User login
Surgeon General report links smoking to diseases beyond cancer
While smoking rates have dropped precipitously since the landmark 1964 Surgeon General’s report, "Smoking and Health," smoking is still the leading cause of preventable disease and death in the United States and is now causally linked to additional diseases and conditions across most organ systems.
A new Surgeon General's report, "The Health Consequences of Smoking – 50 Years of Progress," released at a White House event Jan. 17, synthesizes original and review evidence in an effort to further federal antismoking efforts.
The report causally links cigarette smoking to type 2 diabetes, rheumatoid arthritis, ectopic pregnancy, and erectile dysfunction. Secondhand smoke is now causally linked to cancers, respiratory diseases, and cardiovascular diseases, as well as adverse effects on the health of children, the authors wrote.
In addition, the 980-page report establishes that secondhand smoke is a cause of stroke, and that smoking increases the risk of dying in cancer patients and cancer survivors.
Another finding: Cigarette smokers today have a higher risk of lung cancer than did those who smoked in 1964, because of the higher number of chemical additives now used.
Women smokers now have the same risk of death from lung cancer as that of men and a higher relative risk of dying from coronary heart disease than that of men. Because of smoking, the number of women dying from chronic obstructive pulmonary disease (COPD) is now higher than in men.
With this report, the federal government is launching a new effort to prevent children from using tobacco.
"Today, we’re asking Americans to join a sustained effort to make the next generation a tobacco-free generation," Health and Human Services Secretary Kathleen Sebelius said in a statement. "This is not something the federal government can do alone. We need to partner with the business community, local elected officials, schools and universities, the medical community, the faith community, and committed citizens in communities across the country to make the next generation tobacco free."
The report finds that youth smoking rates declined by 50% between 1997 and 2011, but 3,200 children under age 18 still start smoking each day, and an additional 2,100 youth and young adults become daily smokers. The report places most of the blame for continued interest in smoking on the tobacco industry, saying it has used "aggressive strategies" to deliberately mislead the public about the harms of smoking.
Acting Surgeon General Boris Lushniak noted that smoking rates are disproportionately higher among people with less education and lower incomes, among the mentally ill, and among gay, lesbian, bisexual, and transgender individuals.
Dr. Lushniak and other officials at the White House event called for greater tobacco control efforts, including stricter regulation. The Food and Drug Administration was given the power to regulate tobacco through the 2009 Family Smoking Prevention and Tobacco Control Act.
FDA Commissioner Margaret Hamburg said that the agency is ready to take action.
"The FDA is funding and conducting regulatory science research on tobacco products, enforcing the laws that reduce the access and attractiveness of tobacco products to young people, and preparing to launch an unprecedented national public education campaign to prevent youth tobacco use," Dr. Hamburg said in a statement.
Dr. Thomas R. Frieden, director of the Centers for Disease Control and Prevention, noted that while states collect some $80 per person a year in tobacco taxes and payments from the 1998 tobacco industry master settlement agreement, they spend an average of $1.50 per person on control.
The CDC has urged states to spend $12 per person on control, Dr. Frieden said.
Dr. Lushniak noted that for current and about-to-start smokers, "the clock is ticking – they can’t wait for slow and steady progress to end the epidemic. Enough is enough."
On Twitter @aliciaault
While smoking rates have dropped precipitously since the landmark 1964 Surgeon General’s report, "Smoking and Health," smoking is still the leading cause of preventable disease and death in the United States and is now causally linked to additional diseases and conditions across most organ systems.
A new Surgeon General's report, "The Health Consequences of Smoking – 50 Years of Progress," released at a White House event Jan. 17, synthesizes original and review evidence in an effort to further federal antismoking efforts.
The report causally links cigarette smoking to type 2 diabetes, rheumatoid arthritis, ectopic pregnancy, and erectile dysfunction. Secondhand smoke is now causally linked to cancers, respiratory diseases, and cardiovascular diseases, as well as adverse effects on the health of children, the authors wrote.
In addition, the 980-page report establishes that secondhand smoke is a cause of stroke, and that smoking increases the risk of dying in cancer patients and cancer survivors.
Another finding: Cigarette smokers today have a higher risk of lung cancer than did those who smoked in 1964, because of the higher number of chemical additives now used.
Women smokers now have the same risk of death from lung cancer as that of men and a higher relative risk of dying from coronary heart disease than that of men. Because of smoking, the number of women dying from chronic obstructive pulmonary disease (COPD) is now higher than in men.
With this report, the federal government is launching a new effort to prevent children from using tobacco.
"Today, we’re asking Americans to join a sustained effort to make the next generation a tobacco-free generation," Health and Human Services Secretary Kathleen Sebelius said in a statement. "This is not something the federal government can do alone. We need to partner with the business community, local elected officials, schools and universities, the medical community, the faith community, and committed citizens in communities across the country to make the next generation tobacco free."
The report finds that youth smoking rates declined by 50% between 1997 and 2011, but 3,200 children under age 18 still start smoking each day, and an additional 2,100 youth and young adults become daily smokers. The report places most of the blame for continued interest in smoking on the tobacco industry, saying it has used "aggressive strategies" to deliberately mislead the public about the harms of smoking.
Acting Surgeon General Boris Lushniak noted that smoking rates are disproportionately higher among people with less education and lower incomes, among the mentally ill, and among gay, lesbian, bisexual, and transgender individuals.
Dr. Lushniak and other officials at the White House event called for greater tobacco control efforts, including stricter regulation. The Food and Drug Administration was given the power to regulate tobacco through the 2009 Family Smoking Prevention and Tobacco Control Act.
FDA Commissioner Margaret Hamburg said that the agency is ready to take action.
"The FDA is funding and conducting regulatory science research on tobacco products, enforcing the laws that reduce the access and attractiveness of tobacco products to young people, and preparing to launch an unprecedented national public education campaign to prevent youth tobacco use," Dr. Hamburg said in a statement.
Dr. Thomas R. Frieden, director of the Centers for Disease Control and Prevention, noted that while states collect some $80 per person a year in tobacco taxes and payments from the 1998 tobacco industry master settlement agreement, they spend an average of $1.50 per person on control.
The CDC has urged states to spend $12 per person on control, Dr. Frieden said.
Dr. Lushniak noted that for current and about-to-start smokers, "the clock is ticking – they can’t wait for slow and steady progress to end the epidemic. Enough is enough."
On Twitter @aliciaault
While smoking rates have dropped precipitously since the landmark 1964 Surgeon General’s report, "Smoking and Health," smoking is still the leading cause of preventable disease and death in the United States and is now causally linked to additional diseases and conditions across most organ systems.
A new Surgeon General's report, "The Health Consequences of Smoking – 50 Years of Progress," released at a White House event Jan. 17, synthesizes original and review evidence in an effort to further federal antismoking efforts.
The report causally links cigarette smoking to type 2 diabetes, rheumatoid arthritis, ectopic pregnancy, and erectile dysfunction. Secondhand smoke is now causally linked to cancers, respiratory diseases, and cardiovascular diseases, as well as adverse effects on the health of children, the authors wrote.
In addition, the 980-page report establishes that secondhand smoke is a cause of stroke, and that smoking increases the risk of dying in cancer patients and cancer survivors.
Another finding: Cigarette smokers today have a higher risk of lung cancer than did those who smoked in 1964, because of the higher number of chemical additives now used.
Women smokers now have the same risk of death from lung cancer as that of men and a higher relative risk of dying from coronary heart disease than that of men. Because of smoking, the number of women dying from chronic obstructive pulmonary disease (COPD) is now higher than in men.
With this report, the federal government is launching a new effort to prevent children from using tobacco.
"Today, we’re asking Americans to join a sustained effort to make the next generation a tobacco-free generation," Health and Human Services Secretary Kathleen Sebelius said in a statement. "This is not something the federal government can do alone. We need to partner with the business community, local elected officials, schools and universities, the medical community, the faith community, and committed citizens in communities across the country to make the next generation tobacco free."
The report finds that youth smoking rates declined by 50% between 1997 and 2011, but 3,200 children under age 18 still start smoking each day, and an additional 2,100 youth and young adults become daily smokers. The report places most of the blame for continued interest in smoking on the tobacco industry, saying it has used "aggressive strategies" to deliberately mislead the public about the harms of smoking.
Acting Surgeon General Boris Lushniak noted that smoking rates are disproportionately higher among people with less education and lower incomes, among the mentally ill, and among gay, lesbian, bisexual, and transgender individuals.
Dr. Lushniak and other officials at the White House event called for greater tobacco control efforts, including stricter regulation. The Food and Drug Administration was given the power to regulate tobacco through the 2009 Family Smoking Prevention and Tobacco Control Act.
FDA Commissioner Margaret Hamburg said that the agency is ready to take action.
"The FDA is funding and conducting regulatory science research on tobacco products, enforcing the laws that reduce the access and attractiveness of tobacco products to young people, and preparing to launch an unprecedented national public education campaign to prevent youth tobacco use," Dr. Hamburg said in a statement.
Dr. Thomas R. Frieden, director of the Centers for Disease Control and Prevention, noted that while states collect some $80 per person a year in tobacco taxes and payments from the 1998 tobacco industry master settlement agreement, they spend an average of $1.50 per person on control.
The CDC has urged states to spend $12 per person on control, Dr. Frieden said.
Dr. Lushniak noted that for current and about-to-start smokers, "the clock is ticking – they can’t wait for slow and steady progress to end the epidemic. Enough is enough."
On Twitter @aliciaault
USPSTF calls for gestational diabetes screening after 24 weeks
All pregnant women should be screened for gestational diabetes after 24 weeks of gestation, according to a final recommendation statement from the U.S. Preventive Services Task Force.
The updated statement, published online Jan. 13 in Annals of Internal Medicine, represents "a change in thinking about how the collective benefits of preventing complications during pregnancy and birth – such as birth injuries, preeclampsia, and babies growing too large in the womb – are important according to Dr. Virginia A. Moyer, task force chair.
"The USPSTF determined that screening for and treatment of GDM [gestational diabetes] in women after 24 weeks of gestation are associated with moderate health improvements in the mother and infant through the collective reduction in preeclampsia, macrosomia, and shoulder dystocia. The harms of screening or treatment are considered no greater than small. Therefore, the USPSTF concludes with moderate certainty that the overall net benefit is moderate," Dr. Moyer wrote on behalf of the task force.
The recommendation to screen after 24 weeks is a "B recommendation," based on a systematic review of the literature.
The final recommendation updates the task force’s 2008 recommendation. At that time, USPSTF found insufficient evidence to recommend screening of asymptomatic women, and issued an "I recommendation."
GDM prevalence in the United States is between 1% and 25%, depending on patient demographics and diagnostic thresholds, according to the statement.
Women with GDM are at increased risk for developing type 2 diabetes mellitus, with 15%-60% developing type 2 diabetes within 5-15 years of delivery, Dr. Moyer said in an interview.
"Screening women after 24 weeks of pregnancy helps to identify the disease in women earlier so that they can work with their doctors to identify whether treatment is needed," she added, noting that evidence demonstrates that treatment for gestational diabetes reduces complications.
Evidence reviewed by USPSTF indicates that primary care providers can accurately detect GDM after 24 weeks’ gestation using the two-step 50-g oral glucose challenge test, followed by the oral glucose tolerance test if the screening threshold is met or exceeded (130 mg/dL, 135 mg/dL, or 140 mg/dL [7.21, 7.49, or 7.77 mmol/L]).
In 2013, the American Congress of Obstetricians and Gynecologists recommended use of the two-step approach using the 50-g oral glucose challenge test. Although the American Diabetes Association endorsed the one-step 75-g, 2-hour oral glucose tolerance test, an independent panel supported by the National Institute of Health Consensus Development Program released a draft statement that evidence is insufficient for adopting the one-step approach.
Recommendations of the American Academy of Family Physicians and the Endocrine Society are in line with the updated USPSTF recommendation.
"Clinicians now have good evidence to support continuing to screen pregnant women for diabetes after 24 weeks of pregnancy," Dr. Moyer said.
Although high-risk women can be screened earlier than 24 weeks, the evidence regarding earlier screening remains insufficient for assessing the balance of benefits and risks asymptomatic women, thus the task force issued an "I statement."
Dr. Moyer reported no relevant conflicts of interest.
All pregnant women should be screened for gestational diabetes after 24 weeks of gestation, according to a final recommendation statement from the U.S. Preventive Services Task Force.
The updated statement, published online Jan. 13 in Annals of Internal Medicine, represents "a change in thinking about how the collective benefits of preventing complications during pregnancy and birth – such as birth injuries, preeclampsia, and babies growing too large in the womb – are important according to Dr. Virginia A. Moyer, task force chair.
"The USPSTF determined that screening for and treatment of GDM [gestational diabetes] in women after 24 weeks of gestation are associated with moderate health improvements in the mother and infant through the collective reduction in preeclampsia, macrosomia, and shoulder dystocia. The harms of screening or treatment are considered no greater than small. Therefore, the USPSTF concludes with moderate certainty that the overall net benefit is moderate," Dr. Moyer wrote on behalf of the task force.
The recommendation to screen after 24 weeks is a "B recommendation," based on a systematic review of the literature.
The final recommendation updates the task force’s 2008 recommendation. At that time, USPSTF found insufficient evidence to recommend screening of asymptomatic women, and issued an "I recommendation."
GDM prevalence in the United States is between 1% and 25%, depending on patient demographics and diagnostic thresholds, according to the statement.
Women with GDM are at increased risk for developing type 2 diabetes mellitus, with 15%-60% developing type 2 diabetes within 5-15 years of delivery, Dr. Moyer said in an interview.
"Screening women after 24 weeks of pregnancy helps to identify the disease in women earlier so that they can work with their doctors to identify whether treatment is needed," she added, noting that evidence demonstrates that treatment for gestational diabetes reduces complications.
Evidence reviewed by USPSTF indicates that primary care providers can accurately detect GDM after 24 weeks’ gestation using the two-step 50-g oral glucose challenge test, followed by the oral glucose tolerance test if the screening threshold is met or exceeded (130 mg/dL, 135 mg/dL, or 140 mg/dL [7.21, 7.49, or 7.77 mmol/L]).
In 2013, the American Congress of Obstetricians and Gynecologists recommended use of the two-step approach using the 50-g oral glucose challenge test. Although the American Diabetes Association endorsed the one-step 75-g, 2-hour oral glucose tolerance test, an independent panel supported by the National Institute of Health Consensus Development Program released a draft statement that evidence is insufficient for adopting the one-step approach.
Recommendations of the American Academy of Family Physicians and the Endocrine Society are in line with the updated USPSTF recommendation.
"Clinicians now have good evidence to support continuing to screen pregnant women for diabetes after 24 weeks of pregnancy," Dr. Moyer said.
Although high-risk women can be screened earlier than 24 weeks, the evidence regarding earlier screening remains insufficient for assessing the balance of benefits and risks asymptomatic women, thus the task force issued an "I statement."
Dr. Moyer reported no relevant conflicts of interest.
All pregnant women should be screened for gestational diabetes after 24 weeks of gestation, according to a final recommendation statement from the U.S. Preventive Services Task Force.
The updated statement, published online Jan. 13 in Annals of Internal Medicine, represents "a change in thinking about how the collective benefits of preventing complications during pregnancy and birth – such as birth injuries, preeclampsia, and babies growing too large in the womb – are important according to Dr. Virginia A. Moyer, task force chair.
"The USPSTF determined that screening for and treatment of GDM [gestational diabetes] in women after 24 weeks of gestation are associated with moderate health improvements in the mother and infant through the collective reduction in preeclampsia, macrosomia, and shoulder dystocia. The harms of screening or treatment are considered no greater than small. Therefore, the USPSTF concludes with moderate certainty that the overall net benefit is moderate," Dr. Moyer wrote on behalf of the task force.
The recommendation to screen after 24 weeks is a "B recommendation," based on a systematic review of the literature.
The final recommendation updates the task force’s 2008 recommendation. At that time, USPSTF found insufficient evidence to recommend screening of asymptomatic women, and issued an "I recommendation."
GDM prevalence in the United States is between 1% and 25%, depending on patient demographics and diagnostic thresholds, according to the statement.
Women with GDM are at increased risk for developing type 2 diabetes mellitus, with 15%-60% developing type 2 diabetes within 5-15 years of delivery, Dr. Moyer said in an interview.
"Screening women after 24 weeks of pregnancy helps to identify the disease in women earlier so that they can work with their doctors to identify whether treatment is needed," she added, noting that evidence demonstrates that treatment for gestational diabetes reduces complications.
Evidence reviewed by USPSTF indicates that primary care providers can accurately detect GDM after 24 weeks’ gestation using the two-step 50-g oral glucose challenge test, followed by the oral glucose tolerance test if the screening threshold is met or exceeded (130 mg/dL, 135 mg/dL, or 140 mg/dL [7.21, 7.49, or 7.77 mmol/L]).
In 2013, the American Congress of Obstetricians and Gynecologists recommended use of the two-step approach using the 50-g oral glucose challenge test. Although the American Diabetes Association endorsed the one-step 75-g, 2-hour oral glucose tolerance test, an independent panel supported by the National Institute of Health Consensus Development Program released a draft statement that evidence is insufficient for adopting the one-step approach.
Recommendations of the American Academy of Family Physicians and the Endocrine Society are in line with the updated USPSTF recommendation.
"Clinicians now have good evidence to support continuing to screen pregnant women for diabetes after 24 weeks of pregnancy," Dr. Moyer said.
Although high-risk women can be screened earlier than 24 weeks, the evidence regarding earlier screening remains insufficient for assessing the balance of benefits and risks asymptomatic women, thus the task force issued an "I statement."
Dr. Moyer reported no relevant conflicts of interest.
FROM ANNALS OF INTERNAL MEDICINE
Postpartum care after gestational diabetes is preconception care as well
WASHINGTON – The rise in gestational diabetes cases demands new strategies for active postpartum care that will not only assist with prevention of type 2 diabetes, but will also prepare women for healthy subsequent pregnancies. New approaches to postpartum care aimed at preventing gestational diabetes in subsequent pregnancies were showcased during sessions at the annual meeting of the Diabetes in Pregnancy Study Group of North America.
The problem is a growing one, with the prevalence of gestational diabetes mellitus (GDM) pregnancies ranging from 2% to 10% depending on screening criteria and demographics/population. The prevalence could rise as high as 18% if diagnostic criteria change in the United States to implement the single-step test recommended in 2008 by the International Association of Diabetes and Pregnancy Study Groups (IADPSG) and adopted by the American Diabetes Association and the World Health Organization.
For now, most U.S. obstetricians are continuing to use the two-step approach to diagnosing GDM, following the advice of a National Institutes of Health consensus development panel and the American College of Gynecologists and Obstetricians. Some physicians, however, are lowering their diagnostic thresholds to capture more at-risk women.
And in the meantime, the prevalence of obesity continues to rise, independently increasing the number of pregnancies complicated by GDM. An estimated 50% of GDM is caused by overweight and obesity, according to Dr. Wanda K. Nicholson, director of the diabetes and obesity core at the University of North Carolina’s Center for Women's Health Research in Chapel Hill.
"We’re not even able to manage our current body of patients," she said. "If we expand this number, we’ll definitely need to develop some new strategies." Studies have also shown that approximately 50% of patients with GDM are screened after delivery for persistent glucose intolerance.
"For most of us, preconception care is increasingly about postpartum care," said Dr. Thomas R. Moore, professor and chairman of the department of reproductive medicine at the University of California, San Diego. "We have to find ways of improving our postpartum follow-up [of GDM pregnancies]."
Follow-up should include checking glycemic status, educating and counseling patients on the risks to newborns of both high maternal HbA1c levels and maternal obesity, assisting patients with nutritional and lifestyle change, advising the use of effective contraception until conditions are right for another pregnancy, and ensuring proper folic acid supplementation before conception is attempted, he said.
For ob.gyn. practices, more GDM can mean more physician appointments, more staff dedicated to coordinating visits with physicians and educators, expanded diabetes education, and expanded nutritional counseling. "In particular, many practices will need to expand their capacity for nutritional counseling," Dr. Nicholson said. "We know from large randomized trials that even for women with mild GDM, nutritional counseling makes a difference."
The literature hints at the possible promise of using telephonic nurse management and Internet-based self-monitoring and educational tools to enhance the postpartum care of new mothers who had GDM, she said.
At the University of North Carolina, a program called the Gestational Diabetes Management System (GooDMomS) offers women video-supported lessons on GDM, a Web and mobile self-monitoring diary in which they can track their weight and record time spent exercising postpartum, as well as forums to "ask the staff" questions and to communicate with other mothers. The system, which is being pilot-tested in an NIH-funded randomized study, is meant to enhance – not replace – basic postpartum care.
In terms of postpartum testing for type 2 diabetes, a recently published literature review suggests that systematic efforts to proactively contact patients can have an impact. Programs in which patients were contacted via phone calls, mailings, and other reminders increased postpartum testing rates from an average of 33% up to 60% (Prim. Care Diabetes 2013;7:177-86).
"We can’t even begin to deliver a preconception program to women until we know what her glycemic status is," emphasized Dr. Moore.
A meta-analysis published more than a decade ago showed an overall 80% reduction in the incidence of congenital malformations in women with diabetes who received preconception care as compared to those who did not receive such care. This study has increasingly relevance today, with more than 76 million women worldwide at risk of their pregnancies being complicated by pregestational (existing) diabetes or gestational diabetes, he said.
The analysis (QJM 2001;94:435-44) also showed that "only modest reductions in HbA1c, averaging 17%, can have a profound impact," Dr. Moore said. The patient conversation must go beyond merely recommending that HbA1c levels be as close to normal as possible before conception is attempted, he emphasized.
A recent report from the Maryland Pregnancy Risk Assessment Monitoring System (PRAMS) shows that women with diabetes were the least likely group among women with various preconception medical problems to report contraception use (Contraception 2013;88:263-8). "They had the lowest uptake on contraception," Dr. Moore said.
An estimate of the "downstream savings" of preventing type 2 diabetes in women with GDM was factored into one of two true cost-effectiveness analyses that have been published thus far of the IADPSG criteria, according to Dr. Aaron B. Caughey, chairman of the department of obstetrics and gynecology at the Oregon Health and Science University in Portland.
Both of the two published analyses concluded that the IADPSG criteria are cost effective, but one of them concluded that the criteria would be cost effective only if post-delivery care reduces diabetes incidence (Diabetes Care 2012;35:529-35).
"The short-term costs (of the one-step approach) are easy to figure out," said Dr. Caughey, an ob.gyn and health economist who also is director of the university’s Center for Women’s Health. "The long-term costs are really tough ... we don’t know, what the impact 20 years downstream is going to be."
Providers at Oregon Health and Science University have been encouraged to adopt the one-step approach and, thus far, the change has resulted in a doubling of GDM incidence from approximately 6% to 12%, he noted.
Dr. Caughey, Dr. Moore, and Dr. Nicholson each reported that they had no conflicts of interest to disclose.
WASHINGTON – The rise in gestational diabetes cases demands new strategies for active postpartum care that will not only assist with prevention of type 2 diabetes, but will also prepare women for healthy subsequent pregnancies. New approaches to postpartum care aimed at preventing gestational diabetes in subsequent pregnancies were showcased during sessions at the annual meeting of the Diabetes in Pregnancy Study Group of North America.
The problem is a growing one, with the prevalence of gestational diabetes mellitus (GDM) pregnancies ranging from 2% to 10% depending on screening criteria and demographics/population. The prevalence could rise as high as 18% if diagnostic criteria change in the United States to implement the single-step test recommended in 2008 by the International Association of Diabetes and Pregnancy Study Groups (IADPSG) and adopted by the American Diabetes Association and the World Health Organization.
For now, most U.S. obstetricians are continuing to use the two-step approach to diagnosing GDM, following the advice of a National Institutes of Health consensus development panel and the American College of Gynecologists and Obstetricians. Some physicians, however, are lowering their diagnostic thresholds to capture more at-risk women.
And in the meantime, the prevalence of obesity continues to rise, independently increasing the number of pregnancies complicated by GDM. An estimated 50% of GDM is caused by overweight and obesity, according to Dr. Wanda K. Nicholson, director of the diabetes and obesity core at the University of North Carolina’s Center for Women's Health Research in Chapel Hill.
"We’re not even able to manage our current body of patients," she said. "If we expand this number, we’ll definitely need to develop some new strategies." Studies have also shown that approximately 50% of patients with GDM are screened after delivery for persistent glucose intolerance.
"For most of us, preconception care is increasingly about postpartum care," said Dr. Thomas R. Moore, professor and chairman of the department of reproductive medicine at the University of California, San Diego. "We have to find ways of improving our postpartum follow-up [of GDM pregnancies]."
Follow-up should include checking glycemic status, educating and counseling patients on the risks to newborns of both high maternal HbA1c levels and maternal obesity, assisting patients with nutritional and lifestyle change, advising the use of effective contraception until conditions are right for another pregnancy, and ensuring proper folic acid supplementation before conception is attempted, he said.
For ob.gyn. practices, more GDM can mean more physician appointments, more staff dedicated to coordinating visits with physicians and educators, expanded diabetes education, and expanded nutritional counseling. "In particular, many practices will need to expand their capacity for nutritional counseling," Dr. Nicholson said. "We know from large randomized trials that even for women with mild GDM, nutritional counseling makes a difference."
The literature hints at the possible promise of using telephonic nurse management and Internet-based self-monitoring and educational tools to enhance the postpartum care of new mothers who had GDM, she said.
At the University of North Carolina, a program called the Gestational Diabetes Management System (GooDMomS) offers women video-supported lessons on GDM, a Web and mobile self-monitoring diary in which they can track their weight and record time spent exercising postpartum, as well as forums to "ask the staff" questions and to communicate with other mothers. The system, which is being pilot-tested in an NIH-funded randomized study, is meant to enhance – not replace – basic postpartum care.
In terms of postpartum testing for type 2 diabetes, a recently published literature review suggests that systematic efforts to proactively contact patients can have an impact. Programs in which patients were contacted via phone calls, mailings, and other reminders increased postpartum testing rates from an average of 33% up to 60% (Prim. Care Diabetes 2013;7:177-86).
"We can’t even begin to deliver a preconception program to women until we know what her glycemic status is," emphasized Dr. Moore.
A meta-analysis published more than a decade ago showed an overall 80% reduction in the incidence of congenital malformations in women with diabetes who received preconception care as compared to those who did not receive such care. This study has increasingly relevance today, with more than 76 million women worldwide at risk of their pregnancies being complicated by pregestational (existing) diabetes or gestational diabetes, he said.
The analysis (QJM 2001;94:435-44) also showed that "only modest reductions in HbA1c, averaging 17%, can have a profound impact," Dr. Moore said. The patient conversation must go beyond merely recommending that HbA1c levels be as close to normal as possible before conception is attempted, he emphasized.
A recent report from the Maryland Pregnancy Risk Assessment Monitoring System (PRAMS) shows that women with diabetes were the least likely group among women with various preconception medical problems to report contraception use (Contraception 2013;88:263-8). "They had the lowest uptake on contraception," Dr. Moore said.
An estimate of the "downstream savings" of preventing type 2 diabetes in women with GDM was factored into one of two true cost-effectiveness analyses that have been published thus far of the IADPSG criteria, according to Dr. Aaron B. Caughey, chairman of the department of obstetrics and gynecology at the Oregon Health and Science University in Portland.
Both of the two published analyses concluded that the IADPSG criteria are cost effective, but one of them concluded that the criteria would be cost effective only if post-delivery care reduces diabetes incidence (Diabetes Care 2012;35:529-35).
"The short-term costs (of the one-step approach) are easy to figure out," said Dr. Caughey, an ob.gyn and health economist who also is director of the university’s Center for Women’s Health. "The long-term costs are really tough ... we don’t know, what the impact 20 years downstream is going to be."
Providers at Oregon Health and Science University have been encouraged to adopt the one-step approach and, thus far, the change has resulted in a doubling of GDM incidence from approximately 6% to 12%, he noted.
Dr. Caughey, Dr. Moore, and Dr. Nicholson each reported that they had no conflicts of interest to disclose.
WASHINGTON – The rise in gestational diabetes cases demands new strategies for active postpartum care that will not only assist with prevention of type 2 diabetes, but will also prepare women for healthy subsequent pregnancies. New approaches to postpartum care aimed at preventing gestational diabetes in subsequent pregnancies were showcased during sessions at the annual meeting of the Diabetes in Pregnancy Study Group of North America.
The problem is a growing one, with the prevalence of gestational diabetes mellitus (GDM) pregnancies ranging from 2% to 10% depending on screening criteria and demographics/population. The prevalence could rise as high as 18% if diagnostic criteria change in the United States to implement the single-step test recommended in 2008 by the International Association of Diabetes and Pregnancy Study Groups (IADPSG) and adopted by the American Diabetes Association and the World Health Organization.
For now, most U.S. obstetricians are continuing to use the two-step approach to diagnosing GDM, following the advice of a National Institutes of Health consensus development panel and the American College of Gynecologists and Obstetricians. Some physicians, however, are lowering their diagnostic thresholds to capture more at-risk women.
And in the meantime, the prevalence of obesity continues to rise, independently increasing the number of pregnancies complicated by GDM. An estimated 50% of GDM is caused by overweight and obesity, according to Dr. Wanda K. Nicholson, director of the diabetes and obesity core at the University of North Carolina’s Center for Women's Health Research in Chapel Hill.
"We’re not even able to manage our current body of patients," she said. "If we expand this number, we’ll definitely need to develop some new strategies." Studies have also shown that approximately 50% of patients with GDM are screened after delivery for persistent glucose intolerance.
"For most of us, preconception care is increasingly about postpartum care," said Dr. Thomas R. Moore, professor and chairman of the department of reproductive medicine at the University of California, San Diego. "We have to find ways of improving our postpartum follow-up [of GDM pregnancies]."
Follow-up should include checking glycemic status, educating and counseling patients on the risks to newborns of both high maternal HbA1c levels and maternal obesity, assisting patients with nutritional and lifestyle change, advising the use of effective contraception until conditions are right for another pregnancy, and ensuring proper folic acid supplementation before conception is attempted, he said.
For ob.gyn. practices, more GDM can mean more physician appointments, more staff dedicated to coordinating visits with physicians and educators, expanded diabetes education, and expanded nutritional counseling. "In particular, many practices will need to expand their capacity for nutritional counseling," Dr. Nicholson said. "We know from large randomized trials that even for women with mild GDM, nutritional counseling makes a difference."
The literature hints at the possible promise of using telephonic nurse management and Internet-based self-monitoring and educational tools to enhance the postpartum care of new mothers who had GDM, she said.
At the University of North Carolina, a program called the Gestational Diabetes Management System (GooDMomS) offers women video-supported lessons on GDM, a Web and mobile self-monitoring diary in which they can track their weight and record time spent exercising postpartum, as well as forums to "ask the staff" questions and to communicate with other mothers. The system, which is being pilot-tested in an NIH-funded randomized study, is meant to enhance – not replace – basic postpartum care.
In terms of postpartum testing for type 2 diabetes, a recently published literature review suggests that systematic efforts to proactively contact patients can have an impact. Programs in which patients were contacted via phone calls, mailings, and other reminders increased postpartum testing rates from an average of 33% up to 60% (Prim. Care Diabetes 2013;7:177-86).
"We can’t even begin to deliver a preconception program to women until we know what her glycemic status is," emphasized Dr. Moore.
A meta-analysis published more than a decade ago showed an overall 80% reduction in the incidence of congenital malformations in women with diabetes who received preconception care as compared to those who did not receive such care. This study has increasingly relevance today, with more than 76 million women worldwide at risk of their pregnancies being complicated by pregestational (existing) diabetes or gestational diabetes, he said.
The analysis (QJM 2001;94:435-44) also showed that "only modest reductions in HbA1c, averaging 17%, can have a profound impact," Dr. Moore said. The patient conversation must go beyond merely recommending that HbA1c levels be as close to normal as possible before conception is attempted, he emphasized.
A recent report from the Maryland Pregnancy Risk Assessment Monitoring System (PRAMS) shows that women with diabetes were the least likely group among women with various preconception medical problems to report contraception use (Contraception 2013;88:263-8). "They had the lowest uptake on contraception," Dr. Moore said.
An estimate of the "downstream savings" of preventing type 2 diabetes in women with GDM was factored into one of two true cost-effectiveness analyses that have been published thus far of the IADPSG criteria, according to Dr. Aaron B. Caughey, chairman of the department of obstetrics and gynecology at the Oregon Health and Science University in Portland.
Both of the two published analyses concluded that the IADPSG criteria are cost effective, but one of them concluded that the criteria would be cost effective only if post-delivery care reduces diabetes incidence (Diabetes Care 2012;35:529-35).
"The short-term costs (of the one-step approach) are easy to figure out," said Dr. Caughey, an ob.gyn and health economist who also is director of the university’s Center for Women’s Health. "The long-term costs are really tough ... we don’t know, what the impact 20 years downstream is going to be."
Providers at Oregon Health and Science University have been encouraged to adopt the one-step approach and, thus far, the change has resulted in a doubling of GDM incidence from approximately 6% to 12%, he noted.
Dr. Caughey, Dr. Moore, and Dr. Nicholson each reported that they had no conflicts of interest to disclose.
AT THE ANNUAL MEETING OF THE DIABETES IN PREGNANCY STUDY GROUP
In utero exposure to dyslipidemia magnifies LDL risk in offspring
DALLAS – Elevated LDL cholesterol in women before childbirth was associated with a fivefold increased risk of elevated LDL 2 decades later in their young adult offspring in a new analysis from the Framingham Heart Study.
In contrast, paternal elevation of LDL pre pregnancy was not associated with increased likelihood of hyperlipidemia in children at age 20 years.
In utero exposure to maternal dyslipidemia appears to have lasting adverse consequences in terms of cardiovascular disease risk. If confirmed, the implications of the findings are huge, given that an estimated 25% of American women of childbearing age have elevated LDL, according to National Health and Nutrition Examination Survey data, Dr. Michael M. Mendelson noted at the American Heart Association scientific sessions.
"We postulate that identifying young women of childbearing age with dyslipidemia and reducing abnormal LDL with lipid-specific healthy lifestyle interventions may further reduce the transgenerational cycle of dyslipidemia and cardiovascular disease risk," declared Dr. Mendelson of Boston Children’s Hospital.
He presented prospectively acquired data on 343 maternal-child pairs enrolled in the Framingham Heart Study. Parental serum lipids were measured roughly 3 years prior to childbirth and again 20 years later, when the now young-adult offspring also had their serum lipids measured as part of their first comprehensive assessment as Framingham participants.
Women with an LDL level greater than 130 mg/dL at their prebirth assessment were fivefold more likely to have young-adult offspring with an elevated LDL in a multivariate analysis adjusted for maternal age and offspring gender. With further adjustment for prepregnancy maternal body mass index, smoking status, and genetic variants known to be associated with LDL level – most notably familial hypercholesterolemia – maternal dyslipidemia pre pregnancy remained associated with a highly significant 3.7-fold increased risk of dyslipidemia in young-adult offspring (P = .004).
In contrast, high paternal LDL pre pregnancy was not associated with increased odds of adult dyslipidemia in the offspring. And neither high maternal nor paternal LDL measured 20 years after childbirth was linked to increased likelihood of dyslipidemia in 20-year-old children in the fully adjusted multivariate logistic regression analysis.
The Framingham Heart Study is funded by the National Institutes of Health. Dr. Mendelson reported having no financial conflicts of interest.
DALLAS – Elevated LDL cholesterol in women before childbirth was associated with a fivefold increased risk of elevated LDL 2 decades later in their young adult offspring in a new analysis from the Framingham Heart Study.
In contrast, paternal elevation of LDL pre pregnancy was not associated with increased likelihood of hyperlipidemia in children at age 20 years.
In utero exposure to maternal dyslipidemia appears to have lasting adverse consequences in terms of cardiovascular disease risk. If confirmed, the implications of the findings are huge, given that an estimated 25% of American women of childbearing age have elevated LDL, according to National Health and Nutrition Examination Survey data, Dr. Michael M. Mendelson noted at the American Heart Association scientific sessions.
"We postulate that identifying young women of childbearing age with dyslipidemia and reducing abnormal LDL with lipid-specific healthy lifestyle interventions may further reduce the transgenerational cycle of dyslipidemia and cardiovascular disease risk," declared Dr. Mendelson of Boston Children’s Hospital.
He presented prospectively acquired data on 343 maternal-child pairs enrolled in the Framingham Heart Study. Parental serum lipids were measured roughly 3 years prior to childbirth and again 20 years later, when the now young-adult offspring also had their serum lipids measured as part of their first comprehensive assessment as Framingham participants.
Women with an LDL level greater than 130 mg/dL at their prebirth assessment were fivefold more likely to have young-adult offspring with an elevated LDL in a multivariate analysis adjusted for maternal age and offspring gender. With further adjustment for prepregnancy maternal body mass index, smoking status, and genetic variants known to be associated with LDL level – most notably familial hypercholesterolemia – maternal dyslipidemia pre pregnancy remained associated with a highly significant 3.7-fold increased risk of dyslipidemia in young-adult offspring (P = .004).
In contrast, high paternal LDL pre pregnancy was not associated with increased odds of adult dyslipidemia in the offspring. And neither high maternal nor paternal LDL measured 20 years after childbirth was linked to increased likelihood of dyslipidemia in 20-year-old children in the fully adjusted multivariate logistic regression analysis.
The Framingham Heart Study is funded by the National Institutes of Health. Dr. Mendelson reported having no financial conflicts of interest.
DALLAS – Elevated LDL cholesterol in women before childbirth was associated with a fivefold increased risk of elevated LDL 2 decades later in their young adult offspring in a new analysis from the Framingham Heart Study.
In contrast, paternal elevation of LDL pre pregnancy was not associated with increased likelihood of hyperlipidemia in children at age 20 years.
In utero exposure to maternal dyslipidemia appears to have lasting adverse consequences in terms of cardiovascular disease risk. If confirmed, the implications of the findings are huge, given that an estimated 25% of American women of childbearing age have elevated LDL, according to National Health and Nutrition Examination Survey data, Dr. Michael M. Mendelson noted at the American Heart Association scientific sessions.
"We postulate that identifying young women of childbearing age with dyslipidemia and reducing abnormal LDL with lipid-specific healthy lifestyle interventions may further reduce the transgenerational cycle of dyslipidemia and cardiovascular disease risk," declared Dr. Mendelson of Boston Children’s Hospital.
He presented prospectively acquired data on 343 maternal-child pairs enrolled in the Framingham Heart Study. Parental serum lipids were measured roughly 3 years prior to childbirth and again 20 years later, when the now young-adult offspring also had their serum lipids measured as part of their first comprehensive assessment as Framingham participants.
Women with an LDL level greater than 130 mg/dL at their prebirth assessment were fivefold more likely to have young-adult offspring with an elevated LDL in a multivariate analysis adjusted for maternal age and offspring gender. With further adjustment for prepregnancy maternal body mass index, smoking status, and genetic variants known to be associated with LDL level – most notably familial hypercholesterolemia – maternal dyslipidemia pre pregnancy remained associated with a highly significant 3.7-fold increased risk of dyslipidemia in young-adult offspring (P = .004).
In contrast, high paternal LDL pre pregnancy was not associated with increased odds of adult dyslipidemia in the offspring. And neither high maternal nor paternal LDL measured 20 years after childbirth was linked to increased likelihood of dyslipidemia in 20-year-old children in the fully adjusted multivariate logistic regression analysis.
The Framingham Heart Study is funded by the National Institutes of Health. Dr. Mendelson reported having no financial conflicts of interest.
AT THE AHA SCIENTIFIC SESSIONS
Major finding: With further adjustment for prepregnancy maternal body mass index, smoking status, and genetic variants known to be associated with LDL level – most notably familial hypercholesterolemia – maternal dyslipidemia before pregnancy remained associated with a highly significant 3.7-fold increased risk of dyslipidemia in young-adult offspring (P = .004).
Data source: Prospectively acquired data on 343 maternal-child pairs enrolled in the Framingham Heart Study.
Disclosures: The study is funded by the National Institutes of Health. The presenter reported having no financial disclosures.
The latest guidance from ACOG on hypertension in pregnancy
In November 2013, the American College of Obstetricians and Gynecologists (ACOG) published a 99-page report on hypertensive disorders in pregnancy. The classification, diagnosis, prediction, prevention, and management of gestational hypertension, preeclampsia, and chronic hypertension were addressed, although preeclampsia was the main focus.
In this audiocast, Dr. Pauli sifts through the guidelines and discusses the changes that will be implemented at her institution.
Dr. Pauli and John T. Repke, MD, are co-authors of the Update on Obstetrics (January 2014).
In November 2013, the American College of Obstetricians and Gynecologists (ACOG) published a 99-page report on hypertensive disorders in pregnancy. The classification, diagnosis, prediction, prevention, and management of gestational hypertension, preeclampsia, and chronic hypertension were addressed, although preeclampsia was the main focus.
In this audiocast, Dr. Pauli sifts through the guidelines and discusses the changes that will be implemented at her institution.
Dr. Pauli and John T. Repke, MD, are co-authors of the Update on Obstetrics (January 2014).
In November 2013, the American College of Obstetricians and Gynecologists (ACOG) published a 99-page report on hypertensive disorders in pregnancy. The classification, diagnosis, prediction, prevention, and management of gestational hypertension, preeclampsia, and chronic hypertension were addressed, although preeclampsia was the main focus.
In this audiocast, Dr. Pauli sifts through the guidelines and discusses the changes that will be implemented at her institution.
Dr. Pauli and John T. Repke, MD, are co-authors of the Update on Obstetrics (January 2014).
Wide variation found in early pregnancy beta-HCG values
SAN FRANCISCO – Beta-HCG values can vary widely in early pregnancy, and should not be used in isolation to declare a pregnancy to be abnormal, according to Dr. Jody Steinauer.
In 366 women with vaginal bleeding or pain who went on to have a normal intrauterine pregnancy, the lowest level of beta-HCG at which an intrauterine pregnancy could be seen was 390 mIU/mL. The same study reported that women with beta-HCG levels as high as 3,510 mIU/mL in whom no gestational sac can be seen may still have normal pregnancies (Obstet. Gynecol. 2013;121:65-70).
The widely accepted upper cutoff value is 1,500-2,000 mIU/mL, Dr. Steinauer said at a conference on women’s health sponsored by the University of California, San Francisco. Yet "it’s not a hard-and-fast rule. We have all heard of patients who had even higher beta-HCG levels and went on to have normal singleton pregnancies." On the lower end of beta-HCG values, the attitude may be "her beta-HCG is only 700 mIU/mL, so I’m not expecting to see a gestational sac" on ultrasound. Dr. Steinauer said.
In the current study, using a beta-HCG cutoff of 1,500 mIU/mL when no gestational sac can be seen would identify 80% of normal intrauterine pregnancies. A 2,000-mIU/mL cutoff would identify 91% of normal pregnancies. With either cutoff value, "there would have been a high proportion of normal pregnancies that were called abnormal," said Dr. Steinauer of the university.
New criteria released by the Society of Radiologists in Ultrasound for diagnosing a nonviable pregnancy early in the first trimester also address beta-HCG levels. If no gestational sac is visible at a beta-HCG level greater than 2,000 mIU/mL, an ectopic pregnancy is 19 times more likely than a viable pregnancy, but a nonviable intrauterine pregnancy still is twice as likely as an ectopic pregnancy (N. Engl. J. Med. 2013;369:1443-51).
Those estimates apply to the general U.S. population, Dr. Steinauer said, and may vary depending on patient population.
In women with beta-HCG levels of 2,000-3,000 mIU/mL, there will be 19 ectopic pregnancies and 38 nonviable pregnancies for every viable pregnancy. Yet as many as 2% of women with values this high can have viable pregnancies, according to the society’s criteria. Once the beta-HCG level climbs higher than 3,000 mIU/mL and no gestational sac is visible, an ectopic pregnancy is 70 times more likely than a viable pregnancy.
Ultrasound measurements of the yolk sac or fetal pole also can help diagnose early pregnancy loss, but these are more variable and thus less helpful than gestational sac measurements.
Studies have shown that when no embryo is seen with a mean gestational sac diameter of 2 mm, a diagnosis of early pregnancy loss will be false in 0.5% of cases in which no yolk sac is seen and in 0.4% of cases with a yolk sac. The false-positive rate approaches 0, however, with a mean gestational sac diameter of 21 mm, she said.
Several studies have shown that an inability to find cardiac activity when the fetal pole measures 5 mm does not confirm a diagnosis of early pregnancy loss because 8.3% of cases will be false positives under those criteria. A fetal pole measuring at least 5.3 mm with no cardiac activity, however, eliminates the possibility of a false-positive diagnosis.
The criteria released by the Society of Radiologists in Ultrasound express concern about the difficulty for an average ultrasonographer to detect fractions of millimeters in differences, so they allow a margin of error. The cutoff for a diagnosis of early pregnancy loss is extended to a mean gestational sac diameter of 25 mm with no embryo, or a fetal pole measuring 7 mm with no cardiac activity.
Dr. Steinauer reported having no relevant financial disclosures.
On Twitter @sherryboschert
SAN FRANCISCO – Beta-HCG values can vary widely in early pregnancy, and should not be used in isolation to declare a pregnancy to be abnormal, according to Dr. Jody Steinauer.
In 366 women with vaginal bleeding or pain who went on to have a normal intrauterine pregnancy, the lowest level of beta-HCG at which an intrauterine pregnancy could be seen was 390 mIU/mL. The same study reported that women with beta-HCG levels as high as 3,510 mIU/mL in whom no gestational sac can be seen may still have normal pregnancies (Obstet. Gynecol. 2013;121:65-70).
The widely accepted upper cutoff value is 1,500-2,000 mIU/mL, Dr. Steinauer said at a conference on women’s health sponsored by the University of California, San Francisco. Yet "it’s not a hard-and-fast rule. We have all heard of patients who had even higher beta-HCG levels and went on to have normal singleton pregnancies." On the lower end of beta-HCG values, the attitude may be "her beta-HCG is only 700 mIU/mL, so I’m not expecting to see a gestational sac" on ultrasound. Dr. Steinauer said.
In the current study, using a beta-HCG cutoff of 1,500 mIU/mL when no gestational sac can be seen would identify 80% of normal intrauterine pregnancies. A 2,000-mIU/mL cutoff would identify 91% of normal pregnancies. With either cutoff value, "there would have been a high proportion of normal pregnancies that were called abnormal," said Dr. Steinauer of the university.
New criteria released by the Society of Radiologists in Ultrasound for diagnosing a nonviable pregnancy early in the first trimester also address beta-HCG levels. If no gestational sac is visible at a beta-HCG level greater than 2,000 mIU/mL, an ectopic pregnancy is 19 times more likely than a viable pregnancy, but a nonviable intrauterine pregnancy still is twice as likely as an ectopic pregnancy (N. Engl. J. Med. 2013;369:1443-51).
Those estimates apply to the general U.S. population, Dr. Steinauer said, and may vary depending on patient population.
In women with beta-HCG levels of 2,000-3,000 mIU/mL, there will be 19 ectopic pregnancies and 38 nonviable pregnancies for every viable pregnancy. Yet as many as 2% of women with values this high can have viable pregnancies, according to the society’s criteria. Once the beta-HCG level climbs higher than 3,000 mIU/mL and no gestational sac is visible, an ectopic pregnancy is 70 times more likely than a viable pregnancy.
Ultrasound measurements of the yolk sac or fetal pole also can help diagnose early pregnancy loss, but these are more variable and thus less helpful than gestational sac measurements.
Studies have shown that when no embryo is seen with a mean gestational sac diameter of 2 mm, a diagnosis of early pregnancy loss will be false in 0.5% of cases in which no yolk sac is seen and in 0.4% of cases with a yolk sac. The false-positive rate approaches 0, however, with a mean gestational sac diameter of 21 mm, she said.
Several studies have shown that an inability to find cardiac activity when the fetal pole measures 5 mm does not confirm a diagnosis of early pregnancy loss because 8.3% of cases will be false positives under those criteria. A fetal pole measuring at least 5.3 mm with no cardiac activity, however, eliminates the possibility of a false-positive diagnosis.
The criteria released by the Society of Radiologists in Ultrasound express concern about the difficulty for an average ultrasonographer to detect fractions of millimeters in differences, so they allow a margin of error. The cutoff for a diagnosis of early pregnancy loss is extended to a mean gestational sac diameter of 25 mm with no embryo, or a fetal pole measuring 7 mm with no cardiac activity.
Dr. Steinauer reported having no relevant financial disclosures.
On Twitter @sherryboschert
SAN FRANCISCO – Beta-HCG values can vary widely in early pregnancy, and should not be used in isolation to declare a pregnancy to be abnormal, according to Dr. Jody Steinauer.
In 366 women with vaginal bleeding or pain who went on to have a normal intrauterine pregnancy, the lowest level of beta-HCG at which an intrauterine pregnancy could be seen was 390 mIU/mL. The same study reported that women with beta-HCG levels as high as 3,510 mIU/mL in whom no gestational sac can be seen may still have normal pregnancies (Obstet. Gynecol. 2013;121:65-70).
The widely accepted upper cutoff value is 1,500-2,000 mIU/mL, Dr. Steinauer said at a conference on women’s health sponsored by the University of California, San Francisco. Yet "it’s not a hard-and-fast rule. We have all heard of patients who had even higher beta-HCG levels and went on to have normal singleton pregnancies." On the lower end of beta-HCG values, the attitude may be "her beta-HCG is only 700 mIU/mL, so I’m not expecting to see a gestational sac" on ultrasound. Dr. Steinauer said.
In the current study, using a beta-HCG cutoff of 1,500 mIU/mL when no gestational sac can be seen would identify 80% of normal intrauterine pregnancies. A 2,000-mIU/mL cutoff would identify 91% of normal pregnancies. With either cutoff value, "there would have been a high proportion of normal pregnancies that were called abnormal," said Dr. Steinauer of the university.
New criteria released by the Society of Radiologists in Ultrasound for diagnosing a nonviable pregnancy early in the first trimester also address beta-HCG levels. If no gestational sac is visible at a beta-HCG level greater than 2,000 mIU/mL, an ectopic pregnancy is 19 times more likely than a viable pregnancy, but a nonviable intrauterine pregnancy still is twice as likely as an ectopic pregnancy (N. Engl. J. Med. 2013;369:1443-51).
Those estimates apply to the general U.S. population, Dr. Steinauer said, and may vary depending on patient population.
In women with beta-HCG levels of 2,000-3,000 mIU/mL, there will be 19 ectopic pregnancies and 38 nonviable pregnancies for every viable pregnancy. Yet as many as 2% of women with values this high can have viable pregnancies, according to the society’s criteria. Once the beta-HCG level climbs higher than 3,000 mIU/mL and no gestational sac is visible, an ectopic pregnancy is 70 times more likely than a viable pregnancy.
Ultrasound measurements of the yolk sac or fetal pole also can help diagnose early pregnancy loss, but these are more variable and thus less helpful than gestational sac measurements.
Studies have shown that when no embryo is seen with a mean gestational sac diameter of 2 mm, a diagnosis of early pregnancy loss will be false in 0.5% of cases in which no yolk sac is seen and in 0.4% of cases with a yolk sac. The false-positive rate approaches 0, however, with a mean gestational sac diameter of 21 mm, she said.
Several studies have shown that an inability to find cardiac activity when the fetal pole measures 5 mm does not confirm a diagnosis of early pregnancy loss because 8.3% of cases will be false positives under those criteria. A fetal pole measuring at least 5.3 mm with no cardiac activity, however, eliminates the possibility of a false-positive diagnosis.
The criteria released by the Society of Radiologists in Ultrasound express concern about the difficulty for an average ultrasonographer to detect fractions of millimeters in differences, so they allow a margin of error. The cutoff for a diagnosis of early pregnancy loss is extended to a mean gestational sac diameter of 25 mm with no embryo, or a fetal pole measuring 7 mm with no cardiac activity.
Dr. Steinauer reported having no relevant financial disclosures.
On Twitter @sherryboschert
EXPERT ANALYSIS FROM A CONFERENCE ON WOMEN’S HEALTH
CDC Resources Aid Contraceptive Selection
SAN FRANCISCO – Pop quiz: Do you know which contraceptive methods are safe for women who are postpartum or who smoke? How about for women who have diabetic nephropathy or migraine with aura?
Handy online tools and smart-phone apps from the Centers for Disease Control and Prevention now offer recommendations for contraceptive choices. The U.S. Medical Eligibility Criteria for Contraception, or MEC (MMWR 2010;59:1-6), now has a newer companion document, the U.S. Selected Practice Recommendations for Contraception, released by the U.S. Centers for Disease Control and Prevention (MMWR 2013;62:1-46), Dr. Jody Steinauer said at a conference on women’s health sponsored by the University of California, San Francisco.
The U.S. version of the MEC features four color-coded tables that compare contraceptive methods and more than 60 medical conditions or characteristics. Each pair of method and condition is scored for safety on a scale of 1-4.
A score of 1 (color-coded dark green) means there are no concerns about the method to be used in that scenario. A score of 2 (light green) means that generally the advantages outweigh the risks of the method, said Dr. Steinauer, of the university.
A score of 3 (pink) suggests there is a "relative contraindication" in which there are some concerns, but the advantages of using that contraceptive method outweigh the risks for that patient. "You might talk with the woman about using a different method and if she decides to use the method with a 3 score, you would counsel her about the risks but would still prescribe it," she said. A score of 4 (red) means there’s an absolute contraindication to the method for that condition.
The MEC is available in an app for iPhone or iPad. The app lists choices by condition or by method; the online tables can be printed out and the options viewed all at once, she said.
"We have these hanging around at our clinic, and all of the residents carry them on their iPhones as well," she said. "It’s a really useful document."
The companion Selected Practice Recommendations for Contraception provide advice on how to use contraceptive methods in clinical practice. For example, there are recommendations on how long to use a backup contraceptive method once it has been started, special considerations for each method, and what to do when there are missed or late doses.
Dr. Steinauer reported having no financial disclosures,
On Twitter @sherryboschert
SAN FRANCISCO – Pop quiz: Do you know which contraceptive methods are safe for women who are postpartum or who smoke? How about for women who have diabetic nephropathy or migraine with aura?
Handy online tools and smart-phone apps from the Centers for Disease Control and Prevention now offer recommendations for contraceptive choices. The U.S. Medical Eligibility Criteria for Contraception, or MEC (MMWR 2010;59:1-6), now has a newer companion document, the U.S. Selected Practice Recommendations for Contraception, released by the U.S. Centers for Disease Control and Prevention (MMWR 2013;62:1-46), Dr. Jody Steinauer said at a conference on women’s health sponsored by the University of California, San Francisco.
The U.S. version of the MEC features four color-coded tables that compare contraceptive methods and more than 60 medical conditions or characteristics. Each pair of method and condition is scored for safety on a scale of 1-4.
A score of 1 (color-coded dark green) means there are no concerns about the method to be used in that scenario. A score of 2 (light green) means that generally the advantages outweigh the risks of the method, said Dr. Steinauer, of the university.
A score of 3 (pink) suggests there is a "relative contraindication" in which there are some concerns, but the advantages of using that contraceptive method outweigh the risks for that patient. "You might talk with the woman about using a different method and if she decides to use the method with a 3 score, you would counsel her about the risks but would still prescribe it," she said. A score of 4 (red) means there’s an absolute contraindication to the method for that condition.
The MEC is available in an app for iPhone or iPad. The app lists choices by condition or by method; the online tables can be printed out and the options viewed all at once, she said.
"We have these hanging around at our clinic, and all of the residents carry them on their iPhones as well," she said. "It’s a really useful document."
The companion Selected Practice Recommendations for Contraception provide advice on how to use contraceptive methods in clinical practice. For example, there are recommendations on how long to use a backup contraceptive method once it has been started, special considerations for each method, and what to do when there are missed or late doses.
Dr. Steinauer reported having no financial disclosures,
On Twitter @sherryboschert
SAN FRANCISCO – Pop quiz: Do you know which contraceptive methods are safe for women who are postpartum or who smoke? How about for women who have diabetic nephropathy or migraine with aura?
Handy online tools and smart-phone apps from the Centers for Disease Control and Prevention now offer recommendations for contraceptive choices. The U.S. Medical Eligibility Criteria for Contraception, or MEC (MMWR 2010;59:1-6), now has a newer companion document, the U.S. Selected Practice Recommendations for Contraception, released by the U.S. Centers for Disease Control and Prevention (MMWR 2013;62:1-46), Dr. Jody Steinauer said at a conference on women’s health sponsored by the University of California, San Francisco.
The U.S. version of the MEC features four color-coded tables that compare contraceptive methods and more than 60 medical conditions or characteristics. Each pair of method and condition is scored for safety on a scale of 1-4.
A score of 1 (color-coded dark green) means there are no concerns about the method to be used in that scenario. A score of 2 (light green) means that generally the advantages outweigh the risks of the method, said Dr. Steinauer, of the university.
A score of 3 (pink) suggests there is a "relative contraindication" in which there are some concerns, but the advantages of using that contraceptive method outweigh the risks for that patient. "You might talk with the woman about using a different method and if she decides to use the method with a 3 score, you would counsel her about the risks but would still prescribe it," she said. A score of 4 (red) means there’s an absolute contraindication to the method for that condition.
The MEC is available in an app for iPhone or iPad. The app lists choices by condition or by method; the online tables can be printed out and the options viewed all at once, she said.
"We have these hanging around at our clinic, and all of the residents carry them on their iPhones as well," she said. "It’s a really useful document."
The companion Selected Practice Recommendations for Contraception provide advice on how to use contraceptive methods in clinical practice. For example, there are recommendations on how long to use a backup contraceptive method once it has been started, special considerations for each method, and what to do when there are missed or late doses.
Dr. Steinauer reported having no financial disclosures,
On Twitter @sherryboschert
EXPERT ANALYSIS AT A CONFERENCE ON WOMEN’S HEALTH
How can pregnant women safely relieve low-back pain?
Evidence Summary
Even though clinical research is lacking, acetaminophen is widely used to relieve low-back pain with no documented teratogenic effect (US Food and Drug Administration [FDA] category B). Nonsteroidal anti-inflammatory drugs are classified as FDA category D in the third trimester because of their documented association with oligohydramnios, premature closure of the ductus arteriosus, nephrotoxicity, and periventricular hemorrhage in the fetus.1 Opioids are category C and a poor choice to treat low-back pain in pregnancy.2
Physical therapy and water aerobics relieve pain, reduce sick days
A 2007 Cochrane review of interventions for treating back pain in pregnancy analyzed eight studies with a total of 1305 patients that examined the effects of adding physical therapy and acupuncture to usual care.3 In one RCT, 407 patients with and without pain received five 30-minute individualized physical therapy exercise sessions, two 45-minute group physical therapy classes, or standard care.3,4 Low-back pain decreased with group physical therapy (P <.05; number needed to treat [NNT] = 3.2) and individual therapy (NNT = 2.1). Patients who received individual therapy had a 12% decrease in sick days.
A prospective trial of 258 patients, half of whom did water aerobics and half physical therapy, showed comparable results for the two interventions (NNT = 11.4 for decreased sick days; odds ratio = 0.38, 95% confidence interval [CI], 0.16–0.88).3
Acupuncture reduces pain and analgesic use
A prospective, randomized open study cited in the 2007 Cochrane review divided 72 patients at 24 to 37 weeks’ gestational age into a group that received acupuncture plus standard care and a standard-care–only control group.3,5 Treatment sessions occurred one or two times per week until delivery or recovery. The acupuncture group reported decreased pain (60% vs 14% for controls; P <.01; NNT = 2.2) and improved function (43% vs 9% for controls; P <.001; NNT = 2.9). There was also a difference in analgesic use: 0% for the acupuncture group vs 14% for controls; P <.05; NNT = 7.1.
A 2009 RCT divided 159 patients at 25 to 38 weeks’ gestational age into three groups: auricular acupuncture at specific points for one week, sham auricular acupuncture at nonspecific points for one week, and controls. At the end of Week 1, 80% of the acupuncture group had a clinically significant reduction in pain compared with 56% in the sham acupuncture group and 36% in the control group (P = .001 acupuncture vs sham, NNT = 4.2; P <.0001 acupuncture vs controls, NNT = 2.3).6
Osteopathic manipulative therapy (OMT) decreases disability, but not pain
A 2010 RCT divided 144 third trimester patients into three groups that received usual obstetric care, sham ultrasound therapy plus usual obstetric care, or OMT.7 Pain remained similar among the three groups throughout the study. Using the 24-point Roland-Morris Disability Questionnaire, OMT decreased disability by 0.72 points (95% CI, 0.31–1.14; P <.001) compared with 0.35 points in the usual obstetric-care–only group (95% CI, −0.06 to 0.76; P = .09). Ultrasound had no effect.
Corticosteroid injection reduces pain in a small trial
A small RCT of injection with the corticosteroid triamcinolone at the sacrospinous ligament insertion in 36 women with low-back pain showed significant reduction in pain in 17 of 18 women in the triamcinolone group compared with 9 of 18 women in the control group (P <.01; NNT = 2).8
Evidence lacking on maternity support garments
A poor-quality systematic review of 10 studies (N = 1909) of maternity support garments found insufficient evidence because of the heterogeneity of the trials.9
RECOMMENDATIONS
The American College of Obstetricians and Gynecologists suggests the following measures to prevent and treat low-back pain in pregnancy:10
• wear low-heeled (not flat) shoes with good arch support
• get help when lifting heavy objects
• place one foot on a stool or box when standing for long periods
• place a board between the mattress and box spring if the bed is too soft
• squat down, bend knees, and keep back straight when lifting
• sit in chairs with good back support or use a small pillow to provide support
• sleep on side with pillows between knees for support
• apply heat, cold, or massage to the painful area.
WE WANT TO HEAR FROM YOU!
Drop us a line and let us know what you think about current articles, which topics you'd like to see covered in future issues, and what challenges you face in daily practice. Tell us what you think by emailing us at: [email protected]This article was originally published in The Journal of Family Practice (J Fam Pract. 2013;62(5):260, 268).
Black RA, Hill DA. Over-the-counter medications in pregnancy. Am Fam Physician. 2003;67(12):2517–2524.
Vermani E, Mittal R, Weeks A. Pelvic girdle pain and low back pain in pregnancy: a review. Pain Pract. 2010;10(1):60–71.
Pennick VE, Young G. Interventions for preventing and treating pelvic and back pain in pregnancy. Cochrane Database Syst Rev. 2007;(2):CD001139.
Ostgaard HC, Zetherstrom G, Roos-Hansson E, et al. Reduction of back and posterior pelvic pain in pregnancy. Spine. 1994;19(8):894–900.
Kvorning N, Holmberg C, Grennert L, et al. Acupuncture relieves pelvic and low back pain in late pregnancy. Acta Obstet Gynecol Scand. 2004;83(3):246–250.
Wang SM, Dezinno P, Lin EC, et al. Auricular acupuncture as a treatment for pregnant women who have low back and posterior pelvic pain: A pilot study. Am J Obstet Gynecol. 2009;201(3):271.e1–e9.
Licciardone J, Buchanan S, Hensel K, King H, Fulda K, Stoll S. Osteopathic manipulative treatment of back pain and related symptoms during pregnancy: A randomized controlled trial. Am J Obstet Gynecol. 2010;202(1):43.e1–e8.
Torstensson T, Lindgren A, Kristiansson P. Corticosteroid injection treatment to the ischiadic spine reduced pain in women with long-lasting sacral low back pain with onset during pregnancy: A randomized, double blind, controlled trial. Spine. 2009;34(21):2254–2258.
Ho SS, Yu WW, Lao TT, et al. Effectiveness of maternity support belts in reducing low back pain during pregnancy: A review. J Clin Nurs. 2009;18(11):1523–1532.
American College of Obstetricians and Gynecologists. Patient education guidelines for easing back pain during pregnancy. http://www.acog.org/~/media/For%20Patients/faq115.pdf?dmc=1ts=20130118T1434071958. Published August 2011. Accessed December 10, 2012.
Evidence Summary
Even though clinical research is lacking, acetaminophen is widely used to relieve low-back pain with no documented teratogenic effect (US Food and Drug Administration [FDA] category B). Nonsteroidal anti-inflammatory drugs are classified as FDA category D in the third trimester because of their documented association with oligohydramnios, premature closure of the ductus arteriosus, nephrotoxicity, and periventricular hemorrhage in the fetus.1 Opioids are category C and a poor choice to treat low-back pain in pregnancy.2
Physical therapy and water aerobics relieve pain, reduce sick days
A 2007 Cochrane review of interventions for treating back pain in pregnancy analyzed eight studies with a total of 1305 patients that examined the effects of adding physical therapy and acupuncture to usual care.3 In one RCT, 407 patients with and without pain received five 30-minute individualized physical therapy exercise sessions, two 45-minute group physical therapy classes, or standard care.3,4 Low-back pain decreased with group physical therapy (P <.05; number needed to treat [NNT] = 3.2) and individual therapy (NNT = 2.1). Patients who received individual therapy had a 12% decrease in sick days.
A prospective trial of 258 patients, half of whom did water aerobics and half physical therapy, showed comparable results for the two interventions (NNT = 11.4 for decreased sick days; odds ratio = 0.38, 95% confidence interval [CI], 0.16–0.88).3
Acupuncture reduces pain and analgesic use
A prospective, randomized open study cited in the 2007 Cochrane review divided 72 patients at 24 to 37 weeks’ gestational age into a group that received acupuncture plus standard care and a standard-care–only control group.3,5 Treatment sessions occurred one or two times per week until delivery or recovery. The acupuncture group reported decreased pain (60% vs 14% for controls; P <.01; NNT = 2.2) and improved function (43% vs 9% for controls; P <.001; NNT = 2.9). There was also a difference in analgesic use: 0% for the acupuncture group vs 14% for controls; P <.05; NNT = 7.1.
A 2009 RCT divided 159 patients at 25 to 38 weeks’ gestational age into three groups: auricular acupuncture at specific points for one week, sham auricular acupuncture at nonspecific points for one week, and controls. At the end of Week 1, 80% of the acupuncture group had a clinically significant reduction in pain compared with 56% in the sham acupuncture group and 36% in the control group (P = .001 acupuncture vs sham, NNT = 4.2; P <.0001 acupuncture vs controls, NNT = 2.3).6
Osteopathic manipulative therapy (OMT) decreases disability, but not pain
A 2010 RCT divided 144 third trimester patients into three groups that received usual obstetric care, sham ultrasound therapy plus usual obstetric care, or OMT.7 Pain remained similar among the three groups throughout the study. Using the 24-point Roland-Morris Disability Questionnaire, OMT decreased disability by 0.72 points (95% CI, 0.31–1.14; P <.001) compared with 0.35 points in the usual obstetric-care–only group (95% CI, −0.06 to 0.76; P = .09). Ultrasound had no effect.
Corticosteroid injection reduces pain in a small trial
A small RCT of injection with the corticosteroid triamcinolone at the sacrospinous ligament insertion in 36 women with low-back pain showed significant reduction in pain in 17 of 18 women in the triamcinolone group compared with 9 of 18 women in the control group (P <.01; NNT = 2).8
Evidence lacking on maternity support garments
A poor-quality systematic review of 10 studies (N = 1909) of maternity support garments found insufficient evidence because of the heterogeneity of the trials.9
RECOMMENDATIONS
The American College of Obstetricians and Gynecologists suggests the following measures to prevent and treat low-back pain in pregnancy:10
• wear low-heeled (not flat) shoes with good arch support
• get help when lifting heavy objects
• place one foot on a stool or box when standing for long periods
• place a board between the mattress and box spring if the bed is too soft
• squat down, bend knees, and keep back straight when lifting
• sit in chairs with good back support or use a small pillow to provide support
• sleep on side with pillows between knees for support
• apply heat, cold, or massage to the painful area.
WE WANT TO HEAR FROM YOU!
Drop us a line and let us know what you think about current articles, which topics you'd like to see covered in future issues, and what challenges you face in daily practice. Tell us what you think by emailing us at: [email protected]This article was originally published in The Journal of Family Practice (J Fam Pract. 2013;62(5):260, 268).
Evidence Summary
Even though clinical research is lacking, acetaminophen is widely used to relieve low-back pain with no documented teratogenic effect (US Food and Drug Administration [FDA] category B). Nonsteroidal anti-inflammatory drugs are classified as FDA category D in the third trimester because of their documented association with oligohydramnios, premature closure of the ductus arteriosus, nephrotoxicity, and periventricular hemorrhage in the fetus.1 Opioids are category C and a poor choice to treat low-back pain in pregnancy.2
Physical therapy and water aerobics relieve pain, reduce sick days
A 2007 Cochrane review of interventions for treating back pain in pregnancy analyzed eight studies with a total of 1305 patients that examined the effects of adding physical therapy and acupuncture to usual care.3 In one RCT, 407 patients with and without pain received five 30-minute individualized physical therapy exercise sessions, two 45-minute group physical therapy classes, or standard care.3,4 Low-back pain decreased with group physical therapy (P <.05; number needed to treat [NNT] = 3.2) and individual therapy (NNT = 2.1). Patients who received individual therapy had a 12% decrease in sick days.
A prospective trial of 258 patients, half of whom did water aerobics and half physical therapy, showed comparable results for the two interventions (NNT = 11.4 for decreased sick days; odds ratio = 0.38, 95% confidence interval [CI], 0.16–0.88).3
Acupuncture reduces pain and analgesic use
A prospective, randomized open study cited in the 2007 Cochrane review divided 72 patients at 24 to 37 weeks’ gestational age into a group that received acupuncture plus standard care and a standard-care–only control group.3,5 Treatment sessions occurred one or two times per week until delivery or recovery. The acupuncture group reported decreased pain (60% vs 14% for controls; P <.01; NNT = 2.2) and improved function (43% vs 9% for controls; P <.001; NNT = 2.9). There was also a difference in analgesic use: 0% for the acupuncture group vs 14% for controls; P <.05; NNT = 7.1.
A 2009 RCT divided 159 patients at 25 to 38 weeks’ gestational age into three groups: auricular acupuncture at specific points for one week, sham auricular acupuncture at nonspecific points for one week, and controls. At the end of Week 1, 80% of the acupuncture group had a clinically significant reduction in pain compared with 56% in the sham acupuncture group and 36% in the control group (P = .001 acupuncture vs sham, NNT = 4.2; P <.0001 acupuncture vs controls, NNT = 2.3).6
Osteopathic manipulative therapy (OMT) decreases disability, but not pain
A 2010 RCT divided 144 third trimester patients into three groups that received usual obstetric care, sham ultrasound therapy plus usual obstetric care, or OMT.7 Pain remained similar among the three groups throughout the study. Using the 24-point Roland-Morris Disability Questionnaire, OMT decreased disability by 0.72 points (95% CI, 0.31–1.14; P <.001) compared with 0.35 points in the usual obstetric-care–only group (95% CI, −0.06 to 0.76; P = .09). Ultrasound had no effect.
Corticosteroid injection reduces pain in a small trial
A small RCT of injection with the corticosteroid triamcinolone at the sacrospinous ligament insertion in 36 women with low-back pain showed significant reduction in pain in 17 of 18 women in the triamcinolone group compared with 9 of 18 women in the control group (P <.01; NNT = 2).8
Evidence lacking on maternity support garments
A poor-quality systematic review of 10 studies (N = 1909) of maternity support garments found insufficient evidence because of the heterogeneity of the trials.9
RECOMMENDATIONS
The American College of Obstetricians and Gynecologists suggests the following measures to prevent and treat low-back pain in pregnancy:10
• wear low-heeled (not flat) shoes with good arch support
• get help when lifting heavy objects
• place one foot on a stool or box when standing for long periods
• place a board between the mattress and box spring if the bed is too soft
• squat down, bend knees, and keep back straight when lifting
• sit in chairs with good back support or use a small pillow to provide support
• sleep on side with pillows between knees for support
• apply heat, cold, or massage to the painful area.
WE WANT TO HEAR FROM YOU!
Drop us a line and let us know what you think about current articles, which topics you'd like to see covered in future issues, and what challenges you face in daily practice. Tell us what you think by emailing us at: [email protected]This article was originally published in The Journal of Family Practice (J Fam Pract. 2013;62(5):260, 268).
Black RA, Hill DA. Over-the-counter medications in pregnancy. Am Fam Physician. 2003;67(12):2517–2524.
Vermani E, Mittal R, Weeks A. Pelvic girdle pain and low back pain in pregnancy: a review. Pain Pract. 2010;10(1):60–71.
Pennick VE, Young G. Interventions for preventing and treating pelvic and back pain in pregnancy. Cochrane Database Syst Rev. 2007;(2):CD001139.
Ostgaard HC, Zetherstrom G, Roos-Hansson E, et al. Reduction of back and posterior pelvic pain in pregnancy. Spine. 1994;19(8):894–900.
Kvorning N, Holmberg C, Grennert L, et al. Acupuncture relieves pelvic and low back pain in late pregnancy. Acta Obstet Gynecol Scand. 2004;83(3):246–250.
Wang SM, Dezinno P, Lin EC, et al. Auricular acupuncture as a treatment for pregnant women who have low back and posterior pelvic pain: A pilot study. Am J Obstet Gynecol. 2009;201(3):271.e1–e9.
Licciardone J, Buchanan S, Hensel K, King H, Fulda K, Stoll S. Osteopathic manipulative treatment of back pain and related symptoms during pregnancy: A randomized controlled trial. Am J Obstet Gynecol. 2010;202(1):43.e1–e8.
Torstensson T, Lindgren A, Kristiansson P. Corticosteroid injection treatment to the ischiadic spine reduced pain in women with long-lasting sacral low back pain with onset during pregnancy: A randomized, double blind, controlled trial. Spine. 2009;34(21):2254–2258.
Ho SS, Yu WW, Lao TT, et al. Effectiveness of maternity support belts in reducing low back pain during pregnancy: A review. J Clin Nurs. 2009;18(11):1523–1532.
American College of Obstetricians and Gynecologists. Patient education guidelines for easing back pain during pregnancy. http://www.acog.org/~/media/For%20Patients/faq115.pdf?dmc=1ts=20130118T1434071958. Published August 2011. Accessed December 10, 2012.
Black RA, Hill DA. Over-the-counter medications in pregnancy. Am Fam Physician. 2003;67(12):2517–2524.
Vermani E, Mittal R, Weeks A. Pelvic girdle pain and low back pain in pregnancy: a review. Pain Pract. 2010;10(1):60–71.
Pennick VE, Young G. Interventions for preventing and treating pelvic and back pain in pregnancy. Cochrane Database Syst Rev. 2007;(2):CD001139.
Ostgaard HC, Zetherstrom G, Roos-Hansson E, et al. Reduction of back and posterior pelvic pain in pregnancy. Spine. 1994;19(8):894–900.
Kvorning N, Holmberg C, Grennert L, et al. Acupuncture relieves pelvic and low back pain in late pregnancy. Acta Obstet Gynecol Scand. 2004;83(3):246–250.
Wang SM, Dezinno P, Lin EC, et al. Auricular acupuncture as a treatment for pregnant women who have low back and posterior pelvic pain: A pilot study. Am J Obstet Gynecol. 2009;201(3):271.e1–e9.
Licciardone J, Buchanan S, Hensel K, King H, Fulda K, Stoll S. Osteopathic manipulative treatment of back pain and related symptoms during pregnancy: A randomized controlled trial. Am J Obstet Gynecol. 2010;202(1):43.e1–e8.
Torstensson T, Lindgren A, Kristiansson P. Corticosteroid injection treatment to the ischiadic spine reduced pain in women with long-lasting sacral low back pain with onset during pregnancy: A randomized, double blind, controlled trial. Spine. 2009;34(21):2254–2258.
Ho SS, Yu WW, Lao TT, et al. Effectiveness of maternity support belts in reducing low back pain during pregnancy: A review. J Clin Nurs. 2009;18(11):1523–1532.
American College of Obstetricians and Gynecologists. Patient education guidelines for easing back pain during pregnancy. http://www.acog.org/~/media/For%20Patients/faq115.pdf?dmc=1ts=20130118T1434071958. Published August 2011. Accessed December 10, 2012.
Update on Obstetrics
Over the past 20 years, the incidence of preeclampsia in the United States has increased 25%,1 and the disorder is a leading cause of morbidity and death among both mothers and infants. Although considerable progress has been achieved in elucidating the pathophysiology of preeclampsia, greater understanding has not yet carried over into improved clinical practice.
To address this disconnect between data and practice, the American College of Obstetricians and Gynecologists (ACOG) issued a 99-page document in November 2013 to help establish best practices in the diagnosis and management of hypertensive disorders in pregnancy. We begin this article with a look at its major recommendations.
Other notable developments in obstetrics over the past year have been the rapid evolution of noninvasive prenatal testing and the publication of new guidance on screening, diagnosis, and management of gestational diabetes, all of which are addressed in this article.
ACOG AIMS TO CLARIFY BEST PRACTICES IN THE MANAGEMENT OF HYPERTENSION IN PREGNANCY
American College of Obstetricians and Gynecologists, Task Force on Hypertension in Pregnancy. Hypertension in Pregnancy. Washington, DC: ACOG; November 2013.
The biggest news of the past year is probably the November 2013 report on hypertension in pregnancy from ACOG, which was developed with three goals in mind:
to summarize current knowledge
to provide best-practice guidelines
to identify areas in which further research is needed.
The classification, diagnosis, prediction, prevention, and management of gestational hypertension, preeclampsia, and chronic hypertension are addressed in the report. Although space constraints prevent us from summarizing the entire document, we would like to highlight the biggest changes and most relevant additions to clinical practice.
Notable recommendations
Classification. Preeclampsia is no longer characterized as “mild” or “severe” but as “preeclampsia without severe features” and “preeclampsia with severe features.” As justification for these changes, the ACOG Task Force on Hypertension in Pregnancy noted that preeclampsia is progressive by nature, so a characterization of “mild” disease is appropriate only at the time of diagnosis. Therefore, “appropriate management mandates frequent reevaluation for severe features.”
Diagnosis of proteinuria. The options are a 24-hour urine collection demonstrating more than 300 mg of protein or a single-specimen urine protein:creatinine ratio of 0.3 mg/dL or higher. Dipstick values should only be used if these quantitative measures are unavailable.
Signs of severe disease. Fetal growth restriction and proteinuria of more than 5 g/24 hr are no longer considered defining features of severe disease.
Severe features now include any of these:
systolic blood pressure (BP) of 160 mm Hg or higher, or diastolic BP of 110 mm Hg or higher on two occasions at least 4 hours apart while the patient is on bed rest (unless antihypertensive therapy is initiated before this time)
thrombocytopenia (platelets <100 x 109/L)
impaired liver function, as indicated by abnormally elevated blood concentrations of liver enzymes (to twice normal concentration) and/or severe, persistent right upper quadrant or epigastric pain unresponsive to medication and not accounted for by alternative diagnoses
progressive renal insufficiency (serum creatinine concentration >1.1 mg/dL or a doubling of the serum creatinine concentration in the absence of other renal disease)
pulmonary edema
new-onset visual or central nervous system disturbances.
Related Article: Does an unfavorable cervix preclude induction of labor at term in women who have gestational hypertension or mild preeclampsia? George Macones, MD (Examining the Evidence, December 2012)
Screening for preeclampsia. The use of Doppler studies and serum biomarkers is not recommended, as there is no evidence that early identification translates to improved outcomes.
Prevention of preeclampsia. Low-dose aspirin (60–80 mg/d, starting in the late first trimester) should be offered as primary prevention to:
women with a history of early-onset preeclampsia and delivery before 34 weeks’ gestation
women with a history of preeclampsia in multiple pregnancies
other high-risk patients (chronic hypertension, diabetes).
No other treatments (vitamin C or E, salt restriction, or bed rest) are recommended for the prevention of preeclampsia, although calcium supplementation may be recommended for women with a low baseline dietary intake of calcium.
Use of magnesium sulfate. Universal prophylaxis with magnesium sulfate is not recommended for preeclampsia unless severe features are present or the patient’s clinical condition changes to severe during labor.
Timing of delivery. Recommendations for delivery for patients with hypertensive disorders are:
gestational hypertension or preeclampsia without severe features: 37 weeks’ gestation
preeclampsia with severe features: by 34 weeks
chronic hypertension: not before 38 weeks
chronic hypertension with superimposed preeclampsia: 34 or 37 weeks, depending on the presence of severe features.
All of these recommendations are contingent upon the clinical status of the patient and her fetus. For example, if the fetus develops severe growth restriction (<5%) or oligohydramnios, delivery may be recommended regardless of gestational age, based on fetal testing and maternal stability.
Related Article: A stepwise approach to managing eclampsia and other hypertensive emergencies Baha M. Sibai (October 2013)
Postpartum hypertension. The need for recognition of hypertension in the postpartum period is emphasized, as well as appropriate management, using the following guidelines:
Be aware that BP decreases initially after delivery and then increases 3 to 6 days postpartum, requiring vigilance on the part of the clinician. For this reason, BP monitoring is recommended 72 hours postpartum (inpatient or outpatient) and again in 7 to 10 days in women diagnosed with a hypertensive disorder of pregnancy.
Counsel patients who experience a hypertensive disorder and/or preeclampsia during pregnancy about postpartum preeclampsia, providing strict precautions and explicit instructions regarding its signs and symptoms
If BP remains elevated after the first postpartum day, consider discontinuing nonsteroidal anti-inflammatory drugs (NSAIDs), as they may be related to hypertension
Treat BP that remains above 150/100 mm Hg with antihypertensive therapy
If postpartum preeclampsia is suspected, administer magnesium sulfate for 24 hours.
A culture shift is needed
At our institution, the most significant potential changes to clinical practice may be the elimination of universal magnesium sulfate prophylaxis and the removal of severe fetal growth restriction from the definition of “severe” preeclampsia.
Also, because the use of NSAIDs is widespread for postpartum pain control, a culture change is needed if we are to follow the postpartum recommendations.
WHAT THIS EVIDENCE MEANS FOR PRACTICE
Although we suggest that you read the ACOG report thoroughly, remember that its recommendations are only that—recommendations. Also keep in mind that only six of the approximately 60 “recommendations” provided in this report were accompanied by both high-quality evidence and a strong recommendation. There still is room for clinical judgment and individualization of management to specific patient populations.
NONINVASIVE PRENATAL SCREENING IS EXPANDING RAPIDLY—BUT DON’T THROW OUT THAT CVS KIT JUST YET!
ACOG Committee on Genetics.Committee Opinion #545: Noninvasive prenatal testing for fetal aneuploidy. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2012;120(6):1532–1534.
Mennuti MT, Cherry AM, Morrissette JJD, Dugoff L. Is it time to sound an alarm about false-positive cell-free DNA testing for fetal aneuploidy? Am J Obstet Gynecol. 2013;209(5):415–419.
In last year’s Update in Obstetrics, we discussed noninvasive prenatal genetic screening via cell-free fetal DNA, noting that it is a safer (no risk of miscarriage) and faster (starting at 10 weeks’ gestation) way to screen for aneuploidy. The sensitivity and specificity of this test for Trisomy 21 and 18 are over 99%, with slightly lower sensitivity for Trisomy 13 and sex chromosome abnormalities and a false-positive rate of 0.5%.
In a committee opinion published in December 2012, ACOG concluded that
cell-free fetal DNA is an appropriate screening option only for specific groups of patients at risk for aneuploidy:
women older than age 35
women with fetal ultrasonography findings that are concerning for aneuploidy
women with aneuploidy in a prior pregnancy
women with abnormal first- or second-trimester genetic screening tests
parents with a balanced translocation and an increased risk of Trisomy 21 or 13.
Noninvasive aneuploidy testing is for screening only
Negative results are not diagnostic, and all positive results should be confirmed with invasive testing (chorionic villus sampling [CVS] or amniocentesis).
ACOG does not recommend routine use of cell-free fetal DNA without a comprehensive history and adequate patient counseling, as well as a designation of “high risk.”
Over the past year, more options have become available for aneuploidy screening via cell-free fetal DNA, including screening in twin gestations (for Trisomy 21, 18, 13, and the presence of a Y chromosome only) and screening for:
22q deletion (DiGeorge syndrome)
5p– (Cri-du-chat syndrome)
15q (Prader-Willi and Angelman syndromes)
1p (1p36 deletion syndrome)
Trisomy 16
Trisomy 22.
At this rate, the genetic information potentially available via noninvasive testing seems unlimited. It is easy to see how the fact that this is a screening test—not a diagnostic test—could get lost in the excitement.
Other limitations: Noninvasive testing is still not validated in low-risk patients, and the false-positive risk may be higher than original estimates.
Related Article: Noninvasive prenatal DNA testing: A survey of who is using it, and how (Audiocast, June 2013)
The problem of false positives
This issue was addressed by Mennuti and colleagues, who presented eight cases of abnormal cell-free fetal DNA results that were not confirmed by invasive testing.
There are few prospective data about the source of false-positive results; potential mechanisms include an inadequate fetal fraction of cell-free DNA, maternal or placental mosaicism, and a vanishing twin.
Mennuti and colleagues propose that a registry of false-positive and false-negative results be established to gather further data. They also note that as low-risk patients and aneuploidies of lower and lower prevalence are incorporated into noninvasive testing, the false-positive rate will rise. Their findings have implications for patient counseling, patient distress, invasive testing, and reimbursement.
WHAT THIS EVIDENCE MEANS FOR PRACTICE
Cell-free fetal DNA is a rapidly expanding screening technology, but it is not ready to replace diagnostic testing. Don’t throw away those CVS and amniocentesis kits just yet—we are a long way from a completely noninvasive world.
WHEN IT COMES TO GESTATIONAL DIABETES, LESS MAY BE MORE
ACOG Committee on Practice Bulletins–Obstetrics. Practice Bulletin #137: Gestational diabetes mellitus. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2013;122(2 Pt 1):406–416.
Gestational diabetes accounts for 90% of diabetic pregnancies, and its incidence has been increasing in the United States along with the obesity epidemic. In recent years, there has been some debate about the best way to screen for, diagnose, and treat gestational diabetes.
Multiple criteria exist for a positive 1-hour (130–140 mg/dL) or 3-hour glucose tolerance test (Carpenter and Coustan vs National Diabetes Data Group), without comparative trials or consensus as to which version is best. Lower cutoffs increase the rate of gestational diabetes by as much as 50%, whereas higher cutoffs lower the false-positive rate and reduce the need for additional tests.
Some groups have recommended moving away from the traditional two-step process to a one-step approach that utilizes the 2-hour, 75-g glucose tolerance test commonly used outside of pregnancy. They argue that this approach would simplify and standardize the process and could improve outcomes in “borderline” pregnancies that would have been missed by less stringent guidelines.
However, the baseline rate of gestational diabetes using this one-step approach would likely increase from 7% to 18% or higher, depending on the patient population. Such an increase would trigger a huge rise in costs and resources needed to care for these patients, without data on outcomes or appropriate therapy for this expanded group of women with gestational diabetes.
Treatment isn't clear-cut, either
Treatment of gestational diabetes centers on labor-intensive glucose monitoring, nutritional interventions, and insulin therapy.
Until recently, the use of oral hypoglycemic agents was not recommended due to limited data. Multiple studies now have been performed to evaluate the safety and efficacy of glyburide and metformin in pregnancy, demonstrating glucose control similar to that achieved with insulin without short-term adverse effects in the mother or newborn. However, as many as 20% to 40% of women using glyburide and 50% of those using metformin require the addition of insulin for adequate glucose control. The long-term effects of these medications are unknown.
Related Article: Does myo-inositol supplementation reduce the rate of gestational diabetes in pregnant women with a family history of type 2 diabetes? E. Albert Reece, MD, PhD, MBA (Examining the Evidence, June 2013)
ACOG weighs in
In an attempt to clarify optimal screening, diagnosis, and treatment, ACOG updated its practice bulletin on gestational diabetes in August 2013. Among its recommendations:
Avoid the 2-hour glucose tolerance test because there is no demonstrated benefit for the increased number of mothers (and their fetuses) that would be identified by this approach. Rather, use the two-step approach of a 1-hour 50-g glucose tolerance test followed by a 3-hour 100-g glucose tolerance test.
In regard to the 1-hour test, ACOG finds either 135 or 140 mg/dL acceptable as a cutoff but recommends that each practice choose one value as a standard and use it consistently. For the 3-hour test, ACOG recommends that each practice choose the version that best fits its population and prevalence of diabetes. At our institution, for example, we have chosen a 1-hour cutoff of 140 mg/dL and the National Diabetes Data Group criteria for the 3-hour test (105, 190, 165, and 145 mg/dL).
Oral glyburide or metformin may be used to treat gestational diabetes, but glyburide (starting at 2.5 mg/d) may be the better choice for glucose control.
WHAT THIS EVIDENCE MEANS FOR PRACTICE
Use the two-step screening process to identify gestational diabetes, picking your cutoffs and sticking to them. Oral hypoglycemic agents now are acceptable for treatment, but be prepared to go back to insulin if necessary.
WE WANT TO HEAR FROM YOU. Tell us what you think.
Reference
Wallis AB, Saftlas AF, Hsia J, Atrash HK. Secular trends in the rates of preeclampsia, eclampsia, and gestational hypertension, United States, 1987–2004. Am J Hypertens. 2008;21(5):521–526.
Over the past 20 years, the incidence of preeclampsia in the United States has increased 25%,1 and the disorder is a leading cause of morbidity and death among both mothers and infants. Although considerable progress has been achieved in elucidating the pathophysiology of preeclampsia, greater understanding has not yet carried over into improved clinical practice.
To address this disconnect between data and practice, the American College of Obstetricians and Gynecologists (ACOG) issued a 99-page document in November 2013 to help establish best practices in the diagnosis and management of hypertensive disorders in pregnancy. We begin this article with a look at its major recommendations.
Other notable developments in obstetrics over the past year have been the rapid evolution of noninvasive prenatal testing and the publication of new guidance on screening, diagnosis, and management of gestational diabetes, all of which are addressed in this article.
ACOG AIMS TO CLARIFY BEST PRACTICES IN THE MANAGEMENT OF HYPERTENSION IN PREGNANCY
American College of Obstetricians and Gynecologists, Task Force on Hypertension in Pregnancy. Hypertension in Pregnancy. Washington, DC: ACOG; November 2013.
The biggest news of the past year is probably the November 2013 report on hypertension in pregnancy from ACOG, which was developed with three goals in mind:
to summarize current knowledge
to provide best-practice guidelines
to identify areas in which further research is needed.
The classification, diagnosis, prediction, prevention, and management of gestational hypertension, preeclampsia, and chronic hypertension are addressed in the report. Although space constraints prevent us from summarizing the entire document, we would like to highlight the biggest changes and most relevant additions to clinical practice.
Notable recommendations
Classification. Preeclampsia is no longer characterized as “mild” or “severe” but as “preeclampsia without severe features” and “preeclampsia with severe features.” As justification for these changes, the ACOG Task Force on Hypertension in Pregnancy noted that preeclampsia is progressive by nature, so a characterization of “mild” disease is appropriate only at the time of diagnosis. Therefore, “appropriate management mandates frequent reevaluation for severe features.”
Diagnosis of proteinuria. The options are a 24-hour urine collection demonstrating more than 300 mg of protein or a single-specimen urine protein:creatinine ratio of 0.3 mg/dL or higher. Dipstick values should only be used if these quantitative measures are unavailable.
Signs of severe disease. Fetal growth restriction and proteinuria of more than 5 g/24 hr are no longer considered defining features of severe disease.
Severe features now include any of these:
systolic blood pressure (BP) of 160 mm Hg or higher, or diastolic BP of 110 mm Hg or higher on two occasions at least 4 hours apart while the patient is on bed rest (unless antihypertensive therapy is initiated before this time)
thrombocytopenia (platelets <100 x 109/L)
impaired liver function, as indicated by abnormally elevated blood concentrations of liver enzymes (to twice normal concentration) and/or severe, persistent right upper quadrant or epigastric pain unresponsive to medication and not accounted for by alternative diagnoses
progressive renal insufficiency (serum creatinine concentration >1.1 mg/dL or a doubling of the serum creatinine concentration in the absence of other renal disease)
pulmonary edema
new-onset visual or central nervous system disturbances.
Related Article: Does an unfavorable cervix preclude induction of labor at term in women who have gestational hypertension or mild preeclampsia? George Macones, MD (Examining the Evidence, December 2012)
Screening for preeclampsia. The use of Doppler studies and serum biomarkers is not recommended, as there is no evidence that early identification translates to improved outcomes.
Prevention of preeclampsia. Low-dose aspirin (60–80 mg/d, starting in the late first trimester) should be offered as primary prevention to:
women with a history of early-onset preeclampsia and delivery before 34 weeks’ gestation
women with a history of preeclampsia in multiple pregnancies
other high-risk patients (chronic hypertension, diabetes).
No other treatments (vitamin C or E, salt restriction, or bed rest) are recommended for the prevention of preeclampsia, although calcium supplementation may be recommended for women with a low baseline dietary intake of calcium.
Use of magnesium sulfate. Universal prophylaxis with magnesium sulfate is not recommended for preeclampsia unless severe features are present or the patient’s clinical condition changes to severe during labor.
Timing of delivery. Recommendations for delivery for patients with hypertensive disorders are:
gestational hypertension or preeclampsia without severe features: 37 weeks’ gestation
preeclampsia with severe features: by 34 weeks
chronic hypertension: not before 38 weeks
chronic hypertension with superimposed preeclampsia: 34 or 37 weeks, depending on the presence of severe features.
All of these recommendations are contingent upon the clinical status of the patient and her fetus. For example, if the fetus develops severe growth restriction (<5%) or oligohydramnios, delivery may be recommended regardless of gestational age, based on fetal testing and maternal stability.
Related Article: A stepwise approach to managing eclampsia and other hypertensive emergencies Baha M. Sibai (October 2013)
Postpartum hypertension. The need for recognition of hypertension in the postpartum period is emphasized, as well as appropriate management, using the following guidelines:
Be aware that BP decreases initially after delivery and then increases 3 to 6 days postpartum, requiring vigilance on the part of the clinician. For this reason, BP monitoring is recommended 72 hours postpartum (inpatient or outpatient) and again in 7 to 10 days in women diagnosed with a hypertensive disorder of pregnancy.
Counsel patients who experience a hypertensive disorder and/or preeclampsia during pregnancy about postpartum preeclampsia, providing strict precautions and explicit instructions regarding its signs and symptoms
If BP remains elevated after the first postpartum day, consider discontinuing nonsteroidal anti-inflammatory drugs (NSAIDs), as they may be related to hypertension
Treat BP that remains above 150/100 mm Hg with antihypertensive therapy
If postpartum preeclampsia is suspected, administer magnesium sulfate for 24 hours.
A culture shift is needed
At our institution, the most significant potential changes to clinical practice may be the elimination of universal magnesium sulfate prophylaxis and the removal of severe fetal growth restriction from the definition of “severe” preeclampsia.
Also, because the use of NSAIDs is widespread for postpartum pain control, a culture change is needed if we are to follow the postpartum recommendations.
WHAT THIS EVIDENCE MEANS FOR PRACTICE
Although we suggest that you read the ACOG report thoroughly, remember that its recommendations are only that—recommendations. Also keep in mind that only six of the approximately 60 “recommendations” provided in this report were accompanied by both high-quality evidence and a strong recommendation. There still is room for clinical judgment and individualization of management to specific patient populations.
NONINVASIVE PRENATAL SCREENING IS EXPANDING RAPIDLY—BUT DON’T THROW OUT THAT CVS KIT JUST YET!
ACOG Committee on Genetics.Committee Opinion #545: Noninvasive prenatal testing for fetal aneuploidy. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2012;120(6):1532–1534.
Mennuti MT, Cherry AM, Morrissette JJD, Dugoff L. Is it time to sound an alarm about false-positive cell-free DNA testing for fetal aneuploidy? Am J Obstet Gynecol. 2013;209(5):415–419.
In last year’s Update in Obstetrics, we discussed noninvasive prenatal genetic screening via cell-free fetal DNA, noting that it is a safer (no risk of miscarriage) and faster (starting at 10 weeks’ gestation) way to screen for aneuploidy. The sensitivity and specificity of this test for Trisomy 21 and 18 are over 99%, with slightly lower sensitivity for Trisomy 13 and sex chromosome abnormalities and a false-positive rate of 0.5%.
In a committee opinion published in December 2012, ACOG concluded that
cell-free fetal DNA is an appropriate screening option only for specific groups of patients at risk for aneuploidy:
women older than age 35
women with fetal ultrasonography findings that are concerning for aneuploidy
women with aneuploidy in a prior pregnancy
women with abnormal first- or second-trimester genetic screening tests
parents with a balanced translocation and an increased risk of Trisomy 21 or 13.
Noninvasive aneuploidy testing is for screening only
Negative results are not diagnostic, and all positive results should be confirmed with invasive testing (chorionic villus sampling [CVS] or amniocentesis).
ACOG does not recommend routine use of cell-free fetal DNA without a comprehensive history and adequate patient counseling, as well as a designation of “high risk.”
Over the past year, more options have become available for aneuploidy screening via cell-free fetal DNA, including screening in twin gestations (for Trisomy 21, 18, 13, and the presence of a Y chromosome only) and screening for:
22q deletion (DiGeorge syndrome)
5p– (Cri-du-chat syndrome)
15q (Prader-Willi and Angelman syndromes)
1p (1p36 deletion syndrome)
Trisomy 16
Trisomy 22.
At this rate, the genetic information potentially available via noninvasive testing seems unlimited. It is easy to see how the fact that this is a screening test—not a diagnostic test—could get lost in the excitement.
Other limitations: Noninvasive testing is still not validated in low-risk patients, and the false-positive risk may be higher than original estimates.
Related Article: Noninvasive prenatal DNA testing: A survey of who is using it, and how (Audiocast, June 2013)
The problem of false positives
This issue was addressed by Mennuti and colleagues, who presented eight cases of abnormal cell-free fetal DNA results that were not confirmed by invasive testing.
There are few prospective data about the source of false-positive results; potential mechanisms include an inadequate fetal fraction of cell-free DNA, maternal or placental mosaicism, and a vanishing twin.
Mennuti and colleagues propose that a registry of false-positive and false-negative results be established to gather further data. They also note that as low-risk patients and aneuploidies of lower and lower prevalence are incorporated into noninvasive testing, the false-positive rate will rise. Their findings have implications for patient counseling, patient distress, invasive testing, and reimbursement.
WHAT THIS EVIDENCE MEANS FOR PRACTICE
Cell-free fetal DNA is a rapidly expanding screening technology, but it is not ready to replace diagnostic testing. Don’t throw away those CVS and amniocentesis kits just yet—we are a long way from a completely noninvasive world.
WHEN IT COMES TO GESTATIONAL DIABETES, LESS MAY BE MORE
ACOG Committee on Practice Bulletins–Obstetrics. Practice Bulletin #137: Gestational diabetes mellitus. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2013;122(2 Pt 1):406–416.
Gestational diabetes accounts for 90% of diabetic pregnancies, and its incidence has been increasing in the United States along with the obesity epidemic. In recent years, there has been some debate about the best way to screen for, diagnose, and treat gestational diabetes.
Multiple criteria exist for a positive 1-hour (130–140 mg/dL) or 3-hour glucose tolerance test (Carpenter and Coustan vs National Diabetes Data Group), without comparative trials or consensus as to which version is best. Lower cutoffs increase the rate of gestational diabetes by as much as 50%, whereas higher cutoffs lower the false-positive rate and reduce the need for additional tests.
Some groups have recommended moving away from the traditional two-step process to a one-step approach that utilizes the 2-hour, 75-g glucose tolerance test commonly used outside of pregnancy. They argue that this approach would simplify and standardize the process and could improve outcomes in “borderline” pregnancies that would have been missed by less stringent guidelines.
However, the baseline rate of gestational diabetes using this one-step approach would likely increase from 7% to 18% or higher, depending on the patient population. Such an increase would trigger a huge rise in costs and resources needed to care for these patients, without data on outcomes or appropriate therapy for this expanded group of women with gestational diabetes.
Treatment isn't clear-cut, either
Treatment of gestational diabetes centers on labor-intensive glucose monitoring, nutritional interventions, and insulin therapy.
Until recently, the use of oral hypoglycemic agents was not recommended due to limited data. Multiple studies now have been performed to evaluate the safety and efficacy of glyburide and metformin in pregnancy, demonstrating glucose control similar to that achieved with insulin without short-term adverse effects in the mother or newborn. However, as many as 20% to 40% of women using glyburide and 50% of those using metformin require the addition of insulin for adequate glucose control. The long-term effects of these medications are unknown.
Related Article: Does myo-inositol supplementation reduce the rate of gestational diabetes in pregnant women with a family history of type 2 diabetes? E. Albert Reece, MD, PhD, MBA (Examining the Evidence, June 2013)
ACOG weighs in
In an attempt to clarify optimal screening, diagnosis, and treatment, ACOG updated its practice bulletin on gestational diabetes in August 2013. Among its recommendations:
Avoid the 2-hour glucose tolerance test because there is no demonstrated benefit for the increased number of mothers (and their fetuses) that would be identified by this approach. Rather, use the two-step approach of a 1-hour 50-g glucose tolerance test followed by a 3-hour 100-g glucose tolerance test.
In regard to the 1-hour test, ACOG finds either 135 or 140 mg/dL acceptable as a cutoff but recommends that each practice choose one value as a standard and use it consistently. For the 3-hour test, ACOG recommends that each practice choose the version that best fits its population and prevalence of diabetes. At our institution, for example, we have chosen a 1-hour cutoff of 140 mg/dL and the National Diabetes Data Group criteria for the 3-hour test (105, 190, 165, and 145 mg/dL).
Oral glyburide or metformin may be used to treat gestational diabetes, but glyburide (starting at 2.5 mg/d) may be the better choice for glucose control.
WHAT THIS EVIDENCE MEANS FOR PRACTICE
Use the two-step screening process to identify gestational diabetes, picking your cutoffs and sticking to them. Oral hypoglycemic agents now are acceptable for treatment, but be prepared to go back to insulin if necessary.
WE WANT TO HEAR FROM YOU. Tell us what you think.
Over the past 20 years, the incidence of preeclampsia in the United States has increased 25%,1 and the disorder is a leading cause of morbidity and death among both mothers and infants. Although considerable progress has been achieved in elucidating the pathophysiology of preeclampsia, greater understanding has not yet carried over into improved clinical practice.
To address this disconnect between data and practice, the American College of Obstetricians and Gynecologists (ACOG) issued a 99-page document in November 2013 to help establish best practices in the diagnosis and management of hypertensive disorders in pregnancy. We begin this article with a look at its major recommendations.
Other notable developments in obstetrics over the past year have been the rapid evolution of noninvasive prenatal testing and the publication of new guidance on screening, diagnosis, and management of gestational diabetes, all of which are addressed in this article.
ACOG AIMS TO CLARIFY BEST PRACTICES IN THE MANAGEMENT OF HYPERTENSION IN PREGNANCY
American College of Obstetricians and Gynecologists, Task Force on Hypertension in Pregnancy. Hypertension in Pregnancy. Washington, DC: ACOG; November 2013.
The biggest news of the past year is probably the November 2013 report on hypertension in pregnancy from ACOG, which was developed with three goals in mind:
to summarize current knowledge
to provide best-practice guidelines
to identify areas in which further research is needed.
The classification, diagnosis, prediction, prevention, and management of gestational hypertension, preeclampsia, and chronic hypertension are addressed in the report. Although space constraints prevent us from summarizing the entire document, we would like to highlight the biggest changes and most relevant additions to clinical practice.
Notable recommendations
Classification. Preeclampsia is no longer characterized as “mild” or “severe” but as “preeclampsia without severe features” and “preeclampsia with severe features.” As justification for these changes, the ACOG Task Force on Hypertension in Pregnancy noted that preeclampsia is progressive by nature, so a characterization of “mild” disease is appropriate only at the time of diagnosis. Therefore, “appropriate management mandates frequent reevaluation for severe features.”
Diagnosis of proteinuria. The options are a 24-hour urine collection demonstrating more than 300 mg of protein or a single-specimen urine protein:creatinine ratio of 0.3 mg/dL or higher. Dipstick values should only be used if these quantitative measures are unavailable.
Signs of severe disease. Fetal growth restriction and proteinuria of more than 5 g/24 hr are no longer considered defining features of severe disease.
Severe features now include any of these:
systolic blood pressure (BP) of 160 mm Hg or higher, or diastolic BP of 110 mm Hg or higher on two occasions at least 4 hours apart while the patient is on bed rest (unless antihypertensive therapy is initiated before this time)
thrombocytopenia (platelets <100 x 109/L)
impaired liver function, as indicated by abnormally elevated blood concentrations of liver enzymes (to twice normal concentration) and/or severe, persistent right upper quadrant or epigastric pain unresponsive to medication and not accounted for by alternative diagnoses
progressive renal insufficiency (serum creatinine concentration >1.1 mg/dL or a doubling of the serum creatinine concentration in the absence of other renal disease)
pulmonary edema
new-onset visual or central nervous system disturbances.
Related Article: Does an unfavorable cervix preclude induction of labor at term in women who have gestational hypertension or mild preeclampsia? George Macones, MD (Examining the Evidence, December 2012)
Screening for preeclampsia. The use of Doppler studies and serum biomarkers is not recommended, as there is no evidence that early identification translates to improved outcomes.
Prevention of preeclampsia. Low-dose aspirin (60–80 mg/d, starting in the late first trimester) should be offered as primary prevention to:
women with a history of early-onset preeclampsia and delivery before 34 weeks’ gestation
women with a history of preeclampsia in multiple pregnancies
other high-risk patients (chronic hypertension, diabetes).
No other treatments (vitamin C or E, salt restriction, or bed rest) are recommended for the prevention of preeclampsia, although calcium supplementation may be recommended for women with a low baseline dietary intake of calcium.
Use of magnesium sulfate. Universal prophylaxis with magnesium sulfate is not recommended for preeclampsia unless severe features are present or the patient’s clinical condition changes to severe during labor.
Timing of delivery. Recommendations for delivery for patients with hypertensive disorders are:
gestational hypertension or preeclampsia without severe features: 37 weeks’ gestation
preeclampsia with severe features: by 34 weeks
chronic hypertension: not before 38 weeks
chronic hypertension with superimposed preeclampsia: 34 or 37 weeks, depending on the presence of severe features.
All of these recommendations are contingent upon the clinical status of the patient and her fetus. For example, if the fetus develops severe growth restriction (<5%) or oligohydramnios, delivery may be recommended regardless of gestational age, based on fetal testing and maternal stability.
Related Article: A stepwise approach to managing eclampsia and other hypertensive emergencies Baha M. Sibai (October 2013)
Postpartum hypertension. The need for recognition of hypertension in the postpartum period is emphasized, as well as appropriate management, using the following guidelines:
Be aware that BP decreases initially after delivery and then increases 3 to 6 days postpartum, requiring vigilance on the part of the clinician. For this reason, BP monitoring is recommended 72 hours postpartum (inpatient or outpatient) and again in 7 to 10 days in women diagnosed with a hypertensive disorder of pregnancy.
Counsel patients who experience a hypertensive disorder and/or preeclampsia during pregnancy about postpartum preeclampsia, providing strict precautions and explicit instructions regarding its signs and symptoms
If BP remains elevated after the first postpartum day, consider discontinuing nonsteroidal anti-inflammatory drugs (NSAIDs), as they may be related to hypertension
Treat BP that remains above 150/100 mm Hg with antihypertensive therapy
If postpartum preeclampsia is suspected, administer magnesium sulfate for 24 hours.
A culture shift is needed
At our institution, the most significant potential changes to clinical practice may be the elimination of universal magnesium sulfate prophylaxis and the removal of severe fetal growth restriction from the definition of “severe” preeclampsia.
Also, because the use of NSAIDs is widespread for postpartum pain control, a culture change is needed if we are to follow the postpartum recommendations.
WHAT THIS EVIDENCE MEANS FOR PRACTICE
Although we suggest that you read the ACOG report thoroughly, remember that its recommendations are only that—recommendations. Also keep in mind that only six of the approximately 60 “recommendations” provided in this report were accompanied by both high-quality evidence and a strong recommendation. There still is room for clinical judgment and individualization of management to specific patient populations.
NONINVASIVE PRENATAL SCREENING IS EXPANDING RAPIDLY—BUT DON’T THROW OUT THAT CVS KIT JUST YET!
ACOG Committee on Genetics.Committee Opinion #545: Noninvasive prenatal testing for fetal aneuploidy. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2012;120(6):1532–1534.
Mennuti MT, Cherry AM, Morrissette JJD, Dugoff L. Is it time to sound an alarm about false-positive cell-free DNA testing for fetal aneuploidy? Am J Obstet Gynecol. 2013;209(5):415–419.
In last year’s Update in Obstetrics, we discussed noninvasive prenatal genetic screening via cell-free fetal DNA, noting that it is a safer (no risk of miscarriage) and faster (starting at 10 weeks’ gestation) way to screen for aneuploidy. The sensitivity and specificity of this test for Trisomy 21 and 18 are over 99%, with slightly lower sensitivity for Trisomy 13 and sex chromosome abnormalities and a false-positive rate of 0.5%.
In a committee opinion published in December 2012, ACOG concluded that
cell-free fetal DNA is an appropriate screening option only for specific groups of patients at risk for aneuploidy:
women older than age 35
women with fetal ultrasonography findings that are concerning for aneuploidy
women with aneuploidy in a prior pregnancy
women with abnormal first- or second-trimester genetic screening tests
parents with a balanced translocation and an increased risk of Trisomy 21 or 13.
Noninvasive aneuploidy testing is for screening only
Negative results are not diagnostic, and all positive results should be confirmed with invasive testing (chorionic villus sampling [CVS] or amniocentesis).
ACOG does not recommend routine use of cell-free fetal DNA without a comprehensive history and adequate patient counseling, as well as a designation of “high risk.”
Over the past year, more options have become available for aneuploidy screening via cell-free fetal DNA, including screening in twin gestations (for Trisomy 21, 18, 13, and the presence of a Y chromosome only) and screening for:
22q deletion (DiGeorge syndrome)
5p– (Cri-du-chat syndrome)
15q (Prader-Willi and Angelman syndromes)
1p (1p36 deletion syndrome)
Trisomy 16
Trisomy 22.
At this rate, the genetic information potentially available via noninvasive testing seems unlimited. It is easy to see how the fact that this is a screening test—not a diagnostic test—could get lost in the excitement.
Other limitations: Noninvasive testing is still not validated in low-risk patients, and the false-positive risk may be higher than original estimates.
Related Article: Noninvasive prenatal DNA testing: A survey of who is using it, and how (Audiocast, June 2013)
The problem of false positives
This issue was addressed by Mennuti and colleagues, who presented eight cases of abnormal cell-free fetal DNA results that were not confirmed by invasive testing.
There are few prospective data about the source of false-positive results; potential mechanisms include an inadequate fetal fraction of cell-free DNA, maternal or placental mosaicism, and a vanishing twin.
Mennuti and colleagues propose that a registry of false-positive and false-negative results be established to gather further data. They also note that as low-risk patients and aneuploidies of lower and lower prevalence are incorporated into noninvasive testing, the false-positive rate will rise. Their findings have implications for patient counseling, patient distress, invasive testing, and reimbursement.
WHAT THIS EVIDENCE MEANS FOR PRACTICE
Cell-free fetal DNA is a rapidly expanding screening technology, but it is not ready to replace diagnostic testing. Don’t throw away those CVS and amniocentesis kits just yet—we are a long way from a completely noninvasive world.
WHEN IT COMES TO GESTATIONAL DIABETES, LESS MAY BE MORE
ACOG Committee on Practice Bulletins–Obstetrics. Practice Bulletin #137: Gestational diabetes mellitus. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2013;122(2 Pt 1):406–416.
Gestational diabetes accounts for 90% of diabetic pregnancies, and its incidence has been increasing in the United States along with the obesity epidemic. In recent years, there has been some debate about the best way to screen for, diagnose, and treat gestational diabetes.
Multiple criteria exist for a positive 1-hour (130–140 mg/dL) or 3-hour glucose tolerance test (Carpenter and Coustan vs National Diabetes Data Group), without comparative trials or consensus as to which version is best. Lower cutoffs increase the rate of gestational diabetes by as much as 50%, whereas higher cutoffs lower the false-positive rate and reduce the need for additional tests.
Some groups have recommended moving away from the traditional two-step process to a one-step approach that utilizes the 2-hour, 75-g glucose tolerance test commonly used outside of pregnancy. They argue that this approach would simplify and standardize the process and could improve outcomes in “borderline” pregnancies that would have been missed by less stringent guidelines.
However, the baseline rate of gestational diabetes using this one-step approach would likely increase from 7% to 18% or higher, depending on the patient population. Such an increase would trigger a huge rise in costs and resources needed to care for these patients, without data on outcomes or appropriate therapy for this expanded group of women with gestational diabetes.
Treatment isn't clear-cut, either
Treatment of gestational diabetes centers on labor-intensive glucose monitoring, nutritional interventions, and insulin therapy.
Until recently, the use of oral hypoglycemic agents was not recommended due to limited data. Multiple studies now have been performed to evaluate the safety and efficacy of glyburide and metformin in pregnancy, demonstrating glucose control similar to that achieved with insulin without short-term adverse effects in the mother or newborn. However, as many as 20% to 40% of women using glyburide and 50% of those using metformin require the addition of insulin for adequate glucose control. The long-term effects of these medications are unknown.
Related Article: Does myo-inositol supplementation reduce the rate of gestational diabetes in pregnant women with a family history of type 2 diabetes? E. Albert Reece, MD, PhD, MBA (Examining the Evidence, June 2013)
ACOG weighs in
In an attempt to clarify optimal screening, diagnosis, and treatment, ACOG updated its practice bulletin on gestational diabetes in August 2013. Among its recommendations:
Avoid the 2-hour glucose tolerance test because there is no demonstrated benefit for the increased number of mothers (and their fetuses) that would be identified by this approach. Rather, use the two-step approach of a 1-hour 50-g glucose tolerance test followed by a 3-hour 100-g glucose tolerance test.
In regard to the 1-hour test, ACOG finds either 135 or 140 mg/dL acceptable as a cutoff but recommends that each practice choose one value as a standard and use it consistently. For the 3-hour test, ACOG recommends that each practice choose the version that best fits its population and prevalence of diabetes. At our institution, for example, we have chosen a 1-hour cutoff of 140 mg/dL and the National Diabetes Data Group criteria for the 3-hour test (105, 190, 165, and 145 mg/dL).
Oral glyburide or metformin may be used to treat gestational diabetes, but glyburide (starting at 2.5 mg/d) may be the better choice for glucose control.
WHAT THIS EVIDENCE MEANS FOR PRACTICE
Use the two-step screening process to identify gestational diabetes, picking your cutoffs and sticking to them. Oral hypoglycemic agents now are acceptable for treatment, but be prepared to go back to insulin if necessary.
WE WANT TO HEAR FROM YOU. Tell us what you think.
Reference
Wallis AB, Saftlas AF, Hsia J, Atrash HK. Secular trends in the rates of preeclampsia, eclampsia, and gestational hypertension, United States, 1987–2004. Am J Hypertens. 2008;21(5):521–526.
Reference
Wallis AB, Saftlas AF, Hsia J, Atrash HK. Secular trends in the rates of preeclampsia, eclampsia, and gestational hypertension, United States, 1987–2004. Am J Hypertens. 2008;21(5):521–526.
ART overrepresented among low-birth weight infants
Infants born with the use of assisted reproductive technology make up a disproportionately large fraction of low-birth-weight infants, compared with the general birth population, the Centers for Disease Control and Prevention said.
In 2010, 5.6% of all low-birth-weight infants were conceived through ART. In comparison, the overall proportion of infants born who were ART infants was 1.5% that year. ART infants also "accounted for approximately 4.0% of all preterm births" in 2010, the CDC noted.
That same year, ART infants were overrepresented among multiple births, with 19.2% of twin and 32.5% of triplet and higher-order births involving ART. These numbers, however, are the latest in a downward trend, as "the percentage of ART-conceived multiple infants declined by 13.0%" from 2000 to 2010, the CDC reported (MMWR Surveill. Summ. 2013;62(9):1-24).
According to data from the CDC's National ART Surveillance System, there were 147,260 ART procedures performed in 2010 at 443 fertility clinics, resulting in 47,090 live-birth deliveries and 61,564 infants.
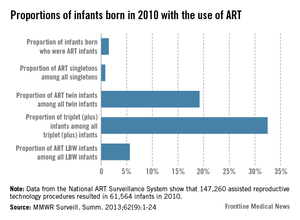
Infants born with the use of assisted reproductive technology make up a disproportionately large fraction of low-birth-weight infants, compared with the general birth population, the Centers for Disease Control and Prevention said.
In 2010, 5.6% of all low-birth-weight infants were conceived through ART. In comparison, the overall proportion of infants born who were ART infants was 1.5% that year. ART infants also "accounted for approximately 4.0% of all preterm births" in 2010, the CDC noted.
That same year, ART infants were overrepresented among multiple births, with 19.2% of twin and 32.5% of triplet and higher-order births involving ART. These numbers, however, are the latest in a downward trend, as "the percentage of ART-conceived multiple infants declined by 13.0%" from 2000 to 2010, the CDC reported (MMWR Surveill. Summ. 2013;62(9):1-24).
According to data from the CDC's National ART Surveillance System, there were 147,260 ART procedures performed in 2010 at 443 fertility clinics, resulting in 47,090 live-birth deliveries and 61,564 infants.

Infants born with the use of assisted reproductive technology make up a disproportionately large fraction of low-birth-weight infants, compared with the general birth population, the Centers for Disease Control and Prevention said.
In 2010, 5.6% of all low-birth-weight infants were conceived through ART. In comparison, the overall proportion of infants born who were ART infants was 1.5% that year. ART infants also "accounted for approximately 4.0% of all preterm births" in 2010, the CDC noted.
That same year, ART infants were overrepresented among multiple births, with 19.2% of twin and 32.5% of triplet and higher-order births involving ART. These numbers, however, are the latest in a downward trend, as "the percentage of ART-conceived multiple infants declined by 13.0%" from 2000 to 2010, the CDC reported (MMWR Surveill. Summ. 2013;62(9):1-24).
According to data from the CDC's National ART Surveillance System, there were 147,260 ART procedures performed in 2010 at 443 fertility clinics, resulting in 47,090 live-birth deliveries and 61,564 infants.

FROM MMWR