User login
Medications provide best risk-to-benefit ratio for weight loss, says expert
Lifestyle changes result in the least weight loss and may be safest, while surgery provides the most weight loss and has the greatest risk. Antiobesity medications, especially the newer ones used in combination with lifestyle changes, can provide significant and sustained weight loss with manageable side effects, said Daniel Bessesen, MD, a professor in the endocrinology, diabetes, and metabolism at University of Colorado at Denver, Aurora.
New and more effective antiobesity medications have given internists more potential options to discuss with their patients, Dr. Bessesen said. He reviewed the pros and cons of the different options.
Medications are indicated for patients with a body mass index greater than 30, including those with a weight-related comorbidity, Dr. Bessesen said. The average weight loss is 5%-15% over 3-6 months but may vary greatly. Insurance often does not cover the medication costs.
Older FDA-approved antiobesity medications
Phentermine is the most widely prescribed antiobesity medication, partly because it is the only option most people can afford out of pocket. Dr. Bessesen presented recent data showing that long-term use of phentermine was associated with greater weight loss and that patients continuously taking phentermine for 24 months lost 7.5% of their weight.
Phentermine suppresses appetite by increasing norepinephrine production. Dr. Bessesen warned that internists should be careful when prescribing it to patients with mental conditions, because it acts as a stimulant. Early studies raised concerns about the risk of cardiovascular disease (CVD) in patients taking phentermine. However, analysis of data from over 13,000 individuals showed no evidence of a relationship between phentermine exposure and CVD events.
“These data provide some reassurance that it could be used in patients with CVD risk,” he noted. Phentermine can also be combined with topiramate extended release, a combination that provides greater efficacy (up to 10% weight loss) with fewer side effects. However, this combination is less effective in patients with diabetes than in those without.
Additional treatment options included orlistat and naltrexone sustained release/bupropion SR. Orlistat is a good treatment alternative for patients with constipation and is the safest option among older anti-obesity medications, whereas naltrexone SR/bupropion SR may be useful in patients with food cravings. However, there is more variability in the individual-level benefit from these agents compared to phentermine and phentermine/topiramate ER, Dr. Bessesen said.
Newer anti‐obesity medications
Liraglutide, an agent used for the management of type 2 diabetes, has recently been approved for weight loss. Liraglutide causes moderate weight loss, and it may reduce the risk of CVD. However, there are tolerability issues, such as nausea and other risks, and Dr. Bessesen advises internists to “start at low doses and increase slowly.”
Semaglutide is the newest and most effective antiobesity drug approved by the Food and Drug Administration, providing sustained weight loss of 8% for up to 48 weeks after starting treatment. Although its efficacy is lower in patients with diabetes, Dr. Bessesen noted that “this is common for antiobesity agents, and clinicians should not refrain from prescribing it in this population.”
Setmelanotide is another new medication approved for chronic weight management in patients with monogenic obesity. This medication can be considered for patients with early-onset severe obesity with abnormal feeding behavior.
Commenting on barriers to access to new antiobesity medications, Dr. Bessesen said that “the high cost of these medications is a substantial problem, but as more companies become involved and products are on the market for a longer period of time, I am hopeful that prices will come down.”
Emerging antiobesity medications
Dr. Bessesen presented recent phase 3 data showing that treatment with tirzepatide provided sustained chronic loss and improved cardiometabolic measures with no diet. Tirzepatide, which targets receptors for glucagonlike peptide–1 and glucose-dependent insulinotropic polypeptide, is used for the management of type 2 diabetes and is expected to be reviewed soon by the FDA for its use in weight management.
A semaglutide/cagrilintide combination may also provide a new treatment option for patients with obesity. In a phase 1b trial, semaglutide/cagrilintide treatment resulted in up to 17% weight loss in patients with obesity who were otherwise healthy; however, phase 2 and 3 data are needed to confirm its efficacy.
A ‘holistic approach’
When deciding whether to prescribe antiobesity medications, Dr. Bessesen noted that medications are better than exercise alone. Factors to consider when deciding whether to prescribe drugs, as well as which ones, include costs, local regulatory guidelines, requirement for long-term use, and patient comorbidities.
He also stated that lifestyle changes, such as adopting healthy nutrition and exercising regularly, are also important and can enhance weight loss when combined with medications.
Richele Corrado, DO, MPH, agreed that lifestyle management in combination with medications may provide greater weight loss than each of these interventions alone.
“If you look at the data, exercise doesn’t help you lose much weight,” said Dr. Corrado, a staff internist and obesity medicine specialist at Walter Reed National Military Medical Center in Bethesda, Md., who spoke at the same session. She added that she has many patients who struggle to lose weight despite having a healthy lifestyle. “It’s important to discuss with these patients about medications and surgery.”
Dr. Bessesen noted that management of mental health and emotional well-being should also be an integral part of obesity management. “Treatment for obesity may be more successful when underlying psychological conditions such as depression, childhood sexual trauma, or anxiety are addressed and treated,” he said.
Dr. Bessesen was involved in the study of the efficacy of semaglutide/cagrilintide. He does not have any financial conflicts with the companies that make other mentioned medications. He has received research grants or contracts from Novo Nordisk, honoraria from Novo Nordisk, and consultantship from Eli Lilly. Dr. Corrado reported no relevant financial conflicts.
Lifestyle changes result in the least weight loss and may be safest, while surgery provides the most weight loss and has the greatest risk. Antiobesity medications, especially the newer ones used in combination with lifestyle changes, can provide significant and sustained weight loss with manageable side effects, said Daniel Bessesen, MD, a professor in the endocrinology, diabetes, and metabolism at University of Colorado at Denver, Aurora.
New and more effective antiobesity medications have given internists more potential options to discuss with their patients, Dr. Bessesen said. He reviewed the pros and cons of the different options.
Medications are indicated for patients with a body mass index greater than 30, including those with a weight-related comorbidity, Dr. Bessesen said. The average weight loss is 5%-15% over 3-6 months but may vary greatly. Insurance often does not cover the medication costs.
Older FDA-approved antiobesity medications
Phentermine is the most widely prescribed antiobesity medication, partly because it is the only option most people can afford out of pocket. Dr. Bessesen presented recent data showing that long-term use of phentermine was associated with greater weight loss and that patients continuously taking phentermine for 24 months lost 7.5% of their weight.
Phentermine suppresses appetite by increasing norepinephrine production. Dr. Bessesen warned that internists should be careful when prescribing it to patients with mental conditions, because it acts as a stimulant. Early studies raised concerns about the risk of cardiovascular disease (CVD) in patients taking phentermine. However, analysis of data from over 13,000 individuals showed no evidence of a relationship between phentermine exposure and CVD events.
“These data provide some reassurance that it could be used in patients with CVD risk,” he noted. Phentermine can also be combined with topiramate extended release, a combination that provides greater efficacy (up to 10% weight loss) with fewer side effects. However, this combination is less effective in patients with diabetes than in those without.
Additional treatment options included orlistat and naltrexone sustained release/bupropion SR. Orlistat is a good treatment alternative for patients with constipation and is the safest option among older anti-obesity medications, whereas naltrexone SR/bupropion SR may be useful in patients with food cravings. However, there is more variability in the individual-level benefit from these agents compared to phentermine and phentermine/topiramate ER, Dr. Bessesen said.
Newer anti‐obesity medications
Liraglutide, an agent used for the management of type 2 diabetes, has recently been approved for weight loss. Liraglutide causes moderate weight loss, and it may reduce the risk of CVD. However, there are tolerability issues, such as nausea and other risks, and Dr. Bessesen advises internists to “start at low doses and increase slowly.”
Semaglutide is the newest and most effective antiobesity drug approved by the Food and Drug Administration, providing sustained weight loss of 8% for up to 48 weeks after starting treatment. Although its efficacy is lower in patients with diabetes, Dr. Bessesen noted that “this is common for antiobesity agents, and clinicians should not refrain from prescribing it in this population.”
Setmelanotide is another new medication approved for chronic weight management in patients with monogenic obesity. This medication can be considered for patients with early-onset severe obesity with abnormal feeding behavior.
Commenting on barriers to access to new antiobesity medications, Dr. Bessesen said that “the high cost of these medications is a substantial problem, but as more companies become involved and products are on the market for a longer period of time, I am hopeful that prices will come down.”
Emerging antiobesity medications
Dr. Bessesen presented recent phase 3 data showing that treatment with tirzepatide provided sustained chronic loss and improved cardiometabolic measures with no diet. Tirzepatide, which targets receptors for glucagonlike peptide–1 and glucose-dependent insulinotropic polypeptide, is used for the management of type 2 diabetes and is expected to be reviewed soon by the FDA for its use in weight management.
A semaglutide/cagrilintide combination may also provide a new treatment option for patients with obesity. In a phase 1b trial, semaglutide/cagrilintide treatment resulted in up to 17% weight loss in patients with obesity who were otherwise healthy; however, phase 2 and 3 data are needed to confirm its efficacy.
A ‘holistic approach’
When deciding whether to prescribe antiobesity medications, Dr. Bessesen noted that medications are better than exercise alone. Factors to consider when deciding whether to prescribe drugs, as well as which ones, include costs, local regulatory guidelines, requirement for long-term use, and patient comorbidities.
He also stated that lifestyle changes, such as adopting healthy nutrition and exercising regularly, are also important and can enhance weight loss when combined with medications.
Richele Corrado, DO, MPH, agreed that lifestyle management in combination with medications may provide greater weight loss than each of these interventions alone.
“If you look at the data, exercise doesn’t help you lose much weight,” said Dr. Corrado, a staff internist and obesity medicine specialist at Walter Reed National Military Medical Center in Bethesda, Md., who spoke at the same session. She added that she has many patients who struggle to lose weight despite having a healthy lifestyle. “It’s important to discuss with these patients about medications and surgery.”
Dr. Bessesen noted that management of mental health and emotional well-being should also be an integral part of obesity management. “Treatment for obesity may be more successful when underlying psychological conditions such as depression, childhood sexual trauma, or anxiety are addressed and treated,” he said.
Dr. Bessesen was involved in the study of the efficacy of semaglutide/cagrilintide. He does not have any financial conflicts with the companies that make other mentioned medications. He has received research grants or contracts from Novo Nordisk, honoraria from Novo Nordisk, and consultantship from Eli Lilly. Dr. Corrado reported no relevant financial conflicts.
Lifestyle changes result in the least weight loss and may be safest, while surgery provides the most weight loss and has the greatest risk. Antiobesity medications, especially the newer ones used in combination with lifestyle changes, can provide significant and sustained weight loss with manageable side effects, said Daniel Bessesen, MD, a professor in the endocrinology, diabetes, and metabolism at University of Colorado at Denver, Aurora.
New and more effective antiobesity medications have given internists more potential options to discuss with their patients, Dr. Bessesen said. He reviewed the pros and cons of the different options.
Medications are indicated for patients with a body mass index greater than 30, including those with a weight-related comorbidity, Dr. Bessesen said. The average weight loss is 5%-15% over 3-6 months but may vary greatly. Insurance often does not cover the medication costs.
Older FDA-approved antiobesity medications
Phentermine is the most widely prescribed antiobesity medication, partly because it is the only option most people can afford out of pocket. Dr. Bessesen presented recent data showing that long-term use of phentermine was associated with greater weight loss and that patients continuously taking phentermine for 24 months lost 7.5% of their weight.
Phentermine suppresses appetite by increasing norepinephrine production. Dr. Bessesen warned that internists should be careful when prescribing it to patients with mental conditions, because it acts as a stimulant. Early studies raised concerns about the risk of cardiovascular disease (CVD) in patients taking phentermine. However, analysis of data from over 13,000 individuals showed no evidence of a relationship between phentermine exposure and CVD events.
“These data provide some reassurance that it could be used in patients with CVD risk,” he noted. Phentermine can also be combined with topiramate extended release, a combination that provides greater efficacy (up to 10% weight loss) with fewer side effects. However, this combination is less effective in patients with diabetes than in those without.
Additional treatment options included orlistat and naltrexone sustained release/bupropion SR. Orlistat is a good treatment alternative for patients with constipation and is the safest option among older anti-obesity medications, whereas naltrexone SR/bupropion SR may be useful in patients with food cravings. However, there is more variability in the individual-level benefit from these agents compared to phentermine and phentermine/topiramate ER, Dr. Bessesen said.
Newer anti‐obesity medications
Liraglutide, an agent used for the management of type 2 diabetes, has recently been approved for weight loss. Liraglutide causes moderate weight loss, and it may reduce the risk of CVD. However, there are tolerability issues, such as nausea and other risks, and Dr. Bessesen advises internists to “start at low doses and increase slowly.”
Semaglutide is the newest and most effective antiobesity drug approved by the Food and Drug Administration, providing sustained weight loss of 8% for up to 48 weeks after starting treatment. Although its efficacy is lower in patients with diabetes, Dr. Bessesen noted that “this is common for antiobesity agents, and clinicians should not refrain from prescribing it in this population.”
Setmelanotide is another new medication approved for chronic weight management in patients with monogenic obesity. This medication can be considered for patients with early-onset severe obesity with abnormal feeding behavior.
Commenting on barriers to access to new antiobesity medications, Dr. Bessesen said that “the high cost of these medications is a substantial problem, but as more companies become involved and products are on the market for a longer period of time, I am hopeful that prices will come down.”
Emerging antiobesity medications
Dr. Bessesen presented recent phase 3 data showing that treatment with tirzepatide provided sustained chronic loss and improved cardiometabolic measures with no diet. Tirzepatide, which targets receptors for glucagonlike peptide–1 and glucose-dependent insulinotropic polypeptide, is used for the management of type 2 diabetes and is expected to be reviewed soon by the FDA for its use in weight management.
A semaglutide/cagrilintide combination may also provide a new treatment option for patients with obesity. In a phase 1b trial, semaglutide/cagrilintide treatment resulted in up to 17% weight loss in patients with obesity who were otherwise healthy; however, phase 2 and 3 data are needed to confirm its efficacy.
A ‘holistic approach’
When deciding whether to prescribe antiobesity medications, Dr. Bessesen noted that medications are better than exercise alone. Factors to consider when deciding whether to prescribe drugs, as well as which ones, include costs, local regulatory guidelines, requirement for long-term use, and patient comorbidities.
He also stated that lifestyle changes, such as adopting healthy nutrition and exercising regularly, are also important and can enhance weight loss when combined with medications.
Richele Corrado, DO, MPH, agreed that lifestyle management in combination with medications may provide greater weight loss than each of these interventions alone.
“If you look at the data, exercise doesn’t help you lose much weight,” said Dr. Corrado, a staff internist and obesity medicine specialist at Walter Reed National Military Medical Center in Bethesda, Md., who spoke at the same session. She added that she has many patients who struggle to lose weight despite having a healthy lifestyle. “It’s important to discuss with these patients about medications and surgery.”
Dr. Bessesen noted that management of mental health and emotional well-being should also be an integral part of obesity management. “Treatment for obesity may be more successful when underlying psychological conditions such as depression, childhood sexual trauma, or anxiety are addressed and treated,” he said.
Dr. Bessesen was involved in the study of the efficacy of semaglutide/cagrilintide. He does not have any financial conflicts with the companies that make other mentioned medications. He has received research grants or contracts from Novo Nordisk, honoraria from Novo Nordisk, and consultantship from Eli Lilly. Dr. Corrado reported no relevant financial conflicts.
AT INTERNAL MEDICINE 2023
Lose weight, gain huge debt: N.Y. provider has sued more than 300 patients who had bariatric surgery
Seven months after Lahavah Wallace’s weight-loss operation, a New York bariatric surgery practice sued her, accusing her of “intentionally” failing to pay nearly $18,000 of her bill.
Long Island Minimally Invasive Surgery, which does business as the New York Bariatric Group, went on to accuse Ms. Wallace of “embezzlement,” alleging she kept insurance payments that should have been turned over to the practice.
Ms. Wallace denies the allegations, which the bariatric practice has leveled against patients in hundreds of debt-collection lawsuits filed over the past 4 years, court records in New York state show.
In about 60 cases, the lawsuits demanded $100,000 or more from patients. Some patients were found liable for tens of thousands of dollars in interest charges or wound up shackled with debt that could take a decade or more to shake. Others are facing the likely prospect of six-figure financial penalties, court records show.
Backed by a major private equity firm, the bariatric practice spends millions each year on advertisements featuring patients who have dropped 100 pounds or more after bariatric procedures, sometimes having had a portion of their stomachs removed. The ads have run on TV, online, and on New York City subway posters.
The online ads, often showcasing the slogan “Stop obesity for life,” appealed to Ms. Wallace, who lives in Brooklyn and works as a legal assistant for the state of New York. She said she turned over checks from her insurer to the bariatric group and was stunned when the medical practice hauled her into court citing an “out-of-network payment agreement” she had signed before her surgery.
“I really didn’t know what I was signing,” Ms. Wallace told KFF Health News. “I didn’t pay enough attention.”
Shawn Garber, MD, a bariatric surgeon who founded the practice in 2000 on Long Island and serves as its CEO, said that “prior to rendering services” his office staff advises patients of the costs and their responsibility to pay the bill.
The bariatric group has cited these out-of-network payment agreements in at least 300 lawsuits filed against patients from January 2019 to 2022 demanding nearly $19 million to cover medical bills, interest charges, and attorney’s fees, a KFF Health News review of New York state court records found.
Danny De Voe, a partner at Sahn Ward Braff Koblenz law firm in Uniondale, N.Y., who filed many of those suits, declined to comment, citing attorney-client privilege.
In most cases, the medical practice had agreed to accept an insurance company’s out-of-network rate as full payment for its services – with caveats, according to court filings.
In the agreements they signed, patients promised to pay any coinsurance, meeting any deductible, and pass on to the medical practice any reimbursement checks they received from their health plans within 7 days.
KFF Health News found – while legal fees and other costs can layer on thousands more.
Elisabeth Benjamin, a lawyer with the Community Service Society of New York, said conflicts can arise when insurers send checks to pay for out-of-network medical services to patients rather than reimbursing a medical provider directly.
“We would prefer to see regulators step in and stop that practice,” she said, adding it “causes tension between providers and patients.”
That’s certainly true for Ms. Wallace. The surgery practice sued her in August 2022demanding $17,981 in fees it said remained unpaid after her January 2022 laparoscopic sleeve gastrectomy, an operation in which much of the stomach is removed to assist weight loss.
The lawsuit also tacked on a demand for $5,993 in attorney’s fees, court records show.
The suit alleges Ms. Wallace signed the contract even though she “had no intention” of paying her bills. The complaint goes on to accuse her of “committing embezzlement” by “willfully, intentionally, deliberately and maliciously” depositing checks from her health plan into her personal account.
The suit doesn’t include details to substantiate these claims, and Ms. Wallace said in her court response they are not true. Ms. Wallace said she turned over checks for the charges.
“They billed the insurance for everything they possibly could,” Ms. Wallace said.
In September, Ms. Wallace filed for bankruptcy, hoping to discharge the bariatric care debt along with about $4,700 in unrelated credit card charges.
The medical practice fired back in November by filing an “adversary complaint” in her Brooklyn bankruptcy court proceeding that argues her medical debt should not be forgiven because Ms. Wallace committed fraud.
The adversary complaint, which is pending in the bankruptcy case, accuses Ms. Wallace of “fraudulently” inducing the surgery center to perform “elective medical procedures” without requiring payment up front.
Both the harsh wording and claims of wrongdoing have infuriated Ms. Wallace and her attorney, Jacob Silver, of Brooklyn.
Mr. Silver wants the medical practice to turn over records of the payments received from Ms. Wallace. “There is no fraud here,” he said. “This is frivolous. We are taking a no-settlement position.”
Gaining debt
Few patients sued by the bariatric practice mount a defense in court and those who do fight often lose, court records show.
The medical practice won default judgments totaling nearly $6 million in about 90 of the 300 cases in the sample reviewed by KFF Health News. Default judgments are entered when the defendant fails to respond.
Many cases either are pending, or it is not clear from court filings how they were resolved.
Some patients tried to argue that the fees were too high or that they didn’t understand going in how much they could owe. One woman, trying to push back against a demand for more than $100,000, said in a legal filing that she “was given numerous papers to sign without anyone of the staff members explaining to me what it actually meant.” Another patient, who was sued for more than $40,000, wrote: “I don’t have the means to pay this bill.”
Among the cases described in court records:
- A Westchester County, N.Y., woman was sued for $102,556 and settled for $72,000 in May 2021. She agreed to pay $7,500 upon signing the settlement and $500 a month from September 2021 to May 2032.
- A Peekskill, N.Y., woman in a December 2019 judgment was held liable for $384,092, which included $94,047 in interest.
- A Newburgh, N.Y., man was sued in 2021 for $252,309 in medical bills, 12% interest, and $84,103 in attorneys’ fees. The case is pending.
Robert Cohen, a longtime attorney for the bariatric practice, testified in a November 2021 hearing that the lawyers take “a contingency fee of one-third of our recovery” in these cases. In that case, Mr. Cohen had requested $13,578 based on his contingency fee arrangement. He testified that he spent 7.3 hours on the case and that his customary billing rate was $475 per hour, which came to $3,467.50. The judge awarded the lower amount, according to a transcript of the hearing.
Teresa LaMasters, MD, president of the American Society for Metabolic and Bariatric Surgery, said suing patients for large sums “is not a common practice” among bariatric surgeons.
“This is not what the vast majority in the field would espouse,” she said.
But Dr. Garber, the NYBG’s chief executive, suggested patients deserve blame.
“These lawsuits stem from these patients stealing the insurance money rather than forwarding it onto NYBG as they are morally and contractually obligated to do,” Dr. Garber wrote in an email to KFF Health News.
Dr. Garber added: “The issue is not with what we bill, but rather with the fact that the insurance companies refuse to send payment directly to us.”
‘A kooky system’
Defense attorneys argue that many patients don’t fully comprehend the perils of failing to pay on time – for whatever reason.
In a few cases, patients admitted pocketing checks they were obligated to turn over to the medical practice. But for the most part, court records don’t specify how many such checks were issued and for what amounts – or whether the patient improperly cashed them.
“It’s a kooky system,” said Paul Brite, an attorney who has faced off against the bariatric practice in court.
“You sign these documents that could cost you tons of money. It shouldn’t be that way,” he said. “This can ruin their financial life.”
New York lawmakers have acted to limit the damage from medical debt, including “surprise bills.”
In November, Democratic Gov. Kathy Hochul signed legislation that prohibits health care providers from slapping liens on a primary residence or garnishing wages.
But contracts with onerous repayment terms represent an “evolving area of law” and an alarming “new twist” on concerns over medical debt, said Ms. Benjamin, the community service society lawyer.
She said contract “accelerator clauses” that trigger severe penalties if patients miss payments should not be permitted for medical debt.
“If you default, the full amount is due,” she said. “This is really a bummer.”
‘Fair market value’
The debt collection lawsuits argue that weight-loss patients had agreed to pay “fair market value” for services – and the doctors are only trying to secure money they are due.
But some prices far exceed typical insurance payments for obesity treatments across the country, according to a medical billing data registry. Surgeons performed about 200,000 bariatric operations in 2020, according to the bariatric surgery society.
Ms. Wallace, the Brooklyn legal assistant, was billed $60,500 for her lap sleeve gastrectomy, though how much her insurance actually paid remains to be hashed out in court.
Michael Arrigo, a California medical billing expert at No World Borders, called the prices “outrageous” and “unreasonable and, in fact, likely unconscionable.”
“I disagree that these are fair market charges,” he said.
Dr. LaMasters called the gastrectomy price billed to Ms. Wallace “really expensive” and “a severe outlier.” While charges vary by region, she quoted a typical price of around $22,000.
Dr. Garber said NYBG “bills at usual and customary rates” determined by Fair Health, a New York City-based repository of insurance claims data. Fair Health “sets these rates based upon the acceptable price for our geographic location,” he said.
But Rachel Kent, Fair Health’s senior director of marketing, told KFF Health News that the group “does not set rates, nor determine or take any position on what constitutes ‘usual and customary rates.’ ” Instead, it reports the prices providers are charging in a given area.
Overall, Fair Health data shows huge price variations even in adjacent ZIP codes in the metro area. In Long Island’s Roslyn Heights neighborhood, where NYBG is based, Fair Health lists the out-of-network price charged by providers in the area as $60,500, the figure Ms. Wallace was billed.
But in several other New York City–area ZIP codes the price charged for the gastrectomy procedure hovers around $20,000, according to the data bank. The price in Manhattan is $17,500, for instance, according to Fair Health.
Nationwide, the average cost in 2021 for bariatric surgery done in a hospital was $32,868, according to a KFF analysis of health insurance claims.
Private equity arrives
Dr. Garber said in a court affidavit in May 2022 that he founded the bariatric practice “with a singular focus: providing safe, effective care to patients suffering from obesity and its resulting complications.”
Under his leadership, the practice has “developed into New York’s elite institution for obesity treatment,” Dr. Garber said. He said the group’s surgeons are “highly sought after to train other bariatric surgeons throughout the country and are active in the development of new, cutting-edge bariatric surgery techniques.”
In 2017, Dr. Garber and partners agreed on a business plan to help spur growth and “attract private equity investment,” according to the affidavit.
They formed a separate company to handle the bariatric practice’s business side. Known as management services organizations, such companies provide a way for private equity investors to circumvent laws in some states that prohibit nonphysicians from owning a stake in a medical practice.
In August 2019, the private equity firm Sentinel Capital Partners bought 65% of the MSO for $156.5 million, according to Dr. Garber’s affidavit. The management company is now known as New You Bariatric Group. The private equity firm did not respond to requests for comment.
Dr. Garber, in a September 2021 American Society for Metabolic and Bariatric Surgery webinar viewable online, said the weight-loss practice spends $6 million a year on media and marketing directly to patients – and is on a roll. Nationally, bariatric surgery is growing 6% annually, he said. NYBG boasts two dozen offices in the tri-state area of New York, New Jersey, and Connecticut and is poised to expand into more states.
“Since private equity, we’ve been growing at 30%-40% year over year,” Dr. Garber said.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Seven months after Lahavah Wallace’s weight-loss operation, a New York bariatric surgery practice sued her, accusing her of “intentionally” failing to pay nearly $18,000 of her bill.
Long Island Minimally Invasive Surgery, which does business as the New York Bariatric Group, went on to accuse Ms. Wallace of “embezzlement,” alleging she kept insurance payments that should have been turned over to the practice.
Ms. Wallace denies the allegations, which the bariatric practice has leveled against patients in hundreds of debt-collection lawsuits filed over the past 4 years, court records in New York state show.
In about 60 cases, the lawsuits demanded $100,000 or more from patients. Some patients were found liable for tens of thousands of dollars in interest charges or wound up shackled with debt that could take a decade or more to shake. Others are facing the likely prospect of six-figure financial penalties, court records show.
Backed by a major private equity firm, the bariatric practice spends millions each year on advertisements featuring patients who have dropped 100 pounds or more after bariatric procedures, sometimes having had a portion of their stomachs removed. The ads have run on TV, online, and on New York City subway posters.
The online ads, often showcasing the slogan “Stop obesity for life,” appealed to Ms. Wallace, who lives in Brooklyn and works as a legal assistant for the state of New York. She said she turned over checks from her insurer to the bariatric group and was stunned when the medical practice hauled her into court citing an “out-of-network payment agreement” she had signed before her surgery.
“I really didn’t know what I was signing,” Ms. Wallace told KFF Health News. “I didn’t pay enough attention.”
Shawn Garber, MD, a bariatric surgeon who founded the practice in 2000 on Long Island and serves as its CEO, said that “prior to rendering services” his office staff advises patients of the costs and their responsibility to pay the bill.
The bariatric group has cited these out-of-network payment agreements in at least 300 lawsuits filed against patients from January 2019 to 2022 demanding nearly $19 million to cover medical bills, interest charges, and attorney’s fees, a KFF Health News review of New York state court records found.
Danny De Voe, a partner at Sahn Ward Braff Koblenz law firm in Uniondale, N.Y., who filed many of those suits, declined to comment, citing attorney-client privilege.
In most cases, the medical practice had agreed to accept an insurance company’s out-of-network rate as full payment for its services – with caveats, according to court filings.
In the agreements they signed, patients promised to pay any coinsurance, meeting any deductible, and pass on to the medical practice any reimbursement checks they received from their health plans within 7 days.
KFF Health News found – while legal fees and other costs can layer on thousands more.
Elisabeth Benjamin, a lawyer with the Community Service Society of New York, said conflicts can arise when insurers send checks to pay for out-of-network medical services to patients rather than reimbursing a medical provider directly.
“We would prefer to see regulators step in and stop that practice,” she said, adding it “causes tension between providers and patients.”
That’s certainly true for Ms. Wallace. The surgery practice sued her in August 2022demanding $17,981 in fees it said remained unpaid after her January 2022 laparoscopic sleeve gastrectomy, an operation in which much of the stomach is removed to assist weight loss.
The lawsuit also tacked on a demand for $5,993 in attorney’s fees, court records show.
The suit alleges Ms. Wallace signed the contract even though she “had no intention” of paying her bills. The complaint goes on to accuse her of “committing embezzlement” by “willfully, intentionally, deliberately and maliciously” depositing checks from her health plan into her personal account.
The suit doesn’t include details to substantiate these claims, and Ms. Wallace said in her court response they are not true. Ms. Wallace said she turned over checks for the charges.
“They billed the insurance for everything they possibly could,” Ms. Wallace said.
In September, Ms. Wallace filed for bankruptcy, hoping to discharge the bariatric care debt along with about $4,700 in unrelated credit card charges.
The medical practice fired back in November by filing an “adversary complaint” in her Brooklyn bankruptcy court proceeding that argues her medical debt should not be forgiven because Ms. Wallace committed fraud.
The adversary complaint, which is pending in the bankruptcy case, accuses Ms. Wallace of “fraudulently” inducing the surgery center to perform “elective medical procedures” without requiring payment up front.
Both the harsh wording and claims of wrongdoing have infuriated Ms. Wallace and her attorney, Jacob Silver, of Brooklyn.
Mr. Silver wants the medical practice to turn over records of the payments received from Ms. Wallace. “There is no fraud here,” he said. “This is frivolous. We are taking a no-settlement position.”
Gaining debt
Few patients sued by the bariatric practice mount a defense in court and those who do fight often lose, court records show.
The medical practice won default judgments totaling nearly $6 million in about 90 of the 300 cases in the sample reviewed by KFF Health News. Default judgments are entered when the defendant fails to respond.
Many cases either are pending, or it is not clear from court filings how they were resolved.
Some patients tried to argue that the fees were too high or that they didn’t understand going in how much they could owe. One woman, trying to push back against a demand for more than $100,000, said in a legal filing that she “was given numerous papers to sign without anyone of the staff members explaining to me what it actually meant.” Another patient, who was sued for more than $40,000, wrote: “I don’t have the means to pay this bill.”
Among the cases described in court records:
- A Westchester County, N.Y., woman was sued for $102,556 and settled for $72,000 in May 2021. She agreed to pay $7,500 upon signing the settlement and $500 a month from September 2021 to May 2032.
- A Peekskill, N.Y., woman in a December 2019 judgment was held liable for $384,092, which included $94,047 in interest.
- A Newburgh, N.Y., man was sued in 2021 for $252,309 in medical bills, 12% interest, and $84,103 in attorneys’ fees. The case is pending.
Robert Cohen, a longtime attorney for the bariatric practice, testified in a November 2021 hearing that the lawyers take “a contingency fee of one-third of our recovery” in these cases. In that case, Mr. Cohen had requested $13,578 based on his contingency fee arrangement. He testified that he spent 7.3 hours on the case and that his customary billing rate was $475 per hour, which came to $3,467.50. The judge awarded the lower amount, according to a transcript of the hearing.
Teresa LaMasters, MD, president of the American Society for Metabolic and Bariatric Surgery, said suing patients for large sums “is not a common practice” among bariatric surgeons.
“This is not what the vast majority in the field would espouse,” she said.
But Dr. Garber, the NYBG’s chief executive, suggested patients deserve blame.
“These lawsuits stem from these patients stealing the insurance money rather than forwarding it onto NYBG as they are morally and contractually obligated to do,” Dr. Garber wrote in an email to KFF Health News.
Dr. Garber added: “The issue is not with what we bill, but rather with the fact that the insurance companies refuse to send payment directly to us.”
‘A kooky system’
Defense attorneys argue that many patients don’t fully comprehend the perils of failing to pay on time – for whatever reason.
In a few cases, patients admitted pocketing checks they were obligated to turn over to the medical practice. But for the most part, court records don’t specify how many such checks were issued and for what amounts – or whether the patient improperly cashed them.
“It’s a kooky system,” said Paul Brite, an attorney who has faced off against the bariatric practice in court.
“You sign these documents that could cost you tons of money. It shouldn’t be that way,” he said. “This can ruin their financial life.”
New York lawmakers have acted to limit the damage from medical debt, including “surprise bills.”
In November, Democratic Gov. Kathy Hochul signed legislation that prohibits health care providers from slapping liens on a primary residence or garnishing wages.
But contracts with onerous repayment terms represent an “evolving area of law” and an alarming “new twist” on concerns over medical debt, said Ms. Benjamin, the community service society lawyer.
She said contract “accelerator clauses” that trigger severe penalties if patients miss payments should not be permitted for medical debt.
“If you default, the full amount is due,” she said. “This is really a bummer.”
‘Fair market value’
The debt collection lawsuits argue that weight-loss patients had agreed to pay “fair market value” for services – and the doctors are only trying to secure money they are due.
But some prices far exceed typical insurance payments for obesity treatments across the country, according to a medical billing data registry. Surgeons performed about 200,000 bariatric operations in 2020, according to the bariatric surgery society.
Ms. Wallace, the Brooklyn legal assistant, was billed $60,500 for her lap sleeve gastrectomy, though how much her insurance actually paid remains to be hashed out in court.
Michael Arrigo, a California medical billing expert at No World Borders, called the prices “outrageous” and “unreasonable and, in fact, likely unconscionable.”
“I disagree that these are fair market charges,” he said.
Dr. LaMasters called the gastrectomy price billed to Ms. Wallace “really expensive” and “a severe outlier.” While charges vary by region, she quoted a typical price of around $22,000.
Dr. Garber said NYBG “bills at usual and customary rates” determined by Fair Health, a New York City-based repository of insurance claims data. Fair Health “sets these rates based upon the acceptable price for our geographic location,” he said.
But Rachel Kent, Fair Health’s senior director of marketing, told KFF Health News that the group “does not set rates, nor determine or take any position on what constitutes ‘usual and customary rates.’ ” Instead, it reports the prices providers are charging in a given area.
Overall, Fair Health data shows huge price variations even in adjacent ZIP codes in the metro area. In Long Island’s Roslyn Heights neighborhood, where NYBG is based, Fair Health lists the out-of-network price charged by providers in the area as $60,500, the figure Ms. Wallace was billed.
But in several other New York City–area ZIP codes the price charged for the gastrectomy procedure hovers around $20,000, according to the data bank. The price in Manhattan is $17,500, for instance, according to Fair Health.
Nationwide, the average cost in 2021 for bariatric surgery done in a hospital was $32,868, according to a KFF analysis of health insurance claims.
Private equity arrives
Dr. Garber said in a court affidavit in May 2022 that he founded the bariatric practice “with a singular focus: providing safe, effective care to patients suffering from obesity and its resulting complications.”
Under his leadership, the practice has “developed into New York’s elite institution for obesity treatment,” Dr. Garber said. He said the group’s surgeons are “highly sought after to train other bariatric surgeons throughout the country and are active in the development of new, cutting-edge bariatric surgery techniques.”
In 2017, Dr. Garber and partners agreed on a business plan to help spur growth and “attract private equity investment,” according to the affidavit.
They formed a separate company to handle the bariatric practice’s business side. Known as management services organizations, such companies provide a way for private equity investors to circumvent laws in some states that prohibit nonphysicians from owning a stake in a medical practice.
In August 2019, the private equity firm Sentinel Capital Partners bought 65% of the MSO for $156.5 million, according to Dr. Garber’s affidavit. The management company is now known as New You Bariatric Group. The private equity firm did not respond to requests for comment.
Dr. Garber, in a September 2021 American Society for Metabolic and Bariatric Surgery webinar viewable online, said the weight-loss practice spends $6 million a year on media and marketing directly to patients – and is on a roll. Nationally, bariatric surgery is growing 6% annually, he said. NYBG boasts two dozen offices in the tri-state area of New York, New Jersey, and Connecticut and is poised to expand into more states.
“Since private equity, we’ve been growing at 30%-40% year over year,” Dr. Garber said.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Seven months after Lahavah Wallace’s weight-loss operation, a New York bariatric surgery practice sued her, accusing her of “intentionally” failing to pay nearly $18,000 of her bill.
Long Island Minimally Invasive Surgery, which does business as the New York Bariatric Group, went on to accuse Ms. Wallace of “embezzlement,” alleging she kept insurance payments that should have been turned over to the practice.
Ms. Wallace denies the allegations, which the bariatric practice has leveled against patients in hundreds of debt-collection lawsuits filed over the past 4 years, court records in New York state show.
In about 60 cases, the lawsuits demanded $100,000 or more from patients. Some patients were found liable for tens of thousands of dollars in interest charges or wound up shackled with debt that could take a decade or more to shake. Others are facing the likely prospect of six-figure financial penalties, court records show.
Backed by a major private equity firm, the bariatric practice spends millions each year on advertisements featuring patients who have dropped 100 pounds or more after bariatric procedures, sometimes having had a portion of their stomachs removed. The ads have run on TV, online, and on New York City subway posters.
The online ads, often showcasing the slogan “Stop obesity for life,” appealed to Ms. Wallace, who lives in Brooklyn and works as a legal assistant for the state of New York. She said she turned over checks from her insurer to the bariatric group and was stunned when the medical practice hauled her into court citing an “out-of-network payment agreement” she had signed before her surgery.
“I really didn’t know what I was signing,” Ms. Wallace told KFF Health News. “I didn’t pay enough attention.”
Shawn Garber, MD, a bariatric surgeon who founded the practice in 2000 on Long Island and serves as its CEO, said that “prior to rendering services” his office staff advises patients of the costs and their responsibility to pay the bill.
The bariatric group has cited these out-of-network payment agreements in at least 300 lawsuits filed against patients from January 2019 to 2022 demanding nearly $19 million to cover medical bills, interest charges, and attorney’s fees, a KFF Health News review of New York state court records found.
Danny De Voe, a partner at Sahn Ward Braff Koblenz law firm in Uniondale, N.Y., who filed many of those suits, declined to comment, citing attorney-client privilege.
In most cases, the medical practice had agreed to accept an insurance company’s out-of-network rate as full payment for its services – with caveats, according to court filings.
In the agreements they signed, patients promised to pay any coinsurance, meeting any deductible, and pass on to the medical practice any reimbursement checks they received from their health plans within 7 days.
KFF Health News found – while legal fees and other costs can layer on thousands more.
Elisabeth Benjamin, a lawyer with the Community Service Society of New York, said conflicts can arise when insurers send checks to pay for out-of-network medical services to patients rather than reimbursing a medical provider directly.
“We would prefer to see regulators step in and stop that practice,” she said, adding it “causes tension between providers and patients.”
That’s certainly true for Ms. Wallace. The surgery practice sued her in August 2022demanding $17,981 in fees it said remained unpaid after her January 2022 laparoscopic sleeve gastrectomy, an operation in which much of the stomach is removed to assist weight loss.
The lawsuit also tacked on a demand for $5,993 in attorney’s fees, court records show.
The suit alleges Ms. Wallace signed the contract even though she “had no intention” of paying her bills. The complaint goes on to accuse her of “committing embezzlement” by “willfully, intentionally, deliberately and maliciously” depositing checks from her health plan into her personal account.
The suit doesn’t include details to substantiate these claims, and Ms. Wallace said in her court response they are not true. Ms. Wallace said she turned over checks for the charges.
“They billed the insurance for everything they possibly could,” Ms. Wallace said.
In September, Ms. Wallace filed for bankruptcy, hoping to discharge the bariatric care debt along with about $4,700 in unrelated credit card charges.
The medical practice fired back in November by filing an “adversary complaint” in her Brooklyn bankruptcy court proceeding that argues her medical debt should not be forgiven because Ms. Wallace committed fraud.
The adversary complaint, which is pending in the bankruptcy case, accuses Ms. Wallace of “fraudulently” inducing the surgery center to perform “elective medical procedures” without requiring payment up front.
Both the harsh wording and claims of wrongdoing have infuriated Ms. Wallace and her attorney, Jacob Silver, of Brooklyn.
Mr. Silver wants the medical practice to turn over records of the payments received from Ms. Wallace. “There is no fraud here,” he said. “This is frivolous. We are taking a no-settlement position.”
Gaining debt
Few patients sued by the bariatric practice mount a defense in court and those who do fight often lose, court records show.
The medical practice won default judgments totaling nearly $6 million in about 90 of the 300 cases in the sample reviewed by KFF Health News. Default judgments are entered when the defendant fails to respond.
Many cases either are pending, or it is not clear from court filings how they were resolved.
Some patients tried to argue that the fees were too high or that they didn’t understand going in how much they could owe. One woman, trying to push back against a demand for more than $100,000, said in a legal filing that she “was given numerous papers to sign without anyone of the staff members explaining to me what it actually meant.” Another patient, who was sued for more than $40,000, wrote: “I don’t have the means to pay this bill.”
Among the cases described in court records:
- A Westchester County, N.Y., woman was sued for $102,556 and settled for $72,000 in May 2021. She agreed to pay $7,500 upon signing the settlement and $500 a month from September 2021 to May 2032.
- A Peekskill, N.Y., woman in a December 2019 judgment was held liable for $384,092, which included $94,047 in interest.
- A Newburgh, N.Y., man was sued in 2021 for $252,309 in medical bills, 12% interest, and $84,103 in attorneys’ fees. The case is pending.
Robert Cohen, a longtime attorney for the bariatric practice, testified in a November 2021 hearing that the lawyers take “a contingency fee of one-third of our recovery” in these cases. In that case, Mr. Cohen had requested $13,578 based on his contingency fee arrangement. He testified that he spent 7.3 hours on the case and that his customary billing rate was $475 per hour, which came to $3,467.50. The judge awarded the lower amount, according to a transcript of the hearing.
Teresa LaMasters, MD, president of the American Society for Metabolic and Bariatric Surgery, said suing patients for large sums “is not a common practice” among bariatric surgeons.
“This is not what the vast majority in the field would espouse,” she said.
But Dr. Garber, the NYBG’s chief executive, suggested patients deserve blame.
“These lawsuits stem from these patients stealing the insurance money rather than forwarding it onto NYBG as they are morally and contractually obligated to do,” Dr. Garber wrote in an email to KFF Health News.
Dr. Garber added: “The issue is not with what we bill, but rather with the fact that the insurance companies refuse to send payment directly to us.”
‘A kooky system’
Defense attorneys argue that many patients don’t fully comprehend the perils of failing to pay on time – for whatever reason.
In a few cases, patients admitted pocketing checks they were obligated to turn over to the medical practice. But for the most part, court records don’t specify how many such checks were issued and for what amounts – or whether the patient improperly cashed them.
“It’s a kooky system,” said Paul Brite, an attorney who has faced off against the bariatric practice in court.
“You sign these documents that could cost you tons of money. It shouldn’t be that way,” he said. “This can ruin their financial life.”
New York lawmakers have acted to limit the damage from medical debt, including “surprise bills.”
In November, Democratic Gov. Kathy Hochul signed legislation that prohibits health care providers from slapping liens on a primary residence or garnishing wages.
But contracts with onerous repayment terms represent an “evolving area of law” and an alarming “new twist” on concerns over medical debt, said Ms. Benjamin, the community service society lawyer.
She said contract “accelerator clauses” that trigger severe penalties if patients miss payments should not be permitted for medical debt.
“If you default, the full amount is due,” she said. “This is really a bummer.”
‘Fair market value’
The debt collection lawsuits argue that weight-loss patients had agreed to pay “fair market value” for services – and the doctors are only trying to secure money they are due.
But some prices far exceed typical insurance payments for obesity treatments across the country, according to a medical billing data registry. Surgeons performed about 200,000 bariatric operations in 2020, according to the bariatric surgery society.
Ms. Wallace, the Brooklyn legal assistant, was billed $60,500 for her lap sleeve gastrectomy, though how much her insurance actually paid remains to be hashed out in court.
Michael Arrigo, a California medical billing expert at No World Borders, called the prices “outrageous” and “unreasonable and, in fact, likely unconscionable.”
“I disagree that these are fair market charges,” he said.
Dr. LaMasters called the gastrectomy price billed to Ms. Wallace “really expensive” and “a severe outlier.” While charges vary by region, she quoted a typical price of around $22,000.
Dr. Garber said NYBG “bills at usual and customary rates” determined by Fair Health, a New York City-based repository of insurance claims data. Fair Health “sets these rates based upon the acceptable price for our geographic location,” he said.
But Rachel Kent, Fair Health’s senior director of marketing, told KFF Health News that the group “does not set rates, nor determine or take any position on what constitutes ‘usual and customary rates.’ ” Instead, it reports the prices providers are charging in a given area.
Overall, Fair Health data shows huge price variations even in adjacent ZIP codes in the metro area. In Long Island’s Roslyn Heights neighborhood, where NYBG is based, Fair Health lists the out-of-network price charged by providers in the area as $60,500, the figure Ms. Wallace was billed.
But in several other New York City–area ZIP codes the price charged for the gastrectomy procedure hovers around $20,000, according to the data bank. The price in Manhattan is $17,500, for instance, according to Fair Health.
Nationwide, the average cost in 2021 for bariatric surgery done in a hospital was $32,868, according to a KFF analysis of health insurance claims.
Private equity arrives
Dr. Garber said in a court affidavit in May 2022 that he founded the bariatric practice “with a singular focus: providing safe, effective care to patients suffering from obesity and its resulting complications.”
Under his leadership, the practice has “developed into New York’s elite institution for obesity treatment,” Dr. Garber said. He said the group’s surgeons are “highly sought after to train other bariatric surgeons throughout the country and are active in the development of new, cutting-edge bariatric surgery techniques.”
In 2017, Dr. Garber and partners agreed on a business plan to help spur growth and “attract private equity investment,” according to the affidavit.
They formed a separate company to handle the bariatric practice’s business side. Known as management services organizations, such companies provide a way for private equity investors to circumvent laws in some states that prohibit nonphysicians from owning a stake in a medical practice.
In August 2019, the private equity firm Sentinel Capital Partners bought 65% of the MSO for $156.5 million, according to Dr. Garber’s affidavit. The management company is now known as New You Bariatric Group. The private equity firm did not respond to requests for comment.
Dr. Garber, in a September 2021 American Society for Metabolic and Bariatric Surgery webinar viewable online, said the weight-loss practice spends $6 million a year on media and marketing directly to patients – and is on a roll. Nationally, bariatric surgery is growing 6% annually, he said. NYBG boasts two dozen offices in the tri-state area of New York, New Jersey, and Connecticut and is poised to expand into more states.
“Since private equity, we’ve been growing at 30%-40% year over year,” Dr. Garber said.
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Bariatric surgery cuts risk for obesity-related cancers in half: Study
For years evidence has pointed to multiple health benefits associated with bariatric surgery, including improvements in diabetes, sleep apnea, and blood pressure. Now researchers are adding cutting cancer risk by more than half to the list.
compared with 8.9% of their peers who did not undergo such surgery.
“We did see a difference in breast cancer, colon cancer, liver cancer, and ovarian cancer incidence. ... with patients in the bariatric surgery group having lower incidence of these four types of cancers when compared to the nonsurgical control group,” said Vibhu Chittajallu, MD, lead author and a gastroenterology fellow at Case Western Reserve University and University Hospitals in Cleveland.
The obesity epidemic is “one of the most serious health challenges in the United States today,” Dr. Chittajallu added at an April 27 media briefing during which select research was previewed for the annual Digestive Disease Week®. Obesity has been associated with multiple serious illnesses, including type 2 diabetes, heart disease, and cancer.
Obesity is also common. The Centers for Disease Control and Prevention reports that nearly 42% of American adults have obesity, and rates continue to rise.
Dr. Chittajallu and colleagues used billing codes in a national database to identify 55,789 patients with obesity who underwent bariatric surgery (sleeve gastrectomy, gastric bypass, or gastric band procedures) and a control group of the same size who did not have surgery.
Investigators controlled for risk factors that contribute to cancer development, including smoking history, alcohol use, heart disease, and hormone therapies.
Key findings
In 10 years of follow-up, 2,206 patients who underwent bariatric surgery developed an obesity-associated cancer, compared with 4,960 patients who did not have bariatric surgery.
The bariatric surgery group had lower numbers of new cases for six types of cancers (Table 1).
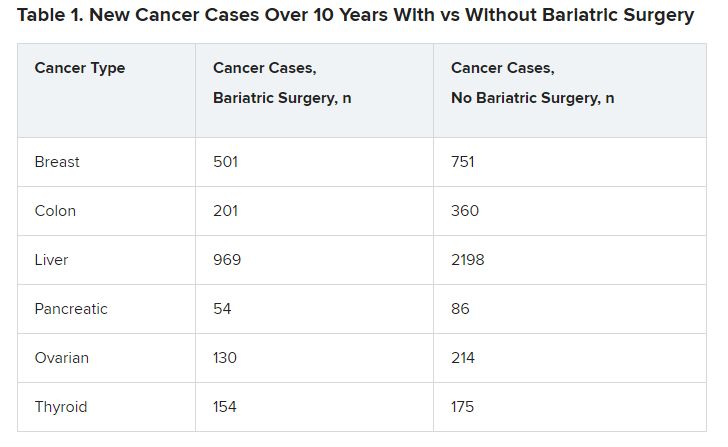
The differences were significant in four cancer types associated with obesity: breast cancer (P = .001), colon cancer (P < .01), liver cancer (P < .01), and ovarian cancer (P = .002).
The incidence of several other cancers, including renal carcinoma, and rectal and endometrial cancers, was not significantly different between the groups.
The mechanisms underlying excess cancer cases in patients with obesity are not completely understood, Dr. Chittajallu said. Bariatric surgery has been shown to decrease excess inflammation, elevate insulin, and moderate hormone levels.
‘Fascinating’ study but questions remain
The study is “fascinating,” said Loren Laine, MD, moderator of the media briefing. “Obesity is clearly associated with a number of different cancers, and that’s very important. So, it makes logical sense that if you lose weight, you will reduce that risk.”
Although investigators controlled for several known cancer risk factors, there are some they couldn’t control for because they were not included in the database, and there could be unknowns that also affected the results, noted Dr. Laine, who is professor of medicine (digestive diseases) and chief of digestive health at Yale University in New Haven, Conn.
“You have to be circumspect when you look at retrospective observational studies,” he added.
It would be helpful to know when most cancers developed over the 10 years, Dr. Laine said. Dr. Chittajallu responded that the research team did not include cancers that developed in the first year after bariatric surgery to minimize incidental findings, but he did not provide a timeline for the cancers that developed.
Another unanswered question, Dr. Laine said, is whether a dose-response relationship exists. If future research shows that the more weight a person loses, the more likely they are to have a reduction in cancer risk, “that would be fascinating,” he said. Also, it would be interesting to know if endoscopic interventions and weight-loss medications decrease cancer risks in people with obesity.
More research is needed to understand how bariatric surgery affects cancer risk, Dr. Chittajallu said. “But the significant findings from this study suggest it’s an exciting avenue for further study.”
DDW 2023 will be held May 6-9 in Chicago and virtually.
The study was independently supported. Dr. Chittajallu and Dr. Laine have reported no relevant financial relationships.
The meeting is sponsored by the American Gastroenterological Association, the American Association for the Study of Liver Diseases, the American Society for Gastrointestinal Endoscopy, and the Society for Surgery of the Alimentary Tract.
A version of this article first appeared on Medscape.com.
For years evidence has pointed to multiple health benefits associated with bariatric surgery, including improvements in diabetes, sleep apnea, and blood pressure. Now researchers are adding cutting cancer risk by more than half to the list.
compared with 8.9% of their peers who did not undergo such surgery.
“We did see a difference in breast cancer, colon cancer, liver cancer, and ovarian cancer incidence. ... with patients in the bariatric surgery group having lower incidence of these four types of cancers when compared to the nonsurgical control group,” said Vibhu Chittajallu, MD, lead author and a gastroenterology fellow at Case Western Reserve University and University Hospitals in Cleveland.
The obesity epidemic is “one of the most serious health challenges in the United States today,” Dr. Chittajallu added at an April 27 media briefing during which select research was previewed for the annual Digestive Disease Week®. Obesity has been associated with multiple serious illnesses, including type 2 diabetes, heart disease, and cancer.
Obesity is also common. The Centers for Disease Control and Prevention reports that nearly 42% of American adults have obesity, and rates continue to rise.
Dr. Chittajallu and colleagues used billing codes in a national database to identify 55,789 patients with obesity who underwent bariatric surgery (sleeve gastrectomy, gastric bypass, or gastric band procedures) and a control group of the same size who did not have surgery.
Investigators controlled for risk factors that contribute to cancer development, including smoking history, alcohol use, heart disease, and hormone therapies.
Key findings
In 10 years of follow-up, 2,206 patients who underwent bariatric surgery developed an obesity-associated cancer, compared with 4,960 patients who did not have bariatric surgery.
The bariatric surgery group had lower numbers of new cases for six types of cancers (Table 1).

The differences were significant in four cancer types associated with obesity: breast cancer (P = .001), colon cancer (P < .01), liver cancer (P < .01), and ovarian cancer (P = .002).
The incidence of several other cancers, including renal carcinoma, and rectal and endometrial cancers, was not significantly different between the groups.
The mechanisms underlying excess cancer cases in patients with obesity are not completely understood, Dr. Chittajallu said. Bariatric surgery has been shown to decrease excess inflammation, elevate insulin, and moderate hormone levels.
‘Fascinating’ study but questions remain
The study is “fascinating,” said Loren Laine, MD, moderator of the media briefing. “Obesity is clearly associated with a number of different cancers, and that’s very important. So, it makes logical sense that if you lose weight, you will reduce that risk.”
Although investigators controlled for several known cancer risk factors, there are some they couldn’t control for because they were not included in the database, and there could be unknowns that also affected the results, noted Dr. Laine, who is professor of medicine (digestive diseases) and chief of digestive health at Yale University in New Haven, Conn.
“You have to be circumspect when you look at retrospective observational studies,” he added.
It would be helpful to know when most cancers developed over the 10 years, Dr. Laine said. Dr. Chittajallu responded that the research team did not include cancers that developed in the first year after bariatric surgery to minimize incidental findings, but he did not provide a timeline for the cancers that developed.
Another unanswered question, Dr. Laine said, is whether a dose-response relationship exists. If future research shows that the more weight a person loses, the more likely they are to have a reduction in cancer risk, “that would be fascinating,” he said. Also, it would be interesting to know if endoscopic interventions and weight-loss medications decrease cancer risks in people with obesity.
More research is needed to understand how bariatric surgery affects cancer risk, Dr. Chittajallu said. “But the significant findings from this study suggest it’s an exciting avenue for further study.”
DDW 2023 will be held May 6-9 in Chicago and virtually.
The study was independently supported. Dr. Chittajallu and Dr. Laine have reported no relevant financial relationships.
The meeting is sponsored by the American Gastroenterological Association, the American Association for the Study of Liver Diseases, the American Society for Gastrointestinal Endoscopy, and the Society for Surgery of the Alimentary Tract.
A version of this article first appeared on Medscape.com.
For years evidence has pointed to multiple health benefits associated with bariatric surgery, including improvements in diabetes, sleep apnea, and blood pressure. Now researchers are adding cutting cancer risk by more than half to the list.
compared with 8.9% of their peers who did not undergo such surgery.
“We did see a difference in breast cancer, colon cancer, liver cancer, and ovarian cancer incidence. ... with patients in the bariatric surgery group having lower incidence of these four types of cancers when compared to the nonsurgical control group,” said Vibhu Chittajallu, MD, lead author and a gastroenterology fellow at Case Western Reserve University and University Hospitals in Cleveland.
The obesity epidemic is “one of the most serious health challenges in the United States today,” Dr. Chittajallu added at an April 27 media briefing during which select research was previewed for the annual Digestive Disease Week®. Obesity has been associated with multiple serious illnesses, including type 2 diabetes, heart disease, and cancer.
Obesity is also common. The Centers for Disease Control and Prevention reports that nearly 42% of American adults have obesity, and rates continue to rise.
Dr. Chittajallu and colleagues used billing codes in a national database to identify 55,789 patients with obesity who underwent bariatric surgery (sleeve gastrectomy, gastric bypass, or gastric band procedures) and a control group of the same size who did not have surgery.
Investigators controlled for risk factors that contribute to cancer development, including smoking history, alcohol use, heart disease, and hormone therapies.
Key findings
In 10 years of follow-up, 2,206 patients who underwent bariatric surgery developed an obesity-associated cancer, compared with 4,960 patients who did not have bariatric surgery.
The bariatric surgery group had lower numbers of new cases for six types of cancers (Table 1).

The differences were significant in four cancer types associated with obesity: breast cancer (P = .001), colon cancer (P < .01), liver cancer (P < .01), and ovarian cancer (P = .002).
The incidence of several other cancers, including renal carcinoma, and rectal and endometrial cancers, was not significantly different between the groups.
The mechanisms underlying excess cancer cases in patients with obesity are not completely understood, Dr. Chittajallu said. Bariatric surgery has been shown to decrease excess inflammation, elevate insulin, and moderate hormone levels.
‘Fascinating’ study but questions remain
The study is “fascinating,” said Loren Laine, MD, moderator of the media briefing. “Obesity is clearly associated with a number of different cancers, and that’s very important. So, it makes logical sense that if you lose weight, you will reduce that risk.”
Although investigators controlled for several known cancer risk factors, there are some they couldn’t control for because they were not included in the database, and there could be unknowns that also affected the results, noted Dr. Laine, who is professor of medicine (digestive diseases) and chief of digestive health at Yale University in New Haven, Conn.
“You have to be circumspect when you look at retrospective observational studies,” he added.
It would be helpful to know when most cancers developed over the 10 years, Dr. Laine said. Dr. Chittajallu responded that the research team did not include cancers that developed in the first year after bariatric surgery to minimize incidental findings, but he did not provide a timeline for the cancers that developed.
Another unanswered question, Dr. Laine said, is whether a dose-response relationship exists. If future research shows that the more weight a person loses, the more likely they are to have a reduction in cancer risk, “that would be fascinating,” he said. Also, it would be interesting to know if endoscopic interventions and weight-loss medications decrease cancer risks in people with obesity.
More research is needed to understand how bariatric surgery affects cancer risk, Dr. Chittajallu said. “But the significant findings from this study suggest it’s an exciting avenue for further study.”
DDW 2023 will be held May 6-9 in Chicago and virtually.
The study was independently supported. Dr. Chittajallu and Dr. Laine have reported no relevant financial relationships.
The meeting is sponsored by the American Gastroenterological Association, the American Association for the Study of Liver Diseases, the American Society for Gastrointestinal Endoscopy, and the Society for Surgery of the Alimentary Tract.
A version of this article first appeared on Medscape.com.
AT DDW 2023
Obesity drugs overpriced, change needed to tackle issue
The lowest available national prices of drugs to treat obesity are up to 20 times higher than the estimated cost of profitable generic versions of the same agents, according to a new analysis.
The findings by Jacob Levi, MBBS, and colleagues were published in Obesity.
“Our study highlights the inequality in pricing that exists for effective antiobesity medications, which are largely unaffordable in most countries,” Dr. Levi, from Royal Free Hospital NHS Trust, London, said in a press release.
“We show that these drugs can actually be produced and sold profitably for low prices,” he summarized. “A public health approach that prioritizes improving access to medications should be adopted, instead of allowing companies to maximize profits,” Dr. Levi urged.
Dr. Levi and colleagues studied the oral agents orlistat, naltrexone/bupropion, topiramate/phentermine, and semaglutide, and subcutaneous liraglutide, semaglutide, and tirzepatide (all approved by the U.S. Food and Drug Administration to treat obesity, except for oral semaglutide and subcutaneous tirzepatide, which are not yet approved to treat obesity in the absence of type 2 diabetes).
“Worldwide, more people are dying from diabetes and clinical obesity than HIV, tuberculosis, and malaria combined now,” senior author Andrew Hill, MD, department of pharmacology and therapeutics, University of Liverpool, England, pointed out.
We need to repeat the low-cost success story with obesity drugs
“Millions of lives have been saved by treating infectious diseases at low cost in poor countries,” Dr. Hill continued. “Now we need to repeat this medical success story, with mass treatment of diabetes and clinical obesity at low prices.”
However, in an accompanying editorial, Eric A. Finkelstein, MD, and Junxing Chay, PhD, Duke-NUS Medical School, Singapore, maintain that “It would be great if everyone had affordable access to all medicines that might improve their health. Yet that is simply not possible, nor will it ever be.”
“What is truly needed is a better way to ration the health care dollars currently available in efforts to maximize population health. That is the challenge ahead not just for [antiobesity medications] but for all treatments,” they say.
“Greater use of cost-effectiveness analysis and direct negotiations, while maintaining the patent system, represents an appropriate approach for allocating scarce health care resources in the United States and beyond,” they continue.
Lowest current patented drug prices vs. estimated generic drug prices
New medications for obesity were highly effective in recent clinical trials, but high prices limit the ability of patients to get these medications, Dr. Levi and colleagues write.
They analyzed prices for obesity drugs in 16 low-, middle-, and high-income countries: Australia, Bangladesh, China, France, Germany, India, Kenya, Morocco, Norway, Peru, Pakistan, South Africa, Turkey, the United Kingdom, the United States, and Vietnam.
The researchers assessed the price of a 30-day supply of each of the studied branded drugs based on the lowest available price (in 2021 U.S. dollars) from multiple online national price databases.
Then they calculated the estimated minimum price of a 30-day supply of a potential generic version of these drugs, which included the cost of the active medicinal ingredients, the excipients (nonactive ingredients), the prefilled injectable device plus needles (for subcutaneous drugs), transportation, 10% profit, and 27% tax on profit.
The national prices of the branded medications for obesity were significantly higher than the estimated minimum prices of potential generic drugs (see Table).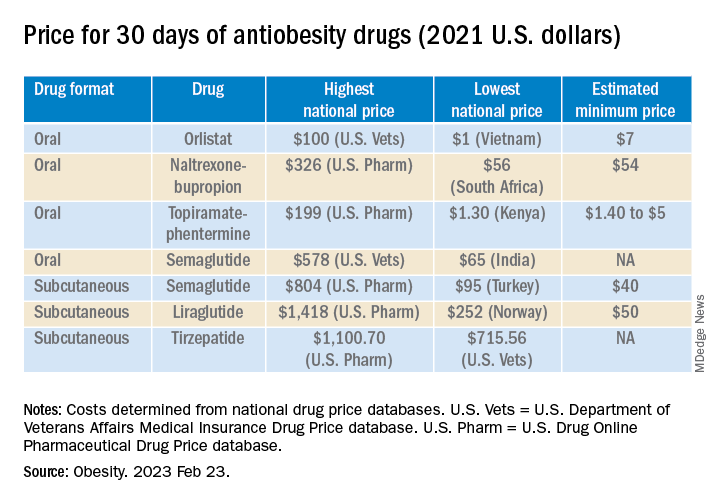
The highest national price for a branded oral drug for obesity vs. the estimated minimum price for a potential generic version was $100 vs. $7 for orlistat, $199 vs. $5 for phentermine/topiramate, and $326 vs. $54 for naltrexone/bupropion, for a 30-day supply.
There was an even greater difference between highest national branded drug price vs. estimated minimum generic drug price for the newer subcutaneously injectable drugs for obesity.
For example, the price of a 30-day course of subcutaneous semaglutide ranged from $804 (United States) to $95 (Turkey), while the estimated minimum potential generic drug price was $40 (which is 20 times lower).
The study was funded by grants from the Make Medicines Affordable/International Treatment Preparedness Coalition and from the National Heart, Lung, and Blood Institute of the National Institutes of Health. Coauthor Francois Venter has reported receiving support from the Bill and Melinda Gates Foundation, U.S. Agency for International Development, Unitaid, SA Medical Research Council, Foundation for Innovative New Diagnostics, the Children’s Investment Fund Foundation, Gilead, ViiV, Mylan, Merck, Adcock Ingram, Aspen, Abbott, Roche, Johnson & Johnson, Sanofi, Virology Education, SA HIV Clinicians Society, and Dira Sengwe. The other authors and Dr. Chay have reported no relevant financial relationships. Dr. Finkelstein has reported receiving support for serving on the WW scientific advisory board and an educational grant unrelated to the present work from Novo Nordisk.
A version of this article first appeared on Medscape.com.
The lowest available national prices of drugs to treat obesity are up to 20 times higher than the estimated cost of profitable generic versions of the same agents, according to a new analysis.
The findings by Jacob Levi, MBBS, and colleagues were published in Obesity.
“Our study highlights the inequality in pricing that exists for effective antiobesity medications, which are largely unaffordable in most countries,” Dr. Levi, from Royal Free Hospital NHS Trust, London, said in a press release.
“We show that these drugs can actually be produced and sold profitably for low prices,” he summarized. “A public health approach that prioritizes improving access to medications should be adopted, instead of allowing companies to maximize profits,” Dr. Levi urged.
Dr. Levi and colleagues studied the oral agents orlistat, naltrexone/bupropion, topiramate/phentermine, and semaglutide, and subcutaneous liraglutide, semaglutide, and tirzepatide (all approved by the U.S. Food and Drug Administration to treat obesity, except for oral semaglutide and subcutaneous tirzepatide, which are not yet approved to treat obesity in the absence of type 2 diabetes).
“Worldwide, more people are dying from diabetes and clinical obesity than HIV, tuberculosis, and malaria combined now,” senior author Andrew Hill, MD, department of pharmacology and therapeutics, University of Liverpool, England, pointed out.
We need to repeat the low-cost success story with obesity drugs
“Millions of lives have been saved by treating infectious diseases at low cost in poor countries,” Dr. Hill continued. “Now we need to repeat this medical success story, with mass treatment of diabetes and clinical obesity at low prices.”
However, in an accompanying editorial, Eric A. Finkelstein, MD, and Junxing Chay, PhD, Duke-NUS Medical School, Singapore, maintain that “It would be great if everyone had affordable access to all medicines that might improve their health. Yet that is simply not possible, nor will it ever be.”
“What is truly needed is a better way to ration the health care dollars currently available in efforts to maximize population health. That is the challenge ahead not just for [antiobesity medications] but for all treatments,” they say.
“Greater use of cost-effectiveness analysis and direct negotiations, while maintaining the patent system, represents an appropriate approach for allocating scarce health care resources in the United States and beyond,” they continue.
Lowest current patented drug prices vs. estimated generic drug prices
New medications for obesity were highly effective in recent clinical trials, but high prices limit the ability of patients to get these medications, Dr. Levi and colleagues write.
They analyzed prices for obesity drugs in 16 low-, middle-, and high-income countries: Australia, Bangladesh, China, France, Germany, India, Kenya, Morocco, Norway, Peru, Pakistan, South Africa, Turkey, the United Kingdom, the United States, and Vietnam.
The researchers assessed the price of a 30-day supply of each of the studied branded drugs based on the lowest available price (in 2021 U.S. dollars) from multiple online national price databases.
Then they calculated the estimated minimum price of a 30-day supply of a potential generic version of these drugs, which included the cost of the active medicinal ingredients, the excipients (nonactive ingredients), the prefilled injectable device plus needles (for subcutaneous drugs), transportation, 10% profit, and 27% tax on profit.
The national prices of the branded medications for obesity were significantly higher than the estimated minimum prices of potential generic drugs (see Table).
The highest national price for a branded oral drug for obesity vs. the estimated minimum price for a potential generic version was $100 vs. $7 for orlistat, $199 vs. $5 for phentermine/topiramate, and $326 vs. $54 for naltrexone/bupropion, for a 30-day supply.
There was an even greater difference between highest national branded drug price vs. estimated minimum generic drug price for the newer subcutaneously injectable drugs for obesity.
For example, the price of a 30-day course of subcutaneous semaglutide ranged from $804 (United States) to $95 (Turkey), while the estimated minimum potential generic drug price was $40 (which is 20 times lower).
The study was funded by grants from the Make Medicines Affordable/International Treatment Preparedness Coalition and from the National Heart, Lung, and Blood Institute of the National Institutes of Health. Coauthor Francois Venter has reported receiving support from the Bill and Melinda Gates Foundation, U.S. Agency for International Development, Unitaid, SA Medical Research Council, Foundation for Innovative New Diagnostics, the Children’s Investment Fund Foundation, Gilead, ViiV, Mylan, Merck, Adcock Ingram, Aspen, Abbott, Roche, Johnson & Johnson, Sanofi, Virology Education, SA HIV Clinicians Society, and Dira Sengwe. The other authors and Dr. Chay have reported no relevant financial relationships. Dr. Finkelstein has reported receiving support for serving on the WW scientific advisory board and an educational grant unrelated to the present work from Novo Nordisk.
A version of this article first appeared on Medscape.com.
The lowest available national prices of drugs to treat obesity are up to 20 times higher than the estimated cost of profitable generic versions of the same agents, according to a new analysis.
The findings by Jacob Levi, MBBS, and colleagues were published in Obesity.
“Our study highlights the inequality in pricing that exists for effective antiobesity medications, which are largely unaffordable in most countries,” Dr. Levi, from Royal Free Hospital NHS Trust, London, said in a press release.
“We show that these drugs can actually be produced and sold profitably for low prices,” he summarized. “A public health approach that prioritizes improving access to medications should be adopted, instead of allowing companies to maximize profits,” Dr. Levi urged.
Dr. Levi and colleagues studied the oral agents orlistat, naltrexone/bupropion, topiramate/phentermine, and semaglutide, and subcutaneous liraglutide, semaglutide, and tirzepatide (all approved by the U.S. Food and Drug Administration to treat obesity, except for oral semaglutide and subcutaneous tirzepatide, which are not yet approved to treat obesity in the absence of type 2 diabetes).
“Worldwide, more people are dying from diabetes and clinical obesity than HIV, tuberculosis, and malaria combined now,” senior author Andrew Hill, MD, department of pharmacology and therapeutics, University of Liverpool, England, pointed out.
We need to repeat the low-cost success story with obesity drugs
“Millions of lives have been saved by treating infectious diseases at low cost in poor countries,” Dr. Hill continued. “Now we need to repeat this medical success story, with mass treatment of diabetes and clinical obesity at low prices.”
However, in an accompanying editorial, Eric A. Finkelstein, MD, and Junxing Chay, PhD, Duke-NUS Medical School, Singapore, maintain that “It would be great if everyone had affordable access to all medicines that might improve their health. Yet that is simply not possible, nor will it ever be.”
“What is truly needed is a better way to ration the health care dollars currently available in efforts to maximize population health. That is the challenge ahead not just for [antiobesity medications] but for all treatments,” they say.
“Greater use of cost-effectiveness analysis and direct negotiations, while maintaining the patent system, represents an appropriate approach for allocating scarce health care resources in the United States and beyond,” they continue.
Lowest current patented drug prices vs. estimated generic drug prices
New medications for obesity were highly effective in recent clinical trials, but high prices limit the ability of patients to get these medications, Dr. Levi and colleagues write.
They analyzed prices for obesity drugs in 16 low-, middle-, and high-income countries: Australia, Bangladesh, China, France, Germany, India, Kenya, Morocco, Norway, Peru, Pakistan, South Africa, Turkey, the United Kingdom, the United States, and Vietnam.
The researchers assessed the price of a 30-day supply of each of the studied branded drugs based on the lowest available price (in 2021 U.S. dollars) from multiple online national price databases.
Then they calculated the estimated minimum price of a 30-day supply of a potential generic version of these drugs, which included the cost of the active medicinal ingredients, the excipients (nonactive ingredients), the prefilled injectable device plus needles (for subcutaneous drugs), transportation, 10% profit, and 27% tax on profit.
The national prices of the branded medications for obesity were significantly higher than the estimated minimum prices of potential generic drugs (see Table).
The highest national price for a branded oral drug for obesity vs. the estimated minimum price for a potential generic version was $100 vs. $7 for orlistat, $199 vs. $5 for phentermine/topiramate, and $326 vs. $54 for naltrexone/bupropion, for a 30-day supply.
There was an even greater difference between highest national branded drug price vs. estimated minimum generic drug price for the newer subcutaneously injectable drugs for obesity.
For example, the price of a 30-day course of subcutaneous semaglutide ranged from $804 (United States) to $95 (Turkey), while the estimated minimum potential generic drug price was $40 (which is 20 times lower).
The study was funded by grants from the Make Medicines Affordable/International Treatment Preparedness Coalition and from the National Heart, Lung, and Blood Institute of the National Institutes of Health. Coauthor Francois Venter has reported receiving support from the Bill and Melinda Gates Foundation, U.S. Agency for International Development, Unitaid, SA Medical Research Council, Foundation for Innovative New Diagnostics, the Children’s Investment Fund Foundation, Gilead, ViiV, Mylan, Merck, Adcock Ingram, Aspen, Abbott, Roche, Johnson & Johnson, Sanofi, Virology Education, SA HIV Clinicians Society, and Dira Sengwe. The other authors and Dr. Chay have reported no relevant financial relationships. Dr. Finkelstein has reported receiving support for serving on the WW scientific advisory board and an educational grant unrelated to the present work from Novo Nordisk.
A version of this article first appeared on Medscape.com.
Tirzepatide scores win in second obesity trial, SURMOUNT-2
The “twincretin” tirzepatide (Mounjaro) has proven successful in SURMOUNT-2, the second pivotal trial for the drug as an antiobesity agent, according to top-line results reported April 27 by tirzepatide’s manufacturer, Lilly, in a press release. The company reveals that tirzepatide achieved both of its primary endpoints in the trial, as well as all its key secondary endpoints.
The findings pave the way for tirzepatide to likely receive Food and Drug Administration approval as a treatment for obesity, perhaps before the end of 2023.
Tirzepatide received FDA approval in May 2022 for the treatment of type 2 diabetes in adults, under the brand name Mounjaro, and some people have already been using it off-label to treat obesity.
Tirzepatide is a dual glucagonlike peptide–1 (GLP-1) agonist and glucose-dependent insulinotropic polypeptide agonist. Several GLP-1 receptor agonists are already approved in the United States, including semaglutide, a once-weekly injection, which is approved as Wegovy for patients with obesity and as Ozempic for treatment of type 2 diabetes.
These agents have been incredibly popular among celebrity influencers, and with use of the #Ozempic hashtag and others on social media, this has led to unprecedented use of these products for weight loss, often among those who do not even have obesity or type 2 diabetes. Subsequently, patients with type 2 diabetes and obesity who need them have often struggled to obtain them, owing to shortages following this phenomenon.
SURMOUNT-2: Weight loss around 15%, less than seen in SURMOUNT-1
SURMOUNT-2 enrolled 938 adults with overweight or obesity and type 2 diabetes and had dual primary endpoints that both focused on weight loss, compared with placebo.
The first completed pivotal trial of tirzepatide for weight loss, SURMOUNT-1, enrolled people with overweight or obesity but no diabetes and had its main results reported in 2022. At the time, the weight loss achieved with tirzepatide, was described as “unprecedented,” with those given the highest dose in that trial (15 mg subcutaneously per week) losing an average of 20%-22% of body weight over 72 weeks, depending on the specific statistical analysis used.
For SURMOUNT-2’s first primary endpoint, 72 weeks of weekly subcutaneous injections with tirzepatide at dosages of 10 mg or 15 mg led to an average weight loss from baseline of 13.4% and 15.7%, respectively, compared with an average loss of 3.3% from baseline in the placebo-treated control arm.
For the second primary endpoint, 81.6% of people on the 10-mg dose and 86.4% on the 15-mg dose achieved at least 5% weight loss from baseline, compared with 30.5% of controls who had at least 5% weight loss from baseline.
In one key secondary endpoint, tirzepatide at dosages of 10 mg or 15 mg weekly produced at least a 15% cut in weight from baseline in 41.4% and 51.8% of participants, respectively, compared with a 2.6% rate of this endpoint in the placebo controls.
So the extent of weight loss seen in in SURMOUNT-2 was somewhat less than was reported in SURMOUNT-1, a finding consistent with many prior studies of incretin-based weight-loss agents, which seem to pack a more potent weight-loss punch in people without type 2 diabetes.
Lilly did not specifically report the treatment effect of tirzepatide on hemoglobin A1c in SURMOUNT-2, only saying that the effect was similar to what had been seen in the series of five SURPASS trials that led to the approval of tirzepatide for type 2 diabetes.
Lilly also reported that the safety profile of tirzepatide in SURMOUNT-2 generally matched what was seen in SURMOUNT-1 as well as in the SURPASS trials. The most common adverse events in SURMOUNT-2 involved gastrointestinal symptoms, such as nausea, diarrhea, and vomiting; these were generally mild to moderate in severity and clustered during the dose-escalation phase at the start of treatment. Treatment discontinuations caused by adverse effects were 3.8% on the 10-mg dosage, 7.4% on the 15-mg dosage, and 3.8% on placebo.
SURMOUNT-2 enrolled patients in the United States, Puerto Rico, and five other countries. All participants also received interventions designed to reduce their calorie intake and increase their physical activity.
More SURMOUNT-2 results at ADA in June
Lilly also announced that researchers would report more complete results from SURMOUNT-2 at the 2023 scientific sessions of the American Diabetes Association, being held in San Diego in late June, and publish the findings in a major medical journal.
Results from two additional phase 3 trials of tirzepatide in people with overweight or obesity, SURMOUNT-3 and SURMOUNT-4, are expected later in 2023.
Lilly started an application to the FDA for an indication for weight loss in October 2022 under a fast track designation by the agency, and the data collected in SURMOUNT-2 are expected to complete this application, which would then be subject to an FDA decision within about 6 months. Lilly said in its April 27 press release that it anticipates an FDA decision on this application may occur before the end of 2023.
SURMOUNT-2 and all of the other tirzepatide trials were sponsored by Lilly.
A version of this article first appeared on Medscape.com.
The “twincretin” tirzepatide (Mounjaro) has proven successful in SURMOUNT-2, the second pivotal trial for the drug as an antiobesity agent, according to top-line results reported April 27 by tirzepatide’s manufacturer, Lilly, in a press release. The company reveals that tirzepatide achieved both of its primary endpoints in the trial, as well as all its key secondary endpoints.
The findings pave the way for tirzepatide to likely receive Food and Drug Administration approval as a treatment for obesity, perhaps before the end of 2023.
Tirzepatide received FDA approval in May 2022 for the treatment of type 2 diabetes in adults, under the brand name Mounjaro, and some people have already been using it off-label to treat obesity.
Tirzepatide is a dual glucagonlike peptide–1 (GLP-1) agonist and glucose-dependent insulinotropic polypeptide agonist. Several GLP-1 receptor agonists are already approved in the United States, including semaglutide, a once-weekly injection, which is approved as Wegovy for patients with obesity and as Ozempic for treatment of type 2 diabetes.
These agents have been incredibly popular among celebrity influencers, and with use of the #Ozempic hashtag and others on social media, this has led to unprecedented use of these products for weight loss, often among those who do not even have obesity or type 2 diabetes. Subsequently, patients with type 2 diabetes and obesity who need them have often struggled to obtain them, owing to shortages following this phenomenon.
SURMOUNT-2: Weight loss around 15%, less than seen in SURMOUNT-1
SURMOUNT-2 enrolled 938 adults with overweight or obesity and type 2 diabetes and had dual primary endpoints that both focused on weight loss, compared with placebo.
The first completed pivotal trial of tirzepatide for weight loss, SURMOUNT-1, enrolled people with overweight or obesity but no diabetes and had its main results reported in 2022. At the time, the weight loss achieved with tirzepatide, was described as “unprecedented,” with those given the highest dose in that trial (15 mg subcutaneously per week) losing an average of 20%-22% of body weight over 72 weeks, depending on the specific statistical analysis used.
For SURMOUNT-2’s first primary endpoint, 72 weeks of weekly subcutaneous injections with tirzepatide at dosages of 10 mg or 15 mg led to an average weight loss from baseline of 13.4% and 15.7%, respectively, compared with an average loss of 3.3% from baseline in the placebo-treated control arm.
For the second primary endpoint, 81.6% of people on the 10-mg dose and 86.4% on the 15-mg dose achieved at least 5% weight loss from baseline, compared with 30.5% of controls who had at least 5% weight loss from baseline.
In one key secondary endpoint, tirzepatide at dosages of 10 mg or 15 mg weekly produced at least a 15% cut in weight from baseline in 41.4% and 51.8% of participants, respectively, compared with a 2.6% rate of this endpoint in the placebo controls.
So the extent of weight loss seen in in SURMOUNT-2 was somewhat less than was reported in SURMOUNT-1, a finding consistent with many prior studies of incretin-based weight-loss agents, which seem to pack a more potent weight-loss punch in people without type 2 diabetes.
Lilly did not specifically report the treatment effect of tirzepatide on hemoglobin A1c in SURMOUNT-2, only saying that the effect was similar to what had been seen in the series of five SURPASS trials that led to the approval of tirzepatide for type 2 diabetes.
Lilly also reported that the safety profile of tirzepatide in SURMOUNT-2 generally matched what was seen in SURMOUNT-1 as well as in the SURPASS trials. The most common adverse events in SURMOUNT-2 involved gastrointestinal symptoms, such as nausea, diarrhea, and vomiting; these were generally mild to moderate in severity and clustered during the dose-escalation phase at the start of treatment. Treatment discontinuations caused by adverse effects were 3.8% on the 10-mg dosage, 7.4% on the 15-mg dosage, and 3.8% on placebo.
SURMOUNT-2 enrolled patients in the United States, Puerto Rico, and five other countries. All participants also received interventions designed to reduce their calorie intake and increase their physical activity.
More SURMOUNT-2 results at ADA in June
Lilly also announced that researchers would report more complete results from SURMOUNT-2 at the 2023 scientific sessions of the American Diabetes Association, being held in San Diego in late June, and publish the findings in a major medical journal.
Results from two additional phase 3 trials of tirzepatide in people with overweight or obesity, SURMOUNT-3 and SURMOUNT-4, are expected later in 2023.
Lilly started an application to the FDA for an indication for weight loss in October 2022 under a fast track designation by the agency, and the data collected in SURMOUNT-2 are expected to complete this application, which would then be subject to an FDA decision within about 6 months. Lilly said in its April 27 press release that it anticipates an FDA decision on this application may occur before the end of 2023.
SURMOUNT-2 and all of the other tirzepatide trials were sponsored by Lilly.
A version of this article first appeared on Medscape.com.
The “twincretin” tirzepatide (Mounjaro) has proven successful in SURMOUNT-2, the second pivotal trial for the drug as an antiobesity agent, according to top-line results reported April 27 by tirzepatide’s manufacturer, Lilly, in a press release. The company reveals that tirzepatide achieved both of its primary endpoints in the trial, as well as all its key secondary endpoints.
The findings pave the way for tirzepatide to likely receive Food and Drug Administration approval as a treatment for obesity, perhaps before the end of 2023.
Tirzepatide received FDA approval in May 2022 for the treatment of type 2 diabetes in adults, under the brand name Mounjaro, and some people have already been using it off-label to treat obesity.
Tirzepatide is a dual glucagonlike peptide–1 (GLP-1) agonist and glucose-dependent insulinotropic polypeptide agonist. Several GLP-1 receptor agonists are already approved in the United States, including semaglutide, a once-weekly injection, which is approved as Wegovy for patients with obesity and as Ozempic for treatment of type 2 diabetes.
These agents have been incredibly popular among celebrity influencers, and with use of the #Ozempic hashtag and others on social media, this has led to unprecedented use of these products for weight loss, often among those who do not even have obesity or type 2 diabetes. Subsequently, patients with type 2 diabetes and obesity who need them have often struggled to obtain them, owing to shortages following this phenomenon.
SURMOUNT-2: Weight loss around 15%, less than seen in SURMOUNT-1
SURMOUNT-2 enrolled 938 adults with overweight or obesity and type 2 diabetes and had dual primary endpoints that both focused on weight loss, compared with placebo.
The first completed pivotal trial of tirzepatide for weight loss, SURMOUNT-1, enrolled people with overweight or obesity but no diabetes and had its main results reported in 2022. At the time, the weight loss achieved with tirzepatide, was described as “unprecedented,” with those given the highest dose in that trial (15 mg subcutaneously per week) losing an average of 20%-22% of body weight over 72 weeks, depending on the specific statistical analysis used.
For SURMOUNT-2’s first primary endpoint, 72 weeks of weekly subcutaneous injections with tirzepatide at dosages of 10 mg or 15 mg led to an average weight loss from baseline of 13.4% and 15.7%, respectively, compared with an average loss of 3.3% from baseline in the placebo-treated control arm.
For the second primary endpoint, 81.6% of people on the 10-mg dose and 86.4% on the 15-mg dose achieved at least 5% weight loss from baseline, compared with 30.5% of controls who had at least 5% weight loss from baseline.
In one key secondary endpoint, tirzepatide at dosages of 10 mg or 15 mg weekly produced at least a 15% cut in weight from baseline in 41.4% and 51.8% of participants, respectively, compared with a 2.6% rate of this endpoint in the placebo controls.
So the extent of weight loss seen in in SURMOUNT-2 was somewhat less than was reported in SURMOUNT-1, a finding consistent with many prior studies of incretin-based weight-loss agents, which seem to pack a more potent weight-loss punch in people without type 2 diabetes.
Lilly did not specifically report the treatment effect of tirzepatide on hemoglobin A1c in SURMOUNT-2, only saying that the effect was similar to what had been seen in the series of five SURPASS trials that led to the approval of tirzepatide for type 2 diabetes.
Lilly also reported that the safety profile of tirzepatide in SURMOUNT-2 generally matched what was seen in SURMOUNT-1 as well as in the SURPASS trials. The most common adverse events in SURMOUNT-2 involved gastrointestinal symptoms, such as nausea, diarrhea, and vomiting; these were generally mild to moderate in severity and clustered during the dose-escalation phase at the start of treatment. Treatment discontinuations caused by adverse effects were 3.8% on the 10-mg dosage, 7.4% on the 15-mg dosage, and 3.8% on placebo.
SURMOUNT-2 enrolled patients in the United States, Puerto Rico, and five other countries. All participants also received interventions designed to reduce their calorie intake and increase their physical activity.
More SURMOUNT-2 results at ADA in June
Lilly also announced that researchers would report more complete results from SURMOUNT-2 at the 2023 scientific sessions of the American Diabetes Association, being held in San Diego in late June, and publish the findings in a major medical journal.
Results from two additional phase 3 trials of tirzepatide in people with overweight or obesity, SURMOUNT-3 and SURMOUNT-4, are expected later in 2023.
Lilly started an application to the FDA for an indication for weight loss in October 2022 under a fast track designation by the agency, and the data collected in SURMOUNT-2 are expected to complete this application, which would then be subject to an FDA decision within about 6 months. Lilly said in its April 27 press release that it anticipates an FDA decision on this application may occur before the end of 2023.
SURMOUNT-2 and all of the other tirzepatide trials were sponsored by Lilly.
A version of this article first appeared on Medscape.com.
BMI is a flawed measure of obesity. What are alternatives?
“BMI is trash. Full stop.” This controversial tweet, which received thousands of likes and retweets, was cited in a recent article by one doctor on when physicians might stop using body mass index (BMI) to diagnose obesity.
BMI has for years been the consensus default method for assessing whether a person is overweight or has obesity, and is still widely used as the gatekeeper metric for treatment eligibility for certain weight-loss agents and bariatric surgery.
an important determinant of the cardiometabolic consequences of fat.
Alternative metrics include waist circumference and/or waist-to-height ratio (WHtR); imaging methods such as CT, MRI, and dual-energy x-ray absorptiometry (DXA); and bioelectrical impedance to assess fat volume and location. All have made some inroads on the tight grip BMI has had on obesity assessment.
Chances are, however, that BMI will not fade away anytime soon given how entrenched it has become in clinical practice and for insurance coverage, as well as its relative simplicity and precision.
“BMI is embedded in a wide range of guidelines on the use of medications and surgery. It’s embedded in Food and Drug Administration regulations and for billing and insurance coverage. It would take extremely strong data and years of work to undo the infrastructure built around BMI and replace it with something else. I don’t see that happening [anytime soon],” commented Daniel H. Bessesen, MD, a professor at the University of Colorado at Denver, Aurora, and chief of endocrinology for Denver Health.
“It would be almost impossible to replace all the studies that have used BMI with investigations using some other measure,” he said.
BMI Is ‘imperfect’
The entrenched position of BMI as the go-to metric doesn’t keep detractors from weighing in. As noted in a commentary on current clinical challenges surrounding obesity recently published in Annals of Internal Medicine, the journal’s editor-in-chief, Christine Laine, MD, and senior deputy editor Christina C. Wee, MD, listed six top issues clinicians must deal with, one of which, they say, is the need for a better measure of obesity than BMI.
“Unfortunately, BMI is an imperfect measure of body composition that differs with ethnicity, sex, body frame, and muscle mass,” noted Dr. Laine and Dr. Wee.
BMI is based on a person’s weight in kilograms divided by the square of their height in meters. A “healthy” BMI is between 18.5 and 24.9 kg/m2, overweight is 25-29.9, and 30 or greater is considered to represent obesity. However, certain ethnic groups have lower cutoffs for overweight or obesity because of evidence that such individuals can be at higher risk of obesity-related comorbidities at lower BMIs.
“BMI was chosen as the initial screening tool [for obesity] not because anyone thought it was perfect or the best measure but because of its simplicity. All you need is height, weight, and a calculator,” Dr. Wee said in an interview.
Numerous online calculators are available, including one from the Centers for Disease Control and Prevention where height in feet and inches and weight in pounds can be entered to generate the BMI.
BMI is also inherently limited by being “a proxy for adiposity” and not a direct measure, added Dr. Wee, who is also director of the Obesity Research Program of Beth Israel Deaconess Medical Center, Boston.
As such, BMI can’t distinguish between fat and muscle because it relies on weight only to gauge adiposity, noted Tiffany Powell-Wiley, MD, an obesity researcher at the National Heart, Lung, and Blood Institute in Bethesda, Md. Another shortcoming of BMI is that it “is good for distinguishing population-level risk for cardiovascular disease and other chronic diseases, but it does not help as much for distinguishing risk at an individual level,” she said in an interview.
These and other drawbacks have prompted researchers to look for other useful metrics. WHtR, for example, has recently made headway as a potential BMI alternative or complement.
The case for WHtR
Concern about overreliance on BMI despite its limitations is not new. In 2015, an American Heart Association scientific statement from the group’s Obesity Committee concluded that “BMI alone, even with lower thresholds, is a useful but not an ideal tool for identification of obesity or assessment of cardiovascular risk,” especially for people from Asian, Black, Hispanic, and Pacific Islander populations.
The writing panel also recommended that clinicians measure waist circumference annually and use that information along with BMI “to better gauge cardiovascular risk in diverse populations.”
Momentum for moving beyond BMI alone has continued to build following the AHA statement.
In September 2022, the National Institute for Health and Care Excellence, which sets policies for the United Kingdom’s National Health Service, revised its guidancefor assessment and management of people with obesity. The updated guidance recommends that when clinicians assess “adults with BMI below 35 kg/m2, measure and use their WHtR, as well as their BMI, as a practical estimate of central adiposity and use these measurements to help to assess and predict health risks.”
NICE released an extensive literature review with the revision, and based on the evidence, said that “using waist-to-height ratio as well as BMI would help give a practical estimate of central adiposity in adults with BMI under 35 kg/m2. This would in turn help professionals assess and predict health risks.”
However, the review added that, “because people with a BMI over 35 kg/m2 are always likely to have a high WHtR, the committee recognized that it may not be a useful addition for predicting health risks in this group.” The 2022 NICE review also said that it is “important to estimate central adiposity when assessing future health risks, including for people whose BMI is in the healthy-weight category.”
This new emphasis by NICE on measuring and using WHtR as part of obesity assessment “represents an important change in population health policy,” commented Dr. Powell-Wiley. “I expect more professional organizations will endorse use of waist circumference or waist-to-height ratio now that NICE has taken this step,” she predicted.
Waist circumference and WHtR may become standard measures of adiposity in clinical practice over the next 5-10 years.
The recent move by NICE to highlight a complementary role for WHtR “is another acknowledgment that BMI is an imperfect tool for stratifying cardiometabolic risk in a diverse population, especially in people with lower BMIs” because of its variability, commented Jamie Almandoz, MD, medical director of the weight wellness program at UT Southwestern Medical Center, Dallas.
WHtR vs. BMI
Another recent step forward for WHtR came with the publication of a post hoc analysis of data collected in the PARADIGM-HF trial, a study that had the primary purpose of comparing two medications for improving outcomes in more than 8,000 patients with heart failure with reduced ejection fraction.
The new analysis showed that “two indices that incorporate waist circumference and height, but not weight, showed a clearer association between greater adiposity and a higher risk of heart failure hospitalization,” compared with BMI.
WHtR was one of the two indices identified as being a better correlate for the adverse effect of excess adiposity compared with BMI.
The authors of the post hoc analysis did not design their analysis to compare WHtR with BMI. Instead, their goal was to better understand what’s known as the “obesity paradox” in people with heart failure with reduced ejection fraction: The recurring observation that, when these patients with heart failure have lower BMIs they fare worse, with higher rates of mortality and adverse cardiovascular outcomes, compared with patients with higher BMIs.
The new analysis showed that this paradox disappeared when WHtR was substituted for BMI as the obesity metric.
This “provides meaningful data about the superiority of WHtR, compared with BMI, for predicting heart failure outcomes,” said Dr. Powell-Wiley, although she cautioned that the analysis was limited by scant data in diverse populations and did not look at other important cardiovascular disease outcomes. While Dr. Powell-Wiley does not think that WHtR needs assessment in a prospective, controlled trial, she called for analysis of pooled prospective studies with more diverse populations to better document the advantages of WHtR over BMI.
The PARADIGM-HF post hoc analysis shows again how flawed BMI is for health assessment and the relative importance of an individualized understanding of a person’s body composition, Dr. Almandoz said in an interview. “As we collect more data, there is increasing awareness of how imperfect BMI is.”
Measuring waist circumference is tricky
Although WHtR looks promising as a substitute for or add-on to BMI, it has its own limitations, particularly the challenge of accurately measuring waist circumference.
Measuring waist circumference “not only takes more time but requires the assessor to be well trained about where to put the tape measure and making sure it’s measured at the same place each time,” even when different people take serial measurements from individual patients, noted Dr. Wee. Determining waist circumference can also be technically difficult when done on larger people, she added, and collectively these challenges make waist circumference “less reproducible from measurement to measurement.”
“It’s relatively clear how to standardize measurement of weight and height, but there is a huge amount of variability when the waist is measured,” agreed Dr. Almandoz. “And waist circumference also differs by ethnicity, race, sex, and body frame. There are significant differences in waist circumference levels that associate with increased health risks” between, for example, White and South Asian people.
Another limitation of waist circumference and WHtR is that they “cannot differentiate between visceral and abdominal subcutaneous adipose tissue, which are vastly different regarding cardiometabolic risk, commented Ian Neeland, MD, director of cardiovascular prevention at the University Hospitals Harrington Heart & Vascular Institute, Cleveland.
The imaging option
“Waist-to-height ratio is not the ultimate answer,” Dr. Neeland said in an interview. He instead endorsed “advanced imaging for body fat distribution,” such as CT or MRI scans, as his pick for what should be the standard obesity metric, “given that it is much more specific and actionable for both risk assessment and response to therapy. I expect slow but steady advancements that move away from BMI cutoffs, for example for bariatric surgery, given that BMI is an imprecise and crude tool.”
But although imaging with methods like CT and MRI may provide the best accuracy and precision for tracking the volume of a person’s cardiometabolically dangerous fat, they are also hampered by relatively high cost and, for CT and DXA, the issue of radiation exposure.
“CT, MRI, and DXA scans give more in-depth assessment of body composition, but should we expose people to the radiation and the cost?” Dr. Almandoz wondered.
“Height, weight, and waist circumference cost nothing to obtain,” creating a big relative disadvantage for imaging, said Naveed Sattar, MD, professor of metabolic medicine at the University of Glasgow.
“Data would need to show that imaging gives clinicians substantially more information about future risk” to justify its price, Dr. Sattar emphasized.
BMI’s limits mean adding on
Regardless of whichever alternatives to BMI end up getting used most, experts generally agree that BMI alone is looking increasingly inadequate.
“Over the next 5 years, BMI will come to be seen as a screening tool that categorizes people into general risk groups” that also needs “other metrics and variables, such as age, race, ethnicity, family history, blood glucose, and blood pressure to better describe health risk in an individual,” predicted Dr. Bessesen.
The endorsement of WHtR by NICE “will lead to more research into how to incorporate WHtR into routine practice. We need more evidence to translate what NICE said into practice,” said Dr. Sattar. “I don’t think we’ll see a shift away from BMI, but we’ll add alternative measures that are particularly useful in certain patients.”
“Because we live in diverse societies, we need to individualize risk assessment and couple that with technology that makes analysis of body composition more accessible,” agreed Dr. Almandoz. He noted that the UT Southwestern weight wellness program where he practices has, for about the past decade, routinely collected waist circumference and bioelectrical impedance data as well as BMI on all people seen in the practice for obesity concerns. Making these additional measurements on a routine basis also helps strengthen patient engagement.
“We get into trouble when we make rigid health policy and clinical decisions based on BMI alone without looking at the patient holistically,” said Dr. Wee. “Patients are more than arbitrary numbers, and clinicians should make clinical decisions based on the totality of evidence for each individual patient.”
Dr. Bessesen, Dr. Wee, Dr. Powell-Wiley, and Dr. Almandoz reported no relevant financial relationships. Dr. Neeland has reported being a consultant for Merck. Dr. Sattar has reported being a consultant or speaker for Abbott Laboratories, Afimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceuticals, Janssen, MSD, Novartis, Novo Nordisk, Pfizer, Roche Diagnostics, and Sanofi.
A version of this article originally appeared on Medscape.com.
“BMI is trash. Full stop.” This controversial tweet, which received thousands of likes and retweets, was cited in a recent article by one doctor on when physicians might stop using body mass index (BMI) to diagnose obesity.
BMI has for years been the consensus default method for assessing whether a person is overweight or has obesity, and is still widely used as the gatekeeper metric for treatment eligibility for certain weight-loss agents and bariatric surgery.
an important determinant of the cardiometabolic consequences of fat.
Alternative metrics include waist circumference and/or waist-to-height ratio (WHtR); imaging methods such as CT, MRI, and dual-energy x-ray absorptiometry (DXA); and bioelectrical impedance to assess fat volume and location. All have made some inroads on the tight grip BMI has had on obesity assessment.
Chances are, however, that BMI will not fade away anytime soon given how entrenched it has become in clinical practice and for insurance coverage, as well as its relative simplicity and precision.
“BMI is embedded in a wide range of guidelines on the use of medications and surgery. It’s embedded in Food and Drug Administration regulations and for billing and insurance coverage. It would take extremely strong data and years of work to undo the infrastructure built around BMI and replace it with something else. I don’t see that happening [anytime soon],” commented Daniel H. Bessesen, MD, a professor at the University of Colorado at Denver, Aurora, and chief of endocrinology for Denver Health.
“It would be almost impossible to replace all the studies that have used BMI with investigations using some other measure,” he said.
BMI Is ‘imperfect’
The entrenched position of BMI as the go-to metric doesn’t keep detractors from weighing in. As noted in a commentary on current clinical challenges surrounding obesity recently published in Annals of Internal Medicine, the journal’s editor-in-chief, Christine Laine, MD, and senior deputy editor Christina C. Wee, MD, listed six top issues clinicians must deal with, one of which, they say, is the need for a better measure of obesity than BMI.
“Unfortunately, BMI is an imperfect measure of body composition that differs with ethnicity, sex, body frame, and muscle mass,” noted Dr. Laine and Dr. Wee.
BMI is based on a person’s weight in kilograms divided by the square of their height in meters. A “healthy” BMI is between 18.5 and 24.9 kg/m2, overweight is 25-29.9, and 30 or greater is considered to represent obesity. However, certain ethnic groups have lower cutoffs for overweight or obesity because of evidence that such individuals can be at higher risk of obesity-related comorbidities at lower BMIs.
“BMI was chosen as the initial screening tool [for obesity] not because anyone thought it was perfect or the best measure but because of its simplicity. All you need is height, weight, and a calculator,” Dr. Wee said in an interview.
Numerous online calculators are available, including one from the Centers for Disease Control and Prevention where height in feet and inches and weight in pounds can be entered to generate the BMI.
BMI is also inherently limited by being “a proxy for adiposity” and not a direct measure, added Dr. Wee, who is also director of the Obesity Research Program of Beth Israel Deaconess Medical Center, Boston.
As such, BMI can’t distinguish between fat and muscle because it relies on weight only to gauge adiposity, noted Tiffany Powell-Wiley, MD, an obesity researcher at the National Heart, Lung, and Blood Institute in Bethesda, Md. Another shortcoming of BMI is that it “is good for distinguishing population-level risk for cardiovascular disease and other chronic diseases, but it does not help as much for distinguishing risk at an individual level,” she said in an interview.
These and other drawbacks have prompted researchers to look for other useful metrics. WHtR, for example, has recently made headway as a potential BMI alternative or complement.
The case for WHtR
Concern about overreliance on BMI despite its limitations is not new. In 2015, an American Heart Association scientific statement from the group’s Obesity Committee concluded that “BMI alone, even with lower thresholds, is a useful but not an ideal tool for identification of obesity or assessment of cardiovascular risk,” especially for people from Asian, Black, Hispanic, and Pacific Islander populations.
The writing panel also recommended that clinicians measure waist circumference annually and use that information along with BMI “to better gauge cardiovascular risk in diverse populations.”
Momentum for moving beyond BMI alone has continued to build following the AHA statement.
In September 2022, the National Institute for Health and Care Excellence, which sets policies for the United Kingdom’s National Health Service, revised its guidancefor assessment and management of people with obesity. The updated guidance recommends that when clinicians assess “adults with BMI below 35 kg/m2, measure and use their WHtR, as well as their BMI, as a practical estimate of central adiposity and use these measurements to help to assess and predict health risks.”
NICE released an extensive literature review with the revision, and based on the evidence, said that “using waist-to-height ratio as well as BMI would help give a practical estimate of central adiposity in adults with BMI under 35 kg/m2. This would in turn help professionals assess and predict health risks.”
However, the review added that, “because people with a BMI over 35 kg/m2 are always likely to have a high WHtR, the committee recognized that it may not be a useful addition for predicting health risks in this group.” The 2022 NICE review also said that it is “important to estimate central adiposity when assessing future health risks, including for people whose BMI is in the healthy-weight category.”
This new emphasis by NICE on measuring and using WHtR as part of obesity assessment “represents an important change in population health policy,” commented Dr. Powell-Wiley. “I expect more professional organizations will endorse use of waist circumference or waist-to-height ratio now that NICE has taken this step,” she predicted.
Waist circumference and WHtR may become standard measures of adiposity in clinical practice over the next 5-10 years.
The recent move by NICE to highlight a complementary role for WHtR “is another acknowledgment that BMI is an imperfect tool for stratifying cardiometabolic risk in a diverse population, especially in people with lower BMIs” because of its variability, commented Jamie Almandoz, MD, medical director of the weight wellness program at UT Southwestern Medical Center, Dallas.
WHtR vs. BMI
Another recent step forward for WHtR came with the publication of a post hoc analysis of data collected in the PARADIGM-HF trial, a study that had the primary purpose of comparing two medications for improving outcomes in more than 8,000 patients with heart failure with reduced ejection fraction.
The new analysis showed that “two indices that incorporate waist circumference and height, but not weight, showed a clearer association between greater adiposity and a higher risk of heart failure hospitalization,” compared with BMI.
WHtR was one of the two indices identified as being a better correlate for the adverse effect of excess adiposity compared with BMI.
The authors of the post hoc analysis did not design their analysis to compare WHtR with BMI. Instead, their goal was to better understand what’s known as the “obesity paradox” in people with heart failure with reduced ejection fraction: The recurring observation that, when these patients with heart failure have lower BMIs they fare worse, with higher rates of mortality and adverse cardiovascular outcomes, compared with patients with higher BMIs.
The new analysis showed that this paradox disappeared when WHtR was substituted for BMI as the obesity metric.
This “provides meaningful data about the superiority of WHtR, compared with BMI, for predicting heart failure outcomes,” said Dr. Powell-Wiley, although she cautioned that the analysis was limited by scant data in diverse populations and did not look at other important cardiovascular disease outcomes. While Dr. Powell-Wiley does not think that WHtR needs assessment in a prospective, controlled trial, she called for analysis of pooled prospective studies with more diverse populations to better document the advantages of WHtR over BMI.
The PARADIGM-HF post hoc analysis shows again how flawed BMI is for health assessment and the relative importance of an individualized understanding of a person’s body composition, Dr. Almandoz said in an interview. “As we collect more data, there is increasing awareness of how imperfect BMI is.”
Measuring waist circumference is tricky
Although WHtR looks promising as a substitute for or add-on to BMI, it has its own limitations, particularly the challenge of accurately measuring waist circumference.
Measuring waist circumference “not only takes more time but requires the assessor to be well trained about where to put the tape measure and making sure it’s measured at the same place each time,” even when different people take serial measurements from individual patients, noted Dr. Wee. Determining waist circumference can also be technically difficult when done on larger people, she added, and collectively these challenges make waist circumference “less reproducible from measurement to measurement.”
“It’s relatively clear how to standardize measurement of weight and height, but there is a huge amount of variability when the waist is measured,” agreed Dr. Almandoz. “And waist circumference also differs by ethnicity, race, sex, and body frame. There are significant differences in waist circumference levels that associate with increased health risks” between, for example, White and South Asian people.
Another limitation of waist circumference and WHtR is that they “cannot differentiate between visceral and abdominal subcutaneous adipose tissue, which are vastly different regarding cardiometabolic risk, commented Ian Neeland, MD, director of cardiovascular prevention at the University Hospitals Harrington Heart & Vascular Institute, Cleveland.
The imaging option
“Waist-to-height ratio is not the ultimate answer,” Dr. Neeland said in an interview. He instead endorsed “advanced imaging for body fat distribution,” such as CT or MRI scans, as his pick for what should be the standard obesity metric, “given that it is much more specific and actionable for both risk assessment and response to therapy. I expect slow but steady advancements that move away from BMI cutoffs, for example for bariatric surgery, given that BMI is an imprecise and crude tool.”
But although imaging with methods like CT and MRI may provide the best accuracy and precision for tracking the volume of a person’s cardiometabolically dangerous fat, they are also hampered by relatively high cost and, for CT and DXA, the issue of radiation exposure.
“CT, MRI, and DXA scans give more in-depth assessment of body composition, but should we expose people to the radiation and the cost?” Dr. Almandoz wondered.
“Height, weight, and waist circumference cost nothing to obtain,” creating a big relative disadvantage for imaging, said Naveed Sattar, MD, professor of metabolic medicine at the University of Glasgow.
“Data would need to show that imaging gives clinicians substantially more information about future risk” to justify its price, Dr. Sattar emphasized.
BMI’s limits mean adding on
Regardless of whichever alternatives to BMI end up getting used most, experts generally agree that BMI alone is looking increasingly inadequate.
“Over the next 5 years, BMI will come to be seen as a screening tool that categorizes people into general risk groups” that also needs “other metrics and variables, such as age, race, ethnicity, family history, blood glucose, and blood pressure to better describe health risk in an individual,” predicted Dr. Bessesen.
The endorsement of WHtR by NICE “will lead to more research into how to incorporate WHtR into routine practice. We need more evidence to translate what NICE said into practice,” said Dr. Sattar. “I don’t think we’ll see a shift away from BMI, but we’ll add alternative measures that are particularly useful in certain patients.”
“Because we live in diverse societies, we need to individualize risk assessment and couple that with technology that makes analysis of body composition more accessible,” agreed Dr. Almandoz. He noted that the UT Southwestern weight wellness program where he practices has, for about the past decade, routinely collected waist circumference and bioelectrical impedance data as well as BMI on all people seen in the practice for obesity concerns. Making these additional measurements on a routine basis also helps strengthen patient engagement.
“We get into trouble when we make rigid health policy and clinical decisions based on BMI alone without looking at the patient holistically,” said Dr. Wee. “Patients are more than arbitrary numbers, and clinicians should make clinical decisions based on the totality of evidence for each individual patient.”
Dr. Bessesen, Dr. Wee, Dr. Powell-Wiley, and Dr. Almandoz reported no relevant financial relationships. Dr. Neeland has reported being a consultant for Merck. Dr. Sattar has reported being a consultant or speaker for Abbott Laboratories, Afimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceuticals, Janssen, MSD, Novartis, Novo Nordisk, Pfizer, Roche Diagnostics, and Sanofi.
A version of this article originally appeared on Medscape.com.
“BMI is trash. Full stop.” This controversial tweet, which received thousands of likes and retweets, was cited in a recent article by one doctor on when physicians might stop using body mass index (BMI) to diagnose obesity.
BMI has for years been the consensus default method for assessing whether a person is overweight or has obesity, and is still widely used as the gatekeeper metric for treatment eligibility for certain weight-loss agents and bariatric surgery.
an important determinant of the cardiometabolic consequences of fat.
Alternative metrics include waist circumference and/or waist-to-height ratio (WHtR); imaging methods such as CT, MRI, and dual-energy x-ray absorptiometry (DXA); and bioelectrical impedance to assess fat volume and location. All have made some inroads on the tight grip BMI has had on obesity assessment.
Chances are, however, that BMI will not fade away anytime soon given how entrenched it has become in clinical practice and for insurance coverage, as well as its relative simplicity and precision.
“BMI is embedded in a wide range of guidelines on the use of medications and surgery. It’s embedded in Food and Drug Administration regulations and for billing and insurance coverage. It would take extremely strong data and years of work to undo the infrastructure built around BMI and replace it with something else. I don’t see that happening [anytime soon],” commented Daniel H. Bessesen, MD, a professor at the University of Colorado at Denver, Aurora, and chief of endocrinology for Denver Health.
“It would be almost impossible to replace all the studies that have used BMI with investigations using some other measure,” he said.
BMI Is ‘imperfect’
The entrenched position of BMI as the go-to metric doesn’t keep detractors from weighing in. As noted in a commentary on current clinical challenges surrounding obesity recently published in Annals of Internal Medicine, the journal’s editor-in-chief, Christine Laine, MD, and senior deputy editor Christina C. Wee, MD, listed six top issues clinicians must deal with, one of which, they say, is the need for a better measure of obesity than BMI.
“Unfortunately, BMI is an imperfect measure of body composition that differs with ethnicity, sex, body frame, and muscle mass,” noted Dr. Laine and Dr. Wee.
BMI is based on a person’s weight in kilograms divided by the square of their height in meters. A “healthy” BMI is between 18.5 and 24.9 kg/m2, overweight is 25-29.9, and 30 or greater is considered to represent obesity. However, certain ethnic groups have lower cutoffs for overweight or obesity because of evidence that such individuals can be at higher risk of obesity-related comorbidities at lower BMIs.
“BMI was chosen as the initial screening tool [for obesity] not because anyone thought it was perfect or the best measure but because of its simplicity. All you need is height, weight, and a calculator,” Dr. Wee said in an interview.
Numerous online calculators are available, including one from the Centers for Disease Control and Prevention where height in feet and inches and weight in pounds can be entered to generate the BMI.
BMI is also inherently limited by being “a proxy for adiposity” and not a direct measure, added Dr. Wee, who is also director of the Obesity Research Program of Beth Israel Deaconess Medical Center, Boston.
As such, BMI can’t distinguish between fat and muscle because it relies on weight only to gauge adiposity, noted Tiffany Powell-Wiley, MD, an obesity researcher at the National Heart, Lung, and Blood Institute in Bethesda, Md. Another shortcoming of BMI is that it “is good for distinguishing population-level risk for cardiovascular disease and other chronic diseases, but it does not help as much for distinguishing risk at an individual level,” she said in an interview.
These and other drawbacks have prompted researchers to look for other useful metrics. WHtR, for example, has recently made headway as a potential BMI alternative or complement.
The case for WHtR
Concern about overreliance on BMI despite its limitations is not new. In 2015, an American Heart Association scientific statement from the group’s Obesity Committee concluded that “BMI alone, even with lower thresholds, is a useful but not an ideal tool for identification of obesity or assessment of cardiovascular risk,” especially for people from Asian, Black, Hispanic, and Pacific Islander populations.
The writing panel also recommended that clinicians measure waist circumference annually and use that information along with BMI “to better gauge cardiovascular risk in diverse populations.”
Momentum for moving beyond BMI alone has continued to build following the AHA statement.
In September 2022, the National Institute for Health and Care Excellence, which sets policies for the United Kingdom’s National Health Service, revised its guidancefor assessment and management of people with obesity. The updated guidance recommends that when clinicians assess “adults with BMI below 35 kg/m2, measure and use their WHtR, as well as their BMI, as a practical estimate of central adiposity and use these measurements to help to assess and predict health risks.”
NICE released an extensive literature review with the revision, and based on the evidence, said that “using waist-to-height ratio as well as BMI would help give a practical estimate of central adiposity in adults with BMI under 35 kg/m2. This would in turn help professionals assess and predict health risks.”
However, the review added that, “because people with a BMI over 35 kg/m2 are always likely to have a high WHtR, the committee recognized that it may not be a useful addition for predicting health risks in this group.” The 2022 NICE review also said that it is “important to estimate central adiposity when assessing future health risks, including for people whose BMI is in the healthy-weight category.”
This new emphasis by NICE on measuring and using WHtR as part of obesity assessment “represents an important change in population health policy,” commented Dr. Powell-Wiley. “I expect more professional organizations will endorse use of waist circumference or waist-to-height ratio now that NICE has taken this step,” she predicted.
Waist circumference and WHtR may become standard measures of adiposity in clinical practice over the next 5-10 years.
The recent move by NICE to highlight a complementary role for WHtR “is another acknowledgment that BMI is an imperfect tool for stratifying cardiometabolic risk in a diverse population, especially in people with lower BMIs” because of its variability, commented Jamie Almandoz, MD, medical director of the weight wellness program at UT Southwestern Medical Center, Dallas.
WHtR vs. BMI
Another recent step forward for WHtR came with the publication of a post hoc analysis of data collected in the PARADIGM-HF trial, a study that had the primary purpose of comparing two medications for improving outcomes in more than 8,000 patients with heart failure with reduced ejection fraction.
The new analysis showed that “two indices that incorporate waist circumference and height, but not weight, showed a clearer association between greater adiposity and a higher risk of heart failure hospitalization,” compared with BMI.
WHtR was one of the two indices identified as being a better correlate for the adverse effect of excess adiposity compared with BMI.
The authors of the post hoc analysis did not design their analysis to compare WHtR with BMI. Instead, their goal was to better understand what’s known as the “obesity paradox” in people with heart failure with reduced ejection fraction: The recurring observation that, when these patients with heart failure have lower BMIs they fare worse, with higher rates of mortality and adverse cardiovascular outcomes, compared with patients with higher BMIs.
The new analysis showed that this paradox disappeared when WHtR was substituted for BMI as the obesity metric.
This “provides meaningful data about the superiority of WHtR, compared with BMI, for predicting heart failure outcomes,” said Dr. Powell-Wiley, although she cautioned that the analysis was limited by scant data in diverse populations and did not look at other important cardiovascular disease outcomes. While Dr. Powell-Wiley does not think that WHtR needs assessment in a prospective, controlled trial, she called for analysis of pooled prospective studies with more diverse populations to better document the advantages of WHtR over BMI.
The PARADIGM-HF post hoc analysis shows again how flawed BMI is for health assessment and the relative importance of an individualized understanding of a person’s body composition, Dr. Almandoz said in an interview. “As we collect more data, there is increasing awareness of how imperfect BMI is.”
Measuring waist circumference is tricky
Although WHtR looks promising as a substitute for or add-on to BMI, it has its own limitations, particularly the challenge of accurately measuring waist circumference.
Measuring waist circumference “not only takes more time but requires the assessor to be well trained about where to put the tape measure and making sure it’s measured at the same place each time,” even when different people take serial measurements from individual patients, noted Dr. Wee. Determining waist circumference can also be technically difficult when done on larger people, she added, and collectively these challenges make waist circumference “less reproducible from measurement to measurement.”
“It’s relatively clear how to standardize measurement of weight and height, but there is a huge amount of variability when the waist is measured,” agreed Dr. Almandoz. “And waist circumference also differs by ethnicity, race, sex, and body frame. There are significant differences in waist circumference levels that associate with increased health risks” between, for example, White and South Asian people.
Another limitation of waist circumference and WHtR is that they “cannot differentiate between visceral and abdominal subcutaneous adipose tissue, which are vastly different regarding cardiometabolic risk, commented Ian Neeland, MD, director of cardiovascular prevention at the University Hospitals Harrington Heart & Vascular Institute, Cleveland.
The imaging option
“Waist-to-height ratio is not the ultimate answer,” Dr. Neeland said in an interview. He instead endorsed “advanced imaging for body fat distribution,” such as CT or MRI scans, as his pick for what should be the standard obesity metric, “given that it is much more specific and actionable for both risk assessment and response to therapy. I expect slow but steady advancements that move away from BMI cutoffs, for example for bariatric surgery, given that BMI is an imprecise and crude tool.”
But although imaging with methods like CT and MRI may provide the best accuracy and precision for tracking the volume of a person’s cardiometabolically dangerous fat, they are also hampered by relatively high cost and, for CT and DXA, the issue of radiation exposure.
“CT, MRI, and DXA scans give more in-depth assessment of body composition, but should we expose people to the radiation and the cost?” Dr. Almandoz wondered.
“Height, weight, and waist circumference cost nothing to obtain,” creating a big relative disadvantage for imaging, said Naveed Sattar, MD, professor of metabolic medicine at the University of Glasgow.
“Data would need to show that imaging gives clinicians substantially more information about future risk” to justify its price, Dr. Sattar emphasized.
BMI’s limits mean adding on
Regardless of whichever alternatives to BMI end up getting used most, experts generally agree that BMI alone is looking increasingly inadequate.
“Over the next 5 years, BMI will come to be seen as a screening tool that categorizes people into general risk groups” that also needs “other metrics and variables, such as age, race, ethnicity, family history, blood glucose, and blood pressure to better describe health risk in an individual,” predicted Dr. Bessesen.
The endorsement of WHtR by NICE “will lead to more research into how to incorporate WHtR into routine practice. We need more evidence to translate what NICE said into practice,” said Dr. Sattar. “I don’t think we’ll see a shift away from BMI, but we’ll add alternative measures that are particularly useful in certain patients.”
“Because we live in diverse societies, we need to individualize risk assessment and couple that with technology that makes analysis of body composition more accessible,” agreed Dr. Almandoz. He noted that the UT Southwestern weight wellness program where he practices has, for about the past decade, routinely collected waist circumference and bioelectrical impedance data as well as BMI on all people seen in the practice for obesity concerns. Making these additional measurements on a routine basis also helps strengthen patient engagement.
“We get into trouble when we make rigid health policy and clinical decisions based on BMI alone without looking at the patient holistically,” said Dr. Wee. “Patients are more than arbitrary numbers, and clinicians should make clinical decisions based on the totality of evidence for each individual patient.”
Dr. Bessesen, Dr. Wee, Dr. Powell-Wiley, and Dr. Almandoz reported no relevant financial relationships. Dr. Neeland has reported being a consultant for Merck. Dr. Sattar has reported being a consultant or speaker for Abbott Laboratories, Afimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceuticals, Janssen, MSD, Novartis, Novo Nordisk, Pfizer, Roche Diagnostics, and Sanofi.
A version of this article originally appeared on Medscape.com.
Child’s health improves by applying new obesity guidelines
At age 15 years, Maya was referred by her primary care provider to our pediatric obesity center. She weighed 151 kg and had a body mass index (BMI) over 48 kg/m2. One year earlier, she had been diagnosed with hypertension and prediabetes.
A review of her growth charts showed that she had been in the 95th percentile at age 8 years. Her weight had steadily risen, with an exponential increase of 55 lb between 2020 and 2022, during the COVID-19 pandemic. Her primary care provider monitored her from age 8 to 12 years, providing nutrition and physical activity counseling.
In February, the American Academy of Pediatrics released new clinical practice guidelines for managing childhood obesity. A better understanding of the pathophysiology has challenged the old-worn concept of lack of will power and personal responsibility as the cause of obesity, which has been the basis for weight-related bias and stigma. The updated guidelines have also been influenced by lifestyle intervention studies, the US Food and Drug Administration approval of new anti-obesity medications, and the 2013 designation of obesity as a disease by the American Medical Association.
We used these updated guidelines in our approach to treating Maya.
Starting with the assessment
In the new AAP guidelines, assessing the genetic, environmental, and social-determinant risks for obesity form the basis for evaluation and intervention. Following this approach, we conducted a complete medical evaluation of Maya, including a review of her symptoms and her family history along with a physical examination to assess for comorbidities and other cause of obesity (for example, genetic, hypothyroidism).
We also collected information regarding her diet and behaviors (for example, drinking sweet beverages, fruit and vegetable intake, parent feeding style, portion sizes, emotional eating, hyperphagia), physical activity behaviors (for example, physical education, organized sports), screen time, social drivers of health (for example, food insecurity, neighborhood, school environment), family and household factors (for example, family composition, support, number of caregivers, parenting style) and mental and physical health (autism, attention-deficit/hyperactivity disorder, history of being bullied, developmental and physical disabilities). Because Maya had a BMI of 48, she met the criterion for severe obesity, which is having a BMI at least 120% of the 95th percentile.
The guidelines use BMI as a criterion for screening for obesity because it is inexpensive and easy to obtain in the clinic setting. The Centers for Disease Control and Prevention growth chart uses BMI as well. Recently, there has been controversy about solely using BMI to define obesity, which is a point that the guidelines address by emphasizing evaluation of the whole child along with BMI to make a diagnosis of obesity.
The child’s age and the severity of their obesity drive the evaluation for comorbidities and treatment. In children aged 10 years or older, pediatricians and other primary care providers should evaluate for lipid abnormalities, abnormal glucose metabolism, and abnormal liver function in children and adolescents with obesity (BMI ≥ 95th percentile).
Maya presented with snoring, early-morning headaches, daytime sleepiness, and abdominal pain. A sleep study revealed an apnea-hypopnea index of 15, indicating obstructive sleep apnea, and she was placed on a continuous positive airway pressure machine.
Her laboratory studies showed elevated triglycerides of 169 mg/dL and abnormal ALT (123 IU/L). Potential causes of elevated liver function test results (such as abnormal ceruloplasmin levels or infectious or autoimmune hepatitis) were excluded, and a liver ultrasound with elastography indicated steatohepatitis. Maya was referred to gastroenterology for nonalcoholic fatty liver disease.
Maya experienced depressive symptoms, including difficulty with peer relationships and declining academic performance. Her Patient Health Questionnaire–9 score was 21, with a moderate impact on her daily functioning. Prior attempts at counseling had been sporadic and not helpful. She was diagnosed with intermittent moderate clinical depression, started on a selective serotonin reuptake inhibitor, and resumed counseling with a new therapist.
Considering treatment options
Based on shared decision-making, our team began a more intensive lifestyle behavior treatment as recommended in the updated guidelines. Maya chose to decrease sugar-sweetened beverages as her initial nutrition goal, a change that can lead to a reduction of liver function test results and triglycerides, even in the absence of weight loss.
As emphasized in the guidelines, we stressed the importance of managing obesity and comorbidities concurrently to the family. In addition to lifestyle behavior intervention, once her mental health stabilized, Maya and her mother opted for bariatric surgery. Sleeve gastrectomy was elected because she met the criteria.
If the child already has obesity, the guidelines discourage watchful waiting (that is, the expectation that the child will grow into their weight) as Maya’s primary care provider had done when she was younger. The staged treatment approach where progressively more intensive interventions are adopted (a hallmark of the 2007 guidelines) is no longer recommended. Rather, the primary care provider should offer treatment options guided by age, severity of obesity, and comorbidities.
Maya completed a bariatric preoperative program, extensive mental health evaluation, and tolerated the sleeve gastrectomy well with no complications. At her 6-month postoperative visit, she had lost 99 lb (45 kg) since the surgery, with an 18% decline in BMI. She is taking daily multivitamins as well as calcium and vitamin D. She continues to incorporate healthy eating into her life, with a focus on adequate protein intake and is exercising three to four times per week in the apartment complex gym. She reports better physical and mental health, her school performance has improved, and she still receives regular counseling.
Maya’s story outlines the benefits of early and intensive intervention as recommended by the new AAP guidelines. The shift from some of the earlier recommendations is partly driven by the persistence of childhood obesity into adulthood, especially for older children with serious psychosocial and physical comorbidities. Hopefully by implementing the new guidelines, the physician can provide empathetic, bias-free, and effective care that recognizes the needs and environment of the whole child.
Dr. Salhah is a pediatric endocrinology fellow at Nationwide Children’s Hospital, Columbus, Ohio. Dr. Eneli is director of the Center for Healthy Weight and Nutrition at Nationwide Children’s Hospital. Dr. Salhah reported no conflicts of interest. Dr. Eneli reported receiving research grants and income from the National Institutes of Health, the AAP, and the National Academy of Medicine.
A version of this article first appeared on Medscape.com.
At age 15 years, Maya was referred by her primary care provider to our pediatric obesity center. She weighed 151 kg and had a body mass index (BMI) over 48 kg/m2. One year earlier, she had been diagnosed with hypertension and prediabetes.
A review of her growth charts showed that she had been in the 95th percentile at age 8 years. Her weight had steadily risen, with an exponential increase of 55 lb between 2020 and 2022, during the COVID-19 pandemic. Her primary care provider monitored her from age 8 to 12 years, providing nutrition and physical activity counseling.
In February, the American Academy of Pediatrics released new clinical practice guidelines for managing childhood obesity. A better understanding of the pathophysiology has challenged the old-worn concept of lack of will power and personal responsibility as the cause of obesity, which has been the basis for weight-related bias and stigma. The updated guidelines have also been influenced by lifestyle intervention studies, the US Food and Drug Administration approval of new anti-obesity medications, and the 2013 designation of obesity as a disease by the American Medical Association.
We used these updated guidelines in our approach to treating Maya.
Starting with the assessment
In the new AAP guidelines, assessing the genetic, environmental, and social-determinant risks for obesity form the basis for evaluation and intervention. Following this approach, we conducted a complete medical evaluation of Maya, including a review of her symptoms and her family history along with a physical examination to assess for comorbidities and other cause of obesity (for example, genetic, hypothyroidism).
We also collected information regarding her diet and behaviors (for example, drinking sweet beverages, fruit and vegetable intake, parent feeding style, portion sizes, emotional eating, hyperphagia), physical activity behaviors (for example, physical education, organized sports), screen time, social drivers of health (for example, food insecurity, neighborhood, school environment), family and household factors (for example, family composition, support, number of caregivers, parenting style) and mental and physical health (autism, attention-deficit/hyperactivity disorder, history of being bullied, developmental and physical disabilities). Because Maya had a BMI of 48, she met the criterion for severe obesity, which is having a BMI at least 120% of the 95th percentile.
The guidelines use BMI as a criterion for screening for obesity because it is inexpensive and easy to obtain in the clinic setting. The Centers for Disease Control and Prevention growth chart uses BMI as well. Recently, there has been controversy about solely using BMI to define obesity, which is a point that the guidelines address by emphasizing evaluation of the whole child along with BMI to make a diagnosis of obesity.
The child’s age and the severity of their obesity drive the evaluation for comorbidities and treatment. In children aged 10 years or older, pediatricians and other primary care providers should evaluate for lipid abnormalities, abnormal glucose metabolism, and abnormal liver function in children and adolescents with obesity (BMI ≥ 95th percentile).
Maya presented with snoring, early-morning headaches, daytime sleepiness, and abdominal pain. A sleep study revealed an apnea-hypopnea index of 15, indicating obstructive sleep apnea, and she was placed on a continuous positive airway pressure machine.
Her laboratory studies showed elevated triglycerides of 169 mg/dL and abnormal ALT (123 IU/L). Potential causes of elevated liver function test results (such as abnormal ceruloplasmin levels or infectious or autoimmune hepatitis) were excluded, and a liver ultrasound with elastography indicated steatohepatitis. Maya was referred to gastroenterology for nonalcoholic fatty liver disease.
Maya experienced depressive symptoms, including difficulty with peer relationships and declining academic performance. Her Patient Health Questionnaire–9 score was 21, with a moderate impact on her daily functioning. Prior attempts at counseling had been sporadic and not helpful. She was diagnosed with intermittent moderate clinical depression, started on a selective serotonin reuptake inhibitor, and resumed counseling with a new therapist.
Considering treatment options
Based on shared decision-making, our team began a more intensive lifestyle behavior treatment as recommended in the updated guidelines. Maya chose to decrease sugar-sweetened beverages as her initial nutrition goal, a change that can lead to a reduction of liver function test results and triglycerides, even in the absence of weight loss.
As emphasized in the guidelines, we stressed the importance of managing obesity and comorbidities concurrently to the family. In addition to lifestyle behavior intervention, once her mental health stabilized, Maya and her mother opted for bariatric surgery. Sleeve gastrectomy was elected because she met the criteria.
If the child already has obesity, the guidelines discourage watchful waiting (that is, the expectation that the child will grow into their weight) as Maya’s primary care provider had done when she was younger. The staged treatment approach where progressively more intensive interventions are adopted (a hallmark of the 2007 guidelines) is no longer recommended. Rather, the primary care provider should offer treatment options guided by age, severity of obesity, and comorbidities.
Maya completed a bariatric preoperative program, extensive mental health evaluation, and tolerated the sleeve gastrectomy well with no complications. At her 6-month postoperative visit, she had lost 99 lb (45 kg) since the surgery, with an 18% decline in BMI. She is taking daily multivitamins as well as calcium and vitamin D. She continues to incorporate healthy eating into her life, with a focus on adequate protein intake and is exercising three to four times per week in the apartment complex gym. She reports better physical and mental health, her school performance has improved, and she still receives regular counseling.
Maya’s story outlines the benefits of early and intensive intervention as recommended by the new AAP guidelines. The shift from some of the earlier recommendations is partly driven by the persistence of childhood obesity into adulthood, especially for older children with serious psychosocial and physical comorbidities. Hopefully by implementing the new guidelines, the physician can provide empathetic, bias-free, and effective care that recognizes the needs and environment of the whole child.
Dr. Salhah is a pediatric endocrinology fellow at Nationwide Children’s Hospital, Columbus, Ohio. Dr. Eneli is director of the Center for Healthy Weight and Nutrition at Nationwide Children’s Hospital. Dr. Salhah reported no conflicts of interest. Dr. Eneli reported receiving research grants and income from the National Institutes of Health, the AAP, and the National Academy of Medicine.
A version of this article first appeared on Medscape.com.
At age 15 years, Maya was referred by her primary care provider to our pediatric obesity center. She weighed 151 kg and had a body mass index (BMI) over 48 kg/m2. One year earlier, she had been diagnosed with hypertension and prediabetes.
A review of her growth charts showed that she had been in the 95th percentile at age 8 years. Her weight had steadily risen, with an exponential increase of 55 lb between 2020 and 2022, during the COVID-19 pandemic. Her primary care provider monitored her from age 8 to 12 years, providing nutrition and physical activity counseling.
In February, the American Academy of Pediatrics released new clinical practice guidelines for managing childhood obesity. A better understanding of the pathophysiology has challenged the old-worn concept of lack of will power and personal responsibility as the cause of obesity, which has been the basis for weight-related bias and stigma. The updated guidelines have also been influenced by lifestyle intervention studies, the US Food and Drug Administration approval of new anti-obesity medications, and the 2013 designation of obesity as a disease by the American Medical Association.
We used these updated guidelines in our approach to treating Maya.
Starting with the assessment
In the new AAP guidelines, assessing the genetic, environmental, and social-determinant risks for obesity form the basis for evaluation and intervention. Following this approach, we conducted a complete medical evaluation of Maya, including a review of her symptoms and her family history along with a physical examination to assess for comorbidities and other cause of obesity (for example, genetic, hypothyroidism).
We also collected information regarding her diet and behaviors (for example, drinking sweet beverages, fruit and vegetable intake, parent feeding style, portion sizes, emotional eating, hyperphagia), physical activity behaviors (for example, physical education, organized sports), screen time, social drivers of health (for example, food insecurity, neighborhood, school environment), family and household factors (for example, family composition, support, number of caregivers, parenting style) and mental and physical health (autism, attention-deficit/hyperactivity disorder, history of being bullied, developmental and physical disabilities). Because Maya had a BMI of 48, she met the criterion for severe obesity, which is having a BMI at least 120% of the 95th percentile.
The guidelines use BMI as a criterion for screening for obesity because it is inexpensive and easy to obtain in the clinic setting. The Centers for Disease Control and Prevention growth chart uses BMI as well. Recently, there has been controversy about solely using BMI to define obesity, which is a point that the guidelines address by emphasizing evaluation of the whole child along with BMI to make a diagnosis of obesity.
The child’s age and the severity of their obesity drive the evaluation for comorbidities and treatment. In children aged 10 years or older, pediatricians and other primary care providers should evaluate for lipid abnormalities, abnormal glucose metabolism, and abnormal liver function in children and adolescents with obesity (BMI ≥ 95th percentile).
Maya presented with snoring, early-morning headaches, daytime sleepiness, and abdominal pain. A sleep study revealed an apnea-hypopnea index of 15, indicating obstructive sleep apnea, and she was placed on a continuous positive airway pressure machine.
Her laboratory studies showed elevated triglycerides of 169 mg/dL and abnormal ALT (123 IU/L). Potential causes of elevated liver function test results (such as abnormal ceruloplasmin levels or infectious or autoimmune hepatitis) were excluded, and a liver ultrasound with elastography indicated steatohepatitis. Maya was referred to gastroenterology for nonalcoholic fatty liver disease.
Maya experienced depressive symptoms, including difficulty with peer relationships and declining academic performance. Her Patient Health Questionnaire–9 score was 21, with a moderate impact on her daily functioning. Prior attempts at counseling had been sporadic and not helpful. She was diagnosed with intermittent moderate clinical depression, started on a selective serotonin reuptake inhibitor, and resumed counseling with a new therapist.
Considering treatment options
Based on shared decision-making, our team began a more intensive lifestyle behavior treatment as recommended in the updated guidelines. Maya chose to decrease sugar-sweetened beverages as her initial nutrition goal, a change that can lead to a reduction of liver function test results and triglycerides, even in the absence of weight loss.
As emphasized in the guidelines, we stressed the importance of managing obesity and comorbidities concurrently to the family. In addition to lifestyle behavior intervention, once her mental health stabilized, Maya and her mother opted for bariatric surgery. Sleeve gastrectomy was elected because she met the criteria.
If the child already has obesity, the guidelines discourage watchful waiting (that is, the expectation that the child will grow into their weight) as Maya’s primary care provider had done when she was younger. The staged treatment approach where progressively more intensive interventions are adopted (a hallmark of the 2007 guidelines) is no longer recommended. Rather, the primary care provider should offer treatment options guided by age, severity of obesity, and comorbidities.
Maya completed a bariatric preoperative program, extensive mental health evaluation, and tolerated the sleeve gastrectomy well with no complications. At her 6-month postoperative visit, she had lost 99 lb (45 kg) since the surgery, with an 18% decline in BMI. She is taking daily multivitamins as well as calcium and vitamin D. She continues to incorporate healthy eating into her life, with a focus on adequate protein intake and is exercising three to four times per week in the apartment complex gym. She reports better physical and mental health, her school performance has improved, and she still receives regular counseling.
Maya’s story outlines the benefits of early and intensive intervention as recommended by the new AAP guidelines. The shift from some of the earlier recommendations is partly driven by the persistence of childhood obesity into adulthood, especially for older children with serious psychosocial and physical comorbidities. Hopefully by implementing the new guidelines, the physician can provide empathetic, bias-free, and effective care that recognizes the needs and environment of the whole child.
Dr. Salhah is a pediatric endocrinology fellow at Nationwide Children’s Hospital, Columbus, Ohio. Dr. Eneli is director of the Center for Healthy Weight and Nutrition at Nationwide Children’s Hospital. Dr. Salhah reported no conflicts of interest. Dr. Eneli reported receiving research grants and income from the National Institutes of Health, the AAP, and the National Academy of Medicine.
A version of this article first appeared on Medscape.com.
Weight gain and excessive fatigue
This patient's clinical presentation and laboratory findings are consistent with a diagnosis of Cushing syndrome (CS).
CS is a rare endocrine disease caused by prolonged exposure to high circulating cortisol levels. Exogenous hypercortisolism is the most common cause of CS. It is largely iatrogenic and results from the prolonged use of glucocorticoids. Less frequently, endogenous CS may occur as the result of excessive production of cortisol by adrenal glands. Endogenous CS can be ACTH-dependent or ACTH-independent. ACTH-dependent CS results from ACTH-secreting pituitary adenomas (Cushing disease) and ectopic ACTH secretion by neoplasms, whereas adrenal hyperplasia, adenoma, and carcinoma are the primary causes of ACTH-independent CS.
The annual incidence and prevalence of CS are unknown; the reported incidence of newly diagnosed cases has ranged from 1.2 to 2.4 per million people per year. Women are affected more often than are men, with a peak of incidence in the third to fourth decade of life. CS is associated with various metabolic, psychiatric, musculoskeletal, and cardiovascular comorbidities. Untreated, it is associated with increased mortality, typically as the result of cardiovascular and infectious complications; however, even in appropriately treated patients, mortality is elevated.
The chronic elevations of glucocorticoid concentrations in CS result in its characteristic phenotype, which includes weight gain, moon-shaped face, buffalo hump, muscle weakness, increased bruising, skin atrophy, red abdominal striae, menstrual irregularities, hirsutism, and acne. It is also associated with numerous comorbidities including diabetes, hypertension, hypercholesterolemia, and osteoporosis. Patients often experience mental health complications, such as depression, emotional lability, and cognitive dysfunction.
Given the rarity of CS and the fact that these symptoms overlap with other conditions, delayed diagnosis is common. The current obesity epidemic also poses diagnostic challenges because true CS can be difficult to differentiate from metabolic syndrome. The duration of hypercortisolism appears to be the most significant factor associated with the degree of morbidity and preterm mortality in CS; thus, an accurate diagnosis as early as possible is important.
Screening and diagnostic tests for CS evaluate cortisol secretion. Available options include late-night salivary cortisol (LNSC), impaired glucocorticoid feedback with overnight 1-mg DST or low-dose 2-day dexamethasone test (LDDT) and increased bioavailable cortisol with 24-hour UFC.
A 2021 consensus statement by Fleseriu and colleagues provides recommendations for the diagnosis of CS. If CS is suspected: begin with UFC, LNSC, or both; DST is an option if LNSC not feasible. If CS because of adrenal tumor is suspected: begin with DST because LNSC has lower specificity in these patients. To confirm CS, any of these tests can be used.
An individualized approach is recommended for the treatment of CS. The optimal approach for iatrogenic CS is to slowly taper exogenous steroids. Chronic exposure to steroids can suppress adrenal functioning; as such, recovery may take several months. Surgical resection is the first-line option for hypercortisolism because of Cushing disease, adrenal tumor, or ectopic tumor. Patients should be closely monitored after surgery to evaluate for possible recurrence. Radiotherapy may be recommended after failed transsphenoidal surgery or in Cushing disease with mass effect or invasion of surrounding structures. Medical therapy, such as pasireotide, cabergoline, and mifepristone, are also sometimes used. In addition, the treatment of comorbidities, such as obesity and type 2 diabetes, hypertension, osteoporosis, psychiatric issues, and electrolyte disorders, is critical.
Courtney Whittle, MD, MSW, Diplomate of ABOM, Pediatric Lead, Obesity Champion, TSPMG, Weight A Minute Clinic, Atlanta, Georgia.
Courtney Whittle, MD, MSW, Diplomate of ABOM, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
This patient's clinical presentation and laboratory findings are consistent with a diagnosis of Cushing syndrome (CS).
CS is a rare endocrine disease caused by prolonged exposure to high circulating cortisol levels. Exogenous hypercortisolism is the most common cause of CS. It is largely iatrogenic and results from the prolonged use of glucocorticoids. Less frequently, endogenous CS may occur as the result of excessive production of cortisol by adrenal glands. Endogenous CS can be ACTH-dependent or ACTH-independent. ACTH-dependent CS results from ACTH-secreting pituitary adenomas (Cushing disease) and ectopic ACTH secretion by neoplasms, whereas adrenal hyperplasia, adenoma, and carcinoma are the primary causes of ACTH-independent CS.
The annual incidence and prevalence of CS are unknown; the reported incidence of newly diagnosed cases has ranged from 1.2 to 2.4 per million people per year. Women are affected more often than are men, with a peak of incidence in the third to fourth decade of life. CS is associated with various metabolic, psychiatric, musculoskeletal, and cardiovascular comorbidities. Untreated, it is associated with increased mortality, typically as the result of cardiovascular and infectious complications; however, even in appropriately treated patients, mortality is elevated.
The chronic elevations of glucocorticoid concentrations in CS result in its characteristic phenotype, which includes weight gain, moon-shaped face, buffalo hump, muscle weakness, increased bruising, skin atrophy, red abdominal striae, menstrual irregularities, hirsutism, and acne. It is also associated with numerous comorbidities including diabetes, hypertension, hypercholesterolemia, and osteoporosis. Patients often experience mental health complications, such as depression, emotional lability, and cognitive dysfunction.
Given the rarity of CS and the fact that these symptoms overlap with other conditions, delayed diagnosis is common. The current obesity epidemic also poses diagnostic challenges because true CS can be difficult to differentiate from metabolic syndrome. The duration of hypercortisolism appears to be the most significant factor associated with the degree of morbidity and preterm mortality in CS; thus, an accurate diagnosis as early as possible is important.
Screening and diagnostic tests for CS evaluate cortisol secretion. Available options include late-night salivary cortisol (LNSC), impaired glucocorticoid feedback with overnight 1-mg DST or low-dose 2-day dexamethasone test (LDDT) and increased bioavailable cortisol with 24-hour UFC.
A 2021 consensus statement by Fleseriu and colleagues provides recommendations for the diagnosis of CS. If CS is suspected: begin with UFC, LNSC, or both; DST is an option if LNSC not feasible. If CS because of adrenal tumor is suspected: begin with DST because LNSC has lower specificity in these patients. To confirm CS, any of these tests can be used.
An individualized approach is recommended for the treatment of CS. The optimal approach for iatrogenic CS is to slowly taper exogenous steroids. Chronic exposure to steroids can suppress adrenal functioning; as such, recovery may take several months. Surgical resection is the first-line option for hypercortisolism because of Cushing disease, adrenal tumor, or ectopic tumor. Patients should be closely monitored after surgery to evaluate for possible recurrence. Radiotherapy may be recommended after failed transsphenoidal surgery or in Cushing disease with mass effect or invasion of surrounding structures. Medical therapy, such as pasireotide, cabergoline, and mifepristone, are also sometimes used. In addition, the treatment of comorbidities, such as obesity and type 2 diabetes, hypertension, osteoporosis, psychiatric issues, and electrolyte disorders, is critical.
Courtney Whittle, MD, MSW, Diplomate of ABOM, Pediatric Lead, Obesity Champion, TSPMG, Weight A Minute Clinic, Atlanta, Georgia.
Courtney Whittle, MD, MSW, Diplomate of ABOM, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
This patient's clinical presentation and laboratory findings are consistent with a diagnosis of Cushing syndrome (CS).
CS is a rare endocrine disease caused by prolonged exposure to high circulating cortisol levels. Exogenous hypercortisolism is the most common cause of CS. It is largely iatrogenic and results from the prolonged use of glucocorticoids. Less frequently, endogenous CS may occur as the result of excessive production of cortisol by adrenal glands. Endogenous CS can be ACTH-dependent or ACTH-independent. ACTH-dependent CS results from ACTH-secreting pituitary adenomas (Cushing disease) and ectopic ACTH secretion by neoplasms, whereas adrenal hyperplasia, adenoma, and carcinoma are the primary causes of ACTH-independent CS.
The annual incidence and prevalence of CS are unknown; the reported incidence of newly diagnosed cases has ranged from 1.2 to 2.4 per million people per year. Women are affected more often than are men, with a peak of incidence in the third to fourth decade of life. CS is associated with various metabolic, psychiatric, musculoskeletal, and cardiovascular comorbidities. Untreated, it is associated with increased mortality, typically as the result of cardiovascular and infectious complications; however, even in appropriately treated patients, mortality is elevated.
The chronic elevations of glucocorticoid concentrations in CS result in its characteristic phenotype, which includes weight gain, moon-shaped face, buffalo hump, muscle weakness, increased bruising, skin atrophy, red abdominal striae, menstrual irregularities, hirsutism, and acne. It is also associated with numerous comorbidities including diabetes, hypertension, hypercholesterolemia, and osteoporosis. Patients often experience mental health complications, such as depression, emotional lability, and cognitive dysfunction.
Given the rarity of CS and the fact that these symptoms overlap with other conditions, delayed diagnosis is common. The current obesity epidemic also poses diagnostic challenges because true CS can be difficult to differentiate from metabolic syndrome. The duration of hypercortisolism appears to be the most significant factor associated with the degree of morbidity and preterm mortality in CS; thus, an accurate diagnosis as early as possible is important.
Screening and diagnostic tests for CS evaluate cortisol secretion. Available options include late-night salivary cortisol (LNSC), impaired glucocorticoid feedback with overnight 1-mg DST or low-dose 2-day dexamethasone test (LDDT) and increased bioavailable cortisol with 24-hour UFC.
A 2021 consensus statement by Fleseriu and colleagues provides recommendations for the diagnosis of CS. If CS is suspected: begin with UFC, LNSC, or both; DST is an option if LNSC not feasible. If CS because of adrenal tumor is suspected: begin with DST because LNSC has lower specificity in these patients. To confirm CS, any of these tests can be used.
An individualized approach is recommended for the treatment of CS. The optimal approach for iatrogenic CS is to slowly taper exogenous steroids. Chronic exposure to steroids can suppress adrenal functioning; as such, recovery may take several months. Surgical resection is the first-line option for hypercortisolism because of Cushing disease, adrenal tumor, or ectopic tumor. Patients should be closely monitored after surgery to evaluate for possible recurrence. Radiotherapy may be recommended after failed transsphenoidal surgery or in Cushing disease with mass effect or invasion of surrounding structures. Medical therapy, such as pasireotide, cabergoline, and mifepristone, are also sometimes used. In addition, the treatment of comorbidities, such as obesity and type 2 diabetes, hypertension, osteoporosis, psychiatric issues, and electrolyte disorders, is critical.
Courtney Whittle, MD, MSW, Diplomate of ABOM, Pediatric Lead, Obesity Champion, TSPMG, Weight A Minute Clinic, Atlanta, Georgia.
Courtney Whittle, MD, MSW, Diplomate of ABOM, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
A 37-year-old woman presents with reports of insomnia, weight gain (approximately 12 lb over the last 9 months), and excessive fatigue. The patient's medical history is significant for hypertension (diagnosed 4 years earlier) and depression (diagnosed 7 years earlier). Her current medications include lisinopril 10 mg/d, bupropion 75 mg/d, and venlafaxine 75 mg/d. There is no history of alcohol or drug abuse; family history is unremarkable. The patient's height and weight are 5 ft 5 in and 182 lb (body mass index of 30.3).
During physical examination, facial hirsutism is observed along with increased adipose tissue in the face (moon-shaped face), upper back at the base of the neck (buffalo hump), and abdomen. Vertical red abdominal striae are present. Several bruises are observed on the patient's thighs and arms; when questioned, she reports noting an increased tendency to bruise in recent months.
Pertinent laboratory findings include urinary free cortisol excretion (UFC) 324 mcg/24 h, 1-mg dexamethasone suppression test (DST) with a cortisol value of 3.64 mcg/dL (100.42 nmol/L), and adrenocorticotropic hormone (ACTH) level of 84.9 pg/mL.
Obesity Treatment
Optimal time period for weight loss drugs: Debate continues
After bariatric surgery in 2014, Kristal Hartman still struggled to manage her weight long term. It took her over a year to lose 100 pounds, a loss she initially maintained, but then gradually her body mass index (BMI) started creeping up again.
“The body kind of has a set point, and you have to constantly trick it because it is going to start to gain weight again,” Ms. Hartman, who is on the national board of directors for the Obesity Action Coalition, said in an interview.
So, 2.5 years after her surgery, Ms. Hartman began weekly subcutaneous injections of the glucagonlike peptide–1 (GLP-1) agonist semaglutide, a medication that is now almost infamous because of its popularity among celebrities and social media influencers.
Branded as Ozempic for type 2 diabetes and Wegovy for obesity, both contain semaglutide but in slightly different doses. The popularity of the medication has led to shortages for those living with type 2 diabetes and/or obesity. And other medications are waiting in the wings that work on GLP-1 and other hormones that regulate appetite, such as the twincretin tirzepatide (Mounjaro), another weekly injection, approved by the Food and Drug Administration in May 2022 for type 2 diabetes and awaiting approval for obesity.
Ms. Hartman said taking semaglutide helped her not only lose weight but also “curb [her] obsessive thoughts over food.” To maintain a BMI within the healthy range, as well as taking the GLP-1 agonist, Ms. Hartman relies on other strategies, including exercise, and mental health support.
“Physicians really need to be open to these FDA-approved medications as one of many tools in the toolbox for patients with obesity. It’s just like any other chronic disease state, when they are thinking of using these, they need to think about long-term use ... in patients who have obesity, not just [among those people] who just want to lose 5-10 pounds. That’s not what these drugs are designed for. They are for people who are actually living with the chronic disease of obesity every day of their lives,” she emphasized.
On average, patients lose 25%-40% of their total body weight following bariatric surgery, said Teresa LeMasters, MD, president of the American Society for Metabolic & Bariatric Surgery. However, there typically is a “small” weight regain after surgery.
“For most patients, it is a small 5-10 pounds, but for some others, it can be significant,” said Dr. LeMasters, a bariatric surgeon at UnityPoint Clinic, Des Moines, Iowa.
“We do still see some patients– anywhere from 10% to 30% – who will have some [significant] weight regain, and so then we will look at that,” she noted. In those cases, the disease of obesity “is definitely still present.”
Medications can counter weight regain after surgery
For patients who don’t reach their weight loss goals after bariatric surgery, Dr. LeMasters said it’s appropriate to consider adding an anti-obesity medication. The newer GLP-1 agonists can lead to a loss of around 15% of body weight in some patients.
or even just to optimize their initial response to surgery if they are starting at a very, very severe point of disease,” she explained.
She noted, however, that some patients shouldn’t be prescribed GLP-1 agonists, including those with a history of thyroid cancer or pancreatitis.
Caroline M. Apovian, MD, codirector of the center for weight management and wellness and professor of medicine at Harvard Medical School, Boston, said in an interview that the physiology behind bariatric surgery and that of the newer obesity medications is somewhat aligned.
“In order to reduce ... body weight permanently you need adjustments. We learned that you need the adjustments of the hormones [that affect appetite, such as GLP-1], and that’s why bariatric surgery works because ... [it] provides the most durable and the most effective treatment for obesity ... because [with surgery] you are adjusting the secretion and timing of many of the hormones that regulate body weight,” she explained.
So, when people are taking GLP-1 agonists for obesity, with or without surgery, these medications “are meant and were approved by the FDA to be taken indefinitely. They are not [for the] short term,” Dr. Apovian noted.
Benjamin O’Donnell, MD, an associate professor at Ohio State University Wexner Medical Center, Columbus, agreed that the newer anti-obesity medications can be very effective; however, he expressed uncertainty about prescribing these medications for years and years.
“If somebody has obesity, they need medicine to help them manage appetite and maintain a lower, healthier weight. It would make sense that they would just stay on the medicine,” he noted.
But he qualified: “I have a hard time committing to saying that someone should take this medication for the rest of their life. Part of my hesitation is that the medications are expensive, so we’ve had a hard time with people staying on them, mostly because of insurance formulary changes.”
Why stop the medications? Side effects and lack of insurance coverage
Many people have to discontinue these newer medications for that exact reason.
When Ms. Hartman’s insurance coverage lapsed, she had to go without semaglutide for a while.
“At that time, I absolutely gained weight back up into an abnormal BMI range,” Ms. Hartman said. When she was able to resume the medication, she lost weight again and her BMI returned to normal range.
These medications currently cost around $1,400 per month in the United States, unless patients can access initiatives such as company coupons. Some insurers, including state-subsidized Medicare and Medicaid, don’t cover the new medications.
Dr. O’Donnell said, “More accessibility for more people would help in the big picture.”
Other patients stop taking GLP-1 agonists because they experience side effects, such as nausea.
“Gastrointestinal complaints ... are the number one reason for people to come off the medication,” said Disha Narang, MD, an endocrinologist and obesity medicine specialist at Northwestern Medicine Lake Forest (Ill.) Hospital.
“It is an elective therapy, so it is not mandatory that someone take it. So if they are not feeling well or they are sick, then that’s a major reason for coming off of it,” she said.
Dan Bessesen, MD, professor of medicine at the University of Colorado at Denver, Aurora, and chief of endocrinology, agreed.
Patients are unlikely to stay on these medications if they feel nauseous or experience vomiting, he said. Although he noted there are options to try to counter this, such as starting patients on a very low dose of the drug and up-titrating slowly. This method requires good coordination between the patient and physician.
Goutham Rao, MD, a professor of medicine at Case Western Reserve University and head of the weight-loss initiative Fitter Me at University Hospitals, both in Cleveland, said that prior to prescribing GLP-1 agonists for weight loss, he sets four basic, nonnegotiable goals for patients: “to have breakfast within 30 minutes of getting up, to drink just water, no food or drink after 7:00 p.m. except for water, and 30 minutes of continuous exercise per day, which is typically, for older clientele, walking.”
This, he said, can help establish good habits because if “patients are not engaged psychologically in weight loss ... they expect the medication to do [all] the work.”
Most regain weight after stopping obesity medications
As Ms. Hartman’s story illustrates, discontinuing the medications often leads to weight regain.
“Without the medicine, there are a variety of things that will happen. Appetite will tend to increase, and so [patients] will gradually tend to eat more over time,” Dr. Bessesen noted.
“So it may take a long time for the weight regain to happen, but in every study where an obesity medicine has been used, and then it is stopped, the weight goes back to where it was on lifestyle alone,” he added.
In the STEP 1 trial, almost 2,000 patients who were either overweight or living with obesity were randomized 2:1 to semaglutide, titrated up to 2.4 mg each week by week 16, or placebo in addition to lifestyle modification. After 68 weeks, those in the semaglutide group had a mean weight loss of 14.9%, compared with 2.4% in the placebo group.
Patients were also followed in a 1-year extension of the trial, published in Diabetes, Obesity, and Metabolism.
Within 1 year of stopping treatment, participants regained two thirds of the weight they had initially lost.
Hence, Dr. Bessesen stressed that a total rethink of how obesity is approached is needed among most physicians.
“I think in the future treating obesity with medications should be like treating hypertension and diabetes, something most primary care doctors are comfortable doing, but that’s going to take a little work and practice on the part of clinicians to really have a comfortable conversation about risks, and benefits, with patients,” he said.
“I would encourage primary care doctors to learn more about the treatment of obesity, and learn more about bias and stigma, and think about how they can deliver care that is compassionate and competent,” he concluded.
A version of this article first appeared on Medscape.com.
After bariatric surgery in 2014, Kristal Hartman still struggled to manage her weight long term. It took her over a year to lose 100 pounds, a loss she initially maintained, but then gradually her body mass index (BMI) started creeping up again.
“The body kind of has a set point, and you have to constantly trick it because it is going to start to gain weight again,” Ms. Hartman, who is on the national board of directors for the Obesity Action Coalition, said in an interview.
So, 2.5 years after her surgery, Ms. Hartman began weekly subcutaneous injections of the glucagonlike peptide–1 (GLP-1) agonist semaglutide, a medication that is now almost infamous because of its popularity among celebrities and social media influencers.
Branded as Ozempic for type 2 diabetes and Wegovy for obesity, both contain semaglutide but in slightly different doses. The popularity of the medication has led to shortages for those living with type 2 diabetes and/or obesity. And other medications are waiting in the wings that work on GLP-1 and other hormones that regulate appetite, such as the twincretin tirzepatide (Mounjaro), another weekly injection, approved by the Food and Drug Administration in May 2022 for type 2 diabetes and awaiting approval for obesity.
Ms. Hartman said taking semaglutide helped her not only lose weight but also “curb [her] obsessive thoughts over food.” To maintain a BMI within the healthy range, as well as taking the GLP-1 agonist, Ms. Hartman relies on other strategies, including exercise, and mental health support.
“Physicians really need to be open to these FDA-approved medications as one of many tools in the toolbox for patients with obesity. It’s just like any other chronic disease state, when they are thinking of using these, they need to think about long-term use ... in patients who have obesity, not just [among those people] who just want to lose 5-10 pounds. That’s not what these drugs are designed for. They are for people who are actually living with the chronic disease of obesity every day of their lives,” she emphasized.
On average, patients lose 25%-40% of their total body weight following bariatric surgery, said Teresa LeMasters, MD, president of the American Society for Metabolic & Bariatric Surgery. However, there typically is a “small” weight regain after surgery.
“For most patients, it is a small 5-10 pounds, but for some others, it can be significant,” said Dr. LeMasters, a bariatric surgeon at UnityPoint Clinic, Des Moines, Iowa.
“We do still see some patients– anywhere from 10% to 30% – who will have some [significant] weight regain, and so then we will look at that,” she noted. In those cases, the disease of obesity “is definitely still present.”
Medications can counter weight regain after surgery
For patients who don’t reach their weight loss goals after bariatric surgery, Dr. LeMasters said it’s appropriate to consider adding an anti-obesity medication. The newer GLP-1 agonists can lead to a loss of around 15% of body weight in some patients.
or even just to optimize their initial response to surgery if they are starting at a very, very severe point of disease,” she explained.
She noted, however, that some patients shouldn’t be prescribed GLP-1 agonists, including those with a history of thyroid cancer or pancreatitis.
Caroline M. Apovian, MD, codirector of the center for weight management and wellness and professor of medicine at Harvard Medical School, Boston, said in an interview that the physiology behind bariatric surgery and that of the newer obesity medications is somewhat aligned.
“In order to reduce ... body weight permanently you need adjustments. We learned that you need the adjustments of the hormones [that affect appetite, such as GLP-1], and that’s why bariatric surgery works because ... [it] provides the most durable and the most effective treatment for obesity ... because [with surgery] you are adjusting the secretion and timing of many of the hormones that regulate body weight,” she explained.
So, when people are taking GLP-1 agonists for obesity, with or without surgery, these medications “are meant and were approved by the FDA to be taken indefinitely. They are not [for the] short term,” Dr. Apovian noted.
Benjamin O’Donnell, MD, an associate professor at Ohio State University Wexner Medical Center, Columbus, agreed that the newer anti-obesity medications can be very effective; however, he expressed uncertainty about prescribing these medications for years and years.
“If somebody has obesity, they need medicine to help them manage appetite and maintain a lower, healthier weight. It would make sense that they would just stay on the medicine,” he noted.
But he qualified: “I have a hard time committing to saying that someone should take this medication for the rest of their life. Part of my hesitation is that the medications are expensive, so we’ve had a hard time with people staying on them, mostly because of insurance formulary changes.”
Why stop the medications? Side effects and lack of insurance coverage
Many people have to discontinue these newer medications for that exact reason.
When Ms. Hartman’s insurance coverage lapsed, she had to go without semaglutide for a while.
“At that time, I absolutely gained weight back up into an abnormal BMI range,” Ms. Hartman said. When she was able to resume the medication, she lost weight again and her BMI returned to normal range.
These medications currently cost around $1,400 per month in the United States, unless patients can access initiatives such as company coupons. Some insurers, including state-subsidized Medicare and Medicaid, don’t cover the new medications.
Dr. O’Donnell said, “More accessibility for more people would help in the big picture.”
Other patients stop taking GLP-1 agonists because they experience side effects, such as nausea.
“Gastrointestinal complaints ... are the number one reason for people to come off the medication,” said Disha Narang, MD, an endocrinologist and obesity medicine specialist at Northwestern Medicine Lake Forest (Ill.) Hospital.
“It is an elective therapy, so it is not mandatory that someone take it. So if they are not feeling well or they are sick, then that’s a major reason for coming off of it,” she said.
Dan Bessesen, MD, professor of medicine at the University of Colorado at Denver, Aurora, and chief of endocrinology, agreed.
Patients are unlikely to stay on these medications if they feel nauseous or experience vomiting, he said. Although he noted there are options to try to counter this, such as starting patients on a very low dose of the drug and up-titrating slowly. This method requires good coordination between the patient and physician.
Goutham Rao, MD, a professor of medicine at Case Western Reserve University and head of the weight-loss initiative Fitter Me at University Hospitals, both in Cleveland, said that prior to prescribing GLP-1 agonists for weight loss, he sets four basic, nonnegotiable goals for patients: “to have breakfast within 30 minutes of getting up, to drink just water, no food or drink after 7:00 p.m. except for water, and 30 minutes of continuous exercise per day, which is typically, for older clientele, walking.”
This, he said, can help establish good habits because if “patients are not engaged psychologically in weight loss ... they expect the medication to do [all] the work.”
Most regain weight after stopping obesity medications
As Ms. Hartman’s story illustrates, discontinuing the medications often leads to weight regain.
“Without the medicine, there are a variety of things that will happen. Appetite will tend to increase, and so [patients] will gradually tend to eat more over time,” Dr. Bessesen noted.
“So it may take a long time for the weight regain to happen, but in every study where an obesity medicine has been used, and then it is stopped, the weight goes back to where it was on lifestyle alone,” he added.
In the STEP 1 trial, almost 2,000 patients who were either overweight or living with obesity were randomized 2:1 to semaglutide, titrated up to 2.4 mg each week by week 16, or placebo in addition to lifestyle modification. After 68 weeks, those in the semaglutide group had a mean weight loss of 14.9%, compared with 2.4% in the placebo group.
Patients were also followed in a 1-year extension of the trial, published in Diabetes, Obesity, and Metabolism.
Within 1 year of stopping treatment, participants regained two thirds of the weight they had initially lost.
Hence, Dr. Bessesen stressed that a total rethink of how obesity is approached is needed among most physicians.
“I think in the future treating obesity with medications should be like treating hypertension and diabetes, something most primary care doctors are comfortable doing, but that’s going to take a little work and practice on the part of clinicians to really have a comfortable conversation about risks, and benefits, with patients,” he said.
“I would encourage primary care doctors to learn more about the treatment of obesity, and learn more about bias and stigma, and think about how they can deliver care that is compassionate and competent,” he concluded.
A version of this article first appeared on Medscape.com.
After bariatric surgery in 2014, Kristal Hartman still struggled to manage her weight long term. It took her over a year to lose 100 pounds, a loss she initially maintained, but then gradually her body mass index (BMI) started creeping up again.
“The body kind of has a set point, and you have to constantly trick it because it is going to start to gain weight again,” Ms. Hartman, who is on the national board of directors for the Obesity Action Coalition, said in an interview.
So, 2.5 years after her surgery, Ms. Hartman began weekly subcutaneous injections of the glucagonlike peptide–1 (GLP-1) agonist semaglutide, a medication that is now almost infamous because of its popularity among celebrities and social media influencers.
Branded as Ozempic for type 2 diabetes and Wegovy for obesity, both contain semaglutide but in slightly different doses. The popularity of the medication has led to shortages for those living with type 2 diabetes and/or obesity. And other medications are waiting in the wings that work on GLP-1 and other hormones that regulate appetite, such as the twincretin tirzepatide (Mounjaro), another weekly injection, approved by the Food and Drug Administration in May 2022 for type 2 diabetes and awaiting approval for obesity.
Ms. Hartman said taking semaglutide helped her not only lose weight but also “curb [her] obsessive thoughts over food.” To maintain a BMI within the healthy range, as well as taking the GLP-1 agonist, Ms. Hartman relies on other strategies, including exercise, and mental health support.
“Physicians really need to be open to these FDA-approved medications as one of many tools in the toolbox for patients with obesity. It’s just like any other chronic disease state, when they are thinking of using these, they need to think about long-term use ... in patients who have obesity, not just [among those people] who just want to lose 5-10 pounds. That’s not what these drugs are designed for. They are for people who are actually living with the chronic disease of obesity every day of their lives,” she emphasized.
On average, patients lose 25%-40% of their total body weight following bariatric surgery, said Teresa LeMasters, MD, president of the American Society for Metabolic & Bariatric Surgery. However, there typically is a “small” weight regain after surgery.
“For most patients, it is a small 5-10 pounds, but for some others, it can be significant,” said Dr. LeMasters, a bariatric surgeon at UnityPoint Clinic, Des Moines, Iowa.
“We do still see some patients– anywhere from 10% to 30% – who will have some [significant] weight regain, and so then we will look at that,” she noted. In those cases, the disease of obesity “is definitely still present.”
Medications can counter weight regain after surgery
For patients who don’t reach their weight loss goals after bariatric surgery, Dr. LeMasters said it’s appropriate to consider adding an anti-obesity medication. The newer GLP-1 agonists can lead to a loss of around 15% of body weight in some patients.
or even just to optimize their initial response to surgery if they are starting at a very, very severe point of disease,” she explained.
She noted, however, that some patients shouldn’t be prescribed GLP-1 agonists, including those with a history of thyroid cancer or pancreatitis.
Caroline M. Apovian, MD, codirector of the center for weight management and wellness and professor of medicine at Harvard Medical School, Boston, said in an interview that the physiology behind bariatric surgery and that of the newer obesity medications is somewhat aligned.
“In order to reduce ... body weight permanently you need adjustments. We learned that you need the adjustments of the hormones [that affect appetite, such as GLP-1], and that’s why bariatric surgery works because ... [it] provides the most durable and the most effective treatment for obesity ... because [with surgery] you are adjusting the secretion and timing of many of the hormones that regulate body weight,” she explained.
So, when people are taking GLP-1 agonists for obesity, with or without surgery, these medications “are meant and were approved by the FDA to be taken indefinitely. They are not [for the] short term,” Dr. Apovian noted.
Benjamin O’Donnell, MD, an associate professor at Ohio State University Wexner Medical Center, Columbus, agreed that the newer anti-obesity medications can be very effective; however, he expressed uncertainty about prescribing these medications for years and years.
“If somebody has obesity, they need medicine to help them manage appetite and maintain a lower, healthier weight. It would make sense that they would just stay on the medicine,” he noted.
But he qualified: “I have a hard time committing to saying that someone should take this medication for the rest of their life. Part of my hesitation is that the medications are expensive, so we’ve had a hard time with people staying on them, mostly because of insurance formulary changes.”
Why stop the medications? Side effects and lack of insurance coverage
Many people have to discontinue these newer medications for that exact reason.
When Ms. Hartman’s insurance coverage lapsed, she had to go without semaglutide for a while.
“At that time, I absolutely gained weight back up into an abnormal BMI range,” Ms. Hartman said. When she was able to resume the medication, she lost weight again and her BMI returned to normal range.
These medications currently cost around $1,400 per month in the United States, unless patients can access initiatives such as company coupons. Some insurers, including state-subsidized Medicare and Medicaid, don’t cover the new medications.
Dr. O’Donnell said, “More accessibility for more people would help in the big picture.”
Other patients stop taking GLP-1 agonists because they experience side effects, such as nausea.
“Gastrointestinal complaints ... are the number one reason for people to come off the medication,” said Disha Narang, MD, an endocrinologist and obesity medicine specialist at Northwestern Medicine Lake Forest (Ill.) Hospital.
“It is an elective therapy, so it is not mandatory that someone take it. So if they are not feeling well or they are sick, then that’s a major reason for coming off of it,” she said.
Dan Bessesen, MD, professor of medicine at the University of Colorado at Denver, Aurora, and chief of endocrinology, agreed.
Patients are unlikely to stay on these medications if they feel nauseous or experience vomiting, he said. Although he noted there are options to try to counter this, such as starting patients on a very low dose of the drug and up-titrating slowly. This method requires good coordination between the patient and physician.
Goutham Rao, MD, a professor of medicine at Case Western Reserve University and head of the weight-loss initiative Fitter Me at University Hospitals, both in Cleveland, said that prior to prescribing GLP-1 agonists for weight loss, he sets four basic, nonnegotiable goals for patients: “to have breakfast within 30 minutes of getting up, to drink just water, no food or drink after 7:00 p.m. except for water, and 30 minutes of continuous exercise per day, which is typically, for older clientele, walking.”
This, he said, can help establish good habits because if “patients are not engaged psychologically in weight loss ... they expect the medication to do [all] the work.”
Most regain weight after stopping obesity medications
As Ms. Hartman’s story illustrates, discontinuing the medications often leads to weight regain.
“Without the medicine, there are a variety of things that will happen. Appetite will tend to increase, and so [patients] will gradually tend to eat more over time,” Dr. Bessesen noted.
“So it may take a long time for the weight regain to happen, but in every study where an obesity medicine has been used, and then it is stopped, the weight goes back to where it was on lifestyle alone,” he added.
In the STEP 1 trial, almost 2,000 patients who were either overweight or living with obesity were randomized 2:1 to semaglutide, titrated up to 2.4 mg each week by week 16, or placebo in addition to lifestyle modification. After 68 weeks, those in the semaglutide group had a mean weight loss of 14.9%, compared with 2.4% in the placebo group.
Patients were also followed in a 1-year extension of the trial, published in Diabetes, Obesity, and Metabolism.
Within 1 year of stopping treatment, participants regained two thirds of the weight they had initially lost.
Hence, Dr. Bessesen stressed that a total rethink of how obesity is approached is needed among most physicians.
“I think in the future treating obesity with medications should be like treating hypertension and diabetes, something most primary care doctors are comfortable doing, but that’s going to take a little work and practice on the part of clinicians to really have a comfortable conversation about risks, and benefits, with patients,” he said.
“I would encourage primary care doctors to learn more about the treatment of obesity, and learn more about bias and stigma, and think about how they can deliver care that is compassionate and competent,” he concluded.
A version of this article first appeared on Medscape.com.