User login
Advisory on youth mental health crisis gets mixed reviews
The advisory on youth mental health from Surgeon General Vivek Murthy, MD, casts a necessary spotlight on the crisis, clinical psychiatrists say. But some think it could have produced more specifics about funding and payment parity for reimbursement.
The 53-page advisory says that about one in five U.S. children and adolescents aged 3-17 suffer from a mental, emotional, developmental, or behavioral disorder. In the decade before COVID, feelings of sadness and hopelessness, as well as suicidal behaviors, were on the rise. The pandemic has exacerbated symptoms of anxiety, depression, and other mental health issues in young people. Compared with 2019, ED visits in early 2021 for suspected suicide attempts rose 51% in adolescent girls and 4% in boys. “Depressive and anxiety symptoms doubled during the pandemic,” the advisory said.
Scope of the advisory
The advisory, released Dec. 7, covers all sectors and considers all social and policy factors that might be contributing to this crisis, said Jessica (Jessi) Gold, MD, MS, an assistant professor in the department of psychiatry at Washington University, St. Louis.
“It is always possible to reimagine health care to be more patient centered and mental health forward.” But changes of this magnitude take time, Dr. Gold, also director of wellness, engagement, and outreach at the university, said in an interview.
She has seen the impact of the pandemic firsthand in her clinic among students and frontline health care workers aged 18-30. People in that age group “feel everything deeply,” Dr. Gold said. Emotions tied to COVID-19 are just a part of it. Confounding factors, such as climate change, racism, and school shootings all contribute to their overall mental health.
Some children and adolescents with social anxiety have fared better during the pandemic, but those who are part of demographic groups such as racial and ethnic minorities, LGBTQ individuals, low-income youth, and those involved in juvenile justice or welfare systems face a higher risk of mental health challenges, the pandemic notwithstanding.
In her work with schools, Denese Shervington, MD, MPH, has witnessed more mental health challenges related to isolation and separation. “There’s an overall worry about the loss of what used to be, the seeming predictability and certainty of prepandemic life,” said Dr. Shervington, clinical professor of psychiatry at Tulane University, and president and CEO of the Institute for Women and Ethnic Studies, both in New Orleans.
A systems of care plan
The advisory lists actionable items for health care and 10 other industry sectors to improve mental health of children and young adults.
Health care organizations and professionals were advised to take the following six steps:
- Implement trauma-informed care principles and other prevention strategies. This may involve referring patients to resources such as economic and legal supports, school enrichment programs, and educating families on healthy child development in the clinic.
- Routinely screen children for mental health challenges and risk factors such as adverse childhood experiences during primary care well-visits or annual physicals, or at schools or EDs. Primary care physicians should use principles of trauma-informed care to conduct these screenings.
- Screen parents, caregivers, and other family members for depression, intimate partner violence, substance use, and other challenges. These can be done in tandem with broader assessments of social determinants of health such as food or housing insecurity.
- Combine efforts of clinical staff with trusted community partners and child welfare and juvenile justice. Hospital-based violence intervention programs, for example, identify patients at risk of repeat violent injury and refer them to hospital- and community-based resources.
- Build multidisciplinary teams, enlisting children and families to develop services that are tailored to their needs for screening and treatment. Such services should reflect cultural diversity and offered in multiple languages.
- Support the well-being of mental health workers and community leaders to foster their ability to help youth and their families.
Dr. Murthy is talking about a “systems of care” approach, in which all sectors that touch children and youth – not just health care – must work together and do their jobs effectively but collaboratively to address this public health crisis, said Aradhana (Bela) Sood MD, MSHA, FAACAP, senior professor of child mental health policy at Virginia Commonwealth University, Richmond. “An investment in infrastructure support of positive mental health in early childhood, be it in schools, communities, or family well-being will lead to a future where illness is not the result of major preventable societal factors, such as a lack of social supports and trauma.”
Changes will ‘take a lot of buy-in’
The recommendations are actionable in the real world – but there are a lot of them, said Dr. Gold. Dr. Murthy doesn’t specify what the plan is to accomplish these metrics or fund them, she added. He “has money and funders like foundations as steps, but foundations have also suffered in the pandemic, so it is not that simple.” Many of these changes are wide in scope and will take a lot of buy-in.
Dr. Shervington would like to have seen more of a focus on educator well-being, given that young people spend a lot of time in educational settings.
“My organization just completed a study in New Orleans that showed teachers having elevated levels of trauma-based conditions since the pandemic,” she said. Schools are indeed a key place to support holistic mental health by focusing on school climate, Dr. Sood added. “If school administrators became uniformly consistent with recognizing the importance of psychological wellness as a prerequisite of good learning, they will create environments where teachers are keenly aware of a child’s mental wellness and make reduction of bullying, wellness check-ins, [and] school-based mental health clinics a priority.
“These are ways nonmedical, community-based supports can enhance student well-being, and reduce depression and other mental health conditions,” Dr. Sood added.
Child psychiatrists stretched ‘even thinner’
Despite mental health parity rules, health plans have not been held accountable. That failure, combined with excessive demands for prior authorization for mental health treatments “have led to dangerous shortages of psychiatrists able to accept insurance,” said Paul S. Nestadt, MD, an assistant professor and public mental health researcher at Johns Hopkins University, Baltimore.
“This is particularly true for child psychiatrists, who are stretched even thinner than those of us in general practice,” Dr. Nestadt said.
While he doesn’t address it head on, Dr. Murthy uses classic parity language when he states that “mental health is no less important than physical health,” said Dr. Nestadt, who consulted with the surgeon general on developing this advisory. “While many of us would have liked to see parity highlighted more directly, this advisory was designed to be an overview.”
Highlighting social media, gun violence
Dr. Nestadt said he was pleased that the advisory emphasized the importance of restricting access to lethal means in preventing youth suicide.
“With youth suicide rates rising faster than in other age groups, and suicide mortality tied so closely to method availability, the surgeon general made the right choice in highlighting the role of guns in suicide,” he said.
The advisory also discussed the role of media and social media companies in addressing the crisis, which is important, said Dr. Gold.
“I believe very strongly that the way we talk about and portray mental health in the media matters,” she said. “I have seen it matter in the clinic with patients. They’ll wonder if someone will think they are now violent if they are diagnosed with a mental illness. Stories change the narrative.”
While the advisory isn’t perfect, the state of youth mental health “will only get worse if we don’t do something,” noted Dr. Gold. “It is critical that this is validated and discussed at the highest level and messages like Dr. Murthy’s get heard.”
Dr. Gold, Dr. Shervington, and Dr. Sood had no disclosures. Dr. Nestadt disclosed serving as a consultant to the surgeon general advisory.
The advisory on youth mental health from Surgeon General Vivek Murthy, MD, casts a necessary spotlight on the crisis, clinical psychiatrists say. But some think it could have produced more specifics about funding and payment parity for reimbursement.
The 53-page advisory says that about one in five U.S. children and adolescents aged 3-17 suffer from a mental, emotional, developmental, or behavioral disorder. In the decade before COVID, feelings of sadness and hopelessness, as well as suicidal behaviors, were on the rise. The pandemic has exacerbated symptoms of anxiety, depression, and other mental health issues in young people. Compared with 2019, ED visits in early 2021 for suspected suicide attempts rose 51% in adolescent girls and 4% in boys. “Depressive and anxiety symptoms doubled during the pandemic,” the advisory said.
Scope of the advisory
The advisory, released Dec. 7, covers all sectors and considers all social and policy factors that might be contributing to this crisis, said Jessica (Jessi) Gold, MD, MS, an assistant professor in the department of psychiatry at Washington University, St. Louis.
“It is always possible to reimagine health care to be more patient centered and mental health forward.” But changes of this magnitude take time, Dr. Gold, also director of wellness, engagement, and outreach at the university, said in an interview.
She has seen the impact of the pandemic firsthand in her clinic among students and frontline health care workers aged 18-30. People in that age group “feel everything deeply,” Dr. Gold said. Emotions tied to COVID-19 are just a part of it. Confounding factors, such as climate change, racism, and school shootings all contribute to their overall mental health.
Some children and adolescents with social anxiety have fared better during the pandemic, but those who are part of demographic groups such as racial and ethnic minorities, LGBTQ individuals, low-income youth, and those involved in juvenile justice or welfare systems face a higher risk of mental health challenges, the pandemic notwithstanding.
In her work with schools, Denese Shervington, MD, MPH, has witnessed more mental health challenges related to isolation and separation. “There’s an overall worry about the loss of what used to be, the seeming predictability and certainty of prepandemic life,” said Dr. Shervington, clinical professor of psychiatry at Tulane University, and president and CEO of the Institute for Women and Ethnic Studies, both in New Orleans.
A systems of care plan
The advisory lists actionable items for health care and 10 other industry sectors to improve mental health of children and young adults.
Health care organizations and professionals were advised to take the following six steps:
- Implement trauma-informed care principles and other prevention strategies. This may involve referring patients to resources such as economic and legal supports, school enrichment programs, and educating families on healthy child development in the clinic.
- Routinely screen children for mental health challenges and risk factors such as adverse childhood experiences during primary care well-visits or annual physicals, or at schools or EDs. Primary care physicians should use principles of trauma-informed care to conduct these screenings.
- Screen parents, caregivers, and other family members for depression, intimate partner violence, substance use, and other challenges. These can be done in tandem with broader assessments of social determinants of health such as food or housing insecurity.
- Combine efforts of clinical staff with trusted community partners and child welfare and juvenile justice. Hospital-based violence intervention programs, for example, identify patients at risk of repeat violent injury and refer them to hospital- and community-based resources.
- Build multidisciplinary teams, enlisting children and families to develop services that are tailored to their needs for screening and treatment. Such services should reflect cultural diversity and offered in multiple languages.
- Support the well-being of mental health workers and community leaders to foster their ability to help youth and their families.
Dr. Murthy is talking about a “systems of care” approach, in which all sectors that touch children and youth – not just health care – must work together and do their jobs effectively but collaboratively to address this public health crisis, said Aradhana (Bela) Sood MD, MSHA, FAACAP, senior professor of child mental health policy at Virginia Commonwealth University, Richmond. “An investment in infrastructure support of positive mental health in early childhood, be it in schools, communities, or family well-being will lead to a future where illness is not the result of major preventable societal factors, such as a lack of social supports and trauma.”
Changes will ‘take a lot of buy-in’
The recommendations are actionable in the real world – but there are a lot of them, said Dr. Gold. Dr. Murthy doesn’t specify what the plan is to accomplish these metrics or fund them, she added. He “has money and funders like foundations as steps, but foundations have also suffered in the pandemic, so it is not that simple.” Many of these changes are wide in scope and will take a lot of buy-in.
Dr. Shervington would like to have seen more of a focus on educator well-being, given that young people spend a lot of time in educational settings.
“My organization just completed a study in New Orleans that showed teachers having elevated levels of trauma-based conditions since the pandemic,” she said. Schools are indeed a key place to support holistic mental health by focusing on school climate, Dr. Sood added. “If school administrators became uniformly consistent with recognizing the importance of psychological wellness as a prerequisite of good learning, they will create environments where teachers are keenly aware of a child’s mental wellness and make reduction of bullying, wellness check-ins, [and] school-based mental health clinics a priority.
“These are ways nonmedical, community-based supports can enhance student well-being, and reduce depression and other mental health conditions,” Dr. Sood added.
Child psychiatrists stretched ‘even thinner’
Despite mental health parity rules, health plans have not been held accountable. That failure, combined with excessive demands for prior authorization for mental health treatments “have led to dangerous shortages of psychiatrists able to accept insurance,” said Paul S. Nestadt, MD, an assistant professor and public mental health researcher at Johns Hopkins University, Baltimore.
“This is particularly true for child psychiatrists, who are stretched even thinner than those of us in general practice,” Dr. Nestadt said.
While he doesn’t address it head on, Dr. Murthy uses classic parity language when he states that “mental health is no less important than physical health,” said Dr. Nestadt, who consulted with the surgeon general on developing this advisory. “While many of us would have liked to see parity highlighted more directly, this advisory was designed to be an overview.”
Highlighting social media, gun violence
Dr. Nestadt said he was pleased that the advisory emphasized the importance of restricting access to lethal means in preventing youth suicide.
“With youth suicide rates rising faster than in other age groups, and suicide mortality tied so closely to method availability, the surgeon general made the right choice in highlighting the role of guns in suicide,” he said.
The advisory also discussed the role of media and social media companies in addressing the crisis, which is important, said Dr. Gold.
“I believe very strongly that the way we talk about and portray mental health in the media matters,” she said. “I have seen it matter in the clinic with patients. They’ll wonder if someone will think they are now violent if they are diagnosed with a mental illness. Stories change the narrative.”
While the advisory isn’t perfect, the state of youth mental health “will only get worse if we don’t do something,” noted Dr. Gold. “It is critical that this is validated and discussed at the highest level and messages like Dr. Murthy’s get heard.”
Dr. Gold, Dr. Shervington, and Dr. Sood had no disclosures. Dr. Nestadt disclosed serving as a consultant to the surgeon general advisory.
The advisory on youth mental health from Surgeon General Vivek Murthy, MD, casts a necessary spotlight on the crisis, clinical psychiatrists say. But some think it could have produced more specifics about funding and payment parity for reimbursement.
The 53-page advisory says that about one in five U.S. children and adolescents aged 3-17 suffer from a mental, emotional, developmental, or behavioral disorder. In the decade before COVID, feelings of sadness and hopelessness, as well as suicidal behaviors, were on the rise. The pandemic has exacerbated symptoms of anxiety, depression, and other mental health issues in young people. Compared with 2019, ED visits in early 2021 for suspected suicide attempts rose 51% in adolescent girls and 4% in boys. “Depressive and anxiety symptoms doubled during the pandemic,” the advisory said.
Scope of the advisory
The advisory, released Dec. 7, covers all sectors and considers all social and policy factors that might be contributing to this crisis, said Jessica (Jessi) Gold, MD, MS, an assistant professor in the department of psychiatry at Washington University, St. Louis.
“It is always possible to reimagine health care to be more patient centered and mental health forward.” But changes of this magnitude take time, Dr. Gold, also director of wellness, engagement, and outreach at the university, said in an interview.
She has seen the impact of the pandemic firsthand in her clinic among students and frontline health care workers aged 18-30. People in that age group “feel everything deeply,” Dr. Gold said. Emotions tied to COVID-19 are just a part of it. Confounding factors, such as climate change, racism, and school shootings all contribute to their overall mental health.
Some children and adolescents with social anxiety have fared better during the pandemic, but those who are part of demographic groups such as racial and ethnic minorities, LGBTQ individuals, low-income youth, and those involved in juvenile justice or welfare systems face a higher risk of mental health challenges, the pandemic notwithstanding.
In her work with schools, Denese Shervington, MD, MPH, has witnessed more mental health challenges related to isolation and separation. “There’s an overall worry about the loss of what used to be, the seeming predictability and certainty of prepandemic life,” said Dr. Shervington, clinical professor of psychiatry at Tulane University, and president and CEO of the Institute for Women and Ethnic Studies, both in New Orleans.
A systems of care plan
The advisory lists actionable items for health care and 10 other industry sectors to improve mental health of children and young adults.
Health care organizations and professionals were advised to take the following six steps:
- Implement trauma-informed care principles and other prevention strategies. This may involve referring patients to resources such as economic and legal supports, school enrichment programs, and educating families on healthy child development in the clinic.
- Routinely screen children for mental health challenges and risk factors such as adverse childhood experiences during primary care well-visits or annual physicals, or at schools or EDs. Primary care physicians should use principles of trauma-informed care to conduct these screenings.
- Screen parents, caregivers, and other family members for depression, intimate partner violence, substance use, and other challenges. These can be done in tandem with broader assessments of social determinants of health such as food or housing insecurity.
- Combine efforts of clinical staff with trusted community partners and child welfare and juvenile justice. Hospital-based violence intervention programs, for example, identify patients at risk of repeat violent injury and refer them to hospital- and community-based resources.
- Build multidisciplinary teams, enlisting children and families to develop services that are tailored to their needs for screening and treatment. Such services should reflect cultural diversity and offered in multiple languages.
- Support the well-being of mental health workers and community leaders to foster their ability to help youth and their families.
Dr. Murthy is talking about a “systems of care” approach, in which all sectors that touch children and youth – not just health care – must work together and do their jobs effectively but collaboratively to address this public health crisis, said Aradhana (Bela) Sood MD, MSHA, FAACAP, senior professor of child mental health policy at Virginia Commonwealth University, Richmond. “An investment in infrastructure support of positive mental health in early childhood, be it in schools, communities, or family well-being will lead to a future where illness is not the result of major preventable societal factors, such as a lack of social supports and trauma.”
Changes will ‘take a lot of buy-in’
The recommendations are actionable in the real world – but there are a lot of them, said Dr. Gold. Dr. Murthy doesn’t specify what the plan is to accomplish these metrics or fund them, she added. He “has money and funders like foundations as steps, but foundations have also suffered in the pandemic, so it is not that simple.” Many of these changes are wide in scope and will take a lot of buy-in.
Dr. Shervington would like to have seen more of a focus on educator well-being, given that young people spend a lot of time in educational settings.
“My organization just completed a study in New Orleans that showed teachers having elevated levels of trauma-based conditions since the pandemic,” she said. Schools are indeed a key place to support holistic mental health by focusing on school climate, Dr. Sood added. “If school administrators became uniformly consistent with recognizing the importance of psychological wellness as a prerequisite of good learning, they will create environments where teachers are keenly aware of a child’s mental wellness and make reduction of bullying, wellness check-ins, [and] school-based mental health clinics a priority.
“These are ways nonmedical, community-based supports can enhance student well-being, and reduce depression and other mental health conditions,” Dr. Sood added.
Child psychiatrists stretched ‘even thinner’
Despite mental health parity rules, health plans have not been held accountable. That failure, combined with excessive demands for prior authorization for mental health treatments “have led to dangerous shortages of psychiatrists able to accept insurance,” said Paul S. Nestadt, MD, an assistant professor and public mental health researcher at Johns Hopkins University, Baltimore.
“This is particularly true for child psychiatrists, who are stretched even thinner than those of us in general practice,” Dr. Nestadt said.
While he doesn’t address it head on, Dr. Murthy uses classic parity language when he states that “mental health is no less important than physical health,” said Dr. Nestadt, who consulted with the surgeon general on developing this advisory. “While many of us would have liked to see parity highlighted more directly, this advisory was designed to be an overview.”
Highlighting social media, gun violence
Dr. Nestadt said he was pleased that the advisory emphasized the importance of restricting access to lethal means in preventing youth suicide.
“With youth suicide rates rising faster than in other age groups, and suicide mortality tied so closely to method availability, the surgeon general made the right choice in highlighting the role of guns in suicide,” he said.
The advisory also discussed the role of media and social media companies in addressing the crisis, which is important, said Dr. Gold.
“I believe very strongly that the way we talk about and portray mental health in the media matters,” she said. “I have seen it matter in the clinic with patients. They’ll wonder if someone will think they are now violent if they are diagnosed with a mental illness. Stories change the narrative.”
While the advisory isn’t perfect, the state of youth mental health “will only get worse if we don’t do something,” noted Dr. Gold. “It is critical that this is validated and discussed at the highest level and messages like Dr. Murthy’s get heard.”
Dr. Gold, Dr. Shervington, and Dr. Sood had no disclosures. Dr. Nestadt disclosed serving as a consultant to the surgeon general advisory.
Telemedicine helps SCD patients survive COVID, but more need access
ATLANTA – , according to an investigator at the annual meeting of the American Society of Hematology.
During the first COVID-19 wave in the summer of 2020, Atlanta’s Grady Sickle Cell Center, the nation’s largest adult sickle cell center, recorded two deaths among the 20 COVID-19_infected patients seen there, said Fuad El Rassi, MD, of Emory University, Atlanta.
Virtual visits, launched to deliver health care needs in the wake of a Georgia’s 2020 statewide shelter-in-place order, helped protect patients from COVID-19 infection, Dr. El Rassi said in a press conference at the meeting.
“The patients’ diligence and awareness to stay home during the pandemic have proven crucial to reducing morbidity and mortality in this vulnerable population,” he said. “The option of having virtual visits for health care delivery was key and should be utilized further in sickle cell care.”
However, virtual visits and other best practices to prevent and treat COVID-19 in patients with sickle cell disease can be challenging to implement outside of large, specialized centers such as Grady.
“The majority of sickle cell patients in major metropolitan areas are not plugged into dedicated sickle cell centers, and that’s a key issue,” said Dr. El Rassi.
“There’s a huge shortage of such clinics around major metropolitan areas, and that restricts things for the general population, unfortunately.”
COVID-19 prevention remains a challenge, no matter where patients are treated. Only about 50% of the center’s sickle cell disease patients are immunized, according to Dr. El Rassi, who added that assessment of vaccine response among those patients is ongoing.
Ifeyinwa (Ify) Osunkwo, MD, MPH, a sickle cell disease specialist, said long-term sustainability of virtual visits depends greatly on states’ continuation of laws or policies that facilitate access to telemedicine. A total of 22 states changed laws or policies during the pandemic to promote access to telemedicine, according to the Commonwealth Fund.
Virtual care is more challenging in states where expanded telemedicine coverage is not available or is ended, said Dr. Osunkwo, director of the Sickle Cell Enterprise at Levine Cancer Institute. The institute is part of Atrium Health, a large health system that operates in four states.
“We are no longer able to do virtual visits for our South Carolinian patients, even though across the border in North Carolina, you can still provide virtual care,” Dr. Osunkwo said in an interview.
“Sickle cell patients suffer from social determinants [of health], so getting to their doctor when they have a regular outpatient visit is kind of hard,” she added. “And having that virtual option actually makes them more adherent, and they have better access to care overall.”
In the study presented at the ASH meeting by Dr. El Rassi and colleagues, there were a total of 55 patients with COVID-19 among the 1,343 sickle cell disease patients they tracked. Of the 55 patients with COVID-19, 28 were female and 27 were male, and 35% were on hydroxyurea for disease modification.
Among these 55 patients with COVID-19, 44 (80%) were hospitalized, and the hospitalizations of 15 (27%) were deemed related to COVID-19 signs and symptoms, Dr. El Rassi said. Twelve of the 55 patients (22%) had emergency visits, including 5 (9%) because of COVID-19 symptoms, he added.
The two deaths from COVID-19 occurred in June and July 2020, said Dr. El Rassi, adding that those patients were among 20 total cases diagnosed from March to September of 2020.
Over the second reported wave of COVID-19, from October 2020 to March 2021, there were no deaths seen among 35 total COVID-19 cases, according to the report at the ASH meeting.
In an interview, Kaitlin Strumph, MD, a sickle cell disease specialist at the Children’s Hospital at Montefiore in New York, noted that patients with sickle cell disease who contract COVID-19 are considered at high risk for morbidity and mortality.
“Patients and providers should not let down their guard,” Dr. Strumph said in an interview. “The best way to protect people from COVID-19 right now is prevention, and vaccinations are the key to further improving outcomes.”
Virtual visits can help bridge gaps in care for patients with sickle cell disease, said Dr. Strumph, given that limited access to care is a large driver of health disparities in this population.
“Telemedicine allows patients to stay home and limit their exposure to COVID-19 out in the community and at the hospital,” she said. “I think most providers feel confident that virtual visits are a huge benefit for the community, and we hope they are here to stay.”
Dr. El Rassi reported disclosures related to Cyclerion, Novartis, Pfizer, Global Blood Therapeutics and bluebird bio.
ATLANTA – , according to an investigator at the annual meeting of the American Society of Hematology.
During the first COVID-19 wave in the summer of 2020, Atlanta’s Grady Sickle Cell Center, the nation’s largest adult sickle cell center, recorded two deaths among the 20 COVID-19_infected patients seen there, said Fuad El Rassi, MD, of Emory University, Atlanta.
Virtual visits, launched to deliver health care needs in the wake of a Georgia’s 2020 statewide shelter-in-place order, helped protect patients from COVID-19 infection, Dr. El Rassi said in a press conference at the meeting.
“The patients’ diligence and awareness to stay home during the pandemic have proven crucial to reducing morbidity and mortality in this vulnerable population,” he said. “The option of having virtual visits for health care delivery was key and should be utilized further in sickle cell care.”
However, virtual visits and other best practices to prevent and treat COVID-19 in patients with sickle cell disease can be challenging to implement outside of large, specialized centers such as Grady.
“The majority of sickle cell patients in major metropolitan areas are not plugged into dedicated sickle cell centers, and that’s a key issue,” said Dr. El Rassi.
“There’s a huge shortage of such clinics around major metropolitan areas, and that restricts things for the general population, unfortunately.”
COVID-19 prevention remains a challenge, no matter where patients are treated. Only about 50% of the center’s sickle cell disease patients are immunized, according to Dr. El Rassi, who added that assessment of vaccine response among those patients is ongoing.
Ifeyinwa (Ify) Osunkwo, MD, MPH, a sickle cell disease specialist, said long-term sustainability of virtual visits depends greatly on states’ continuation of laws or policies that facilitate access to telemedicine. A total of 22 states changed laws or policies during the pandemic to promote access to telemedicine, according to the Commonwealth Fund.
Virtual care is more challenging in states where expanded telemedicine coverage is not available or is ended, said Dr. Osunkwo, director of the Sickle Cell Enterprise at Levine Cancer Institute. The institute is part of Atrium Health, a large health system that operates in four states.
“We are no longer able to do virtual visits for our South Carolinian patients, even though across the border in North Carolina, you can still provide virtual care,” Dr. Osunkwo said in an interview.
“Sickle cell patients suffer from social determinants [of health], so getting to their doctor when they have a regular outpatient visit is kind of hard,” she added. “And having that virtual option actually makes them more adherent, and they have better access to care overall.”
In the study presented at the ASH meeting by Dr. El Rassi and colleagues, there were a total of 55 patients with COVID-19 among the 1,343 sickle cell disease patients they tracked. Of the 55 patients with COVID-19, 28 were female and 27 were male, and 35% were on hydroxyurea for disease modification.
Among these 55 patients with COVID-19, 44 (80%) were hospitalized, and the hospitalizations of 15 (27%) were deemed related to COVID-19 signs and symptoms, Dr. El Rassi said. Twelve of the 55 patients (22%) had emergency visits, including 5 (9%) because of COVID-19 symptoms, he added.
The two deaths from COVID-19 occurred in June and July 2020, said Dr. El Rassi, adding that those patients were among 20 total cases diagnosed from March to September of 2020.
Over the second reported wave of COVID-19, from October 2020 to March 2021, there were no deaths seen among 35 total COVID-19 cases, according to the report at the ASH meeting.
In an interview, Kaitlin Strumph, MD, a sickle cell disease specialist at the Children’s Hospital at Montefiore in New York, noted that patients with sickle cell disease who contract COVID-19 are considered at high risk for morbidity and mortality.
“Patients and providers should not let down their guard,” Dr. Strumph said in an interview. “The best way to protect people from COVID-19 right now is prevention, and vaccinations are the key to further improving outcomes.”
Virtual visits can help bridge gaps in care for patients with sickle cell disease, said Dr. Strumph, given that limited access to care is a large driver of health disparities in this population.
“Telemedicine allows patients to stay home and limit their exposure to COVID-19 out in the community and at the hospital,” she said. “I think most providers feel confident that virtual visits are a huge benefit for the community, and we hope they are here to stay.”
Dr. El Rassi reported disclosures related to Cyclerion, Novartis, Pfizer, Global Blood Therapeutics and bluebird bio.
ATLANTA – , according to an investigator at the annual meeting of the American Society of Hematology.
During the first COVID-19 wave in the summer of 2020, Atlanta’s Grady Sickle Cell Center, the nation’s largest adult sickle cell center, recorded two deaths among the 20 COVID-19_infected patients seen there, said Fuad El Rassi, MD, of Emory University, Atlanta.
Virtual visits, launched to deliver health care needs in the wake of a Georgia’s 2020 statewide shelter-in-place order, helped protect patients from COVID-19 infection, Dr. El Rassi said in a press conference at the meeting.
“The patients’ diligence and awareness to stay home during the pandemic have proven crucial to reducing morbidity and mortality in this vulnerable population,” he said. “The option of having virtual visits for health care delivery was key and should be utilized further in sickle cell care.”
However, virtual visits and other best practices to prevent and treat COVID-19 in patients with sickle cell disease can be challenging to implement outside of large, specialized centers such as Grady.
“The majority of sickle cell patients in major metropolitan areas are not plugged into dedicated sickle cell centers, and that’s a key issue,” said Dr. El Rassi.
“There’s a huge shortage of such clinics around major metropolitan areas, and that restricts things for the general population, unfortunately.”
COVID-19 prevention remains a challenge, no matter where patients are treated. Only about 50% of the center’s sickle cell disease patients are immunized, according to Dr. El Rassi, who added that assessment of vaccine response among those patients is ongoing.
Ifeyinwa (Ify) Osunkwo, MD, MPH, a sickle cell disease specialist, said long-term sustainability of virtual visits depends greatly on states’ continuation of laws or policies that facilitate access to telemedicine. A total of 22 states changed laws or policies during the pandemic to promote access to telemedicine, according to the Commonwealth Fund.
Virtual care is more challenging in states where expanded telemedicine coverage is not available or is ended, said Dr. Osunkwo, director of the Sickle Cell Enterprise at Levine Cancer Institute. The institute is part of Atrium Health, a large health system that operates in four states.
“We are no longer able to do virtual visits for our South Carolinian patients, even though across the border in North Carolina, you can still provide virtual care,” Dr. Osunkwo said in an interview.
“Sickle cell patients suffer from social determinants [of health], so getting to their doctor when they have a regular outpatient visit is kind of hard,” she added. “And having that virtual option actually makes them more adherent, and they have better access to care overall.”
In the study presented at the ASH meeting by Dr. El Rassi and colleagues, there were a total of 55 patients with COVID-19 among the 1,343 sickle cell disease patients they tracked. Of the 55 patients with COVID-19, 28 were female and 27 were male, and 35% were on hydroxyurea for disease modification.
Among these 55 patients with COVID-19, 44 (80%) were hospitalized, and the hospitalizations of 15 (27%) were deemed related to COVID-19 signs and symptoms, Dr. El Rassi said. Twelve of the 55 patients (22%) had emergency visits, including 5 (9%) because of COVID-19 symptoms, he added.
The two deaths from COVID-19 occurred in June and July 2020, said Dr. El Rassi, adding that those patients were among 20 total cases diagnosed from March to September of 2020.
Over the second reported wave of COVID-19, from October 2020 to March 2021, there were no deaths seen among 35 total COVID-19 cases, according to the report at the ASH meeting.
In an interview, Kaitlin Strumph, MD, a sickle cell disease specialist at the Children’s Hospital at Montefiore in New York, noted that patients with sickle cell disease who contract COVID-19 are considered at high risk for morbidity and mortality.
“Patients and providers should not let down their guard,” Dr. Strumph said in an interview. “The best way to protect people from COVID-19 right now is prevention, and vaccinations are the key to further improving outcomes.”
Virtual visits can help bridge gaps in care for patients with sickle cell disease, said Dr. Strumph, given that limited access to care is a large driver of health disparities in this population.
“Telemedicine allows patients to stay home and limit their exposure to COVID-19 out in the community and at the hospital,” she said. “I think most providers feel confident that virtual visits are a huge benefit for the community, and we hope they are here to stay.”
Dr. El Rassi reported disclosures related to Cyclerion, Novartis, Pfizer, Global Blood Therapeutics and bluebird bio.
FROM ASH 2021
iPLEDGE rollout described as a failure, chaotic, and a disaster
The that launched on Dec. 13, and what can be done to fix it.
By most accounts, the rollout was disastrous, chaotic, and a failure. Dermatologists on Twitter and elsewhere are angry and frustrated, with some calling for a temporary halt to the program until the bugs can be ironed out.
On Twitter Dec. 15, the Academy posted: “Due to the unacceptable situation with #iPLEDGE, the @US_FDA has convened an emergency meeting with AADA representatives tomorrow, December 16.”
The switch to a new platform was met with frustration from physicians, pharmacists, and patients alike. The new website crashed repeatedly, with physicians and patients complaining they got locked out or bounced off the platform when they attempted to follow instructions to enter information. Calls to obtain support from a live person often required hours on hold, several said.
The new approach to the isotretinoin risk-mitigation program itself isn’t under fire. It was welcomed by dermatologists and others who had long requested the change. Instead of three risk categories (females of reproductive potential, females not of reproductive potential, and males), there are now two (those who can get pregnant and those who cannot). Advocates for the change said it will make the experience more inclusive for transgender patients. The previous categories, some contended, were a barrier to access to care.
Because isotretinoin (Absorica, Amnesteem, Claravis, others), an oral retinoid used to treat severe forms of acne, is teratogenic, with a high risk of birth defects, and has also been associated with other health issues, those who take the medication who are able to get pregnant must take contraceptive precautions. The risk evaluation and mitigation program (REMS), mandated by the FDA, stipulates that physicians, patients, and pharmacists prescribing, using, or dispensing the drug must all be registered with requirements that include the use of two forms of an effective contraceptive and regular pregnancy tests by those capable of becoming pregnant.
A day of frustration
Before navigating the new website, a new log-on name was needed, said Ilona J. Frieden, MD, chair of the AADA’s iPLEDGE Workgroup and professor of dermatology at the University of California, San Francisco. “They made you create a month-day-year date of personal significance.” When she tried to log on, she got locked out, she said in an interview.
The transition from the old website to the new, which Dr. Frieden said is now administered by a different vendor, was done quickly. The previous website shut down Dec. 10, and the new one launched Dec. 13, the first day for the new approach.
“A slower rollout would have helped,” Dr. Frieden said. While she and other dermatologists said they offered input previously on how to make the transition go more smoothly, no one seemed to want that help. “We did have a listening session with the FDA,” Dr. Frieden said. That was before the scheduled meeting of Dec. 16.
Neil S. Goldberg, MD, a dermatologist in Westchester County, New York, also was frustrated with the rollout. “The week before the transition, one of my staff had to call iPLEDGE. They had a 177-minute wait to get to a human.
“They want us to register patients online now instead of signing forms in the office, but the links to view, download, or print don’t work,” Dr. Goldberg said in an interview.
This was after receiving information from the iPLEDGE REMS program, which stated, “The iPLEDGE REMS website will be updated to a modernized platform. All program materials and educational tools will be now available to you at the click of a button.’’
Dr. Goldberg also received calls from three patients who reported that they couldn’t complete the quiz that is required of patients capable of reproducing to demonstrate their comprehension about risk. Without the completed quiz, required monthly, the prescription can’t be refilled.
“It’s chaotic,” said Howa Yeung, MD, assistant professor of dermatology at Emory University, Atlanta. “The change is sudden, it’s a major change in the workflow. The process of reverification [required] is not that hard, but a lot of people have trouble even logging into the platform.”
What would help? To have a human on the phone to help navigate the system, Dr. Yeung said.
The glitches are delaying prescriptions for established patients and new ones as well, Dr. Yeung said. Existing patients who can get pregnant have 7 days after their negative pregnancy test to get their prescription filled. “And over the weekend the website was down,” he said, so that was a 2-day delay.
“The information we have and were told to use doesn’t match what is in their database,” said Mitesh Patel, PharmD, owner of Sunshine Pharmacy in White Plains, N.Y., who said pharmacists are experiencing issues with the new platform similar to those of doctors.
Twitter users had a lot to say, as well. Jack Resneck Jr., MD, professor of dermatology at the University of California, San Francisco, tweeted: “#Accutane has basically been pulled from market by utter incompetence of @SyneosHealth hired by @US_FDA to administer risk mgmt program.”
Dr. Resneck, president-elect of the American Medical Association, noted the crashed website, help line with 6-hour hold times, and patients unable to get the drug.
Adewole Adamson, MD, a dermatologist at the University of Texas, Austin, tweeted, “Dermatologists around the US are BIG mad about the current accutane debacle brought on by @SyneosHealth and @US_FDA. What a disaster for patient care!”
Several called for the FDA to immediately halt the program and let physicians manage the risk until the platform could be improved.
Are fixes in sight?
On Tuesday, Dec. 14, AADA President Kenneth J. Tomecki, MD, issued a statement expressing disappointment about the transition.
“In advance of this transition, the AADA engaged the FDA and the iPLEDGE administrator, Syneos Health, about the numerous workflow concerns raised by dermatologists and how the impending changes would threaten patient access to necessary medication. Those concerns have become a reality across the country and we’re working to ensure patients can maintain safe and appropriate access to the treatment they need.”
The AADA, the statement continues, supports efforts to streamline the program while keeping patient safety and incorporating input from physicians.
“We are very aware of the problems with the implementation of the iPLEDGE program,” FDA spokesperson Charlie Kohler said in an email. “We are continuing to work closely with the isotretinoin manufacturers to ensure that they implement a smoothly functioning iPLEDGE REMS program and that patient care is not interrupted.”
“Syneos Health appreciates the concern about iPLEDGE,” said Gary Gatyas, a spokesperson for Syneos Health. “While Syneos Health does not maintain the iPLEDGE system or contact center, we are doing what we can to help the responsible parties with a resolution.” Meanwhile, he recommended that people contact the call center.
He did not respond immediately to questions about who is responsible for maintaining the system and call center.
Dr. Goldberg, Dr. Frieden, and Dr. Yeung have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The that launched on Dec. 13, and what can be done to fix it.
By most accounts, the rollout was disastrous, chaotic, and a failure. Dermatologists on Twitter and elsewhere are angry and frustrated, with some calling for a temporary halt to the program until the bugs can be ironed out.
On Twitter Dec. 15, the Academy posted: “Due to the unacceptable situation with #iPLEDGE, the @US_FDA has convened an emergency meeting with AADA representatives tomorrow, December 16.”
The switch to a new platform was met with frustration from physicians, pharmacists, and patients alike. The new website crashed repeatedly, with physicians and patients complaining they got locked out or bounced off the platform when they attempted to follow instructions to enter information. Calls to obtain support from a live person often required hours on hold, several said.
The new approach to the isotretinoin risk-mitigation program itself isn’t under fire. It was welcomed by dermatologists and others who had long requested the change. Instead of three risk categories (females of reproductive potential, females not of reproductive potential, and males), there are now two (those who can get pregnant and those who cannot). Advocates for the change said it will make the experience more inclusive for transgender patients. The previous categories, some contended, were a barrier to access to care.
Because isotretinoin (Absorica, Amnesteem, Claravis, others), an oral retinoid used to treat severe forms of acne, is teratogenic, with a high risk of birth defects, and has also been associated with other health issues, those who take the medication who are able to get pregnant must take contraceptive precautions. The risk evaluation and mitigation program (REMS), mandated by the FDA, stipulates that physicians, patients, and pharmacists prescribing, using, or dispensing the drug must all be registered with requirements that include the use of two forms of an effective contraceptive and regular pregnancy tests by those capable of becoming pregnant.
A day of frustration
Before navigating the new website, a new log-on name was needed, said Ilona J. Frieden, MD, chair of the AADA’s iPLEDGE Workgroup and professor of dermatology at the University of California, San Francisco. “They made you create a month-day-year date of personal significance.” When she tried to log on, she got locked out, she said in an interview.
The transition from the old website to the new, which Dr. Frieden said is now administered by a different vendor, was done quickly. The previous website shut down Dec. 10, and the new one launched Dec. 13, the first day for the new approach.
“A slower rollout would have helped,” Dr. Frieden said. While she and other dermatologists said they offered input previously on how to make the transition go more smoothly, no one seemed to want that help. “We did have a listening session with the FDA,” Dr. Frieden said. That was before the scheduled meeting of Dec. 16.
Neil S. Goldberg, MD, a dermatologist in Westchester County, New York, also was frustrated with the rollout. “The week before the transition, one of my staff had to call iPLEDGE. They had a 177-minute wait to get to a human.
“They want us to register patients online now instead of signing forms in the office, but the links to view, download, or print don’t work,” Dr. Goldberg said in an interview.
This was after receiving information from the iPLEDGE REMS program, which stated, “The iPLEDGE REMS website will be updated to a modernized platform. All program materials and educational tools will be now available to you at the click of a button.’’
Dr. Goldberg also received calls from three patients who reported that they couldn’t complete the quiz that is required of patients capable of reproducing to demonstrate their comprehension about risk. Without the completed quiz, required monthly, the prescription can’t be refilled.
“It’s chaotic,” said Howa Yeung, MD, assistant professor of dermatology at Emory University, Atlanta. “The change is sudden, it’s a major change in the workflow. The process of reverification [required] is not that hard, but a lot of people have trouble even logging into the platform.”
What would help? To have a human on the phone to help navigate the system, Dr. Yeung said.
The glitches are delaying prescriptions for established patients and new ones as well, Dr. Yeung said. Existing patients who can get pregnant have 7 days after their negative pregnancy test to get their prescription filled. “And over the weekend the website was down,” he said, so that was a 2-day delay.
“The information we have and were told to use doesn’t match what is in their database,” said Mitesh Patel, PharmD, owner of Sunshine Pharmacy in White Plains, N.Y., who said pharmacists are experiencing issues with the new platform similar to those of doctors.
Twitter users had a lot to say, as well. Jack Resneck Jr., MD, professor of dermatology at the University of California, San Francisco, tweeted: “#Accutane has basically been pulled from market by utter incompetence of @SyneosHealth hired by @US_FDA to administer risk mgmt program.”
Dr. Resneck, president-elect of the American Medical Association, noted the crashed website, help line with 6-hour hold times, and patients unable to get the drug.
Adewole Adamson, MD, a dermatologist at the University of Texas, Austin, tweeted, “Dermatologists around the US are BIG mad about the current accutane debacle brought on by @SyneosHealth and @US_FDA. What a disaster for patient care!”
Several called for the FDA to immediately halt the program and let physicians manage the risk until the platform could be improved.
Are fixes in sight?
On Tuesday, Dec. 14, AADA President Kenneth J. Tomecki, MD, issued a statement expressing disappointment about the transition.
“In advance of this transition, the AADA engaged the FDA and the iPLEDGE administrator, Syneos Health, about the numerous workflow concerns raised by dermatologists and how the impending changes would threaten patient access to necessary medication. Those concerns have become a reality across the country and we’re working to ensure patients can maintain safe and appropriate access to the treatment they need.”
The AADA, the statement continues, supports efforts to streamline the program while keeping patient safety and incorporating input from physicians.
“We are very aware of the problems with the implementation of the iPLEDGE program,” FDA spokesperson Charlie Kohler said in an email. “We are continuing to work closely with the isotretinoin manufacturers to ensure that they implement a smoothly functioning iPLEDGE REMS program and that patient care is not interrupted.”
“Syneos Health appreciates the concern about iPLEDGE,” said Gary Gatyas, a spokesperson for Syneos Health. “While Syneos Health does not maintain the iPLEDGE system or contact center, we are doing what we can to help the responsible parties with a resolution.” Meanwhile, he recommended that people contact the call center.
He did not respond immediately to questions about who is responsible for maintaining the system and call center.
Dr. Goldberg, Dr. Frieden, and Dr. Yeung have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The that launched on Dec. 13, and what can be done to fix it.
By most accounts, the rollout was disastrous, chaotic, and a failure. Dermatologists on Twitter and elsewhere are angry and frustrated, with some calling for a temporary halt to the program until the bugs can be ironed out.
On Twitter Dec. 15, the Academy posted: “Due to the unacceptable situation with #iPLEDGE, the @US_FDA has convened an emergency meeting with AADA representatives tomorrow, December 16.”
The switch to a new platform was met with frustration from physicians, pharmacists, and patients alike. The new website crashed repeatedly, with physicians and patients complaining they got locked out or bounced off the platform when they attempted to follow instructions to enter information. Calls to obtain support from a live person often required hours on hold, several said.
The new approach to the isotretinoin risk-mitigation program itself isn’t under fire. It was welcomed by dermatologists and others who had long requested the change. Instead of three risk categories (females of reproductive potential, females not of reproductive potential, and males), there are now two (those who can get pregnant and those who cannot). Advocates for the change said it will make the experience more inclusive for transgender patients. The previous categories, some contended, were a barrier to access to care.
Because isotretinoin (Absorica, Amnesteem, Claravis, others), an oral retinoid used to treat severe forms of acne, is teratogenic, with a high risk of birth defects, and has also been associated with other health issues, those who take the medication who are able to get pregnant must take contraceptive precautions. The risk evaluation and mitigation program (REMS), mandated by the FDA, stipulates that physicians, patients, and pharmacists prescribing, using, or dispensing the drug must all be registered with requirements that include the use of two forms of an effective contraceptive and regular pregnancy tests by those capable of becoming pregnant.
A day of frustration
Before navigating the new website, a new log-on name was needed, said Ilona J. Frieden, MD, chair of the AADA’s iPLEDGE Workgroup and professor of dermatology at the University of California, San Francisco. “They made you create a month-day-year date of personal significance.” When she tried to log on, she got locked out, she said in an interview.
The transition from the old website to the new, which Dr. Frieden said is now administered by a different vendor, was done quickly. The previous website shut down Dec. 10, and the new one launched Dec. 13, the first day for the new approach.
“A slower rollout would have helped,” Dr. Frieden said. While she and other dermatologists said they offered input previously on how to make the transition go more smoothly, no one seemed to want that help. “We did have a listening session with the FDA,” Dr. Frieden said. That was before the scheduled meeting of Dec. 16.
Neil S. Goldberg, MD, a dermatologist in Westchester County, New York, also was frustrated with the rollout. “The week before the transition, one of my staff had to call iPLEDGE. They had a 177-minute wait to get to a human.
“They want us to register patients online now instead of signing forms in the office, but the links to view, download, or print don’t work,” Dr. Goldberg said in an interview.
This was after receiving information from the iPLEDGE REMS program, which stated, “The iPLEDGE REMS website will be updated to a modernized platform. All program materials and educational tools will be now available to you at the click of a button.’’
Dr. Goldberg also received calls from three patients who reported that they couldn’t complete the quiz that is required of patients capable of reproducing to demonstrate their comprehension about risk. Without the completed quiz, required monthly, the prescription can’t be refilled.
“It’s chaotic,” said Howa Yeung, MD, assistant professor of dermatology at Emory University, Atlanta. “The change is sudden, it’s a major change in the workflow. The process of reverification [required] is not that hard, but a lot of people have trouble even logging into the platform.”
What would help? To have a human on the phone to help navigate the system, Dr. Yeung said.
The glitches are delaying prescriptions for established patients and new ones as well, Dr. Yeung said. Existing patients who can get pregnant have 7 days after their negative pregnancy test to get their prescription filled. “And over the weekend the website was down,” he said, so that was a 2-day delay.
“The information we have and were told to use doesn’t match what is in their database,” said Mitesh Patel, PharmD, owner of Sunshine Pharmacy in White Plains, N.Y., who said pharmacists are experiencing issues with the new platform similar to those of doctors.
Twitter users had a lot to say, as well. Jack Resneck Jr., MD, professor of dermatology at the University of California, San Francisco, tweeted: “#Accutane has basically been pulled from market by utter incompetence of @SyneosHealth hired by @US_FDA to administer risk mgmt program.”
Dr. Resneck, president-elect of the American Medical Association, noted the crashed website, help line with 6-hour hold times, and patients unable to get the drug.
Adewole Adamson, MD, a dermatologist at the University of Texas, Austin, tweeted, “Dermatologists around the US are BIG mad about the current accutane debacle brought on by @SyneosHealth and @US_FDA. What a disaster for patient care!”
Several called for the FDA to immediately halt the program and let physicians manage the risk until the platform could be improved.
Are fixes in sight?
On Tuesday, Dec. 14, AADA President Kenneth J. Tomecki, MD, issued a statement expressing disappointment about the transition.
“In advance of this transition, the AADA engaged the FDA and the iPLEDGE administrator, Syneos Health, about the numerous workflow concerns raised by dermatologists and how the impending changes would threaten patient access to necessary medication. Those concerns have become a reality across the country and we’re working to ensure patients can maintain safe and appropriate access to the treatment they need.”
The AADA, the statement continues, supports efforts to streamline the program while keeping patient safety and incorporating input from physicians.
“We are very aware of the problems with the implementation of the iPLEDGE program,” FDA spokesperson Charlie Kohler said in an email. “We are continuing to work closely with the isotretinoin manufacturers to ensure that they implement a smoothly functioning iPLEDGE REMS program and that patient care is not interrupted.”
“Syneos Health appreciates the concern about iPLEDGE,” said Gary Gatyas, a spokesperson for Syneos Health. “While Syneos Health does not maintain the iPLEDGE system or contact center, we are doing what we can to help the responsible parties with a resolution.” Meanwhile, he recommended that people contact the call center.
He did not respond immediately to questions about who is responsible for maintaining the system and call center.
Dr. Goldberg, Dr. Frieden, and Dr. Yeung have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A pandemic silver lining? Dramatic drop in teen drug use
Illicit drug use among U.S. teenagers dropped sharply in 2021, likely because of stay-at-home orders and other restrictions on social activities due to the COVID-19 pandemic.
The latest findings, from the Monitoring the Future survey, represent the largest 1-year decrease in overall illicit drug use reported since the survey began in 1975.
“We have never seen such dramatic decreases in drug use among teens in just a 1-year period,” Nora Volkow, MD, director of the National Institute on Drug Abuse (NIDA), said in a news release.
“These data are unprecedented and highlight one unexpected potential consequence of the COVID-19 pandemic, which caused seismic shifts in the day-to-day lives of adolescents,” said Dr. Volkow.
The annual Monitoring the Future survey is conducted by researchers at the University of Michigan, Ann Arbor, and funded by NIDA, to assess drug and alcohol use and related attitudes among adolescent students across the United States.
This year’s self-reported survey included 32,260 students in grades 8, 10, and 12 across 319 public and private schools.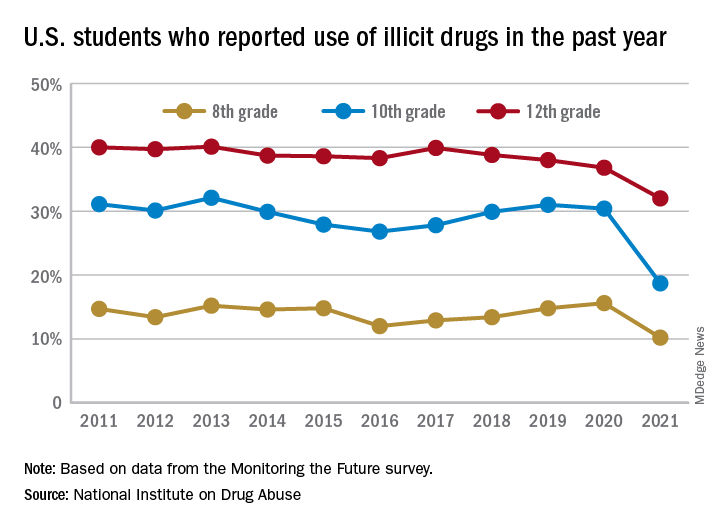
Compared with 2020, the percentage of students reporting any illicit drug use (other than marijuana) in 2021 decreased significantly for 8th graders (down 5.4%), 10th graders (down 11.7%), and 12th graders (down 4.8%).
For alcohol, about 47% of 12th graders and 29% of 10th graders said they drank alcohol in 2021, down significantly from 55% and 41%, respectively, in 2020. The percentage of 8th graders who said they drank alcohol remained stable (17% in 2021 and 20% in 2020).
For teen vaping, about 27% of 12th graders and 20% of 10th graders said they had vaped nicotine in 2021, down significantly from nearly 35% and 31%, respectively, in 2020. Fewer 8th graders also vaped nicotine in 2021 compared with 2020 (12% vs. 17%).
For marijuana, use dropped significantly for all three grades in 2021 compared with 2020. About 31% of 12th graders and 17% of 10th graders said they used marijuana in 2021, down from 35% and 28% in 2020. Among 8th graders, 7% used marijuana in 2021, down from 11% in 2020.
The latest survey also shows significant declines in use of a range of other drugs for many of the age cohorts, including cocaine, hallucinogens, and nonmedical use of amphetamines, tranquilizers, and prescription opioids.
“We knew that this year’s data would illuminate how the COVID-19 pandemic may have impacted substance use among young people, and in the coming years, we will find out whether those impacts are long-lasting as we continue tracking the drug use patterns of these unique cohorts of adolescents,” Richard A. Miech, PhD, who heads the Monitoring the Future study at the University of Michigan, said in the news release.
“Moving forward, it will be crucial to identify the pivotal elements of this past year that contributed to decreased drug use – whether related to drug availability, family involvement, differences in peer pressure, or other factors – and harness them to inform future prevention efforts,” Dr. Volkow added.
In 2021, students across all age groups reported moderate increases in feelings of boredom, anxiety, depression, loneliness, worry, difficulty sleeping, and other negative mental health indicators since the beginning of the pandemic.
A version of this article first appeared on Medscape.com.
Illicit drug use among U.S. teenagers dropped sharply in 2021, likely because of stay-at-home orders and other restrictions on social activities due to the COVID-19 pandemic.
The latest findings, from the Monitoring the Future survey, represent the largest 1-year decrease in overall illicit drug use reported since the survey began in 1975.
“We have never seen such dramatic decreases in drug use among teens in just a 1-year period,” Nora Volkow, MD, director of the National Institute on Drug Abuse (NIDA), said in a news release.
“These data are unprecedented and highlight one unexpected potential consequence of the COVID-19 pandemic, which caused seismic shifts in the day-to-day lives of adolescents,” said Dr. Volkow.
The annual Monitoring the Future survey is conducted by researchers at the University of Michigan, Ann Arbor, and funded by NIDA, to assess drug and alcohol use and related attitudes among adolescent students across the United States.
This year’s self-reported survey included 32,260 students in grades 8, 10, and 12 across 319 public and private schools.
Compared with 2020, the percentage of students reporting any illicit drug use (other than marijuana) in 2021 decreased significantly for 8th graders (down 5.4%), 10th graders (down 11.7%), and 12th graders (down 4.8%).
For alcohol, about 47% of 12th graders and 29% of 10th graders said they drank alcohol in 2021, down significantly from 55% and 41%, respectively, in 2020. The percentage of 8th graders who said they drank alcohol remained stable (17% in 2021 and 20% in 2020).
For teen vaping, about 27% of 12th graders and 20% of 10th graders said they had vaped nicotine in 2021, down significantly from nearly 35% and 31%, respectively, in 2020. Fewer 8th graders also vaped nicotine in 2021 compared with 2020 (12% vs. 17%).
For marijuana, use dropped significantly for all three grades in 2021 compared with 2020. About 31% of 12th graders and 17% of 10th graders said they used marijuana in 2021, down from 35% and 28% in 2020. Among 8th graders, 7% used marijuana in 2021, down from 11% in 2020.
The latest survey also shows significant declines in use of a range of other drugs for many of the age cohorts, including cocaine, hallucinogens, and nonmedical use of amphetamines, tranquilizers, and prescription opioids.
“We knew that this year’s data would illuminate how the COVID-19 pandemic may have impacted substance use among young people, and in the coming years, we will find out whether those impacts are long-lasting as we continue tracking the drug use patterns of these unique cohorts of adolescents,” Richard A. Miech, PhD, who heads the Monitoring the Future study at the University of Michigan, said in the news release.
“Moving forward, it will be crucial to identify the pivotal elements of this past year that contributed to decreased drug use – whether related to drug availability, family involvement, differences in peer pressure, or other factors – and harness them to inform future prevention efforts,” Dr. Volkow added.
In 2021, students across all age groups reported moderate increases in feelings of boredom, anxiety, depression, loneliness, worry, difficulty sleeping, and other negative mental health indicators since the beginning of the pandemic.
A version of this article first appeared on Medscape.com.
Illicit drug use among U.S. teenagers dropped sharply in 2021, likely because of stay-at-home orders and other restrictions on social activities due to the COVID-19 pandemic.
The latest findings, from the Monitoring the Future survey, represent the largest 1-year decrease in overall illicit drug use reported since the survey began in 1975.
“We have never seen such dramatic decreases in drug use among teens in just a 1-year period,” Nora Volkow, MD, director of the National Institute on Drug Abuse (NIDA), said in a news release.
“These data are unprecedented and highlight one unexpected potential consequence of the COVID-19 pandemic, which caused seismic shifts in the day-to-day lives of adolescents,” said Dr. Volkow.
The annual Monitoring the Future survey is conducted by researchers at the University of Michigan, Ann Arbor, and funded by NIDA, to assess drug and alcohol use and related attitudes among adolescent students across the United States.
This year’s self-reported survey included 32,260 students in grades 8, 10, and 12 across 319 public and private schools.
Compared with 2020, the percentage of students reporting any illicit drug use (other than marijuana) in 2021 decreased significantly for 8th graders (down 5.4%), 10th graders (down 11.7%), and 12th graders (down 4.8%).
For alcohol, about 47% of 12th graders and 29% of 10th graders said they drank alcohol in 2021, down significantly from 55% and 41%, respectively, in 2020. The percentage of 8th graders who said they drank alcohol remained stable (17% in 2021 and 20% in 2020).
For teen vaping, about 27% of 12th graders and 20% of 10th graders said they had vaped nicotine in 2021, down significantly from nearly 35% and 31%, respectively, in 2020. Fewer 8th graders also vaped nicotine in 2021 compared with 2020 (12% vs. 17%).
For marijuana, use dropped significantly for all three grades in 2021 compared with 2020. About 31% of 12th graders and 17% of 10th graders said they used marijuana in 2021, down from 35% and 28% in 2020. Among 8th graders, 7% used marijuana in 2021, down from 11% in 2020.
The latest survey also shows significant declines in use of a range of other drugs for many of the age cohorts, including cocaine, hallucinogens, and nonmedical use of amphetamines, tranquilizers, and prescription opioids.
“We knew that this year’s data would illuminate how the COVID-19 pandemic may have impacted substance use among young people, and in the coming years, we will find out whether those impacts are long-lasting as we continue tracking the drug use patterns of these unique cohorts of adolescents,” Richard A. Miech, PhD, who heads the Monitoring the Future study at the University of Michigan, said in the news release.
“Moving forward, it will be crucial to identify the pivotal elements of this past year that contributed to decreased drug use – whether related to drug availability, family involvement, differences in peer pressure, or other factors – and harness them to inform future prevention efforts,” Dr. Volkow added.
In 2021, students across all age groups reported moderate increases in feelings of boredom, anxiety, depression, loneliness, worry, difficulty sleeping, and other negative mental health indicators since the beginning of the pandemic.
A version of this article first appeared on Medscape.com.
Abrocitinib efficacy dose-dependent, similar across AD age groups
and was comparable in patients aged 51 years and older, results from a post hoc analysis of four trials showed.
Abrocitinib (Cibinqo) is an oral, once-daily, Janus kinase 1 selective inhibitor that has shown good efficacy and safety as monotherapy or combined with topical therapy for treatment of patients with moderate to severe AD. The agent was approved in mid-December in Europe for the treatment of moderate to severe AD in adults who are candidates for systemic therapy and is currently under review by the Food and Drug Administration.
“We know that responses to, and adverse events associated with, systemic therapies may vary among patients of different ages,” Andrew F. Alexis, MD, MPH, said during a late-breaking abstract session at the Revolutionizing Atopic Dermatitis virtual symposium. “The efficacy and safety of abrocitinib monotherapy were previously evaluated in adolescent and adult subpopulations from controlled clinical trials in patients with moderate to severe AD. The objective of the current study was to assess the impact of age on short-term responses to abrocitinib treatment in patients with moderate to severe AD.”
Dr. Alexis, professor of clinical dermatology at Weill Cornell Medicine, New York, and colleagues performed a post hoc analysis across four randomized, double-blind studies that was stratified by age group: 12-17 years, 18-40 years, 41-50 years, and 51 years and older. Efficacy data were assessed separately for patients in the monotherapy pool and in the JADE COMPARE trial. The monotherapy pool included patients from one phase 2b study and two phase 3 studies who received abrocitinib 200 mg, abrocitinib 100 mg, or placebo monotherapy for 12 weeks (JADE-MONO-1 and JADE-MONO-2).
The JADE COMPARE pool included patients who received abrocitinib 200 mg, abrocitinib 100 mg, or placebo, plus medicated topical therapy for 16 weeks. Data from patients in all four trials were pooled for the analysis of treatment-emergent adverse events. Efficacy points analyzed were the Investigator Global Assessment (IGA) score of 0/1 (clear or almost clear), a 75% reduction from baseline in the Eczema Area and Severity Index (EASI-75), or Peak Pruritus Numeric Rating Scale score (PP-NRS4) at week 12 for the monotherapy pool and at week 16 for COMPARE.
In the monotherapy pool, the proportions of patients ages 12-17 years, 18-40 years, 41-50 years, and 51 years and older who achieved an IGA 0/1 response at 12 weeks were 31.3%, 40.2%, 43.8%, and 50.8% (abrocitinib 200 mg); 22%, 23.7%, 22.4%, and 40.8% (abrocitinib 100 mg); and 8.7%, 8%, 3.3%, and 10% (placebo).
In JADE COMPARE, the proportions of patients aged 18-40 years, 41-50 years, and 51 years and older who achieved an IGA 0/1 response were 50%, 53.2%, and 34.8% (abrocitinib 200 mg); 36.9%, 37.1%, and 26.1% (abrocitinib 100 mg); and 12%, 11.8%, and 16.7% (placebo) at 16 weeks. Similar trends were observed for EASI-75 and PP-NRS4 responses at 12 weeks.
Across all age groups, the most common treatment-emergent adverse events were infections/infestations and gastrointestinal effects; most cases were mild or moderate. Nausea was more frequent in the two younger age groups and was dose related: For abrocitinib 200 mg and abrocitinib 100 mg, respectively, the rates of nausea were 18.8% and 7.8% in patients aged 12-17 years; 17.1% and 6.4% in patients aged 18-40 years; and 7.1% and 3.3% in patients aged 51 and older.
“Efficacy responses in patients 51 years of age and older were comparable to those in other age groups,” concluded Dr. Alexis, vice chair for diversity and inclusion in the department of dermatology at Weill Cornell. “The safety profile was consistent across age ranges and was similar to that reported previously.”
The investigators found that treatment response to abrocitinib “in the absence or presence of medicated topical therapy was fairly consistent across age groups, showed similar dose-dependency, and importantly, did not show reduced efficacy in older adults as measured by lesional severity, extent, and itch at 4 months,” said Raj Chovatiya, MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, who was asked to comment on the study.
“Furthermore, the safety profile was consistent across all adults, though notably, nausea was more common among younger age groups, highlighting an area of future investigation,” he added. “Overall, these data show that abrocitinib is associated with similar short-term responses across adulthood and underscore the importance of the JAK-STAT pathway in the underlying pathophysiology of AD in different age groups. It will be interesting to see how these data reflect the real-world setting with both short- and long-term outcomes in a heterogeneous patient population.”
In the interview, Dr. Chovatiya said, “the next frontier in personalized therapy for AD involves deeper clinical phenotyping of our patients and a better understanding of how efficacy and safety vary across patient groups.” For example, he noted, “AD in earlier versus later adulthood may be associated with different clinical signs, symptoms, comorbidities, and other measures of patient burden, and thus, may be associated with different treatment responses to systemic therapy.”
Dr. Alexis disclosed that he has served as an adviser to, or has received consulting fees from, Leo, Galderma, Pfizer, Sanofi-Regeneron, Dermavant, Beiersdorf, Valeant, L’Oréal, BMS, Bausch Health, UCB, Vyne, Arcutis, Janssen, Allergan, Almirall, AbbVie, Sol-Gel, and Amgen.
Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Arena, Arcutis, Incyte, Pfizer, Regeneron, and Sanofi-Genzyme.
A version of this article first appeared on Medscape.com.
and was comparable in patients aged 51 years and older, results from a post hoc analysis of four trials showed.
Abrocitinib (Cibinqo) is an oral, once-daily, Janus kinase 1 selective inhibitor that has shown good efficacy and safety as monotherapy or combined with topical therapy for treatment of patients with moderate to severe AD. The agent was approved in mid-December in Europe for the treatment of moderate to severe AD in adults who are candidates for systemic therapy and is currently under review by the Food and Drug Administration.
“We know that responses to, and adverse events associated with, systemic therapies may vary among patients of different ages,” Andrew F. Alexis, MD, MPH, said during a late-breaking abstract session at the Revolutionizing Atopic Dermatitis virtual symposium. “The efficacy and safety of abrocitinib monotherapy were previously evaluated in adolescent and adult subpopulations from controlled clinical trials in patients with moderate to severe AD. The objective of the current study was to assess the impact of age on short-term responses to abrocitinib treatment in patients with moderate to severe AD.”
Dr. Alexis, professor of clinical dermatology at Weill Cornell Medicine, New York, and colleagues performed a post hoc analysis across four randomized, double-blind studies that was stratified by age group: 12-17 years, 18-40 years, 41-50 years, and 51 years and older. Efficacy data were assessed separately for patients in the monotherapy pool and in the JADE COMPARE trial. The monotherapy pool included patients from one phase 2b study and two phase 3 studies who received abrocitinib 200 mg, abrocitinib 100 mg, or placebo monotherapy for 12 weeks (JADE-MONO-1 and JADE-MONO-2).
The JADE COMPARE pool included patients who received abrocitinib 200 mg, abrocitinib 100 mg, or placebo, plus medicated topical therapy for 16 weeks. Data from patients in all four trials were pooled for the analysis of treatment-emergent adverse events. Efficacy points analyzed were the Investigator Global Assessment (IGA) score of 0/1 (clear or almost clear), a 75% reduction from baseline in the Eczema Area and Severity Index (EASI-75), or Peak Pruritus Numeric Rating Scale score (PP-NRS4) at week 12 for the monotherapy pool and at week 16 for COMPARE.
In the monotherapy pool, the proportions of patients ages 12-17 years, 18-40 years, 41-50 years, and 51 years and older who achieved an IGA 0/1 response at 12 weeks were 31.3%, 40.2%, 43.8%, and 50.8% (abrocitinib 200 mg); 22%, 23.7%, 22.4%, and 40.8% (abrocitinib 100 mg); and 8.7%, 8%, 3.3%, and 10% (placebo).
In JADE COMPARE, the proportions of patients aged 18-40 years, 41-50 years, and 51 years and older who achieved an IGA 0/1 response were 50%, 53.2%, and 34.8% (abrocitinib 200 mg); 36.9%, 37.1%, and 26.1% (abrocitinib 100 mg); and 12%, 11.8%, and 16.7% (placebo) at 16 weeks. Similar trends were observed for EASI-75 and PP-NRS4 responses at 12 weeks.
Across all age groups, the most common treatment-emergent adverse events were infections/infestations and gastrointestinal effects; most cases were mild or moderate. Nausea was more frequent in the two younger age groups and was dose related: For abrocitinib 200 mg and abrocitinib 100 mg, respectively, the rates of nausea were 18.8% and 7.8% in patients aged 12-17 years; 17.1% and 6.4% in patients aged 18-40 years; and 7.1% and 3.3% in patients aged 51 and older.
“Efficacy responses in patients 51 years of age and older were comparable to those in other age groups,” concluded Dr. Alexis, vice chair for diversity and inclusion in the department of dermatology at Weill Cornell. “The safety profile was consistent across age ranges and was similar to that reported previously.”
The investigators found that treatment response to abrocitinib “in the absence or presence of medicated topical therapy was fairly consistent across age groups, showed similar dose-dependency, and importantly, did not show reduced efficacy in older adults as measured by lesional severity, extent, and itch at 4 months,” said Raj Chovatiya, MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, who was asked to comment on the study.
“Furthermore, the safety profile was consistent across all adults, though notably, nausea was more common among younger age groups, highlighting an area of future investigation,” he added. “Overall, these data show that abrocitinib is associated with similar short-term responses across adulthood and underscore the importance of the JAK-STAT pathway in the underlying pathophysiology of AD in different age groups. It will be interesting to see how these data reflect the real-world setting with both short- and long-term outcomes in a heterogeneous patient population.”
In the interview, Dr. Chovatiya said, “the next frontier in personalized therapy for AD involves deeper clinical phenotyping of our patients and a better understanding of how efficacy and safety vary across patient groups.” For example, he noted, “AD in earlier versus later adulthood may be associated with different clinical signs, symptoms, comorbidities, and other measures of patient burden, and thus, may be associated with different treatment responses to systemic therapy.”
Dr. Alexis disclosed that he has served as an adviser to, or has received consulting fees from, Leo, Galderma, Pfizer, Sanofi-Regeneron, Dermavant, Beiersdorf, Valeant, L’Oréal, BMS, Bausch Health, UCB, Vyne, Arcutis, Janssen, Allergan, Almirall, AbbVie, Sol-Gel, and Amgen.
Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Arena, Arcutis, Incyte, Pfizer, Regeneron, and Sanofi-Genzyme.
A version of this article first appeared on Medscape.com.
and was comparable in patients aged 51 years and older, results from a post hoc analysis of four trials showed.
Abrocitinib (Cibinqo) is an oral, once-daily, Janus kinase 1 selective inhibitor that has shown good efficacy and safety as monotherapy or combined with topical therapy for treatment of patients with moderate to severe AD. The agent was approved in mid-December in Europe for the treatment of moderate to severe AD in adults who are candidates for systemic therapy and is currently under review by the Food and Drug Administration.
“We know that responses to, and adverse events associated with, systemic therapies may vary among patients of different ages,” Andrew F. Alexis, MD, MPH, said during a late-breaking abstract session at the Revolutionizing Atopic Dermatitis virtual symposium. “The efficacy and safety of abrocitinib monotherapy were previously evaluated in adolescent and adult subpopulations from controlled clinical trials in patients with moderate to severe AD. The objective of the current study was to assess the impact of age on short-term responses to abrocitinib treatment in patients with moderate to severe AD.”
Dr. Alexis, professor of clinical dermatology at Weill Cornell Medicine, New York, and colleagues performed a post hoc analysis across four randomized, double-blind studies that was stratified by age group: 12-17 years, 18-40 years, 41-50 years, and 51 years and older. Efficacy data were assessed separately for patients in the monotherapy pool and in the JADE COMPARE trial. The monotherapy pool included patients from one phase 2b study and two phase 3 studies who received abrocitinib 200 mg, abrocitinib 100 mg, or placebo monotherapy for 12 weeks (JADE-MONO-1 and JADE-MONO-2).
The JADE COMPARE pool included patients who received abrocitinib 200 mg, abrocitinib 100 mg, or placebo, plus medicated topical therapy for 16 weeks. Data from patients in all four trials were pooled for the analysis of treatment-emergent adverse events. Efficacy points analyzed were the Investigator Global Assessment (IGA) score of 0/1 (clear or almost clear), a 75% reduction from baseline in the Eczema Area and Severity Index (EASI-75), or Peak Pruritus Numeric Rating Scale score (PP-NRS4) at week 12 for the monotherapy pool and at week 16 for COMPARE.
In the monotherapy pool, the proportions of patients ages 12-17 years, 18-40 years, 41-50 years, and 51 years and older who achieved an IGA 0/1 response at 12 weeks were 31.3%, 40.2%, 43.8%, and 50.8% (abrocitinib 200 mg); 22%, 23.7%, 22.4%, and 40.8% (abrocitinib 100 mg); and 8.7%, 8%, 3.3%, and 10% (placebo).
In JADE COMPARE, the proportions of patients aged 18-40 years, 41-50 years, and 51 years and older who achieved an IGA 0/1 response were 50%, 53.2%, and 34.8% (abrocitinib 200 mg); 36.9%, 37.1%, and 26.1% (abrocitinib 100 mg); and 12%, 11.8%, and 16.7% (placebo) at 16 weeks. Similar trends were observed for EASI-75 and PP-NRS4 responses at 12 weeks.
Across all age groups, the most common treatment-emergent adverse events were infections/infestations and gastrointestinal effects; most cases were mild or moderate. Nausea was more frequent in the two younger age groups and was dose related: For abrocitinib 200 mg and abrocitinib 100 mg, respectively, the rates of nausea were 18.8% and 7.8% in patients aged 12-17 years; 17.1% and 6.4% in patients aged 18-40 years; and 7.1% and 3.3% in patients aged 51 and older.
“Efficacy responses in patients 51 years of age and older were comparable to those in other age groups,” concluded Dr. Alexis, vice chair for diversity and inclusion in the department of dermatology at Weill Cornell. “The safety profile was consistent across age ranges and was similar to that reported previously.”
The investigators found that treatment response to abrocitinib “in the absence or presence of medicated topical therapy was fairly consistent across age groups, showed similar dose-dependency, and importantly, did not show reduced efficacy in older adults as measured by lesional severity, extent, and itch at 4 months,” said Raj Chovatiya, MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, who was asked to comment on the study.
“Furthermore, the safety profile was consistent across all adults, though notably, nausea was more common among younger age groups, highlighting an area of future investigation,” he added. “Overall, these data show that abrocitinib is associated with similar short-term responses across adulthood and underscore the importance of the JAK-STAT pathway in the underlying pathophysiology of AD in different age groups. It will be interesting to see how these data reflect the real-world setting with both short- and long-term outcomes in a heterogeneous patient population.”
In the interview, Dr. Chovatiya said, “the next frontier in personalized therapy for AD involves deeper clinical phenotyping of our patients and a better understanding of how efficacy and safety vary across patient groups.” For example, he noted, “AD in earlier versus later adulthood may be associated with different clinical signs, symptoms, comorbidities, and other measures of patient burden, and thus, may be associated with different treatment responses to systemic therapy.”
Dr. Alexis disclosed that he has served as an adviser to, or has received consulting fees from, Leo, Galderma, Pfizer, Sanofi-Regeneron, Dermavant, Beiersdorf, Valeant, L’Oréal, BMS, Bausch Health, UCB, Vyne, Arcutis, Janssen, Allergan, Almirall, AbbVie, Sol-Gel, and Amgen.
Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Arena, Arcutis, Incyte, Pfizer, Regeneron, and Sanofi-Genzyme.
A version of this article first appeared on Medscape.com.
FROM REVOLUTIONIZING AD 2021
Delayed umbilical cord clamping improves outcomes in very preterm infants
Delayed umbilical cord clamping for at least 60 seconds after birth significantly reduced death or disability in infants of less than 30 weeks’ gestation, according to data from nearly 1,500 infants.
The burden of disability and mortality for babies born before 30 weeks’ gestation remains high, especially in low- and middle-income countries, wrote Kristy P. Robledo, PhD, of the University of Sydney, Australia, and colleagues. Delayed clamping of the umbilical cord is a simple procedure that may improve mortality in this population, but more research is needed; recommended times to delayed clamping range from 30 seconds to 3 minutes, they noted.
In a study published in The Lancet Child & Adolescent Health, the researchers randomized 767 very preterm infants to delayed clamping at least 60 seconds after birth and 764 to immediate clamping. Of these, 384 were multiple births (who were individually randomized), 862 were male, and 505 were born before 27 weeks’ gestation. The primary outcome was death or disability at 2 years of age. Major disability was defined as cerebral palsy, severe visual loss, deafness requiring a hearing aid or cochlear implants, major language or speech problems, or cognitive delay at 2 years corrected age. The median time to clamping was 60 seconds in the delayed group and 5 seconds in the immediate group.
Primary outcome data were available for 1,419 infants. Death or major disability occurred in 29% of infants assigned to delayed clamping compared to 34% of those assigned to immediate clamping (relative risk 0.83, P = .010). The infants were part of the APTS Childhood Follow-Up Study, an open-label superiority trial conducted in Australia and New Zealand.
By age 2 years, 8% of infants in the delayed group and 11% of those in the immediate group had died; 23% and 26%, respectively, met criteria for major disability. The impact of delayed clamping translates to a 30% reduction in relative risk of mortality at 2 years of age, but no significant impact on major disability, the researchers wrote.
The findings were limited by several factors including the unblinded study design, lack of data on heart rate or time to first breath, and the clamping prior to 60 seconds in 26% of infants in the delayed group based on clinical concerns for these specific infants, the researchers noted.
However, the results were strengthened by the large size, low risk of bias, and specific primary outcome, they said. The data support findings from recent systematic reviews and highlight the need for further trials to evaluate delayed clamping at different time points, with larger populations, inclusion of time to first breath and heart rate, and improved measures of disability, the researchers added.
In clinical practice, “Given that aiming to delay cord clamping for 60 seconds or more improved 2-year outcomes and short-term hematological measures with no evidence of significant harm, it seems reasonable to conclude that delayed clamping is appropriate as standard care in very preterm infants,” they concluded.
Accepting simple intervention could have great impact
This study is important in light of the overwhelming burden of preterm birth on the health care system and society as a whole, Lisette D. Tanner, MD, of Emory University, Atlanta, said in an interview.
“Preterm birth is associated with billions in health care costs each year, and a large portion of that money is directed to the complications associated with preterm birth, such as early intervention services, educational support, and ongoing medical care,” Dr. Tanner said. “This study is particularly timely, as we are quickly approaching 2030, the deadline for achieving the United Nations Sustainable Development Goal of ending preventable deaths of newborns and children under 5 years of age,” she said. The goal involves “all countries aiming to reduce neonatal mortality to at least as low as 12 per 1,000 live births and under-5 mortality to at least as low as 25 per 1,000 live births. Effective treatments to reduce infant and child mortality would make strong inroads toward this goal,” she explained.
Dr. Tanner said she was not surprised by the findings because previous studies have shown similar results. “However, the large, multicenter nature of this study provides additional weight to recommendations to delay cord clamping as standard practice,” she said.
“The findings of this study support the recommendations of a number of large organizations,” said Dr. Tanner. “The World Health Organization recommends that the umbilical cord not be clamped earlier than 1 minute after birth in term or preterm infants who do not require positive pressure ventilation. The American College of Obstetricians and Gynecologists and the American Academy of Pediatrics now recommend a delay in umbilical cord clamping in vigorous term and preterm infants for at least 30–60 seconds after birth,” she said. “The Royal College of Obstetricians and Gynaecologists also recommends deferring umbilical cord clamping for healthy term and preterm infants for at least 2 minutes after birth,” she added.
However, “the delay in adoption of this guidelines in practice appears to be related to some concerns regarding universal adoption of this approach,” Dr. Tanner noted. “Some clinicians have suggested that delayed cord clamping could delay vital neonatal resuscitative efforts, leading to worse neonatal outcomes, but this concern has not been borne out in the data, as all guidelines specifically state that this intervention is for vigorous newborns,” she said. “In fact, in preterm infants, delayed cord clamping is associated with improved transitional circulation, decreased need for blood transfusion, and lower incidence of necrotizing enterocolitis and intraventricular hemorrhage,” Dr. Tanner emphasized. “Additionally, concerns persist that delayed cord clamping could lead to excessive transfusion with resultant polycythemia. Again, no data have supported this claim to date,” she said.
“Finally, some clinicians are concerned that delayed clamping could lead to delay in addressing maternal complications of birth such as hemorrhage, but studies have shown the opposite; delayed umbilical cord clamping has not been associated with an increased risk of postpartum hemorrhage or increased blood loss at delivery, nor has it been with a difference in the need for blood transfusion,” said Dr. Tanner.
Ideally, practitioners will become more comfortable in delaying cord clamping as a routine practice as more data demonstrating the safety and benefit of this easy intervention are disseminated, she said.
Additional research delineating which gestational ages benefit most from delayed cord clamping would help direct education efforts to implement this intervention, Dr. Tanner noted.
The study was funded by the Australian National Health and Medical Research Council. The researchers and Dr. Tanner had no financial conflicts to disclose.
Delayed umbilical cord clamping for at least 60 seconds after birth significantly reduced death or disability in infants of less than 30 weeks’ gestation, according to data from nearly 1,500 infants.
The burden of disability and mortality for babies born before 30 weeks’ gestation remains high, especially in low- and middle-income countries, wrote Kristy P. Robledo, PhD, of the University of Sydney, Australia, and colleagues. Delayed clamping of the umbilical cord is a simple procedure that may improve mortality in this population, but more research is needed; recommended times to delayed clamping range from 30 seconds to 3 minutes, they noted.
In a study published in The Lancet Child & Adolescent Health, the researchers randomized 767 very preterm infants to delayed clamping at least 60 seconds after birth and 764 to immediate clamping. Of these, 384 were multiple births (who were individually randomized), 862 were male, and 505 were born before 27 weeks’ gestation. The primary outcome was death or disability at 2 years of age. Major disability was defined as cerebral palsy, severe visual loss, deafness requiring a hearing aid or cochlear implants, major language or speech problems, or cognitive delay at 2 years corrected age. The median time to clamping was 60 seconds in the delayed group and 5 seconds in the immediate group.
Primary outcome data were available for 1,419 infants. Death or major disability occurred in 29% of infants assigned to delayed clamping compared to 34% of those assigned to immediate clamping (relative risk 0.83, P = .010). The infants were part of the APTS Childhood Follow-Up Study, an open-label superiority trial conducted in Australia and New Zealand.
By age 2 years, 8% of infants in the delayed group and 11% of those in the immediate group had died; 23% and 26%, respectively, met criteria for major disability. The impact of delayed clamping translates to a 30% reduction in relative risk of mortality at 2 years of age, but no significant impact on major disability, the researchers wrote.
The findings were limited by several factors including the unblinded study design, lack of data on heart rate or time to first breath, and the clamping prior to 60 seconds in 26% of infants in the delayed group based on clinical concerns for these specific infants, the researchers noted.
However, the results were strengthened by the large size, low risk of bias, and specific primary outcome, they said. The data support findings from recent systematic reviews and highlight the need for further trials to evaluate delayed clamping at different time points, with larger populations, inclusion of time to first breath and heart rate, and improved measures of disability, the researchers added.
In clinical practice, “Given that aiming to delay cord clamping for 60 seconds or more improved 2-year outcomes and short-term hematological measures with no evidence of significant harm, it seems reasonable to conclude that delayed clamping is appropriate as standard care in very preterm infants,” they concluded.
Accepting simple intervention could have great impact
This study is important in light of the overwhelming burden of preterm birth on the health care system and society as a whole, Lisette D. Tanner, MD, of Emory University, Atlanta, said in an interview.
“Preterm birth is associated with billions in health care costs each year, and a large portion of that money is directed to the complications associated with preterm birth, such as early intervention services, educational support, and ongoing medical care,” Dr. Tanner said. “This study is particularly timely, as we are quickly approaching 2030, the deadline for achieving the United Nations Sustainable Development Goal of ending preventable deaths of newborns and children under 5 years of age,” she said. The goal involves “all countries aiming to reduce neonatal mortality to at least as low as 12 per 1,000 live births and under-5 mortality to at least as low as 25 per 1,000 live births. Effective treatments to reduce infant and child mortality would make strong inroads toward this goal,” she explained.
Dr. Tanner said she was not surprised by the findings because previous studies have shown similar results. “However, the large, multicenter nature of this study provides additional weight to recommendations to delay cord clamping as standard practice,” she said.
“The findings of this study support the recommendations of a number of large organizations,” said Dr. Tanner. “The World Health Organization recommends that the umbilical cord not be clamped earlier than 1 minute after birth in term or preterm infants who do not require positive pressure ventilation. The American College of Obstetricians and Gynecologists and the American Academy of Pediatrics now recommend a delay in umbilical cord clamping in vigorous term and preterm infants for at least 30–60 seconds after birth,” she said. “The Royal College of Obstetricians and Gynaecologists also recommends deferring umbilical cord clamping for healthy term and preterm infants for at least 2 minutes after birth,” she added.
However, “the delay in adoption of this guidelines in practice appears to be related to some concerns regarding universal adoption of this approach,” Dr. Tanner noted. “Some clinicians have suggested that delayed cord clamping could delay vital neonatal resuscitative efforts, leading to worse neonatal outcomes, but this concern has not been borne out in the data, as all guidelines specifically state that this intervention is for vigorous newborns,” she said. “In fact, in preterm infants, delayed cord clamping is associated with improved transitional circulation, decreased need for blood transfusion, and lower incidence of necrotizing enterocolitis and intraventricular hemorrhage,” Dr. Tanner emphasized. “Additionally, concerns persist that delayed cord clamping could lead to excessive transfusion with resultant polycythemia. Again, no data have supported this claim to date,” she said.
“Finally, some clinicians are concerned that delayed clamping could lead to delay in addressing maternal complications of birth such as hemorrhage, but studies have shown the opposite; delayed umbilical cord clamping has not been associated with an increased risk of postpartum hemorrhage or increased blood loss at delivery, nor has it been with a difference in the need for blood transfusion,” said Dr. Tanner.
Ideally, practitioners will become more comfortable in delaying cord clamping as a routine practice as more data demonstrating the safety and benefit of this easy intervention are disseminated, she said.
Additional research delineating which gestational ages benefit most from delayed cord clamping would help direct education efforts to implement this intervention, Dr. Tanner noted.
The study was funded by the Australian National Health and Medical Research Council. The researchers and Dr. Tanner had no financial conflicts to disclose.
Delayed umbilical cord clamping for at least 60 seconds after birth significantly reduced death or disability in infants of less than 30 weeks’ gestation, according to data from nearly 1,500 infants.
The burden of disability and mortality for babies born before 30 weeks’ gestation remains high, especially in low- and middle-income countries, wrote Kristy P. Robledo, PhD, of the University of Sydney, Australia, and colleagues. Delayed clamping of the umbilical cord is a simple procedure that may improve mortality in this population, but more research is needed; recommended times to delayed clamping range from 30 seconds to 3 minutes, they noted.
In a study published in The Lancet Child & Adolescent Health, the researchers randomized 767 very preterm infants to delayed clamping at least 60 seconds after birth and 764 to immediate clamping. Of these, 384 were multiple births (who were individually randomized), 862 were male, and 505 were born before 27 weeks’ gestation. The primary outcome was death or disability at 2 years of age. Major disability was defined as cerebral palsy, severe visual loss, deafness requiring a hearing aid or cochlear implants, major language or speech problems, or cognitive delay at 2 years corrected age. The median time to clamping was 60 seconds in the delayed group and 5 seconds in the immediate group.
Primary outcome data were available for 1,419 infants. Death or major disability occurred in 29% of infants assigned to delayed clamping compared to 34% of those assigned to immediate clamping (relative risk 0.83, P = .010). The infants were part of the APTS Childhood Follow-Up Study, an open-label superiority trial conducted in Australia and New Zealand.
By age 2 years, 8% of infants in the delayed group and 11% of those in the immediate group had died; 23% and 26%, respectively, met criteria for major disability. The impact of delayed clamping translates to a 30% reduction in relative risk of mortality at 2 years of age, but no significant impact on major disability, the researchers wrote.
The findings were limited by several factors including the unblinded study design, lack of data on heart rate or time to first breath, and the clamping prior to 60 seconds in 26% of infants in the delayed group based on clinical concerns for these specific infants, the researchers noted.
However, the results were strengthened by the large size, low risk of bias, and specific primary outcome, they said. The data support findings from recent systematic reviews and highlight the need for further trials to evaluate delayed clamping at different time points, with larger populations, inclusion of time to first breath and heart rate, and improved measures of disability, the researchers added.
In clinical practice, “Given that aiming to delay cord clamping for 60 seconds or more improved 2-year outcomes and short-term hematological measures with no evidence of significant harm, it seems reasonable to conclude that delayed clamping is appropriate as standard care in very preterm infants,” they concluded.
Accepting simple intervention could have great impact
This study is important in light of the overwhelming burden of preterm birth on the health care system and society as a whole, Lisette D. Tanner, MD, of Emory University, Atlanta, said in an interview.
“Preterm birth is associated with billions in health care costs each year, and a large portion of that money is directed to the complications associated with preterm birth, such as early intervention services, educational support, and ongoing medical care,” Dr. Tanner said. “This study is particularly timely, as we are quickly approaching 2030, the deadline for achieving the United Nations Sustainable Development Goal of ending preventable deaths of newborns and children under 5 years of age,” she said. The goal involves “all countries aiming to reduce neonatal mortality to at least as low as 12 per 1,000 live births and under-5 mortality to at least as low as 25 per 1,000 live births. Effective treatments to reduce infant and child mortality would make strong inroads toward this goal,” she explained.
Dr. Tanner said she was not surprised by the findings because previous studies have shown similar results. “However, the large, multicenter nature of this study provides additional weight to recommendations to delay cord clamping as standard practice,” she said.
“The findings of this study support the recommendations of a number of large organizations,” said Dr. Tanner. “The World Health Organization recommends that the umbilical cord not be clamped earlier than 1 minute after birth in term or preterm infants who do not require positive pressure ventilation. The American College of Obstetricians and Gynecologists and the American Academy of Pediatrics now recommend a delay in umbilical cord clamping in vigorous term and preterm infants for at least 30–60 seconds after birth,” she said. “The Royal College of Obstetricians and Gynaecologists also recommends deferring umbilical cord clamping for healthy term and preterm infants for at least 2 minutes after birth,” she added.
However, “the delay in adoption of this guidelines in practice appears to be related to some concerns regarding universal adoption of this approach,” Dr. Tanner noted. “Some clinicians have suggested that delayed cord clamping could delay vital neonatal resuscitative efforts, leading to worse neonatal outcomes, but this concern has not been borne out in the data, as all guidelines specifically state that this intervention is for vigorous newborns,” she said. “In fact, in preterm infants, delayed cord clamping is associated with improved transitional circulation, decreased need for blood transfusion, and lower incidence of necrotizing enterocolitis and intraventricular hemorrhage,” Dr. Tanner emphasized. “Additionally, concerns persist that delayed cord clamping could lead to excessive transfusion with resultant polycythemia. Again, no data have supported this claim to date,” she said.
“Finally, some clinicians are concerned that delayed clamping could lead to delay in addressing maternal complications of birth such as hemorrhage, but studies have shown the opposite; delayed umbilical cord clamping has not been associated with an increased risk of postpartum hemorrhage or increased blood loss at delivery, nor has it been with a difference in the need for blood transfusion,” said Dr. Tanner.
Ideally, practitioners will become more comfortable in delaying cord clamping as a routine practice as more data demonstrating the safety and benefit of this easy intervention are disseminated, she said.
Additional research delineating which gestational ages benefit most from delayed cord clamping would help direct education efforts to implement this intervention, Dr. Tanner noted.
The study was funded by the Australian National Health and Medical Research Council. The researchers and Dr. Tanner had no financial conflicts to disclose.
FROM THE LANCET CHILD & ADOLESCENT HEALTH
ASH studies look at racial disparities in ALL care, outcomes
Among almost 25,000 children and young adults up to 31 years old, all of whom participated in Children’s Oncology Group studies since 2004, 5-year event free survival (EFS) was 87.4% for White patients, 82.8% for Hispanic patients, and 81.9% for Black patients.
When socioeconomics and disease characteristics such as CNS involvement, white blood cell lineage, and induction status were taken into account, the risk of having a survival event fell from 37% to 11% higher for Hispanic patients versus White patients but from 45% to 32% for Black patients versus White patients.
However, there was no explicit adjustment in the study for acuity at presentation, body mass index, adherence to protocols, or Philadelphia chromosome (PH)-like disease, which is more common among Hispanic patients.
Even so, lead investigator Sumit Gupta, MD, a pediatric blood cancer specialist at the University of Toronto, said that even with the potential confounders, lingering differences in outcomes raise questions about equal access to care and other matters, and suggest that there are still “uncomfortable things to consider, things like ... structural racism” and a system that delivers “systemically different care to patients across racial” groups.
Another report presented at the meeting with 295 patients 18-40 years old found that Hispanic patients had 3-year overall survival comparable to that of White patients despite a higher prevalence of PH-like disease, perhaps because Hispanic patients had higher treatment adherence than did White patients at 76% versus 56%, said lead investigator Lori Muffly, MD, a bone and marrow transplant specialist at Stanford (Calif.) University.
However, Hispanic ALL patients were underrepresented in the study because the investigators didn’t recruit in Texas and Florida, states with higher percentages of young Hispanic ALL patients, and recruitment in California fell short of the prevalence of young Hispanic patients in that state. The work was a substudy of CALGB 10403, a trial of pediatric regimens in adolescents and young adults.
“It’s a relatively easy maneuver, going to where the patients are. When groups are thinking about multicenter trials, it has to be part of the dialogue from the beginning,” Dr. Muffly said.
Black patients in the review had fewer days in treatment and a higher prevalence of T-cell disease, and didn’t do as well as other groups.
Together, the studies “offer insight into the magnitude of racial and ethnic disparities in care among young people with” ALL, said Mikkael Sekeres, MD, chief of the division of hematology at the University of Miami, who moderated the presentations.
Dr. Gupta and his team found outcome differences only in relapsed B-cell ALL, not T-cell disease. B-cell disease has a more rigorous maintenance schedule, so it could be that there’s a difference in sticking to follow-up between various groups or less rigorous monitoring by pediatric oncologists in some groups, he said.
Dr. Gupta’s study was funded by the National Cancer Institute and others. Dr. Muffly didn’t report a funding source, but reported ties to Pfizer, Amgen, and other companies. Dr. Gupta is involved with Jazz Pharmaceuticals.
Among almost 25,000 children and young adults up to 31 years old, all of whom participated in Children’s Oncology Group studies since 2004, 5-year event free survival (EFS) was 87.4% for White patients, 82.8% for Hispanic patients, and 81.9% for Black patients.
When socioeconomics and disease characteristics such as CNS involvement, white blood cell lineage, and induction status were taken into account, the risk of having a survival event fell from 37% to 11% higher for Hispanic patients versus White patients but from 45% to 32% for Black patients versus White patients.
However, there was no explicit adjustment in the study for acuity at presentation, body mass index, adherence to protocols, or Philadelphia chromosome (PH)-like disease, which is more common among Hispanic patients.
Even so, lead investigator Sumit Gupta, MD, a pediatric blood cancer specialist at the University of Toronto, said that even with the potential confounders, lingering differences in outcomes raise questions about equal access to care and other matters, and suggest that there are still “uncomfortable things to consider, things like ... structural racism” and a system that delivers “systemically different care to patients across racial” groups.
Another report presented at the meeting with 295 patients 18-40 years old found that Hispanic patients had 3-year overall survival comparable to that of White patients despite a higher prevalence of PH-like disease, perhaps because Hispanic patients had higher treatment adherence than did White patients at 76% versus 56%, said lead investigator Lori Muffly, MD, a bone and marrow transplant specialist at Stanford (Calif.) University.
However, Hispanic ALL patients were underrepresented in the study because the investigators didn’t recruit in Texas and Florida, states with higher percentages of young Hispanic ALL patients, and recruitment in California fell short of the prevalence of young Hispanic patients in that state. The work was a substudy of CALGB 10403, a trial of pediatric regimens in adolescents and young adults.
“It’s a relatively easy maneuver, going to where the patients are. When groups are thinking about multicenter trials, it has to be part of the dialogue from the beginning,” Dr. Muffly said.
Black patients in the review had fewer days in treatment and a higher prevalence of T-cell disease, and didn’t do as well as other groups.
Together, the studies “offer insight into the magnitude of racial and ethnic disparities in care among young people with” ALL, said Mikkael Sekeres, MD, chief of the division of hematology at the University of Miami, who moderated the presentations.
Dr. Gupta and his team found outcome differences only in relapsed B-cell ALL, not T-cell disease. B-cell disease has a more rigorous maintenance schedule, so it could be that there’s a difference in sticking to follow-up between various groups or less rigorous monitoring by pediatric oncologists in some groups, he said.
Dr. Gupta’s study was funded by the National Cancer Institute and others. Dr. Muffly didn’t report a funding source, but reported ties to Pfizer, Amgen, and other companies. Dr. Gupta is involved with Jazz Pharmaceuticals.
Among almost 25,000 children and young adults up to 31 years old, all of whom participated in Children’s Oncology Group studies since 2004, 5-year event free survival (EFS) was 87.4% for White patients, 82.8% for Hispanic patients, and 81.9% for Black patients.
When socioeconomics and disease characteristics such as CNS involvement, white blood cell lineage, and induction status were taken into account, the risk of having a survival event fell from 37% to 11% higher for Hispanic patients versus White patients but from 45% to 32% for Black patients versus White patients.
However, there was no explicit adjustment in the study for acuity at presentation, body mass index, adherence to protocols, or Philadelphia chromosome (PH)-like disease, which is more common among Hispanic patients.
Even so, lead investigator Sumit Gupta, MD, a pediatric blood cancer specialist at the University of Toronto, said that even with the potential confounders, lingering differences in outcomes raise questions about equal access to care and other matters, and suggest that there are still “uncomfortable things to consider, things like ... structural racism” and a system that delivers “systemically different care to patients across racial” groups.
Another report presented at the meeting with 295 patients 18-40 years old found that Hispanic patients had 3-year overall survival comparable to that of White patients despite a higher prevalence of PH-like disease, perhaps because Hispanic patients had higher treatment adherence than did White patients at 76% versus 56%, said lead investigator Lori Muffly, MD, a bone and marrow transplant specialist at Stanford (Calif.) University.
However, Hispanic ALL patients were underrepresented in the study because the investigators didn’t recruit in Texas and Florida, states with higher percentages of young Hispanic ALL patients, and recruitment in California fell short of the prevalence of young Hispanic patients in that state. The work was a substudy of CALGB 10403, a trial of pediatric regimens in adolescents and young adults.
“It’s a relatively easy maneuver, going to where the patients are. When groups are thinking about multicenter trials, it has to be part of the dialogue from the beginning,” Dr. Muffly said.
Black patients in the review had fewer days in treatment and a higher prevalence of T-cell disease, and didn’t do as well as other groups.
Together, the studies “offer insight into the magnitude of racial and ethnic disparities in care among young people with” ALL, said Mikkael Sekeres, MD, chief of the division of hematology at the University of Miami, who moderated the presentations.
Dr. Gupta and his team found outcome differences only in relapsed B-cell ALL, not T-cell disease. B-cell disease has a more rigorous maintenance schedule, so it could be that there’s a difference in sticking to follow-up between various groups or less rigorous monitoring by pediatric oncologists in some groups, he said.
Dr. Gupta’s study was funded by the National Cancer Institute and others. Dr. Muffly didn’t report a funding source, but reported ties to Pfizer, Amgen, and other companies. Dr. Gupta is involved with Jazz Pharmaceuticals.
FROM ASH 2021
Adaptive therapy borrows from nature to keep rhabdomyosarcoma in check
In 1859, Charles Darwin published “On the Origin of Species,” which outlined his world-shaking theory of evolution and its core principle of natural selection caused by environmental pressures that may determine whether an organism adapts and survives, or remains static, languishes, and eventually dies out.
The same forces that have influenced the size and shape of the beaks of finches in the Galapagos Islands, the length of giraffe necks in Africa, and the intestinal microbiomes of the nearly 8 billion human inhabitants of this planet also control whether malignant cells thrive and multiply, wither and die when assaulted by chemotherapy, or go into hiding, mutating and waiting for their next opportunity to erupt again and metastasize.
The ability of malignant cells to adapt to environmental pressures is “cancer’s most lethal and sophisticated property,” said Damon R. Reed, MD, program leader of the adolescent/young adult program at Moffitt Cancer Center in Tampa, Fla.
Dr. Reed and colleagues are developing methods to meet cancer on its own terms, applying evolutionary principles to the treatment of childhood fusion-positive rhabdomyosarcoma in an innovative, and some would say audacious, clinical trial.
Adaptive versus conventional therapy
The trial, now recruiting, is designed to evaluate each of four different strategies for chemotherapy schedules in patients with newly diagnosed metastatic fusion-positive rhabdomyosarcoma.
The trial contains four arms, three of which consist of either conventional chemotherapy based on published clinical trials, moving a second-line therapy to the first line, or adding maintenance therapy, all of which have the goal of inducing as many complete remissions as possible.
The remaining adaptive therapy arm, however, is entirely novel in approach, with therapy using a combination of chemotherapy drugs that will be started and interrupted based on tumor responses, with resumption of therapy on an adaptive schedule unique to each patient. The goal of treatment for patients enrolled in this arm will be prolongation of the time to disease progression, rather than complete remission.
Although some people might consider the adaptive therapy approach to be sacrificing the hope for a cure in exchange for palliation, the hard truth is that patients with fusion-positive rhabdomyosarcoma (in contrast to those with fusion-negative disease) have a dismal prognosis following relapse after up-front intensified therapy.
Instead, because a cure is exceedingly unlikely in patients with metastatic disease, the conventional idea of delivering the maximum tolerated dose of chemotherapy until disease progression could be replaced by an approach based on understanding of the evolution of cancer cells under selective pressures, Dr. Reed and colleagues contend.
“Although adaptive therapy would represent a major paradigm shift in pediatric oncology, this approach would exploit the chemotherapy-sensitive population to prevent the emergence of resistant populations, optimizing tumor control with less toxicity,” they wrote in a commentary published online in the journal Cancer.1
Poor survival with advanced disease
Childhood rhabdomyosarcoma (RMS) is a form of soft tissue sarcoma of mesenchymal origin. Approximately 25% of cases are parameningeal, arising from sites adjacent to the meninges of the nasopharynx, middle ear, paranasal sinuses, orbit, and other regions of the head and neck. Approximately 31% of cases arise in the genitourinary tract and 13% in the extremities, and other tumors occur less commonly in the trunk, chest wall, perineal/anal region, and abdomen.
The overall 5-year survival rate is approximately 71%.1
However, for patients with high-risk disease, a group that includes children 10 years of age or older with widespread disease with or without an activating PAX/FOX01 gene fusion, 5-year survival ranges from just 20% to 30% (Cancer Facts & Figures 2020).
“Among patients with metastatic disease, there is a clear difference in overall survival between those who have fusion-positive disease, where the 5-year overall survival is about 19%, and patients with fusion-negative disease,” said Douglas S. Hawkins, MD, chair of the children’s oncology group and professor of pediatrics at the University of Washington, Seattle, and associate chief in the division of hematology/oncology at Seattle Children’s Hospital.
Patients with fusion-negative disease can be further classified into those with multiple metastatic sites, with a 5-year overall survival rate of approximately 45%, and those with a single metastatic site, with a 5-year overall survival rate of 70%, he said in an interview.
“So when we talk about metastatic rhabdomyosarcoma, there actually is a diversity of outcomes, between really bad – those with fusion-positive disease – and not terrible – not great, but not terrible – for a selected group of patients with fusion-negative disease,” Dr. Hawkins said.
The poor prognosis for patients with metastatic fusion-positive disease prompted Dr. Reed and colleagues to rethink the entire approach to advanced cancers.
“If someone has a sarcoma, we know that we need to do surgery and radiation to the area, we know that localized disease does better than metastatic disease, and we generally hit it with some kind of chemotherapy that we call ‘standard of care,’ ” he said in an interview.
This approach is largely effective in some forms of cancer of bone and soft tissues, such as Ewing sarcoma, he notes, which has 5-year survival rates below 20% when treated with surgery and radiation only, but with the addition of chemotherapy has 5-year overall survival rates as high as 80%.
“At other times, with other sarcomas, the cure rate is abysmal, but we still call it standard of care,” Dr. Reed said.
For example, patients with metastatic fusion-positive RMS may have an initial response to chemotherapy, but most will eventually experience relapse and die of the disease.
“With some of the most common treatments, 70% of patients will have their cancers shrink by more than 50%, which is a major response, but the vast majority of them will have a recurrence later on,” Dr. Hawkins said.
He noted that the standard of care for patients with metastatic rhabdomyosarcoma, both with and without the PAX/FOX01 fusion, is chemotherapy, generally with the VAC regimen (vincristine, actinomycin D, and cyclophosphamide), although other agents such as doxorubicin, ifosfamide, etoposide, or irinotecan have also been tried, with little effect on event-free survival or overall survival rates.
A life too brief
Ricky Huff and his family know the course that the disease can take only too well. In 2015, his 5-month-old son, Theo, was diagnosed with metastatic rhabdomyosarcoma and put under the care of Damon Reed at Moffitt.
“During the whole course of treatment – I’m sure like many other parents – apart from relying on Damon and his treatment expertise to try to determine the best treatment options, I was reading everything under the sun to try to get a working knowledge of what Theo was up against, what his treatment and clinical trial options were, and what was the state of the science,” Mr. Huff says.
Unfortunately, the characteristics of Theo’s disease, including his very young age at onset and diagnosis of stage 4 metastatic disease, conspired against him, and despite undergoing 14 months of chemotherapy, Theo died of the disease in October 2016, 5 months shy of what would have been his second birthday.
In their grief, Mr. Huff, a real estate lawyer with a practice in Clearwater, Fla., and his wife, Leah, were determined to help other families of children with cancer and settled on the National Pediatric Cancer Foundation. Mr. Huff joined the board of directors of the foundation, which is collaborating with Moffitt Cancer Center on the adaptive therapy trial.
An evolutionary primer (cancer edition)
To get a better idea of just how adaptive therapy works, it is helpful to view cancer cells through the lens of species development, adaptation, extinction, and evolution.
“Cancer cells compete against each other in a dynamic environment. Their tumor ecosystems exhibit spatial and temporal fluctuations in blood-borne nutrients, oxygen, growth factors, immune cells, and hormones,” Dr. Reed and colleagues wrote.
These influences can affect genetically identical cancer cells, which may begin to diverge from one another depending on their location in a tumor and the availability of nutrients, which in turn can result in two once-identical cells exhibiting different transcription rates for growth factors.
“Ultimately, this may affect the rate of progression through the cell cycle, leading to distinct rates of proliferation and mutational acquisition,” they wrote.
The diverging subpopulations will begin to develop different methods for adapting to the tumor microenvironment, with unique strategies for both accelerating growth and avoiding hazards such as chemotherapy drugs or radiation, the investigators explained.
“By the time a cancer becomes clinically apparent, cancer cells have transformed from a single clone into a diverse community of cell types evolving in response to a spatially and temporally heterogeneous tumor environment. Theoretically, a 10-gram cancer may contain the same order of magnitude of cancer cells as there are humans on earth, with tremendous diversity of phenotypes and environments,” they wrote.
Survival of the fittest
The competition of individuals within and between species described by Darwin also applies to cancer cells, in their interactions both with each other and with stromal cells and immune cells resulting in “the progressive replacement of less fit phenotypes by those that are more fit,” Dr. Reed and colleagues explained.
And just like the old joke about two hikers trying to escape from a charging grizzly bear (one says, “This is futile – we can’t outrun a grizzly,” and the other says, “I only have to outrun you!”), cancer cells only need to be more resistant to therapeutic attack than normal cells that are critical to function.
“This may explain why initial responses in certain solid tumors (notably rhabdomyosarcoma) do not predict eventual survival. The sensitivities of the dominant cancer cell populations dictate the initial response, but it is the ecology and evolution of the rare and more resistant populations that determine cure or relapse,” they wrote.
The endangered species list
As with many types of cancer, the current approach to treating pediatric sarcomas with curative intent is with a “first strike” approach, treating patients with surgery, radiation, and cytotoxic chemotherapy at the maximum tolerated dose for as long as needed or until unacceptable toxicities occur, with the intention of wiping out all cancer cells without permanently injuring normal cells.
The evolutionary analogy to this approach is a mass extinction event such as the meteor strike that is believed to have wiped out the dinosaurs roughly 66 million years ago. Fossil evidence suggests that the cataclysmic event resulted in the atmosphere being blanketed with dust particles that blocked sunlight and caused massive die-off of plants that dinosaurs needed to survive and were ill-adapted to do without.
In contrast, populations of smaller, more adaptable species of microbes, insects, and animals, including our mammalian ancestors, were able to survive and eventually flourish.
Many patients with localized cancers may be cured with up-front therapy, but others will have residual disease from populations of cells that are intrinsically resistant to therapy or have developed new evasion strategies.
Strike two and the MVP
Dr. Reed and colleagues liken the approach of second-line therapy for treatment of relapsed or refractory disease to the concept of “background extinctions,” using the fate of the passenger pigeon as an example of how a second-strike therapeutic strategy works.
Although the popular conception is that the passenger pigeon was hunted to extinction by humans, the species in fact died out because of many different factors, including loss of habitat, isolation of populations leading to a loss of genetic diversity, and disruption of breeding habits.
“Once first strikes of deforestation and hunting reduced the birds to small, fragmented populations, a series of what would otherwise have been minor second strikes pushed the passenger pigeon below its extinction threshold, or minimum viable population,” they said.
The analogy, as it applies to cancer therapy, is the use of second-line or follow-on therapy with one or more agents that the residual cells are at least in theory not resistant to. In the case of fusion-positive rhabdomyosarcoma, the drug most commonly added in the second-strike approach is vinorelbine.2
“Second strikes should be timed to occur around the time when the first strike has achieved its greatest effect, presumably at the point when the disease becomes clinically undetectable or at a measurable nadir,” Dr. Reed and colleagues wrote. “Ideally, second-strike therapies should have modes of action that require different resistance strategies by the cancer cells than those needed for resistance to the first strike.”
Adaptive therapy
As Dr. Reed and colleagues note, despite optimal therapy, 94% of patients with metastatic fusion-positive rhabdomyosarcoma will experience a relapse within 3 years of diagnosis.1 Clearly the scorched earth or “throw everything you have it” approach no longer works, and that’s where adaptive therapy comes in.
Here again, the authors rely on nature, or rather human interaction with nature, to devise a strategy for keeping the disease at bay when extinction of all cancerous cells cannot be achieved.
They cite the example of agricultural integrated pest management, which seeks to keep harmful insects in check by treating them to suppress but not completely destroy a population, then stopping the use of pesticides, and resuming only when the insect population spikes and again becomes a threat to crops.
“The goal is to limit crop damage while retaining the sensitivity of the insects to the pesticides. Resistance most often comes at a cost. In the absence of the pesticide, sensitive individuals will outcompete resistant individuals,” they wrote.
Adaptive therapy uses the same approach to reduce selection pressures that foster resistance, with patients treated only until a specific, predetermined response is achieved in the dominant population of chemosensitive cells. The treatment is then interrupted and reintroduced only when the tumor rebounds to a certain predetermined size.
In this scenario, cells that retain sensitivity to chemotherapy will be able to reproduce and proliferate more rapidly than drug-resistant cells, and the therapy can then be reintroduced. This strategy is less likely to cause the development and proliferation of resistant cells than conventional intensified chemotherapy, Dr. Reed and colleagues contend.
Putting it to the test
The clinical trial that Dr. Reed and colleagues have initiated, officially titled “Evolutionary Inspired Therapy for Newly Diagnosed, Metastatic, Fusion Positive Rhabdomyosarcoma,” (NCT04388839) contains four arms: three experimental and one active comparator arm.
“We won’t randomize; we don’t feel that it would be fair to randomize patients, because these arms are so different from each other,” Dr. Reed said.
Arm A is the experimental first-strike arm, a 42-week course containing cyclophosphamide delivered intravenously over 60 minutes at a dose ranging from 220 mg to 1200 mg, vinorelbine delivered in an IV push over 6-10 minutes with a dose ranging from 4 mg to 25 mg, and actinomycin D administered via IV over 3-5 minutes at a dose ranging from 0.025 mg to 0.04 mg.
“The idea is that we take the standard of care, and we add a drug – vinorelbine – to make it stronger,” Dr. Reed said. “The idea is that the resistant cell, the cell that escapes, if we start hitting it on day 1 with vinorelbine, we might be able to drive it to extinction.”
Arm B, the second experimental arm, is the second-strike and maintenance arm, in which patients will receive conventional doses of vincristine, actinomycin D, and cyclophosphamide (VAC) until complete response (CR) for 12-42 weeks, and will then be switched to up to 2 years of maintenance with vinorelbine and oral cyclophosphamide.
“Vinorelbine will be added when the cancer is declining or first goes into remission. We try not to wait 42 weeks, which is too long we think, by which time the cancer may be fully adapted and resistant,” he explained.
Arm C is the adaptive therapy arm, in which patients will receive VAC that starts and stops based on response, with the goal of prolonging time to disease progression rather than achieving CR.
Arm D is the active comparator arm, consisting of conventional chemotherapy based on published clinical trials, such as VAC for 42 weeks, or other standard-of-care regimens that may include irinotecan, doxorubicin, ifosfamide, and/or etoposide.
A change in thinking
Dr. Reed acknowledges that Arm C, the adaptive therapy arm, “definitely represents a change in thinking for pediatric oncology.”
“The idea is that if you could do this perfectly well, you would be able to take a patient who is diagnosed today and essentially ‘pause’ their disease for a while. Then 5 years from now, if there is a better medicine, you would have gotten that patient to that medicine.”
The optimal approach to treating metastatic fusion-positive rhabdomyosarcoma may be similar to that used for treatment of acute lymphoblastic leukemia, with induction, consolidation, and maintenance and the option of delayed intensification, he said.
“But we’re so far away from knowing which series to do that we just need to show that any series – any changing it up – is helpful.”
Dr. Reed said that when he started presenting the concept of adaptive therapy in clinical meetings in 2017, “I was told to come up with a better idea. There were several people who instantly got it, but most people would instantly get angry.”
The common refrain was that adaptive therapy was “giving up.”
But minds began to change in 2018, following presentation at the annual meeting of the American Society of Clinical Oncology of a European study showing that adding 6 months of low-dose chemotherapy maintenance to standard therapy improved the 5-year overall survival rate of pediatric rhabdomyosarcoma from 73.7% to 86.6%.2
Before presenting the idea of adaptive therapy to his colleagues, he ran it by the parents of children with advanced sarcomas, and many were on board with it, he said.
Ricky Huff said that had the option of adaptive therapy been available for Theo, he and his wife would have been willing to try it.
“Of course, everyone has the ability in hindsight to apply critical thinking to decisions that you made or could have made,” he said. “I think is true for many parents, who if they’re presented with information about options will say ‘well if there’s a 1 percent chance, I want that chance for my child, especially for a 5-month-old.”
The decision to choose adaptive therapy is a difficult decision to make, whether for oneself or for one’s son, because it isn’t curative.
“My wife and I have since had a conversation about this, and I do think we would have considered it, although through a lot of difficult conversations,” he said.
“After we got the pathology, knowing that it was metastatic, fusion-positive, and given his age, just doing a brief literature review on my own, I knew what we were up against using 20-year-old treatments, and that the chance of a cure was very, very small.”
If parents of children with metastatic, poor-prognosis rhabdomyosarcoma could be made to understand that adaptive therapy would entail shorter and fewer hospital stays, and cumulatively less toxic chemotherapy, and could prolong the lives of their children, the option might be more acceptable, he said.
And as Dr. Reed mentioned, prolonging time to progression offers hope of additional therapies to come.
“The whole time that my son was being treated, I hoped that there was going to be something else that came out, that a new trial would be launched because they found a way to drug a mutation, or treat it with immunotherapy – something that was going to give us a better option.”
Asked whether he would be willing to share his experiences in this article, Mr. Huff said that “I am willing to, in whatever small way I can, make an impact, and hopefully save another family from what we experienced.”
References
1. Reed DR et al. Cancer. 2020 Jun 1;126(11):2577-87 2. Bisogno G et al. J Clin Oncol. 2018;36:18_suppl,LBA-2
In 1859, Charles Darwin published “On the Origin of Species,” which outlined his world-shaking theory of evolution and its core principle of natural selection caused by environmental pressures that may determine whether an organism adapts and survives, or remains static, languishes, and eventually dies out.
The same forces that have influenced the size and shape of the beaks of finches in the Galapagos Islands, the length of giraffe necks in Africa, and the intestinal microbiomes of the nearly 8 billion human inhabitants of this planet also control whether malignant cells thrive and multiply, wither and die when assaulted by chemotherapy, or go into hiding, mutating and waiting for their next opportunity to erupt again and metastasize.
The ability of malignant cells to adapt to environmental pressures is “cancer’s most lethal and sophisticated property,” said Damon R. Reed, MD, program leader of the adolescent/young adult program at Moffitt Cancer Center in Tampa, Fla.
Dr. Reed and colleagues are developing methods to meet cancer on its own terms, applying evolutionary principles to the treatment of childhood fusion-positive rhabdomyosarcoma in an innovative, and some would say audacious, clinical trial.
Adaptive versus conventional therapy
The trial, now recruiting, is designed to evaluate each of four different strategies for chemotherapy schedules in patients with newly diagnosed metastatic fusion-positive rhabdomyosarcoma.
The trial contains four arms, three of which consist of either conventional chemotherapy based on published clinical trials, moving a second-line therapy to the first line, or adding maintenance therapy, all of which have the goal of inducing as many complete remissions as possible.
The remaining adaptive therapy arm, however, is entirely novel in approach, with therapy using a combination of chemotherapy drugs that will be started and interrupted based on tumor responses, with resumption of therapy on an adaptive schedule unique to each patient. The goal of treatment for patients enrolled in this arm will be prolongation of the time to disease progression, rather than complete remission.
Although some people might consider the adaptive therapy approach to be sacrificing the hope for a cure in exchange for palliation, the hard truth is that patients with fusion-positive rhabdomyosarcoma (in contrast to those with fusion-negative disease) have a dismal prognosis following relapse after up-front intensified therapy.
Instead, because a cure is exceedingly unlikely in patients with metastatic disease, the conventional idea of delivering the maximum tolerated dose of chemotherapy until disease progression could be replaced by an approach based on understanding of the evolution of cancer cells under selective pressures, Dr. Reed and colleagues contend.
“Although adaptive therapy would represent a major paradigm shift in pediatric oncology, this approach would exploit the chemotherapy-sensitive population to prevent the emergence of resistant populations, optimizing tumor control with less toxicity,” they wrote in a commentary published online in the journal Cancer.1
Poor survival with advanced disease
Childhood rhabdomyosarcoma (RMS) is a form of soft tissue sarcoma of mesenchymal origin. Approximately 25% of cases are parameningeal, arising from sites adjacent to the meninges of the nasopharynx, middle ear, paranasal sinuses, orbit, and other regions of the head and neck. Approximately 31% of cases arise in the genitourinary tract and 13% in the extremities, and other tumors occur less commonly in the trunk, chest wall, perineal/anal region, and abdomen.
The overall 5-year survival rate is approximately 71%.1
However, for patients with high-risk disease, a group that includes children 10 years of age or older with widespread disease with or without an activating PAX/FOX01 gene fusion, 5-year survival ranges from just 20% to 30% (Cancer Facts & Figures 2020).
“Among patients with metastatic disease, there is a clear difference in overall survival between those who have fusion-positive disease, where the 5-year overall survival is about 19%, and patients with fusion-negative disease,” said Douglas S. Hawkins, MD, chair of the children’s oncology group and professor of pediatrics at the University of Washington, Seattle, and associate chief in the division of hematology/oncology at Seattle Children’s Hospital.
Patients with fusion-negative disease can be further classified into those with multiple metastatic sites, with a 5-year overall survival rate of approximately 45%, and those with a single metastatic site, with a 5-year overall survival rate of 70%, he said in an interview.
“So when we talk about metastatic rhabdomyosarcoma, there actually is a diversity of outcomes, between really bad – those with fusion-positive disease – and not terrible – not great, but not terrible – for a selected group of patients with fusion-negative disease,” Dr. Hawkins said.
The poor prognosis for patients with metastatic fusion-positive disease prompted Dr. Reed and colleagues to rethink the entire approach to advanced cancers.
“If someone has a sarcoma, we know that we need to do surgery and radiation to the area, we know that localized disease does better than metastatic disease, and we generally hit it with some kind of chemotherapy that we call ‘standard of care,’ ” he said in an interview.
This approach is largely effective in some forms of cancer of bone and soft tissues, such as Ewing sarcoma, he notes, which has 5-year survival rates below 20% when treated with surgery and radiation only, but with the addition of chemotherapy has 5-year overall survival rates as high as 80%.
“At other times, with other sarcomas, the cure rate is abysmal, but we still call it standard of care,” Dr. Reed said.
For example, patients with metastatic fusion-positive RMS may have an initial response to chemotherapy, but most will eventually experience relapse and die of the disease.
“With some of the most common treatments, 70% of patients will have their cancers shrink by more than 50%, which is a major response, but the vast majority of them will have a recurrence later on,” Dr. Hawkins said.
He noted that the standard of care for patients with metastatic rhabdomyosarcoma, both with and without the PAX/FOX01 fusion, is chemotherapy, generally with the VAC regimen (vincristine, actinomycin D, and cyclophosphamide), although other agents such as doxorubicin, ifosfamide, etoposide, or irinotecan have also been tried, with little effect on event-free survival or overall survival rates.
A life too brief
Ricky Huff and his family know the course that the disease can take only too well. In 2015, his 5-month-old son, Theo, was diagnosed with metastatic rhabdomyosarcoma and put under the care of Damon Reed at Moffitt.
“During the whole course of treatment – I’m sure like many other parents – apart from relying on Damon and his treatment expertise to try to determine the best treatment options, I was reading everything under the sun to try to get a working knowledge of what Theo was up against, what his treatment and clinical trial options were, and what was the state of the science,” Mr. Huff says.
Unfortunately, the characteristics of Theo’s disease, including his very young age at onset and diagnosis of stage 4 metastatic disease, conspired against him, and despite undergoing 14 months of chemotherapy, Theo died of the disease in October 2016, 5 months shy of what would have been his second birthday.
In their grief, Mr. Huff, a real estate lawyer with a practice in Clearwater, Fla., and his wife, Leah, were determined to help other families of children with cancer and settled on the National Pediatric Cancer Foundation. Mr. Huff joined the board of directors of the foundation, which is collaborating with Moffitt Cancer Center on the adaptive therapy trial.
An evolutionary primer (cancer edition)
To get a better idea of just how adaptive therapy works, it is helpful to view cancer cells through the lens of species development, adaptation, extinction, and evolution.
“Cancer cells compete against each other in a dynamic environment. Their tumor ecosystems exhibit spatial and temporal fluctuations in blood-borne nutrients, oxygen, growth factors, immune cells, and hormones,” Dr. Reed and colleagues wrote.
These influences can affect genetically identical cancer cells, which may begin to diverge from one another depending on their location in a tumor and the availability of nutrients, which in turn can result in two once-identical cells exhibiting different transcription rates for growth factors.
“Ultimately, this may affect the rate of progression through the cell cycle, leading to distinct rates of proliferation and mutational acquisition,” they wrote.
The diverging subpopulations will begin to develop different methods for adapting to the tumor microenvironment, with unique strategies for both accelerating growth and avoiding hazards such as chemotherapy drugs or radiation, the investigators explained.
“By the time a cancer becomes clinically apparent, cancer cells have transformed from a single clone into a diverse community of cell types evolving in response to a spatially and temporally heterogeneous tumor environment. Theoretically, a 10-gram cancer may contain the same order of magnitude of cancer cells as there are humans on earth, with tremendous diversity of phenotypes and environments,” they wrote.
Survival of the fittest
The competition of individuals within and between species described by Darwin also applies to cancer cells, in their interactions both with each other and with stromal cells and immune cells resulting in “the progressive replacement of less fit phenotypes by those that are more fit,” Dr. Reed and colleagues explained.
And just like the old joke about two hikers trying to escape from a charging grizzly bear (one says, “This is futile – we can’t outrun a grizzly,” and the other says, “I only have to outrun you!”), cancer cells only need to be more resistant to therapeutic attack than normal cells that are critical to function.
“This may explain why initial responses in certain solid tumors (notably rhabdomyosarcoma) do not predict eventual survival. The sensitivities of the dominant cancer cell populations dictate the initial response, but it is the ecology and evolution of the rare and more resistant populations that determine cure or relapse,” they wrote.
The endangered species list
As with many types of cancer, the current approach to treating pediatric sarcomas with curative intent is with a “first strike” approach, treating patients with surgery, radiation, and cytotoxic chemotherapy at the maximum tolerated dose for as long as needed or until unacceptable toxicities occur, with the intention of wiping out all cancer cells without permanently injuring normal cells.
The evolutionary analogy to this approach is a mass extinction event such as the meteor strike that is believed to have wiped out the dinosaurs roughly 66 million years ago. Fossil evidence suggests that the cataclysmic event resulted in the atmosphere being blanketed with dust particles that blocked sunlight and caused massive die-off of plants that dinosaurs needed to survive and were ill-adapted to do without.
In contrast, populations of smaller, more adaptable species of microbes, insects, and animals, including our mammalian ancestors, were able to survive and eventually flourish.
Many patients with localized cancers may be cured with up-front therapy, but others will have residual disease from populations of cells that are intrinsically resistant to therapy or have developed new evasion strategies.
Strike two and the MVP
Dr. Reed and colleagues liken the approach of second-line therapy for treatment of relapsed or refractory disease to the concept of “background extinctions,” using the fate of the passenger pigeon as an example of how a second-strike therapeutic strategy works.
Although the popular conception is that the passenger pigeon was hunted to extinction by humans, the species in fact died out because of many different factors, including loss of habitat, isolation of populations leading to a loss of genetic diversity, and disruption of breeding habits.
“Once first strikes of deforestation and hunting reduced the birds to small, fragmented populations, a series of what would otherwise have been minor second strikes pushed the passenger pigeon below its extinction threshold, or minimum viable population,” they said.
The analogy, as it applies to cancer therapy, is the use of second-line or follow-on therapy with one or more agents that the residual cells are at least in theory not resistant to. In the case of fusion-positive rhabdomyosarcoma, the drug most commonly added in the second-strike approach is vinorelbine.2
“Second strikes should be timed to occur around the time when the first strike has achieved its greatest effect, presumably at the point when the disease becomes clinically undetectable or at a measurable nadir,” Dr. Reed and colleagues wrote. “Ideally, second-strike therapies should have modes of action that require different resistance strategies by the cancer cells than those needed for resistance to the first strike.”
Adaptive therapy
As Dr. Reed and colleagues note, despite optimal therapy, 94% of patients with metastatic fusion-positive rhabdomyosarcoma will experience a relapse within 3 years of diagnosis.1 Clearly the scorched earth or “throw everything you have it” approach no longer works, and that’s where adaptive therapy comes in.
Here again, the authors rely on nature, or rather human interaction with nature, to devise a strategy for keeping the disease at bay when extinction of all cancerous cells cannot be achieved.
They cite the example of agricultural integrated pest management, which seeks to keep harmful insects in check by treating them to suppress but not completely destroy a population, then stopping the use of pesticides, and resuming only when the insect population spikes and again becomes a threat to crops.
“The goal is to limit crop damage while retaining the sensitivity of the insects to the pesticides. Resistance most often comes at a cost. In the absence of the pesticide, sensitive individuals will outcompete resistant individuals,” they wrote.
Adaptive therapy uses the same approach to reduce selection pressures that foster resistance, with patients treated only until a specific, predetermined response is achieved in the dominant population of chemosensitive cells. The treatment is then interrupted and reintroduced only when the tumor rebounds to a certain predetermined size.
In this scenario, cells that retain sensitivity to chemotherapy will be able to reproduce and proliferate more rapidly than drug-resistant cells, and the therapy can then be reintroduced. This strategy is less likely to cause the development and proliferation of resistant cells than conventional intensified chemotherapy, Dr. Reed and colleagues contend.
Putting it to the test
The clinical trial that Dr. Reed and colleagues have initiated, officially titled “Evolutionary Inspired Therapy for Newly Diagnosed, Metastatic, Fusion Positive Rhabdomyosarcoma,” (NCT04388839) contains four arms: three experimental and one active comparator arm.
“We won’t randomize; we don’t feel that it would be fair to randomize patients, because these arms are so different from each other,” Dr. Reed said.
Arm A is the experimental first-strike arm, a 42-week course containing cyclophosphamide delivered intravenously over 60 minutes at a dose ranging from 220 mg to 1200 mg, vinorelbine delivered in an IV push over 6-10 minutes with a dose ranging from 4 mg to 25 mg, and actinomycin D administered via IV over 3-5 minutes at a dose ranging from 0.025 mg to 0.04 mg.
“The idea is that we take the standard of care, and we add a drug – vinorelbine – to make it stronger,” Dr. Reed said. “The idea is that the resistant cell, the cell that escapes, if we start hitting it on day 1 with vinorelbine, we might be able to drive it to extinction.”
Arm B, the second experimental arm, is the second-strike and maintenance arm, in which patients will receive conventional doses of vincristine, actinomycin D, and cyclophosphamide (VAC) until complete response (CR) for 12-42 weeks, and will then be switched to up to 2 years of maintenance with vinorelbine and oral cyclophosphamide.
“Vinorelbine will be added when the cancer is declining or first goes into remission. We try not to wait 42 weeks, which is too long we think, by which time the cancer may be fully adapted and resistant,” he explained.
Arm C is the adaptive therapy arm, in which patients will receive VAC that starts and stops based on response, with the goal of prolonging time to disease progression rather than achieving CR.
Arm D is the active comparator arm, consisting of conventional chemotherapy based on published clinical trials, such as VAC for 42 weeks, or other standard-of-care regimens that may include irinotecan, doxorubicin, ifosfamide, and/or etoposide.
A change in thinking
Dr. Reed acknowledges that Arm C, the adaptive therapy arm, “definitely represents a change in thinking for pediatric oncology.”
“The idea is that if you could do this perfectly well, you would be able to take a patient who is diagnosed today and essentially ‘pause’ their disease for a while. Then 5 years from now, if there is a better medicine, you would have gotten that patient to that medicine.”
The optimal approach to treating metastatic fusion-positive rhabdomyosarcoma may be similar to that used for treatment of acute lymphoblastic leukemia, with induction, consolidation, and maintenance and the option of delayed intensification, he said.
“But we’re so far away from knowing which series to do that we just need to show that any series – any changing it up – is helpful.”
Dr. Reed said that when he started presenting the concept of adaptive therapy in clinical meetings in 2017, “I was told to come up with a better idea. There were several people who instantly got it, but most people would instantly get angry.”
The common refrain was that adaptive therapy was “giving up.”
But minds began to change in 2018, following presentation at the annual meeting of the American Society of Clinical Oncology of a European study showing that adding 6 months of low-dose chemotherapy maintenance to standard therapy improved the 5-year overall survival rate of pediatric rhabdomyosarcoma from 73.7% to 86.6%.2
Before presenting the idea of adaptive therapy to his colleagues, he ran it by the parents of children with advanced sarcomas, and many were on board with it, he said.
Ricky Huff said that had the option of adaptive therapy been available for Theo, he and his wife would have been willing to try it.
“Of course, everyone has the ability in hindsight to apply critical thinking to decisions that you made or could have made,” he said. “I think is true for many parents, who if they’re presented with information about options will say ‘well if there’s a 1 percent chance, I want that chance for my child, especially for a 5-month-old.”
The decision to choose adaptive therapy is a difficult decision to make, whether for oneself or for one’s son, because it isn’t curative.
“My wife and I have since had a conversation about this, and I do think we would have considered it, although through a lot of difficult conversations,” he said.
“After we got the pathology, knowing that it was metastatic, fusion-positive, and given his age, just doing a brief literature review on my own, I knew what we were up against using 20-year-old treatments, and that the chance of a cure was very, very small.”
If parents of children with metastatic, poor-prognosis rhabdomyosarcoma could be made to understand that adaptive therapy would entail shorter and fewer hospital stays, and cumulatively less toxic chemotherapy, and could prolong the lives of their children, the option might be more acceptable, he said.
And as Dr. Reed mentioned, prolonging time to progression offers hope of additional therapies to come.
“The whole time that my son was being treated, I hoped that there was going to be something else that came out, that a new trial would be launched because they found a way to drug a mutation, or treat it with immunotherapy – something that was going to give us a better option.”
Asked whether he would be willing to share his experiences in this article, Mr. Huff said that “I am willing to, in whatever small way I can, make an impact, and hopefully save another family from what we experienced.”
References
1. Reed DR et al. Cancer. 2020 Jun 1;126(11):2577-87 2. Bisogno G et al. J Clin Oncol. 2018;36:18_suppl,LBA-2
In 1859, Charles Darwin published “On the Origin of Species,” which outlined his world-shaking theory of evolution and its core principle of natural selection caused by environmental pressures that may determine whether an organism adapts and survives, or remains static, languishes, and eventually dies out.
The same forces that have influenced the size and shape of the beaks of finches in the Galapagos Islands, the length of giraffe necks in Africa, and the intestinal microbiomes of the nearly 8 billion human inhabitants of this planet also control whether malignant cells thrive and multiply, wither and die when assaulted by chemotherapy, or go into hiding, mutating and waiting for their next opportunity to erupt again and metastasize.
The ability of malignant cells to adapt to environmental pressures is “cancer’s most lethal and sophisticated property,” said Damon R. Reed, MD, program leader of the adolescent/young adult program at Moffitt Cancer Center in Tampa, Fla.
Dr. Reed and colleagues are developing methods to meet cancer on its own terms, applying evolutionary principles to the treatment of childhood fusion-positive rhabdomyosarcoma in an innovative, and some would say audacious, clinical trial.
Adaptive versus conventional therapy
The trial, now recruiting, is designed to evaluate each of four different strategies for chemotherapy schedules in patients with newly diagnosed metastatic fusion-positive rhabdomyosarcoma.
The trial contains four arms, three of which consist of either conventional chemotherapy based on published clinical trials, moving a second-line therapy to the first line, or adding maintenance therapy, all of which have the goal of inducing as many complete remissions as possible.
The remaining adaptive therapy arm, however, is entirely novel in approach, with therapy using a combination of chemotherapy drugs that will be started and interrupted based on tumor responses, with resumption of therapy on an adaptive schedule unique to each patient. The goal of treatment for patients enrolled in this arm will be prolongation of the time to disease progression, rather than complete remission.
Although some people might consider the adaptive therapy approach to be sacrificing the hope for a cure in exchange for palliation, the hard truth is that patients with fusion-positive rhabdomyosarcoma (in contrast to those with fusion-negative disease) have a dismal prognosis following relapse after up-front intensified therapy.
Instead, because a cure is exceedingly unlikely in patients with metastatic disease, the conventional idea of delivering the maximum tolerated dose of chemotherapy until disease progression could be replaced by an approach based on understanding of the evolution of cancer cells under selective pressures, Dr. Reed and colleagues contend.
“Although adaptive therapy would represent a major paradigm shift in pediatric oncology, this approach would exploit the chemotherapy-sensitive population to prevent the emergence of resistant populations, optimizing tumor control with less toxicity,” they wrote in a commentary published online in the journal Cancer.1
Poor survival with advanced disease
Childhood rhabdomyosarcoma (RMS) is a form of soft tissue sarcoma of mesenchymal origin. Approximately 25% of cases are parameningeal, arising from sites adjacent to the meninges of the nasopharynx, middle ear, paranasal sinuses, orbit, and other regions of the head and neck. Approximately 31% of cases arise in the genitourinary tract and 13% in the extremities, and other tumors occur less commonly in the trunk, chest wall, perineal/anal region, and abdomen.
The overall 5-year survival rate is approximately 71%.1
However, for patients with high-risk disease, a group that includes children 10 years of age or older with widespread disease with or without an activating PAX/FOX01 gene fusion, 5-year survival ranges from just 20% to 30% (Cancer Facts & Figures 2020).
“Among patients with metastatic disease, there is a clear difference in overall survival between those who have fusion-positive disease, where the 5-year overall survival is about 19%, and patients with fusion-negative disease,” said Douglas S. Hawkins, MD, chair of the children’s oncology group and professor of pediatrics at the University of Washington, Seattle, and associate chief in the division of hematology/oncology at Seattle Children’s Hospital.
Patients with fusion-negative disease can be further classified into those with multiple metastatic sites, with a 5-year overall survival rate of approximately 45%, and those with a single metastatic site, with a 5-year overall survival rate of 70%, he said in an interview.
“So when we talk about metastatic rhabdomyosarcoma, there actually is a diversity of outcomes, between really bad – those with fusion-positive disease – and not terrible – not great, but not terrible – for a selected group of patients with fusion-negative disease,” Dr. Hawkins said.
The poor prognosis for patients with metastatic fusion-positive disease prompted Dr. Reed and colleagues to rethink the entire approach to advanced cancers.
“If someone has a sarcoma, we know that we need to do surgery and radiation to the area, we know that localized disease does better than metastatic disease, and we generally hit it with some kind of chemotherapy that we call ‘standard of care,’ ” he said in an interview.
This approach is largely effective in some forms of cancer of bone and soft tissues, such as Ewing sarcoma, he notes, which has 5-year survival rates below 20% when treated with surgery and radiation only, but with the addition of chemotherapy has 5-year overall survival rates as high as 80%.
“At other times, with other sarcomas, the cure rate is abysmal, but we still call it standard of care,” Dr. Reed said.
For example, patients with metastatic fusion-positive RMS may have an initial response to chemotherapy, but most will eventually experience relapse and die of the disease.
“With some of the most common treatments, 70% of patients will have their cancers shrink by more than 50%, which is a major response, but the vast majority of them will have a recurrence later on,” Dr. Hawkins said.
He noted that the standard of care for patients with metastatic rhabdomyosarcoma, both with and without the PAX/FOX01 fusion, is chemotherapy, generally with the VAC regimen (vincristine, actinomycin D, and cyclophosphamide), although other agents such as doxorubicin, ifosfamide, etoposide, or irinotecan have also been tried, with little effect on event-free survival or overall survival rates.
A life too brief
Ricky Huff and his family know the course that the disease can take only too well. In 2015, his 5-month-old son, Theo, was diagnosed with metastatic rhabdomyosarcoma and put under the care of Damon Reed at Moffitt.
“During the whole course of treatment – I’m sure like many other parents – apart from relying on Damon and his treatment expertise to try to determine the best treatment options, I was reading everything under the sun to try to get a working knowledge of what Theo was up against, what his treatment and clinical trial options were, and what was the state of the science,” Mr. Huff says.
Unfortunately, the characteristics of Theo’s disease, including his very young age at onset and diagnosis of stage 4 metastatic disease, conspired against him, and despite undergoing 14 months of chemotherapy, Theo died of the disease in October 2016, 5 months shy of what would have been his second birthday.
In their grief, Mr. Huff, a real estate lawyer with a practice in Clearwater, Fla., and his wife, Leah, were determined to help other families of children with cancer and settled on the National Pediatric Cancer Foundation. Mr. Huff joined the board of directors of the foundation, which is collaborating with Moffitt Cancer Center on the adaptive therapy trial.
An evolutionary primer (cancer edition)
To get a better idea of just how adaptive therapy works, it is helpful to view cancer cells through the lens of species development, adaptation, extinction, and evolution.
“Cancer cells compete against each other in a dynamic environment. Their tumor ecosystems exhibit spatial and temporal fluctuations in blood-borne nutrients, oxygen, growth factors, immune cells, and hormones,” Dr. Reed and colleagues wrote.
These influences can affect genetically identical cancer cells, which may begin to diverge from one another depending on their location in a tumor and the availability of nutrients, which in turn can result in two once-identical cells exhibiting different transcription rates for growth factors.
“Ultimately, this may affect the rate of progression through the cell cycle, leading to distinct rates of proliferation and mutational acquisition,” they wrote.
The diverging subpopulations will begin to develop different methods for adapting to the tumor microenvironment, with unique strategies for both accelerating growth and avoiding hazards such as chemotherapy drugs or radiation, the investigators explained.
“By the time a cancer becomes clinically apparent, cancer cells have transformed from a single clone into a diverse community of cell types evolving in response to a spatially and temporally heterogeneous tumor environment. Theoretically, a 10-gram cancer may contain the same order of magnitude of cancer cells as there are humans on earth, with tremendous diversity of phenotypes and environments,” they wrote.
Survival of the fittest
The competition of individuals within and between species described by Darwin also applies to cancer cells, in their interactions both with each other and with stromal cells and immune cells resulting in “the progressive replacement of less fit phenotypes by those that are more fit,” Dr. Reed and colleagues explained.
And just like the old joke about two hikers trying to escape from a charging grizzly bear (one says, “This is futile – we can’t outrun a grizzly,” and the other says, “I only have to outrun you!”), cancer cells only need to be more resistant to therapeutic attack than normal cells that are critical to function.
“This may explain why initial responses in certain solid tumors (notably rhabdomyosarcoma) do not predict eventual survival. The sensitivities of the dominant cancer cell populations dictate the initial response, but it is the ecology and evolution of the rare and more resistant populations that determine cure or relapse,” they wrote.
The endangered species list
As with many types of cancer, the current approach to treating pediatric sarcomas with curative intent is with a “first strike” approach, treating patients with surgery, radiation, and cytotoxic chemotherapy at the maximum tolerated dose for as long as needed or until unacceptable toxicities occur, with the intention of wiping out all cancer cells without permanently injuring normal cells.
The evolutionary analogy to this approach is a mass extinction event such as the meteor strike that is believed to have wiped out the dinosaurs roughly 66 million years ago. Fossil evidence suggests that the cataclysmic event resulted in the atmosphere being blanketed with dust particles that blocked sunlight and caused massive die-off of plants that dinosaurs needed to survive and were ill-adapted to do without.
In contrast, populations of smaller, more adaptable species of microbes, insects, and animals, including our mammalian ancestors, were able to survive and eventually flourish.
Many patients with localized cancers may be cured with up-front therapy, but others will have residual disease from populations of cells that are intrinsically resistant to therapy or have developed new evasion strategies.
Strike two and the MVP
Dr. Reed and colleagues liken the approach of second-line therapy for treatment of relapsed or refractory disease to the concept of “background extinctions,” using the fate of the passenger pigeon as an example of how a second-strike therapeutic strategy works.
Although the popular conception is that the passenger pigeon was hunted to extinction by humans, the species in fact died out because of many different factors, including loss of habitat, isolation of populations leading to a loss of genetic diversity, and disruption of breeding habits.
“Once first strikes of deforestation and hunting reduced the birds to small, fragmented populations, a series of what would otherwise have been minor second strikes pushed the passenger pigeon below its extinction threshold, or minimum viable population,” they said.
The analogy, as it applies to cancer therapy, is the use of second-line or follow-on therapy with one or more agents that the residual cells are at least in theory not resistant to. In the case of fusion-positive rhabdomyosarcoma, the drug most commonly added in the second-strike approach is vinorelbine.2
“Second strikes should be timed to occur around the time when the first strike has achieved its greatest effect, presumably at the point when the disease becomes clinically undetectable or at a measurable nadir,” Dr. Reed and colleagues wrote. “Ideally, second-strike therapies should have modes of action that require different resistance strategies by the cancer cells than those needed for resistance to the first strike.”
Adaptive therapy
As Dr. Reed and colleagues note, despite optimal therapy, 94% of patients with metastatic fusion-positive rhabdomyosarcoma will experience a relapse within 3 years of diagnosis.1 Clearly the scorched earth or “throw everything you have it” approach no longer works, and that’s where adaptive therapy comes in.
Here again, the authors rely on nature, or rather human interaction with nature, to devise a strategy for keeping the disease at bay when extinction of all cancerous cells cannot be achieved.
They cite the example of agricultural integrated pest management, which seeks to keep harmful insects in check by treating them to suppress but not completely destroy a population, then stopping the use of pesticides, and resuming only when the insect population spikes and again becomes a threat to crops.
“The goal is to limit crop damage while retaining the sensitivity of the insects to the pesticides. Resistance most often comes at a cost. In the absence of the pesticide, sensitive individuals will outcompete resistant individuals,” they wrote.
Adaptive therapy uses the same approach to reduce selection pressures that foster resistance, with patients treated only until a specific, predetermined response is achieved in the dominant population of chemosensitive cells. The treatment is then interrupted and reintroduced only when the tumor rebounds to a certain predetermined size.
In this scenario, cells that retain sensitivity to chemotherapy will be able to reproduce and proliferate more rapidly than drug-resistant cells, and the therapy can then be reintroduced. This strategy is less likely to cause the development and proliferation of resistant cells than conventional intensified chemotherapy, Dr. Reed and colleagues contend.
Putting it to the test
The clinical trial that Dr. Reed and colleagues have initiated, officially titled “Evolutionary Inspired Therapy for Newly Diagnosed, Metastatic, Fusion Positive Rhabdomyosarcoma,” (NCT04388839) contains four arms: three experimental and one active comparator arm.
“We won’t randomize; we don’t feel that it would be fair to randomize patients, because these arms are so different from each other,” Dr. Reed said.
Arm A is the experimental first-strike arm, a 42-week course containing cyclophosphamide delivered intravenously over 60 minutes at a dose ranging from 220 mg to 1200 mg, vinorelbine delivered in an IV push over 6-10 minutes with a dose ranging from 4 mg to 25 mg, and actinomycin D administered via IV over 3-5 minutes at a dose ranging from 0.025 mg to 0.04 mg.
“The idea is that we take the standard of care, and we add a drug – vinorelbine – to make it stronger,” Dr. Reed said. “The idea is that the resistant cell, the cell that escapes, if we start hitting it on day 1 with vinorelbine, we might be able to drive it to extinction.”
Arm B, the second experimental arm, is the second-strike and maintenance arm, in which patients will receive conventional doses of vincristine, actinomycin D, and cyclophosphamide (VAC) until complete response (CR) for 12-42 weeks, and will then be switched to up to 2 years of maintenance with vinorelbine and oral cyclophosphamide.
“Vinorelbine will be added when the cancer is declining or first goes into remission. We try not to wait 42 weeks, which is too long we think, by which time the cancer may be fully adapted and resistant,” he explained.
Arm C is the adaptive therapy arm, in which patients will receive VAC that starts and stops based on response, with the goal of prolonging time to disease progression rather than achieving CR.
Arm D is the active comparator arm, consisting of conventional chemotherapy based on published clinical trials, such as VAC for 42 weeks, or other standard-of-care regimens that may include irinotecan, doxorubicin, ifosfamide, and/or etoposide.
A change in thinking
Dr. Reed acknowledges that Arm C, the adaptive therapy arm, “definitely represents a change in thinking for pediatric oncology.”
“The idea is that if you could do this perfectly well, you would be able to take a patient who is diagnosed today and essentially ‘pause’ their disease for a while. Then 5 years from now, if there is a better medicine, you would have gotten that patient to that medicine.”
The optimal approach to treating metastatic fusion-positive rhabdomyosarcoma may be similar to that used for treatment of acute lymphoblastic leukemia, with induction, consolidation, and maintenance and the option of delayed intensification, he said.
“But we’re so far away from knowing which series to do that we just need to show that any series – any changing it up – is helpful.”
Dr. Reed said that when he started presenting the concept of adaptive therapy in clinical meetings in 2017, “I was told to come up with a better idea. There were several people who instantly got it, but most people would instantly get angry.”
The common refrain was that adaptive therapy was “giving up.”
But minds began to change in 2018, following presentation at the annual meeting of the American Society of Clinical Oncology of a European study showing that adding 6 months of low-dose chemotherapy maintenance to standard therapy improved the 5-year overall survival rate of pediatric rhabdomyosarcoma from 73.7% to 86.6%.2
Before presenting the idea of adaptive therapy to his colleagues, he ran it by the parents of children with advanced sarcomas, and many were on board with it, he said.
Ricky Huff said that had the option of adaptive therapy been available for Theo, he and his wife would have been willing to try it.
“Of course, everyone has the ability in hindsight to apply critical thinking to decisions that you made or could have made,” he said. “I think is true for many parents, who if they’re presented with information about options will say ‘well if there’s a 1 percent chance, I want that chance for my child, especially for a 5-month-old.”
The decision to choose adaptive therapy is a difficult decision to make, whether for oneself or for one’s son, because it isn’t curative.
“My wife and I have since had a conversation about this, and I do think we would have considered it, although through a lot of difficult conversations,” he said.
“After we got the pathology, knowing that it was metastatic, fusion-positive, and given his age, just doing a brief literature review on my own, I knew what we were up against using 20-year-old treatments, and that the chance of a cure was very, very small.”
If parents of children with metastatic, poor-prognosis rhabdomyosarcoma could be made to understand that adaptive therapy would entail shorter and fewer hospital stays, and cumulatively less toxic chemotherapy, and could prolong the lives of their children, the option might be more acceptable, he said.
And as Dr. Reed mentioned, prolonging time to progression offers hope of additional therapies to come.
“The whole time that my son was being treated, I hoped that there was going to be something else that came out, that a new trial would be launched because they found a way to drug a mutation, or treat it with immunotherapy – something that was going to give us a better option.”
Asked whether he would be willing to share his experiences in this article, Mr. Huff said that “I am willing to, in whatever small way I can, make an impact, and hopefully save another family from what we experienced.”
References
1. Reed DR et al. Cancer. 2020 Jun 1;126(11):2577-87 2. Bisogno G et al. J Clin Oncol. 2018;36:18_suppl,LBA-2
Spice in breast milk could shape taste preferences later
They say you are what you eat, but scientists have long wondered whether breastfeeding babies are what their mothers eat, too. Their question: How much of a nursing mother’s diet eventually plays a role in a child’s food preferences later in life?
The aroma, taste, and makeup of breast milk change from day to day, based mostly on the mother’s diet. But previous research has already shown that the foods a mother eats do not directly translate into the same smells and tastes of that food in breast milk. Some substances from the mother’s diet enter her breast milk, some don’t, and even ones that do may have a different scent or flavor than what the mother experiences.
But a new study suggests that the active ingredient in black pepper makes its way into breast milk and may help the infant develop a tolerance to pepper later. The researchers published their findings in the journal Molecular Nutrition & Food Research.
Pinch of pepper
The study authors thought that maybe some food preferences could result from sensory programming that occurs through breast milk in the first few months of life. Though past studies have looked at which odor-producing substances transfer into breast milk, not many have explored specific substances that give food its distinctive flavor, or even what makes up the taste of breast milk. So they decided to investigate what happens when a mother consumes a meal containing three specific compounds: those that give pepper, chili, and ginger their particularly pungent flavors.
The researchers recruited 18 healthy, nonsmoking, nursing mothers who were producing more than enough milk for their baby’s needs. Their breastfeeding children ranged in age from 8 weeks to 1 year old. The women all ate a curry dish after having spent 2 days avoiding onion, garlic, and the spices in the curry. Then they provided pumped breast milk samples at 1, 2, and 3 hours after eating the curry.
Within an hour of the women eating the curry, the scientists were able to detect piperine, the compound that gives black pepper its bite, in the mothers’ breast milk. They did not find the compounds from ginger, chili, or curcumin – the main active ingredient in turmeric – in the breast milk. The piperine remained there for several hours, but there wasn’t enough for an adult to be able to taste it. It wasn’t possible to reliably tell whether the infants could consciously detect the flavor, but the researchers don’t think it’s likely they did.
But the scientists do suggest it’s possible that the piperine in breast milk could regularly activate a protein that detects pungent or potentially harmful substances. This is the same protein that produces the sensation of heat when eating a spicy food. If the piperine frequently activates that protein in a nursing baby at levels too low for the baby to notice, it may increase the baby’s tolerance for similar spicy substances later in life.
Ultimately, the findings suggest that .
A version of this story first appeared on WebMD.com.
They say you are what you eat, but scientists have long wondered whether breastfeeding babies are what their mothers eat, too. Their question: How much of a nursing mother’s diet eventually plays a role in a child’s food preferences later in life?
The aroma, taste, and makeup of breast milk change from day to day, based mostly on the mother’s diet. But previous research has already shown that the foods a mother eats do not directly translate into the same smells and tastes of that food in breast milk. Some substances from the mother’s diet enter her breast milk, some don’t, and even ones that do may have a different scent or flavor than what the mother experiences.
But a new study suggests that the active ingredient in black pepper makes its way into breast milk and may help the infant develop a tolerance to pepper later. The researchers published their findings in the journal Molecular Nutrition & Food Research.
Pinch of pepper
The study authors thought that maybe some food preferences could result from sensory programming that occurs through breast milk in the first few months of life. Though past studies have looked at which odor-producing substances transfer into breast milk, not many have explored specific substances that give food its distinctive flavor, or even what makes up the taste of breast milk. So they decided to investigate what happens when a mother consumes a meal containing three specific compounds: those that give pepper, chili, and ginger their particularly pungent flavors.
The researchers recruited 18 healthy, nonsmoking, nursing mothers who were producing more than enough milk for their baby’s needs. Their breastfeeding children ranged in age from 8 weeks to 1 year old. The women all ate a curry dish after having spent 2 days avoiding onion, garlic, and the spices in the curry. Then they provided pumped breast milk samples at 1, 2, and 3 hours after eating the curry.
Within an hour of the women eating the curry, the scientists were able to detect piperine, the compound that gives black pepper its bite, in the mothers’ breast milk. They did not find the compounds from ginger, chili, or curcumin – the main active ingredient in turmeric – in the breast milk. The piperine remained there for several hours, but there wasn’t enough for an adult to be able to taste it. It wasn’t possible to reliably tell whether the infants could consciously detect the flavor, but the researchers don’t think it’s likely they did.
But the scientists do suggest it’s possible that the piperine in breast milk could regularly activate a protein that detects pungent or potentially harmful substances. This is the same protein that produces the sensation of heat when eating a spicy food. If the piperine frequently activates that protein in a nursing baby at levels too low for the baby to notice, it may increase the baby’s tolerance for similar spicy substances later in life.
Ultimately, the findings suggest that .
A version of this story first appeared on WebMD.com.
They say you are what you eat, but scientists have long wondered whether breastfeeding babies are what their mothers eat, too. Their question: How much of a nursing mother’s diet eventually plays a role in a child’s food preferences later in life?
The aroma, taste, and makeup of breast milk change from day to day, based mostly on the mother’s diet. But previous research has already shown that the foods a mother eats do not directly translate into the same smells and tastes of that food in breast milk. Some substances from the mother’s diet enter her breast milk, some don’t, and even ones that do may have a different scent or flavor than what the mother experiences.
But a new study suggests that the active ingredient in black pepper makes its way into breast milk and may help the infant develop a tolerance to pepper later. The researchers published their findings in the journal Molecular Nutrition & Food Research.
Pinch of pepper
The study authors thought that maybe some food preferences could result from sensory programming that occurs through breast milk in the first few months of life. Though past studies have looked at which odor-producing substances transfer into breast milk, not many have explored specific substances that give food its distinctive flavor, or even what makes up the taste of breast milk. So they decided to investigate what happens when a mother consumes a meal containing three specific compounds: those that give pepper, chili, and ginger their particularly pungent flavors.
The researchers recruited 18 healthy, nonsmoking, nursing mothers who were producing more than enough milk for their baby’s needs. Their breastfeeding children ranged in age from 8 weeks to 1 year old. The women all ate a curry dish after having spent 2 days avoiding onion, garlic, and the spices in the curry. Then they provided pumped breast milk samples at 1, 2, and 3 hours after eating the curry.
Within an hour of the women eating the curry, the scientists were able to detect piperine, the compound that gives black pepper its bite, in the mothers’ breast milk. They did not find the compounds from ginger, chili, or curcumin – the main active ingredient in turmeric – in the breast milk. The piperine remained there for several hours, but there wasn’t enough for an adult to be able to taste it. It wasn’t possible to reliably tell whether the infants could consciously detect the flavor, but the researchers don’t think it’s likely they did.
But the scientists do suggest it’s possible that the piperine in breast milk could regularly activate a protein that detects pungent or potentially harmful substances. This is the same protein that produces the sensation of heat when eating a spicy food. If the piperine frequently activates that protein in a nursing baby at levels too low for the baby to notice, it may increase the baby’s tolerance for similar spicy substances later in life.
Ultimately, the findings suggest that .
A version of this story first appeared on WebMD.com.
FROM MOLECULAR NUTRITION & FOOD RESEARCH
Children and COVID: Weekly cases resume their climb
After a brief lull in activity, weekly COVID-19 cases in children returned to the upward trend that began in early November, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
according to the Centers for Disease Control and Prevention.
New COVID-19 cases were up by 23.5% for the week of Dec. 3-9, after a 2-week period that saw a drop and then just a slight increase, the AAP and CHA said in their latest weekly COVID report. There were 164,000 new cases from Dec. 3 to Dec. 9 in 46 states (Alabama, Nebraska, and Texas stopped reporting over the summer of 2021 and New York has never reported by age), the District of Columbia, New York City, Puerto Rico, and Guam.
The increase occurred across all four regions of the country, but the largest share came in the Midwest, with over 65,000 new cases, followed by the West (just over 35,000), the Northeast (just under 35,000), and the South (close to 28,000), the AAP/CHA data show.
The 7.2 million cumulative cases in children as of Dec. 9 represent 17.2% of all cases reported in the United States since the start of the pandemic, with available state reports showing that proportion ranges from 12.3% in Florida to 26.1% in Vermont. Alaska has the highest incidence of COVID at 19,000 cases per 100,000 children, and Hawaii has the lowest (5,300 per 100,000) among the states currently reporting, the AAP and CHA said.
State reporting on vaccinations shows that 37% of children aged 5-11 years in Massachusetts have received at least one dose, the highest of any state, while West Virginia is lowest at just 4%. The highest vaccination rate for children aged 12-17 goes to Massachusetts at 84%, with Wyoming lowest at 37%, the AAP said in a separate report.
Nationally, new vaccinations fell by a third during the week of Dec. 7-13, compared with the previous week, with the largest decline (34.7%) coming from the 5- to 11-year-olds, who still represented the majority (almost 84%) of the 430,000 new child vaccinations received, according to the CDC’s COVID Data Tracker. Corresponding declines for the last week were 27.5% for 12- to 15-year-olds and 22.7% for those aged 16-17.
Altogether, 21.2 million children aged 5-17 had received at least one dose and 16.0 million were fully vaccinated as of Dec. 13. By age group, 19.2% of children aged 5-11 years have gotten at least one dose and 9.6% are fully vaccinated, compared with 62.1% and 52.3%, respectively, among children aged 12-17, the CDC said.
After a brief lull in activity, weekly COVID-19 cases in children returned to the upward trend that began in early November, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.

according to the Centers for Disease Control and Prevention.
New COVID-19 cases were up by 23.5% for the week of Dec. 3-9, after a 2-week period that saw a drop and then just a slight increase, the AAP and CHA said in their latest weekly COVID report. There were 164,000 new cases from Dec. 3 to Dec. 9 in 46 states (Alabama, Nebraska, and Texas stopped reporting over the summer of 2021 and New York has never reported by age), the District of Columbia, New York City, Puerto Rico, and Guam.
The increase occurred across all four regions of the country, but the largest share came in the Midwest, with over 65,000 new cases, followed by the West (just over 35,000), the Northeast (just under 35,000), and the South (close to 28,000), the AAP/CHA data show.
The 7.2 million cumulative cases in children as of Dec. 9 represent 17.2% of all cases reported in the United States since the start of the pandemic, with available state reports showing that proportion ranges from 12.3% in Florida to 26.1% in Vermont. Alaska has the highest incidence of COVID at 19,000 cases per 100,000 children, and Hawaii has the lowest (5,300 per 100,000) among the states currently reporting, the AAP and CHA said.
State reporting on vaccinations shows that 37% of children aged 5-11 years in Massachusetts have received at least one dose, the highest of any state, while West Virginia is lowest at just 4%. The highest vaccination rate for children aged 12-17 goes to Massachusetts at 84%, with Wyoming lowest at 37%, the AAP said in a separate report.
Nationally, new vaccinations fell by a third during the week of Dec. 7-13, compared with the previous week, with the largest decline (34.7%) coming from the 5- to 11-year-olds, who still represented the majority (almost 84%) of the 430,000 new child vaccinations received, according to the CDC’s COVID Data Tracker. Corresponding declines for the last week were 27.5% for 12- to 15-year-olds and 22.7% for those aged 16-17.
Altogether, 21.2 million children aged 5-17 had received at least one dose and 16.0 million were fully vaccinated as of Dec. 13. By age group, 19.2% of children aged 5-11 years have gotten at least one dose and 9.6% are fully vaccinated, compared with 62.1% and 52.3%, respectively, among children aged 12-17, the CDC said.
After a brief lull in activity, weekly COVID-19 cases in children returned to the upward trend that began in early November, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.

according to the Centers for Disease Control and Prevention.
New COVID-19 cases were up by 23.5% for the week of Dec. 3-9, after a 2-week period that saw a drop and then just a slight increase, the AAP and CHA said in their latest weekly COVID report. There were 164,000 new cases from Dec. 3 to Dec. 9 in 46 states (Alabama, Nebraska, and Texas stopped reporting over the summer of 2021 and New York has never reported by age), the District of Columbia, New York City, Puerto Rico, and Guam.
The increase occurred across all four regions of the country, but the largest share came in the Midwest, with over 65,000 new cases, followed by the West (just over 35,000), the Northeast (just under 35,000), and the South (close to 28,000), the AAP/CHA data show.
The 7.2 million cumulative cases in children as of Dec. 9 represent 17.2% of all cases reported in the United States since the start of the pandemic, with available state reports showing that proportion ranges from 12.3% in Florida to 26.1% in Vermont. Alaska has the highest incidence of COVID at 19,000 cases per 100,000 children, and Hawaii has the lowest (5,300 per 100,000) among the states currently reporting, the AAP and CHA said.
State reporting on vaccinations shows that 37% of children aged 5-11 years in Massachusetts have received at least one dose, the highest of any state, while West Virginia is lowest at just 4%. The highest vaccination rate for children aged 12-17 goes to Massachusetts at 84%, with Wyoming lowest at 37%, the AAP said in a separate report.
Nationally, new vaccinations fell by a third during the week of Dec. 7-13, compared with the previous week, with the largest decline (34.7%) coming from the 5- to 11-year-olds, who still represented the majority (almost 84%) of the 430,000 new child vaccinations received, according to the CDC’s COVID Data Tracker. Corresponding declines for the last week were 27.5% for 12- to 15-year-olds and 22.7% for those aged 16-17.
Altogether, 21.2 million children aged 5-17 had received at least one dose and 16.0 million were fully vaccinated as of Dec. 13. By age group, 19.2% of children aged 5-11 years have gotten at least one dose and 9.6% are fully vaccinated, compared with 62.1% and 52.3%, respectively, among children aged 12-17, the CDC said.