User login
VIDEO – IBD meds found safe during pregnancy
CHICAGO – Medications to treat inflammatory bowel disease were well tolerated in pregnancy and weren’t linked to worse maternal-fetal outcomes, according to a study of more than 1,000 pregnant women.
Despite many physicians’ concerns about the safety of azathioprine and biologic agents during pregnancy, maternal exposure to those IBD medications was not associated with an increase in adverse outcomes such as preterm birth or congenital anomalies, explained Dr. Uma Mahadevan of the University of California, San Francisco Center for Colitis and Crohn’s Disease.
"If you continue [the medications] and control your disease and have a healthy pregnancy, that’s the key to having a healthy baby," Dr. Mahadevan observed.
In a video interview at the annual Digestive Disease Week, Dr. Mahadevan discussed the study’s findings, whether combination therapy has an impact on the risk of preterm birth, and what effect IBD medication exposure may have had on children’s achievement of developmental milestones up to age 4 years.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Medications to treat inflammatory bowel disease were well tolerated in pregnancy and weren’t linked to worse maternal-fetal outcomes, according to a study of more than 1,000 pregnant women.
Despite many physicians’ concerns about the safety of azathioprine and biologic agents during pregnancy, maternal exposure to those IBD medications was not associated with an increase in adverse outcomes such as preterm birth or congenital anomalies, explained Dr. Uma Mahadevan of the University of California, San Francisco Center for Colitis and Crohn’s Disease.
"If you continue [the medications] and control your disease and have a healthy pregnancy, that’s the key to having a healthy baby," Dr. Mahadevan observed.
In a video interview at the annual Digestive Disease Week, Dr. Mahadevan discussed the study’s findings, whether combination therapy has an impact on the risk of preterm birth, and what effect IBD medication exposure may have had on children’s achievement of developmental milestones up to age 4 years.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Medications to treat inflammatory bowel disease were well tolerated in pregnancy and weren’t linked to worse maternal-fetal outcomes, according to a study of more than 1,000 pregnant women.
Despite many physicians’ concerns about the safety of azathioprine and biologic agents during pregnancy, maternal exposure to those IBD medications was not associated with an increase in adverse outcomes such as preterm birth or congenital anomalies, explained Dr. Uma Mahadevan of the University of California, San Francisco Center for Colitis and Crohn’s Disease.
"If you continue [the medications] and control your disease and have a healthy pregnancy, that’s the key to having a healthy baby," Dr. Mahadevan observed.
In a video interview at the annual Digestive Disease Week, Dr. Mahadevan discussed the study’s findings, whether combination therapy has an impact on the risk of preterm birth, and what effect IBD medication exposure may have had on children’s achievement of developmental milestones up to age 4 years.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
At DDW 2014
IBD Meds Found Safe During Pregnancy
CHICAGO – Medications to treat inflammatory bowel disease were well tolerated in pregnancy and weren’t linked to worse maternal-fetal outcomes, according to a study of more than 1,000 pregnant women.
Despite many physicians’ concerns about the safety of azathioprine and biologic agents during pregnancy, maternal exposure to those IBD medications was not associated with an increase in adverse outcomes such as preterm birth or congenital anomalies, explained Dr. Uma Mahadevan of the University of California, San Francisco Center for Colitis and Crohn’s Disease.
"If you continue [the medications] and control your disease and have a healthy pregnancy, that’s the key to having a healthy baby," Dr. Mahadevan observed.
In a video interview at the annual Digestive Disease Week, Dr. Mahadevan discussed the study’s findings, whether combination therapy has an impact on the risk of preterm birth, and what effect IBD medication exposure may have had on children’s achievement of developmental milestones up to age 4 years.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Medications to treat inflammatory bowel disease were well tolerated in pregnancy and weren’t linked to worse maternal-fetal outcomes, according to a study of more than 1,000 pregnant women.
Despite many physicians’ concerns about the safety of azathioprine and biologic agents during pregnancy, maternal exposure to those IBD medications was not associated with an increase in adverse outcomes such as preterm birth or congenital anomalies, explained Dr. Uma Mahadevan of the University of California, San Francisco Center for Colitis and Crohn’s Disease.
"If you continue [the medications] and control your disease and have a healthy pregnancy, that’s the key to having a healthy baby," Dr. Mahadevan observed.
In a video interview at the annual Digestive Disease Week, Dr. Mahadevan discussed the study’s findings, whether combination therapy has an impact on the risk of preterm birth, and what effect IBD medication exposure may have had on children’s achievement of developmental milestones up to age 4 years.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Medications to treat inflammatory bowel disease were well tolerated in pregnancy and weren’t linked to worse maternal-fetal outcomes, according to a study of more than 1,000 pregnant women.
Despite many physicians’ concerns about the safety of azathioprine and biologic agents during pregnancy, maternal exposure to those IBD medications was not associated with an increase in adverse outcomes such as preterm birth or congenital anomalies, explained Dr. Uma Mahadevan of the University of California, San Francisco Center for Colitis and Crohn’s Disease.
"If you continue [the medications] and control your disease and have a healthy pregnancy, that’s the key to having a healthy baby," Dr. Mahadevan observed.
In a video interview at the annual Digestive Disease Week, Dr. Mahadevan discussed the study’s findings, whether combination therapy has an impact on the risk of preterm birth, and what effect IBD medication exposure may have had on children’s achievement of developmental milestones up to age 4 years.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
At DDW 2014
Barrier film during repeat C-section confers no clinical benefit
CHICAGO – The use of a sodium hyaluronate–carboxycellulose adhesion barrier during primary cesarean delivery was not associated with decreased delivery time or improved adhesion scores during a first repeat cesarean delivery in a cohort study involving 97 women.
The incision to delivery time among 71 women who did not receive sodium hyaluronate–carboxycellulose (HA-CC) barrier film during their primary cesarean delivery was 9.5 minutes, compared with 10.6 minutes in 26 women who did receive HA-CC barrier film, Dr. Maria Gaspar-Oishi of the University of Hawaii, Honolulu, reported at the annual meeting of the American Congress of Obstetricians and Gynecologists.
Average blood loss and adhesion scores also were similar in the two groups. Blood loss was 564 mL and 563 mL, in the groups, respectively; mean fascia adhesion scores were 1.45 and 1.31; and mean intraperitoneal adhesion scores were 1.11 and 0.92, said Dr. Gaspar-Oishi, whose paper received a Donald F. Richardson Memorial Prize Paper award as one of the best papers by a junior fellow at the meeting.
Patients included in the combined prospective/retrospective cohort study were women who gave birth at a single center. Intraoperative data were collected prospectively at the time of first repeat cesarean delivery, and a retrospective chart study was performed for each subject to determine whether HA-CC barrier film was used during the primary cesarean delivery. The two study groups were similar with respect to age, body mass index, parity, and gestational age.
HA-CC barrier film is commonly used in gynecologic and other surgeries in an effort to reduce the risk of postoperative adhesions, which can result in significant morbidity – including pain, infertility, and bowel obstruction, Dr. Gaspar-Oishi said.
Data with respect to the use of HA-CC barrier film for cesarean delivery, however, are limited. In fact, this is the first prospective study to show that barrier film use does not improve delivery time or adhesion severity in subsequent cesarean deliveries, she noted.
The findings support a recent commentary in Obstetrics & Gynecology, which stated that the routine use of HA-CC barrier film is ill advised because of a lack of evidence regarding clinical benefit, she noted.
Although limited by the fact that about 80% of the women in the study were Asian American (an accurate reflection of the population served by the study hospital), the findings are nonetheless important, because an estimated 90% of all primary cesarean deliveries will result in a repeat cesarean delivery, and because HA-CC barrier film use can be costly, she said.
Additional study in a more diverse population is needed to allow for more generalized conclusions, she noted.
Dr. Gaspar-Oishi reported having no relevant financial disclosures.
CHICAGO – The use of a sodium hyaluronate–carboxycellulose adhesion barrier during primary cesarean delivery was not associated with decreased delivery time or improved adhesion scores during a first repeat cesarean delivery in a cohort study involving 97 women.
The incision to delivery time among 71 women who did not receive sodium hyaluronate–carboxycellulose (HA-CC) barrier film during their primary cesarean delivery was 9.5 minutes, compared with 10.6 minutes in 26 women who did receive HA-CC barrier film, Dr. Maria Gaspar-Oishi of the University of Hawaii, Honolulu, reported at the annual meeting of the American Congress of Obstetricians and Gynecologists.
Average blood loss and adhesion scores also were similar in the two groups. Blood loss was 564 mL and 563 mL, in the groups, respectively; mean fascia adhesion scores were 1.45 and 1.31; and mean intraperitoneal adhesion scores were 1.11 and 0.92, said Dr. Gaspar-Oishi, whose paper received a Donald F. Richardson Memorial Prize Paper award as one of the best papers by a junior fellow at the meeting.
Patients included in the combined prospective/retrospective cohort study were women who gave birth at a single center. Intraoperative data were collected prospectively at the time of first repeat cesarean delivery, and a retrospective chart study was performed for each subject to determine whether HA-CC barrier film was used during the primary cesarean delivery. The two study groups were similar with respect to age, body mass index, parity, and gestational age.
HA-CC barrier film is commonly used in gynecologic and other surgeries in an effort to reduce the risk of postoperative adhesions, which can result in significant morbidity – including pain, infertility, and bowel obstruction, Dr. Gaspar-Oishi said.
Data with respect to the use of HA-CC barrier film for cesarean delivery, however, are limited. In fact, this is the first prospective study to show that barrier film use does not improve delivery time or adhesion severity in subsequent cesarean deliveries, she noted.
The findings support a recent commentary in Obstetrics & Gynecology, which stated that the routine use of HA-CC barrier film is ill advised because of a lack of evidence regarding clinical benefit, she noted.
Although limited by the fact that about 80% of the women in the study were Asian American (an accurate reflection of the population served by the study hospital), the findings are nonetheless important, because an estimated 90% of all primary cesarean deliveries will result in a repeat cesarean delivery, and because HA-CC barrier film use can be costly, she said.
Additional study in a more diverse population is needed to allow for more generalized conclusions, she noted.
Dr. Gaspar-Oishi reported having no relevant financial disclosures.
CHICAGO – The use of a sodium hyaluronate–carboxycellulose adhesion barrier during primary cesarean delivery was not associated with decreased delivery time or improved adhesion scores during a first repeat cesarean delivery in a cohort study involving 97 women.
The incision to delivery time among 71 women who did not receive sodium hyaluronate–carboxycellulose (HA-CC) barrier film during their primary cesarean delivery was 9.5 minutes, compared with 10.6 minutes in 26 women who did receive HA-CC barrier film, Dr. Maria Gaspar-Oishi of the University of Hawaii, Honolulu, reported at the annual meeting of the American Congress of Obstetricians and Gynecologists.
Average blood loss and adhesion scores also were similar in the two groups. Blood loss was 564 mL and 563 mL, in the groups, respectively; mean fascia adhesion scores were 1.45 and 1.31; and mean intraperitoneal adhesion scores were 1.11 and 0.92, said Dr. Gaspar-Oishi, whose paper received a Donald F. Richardson Memorial Prize Paper award as one of the best papers by a junior fellow at the meeting.
Patients included in the combined prospective/retrospective cohort study were women who gave birth at a single center. Intraoperative data were collected prospectively at the time of first repeat cesarean delivery, and a retrospective chart study was performed for each subject to determine whether HA-CC barrier film was used during the primary cesarean delivery. The two study groups were similar with respect to age, body mass index, parity, and gestational age.
HA-CC barrier film is commonly used in gynecologic and other surgeries in an effort to reduce the risk of postoperative adhesions, which can result in significant morbidity – including pain, infertility, and bowel obstruction, Dr. Gaspar-Oishi said.
Data with respect to the use of HA-CC barrier film for cesarean delivery, however, are limited. In fact, this is the first prospective study to show that barrier film use does not improve delivery time or adhesion severity in subsequent cesarean deliveries, she noted.
The findings support a recent commentary in Obstetrics & Gynecology, which stated that the routine use of HA-CC barrier film is ill advised because of a lack of evidence regarding clinical benefit, she noted.
Although limited by the fact that about 80% of the women in the study were Asian American (an accurate reflection of the population served by the study hospital), the findings are nonetheless important, because an estimated 90% of all primary cesarean deliveries will result in a repeat cesarean delivery, and because HA-CC barrier film use can be costly, she said.
Additional study in a more diverse population is needed to allow for more generalized conclusions, she noted.
Dr. Gaspar-Oishi reported having no relevant financial disclosures.
AT THE ACOG ANNUAL CLINICAL MEETING
Key clinical point: HA-CC barrier film doesn’t reduce adhesions when used during a first repeat C-section.
Major finding: Incision to delivery time in those who did not receive HA-CC barrier film was 9.5 minutes vs. 10.6 minutes in those who did.
Data source: A combined prospective/retrospective cohort study of 97 women.
Disclosures: Dr. Gaspar-Oishi reported having no relevant financial disclosures.
VIDEO – National effort to reduce maternal deaths and morbidities gains traction
CHICAGO – The 3,200 U. S. hospitals that offer obstetric care are being urged to reduce maternal morbidity and mortality by joining the National Partnership for Maternal Safety. The initial aim of the collaborative is to implement priority bundles for the three most common preventable causes of maternal death and severe morbidity: obstetric hemorrhage, severe hypertension in pregnancy, and peripartum venous thromboembolism.
Efforts by Dr. Elliott Main to turn maternal mortality reviews into quality improvement materials for preeclampsia and hemorrhage were instrumental in the efforts. In this exclusive video, Dr. Main, medical director of the California Maternal Quality Care Collaborative and chair of obstetrics and gynecology at California Pacific Medical Center in San Francisco, discusses how those efforts are beginning to take root nationally with Dr. Mary Elizabeth D’Alton, chair of obstetrics and gynecology at Columbia University, New York, and Dr. Sarah J. Kilpatrick, chair of obstetrics and gynecology at Cedars-Sinai, Los Angeles.
Dr. Main interviewed Dr. D’Alton and Dr. Kilpatrick at the 2014 ACOG annual clinical meeting after they presented the Edith Louise Potter Memorial Lecture titled "Maternal Morbidity and Mortality in the U.S.: Time to Wake Up and Take the Lead."
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – The 3,200 U. S. hospitals that offer obstetric care are being urged to reduce maternal morbidity and mortality by joining the National Partnership for Maternal Safety. The initial aim of the collaborative is to implement priority bundles for the three most common preventable causes of maternal death and severe morbidity: obstetric hemorrhage, severe hypertension in pregnancy, and peripartum venous thromboembolism.
Efforts by Dr. Elliott Main to turn maternal mortality reviews into quality improvement materials for preeclampsia and hemorrhage were instrumental in the efforts. In this exclusive video, Dr. Main, medical director of the California Maternal Quality Care Collaborative and chair of obstetrics and gynecology at California Pacific Medical Center in San Francisco, discusses how those efforts are beginning to take root nationally with Dr. Mary Elizabeth D’Alton, chair of obstetrics and gynecology at Columbia University, New York, and Dr. Sarah J. Kilpatrick, chair of obstetrics and gynecology at Cedars-Sinai, Los Angeles.
Dr. Main interviewed Dr. D’Alton and Dr. Kilpatrick at the 2014 ACOG annual clinical meeting after they presented the Edith Louise Potter Memorial Lecture titled "Maternal Morbidity and Mortality in the U.S.: Time to Wake Up and Take the Lead."
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – The 3,200 U. S. hospitals that offer obstetric care are being urged to reduce maternal morbidity and mortality by joining the National Partnership for Maternal Safety. The initial aim of the collaborative is to implement priority bundles for the three most common preventable causes of maternal death and severe morbidity: obstetric hemorrhage, severe hypertension in pregnancy, and peripartum venous thromboembolism.
Efforts by Dr. Elliott Main to turn maternal mortality reviews into quality improvement materials for preeclampsia and hemorrhage were instrumental in the efforts. In this exclusive video, Dr. Main, medical director of the California Maternal Quality Care Collaborative and chair of obstetrics and gynecology at California Pacific Medical Center in San Francisco, discusses how those efforts are beginning to take root nationally with Dr. Mary Elizabeth D’Alton, chair of obstetrics and gynecology at Columbia University, New York, and Dr. Sarah J. Kilpatrick, chair of obstetrics and gynecology at Cedars-Sinai, Los Angeles.
Dr. Main interviewed Dr. D’Alton and Dr. Kilpatrick at the 2014 ACOG annual clinical meeting after they presented the Edith Louise Potter Memorial Lecture titled "Maternal Morbidity and Mortality in the U.S.: Time to Wake Up and Take the Lead."
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
EXPERT ANALYSIS FROM THE ACOG ANNUAL CLINICAL MEETING
VIDEO: Cervical length at 16-22 weeks predicts preterm birth
CHICAGO – Transvaginal ultrasound measures of cervical length at 16-22 weeks gestation can identify patients who have a short cervix and are at risk of preterm birth, allowing selection of patients for vaginal progesterone therapy.
At the annual meeting of the American Congress of Obstetricians and Gynecologists, Dr. Eric Strand, director of the division of general obstetrics and gynecology at Washington University, St. Louis, interviews Dr. George Andrew Macones, the Mitchell and Elaine Yanow Professor and chair of the department of obstetrics and gynecology at Washington University, about these findings and how clinicians can get free training in performing transvaginal screening to determine cervical length. Dr. Macones presented the March of Dimes Annual Lecture entitled "Preventing Preterm Birth: Recent Progress and Future Directions."
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Transvaginal ultrasound measures of cervical length at 16-22 weeks gestation can identify patients who have a short cervix and are at risk of preterm birth, allowing selection of patients for vaginal progesterone therapy.
At the annual meeting of the American Congress of Obstetricians and Gynecologists, Dr. Eric Strand, director of the division of general obstetrics and gynecology at Washington University, St. Louis, interviews Dr. George Andrew Macones, the Mitchell and Elaine Yanow Professor and chair of the department of obstetrics and gynecology at Washington University, about these findings and how clinicians can get free training in performing transvaginal screening to determine cervical length. Dr. Macones presented the March of Dimes Annual Lecture entitled "Preventing Preterm Birth: Recent Progress and Future Directions."
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – Transvaginal ultrasound measures of cervical length at 16-22 weeks gestation can identify patients who have a short cervix and are at risk of preterm birth, allowing selection of patients for vaginal progesterone therapy.
At the annual meeting of the American Congress of Obstetricians and Gynecologists, Dr. Eric Strand, director of the division of general obstetrics and gynecology at Washington University, St. Louis, interviews Dr. George Andrew Macones, the Mitchell and Elaine Yanow Professor and chair of the department of obstetrics and gynecology at Washington University, about these findings and how clinicians can get free training in performing transvaginal screening to determine cervical length. Dr. Macones presented the March of Dimes Annual Lecture entitled "Preventing Preterm Birth: Recent Progress and Future Directions."
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE ACOG ANNUAL CLINICAL MEETING
Noninvasive prenatal testing: Where we are and where we’re going
The introduction of amniocentesis in the 1960s brought to prenatal diagnosticians the ability to detect fetal chromosome abnormalities and certain structural defects (including neural tube defects). Since that time, a goal for these practitioners has been the development of effective screening algorithms to better identify women at high risk for detectable fetal abnormalities in concert with the advent of safer and more accessible diagnostic tests, with the eventual aim being the development of a noninvasive prenatal diagnostic test.
Postamniocentesis advancements have included the identification of maternal serum analytes as well as the incorporation of first-trimester ultrasonographic measurements of the fetal nuchal translucency (NT) and nasal bone, all associated with an improved ability to identify women at increased risk for fetal trisomies 21 and 18 as well as some other fetal abnormalities. In addition, targeted ultrasound has greatly improved the ability to detect fetal structural and growth abnormalities in women of all risk levels, although it remains a highly subjective process with considerable inter/intraoperator and equipment variability.
Related article: NIPT is expanding rapidly--but don't throw out that CVS kit just yet! (Update on Obstetrics; Jaimey M. Pauli, MD, and John T. Repke, MD; January 2014)
Noninvasive prenatal screening has the advantages of being noninvasive and carrying no increased risk for fetal loss compared with chorionic villus sampling (CVS) and amniocentesis, which are associated with a small increased risk for pregnancy loss (1/500 to 1/1,500 over baseline risk for loss). However, noninvasive screening is limited compared with diagnostic procedures because it provides only a risk adjustment rather than a definitive diagnostic outcome and is mostly limited to assessment for fetal trisomies 18 and 21.
Targeted ultrasound can identify structural abnormalities associated with other chromosomal, genetic, and genomic abnormalities, but again depends on operator experience, equipment used, maternal habitus, and fetal position. Accordingly, considerable interest has remained in developing a more effective approach for detecting fetal aneuploidy and other fetal abnormalities, including assays that eventually could serve to provide noninvasive prenatal diagnosis.
RECENT ADVANCES BRING US CLOSER TO OUR ULTIMATE GOAL
The recent introduction of circulating cell-free nucleic acids (ccfna) technologies for prenatal screening for common fetal aneuploidies, better known as noninvasive prenatal testing, or NIPT, has presented a far more effective prenatal screening protocol for certain groups of women compared with the aforementioned screening algorithms that rely on measurements of the fetal NT in the late first trimester and maternal serum measurements of analytes in the first and second trimesters.
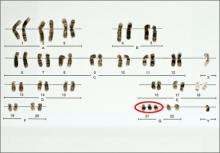
Currently, four NIPT screening products are available commercially in the United States: MaterniT21 Plus (Sequenom, San Diego, California); Verifi (Illumina, San Diego, California); Harmony Prenatal Test (Ariosa Diagnostics, San Jose, California); and Panorama Prenatal Test (Natera, San Carlos, California). While the technologies and algorithms used by each of the companies differ, they all rely on the premise that 5% to 10% of ccfna in maternal blood are fetal in nature.1 Calculating the ratios of the expected amount of each chromosome-specific nucleic acid to that actually measured in the sample, a prediction of a normal or abnormal complement for that specific chromosome is then made. None of the commercially available tests specifically identify fetal DNA or differentiate fetal from maternal DNA.
Current validation studies have thus far limited the offering of NIPT to women at increased risk for fetal aneuploidy, including those:2–6
- of advanced maternal age
- with a positive conventional screening test
- with abnormal ultrasound results suggestive of aneuploidy, or
- who have had a prior pregnancy with a chromosome aneuploidy found in the NIPT panel.
Studies of all available technologies tested on women at increased risk for fetal aneuploidy have thus far shown considerably higher sensitivities and specificities and detection rates for fetal trisomies 21, 18, and 13 than conventional screening algorithms, although detection rates for trisomy 13 are slightly lower than those observed for trisomies 21 and 18.
WE STILL HAVE MANY HURDLES TO LEAP
However, the groups of women at high risk for fetal aneuploidy just outlined represent only a small segment of the community of pregnant women. A multicenter study involving 1,914 women published February 2014 in the New England Journal of Medicine7 showed considerably and significantly lower false-positive rates and higher positive predictive values for the detection of trisomies 21 and 18 by NIPT compared with conventional fetal aneuploidy screening. This study incorporated women at low risk for fetal aneuploidies in the study cohort, although women at high risk (based on the stated range of maternal age) also were included in the cohort. Unfortunately, no information was provided in the report about the percentage of low-risk women among the study participants.
Related articles:
Noninvasive prenatal DNA testing: Who is using it, and how? Audiocast, June 2013
Noninvasive prenatal DNA tests are unproven and costly David A. Carpenter, MD (Comment & Controversy; September 2013)
Another concern about the published accuracy of NIPT clinical assays was recently sounded by Menutti and colleagues.8 The authors cited recent cases of positive NIPT outcomes for fetal trisomies 18 and 13 that were not confirmed by diagnostic testing of the pregnancies in question. The authors pondered whether such cases may reflect a limitation of the positive predictive values attributed to NIPT assays and that such limitations may carry profound inaccuracies in determining the accuracy of such protocols for rare aneuploidies.
While the improved detection rates for NIPT compared with conventional screening are not surprising, guidelines published by the American College of Obstetricians and Gynecologists still do not recommend the use of NIPT for the screening of low-risk women because of insufficient evaluation of ccfna technologies in the screening of such pregnancies.3 This also applies to twin pregnancies, despite preliminary studies showing comparable detection of trisomies 18 and 21 in such pregnancies compared with singleton pregnancies.3,9
There are no direct comparative studies of the four commercially available screening products, thus precluding a robust comparison and determination of the best existing method to use.
SO, WHERE ARE WE WITH NIPT EXACTLY?
The recent introduction of NIPT into routine obstetric care has left many clinicians with a wide range of questions, many of which cannot be answered because of little or no information, robust or otherwise, to formulate an accurate and cogent response. So let’s state what we know based on the available evidence, recognizing that this will likely change, perhaps considerably, in the weeks and months ahead.
NIPT is a far superior approach, compared with conventional screening approaches, to screening for fetal trisomies 21, 18, and 13 in women carrying singleton pregnancies who are at an increased risk for fetal chromosome abnormalities.
In our current understanding of prenatal screening and diagnosis, NIPT does not provide either the comprehensive approach or the diagnostic accuracy associated with CVS and amniocentesis. As such, NIPT is not a suitable replacement for prenatal diagnostic procedures.
However, its application to screening a low-risk population for the common fetal aneuploidies, as well as in twin pregnancies, has been supported by initial studies, and the inclusion of other clinical outcomes—including other chromosome abnormalities, such as X and Y aneuploidies, trisomy 16, and triploidy10,11 and certain genomic abnormalities (eg, 22q deletions)—in the screening algorithm will expand the future clinical applications of NIPT screening.
DOES NIPT CHANGE OUR CONCEPTS OF SCREENING AND DIAGNOSIS?
This question is simple but profound and is perhaps the most important to be asked and addressed. Is a screening algorithm that has a similar sensitivity and specificity to that of CVS and amniocentesis for the most common fetal trisomies in the first and second trimesters sufficient to replace invasive testing for most women? Does the ability to detect fetal genomic abnormalities with microarray analyses of fetal cells obtained by CVS or amniocentesis provide a far greater benefit than that possible with any screening algorithm?
With renewed interest in the cost of health-care screening and diagnosis, we need to consider how comprehensive and accurate our prenatal screening and diagnostic tests should be and whether such improvements are desired or even possible from a clinical or economic viewpoint. In addition, the development of new technologies, such as the capture and analysis of fetal cells in maternal blood, presents the potential for a direct diagnostic fetal assay without the risks of an invasive procedure.
BIAS-FREE COUNSELING CANNOT BE OVERLOOKED
That being said, the current role of NIPT and other screening protocols in obstetric care needs to be clearly communicated to women who are considering their fetal assessment options, with emphasis placed on the capabilities and limitations of prenatal screening (even the newer ccfna-based options), the actual risks associated with invasive testing, and the ability of invasive testing to provide expanded fetal information with the use of microarray analyses.
As it has been from the beginning of prenatal testing in the 1960s, counseling continues to be the most important part of the prenatal screening and diagnostic process and it is needed to facilitate clinical decisions made by women and couples. Counseling must include an accurate communication of the risks, benefits, and limitations of the aforementioned options and issues, and should be provided in a manner that strives to be free of bias, direction, and the personal opinions of the counselor.
In order to provide such counseling, we must remain informed of the ongoing work in the field of prenatal testing, a task that has become more challenging with the rapid release of a considerable amount of new information on prenatal screening technologies over the past 2 years. This will likely continue, and perhaps become even more frenetic, with the expected release of additional information on the clinical applications of ccfna technologies in the near future as well as the development of new technologies applicable for the screening and diagnosis of fetal abnormalities.
WE WANT TO HEAR FROM YOU!
Share your thoughts on this article. Send your letter to: [email protected] Please include the city and state in which you practice.
- Lo YM, Corbetta N, Chamberlain PF, et al. Presence of fetal DNA in maternal plasma and serum. Lancet. 1997;350(9076):485–487.
- Ashoor G, Syngelaki A, Wagner M, Birdir C, Nicolaides KH. Chromosome-selective sequencing of maternal plasma cell–free DNA for first-trimester detection of trisomy 21 and trisomy 18. Am J Obstet Gynecol. 2012;206(4):322.e1–e5.
- American College of Obstetricians and Gynecologists Committee on Genetics. Committee Opinion No. 545: Noninvasive prenatal testing for fetal aneuploidy. Obstet Gynecol. 2012;120(6):1532–1534.
- Bianchi DW, Platt LD, Goldberg JD, et al; MatErnal Blood IS Source to Accurately diagnose fetal aneuploidy (MELISSA) Study Group. Genome-wide fetal aneuploidy detection by maternal plasma DNA-sequencing. Obstet Gynecol. 2012;119(5):890–901.
- Palomaki GE, Kloza EM, Lambert-Messerlian GM, et al. DNA sequencing of maternal plasma to detect Down syndrome: An international clinical validation study. Genet Med. 2011;13(11):913–920.
- Palomaki GE, Deciu C, Kloza EM, et al. DNA sequencing of maternal plasma reliably identifies trisomy 18 and trisomy 13 as well as Down syndrome: An international collaborative study. Genet Med. 2012;14(3):296–305.
- Bianchi DW, Parker RL, Wentworth J, et al; CARE Study Group. DNA sequencing versus standard prenatal aneuploidy screening. N Engl J Med. 2014;370(9):799–808.
- Menutti MT, Cherry AM, Morrissette JJ, Dugoff L. Is it time to sound an alarm about false-positive cell-free DNA testing for fetal aneuploidy. Am J Obstet Gynecol. 2013;209(5):415−419.
- Canick JA, Kloza EM, Lambert-Messerlian GM, et al. DNA sequencing of maternal plasma to identify Down syndrome and other trisomies in multiple gestations. Prenat Diagn. 2012;32(8):730–734.
- Nicolaides KH, Syngelaki A, Gil MM, Quezada MS, Zinevich Y. Prenatal detection of fetal triploidy from cell-free DNA testing in maternal blood [published online ahead of print October 10, 2013]. Fetal Diagn Ther.
- Semango-Sprouse C, Banjevic M, Ryan A, et al. SNP-based non-invasive prenatal testing detects sex chromosome aneuploidies with high accuracy. Prenat Diagn. 2013;33(7):643–649.
The introduction of amniocentesis in the 1960s brought to prenatal diagnosticians the ability to detect fetal chromosome abnormalities and certain structural defects (including neural tube defects). Since that time, a goal for these practitioners has been the development of effective screening algorithms to better identify women at high risk for detectable fetal abnormalities in concert with the advent of safer and more accessible diagnostic tests, with the eventual aim being the development of a noninvasive prenatal diagnostic test.
Postamniocentesis advancements have included the identification of maternal serum analytes as well as the incorporation of first-trimester ultrasonographic measurements of the fetal nuchal translucency (NT) and nasal bone, all associated with an improved ability to identify women at increased risk for fetal trisomies 21 and 18 as well as some other fetal abnormalities. In addition, targeted ultrasound has greatly improved the ability to detect fetal structural and growth abnormalities in women of all risk levels, although it remains a highly subjective process with considerable inter/intraoperator and equipment variability.
Related article: NIPT is expanding rapidly--but don't throw out that CVS kit just yet! (Update on Obstetrics; Jaimey M. Pauli, MD, and John T. Repke, MD; January 2014)
Noninvasive prenatal screening has the advantages of being noninvasive and carrying no increased risk for fetal loss compared with chorionic villus sampling (CVS) and amniocentesis, which are associated with a small increased risk for pregnancy loss (1/500 to 1/1,500 over baseline risk for loss). However, noninvasive screening is limited compared with diagnostic procedures because it provides only a risk adjustment rather than a definitive diagnostic outcome and is mostly limited to assessment for fetal trisomies 18 and 21.
Targeted ultrasound can identify structural abnormalities associated with other chromosomal, genetic, and genomic abnormalities, but again depends on operator experience, equipment used, maternal habitus, and fetal position. Accordingly, considerable interest has remained in developing a more effective approach for detecting fetal aneuploidy and other fetal abnormalities, including assays that eventually could serve to provide noninvasive prenatal diagnosis.
RECENT ADVANCES BRING US CLOSER TO OUR ULTIMATE GOAL
The recent introduction of circulating cell-free nucleic acids (ccfna) technologies for prenatal screening for common fetal aneuploidies, better known as noninvasive prenatal testing, or NIPT, has presented a far more effective prenatal screening protocol for certain groups of women compared with the aforementioned screening algorithms that rely on measurements of the fetal NT in the late first trimester and maternal serum measurements of analytes in the first and second trimesters.
Currently, four NIPT screening products are available commercially in the United States: MaterniT21 Plus (Sequenom, San Diego, California); Verifi (Illumina, San Diego, California); Harmony Prenatal Test (Ariosa Diagnostics, San Jose, California); and Panorama Prenatal Test (Natera, San Carlos, California). While the technologies and algorithms used by each of the companies differ, they all rely on the premise that 5% to 10% of ccfna in maternal blood are fetal in nature.1 Calculating the ratios of the expected amount of each chromosome-specific nucleic acid to that actually measured in the sample, a prediction of a normal or abnormal complement for that specific chromosome is then made. None of the commercially available tests specifically identify fetal DNA or differentiate fetal from maternal DNA.
Current validation studies have thus far limited the offering of NIPT to women at increased risk for fetal aneuploidy, including those:2–6
- of advanced maternal age
- with a positive conventional screening test
- with abnormal ultrasound results suggestive of aneuploidy, or
- who have had a prior pregnancy with a chromosome aneuploidy found in the NIPT panel.
Studies of all available technologies tested on women at increased risk for fetal aneuploidy have thus far shown considerably higher sensitivities and specificities and detection rates for fetal trisomies 21, 18, and 13 than conventional screening algorithms, although detection rates for trisomy 13 are slightly lower than those observed for trisomies 21 and 18.
WE STILL HAVE MANY HURDLES TO LEAP
However, the groups of women at high risk for fetal aneuploidy just outlined represent only a small segment of the community of pregnant women. A multicenter study involving 1,914 women published February 2014 in the New England Journal of Medicine7 showed considerably and significantly lower false-positive rates and higher positive predictive values for the detection of trisomies 21 and 18 by NIPT compared with conventional fetal aneuploidy screening. This study incorporated women at low risk for fetal aneuploidies in the study cohort, although women at high risk (based on the stated range of maternal age) also were included in the cohort. Unfortunately, no information was provided in the report about the percentage of low-risk women among the study participants.
Related articles:
Noninvasive prenatal DNA testing: Who is using it, and how? Audiocast, June 2013
Noninvasive prenatal DNA tests are unproven and costly David A. Carpenter, MD (Comment & Controversy; September 2013)
Another concern about the published accuracy of NIPT clinical assays was recently sounded by Menutti and colleagues.8 The authors cited recent cases of positive NIPT outcomes for fetal trisomies 18 and 13 that were not confirmed by diagnostic testing of the pregnancies in question. The authors pondered whether such cases may reflect a limitation of the positive predictive values attributed to NIPT assays and that such limitations may carry profound inaccuracies in determining the accuracy of such protocols for rare aneuploidies.
While the improved detection rates for NIPT compared with conventional screening are not surprising, guidelines published by the American College of Obstetricians and Gynecologists still do not recommend the use of NIPT for the screening of low-risk women because of insufficient evaluation of ccfna technologies in the screening of such pregnancies.3 This also applies to twin pregnancies, despite preliminary studies showing comparable detection of trisomies 18 and 21 in such pregnancies compared with singleton pregnancies.3,9
There are no direct comparative studies of the four commercially available screening products, thus precluding a robust comparison and determination of the best existing method to use.
SO, WHERE ARE WE WITH NIPT EXACTLY?
The recent introduction of NIPT into routine obstetric care has left many clinicians with a wide range of questions, many of which cannot be answered because of little or no information, robust or otherwise, to formulate an accurate and cogent response. So let’s state what we know based on the available evidence, recognizing that this will likely change, perhaps considerably, in the weeks and months ahead.
NIPT is a far superior approach, compared with conventional screening approaches, to screening for fetal trisomies 21, 18, and 13 in women carrying singleton pregnancies who are at an increased risk for fetal chromosome abnormalities.
In our current understanding of prenatal screening and diagnosis, NIPT does not provide either the comprehensive approach or the diagnostic accuracy associated with CVS and amniocentesis. As such, NIPT is not a suitable replacement for prenatal diagnostic procedures.
However, its application to screening a low-risk population for the common fetal aneuploidies, as well as in twin pregnancies, has been supported by initial studies, and the inclusion of other clinical outcomes—including other chromosome abnormalities, such as X and Y aneuploidies, trisomy 16, and triploidy10,11 and certain genomic abnormalities (eg, 22q deletions)—in the screening algorithm will expand the future clinical applications of NIPT screening.
DOES NIPT CHANGE OUR CONCEPTS OF SCREENING AND DIAGNOSIS?
This question is simple but profound and is perhaps the most important to be asked and addressed. Is a screening algorithm that has a similar sensitivity and specificity to that of CVS and amniocentesis for the most common fetal trisomies in the first and second trimesters sufficient to replace invasive testing for most women? Does the ability to detect fetal genomic abnormalities with microarray analyses of fetal cells obtained by CVS or amniocentesis provide a far greater benefit than that possible with any screening algorithm?
With renewed interest in the cost of health-care screening and diagnosis, we need to consider how comprehensive and accurate our prenatal screening and diagnostic tests should be and whether such improvements are desired or even possible from a clinical or economic viewpoint. In addition, the development of new technologies, such as the capture and analysis of fetal cells in maternal blood, presents the potential for a direct diagnostic fetal assay without the risks of an invasive procedure.
BIAS-FREE COUNSELING CANNOT BE OVERLOOKED
That being said, the current role of NIPT and other screening protocols in obstetric care needs to be clearly communicated to women who are considering their fetal assessment options, with emphasis placed on the capabilities and limitations of prenatal screening (even the newer ccfna-based options), the actual risks associated with invasive testing, and the ability of invasive testing to provide expanded fetal information with the use of microarray analyses.
As it has been from the beginning of prenatal testing in the 1960s, counseling continues to be the most important part of the prenatal screening and diagnostic process and it is needed to facilitate clinical decisions made by women and couples. Counseling must include an accurate communication of the risks, benefits, and limitations of the aforementioned options and issues, and should be provided in a manner that strives to be free of bias, direction, and the personal opinions of the counselor.
In order to provide such counseling, we must remain informed of the ongoing work in the field of prenatal testing, a task that has become more challenging with the rapid release of a considerable amount of new information on prenatal screening technologies over the past 2 years. This will likely continue, and perhaps become even more frenetic, with the expected release of additional information on the clinical applications of ccfna technologies in the near future as well as the development of new technologies applicable for the screening and diagnosis of fetal abnormalities.
WE WANT TO HEAR FROM YOU!
Share your thoughts on this article. Send your letter to: [email protected] Please include the city and state in which you practice.
The introduction of amniocentesis in the 1960s brought to prenatal diagnosticians the ability to detect fetal chromosome abnormalities and certain structural defects (including neural tube defects). Since that time, a goal for these practitioners has been the development of effective screening algorithms to better identify women at high risk for detectable fetal abnormalities in concert with the advent of safer and more accessible diagnostic tests, with the eventual aim being the development of a noninvasive prenatal diagnostic test.
Postamniocentesis advancements have included the identification of maternal serum analytes as well as the incorporation of first-trimester ultrasonographic measurements of the fetal nuchal translucency (NT) and nasal bone, all associated with an improved ability to identify women at increased risk for fetal trisomies 21 and 18 as well as some other fetal abnormalities. In addition, targeted ultrasound has greatly improved the ability to detect fetal structural and growth abnormalities in women of all risk levels, although it remains a highly subjective process with considerable inter/intraoperator and equipment variability.
Related article: NIPT is expanding rapidly--but don't throw out that CVS kit just yet! (Update on Obstetrics; Jaimey M. Pauli, MD, and John T. Repke, MD; January 2014)
Noninvasive prenatal screening has the advantages of being noninvasive and carrying no increased risk for fetal loss compared with chorionic villus sampling (CVS) and amniocentesis, which are associated with a small increased risk for pregnancy loss (1/500 to 1/1,500 over baseline risk for loss). However, noninvasive screening is limited compared with diagnostic procedures because it provides only a risk adjustment rather than a definitive diagnostic outcome and is mostly limited to assessment for fetal trisomies 18 and 21.
Targeted ultrasound can identify structural abnormalities associated with other chromosomal, genetic, and genomic abnormalities, but again depends on operator experience, equipment used, maternal habitus, and fetal position. Accordingly, considerable interest has remained in developing a more effective approach for detecting fetal aneuploidy and other fetal abnormalities, including assays that eventually could serve to provide noninvasive prenatal diagnosis.
RECENT ADVANCES BRING US CLOSER TO OUR ULTIMATE GOAL
The recent introduction of circulating cell-free nucleic acids (ccfna) technologies for prenatal screening for common fetal aneuploidies, better known as noninvasive prenatal testing, or NIPT, has presented a far more effective prenatal screening protocol for certain groups of women compared with the aforementioned screening algorithms that rely on measurements of the fetal NT in the late first trimester and maternal serum measurements of analytes in the first and second trimesters.
Currently, four NIPT screening products are available commercially in the United States: MaterniT21 Plus (Sequenom, San Diego, California); Verifi (Illumina, San Diego, California); Harmony Prenatal Test (Ariosa Diagnostics, San Jose, California); and Panorama Prenatal Test (Natera, San Carlos, California). While the technologies and algorithms used by each of the companies differ, they all rely on the premise that 5% to 10% of ccfna in maternal blood are fetal in nature.1 Calculating the ratios of the expected amount of each chromosome-specific nucleic acid to that actually measured in the sample, a prediction of a normal or abnormal complement for that specific chromosome is then made. None of the commercially available tests specifically identify fetal DNA or differentiate fetal from maternal DNA.
Current validation studies have thus far limited the offering of NIPT to women at increased risk for fetal aneuploidy, including those:2–6
- of advanced maternal age
- with a positive conventional screening test
- with abnormal ultrasound results suggestive of aneuploidy, or
- who have had a prior pregnancy with a chromosome aneuploidy found in the NIPT panel.
Studies of all available technologies tested on women at increased risk for fetal aneuploidy have thus far shown considerably higher sensitivities and specificities and detection rates for fetal trisomies 21, 18, and 13 than conventional screening algorithms, although detection rates for trisomy 13 are slightly lower than those observed for trisomies 21 and 18.
WE STILL HAVE MANY HURDLES TO LEAP
However, the groups of women at high risk for fetal aneuploidy just outlined represent only a small segment of the community of pregnant women. A multicenter study involving 1,914 women published February 2014 in the New England Journal of Medicine7 showed considerably and significantly lower false-positive rates and higher positive predictive values for the detection of trisomies 21 and 18 by NIPT compared with conventional fetal aneuploidy screening. This study incorporated women at low risk for fetal aneuploidies in the study cohort, although women at high risk (based on the stated range of maternal age) also were included in the cohort. Unfortunately, no information was provided in the report about the percentage of low-risk women among the study participants.
Related articles:
Noninvasive prenatal DNA testing: Who is using it, and how? Audiocast, June 2013
Noninvasive prenatal DNA tests are unproven and costly David A. Carpenter, MD (Comment & Controversy; September 2013)
Another concern about the published accuracy of NIPT clinical assays was recently sounded by Menutti and colleagues.8 The authors cited recent cases of positive NIPT outcomes for fetal trisomies 18 and 13 that were not confirmed by diagnostic testing of the pregnancies in question. The authors pondered whether such cases may reflect a limitation of the positive predictive values attributed to NIPT assays and that such limitations may carry profound inaccuracies in determining the accuracy of such protocols for rare aneuploidies.
While the improved detection rates for NIPT compared with conventional screening are not surprising, guidelines published by the American College of Obstetricians and Gynecologists still do not recommend the use of NIPT for the screening of low-risk women because of insufficient evaluation of ccfna technologies in the screening of such pregnancies.3 This also applies to twin pregnancies, despite preliminary studies showing comparable detection of trisomies 18 and 21 in such pregnancies compared with singleton pregnancies.3,9
There are no direct comparative studies of the four commercially available screening products, thus precluding a robust comparison and determination of the best existing method to use.
SO, WHERE ARE WE WITH NIPT EXACTLY?
The recent introduction of NIPT into routine obstetric care has left many clinicians with a wide range of questions, many of which cannot be answered because of little or no information, robust or otherwise, to formulate an accurate and cogent response. So let’s state what we know based on the available evidence, recognizing that this will likely change, perhaps considerably, in the weeks and months ahead.
NIPT is a far superior approach, compared with conventional screening approaches, to screening for fetal trisomies 21, 18, and 13 in women carrying singleton pregnancies who are at an increased risk for fetal chromosome abnormalities.
In our current understanding of prenatal screening and diagnosis, NIPT does not provide either the comprehensive approach or the diagnostic accuracy associated with CVS and amniocentesis. As such, NIPT is not a suitable replacement for prenatal diagnostic procedures.
However, its application to screening a low-risk population for the common fetal aneuploidies, as well as in twin pregnancies, has been supported by initial studies, and the inclusion of other clinical outcomes—including other chromosome abnormalities, such as X and Y aneuploidies, trisomy 16, and triploidy10,11 and certain genomic abnormalities (eg, 22q deletions)—in the screening algorithm will expand the future clinical applications of NIPT screening.
DOES NIPT CHANGE OUR CONCEPTS OF SCREENING AND DIAGNOSIS?
This question is simple but profound and is perhaps the most important to be asked and addressed. Is a screening algorithm that has a similar sensitivity and specificity to that of CVS and amniocentesis for the most common fetal trisomies in the first and second trimesters sufficient to replace invasive testing for most women? Does the ability to detect fetal genomic abnormalities with microarray analyses of fetal cells obtained by CVS or amniocentesis provide a far greater benefit than that possible with any screening algorithm?
With renewed interest in the cost of health-care screening and diagnosis, we need to consider how comprehensive and accurate our prenatal screening and diagnostic tests should be and whether such improvements are desired or even possible from a clinical or economic viewpoint. In addition, the development of new technologies, such as the capture and analysis of fetal cells in maternal blood, presents the potential for a direct diagnostic fetal assay without the risks of an invasive procedure.
BIAS-FREE COUNSELING CANNOT BE OVERLOOKED
That being said, the current role of NIPT and other screening protocols in obstetric care needs to be clearly communicated to women who are considering their fetal assessment options, with emphasis placed on the capabilities and limitations of prenatal screening (even the newer ccfna-based options), the actual risks associated with invasive testing, and the ability of invasive testing to provide expanded fetal information with the use of microarray analyses.
As it has been from the beginning of prenatal testing in the 1960s, counseling continues to be the most important part of the prenatal screening and diagnostic process and it is needed to facilitate clinical decisions made by women and couples. Counseling must include an accurate communication of the risks, benefits, and limitations of the aforementioned options and issues, and should be provided in a manner that strives to be free of bias, direction, and the personal opinions of the counselor.
In order to provide such counseling, we must remain informed of the ongoing work in the field of prenatal testing, a task that has become more challenging with the rapid release of a considerable amount of new information on prenatal screening technologies over the past 2 years. This will likely continue, and perhaps become even more frenetic, with the expected release of additional information on the clinical applications of ccfna technologies in the near future as well as the development of new technologies applicable for the screening and diagnosis of fetal abnormalities.
WE WANT TO HEAR FROM YOU!
Share your thoughts on this article. Send your letter to: [email protected] Please include the city and state in which you practice.
- Lo YM, Corbetta N, Chamberlain PF, et al. Presence of fetal DNA in maternal plasma and serum. Lancet. 1997;350(9076):485–487.
- Ashoor G, Syngelaki A, Wagner M, Birdir C, Nicolaides KH. Chromosome-selective sequencing of maternal plasma cell–free DNA for first-trimester detection of trisomy 21 and trisomy 18. Am J Obstet Gynecol. 2012;206(4):322.e1–e5.
- American College of Obstetricians and Gynecologists Committee on Genetics. Committee Opinion No. 545: Noninvasive prenatal testing for fetal aneuploidy. Obstet Gynecol. 2012;120(6):1532–1534.
- Bianchi DW, Platt LD, Goldberg JD, et al; MatErnal Blood IS Source to Accurately diagnose fetal aneuploidy (MELISSA) Study Group. Genome-wide fetal aneuploidy detection by maternal plasma DNA-sequencing. Obstet Gynecol. 2012;119(5):890–901.
- Palomaki GE, Kloza EM, Lambert-Messerlian GM, et al. DNA sequencing of maternal plasma to detect Down syndrome: An international clinical validation study. Genet Med. 2011;13(11):913–920.
- Palomaki GE, Deciu C, Kloza EM, et al. DNA sequencing of maternal plasma reliably identifies trisomy 18 and trisomy 13 as well as Down syndrome: An international collaborative study. Genet Med. 2012;14(3):296–305.
- Bianchi DW, Parker RL, Wentworth J, et al; CARE Study Group. DNA sequencing versus standard prenatal aneuploidy screening. N Engl J Med. 2014;370(9):799–808.
- Menutti MT, Cherry AM, Morrissette JJ, Dugoff L. Is it time to sound an alarm about false-positive cell-free DNA testing for fetal aneuploidy. Am J Obstet Gynecol. 2013;209(5):415−419.
- Canick JA, Kloza EM, Lambert-Messerlian GM, et al. DNA sequencing of maternal plasma to identify Down syndrome and other trisomies in multiple gestations. Prenat Diagn. 2012;32(8):730–734.
- Nicolaides KH, Syngelaki A, Gil MM, Quezada MS, Zinevich Y. Prenatal detection of fetal triploidy from cell-free DNA testing in maternal blood [published online ahead of print October 10, 2013]. Fetal Diagn Ther.
- Semango-Sprouse C, Banjevic M, Ryan A, et al. SNP-based non-invasive prenatal testing detects sex chromosome aneuploidies with high accuracy. Prenat Diagn. 2013;33(7):643–649.
- Lo YM, Corbetta N, Chamberlain PF, et al. Presence of fetal DNA in maternal plasma and serum. Lancet. 1997;350(9076):485–487.
- Ashoor G, Syngelaki A, Wagner M, Birdir C, Nicolaides KH. Chromosome-selective sequencing of maternal plasma cell–free DNA for first-trimester detection of trisomy 21 and trisomy 18. Am J Obstet Gynecol. 2012;206(4):322.e1–e5.
- American College of Obstetricians and Gynecologists Committee on Genetics. Committee Opinion No. 545: Noninvasive prenatal testing for fetal aneuploidy. Obstet Gynecol. 2012;120(6):1532–1534.
- Bianchi DW, Platt LD, Goldberg JD, et al; MatErnal Blood IS Source to Accurately diagnose fetal aneuploidy (MELISSA) Study Group. Genome-wide fetal aneuploidy detection by maternal plasma DNA-sequencing. Obstet Gynecol. 2012;119(5):890–901.
- Palomaki GE, Kloza EM, Lambert-Messerlian GM, et al. DNA sequencing of maternal plasma to detect Down syndrome: An international clinical validation study. Genet Med. 2011;13(11):913–920.
- Palomaki GE, Deciu C, Kloza EM, et al. DNA sequencing of maternal plasma reliably identifies trisomy 18 and trisomy 13 as well as Down syndrome: An international collaborative study. Genet Med. 2012;14(3):296–305.
- Bianchi DW, Parker RL, Wentworth J, et al; CARE Study Group. DNA sequencing versus standard prenatal aneuploidy screening. N Engl J Med. 2014;370(9):799–808.
- Menutti MT, Cherry AM, Morrissette JJ, Dugoff L. Is it time to sound an alarm about false-positive cell-free DNA testing for fetal aneuploidy. Am J Obstet Gynecol. 2013;209(5):415−419.
- Canick JA, Kloza EM, Lambert-Messerlian GM, et al. DNA sequencing of maternal plasma to identify Down syndrome and other trisomies in multiple gestations. Prenat Diagn. 2012;32(8):730–734.
- Nicolaides KH, Syngelaki A, Gil MM, Quezada MS, Zinevich Y. Prenatal detection of fetal triploidy from cell-free DNA testing in maternal blood [published online ahead of print October 10, 2013]. Fetal Diagn Ther.
- Semango-Sprouse C, Banjevic M, Ryan A, et al. SNP-based non-invasive prenatal testing detects sex chromosome aneuploidies with high accuracy. Prenat Diagn. 2013;33(7):643–649.
PAMG-1 is cost effective in questionable preterm PROM
CHICAGO – The placental alpha-microglobulin-1 test is superior from a cost-benefit perspective to the combined traditional diagnostic test of pooling, nitrazine, and ferning for screening in the setting of questionable preterm premature rupture of membranes, Dr. Nelson C. Echebiri reported at the annual meeting of the American Congress of Obstetricians and Gynecologists.
The PAMG-1 also should be used if the traditional test result is equivocal or inconclusive, he said.
The cost of PAMG-1 is about $85, compared with $14 for the traditional diagnostic test. Because of the added expense, testing all patients who present with possible preterm premature rupture of membranes (PROM) and those whose diagnosis is obvious would be counterproductive, Dr. Echebiri said.
The PAMG-1 test is easy to use and has been shown in multiple studies to have high sensitivity and specificity for detecting PAMG-1 – a marker for amniotic fluid. Dr. Echebiri said he felt the test would be a good noninvasive alternative to the invasive standard test of indigo carmine dye injection into the amniotic cavity.
For the study, a decision analysis was constructed to estimate the cost benefit of the PAMG-1 test as compared with the traditional test with respect to preterm delivery costs, which average $47,000. The analysis was based on the setting of potential preterm PROM at 34 weeks to less than 37 weeks of gestation as applied to 1,000 theoretical patients, 1,000 times each. Cost estimates reflected 2013 dollars.
When used according to the decision analysis, the estimated annual net benefit with the noninvasive placental alpha-microglobulin-1 (PAMG-1) test was $12,215/person tested, compared with $4,737/person tested using the traditional test. The PAMG-1 test was cost-beneficial in cases in which the probability of preterm PROM was less than 42% (net savings of $5,000-$32,000 per person tested vs. $5,000-$9,000 per person with the traditional test.
The PAMG-1 test was not associated with any savings in cases with 44% or greater probability of preterm PROM, said Dr. Echebiri of the University of Buffalo, N.Y., whose paper received a Donald F. Richardson Memorial Prize Paper Award as one of the best papers by a junior fellow at the meeting.
Dr. Echebiri reported having no disclosures.
CHICAGO – The placental alpha-microglobulin-1 test is superior from a cost-benefit perspective to the combined traditional diagnostic test of pooling, nitrazine, and ferning for screening in the setting of questionable preterm premature rupture of membranes, Dr. Nelson C. Echebiri reported at the annual meeting of the American Congress of Obstetricians and Gynecologists.
The PAMG-1 also should be used if the traditional test result is equivocal or inconclusive, he said.
The cost of PAMG-1 is about $85, compared with $14 for the traditional diagnostic test. Because of the added expense, testing all patients who present with possible preterm premature rupture of membranes (PROM) and those whose diagnosis is obvious would be counterproductive, Dr. Echebiri said.
The PAMG-1 test is easy to use and has been shown in multiple studies to have high sensitivity and specificity for detecting PAMG-1 – a marker for amniotic fluid. Dr. Echebiri said he felt the test would be a good noninvasive alternative to the invasive standard test of indigo carmine dye injection into the amniotic cavity.
For the study, a decision analysis was constructed to estimate the cost benefit of the PAMG-1 test as compared with the traditional test with respect to preterm delivery costs, which average $47,000. The analysis was based on the setting of potential preterm PROM at 34 weeks to less than 37 weeks of gestation as applied to 1,000 theoretical patients, 1,000 times each. Cost estimates reflected 2013 dollars.
When used according to the decision analysis, the estimated annual net benefit with the noninvasive placental alpha-microglobulin-1 (PAMG-1) test was $12,215/person tested, compared with $4,737/person tested using the traditional test. The PAMG-1 test was cost-beneficial in cases in which the probability of preterm PROM was less than 42% (net savings of $5,000-$32,000 per person tested vs. $5,000-$9,000 per person with the traditional test.
The PAMG-1 test was not associated with any savings in cases with 44% or greater probability of preterm PROM, said Dr. Echebiri of the University of Buffalo, N.Y., whose paper received a Donald F. Richardson Memorial Prize Paper Award as one of the best papers by a junior fellow at the meeting.
Dr. Echebiri reported having no disclosures.
CHICAGO – The placental alpha-microglobulin-1 test is superior from a cost-benefit perspective to the combined traditional diagnostic test of pooling, nitrazine, and ferning for screening in the setting of questionable preterm premature rupture of membranes, Dr. Nelson C. Echebiri reported at the annual meeting of the American Congress of Obstetricians and Gynecologists.
The PAMG-1 also should be used if the traditional test result is equivocal or inconclusive, he said.
The cost of PAMG-1 is about $85, compared with $14 for the traditional diagnostic test. Because of the added expense, testing all patients who present with possible preterm premature rupture of membranes (PROM) and those whose diagnosis is obvious would be counterproductive, Dr. Echebiri said.
The PAMG-1 test is easy to use and has been shown in multiple studies to have high sensitivity and specificity for detecting PAMG-1 – a marker for amniotic fluid. Dr. Echebiri said he felt the test would be a good noninvasive alternative to the invasive standard test of indigo carmine dye injection into the amniotic cavity.
For the study, a decision analysis was constructed to estimate the cost benefit of the PAMG-1 test as compared with the traditional test with respect to preterm delivery costs, which average $47,000. The analysis was based on the setting of potential preterm PROM at 34 weeks to less than 37 weeks of gestation as applied to 1,000 theoretical patients, 1,000 times each. Cost estimates reflected 2013 dollars.
When used according to the decision analysis, the estimated annual net benefit with the noninvasive placental alpha-microglobulin-1 (PAMG-1) test was $12,215/person tested, compared with $4,737/person tested using the traditional test. The PAMG-1 test was cost-beneficial in cases in which the probability of preterm PROM was less than 42% (net savings of $5,000-$32,000 per person tested vs. $5,000-$9,000 per person with the traditional test.
The PAMG-1 test was not associated with any savings in cases with 44% or greater probability of preterm PROM, said Dr. Echebiri of the University of Buffalo, N.Y., whose paper received a Donald F. Richardson Memorial Prize Paper Award as one of the best papers by a junior fellow at the meeting.
Dr. Echebiri reported having no disclosures.
AT THE ACOG ANNUAL CLINICAL MEETING
Does your obstetric unit have a protocol for treating amniotic fluid embolism?
Amniotic fluid embolism (AFE) occurs in about 1 in 20,000 to 1 in 40,000 deliveries.1,2 Although the condition is rare, the case fatality rate is high, and AFE is a common cause of maternal death in developed countries. AFE cannot be predicted or prevented. Moreover, the condition is difficult to precisely define and is often a diagnosis of exclusion.
AFE should be considered in the differential diagnosis of a pregnant woman with sudden onset of shortness of breath, hypotension, or cardiac arrhythmia or arrest, followed by coagulopathy and hemorrhage. Premonitory symptoms, including restlessness, confusion, disorientation, agitation, chills, nausea, numbness, and tingling, are commonly reported just before the cardiorespiratory collapse. AFE is less likely if the initial obstetric event is hemorrhage in the absence of cardiorespiratory compromise or a preceding coagulopathy.3
Typically, the onset is just before birth, during birth, or within the first few hours after delivery. In the United Kingdom, which has a robust centralized registry for reporting AFE, about 56% of cases occur before birth and 44% after birth.4
Related article: Is the incidence of amniotic fluid embolism rising? John T. Repke, MD (Examining the Evidence, August 2010)
The resources available to obstetric units vary greatly. Each unit needs to assess its resources and develop an AFE treatment protocol that builds on the unique strengths of the unit. Treatment of AFE requires the coordinated actions of anesthesiologists, obstetricians, nurses, the blood bank, pharmacy, and cardiovascular specialists. Coordinated activity among the members of such a large multidisciplinary team requires a written protocol that is practiced on a regular basis.
Six important components of a multidisciplinary response to AFE treatment protocol are:
- high-quality cardiopulmonary resuscitation (CPR)
- a protocol for massive transfusion
- treatment of diffuse bleeding and coagulopathy
- treatment of uterine and pelvic bleeding
- extracorporeal lung and heart support
- post-AFE intensive care.
1. Initiate high-quality CPR
Hypotension and hypoxemia due to cardiac and pulmonary dysfunction are prominent features of AFE. Dysrythmias such as pulseless electrical activity, bradycardia, ventricular fibrillation, and asystole are common. Rapid institution of high-quality CPR is critical to the survival of women with AFE.
Interventions often used in CPR of patients with AFE include initiation of high-quality chest compressions, early defibrillation if indicated, immediate administration of 100% oxygen by mask ventilation followed by early intubation, and rapid establishment of peripheral, arterial, and central venous access. Volume assessment, fluid replacement, and administration of vasopressors and inotropes are also important.
CPR of pregnant women requires special interventions, including maximal left lateral displacement of the uterus to reduce compression of the descending aorta and vena cava. Lateral displacement of the uterus can be accomplished by left lateral tilt or by manual uterine displacement. To optimize the effectiveness of chest compressions, many experts recommend placing the woman in a supine position and using manual uterine displacement rather than a left lateral tilt.5 For chest compressions, the hands should be placed just above the center of the sternum to adjust for the elevation of the diaphragm caused by the gravid uterus.
The gravid uterus can compromise the effectiveness of CPR. Fetal viability and neurologic outcome are best if delivery occurs within 5 minutes of the onset of cardiopulmonary arrest. If the gestational age of the fetus is consistent with extrauterine viability and initial CPR has not restored cardiac function, it is best to initiate fetal delivery within 4 minutes of the onset of cardiopulmonary arrest with the intent to deliver the fetus within 5 minutes.6,7 If the fetus is beyond 20 weeks’ gestational age, delivery early in the course of CPR improves the effectiveness of maternal resuscitation and may increase the probability of maternal survival.
In one study of the response of anesthesiologists, obstetricians, and nurses to a simulated cardiac arrest caused by an AFE, the participants did not routinely use defibrillation when indicated, did not place a firm support under the back for chest compressions, and did not switch the provider of chest compressions every 2 minutes.8 This study indicates that additional training and routinely scheduled multidisciplinary simulation of the response to cardiopulmonary arrest could improve the quality of our CPR.
2. Use a massive transfusion protocol
Severe coagulopathy and diffuse bleeding are commonly encountered in AFE. Target goals for the replacement of blood products include:
- hemoglobin concentration ≥8 g/dL
- fibrinogen ≥150 to 200 mg/dL
- platelets ≥50,000/μL
- prothrombin time international normalized ratio (INR) ≤1.5.
Most massive transfusion protocols provide for the rapid delivery of 4 to 8 units of red blood cells and a similar number of units of fresh frozen plasma to the patient’s bedside. In the management of AFE, 20 to 30 units of red blood cells and a similar quantity of fresh frozen plasma may need to be transfused. Cryoprecipitate takes 20 to 30 minutes to thaw, so preparations to transfuse cryoprecipitate should be initiated as soon as the massive transfusion protocol is triggered. A case of AFE can completely empty the blood bank of all available blood products and necessitate the use of alternative agents.
Lyophilized fibrinogen concentrate (RiaSTAP) is approved by the US Food and Drug Administration for the treatment of congenital hypofibrinogenemia and also may be useful to replace fibrinogen in cases of AFE. In many hospitals, large quantities of fresh frozen plasma are not immediately available; lyophilized fibrinogen concentrate may be especially useful in these settings. Another advantage of fibrinogen concentrate is that large amounts of fibrinogen can be administered in a small volume of intravenous fluid. Fibrinogen concentrate typically is used at a dose of 70 mg/kg of body weight.9,10
Intraoperative red cell salvage occasionally is used in cases of obstetric hemorrhage. In one case report of the use of red cell salvage with leukocyte depletion filtration during treatment of an AFE, acute hypotension developed in the patient after the transfusion of salvaged red cells.11 This case report raises safety concerns about the use of salvaged cells in women with severe AFE.
Related article: 10 practical, evidence-based recommendations for the management of severe postpartum hemorrhage Baha M. Sibai, MD (June 2011)
3. Treat diffuse bleeding and coagulopathy
In addition to the initiation of the massive transfusion protocol, additional treatments that may be helpful in managing the coagulopathy of AFE include tranexamic acid, recombinant factor VIIa (rFVIIa), and exchange transfusion.
AFE is often associated with hyperfibrinolysis, which can cause excessive bleeding.12 Tranexamic acid blocks the lysine binding sites on plasminogen and thereby reduces the lysis of fibrin clots. Clinical trials in patients who have undergone trauma have demonstrated that the administration of tranexamic acid reduces blood loss.13 The dose of tranexamic acid is approximately 10 to 20 mg/kg of body weight, or approximately 1 g.
Controversy exists about the use of rFVIIa to treat the coagulopathy and bleeding caused by AFE. Some authorities believe that rFVIIa is associated with an increased AFE case fatality rate.14 Other authorities believe rFVIIa may be useful in the treatment of AFE coagulopathy, especially when bleeding persists despite aggressive blood and component replacement.”15 The dose of rFVIIa is approximately 90 µg/kg of body weight. rFVIIa is extremely expensive.
Exchange transfusion has been used successfully to treat AFE.16 In women with AFE, exchange transfusion removes circulating cells, cell fragments, and substances that trigger systemic anaphylaxis and coagulopathy, thereby enhancing rapid recovery.
Related article: Act fast when confronted by a coagulopathy postpartum Robert L. Barbieri, MD (Editorial; March 2012)
4. Treat uterine and pelvic bleeding
Obstetrician-gynecologists are experts in the control of uterine and pelvic bleeding. Interventions that commonly are used to control uterine and pelvic bleeding in cases of postpartum hemorrhage, uterine rupture, or placenta accreta also can be applied in cases of AFE with uncontrolled uterine and pelvic bleeding. These techniques include:
- use of uterine compression sutures
- the Bakri balloon
- a uterine tourniquet
- vascular clamps on the ovarian vessels.17,18
In many cases of AFE, total or supracervical hysterectomy is necessary to control uterine bleeding. Uterine artery embolization, if available, has been reported to be helpful in select cases. However, many women with AFE are too unstable to survive transfer to an interventional radiology suite. Additional interventions to control bleeding include hypogastric artery ligation, infrarenal aortic compression, and pelvic packing.
Cross-clamping the aorta below the renal vessels can reduce blood flow to the pelvis and provide time for cardiopulmonary and volume resuscitation. Alternatively, placing pressure on the infrarenal aorta with a sponge or directly by hand can help reduce blood flow to the pelvis.19
In many cases of AFE, pelvic hemorrhage is difficult to control. Even if surgical pedicles are ligated securely, the coagulopathy of AFE may cause persistent oozing from areas of minor tissue trauma. Uncontrolled blood loss can be a proximate cause of death in women with AFE. All written protocols for responding to an AFE should include a plan to use pelvic packing for patients in whom standard operative procedures do not produce adequate control of bleeding. A “mushroom,” “parachute,” or “umbrella” pack has been reported to help stabilize the severely ill patient with pelvic bleeding and permit effective resuscitation and blood product replacement.20
Related articles:
A stitch in time: The B-Lynch, Hayman and Pereira uterine compression sutures Robert L. Barbieri, MD (Editorial, December 2012)
Have you made the best use of the Bakri balloon in PPH? Robert L. Barbieri, MD (Editorial, July 2011)
5. Consider extracorporeal lung and heart support
In many cases of AFE, both lung and cardiac function are severely compromised. Both veno-arterial extracorporeal membrane oxygenation (VA-ECMO) and full cardiopulmonary bypass provide support for the failing lung and heart. Based on a small number of case reports, extracorporeal lung and heart support appear to be useful in the treatment of AFE.21–26 Using the Seldinger technique,27 it is technically feasible to rapidly access a major vein and artery to provide the input and output ports for VA-ECMO. Unlike the cardiopulmonary bypass pump, the VA-ECMO pump does not have a reservoir that needs to be primed with blood and is smaller and more portable. To provide a patient with VA-ECMO or cardiopulmonary bypass, a cardiac interventionist and a perfusionist must be available. Extracorporeal lung and heart support require heparinization of the patient’s blood, which may result in increased bleeding. Both VA-ECMO and cardiopulmonary bypass, along with the diseases for which they are used, may cause renal dysfunction, neurologic injury, and infection.28
Alternative approaches that provide support of the heart—but not lung—are the Impella pump, TandemHeart, and the intra-aortic balloon pump. An alternative that provides lung support—but not cardiac support—is veno-venous ECMO.
In developing a written protocol for responding to an AFE, obstetricians should explore the potential availability of VA-ECMO, cardiopulmonary bypass, or other cardiopulmonary support devices as options for patients who have not responded to standard treatment of AFE and are at high risk of death.
6. Post-AFE intensive care
After stabilization, most women with AFE will require intensive care for 48 to 96 hours. Some experts have proposed that all survivors of cardiopulmonary arrest who are successfully resuscitated and stabilized be transferred to hospitals that specialize in post−cardiac arrest care to improve outcomes.
Assessment of organ injury is important after an AFE. In addition, encephalopathy is a common complication of AFE, and sequential neurologic examination is a priority. Therapeutic hypothermia (TH) may help to preserve neurologic function after AFE.29 However, TH may cause a mild coagulopathy by inhibiting platelet activation and enzyme activity of clotting factors. Because coagulopathy is a prominent feature of AFE, TH may be contraindicated if the patient has a clinically significant baseline coagulopathy.30
DEVELOP AN AFE PROTOCOL AND PRACTICE THE COMPONENTS
Practicing the components of obstetric protocols can improve unit performance and patient outcomes.31 The components of an AFE protocol, as described in this article, include high-quality CPR, a protocol for massive transfusion, treatment of diffuse bleeding and coagulopathy, treatment of uterine and pelvic bleeding, extracorporeal lung and heart support, and post-AFE intensive care. Practicing these components of an AFE protocol will enhance performance across many common obstetric complications including postpartum hemorrhage, uterine rupture, placenta accreta, and pulmonary embolism.
When Chesley “Sully” Sullenberger and his copilot landed Flight 1549 in the Hudson River in New York, he had never practiced that specific response to twin engine failure, but he had practiced many emergency responses involving related scenarios. The combination of exceptional flight experience and years of practicing the response to emergency scenarios in simulation exercises permitted him and his copilot to execute a uniquely clever plan to solve a life-threatening
emergency. In a related way, practicing the components of AFE treatment will help obstetricians, obstetric anesthesiologists, and their multidisciplinary team to improve the responses to all major obstetric emergencies.
INSTANT POLL
Does your obstetric unit have a written protocol for treating an amniotic fluid embolism (AFE)? Has your obstetric unit practiced any of the components of the AFE treatment protocol: 1) high-quality cardiopulmonary resuscitation, 2) a protocol for massive transfusion protocol, 3) treatment of diffuse bleeding and coagulopathy, 4) treatment of uterine and pelvic bleeding, 5) extracorporeal lung and heart support, and 6) post-AFE intensive care?
Tell us—at [email protected]. Please include your name and the city and state in which you practice.
- Kramer MS, Rouleau J, Baskett TF, Joseph KS; Maternal Health Study Group of the Canadian Perinatal Surveillance System. Amniotic-fluid embolism and medical induction of labour: A retrospective, population-based cohort study. Lancet. 2006;368(9545):1444–1448.
- Abenhaim HA, Azoulay L, Kramer MS, Leduc L. Incidence and risk factors of amniotic fluid embolism: A population-based study on 3 million births in the United States. Am J Obstet Gynecol. 2008;199(1):49.e1–49.e8.
- Tuffnell D, Knight M, Plaat F. Amniotic fluid embolism—An update. Anaesthesia. 2011;66(1):3–6.
- Knight M, Tuffnell D, Brocklehurst P, Spark P, Kurinczuk JJ; UK Obstetric Surveillance System. Incidence and risk factors for amniotic-fluid embolism. Obstet Gynecol. 2010;115(5):910–917.
- Kundra P, Khanna S, Habeebullahg S, Ravishankar M. Manual displacement of the uterus during Caesarean section. Anaesthesia. 2007;62(5):460–465.
- Katz VL, Dotters DJ, Droegemueller W. Perimortem cesarean delivery. Obstet Gynecol. 1986;68(4):571–576.
- Katz V, Balderston K, DeFreest M. Perimortem cesarean delivery: Were our assumptions correct? Am J Obstet Gynecol. 2005;192(6):1916–1920.
- Lipman SS, Daniels KI, Carvalho B, et al. Deficits in the provision of cardiopulmonary resuscitation during simulated obstetric crises. Am J Obstet Gynecol. 2010;203(2):179.e1–179.e5.
- Bell SF, Rayment R, Collins PW, Collis RE. The use of fibrinogen concentrate to correct hypofibrinogenaemia rapidly during obstetric haemorrhage. Int J Obstet Anesth. 2010;19(2):218–223.
- Sorensen B, Tang M, Larsen OH, Laursen PN, Fenger-Eriksen C, Rea CJ. The role of fibrinogen: A new paradigm in the treatment of coagulopathic bleeding. Thromb Res. 2011;128(Suppl 1):S13–S16.
- Rogers WK, Wernimont SA, Kumar GC, Bennett E, Chestnut DH. Acute hypotension associated with intraoperative cell salvage using a leukocyte depletion filter during management of obstetric hemorrhage due to amniotic fluid embolism. Anesth Analg. 2013;117(2):449–452.
- Collins NF, Bloor M, McDonnell NJ. Hyperfibrinolysis diagnosed by rotational thromboelastometry in a case of suspected amniotic fluid embolism. Int J Obstet Anesth. 2013;22(1):71–76.
- Ker K, Edwards P, Perel P, Shakur H, Roberts I. Effect of tranexamic acid on surgical bleeding: Systematic review and cumulative meta-analysis. BMJ. 2012;344:e3054.
- Leighton BL, Wall MH, Lockhart EM, Phillips LE, Zatta AJ. Use of recombinant factor VIIa in patients with amniotic fluid embolism: A systematic review of case reports. Anesthesiology. 2011;115(6):1201–1208.
- Huber AW, Raio L, Alberio L, Ghezzi F, Surbek DV. Recombinant human factor VIIa prevents hysterectomy in severe postpartum hemorrhage: single center study. J Perinat Med. 2011;40(1):43–49.
- Dodgson J, Martin J, Boswell J, Goodall HB, Smith R. Probable amniotic fluid embolism precipitated by amniocentesis and treated by exchange transfusion. Brit Med J (Clin Res Ed). 1987;294(6583):1322–1323.
- Barbieri RL. A stitch in time: The B-Lynch, Hayman and Pereira uterine compression sutures. OBG Manage. 2012;24(12):6, 8, 10, 11.
- Barbieri RL. Have you made the best use of the Bakri balloon in PPH? OBG Manage. 2011;23(7):6, 8, 9.
- Belfort MA, Zimmerman J, Schemmer G, Oldroyd R, Smilanich R, Pearce M. Aortic compression and cross clamping in a case of placenta percreta and amniotic fluid embolism: A case report. AJP Rep. 2011;1(1):33–36.
- Dildy GA, Scott JR, Saffer CS, Belfort MA. An
effective pressure pack for severe pelvic hemorrhage. Obstet Gynecol. 2006;108(5):1222–1226. - Stanten RD, Iverson LI, Daugharty TM, Lovett SM, Terry C, Blumenstock E. Amniotic fluid embolism causing catastrophic pulmonary vasoconstriction: Diagnosis by transesophageal echocardiogram and treatment by cardiopulmonary bypass. Obstet Gynecol. 2003;102(3):496–498.
- Ho CH, Chen KB, Liu SK, Liu YF, Cheng HC, Wu RS. Early application of extracorporeal membrane oxygenation in a patient with amniotic fluid embolism. Acta Anaesthesiol Taiwan. 2009;47(2):99–102.
- Shen HP, Chang WC, Yeh LS, Ho M. Amniotic fluid embolism treated with emergency extracorporeal membrane oxygenation: A case report.
J Reprod Med. 2009;54(11–12):706–708. - Lee PH, Shulman MS, Vellayappan U, Symes JF, Olenchock SA Jr. Surgical treatment of amniotic fluid embolism with cardiopulmonary collapse. Ann Thorac Surg. 2010;90(5):1694–1696.
- Firstenberg MS, Abel E, Blais D, et al. Temporary extracorporeal circulatory support and pulmonary embolectomy for catastrophic amniotic fluid embolism. Heart Surg Forum. 2011;14(3):E157–E159.
- Ecker JL, Solt K, Fitzsimons MG, MacGillivray TE. Case records of the Massachusetts General Hospital. Case 40-2012: A 43-year-old woman with cardiorespiratory arrest after a cesarean section. N Engl J Med. 2012;367(26):2528–2536.
- Seldinger SI. Catheter replacement of the needle in percutaneous arteriography; a new technique. Acta Radiol. 1953;39(5):368–376.
- Cheng R, Hachamovitch R, Kittelson M, et al. Complications of extracorporeal membrane oxygenation for treatment of cardiogenic shock and cardiac arrest: A meta-analysis of 1,866 adult patients. Ann Thorac Surg. 2013; epub Nov 8.
- Rittenberger JC, Kelly E, Jang D, Greer K, Heffner A. Successful outcome utilizing hypothermia after cardiac arrest in pregnancy: A case report. Crit Care Med. 2008;36(4):1354–1356.
- Michelson AD, MacGregor H, Barnard MR, Krestin AS, Rohrer MJ, Valeri CR. Reversible inhibition of human platelet activation by hypothermia in vivo and in vitro. Thromb Haemost. 1994;71(5):633–640.
- Rivzi F, Mackey R, Barrett T, McKenna P, Geary M. Successful reduction of massive postpartum haemorrhage by use of guidelines and staff education. BJOG. 2004;111(5):495–498.
Amniotic fluid embolism (AFE) occurs in about 1 in 20,000 to 1 in 40,000 deliveries.1,2 Although the condition is rare, the case fatality rate is high, and AFE is a common cause of maternal death in developed countries. AFE cannot be predicted or prevented. Moreover, the condition is difficult to precisely define and is often a diagnosis of exclusion.
AFE should be considered in the differential diagnosis of a pregnant woman with sudden onset of shortness of breath, hypotension, or cardiac arrhythmia or arrest, followed by coagulopathy and hemorrhage. Premonitory symptoms, including restlessness, confusion, disorientation, agitation, chills, nausea, numbness, and tingling, are commonly reported just before the cardiorespiratory collapse. AFE is less likely if the initial obstetric event is hemorrhage in the absence of cardiorespiratory compromise or a preceding coagulopathy.3
Typically, the onset is just before birth, during birth, or within the first few hours after delivery. In the United Kingdom, which has a robust centralized registry for reporting AFE, about 56% of cases occur before birth and 44% after birth.4
Related article: Is the incidence of amniotic fluid embolism rising? John T. Repke, MD (Examining the Evidence, August 2010)
The resources available to obstetric units vary greatly. Each unit needs to assess its resources and develop an AFE treatment protocol that builds on the unique strengths of the unit. Treatment of AFE requires the coordinated actions of anesthesiologists, obstetricians, nurses, the blood bank, pharmacy, and cardiovascular specialists. Coordinated activity among the members of such a large multidisciplinary team requires a written protocol that is practiced on a regular basis.
Six important components of a multidisciplinary response to AFE treatment protocol are:
- high-quality cardiopulmonary resuscitation (CPR)
- a protocol for massive transfusion
- treatment of diffuse bleeding and coagulopathy
- treatment of uterine and pelvic bleeding
- extracorporeal lung and heart support
- post-AFE intensive care.
1. Initiate high-quality CPR
Hypotension and hypoxemia due to cardiac and pulmonary dysfunction are prominent features of AFE. Dysrythmias such as pulseless electrical activity, bradycardia, ventricular fibrillation, and asystole are common. Rapid institution of high-quality CPR is critical to the survival of women with AFE.
Interventions often used in CPR of patients with AFE include initiation of high-quality chest compressions, early defibrillation if indicated, immediate administration of 100% oxygen by mask ventilation followed by early intubation, and rapid establishment of peripheral, arterial, and central venous access. Volume assessment, fluid replacement, and administration of vasopressors and inotropes are also important.
CPR of pregnant women requires special interventions, including maximal left lateral displacement of the uterus to reduce compression of the descending aorta and vena cava. Lateral displacement of the uterus can be accomplished by left lateral tilt or by manual uterine displacement. To optimize the effectiveness of chest compressions, many experts recommend placing the woman in a supine position and using manual uterine displacement rather than a left lateral tilt.5 For chest compressions, the hands should be placed just above the center of the sternum to adjust for the elevation of the diaphragm caused by the gravid uterus.
The gravid uterus can compromise the effectiveness of CPR. Fetal viability and neurologic outcome are best if delivery occurs within 5 minutes of the onset of cardiopulmonary arrest. If the gestational age of the fetus is consistent with extrauterine viability and initial CPR has not restored cardiac function, it is best to initiate fetal delivery within 4 minutes of the onset of cardiopulmonary arrest with the intent to deliver the fetus within 5 minutes.6,7 If the fetus is beyond 20 weeks’ gestational age, delivery early in the course of CPR improves the effectiveness of maternal resuscitation and may increase the probability of maternal survival.
In one study of the response of anesthesiologists, obstetricians, and nurses to a simulated cardiac arrest caused by an AFE, the participants did not routinely use defibrillation when indicated, did not place a firm support under the back for chest compressions, and did not switch the provider of chest compressions every 2 minutes.8 This study indicates that additional training and routinely scheduled multidisciplinary simulation of the response to cardiopulmonary arrest could improve the quality of our CPR.
2. Use a massive transfusion protocol
Severe coagulopathy and diffuse bleeding are commonly encountered in AFE. Target goals for the replacement of blood products include:
- hemoglobin concentration ≥8 g/dL
- fibrinogen ≥150 to 200 mg/dL
- platelets ≥50,000/μL
- prothrombin time international normalized ratio (INR) ≤1.5.
Most massive transfusion protocols provide for the rapid delivery of 4 to 8 units of red blood cells and a similar number of units of fresh frozen plasma to the patient’s bedside. In the management of AFE, 20 to 30 units of red blood cells and a similar quantity of fresh frozen plasma may need to be transfused. Cryoprecipitate takes 20 to 30 minutes to thaw, so preparations to transfuse cryoprecipitate should be initiated as soon as the massive transfusion protocol is triggered. A case of AFE can completely empty the blood bank of all available blood products and necessitate the use of alternative agents.
Lyophilized fibrinogen concentrate (RiaSTAP) is approved by the US Food and Drug Administration for the treatment of congenital hypofibrinogenemia and also may be useful to replace fibrinogen in cases of AFE. In many hospitals, large quantities of fresh frozen plasma are not immediately available; lyophilized fibrinogen concentrate may be especially useful in these settings. Another advantage of fibrinogen concentrate is that large amounts of fibrinogen can be administered in a small volume of intravenous fluid. Fibrinogen concentrate typically is used at a dose of 70 mg/kg of body weight.9,10
Intraoperative red cell salvage occasionally is used in cases of obstetric hemorrhage. In one case report of the use of red cell salvage with leukocyte depletion filtration during treatment of an AFE, acute hypotension developed in the patient after the transfusion of salvaged red cells.11 This case report raises safety concerns about the use of salvaged cells in women with severe AFE.
Related article: 10 practical, evidence-based recommendations for the management of severe postpartum hemorrhage Baha M. Sibai, MD (June 2011)
3. Treat diffuse bleeding and coagulopathy
In addition to the initiation of the massive transfusion protocol, additional treatments that may be helpful in managing the coagulopathy of AFE include tranexamic acid, recombinant factor VIIa (rFVIIa), and exchange transfusion.
AFE is often associated with hyperfibrinolysis, which can cause excessive bleeding.12 Tranexamic acid blocks the lysine binding sites on plasminogen and thereby reduces the lysis of fibrin clots. Clinical trials in patients who have undergone trauma have demonstrated that the administration of tranexamic acid reduces blood loss.13 The dose of tranexamic acid is approximately 10 to 20 mg/kg of body weight, or approximately 1 g.
Controversy exists about the use of rFVIIa to treat the coagulopathy and bleeding caused by AFE. Some authorities believe that rFVIIa is associated with an increased AFE case fatality rate.14 Other authorities believe rFVIIa may be useful in the treatment of AFE coagulopathy, especially when bleeding persists despite aggressive blood and component replacement.”15 The dose of rFVIIa is approximately 90 µg/kg of body weight. rFVIIa is extremely expensive.
Exchange transfusion has been used successfully to treat AFE.16 In women with AFE, exchange transfusion removes circulating cells, cell fragments, and substances that trigger systemic anaphylaxis and coagulopathy, thereby enhancing rapid recovery.
Related article: Act fast when confronted by a coagulopathy postpartum Robert L. Barbieri, MD (Editorial; March 2012)
4. Treat uterine and pelvic bleeding
Obstetrician-gynecologists are experts in the control of uterine and pelvic bleeding. Interventions that commonly are used to control uterine and pelvic bleeding in cases of postpartum hemorrhage, uterine rupture, or placenta accreta also can be applied in cases of AFE with uncontrolled uterine and pelvic bleeding. These techniques include:
- use of uterine compression sutures
- the Bakri balloon
- a uterine tourniquet
- vascular clamps on the ovarian vessels.17,18
In many cases of AFE, total or supracervical hysterectomy is necessary to control uterine bleeding. Uterine artery embolization, if available, has been reported to be helpful in select cases. However, many women with AFE are too unstable to survive transfer to an interventional radiology suite. Additional interventions to control bleeding include hypogastric artery ligation, infrarenal aortic compression, and pelvic packing.
Cross-clamping the aorta below the renal vessels can reduce blood flow to the pelvis and provide time for cardiopulmonary and volume resuscitation. Alternatively, placing pressure on the infrarenal aorta with a sponge or directly by hand can help reduce blood flow to the pelvis.19
In many cases of AFE, pelvic hemorrhage is difficult to control. Even if surgical pedicles are ligated securely, the coagulopathy of AFE may cause persistent oozing from areas of minor tissue trauma. Uncontrolled blood loss can be a proximate cause of death in women with AFE. All written protocols for responding to an AFE should include a plan to use pelvic packing for patients in whom standard operative procedures do not produce adequate control of bleeding. A “mushroom,” “parachute,” or “umbrella” pack has been reported to help stabilize the severely ill patient with pelvic bleeding and permit effective resuscitation and blood product replacement.20
Related articles:
A stitch in time: The B-Lynch, Hayman and Pereira uterine compression sutures Robert L. Barbieri, MD (Editorial, December 2012)
Have you made the best use of the Bakri balloon in PPH? Robert L. Barbieri, MD (Editorial, July 2011)
5. Consider extracorporeal lung and heart support
In many cases of AFE, both lung and cardiac function are severely compromised. Both veno-arterial extracorporeal membrane oxygenation (VA-ECMO) and full cardiopulmonary bypass provide support for the failing lung and heart. Based on a small number of case reports, extracorporeal lung and heart support appear to be useful in the treatment of AFE.21–26 Using the Seldinger technique,27 it is technically feasible to rapidly access a major vein and artery to provide the input and output ports for VA-ECMO. Unlike the cardiopulmonary bypass pump, the VA-ECMO pump does not have a reservoir that needs to be primed with blood and is smaller and more portable. To provide a patient with VA-ECMO or cardiopulmonary bypass, a cardiac interventionist and a perfusionist must be available. Extracorporeal lung and heart support require heparinization of the patient’s blood, which may result in increased bleeding. Both VA-ECMO and cardiopulmonary bypass, along with the diseases for which they are used, may cause renal dysfunction, neurologic injury, and infection.28
Alternative approaches that provide support of the heart—but not lung—are the Impella pump, TandemHeart, and the intra-aortic balloon pump. An alternative that provides lung support—but not cardiac support—is veno-venous ECMO.
In developing a written protocol for responding to an AFE, obstetricians should explore the potential availability of VA-ECMO, cardiopulmonary bypass, or other cardiopulmonary support devices as options for patients who have not responded to standard treatment of AFE and are at high risk of death.
6. Post-AFE intensive care
After stabilization, most women with AFE will require intensive care for 48 to 96 hours. Some experts have proposed that all survivors of cardiopulmonary arrest who are successfully resuscitated and stabilized be transferred to hospitals that specialize in post−cardiac arrest care to improve outcomes.
Assessment of organ injury is important after an AFE. In addition, encephalopathy is a common complication of AFE, and sequential neurologic examination is a priority. Therapeutic hypothermia (TH) may help to preserve neurologic function after AFE.29 However, TH may cause a mild coagulopathy by inhibiting platelet activation and enzyme activity of clotting factors. Because coagulopathy is a prominent feature of AFE, TH may be contraindicated if the patient has a clinically significant baseline coagulopathy.30
DEVELOP AN AFE PROTOCOL AND PRACTICE THE COMPONENTS
Practicing the components of obstetric protocols can improve unit performance and patient outcomes.31 The components of an AFE protocol, as described in this article, include high-quality CPR, a protocol for massive transfusion, treatment of diffuse bleeding and coagulopathy, treatment of uterine and pelvic bleeding, extracorporeal lung and heart support, and post-AFE intensive care. Practicing these components of an AFE protocol will enhance performance across many common obstetric complications including postpartum hemorrhage, uterine rupture, placenta accreta, and pulmonary embolism.
When Chesley “Sully” Sullenberger and his copilot landed Flight 1549 in the Hudson River in New York, he had never practiced that specific response to twin engine failure, but he had practiced many emergency responses involving related scenarios. The combination of exceptional flight experience and years of practicing the response to emergency scenarios in simulation exercises permitted him and his copilot to execute a uniquely clever plan to solve a life-threatening
emergency. In a related way, practicing the components of AFE treatment will help obstetricians, obstetric anesthesiologists, and their multidisciplinary team to improve the responses to all major obstetric emergencies.
INSTANT POLL
Does your obstetric unit have a written protocol for treating an amniotic fluid embolism (AFE)? Has your obstetric unit practiced any of the components of the AFE treatment protocol: 1) high-quality cardiopulmonary resuscitation, 2) a protocol for massive transfusion protocol, 3) treatment of diffuse bleeding and coagulopathy, 4) treatment of uterine and pelvic bleeding, 5) extracorporeal lung and heart support, and 6) post-AFE intensive care?
Tell us—at [email protected]. Please include your name and the city and state in which you practice.
Amniotic fluid embolism (AFE) occurs in about 1 in 20,000 to 1 in 40,000 deliveries.1,2 Although the condition is rare, the case fatality rate is high, and AFE is a common cause of maternal death in developed countries. AFE cannot be predicted or prevented. Moreover, the condition is difficult to precisely define and is often a diagnosis of exclusion.
AFE should be considered in the differential diagnosis of a pregnant woman with sudden onset of shortness of breath, hypotension, or cardiac arrhythmia or arrest, followed by coagulopathy and hemorrhage. Premonitory symptoms, including restlessness, confusion, disorientation, agitation, chills, nausea, numbness, and tingling, are commonly reported just before the cardiorespiratory collapse. AFE is less likely if the initial obstetric event is hemorrhage in the absence of cardiorespiratory compromise or a preceding coagulopathy.3
Typically, the onset is just before birth, during birth, or within the first few hours after delivery. In the United Kingdom, which has a robust centralized registry for reporting AFE, about 56% of cases occur before birth and 44% after birth.4
Related article: Is the incidence of amniotic fluid embolism rising? John T. Repke, MD (Examining the Evidence, August 2010)
The resources available to obstetric units vary greatly. Each unit needs to assess its resources and develop an AFE treatment protocol that builds on the unique strengths of the unit. Treatment of AFE requires the coordinated actions of anesthesiologists, obstetricians, nurses, the blood bank, pharmacy, and cardiovascular specialists. Coordinated activity among the members of such a large multidisciplinary team requires a written protocol that is practiced on a regular basis.
Six important components of a multidisciplinary response to AFE treatment protocol are:
- high-quality cardiopulmonary resuscitation (CPR)
- a protocol for massive transfusion
- treatment of diffuse bleeding and coagulopathy
- treatment of uterine and pelvic bleeding
- extracorporeal lung and heart support
- post-AFE intensive care.
1. Initiate high-quality CPR
Hypotension and hypoxemia due to cardiac and pulmonary dysfunction are prominent features of AFE. Dysrythmias such as pulseless electrical activity, bradycardia, ventricular fibrillation, and asystole are common. Rapid institution of high-quality CPR is critical to the survival of women with AFE.
Interventions often used in CPR of patients with AFE include initiation of high-quality chest compressions, early defibrillation if indicated, immediate administration of 100% oxygen by mask ventilation followed by early intubation, and rapid establishment of peripheral, arterial, and central venous access. Volume assessment, fluid replacement, and administration of vasopressors and inotropes are also important.
CPR of pregnant women requires special interventions, including maximal left lateral displacement of the uterus to reduce compression of the descending aorta and vena cava. Lateral displacement of the uterus can be accomplished by left lateral tilt or by manual uterine displacement. To optimize the effectiveness of chest compressions, many experts recommend placing the woman in a supine position and using manual uterine displacement rather than a left lateral tilt.5 For chest compressions, the hands should be placed just above the center of the sternum to adjust for the elevation of the diaphragm caused by the gravid uterus.
The gravid uterus can compromise the effectiveness of CPR. Fetal viability and neurologic outcome are best if delivery occurs within 5 minutes of the onset of cardiopulmonary arrest. If the gestational age of the fetus is consistent with extrauterine viability and initial CPR has not restored cardiac function, it is best to initiate fetal delivery within 4 minutes of the onset of cardiopulmonary arrest with the intent to deliver the fetus within 5 minutes.6,7 If the fetus is beyond 20 weeks’ gestational age, delivery early in the course of CPR improves the effectiveness of maternal resuscitation and may increase the probability of maternal survival.
In one study of the response of anesthesiologists, obstetricians, and nurses to a simulated cardiac arrest caused by an AFE, the participants did not routinely use defibrillation when indicated, did not place a firm support under the back for chest compressions, and did not switch the provider of chest compressions every 2 minutes.8 This study indicates that additional training and routinely scheduled multidisciplinary simulation of the response to cardiopulmonary arrest could improve the quality of our CPR.
2. Use a massive transfusion protocol
Severe coagulopathy and diffuse bleeding are commonly encountered in AFE. Target goals for the replacement of blood products include:
- hemoglobin concentration ≥8 g/dL
- fibrinogen ≥150 to 200 mg/dL
- platelets ≥50,000/μL
- prothrombin time international normalized ratio (INR) ≤1.5.
Most massive transfusion protocols provide for the rapid delivery of 4 to 8 units of red blood cells and a similar number of units of fresh frozen plasma to the patient’s bedside. In the management of AFE, 20 to 30 units of red blood cells and a similar quantity of fresh frozen plasma may need to be transfused. Cryoprecipitate takes 20 to 30 minutes to thaw, so preparations to transfuse cryoprecipitate should be initiated as soon as the massive transfusion protocol is triggered. A case of AFE can completely empty the blood bank of all available blood products and necessitate the use of alternative agents.
Lyophilized fibrinogen concentrate (RiaSTAP) is approved by the US Food and Drug Administration for the treatment of congenital hypofibrinogenemia and also may be useful to replace fibrinogen in cases of AFE. In many hospitals, large quantities of fresh frozen plasma are not immediately available; lyophilized fibrinogen concentrate may be especially useful in these settings. Another advantage of fibrinogen concentrate is that large amounts of fibrinogen can be administered in a small volume of intravenous fluid. Fibrinogen concentrate typically is used at a dose of 70 mg/kg of body weight.9,10
Intraoperative red cell salvage occasionally is used in cases of obstetric hemorrhage. In one case report of the use of red cell salvage with leukocyte depletion filtration during treatment of an AFE, acute hypotension developed in the patient after the transfusion of salvaged red cells.11 This case report raises safety concerns about the use of salvaged cells in women with severe AFE.
Related article: 10 practical, evidence-based recommendations for the management of severe postpartum hemorrhage Baha M. Sibai, MD (June 2011)
3. Treat diffuse bleeding and coagulopathy
In addition to the initiation of the massive transfusion protocol, additional treatments that may be helpful in managing the coagulopathy of AFE include tranexamic acid, recombinant factor VIIa (rFVIIa), and exchange transfusion.
AFE is often associated with hyperfibrinolysis, which can cause excessive bleeding.12 Tranexamic acid blocks the lysine binding sites on plasminogen and thereby reduces the lysis of fibrin clots. Clinical trials in patients who have undergone trauma have demonstrated that the administration of tranexamic acid reduces blood loss.13 The dose of tranexamic acid is approximately 10 to 20 mg/kg of body weight, or approximately 1 g.
Controversy exists about the use of rFVIIa to treat the coagulopathy and bleeding caused by AFE. Some authorities believe that rFVIIa is associated with an increased AFE case fatality rate.14 Other authorities believe rFVIIa may be useful in the treatment of AFE coagulopathy, especially when bleeding persists despite aggressive blood and component replacement.”15 The dose of rFVIIa is approximately 90 µg/kg of body weight. rFVIIa is extremely expensive.
Exchange transfusion has been used successfully to treat AFE.16 In women with AFE, exchange transfusion removes circulating cells, cell fragments, and substances that trigger systemic anaphylaxis and coagulopathy, thereby enhancing rapid recovery.
Related article: Act fast when confronted by a coagulopathy postpartum Robert L. Barbieri, MD (Editorial; March 2012)
4. Treat uterine and pelvic bleeding
Obstetrician-gynecologists are experts in the control of uterine and pelvic bleeding. Interventions that commonly are used to control uterine and pelvic bleeding in cases of postpartum hemorrhage, uterine rupture, or placenta accreta also can be applied in cases of AFE with uncontrolled uterine and pelvic bleeding. These techniques include:
- use of uterine compression sutures
- the Bakri balloon
- a uterine tourniquet
- vascular clamps on the ovarian vessels.17,18
In many cases of AFE, total or supracervical hysterectomy is necessary to control uterine bleeding. Uterine artery embolization, if available, has been reported to be helpful in select cases. However, many women with AFE are too unstable to survive transfer to an interventional radiology suite. Additional interventions to control bleeding include hypogastric artery ligation, infrarenal aortic compression, and pelvic packing.
Cross-clamping the aorta below the renal vessels can reduce blood flow to the pelvis and provide time for cardiopulmonary and volume resuscitation. Alternatively, placing pressure on the infrarenal aorta with a sponge or directly by hand can help reduce blood flow to the pelvis.19
In many cases of AFE, pelvic hemorrhage is difficult to control. Even if surgical pedicles are ligated securely, the coagulopathy of AFE may cause persistent oozing from areas of minor tissue trauma. Uncontrolled blood loss can be a proximate cause of death in women with AFE. All written protocols for responding to an AFE should include a plan to use pelvic packing for patients in whom standard operative procedures do not produce adequate control of bleeding. A “mushroom,” “parachute,” or “umbrella” pack has been reported to help stabilize the severely ill patient with pelvic bleeding and permit effective resuscitation and blood product replacement.20
Related articles:
A stitch in time: The B-Lynch, Hayman and Pereira uterine compression sutures Robert L. Barbieri, MD (Editorial, December 2012)
Have you made the best use of the Bakri balloon in PPH? Robert L. Barbieri, MD (Editorial, July 2011)
5. Consider extracorporeal lung and heart support
In many cases of AFE, both lung and cardiac function are severely compromised. Both veno-arterial extracorporeal membrane oxygenation (VA-ECMO) and full cardiopulmonary bypass provide support for the failing lung and heart. Based on a small number of case reports, extracorporeal lung and heart support appear to be useful in the treatment of AFE.21–26 Using the Seldinger technique,27 it is technically feasible to rapidly access a major vein and artery to provide the input and output ports for VA-ECMO. Unlike the cardiopulmonary bypass pump, the VA-ECMO pump does not have a reservoir that needs to be primed with blood and is smaller and more portable. To provide a patient with VA-ECMO or cardiopulmonary bypass, a cardiac interventionist and a perfusionist must be available. Extracorporeal lung and heart support require heparinization of the patient’s blood, which may result in increased bleeding. Both VA-ECMO and cardiopulmonary bypass, along with the diseases for which they are used, may cause renal dysfunction, neurologic injury, and infection.28
Alternative approaches that provide support of the heart—but not lung—are the Impella pump, TandemHeart, and the intra-aortic balloon pump. An alternative that provides lung support—but not cardiac support—is veno-venous ECMO.
In developing a written protocol for responding to an AFE, obstetricians should explore the potential availability of VA-ECMO, cardiopulmonary bypass, or other cardiopulmonary support devices as options for patients who have not responded to standard treatment of AFE and are at high risk of death.
6. Post-AFE intensive care
After stabilization, most women with AFE will require intensive care for 48 to 96 hours. Some experts have proposed that all survivors of cardiopulmonary arrest who are successfully resuscitated and stabilized be transferred to hospitals that specialize in post−cardiac arrest care to improve outcomes.
Assessment of organ injury is important after an AFE. In addition, encephalopathy is a common complication of AFE, and sequential neurologic examination is a priority. Therapeutic hypothermia (TH) may help to preserve neurologic function after AFE.29 However, TH may cause a mild coagulopathy by inhibiting platelet activation and enzyme activity of clotting factors. Because coagulopathy is a prominent feature of AFE, TH may be contraindicated if the patient has a clinically significant baseline coagulopathy.30
DEVELOP AN AFE PROTOCOL AND PRACTICE THE COMPONENTS
Practicing the components of obstetric protocols can improve unit performance and patient outcomes.31 The components of an AFE protocol, as described in this article, include high-quality CPR, a protocol for massive transfusion, treatment of diffuse bleeding and coagulopathy, treatment of uterine and pelvic bleeding, extracorporeal lung and heart support, and post-AFE intensive care. Practicing these components of an AFE protocol will enhance performance across many common obstetric complications including postpartum hemorrhage, uterine rupture, placenta accreta, and pulmonary embolism.
When Chesley “Sully” Sullenberger and his copilot landed Flight 1549 in the Hudson River in New York, he had never practiced that specific response to twin engine failure, but he had practiced many emergency responses involving related scenarios. The combination of exceptional flight experience and years of practicing the response to emergency scenarios in simulation exercises permitted him and his copilot to execute a uniquely clever plan to solve a life-threatening
emergency. In a related way, practicing the components of AFE treatment will help obstetricians, obstetric anesthesiologists, and their multidisciplinary team to improve the responses to all major obstetric emergencies.
INSTANT POLL
Does your obstetric unit have a written protocol for treating an amniotic fluid embolism (AFE)? Has your obstetric unit practiced any of the components of the AFE treatment protocol: 1) high-quality cardiopulmonary resuscitation, 2) a protocol for massive transfusion protocol, 3) treatment of diffuse bleeding and coagulopathy, 4) treatment of uterine and pelvic bleeding, 5) extracorporeal lung and heart support, and 6) post-AFE intensive care?
Tell us—at [email protected]. Please include your name and the city and state in which you practice.
- Kramer MS, Rouleau J, Baskett TF, Joseph KS; Maternal Health Study Group of the Canadian Perinatal Surveillance System. Amniotic-fluid embolism and medical induction of labour: A retrospective, population-based cohort study. Lancet. 2006;368(9545):1444–1448.
- Abenhaim HA, Azoulay L, Kramer MS, Leduc L. Incidence and risk factors of amniotic fluid embolism: A population-based study on 3 million births in the United States. Am J Obstet Gynecol. 2008;199(1):49.e1–49.e8.
- Tuffnell D, Knight M, Plaat F. Amniotic fluid embolism—An update. Anaesthesia. 2011;66(1):3–6.
- Knight M, Tuffnell D, Brocklehurst P, Spark P, Kurinczuk JJ; UK Obstetric Surveillance System. Incidence and risk factors for amniotic-fluid embolism. Obstet Gynecol. 2010;115(5):910–917.
- Kundra P, Khanna S, Habeebullahg S, Ravishankar M. Manual displacement of the uterus during Caesarean section. Anaesthesia. 2007;62(5):460–465.
- Katz VL, Dotters DJ, Droegemueller W. Perimortem cesarean delivery. Obstet Gynecol. 1986;68(4):571–576.
- Katz V, Balderston K, DeFreest M. Perimortem cesarean delivery: Were our assumptions correct? Am J Obstet Gynecol. 2005;192(6):1916–1920.
- Lipman SS, Daniels KI, Carvalho B, et al. Deficits in the provision of cardiopulmonary resuscitation during simulated obstetric crises. Am J Obstet Gynecol. 2010;203(2):179.e1–179.e5.
- Bell SF, Rayment R, Collins PW, Collis RE. The use of fibrinogen concentrate to correct hypofibrinogenaemia rapidly during obstetric haemorrhage. Int J Obstet Anesth. 2010;19(2):218–223.
- Sorensen B, Tang M, Larsen OH, Laursen PN, Fenger-Eriksen C, Rea CJ. The role of fibrinogen: A new paradigm in the treatment of coagulopathic bleeding. Thromb Res. 2011;128(Suppl 1):S13–S16.
- Rogers WK, Wernimont SA, Kumar GC, Bennett E, Chestnut DH. Acute hypotension associated with intraoperative cell salvage using a leukocyte depletion filter during management of obstetric hemorrhage due to amniotic fluid embolism. Anesth Analg. 2013;117(2):449–452.
- Collins NF, Bloor M, McDonnell NJ. Hyperfibrinolysis diagnosed by rotational thromboelastometry in a case of suspected amniotic fluid embolism. Int J Obstet Anesth. 2013;22(1):71–76.
- Ker K, Edwards P, Perel P, Shakur H, Roberts I. Effect of tranexamic acid on surgical bleeding: Systematic review and cumulative meta-analysis. BMJ. 2012;344:e3054.
- Leighton BL, Wall MH, Lockhart EM, Phillips LE, Zatta AJ. Use of recombinant factor VIIa in patients with amniotic fluid embolism: A systematic review of case reports. Anesthesiology. 2011;115(6):1201–1208.
- Huber AW, Raio L, Alberio L, Ghezzi F, Surbek DV. Recombinant human factor VIIa prevents hysterectomy in severe postpartum hemorrhage: single center study. J Perinat Med. 2011;40(1):43–49.
- Dodgson J, Martin J, Boswell J, Goodall HB, Smith R. Probable amniotic fluid embolism precipitated by amniocentesis and treated by exchange transfusion. Brit Med J (Clin Res Ed). 1987;294(6583):1322–1323.
- Barbieri RL. A stitch in time: The B-Lynch, Hayman and Pereira uterine compression sutures. OBG Manage. 2012;24(12):6, 8, 10, 11.
- Barbieri RL. Have you made the best use of the Bakri balloon in PPH? OBG Manage. 2011;23(7):6, 8, 9.
- Belfort MA, Zimmerman J, Schemmer G, Oldroyd R, Smilanich R, Pearce M. Aortic compression and cross clamping in a case of placenta percreta and amniotic fluid embolism: A case report. AJP Rep. 2011;1(1):33–36.
- Dildy GA, Scott JR, Saffer CS, Belfort MA. An
effective pressure pack for severe pelvic hemorrhage. Obstet Gynecol. 2006;108(5):1222–1226. - Stanten RD, Iverson LI, Daugharty TM, Lovett SM, Terry C, Blumenstock E. Amniotic fluid embolism causing catastrophic pulmonary vasoconstriction: Diagnosis by transesophageal echocardiogram and treatment by cardiopulmonary bypass. Obstet Gynecol. 2003;102(3):496–498.
- Ho CH, Chen KB, Liu SK, Liu YF, Cheng HC, Wu RS. Early application of extracorporeal membrane oxygenation in a patient with amniotic fluid embolism. Acta Anaesthesiol Taiwan. 2009;47(2):99–102.
- Shen HP, Chang WC, Yeh LS, Ho M. Amniotic fluid embolism treated with emergency extracorporeal membrane oxygenation: A case report.
J Reprod Med. 2009;54(11–12):706–708. - Lee PH, Shulman MS, Vellayappan U, Symes JF, Olenchock SA Jr. Surgical treatment of amniotic fluid embolism with cardiopulmonary collapse. Ann Thorac Surg. 2010;90(5):1694–1696.
- Firstenberg MS, Abel E, Blais D, et al. Temporary extracorporeal circulatory support and pulmonary embolectomy for catastrophic amniotic fluid embolism. Heart Surg Forum. 2011;14(3):E157–E159.
- Ecker JL, Solt K, Fitzsimons MG, MacGillivray TE. Case records of the Massachusetts General Hospital. Case 40-2012: A 43-year-old woman with cardiorespiratory arrest after a cesarean section. N Engl J Med. 2012;367(26):2528–2536.
- Seldinger SI. Catheter replacement of the needle in percutaneous arteriography; a new technique. Acta Radiol. 1953;39(5):368–376.
- Cheng R, Hachamovitch R, Kittelson M, et al. Complications of extracorporeal membrane oxygenation for treatment of cardiogenic shock and cardiac arrest: A meta-analysis of 1,866 adult patients. Ann Thorac Surg. 2013; epub Nov 8.
- Rittenberger JC, Kelly E, Jang D, Greer K, Heffner A. Successful outcome utilizing hypothermia after cardiac arrest in pregnancy: A case report. Crit Care Med. 2008;36(4):1354–1356.
- Michelson AD, MacGregor H, Barnard MR, Krestin AS, Rohrer MJ, Valeri CR. Reversible inhibition of human platelet activation by hypothermia in vivo and in vitro. Thromb Haemost. 1994;71(5):633–640.
- Rivzi F, Mackey R, Barrett T, McKenna P, Geary M. Successful reduction of massive postpartum haemorrhage by use of guidelines and staff education. BJOG. 2004;111(5):495–498.
- Kramer MS, Rouleau J, Baskett TF, Joseph KS; Maternal Health Study Group of the Canadian Perinatal Surveillance System. Amniotic-fluid embolism and medical induction of labour: A retrospective, population-based cohort study. Lancet. 2006;368(9545):1444–1448.
- Abenhaim HA, Azoulay L, Kramer MS, Leduc L. Incidence and risk factors of amniotic fluid embolism: A population-based study on 3 million births in the United States. Am J Obstet Gynecol. 2008;199(1):49.e1–49.e8.
- Tuffnell D, Knight M, Plaat F. Amniotic fluid embolism—An update. Anaesthesia. 2011;66(1):3–6.
- Knight M, Tuffnell D, Brocklehurst P, Spark P, Kurinczuk JJ; UK Obstetric Surveillance System. Incidence and risk factors for amniotic-fluid embolism. Obstet Gynecol. 2010;115(5):910–917.
- Kundra P, Khanna S, Habeebullahg S, Ravishankar M. Manual displacement of the uterus during Caesarean section. Anaesthesia. 2007;62(5):460–465.
- Katz VL, Dotters DJ, Droegemueller W. Perimortem cesarean delivery. Obstet Gynecol. 1986;68(4):571–576.
- Katz V, Balderston K, DeFreest M. Perimortem cesarean delivery: Were our assumptions correct? Am J Obstet Gynecol. 2005;192(6):1916–1920.
- Lipman SS, Daniels KI, Carvalho B, et al. Deficits in the provision of cardiopulmonary resuscitation during simulated obstetric crises. Am J Obstet Gynecol. 2010;203(2):179.e1–179.e5.
- Bell SF, Rayment R, Collins PW, Collis RE. The use of fibrinogen concentrate to correct hypofibrinogenaemia rapidly during obstetric haemorrhage. Int J Obstet Anesth. 2010;19(2):218–223.
- Sorensen B, Tang M, Larsen OH, Laursen PN, Fenger-Eriksen C, Rea CJ. The role of fibrinogen: A new paradigm in the treatment of coagulopathic bleeding. Thromb Res. 2011;128(Suppl 1):S13–S16.
- Rogers WK, Wernimont SA, Kumar GC, Bennett E, Chestnut DH. Acute hypotension associated with intraoperative cell salvage using a leukocyte depletion filter during management of obstetric hemorrhage due to amniotic fluid embolism. Anesth Analg. 2013;117(2):449–452.
- Collins NF, Bloor M, McDonnell NJ. Hyperfibrinolysis diagnosed by rotational thromboelastometry in a case of suspected amniotic fluid embolism. Int J Obstet Anesth. 2013;22(1):71–76.
- Ker K, Edwards P, Perel P, Shakur H, Roberts I. Effect of tranexamic acid on surgical bleeding: Systematic review and cumulative meta-analysis. BMJ. 2012;344:e3054.
- Leighton BL, Wall MH, Lockhart EM, Phillips LE, Zatta AJ. Use of recombinant factor VIIa in patients with amniotic fluid embolism: A systematic review of case reports. Anesthesiology. 2011;115(6):1201–1208.
- Huber AW, Raio L, Alberio L, Ghezzi F, Surbek DV. Recombinant human factor VIIa prevents hysterectomy in severe postpartum hemorrhage: single center study. J Perinat Med. 2011;40(1):43–49.
- Dodgson J, Martin J, Boswell J, Goodall HB, Smith R. Probable amniotic fluid embolism precipitated by amniocentesis and treated by exchange transfusion. Brit Med J (Clin Res Ed). 1987;294(6583):1322–1323.
- Barbieri RL. A stitch in time: The B-Lynch, Hayman and Pereira uterine compression sutures. OBG Manage. 2012;24(12):6, 8, 10, 11.
- Barbieri RL. Have you made the best use of the Bakri balloon in PPH? OBG Manage. 2011;23(7):6, 8, 9.
- Belfort MA, Zimmerman J, Schemmer G, Oldroyd R, Smilanich R, Pearce M. Aortic compression and cross clamping in a case of placenta percreta and amniotic fluid embolism: A case report. AJP Rep. 2011;1(1):33–36.
- Dildy GA, Scott JR, Saffer CS, Belfort MA. An
effective pressure pack for severe pelvic hemorrhage. Obstet Gynecol. 2006;108(5):1222–1226. - Stanten RD, Iverson LI, Daugharty TM, Lovett SM, Terry C, Blumenstock E. Amniotic fluid embolism causing catastrophic pulmonary vasoconstriction: Diagnosis by transesophageal echocardiogram and treatment by cardiopulmonary bypass. Obstet Gynecol. 2003;102(3):496–498.
- Ho CH, Chen KB, Liu SK, Liu YF, Cheng HC, Wu RS. Early application of extracorporeal membrane oxygenation in a patient with amniotic fluid embolism. Acta Anaesthesiol Taiwan. 2009;47(2):99–102.
- Shen HP, Chang WC, Yeh LS, Ho M. Amniotic fluid embolism treated with emergency extracorporeal membrane oxygenation: A case report.
J Reprod Med. 2009;54(11–12):706–708. - Lee PH, Shulman MS, Vellayappan U, Symes JF, Olenchock SA Jr. Surgical treatment of amniotic fluid embolism with cardiopulmonary collapse. Ann Thorac Surg. 2010;90(5):1694–1696.
- Firstenberg MS, Abel E, Blais D, et al. Temporary extracorporeal circulatory support and pulmonary embolectomy for catastrophic amniotic fluid embolism. Heart Surg Forum. 2011;14(3):E157–E159.
- Ecker JL, Solt K, Fitzsimons MG, MacGillivray TE. Case records of the Massachusetts General Hospital. Case 40-2012: A 43-year-old woman with cardiorespiratory arrest after a cesarean section. N Engl J Med. 2012;367(26):2528–2536.
- Seldinger SI. Catheter replacement of the needle in percutaneous arteriography; a new technique. Acta Radiol. 1953;39(5):368–376.
- Cheng R, Hachamovitch R, Kittelson M, et al. Complications of extracorporeal membrane oxygenation for treatment of cardiogenic shock and cardiac arrest: A meta-analysis of 1,866 adult patients. Ann Thorac Surg. 2013; epub Nov 8.
- Rittenberger JC, Kelly E, Jang D, Greer K, Heffner A. Successful outcome utilizing hypothermia after cardiac arrest in pregnancy: A case report. Crit Care Med. 2008;36(4):1354–1356.
- Michelson AD, MacGregor H, Barnard MR, Krestin AS, Rohrer MJ, Valeri CR. Reversible inhibition of human platelet activation by hypothermia in vivo and in vitro. Thromb Haemost. 1994;71(5):633–640.
- Rivzi F, Mackey R, Barrett T, McKenna P, Geary M. Successful reduction of massive postpartum haemorrhage by use of guidelines and staff education. BJOG. 2004;111(5):495–498.
Case Report: Headache in a Postpartum Patient With Essential Thrombocytosis
Case
At presentation, the patient was in no apparent distress and had normal vital signs, including normal blood pressure. On physical examination, she was alert and oriented, with unremarkable head, neck, fundoscopic, cardiac, pulmonary, skin, and neurological examinations. Laboratory results were only notable for a platelet count of 677 x 109/L. The remainder of the complete blood count, chemistry, and coagulation panels was all within normal limits. A noncontrast computed tomography (CT) of the brain was unremarkable. She was treated symptomatically with ketorolac, diphenhydramine, and metoclopramide, but received no relief.
Based on the patient’s history of ET and postpartum state, there was a high suspicion for cerebral venous thrombosis (CVT). Magnetic resonance imaging/magnetic resonance venography (MRI/MRV) of the brain was obtained, which revealed near-complete thrombosis of the vein of Galen and straight sinus (Figure). Neurology and hematology services were consulted emergently, and the patient was started on a heparin drip and admitted to the neurosurgical intensive care unit. Hydroxyurea was also given for cytoreduction, and platelets normalized to 305 x 109/L within 1 day of treatment. The patient’s neurological examination remained nonfocal, her headache gradually resolved, and she was discharged on hospital day 3 on oral anticoagulants.
Discussion
Essential thrombocytosis is a chronic myeloproliferative disorder characterized by clonal proliferation of the megakaryocyte cell line due to a defect in a pluripotent hematopoietic stem cell.1 The estimated annual incidence is 2.5 per 100,000 individuals.2 It affects patients of both genders, most commonly between ages 50 to 70 years.3
Up to 25% of patients with ET experience neurological complications, mainly occlusive strokes, chronic headache, and dizziness.4 Since pregnancy also increases thrombogenicity, and CVT occurs in 12 per 100,000 deliveries,5 it should be considered in any pregnant or postpartum patient with neurological symptoms.
Diagnosis of ET
Essential thrombocytosis is a diagnosis of exclusion, and no clinical or laboratory finding is diagnostic.1 Although patients with ET are at increased risk for bleeding, these events usually involve mucosal and cutaneous sites and are clinically insignificant. Thrombotic complications account for the majority of the morbidity and mortality associated with ET and can be both arterial and venous. In one case series, roughly 50% patients with ET experienced a thrombotic event within 9 years from diagnosis.6 Thrombotic complications are seen most commonly in patients younger than age 55 years at diagnosis. Common sites of thrombosis include the deep veins of the lower extremities, pulmonary vessels, hepatic vein, portal vein, digital microvasculature, and placenta.1
Despite the high rate of thrombotic complications in ET, there is little data on neurological complications and few reports of CVT in patients with ET.7-9 In one chart review of 70 patients with ET, researchers identified 18 patients who developed neurological complications,4 the most common of which was cerebrovascular accident/transient ischemic attack. Only one patient in the series had a CVT. The majority of patients with neurological events were female and no clinical or laboratory parameter predicted the risk of neurological event.
Thrombocytosis
Thrombocytosis is classified as either primary/essential or secondary/reactive. The majority of thrombocytosis is secondary/reactive and is caused by a variety of inflammatory conditions, such as infection and malignancy. Unlike ET, secondary thrombocytosis is not associated with any bleeding or thrombotic complications and does not require direct treatment.1
Treatment of ET
The decision to treat ET is based on clinical parameters and is usually reserved for patients at high-risk of thrombosis, including those with history of thrombosis or platelets >1,500 x 109/L.1 The first-line agent for treatment of essential thrombocytosis, hydroxyurea has been shown to decrease thrombotic episodes.10 Other cytoreductive agents include anagrilide and interferon-α, the use of which is considered safe in pregnancy.1
Cerebral Venous Thrombosis
Cerebral venous thrombosis is a rare but potentially fatal condition. Patients may present with headache (95%), seizures (47%), and vision changes (41%), and may or may not have focal or generalized neurological deficits.11 Thrombosis can lead to venous obstruction, resulting in increased intracranial pressure and ultimately cerebral herniation and death. Predisposing risk factors for CVT include thrombophilic and procoagulant disorders, maxillofacial infections, trauma, malignancy, and vasculitides. Among female patients with CVT, studies show approximately 50% were on oral contraceptives and 20% were pregnant or in the postpartum period at the time of the event. Nearly half of patients with CVT have multiple risk factors.12
Diagnosis of CVT
Diagnosis of CVT requires a high clinical suspicion. Lumbar puncture usually reveals high opening pressure with normal cerebrospinal fluid analysis.13 The classic finding of CVT on a contrast-enhanced CT scan is the “empty delta sign,” which is a central hypointensity within the superior sagittal sinus secondary due to slow/absent flow surrounded by contrast enhancement in a triangular shape. An ischemic infarction crossing arterial boundaries or near a venous sinus—with or without a hemorrhagic component—is also suggestive of CVT. However, CT scans are read as normal or indeterminate in up to 30% patients.14 Moreover, while CT venography may visualize the cerebral venous system, there is a false-negative rate of 25%.15 Therefore, MRI with MRV is the gold standard for diagnosis.
Pregnancy and CVT
As previously noted, pregnant or postpartum patients are at an increased risk of developing CVT. During pregnancy, decreased levels of protein S inhibitors and increased levels of fibrinogen, clotting factors, and protein C inhibitors lead to increased thrombogenicity.16 Older maternal age, pregnancy-related hypertension, peripartum infections, and cesarean delivery also increase the risk of CVT.16
Patients with CVT associated with pregnancy have more acute onset of symptoms and neurological findings, and 2% to 10% less mortality than patients with CVT secondary to other etiologies. Cerebral venous thrombosis should be considered in the differential diagnosis of any pregnant or postpartum patient presenting with neurological symptoms.
Treatment of CVT
Regardless of underlying etiology or symptom duration, all patients with CVT should receive anticoagulation therapy. Unfractionated or low-molecular weight heparin has been shown to decrease thrombus propagation, increase the rate of recanalization, and improve long-term outcomes—even in the presence of intracranial hemorrhage.17 In the 30% to 40% of patients with CVT who present with intracranial hemorrhage, anticoagulation decreases mortality and is not associated with new or enlarged bleeding.17-19 Patients who continue to deteriorate despite anticoagulation therapy may benefit from endovascular thrombolysis or decompressive hemicraniectomy.20 In a study of patients with CVT, 80% had full recovery, 6% had minor disability, and 14% had a poor outcome.12
Conclusion
This case describes a patient with two risk factors for CVT, a life-threatening condition. It should be considered in patients with ET and/or pregnant and postpartum patients presenting with headache or other neurological symptoms. Magnetic resonance venography is the diagnostic imaging of choice as the CVT may not be visible on CT. All patients with thrombosis and ET should receive anticoagulation therapy and hydroxyurea to prevent cerebral herniation and death.
Dr Canders is a resident, department of emergency medicine, David Geffen School of Medicine, University of California, Los Angeles. Dr Taira is a clinical assistant professor, department of emergency medicine, Los Angeles County Hospital/University of Southern California, Los Angeles.
Disclosure: The authors report no financial disclosures or conflicts of interests related to this article.
- Schafer AI. Thrombocytosis and thrombocythemia. Blood Rev. 2001;15(4):159-166.
- Mesa RA, Silverstein MN, Jacobsen SJ, Wollan PC, Tefferi A. Population-based incidence and survival figures in essential thrombocythemia and agnogenic myeloid metaplasia: an Olmsted County Study, 1976-1995. Am J Hematol. 1999;61():10-15.
- Tefferi A. Recent progress in the pathogenesis and management of essential thrombocythemia. Leuk Res. 2001;25(5):369-377.
- Kesler A, Ellis MH, Manor Y, Gadoth N, Lishner M. Neurological complications of essential thrombocytosis (ET). Acta Neurol Scand. 2000;102(5):299-302.
- Cantú C, Barinagarrementeria F. Cerebral venous thrombosis associated with pregnancy and puerperium: Review of 67 cases. Stroke. 1993;24(12):1880-1884.
- Bazzan M, Tamponi G, Schinco P, Vaccarino A, Foli C, Gallone G, et al. Thrombosis-free survival and life expectancy in 187 consecutive patients with essential thrombocythemia. Ann Hematol. 1999;78(12):539-543.
- Walther EU, Tiecks FP, Haberl RL. Cranial sinus thrombosis associated with essential thrombocythemia followed by heparin-associated thrombocytopenia. Neurology. 1996;47(1):300-301.
- McDonald TD, Tatemichi TK, Kranzler SJ, Chi L, Hilal SK, Mohr JP. Thrombosis of the superior sagittal sinus associated with essential thrombocytosis followed by MRI during anticoagulant therapy. Neurology. 1989;39(11):1554-1555.
- Iob I, Scanarini M, Andrioli GC, Pardatscher K. Thrombosis of the superior sagittal sinus associated with idiopathic thrombocytosis. Surg Neurol. 1979;11(6):439-41.
- Cortelazzo S, Finazzi G, Ruggeri M, Vestri O, Galli M, Rodeghiero F, et al. Hydroxyurea for patients with essential thrombocythemia and a high risk of thrombosis. N Engl J Med. 1995;332(17):1132-1136.
- de Bruijn SF, de Haan RJ, Stam J. Clinical features and prognostic factors of cerebral venous sinus thrombosis in a prospective series of 59 patients. For The Cerebral Venous Sinus Thrombosis Study Group. J Neurol Neurosurg Psychiatry. 2001;70(1):105-108.
- Ferro JM, Canhão P, Stam J, Bousser MG, Barinagarrementeria F; ISCVT Investigators. Prognosis of cerebral vein and dural sinus thrombosis: results of the International Study on Cerebral Vein and Dural Sinus Thrombosis (ISCVT). Stroke. 2004;35(3):664-670.
- DeLashaw MR, Vizioli TL, Jr, Counselman FL. Headache and seizure in a young woman postpartum. J Emerg Med. 2005;29(3):289-293.
- Bousser MG. Cerebral venous thrombosis: diagnosis and management. J Neurol. 2000;247(4):252-258.
- Grover M. Cerebral venous thrombosis as a cause of acute headache. J Am Board Fam Pract. 2004;17(4):295-298.
- Martinelli I, Sacchi E, Landi G, Taioli E, Duca F, Mannucci PM. High risk of cerebral venous thrombosis in carriers of prothrombic-gene mutation and in users of oral contraceptives. N Engl J Med. 1988;338(25):1793-1797.
- Star M, Flaster M. Advances and controversies in the management of cerebral venous thrombosis. Neurol Clin. 2013;31(3):765-783.
- Einhäupl KM, Villringer A, Meister W, Mehraein S, Garner C, Pellkofer M, et al. Heparin treatment in sinus venous thrombosis. Lancet. 1991;338(8767):597-600.
- Preter M, Tzourio C, Ameri A, Bousser MG. Long-term prognosis in cerebral venous thrombosis: follow-up of 77 patients. Stroke 1996;27(2):243-246.
- Coutinho JM, Stam J. How to treat cerebral venous and sinus thrombosis. J Thromb Haemost. 2010;8(5):877-883.
Case
At presentation, the patient was in no apparent distress and had normal vital signs, including normal blood pressure. On physical examination, she was alert and oriented, with unremarkable head, neck, fundoscopic, cardiac, pulmonary, skin, and neurological examinations. Laboratory results were only notable for a platelet count of 677 x 109/L. The remainder of the complete blood count, chemistry, and coagulation panels was all within normal limits. A noncontrast computed tomography (CT) of the brain was unremarkable. She was treated symptomatically with ketorolac, diphenhydramine, and metoclopramide, but received no relief.
Based on the patient’s history of ET and postpartum state, there was a high suspicion for cerebral venous thrombosis (CVT). Magnetic resonance imaging/magnetic resonance venography (MRI/MRV) of the brain was obtained, which revealed near-complete thrombosis of the vein of Galen and straight sinus (Figure). Neurology and hematology services were consulted emergently, and the patient was started on a heparin drip and admitted to the neurosurgical intensive care unit. Hydroxyurea was also given for cytoreduction, and platelets normalized to 305 x 109/L within 1 day of treatment. The patient’s neurological examination remained nonfocal, her headache gradually resolved, and she was discharged on hospital day 3 on oral anticoagulants.
Discussion
Essential thrombocytosis is a chronic myeloproliferative disorder characterized by clonal proliferation of the megakaryocyte cell line due to a defect in a pluripotent hematopoietic stem cell.1 The estimated annual incidence is 2.5 per 100,000 individuals.2 It affects patients of both genders, most commonly between ages 50 to 70 years.3
Up to 25% of patients with ET experience neurological complications, mainly occlusive strokes, chronic headache, and dizziness.4 Since pregnancy also increases thrombogenicity, and CVT occurs in 12 per 100,000 deliveries,5 it should be considered in any pregnant or postpartum patient with neurological symptoms.
Diagnosis of ET
Essential thrombocytosis is a diagnosis of exclusion, and no clinical or laboratory finding is diagnostic.1 Although patients with ET are at increased risk for bleeding, these events usually involve mucosal and cutaneous sites and are clinically insignificant. Thrombotic complications account for the majority of the morbidity and mortality associated with ET and can be both arterial and venous. In one case series, roughly 50% patients with ET experienced a thrombotic event within 9 years from diagnosis.6 Thrombotic complications are seen most commonly in patients younger than age 55 years at diagnosis. Common sites of thrombosis include the deep veins of the lower extremities, pulmonary vessels, hepatic vein, portal vein, digital microvasculature, and placenta.1
Despite the high rate of thrombotic complications in ET, there is little data on neurological complications and few reports of CVT in patients with ET.7-9 In one chart review of 70 patients with ET, researchers identified 18 patients who developed neurological complications,4 the most common of which was cerebrovascular accident/transient ischemic attack. Only one patient in the series had a CVT. The majority of patients with neurological events were female and no clinical or laboratory parameter predicted the risk of neurological event.
Thrombocytosis
Thrombocytosis is classified as either primary/essential or secondary/reactive. The majority of thrombocytosis is secondary/reactive and is caused by a variety of inflammatory conditions, such as infection and malignancy. Unlike ET, secondary thrombocytosis is not associated with any bleeding or thrombotic complications and does not require direct treatment.1
Treatment of ET
The decision to treat ET is based on clinical parameters and is usually reserved for patients at high-risk of thrombosis, including those with history of thrombosis or platelets >1,500 x 109/L.1 The first-line agent for treatment of essential thrombocytosis, hydroxyurea has been shown to decrease thrombotic episodes.10 Other cytoreductive agents include anagrilide and interferon-α, the use of which is considered safe in pregnancy.1
Cerebral Venous Thrombosis
Cerebral venous thrombosis is a rare but potentially fatal condition. Patients may present with headache (95%), seizures (47%), and vision changes (41%), and may or may not have focal or generalized neurological deficits.11 Thrombosis can lead to venous obstruction, resulting in increased intracranial pressure and ultimately cerebral herniation and death. Predisposing risk factors for CVT include thrombophilic and procoagulant disorders, maxillofacial infections, trauma, malignancy, and vasculitides. Among female patients with CVT, studies show approximately 50% were on oral contraceptives and 20% were pregnant or in the postpartum period at the time of the event. Nearly half of patients with CVT have multiple risk factors.12
Diagnosis of CVT
Diagnosis of CVT requires a high clinical suspicion. Lumbar puncture usually reveals high opening pressure with normal cerebrospinal fluid analysis.13 The classic finding of CVT on a contrast-enhanced CT scan is the “empty delta sign,” which is a central hypointensity within the superior sagittal sinus secondary due to slow/absent flow surrounded by contrast enhancement in a triangular shape. An ischemic infarction crossing arterial boundaries or near a venous sinus—with or without a hemorrhagic component—is also suggestive of CVT. However, CT scans are read as normal or indeterminate in up to 30% patients.14 Moreover, while CT venography may visualize the cerebral venous system, there is a false-negative rate of 25%.15 Therefore, MRI with MRV is the gold standard for diagnosis.
Pregnancy and CVT
As previously noted, pregnant or postpartum patients are at an increased risk of developing CVT. During pregnancy, decreased levels of protein S inhibitors and increased levels of fibrinogen, clotting factors, and protein C inhibitors lead to increased thrombogenicity.16 Older maternal age, pregnancy-related hypertension, peripartum infections, and cesarean delivery also increase the risk of CVT.16
Patients with CVT associated with pregnancy have more acute onset of symptoms and neurological findings, and 2% to 10% less mortality than patients with CVT secondary to other etiologies. Cerebral venous thrombosis should be considered in the differential diagnosis of any pregnant or postpartum patient presenting with neurological symptoms.
Treatment of CVT
Regardless of underlying etiology or symptom duration, all patients with CVT should receive anticoagulation therapy. Unfractionated or low-molecular weight heparin has been shown to decrease thrombus propagation, increase the rate of recanalization, and improve long-term outcomes—even in the presence of intracranial hemorrhage.17 In the 30% to 40% of patients with CVT who present with intracranial hemorrhage, anticoagulation decreases mortality and is not associated with new or enlarged bleeding.17-19 Patients who continue to deteriorate despite anticoagulation therapy may benefit from endovascular thrombolysis or decompressive hemicraniectomy.20 In a study of patients with CVT, 80% had full recovery, 6% had minor disability, and 14% had a poor outcome.12
Conclusion
This case describes a patient with two risk factors for CVT, a life-threatening condition. It should be considered in patients with ET and/or pregnant and postpartum patients presenting with headache or other neurological symptoms. Magnetic resonance venography is the diagnostic imaging of choice as the CVT may not be visible on CT. All patients with thrombosis and ET should receive anticoagulation therapy and hydroxyurea to prevent cerebral herniation and death.
Dr Canders is a resident, department of emergency medicine, David Geffen School of Medicine, University of California, Los Angeles. Dr Taira is a clinical assistant professor, department of emergency medicine, Los Angeles County Hospital/University of Southern California, Los Angeles.
Disclosure: The authors report no financial disclosures or conflicts of interests related to this article.
Case
At presentation, the patient was in no apparent distress and had normal vital signs, including normal blood pressure. On physical examination, she was alert and oriented, with unremarkable head, neck, fundoscopic, cardiac, pulmonary, skin, and neurological examinations. Laboratory results were only notable for a platelet count of 677 x 109/L. The remainder of the complete blood count, chemistry, and coagulation panels was all within normal limits. A noncontrast computed tomography (CT) of the brain was unremarkable. She was treated symptomatically with ketorolac, diphenhydramine, and metoclopramide, but received no relief.
Based on the patient’s history of ET and postpartum state, there was a high suspicion for cerebral venous thrombosis (CVT). Magnetic resonance imaging/magnetic resonance venography (MRI/MRV) of the brain was obtained, which revealed near-complete thrombosis of the vein of Galen and straight sinus (Figure). Neurology and hematology services were consulted emergently, and the patient was started on a heparin drip and admitted to the neurosurgical intensive care unit. Hydroxyurea was also given for cytoreduction, and platelets normalized to 305 x 109/L within 1 day of treatment. The patient’s neurological examination remained nonfocal, her headache gradually resolved, and she was discharged on hospital day 3 on oral anticoagulants.
Discussion
Essential thrombocytosis is a chronic myeloproliferative disorder characterized by clonal proliferation of the megakaryocyte cell line due to a defect in a pluripotent hematopoietic stem cell.1 The estimated annual incidence is 2.5 per 100,000 individuals.2 It affects patients of both genders, most commonly between ages 50 to 70 years.3
Up to 25% of patients with ET experience neurological complications, mainly occlusive strokes, chronic headache, and dizziness.4 Since pregnancy also increases thrombogenicity, and CVT occurs in 12 per 100,000 deliveries,5 it should be considered in any pregnant or postpartum patient with neurological symptoms.
Diagnosis of ET
Essential thrombocytosis is a diagnosis of exclusion, and no clinical or laboratory finding is diagnostic.1 Although patients with ET are at increased risk for bleeding, these events usually involve mucosal and cutaneous sites and are clinically insignificant. Thrombotic complications account for the majority of the morbidity and mortality associated with ET and can be both arterial and venous. In one case series, roughly 50% patients with ET experienced a thrombotic event within 9 years from diagnosis.6 Thrombotic complications are seen most commonly in patients younger than age 55 years at diagnosis. Common sites of thrombosis include the deep veins of the lower extremities, pulmonary vessels, hepatic vein, portal vein, digital microvasculature, and placenta.1
Despite the high rate of thrombotic complications in ET, there is little data on neurological complications and few reports of CVT in patients with ET.7-9 In one chart review of 70 patients with ET, researchers identified 18 patients who developed neurological complications,4 the most common of which was cerebrovascular accident/transient ischemic attack. Only one patient in the series had a CVT. The majority of patients with neurological events were female and no clinical or laboratory parameter predicted the risk of neurological event.
Thrombocytosis
Thrombocytosis is classified as either primary/essential or secondary/reactive. The majority of thrombocytosis is secondary/reactive and is caused by a variety of inflammatory conditions, such as infection and malignancy. Unlike ET, secondary thrombocytosis is not associated with any bleeding or thrombotic complications and does not require direct treatment.1
Treatment of ET
The decision to treat ET is based on clinical parameters and is usually reserved for patients at high-risk of thrombosis, including those with history of thrombosis or platelets >1,500 x 109/L.1 The first-line agent for treatment of essential thrombocytosis, hydroxyurea has been shown to decrease thrombotic episodes.10 Other cytoreductive agents include anagrilide and interferon-α, the use of which is considered safe in pregnancy.1
Cerebral Venous Thrombosis
Cerebral venous thrombosis is a rare but potentially fatal condition. Patients may present with headache (95%), seizures (47%), and vision changes (41%), and may or may not have focal or generalized neurological deficits.11 Thrombosis can lead to venous obstruction, resulting in increased intracranial pressure and ultimately cerebral herniation and death. Predisposing risk factors for CVT include thrombophilic and procoagulant disorders, maxillofacial infections, trauma, malignancy, and vasculitides. Among female patients with CVT, studies show approximately 50% were on oral contraceptives and 20% were pregnant or in the postpartum period at the time of the event. Nearly half of patients with CVT have multiple risk factors.12
Diagnosis of CVT
Diagnosis of CVT requires a high clinical suspicion. Lumbar puncture usually reveals high opening pressure with normal cerebrospinal fluid analysis.13 The classic finding of CVT on a contrast-enhanced CT scan is the “empty delta sign,” which is a central hypointensity within the superior sagittal sinus secondary due to slow/absent flow surrounded by contrast enhancement in a triangular shape. An ischemic infarction crossing arterial boundaries or near a venous sinus—with or without a hemorrhagic component—is also suggestive of CVT. However, CT scans are read as normal or indeterminate in up to 30% patients.14 Moreover, while CT venography may visualize the cerebral venous system, there is a false-negative rate of 25%.15 Therefore, MRI with MRV is the gold standard for diagnosis.
Pregnancy and CVT
As previously noted, pregnant or postpartum patients are at an increased risk of developing CVT. During pregnancy, decreased levels of protein S inhibitors and increased levels of fibrinogen, clotting factors, and protein C inhibitors lead to increased thrombogenicity.16 Older maternal age, pregnancy-related hypertension, peripartum infections, and cesarean delivery also increase the risk of CVT.16
Patients with CVT associated with pregnancy have more acute onset of symptoms and neurological findings, and 2% to 10% less mortality than patients with CVT secondary to other etiologies. Cerebral venous thrombosis should be considered in the differential diagnosis of any pregnant or postpartum patient presenting with neurological symptoms.
Treatment of CVT
Regardless of underlying etiology or symptom duration, all patients with CVT should receive anticoagulation therapy. Unfractionated or low-molecular weight heparin has been shown to decrease thrombus propagation, increase the rate of recanalization, and improve long-term outcomes—even in the presence of intracranial hemorrhage.17 In the 30% to 40% of patients with CVT who present with intracranial hemorrhage, anticoagulation decreases mortality and is not associated with new or enlarged bleeding.17-19 Patients who continue to deteriorate despite anticoagulation therapy may benefit from endovascular thrombolysis or decompressive hemicraniectomy.20 In a study of patients with CVT, 80% had full recovery, 6% had minor disability, and 14% had a poor outcome.12
Conclusion
This case describes a patient with two risk factors for CVT, a life-threatening condition. It should be considered in patients with ET and/or pregnant and postpartum patients presenting with headache or other neurological symptoms. Magnetic resonance venography is the diagnostic imaging of choice as the CVT may not be visible on CT. All patients with thrombosis and ET should receive anticoagulation therapy and hydroxyurea to prevent cerebral herniation and death.
Dr Canders is a resident, department of emergency medicine, David Geffen School of Medicine, University of California, Los Angeles. Dr Taira is a clinical assistant professor, department of emergency medicine, Los Angeles County Hospital/University of Southern California, Los Angeles.
Disclosure: The authors report no financial disclosures or conflicts of interests related to this article.
- Schafer AI. Thrombocytosis and thrombocythemia. Blood Rev. 2001;15(4):159-166.
- Mesa RA, Silverstein MN, Jacobsen SJ, Wollan PC, Tefferi A. Population-based incidence and survival figures in essential thrombocythemia and agnogenic myeloid metaplasia: an Olmsted County Study, 1976-1995. Am J Hematol. 1999;61():10-15.
- Tefferi A. Recent progress in the pathogenesis and management of essential thrombocythemia. Leuk Res. 2001;25(5):369-377.
- Kesler A, Ellis MH, Manor Y, Gadoth N, Lishner M. Neurological complications of essential thrombocytosis (ET). Acta Neurol Scand. 2000;102(5):299-302.
- Cantú C, Barinagarrementeria F. Cerebral venous thrombosis associated with pregnancy and puerperium: Review of 67 cases. Stroke. 1993;24(12):1880-1884.
- Bazzan M, Tamponi G, Schinco P, Vaccarino A, Foli C, Gallone G, et al. Thrombosis-free survival and life expectancy in 187 consecutive patients with essential thrombocythemia. Ann Hematol. 1999;78(12):539-543.
- Walther EU, Tiecks FP, Haberl RL. Cranial sinus thrombosis associated with essential thrombocythemia followed by heparin-associated thrombocytopenia. Neurology. 1996;47(1):300-301.
- McDonald TD, Tatemichi TK, Kranzler SJ, Chi L, Hilal SK, Mohr JP. Thrombosis of the superior sagittal sinus associated with essential thrombocytosis followed by MRI during anticoagulant therapy. Neurology. 1989;39(11):1554-1555.
- Iob I, Scanarini M, Andrioli GC, Pardatscher K. Thrombosis of the superior sagittal sinus associated with idiopathic thrombocytosis. Surg Neurol. 1979;11(6):439-41.
- Cortelazzo S, Finazzi G, Ruggeri M, Vestri O, Galli M, Rodeghiero F, et al. Hydroxyurea for patients with essential thrombocythemia and a high risk of thrombosis. N Engl J Med. 1995;332(17):1132-1136.
- de Bruijn SF, de Haan RJ, Stam J. Clinical features and prognostic factors of cerebral venous sinus thrombosis in a prospective series of 59 patients. For The Cerebral Venous Sinus Thrombosis Study Group. J Neurol Neurosurg Psychiatry. 2001;70(1):105-108.
- Ferro JM, Canhão P, Stam J, Bousser MG, Barinagarrementeria F; ISCVT Investigators. Prognosis of cerebral vein and dural sinus thrombosis: results of the International Study on Cerebral Vein and Dural Sinus Thrombosis (ISCVT). Stroke. 2004;35(3):664-670.
- DeLashaw MR, Vizioli TL, Jr, Counselman FL. Headache and seizure in a young woman postpartum. J Emerg Med. 2005;29(3):289-293.
- Bousser MG. Cerebral venous thrombosis: diagnosis and management. J Neurol. 2000;247(4):252-258.
- Grover M. Cerebral venous thrombosis as a cause of acute headache. J Am Board Fam Pract. 2004;17(4):295-298.
- Martinelli I, Sacchi E, Landi G, Taioli E, Duca F, Mannucci PM. High risk of cerebral venous thrombosis in carriers of prothrombic-gene mutation and in users of oral contraceptives. N Engl J Med. 1988;338(25):1793-1797.
- Star M, Flaster M. Advances and controversies in the management of cerebral venous thrombosis. Neurol Clin. 2013;31(3):765-783.
- Einhäupl KM, Villringer A, Meister W, Mehraein S, Garner C, Pellkofer M, et al. Heparin treatment in sinus venous thrombosis. Lancet. 1991;338(8767):597-600.
- Preter M, Tzourio C, Ameri A, Bousser MG. Long-term prognosis in cerebral venous thrombosis: follow-up of 77 patients. Stroke 1996;27(2):243-246.
- Coutinho JM, Stam J. How to treat cerebral venous and sinus thrombosis. J Thromb Haemost. 2010;8(5):877-883.
- Schafer AI. Thrombocytosis and thrombocythemia. Blood Rev. 2001;15(4):159-166.
- Mesa RA, Silverstein MN, Jacobsen SJ, Wollan PC, Tefferi A. Population-based incidence and survival figures in essential thrombocythemia and agnogenic myeloid metaplasia: an Olmsted County Study, 1976-1995. Am J Hematol. 1999;61():10-15.
- Tefferi A. Recent progress in the pathogenesis and management of essential thrombocythemia. Leuk Res. 2001;25(5):369-377.
- Kesler A, Ellis MH, Manor Y, Gadoth N, Lishner M. Neurological complications of essential thrombocytosis (ET). Acta Neurol Scand. 2000;102(5):299-302.
- Cantú C, Barinagarrementeria F. Cerebral venous thrombosis associated with pregnancy and puerperium: Review of 67 cases. Stroke. 1993;24(12):1880-1884.
- Bazzan M, Tamponi G, Schinco P, Vaccarino A, Foli C, Gallone G, et al. Thrombosis-free survival and life expectancy in 187 consecutive patients with essential thrombocythemia. Ann Hematol. 1999;78(12):539-543.
- Walther EU, Tiecks FP, Haberl RL. Cranial sinus thrombosis associated with essential thrombocythemia followed by heparin-associated thrombocytopenia. Neurology. 1996;47(1):300-301.
- McDonald TD, Tatemichi TK, Kranzler SJ, Chi L, Hilal SK, Mohr JP. Thrombosis of the superior sagittal sinus associated with essential thrombocytosis followed by MRI during anticoagulant therapy. Neurology. 1989;39(11):1554-1555.
- Iob I, Scanarini M, Andrioli GC, Pardatscher K. Thrombosis of the superior sagittal sinus associated with idiopathic thrombocytosis. Surg Neurol. 1979;11(6):439-41.
- Cortelazzo S, Finazzi G, Ruggeri M, Vestri O, Galli M, Rodeghiero F, et al. Hydroxyurea for patients with essential thrombocythemia and a high risk of thrombosis. N Engl J Med. 1995;332(17):1132-1136.
- de Bruijn SF, de Haan RJ, Stam J. Clinical features and prognostic factors of cerebral venous sinus thrombosis in a prospective series of 59 patients. For The Cerebral Venous Sinus Thrombosis Study Group. J Neurol Neurosurg Psychiatry. 2001;70(1):105-108.
- Ferro JM, Canhão P, Stam J, Bousser MG, Barinagarrementeria F; ISCVT Investigators. Prognosis of cerebral vein and dural sinus thrombosis: results of the International Study on Cerebral Vein and Dural Sinus Thrombosis (ISCVT). Stroke. 2004;35(3):664-670.
- DeLashaw MR, Vizioli TL, Jr, Counselman FL. Headache and seizure in a young woman postpartum. J Emerg Med. 2005;29(3):289-293.
- Bousser MG. Cerebral venous thrombosis: diagnosis and management. J Neurol. 2000;247(4):252-258.
- Grover M. Cerebral venous thrombosis as a cause of acute headache. J Am Board Fam Pract. 2004;17(4):295-298.
- Martinelli I, Sacchi E, Landi G, Taioli E, Duca F, Mannucci PM. High risk of cerebral venous thrombosis in carriers of prothrombic-gene mutation and in users of oral contraceptives. N Engl J Med. 1988;338(25):1793-1797.
- Star M, Flaster M. Advances and controversies in the management of cerebral venous thrombosis. Neurol Clin. 2013;31(3):765-783.
- Einhäupl KM, Villringer A, Meister W, Mehraein S, Garner C, Pellkofer M, et al. Heparin treatment in sinus venous thrombosis. Lancet. 1991;338(8767):597-600.
- Preter M, Tzourio C, Ameri A, Bousser MG. Long-term prognosis in cerebral venous thrombosis: follow-up of 77 patients. Stroke 1996;27(2):243-246.
- Coutinho JM, Stam J. How to treat cerebral venous and sinus thrombosis. J Thromb Haemost. 2010;8(5):877-883.
Who will be found at fault?
I am always interested in the Medical Verdicts section (it’s like watching a car accident—you can’t look away). I recently was involved in an argument in a physician-only online forum where a participant presented a case of a failed homebirth brought into a hospital with an obstetrics (OB) service. A call was allegedly placed to one of the ObGyns who refused to come to the hospital and attend the patient, claiming that since the hospital had negotiated no contract with the ObGyns for unassigned coverage, he was not obligated to do so. The baby died. A lawsuit was filed.
I argued that, per the Emergency Medical Treatment and Active Labor Act (EMTALA), the hospital is required to have an unassigned call roster for OB. Usually it is an obligation of privileges and is outlined in the medical staff bylaws; sometimes payment is offered by the hospital, sometimes not.
A spirited discussion ensued. A bunch of emergency department (ED) physicians said that they practice in hospitals where no one can be “forced to take a call for unassigned patients for free against their will” and that this scenario is quite common. They felt there is nothing in EMTALA that requires a hospital to have on-call physicians available. They said it is common to be unable to find specialists, surgeons, or ObGyns willing to come in and take these patients.
I still don’t know what our legal obligation is under EMTALA, but I had never heard of an OB service without a provision for unassigned patients.
If this story is true, and not just an apocryphal tale, who will be found at fault? (Probably everyone.)
Deborah Owen, MD
Spokane, Washington
Shirley Pruitt, RN, JD, responds
The obligation to maintain a call schedule is imposed on hospitals by a section of the Medicare statute that refers back to the EMTALA obligations.1 Generally, if a hospital provides specialized services to the public, it is required to provide these services through ED on-call coverage. Each hospital has the discretion to maintain the on-call list in a manner to best meet the needs of its patients. The medical staff bylaws or policies and procedures must define 1) the responsibility of on-call physicians to respond, examine, and treat patients with emergency medical conditions, and 2) the procedures to be followed when a particular specialty is not available or the on-call physician cannot respond because of situations beyond his or her control. The Centers for Medicare and Medicaid Services (CMS) may impose a penalty on a physician who fails to respond to an emergency situation when he or she is assigned as the on-call physician.
There is no Federal law through CMS/EMTALA requiring a specialist to participate in the on-call list. However, a specialist who refuses to participate in the on-call list may not take “selective call” and agree to see patients with whom the specialist has a prior existing relationship while refusing to see other patients with whom there is no such relationship.
Disclaimer
This information should not be construed as business, risk management, or legal advice or legal opinion.
Shirley M. Pruitt, RN, JD
Yates, McLamb & Weyher, LLP
Raleigh, North Carolina
OBG Management Contributing Editor
Do you have a DIAGNOSTIC IN-SIGHT?
Read What is causing her abdominal pain? by Chetan Narasanna, MD, Reginald Griffin, MD, Michael S. Nussbaum, MD, and Andrew M. Kaunitz, MD.
Submit a query for your image-based case to [email protected]
Reference
- The Public Health and Welfare, 42 USC §1395dd et seq (2011).
I am always interested in the Medical Verdicts section (it’s like watching a car accident—you can’t look away). I recently was involved in an argument in a physician-only online forum where a participant presented a case of a failed homebirth brought into a hospital with an obstetrics (OB) service. A call was allegedly placed to one of the ObGyns who refused to come to the hospital and attend the patient, claiming that since the hospital had negotiated no contract with the ObGyns for unassigned coverage, he was not obligated to do so. The baby died. A lawsuit was filed.
I argued that, per the Emergency Medical Treatment and Active Labor Act (EMTALA), the hospital is required to have an unassigned call roster for OB. Usually it is an obligation of privileges and is outlined in the medical staff bylaws; sometimes payment is offered by the hospital, sometimes not.
A spirited discussion ensued. A bunch of emergency department (ED) physicians said that they practice in hospitals where no one can be “forced to take a call for unassigned patients for free against their will” and that this scenario is quite common. They felt there is nothing in EMTALA that requires a hospital to have on-call physicians available. They said it is common to be unable to find specialists, surgeons, or ObGyns willing to come in and take these patients.
I still don’t know what our legal obligation is under EMTALA, but I had never heard of an OB service without a provision for unassigned patients.
If this story is true, and not just an apocryphal tale, who will be found at fault? (Probably everyone.)
Deborah Owen, MD
Spokane, Washington
Shirley Pruitt, RN, JD, responds
The obligation to maintain a call schedule is imposed on hospitals by a section of the Medicare statute that refers back to the EMTALA obligations.1 Generally, if a hospital provides specialized services to the public, it is required to provide these services through ED on-call coverage. Each hospital has the discretion to maintain the on-call list in a manner to best meet the needs of its patients. The medical staff bylaws or policies and procedures must define 1) the responsibility of on-call physicians to respond, examine, and treat patients with emergency medical conditions, and 2) the procedures to be followed when a particular specialty is not available or the on-call physician cannot respond because of situations beyond his or her control. The Centers for Medicare and Medicaid Services (CMS) may impose a penalty on a physician who fails to respond to an emergency situation when he or she is assigned as the on-call physician.
There is no Federal law through CMS/EMTALA requiring a specialist to participate in the on-call list. However, a specialist who refuses to participate in the on-call list may not take “selective call” and agree to see patients with whom the specialist has a prior existing relationship while refusing to see other patients with whom there is no such relationship.
Disclaimer
This information should not be construed as business, risk management, or legal advice or legal opinion.
Shirley M. Pruitt, RN, JD
Yates, McLamb & Weyher, LLP
Raleigh, North Carolina
OBG Management Contributing Editor
Do you have a DIAGNOSTIC IN-SIGHT?
Read What is causing her abdominal pain? by Chetan Narasanna, MD, Reginald Griffin, MD, Michael S. Nussbaum, MD, and Andrew M. Kaunitz, MD.
Submit a query for your image-based case to [email protected]
I am always interested in the Medical Verdicts section (it’s like watching a car accident—you can’t look away). I recently was involved in an argument in a physician-only online forum where a participant presented a case of a failed homebirth brought into a hospital with an obstetrics (OB) service. A call was allegedly placed to one of the ObGyns who refused to come to the hospital and attend the patient, claiming that since the hospital had negotiated no contract with the ObGyns for unassigned coverage, he was not obligated to do so. The baby died. A lawsuit was filed.
I argued that, per the Emergency Medical Treatment and Active Labor Act (EMTALA), the hospital is required to have an unassigned call roster for OB. Usually it is an obligation of privileges and is outlined in the medical staff bylaws; sometimes payment is offered by the hospital, sometimes not.
A spirited discussion ensued. A bunch of emergency department (ED) physicians said that they practice in hospitals where no one can be “forced to take a call for unassigned patients for free against their will” and that this scenario is quite common. They felt there is nothing in EMTALA that requires a hospital to have on-call physicians available. They said it is common to be unable to find specialists, surgeons, or ObGyns willing to come in and take these patients.
I still don’t know what our legal obligation is under EMTALA, but I had never heard of an OB service without a provision for unassigned patients.
If this story is true, and not just an apocryphal tale, who will be found at fault? (Probably everyone.)
Deborah Owen, MD
Spokane, Washington
Shirley Pruitt, RN, JD, responds
The obligation to maintain a call schedule is imposed on hospitals by a section of the Medicare statute that refers back to the EMTALA obligations.1 Generally, if a hospital provides specialized services to the public, it is required to provide these services through ED on-call coverage. Each hospital has the discretion to maintain the on-call list in a manner to best meet the needs of its patients. The medical staff bylaws or policies and procedures must define 1) the responsibility of on-call physicians to respond, examine, and treat patients with emergency medical conditions, and 2) the procedures to be followed when a particular specialty is not available or the on-call physician cannot respond because of situations beyond his or her control. The Centers for Medicare and Medicaid Services (CMS) may impose a penalty on a physician who fails to respond to an emergency situation when he or she is assigned as the on-call physician.
There is no Federal law through CMS/EMTALA requiring a specialist to participate in the on-call list. However, a specialist who refuses to participate in the on-call list may not take “selective call” and agree to see patients with whom the specialist has a prior existing relationship while refusing to see other patients with whom there is no such relationship.
Disclaimer
This information should not be construed as business, risk management, or legal advice or legal opinion.
Shirley M. Pruitt, RN, JD
Yates, McLamb & Weyher, LLP
Raleigh, North Carolina
OBG Management Contributing Editor
Do you have a DIAGNOSTIC IN-SIGHT?
Read What is causing her abdominal pain? by Chetan Narasanna, MD, Reginald Griffin, MD, Michael S. Nussbaum, MD, and Andrew M. Kaunitz, MD.
Submit a query for your image-based case to [email protected]
Reference
- The Public Health and Welfare, 42 USC §1395dd et seq (2011).
Reference
- The Public Health and Welfare, 42 USC §1395dd et seq (2011).
WE LIKE TO HEAR FROM YOU!
Readers are always encouraged to “Tell us what you think!” We want to know your feedback regarding current articles, topics you’d like to see covered in future issues, and what challenges you face in daily practice.
Here’s a letter from an avid reader about emergency department on-call obligations, and a response from our legal expert.
What’s your opinion? Email us at [email protected]. Please include your name and the city and state in which you practice.