers in skin will be useful in studies of neurodegenerative diseases.”
He noted that one study limitation is that inflammatory cytokines such as IL-6 are highly nonspecific, and levels are elevated in various diseases associated with inflammation.
“It is highly unlikely that measuring IL-6 will be useful as a diagnostic tool. However, it does have potential as a biomarker for measuring the impact of treatments aimed at reducing inflammation. As the authors point out, it’s more likely that clinicians will require a panel of biomarkers rather than only measuring IL-6,” he said.
The study was funded by Fundação para a Ciência e Tecnologia. The investigators disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ACS OMEGA
Poor sleep quality as a teen may up MS risk in adulthood
Too little sleep or poor sleep quality during the teen years can significantly increase the risk for multiple sclerosis (MS) during adulthood, new research suggests.
In a large case-control study, individuals who slept less than 7 hours a night on average during adolescence were 40% more likely to develop MS later on. The risk was even higher for those who rated their sleep quality as bad.
On the other hand, MS was significantly less common among individuals who slept longer as teens – indicating a possible protective benefit.
While sleep duration has been associated with mortality or disease risk for other conditions, sleep quality usually has little to no effect on risk, lead investigator Torbjörn Åkerstedt, PhD, sleep researcher and professor of psychology, department of neuroscience, Karolinska Institutet, Stockholm, told this news organization.
“I hadn’t really expected that, but those results were quite strong, even stronger than sleep duration,” Dr. Åkerstedt said.
“We don’t really know why this is happening in young age, but the most suitable explanation is that the brain in still developing quite a bit, and you’re interfering with it,” he added.
The findings were published online in the Journal of Neurology, Neurosurgery and Psychiatry.
Strong association
Other studies have tied sleep deprivation to increased risk for serious illness, but the link between sleep and MS risk isn’t as well studied.
Previous research by Dr. Åkerstedt showed that the risk for MS was higher among individuals who took part in shift work before the age of 20. However, the impact of sleep duration or quality among teens was unknown.
The current Swedish population-based case-control study included 2,075 patients with MS and 3,164 without the disorder. All participants were asked to recall how many hours on average they slept per night between the ages of 15 and 19 years and to rate their sleep quality during that time.
Results showed that individuals who slept fewer than 7 hours a night during their teen years were 40% more likely to have MS as adults (odds ratio [OR], 1.4; 95% confidence interval [CI], 1.1-1.7).
Poor sleep quality increased MS risk even more (OR, 1.5; 95% CI, 1.3-1.9).
The association remained strong even after adjustment for additional sleep on weekends and breaks and excluding shift workers.
Long sleep ‘apparently good’
The researchers also conducted several sensitivity studies to rule out confounders that might bias the association, such as excluding participants who reported currently experiencing less sleep or poor sleep.
“You would expect that people who are suffering from sleep problems today would be the people who reported sleep problems during their youth,” but that didn’t happen, Dr. Åkerstedt noted.
The investigators also entered data on sleep duration and sleep quality at the same time, thinking the data would cancel each other out. However, the association remained the same.
“Quite often you see that sleep duration would eliminate the effect of sleep complaints in the prediction of disease, but here both remain significant when they are entered at the same time,” Dr. Åkerstedt said. “You get the feeling that this might mean they act together to produce results,” he added.
“One other thing that surprised me is that long sleep was apparently good,” said Dr. Åkerstedt.
The investigators have conducted several studies on sleep duration and mortality. In recent research, they found that both short sleep and long sleep predicted mortality – “and often, long sleep is a stronger predictor than short sleep,” he said.
Underestimated problem?
Commenting on the findings, Kathleen Zackowski, PhD, associate vice president of research for the National Multiple Sclerosis Society in Baltimore, noted that participants were asked to rate their own sleep quality during adolescence, a subjective report that may mean sleep quality has an even larger association with MS risk.
“That they found a result with sleep quality says to me that there probably is a bigger problem, because I don’t know if people over- or underestimate their sleep quality,” said Dr. Zackowski, who was not involved with the research.
“If we could get to that sleep quality question a little more objectively, I bet that we’d find there’s a lot more to the story,” she said.
That’s a story the researchers would like to explore, Dr. Åkerstedt reported. Designing a prospective study that more closely tracks sleeping habits during adolescence and follows individuals through adulthood could provide valuable information about how sleep quality and duration affect immune system development and MS risk, he said.
Dr. Zackowski said clinicians know that MS is not caused just by a genetic abnormality and that other environmental lifestyle factors seem to play a part.
“If we find out that sleep is one of those lifestyle factors, this is very changeable,” she added.
The study was funded by the Swedish Research Council, the Swedish Research Council for Health, Working Life and Welfare, the Swedish Brain Foundation, AFA Insurance, the European Aviation Safety Authority, the Tercentenary Fund of the Bank of Sweden, the Margaretha af Ugglas Foundation, the Swedish Foundation for MS Research, and NEURO Sweden. Dr. Åkerstadt has been supported by Tercentenary Fund of Bank of Sweden, AFA Insurance, and the European Aviation Safety Authority. Dr. Zackowski reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Too little sleep or poor sleep quality during the teen years can significantly increase the risk for multiple sclerosis (MS) during adulthood, new research suggests.
In a large case-control study, individuals who slept less than 7 hours a night on average during adolescence were 40% more likely to develop MS later on. The risk was even higher for those who rated their sleep quality as bad.
On the other hand, MS was significantly less common among individuals who slept longer as teens – indicating a possible protective benefit.
While sleep duration has been associated with mortality or disease risk for other conditions, sleep quality usually has little to no effect on risk, lead investigator Torbjörn Åkerstedt, PhD, sleep researcher and professor of psychology, department of neuroscience, Karolinska Institutet, Stockholm, told this news organization.
“I hadn’t really expected that, but those results were quite strong, even stronger than sleep duration,” Dr. Åkerstedt said.
“We don’t really know why this is happening in young age, but the most suitable explanation is that the brain in still developing quite a bit, and you’re interfering with it,” he added.
The findings were published online in the Journal of Neurology, Neurosurgery and Psychiatry.
Strong association
Other studies have tied sleep deprivation to increased risk for serious illness, but the link between sleep and MS risk isn’t as well studied.
Previous research by Dr. Åkerstedt showed that the risk for MS was higher among individuals who took part in shift work before the age of 20. However, the impact of sleep duration or quality among teens was unknown.
The current Swedish population-based case-control study included 2,075 patients with MS and 3,164 without the disorder. All participants were asked to recall how many hours on average they slept per night between the ages of 15 and 19 years and to rate their sleep quality during that time.
Results showed that individuals who slept fewer than 7 hours a night during their teen years were 40% more likely to have MS as adults (odds ratio [OR], 1.4; 95% confidence interval [CI], 1.1-1.7).
Poor sleep quality increased MS risk even more (OR, 1.5; 95% CI, 1.3-1.9).
The association remained strong even after adjustment for additional sleep on weekends and breaks and excluding shift workers.
Long sleep ‘apparently good’
The researchers also conducted several sensitivity studies to rule out confounders that might bias the association, such as excluding participants who reported currently experiencing less sleep or poor sleep.
“You would expect that people who are suffering from sleep problems today would be the people who reported sleep problems during their youth,” but that didn’t happen, Dr. Åkerstedt noted.
The investigators also entered data on sleep duration and sleep quality at the same time, thinking the data would cancel each other out. However, the association remained the same.
“Quite often you see that sleep duration would eliminate the effect of sleep complaints in the prediction of disease, but here both remain significant when they are entered at the same time,” Dr. Åkerstedt said. “You get the feeling that this might mean they act together to produce results,” he added.
“One other thing that surprised me is that long sleep was apparently good,” said Dr. Åkerstedt.
The investigators have conducted several studies on sleep duration and mortality. In recent research, they found that both short sleep and long sleep predicted mortality – “and often, long sleep is a stronger predictor than short sleep,” he said.
Underestimated problem?
Commenting on the findings, Kathleen Zackowski, PhD, associate vice president of research for the National Multiple Sclerosis Society in Baltimore, noted that participants were asked to rate their own sleep quality during adolescence, a subjective report that may mean sleep quality has an even larger association with MS risk.
“That they found a result with sleep quality says to me that there probably is a bigger problem, because I don’t know if people over- or underestimate their sleep quality,” said Dr. Zackowski, who was not involved with the research.
“If we could get to that sleep quality question a little more objectively, I bet that we’d find there’s a lot more to the story,” she said.
That’s a story the researchers would like to explore, Dr. Åkerstedt reported. Designing a prospective study that more closely tracks sleeping habits during adolescence and follows individuals through adulthood could provide valuable information about how sleep quality and duration affect immune system development and MS risk, he said.
Dr. Zackowski said clinicians know that MS is not caused just by a genetic abnormality and that other environmental lifestyle factors seem to play a part.
“If we find out that sleep is one of those lifestyle factors, this is very changeable,” she added.
The study was funded by the Swedish Research Council, the Swedish Research Council for Health, Working Life and Welfare, the Swedish Brain Foundation, AFA Insurance, the European Aviation Safety Authority, the Tercentenary Fund of the Bank of Sweden, the Margaretha af Ugglas Foundation, the Swedish Foundation for MS Research, and NEURO Sweden. Dr. Åkerstadt has been supported by Tercentenary Fund of Bank of Sweden, AFA Insurance, and the European Aviation Safety Authority. Dr. Zackowski reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Too little sleep or poor sleep quality during the teen years can significantly increase the risk for multiple sclerosis (MS) during adulthood, new research suggests.
In a large case-control study, individuals who slept less than 7 hours a night on average during adolescence were 40% more likely to develop MS later on. The risk was even higher for those who rated their sleep quality as bad.
On the other hand, MS was significantly less common among individuals who slept longer as teens – indicating a possible protective benefit.
While sleep duration has been associated with mortality or disease risk for other conditions, sleep quality usually has little to no effect on risk, lead investigator Torbjörn Åkerstedt, PhD, sleep researcher and professor of psychology, department of neuroscience, Karolinska Institutet, Stockholm, told this news organization.
“I hadn’t really expected that, but those results were quite strong, even stronger than sleep duration,” Dr. Åkerstedt said.
“We don’t really know why this is happening in young age, but the most suitable explanation is that the brain in still developing quite a bit, and you’re interfering with it,” he added.
The findings were published online in the Journal of Neurology, Neurosurgery and Psychiatry.
Strong association
Other studies have tied sleep deprivation to increased risk for serious illness, but the link between sleep and MS risk isn’t as well studied.
Previous research by Dr. Åkerstedt showed that the risk for MS was higher among individuals who took part in shift work before the age of 20. However, the impact of sleep duration or quality among teens was unknown.
The current Swedish population-based case-control study included 2,075 patients with MS and 3,164 without the disorder. All participants were asked to recall how many hours on average they slept per night between the ages of 15 and 19 years and to rate their sleep quality during that time.
Results showed that individuals who slept fewer than 7 hours a night during their teen years were 40% more likely to have MS as adults (odds ratio [OR], 1.4; 95% confidence interval [CI], 1.1-1.7).
Poor sleep quality increased MS risk even more (OR, 1.5; 95% CI, 1.3-1.9).
The association remained strong even after adjustment for additional sleep on weekends and breaks and excluding shift workers.
Long sleep ‘apparently good’
The researchers also conducted several sensitivity studies to rule out confounders that might bias the association, such as excluding participants who reported currently experiencing less sleep or poor sleep.
“You would expect that people who are suffering from sleep problems today would be the people who reported sleep problems during their youth,” but that didn’t happen, Dr. Åkerstedt noted.
The investigators also entered data on sleep duration and sleep quality at the same time, thinking the data would cancel each other out. However, the association remained the same.
“Quite often you see that sleep duration would eliminate the effect of sleep complaints in the prediction of disease, but here both remain significant when they are entered at the same time,” Dr. Åkerstedt said. “You get the feeling that this might mean they act together to produce results,” he added.
“One other thing that surprised me is that long sleep was apparently good,” said Dr. Åkerstedt.
The investigators have conducted several studies on sleep duration and mortality. In recent research, they found that both short sleep and long sleep predicted mortality – “and often, long sleep is a stronger predictor than short sleep,” he said.
Underestimated problem?
Commenting on the findings, Kathleen Zackowski, PhD, associate vice president of research for the National Multiple Sclerosis Society in Baltimore, noted that participants were asked to rate their own sleep quality during adolescence, a subjective report that may mean sleep quality has an even larger association with MS risk.
“That they found a result with sleep quality says to me that there probably is a bigger problem, because I don’t know if people over- or underestimate their sleep quality,” said Dr. Zackowski, who was not involved with the research.
“If we could get to that sleep quality question a little more objectively, I bet that we’d find there’s a lot more to the story,” she said.
That’s a story the researchers would like to explore, Dr. Åkerstedt reported. Designing a prospective study that more closely tracks sleeping habits during adolescence and follows individuals through adulthood could provide valuable information about how sleep quality and duration affect immune system development and MS risk, he said.
Dr. Zackowski said clinicians know that MS is not caused just by a genetic abnormality and that other environmental lifestyle factors seem to play a part.
“If we find out that sleep is one of those lifestyle factors, this is very changeable,” she added.
The study was funded by the Swedish Research Council, the Swedish Research Council for Health, Working Life and Welfare, the Swedish Brain Foundation, AFA Insurance, the European Aviation Safety Authority, the Tercentenary Fund of the Bank of Sweden, the Margaretha af Ugglas Foundation, the Swedish Foundation for MS Research, and NEURO Sweden. Dr. Åkerstadt has been supported by Tercentenary Fund of Bank of Sweden, AFA Insurance, and the European Aviation Safety Authority. Dr. Zackowski reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Surgeon General says 13-year-olds shouldn’t be on social media
The U.S. Surgeon General says 13 years old is too young to begin using social media.
Most social media platforms including TikTok, Snapchat, Instagram, and Facebook allow users to create accounts if they say they are at least 13 years old.
“I, personally, based on the data I’ve seen, believe that 13 is too early. ... It’s a time where it’s really important for us to be thoughtful about what’s going into how they think about their own self-worth and their relationships, and the skewed and often distorted environment of social media often does a disservice to many of those children,” U.S. Surgeon General Vivek Murthy, MD, told CNN.
Research has shown that teens are susceptible to cyberbullying and serious mental health impacts from social media usage and online activity during an era when the influence of the Internet has become everywhere for young people.
According to the Pew Research Center, 95% of teens age 13 and up have a smartphone, and 97% of teens say they use the Internet daily. Among 13- and 14-year-olds, 61% say they use TikTok and 51% say they use Snapchat. Older teens ages 15-17 use those social media platforms at higher rates, with 71% saying they use TikTok and 65% using Snapchat.
“If parents can band together and say you know, as a group, we’re not going to allow our kids to use social media until 16 or 17 or 18 or whatever age they choose, that’s a much more effective strategy in making sure your kids don’t get exposed to harm early,” Dr. Murthy said.
A version of this article originally appeared on WebMD.com.
The U.S. Surgeon General says 13 years old is too young to begin using social media.
Most social media platforms including TikTok, Snapchat, Instagram, and Facebook allow users to create accounts if they say they are at least 13 years old.
“I, personally, based on the data I’ve seen, believe that 13 is too early. ... It’s a time where it’s really important for us to be thoughtful about what’s going into how they think about their own self-worth and their relationships, and the skewed and often distorted environment of social media often does a disservice to many of those children,” U.S. Surgeon General Vivek Murthy, MD, told CNN.
Research has shown that teens are susceptible to cyberbullying and serious mental health impacts from social media usage and online activity during an era when the influence of the Internet has become everywhere for young people.
According to the Pew Research Center, 95% of teens age 13 and up have a smartphone, and 97% of teens say they use the Internet daily. Among 13- and 14-year-olds, 61% say they use TikTok and 51% say they use Snapchat. Older teens ages 15-17 use those social media platforms at higher rates, with 71% saying they use TikTok and 65% using Snapchat.
“If parents can band together and say you know, as a group, we’re not going to allow our kids to use social media until 16 or 17 or 18 or whatever age they choose, that’s a much more effective strategy in making sure your kids don’t get exposed to harm early,” Dr. Murthy said.
A version of this article originally appeared on WebMD.com.
The U.S. Surgeon General says 13 years old is too young to begin using social media.
Most social media platforms including TikTok, Snapchat, Instagram, and Facebook allow users to create accounts if they say they are at least 13 years old.
“I, personally, based on the data I’ve seen, believe that 13 is too early. ... It’s a time where it’s really important for us to be thoughtful about what’s going into how they think about their own self-worth and their relationships, and the skewed and often distorted environment of social media often does a disservice to many of those children,” U.S. Surgeon General Vivek Murthy, MD, told CNN.
Research has shown that teens are susceptible to cyberbullying and serious mental health impacts from social media usage and online activity during an era when the influence of the Internet has become everywhere for young people.
According to the Pew Research Center, 95% of teens age 13 and up have a smartphone, and 97% of teens say they use the Internet daily. Among 13- and 14-year-olds, 61% say they use TikTok and 51% say they use Snapchat. Older teens ages 15-17 use those social media platforms at higher rates, with 71% saying they use TikTok and 65% using Snapchat.
“If parents can band together and say you know, as a group, we’re not going to allow our kids to use social media until 16 or 17 or 18 or whatever age they choose, that’s a much more effective strategy in making sure your kids don’t get exposed to harm early,” Dr. Murthy said.
A version of this article originally appeared on WebMD.com.
Six healthy lifestyle habits linked to slowed memory decline
Investigators found that a healthy diet, cognitive activity, regular physical exercise, not smoking, and abstaining from alcohol were significantly linked to slowed cognitive decline irrespective of APOE4 status.
After adjusting for health and socioeconomic factors, investigators found that each individual healthy behavior was associated with a slower-than-average decline in memory over a decade. A healthy diet emerged as the strongest deterrent, followed by cognitive activity and physical exercise.
“A healthy lifestyle is associated with slower memory decline, even in the presence of the APOE4 allele,” study investigators led by Jianping Jia, MD, PhD, of the Innovation Center for Neurological Disorders and the department of neurology, Xuan Wu Hospital, Capital Medical University, Beijing, write.
“This study might offer important information to protect older adults against memory decline,” they add.
The study was published online in the BMJ.
Preventing memory decline
Memory “continuously declines as people age,” but age-related memory decline is not necessarily a prodrome of dementia and can “merely be senescent forgetfulness,” the investigators note. This can be “reversed or [can] become stable,” instead of progressing to a pathologic state.
Factors affecting memory include aging, APOE4 genotype, chronic diseases, and lifestyle patterns, with lifestyle “receiving increasing attention as a modifiable behavior.”
Nevertheless, few studies have focused on the impact of lifestyle on memory, and those that have are mostly cross-sectional and also “did not consider the interaction between a healthy lifestyle and genetic risk,” the researchers note.
To investigate, the researchers conducted a longitudinal study, known as the China Cognition and Aging Study, that considered genetic risk as well as lifestyle factors.
The study began in 2009 and concluded in 2019. Participants were evaluated and underwent neuropsychological testing in 2012, 2014, 2016, and at the study’s conclusion.
Participants (n = 29,072; mean [SD] age, 72.23 [6.61] years; 48.54% women; 20.43% APOE4 carriers) were required to have normal cognitive function at baseline. Data on those whose condition progressed to mild cognitive impairment (MCI) or dementia during the follow-up period were excluded after their diagnosis.
The Mini–Mental State Examination was used to assess global cognitive function. Memory function was assessed using the World Health Organization/University of California, Los Angeles Auditory Verbal Learning Test.
“Lifestyle” consisted of six modifiable factors: physical exercise (weekly frequency and total time), smoking (current, former, or never-smokers), alcohol consumption (never drank, drank occasionally, low to excess drinking, and heavy drinking), diet (daily intake of 12 food items: fruits, vegetables, fish, meat, dairy products, salt, oil, eggs, cereals, legumes, nuts, tea), cognitive activity (writing, reading, playing cards, mahjong, other games), and social contact (participating in meetings, attending parties, visiting friends/relatives, traveling, chatting online).
Participants’ lifestyles were scored on the basis of the number of healthy factors they engaged in.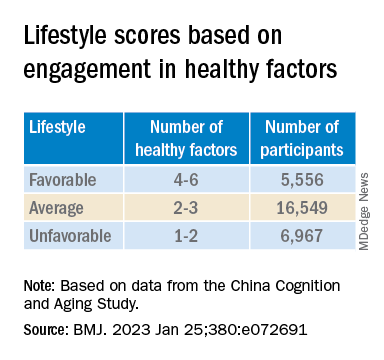
Participants were also stratified by APOE genotype into APOE4 carriers and noncarriers.
Demographic and other items of health information, including the presence of medical illness, were used as covariates. The researchers also included the “learning effect of each participant as a covariate, due to repeated cognitive assessments.”
Important for public health
During the 10-year period, 7,164 participants died, and 3,567 stopped participating.
Participants in the favorable and average groups showed slower memory decline per increased year of age (0.007 [0.005-0.009], P < .001; and 0.002 [0 .000-0.003], P = .033 points higher, respectively), compared with those in the unfavorable group.
Healthy diet had the strongest protective effect on memory.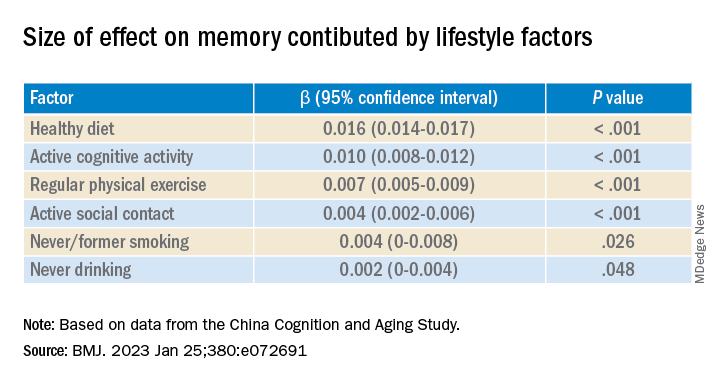
Memory decline occurred faster in APOE4 vesus non-APOE4 carriers (0.002 points/year [95% confidence interval, 0.001-0.003]; P = .007).
But APOE4 carriers with favorable and average lifestyles showed slower memory decline (0.027 [0.023-0.031] and 0.014 [0.010-0.019], respectively), compared with those with unfavorable lifestyles. Similar findings were obtained in non-APOE4 carriers.
Those with favorable or average lifestyle were respectively almost 90% and 30% less likely to develop dementia or MCI, compared with those with an unfavorable lifestyle.
The authors acknowledge the study’s limitations, including its observational design and the potential for measurement errors, owing to self-reporting of lifestyle factors. Additionally, some participants did not return for follow-up evaluations, leading to potential selection bias.
Nevertheless, the findings “might offer important information for public health to protect older [people] against memory decline,” they note – especially since the study “provides evidence that these effects also include individuals with the APOE4 allele.”
‘Important, encouraging’ research
In a comment, Severine Sabia, PhD, a senior researcher at the Université Paris Cité, INSERM Institut National de la Santé et de la Recherche Medicalé, France, called the findings “important and encouraging.”
However, said Dr. Sabia, who was not involved with the study, “there remain important research questions that need to be investigated in order to identify key behaviors: which combination, the cutoff of risk, and when to intervene.”
Future research on prevention “should examine a wider range of possible risk factors” and should also “identify specific exposures associated with the greatest risk, while also considering the risk threshold and age at exposure for each one.”
In an accompanying editorial, Dr. Sabia and co-author Archana Singh-Manoux, PhD, note that the risk of cognitive decline and dementia are probably determined by multiple factors.
They liken it to the “multifactorial risk paradigm introduced by the Framingham study,” which has “led to a substantial reduction in cardiovascular disease.” A similar approach could be used with dementia prevention, they suggest.
The authors received support from the Xuanwu Hospital of Capital Medical University for the submitted work. One of the authors received a grant from the French National Research Agency. The other authors have disclosed no relevant financial relationships. Dr. Sabia received grant funding from the French National Research Agency. Dr. Singh-Manoux received grants from the National Institute on Aging of the National Institutes of Health.
A version of this article first appeared on Medscape.com.
Investigators found that a healthy diet, cognitive activity, regular physical exercise, not smoking, and abstaining from alcohol were significantly linked to slowed cognitive decline irrespective of APOE4 status.
After adjusting for health and socioeconomic factors, investigators found that each individual healthy behavior was associated with a slower-than-average decline in memory over a decade. A healthy diet emerged as the strongest deterrent, followed by cognitive activity and physical exercise.
“A healthy lifestyle is associated with slower memory decline, even in the presence of the APOE4 allele,” study investigators led by Jianping Jia, MD, PhD, of the Innovation Center for Neurological Disorders and the department of neurology, Xuan Wu Hospital, Capital Medical University, Beijing, write.
“This study might offer important information to protect older adults against memory decline,” they add.
The study was published online in the BMJ.
Preventing memory decline
Memory “continuously declines as people age,” but age-related memory decline is not necessarily a prodrome of dementia and can “merely be senescent forgetfulness,” the investigators note. This can be “reversed or [can] become stable,” instead of progressing to a pathologic state.
Factors affecting memory include aging, APOE4 genotype, chronic diseases, and lifestyle patterns, with lifestyle “receiving increasing attention as a modifiable behavior.”
Nevertheless, few studies have focused on the impact of lifestyle on memory, and those that have are mostly cross-sectional and also “did not consider the interaction between a healthy lifestyle and genetic risk,” the researchers note.
To investigate, the researchers conducted a longitudinal study, known as the China Cognition and Aging Study, that considered genetic risk as well as lifestyle factors.
The study began in 2009 and concluded in 2019. Participants were evaluated and underwent neuropsychological testing in 2012, 2014, 2016, and at the study’s conclusion.
Participants (n = 29,072; mean [SD] age, 72.23 [6.61] years; 48.54% women; 20.43% APOE4 carriers) were required to have normal cognitive function at baseline. Data on those whose condition progressed to mild cognitive impairment (MCI) or dementia during the follow-up period were excluded after their diagnosis.
The Mini–Mental State Examination was used to assess global cognitive function. Memory function was assessed using the World Health Organization/University of California, Los Angeles Auditory Verbal Learning Test.
“Lifestyle” consisted of six modifiable factors: physical exercise (weekly frequency and total time), smoking (current, former, or never-smokers), alcohol consumption (never drank, drank occasionally, low to excess drinking, and heavy drinking), diet (daily intake of 12 food items: fruits, vegetables, fish, meat, dairy products, salt, oil, eggs, cereals, legumes, nuts, tea), cognitive activity (writing, reading, playing cards, mahjong, other games), and social contact (participating in meetings, attending parties, visiting friends/relatives, traveling, chatting online).
Participants’ lifestyles were scored on the basis of the number of healthy factors they engaged in.
Participants were also stratified by APOE genotype into APOE4 carriers and noncarriers.
Demographic and other items of health information, including the presence of medical illness, were used as covariates. The researchers also included the “learning effect of each participant as a covariate, due to repeated cognitive assessments.”
Important for public health
During the 10-year period, 7,164 participants died, and 3,567 stopped participating.
Participants in the favorable and average groups showed slower memory decline per increased year of age (0.007 [0.005-0.009], P < .001; and 0.002 [0 .000-0.003], P = .033 points higher, respectively), compared with those in the unfavorable group.
Healthy diet had the strongest protective effect on memory.
Memory decline occurred faster in APOE4 vesus non-APOE4 carriers (0.002 points/year [95% confidence interval, 0.001-0.003]; P = .007).
But APOE4 carriers with favorable and average lifestyles showed slower memory decline (0.027 [0.023-0.031] and 0.014 [0.010-0.019], respectively), compared with those with unfavorable lifestyles. Similar findings were obtained in non-APOE4 carriers.
Those with favorable or average lifestyle were respectively almost 90% and 30% less likely to develop dementia or MCI, compared with those with an unfavorable lifestyle.
The authors acknowledge the study’s limitations, including its observational design and the potential for measurement errors, owing to self-reporting of lifestyle factors. Additionally, some participants did not return for follow-up evaluations, leading to potential selection bias.
Nevertheless, the findings “might offer important information for public health to protect older [people] against memory decline,” they note – especially since the study “provides evidence that these effects also include individuals with the APOE4 allele.”
‘Important, encouraging’ research
In a comment, Severine Sabia, PhD, a senior researcher at the Université Paris Cité, INSERM Institut National de la Santé et de la Recherche Medicalé, France, called the findings “important and encouraging.”
However, said Dr. Sabia, who was not involved with the study, “there remain important research questions that need to be investigated in order to identify key behaviors: which combination, the cutoff of risk, and when to intervene.”
Future research on prevention “should examine a wider range of possible risk factors” and should also “identify specific exposures associated with the greatest risk, while also considering the risk threshold and age at exposure for each one.”
In an accompanying editorial, Dr. Sabia and co-author Archana Singh-Manoux, PhD, note that the risk of cognitive decline and dementia are probably determined by multiple factors.
They liken it to the “multifactorial risk paradigm introduced by the Framingham study,” which has “led to a substantial reduction in cardiovascular disease.” A similar approach could be used with dementia prevention, they suggest.
The authors received support from the Xuanwu Hospital of Capital Medical University for the submitted work. One of the authors received a grant from the French National Research Agency. The other authors have disclosed no relevant financial relationships. Dr. Sabia received grant funding from the French National Research Agency. Dr. Singh-Manoux received grants from the National Institute on Aging of the National Institutes of Health.
A version of this article first appeared on Medscape.com.
Investigators found that a healthy diet, cognitive activity, regular physical exercise, not smoking, and abstaining from alcohol were significantly linked to slowed cognitive decline irrespective of APOE4 status.
After adjusting for health and socioeconomic factors, investigators found that each individual healthy behavior was associated with a slower-than-average decline in memory over a decade. A healthy diet emerged as the strongest deterrent, followed by cognitive activity and physical exercise.
“A healthy lifestyle is associated with slower memory decline, even in the presence of the APOE4 allele,” study investigators led by Jianping Jia, MD, PhD, of the Innovation Center for Neurological Disorders and the department of neurology, Xuan Wu Hospital, Capital Medical University, Beijing, write.
“This study might offer important information to protect older adults against memory decline,” they add.
The study was published online in the BMJ.
Preventing memory decline
Memory “continuously declines as people age,” but age-related memory decline is not necessarily a prodrome of dementia and can “merely be senescent forgetfulness,” the investigators note. This can be “reversed or [can] become stable,” instead of progressing to a pathologic state.
Factors affecting memory include aging, APOE4 genotype, chronic diseases, and lifestyle patterns, with lifestyle “receiving increasing attention as a modifiable behavior.”
Nevertheless, few studies have focused on the impact of lifestyle on memory, and those that have are mostly cross-sectional and also “did not consider the interaction between a healthy lifestyle and genetic risk,” the researchers note.
To investigate, the researchers conducted a longitudinal study, known as the China Cognition and Aging Study, that considered genetic risk as well as lifestyle factors.
The study began in 2009 and concluded in 2019. Participants were evaluated and underwent neuropsychological testing in 2012, 2014, 2016, and at the study’s conclusion.
Participants (n = 29,072; mean [SD] age, 72.23 [6.61] years; 48.54% women; 20.43% APOE4 carriers) were required to have normal cognitive function at baseline. Data on those whose condition progressed to mild cognitive impairment (MCI) or dementia during the follow-up period were excluded after their diagnosis.
The Mini–Mental State Examination was used to assess global cognitive function. Memory function was assessed using the World Health Organization/University of California, Los Angeles Auditory Verbal Learning Test.
“Lifestyle” consisted of six modifiable factors: physical exercise (weekly frequency and total time), smoking (current, former, or never-smokers), alcohol consumption (never drank, drank occasionally, low to excess drinking, and heavy drinking), diet (daily intake of 12 food items: fruits, vegetables, fish, meat, dairy products, salt, oil, eggs, cereals, legumes, nuts, tea), cognitive activity (writing, reading, playing cards, mahjong, other games), and social contact (participating in meetings, attending parties, visiting friends/relatives, traveling, chatting online).
Participants’ lifestyles were scored on the basis of the number of healthy factors they engaged in.
Participants were also stratified by APOE genotype into APOE4 carriers and noncarriers.
Demographic and other items of health information, including the presence of medical illness, were used as covariates. The researchers also included the “learning effect of each participant as a covariate, due to repeated cognitive assessments.”
Important for public health
During the 10-year period, 7,164 participants died, and 3,567 stopped participating.
Participants in the favorable and average groups showed slower memory decline per increased year of age (0.007 [0.005-0.009], P < .001; and 0.002 [0 .000-0.003], P = .033 points higher, respectively), compared with those in the unfavorable group.
Healthy diet had the strongest protective effect on memory.
Memory decline occurred faster in APOE4 vesus non-APOE4 carriers (0.002 points/year [95% confidence interval, 0.001-0.003]; P = .007).
But APOE4 carriers with favorable and average lifestyles showed slower memory decline (0.027 [0.023-0.031] and 0.014 [0.010-0.019], respectively), compared with those with unfavorable lifestyles. Similar findings were obtained in non-APOE4 carriers.
Those with favorable or average lifestyle were respectively almost 90% and 30% less likely to develop dementia or MCI, compared with those with an unfavorable lifestyle.
The authors acknowledge the study’s limitations, including its observational design and the potential for measurement errors, owing to self-reporting of lifestyle factors. Additionally, some participants did not return for follow-up evaluations, leading to potential selection bias.
Nevertheless, the findings “might offer important information for public health to protect older [people] against memory decline,” they note – especially since the study “provides evidence that these effects also include individuals with the APOE4 allele.”
‘Important, encouraging’ research
In a comment, Severine Sabia, PhD, a senior researcher at the Université Paris Cité, INSERM Institut National de la Santé et de la Recherche Medicalé, France, called the findings “important and encouraging.”
However, said Dr. Sabia, who was not involved with the study, “there remain important research questions that need to be investigated in order to identify key behaviors: which combination, the cutoff of risk, and when to intervene.”
Future research on prevention “should examine a wider range of possible risk factors” and should also “identify specific exposures associated with the greatest risk, while also considering the risk threshold and age at exposure for each one.”
In an accompanying editorial, Dr. Sabia and co-author Archana Singh-Manoux, PhD, note that the risk of cognitive decline and dementia are probably determined by multiple factors.
They liken it to the “multifactorial risk paradigm introduced by the Framingham study,” which has “led to a substantial reduction in cardiovascular disease.” A similar approach could be used with dementia prevention, they suggest.
The authors received support from the Xuanwu Hospital of Capital Medical University for the submitted work. One of the authors received a grant from the French National Research Agency. The other authors have disclosed no relevant financial relationships. Dr. Sabia received grant funding from the French National Research Agency. Dr. Singh-Manoux received grants from the National Institute on Aging of the National Institutes of Health.
A version of this article first appeared on Medscape.com.
FROM THE BMJ
Even one head injury boosts all-cause mortality risk
An analysis of more than 13,000 adult participants in the Atherosclerosis Risk in Communities (ARIC) study showed a dose-response pattern in which one head injury was linked to a 66% increased risk for all-cause mortality, and two or more head injuries were associated with twice the risk in comparison with no head injuries.
These findings underscore the importance of preventing head injuries and of swift clinical intervention once a head injury occurs, lead author Holly Elser, MD, PhD, department of neurology, Hospital of the University of Pennsylvania, Philadelphia, told this news organization.
“Clinicians should counsel patients who are at risk for falls about head injuries and ensure patients are promptly evaluated in the hospital setting if they do have a fall – especially with loss of consciousness or other symptoms, such as headache or dizziness,” Dr. Elser added.
The findings were published online in JAMA Neurology.
Consistent evidence
There is “pretty consistent evidence” that mortality rates are increased in the short term after head injury, predominantly among hospitalized patients, Dr. Elser noted.
“But there’s less evidence about the long-term mortality implications of head injuries and less evidence from adults living in the community,” she added.
The analysis included 13,037 participants in the ARIC study, an ongoing study involving adults aged 45-65 years who were recruited from four geographically and racially diverse U.S. communities. The mean age at baseline (1987-1989) was 54 years; 57.7% were women; and 27.9% were Black.
Study participants are followed at routine in-person visits and semiannually via telephone.
Data on head injuries came from hospital diagnostic codes and self-reports. These reports included information on the number of injuries and whether the injury required medical care and involved loss of consciousness.
During the 27-year follow-up, 18.4% of the study sample had at least one head injury. Injuries occurred more frequently among women, which may reflect the predominance of women in the study population, said Dr. Elser.
Overall, about 56% of participants died during the study period. The estimated median amount of survival time after head injury was 4.7 years.
The most common causes of death were neoplasm, cardiovascular disease, and neurologic disorders. Regarding specific neurologic causes of death, the researchers found that 62.2% of deaths were due to neurodegenerative disease among individuals with head injury, vs. 51.4% among those without head injury.
This, said Dr. Elser, raises the possibility of reverse causality. “If you have a neurodegenerative disorder like Alzheimer’s disease dementia or Parkinson’s disease that leads to difficulty walking, you may be more likely to fall and have a head injury. The head injury in turn may lead to increased mortality,” she noted.
However, she stressed that the data on cause-specific mortality are exploratory. “Our research motivates future studies that really examine this time-dependent relationship between neurodegenerative disease and head injuries,” Dr. Elser said.
Dose-dependent response
In the unadjusted analysis, the hazard ratio of mortality among individuals with head injury was 2.21 (95% confidence interval, 2.09-2.34) compared with those who did not have head injury.
The association remained significant with adjustment for sociodemographic factors (HR, 1.99; 95% CI, 1.88-2.11) and with additional adjustment for vascular risk factors (HR, 1.92; 95% CI, 1.81-2.03).
The findings also showed a dose-response pattern in the association of head injuries with mortality. Compared with participants who did not have head injury, the HR was 1.66 (95% CI, 1.56-1.77) for those with one head injury and 2.11 (95% CI, 1.89-2.37) for those with two or more head injuries.
“It’s not as though once you’ve had one head injury, you’ve accrued all the damage you possibly can. We see pretty clearly here that recurrent head injury further increased the rate of deaths from all causes,” said Dr. Elser.
Injury severity was determined from hospital diagnostic codes using established algorithms. Results showed that mortality rates were increased with even mild head injury.
Interestingly, the association between head injury and all-cause mortality was weaker among those whose injuries were self-reported. One possibility is that these injuries were less severe, Dr. Elser noted.
“If you have head injury that’s mild enough that you don’t need to go to the hospital, it’s probably going to confer less long-term health risks than one that’s severe enough that you needed to be examined in an acute care setting,” she said.
Results were similar by race and for sex. “Even though there were more women with head injuries, the rate of mortality associated with head injury doesn’t differ from the rate among men,” Dr. Elser reported.
However, the association was stronger among those younger than 54 years at baseline (HR, 2.26) compared with older individuals (HR, 2.0) in the model that adjusted for demographics and lifestyle factors.
This may be explained by the reference group (those without a head injury) – the mortality rate was in general higher for the older participants, said Dr. Elser. It could also be that younger adults are more likely to have severe head injuries from, for example, motor vehicle accidents or violence, she added.
These new findings underscore the importance of public health measures, such as seatbelt laws, to reduce head injuries, the investigators note.
They add that clinicians with patients at risk for head injuries may recommend steps to lessen the risk of falls, such as having access to durable medical equipment, and ensuring driver safety.
Shorter life span
Commenting for this news organization, Frank Conidi, MD, director of the Florida Center for Headache and Sports Neurology in Port St. Lucie and past president of the Florida Society of Neurology, said the large number of participants “adds validity” to the finding that individuals with head injury are likely to have a shorter life span than those who do not suffer head trauma – and that this “was not purely by chance or from other causes.”
However, patients may not have accurately reported head injuries, in which case the rate of injury in the self-report subgroup would not reflect the actual incidence, noted Dr. Conidi, who was not involved with the research.
“In my practice, most patients have little knowledge as to the signs and symptoms of concussion and traumatic brain injury. Most think there needs to be some form of loss of consciousness to have a head injury, which is of course not true,” he said.
Dr. Conidi added that the finding of a higher incidence of death from neurodegenerative disorders supports the generally accepted consensus view that about 30% of patients with traumatic brain injury experience progression of symptoms and are at risk for early dementia.
The ARIC study is supported by the National Heart, Lung, and Blood Institute. Dr. Elser and Dr. Conidi have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
An analysis of more than 13,000 adult participants in the Atherosclerosis Risk in Communities (ARIC) study showed a dose-response pattern in which one head injury was linked to a 66% increased risk for all-cause mortality, and two or more head injuries were associated with twice the risk in comparison with no head injuries.
These findings underscore the importance of preventing head injuries and of swift clinical intervention once a head injury occurs, lead author Holly Elser, MD, PhD, department of neurology, Hospital of the University of Pennsylvania, Philadelphia, told this news organization.
“Clinicians should counsel patients who are at risk for falls about head injuries and ensure patients are promptly evaluated in the hospital setting if they do have a fall – especially with loss of consciousness or other symptoms, such as headache or dizziness,” Dr. Elser added.
The findings were published online in JAMA Neurology.
Consistent evidence
There is “pretty consistent evidence” that mortality rates are increased in the short term after head injury, predominantly among hospitalized patients, Dr. Elser noted.
“But there’s less evidence about the long-term mortality implications of head injuries and less evidence from adults living in the community,” she added.
The analysis included 13,037 participants in the ARIC study, an ongoing study involving adults aged 45-65 years who were recruited from four geographically and racially diverse U.S. communities. The mean age at baseline (1987-1989) was 54 years; 57.7% were women; and 27.9% were Black.
Study participants are followed at routine in-person visits and semiannually via telephone.
Data on head injuries came from hospital diagnostic codes and self-reports. These reports included information on the number of injuries and whether the injury required medical care and involved loss of consciousness.
During the 27-year follow-up, 18.4% of the study sample had at least one head injury. Injuries occurred more frequently among women, which may reflect the predominance of women in the study population, said Dr. Elser.
Overall, about 56% of participants died during the study period. The estimated median amount of survival time after head injury was 4.7 years.
The most common causes of death were neoplasm, cardiovascular disease, and neurologic disorders. Regarding specific neurologic causes of death, the researchers found that 62.2% of deaths were due to neurodegenerative disease among individuals with head injury, vs. 51.4% among those without head injury.
This, said Dr. Elser, raises the possibility of reverse causality. “If you have a neurodegenerative disorder like Alzheimer’s disease dementia or Parkinson’s disease that leads to difficulty walking, you may be more likely to fall and have a head injury. The head injury in turn may lead to increased mortality,” she noted.
However, she stressed that the data on cause-specific mortality are exploratory. “Our research motivates future studies that really examine this time-dependent relationship between neurodegenerative disease and head injuries,” Dr. Elser said.
Dose-dependent response
In the unadjusted analysis, the hazard ratio of mortality among individuals with head injury was 2.21 (95% confidence interval, 2.09-2.34) compared with those who did not have head injury.
The association remained significant with adjustment for sociodemographic factors (HR, 1.99; 95% CI, 1.88-2.11) and with additional adjustment for vascular risk factors (HR, 1.92; 95% CI, 1.81-2.03).
The findings also showed a dose-response pattern in the association of head injuries with mortality. Compared with participants who did not have head injury, the HR was 1.66 (95% CI, 1.56-1.77) for those with one head injury and 2.11 (95% CI, 1.89-2.37) for those with two or more head injuries.
“It’s not as though once you’ve had one head injury, you’ve accrued all the damage you possibly can. We see pretty clearly here that recurrent head injury further increased the rate of deaths from all causes,” said Dr. Elser.
Injury severity was determined from hospital diagnostic codes using established algorithms. Results showed that mortality rates were increased with even mild head injury.
Interestingly, the association between head injury and all-cause mortality was weaker among those whose injuries were self-reported. One possibility is that these injuries were less severe, Dr. Elser noted.
“If you have head injury that’s mild enough that you don’t need to go to the hospital, it’s probably going to confer less long-term health risks than one that’s severe enough that you needed to be examined in an acute care setting,” she said.
Results were similar by race and for sex. “Even though there were more women with head injuries, the rate of mortality associated with head injury doesn’t differ from the rate among men,” Dr. Elser reported.
However, the association was stronger among those younger than 54 years at baseline (HR, 2.26) compared with older individuals (HR, 2.0) in the model that adjusted for demographics and lifestyle factors.
This may be explained by the reference group (those without a head injury) – the mortality rate was in general higher for the older participants, said Dr. Elser. It could also be that younger adults are more likely to have severe head injuries from, for example, motor vehicle accidents or violence, she added.
These new findings underscore the importance of public health measures, such as seatbelt laws, to reduce head injuries, the investigators note.
They add that clinicians with patients at risk for head injuries may recommend steps to lessen the risk of falls, such as having access to durable medical equipment, and ensuring driver safety.
Shorter life span
Commenting for this news organization, Frank Conidi, MD, director of the Florida Center for Headache and Sports Neurology in Port St. Lucie and past president of the Florida Society of Neurology, said the large number of participants “adds validity” to the finding that individuals with head injury are likely to have a shorter life span than those who do not suffer head trauma – and that this “was not purely by chance or from other causes.”
However, patients may not have accurately reported head injuries, in which case the rate of injury in the self-report subgroup would not reflect the actual incidence, noted Dr. Conidi, who was not involved with the research.
“In my practice, most patients have little knowledge as to the signs and symptoms of concussion and traumatic brain injury. Most think there needs to be some form of loss of consciousness to have a head injury, which is of course not true,” he said.
Dr. Conidi added that the finding of a higher incidence of death from neurodegenerative disorders supports the generally accepted consensus view that about 30% of patients with traumatic brain injury experience progression of symptoms and are at risk for early dementia.
The ARIC study is supported by the National Heart, Lung, and Blood Institute. Dr. Elser and Dr. Conidi have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
An analysis of more than 13,000 adult participants in the Atherosclerosis Risk in Communities (ARIC) study showed a dose-response pattern in which one head injury was linked to a 66% increased risk for all-cause mortality, and two or more head injuries were associated with twice the risk in comparison with no head injuries.
These findings underscore the importance of preventing head injuries and of swift clinical intervention once a head injury occurs, lead author Holly Elser, MD, PhD, department of neurology, Hospital of the University of Pennsylvania, Philadelphia, told this news organization.
“Clinicians should counsel patients who are at risk for falls about head injuries and ensure patients are promptly evaluated in the hospital setting if they do have a fall – especially with loss of consciousness or other symptoms, such as headache or dizziness,” Dr. Elser added.
The findings were published online in JAMA Neurology.
Consistent evidence
There is “pretty consistent evidence” that mortality rates are increased in the short term after head injury, predominantly among hospitalized patients, Dr. Elser noted.
“But there’s less evidence about the long-term mortality implications of head injuries and less evidence from adults living in the community,” she added.
The analysis included 13,037 participants in the ARIC study, an ongoing study involving adults aged 45-65 years who were recruited from four geographically and racially diverse U.S. communities. The mean age at baseline (1987-1989) was 54 years; 57.7% were women; and 27.9% were Black.
Study participants are followed at routine in-person visits and semiannually via telephone.
Data on head injuries came from hospital diagnostic codes and self-reports. These reports included information on the number of injuries and whether the injury required medical care and involved loss of consciousness.
During the 27-year follow-up, 18.4% of the study sample had at least one head injury. Injuries occurred more frequently among women, which may reflect the predominance of women in the study population, said Dr. Elser.
Overall, about 56% of participants died during the study period. The estimated median amount of survival time after head injury was 4.7 years.
The most common causes of death were neoplasm, cardiovascular disease, and neurologic disorders. Regarding specific neurologic causes of death, the researchers found that 62.2% of deaths were due to neurodegenerative disease among individuals with head injury, vs. 51.4% among those without head injury.
This, said Dr. Elser, raises the possibility of reverse causality. “If you have a neurodegenerative disorder like Alzheimer’s disease dementia or Parkinson’s disease that leads to difficulty walking, you may be more likely to fall and have a head injury. The head injury in turn may lead to increased mortality,” she noted.
However, she stressed that the data on cause-specific mortality are exploratory. “Our research motivates future studies that really examine this time-dependent relationship between neurodegenerative disease and head injuries,” Dr. Elser said.
Dose-dependent response
In the unadjusted analysis, the hazard ratio of mortality among individuals with head injury was 2.21 (95% confidence interval, 2.09-2.34) compared with those who did not have head injury.
The association remained significant with adjustment for sociodemographic factors (HR, 1.99; 95% CI, 1.88-2.11) and with additional adjustment for vascular risk factors (HR, 1.92; 95% CI, 1.81-2.03).
The findings also showed a dose-response pattern in the association of head injuries with mortality. Compared with participants who did not have head injury, the HR was 1.66 (95% CI, 1.56-1.77) for those with one head injury and 2.11 (95% CI, 1.89-2.37) for those with two or more head injuries.
“It’s not as though once you’ve had one head injury, you’ve accrued all the damage you possibly can. We see pretty clearly here that recurrent head injury further increased the rate of deaths from all causes,” said Dr. Elser.
Injury severity was determined from hospital diagnostic codes using established algorithms. Results showed that mortality rates were increased with even mild head injury.
Interestingly, the association between head injury and all-cause mortality was weaker among those whose injuries were self-reported. One possibility is that these injuries were less severe, Dr. Elser noted.
“If you have head injury that’s mild enough that you don’t need to go to the hospital, it’s probably going to confer less long-term health risks than one that’s severe enough that you needed to be examined in an acute care setting,” she said.
Results were similar by race and for sex. “Even though there were more women with head injuries, the rate of mortality associated with head injury doesn’t differ from the rate among men,” Dr. Elser reported.
However, the association was stronger among those younger than 54 years at baseline (HR, 2.26) compared with older individuals (HR, 2.0) in the model that adjusted for demographics and lifestyle factors.
This may be explained by the reference group (those without a head injury) – the mortality rate was in general higher for the older participants, said Dr. Elser. It could also be that younger adults are more likely to have severe head injuries from, for example, motor vehicle accidents or violence, she added.
These new findings underscore the importance of public health measures, such as seatbelt laws, to reduce head injuries, the investigators note.
They add that clinicians with patients at risk for head injuries may recommend steps to lessen the risk of falls, such as having access to durable medical equipment, and ensuring driver safety.
Shorter life span
Commenting for this news organization, Frank Conidi, MD, director of the Florida Center for Headache and Sports Neurology in Port St. Lucie and past president of the Florida Society of Neurology, said the large number of participants “adds validity” to the finding that individuals with head injury are likely to have a shorter life span than those who do not suffer head trauma – and that this “was not purely by chance or from other causes.”
However, patients may not have accurately reported head injuries, in which case the rate of injury in the self-report subgroup would not reflect the actual incidence, noted Dr. Conidi, who was not involved with the research.
“In my practice, most patients have little knowledge as to the signs and symptoms of concussion and traumatic brain injury. Most think there needs to be some form of loss of consciousness to have a head injury, which is of course not true,” he said.
Dr. Conidi added that the finding of a higher incidence of death from neurodegenerative disorders supports the generally accepted consensus view that about 30% of patients with traumatic brain injury experience progression of symptoms and are at risk for early dementia.
The ARIC study is supported by the National Heart, Lung, and Blood Institute. Dr. Elser and Dr. Conidi have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM JAMA NEUROLOGY
Tips and tools to help you manage ADHD in children, adolescents
THE CASE
James B* is a 7-year-old Black child who presented to his primary care physician (PCP) for a well-child visit. During preventive health screening, James’ mother expressed concerns about his behavior, characterizing him as immature, aggressive, destructive, and occasionally self-loathing. She described him as physically uncoordinated, struggling to keep up with his peers in sports, and tiring after 20 minutes of activity. James slept 10 hours nightly but was often restless and snored intermittently. As a second grader, his academic achievement was not progressing, and he had become increasingly inattentive at home and at school. James’ mother offered several examples of his fighting with his siblings, noncompliance with morning routines, and avoidance of learning activities. Additionally, his mother expressed concern that James, as a Black child, might eventually be unfairly labeled as a problem child by his teachers or held back a grade level in school.
Although James did not have a family history of developmental delays or learning disorders, he had not met any milestones on time for gross or fine motor, language, cognitive, and social-emotional skills. James had a history of chronic otitis media, for which pressure equalizer tubes were inserted at age 2 years. He had not had any major physical injuries, psychological trauma, recent life transitions, or adverse childhood events. When asked, James’ mother acknowledged symptoms of maternal depression but alluded to faith-based reasons for not seeking treatment for herself.
James’ physical examination was unremarkable. His height, weight, and vitals were all within normal limits. However, he had some difficulty with verbal articulation and expression and showed signs of a possible vocal tic. Based on James’ presentation, his PCP suspected attention-deficit/hyperactivity disorder (ADHD), as well as neurodevelopmental delays.
The PCP gave James’ mother the Strengths and Difficulties Questionnaire to complete and the Vanderbilt Assessment Scales for her and James’ teacher to fill out independently and return to the clinic. The PCP also instructed James’ mother on how to use a sleep diary to maintain a 1-month log of his sleep patterns and habits. The PCP consulted the integrated behavioral health clinician (IBHC; a clinical social worker embedded in the primary care clinic) and made a warm handoff for the IBHC to further assess James’ maladaptive behaviors and interactions.
●
* The patient’s name has been changed to protect his identity.
James is one of more than 6 million children, ages 3 to 17 years, in the United States who live with ADHD.1,2 ADHD is the most common neurodevelopmental disorder among children, and it affects multiple cognitive and behavioral domains throughout the lifespan.3 Children with ADHD often initially present in primary care settings; thus, PCPs are well positioned to diagnose the disorder and provide longitudinal treatment. This Behavioral Health Consult reviews clinical assessment and practice guidelines, as well as treatment recommendations applicable across different areas of influence—individual, family, community, and systems—for PCPs and IBHCs to use in managing ADHD in children.
ADHD features can vary by age and sex
ADHD is a persistent pattern of inattention or hyperactivity and impulsivity interfering with functioning or development in childhood and functioning later in adulthood. ADHD symptoms manifest prior to age 12 years and must occur in 2 or more settings.4 Symptoms should not be better explained by another psychiatric disorder or occur exclusively during the course of another disorder (TABLE 1).4
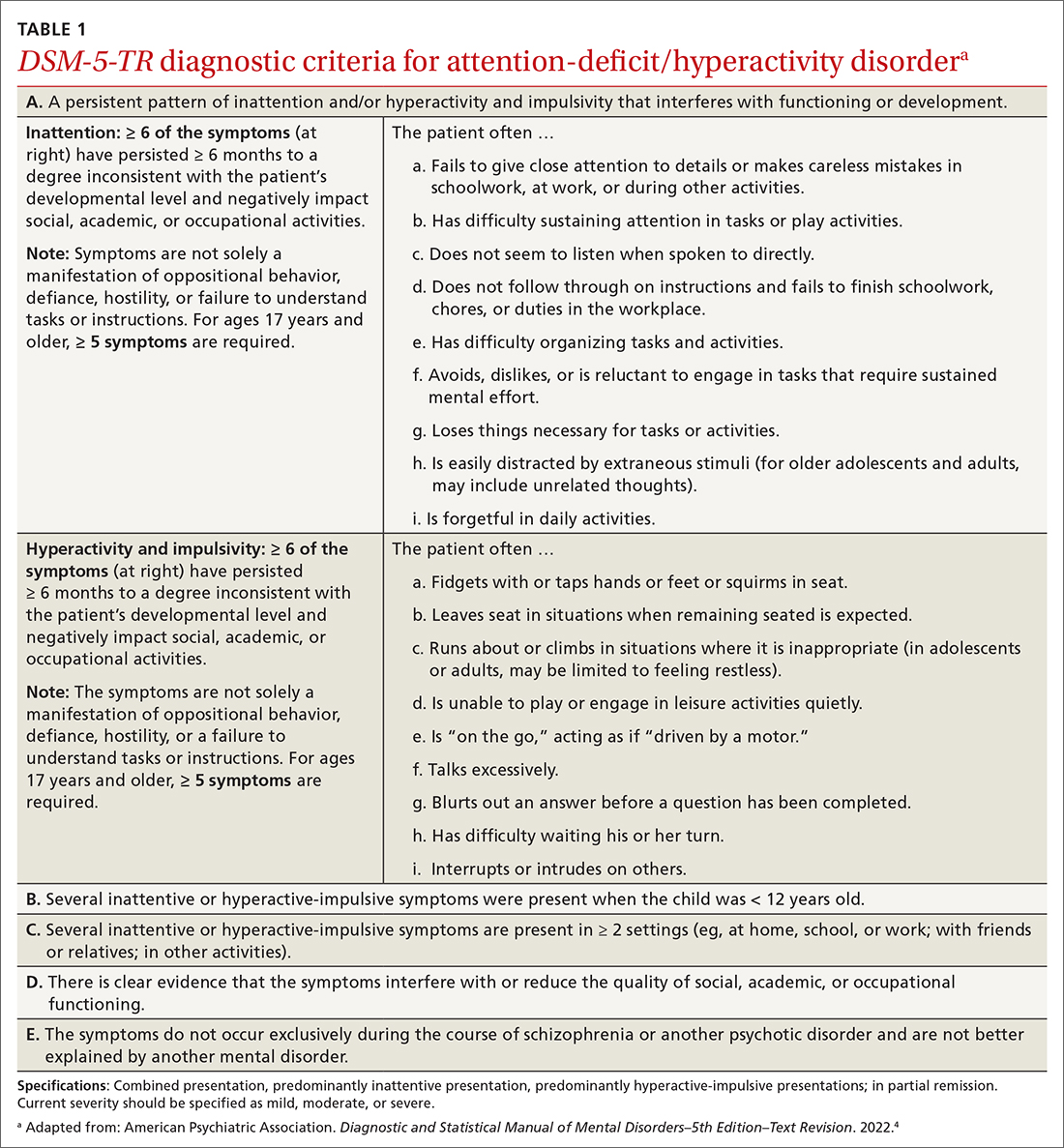
The rate of heritability is high, with significant incidence among first-degree relatives.4 Children with ADHD show executive functioning deficits in 1 or more cognitive domains (eg, visuospatial, memory, inhibitions, decision making, and reward regulation).4,5 The prevalence of ADHD nationally is approximately 9.8% (2.2%, ages 3-5 years; 10%, ages 6-11 years; 13.2%, ages 12-17 years) in children and adolescents; worldwide prevalence is 7.2%.1,6 It persists among 2.6% to 6.8% of adults worldwide.7
Research has shown that boys ages 6 to 11 years are significantly more likely than girls to exhibit attention-getting, externalizing behaviors or conduct problems (eg, hyperactivity, impulsivity, disruption, aggression).1,6 On the other hand, girls ages 12 to 17 years tend to display internalized (eg, depressed mood, anxiety, low self-esteem) or inattentive behaviors, which clinicians and educators may assess as less severe and warranting fewer supportive measures.1
The prevalence of ADHD and its associated factors, which evolve through maturation, underscore the importance of persistent, patient-centered, and collaborative PCP and IBHC clinical management.
Continue to: Begin with a screening tool, move to a clinical interview
Begin with a screening tool, move to a clinical interview
When caregivers express concerns about their child’s behavior, focus, mood, learning, and socialization, consider initiating a multimodal evaluation for ADHD.5,8 Embarking on an ADHD assessment can require extended or multiple visits to arrive at the diagnosis, followed by still more visits to confirm a course of care and adjust medications. The integrative care approach described in the patient case and elaborated on later in this article can help facilitate assessment and treatment of ADHD.9
Signs of ADHD may be observed at initial screening using a tool such as the Ages & Stages Questionnaire (https://agesandstages.com/products-pricing/asq3/) to reveal indications of norm deviations or delays commensurate with ADHD.10 However, to substantiate the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, Text Revision criteria for an accurate diagnosis,4 the American Academy of Pediatrics (AAP) clinical practice guidelines require a thorough clinical interview, administration of a standardized assessment tool, and review of objective reports in conjunction with a physical examination and psychosocial evaluation.6 Standardized measures of psychological, neurocognitive, and academic achievement reported by caregivers and collateral contacts (eg, teachers, counselors, coaches, care providers) are needed to maximize data objectivity and symptom accuracy across settings (TABLE 210-17). Additionally, periodic reassessment is recommended to validate changes in diagnostic subtype and treatment plans due to the chronic and dynamic nature of ADHD.
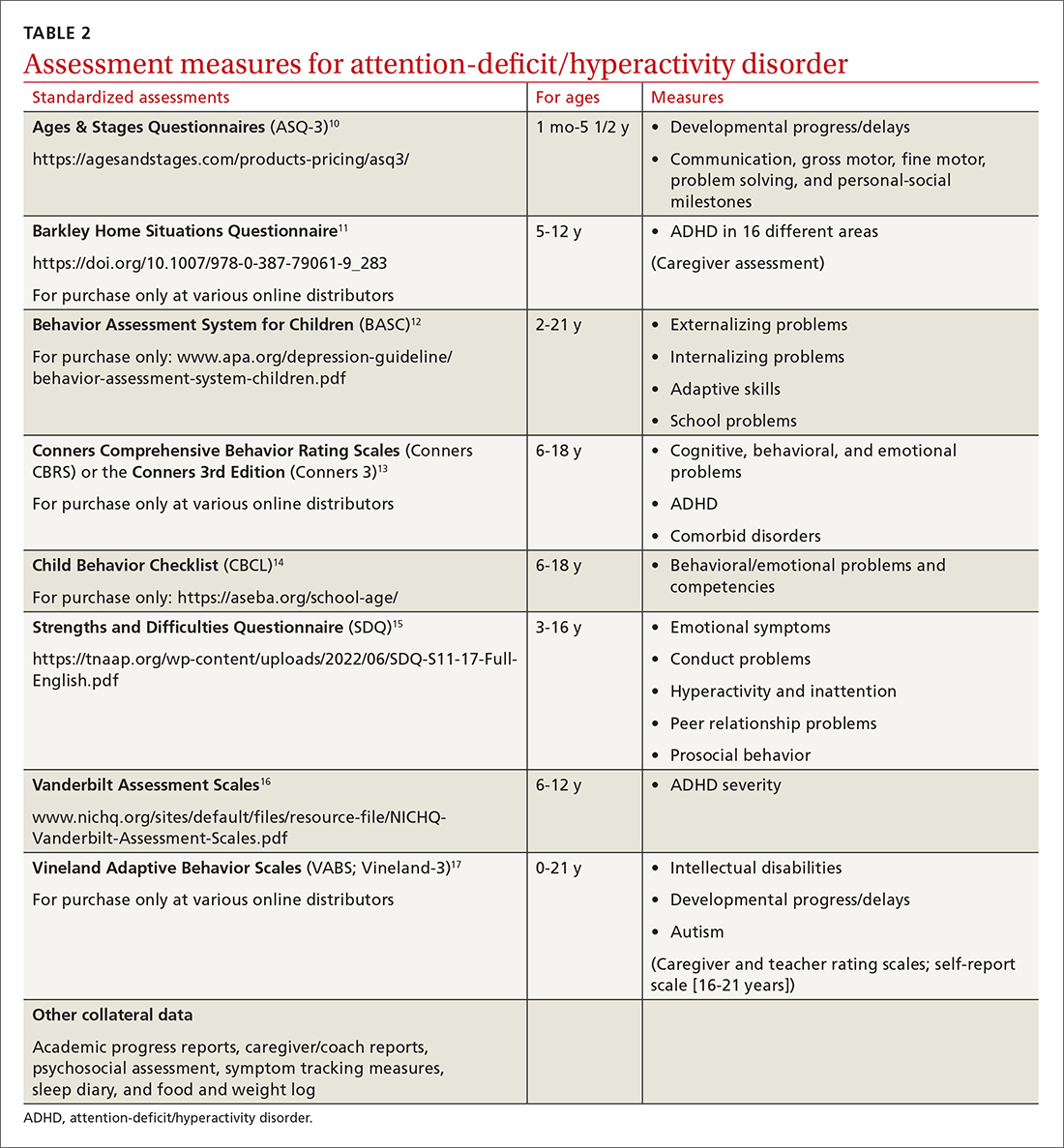
Consider comorbidities and alternate diagnoses
The diagnostic possibility of ADHD should also prompt consideration of other childhood disorders due to the high potential for comorbidities.4,6 In a 2016 study, approximately 64% of
Various medical disorders may manifest with similar signs or symptoms to ADHD, such as thyroid disorders, seizure disorders, adverse drug effects, anemia, genetic anomalies, and others.6,19
If there are behavioral concerns or developmental delays associated with tall stature for age or pubertal or testicular development anomalies, consult a geneticist and a developmental pediatrician for targeted testing and neurodevelopmental assessment, respectively. For example, ADHD is a common comorbidity among boys who also have XYY syndrome (Jacobs syndrome). However, due to the variability of symptoms and severity, XYY syndrome often goes undiagnosed, leaving a host of compounding pervasive and developmental problems untreated. Overall, more than two-thirds of patients with ADHD and a co-occurring condition are either inaccurately diagnosed or not referred for additional assessment and adjunct treatment.21
Continue to: Risks that arise over time
Risks that arise over time. As ADHD persists, adolescents are at greater risk for psychiatric comorbidities, suicidality, and functional impairments (eg, risky behaviors, occupational problems, truancy, delinquency, and poor self-esteem).4,8 Adolescents with internalized behaviors are more likely to experience comorbid depressive disorders with increased risk for self-harm.4,5,8 As adolescents age and their sense of autonomy increases, there is a tendency among those who have received a diagnosis of ADHD to minimize symptoms and decrease the frequency of routine clinic visits along with medication use and treatment compliance.3 Additionally, abuse, misuse, and misappropriation of stimulants among teens and young adults are commonplace.
Wide-scope, multidisciplinary evaluation and close clinical management reduce the potential for imprecise diagnoses, particularly at critical developmental junctures. AAP suggests that PCPs can treat mild and moderate cases of ADHD, but if the treating clinician does not have adequate training, experience, time, or clinical support to manage this condition, early referral is warranted.6
A guide to pharmacotherapy
Approximately 77% of children ages 2 to 17 years with a diagnosis of ADHD receive any form of treatment.2 Treatment for ADHD can include behavioral therapy and medication.2 AAP clinical practice guidelines caution against prescribing medications for children younger than 6 years, relying instead on caregiver-, teacher-, or clinician-administered behavioral strategies and parental training in behavioral modification. For children and adolescents between ages 6 and 18 years, first-line treatment includes pharmacotherapy balanced with behavioral therapy, academic modifications, and educational supports (eg, 504 Plan, individualized education plan [IEP]).6
Psychostimulants are preferred. These agents (eg, methylphenidate, amphetamine) remain the most efficacious class of medications to reduce hyperactivity and inattentiveness and to improve function. While long-acting psychostimulants are associated with better medication adherence and adverse-effect tolerance than are short-acting forms, the latter offer more flexibility in dosing. Start by titrating any stimulant to the lowest effective dose; reassess monthly until potential rebound effects stabilize.
Due to potential adverse effects of this class of medication, screen for any family history or personal risk for structural or electrical cardiac anomalies before starting pharmacotherapy. If any such risks exist, arrange for further cardiac evaluation before initiating medication.6 Adverse effects of stimulants include reduced appetite, gastrointestinal symptoms, headaches, anxiousness, parasomnia, tachycardia, and hypertension.
Continue to: Once medication is stabilized...
Once medication is stabilized, monitor treatment 2 to 3 times per year thereafter; watch for longer-term adverse effects such as weight loss, decreased growth rate, and psychiatric comorbidities including the Food and Drug Administration (FDA)’s black box warning of increased risk for suicidality.5,6,22
Other options. The optimal duration of psychostimulant use remains debatable, as existing evidence does not support its long-term use (10 years) over other interventions, such as nonstimulants and nonmedicinal therapies.22 Although backed by less evidence, additional medications indicated for the treatment of ADHD include: (1) atomoxetine, a selective norepinephrine reuptake inhibitor, and (2) the selective alpha-2 adrenergic agonists, extended-release guanfacine and extended-release clonidine (third-line agent).22
Adverse effects of these FDA-approved medications are similar to those observed in stimulant medications. Evaluation of cardiac risks is recommended before starting nonstimulant medications. The alpha-2 adrenergic agonists may also be used as adjunct therapies to stimulants. Before stopping an alpha-2 adrenergic agonist, taper the dosage slowly to avoid the risk for rebound hypertension.6,23 Given the wide variety of medication options and variability of effects, it may be necessary to try different medications as children grow and their symptoms and capacity to manage them change. Additional guidance on FDA-approved medications is available at www.ADHDMedicationGuide.com.
How multilevel care coordination can work
As with other chronic or developmental conditions, the treatment of ADHD requires an interdisciplinary perspective. Continuous, comprehensive case management can help patients overcome obstacles to wellness by balancing the resolution of problems with the development of resilience. Well-documented collaboration of subspecialists, educators, and other stakeholders engaged in ADHD care at multiple levels (individual, family, community, and health care system) increases the likelihood of meaningful, sustainable gains. Using a patient-centered medical home framework, IBHCs or other allied health professionals embedded in, or co-located with, primary care settings can be key to accessing evidence-based treatments that include: psycho-education and mindfulness-based stress reduction training for caregivers24,25; occupational,26 cognitive behavioral,27 or family therapies28,29; neuro-feedback; computer-based attention training; group- or community-based interventions; and academic and social supports.5,8
Treatment approaches that capitalize on children’s neurologic and psychological plasticity and fortify self-efficacy with developmentally appropriate tools empower them to surmount ADHD symptoms over time.23 Facilitating children’s resilience within a developmental framework and health system’s capacities with socio-culturally relevant approaches, consultation, and research can optimize outcomes and mitigate pervasiveness into adulthood. While the patient is at the center of treatment, it is important to consider the family, school, and communities in which the child lives, learns, and plays. PCPs and IBHCs together can consider a “try and track” method to follow progress, changes, and outcomes over time. With this method, the physician can employ approaches that focus on the patient, caregiver, or the caregiver–child interaction (TABLE 3).
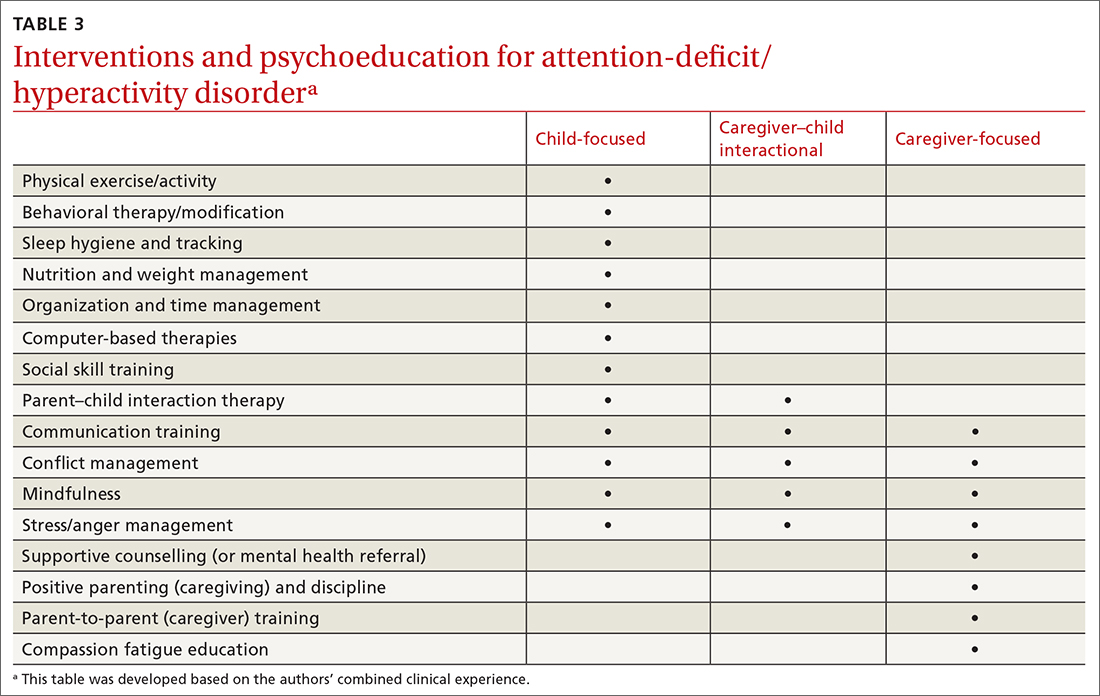
Continue to: Assess patients' needs and the resources available
Assess patients’ needs and the resources available throughout the system of care beyond the primary care setting. Stay abreast of hospital policies, health care insurance coverage, and community- and school-based health programs, and any gaps in adequate and equitable assessment and treatment. For example, while clinical recommendations include psychiatric care, health insurance availability or limits in coverage may dissuade caregivers from seeking help or limit initial or long-term access to resources for help.30 Integrating or advocating for clinic support resources or staffing to assist patients in navigating and mitigating challenges may lessen the management burden and increase the likelihood and longevity of favorable health outcomes.
Steps to ensuring health care equity
Among children of historically marginalized and racial and ethnic minority groups or those of populations affected by health disparities, ADHD symptoms and needs are often masked by structural biases that lead to inequitable care and outcomes, as well as treatment misprioritization or delays.31 In particular, evidence has shown that recognition and diagnostic specificity of ADHD and comorbidities, not prevalence, vary more widely among minority than among nonminority populations,32 contributing to the 23% of children with ADHD who receive no treatment at all.2
Understand caregiver concerns. This diagnosis discrepancy is correlated with symptom rating sensitivities (eg, reliability, perception, accuracy) among informants and how caregivers observe, perceive, appreciate, understand, and report behaviors. This discrepancy is also related to cultural belief differences, physician–patient communication variants, and a litany of other socioeconomic determinants.2,4,31 Caregivers from some cultural, ethnic, or socioeconomic backgrounds may be doubtful of psychiatric assessment, diagnoses, treatment, or medication, and that can impact how children are engaged in clinical and educational settings from the outset.31 In the case we described, James’ mother was initially hesitant to explore psychotropic medications and was concerned about stigmatization within the school system. She also seemed to avoid psychiatric treatment for her own depressive symptoms due to cultural and religious beliefs.
Health care provider concerns. Some PCPs may hesitate to explore medications due to limited knowledge and skill in dosing and titrating based on a child’s age, stage, and symptoms, and a perceived lack of competence in managing ADHD. This, too, can indirectly perpetuate existing health disparities. Furthermore, ADHD symptoms may be deemed a secondary or tertiary concern if other complex or urgent medical or undifferentiated developmental problems manifest.
Compounding matters is the limited dissemination of empiric research articles (including randomized controlled trials with representative samples) and limited education on the effectiveness and safety of psychopharmacologic interventions across the lifespan and different cultural and ethnic groups.4 Consequently, patients who struggle with unmanaged ADHD symptoms are more likely to have chronic mental health disorders, maladaptive behaviors, and other co-occurring conditions contributing to the complexity of individual needs, health care burdens, or justice system involvement; this is particularly true for those of racial and ethnic minorities.33
Continue to: Impact of the COVID-19 pandemic
Impact of the COVID-19 pandemic. Patients—particularly those in minority or health disparity populations—who under normal circumstances might have been hesitant to seek help may have felt even more reluctant to do so during the COVID-19 pandemic. We have not yet learned the degree to which limited availability of preventive health care services, decreased routine visits, and fluctuating insurance coverage has impacted the diagnosis, management, or severity of childhood disorders during the past 2 years. Reports of national findings indicate that prolonged periods out of school and reduced daily structure were associated with increased disruptions in mood, sleep, and appetite, particularly among children with pre-existing pathologies. Evidence suggests that school-aged children experienced more anxiety, regressive behaviors, and parasomnias than they did before the pandemic, while adolescents experienced more isolation and depressive symptoms.34,35
However, there remains a paucity of large-scale or representative studies that use an intersectional lens to examine the influence of COVID-19 on children with ADHD. Therefore, PCPs and IBHCs should refocus attention on possibly undiagnosed, stagnated, or regressed ADHD cases, as well as the adults who care for them. (See “5 ways to overcome Tx barriers and promote health equity.”)
SIDEBAR
5 ways to overcome Tx barriers and promote health equitya
1. Inquire about cultural or ethnic beliefs and behaviors and socioeconomic barriers.
2. Establish trust or assuage mistrust by exploring and dispelling misinformation.
3. Offer accessible, feasible, and sustainable evidence-based interventions.
4. Encourage autonomy and selfdetermination throughout the health care process.
5. Connect caregivers and children with clinical, community, and school-based resources and coordinators.
a These recommendations are based on the authors’ combined clinical experience.
THE CASE
During a follow-up visit 1 month later, the PCP confirmed the clinical impression of ADHD combined presentation with a clinical interview and review of the Strengths and Difficulties Questionnaire completed by James’ mother and the Vanderbilt Assessment Scales completed by James’ mother and teacher. The sleep diary indicated potential problems and apneas worthy of consults for pulmonary function testing, a sleep study, and otolaryngology examination. The PCP informed James’ mother on sleep hygiene strategies and ADHD medication options. She indicated that she wanted to pursue the referrals and behavioral modifications before starting any medication trial.
The PCP referred James to a developmental pediatrician for in-depth assessment of his overall development, learning, and functioning. The developmental pediatrician ultimately confirmed the diagnosis of ADHD, as well as motor and speech delays warranting physical, occupational, and speech therapies. The developmental pediatrician also referred James for targeted genetic testing because she suspected a genetic disorder (eg, XYY syndrome).
The PCP reconnected James and his mother to the IBHC to facilitate subspecialty and school-based care coordination and to provide in-office and home-based interventions. The IBHC assessed James’ emotional dysregulation and impulsivity as adversely impacting his interpersonal relationships and planned to address these issues with behavioral and parent–child interaction therapies and skills training during the course of 6 to 12 visits. James’ mother was encouraged to engage his teacher on his academic performance and to initiate a 504 Plan or IEP for in-school accommodations and support. The IBHC aided in tracking his assessments, referrals, follow-ups, access barriers, and treatment goals.
After 6 months, James had made only modest progress, and his mother requested that he begin a trial of medication. Based on his weight, symptoms, behavior patterns, and sleep habits, the PCP prescribed extended-release dexmethylphenidate 10 mg each morning, then extended-release clonidine 0.1 mg nightly. With team-based clinical management of pharmacologic, behavioral, physical, speech, and occupational therapies, James’ behavior and sleep improved, and the signs of a vocal tic diminished.
By the next school year, James demonstrated a marked improvement in impulse control, attention, and academic functioning. He followed up with the PCP at least quarterly for reassessment of his symptoms, growth, and experience of adverse effects, and to titrate medications accordingly. James and his mother continued to work closely with the IBHC monthly to engage interventions and to monitor his progress at home and school.
CORRESPONDENCE
Sundania J. W. Wonnum, PhD, LCSW, National Institute on Minority Health and Health Disparities, 6707 Democracy Boulevard, Suite 800, Bethesda, MD 20892; [email protected]
1. Bitsko RH, Claussen AH, Lichstein J, et al. Mental health surveillance among children—United States, 2013-2019. MMWR Suppl. 2022;71:1-42. doi: 10.15585/mmwr.su7102a1
2. Danielson ML, Holbrook JR, Blumberg SJ, et al. State-level estimates of the prevalence of parent-reported ADHD diagnosis and treatment among U.S. children and adolescents, 2016 to 2019. J Atten Disord. 2022;26:1685-1697. doi: 10.1177/10870547221099961
3. Faraone SV, Banaschewski T, Coghill D, et al. The World Federation of ADHD International Consensus Statement: 208 evidence-based conclusions about the disorder. Neurosci Biobehav Rev. 2021;128:789-818. doi: 10.1016/j.neubiorev.2021.01.022
4. American Psychiatric Association
5. Brahmbhatt K, Hilty DM, Mina H, et al. Diagnosis and treatment of attention deficit hyperactivity disorder during adolescence in the primary care setting: a concise review. J Adolesc Health. 2016;59:135-143. doi: 10.1016/j.jadohealth.2016.03.025
6. Wolraich ML, Hagan JF, Allan C, et al. AAP Subcommittee on Children and Adolescents with Attention-Deficit/Hyperactivity Disorder. Clinical Practice Guideline for the Diagnosis, Evaluation, and Treatment of Attention-Deficit/Hyperactivity Disorder in Children and Adolescents. Pediatrics. 2019;144:e20192528. doi: 10.1542/peds.2019-2528
7. Song P, Zha M, Yang Q, et al. The prevalence of adult attention-deficit hyperactivity disorder: a global systematic review and meta-analysis. J Glob Health. 2021;11:04009. doi: 10.7189/jogh.11.04009
8. Chang JG, Cimino FM, Gossa W. ADHD in children: common questions and answers. Am Fam Physician. 2020;102:592-602.
9. Asarnow JR, Rozenman M, Wiblin J, et al. Integrated medical-behavioral care compared with usual primary care for child and adolescent behavioral health: a meta-analysis. JAMA Pediatr. 2015;169:929-937. doi: 10.1001/jamapediatrics.2015.1141
10. Squires J, Bricker D. Ages & Stages Questionnaires®. 3rd ed (ASQ®-3). Paul H. Brookes Publishing Co., Inc; 2009.
11. DuPaul GJ, Barkley RA. Situational variability of attention problems: psychometric properties of the Revised Home and School Situations Questionnaires. J Clin Child Psychol. 1992;21:178-188. doi.org/10.1207/s15374424jccp2102_10
12. Merenda PF. BASC: behavior assessment system for children. Meas Eval Counsel Develop. 1996;28:229-232.
13. Conners CK. Conners, 3rd ed manual. Multi-Health Systems. 2008.
14. Achenbach TM. The Child Behavior Checklist and related instruments. In: Maruish ME, ed. The Use of Psychological Testing for Treatment Planning and Outcomes Assessment. Lawrence Erlbaum Associates Publishers; 1999:429-466.
15. Goodman R. The extended version of the Strengths and Difficulties Questionnaire as a guide to child psychiatric caseness and consequent burden. J Child Psychol Psychiatry. 1999;40:791-799.
16. Wolraich ML, Lambert W, Doffing MA, et al. Psychometric properties of the Vanderbilt ADHD Diagnostic Parent Rating Scale in a referred population. J Pediatr Psychol. 2003;28:559-567. doi: 10.1093/jpepsy/jsg046
17. Sparrow SS, Cicchetti DV. The Vineland Adaptive Behavior Scales. In: Newmark CS, ed. Major Psychological Assessment Instruments. Vol 2. Allyn & Bacon; 2003:199-231.
18. Danielson ML, Bitsko RH, Ghandour RM, et al. Prevalence of parent-reported ADHD diagnosis and associated treatment among U.S. children and adolescents, 2016. J Clin Child Adolesc Psychol. 2018;47:199-212. doi: 10.1080/15374416.2017.1417860
19. Ghriwati NA, Langberg JM, Gardner W, et al. Impact of mental health comorbidities on the community-based pediatric treatment and outcomes of children with attention deficit hyperactivity disorder. J Dev Behav Ped. 2017;38:20-28. doi: 10.1097/DBP.0000000000000359
20. Niclasen J, Obel C, Homøe P, et al. Associations between otitis media and child behavioural and learning difficulties: results from a Danish Cohort. Int J Ped Otorhinolaryngol. 2016;84:12-20. doi: 10.1016/j.ijporl.2016.02.017
21. Ross JL Roeltgen DP Kushner H, et al. Behavioral and social phenotypes in boys with 47,XYY syndrome or 47,XXY Klinefelter syndrome. doi: 10.1542/peds.2011-0719
22. Mechler K, Banaschewski T, Hohmann S, et al. Evidence-based pharmacological treatment options for ADHD in children and adolescents. Pharmacol Ther. 2022;230:107940. doi: 10.1016/j.pharmthera.2021.107940
23. Mishra J, Merzenich MM, Sagar R. Accessible online neuroplasticity-targeted training for children with ADHD. Child Adolesc Psychiatry Ment Health. 2013;7:38. doi: 10.1186/1753-2000-7-38
24. Neece CL. Mindfulness-based stress reduction for parents of young children with developmental delays: implications for parental mental health and child behavior problems. J Applied Res Intellect Disabil. 2014;27:174-186. doi: 10.1111/jar.12064
25. Petcharat M, Liehr P. Mindfulness training for parents of children with special needs: guidance for nurses in mental health practice. J Child Adolesc Psychiatr Nursing. 2017;30:35-46. doi: 10.1111/jcap.12169
26. Hahn-Markowitz J, Burger I, Manor I, et al. Efficacy of cognitive-functional (Cog-Fun) occupational therapy intervention among children with ADHD: an RCT. J Atten Disord. 2020;24:655-666. doi: 10.1177/1087054716666955
27. Young Z, Moghaddam N, Tickle A. The efficacy of cognitive behavioral therapy for adults with ADHD: a systematic review and meta-analysis of randomized controlled trials. J Atten Disord. 2020;24:875-888.
28. Carr AW, Bean RA, Nelson KF. Childhood attention-deficit hyperactivity disorder: family therapy from an attachment based perspective. Child Youth Serv Rev. 2020;119:105666.
29. Robin AL. Family therapy for adolescents with ADHD. Child Adolesc Psychiatr Clin N Am. 2014;23:747-756. doi: 10.1016/j.chc.2014.06.001
30. Cattoi B, Alpern I, Katz JS, et al. The adverse health outcomes, economic burden, and public health implications of unmanaged attention deficit hyperactivity disorder (ADHD): a call to action resulting from CHADD summit, Washington, DC, October 17, 2019. J Atten Disord. 2022;26:807-808. doi: 10.1177/10870547211036754
31. Hinojosa MS, Hinojosa R, Nguyen J. Shared decision making and treatment for minority children with ADHD. J Transcult Nurs. 2020;31:135-143. doi: 10.1177/1043659619853021
32. Slobodin O, Masalha R. Challenges in ADHD care for ethnic minority children: a review of the current literature. Transcult Psychiatry. 2020;57:468-483. doi: 10.1177/1363461520902885
33. Retz W, Ginsberg Y, Turner D, et al. Attention-deficit/hyperactivity disorder (ADHD), antisociality and delinquent behavior over the lifespan. Neurosci Biobehav Rev. 2021;120:236-248. doi: 10.1016/j.neubiorev.2020.11.025
34. Del Sol Calderon P, Izquierdo A, Garcia Moreno M. Effects of the pandemic on the mental health of children and adolescents. Review and current scientific evidence of the SARS-COV2 pandemic. Eur Psychiatry. 2021;64:S223-S224. doi: 10.1192/j.eurpsy.2021.597
35. Insa I, Alda JA. Attention deficit hyperactivity disorder (ADHD) & COVID-19: attention deficit hyperactivity disorder: consequences of the 1st wave. Eur Psychiatry. 2021;64:S660. doi: 10.1192/j.eurpsy.2021.1752
THE CASE
James B* is a 7-year-old Black child who presented to his primary care physician (PCP) for a well-child visit. During preventive health screening, James’ mother expressed concerns about his behavior, characterizing him as immature, aggressive, destructive, and occasionally self-loathing. She described him as physically uncoordinated, struggling to keep up with his peers in sports, and tiring after 20 minutes of activity. James slept 10 hours nightly but was often restless and snored intermittently. As a second grader, his academic achievement was not progressing, and he had become increasingly inattentive at home and at school. James’ mother offered several examples of his fighting with his siblings, noncompliance with morning routines, and avoidance of learning activities. Additionally, his mother expressed concern that James, as a Black child, might eventually be unfairly labeled as a problem child by his teachers or held back a grade level in school.
Although James did not have a family history of developmental delays or learning disorders, he had not met any milestones on time for gross or fine motor, language, cognitive, and social-emotional skills. James had a history of chronic otitis media, for which pressure equalizer tubes were inserted at age 2 years. He had not had any major physical injuries, psychological trauma, recent life transitions, or adverse childhood events. When asked, James’ mother acknowledged symptoms of maternal depression but alluded to faith-based reasons for not seeking treatment for herself.
James’ physical examination was unremarkable. His height, weight, and vitals were all within normal limits. However, he had some difficulty with verbal articulation and expression and showed signs of a possible vocal tic. Based on James’ presentation, his PCP suspected attention-deficit/hyperactivity disorder (ADHD), as well as neurodevelopmental delays.
The PCP gave James’ mother the Strengths and Difficulties Questionnaire to complete and the Vanderbilt Assessment Scales for her and James’ teacher to fill out independently and return to the clinic. The PCP also instructed James’ mother on how to use a sleep diary to maintain a 1-month log of his sleep patterns and habits. The PCP consulted the integrated behavioral health clinician (IBHC; a clinical social worker embedded in the primary care clinic) and made a warm handoff for the IBHC to further assess James’ maladaptive behaviors and interactions.
●
* The patient’s name has been changed to protect his identity.
James is one of more than 6 million children, ages 3 to 17 years, in the United States who live with ADHD.1,2 ADHD is the most common neurodevelopmental disorder among children, and it affects multiple cognitive and behavioral domains throughout the lifespan.3 Children with ADHD often initially present in primary care settings; thus, PCPs are well positioned to diagnose the disorder and provide longitudinal treatment. This Behavioral Health Consult reviews clinical assessment and practice guidelines, as well as treatment recommendations applicable across different areas of influence—individual, family, community, and systems—for PCPs and IBHCs to use in managing ADHD in children.
ADHD features can vary by age and sex
ADHD is a persistent pattern of inattention or hyperactivity and impulsivity interfering with functioning or development in childhood and functioning later in adulthood. ADHD symptoms manifest prior to age 12 years and must occur in 2 or more settings.4 Symptoms should not be better explained by another psychiatric disorder or occur exclusively during the course of another disorder (TABLE 1).4

The rate of heritability is high, with significant incidence among first-degree relatives.4 Children with ADHD show executive functioning deficits in 1 or more cognitive domains (eg, visuospatial, memory, inhibitions, decision making, and reward regulation).4,5 The prevalence of ADHD nationally is approximately 9.8% (2.2%, ages 3-5 years; 10%, ages 6-11 years; 13.2%, ages 12-17 years) in children and adolescents; worldwide prevalence is 7.2%.1,6 It persists among 2.6% to 6.8% of adults worldwide.7
Research has shown that boys ages 6 to 11 years are significantly more likely than girls to exhibit attention-getting, externalizing behaviors or conduct problems (eg, hyperactivity, impulsivity, disruption, aggression).1,6 On the other hand, girls ages 12 to 17 years tend to display internalized (eg, depressed mood, anxiety, low self-esteem) or inattentive behaviors, which clinicians and educators may assess as less severe and warranting fewer supportive measures.1
The prevalence of ADHD and its associated factors, which evolve through maturation, underscore the importance of persistent, patient-centered, and collaborative PCP and IBHC clinical management.
Continue to: Begin with a screening tool, move to a clinical interview
Begin with a screening tool, move to a clinical interview
When caregivers express concerns about their child’s behavior, focus, mood, learning, and socialization, consider initiating a multimodal evaluation for ADHD.5,8 Embarking on an ADHD assessment can require extended or multiple visits to arrive at the diagnosis, followed by still more visits to confirm a course of care and adjust medications. The integrative care approach described in the patient case and elaborated on later in this article can help facilitate assessment and treatment of ADHD.9
Signs of ADHD may be observed at initial screening using a tool such as the Ages & Stages Questionnaire (https://agesandstages.com/products-pricing/asq3/) to reveal indications of norm deviations or delays commensurate with ADHD.10 However, to substantiate the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, Text Revision criteria for an accurate diagnosis,4 the American Academy of Pediatrics (AAP) clinical practice guidelines require a thorough clinical interview, administration of a standardized assessment tool, and review of objective reports in conjunction with a physical examination and psychosocial evaluation.6 Standardized measures of psychological, neurocognitive, and academic achievement reported by caregivers and collateral contacts (eg, teachers, counselors, coaches, care providers) are needed to maximize data objectivity and symptom accuracy across settings (TABLE 210-17). Additionally, periodic reassessment is recommended to validate changes in diagnostic subtype and treatment plans due to the chronic and dynamic nature of ADHD.

Consider comorbidities and alternate diagnoses
The diagnostic possibility of ADHD should also prompt consideration of other childhood disorders due to the high potential for comorbidities.4,6 In a 2016 study, approximately 64% of
Various medical disorders may manifest with similar signs or symptoms to ADHD, such as thyroid disorders, seizure disorders, adverse drug effects, anemia, genetic anomalies, and others.6,19
If there are behavioral concerns or developmental delays associated with tall stature for age or pubertal or testicular development anomalies, consult a geneticist and a developmental pediatrician for targeted testing and neurodevelopmental assessment, respectively. For example, ADHD is a common comorbidity among boys who also have XYY syndrome (Jacobs syndrome). However, due to the variability of symptoms and severity, XYY syndrome often goes undiagnosed, leaving a host of compounding pervasive and developmental problems untreated. Overall, more than two-thirds of patients with ADHD and a co-occurring condition are either inaccurately diagnosed or not referred for additional assessment and adjunct treatment.21
Continue to: Risks that arise over time
Risks that arise over time. As ADHD persists, adolescents are at greater risk for psychiatric comorbidities, suicidality, and functional impairments (eg, risky behaviors, occupational problems, truancy, delinquency, and poor self-esteem).4,8 Adolescents with internalized behaviors are more likely to experience comorbid depressive disorders with increased risk for self-harm.4,5,8 As adolescents age and their sense of autonomy increases, there is a tendency among those who have received a diagnosis of ADHD to minimize symptoms and decrease the frequency of routine clinic visits along with medication use and treatment compliance.3 Additionally, abuse, misuse, and misappropriation of stimulants among teens and young adults are commonplace.
Wide-scope, multidisciplinary evaluation and close clinical management reduce the potential for imprecise diagnoses, particularly at critical developmental junctures. AAP suggests that PCPs can treat mild and moderate cases of ADHD, but if the treating clinician does not have adequate training, experience, time, or clinical support to manage this condition, early referral is warranted.6
A guide to pharmacotherapy
Approximately 77% of children ages 2 to 17 years with a diagnosis of ADHD receive any form of treatment.2 Treatment for ADHD can include behavioral therapy and medication.2 AAP clinical practice guidelines caution against prescribing medications for children younger than 6 years, relying instead on caregiver-, teacher-, or clinician-administered behavioral strategies and parental training in behavioral modification. For children and adolescents between ages 6 and 18 years, first-line treatment includes pharmacotherapy balanced with behavioral therapy, academic modifications, and educational supports (eg, 504 Plan, individualized education plan [IEP]).6
Psychostimulants are preferred. These agents (eg, methylphenidate, amphetamine) remain the most efficacious class of medications to reduce hyperactivity and inattentiveness and to improve function. While long-acting psychostimulants are associated with better medication adherence and adverse-effect tolerance than are short-acting forms, the latter offer more flexibility in dosing. Start by titrating any stimulant to the lowest effective dose; reassess monthly until potential rebound effects stabilize.
Due to potential adverse effects of this class of medication, screen for any family history or personal risk for structural or electrical cardiac anomalies before starting pharmacotherapy. If any such risks exist, arrange for further cardiac evaluation before initiating medication.6 Adverse effects of stimulants include reduced appetite, gastrointestinal symptoms, headaches, anxiousness, parasomnia, tachycardia, and hypertension.
Continue to: Once medication is stabilized...
Once medication is stabilized, monitor treatment 2 to 3 times per year thereafter; watch for longer-term adverse effects such as weight loss, decreased growth rate, and psychiatric comorbidities including the Food and Drug Administration (FDA)’s black box warning of increased risk for suicidality.5,6,22
Other options. The optimal duration of psychostimulant use remains debatable, as existing evidence does not support its long-term use (10 years) over other interventions, such as nonstimulants and nonmedicinal therapies.22 Although backed by less evidence, additional medications indicated for the treatment of ADHD include: (1) atomoxetine, a selective norepinephrine reuptake inhibitor, and (2) the selective alpha-2 adrenergic agonists, extended-release guanfacine and extended-release clonidine (third-line agent).22
Adverse effects of these FDA-approved medications are similar to those observed in stimulant medications. Evaluation of cardiac risks is recommended before starting nonstimulant medications. The alpha-2 adrenergic agonists may also be used as adjunct therapies to stimulants. Before stopping an alpha-2 adrenergic agonist, taper the dosage slowly to avoid the risk for rebound hypertension.6,23 Given the wide variety of medication options and variability of effects, it may be necessary to try different medications as children grow and their symptoms and capacity to manage them change. Additional guidance on FDA-approved medications is available at www.ADHDMedicationGuide.com.
How multilevel care coordination can work
As with other chronic or developmental conditions, the treatment of ADHD requires an interdisciplinary perspective. Continuous, comprehensive case management can help patients overcome obstacles to wellness by balancing the resolution of problems with the development of resilience. Well-documented collaboration of subspecialists, educators, and other stakeholders engaged in ADHD care at multiple levels (individual, family, community, and health care system) increases the likelihood of meaningful, sustainable gains. Using a patient-centered medical home framework, IBHCs or other allied health professionals embedded in, or co-located with, primary care settings can be key to accessing evidence-based treatments that include: psycho-education and mindfulness-based stress reduction training for caregivers24,25; occupational,26 cognitive behavioral,27 or family therapies28,29; neuro-feedback; computer-based attention training; group- or community-based interventions; and academic and social supports.5,8
Treatment approaches that capitalize on children’s neurologic and psychological plasticity and fortify self-efficacy with developmentally appropriate tools empower them to surmount ADHD symptoms over time.23 Facilitating children’s resilience within a developmental framework and health system’s capacities with socio-culturally relevant approaches, consultation, and research can optimize outcomes and mitigate pervasiveness into adulthood. While the patient is at the center of treatment, it is important to consider the family, school, and communities in which the child lives, learns, and plays. PCPs and IBHCs together can consider a “try and track” method to follow progress, changes, and outcomes over time. With this method, the physician can employ approaches that focus on the patient, caregiver, or the caregiver–child interaction (TABLE 3).

Continue to: Assess patients' needs and the resources available
Assess patients’ needs and the resources available throughout the system of care beyond the primary care setting. Stay abreast of hospital policies, health care insurance coverage, and community- and school-based health programs, and any gaps in adequate and equitable assessment and treatment. For example, while clinical recommendations include psychiatric care, health insurance availability or limits in coverage may dissuade caregivers from seeking help or limit initial or long-term access to resources for help.30 Integrating or advocating for clinic support resources or staffing to assist patients in navigating and mitigating challenges may lessen the management burden and increase the likelihood and longevity of favorable health outcomes.
Steps to ensuring health care equity
Among children of historically marginalized and racial and ethnic minority groups or those of populations affected by health disparities, ADHD symptoms and needs are often masked by structural biases that lead to inequitable care and outcomes, as well as treatment misprioritization or delays.31 In particular, evidence has shown that recognition and diagnostic specificity of ADHD and comorbidities, not prevalence, vary more widely among minority than among nonminority populations,32 contributing to the 23% of children with ADHD who receive no treatment at all.2
Understand caregiver concerns. This diagnosis discrepancy is correlated with symptom rating sensitivities (eg, reliability, perception, accuracy) among informants and how caregivers observe, perceive, appreciate, understand, and report behaviors. This discrepancy is also related to cultural belief differences, physician–patient communication variants, and a litany of other socioeconomic determinants.2,4,31 Caregivers from some cultural, ethnic, or socioeconomic backgrounds may be doubtful of psychiatric assessment, diagnoses, treatment, or medication, and that can impact how children are engaged in clinical and educational settings from the outset.31 In the case we described, James’ mother was initially hesitant to explore psychotropic medications and was concerned about stigmatization within the school system. She also seemed to avoid psychiatric treatment for her own depressive symptoms due to cultural and religious beliefs.
Health care provider concerns. Some PCPs may hesitate to explore medications due to limited knowledge and skill in dosing and titrating based on a child’s age, stage, and symptoms, and a perceived lack of competence in managing ADHD. This, too, can indirectly perpetuate existing health disparities. Furthermore, ADHD symptoms may be deemed a secondary or tertiary concern if other complex or urgent medical or undifferentiated developmental problems manifest.
Compounding matters is the limited dissemination of empiric research articles (including randomized controlled trials with representative samples) and limited education on the effectiveness and safety of psychopharmacologic interventions across the lifespan and different cultural and ethnic groups.4 Consequently, patients who struggle with unmanaged ADHD symptoms are more likely to have chronic mental health disorders, maladaptive behaviors, and other co-occurring conditions contributing to the complexity of individual needs, health care burdens, or justice system involvement; this is particularly true for those of racial and ethnic minorities.33
Continue to: Impact of the COVID-19 pandemic
Impact of the COVID-19 pandemic. Patients—particularly those in minority or health disparity populations—who under normal circumstances might have been hesitant to seek help may have felt even more reluctant to do so during the COVID-19 pandemic. We have not yet learned the degree to which limited availability of preventive health care services, decreased routine visits, and fluctuating insurance coverage has impacted the diagnosis, management, or severity of childhood disorders during the past 2 years. Reports of national findings indicate that prolonged periods out of school and reduced daily structure were associated with increased disruptions in mood, sleep, and appetite, particularly among children with pre-existing pathologies. Evidence suggests that school-aged children experienced more anxiety, regressive behaviors, and parasomnias than they did before the pandemic, while adolescents experienced more isolation and depressive symptoms.34,35
However, there remains a paucity of large-scale or representative studies that use an intersectional lens to examine the influence of COVID-19 on children with ADHD. Therefore, PCPs and IBHCs should refocus attention on possibly undiagnosed, stagnated, or regressed ADHD cases, as well as the adults who care for them. (See “5 ways to overcome Tx barriers and promote health equity.”)
SIDEBAR
5 ways to overcome Tx barriers and promote health equitya
1. Inquire about cultural or ethnic beliefs and behaviors and socioeconomic barriers.
2. Establish trust or assuage mistrust by exploring and dispelling misinformation.
3. Offer accessible, feasible, and sustainable evidence-based interventions.
4. Encourage autonomy and selfdetermination throughout the health care process.
5. Connect caregivers and children with clinical, community, and school-based resources and coordinators.
a These recommendations are based on the authors’ combined clinical experience.
THE CASE
During a follow-up visit 1 month later, the PCP confirmed the clinical impression of ADHD combined presentation with a clinical interview and review of the Strengths and Difficulties Questionnaire completed by James’ mother and the Vanderbilt Assessment Scales completed by James’ mother and teacher. The sleep diary indicated potential problems and apneas worthy of consults for pulmonary function testing, a sleep study, and otolaryngology examination. The PCP informed James’ mother on sleep hygiene strategies and ADHD medication options. She indicated that she wanted to pursue the referrals and behavioral modifications before starting any medication trial.
The PCP referred James to a developmental pediatrician for in-depth assessment of his overall development, learning, and functioning. The developmental pediatrician ultimately confirmed the diagnosis of ADHD, as well as motor and speech delays warranting physical, occupational, and speech therapies. The developmental pediatrician also referred James for targeted genetic testing because she suspected a genetic disorder (eg, XYY syndrome).
The PCP reconnected James and his mother to the IBHC to facilitate subspecialty and school-based care coordination and to provide in-office and home-based interventions. The IBHC assessed James’ emotional dysregulation and impulsivity as adversely impacting his interpersonal relationships and planned to address these issues with behavioral and parent–child interaction therapies and skills training during the course of 6 to 12 visits. James’ mother was encouraged to engage his teacher on his academic performance and to initiate a 504 Plan or IEP for in-school accommodations and support. The IBHC aided in tracking his assessments, referrals, follow-ups, access barriers, and treatment goals.
After 6 months, James had made only modest progress, and his mother requested that he begin a trial of medication. Based on his weight, symptoms, behavior patterns, and sleep habits, the PCP prescribed extended-release dexmethylphenidate 10 mg each morning, then extended-release clonidine 0.1 mg nightly. With team-based clinical management of pharmacologic, behavioral, physical, speech, and occupational therapies, James’ behavior and sleep improved, and the signs of a vocal tic diminished.
By the next school year, James demonstrated a marked improvement in impulse control, attention, and academic functioning. He followed up with the PCP at least quarterly for reassessment of his symptoms, growth, and experience of adverse effects, and to titrate medications accordingly. James and his mother continued to work closely with the IBHC monthly to engage interventions and to monitor his progress at home and school.
CORRESPONDENCE
Sundania J. W. Wonnum, PhD, LCSW, National Institute on Minority Health and Health Disparities, 6707 Democracy Boulevard, Suite 800, Bethesda, MD 20892; [email protected]
THE CASE
James B* is a 7-year-old Black child who presented to his primary care physician (PCP) for a well-child visit. During preventive health screening, James’ mother expressed concerns about his behavior, characterizing him as immature, aggressive, destructive, and occasionally self-loathing. She described him as physically uncoordinated, struggling to keep up with his peers in sports, and tiring after 20 minutes of activity. James slept 10 hours nightly but was often restless and snored intermittently. As a second grader, his academic achievement was not progressing, and he had become increasingly inattentive at home and at school. James’ mother offered several examples of his fighting with his siblings, noncompliance with morning routines, and avoidance of learning activities. Additionally, his mother expressed concern that James, as a Black child, might eventually be unfairly labeled as a problem child by his teachers or held back a grade level in school.
Although James did not have a family history of developmental delays or learning disorders, he had not met any milestones on time for gross or fine motor, language, cognitive, and social-emotional skills. James had a history of chronic otitis media, for which pressure equalizer tubes were inserted at age 2 years. He had not had any major physical injuries, psychological trauma, recent life transitions, or adverse childhood events. When asked, James’ mother acknowledged symptoms of maternal depression but alluded to faith-based reasons for not seeking treatment for herself.
James’ physical examination was unremarkable. His height, weight, and vitals were all within normal limits. However, he had some difficulty with verbal articulation and expression and showed signs of a possible vocal tic. Based on James’ presentation, his PCP suspected attention-deficit/hyperactivity disorder (ADHD), as well as neurodevelopmental delays.
The PCP gave James’ mother the Strengths and Difficulties Questionnaire to complete and the Vanderbilt Assessment Scales for her and James’ teacher to fill out independently and return to the clinic. The PCP also instructed James’ mother on how to use a sleep diary to maintain a 1-month log of his sleep patterns and habits. The PCP consulted the integrated behavioral health clinician (IBHC; a clinical social worker embedded in the primary care clinic) and made a warm handoff for the IBHC to further assess James’ maladaptive behaviors and interactions.
●
* The patient’s name has been changed to protect his identity.
James is one of more than 6 million children, ages 3 to 17 years, in the United States who live with ADHD.1,2 ADHD is the most common neurodevelopmental disorder among children, and it affects multiple cognitive and behavioral domains throughout the lifespan.3 Children with ADHD often initially present in primary care settings; thus, PCPs are well positioned to diagnose the disorder and provide longitudinal treatment. This Behavioral Health Consult reviews clinical assessment and practice guidelines, as well as treatment recommendations applicable across different areas of influence—individual, family, community, and systems—for PCPs and IBHCs to use in managing ADHD in children.
ADHD features can vary by age and sex
ADHD is a persistent pattern of inattention or hyperactivity and impulsivity interfering with functioning or development in childhood and functioning later in adulthood. ADHD symptoms manifest prior to age 12 years and must occur in 2 or more settings.4 Symptoms should not be better explained by another psychiatric disorder or occur exclusively during the course of another disorder (TABLE 1).4

The rate of heritability is high, with significant incidence among first-degree relatives.4 Children with ADHD show executive functioning deficits in 1 or more cognitive domains (eg, visuospatial, memory, inhibitions, decision making, and reward regulation).4,5 The prevalence of ADHD nationally is approximately 9.8% (2.2%, ages 3-5 years; 10%, ages 6-11 years; 13.2%, ages 12-17 years) in children and adolescents; worldwide prevalence is 7.2%.1,6 It persists among 2.6% to 6.8% of adults worldwide.7
Research has shown that boys ages 6 to 11 years are significantly more likely than girls to exhibit attention-getting, externalizing behaviors or conduct problems (eg, hyperactivity, impulsivity, disruption, aggression).1,6 On the other hand, girls ages 12 to 17 years tend to display internalized (eg, depressed mood, anxiety, low self-esteem) or inattentive behaviors, which clinicians and educators may assess as less severe and warranting fewer supportive measures.1
The prevalence of ADHD and its associated factors, which evolve through maturation, underscore the importance of persistent, patient-centered, and collaborative PCP and IBHC clinical management.
Continue to: Begin with a screening tool, move to a clinical interview
Begin with a screening tool, move to a clinical interview
When caregivers express concerns about their child’s behavior, focus, mood, learning, and socialization, consider initiating a multimodal evaluation for ADHD.5,8 Embarking on an ADHD assessment can require extended or multiple visits to arrive at the diagnosis, followed by still more visits to confirm a course of care and adjust medications. The integrative care approach described in the patient case and elaborated on later in this article can help facilitate assessment and treatment of ADHD.9
Signs of ADHD may be observed at initial screening using a tool such as the Ages & Stages Questionnaire (https://agesandstages.com/products-pricing/asq3/) to reveal indications of norm deviations or delays commensurate with ADHD.10 However, to substantiate the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, Text Revision criteria for an accurate diagnosis,4 the American Academy of Pediatrics (AAP) clinical practice guidelines require a thorough clinical interview, administration of a standardized assessment tool, and review of objective reports in conjunction with a physical examination and psychosocial evaluation.6 Standardized measures of psychological, neurocognitive, and academic achievement reported by caregivers and collateral contacts (eg, teachers, counselors, coaches, care providers) are needed to maximize data objectivity and symptom accuracy across settings (TABLE 210-17). Additionally, periodic reassessment is recommended to validate changes in diagnostic subtype and treatment plans due to the chronic and dynamic nature of ADHD.

Consider comorbidities and alternate diagnoses
The diagnostic possibility of ADHD should also prompt consideration of other childhood disorders due to the high potential for comorbidities.4,6 In a 2016 study, approximately 64% of
Various medical disorders may manifest with similar signs or symptoms to ADHD, such as thyroid disorders, seizure disorders, adverse drug effects, anemia, genetic anomalies, and others.6,19
If there are behavioral concerns or developmental delays associated with tall stature for age or pubertal or testicular development anomalies, consult a geneticist and a developmental pediatrician for targeted testing and neurodevelopmental assessment, respectively. For example, ADHD is a common comorbidity among boys who also have XYY syndrome (Jacobs syndrome). However, due to the variability of symptoms and severity, XYY syndrome often goes undiagnosed, leaving a host of compounding pervasive and developmental problems untreated. Overall, more than two-thirds of patients with ADHD and a co-occurring condition are either inaccurately diagnosed or not referred for additional assessment and adjunct treatment.21
Continue to: Risks that arise over time
Risks that arise over time. As ADHD persists, adolescents are at greater risk for psychiatric comorbidities, suicidality, and functional impairments (eg, risky behaviors, occupational problems, truancy, delinquency, and poor self-esteem).4,8 Adolescents with internalized behaviors are more likely to experience comorbid depressive disorders with increased risk for self-harm.4,5,8 As adolescents age and their sense of autonomy increases, there is a tendency among those who have received a diagnosis of ADHD to minimize symptoms and decrease the frequency of routine clinic visits along with medication use and treatment compliance.3 Additionally, abuse, misuse, and misappropriation of stimulants among teens and young adults are commonplace.
Wide-scope, multidisciplinary evaluation and close clinical management reduce the potential for imprecise diagnoses, particularly at critical developmental junctures. AAP suggests that PCPs can treat mild and moderate cases of ADHD, but if the treating clinician does not have adequate training, experience, time, or clinical support to manage this condition, early referral is warranted.6
A guide to pharmacotherapy
Approximately 77% of children ages 2 to 17 years with a diagnosis of ADHD receive any form of treatment.2 Treatment for ADHD can include behavioral therapy and medication.2 AAP clinical practice guidelines caution against prescribing medications for children younger than 6 years, relying instead on caregiver-, teacher-, or clinician-administered behavioral strategies and parental training in behavioral modification. For children and adolescents between ages 6 and 18 years, first-line treatment includes pharmacotherapy balanced with behavioral therapy, academic modifications, and educational supports (eg, 504 Plan, individualized education plan [IEP]).6
Psychostimulants are preferred. These agents (eg, methylphenidate, amphetamine) remain the most efficacious class of medications to reduce hyperactivity and inattentiveness and to improve function. While long-acting psychostimulants are associated with better medication adherence and adverse-effect tolerance than are short-acting forms, the latter offer more flexibility in dosing. Start by titrating any stimulant to the lowest effective dose; reassess monthly until potential rebound effects stabilize.
Due to potential adverse effects of this class of medication, screen for any family history or personal risk for structural or electrical cardiac anomalies before starting pharmacotherapy. If any such risks exist, arrange for further cardiac evaluation before initiating medication.6 Adverse effects of stimulants include reduced appetite, gastrointestinal symptoms, headaches, anxiousness, parasomnia, tachycardia, and hypertension.
Continue to: Once medication is stabilized...
Once medication is stabilized, monitor treatment 2 to 3 times per year thereafter; watch for longer-term adverse effects such as weight loss, decreased growth rate, and psychiatric comorbidities including the Food and Drug Administration (FDA)’s black box warning of increased risk for suicidality.5,6,22
Other options. The optimal duration of psychostimulant use remains debatable, as existing evidence does not support its long-term use (10 years) over other interventions, such as nonstimulants and nonmedicinal therapies.22 Although backed by less evidence, additional medications indicated for the treatment of ADHD include: (1) atomoxetine, a selective norepinephrine reuptake inhibitor, and (2) the selective alpha-2 adrenergic agonists, extended-release guanfacine and extended-release clonidine (third-line agent).22
Adverse effects of these FDA-approved medications are similar to those observed in stimulant medications. Evaluation of cardiac risks is recommended before starting nonstimulant medications. The alpha-2 adrenergic agonists may also be used as adjunct therapies to stimulants. Before stopping an alpha-2 adrenergic agonist, taper the dosage slowly to avoid the risk for rebound hypertension.6,23 Given the wide variety of medication options and variability of effects, it may be necessary to try different medications as children grow and their symptoms and capacity to manage them change. Additional guidance on FDA-approved medications is available at www.ADHDMedicationGuide.com.
How multilevel care coordination can work
As with other chronic or developmental conditions, the treatment of ADHD requires an interdisciplinary perspective. Continuous, comprehensive case management can help patients overcome obstacles to wellness by balancing the resolution of problems with the development of resilience. Well-documented collaboration of subspecialists, educators, and other stakeholders engaged in ADHD care at multiple levels (individual, family, community, and health care system) increases the likelihood of meaningful, sustainable gains. Using a patient-centered medical home framework, IBHCs or other allied health professionals embedded in, or co-located with, primary care settings can be key to accessing evidence-based treatments that include: psycho-education and mindfulness-based stress reduction training for caregivers24,25; occupational,26 cognitive behavioral,27 or family therapies28,29; neuro-feedback; computer-based attention training; group- or community-based interventions; and academic and social supports.5,8
Treatment approaches that capitalize on children’s neurologic and psychological plasticity and fortify self-efficacy with developmentally appropriate tools empower them to surmount ADHD symptoms over time.23 Facilitating children’s resilience within a developmental framework and health system’s capacities with socio-culturally relevant approaches, consultation, and research can optimize outcomes and mitigate pervasiveness into adulthood. While the patient is at the center of treatment, it is important to consider the family, school, and communities in which the child lives, learns, and plays. PCPs and IBHCs together can consider a “try and track” method to follow progress, changes, and outcomes over time. With this method, the physician can employ approaches that focus on the patient, caregiver, or the caregiver–child interaction (TABLE 3).

Continue to: Assess patients' needs and the resources available
Assess patients’ needs and the resources available throughout the system of care beyond the primary care setting. Stay abreast of hospital policies, health care insurance coverage, and community- and school-based health programs, and any gaps in adequate and equitable assessment and treatment. For example, while clinical recommendations include psychiatric care, health insurance availability or limits in coverage may dissuade caregivers from seeking help or limit initial or long-term access to resources for help.30 Integrating or advocating for clinic support resources or staffing to assist patients in navigating and mitigating challenges may lessen the management burden and increase the likelihood and longevity of favorable health outcomes.
Steps to ensuring health care equity
Among children of historically marginalized and racial and ethnic minority groups or those of populations affected by health disparities, ADHD symptoms and needs are often masked by structural biases that lead to inequitable care and outcomes, as well as treatment misprioritization or delays.31 In particular, evidence has shown that recognition and diagnostic specificity of ADHD and comorbidities, not prevalence, vary more widely among minority than among nonminority populations,32 contributing to the 23% of children with ADHD who receive no treatment at all.2
Understand caregiver concerns. This diagnosis discrepancy is correlated with symptom rating sensitivities (eg, reliability, perception, accuracy) among informants and how caregivers observe, perceive, appreciate, understand, and report behaviors. This discrepancy is also related to cultural belief differences, physician–patient communication variants, and a litany of other socioeconomic determinants.2,4,31 Caregivers from some cultural, ethnic, or socioeconomic backgrounds may be doubtful of psychiatric assessment, diagnoses, treatment, or medication, and that can impact how children are engaged in clinical and educational settings from the outset.31 In the case we described, James’ mother was initially hesitant to explore psychotropic medications and was concerned about stigmatization within the school system. She also seemed to avoid psychiatric treatment for her own depressive symptoms due to cultural and religious beliefs.
Health care provider concerns. Some PCPs may hesitate to explore medications due to limited knowledge and skill in dosing and titrating based on a child’s age, stage, and symptoms, and a perceived lack of competence in managing ADHD. This, too, can indirectly perpetuate existing health disparities. Furthermore, ADHD symptoms may be deemed a secondary or tertiary concern if other complex or urgent medical or undifferentiated developmental problems manifest.
Compounding matters is the limited dissemination of empiric research articles (including randomized controlled trials with representative samples) and limited education on the effectiveness and safety of psychopharmacologic interventions across the lifespan and different cultural and ethnic groups.4 Consequently, patients who struggle with unmanaged ADHD symptoms are more likely to have chronic mental health disorders, maladaptive behaviors, and other co-occurring conditions contributing to the complexity of individual needs, health care burdens, or justice system involvement; this is particularly true for those of racial and ethnic minorities.33
Continue to: Impact of the COVID-19 pandemic
Impact of the COVID-19 pandemic. Patients—particularly those in minority or health disparity populations—who under normal circumstances might have been hesitant to seek help may have felt even more reluctant to do so during the COVID-19 pandemic. We have not yet learned the degree to which limited availability of preventive health care services, decreased routine visits, and fluctuating insurance coverage has impacted the diagnosis, management, or severity of childhood disorders during the past 2 years. Reports of national findings indicate that prolonged periods out of school and reduced daily structure were associated with increased disruptions in mood, sleep, and appetite, particularly among children with pre-existing pathologies. Evidence suggests that school-aged children experienced more anxiety, regressive behaviors, and parasomnias than they did before the pandemic, while adolescents experienced more isolation and depressive symptoms.34,35
However, there remains a paucity of large-scale or representative studies that use an intersectional lens to examine the influence of COVID-19 on children with ADHD. Therefore, PCPs and IBHCs should refocus attention on possibly undiagnosed, stagnated, or regressed ADHD cases, as well as the adults who care for them. (See “5 ways to overcome Tx barriers and promote health equity.”)
SIDEBAR
5 ways to overcome Tx barriers and promote health equitya
1. Inquire about cultural or ethnic beliefs and behaviors and socioeconomic barriers.
2. Establish trust or assuage mistrust by exploring and dispelling misinformation.
3. Offer accessible, feasible, and sustainable evidence-based interventions.
4. Encourage autonomy and selfdetermination throughout the health care process.
5. Connect caregivers and children with clinical, community, and school-based resources and coordinators.
a These recommendations are based on the authors’ combined clinical experience.
THE CASE
During a follow-up visit 1 month later, the PCP confirmed the clinical impression of ADHD combined presentation with a clinical interview and review of the Strengths and Difficulties Questionnaire completed by James’ mother and the Vanderbilt Assessment Scales completed by James’ mother and teacher. The sleep diary indicated potential problems and apneas worthy of consults for pulmonary function testing, a sleep study, and otolaryngology examination. The PCP informed James’ mother on sleep hygiene strategies and ADHD medication options. She indicated that she wanted to pursue the referrals and behavioral modifications before starting any medication trial.
The PCP referred James to a developmental pediatrician for in-depth assessment of his overall development, learning, and functioning. The developmental pediatrician ultimately confirmed the diagnosis of ADHD, as well as motor and speech delays warranting physical, occupational, and speech therapies. The developmental pediatrician also referred James for targeted genetic testing because she suspected a genetic disorder (eg, XYY syndrome).
The PCP reconnected James and his mother to the IBHC to facilitate subspecialty and school-based care coordination and to provide in-office and home-based interventions. The IBHC assessed James’ emotional dysregulation and impulsivity as adversely impacting his interpersonal relationships and planned to address these issues with behavioral and parent–child interaction therapies and skills training during the course of 6 to 12 visits. James’ mother was encouraged to engage his teacher on his academic performance and to initiate a 504 Plan or IEP for in-school accommodations and support. The IBHC aided in tracking his assessments, referrals, follow-ups, access barriers, and treatment goals.
After 6 months, James had made only modest progress, and his mother requested that he begin a trial of medication. Based on his weight, symptoms, behavior patterns, and sleep habits, the PCP prescribed extended-release dexmethylphenidate 10 mg each morning, then extended-release clonidine 0.1 mg nightly. With team-based clinical management of pharmacologic, behavioral, physical, speech, and occupational therapies, James’ behavior and sleep improved, and the signs of a vocal tic diminished.
By the next school year, James demonstrated a marked improvement in impulse control, attention, and academic functioning. He followed up with the PCP at least quarterly for reassessment of his symptoms, growth, and experience of adverse effects, and to titrate medications accordingly. James and his mother continued to work closely with the IBHC monthly to engage interventions and to monitor his progress at home and school.
CORRESPONDENCE
Sundania J. W. Wonnum, PhD, LCSW, National Institute on Minority Health and Health Disparities, 6707 Democracy Boulevard, Suite 800, Bethesda, MD 20892; [email protected]
1. Bitsko RH, Claussen AH, Lichstein J, et al. Mental health surveillance among children—United States, 2013-2019. MMWR Suppl. 2022;71:1-42. doi: 10.15585/mmwr.su7102a1
2. Danielson ML, Holbrook JR, Blumberg SJ, et al. State-level estimates of the prevalence of parent-reported ADHD diagnosis and treatment among U.S. children and adolescents, 2016 to 2019. J Atten Disord. 2022;26:1685-1697. doi: 10.1177/10870547221099961
3. Faraone SV, Banaschewski T, Coghill D, et al. The World Federation of ADHD International Consensus Statement: 208 evidence-based conclusions about the disorder. Neurosci Biobehav Rev. 2021;128:789-818. doi: 10.1016/j.neubiorev.2021.01.022
4. American Psychiatric Association
5. Brahmbhatt K, Hilty DM, Mina H, et al. Diagnosis and treatment of attention deficit hyperactivity disorder during adolescence in the primary care setting: a concise review. J Adolesc Health. 2016;59:135-143. doi: 10.1016/j.jadohealth.2016.03.025
6. Wolraich ML, Hagan JF, Allan C, et al. AAP Subcommittee on Children and Adolescents with Attention-Deficit/Hyperactivity Disorder. Clinical Practice Guideline for the Diagnosis, Evaluation, and Treatment of Attention-Deficit/Hyperactivity Disorder in Children and Adolescents. Pediatrics. 2019;144:e20192528. doi: 10.1542/peds.2019-2528
7. Song P, Zha M, Yang Q, et al. The prevalence of adult attention-deficit hyperactivity disorder: a global systematic review and meta-analysis. J Glob Health. 2021;11:04009. doi: 10.7189/jogh.11.04009
8. Chang JG, Cimino FM, Gossa W. ADHD in children: common questions and answers. Am Fam Physician. 2020;102:592-602.
9. Asarnow JR, Rozenman M, Wiblin J, et al. Integrated medical-behavioral care compared with usual primary care for child and adolescent behavioral health: a meta-analysis. JAMA Pediatr. 2015;169:929-937. doi: 10.1001/jamapediatrics.2015.1141
10. Squires J, Bricker D. Ages & Stages Questionnaires®. 3rd ed (ASQ®-3). Paul H. Brookes Publishing Co., Inc; 2009.
11. DuPaul GJ, Barkley RA. Situational variability of attention problems: psychometric properties of the Revised Home and School Situations Questionnaires. J Clin Child Psychol. 1992;21:178-188. doi.org/10.1207/s15374424jccp2102_10
12. Merenda PF. BASC: behavior assessment system for children. Meas Eval Counsel Develop. 1996;28:229-232.
13. Conners CK. Conners, 3rd ed manual. Multi-Health Systems. 2008.
14. Achenbach TM. The Child Behavior Checklist and related instruments. In: Maruish ME, ed. The Use of Psychological Testing for Treatment Planning and Outcomes Assessment. Lawrence Erlbaum Associates Publishers; 1999:429-466.
15. Goodman R. The extended version of the Strengths and Difficulties Questionnaire as a guide to child psychiatric caseness and consequent burden. J Child Psychol Psychiatry. 1999;40:791-799.
16. Wolraich ML, Lambert W, Doffing MA, et al. Psychometric properties of the Vanderbilt ADHD Diagnostic Parent Rating Scale in a referred population. J Pediatr Psychol. 2003;28:559-567. doi: 10.1093/jpepsy/jsg046
17. Sparrow SS, Cicchetti DV. The Vineland Adaptive Behavior Scales. In: Newmark CS, ed. Major Psychological Assessment Instruments. Vol 2. Allyn & Bacon; 2003:199-231.
18. Danielson ML, Bitsko RH, Ghandour RM, et al. Prevalence of parent-reported ADHD diagnosis and associated treatment among U.S. children and adolescents, 2016. J Clin Child Adolesc Psychol. 2018;47:199-212. doi: 10.1080/15374416.2017.1417860
19. Ghriwati NA, Langberg JM, Gardner W, et al. Impact of mental health comorbidities on the community-based pediatric treatment and outcomes of children with attention deficit hyperactivity disorder. J Dev Behav Ped. 2017;38:20-28. doi: 10.1097/DBP.0000000000000359
20. Niclasen J, Obel C, Homøe P, et al. Associations between otitis media and child behavioural and learning difficulties: results from a Danish Cohort. Int J Ped Otorhinolaryngol. 2016;84:12-20. doi: 10.1016/j.ijporl.2016.02.017
21. Ross JL Roeltgen DP Kushner H, et al. Behavioral and social phenotypes in boys with 47,XYY syndrome or 47,XXY Klinefelter syndrome. doi: 10.1542/peds.2011-0719
22. Mechler K, Banaschewski T, Hohmann S, et al. Evidence-based pharmacological treatment options for ADHD in children and adolescents. Pharmacol Ther. 2022;230:107940. doi: 10.1016/j.pharmthera.2021.107940
23. Mishra J, Merzenich MM, Sagar R. Accessible online neuroplasticity-targeted training for children with ADHD. Child Adolesc Psychiatry Ment Health. 2013;7:38. doi: 10.1186/1753-2000-7-38
24. Neece CL. Mindfulness-based stress reduction for parents of young children with developmental delays: implications for parental mental health and child behavior problems. J Applied Res Intellect Disabil. 2014;27:174-186. doi: 10.1111/jar.12064
25. Petcharat M, Liehr P. Mindfulness training for parents of children with special needs: guidance for nurses in mental health practice. J Child Adolesc Psychiatr Nursing. 2017;30:35-46. doi: 10.1111/jcap.12169
26. Hahn-Markowitz J, Burger I, Manor I, et al. Efficacy of cognitive-functional (Cog-Fun) occupational therapy intervention among children with ADHD: an RCT. J Atten Disord. 2020;24:655-666. doi: 10.1177/1087054716666955
27. Young Z, Moghaddam N, Tickle A. The efficacy of cognitive behavioral therapy for adults with ADHD: a systematic review and meta-analysis of randomized controlled trials. J Atten Disord. 2020;24:875-888.
28. Carr AW, Bean RA, Nelson KF. Childhood attention-deficit hyperactivity disorder: family therapy from an attachment based perspective. Child Youth Serv Rev. 2020;119:105666.
29. Robin AL. Family therapy for adolescents with ADHD. Child Adolesc Psychiatr Clin N Am. 2014;23:747-756. doi: 10.1016/j.chc.2014.06.001
30. Cattoi B, Alpern I, Katz JS, et al. The adverse health outcomes, economic burden, and public health implications of unmanaged attention deficit hyperactivity disorder (ADHD): a call to action resulting from CHADD summit, Washington, DC, October 17, 2019. J Atten Disord. 2022;26:807-808. doi: 10.1177/10870547211036754
31. Hinojosa MS, Hinojosa R, Nguyen J. Shared decision making and treatment for minority children with ADHD. J Transcult Nurs. 2020;31:135-143. doi: 10.1177/1043659619853021
32. Slobodin O, Masalha R. Challenges in ADHD care for ethnic minority children: a review of the current literature. Transcult Psychiatry. 2020;57:468-483. doi: 10.1177/1363461520902885
33. Retz W, Ginsberg Y, Turner D, et al. Attention-deficit/hyperactivity disorder (ADHD), antisociality and delinquent behavior over the lifespan. Neurosci Biobehav Rev. 2021;120:236-248. doi: 10.1016/j.neubiorev.2020.11.025
34. Del Sol Calderon P, Izquierdo A, Garcia Moreno M. Effects of the pandemic on the mental health of children and adolescents. Review and current scientific evidence of the SARS-COV2 pandemic. Eur Psychiatry. 2021;64:S223-S224. doi: 10.1192/j.eurpsy.2021.597
35. Insa I, Alda JA. Attention deficit hyperactivity disorder (ADHD) & COVID-19: attention deficit hyperactivity disorder: consequences of the 1st wave. Eur Psychiatry. 2021;64:S660. doi: 10.1192/j.eurpsy.2021.1752
1. Bitsko RH, Claussen AH, Lichstein J, et al. Mental health surveillance among children—United States, 2013-2019. MMWR Suppl. 2022;71:1-42. doi: 10.15585/mmwr.su7102a1
2. Danielson ML, Holbrook JR, Blumberg SJ, et al. State-level estimates of the prevalence of parent-reported ADHD diagnosis and treatment among U.S. children and adolescents, 2016 to 2019. J Atten Disord. 2022;26:1685-1697. doi: 10.1177/10870547221099961
3. Faraone SV, Banaschewski T, Coghill D, et al. The World Federation of ADHD International Consensus Statement: 208 evidence-based conclusions about the disorder. Neurosci Biobehav Rev. 2021;128:789-818. doi: 10.1016/j.neubiorev.2021.01.022
4. American Psychiatric Association
5. Brahmbhatt K, Hilty DM, Mina H, et al. Diagnosis and treatment of attention deficit hyperactivity disorder during adolescence in the primary care setting: a concise review. J Adolesc Health. 2016;59:135-143. doi: 10.1016/j.jadohealth.2016.03.025
6. Wolraich ML, Hagan JF, Allan C, et al. AAP Subcommittee on Children and Adolescents with Attention-Deficit/Hyperactivity Disorder. Clinical Practice Guideline for the Diagnosis, Evaluation, and Treatment of Attention-Deficit/Hyperactivity Disorder in Children and Adolescents. Pediatrics. 2019;144:e20192528. doi: 10.1542/peds.2019-2528
7. Song P, Zha M, Yang Q, et al. The prevalence of adult attention-deficit hyperactivity disorder: a global systematic review and meta-analysis. J Glob Health. 2021;11:04009. doi: 10.7189/jogh.11.04009
8. Chang JG, Cimino FM, Gossa W. ADHD in children: common questions and answers. Am Fam Physician. 2020;102:592-602.
9. Asarnow JR, Rozenman M, Wiblin J, et al. Integrated medical-behavioral care compared with usual primary care for child and adolescent behavioral health: a meta-analysis. JAMA Pediatr. 2015;169:929-937. doi: 10.1001/jamapediatrics.2015.1141
10. Squires J, Bricker D. Ages & Stages Questionnaires®. 3rd ed (ASQ®-3). Paul H. Brookes Publishing Co., Inc; 2009.
11. DuPaul GJ, Barkley RA. Situational variability of attention problems: psychometric properties of the Revised Home and School Situations Questionnaires. J Clin Child Psychol. 1992;21:178-188. doi.org/10.1207/s15374424jccp2102_10
12. Merenda PF. BASC: behavior assessment system for children. Meas Eval Counsel Develop. 1996;28:229-232.
13. Conners CK. Conners, 3rd ed manual. Multi-Health Systems. 2008.
14. Achenbach TM. The Child Behavior Checklist and related instruments. In: Maruish ME, ed. The Use of Psychological Testing for Treatment Planning and Outcomes Assessment. Lawrence Erlbaum Associates Publishers; 1999:429-466.
15. Goodman R. The extended version of the Strengths and Difficulties Questionnaire as a guide to child psychiatric caseness and consequent burden. J Child Psychol Psychiatry. 1999;40:791-799.
16. Wolraich ML, Lambert W, Doffing MA, et al. Psychometric properties of the Vanderbilt ADHD Diagnostic Parent Rating Scale in a referred population. J Pediatr Psychol. 2003;28:559-567. doi: 10.1093/jpepsy/jsg046
17. Sparrow SS, Cicchetti DV. The Vineland Adaptive Behavior Scales. In: Newmark CS, ed. Major Psychological Assessment Instruments. Vol 2. Allyn & Bacon; 2003:199-231.
18. Danielson ML, Bitsko RH, Ghandour RM, et al. Prevalence of parent-reported ADHD diagnosis and associated treatment among U.S. children and adolescents, 2016. J Clin Child Adolesc Psychol. 2018;47:199-212. doi: 10.1080/15374416.2017.1417860
19. Ghriwati NA, Langberg JM, Gardner W, et al. Impact of mental health comorbidities on the community-based pediatric treatment and outcomes of children with attention deficit hyperactivity disorder. J Dev Behav Ped. 2017;38:20-28. doi: 10.1097/DBP.0000000000000359
20. Niclasen J, Obel C, Homøe P, et al. Associations between otitis media and child behavioural and learning difficulties: results from a Danish Cohort. Int J Ped Otorhinolaryngol. 2016;84:12-20. doi: 10.1016/j.ijporl.2016.02.017
21. Ross JL Roeltgen DP Kushner H, et al. Behavioral and social phenotypes in boys with 47,XYY syndrome or 47,XXY Klinefelter syndrome. doi: 10.1542/peds.2011-0719
22. Mechler K, Banaschewski T, Hohmann S, et al. Evidence-based pharmacological treatment options for ADHD in children and adolescents. Pharmacol Ther. 2022;230:107940. doi: 10.1016/j.pharmthera.2021.107940
23. Mishra J, Merzenich MM, Sagar R. Accessible online neuroplasticity-targeted training for children with ADHD. Child Adolesc Psychiatry Ment Health. 2013;7:38. doi: 10.1186/1753-2000-7-38
24. Neece CL. Mindfulness-based stress reduction for parents of young children with developmental delays: implications for parental mental health and child behavior problems. J Applied Res Intellect Disabil. 2014;27:174-186. doi: 10.1111/jar.12064
25. Petcharat M, Liehr P. Mindfulness training for parents of children with special needs: guidance for nurses in mental health practice. J Child Adolesc Psychiatr Nursing. 2017;30:35-46. doi: 10.1111/jcap.12169
26. Hahn-Markowitz J, Burger I, Manor I, et al. Efficacy of cognitive-functional (Cog-Fun) occupational therapy intervention among children with ADHD: an RCT. J Atten Disord. 2020;24:655-666. doi: 10.1177/1087054716666955
27. Young Z, Moghaddam N, Tickle A. The efficacy of cognitive behavioral therapy for adults with ADHD: a systematic review and meta-analysis of randomized controlled trials. J Atten Disord. 2020;24:875-888.
28. Carr AW, Bean RA, Nelson KF. Childhood attention-deficit hyperactivity disorder: family therapy from an attachment based perspective. Child Youth Serv Rev. 2020;119:105666.
29. Robin AL. Family therapy for adolescents with ADHD. Child Adolesc Psychiatr Clin N Am. 2014;23:747-756. doi: 10.1016/j.chc.2014.06.001
30. Cattoi B, Alpern I, Katz JS, et al. The adverse health outcomes, economic burden, and public health implications of unmanaged attention deficit hyperactivity disorder (ADHD): a call to action resulting from CHADD summit, Washington, DC, October 17, 2019. J Atten Disord. 2022;26:807-808. doi: 10.1177/10870547211036754
31. Hinojosa MS, Hinojosa R, Nguyen J. Shared decision making and treatment for minority children with ADHD. J Transcult Nurs. 2020;31:135-143. doi: 10.1177/1043659619853021
32. Slobodin O, Masalha R. Challenges in ADHD care for ethnic minority children: a review of the current literature. Transcult Psychiatry. 2020;57:468-483. doi: 10.1177/1363461520902885
33. Retz W, Ginsberg Y, Turner D, et al. Attention-deficit/hyperactivity disorder (ADHD), antisociality and delinquent behavior over the lifespan. Neurosci Biobehav Rev. 2021;120:236-248. doi: 10.1016/j.neubiorev.2020.11.025
34. Del Sol Calderon P, Izquierdo A, Garcia Moreno M. Effects of the pandemic on the mental health of children and adolescents. Review and current scientific evidence of the SARS-COV2 pandemic. Eur Psychiatry. 2021;64:S223-S224. doi: 10.1192/j.eurpsy.2021.597
35. Insa I, Alda JA. Attention deficit hyperactivity disorder (ADHD) & COVID-19: attention deficit hyperactivity disorder: consequences of the 1st wave. Eur Psychiatry. 2021;64:S660. doi: 10.1192/j.eurpsy.2021.1752
Does physical exercise reduce dementia-associated agitation?
Evidence summary
Mixed results on exercise’s effect on neuropsychiatric symptoms
A 2020 systematic review and meta-analysis of 18 RCTs investigated the effect of home-based physical activity on several markers of behavioral and psychological symptoms of dementia (BPSD). These symptoms were measured using the caregiver-completed neuropsychiatric inventory (NPI), which includes agitation. There was substantial heterogeneity between trials; however, 4 RCTs (472 patients) were included in a meta-analysis of the NPI. These RCTs were nonblinded, given the nature of the intervention.1
Interventions to enhance physical activity ranged from 12 weeks to 2 years in duration, with 2 to 8 contacts from the study team per week. The type of physical activity varied and included cardiorespiratory endurance, balance training, resistance training, and activities of daily living training.1
Exercise was associated with significantly fewer symptoms on the NPI, although the effect size was small (standard mean difference [SMD] = –0.37; 95% CI, –0.57 to –0.17). Heterogeneity in the interventions and assessments were limitations to this meta-analysis.1
A 2015 systematic review and meta-analysis of 18 RCTs compared the effect of exercise interventions against a control group for the treatment of BPSD, utilizing 10 behavioral and 2 neurovegetative components of the NPI (each scored from 0 to 5) in patients with dementia. Studies were included if they used ≥ 1 exercise intervention compared to a control or usual care group without additional exercise recommendations. Thirteen studies had a multicomponent training intervention (≥ 2 exercise types grouped together in the same training session), 2 used tai chi, 4 used walking, and 1 used dance and movement therapy. These RCTs were conducted in a variety of settings, including community-dwelling and long-term care facilities (n = 6427 patients).2
Exercise did not reduce global BPSD (N = 4441 patients), with a weighted mean difference (WMD) of −3.9 (95% CI, −9.0 to 1.2; P = .13).
A 2017 hospital-based RCT evaluated the effects of a short-term exercise program on neuropsychiatric signs and symptoms in patients with dementia in 3 specialized dementia care wards (N = 85). Patients had a diagnosis of dementia, minimum length of stay of 1 week, no delirium, and the ability to perform the Timed Up and Go Test. The intervention group included a 2-week exercise program of four 20-minute exercise sessions per day on 3 days per week, involving strengthening or endurance exercises, in addition to treatment as usual. The control group included a 2-week period of social-stimulation programs consisting of table games for 120 minutes per week, in addition to treatment as usual.3
Of 85 patients randomized, 15 (18%) were lost to follow-up (14 of whom were discharged early from the hospital). Among the 70 patients included in the final analysis, the mean age was 80 years; 47% were female and 53% male; and the mean Mini-Mental Status Examination score was 18.3 (≤ 23 indicates dementia). In both groups, most patients had moderate dementia, moderate neuropsychiatric signs and symptoms, and a low level of psychotic symptoms. Patients in the intervention group had a higher adherence rate compared with those in the control group.3
Continue to: The primary outcome...
The primary outcome was neuropsychiatric signs and symptoms as measured by the Alzheimer’s Disease Cooperative Study–Clinical Global Impression of Change (ADCS-CGIC). Compared to the control group, the intervention group experienced greater improvement on the ADCS-CGIC dimensions of emotional agitation (
A 2016 factorial cluster RCT of 16 nursing homes (with at least 60% of the population having dementia) compared the use of person-centered care vs person-centered care plus at least 1 randomly assigned additional intervention (eg, antipsychotic medication use review, social interaction interventions, and exercise over a period of 9 months) (n = 277, with 193 analyzed per protocol). Exercise was implemented at 1 hour per week or at an increase of 20% above baseline and compared with a control group with no change in exercise.4
Exercise significantly improved neuropsychiatric symptoms. The baseline NPI score of 14.54 improved by –3.59 (95% CI, –7.08 to –0.09; P < .05). However, none of the study interventions significantly improved the agitation-specific scores. The primary limitation of this study was that antipsychotic prescribing was at the discretion of the provider and not according to a protocol. In addition, the authors noted that the trial was inadequately powered to correct for testing 3 primary outcomes.4
Editor’s takeaway
Dementia and dementia with agitation are challenging conditions to treat. Disappointingly, physical exercise had inconsistent and generally minimal effect on agitation in dementia. Nevertheless, exercise had other positive effects. So, considering the benefits that exercise does provide, its low cost, and its limited adverse effects, exercise remains a small tool to address a big problem.
1. de Almeida SIL, Gomes da Silva M, de Dias Marques ASP. Home-based physical activity programs for people with dementia: systematic review and meta-analysis. Gerontologist. 2020;60:600-608. doi: 10.1093/geront/gnz176
2. de Souto Barreto P, Demougeot L, Pillard F, et al. Exercise training for managing behavioral and psychological symptoms in people with dementia: a systematic review and meta-analysis. Ageing Res Rev. 2015;24(pt B):274-285. doi: 10.1016/j.arr.2015.09.001
3. Fleiner T, Dauth H, Gersie M, et al. Structured physical exercise improves neuropsychiatric symptoms in acute dementia care: a hospital-based RCT. Alzheimers Res Ther. 2017;9:68. doi: 10.1186/s13195-017-0289-z
4. Ballard C, Orrell M, YongZhong S, et al. Impact of antipsychotic review and nonpharmacological intervention on antipsychotic use, neuropsychiatric symptoms, and mortality in people with dementia living in nursing homes: a factorial cluster-randomized controlled trial by the Well-Being and Health for People With Dementia (WHELD) Program. Am J Psychiatry. 2016;173:252-262. doi: 10.1176/appi.ajp.2015.15010130
Evidence summary
Mixed results on exercise’s effect on neuropsychiatric symptoms
A 2020 systematic review and meta-analysis of 18 RCTs investigated the effect of home-based physical activity on several markers of behavioral and psychological symptoms of dementia (BPSD). These symptoms were measured using the caregiver-completed neuropsychiatric inventory (NPI), which includes agitation. There was substantial heterogeneity between trials; however, 4 RCTs (472 patients) were included in a meta-analysis of the NPI. These RCTs were nonblinded, given the nature of the intervention.1
Interventions to enhance physical activity ranged from 12 weeks to 2 years in duration, with 2 to 8 contacts from the study team per week. The type of physical activity varied and included cardiorespiratory endurance, balance training, resistance training, and activities of daily living training.1
Exercise was associated with significantly fewer symptoms on the NPI, although the effect size was small (standard mean difference [SMD] = –0.37; 95% CI, –0.57 to –0.17). Heterogeneity in the interventions and assessments were limitations to this meta-analysis.1
A 2015 systematic review and meta-analysis of 18 RCTs compared the effect of exercise interventions against a control group for the treatment of BPSD, utilizing 10 behavioral and 2 neurovegetative components of the NPI (each scored from 0 to 5) in patients with dementia. Studies were included if they used ≥ 1 exercise intervention compared to a control or usual care group without additional exercise recommendations. Thirteen studies had a multicomponent training intervention (≥ 2 exercise types grouped together in the same training session), 2 used tai chi, 4 used walking, and 1 used dance and movement therapy. These RCTs were conducted in a variety of settings, including community-dwelling and long-term care facilities (n = 6427 patients).2
Exercise did not reduce global BPSD (N = 4441 patients), with a weighted mean difference (WMD) of −3.9 (95% CI, −9.0 to 1.2; P = .13).
A 2017 hospital-based RCT evaluated the effects of a short-term exercise program on neuropsychiatric signs and symptoms in patients with dementia in 3 specialized dementia care wards (N = 85). Patients had a diagnosis of dementia, minimum length of stay of 1 week, no delirium, and the ability to perform the Timed Up and Go Test. The intervention group included a 2-week exercise program of four 20-minute exercise sessions per day on 3 days per week, involving strengthening or endurance exercises, in addition to treatment as usual. The control group included a 2-week period of social-stimulation programs consisting of table games for 120 minutes per week, in addition to treatment as usual.3
Of 85 patients randomized, 15 (18%) were lost to follow-up (14 of whom were discharged early from the hospital). Among the 70 patients included in the final analysis, the mean age was 80 years; 47% were female and 53% male; and the mean Mini-Mental Status Examination score was 18.3 (≤ 23 indicates dementia). In both groups, most patients had moderate dementia, moderate neuropsychiatric signs and symptoms, and a low level of psychotic symptoms. Patients in the intervention group had a higher adherence rate compared with those in the control group.3
Continue to: The primary outcome...
The primary outcome was neuropsychiatric signs and symptoms as measured by the Alzheimer’s Disease Cooperative Study–Clinical Global Impression of Change (ADCS-CGIC). Compared to the control group, the intervention group experienced greater improvement on the ADCS-CGIC dimensions of emotional agitation (
A 2016 factorial cluster RCT of 16 nursing homes (with at least 60% of the population having dementia) compared the use of person-centered care vs person-centered care plus at least 1 randomly assigned additional intervention (eg, antipsychotic medication use review, social interaction interventions, and exercise over a period of 9 months) (n = 277, with 193 analyzed per protocol). Exercise was implemented at 1 hour per week or at an increase of 20% above baseline and compared with a control group with no change in exercise.4
Exercise significantly improved neuropsychiatric symptoms. The baseline NPI score of 14.54 improved by –3.59 (95% CI, –7.08 to –0.09; P < .05). However, none of the study interventions significantly improved the agitation-specific scores. The primary limitation of this study was that antipsychotic prescribing was at the discretion of the provider and not according to a protocol. In addition, the authors noted that the trial was inadequately powered to correct for testing 3 primary outcomes.4
Editor’s takeaway
Dementia and dementia with agitation are challenging conditions to treat. Disappointingly, physical exercise had inconsistent and generally minimal effect on agitation in dementia. Nevertheless, exercise had other positive effects. So, considering the benefits that exercise does provide, its low cost, and its limited adverse effects, exercise remains a small tool to address a big problem.
Evidence summary
Mixed results on exercise’s effect on neuropsychiatric symptoms
A 2020 systematic review and meta-analysis of 18 RCTs investigated the effect of home-based physical activity on several markers of behavioral and psychological symptoms of dementia (BPSD). These symptoms were measured using the caregiver-completed neuropsychiatric inventory (NPI), which includes agitation. There was substantial heterogeneity between trials; however, 4 RCTs (472 patients) were included in a meta-analysis of the NPI. These RCTs were nonblinded, given the nature of the intervention.1
Interventions to enhance physical activity ranged from 12 weeks to 2 years in duration, with 2 to 8 contacts from the study team per week. The type of physical activity varied and included cardiorespiratory endurance, balance training, resistance training, and activities of daily living training.1
Exercise was associated with significantly fewer symptoms on the NPI, although the effect size was small (standard mean difference [SMD] = –0.37; 95% CI, –0.57 to –0.17). Heterogeneity in the interventions and assessments were limitations to this meta-analysis.1
A 2015 systematic review and meta-analysis of 18 RCTs compared the effect of exercise interventions against a control group for the treatment of BPSD, utilizing 10 behavioral and 2 neurovegetative components of the NPI (each scored from 0 to 5) in patients with dementia. Studies were included if they used ≥ 1 exercise intervention compared to a control or usual care group without additional exercise recommendations. Thirteen studies had a multicomponent training intervention (≥ 2 exercise types grouped together in the same training session), 2 used tai chi, 4 used walking, and 1 used dance and movement therapy. These RCTs were conducted in a variety of settings, including community-dwelling and long-term care facilities (n = 6427 patients).2
Exercise did not reduce global BPSD (N = 4441 patients), with a weighted mean difference (WMD) of −3.9 (95% CI, −9.0 to 1.2; P = .13).
A 2017 hospital-based RCT evaluated the effects of a short-term exercise program on neuropsychiatric signs and symptoms in patients with dementia in 3 specialized dementia care wards (N = 85). Patients had a diagnosis of dementia, minimum length of stay of 1 week, no delirium, and the ability to perform the Timed Up and Go Test. The intervention group included a 2-week exercise program of four 20-minute exercise sessions per day on 3 days per week, involving strengthening or endurance exercises, in addition to treatment as usual. The control group included a 2-week period of social-stimulation programs consisting of table games for 120 minutes per week, in addition to treatment as usual.3
Of 85 patients randomized, 15 (18%) were lost to follow-up (14 of whom were discharged early from the hospital). Among the 70 patients included in the final analysis, the mean age was 80 years; 47% were female and 53% male; and the mean Mini-Mental Status Examination score was 18.3 (≤ 23 indicates dementia). In both groups, most patients had moderate dementia, moderate neuropsychiatric signs and symptoms, and a low level of psychotic symptoms. Patients in the intervention group had a higher adherence rate compared with those in the control group.3
Continue to: The primary outcome...
The primary outcome was neuropsychiatric signs and symptoms as measured by the Alzheimer’s Disease Cooperative Study–Clinical Global Impression of Change (ADCS-CGIC). Compared to the control group, the intervention group experienced greater improvement on the ADCS-CGIC dimensions of emotional agitation (
A 2016 factorial cluster RCT of 16 nursing homes (with at least 60% of the population having dementia) compared the use of person-centered care vs person-centered care plus at least 1 randomly assigned additional intervention (eg, antipsychotic medication use review, social interaction interventions, and exercise over a period of 9 months) (n = 277, with 193 analyzed per protocol). Exercise was implemented at 1 hour per week or at an increase of 20% above baseline and compared with a control group with no change in exercise.4
Exercise significantly improved neuropsychiatric symptoms. The baseline NPI score of 14.54 improved by –3.59 (95% CI, –7.08 to –0.09; P < .05). However, none of the study interventions significantly improved the agitation-specific scores. The primary limitation of this study was that antipsychotic prescribing was at the discretion of the provider and not according to a protocol. In addition, the authors noted that the trial was inadequately powered to correct for testing 3 primary outcomes.4
Editor’s takeaway
Dementia and dementia with agitation are challenging conditions to treat. Disappointingly, physical exercise had inconsistent and generally minimal effect on agitation in dementia. Nevertheless, exercise had other positive effects. So, considering the benefits that exercise does provide, its low cost, and its limited adverse effects, exercise remains a small tool to address a big problem.
1. de Almeida SIL, Gomes da Silva M, de Dias Marques ASP. Home-based physical activity programs for people with dementia: systematic review and meta-analysis. Gerontologist. 2020;60:600-608. doi: 10.1093/geront/gnz176
2. de Souto Barreto P, Demougeot L, Pillard F, et al. Exercise training for managing behavioral and psychological symptoms in people with dementia: a systematic review and meta-analysis. Ageing Res Rev. 2015;24(pt B):274-285. doi: 10.1016/j.arr.2015.09.001
3. Fleiner T, Dauth H, Gersie M, et al. Structured physical exercise improves neuropsychiatric symptoms in acute dementia care: a hospital-based RCT. Alzheimers Res Ther. 2017;9:68. doi: 10.1186/s13195-017-0289-z
4. Ballard C, Orrell M, YongZhong S, et al. Impact of antipsychotic review and nonpharmacological intervention on antipsychotic use, neuropsychiatric symptoms, and mortality in people with dementia living in nursing homes: a factorial cluster-randomized controlled trial by the Well-Being and Health for People With Dementia (WHELD) Program. Am J Psychiatry. 2016;173:252-262. doi: 10.1176/appi.ajp.2015.15010130
1. de Almeida SIL, Gomes da Silva M, de Dias Marques ASP. Home-based physical activity programs for people with dementia: systematic review and meta-analysis. Gerontologist. 2020;60:600-608. doi: 10.1093/geront/gnz176
2. de Souto Barreto P, Demougeot L, Pillard F, et al. Exercise training for managing behavioral and psychological symptoms in people with dementia: a systematic review and meta-analysis. Ageing Res Rev. 2015;24(pt B):274-285. doi: 10.1016/j.arr.2015.09.001
3. Fleiner T, Dauth H, Gersie M, et al. Structured physical exercise improves neuropsychiatric symptoms in acute dementia care: a hospital-based RCT. Alzheimers Res Ther. 2017;9:68. doi: 10.1186/s13195-017-0289-z
4. Ballard C, Orrell M, YongZhong S, et al. Impact of antipsychotic review and nonpharmacological intervention on antipsychotic use, neuropsychiatric symptoms, and mortality in people with dementia living in nursing homes: a factorial cluster-randomized controlled trial by the Well-Being and Health for People With Dementia (WHELD) Program. Am J Psychiatry. 2016;173:252-262. doi: 10.1176/appi.ajp.2015.15010130
EVIDENCE-BASED ANSWER:
Not consistently. Physical exer- cise demonstrates inconsistent benefit for neuropsychiatric symptoms, including agitation, in patients with dementia (strength of recommendation: B, inconsistent meta-analyses, 2 small randomized controlled trials [RCTs]). The care setting and the modality, frequency, and duration of exercise varied across trials; the impact of these factors is not known.
Pediatricians, specialists largely agree on ASD diagnoses
General pediatricians and a multidisciplinary team of specialists agreed most of the time on which children should be diagnosed with autism spectrum disorder (ASD), data from a new study suggest.
But when it came to ruling out ASD, the agreement rate was much lower.
The study by Melanie Penner, MSc, MD, with the Autism Research Centre at Bloorview Research Institute, Toronto, and colleagues found that 89% of the time when a physician determined a child had ASD, the multidisciplinary team agreed. But when a pediatrician thought a child did not have ASD, the multidisciplinary team agreed only 60% of the time. The study was published in JAMA Network Open.
Multidisciplinary team model can’t keep up with demand
The findings are important as many guidelines recommend multidisciplinary teams (MDTs) for all ASD diagnostic assessment. However, the resources for this model can’t meet the demand of children needing a diagnosis and can lead to long waits for ASD therapies.
In Canada, the researchers note, the average wait time from referral to receipt of ASD diagnosis has been reported as 7 months and “has likely lengthened since the COVID-19 pandemic.”
Jennifer Gerdts, PhD, an attending psychologist at the Seattle Children’s Autism Center, said in an interview that the wait there for diagnosis in children older than 4 is “multiple years,” a length of time that’s common across the United States. Meanwhile, in many states families can’t access services without a diagnosis.
Expanding capacity with diagnoses by general pediatricians may improve access, but the diagnostic accuracy is critical.
Dr. Gerdts, who was not part of the study, said this research is “hugely important in the work that is under way to build community capacity for diagnostic evaluation.”
She said this study shows that not all diagnoses need the resources of a multiple-disciplinary team and that “pediatricians can do it, too, and they can do it pretty accurately.” Dr. Gerdts evaluates children for autism and helps train pediatricians to make diagnoses.
Pediatricians, specialist team completed blinded assessments
The 17 pediatricians in the study and the specialist team independently completed blinded assessment and each recorded a decision on whether the child had ASD. The prospective diagnostic study was conducted in a specialist assessment center in Toronto and in general pediatrician practices in Ontario from June 2016 to March 2020.
Children were younger than 5.5 years, did not have an ASD diagnosis and were referred because there was a development concern. The pediatricians referred 106 children (75% boys; average age, 3.5 years). More than half (57%) of the participating children were from minority racial and ethnic groups.
The children were randomly assigned to two groups: One included children who had their MDT visits before their pediatrician assessment and the other group included those who had their MDT visits after their pediatrician assessment.
The MDT diagnosed more than two-thirds of the children (68%) with ASD.
Sensitivity and specificity of the pediatrician assessments, compared with that of the specialist team, were 0.75 (95% confidence interval, 0.67-0.83) and 0.79 (95% CI, 0.62-0.91), respectively.
A look at pediatricians’ accuracy
Pediatricians reported the decisions they would have made had the child not been in the study.
- In 69% of the true-positive cases, pediatricians would have given an ASD diagnosis.
- In 44% of true-negative cases, they would have told the family the child did not have autism; in 30% of those case, they would give alternative diagnoses (most commonly ADHD and language delay).
- The pediatrician would have diagnosed ASD in only one of the seven false-positive cases and would refer those patients to a subspecialist 71% of the time.
- In false-negative cases, the pediatrician would incorrectly tell the family the child does not have autism 44% of the time.
Regarding the false-negative cases, the authors wrote, “more caution is needed for pediatricians when definitively ruling out ASD, which might result in diagnostic delays.”
Confidence is key
Physician confidence was also correlated with accuracy.
The authors wrote: “Among true-positive cases (MDT and pediatrician agree the child has ASD), the pediatrician was certain or very certain 80% of the time (43 cases) and the MDT was certain or very certain 96% of the time (52 cases). As such, if pediatricians conferred ASD diagnoses when feeling certain or very certain, they would make 46 correct diagnoses and 2 incorrect diagnoses.”
The high accuracy of diagnosis when physicians are confident suggests “listening to that sense of certainty is important,” Dr. Gerdts said. Conversely, these numbers show when a physician is uncertain about diagnosing ASD, they should listen to that instinct, too, and refer.
The results of the study support having general pediatricians diagnose and move forward with their patients when the signs of ASD are more definitive, saving the less-certain cases for the more resource-intensive teams to diagnose. Many states are moving toward that “tiered” system, Dr. Gerdts said.
“For many, and in fact most children, general pediatricians are pretty accurate when making an autism diagnosis,” she said.
“Let’s get [general pediatricians] confident in recognizing when this is outside their skill and ability level,” she said. “If you’re not sure, it is better to refer them on than to misdiagnose them.”
The important missing piece she said is how to support them “so they don’t feel pressure to make that call,” Dr. Gerdts said.
This project was funded by a grant from the Bloorview Research Institute, a grant from the Canadian Institutes of Health Research and a grant from the Canadian Institutes of Health. Three coauthors consult for and receive grants from several pharmaceutical companies and other organizations. Dr. Gerdts declared no relevant financial relationships.
General pediatricians and a multidisciplinary team of specialists agreed most of the time on which children should be diagnosed with autism spectrum disorder (ASD), data from a new study suggest.
But when it came to ruling out ASD, the agreement rate was much lower.
The study by Melanie Penner, MSc, MD, with the Autism Research Centre at Bloorview Research Institute, Toronto, and colleagues found that 89% of the time when a physician determined a child had ASD, the multidisciplinary team agreed. But when a pediatrician thought a child did not have ASD, the multidisciplinary team agreed only 60% of the time. The study was published in JAMA Network Open.
Multidisciplinary team model can’t keep up with demand
The findings are important as many guidelines recommend multidisciplinary teams (MDTs) for all ASD diagnostic assessment. However, the resources for this model can’t meet the demand of children needing a diagnosis and can lead to long waits for ASD therapies.
In Canada, the researchers note, the average wait time from referral to receipt of ASD diagnosis has been reported as 7 months and “has likely lengthened since the COVID-19 pandemic.”
Jennifer Gerdts, PhD, an attending psychologist at the Seattle Children’s Autism Center, said in an interview that the wait there for diagnosis in children older than 4 is “multiple years,” a length of time that’s common across the United States. Meanwhile, in many states families can’t access services without a diagnosis.
Expanding capacity with diagnoses by general pediatricians may improve access, but the diagnostic accuracy is critical.
Dr. Gerdts, who was not part of the study, said this research is “hugely important in the work that is under way to build community capacity for diagnostic evaluation.”
She said this study shows that not all diagnoses need the resources of a multiple-disciplinary team and that “pediatricians can do it, too, and they can do it pretty accurately.” Dr. Gerdts evaluates children for autism and helps train pediatricians to make diagnoses.
Pediatricians, specialist team completed blinded assessments
The 17 pediatricians in the study and the specialist team independently completed blinded assessment and each recorded a decision on whether the child had ASD. The prospective diagnostic study was conducted in a specialist assessment center in Toronto and in general pediatrician practices in Ontario from June 2016 to March 2020.
Children were younger than 5.5 years, did not have an ASD diagnosis and were referred because there was a development concern. The pediatricians referred 106 children (75% boys; average age, 3.5 years). More than half (57%) of the participating children were from minority racial and ethnic groups.
The children were randomly assigned to two groups: One included children who had their MDT visits before their pediatrician assessment and the other group included those who had their MDT visits after their pediatrician assessment.
The MDT diagnosed more than two-thirds of the children (68%) with ASD.
Sensitivity and specificity of the pediatrician assessments, compared with that of the specialist team, were 0.75 (95% confidence interval, 0.67-0.83) and 0.79 (95% CI, 0.62-0.91), respectively.
A look at pediatricians’ accuracy
Pediatricians reported the decisions they would have made had the child not been in the study.
- In 69% of the true-positive cases, pediatricians would have given an ASD diagnosis.
- In 44% of true-negative cases, they would have told the family the child did not have autism; in 30% of those case, they would give alternative diagnoses (most commonly ADHD and language delay).
- The pediatrician would have diagnosed ASD in only one of the seven false-positive cases and would refer those patients to a subspecialist 71% of the time.
- In false-negative cases, the pediatrician would incorrectly tell the family the child does not have autism 44% of the time.
Regarding the false-negative cases, the authors wrote, “more caution is needed for pediatricians when definitively ruling out ASD, which might result in diagnostic delays.”
Confidence is key
Physician confidence was also correlated with accuracy.
The authors wrote: “Among true-positive cases (MDT and pediatrician agree the child has ASD), the pediatrician was certain or very certain 80% of the time (43 cases) and the MDT was certain or very certain 96% of the time (52 cases). As such, if pediatricians conferred ASD diagnoses when feeling certain or very certain, they would make 46 correct diagnoses and 2 incorrect diagnoses.”
The high accuracy of diagnosis when physicians are confident suggests “listening to that sense of certainty is important,” Dr. Gerdts said. Conversely, these numbers show when a physician is uncertain about diagnosing ASD, they should listen to that instinct, too, and refer.
The results of the study support having general pediatricians diagnose and move forward with their patients when the signs of ASD are more definitive, saving the less-certain cases for the more resource-intensive teams to diagnose. Many states are moving toward that “tiered” system, Dr. Gerdts said.
“For many, and in fact most children, general pediatricians are pretty accurate when making an autism diagnosis,” she said.
“Let’s get [general pediatricians] confident in recognizing when this is outside their skill and ability level,” she said. “If you’re not sure, it is better to refer them on than to misdiagnose them.”
The important missing piece she said is how to support them “so they don’t feel pressure to make that call,” Dr. Gerdts said.
This project was funded by a grant from the Bloorview Research Institute, a grant from the Canadian Institutes of Health Research and a grant from the Canadian Institutes of Health. Three coauthors consult for and receive grants from several pharmaceutical companies and other organizations. Dr. Gerdts declared no relevant financial relationships.
General pediatricians and a multidisciplinary team of specialists agreed most of the time on which children should be diagnosed with autism spectrum disorder (ASD), data from a new study suggest.
But when it came to ruling out ASD, the agreement rate was much lower.
The study by Melanie Penner, MSc, MD, with the Autism Research Centre at Bloorview Research Institute, Toronto, and colleagues found that 89% of the time when a physician determined a child had ASD, the multidisciplinary team agreed. But when a pediatrician thought a child did not have ASD, the multidisciplinary team agreed only 60% of the time. The study was published in JAMA Network Open.
Multidisciplinary team model can’t keep up with demand
The findings are important as many guidelines recommend multidisciplinary teams (MDTs) for all ASD diagnostic assessment. However, the resources for this model can’t meet the demand of children needing a diagnosis and can lead to long waits for ASD therapies.
In Canada, the researchers note, the average wait time from referral to receipt of ASD diagnosis has been reported as 7 months and “has likely lengthened since the COVID-19 pandemic.”
Jennifer Gerdts, PhD, an attending psychologist at the Seattle Children’s Autism Center, said in an interview that the wait there for diagnosis in children older than 4 is “multiple years,” a length of time that’s common across the United States. Meanwhile, in many states families can’t access services without a diagnosis.
Expanding capacity with diagnoses by general pediatricians may improve access, but the diagnostic accuracy is critical.
Dr. Gerdts, who was not part of the study, said this research is “hugely important in the work that is under way to build community capacity for diagnostic evaluation.”
She said this study shows that not all diagnoses need the resources of a multiple-disciplinary team and that “pediatricians can do it, too, and they can do it pretty accurately.” Dr. Gerdts evaluates children for autism and helps train pediatricians to make diagnoses.
Pediatricians, specialist team completed blinded assessments
The 17 pediatricians in the study and the specialist team independently completed blinded assessment and each recorded a decision on whether the child had ASD. The prospective diagnostic study was conducted in a specialist assessment center in Toronto and in general pediatrician practices in Ontario from June 2016 to March 2020.
Children were younger than 5.5 years, did not have an ASD diagnosis and were referred because there was a development concern. The pediatricians referred 106 children (75% boys; average age, 3.5 years). More than half (57%) of the participating children were from minority racial and ethnic groups.
The children were randomly assigned to two groups: One included children who had their MDT visits before their pediatrician assessment and the other group included those who had their MDT visits after their pediatrician assessment.
The MDT diagnosed more than two-thirds of the children (68%) with ASD.
Sensitivity and specificity of the pediatrician assessments, compared with that of the specialist team, were 0.75 (95% confidence interval, 0.67-0.83) and 0.79 (95% CI, 0.62-0.91), respectively.
A look at pediatricians’ accuracy
Pediatricians reported the decisions they would have made had the child not been in the study.
- In 69% of the true-positive cases, pediatricians would have given an ASD diagnosis.
- In 44% of true-negative cases, they would have told the family the child did not have autism; in 30% of those case, they would give alternative diagnoses (most commonly ADHD and language delay).
- The pediatrician would have diagnosed ASD in only one of the seven false-positive cases and would refer those patients to a subspecialist 71% of the time.
- In false-negative cases, the pediatrician would incorrectly tell the family the child does not have autism 44% of the time.
Regarding the false-negative cases, the authors wrote, “more caution is needed for pediatricians when definitively ruling out ASD, which might result in diagnostic delays.”
Confidence is key
Physician confidence was also correlated with accuracy.
The authors wrote: “Among true-positive cases (MDT and pediatrician agree the child has ASD), the pediatrician was certain or very certain 80% of the time (43 cases) and the MDT was certain or very certain 96% of the time (52 cases). As such, if pediatricians conferred ASD diagnoses when feeling certain or very certain, they would make 46 correct diagnoses and 2 incorrect diagnoses.”
The high accuracy of diagnosis when physicians are confident suggests “listening to that sense of certainty is important,” Dr. Gerdts said. Conversely, these numbers show when a physician is uncertain about diagnosing ASD, they should listen to that instinct, too, and refer.
The results of the study support having general pediatricians diagnose and move forward with their patients when the signs of ASD are more definitive, saving the less-certain cases for the more resource-intensive teams to diagnose. Many states are moving toward that “tiered” system, Dr. Gerdts said.
“For many, and in fact most children, general pediatricians are pretty accurate when making an autism diagnosis,” she said.
“Let’s get [general pediatricians] confident in recognizing when this is outside their skill and ability level,” she said. “If you’re not sure, it is better to refer them on than to misdiagnose them.”
The important missing piece she said is how to support them “so they don’t feel pressure to make that call,” Dr. Gerdts said.
This project was funded by a grant from the Bloorview Research Institute, a grant from the Canadian Institutes of Health Research and a grant from the Canadian Institutes of Health. Three coauthors consult for and receive grants from several pharmaceutical companies and other organizations. Dr. Gerdts declared no relevant financial relationships.
FROM JAMA NETWORK OPEN






