User login
Does the risk of unplanned pregnancy outweigh the risk of VTE from hormonal contraception?
Let’s increase our use of IUDs and improve contraceptive effectiveness in this country
Robert L. Barbieri, MD (Editorial, August 2012)
Let’s increase our use of implants and DMPA and improve contraceptive effectiveness in this country
Robert L. Barbieri, MD (Editorial, September 2012)
It is well established that combined hormonal contraception increases the risk of venous thromboembolism (VTE), both deep venous thrombosis (DVT) and pulmonary embolism (PE).1 Concerns exist that drospirenone-containing combined oral contraceptives (OCs), the norelgestromin patch, and the etonogestrel vaginal ring may increase the risk of VTE, compared with second-generation OCs, although results from studies evaluating the thromboembolic risk of these products are conflicting.1,2
An April 2012 safety communication from the US Food and Drug Administration (FDA) reported that “drospirenone-containing birth control pills may be associated with a higher risk for blood clots than other progestin-containing pills.”3 These pills now carry revised drug labels stating that epidemiologic studies that compared the risk of VTE reported that the risk ranged from no increase to a three-fold increase.3
Together, these studies and the FDA warning have garnered a lot of publicity and caused confusion and concern, leading both patients and providers to ask, “Are these specific products really safe?”
What is the baseline risk?
For nonusers of hormonal contraception, the baseline risk of VTE is 1 to 5 events per 10,000 woman-years.1,3-5 Variables that increase a woman’s risk of VTE include1:
- advanced age
- obesity
- immobility
- hematologic disorders
- pregnancy.
Estrogen-containing OCs with second-generation progestins (levonorgestrel, norgestimate, and norethindrone) have a risk of VTE of approximately 3 to 9 events per 10,000 woman-years.1,3-5
When study results conflict
The relative risk of VTE associated with drospirenone-containing OCs, compared with second-generation pills, ranges from 0.9 to 3.3. The relative risk is 1.2 to 2.2 for the norelgestromin patch, and 1.6 to 1.9 for the etonogestrel ring.1-4
All of the studies addressing the increased risk of VTE with drospirenone, the patch, and the ring have some limitations, such as the use of retrospective data, selection bias, study design, or inclusion of multiple pill regimens. However, most of the studies that found no association between these methods and VTE were industry-funded.2 Criticisms of these studies have led to disagreement about the risk; it is unclear whether a definitive study ever can be designed and performed.2
Worst-case scenario. Using data from only those studies that show an increased risk of VTE, the increased number of VTE events above that conferred by a second-generation progestin would be approximately2:
- ring: 3–5/10,000 woman-years
- patch: 3–8/10,000 woman-years
- drospirenone pill: 5–10/10,000 woman-years (risk may be highest in the first year of use3).
Adding perspective
The risks of hormonal contraception must be weighed against the consequences of using no contraception: 43 million women in the United States are sexually active but do not wish to become pregnant. Without contraception, 85% will be pregnant within 1 year.6 The risk of mortality during pregnancy in the United States is 1.8 deaths per 10,000 live births (5.5 deaths per 10,000 live births for women older than 39 years).7The prevalence of VTE during pregnancy is 5 to 29 events per 10,000 women; during the postpartum period, the prevalence is 40 to 65 events per 10,000 women (although some quote the VTE risk during the postpartum period to be as high as 200 to 400 events per 10,000 women).3,5,8
An unplanned pregnancy is more likely than a planned pregnancy to have a poor perinatal outcome or to end in abortion. The socioeconomic benefits of planning pregnancies must also be considered.

Hormonal contraception confers benefits beyond the prevention of pregnancy. In addition to a 50% reduction in the rate of endometrial cancer and a 27% reduction in the rate of ovarian cancer (and an even greater reduction for women who take OCs longer than 5 years), there are other benefits to hormonal contraception, such as reduced acne, dysmenorrhea, and menorrhagia.9
Individualize your care
When choosing a method of contraception, it is important not only to consider thromboembolic risk but also:
- previous contraceptive experiences
- previous pregnancies
- patient preference
- efficacy
- individual health factors
- cost.
For instance, even though the risk of VTE may be slightly increased among women using the norelgestromin patch, compliance rates are higher with the patch than with the pill.10 A woman with two unplanned pregnancies while taking the pill who reports having difficulty adhering to a daily regimen is a different patch candidate than a woman who has successfully planned two pregnancies using OCs.
For many women, a weekly or monthly reversible contraceptive is the most desirable method. In addition to these more quantifiable factors, some women prefer a specific brand of pill or delivery method—and satisfaction is a key component of contraception adherence.
Educate your patient
I favor the approach of providing as much data as possible. Patients may read the black box warning in the package inserts for drospirenone-containing pills or the norelgestromin patch, find news sources that inaccurately report risk to garner the most compelling headline, or stumble across plaintiff’s lawyers advertising lawsuits for drospirenone-containing pills, the contraceptive ring, and the patch. I can best counter confusion or misinformation by providing accurate information and putting possible risks into perspective up front. I now explain that the risk for VTE may be higher with certain pills, the ring, and the patch, but there just aren’t enough high-quality data to be certain. I also explain that risk may mean different things for different patients, based on medical history and previous experiences. I have found that my patients appreciate the full disclosure.
Overall, the benefits of combined hormonal contraception with all methods outweigh the risk of VTE. In addition, issues related to switching contraceptive methods may increase the risk of an unplanned pregnancy. In 1995, when the United Kingdom warned that desogestrel pills carried an increased risk of VTE but were still “safe,” the incidence of unplanned pregnancies and abortions increased.2,11 The data regarding the risk of VTE associated with drospirenone, the patch, and the ring should not be an impetus for sweeping generalizations, but rather an opportunity to educate our patients (and ourselves) and to further individualize care.
Do you agree that the benefits of combined hormonal contraception with all methods outweigh the VTE risk? Why or why not? What do you do in your practice? Click here
Click here to find additional articles on contraception published in OBG Management
in 2012.
1. Lidegaard Ø, Milsom I, Geirsson R, Skjeldestad F. Hormonal contraception and venous thromboembolism. Acta Obstet Gynecol Scand. 2012;91(7):769-778.
2. Raymond EG, Burke AE, Espey E. Combined hormonal contraceptives and venous thromboembolism: putting the risks into perspective. Obstet Gynecol. 2012;119(5):1039-1044.
3. US Food and Drug Administration. Safety Announcement. Updated information about the risk of blood clots in women taking birth control pills containing drospirenone. http://www.fda.gov/Drugs/DrugSafety/ucm299305.htm. Published April 10 2012. Accessed September 11, 2012.
4. Lidegaard Ø, Nielsen L, Skovlund C, Løkkegaard E. Venous thrombosis in users of non-oral hormonal contraception: follow-up study Denmark 2001-10 [published online ahead of print May 10, 2012]. BMJ. 2012:344-353:e2990. doi: 10.1136 /bmj.e2990.
5. Oral contraceptives and the risk of thromboembolism: an update. Clinical practice Guideline No. 252. Society of Obstetricians and Gynaecologists of Canada. 2010;252. http://www.sogc.org/guidelines/documents/gui252CPG1012E.pdf. Accessed September 5, 2012.
6. Fact Sheet Contraceptive use in the United States. Guttmacher Institute. http://www.guttmacher.org/pubs/fb_contr_use.html. Published July 2012. Accessed September 5 2012.
7. Berg CJ, Callaghan WM, Syverson C, Henderson Z. Pregnancy related mortality in the United States 1998 to 2005. Obstet Gynecol. 2010;116(6):1302-1309.
8. James A. Committee on Practice Bulletins-Obstetrics. ACOG Practice Bulletin No. 123. Thromboembolism in pregnancy. Obstet Gynecol. 2011;118(3):718-729.
9. Committee on Practice Bulletins-Gynecology. ACOG Practice Bulletin No. 110. Noncontraceptive uses of hormonal contraception. Obstet Gynecol. 2010;115(1):206-218.
10. Archer DF, Bigrigg A, Smallwood GH, Shangold GA, Creasy GW, Fisher AC. Assessment of compliance with a weekly contraceptive patch (OrthoEvra/Evra) among North American women. Fertil Steril. 2002;77(2 suppl 2):S27-S31.
11. Furedi A. The public health implications of the 1995 ‘pill scare.’ Hum Reprod Update. 1999;5(6):621-626.
Let’s increase our use of IUDs and improve contraceptive effectiveness in this country
Robert L. Barbieri, MD (Editorial, August 2012)
Let’s increase our use of implants and DMPA and improve contraceptive effectiveness in this country
Robert L. Barbieri, MD (Editorial, September 2012)
It is well established that combined hormonal contraception increases the risk of venous thromboembolism (VTE), both deep venous thrombosis (DVT) and pulmonary embolism (PE).1 Concerns exist that drospirenone-containing combined oral contraceptives (OCs), the norelgestromin patch, and the etonogestrel vaginal ring may increase the risk of VTE, compared with second-generation OCs, although results from studies evaluating the thromboembolic risk of these products are conflicting.1,2
An April 2012 safety communication from the US Food and Drug Administration (FDA) reported that “drospirenone-containing birth control pills may be associated with a higher risk for blood clots than other progestin-containing pills.”3 These pills now carry revised drug labels stating that epidemiologic studies that compared the risk of VTE reported that the risk ranged from no increase to a three-fold increase.3
Together, these studies and the FDA warning have garnered a lot of publicity and caused confusion and concern, leading both patients and providers to ask, “Are these specific products really safe?”
What is the baseline risk?
For nonusers of hormonal contraception, the baseline risk of VTE is 1 to 5 events per 10,000 woman-years.1,3-5 Variables that increase a woman’s risk of VTE include1:
- advanced age
- obesity
- immobility
- hematologic disorders
- pregnancy.
Estrogen-containing OCs with second-generation progestins (levonorgestrel, norgestimate, and norethindrone) have a risk of VTE of approximately 3 to 9 events per 10,000 woman-years.1,3-5
When study results conflict
The relative risk of VTE associated with drospirenone-containing OCs, compared with second-generation pills, ranges from 0.9 to 3.3. The relative risk is 1.2 to 2.2 for the norelgestromin patch, and 1.6 to 1.9 for the etonogestrel ring.1-4
All of the studies addressing the increased risk of VTE with drospirenone, the patch, and the ring have some limitations, such as the use of retrospective data, selection bias, study design, or inclusion of multiple pill regimens. However, most of the studies that found no association between these methods and VTE were industry-funded.2 Criticisms of these studies have led to disagreement about the risk; it is unclear whether a definitive study ever can be designed and performed.2
Worst-case scenario. Using data from only those studies that show an increased risk of VTE, the increased number of VTE events above that conferred by a second-generation progestin would be approximately2:
- ring: 3–5/10,000 woman-years
- patch: 3–8/10,000 woman-years
- drospirenone pill: 5–10/10,000 woman-years (risk may be highest in the first year of use3).
Adding perspective
The risks of hormonal contraception must be weighed against the consequences of using no contraception: 43 million women in the United States are sexually active but do not wish to become pregnant. Without contraception, 85% will be pregnant within 1 year.6 The risk of mortality during pregnancy in the United States is 1.8 deaths per 10,000 live births (5.5 deaths per 10,000 live births for women older than 39 years).7The prevalence of VTE during pregnancy is 5 to 29 events per 10,000 women; during the postpartum period, the prevalence is 40 to 65 events per 10,000 women (although some quote the VTE risk during the postpartum period to be as high as 200 to 400 events per 10,000 women).3,5,8
An unplanned pregnancy is more likely than a planned pregnancy to have a poor perinatal outcome or to end in abortion. The socioeconomic benefits of planning pregnancies must also be considered.

Hormonal contraception confers benefits beyond the prevention of pregnancy. In addition to a 50% reduction in the rate of endometrial cancer and a 27% reduction in the rate of ovarian cancer (and an even greater reduction for women who take OCs longer than 5 years), there are other benefits to hormonal contraception, such as reduced acne, dysmenorrhea, and menorrhagia.9
Individualize your care
When choosing a method of contraception, it is important not only to consider thromboembolic risk but also:
- previous contraceptive experiences
- previous pregnancies
- patient preference
- efficacy
- individual health factors
- cost.
For instance, even though the risk of VTE may be slightly increased among women using the norelgestromin patch, compliance rates are higher with the patch than with the pill.10 A woman with two unplanned pregnancies while taking the pill who reports having difficulty adhering to a daily regimen is a different patch candidate than a woman who has successfully planned two pregnancies using OCs.
For many women, a weekly or monthly reversible contraceptive is the most desirable method. In addition to these more quantifiable factors, some women prefer a specific brand of pill or delivery method—and satisfaction is a key component of contraception adherence.
Educate your patient
I favor the approach of providing as much data as possible. Patients may read the black box warning in the package inserts for drospirenone-containing pills or the norelgestromin patch, find news sources that inaccurately report risk to garner the most compelling headline, or stumble across plaintiff’s lawyers advertising lawsuits for drospirenone-containing pills, the contraceptive ring, and the patch. I can best counter confusion or misinformation by providing accurate information and putting possible risks into perspective up front. I now explain that the risk for VTE may be higher with certain pills, the ring, and the patch, but there just aren’t enough high-quality data to be certain. I also explain that risk may mean different things for different patients, based on medical history and previous experiences. I have found that my patients appreciate the full disclosure.
Overall, the benefits of combined hormonal contraception with all methods outweigh the risk of VTE. In addition, issues related to switching contraceptive methods may increase the risk of an unplanned pregnancy. In 1995, when the United Kingdom warned that desogestrel pills carried an increased risk of VTE but were still “safe,” the incidence of unplanned pregnancies and abortions increased.2,11 The data regarding the risk of VTE associated with drospirenone, the patch, and the ring should not be an impetus for sweeping generalizations, but rather an opportunity to educate our patients (and ourselves) and to further individualize care.
Do you agree that the benefits of combined hormonal contraception with all methods outweigh the VTE risk? Why or why not? What do you do in your practice? Click here
Click here to find additional articles on contraception published in OBG Management
in 2012.
Let’s increase our use of IUDs and improve contraceptive effectiveness in this country
Robert L. Barbieri, MD (Editorial, August 2012)
Let’s increase our use of implants and DMPA and improve contraceptive effectiveness in this country
Robert L. Barbieri, MD (Editorial, September 2012)
It is well established that combined hormonal contraception increases the risk of venous thromboembolism (VTE), both deep venous thrombosis (DVT) and pulmonary embolism (PE).1 Concerns exist that drospirenone-containing combined oral contraceptives (OCs), the norelgestromin patch, and the etonogestrel vaginal ring may increase the risk of VTE, compared with second-generation OCs, although results from studies evaluating the thromboembolic risk of these products are conflicting.1,2
An April 2012 safety communication from the US Food and Drug Administration (FDA) reported that “drospirenone-containing birth control pills may be associated with a higher risk for blood clots than other progestin-containing pills.”3 These pills now carry revised drug labels stating that epidemiologic studies that compared the risk of VTE reported that the risk ranged from no increase to a three-fold increase.3
Together, these studies and the FDA warning have garnered a lot of publicity and caused confusion and concern, leading both patients and providers to ask, “Are these specific products really safe?”
What is the baseline risk?
For nonusers of hormonal contraception, the baseline risk of VTE is 1 to 5 events per 10,000 woman-years.1,3-5 Variables that increase a woman’s risk of VTE include1:
- advanced age
- obesity
- immobility
- hematologic disorders
- pregnancy.
Estrogen-containing OCs with second-generation progestins (levonorgestrel, norgestimate, and norethindrone) have a risk of VTE of approximately 3 to 9 events per 10,000 woman-years.1,3-5
When study results conflict
The relative risk of VTE associated with drospirenone-containing OCs, compared with second-generation pills, ranges from 0.9 to 3.3. The relative risk is 1.2 to 2.2 for the norelgestromin patch, and 1.6 to 1.9 for the etonogestrel ring.1-4
All of the studies addressing the increased risk of VTE with drospirenone, the patch, and the ring have some limitations, such as the use of retrospective data, selection bias, study design, or inclusion of multiple pill regimens. However, most of the studies that found no association between these methods and VTE were industry-funded.2 Criticisms of these studies have led to disagreement about the risk; it is unclear whether a definitive study ever can be designed and performed.2
Worst-case scenario. Using data from only those studies that show an increased risk of VTE, the increased number of VTE events above that conferred by a second-generation progestin would be approximately2:
- ring: 3–5/10,000 woman-years
- patch: 3–8/10,000 woman-years
- drospirenone pill: 5–10/10,000 woman-years (risk may be highest in the first year of use3).
Adding perspective
The risks of hormonal contraception must be weighed against the consequences of using no contraception: 43 million women in the United States are sexually active but do not wish to become pregnant. Without contraception, 85% will be pregnant within 1 year.6 The risk of mortality during pregnancy in the United States is 1.8 deaths per 10,000 live births (5.5 deaths per 10,000 live births for women older than 39 years).7The prevalence of VTE during pregnancy is 5 to 29 events per 10,000 women; during the postpartum period, the prevalence is 40 to 65 events per 10,000 women (although some quote the VTE risk during the postpartum period to be as high as 200 to 400 events per 10,000 women).3,5,8
An unplanned pregnancy is more likely than a planned pregnancy to have a poor perinatal outcome or to end in abortion. The socioeconomic benefits of planning pregnancies must also be considered.

Hormonal contraception confers benefits beyond the prevention of pregnancy. In addition to a 50% reduction in the rate of endometrial cancer and a 27% reduction in the rate of ovarian cancer (and an even greater reduction for women who take OCs longer than 5 years), there are other benefits to hormonal contraception, such as reduced acne, dysmenorrhea, and menorrhagia.9
Individualize your care
When choosing a method of contraception, it is important not only to consider thromboembolic risk but also:
- previous contraceptive experiences
- previous pregnancies
- patient preference
- efficacy
- individual health factors
- cost.
For instance, even though the risk of VTE may be slightly increased among women using the norelgestromin patch, compliance rates are higher with the patch than with the pill.10 A woman with two unplanned pregnancies while taking the pill who reports having difficulty adhering to a daily regimen is a different patch candidate than a woman who has successfully planned two pregnancies using OCs.
For many women, a weekly or monthly reversible contraceptive is the most desirable method. In addition to these more quantifiable factors, some women prefer a specific brand of pill or delivery method—and satisfaction is a key component of contraception adherence.
Educate your patient
I favor the approach of providing as much data as possible. Patients may read the black box warning in the package inserts for drospirenone-containing pills or the norelgestromin patch, find news sources that inaccurately report risk to garner the most compelling headline, or stumble across plaintiff’s lawyers advertising lawsuits for drospirenone-containing pills, the contraceptive ring, and the patch. I can best counter confusion or misinformation by providing accurate information and putting possible risks into perspective up front. I now explain that the risk for VTE may be higher with certain pills, the ring, and the patch, but there just aren’t enough high-quality data to be certain. I also explain that risk may mean different things for different patients, based on medical history and previous experiences. I have found that my patients appreciate the full disclosure.
Overall, the benefits of combined hormonal contraception with all methods outweigh the risk of VTE. In addition, issues related to switching contraceptive methods may increase the risk of an unplanned pregnancy. In 1995, when the United Kingdom warned that desogestrel pills carried an increased risk of VTE but were still “safe,” the incidence of unplanned pregnancies and abortions increased.2,11 The data regarding the risk of VTE associated with drospirenone, the patch, and the ring should not be an impetus for sweeping generalizations, but rather an opportunity to educate our patients (and ourselves) and to further individualize care.
Do you agree that the benefits of combined hormonal contraception with all methods outweigh the VTE risk? Why or why not? What do you do in your practice? Click here
Click here to find additional articles on contraception published in OBG Management
in 2012.
1. Lidegaard Ø, Milsom I, Geirsson R, Skjeldestad F. Hormonal contraception and venous thromboembolism. Acta Obstet Gynecol Scand. 2012;91(7):769-778.
2. Raymond EG, Burke AE, Espey E. Combined hormonal contraceptives and venous thromboembolism: putting the risks into perspective. Obstet Gynecol. 2012;119(5):1039-1044.
3. US Food and Drug Administration. Safety Announcement. Updated information about the risk of blood clots in women taking birth control pills containing drospirenone. http://www.fda.gov/Drugs/DrugSafety/ucm299305.htm. Published April 10 2012. Accessed September 11, 2012.
4. Lidegaard Ø, Nielsen L, Skovlund C, Løkkegaard E. Venous thrombosis in users of non-oral hormonal contraception: follow-up study Denmark 2001-10 [published online ahead of print May 10, 2012]. BMJ. 2012:344-353:e2990. doi: 10.1136 /bmj.e2990.
5. Oral contraceptives and the risk of thromboembolism: an update. Clinical practice Guideline No. 252. Society of Obstetricians and Gynaecologists of Canada. 2010;252. http://www.sogc.org/guidelines/documents/gui252CPG1012E.pdf. Accessed September 5, 2012.
6. Fact Sheet Contraceptive use in the United States. Guttmacher Institute. http://www.guttmacher.org/pubs/fb_contr_use.html. Published July 2012. Accessed September 5 2012.
7. Berg CJ, Callaghan WM, Syverson C, Henderson Z. Pregnancy related mortality in the United States 1998 to 2005. Obstet Gynecol. 2010;116(6):1302-1309.
8. James A. Committee on Practice Bulletins-Obstetrics. ACOG Practice Bulletin No. 123. Thromboembolism in pregnancy. Obstet Gynecol. 2011;118(3):718-729.
9. Committee on Practice Bulletins-Gynecology. ACOG Practice Bulletin No. 110. Noncontraceptive uses of hormonal contraception. Obstet Gynecol. 2010;115(1):206-218.
10. Archer DF, Bigrigg A, Smallwood GH, Shangold GA, Creasy GW, Fisher AC. Assessment of compliance with a weekly contraceptive patch (OrthoEvra/Evra) among North American women. Fertil Steril. 2002;77(2 suppl 2):S27-S31.
11. Furedi A. The public health implications of the 1995 ‘pill scare.’ Hum Reprod Update. 1999;5(6):621-626.
1. Lidegaard Ø, Milsom I, Geirsson R, Skjeldestad F. Hormonal contraception and venous thromboembolism. Acta Obstet Gynecol Scand. 2012;91(7):769-778.
2. Raymond EG, Burke AE, Espey E. Combined hormonal contraceptives and venous thromboembolism: putting the risks into perspective. Obstet Gynecol. 2012;119(5):1039-1044.
3. US Food and Drug Administration. Safety Announcement. Updated information about the risk of blood clots in women taking birth control pills containing drospirenone. http://www.fda.gov/Drugs/DrugSafety/ucm299305.htm. Published April 10 2012. Accessed September 11, 2012.
4. Lidegaard Ø, Nielsen L, Skovlund C, Løkkegaard E. Venous thrombosis in users of non-oral hormonal contraception: follow-up study Denmark 2001-10 [published online ahead of print May 10, 2012]. BMJ. 2012:344-353:e2990. doi: 10.1136 /bmj.e2990.
5. Oral contraceptives and the risk of thromboembolism: an update. Clinical practice Guideline No. 252. Society of Obstetricians and Gynaecologists of Canada. 2010;252. http://www.sogc.org/guidelines/documents/gui252CPG1012E.pdf. Accessed September 5, 2012.
6. Fact Sheet Contraceptive use in the United States. Guttmacher Institute. http://www.guttmacher.org/pubs/fb_contr_use.html. Published July 2012. Accessed September 5 2012.
7. Berg CJ, Callaghan WM, Syverson C, Henderson Z. Pregnancy related mortality in the United States 1998 to 2005. Obstet Gynecol. 2010;116(6):1302-1309.
8. James A. Committee on Practice Bulletins-Obstetrics. ACOG Practice Bulletin No. 123. Thromboembolism in pregnancy. Obstet Gynecol. 2011;118(3):718-729.
9. Committee on Practice Bulletins-Gynecology. ACOG Practice Bulletin No. 110. Noncontraceptive uses of hormonal contraception. Obstet Gynecol. 2010;115(1):206-218.
10. Archer DF, Bigrigg A, Smallwood GH, Shangold GA, Creasy GW, Fisher AC. Assessment of compliance with a weekly contraceptive patch (OrthoEvra/Evra) among North American women. Fertil Steril. 2002;77(2 suppl 2):S27-S31.
11. Furedi A. The public health implications of the 1995 ‘pill scare.’ Hum Reprod Update. 1999;5(6):621-626.
FDA Approves Oral MS Drug Teriflunomide
The Food and Drug Administration has approved the latest oral therapy for relapsing multiple sclerosis, teriflunomide (Aubagio), but it will carry a strong warning that it increases the risk of fetal harm.
The drug, made by the Genzyme division of Sanofi Aventis, is the second oral tablet approved by the agency for MS. Novartis’ fingolimod (Gilenya) was approved in 2010.
Teriflunomide is the active metabolite of leflunomide, a synthetic, low-molecular-weight drug that was approved by the FDA in 1998 for the treatment of rheumatoid arthritis.
The most common side effects with teriflunomide were diarrhea, abnormal liver tests, nausea, and hair loss, the agency noted in a written statement. The drug will carry a boxed warning on the risk of liver toxicity. Clinicians are being advised to conduct liver function tests before initiating treatment and during therapy.
The warning also will highlight teriflunomide’s increased potential for causing birth defects. Animal studies suggested that the drug is a teratogen. Thus, it is being placed in pregnancy category X, which means that women of childbearing age must have a negative pregnancy test before starting teriflunomide therapy, and also must use effective birth control during treatment.
According to the FDA, the relapse rate for patients taking teriflunomide in trials was 30% lower than for those taking a placebo. Those results were first reported in late 2010 in the Teriflunomide Multiple Sclerosis Oral (TEMSO) trial.
"Multiple sclerosis can impair movement, sensation, and thinking, so it is important to have a variety of treatment options available to patients," Dr. Russell Katz, director of the Division of Neurology Products in the FDA’s Center for Drug Evaluation and Research, said in a written statement.
"We are greatly encouraged to see a new oral therapeutic option become available to people living with MS," Dr. Timothy Coetzee, chief research officer at the National Multiple Sclerosis Society, said in a statement provided by Genzyme.
The neurological disorder is thought to affect more than 350,000 Americans, and 2.5 million people worldwide. Symptoms usually begin between ages 20 and 40, and women are affected twice as often as are men. The relapsing phase of the disease is the most common.
Teriflunomide patients will receive a medication guide that outlines the drug’s potential harms.
The Food and Drug Administration has approved the latest oral therapy for relapsing multiple sclerosis, teriflunomide (Aubagio), but it will carry a strong warning that it increases the risk of fetal harm.
The drug, made by the Genzyme division of Sanofi Aventis, is the second oral tablet approved by the agency for MS. Novartis’ fingolimod (Gilenya) was approved in 2010.
Teriflunomide is the active metabolite of leflunomide, a synthetic, low-molecular-weight drug that was approved by the FDA in 1998 for the treatment of rheumatoid arthritis.
The most common side effects with teriflunomide were diarrhea, abnormal liver tests, nausea, and hair loss, the agency noted in a written statement. The drug will carry a boxed warning on the risk of liver toxicity. Clinicians are being advised to conduct liver function tests before initiating treatment and during therapy.
The warning also will highlight teriflunomide’s increased potential for causing birth defects. Animal studies suggested that the drug is a teratogen. Thus, it is being placed in pregnancy category X, which means that women of childbearing age must have a negative pregnancy test before starting teriflunomide therapy, and also must use effective birth control during treatment.
According to the FDA, the relapse rate for patients taking teriflunomide in trials was 30% lower than for those taking a placebo. Those results were first reported in late 2010 in the Teriflunomide Multiple Sclerosis Oral (TEMSO) trial.
"Multiple sclerosis can impair movement, sensation, and thinking, so it is important to have a variety of treatment options available to patients," Dr. Russell Katz, director of the Division of Neurology Products in the FDA’s Center for Drug Evaluation and Research, said in a written statement.
"We are greatly encouraged to see a new oral therapeutic option become available to people living with MS," Dr. Timothy Coetzee, chief research officer at the National Multiple Sclerosis Society, said in a statement provided by Genzyme.
The neurological disorder is thought to affect more than 350,000 Americans, and 2.5 million people worldwide. Symptoms usually begin between ages 20 and 40, and women are affected twice as often as are men. The relapsing phase of the disease is the most common.
Teriflunomide patients will receive a medication guide that outlines the drug’s potential harms.
The Food and Drug Administration has approved the latest oral therapy for relapsing multiple sclerosis, teriflunomide (Aubagio), but it will carry a strong warning that it increases the risk of fetal harm.
The drug, made by the Genzyme division of Sanofi Aventis, is the second oral tablet approved by the agency for MS. Novartis’ fingolimod (Gilenya) was approved in 2010.
Teriflunomide is the active metabolite of leflunomide, a synthetic, low-molecular-weight drug that was approved by the FDA in 1998 for the treatment of rheumatoid arthritis.
The most common side effects with teriflunomide were diarrhea, abnormal liver tests, nausea, and hair loss, the agency noted in a written statement. The drug will carry a boxed warning on the risk of liver toxicity. Clinicians are being advised to conduct liver function tests before initiating treatment and during therapy.
The warning also will highlight teriflunomide’s increased potential for causing birth defects. Animal studies suggested that the drug is a teratogen. Thus, it is being placed in pregnancy category X, which means that women of childbearing age must have a negative pregnancy test before starting teriflunomide therapy, and also must use effective birth control during treatment.
According to the FDA, the relapse rate for patients taking teriflunomide in trials was 30% lower than for those taking a placebo. Those results were first reported in late 2010 in the Teriflunomide Multiple Sclerosis Oral (TEMSO) trial.
"Multiple sclerosis can impair movement, sensation, and thinking, so it is important to have a variety of treatment options available to patients," Dr. Russell Katz, director of the Division of Neurology Products in the FDA’s Center for Drug Evaluation and Research, said in a written statement.
"We are greatly encouraged to see a new oral therapeutic option become available to people living with MS," Dr. Timothy Coetzee, chief research officer at the National Multiple Sclerosis Society, said in a statement provided by Genzyme.
The neurological disorder is thought to affect more than 350,000 Americans, and 2.5 million people worldwide. Symptoms usually begin between ages 20 and 40, and women are affected twice as often as are men. The relapsing phase of the disease is the most common.
Teriflunomide patients will receive a medication guide that outlines the drug’s potential harms.
Secondary Headaches Flag Medical Conditions in Pregnancy
LOS ANGELES – Secondary headaches in pregnant women are likely to be associated with an additional medical complaint, according to a retrospective study of patients who presented to a tertiary-care center.
A team led by Dr. Matthew S. Robbins of Montefiore Headache Center in the Bronx, N.Y., assessed characteristics of 68 consecutive women who had an inpatient neurologic consultation for acute headache in pregnancy over a 2.5-year period at Montefiore Medical Center.
Secondary headaches were diagnosed in 57% of these women. Women with secondary headaches were more likely to have hypertension and some other chief complaint along with headache. Women with primary headaches were more likely to have a history of migraine and to have photophobia, phonophobia, and lacrimation on exam.
Three cases (4%) were noteworthy in that the headache led to discovery of an unrecognized pregnancy. All of these patients had secondary headache, and life-threatening but treatable medical conditions.
"In the acute care setting, vigilance for secondary headache in the pregnant population should be heightened, particularly in those patients who may have headache in addition to other symptoms, the lack of a migraine history, and elevated blood pressure acutely," Dr. Robbins recommended. "Because of our sad corollary [of three cases], we should ... always consider a pregnancy test in all women presenting with headache during childbearing years."
He acknowledged that a study limitation was the highly selected patient population. "This is not only patients presenting to the acute care setting, but patients who are then referred by the [emergency department] doctors or obstetricians for a neurologic consultation. So we are probably not capturing the routine cases of preeclampsia that are obvious, or other diagnoses that, in our center, the obstetricians don’t feel warrant neurologic consultation," he commented at the annual meeting of the American Headache Society.
Session attendee Dr. Peter J. Goadsby of the University of California, San Francisco, noted that about a fifth of patients with primary headache were classified as having an abnormal neurologic exam. "That wouldn’t be consistent with primary headache, would it? So what’s abnormal mean?" he asked.
"Most of the abnormal exam findings in the primary headache group were sensory abnormalities," such as unilateral facial numbness, which – although documented as abnormal – might not be clinically relevant. "It was up to the discretion of the treating team whether that patient had a primary or secondary diagnosis with the exam and abnormality combined," Dr. Robbins replied.
"Clinical experience, at least ours, suggests that consultation in the acute care setting for headache in pregnant women is not such an uncommon experience," he noted, explaining the study’s rationale. However, "there are very few guidelines that address this. It is mostly review articles in the obstetrics and gynecology literature that are not really validated, and clinical series are not well reported."
On average, the 68 women the investigators studied were 29 years old, and they were predominantly Hispanic (43%) and black (41%). The mean gestational age was 28.6 weeks, with 60% of women in their third trimester.
In terms of diagnoses, made via the ICHD-II (International Classification of Headache Disorders, second edition) system, 57% of the women had secondary headache and 43% had primary headache.
Headache class was predominantly migraine (43%), preeclampsia or eclampsia (25%), intercurrent infection (7%), and pituitary adenoma/apoplexy (6%).
The patients with primary headache and the patients with secondary headache were statistically indistinguishable in terms of most demographic, pregnancy, and clinical characteristics, according to Dr. Robbins.
But patients in the secondary headache group were less likely to have a history of migraine (39% vs. 90%; P less than .0001) and, on exam, photophobia (67% vs. 93%; P = .02), phonophobia (36% vs. 79%; P = .0004), and lacrimation (0% vs. 17%; P = .01).
On the flip side, those in the secondary headache group were more likely to have headache plus some other chief complaint such as seizure, shortness of breath, or visual disturbances (54% vs. 14%; P = .0009) and hypertension (49% vs. 0%; P less than .0001).
Of the three cases in which headache led to the discovery of an unrecognized pregnancy, one had diagnoses of PRES (posterior reversible encephalopathy syndrome) and eclampsia; one had diagnoses of PRES, RCVS (reversible cerebral vasoconstriction syndrome), and eclampsia; and one had diagnoses of hyperkalemia and renal failure in systemic lupus erythematous.
Dr. Robbins disclosed no relevant conflicts of interest.
LOS ANGELES – Secondary headaches in pregnant women are likely to be associated with an additional medical complaint, according to a retrospective study of patients who presented to a tertiary-care center.
A team led by Dr. Matthew S. Robbins of Montefiore Headache Center in the Bronx, N.Y., assessed characteristics of 68 consecutive women who had an inpatient neurologic consultation for acute headache in pregnancy over a 2.5-year period at Montefiore Medical Center.
Secondary headaches were diagnosed in 57% of these women. Women with secondary headaches were more likely to have hypertension and some other chief complaint along with headache. Women with primary headaches were more likely to have a history of migraine and to have photophobia, phonophobia, and lacrimation on exam.
Three cases (4%) were noteworthy in that the headache led to discovery of an unrecognized pregnancy. All of these patients had secondary headache, and life-threatening but treatable medical conditions.
"In the acute care setting, vigilance for secondary headache in the pregnant population should be heightened, particularly in those patients who may have headache in addition to other symptoms, the lack of a migraine history, and elevated blood pressure acutely," Dr. Robbins recommended. "Because of our sad corollary [of three cases], we should ... always consider a pregnancy test in all women presenting with headache during childbearing years."
He acknowledged that a study limitation was the highly selected patient population. "This is not only patients presenting to the acute care setting, but patients who are then referred by the [emergency department] doctors or obstetricians for a neurologic consultation. So we are probably not capturing the routine cases of preeclampsia that are obvious, or other diagnoses that, in our center, the obstetricians don’t feel warrant neurologic consultation," he commented at the annual meeting of the American Headache Society.
Session attendee Dr. Peter J. Goadsby of the University of California, San Francisco, noted that about a fifth of patients with primary headache were classified as having an abnormal neurologic exam. "That wouldn’t be consistent with primary headache, would it? So what’s abnormal mean?" he asked.
"Most of the abnormal exam findings in the primary headache group were sensory abnormalities," such as unilateral facial numbness, which – although documented as abnormal – might not be clinically relevant. "It was up to the discretion of the treating team whether that patient had a primary or secondary diagnosis with the exam and abnormality combined," Dr. Robbins replied.
"Clinical experience, at least ours, suggests that consultation in the acute care setting for headache in pregnant women is not such an uncommon experience," he noted, explaining the study’s rationale. However, "there are very few guidelines that address this. It is mostly review articles in the obstetrics and gynecology literature that are not really validated, and clinical series are not well reported."
On average, the 68 women the investigators studied were 29 years old, and they were predominantly Hispanic (43%) and black (41%). The mean gestational age was 28.6 weeks, with 60% of women in their third trimester.
In terms of diagnoses, made via the ICHD-II (International Classification of Headache Disorders, second edition) system, 57% of the women had secondary headache and 43% had primary headache.
Headache class was predominantly migraine (43%), preeclampsia or eclampsia (25%), intercurrent infection (7%), and pituitary adenoma/apoplexy (6%).
The patients with primary headache and the patients with secondary headache were statistically indistinguishable in terms of most demographic, pregnancy, and clinical characteristics, according to Dr. Robbins.
But patients in the secondary headache group were less likely to have a history of migraine (39% vs. 90%; P less than .0001) and, on exam, photophobia (67% vs. 93%; P = .02), phonophobia (36% vs. 79%; P = .0004), and lacrimation (0% vs. 17%; P = .01).
On the flip side, those in the secondary headache group were more likely to have headache plus some other chief complaint such as seizure, shortness of breath, or visual disturbances (54% vs. 14%; P = .0009) and hypertension (49% vs. 0%; P less than .0001).
Of the three cases in which headache led to the discovery of an unrecognized pregnancy, one had diagnoses of PRES (posterior reversible encephalopathy syndrome) and eclampsia; one had diagnoses of PRES, RCVS (reversible cerebral vasoconstriction syndrome), and eclampsia; and one had diagnoses of hyperkalemia and renal failure in systemic lupus erythematous.
Dr. Robbins disclosed no relevant conflicts of interest.
LOS ANGELES – Secondary headaches in pregnant women are likely to be associated with an additional medical complaint, according to a retrospective study of patients who presented to a tertiary-care center.
A team led by Dr. Matthew S. Robbins of Montefiore Headache Center in the Bronx, N.Y., assessed characteristics of 68 consecutive women who had an inpatient neurologic consultation for acute headache in pregnancy over a 2.5-year period at Montefiore Medical Center.
Secondary headaches were diagnosed in 57% of these women. Women with secondary headaches were more likely to have hypertension and some other chief complaint along with headache. Women with primary headaches were more likely to have a history of migraine and to have photophobia, phonophobia, and lacrimation on exam.
Three cases (4%) were noteworthy in that the headache led to discovery of an unrecognized pregnancy. All of these patients had secondary headache, and life-threatening but treatable medical conditions.
"In the acute care setting, vigilance for secondary headache in the pregnant population should be heightened, particularly in those patients who may have headache in addition to other symptoms, the lack of a migraine history, and elevated blood pressure acutely," Dr. Robbins recommended. "Because of our sad corollary [of three cases], we should ... always consider a pregnancy test in all women presenting with headache during childbearing years."
He acknowledged that a study limitation was the highly selected patient population. "This is not only patients presenting to the acute care setting, but patients who are then referred by the [emergency department] doctors or obstetricians for a neurologic consultation. So we are probably not capturing the routine cases of preeclampsia that are obvious, or other diagnoses that, in our center, the obstetricians don’t feel warrant neurologic consultation," he commented at the annual meeting of the American Headache Society.
Session attendee Dr. Peter J. Goadsby of the University of California, San Francisco, noted that about a fifth of patients with primary headache were classified as having an abnormal neurologic exam. "That wouldn’t be consistent with primary headache, would it? So what’s abnormal mean?" he asked.
"Most of the abnormal exam findings in the primary headache group were sensory abnormalities," such as unilateral facial numbness, which – although documented as abnormal – might not be clinically relevant. "It was up to the discretion of the treating team whether that patient had a primary or secondary diagnosis with the exam and abnormality combined," Dr. Robbins replied.
"Clinical experience, at least ours, suggests that consultation in the acute care setting for headache in pregnant women is not such an uncommon experience," he noted, explaining the study’s rationale. However, "there are very few guidelines that address this. It is mostly review articles in the obstetrics and gynecology literature that are not really validated, and clinical series are not well reported."
On average, the 68 women the investigators studied were 29 years old, and they were predominantly Hispanic (43%) and black (41%). The mean gestational age was 28.6 weeks, with 60% of women in their third trimester.
In terms of diagnoses, made via the ICHD-II (International Classification of Headache Disorders, second edition) system, 57% of the women had secondary headache and 43% had primary headache.
Headache class was predominantly migraine (43%), preeclampsia or eclampsia (25%), intercurrent infection (7%), and pituitary adenoma/apoplexy (6%).
The patients with primary headache and the patients with secondary headache were statistically indistinguishable in terms of most demographic, pregnancy, and clinical characteristics, according to Dr. Robbins.
But patients in the secondary headache group were less likely to have a history of migraine (39% vs. 90%; P less than .0001) and, on exam, photophobia (67% vs. 93%; P = .02), phonophobia (36% vs. 79%; P = .0004), and lacrimation (0% vs. 17%; P = .01).
On the flip side, those in the secondary headache group were more likely to have headache plus some other chief complaint such as seizure, shortness of breath, or visual disturbances (54% vs. 14%; P = .0009) and hypertension (49% vs. 0%; P less than .0001).
Of the three cases in which headache led to the discovery of an unrecognized pregnancy, one had diagnoses of PRES (posterior reversible encephalopathy syndrome) and eclampsia; one had diagnoses of PRES, RCVS (reversible cerebral vasoconstriction syndrome), and eclampsia; and one had diagnoses of hyperkalemia and renal failure in systemic lupus erythematous.
Dr. Robbins disclosed no relevant conflicts of interest.
AT THE ANNUAL MEETING OF THE AMERICAN HEADACHE SOCIETY
Major Finding: Secondary headaches were seen in 57% of 68 consecutive women who had an inpatient neurologic consultation for acute headache in pregnancy.
Data Source: Researchers conducted a retrospective study of 68 women who were referred for inpatient neurologic consultation because of acute headache in pregnancy.
Disclosures: Dr. Robbins disclosed no relevant conflicts of interest.
Pregnant women taking antihypertensives—some shown to cause fetal risk—in increasing numbers
New research in the American Heart Association journal Hypertension indicates that almost 5% of pregnant women are prescribed antihypertensives, including some drugs that are contraindicated for mothers or their babies.
“Use of high blood pressure drugs during pregnancy is becoming increasingly common,” said Brian T. Bateman, MD, lead author and Assistant Professor of Anesthesia at Harvard Medical School in Boston, Massachusetts. "While we know high blood pressure, or hypertension, occurs in about 6% to 8% of all pregnancies, we know little about how women and their doctors treat the condition."1
A database of more than 1 million Medicaid patients was studied. Of those patients, 48,453 (4.4%) filled prescriptions for high blood pressure drugs during their pregnancies.1
- Antihypertensive drug use increased from 3.5% to 4.9% between 2000 and 2006.
- Antihypertensive drug users were older than nonusers, more likely to have diabetes or kidney disease, and more likely to be white or black than Hispanic or Asian.
- Nearly 2% of pregnant women filled prescriptions for antihypertensive drugs during the first trimester; 1.7% during the second trimester; and 3.2% during the third trimester.
- Angiotensin-converting-enzyme (ACE) inhibitors and angiotensin receptor blockers—both of which have been shown to have harmful side effects during pregnancy—were used by 4.9% of users in the second trimester, and 1.1% in the third trimester.
Research is needed because there are limited data on the safety, efficacy, and proper use of antihypertensive drugs during pregnancy.
“These drugs can cause poor growth, kidney problems, and even death of the newborn, wrote Bateman. “If women are taking one of these blood pressure medications and they become pregnant or plan to do so, they and their doctors should discuss treatment choices during pregnancy.”1
For access to the abstract, click here.
We want to hear from you! Tell us what you think.
1. More pregnant women taking high blood pressure drugs, yet safety unclear (news release). GlobeNewswire.com. http://www.globenewswire.com/newsroom/news.html?d=10004131. Published September 10, 2012. Accessed September 11, 2012.
2. Bateman BT, Hernandez-Diaz S, Huybrechts KF, et al. Patterns of outpatient antihypertensive medication use during pregnancy in a Medicaid population [published online ahead of print September 10, 2012]. Hypertension. doi: 10.1161/HYPERTENSIONAHA.112.197095.
New research in the American Heart Association journal Hypertension indicates that almost 5% of pregnant women are prescribed antihypertensives, including some drugs that are contraindicated for mothers or their babies.
“Use of high blood pressure drugs during pregnancy is becoming increasingly common,” said Brian T. Bateman, MD, lead author and Assistant Professor of Anesthesia at Harvard Medical School in Boston, Massachusetts. "While we know high blood pressure, or hypertension, occurs in about 6% to 8% of all pregnancies, we know little about how women and their doctors treat the condition."1
A database of more than 1 million Medicaid patients was studied. Of those patients, 48,453 (4.4%) filled prescriptions for high blood pressure drugs during their pregnancies.1
- Antihypertensive drug use increased from 3.5% to 4.9% between 2000 and 2006.
- Antihypertensive drug users were older than nonusers, more likely to have diabetes or kidney disease, and more likely to be white or black than Hispanic or Asian.
- Nearly 2% of pregnant women filled prescriptions for antihypertensive drugs during the first trimester; 1.7% during the second trimester; and 3.2% during the third trimester.
- Angiotensin-converting-enzyme (ACE) inhibitors and angiotensin receptor blockers—both of which have been shown to have harmful side effects during pregnancy—were used by 4.9% of users in the second trimester, and 1.1% in the third trimester.
Research is needed because there are limited data on the safety, efficacy, and proper use of antihypertensive drugs during pregnancy.
“These drugs can cause poor growth, kidney problems, and even death of the newborn, wrote Bateman. “If women are taking one of these blood pressure medications and they become pregnant or plan to do so, they and their doctors should discuss treatment choices during pregnancy.”1
For access to the abstract, click here.
We want to hear from you! Tell us what you think.
New research in the American Heart Association journal Hypertension indicates that almost 5% of pregnant women are prescribed antihypertensives, including some drugs that are contraindicated for mothers or their babies.
“Use of high blood pressure drugs during pregnancy is becoming increasingly common,” said Brian T. Bateman, MD, lead author and Assistant Professor of Anesthesia at Harvard Medical School in Boston, Massachusetts. "While we know high blood pressure, or hypertension, occurs in about 6% to 8% of all pregnancies, we know little about how women and their doctors treat the condition."1
A database of more than 1 million Medicaid patients was studied. Of those patients, 48,453 (4.4%) filled prescriptions for high blood pressure drugs during their pregnancies.1
- Antihypertensive drug use increased from 3.5% to 4.9% between 2000 and 2006.
- Antihypertensive drug users were older than nonusers, more likely to have diabetes or kidney disease, and more likely to be white or black than Hispanic or Asian.
- Nearly 2% of pregnant women filled prescriptions for antihypertensive drugs during the first trimester; 1.7% during the second trimester; and 3.2% during the third trimester.
- Angiotensin-converting-enzyme (ACE) inhibitors and angiotensin receptor blockers—both of which have been shown to have harmful side effects during pregnancy—were used by 4.9% of users in the second trimester, and 1.1% in the third trimester.
Research is needed because there are limited data on the safety, efficacy, and proper use of antihypertensive drugs during pregnancy.
“These drugs can cause poor growth, kidney problems, and even death of the newborn, wrote Bateman. “If women are taking one of these blood pressure medications and they become pregnant or plan to do so, they and their doctors should discuss treatment choices during pregnancy.”1
For access to the abstract, click here.
We want to hear from you! Tell us what you think.
1. More pregnant women taking high blood pressure drugs, yet safety unclear (news release). GlobeNewswire.com. http://www.globenewswire.com/newsroom/news.html?d=10004131. Published September 10, 2012. Accessed September 11, 2012.
2. Bateman BT, Hernandez-Diaz S, Huybrechts KF, et al. Patterns of outpatient antihypertensive medication use during pregnancy in a Medicaid population [published online ahead of print September 10, 2012]. Hypertension. doi: 10.1161/HYPERTENSIONAHA.112.197095.
1. More pregnant women taking high blood pressure drugs, yet safety unclear (news release). GlobeNewswire.com. http://www.globenewswire.com/newsroom/news.html?d=10004131. Published September 10, 2012. Accessed September 11, 2012.
2. Bateman BT, Hernandez-Diaz S, Huybrechts KF, et al. Patterns of outpatient antihypertensive medication use during pregnancy in a Medicaid population [published online ahead of print September 10, 2012]. Hypertension. doi: 10.1161/HYPERTENSIONAHA.112.197095.
Is elective delivery at 37 weeks’ gestation safe in uncomplicated twin pregnancies?
In uncomplicated twin pregnancies, timing of delivery remains an area of controversy. The optimal length of gestation appears to be shorter in twins, compared with singleton pregnancies. Although population-based studies are limited by the inclusion of complicated pregnancies, data suggest that the nadir in perinatal mortality occurs at 37 to 39 weeks in twins versus 39 to 41 weeks in singletons.1 It is reasonable, therefore, to consider elective delivery of uncomplicated twin pregnancies prior to 39 weeks’ gestation, when elective delivery of singleton pregnancies becomes an option. The American College of Obstetricians and Gynecologists suggests that it is safe to continue uncomplicated twin pregnancies beyond 37 weeks, although there is a lack of prospective data to establish the optimal time of delivery.
Details of the study
Enter Dodd and colleagues, who conducted their randomized, controlled trial to assess the risks and benefits of elective delivery of twins at 37 weeks. In this multicenter study from Australia, women who had uncomplicated twin pregnancies at 36 6/7 weeks or beyond with no contraindication to continuing the pregnancy were randomly assigned to:
- an elective birth group, with planned delivery at 37 weeks, or
- a “standard care” group, with delivery planned at 38 weeks or later.
The primary outcome was a composite of serious adverse outcome for the infants, including death and serious morbidity.
The elective birth group delivered at a mean (SD) gestational age of 37.3 (0.4) weeks, versus 37.9 (0.5) weeks for the standard care group. In both groups, more than 80% of pregnancies were dichorionic, and elective cesarean delivery was performed in approximately one-third of women.
Although the primary outcome was less common in the elective birth group, the difference was due entirely to a higher rate of birthweight below the third centile (for gestational age at birth and infant sex) in the standard care group, with no difference in any other individual outcome. The only neonatal death occurred 27 days after birth as a result of group B streptococcus sepsis in one newborn from the standard care group. Induction of labor occurred more frequently in the elective birth group than in the standard care group (50.9% vs 37.8%; P = .046).
Strengths and weaknesses of the trial
Among the strengths of this study is the fact that it is the largest published randomized, controlled trial addressing the timing of delivery in uncomplicated twin pregnancies. It also appears to have been well conducted and appropriately analyzed.
The main weakness is that nearly all primary outcomes in both groups involved birthweights below the 3rd centile rather than mortality or clinical outcomes associated with significant morbidity in the newborn period.
No information on ultrasonographic monitoring of fetal growth was provided; it is possible that more aggressive screening could have identified growth restriction, rendering some patients ineligible for randomization and ultimate inclusion in the trial. In addition, nearly 20% of pregnancies were monochorionic; these gestations are typically delivered at early gestational ages due to higher rates of intrauterine mortality.2
This study provides reassurance that elective delivery at 37 weeks is likely to be safe and could reduce the rate of intrauterine growth restriction. Achieving this reduction would require a higher rate of induction of labor in this population.
An important consideration is the association between elective cesarean delivery and neonatal respiratory disorders, which has been noted in twins3 as well as in singletons.4 Therefore, unless there is an indication for earlier delivery, it may be reasonable to await scheduled cesarean delivery at 38 weeks.
Because of a higher rate of intrauterine mortality among monochorionic twins, it is prudent to deliver these pregnancies by 37 weeks.
Stephen T. Chasen, MD
We want to hear from you! Tell us what you think.
Does mediolateral episiotomy reduce the risk of anal sphincter injury in operative vaginal delivery?
Errol R. Norwitz, MD, PhD (Examining the Evidence, August 2012)
What is the optimal interval for osteoporosis screening in postmenopausal women before fracture occurrence and osteoporosis treatment initiation?
Steven R. Goldstein, MD (Examining the Evidence, August 2012)
Does long-acting reversible contraception prevent unintended pregnancy better than OCs, transdermal patch, and vaginal ring—regardless of a patient’s age?
Andrew M. Kaunitz, MD (Examining the Evidence, July 2012)
In women who have stress incontinence and intrinsic sphincter deficiency, which midurethral sling produces the best long-term results?
Ladin A. Yurteri-Kaplan MD, and Amy J. Park, MD (Examining the Evidence, June 2012)
1. Kahn B, Lumey LH, Zybert PA, et al. Prospective risk of fetal death in singleton, twin, and triplet gestations: implications for practice. Obstet Gynecol. 2003;102(4):685-692.
2. Breathnach FM, McAuliff FM, Geary M, et al. Perinatal Ireland Research Consortium. Optimum timing for planned delivery of uncomplicated monochorionic and dichorionic twin pregnancies. Obstet Gynecol. 2012;119(1):50-59.
3. Chasen ST, Madden A, Chervenak FA. Cesarean delivery of twins and neonatal respiratory disorders. Am J Obstet Gynecol. 1999;181(5 pt 1):1052-1056.
4. Tita AT, Landon MB, Spong CY, et al. Eunice Kennedy Shriver NICHD Maternal-Fetal Medicine Units Network. Timing of elective repeat cesarean delivery at term and neonatal outcomes. N Engl J Med. 2009;360(2):111-120.
In uncomplicated twin pregnancies, timing of delivery remains an area of controversy. The optimal length of gestation appears to be shorter in twins, compared with singleton pregnancies. Although population-based studies are limited by the inclusion of complicated pregnancies, data suggest that the nadir in perinatal mortality occurs at 37 to 39 weeks in twins versus 39 to 41 weeks in singletons.1 It is reasonable, therefore, to consider elective delivery of uncomplicated twin pregnancies prior to 39 weeks’ gestation, when elective delivery of singleton pregnancies becomes an option. The American College of Obstetricians and Gynecologists suggests that it is safe to continue uncomplicated twin pregnancies beyond 37 weeks, although there is a lack of prospective data to establish the optimal time of delivery.
Details of the study
Enter Dodd and colleagues, who conducted their randomized, controlled trial to assess the risks and benefits of elective delivery of twins at 37 weeks. In this multicenter study from Australia, women who had uncomplicated twin pregnancies at 36 6/7 weeks or beyond with no contraindication to continuing the pregnancy were randomly assigned to:
- an elective birth group, with planned delivery at 37 weeks, or
- a “standard care” group, with delivery planned at 38 weeks or later.
The primary outcome was a composite of serious adverse outcome for the infants, including death and serious morbidity.
The elective birth group delivered at a mean (SD) gestational age of 37.3 (0.4) weeks, versus 37.9 (0.5) weeks for the standard care group. In both groups, more than 80% of pregnancies were dichorionic, and elective cesarean delivery was performed in approximately one-third of women.
Although the primary outcome was less common in the elective birth group, the difference was due entirely to a higher rate of birthweight below the third centile (for gestational age at birth and infant sex) in the standard care group, with no difference in any other individual outcome. The only neonatal death occurred 27 days after birth as a result of group B streptococcus sepsis in one newborn from the standard care group. Induction of labor occurred more frequently in the elective birth group than in the standard care group (50.9% vs 37.8%; P = .046).
Strengths and weaknesses of the trial
Among the strengths of this study is the fact that it is the largest published randomized, controlled trial addressing the timing of delivery in uncomplicated twin pregnancies. It also appears to have been well conducted and appropriately analyzed.
The main weakness is that nearly all primary outcomes in both groups involved birthweights below the 3rd centile rather than mortality or clinical outcomes associated with significant morbidity in the newborn period.
No information on ultrasonographic monitoring of fetal growth was provided; it is possible that more aggressive screening could have identified growth restriction, rendering some patients ineligible for randomization and ultimate inclusion in the trial. In addition, nearly 20% of pregnancies were monochorionic; these gestations are typically delivered at early gestational ages due to higher rates of intrauterine mortality.2
This study provides reassurance that elective delivery at 37 weeks is likely to be safe and could reduce the rate of intrauterine growth restriction. Achieving this reduction would require a higher rate of induction of labor in this population.
An important consideration is the association between elective cesarean delivery and neonatal respiratory disorders, which has been noted in twins3 as well as in singletons.4 Therefore, unless there is an indication for earlier delivery, it may be reasonable to await scheduled cesarean delivery at 38 weeks.
Because of a higher rate of intrauterine mortality among monochorionic twins, it is prudent to deliver these pregnancies by 37 weeks.
Stephen T. Chasen, MD
We want to hear from you! Tell us what you think.
Does mediolateral episiotomy reduce the risk of anal sphincter injury in operative vaginal delivery?
Errol R. Norwitz, MD, PhD (Examining the Evidence, August 2012)
What is the optimal interval for osteoporosis screening in postmenopausal women before fracture occurrence and osteoporosis treatment initiation?
Steven R. Goldstein, MD (Examining the Evidence, August 2012)
Does long-acting reversible contraception prevent unintended pregnancy better than OCs, transdermal patch, and vaginal ring—regardless of a patient’s age?
Andrew M. Kaunitz, MD (Examining the Evidence, July 2012)
In women who have stress incontinence and intrinsic sphincter deficiency, which midurethral sling produces the best long-term results?
Ladin A. Yurteri-Kaplan MD, and Amy J. Park, MD (Examining the Evidence, June 2012)
In uncomplicated twin pregnancies, timing of delivery remains an area of controversy. The optimal length of gestation appears to be shorter in twins, compared with singleton pregnancies. Although population-based studies are limited by the inclusion of complicated pregnancies, data suggest that the nadir in perinatal mortality occurs at 37 to 39 weeks in twins versus 39 to 41 weeks in singletons.1 It is reasonable, therefore, to consider elective delivery of uncomplicated twin pregnancies prior to 39 weeks’ gestation, when elective delivery of singleton pregnancies becomes an option. The American College of Obstetricians and Gynecologists suggests that it is safe to continue uncomplicated twin pregnancies beyond 37 weeks, although there is a lack of prospective data to establish the optimal time of delivery.
Details of the study
Enter Dodd and colleagues, who conducted their randomized, controlled trial to assess the risks and benefits of elective delivery of twins at 37 weeks. In this multicenter study from Australia, women who had uncomplicated twin pregnancies at 36 6/7 weeks or beyond with no contraindication to continuing the pregnancy were randomly assigned to:
- an elective birth group, with planned delivery at 37 weeks, or
- a “standard care” group, with delivery planned at 38 weeks or later.
The primary outcome was a composite of serious adverse outcome for the infants, including death and serious morbidity.
The elective birth group delivered at a mean (SD) gestational age of 37.3 (0.4) weeks, versus 37.9 (0.5) weeks for the standard care group. In both groups, more than 80% of pregnancies were dichorionic, and elective cesarean delivery was performed in approximately one-third of women.
Although the primary outcome was less common in the elective birth group, the difference was due entirely to a higher rate of birthweight below the third centile (for gestational age at birth and infant sex) in the standard care group, with no difference in any other individual outcome. The only neonatal death occurred 27 days after birth as a result of group B streptococcus sepsis in one newborn from the standard care group. Induction of labor occurred more frequently in the elective birth group than in the standard care group (50.9% vs 37.8%; P = .046).
Strengths and weaknesses of the trial
Among the strengths of this study is the fact that it is the largest published randomized, controlled trial addressing the timing of delivery in uncomplicated twin pregnancies. It also appears to have been well conducted and appropriately analyzed.
The main weakness is that nearly all primary outcomes in both groups involved birthweights below the 3rd centile rather than mortality or clinical outcomes associated with significant morbidity in the newborn period.
No information on ultrasonographic monitoring of fetal growth was provided; it is possible that more aggressive screening could have identified growth restriction, rendering some patients ineligible for randomization and ultimate inclusion in the trial. In addition, nearly 20% of pregnancies were monochorionic; these gestations are typically delivered at early gestational ages due to higher rates of intrauterine mortality.2
This study provides reassurance that elective delivery at 37 weeks is likely to be safe and could reduce the rate of intrauterine growth restriction. Achieving this reduction would require a higher rate of induction of labor in this population.
An important consideration is the association between elective cesarean delivery and neonatal respiratory disorders, which has been noted in twins3 as well as in singletons.4 Therefore, unless there is an indication for earlier delivery, it may be reasonable to await scheduled cesarean delivery at 38 weeks.
Because of a higher rate of intrauterine mortality among monochorionic twins, it is prudent to deliver these pregnancies by 37 weeks.
Stephen T. Chasen, MD
We want to hear from you! Tell us what you think.
Does mediolateral episiotomy reduce the risk of anal sphincter injury in operative vaginal delivery?
Errol R. Norwitz, MD, PhD (Examining the Evidence, August 2012)
What is the optimal interval for osteoporosis screening in postmenopausal women before fracture occurrence and osteoporosis treatment initiation?
Steven R. Goldstein, MD (Examining the Evidence, August 2012)
Does long-acting reversible contraception prevent unintended pregnancy better than OCs, transdermal patch, and vaginal ring—regardless of a patient’s age?
Andrew M. Kaunitz, MD (Examining the Evidence, July 2012)
In women who have stress incontinence and intrinsic sphincter deficiency, which midurethral sling produces the best long-term results?
Ladin A. Yurteri-Kaplan MD, and Amy J. Park, MD (Examining the Evidence, June 2012)
1. Kahn B, Lumey LH, Zybert PA, et al. Prospective risk of fetal death in singleton, twin, and triplet gestations: implications for practice. Obstet Gynecol. 2003;102(4):685-692.
2. Breathnach FM, McAuliff FM, Geary M, et al. Perinatal Ireland Research Consortium. Optimum timing for planned delivery of uncomplicated monochorionic and dichorionic twin pregnancies. Obstet Gynecol. 2012;119(1):50-59.
3. Chasen ST, Madden A, Chervenak FA. Cesarean delivery of twins and neonatal respiratory disorders. Am J Obstet Gynecol. 1999;181(5 pt 1):1052-1056.
4. Tita AT, Landon MB, Spong CY, et al. Eunice Kennedy Shriver NICHD Maternal-Fetal Medicine Units Network. Timing of elective repeat cesarean delivery at term and neonatal outcomes. N Engl J Med. 2009;360(2):111-120.
1. Kahn B, Lumey LH, Zybert PA, et al. Prospective risk of fetal death in singleton, twin, and triplet gestations: implications for practice. Obstet Gynecol. 2003;102(4):685-692.
2. Breathnach FM, McAuliff FM, Geary M, et al. Perinatal Ireland Research Consortium. Optimum timing for planned delivery of uncomplicated monochorionic and dichorionic twin pregnancies. Obstet Gynecol. 2012;119(1):50-59.
3. Chasen ST, Madden A, Chervenak FA. Cesarean delivery of twins and neonatal respiratory disorders. Am J Obstet Gynecol. 1999;181(5 pt 1):1052-1056.
4. Tita AT, Landon MB, Spong CY, et al. Eunice Kennedy Shriver NICHD Maternal-Fetal Medicine Units Network. Timing of elective repeat cesarean delivery at term and neonatal outcomes. N Engl J Med. 2009;360(2):111-120.
Women’s health under the Affordable Care Act: What is covered?
Lay midwives and the ObGyn: Is collaboration risky?
(May 2012)
How state budget crises are putting the squeeze on Medicaid (and you)
(February 2012)
Is private ObGyn practice on its way out?
(October 2011)
14 questions (and answers) about health reform and you
with Janelle Yates, Senior Editor (August 2010)
For the first half of 2012, the big question was: Will anything be covered under the Affordable Care Act (ACA)? After considering constitutional challenges to the Act that had the potential to invalidate the entire law, the US Supreme Court ruled, on June 28, that the ACA met constitutional muster in National Federation of Independent Business v. Sebelius (2012).
Now that the Court has upheld the ACA, let’s review the major women’s health services included under the law. This Web version incorporates 10 more women's health provisions from the ACA, from smoking cessation to young women’s breast cancer, that were not in the print version.

Preventive services guaranteed without copays
A major component of the health reform law went into effect August 1, 2012; it requires most health plans to cover women’s preventive services without requiring enrollees to pay a copay or deductibles. This provision reflects Congress’ understanding that women have a longer life expectancy and bear a greater burden of chronic disease, disability, and reproductive and gender-specific conditions. In addition, women often have a different response to treatment than men do.
The federal Department of Health and Human Services (HHS) estimates that Americans use preventive services at only about half of the recommended rate. By 2013, as many as 73 million individuals will benefit from preventive care offered under the law.
The American Congress of Obstetricians and Gynecologists (ACOG) worked with the Institute of Medicine (IOM)—which was charged with advising HHS—to encourage the inclusion of women’s preventive services specified in ACOG guidelines to ensure women’s health and well-being. As ACOG Executive Vice President Hal C. Lawrence, MD, told the IOM in January 2011:
- The College’s clinical guidelines…offer an excellent resource…and encompass the entire field of women’s preventive care. Our guidance is based on the best available evidence and is developed by committees with expertise reflecting the breadth of women’s health care and subject to a rigorous conflict of interest policy.
Dr. Lawrence further urged the IOM “to recommend coverage of the following services and products without cost-sharing”:
- well-woman visits
- preconception care
- family planning counseling and services
- HIV screening (for women at average risk)
- screening for intimate partner violence
- testing for human papillomavirus (HPV) as part of cervical cancer screening.
ACOG’s recommendations were approved by the IOM and, subsequently, by HHS. As a result, all private health plans that began on or after September 30, 2010, are required to cover these services at no out-of-pocket cost to patients (TABLE).
Women’s preventive services guaranteed under ACA*
| Service | Frequency | HHS guidelines for health insurance coverage |
|---|---|---|
| Well-woman visit | Annual for adult women, although HHS recognizes that several visits may be needed to obtain all necessary recommended preventive services, depending on a woman’s health status, health needs, and other risk factors** | The visit should focus on preventive services that are appropriate for the patient’s age and development, including preconception and prenatal care. This visit should, where appropriate, include other preventive services listed in this set of guidelines, as well as others referenced in section 2713 |
| Screening for gestational diabetes | Between 24 and 28 weeks of gestation and at the first prenatal visit for pregnant women identified to be at high risk for diabetes | |
| Testing for human papillomavirus (HPV) | At age 30 and older, no more frequently than every 3 years | High-risk HPV DNA testing in women who have normal cervical cytology |
| Counseling about sexually transmitted infection (STI) | Annual | All sexually active women |
| Counseling about and screening for HIV | Annual | All sexually active women |
| Counseling about and provision of contraception† | As prescribed | All FDA-approved contraceptive methods and sterilization procedures. Counseling for all women with reproductive capacity |
| Breastfeeding support, supplies, and counseling | In conjunction with each birth | Comprehensive lactation support and counseling by a trained provider during pregnancy or postpartum (or both), as well as costs for renting breastfeeding equipment |
| Screening for and counseling about interpersonal and domestic violence | Annual | |
| HHS = Health and Human Services * HHS guidelines are effective August 1, 2011. Nongrandfathered plans and insurers are required to provide coverage without cost-sharing consistent with HHS guidelines in the first plan year (in the individual market, policy year) that begins on or after August 1, 2012. ** The July 2011 Institute of Medicine report titled “Clinical preventive services for women: closing the gap” lists recommendations on individual preventive services that may be obtained during a well-woman preventive service visit. † Group health plans sponsored by certain religious employers, and group health insurance coverage in connection with such plans, are exempt from the requirement to cover contraceptive services. SOURCE: Adapted from Healthcare.gov. Affordable Care Act Expands Prevention Coverage for Women’s Health and Well-Being. http://www.hrsa.gov/womensguidelines/. Accessed August 8, 2012. | ||
Contraceptive mandate triggers a firestorm
On February 10, 2012, under pressure from religious groups, the Obama Administration offered a religious exemption to the contraception mandate for certain employers and group health plans. Under this “accommodation,” certain religious employers are exempt from the requirement to cover contraceptive services in their group health plans. An employer qualifies for this exemption if it:
- has the inculcation of religious values as one of its purposes
- primarily employs individuals who share its religious tenets
- primarily serves individuals who share its religious tenets, and
- qualifies for nonprofit status under
Internal Revenue Service (IRS) rules. At the same time that the Obama Administration wanted to accommodate employers’ religious beliefs, it also wanted to ensure that every woman would have access to free preventive care, including contraceptive services, regardless of where she works. And so while the Administration requires insurers to offer group health plan coverage without contraceptive coverage to religious-affiliated organizations, it also requires insurers to provide contraceptive coverage directly to individuals covered under the organization’s group health plan with no cost sharing.
This contraceptive mandate—even with the accommodation—has set off a firestorm on Capitol Hill that will eventually be settled in the courts.
Medicaid expansion falls short of original goal
In National Federation of Independent Business v. Sebelius, the plaintiffs asked the Supreme Court to rule on the federal government’s authority to require states to expand their Medicaid programs. Medicaid costs are typically shared by the federal and state governments. Under the ACA, state Medicaid programs were required to cover nearly all individuals who have incomes below 133% of the federal poverty level—$30,656 for a family of four in 2012—paid entirely by the federal government from 2014 through 2016. After that, the federal share gradually declines to, and then stays at, 90%. States that did not expand their Medicaid programs risked losing all federal Medicaid funding.
The Court ruled that the federal government can expand Medicaid but can’t penalize states that don’t accept the expansion mandate—effectively turning the mandate into a state option. States will receive the additional federal funds if they expand coverage, but states that don’t expand will not be penalized by losing existing federal funds for other parts of the program.
Since the ruling, a number of governors have announced that they will not expand their Medicaid programs, including governors of Florida and Louisiana. Those two states alone are home to 20% of all individuals intended to be covered under the Medicaid expansion.
This part of the ACA is particularly important to women because it strikes, for the first time, the requirement that a low-income woman must be pregnant to receive Medicaid coverage.
The figure below shows the dramatic potential improvement in coverage for women if all states fully implement the Medicaid expansion. Time, court decisions, elections, and state budget fights will determine how much of this change is realized for women’s health.
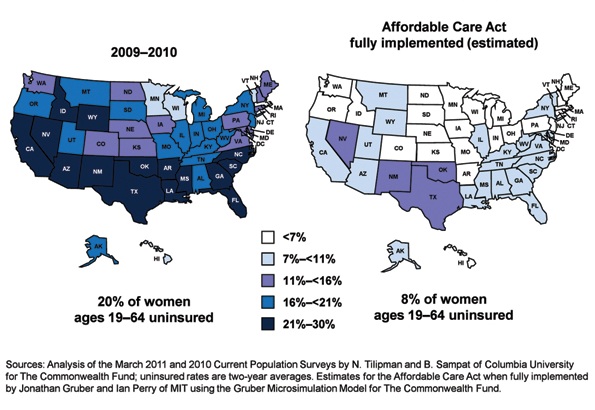
Percentage of insured women will increase under ACA
Percentage of women aged 19 to 64 years who were uninsured in 2009–2012 and under the Affordable Care Act when fully implemented.
SOURCE: Commonwealth Fund. Analysis of the March 2011 and 2010 Current Population Surveys by N. Tilipman and B. Sampat of Columbia University.
Women gain direct access to ObGyns
The ACA guarantees women in all states and all plans direct access to their ObGyns. Before the ACA, women in nine states lacked this guarantee, and women in 16 other states had only limited direct access. Now, a woman can go directly to her ObGyn without having to get a referral from her primary care physician or insurer.
Direct access is especially important because the ACA establishes new delivery systems, such as medical homes and accountable care organizations, designed to capture patients to maximize savings. An ObGyn does not have to be the patient’s primary care provider, and the patient’s access to her ObGyn cannot be limited to a certain number of visits or types of services.
ACA encourages states to cover family-planning services
Under the ACA, states have an easier time covering family-planning services, up to the same eligibility levels as pregnant women. Family planning is still an optional service that a state can choose to extend to women who have incomes above the Medicaid income eligibility level but, before the ACA was enacted, states had to apply to HHS for permission to waive the federal rules, often a very cumbersome process.
Prior to the ACA, 27 states had family planning waivers to provide services to nonpregnant women who had incomes above the Medicaid eligibility level—most at or near 200% of poverty. Now, states can provide family planning services to this population without federal approval.
Don’t miss Dr. Robert L. Barbieri’s October article titled “Gynecologic care across a woman’s life”
Insurance reforms end lifetime limits on coverage
Insurance reforms are important to us and our patients. The better the private health insurance system works—allowing us to provide our best possible care to patients and making sure they can see us when they need our care—the less our nation relies on the public safety net.
Beginning in 2010, the ACA eliminated all lifetime limits on how much insurance companies would cover when beneficiaries get sick; it also bans insurance companies from dropping people from coverage when they get sick. So if your patient has private health insurance and has faithfully paid her premiums and hasn’t committed fraud, her insurer cannot drop her or impose a limit on her coverage once she claims benefits.
This may be especially important for patients who need the most care, such as those who have cancer or another long-term, expensive, and unforeseen diagnosis. Because of this provision, you will not have to worry about your patient losing coverage in the middle of a long course of treatment.
The insurance practice of charging women more than men for equivalent policies ended on January 1, 2011, making insurance more affordable for our patients. Insurers in the individual and small group markets are allowed to vary premiums only for age, geographic location, family size, and tobacco use, not for gender—another important aspect of the law.
2014 is a key year in health reform
Exchanges begin
In 2014, under the ACA, state health insurance exchanges become reality.
An exchange is a marketplace where people can shop for health insurance; private health insurers can market their insurance products in state and multistate exchanges if they comply with new federal insurance reforms established in the ACA and offer the minimum benefits packages established by each state. Exchanges are intended to offer patients a choice of health insurance plans that are affordable, comprehensive, and easy to compare. Low-income individuals will be able to purchase private insurance in the exchanges with the federal premium subsidies or tax credits.
Insurers wanting to market their policies in an exchange may not deny coverage for preexisting conditions, including pregnancy, domestic violence, and previous cesarean delivery. They can’t deny coverage on the basis of an individual’s medical history, health status, genetic information, or disability. And they can’t impose waiting periods longer than 90 days before coverage takes effect, including 9-month waiting periods before maternity coverage.
Essential benefits are established
The ACA sets a minimum standard of health-care coverage that must be included in nearly every private insurance policy. The intent is that every person in the United States, regardless of where they live, who employs them, and what their income is, should have access to the same basic care.
Effective January 1, 2014, all insurance plans, except plans that existed before the ACA was enacted on March 23, 2010, must offer an “essential health benefits” (EHB) package, which must include:
- ambulatory patient services
- emergency services
- hospitalization
- maternity and newborn care
- mental health and substance use disorder services
- prescription drugs
- rehabilitative and habilitative services and devices
- laboratory services
- women’s preventive and wellness services and chronic disease management
- pediatric services, including oral and vision care.
Last December, HHS surprised many by giving states flexibility to design their own EHB packages, as long as the packages included each service on the list.
To choose its EHB package, a state must select a “benchmark” plan from the top- selling plans in four markets: federal and state public employee plans, commercial HMO plans, and small business plans. If a state doesn’t select a benchmark plan, the EHB defaults to the largest small-group market plan in the state. Each state must also choose an EHB package for its Medicaid program using the same 10 benefit categories.
State EHB plans must follow ACA requirements on annual and lifetime dollar limits but may impose limits on the scope and duration of coverage.
As for state-mandated benefits, if a state selects an EHB package that does not include a benefit already mandated by the state, the state must fund coverage for that service on its own—a decision HHS has promised to revisit in 2016.
Abortion decisions reside with the states
ACA requirements regarding abortion coverage 1) take effect in 2014 and 2) apply only to private health insurance plans marketed in the state exchanges that 3) cover abortions beyond those eligible for Medicaid coverage now, which are those that involve cases of rape or incest or that are necessary to save the life of the mother. Medicaid coverage for these categories of abortion is allowed under the Hyde Amendment.
Each insurer marketing a health plan in an exchange can determine whether or not its plan will cover abortion and, if it does, whether coverage will be limited to or go beyond those allowed under the Hyde Amendment. No federal tax or premium subsidies may be used to pay for abortions beyond those permitted by the Hyde Amendment.
The Secretary of HHS must ensure that at least one plan in each state exchange covers abortion, and that at least one plan either covers no abortions or limits abortions to those allowed under the Hyde Amendment. Insurers who offer abortion coverage beyond Hyde have to comply with a number of administrative requirements.
Congress was clear that the ultimate decisions about abortion should be made at the state rather than the federal level, and it gave states the ultimate trump card: Any state can pass legislation that prohibits any plan from offering abortion coverage of any kind within that state’s exchange. Any state can prohibit insurers offering plans within that state’s exchange from including any abortion coverage.
10 additional health provisions under the ACA
1. Creation of women's medical homes
The law points the way for creation of medical homes for women in the Medicare and Medicaid programs. The bill establishes an Innovation Center within the Centers for Medicare and Medicaid Services that has broad authority to evaluate, test, and adopt systems that foster patient-centered care, improve quality, and contain costs under Medicare, Medicaid, and the Children’s Health Insurance Program (CHIP)—and this includes patient-centered medical homes that address women’s unique health needs. ObGyn practices are eligible to participate and to receive additional reimbursement if they do.
2. Smoking-cessation counseling in pregnancy
The framers of the ACA recognized the large negative impact that smoking has on health, especially during pregnancy. Studies suggest that the intervention of a physician—most notably, counseling of the patient to quit smoking—has strong potential to modify this behavior. The new law provides reimbursement for this intervention. There are no copays or deductibles for patients, and smoking-cessation services can include diagnostic, therapeutic, and counseling modalities in addition to prescription of pharmacotherapy.
Before this bill became law, only 24 state Medicaid programs paid ObGyns or other physicians for smoking-cessation counseling of pregnant patients, and five states provided no coverage at all. Now, all pregnant Medicaid patients can get this counseling, and you’ll be paid for this important service.
3. Payments to nonphysician providers in freestanding birth centers
Before the ACA became law, Medicaid was authorized to pay hospitals and other facilities operated by and under the supervision of a physician; no payments were authorized for services of an ambulatory center operated by other health professionals. The ACA authorizes Medicaid payments to state-recognized freestanding birth centers not operated by or under the supervision of a physician. A state that doesn’t currently license birth centers must pass legislation and license these centers before the centers can receive these payments.
Medicaid will also reimburse providers who practice in state-recognized freestanding birth centers, as long as the individuals are practicing within their state’s scope of practice laws and regulations. Because the type of provider is not specified but instead left up to each state’s scope of practice laws and regulations, this provision could allow for separate provider payments for physicians, certified nurse midwives, certified professional midwives, and doulas.
4. Immigrant coverage
Legal immigrants are bound by the individual coverage mandate and must purchase health insurance. These individuals are eligible for income-related premium credits and subsidies for insurance purchased through an exchange. Legal immigrants who are barred from Medicaid during their first 5 years in the United States (by earlier law) are eligible for premium credits only.
Undocumented immigrants are not eligible for Medicaid, premium credits, or subsidies and are barred from purchasing insurance in the exchange, even with their own money.
5. Postpartum depression
Health reform will help bring perinatal and postpartum depression out of the shadows by providing federal funds for research, patient education, and clinical treatment. For example, the federal Department of Health and Human Services (HHS) will:
- conduct research into the causes of, and treatments for, postpartum conditions
- create a national public awareness campaign to increase awareness and knowledge of postpartum depression and psychosis
- provide grants to study the benefits of screening for postpartum depression and psychosis
- establish grants to deliver or enhance outpatient, inpatient, and home-based health and support services, including case management and comprehensive treatment services for individuals with, or at risk for, postpartum conditions.
The National Institute of Mental Health is encouraged to conduct a 10-year longitudinal study on the mental health consequences of pregnancy. This study is intended to focus on perinatal depression.
Community health centers will be eligible for grants in 2012 (as they were in 2011) to the tune of $3 million for inpatient and outpatient counseling and services.
And a federal public awareness campaign will educate the public through radio and television ads.
These endeavors point to the need for ObGyns to familiarize themselves with postpartum depression—if they aren’t already well versed in the subject—because patients are likely to become more aware of this issue and look to their ObGyns for answers.
6. Maternal home visits
Congress established a new Maternal, Infant, and Early Childhood Home Visiting program to improve maternal and fetal health in underserved areas of our country. This program will provide funds to states, tribes, and territories to develop and implement evidence-based home-visitation models to reduce infant and maternal mortality and its causes by producing improvements in:
- prenatal, maternal, and newborn health
- child health and development
- parenting skills
- school readiness
- juvenile delinquency
- family economic self-sufficiency.
These programs will have to demonstrate effectiveness and improved outcomes. HHS recently requested suggestions on ways of demonstrating the effectiveness of home-visiting program models for pregnant women, expectant fathers, and caregivers of children from birth through entry into kindergarten.
The law appropriates $350 million to this program in 2012 and $400 million in both 2013 and 2014.
7. Assistance for pregnant students
A new Pregnancy Assistance Fund—$25 million annually over 10 years (fiscal years 2010–2019)—requires the Secretary of HHS (in collaboration with the Secretary of Education) to establish a state grant program to help pregnant and parenting teens and young women. The aim of this program is to help teens who become pregnant and who choose to bring their pregnancies to term or keep their babies, or both, to stay in school. Grants will go to institutions of higher education, high schools and community service centers, as well as state attorneys general.
Institutions that receive grants must work with providers to meet specific practical needs of pregnant or parenting students:
- housing
- childcare
- parenting education
- postpartum counseling
- assistance in finding and accessing needed services
- referrals for prenatal care and delivery, infant or foster care, or adoption.
Funds to attorneys general will be used to combat domestic violence among pregnant teens.
8. Young women's breast cancer
A new program is intended to help educate young women about the importance of breast health and screening, in two ways:
- The National Institutes of Health (NIH) will conduct research to develop and test screening measures for prevention and early detection of breast cancer in women 15 to 44 years old.
- The US Department of HHS will create a national awareness campaign, with $9 million in funding each year from 2010 to 2014, to encourage young women to talk with their doctors about breast cancer and early detection.
ObGyns can expect to see more interest and questions about breast health among young women and their mothers. It pays to be prepared with good information for these important conversations.
9. Personal responsibility education
From 2010 through 2014, each state will receive funds for personal responsibility education programs aimed at reducing pregnancy in youths. Funds are $75 million for each fiscal year, allocated to each state depending on the size of its youth population but not intended to be less than $250,000 per state.
Educational programs eligible for federal funds must include both abstinence and contraception information for prevention of teenage pregnancy and sexually transmitted infections, including HIV/AIDS, as well as three or more adulthood-preparation subjects.
10. Community-based support of Patient-Centered Medical Homes
Federal funding is available to states for the development of community-based health teams to support medical homes run by primary care practices. These teams may include specialists, nurses, pharmacists, nutritionists, dieticians, social workers, behavioral and mental health providers, and physician assistants. Primary care practices in this program function as medical homes and are responsible for addressing a patient’s personal health-care needs. The team links the medical home to community support services for its patients.
Eligible ObGyn practices can qualify as primary care practices, and ObGyns are eligible to serve as specialist members of the community-based health team.
ACA is a mixed bag for ObGyns
Women have much to gain from the provisions of the ACA. It’s also true that many parts of the law are terrible for practicing ObGyns, including the Independent Payment Advisory Board (IPAB) and the absence of meaningful medical liability reform. For more on these issues, see “Is private ObGyn practice on its way out?” which appears in the October 2011 issue of OBG Management (available in the archive at obgmanagement.com). ACOG is committed to working with Congress to repeal or remedy those aspects of the law.
A recent study reveals that almost one-third of physicians are no longer accepting Medicaid patients
If all states expanded Medicaid to cover people with incomes at or below 138% of the federal poverty level in 2014, as the Affordable Care Act (ACA) proposes, 23 million people would become eligible for the program.1
That statistic prompts important questions:
- Would the health-care workforce be able to meet the demand of caring for all these new patients?
- Would it be willing?
A recent analysis of data from 4,326 office-based physicians suggests that the answer to both questions is “No”: Almost one-third of these providers were already declining to accept new Medicaid patients in 2011.2
Although 96% of physicians in the analysis accepted new patients in 2011, the percentage of physicians accepting new patients covered by Medicaid was lower (69%), as was the percentage accepting new self-paying patients (91.7%), patients covered by Medicare (83%), and patients with private insurance (82%).2
Physicians who were in solo practice were 23.5% less likely to accept new Medicaid patients, compared with those who practiced in an office with 10 or more other physicians.2
The data from this study come from the 2011 National Ambulatory Medical Care Survey Electronic Medical Records Supplement, a survey conducted by the Centers for Disease Control and Prevention National Center for Health Statistics. The survey included questions exploring whether physicians were accepting new patients.2
Earlier studies have found that the low reimbursement levels for care delivered through Medicaid has deterred many physicians from accepting patients.3
The view in the ObGyn specialty
The findings of this analysis were not broken down by specialty—only by primary care versus non–primary care. To get an idea of conditions in the ObGyn specialty, OBG Management surveyed the members of its Virtual Board of Editors (VBE). Of the 117 members contacted, 61 responded—a response rate of 52.1%. Roughly three-quarters (75.4%) reported that they currently treat patients covered by Medicaid, but only 60.7% are accepting new patients covered by Medicaid. Twenty-one percent of respondents reported that they have not and will not accept patients covered by Medicaid.
When asked to comment on their level of satisfaction with Medicaid, the most common response among VBE members was dissatisfaction due to “insufficient reimbursement.”
“I am not satisfied with Medicaid,” commented one VBE member. “The reimbursement is terrible….I have certainly thought of stopping care for Medicaid patients and, if Congress ever allows the big cuts to reimbursement that are threatened every year, I think I would stop.”
Another VBE member reported extreme dissatisfaction with Medicaid because of “lousy” reimbursement. He also pointed to “all the paperwork and crazy regulations that require inordinate time and additional personnel just to handle….and then [the claim] gets denied for reasons beyond reason.” He added that physicians who do accept Medicaid “are on the fast track to sainthood.”
Other reasons for refusing to accept patients with Medicaid (or, if Medicaid was accepted, for high levels of aggravation with the program):
- payment rejections
- too many different categories of coverage “that patients are completely uninformed about”
- difficulty finding a specialist who will manage high-risk patients covered by Medicaid
- red tape
- the complex health problems that Medicaid patients tend to have, compared with patients who have other types of coverage.
One VBE member summed up his feelings in one word: “Phooey.”
Several VBE members suggested that health reform should focus on the Medicaid program.
“These plans are just sucking up the state’s money and paying docs peanuts and their administrators big bucks!” wrote Mary Vanko, MD, of Munster, Indiana.
“I’m tired of how much Medicaid is being abused by people,” commented another VBE member. “People using other people’s cards, people with regular insurance getting Medicaid to cover their copays. The whole system needs reform!”
Some physicians were satisfied with Medicaid
Among the respondents were several who reported being satisfied with the program, including one who called the experience “good” and another who reported being “shielded from the reimbursement issues.”
“I have no problems with Medicaid,” wrote another.
—Janelle Yates, Senior Editor
References
1. Kenney GM, Dubay L, Zuckerman S, Huntress M. Making the Medicaid Expansion an ACA Option: How Many Low-Income Americans Could Remain Uninsured? Washington, DC: Urban Institute Health Policy Center; June 29, 2012. http://www.urban.org/UploadedPDF/412606-Making-the-Medicaid-Expansion-an-ACA-Option.pdf. Accessed August 18, 2012.
2. Decker SL. In 2011 nearly one-third of physicians said they would not accept new Medicaid patients, but rising fees may help. Health Affairs. 2012;31(8):1673–1679.
3. Centers for Disease Control and Prevention. QuickStats: percentage of office-based physicians accepting new patients, by types of payment accepted—United States, 1999–2000 and 2008–2009. MMWR Morb Mortal Wkly Rep. 2011;60(27):928.
We want to hear from you! Tell us what you think.
Lay midwives and the ObGyn: Is collaboration risky?
(May 2012)
How state budget crises are putting the squeeze on Medicaid (and you)
(February 2012)
Is private ObGyn practice on its way out?
(October 2011)
14 questions (and answers) about health reform and you
with Janelle Yates, Senior Editor (August 2010)
For the first half of 2012, the big question was: Will anything be covered under the Affordable Care Act (ACA)? After considering constitutional challenges to the Act that had the potential to invalidate the entire law, the US Supreme Court ruled, on June 28, that the ACA met constitutional muster in National Federation of Independent Business v. Sebelius (2012).
Now that the Court has upheld the ACA, let’s review the major women’s health services included under the law. This Web version incorporates 10 more women's health provisions from the ACA, from smoking cessation to young women’s breast cancer, that were not in the print version.

Preventive services guaranteed without copays
A major component of the health reform law went into effect August 1, 2012; it requires most health plans to cover women’s preventive services without requiring enrollees to pay a copay or deductibles. This provision reflects Congress’ understanding that women have a longer life expectancy and bear a greater burden of chronic disease, disability, and reproductive and gender-specific conditions. In addition, women often have a different response to treatment than men do.
The federal Department of Health and Human Services (HHS) estimates that Americans use preventive services at only about half of the recommended rate. By 2013, as many as 73 million individuals will benefit from preventive care offered under the law.
The American Congress of Obstetricians and Gynecologists (ACOG) worked with the Institute of Medicine (IOM)—which was charged with advising HHS—to encourage the inclusion of women’s preventive services specified in ACOG guidelines to ensure women’s health and well-being. As ACOG Executive Vice President Hal C. Lawrence, MD, told the IOM in January 2011:
- The College’s clinical guidelines…offer an excellent resource…and encompass the entire field of women’s preventive care. Our guidance is based on the best available evidence and is developed by committees with expertise reflecting the breadth of women’s health care and subject to a rigorous conflict of interest policy.
Dr. Lawrence further urged the IOM “to recommend coverage of the following services and products without cost-sharing”:
- well-woman visits
- preconception care
- family planning counseling and services
- HIV screening (for women at average risk)
- screening for intimate partner violence
- testing for human papillomavirus (HPV) as part of cervical cancer screening.
ACOG’s recommendations were approved by the IOM and, subsequently, by HHS. As a result, all private health plans that began on or after September 30, 2010, are required to cover these services at no out-of-pocket cost to patients (TABLE).
Women’s preventive services guaranteed under ACA*
| Service | Frequency | HHS guidelines for health insurance coverage |
|---|---|---|
| Well-woman visit | Annual for adult women, although HHS recognizes that several visits may be needed to obtain all necessary recommended preventive services, depending on a woman’s health status, health needs, and other risk factors** | The visit should focus on preventive services that are appropriate for the patient’s age and development, including preconception and prenatal care. This visit should, where appropriate, include other preventive services listed in this set of guidelines, as well as others referenced in section 2713 |
| Screening for gestational diabetes | Between 24 and 28 weeks of gestation and at the first prenatal visit for pregnant women identified to be at high risk for diabetes | |
| Testing for human papillomavirus (HPV) | At age 30 and older, no more frequently than every 3 years | High-risk HPV DNA testing in women who have normal cervical cytology |
| Counseling about sexually transmitted infection (STI) | Annual | All sexually active women |
| Counseling about and screening for HIV | Annual | All sexually active women |
| Counseling about and provision of contraception† | As prescribed | All FDA-approved contraceptive methods and sterilization procedures. Counseling for all women with reproductive capacity |
| Breastfeeding support, supplies, and counseling | In conjunction with each birth | Comprehensive lactation support and counseling by a trained provider during pregnancy or postpartum (or both), as well as costs for renting breastfeeding equipment |
| Screening for and counseling about interpersonal and domestic violence | Annual | |
| HHS = Health and Human Services * HHS guidelines are effective August 1, 2011. Nongrandfathered plans and insurers are required to provide coverage without cost-sharing consistent with HHS guidelines in the first plan year (in the individual market, policy year) that begins on or after August 1, 2012. ** The July 2011 Institute of Medicine report titled “Clinical preventive services for women: closing the gap” lists recommendations on individual preventive services that may be obtained during a well-woman preventive service visit. † Group health plans sponsored by certain religious employers, and group health insurance coverage in connection with such plans, are exempt from the requirement to cover contraceptive services. SOURCE: Adapted from Healthcare.gov. Affordable Care Act Expands Prevention Coverage for Women’s Health and Well-Being. http://www.hrsa.gov/womensguidelines/. Accessed August 8, 2012. | ||
Contraceptive mandate triggers a firestorm
On February 10, 2012, under pressure from religious groups, the Obama Administration offered a religious exemption to the contraception mandate for certain employers and group health plans. Under this “accommodation,” certain religious employers are exempt from the requirement to cover contraceptive services in their group health plans. An employer qualifies for this exemption if it:
- has the inculcation of religious values as one of its purposes
- primarily employs individuals who share its religious tenets
- primarily serves individuals who share its religious tenets, and
- qualifies for nonprofit status under
Internal Revenue Service (IRS) rules. At the same time that the Obama Administration wanted to accommodate employers’ religious beliefs, it also wanted to ensure that every woman would have access to free preventive care, including contraceptive services, regardless of where she works. And so while the Administration requires insurers to offer group health plan coverage without contraceptive coverage to religious-affiliated organizations, it also requires insurers to provide contraceptive coverage directly to individuals covered under the organization’s group health plan with no cost sharing.
This contraceptive mandate—even with the accommodation—has set off a firestorm on Capitol Hill that will eventually be settled in the courts.
Medicaid expansion falls short of original goal
In National Federation of Independent Business v. Sebelius, the plaintiffs asked the Supreme Court to rule on the federal government’s authority to require states to expand their Medicaid programs. Medicaid costs are typically shared by the federal and state governments. Under the ACA, state Medicaid programs were required to cover nearly all individuals who have incomes below 133% of the federal poverty level—$30,656 for a family of four in 2012—paid entirely by the federal government from 2014 through 2016. After that, the federal share gradually declines to, and then stays at, 90%. States that did not expand their Medicaid programs risked losing all federal Medicaid funding.
The Court ruled that the federal government can expand Medicaid but can’t penalize states that don’t accept the expansion mandate—effectively turning the mandate into a state option. States will receive the additional federal funds if they expand coverage, but states that don’t expand will not be penalized by losing existing federal funds for other parts of the program.
Since the ruling, a number of governors have announced that they will not expand their Medicaid programs, including governors of Florida and Louisiana. Those two states alone are home to 20% of all individuals intended to be covered under the Medicaid expansion.
This part of the ACA is particularly important to women because it strikes, for the first time, the requirement that a low-income woman must be pregnant to receive Medicaid coverage.
The figure below shows the dramatic potential improvement in coverage for women if all states fully implement the Medicaid expansion. Time, court decisions, elections, and state budget fights will determine how much of this change is realized for women’s health.

Percentage of insured women will increase under ACA
Percentage of women aged 19 to 64 years who were uninsured in 2009–2012 and under the Affordable Care Act when fully implemented.
SOURCE: Commonwealth Fund. Analysis of the March 2011 and 2010 Current Population Surveys by N. Tilipman and B. Sampat of Columbia University.
Women gain direct access to ObGyns
The ACA guarantees women in all states and all plans direct access to their ObGyns. Before the ACA, women in nine states lacked this guarantee, and women in 16 other states had only limited direct access. Now, a woman can go directly to her ObGyn without having to get a referral from her primary care physician or insurer.
Direct access is especially important because the ACA establishes new delivery systems, such as medical homes and accountable care organizations, designed to capture patients to maximize savings. An ObGyn does not have to be the patient’s primary care provider, and the patient’s access to her ObGyn cannot be limited to a certain number of visits or types of services.
ACA encourages states to cover family-planning services
Under the ACA, states have an easier time covering family-planning services, up to the same eligibility levels as pregnant women. Family planning is still an optional service that a state can choose to extend to women who have incomes above the Medicaid income eligibility level but, before the ACA was enacted, states had to apply to HHS for permission to waive the federal rules, often a very cumbersome process.
Prior to the ACA, 27 states had family planning waivers to provide services to nonpregnant women who had incomes above the Medicaid eligibility level—most at or near 200% of poverty. Now, states can provide family planning services to this population without federal approval.
Don’t miss Dr. Robert L. Barbieri’s October article titled “Gynecologic care across a woman’s life”
Insurance reforms end lifetime limits on coverage
Insurance reforms are important to us and our patients. The better the private health insurance system works—allowing us to provide our best possible care to patients and making sure they can see us when they need our care—the less our nation relies on the public safety net.
Beginning in 2010, the ACA eliminated all lifetime limits on how much insurance companies would cover when beneficiaries get sick; it also bans insurance companies from dropping people from coverage when they get sick. So if your patient has private health insurance and has faithfully paid her premiums and hasn’t committed fraud, her insurer cannot drop her or impose a limit on her coverage once she claims benefits.
This may be especially important for patients who need the most care, such as those who have cancer or another long-term, expensive, and unforeseen diagnosis. Because of this provision, you will not have to worry about your patient losing coverage in the middle of a long course of treatment.
The insurance practice of charging women more than men for equivalent policies ended on January 1, 2011, making insurance more affordable for our patients. Insurers in the individual and small group markets are allowed to vary premiums only for age, geographic location, family size, and tobacco use, not for gender—another important aspect of the law.
2014 is a key year in health reform
Exchanges begin
In 2014, under the ACA, state health insurance exchanges become reality.
An exchange is a marketplace where people can shop for health insurance; private health insurers can market their insurance products in state and multistate exchanges if they comply with new federal insurance reforms established in the ACA and offer the minimum benefits packages established by each state. Exchanges are intended to offer patients a choice of health insurance plans that are affordable, comprehensive, and easy to compare. Low-income individuals will be able to purchase private insurance in the exchanges with the federal premium subsidies or tax credits.
Insurers wanting to market their policies in an exchange may not deny coverage for preexisting conditions, including pregnancy, domestic violence, and previous cesarean delivery. They can’t deny coverage on the basis of an individual’s medical history, health status, genetic information, or disability. And they can’t impose waiting periods longer than 90 days before coverage takes effect, including 9-month waiting periods before maternity coverage.
Essential benefits are established
The ACA sets a minimum standard of health-care coverage that must be included in nearly every private insurance policy. The intent is that every person in the United States, regardless of where they live, who employs them, and what their income is, should have access to the same basic care.
Effective January 1, 2014, all insurance plans, except plans that existed before the ACA was enacted on March 23, 2010, must offer an “essential health benefits” (EHB) package, which must include:
- ambulatory patient services
- emergency services
- hospitalization
- maternity and newborn care
- mental health and substance use disorder services
- prescription drugs
- rehabilitative and habilitative services and devices
- laboratory services
- women’s preventive and wellness services and chronic disease management
- pediatric services, including oral and vision care.
Last December, HHS surprised many by giving states flexibility to design their own EHB packages, as long as the packages included each service on the list.
To choose its EHB package, a state must select a “benchmark” plan from the top- selling plans in four markets: federal and state public employee plans, commercial HMO plans, and small business plans. If a state doesn’t select a benchmark plan, the EHB defaults to the largest small-group market plan in the state. Each state must also choose an EHB package for its Medicaid program using the same 10 benefit categories.
State EHB plans must follow ACA requirements on annual and lifetime dollar limits but may impose limits on the scope and duration of coverage.
As for state-mandated benefits, if a state selects an EHB package that does not include a benefit already mandated by the state, the state must fund coverage for that service on its own—a decision HHS has promised to revisit in 2016.
Abortion decisions reside with the states
ACA requirements regarding abortion coverage 1) take effect in 2014 and 2) apply only to private health insurance plans marketed in the state exchanges that 3) cover abortions beyond those eligible for Medicaid coverage now, which are those that involve cases of rape or incest or that are necessary to save the life of the mother. Medicaid coverage for these categories of abortion is allowed under the Hyde Amendment.
Each insurer marketing a health plan in an exchange can determine whether or not its plan will cover abortion and, if it does, whether coverage will be limited to or go beyond those allowed under the Hyde Amendment. No federal tax or premium subsidies may be used to pay for abortions beyond those permitted by the Hyde Amendment.
The Secretary of HHS must ensure that at least one plan in each state exchange covers abortion, and that at least one plan either covers no abortions or limits abortions to those allowed under the Hyde Amendment. Insurers who offer abortion coverage beyond Hyde have to comply with a number of administrative requirements.
Congress was clear that the ultimate decisions about abortion should be made at the state rather than the federal level, and it gave states the ultimate trump card: Any state can pass legislation that prohibits any plan from offering abortion coverage of any kind within that state’s exchange. Any state can prohibit insurers offering plans within that state’s exchange from including any abortion coverage.
10 additional health provisions under the ACA
1. Creation of women's medical homes
The law points the way for creation of medical homes for women in the Medicare and Medicaid programs. The bill establishes an Innovation Center within the Centers for Medicare and Medicaid Services that has broad authority to evaluate, test, and adopt systems that foster patient-centered care, improve quality, and contain costs under Medicare, Medicaid, and the Children’s Health Insurance Program (CHIP)—and this includes patient-centered medical homes that address women’s unique health needs. ObGyn practices are eligible to participate and to receive additional reimbursement if they do.
2. Smoking-cessation counseling in pregnancy
The framers of the ACA recognized the large negative impact that smoking has on health, especially during pregnancy. Studies suggest that the intervention of a physician—most notably, counseling of the patient to quit smoking—has strong potential to modify this behavior. The new law provides reimbursement for this intervention. There are no copays or deductibles for patients, and smoking-cessation services can include diagnostic, therapeutic, and counseling modalities in addition to prescription of pharmacotherapy.
Before this bill became law, only 24 state Medicaid programs paid ObGyns or other physicians for smoking-cessation counseling of pregnant patients, and five states provided no coverage at all. Now, all pregnant Medicaid patients can get this counseling, and you’ll be paid for this important service.
3. Payments to nonphysician providers in freestanding birth centers
Before the ACA became law, Medicaid was authorized to pay hospitals and other facilities operated by and under the supervision of a physician; no payments were authorized for services of an ambulatory center operated by other health professionals. The ACA authorizes Medicaid payments to state-recognized freestanding birth centers not operated by or under the supervision of a physician. A state that doesn’t currently license birth centers must pass legislation and license these centers before the centers can receive these payments.
Medicaid will also reimburse providers who practice in state-recognized freestanding birth centers, as long as the individuals are practicing within their state’s scope of practice laws and regulations. Because the type of provider is not specified but instead left up to each state’s scope of practice laws and regulations, this provision could allow for separate provider payments for physicians, certified nurse midwives, certified professional midwives, and doulas.
4. Immigrant coverage
Legal immigrants are bound by the individual coverage mandate and must purchase health insurance. These individuals are eligible for income-related premium credits and subsidies for insurance purchased through an exchange. Legal immigrants who are barred from Medicaid during their first 5 years in the United States (by earlier law) are eligible for premium credits only.
Undocumented immigrants are not eligible for Medicaid, premium credits, or subsidies and are barred from purchasing insurance in the exchange, even with their own money.
5. Postpartum depression
Health reform will help bring perinatal and postpartum depression out of the shadows by providing federal funds for research, patient education, and clinical treatment. For example, the federal Department of Health and Human Services (HHS) will:
- conduct research into the causes of, and treatments for, postpartum conditions
- create a national public awareness campaign to increase awareness and knowledge of postpartum depression and psychosis
- provide grants to study the benefits of screening for postpartum depression and psychosis
- establish grants to deliver or enhance outpatient, inpatient, and home-based health and support services, including case management and comprehensive treatment services for individuals with, or at risk for, postpartum conditions.
The National Institute of Mental Health is encouraged to conduct a 10-year longitudinal study on the mental health consequences of pregnancy. This study is intended to focus on perinatal depression.
Community health centers will be eligible for grants in 2012 (as they were in 2011) to the tune of $3 million for inpatient and outpatient counseling and services.
And a federal public awareness campaign will educate the public through radio and television ads.
These endeavors point to the need for ObGyns to familiarize themselves with postpartum depression—if they aren’t already well versed in the subject—because patients are likely to become more aware of this issue and look to their ObGyns for answers.
6. Maternal home visits
Congress established a new Maternal, Infant, and Early Childhood Home Visiting program to improve maternal and fetal health in underserved areas of our country. This program will provide funds to states, tribes, and territories to develop and implement evidence-based home-visitation models to reduce infant and maternal mortality and its causes by producing improvements in:
- prenatal, maternal, and newborn health
- child health and development
- parenting skills
- school readiness
- juvenile delinquency
- family economic self-sufficiency.
These programs will have to demonstrate effectiveness and improved outcomes. HHS recently requested suggestions on ways of demonstrating the effectiveness of home-visiting program models for pregnant women, expectant fathers, and caregivers of children from birth through entry into kindergarten.
The law appropriates $350 million to this program in 2012 and $400 million in both 2013 and 2014.
7. Assistance for pregnant students
A new Pregnancy Assistance Fund—$25 million annually over 10 years (fiscal years 2010–2019)—requires the Secretary of HHS (in collaboration with the Secretary of Education) to establish a state grant program to help pregnant and parenting teens and young women. The aim of this program is to help teens who become pregnant and who choose to bring their pregnancies to term or keep their babies, or both, to stay in school. Grants will go to institutions of higher education, high schools and community service centers, as well as state attorneys general.
Institutions that receive grants must work with providers to meet specific practical needs of pregnant or parenting students:
- housing
- childcare
- parenting education
- postpartum counseling
- assistance in finding and accessing needed services
- referrals for prenatal care and delivery, infant or foster care, or adoption.
Funds to attorneys general will be used to combat domestic violence among pregnant teens.
8. Young women's breast cancer
A new program is intended to help educate young women about the importance of breast health and screening, in two ways:
- The National Institutes of Health (NIH) will conduct research to develop and test screening measures for prevention and early detection of breast cancer in women 15 to 44 years old.
- The US Department of HHS will create a national awareness campaign, with $9 million in funding each year from 2010 to 2014, to encourage young women to talk with their doctors about breast cancer and early detection.
ObGyns can expect to see more interest and questions about breast health among young women and their mothers. It pays to be prepared with good information for these important conversations.
9. Personal responsibility education
From 2010 through 2014, each state will receive funds for personal responsibility education programs aimed at reducing pregnancy in youths. Funds are $75 million for each fiscal year, allocated to each state depending on the size of its youth population but not intended to be less than $250,000 per state.
Educational programs eligible for federal funds must include both abstinence and contraception information for prevention of teenage pregnancy and sexually transmitted infections, including HIV/AIDS, as well as three or more adulthood-preparation subjects.
10. Community-based support of Patient-Centered Medical Homes
Federal funding is available to states for the development of community-based health teams to support medical homes run by primary care practices. These teams may include specialists, nurses, pharmacists, nutritionists, dieticians, social workers, behavioral and mental health providers, and physician assistants. Primary care practices in this program function as medical homes and are responsible for addressing a patient’s personal health-care needs. The team links the medical home to community support services for its patients.
Eligible ObGyn practices can qualify as primary care practices, and ObGyns are eligible to serve as specialist members of the community-based health team.
ACA is a mixed bag for ObGyns
Women have much to gain from the provisions of the ACA. It’s also true that many parts of the law are terrible for practicing ObGyns, including the Independent Payment Advisory Board (IPAB) and the absence of meaningful medical liability reform. For more on these issues, see “Is private ObGyn practice on its way out?” which appears in the October 2011 issue of OBG Management (available in the archive at obgmanagement.com). ACOG is committed to working with Congress to repeal or remedy those aspects of the law.
A recent study reveals that almost one-third of physicians are no longer accepting Medicaid patients
If all states expanded Medicaid to cover people with incomes at or below 138% of the federal poverty level in 2014, as the Affordable Care Act (ACA) proposes, 23 million people would become eligible for the program.1
That statistic prompts important questions:
- Would the health-care workforce be able to meet the demand of caring for all these new patients?
- Would it be willing?
A recent analysis of data from 4,326 office-based physicians suggests that the answer to both questions is “No”: Almost one-third of these providers were already declining to accept new Medicaid patients in 2011.2
Although 96% of physicians in the analysis accepted new patients in 2011, the percentage of physicians accepting new patients covered by Medicaid was lower (69%), as was the percentage accepting new self-paying patients (91.7%), patients covered by Medicare (83%), and patients with private insurance (82%).2
Physicians who were in solo practice were 23.5% less likely to accept new Medicaid patients, compared with those who practiced in an office with 10 or more other physicians.2
The data from this study come from the 2011 National Ambulatory Medical Care Survey Electronic Medical Records Supplement, a survey conducted by the Centers for Disease Control and Prevention National Center for Health Statistics. The survey included questions exploring whether physicians were accepting new patients.2
Earlier studies have found that the low reimbursement levels for care delivered through Medicaid has deterred many physicians from accepting patients.3
The view in the ObGyn specialty
The findings of this analysis were not broken down by specialty—only by primary care versus non–primary care. To get an idea of conditions in the ObGyn specialty, OBG Management surveyed the members of its Virtual Board of Editors (VBE). Of the 117 members contacted, 61 responded—a response rate of 52.1%. Roughly three-quarters (75.4%) reported that they currently treat patients covered by Medicaid, but only 60.7% are accepting new patients covered by Medicaid. Twenty-one percent of respondents reported that they have not and will not accept patients covered by Medicaid.
When asked to comment on their level of satisfaction with Medicaid, the most common response among VBE members was dissatisfaction due to “insufficient reimbursement.”
“I am not satisfied with Medicaid,” commented one VBE member. “The reimbursement is terrible….I have certainly thought of stopping care for Medicaid patients and, if Congress ever allows the big cuts to reimbursement that are threatened every year, I think I would stop.”
Another VBE member reported extreme dissatisfaction with Medicaid because of “lousy” reimbursement. He also pointed to “all the paperwork and crazy regulations that require inordinate time and additional personnel just to handle….and then [the claim] gets denied for reasons beyond reason.” He added that physicians who do accept Medicaid “are on the fast track to sainthood.”
Other reasons for refusing to accept patients with Medicaid (or, if Medicaid was accepted, for high levels of aggravation with the program):
- payment rejections
- too many different categories of coverage “that patients are completely uninformed about”
- difficulty finding a specialist who will manage high-risk patients covered by Medicaid
- red tape
- the complex health problems that Medicaid patients tend to have, compared with patients who have other types of coverage.
One VBE member summed up his feelings in one word: “Phooey.”
Several VBE members suggested that health reform should focus on the Medicaid program.
“These plans are just sucking up the state’s money and paying docs peanuts and their administrators big bucks!” wrote Mary Vanko, MD, of Munster, Indiana.
“I’m tired of how much Medicaid is being abused by people,” commented another VBE member. “People using other people’s cards, people with regular insurance getting Medicaid to cover their copays. The whole system needs reform!”
Some physicians were satisfied with Medicaid
Among the respondents were several who reported being satisfied with the program, including one who called the experience “good” and another who reported being “shielded from the reimbursement issues.”
“I have no problems with Medicaid,” wrote another.
—Janelle Yates, Senior Editor
References
1. Kenney GM, Dubay L, Zuckerman S, Huntress M. Making the Medicaid Expansion an ACA Option: How Many Low-Income Americans Could Remain Uninsured? Washington, DC: Urban Institute Health Policy Center; June 29, 2012. http://www.urban.org/UploadedPDF/412606-Making-the-Medicaid-Expansion-an-ACA-Option.pdf. Accessed August 18, 2012.
2. Decker SL. In 2011 nearly one-third of physicians said they would not accept new Medicaid patients, but rising fees may help. Health Affairs. 2012;31(8):1673–1679.
3. Centers for Disease Control and Prevention. QuickStats: percentage of office-based physicians accepting new patients, by types of payment accepted—United States, 1999–2000 and 2008–2009. MMWR Morb Mortal Wkly Rep. 2011;60(27):928.
We want to hear from you! Tell us what you think.
Lay midwives and the ObGyn: Is collaboration risky?
(May 2012)
How state budget crises are putting the squeeze on Medicaid (and you)
(February 2012)
Is private ObGyn practice on its way out?
(October 2011)
14 questions (and answers) about health reform and you
with Janelle Yates, Senior Editor (August 2010)
For the first half of 2012, the big question was: Will anything be covered under the Affordable Care Act (ACA)? After considering constitutional challenges to the Act that had the potential to invalidate the entire law, the US Supreme Court ruled, on June 28, that the ACA met constitutional muster in National Federation of Independent Business v. Sebelius (2012).
Now that the Court has upheld the ACA, let’s review the major women’s health services included under the law. This Web version incorporates 10 more women's health provisions from the ACA, from smoking cessation to young women’s breast cancer, that were not in the print version.

Preventive services guaranteed without copays
A major component of the health reform law went into effect August 1, 2012; it requires most health plans to cover women’s preventive services without requiring enrollees to pay a copay or deductibles. This provision reflects Congress’ understanding that women have a longer life expectancy and bear a greater burden of chronic disease, disability, and reproductive and gender-specific conditions. In addition, women often have a different response to treatment than men do.
The federal Department of Health and Human Services (HHS) estimates that Americans use preventive services at only about half of the recommended rate. By 2013, as many as 73 million individuals will benefit from preventive care offered under the law.
The American Congress of Obstetricians and Gynecologists (ACOG) worked with the Institute of Medicine (IOM)—which was charged with advising HHS—to encourage the inclusion of women’s preventive services specified in ACOG guidelines to ensure women’s health and well-being. As ACOG Executive Vice President Hal C. Lawrence, MD, told the IOM in January 2011:
- The College’s clinical guidelines…offer an excellent resource…and encompass the entire field of women’s preventive care. Our guidance is based on the best available evidence and is developed by committees with expertise reflecting the breadth of women’s health care and subject to a rigorous conflict of interest policy.
Dr. Lawrence further urged the IOM “to recommend coverage of the following services and products without cost-sharing”:
- well-woman visits
- preconception care
- family planning counseling and services
- HIV screening (for women at average risk)
- screening for intimate partner violence
- testing for human papillomavirus (HPV) as part of cervical cancer screening.
ACOG’s recommendations were approved by the IOM and, subsequently, by HHS. As a result, all private health plans that began on or after September 30, 2010, are required to cover these services at no out-of-pocket cost to patients (TABLE).
Women’s preventive services guaranteed under ACA*
| Service | Frequency | HHS guidelines for health insurance coverage |
|---|---|---|
| Well-woman visit | Annual for adult women, although HHS recognizes that several visits may be needed to obtain all necessary recommended preventive services, depending on a woman’s health status, health needs, and other risk factors** | The visit should focus on preventive services that are appropriate for the patient’s age and development, including preconception and prenatal care. This visit should, where appropriate, include other preventive services listed in this set of guidelines, as well as others referenced in section 2713 |
| Screening for gestational diabetes | Between 24 and 28 weeks of gestation and at the first prenatal visit for pregnant women identified to be at high risk for diabetes | |
| Testing for human papillomavirus (HPV) | At age 30 and older, no more frequently than every 3 years | High-risk HPV DNA testing in women who have normal cervical cytology |
| Counseling about sexually transmitted infection (STI) | Annual | All sexually active women |
| Counseling about and screening for HIV | Annual | All sexually active women |
| Counseling about and provision of contraception† | As prescribed | All FDA-approved contraceptive methods and sterilization procedures. Counseling for all women with reproductive capacity |
| Breastfeeding support, supplies, and counseling | In conjunction with each birth | Comprehensive lactation support and counseling by a trained provider during pregnancy or postpartum (or both), as well as costs for renting breastfeeding equipment |
| Screening for and counseling about interpersonal and domestic violence | Annual | |
| HHS = Health and Human Services * HHS guidelines are effective August 1, 2011. Nongrandfathered plans and insurers are required to provide coverage without cost-sharing consistent with HHS guidelines in the first plan year (in the individual market, policy year) that begins on or after August 1, 2012. ** The July 2011 Institute of Medicine report titled “Clinical preventive services for women: closing the gap” lists recommendations on individual preventive services that may be obtained during a well-woman preventive service visit. † Group health plans sponsored by certain religious employers, and group health insurance coverage in connection with such plans, are exempt from the requirement to cover contraceptive services. SOURCE: Adapted from Healthcare.gov. Affordable Care Act Expands Prevention Coverage for Women’s Health and Well-Being. http://www.hrsa.gov/womensguidelines/. Accessed August 8, 2012. | ||
Contraceptive mandate triggers a firestorm
On February 10, 2012, under pressure from religious groups, the Obama Administration offered a religious exemption to the contraception mandate for certain employers and group health plans. Under this “accommodation,” certain religious employers are exempt from the requirement to cover contraceptive services in their group health plans. An employer qualifies for this exemption if it:
- has the inculcation of religious values as one of its purposes
- primarily employs individuals who share its religious tenets
- primarily serves individuals who share its religious tenets, and
- qualifies for nonprofit status under
Internal Revenue Service (IRS) rules. At the same time that the Obama Administration wanted to accommodate employers’ religious beliefs, it also wanted to ensure that every woman would have access to free preventive care, including contraceptive services, regardless of where she works. And so while the Administration requires insurers to offer group health plan coverage without contraceptive coverage to religious-affiliated organizations, it also requires insurers to provide contraceptive coverage directly to individuals covered under the organization’s group health plan with no cost sharing.
This contraceptive mandate—even with the accommodation—has set off a firestorm on Capitol Hill that will eventually be settled in the courts.
Medicaid expansion falls short of original goal
In National Federation of Independent Business v. Sebelius, the plaintiffs asked the Supreme Court to rule on the federal government’s authority to require states to expand their Medicaid programs. Medicaid costs are typically shared by the federal and state governments. Under the ACA, state Medicaid programs were required to cover nearly all individuals who have incomes below 133% of the federal poverty level—$30,656 for a family of four in 2012—paid entirely by the federal government from 2014 through 2016. After that, the federal share gradually declines to, and then stays at, 90%. States that did not expand their Medicaid programs risked losing all federal Medicaid funding.
The Court ruled that the federal government can expand Medicaid but can’t penalize states that don’t accept the expansion mandate—effectively turning the mandate into a state option. States will receive the additional federal funds if they expand coverage, but states that don’t expand will not be penalized by losing existing federal funds for other parts of the program.
Since the ruling, a number of governors have announced that they will not expand their Medicaid programs, including governors of Florida and Louisiana. Those two states alone are home to 20% of all individuals intended to be covered under the Medicaid expansion.
This part of the ACA is particularly important to women because it strikes, for the first time, the requirement that a low-income woman must be pregnant to receive Medicaid coverage.
The figure below shows the dramatic potential improvement in coverage for women if all states fully implement the Medicaid expansion. Time, court decisions, elections, and state budget fights will determine how much of this change is realized for women’s health.

Percentage of insured women will increase under ACA
Percentage of women aged 19 to 64 years who were uninsured in 2009–2012 and under the Affordable Care Act when fully implemented.
SOURCE: Commonwealth Fund. Analysis of the March 2011 and 2010 Current Population Surveys by N. Tilipman and B. Sampat of Columbia University.
Women gain direct access to ObGyns
The ACA guarantees women in all states and all plans direct access to their ObGyns. Before the ACA, women in nine states lacked this guarantee, and women in 16 other states had only limited direct access. Now, a woman can go directly to her ObGyn without having to get a referral from her primary care physician or insurer.
Direct access is especially important because the ACA establishes new delivery systems, such as medical homes and accountable care organizations, designed to capture patients to maximize savings. An ObGyn does not have to be the patient’s primary care provider, and the patient’s access to her ObGyn cannot be limited to a certain number of visits or types of services.
ACA encourages states to cover family-planning services
Under the ACA, states have an easier time covering family-planning services, up to the same eligibility levels as pregnant women. Family planning is still an optional service that a state can choose to extend to women who have incomes above the Medicaid income eligibility level but, before the ACA was enacted, states had to apply to HHS for permission to waive the federal rules, often a very cumbersome process.
Prior to the ACA, 27 states had family planning waivers to provide services to nonpregnant women who had incomes above the Medicaid eligibility level—most at or near 200% of poverty. Now, states can provide family planning services to this population without federal approval.
Don’t miss Dr. Robert L. Barbieri’s October article titled “Gynecologic care across a woman’s life”
Insurance reforms end lifetime limits on coverage
Insurance reforms are important to us and our patients. The better the private health insurance system works—allowing us to provide our best possible care to patients and making sure they can see us when they need our care—the less our nation relies on the public safety net.
Beginning in 2010, the ACA eliminated all lifetime limits on how much insurance companies would cover when beneficiaries get sick; it also bans insurance companies from dropping people from coverage when they get sick. So if your patient has private health insurance and has faithfully paid her premiums and hasn’t committed fraud, her insurer cannot drop her or impose a limit on her coverage once she claims benefits.
This may be especially important for patients who need the most care, such as those who have cancer or another long-term, expensive, and unforeseen diagnosis. Because of this provision, you will not have to worry about your patient losing coverage in the middle of a long course of treatment.
The insurance practice of charging women more than men for equivalent policies ended on January 1, 2011, making insurance more affordable for our patients. Insurers in the individual and small group markets are allowed to vary premiums only for age, geographic location, family size, and tobacco use, not for gender—another important aspect of the law.
2014 is a key year in health reform
Exchanges begin
In 2014, under the ACA, state health insurance exchanges become reality.
An exchange is a marketplace where people can shop for health insurance; private health insurers can market their insurance products in state and multistate exchanges if they comply with new federal insurance reforms established in the ACA and offer the minimum benefits packages established by each state. Exchanges are intended to offer patients a choice of health insurance plans that are affordable, comprehensive, and easy to compare. Low-income individuals will be able to purchase private insurance in the exchanges with the federal premium subsidies or tax credits.
Insurers wanting to market their policies in an exchange may not deny coverage for preexisting conditions, including pregnancy, domestic violence, and previous cesarean delivery. They can’t deny coverage on the basis of an individual’s medical history, health status, genetic information, or disability. And they can’t impose waiting periods longer than 90 days before coverage takes effect, including 9-month waiting periods before maternity coverage.
Essential benefits are established
The ACA sets a minimum standard of health-care coverage that must be included in nearly every private insurance policy. The intent is that every person in the United States, regardless of where they live, who employs them, and what their income is, should have access to the same basic care.
Effective January 1, 2014, all insurance plans, except plans that existed before the ACA was enacted on March 23, 2010, must offer an “essential health benefits” (EHB) package, which must include:
- ambulatory patient services
- emergency services
- hospitalization
- maternity and newborn care
- mental health and substance use disorder services
- prescription drugs
- rehabilitative and habilitative services and devices
- laboratory services
- women’s preventive and wellness services and chronic disease management
- pediatric services, including oral and vision care.
Last December, HHS surprised many by giving states flexibility to design their own EHB packages, as long as the packages included each service on the list.
To choose its EHB package, a state must select a “benchmark” plan from the top- selling plans in four markets: federal and state public employee plans, commercial HMO plans, and small business plans. If a state doesn’t select a benchmark plan, the EHB defaults to the largest small-group market plan in the state. Each state must also choose an EHB package for its Medicaid program using the same 10 benefit categories.
State EHB plans must follow ACA requirements on annual and lifetime dollar limits but may impose limits on the scope and duration of coverage.
As for state-mandated benefits, if a state selects an EHB package that does not include a benefit already mandated by the state, the state must fund coverage for that service on its own—a decision HHS has promised to revisit in 2016.
Abortion decisions reside with the states
ACA requirements regarding abortion coverage 1) take effect in 2014 and 2) apply only to private health insurance plans marketed in the state exchanges that 3) cover abortions beyond those eligible for Medicaid coverage now, which are those that involve cases of rape or incest or that are necessary to save the life of the mother. Medicaid coverage for these categories of abortion is allowed under the Hyde Amendment.
Each insurer marketing a health plan in an exchange can determine whether or not its plan will cover abortion and, if it does, whether coverage will be limited to or go beyond those allowed under the Hyde Amendment. No federal tax or premium subsidies may be used to pay for abortions beyond those permitted by the Hyde Amendment.
The Secretary of HHS must ensure that at least one plan in each state exchange covers abortion, and that at least one plan either covers no abortions or limits abortions to those allowed under the Hyde Amendment. Insurers who offer abortion coverage beyond Hyde have to comply with a number of administrative requirements.
Congress was clear that the ultimate decisions about abortion should be made at the state rather than the federal level, and it gave states the ultimate trump card: Any state can pass legislation that prohibits any plan from offering abortion coverage of any kind within that state’s exchange. Any state can prohibit insurers offering plans within that state’s exchange from including any abortion coverage.
10 additional health provisions under the ACA
1. Creation of women's medical homes
The law points the way for creation of medical homes for women in the Medicare and Medicaid programs. The bill establishes an Innovation Center within the Centers for Medicare and Medicaid Services that has broad authority to evaluate, test, and adopt systems that foster patient-centered care, improve quality, and contain costs under Medicare, Medicaid, and the Children’s Health Insurance Program (CHIP)—and this includes patient-centered medical homes that address women’s unique health needs. ObGyn practices are eligible to participate and to receive additional reimbursement if they do.
2. Smoking-cessation counseling in pregnancy
The framers of the ACA recognized the large negative impact that smoking has on health, especially during pregnancy. Studies suggest that the intervention of a physician—most notably, counseling of the patient to quit smoking—has strong potential to modify this behavior. The new law provides reimbursement for this intervention. There are no copays or deductibles for patients, and smoking-cessation services can include diagnostic, therapeutic, and counseling modalities in addition to prescription of pharmacotherapy.
Before this bill became law, only 24 state Medicaid programs paid ObGyns or other physicians for smoking-cessation counseling of pregnant patients, and five states provided no coverage at all. Now, all pregnant Medicaid patients can get this counseling, and you’ll be paid for this important service.
3. Payments to nonphysician providers in freestanding birth centers
Before the ACA became law, Medicaid was authorized to pay hospitals and other facilities operated by and under the supervision of a physician; no payments were authorized for services of an ambulatory center operated by other health professionals. The ACA authorizes Medicaid payments to state-recognized freestanding birth centers not operated by or under the supervision of a physician. A state that doesn’t currently license birth centers must pass legislation and license these centers before the centers can receive these payments.
Medicaid will also reimburse providers who practice in state-recognized freestanding birth centers, as long as the individuals are practicing within their state’s scope of practice laws and regulations. Because the type of provider is not specified but instead left up to each state’s scope of practice laws and regulations, this provision could allow for separate provider payments for physicians, certified nurse midwives, certified professional midwives, and doulas.
4. Immigrant coverage
Legal immigrants are bound by the individual coverage mandate and must purchase health insurance. These individuals are eligible for income-related premium credits and subsidies for insurance purchased through an exchange. Legal immigrants who are barred from Medicaid during their first 5 years in the United States (by earlier law) are eligible for premium credits only.
Undocumented immigrants are not eligible for Medicaid, premium credits, or subsidies and are barred from purchasing insurance in the exchange, even with their own money.
5. Postpartum depression
Health reform will help bring perinatal and postpartum depression out of the shadows by providing federal funds for research, patient education, and clinical treatment. For example, the federal Department of Health and Human Services (HHS) will:
- conduct research into the causes of, and treatments for, postpartum conditions
- create a national public awareness campaign to increase awareness and knowledge of postpartum depression and psychosis
- provide grants to study the benefits of screening for postpartum depression and psychosis
- establish grants to deliver or enhance outpatient, inpatient, and home-based health and support services, including case management and comprehensive treatment services for individuals with, or at risk for, postpartum conditions.
The National Institute of Mental Health is encouraged to conduct a 10-year longitudinal study on the mental health consequences of pregnancy. This study is intended to focus on perinatal depression.
Community health centers will be eligible for grants in 2012 (as they were in 2011) to the tune of $3 million for inpatient and outpatient counseling and services.
And a federal public awareness campaign will educate the public through radio and television ads.
These endeavors point to the need for ObGyns to familiarize themselves with postpartum depression—if they aren’t already well versed in the subject—because patients are likely to become more aware of this issue and look to their ObGyns for answers.
6. Maternal home visits
Congress established a new Maternal, Infant, and Early Childhood Home Visiting program to improve maternal and fetal health in underserved areas of our country. This program will provide funds to states, tribes, and territories to develop and implement evidence-based home-visitation models to reduce infant and maternal mortality and its causes by producing improvements in:
- prenatal, maternal, and newborn health
- child health and development
- parenting skills
- school readiness
- juvenile delinquency
- family economic self-sufficiency.
These programs will have to demonstrate effectiveness and improved outcomes. HHS recently requested suggestions on ways of demonstrating the effectiveness of home-visiting program models for pregnant women, expectant fathers, and caregivers of children from birth through entry into kindergarten.
The law appropriates $350 million to this program in 2012 and $400 million in both 2013 and 2014.
7. Assistance for pregnant students
A new Pregnancy Assistance Fund—$25 million annually over 10 years (fiscal years 2010–2019)—requires the Secretary of HHS (in collaboration with the Secretary of Education) to establish a state grant program to help pregnant and parenting teens and young women. The aim of this program is to help teens who become pregnant and who choose to bring their pregnancies to term or keep their babies, or both, to stay in school. Grants will go to institutions of higher education, high schools and community service centers, as well as state attorneys general.
Institutions that receive grants must work with providers to meet specific practical needs of pregnant or parenting students:
- housing
- childcare
- parenting education
- postpartum counseling
- assistance in finding and accessing needed services
- referrals for prenatal care and delivery, infant or foster care, or adoption.
Funds to attorneys general will be used to combat domestic violence among pregnant teens.
8. Young women's breast cancer
A new program is intended to help educate young women about the importance of breast health and screening, in two ways:
- The National Institutes of Health (NIH) will conduct research to develop and test screening measures for prevention and early detection of breast cancer in women 15 to 44 years old.
- The US Department of HHS will create a national awareness campaign, with $9 million in funding each year from 2010 to 2014, to encourage young women to talk with their doctors about breast cancer and early detection.
ObGyns can expect to see more interest and questions about breast health among young women and their mothers. It pays to be prepared with good information for these important conversations.
9. Personal responsibility education
From 2010 through 2014, each state will receive funds for personal responsibility education programs aimed at reducing pregnancy in youths. Funds are $75 million for each fiscal year, allocated to each state depending on the size of its youth population but not intended to be less than $250,000 per state.
Educational programs eligible for federal funds must include both abstinence and contraception information for prevention of teenage pregnancy and sexually transmitted infections, including HIV/AIDS, as well as three or more adulthood-preparation subjects.
10. Community-based support of Patient-Centered Medical Homes
Federal funding is available to states for the development of community-based health teams to support medical homes run by primary care practices. These teams may include specialists, nurses, pharmacists, nutritionists, dieticians, social workers, behavioral and mental health providers, and physician assistants. Primary care practices in this program function as medical homes and are responsible for addressing a patient’s personal health-care needs. The team links the medical home to community support services for its patients.
Eligible ObGyn practices can qualify as primary care practices, and ObGyns are eligible to serve as specialist members of the community-based health team.
ACA is a mixed bag for ObGyns
Women have much to gain from the provisions of the ACA. It’s also true that many parts of the law are terrible for practicing ObGyns, including the Independent Payment Advisory Board (IPAB) and the absence of meaningful medical liability reform. For more on these issues, see “Is private ObGyn practice on its way out?” which appears in the October 2011 issue of OBG Management (available in the archive at obgmanagement.com). ACOG is committed to working with Congress to repeal or remedy those aspects of the law.
A recent study reveals that almost one-third of physicians are no longer accepting Medicaid patients
If all states expanded Medicaid to cover people with incomes at or below 138% of the federal poverty level in 2014, as the Affordable Care Act (ACA) proposes, 23 million people would become eligible for the program.1
That statistic prompts important questions:
- Would the health-care workforce be able to meet the demand of caring for all these new patients?
- Would it be willing?
A recent analysis of data from 4,326 office-based physicians suggests that the answer to both questions is “No”: Almost one-third of these providers were already declining to accept new Medicaid patients in 2011.2
Although 96% of physicians in the analysis accepted new patients in 2011, the percentage of physicians accepting new patients covered by Medicaid was lower (69%), as was the percentage accepting new self-paying patients (91.7%), patients covered by Medicare (83%), and patients with private insurance (82%).2
Physicians who were in solo practice were 23.5% less likely to accept new Medicaid patients, compared with those who practiced in an office with 10 or more other physicians.2
The data from this study come from the 2011 National Ambulatory Medical Care Survey Electronic Medical Records Supplement, a survey conducted by the Centers for Disease Control and Prevention National Center for Health Statistics. The survey included questions exploring whether physicians were accepting new patients.2
Earlier studies have found that the low reimbursement levels for care delivered through Medicaid has deterred many physicians from accepting patients.3
The view in the ObGyn specialty
The findings of this analysis were not broken down by specialty—only by primary care versus non–primary care. To get an idea of conditions in the ObGyn specialty, OBG Management surveyed the members of its Virtual Board of Editors (VBE). Of the 117 members contacted, 61 responded—a response rate of 52.1%. Roughly three-quarters (75.4%) reported that they currently treat patients covered by Medicaid, but only 60.7% are accepting new patients covered by Medicaid. Twenty-one percent of respondents reported that they have not and will not accept patients covered by Medicaid.
When asked to comment on their level of satisfaction with Medicaid, the most common response among VBE members was dissatisfaction due to “insufficient reimbursement.”
“I am not satisfied with Medicaid,” commented one VBE member. “The reimbursement is terrible….I have certainly thought of stopping care for Medicaid patients and, if Congress ever allows the big cuts to reimbursement that are threatened every year, I think I would stop.”
Another VBE member reported extreme dissatisfaction with Medicaid because of “lousy” reimbursement. He also pointed to “all the paperwork and crazy regulations that require inordinate time and additional personnel just to handle….and then [the claim] gets denied for reasons beyond reason.” He added that physicians who do accept Medicaid “are on the fast track to sainthood.”
Other reasons for refusing to accept patients with Medicaid (or, if Medicaid was accepted, for high levels of aggravation with the program):
- payment rejections
- too many different categories of coverage “that patients are completely uninformed about”
- difficulty finding a specialist who will manage high-risk patients covered by Medicaid
- red tape
- the complex health problems that Medicaid patients tend to have, compared with patients who have other types of coverage.
One VBE member summed up his feelings in one word: “Phooey.”
Several VBE members suggested that health reform should focus on the Medicaid program.
“These plans are just sucking up the state’s money and paying docs peanuts and their administrators big bucks!” wrote Mary Vanko, MD, of Munster, Indiana.
“I’m tired of how much Medicaid is being abused by people,” commented another VBE member. “People using other people’s cards, people with regular insurance getting Medicaid to cover their copays. The whole system needs reform!”
Some physicians were satisfied with Medicaid
Among the respondents were several who reported being satisfied with the program, including one who called the experience “good” and another who reported being “shielded from the reimbursement issues.”
“I have no problems with Medicaid,” wrote another.
—Janelle Yates, Senior Editor
References
1. Kenney GM, Dubay L, Zuckerman S, Huntress M. Making the Medicaid Expansion an ACA Option: How Many Low-Income Americans Could Remain Uninsured? Washington, DC: Urban Institute Health Policy Center; June 29, 2012. http://www.urban.org/UploadedPDF/412606-Making-the-Medicaid-Expansion-an-ACA-Option.pdf. Accessed August 18, 2012.
2. Decker SL. In 2011 nearly one-third of physicians said they would not accept new Medicaid patients, but rising fees may help. Health Affairs. 2012;31(8):1673–1679.
3. Centers for Disease Control and Prevention. QuickStats: percentage of office-based physicians accepting new patients, by types of payment accepted—United States, 1999–2000 and 2008–2009. MMWR Morb Mortal Wkly Rep. 2011;60(27):928.
We want to hear from you! Tell us what you think.
Pregnancy in Lupus Poses Unique Challenges
CHICAGO – The risk of active disease in pregnant women with systemic lupus erythematosus far outweighs the risks of most medications.
Indeed, the risk of pregnancy loss doubles if lupus is active during pregnancy and jumps fourfold if the autoimmune disease is active in the 3 months before conception.
"My general rule is that the inflammation of active lupus is more dangerous to a pregnancy than medications," said Dr. Megan Clowse, director of the Duke University Autoimmunity in Pregnancy Registry in Durham, N.C. "We don’t have a medication that will cause a 40% pregnancy loss. So I think it’s important to continue medications within this population, although some drugs are certainly better than others."
She advocates the use of hydroxychloroquine (Plaquenil) as the best way to prevent a systemic lupus erythematosus (SLE) flare. It’s a pregnancy category C drug because of reports of hearing abnormalities in children exposed to high doses of chloroquine (Aralen), but there are no clear fetal risks with hydroxychloroquine itself, particularly at the doses used by rheumatologists, she said. Moreover, several reports have suggested that hydroxychloroquine discontinuation during pregnancy may precipitate SLE flares, thereby worsening pregnancy prognosis.
The one exception to hydroxychloroquine use in SLE is the patient who’s been off the drug for years and is no longer symptomatic but becomes pregnant. "I don’t force those women back on their hydroxychloroquine, but basically everybody else should be on it," Dr. Clowse said at a symposium sponsored by the American College of Rheumatology.
For women with more active SLE, consider the use of azathioprine (Azasan, Imuran). It is a pregnancy category D drug due to the risk for fetal immunosuppression at delivery, but its use in SLE pregnancy is supported by extensive data among renal transplant patients, who have a higher rate of prematurity than SLE patients.
Azathioprine is most effective in preventing SLE flare in pregnancy, but Dr. Clowse said she has not been as impressed with its ability to treat flares during pregnancy. In her own recent study, azathioprine did not prevent pregnancy loss, and was actually associated with higher rates of loss among women with high-activity SLE.
When SLE flares occur, they need to be treated promptly, she stressed. Prednisone is probably the most effective treatment, although intravenous immunoglobulin may be a reasonable option. Prednisone is metabolized by the placenta, allowing only 10% of the maternal dose to reach the fetus. In contrast, the fluorinated corticosteroid betamethasone (Betnesol injection) is not metabolized and is transferred to the fetus, so it should not be used during pregnancy. Prednisone may decrease birth weight and is associated with a modest, threefold increased risk for cleft lip and palate, but this is really a first-trimester risk, she noted.
Drugs to avoid in pregnancy include cyclophosphamide (Cytoxan), mycophenolate mofetil (CellCept), and belimumab (Benlysta). A review of 26 pregnancies in 18 renal transplant patients receiving mycophenolate reported that 11 ended in spontaneous abortions and 10 in preterm births, and there were 4 cases of congenital anomalies (Transplantation 2006;82:1698-702). Not only was the 26% birth defect rate staggering compared with the 3% seen in the general population, but three of the infants had microtia. This was not simply underdeveloped pinnae, but the ears were located in the wrong place on the head, resulting in the babies being deaf, Dr. Clowse warned.
Although teratogenicity has not been observed in animals on belimumab, GlaxoSmithKline recently launched a global Benlysta Pregnancy Registry to prospectively collect data on birth defects and other pregnancy outcomes from women receiving belimumab within the 4 months prior to and/or during pregnancy (telephone: 1-877-681-6296).
Dr. Clowse said she recently began putting most of her pregnant SLE patients on aspirin 81 mg/day to prevent preeclampsia. Data from high-risk, nonlupus patients suggest aspirin may decrease the risk of preterm birth, preeclampsia, maternal hypertension, and low-birth weight without increasing the risk of congenital anomalies or ductus arteriosus closure observed with regular aspirin.
"It also, to be honest, takes away that whole discussion of low anticardiolipin antibodies and what do you do," she added.
Pregnant women with SLE should be closely monitored by a rheumatologist and an obstetrician skilled in high-risk pregnancies. Key parameters to monitor are hypertension, particularly in the first trimester, as this can increase the risk of pregnancy loss, preterm birth, and preeclampsia by up to 40%; and proteinuria, which may rise modestly during pregnancy due to increased renal function. More than a doubling or a 1-g increase of proteinuria, however, should raise the alarm, Dr. Clowse said.
To achieve the best possible pregnancy outcome, women with SLE should be advised to avoid pregnancy when SLE is active. Dr. Clowse said it’s recently come to her attention that in North Carolina and other parts of the United States where abstinence-only education has been the law of the land for the past dozen years, this may mean having that first conversation about contraception with a patient already in her 20s.
"I think it’s really important that we address this with our rheumatology patients," she said.
Dr. Clowse said progesterone-only contraceptives are probably the safest method in SLE patients, with medroxyprogesterone injection (Depo-Provera) generally well tolerated, and the 3-year etonogestrel implant (Implanon) is better tolerated than the levonorgestrel implant (Norplant). She also advises her SLE patients to use barrier methods and to pick up levonorgestrel (Plan B One-Step) on the way home from the clinic to have on hand for emergencies.
Dr. Clowse reported consulting for UCB and receiving grant support from the Arthritis Foundation.
CHICAGO – The risk of active disease in pregnant women with systemic lupus erythematosus far outweighs the risks of most medications.
Indeed, the risk of pregnancy loss doubles if lupus is active during pregnancy and jumps fourfold if the autoimmune disease is active in the 3 months before conception.
"My general rule is that the inflammation of active lupus is more dangerous to a pregnancy than medications," said Dr. Megan Clowse, director of the Duke University Autoimmunity in Pregnancy Registry in Durham, N.C. "We don’t have a medication that will cause a 40% pregnancy loss. So I think it’s important to continue medications within this population, although some drugs are certainly better than others."
She advocates the use of hydroxychloroquine (Plaquenil) as the best way to prevent a systemic lupus erythematosus (SLE) flare. It’s a pregnancy category C drug because of reports of hearing abnormalities in children exposed to high doses of chloroquine (Aralen), but there are no clear fetal risks with hydroxychloroquine itself, particularly at the doses used by rheumatologists, she said. Moreover, several reports have suggested that hydroxychloroquine discontinuation during pregnancy may precipitate SLE flares, thereby worsening pregnancy prognosis.
The one exception to hydroxychloroquine use in SLE is the patient who’s been off the drug for years and is no longer symptomatic but becomes pregnant. "I don’t force those women back on their hydroxychloroquine, but basically everybody else should be on it," Dr. Clowse said at a symposium sponsored by the American College of Rheumatology.
For women with more active SLE, consider the use of azathioprine (Azasan, Imuran). It is a pregnancy category D drug due to the risk for fetal immunosuppression at delivery, but its use in SLE pregnancy is supported by extensive data among renal transplant patients, who have a higher rate of prematurity than SLE patients.
Azathioprine is most effective in preventing SLE flare in pregnancy, but Dr. Clowse said she has not been as impressed with its ability to treat flares during pregnancy. In her own recent study, azathioprine did not prevent pregnancy loss, and was actually associated with higher rates of loss among women with high-activity SLE.
When SLE flares occur, they need to be treated promptly, she stressed. Prednisone is probably the most effective treatment, although intravenous immunoglobulin may be a reasonable option. Prednisone is metabolized by the placenta, allowing only 10% of the maternal dose to reach the fetus. In contrast, the fluorinated corticosteroid betamethasone (Betnesol injection) is not metabolized and is transferred to the fetus, so it should not be used during pregnancy. Prednisone may decrease birth weight and is associated with a modest, threefold increased risk for cleft lip and palate, but this is really a first-trimester risk, she noted.
Drugs to avoid in pregnancy include cyclophosphamide (Cytoxan), mycophenolate mofetil (CellCept), and belimumab (Benlysta). A review of 26 pregnancies in 18 renal transplant patients receiving mycophenolate reported that 11 ended in spontaneous abortions and 10 in preterm births, and there were 4 cases of congenital anomalies (Transplantation 2006;82:1698-702). Not only was the 26% birth defect rate staggering compared with the 3% seen in the general population, but three of the infants had microtia. This was not simply underdeveloped pinnae, but the ears were located in the wrong place on the head, resulting in the babies being deaf, Dr. Clowse warned.
Although teratogenicity has not been observed in animals on belimumab, GlaxoSmithKline recently launched a global Benlysta Pregnancy Registry to prospectively collect data on birth defects and other pregnancy outcomes from women receiving belimumab within the 4 months prior to and/or during pregnancy (telephone: 1-877-681-6296).
Dr. Clowse said she recently began putting most of her pregnant SLE patients on aspirin 81 mg/day to prevent preeclampsia. Data from high-risk, nonlupus patients suggest aspirin may decrease the risk of preterm birth, preeclampsia, maternal hypertension, and low-birth weight without increasing the risk of congenital anomalies or ductus arteriosus closure observed with regular aspirin.
"It also, to be honest, takes away that whole discussion of low anticardiolipin antibodies and what do you do," she added.
Pregnant women with SLE should be closely monitored by a rheumatologist and an obstetrician skilled in high-risk pregnancies. Key parameters to monitor are hypertension, particularly in the first trimester, as this can increase the risk of pregnancy loss, preterm birth, and preeclampsia by up to 40%; and proteinuria, which may rise modestly during pregnancy due to increased renal function. More than a doubling or a 1-g increase of proteinuria, however, should raise the alarm, Dr. Clowse said.
To achieve the best possible pregnancy outcome, women with SLE should be advised to avoid pregnancy when SLE is active. Dr. Clowse said it’s recently come to her attention that in North Carolina and other parts of the United States where abstinence-only education has been the law of the land for the past dozen years, this may mean having that first conversation about contraception with a patient already in her 20s.
"I think it’s really important that we address this with our rheumatology patients," she said.
Dr. Clowse said progesterone-only contraceptives are probably the safest method in SLE patients, with medroxyprogesterone injection (Depo-Provera) generally well tolerated, and the 3-year etonogestrel implant (Implanon) is better tolerated than the levonorgestrel implant (Norplant). She also advises her SLE patients to use barrier methods and to pick up levonorgestrel (Plan B One-Step) on the way home from the clinic to have on hand for emergencies.
Dr. Clowse reported consulting for UCB and receiving grant support from the Arthritis Foundation.
CHICAGO – The risk of active disease in pregnant women with systemic lupus erythematosus far outweighs the risks of most medications.
Indeed, the risk of pregnancy loss doubles if lupus is active during pregnancy and jumps fourfold if the autoimmune disease is active in the 3 months before conception.
"My general rule is that the inflammation of active lupus is more dangerous to a pregnancy than medications," said Dr. Megan Clowse, director of the Duke University Autoimmunity in Pregnancy Registry in Durham, N.C. "We don’t have a medication that will cause a 40% pregnancy loss. So I think it’s important to continue medications within this population, although some drugs are certainly better than others."
She advocates the use of hydroxychloroquine (Plaquenil) as the best way to prevent a systemic lupus erythematosus (SLE) flare. It’s a pregnancy category C drug because of reports of hearing abnormalities in children exposed to high doses of chloroquine (Aralen), but there are no clear fetal risks with hydroxychloroquine itself, particularly at the doses used by rheumatologists, she said. Moreover, several reports have suggested that hydroxychloroquine discontinuation during pregnancy may precipitate SLE flares, thereby worsening pregnancy prognosis.
The one exception to hydroxychloroquine use in SLE is the patient who’s been off the drug for years and is no longer symptomatic but becomes pregnant. "I don’t force those women back on their hydroxychloroquine, but basically everybody else should be on it," Dr. Clowse said at a symposium sponsored by the American College of Rheumatology.
For women with more active SLE, consider the use of azathioprine (Azasan, Imuran). It is a pregnancy category D drug due to the risk for fetal immunosuppression at delivery, but its use in SLE pregnancy is supported by extensive data among renal transplant patients, who have a higher rate of prematurity than SLE patients.
Azathioprine is most effective in preventing SLE flare in pregnancy, but Dr. Clowse said she has not been as impressed with its ability to treat flares during pregnancy. In her own recent study, azathioprine did not prevent pregnancy loss, and was actually associated with higher rates of loss among women with high-activity SLE.
When SLE flares occur, they need to be treated promptly, she stressed. Prednisone is probably the most effective treatment, although intravenous immunoglobulin may be a reasonable option. Prednisone is metabolized by the placenta, allowing only 10% of the maternal dose to reach the fetus. In contrast, the fluorinated corticosteroid betamethasone (Betnesol injection) is not metabolized and is transferred to the fetus, so it should not be used during pregnancy. Prednisone may decrease birth weight and is associated with a modest, threefold increased risk for cleft lip and palate, but this is really a first-trimester risk, she noted.
Drugs to avoid in pregnancy include cyclophosphamide (Cytoxan), mycophenolate mofetil (CellCept), and belimumab (Benlysta). A review of 26 pregnancies in 18 renal transplant patients receiving mycophenolate reported that 11 ended in spontaneous abortions and 10 in preterm births, and there were 4 cases of congenital anomalies (Transplantation 2006;82:1698-702). Not only was the 26% birth defect rate staggering compared with the 3% seen in the general population, but three of the infants had microtia. This was not simply underdeveloped pinnae, but the ears were located in the wrong place on the head, resulting in the babies being deaf, Dr. Clowse warned.
Although teratogenicity has not been observed in animals on belimumab, GlaxoSmithKline recently launched a global Benlysta Pregnancy Registry to prospectively collect data on birth defects and other pregnancy outcomes from women receiving belimumab within the 4 months prior to and/or during pregnancy (telephone: 1-877-681-6296).
Dr. Clowse said she recently began putting most of her pregnant SLE patients on aspirin 81 mg/day to prevent preeclampsia. Data from high-risk, nonlupus patients suggest aspirin may decrease the risk of preterm birth, preeclampsia, maternal hypertension, and low-birth weight without increasing the risk of congenital anomalies or ductus arteriosus closure observed with regular aspirin.
"It also, to be honest, takes away that whole discussion of low anticardiolipin antibodies and what do you do," she added.
Pregnant women with SLE should be closely monitored by a rheumatologist and an obstetrician skilled in high-risk pregnancies. Key parameters to monitor are hypertension, particularly in the first trimester, as this can increase the risk of pregnancy loss, preterm birth, and preeclampsia by up to 40%; and proteinuria, which may rise modestly during pregnancy due to increased renal function. More than a doubling or a 1-g increase of proteinuria, however, should raise the alarm, Dr. Clowse said.
To achieve the best possible pregnancy outcome, women with SLE should be advised to avoid pregnancy when SLE is active. Dr. Clowse said it’s recently come to her attention that in North Carolina and other parts of the United States where abstinence-only education has been the law of the land for the past dozen years, this may mean having that first conversation about contraception with a patient already in her 20s.
"I think it’s really important that we address this with our rheumatology patients," she said.
Dr. Clowse said progesterone-only contraceptives are probably the safest method in SLE patients, with medroxyprogesterone injection (Depo-Provera) generally well tolerated, and the 3-year etonogestrel implant (Implanon) is better tolerated than the levonorgestrel implant (Norplant). She also advises her SLE patients to use barrier methods and to pick up levonorgestrel (Plan B One-Step) on the way home from the clinic to have on hand for emergencies.
Dr. Clowse reported consulting for UCB and receiving grant support from the Arthritis Foundation.
EXPERT ANALYSIS FROM A SYMPOSIUM SPONSORED BY THE AMERICAN COLLEGE OF RHEUMATOLOGY
Ovarian Cortex Autografts in Cancer Survivors Yield Live Births
SANTA BARBARA, CALIF. – Ovarian cortex orthotransplantation has resulted in the live births of at least 21 babies to cancer survivors in Europe, where the technique was pioneered and is being refined, Dr. Antonio Pellicer reported at a meeting on in vitro fertilization and embryo transfer.
Unlike the freezing of oocytes or embryos to preserve potential fertility, which requires ovarian stimulation, the ovarian cortex can be harvested from a cancer patient without delay, permitting immediate initiation of chemotherapy and/or radiation therapy. The tissue is cryopreserved until the patient is in remission.
If cancer treatment results in premature ovarian failure and the patient wishes to become pregnant, her autologous ovarian cortex can then be reintroduced.
Ovarian function generally resumes within 3-4 months, said Dr. Pellicer, professor of obstetrics and gynecology and dean of the medical school at the University of Valencia (Spain). Follicle stimulating hormone rates do not reach normal levels, but are sufficient in many cases for resumption of menses and pregnancy, either naturally or through assisted reproductive techniques.
The technique is currently believed to be safe for breast cancer patients and those with Hodgkin’s and non-Hodgkin’s lymphoma, based on histologic and immunologic evaluations of harvested ovarian tissue, Dr. Pellicer said at the meeting, which was sponsored by the University of California, Los Angeles.
It is considered unsafe for patients with leukemia, as metastatic cells might well circulate through the bloodstream to the ovaries. Because of its highly metastatic potential, Ewing’s sarcoma is also considered a contraindication for the procedure, according to Dr. Pellicer.
The technique offers hope, potentially, for prepubertal girls and adolescents with other types of cancer, as well as adult cancer patients, although much remains unknown about the viability and usefulness of the treatment, explained Dr. Pellicer.
At the Valencia Program of Fertility Preservation, more than 600 cancer patients from across Spain have undergone removal of the ovarian cortex around the time of diagnosis, said Dr. Pellicer.
He reported on results in 583 of those patients who were treated since 2005, 55% of whom had been diagnosed with breast cancer.
Regular menses and fertility were restored in some patients who received ovarian autografts, said Dr. Pellicer. In all, 16 pregnancies and 3 live births have occurred, some following in vitro fertilization and some following natural conception.
Those results, along with published studies from programs in France, Germany, Denmark, Belgium, and other countries, indicate that at least 21 and perhaps 23 or more live births have resulted from the technique.
The problem, as Dr. Pellicer sees it, is a lack of cohesive follow-up or evidence that would put those births into perspective.
"We don’t know the number of failed attempts," he said. "There are no registries. There are no real data. Are we doing something which is really helpful? Or are the unsuccessful cases more [typical] than the successful cases?"
"This is a concern to me," he said.
Responding to a question from an audience member, Dr. Pellicer acknowledged that the removal of one ovarian cortex prior to cancer treatment might diminish fertility potential rather than enhance it, because some cancer patients conceive naturally following remission.
Dr. Pellicer reported that he had no relevant financial relationships to disclose.
SANTA BARBARA, CALIF. – Ovarian cortex orthotransplantation has resulted in the live births of at least 21 babies to cancer survivors in Europe, where the technique was pioneered and is being refined, Dr. Antonio Pellicer reported at a meeting on in vitro fertilization and embryo transfer.
Unlike the freezing of oocytes or embryos to preserve potential fertility, which requires ovarian stimulation, the ovarian cortex can be harvested from a cancer patient without delay, permitting immediate initiation of chemotherapy and/or radiation therapy. The tissue is cryopreserved until the patient is in remission.
If cancer treatment results in premature ovarian failure and the patient wishes to become pregnant, her autologous ovarian cortex can then be reintroduced.
Ovarian function generally resumes within 3-4 months, said Dr. Pellicer, professor of obstetrics and gynecology and dean of the medical school at the University of Valencia (Spain). Follicle stimulating hormone rates do not reach normal levels, but are sufficient in many cases for resumption of menses and pregnancy, either naturally or through assisted reproductive techniques.
The technique is currently believed to be safe for breast cancer patients and those with Hodgkin’s and non-Hodgkin’s lymphoma, based on histologic and immunologic evaluations of harvested ovarian tissue, Dr. Pellicer said at the meeting, which was sponsored by the University of California, Los Angeles.
It is considered unsafe for patients with leukemia, as metastatic cells might well circulate through the bloodstream to the ovaries. Because of its highly metastatic potential, Ewing’s sarcoma is also considered a contraindication for the procedure, according to Dr. Pellicer.
The technique offers hope, potentially, for prepubertal girls and adolescents with other types of cancer, as well as adult cancer patients, although much remains unknown about the viability and usefulness of the treatment, explained Dr. Pellicer.
At the Valencia Program of Fertility Preservation, more than 600 cancer patients from across Spain have undergone removal of the ovarian cortex around the time of diagnosis, said Dr. Pellicer.
He reported on results in 583 of those patients who were treated since 2005, 55% of whom had been diagnosed with breast cancer.
Regular menses and fertility were restored in some patients who received ovarian autografts, said Dr. Pellicer. In all, 16 pregnancies and 3 live births have occurred, some following in vitro fertilization and some following natural conception.
Those results, along with published studies from programs in France, Germany, Denmark, Belgium, and other countries, indicate that at least 21 and perhaps 23 or more live births have resulted from the technique.
The problem, as Dr. Pellicer sees it, is a lack of cohesive follow-up or evidence that would put those births into perspective.
"We don’t know the number of failed attempts," he said. "There are no registries. There are no real data. Are we doing something which is really helpful? Or are the unsuccessful cases more [typical] than the successful cases?"
"This is a concern to me," he said.
Responding to a question from an audience member, Dr. Pellicer acknowledged that the removal of one ovarian cortex prior to cancer treatment might diminish fertility potential rather than enhance it, because some cancer patients conceive naturally following remission.
Dr. Pellicer reported that he had no relevant financial relationships to disclose.
SANTA BARBARA, CALIF. – Ovarian cortex orthotransplantation has resulted in the live births of at least 21 babies to cancer survivors in Europe, where the technique was pioneered and is being refined, Dr. Antonio Pellicer reported at a meeting on in vitro fertilization and embryo transfer.
Unlike the freezing of oocytes or embryos to preserve potential fertility, which requires ovarian stimulation, the ovarian cortex can be harvested from a cancer patient without delay, permitting immediate initiation of chemotherapy and/or radiation therapy. The tissue is cryopreserved until the patient is in remission.
If cancer treatment results in premature ovarian failure and the patient wishes to become pregnant, her autologous ovarian cortex can then be reintroduced.
Ovarian function generally resumes within 3-4 months, said Dr. Pellicer, professor of obstetrics and gynecology and dean of the medical school at the University of Valencia (Spain). Follicle stimulating hormone rates do not reach normal levels, but are sufficient in many cases for resumption of menses and pregnancy, either naturally or through assisted reproductive techniques.
The technique is currently believed to be safe for breast cancer patients and those with Hodgkin’s and non-Hodgkin’s lymphoma, based on histologic and immunologic evaluations of harvested ovarian tissue, Dr. Pellicer said at the meeting, which was sponsored by the University of California, Los Angeles.
It is considered unsafe for patients with leukemia, as metastatic cells might well circulate through the bloodstream to the ovaries. Because of its highly metastatic potential, Ewing’s sarcoma is also considered a contraindication for the procedure, according to Dr. Pellicer.
The technique offers hope, potentially, for prepubertal girls and adolescents with other types of cancer, as well as adult cancer patients, although much remains unknown about the viability and usefulness of the treatment, explained Dr. Pellicer.
At the Valencia Program of Fertility Preservation, more than 600 cancer patients from across Spain have undergone removal of the ovarian cortex around the time of diagnosis, said Dr. Pellicer.
He reported on results in 583 of those patients who were treated since 2005, 55% of whom had been diagnosed with breast cancer.
Regular menses and fertility were restored in some patients who received ovarian autografts, said Dr. Pellicer. In all, 16 pregnancies and 3 live births have occurred, some following in vitro fertilization and some following natural conception.
Those results, along with published studies from programs in France, Germany, Denmark, Belgium, and other countries, indicate that at least 21 and perhaps 23 or more live births have resulted from the technique.
The problem, as Dr. Pellicer sees it, is a lack of cohesive follow-up or evidence that would put those births into perspective.
"We don’t know the number of failed attempts," he said. "There are no registries. There are no real data. Are we doing something which is really helpful? Or are the unsuccessful cases more [typical] than the successful cases?"
"This is a concern to me," he said.
Responding to a question from an audience member, Dr. Pellicer acknowledged that the removal of one ovarian cortex prior to cancer treatment might diminish fertility potential rather than enhance it, because some cancer patients conceive naturally following remission.
Dr. Pellicer reported that he had no relevant financial relationships to disclose.
AT A MEETING ON IN VITRO FERTILIZATION AND EMBRYO TRANSFER
Major Finding: Sixteen pregnancies and three live births have occurred, some following in vitro fertilization and some following natural conception.
Data Source: This was a study of 583 patients who received ovarian autografts since 2005, 55% of whom had been diagnosed with breast cancer.
Disclosures: Dr. Pellicer reported that he had no relevant financial relationships to disclose.
Breast Cancer During Pregnancy Can Be Treated as in Nonpregnant Women
It appears that breast cancer diagnosed during pregnancy can be treated much the same as breast cancer diagnosed in nonpregnant women without substantially raising the risks to mother or child, according to a study published online August 16 in the Lancet Oncology .
This conclusion, from an observational study involving 447 European women included in registries of cancers diagnosed during pregnancy, must still be validated in other studies. But until then, the current evidence indicates that pregnancy outcomes are not significantly different between women who receive breast cancer chemotherapy during the second or third trimesters and those who wait until after delivery to start treatment, said Dr. Sibylle Loibl of the German Breast Group, Neu-Isenburg, Germany, and her associates.
In this study, infants exposed to their mothers’ breast cancer chemotherapy while in utero had slightly lower birth weights and slightly more complications than those not exposed to chemotherapy, but these differences were not clinically significant.
Breast cancer diagnosed during pregnancy is rare, estimated to occur in less than 1% of breast cancers in Europe. But its incidence is increasing in high-income countries due to the trend of women delaying childbirth until they are older, when breast cancer is more prevalent.
The German Breast Group established its Breast Cancer During Pregnancy registry in 2003 and expanded it to include cases in the Netherlands, the United Kingdom, Poland, Italy, and the Czech Republic in 2009. In the same time period, Belgium also established a registry of all cancers diagnosed during pregnancy. Dr. Loibl and her colleagues assessed outcomes in 447 cases from these registries in which women were diagnosed as having early (413 patients) or metastatic (34 patients) breast cancer while pregnant (Lancet Oncol. 2012 Aug. 15 [doi:10.1016/S1470-2045(12)70261-9]).
The median gestational age at diagnosis was 24 weeks (range, 5-40 weeks), and the median age of the women was 33 years (range, 22-51 years).
Data on chemotherapy were available for 368 women. Of these, 197 received chemotherapy while pregnant and 171 received it after delivery.
Overall, 1,187 cycles of chemotherapy were given, and 63% of these were given during pregnancy. The women received a median of four cycles (range, one to eight cycles) during pregnancy.
A total of 90% of those treated during pregnancy received an anthracyline; 8% received a combination of cyclophosphamide, methotrexate, and fluorouracil; and 7% received a taxane. None of the women received trastuzumab, endocrine therapy, or radiotherapy during pregnancy.
Women with early breast cancer who opted for chemotherapy during pregnancy tended to have more advanced disease, with more unfavorable tumor stage and nodal status, than did those who chose to begin chemotherapy after delivery. After the data were adjusted to account for this difference, the researchers found no significant difference between the two groups in disease-free or overall survival.
The estimated 3-year disease-free survival was 70.2% in women with early disease who underwent chemotherapy while pregnant and 74.3% in those who waited until after delivery. Similarly, the estimated overall 3-year survival was 84.9% in women with early breast cancer who underwent chemotherapy while pregnant and 87.4% in those who delayed chemotherapy until after delivery.
The estimated 5-year disease-free survival was 61.1% in women who had chemotherapy while pregnant and 64.4% in those who waited, and the estimated 5-year overall survival was 77% and 82.4%, respectively.
Data were available for 373 newborns, of whom 203 had been exposed to chemotherapy in utero and 170 had not.
Birth weight was slightly lower in the exposed than in the nonexposed infants, but this difference was judged to be "clinically irrelevant" because it didn’t affect the health of the babies, Dr. Loibl and her associates said.
Moreover, there were no significant differences between the two groups in major birth defects, infant height, Apgar scores, hemoglobin concentration, leukocyte counts, thrombocyte counts, or alopecia. And there was no significant difference in the proportion of infants discharged with their mothers (34% vs. 41%).
Adverse events occurred more often when chemotherapy was received during pregnancy (15%) than when it was delayed (4%). However, this difference was attributed to the higher rates of preterm labor and premature rupture of the membrane among exposed pregnancies. "Most complications were reported in babies who were delivered prematurely, regardless of exposure to chemotherapy," the investigators said.
The data were not adequate to determine why women who received chemotherapy had a higher rate of preterm delivery. Both physical stress and psychological stress may have played a role, and it is possible that women who received chemotherapy were more prone to infections that may have triggered labor.
In addition, the cytotoxic agents themselves may have hastened labor through some as yet unknown mechanism. However, the rate of preeclampsia was similar between the two groups, so oxidative stress, which is known to be induced by cytoxic agents, was not responsible.
Further study of the data being collected in the registries of cancers diagnosed during pregnancy will likely shed light on these issues. Dr. Loibl and her colleagues are now performing a matched-pair analysis to assess whether the prognosis of breast cancer in nonpregnant women differs from that in pregnant women when the latter are treated according to current guidelines.
This study was supported by BANSS Foundation, University Hospital Frankfurt, the German Breast Group, Research Foundation-Flanders, Clinical Research Fund-UZ Gasthuisberg, and the Belgian Cancer Plan. No financial conflicts of interest were reported.
Future studies should address not just the toxic effects of chemotherapy during pregnancy but also the pharmacokinetics of cytotoxic agents in pregnant women, because the physiologic changes of pregnancy can greatly affect drug disposition, said Dr. Olivier Mir and Dr. Paul Berveiller.
"Whether doses should be increased in this population is uncertain because such increases could result in severe thrombocytopenia, neutropenia, and infection, with potentially devastating consequences for both mother and baby," they noted.
More research also is needed to determine whether the slightly increased fetal risks identified by Dr. Loibl and her colleagues could be minimized with better drug selection and dosing, they added.
Dr. Mir and Dr. Berveiller are in the Cancer Associated With Pregnancy Network, Paris. Dr. Mir is also in the department of medical oncology at Hôpital Cochin at the Université René Descartes, Paris. Dr. Mir reported ties to Roche, Pfizer, and Servier. These remarks were taken from their editorial comment accompanying Dr. Loibl’s report (Lancet Oncol. 2012 Aug. 15 [doi:10.1016/S1470-2045(12)70331-5]).
Future studies should address not just the toxic effects of chemotherapy during pregnancy but also the pharmacokinetics of cytotoxic agents in pregnant women, because the physiologic changes of pregnancy can greatly affect drug disposition, said Dr. Olivier Mir and Dr. Paul Berveiller.
"Whether doses should be increased in this population is uncertain because such increases could result in severe thrombocytopenia, neutropenia, and infection, with potentially devastating consequences for both mother and baby," they noted.
More research also is needed to determine whether the slightly increased fetal risks identified by Dr. Loibl and her colleagues could be minimized with better drug selection and dosing, they added.
Dr. Mir and Dr. Berveiller are in the Cancer Associated With Pregnancy Network, Paris. Dr. Mir is also in the department of medical oncology at Hôpital Cochin at the Université René Descartes, Paris. Dr. Mir reported ties to Roche, Pfizer, and Servier. These remarks were taken from their editorial comment accompanying Dr. Loibl’s report (Lancet Oncol. 2012 Aug. 15 [doi:10.1016/S1470-2045(12)70331-5]).
Future studies should address not just the toxic effects of chemotherapy during pregnancy but also the pharmacokinetics of cytotoxic agents in pregnant women, because the physiologic changes of pregnancy can greatly affect drug disposition, said Dr. Olivier Mir and Dr. Paul Berveiller.
"Whether doses should be increased in this population is uncertain because such increases could result in severe thrombocytopenia, neutropenia, and infection, with potentially devastating consequences for both mother and baby," they noted.
More research also is needed to determine whether the slightly increased fetal risks identified by Dr. Loibl and her colleagues could be minimized with better drug selection and dosing, they added.
Dr. Mir and Dr. Berveiller are in the Cancer Associated With Pregnancy Network, Paris. Dr. Mir is also in the department of medical oncology at Hôpital Cochin at the Université René Descartes, Paris. Dr. Mir reported ties to Roche, Pfizer, and Servier. These remarks were taken from their editorial comment accompanying Dr. Loibl’s report (Lancet Oncol. 2012 Aug. 15 [doi:10.1016/S1470-2045(12)70331-5]).
It appears that breast cancer diagnosed during pregnancy can be treated much the same as breast cancer diagnosed in nonpregnant women without substantially raising the risks to mother or child, according to a study published online August 16 in the Lancet Oncology .
This conclusion, from an observational study involving 447 European women included in registries of cancers diagnosed during pregnancy, must still be validated in other studies. But until then, the current evidence indicates that pregnancy outcomes are not significantly different between women who receive breast cancer chemotherapy during the second or third trimesters and those who wait until after delivery to start treatment, said Dr. Sibylle Loibl of the German Breast Group, Neu-Isenburg, Germany, and her associates.
In this study, infants exposed to their mothers’ breast cancer chemotherapy while in utero had slightly lower birth weights and slightly more complications than those not exposed to chemotherapy, but these differences were not clinically significant.
Breast cancer diagnosed during pregnancy is rare, estimated to occur in less than 1% of breast cancers in Europe. But its incidence is increasing in high-income countries due to the trend of women delaying childbirth until they are older, when breast cancer is more prevalent.
The German Breast Group established its Breast Cancer During Pregnancy registry in 2003 and expanded it to include cases in the Netherlands, the United Kingdom, Poland, Italy, and the Czech Republic in 2009. In the same time period, Belgium also established a registry of all cancers diagnosed during pregnancy. Dr. Loibl and her colleagues assessed outcomes in 447 cases from these registries in which women were diagnosed as having early (413 patients) or metastatic (34 patients) breast cancer while pregnant (Lancet Oncol. 2012 Aug. 15 [doi:10.1016/S1470-2045(12)70261-9]).
The median gestational age at diagnosis was 24 weeks (range, 5-40 weeks), and the median age of the women was 33 years (range, 22-51 years).
Data on chemotherapy were available for 368 women. Of these, 197 received chemotherapy while pregnant and 171 received it after delivery.
Overall, 1,187 cycles of chemotherapy were given, and 63% of these were given during pregnancy. The women received a median of four cycles (range, one to eight cycles) during pregnancy.
A total of 90% of those treated during pregnancy received an anthracyline; 8% received a combination of cyclophosphamide, methotrexate, and fluorouracil; and 7% received a taxane. None of the women received trastuzumab, endocrine therapy, or radiotherapy during pregnancy.
Women with early breast cancer who opted for chemotherapy during pregnancy tended to have more advanced disease, with more unfavorable tumor stage and nodal status, than did those who chose to begin chemotherapy after delivery. After the data were adjusted to account for this difference, the researchers found no significant difference between the two groups in disease-free or overall survival.
The estimated 3-year disease-free survival was 70.2% in women with early disease who underwent chemotherapy while pregnant and 74.3% in those who waited until after delivery. Similarly, the estimated overall 3-year survival was 84.9% in women with early breast cancer who underwent chemotherapy while pregnant and 87.4% in those who delayed chemotherapy until after delivery.
The estimated 5-year disease-free survival was 61.1% in women who had chemotherapy while pregnant and 64.4% in those who waited, and the estimated 5-year overall survival was 77% and 82.4%, respectively.
Data were available for 373 newborns, of whom 203 had been exposed to chemotherapy in utero and 170 had not.
Birth weight was slightly lower in the exposed than in the nonexposed infants, but this difference was judged to be "clinically irrelevant" because it didn’t affect the health of the babies, Dr. Loibl and her associates said.
Moreover, there were no significant differences between the two groups in major birth defects, infant height, Apgar scores, hemoglobin concentration, leukocyte counts, thrombocyte counts, or alopecia. And there was no significant difference in the proportion of infants discharged with their mothers (34% vs. 41%).
Adverse events occurred more often when chemotherapy was received during pregnancy (15%) than when it was delayed (4%). However, this difference was attributed to the higher rates of preterm labor and premature rupture of the membrane among exposed pregnancies. "Most complications were reported in babies who were delivered prematurely, regardless of exposure to chemotherapy," the investigators said.
The data were not adequate to determine why women who received chemotherapy had a higher rate of preterm delivery. Both physical stress and psychological stress may have played a role, and it is possible that women who received chemotherapy were more prone to infections that may have triggered labor.
In addition, the cytotoxic agents themselves may have hastened labor through some as yet unknown mechanism. However, the rate of preeclampsia was similar between the two groups, so oxidative stress, which is known to be induced by cytoxic agents, was not responsible.
Further study of the data being collected in the registries of cancers diagnosed during pregnancy will likely shed light on these issues. Dr. Loibl and her colleagues are now performing a matched-pair analysis to assess whether the prognosis of breast cancer in nonpregnant women differs from that in pregnant women when the latter are treated according to current guidelines.
This study was supported by BANSS Foundation, University Hospital Frankfurt, the German Breast Group, Research Foundation-Flanders, Clinical Research Fund-UZ Gasthuisberg, and the Belgian Cancer Plan. No financial conflicts of interest were reported.
It appears that breast cancer diagnosed during pregnancy can be treated much the same as breast cancer diagnosed in nonpregnant women without substantially raising the risks to mother or child, according to a study published online August 16 in the Lancet Oncology .
This conclusion, from an observational study involving 447 European women included in registries of cancers diagnosed during pregnancy, must still be validated in other studies. But until then, the current evidence indicates that pregnancy outcomes are not significantly different between women who receive breast cancer chemotherapy during the second or third trimesters and those who wait until after delivery to start treatment, said Dr. Sibylle Loibl of the German Breast Group, Neu-Isenburg, Germany, and her associates.
In this study, infants exposed to their mothers’ breast cancer chemotherapy while in utero had slightly lower birth weights and slightly more complications than those not exposed to chemotherapy, but these differences were not clinically significant.
Breast cancer diagnosed during pregnancy is rare, estimated to occur in less than 1% of breast cancers in Europe. But its incidence is increasing in high-income countries due to the trend of women delaying childbirth until they are older, when breast cancer is more prevalent.
The German Breast Group established its Breast Cancer During Pregnancy registry in 2003 and expanded it to include cases in the Netherlands, the United Kingdom, Poland, Italy, and the Czech Republic in 2009. In the same time period, Belgium also established a registry of all cancers diagnosed during pregnancy. Dr. Loibl and her colleagues assessed outcomes in 447 cases from these registries in which women were diagnosed as having early (413 patients) or metastatic (34 patients) breast cancer while pregnant (Lancet Oncol. 2012 Aug. 15 [doi:10.1016/S1470-2045(12)70261-9]).
The median gestational age at diagnosis was 24 weeks (range, 5-40 weeks), and the median age of the women was 33 years (range, 22-51 years).
Data on chemotherapy were available for 368 women. Of these, 197 received chemotherapy while pregnant and 171 received it after delivery.
Overall, 1,187 cycles of chemotherapy were given, and 63% of these were given during pregnancy. The women received a median of four cycles (range, one to eight cycles) during pregnancy.
A total of 90% of those treated during pregnancy received an anthracyline; 8% received a combination of cyclophosphamide, methotrexate, and fluorouracil; and 7% received a taxane. None of the women received trastuzumab, endocrine therapy, or radiotherapy during pregnancy.
Women with early breast cancer who opted for chemotherapy during pregnancy tended to have more advanced disease, with more unfavorable tumor stage and nodal status, than did those who chose to begin chemotherapy after delivery. After the data were adjusted to account for this difference, the researchers found no significant difference between the two groups in disease-free or overall survival.
The estimated 3-year disease-free survival was 70.2% in women with early disease who underwent chemotherapy while pregnant and 74.3% in those who waited until after delivery. Similarly, the estimated overall 3-year survival was 84.9% in women with early breast cancer who underwent chemotherapy while pregnant and 87.4% in those who delayed chemotherapy until after delivery.
The estimated 5-year disease-free survival was 61.1% in women who had chemotherapy while pregnant and 64.4% in those who waited, and the estimated 5-year overall survival was 77% and 82.4%, respectively.
Data were available for 373 newborns, of whom 203 had been exposed to chemotherapy in utero and 170 had not.
Birth weight was slightly lower in the exposed than in the nonexposed infants, but this difference was judged to be "clinically irrelevant" because it didn’t affect the health of the babies, Dr. Loibl and her associates said.
Moreover, there were no significant differences between the two groups in major birth defects, infant height, Apgar scores, hemoglobin concentration, leukocyte counts, thrombocyte counts, or alopecia. And there was no significant difference in the proportion of infants discharged with their mothers (34% vs. 41%).
Adverse events occurred more often when chemotherapy was received during pregnancy (15%) than when it was delayed (4%). However, this difference was attributed to the higher rates of preterm labor and premature rupture of the membrane among exposed pregnancies. "Most complications were reported in babies who were delivered prematurely, regardless of exposure to chemotherapy," the investigators said.
The data were not adequate to determine why women who received chemotherapy had a higher rate of preterm delivery. Both physical stress and psychological stress may have played a role, and it is possible that women who received chemotherapy were more prone to infections that may have triggered labor.
In addition, the cytotoxic agents themselves may have hastened labor through some as yet unknown mechanism. However, the rate of preeclampsia was similar between the two groups, so oxidative stress, which is known to be induced by cytoxic agents, was not responsible.
Further study of the data being collected in the registries of cancers diagnosed during pregnancy will likely shed light on these issues. Dr. Loibl and her colleagues are now performing a matched-pair analysis to assess whether the prognosis of breast cancer in nonpregnant women differs from that in pregnant women when the latter are treated according to current guidelines.
This study was supported by BANSS Foundation, University Hospital Frankfurt, the German Breast Group, Research Foundation-Flanders, Clinical Research Fund-UZ Gasthuisberg, and the Belgian Cancer Plan. No financial conflicts of interest were reported.
FROM THE LANCET ONCOLOGY
Major Finding: Rates of low birth weight and adverse events were slightly higher when women underwent breast cancer chemotherapy while pregnant than when they delayed chemotherapy until after delivery, but the small differences were deemed clinically irrelevant.
Data Source: An observational study of pregnancy outcomes in European women diagnosed as having breast cancer during pregnancy, of whom 197 received chemotherapy while pregnant and 171 delayed chemotherapy until after delivery.
Disclosures: This study was supported by BANSS Foundation, University Hospital Frankfurt, the German Breast Group, Research Foundation-Flanders, Clinical Research Fund-UZ Gasthuisberg, and the Belgian Cancer Plan. No financial conflicts of interest were reported.
Complacency Is the Enemy in Transvaginal Follicle Aspiration
SANTA BARBARA, CALIF. – A third of the audience attending a specialty conference on in vitro fertilization reported hospitalizing a patient for pelvic infection following transvaginal follicle aspiration, and the same percentage reported performing a laparoscopy or laparotomy for bleeding following the procedure.
Bowel and ureteral injuries and retroperitoneal hematomas also were reported, but at far lower rates, Dr. David R. Meldrum said at a conference on in vitro fertilization and embryo transfer sponsored by the University of California, Los Angeles.
While the instant survey was unscientific and represented complications only among specialists who perform many transvaginal procedures, the high rate of serious complications was noteworthy, said Dr. Meldrum, scientific director of Reproductive Partners Medical Group in Redondo Beach, Calif., and director of the annual course on in vitro fertilization and embryo transfer.
"I’ve talked to several people who feel that complications of this procedure are underreported," he said.
"The last thing you want to be is complacent," Dr. Meldrum continued. "As a pilot, I know ... if you ever get complacent about something you should be paying maximal attention to," it will "bite you in the bottom."
Dr. Meldrum noted that transvaginal oocyte retrieval is highly efficacious and generally well tolerated under intravenous sedation. It is not, however, a risk-free procedure.
To reduce the potential for complications, he recommended using a 16- or 17-gauge needle with a very sharp tip and rinsing both the needles and collection tubing before use.
The ovary should be positioned very close to the transducer. Use "firm, constant pressure" of the probe, abdominal pressure, and a tenaculum, he suggested.
During the procedure, the needle should be rotated during movement to avoid bending the needle in any direction.
The ovary should be entered high, away from lower structures. "Don’t have a hint of anything between the ovary and the posterior pelvic wall," he said. In case of positioning difficulties, "keep in mind you can come across from the other side."
"Keep the needle tip well visualized and within the ovary," he said.
Dr. Meldrum reminded the audience that retroperitoneal bleeding can usually be managed by pressure from above when it is recognized during a procedure, but that symptoms may develop hours after the procedure. Abscesses may present up to 6 weeks following a transvaginal follicle aspiration.
Even during normal, successful aspirations, severe pain may occur during the procedure and for several days afterward, in a small percentage of patients.
Dr. Meldrum noted that Tylenol with codeine, a commonly prescribed analgesic, will be ineffective in approximately 1 of 10 patients who don’t metabolize codeine.
He reported that he had no relevant conflicts of interest.
SANTA BARBARA, CALIF. – A third of the audience attending a specialty conference on in vitro fertilization reported hospitalizing a patient for pelvic infection following transvaginal follicle aspiration, and the same percentage reported performing a laparoscopy or laparotomy for bleeding following the procedure.
Bowel and ureteral injuries and retroperitoneal hematomas also were reported, but at far lower rates, Dr. David R. Meldrum said at a conference on in vitro fertilization and embryo transfer sponsored by the University of California, Los Angeles.
While the instant survey was unscientific and represented complications only among specialists who perform many transvaginal procedures, the high rate of serious complications was noteworthy, said Dr. Meldrum, scientific director of Reproductive Partners Medical Group in Redondo Beach, Calif., and director of the annual course on in vitro fertilization and embryo transfer.
"I’ve talked to several people who feel that complications of this procedure are underreported," he said.
"The last thing you want to be is complacent," Dr. Meldrum continued. "As a pilot, I know ... if you ever get complacent about something you should be paying maximal attention to," it will "bite you in the bottom."
Dr. Meldrum noted that transvaginal oocyte retrieval is highly efficacious and generally well tolerated under intravenous sedation. It is not, however, a risk-free procedure.
To reduce the potential for complications, he recommended using a 16- or 17-gauge needle with a very sharp tip and rinsing both the needles and collection tubing before use.
The ovary should be positioned very close to the transducer. Use "firm, constant pressure" of the probe, abdominal pressure, and a tenaculum, he suggested.
During the procedure, the needle should be rotated during movement to avoid bending the needle in any direction.
The ovary should be entered high, away from lower structures. "Don’t have a hint of anything between the ovary and the posterior pelvic wall," he said. In case of positioning difficulties, "keep in mind you can come across from the other side."
"Keep the needle tip well visualized and within the ovary," he said.
Dr. Meldrum reminded the audience that retroperitoneal bleeding can usually be managed by pressure from above when it is recognized during a procedure, but that symptoms may develop hours after the procedure. Abscesses may present up to 6 weeks following a transvaginal follicle aspiration.
Even during normal, successful aspirations, severe pain may occur during the procedure and for several days afterward, in a small percentage of patients.
Dr. Meldrum noted that Tylenol with codeine, a commonly prescribed analgesic, will be ineffective in approximately 1 of 10 patients who don’t metabolize codeine.
He reported that he had no relevant conflicts of interest.
SANTA BARBARA, CALIF. – A third of the audience attending a specialty conference on in vitro fertilization reported hospitalizing a patient for pelvic infection following transvaginal follicle aspiration, and the same percentage reported performing a laparoscopy or laparotomy for bleeding following the procedure.
Bowel and ureteral injuries and retroperitoneal hematomas also were reported, but at far lower rates, Dr. David R. Meldrum said at a conference on in vitro fertilization and embryo transfer sponsored by the University of California, Los Angeles.
While the instant survey was unscientific and represented complications only among specialists who perform many transvaginal procedures, the high rate of serious complications was noteworthy, said Dr. Meldrum, scientific director of Reproductive Partners Medical Group in Redondo Beach, Calif., and director of the annual course on in vitro fertilization and embryo transfer.
"I’ve talked to several people who feel that complications of this procedure are underreported," he said.
"The last thing you want to be is complacent," Dr. Meldrum continued. "As a pilot, I know ... if you ever get complacent about something you should be paying maximal attention to," it will "bite you in the bottom."
Dr. Meldrum noted that transvaginal oocyte retrieval is highly efficacious and generally well tolerated under intravenous sedation. It is not, however, a risk-free procedure.
To reduce the potential for complications, he recommended using a 16- or 17-gauge needle with a very sharp tip and rinsing both the needles and collection tubing before use.
The ovary should be positioned very close to the transducer. Use "firm, constant pressure" of the probe, abdominal pressure, and a tenaculum, he suggested.
During the procedure, the needle should be rotated during movement to avoid bending the needle in any direction.
The ovary should be entered high, away from lower structures. "Don’t have a hint of anything between the ovary and the posterior pelvic wall," he said. In case of positioning difficulties, "keep in mind you can come across from the other side."
"Keep the needle tip well visualized and within the ovary," he said.
Dr. Meldrum reminded the audience that retroperitoneal bleeding can usually be managed by pressure from above when it is recognized during a procedure, but that symptoms may develop hours after the procedure. Abscesses may present up to 6 weeks following a transvaginal follicle aspiration.
Even during normal, successful aspirations, severe pain may occur during the procedure and for several days afterward, in a small percentage of patients.
Dr. Meldrum noted that Tylenol with codeine, a commonly prescribed analgesic, will be ineffective in approximately 1 of 10 patients who don’t metabolize codeine.
He reported that he had no relevant conflicts of interest.
EXPERT ANALYSIS FROM A CONFERENCE ON IN VITRO FERTILIZATION AND EMBRYO TRANSFER





