User login
Physicians want to vaccinate, but money gets in the way
While almost all internists and family physicians feel a responsibility to ask about patients’ immunization status at least annually, most refer them to another source for all but basic seasonal vaccines.
A national survey determined that less than a third of these primary care providers stocked all 11 of the recommended adult vaccines. Financial issues were the biggest barrier to keeping all of the vaccines on hand and administering them, Dr. Laura Hurley and her colleagues wrote in the February issue of Annals of Internal Medicine (Ann. Int. Med. 2014;160:161-70).
"Although primary care physicians appear motivated to ensure that patients are up to date on vaccinations, many barriers exist. Implementation of system changes, including adopting practices that improve communication between primary care physicians and alternate vaccinators, more widespread use of effective tools ... and removing policy-related barriers, could improve adult vaccination in the United States," wrote Dr. Hurley of the Wellington E. Webb Center for Primary Care, Denver, and her coauthors.
Conducted by the Vaccine Policy Collaborative Initiative (VPCI), the survey assessed how responsible primary care physicians feel about adult vaccination, how often they assess and provide it, and what the barriers are to how vaccination is handled. The survey was mailed to 443 general internists and 409 family physicians, of whom 352 and 255 physicians, respectively, responded.
Ninety-seven percent of both groups said they assessed vaccination status at annual visits, but only about a third reported doing so at each visit.
Automated systems were fairly low down on the list of assessment techniques, although more family doctors than internists reported using them (36% vs. 8%). About half of all the respondents said it was moderately to very difficult to get up-to-date information on anything but the seasonal influenza vaccine.
In addition to the flu vaccine, most physicians checked on the status of pneumococcal; tetanus, diphtheria, and pertussis (Td and Tdap); and shingles vaccines. Family doctors were more likely to check on hepatitis A and B; MMR; and varicella vaccines.
Most physicians reported stocking influenza, pneumococcal, Td, and Tdap vaccines. Family physicians were more likely to stock both hepatitis vaccines, as well as those for human papillomavirus, varicella, meningitis, and shingles. Thirty-one percent of family physicians and 20% of internists said they routinely stocked all 11 of the recommended adult vaccines.
Money issues were the biggest barriers to stocking and administering vaccines, the respondents said. Commonly cited problems were lack of reimbursement for purchasing and administering; lack of insurance coverage for vaccines; and the up-front cost of buying the vaccine. Some providers worried the vaccine would expire before it could all be used, and others said patients simply refused them because they couldn’t pay.
Physicians in private and small group practices reported the biggest financial barriers, as did those who served a large number of Medicare Part D patients.
But even if they couldn’t provide the service themselves, respondents were sending patients to places that could – mostly pharmacies and public health departments. Still, financial issues were the biggest reasons for referrals; lack of insurance coverage or low insurance reimbursement figured prominently.
That can be a tricky proposition, however, said Dr. Jonathan Temte, chair of the Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices.
Section 317 of the Public Health Service Act provides federally purchased vaccine for free immunizations for qualifying patients, including uninsured adults. But underinsured adults or those who have very high insurance deductibles can’t get free immunizations with 317-purchased vaccines.
"Changes in the federal 317 rules prohibit adult patients who are insured from receiving vaccines without charge through public health agencies," Dr. Temte said in an interview. "Accordingly, the practice of ‘referring adults to public health’ for immunization doesn’t work too well."
When they did send patients to another source, most respondents were able to follow up on vaccination status. The most common way was to communicate with the vaccination provider. About a third of the two groups used an immunization information system. The information was usually recorded in the patient’s electronic health record or in a written chart or vaccine record.
"Of note," the authors said, "almost all physicians agreed that it was the primary care physician’s responsibility to see that patients receive recommended vaccines, even if they did so elsewhere."
The CDC sponsored the survey; none of the authors had any financial disclosures.
While almost all internists and family physicians feel a responsibility to ask about patients’ immunization status at least annually, most refer them to another source for all but basic seasonal vaccines.
A national survey determined that less than a third of these primary care providers stocked all 11 of the recommended adult vaccines. Financial issues were the biggest barrier to keeping all of the vaccines on hand and administering them, Dr. Laura Hurley and her colleagues wrote in the February issue of Annals of Internal Medicine (Ann. Int. Med. 2014;160:161-70).
"Although primary care physicians appear motivated to ensure that patients are up to date on vaccinations, many barriers exist. Implementation of system changes, including adopting practices that improve communication between primary care physicians and alternate vaccinators, more widespread use of effective tools ... and removing policy-related barriers, could improve adult vaccination in the United States," wrote Dr. Hurley of the Wellington E. Webb Center for Primary Care, Denver, and her coauthors.
Conducted by the Vaccine Policy Collaborative Initiative (VPCI), the survey assessed how responsible primary care physicians feel about adult vaccination, how often they assess and provide it, and what the barriers are to how vaccination is handled. The survey was mailed to 443 general internists and 409 family physicians, of whom 352 and 255 physicians, respectively, responded.
Ninety-seven percent of both groups said they assessed vaccination status at annual visits, but only about a third reported doing so at each visit.
Automated systems were fairly low down on the list of assessment techniques, although more family doctors than internists reported using them (36% vs. 8%). About half of all the respondents said it was moderately to very difficult to get up-to-date information on anything but the seasonal influenza vaccine.
In addition to the flu vaccine, most physicians checked on the status of pneumococcal; tetanus, diphtheria, and pertussis (Td and Tdap); and shingles vaccines. Family doctors were more likely to check on hepatitis A and B; MMR; and varicella vaccines.
Most physicians reported stocking influenza, pneumococcal, Td, and Tdap vaccines. Family physicians were more likely to stock both hepatitis vaccines, as well as those for human papillomavirus, varicella, meningitis, and shingles. Thirty-one percent of family physicians and 20% of internists said they routinely stocked all 11 of the recommended adult vaccines.
Money issues were the biggest barriers to stocking and administering vaccines, the respondents said. Commonly cited problems were lack of reimbursement for purchasing and administering; lack of insurance coverage for vaccines; and the up-front cost of buying the vaccine. Some providers worried the vaccine would expire before it could all be used, and others said patients simply refused them because they couldn’t pay.
Physicians in private and small group practices reported the biggest financial barriers, as did those who served a large number of Medicare Part D patients.
But even if they couldn’t provide the service themselves, respondents were sending patients to places that could – mostly pharmacies and public health departments. Still, financial issues were the biggest reasons for referrals; lack of insurance coverage or low insurance reimbursement figured prominently.
That can be a tricky proposition, however, said Dr. Jonathan Temte, chair of the Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices.
Section 317 of the Public Health Service Act provides federally purchased vaccine for free immunizations for qualifying patients, including uninsured adults. But underinsured adults or those who have very high insurance deductibles can’t get free immunizations with 317-purchased vaccines.
"Changes in the federal 317 rules prohibit adult patients who are insured from receiving vaccines without charge through public health agencies," Dr. Temte said in an interview. "Accordingly, the practice of ‘referring adults to public health’ for immunization doesn’t work too well."
When they did send patients to another source, most respondents were able to follow up on vaccination status. The most common way was to communicate with the vaccination provider. About a third of the two groups used an immunization information system. The information was usually recorded in the patient’s electronic health record or in a written chart or vaccine record.
"Of note," the authors said, "almost all physicians agreed that it was the primary care physician’s responsibility to see that patients receive recommended vaccines, even if they did so elsewhere."
The CDC sponsored the survey; none of the authors had any financial disclosures.
While almost all internists and family physicians feel a responsibility to ask about patients’ immunization status at least annually, most refer them to another source for all but basic seasonal vaccines.
A national survey determined that less than a third of these primary care providers stocked all 11 of the recommended adult vaccines. Financial issues were the biggest barrier to keeping all of the vaccines on hand and administering them, Dr. Laura Hurley and her colleagues wrote in the February issue of Annals of Internal Medicine (Ann. Int. Med. 2014;160:161-70).
"Although primary care physicians appear motivated to ensure that patients are up to date on vaccinations, many barriers exist. Implementation of system changes, including adopting practices that improve communication between primary care physicians and alternate vaccinators, more widespread use of effective tools ... and removing policy-related barriers, could improve adult vaccination in the United States," wrote Dr. Hurley of the Wellington E. Webb Center for Primary Care, Denver, and her coauthors.
Conducted by the Vaccine Policy Collaborative Initiative (VPCI), the survey assessed how responsible primary care physicians feel about adult vaccination, how often they assess and provide it, and what the barriers are to how vaccination is handled. The survey was mailed to 443 general internists and 409 family physicians, of whom 352 and 255 physicians, respectively, responded.
Ninety-seven percent of both groups said they assessed vaccination status at annual visits, but only about a third reported doing so at each visit.
Automated systems were fairly low down on the list of assessment techniques, although more family doctors than internists reported using them (36% vs. 8%). About half of all the respondents said it was moderately to very difficult to get up-to-date information on anything but the seasonal influenza vaccine.
In addition to the flu vaccine, most physicians checked on the status of pneumococcal; tetanus, diphtheria, and pertussis (Td and Tdap); and shingles vaccines. Family doctors were more likely to check on hepatitis A and B; MMR; and varicella vaccines.
Most physicians reported stocking influenza, pneumococcal, Td, and Tdap vaccines. Family physicians were more likely to stock both hepatitis vaccines, as well as those for human papillomavirus, varicella, meningitis, and shingles. Thirty-one percent of family physicians and 20% of internists said they routinely stocked all 11 of the recommended adult vaccines.
Money issues were the biggest barriers to stocking and administering vaccines, the respondents said. Commonly cited problems were lack of reimbursement for purchasing and administering; lack of insurance coverage for vaccines; and the up-front cost of buying the vaccine. Some providers worried the vaccine would expire before it could all be used, and others said patients simply refused them because they couldn’t pay.
Physicians in private and small group practices reported the biggest financial barriers, as did those who served a large number of Medicare Part D patients.
But even if they couldn’t provide the service themselves, respondents were sending patients to places that could – mostly pharmacies and public health departments. Still, financial issues were the biggest reasons for referrals; lack of insurance coverage or low insurance reimbursement figured prominently.
That can be a tricky proposition, however, said Dr. Jonathan Temte, chair of the Centers for Disease Control and Prevention’s (CDC’s) Advisory Committee on Immunization Practices.
Section 317 of the Public Health Service Act provides federally purchased vaccine for free immunizations for qualifying patients, including uninsured adults. But underinsured adults or those who have very high insurance deductibles can’t get free immunizations with 317-purchased vaccines.
"Changes in the federal 317 rules prohibit adult patients who are insured from receiving vaccines without charge through public health agencies," Dr. Temte said in an interview. "Accordingly, the practice of ‘referring adults to public health’ for immunization doesn’t work too well."
When they did send patients to another source, most respondents were able to follow up on vaccination status. The most common way was to communicate with the vaccination provider. About a third of the two groups used an immunization information system. The information was usually recorded in the patient’s electronic health record or in a written chart or vaccine record.
"Of note," the authors said, "almost all physicians agreed that it was the primary care physician’s responsibility to see that patients receive recommended vaccines, even if they did so elsewhere."
The CDC sponsored the survey; none of the authors had any financial disclosures.
FROM ANNALS OF INTERNAL MEDICINE
Major finding: Although about 90% of primary care providers assessed patients’ vaccination status at least once a year, less than a third stocked all 11 of the recommended adult vaccines.
Data source: The survey was mailed to 443 general internists and 409 family physicians.
Disclosures: The CDC sponsored the survey; none of the authors had any financial disclosures.
2014 childhood and adolescent immunization schedule now available
The 2014 childhood and adolescent immunization schedule has been approved, with additions that include the use of one of the meningococcal conjugate vaccines (Menveo) in certain groups of high-risk infants and a list specifying the groups of people at increased risk of hepatitis A.
The schedule will be published in the February 2014 issue of Pediatrics, and is being made available online on Jan. 31 (Pediatrics 2014 [doi: 10.1542/peds.2013-3965]).
Guidance on the use of Menveo (Meningococcal Groups A, C, W-135, and Y Oligosaccharide Diphtheria CRM197 conjugate vaccine) for certain groups of infants at increased risk of disease starting at age 2 months has been added to the meningococcal vaccine footnote. This is based on the Food and Drug Administration licensure of Menveo for use starting at age 2 months in August 2013. Some of the high-risk categories include anatomic or functional asplenia, including sickle cell disease; children with persistent complement component deficiency; and those who travel to or live in an area hyperendemic area for meningococcal disease.
This is the first time that a meningococcal vaccine has been available for use starting at age 2 months, Dr. H. Cody Meissner, professor of pediatrics at Tufts University, Boston, pointed out in an interview.
The hepatitis A vaccine footnote now provides a list of groups at increased risk for hepatitis A. While these groups are well recognized, "it was useful to itemize these groups in the footnote," noted Dr. Meissner, who is a member of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices work group on the harmonized immunization schedule.
The list includes people traveling to or working in countries where there is a high or intermediate endemicity of infection; men having sex with men; people with clotting factor disorders; people with chronic liver disease; users of injection and noninjection illicit drugs; and personal contacts – such as household contacts or regular babysitters – of international adoptees during the first 2 months of arrival in the United States "from a country with high or intermediate endemicity."
The footnote on the tetanus and diphtheria toxoids and acellular pertussis (Tdap) vaccine now states that a dose of the vaccine is recommended for pregnant adolescents every time they get pregnant – preferably during week 27 through week 36 of gestation. Last year, the American Academy of Pediatrics agreed that this vaccine should be given to a pregnant woman, but it withheld the recommendation to vaccinate during every pregnancy until more data became available, Dr. Meissner said. "Additional data now indicate the safety and efficacy of administration of Tdap each time a woman becomes pregnant." This will protect most infants during the first 2 months of life when pertussis can be most severe and until they receive their first DTaP dose at 2 months, he noted.
Other changes include clarification of the intervals between doses in the human papillomavirus (HPV) vaccines footnote, to avoid any misunderstanding of the schedule, said Dr. Meissner, who is also chief of pediatric infectious disease at the Floating Hospital for Children at Tufts Medical Center.
The recommendation for pneumococcal vaccines has not changed from last year, but the footnote provides clarification about the recommendations for PCV13 (Prevnar 13) and PPSV23 in children and adolescents, "which have been stratified according to age and according to degree of risk," he added.
The footnote on Haemophilus influenzae type b (Hib) conjugate vaccine now includes clarification of who should receive the vaccine if immunocompromised.
This is the second year that recommendations and footnotes for ages 0-18 years are included in one schedule, as opposed to previous years, when there were separate schedules and footnotes for 0-7 years and 8-18 years.
The 2014 schedules have been approved by the American Academy of Pediatrics, the Advisory Committee on Immunization Practices, the American Academy of Family Physicians, and the American College of Obstetricians and Gynecologists.
Dr. Meissner said he has no relevant financial disclosures.
You can view the 2014 schedule at our Resources section here.
The 2014 childhood and adolescent immunization schedule has been approved, with additions that include the use of one of the meningococcal conjugate vaccines (Menveo) in certain groups of high-risk infants and a list specifying the groups of people at increased risk of hepatitis A.
The schedule will be published in the February 2014 issue of Pediatrics, and is being made available online on Jan. 31 (Pediatrics 2014 [doi: 10.1542/peds.2013-3965]).
Guidance on the use of Menveo (Meningococcal Groups A, C, W-135, and Y Oligosaccharide Diphtheria CRM197 conjugate vaccine) for certain groups of infants at increased risk of disease starting at age 2 months has been added to the meningococcal vaccine footnote. This is based on the Food and Drug Administration licensure of Menveo for use starting at age 2 months in August 2013. Some of the high-risk categories include anatomic or functional asplenia, including sickle cell disease; children with persistent complement component deficiency; and those who travel to or live in an area hyperendemic area for meningococcal disease.
This is the first time that a meningococcal vaccine has been available for use starting at age 2 months, Dr. H. Cody Meissner, professor of pediatrics at Tufts University, Boston, pointed out in an interview.
The hepatitis A vaccine footnote now provides a list of groups at increased risk for hepatitis A. While these groups are well recognized, "it was useful to itemize these groups in the footnote," noted Dr. Meissner, who is a member of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices work group on the harmonized immunization schedule.
The list includes people traveling to or working in countries where there is a high or intermediate endemicity of infection; men having sex with men; people with clotting factor disorders; people with chronic liver disease; users of injection and noninjection illicit drugs; and personal contacts – such as household contacts or regular babysitters – of international adoptees during the first 2 months of arrival in the United States "from a country with high or intermediate endemicity."
The footnote on the tetanus and diphtheria toxoids and acellular pertussis (Tdap) vaccine now states that a dose of the vaccine is recommended for pregnant adolescents every time they get pregnant – preferably during week 27 through week 36 of gestation. Last year, the American Academy of Pediatrics agreed that this vaccine should be given to a pregnant woman, but it withheld the recommendation to vaccinate during every pregnancy until more data became available, Dr. Meissner said. "Additional data now indicate the safety and efficacy of administration of Tdap each time a woman becomes pregnant." This will protect most infants during the first 2 months of life when pertussis can be most severe and until they receive their first DTaP dose at 2 months, he noted.
Other changes include clarification of the intervals between doses in the human papillomavirus (HPV) vaccines footnote, to avoid any misunderstanding of the schedule, said Dr. Meissner, who is also chief of pediatric infectious disease at the Floating Hospital for Children at Tufts Medical Center.
The recommendation for pneumococcal vaccines has not changed from last year, but the footnote provides clarification about the recommendations for PCV13 (Prevnar 13) and PPSV23 in children and adolescents, "which have been stratified according to age and according to degree of risk," he added.
The footnote on Haemophilus influenzae type b (Hib) conjugate vaccine now includes clarification of who should receive the vaccine if immunocompromised.
This is the second year that recommendations and footnotes for ages 0-18 years are included in one schedule, as opposed to previous years, when there were separate schedules and footnotes for 0-7 years and 8-18 years.
The 2014 schedules have been approved by the American Academy of Pediatrics, the Advisory Committee on Immunization Practices, the American Academy of Family Physicians, and the American College of Obstetricians and Gynecologists.
Dr. Meissner said he has no relevant financial disclosures.
You can view the 2014 schedule at our Resources section here.
The 2014 childhood and adolescent immunization schedule has been approved, with additions that include the use of one of the meningococcal conjugate vaccines (Menveo) in certain groups of high-risk infants and a list specifying the groups of people at increased risk of hepatitis A.
The schedule will be published in the February 2014 issue of Pediatrics, and is being made available online on Jan. 31 (Pediatrics 2014 [doi: 10.1542/peds.2013-3965]).
Guidance on the use of Menveo (Meningococcal Groups A, C, W-135, and Y Oligosaccharide Diphtheria CRM197 conjugate vaccine) for certain groups of infants at increased risk of disease starting at age 2 months has been added to the meningococcal vaccine footnote. This is based on the Food and Drug Administration licensure of Menveo for use starting at age 2 months in August 2013. Some of the high-risk categories include anatomic or functional asplenia, including sickle cell disease; children with persistent complement component deficiency; and those who travel to or live in an area hyperendemic area for meningococcal disease.
This is the first time that a meningococcal vaccine has been available for use starting at age 2 months, Dr. H. Cody Meissner, professor of pediatrics at Tufts University, Boston, pointed out in an interview.
The hepatitis A vaccine footnote now provides a list of groups at increased risk for hepatitis A. While these groups are well recognized, "it was useful to itemize these groups in the footnote," noted Dr. Meissner, who is a member of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices work group on the harmonized immunization schedule.
The list includes people traveling to or working in countries where there is a high or intermediate endemicity of infection; men having sex with men; people with clotting factor disorders; people with chronic liver disease; users of injection and noninjection illicit drugs; and personal contacts – such as household contacts or regular babysitters – of international adoptees during the first 2 months of arrival in the United States "from a country with high or intermediate endemicity."
The footnote on the tetanus and diphtheria toxoids and acellular pertussis (Tdap) vaccine now states that a dose of the vaccine is recommended for pregnant adolescents every time they get pregnant – preferably during week 27 through week 36 of gestation. Last year, the American Academy of Pediatrics agreed that this vaccine should be given to a pregnant woman, but it withheld the recommendation to vaccinate during every pregnancy until more data became available, Dr. Meissner said. "Additional data now indicate the safety and efficacy of administration of Tdap each time a woman becomes pregnant." This will protect most infants during the first 2 months of life when pertussis can be most severe and until they receive their first DTaP dose at 2 months, he noted.
Other changes include clarification of the intervals between doses in the human papillomavirus (HPV) vaccines footnote, to avoid any misunderstanding of the schedule, said Dr. Meissner, who is also chief of pediatric infectious disease at the Floating Hospital for Children at Tufts Medical Center.
The recommendation for pneumococcal vaccines has not changed from last year, but the footnote provides clarification about the recommendations for PCV13 (Prevnar 13) and PPSV23 in children and adolescents, "which have been stratified according to age and according to degree of risk," he added.
The footnote on Haemophilus influenzae type b (Hib) conjugate vaccine now includes clarification of who should receive the vaccine if immunocompromised.
This is the second year that recommendations and footnotes for ages 0-18 years are included in one schedule, as opposed to previous years, when there were separate schedules and footnotes for 0-7 years and 8-18 years.
The 2014 schedules have been approved by the American Academy of Pediatrics, the Advisory Committee on Immunization Practices, the American Academy of Family Physicians, and the American College of Obstetricians and Gynecologists.
Dr. Meissner said he has no relevant financial disclosures.
You can view the 2014 schedule at our Resources section here.
FROM PEDIATRICS
Hyperprolactinemia: Causes and Treatments
A 31-year-old woman is referred by her Ob-Gyn for elevated prolactin. She initially presented with a three-month history of amenorrhea, a negative home pregnancy test, and 100% compliance with condom use. She denies hirsutism and acne but admits to thin milky nipple discharge upon squeezing (but not spontaneous).
Two weeks ago, her Ob-Gyn ordered labs; results were negative for serum beta human chorionic gonadotropin and within normal ranges for thyroid-stimulating hormone (TSH), luteinizing hormone, follicle-stimulating hormone, estradiol, free and total testosterone, dehydroepiandrosterone sulfate (DHEAs), complete chemistry panel, and complete blood count. Her serum prolactin level was 110 ng/mL (normal, 3 to 27 ng/mL).
Q: How is prolactin physiologically regulated?
The primary role of prolactin, which is produced by lactotroph cells in the anterior pituitary gland, is to stimulate lactation and breast development. Prolactin is regulated by dopamine (also known as prolactin inhibitory hormone), which is secreted from the hypothalamus via an inhibitory pathway unique to the hypothalamus-pituitary hormone system. Dopamine essentially suppresses prolactin.
Other hormones can have a stimulatory effect on the anterior pituitary gland and thus increase prolactin levels. Estrogen can induce lactotroph hyperplasia and elevated prolactin; however, this is only clinically relevant in the context of estrogen surge during pregnancy. (Estrogen therapy, such as oral contraception or hormone replacement therapy, on the other hand, is targeted to “normal” estrogen levels.) Thyrotropin-releasing hormone (TRH) from the hypothalamus also stimulates the anterior pituitary gland, so patients with inadequately treated or untreated primary hypothyroidism will have mildly elevated prolactin.
Neurogenic stimuli of the chest wall, through nipple suckling or varicella zoster infection (shingles), can also increase prolactin secretion. And since prolactin is eliminated by the liver (75%) and the kidney (25%), significant liver disease and/or renal insufficiency can raise prolactin levels, due to decreased clearance.
What are the possible etiologies for elevated prolactin? See answer on the next page...
Q: What are the possible etiologies for elevated prolactin?
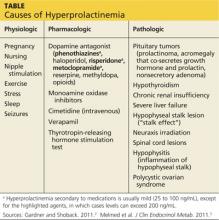
The causes of hyperprolactinemia fall into three categories: physiologic, pharmacologic, and pathologic.2 The table provides examples from each category.
A nonsecretory pituitary adenoma or any lesion in the brain that would disrupt the hypophyseal stalk may interfere with dopamine’s inhibitory control and thereby increase prolactin. This is called the stalk effect. It is important to note that not all MRI-proven pituitary adenomas are prolactin secreting, even in the presence of hyperprolactinemia. According to an autopsy series, about 12% of the general population had pituitary microadenoma.3
There is rough correlation between prolactinoma size and level of prolactin. Large nonsecretory pituitary adenomas have prolactin levels less than 150 ng/mL. Microprolactinomas (< 1 cm) are usually in the range of 100 to 250 ng/mL, while macroprolactinomas (> 1 cm) are generally
≥ 250 ng/mL. If the tumor is very large and invades the cavernous sinus, prolactin can measure in the 1,000s.3
Polycystic ovarian syndrome (PCOS) is a common disorder affecting women of reproductive age and the most common cause of underlying ovulatory problems. Patients with PCOS can have mildly elevated prolactin; the exact mechanism of hyperprolactinemia in PCOS is unknown. One theory is that constant high levels of estrogen experienced in PCOS would stimulate prolactin production. It is important to rule out other causes of hyperprolactinemia before making the diagnosis of PCOS.
What is the clinical significance of elevated prolactin? Why do we have to work up and treat it? See answer on the next page...
Q: What is the clinical significance of elevated prolactin? Why do we have to work up and treat it?
By physiologic mechanisms not completely understood, hyperprolactinemia can interrupt the gonadal axis, leading to hypogonadism. In women, it can cause irregular menstrual cycles, oligomenorrhea, amenorrhea, and infertility. In men, it can lower testosterone levels. Long-term effects include declining bone mineral density due to insufficient estrogen in women or testosterone in men.
With macroadenoma, the size of the tumor can have a mass effect such as headache and visual defect by compressing the optic chiasm (bitemporal hemianopsia), which may lead to permanent vision loss if left untreated. Referral to an ophthalmologist may be necessary for formal visual field examination.
How is hyperprolactinemia treated? See answer on the next page...
Q: How is hyperprolactinemia treated?
There are three options for treatment: medication, surgery, and radiation.
Dopamine agonists (bromocriptine, cabergoline) are effective in normalizing prolactin and reducing the size of the tumor in the majority of cases. However, some patients may require long-term treatment. Bromocriptine has been used since the late 1970s, but, due to better tolerance and less frequent dosing, cabergoline is the preferred agent.3
Transsphenoidal surgery is indicated for patients who are intolerant to medication, who have a medication-resistant tumor or significant mass effect, or who prefer definitive treatment. Women of childbearing age with a macroadenoma might consider surgery due to the risk for tumor expansion during pregnancy (estrogen effect) and risk for pituitary apoplexy (hemorrhage or infarct of the pituitary gland). Surgical risk is usually low with a neurosurgeon who has extensive experience.
Radiation can be considered for large tumors that are resistant to medication. It can be used as adjunctive therapy to surgery, since reducing the size of the tumor can make the surgical field smaller. In some medication-resistant tumors, radiation can raise sensitivity to medication.
What does follow-up entail? See next page for answer...
Q: What does follow-up entail?
Once medication is initiated or dosage is adjusted, have the patient follow up in one month and recheck the prolactin level to assess responsiveness to medication (as well as medication adherence). When a therapeutic prolactin level is achieved, recheck the prolactin and have the patient follow up at three and six months and then every six months thereafter.3
MRI of the pituitary gland should be performed at baseline, then in six months to assess tumor response to medication, and then at 12 and 24 months.3 If tumor regression has stabilized or if the tumor has shrunk to a nondetectable size, consider discontinuing the dopamine agonist. If medication is discontinued, recheck prolactin every three months for the first year; if it remains in normal reference range, simply check serum prolactin annually.3
See next page for summary.
See next page for references.
REFERENCES
1. Jameson JL. Harrison’s Endocrinology. 18th ed. China: McGraw-Hill; 2010.
2. Gardner D, Shoback D. Greenspan’s Basic & Clinical Endocrinology. 9th ed. China: McGraw-Hill; 2011.
3. Melmed S, Casanueva FF, Hoffman AR, et al. Diagnosis and treatment of hyperprolactinemia: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2011;96(2):273-288.
A 31-year-old woman is referred by her Ob-Gyn for elevated prolactin. She initially presented with a three-month history of amenorrhea, a negative home pregnancy test, and 100% compliance with condom use. She denies hirsutism and acne but admits to thin milky nipple discharge upon squeezing (but not spontaneous).
Two weeks ago, her Ob-Gyn ordered labs; results were negative for serum beta human chorionic gonadotropin and within normal ranges for thyroid-stimulating hormone (TSH), luteinizing hormone, follicle-stimulating hormone, estradiol, free and total testosterone, dehydroepiandrosterone sulfate (DHEAs), complete chemistry panel, and complete blood count. Her serum prolactin level was 110 ng/mL (normal, 3 to 27 ng/mL).
Q: How is prolactin physiologically regulated?
The primary role of prolactin, which is produced by lactotroph cells in the anterior pituitary gland, is to stimulate lactation and breast development. Prolactin is regulated by dopamine (also known as prolactin inhibitory hormone), which is secreted from the hypothalamus via an inhibitory pathway unique to the hypothalamus-pituitary hormone system. Dopamine essentially suppresses prolactin.
Other hormones can have a stimulatory effect on the anterior pituitary gland and thus increase prolactin levels. Estrogen can induce lactotroph hyperplasia and elevated prolactin; however, this is only clinically relevant in the context of estrogen surge during pregnancy. (Estrogen therapy, such as oral contraception or hormone replacement therapy, on the other hand, is targeted to “normal” estrogen levels.) Thyrotropin-releasing hormone (TRH) from the hypothalamus also stimulates the anterior pituitary gland, so patients with inadequately treated or untreated primary hypothyroidism will have mildly elevated prolactin.
Neurogenic stimuli of the chest wall, through nipple suckling or varicella zoster infection (shingles), can also increase prolactin secretion. And since prolactin is eliminated by the liver (75%) and the kidney (25%), significant liver disease and/or renal insufficiency can raise prolactin levels, due to decreased clearance.
What are the possible etiologies for elevated prolactin? See answer on the next page...
Q: What are the possible etiologies for elevated prolactin?
The causes of hyperprolactinemia fall into three categories: physiologic, pharmacologic, and pathologic.2 The table provides examples from each category.
A nonsecretory pituitary adenoma or any lesion in the brain that would disrupt the hypophyseal stalk may interfere with dopamine’s inhibitory control and thereby increase prolactin. This is called the stalk effect. It is important to note that not all MRI-proven pituitary adenomas are prolactin secreting, even in the presence of hyperprolactinemia. According to an autopsy series, about 12% of the general population had pituitary microadenoma.3
There is rough correlation between prolactinoma size and level of prolactin. Large nonsecretory pituitary adenomas have prolactin levels less than 150 ng/mL. Microprolactinomas (< 1 cm) are usually in the range of 100 to 250 ng/mL, while macroprolactinomas (> 1 cm) are generally
≥ 250 ng/mL. If the tumor is very large and invades the cavernous sinus, prolactin can measure in the 1,000s.3
Polycystic ovarian syndrome (PCOS) is a common disorder affecting women of reproductive age and the most common cause of underlying ovulatory problems. Patients with PCOS can have mildly elevated prolactin; the exact mechanism of hyperprolactinemia in PCOS is unknown. One theory is that constant high levels of estrogen experienced in PCOS would stimulate prolactin production. It is important to rule out other causes of hyperprolactinemia before making the diagnosis of PCOS.
What is the clinical significance of elevated prolactin? Why do we have to work up and treat it? See answer on the next page...
Q: What is the clinical significance of elevated prolactin? Why do we have to work up and treat it?
By physiologic mechanisms not completely understood, hyperprolactinemia can interrupt the gonadal axis, leading to hypogonadism. In women, it can cause irregular menstrual cycles, oligomenorrhea, amenorrhea, and infertility. In men, it can lower testosterone levels. Long-term effects include declining bone mineral density due to insufficient estrogen in women or testosterone in men.
With macroadenoma, the size of the tumor can have a mass effect such as headache and visual defect by compressing the optic chiasm (bitemporal hemianopsia), which may lead to permanent vision loss if left untreated. Referral to an ophthalmologist may be necessary for formal visual field examination.
How is hyperprolactinemia treated? See answer on the next page...
Q: How is hyperprolactinemia treated?
There are three options for treatment: medication, surgery, and radiation.
Dopamine agonists (bromocriptine, cabergoline) are effective in normalizing prolactin and reducing the size of the tumor in the majority of cases. However, some patients may require long-term treatment. Bromocriptine has been used since the late 1970s, but, due to better tolerance and less frequent dosing, cabergoline is the preferred agent.3
Transsphenoidal surgery is indicated for patients who are intolerant to medication, who have a medication-resistant tumor or significant mass effect, or who prefer definitive treatment. Women of childbearing age with a macroadenoma might consider surgery due to the risk for tumor expansion during pregnancy (estrogen effect) and risk for pituitary apoplexy (hemorrhage or infarct of the pituitary gland). Surgical risk is usually low with a neurosurgeon who has extensive experience.
Radiation can be considered for large tumors that are resistant to medication. It can be used as adjunctive therapy to surgery, since reducing the size of the tumor can make the surgical field smaller. In some medication-resistant tumors, radiation can raise sensitivity to medication.
What does follow-up entail? See next page for answer...
Q: What does follow-up entail?
Once medication is initiated or dosage is adjusted, have the patient follow up in one month and recheck the prolactin level to assess responsiveness to medication (as well as medication adherence). When a therapeutic prolactin level is achieved, recheck the prolactin and have the patient follow up at three and six months and then every six months thereafter.3
MRI of the pituitary gland should be performed at baseline, then in six months to assess tumor response to medication, and then at 12 and 24 months.3 If tumor regression has stabilized or if the tumor has shrunk to a nondetectable size, consider discontinuing the dopamine agonist. If medication is discontinued, recheck prolactin every three months for the first year; if it remains in normal reference range, simply check serum prolactin annually.3
See next page for summary.
See next page for references.
REFERENCES
1. Jameson JL. Harrison’s Endocrinology. 18th ed. China: McGraw-Hill; 2010.
2. Gardner D, Shoback D. Greenspan’s Basic & Clinical Endocrinology. 9th ed. China: McGraw-Hill; 2011.
3. Melmed S, Casanueva FF, Hoffman AR, et al. Diagnosis and treatment of hyperprolactinemia: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2011;96(2):273-288.
A 31-year-old woman is referred by her Ob-Gyn for elevated prolactin. She initially presented with a three-month history of amenorrhea, a negative home pregnancy test, and 100% compliance with condom use. She denies hirsutism and acne but admits to thin milky nipple discharge upon squeezing (but not spontaneous).
Two weeks ago, her Ob-Gyn ordered labs; results were negative for serum beta human chorionic gonadotropin and within normal ranges for thyroid-stimulating hormone (TSH), luteinizing hormone, follicle-stimulating hormone, estradiol, free and total testosterone, dehydroepiandrosterone sulfate (DHEAs), complete chemistry panel, and complete blood count. Her serum prolactin level was 110 ng/mL (normal, 3 to 27 ng/mL).
Q: How is prolactin physiologically regulated?
The primary role of prolactin, which is produced by lactotroph cells in the anterior pituitary gland, is to stimulate lactation and breast development. Prolactin is regulated by dopamine (also known as prolactin inhibitory hormone), which is secreted from the hypothalamus via an inhibitory pathway unique to the hypothalamus-pituitary hormone system. Dopamine essentially suppresses prolactin.
Other hormones can have a stimulatory effect on the anterior pituitary gland and thus increase prolactin levels. Estrogen can induce lactotroph hyperplasia and elevated prolactin; however, this is only clinically relevant in the context of estrogen surge during pregnancy. (Estrogen therapy, such as oral contraception or hormone replacement therapy, on the other hand, is targeted to “normal” estrogen levels.) Thyrotropin-releasing hormone (TRH) from the hypothalamus also stimulates the anterior pituitary gland, so patients with inadequately treated or untreated primary hypothyroidism will have mildly elevated prolactin.
Neurogenic stimuli of the chest wall, through nipple suckling or varicella zoster infection (shingles), can also increase prolactin secretion. And since prolactin is eliminated by the liver (75%) and the kidney (25%), significant liver disease and/or renal insufficiency can raise prolactin levels, due to decreased clearance.
What are the possible etiologies for elevated prolactin? See answer on the next page...
Q: What are the possible etiologies for elevated prolactin?
The causes of hyperprolactinemia fall into three categories: physiologic, pharmacologic, and pathologic.2 The table provides examples from each category.
A nonsecretory pituitary adenoma or any lesion in the brain that would disrupt the hypophyseal stalk may interfere with dopamine’s inhibitory control and thereby increase prolactin. This is called the stalk effect. It is important to note that not all MRI-proven pituitary adenomas are prolactin secreting, even in the presence of hyperprolactinemia. According to an autopsy series, about 12% of the general population had pituitary microadenoma.3
There is rough correlation between prolactinoma size and level of prolactin. Large nonsecretory pituitary adenomas have prolactin levels less than 150 ng/mL. Microprolactinomas (< 1 cm) are usually in the range of 100 to 250 ng/mL, while macroprolactinomas (> 1 cm) are generally
≥ 250 ng/mL. If the tumor is very large and invades the cavernous sinus, prolactin can measure in the 1,000s.3
Polycystic ovarian syndrome (PCOS) is a common disorder affecting women of reproductive age and the most common cause of underlying ovulatory problems. Patients with PCOS can have mildly elevated prolactin; the exact mechanism of hyperprolactinemia in PCOS is unknown. One theory is that constant high levels of estrogen experienced in PCOS would stimulate prolactin production. It is important to rule out other causes of hyperprolactinemia before making the diagnosis of PCOS.
What is the clinical significance of elevated prolactin? Why do we have to work up and treat it? See answer on the next page...
Q: What is the clinical significance of elevated prolactin? Why do we have to work up and treat it?
By physiologic mechanisms not completely understood, hyperprolactinemia can interrupt the gonadal axis, leading to hypogonadism. In women, it can cause irregular menstrual cycles, oligomenorrhea, amenorrhea, and infertility. In men, it can lower testosterone levels. Long-term effects include declining bone mineral density due to insufficient estrogen in women or testosterone in men.
With macroadenoma, the size of the tumor can have a mass effect such as headache and visual defect by compressing the optic chiasm (bitemporal hemianopsia), which may lead to permanent vision loss if left untreated. Referral to an ophthalmologist may be necessary for formal visual field examination.
How is hyperprolactinemia treated? See answer on the next page...
Q: How is hyperprolactinemia treated?
There are three options for treatment: medication, surgery, and radiation.
Dopamine agonists (bromocriptine, cabergoline) are effective in normalizing prolactin and reducing the size of the tumor in the majority of cases. However, some patients may require long-term treatment. Bromocriptine has been used since the late 1970s, but, due to better tolerance and less frequent dosing, cabergoline is the preferred agent.3
Transsphenoidal surgery is indicated for patients who are intolerant to medication, who have a medication-resistant tumor or significant mass effect, or who prefer definitive treatment. Women of childbearing age with a macroadenoma might consider surgery due to the risk for tumor expansion during pregnancy (estrogen effect) and risk for pituitary apoplexy (hemorrhage or infarct of the pituitary gland). Surgical risk is usually low with a neurosurgeon who has extensive experience.
Radiation can be considered for large tumors that are resistant to medication. It can be used as adjunctive therapy to surgery, since reducing the size of the tumor can make the surgical field smaller. In some medication-resistant tumors, radiation can raise sensitivity to medication.
What does follow-up entail? See next page for answer...
Q: What does follow-up entail?
Once medication is initiated or dosage is adjusted, have the patient follow up in one month and recheck the prolactin level to assess responsiveness to medication (as well as medication adherence). When a therapeutic prolactin level is achieved, recheck the prolactin and have the patient follow up at three and six months and then every six months thereafter.3
MRI of the pituitary gland should be performed at baseline, then in six months to assess tumor response to medication, and then at 12 and 24 months.3 If tumor regression has stabilized or if the tumor has shrunk to a nondetectable size, consider discontinuing the dopamine agonist. If medication is discontinued, recheck prolactin every three months for the first year; if it remains in normal reference range, simply check serum prolactin annually.3
See next page for summary.
See next page for references.
REFERENCES
1. Jameson JL. Harrison’s Endocrinology. 18th ed. China: McGraw-Hill; 2010.
2. Gardner D, Shoback D. Greenspan’s Basic & Clinical Endocrinology. 9th ed. China: McGraw-Hill; 2011.
3. Melmed S, Casanueva FF, Hoffman AR, et al. Diagnosis and treatment of hyperprolactinemia: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2011;96(2):273-288.
House passes bill to ban federal funding of abortion
The U.S. House of Representatives has passed a bill that would bar the use of any federal funds in paying for an abortion, including tax subsidies to purchase insurance under the Affordable Care Act.
On Jan. 28, the House voted 227 to 188, with one lawmaker voting "present," to approve H.R. 7, the No Taxpayer Funding for Abortion Act. The bill would make permanent the Hyde Amendment, an annual rider placed on spending bills that prohibits federal funds from being used to pay for abortions. H.R. 7 would also bar individuals and small businesses from using federal tax subsidies under the Affordable Care Act (ACA) to purchase health plans that include coverage for abortion.
The bill includes exceptions in cases of rape or incest, or when the mother’s life is endangered.
The bill is not expected to advance in the Senate.
The bill’s sponsor, Rep. Chris Smith (R-N.J.), said that without the new legislation, the ACA would create a massive expansion of public funding for abortion. He estimated that of the 112 health plans available to members of Congress and their staff through the ACA, 103 include coverage of abortion services.
But reproductive health advocates said the bill interferes with a woman’s private health care decisions and attempts to restrict the health care services that can be offered in private insurance plans.
"This bill is as callous as it is cynical, denying insurance coverage of critical health care to millions of women across the U.S. and worsening especially the hardship of women already struggling under challenging economic conditions," Nancy Northup, president and CEO of the Center for Reproductive Rights, said in a statement.
On Twitter @maryellenny
The U.S. House of Representatives has passed a bill that would bar the use of any federal funds in paying for an abortion, including tax subsidies to purchase insurance under the Affordable Care Act.
On Jan. 28, the House voted 227 to 188, with one lawmaker voting "present," to approve H.R. 7, the No Taxpayer Funding for Abortion Act. The bill would make permanent the Hyde Amendment, an annual rider placed on spending bills that prohibits federal funds from being used to pay for abortions. H.R. 7 would also bar individuals and small businesses from using federal tax subsidies under the Affordable Care Act (ACA) to purchase health plans that include coverage for abortion.
The bill includes exceptions in cases of rape or incest, or when the mother’s life is endangered.
The bill is not expected to advance in the Senate.
The bill’s sponsor, Rep. Chris Smith (R-N.J.), said that without the new legislation, the ACA would create a massive expansion of public funding for abortion. He estimated that of the 112 health plans available to members of Congress and their staff through the ACA, 103 include coverage of abortion services.
But reproductive health advocates said the bill interferes with a woman’s private health care decisions and attempts to restrict the health care services that can be offered in private insurance plans.
"This bill is as callous as it is cynical, denying insurance coverage of critical health care to millions of women across the U.S. and worsening especially the hardship of women already struggling under challenging economic conditions," Nancy Northup, president and CEO of the Center for Reproductive Rights, said in a statement.
On Twitter @maryellenny
The U.S. House of Representatives has passed a bill that would bar the use of any federal funds in paying for an abortion, including tax subsidies to purchase insurance under the Affordable Care Act.
On Jan. 28, the House voted 227 to 188, with one lawmaker voting "present," to approve H.R. 7, the No Taxpayer Funding for Abortion Act. The bill would make permanent the Hyde Amendment, an annual rider placed on spending bills that prohibits federal funds from being used to pay for abortions. H.R. 7 would also bar individuals and small businesses from using federal tax subsidies under the Affordable Care Act (ACA) to purchase health plans that include coverage for abortion.
The bill includes exceptions in cases of rape or incest, or when the mother’s life is endangered.
The bill is not expected to advance in the Senate.
The bill’s sponsor, Rep. Chris Smith (R-N.J.), said that without the new legislation, the ACA would create a massive expansion of public funding for abortion. He estimated that of the 112 health plans available to members of Congress and their staff through the ACA, 103 include coverage of abortion services.
But reproductive health advocates said the bill interferes with a woman’s private health care decisions and attempts to restrict the health care services that can be offered in private insurance plans.
"This bill is as callous as it is cynical, denying insurance coverage of critical health care to millions of women across the U.S. and worsening especially the hardship of women already struggling under challenging economic conditions," Nancy Northup, president and CEO of the Center for Reproductive Rights, said in a statement.
On Twitter @maryellenny
We need honest, respectful discussion about the risks of home birth
November 2013
We need honest, respectful discussion about the risks of home birth
I object to the tone of the recent commentary by Dr. Errol Norwitz. Although I agree with the concerns he raised, I object to his “tactics.” The subject line of the email I received about this commentary (“Let’s keep home delivery where it belongs…for pizza!”) and some of the comments in the article were inflammatory and almost seem to be making fun of a topic about which many women in our country feel passionately.
As health-care providers, we are skilled at explaining risks and helping people understand how their choices and actions affect those risks. We do this daily without offering insult to patients regarding their beliefs. You don’t need to convince ObGyns that home birth is unsafe—I daresay that most of them already believe that. Awareness of the data and even some of Dr. Norwitz’s insights surely are valuable to the people who are actually pursuing and performing home births—but I feel that the subject line and closing remarks were mean-spirited and serve only to push practitioners and even patients—who need information to make informed decisions—farther away from what Dr. Norwitz argues is the safest care. No one responds well to mockery.
I practice the full scope of nurse-midwifery in a hospital-based practice with excellent physicians to support me and my patients. I do not attend home births, and I, too, have concerns about some home-birth practices and statistics. Honest, respectful communication about the facts is what is needed to improve patient safety and outcomes—not marginalization and disrespect.
Jamie A. Otremba, CNM
Waconia, Minnesota
Hospital deliveries can have problems, too
This article was insulting. When the study by Cheng and colleagues1 was first published, it received enormous criticism, so why has it been revived? Planned home birth with an appropriate provider is safe. The absolute numbers for maternal and newborn problems are very low but sound high when couched in warning language. Let’s not forget the mothers and babies who have problems in the hospital. In fact, the United States has a terrible record when it comes to maternal and neonatal morbidity and mortality—figures based almost entirely on hospital deliveries.
Low-risk women choose home birth to avoid a delivery that often results in far too many non-evidence-based interventions. Why don’t we change the system to make it work? If ObGyns would participate in a collaborative system of care that allows seamless integration of midwives into the acute-care setting when women are no longer low-risk, home-birth statistics would improve tremendously.
ObGyns are surgeons and specialists in problem-solving. Midwives are specialists in low-risk care, and they are adept at continuously assessing their patients to assure that they remain low-risk, consulting or transferring when necessary. Far too many physicians are “turf-conscious,” refusing to collaborate with midwives. It sounds to me as though the risk in home birth is iatrogenic, caused by the very physicians who claim that it is unsafe.
Chris Hilderbrandt, ARNP, CNM
Largo, Florida
Why are real changes taking so long?
I believe that the risk figures—bolded and supersized at the beginning of Dr. Norwitz’s commentary—are somewhat misleading. The intended message is clear but something very important is missing: The scores and risk values for home births were not isolated and compared between skilled midwives attending the births versus unskilled and untrained “other midwives.” In that light, when one considers the difference in risk—0.37% risk for an Apgar score of less than 4 for a home birth (which involves mainly nonprofessional labor attendants) and 0.24% for a hospitalized, medically managed birth, it is remarkable that the difference—0.13%—is so small.
I realize the importance of striving to bring any risk close to zero, but the way the data were presented unfairly implies that home births are essentially more risky than hospital births. It simply may be that they are attended by less-skilled people.
In addition, Dr. Norwitz noted that, in countries where home birth is integrated into the health-care system, there is a twofold to threefold increase in neonatal death. However, a 2011 article by van der Kooy and colleagues2 contradicts that claim, showing no significant difference in neonatal deaths between planned home birth and hospital birth for low-risk women in the Netherlands. I suppose, depending on the vantage point, there is a study somewhere that will support any of our beliefs.
Please make no mistake. I have never been a proponent of home birth. Even a small risk becomes 100% for the individual who suffers any kind of ill effects or damage. But I am a promoter of natural birth and minimal medical intervention. I abandoned obstetrics years ago when I saw firsthand that the increasing focus on technology and medical intervention moved the woman and her experience to the outer margins. The situation has not improved, and the strides made early on by the women’s movement have all but disappeared as women are aggressively convinced that technology and “controlled” interventions will grant them safe passage. In the process, they lose a sense of their autonomy and participation in one of the most important events in their lives.
At the end of the article, Dr. Norwitz leaves us with a notion that obstetric care providers need to do more in the way of providing emotional and social support. My question is… What in the world are you waiting for?
Obstetric care has a very long way to go before the focus moves meaningfully toward handing back some of the power of choice to the pregnant woman, something early feminists championed. That would require the obstetrician to honor and listen to the woman as she begins to formulate a delivery plan, to support her input at every step of decision-making, to provide her with or refer her to sources of information about delivery alternatives—the very types of alternatives that are readily available in midwife-attended births.
Jacquelynn Cunliffe, MSN, PhD
Wayne, Pennsylvania
Dr. Norwitz responds
We all want a healthy infant
Regardless of the type of training or level of experience, all obstetric care providers want the same outcome: an uncomplicated delivery of a healthy infant under conditions that are safe and supportive.
Contrary to popular belief, Nature is not a good obstetrician. It is estimated that between 1 in 50 and 1 in 500 fetuses reach maturity in utero and are subsequently involved in a catastrophic event that results in severe neurologic damage or perinatal death.3 Many of these catastrophic events occur during labor. Even a woman categorized as “low-risk” throughout her pregnancy can become “high-risk” in a matter of minutes if she develops a complication during labor such as cord prolapse or placental abruption. Risk factors for such intrapartum complications have been described, but these complications can develop in anyone at any time, even in women with no risk factors at all.
As noted in the letter from Dr. Cunliffe and the article by Cheng and colleagues,4 the level of training of the person attending the planned home birth may well affect the outcome. The less skilled the provider, the less likely he or she is to anticipate and recognize a complication and the more likely an adverse event. The existing literature on this topic should not be interpreted as a criticism of the training or skill of certified nurse midwives. Even the most skilled birth attendant is ill-prepared to deal with the potential catastrophe of an intrapartum complication during a planned home birth, given the limited resources of a home environment.
The issue of planned home birth is an emotive one. Although every effort should be made to ensure that the birthing experience is a positive one, it should not be done at the expense of safety. Ms. Hilderbrandt’s claim that “Planned home birth with an appropriate provider is safe” is not supported by the existing data. Even in countries where home births are integrated fully into the medical care system, such deliveries are associated with a twofold to threefold increase in the odds of neonatal death.5 In the United States, where no such integration exists, a planned home birth is simply dangerous, although the absolute risk of a serious adverse event is low.
If a pregnant woman ever wants to know the safest place to deliver her baby, the unequivocal answer is: in a hospital setting. The question of who is best suited to attend the birth is far less critical, so long as the person—either a physician or a certified nurse midwife—has completed an accredited training program.
WE WANT TO HEAR FROM YOU!
Drop us a line and let us know what you think about current articles, which topics you'd like to see covered in future issues, and what challenges you face in daily practice. Tell us what you think by emailing us at: [email protected]
- Cheng YW, Snowden JM, King TL, Caughey AB. Selected perinatal outcomes associated with planned home births in the United States. Am J Obstet Gynecol. 2013;209(4):325.e1–e8. doi:10.1016/j.ajog.2013.06.022.
- van der Kooy J, Poeran J, de Graaf JP, et al. Planned home births compared with planned hospital births in the Netherlands: Intrapartum and early neonatal death in low-risk pregnancies. Obstet Gynecol. 2011;118(5):1037–1046.
- Feldman GB, Freiman JA. Prophylactic cesarean section at term? N Engl J Med. 1985;312(19):1264.
- Cheng YW, Snowden JM, King TL, Caughey AB. Selected perinatal outcomes associated with planned home births in the United States. Am J Obstet Gynecol. 2013;209(4):325.e1–e8.
- Wax JR, Lucas FL, Lamont M, et al. Maternal and newborn outcomes in planned home birth vs planned hospital births: A meta-analysis. Am J Obstet Gynecol. 2012;203(3):243.e1–e8.
November 2013
We need honest, respectful discussion about the risks of home birth
I object to the tone of the recent commentary by Dr. Errol Norwitz. Although I agree with the concerns he raised, I object to his “tactics.” The subject line of the email I received about this commentary (“Let’s keep home delivery where it belongs…for pizza!”) and some of the comments in the article were inflammatory and almost seem to be making fun of a topic about which many women in our country feel passionately.
As health-care providers, we are skilled at explaining risks and helping people understand how their choices and actions affect those risks. We do this daily without offering insult to patients regarding their beliefs. You don’t need to convince ObGyns that home birth is unsafe—I daresay that most of them already believe that. Awareness of the data and even some of Dr. Norwitz’s insights surely are valuable to the people who are actually pursuing and performing home births—but I feel that the subject line and closing remarks were mean-spirited and serve only to push practitioners and even patients—who need information to make informed decisions—farther away from what Dr. Norwitz argues is the safest care. No one responds well to mockery.
I practice the full scope of nurse-midwifery in a hospital-based practice with excellent physicians to support me and my patients. I do not attend home births, and I, too, have concerns about some home-birth practices and statistics. Honest, respectful communication about the facts is what is needed to improve patient safety and outcomes—not marginalization and disrespect.
Jamie A. Otremba, CNM
Waconia, Minnesota
Hospital deliveries can have problems, too
This article was insulting. When the study by Cheng and colleagues1 was first published, it received enormous criticism, so why has it been revived? Planned home birth with an appropriate provider is safe. The absolute numbers for maternal and newborn problems are very low but sound high when couched in warning language. Let’s not forget the mothers and babies who have problems in the hospital. In fact, the United States has a terrible record when it comes to maternal and neonatal morbidity and mortality—figures based almost entirely on hospital deliveries.
Low-risk women choose home birth to avoid a delivery that often results in far too many non-evidence-based interventions. Why don’t we change the system to make it work? If ObGyns would participate in a collaborative system of care that allows seamless integration of midwives into the acute-care setting when women are no longer low-risk, home-birth statistics would improve tremendously.
ObGyns are surgeons and specialists in problem-solving. Midwives are specialists in low-risk care, and they are adept at continuously assessing their patients to assure that they remain low-risk, consulting or transferring when necessary. Far too many physicians are “turf-conscious,” refusing to collaborate with midwives. It sounds to me as though the risk in home birth is iatrogenic, caused by the very physicians who claim that it is unsafe.
Chris Hilderbrandt, ARNP, CNM
Largo, Florida
Why are real changes taking so long?
I believe that the risk figures—bolded and supersized at the beginning of Dr. Norwitz’s commentary—are somewhat misleading. The intended message is clear but something very important is missing: The scores and risk values for home births were not isolated and compared between skilled midwives attending the births versus unskilled and untrained “other midwives.” In that light, when one considers the difference in risk—0.37% risk for an Apgar score of less than 4 for a home birth (which involves mainly nonprofessional labor attendants) and 0.24% for a hospitalized, medically managed birth, it is remarkable that the difference—0.13%—is so small.
I realize the importance of striving to bring any risk close to zero, but the way the data were presented unfairly implies that home births are essentially more risky than hospital births. It simply may be that they are attended by less-skilled people.
In addition, Dr. Norwitz noted that, in countries where home birth is integrated into the health-care system, there is a twofold to threefold increase in neonatal death. However, a 2011 article by van der Kooy and colleagues2 contradicts that claim, showing no significant difference in neonatal deaths between planned home birth and hospital birth for low-risk women in the Netherlands. I suppose, depending on the vantage point, there is a study somewhere that will support any of our beliefs.
Please make no mistake. I have never been a proponent of home birth. Even a small risk becomes 100% for the individual who suffers any kind of ill effects or damage. But I am a promoter of natural birth and minimal medical intervention. I abandoned obstetrics years ago when I saw firsthand that the increasing focus on technology and medical intervention moved the woman and her experience to the outer margins. The situation has not improved, and the strides made early on by the women’s movement have all but disappeared as women are aggressively convinced that technology and “controlled” interventions will grant them safe passage. In the process, they lose a sense of their autonomy and participation in one of the most important events in their lives.
At the end of the article, Dr. Norwitz leaves us with a notion that obstetric care providers need to do more in the way of providing emotional and social support. My question is… What in the world are you waiting for?
Obstetric care has a very long way to go before the focus moves meaningfully toward handing back some of the power of choice to the pregnant woman, something early feminists championed. That would require the obstetrician to honor and listen to the woman as she begins to formulate a delivery plan, to support her input at every step of decision-making, to provide her with or refer her to sources of information about delivery alternatives—the very types of alternatives that are readily available in midwife-attended births.
Jacquelynn Cunliffe, MSN, PhD
Wayne, Pennsylvania
Dr. Norwitz responds
We all want a healthy infant
Regardless of the type of training or level of experience, all obstetric care providers want the same outcome: an uncomplicated delivery of a healthy infant under conditions that are safe and supportive.
Contrary to popular belief, Nature is not a good obstetrician. It is estimated that between 1 in 50 and 1 in 500 fetuses reach maturity in utero and are subsequently involved in a catastrophic event that results in severe neurologic damage or perinatal death.3 Many of these catastrophic events occur during labor. Even a woman categorized as “low-risk” throughout her pregnancy can become “high-risk” in a matter of minutes if she develops a complication during labor such as cord prolapse or placental abruption. Risk factors for such intrapartum complications have been described, but these complications can develop in anyone at any time, even in women with no risk factors at all.
As noted in the letter from Dr. Cunliffe and the article by Cheng and colleagues,4 the level of training of the person attending the planned home birth may well affect the outcome. The less skilled the provider, the less likely he or she is to anticipate and recognize a complication and the more likely an adverse event. The existing literature on this topic should not be interpreted as a criticism of the training or skill of certified nurse midwives. Even the most skilled birth attendant is ill-prepared to deal with the potential catastrophe of an intrapartum complication during a planned home birth, given the limited resources of a home environment.
The issue of planned home birth is an emotive one. Although every effort should be made to ensure that the birthing experience is a positive one, it should not be done at the expense of safety. Ms. Hilderbrandt’s claim that “Planned home birth with an appropriate provider is safe” is not supported by the existing data. Even in countries where home births are integrated fully into the medical care system, such deliveries are associated with a twofold to threefold increase in the odds of neonatal death.5 In the United States, where no such integration exists, a planned home birth is simply dangerous, although the absolute risk of a serious adverse event is low.
If a pregnant woman ever wants to know the safest place to deliver her baby, the unequivocal answer is: in a hospital setting. The question of who is best suited to attend the birth is far less critical, so long as the person—either a physician or a certified nurse midwife—has completed an accredited training program.
WE WANT TO HEAR FROM YOU!
Drop us a line and let us know what you think about current articles, which topics you'd like to see covered in future issues, and what challenges you face in daily practice. Tell us what you think by emailing us at: [email protected]
November 2013
We need honest, respectful discussion about the risks of home birth
I object to the tone of the recent commentary by Dr. Errol Norwitz. Although I agree with the concerns he raised, I object to his “tactics.” The subject line of the email I received about this commentary (“Let’s keep home delivery where it belongs…for pizza!”) and some of the comments in the article were inflammatory and almost seem to be making fun of a topic about which many women in our country feel passionately.
As health-care providers, we are skilled at explaining risks and helping people understand how their choices and actions affect those risks. We do this daily without offering insult to patients regarding their beliefs. You don’t need to convince ObGyns that home birth is unsafe—I daresay that most of them already believe that. Awareness of the data and even some of Dr. Norwitz’s insights surely are valuable to the people who are actually pursuing and performing home births—but I feel that the subject line and closing remarks were mean-spirited and serve only to push practitioners and even patients—who need information to make informed decisions—farther away from what Dr. Norwitz argues is the safest care. No one responds well to mockery.
I practice the full scope of nurse-midwifery in a hospital-based practice with excellent physicians to support me and my patients. I do not attend home births, and I, too, have concerns about some home-birth practices and statistics. Honest, respectful communication about the facts is what is needed to improve patient safety and outcomes—not marginalization and disrespect.
Jamie A. Otremba, CNM
Waconia, Minnesota
Hospital deliveries can have problems, too
This article was insulting. When the study by Cheng and colleagues1 was first published, it received enormous criticism, so why has it been revived? Planned home birth with an appropriate provider is safe. The absolute numbers for maternal and newborn problems are very low but sound high when couched in warning language. Let’s not forget the mothers and babies who have problems in the hospital. In fact, the United States has a terrible record when it comes to maternal and neonatal morbidity and mortality—figures based almost entirely on hospital deliveries.
Low-risk women choose home birth to avoid a delivery that often results in far too many non-evidence-based interventions. Why don’t we change the system to make it work? If ObGyns would participate in a collaborative system of care that allows seamless integration of midwives into the acute-care setting when women are no longer low-risk, home-birth statistics would improve tremendously.
ObGyns are surgeons and specialists in problem-solving. Midwives are specialists in low-risk care, and they are adept at continuously assessing their patients to assure that they remain low-risk, consulting or transferring when necessary. Far too many physicians are “turf-conscious,” refusing to collaborate with midwives. It sounds to me as though the risk in home birth is iatrogenic, caused by the very physicians who claim that it is unsafe.
Chris Hilderbrandt, ARNP, CNM
Largo, Florida
Why are real changes taking so long?
I believe that the risk figures—bolded and supersized at the beginning of Dr. Norwitz’s commentary—are somewhat misleading. The intended message is clear but something very important is missing: The scores and risk values for home births were not isolated and compared between skilled midwives attending the births versus unskilled and untrained “other midwives.” In that light, when one considers the difference in risk—0.37% risk for an Apgar score of less than 4 for a home birth (which involves mainly nonprofessional labor attendants) and 0.24% for a hospitalized, medically managed birth, it is remarkable that the difference—0.13%—is so small.
I realize the importance of striving to bring any risk close to zero, but the way the data were presented unfairly implies that home births are essentially more risky than hospital births. It simply may be that they are attended by less-skilled people.
In addition, Dr. Norwitz noted that, in countries where home birth is integrated into the health-care system, there is a twofold to threefold increase in neonatal death. However, a 2011 article by van der Kooy and colleagues2 contradicts that claim, showing no significant difference in neonatal deaths between planned home birth and hospital birth for low-risk women in the Netherlands. I suppose, depending on the vantage point, there is a study somewhere that will support any of our beliefs.
Please make no mistake. I have never been a proponent of home birth. Even a small risk becomes 100% for the individual who suffers any kind of ill effects or damage. But I am a promoter of natural birth and minimal medical intervention. I abandoned obstetrics years ago when I saw firsthand that the increasing focus on technology and medical intervention moved the woman and her experience to the outer margins. The situation has not improved, and the strides made early on by the women’s movement have all but disappeared as women are aggressively convinced that technology and “controlled” interventions will grant them safe passage. In the process, they lose a sense of their autonomy and participation in one of the most important events in their lives.
At the end of the article, Dr. Norwitz leaves us with a notion that obstetric care providers need to do more in the way of providing emotional and social support. My question is… What in the world are you waiting for?
Obstetric care has a very long way to go before the focus moves meaningfully toward handing back some of the power of choice to the pregnant woman, something early feminists championed. That would require the obstetrician to honor and listen to the woman as she begins to formulate a delivery plan, to support her input at every step of decision-making, to provide her with or refer her to sources of information about delivery alternatives—the very types of alternatives that are readily available in midwife-attended births.
Jacquelynn Cunliffe, MSN, PhD
Wayne, Pennsylvania
Dr. Norwitz responds
We all want a healthy infant
Regardless of the type of training or level of experience, all obstetric care providers want the same outcome: an uncomplicated delivery of a healthy infant under conditions that are safe and supportive.
Contrary to popular belief, Nature is not a good obstetrician. It is estimated that between 1 in 50 and 1 in 500 fetuses reach maturity in utero and are subsequently involved in a catastrophic event that results in severe neurologic damage or perinatal death.3 Many of these catastrophic events occur during labor. Even a woman categorized as “low-risk” throughout her pregnancy can become “high-risk” in a matter of minutes if she develops a complication during labor such as cord prolapse or placental abruption. Risk factors for such intrapartum complications have been described, but these complications can develop in anyone at any time, even in women with no risk factors at all.
As noted in the letter from Dr. Cunliffe and the article by Cheng and colleagues,4 the level of training of the person attending the planned home birth may well affect the outcome. The less skilled the provider, the less likely he or she is to anticipate and recognize a complication and the more likely an adverse event. The existing literature on this topic should not be interpreted as a criticism of the training or skill of certified nurse midwives. Even the most skilled birth attendant is ill-prepared to deal with the potential catastrophe of an intrapartum complication during a planned home birth, given the limited resources of a home environment.
The issue of planned home birth is an emotive one. Although every effort should be made to ensure that the birthing experience is a positive one, it should not be done at the expense of safety. Ms. Hilderbrandt’s claim that “Planned home birth with an appropriate provider is safe” is not supported by the existing data. Even in countries where home births are integrated fully into the medical care system, such deliveries are associated with a twofold to threefold increase in the odds of neonatal death.5 In the United States, where no such integration exists, a planned home birth is simply dangerous, although the absolute risk of a serious adverse event is low.
If a pregnant woman ever wants to know the safest place to deliver her baby, the unequivocal answer is: in a hospital setting. The question of who is best suited to attend the birth is far less critical, so long as the person—either a physician or a certified nurse midwife—has completed an accredited training program.
WE WANT TO HEAR FROM YOU!
Drop us a line and let us know what you think about current articles, which topics you'd like to see covered in future issues, and what challenges you face in daily practice. Tell us what you think by emailing us at: [email protected]
- Cheng YW, Snowden JM, King TL, Caughey AB. Selected perinatal outcomes associated with planned home births in the United States. Am J Obstet Gynecol. 2013;209(4):325.e1–e8. doi:10.1016/j.ajog.2013.06.022.
- van der Kooy J, Poeran J, de Graaf JP, et al. Planned home births compared with planned hospital births in the Netherlands: Intrapartum and early neonatal death in low-risk pregnancies. Obstet Gynecol. 2011;118(5):1037–1046.
- Feldman GB, Freiman JA. Prophylactic cesarean section at term? N Engl J Med. 1985;312(19):1264.
- Cheng YW, Snowden JM, King TL, Caughey AB. Selected perinatal outcomes associated with planned home births in the United States. Am J Obstet Gynecol. 2013;209(4):325.e1–e8.
- Wax JR, Lucas FL, Lamont M, et al. Maternal and newborn outcomes in planned home birth vs planned hospital births: A meta-analysis. Am J Obstet Gynecol. 2012;203(3):243.e1–e8.
- Cheng YW, Snowden JM, King TL, Caughey AB. Selected perinatal outcomes associated with planned home births in the United States. Am J Obstet Gynecol. 2013;209(4):325.e1–e8. doi:10.1016/j.ajog.2013.06.022.
- van der Kooy J, Poeran J, de Graaf JP, et al. Planned home births compared with planned hospital births in the Netherlands: Intrapartum and early neonatal death in low-risk pregnancies. Obstet Gynecol. 2011;118(5):1037–1046.
- Feldman GB, Freiman JA. Prophylactic cesarean section at term? N Engl J Med. 1985;312(19):1264.
- Cheng YW, Snowden JM, King TL, Caughey AB. Selected perinatal outcomes associated with planned home births in the United States. Am J Obstet Gynecol. 2013;209(4):325.e1–e8.
- Wax JR, Lucas FL, Lamont M, et al. Maternal and newborn outcomes in planned home birth vs planned hospital births: A meta-analysis. Am J Obstet Gynecol. 2012;203(3):243.e1–e8.
Small declines in primary cesarean births show slowly reversing trend
Just over one in five live, singleton births in 2012 were delivered by cesarean section for first-time mothers, showing slight declines in the rates since 2009 among the U.S. states included in a recent report.
The overall primary cesarean delivery rate was 21.5% for the 38 states – plus the District of Columbia and New York City – that had implemented the 2003 U.S. Standard Certificate of Live Birth (revised) by, at latest, Jan. 1, 2012, according to a National Vital Statistics Report from the National Center for Health Statistics.
State-specific rates in 2012 ranged from a low of 12.5% in Utah to a high of 26.9% in Florida and Louisiana, the investigators reported Jan. 23 (Natl. Vital Stat. Rep. 2014;63:1-10).
The report did not include any data from states that used the 1989 U.S. Standard Certificate of Live Birth (unrevised) through 2012 since the 1989 certificate reports on the "method of delivery" differently than the 2003 revised certificate. The data are, therefore, not generalizable to the entire country since the births are not a random sample or representative of the national demographics, especially in terms of Hispanics’ distribution.
Instead, the report’s data are presented based on when states implemented the revised 2003 certificate, a process that occurred gradually over about a decade. Among the 19 states (excluding New York City) that had implemented the 2003 certificate by Jan. 1, 2006, the primary cesarean rate first increased from 21.9% in 2006 to 22.4% in 2009 and then dropped to 21.9% in 2012. The lower rate in 2012 resulted from declines in 11 of the 19 states since 2009 while the other eight states had no significant changes in their rates.
The 28 states plus New York City that implemented the 2003 certificate by Jan. 1, 2009, also showed a decline from 2009, when the primary cesarean rate was 22.1%, to 2012, when the rate was 21.5%. This overall decline resulted from decreases in the cesarean rates in 16 of the 29 total areas included, while the other 13 areas’ rates did not significantly change. Utah’s rate during this time dropped 15%, while Delaware, New York, New York City, North Dakota, and Oregon saw drops of 5% to 10% from 2009 to 2012.
"Although significant declines were observed for total 2006 and 2009 revised reporting area rates and for many state-specific rates between 2009 and 2012, the pace of the decline has slowed," the authors wrote. For example, only two of the 19 states using the revised certificate by 2006 saw declines from 2011 to 2012, even though 13 states from this group saw declines from 2009 to 2010.
Few changes occurred in cesarean rates based on gestational age at the state level, with the only statistically significant change across multiple states occurring among babies born at 38 weeks. Among 18 of the 29 areas that adopted the 2003 certificate by 2009, the rate for this gestational age dropped an average of 10% from 2009 to 2012.
The decline varied from a 5% drop in Michigan to an 18% drop in Utah. States seeing a drop of at least 10% in primary cesarean rates at 38 weeks for those years included Georgia, Kansas, Nebraska, New Mexico, New York, New York City, Ohio, Oregon, Utah, and Washington. The overall primary cesarean rates for all 29 areas combined did show decreases for each gestational age from 37 weeks to 41 weeks and for 42 or more weeks.
The report was funded by the National Center for Health Statistics. No disclosures were reported.
The new CDC data on cesarean section rates across the country, which show considerable variation from state to state, reflect the individualistic nature of U.S. citizens and indicate that this method of delivery is influenced by multiple factors, such as location, a milieu of ethnic backgrounds and mores, culture, and available options for childbirth. For example, rates may be high in one state because physicians are more vulnerable to liability suits and, therefore, elect to perform cesarean sections more often than vaginal deliveries.

|
|
Whereas women in other countries might defer to what their doctor may recommend or to national guidelines, the CDC data reveal that women and their physicians can and will make their own decisions about the delivery of their babies, based on geographic locale, cultural background of the patient, and perception of cesarean section vs. vaginal birth.
Fortunately, the morbidity and mortality associated with cesarean or vaginal delivery is significantly reduced today, allowing most women and their physicians, in general, the flexibility to pursue options based on preference. For ob.gyns. reviewing the CDC report, what remains important is the patient. The cesarean section rates today, which are slightly lower than the rates from several years ago, are neither too high nor too low, but reflect a cultural trend that is relevant to this time in our history. In another several years, we may see a shift in the opposite direction. It will all depend upon popular perceptions, preferences of women and their doctors, and the impact of health care clinicians on pregnancy and delivery.
Dr. E. Albert Reece is vice president for medical affairs at the University of Maryland, Baltimore, dean of the school of medicine, and the John Z. and Akiko K. Bowers Distinguished Professor in obstetrics and gynecology. He made these comments in an interview. He had no relevant financial disclosures.
The new CDC data on cesarean section rates across the country, which show considerable variation from state to state, reflect the individualistic nature of U.S. citizens and indicate that this method of delivery is influenced by multiple factors, such as location, a milieu of ethnic backgrounds and mores, culture, and available options for childbirth. For example, rates may be high in one state because physicians are more vulnerable to liability suits and, therefore, elect to perform cesarean sections more often than vaginal deliveries.

|
|
Whereas women in other countries might defer to what their doctor may recommend or to national guidelines, the CDC data reveal that women and their physicians can and will make their own decisions about the delivery of their babies, based on geographic locale, cultural background of the patient, and perception of cesarean section vs. vaginal birth.
Fortunately, the morbidity and mortality associated with cesarean or vaginal delivery is significantly reduced today, allowing most women and their physicians, in general, the flexibility to pursue options based on preference. For ob.gyns. reviewing the CDC report, what remains important is the patient. The cesarean section rates today, which are slightly lower than the rates from several years ago, are neither too high nor too low, but reflect a cultural trend that is relevant to this time in our history. In another several years, we may see a shift in the opposite direction. It will all depend upon popular perceptions, preferences of women and their doctors, and the impact of health care clinicians on pregnancy and delivery.
Dr. E. Albert Reece is vice president for medical affairs at the University of Maryland, Baltimore, dean of the school of medicine, and the John Z. and Akiko K. Bowers Distinguished Professor in obstetrics and gynecology. He made these comments in an interview. He had no relevant financial disclosures.
The new CDC data on cesarean section rates across the country, which show considerable variation from state to state, reflect the individualistic nature of U.S. citizens and indicate that this method of delivery is influenced by multiple factors, such as location, a milieu of ethnic backgrounds and mores, culture, and available options for childbirth. For example, rates may be high in one state because physicians are more vulnerable to liability suits and, therefore, elect to perform cesarean sections more often than vaginal deliveries.

|
|
Whereas women in other countries might defer to what their doctor may recommend or to national guidelines, the CDC data reveal that women and their physicians can and will make their own decisions about the delivery of their babies, based on geographic locale, cultural background of the patient, and perception of cesarean section vs. vaginal birth.
Fortunately, the morbidity and mortality associated with cesarean or vaginal delivery is significantly reduced today, allowing most women and their physicians, in general, the flexibility to pursue options based on preference. For ob.gyns. reviewing the CDC report, what remains important is the patient. The cesarean section rates today, which are slightly lower than the rates from several years ago, are neither too high nor too low, but reflect a cultural trend that is relevant to this time in our history. In another several years, we may see a shift in the opposite direction. It will all depend upon popular perceptions, preferences of women and their doctors, and the impact of health care clinicians on pregnancy and delivery.
Dr. E. Albert Reece is vice president for medical affairs at the University of Maryland, Baltimore, dean of the school of medicine, and the John Z. and Akiko K. Bowers Distinguished Professor in obstetrics and gynecology. He made these comments in an interview. He had no relevant financial disclosures.
Just over one in five live, singleton births in 2012 were delivered by cesarean section for first-time mothers, showing slight declines in the rates since 2009 among the U.S. states included in a recent report.
The overall primary cesarean delivery rate was 21.5% for the 38 states – plus the District of Columbia and New York City – that had implemented the 2003 U.S. Standard Certificate of Live Birth (revised) by, at latest, Jan. 1, 2012, according to a National Vital Statistics Report from the National Center for Health Statistics.
State-specific rates in 2012 ranged from a low of 12.5% in Utah to a high of 26.9% in Florida and Louisiana, the investigators reported Jan. 23 (Natl. Vital Stat. Rep. 2014;63:1-10).
The report did not include any data from states that used the 1989 U.S. Standard Certificate of Live Birth (unrevised) through 2012 since the 1989 certificate reports on the "method of delivery" differently than the 2003 revised certificate. The data are, therefore, not generalizable to the entire country since the births are not a random sample or representative of the national demographics, especially in terms of Hispanics’ distribution.
Instead, the report’s data are presented based on when states implemented the revised 2003 certificate, a process that occurred gradually over about a decade. Among the 19 states (excluding New York City) that had implemented the 2003 certificate by Jan. 1, 2006, the primary cesarean rate first increased from 21.9% in 2006 to 22.4% in 2009 and then dropped to 21.9% in 2012. The lower rate in 2012 resulted from declines in 11 of the 19 states since 2009 while the other eight states had no significant changes in their rates.
The 28 states plus New York City that implemented the 2003 certificate by Jan. 1, 2009, also showed a decline from 2009, when the primary cesarean rate was 22.1%, to 2012, when the rate was 21.5%. This overall decline resulted from decreases in the cesarean rates in 16 of the 29 total areas included, while the other 13 areas’ rates did not significantly change. Utah’s rate during this time dropped 15%, while Delaware, New York, New York City, North Dakota, and Oregon saw drops of 5% to 10% from 2009 to 2012.
"Although significant declines were observed for total 2006 and 2009 revised reporting area rates and for many state-specific rates between 2009 and 2012, the pace of the decline has slowed," the authors wrote. For example, only two of the 19 states using the revised certificate by 2006 saw declines from 2011 to 2012, even though 13 states from this group saw declines from 2009 to 2010.
Few changes occurred in cesarean rates based on gestational age at the state level, with the only statistically significant change across multiple states occurring among babies born at 38 weeks. Among 18 of the 29 areas that adopted the 2003 certificate by 2009, the rate for this gestational age dropped an average of 10% from 2009 to 2012.
The decline varied from a 5% drop in Michigan to an 18% drop in Utah. States seeing a drop of at least 10% in primary cesarean rates at 38 weeks for those years included Georgia, Kansas, Nebraska, New Mexico, New York, New York City, Ohio, Oregon, Utah, and Washington. The overall primary cesarean rates for all 29 areas combined did show decreases for each gestational age from 37 weeks to 41 weeks and for 42 or more weeks.
The report was funded by the National Center for Health Statistics. No disclosures were reported.
Just over one in five live, singleton births in 2012 were delivered by cesarean section for first-time mothers, showing slight declines in the rates since 2009 among the U.S. states included in a recent report.
The overall primary cesarean delivery rate was 21.5% for the 38 states – plus the District of Columbia and New York City – that had implemented the 2003 U.S. Standard Certificate of Live Birth (revised) by, at latest, Jan. 1, 2012, according to a National Vital Statistics Report from the National Center for Health Statistics.
State-specific rates in 2012 ranged from a low of 12.5% in Utah to a high of 26.9% in Florida and Louisiana, the investigators reported Jan. 23 (Natl. Vital Stat. Rep. 2014;63:1-10).
The report did not include any data from states that used the 1989 U.S. Standard Certificate of Live Birth (unrevised) through 2012 since the 1989 certificate reports on the "method of delivery" differently than the 2003 revised certificate. The data are, therefore, not generalizable to the entire country since the births are not a random sample or representative of the national demographics, especially in terms of Hispanics’ distribution.
Instead, the report’s data are presented based on when states implemented the revised 2003 certificate, a process that occurred gradually over about a decade. Among the 19 states (excluding New York City) that had implemented the 2003 certificate by Jan. 1, 2006, the primary cesarean rate first increased from 21.9% in 2006 to 22.4% in 2009 and then dropped to 21.9% in 2012. The lower rate in 2012 resulted from declines in 11 of the 19 states since 2009 while the other eight states had no significant changes in their rates.
The 28 states plus New York City that implemented the 2003 certificate by Jan. 1, 2009, also showed a decline from 2009, when the primary cesarean rate was 22.1%, to 2012, when the rate was 21.5%. This overall decline resulted from decreases in the cesarean rates in 16 of the 29 total areas included, while the other 13 areas’ rates did not significantly change. Utah’s rate during this time dropped 15%, while Delaware, New York, New York City, North Dakota, and Oregon saw drops of 5% to 10% from 2009 to 2012.
"Although significant declines were observed for total 2006 and 2009 revised reporting area rates and for many state-specific rates between 2009 and 2012, the pace of the decline has slowed," the authors wrote. For example, only two of the 19 states using the revised certificate by 2006 saw declines from 2011 to 2012, even though 13 states from this group saw declines from 2009 to 2010.
Few changes occurred in cesarean rates based on gestational age at the state level, with the only statistically significant change across multiple states occurring among babies born at 38 weeks. Among 18 of the 29 areas that adopted the 2003 certificate by 2009, the rate for this gestational age dropped an average of 10% from 2009 to 2012.
The decline varied from a 5% drop in Michigan to an 18% drop in Utah. States seeing a drop of at least 10% in primary cesarean rates at 38 weeks for those years included Georgia, Kansas, Nebraska, New Mexico, New York, New York City, Ohio, Oregon, Utah, and Washington. The overall primary cesarean rates for all 29 areas combined did show decreases for each gestational age from 37 weeks to 41 weeks and for 42 or more weeks.
The report was funded by the National Center for Health Statistics. No disclosures were reported.
FROM NATIONAL VITAL STATISTICS REPORTS
Major finding: The primary singleton cesarean delivery rate for 38 states, the District of Columbia, and New York City was 21.5% in 2012, with a decline among 19 of those states from 22.4% in 2009 to 21.9% in 2012 and among 28 of those states plus New York City from 22.1% in 2009 to 21.5% in 2012.
Data source: The findings are based on 100% of singleton births for 2006-2012 among the 19 states that implemented the 2003 U.S. Standard Certificate of Live Birth by Jan. 1, 2006; for 2009-2012 among the 28 states plus New York City, that implemented the 2003 certificate by Jan. 1, 2009; and for 2012 among the additional states that implemented the certificate by Jan. 1, 2012.
Disclosures: The report was funded by the National Center for Health Statistics. No disclosures were reported.
Antidotes, detoxification agents, and pregnancy
By their very nature, antidotes and detoxification agents are needed in situations where the health and well-being of the mother are in jeopardy. In nearly all such cases, the mother’s condition will take priority over the safety of the embryo-fetus. Only two of the drugs (ethanol and penicillamine) are known to cause embryo or fetal harm but, for most of these drugs, the reported human pregnancy experience is very limited or absent. Nevertheless, pregnant women should be treated the same way as nonpregnant women.
Activated charcoal prevents absorption of substances from the gut and is no risk to the mother or her pregnancy. Similarly, ipecac syrup, which is used to induce vomiting, is safe in pregnancy.
Several agents are available for the reversal of opioid (natural or synthetic) overdose that is causing respiratory depression and/or marked sedation: naloxone, naltrexone, and nalmefene, a long-acting derivative of naltrexone (plasma half-life about 10 hours). Of the three agents, naloxone is the one for which there is the most human pregnancy experience. It has no intrinsic respiratory depressive activity or other narcotic effects of its own. All of these agents can be used in pregnancy for acute narcotic overdose.
Acetylcysteine is used to prevent or lessen hepatic injury following the ingestion of potentially hepatic toxic doses of acetaminophen. The antidote is not teratogenic or embryo toxic, and limited human pregnancy data have not shown fetal toxicity. After IV administration, acetylcysteine crosses the placenta in sufficient amounts to achieve protective serum levels in the fetus.
Potentially life-threatening digoxin overdose can be treated with IV digoxin immune Fab (ovine). The use of the agent has been reported in 44 pregnancies, but none of the cases involved digitalis overdose (all women had severe preeclampsia). No fetal harm secondary to the drug was observed.
Flumazenil is indicated for the reversal of benzodiazepine overdose. The drug is not teratogenic or embryo-fetal toxic in animals at systemic exposures near those obtained in humans. Based on very limited data, it appears to cross the human placenta and to reverse the depressive effects of benzodiazepines on the fetus.
Fomepizole is used for the treatment of ethylene glycol or methanol ingestion. It inhibits alcohol dehydrogenase, an enzyme that catalyzes the oxidation of the two chemicals to their toxic metabolites. The drug was not teratogenic in mice, but only one case of human pregnancy exposure has been reported, and the pregnancy outcome was unknown. Ethanol also has been used for poisonings with these two chemicals. Although the fetal effects of this short-term (24-48 hours) use have not been studied, neurotoxicity is a potential complication.
Glucarpidase is indicated for the treatment of toxic plasma methotrexate levels. It converts methotrexate to inactive metabolites. There are no reports of its use in human or animal pregnancies. Human reports are unlikely because methotrexate is contraindicated in pregnancy.
There are six agents available to treat heavy metal (arsenic, gold, iron, lead, and mercury) intoxication: deferasirox (iron), deferoxamine (iron), dimercaprol (arsenic, gold, lead, and mercury), edetate calcium disodium (lead), penicillamine (copper and mercury), and succimer (lead).
Deferasirox is indicated for chronic iron overload due to blood transfusions. Three reports have described its use in the first half of pregnancy without embryo or fetal harm. Deferoxamine is used for the treatment of both acute and chronic iron overload. Although the drug causes toxicity in two animal species, the human pregnancy experience is substantial, and no embryo or fetal adverse effects attributable to the agent have been reported. Dimercaprol (British anti-Lewisite; BAL) is used for the treatment of arsenic, gold, and acute mercury poisoning (not effective for chronic mercury poisoning). It is also combined with edetate calcium disodium for lead poisoning. High doses are embryotoxic and teratogenic in mice. The published human pregnancy experience is limited and all involved exposures after the first trimester. High levels of arsenic or lead were found in the newborns in two cases.
Edetate calcium disodium forms stable chelates with a number of metals, but it is primarily used for lead overdose, either alone or in combination with dimercaprol. There are only a few reports of its use in human pregnancy, all occurring late in gestation. A potential complication of therapy is maternal hypotension that could jeopardize placental perfusion. The agent also chelates zinc, resulting in zinc deficiency. This mechanism was thought to be involved in the teratogenic effects seen in animals.
Penicillamine has been used in mercury poisoning (one report), in addition to its indication as a chelating agent for copper in the treatment of Wilson’s disease. Exposure in the first trimester is related to a risk of connective tissue anomalies, primarily cutis laxa. Succimer (dimercaptosuccinic acid; DMSA) has been used for lead, arsenic, mercury, and cadmium poisoning. It also chelates zinc quite effectively. The agent is toxic and/or teratogenic in mice and rats, but some of the effects may have been secondary to zinc deficiency. Because of the complete absence of human pregnancy experience, antidotes other than succimer probably are preferable.
Lanthanum carbonate and sevelamer are indicated to reduce serum phosphate levels in patients with end-stage renal disease. The drugs bind dietary phosphate from food during digestion in the gut. There are no reports of their use in human pregnancy. The systemic bioavailability is minimal, and the drugs should have no effect on the embryo or fetus. However, they may prevent intestinal vitamin absorption, especially of fat-soluble vitamins.
The cholinergic agent physostigmine is capable of reversing the central nervous system effects of anticholinergics, such as scopolamine and tricyclic antidepressants. The reported human pregnancy experience is limited to the third trimester.
Methylene blue has been used for cyanide poisoning. In humans, it is teratogenic and fetal toxic when given by intra-amniotic injection, but its oral use as an antidote in pregnancy has not been reported. The cyanide antidote package contains amyl nitrite, sodium nitrite, and sodium thiosulfate. The effects of these agents on human pregnancy also are unknown, as are the effects of high-dose hydroxocobalamin, an analogue of vitamin B12 also used in cyanide poisoning.
Pralidoxime (2-PAM) reactivates cholinesterase that has been inactivated by organophosphate pesticides and chemicals with anticholinesterase activity, thereby relieving the paralysis of the muscles of respiration. The drug is available in an autoinjector that can be used rapidly in cases of exposure to nerve agents possessing anticholinesterase activity (organophosphate poisoning). Animal reproduction tests have not been conducted with pralidoxime, and the human pregnancy experience is limited to a few cases of insecticide poisoning (second and third trimesters). Healthy infants were later delivered in these cases.
Four antivenins are commercially available for acute envenomation: black widow spider antivenin, Centruroides (scorpion) immune F(ab\')2 (equine), crotalidae polyvalent immune Fab (ovine) (North American rattlesnake), and North American coral snake antivenin (equine). In addition, botulism antitoxin heptavalent (equine) is used for food poisoning caused by the neurotoxic bacterium Clostridium botulinum. Animal reproduction studies have not been conducted with these products, and human reports are limited or absent.
Sapropterin, a cofactor for the enzyme phenylalanine hydroxylase, reduces blood phenylalanine levels in patients with phenylketonuria. The drug is given daily if diet alone does not control maternal phenylalanine levels. Use of the drug in human pregnancy has not been reported.
A number of other agents can be classified as antidotes, in addition to their primary indications, because they can reverse the toxic effects of other agents. These antidotes include atropine (severe bradycardia, poisonings with organophosphates and carbamates), calcium chloride or gluconate (severe hypocalcemia, calcium-channel-blocker overdose, exposure to hydrofluoric acid), glucagon (hypoglycemia), folinic acid (methotrexate overdose), protamine (heparin overdose), pyridoxine (isoniazid-induced seizures; adjunct in ethylene glycol poisoning), and vitamin K (phytonadione) (warfarin overdose). The pregnancy data are extensive for many of these agents and are not suggestive of significant embryo or fetal risk.
Mr. Briggs is a pharmacist clinical specialist at the outpatient clinics of Memorial Care Center for Women at Miller Children’s Hospital in Long Beach, Calif.; clinical professor of pharmacy at the University of California, San Francisco; and adjunct professor of pharmacy at the University of Southern California, Los Angeles, and Washington State University, Spokane. He also is coauthor of "Drugs in Pregnancy and Lactation," and coeditor of "Diseases, Complications, and Drug Therapy in Obstetrics." He had no relevant financial disclosures. Contact him at [email protected].
By their very nature, antidotes and detoxification agents are needed in situations where the health and well-being of the mother are in jeopardy. In nearly all such cases, the mother’s condition will take priority over the safety of the embryo-fetus. Only two of the drugs (ethanol and penicillamine) are known to cause embryo or fetal harm but, for most of these drugs, the reported human pregnancy experience is very limited or absent. Nevertheless, pregnant women should be treated the same way as nonpregnant women.
Activated charcoal prevents absorption of substances from the gut and is no risk to the mother or her pregnancy. Similarly, ipecac syrup, which is used to induce vomiting, is safe in pregnancy.
Several agents are available for the reversal of opioid (natural or synthetic) overdose that is causing respiratory depression and/or marked sedation: naloxone, naltrexone, and nalmefene, a long-acting derivative of naltrexone (plasma half-life about 10 hours). Of the three agents, naloxone is the one for which there is the most human pregnancy experience. It has no intrinsic respiratory depressive activity or other narcotic effects of its own. All of these agents can be used in pregnancy for acute narcotic overdose.
Acetylcysteine is used to prevent or lessen hepatic injury following the ingestion of potentially hepatic toxic doses of acetaminophen. The antidote is not teratogenic or embryo toxic, and limited human pregnancy data have not shown fetal toxicity. After IV administration, acetylcysteine crosses the placenta in sufficient amounts to achieve protective serum levels in the fetus.
Potentially life-threatening digoxin overdose can be treated with IV digoxin immune Fab (ovine). The use of the agent has been reported in 44 pregnancies, but none of the cases involved digitalis overdose (all women had severe preeclampsia). No fetal harm secondary to the drug was observed.
Flumazenil is indicated for the reversal of benzodiazepine overdose. The drug is not teratogenic or embryo-fetal toxic in animals at systemic exposures near those obtained in humans. Based on very limited data, it appears to cross the human placenta and to reverse the depressive effects of benzodiazepines on the fetus.
Fomepizole is used for the treatment of ethylene glycol or methanol ingestion. It inhibits alcohol dehydrogenase, an enzyme that catalyzes the oxidation of the two chemicals to their toxic metabolites. The drug was not teratogenic in mice, but only one case of human pregnancy exposure has been reported, and the pregnancy outcome was unknown. Ethanol also has been used for poisonings with these two chemicals. Although the fetal effects of this short-term (24-48 hours) use have not been studied, neurotoxicity is a potential complication.
Glucarpidase is indicated for the treatment of toxic plasma methotrexate levels. It converts methotrexate to inactive metabolites. There are no reports of its use in human or animal pregnancies. Human reports are unlikely because methotrexate is contraindicated in pregnancy.
There are six agents available to treat heavy metal (arsenic, gold, iron, lead, and mercury) intoxication: deferasirox (iron), deferoxamine (iron), dimercaprol (arsenic, gold, lead, and mercury), edetate calcium disodium (lead), penicillamine (copper and mercury), and succimer (lead).
Deferasirox is indicated for chronic iron overload due to blood transfusions. Three reports have described its use in the first half of pregnancy without embryo or fetal harm. Deferoxamine is used for the treatment of both acute and chronic iron overload. Although the drug causes toxicity in two animal species, the human pregnancy experience is substantial, and no embryo or fetal adverse effects attributable to the agent have been reported. Dimercaprol (British anti-Lewisite; BAL) is used for the treatment of arsenic, gold, and acute mercury poisoning (not effective for chronic mercury poisoning). It is also combined with edetate calcium disodium for lead poisoning. High doses are embryotoxic and teratogenic in mice. The published human pregnancy experience is limited and all involved exposures after the first trimester. High levels of arsenic or lead were found in the newborns in two cases.
Edetate calcium disodium forms stable chelates with a number of metals, but it is primarily used for lead overdose, either alone or in combination with dimercaprol. There are only a few reports of its use in human pregnancy, all occurring late in gestation. A potential complication of therapy is maternal hypotension that could jeopardize placental perfusion. The agent also chelates zinc, resulting in zinc deficiency. This mechanism was thought to be involved in the teratogenic effects seen in animals.
Penicillamine has been used in mercury poisoning (one report), in addition to its indication as a chelating agent for copper in the treatment of Wilson’s disease. Exposure in the first trimester is related to a risk of connective tissue anomalies, primarily cutis laxa. Succimer (dimercaptosuccinic acid; DMSA) has been used for lead, arsenic, mercury, and cadmium poisoning. It also chelates zinc quite effectively. The agent is toxic and/or teratogenic in mice and rats, but some of the effects may have been secondary to zinc deficiency. Because of the complete absence of human pregnancy experience, antidotes other than succimer probably are preferable.
Lanthanum carbonate and sevelamer are indicated to reduce serum phosphate levels in patients with end-stage renal disease. The drugs bind dietary phosphate from food during digestion in the gut. There are no reports of their use in human pregnancy. The systemic bioavailability is minimal, and the drugs should have no effect on the embryo or fetus. However, they may prevent intestinal vitamin absorption, especially of fat-soluble vitamins.
The cholinergic agent physostigmine is capable of reversing the central nervous system effects of anticholinergics, such as scopolamine and tricyclic antidepressants. The reported human pregnancy experience is limited to the third trimester.
Methylene blue has been used for cyanide poisoning. In humans, it is teratogenic and fetal toxic when given by intra-amniotic injection, but its oral use as an antidote in pregnancy has not been reported. The cyanide antidote package contains amyl nitrite, sodium nitrite, and sodium thiosulfate. The effects of these agents on human pregnancy also are unknown, as are the effects of high-dose hydroxocobalamin, an analogue of vitamin B12 also used in cyanide poisoning.
Pralidoxime (2-PAM) reactivates cholinesterase that has been inactivated by organophosphate pesticides and chemicals with anticholinesterase activity, thereby relieving the paralysis of the muscles of respiration. The drug is available in an autoinjector that can be used rapidly in cases of exposure to nerve agents possessing anticholinesterase activity (organophosphate poisoning). Animal reproduction tests have not been conducted with pralidoxime, and the human pregnancy experience is limited to a few cases of insecticide poisoning (second and third trimesters). Healthy infants were later delivered in these cases.
Four antivenins are commercially available for acute envenomation: black widow spider antivenin, Centruroides (scorpion) immune F(ab\')2 (equine), crotalidae polyvalent immune Fab (ovine) (North American rattlesnake), and North American coral snake antivenin (equine). In addition, botulism antitoxin heptavalent (equine) is used for food poisoning caused by the neurotoxic bacterium Clostridium botulinum. Animal reproduction studies have not been conducted with these products, and human reports are limited or absent.
Sapropterin, a cofactor for the enzyme phenylalanine hydroxylase, reduces blood phenylalanine levels in patients with phenylketonuria. The drug is given daily if diet alone does not control maternal phenylalanine levels. Use of the drug in human pregnancy has not been reported.
A number of other agents can be classified as antidotes, in addition to their primary indications, because they can reverse the toxic effects of other agents. These antidotes include atropine (severe bradycardia, poisonings with organophosphates and carbamates), calcium chloride or gluconate (severe hypocalcemia, calcium-channel-blocker overdose, exposure to hydrofluoric acid), glucagon (hypoglycemia), folinic acid (methotrexate overdose), protamine (heparin overdose), pyridoxine (isoniazid-induced seizures; adjunct in ethylene glycol poisoning), and vitamin K (phytonadione) (warfarin overdose). The pregnancy data are extensive for many of these agents and are not suggestive of significant embryo or fetal risk.
Mr. Briggs is a pharmacist clinical specialist at the outpatient clinics of Memorial Care Center for Women at Miller Children’s Hospital in Long Beach, Calif.; clinical professor of pharmacy at the University of California, San Francisco; and adjunct professor of pharmacy at the University of Southern California, Los Angeles, and Washington State University, Spokane. He also is coauthor of "Drugs in Pregnancy and Lactation," and coeditor of "Diseases, Complications, and Drug Therapy in Obstetrics." He had no relevant financial disclosures. Contact him at [email protected].
By their very nature, antidotes and detoxification agents are needed in situations where the health and well-being of the mother are in jeopardy. In nearly all such cases, the mother’s condition will take priority over the safety of the embryo-fetus. Only two of the drugs (ethanol and penicillamine) are known to cause embryo or fetal harm but, for most of these drugs, the reported human pregnancy experience is very limited or absent. Nevertheless, pregnant women should be treated the same way as nonpregnant women.
Activated charcoal prevents absorption of substances from the gut and is no risk to the mother or her pregnancy. Similarly, ipecac syrup, which is used to induce vomiting, is safe in pregnancy.
Several agents are available for the reversal of opioid (natural or synthetic) overdose that is causing respiratory depression and/or marked sedation: naloxone, naltrexone, and nalmefene, a long-acting derivative of naltrexone (plasma half-life about 10 hours). Of the three agents, naloxone is the one for which there is the most human pregnancy experience. It has no intrinsic respiratory depressive activity or other narcotic effects of its own. All of these agents can be used in pregnancy for acute narcotic overdose.
Acetylcysteine is used to prevent or lessen hepatic injury following the ingestion of potentially hepatic toxic doses of acetaminophen. The antidote is not teratogenic or embryo toxic, and limited human pregnancy data have not shown fetal toxicity. After IV administration, acetylcysteine crosses the placenta in sufficient amounts to achieve protective serum levels in the fetus.
Potentially life-threatening digoxin overdose can be treated with IV digoxin immune Fab (ovine). The use of the agent has been reported in 44 pregnancies, but none of the cases involved digitalis overdose (all women had severe preeclampsia). No fetal harm secondary to the drug was observed.
Flumazenil is indicated for the reversal of benzodiazepine overdose. The drug is not teratogenic or embryo-fetal toxic in animals at systemic exposures near those obtained in humans. Based on very limited data, it appears to cross the human placenta and to reverse the depressive effects of benzodiazepines on the fetus.
Fomepizole is used for the treatment of ethylene glycol or methanol ingestion. It inhibits alcohol dehydrogenase, an enzyme that catalyzes the oxidation of the two chemicals to their toxic metabolites. The drug was not teratogenic in mice, but only one case of human pregnancy exposure has been reported, and the pregnancy outcome was unknown. Ethanol also has been used for poisonings with these two chemicals. Although the fetal effects of this short-term (24-48 hours) use have not been studied, neurotoxicity is a potential complication.
Glucarpidase is indicated for the treatment of toxic plasma methotrexate levels. It converts methotrexate to inactive metabolites. There are no reports of its use in human or animal pregnancies. Human reports are unlikely because methotrexate is contraindicated in pregnancy.
There are six agents available to treat heavy metal (arsenic, gold, iron, lead, and mercury) intoxication: deferasirox (iron), deferoxamine (iron), dimercaprol (arsenic, gold, lead, and mercury), edetate calcium disodium (lead), penicillamine (copper and mercury), and succimer (lead).
Deferasirox is indicated for chronic iron overload due to blood transfusions. Three reports have described its use in the first half of pregnancy without embryo or fetal harm. Deferoxamine is used for the treatment of both acute and chronic iron overload. Although the drug causes toxicity in two animal species, the human pregnancy experience is substantial, and no embryo or fetal adverse effects attributable to the agent have been reported. Dimercaprol (British anti-Lewisite; BAL) is used for the treatment of arsenic, gold, and acute mercury poisoning (not effective for chronic mercury poisoning). It is also combined with edetate calcium disodium for lead poisoning. High doses are embryotoxic and teratogenic in mice. The published human pregnancy experience is limited and all involved exposures after the first trimester. High levels of arsenic or lead were found in the newborns in two cases.
Edetate calcium disodium forms stable chelates with a number of metals, but it is primarily used for lead overdose, either alone or in combination with dimercaprol. There are only a few reports of its use in human pregnancy, all occurring late in gestation. A potential complication of therapy is maternal hypotension that could jeopardize placental perfusion. The agent also chelates zinc, resulting in zinc deficiency. This mechanism was thought to be involved in the teratogenic effects seen in animals.
Penicillamine has been used in mercury poisoning (one report), in addition to its indication as a chelating agent for copper in the treatment of Wilson’s disease. Exposure in the first trimester is related to a risk of connective tissue anomalies, primarily cutis laxa. Succimer (dimercaptosuccinic acid; DMSA) has been used for lead, arsenic, mercury, and cadmium poisoning. It also chelates zinc quite effectively. The agent is toxic and/or teratogenic in mice and rats, but some of the effects may have been secondary to zinc deficiency. Because of the complete absence of human pregnancy experience, antidotes other than succimer probably are preferable.
Lanthanum carbonate and sevelamer are indicated to reduce serum phosphate levels in patients with end-stage renal disease. The drugs bind dietary phosphate from food during digestion in the gut. There are no reports of their use in human pregnancy. The systemic bioavailability is minimal, and the drugs should have no effect on the embryo or fetus. However, they may prevent intestinal vitamin absorption, especially of fat-soluble vitamins.
The cholinergic agent physostigmine is capable of reversing the central nervous system effects of anticholinergics, such as scopolamine and tricyclic antidepressants. The reported human pregnancy experience is limited to the third trimester.
Methylene blue has been used for cyanide poisoning. In humans, it is teratogenic and fetal toxic when given by intra-amniotic injection, but its oral use as an antidote in pregnancy has not been reported. The cyanide antidote package contains amyl nitrite, sodium nitrite, and sodium thiosulfate. The effects of these agents on human pregnancy also are unknown, as are the effects of high-dose hydroxocobalamin, an analogue of vitamin B12 also used in cyanide poisoning.
Pralidoxime (2-PAM) reactivates cholinesterase that has been inactivated by organophosphate pesticides and chemicals with anticholinesterase activity, thereby relieving the paralysis of the muscles of respiration. The drug is available in an autoinjector that can be used rapidly in cases of exposure to nerve agents possessing anticholinesterase activity (organophosphate poisoning). Animal reproduction tests have not been conducted with pralidoxime, and the human pregnancy experience is limited to a few cases of insecticide poisoning (second and third trimesters). Healthy infants were later delivered in these cases.
Four antivenins are commercially available for acute envenomation: black widow spider antivenin, Centruroides (scorpion) immune F(ab\')2 (equine), crotalidae polyvalent immune Fab (ovine) (North American rattlesnake), and North American coral snake antivenin (equine). In addition, botulism antitoxin heptavalent (equine) is used for food poisoning caused by the neurotoxic bacterium Clostridium botulinum. Animal reproduction studies have not been conducted with these products, and human reports are limited or absent.
Sapropterin, a cofactor for the enzyme phenylalanine hydroxylase, reduces blood phenylalanine levels in patients with phenylketonuria. The drug is given daily if diet alone does not control maternal phenylalanine levels. Use of the drug in human pregnancy has not been reported.
A number of other agents can be classified as antidotes, in addition to their primary indications, because they can reverse the toxic effects of other agents. These antidotes include atropine (severe bradycardia, poisonings with organophosphates and carbamates), calcium chloride or gluconate (severe hypocalcemia, calcium-channel-blocker overdose, exposure to hydrofluoric acid), glucagon (hypoglycemia), folinic acid (methotrexate overdose), protamine (heparin overdose), pyridoxine (isoniazid-induced seizures; adjunct in ethylene glycol poisoning), and vitamin K (phytonadione) (warfarin overdose). The pregnancy data are extensive for many of these agents and are not suggestive of significant embryo or fetal risk.
Mr. Briggs is a pharmacist clinical specialist at the outpatient clinics of Memorial Care Center for Women at Miller Children’s Hospital in Long Beach, Calif.; clinical professor of pharmacy at the University of California, San Francisco; and adjunct professor of pharmacy at the University of Southern California, Los Angeles, and Washington State University, Spokane. He also is coauthor of "Drugs in Pregnancy and Lactation," and coeditor of "Diseases, Complications, and Drug Therapy in Obstetrics." He had no relevant financial disclosures. Contact him at [email protected].
Excessive GBS sepsis evaluations of neonates curbed
Excessive evaluations of well-appearing neonates declined markedly, as intended, when the most recent (2010) CDC guidelines for assessing early-onset group B streptococcal sepsis were implemented, according to a report from one hospital published online Jan. 20 in Pediatrics.
This decrease was accompanied by a marked drop in health care costs and was not associated with any apparent harmful effects, indicating that the evaluations that were avoided were indeed unnecessary, said Dr. Sagori Mukhopadhyay of Boston Children’s Hospital and Harvard Medical School and her associates.
The guidelines from the Centers for Disease Control and Prevention (CDC) were revised so that healthy-appearing infants born to mothers who had inadequate intrapartum antibiotic prophylaxis for group B streptococcus would only be evaluated for early-onset GBS sepsis if they had additional indications: gestational age of less than 37 weeks, rupture of membranes that preceded delivery by 18 hours or longer, maternal fever, or chorioamnionitis. Preliminary studies indicated that implementing these more restrictive guidelines would decrease excessive evaluations of healthy newborns by approximately 25%, "but the real-life impact of changes in medical policy is rarely straightforward," the investigators said.
There was concern that some well-appearing infants who weren’t closely evaluated might go on to develop early-onset GBS sepsis that could have been prevented. So Dr. Mukhopadhyay and her colleagues performed a retrospective cohort study involving 14,286 live births to assess the effect of implementing the new guidelines.
They compared infants born at Brigham and Women’s Hospital, Boston, during two time periods: 7,282 born the year before the new guidelines were adopted (spring 2009–spring 2010), and 7,004 born the year afterward (spring 2011–spring 2012).
Typical evaluations for early-onset GBS sepsis involved immediate transfer to the neonatal intensive care unit, measurement of vital signs, blood sampling, peripheral IV catheter insertion, empiric antibiotic therapy, and close observation for several hours.
The number of such evaluations declined substantially after implementation of the new guidelines, from 920 to 476. This represents a rate decrease from 126 per 1,000 live births to 68 per 1,000 live births, the investigators reported (Pediatrics 2014;33:196-203).
The number of infants given empiric antibiotics also declined, from 527 to 365.
There was no increase in the number of infants who developed early-onset GBS sepsis between the two study periods. The overall rate of culture-confirmed infection was 0.25 per 1,000 live births in 2009-2010, which was not significantly different from the rate of 0.39 per 1,000 in 2011-2012.
Costs of care dramatically decreased along with the decline in "unnecessary" newborn evaluations. "The resources expended over the study periods among asymptomatic infants at risk for early-onset sepsis were considerable: more than 2,200 nursing hours were required to evaluate 1,396 infants, including 890 uninfected infants treated with antibiotics, at an estimated cost of nearly $400,000. These efforts detected only 2 early-onset sepsis cases," Dr. Mukhopadhyay and her associates said.
They added that the American Academy of Pediatrics’ Committee on the Fetus and Newborn recently published its own guidelines for early-onset sepsis evaluation and prevention. Because these AAP guidelines differ somewhat from the CDC guidelines, implementing them will likely have a somewhat different effect on the frequency and costs of such evaluations, the investigators said.
This study was supported by the National Institutes of Health, the Harvard Catalyst/Harvard Clinical and Translational Science Center, the National Center for Research Resources, the National Center for Advancing Translational Sciences, and Harvard University. No financial conflicts of interest were reported.
Excessive evaluations of well-appearing neonates declined markedly, as intended, when the most recent (2010) CDC guidelines for assessing early-onset group B streptococcal sepsis were implemented, according to a report from one hospital published online Jan. 20 in Pediatrics.
This decrease was accompanied by a marked drop in health care costs and was not associated with any apparent harmful effects, indicating that the evaluations that were avoided were indeed unnecessary, said Dr. Sagori Mukhopadhyay of Boston Children’s Hospital and Harvard Medical School and her associates.
The guidelines from the Centers for Disease Control and Prevention (CDC) were revised so that healthy-appearing infants born to mothers who had inadequate intrapartum antibiotic prophylaxis for group B streptococcus would only be evaluated for early-onset GBS sepsis if they had additional indications: gestational age of less than 37 weeks, rupture of membranes that preceded delivery by 18 hours or longer, maternal fever, or chorioamnionitis. Preliminary studies indicated that implementing these more restrictive guidelines would decrease excessive evaluations of healthy newborns by approximately 25%, "but the real-life impact of changes in medical policy is rarely straightforward," the investigators said.
There was concern that some well-appearing infants who weren’t closely evaluated might go on to develop early-onset GBS sepsis that could have been prevented. So Dr. Mukhopadhyay and her colleagues performed a retrospective cohort study involving 14,286 live births to assess the effect of implementing the new guidelines.
They compared infants born at Brigham and Women’s Hospital, Boston, during two time periods: 7,282 born the year before the new guidelines were adopted (spring 2009–spring 2010), and 7,004 born the year afterward (spring 2011–spring 2012).
Typical evaluations for early-onset GBS sepsis involved immediate transfer to the neonatal intensive care unit, measurement of vital signs, blood sampling, peripheral IV catheter insertion, empiric antibiotic therapy, and close observation for several hours.
The number of such evaluations declined substantially after implementation of the new guidelines, from 920 to 476. This represents a rate decrease from 126 per 1,000 live births to 68 per 1,000 live births, the investigators reported (Pediatrics 2014;33:196-203).
The number of infants given empiric antibiotics also declined, from 527 to 365.
There was no increase in the number of infants who developed early-onset GBS sepsis between the two study periods. The overall rate of culture-confirmed infection was 0.25 per 1,000 live births in 2009-2010, which was not significantly different from the rate of 0.39 per 1,000 in 2011-2012.
Costs of care dramatically decreased along with the decline in "unnecessary" newborn evaluations. "The resources expended over the study periods among asymptomatic infants at risk for early-onset sepsis were considerable: more than 2,200 nursing hours were required to evaluate 1,396 infants, including 890 uninfected infants treated with antibiotics, at an estimated cost of nearly $400,000. These efforts detected only 2 early-onset sepsis cases," Dr. Mukhopadhyay and her associates said.
They added that the American Academy of Pediatrics’ Committee on the Fetus and Newborn recently published its own guidelines for early-onset sepsis evaluation and prevention. Because these AAP guidelines differ somewhat from the CDC guidelines, implementing them will likely have a somewhat different effect on the frequency and costs of such evaluations, the investigators said.
This study was supported by the National Institutes of Health, the Harvard Catalyst/Harvard Clinical and Translational Science Center, the National Center for Research Resources, the National Center for Advancing Translational Sciences, and Harvard University. No financial conflicts of interest were reported.
Excessive evaluations of well-appearing neonates declined markedly, as intended, when the most recent (2010) CDC guidelines for assessing early-onset group B streptococcal sepsis were implemented, according to a report from one hospital published online Jan. 20 in Pediatrics.
This decrease was accompanied by a marked drop in health care costs and was not associated with any apparent harmful effects, indicating that the evaluations that were avoided were indeed unnecessary, said Dr. Sagori Mukhopadhyay of Boston Children’s Hospital and Harvard Medical School and her associates.
The guidelines from the Centers for Disease Control and Prevention (CDC) were revised so that healthy-appearing infants born to mothers who had inadequate intrapartum antibiotic prophylaxis for group B streptococcus would only be evaluated for early-onset GBS sepsis if they had additional indications: gestational age of less than 37 weeks, rupture of membranes that preceded delivery by 18 hours or longer, maternal fever, or chorioamnionitis. Preliminary studies indicated that implementing these more restrictive guidelines would decrease excessive evaluations of healthy newborns by approximately 25%, "but the real-life impact of changes in medical policy is rarely straightforward," the investigators said.
There was concern that some well-appearing infants who weren’t closely evaluated might go on to develop early-onset GBS sepsis that could have been prevented. So Dr. Mukhopadhyay and her colleagues performed a retrospective cohort study involving 14,286 live births to assess the effect of implementing the new guidelines.
They compared infants born at Brigham and Women’s Hospital, Boston, during two time periods: 7,282 born the year before the new guidelines were adopted (spring 2009–spring 2010), and 7,004 born the year afterward (spring 2011–spring 2012).
Typical evaluations for early-onset GBS sepsis involved immediate transfer to the neonatal intensive care unit, measurement of vital signs, blood sampling, peripheral IV catheter insertion, empiric antibiotic therapy, and close observation for several hours.
The number of such evaluations declined substantially after implementation of the new guidelines, from 920 to 476. This represents a rate decrease from 126 per 1,000 live births to 68 per 1,000 live births, the investigators reported (Pediatrics 2014;33:196-203).
The number of infants given empiric antibiotics also declined, from 527 to 365.
There was no increase in the number of infants who developed early-onset GBS sepsis between the two study periods. The overall rate of culture-confirmed infection was 0.25 per 1,000 live births in 2009-2010, which was not significantly different from the rate of 0.39 per 1,000 in 2011-2012.
Costs of care dramatically decreased along with the decline in "unnecessary" newborn evaluations. "The resources expended over the study periods among asymptomatic infants at risk for early-onset sepsis were considerable: more than 2,200 nursing hours were required to evaluate 1,396 infants, including 890 uninfected infants treated with antibiotics, at an estimated cost of nearly $400,000. These efforts detected only 2 early-onset sepsis cases," Dr. Mukhopadhyay and her associates said.
They added that the American Academy of Pediatrics’ Committee on the Fetus and Newborn recently published its own guidelines for early-onset sepsis evaluation and prevention. Because these AAP guidelines differ somewhat from the CDC guidelines, implementing them will likely have a somewhat different effect on the frequency and costs of such evaluations, the investigators said.
This study was supported by the National Institutes of Health, the Harvard Catalyst/Harvard Clinical and Translational Science Center, the National Center for Research Resources, the National Center for Advancing Translational Sciences, and Harvard University. No financial conflicts of interest were reported.
FROM PEDIATRICS
Major finding: The number of evaluations of well-appearing neonates for early-onset GBS sepsis declined substantially after implementation of new CDC guidelines, from 920 to 476.
Data source: A retrospective cohort study comparing the outcomes of 7,282 live births that occurred before new CDC guidelines on evaluating neonates for GBS sepsis were implemented with 7,004 that occurred afterward.
Disclosures: This study was supported by the National Institutes of Health, the Harvard Catalyst/Harvard Clinical and Translational Science Center, the National Center for Research Resources, the National Center for Advancing Translational Sciences, and Harvard University. No financial conflicts of interest were reported.
FDA clears assay to help diagnose developmental delay, intellectual disabilities
The Food and Drug Administration authorized for marketing a postnatal diagnostic test that helps detect genetic causes of developmental delay or intellectual disabilities in children, but it is not intended to be used as a stand-alone test, the agency announced on Jan 17.
With a blood sample, the CytoScan Dx Assay analyzes the entire genome and detects chromosomal variations in regions of the genome that are associated with intellectual and developmental disabilities, such as DiGeorge syndrome, according to the FDA statement. The statement emphasized that is should not be used as a stand-alone diagnostic test, or for preimplantation or prenatal testing or screening; population screening; "or for the detection of, or screening for acquired or genetic aberrations occurring after birth, such as cancer."
Moreover, test results "should only be used in conjunction with other clinical and diagnostic findings, consistent with professional standards of practice, including confirmation by alternative methods, evaluation of parental samples, clinical genetic evaluation, and counseling as appropriate," the statement said. Results should only be interpreted by health care professionals who are board certified in clinical cytogenetics or molecular genetics.
Evaluation of the test included a comparison of results of almost 1,000 blood samples, which found the test was better at detecting certain chromosomal abnormalities than karyotyping and fluorescence in situ hybridization (FISH) chromosomal tests.
"This new tool may help in the identification of possible causes of a child’s developmental delay or intellectual disability, allowing health care providers and parents to intervene with appropriate care and support for the child," Alberto Gutierrez, Ph.D., director of the office of in vitro diagnostics and radiological health in the FDA’s Center for Devices and Radiological Health, said in the statement.
The test is manufactured by Affymetrix.
The Food and Drug Administration authorized for marketing a postnatal diagnostic test that helps detect genetic causes of developmental delay or intellectual disabilities in children, but it is not intended to be used as a stand-alone test, the agency announced on Jan 17.
With a blood sample, the CytoScan Dx Assay analyzes the entire genome and detects chromosomal variations in regions of the genome that are associated with intellectual and developmental disabilities, such as DiGeorge syndrome, according to the FDA statement. The statement emphasized that is should not be used as a stand-alone diagnostic test, or for preimplantation or prenatal testing or screening; population screening; "or for the detection of, or screening for acquired or genetic aberrations occurring after birth, such as cancer."
Moreover, test results "should only be used in conjunction with other clinical and diagnostic findings, consistent with professional standards of practice, including confirmation by alternative methods, evaluation of parental samples, clinical genetic evaluation, and counseling as appropriate," the statement said. Results should only be interpreted by health care professionals who are board certified in clinical cytogenetics or molecular genetics.
Evaluation of the test included a comparison of results of almost 1,000 blood samples, which found the test was better at detecting certain chromosomal abnormalities than karyotyping and fluorescence in situ hybridization (FISH) chromosomal tests.
"This new tool may help in the identification of possible causes of a child’s developmental delay or intellectual disability, allowing health care providers and parents to intervene with appropriate care and support for the child," Alberto Gutierrez, Ph.D., director of the office of in vitro diagnostics and radiological health in the FDA’s Center for Devices and Radiological Health, said in the statement.
The test is manufactured by Affymetrix.
The Food and Drug Administration authorized for marketing a postnatal diagnostic test that helps detect genetic causes of developmental delay or intellectual disabilities in children, but it is not intended to be used as a stand-alone test, the agency announced on Jan 17.
With a blood sample, the CytoScan Dx Assay analyzes the entire genome and detects chromosomal variations in regions of the genome that are associated with intellectual and developmental disabilities, such as DiGeorge syndrome, according to the FDA statement. The statement emphasized that is should not be used as a stand-alone diagnostic test, or for preimplantation or prenatal testing or screening; population screening; "or for the detection of, or screening for acquired or genetic aberrations occurring after birth, such as cancer."
Moreover, test results "should only be used in conjunction with other clinical and diagnostic findings, consistent with professional standards of practice, including confirmation by alternative methods, evaluation of parental samples, clinical genetic evaluation, and counseling as appropriate," the statement said. Results should only be interpreted by health care professionals who are board certified in clinical cytogenetics or molecular genetics.
Evaluation of the test included a comparison of results of almost 1,000 blood samples, which found the test was better at detecting certain chromosomal abnormalities than karyotyping and fluorescence in situ hybridization (FISH) chromosomal tests.
"This new tool may help in the identification of possible causes of a child’s developmental delay or intellectual disability, allowing health care providers and parents to intervene with appropriate care and support for the child," Alberto Gutierrez, Ph.D., director of the office of in vitro diagnostics and radiological health in the FDA’s Center for Devices and Radiological Health, said in the statement.
The test is manufactured by Affymetrix.
Look beyond FDA for safe pregnancy prescribing
SAN FRANCISCO – Because of significant limitations in the Food and Drug Administration’s pregnancy categories, it’s important to have another reliable resource handy to guide prescribing during pregnancy, Dr. Elizabeth S. Harleman believes.
Three out of four attendees at a conference on women’s health sponsored by the University of California, San Francisco, said that they use the FDA categories to determine a drug’s safety during pregnancy, an informal electronic poll at the meeting showed. There are good reasons for that, Dr. Harleman told the audience. The FDA categories are easily accessible, quick to read, and provide some information.
But they’re also hard to remember and don’t provide sufficient information, she added. "I think it’s going to become increasingly important, as we take care of more women with chronic diseases earlier in life, to think about the implications of conceiving while on the medications that we’re prescribing," said Dr. Harleman, of the department of medicine at the university.
The FDA categories do not provide any gradation of risk. Drugs in the A or B categories are not necessarily safer than category D drugs, she said. A medication can get a B categorization without any human data. The FDA categories give no information about dosing or the timing of administration. "If you give a drug in the first trimester, there may be very different implications than if you give a drug in the third trimester, and you can’t get that information from this classification system," she said.
There’s also no information on the change in risk from one category to another. "We don’t know if this increases the risk from 1% to 5% or from 1% to 80%," Dr. Harleman said. "It’s very hard to make an informed decision about prescribing a medication just using the FDA classification."
Approximately 60% of drugs in category X have no human data behind that classification. A drug can end up in category X simply because it has no utility during pregnancy, such as oral contraceptives. Physicians who don’t know that, however, may erroneously tell a woman who conceived while on an oral contraceptive that the drug increases her baby’s risk for malformation.
The FDA categories rarely get updated. "It’s really more of a legal system" than a helpful clinical tool, she said.
Half of pregnant women who are on medications take category C, D, or X drugs, studies have shown. One in six women of reproductive age receives a category D or X drug, mainly antibiotics, anticonvulsants, statins, benzodiazepines, or warfarin. A study of 488,175 women found that those taking category D or X drugs were no more likely to have documentation in their physicians’ charts of a discussion about contraception.
"In our hurried practices, sometimes it’s hard to remember this, but we need to think about which drugs are absolutely contraindicated and make sure that we are discussing them with our patients," Dr. Harleman said.
The main drugs to avoid during pregnancy are ACE inhibitors, tetracycline, fluoroquinolones, systemic retinoids, warfarin, valproic acid, nonsteroidal anti-inflammatory drugs, and live vaccines. "It’s a pretty short list," she said. "A lot of other drugs are safe in pregnancy; we may just not have as much familiarity with using them."
Dr. Harleman gave three pieces of advice on prescribing during pregnancy: Think hard before starting or stopping any medication. Remember that changes in blood volume and metabolism during pregnancy may mandate increased frequency of dosing or higher doses. And keep one of the following pregnancy prescribing resources available instead of relying solely on the FDA categories:
• The clinical reference tool Micromedex. It has an excellent "ReproRisk" section, she said (micromedex.com).
• Reprotox, a service of the nonprofit Reproductive Toxicology Center, Washington. It synthesizes all the published information on drug safety and provides quick-take summaries that Dr. Harleman believes are worth the low fee for use (reprotox.org/Default.aspx).
• The American College of Physicians’ "Medical Care of the Pregnant Patient," 2nd ed. (Hanover, Pa.: Sheridan Press, 2008) is a good source.
• Motherisk is a service affiliated with the Hospital for Sick Children, Toronto (motherisk.org).
• MotherToBaby is a service of the Organization of Teratology Information Specialists (mothertobaby.org).
• The reference guide "Drugs in Pregnancy and Lactation: A Reference Guide to Fetal and Neonatal Risk," 9th ed. (Philadelphia: Lippincott Williams and Wilkins, 2011) is a good source.
• The book "Medications and Mother’s Milk" (Amarillo, Tex.: Hale Publishing, 2012) is helpful.
Dr. Harleman reported having no financial disclosures.
[email protected]
On Twitter @sherryboschert
SAN FRANCISCO – Because of significant limitations in the Food and Drug Administration’s pregnancy categories, it’s important to have another reliable resource handy to guide prescribing during pregnancy, Dr. Elizabeth S. Harleman believes.
Three out of four attendees at a conference on women’s health sponsored by the University of California, San Francisco, said that they use the FDA categories to determine a drug’s safety during pregnancy, an informal electronic poll at the meeting showed. There are good reasons for that, Dr. Harleman told the audience. The FDA categories are easily accessible, quick to read, and provide some information.
But they’re also hard to remember and don’t provide sufficient information, she added. "I think it’s going to become increasingly important, as we take care of more women with chronic diseases earlier in life, to think about the implications of conceiving while on the medications that we’re prescribing," said Dr. Harleman, of the department of medicine at the university.
The FDA categories do not provide any gradation of risk. Drugs in the A or B categories are not necessarily safer than category D drugs, she said. A medication can get a B categorization without any human data. The FDA categories give no information about dosing or the timing of administration. "If you give a drug in the first trimester, there may be very different implications than if you give a drug in the third trimester, and you can’t get that information from this classification system," she said.
There’s also no information on the change in risk from one category to another. "We don’t know if this increases the risk from 1% to 5% or from 1% to 80%," Dr. Harleman said. "It’s very hard to make an informed decision about prescribing a medication just using the FDA classification."
Approximately 60% of drugs in category X have no human data behind that classification. A drug can end up in category X simply because it has no utility during pregnancy, such as oral contraceptives. Physicians who don’t know that, however, may erroneously tell a woman who conceived while on an oral contraceptive that the drug increases her baby’s risk for malformation.
The FDA categories rarely get updated. "It’s really more of a legal system" than a helpful clinical tool, she said.
Half of pregnant women who are on medications take category C, D, or X drugs, studies have shown. One in six women of reproductive age receives a category D or X drug, mainly antibiotics, anticonvulsants, statins, benzodiazepines, or warfarin. A study of 488,175 women found that those taking category D or X drugs were no more likely to have documentation in their physicians’ charts of a discussion about contraception.
"In our hurried practices, sometimes it’s hard to remember this, but we need to think about which drugs are absolutely contraindicated and make sure that we are discussing them with our patients," Dr. Harleman said.
The main drugs to avoid during pregnancy are ACE inhibitors, tetracycline, fluoroquinolones, systemic retinoids, warfarin, valproic acid, nonsteroidal anti-inflammatory drugs, and live vaccines. "It’s a pretty short list," she said. "A lot of other drugs are safe in pregnancy; we may just not have as much familiarity with using them."
Dr. Harleman gave three pieces of advice on prescribing during pregnancy: Think hard before starting or stopping any medication. Remember that changes in blood volume and metabolism during pregnancy may mandate increased frequency of dosing or higher doses. And keep one of the following pregnancy prescribing resources available instead of relying solely on the FDA categories:
• The clinical reference tool Micromedex. It has an excellent "ReproRisk" section, she said (micromedex.com).
• Reprotox, a service of the nonprofit Reproductive Toxicology Center, Washington. It synthesizes all the published information on drug safety and provides quick-take summaries that Dr. Harleman believes are worth the low fee for use (reprotox.org/Default.aspx).
• The American College of Physicians’ "Medical Care of the Pregnant Patient," 2nd ed. (Hanover, Pa.: Sheridan Press, 2008) is a good source.
• Motherisk is a service affiliated with the Hospital for Sick Children, Toronto (motherisk.org).
• MotherToBaby is a service of the Organization of Teratology Information Specialists (mothertobaby.org).
• The reference guide "Drugs in Pregnancy and Lactation: A Reference Guide to Fetal and Neonatal Risk," 9th ed. (Philadelphia: Lippincott Williams and Wilkins, 2011) is a good source.
• The book "Medications and Mother’s Milk" (Amarillo, Tex.: Hale Publishing, 2012) is helpful.
Dr. Harleman reported having no financial disclosures.
[email protected]
On Twitter @sherryboschert
SAN FRANCISCO – Because of significant limitations in the Food and Drug Administration’s pregnancy categories, it’s important to have another reliable resource handy to guide prescribing during pregnancy, Dr. Elizabeth S. Harleman believes.
Three out of four attendees at a conference on women’s health sponsored by the University of California, San Francisco, said that they use the FDA categories to determine a drug’s safety during pregnancy, an informal electronic poll at the meeting showed. There are good reasons for that, Dr. Harleman told the audience. The FDA categories are easily accessible, quick to read, and provide some information.
But they’re also hard to remember and don’t provide sufficient information, she added. "I think it’s going to become increasingly important, as we take care of more women with chronic diseases earlier in life, to think about the implications of conceiving while on the medications that we’re prescribing," said Dr. Harleman, of the department of medicine at the university.
The FDA categories do not provide any gradation of risk. Drugs in the A or B categories are not necessarily safer than category D drugs, she said. A medication can get a B categorization without any human data. The FDA categories give no information about dosing or the timing of administration. "If you give a drug in the first trimester, there may be very different implications than if you give a drug in the third trimester, and you can’t get that information from this classification system," she said.
There’s also no information on the change in risk from one category to another. "We don’t know if this increases the risk from 1% to 5% or from 1% to 80%," Dr. Harleman said. "It’s very hard to make an informed decision about prescribing a medication just using the FDA classification."
Approximately 60% of drugs in category X have no human data behind that classification. A drug can end up in category X simply because it has no utility during pregnancy, such as oral contraceptives. Physicians who don’t know that, however, may erroneously tell a woman who conceived while on an oral contraceptive that the drug increases her baby’s risk for malformation.
The FDA categories rarely get updated. "It’s really more of a legal system" than a helpful clinical tool, she said.
Half of pregnant women who are on medications take category C, D, or X drugs, studies have shown. One in six women of reproductive age receives a category D or X drug, mainly antibiotics, anticonvulsants, statins, benzodiazepines, or warfarin. A study of 488,175 women found that those taking category D or X drugs were no more likely to have documentation in their physicians’ charts of a discussion about contraception.
"In our hurried practices, sometimes it’s hard to remember this, but we need to think about which drugs are absolutely contraindicated and make sure that we are discussing them with our patients," Dr. Harleman said.
The main drugs to avoid during pregnancy are ACE inhibitors, tetracycline, fluoroquinolones, systemic retinoids, warfarin, valproic acid, nonsteroidal anti-inflammatory drugs, and live vaccines. "It’s a pretty short list," she said. "A lot of other drugs are safe in pregnancy; we may just not have as much familiarity with using them."
Dr. Harleman gave three pieces of advice on prescribing during pregnancy: Think hard before starting or stopping any medication. Remember that changes in blood volume and metabolism during pregnancy may mandate increased frequency of dosing or higher doses. And keep one of the following pregnancy prescribing resources available instead of relying solely on the FDA categories:
• The clinical reference tool Micromedex. It has an excellent "ReproRisk" section, she said (micromedex.com).
• Reprotox, a service of the nonprofit Reproductive Toxicology Center, Washington. It synthesizes all the published information on drug safety and provides quick-take summaries that Dr. Harleman believes are worth the low fee for use (reprotox.org/Default.aspx).
• The American College of Physicians’ "Medical Care of the Pregnant Patient," 2nd ed. (Hanover, Pa.: Sheridan Press, 2008) is a good source.
• Motherisk is a service affiliated with the Hospital for Sick Children, Toronto (motherisk.org).
• MotherToBaby is a service of the Organization of Teratology Information Specialists (mothertobaby.org).
• The reference guide "Drugs in Pregnancy and Lactation: A Reference Guide to Fetal and Neonatal Risk," 9th ed. (Philadelphia: Lippincott Williams and Wilkins, 2011) is a good source.
• The book "Medications and Mother’s Milk" (Amarillo, Tex.: Hale Publishing, 2012) is helpful.
Dr. Harleman reported having no financial disclosures.
[email protected]
On Twitter @sherryboschert
EXPERT ANALYSIS FROM A CONFERENCE ON WOMEN’S HEALTH