User login
Cell-free DNA screening for women at low risk for fetal aneuploidy
CASE: Low-risk patient requests cell-free DNA screening
Ms. Smith is a 25-year-old woman (G1P0) presenting at 10 weeks’ gestation for her first prenatal visit. She requests cell-free DNA (cfDNA) screening to test for fetal aneuploidy. You explain that the current recommendations are for traditional screening, and inform her that her insurance may not cover the cost of cfDNA screening. She is anxious to learn the sex of her fetus as early as possible, and indicates that she would like to pursue cfDNA. After further discussion of the pros and cons, you order the test.
Prenatal screening is currently recommended in pregnancy for a number of genetic disorders, chromosomal aneuploidy, and structural birth defects in the fetus, regardless of maternal age or family history. There is a broad range of sonographic and maternal serum-based options available for carrying out aneuploidy risk assessment in the first and/or second trimester.
In addition, cfDNA screening for fetal aneuploidy has been clinically available since 2011 and has seen tremendous uptake, particularly in the high-risk population. Recent data indicate that cfDNA screening likewise has very high sensitivity and specificity for trisomy 21 in the low-risk population.1,2
Many low-risk patients are asking providers about the pros and cons of cfDNA screening, and the appropriateness of this test as a primary screen, including in low-risk patients, is the focus of this article.
What is cfDNA?
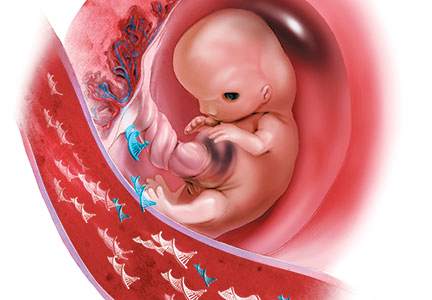
cfDNA consists of small (<200 base pairs) fragments of DNA that are present in the maternal serum. After 10 weeks of gestation, about 10% of the total circulating cfDNA in the maternal serum is derived from the placenta and can therefore be used to test for fetal disorders (FIGURE).3
Although cfDNA screening has been reported to be possible for many different types of genetic conditions, such as RhD type and single-gene disorders such as achondroplasia,4 most clinical testing is done for fetal chromosomal disorders, including trisomies 13, 18, and 21 and the sex chromosomes. In addition, some laboratories provide testing for other trisomies (16 and 22), as well as some of the microdeletion syndromes (22q11.2, 1p36, Prader Willi syndrome, and others).5
Analysis of cfDNA to assess the risk for aneuploidy is done using a number of different approaches; these generally all include next-generation sequencing with advanced bioinformatics analyses.3,6–9 Although the laboratories use somewhat different techniques, all of them share very high sensitivity and specificity for detection of trisomy 21 (TABLE 1).10
Sensitivities for trisomy 13 and sex chromosomal abnormalities are somewhat lower, but the specificity is greater than 99% for each condition, meaning that false-positive rates are very low.
The accuracy of cfDNA in identifying chromosomal aneuploidy depends on several factors, including the relative amount of fetal to maternal DNA, the chance that a chromosome abnormality is present (that is, the risk based on maternal age or results of other screening), and other factors such as the presence of twins or a nonviable second fetus, or the presence of placental mosaicism.
Because of these variables, both false-positive and false-negative results can occur, and the test is not diagnostic but rather is considered a screening test. A positive result does not mean that the fetus is definitely affected with aneuploidy.
What are the advantages of cfDNA screening for low-risk patients?
There are several benefits of cfDNA screening versus traditional screening or diagnostic testing, which are the other options available (TABLE 2). For Down syndrome, the detection rate is higher and the false-positive rate is lower than that seen with traditional aneuploidy screening using serum analytes and nuchal translucency ultrasonography.1,2
TABLE 2 Pros and cons of cfDNA screening in low-risk patients
Pros
- High detection rate and very low false-positive rate
- Can be performed any time after 10 weeks’ gestation
- Requires a single blood test at any gestational age
- Results presented in simple “Yes” or “No” format
- As with other screening tests, cfDNA provides a noninvasive determination of risk
Cons
- Tests for a limited range of conditions, which are rare in low-risk patients
- Is not as comprehensive or definitive as diagnostic testing with amniocentesis or chorionic villus sampling
- Results do not adjust for patient’s prior risk
- Positive results require calculation and interpretation of positive predictive value by provider
- Low fetal DNA and other factors can lead to test failure in some cases
- Cannot be used with vanishing twin
- Can reveal unsuspected maternal conditions of uncertain significance
The test can be done any time after 10 weeks’ gestation without the narrow gestational-age windows required or the need for accurate gestational age determination using traditional screening to accurately interpret results. cfDNA screening involves a single blood test that does not require integration with multiple serum markers or ultrasound findings. Finally, results are generally presented in a simple “Yes” or “No” format that is easy for providers and patients to understand.
CASE Continued
Your patient’s results are positive for trisomy 13. Her understanding is that the test is more than 99% accurate, and she interprets this to mean that the chance of trisomy 13 in her fetus is more than 99%. She is distraught and asks about pregnancy termination.
What are the limitations of cfDNA screening?
Similar to other noninvasive screening tests, cfDNA screening does not carry direct risk to the pregnancy. However, there are limitations to this testing. As a result, the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) recently have stated that traditional screening is the most appropriate option for most women.11,12
One reason that cfDNA screening may not be the best choice for low-risk women is that Down syndrome is quite uncommon in this group, so cfDNA screening is a very precise test for a rare condition. Traditional multiple marker screening, on the other hand, is more effective at signaling risk for the broad range of adverse perinatal outcomes that can affect a pregnancy, including other structural birth defects, as well as such obstetric complications as preterm birth, preeclampsia, and fetal growth restriction.13,14
Many women who undergo cfDNA screening are under the impression that they have had a definitive test for all birth defects when, in fact, the coverage of cfDNA for all possible birth defects in a low-risk woman is very limited; her residual risk for a birth defect is little changed by a normal cfDNA result.
The ease of obtaining a blood sample for cfDNA screening is an advantage of the test. However, because it is simple to perform, it often is done with inadequate pretest counseling or consideration. Just because the test is easily obtained does not negate the need for adequate discussion to assure that each woman understands what the test can and cannot measure, and the possible outcomes of testing.
Another perceived benefit of cfDNA screening is the simple presentation of results. While reports vary, they generally provide very dichotomized results. Aneuploidy risk is reported as “Positive” or “Negative,” or as “Detected” or “Not detected.”
Some laboratories report the chance of aneuploidy; this is almost always stated to be more than 99% in patients at increased risk, and less than 1 in 10,000 in patients at low risk.
All of these results suggest a near diagnostic certainty. However, this reporting is oversimplified and misleading, as it does not account for each patient’s prior or background risk. The chance that a positive result is a true positive is very different in a 20-year-old versus a 35-year-old woman, yet the reports do not reflect this difference in positive predictive value (PPV). See TABLE 1.
Accurate interpretation of risk for the individual patient, therefore, requires calculation by the provider; this can be done through an online calculator available through the Perinatal Quality Foundation (www.perina talquality.org).
CASE Continued
You explain to your patient that the chance her fetus has trisomy 13 is far lower than 99%, based in part on the very low prior risk given her age. You calculate the PPV using an online calculator, which estimates that there is only a 7% chance that this is a true positive result.
As mentioned earlier, there has been a tremendously rapid uptake of cfDNA screening. Given wider use by practitioners not as familiar with the complexities of genetic testing and statistical analysis, misunderstanding of the test characteristics carries risks if inappropriate recommendations or decisions are made or actions taken.
Most low-risk patients do not request or desire diagnostic testing. It is important during pretest counseling to explain that cfDNA cannot detect all significant chromosomal aneuploidies. Some serious abnormalities will be undetected; therefore, some women may prefer more comprehensive prenatal testing (TABLE 3).
TABLE 3 Checklist for pretest counseling for cfDNA28
- cfDNA screening is the most accurate screening test for trisomy 21
- cfDNA is a screening test, and false-positive and false-negative results can and do occur
- Diagnostic confirmation with chorionic villus sampling or amniocentesis is recommended for women with abnormal cfDNA results
- A negative cfDNA result decreases risk but does not rule out trisomy 21 and other chromosomal conditions
- cfDNA does not test for all chromosomal conditions
- Women who desire definitive information about chromosome conditions in the pregnancy should consider diagnostic testing with chorionic villus sampling or amniocentesis
- All genetic testing is optional. Whether a woman chooses to have a screening test, a diagnostic test, or no testing is a personal decision; any are reasonable options for any pregnant woman.
ACOG recommends that diagnostic testing should be available to all pregnant women, regardless of age.15 In prenatal series, trisomies 13, 18, and 21 make up approximately two-thirds of all clinically significant aneuploidies.16,17 Given that cfDNA detects only these aneuploidies, the other third will not be identified prenatally in patients who choose cfDNA. Traditional aneuploidy screening has been demonstrated to detect a broader range of these less common but clinically important chromosomal abnormalities.18
In one study of women found to be at increased risk based on traditional multiple marker screening, if cfDNA were chosen instead of diagnostic testing, 17% of the aneuploidies present in this group would not have been detected.18 Of all high-risk women in this study, 2%, or 1 in 50, had a chromosomal abnormality detectable by amniocentesis but not with cfDNA.
Successful tests require adequate placental DNA
Accurate interpretation of cfDNA screening also requires that an adequate quantity of placental DNA be present; this is often referred to as the “fetal fraction.” In some cases, the placental DNA volume is too low for accurate analysis, particularly in obese patients and women with specific chromosomal abnormalities.
Some laboratories measure this and do not report a result if the fetal fraction is below a specific cut-off, typically about 4%. Other laboratories do not measure or exclude cases with too little fetal DNA, raising concern that this could result in missing cases of aneuploidy. It has been noted that a placental DNA fraction of less than 8% is associated with less accurate test results, even if results are returned.8
Low fetal fraction also has been associated with maternal obesity, and in one study cfDNA failed to provide a result in 20% of women weighing more than 250 lb and 50% of women weighing more than 350 lb.19 Therefore, cfDNA is not the best option for obese women (TABLE 4).
TABLE 4 Appropriateness of cfDNA screeningin specific clinical circumstances
Optimal candidates for cfDNA screening
- High risk for trisomy based on maternal age (≥35 years)
- Ultrasound findings suggesting trisomy 13, 18, or 21
- History of prior pregnancy with trisomy 13, 18, or 21
- Positive traditional screening test
- Parental balanced Robertsonian translocation associated with risk for trisomy 13 or 21
Less optimal candidates
- Low risk for trisomy based on age and/or low risk traditional screening
- Ultrasound structural anomalies other than those specifically suggesting trisomy 13, 18, or 21
- High risk for nonchromosomal genetic disorder
- Comprehensive genetic diagnosis desired
- Maternal malignancy
- Maternal organ transplant
- Maternal sex chromosomal mosaicism or other chromosomal abnormality
- Maternal obesity
- Gestational age <10 weeks
While the free fraction is relatively constant from 10 to 22 weeks’ gestation, it is lower earlier than 10 weeks’ gestation and less likely to provide a result. For this reason, the test should not be attempted before 10 weeks’ gestation.
Recent evidence indicates that low fetal DNA fraction is associated with some chromosome abnormalities. Given this association, women with failed cfDNA results should be counseled and offered appropriate follow-up. As the association appears to be greater for trisomies 13 and 18, and triploidy, a careful ultrasound is likely to detect abnormalities in many such cases. However, it also is appropriate to offer the option of diagnostic testing, given the very high risk.
A repeat cfDNA test will be successful in some cases. Whether the patient chooses to attempt cfDNA again may depend in part on maternal body mass index (BMI), as well as gestational age—a patient at a more advanced gestation may not wish to delay obtaining definitive information given the high risk.
cfDNA screening has a low false-positive rate
One of the greatest benefits of cfDNA screening is a lower false-positive rate than is reported with traditional screening. However, when “no results” cases are also considered, the percentage of patients who require follow-up after cfDNA is close to that of traditional screening.
The chance of test failure is reported to be 0.9% to 8.1%,7,9,10 and varies in part by whether the laboratory measures fetal fraction and requires a minimum concentration.
A recent meta-analysis estimated the overall test failure rate at 3%.10 When comparing cfDNA to traditional screening, if “no results” cases are included with the “screen positive” group, the benefits of cfDNA over traditional screening are much less clear, particularly in a low-risk population.
ACOG: Offer traditional multiple-marker screening first
While multiple marker and cfDNA screening have differing performance characteristics, there are no data to support doing both tests concurrently. In fact, in a recent survey of nearly 200 women presented with different testing scenarios, women found it preferable and more reassuring to have a positive traditional screen followed by normal cfDNA results, rather that discrepant results of the 2 tests done concurrently.20
For many reasons, the approach recommended by ACOG and SMFM is to offer traditional multiple-marker screening first, and cfDNA screening or diagnostic testing as a follow-up for patients that screen positive. In that scenario, the benefits and limitations of diagnostic testing versus follow-up with cfDNA screening should be explained carefully.
In all patients who have a positive cfDNA result, diagnostic testing for confirmation should be offered and strongly recommended prior to pregnancy termination if that is considered. Even if a structural abnormality is present and a true positive result is highly likely, karyotyping is important to determine if there may be an inherited translocation putting subsequent pregnancies at higher risk.
Components of pretest counseling
A woman of any age can have a fetus with trisomy or another chromosomal abnormality, and some women prefer diagnostic testing or no testing regardless of age. It is therefore appropriate to offer diagnostic testing, screening, or the option of no testing to all women.
Recent studies have demonstrated that providing well-informed access to all prenatal tests results in more informed choices and no increase in uptake of invasive testing.22 However, the offer of prenatal testing requires discussion of the pros and cons of all test options, including the detection rates of all significant abnormalities, the screen positive rates, and recommended follow-up if an abnormal result is obtained. See TABLE 4.
Cost-effectiveness
Although the detection rate of cfDNA for trisomy 21 is higher than that of traditional screening, the detection rate of traditional screening is also quite high at lower cost. For low-risk women, therefore, traditional screening provides a less expensive alternative to cfDNA. Because aneuploidy is rare in low-risk patients, the residual chance of aneuploidy after a normal traditional screen is very low, and the cost per additional case of Down syndrome detected by cfDNA is very high.
In one study, this was estimated at $3.6 million.23 These authors suggested that, at present, cfDNA is optimally used as a secondary screen for high-risk women. Other cost analyses also have demonstrated that the most cost-effective strategy is a model in which cfDNA is used as a follow-up test in patients found to be screen positive by traditional screening.15,24 A recent cost utility analysis compared outcomes of 6 approaches to prenatal screening, including sequential screening, cfDNA screening, nuchal translucency only, and diagnostic testing with microarray (alone, in combination, or in sequence).
The clinical outcomes included fetal abnormalities detected, taking into account all chromosomal abnormalities, as well as failed cfDNA tests. For younger women (<40 yr), traditional sequential screening provided the highest detection of all abnormalities and was the optimal testing strategy, while cfDNA was preferable for women aged 40 or older, given the higher prevalence of trisomy 21.20
Incidental findings
Given that the cfDNA in maternal serum is a mixture of maternal and placental DNA, a number of biologic phenomena can cause a false-positive cfDNA result. In many cases, these false-positives reveal unanticipated or unexpected maternal conditions and information that the woman may have preferred not to know. A few cases of maternal malignancies with chromosomal abnormalities within the tumor have been reported in patients with false-positive cfDNA results.26
These case reports have raised the question about the need for further evaluation for maternal malignancy in women with false-positive results. Maternal genetic disorders also can cause false-positive results, and may lead to unanticipated detection of adult-onset conditions. In some cases, positive results for sex chromosomal aneuploidy can occur in pregnant women who themselves have a sex chromosomal abnormality, often in mosaic form and previously undiagnosed.27
Again, this has led to discussion of the possible health benefit of karyotyping women who have a false-positive cfDNA result to rule out a mosaic chromosomal abnormality in the mother.
At this time, the clinical utility of such investigations is unknown and there are no recommendations regarding appropriate follow-up for such cases.
CASE Resolved
Given the results of her cfDNA screening, your patient opts to undergo diagnostic testing. In that testing, trisomy 13 is ruled out and she goes on to have a healthy daughter.
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Bianchi DW, Parker RL, Wentworth J, et al. DNA sequencing versus standard prenatal aneuploidy screening. N Engl J Med. 2014;370(9):799–808.
- Norton ME, Jacobsson B, Swamy GK, et al. Cell-free DNA analysis for noninvasive examination of trisomy. N Engl J Med. 2015;372(17):1589–1597.
- Norton ME, Brar H, Weiss J, et al. Non-Invasive Chromosomal Evaluation (NICE) study: results of a multicenter prospective cohort study for detection of fetal trisomy 21 and trisomy 18. Am J Obstet Gynecol. 2012;207(2):137.e1–e8.
- Chitty LS, Mason S, Barrett AN, et al. Non-invasive prenatal diagnosis of achondroplasia and thanatophoric dysplasia: next-generation sequencing allows for a safer, more accurate, and comprehensive approach. Prenat Diagn. 2015;35(7):656–662.
- Wapner RJ, Babiarz JE, Levy B, et al. Expanding the scope of noninvasive prenatal testing: detection of fetal microdeletion syndromes. Am J Obstet Gynecol. 2015;212(3):332.e1–e9.
- Bianchi DW, Platt LD, Goldberg JD, Abuhamad AZ, Sehnert AJ, Rava RP. Genome-wide fetal aneuploidy detection by maternal plasma DNA sequencing. Obstet Gynecol. 2012;119(5):890–901.
- Palomaki GE, Kloza EM, Lambert-Messerlian GM, et al. DNA sequencing of maternal plasma to detect Down syndrome: an international clinical validation study. Genet Med. 2011;13(11):913–920.
- Sparks AB, Wang ET, Struble CA, et al. DNA sequencing of maternal plasma to detect Down syndrome: an international clinical validation study [abstract]. Proceedings of the ISPD 16th International Conference on Prenatal Diagnosis and Therapy; Miami, Florida; June 3–6, 2012. Prenat Diagn. 2012;32(suppl 1):s3–s9.
- Zimmermann B, Hill M, Gemelos G, et al. Noninvasive prenatal aneuploidy testing of chromosomes 13, 18, 21, X, and Y, using targeted sequencing of polymorphic loci. Prenat Diagn. 2012;32(13):1233–1241.
- Gil MM, Quezada MS, Revello R, Akolekar R, Niclaides KH. Analysis of cell-free DNA in maternal blood in screening for fetal aneuploidies: updated meta-analysis. Ultrasound Obstet Gynecol. 2015;45(3):249–266.
- American College of Obstetricians and Gynecologists. Committee Opinion No. 640: cell-free DNA screening for fetal aneuploidy. Obstet Gynecol. 2015;126(3):e31–e37.
- Society for Maternal-Fetal Medicine (SMFM) Publications Committee. Prenatal aneuploidy screening using cell-free DNA. Am J Obstet Gynecol. 2015;212(6):711–716.
- Baer RJ, Currier RJ, Norton ME, et al. Obstetric, perinatal, and fetal outcomes in pregnancies with false-positive integrated screening results. Obstet Gynecol. 2014;123(3):603–609.
- Dugoff L; Society for Maternal-Fetal Medicine. First- and second-trimester maternal serum markers for aneuploidy and adverse obstetric outcomes. Obstet Gynecol. 2010;115(5):1052–1061.
- American College of Obstetricians and Gynecologists. Practice bulletin No. 88: invasive prenatal testing for aneuploidy. Obstet Gynecol. 2007;110(6):1459–1467.
- Alamillo CM, Krantz D, Evans M, Fiddler M, Pergament E. Nearly a third of abnormalities found after first-trimester screening are different than expected: 10-year experience from a single center. Prenat Diagn. 2013;33(3):251–256.
- Wellesley D, Dolk H, Boyd PA, et al. Rare chromosome abnormalities, prevalence and prenatal diagnosis rates from population-based congenital anomaly registers in Europe. Eur J Hum Genet. 2012;20(5):521–526.
- Norton ME, Jelliffe-Pawlowski LL, Currier RJ. Chromosome abnormalities detected by current prenatal screening and noninvasive prenatal testing. Obstet Gynecol. 2014;124(5):979–986.
- Ashoor G, Syngelaki A, Poon LC, Rezende JC, Nicolaides KH. Fetal fraction in maternal plasma cell-free DNA at 11-13 weeks’ gestation: relation to maternal and fetal characteristics. Ultrasound Obstet Gynecol. 2013;41(1):26–32.
- Kaimal AJ, Norton ME, Kuppermann M. Prenatal testing in the genomic age: clinical outcomes, quality of life, and costs. Obstet Gynecol. 2015;126(4):737–746.
- Wapner RJ, Martin CL, Levy B, et al. Chromosomal microarray versus karyotyping for prenatal diagnosis. N Engl J Med. 2012;367(23):2175–2184.
- Kuppermann M, Pena S, Bishop JT, et al. Effect of enhanced information, values clarification, and removal of financial barriers on use of prenatal genetic testing: a randomized clinical trial. JAMA. 2014;312(12):1210–1217.
- Cuckle H, Benn P, Pergament E. Maternal cfDNA screening for Down syndrome—a cost sensitivity analysis. Prenat Diagn. 2013;33(7):636–642.
- Beulen L, Grutters JPC, Faas BH, Feenstra I, van Vugt JMG, Bekker MN. The consequences of implementing non-invasive prenatal testing in Dutch national health care: a cost-effectiveness analysis. Eur J Obstet Gynecol Reprod Biol. 2014;182:53–61.
- Okun N, Teitelbaum M, Huang T, Dewa CS, Hoch JS. The price of performance: a cost and performance analysis of the implementation of cell-free fetal DNA testing for Down syndrome in Ontario, Canada: Cost and performance analysis of cfDNA testing for Down syndrome in Ontario. Prenat Diagn. 2014;34(4):350–356.
- Bianchi DW, Chudova D, Sehnert AJ, et al. Noninvasive prenatal testing and incidental detection of occult maternal malignancies. JAMA. 2015;314(2):162–169.
- Wang Y, Chen Y, Tian F, et al. Maternal mosaicism is a significant contributor to discordant sex chromosomal aneuploidies associated with noninvasive prenatal testing. Clin Chem. 2014;60(1):251–259.
- Norton ME, Jelliffe-Pawlowski LL, Currier RJ. Chromosome abnormalities detected by current prenatal screening and noninvasive prenatal testing. Obstet Gynecol. 2014;124(5):979–986Society for Maternal-Fetal Medicine (SMFM) Publications Committee. Prenatal aneuploidy screening using cell-free DNA. Am J Obstet Gynecol. 2015;212(6):711–716.
CASE: Low-risk patient requests cell-free DNA screening
Ms. Smith is a 25-year-old woman (G1P0) presenting at 10 weeks’ gestation for her first prenatal visit. She requests cell-free DNA (cfDNA) screening to test for fetal aneuploidy. You explain that the current recommendations are for traditional screening, and inform her that her insurance may not cover the cost of cfDNA screening. She is anxious to learn the sex of her fetus as early as possible, and indicates that she would like to pursue cfDNA. After further discussion of the pros and cons, you order the test.
Prenatal screening is currently recommended in pregnancy for a number of genetic disorders, chromosomal aneuploidy, and structural birth defects in the fetus, regardless of maternal age or family history. There is a broad range of sonographic and maternal serum-based options available for carrying out aneuploidy risk assessment in the first and/or second trimester.
In addition, cfDNA screening for fetal aneuploidy has been clinically available since 2011 and has seen tremendous uptake, particularly in the high-risk population. Recent data indicate that cfDNA screening likewise has very high sensitivity and specificity for trisomy 21 in the low-risk population.1,2
Many low-risk patients are asking providers about the pros and cons of cfDNA screening, and the appropriateness of this test as a primary screen, including in low-risk patients, is the focus of this article.
What is cfDNA?
cfDNA consists of small (<200 base pairs) fragments of DNA that are present in the maternal serum. After 10 weeks of gestation, about 10% of the total circulating cfDNA in the maternal serum is derived from the placenta and can therefore be used to test for fetal disorders (FIGURE).3
Although cfDNA screening has been reported to be possible for many different types of genetic conditions, such as RhD type and single-gene disorders such as achondroplasia,4 most clinical testing is done for fetal chromosomal disorders, including trisomies 13, 18, and 21 and the sex chromosomes. In addition, some laboratories provide testing for other trisomies (16 and 22), as well as some of the microdeletion syndromes (22q11.2, 1p36, Prader Willi syndrome, and others).5
Analysis of cfDNA to assess the risk for aneuploidy is done using a number of different approaches; these generally all include next-generation sequencing with advanced bioinformatics analyses.3,6–9 Although the laboratories use somewhat different techniques, all of them share very high sensitivity and specificity for detection of trisomy 21 (TABLE 1).10
Sensitivities for trisomy 13 and sex chromosomal abnormalities are somewhat lower, but the specificity is greater than 99% for each condition, meaning that false-positive rates are very low.
The accuracy of cfDNA in identifying chromosomal aneuploidy depends on several factors, including the relative amount of fetal to maternal DNA, the chance that a chromosome abnormality is present (that is, the risk based on maternal age or results of other screening), and other factors such as the presence of twins or a nonviable second fetus, or the presence of placental mosaicism.
Because of these variables, both false-positive and false-negative results can occur, and the test is not diagnostic but rather is considered a screening test. A positive result does not mean that the fetus is definitely affected with aneuploidy.
What are the advantages of cfDNA screening for low-risk patients?
There are several benefits of cfDNA screening versus traditional screening or diagnostic testing, which are the other options available (TABLE 2). For Down syndrome, the detection rate is higher and the false-positive rate is lower than that seen with traditional aneuploidy screening using serum analytes and nuchal translucency ultrasonography.1,2
TABLE 2 Pros and cons of cfDNA screening in low-risk patients
Pros
- High detection rate and very low false-positive rate
- Can be performed any time after 10 weeks’ gestation
- Requires a single blood test at any gestational age
- Results presented in simple “Yes” or “No” format
- As with other screening tests, cfDNA provides a noninvasive determination of risk
Cons
- Tests for a limited range of conditions, which are rare in low-risk patients
- Is not as comprehensive or definitive as diagnostic testing with amniocentesis or chorionic villus sampling
- Results do not adjust for patient’s prior risk
- Positive results require calculation and interpretation of positive predictive value by provider
- Low fetal DNA and other factors can lead to test failure in some cases
- Cannot be used with vanishing twin
- Can reveal unsuspected maternal conditions of uncertain significance
The test can be done any time after 10 weeks’ gestation without the narrow gestational-age windows required or the need for accurate gestational age determination using traditional screening to accurately interpret results. cfDNA screening involves a single blood test that does not require integration with multiple serum markers or ultrasound findings. Finally, results are generally presented in a simple “Yes” or “No” format that is easy for providers and patients to understand.
CASE Continued
Your patient’s results are positive for trisomy 13. Her understanding is that the test is more than 99% accurate, and she interprets this to mean that the chance of trisomy 13 in her fetus is more than 99%. She is distraught and asks about pregnancy termination.
What are the limitations of cfDNA screening?
Similar to other noninvasive screening tests, cfDNA screening does not carry direct risk to the pregnancy. However, there are limitations to this testing. As a result, the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) recently have stated that traditional screening is the most appropriate option for most women.11,12
One reason that cfDNA screening may not be the best choice for low-risk women is that Down syndrome is quite uncommon in this group, so cfDNA screening is a very precise test for a rare condition. Traditional multiple marker screening, on the other hand, is more effective at signaling risk for the broad range of adverse perinatal outcomes that can affect a pregnancy, including other structural birth defects, as well as such obstetric complications as preterm birth, preeclampsia, and fetal growth restriction.13,14
Many women who undergo cfDNA screening are under the impression that they have had a definitive test for all birth defects when, in fact, the coverage of cfDNA for all possible birth defects in a low-risk woman is very limited; her residual risk for a birth defect is little changed by a normal cfDNA result.
The ease of obtaining a blood sample for cfDNA screening is an advantage of the test. However, because it is simple to perform, it often is done with inadequate pretest counseling or consideration. Just because the test is easily obtained does not negate the need for adequate discussion to assure that each woman understands what the test can and cannot measure, and the possible outcomes of testing.
Another perceived benefit of cfDNA screening is the simple presentation of results. While reports vary, they generally provide very dichotomized results. Aneuploidy risk is reported as “Positive” or “Negative,” or as “Detected” or “Not detected.”
Some laboratories report the chance of aneuploidy; this is almost always stated to be more than 99% in patients at increased risk, and less than 1 in 10,000 in patients at low risk.
All of these results suggest a near diagnostic certainty. However, this reporting is oversimplified and misleading, as it does not account for each patient’s prior or background risk. The chance that a positive result is a true positive is very different in a 20-year-old versus a 35-year-old woman, yet the reports do not reflect this difference in positive predictive value (PPV). See TABLE 1.
Accurate interpretation of risk for the individual patient, therefore, requires calculation by the provider; this can be done through an online calculator available through the Perinatal Quality Foundation (www.perina talquality.org).
CASE Continued
You explain to your patient that the chance her fetus has trisomy 13 is far lower than 99%, based in part on the very low prior risk given her age. You calculate the PPV using an online calculator, which estimates that there is only a 7% chance that this is a true positive result.
As mentioned earlier, there has been a tremendously rapid uptake of cfDNA screening. Given wider use by practitioners not as familiar with the complexities of genetic testing and statistical analysis, misunderstanding of the test characteristics carries risks if inappropriate recommendations or decisions are made or actions taken.
Most low-risk patients do not request or desire diagnostic testing. It is important during pretest counseling to explain that cfDNA cannot detect all significant chromosomal aneuploidies. Some serious abnormalities will be undetected; therefore, some women may prefer more comprehensive prenatal testing (TABLE 3).
TABLE 3 Checklist for pretest counseling for cfDNA28
- cfDNA screening is the most accurate screening test for trisomy 21
- cfDNA is a screening test, and false-positive and false-negative results can and do occur
- Diagnostic confirmation with chorionic villus sampling or amniocentesis is recommended for women with abnormal cfDNA results
- A negative cfDNA result decreases risk but does not rule out trisomy 21 and other chromosomal conditions
- cfDNA does not test for all chromosomal conditions
- Women who desire definitive information about chromosome conditions in the pregnancy should consider diagnostic testing with chorionic villus sampling or amniocentesis
- All genetic testing is optional. Whether a woman chooses to have a screening test, a diagnostic test, or no testing is a personal decision; any are reasonable options for any pregnant woman.
ACOG recommends that diagnostic testing should be available to all pregnant women, regardless of age.15 In prenatal series, trisomies 13, 18, and 21 make up approximately two-thirds of all clinically significant aneuploidies.16,17 Given that cfDNA detects only these aneuploidies, the other third will not be identified prenatally in patients who choose cfDNA. Traditional aneuploidy screening has been demonstrated to detect a broader range of these less common but clinically important chromosomal abnormalities.18
In one study of women found to be at increased risk based on traditional multiple marker screening, if cfDNA were chosen instead of diagnostic testing, 17% of the aneuploidies present in this group would not have been detected.18 Of all high-risk women in this study, 2%, or 1 in 50, had a chromosomal abnormality detectable by amniocentesis but not with cfDNA.
Successful tests require adequate placental DNA
Accurate interpretation of cfDNA screening also requires that an adequate quantity of placental DNA be present; this is often referred to as the “fetal fraction.” In some cases, the placental DNA volume is too low for accurate analysis, particularly in obese patients and women with specific chromosomal abnormalities.
Some laboratories measure this and do not report a result if the fetal fraction is below a specific cut-off, typically about 4%. Other laboratories do not measure or exclude cases with too little fetal DNA, raising concern that this could result in missing cases of aneuploidy. It has been noted that a placental DNA fraction of less than 8% is associated with less accurate test results, even if results are returned.8
Low fetal fraction also has been associated with maternal obesity, and in one study cfDNA failed to provide a result in 20% of women weighing more than 250 lb and 50% of women weighing more than 350 lb.19 Therefore, cfDNA is not the best option for obese women (TABLE 4).
TABLE 4 Appropriateness of cfDNA screeningin specific clinical circumstances
Optimal candidates for cfDNA screening
- High risk for trisomy based on maternal age (≥35 years)
- Ultrasound findings suggesting trisomy 13, 18, or 21
- History of prior pregnancy with trisomy 13, 18, or 21
- Positive traditional screening test
- Parental balanced Robertsonian translocation associated with risk for trisomy 13 or 21
Less optimal candidates
- Low risk for trisomy based on age and/or low risk traditional screening
- Ultrasound structural anomalies other than those specifically suggesting trisomy 13, 18, or 21
- High risk for nonchromosomal genetic disorder
- Comprehensive genetic diagnosis desired
- Maternal malignancy
- Maternal organ transplant
- Maternal sex chromosomal mosaicism or other chromosomal abnormality
- Maternal obesity
- Gestational age <10 weeks
While the free fraction is relatively constant from 10 to 22 weeks’ gestation, it is lower earlier than 10 weeks’ gestation and less likely to provide a result. For this reason, the test should not be attempted before 10 weeks’ gestation.
Recent evidence indicates that low fetal DNA fraction is associated with some chromosome abnormalities. Given this association, women with failed cfDNA results should be counseled and offered appropriate follow-up. As the association appears to be greater for trisomies 13 and 18, and triploidy, a careful ultrasound is likely to detect abnormalities in many such cases. However, it also is appropriate to offer the option of diagnostic testing, given the very high risk.
A repeat cfDNA test will be successful in some cases. Whether the patient chooses to attempt cfDNA again may depend in part on maternal body mass index (BMI), as well as gestational age—a patient at a more advanced gestation may not wish to delay obtaining definitive information given the high risk.
cfDNA screening has a low false-positive rate
One of the greatest benefits of cfDNA screening is a lower false-positive rate than is reported with traditional screening. However, when “no results” cases are also considered, the percentage of patients who require follow-up after cfDNA is close to that of traditional screening.
The chance of test failure is reported to be 0.9% to 8.1%,7,9,10 and varies in part by whether the laboratory measures fetal fraction and requires a minimum concentration.
A recent meta-analysis estimated the overall test failure rate at 3%.10 When comparing cfDNA to traditional screening, if “no results” cases are included with the “screen positive” group, the benefits of cfDNA over traditional screening are much less clear, particularly in a low-risk population.
ACOG: Offer traditional multiple-marker screening first
While multiple marker and cfDNA screening have differing performance characteristics, there are no data to support doing both tests concurrently. In fact, in a recent survey of nearly 200 women presented with different testing scenarios, women found it preferable and more reassuring to have a positive traditional screen followed by normal cfDNA results, rather that discrepant results of the 2 tests done concurrently.20
For many reasons, the approach recommended by ACOG and SMFM is to offer traditional multiple-marker screening first, and cfDNA screening or diagnostic testing as a follow-up for patients that screen positive. In that scenario, the benefits and limitations of diagnostic testing versus follow-up with cfDNA screening should be explained carefully.
In all patients who have a positive cfDNA result, diagnostic testing for confirmation should be offered and strongly recommended prior to pregnancy termination if that is considered. Even if a structural abnormality is present and a true positive result is highly likely, karyotyping is important to determine if there may be an inherited translocation putting subsequent pregnancies at higher risk.
Components of pretest counseling
A woman of any age can have a fetus with trisomy or another chromosomal abnormality, and some women prefer diagnostic testing or no testing regardless of age. It is therefore appropriate to offer diagnostic testing, screening, or the option of no testing to all women.
Recent studies have demonstrated that providing well-informed access to all prenatal tests results in more informed choices and no increase in uptake of invasive testing.22 However, the offer of prenatal testing requires discussion of the pros and cons of all test options, including the detection rates of all significant abnormalities, the screen positive rates, and recommended follow-up if an abnormal result is obtained. See TABLE 4.
Cost-effectiveness
Although the detection rate of cfDNA for trisomy 21 is higher than that of traditional screening, the detection rate of traditional screening is also quite high at lower cost. For low-risk women, therefore, traditional screening provides a less expensive alternative to cfDNA. Because aneuploidy is rare in low-risk patients, the residual chance of aneuploidy after a normal traditional screen is very low, and the cost per additional case of Down syndrome detected by cfDNA is very high.
In one study, this was estimated at $3.6 million.23 These authors suggested that, at present, cfDNA is optimally used as a secondary screen for high-risk women. Other cost analyses also have demonstrated that the most cost-effective strategy is a model in which cfDNA is used as a follow-up test in patients found to be screen positive by traditional screening.15,24 A recent cost utility analysis compared outcomes of 6 approaches to prenatal screening, including sequential screening, cfDNA screening, nuchal translucency only, and diagnostic testing with microarray (alone, in combination, or in sequence).
The clinical outcomes included fetal abnormalities detected, taking into account all chromosomal abnormalities, as well as failed cfDNA tests. For younger women (<40 yr), traditional sequential screening provided the highest detection of all abnormalities and was the optimal testing strategy, while cfDNA was preferable for women aged 40 or older, given the higher prevalence of trisomy 21.20
Incidental findings
Given that the cfDNA in maternal serum is a mixture of maternal and placental DNA, a number of biologic phenomena can cause a false-positive cfDNA result. In many cases, these false-positives reveal unanticipated or unexpected maternal conditions and information that the woman may have preferred not to know. A few cases of maternal malignancies with chromosomal abnormalities within the tumor have been reported in patients with false-positive cfDNA results.26
These case reports have raised the question about the need for further evaluation for maternal malignancy in women with false-positive results. Maternal genetic disorders also can cause false-positive results, and may lead to unanticipated detection of adult-onset conditions. In some cases, positive results for sex chromosomal aneuploidy can occur in pregnant women who themselves have a sex chromosomal abnormality, often in mosaic form and previously undiagnosed.27
Again, this has led to discussion of the possible health benefit of karyotyping women who have a false-positive cfDNA result to rule out a mosaic chromosomal abnormality in the mother.
At this time, the clinical utility of such investigations is unknown and there are no recommendations regarding appropriate follow-up for such cases.
CASE Resolved
Given the results of her cfDNA screening, your patient opts to undergo diagnostic testing. In that testing, trisomy 13 is ruled out and she goes on to have a healthy daughter.
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
CASE: Low-risk patient requests cell-free DNA screening
Ms. Smith is a 25-year-old woman (G1P0) presenting at 10 weeks’ gestation for her first prenatal visit. She requests cell-free DNA (cfDNA) screening to test for fetal aneuploidy. You explain that the current recommendations are for traditional screening, and inform her that her insurance may not cover the cost of cfDNA screening. She is anxious to learn the sex of her fetus as early as possible, and indicates that she would like to pursue cfDNA. After further discussion of the pros and cons, you order the test.
Prenatal screening is currently recommended in pregnancy for a number of genetic disorders, chromosomal aneuploidy, and structural birth defects in the fetus, regardless of maternal age or family history. There is a broad range of sonographic and maternal serum-based options available for carrying out aneuploidy risk assessment in the first and/or second trimester.
In addition, cfDNA screening for fetal aneuploidy has been clinically available since 2011 and has seen tremendous uptake, particularly in the high-risk population. Recent data indicate that cfDNA screening likewise has very high sensitivity and specificity for trisomy 21 in the low-risk population.1,2
Many low-risk patients are asking providers about the pros and cons of cfDNA screening, and the appropriateness of this test as a primary screen, including in low-risk patients, is the focus of this article.
What is cfDNA?
cfDNA consists of small (<200 base pairs) fragments of DNA that are present in the maternal serum. After 10 weeks of gestation, about 10% of the total circulating cfDNA in the maternal serum is derived from the placenta and can therefore be used to test for fetal disorders (FIGURE).3
Although cfDNA screening has been reported to be possible for many different types of genetic conditions, such as RhD type and single-gene disorders such as achondroplasia,4 most clinical testing is done for fetal chromosomal disorders, including trisomies 13, 18, and 21 and the sex chromosomes. In addition, some laboratories provide testing for other trisomies (16 and 22), as well as some of the microdeletion syndromes (22q11.2, 1p36, Prader Willi syndrome, and others).5
Analysis of cfDNA to assess the risk for aneuploidy is done using a number of different approaches; these generally all include next-generation sequencing with advanced bioinformatics analyses.3,6–9 Although the laboratories use somewhat different techniques, all of them share very high sensitivity and specificity for detection of trisomy 21 (TABLE 1).10
Sensitivities for trisomy 13 and sex chromosomal abnormalities are somewhat lower, but the specificity is greater than 99% for each condition, meaning that false-positive rates are very low.
The accuracy of cfDNA in identifying chromosomal aneuploidy depends on several factors, including the relative amount of fetal to maternal DNA, the chance that a chromosome abnormality is present (that is, the risk based on maternal age or results of other screening), and other factors such as the presence of twins or a nonviable second fetus, or the presence of placental mosaicism.
Because of these variables, both false-positive and false-negative results can occur, and the test is not diagnostic but rather is considered a screening test. A positive result does not mean that the fetus is definitely affected with aneuploidy.
What are the advantages of cfDNA screening for low-risk patients?
There are several benefits of cfDNA screening versus traditional screening or diagnostic testing, which are the other options available (TABLE 2). For Down syndrome, the detection rate is higher and the false-positive rate is lower than that seen with traditional aneuploidy screening using serum analytes and nuchal translucency ultrasonography.1,2
TABLE 2 Pros and cons of cfDNA screening in low-risk patients
Pros
- High detection rate and very low false-positive rate
- Can be performed any time after 10 weeks’ gestation
- Requires a single blood test at any gestational age
- Results presented in simple “Yes” or “No” format
- As with other screening tests, cfDNA provides a noninvasive determination of risk
Cons
- Tests for a limited range of conditions, which are rare in low-risk patients
- Is not as comprehensive or definitive as diagnostic testing with amniocentesis or chorionic villus sampling
- Results do not adjust for patient’s prior risk
- Positive results require calculation and interpretation of positive predictive value by provider
- Low fetal DNA and other factors can lead to test failure in some cases
- Cannot be used with vanishing twin
- Can reveal unsuspected maternal conditions of uncertain significance
The test can be done any time after 10 weeks’ gestation without the narrow gestational-age windows required or the need for accurate gestational age determination using traditional screening to accurately interpret results. cfDNA screening involves a single blood test that does not require integration with multiple serum markers or ultrasound findings. Finally, results are generally presented in a simple “Yes” or “No” format that is easy for providers and patients to understand.
CASE Continued
Your patient’s results are positive for trisomy 13. Her understanding is that the test is more than 99% accurate, and she interprets this to mean that the chance of trisomy 13 in her fetus is more than 99%. She is distraught and asks about pregnancy termination.
What are the limitations of cfDNA screening?
Similar to other noninvasive screening tests, cfDNA screening does not carry direct risk to the pregnancy. However, there are limitations to this testing. As a result, the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) recently have stated that traditional screening is the most appropriate option for most women.11,12
One reason that cfDNA screening may not be the best choice for low-risk women is that Down syndrome is quite uncommon in this group, so cfDNA screening is a very precise test for a rare condition. Traditional multiple marker screening, on the other hand, is more effective at signaling risk for the broad range of adverse perinatal outcomes that can affect a pregnancy, including other structural birth defects, as well as such obstetric complications as preterm birth, preeclampsia, and fetal growth restriction.13,14
Many women who undergo cfDNA screening are under the impression that they have had a definitive test for all birth defects when, in fact, the coverage of cfDNA for all possible birth defects in a low-risk woman is very limited; her residual risk for a birth defect is little changed by a normal cfDNA result.
The ease of obtaining a blood sample for cfDNA screening is an advantage of the test. However, because it is simple to perform, it often is done with inadequate pretest counseling or consideration. Just because the test is easily obtained does not negate the need for adequate discussion to assure that each woman understands what the test can and cannot measure, and the possible outcomes of testing.
Another perceived benefit of cfDNA screening is the simple presentation of results. While reports vary, they generally provide very dichotomized results. Aneuploidy risk is reported as “Positive” or “Negative,” or as “Detected” or “Not detected.”
Some laboratories report the chance of aneuploidy; this is almost always stated to be more than 99% in patients at increased risk, and less than 1 in 10,000 in patients at low risk.
All of these results suggest a near diagnostic certainty. However, this reporting is oversimplified and misleading, as it does not account for each patient’s prior or background risk. The chance that a positive result is a true positive is very different in a 20-year-old versus a 35-year-old woman, yet the reports do not reflect this difference in positive predictive value (PPV). See TABLE 1.
Accurate interpretation of risk for the individual patient, therefore, requires calculation by the provider; this can be done through an online calculator available through the Perinatal Quality Foundation (www.perina talquality.org).
CASE Continued
You explain to your patient that the chance her fetus has trisomy 13 is far lower than 99%, based in part on the very low prior risk given her age. You calculate the PPV using an online calculator, which estimates that there is only a 7% chance that this is a true positive result.
As mentioned earlier, there has been a tremendously rapid uptake of cfDNA screening. Given wider use by practitioners not as familiar with the complexities of genetic testing and statistical analysis, misunderstanding of the test characteristics carries risks if inappropriate recommendations or decisions are made or actions taken.
Most low-risk patients do not request or desire diagnostic testing. It is important during pretest counseling to explain that cfDNA cannot detect all significant chromosomal aneuploidies. Some serious abnormalities will be undetected; therefore, some women may prefer more comprehensive prenatal testing (TABLE 3).
TABLE 3 Checklist for pretest counseling for cfDNA28
- cfDNA screening is the most accurate screening test for trisomy 21
- cfDNA is a screening test, and false-positive and false-negative results can and do occur
- Diagnostic confirmation with chorionic villus sampling or amniocentesis is recommended for women with abnormal cfDNA results
- A negative cfDNA result decreases risk but does not rule out trisomy 21 and other chromosomal conditions
- cfDNA does not test for all chromosomal conditions
- Women who desire definitive information about chromosome conditions in the pregnancy should consider diagnostic testing with chorionic villus sampling or amniocentesis
- All genetic testing is optional. Whether a woman chooses to have a screening test, a diagnostic test, or no testing is a personal decision; any are reasonable options for any pregnant woman.
ACOG recommends that diagnostic testing should be available to all pregnant women, regardless of age.15 In prenatal series, trisomies 13, 18, and 21 make up approximately two-thirds of all clinically significant aneuploidies.16,17 Given that cfDNA detects only these aneuploidies, the other third will not be identified prenatally in patients who choose cfDNA. Traditional aneuploidy screening has been demonstrated to detect a broader range of these less common but clinically important chromosomal abnormalities.18
In one study of women found to be at increased risk based on traditional multiple marker screening, if cfDNA were chosen instead of diagnostic testing, 17% of the aneuploidies present in this group would not have been detected.18 Of all high-risk women in this study, 2%, or 1 in 50, had a chromosomal abnormality detectable by amniocentesis but not with cfDNA.
Successful tests require adequate placental DNA
Accurate interpretation of cfDNA screening also requires that an adequate quantity of placental DNA be present; this is often referred to as the “fetal fraction.” In some cases, the placental DNA volume is too low for accurate analysis, particularly in obese patients and women with specific chromosomal abnormalities.
Some laboratories measure this and do not report a result if the fetal fraction is below a specific cut-off, typically about 4%. Other laboratories do not measure or exclude cases with too little fetal DNA, raising concern that this could result in missing cases of aneuploidy. It has been noted that a placental DNA fraction of less than 8% is associated with less accurate test results, even if results are returned.8
Low fetal fraction also has been associated with maternal obesity, and in one study cfDNA failed to provide a result in 20% of women weighing more than 250 lb and 50% of women weighing more than 350 lb.19 Therefore, cfDNA is not the best option for obese women (TABLE 4).
TABLE 4 Appropriateness of cfDNA screeningin specific clinical circumstances
Optimal candidates for cfDNA screening
- High risk for trisomy based on maternal age (≥35 years)
- Ultrasound findings suggesting trisomy 13, 18, or 21
- History of prior pregnancy with trisomy 13, 18, or 21
- Positive traditional screening test
- Parental balanced Robertsonian translocation associated with risk for trisomy 13 or 21
Less optimal candidates
- Low risk for trisomy based on age and/or low risk traditional screening
- Ultrasound structural anomalies other than those specifically suggesting trisomy 13, 18, or 21
- High risk for nonchromosomal genetic disorder
- Comprehensive genetic diagnosis desired
- Maternal malignancy
- Maternal organ transplant
- Maternal sex chromosomal mosaicism or other chromosomal abnormality
- Maternal obesity
- Gestational age <10 weeks
While the free fraction is relatively constant from 10 to 22 weeks’ gestation, it is lower earlier than 10 weeks’ gestation and less likely to provide a result. For this reason, the test should not be attempted before 10 weeks’ gestation.
Recent evidence indicates that low fetal DNA fraction is associated with some chromosome abnormalities. Given this association, women with failed cfDNA results should be counseled and offered appropriate follow-up. As the association appears to be greater for trisomies 13 and 18, and triploidy, a careful ultrasound is likely to detect abnormalities in many such cases. However, it also is appropriate to offer the option of diagnostic testing, given the very high risk.
A repeat cfDNA test will be successful in some cases. Whether the patient chooses to attempt cfDNA again may depend in part on maternal body mass index (BMI), as well as gestational age—a patient at a more advanced gestation may not wish to delay obtaining definitive information given the high risk.
cfDNA screening has a low false-positive rate
One of the greatest benefits of cfDNA screening is a lower false-positive rate than is reported with traditional screening. However, when “no results” cases are also considered, the percentage of patients who require follow-up after cfDNA is close to that of traditional screening.
The chance of test failure is reported to be 0.9% to 8.1%,7,9,10 and varies in part by whether the laboratory measures fetal fraction and requires a minimum concentration.
A recent meta-analysis estimated the overall test failure rate at 3%.10 When comparing cfDNA to traditional screening, if “no results” cases are included with the “screen positive” group, the benefits of cfDNA over traditional screening are much less clear, particularly in a low-risk population.
ACOG: Offer traditional multiple-marker screening first
While multiple marker and cfDNA screening have differing performance characteristics, there are no data to support doing both tests concurrently. In fact, in a recent survey of nearly 200 women presented with different testing scenarios, women found it preferable and more reassuring to have a positive traditional screen followed by normal cfDNA results, rather that discrepant results of the 2 tests done concurrently.20
For many reasons, the approach recommended by ACOG and SMFM is to offer traditional multiple-marker screening first, and cfDNA screening or diagnostic testing as a follow-up for patients that screen positive. In that scenario, the benefits and limitations of diagnostic testing versus follow-up with cfDNA screening should be explained carefully.
In all patients who have a positive cfDNA result, diagnostic testing for confirmation should be offered and strongly recommended prior to pregnancy termination if that is considered. Even if a structural abnormality is present and a true positive result is highly likely, karyotyping is important to determine if there may be an inherited translocation putting subsequent pregnancies at higher risk.
Components of pretest counseling
A woman of any age can have a fetus with trisomy or another chromosomal abnormality, and some women prefer diagnostic testing or no testing regardless of age. It is therefore appropriate to offer diagnostic testing, screening, or the option of no testing to all women.
Recent studies have demonstrated that providing well-informed access to all prenatal tests results in more informed choices and no increase in uptake of invasive testing.22 However, the offer of prenatal testing requires discussion of the pros and cons of all test options, including the detection rates of all significant abnormalities, the screen positive rates, and recommended follow-up if an abnormal result is obtained. See TABLE 4.
Cost-effectiveness
Although the detection rate of cfDNA for trisomy 21 is higher than that of traditional screening, the detection rate of traditional screening is also quite high at lower cost. For low-risk women, therefore, traditional screening provides a less expensive alternative to cfDNA. Because aneuploidy is rare in low-risk patients, the residual chance of aneuploidy after a normal traditional screen is very low, and the cost per additional case of Down syndrome detected by cfDNA is very high.
In one study, this was estimated at $3.6 million.23 These authors suggested that, at present, cfDNA is optimally used as a secondary screen for high-risk women. Other cost analyses also have demonstrated that the most cost-effective strategy is a model in which cfDNA is used as a follow-up test in patients found to be screen positive by traditional screening.15,24 A recent cost utility analysis compared outcomes of 6 approaches to prenatal screening, including sequential screening, cfDNA screening, nuchal translucency only, and diagnostic testing with microarray (alone, in combination, or in sequence).
The clinical outcomes included fetal abnormalities detected, taking into account all chromosomal abnormalities, as well as failed cfDNA tests. For younger women (<40 yr), traditional sequential screening provided the highest detection of all abnormalities and was the optimal testing strategy, while cfDNA was preferable for women aged 40 or older, given the higher prevalence of trisomy 21.20
Incidental findings
Given that the cfDNA in maternal serum is a mixture of maternal and placental DNA, a number of biologic phenomena can cause a false-positive cfDNA result. In many cases, these false-positives reveal unanticipated or unexpected maternal conditions and information that the woman may have preferred not to know. A few cases of maternal malignancies with chromosomal abnormalities within the tumor have been reported in patients with false-positive cfDNA results.26
These case reports have raised the question about the need for further evaluation for maternal malignancy in women with false-positive results. Maternal genetic disorders also can cause false-positive results, and may lead to unanticipated detection of adult-onset conditions. In some cases, positive results for sex chromosomal aneuploidy can occur in pregnant women who themselves have a sex chromosomal abnormality, often in mosaic form and previously undiagnosed.27
Again, this has led to discussion of the possible health benefit of karyotyping women who have a false-positive cfDNA result to rule out a mosaic chromosomal abnormality in the mother.
At this time, the clinical utility of such investigations is unknown and there are no recommendations regarding appropriate follow-up for such cases.
CASE Resolved
Given the results of her cfDNA screening, your patient opts to undergo diagnostic testing. In that testing, trisomy 13 is ruled out and she goes on to have a healthy daughter.
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Bianchi DW, Parker RL, Wentworth J, et al. DNA sequencing versus standard prenatal aneuploidy screening. N Engl J Med. 2014;370(9):799–808.
- Norton ME, Jacobsson B, Swamy GK, et al. Cell-free DNA analysis for noninvasive examination of trisomy. N Engl J Med. 2015;372(17):1589–1597.
- Norton ME, Brar H, Weiss J, et al. Non-Invasive Chromosomal Evaluation (NICE) study: results of a multicenter prospective cohort study for detection of fetal trisomy 21 and trisomy 18. Am J Obstet Gynecol. 2012;207(2):137.e1–e8.
- Chitty LS, Mason S, Barrett AN, et al. Non-invasive prenatal diagnosis of achondroplasia and thanatophoric dysplasia: next-generation sequencing allows for a safer, more accurate, and comprehensive approach. Prenat Diagn. 2015;35(7):656–662.
- Wapner RJ, Babiarz JE, Levy B, et al. Expanding the scope of noninvasive prenatal testing: detection of fetal microdeletion syndromes. Am J Obstet Gynecol. 2015;212(3):332.e1–e9.
- Bianchi DW, Platt LD, Goldberg JD, Abuhamad AZ, Sehnert AJ, Rava RP. Genome-wide fetal aneuploidy detection by maternal plasma DNA sequencing. Obstet Gynecol. 2012;119(5):890–901.
- Palomaki GE, Kloza EM, Lambert-Messerlian GM, et al. DNA sequencing of maternal plasma to detect Down syndrome: an international clinical validation study. Genet Med. 2011;13(11):913–920.
- Sparks AB, Wang ET, Struble CA, et al. DNA sequencing of maternal plasma to detect Down syndrome: an international clinical validation study [abstract]. Proceedings of the ISPD 16th International Conference on Prenatal Diagnosis and Therapy; Miami, Florida; June 3–6, 2012. Prenat Diagn. 2012;32(suppl 1):s3–s9.
- Zimmermann B, Hill M, Gemelos G, et al. Noninvasive prenatal aneuploidy testing of chromosomes 13, 18, 21, X, and Y, using targeted sequencing of polymorphic loci. Prenat Diagn. 2012;32(13):1233–1241.
- Gil MM, Quezada MS, Revello R, Akolekar R, Niclaides KH. Analysis of cell-free DNA in maternal blood in screening for fetal aneuploidies: updated meta-analysis. Ultrasound Obstet Gynecol. 2015;45(3):249–266.
- American College of Obstetricians and Gynecologists. Committee Opinion No. 640: cell-free DNA screening for fetal aneuploidy. Obstet Gynecol. 2015;126(3):e31–e37.
- Society for Maternal-Fetal Medicine (SMFM) Publications Committee. Prenatal aneuploidy screening using cell-free DNA. Am J Obstet Gynecol. 2015;212(6):711–716.
- Baer RJ, Currier RJ, Norton ME, et al. Obstetric, perinatal, and fetal outcomes in pregnancies with false-positive integrated screening results. Obstet Gynecol. 2014;123(3):603–609.
- Dugoff L; Society for Maternal-Fetal Medicine. First- and second-trimester maternal serum markers for aneuploidy and adverse obstetric outcomes. Obstet Gynecol. 2010;115(5):1052–1061.
- American College of Obstetricians and Gynecologists. Practice bulletin No. 88: invasive prenatal testing for aneuploidy. Obstet Gynecol. 2007;110(6):1459–1467.
- Alamillo CM, Krantz D, Evans M, Fiddler M, Pergament E. Nearly a third of abnormalities found after first-trimester screening are different than expected: 10-year experience from a single center. Prenat Diagn. 2013;33(3):251–256.
- Wellesley D, Dolk H, Boyd PA, et al. Rare chromosome abnormalities, prevalence and prenatal diagnosis rates from population-based congenital anomaly registers in Europe. Eur J Hum Genet. 2012;20(5):521–526.
- Norton ME, Jelliffe-Pawlowski LL, Currier RJ. Chromosome abnormalities detected by current prenatal screening and noninvasive prenatal testing. Obstet Gynecol. 2014;124(5):979–986.
- Ashoor G, Syngelaki A, Poon LC, Rezende JC, Nicolaides KH. Fetal fraction in maternal plasma cell-free DNA at 11-13 weeks’ gestation: relation to maternal and fetal characteristics. Ultrasound Obstet Gynecol. 2013;41(1):26–32.
- Kaimal AJ, Norton ME, Kuppermann M. Prenatal testing in the genomic age: clinical outcomes, quality of life, and costs. Obstet Gynecol. 2015;126(4):737–746.
- Wapner RJ, Martin CL, Levy B, et al. Chromosomal microarray versus karyotyping for prenatal diagnosis. N Engl J Med. 2012;367(23):2175–2184.
- Kuppermann M, Pena S, Bishop JT, et al. Effect of enhanced information, values clarification, and removal of financial barriers on use of prenatal genetic testing: a randomized clinical trial. JAMA. 2014;312(12):1210–1217.
- Cuckle H, Benn P, Pergament E. Maternal cfDNA screening for Down syndrome—a cost sensitivity analysis. Prenat Diagn. 2013;33(7):636–642.
- Beulen L, Grutters JPC, Faas BH, Feenstra I, van Vugt JMG, Bekker MN. The consequences of implementing non-invasive prenatal testing in Dutch national health care: a cost-effectiveness analysis. Eur J Obstet Gynecol Reprod Biol. 2014;182:53–61.
- Okun N, Teitelbaum M, Huang T, Dewa CS, Hoch JS. The price of performance: a cost and performance analysis of the implementation of cell-free fetal DNA testing for Down syndrome in Ontario, Canada: Cost and performance analysis of cfDNA testing for Down syndrome in Ontario. Prenat Diagn. 2014;34(4):350–356.
- Bianchi DW, Chudova D, Sehnert AJ, et al. Noninvasive prenatal testing and incidental detection of occult maternal malignancies. JAMA. 2015;314(2):162–169.
- Wang Y, Chen Y, Tian F, et al. Maternal mosaicism is a significant contributor to discordant sex chromosomal aneuploidies associated with noninvasive prenatal testing. Clin Chem. 2014;60(1):251–259.
- Norton ME, Jelliffe-Pawlowski LL, Currier RJ. Chromosome abnormalities detected by current prenatal screening and noninvasive prenatal testing. Obstet Gynecol. 2014;124(5):979–986Society for Maternal-Fetal Medicine (SMFM) Publications Committee. Prenatal aneuploidy screening using cell-free DNA. Am J Obstet Gynecol. 2015;212(6):711–716.
- Bianchi DW, Parker RL, Wentworth J, et al. DNA sequencing versus standard prenatal aneuploidy screening. N Engl J Med. 2014;370(9):799–808.
- Norton ME, Jacobsson B, Swamy GK, et al. Cell-free DNA analysis for noninvasive examination of trisomy. N Engl J Med. 2015;372(17):1589–1597.
- Norton ME, Brar H, Weiss J, et al. Non-Invasive Chromosomal Evaluation (NICE) study: results of a multicenter prospective cohort study for detection of fetal trisomy 21 and trisomy 18. Am J Obstet Gynecol. 2012;207(2):137.e1–e8.
- Chitty LS, Mason S, Barrett AN, et al. Non-invasive prenatal diagnosis of achondroplasia and thanatophoric dysplasia: next-generation sequencing allows for a safer, more accurate, and comprehensive approach. Prenat Diagn. 2015;35(7):656–662.
- Wapner RJ, Babiarz JE, Levy B, et al. Expanding the scope of noninvasive prenatal testing: detection of fetal microdeletion syndromes. Am J Obstet Gynecol. 2015;212(3):332.e1–e9.
- Bianchi DW, Platt LD, Goldberg JD, Abuhamad AZ, Sehnert AJ, Rava RP. Genome-wide fetal aneuploidy detection by maternal plasma DNA sequencing. Obstet Gynecol. 2012;119(5):890–901.
- Palomaki GE, Kloza EM, Lambert-Messerlian GM, et al. DNA sequencing of maternal plasma to detect Down syndrome: an international clinical validation study. Genet Med. 2011;13(11):913–920.
- Sparks AB, Wang ET, Struble CA, et al. DNA sequencing of maternal plasma to detect Down syndrome: an international clinical validation study [abstract]. Proceedings of the ISPD 16th International Conference on Prenatal Diagnosis and Therapy; Miami, Florida; June 3–6, 2012. Prenat Diagn. 2012;32(suppl 1):s3–s9.
- Zimmermann B, Hill M, Gemelos G, et al. Noninvasive prenatal aneuploidy testing of chromosomes 13, 18, 21, X, and Y, using targeted sequencing of polymorphic loci. Prenat Diagn. 2012;32(13):1233–1241.
- Gil MM, Quezada MS, Revello R, Akolekar R, Niclaides KH. Analysis of cell-free DNA in maternal blood in screening for fetal aneuploidies: updated meta-analysis. Ultrasound Obstet Gynecol. 2015;45(3):249–266.
- American College of Obstetricians and Gynecologists. Committee Opinion No. 640: cell-free DNA screening for fetal aneuploidy. Obstet Gynecol. 2015;126(3):e31–e37.
- Society for Maternal-Fetal Medicine (SMFM) Publications Committee. Prenatal aneuploidy screening using cell-free DNA. Am J Obstet Gynecol. 2015;212(6):711–716.
- Baer RJ, Currier RJ, Norton ME, et al. Obstetric, perinatal, and fetal outcomes in pregnancies with false-positive integrated screening results. Obstet Gynecol. 2014;123(3):603–609.
- Dugoff L; Society for Maternal-Fetal Medicine. First- and second-trimester maternal serum markers for aneuploidy and adverse obstetric outcomes. Obstet Gynecol. 2010;115(5):1052–1061.
- American College of Obstetricians and Gynecologists. Practice bulletin No. 88: invasive prenatal testing for aneuploidy. Obstet Gynecol. 2007;110(6):1459–1467.
- Alamillo CM, Krantz D, Evans M, Fiddler M, Pergament E. Nearly a third of abnormalities found after first-trimester screening are different than expected: 10-year experience from a single center. Prenat Diagn. 2013;33(3):251–256.
- Wellesley D, Dolk H, Boyd PA, et al. Rare chromosome abnormalities, prevalence and prenatal diagnosis rates from population-based congenital anomaly registers in Europe. Eur J Hum Genet. 2012;20(5):521–526.
- Norton ME, Jelliffe-Pawlowski LL, Currier RJ. Chromosome abnormalities detected by current prenatal screening and noninvasive prenatal testing. Obstet Gynecol. 2014;124(5):979–986.
- Ashoor G, Syngelaki A, Poon LC, Rezende JC, Nicolaides KH. Fetal fraction in maternal plasma cell-free DNA at 11-13 weeks’ gestation: relation to maternal and fetal characteristics. Ultrasound Obstet Gynecol. 2013;41(1):26–32.
- Kaimal AJ, Norton ME, Kuppermann M. Prenatal testing in the genomic age: clinical outcomes, quality of life, and costs. Obstet Gynecol. 2015;126(4):737–746.
- Wapner RJ, Martin CL, Levy B, et al. Chromosomal microarray versus karyotyping for prenatal diagnosis. N Engl J Med. 2012;367(23):2175–2184.
- Kuppermann M, Pena S, Bishop JT, et al. Effect of enhanced information, values clarification, and removal of financial barriers on use of prenatal genetic testing: a randomized clinical trial. JAMA. 2014;312(12):1210–1217.
- Cuckle H, Benn P, Pergament E. Maternal cfDNA screening for Down syndrome—a cost sensitivity analysis. Prenat Diagn. 2013;33(7):636–642.
- Beulen L, Grutters JPC, Faas BH, Feenstra I, van Vugt JMG, Bekker MN. The consequences of implementing non-invasive prenatal testing in Dutch national health care: a cost-effectiveness analysis. Eur J Obstet Gynecol Reprod Biol. 2014;182:53–61.
- Okun N, Teitelbaum M, Huang T, Dewa CS, Hoch JS. The price of performance: a cost and performance analysis of the implementation of cell-free fetal DNA testing for Down syndrome in Ontario, Canada: Cost and performance analysis of cfDNA testing for Down syndrome in Ontario. Prenat Diagn. 2014;34(4):350–356.
- Bianchi DW, Chudova D, Sehnert AJ, et al. Noninvasive prenatal testing and incidental detection of occult maternal malignancies. JAMA. 2015;314(2):162–169.
- Wang Y, Chen Y, Tian F, et al. Maternal mosaicism is a significant contributor to discordant sex chromosomal aneuploidies associated with noninvasive prenatal testing. Clin Chem. 2014;60(1):251–259.
- Norton ME, Jelliffe-Pawlowski LL, Currier RJ. Chromosome abnormalities detected by current prenatal screening and noninvasive prenatal testing. Obstet Gynecol. 2014;124(5):979–986Society for Maternal-Fetal Medicine (SMFM) Publications Committee. Prenatal aneuploidy screening using cell-free DNA. Am J Obstet Gynecol. 2015;212(6):711–716.
In this Article
- Pros and cons of cfDNA in low-risk patients
- Optimal and less optimal candidates for cfDNA screening
- Checklist for pretest counseling for cfDNA
Should you adopt the practice of vaginal cleansing with povidone-iodine prior to cesarean delivery?
There are approximately 4,000,000 births annually in the United States, and about 32% of them occur by cesarean delivery. Compared with vaginal birth, cesarean delivery is associated with an increased risk of endometritis (defined as fever plus uterine or abdominal tenderness). Although surgical complications cannot be eliminated entirely, surgeons are deeply dedicated to the continuous improvement of surgical practice in order to reduce the risk of complications.
With cesarean delivery, many surgical practices have been adopted universally to reduce postoperative complications, including administration of intravenous (IV) antibiotics before skin incision to minimize postoperative infection and the use of postoperative mechanical or pharmacologic interventions to help prevent venous thromboembolism and pulmonary embolism. Preoperative vaginal cleansing with povidone-iodine may reduce the risk of postoperative endometritis, but the practice is not currently common in the United States.
Should you adopt a policy of preoperative vaginal cleansing prior to cesarean delivery? The data suggest perhaps you should.
Data-driven support for povidone-iodine precesareanThree large randomized trials published within the past 10 years concluded that preoperative vaginal cleansing with povidone-iodine reduced the risk of postcesarean endometritis in women who also received prophylactic IV antibiotics (TABLE).1−3 Vaginal cleansing did not reduce the rate of postpartum fever or wound infection in these studies.
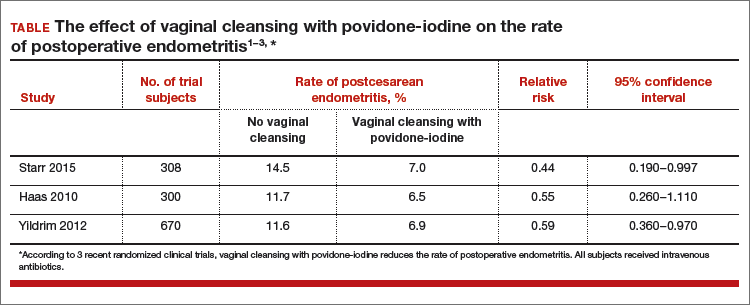
Clinical factors that increased the risk of postpartum endometritis independent of vaginal cleansing included:
- extended duration of cesarean surgery
- being in labor prior to cesarean delivery
- ruptured membranes
- advanced cervical examination
- maternal anemia
- use of intrapartum internal monitors
- prior history of genitourinary infection.
Authors of two recent, large nonrandomized studies also have reported that vaginal cleansing reduced the risk of postcesarean endometritis.4,5 By contrast, investigators from one large trial from 2001 did not observe a decrease in endometritis with vaginal cleansing.6
To test the impact of metronidazole vaginal gel on post‑cesarean endometritis, 224 women undergoing cesarean delivery for various indications were randomly assigned to placebo vaginal gel or metronidazole vaginal gel 5 g prior to surgery initiation.1 Most women also received intravenous antibiotics. The rates of endometritis were 17% and 7% in the placebo and metronidazole groups, respectively (relative risk, 0.42; 95% confidence interval, 0.19−0.92).
Vaginal antibiotic administration shows promise as an alternative to povidone-iodine cleansing in the prevention of postcesarean endometritis. Additional randomized clinical trials are necessary to fully evaluate the benefits and risks of this practice.
Reference
1. Pitt C, Sanchez-Ramos L, Kaunitz AM. Adjunctive intravaginal metronidazole for the prevention of postcesarean endometritis: a randomized controlled trial. Obstet Gynecol. 2001;98(5 pt 1):745−750.
Cochrane review of precesarean vaginal cleansingAuthors of a Cochrane review, in which they synthesized 7 studies involving 2,635 women, reported that vaginal cleansing with povidone-iodine immediately before cesarean delivery was associated with a reduced risk of postcesarean endometritis: 8.3% vs 4.3% in the control and vaginal cleansing groups, respectively, (risk ratio [RR], 0.45; 95% confidence interval [CI], 0.25−0.81).7
The positive effect of vaginal cleansing was particularly noteworthy in the subgroup of women with ruptured membranes (3 trials involving 272 women). The rates of endometritis in the control versus vaginal cleansing groups were 17.9% and 4.3%, respectively (RR, 0.24; 95% CI, 0.10−0.55).
Women who went into labor prior to cesarean delivery (523 women from 3 trials) also benefitted from vaginal cleansing, with endometritis rates of 13.0% and 7.4% in the control and vaginal cleansing groups, respectively (RR, 0.56; 95% CI, 0.34−0.95).
In this review, again, vaginal cleansing did not significantly reduce the rate of postoperative fever or wound infection.
The American College of Obstetricians and Gynecologists has noted that chlorohexidine gluconate solutions with high concentrations of alcohol are contraindicated for vaginal cleansing.1 However, although not approved for vaginal cleansing, solutions of chlorohexidine gluconate with low alcohol content (4% alcohol concentration) are safe and may be effective for off-label use as vaginal cleansings.
Reference
1. American College of Obstetricians and Gynecologists Women’s Health Care Physicians; Committee on Gynecologic Practice. Committee Opinion No. 571: solutions for surgical cleansing of the vagina. Obstet Gynecol. 2013;122(3):718−720.
Is vaginal cleansing prior to cesarean delivery best practice?In the United States, precesarean vaginal cleansing is not a common practice. To close the gap between current practice and what is potentially a best practice, two approaches to using vaginal cleansing could be instituted in delivery units.
Approach #1: A liberal clinical protocol. In this scenario, all women (who are not allergic to iodine or povidone-iodine) undergoing cesarean delivery should undergo vaginal cleansing. The World Health Organization conditionally recommends vaginal cleansing for all women undergoing a cesarean delivery.8
Approach #2: A focused clinical protocol. For this protocol, only women (again, who are not allergic to iodine or povidone-iodine) who have ruptured membranes or are in labor upon advanced cervical examination should receive vaginal cleansing.
The advantage of a liberal protocol is that vaginal preparation becomes embedded within the standard practice of cesarean delivery and, hence, is seldom overlooked. The upside of the focused protocol is that only those women most likely to benefit receive the intervention.
Tell me what you thinkWill you consider using vaginal cleansing in your practice? Please let me know your views on vaginal cleansing for cesarean delivery, as well as your clinical pearls on cesarean delivery surgery, at obgmanagement.com. In addition, weigh in on the Quick Poll posted to OBG Management’s homepage. Send your letter to the editor to [email protected].
- Starr RV, Zurawski J, Ismail M. Preoperative vaginal preparation with povidone-iodine and the risk of postcesarean endometritis. Obstet Gynecol. 2015;105(5 pt 1):1024–1029.
- Haas DM, Pazouki F, Smith RR, et al. Vaginal cleansing before cesarean delivery to reduce postoperative infectious morbidity: a randomized, controlled trial. Am J Obstet Gynecol. 2010;202(3):310.e1–e6.
- Yildrim G, Gungorduk K, Asicioglu O, et al. Does vaginal preparation with povidone-iodine prior to cesarean delivery reduce the risk of endometritis?A randomized controlled trial. J Matern Fetal Neonatal Med. 2012;25(11):2316–2321.
- Asghania M, Mirblouk F, Shakiba M, Faraji R. Preoperative vaginal preparation with povidone-iodine on post-cesarean infectious morbidity. J Obstet Gynaecol. 2011;31(5):400–403.
- Memon S, Qazi RA, Bibi S, Parveen N. Effect of preoperative vaginal cleansing with an antiseptic solution to reduce post caesarean infectious morbidity. J Pak Med Assoc. 2011;61(12):1179–1183.
- Reid VC, Hartmann KE, McMahon M, Fry EP. Vaginal preparation with povidone-iodine and postcesarean infectious morbidity: a randomized controlled trial. Obstet Gynecol. 2001;97(1):147–152.
- Haas DM, Morgan S, Contreras K. Vaginal preparation with antiseptic solution before cesarean section for preventing postoperative infections. Cochrane Database Syst Rev. 2014;12:CD007892.
- Yildrim G, Gungorduk K, Asicioglu O, et al. Does vaginal preparation with povidone-iodine prior to
There are approximately 4,000,000 births annually in the United States, and about 32% of them occur by cesarean delivery. Compared with vaginal birth, cesarean delivery is associated with an increased risk of endometritis (defined as fever plus uterine or abdominal tenderness). Although surgical complications cannot be eliminated entirely, surgeons are deeply dedicated to the continuous improvement of surgical practice in order to reduce the risk of complications.
With cesarean delivery, many surgical practices have been adopted universally to reduce postoperative complications, including administration of intravenous (IV) antibiotics before skin incision to minimize postoperative infection and the use of postoperative mechanical or pharmacologic interventions to help prevent venous thromboembolism and pulmonary embolism. Preoperative vaginal cleansing with povidone-iodine may reduce the risk of postoperative endometritis, but the practice is not currently common in the United States.
Should you adopt a policy of preoperative vaginal cleansing prior to cesarean delivery? The data suggest perhaps you should.
Data-driven support for povidone-iodine precesareanThree large randomized trials published within the past 10 years concluded that preoperative vaginal cleansing with povidone-iodine reduced the risk of postcesarean endometritis in women who also received prophylactic IV antibiotics (TABLE).1−3 Vaginal cleansing did not reduce the rate of postpartum fever or wound infection in these studies.

Clinical factors that increased the risk of postpartum endometritis independent of vaginal cleansing included:
- extended duration of cesarean surgery
- being in labor prior to cesarean delivery
- ruptured membranes
- advanced cervical examination
- maternal anemia
- use of intrapartum internal monitors
- prior history of genitourinary infection.
Authors of two recent, large nonrandomized studies also have reported that vaginal cleansing reduced the risk of postcesarean endometritis.4,5 By contrast, investigators from one large trial from 2001 did not observe a decrease in endometritis with vaginal cleansing.6
To test the impact of metronidazole vaginal gel on post‑cesarean endometritis, 224 women undergoing cesarean delivery for various indications were randomly assigned to placebo vaginal gel or metronidazole vaginal gel 5 g prior to surgery initiation.1 Most women also received intravenous antibiotics. The rates of endometritis were 17% and 7% in the placebo and metronidazole groups, respectively (relative risk, 0.42; 95% confidence interval, 0.19−0.92).
Vaginal antibiotic administration shows promise as an alternative to povidone-iodine cleansing in the prevention of postcesarean endometritis. Additional randomized clinical trials are necessary to fully evaluate the benefits and risks of this practice.
Reference
1. Pitt C, Sanchez-Ramos L, Kaunitz AM. Adjunctive intravaginal metronidazole for the prevention of postcesarean endometritis: a randomized controlled trial. Obstet Gynecol. 2001;98(5 pt 1):745−750.
Cochrane review of precesarean vaginal cleansingAuthors of a Cochrane review, in which they synthesized 7 studies involving 2,635 women, reported that vaginal cleansing with povidone-iodine immediately before cesarean delivery was associated with a reduced risk of postcesarean endometritis: 8.3% vs 4.3% in the control and vaginal cleansing groups, respectively, (risk ratio [RR], 0.45; 95% confidence interval [CI], 0.25−0.81).7
The positive effect of vaginal cleansing was particularly noteworthy in the subgroup of women with ruptured membranes (3 trials involving 272 women). The rates of endometritis in the control versus vaginal cleansing groups were 17.9% and 4.3%, respectively (RR, 0.24; 95% CI, 0.10−0.55).
Women who went into labor prior to cesarean delivery (523 women from 3 trials) also benefitted from vaginal cleansing, with endometritis rates of 13.0% and 7.4% in the control and vaginal cleansing groups, respectively (RR, 0.56; 95% CI, 0.34−0.95).
In this review, again, vaginal cleansing did not significantly reduce the rate of postoperative fever or wound infection.
The American College of Obstetricians and Gynecologists has noted that chlorohexidine gluconate solutions with high concentrations of alcohol are contraindicated for vaginal cleansing.1 However, although not approved for vaginal cleansing, solutions of chlorohexidine gluconate with low alcohol content (4% alcohol concentration) are safe and may be effective for off-label use as vaginal cleansings.
Reference
1. American College of Obstetricians and Gynecologists Women’s Health Care Physicians; Committee on Gynecologic Practice. Committee Opinion No. 571: solutions for surgical cleansing of the vagina. Obstet Gynecol. 2013;122(3):718−720.
Is vaginal cleansing prior to cesarean delivery best practice?In the United States, precesarean vaginal cleansing is not a common practice. To close the gap between current practice and what is potentially a best practice, two approaches to using vaginal cleansing could be instituted in delivery units.
Approach #1: A liberal clinical protocol. In this scenario, all women (who are not allergic to iodine or povidone-iodine) undergoing cesarean delivery should undergo vaginal cleansing. The World Health Organization conditionally recommends vaginal cleansing for all women undergoing a cesarean delivery.8
Approach #2: A focused clinical protocol. For this protocol, only women (again, who are not allergic to iodine or povidone-iodine) who have ruptured membranes or are in labor upon advanced cervical examination should receive vaginal cleansing.
The advantage of a liberal protocol is that vaginal preparation becomes embedded within the standard practice of cesarean delivery and, hence, is seldom overlooked. The upside of the focused protocol is that only those women most likely to benefit receive the intervention.
Tell me what you thinkWill you consider using vaginal cleansing in your practice? Please let me know your views on vaginal cleansing for cesarean delivery, as well as your clinical pearls on cesarean delivery surgery, at obgmanagement.com. In addition, weigh in on the Quick Poll posted to OBG Management’s homepage. Send your letter to the editor to [email protected].
There are approximately 4,000,000 births annually in the United States, and about 32% of them occur by cesarean delivery. Compared with vaginal birth, cesarean delivery is associated with an increased risk of endometritis (defined as fever plus uterine or abdominal tenderness). Although surgical complications cannot be eliminated entirely, surgeons are deeply dedicated to the continuous improvement of surgical practice in order to reduce the risk of complications.
With cesarean delivery, many surgical practices have been adopted universally to reduce postoperative complications, including administration of intravenous (IV) antibiotics before skin incision to minimize postoperative infection and the use of postoperative mechanical or pharmacologic interventions to help prevent venous thromboembolism and pulmonary embolism. Preoperative vaginal cleansing with povidone-iodine may reduce the risk of postoperative endometritis, but the practice is not currently common in the United States.
Should you adopt a policy of preoperative vaginal cleansing prior to cesarean delivery? The data suggest perhaps you should.
Data-driven support for povidone-iodine precesareanThree large randomized trials published within the past 10 years concluded that preoperative vaginal cleansing with povidone-iodine reduced the risk of postcesarean endometritis in women who also received prophylactic IV antibiotics (TABLE).1−3 Vaginal cleansing did not reduce the rate of postpartum fever or wound infection in these studies.

Clinical factors that increased the risk of postpartum endometritis independent of vaginal cleansing included:
- extended duration of cesarean surgery
- being in labor prior to cesarean delivery
- ruptured membranes
- advanced cervical examination
- maternal anemia
- use of intrapartum internal monitors
- prior history of genitourinary infection.
Authors of two recent, large nonrandomized studies also have reported that vaginal cleansing reduced the risk of postcesarean endometritis.4,5 By contrast, investigators from one large trial from 2001 did not observe a decrease in endometritis with vaginal cleansing.6
To test the impact of metronidazole vaginal gel on post‑cesarean endometritis, 224 women undergoing cesarean delivery for various indications were randomly assigned to placebo vaginal gel or metronidazole vaginal gel 5 g prior to surgery initiation.1 Most women also received intravenous antibiotics. The rates of endometritis were 17% and 7% in the placebo and metronidazole groups, respectively (relative risk, 0.42; 95% confidence interval, 0.19−0.92).
Vaginal antibiotic administration shows promise as an alternative to povidone-iodine cleansing in the prevention of postcesarean endometritis. Additional randomized clinical trials are necessary to fully evaluate the benefits and risks of this practice.
Reference
1. Pitt C, Sanchez-Ramos L, Kaunitz AM. Adjunctive intravaginal metronidazole for the prevention of postcesarean endometritis: a randomized controlled trial. Obstet Gynecol. 2001;98(5 pt 1):745−750.
Cochrane review of precesarean vaginal cleansingAuthors of a Cochrane review, in which they synthesized 7 studies involving 2,635 women, reported that vaginal cleansing with povidone-iodine immediately before cesarean delivery was associated with a reduced risk of postcesarean endometritis: 8.3% vs 4.3% in the control and vaginal cleansing groups, respectively, (risk ratio [RR], 0.45; 95% confidence interval [CI], 0.25−0.81).7
The positive effect of vaginal cleansing was particularly noteworthy in the subgroup of women with ruptured membranes (3 trials involving 272 women). The rates of endometritis in the control versus vaginal cleansing groups were 17.9% and 4.3%, respectively (RR, 0.24; 95% CI, 0.10−0.55).
Women who went into labor prior to cesarean delivery (523 women from 3 trials) also benefitted from vaginal cleansing, with endometritis rates of 13.0% and 7.4% in the control and vaginal cleansing groups, respectively (RR, 0.56; 95% CI, 0.34−0.95).
In this review, again, vaginal cleansing did not significantly reduce the rate of postoperative fever or wound infection.
The American College of Obstetricians and Gynecologists has noted that chlorohexidine gluconate solutions with high concentrations of alcohol are contraindicated for vaginal cleansing.1 However, although not approved for vaginal cleansing, solutions of chlorohexidine gluconate with low alcohol content (4% alcohol concentration) are safe and may be effective for off-label use as vaginal cleansings.
Reference
1. American College of Obstetricians and Gynecologists Women’s Health Care Physicians; Committee on Gynecologic Practice. Committee Opinion No. 571: solutions for surgical cleansing of the vagina. Obstet Gynecol. 2013;122(3):718−720.
Is vaginal cleansing prior to cesarean delivery best practice?In the United States, precesarean vaginal cleansing is not a common practice. To close the gap between current practice and what is potentially a best practice, two approaches to using vaginal cleansing could be instituted in delivery units.
Approach #1: A liberal clinical protocol. In this scenario, all women (who are not allergic to iodine or povidone-iodine) undergoing cesarean delivery should undergo vaginal cleansing. The World Health Organization conditionally recommends vaginal cleansing for all women undergoing a cesarean delivery.8
Approach #2: A focused clinical protocol. For this protocol, only women (again, who are not allergic to iodine or povidone-iodine) who have ruptured membranes or are in labor upon advanced cervical examination should receive vaginal cleansing.
The advantage of a liberal protocol is that vaginal preparation becomes embedded within the standard practice of cesarean delivery and, hence, is seldom overlooked. The upside of the focused protocol is that only those women most likely to benefit receive the intervention.
Tell me what you thinkWill you consider using vaginal cleansing in your practice? Please let me know your views on vaginal cleansing for cesarean delivery, as well as your clinical pearls on cesarean delivery surgery, at obgmanagement.com. In addition, weigh in on the Quick Poll posted to OBG Management’s homepage. Send your letter to the editor to [email protected].
- Starr RV, Zurawski J, Ismail M. Preoperative vaginal preparation with povidone-iodine and the risk of postcesarean endometritis. Obstet Gynecol. 2015;105(5 pt 1):1024–1029.
- Haas DM, Pazouki F, Smith RR, et al. Vaginal cleansing before cesarean delivery to reduce postoperative infectious morbidity: a randomized, controlled trial. Am J Obstet Gynecol. 2010;202(3):310.e1–e6.
- Yildrim G, Gungorduk K, Asicioglu O, et al. Does vaginal preparation with povidone-iodine prior to cesarean delivery reduce the risk of endometritis?A randomized controlled trial. J Matern Fetal Neonatal Med. 2012;25(11):2316–2321.
- Asghania M, Mirblouk F, Shakiba M, Faraji R. Preoperative vaginal preparation with povidone-iodine on post-cesarean infectious morbidity. J Obstet Gynaecol. 2011;31(5):400–403.
- Memon S, Qazi RA, Bibi S, Parveen N. Effect of preoperative vaginal cleansing with an antiseptic solution to reduce post caesarean infectious morbidity. J Pak Med Assoc. 2011;61(12):1179–1183.
- Reid VC, Hartmann KE, McMahon M, Fry EP. Vaginal preparation with povidone-iodine and postcesarean infectious morbidity: a randomized controlled trial. Obstet Gynecol. 2001;97(1):147–152.
- Haas DM, Morgan S, Contreras K. Vaginal preparation with antiseptic solution before cesarean section for preventing postoperative infections. Cochrane Database Syst Rev. 2014;12:CD007892.
- Yildrim G, Gungorduk K, Asicioglu O, et al. Does vaginal preparation with povidone-iodine prior to
- Starr RV, Zurawski J, Ismail M. Preoperative vaginal preparation with povidone-iodine and the risk of postcesarean endometritis. Obstet Gynecol. 2015;105(5 pt 1):1024–1029.
- Haas DM, Pazouki F, Smith RR, et al. Vaginal cleansing before cesarean delivery to reduce postoperative infectious morbidity: a randomized, controlled trial. Am J Obstet Gynecol. 2010;202(3):310.e1–e6.
- Yildrim G, Gungorduk K, Asicioglu O, et al. Does vaginal preparation with povidone-iodine prior to cesarean delivery reduce the risk of endometritis?A randomized controlled trial. J Matern Fetal Neonatal Med. 2012;25(11):2316–2321.
- Asghania M, Mirblouk F, Shakiba M, Faraji R. Preoperative vaginal preparation with povidone-iodine on post-cesarean infectious morbidity. J Obstet Gynaecol. 2011;31(5):400–403.
- Memon S, Qazi RA, Bibi S, Parveen N. Effect of preoperative vaginal cleansing with an antiseptic solution to reduce post caesarean infectious morbidity. J Pak Med Assoc. 2011;61(12):1179–1183.
- Reid VC, Hartmann KE, McMahon M, Fry EP. Vaginal preparation with povidone-iodine and postcesarean infectious morbidity: a randomized controlled trial. Obstet Gynecol. 2001;97(1):147–152.
- Haas DM, Morgan S, Contreras K. Vaginal preparation with antiseptic solution before cesarean section for preventing postoperative infections. Cochrane Database Syst Rev. 2014;12:CD007892.
- Yildrim G, Gungorduk K, Asicioglu O, et al. Does vaginal preparation with povidone-iodine prior to
Can transabdominal ultrasound exclude short cervix?
Preterm birth (PTB) remains a major cause of perinatal morbidity and mortality, and so its prediction and prevention are 2 of the most important issues in obstetrics. Cervical length (CL) measured by ultrasound has been shown to be the best predictor; several interventions (vaginal progesterone and cerclage) have been shown to be effective at reducing PTB if a short CL is identified. In fact, both the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) recommend CL being measured every 2 weeks from 16 to 23 weeks in singletons with prior spontaneous PTB (sPTB), with cerclage placed for CL less than 25 mm. Moreover, both ACOG and SMFM recommend that “universal CL screening” (CL measured in singletons without a prior sPTB) be considered as a single measurement at about 18 to 23 weeks.
Details of the study
Rhoades and colleagues present data on CL screening done by transabdominal ultrasound (TAU), as an alternative to transvaginal ultrasound (TVU). This study confirms early data:
- TAU cannot visualize CL in several women (20.6%).
- To make sure a high sensitivity (92.9% in this study) is achieved to detect a TVU CL less than 30 mm, a high cutoff (in this case 35 mm) needs to be used with TAU. Nonetheless, 7% of women with a short TVU CL would not be detected, raising clinical and legal issues.
- A high percentage (in this case 32.4%; 103/318) of women screened by TAU would screen positive (TAU CL less than 35 mm) and therefore need to have a TVU anyway.
- Overall, more than 50% (in this study 53%–20.6% because TAU could not visualize CL, and 32.4% because TAU was less than 35 mm) of women having TAU CL screening would need to have TVU anyway! In the largest study comparing TAU to TVU CL screening (TABLE1–6), 66% of women screened by TAU would have to be screened also by TVU.5
There are several other reasons why TVU is considered the gold standard for CL screening, and instead TAU CL should be avoided as possible. All randomized controlled trials that showed benefit from interventions (vaginal progesterone, cerclage, pessary) aimed at decreasing PTB in women with short CL used TVU CL screening and never TAU CL screening. In addition, TAU CL is less accurate than TVU CL screening. On TAU, fetal parts can obscure the cervix, obesity makes it hard to visualize CL, the distance between probe and cervix is longer, manual pressure can mask CL shortening, and bladder filling can elongate CL.7 Cost-effectiveness studies show that TVU CL screening is more effective, and less costly, compared with TAU CL screening, even in singletons without a prior sPTB.8
Societies such as ACOG and SMFM all have recommended TVU CL for prediction and prevention of PTB, over TAU CL.9,10 Importantly, a TVU CL should be done by sonographers educated and trained formally, through such programs as those made available by SMFM.11
What this evidence means for practice
If CL assessment is done, TVU should be preferred, as it is the gold standard, and not TAU.
>>Vincenzo Berghella, MD
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Saul LL, Kurtzman JT, Hagemann C, Ghamsary M, Wing DA. Is transabdominal sonography of the cervix after voiding a reliable method of cervical length assessment? J Ultrasound Med. 2008;27(9):1305−1311.
- Stone PR, Chan EH, McCowan LM, Taylor RS, Mitchell JM; SCOPE Consortium. Aust N Z J Obstet Gynaecol. 2010;50(6):523−527.
- To MS, Skentou C, Cicero S, Nicolaides KH. Cervical assessment at the routine 23-weeks’ scan: problems with transabdominal sonography. Ultrasound Obstet Gynecol 2000;15(4):292−296.
- Hernandez-Andrade E, Romero R, Ahn H, et al. Transabdominal evaluation of uterine cervical length during pregnancy fails to identify a substantial number of women with a short cervix. 2012;25(9):1682−1689.
- Friedman AM, Srinivas SK, Parry S, et al. Can transabdominal ultrasound be used as a screening test for short cervical length? Am J Obstet Gynecol. 2013;208(3):190.e1−e7.
- Rhoades JS, Park JM, Stout MJ, Macones GA, Cahill AG, Tuuli MG. Can transabdominal cervical length measurement exclude short cervix? 2015 Nov 2. [Epub ahead of print]
- Berghella V, Bega G, Tolosa JE, Berghella M. Ultrasound assessment of the cervix. Clin Obstet Gynecol. 2003; 46(4):947–623.
- Miller ES, Grobman WA. Cost-effectiveness of transabdominal ultrasound for cervical length screening for preterm birth prevention. Am J Obstet Gynecol. 2013;209(6): 546.e1–e6.
- American College of Obstetricians and Gynecologists. Practice bulletin No. 130: prediction and prevention of preterm birth. Obstet Gynecol. 2012;120(4):964–973.
- Society for Maternal-Fetal Medicine Publications Committee; Berghella V. Progesterone and preterm birth prevention: translating clinical trial data into clinical practice. Am J Obstet Gynecol. 2012;206(5):376–386.
- Cervical Length Education and Review (CLEAR) guidelines. https://clear.perinatalquality.org. Published 2015. Accessed December 15, 2015.
Preterm birth (PTB) remains a major cause of perinatal morbidity and mortality, and so its prediction and prevention are 2 of the most important issues in obstetrics. Cervical length (CL) measured by ultrasound has been shown to be the best predictor; several interventions (vaginal progesterone and cerclage) have been shown to be effective at reducing PTB if a short CL is identified. In fact, both the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) recommend CL being measured every 2 weeks from 16 to 23 weeks in singletons with prior spontaneous PTB (sPTB), with cerclage placed for CL less than 25 mm. Moreover, both ACOG and SMFM recommend that “universal CL screening” (CL measured in singletons without a prior sPTB) be considered as a single measurement at about 18 to 23 weeks.
Details of the study
Rhoades and colleagues present data on CL screening done by transabdominal ultrasound (TAU), as an alternative to transvaginal ultrasound (TVU). This study confirms early data:
- TAU cannot visualize CL in several women (20.6%).
- To make sure a high sensitivity (92.9% in this study) is achieved to detect a TVU CL less than 30 mm, a high cutoff (in this case 35 mm) needs to be used with TAU. Nonetheless, 7% of women with a short TVU CL would not be detected, raising clinical and legal issues.
- A high percentage (in this case 32.4%; 103/318) of women screened by TAU would screen positive (TAU CL less than 35 mm) and therefore need to have a TVU anyway.
- Overall, more than 50% (in this study 53%–20.6% because TAU could not visualize CL, and 32.4% because TAU was less than 35 mm) of women having TAU CL screening would need to have TVU anyway! In the largest study comparing TAU to TVU CL screening (TABLE1–6), 66% of women screened by TAU would have to be screened also by TVU.5
There are several other reasons why TVU is considered the gold standard for CL screening, and instead TAU CL should be avoided as possible. All randomized controlled trials that showed benefit from interventions (vaginal progesterone, cerclage, pessary) aimed at decreasing PTB in women with short CL used TVU CL screening and never TAU CL screening. In addition, TAU CL is less accurate than TVU CL screening. On TAU, fetal parts can obscure the cervix, obesity makes it hard to visualize CL, the distance between probe and cervix is longer, manual pressure can mask CL shortening, and bladder filling can elongate CL.7 Cost-effectiveness studies show that TVU CL screening is more effective, and less costly, compared with TAU CL screening, even in singletons without a prior sPTB.8
Societies such as ACOG and SMFM all have recommended TVU CL for prediction and prevention of PTB, over TAU CL.9,10 Importantly, a TVU CL should be done by sonographers educated and trained formally, through such programs as those made available by SMFM.11
What this evidence means for practice
If CL assessment is done, TVU should be preferred, as it is the gold standard, and not TAU.
>>Vincenzo Berghella, MD
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Preterm birth (PTB) remains a major cause of perinatal morbidity and mortality, and so its prediction and prevention are 2 of the most important issues in obstetrics. Cervical length (CL) measured by ultrasound has been shown to be the best predictor; several interventions (vaginal progesterone and cerclage) have been shown to be effective at reducing PTB if a short CL is identified. In fact, both the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM) recommend CL being measured every 2 weeks from 16 to 23 weeks in singletons with prior spontaneous PTB (sPTB), with cerclage placed for CL less than 25 mm. Moreover, both ACOG and SMFM recommend that “universal CL screening” (CL measured in singletons without a prior sPTB) be considered as a single measurement at about 18 to 23 weeks.
Details of the study
Rhoades and colleagues present data on CL screening done by transabdominal ultrasound (TAU), as an alternative to transvaginal ultrasound (TVU). This study confirms early data:
- TAU cannot visualize CL in several women (20.6%).
- To make sure a high sensitivity (92.9% in this study) is achieved to detect a TVU CL less than 30 mm, a high cutoff (in this case 35 mm) needs to be used with TAU. Nonetheless, 7% of women with a short TVU CL would not be detected, raising clinical and legal issues.
- A high percentage (in this case 32.4%; 103/318) of women screened by TAU would screen positive (TAU CL less than 35 mm) and therefore need to have a TVU anyway.
- Overall, more than 50% (in this study 53%–20.6% because TAU could not visualize CL, and 32.4% because TAU was less than 35 mm) of women having TAU CL screening would need to have TVU anyway! In the largest study comparing TAU to TVU CL screening (TABLE1–6), 66% of women screened by TAU would have to be screened also by TVU.5
There are several other reasons why TVU is considered the gold standard for CL screening, and instead TAU CL should be avoided as possible. All randomized controlled trials that showed benefit from interventions (vaginal progesterone, cerclage, pessary) aimed at decreasing PTB in women with short CL used TVU CL screening and never TAU CL screening. In addition, TAU CL is less accurate than TVU CL screening. On TAU, fetal parts can obscure the cervix, obesity makes it hard to visualize CL, the distance between probe and cervix is longer, manual pressure can mask CL shortening, and bladder filling can elongate CL.7 Cost-effectiveness studies show that TVU CL screening is more effective, and less costly, compared with TAU CL screening, even in singletons without a prior sPTB.8
Societies such as ACOG and SMFM all have recommended TVU CL for prediction and prevention of PTB, over TAU CL.9,10 Importantly, a TVU CL should be done by sonographers educated and trained formally, through such programs as those made available by SMFM.11
What this evidence means for practice
If CL assessment is done, TVU should be preferred, as it is the gold standard, and not TAU.
>>Vincenzo Berghella, MD
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Saul LL, Kurtzman JT, Hagemann C, Ghamsary M, Wing DA. Is transabdominal sonography of the cervix after voiding a reliable method of cervical length assessment? J Ultrasound Med. 2008;27(9):1305−1311.
- Stone PR, Chan EH, McCowan LM, Taylor RS, Mitchell JM; SCOPE Consortium. Aust N Z J Obstet Gynaecol. 2010;50(6):523−527.
- To MS, Skentou C, Cicero S, Nicolaides KH. Cervical assessment at the routine 23-weeks’ scan: problems with transabdominal sonography. Ultrasound Obstet Gynecol 2000;15(4):292−296.
- Hernandez-Andrade E, Romero R, Ahn H, et al. Transabdominal evaluation of uterine cervical length during pregnancy fails to identify a substantial number of women with a short cervix. 2012;25(9):1682−1689.
- Friedman AM, Srinivas SK, Parry S, et al. Can transabdominal ultrasound be used as a screening test for short cervical length? Am J Obstet Gynecol. 2013;208(3):190.e1−e7.
- Rhoades JS, Park JM, Stout MJ, Macones GA, Cahill AG, Tuuli MG. Can transabdominal cervical length measurement exclude short cervix? 2015 Nov 2. [Epub ahead of print]
- Berghella V, Bega G, Tolosa JE, Berghella M. Ultrasound assessment of the cervix. Clin Obstet Gynecol. 2003; 46(4):947–623.
- Miller ES, Grobman WA. Cost-effectiveness of transabdominal ultrasound for cervical length screening for preterm birth prevention. Am J Obstet Gynecol. 2013;209(6): 546.e1–e6.
- American College of Obstetricians and Gynecologists. Practice bulletin No. 130: prediction and prevention of preterm birth. Obstet Gynecol. 2012;120(4):964–973.
- Society for Maternal-Fetal Medicine Publications Committee; Berghella V. Progesterone and preterm birth prevention: translating clinical trial data into clinical practice. Am J Obstet Gynecol. 2012;206(5):376–386.
- Cervical Length Education and Review (CLEAR) guidelines. https://clear.perinatalquality.org. Published 2015. Accessed December 15, 2015.
- Saul LL, Kurtzman JT, Hagemann C, Ghamsary M, Wing DA. Is transabdominal sonography of the cervix after voiding a reliable method of cervical length assessment? J Ultrasound Med. 2008;27(9):1305−1311.
- Stone PR, Chan EH, McCowan LM, Taylor RS, Mitchell JM; SCOPE Consortium. Aust N Z J Obstet Gynaecol. 2010;50(6):523−527.
- To MS, Skentou C, Cicero S, Nicolaides KH. Cervical assessment at the routine 23-weeks’ scan: problems with transabdominal sonography. Ultrasound Obstet Gynecol 2000;15(4):292−296.
- Hernandez-Andrade E, Romero R, Ahn H, et al. Transabdominal evaluation of uterine cervical length during pregnancy fails to identify a substantial number of women with a short cervix. 2012;25(9):1682−1689.
- Friedman AM, Srinivas SK, Parry S, et al. Can transabdominal ultrasound be used as a screening test for short cervical length? Am J Obstet Gynecol. 2013;208(3):190.e1−e7.
- Rhoades JS, Park JM, Stout MJ, Macones GA, Cahill AG, Tuuli MG. Can transabdominal cervical length measurement exclude short cervix? 2015 Nov 2. [Epub ahead of print]
- Berghella V, Bega G, Tolosa JE, Berghella M. Ultrasound assessment of the cervix. Clin Obstet Gynecol. 2003; 46(4):947–623.
- Miller ES, Grobman WA. Cost-effectiveness of transabdominal ultrasound for cervical length screening for preterm birth prevention. Am J Obstet Gynecol. 2013;209(6): 546.e1–e6.
- American College of Obstetricians and Gynecologists. Practice bulletin No. 130: prediction and prevention of preterm birth. Obstet Gynecol. 2012;120(4):964–973.
- Society for Maternal-Fetal Medicine Publications Committee; Berghella V. Progesterone and preterm birth prevention: translating clinical trial data into clinical practice. Am J Obstet Gynecol. 2012;206(5):376–386.
- Cervical Length Education and Review (CLEAR) guidelines. https://clear.perinatalquality.org. Published 2015. Accessed December 15, 2015.
Twice as many adverse events with planned out-of-hospital births
The rates of several adverse outcomes appear to be twice as high among planned out-of-hospital births than planned in-hospital births, based on a study in Oregon that relied on more accurate statistical information about planned birth location.
Out-of-hospital births also were associated with fewer obstetrical procedures, and with increased odds of unassisted vaginal delivery.
Nationally, and in most individual states, data for births that were intended to occur in a hospital can not be separated from data for births that were intended to occur at home or at another nonhospital location such as a birth center but required transfer to a hospital. In effect, intrapartum home-to-hospital transfers were inaccurately counted as in-hospital births, said Dr. Jonathan M. Snowden of the departments of ob.gyn. and public health and preventive medicine, Oregon Health and Science University, Portland, and his associates.
In 2012, Oregon began requiring that birth certificates document both the planned place of delivery at the onset of labor and the actual site of delivery. This change allowed researchers to use birth certificate data to assess more accurately maternal and neonatal outcomes by birth plan.
Dr. Snowden and his associates analyzed data from Oregon birth and death certificates during 2012-2013. They focused on 79,727 singleton term deliveries in which 95.2% of the mothers planned to deliver in hospital and did so, 4% planned and completed an out-of-hospital delivery (1,968 at home and 1,235 at a birth center), and 601 women (0.8%) planned an out-of-hospital delivery but ended up delivering in hospital after an intrapartum transfer.
Of mothers who planned an out-of-hospital delivery, 16.5% actually required transfer to a hospital and gave birth there. Misclassifying these births as simply “in-hospital” according to the previous statistical methods “caused rates of adverse outcomes among planned out-of-hospital births to be underestimated (in some cases, substantially),” the investigators said in a study published online Dec. 31 in the New England Journal of Medicine (doi: 10.1056/NEJMsa1501738).
Before intrapartum transfers were classified more accurately, rates of fetal, perinatal, and neonatal death did not differ significantly between in-hospital and out-of-hospital deliveries. After reclassification, death rates were higher for out-of-hospital than for in-hospital births: the fetal death rate was 2.4 vs. 1.2/1,000 deliveries, the perinatal death rate was 3.9 vs. 1.8 deaths/1,000 deliveries, and the neonatal death rate was 1.6 vs. 0.6/1,000 deliveries.
Using the more accurate data, rates of depressed 5-minute Apgar scores, neonatal seizures, and maternal blood transfusions were significantly higher for out-of-hospital than for in-hospital deliveries.
It is important to note that rates of all serious adverse outcomes were very low across the study groups, and that the absolute differences in risk between in-hospital and out-of-hospital delivery were correspondingly small, Dr. Snowden and his associates said.
Alternatively, the odds of NICU admission were lower with planned out-of-hospital births (adjusted odds ratio, 0.71; 95% confidence interval, 0.55-0.92).
Also, planned out-of-hospital birth remained strongly associated with decreased odds of induced labor (adjusted OR, 0.11; 95% CI, 0.09-0.12), cesarean delivery (adjusted OR, 0.18; 95% CI, 0.16-0.22), and other obstetrical procedures, and with increased odds of unassisted vaginal delivery (adjusted OR, 5.63; 95% CI, 4.84-6.55), the researchers wrote.
The rates of several adverse outcomes appear to be twice as high among planned out-of-hospital births than planned in-hospital births, based on a study in Oregon that relied on more accurate statistical information about planned birth location.
Out-of-hospital births also were associated with fewer obstetrical procedures, and with increased odds of unassisted vaginal delivery.
Nationally, and in most individual states, data for births that were intended to occur in a hospital can not be separated from data for births that were intended to occur at home or at another nonhospital location such as a birth center but required transfer to a hospital. In effect, intrapartum home-to-hospital transfers were inaccurately counted as in-hospital births, said Dr. Jonathan M. Snowden of the departments of ob.gyn. and public health and preventive medicine, Oregon Health and Science University, Portland, and his associates.
In 2012, Oregon began requiring that birth certificates document both the planned place of delivery at the onset of labor and the actual site of delivery. This change allowed researchers to use birth certificate data to assess more accurately maternal and neonatal outcomes by birth plan.
Dr. Snowden and his associates analyzed data from Oregon birth and death certificates during 2012-2013. They focused on 79,727 singleton term deliveries in which 95.2% of the mothers planned to deliver in hospital and did so, 4% planned and completed an out-of-hospital delivery (1,968 at home and 1,235 at a birth center), and 601 women (0.8%) planned an out-of-hospital delivery but ended up delivering in hospital after an intrapartum transfer.
Of mothers who planned an out-of-hospital delivery, 16.5% actually required transfer to a hospital and gave birth there. Misclassifying these births as simply “in-hospital” according to the previous statistical methods “caused rates of adverse outcomes among planned out-of-hospital births to be underestimated (in some cases, substantially),” the investigators said in a study published online Dec. 31 in the New England Journal of Medicine (doi: 10.1056/NEJMsa1501738).
Before intrapartum transfers were classified more accurately, rates of fetal, perinatal, and neonatal death did not differ significantly between in-hospital and out-of-hospital deliveries. After reclassification, death rates were higher for out-of-hospital than for in-hospital births: the fetal death rate was 2.4 vs. 1.2/1,000 deliveries, the perinatal death rate was 3.9 vs. 1.8 deaths/1,000 deliveries, and the neonatal death rate was 1.6 vs. 0.6/1,000 deliveries.
Using the more accurate data, rates of depressed 5-minute Apgar scores, neonatal seizures, and maternal blood transfusions were significantly higher for out-of-hospital than for in-hospital deliveries.
It is important to note that rates of all serious adverse outcomes were very low across the study groups, and that the absolute differences in risk between in-hospital and out-of-hospital delivery were correspondingly small, Dr. Snowden and his associates said.
Alternatively, the odds of NICU admission were lower with planned out-of-hospital births (adjusted odds ratio, 0.71; 95% confidence interval, 0.55-0.92).
Also, planned out-of-hospital birth remained strongly associated with decreased odds of induced labor (adjusted OR, 0.11; 95% CI, 0.09-0.12), cesarean delivery (adjusted OR, 0.18; 95% CI, 0.16-0.22), and other obstetrical procedures, and with increased odds of unassisted vaginal delivery (adjusted OR, 5.63; 95% CI, 4.84-6.55), the researchers wrote.
The rates of several adverse outcomes appear to be twice as high among planned out-of-hospital births than planned in-hospital births, based on a study in Oregon that relied on more accurate statistical information about planned birth location.
Out-of-hospital births also were associated with fewer obstetrical procedures, and with increased odds of unassisted vaginal delivery.
Nationally, and in most individual states, data for births that were intended to occur in a hospital can not be separated from data for births that were intended to occur at home or at another nonhospital location such as a birth center but required transfer to a hospital. In effect, intrapartum home-to-hospital transfers were inaccurately counted as in-hospital births, said Dr. Jonathan M. Snowden of the departments of ob.gyn. and public health and preventive medicine, Oregon Health and Science University, Portland, and his associates.
In 2012, Oregon began requiring that birth certificates document both the planned place of delivery at the onset of labor and the actual site of delivery. This change allowed researchers to use birth certificate data to assess more accurately maternal and neonatal outcomes by birth plan.
Dr. Snowden and his associates analyzed data from Oregon birth and death certificates during 2012-2013. They focused on 79,727 singleton term deliveries in which 95.2% of the mothers planned to deliver in hospital and did so, 4% planned and completed an out-of-hospital delivery (1,968 at home and 1,235 at a birth center), and 601 women (0.8%) planned an out-of-hospital delivery but ended up delivering in hospital after an intrapartum transfer.
Of mothers who planned an out-of-hospital delivery, 16.5% actually required transfer to a hospital and gave birth there. Misclassifying these births as simply “in-hospital” according to the previous statistical methods “caused rates of adverse outcomes among planned out-of-hospital births to be underestimated (in some cases, substantially),” the investigators said in a study published online Dec. 31 in the New England Journal of Medicine (doi: 10.1056/NEJMsa1501738).
Before intrapartum transfers were classified more accurately, rates of fetal, perinatal, and neonatal death did not differ significantly between in-hospital and out-of-hospital deliveries. After reclassification, death rates were higher for out-of-hospital than for in-hospital births: the fetal death rate was 2.4 vs. 1.2/1,000 deliveries, the perinatal death rate was 3.9 vs. 1.8 deaths/1,000 deliveries, and the neonatal death rate was 1.6 vs. 0.6/1,000 deliveries.
Using the more accurate data, rates of depressed 5-minute Apgar scores, neonatal seizures, and maternal blood transfusions were significantly higher for out-of-hospital than for in-hospital deliveries.
It is important to note that rates of all serious adverse outcomes were very low across the study groups, and that the absolute differences in risk between in-hospital and out-of-hospital delivery were correspondingly small, Dr. Snowden and his associates said.
Alternatively, the odds of NICU admission were lower with planned out-of-hospital births (adjusted odds ratio, 0.71; 95% confidence interval, 0.55-0.92).
Also, planned out-of-hospital birth remained strongly associated with decreased odds of induced labor (adjusted OR, 0.11; 95% CI, 0.09-0.12), cesarean delivery (adjusted OR, 0.18; 95% CI, 0.16-0.22), and other obstetrical procedures, and with increased odds of unassisted vaginal delivery (adjusted OR, 5.63; 95% CI, 4.84-6.55), the researchers wrote.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: More accurate statistical methods show higher rates of several adverse outcomes among planned out-of-hospital births.
Major finding: 16.5% of mothers who planned an out-of-hospital delivery actually required transfer to a hospital and gave birth there, causing substantial underestimation of adverse outcomes for planned out-of-hospital deliveries.
Data source: A population-based retrospective cohort study involving 79,727 singleton term deliveries in Oregon during a 2-year period.
Disclosures: This study was principally supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development, and also by the Oregon Building Independent Careers in Women’s Health Scholars, the Jonas Doctoral Scholars Program, Sigma Theta Tau Beta Psi, and the American College of Nurse-Midwives.
Is metformin a future preeclampsia treatment?
Metformin decreased toxin levels implicated in preeclampsia, while also helping to heal injured omental blood vessels, researchers in Australia have reported.
Since the drug, a common treatment for type 2 diabetes, is already proven safe during pregnancy, the study’s lead author Dr. Fiona Brownfoot said clinical trials to test metformin’s efficacy in preventing and treating preeclampsia are “justified”. The findings were published online Dec. 22 in the American Journal of Obstetrics and Gynecology (doi: 10.1016/j.ajog.2015.12.019).
Preeclampsia is characterized by elevated levels of soluble vascular endothelial growth factor receptor 1 and soluble endoglin, toxins produced by the placenta. When elevated, these toxins tend to damage endothelial cells, manifesting clinically as hypertension and multisystem organ injury. Preeclampsia occurs in 5%-8% of pregnancies, presenting after week 20 of gestation with new-onset hypertension and proteinuria. Since there have been no agents or drugs to decrease the production of these toxins in pregnancy, the only treatment options for preeclampsia are expectant management and delivery.
Dr. Brownfoot, a researcher in the Translational Obstetrics Group in the department of obstetrics and gynecology at the University of Melbourne and her associates found that when endothelial and placental cells taken from 23 women diagnosed with severe preterm proteinuric preeclampsia were incubated with metformin, there were improvements in endothelial dysfunction when compared with 25 gestationally matched controls. Metformin was also shown to improve whole blood vessel angiogenesis impaired by elevated toxin levels.
“Metformin appears to be the aspirin of the 21st century,” Dr. Roberto Romero, editor-in-chief of the American Journal of Obstetrics and Gynecology, said in a statement.
Noting metformin’s use in diabetes as well as in polycystic ovarian disease, and its recently discovered anticancer properties, Dr. Romero said the recent findings are “exciting and promising,” particularly since antiangiogenesis occurs not only in preeclampsia, but in other pregnancy complications such as fetal death, fetal growth restriction, and premature labor. He called for systematic reviews of previous randomized clinical trials of metformin in pregnant women, as well as new randomized studies to determine the effectiveness of metformin in preventing preeclampsia.
The work was funded by the National Health and Medical Research Council of Australia. The researchers reported having no conflicts of interest.
On Twitter @whitneymcknight
Metformin decreased toxin levels implicated in preeclampsia, while also helping to heal injured omental blood vessels, researchers in Australia have reported.
Since the drug, a common treatment for type 2 diabetes, is already proven safe during pregnancy, the study’s lead author Dr. Fiona Brownfoot said clinical trials to test metformin’s efficacy in preventing and treating preeclampsia are “justified”. The findings were published online Dec. 22 in the American Journal of Obstetrics and Gynecology (doi: 10.1016/j.ajog.2015.12.019).
Preeclampsia is characterized by elevated levels of soluble vascular endothelial growth factor receptor 1 and soluble endoglin, toxins produced by the placenta. When elevated, these toxins tend to damage endothelial cells, manifesting clinically as hypertension and multisystem organ injury. Preeclampsia occurs in 5%-8% of pregnancies, presenting after week 20 of gestation with new-onset hypertension and proteinuria. Since there have been no agents or drugs to decrease the production of these toxins in pregnancy, the only treatment options for preeclampsia are expectant management and delivery.
Dr. Brownfoot, a researcher in the Translational Obstetrics Group in the department of obstetrics and gynecology at the University of Melbourne and her associates found that when endothelial and placental cells taken from 23 women diagnosed with severe preterm proteinuric preeclampsia were incubated with metformin, there were improvements in endothelial dysfunction when compared with 25 gestationally matched controls. Metformin was also shown to improve whole blood vessel angiogenesis impaired by elevated toxin levels.
“Metformin appears to be the aspirin of the 21st century,” Dr. Roberto Romero, editor-in-chief of the American Journal of Obstetrics and Gynecology, said in a statement.
Noting metformin’s use in diabetes as well as in polycystic ovarian disease, and its recently discovered anticancer properties, Dr. Romero said the recent findings are “exciting and promising,” particularly since antiangiogenesis occurs not only in preeclampsia, but in other pregnancy complications such as fetal death, fetal growth restriction, and premature labor. He called for systematic reviews of previous randomized clinical trials of metformin in pregnant women, as well as new randomized studies to determine the effectiveness of metformin in preventing preeclampsia.
The work was funded by the National Health and Medical Research Council of Australia. The researchers reported having no conflicts of interest.
On Twitter @whitneymcknight
Metformin decreased toxin levels implicated in preeclampsia, while also helping to heal injured omental blood vessels, researchers in Australia have reported.
Since the drug, a common treatment for type 2 diabetes, is already proven safe during pregnancy, the study’s lead author Dr. Fiona Brownfoot said clinical trials to test metformin’s efficacy in preventing and treating preeclampsia are “justified”. The findings were published online Dec. 22 in the American Journal of Obstetrics and Gynecology (doi: 10.1016/j.ajog.2015.12.019).
Preeclampsia is characterized by elevated levels of soluble vascular endothelial growth factor receptor 1 and soluble endoglin, toxins produced by the placenta. When elevated, these toxins tend to damage endothelial cells, manifesting clinically as hypertension and multisystem organ injury. Preeclampsia occurs in 5%-8% of pregnancies, presenting after week 20 of gestation with new-onset hypertension and proteinuria. Since there have been no agents or drugs to decrease the production of these toxins in pregnancy, the only treatment options for preeclampsia are expectant management and delivery.
Dr. Brownfoot, a researcher in the Translational Obstetrics Group in the department of obstetrics and gynecology at the University of Melbourne and her associates found that when endothelial and placental cells taken from 23 women diagnosed with severe preterm proteinuric preeclampsia were incubated with metformin, there were improvements in endothelial dysfunction when compared with 25 gestationally matched controls. Metformin was also shown to improve whole blood vessel angiogenesis impaired by elevated toxin levels.
“Metformin appears to be the aspirin of the 21st century,” Dr. Roberto Romero, editor-in-chief of the American Journal of Obstetrics and Gynecology, said in a statement.
Noting metformin’s use in diabetes as well as in polycystic ovarian disease, and its recently discovered anticancer properties, Dr. Romero said the recent findings are “exciting and promising,” particularly since antiangiogenesis occurs not only in preeclampsia, but in other pregnancy complications such as fetal death, fetal growth restriction, and premature labor. He called for systematic reviews of previous randomized clinical trials of metformin in pregnant women, as well as new randomized studies to determine the effectiveness of metformin in preventing preeclampsia.
The work was funded by the National Health and Medical Research Council of Australia. The researchers reported having no conflicts of interest.
On Twitter @whitneymcknight
FROM THE AMERICAN JOURNAL OF OBSTETRICS AND GYNECOLOGY
ACOG, SMFM support short-term use of magnesium sulfate in pregnancy
The American College of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine continue to support the use of magnesium sulfate injection during pregnancy in limited circumstances and for short periods of time, despite a warning from the Food and Drug Administration that the drug has the potential to harm a developing fetus.
In 2013, the Food and Drug Administration issued a safety announcement warning against the prolonged use of intravenous magnesium sulfate (beyond 5-7 days) to treat preterm labor, citing risks of bone changes and low calcium levels in the developing fetus. Agency officials also changed the Pregnancy category classification for magnesium sulfate from a category A to a category D.
The FDA announcement highlighted that although use of magnesium sulfate for preterm labor longer than 5-7 days was considered “off label,” it is approved for the treatment of preeclampsia.
But in a committee opinion issued on Dec. 22, ACOG and the Society for Maternal-Fetal Medicine (SMFM) pointed out that the FDA changes were prompted by reports of fractures and bone demineralization after magnesium sulfate treatment for “a much longer duration and much higher dose than is currently recommended for obstetric use.” In the cases cited by the FDA, average exposure was 9.6 weeks with an average total maternal dose of 3,700 g, according to the committee opinion (Obstet Gynecol. 2016;127:e52-3.).
Magnesium sulfate has been used and studied in obstetrics for many years without concerns for fractures or bone demineralization being raised, including in recent trials of magnesium for neuroprotection, ACOG and SMFM officials wrote.
The updated committee opinion outlines the continued short-term (less than 48 hours) use of magnesium sulfate for certain conditions:
• Prevention and treatment of seizures in women with preeclampsia or eclampsia.
• Preterm labor at less than 32 weeks’ gestation for fetal neuroprotection.
• Prolonging pregnancy for up to 48 hours to allow for administration of antenatal corticosteroids in women at risk for preterm delivery within 7 days.
“Practitioners should not stop using magnesium sulfate for these indications based on the FDA reclassification,” ACOG and SMFM committee members wrote. “In all of these conditions, prolonged use of magnesium sulfate is never indicated. Therefore, the FDA’s change in the pregnancy classification of magnesium sulfate addresses an unindicated and nonstandard use of this medication.”
The American College of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine continue to support the use of magnesium sulfate injection during pregnancy in limited circumstances and for short periods of time, despite a warning from the Food and Drug Administration that the drug has the potential to harm a developing fetus.
In 2013, the Food and Drug Administration issued a safety announcement warning against the prolonged use of intravenous magnesium sulfate (beyond 5-7 days) to treat preterm labor, citing risks of bone changes and low calcium levels in the developing fetus. Agency officials also changed the Pregnancy category classification for magnesium sulfate from a category A to a category D.
The FDA announcement highlighted that although use of magnesium sulfate for preterm labor longer than 5-7 days was considered “off label,” it is approved for the treatment of preeclampsia.
But in a committee opinion issued on Dec. 22, ACOG and the Society for Maternal-Fetal Medicine (SMFM) pointed out that the FDA changes were prompted by reports of fractures and bone demineralization after magnesium sulfate treatment for “a much longer duration and much higher dose than is currently recommended for obstetric use.” In the cases cited by the FDA, average exposure was 9.6 weeks with an average total maternal dose of 3,700 g, according to the committee opinion (Obstet Gynecol. 2016;127:e52-3.).
Magnesium sulfate has been used and studied in obstetrics for many years without concerns for fractures or bone demineralization being raised, including in recent trials of magnesium for neuroprotection, ACOG and SMFM officials wrote.
The updated committee opinion outlines the continued short-term (less than 48 hours) use of magnesium sulfate for certain conditions:
• Prevention and treatment of seizures in women with preeclampsia or eclampsia.
• Preterm labor at less than 32 weeks’ gestation for fetal neuroprotection.
• Prolonging pregnancy for up to 48 hours to allow for administration of antenatal corticosteroids in women at risk for preterm delivery within 7 days.
“Practitioners should not stop using magnesium sulfate for these indications based on the FDA reclassification,” ACOG and SMFM committee members wrote. “In all of these conditions, prolonged use of magnesium sulfate is never indicated. Therefore, the FDA’s change in the pregnancy classification of magnesium sulfate addresses an unindicated and nonstandard use of this medication.”
The American College of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine continue to support the use of magnesium sulfate injection during pregnancy in limited circumstances and for short periods of time, despite a warning from the Food and Drug Administration that the drug has the potential to harm a developing fetus.
In 2013, the Food and Drug Administration issued a safety announcement warning against the prolonged use of intravenous magnesium sulfate (beyond 5-7 days) to treat preterm labor, citing risks of bone changes and low calcium levels in the developing fetus. Agency officials also changed the Pregnancy category classification for magnesium sulfate from a category A to a category D.
The FDA announcement highlighted that although use of magnesium sulfate for preterm labor longer than 5-7 days was considered “off label,” it is approved for the treatment of preeclampsia.
But in a committee opinion issued on Dec. 22, ACOG and the Society for Maternal-Fetal Medicine (SMFM) pointed out that the FDA changes were prompted by reports of fractures and bone demineralization after magnesium sulfate treatment for “a much longer duration and much higher dose than is currently recommended for obstetric use.” In the cases cited by the FDA, average exposure was 9.6 weeks with an average total maternal dose of 3,700 g, according to the committee opinion (Obstet Gynecol. 2016;127:e52-3.).
Magnesium sulfate has been used and studied in obstetrics for many years without concerns for fractures or bone demineralization being raised, including in recent trials of magnesium for neuroprotection, ACOG and SMFM officials wrote.
The updated committee opinion outlines the continued short-term (less than 48 hours) use of magnesium sulfate for certain conditions:
• Prevention and treatment of seizures in women with preeclampsia or eclampsia.
• Preterm labor at less than 32 weeks’ gestation for fetal neuroprotection.
• Prolonging pregnancy for up to 48 hours to allow for administration of antenatal corticosteroids in women at risk for preterm delivery within 7 days.
“Practitioners should not stop using magnesium sulfate for these indications based on the FDA reclassification,” ACOG and SMFM committee members wrote. “In all of these conditions, prolonged use of magnesium sulfate is never indicated. Therefore, the FDA’s change in the pregnancy classification of magnesium sulfate addresses an unindicated and nonstandard use of this medication.”
Chagas disease: Neither foreign nor untreatable
Chagas disease is a vector-borne parasitic disease, endemic to the Americas, that remains as little recognized by U.S. patients and practitioners as the obscure winged insects that transmit it.
Transmission occurs when triatomine bugs, commonly called “kissing bugs,” pierce the skin to feed and leave behind parasite-infected feces that can enter the bloodstream; pregnant women can also transmit Chagas to their newborns.
About a third of patients infected with Trypanosoma cruzi, the protozoan parasite that causes Chagas, will develop cardiac abnormalities such as cardiomyopathy, arrhythmias, and heart failure – often decades after becoming infected. In the United States, where blood banks began screening for Chagas in 2007, patients without symptoms are likely to learn they are positive only after donating blood.
Conventional wisdom has long maintained that Chagas is limited to Central and South America. But immigration from Chagas-endemic countries, such as El Salvador, Mexico, and Bolivia, means more people are living with the disease in the United States.
“One percent of the Latin American immigrant population we screen [in Los Angeles] has Chagas,” said Dr. Sheba K. Meymandi, cardiologist and director of Center of Excellence for Chagas Disease at Olive View–UCLA Medical Center in Los Angeles, who also works with the city’s health department to detect Chagas. “That’s huge.”
Meanwhile, blood banks are discovering more cases among people without ties to Latin America, and species of kissing bugs native to the southern United States are increasingly recognized as a non-negligible source of Chagas transmission. Of 39 Chagas cases reported to Texas health authorities in 2013 and 2014, 12 were thought to be locally acquired.
Dr. Heather Yun, an infectious disease specialist at the San Antonio Military Medical Center, said risk factors for local transmission are not well established, but “we think people who are living in poverty in substandard housing, people who spend a lot of time outdoors, especially at night, and people involved with direct blood contact with wild game in Southern parts of the United States” may be at higher risk.
A U.S. disease
Evidence is amassing quickly that Chagas is a U.S. disease. But U.S. clinicians still lag in their knowledge of it, say physicians treating Chagas cases. “In medical school we get a 2-hour lecture on it, and it’s always been presented as an exotic disease and one you don’t treat,” Dr. Meymandi said.
The persistent perception of Chagas as a foreign disease means clinicians are inclined to dismiss positive results from a blood screening, particularly from someone who is not from Latin America. Yet cardiologists, ID practitioners, obstetricians, and primary care physicians all need to be aware that cases do occur in the United States and are potentially treatable.
Dr. Laila Woc-Colburn, an infectious disease and tropical medicine specialist at Baylor College of Medicine in Houston, said many people with Chagas never make it to an infectious disease specialist or cardiologist for a work-up. “When you test positive on serology [after a blood donation], you get a letter recommending you consult your physician. Most will go to their primary care doctors, who might say ‘this isn’t a disease in the United States.’ In Houston, that is often the case.”
Dr. Meymandi, who has treated hundreds of patients with Chagas with and without cardiac involvement, said any physician with a potential Chagas case must act. “If you get someone that’s positive, it’s your duty as a physician to confirm the positivity with CDC,” she said.
Dr. Yun concurred. “The most important message is, do something,” she said. “Don’t just assume it’s a false positive.”
Diagnosis is not simple and requires testing beyond the initial ELISA assay used in blood-bank screening. Confirmatory tests must be carried out in coordination with the Centers for Disease Control and Prevention. Also, with no agents approved by the Food and Drug Administration to treat Chagas, treatment is available only through the CDC’s investigational drugs protocol. Both drugs used in Chagas, benznidazole and nifurtimox, come with serious adverse effects that must be closely monitored.
“It’s time consuming, filling out the forms, getting the consent, tracking and sending back lab results to CDC in order to get drugs – it’s not like you can just write a prescription,” Dr. Meymandi said. But, “if you don’t know how to treat the patient or don’t have time, find someone like me,” she noted, adding that she is available to counsel any physician daunted by a potential Chagas case.
Treatment options
No formal clinical algorithm exists for Chagas, but Dr. Meymandi, Dr. Yun, and Dr. Woc-Colburn all pointed to a 2007 JAMA article, which describes diagnosis and treatment protocols, as an important reference for clinicians to start with. It’s “the best approximation of a clinical guideline we have,” Dr. Yun said (JAMA. 2007;298[18]:2171-81. doi:10.1001/jama.298.18.2171).
Dr. Meymandi, who has treated more Chagas patients than has any other U.S. clinician, said that treatment has changed somewhat since the JAMA article was published. In 2007, she said, nifurtimox was the main drug available through CDC, while benznidazole, which is somewhat better tolerated and has shorter treatment duration, has since become the first-line agent.
“We’ve lowered the dose of benznidazole, maxing out at 400 mg/day to decrease the toxicity,” she said. Also, treatment is now being extended to some patients aged 60 years and older.
The decision to treat or not treat, clinicians say, depends on the patient’s age, disease progression, comorbidities and potential serious drug interactions, and willingness to tolerate side effects that, with nifurtimox especially, can include skin sloughing, rash, and psychological and neurologic symptoms including depression and peripheral neuropathy.
“If you don’t have side effects, you’re not taking the drugs,” Dr. Meymandi said. Dr. Woc-Colburn noted that polypharmacy was a major consideration when treating older adults for Chagas. “If I have a patient who has diabetes, obesity, [and] end-stage renal disease, it’s not going to be ideal to give [benznidazole].”
Recent, highly anticipated results from BENEFIT, a large randomized trial (n = 2,854) showed that benznidazole reduced parasite load but was not helpful in halting cardiac damage at 5 years’ follow-up in patients with established Chagas cardiomyopathy (N Engl J Med. 2015 Oct;373:1295-306. doi:10.1056/NEJMoa1507574).
Dr. Meymandi, whose earlier research established that Chagas cardiomyopathy carries significantly higher morbidity and mortality than does non–Chagas cardiomyopathy (Circulation. 2012;126:A18171), said that the BENEFIT results underscore the need for physicians to be bullish in their approach to treating Chagas soon after diagnosis.
“It doesn’t matter if they’re symptomatic or asymptomatic. You can’t wait till they progress to treat. If you wait for the progression of disease you’ve lost the battle. You can’t wait and follow conservatively until you see the complications, because once those complications have started the parasitic load is too high for you to have an impact,” she said.
Dr. Yun said that given the toxicity of current treatment, she hoped to see more studies show clearer evidence of clinical benefit, “either reductions in mortality or reductions in end organ disease.” Most studies “have focused on clearance of parasite, which is important, but it’s not as important decreasing the risk of death or cardiomyopathy or heart failure.”
Rick Tarleton, Ph.D., a biologist the University of Georgia, in Athens, who has worked on Chagas for more than 30 years, said that because Chagas pathology is directly tied to parasite load – and not, as people have suggested in the past, an autoimmune reaction resulting from parasite exposure – drug treatment may prove to be worthwhile even in patients with significant cardiac involvement.
“You get rid of the parasite, you get rid of the progression of the disease,” Dr. Tarleton said. Even the findings from the BENEFIT trial, he said, did not lead him to conclude that treatment in people with established cardiac disease was futile.
“If you’re treating people who are already chronically infected and showing symptoms, the question is not have you reversed the damage, it’s have you stopped accumulating damage,” he noted. “And a 5-year follow-up is probably not long enough to know whether you’ve stopped accumulating.”
“We have drugs, they’re not great, they do have side effects, they don’t always work,” Dr. Tarleton said. “But they’re better than nothing. And they ought to be more widely used.”
Dr. Meymandi said that current supplies of benznidazole at CDC are low and that a dozen patients at her clinic are awaiting treatment. Meanwhile, access may soon be complicated further by the announcement, this month, that KaloBios Pharmaceuticals had bought the rights to seek FDA approval of benznidazole and market it in the United States.
The same company’s CEO came under fire in recent months for acquiring rights to an inexpensive drug to treat toxoplasmosis in AIDS patients, then announcing a price increase from $13.50 to $750 a pill.
“Everyone’s really concerned,” Dr. Meymandi said, “because Chagas is a disease of the poor.”
Chagas disease is a vector-borne parasitic disease, endemic to the Americas, that remains as little recognized by U.S. patients and practitioners as the obscure winged insects that transmit it.
Transmission occurs when triatomine bugs, commonly called “kissing bugs,” pierce the skin to feed and leave behind parasite-infected feces that can enter the bloodstream; pregnant women can also transmit Chagas to their newborns.
About a third of patients infected with Trypanosoma cruzi, the protozoan parasite that causes Chagas, will develop cardiac abnormalities such as cardiomyopathy, arrhythmias, and heart failure – often decades after becoming infected. In the United States, where blood banks began screening for Chagas in 2007, patients without symptoms are likely to learn they are positive only after donating blood.
Conventional wisdom has long maintained that Chagas is limited to Central and South America. But immigration from Chagas-endemic countries, such as El Salvador, Mexico, and Bolivia, means more people are living with the disease in the United States.
“One percent of the Latin American immigrant population we screen [in Los Angeles] has Chagas,” said Dr. Sheba K. Meymandi, cardiologist and director of Center of Excellence for Chagas Disease at Olive View–UCLA Medical Center in Los Angeles, who also works with the city’s health department to detect Chagas. “That’s huge.”
Meanwhile, blood banks are discovering more cases among people without ties to Latin America, and species of kissing bugs native to the southern United States are increasingly recognized as a non-negligible source of Chagas transmission. Of 39 Chagas cases reported to Texas health authorities in 2013 and 2014, 12 were thought to be locally acquired.
Dr. Heather Yun, an infectious disease specialist at the San Antonio Military Medical Center, said risk factors for local transmission are not well established, but “we think people who are living in poverty in substandard housing, people who spend a lot of time outdoors, especially at night, and people involved with direct blood contact with wild game in Southern parts of the United States” may be at higher risk.
A U.S. disease
Evidence is amassing quickly that Chagas is a U.S. disease. But U.S. clinicians still lag in their knowledge of it, say physicians treating Chagas cases. “In medical school we get a 2-hour lecture on it, and it’s always been presented as an exotic disease and one you don’t treat,” Dr. Meymandi said.
The persistent perception of Chagas as a foreign disease means clinicians are inclined to dismiss positive results from a blood screening, particularly from someone who is not from Latin America. Yet cardiologists, ID practitioners, obstetricians, and primary care physicians all need to be aware that cases do occur in the United States and are potentially treatable.
Dr. Laila Woc-Colburn, an infectious disease and tropical medicine specialist at Baylor College of Medicine in Houston, said many people with Chagas never make it to an infectious disease specialist or cardiologist for a work-up. “When you test positive on serology [after a blood donation], you get a letter recommending you consult your physician. Most will go to their primary care doctors, who might say ‘this isn’t a disease in the United States.’ In Houston, that is often the case.”
Dr. Meymandi, who has treated hundreds of patients with Chagas with and without cardiac involvement, said any physician with a potential Chagas case must act. “If you get someone that’s positive, it’s your duty as a physician to confirm the positivity with CDC,” she said.
Dr. Yun concurred. “The most important message is, do something,” she said. “Don’t just assume it’s a false positive.”
Diagnosis is not simple and requires testing beyond the initial ELISA assay used in blood-bank screening. Confirmatory tests must be carried out in coordination with the Centers for Disease Control and Prevention. Also, with no agents approved by the Food and Drug Administration to treat Chagas, treatment is available only through the CDC’s investigational drugs protocol. Both drugs used in Chagas, benznidazole and nifurtimox, come with serious adverse effects that must be closely monitored.
“It’s time consuming, filling out the forms, getting the consent, tracking and sending back lab results to CDC in order to get drugs – it’s not like you can just write a prescription,” Dr. Meymandi said. But, “if you don’t know how to treat the patient or don’t have time, find someone like me,” she noted, adding that she is available to counsel any physician daunted by a potential Chagas case.
Treatment options
No formal clinical algorithm exists for Chagas, but Dr. Meymandi, Dr. Yun, and Dr. Woc-Colburn all pointed to a 2007 JAMA article, which describes diagnosis and treatment protocols, as an important reference for clinicians to start with. It’s “the best approximation of a clinical guideline we have,” Dr. Yun said (JAMA. 2007;298[18]:2171-81. doi:10.1001/jama.298.18.2171).
Dr. Meymandi, who has treated more Chagas patients than has any other U.S. clinician, said that treatment has changed somewhat since the JAMA article was published. In 2007, she said, nifurtimox was the main drug available through CDC, while benznidazole, which is somewhat better tolerated and has shorter treatment duration, has since become the first-line agent.
“We’ve lowered the dose of benznidazole, maxing out at 400 mg/day to decrease the toxicity,” she said. Also, treatment is now being extended to some patients aged 60 years and older.
The decision to treat or not treat, clinicians say, depends on the patient’s age, disease progression, comorbidities and potential serious drug interactions, and willingness to tolerate side effects that, with nifurtimox especially, can include skin sloughing, rash, and psychological and neurologic symptoms including depression and peripheral neuropathy.
“If you don’t have side effects, you’re not taking the drugs,” Dr. Meymandi said. Dr. Woc-Colburn noted that polypharmacy was a major consideration when treating older adults for Chagas. “If I have a patient who has diabetes, obesity, [and] end-stage renal disease, it’s not going to be ideal to give [benznidazole].”
Recent, highly anticipated results from BENEFIT, a large randomized trial (n = 2,854) showed that benznidazole reduced parasite load but was not helpful in halting cardiac damage at 5 years’ follow-up in patients with established Chagas cardiomyopathy (N Engl J Med. 2015 Oct;373:1295-306. doi:10.1056/NEJMoa1507574).
Dr. Meymandi, whose earlier research established that Chagas cardiomyopathy carries significantly higher morbidity and mortality than does non–Chagas cardiomyopathy (Circulation. 2012;126:A18171), said that the BENEFIT results underscore the need for physicians to be bullish in their approach to treating Chagas soon after diagnosis.
“It doesn’t matter if they’re symptomatic or asymptomatic. You can’t wait till they progress to treat. If you wait for the progression of disease you’ve lost the battle. You can’t wait and follow conservatively until you see the complications, because once those complications have started the parasitic load is too high for you to have an impact,” she said.
Dr. Yun said that given the toxicity of current treatment, she hoped to see more studies show clearer evidence of clinical benefit, “either reductions in mortality or reductions in end organ disease.” Most studies “have focused on clearance of parasite, which is important, but it’s not as important decreasing the risk of death or cardiomyopathy or heart failure.”
Rick Tarleton, Ph.D., a biologist the University of Georgia, in Athens, who has worked on Chagas for more than 30 years, said that because Chagas pathology is directly tied to parasite load – and not, as people have suggested in the past, an autoimmune reaction resulting from parasite exposure – drug treatment may prove to be worthwhile even in patients with significant cardiac involvement.
“You get rid of the parasite, you get rid of the progression of the disease,” Dr. Tarleton said. Even the findings from the BENEFIT trial, he said, did not lead him to conclude that treatment in people with established cardiac disease was futile.
“If you’re treating people who are already chronically infected and showing symptoms, the question is not have you reversed the damage, it’s have you stopped accumulating damage,” he noted. “And a 5-year follow-up is probably not long enough to know whether you’ve stopped accumulating.”
“We have drugs, they’re not great, they do have side effects, they don’t always work,” Dr. Tarleton said. “But they’re better than nothing. And they ought to be more widely used.”
Dr. Meymandi said that current supplies of benznidazole at CDC are low and that a dozen patients at her clinic are awaiting treatment. Meanwhile, access may soon be complicated further by the announcement, this month, that KaloBios Pharmaceuticals had bought the rights to seek FDA approval of benznidazole and market it in the United States.
The same company’s CEO came under fire in recent months for acquiring rights to an inexpensive drug to treat toxoplasmosis in AIDS patients, then announcing a price increase from $13.50 to $750 a pill.
“Everyone’s really concerned,” Dr. Meymandi said, “because Chagas is a disease of the poor.”
Chagas disease is a vector-borne parasitic disease, endemic to the Americas, that remains as little recognized by U.S. patients and practitioners as the obscure winged insects that transmit it.
Transmission occurs when triatomine bugs, commonly called “kissing bugs,” pierce the skin to feed and leave behind parasite-infected feces that can enter the bloodstream; pregnant women can also transmit Chagas to their newborns.
About a third of patients infected with Trypanosoma cruzi, the protozoan parasite that causes Chagas, will develop cardiac abnormalities such as cardiomyopathy, arrhythmias, and heart failure – often decades after becoming infected. In the United States, where blood banks began screening for Chagas in 2007, patients without symptoms are likely to learn they are positive only after donating blood.
Conventional wisdom has long maintained that Chagas is limited to Central and South America. But immigration from Chagas-endemic countries, such as El Salvador, Mexico, and Bolivia, means more people are living with the disease in the United States.
“One percent of the Latin American immigrant population we screen [in Los Angeles] has Chagas,” said Dr. Sheba K. Meymandi, cardiologist and director of Center of Excellence for Chagas Disease at Olive View–UCLA Medical Center in Los Angeles, who also works with the city’s health department to detect Chagas. “That’s huge.”
Meanwhile, blood banks are discovering more cases among people without ties to Latin America, and species of kissing bugs native to the southern United States are increasingly recognized as a non-negligible source of Chagas transmission. Of 39 Chagas cases reported to Texas health authorities in 2013 and 2014, 12 were thought to be locally acquired.
Dr. Heather Yun, an infectious disease specialist at the San Antonio Military Medical Center, said risk factors for local transmission are not well established, but “we think people who are living in poverty in substandard housing, people who spend a lot of time outdoors, especially at night, and people involved with direct blood contact with wild game in Southern parts of the United States” may be at higher risk.
A U.S. disease
Evidence is amassing quickly that Chagas is a U.S. disease. But U.S. clinicians still lag in their knowledge of it, say physicians treating Chagas cases. “In medical school we get a 2-hour lecture on it, and it’s always been presented as an exotic disease and one you don’t treat,” Dr. Meymandi said.
The persistent perception of Chagas as a foreign disease means clinicians are inclined to dismiss positive results from a blood screening, particularly from someone who is not from Latin America. Yet cardiologists, ID practitioners, obstetricians, and primary care physicians all need to be aware that cases do occur in the United States and are potentially treatable.
Dr. Laila Woc-Colburn, an infectious disease and tropical medicine specialist at Baylor College of Medicine in Houston, said many people with Chagas never make it to an infectious disease specialist or cardiologist for a work-up. “When you test positive on serology [after a blood donation], you get a letter recommending you consult your physician. Most will go to their primary care doctors, who might say ‘this isn’t a disease in the United States.’ In Houston, that is often the case.”
Dr. Meymandi, who has treated hundreds of patients with Chagas with and without cardiac involvement, said any physician with a potential Chagas case must act. “If you get someone that’s positive, it’s your duty as a physician to confirm the positivity with CDC,” she said.
Dr. Yun concurred. “The most important message is, do something,” she said. “Don’t just assume it’s a false positive.”
Diagnosis is not simple and requires testing beyond the initial ELISA assay used in blood-bank screening. Confirmatory tests must be carried out in coordination with the Centers for Disease Control and Prevention. Also, with no agents approved by the Food and Drug Administration to treat Chagas, treatment is available only through the CDC’s investigational drugs protocol. Both drugs used in Chagas, benznidazole and nifurtimox, come with serious adverse effects that must be closely monitored.
“It’s time consuming, filling out the forms, getting the consent, tracking and sending back lab results to CDC in order to get drugs – it’s not like you can just write a prescription,” Dr. Meymandi said. But, “if you don’t know how to treat the patient or don’t have time, find someone like me,” she noted, adding that she is available to counsel any physician daunted by a potential Chagas case.
Treatment options
No formal clinical algorithm exists for Chagas, but Dr. Meymandi, Dr. Yun, and Dr. Woc-Colburn all pointed to a 2007 JAMA article, which describes diagnosis and treatment protocols, as an important reference for clinicians to start with. It’s “the best approximation of a clinical guideline we have,” Dr. Yun said (JAMA. 2007;298[18]:2171-81. doi:10.1001/jama.298.18.2171).
Dr. Meymandi, who has treated more Chagas patients than has any other U.S. clinician, said that treatment has changed somewhat since the JAMA article was published. In 2007, she said, nifurtimox was the main drug available through CDC, while benznidazole, which is somewhat better tolerated and has shorter treatment duration, has since become the first-line agent.
“We’ve lowered the dose of benznidazole, maxing out at 400 mg/day to decrease the toxicity,” she said. Also, treatment is now being extended to some patients aged 60 years and older.
The decision to treat or not treat, clinicians say, depends on the patient’s age, disease progression, comorbidities and potential serious drug interactions, and willingness to tolerate side effects that, with nifurtimox especially, can include skin sloughing, rash, and psychological and neurologic symptoms including depression and peripheral neuropathy.
“If you don’t have side effects, you’re not taking the drugs,” Dr. Meymandi said. Dr. Woc-Colburn noted that polypharmacy was a major consideration when treating older adults for Chagas. “If I have a patient who has diabetes, obesity, [and] end-stage renal disease, it’s not going to be ideal to give [benznidazole].”
Recent, highly anticipated results from BENEFIT, a large randomized trial (n = 2,854) showed that benznidazole reduced parasite load but was not helpful in halting cardiac damage at 5 years’ follow-up in patients with established Chagas cardiomyopathy (N Engl J Med. 2015 Oct;373:1295-306. doi:10.1056/NEJMoa1507574).
Dr. Meymandi, whose earlier research established that Chagas cardiomyopathy carries significantly higher morbidity and mortality than does non–Chagas cardiomyopathy (Circulation. 2012;126:A18171), said that the BENEFIT results underscore the need for physicians to be bullish in their approach to treating Chagas soon after diagnosis.
“It doesn’t matter if they’re symptomatic or asymptomatic. You can’t wait till they progress to treat. If you wait for the progression of disease you’ve lost the battle. You can’t wait and follow conservatively until you see the complications, because once those complications have started the parasitic load is too high for you to have an impact,” she said.
Dr. Yun said that given the toxicity of current treatment, she hoped to see more studies show clearer evidence of clinical benefit, “either reductions in mortality or reductions in end organ disease.” Most studies “have focused on clearance of parasite, which is important, but it’s not as important decreasing the risk of death or cardiomyopathy or heart failure.”
Rick Tarleton, Ph.D., a biologist the University of Georgia, in Athens, who has worked on Chagas for more than 30 years, said that because Chagas pathology is directly tied to parasite load – and not, as people have suggested in the past, an autoimmune reaction resulting from parasite exposure – drug treatment may prove to be worthwhile even in patients with significant cardiac involvement.
“You get rid of the parasite, you get rid of the progression of the disease,” Dr. Tarleton said. Even the findings from the BENEFIT trial, he said, did not lead him to conclude that treatment in people with established cardiac disease was futile.
“If you’re treating people who are already chronically infected and showing symptoms, the question is not have you reversed the damage, it’s have you stopped accumulating damage,” he noted. “And a 5-year follow-up is probably not long enough to know whether you’ve stopped accumulating.”
“We have drugs, they’re not great, they do have side effects, they don’t always work,” Dr. Tarleton said. “But they’re better than nothing. And they ought to be more widely used.”
Dr. Meymandi said that current supplies of benznidazole at CDC are low and that a dozen patients at her clinic are awaiting treatment. Meanwhile, access may soon be complicated further by the announcement, this month, that KaloBios Pharmaceuticals had bought the rights to seek FDA approval of benznidazole and market it in the United States.
The same company’s CEO came under fire in recent months for acquiring rights to an inexpensive drug to treat toxoplasmosis in AIDS patients, then announcing a price increase from $13.50 to $750 a pill.
“Everyone’s really concerned,” Dr. Meymandi said, “because Chagas is a disease of the poor.”
Pregnancy did not increase Hodgkin lymphoma relapse rate
Women who become pregnant while in remission from Hodgkin lymphoma were not at increased risk for cancer relapse, according to an analysis of data from Swedish health care registries combined with medical records.
Of 449 women who were diagnosed with Hodgkin lymphoma between 1992 and 2009, 144 (32%) became pregnant during follow-up, which started 6 months after diagnosis, when the disease was assumed to be in remission. Only one of these women experienced a pregnancy-associated relapse, which was defined as a relapse occurring during pregnancy or within 5 years of delivery. Of the women who did not become pregnant, 46 had a relapse.
The effect of pregnancy on relapse has been a concern of patients and clinicians, but “our findings suggest that the risk of pregnancy-associated relapse does not need to be taken into account in family planning for women whose Hodgkin lymphoma is in remission,” said Caroline E. Weibull of Karolinska Institutet in Stockholm, and her associates.
The researchers used the nationwide “Swedish Cancer Register” to identify all cases of Hodgkin lymphoma (reporting is mandatory) and merged this data with clinical information from other registries and medical records.
The pregnancy rates were similar among women who had limited- and advanced-stage disease and among women with and without B symptoms at diagnosis – a finding that negates consideration of a so-called “healthy mother effect” in protecting against relapse, they wrote (J Clin Onc. 2015 Dec. 14 [doi:10.1200/JCO.2015.63.3446]).
The researchers also found that the absolute risk for relapse was highest in the first 2-3 years after diagnosis, which suggests that women should be advised, “if possible, to wait 2 years after cessation of treatment before becoming pregnant.” Additionally, the relapse rate more than doubled in women aged 30 years or older at diagnosis, compared with women aged 18-24 years at diagnosis – a finding consistent with previous research, they noted.
Women in the study were aged 18-40 at diagnosis. Follow-up ended on the date of relapse, the date of death, or at the end of 2010, whichever came first.
Women who become pregnant while in remission from Hodgkin lymphoma were not at increased risk for cancer relapse, according to an analysis of data from Swedish health care registries combined with medical records.
Of 449 women who were diagnosed with Hodgkin lymphoma between 1992 and 2009, 144 (32%) became pregnant during follow-up, which started 6 months after diagnosis, when the disease was assumed to be in remission. Only one of these women experienced a pregnancy-associated relapse, which was defined as a relapse occurring during pregnancy or within 5 years of delivery. Of the women who did not become pregnant, 46 had a relapse.
The effect of pregnancy on relapse has been a concern of patients and clinicians, but “our findings suggest that the risk of pregnancy-associated relapse does not need to be taken into account in family planning for women whose Hodgkin lymphoma is in remission,” said Caroline E. Weibull of Karolinska Institutet in Stockholm, and her associates.
The researchers used the nationwide “Swedish Cancer Register” to identify all cases of Hodgkin lymphoma (reporting is mandatory) and merged this data with clinical information from other registries and medical records.
The pregnancy rates were similar among women who had limited- and advanced-stage disease and among women with and without B symptoms at diagnosis – a finding that negates consideration of a so-called “healthy mother effect” in protecting against relapse, they wrote (J Clin Onc. 2015 Dec. 14 [doi:10.1200/JCO.2015.63.3446]).
The researchers also found that the absolute risk for relapse was highest in the first 2-3 years after diagnosis, which suggests that women should be advised, “if possible, to wait 2 years after cessation of treatment before becoming pregnant.” Additionally, the relapse rate more than doubled in women aged 30 years or older at diagnosis, compared with women aged 18-24 years at diagnosis – a finding consistent with previous research, they noted.
Women in the study were aged 18-40 at diagnosis. Follow-up ended on the date of relapse, the date of death, or at the end of 2010, whichever came first.
Women who become pregnant while in remission from Hodgkin lymphoma were not at increased risk for cancer relapse, according to an analysis of data from Swedish health care registries combined with medical records.
Of 449 women who were diagnosed with Hodgkin lymphoma between 1992 and 2009, 144 (32%) became pregnant during follow-up, which started 6 months after diagnosis, when the disease was assumed to be in remission. Only one of these women experienced a pregnancy-associated relapse, which was defined as a relapse occurring during pregnancy or within 5 years of delivery. Of the women who did not become pregnant, 46 had a relapse.
The effect of pregnancy on relapse has been a concern of patients and clinicians, but “our findings suggest that the risk of pregnancy-associated relapse does not need to be taken into account in family planning for women whose Hodgkin lymphoma is in remission,” said Caroline E. Weibull of Karolinska Institutet in Stockholm, and her associates.
The researchers used the nationwide “Swedish Cancer Register” to identify all cases of Hodgkin lymphoma (reporting is mandatory) and merged this data with clinical information from other registries and medical records.
The pregnancy rates were similar among women who had limited- and advanced-stage disease and among women with and without B symptoms at diagnosis – a finding that negates consideration of a so-called “healthy mother effect” in protecting against relapse, they wrote (J Clin Onc. 2015 Dec. 14 [doi:10.1200/JCO.2015.63.3446]).
The researchers also found that the absolute risk for relapse was highest in the first 2-3 years after diagnosis, which suggests that women should be advised, “if possible, to wait 2 years after cessation of treatment before becoming pregnant.” Additionally, the relapse rate more than doubled in women aged 30 years or older at diagnosis, compared with women aged 18-24 years at diagnosis – a finding consistent with previous research, they noted.
Women in the study were aged 18-40 at diagnosis. Follow-up ended on the date of relapse, the date of death, or at the end of 2010, whichever came first.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Pregnancy did not increase the risk of relapse of Hodgkin lymphoma in a population-based study.
Major finding: Of 144 women who became pregnant 6 months or longer after diagnosis of Hodgkin lymphoma, 1 experienced a pregnancy-associated relapse.
Data source: Population-based study utilizing Swedish health care registries and medical records, in which 449 women with Hodgkin lymphoma diagnoses, and 47 relapses, were identified.
Disclosures: The study was supported by the Swedish Cancer Society, the Strategic Research Program in Epidemiology at Karolinska Institutet, the Swedish Society for Medicine, and the Swedish Society for Medical Research.
Ophthalmic drugs in pregnancy and lactation
A number of drugs are available for ophthalmic use. This review focuses on single drug products and, although combination drug products are not discussed, these formulations typically include the drugs reviewed here.
Since it appears that ophthalmic medications are commonly used for a wide range of conditions and ages, one would expect to see numerous reports of their use in the eyes of pregnant or breastfeeding patients. Unfortunately, the opposite is true. The majority of the drugs have no human pregnancy or lactation data. When there are human data, it invariably involves the systemic use of the drug for other indications, rather than its ophthalmic use. Moreover, the animal reproduction data are usually not relevant because they involve systemic drugs (e.g., IV or oral). Consequently, determining the level of risk an ophthalmic drug presents to an embryo and/or fetus is primarily based on time and dose, the two cardinal principles of teratology.
Avoiding exposure during organogenesis – the period when a drug can cause developmental toxicity (altered growth, structural anomalies, functional and/or behavioral deficits, or death) – is usually best, but may not be possible in some cases, including glaucoma, eye infections, and eye surgery. Fortunately, the systemic concentrations of drugs applied topically to the eye are typically thought to be low, even though the levels of most drugs have not been studied. Thus, the risk to the embryo and/or fetus in most cases can be considered low and the drug classified as compatible in pregnancy and breastfeeding.
If a topical eye drug must be used during pregnancy or lactation, teach the patient how to decrease the amount of drug reaching the systemic circulation. This involves placing pressure over the tear duct in the corner of the eye for 1 minute or more, then removing any excess solution with absorbent tissue.
In the sections below, drugs are shown by indication or by pharmacologic class. The term “human eye data” refers to the use of the drug in pregnancy and/or lactation.
Glaucoma
If you are caring for a pregnant patient who is being treated for glaucoma, two recent reviews may be helpful: Surv Ophthalmol. 2011 Jul-Aug;56(4):324-35 and Curr Opin Ophthalmol. 2014 Mar;25(2):93-7.
Sympathomimetics (alpha-adrenergic agonists) include apraclonidine (Iopidine), which has no human eye data, and brimonidine (Alphagan P), which has one case report in pregnancy and breastfeeding showing no fetal or nursing infant harm.
Four of the five beta-adrenergic blockers have no human eye data: betaxolol (Betoptic), carteolol (Ocupress), levobunolol (Betagan), and metipranolol, but there are two case reports for timolol (Betimol, Istalol, Timoptic, Timoptic-XE) showing no fetal harm in one newborn and growth restriction in the other.
Among the miotics, there are limited human eye data for pilocarpine (Isopto Carpine) and no fetal harm was observed. For echothiophate iodide (Phospholine Iodide), there is one case report of a normal full-term infant whose mother was treated with the agent up to 32 weeks’ gestation and then with pilocarpine for 8 weeks (Arch Ophthalmol. 1968 Mar;79[3]:283-5).
Carbonic anhydrase inhibitors include brinzolamide (Azopt), which has no human eye data, and dorzolamide (Trusopt), which has growth restriction in one case treated with fixed combination of dorzolamide and timolol. There are no human eye data for unoprostone isopropyl (Rescula), a synthetic docosanoid.
Of the four prostaglandin analogs, three have no human eye data, bimatoprost (Lumigan), tafluprost (Zioptan), and travoprost (Travatan Z). There were 11 pregnancies exposed to latanoprost (Xalatan). The outcomes of these cases were one lost to follow-up, one miscarriage, and nine infants without congenital anomalies (Am J Ophthalmol. 2004 Aug;138[2]:305-6).
Mitomycin (Mitosol) is an antimetabolite that is given topically to the surgical site of glaucoma filtration surgery. No reports of its use in pregnant humans have been located. According to the manufacturer, clinically significant systemic concentrations are not expected.
Antiseptics
Povidone-iodine (Betadine) is indicated for prepping of the periocular region. There does not appear to be any risk to the embryo-fetus or nursing infant from this indication.
Antihistamines
The four ophthalmic agents in this class are alcaftadine (Lastacaft), azelastine (Optivar), emedastine (Emadine), and epinastine (Elestat). There are no human eye data for these agents but, like antihistamines given systemically, they are probably compatible in pregnancy and lactation.
Antihistamine-mast cell stabilizers
There are no human eye data for bepotastine (Bepreve), ketotifen (Alaway), and olopatadine (Pataday, Patanol). Peak plasma concentrations of bepotastine were 5.1-7.3 ng/mL for 1-2 hours after instillation and were less than 2 ng/mL at 24 hours. It appears that these drugs can also be classified as compatible in pregnancy and lactation.
Anti-infectives
Ten anti-infectives are available for topical treatment of eye infections (systemic concentrations if known): besifloxacin (Besivance) (0.43 ng/mL), ciprofloxacin (less than 2.5 ng/mL), gentamicin, gatifloxacin (Zymaxid) (less than 5 ng/mL), levofloxacin (Iquix) (10.9 ng/mL), moxifloxacin (Vigamox) (2.7 ng/mL), ofloxacin (1.9 ng/mL), sulfacetamide (Isopto Cetamide), and tobramycin. The tenth agent, natamycin (Natacyn), is an antifungal. According to the manufacturer, systemic absorption is not expected.
None of these agents have human eye data, but they are usually considered compatible in pregnancy and breastfeeding when systemic formulations are used, so they should be compatible when used in the eye.
Antivirals
Ganciclovir (Zirgan) has no human eye data but, according to the manufacturer, the daily ophthalmic dose is about 0.04% and 0.1% of the oral and IV doses, respectively. Thus, minimal systemic exposure is expected. Trifluridine (Viroptic) also has no eye human data. As reported in the product information, detectable blood concentrations of the drug were not found in healthy normal subjects indicating that systemic absorption was negligible.
Corticosteroids
Among the nine corticosteroid products, six are suspensions or ointments, one is an injection, and two are implants. In most nonpregnant patients receiving the dexamethasone (Ozurdex) intravitreal implant, plasma dexamethasone concentrations were undetectable (less than 50 pg/mL) but, in some, ranged from 52 pg/mL to 102 pg/mL. There is only one case report describing the use of topical dexamethasone suspension (Maxidex) in pregnancy. In that case, dexamethasone and clindamycin were given in the first and second trimesters and the woman eventually gave birth to a normal full term infant (Int Ophthalmol. 1998-1999;22[2]:85-8).
For the fluocinolone (Retisert) ocular implant, systemic absorption of detectable amounts of the drug have not been observed. In a second report, a patient who had type 1 diabetes and was 6 months pregnant received a 2-mg intravitreal injection of triamcinolone (Triesence) in both eyes. No adverse effects in the mother or fetus were noted (Clin Ophthalmol. 2011;5:439-41).
There are no human eye data for five topical corticosteroids: difluprednate (Durezol), fluorometholone (Flarex, Fluor-OP, FML), loteprednol (Alrex, Lotemax), prednisolone (Econopred), and rimexolone (Vexol). Only two of the five drugs had information about systemic absorption. For difluprednate, blood levels were below the quantification limit (50 ng/mL). Extremely low levels were detected after the use of rimexolone with a mean serum concentration of 130 pg/mL (range less than 80-470 pg/mL).
Cycloplegics-mydriatics
This class includes four anticholinergic agents: atropine, cyclopentolate (Cyclogyl), homatropine, and tropicamide (Mydriacyl). Systemic absorption has not been studied for these drugs and there are no reports of their use in pregnancy or lactation.
Cystine-depleting agents
There are no reports describing the use of cysteamine (Cystaran) in human pregnancy or lactation. Since the drug is given as one drop in each eye every waking hour, transfer to the systemic circulation should be expected, but the amount, if it occurs, has not been reported.
Immunologics
There are no reports on the use of cyclosporine (Restasis) eye drops in human pregnancy or during breastfeeding. However, data for the systemic use of the drug during these conditions has shown it to be low risk. After long term ophthalmic use, blood concentrations of the drug were below the quantitation limit of 0.1 ng/mL.
Local anesthetics
The three drugs in this class are lidocaine, proparacaine (Alcaine), and tetracaine (Altacaine, Tetravisc). There is no information regarding the use of these agents in pregnancy or lactation. Since they are used for brief periods, the risk to an embryo or nursing infant appears to be nil.
Mast cell stabilizers
There are three drugs in this class that can be used topically in the eye: cromolyn, lodoxamide (Alomide), and nedocromil (Alocril). There are no human eye data for these agents. Systemic concentrations of lodoxamide were below the detection limit of 2.5 ng/mL.
Miotics
The two miotics are acetylcholine (Miochol E) and carbachol (Carbastat, Miostat). Both are used immediately before eye surgery. The amount, if any, in the systemic circulation is unknown. No reports of human eye use in pregnancy or lactation have been found.
NSAIDs
The five drugs in this class are bromfenac (Xibrom), diclofenac (Voltaren), flurbiprofen (Ocufen), ketorolac, and nepafenac (Nevanac). Although it has not been studied, the estimated plasma level for bromfenac is less than 50 ng/mL. For diclofenac, the plasma concentration was below the limit of quantification (10 ng/mL). In a study conducted with ketorolac, only 5 of 26 subjects had detectable plasma concentrations (10.7 to 22.5 ng/mL). These levels were about 2% of the peak plasma levels after oral dosing. For nepafenac, the peak plasma concentrations of the parent drug and active metabolite were 0.3 and 0.4 ng/mL, respectively. These data suggest that the risk to the embryo-fetus or a nursing infant are nil.
Photodynamic therapy
Verteporfin (Visudyne) is given intravenously. Three reports have described its use in three pregnancies. In all cases, the infants were healthy at birth and had normal growth (Acta Ophthalmol Scand. 2004 Oct;82[5]:623-4, Eye [Lond]. 2009 Jun;23[6]:1479, and Aust N Z J Obstet Gynaecol. 2009 Apr;49[2]:236-7).
Proteolytic enzymes
No human eye data were found for ocriplasmin (Jetrea), an agent given as an intravitreal injection. Although it was not studied, detectable levels of the drug in the systemic circulation are not expected, according to the manufacturer.
Selective vascular endothelial growth factor antagonists
There are three agents in this class: aflibercept (Eylea), pegaptanib (Macugen), and ranibizumab (Lucentis). There are no human eye data for these drugs. All are given as intravitreal injection. The systemic concentrations of the three drugs were a mean 0.02 mcg/mL; 80 ng/mL (after a dose of 10 times the recommended dose); and an estimated minimum 0.22 ng/mL, respectively. These data suggest that the risk to the embryo-fetus or a nursing infant is nil.
Sympathomimetics (decongestants)
There are four ophthalmic agents in this class and none have human eye data. Naphazoline (Naphcon, Vasocon) is the only one of the four that requires a prescription. No reports describing the systemic absorption, if any, have been found. The other three agents are available over the counter. They are oxymetazoline (Visine L.R.), phenylephrine, and tetrahydrozoline (Opti-Clear). Although the amount reaching the systemic circulation was not provided by the manufacturers for these three agents, a caution was placed on phenylephrine stating that systemic absorption, although rare, might cause alpha-adrenergic effects, such as a rise in blood pressure and reflex atropine-sensitive bradycardia.
The risk to the embryo-fetus or nursing infant from ophthalmic drugs appears to be low, with the possible exception of phenylephrine. Nevertheless, if any of these agents are used in pregnancy or during breastfeeding, careful assessment of the embryo-fetus and nursing infant should be conducted. Moreover, research on the potential effects of ophthalmic drugs on the embryo-fetus and nursing infant are desperately needed.
Mr. Briggs is clinical professor of pharmacy at the University of California, San Francisco; and adjunct professor of pharmacy at the University of Southern California, Los Angeles, and Washington State University, Spokane. He is coauthor of “Drugs in Pregnancy and Lactation,” and coeditor of “Diseases, Complications, and Drug Therapy in Obstetrics.” He has no relevant financial disclosures.
A number of drugs are available for ophthalmic use. This review focuses on single drug products and, although combination drug products are not discussed, these formulations typically include the drugs reviewed here.
Since it appears that ophthalmic medications are commonly used for a wide range of conditions and ages, one would expect to see numerous reports of their use in the eyes of pregnant or breastfeeding patients. Unfortunately, the opposite is true. The majority of the drugs have no human pregnancy or lactation data. When there are human data, it invariably involves the systemic use of the drug for other indications, rather than its ophthalmic use. Moreover, the animal reproduction data are usually not relevant because they involve systemic drugs (e.g., IV or oral). Consequently, determining the level of risk an ophthalmic drug presents to an embryo and/or fetus is primarily based on time and dose, the two cardinal principles of teratology.
Avoiding exposure during organogenesis – the period when a drug can cause developmental toxicity (altered growth, structural anomalies, functional and/or behavioral deficits, or death) – is usually best, but may not be possible in some cases, including glaucoma, eye infections, and eye surgery. Fortunately, the systemic concentrations of drugs applied topically to the eye are typically thought to be low, even though the levels of most drugs have not been studied. Thus, the risk to the embryo and/or fetus in most cases can be considered low and the drug classified as compatible in pregnancy and breastfeeding.
If a topical eye drug must be used during pregnancy or lactation, teach the patient how to decrease the amount of drug reaching the systemic circulation. This involves placing pressure over the tear duct in the corner of the eye for 1 minute or more, then removing any excess solution with absorbent tissue.
In the sections below, drugs are shown by indication or by pharmacologic class. The term “human eye data” refers to the use of the drug in pregnancy and/or lactation.
Glaucoma
If you are caring for a pregnant patient who is being treated for glaucoma, two recent reviews may be helpful: Surv Ophthalmol. 2011 Jul-Aug;56(4):324-35 and Curr Opin Ophthalmol. 2014 Mar;25(2):93-7.
Sympathomimetics (alpha-adrenergic agonists) include apraclonidine (Iopidine), which has no human eye data, and brimonidine (Alphagan P), which has one case report in pregnancy and breastfeeding showing no fetal or nursing infant harm.
Four of the five beta-adrenergic blockers have no human eye data: betaxolol (Betoptic), carteolol (Ocupress), levobunolol (Betagan), and metipranolol, but there are two case reports for timolol (Betimol, Istalol, Timoptic, Timoptic-XE) showing no fetal harm in one newborn and growth restriction in the other.
Among the miotics, there are limited human eye data for pilocarpine (Isopto Carpine) and no fetal harm was observed. For echothiophate iodide (Phospholine Iodide), there is one case report of a normal full-term infant whose mother was treated with the agent up to 32 weeks’ gestation and then with pilocarpine for 8 weeks (Arch Ophthalmol. 1968 Mar;79[3]:283-5).
Carbonic anhydrase inhibitors include brinzolamide (Azopt), which has no human eye data, and dorzolamide (Trusopt), which has growth restriction in one case treated with fixed combination of dorzolamide and timolol. There are no human eye data for unoprostone isopropyl (Rescula), a synthetic docosanoid.
Of the four prostaglandin analogs, three have no human eye data, bimatoprost (Lumigan), tafluprost (Zioptan), and travoprost (Travatan Z). There were 11 pregnancies exposed to latanoprost (Xalatan). The outcomes of these cases were one lost to follow-up, one miscarriage, and nine infants without congenital anomalies (Am J Ophthalmol. 2004 Aug;138[2]:305-6).
Mitomycin (Mitosol) is an antimetabolite that is given topically to the surgical site of glaucoma filtration surgery. No reports of its use in pregnant humans have been located. According to the manufacturer, clinically significant systemic concentrations are not expected.
Antiseptics
Povidone-iodine (Betadine) is indicated for prepping of the periocular region. There does not appear to be any risk to the embryo-fetus or nursing infant from this indication.
Antihistamines
The four ophthalmic agents in this class are alcaftadine (Lastacaft), azelastine (Optivar), emedastine (Emadine), and epinastine (Elestat). There are no human eye data for these agents but, like antihistamines given systemically, they are probably compatible in pregnancy and lactation.
Antihistamine-mast cell stabilizers
There are no human eye data for bepotastine (Bepreve), ketotifen (Alaway), and olopatadine (Pataday, Patanol). Peak plasma concentrations of bepotastine were 5.1-7.3 ng/mL for 1-2 hours after instillation and were less than 2 ng/mL at 24 hours. It appears that these drugs can also be classified as compatible in pregnancy and lactation.
Anti-infectives
Ten anti-infectives are available for topical treatment of eye infections (systemic concentrations if known): besifloxacin (Besivance) (0.43 ng/mL), ciprofloxacin (less than 2.5 ng/mL), gentamicin, gatifloxacin (Zymaxid) (less than 5 ng/mL), levofloxacin (Iquix) (10.9 ng/mL), moxifloxacin (Vigamox) (2.7 ng/mL), ofloxacin (1.9 ng/mL), sulfacetamide (Isopto Cetamide), and tobramycin. The tenth agent, natamycin (Natacyn), is an antifungal. According to the manufacturer, systemic absorption is not expected.
None of these agents have human eye data, but they are usually considered compatible in pregnancy and breastfeeding when systemic formulations are used, so they should be compatible when used in the eye.
Antivirals
Ganciclovir (Zirgan) has no human eye data but, according to the manufacturer, the daily ophthalmic dose is about 0.04% and 0.1% of the oral and IV doses, respectively. Thus, minimal systemic exposure is expected. Trifluridine (Viroptic) also has no eye human data. As reported in the product information, detectable blood concentrations of the drug were not found in healthy normal subjects indicating that systemic absorption was negligible.
Corticosteroids
Among the nine corticosteroid products, six are suspensions or ointments, one is an injection, and two are implants. In most nonpregnant patients receiving the dexamethasone (Ozurdex) intravitreal implant, plasma dexamethasone concentrations were undetectable (less than 50 pg/mL) but, in some, ranged from 52 pg/mL to 102 pg/mL. There is only one case report describing the use of topical dexamethasone suspension (Maxidex) in pregnancy. In that case, dexamethasone and clindamycin were given in the first and second trimesters and the woman eventually gave birth to a normal full term infant (Int Ophthalmol. 1998-1999;22[2]:85-8).
For the fluocinolone (Retisert) ocular implant, systemic absorption of detectable amounts of the drug have not been observed. In a second report, a patient who had type 1 diabetes and was 6 months pregnant received a 2-mg intravitreal injection of triamcinolone (Triesence) in both eyes. No adverse effects in the mother or fetus were noted (Clin Ophthalmol. 2011;5:439-41).
There are no human eye data for five topical corticosteroids: difluprednate (Durezol), fluorometholone (Flarex, Fluor-OP, FML), loteprednol (Alrex, Lotemax), prednisolone (Econopred), and rimexolone (Vexol). Only two of the five drugs had information about systemic absorption. For difluprednate, blood levels were below the quantification limit (50 ng/mL). Extremely low levels were detected after the use of rimexolone with a mean serum concentration of 130 pg/mL (range less than 80-470 pg/mL).
Cycloplegics-mydriatics
This class includes four anticholinergic agents: atropine, cyclopentolate (Cyclogyl), homatropine, and tropicamide (Mydriacyl). Systemic absorption has not been studied for these drugs and there are no reports of their use in pregnancy or lactation.
Cystine-depleting agents
There are no reports describing the use of cysteamine (Cystaran) in human pregnancy or lactation. Since the drug is given as one drop in each eye every waking hour, transfer to the systemic circulation should be expected, but the amount, if it occurs, has not been reported.
Immunologics
There are no reports on the use of cyclosporine (Restasis) eye drops in human pregnancy or during breastfeeding. However, data for the systemic use of the drug during these conditions has shown it to be low risk. After long term ophthalmic use, blood concentrations of the drug were below the quantitation limit of 0.1 ng/mL.
Local anesthetics
The three drugs in this class are lidocaine, proparacaine (Alcaine), and tetracaine (Altacaine, Tetravisc). There is no information regarding the use of these agents in pregnancy or lactation. Since they are used for brief periods, the risk to an embryo or nursing infant appears to be nil.
Mast cell stabilizers
There are three drugs in this class that can be used topically in the eye: cromolyn, lodoxamide (Alomide), and nedocromil (Alocril). There are no human eye data for these agents. Systemic concentrations of lodoxamide were below the detection limit of 2.5 ng/mL.
Miotics
The two miotics are acetylcholine (Miochol E) and carbachol (Carbastat, Miostat). Both are used immediately before eye surgery. The amount, if any, in the systemic circulation is unknown. No reports of human eye use in pregnancy or lactation have been found.
NSAIDs
The five drugs in this class are bromfenac (Xibrom), diclofenac (Voltaren), flurbiprofen (Ocufen), ketorolac, and nepafenac (Nevanac). Although it has not been studied, the estimated plasma level for bromfenac is less than 50 ng/mL. For diclofenac, the plasma concentration was below the limit of quantification (10 ng/mL). In a study conducted with ketorolac, only 5 of 26 subjects had detectable plasma concentrations (10.7 to 22.5 ng/mL). These levels were about 2% of the peak plasma levels after oral dosing. For nepafenac, the peak plasma concentrations of the parent drug and active metabolite were 0.3 and 0.4 ng/mL, respectively. These data suggest that the risk to the embryo-fetus or a nursing infant are nil.
Photodynamic therapy
Verteporfin (Visudyne) is given intravenously. Three reports have described its use in three pregnancies. In all cases, the infants were healthy at birth and had normal growth (Acta Ophthalmol Scand. 2004 Oct;82[5]:623-4, Eye [Lond]. 2009 Jun;23[6]:1479, and Aust N Z J Obstet Gynaecol. 2009 Apr;49[2]:236-7).
Proteolytic enzymes
No human eye data were found for ocriplasmin (Jetrea), an agent given as an intravitreal injection. Although it was not studied, detectable levels of the drug in the systemic circulation are not expected, according to the manufacturer.
Selective vascular endothelial growth factor antagonists
There are three agents in this class: aflibercept (Eylea), pegaptanib (Macugen), and ranibizumab (Lucentis). There are no human eye data for these drugs. All are given as intravitreal injection. The systemic concentrations of the three drugs were a mean 0.02 mcg/mL; 80 ng/mL (after a dose of 10 times the recommended dose); and an estimated minimum 0.22 ng/mL, respectively. These data suggest that the risk to the embryo-fetus or a nursing infant is nil.
Sympathomimetics (decongestants)
There are four ophthalmic agents in this class and none have human eye data. Naphazoline (Naphcon, Vasocon) is the only one of the four that requires a prescription. No reports describing the systemic absorption, if any, have been found. The other three agents are available over the counter. They are oxymetazoline (Visine L.R.), phenylephrine, and tetrahydrozoline (Opti-Clear). Although the amount reaching the systemic circulation was not provided by the manufacturers for these three agents, a caution was placed on phenylephrine stating that systemic absorption, although rare, might cause alpha-adrenergic effects, such as a rise in blood pressure and reflex atropine-sensitive bradycardia.
The risk to the embryo-fetus or nursing infant from ophthalmic drugs appears to be low, with the possible exception of phenylephrine. Nevertheless, if any of these agents are used in pregnancy or during breastfeeding, careful assessment of the embryo-fetus and nursing infant should be conducted. Moreover, research on the potential effects of ophthalmic drugs on the embryo-fetus and nursing infant are desperately needed.
Mr. Briggs is clinical professor of pharmacy at the University of California, San Francisco; and adjunct professor of pharmacy at the University of Southern California, Los Angeles, and Washington State University, Spokane. He is coauthor of “Drugs in Pregnancy and Lactation,” and coeditor of “Diseases, Complications, and Drug Therapy in Obstetrics.” He has no relevant financial disclosures.
A number of drugs are available for ophthalmic use. This review focuses on single drug products and, although combination drug products are not discussed, these formulations typically include the drugs reviewed here.
Since it appears that ophthalmic medications are commonly used for a wide range of conditions and ages, one would expect to see numerous reports of their use in the eyes of pregnant or breastfeeding patients. Unfortunately, the opposite is true. The majority of the drugs have no human pregnancy or lactation data. When there are human data, it invariably involves the systemic use of the drug for other indications, rather than its ophthalmic use. Moreover, the animal reproduction data are usually not relevant because they involve systemic drugs (e.g., IV or oral). Consequently, determining the level of risk an ophthalmic drug presents to an embryo and/or fetus is primarily based on time and dose, the two cardinal principles of teratology.
Avoiding exposure during organogenesis – the period when a drug can cause developmental toxicity (altered growth, structural anomalies, functional and/or behavioral deficits, or death) – is usually best, but may not be possible in some cases, including glaucoma, eye infections, and eye surgery. Fortunately, the systemic concentrations of drugs applied topically to the eye are typically thought to be low, even though the levels of most drugs have not been studied. Thus, the risk to the embryo and/or fetus in most cases can be considered low and the drug classified as compatible in pregnancy and breastfeeding.
If a topical eye drug must be used during pregnancy or lactation, teach the patient how to decrease the amount of drug reaching the systemic circulation. This involves placing pressure over the tear duct in the corner of the eye for 1 minute or more, then removing any excess solution with absorbent tissue.
In the sections below, drugs are shown by indication or by pharmacologic class. The term “human eye data” refers to the use of the drug in pregnancy and/or lactation.
Glaucoma
If you are caring for a pregnant patient who is being treated for glaucoma, two recent reviews may be helpful: Surv Ophthalmol. 2011 Jul-Aug;56(4):324-35 and Curr Opin Ophthalmol. 2014 Mar;25(2):93-7.
Sympathomimetics (alpha-adrenergic agonists) include apraclonidine (Iopidine), which has no human eye data, and brimonidine (Alphagan P), which has one case report in pregnancy and breastfeeding showing no fetal or nursing infant harm.
Four of the five beta-adrenergic blockers have no human eye data: betaxolol (Betoptic), carteolol (Ocupress), levobunolol (Betagan), and metipranolol, but there are two case reports for timolol (Betimol, Istalol, Timoptic, Timoptic-XE) showing no fetal harm in one newborn and growth restriction in the other.
Among the miotics, there are limited human eye data for pilocarpine (Isopto Carpine) and no fetal harm was observed. For echothiophate iodide (Phospholine Iodide), there is one case report of a normal full-term infant whose mother was treated with the agent up to 32 weeks’ gestation and then with pilocarpine for 8 weeks (Arch Ophthalmol. 1968 Mar;79[3]:283-5).
Carbonic anhydrase inhibitors include brinzolamide (Azopt), which has no human eye data, and dorzolamide (Trusopt), which has growth restriction in one case treated with fixed combination of dorzolamide and timolol. There are no human eye data for unoprostone isopropyl (Rescula), a synthetic docosanoid.
Of the four prostaglandin analogs, three have no human eye data, bimatoprost (Lumigan), tafluprost (Zioptan), and travoprost (Travatan Z). There were 11 pregnancies exposed to latanoprost (Xalatan). The outcomes of these cases were one lost to follow-up, one miscarriage, and nine infants without congenital anomalies (Am J Ophthalmol. 2004 Aug;138[2]:305-6).
Mitomycin (Mitosol) is an antimetabolite that is given topically to the surgical site of glaucoma filtration surgery. No reports of its use in pregnant humans have been located. According to the manufacturer, clinically significant systemic concentrations are not expected.
Antiseptics
Povidone-iodine (Betadine) is indicated for prepping of the periocular region. There does not appear to be any risk to the embryo-fetus or nursing infant from this indication.
Antihistamines
The four ophthalmic agents in this class are alcaftadine (Lastacaft), azelastine (Optivar), emedastine (Emadine), and epinastine (Elestat). There are no human eye data for these agents but, like antihistamines given systemically, they are probably compatible in pregnancy and lactation.
Antihistamine-mast cell stabilizers
There are no human eye data for bepotastine (Bepreve), ketotifen (Alaway), and olopatadine (Pataday, Patanol). Peak plasma concentrations of bepotastine were 5.1-7.3 ng/mL for 1-2 hours after instillation and were less than 2 ng/mL at 24 hours. It appears that these drugs can also be classified as compatible in pregnancy and lactation.
Anti-infectives
Ten anti-infectives are available for topical treatment of eye infections (systemic concentrations if known): besifloxacin (Besivance) (0.43 ng/mL), ciprofloxacin (less than 2.5 ng/mL), gentamicin, gatifloxacin (Zymaxid) (less than 5 ng/mL), levofloxacin (Iquix) (10.9 ng/mL), moxifloxacin (Vigamox) (2.7 ng/mL), ofloxacin (1.9 ng/mL), sulfacetamide (Isopto Cetamide), and tobramycin. The tenth agent, natamycin (Natacyn), is an antifungal. According to the manufacturer, systemic absorption is not expected.
None of these agents have human eye data, but they are usually considered compatible in pregnancy and breastfeeding when systemic formulations are used, so they should be compatible when used in the eye.
Antivirals
Ganciclovir (Zirgan) has no human eye data but, according to the manufacturer, the daily ophthalmic dose is about 0.04% and 0.1% of the oral and IV doses, respectively. Thus, minimal systemic exposure is expected. Trifluridine (Viroptic) also has no eye human data. As reported in the product information, detectable blood concentrations of the drug were not found in healthy normal subjects indicating that systemic absorption was negligible.
Corticosteroids
Among the nine corticosteroid products, six are suspensions or ointments, one is an injection, and two are implants. In most nonpregnant patients receiving the dexamethasone (Ozurdex) intravitreal implant, plasma dexamethasone concentrations were undetectable (less than 50 pg/mL) but, in some, ranged from 52 pg/mL to 102 pg/mL. There is only one case report describing the use of topical dexamethasone suspension (Maxidex) in pregnancy. In that case, dexamethasone and clindamycin were given in the first and second trimesters and the woman eventually gave birth to a normal full term infant (Int Ophthalmol. 1998-1999;22[2]:85-8).
For the fluocinolone (Retisert) ocular implant, systemic absorption of detectable amounts of the drug have not been observed. In a second report, a patient who had type 1 diabetes and was 6 months pregnant received a 2-mg intravitreal injection of triamcinolone (Triesence) in both eyes. No adverse effects in the mother or fetus were noted (Clin Ophthalmol. 2011;5:439-41).
There are no human eye data for five topical corticosteroids: difluprednate (Durezol), fluorometholone (Flarex, Fluor-OP, FML), loteprednol (Alrex, Lotemax), prednisolone (Econopred), and rimexolone (Vexol). Only two of the five drugs had information about systemic absorption. For difluprednate, blood levels were below the quantification limit (50 ng/mL). Extremely low levels were detected after the use of rimexolone with a mean serum concentration of 130 pg/mL (range less than 80-470 pg/mL).
Cycloplegics-mydriatics
This class includes four anticholinergic agents: atropine, cyclopentolate (Cyclogyl), homatropine, and tropicamide (Mydriacyl). Systemic absorption has not been studied for these drugs and there are no reports of their use in pregnancy or lactation.
Cystine-depleting agents
There are no reports describing the use of cysteamine (Cystaran) in human pregnancy or lactation. Since the drug is given as one drop in each eye every waking hour, transfer to the systemic circulation should be expected, but the amount, if it occurs, has not been reported.
Immunologics
There are no reports on the use of cyclosporine (Restasis) eye drops in human pregnancy or during breastfeeding. However, data for the systemic use of the drug during these conditions has shown it to be low risk. After long term ophthalmic use, blood concentrations of the drug were below the quantitation limit of 0.1 ng/mL.
Local anesthetics
The three drugs in this class are lidocaine, proparacaine (Alcaine), and tetracaine (Altacaine, Tetravisc). There is no information regarding the use of these agents in pregnancy or lactation. Since they are used for brief periods, the risk to an embryo or nursing infant appears to be nil.
Mast cell stabilizers
There are three drugs in this class that can be used topically in the eye: cromolyn, lodoxamide (Alomide), and nedocromil (Alocril). There are no human eye data for these agents. Systemic concentrations of lodoxamide were below the detection limit of 2.5 ng/mL.
Miotics
The two miotics are acetylcholine (Miochol E) and carbachol (Carbastat, Miostat). Both are used immediately before eye surgery. The amount, if any, in the systemic circulation is unknown. No reports of human eye use in pregnancy or lactation have been found.
NSAIDs
The five drugs in this class are bromfenac (Xibrom), diclofenac (Voltaren), flurbiprofen (Ocufen), ketorolac, and nepafenac (Nevanac). Although it has not been studied, the estimated plasma level for bromfenac is less than 50 ng/mL. For diclofenac, the plasma concentration was below the limit of quantification (10 ng/mL). In a study conducted with ketorolac, only 5 of 26 subjects had detectable plasma concentrations (10.7 to 22.5 ng/mL). These levels were about 2% of the peak plasma levels after oral dosing. For nepafenac, the peak plasma concentrations of the parent drug and active metabolite were 0.3 and 0.4 ng/mL, respectively. These data suggest that the risk to the embryo-fetus or a nursing infant are nil.
Photodynamic therapy
Verteporfin (Visudyne) is given intravenously. Three reports have described its use in three pregnancies. In all cases, the infants were healthy at birth and had normal growth (Acta Ophthalmol Scand. 2004 Oct;82[5]:623-4, Eye [Lond]. 2009 Jun;23[6]:1479, and Aust N Z J Obstet Gynaecol. 2009 Apr;49[2]:236-7).
Proteolytic enzymes
No human eye data were found for ocriplasmin (Jetrea), an agent given as an intravitreal injection. Although it was not studied, detectable levels of the drug in the systemic circulation are not expected, according to the manufacturer.
Selective vascular endothelial growth factor antagonists
There are three agents in this class: aflibercept (Eylea), pegaptanib (Macugen), and ranibizumab (Lucentis). There are no human eye data for these drugs. All are given as intravitreal injection. The systemic concentrations of the three drugs were a mean 0.02 mcg/mL; 80 ng/mL (after a dose of 10 times the recommended dose); and an estimated minimum 0.22 ng/mL, respectively. These data suggest that the risk to the embryo-fetus or a nursing infant is nil.
Sympathomimetics (decongestants)
There are four ophthalmic agents in this class and none have human eye data. Naphazoline (Naphcon, Vasocon) is the only one of the four that requires a prescription. No reports describing the systemic absorption, if any, have been found. The other three agents are available over the counter. They are oxymetazoline (Visine L.R.), phenylephrine, and tetrahydrozoline (Opti-Clear). Although the amount reaching the systemic circulation was not provided by the manufacturers for these three agents, a caution was placed on phenylephrine stating that systemic absorption, although rare, might cause alpha-adrenergic effects, such as a rise in blood pressure and reflex atropine-sensitive bradycardia.
The risk to the embryo-fetus or nursing infant from ophthalmic drugs appears to be low, with the possible exception of phenylephrine. Nevertheless, if any of these agents are used in pregnancy or during breastfeeding, careful assessment of the embryo-fetus and nursing infant should be conducted. Moreover, research on the potential effects of ophthalmic drugs on the embryo-fetus and nursing infant are desperately needed.
Mr. Briggs is clinical professor of pharmacy at the University of California, San Francisco; and adjunct professor of pharmacy at the University of Southern California, Los Angeles, and Washington State University, Spokane. He is coauthor of “Drugs in Pregnancy and Lactation,” and coeditor of “Diseases, Complications, and Drug Therapy in Obstetrics.” He has no relevant financial disclosures.
One-step gestational diabetes screen not beneficial
Switching to the new one-step screen for gestational diabetes, recommended by the International Association of the Diabetes and Pregnancy Study Group and endorsed by the World Health Organization, failed to reduce the rates of large-for-gestational-age neonates, macrosomia, neonatal intensive care unit admissions, preterm births, preeclampsia, and shoulder dystocia, according to a new study.
Clinicians in the United States have traditionally screened for gestational diabetes using a two-step test early in gestation in those patients considered high risk, with a second test at 24-28 weeks of gestation for average-risk patients. Values were considered abnormal if they exceeded those recommended by either the Carpenter and Coustan and Fourth International Workshop–Conference Criteria or the values designated by the National Diabetes Group. The American College of Obstetricians and Gynecologists continues to recommend this two-step approach, according to Dr. R. Klara Feldman of the department of ob.gyn., Kaiser Permanente at Baldwin Park, Montebello, Calif., and her associates.
But based on the results of observational studies, some in the ob.gyn. community thought that expanding the number of women diagnosed and treated early would decrease the number of LGA neonates and related complications. In 2010, the IADPSG and the WHO recommended that instead of the two-step screen, clinicians should perform a hemoglobin A1c test, a random plasma glucose test, or a fasting plasma glucose test at the first prenatal visit to identify undiagnosed pregestational diabetes. For all women not found to have pregestational diabetes, a one-step, 2-hour glucose tolerance test is recommended at 24-28 weeks.
To determine whether switching to the new screening method decreased the number of LGA neonates as hoped, Dr. Feldman and her associates performed a before-after retrospective cohort study comparing the outcomes of 2,972 singleton pregnancies before their hospital and its affiliated clinics switched from the two-step Carpenter-Coustan screen to the one-step IADPSG screen, with 3,094 singleton pregnancies after the change.
The care of patients with gestational diabetes did not change during this period, and the two study groups showed no significant differences in mother’s prenatal weight, mother’s body mass index at initial prenatal visit, the number of office visits during pregnancy, or the percentage of deliveries requiring labor induction.
The rate of diagnosed gestational diabetes increased as expected after the new screen was implemented, from 17% to 27%. However, the primary outcome measure – the percentage of LGA neonates – did not decrease significantly. That rate was 10% in the “before” group and 9% in the “after” group. Similarly, rates of macrosomia, NICU admission, preterm birth, preeclampsia, and shoulder dystocia remained unchanged after implementation of the new screening approach (Obstet Gynecol. 2016;127:10-7. doi: 10.1097/AOG.0000000000001132).
“The cutoffs chosen by the IADPSG may be too low and thus result in too many patients being treated as having gestational diabetes,” they added. “Different cutoff values need to be evaluated, and more attention needs to be focused on controlling prepregnancy BMI.”
The Kaiser Permanente Regional Research Committee supported the study. Dr. Feldman and her associates reported having no relevant financial disclosures.
Switching to the new one-step screen for gestational diabetes, recommended by the International Association of the Diabetes and Pregnancy Study Group and endorsed by the World Health Organization, failed to reduce the rates of large-for-gestational-age neonates, macrosomia, neonatal intensive care unit admissions, preterm births, preeclampsia, and shoulder dystocia, according to a new study.
Clinicians in the United States have traditionally screened for gestational diabetes using a two-step test early in gestation in those patients considered high risk, with a second test at 24-28 weeks of gestation for average-risk patients. Values were considered abnormal if they exceeded those recommended by either the Carpenter and Coustan and Fourth International Workshop–Conference Criteria or the values designated by the National Diabetes Group. The American College of Obstetricians and Gynecologists continues to recommend this two-step approach, according to Dr. R. Klara Feldman of the department of ob.gyn., Kaiser Permanente at Baldwin Park, Montebello, Calif., and her associates.
But based on the results of observational studies, some in the ob.gyn. community thought that expanding the number of women diagnosed and treated early would decrease the number of LGA neonates and related complications. In 2010, the IADPSG and the WHO recommended that instead of the two-step screen, clinicians should perform a hemoglobin A1c test, a random plasma glucose test, or a fasting plasma glucose test at the first prenatal visit to identify undiagnosed pregestational diabetes. For all women not found to have pregestational diabetes, a one-step, 2-hour glucose tolerance test is recommended at 24-28 weeks.
To determine whether switching to the new screening method decreased the number of LGA neonates as hoped, Dr. Feldman and her associates performed a before-after retrospective cohort study comparing the outcomes of 2,972 singleton pregnancies before their hospital and its affiliated clinics switched from the two-step Carpenter-Coustan screen to the one-step IADPSG screen, with 3,094 singleton pregnancies after the change.
The care of patients with gestational diabetes did not change during this period, and the two study groups showed no significant differences in mother’s prenatal weight, mother’s body mass index at initial prenatal visit, the number of office visits during pregnancy, or the percentage of deliveries requiring labor induction.
The rate of diagnosed gestational diabetes increased as expected after the new screen was implemented, from 17% to 27%. However, the primary outcome measure – the percentage of LGA neonates – did not decrease significantly. That rate was 10% in the “before” group and 9% in the “after” group. Similarly, rates of macrosomia, NICU admission, preterm birth, preeclampsia, and shoulder dystocia remained unchanged after implementation of the new screening approach (Obstet Gynecol. 2016;127:10-7. doi: 10.1097/AOG.0000000000001132).
“The cutoffs chosen by the IADPSG may be too low and thus result in too many patients being treated as having gestational diabetes,” they added. “Different cutoff values need to be evaluated, and more attention needs to be focused on controlling prepregnancy BMI.”
The Kaiser Permanente Regional Research Committee supported the study. Dr. Feldman and her associates reported having no relevant financial disclosures.
Switching to the new one-step screen for gestational diabetes, recommended by the International Association of the Diabetes and Pregnancy Study Group and endorsed by the World Health Organization, failed to reduce the rates of large-for-gestational-age neonates, macrosomia, neonatal intensive care unit admissions, preterm births, preeclampsia, and shoulder dystocia, according to a new study.
Clinicians in the United States have traditionally screened for gestational diabetes using a two-step test early in gestation in those patients considered high risk, with a second test at 24-28 weeks of gestation for average-risk patients. Values were considered abnormal if they exceeded those recommended by either the Carpenter and Coustan and Fourth International Workshop–Conference Criteria or the values designated by the National Diabetes Group. The American College of Obstetricians and Gynecologists continues to recommend this two-step approach, according to Dr. R. Klara Feldman of the department of ob.gyn., Kaiser Permanente at Baldwin Park, Montebello, Calif., and her associates.
But based on the results of observational studies, some in the ob.gyn. community thought that expanding the number of women diagnosed and treated early would decrease the number of LGA neonates and related complications. In 2010, the IADPSG and the WHO recommended that instead of the two-step screen, clinicians should perform a hemoglobin A1c test, a random plasma glucose test, or a fasting plasma glucose test at the first prenatal visit to identify undiagnosed pregestational diabetes. For all women not found to have pregestational diabetes, a one-step, 2-hour glucose tolerance test is recommended at 24-28 weeks.
To determine whether switching to the new screening method decreased the number of LGA neonates as hoped, Dr. Feldman and her associates performed a before-after retrospective cohort study comparing the outcomes of 2,972 singleton pregnancies before their hospital and its affiliated clinics switched from the two-step Carpenter-Coustan screen to the one-step IADPSG screen, with 3,094 singleton pregnancies after the change.
The care of patients with gestational diabetes did not change during this period, and the two study groups showed no significant differences in mother’s prenatal weight, mother’s body mass index at initial prenatal visit, the number of office visits during pregnancy, or the percentage of deliveries requiring labor induction.
The rate of diagnosed gestational diabetes increased as expected after the new screen was implemented, from 17% to 27%. However, the primary outcome measure – the percentage of LGA neonates – did not decrease significantly. That rate was 10% in the “before” group and 9% in the “after” group. Similarly, rates of macrosomia, NICU admission, preterm birth, preeclampsia, and shoulder dystocia remained unchanged after implementation of the new screening approach (Obstet Gynecol. 2016;127:10-7. doi: 10.1097/AOG.0000000000001132).
“The cutoffs chosen by the IADPSG may be too low and thus result in too many patients being treated as having gestational diabetes,” they added. “Different cutoff values need to be evaluated, and more attention needs to be focused on controlling prepregnancy BMI.”
The Kaiser Permanente Regional Research Committee supported the study. Dr. Feldman and her associates reported having no relevant financial disclosures.
FROM OBSTETRICS AND GYNECOLOGY
Key clinical point: Switching to the new 1-step screen for gestational diabetes recommended by the International Association of the Diabetes and Pregnancy Study Group failed to reduce the rates of large-for-gestational age neonates, macrosomia, neonatal intensive care unit admissions, preterm births, preeclampsia, or shoulder dystocia.
Major finding: The primary outcome measure – the percentage of LGA neonates – was 10% before switching to the new screen and 9% afterward, a nonsignificant difference.
Data source: A before-after retrospective cohort study comparing outcomes of 2,972 pregnancies before the switch to the new screen against 3,094 afterward.
Disclosures: The Kaiser Permanente Regional Research Committee supported the study. Dr. Feldman and her associates reported having no relevant financial disclosures.