User login
Folic acid supplementation low among women with epilepsy
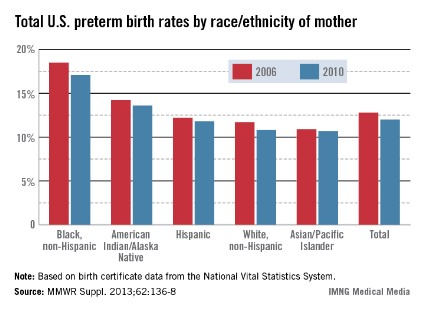
WASHINGTON – Only about 43% of reproductive-age women with epilepsy who completed a Web-based survey were taking a folic acid supplement, according to interim findings from the Epilepsy Birth Control Registry.
Furthermore, only 46% of those considered to be at risk for becoming pregnant (for example, those who were sexually active and who had no history of infertility) reported taking a folic acid supplement, and among respondents who were taking antiepileptic drugs (AEDs), the lowest rate of folic acid supplement use (25%) was in those taking valproate, a folic acid antagonist, Dr. Andrew G. Herzog reported at the annual meeting of the American Epilepsy Society.
Folic acid deficiency is known to be associated with fetal loss and risk of neural tube defects, and valproate is the AED associated with the highest risk of neural tube malformations, said Dr. Herzog of Harvard Medical School, Boston, and Beth Israel Deaconess Medical Center, Wellesley, Mass.
However, respondents taking valproate in this study were the least likely to have a college degree, and having a college degree was found to be associated with greater likelihood of taking a folic acid supplement. In fact, having an associate college degree or higher was the only significant demographic predictor of folic acid use; 50% of those with a college degree were taking folic acid, compared with 39% of those without a college degree, and those with an advanced degree were twice as likely as those with only a high school education to be taking folic acid, but the numbers were insufficient for determining whether AED type or education level predicted folic acid use, Dr. Herzog noted.
Study subjects were women with epilepsy aged 18 to 47 years. The first 650 to complete the survey at the registry site were included in the analysis.
Prior findings from the registry showed that this is largely a population at high risk for pregnancy; about 60% of pregnancies among respondents were unintended, compared with about 47% in the general population, he said.
Yet, the only category of respondents for which folic acid use was more than 50%, including, for example, those at risk for pregnancy, those not at risk for pregnancy, those using birth control, those not using birth control, and those trying to become pregnant, was the latter – those women actively trying to become pregnant, of whom 70% were taking folic acid, he said.
Factors including age, seizure type, and insurance status were not associated with folic acid use.
Importantly, seeing a health care professional within the year prior to the survey also was not associated with increased folic acid use.
This is concerning, given that it has been known for three decades that folic acid deficiency is associated with serious consequences, Dr. Herzog said, suggesting that education about the importance of supplementation may get pushed aside by busy physicians focused more on seizure activity and epilepsy treatment.
Dr. Herzog reported having no disclosures.
WASHINGTON – Only about 43% of reproductive-age women with epilepsy who completed a Web-based survey were taking a folic acid supplement, according to interim findings from the Epilepsy Birth Control Registry.
Furthermore, only 46% of those considered to be at risk for becoming pregnant (for example, those who were sexually active and who had no history of infertility) reported taking a folic acid supplement, and among respondents who were taking antiepileptic drugs (AEDs), the lowest rate of folic acid supplement use (25%) was in those taking valproate, a folic acid antagonist, Dr. Andrew G. Herzog reported at the annual meeting of the American Epilepsy Society.
Folic acid deficiency is known to be associated with fetal loss and risk of neural tube defects, and valproate is the AED associated with the highest risk of neural tube malformations, said Dr. Herzog of Harvard Medical School, Boston, and Beth Israel Deaconess Medical Center, Wellesley, Mass.
However, respondents taking valproate in this study were the least likely to have a college degree, and having a college degree was found to be associated with greater likelihood of taking a folic acid supplement. In fact, having an associate college degree or higher was the only significant demographic predictor of folic acid use; 50% of those with a college degree were taking folic acid, compared with 39% of those without a college degree, and those with an advanced degree were twice as likely as those with only a high school education to be taking folic acid, but the numbers were insufficient for determining whether AED type or education level predicted folic acid use, Dr. Herzog noted.
Study subjects were women with epilepsy aged 18 to 47 years. The first 650 to complete the survey at the registry site were included in the analysis.
Prior findings from the registry showed that this is largely a population at high risk for pregnancy; about 60% of pregnancies among respondents were unintended, compared with about 47% in the general population, he said.
Yet, the only category of respondents for which folic acid use was more than 50%, including, for example, those at risk for pregnancy, those not at risk for pregnancy, those using birth control, those not using birth control, and those trying to become pregnant, was the latter – those women actively trying to become pregnant, of whom 70% were taking folic acid, he said.
Factors including age, seizure type, and insurance status were not associated with folic acid use.
Importantly, seeing a health care professional within the year prior to the survey also was not associated with increased folic acid use.
This is concerning, given that it has been known for three decades that folic acid deficiency is associated with serious consequences, Dr. Herzog said, suggesting that education about the importance of supplementation may get pushed aside by busy physicians focused more on seizure activity and epilepsy treatment.
Dr. Herzog reported having no disclosures.
WASHINGTON – Only about 43% of reproductive-age women with epilepsy who completed a Web-based survey were taking a folic acid supplement, according to interim findings from the Epilepsy Birth Control Registry.
Furthermore, only 46% of those considered to be at risk for becoming pregnant (for example, those who were sexually active and who had no history of infertility) reported taking a folic acid supplement, and among respondents who were taking antiepileptic drugs (AEDs), the lowest rate of folic acid supplement use (25%) was in those taking valproate, a folic acid antagonist, Dr. Andrew G. Herzog reported at the annual meeting of the American Epilepsy Society.
Folic acid deficiency is known to be associated with fetal loss and risk of neural tube defects, and valproate is the AED associated with the highest risk of neural tube malformations, said Dr. Herzog of Harvard Medical School, Boston, and Beth Israel Deaconess Medical Center, Wellesley, Mass.
However, respondents taking valproate in this study were the least likely to have a college degree, and having a college degree was found to be associated with greater likelihood of taking a folic acid supplement. In fact, having an associate college degree or higher was the only significant demographic predictor of folic acid use; 50% of those with a college degree were taking folic acid, compared with 39% of those without a college degree, and those with an advanced degree were twice as likely as those with only a high school education to be taking folic acid, but the numbers were insufficient for determining whether AED type or education level predicted folic acid use, Dr. Herzog noted.
Study subjects were women with epilepsy aged 18 to 47 years. The first 650 to complete the survey at the registry site were included in the analysis.
Prior findings from the registry showed that this is largely a population at high risk for pregnancy; about 60% of pregnancies among respondents were unintended, compared with about 47% in the general population, he said.
Yet, the only category of respondents for which folic acid use was more than 50%, including, for example, those at risk for pregnancy, those not at risk for pregnancy, those using birth control, those not using birth control, and those trying to become pregnant, was the latter – those women actively trying to become pregnant, of whom 70% were taking folic acid, he said.
Factors including age, seizure type, and insurance status were not associated with folic acid use.
Importantly, seeing a health care professional within the year prior to the survey also was not associated with increased folic acid use.
This is concerning, given that it has been known for three decades that folic acid deficiency is associated with serious consequences, Dr. Herzog said, suggesting that education about the importance of supplementation may get pushed aside by busy physicians focused more on seizure activity and epilepsy treatment.
Dr. Herzog reported having no disclosures.
AT AES 2013
Major finding: Only 46% of reproductive-age women with epilepsy who were considered to be at risk for becoming pregnant were taking a folic acid supplement.
Data source: A Web-based survey involving 650 women.
Disclosures: Dr. Herzog reported having no disclosures.
Psychiatric Medications and Lactation: Informing Clinical Decisions
Over the last 2 decades, abundant data on the reproductive safety of medications used to treat psychiatric disorders have become available, filling in many gaps with respect to our knowledge about the safety of commonly used psychiatric medications during pregnancy. But the availability of such safety data with respect to the use of these agents during breastfeeding is less complete.
Because of fears of potential adverse effects on infants associated with psychotropic drug use during lactation, many women with a psychiatric disorder who are treated with a range of psychiatric medications are advised not to breastfeed; or if they choose to breastfeed, they are counseled to avoid taking the essential medication that has made them well. This has been a somewhat intuitive, cautious approach. However, in my 25 years of clinical experience taking care of pregnant and postpartum women with a range of psychiatric disorders, one sad scenario I have frequently witnessed is the woman who decides to defer pharmacologic treatment for severe postpartum psychiatric illness after being counseled to defer treatment given a wish to breastfeed. Those women often have been psychiatrically ill for months while breastfeeding after having decided to defer their own treatment because they do not want to expose the baby to even scant amounts of medication secreted into breast milk associated with use of a needed medicine during lactation.
In a recently published clinical report from the American Academy of Pediatrics committee on drugs, authors suggest that advice not to breastfeed or to uniformly avoid medications while nursing because of possible adverse effects in the infant is often not evidence based and may not be necessary in many cases. The committee states that most drugs do not pose a risk to the mother or infant who is nursing, and that "the benefits of breastfeeding outweigh the risks of exposure to most therapeutic agents via human breast milk" (Pediatrics 2013;132:e796-e809).
The report points out that for certain drugs, a careful risk-benefit analysis is needed, especially for drugs that are concentrated in human milk, those with unproven benefits, and those with long half-lives. Importantly, the report notes say that decisions about the use of medications during lactation have to be made on a case-by-case basis. A concrete example would be exercising appropriate vigilance about the use of these medicines in premature infants with immature metabolism.
The report, published on-line in Pediatrics in August 2013, includes a section on antidepressants, anxiolytics, and antipsychotics. As a resource for clinicians, the report highlights LactMed, part of the National Library of Medicine’s toxicology data network (TOXNET), which provides real-time updated scientific information on the excretion of drugs into breast milk.
The report makes the important distinction regarding the range of clinical decisions that get made in the context of different clinical situations. For example, at our center, patients frequently present with questions about whether to use psychiatric medications while breastfeeding when these women have already been taking the medication during pregnancy for treatment of underlying psychiatric disorder. Others make queries about introduction of pharmacologic therapy in the early postpartum period in the context, for example, of new-onset postpartum depression. Specifically, a woman with a history of psychiatric disorder who is treated with antidepressant during pregnancy may continue that medication across the postpartum period to attenuate risk for postpartum depression, particularly if she has a history of recurrent disease, or depressive relapse when medication has been discontinued. That is clinical scenario differs from that of a woman who develops new onset of depression during the postpartum period.
One part of the AAP report addresses use of certain psychiatric medications in the context of available information from the literature regarding extent of excretion of these medications into breast milk. This section states that many antianxiety drugs, antidepressants, and mood stabilizers are excreted in low concentrations into human milk, with the estimated infant doses under 2% of the weight-adjusted maternal dose. However, the authors also cite small series or case reports where infant plasma levels of some drugs were reported to exceed 10% of maternal plasma concentrations. They list 13 such drugs, which include selective serotonin reuptake inhibitors (SSRIs), antipsychotics, anxiolytics, and mood stabilizers. It is important to keep in mind that the number of these cases is small and represent a very small proportion of cases, when the total denominator of reports in the literature of psychotropic drug use during lactation is considered. For example, olanzapine, a second generation antipsychotic is highlighted as an agent of concern based on one case report (J. Psychopharmacol. 2010;24:121-3).
The take-home message for the clinician is that all psychotropics are excreted into breast milk, even if quantification of the agents in breast milk or infant plasma reveals relatively scant concentration (J. Clin. Psychiatry 2003;64:73-80). If mom takes the medicine coincident with lactation, baby is exposed. At our center, we are usually reluctant to discontinue a medication such as an atypical antipsychotic to treat bipolar disorder in the postpartum period even if the mom chooses to breastfeed considering the extent to which women with bipolar disorder are at a high risk for relapse during the puerperium.
Ironically, we probably have more information regarding the excretion of antidepressants and drugs such as lamotrigine, used as a mood stabilizer, into breast milk than most medicines women take during the postpartum period, with data over the past 15 years suggesting that these medications, like other medications, are excreted into breast milk and are present in infant plasma in extremely sparse concentrations. It is noteworthy that cases of frank newborn toxicity directly associated with mothers who breastfeed on psychiatric medications are extremely few and far between, and are anecdotal at best. For some context, the literature on the effects of SSRI use during pregnancy is vast and prevalence of use of these medications during pregnancy and the postpartum period is substantial; that being said, reports of adverse effects reported in the babies of women who breastfeed while taking an SSRI are scant and thus at least somewhat reassuring.
And yet, consistently, I have witnessed that psychiatric medications are highlighted in the literature as particular agents of concern when it comes to lactation, compared with other medicines, for which only sparse data are available. Whether this reflects a bias about the necessity of treating psychiatric disorders during the postpartum period is unknown. Certainly, the long-term implications for the infant of exposure to low concentrations of psychiatric and nonpsychiatric medications in the context of breastfeeding exposure have yet to be clarified.
Whether a woman treated with a psychiatric medication during the post partum should breastfeed is a prime example of a clinical scenario in which there is no perfect decision, and we need to make the best decision possible, taking into account the available data, and the mother’s psychiatric disorder and her wishes. Some women may be extremely committed to breastfeeding and may choose to breastfeed, acknowledging some of the unknowns regarding these medications during lactation, while other women consider some of the ambiguity associated with the long-term effects of exposure while lactating and may choose not to breastfeed.
It is noteworthy that the AAP committee on drugs concluded the benefits of breastfeeding outweigh the risk of exposure to most therapeutic agents via human milk. And those at our center would certainly suggest that this is the case for psychiatric medications, particularly those used to sustain postpartum maternal psychiatric well-being, which is so critical. As is the case with any clinical decision, and certainly with respect to the use of psychiatric medications during pregnancy and lactation, the decision to treat is contingent on a careful risk-benefit analysis, where the risks of exposure to a medicine is weighed against the risk of untreated psychiatric illness. Even with the well-documented benefits of breastfeeding, nothing should trump the treatment of postpartum psychiatric illness, even if the cost is deferring breastfeeding. Treatment cannot be deferred because of the impact of untreated maternal psychiatric illness on maternal morbidity and on the development of children.
Dr. Cohen is the director of the Center for Women’s Mental Health at Massachusetts General Hospital in Boston, which provides information about reproductive mental health at www.womensmentalhealth.org. To comment, e-mail him at [email protected]. Dr. Cohen has been a consultant to manufacturers of antidepressants and antipsychotic medications.
Over the last 2 decades, abundant data on the reproductive safety of medications used to treat psychiatric disorders have become available, filling in many gaps with respect to our knowledge about the safety of commonly used psychiatric medications during pregnancy. But the availability of such safety data with respect to the use of these agents during breastfeeding is less complete.
Because of fears of potential adverse effects on infants associated with psychotropic drug use during lactation, many women with a psychiatric disorder who are treated with a range of psychiatric medications are advised not to breastfeed; or if they choose to breastfeed, they are counseled to avoid taking the essential medication that has made them well. This has been a somewhat intuitive, cautious approach. However, in my 25 years of clinical experience taking care of pregnant and postpartum women with a range of psychiatric disorders, one sad scenario I have frequently witnessed is the woman who decides to defer pharmacologic treatment for severe postpartum psychiatric illness after being counseled to defer treatment given a wish to breastfeed. Those women often have been psychiatrically ill for months while breastfeeding after having decided to defer their own treatment because they do not want to expose the baby to even scant amounts of medication secreted into breast milk associated with use of a needed medicine during lactation.
In a recently published clinical report from the American Academy of Pediatrics committee on drugs, authors suggest that advice not to breastfeed or to uniformly avoid medications while nursing because of possible adverse effects in the infant is often not evidence based and may not be necessary in many cases. The committee states that most drugs do not pose a risk to the mother or infant who is nursing, and that "the benefits of breastfeeding outweigh the risks of exposure to most therapeutic agents via human breast milk" (Pediatrics 2013;132:e796-e809).
The report points out that for certain drugs, a careful risk-benefit analysis is needed, especially for drugs that are concentrated in human milk, those with unproven benefits, and those with long half-lives. Importantly, the report notes say that decisions about the use of medications during lactation have to be made on a case-by-case basis. A concrete example would be exercising appropriate vigilance about the use of these medicines in premature infants with immature metabolism.
The report, published on-line in Pediatrics in August 2013, includes a section on antidepressants, anxiolytics, and antipsychotics. As a resource for clinicians, the report highlights LactMed, part of the National Library of Medicine’s toxicology data network (TOXNET), which provides real-time updated scientific information on the excretion of drugs into breast milk.
The report makes the important distinction regarding the range of clinical decisions that get made in the context of different clinical situations. For example, at our center, patients frequently present with questions about whether to use psychiatric medications while breastfeeding when these women have already been taking the medication during pregnancy for treatment of underlying psychiatric disorder. Others make queries about introduction of pharmacologic therapy in the early postpartum period in the context, for example, of new-onset postpartum depression. Specifically, a woman with a history of psychiatric disorder who is treated with antidepressant during pregnancy may continue that medication across the postpartum period to attenuate risk for postpartum depression, particularly if she has a history of recurrent disease, or depressive relapse when medication has been discontinued. That is clinical scenario differs from that of a woman who develops new onset of depression during the postpartum period.
One part of the AAP report addresses use of certain psychiatric medications in the context of available information from the literature regarding extent of excretion of these medications into breast milk. This section states that many antianxiety drugs, antidepressants, and mood stabilizers are excreted in low concentrations into human milk, with the estimated infant doses under 2% of the weight-adjusted maternal dose. However, the authors also cite small series or case reports where infant plasma levels of some drugs were reported to exceed 10% of maternal plasma concentrations. They list 13 such drugs, which include selective serotonin reuptake inhibitors (SSRIs), antipsychotics, anxiolytics, and mood stabilizers. It is important to keep in mind that the number of these cases is small and represent a very small proportion of cases, when the total denominator of reports in the literature of psychotropic drug use during lactation is considered. For example, olanzapine, a second generation antipsychotic is highlighted as an agent of concern based on one case report (J. Psychopharmacol. 2010;24:121-3).
The take-home message for the clinician is that all psychotropics are excreted into breast milk, even if quantification of the agents in breast milk or infant plasma reveals relatively scant concentration (J. Clin. Psychiatry 2003;64:73-80). If mom takes the medicine coincident with lactation, baby is exposed. At our center, we are usually reluctant to discontinue a medication such as an atypical antipsychotic to treat bipolar disorder in the postpartum period even if the mom chooses to breastfeed considering the extent to which women with bipolar disorder are at a high risk for relapse during the puerperium.
Ironically, we probably have more information regarding the excretion of antidepressants and drugs such as lamotrigine, used as a mood stabilizer, into breast milk than most medicines women take during the postpartum period, with data over the past 15 years suggesting that these medications, like other medications, are excreted into breast milk and are present in infant plasma in extremely sparse concentrations. It is noteworthy that cases of frank newborn toxicity directly associated with mothers who breastfeed on psychiatric medications are extremely few and far between, and are anecdotal at best. For some context, the literature on the effects of SSRI use during pregnancy is vast and prevalence of use of these medications during pregnancy and the postpartum period is substantial; that being said, reports of adverse effects reported in the babies of women who breastfeed while taking an SSRI are scant and thus at least somewhat reassuring.
And yet, consistently, I have witnessed that psychiatric medications are highlighted in the literature as particular agents of concern when it comes to lactation, compared with other medicines, for which only sparse data are available. Whether this reflects a bias about the necessity of treating psychiatric disorders during the postpartum period is unknown. Certainly, the long-term implications for the infant of exposure to low concentrations of psychiatric and nonpsychiatric medications in the context of breastfeeding exposure have yet to be clarified.
Whether a woman treated with a psychiatric medication during the post partum should breastfeed is a prime example of a clinical scenario in which there is no perfect decision, and we need to make the best decision possible, taking into account the available data, and the mother’s psychiatric disorder and her wishes. Some women may be extremely committed to breastfeeding and may choose to breastfeed, acknowledging some of the unknowns regarding these medications during lactation, while other women consider some of the ambiguity associated with the long-term effects of exposure while lactating and may choose not to breastfeed.
It is noteworthy that the AAP committee on drugs concluded the benefits of breastfeeding outweigh the risk of exposure to most therapeutic agents via human milk. And those at our center would certainly suggest that this is the case for psychiatric medications, particularly those used to sustain postpartum maternal psychiatric well-being, which is so critical. As is the case with any clinical decision, and certainly with respect to the use of psychiatric medications during pregnancy and lactation, the decision to treat is contingent on a careful risk-benefit analysis, where the risks of exposure to a medicine is weighed against the risk of untreated psychiatric illness. Even with the well-documented benefits of breastfeeding, nothing should trump the treatment of postpartum psychiatric illness, even if the cost is deferring breastfeeding. Treatment cannot be deferred because of the impact of untreated maternal psychiatric illness on maternal morbidity and on the development of children.
Dr. Cohen is the director of the Center for Women’s Mental Health at Massachusetts General Hospital in Boston, which provides information about reproductive mental health at www.womensmentalhealth.org. To comment, e-mail him at [email protected]. Dr. Cohen has been a consultant to manufacturers of antidepressants and antipsychotic medications.
Over the last 2 decades, abundant data on the reproductive safety of medications used to treat psychiatric disorders have become available, filling in many gaps with respect to our knowledge about the safety of commonly used psychiatric medications during pregnancy. But the availability of such safety data with respect to the use of these agents during breastfeeding is less complete.
Because of fears of potential adverse effects on infants associated with psychotropic drug use during lactation, many women with a psychiatric disorder who are treated with a range of psychiatric medications are advised not to breastfeed; or if they choose to breastfeed, they are counseled to avoid taking the essential medication that has made them well. This has been a somewhat intuitive, cautious approach. However, in my 25 years of clinical experience taking care of pregnant and postpartum women with a range of psychiatric disorders, one sad scenario I have frequently witnessed is the woman who decides to defer pharmacologic treatment for severe postpartum psychiatric illness after being counseled to defer treatment given a wish to breastfeed. Those women often have been psychiatrically ill for months while breastfeeding after having decided to defer their own treatment because they do not want to expose the baby to even scant amounts of medication secreted into breast milk associated with use of a needed medicine during lactation.
In a recently published clinical report from the American Academy of Pediatrics committee on drugs, authors suggest that advice not to breastfeed or to uniformly avoid medications while nursing because of possible adverse effects in the infant is often not evidence based and may not be necessary in many cases. The committee states that most drugs do not pose a risk to the mother or infant who is nursing, and that "the benefits of breastfeeding outweigh the risks of exposure to most therapeutic agents via human breast milk" (Pediatrics 2013;132:e796-e809).
The report points out that for certain drugs, a careful risk-benefit analysis is needed, especially for drugs that are concentrated in human milk, those with unproven benefits, and those with long half-lives. Importantly, the report notes say that decisions about the use of medications during lactation have to be made on a case-by-case basis. A concrete example would be exercising appropriate vigilance about the use of these medicines in premature infants with immature metabolism.
The report, published on-line in Pediatrics in August 2013, includes a section on antidepressants, anxiolytics, and antipsychotics. As a resource for clinicians, the report highlights LactMed, part of the National Library of Medicine’s toxicology data network (TOXNET), which provides real-time updated scientific information on the excretion of drugs into breast milk.
The report makes the important distinction regarding the range of clinical decisions that get made in the context of different clinical situations. For example, at our center, patients frequently present with questions about whether to use psychiatric medications while breastfeeding when these women have already been taking the medication during pregnancy for treatment of underlying psychiatric disorder. Others make queries about introduction of pharmacologic therapy in the early postpartum period in the context, for example, of new-onset postpartum depression. Specifically, a woman with a history of psychiatric disorder who is treated with antidepressant during pregnancy may continue that medication across the postpartum period to attenuate risk for postpartum depression, particularly if she has a history of recurrent disease, or depressive relapse when medication has been discontinued. That is clinical scenario differs from that of a woman who develops new onset of depression during the postpartum period.
One part of the AAP report addresses use of certain psychiatric medications in the context of available information from the literature regarding extent of excretion of these medications into breast milk. This section states that many antianxiety drugs, antidepressants, and mood stabilizers are excreted in low concentrations into human milk, with the estimated infant doses under 2% of the weight-adjusted maternal dose. However, the authors also cite small series or case reports where infant plasma levels of some drugs were reported to exceed 10% of maternal plasma concentrations. They list 13 such drugs, which include selective serotonin reuptake inhibitors (SSRIs), antipsychotics, anxiolytics, and mood stabilizers. It is important to keep in mind that the number of these cases is small and represent a very small proportion of cases, when the total denominator of reports in the literature of psychotropic drug use during lactation is considered. For example, olanzapine, a second generation antipsychotic is highlighted as an agent of concern based on one case report (J. Psychopharmacol. 2010;24:121-3).
The take-home message for the clinician is that all psychotropics are excreted into breast milk, even if quantification of the agents in breast milk or infant plasma reveals relatively scant concentration (J. Clin. Psychiatry 2003;64:73-80). If mom takes the medicine coincident with lactation, baby is exposed. At our center, we are usually reluctant to discontinue a medication such as an atypical antipsychotic to treat bipolar disorder in the postpartum period even if the mom chooses to breastfeed considering the extent to which women with bipolar disorder are at a high risk for relapse during the puerperium.
Ironically, we probably have more information regarding the excretion of antidepressants and drugs such as lamotrigine, used as a mood stabilizer, into breast milk than most medicines women take during the postpartum period, with data over the past 15 years suggesting that these medications, like other medications, are excreted into breast milk and are present in infant plasma in extremely sparse concentrations. It is noteworthy that cases of frank newborn toxicity directly associated with mothers who breastfeed on psychiatric medications are extremely few and far between, and are anecdotal at best. For some context, the literature on the effects of SSRI use during pregnancy is vast and prevalence of use of these medications during pregnancy and the postpartum period is substantial; that being said, reports of adverse effects reported in the babies of women who breastfeed while taking an SSRI are scant and thus at least somewhat reassuring.
And yet, consistently, I have witnessed that psychiatric medications are highlighted in the literature as particular agents of concern when it comes to lactation, compared with other medicines, for which only sparse data are available. Whether this reflects a bias about the necessity of treating psychiatric disorders during the postpartum period is unknown. Certainly, the long-term implications for the infant of exposure to low concentrations of psychiatric and nonpsychiatric medications in the context of breastfeeding exposure have yet to be clarified.
Whether a woman treated with a psychiatric medication during the post partum should breastfeed is a prime example of a clinical scenario in which there is no perfect decision, and we need to make the best decision possible, taking into account the available data, and the mother’s psychiatric disorder and her wishes. Some women may be extremely committed to breastfeeding and may choose to breastfeed, acknowledging some of the unknowns regarding these medications during lactation, while other women consider some of the ambiguity associated with the long-term effects of exposure while lactating and may choose not to breastfeed.
It is noteworthy that the AAP committee on drugs concluded the benefits of breastfeeding outweigh the risk of exposure to most therapeutic agents via human milk. And those at our center would certainly suggest that this is the case for psychiatric medications, particularly those used to sustain postpartum maternal psychiatric well-being, which is so critical. As is the case with any clinical decision, and certainly with respect to the use of psychiatric medications during pregnancy and lactation, the decision to treat is contingent on a careful risk-benefit analysis, where the risks of exposure to a medicine is weighed against the risk of untreated psychiatric illness. Even with the well-documented benefits of breastfeeding, nothing should trump the treatment of postpartum psychiatric illness, even if the cost is deferring breastfeeding. Treatment cannot be deferred because of the impact of untreated maternal psychiatric illness on maternal morbidity and on the development of children.
Dr. Cohen is the director of the Center for Women’s Mental Health at Massachusetts General Hospital in Boston, which provides information about reproductive mental health at www.womensmentalhealth.org. To comment, e-mail him at [email protected]. Dr. Cohen has been a consultant to manufacturers of antidepressants and antipsychotic medications.
Psychiatric medications and lactation: Informing clinical decisions
Over the last 2 decades, abundant data on the reproductive safety of medications used to treat psychiatric disorders have become available, filling in many gaps with respect to our knowledge about the safety of commonly used psychiatric medications during pregnancy. But the availability of such safety data with respect to the use of these agents during breastfeeding is less complete.
Because of fears of potential adverse effects on infants associated with psychotropic drug use during lactation, many women with a psychiatric disorder who are treated with a range of psychiatric medications are advised not to breastfeed; or if they choose to breastfeed, they are counseled to avoid taking the essential medication that has made them well. This has been a somewhat intuitive, cautious approach. However, in my 25 years of clinical experience taking care of pregnant and postpartum women with a range of psychiatric disorders, one sad scenario I have frequently witnessed is the woman who decides to defer pharmacologic treatment for severe postpartum psychiatric illness after being counseled to defer treatment given a wish to breastfeed. Those women often have been psychiatrically ill for months while breastfeeding after having decided to defer their own treatment because they do not want to expose the baby to even scant amounts of medication secreted into breast milk associated with use of a needed medicine during lactation.
In a recently published clinical report from the American Academy of Pediatrics committee on drugs, authors suggest that advice not to breastfeed or to uniformly avoid medications while nursing because of possible adverse effects in the infant is often not evidence based and may not be necessary in many cases. The committee states that most drugs do not pose a risk to the mother or infant who is nursing, and that "the benefits of breastfeeding outweigh the risks of exposure to most therapeutic agents via human breast milk" (Pediatrics 2013;132:e796-e809).
The report points out that for certain drugs, a careful risk-benefit analysis is needed, especially for drugs that are concentrated in human milk, those with unproven benefits, and those with long half-lives. Importantly, the report notes say that decisions about the use of medications during lactation have to be made on a case-by-case basis. A concrete example would be exercising appropriate vigilance about the use of these medicines in premature infants with immature metabolism.
The report, published on-line in Pediatrics in August 2013, includes a section on antidepressants, anxiolytics, and antipsychotics. As a resource for clinicians, the report highlights LactMed, part of the National Library of Medicine’s toxicology data network (TOXNET), which provides real-time updated scientific information on the excretion of drugs into breast milk.
The report makes the important distinction regarding the range of clinical decisions that get made in the context of different clinical situations. For example, at our center, patients frequently present with questions about whether to use psychiatric medications while breastfeeding when these women have already been taking the medication during pregnancy for treatment of underlying psychiatric disorder. Others make queries about introduction of pharmacologic therapy in the early postpartum period in the context, for example, of new-onset postpartum depression. Specifically, a woman with a history of psychiatric disorder who is treated with antidepressant during pregnancy may continue that medication across the postpartum period to attenuate risk for postpartum depression, particularly if she has a history of recurrent disease, or depressive relapse when medication has been discontinued. That is clinical scenario differs from that of a woman who develops new onset of depression during the postpartum period.
One part of the AAP report addresses use of certain psychiatric medications in the context of available information from the literature regarding extent of excretion of these medications into breast milk. This section states that many antianxiety drugs, antidepressants, and mood stabilizers are excreted in low concentrations into human milk, with the estimated infant doses under 2% of the weight-adjusted maternal dose. However, the authors also cite small series or case reports where infant plasma levels of some drugs were reported to exceed 10% of maternal plasma concentrations. They list 13 such drugs, which include selective serotonin reuptake inhibitors (SSRIs), antipsychotics, anxiolytics, and mood stabilizers. It is important to keep in mind that the number of these cases is small and represent a very small proportion of cases, when the total denominator of reports in the literature of psychotropic drug use during lactation is considered. For example, olanzapine, a second generation antipsychotic is highlighted as an agent of concern based on one case report (J. Psychopharmacol. 2010;24:121-3).
The take-home message for the clinician is that all psychotropics are excreted into breast milk, even if quantification of the agents in breast milk or infant plasma reveals relatively scant concentration (J. Clin. Psychiatry 2003;64:73-80). If mom takes the medicine coincident with lactation, baby is exposed. At our center, we are usually reluctant to discontinue a medication such as an atypical antipsychotic to treat bipolar disorder in the postpartum period even if the mom chooses to breastfeed considering the extent to which women with bipolar disorder are at a high risk for relapse during the puerperium.
Ironically, we probably have more information regarding the excretion of antidepressants and drugs such as lamotrigine, used as a mood stabilizer, into breast milk than most medicines women take during the postpartum period, with data over the past 15 years suggesting that these medications, like other medications, are excreted into breast milk and are present in infant plasma in extremely sparse concentrations. It is noteworthy that cases of frank newborn toxicity directly associated with mothers who breastfeed on psychiatric medications are extremely few and far between, and are anecdotal at best. For some context, the literature on the effects of SSRI use during pregnancy is vast and prevalence of use of these medications during pregnancy and the postpartum period is substantial; that being said, reports of adverse effects reported in the babies of women who breastfeed while taking an SSRI are scant and thus at least somewhat reassuring.
And yet, consistently, I have witnessed that psychiatric medications are highlighted in the literature as particular agents of concern when it comes to lactation, compared with other medicines, for which only sparse data are available. Whether this reflects a bias about the necessity of treating psychiatric disorders during the postpartum period is unknown. Certainly, the long-term implications for the infant of exposure to low concentrations of psychiatric and nonpsychiatric medications in the context of breastfeeding exposure have yet to be clarified.
Whether a woman treated with a psychiatric medication during the post partum should breastfeed is a prime example of a clinical scenario in which there is no perfect decision, and we need to make the best decision possible, taking into account the available data, and the mother’s psychiatric disorder and her wishes. Some women may be extremely committed to breastfeeding and may choose to breastfeed, acknowledging some of the unknowns regarding these medications during lactation, while other women consider some of the ambiguity associated with the long-term effects of exposure while lactating and may choose not to breastfeed.
It is noteworthy that the AAP committee on drugs concluded the benefits of breastfeeding outweigh the risk of exposure to most therapeutic agents via human milk. And those at our center would certainly suggest that this is the case for psychiatric medications, particularly those used to sustain postpartum maternal psychiatric well-being, which is so critical. As is the case with any clinical decision, and certainly with respect to the use of psychiatric medications during pregnancy and lactation, the decision to treat is contingent on a careful risk-benefit analysis, where the risks of exposure to a medicine is weighed against the risk of untreated psychiatric illness. Even with the well-documented benefits of breastfeeding, nothing should trump the treatment of postpartum psychiatric illness, even if the cost is deferring breastfeeding. Treatment cannot be deferred because of the impact of untreated maternal psychiatric illness on maternal morbidity and on the development of children.
Dr. Cohen is the director of the Center for Women’s Mental Health at Massachusetts General Hospital in Boston, which provides information about reproductive mental health at www.womensmentalhealth.org. To comment, e-mail him at [email protected]. Dr. Cohen has been a consultant to manufacturers of antidepressants and antipsychotic medications.
Over the last 2 decades, abundant data on the reproductive safety of medications used to treat psychiatric disorders have become available, filling in many gaps with respect to our knowledge about the safety of commonly used psychiatric medications during pregnancy. But the availability of such safety data with respect to the use of these agents during breastfeeding is less complete.
Because of fears of potential adverse effects on infants associated with psychotropic drug use during lactation, many women with a psychiatric disorder who are treated with a range of psychiatric medications are advised not to breastfeed; or if they choose to breastfeed, they are counseled to avoid taking the essential medication that has made them well. This has been a somewhat intuitive, cautious approach. However, in my 25 years of clinical experience taking care of pregnant and postpartum women with a range of psychiatric disorders, one sad scenario I have frequently witnessed is the woman who decides to defer pharmacologic treatment for severe postpartum psychiatric illness after being counseled to defer treatment given a wish to breastfeed. Those women often have been psychiatrically ill for months while breastfeeding after having decided to defer their own treatment because they do not want to expose the baby to even scant amounts of medication secreted into breast milk associated with use of a needed medicine during lactation.
In a recently published clinical report from the American Academy of Pediatrics committee on drugs, authors suggest that advice not to breastfeed or to uniformly avoid medications while nursing because of possible adverse effects in the infant is often not evidence based and may not be necessary in many cases. The committee states that most drugs do not pose a risk to the mother or infant who is nursing, and that "the benefits of breastfeeding outweigh the risks of exposure to most therapeutic agents via human breast milk" (Pediatrics 2013;132:e796-e809).
The report points out that for certain drugs, a careful risk-benefit analysis is needed, especially for drugs that are concentrated in human milk, those with unproven benefits, and those with long half-lives. Importantly, the report notes say that decisions about the use of medications during lactation have to be made on a case-by-case basis. A concrete example would be exercising appropriate vigilance about the use of these medicines in premature infants with immature metabolism.
The report, published on-line in Pediatrics in August 2013, includes a section on antidepressants, anxiolytics, and antipsychotics. As a resource for clinicians, the report highlights LactMed, part of the National Library of Medicine’s toxicology data network (TOXNET), which provides real-time updated scientific information on the excretion of drugs into breast milk.
The report makes the important distinction regarding the range of clinical decisions that get made in the context of different clinical situations. For example, at our center, patients frequently present with questions about whether to use psychiatric medications while breastfeeding when these women have already been taking the medication during pregnancy for treatment of underlying psychiatric disorder. Others make queries about introduction of pharmacologic therapy in the early postpartum period in the context, for example, of new-onset postpartum depression. Specifically, a woman with a history of psychiatric disorder who is treated with antidepressant during pregnancy may continue that medication across the postpartum period to attenuate risk for postpartum depression, particularly if she has a history of recurrent disease, or depressive relapse when medication has been discontinued. That is clinical scenario differs from that of a woman who develops new onset of depression during the postpartum period.
One part of the AAP report addresses use of certain psychiatric medications in the context of available information from the literature regarding extent of excretion of these medications into breast milk. This section states that many antianxiety drugs, antidepressants, and mood stabilizers are excreted in low concentrations into human milk, with the estimated infant doses under 2% of the weight-adjusted maternal dose. However, the authors also cite small series or case reports where infant plasma levels of some drugs were reported to exceed 10% of maternal plasma concentrations. They list 13 such drugs, which include selective serotonin reuptake inhibitors (SSRIs), antipsychotics, anxiolytics, and mood stabilizers. It is important to keep in mind that the number of these cases is small and represent a very small proportion of cases, when the total denominator of reports in the literature of psychotropic drug use during lactation is considered. For example, olanzapine, a second generation antipsychotic is highlighted as an agent of concern based on one case report (J. Psychopharmacol. 2010;24:121-3).
The take-home message for the clinician is that all psychotropics are excreted into breast milk, even if quantification of the agents in breast milk or infant plasma reveals relatively scant concentration (J. Clin. Psychiatry 2003;64:73-80). If mom takes the medicine coincident with lactation, baby is exposed. At our center, we are usually reluctant to discontinue a medication such as an atypical antipsychotic to treat bipolar disorder in the postpartum period even if the mom chooses to breastfeed considering the extent to which women with bipolar disorder are at a high risk for relapse during the puerperium.
Ironically, we probably have more information regarding the excretion of antidepressants and drugs such as lamotrigine, used as a mood stabilizer, into breast milk than most medicines women take during the postpartum period, with data over the past 15 years suggesting that these medications, like other medications, are excreted into breast milk and are present in infant plasma in extremely sparse concentrations. It is noteworthy that cases of frank newborn toxicity directly associated with mothers who breastfeed on psychiatric medications are extremely few and far between, and are anecdotal at best. For some context, the literature on the effects of SSRI use during pregnancy is vast and prevalence of use of these medications during pregnancy and the postpartum period is substantial; that being said, reports of adverse effects reported in the babies of women who breastfeed while taking an SSRI are scant and thus at least somewhat reassuring.
And yet, consistently, I have witnessed that psychiatric medications are highlighted in the literature as particular agents of concern when it comes to lactation, compared with other medicines, for which only sparse data are available. Whether this reflects a bias about the necessity of treating psychiatric disorders during the postpartum period is unknown. Certainly, the long-term implications for the infant of exposure to low concentrations of psychiatric and nonpsychiatric medications in the context of breastfeeding exposure have yet to be clarified.
Whether a woman treated with a psychiatric medication during the post partum should breastfeed is a prime example of a clinical scenario in which there is no perfect decision, and we need to make the best decision possible, taking into account the available data, and the mother’s psychiatric disorder and her wishes. Some women may be extremely committed to breastfeeding and may choose to breastfeed, acknowledging some of the unknowns regarding these medications during lactation, while other women consider some of the ambiguity associated with the long-term effects of exposure while lactating and may choose not to breastfeed.
It is noteworthy that the AAP committee on drugs concluded the benefits of breastfeeding outweigh the risk of exposure to most therapeutic agents via human milk. And those at our center would certainly suggest that this is the case for psychiatric medications, particularly those used to sustain postpartum maternal psychiatric well-being, which is so critical. As is the case with any clinical decision, and certainly with respect to the use of psychiatric medications during pregnancy and lactation, the decision to treat is contingent on a careful risk-benefit analysis, where the risks of exposure to a medicine is weighed against the risk of untreated psychiatric illness. Even with the well-documented benefits of breastfeeding, nothing should trump the treatment of postpartum psychiatric illness, even if the cost is deferring breastfeeding. Treatment cannot be deferred because of the impact of untreated maternal psychiatric illness on maternal morbidity and on the development of children.
Dr. Cohen is the director of the Center for Women’s Mental Health at Massachusetts General Hospital in Boston, which provides information about reproductive mental health at www.womensmentalhealth.org. To comment, e-mail him at [email protected]. Dr. Cohen has been a consultant to manufacturers of antidepressants and antipsychotic medications.
Over the last 2 decades, abundant data on the reproductive safety of medications used to treat psychiatric disorders have become available, filling in many gaps with respect to our knowledge about the safety of commonly used psychiatric medications during pregnancy. But the availability of such safety data with respect to the use of these agents during breastfeeding is less complete.
Because of fears of potential adverse effects on infants associated with psychotropic drug use during lactation, many women with a psychiatric disorder who are treated with a range of psychiatric medications are advised not to breastfeed; or if they choose to breastfeed, they are counseled to avoid taking the essential medication that has made them well. This has been a somewhat intuitive, cautious approach. However, in my 25 years of clinical experience taking care of pregnant and postpartum women with a range of psychiatric disorders, one sad scenario I have frequently witnessed is the woman who decides to defer pharmacologic treatment for severe postpartum psychiatric illness after being counseled to defer treatment given a wish to breastfeed. Those women often have been psychiatrically ill for months while breastfeeding after having decided to defer their own treatment because they do not want to expose the baby to even scant amounts of medication secreted into breast milk associated with use of a needed medicine during lactation.
In a recently published clinical report from the American Academy of Pediatrics committee on drugs, authors suggest that advice not to breastfeed or to uniformly avoid medications while nursing because of possible adverse effects in the infant is often not evidence based and may not be necessary in many cases. The committee states that most drugs do not pose a risk to the mother or infant who is nursing, and that "the benefits of breastfeeding outweigh the risks of exposure to most therapeutic agents via human breast milk" (Pediatrics 2013;132:e796-e809).
The report points out that for certain drugs, a careful risk-benefit analysis is needed, especially for drugs that are concentrated in human milk, those with unproven benefits, and those with long half-lives. Importantly, the report notes say that decisions about the use of medications during lactation have to be made on a case-by-case basis. A concrete example would be exercising appropriate vigilance about the use of these medicines in premature infants with immature metabolism.
The report, published on-line in Pediatrics in August 2013, includes a section on antidepressants, anxiolytics, and antipsychotics. As a resource for clinicians, the report highlights LactMed, part of the National Library of Medicine’s toxicology data network (TOXNET), which provides real-time updated scientific information on the excretion of drugs into breast milk.
The report makes the important distinction regarding the range of clinical decisions that get made in the context of different clinical situations. For example, at our center, patients frequently present with questions about whether to use psychiatric medications while breastfeeding when these women have already been taking the medication during pregnancy for treatment of underlying psychiatric disorder. Others make queries about introduction of pharmacologic therapy in the early postpartum period in the context, for example, of new-onset postpartum depression. Specifically, a woman with a history of psychiatric disorder who is treated with antidepressant during pregnancy may continue that medication across the postpartum period to attenuate risk for postpartum depression, particularly if she has a history of recurrent disease, or depressive relapse when medication has been discontinued. That is clinical scenario differs from that of a woman who develops new onset of depression during the postpartum period.
One part of the AAP report addresses use of certain psychiatric medications in the context of available information from the literature regarding extent of excretion of these medications into breast milk. This section states that many antianxiety drugs, antidepressants, and mood stabilizers are excreted in low concentrations into human milk, with the estimated infant doses under 2% of the weight-adjusted maternal dose. However, the authors also cite small series or case reports where infant plasma levels of some drugs were reported to exceed 10% of maternal plasma concentrations. They list 13 such drugs, which include selective serotonin reuptake inhibitors (SSRIs), antipsychotics, anxiolytics, and mood stabilizers. It is important to keep in mind that the number of these cases is small and represent a very small proportion of cases, when the total denominator of reports in the literature of psychotropic drug use during lactation is considered. For example, olanzapine, a second generation antipsychotic is highlighted as an agent of concern based on one case report (J. Psychopharmacol. 2010;24:121-3).
The take-home message for the clinician is that all psychotropics are excreted into breast milk, even if quantification of the agents in breast milk or infant plasma reveals relatively scant concentration (J. Clin. Psychiatry 2003;64:73-80). If mom takes the medicine coincident with lactation, baby is exposed. At our center, we are usually reluctant to discontinue a medication such as an atypical antipsychotic to treat bipolar disorder in the postpartum period even if the mom chooses to breastfeed considering the extent to which women with bipolar disorder are at a high risk for relapse during the puerperium.
Ironically, we probably have more information regarding the excretion of antidepressants and drugs such as lamotrigine, used as a mood stabilizer, into breast milk than most medicines women take during the postpartum period, with data over the past 15 years suggesting that these medications, like other medications, are excreted into breast milk and are present in infant plasma in extremely sparse concentrations. It is noteworthy that cases of frank newborn toxicity directly associated with mothers who breastfeed on psychiatric medications are extremely few and far between, and are anecdotal at best. For some context, the literature on the effects of SSRI use during pregnancy is vast and prevalence of use of these medications during pregnancy and the postpartum period is substantial; that being said, reports of adverse effects reported in the babies of women who breastfeed while taking an SSRI are scant and thus at least somewhat reassuring.
And yet, consistently, I have witnessed that psychiatric medications are highlighted in the literature as particular agents of concern when it comes to lactation, compared with other medicines, for which only sparse data are available. Whether this reflects a bias about the necessity of treating psychiatric disorders during the postpartum period is unknown. Certainly, the long-term implications for the infant of exposure to low concentrations of psychiatric and nonpsychiatric medications in the context of breastfeeding exposure have yet to be clarified.
Whether a woman treated with a psychiatric medication during the post partum should breastfeed is a prime example of a clinical scenario in which there is no perfect decision, and we need to make the best decision possible, taking into account the available data, and the mother’s psychiatric disorder and her wishes. Some women may be extremely committed to breastfeeding and may choose to breastfeed, acknowledging some of the unknowns regarding these medications during lactation, while other women consider some of the ambiguity associated with the long-term effects of exposure while lactating and may choose not to breastfeed.
It is noteworthy that the AAP committee on drugs concluded the benefits of breastfeeding outweigh the risk of exposure to most therapeutic agents via human milk. And those at our center would certainly suggest that this is the case for psychiatric medications, particularly those used to sustain postpartum maternal psychiatric well-being, which is so critical. As is the case with any clinical decision, and certainly with respect to the use of psychiatric medications during pregnancy and lactation, the decision to treat is contingent on a careful risk-benefit analysis, where the risks of exposure to a medicine is weighed against the risk of untreated psychiatric illness. Even with the well-documented benefits of breastfeeding, nothing should trump the treatment of postpartum psychiatric illness, even if the cost is deferring breastfeeding. Treatment cannot be deferred because of the impact of untreated maternal psychiatric illness on maternal morbidity and on the development of children.
Dr. Cohen is the director of the Center for Women’s Mental Health at Massachusetts General Hospital in Boston, which provides information about reproductive mental health at www.womensmentalhealth.org. To comment, e-mail him at [email protected]. Dr. Cohen has been a consultant to manufacturers of antidepressants and antipsychotic medications.
Expert provides miscarriage care counseling tips
SAN DIEGO – "It’s not your fault."
Family physicians should relay those words of assurance to patients who present to them with early pregnancy loss, according to Dr. Linda Prine.
"In busy emergency rooms, that’s rarely the kind of thing an ER doc is going to have time to sit down and discuss with the patient," said Dr. Prine, women’s health director of the Institute for Family Health in New York. "They’re trying to make a diagnosis and move the patient on. Here you often have a woman who’s already thinking about what color she’s going to paint the bedroom, or buying a baby crib, or a stroller. This feels like a tragic loss to her. It needs to be explained gently as a natural process and some time taken for understanding. Her partner also needs to hear that it wasn’t her fault or his fault. It wasn’t the sex they had, the snow she shoveled – all of the things that start to go through a woman’s mind to blame herself."
Risk factors for miscarriage include advanced age or very young age, having prior miscarriages, smoking, cocaine use, and fever or infection. Contrary to myth, air travel does not cause miscarriage, she continued, and blunt abdominal trauma, contraceptive use, exercise, the human papillomavirus vaccine, previous abortions, and sexual activity are also not to blame.
Options for miscarriage include expectant management, medication management, and an aspiration procedure. "The patient should be involved in the decision making here," Dr. Prine said at the annual meeting of the American Academy of Family Physicians. "When the women are involved, their mental health outcomes are better. Family physicians are best suited to work through these events with our patients."
The potential risks of expectant management are rare but include infection, the need for emergent uterine aspiration, and bleeding or the need for blood transfusion. The benefits of expectant management include "avoiding the risks, though rare, that can happen with instrumentation such as perforation, introducing infection, or bleeding from a procedure that’s done too vigorously," she noted.
Fever with a tender uterus is a contraindication for expectant management. Incomplete abortions are more likely to have successful expectant management than are fetal demises or anembryonic pregnancies (at day 49, a success rate of 91% vs. 76% and 66%, respectively). "There’s no outside limit for how long you can wait for expectant management," Dr. Prine said. "Usually the woman is not going to want to wait for weeks and weeks, and that becomes the patient-centered limiting factor. How heavy can the bleeding get before she should start to worry about it? If she’s bleeding through two pads an hour for 2 hours back to back, she should call us, though this is rare."
For medical management of an early pregnancy loss, a common protocol involves 800 mcg misoprostol administered vaginally or buccally and repeated in 24 hours if the abortion is incomplete, with vacuum aspiration on day 8 if still incomplete. "Alternative oral regimens cause more GI side effects," Dr. Prine said. Side effects of misoprostol are bleeding, cramping, fever and/or chills, nausea and vomiting, and diarrhea. Misoprostol treatment is safest when used 10 weeks or less after an ultrasound exam confirming the pregnancy loss. "Rule out ectopic pregnancy because the medical treatment for ectopic pregnancy differs from miscarriage treatment," she said. Testing may include an ultrasound, an rh factor screen, hematocrit assessment, and measurement of the serum human chorionic gonadotropin level.
If medical management is not working, "we can repeat the misoprostol again, or she can come into the office for an aspiration procedure," Dr. Prine said.
Sample clinical protocols, patient education sheets, and other resources for providing miscarriage care can be found here.
Dr. Prine said she had no relevant financial conflicts to disclose.
SAN DIEGO – "It’s not your fault."
Family physicians should relay those words of assurance to patients who present to them with early pregnancy loss, according to Dr. Linda Prine.
"In busy emergency rooms, that’s rarely the kind of thing an ER doc is going to have time to sit down and discuss with the patient," said Dr. Prine, women’s health director of the Institute for Family Health in New York. "They’re trying to make a diagnosis and move the patient on. Here you often have a woman who’s already thinking about what color she’s going to paint the bedroom, or buying a baby crib, or a stroller. This feels like a tragic loss to her. It needs to be explained gently as a natural process and some time taken for understanding. Her partner also needs to hear that it wasn’t her fault or his fault. It wasn’t the sex they had, the snow she shoveled – all of the things that start to go through a woman’s mind to blame herself."
Risk factors for miscarriage include advanced age or very young age, having prior miscarriages, smoking, cocaine use, and fever or infection. Contrary to myth, air travel does not cause miscarriage, she continued, and blunt abdominal trauma, contraceptive use, exercise, the human papillomavirus vaccine, previous abortions, and sexual activity are also not to blame.
Options for miscarriage include expectant management, medication management, and an aspiration procedure. "The patient should be involved in the decision making here," Dr. Prine said at the annual meeting of the American Academy of Family Physicians. "When the women are involved, their mental health outcomes are better. Family physicians are best suited to work through these events with our patients."
The potential risks of expectant management are rare but include infection, the need for emergent uterine aspiration, and bleeding or the need for blood transfusion. The benefits of expectant management include "avoiding the risks, though rare, that can happen with instrumentation such as perforation, introducing infection, or bleeding from a procedure that’s done too vigorously," she noted.
Fever with a tender uterus is a contraindication for expectant management. Incomplete abortions are more likely to have successful expectant management than are fetal demises or anembryonic pregnancies (at day 49, a success rate of 91% vs. 76% and 66%, respectively). "There’s no outside limit for how long you can wait for expectant management," Dr. Prine said. "Usually the woman is not going to want to wait for weeks and weeks, and that becomes the patient-centered limiting factor. How heavy can the bleeding get before she should start to worry about it? If she’s bleeding through two pads an hour for 2 hours back to back, she should call us, though this is rare."
For medical management of an early pregnancy loss, a common protocol involves 800 mcg misoprostol administered vaginally or buccally and repeated in 24 hours if the abortion is incomplete, with vacuum aspiration on day 8 if still incomplete. "Alternative oral regimens cause more GI side effects," Dr. Prine said. Side effects of misoprostol are bleeding, cramping, fever and/or chills, nausea and vomiting, and diarrhea. Misoprostol treatment is safest when used 10 weeks or less after an ultrasound exam confirming the pregnancy loss. "Rule out ectopic pregnancy because the medical treatment for ectopic pregnancy differs from miscarriage treatment," she said. Testing may include an ultrasound, an rh factor screen, hematocrit assessment, and measurement of the serum human chorionic gonadotropin level.
If medical management is not working, "we can repeat the misoprostol again, or she can come into the office for an aspiration procedure," Dr. Prine said.
Sample clinical protocols, patient education sheets, and other resources for providing miscarriage care can be found here.
Dr. Prine said she had no relevant financial conflicts to disclose.
SAN DIEGO – "It’s not your fault."
Family physicians should relay those words of assurance to patients who present to them with early pregnancy loss, according to Dr. Linda Prine.
"In busy emergency rooms, that’s rarely the kind of thing an ER doc is going to have time to sit down and discuss with the patient," said Dr. Prine, women’s health director of the Institute for Family Health in New York. "They’re trying to make a diagnosis and move the patient on. Here you often have a woman who’s already thinking about what color she’s going to paint the bedroom, or buying a baby crib, or a stroller. This feels like a tragic loss to her. It needs to be explained gently as a natural process and some time taken for understanding. Her partner also needs to hear that it wasn’t her fault or his fault. It wasn’t the sex they had, the snow she shoveled – all of the things that start to go through a woman’s mind to blame herself."
Risk factors for miscarriage include advanced age or very young age, having prior miscarriages, smoking, cocaine use, and fever or infection. Contrary to myth, air travel does not cause miscarriage, she continued, and blunt abdominal trauma, contraceptive use, exercise, the human papillomavirus vaccine, previous abortions, and sexual activity are also not to blame.
Options for miscarriage include expectant management, medication management, and an aspiration procedure. "The patient should be involved in the decision making here," Dr. Prine said at the annual meeting of the American Academy of Family Physicians. "When the women are involved, their mental health outcomes are better. Family physicians are best suited to work through these events with our patients."
The potential risks of expectant management are rare but include infection, the need for emergent uterine aspiration, and bleeding or the need for blood transfusion. The benefits of expectant management include "avoiding the risks, though rare, that can happen with instrumentation such as perforation, introducing infection, or bleeding from a procedure that’s done too vigorously," she noted.
Fever with a tender uterus is a contraindication for expectant management. Incomplete abortions are more likely to have successful expectant management than are fetal demises or anembryonic pregnancies (at day 49, a success rate of 91% vs. 76% and 66%, respectively). "There’s no outside limit for how long you can wait for expectant management," Dr. Prine said. "Usually the woman is not going to want to wait for weeks and weeks, and that becomes the patient-centered limiting factor. How heavy can the bleeding get before she should start to worry about it? If she’s bleeding through two pads an hour for 2 hours back to back, she should call us, though this is rare."
For medical management of an early pregnancy loss, a common protocol involves 800 mcg misoprostol administered vaginally or buccally and repeated in 24 hours if the abortion is incomplete, with vacuum aspiration on day 8 if still incomplete. "Alternative oral regimens cause more GI side effects," Dr. Prine said. Side effects of misoprostol are bleeding, cramping, fever and/or chills, nausea and vomiting, and diarrhea. Misoprostol treatment is safest when used 10 weeks or less after an ultrasound exam confirming the pregnancy loss. "Rule out ectopic pregnancy because the medical treatment for ectopic pregnancy differs from miscarriage treatment," she said. Testing may include an ultrasound, an rh factor screen, hematocrit assessment, and measurement of the serum human chorionic gonadotropin level.
If medical management is not working, "we can repeat the misoprostol again, or she can come into the office for an aspiration procedure," Dr. Prine said.
Sample clinical protocols, patient education sheets, and other resources for providing miscarriage care can be found here.
Dr. Prine said she had no relevant financial conflicts to disclose.
EXPERT ANALYSIS AT THE AAFP SCIENTIFIC ASSEMBLY
SSRI use in pregnancy doesn’t raise autism risk
A mother’s use of selective serotonin reuptake inhibitors during pregnancy doesn’t appear to raise the risk of autism spectrum disorders in her offspring, according to a report published online Dec. 18 in the New England Journal of Medicine.
In a cohort study of all 626,875 singleton live births that occurred in Denmark between 1996 and 2005, fetal exposure to maternal SSRI use did not significantly increase the risk that the child would be diagnosed as having an autism spectrum disorder during 5-14 years of follow-up, said Dr. Anders Hviid of the department of epidemiology research, Statens Serum Institut, Copenhagen, and his associates.
This large nationwide study had the statistical power to rule out all but a small increase in relative risk, they noted.
Dr. Hviid and his colleagues undertook this study because a recent California case-control study reported that the risk of autism spectrum disorder increased by a factor of two among offspring of women who used antidepressants, particularly SSRIs, during pregnancy, and that this risk was increased by a factor of more than three if the woman used an SSRI during the first trimester (Arch. Gen. Psychiatry 2011;68:1104-12). Evidence from other studies also seemed to bolster this association.
In addition, a causal association is biologically plausible, because people with autism spectrum disorders are known to have increased levels of circulating serotonin, and because serotonin appears to play an important role in early brain development. "Manipulation of serotonin homeostasis can alter neuroanatomical and neurophysiological development and produce enduring behavioral changes in animal models," Dr. Hviid and his associates said.
The investigators analyzed data from the Danish national birth registry, national prescription registry, and national registry of psychiatric diagnoses to assess the 626,875 children, including those who were born to 6,068 mothers who used SSRIs during pregnancy. A total of 3,892 cases of autism spectrum disorder were diagnosed.
In an initial analysis, autism was strongly associated with maternal psychiatric diagnoses and with the use of drugs other than SSRIs during pregnancy. This suggested possible confounding by indication, meaning that any association found between SSRI use and autism might be due to the mother’s underlying psychiatric disorder rather than to the medication she took for it.
During 42,400 person-years of follow-up, there were 52 cases of autism spectrum disorder among the offspring of women who took SSRIs during pregnancy. "In unadjusted analyses, we did find a significantly increased risk of autism spectrum disorder in association with the use of SSRIs during pregnancy.
"In fully adjusted analyses, however, the risk was no longer significant. This was primarily due to adjustment for a number of psychiatric diagnoses [in the mother], which is consistent with the presence of confounding by indication in the unadjusted analysis," the investigators said (N. Engl. J. Med. 2013;369:2406-15 [doi:10.1056/NEJMoa1301449]).
In a further analysis, the researchers found that SSRI use before but not during pregnancy was associated with an increased risk of subsequent autism spectrum disorders in the offspring, similarly suggesting that any association arose from the mother’s underlying psychiatric disorder rather than from the medication.
A sensitivity analysis restricted to the 574,020 pregnancies for which complete information was available yielded the same results as the main analysis: SSRI use during pregnancy did not raise the risk of autism spectrum disorders in the offspring.
One of the strengths of this study was that it was large enough to include 52 cases of autism spectrum disorder, which is more than three times the number of cases assessed in previous studies of this issue.
However, this study was limited in that the prevalence of SSRI use during pregnancy was only 0.97% in this cohort, whereas the prevalence in the United States is estimated to be 5.6%. Similarly, the prevalence of autism spectrum disorders in this cohort was only 0.62%, while it is estimated to be 1.14% in the United States "Thus, our findings may not be generalizable to other countries," Dr. Hviid and his associates said.
This study was funded by the Danish Health and Medicines Authority. No financial conflicts of interest were reported.
A mother’s use of selective serotonin reuptake inhibitors during pregnancy doesn’t appear to raise the risk of autism spectrum disorders in her offspring, according to a report published online Dec. 18 in the New England Journal of Medicine.
In a cohort study of all 626,875 singleton live births that occurred in Denmark between 1996 and 2005, fetal exposure to maternal SSRI use did not significantly increase the risk that the child would be diagnosed as having an autism spectrum disorder during 5-14 years of follow-up, said Dr. Anders Hviid of the department of epidemiology research, Statens Serum Institut, Copenhagen, and his associates.
This large nationwide study had the statistical power to rule out all but a small increase in relative risk, they noted.
Dr. Hviid and his colleagues undertook this study because a recent California case-control study reported that the risk of autism spectrum disorder increased by a factor of two among offspring of women who used antidepressants, particularly SSRIs, during pregnancy, and that this risk was increased by a factor of more than three if the woman used an SSRI during the first trimester (Arch. Gen. Psychiatry 2011;68:1104-12). Evidence from other studies also seemed to bolster this association.
In addition, a causal association is biologically plausible, because people with autism spectrum disorders are known to have increased levels of circulating serotonin, and because serotonin appears to play an important role in early brain development. "Manipulation of serotonin homeostasis can alter neuroanatomical and neurophysiological development and produce enduring behavioral changes in animal models," Dr. Hviid and his associates said.
The investigators analyzed data from the Danish national birth registry, national prescription registry, and national registry of psychiatric diagnoses to assess the 626,875 children, including those who were born to 6,068 mothers who used SSRIs during pregnancy. A total of 3,892 cases of autism spectrum disorder were diagnosed.
In an initial analysis, autism was strongly associated with maternal psychiatric diagnoses and with the use of drugs other than SSRIs during pregnancy. This suggested possible confounding by indication, meaning that any association found between SSRI use and autism might be due to the mother’s underlying psychiatric disorder rather than to the medication she took for it.
During 42,400 person-years of follow-up, there were 52 cases of autism spectrum disorder among the offspring of women who took SSRIs during pregnancy. "In unadjusted analyses, we did find a significantly increased risk of autism spectrum disorder in association with the use of SSRIs during pregnancy.
"In fully adjusted analyses, however, the risk was no longer significant. This was primarily due to adjustment for a number of psychiatric diagnoses [in the mother], which is consistent with the presence of confounding by indication in the unadjusted analysis," the investigators said (N. Engl. J. Med. 2013;369:2406-15 [doi:10.1056/NEJMoa1301449]).
In a further analysis, the researchers found that SSRI use before but not during pregnancy was associated with an increased risk of subsequent autism spectrum disorders in the offspring, similarly suggesting that any association arose from the mother’s underlying psychiatric disorder rather than from the medication.
A sensitivity analysis restricted to the 574,020 pregnancies for which complete information was available yielded the same results as the main analysis: SSRI use during pregnancy did not raise the risk of autism spectrum disorders in the offspring.
One of the strengths of this study was that it was large enough to include 52 cases of autism spectrum disorder, which is more than three times the number of cases assessed in previous studies of this issue.
However, this study was limited in that the prevalence of SSRI use during pregnancy was only 0.97% in this cohort, whereas the prevalence in the United States is estimated to be 5.6%. Similarly, the prevalence of autism spectrum disorders in this cohort was only 0.62%, while it is estimated to be 1.14% in the United States "Thus, our findings may not be generalizable to other countries," Dr. Hviid and his associates said.
This study was funded by the Danish Health and Medicines Authority. No financial conflicts of interest were reported.
A mother’s use of selective serotonin reuptake inhibitors during pregnancy doesn’t appear to raise the risk of autism spectrum disorders in her offspring, according to a report published online Dec. 18 in the New England Journal of Medicine.
In a cohort study of all 626,875 singleton live births that occurred in Denmark between 1996 and 2005, fetal exposure to maternal SSRI use did not significantly increase the risk that the child would be diagnosed as having an autism spectrum disorder during 5-14 years of follow-up, said Dr. Anders Hviid of the department of epidemiology research, Statens Serum Institut, Copenhagen, and his associates.
This large nationwide study had the statistical power to rule out all but a small increase in relative risk, they noted.
Dr. Hviid and his colleagues undertook this study because a recent California case-control study reported that the risk of autism spectrum disorder increased by a factor of two among offspring of women who used antidepressants, particularly SSRIs, during pregnancy, and that this risk was increased by a factor of more than three if the woman used an SSRI during the first trimester (Arch. Gen. Psychiatry 2011;68:1104-12). Evidence from other studies also seemed to bolster this association.
In addition, a causal association is biologically plausible, because people with autism spectrum disorders are known to have increased levels of circulating serotonin, and because serotonin appears to play an important role in early brain development. "Manipulation of serotonin homeostasis can alter neuroanatomical and neurophysiological development and produce enduring behavioral changes in animal models," Dr. Hviid and his associates said.
The investigators analyzed data from the Danish national birth registry, national prescription registry, and national registry of psychiatric diagnoses to assess the 626,875 children, including those who were born to 6,068 mothers who used SSRIs during pregnancy. A total of 3,892 cases of autism spectrum disorder were diagnosed.
In an initial analysis, autism was strongly associated with maternal psychiatric diagnoses and with the use of drugs other than SSRIs during pregnancy. This suggested possible confounding by indication, meaning that any association found between SSRI use and autism might be due to the mother’s underlying psychiatric disorder rather than to the medication she took for it.
During 42,400 person-years of follow-up, there were 52 cases of autism spectrum disorder among the offspring of women who took SSRIs during pregnancy. "In unadjusted analyses, we did find a significantly increased risk of autism spectrum disorder in association with the use of SSRIs during pregnancy.
"In fully adjusted analyses, however, the risk was no longer significant. This was primarily due to adjustment for a number of psychiatric diagnoses [in the mother], which is consistent with the presence of confounding by indication in the unadjusted analysis," the investigators said (N. Engl. J. Med. 2013;369:2406-15 [doi:10.1056/NEJMoa1301449]).
In a further analysis, the researchers found that SSRI use before but not during pregnancy was associated with an increased risk of subsequent autism spectrum disorders in the offspring, similarly suggesting that any association arose from the mother’s underlying psychiatric disorder rather than from the medication.
A sensitivity analysis restricted to the 574,020 pregnancies for which complete information was available yielded the same results as the main analysis: SSRI use during pregnancy did not raise the risk of autism spectrum disorders in the offspring.
One of the strengths of this study was that it was large enough to include 52 cases of autism spectrum disorder, which is more than three times the number of cases assessed in previous studies of this issue.
However, this study was limited in that the prevalence of SSRI use during pregnancy was only 0.97% in this cohort, whereas the prevalence in the United States is estimated to be 5.6%. Similarly, the prevalence of autism spectrum disorders in this cohort was only 0.62%, while it is estimated to be 1.14% in the United States "Thus, our findings may not be generalizable to other countries," Dr. Hviid and his associates said.
This study was funded by the Danish Health and Medicines Authority. No financial conflicts of interest were reported.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: During 42,400 person-years of follow-up, 52 children of women taking SSRIs developed autism spectrum disorder, indicating there was no increased risk for autism among exposed children.
Data Source: A cohort study involving 626,875 births in Denmark between 1996 and 2005, in which 6,068 mothers used SSRIs during pregnancy and after which 3,892 of the offspring developed autism spectrum disorder during follow-up.
Disclosures: This study was funded by the Danish Health and Medicines Authority. No financial conflicts of interest were reported.
Developments and issues in prenatal screening and diagnosis
Prenatal assessments for major chromosomal abnormalities have, like a pendulum, swung over the last 50 years between advancements in screening tests and diagnostic procedures.
In the 1960s, screening for advanced maternal age gave way to diagnostic amniocentesis. Maternal serum alpha-fetoprotein screening for neural tube defects came on the scene in the 1970s, and Down syndrome screening and chorionic villus sampling (CVS) followed in the 1980s. Better ultrasound screening markers were used in combination with biochemistry to advance first-trimester screening in the 1990s and 2000s, leading to a significant decline in diagnostic procedures. Now, free fetal DNA measurement, known as noninvasive prenatal screening, or NIPS, has entered the scene. This development, along with advances in the accuracy of diagnostic lab testing through microarray analysis, will soon lead the pendulum to swing back toward more definitive diagnostic procedures that require either CVS or amniocentesis.
Screening tests provide us with odds adjustments, not definitive answers, and are meant for everyone. Diagnostic tests are meant to give us definitive answers, may have risks, and therefore have traditionally been done on "at-risk" patients. Fundamentally, a screening test gives us an impression, while a diagnostic test gives us harsh reality. As always, there will be trade-offs. No approach is perfect, and no one size fits everyone.
Risks beyond Down syndrome
During the prenatal period, patients will often say, "I’m concerned about having a baby with Down syndrome." What they really mean is that they’re concerned about having a baby with a serious problem, and Down syndrome is the name they know.
Serious problems – a Mendelian disorder, a multifactorial disorder, or a major chromosomal abnormality – affect 2%-3% of all births. Less serious chromosomal abnormalities affect 5%-6% of births. Although advanced maternal age is no longer the sole criterion for deciding who should be offered diagnostic testing, age still is a principal factor for risk determination.
At age 35, the chance of having a baby with Down syndrome is 1 in 380, but the chance of having any chromosomal abnormality detectable by karyotype is 1 in 190. For a 30-year-old, the chance of having a baby with any chromosomal abnormality is 1 in 380, and for a 40-year-old, the risk is 1 in 65.
With the first-trimester screening approach that combines maternal serum free beta-human chorionic gonadotropin (free beta-HCG) and pregnancy-associated plasma protein A (PAPP-A) with fetal nuchal translucency measurement, we are able to detect upward of 85% of fetuses with Down syndrome, or trisomy 21. Yet the disorder is only one of a large number of chromosomal abnormalities observed.
In a recent single-center study of more than 20,000 first-trimester screenings, 5.6% were positive for Down syndrome risk. Of those who subsequently had an amniocentesis or CVS, we found 4% had an abnormal karyotype. Interestingly, 40% of the time the abnormality was not Down syndrome, but another chromosomal abnormality. Similar analyses for trisomies 13 and 18 – the other major abnormalities targeted in first-trimester combined screening – yielded similar statistics (Prenat. Diagn. 2013;33:251-6).
All told, of the screen-positive pregnancies found to have an abnormal karyotype, at least 30% had chromosomal abnormalities outside of those for which they were screen positive. Such findings speak to the limitations of screening as opposed to diagnostic testing, and have implications for patient counseling. Patients should be counseled about the possibility of all chromosome abnormalities – not just Down syndrome.
The NIPS rollout
We have known for well over 100 years that fetal cells cross the placental barrier in small numbers, driving the development of what’s currently known as NIPS. The future of NIPS actually lies in an ever-expanding number of disorders, and will eventually end with sequencing the entire genome.
There are two main methods by which NIPS is done. The original and predominant method uses massive parallel shotgun sequencing, known as next-generation sequencing. This method involves whole-genome amplification and collects enormous amounts of information. Investigators are now attempting to direct amplification at the subchromosome level, mimicking some of what microarray analysis can do.
The second approach uses selected probes, or targeted sequencing, to focus on those sections of DNA that are of interest. Although this method may be cheaper in the short run, one drawback is that new probes will need to be created for each new disorder.
Initially, investigators attempted to isolate nucleated fetal cells from the maternal blood and use them for aneuploidy detection. However, a National Institutes of Health–funded fetal-cell isolation study that ended in 2002 reported disappointing results: Fetal-cell isolation methods had low sensitivity and other technological shortcomings. Subsequently, a number of companies attempted to replicate and improve the work, also without much success.
Concurrent with efforts to use fetal-cell isolation to perform NIPS was the discovery, in the late 1990s by Dr. Dennis Lo, upon whose work next-generation sequencing for NIPS is based, of the presence of circulating cell-free fetal DNA in maternal plasma. The concentration of cell-free fetal DNA may be as much as 5%-8% of the total circulating cell-free DNA in maternal plasma, making free fetal DNA a promising source of fetal genetic material for noninvasive prenatal investigation.
The first high-quality trials on the use of free fetal DNA measurement for detection of trisomy 21 were published in the fall of 2011, and demonstrated up to a 99% detection rate for Down syndrome (less for trisomies 18 and 13). Companies subsequently began to manufacture free fetal DNA tests as off-label products, The tests were initially designated as "noninvasive prenatal diagnosis" tools, but experts around the world objected to the "diagnosis" label, and the terminology shifted to "noninvasive prenatal testing" and finally to "noninvasive prenatal screening"– a designation that I believe accurately reflects its current role.
The uptake in utilization of NIPS has been faster than anyone could have predicted: In just over 2 years, several hundred thousand screening tests have been performed. With 98% to 99% sensitivity, NIPS is an excellent screening test for Down syndrome. However, 99% sensitivity does not equate to 99% positive predictive value, that is, the risk that a patient with a positive screen actually has Down syndrome is much lower.
The published studies of NIPS cite false positive rates of 0.2%-1.0%, but this rate will increase as more disorders are screened. Positive predictive value is directly proportional to the underlying risk. For example, a test with 99% sensitivity and 99% specificity (1% false positive rate) means that a 26-year-old woman who has a positive free fetal DNA test actually has as low as an 11% chance of having a baby with Down syndrome. In older women, who have a higher incidence of having a baby with Down syndrome, a positive NIPS result will have a higher positive predictive value of giving birth to a baby with Down syndrome. However, at the current time, NIPS is an excellent screening test for Down syndrome, but it is not ready for universal primary screening in younger women.
Falsely reassuring are the hyped marketing claims in the United States that NIPS "replaces amnio" and eliminates the need for a nuchal translucency (NT) test. When clinicians abandon performing or referring for a high-quality NT measurement, they can miss or significantly delay the diagnosis of twins/zygosity, growth abnormalities and placentation, cardiac defects, and numerous other anomalies. Similarly, when patients who otherwise would have opted to have CVS or amniocentesis forego having the procedure and have NIPS performed instead, they may regret this decision.
The danger that overreliance on screening tests may paradoxically increase the number of births of babies with otherwise detectable problems was raised in a study led by the late Dr. George Henry, an obstetrician-geneticist in Denver. Curious about declining rates of diagnostic testing in his own practice, Dr. Henry examined trends in his state and found that while the utilization of diagnostic tests had plummeted by 70% over about 15 years, the birth rate of babies born with Down syndrome during this time had doubled among mothers over age 35, and stayed the same among mothers under age 35.
A sociologist-researcher examined the trend that Dr. Henry identified and found that the rise in Down syndrome births was not due to an increase in women electing to keep these pregnancies, but to the fact that the abnormality was not detected in the first place (Fetal Diagn. Ther. 2008;23:308-15). The same risk of screening tests replacing definitive diagnosis exists today with the uptake of NIPS.
The impact of microarray
Microarray technologies have been developed over the past decade. The National Institutes of Health study on chromosomal microarray versus karyotyping, published a year ago, is a game changer. The microarray is analogous to a 15-fold magnifying glass on the karyotype. While the smallest piece of a chromosome that can be evaluated by karyotype analysis is about 5 million base pairs, microarray analysis zooms in on about 200,000 base pairs, allowing us to see small genomic deletions and duplications (copy number variants) that we’ve never seen before.
The trial looked at upward of 4,000 women undergoing CVS or amniocentesis at one of 29 centers. Each diagnostic sample was split in two, with standard karyotyping performed on one portion and chromosomal microarray on the other (N. Engl. J. Med. 2012;367:2175-84).
There were several significant findings: One is that almost one-third of patients have a copy variation now known to be benign. Another is that 2.5% of women who had ultrasound-identified anomalies and normal karyotypes had microdeletion/duplications on the microarray that were clearly associated with a known clinical problem. Moreover, another 3.2% had gains and losses of potential clinical significance. As such, close to 6% of women with ultrasound-identified anomalies and a normal karyotype had clinically relevant copy number variants that only the microarray could find.
When microarray analysis was performed in women whose only indication for prenatal diagnosis was advanced age or an abnormal result on Down syndrome screening, as opposed to an ultrasound-detected anomaly, 0.5%, or 1 in 200, were found to have a pathogenic abnormality that otherwise would have been missed. This is significant: Previously, with karyotyping, we quoted patients a minimum of 1/500 risk of finding a clinically relevant chromosomal anomaly even if the combined report suggested much lower risks of Down syndrome. Now, with microarray, that risk is 1/200. Other studies already published or about to be published show this same level of risk determined by microarray.
With any new technological advance, we get our numerators of the problem before our denominators. The first cases published are those in which clinical or laboratory findings are associated with an abnormality. Only then do researchers go back and look at cases with those findings to test these associations – and only then are the markers sometimes found not to be associated. It will take a number of years to acquire a sizable database on microarray-detected copy number variants. In the end, microarray may help us to explain many of the approximately 1% of serious problems that we have been unable to diagnose until now.
Current decision-making
Ultimately, the future lies with routine, complete genomic sequencing that provides a detailed view of the fetal genome. This is likely to be about 7-10 years away, and the main question on the table is whether it will be performed invasively or with a maternal blood sample.
Today, when a 35-year-old woman comes into my office early in her pregnancy, I will tell her that the risk of having a baby with a chromosomal abnormality is 1 in 190. If she wants to know more, I will explain that the second-trimester quad screen will detect 60% of Down syndrome cases, that the first-trimester combined screen can identify 85% or more, and that the free fetal DNA test will get closer to 99%.
Despite the significant advances in screening, all of these options are still the fundamental equivalent of a Gallup poll. If the patient wants a definitive answer, she will need either an amniocentesis or a CVS, which, in experienced hands, have been shown through an increasing number of studies to be of equal risk. Because there is no such thing as "no" risk when it comes to prenatal diagnostic testing, the question that each patient must answer is, "Where do you want to put that risk – in the test or gambling on the outcome?"
We also have a great cultural divide in the United States. Some people want to know everything, others want to know nothing. Our affluent patients are getting older. Our poorer patients are getting younger. Some people will pay whatever it takes to get the answers they want, while others can or will not pay a dime beyond what their insurance will cover.
There is no one algorithm that can handle these two extremes of patients. Right now, many programs around the country have seen a diminishing number of patients having diagnostic testing – a phenomenon I believe is the result of a false sense of confidence, of people being lulled by the faulty argument that screening protocols can find Down syndrome, so what else is there to know?
Ultimately, a model for younger women would start with "contingent screening," in which patients could start with first-trimester combined screening and move straight to CVS if found to be at "high risk," and end testing if found to be at "low risk." Women who fall in the middle could undergo either free fetal DNA analysis or CVS, and we are developing methods to improve the mathematical processing of data to improve the sensitivity and specificity of all screening programs.
In my program, CVS and microarray are now offered to all patients regardless of age, as the risk that a microarray will find a significant abnormality is at least 1/200, which is the risk we have been quoting to 35-year-old patients for nearly 50 years.
Dr. Evans is president of the Fetal Medicine Foundation and International Fetal Medicine and Surgery Society Foundation, and professor of obstetrics and gynecology at the Icahn School of Medicine at Mount Sinai, New York. Dr. Evans disclosed that he is a consultant for PerkinElmer, a genetics company based in Waltham, Mass.
Prenatal assessments for major chromosomal abnormalities have, like a pendulum, swung over the last 50 years between advancements in screening tests and diagnostic procedures.
In the 1960s, screening for advanced maternal age gave way to diagnostic amniocentesis. Maternal serum alpha-fetoprotein screening for neural tube defects came on the scene in the 1970s, and Down syndrome screening and chorionic villus sampling (CVS) followed in the 1980s. Better ultrasound screening markers were used in combination with biochemistry to advance first-trimester screening in the 1990s and 2000s, leading to a significant decline in diagnostic procedures. Now, free fetal DNA measurement, known as noninvasive prenatal screening, or NIPS, has entered the scene. This development, along with advances in the accuracy of diagnostic lab testing through microarray analysis, will soon lead the pendulum to swing back toward more definitive diagnostic procedures that require either CVS or amniocentesis.
Screening tests provide us with odds adjustments, not definitive answers, and are meant for everyone. Diagnostic tests are meant to give us definitive answers, may have risks, and therefore have traditionally been done on "at-risk" patients. Fundamentally, a screening test gives us an impression, while a diagnostic test gives us harsh reality. As always, there will be trade-offs. No approach is perfect, and no one size fits everyone.
Risks beyond Down syndrome
During the prenatal period, patients will often say, "I’m concerned about having a baby with Down syndrome." What they really mean is that they’re concerned about having a baby with a serious problem, and Down syndrome is the name they know.
Serious problems – a Mendelian disorder, a multifactorial disorder, or a major chromosomal abnormality – affect 2%-3% of all births. Less serious chromosomal abnormalities affect 5%-6% of births. Although advanced maternal age is no longer the sole criterion for deciding who should be offered diagnostic testing, age still is a principal factor for risk determination.
At age 35, the chance of having a baby with Down syndrome is 1 in 380, but the chance of having any chromosomal abnormality detectable by karyotype is 1 in 190. For a 30-year-old, the chance of having a baby with any chromosomal abnormality is 1 in 380, and for a 40-year-old, the risk is 1 in 65.
With the first-trimester screening approach that combines maternal serum free beta-human chorionic gonadotropin (free beta-HCG) and pregnancy-associated plasma protein A (PAPP-A) with fetal nuchal translucency measurement, we are able to detect upward of 85% of fetuses with Down syndrome, or trisomy 21. Yet the disorder is only one of a large number of chromosomal abnormalities observed.
In a recent single-center study of more than 20,000 first-trimester screenings, 5.6% were positive for Down syndrome risk. Of those who subsequently had an amniocentesis or CVS, we found 4% had an abnormal karyotype. Interestingly, 40% of the time the abnormality was not Down syndrome, but another chromosomal abnormality. Similar analyses for trisomies 13 and 18 – the other major abnormalities targeted in first-trimester combined screening – yielded similar statistics (Prenat. Diagn. 2013;33:251-6).
All told, of the screen-positive pregnancies found to have an abnormal karyotype, at least 30% had chromosomal abnormalities outside of those for which they were screen positive. Such findings speak to the limitations of screening as opposed to diagnostic testing, and have implications for patient counseling. Patients should be counseled about the possibility of all chromosome abnormalities – not just Down syndrome.
The NIPS rollout
We have known for well over 100 years that fetal cells cross the placental barrier in small numbers, driving the development of what’s currently known as NIPS. The future of NIPS actually lies in an ever-expanding number of disorders, and will eventually end with sequencing the entire genome.
There are two main methods by which NIPS is done. The original and predominant method uses massive parallel shotgun sequencing, known as next-generation sequencing. This method involves whole-genome amplification and collects enormous amounts of information. Investigators are now attempting to direct amplification at the subchromosome level, mimicking some of what microarray analysis can do.
The second approach uses selected probes, or targeted sequencing, to focus on those sections of DNA that are of interest. Although this method may be cheaper in the short run, one drawback is that new probes will need to be created for each new disorder.
Initially, investigators attempted to isolate nucleated fetal cells from the maternal blood and use them for aneuploidy detection. However, a National Institutes of Health–funded fetal-cell isolation study that ended in 2002 reported disappointing results: Fetal-cell isolation methods had low sensitivity and other technological shortcomings. Subsequently, a number of companies attempted to replicate and improve the work, also without much success.
Concurrent with efforts to use fetal-cell isolation to perform NIPS was the discovery, in the late 1990s by Dr. Dennis Lo, upon whose work next-generation sequencing for NIPS is based, of the presence of circulating cell-free fetal DNA in maternal plasma. The concentration of cell-free fetal DNA may be as much as 5%-8% of the total circulating cell-free DNA in maternal plasma, making free fetal DNA a promising source of fetal genetic material for noninvasive prenatal investigation.
The first high-quality trials on the use of free fetal DNA measurement for detection of trisomy 21 were published in the fall of 2011, and demonstrated up to a 99% detection rate for Down syndrome (less for trisomies 18 and 13). Companies subsequently began to manufacture free fetal DNA tests as off-label products, The tests were initially designated as "noninvasive prenatal diagnosis" tools, but experts around the world objected to the "diagnosis" label, and the terminology shifted to "noninvasive prenatal testing" and finally to "noninvasive prenatal screening"– a designation that I believe accurately reflects its current role.
The uptake in utilization of NIPS has been faster than anyone could have predicted: In just over 2 years, several hundred thousand screening tests have been performed. With 98% to 99% sensitivity, NIPS is an excellent screening test for Down syndrome. However, 99% sensitivity does not equate to 99% positive predictive value, that is, the risk that a patient with a positive screen actually has Down syndrome is much lower.
The published studies of NIPS cite false positive rates of 0.2%-1.0%, but this rate will increase as more disorders are screened. Positive predictive value is directly proportional to the underlying risk. For example, a test with 99% sensitivity and 99% specificity (1% false positive rate) means that a 26-year-old woman who has a positive free fetal DNA test actually has as low as an 11% chance of having a baby with Down syndrome. In older women, who have a higher incidence of having a baby with Down syndrome, a positive NIPS result will have a higher positive predictive value of giving birth to a baby with Down syndrome. However, at the current time, NIPS is an excellent screening test for Down syndrome, but it is not ready for universal primary screening in younger women.
Falsely reassuring are the hyped marketing claims in the United States that NIPS "replaces amnio" and eliminates the need for a nuchal translucency (NT) test. When clinicians abandon performing or referring for a high-quality NT measurement, they can miss or significantly delay the diagnosis of twins/zygosity, growth abnormalities and placentation, cardiac defects, and numerous other anomalies. Similarly, when patients who otherwise would have opted to have CVS or amniocentesis forego having the procedure and have NIPS performed instead, they may regret this decision.
The danger that overreliance on screening tests may paradoxically increase the number of births of babies with otherwise detectable problems was raised in a study led by the late Dr. George Henry, an obstetrician-geneticist in Denver. Curious about declining rates of diagnostic testing in his own practice, Dr. Henry examined trends in his state and found that while the utilization of diagnostic tests had plummeted by 70% over about 15 years, the birth rate of babies born with Down syndrome during this time had doubled among mothers over age 35, and stayed the same among mothers under age 35.
A sociologist-researcher examined the trend that Dr. Henry identified and found that the rise in Down syndrome births was not due to an increase in women electing to keep these pregnancies, but to the fact that the abnormality was not detected in the first place (Fetal Diagn. Ther. 2008;23:308-15). The same risk of screening tests replacing definitive diagnosis exists today with the uptake of NIPS.
The impact of microarray
Microarray technologies have been developed over the past decade. The National Institutes of Health study on chromosomal microarray versus karyotyping, published a year ago, is a game changer. The microarray is analogous to a 15-fold magnifying glass on the karyotype. While the smallest piece of a chromosome that can be evaluated by karyotype analysis is about 5 million base pairs, microarray analysis zooms in on about 200,000 base pairs, allowing us to see small genomic deletions and duplications (copy number variants) that we’ve never seen before.
The trial looked at upward of 4,000 women undergoing CVS or amniocentesis at one of 29 centers. Each diagnostic sample was split in two, with standard karyotyping performed on one portion and chromosomal microarray on the other (N. Engl. J. Med. 2012;367:2175-84).
There were several significant findings: One is that almost one-third of patients have a copy variation now known to be benign. Another is that 2.5% of women who had ultrasound-identified anomalies and normal karyotypes had microdeletion/duplications on the microarray that were clearly associated with a known clinical problem. Moreover, another 3.2% had gains and losses of potential clinical significance. As such, close to 6% of women with ultrasound-identified anomalies and a normal karyotype had clinically relevant copy number variants that only the microarray could find.
When microarray analysis was performed in women whose only indication for prenatal diagnosis was advanced age or an abnormal result on Down syndrome screening, as opposed to an ultrasound-detected anomaly, 0.5%, or 1 in 200, were found to have a pathogenic abnormality that otherwise would have been missed. This is significant: Previously, with karyotyping, we quoted patients a minimum of 1/500 risk of finding a clinically relevant chromosomal anomaly even if the combined report suggested much lower risks of Down syndrome. Now, with microarray, that risk is 1/200. Other studies already published or about to be published show this same level of risk determined by microarray.
With any new technological advance, we get our numerators of the problem before our denominators. The first cases published are those in which clinical or laboratory findings are associated with an abnormality. Only then do researchers go back and look at cases with those findings to test these associations – and only then are the markers sometimes found not to be associated. It will take a number of years to acquire a sizable database on microarray-detected copy number variants. In the end, microarray may help us to explain many of the approximately 1% of serious problems that we have been unable to diagnose until now.
Current decision-making
Ultimately, the future lies with routine, complete genomic sequencing that provides a detailed view of the fetal genome. This is likely to be about 7-10 years away, and the main question on the table is whether it will be performed invasively or with a maternal blood sample.
Today, when a 35-year-old woman comes into my office early in her pregnancy, I will tell her that the risk of having a baby with a chromosomal abnormality is 1 in 190. If she wants to know more, I will explain that the second-trimester quad screen will detect 60% of Down syndrome cases, that the first-trimester combined screen can identify 85% or more, and that the free fetal DNA test will get closer to 99%.
Despite the significant advances in screening, all of these options are still the fundamental equivalent of a Gallup poll. If the patient wants a definitive answer, she will need either an amniocentesis or a CVS, which, in experienced hands, have been shown through an increasing number of studies to be of equal risk. Because there is no such thing as "no" risk when it comes to prenatal diagnostic testing, the question that each patient must answer is, "Where do you want to put that risk – in the test or gambling on the outcome?"
We also have a great cultural divide in the United States. Some people want to know everything, others want to know nothing. Our affluent patients are getting older. Our poorer patients are getting younger. Some people will pay whatever it takes to get the answers they want, while others can or will not pay a dime beyond what their insurance will cover.
There is no one algorithm that can handle these two extremes of patients. Right now, many programs around the country have seen a diminishing number of patients having diagnostic testing – a phenomenon I believe is the result of a false sense of confidence, of people being lulled by the faulty argument that screening protocols can find Down syndrome, so what else is there to know?
Ultimately, a model for younger women would start with "contingent screening," in which patients could start with first-trimester combined screening and move straight to CVS if found to be at "high risk," and end testing if found to be at "low risk." Women who fall in the middle could undergo either free fetal DNA analysis or CVS, and we are developing methods to improve the mathematical processing of data to improve the sensitivity and specificity of all screening programs.
In my program, CVS and microarray are now offered to all patients regardless of age, as the risk that a microarray will find a significant abnormality is at least 1/200, which is the risk we have been quoting to 35-year-old patients for nearly 50 years.
Dr. Evans is president of the Fetal Medicine Foundation and International Fetal Medicine and Surgery Society Foundation, and professor of obstetrics and gynecology at the Icahn School of Medicine at Mount Sinai, New York. Dr. Evans disclosed that he is a consultant for PerkinElmer, a genetics company based in Waltham, Mass.
Prenatal assessments for major chromosomal abnormalities have, like a pendulum, swung over the last 50 years between advancements in screening tests and diagnostic procedures.
In the 1960s, screening for advanced maternal age gave way to diagnostic amniocentesis. Maternal serum alpha-fetoprotein screening for neural tube defects came on the scene in the 1970s, and Down syndrome screening and chorionic villus sampling (CVS) followed in the 1980s. Better ultrasound screening markers were used in combination with biochemistry to advance first-trimester screening in the 1990s and 2000s, leading to a significant decline in diagnostic procedures. Now, free fetal DNA measurement, known as noninvasive prenatal screening, or NIPS, has entered the scene. This development, along with advances in the accuracy of diagnostic lab testing through microarray analysis, will soon lead the pendulum to swing back toward more definitive diagnostic procedures that require either CVS or amniocentesis.
Screening tests provide us with odds adjustments, not definitive answers, and are meant for everyone. Diagnostic tests are meant to give us definitive answers, may have risks, and therefore have traditionally been done on "at-risk" patients. Fundamentally, a screening test gives us an impression, while a diagnostic test gives us harsh reality. As always, there will be trade-offs. No approach is perfect, and no one size fits everyone.
Risks beyond Down syndrome
During the prenatal period, patients will often say, "I’m concerned about having a baby with Down syndrome." What they really mean is that they’re concerned about having a baby with a serious problem, and Down syndrome is the name they know.
Serious problems – a Mendelian disorder, a multifactorial disorder, or a major chromosomal abnormality – affect 2%-3% of all births. Less serious chromosomal abnormalities affect 5%-6% of births. Although advanced maternal age is no longer the sole criterion for deciding who should be offered diagnostic testing, age still is a principal factor for risk determination.
At age 35, the chance of having a baby with Down syndrome is 1 in 380, but the chance of having any chromosomal abnormality detectable by karyotype is 1 in 190. For a 30-year-old, the chance of having a baby with any chromosomal abnormality is 1 in 380, and for a 40-year-old, the risk is 1 in 65.
With the first-trimester screening approach that combines maternal serum free beta-human chorionic gonadotropin (free beta-HCG) and pregnancy-associated plasma protein A (PAPP-A) with fetal nuchal translucency measurement, we are able to detect upward of 85% of fetuses with Down syndrome, or trisomy 21. Yet the disorder is only one of a large number of chromosomal abnormalities observed.
In a recent single-center study of more than 20,000 first-trimester screenings, 5.6% were positive for Down syndrome risk. Of those who subsequently had an amniocentesis or CVS, we found 4% had an abnormal karyotype. Interestingly, 40% of the time the abnormality was not Down syndrome, but another chromosomal abnormality. Similar analyses for trisomies 13 and 18 – the other major abnormalities targeted in first-trimester combined screening – yielded similar statistics (Prenat. Diagn. 2013;33:251-6).
All told, of the screen-positive pregnancies found to have an abnormal karyotype, at least 30% had chromosomal abnormalities outside of those for which they were screen positive. Such findings speak to the limitations of screening as opposed to diagnostic testing, and have implications for patient counseling. Patients should be counseled about the possibility of all chromosome abnormalities – not just Down syndrome.
The NIPS rollout
We have known for well over 100 years that fetal cells cross the placental barrier in small numbers, driving the development of what’s currently known as NIPS. The future of NIPS actually lies in an ever-expanding number of disorders, and will eventually end with sequencing the entire genome.
There are two main methods by which NIPS is done. The original and predominant method uses massive parallel shotgun sequencing, known as next-generation sequencing. This method involves whole-genome amplification and collects enormous amounts of information. Investigators are now attempting to direct amplification at the subchromosome level, mimicking some of what microarray analysis can do.
The second approach uses selected probes, or targeted sequencing, to focus on those sections of DNA that are of interest. Although this method may be cheaper in the short run, one drawback is that new probes will need to be created for each new disorder.
Initially, investigators attempted to isolate nucleated fetal cells from the maternal blood and use them for aneuploidy detection. However, a National Institutes of Health–funded fetal-cell isolation study that ended in 2002 reported disappointing results: Fetal-cell isolation methods had low sensitivity and other technological shortcomings. Subsequently, a number of companies attempted to replicate and improve the work, also without much success.
Concurrent with efforts to use fetal-cell isolation to perform NIPS was the discovery, in the late 1990s by Dr. Dennis Lo, upon whose work next-generation sequencing for NIPS is based, of the presence of circulating cell-free fetal DNA in maternal plasma. The concentration of cell-free fetal DNA may be as much as 5%-8% of the total circulating cell-free DNA in maternal plasma, making free fetal DNA a promising source of fetal genetic material for noninvasive prenatal investigation.
The first high-quality trials on the use of free fetal DNA measurement for detection of trisomy 21 were published in the fall of 2011, and demonstrated up to a 99% detection rate for Down syndrome (less for trisomies 18 and 13). Companies subsequently began to manufacture free fetal DNA tests as off-label products, The tests were initially designated as "noninvasive prenatal diagnosis" tools, but experts around the world objected to the "diagnosis" label, and the terminology shifted to "noninvasive prenatal testing" and finally to "noninvasive prenatal screening"– a designation that I believe accurately reflects its current role.
The uptake in utilization of NIPS has been faster than anyone could have predicted: In just over 2 years, several hundred thousand screening tests have been performed. With 98% to 99% sensitivity, NIPS is an excellent screening test for Down syndrome. However, 99% sensitivity does not equate to 99% positive predictive value, that is, the risk that a patient with a positive screen actually has Down syndrome is much lower.
The published studies of NIPS cite false positive rates of 0.2%-1.0%, but this rate will increase as more disorders are screened. Positive predictive value is directly proportional to the underlying risk. For example, a test with 99% sensitivity and 99% specificity (1% false positive rate) means that a 26-year-old woman who has a positive free fetal DNA test actually has as low as an 11% chance of having a baby with Down syndrome. In older women, who have a higher incidence of having a baby with Down syndrome, a positive NIPS result will have a higher positive predictive value of giving birth to a baby with Down syndrome. However, at the current time, NIPS is an excellent screening test for Down syndrome, but it is not ready for universal primary screening in younger women.
Falsely reassuring are the hyped marketing claims in the United States that NIPS "replaces amnio" and eliminates the need for a nuchal translucency (NT) test. When clinicians abandon performing or referring for a high-quality NT measurement, they can miss or significantly delay the diagnosis of twins/zygosity, growth abnormalities and placentation, cardiac defects, and numerous other anomalies. Similarly, when patients who otherwise would have opted to have CVS or amniocentesis forego having the procedure and have NIPS performed instead, they may regret this decision.
The danger that overreliance on screening tests may paradoxically increase the number of births of babies with otherwise detectable problems was raised in a study led by the late Dr. George Henry, an obstetrician-geneticist in Denver. Curious about declining rates of diagnostic testing in his own practice, Dr. Henry examined trends in his state and found that while the utilization of diagnostic tests had plummeted by 70% over about 15 years, the birth rate of babies born with Down syndrome during this time had doubled among mothers over age 35, and stayed the same among mothers under age 35.
A sociologist-researcher examined the trend that Dr. Henry identified and found that the rise in Down syndrome births was not due to an increase in women electing to keep these pregnancies, but to the fact that the abnormality was not detected in the first place (Fetal Diagn. Ther. 2008;23:308-15). The same risk of screening tests replacing definitive diagnosis exists today with the uptake of NIPS.
The impact of microarray
Microarray technologies have been developed over the past decade. The National Institutes of Health study on chromosomal microarray versus karyotyping, published a year ago, is a game changer. The microarray is analogous to a 15-fold magnifying glass on the karyotype. While the smallest piece of a chromosome that can be evaluated by karyotype analysis is about 5 million base pairs, microarray analysis zooms in on about 200,000 base pairs, allowing us to see small genomic deletions and duplications (copy number variants) that we’ve never seen before.
The trial looked at upward of 4,000 women undergoing CVS or amniocentesis at one of 29 centers. Each diagnostic sample was split in two, with standard karyotyping performed on one portion and chromosomal microarray on the other (N. Engl. J. Med. 2012;367:2175-84).
There were several significant findings: One is that almost one-third of patients have a copy variation now known to be benign. Another is that 2.5% of women who had ultrasound-identified anomalies and normal karyotypes had microdeletion/duplications on the microarray that were clearly associated with a known clinical problem. Moreover, another 3.2% had gains and losses of potential clinical significance. As such, close to 6% of women with ultrasound-identified anomalies and a normal karyotype had clinically relevant copy number variants that only the microarray could find.
When microarray analysis was performed in women whose only indication for prenatal diagnosis was advanced age or an abnormal result on Down syndrome screening, as opposed to an ultrasound-detected anomaly, 0.5%, or 1 in 200, were found to have a pathogenic abnormality that otherwise would have been missed. This is significant: Previously, with karyotyping, we quoted patients a minimum of 1/500 risk of finding a clinically relevant chromosomal anomaly even if the combined report suggested much lower risks of Down syndrome. Now, with microarray, that risk is 1/200. Other studies already published or about to be published show this same level of risk determined by microarray.
With any new technological advance, we get our numerators of the problem before our denominators. The first cases published are those in which clinical or laboratory findings are associated with an abnormality. Only then do researchers go back and look at cases with those findings to test these associations – and only then are the markers sometimes found not to be associated. It will take a number of years to acquire a sizable database on microarray-detected copy number variants. In the end, microarray may help us to explain many of the approximately 1% of serious problems that we have been unable to diagnose until now.
Current decision-making
Ultimately, the future lies with routine, complete genomic sequencing that provides a detailed view of the fetal genome. This is likely to be about 7-10 years away, and the main question on the table is whether it will be performed invasively or with a maternal blood sample.
Today, when a 35-year-old woman comes into my office early in her pregnancy, I will tell her that the risk of having a baby with a chromosomal abnormality is 1 in 190. If she wants to know more, I will explain that the second-trimester quad screen will detect 60% of Down syndrome cases, that the first-trimester combined screen can identify 85% or more, and that the free fetal DNA test will get closer to 99%.
Despite the significant advances in screening, all of these options are still the fundamental equivalent of a Gallup poll. If the patient wants a definitive answer, she will need either an amniocentesis or a CVS, which, in experienced hands, have been shown through an increasing number of studies to be of equal risk. Because there is no such thing as "no" risk when it comes to prenatal diagnostic testing, the question that each patient must answer is, "Where do you want to put that risk – in the test or gambling on the outcome?"
We also have a great cultural divide in the United States. Some people want to know everything, others want to know nothing. Our affluent patients are getting older. Our poorer patients are getting younger. Some people will pay whatever it takes to get the answers they want, while others can or will not pay a dime beyond what their insurance will cover.
There is no one algorithm that can handle these two extremes of patients. Right now, many programs around the country have seen a diminishing number of patients having diagnostic testing – a phenomenon I believe is the result of a false sense of confidence, of people being lulled by the faulty argument that screening protocols can find Down syndrome, so what else is there to know?
Ultimately, a model for younger women would start with "contingent screening," in which patients could start with first-trimester combined screening and move straight to CVS if found to be at "high risk," and end testing if found to be at "low risk." Women who fall in the middle could undergo either free fetal DNA analysis or CVS, and we are developing methods to improve the mathematical processing of data to improve the sensitivity and specificity of all screening programs.
In my program, CVS and microarray are now offered to all patients regardless of age, as the risk that a microarray will find a significant abnormality is at least 1/200, which is the risk we have been quoting to 35-year-old patients for nearly 50 years.
Dr. Evans is president of the Fetal Medicine Foundation and International Fetal Medicine and Surgery Society Foundation, and professor of obstetrics and gynecology at the Icahn School of Medicine at Mount Sinai, New York. Dr. Evans disclosed that he is a consultant for PerkinElmer, a genetics company based in Waltham, Mass.
Pregnancy rate continues downward trend to 12-year low in 2009
The lowest pregnancy rate in more than a decade was reported for 2009, largely driven by declines among teenagers and, to a lesser extent, women under age 30 years.
The 2009 rate of 102.1 pregnancies per 1,000 women was 12% lower than the 1990 peak of 115.8 pregnancies per 1,000 women, reported Sally C. Curtin of the Centers for Disease Control and Prevention’s National Center for Health Statistics and her colleagues in the December issue of an NCHS Data Brief.
Continuing a trend of declining pregnancies, especially since 2007, the rate drop occurred across both married and unmarried women, although the rate among women over age 30 continued to climb gradually from 1990 rates. "It has been suggested that the declining economy, beginning in 2007, has likely played a role in the decreased rates for women under age 40," the authors wrote (NCHS Data Brief, December 2013; No. 136:1-7).
The age group with the highest pregnancy rate also shifted slightly: Those aged 25-29 led with 1.69 million pregnancies in 2009, compared with 1.61 million pregnancies among women aged 20-24. In 1990 and 2000, women aged 20-24 had the highest pregnancy rates.
The most substantial declines in pregnancy rates occurred among teenagers. The rate among all teens in 2009 was 37.9/1,000, down 39% from the 1991 peak of 61.8 pregnancies per 1,000 teens. The fall in the teen pregnancy rate has continued through 2012, with an all-time low of 29.4/1,000 teens. Younger teens in particular have contributed to the decline, with the rate among girls aged 15-17 down 53% since 1990. The drop among 18- and 19-year-olds fell by more than a third (36%) since 1990.
Although black and Hispanic teen pregnancy rates remained at least double the rates among white teens, the decline was still seen across all three demographics. White and black teen pregnancy rates fell 50%, and the Hispanic rate fell 40%, between 1990 and 2009. The authors noted that the teen pregnancy rate declines match up with related trends among teens showing shrinking percentages of sexually experienced teens and increasing rates of contraception use at first intercourse.
Along with the drop in pregnancies has been a drop in abortions, a trend that has continued since 1980 to arrive in 2009 at the lowest rate since 1976. The 2009 rate of 18.5 abortions per 1,000 women is 32% lower than the 1990 rate of 27.4/1,000 women.
However, the abortion rates varied greatly between married and unmarried women: The 2009 rate for unmarried women was 28.9/1,000, almost five times greater than the 6.1/1,000 rate among married women. Accordingly, married women’s 2009 birth rate of 85.6 live births per 1,000 women was 72% higher than that of unmarried women, 49.9/1,000 women.
The data are pulled from three federal sources on live births, abortions, and fetal losses. All states report live births to the CDC’s National Center for Health Statistics through the vital statistics cooperative program of the National Vital Statistics System. Most states report abortion data to the CDC’s National Center for Chronic Disease Prevention and Health Promotion, after which the data are adjusted by the Guttmacher Institute to estimate national totals.
The NCHS National Survey of Family Growth provided data for estimates of fetal losses from the 1995, 2002, and 2006-2010 surveys, plus 1988 data used only for adolescent estimates. The report was funded by the Centers for Disease Control and Prevention.
The lowest pregnancy rate in more than a decade was reported for 2009, largely driven by declines among teenagers and, to a lesser extent, women under age 30 years.
The 2009 rate of 102.1 pregnancies per 1,000 women was 12% lower than the 1990 peak of 115.8 pregnancies per 1,000 women, reported Sally C. Curtin of the Centers for Disease Control and Prevention’s National Center for Health Statistics and her colleagues in the December issue of an NCHS Data Brief.
Continuing a trend of declining pregnancies, especially since 2007, the rate drop occurred across both married and unmarried women, although the rate among women over age 30 continued to climb gradually from 1990 rates. "It has been suggested that the declining economy, beginning in 2007, has likely played a role in the decreased rates for women under age 40," the authors wrote (NCHS Data Brief, December 2013; No. 136:1-7).
The age group with the highest pregnancy rate also shifted slightly: Those aged 25-29 led with 1.69 million pregnancies in 2009, compared with 1.61 million pregnancies among women aged 20-24. In 1990 and 2000, women aged 20-24 had the highest pregnancy rates.
The most substantial declines in pregnancy rates occurred among teenagers. The rate among all teens in 2009 was 37.9/1,000, down 39% from the 1991 peak of 61.8 pregnancies per 1,000 teens. The fall in the teen pregnancy rate has continued through 2012, with an all-time low of 29.4/1,000 teens. Younger teens in particular have contributed to the decline, with the rate among girls aged 15-17 down 53% since 1990. The drop among 18- and 19-year-olds fell by more than a third (36%) since 1990.
Although black and Hispanic teen pregnancy rates remained at least double the rates among white teens, the decline was still seen across all three demographics. White and black teen pregnancy rates fell 50%, and the Hispanic rate fell 40%, between 1990 and 2009. The authors noted that the teen pregnancy rate declines match up with related trends among teens showing shrinking percentages of sexually experienced teens and increasing rates of contraception use at first intercourse.
Along with the drop in pregnancies has been a drop in abortions, a trend that has continued since 1980 to arrive in 2009 at the lowest rate since 1976. The 2009 rate of 18.5 abortions per 1,000 women is 32% lower than the 1990 rate of 27.4/1,000 women.
However, the abortion rates varied greatly between married and unmarried women: The 2009 rate for unmarried women was 28.9/1,000, almost five times greater than the 6.1/1,000 rate among married women. Accordingly, married women’s 2009 birth rate of 85.6 live births per 1,000 women was 72% higher than that of unmarried women, 49.9/1,000 women.
The data are pulled from three federal sources on live births, abortions, and fetal losses. All states report live births to the CDC’s National Center for Health Statistics through the vital statistics cooperative program of the National Vital Statistics System. Most states report abortion data to the CDC’s National Center for Chronic Disease Prevention and Health Promotion, after which the data are adjusted by the Guttmacher Institute to estimate national totals.
The NCHS National Survey of Family Growth provided data for estimates of fetal losses from the 1995, 2002, and 2006-2010 surveys, plus 1988 data used only for adolescent estimates. The report was funded by the Centers for Disease Control and Prevention.
The lowest pregnancy rate in more than a decade was reported for 2009, largely driven by declines among teenagers and, to a lesser extent, women under age 30 years.
The 2009 rate of 102.1 pregnancies per 1,000 women was 12% lower than the 1990 peak of 115.8 pregnancies per 1,000 women, reported Sally C. Curtin of the Centers for Disease Control and Prevention’s National Center for Health Statistics and her colleagues in the December issue of an NCHS Data Brief.
Continuing a trend of declining pregnancies, especially since 2007, the rate drop occurred across both married and unmarried women, although the rate among women over age 30 continued to climb gradually from 1990 rates. "It has been suggested that the declining economy, beginning in 2007, has likely played a role in the decreased rates for women under age 40," the authors wrote (NCHS Data Brief, December 2013; No. 136:1-7).
The age group with the highest pregnancy rate also shifted slightly: Those aged 25-29 led with 1.69 million pregnancies in 2009, compared with 1.61 million pregnancies among women aged 20-24. In 1990 and 2000, women aged 20-24 had the highest pregnancy rates.
The most substantial declines in pregnancy rates occurred among teenagers. The rate among all teens in 2009 was 37.9/1,000, down 39% from the 1991 peak of 61.8 pregnancies per 1,000 teens. The fall in the teen pregnancy rate has continued through 2012, with an all-time low of 29.4/1,000 teens. Younger teens in particular have contributed to the decline, with the rate among girls aged 15-17 down 53% since 1990. The drop among 18- and 19-year-olds fell by more than a third (36%) since 1990.
Although black and Hispanic teen pregnancy rates remained at least double the rates among white teens, the decline was still seen across all three demographics. White and black teen pregnancy rates fell 50%, and the Hispanic rate fell 40%, between 1990 and 2009. The authors noted that the teen pregnancy rate declines match up with related trends among teens showing shrinking percentages of sexually experienced teens and increasing rates of contraception use at first intercourse.
Along with the drop in pregnancies has been a drop in abortions, a trend that has continued since 1980 to arrive in 2009 at the lowest rate since 1976. The 2009 rate of 18.5 abortions per 1,000 women is 32% lower than the 1990 rate of 27.4/1,000 women.
However, the abortion rates varied greatly between married and unmarried women: The 2009 rate for unmarried women was 28.9/1,000, almost five times greater than the 6.1/1,000 rate among married women. Accordingly, married women’s 2009 birth rate of 85.6 live births per 1,000 women was 72% higher than that of unmarried women, 49.9/1,000 women.
The data are pulled from three federal sources on live births, abortions, and fetal losses. All states report live births to the CDC’s National Center for Health Statistics through the vital statistics cooperative program of the National Vital Statistics System. Most states report abortion data to the CDC’s National Center for Chronic Disease Prevention and Health Promotion, after which the data are adjusted by the Guttmacher Institute to estimate national totals.
The NCHS National Survey of Family Growth provided data for estimates of fetal losses from the 1995, 2002, and 2006-2010 surveys, plus 1988 data used only for adolescent estimates. The report was funded by the Centers for Disease Control and Prevention.
FROM THE NATIONAL CENTER FOR HEALTH STATISTICS DATA BRIEF
Major Finding: U.S. pregnancy rates hit a 12-year low in 2009, with a rate of 102.1/1,000 women aged 15-44 years, down 12% since 1990’s peak of 115.8/1,000, because of large declines in teen pregnancy rates and lesser declines in pregnancy rates among women under 30.
Data Source: State reports of live births through the CDC’s vital statistics cooperative program of the National Vital Statistics System, abortion surveillance data reported by most states to the CDC’s National Center for Chronic Disease Prevention and Health Promotion, and fetal loss data collected in the 1995, 2002, and 2006-2010 National Surveys of Family Growth.
Disclosures: Funding for the report came from the CDC.
Long-term GDM Risks Remain Susceptible to Intervention
WASHINGTON – Women with a history of gestational diabetes have a sustained long-term risk of developing type 2 diabetes, and this risk continues to be modifiable with either metformin or lifestyle intervention, according to a 10-year follow-up of women who participated in the National Institutes of Health–funded Diabetes Prevention Program trial.
"Even years out from their index pregnancy, women with a history of gestational diabetes are at a significantly increased and sustained risk [compared with women without a GDM history] of developing type 2 [diabetes] ... despite similar baseline metabolic status and despite similar clinical evidence of risk," said Dr. Vanita R. Aroda, who has served as a subinvestigator in the Diabetes Prevention Program (DPP).
Post-hoc analyses of follow-up data from the Diabetes Prevention Program Outcomes Study (DPPOS) show that while lifestyle intervention was similarly effective in reducing risk for all parous women (regardless of whether they had GDM), metformin was effective only in women with a history of GDM, Dr. Aroda reported at the annual meeting of the Diabetes in Pregnancy Study Group of North America.
The original DPP enrolled more than 3,000 individuals at high risk of developing diabetes. Participants were at least 25 years old, were overweight or obese, and had impaired glucose tolerance with fasting plasma glucose levels of 92-125 mg/dL and glucose levels of 140-199 mg/dL 2 hours after a 75-g oral glucose tolerance test.
Approximately 2,100 of the participants were women, 350 of whom had GDM "about 12 years prior," explained Dr. Aroda of Georgetown University, Washington.
Participants were randomized in the main study to placebo, lifestyle intervention, or metformin for approximately 3 years. In women with a history of GDM, lifestyle intervention and metformin reduced the progression to diabetes over this time by 53% and 50%, compared with placebo.
The effects of metformin were greater in these women than in the population as a whole, for which the risk reduction from metformin was 31%. For women without a history of GDM, the risk reduction from metformin was insignificant. Lifestyle intervention – which involved at least 7% weight loss, 150 minutes of activity per week, and a low-fat diet – was similarly effective for all groups.
The long-term DPPOS began after the DPP concluded, with the majority of the original cohort continuing in the follow-up. All participants in the DPPOS received basic lifestyle education at this point, because of its proven benefits. Those originally randomized to the lifestyle group received additional education/support, while those originally assigned to receive metformin or placebo continued in these study groups.
At a mean of 10 years’ follow-up, women with a history of GDM in the placebo group had a 65% higher risk of developing type 2 diabetes compared with women without a history of GDM – despite the fact these women were approximately 9 years younger than those without the GDM history, and an average of 20 years past their index pregnancy, Dr. Aroda said.
Metformin and lifestyle reduced progression to diabetes similarly among those with a GDM history (41% and 35%), compared with placebo. These intervention effects were greater than in those without a GDM history, who saw a 28% risk reduction from lifestyle and an insignificant reduction with metformin.
"For women with a history of GDM, the effectiveness of the initial interventions – lifestyle and metformin – was sustained. There were very nice risk reductions with both," Dr. Aroda said.
The GDM and non-GDM cohorts of women had a similar baseline body mass index and similar fasting and post–oral glucose tolerance test glucose levels. It is not clear why women without prior GDM had a lesser response to metformin, but "it could be that these women had healthier beta-cell function," she said.
Dr. Aroda reported that she receives consulting fees and grant support from Novo Nordisk and Sanofi-Aventis, as well as grant support from Bristol-Myers Squibb, GlaxoSmithKline, Integrium, Takeda Pharmaceuticals, and Boehringer Ingelheim Vetmedica GmbH.
WASHINGTON – Women with a history of gestational diabetes have a sustained long-term risk of developing type 2 diabetes, and this risk continues to be modifiable with either metformin or lifestyle intervention, according to a 10-year follow-up of women who participated in the National Institutes of Health–funded Diabetes Prevention Program trial.
"Even years out from their index pregnancy, women with a history of gestational diabetes are at a significantly increased and sustained risk [compared with women without a GDM history] of developing type 2 [diabetes] ... despite similar baseline metabolic status and despite similar clinical evidence of risk," said Dr. Vanita R. Aroda, who has served as a subinvestigator in the Diabetes Prevention Program (DPP).
Post-hoc analyses of follow-up data from the Diabetes Prevention Program Outcomes Study (DPPOS) show that while lifestyle intervention was similarly effective in reducing risk for all parous women (regardless of whether they had GDM), metformin was effective only in women with a history of GDM, Dr. Aroda reported at the annual meeting of the Diabetes in Pregnancy Study Group of North America.
The original DPP enrolled more than 3,000 individuals at high risk of developing diabetes. Participants were at least 25 years old, were overweight or obese, and had impaired glucose tolerance with fasting plasma glucose levels of 92-125 mg/dL and glucose levels of 140-199 mg/dL 2 hours after a 75-g oral glucose tolerance test.
Approximately 2,100 of the participants were women, 350 of whom had GDM "about 12 years prior," explained Dr. Aroda of Georgetown University, Washington.
Participants were randomized in the main study to placebo, lifestyle intervention, or metformin for approximately 3 years. In women with a history of GDM, lifestyle intervention and metformin reduced the progression to diabetes over this time by 53% and 50%, compared with placebo.
The effects of metformin were greater in these women than in the population as a whole, for which the risk reduction from metformin was 31%. For women without a history of GDM, the risk reduction from metformin was insignificant. Lifestyle intervention – which involved at least 7% weight loss, 150 minutes of activity per week, and a low-fat diet – was similarly effective for all groups.
The long-term DPPOS began after the DPP concluded, with the majority of the original cohort continuing in the follow-up. All participants in the DPPOS received basic lifestyle education at this point, because of its proven benefits. Those originally randomized to the lifestyle group received additional education/support, while those originally assigned to receive metformin or placebo continued in these study groups.
At a mean of 10 years’ follow-up, women with a history of GDM in the placebo group had a 65% higher risk of developing type 2 diabetes compared with women without a history of GDM – despite the fact these women were approximately 9 years younger than those without the GDM history, and an average of 20 years past their index pregnancy, Dr. Aroda said.
Metformin and lifestyle reduced progression to diabetes similarly among those with a GDM history (41% and 35%), compared with placebo. These intervention effects were greater than in those without a GDM history, who saw a 28% risk reduction from lifestyle and an insignificant reduction with metformin.
"For women with a history of GDM, the effectiveness of the initial interventions – lifestyle and metformin – was sustained. There were very nice risk reductions with both," Dr. Aroda said.
The GDM and non-GDM cohorts of women had a similar baseline body mass index and similar fasting and post–oral glucose tolerance test glucose levels. It is not clear why women without prior GDM had a lesser response to metformin, but "it could be that these women had healthier beta-cell function," she said.
Dr. Aroda reported that she receives consulting fees and grant support from Novo Nordisk and Sanofi-Aventis, as well as grant support from Bristol-Myers Squibb, GlaxoSmithKline, Integrium, Takeda Pharmaceuticals, and Boehringer Ingelheim Vetmedica GmbH.
WASHINGTON – Women with a history of gestational diabetes have a sustained long-term risk of developing type 2 diabetes, and this risk continues to be modifiable with either metformin or lifestyle intervention, according to a 10-year follow-up of women who participated in the National Institutes of Health–funded Diabetes Prevention Program trial.
"Even years out from their index pregnancy, women with a history of gestational diabetes are at a significantly increased and sustained risk [compared with women without a GDM history] of developing type 2 [diabetes] ... despite similar baseline metabolic status and despite similar clinical evidence of risk," said Dr. Vanita R. Aroda, who has served as a subinvestigator in the Diabetes Prevention Program (DPP).
Post-hoc analyses of follow-up data from the Diabetes Prevention Program Outcomes Study (DPPOS) show that while lifestyle intervention was similarly effective in reducing risk for all parous women (regardless of whether they had GDM), metformin was effective only in women with a history of GDM, Dr. Aroda reported at the annual meeting of the Diabetes in Pregnancy Study Group of North America.
The original DPP enrolled more than 3,000 individuals at high risk of developing diabetes. Participants were at least 25 years old, were overweight or obese, and had impaired glucose tolerance with fasting plasma glucose levels of 92-125 mg/dL and glucose levels of 140-199 mg/dL 2 hours after a 75-g oral glucose tolerance test.
Approximately 2,100 of the participants were women, 350 of whom had GDM "about 12 years prior," explained Dr. Aroda of Georgetown University, Washington.
Participants were randomized in the main study to placebo, lifestyle intervention, or metformin for approximately 3 years. In women with a history of GDM, lifestyle intervention and metformin reduced the progression to diabetes over this time by 53% and 50%, compared with placebo.
The effects of metformin were greater in these women than in the population as a whole, for which the risk reduction from metformin was 31%. For women without a history of GDM, the risk reduction from metformin was insignificant. Lifestyle intervention – which involved at least 7% weight loss, 150 minutes of activity per week, and a low-fat diet – was similarly effective for all groups.
The long-term DPPOS began after the DPP concluded, with the majority of the original cohort continuing in the follow-up. All participants in the DPPOS received basic lifestyle education at this point, because of its proven benefits. Those originally randomized to the lifestyle group received additional education/support, while those originally assigned to receive metformin or placebo continued in these study groups.
At a mean of 10 years’ follow-up, women with a history of GDM in the placebo group had a 65% higher risk of developing type 2 diabetes compared with women without a history of GDM – despite the fact these women were approximately 9 years younger than those without the GDM history, and an average of 20 years past their index pregnancy, Dr. Aroda said.
Metformin and lifestyle reduced progression to diabetes similarly among those with a GDM history (41% and 35%), compared with placebo. These intervention effects were greater than in those without a GDM history, who saw a 28% risk reduction from lifestyle and an insignificant reduction with metformin.
"For women with a history of GDM, the effectiveness of the initial interventions – lifestyle and metformin – was sustained. There were very nice risk reductions with both," Dr. Aroda said.
The GDM and non-GDM cohorts of women had a similar baseline body mass index and similar fasting and post–oral glucose tolerance test glucose levels. It is not clear why women without prior GDM had a lesser response to metformin, but "it could be that these women had healthier beta-cell function," she said.
Dr. Aroda reported that she receives consulting fees and grant support from Novo Nordisk and Sanofi-Aventis, as well as grant support from Bristol-Myers Squibb, GlaxoSmithKline, Integrium, Takeda Pharmaceuticals, and Boehringer Ingelheim Vetmedica GmbH.
FROM THE ANNUAL MEETING OF THE DIABETES IN PREGNANCY STUDY GROUP
Long-term GDM risks remain susceptible to intervention
WASHINGTON – Women with a history of gestational diabetes have a sustained long-term risk of developing type 2 diabetes, and this risk continues to be modifiable with either metformin or lifestyle intervention, according to a 10-year follow-up of women who participated in the National Institutes of Health–funded Diabetes Prevention Program trial.
"Even years out from their index pregnancy, women with a history of gestational diabetes are at a significantly increased and sustained risk [compared with women without a GDM history] of developing type 2 [diabetes] ... despite similar baseline metabolic status and despite similar clinical evidence of risk," said Dr. Vanita R. Aroda, who has served as a subinvestigator in the Diabetes Prevention Program (DPP).
Post-hoc analyses of follow-up data from the Diabetes Prevention Program Outcomes Study (DPPOS) show that while lifestyle intervention was similarly effective in reducing risk for all parous women (regardless of whether they had GDM), metformin was effective only in women with a history of GDM, Dr. Aroda reported at the annual meeting of the Diabetes in Pregnancy Study Group of North America.
The original DPP enrolled more than 3,000 individuals at high risk of developing diabetes. Participants were at least 25 years old, were overweight or obese, and had impaired glucose tolerance with fasting plasma glucose levels of 92-125 mg/dL and glucose levels of 140-199 mg/dL 2 hours after a 75-g oral glucose tolerance test.
Approximately 2,100 of the participants were women, 350 of whom had GDM "about 12 years prior," explained Dr. Aroda of Georgetown University, Washington.
Participants were randomized in the main study to placebo, lifestyle intervention, or metformin for approximately 3 years. In women with a history of GDM, lifestyle intervention and metformin reduced the progression to diabetes over this time by 53% and 50%, compared with placebo.
The effects of metformin were greater in these women than in the population as a whole, for which the risk reduction from metformin was 31%. For women without a history of GDM, the risk reduction from metformin was insignificant. Lifestyle intervention – which involved at least 7% weight loss, 150 minutes of activity per week, and a low-fat diet – was similarly effective for all groups.
The long-term DPPOS began after the DPP concluded, with the majority of the original cohort continuing in the follow-up. All participants in the DPPOS received basic lifestyle education at this point, because of its proven benefits. Those originally randomized to the lifestyle group received additional education/support, while those originally assigned to receive metformin or placebo continued in these study groups.
At a mean of 10 years’ follow-up, women with a history of GDM in the placebo group had a 65% higher risk of developing type 2 diabetes compared with women without a history of GDM – despite the fact these women were approximately 9 years younger than those without the GDM history, and an average of 20 years past their index pregnancy, Dr. Aroda said.
Metformin and lifestyle reduced progression to diabetes similarly among those with a GDM history (41% and 35%), compared with placebo. These intervention effects were greater than in those without a GDM history, who saw a 28% risk reduction from lifestyle and an insignificant reduction with metformin.
"For women with a history of GDM, the effectiveness of the initial interventions – lifestyle and metformin – was sustained. There were very nice risk reductions with both," Dr. Aroda said.
The GDM and non-GDM cohorts of women had a similar baseline body mass index and similar fasting and post–oral glucose tolerance test glucose levels. It is not clear why women without prior GDM had a lesser response to metformin, but "it could be that these women had healthier beta-cell function," she said.
Dr. Aroda reported that she receives consulting fees and grant support from Novo Nordisk and Sanofi-Aventis, as well as grant support from Bristol-Myers Squibb, GlaxoSmithKline, Integrium, Takeda Pharmaceuticals, and Boehringer Ingelheim Vetmedica GmbH.
WASHINGTON – Women with a history of gestational diabetes have a sustained long-term risk of developing type 2 diabetes, and this risk continues to be modifiable with either metformin or lifestyle intervention, according to a 10-year follow-up of women who participated in the National Institutes of Health–funded Diabetes Prevention Program trial.
"Even years out from their index pregnancy, women with a history of gestational diabetes are at a significantly increased and sustained risk [compared with women without a GDM history] of developing type 2 [diabetes] ... despite similar baseline metabolic status and despite similar clinical evidence of risk," said Dr. Vanita R. Aroda, who has served as a subinvestigator in the Diabetes Prevention Program (DPP).
Post-hoc analyses of follow-up data from the Diabetes Prevention Program Outcomes Study (DPPOS) show that while lifestyle intervention was similarly effective in reducing risk for all parous women (regardless of whether they had GDM), metformin was effective only in women with a history of GDM, Dr. Aroda reported at the annual meeting of the Diabetes in Pregnancy Study Group of North America.
The original DPP enrolled more than 3,000 individuals at high risk of developing diabetes. Participants were at least 25 years old, were overweight or obese, and had impaired glucose tolerance with fasting plasma glucose levels of 92-125 mg/dL and glucose levels of 140-199 mg/dL 2 hours after a 75-g oral glucose tolerance test.
Approximately 2,100 of the participants were women, 350 of whom had GDM "about 12 years prior," explained Dr. Aroda of Georgetown University, Washington.
Participants were randomized in the main study to placebo, lifestyle intervention, or metformin for approximately 3 years. In women with a history of GDM, lifestyle intervention and metformin reduced the progression to diabetes over this time by 53% and 50%, compared with placebo.
The effects of metformin were greater in these women than in the population as a whole, for which the risk reduction from metformin was 31%. For women without a history of GDM, the risk reduction from metformin was insignificant. Lifestyle intervention – which involved at least 7% weight loss, 150 minutes of activity per week, and a low-fat diet – was similarly effective for all groups.
The long-term DPPOS began after the DPP concluded, with the majority of the original cohort continuing in the follow-up. All participants in the DPPOS received basic lifestyle education at this point, because of its proven benefits. Those originally randomized to the lifestyle group received additional education/support, while those originally assigned to receive metformin or placebo continued in these study groups.
At a mean of 10 years’ follow-up, women with a history of GDM in the placebo group had a 65% higher risk of developing type 2 diabetes compared with women without a history of GDM – despite the fact these women were approximately 9 years younger than those without the GDM history, and an average of 20 years past their index pregnancy, Dr. Aroda said.
Metformin and lifestyle reduced progression to diabetes similarly among those with a GDM history (41% and 35%), compared with placebo. These intervention effects were greater than in those without a GDM history, who saw a 28% risk reduction from lifestyle and an insignificant reduction with metformin.
"For women with a history of GDM, the effectiveness of the initial interventions – lifestyle and metformin – was sustained. There were very nice risk reductions with both," Dr. Aroda said.
The GDM and non-GDM cohorts of women had a similar baseline body mass index and similar fasting and post–oral glucose tolerance test glucose levels. It is not clear why women without prior GDM had a lesser response to metformin, but "it could be that these women had healthier beta-cell function," she said.
Dr. Aroda reported that she receives consulting fees and grant support from Novo Nordisk and Sanofi-Aventis, as well as grant support from Bristol-Myers Squibb, GlaxoSmithKline, Integrium, Takeda Pharmaceuticals, and Boehringer Ingelheim Vetmedica GmbH.
WASHINGTON – Women with a history of gestational diabetes have a sustained long-term risk of developing type 2 diabetes, and this risk continues to be modifiable with either metformin or lifestyle intervention, according to a 10-year follow-up of women who participated in the National Institutes of Health–funded Diabetes Prevention Program trial.
"Even years out from their index pregnancy, women with a history of gestational diabetes are at a significantly increased and sustained risk [compared with women without a GDM history] of developing type 2 [diabetes] ... despite similar baseline metabolic status and despite similar clinical evidence of risk," said Dr. Vanita R. Aroda, who has served as a subinvestigator in the Diabetes Prevention Program (DPP).
Post-hoc analyses of follow-up data from the Diabetes Prevention Program Outcomes Study (DPPOS) show that while lifestyle intervention was similarly effective in reducing risk for all parous women (regardless of whether they had GDM), metformin was effective only in women with a history of GDM, Dr. Aroda reported at the annual meeting of the Diabetes in Pregnancy Study Group of North America.
The original DPP enrolled more than 3,000 individuals at high risk of developing diabetes. Participants were at least 25 years old, were overweight or obese, and had impaired glucose tolerance with fasting plasma glucose levels of 92-125 mg/dL and glucose levels of 140-199 mg/dL 2 hours after a 75-g oral glucose tolerance test.
Approximately 2,100 of the participants were women, 350 of whom had GDM "about 12 years prior," explained Dr. Aroda of Georgetown University, Washington.
Participants were randomized in the main study to placebo, lifestyle intervention, or metformin for approximately 3 years. In women with a history of GDM, lifestyle intervention and metformin reduced the progression to diabetes over this time by 53% and 50%, compared with placebo.
The effects of metformin were greater in these women than in the population as a whole, for which the risk reduction from metformin was 31%. For women without a history of GDM, the risk reduction from metformin was insignificant. Lifestyle intervention – which involved at least 7% weight loss, 150 minutes of activity per week, and a low-fat diet – was similarly effective for all groups.
The long-term DPPOS began after the DPP concluded, with the majority of the original cohort continuing in the follow-up. All participants in the DPPOS received basic lifestyle education at this point, because of its proven benefits. Those originally randomized to the lifestyle group received additional education/support, while those originally assigned to receive metformin or placebo continued in these study groups.
At a mean of 10 years’ follow-up, women with a history of GDM in the placebo group had a 65% higher risk of developing type 2 diabetes compared with women without a history of GDM – despite the fact these women were approximately 9 years younger than those without the GDM history, and an average of 20 years past their index pregnancy, Dr. Aroda said.
Metformin and lifestyle reduced progression to diabetes similarly among those with a GDM history (41% and 35%), compared with placebo. These intervention effects were greater than in those without a GDM history, who saw a 28% risk reduction from lifestyle and an insignificant reduction with metformin.
"For women with a history of GDM, the effectiveness of the initial interventions – lifestyle and metformin – was sustained. There were very nice risk reductions with both," Dr. Aroda said.
The GDM and non-GDM cohorts of women had a similar baseline body mass index and similar fasting and post–oral glucose tolerance test glucose levels. It is not clear why women without prior GDM had a lesser response to metformin, but "it could be that these women had healthier beta-cell function," she said.
Dr. Aroda reported that she receives consulting fees and grant support from Novo Nordisk and Sanofi-Aventis, as well as grant support from Bristol-Myers Squibb, GlaxoSmithKline, Integrium, Takeda Pharmaceuticals, and Boehringer Ingelheim Vetmedica GmbH.
FROM THE ANNUAL MEETING OF THE DIABETES IN PREGNANCY STUDY GROUP
Major finding: Women with a history of GDM who received no intervention for diabetes prevention had a 65% higher risk of developing diabetes at 10-year follow-up compared with parous women without a history of GDM. Metformin and lifestyle reduced progression to diabetes in women with a GDM history by 41% and 35%, respectively, compared with placebo.
Data source: The Diabetes Prevention Program Outcomes Study, a longitudinal follow-up study of the NIH-funded Diabetes Prevention Program randomized prospective study.
Disclosures: Dr. Aroda reported that she receives consulting fees and grant support from Novo Nordisk and Sanofi-Aventis, as well as grant support from Bristol-Myers Squibb, GlaxoSmithKline, Integrium, Takeda Pharmaceuticals, and Boehringer Ingelheim Vetmedica GmbH.
Preterm birth rates show racial/ethnic disparities
The preterm birth rate for black infants in the United States was lower than ever in 2010, but it was still about 60% higher than the rate for white infants, the Centers for Disease Control and Prevention reported.
Non-Hispanic black infants had a rate of preterm births (occurring before 37 weeks’ gestation) of 17.1% in 2010, a decrease of about 8% from the 18.5% reported in 2006, according to final birth certificate data from the National Vital Statistics System.
The total U.S. preterm birth rate in 2010 was 12.0%, with non-Hispanic whites (10.8%) and Asian/Pacific Islanders (10.7%) below the average and Hispanics (11.8%) and American Indian/Alaska Natives (13.6%) joining blacks above the national figure, the CDC said (MMWR Suppl. 2013;62:136-8).
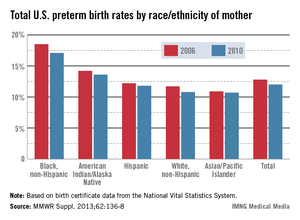
The preterm birth rate for black infants in the United States was lower than ever in 2010, but it was still about 60% higher than the rate for white infants, the Centers for Disease Control and Prevention reported.
Non-Hispanic black infants had a rate of preterm births (occurring before 37 weeks’ gestation) of 17.1% in 2010, a decrease of about 8% from the 18.5% reported in 2006, according to final birth certificate data from the National Vital Statistics System.
The total U.S. preterm birth rate in 2010 was 12.0%, with non-Hispanic whites (10.8%) and Asian/Pacific Islanders (10.7%) below the average and Hispanics (11.8%) and American Indian/Alaska Natives (13.6%) joining blacks above the national figure, the CDC said (MMWR Suppl. 2013;62:136-8).

The preterm birth rate for black infants in the United States was lower than ever in 2010, but it was still about 60% higher than the rate for white infants, the Centers for Disease Control and Prevention reported.
Non-Hispanic black infants had a rate of preterm births (occurring before 37 weeks’ gestation) of 17.1% in 2010, a decrease of about 8% from the 18.5% reported in 2006, according to final birth certificate data from the National Vital Statistics System.
The total U.S. preterm birth rate in 2010 was 12.0%, with non-Hispanic whites (10.8%) and Asian/Pacific Islanders (10.7%) below the average and Hispanics (11.8%) and American Indian/Alaska Natives (13.6%) joining blacks above the national figure, the CDC said (MMWR Suppl. 2013;62:136-8).

FROM MMWR