User login
RSV resurgence likely in wake of COVID-19
The impact of respiratory syncytial virus (RSV)will likely be greater in 2021 and 2022 in the United States than in previous years as a result of the ongoing COVID-19 pandemic, based on data from a simulation-modeling study involving approximately 19 million individuals.
Although RSV usually follows consistent patterns of timing and duration, the disease all but disappeared starting in March 2020 after the introduction of measures to mitigate the spread of COVID-19, Zhe Zheng, MBBS, of Yale University, New Haven, Conn., and colleagues wrote.
However, lifting of mitigation measures has resulted in emergence of RSV in various parts of the world in early 2021, and trends may be similar in the United States, but data are needed to plan for prophylaxis and hospital use, they noted.
In a study published in JAMA Network Open, the researchers developed a simulation model for epidemics of RSV based on historical data. They acquired inpatient records from New York during 2005-2014 and from California during 2003-2011. The primary clinical outcome was the estimated monthly hospitalizations for RSV.
The simulated study population was 19.45 million individuals. After evaluating several scenarios including continued low transmission associated with social distancing and other mitigation measures, the researchers focused on the likely scenario that introduction of RSV from other regions would likely spark RSV epidemics in the United States.
They determined that spring and summer 2021 would show an increase in hospitalizations for RSV. Overall, higher rates of virus introduction from other regions were associated with more intense spring and summer RSV epidemics, with the trade-off of smaller winter epidemics. In the model, the expected RSV epidemic in spring and summer 2021 in New York was small, with a peak incidence of 419 hospitalizations per 100,000 people in April; by contrast, for states with less seasonal variability, such as Florida, the model predicted a larger summer epidemic.
In the model, the mean age of hospitalization for children younger than 5 years for January 2022 was expected to be 1.17 years, compared with 0.84 years in January 2019, the researchers noted.
Across all age groups, the greatest relative increase in the incidence of RSV infection was predicted for children aged 1-4 years (ranging from 82% to 86%), as were lower respiratory infections (87%-101%) and hospitalization (99%-119%), compared with prepandemic levels.
Hospitalizations for children aged 1 year were predicted to double compared with prepandemic seasons; 707 per 100,000 children per year for 2021 and 2022 versus 355 per 100,000 children per year in a typical prepandemic season. However, the largest incidence of lower respiratory infections (30,075 per 100,000) was predicted for infants aged 3-5 months, and the largest incidence of hospitalizations (3,116 per 100,000) was predicted for infants younger than 3 months.
“Without virus importation, the risk of RSV infections across all age groups in the winter of 2021 and 2022 would be greater, as more susceptible individuals were spared from infections in the absence of summer epidemics,” the researchers noted.
The older mean hospitalization age seen in the model was similar to the reported median patient age in Australia both before the pandemic and during the reemergent RSV epidemic.
“This makes intuitive sense, since many children born in 2020 were spared from RSV infection due to the low virus activity; these children will be older when they get infected for the first time during the reemergent epidemics,” the researchers wrote. “Consequently, stakeholders should consider modifying prophylaxis guidelines to include high-risk infants less than 2 years of age for the 2021-2022 season.”
The study findings were limited by several factors including the lack of data on level of virus introduction or on the impact of lack of boosting on infants with only transplacentally acquired RSV antibodies, the researchers noted. Other limitations include the use of historical data and the lack of data on values outside those included in the model, as well as the inability to control for other factors that could influence RSV, such as vaccines or long-lasting antibodies.
However, the results suggest that the rate of imported infections is associated with RSV hospitalizations, and the model effectively captured the RSV epidemics in the United States in spring and summer 2021.
Models can guide clinical preparations
“Health care simulation modeling is a growing field, with very exciting implications,” Lenore Jarvis, MD, of George Washington University, Washington, said in an interview. The field has the potential ability to influence health care in a data-driven way, including, but not limited to, staffing and other hospital operations, as well as patient care decision-making. “In short, accurate modeling and predictions can help us to make informed health care decisions that can lead to increased quality of care, potential cost savings, and even to help save lives,” she said.
Although the details of transmission modeling were not mentioned in the study, the authors evaluated the performances of several models and scenarios. “Scenario 4, for example, was focused on in particular because it best captured the observed dynamics [for RSV] that emerged during the spring and summer of 2021,” Dr. Jarvis said.
“Pediatricians can speak to these trends firsthand. A decrease in expected RSV infections and hospitalizations in 2020, followed by an unprecedented and early increase in RSV infections and severity in 2021, and the factors that the authors account for make sense, such as reintroduction of RSV from other regions and low immunity in the population,” she said. “It also makes sense that, in these transmission modeling scenarios, the expected mean age of hospitalization because of RSV increased with a temporary (hopefully) increase in RSV hospitalizations in the 2021 season, and potentially the 2022 RSV season.”
As for additional research, Dr. Jarvis said she would like to see follow-up data on the RSV transmission modeling. “For example, with scenario 4, does this scenario continue to perform well in other time periods, such as the winter? If the modeling continues to be accurate during other periods of evaluation and reevaluation, this modeling could be very useful in helping pediatric clinics and hospitals to prepare for RSV care and hospital capacity management.”
The study was supported by grants to various researchers from the National Institute of Allergy and Infectious Diseases/National Institutes of Health, the National Center for Advancing Translational Science at the National Institutes of Health, and NIH Roadmap for Medical Research. Lead author Ms. Zheng had no financial conflicts to disclose. Her study coauthors disclosed relationships with companies including AbbVie, Merck, Pfizer, GlaxoSmithKline, MedImmune, and Janssen. Dr. Jarvis had no financial conflicts to disclose and serves on the Pediatric News editorial advisory board.
The impact of respiratory syncytial virus (RSV)will likely be greater in 2021 and 2022 in the United States than in previous years as a result of the ongoing COVID-19 pandemic, based on data from a simulation-modeling study involving approximately 19 million individuals.
Although RSV usually follows consistent patterns of timing and duration, the disease all but disappeared starting in March 2020 after the introduction of measures to mitigate the spread of COVID-19, Zhe Zheng, MBBS, of Yale University, New Haven, Conn., and colleagues wrote.
However, lifting of mitigation measures has resulted in emergence of RSV in various parts of the world in early 2021, and trends may be similar in the United States, but data are needed to plan for prophylaxis and hospital use, they noted.
In a study published in JAMA Network Open, the researchers developed a simulation model for epidemics of RSV based on historical data. They acquired inpatient records from New York during 2005-2014 and from California during 2003-2011. The primary clinical outcome was the estimated monthly hospitalizations for RSV.
The simulated study population was 19.45 million individuals. After evaluating several scenarios including continued low transmission associated with social distancing and other mitigation measures, the researchers focused on the likely scenario that introduction of RSV from other regions would likely spark RSV epidemics in the United States.
They determined that spring and summer 2021 would show an increase in hospitalizations for RSV. Overall, higher rates of virus introduction from other regions were associated with more intense spring and summer RSV epidemics, with the trade-off of smaller winter epidemics. In the model, the expected RSV epidemic in spring and summer 2021 in New York was small, with a peak incidence of 419 hospitalizations per 100,000 people in April; by contrast, for states with less seasonal variability, such as Florida, the model predicted a larger summer epidemic.
In the model, the mean age of hospitalization for children younger than 5 years for January 2022 was expected to be 1.17 years, compared with 0.84 years in January 2019, the researchers noted.
Across all age groups, the greatest relative increase in the incidence of RSV infection was predicted for children aged 1-4 years (ranging from 82% to 86%), as were lower respiratory infections (87%-101%) and hospitalization (99%-119%), compared with prepandemic levels.
Hospitalizations for children aged 1 year were predicted to double compared with prepandemic seasons; 707 per 100,000 children per year for 2021 and 2022 versus 355 per 100,000 children per year in a typical prepandemic season. However, the largest incidence of lower respiratory infections (30,075 per 100,000) was predicted for infants aged 3-5 months, and the largest incidence of hospitalizations (3,116 per 100,000) was predicted for infants younger than 3 months.
“Without virus importation, the risk of RSV infections across all age groups in the winter of 2021 and 2022 would be greater, as more susceptible individuals were spared from infections in the absence of summer epidemics,” the researchers noted.
The older mean hospitalization age seen in the model was similar to the reported median patient age in Australia both before the pandemic and during the reemergent RSV epidemic.
“This makes intuitive sense, since many children born in 2020 were spared from RSV infection due to the low virus activity; these children will be older when they get infected for the first time during the reemergent epidemics,” the researchers wrote. “Consequently, stakeholders should consider modifying prophylaxis guidelines to include high-risk infants less than 2 years of age for the 2021-2022 season.”
The study findings were limited by several factors including the lack of data on level of virus introduction or on the impact of lack of boosting on infants with only transplacentally acquired RSV antibodies, the researchers noted. Other limitations include the use of historical data and the lack of data on values outside those included in the model, as well as the inability to control for other factors that could influence RSV, such as vaccines or long-lasting antibodies.
However, the results suggest that the rate of imported infections is associated with RSV hospitalizations, and the model effectively captured the RSV epidemics in the United States in spring and summer 2021.
Models can guide clinical preparations
“Health care simulation modeling is a growing field, with very exciting implications,” Lenore Jarvis, MD, of George Washington University, Washington, said in an interview. The field has the potential ability to influence health care in a data-driven way, including, but not limited to, staffing and other hospital operations, as well as patient care decision-making. “In short, accurate modeling and predictions can help us to make informed health care decisions that can lead to increased quality of care, potential cost savings, and even to help save lives,” she said.
Although the details of transmission modeling were not mentioned in the study, the authors evaluated the performances of several models and scenarios. “Scenario 4, for example, was focused on in particular because it best captured the observed dynamics [for RSV] that emerged during the spring and summer of 2021,” Dr. Jarvis said.
“Pediatricians can speak to these trends firsthand. A decrease in expected RSV infections and hospitalizations in 2020, followed by an unprecedented and early increase in RSV infections and severity in 2021, and the factors that the authors account for make sense, such as reintroduction of RSV from other regions and low immunity in the population,” she said. “It also makes sense that, in these transmission modeling scenarios, the expected mean age of hospitalization because of RSV increased with a temporary (hopefully) increase in RSV hospitalizations in the 2021 season, and potentially the 2022 RSV season.”
As for additional research, Dr. Jarvis said she would like to see follow-up data on the RSV transmission modeling. “For example, with scenario 4, does this scenario continue to perform well in other time periods, such as the winter? If the modeling continues to be accurate during other periods of evaluation and reevaluation, this modeling could be very useful in helping pediatric clinics and hospitals to prepare for RSV care and hospital capacity management.”
The study was supported by grants to various researchers from the National Institute of Allergy and Infectious Diseases/National Institutes of Health, the National Center for Advancing Translational Science at the National Institutes of Health, and NIH Roadmap for Medical Research. Lead author Ms. Zheng had no financial conflicts to disclose. Her study coauthors disclosed relationships with companies including AbbVie, Merck, Pfizer, GlaxoSmithKline, MedImmune, and Janssen. Dr. Jarvis had no financial conflicts to disclose and serves on the Pediatric News editorial advisory board.
The impact of respiratory syncytial virus (RSV)will likely be greater in 2021 and 2022 in the United States than in previous years as a result of the ongoing COVID-19 pandemic, based on data from a simulation-modeling study involving approximately 19 million individuals.
Although RSV usually follows consistent patterns of timing and duration, the disease all but disappeared starting in March 2020 after the introduction of measures to mitigate the spread of COVID-19, Zhe Zheng, MBBS, of Yale University, New Haven, Conn., and colleagues wrote.
However, lifting of mitigation measures has resulted in emergence of RSV in various parts of the world in early 2021, and trends may be similar in the United States, but data are needed to plan for prophylaxis and hospital use, they noted.
In a study published in JAMA Network Open, the researchers developed a simulation model for epidemics of RSV based on historical data. They acquired inpatient records from New York during 2005-2014 and from California during 2003-2011. The primary clinical outcome was the estimated monthly hospitalizations for RSV.
The simulated study population was 19.45 million individuals. After evaluating several scenarios including continued low transmission associated with social distancing and other mitigation measures, the researchers focused on the likely scenario that introduction of RSV from other regions would likely spark RSV epidemics in the United States.
They determined that spring and summer 2021 would show an increase in hospitalizations for RSV. Overall, higher rates of virus introduction from other regions were associated with more intense spring and summer RSV epidemics, with the trade-off of smaller winter epidemics. In the model, the expected RSV epidemic in spring and summer 2021 in New York was small, with a peak incidence of 419 hospitalizations per 100,000 people in April; by contrast, for states with less seasonal variability, such as Florida, the model predicted a larger summer epidemic.
In the model, the mean age of hospitalization for children younger than 5 years for January 2022 was expected to be 1.17 years, compared with 0.84 years in January 2019, the researchers noted.
Across all age groups, the greatest relative increase in the incidence of RSV infection was predicted for children aged 1-4 years (ranging from 82% to 86%), as were lower respiratory infections (87%-101%) and hospitalization (99%-119%), compared with prepandemic levels.
Hospitalizations for children aged 1 year were predicted to double compared with prepandemic seasons; 707 per 100,000 children per year for 2021 and 2022 versus 355 per 100,000 children per year in a typical prepandemic season. However, the largest incidence of lower respiratory infections (30,075 per 100,000) was predicted for infants aged 3-5 months, and the largest incidence of hospitalizations (3,116 per 100,000) was predicted for infants younger than 3 months.
“Without virus importation, the risk of RSV infections across all age groups in the winter of 2021 and 2022 would be greater, as more susceptible individuals were spared from infections in the absence of summer epidemics,” the researchers noted.
The older mean hospitalization age seen in the model was similar to the reported median patient age in Australia both before the pandemic and during the reemergent RSV epidemic.
“This makes intuitive sense, since many children born in 2020 were spared from RSV infection due to the low virus activity; these children will be older when they get infected for the first time during the reemergent epidemics,” the researchers wrote. “Consequently, stakeholders should consider modifying prophylaxis guidelines to include high-risk infants less than 2 years of age for the 2021-2022 season.”
The study findings were limited by several factors including the lack of data on level of virus introduction or on the impact of lack of boosting on infants with only transplacentally acquired RSV antibodies, the researchers noted. Other limitations include the use of historical data and the lack of data on values outside those included in the model, as well as the inability to control for other factors that could influence RSV, such as vaccines or long-lasting antibodies.
However, the results suggest that the rate of imported infections is associated with RSV hospitalizations, and the model effectively captured the RSV epidemics in the United States in spring and summer 2021.
Models can guide clinical preparations
“Health care simulation modeling is a growing field, with very exciting implications,” Lenore Jarvis, MD, of George Washington University, Washington, said in an interview. The field has the potential ability to influence health care in a data-driven way, including, but not limited to, staffing and other hospital operations, as well as patient care decision-making. “In short, accurate modeling and predictions can help us to make informed health care decisions that can lead to increased quality of care, potential cost savings, and even to help save lives,” she said.
Although the details of transmission modeling were not mentioned in the study, the authors evaluated the performances of several models and scenarios. “Scenario 4, for example, was focused on in particular because it best captured the observed dynamics [for RSV] that emerged during the spring and summer of 2021,” Dr. Jarvis said.
“Pediatricians can speak to these trends firsthand. A decrease in expected RSV infections and hospitalizations in 2020, followed by an unprecedented and early increase in RSV infections and severity in 2021, and the factors that the authors account for make sense, such as reintroduction of RSV from other regions and low immunity in the population,” she said. “It also makes sense that, in these transmission modeling scenarios, the expected mean age of hospitalization because of RSV increased with a temporary (hopefully) increase in RSV hospitalizations in the 2021 season, and potentially the 2022 RSV season.”
As for additional research, Dr. Jarvis said she would like to see follow-up data on the RSV transmission modeling. “For example, with scenario 4, does this scenario continue to perform well in other time periods, such as the winter? If the modeling continues to be accurate during other periods of evaluation and reevaluation, this modeling could be very useful in helping pediatric clinics and hospitals to prepare for RSV care and hospital capacity management.”
The study was supported by grants to various researchers from the National Institute of Allergy and Infectious Diseases/National Institutes of Health, the National Center for Advancing Translational Science at the National Institutes of Health, and NIH Roadmap for Medical Research. Lead author Ms. Zheng had no financial conflicts to disclose. Her study coauthors disclosed relationships with companies including AbbVie, Merck, Pfizer, GlaxoSmithKline, MedImmune, and Janssen. Dr. Jarvis had no financial conflicts to disclose and serves on the Pediatric News editorial advisory board.
FROM JAMA NETWORK OPEN
Visceral fat may help ID heart risk in obese youth
The amount of fat surrounding abdominal organs may help clinicians identify cardiovascular risk in young people with obesity, researchers have found.
Severely overweight children and young adults showed a subtle association between visceral fat and arterial stiffness independent of body mass index (BMI). The association was not present in those of healthy weight, possibly because their visceral fat stores are too small to have a detectable effect on cardiovascular health, according to the researchers, who reported their findings in the latest issue of Pediatric Obesity.
“Those kids with greater visceral fat had stiffer arteries, which can overtax and overstress the system and lead to unfortunate consequences in terms of cardiovascular health down the line,” senior author Joseph M. Kindler, PhD, an assistant professor of nutritional sciences at the University of Georgia, Athens, told this news organization.
The data came from cross-sectional measurements in 605 youth (67% female, 56% non-Black) aged 10-23 years at Cincinnati Children’s Hospital Medical Center. The sample included 236 individuals of healthy weight, 224 with obesity, and 145 with type 2 diabetes.
Visceral fat was assessed with dual-energy x-ray absorptiometry (DXA), a widely used test of bone mineral density screening to assess fracture risk. Carotid-femoral pulse wave velocity (PWV) was used to gauge arterial stiffness, a subclinical sign of cardiovascular disease.
Visceral fat was associated with PWV in all three groups of study subjects (P < .05), the researchers found, whereas the amount of subcutaneous fat was linked to arterial stiffness in obese youth and those with obesity but not those whose weight was considered healthy.
The amount of fat was associated with an additional 1.6% of the variability in arterial stiffness in youth with obesity after accounting for BMI. Subcutaneous fat, meanwhile, did not appear to affect PWV, the researchers found. “In youth with healthy weight, visceral fat, subcutaneous fat, BMI, and waist circumference were not significantly associated with PWV in any analyses,” they write.
The researchers cited a paucity of data on the relationship between visceral fat and cardiovascular disease in children with obesity. Although BMI is a reliable and readily available indicator of risk for disease, DXA “might give us a little more information,” Dr. Kindler, a nutritionist and bone biologist, said. As for clinical use to supplement BMI and waist circumference, he said, “maybe there’s room for visceral fat, but we do need a lot more science to back those decisions down the line.”
For example, what normal visceral fat accumulation during childhood looks like is unknown, he said.
Rigorous longitudinal studies are needed to establish cause and effect, but the new findings offer “a potential connection between visceral fat and cardiovascular disease risk in youth in a relatively large sample,” Wei Shen, MD, MPH, the associate director of the body composition unit at the New York Obesity Nutrition Research Center at Columbia University, New York, said.
Ideally, said Dr. Shen, who was not involved in the latest study, it would be “more credible to use the most accurate measure of visceral fat, the volumetric measurement of visceral fat using MRI” to establish a causal relationship with cardiovascular risk. However, MRI is more expensive and less accessible than DXA. To assess visceral fat in the clinic, “waist circumference may still be a good choice, as it is so convenient to use,” she added.
Dr. Kindler and his colleagues highlighted the need to examine the effect of excess visceral fat as well as intrahepatic fat on youth with type 2 diabetes, who experience cardiovascular complications independent of whether they are obese. In the new study, the positive association between visceral fat and arterial stiffness did not differ between youth with obesity and normal glucose control and those with obesity and type 2 diabetes.
Funding came from the Endocrine Fellows Foundation, the National Institutes of Health, and the University of Georgia Obesity Initiative. Dr. Kindler and Dr. Shen have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The amount of fat surrounding abdominal organs may help clinicians identify cardiovascular risk in young people with obesity, researchers have found.
Severely overweight children and young adults showed a subtle association between visceral fat and arterial stiffness independent of body mass index (BMI). The association was not present in those of healthy weight, possibly because their visceral fat stores are too small to have a detectable effect on cardiovascular health, according to the researchers, who reported their findings in the latest issue of Pediatric Obesity.
“Those kids with greater visceral fat had stiffer arteries, which can overtax and overstress the system and lead to unfortunate consequences in terms of cardiovascular health down the line,” senior author Joseph M. Kindler, PhD, an assistant professor of nutritional sciences at the University of Georgia, Athens, told this news organization.
The data came from cross-sectional measurements in 605 youth (67% female, 56% non-Black) aged 10-23 years at Cincinnati Children’s Hospital Medical Center. The sample included 236 individuals of healthy weight, 224 with obesity, and 145 with type 2 diabetes.
Visceral fat was assessed with dual-energy x-ray absorptiometry (DXA), a widely used test of bone mineral density screening to assess fracture risk. Carotid-femoral pulse wave velocity (PWV) was used to gauge arterial stiffness, a subclinical sign of cardiovascular disease.
Visceral fat was associated with PWV in all three groups of study subjects (P < .05), the researchers found, whereas the amount of subcutaneous fat was linked to arterial stiffness in obese youth and those with obesity but not those whose weight was considered healthy.
The amount of fat was associated with an additional 1.6% of the variability in arterial stiffness in youth with obesity after accounting for BMI. Subcutaneous fat, meanwhile, did not appear to affect PWV, the researchers found. “In youth with healthy weight, visceral fat, subcutaneous fat, BMI, and waist circumference were not significantly associated with PWV in any analyses,” they write.
The researchers cited a paucity of data on the relationship between visceral fat and cardiovascular disease in children with obesity. Although BMI is a reliable and readily available indicator of risk for disease, DXA “might give us a little more information,” Dr. Kindler, a nutritionist and bone biologist, said. As for clinical use to supplement BMI and waist circumference, he said, “maybe there’s room for visceral fat, but we do need a lot more science to back those decisions down the line.”
For example, what normal visceral fat accumulation during childhood looks like is unknown, he said.
Rigorous longitudinal studies are needed to establish cause and effect, but the new findings offer “a potential connection between visceral fat and cardiovascular disease risk in youth in a relatively large sample,” Wei Shen, MD, MPH, the associate director of the body composition unit at the New York Obesity Nutrition Research Center at Columbia University, New York, said.
Ideally, said Dr. Shen, who was not involved in the latest study, it would be “more credible to use the most accurate measure of visceral fat, the volumetric measurement of visceral fat using MRI” to establish a causal relationship with cardiovascular risk. However, MRI is more expensive and less accessible than DXA. To assess visceral fat in the clinic, “waist circumference may still be a good choice, as it is so convenient to use,” she added.
Dr. Kindler and his colleagues highlighted the need to examine the effect of excess visceral fat as well as intrahepatic fat on youth with type 2 diabetes, who experience cardiovascular complications independent of whether they are obese. In the new study, the positive association between visceral fat and arterial stiffness did not differ between youth with obesity and normal glucose control and those with obesity and type 2 diabetes.
Funding came from the Endocrine Fellows Foundation, the National Institutes of Health, and the University of Georgia Obesity Initiative. Dr. Kindler and Dr. Shen have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The amount of fat surrounding abdominal organs may help clinicians identify cardiovascular risk in young people with obesity, researchers have found.
Severely overweight children and young adults showed a subtle association between visceral fat and arterial stiffness independent of body mass index (BMI). The association was not present in those of healthy weight, possibly because their visceral fat stores are too small to have a detectable effect on cardiovascular health, according to the researchers, who reported their findings in the latest issue of Pediatric Obesity.
“Those kids with greater visceral fat had stiffer arteries, which can overtax and overstress the system and lead to unfortunate consequences in terms of cardiovascular health down the line,” senior author Joseph M. Kindler, PhD, an assistant professor of nutritional sciences at the University of Georgia, Athens, told this news organization.
The data came from cross-sectional measurements in 605 youth (67% female, 56% non-Black) aged 10-23 years at Cincinnati Children’s Hospital Medical Center. The sample included 236 individuals of healthy weight, 224 with obesity, and 145 with type 2 diabetes.
Visceral fat was assessed with dual-energy x-ray absorptiometry (DXA), a widely used test of bone mineral density screening to assess fracture risk. Carotid-femoral pulse wave velocity (PWV) was used to gauge arterial stiffness, a subclinical sign of cardiovascular disease.
Visceral fat was associated with PWV in all three groups of study subjects (P < .05), the researchers found, whereas the amount of subcutaneous fat was linked to arterial stiffness in obese youth and those with obesity but not those whose weight was considered healthy.
The amount of fat was associated with an additional 1.6% of the variability in arterial stiffness in youth with obesity after accounting for BMI. Subcutaneous fat, meanwhile, did not appear to affect PWV, the researchers found. “In youth with healthy weight, visceral fat, subcutaneous fat, BMI, and waist circumference were not significantly associated with PWV in any analyses,” they write.
The researchers cited a paucity of data on the relationship between visceral fat and cardiovascular disease in children with obesity. Although BMI is a reliable and readily available indicator of risk for disease, DXA “might give us a little more information,” Dr. Kindler, a nutritionist and bone biologist, said. As for clinical use to supplement BMI and waist circumference, he said, “maybe there’s room for visceral fat, but we do need a lot more science to back those decisions down the line.”
For example, what normal visceral fat accumulation during childhood looks like is unknown, he said.
Rigorous longitudinal studies are needed to establish cause and effect, but the new findings offer “a potential connection between visceral fat and cardiovascular disease risk in youth in a relatively large sample,” Wei Shen, MD, MPH, the associate director of the body composition unit at the New York Obesity Nutrition Research Center at Columbia University, New York, said.
Ideally, said Dr. Shen, who was not involved in the latest study, it would be “more credible to use the most accurate measure of visceral fat, the volumetric measurement of visceral fat using MRI” to establish a causal relationship with cardiovascular risk. However, MRI is more expensive and less accessible than DXA. To assess visceral fat in the clinic, “waist circumference may still be a good choice, as it is so convenient to use,” she added.
Dr. Kindler and his colleagues highlighted the need to examine the effect of excess visceral fat as well as intrahepatic fat on youth with type 2 diabetes, who experience cardiovascular complications independent of whether they are obese. In the new study, the positive association between visceral fat and arterial stiffness did not differ between youth with obesity and normal glucose control and those with obesity and type 2 diabetes.
Funding came from the Endocrine Fellows Foundation, the National Institutes of Health, and the University of Georgia Obesity Initiative. Dr. Kindler and Dr. Shen have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AAP updates guidance on HIV testing and prophylaxis in youth
Pediatricians should take a more proactive role in protecting children and adolescents from HIV infections, according to updated guidance from the American Academy of Pediatrics. The comprehensive new recommendations stress winning the trust and confidence of pediatric patients and reaffirm support for testing and treating adolescents without parental consent where state laws allow.
While the number of HIV-infected people in the United States remains high, most sexually active youth do not believe they are at risk and have never been tested, noted authors Katherine K. Hsu, MD, MPH, of the Massachusetts Department of Public Health and Boston University Medical Center, and Natella Yurievna Rakhmanina, MD, PhD, of Children’s National Hospital and George Washington University, both in Washington.
That is a knowledge gap that pediatricians are well situated to fill. “Pediatricians can play a key role in preventing and controlling HIV infection by promoting risk-reduction counseling and offering routine HIV testing and prophylaxis to adolescent and young adult (youth) patients,” they wrote on Dec. 20, 2021, in their study published in Pediatrics.
Key components of youth encounters, they stressed, is creating safe environments for obtaining an accurate sexual and reproductive health assessment and providing nonstigmatizing risk counseling.
According to Dr. Rakhmanina, major barriers to addressing preventive HIV counseling have included pediatricians’ lack of time, cultural differences, adolescents’ inaccurate responses, discomfort discussing sexual issues, and adolescents’ fear of parent or caregiver notification. Other concerns have been lack of adequate payment and insufficient training in how to talk to adolescents about sexual and reproductive issues.
According to the Centers for Disease Control and Prevention, at year end in 2018 an estimated 1,173,900 people age 13 or older were living with HIV infection in the United States, of whom 47,800 (4%) were adolescents and young adults 13-24 years of age.
These estimates include diagnosed and undiagnosed individuals. Between 2014 and 2018, new diagnoses of HIV infection accounted for 21% (7,817 of 37,515) of all new HIV diagnoses in the United States.
The new AAP clinical report updates policy statements from 2001 and again 2011 that encouraged HIV testing of all sexually active youth.
It reflects changes in epidemiology, advances in diagnostic testing with improved immunoassays, and updated recommendations for HIV testing and postexposure prophylaxis (PEP), as well as new guidance for pre-exposure prophylaxis (PrEP).
A 2017 study found that the 2011 HIV testing guidelines was associated with only a slight increase in HIV screening and a shift toward testing younger people and away from testing on the basis of risk.
Against this backdrop of persistent HIV infection and to-date modest uptake of earlier guidance, the 2021 statement made 14 main recommendations to pediatricians. Among these:
- Foster open discussion of gender and sexual orientation and behavior, as well as reproductive health issues.
- Recognize the clinical presentation of the acute retroviral syndrome, which can present as syndromes resembling infectious mononucleosis and influenza.
- Consider including virologic testing in the diagnostic workup of sexually active youth.
- Consider routine HIV screening for all youth 15 years or older at least once and rescreening high-risk youth. Those at higher risk should be rescreened at least annually, and potentially as frequently as every 3-6 months.
- Youth at substantial risk should be routinely offered PrEP, while PEP with antiretroviral drugs is indicated after unsafe exposures such as unsafe sexual activity, unsafe needle use, or sexual violence. Survivors of sexual violence should have baseline HIV testing and sexually transmitted infection (STI) screening and treatment. They should also be offered mental health and other supportive counseling.
- Test youth who request HIV screening at any time even in the absence of reported risk factors. Although parent or guardian involvement is preferable, in most legal settings the adolescent’s consent should suffice for testing and treatment.
- For youth with a positive HIV test, facilitate and confirm prompt linkage to age-appropriate HIV specialty care.
Will the current report’s recommendations be met with greater uptake than previous iterations? Yes, according to Maria E. Trent, MD, MPH, chief of the division of adolescent/young adult medicine at Johns Hopkins University, Baltimore, but a fundamental first step will be the establishment of honesty and confidentiality. “Pediatricians are essential stakeholders in HIV prevention and intervention efforts in the United States. Recent data, however, suggest that pediatricians often struggle to create the essential alone time with adolescents and young adults to conduct critical sexual health conversations that allow for adequate STI/HIV risk screening,” said Dr. Trent, who was not involved in the report. “Consistently creating that space will be the first task for ensuring adherence to these recommendations.”
Strategies to optimize risk screening for clinical decision support, such as confidential online previsit questionnaires that link to the electronic medical record, may facilitate discussions during the visit while maintaining clinician efficiency, she added.
Furthermore, while one-time general HIV screening during adolescence will be an easy goal, “integrating annual testing, biomedical intervention for PrEP/PEP, and ongoing follow-up and testing for those on biomedical intervention may present practical but not insurmountable challenges,” Dr. Trent said.
When pediatricians recognize that care is suboptimal in practice, ensuring that pediatricians have established linkages to adolescent-friendly services for free or low-cost HIV testing, PrEP/PEP, and HIV management will prevent gaps in care, Dr. Trent continued. “The most exciting development in health care is that telemedicine can now be used to work with young people, giving the practicing pediatrician more opportunities and flexibility to deliver and triage care.”
Will any of the guidelines such as an adolescent’s right to independent consent be considered unacceptable by parents? “While this part of the recommendations is not new, the thought that their adolescent can initiate and receive confidential care for HIV prevention or intervention without their knowledge or consent may initially be challenging to process,” Dr. Trent said. “Ultimately, what I’ve observed in practice is that parents are relieved and often proud of their young person for taking the initiative to engage in self-care to maintain their health and relieved to be involved as a critical support person.”
She added that pediatricians need to make their practice policies clear and have information available for parents on state laws related to confidential care. “They also need to carefully use the electronic health record to avoid errors in disclosures to proxies without patient consent.”
Dr. Rakhmanina agreed there will likely be greater adherence to this round of recommendations. “The culture of addressing sexual and reproductive health issues among adolescents in the U.S. is changing among pediatric providers, and we start seeing more champions of PrEP and HIV testing in our communities,” she said.
This study received no external funding. The authors had no financial relationships or potential conflicts of interest to disclose. Dr. Trent disclosed no competing interests relevant to her comments.
Pediatricians should take a more proactive role in protecting children and adolescents from HIV infections, according to updated guidance from the American Academy of Pediatrics. The comprehensive new recommendations stress winning the trust and confidence of pediatric patients and reaffirm support for testing and treating adolescents without parental consent where state laws allow.
While the number of HIV-infected people in the United States remains high, most sexually active youth do not believe they are at risk and have never been tested, noted authors Katherine K. Hsu, MD, MPH, of the Massachusetts Department of Public Health and Boston University Medical Center, and Natella Yurievna Rakhmanina, MD, PhD, of Children’s National Hospital and George Washington University, both in Washington.
That is a knowledge gap that pediatricians are well situated to fill. “Pediatricians can play a key role in preventing and controlling HIV infection by promoting risk-reduction counseling and offering routine HIV testing and prophylaxis to adolescent and young adult (youth) patients,” they wrote on Dec. 20, 2021, in their study published in Pediatrics.
Key components of youth encounters, they stressed, is creating safe environments for obtaining an accurate sexual and reproductive health assessment and providing nonstigmatizing risk counseling.
According to Dr. Rakhmanina, major barriers to addressing preventive HIV counseling have included pediatricians’ lack of time, cultural differences, adolescents’ inaccurate responses, discomfort discussing sexual issues, and adolescents’ fear of parent or caregiver notification. Other concerns have been lack of adequate payment and insufficient training in how to talk to adolescents about sexual and reproductive issues.
According to the Centers for Disease Control and Prevention, at year end in 2018 an estimated 1,173,900 people age 13 or older were living with HIV infection in the United States, of whom 47,800 (4%) were adolescents and young adults 13-24 years of age.
These estimates include diagnosed and undiagnosed individuals. Between 2014 and 2018, new diagnoses of HIV infection accounted for 21% (7,817 of 37,515) of all new HIV diagnoses in the United States.
The new AAP clinical report updates policy statements from 2001 and again 2011 that encouraged HIV testing of all sexually active youth.
It reflects changes in epidemiology, advances in diagnostic testing with improved immunoassays, and updated recommendations for HIV testing and postexposure prophylaxis (PEP), as well as new guidance for pre-exposure prophylaxis (PrEP).
A 2017 study found that the 2011 HIV testing guidelines was associated with only a slight increase in HIV screening and a shift toward testing younger people and away from testing on the basis of risk.
Against this backdrop of persistent HIV infection and to-date modest uptake of earlier guidance, the 2021 statement made 14 main recommendations to pediatricians. Among these:
- Foster open discussion of gender and sexual orientation and behavior, as well as reproductive health issues.
- Recognize the clinical presentation of the acute retroviral syndrome, which can present as syndromes resembling infectious mononucleosis and influenza.
- Consider including virologic testing in the diagnostic workup of sexually active youth.
- Consider routine HIV screening for all youth 15 years or older at least once and rescreening high-risk youth. Those at higher risk should be rescreened at least annually, and potentially as frequently as every 3-6 months.
- Youth at substantial risk should be routinely offered PrEP, while PEP with antiretroviral drugs is indicated after unsafe exposures such as unsafe sexual activity, unsafe needle use, or sexual violence. Survivors of sexual violence should have baseline HIV testing and sexually transmitted infection (STI) screening and treatment. They should also be offered mental health and other supportive counseling.
- Test youth who request HIV screening at any time even in the absence of reported risk factors. Although parent or guardian involvement is preferable, in most legal settings the adolescent’s consent should suffice for testing and treatment.
- For youth with a positive HIV test, facilitate and confirm prompt linkage to age-appropriate HIV specialty care.
Will the current report’s recommendations be met with greater uptake than previous iterations? Yes, according to Maria E. Trent, MD, MPH, chief of the division of adolescent/young adult medicine at Johns Hopkins University, Baltimore, but a fundamental first step will be the establishment of honesty and confidentiality. “Pediatricians are essential stakeholders in HIV prevention and intervention efforts in the United States. Recent data, however, suggest that pediatricians often struggle to create the essential alone time with adolescents and young adults to conduct critical sexual health conversations that allow for adequate STI/HIV risk screening,” said Dr. Trent, who was not involved in the report. “Consistently creating that space will be the first task for ensuring adherence to these recommendations.”
Strategies to optimize risk screening for clinical decision support, such as confidential online previsit questionnaires that link to the electronic medical record, may facilitate discussions during the visit while maintaining clinician efficiency, she added.
Furthermore, while one-time general HIV screening during adolescence will be an easy goal, “integrating annual testing, biomedical intervention for PrEP/PEP, and ongoing follow-up and testing for those on biomedical intervention may present practical but not insurmountable challenges,” Dr. Trent said.
When pediatricians recognize that care is suboptimal in practice, ensuring that pediatricians have established linkages to adolescent-friendly services for free or low-cost HIV testing, PrEP/PEP, and HIV management will prevent gaps in care, Dr. Trent continued. “The most exciting development in health care is that telemedicine can now be used to work with young people, giving the practicing pediatrician more opportunities and flexibility to deliver and triage care.”
Will any of the guidelines such as an adolescent’s right to independent consent be considered unacceptable by parents? “While this part of the recommendations is not new, the thought that their adolescent can initiate and receive confidential care for HIV prevention or intervention without their knowledge or consent may initially be challenging to process,” Dr. Trent said. “Ultimately, what I’ve observed in practice is that parents are relieved and often proud of their young person for taking the initiative to engage in self-care to maintain their health and relieved to be involved as a critical support person.”
She added that pediatricians need to make their practice policies clear and have information available for parents on state laws related to confidential care. “They also need to carefully use the electronic health record to avoid errors in disclosures to proxies without patient consent.”
Dr. Rakhmanina agreed there will likely be greater adherence to this round of recommendations. “The culture of addressing sexual and reproductive health issues among adolescents in the U.S. is changing among pediatric providers, and we start seeing more champions of PrEP and HIV testing in our communities,” she said.
This study received no external funding. The authors had no financial relationships or potential conflicts of interest to disclose. Dr. Trent disclosed no competing interests relevant to her comments.
Pediatricians should take a more proactive role in protecting children and adolescents from HIV infections, according to updated guidance from the American Academy of Pediatrics. The comprehensive new recommendations stress winning the trust and confidence of pediatric patients and reaffirm support for testing and treating adolescents without parental consent where state laws allow.
While the number of HIV-infected people in the United States remains high, most sexually active youth do not believe they are at risk and have never been tested, noted authors Katherine K. Hsu, MD, MPH, of the Massachusetts Department of Public Health and Boston University Medical Center, and Natella Yurievna Rakhmanina, MD, PhD, of Children’s National Hospital and George Washington University, both in Washington.
That is a knowledge gap that pediatricians are well situated to fill. “Pediatricians can play a key role in preventing and controlling HIV infection by promoting risk-reduction counseling and offering routine HIV testing and prophylaxis to adolescent and young adult (youth) patients,” they wrote on Dec. 20, 2021, in their study published in Pediatrics.
Key components of youth encounters, they stressed, is creating safe environments for obtaining an accurate sexual and reproductive health assessment and providing nonstigmatizing risk counseling.
According to Dr. Rakhmanina, major barriers to addressing preventive HIV counseling have included pediatricians’ lack of time, cultural differences, adolescents’ inaccurate responses, discomfort discussing sexual issues, and adolescents’ fear of parent or caregiver notification. Other concerns have been lack of adequate payment and insufficient training in how to talk to adolescents about sexual and reproductive issues.
According to the Centers for Disease Control and Prevention, at year end in 2018 an estimated 1,173,900 people age 13 or older were living with HIV infection in the United States, of whom 47,800 (4%) were adolescents and young adults 13-24 years of age.
These estimates include diagnosed and undiagnosed individuals. Between 2014 and 2018, new diagnoses of HIV infection accounted for 21% (7,817 of 37,515) of all new HIV diagnoses in the United States.
The new AAP clinical report updates policy statements from 2001 and again 2011 that encouraged HIV testing of all sexually active youth.
It reflects changes in epidemiology, advances in diagnostic testing with improved immunoassays, and updated recommendations for HIV testing and postexposure prophylaxis (PEP), as well as new guidance for pre-exposure prophylaxis (PrEP).
A 2017 study found that the 2011 HIV testing guidelines was associated with only a slight increase in HIV screening and a shift toward testing younger people and away from testing on the basis of risk.
Against this backdrop of persistent HIV infection and to-date modest uptake of earlier guidance, the 2021 statement made 14 main recommendations to pediatricians. Among these:
- Foster open discussion of gender and sexual orientation and behavior, as well as reproductive health issues.
- Recognize the clinical presentation of the acute retroviral syndrome, which can present as syndromes resembling infectious mononucleosis and influenza.
- Consider including virologic testing in the diagnostic workup of sexually active youth.
- Consider routine HIV screening for all youth 15 years or older at least once and rescreening high-risk youth. Those at higher risk should be rescreened at least annually, and potentially as frequently as every 3-6 months.
- Youth at substantial risk should be routinely offered PrEP, while PEP with antiretroviral drugs is indicated after unsafe exposures such as unsafe sexual activity, unsafe needle use, or sexual violence. Survivors of sexual violence should have baseline HIV testing and sexually transmitted infection (STI) screening and treatment. They should also be offered mental health and other supportive counseling.
- Test youth who request HIV screening at any time even in the absence of reported risk factors. Although parent or guardian involvement is preferable, in most legal settings the adolescent’s consent should suffice for testing and treatment.
- For youth with a positive HIV test, facilitate and confirm prompt linkage to age-appropriate HIV specialty care.
Will the current report’s recommendations be met with greater uptake than previous iterations? Yes, according to Maria E. Trent, MD, MPH, chief of the division of adolescent/young adult medicine at Johns Hopkins University, Baltimore, but a fundamental first step will be the establishment of honesty and confidentiality. “Pediatricians are essential stakeholders in HIV prevention and intervention efforts in the United States. Recent data, however, suggest that pediatricians often struggle to create the essential alone time with adolescents and young adults to conduct critical sexual health conversations that allow for adequate STI/HIV risk screening,” said Dr. Trent, who was not involved in the report. “Consistently creating that space will be the first task for ensuring adherence to these recommendations.”
Strategies to optimize risk screening for clinical decision support, such as confidential online previsit questionnaires that link to the electronic medical record, may facilitate discussions during the visit while maintaining clinician efficiency, she added.
Furthermore, while one-time general HIV screening during adolescence will be an easy goal, “integrating annual testing, biomedical intervention for PrEP/PEP, and ongoing follow-up and testing for those on biomedical intervention may present practical but not insurmountable challenges,” Dr. Trent said.
When pediatricians recognize that care is suboptimal in practice, ensuring that pediatricians have established linkages to adolescent-friendly services for free or low-cost HIV testing, PrEP/PEP, and HIV management will prevent gaps in care, Dr. Trent continued. “The most exciting development in health care is that telemedicine can now be used to work with young people, giving the practicing pediatrician more opportunities and flexibility to deliver and triage care.”
Will any of the guidelines such as an adolescent’s right to independent consent be considered unacceptable by parents? “While this part of the recommendations is not new, the thought that their adolescent can initiate and receive confidential care for HIV prevention or intervention without their knowledge or consent may initially be challenging to process,” Dr. Trent said. “Ultimately, what I’ve observed in practice is that parents are relieved and often proud of their young person for taking the initiative to engage in self-care to maintain their health and relieved to be involved as a critical support person.”
She added that pediatricians need to make their practice policies clear and have information available for parents on state laws related to confidential care. “They also need to carefully use the electronic health record to avoid errors in disclosures to proxies without patient consent.”
Dr. Rakhmanina agreed there will likely be greater adherence to this round of recommendations. “The culture of addressing sexual and reproductive health issues among adolescents in the U.S. is changing among pediatric providers, and we start seeing more champions of PrEP and HIV testing in our communities,” she said.
This study received no external funding. The authors had no financial relationships or potential conflicts of interest to disclose. Dr. Trent disclosed no competing interests relevant to her comments.
FROM PEDIATRICS
Voxelotor for sickle cell anemia now down to 4-year-olds
The indication had previously been for patients 12 years old and up, the FDA said in an announcement.
Voxelotor (Oxbryta) was originally approved for sickle cell disease in November 2019 and was described as the first drug that directly inhibits sickle hemoglobin polymerization, the root cause of the disease. It binds and stabilizes hemoglobin to prevent red blood cells from sickling and being destroyed.
Approval for the new indication of use in children down to age 4 was based on data from a phase 2 trial that involved 45 children aged 4-11 years; the results show that 36% had an increase in hemoglobin greater than 1 g/dL by week 24, the FDA said.
“Complications of [sickle cell disease] that can cause irreversible organ damage are known to begin in the first few years of life, which is why earlier intervention is critical,” commented Ted Love, MD, president and CEO of Global Blood Therapeutics, the manufacturer, in a press release.
The company is studying voxelotor in children as young as 9 months old.
The agent was granted an accelerated approval by the FDA, so continued approval depends on additional data to confirm that increases in hemoglobin have clinical benefit.
With the new approvals, voxelotor is now available in 500-mg tablets and the 300-mg tablets for oral suspension. Dosing for ages 12 years and up is 1,500 mg once daily. Dosing for children 4 to up to 12 years old is weight based.
The most common side effects are headache, vomiting, diarrhea, abdominal pain, nausea, rash, and fever.
A version of this article first appeared on Medscape.com.
The indication had previously been for patients 12 years old and up, the FDA said in an announcement.
Voxelotor (Oxbryta) was originally approved for sickle cell disease in November 2019 and was described as the first drug that directly inhibits sickle hemoglobin polymerization, the root cause of the disease. It binds and stabilizes hemoglobin to prevent red blood cells from sickling and being destroyed.
Approval for the new indication of use in children down to age 4 was based on data from a phase 2 trial that involved 45 children aged 4-11 years; the results show that 36% had an increase in hemoglobin greater than 1 g/dL by week 24, the FDA said.
“Complications of [sickle cell disease] that can cause irreversible organ damage are known to begin in the first few years of life, which is why earlier intervention is critical,” commented Ted Love, MD, president and CEO of Global Blood Therapeutics, the manufacturer, in a press release.
The company is studying voxelotor in children as young as 9 months old.
The agent was granted an accelerated approval by the FDA, so continued approval depends on additional data to confirm that increases in hemoglobin have clinical benefit.
With the new approvals, voxelotor is now available in 500-mg tablets and the 300-mg tablets for oral suspension. Dosing for ages 12 years and up is 1,500 mg once daily. Dosing for children 4 to up to 12 years old is weight based.
The most common side effects are headache, vomiting, diarrhea, abdominal pain, nausea, rash, and fever.
A version of this article first appeared on Medscape.com.
The indication had previously been for patients 12 years old and up, the FDA said in an announcement.
Voxelotor (Oxbryta) was originally approved for sickle cell disease in November 2019 and was described as the first drug that directly inhibits sickle hemoglobin polymerization, the root cause of the disease. It binds and stabilizes hemoglobin to prevent red blood cells from sickling and being destroyed.
Approval for the new indication of use in children down to age 4 was based on data from a phase 2 trial that involved 45 children aged 4-11 years; the results show that 36% had an increase in hemoglobin greater than 1 g/dL by week 24, the FDA said.
“Complications of [sickle cell disease] that can cause irreversible organ damage are known to begin in the first few years of life, which is why earlier intervention is critical,” commented Ted Love, MD, president and CEO of Global Blood Therapeutics, the manufacturer, in a press release.
The company is studying voxelotor in children as young as 9 months old.
The agent was granted an accelerated approval by the FDA, so continued approval depends on additional data to confirm that increases in hemoglobin have clinical benefit.
With the new approvals, voxelotor is now available in 500-mg tablets and the 300-mg tablets for oral suspension. Dosing for ages 12 years and up is 1,500 mg once daily. Dosing for children 4 to up to 12 years old is weight based.
The most common side effects are headache, vomiting, diarrhea, abdominal pain, nausea, rash, and fever.
A version of this article first appeared on Medscape.com.
CDC supports ‘test-to-stay’ for COVID- exposed students
The Centers for Disease Control and Prevention has announced that in the following days.
The new guidance, known as the “test-to-stay” protocol, would reduce the number of children who are expected to stay home as a close contact to someone who tested positive for the virus.
“Test-to-stay is an encouraging public health practice to keep our children in schools,” Rochelle Walensky, MD, director of the CDC, said during a White House press briefing.
When a COVID-19 case is identified in a school, the test-to-stay strategy allows schools to implement regular testing rather than quarantine close contacts. If the contacts don’t experience symptoms and test negative at least twice in a seven-day period, they can continue in-person learning. If they test positive, then they are required to isolate.
In recent months, the CDC has collaborated with several school districts across the United States to evaluate test-to-stay programs. On Dec. 17, the CDC published two studies in its Morbidity and Mortality Weekly Report that demonstrated the effectiveness of these programs in limiting the spread of the virus while also keeping students in class.
“CDC is updating our materials to help schools and parents know how to best implement this promising and now-proven practice, along with our multi-layer prevention strategies that will help keep our children in the classroom safely,” Dr. Walensky said. “These studies demonstrated that test-to-stay works to keep unvaccinated children in school safely.”
In one study, researchers analyzed data for public schools in Los Angeles County between Aug. 16 and Oct. 31, where 432 schools implemented test-to-stay and 1,635 did not.
The Los Angeles County Department of Public Health found that COVID-19 cases did not increase among the schools that used the protocol, as compared with schools that didn’t.
Before test-to-stay was implemented, the average daily number of cases was 10 cases per 100,000 students in districts that later adopted the protocol and 20 cases per 100,000 students in districts that didn’t. After the program was implemented, average daily case rates declined in all school districts but remained lower in test-to-stay districts, with 6 cases per 100,000 students as compared with 11 cases per 100,000 students in districts that didn’t do the protocol.
In addition, schools that didn’t use the test-to-stay program “lost substantial in-person school days,” researchers wrote. At the same time, implementing the program “requires resources that might be currently unavailable for some schools,” they added, noting that “a higher percentage of disadvantaged schools” didn’t do the protocol.
The program requires personnel who can track which students need to be tested, their results and when they can come off the list of close contacts, officials told CNN. This can be a challenge for overstretched school nursing staff.
In another study published last week, researchers analyzed data between Aug. 9 and Oct. 29 for 90 schools across 31 districts in Lake County, Ill., that implemented test-to-stay programs. During that time, the schools reported 258 COVID-19 cases and 1,664 close contacts.
The Lake County Health Department examined the number of close contacts that later tested positive and whether the virus further spread from the close contacts to other people. They found that 16 of the close contacts tested positive and that these were all students. No one appeared to transmit the virus to others at school, but nine cases were identified among household contacts.
Overall, study authors wrote, the test-to-stay protocol preserved in-person learning days for students. In addition, regular testing, masking, and physical distancing led to lower virus transmission in school.
“The test-to-stay-programs are really good at balancing the costs and benefits,” Zoe McLaren, a health policy expert at the University of Maryland at Baltimore, told The New York Times.
“What the test-to-stay program does is help us keep COVID cases down, while also trying to make sure we keep kids in school as much as possible, which I think is really important,” she said.
A version of this article first appeared on WebMD.com.
The Centers for Disease Control and Prevention has announced that in the following days.
The new guidance, known as the “test-to-stay” protocol, would reduce the number of children who are expected to stay home as a close contact to someone who tested positive for the virus.
“Test-to-stay is an encouraging public health practice to keep our children in schools,” Rochelle Walensky, MD, director of the CDC, said during a White House press briefing.
When a COVID-19 case is identified in a school, the test-to-stay strategy allows schools to implement regular testing rather than quarantine close contacts. If the contacts don’t experience symptoms and test negative at least twice in a seven-day period, they can continue in-person learning. If they test positive, then they are required to isolate.
In recent months, the CDC has collaborated with several school districts across the United States to evaluate test-to-stay programs. On Dec. 17, the CDC published two studies in its Morbidity and Mortality Weekly Report that demonstrated the effectiveness of these programs in limiting the spread of the virus while also keeping students in class.
“CDC is updating our materials to help schools and parents know how to best implement this promising and now-proven practice, along with our multi-layer prevention strategies that will help keep our children in the classroom safely,” Dr. Walensky said. “These studies demonstrated that test-to-stay works to keep unvaccinated children in school safely.”
In one study, researchers analyzed data for public schools in Los Angeles County between Aug. 16 and Oct. 31, where 432 schools implemented test-to-stay and 1,635 did not.
The Los Angeles County Department of Public Health found that COVID-19 cases did not increase among the schools that used the protocol, as compared with schools that didn’t.
Before test-to-stay was implemented, the average daily number of cases was 10 cases per 100,000 students in districts that later adopted the protocol and 20 cases per 100,000 students in districts that didn’t. After the program was implemented, average daily case rates declined in all school districts but remained lower in test-to-stay districts, with 6 cases per 100,000 students as compared with 11 cases per 100,000 students in districts that didn’t do the protocol.
In addition, schools that didn’t use the test-to-stay program “lost substantial in-person school days,” researchers wrote. At the same time, implementing the program “requires resources that might be currently unavailable for some schools,” they added, noting that “a higher percentage of disadvantaged schools” didn’t do the protocol.
The program requires personnel who can track which students need to be tested, their results and when they can come off the list of close contacts, officials told CNN. This can be a challenge for overstretched school nursing staff.
In another study published last week, researchers analyzed data between Aug. 9 and Oct. 29 for 90 schools across 31 districts in Lake County, Ill., that implemented test-to-stay programs. During that time, the schools reported 258 COVID-19 cases and 1,664 close contacts.
The Lake County Health Department examined the number of close contacts that later tested positive and whether the virus further spread from the close contacts to other people. They found that 16 of the close contacts tested positive and that these were all students. No one appeared to transmit the virus to others at school, but nine cases were identified among household contacts.
Overall, study authors wrote, the test-to-stay protocol preserved in-person learning days for students. In addition, regular testing, masking, and physical distancing led to lower virus transmission in school.
“The test-to-stay-programs are really good at balancing the costs and benefits,” Zoe McLaren, a health policy expert at the University of Maryland at Baltimore, told The New York Times.
“What the test-to-stay program does is help us keep COVID cases down, while also trying to make sure we keep kids in school as much as possible, which I think is really important,” she said.
A version of this article first appeared on WebMD.com.
The Centers for Disease Control and Prevention has announced that in the following days.
The new guidance, known as the “test-to-stay” protocol, would reduce the number of children who are expected to stay home as a close contact to someone who tested positive for the virus.
“Test-to-stay is an encouraging public health practice to keep our children in schools,” Rochelle Walensky, MD, director of the CDC, said during a White House press briefing.
When a COVID-19 case is identified in a school, the test-to-stay strategy allows schools to implement regular testing rather than quarantine close contacts. If the contacts don’t experience symptoms and test negative at least twice in a seven-day period, they can continue in-person learning. If they test positive, then they are required to isolate.
In recent months, the CDC has collaborated with several school districts across the United States to evaluate test-to-stay programs. On Dec. 17, the CDC published two studies in its Morbidity and Mortality Weekly Report that demonstrated the effectiveness of these programs in limiting the spread of the virus while also keeping students in class.
“CDC is updating our materials to help schools and parents know how to best implement this promising and now-proven practice, along with our multi-layer prevention strategies that will help keep our children in the classroom safely,” Dr. Walensky said. “These studies demonstrated that test-to-stay works to keep unvaccinated children in school safely.”
In one study, researchers analyzed data for public schools in Los Angeles County between Aug. 16 and Oct. 31, where 432 schools implemented test-to-stay and 1,635 did not.
The Los Angeles County Department of Public Health found that COVID-19 cases did not increase among the schools that used the protocol, as compared with schools that didn’t.
Before test-to-stay was implemented, the average daily number of cases was 10 cases per 100,000 students in districts that later adopted the protocol and 20 cases per 100,000 students in districts that didn’t. After the program was implemented, average daily case rates declined in all school districts but remained lower in test-to-stay districts, with 6 cases per 100,000 students as compared with 11 cases per 100,000 students in districts that didn’t do the protocol.
In addition, schools that didn’t use the test-to-stay program “lost substantial in-person school days,” researchers wrote. At the same time, implementing the program “requires resources that might be currently unavailable for some schools,” they added, noting that “a higher percentage of disadvantaged schools” didn’t do the protocol.
The program requires personnel who can track which students need to be tested, their results and when they can come off the list of close contacts, officials told CNN. This can be a challenge for overstretched school nursing staff.
In another study published last week, researchers analyzed data between Aug. 9 and Oct. 29 for 90 schools across 31 districts in Lake County, Ill., that implemented test-to-stay programs. During that time, the schools reported 258 COVID-19 cases and 1,664 close contacts.
The Lake County Health Department examined the number of close contacts that later tested positive and whether the virus further spread from the close contacts to other people. They found that 16 of the close contacts tested positive and that these were all students. No one appeared to transmit the virus to others at school, but nine cases were identified among household contacts.
Overall, study authors wrote, the test-to-stay protocol preserved in-person learning days for students. In addition, regular testing, masking, and physical distancing led to lower virus transmission in school.
“The test-to-stay-programs are really good at balancing the costs and benefits,” Zoe McLaren, a health policy expert at the University of Maryland at Baltimore, told The New York Times.
“What the test-to-stay program does is help us keep COVID cases down, while also trying to make sure we keep kids in school as much as possible, which I think is really important,” she said.
A version of this article first appeared on WebMD.com.
Blastomycosislike Pyoderma: Verrucous Hyperpigmented Plaques on the Pretibial Shins
To the Editor:
Blastomycosislike pyoderma (BLP), also commonly referred to as pyoderma vegetans, is a rare cutaneous bacterial infection that often mimics other fungal, inflammatory, or neoplastic disorders.1 It is characterized by a collection of neutrophilic abscesses with pseudoepitheliomatous hyperplasia that coalesce into crusted plaques.
A 15-year-old adolescent girl with a history of type 1 diabetes mellitus was admitted for diabetic ketoacidosis. The patient presented with bilateral pretibial lesions of 6 years’ duration that developed after swimming in a pool following reported trauma to the site. These pruritic plaques had grown slowly and were occasionally tender. Of note, with episodes of hyperglycemia, the lesions developed purulent drainage.
Upon admission to the hospital and subsequent dermatology consultation, physical examination revealed the right pretibial shin had a 15×5-cm, gray-brown, hyperpigmented, verrucous, tender plaque with purulent drainage and overlying crust (Figure 1). The left pretibial shin had a similar smaller lesion (Figure 2). Laboratory test results were notable for a white blood cell count of 41.84 cells/µL (reference range, 3.8–10.5 cells/µL), blood glucose level of 586 mg/dL (reference range, 70–99 mg/dL), and hemoglobin A1c of 11.7% (reference range, 4.0%–5.6%). A biopsy specimen from the right pretibial shin was stained with hematoxylin and eosin for dermatopathologic evaluation as well as sent for tissue culture. Tissue and wound cultures grew Staphylococcus aureus and group B Streptococcus with no fungal or acid-fast bacilli growth.
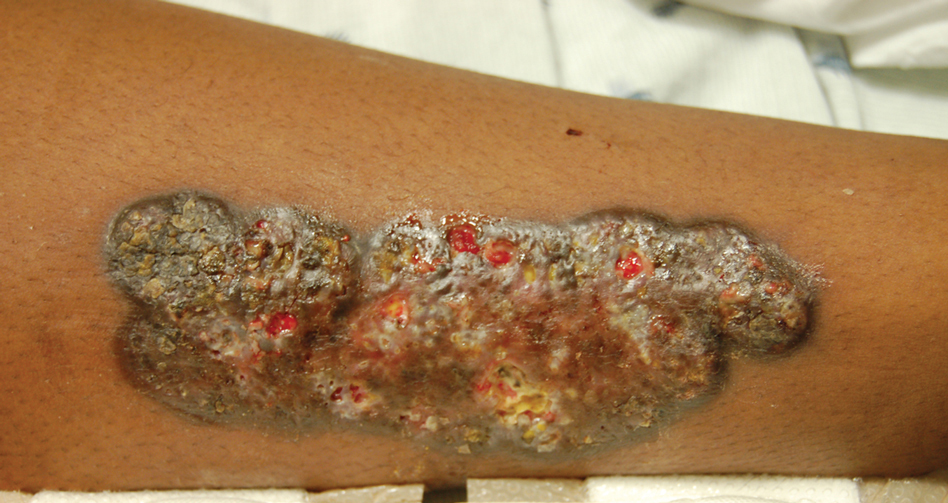
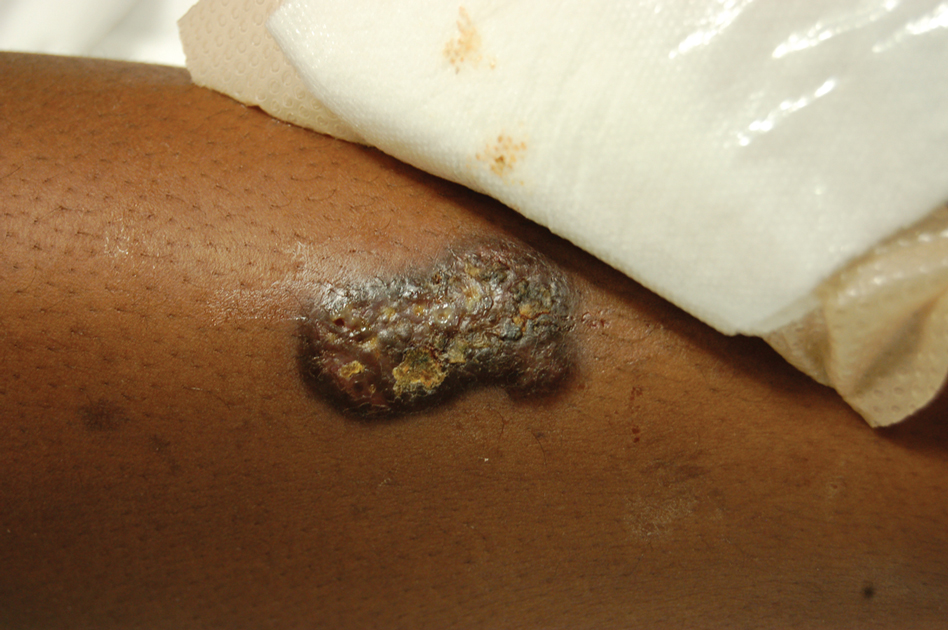
Blood cultures were negative for bacteria. Results of radiographic imaging were negative for osteomyelitis. Biopsy specimens from the right pretibial plaque showed a markedly inflamed, ruptured follicular unit with a dense dermal lympho-neutrophilic infiltrate and overlying pseudoepitheliomatous hyperplasia (Figure 3). Periodic acid–Schiff, Gomori methenamine-silver, acid-fast bacilli, and Giemsa stains were negative for organisms. No granules consistent with a Splendore-Hoeppli phenomenon were observed. These observations were consistent with a diagnosis of BLP.
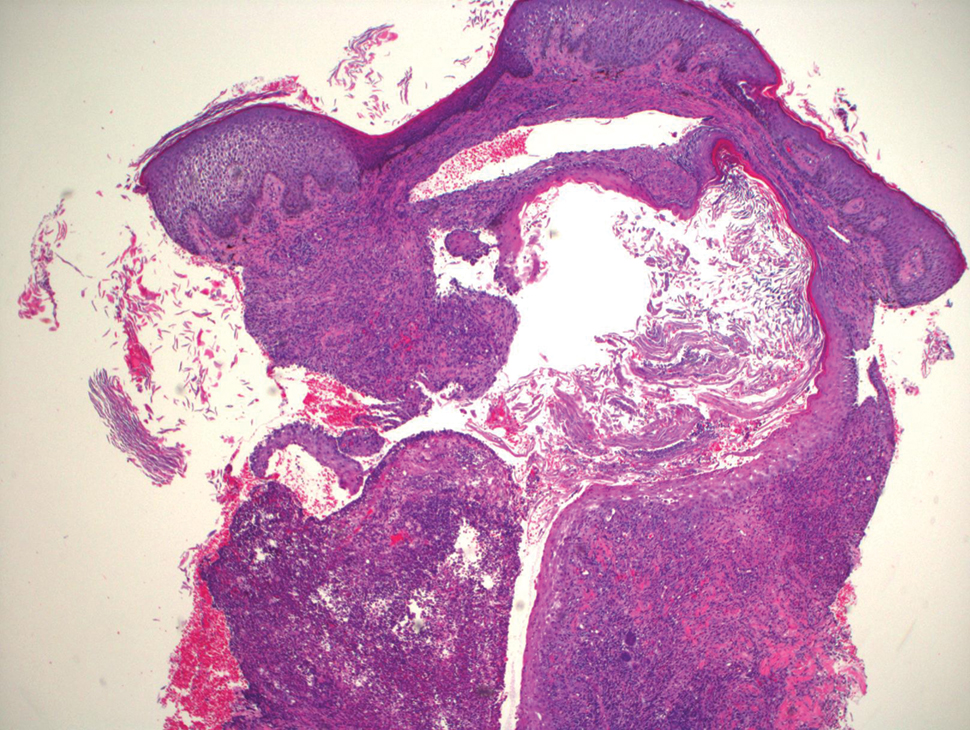
Blastomycosislike pyoderma is a rare cutaneous bacterial infection that often mimics other fungal, inflammatory, or neoplastic disorders.1 Pediatric cases also are uncommon. Blastomycosislike pyoderma most commonly is caused by infection with S aureus or group A streptococci, but several other organisms have been implicated.2 Clinically, BLP is similar to cutaneous botryomycosis, as both are caused by similar organisms.3 However, while BLP is limited to the skin, botryomycosis may involve visceral organs.
Blastomycosislike pyoderma typically presents as verrucous, hyperkeratotic, purulent plaques with raised borders. It most commonly occurs on the face, scalp, axillae, trunk, and distal extremities. Predisposing factors include immunosuppressed states such as poor nutrition, HIV, malignancy, alcoholism, and diabetes mellitus.3,4 Hyperglycemia is thought to suppress helper T cell (TH1)–dependent immunity, which may explain why our patient’s lesions worsened with hyperglycemic episodes.5Histopathology revealed pseudoepitheliomatous hyperplasia with neutrophilic abscesses.1 The distinguishing feature between botryomycosis and BLP is the development of grains known as the Splendore-Hoeppli phenomenon in botryomycosis.6 The grains are eosinophilic and contain the causative infectious agent. The presence of these grains is consistent with botryomycosis but is not pathognomonic, as it also can be found in several bacterial, fungal, and parasitic infections.3,6
The differential diagnosis of BLP includes atypical mycobacterial infection, pyoderma gangrenosum, fungal infection, and tuberculosis verrucosa cutis.7
Although BLP is caused by bacteria, response to systemic antibiotics is variable. Other treatment modalities include dapsone, systemic and intralesional corticosteroids, retinoids, debridement, CO2 laser, and excision.6,8 Lesions typically start out localized, but it is not uncommon for them to spread to distal or vulnerable tissue, such as sites of trauma or inflammation. Our patient was started on oral trimethoprim-sulfamethoxazole and showed improvement, but she worsened with subsequent hyperglycemic episodes when antibiotics were discontinued.
1. Adis¸en E, Tezel F, Gürer MA. Pyoderma vegetans: a case for discussion. Acta Derm Venereol. 2009;89:186-188.
2. Scuderi S, O’Brien B, Robertson I, et al. Heterogeneity of blastomycosis-like pyoderma: a selection of cases from the last 35 years. Australas J Dermatol. 2017;58:139-141.
3. Marschalko, M. Pyoderma vegetans: report on a case and review of data on pyoderma vegetans and cutaneous botryomycosis. Acta Dermatovenerol Alp Pannonica Adriat. 1995;4:55-59.
4. Cerullo L, Zussman J, Young L. An unusual presentation of blastomycosislike pyoderma (pyoderma vegetans) and a review of the literature. Cutis. 2009;84:201-204.
5. Tanaka Y. Immunosuppressive mechanisms in diabetes mellitus [in Japanese]. Nihon Rinsho. 2008;66:2233-2237.
6. Hussein MR. Mucocutaneous Splendore-Hoeppli phenomenon. J Cutan Pathol. 2008;35:979-988.
7. Lee YS, Jung SW, Sim HS, et al. Blastomycosis-like pyoderma with good response to acitretin. Ann Dermatol. 2011;23:365-368.
8. Kobraei KB, Wesson SK. Blastomycosis-like pyoderma: response to systemic retinoid therapy. Int J Dermatol. 2010;49:1336-1338.
To the Editor:
Blastomycosislike pyoderma (BLP), also commonly referred to as pyoderma vegetans, is a rare cutaneous bacterial infection that often mimics other fungal, inflammatory, or neoplastic disorders.1 It is characterized by a collection of neutrophilic abscesses with pseudoepitheliomatous hyperplasia that coalesce into crusted plaques.
A 15-year-old adolescent girl with a history of type 1 diabetes mellitus was admitted for diabetic ketoacidosis. The patient presented with bilateral pretibial lesions of 6 years’ duration that developed after swimming in a pool following reported trauma to the site. These pruritic plaques had grown slowly and were occasionally tender. Of note, with episodes of hyperglycemia, the lesions developed purulent drainage.
Upon admission to the hospital and subsequent dermatology consultation, physical examination revealed the right pretibial shin had a 15×5-cm, gray-brown, hyperpigmented, verrucous, tender plaque with purulent drainage and overlying crust (Figure 1). The left pretibial shin had a similar smaller lesion (Figure 2). Laboratory test results were notable for a white blood cell count of 41.84 cells/µL (reference range, 3.8–10.5 cells/µL), blood glucose level of 586 mg/dL (reference range, 70–99 mg/dL), and hemoglobin A1c of 11.7% (reference range, 4.0%–5.6%). A biopsy specimen from the right pretibial shin was stained with hematoxylin and eosin for dermatopathologic evaluation as well as sent for tissue culture. Tissue and wound cultures grew Staphylococcus aureus and group B Streptococcus with no fungal or acid-fast bacilli growth.


Blood cultures were negative for bacteria. Results of radiographic imaging were negative for osteomyelitis. Biopsy specimens from the right pretibial plaque showed a markedly inflamed, ruptured follicular unit with a dense dermal lympho-neutrophilic infiltrate and overlying pseudoepitheliomatous hyperplasia (Figure 3). Periodic acid–Schiff, Gomori methenamine-silver, acid-fast bacilli, and Giemsa stains were negative for organisms. No granules consistent with a Splendore-Hoeppli phenomenon were observed. These observations were consistent with a diagnosis of BLP.

Blastomycosislike pyoderma is a rare cutaneous bacterial infection that often mimics other fungal, inflammatory, or neoplastic disorders.1 Pediatric cases also are uncommon. Blastomycosislike pyoderma most commonly is caused by infection with S aureus or group A streptococci, but several other organisms have been implicated.2 Clinically, BLP is similar to cutaneous botryomycosis, as both are caused by similar organisms.3 However, while BLP is limited to the skin, botryomycosis may involve visceral organs.
Blastomycosislike pyoderma typically presents as verrucous, hyperkeratotic, purulent plaques with raised borders. It most commonly occurs on the face, scalp, axillae, trunk, and distal extremities. Predisposing factors include immunosuppressed states such as poor nutrition, HIV, malignancy, alcoholism, and diabetes mellitus.3,4 Hyperglycemia is thought to suppress helper T cell (TH1)–dependent immunity, which may explain why our patient’s lesions worsened with hyperglycemic episodes.5Histopathology revealed pseudoepitheliomatous hyperplasia with neutrophilic abscesses.1 The distinguishing feature between botryomycosis and BLP is the development of grains known as the Splendore-Hoeppli phenomenon in botryomycosis.6 The grains are eosinophilic and contain the causative infectious agent. The presence of these grains is consistent with botryomycosis but is not pathognomonic, as it also can be found in several bacterial, fungal, and parasitic infections.3,6
The differential diagnosis of BLP includes atypical mycobacterial infection, pyoderma gangrenosum, fungal infection, and tuberculosis verrucosa cutis.7
Although BLP is caused by bacteria, response to systemic antibiotics is variable. Other treatment modalities include dapsone, systemic and intralesional corticosteroids, retinoids, debridement, CO2 laser, and excision.6,8 Lesions typically start out localized, but it is not uncommon for them to spread to distal or vulnerable tissue, such as sites of trauma or inflammation. Our patient was started on oral trimethoprim-sulfamethoxazole and showed improvement, but she worsened with subsequent hyperglycemic episodes when antibiotics were discontinued.
To the Editor:
Blastomycosislike pyoderma (BLP), also commonly referred to as pyoderma vegetans, is a rare cutaneous bacterial infection that often mimics other fungal, inflammatory, or neoplastic disorders.1 It is characterized by a collection of neutrophilic abscesses with pseudoepitheliomatous hyperplasia that coalesce into crusted plaques.
A 15-year-old adolescent girl with a history of type 1 diabetes mellitus was admitted for diabetic ketoacidosis. The patient presented with bilateral pretibial lesions of 6 years’ duration that developed after swimming in a pool following reported trauma to the site. These pruritic plaques had grown slowly and were occasionally tender. Of note, with episodes of hyperglycemia, the lesions developed purulent drainage.
Upon admission to the hospital and subsequent dermatology consultation, physical examination revealed the right pretibial shin had a 15×5-cm, gray-brown, hyperpigmented, verrucous, tender plaque with purulent drainage and overlying crust (Figure 1). The left pretibial shin had a similar smaller lesion (Figure 2). Laboratory test results were notable for a white blood cell count of 41.84 cells/µL (reference range, 3.8–10.5 cells/µL), blood glucose level of 586 mg/dL (reference range, 70–99 mg/dL), and hemoglobin A1c of 11.7% (reference range, 4.0%–5.6%). A biopsy specimen from the right pretibial shin was stained with hematoxylin and eosin for dermatopathologic evaluation as well as sent for tissue culture. Tissue and wound cultures grew Staphylococcus aureus and group B Streptococcus with no fungal or acid-fast bacilli growth.


Blood cultures were negative for bacteria. Results of radiographic imaging were negative for osteomyelitis. Biopsy specimens from the right pretibial plaque showed a markedly inflamed, ruptured follicular unit with a dense dermal lympho-neutrophilic infiltrate and overlying pseudoepitheliomatous hyperplasia (Figure 3). Periodic acid–Schiff, Gomori methenamine-silver, acid-fast bacilli, and Giemsa stains were negative for organisms. No granules consistent with a Splendore-Hoeppli phenomenon were observed. These observations were consistent with a diagnosis of BLP.

Blastomycosislike pyoderma is a rare cutaneous bacterial infection that often mimics other fungal, inflammatory, or neoplastic disorders.1 Pediatric cases also are uncommon. Blastomycosislike pyoderma most commonly is caused by infection with S aureus or group A streptococci, but several other organisms have been implicated.2 Clinically, BLP is similar to cutaneous botryomycosis, as both are caused by similar organisms.3 However, while BLP is limited to the skin, botryomycosis may involve visceral organs.
Blastomycosislike pyoderma typically presents as verrucous, hyperkeratotic, purulent plaques with raised borders. It most commonly occurs on the face, scalp, axillae, trunk, and distal extremities. Predisposing factors include immunosuppressed states such as poor nutrition, HIV, malignancy, alcoholism, and diabetes mellitus.3,4 Hyperglycemia is thought to suppress helper T cell (TH1)–dependent immunity, which may explain why our patient’s lesions worsened with hyperglycemic episodes.5Histopathology revealed pseudoepitheliomatous hyperplasia with neutrophilic abscesses.1 The distinguishing feature between botryomycosis and BLP is the development of grains known as the Splendore-Hoeppli phenomenon in botryomycosis.6 The grains are eosinophilic and contain the causative infectious agent. The presence of these grains is consistent with botryomycosis but is not pathognomonic, as it also can be found in several bacterial, fungal, and parasitic infections.3,6
The differential diagnosis of BLP includes atypical mycobacterial infection, pyoderma gangrenosum, fungal infection, and tuberculosis verrucosa cutis.7
Although BLP is caused by bacteria, response to systemic antibiotics is variable. Other treatment modalities include dapsone, systemic and intralesional corticosteroids, retinoids, debridement, CO2 laser, and excision.6,8 Lesions typically start out localized, but it is not uncommon for them to spread to distal or vulnerable tissue, such as sites of trauma or inflammation. Our patient was started on oral trimethoprim-sulfamethoxazole and showed improvement, but she worsened with subsequent hyperglycemic episodes when antibiotics were discontinued.
1. Adis¸en E, Tezel F, Gürer MA. Pyoderma vegetans: a case for discussion. Acta Derm Venereol. 2009;89:186-188.
2. Scuderi S, O’Brien B, Robertson I, et al. Heterogeneity of blastomycosis-like pyoderma: a selection of cases from the last 35 years. Australas J Dermatol. 2017;58:139-141.
3. Marschalko, M. Pyoderma vegetans: report on a case and review of data on pyoderma vegetans and cutaneous botryomycosis. Acta Dermatovenerol Alp Pannonica Adriat. 1995;4:55-59.
4. Cerullo L, Zussman J, Young L. An unusual presentation of blastomycosislike pyoderma (pyoderma vegetans) and a review of the literature. Cutis. 2009;84:201-204.
5. Tanaka Y. Immunosuppressive mechanisms in diabetes mellitus [in Japanese]. Nihon Rinsho. 2008;66:2233-2237.
6. Hussein MR. Mucocutaneous Splendore-Hoeppli phenomenon. J Cutan Pathol. 2008;35:979-988.
7. Lee YS, Jung SW, Sim HS, et al. Blastomycosis-like pyoderma with good response to acitretin. Ann Dermatol. 2011;23:365-368.
8. Kobraei KB, Wesson SK. Blastomycosis-like pyoderma: response to systemic retinoid therapy. Int J Dermatol. 2010;49:1336-1338.
1. Adis¸en E, Tezel F, Gürer MA. Pyoderma vegetans: a case for discussion. Acta Derm Venereol. 2009;89:186-188.
2. Scuderi S, O’Brien B, Robertson I, et al. Heterogeneity of blastomycosis-like pyoderma: a selection of cases from the last 35 years. Australas J Dermatol. 2017;58:139-141.
3. Marschalko, M. Pyoderma vegetans: report on a case and review of data on pyoderma vegetans and cutaneous botryomycosis. Acta Dermatovenerol Alp Pannonica Adriat. 1995;4:55-59.
4. Cerullo L, Zussman J, Young L. An unusual presentation of blastomycosislike pyoderma (pyoderma vegetans) and a review of the literature. Cutis. 2009;84:201-204.
5. Tanaka Y. Immunosuppressive mechanisms in diabetes mellitus [in Japanese]. Nihon Rinsho. 2008;66:2233-2237.
6. Hussein MR. Mucocutaneous Splendore-Hoeppli phenomenon. J Cutan Pathol. 2008;35:979-988.
7. Lee YS, Jung SW, Sim HS, et al. Blastomycosis-like pyoderma with good response to acitretin. Ann Dermatol. 2011;23:365-368.
8. Kobraei KB, Wesson SK. Blastomycosis-like pyoderma: response to systemic retinoid therapy. Int J Dermatol. 2010;49:1336-1338.
Practice Points
- Blastomycosislike pyoderma is a rare condition secondary to bacterial infection, but as the name suggests, it also can resemble cutaneous blastomycosis.
- Blastomycosislike pyoderma most commonly occurs in immunocompromised patients.
- The most common histologic findings include suppurative and neutrophilic inflammation with pseudoepitheliomatous hyperplasia.
Pfizer COVID vaccine for younger children hits snag
Pfizer’s COVID-19 vaccine for children ages 2 to 5 years old fizzled in clinical trials, the company said on Friday, signaling a further delay in getting a vaccine to preschoolers just as Omicron bears down on the U.S.
In a news release, Pfizer reported that while its 3-microgram dose – which is less than one-third of the dose given to older children – generated a protective immune response in babies and toddlers ages 6 to 24 months, it didn’t generate adequate immunity in children ages 2 to 5.
The company plans to change its clinical trial to add a third dose for younger children in hopes of improving those results. It also plans to test a third dose of its 10-microgram vaccine for children ages 5 to 12.
If the trials are successful, Pfizer said it would submit data to the FDA for an emergency use authorization (EUA) in the first half of 2022.
That pushes the timeline of getting a vaccine to younger children back by several months. In November, Anthony Fauci, MD, head of the National Institute of Allergy Infectious Diseases, predicted a vaccine would be ready for preschoolers by spring.
“On one hand, parents are understandably disappointed,” said Jill Foster, MD, a pediatric infectious disease doctor at the University of Minnesota Medical School. “On the other, it shows that the system for testing vaccines is working. Children are not little adults and have complex immune systems, so it’s not just a matter of making the dose smaller and expecting that it will work,” she said, noting that data from Moderna’s KidCOVE study in preschoolers is pending.
Until there’s a vaccine, Dr. Foster says parents should protect babies and toddlers by making sure everyone around them is vaccinated, promote the use of face masks for everyone around them and for all children over age 2, and continue to avoid crowded gatherings, particularly those that are indoors.
“Hand sanitizer is important, but this virus, especially the Omicron variant, is very easily spread through the air, so keep the air clear of virus as much as possible,” she said.
While the youngest children are still waiting for an effective vaccine, there was reassuring news Dec. 16 about the safety of Pfizer’s vaccine for school-aged kids – those ages 5 through 11.
Out of more than 7 million doses given since this vaccine was authorized for emergency use in late October, most reactions to the vaccine – including arm pain, swelling, and fatigue – have been mild and gone away quickly, without the need to miss school or see a doctor, the CDC reported to a meeting of its Advisory Committee on Immunization Practices, or ACIP.
Many experts had been waiting to see if this vaccine would cause rare cases of heart inflammation called myocarditis, as a higher dose did in teens and young adults.
The news on this front was excellent. About 6 weeks after this vaccine became available, the CDC says there have been only eight confirmed cases of myocarditis in this age group. Six more cases are under investigation.
To put this risk into context, data collected by the American Academy of Pediatrics and the Children’s Hospital Association shows that about 1% of children who test positive for COVID-19 are hospitalized for their infections, while the risk of getting a case of myocarditis after vaccination is .0002%, making it about 5,000 times more likely that a child would need to be hospitalized for COVID-19 than for myocarditis after vaccination.
John Su, MD, who is a member of the CDC’s Vaccine Safety Team, reported there had been two deaths in children after a COVID-19 vaccination. Both were girls, ages 5 and 6. Both had complicated medical histories for several medical disorders. It’s not clear their deaths were linked to the vaccine, and the causes of their deaths are still under investigation.
A version of this article first appeared on WebMD.com.
Pfizer’s COVID-19 vaccine for children ages 2 to 5 years old fizzled in clinical trials, the company said on Friday, signaling a further delay in getting a vaccine to preschoolers just as Omicron bears down on the U.S.
In a news release, Pfizer reported that while its 3-microgram dose – which is less than one-third of the dose given to older children – generated a protective immune response in babies and toddlers ages 6 to 24 months, it didn’t generate adequate immunity in children ages 2 to 5.
The company plans to change its clinical trial to add a third dose for younger children in hopes of improving those results. It also plans to test a third dose of its 10-microgram vaccine for children ages 5 to 12.
If the trials are successful, Pfizer said it would submit data to the FDA for an emergency use authorization (EUA) in the first half of 2022.
That pushes the timeline of getting a vaccine to younger children back by several months. In November, Anthony Fauci, MD, head of the National Institute of Allergy Infectious Diseases, predicted a vaccine would be ready for preschoolers by spring.
“On one hand, parents are understandably disappointed,” said Jill Foster, MD, a pediatric infectious disease doctor at the University of Minnesota Medical School. “On the other, it shows that the system for testing vaccines is working. Children are not little adults and have complex immune systems, so it’s not just a matter of making the dose smaller and expecting that it will work,” she said, noting that data from Moderna’s KidCOVE study in preschoolers is pending.
Until there’s a vaccine, Dr. Foster says parents should protect babies and toddlers by making sure everyone around them is vaccinated, promote the use of face masks for everyone around them and for all children over age 2, and continue to avoid crowded gatherings, particularly those that are indoors.
“Hand sanitizer is important, but this virus, especially the Omicron variant, is very easily spread through the air, so keep the air clear of virus as much as possible,” she said.
While the youngest children are still waiting for an effective vaccine, there was reassuring news Dec. 16 about the safety of Pfizer’s vaccine for school-aged kids – those ages 5 through 11.
Out of more than 7 million doses given since this vaccine was authorized for emergency use in late October, most reactions to the vaccine – including arm pain, swelling, and fatigue – have been mild and gone away quickly, without the need to miss school or see a doctor, the CDC reported to a meeting of its Advisory Committee on Immunization Practices, or ACIP.
Many experts had been waiting to see if this vaccine would cause rare cases of heart inflammation called myocarditis, as a higher dose did in teens and young adults.
The news on this front was excellent. About 6 weeks after this vaccine became available, the CDC says there have been only eight confirmed cases of myocarditis in this age group. Six more cases are under investigation.
To put this risk into context, data collected by the American Academy of Pediatrics and the Children’s Hospital Association shows that about 1% of children who test positive for COVID-19 are hospitalized for their infections, while the risk of getting a case of myocarditis after vaccination is .0002%, making it about 5,000 times more likely that a child would need to be hospitalized for COVID-19 than for myocarditis after vaccination.
John Su, MD, who is a member of the CDC’s Vaccine Safety Team, reported there had been two deaths in children after a COVID-19 vaccination. Both were girls, ages 5 and 6. Both had complicated medical histories for several medical disorders. It’s not clear their deaths were linked to the vaccine, and the causes of their deaths are still under investigation.
A version of this article first appeared on WebMD.com.
Pfizer’s COVID-19 vaccine for children ages 2 to 5 years old fizzled in clinical trials, the company said on Friday, signaling a further delay in getting a vaccine to preschoolers just as Omicron bears down on the U.S.
In a news release, Pfizer reported that while its 3-microgram dose – which is less than one-third of the dose given to older children – generated a protective immune response in babies and toddlers ages 6 to 24 months, it didn’t generate adequate immunity in children ages 2 to 5.
The company plans to change its clinical trial to add a third dose for younger children in hopes of improving those results. It also plans to test a third dose of its 10-microgram vaccine for children ages 5 to 12.
If the trials are successful, Pfizer said it would submit data to the FDA for an emergency use authorization (EUA) in the first half of 2022.
That pushes the timeline of getting a vaccine to younger children back by several months. In November, Anthony Fauci, MD, head of the National Institute of Allergy Infectious Diseases, predicted a vaccine would be ready for preschoolers by spring.
“On one hand, parents are understandably disappointed,” said Jill Foster, MD, a pediatric infectious disease doctor at the University of Minnesota Medical School. “On the other, it shows that the system for testing vaccines is working. Children are not little adults and have complex immune systems, so it’s not just a matter of making the dose smaller and expecting that it will work,” she said, noting that data from Moderna’s KidCOVE study in preschoolers is pending.
Until there’s a vaccine, Dr. Foster says parents should protect babies and toddlers by making sure everyone around them is vaccinated, promote the use of face masks for everyone around them and for all children over age 2, and continue to avoid crowded gatherings, particularly those that are indoors.
“Hand sanitizer is important, but this virus, especially the Omicron variant, is very easily spread through the air, so keep the air clear of virus as much as possible,” she said.
While the youngest children are still waiting for an effective vaccine, there was reassuring news Dec. 16 about the safety of Pfizer’s vaccine for school-aged kids – those ages 5 through 11.
Out of more than 7 million doses given since this vaccine was authorized for emergency use in late October, most reactions to the vaccine – including arm pain, swelling, and fatigue – have been mild and gone away quickly, without the need to miss school or see a doctor, the CDC reported to a meeting of its Advisory Committee on Immunization Practices, or ACIP.
Many experts had been waiting to see if this vaccine would cause rare cases of heart inflammation called myocarditis, as a higher dose did in teens and young adults.
The news on this front was excellent. About 6 weeks after this vaccine became available, the CDC says there have been only eight confirmed cases of myocarditis in this age group. Six more cases are under investigation.
To put this risk into context, data collected by the American Academy of Pediatrics and the Children’s Hospital Association shows that about 1% of children who test positive for COVID-19 are hospitalized for their infections, while the risk of getting a case of myocarditis after vaccination is .0002%, making it about 5,000 times more likely that a child would need to be hospitalized for COVID-19 than for myocarditis after vaccination.
John Su, MD, who is a member of the CDC’s Vaccine Safety Team, reported there had been two deaths in children after a COVID-19 vaccination. Both were girls, ages 5 and 6. Both had complicated medical histories for several medical disorders. It’s not clear their deaths were linked to the vaccine, and the causes of their deaths are still under investigation.
A version of this article first appeared on WebMD.com.
Fitusiran: Great ‘leap forward’ in hemophilia treatment
Fitusiran is a small interfering RNA molecule that blocks antithrombin production in liver cells. Instead of taking the traditional approach in hemophilia treatment of boosting the coagulation cascade by replacing what’s missing, the idea of fitusiran is to short circuit the body’s anticoagulation system by targeting antithrombin.
Patients in two trials presented at the meeting, ATLAS-A/B and ATLAS-INH, had about a 90% reduction in their annualized bleeding rates when treated with prophylactic fitusiran, with half or more having no bleeds that required treatment during the 9-month trials. The median annualized bleeding rate fell to 0, trial investigators reported at the meeting.
These findings held in both hemophilia A and B with and without inhibitors, which are antibodies formed against exogenous clotting factors, and on subanalysis of spontaneous and joint bleeding rates. Reduced bleeding was associated with substantial improvements in health-care related quality of life, particularly in the physical health domain.
A question about study design
An audience member at ASH noted that the trials didn’t compare fitusiran against prophylactic treatment, which is standard of care for hemophilia, but rather against episodic treatment – concentrated factors or bypassing agents in subjects with inhibitors – once subjects in the control groups started to bleed.
Still, the numbers reported in the studies “have never been achieved with standard prophylaxis in the past.” Furthermore, standard prophylaxis requires lifelong intravenous infusions, sometimes several a week, said lead ATLAS-A/B investigator Alok Srivastava, MD, a hematologist at the Christian Medical College in Vellore, India.
Fitusiran was dosed in the studies as a once-a-month 80 mg subcutaneous injection, so is much less bothersome. Also, it seems likely that some patients will only need dosing every other month. Maker Sanofi Genzyme is exploring lower and less frequent dosing to reduce thrombotic event risks that emerged in earlier studies, said Steven Pipe, MD, a pediatric hematologist at the University of Michigan, Ann Arbor, and the senior investigator on ATLAS-A/B, which assessed fitusiran in patients without inhibitors.
Serious thrombotic events occurred in two fitusiran patients in the trials, one of which led to discontinuation.
No pricing information
Overall, “I think [fitusiran] is a tremendous leap forward” with “the opportunity to transform the day-to-day lives of patients,” particularly those with hemophilia B, who have limited treatment options, Dr. Pipe said.
If approved for the U.S. market, fitusiran will go up against the monoclonal antibody emicizumab (Hemlibra), a subcutaneous injection dosed weekly to monthly that mimics the function of factor VIII, so it’s approved only for hemophilia A with or without inhibitors.
Several audience members at ASH noted that a major consideration for fitusiran, if approved, will be its cost. There’s no pricing information yet, but annual list price for emicizumab is reported to be in the $500,000 range.
For hemophilia A, “it will come to what proves to be the most efficacious and safe, with also consideration given to pricing,” Nigel Key, MD, a hematologist at the University of North Carolina at Chapel Hill, said in a comment.
Trial details
In ATLAS-A/B, 80 male patients were randomized to prophylactic fitusiran once monthly and 40 to continue with clotting factors as needed for bleeding. Just over 20% had hemophilia B, the rest hemophilia A. The mean age was 34 years, and subjects had a mean of about 12 bleeds in the 6 months leading up to the study.
Half of the fitusiran group had no treated bleeds during the study period versus only 5% in the control arm.
The five treatment emergent serious adverse events in the fitusiran arm included cholelithiasis in two subjects, plus cholecystitis, lower respiratory tract infection, and asthma in one each. Two fitusiran patients discontinued treatment because of cholecystitis and increased alanine aminotransferase.
ATLAS-INH had the same study design, and investigated patients with inhibitors; again, just over 20% had hemophilia B, the rest A. Mean age was 28 years, and patients had a mean of about 13 bleeds over the 6 months before the study. A total of 38 subjects were randomized to fitusiran, and 19 to bypassing agents as needed.
Almost 66% of fitusiran patients had no treated bleeds versus about 5% in the control arm.
There “was far less bleeding both for hemophilia A and B. It’s definitely a clinically meaningful and important” effect, said ATLAS-INH lead investigator Guy Young, MD, director of the hemostasis and thrombosis program at Children’s Hospital Los Angeles.
He said the improved quality of life with fitusiran noted in both trials was “not surprising. If you only have to dose once a month subcutaneously, and you are not bleeding, of course your quality of life is going to improve.”
Both fitusiran thrombotic events occurred in ATLAS-INH. One patient developed deep vein thrombosis, subclavian vein thrombosis, and superficial thrombophlebitis but stayed in the study. Another discontinued after developing suspected spinal vessel thrombosis. Serious adverse events among five other subjects included acute cholecystitis and hematuria.
Fitusiran was associated with liver enzyme elevations in both trials, but they were generally mild to moderate.
The studies were funded by fitusiran maker Sanofi Genzyme. Several investigators were employees. Dr. Pipe is a consultant, Dr. Srivastava is a researcher and adviser, and Dr. Young is a speaker and consultant for the company and disclosed honoraria from it. Dr. Key had no relevant disclosures.
Fitusiran is a small interfering RNA molecule that blocks antithrombin production in liver cells. Instead of taking the traditional approach in hemophilia treatment of boosting the coagulation cascade by replacing what’s missing, the idea of fitusiran is to short circuit the body’s anticoagulation system by targeting antithrombin.
Patients in two trials presented at the meeting, ATLAS-A/B and ATLAS-INH, had about a 90% reduction in their annualized bleeding rates when treated with prophylactic fitusiran, with half or more having no bleeds that required treatment during the 9-month trials. The median annualized bleeding rate fell to 0, trial investigators reported at the meeting.
These findings held in both hemophilia A and B with and without inhibitors, which are antibodies formed against exogenous clotting factors, and on subanalysis of spontaneous and joint bleeding rates. Reduced bleeding was associated with substantial improvements in health-care related quality of life, particularly in the physical health domain.
A question about study design
An audience member at ASH noted that the trials didn’t compare fitusiran against prophylactic treatment, which is standard of care for hemophilia, but rather against episodic treatment – concentrated factors or bypassing agents in subjects with inhibitors – once subjects in the control groups started to bleed.
Still, the numbers reported in the studies “have never been achieved with standard prophylaxis in the past.” Furthermore, standard prophylaxis requires lifelong intravenous infusions, sometimes several a week, said lead ATLAS-A/B investigator Alok Srivastava, MD, a hematologist at the Christian Medical College in Vellore, India.
Fitusiran was dosed in the studies as a once-a-month 80 mg subcutaneous injection, so is much less bothersome. Also, it seems likely that some patients will only need dosing every other month. Maker Sanofi Genzyme is exploring lower and less frequent dosing to reduce thrombotic event risks that emerged in earlier studies, said Steven Pipe, MD, a pediatric hematologist at the University of Michigan, Ann Arbor, and the senior investigator on ATLAS-A/B, which assessed fitusiran in patients without inhibitors.
Serious thrombotic events occurred in two fitusiran patients in the trials, one of which led to discontinuation.
No pricing information
Overall, “I think [fitusiran] is a tremendous leap forward” with “the opportunity to transform the day-to-day lives of patients,” particularly those with hemophilia B, who have limited treatment options, Dr. Pipe said.
If approved for the U.S. market, fitusiran will go up against the monoclonal antibody emicizumab (Hemlibra), a subcutaneous injection dosed weekly to monthly that mimics the function of factor VIII, so it’s approved only for hemophilia A with or without inhibitors.
Several audience members at ASH noted that a major consideration for fitusiran, if approved, will be its cost. There’s no pricing information yet, but annual list price for emicizumab is reported to be in the $500,000 range.
For hemophilia A, “it will come to what proves to be the most efficacious and safe, with also consideration given to pricing,” Nigel Key, MD, a hematologist at the University of North Carolina at Chapel Hill, said in a comment.
Trial details
In ATLAS-A/B, 80 male patients were randomized to prophylactic fitusiran once monthly and 40 to continue with clotting factors as needed for bleeding. Just over 20% had hemophilia B, the rest hemophilia A. The mean age was 34 years, and subjects had a mean of about 12 bleeds in the 6 months leading up to the study.
Half of the fitusiran group had no treated bleeds during the study period versus only 5% in the control arm.
The five treatment emergent serious adverse events in the fitusiran arm included cholelithiasis in two subjects, plus cholecystitis, lower respiratory tract infection, and asthma in one each. Two fitusiran patients discontinued treatment because of cholecystitis and increased alanine aminotransferase.
ATLAS-INH had the same study design, and investigated patients with inhibitors; again, just over 20% had hemophilia B, the rest A. Mean age was 28 years, and patients had a mean of about 13 bleeds over the 6 months before the study. A total of 38 subjects were randomized to fitusiran, and 19 to bypassing agents as needed.
Almost 66% of fitusiran patients had no treated bleeds versus about 5% in the control arm.
There “was far less bleeding both for hemophilia A and B. It’s definitely a clinically meaningful and important” effect, said ATLAS-INH lead investigator Guy Young, MD, director of the hemostasis and thrombosis program at Children’s Hospital Los Angeles.
He said the improved quality of life with fitusiran noted in both trials was “not surprising. If you only have to dose once a month subcutaneously, and you are not bleeding, of course your quality of life is going to improve.”
Both fitusiran thrombotic events occurred in ATLAS-INH. One patient developed deep vein thrombosis, subclavian vein thrombosis, and superficial thrombophlebitis but stayed in the study. Another discontinued after developing suspected spinal vessel thrombosis. Serious adverse events among five other subjects included acute cholecystitis and hematuria.
Fitusiran was associated with liver enzyme elevations in both trials, but they were generally mild to moderate.
The studies were funded by fitusiran maker Sanofi Genzyme. Several investigators were employees. Dr. Pipe is a consultant, Dr. Srivastava is a researcher and adviser, and Dr. Young is a speaker and consultant for the company and disclosed honoraria from it. Dr. Key had no relevant disclosures.
Fitusiran is a small interfering RNA molecule that blocks antithrombin production in liver cells. Instead of taking the traditional approach in hemophilia treatment of boosting the coagulation cascade by replacing what’s missing, the idea of fitusiran is to short circuit the body’s anticoagulation system by targeting antithrombin.
Patients in two trials presented at the meeting, ATLAS-A/B and ATLAS-INH, had about a 90% reduction in their annualized bleeding rates when treated with prophylactic fitusiran, with half or more having no bleeds that required treatment during the 9-month trials. The median annualized bleeding rate fell to 0, trial investigators reported at the meeting.
These findings held in both hemophilia A and B with and without inhibitors, which are antibodies formed against exogenous clotting factors, and on subanalysis of spontaneous and joint bleeding rates. Reduced bleeding was associated with substantial improvements in health-care related quality of life, particularly in the physical health domain.
A question about study design
An audience member at ASH noted that the trials didn’t compare fitusiran against prophylactic treatment, which is standard of care for hemophilia, but rather against episodic treatment – concentrated factors or bypassing agents in subjects with inhibitors – once subjects in the control groups started to bleed.
Still, the numbers reported in the studies “have never been achieved with standard prophylaxis in the past.” Furthermore, standard prophylaxis requires lifelong intravenous infusions, sometimes several a week, said lead ATLAS-A/B investigator Alok Srivastava, MD, a hematologist at the Christian Medical College in Vellore, India.
Fitusiran was dosed in the studies as a once-a-month 80 mg subcutaneous injection, so is much less bothersome. Also, it seems likely that some patients will only need dosing every other month. Maker Sanofi Genzyme is exploring lower and less frequent dosing to reduce thrombotic event risks that emerged in earlier studies, said Steven Pipe, MD, a pediatric hematologist at the University of Michigan, Ann Arbor, and the senior investigator on ATLAS-A/B, which assessed fitusiran in patients without inhibitors.
Serious thrombotic events occurred in two fitusiran patients in the trials, one of which led to discontinuation.
No pricing information
Overall, “I think [fitusiran] is a tremendous leap forward” with “the opportunity to transform the day-to-day lives of patients,” particularly those with hemophilia B, who have limited treatment options, Dr. Pipe said.
If approved for the U.S. market, fitusiran will go up against the monoclonal antibody emicizumab (Hemlibra), a subcutaneous injection dosed weekly to monthly that mimics the function of factor VIII, so it’s approved only for hemophilia A with or without inhibitors.
Several audience members at ASH noted that a major consideration for fitusiran, if approved, will be its cost. There’s no pricing information yet, but annual list price for emicizumab is reported to be in the $500,000 range.
For hemophilia A, “it will come to what proves to be the most efficacious and safe, with also consideration given to pricing,” Nigel Key, MD, a hematologist at the University of North Carolina at Chapel Hill, said in a comment.
Trial details
In ATLAS-A/B, 80 male patients were randomized to prophylactic fitusiran once monthly and 40 to continue with clotting factors as needed for bleeding. Just over 20% had hemophilia B, the rest hemophilia A. The mean age was 34 years, and subjects had a mean of about 12 bleeds in the 6 months leading up to the study.
Half of the fitusiran group had no treated bleeds during the study period versus only 5% in the control arm.
The five treatment emergent serious adverse events in the fitusiran arm included cholelithiasis in two subjects, plus cholecystitis, lower respiratory tract infection, and asthma in one each. Two fitusiran patients discontinued treatment because of cholecystitis and increased alanine aminotransferase.
ATLAS-INH had the same study design, and investigated patients with inhibitors; again, just over 20% had hemophilia B, the rest A. Mean age was 28 years, and patients had a mean of about 13 bleeds over the 6 months before the study. A total of 38 subjects were randomized to fitusiran, and 19 to bypassing agents as needed.
Almost 66% of fitusiran patients had no treated bleeds versus about 5% in the control arm.
There “was far less bleeding both for hemophilia A and B. It’s definitely a clinically meaningful and important” effect, said ATLAS-INH lead investigator Guy Young, MD, director of the hemostasis and thrombosis program at Children’s Hospital Los Angeles.
He said the improved quality of life with fitusiran noted in both trials was “not surprising. If you only have to dose once a month subcutaneously, and you are not bleeding, of course your quality of life is going to improve.”
Both fitusiran thrombotic events occurred in ATLAS-INH. One patient developed deep vein thrombosis, subclavian vein thrombosis, and superficial thrombophlebitis but stayed in the study. Another discontinued after developing suspected spinal vessel thrombosis. Serious adverse events among five other subjects included acute cholecystitis and hematuria.
Fitusiran was associated with liver enzyme elevations in both trials, but they were generally mild to moderate.
The studies were funded by fitusiran maker Sanofi Genzyme. Several investigators were employees. Dr. Pipe is a consultant, Dr. Srivastava is a researcher and adviser, and Dr. Young is a speaker and consultant for the company and disclosed honoraria from it. Dr. Key had no relevant disclosures.
FROM ASH 2021
Genetic tests prompt therapy adjustments in children with epilepsy
Physicians at a Boston hospital adjusted medical management for nearly three-quarters of patients with infantile- or childhood-onset epilepsy who were diagnosed with genetic epilepsy, researchers reported at the annual meeting of the American Epilepsy Society. The findings provide new insight into the usefulness of genetic tests in children with epilepsy of unknown cause.
“. Genetic testing should be included as part of the standard evaluation of individuals with unexplained pediatric epilepsy as a means of achieving diagnostic precision and informing clinical management,” study lead author Isabel Haviland, MD, a neurologist with Boston Children’s Hospital/Harvard Medical School, said in an interview.
According to Dr. Haviland, the causes of epilepsy are unexplained in an estimated two-thirds of pediatric epilepsy cases. “Increasingly, when genetic testing is available, previously unexplained cases of pediatric epilepsy are being found to have single-gene etiologies,” she said. “Though a genetic diagnosis in this population has implications for medical care, the direct impact on medical management in a clinical setting has not been measured. We aimed to describe the impact of genetic diagnosis on medical management in a cohort of individuals with pediatric epilepsy.”
Researchers tracked 602 patients at Boston Children’s Hospital who received next-generation gene sequencing testing from 2012 to 2019. Of those, Dr. Haviland said, 152 (25%) had a positive result that indicated genetic epilepsy (46% female, median age of onset = 6 months [2-15 months]). These patients were included in the study.
“We documented an impact on medical management in nearly three-fourths of participants (72%),” Dr. Haviland said. “A genetic diagnosis affected at least one of four categories of medical management, including care coordination (48%), treatment (45%), counseling about a change in prognosis (28%), and change in diagnosis for a few individuals who had a prior established diagnosis (1%).”
As examples, she mentioned three cases:
- Testing revealed that a subject has a disease-causing genetic variant in a gene called PRRT2. “This gene is involved in the release of neurotransmitters in the brain,” Dr. Haviland said. “Thanks to his diagnosis, he was treated with the antiseizure medication oxcarbazepine, which is often effective for epilepsy caused by variants in this gene. He had excellent response to the medication and later became seizure free.”
- A subject had a variation in the SCN1A gene that causes types of epilepsy. “At the time of his diagnosis, there was a trial for a medication called fenfluramine being offered for individuals with SCN1A variants, and his family elected to participate,” she said. “This medication was later approved by the [Food and Drug Administration] for SCN1A-related epilepsy.”
- Testing identified disease-causing variant in the GRIN2A gene in another subject. “This gene is involved in brain cell communication,” Dr. Haviland said. “This individual was treated with memantine, which acts on the specific biological pathway affected by the gene. This treatment would not have been considered without the genetic diagnosis as it is currently only approved for Alzheimer’s disease.”
In addition, Dr. Haviland said, researchers found that “there was impact on medical management both in those with earlier age of epilepsy onset (under 2 years) and those with later age of onset, as well as both in those with developmental disorders (such as autism spectrum disorder, intellectual disability and developmental delay) and those with normal development.
As for the cost of genetic tests, Dr. Haviland pointed to a 2019 study that she said estimated epilepsy panel testing runs from $1,500 to $7,500, and the whole exome sequencing from $4,500 to $7,000. “Insurers sometimes cover testing, but not always,” she said. “In some cases, insurance will only cover testing if it is documented that results will directly alter medical management, which highlights the importance of our findings.”
No study funding was reported. Dr. Haviland and several other authors report no disclosures. One author reports consulting fees from Takeda, Zogenix, Marinus, and FOXG1 Research Foundation. Another author reports research support from the International Foundation for CDKL5 Research.
Physicians at a Boston hospital adjusted medical management for nearly three-quarters of patients with infantile- or childhood-onset epilepsy who were diagnosed with genetic epilepsy, researchers reported at the annual meeting of the American Epilepsy Society. The findings provide new insight into the usefulness of genetic tests in children with epilepsy of unknown cause.
“. Genetic testing should be included as part of the standard evaluation of individuals with unexplained pediatric epilepsy as a means of achieving diagnostic precision and informing clinical management,” study lead author Isabel Haviland, MD, a neurologist with Boston Children’s Hospital/Harvard Medical School, said in an interview.
According to Dr. Haviland, the causes of epilepsy are unexplained in an estimated two-thirds of pediatric epilepsy cases. “Increasingly, when genetic testing is available, previously unexplained cases of pediatric epilepsy are being found to have single-gene etiologies,” she said. “Though a genetic diagnosis in this population has implications for medical care, the direct impact on medical management in a clinical setting has not been measured. We aimed to describe the impact of genetic diagnosis on medical management in a cohort of individuals with pediatric epilepsy.”
Researchers tracked 602 patients at Boston Children’s Hospital who received next-generation gene sequencing testing from 2012 to 2019. Of those, Dr. Haviland said, 152 (25%) had a positive result that indicated genetic epilepsy (46% female, median age of onset = 6 months [2-15 months]). These patients were included in the study.
“We documented an impact on medical management in nearly three-fourths of participants (72%),” Dr. Haviland said. “A genetic diagnosis affected at least one of four categories of medical management, including care coordination (48%), treatment (45%), counseling about a change in prognosis (28%), and change in diagnosis for a few individuals who had a prior established diagnosis (1%).”
As examples, she mentioned three cases:
- Testing revealed that a subject has a disease-causing genetic variant in a gene called PRRT2. “This gene is involved in the release of neurotransmitters in the brain,” Dr. Haviland said. “Thanks to his diagnosis, he was treated with the antiseizure medication oxcarbazepine, which is often effective for epilepsy caused by variants in this gene. He had excellent response to the medication and later became seizure free.”
- A subject had a variation in the SCN1A gene that causes types of epilepsy. “At the time of his diagnosis, there was a trial for a medication called fenfluramine being offered for individuals with SCN1A variants, and his family elected to participate,” she said. “This medication was later approved by the [Food and Drug Administration] for SCN1A-related epilepsy.”
- Testing identified disease-causing variant in the GRIN2A gene in another subject. “This gene is involved in brain cell communication,” Dr. Haviland said. “This individual was treated with memantine, which acts on the specific biological pathway affected by the gene. This treatment would not have been considered without the genetic diagnosis as it is currently only approved for Alzheimer’s disease.”
In addition, Dr. Haviland said, researchers found that “there was impact on medical management both in those with earlier age of epilepsy onset (under 2 years) and those with later age of onset, as well as both in those with developmental disorders (such as autism spectrum disorder, intellectual disability and developmental delay) and those with normal development.
As for the cost of genetic tests, Dr. Haviland pointed to a 2019 study that she said estimated epilepsy panel testing runs from $1,500 to $7,500, and the whole exome sequencing from $4,500 to $7,000. “Insurers sometimes cover testing, but not always,” she said. “In some cases, insurance will only cover testing if it is documented that results will directly alter medical management, which highlights the importance of our findings.”
No study funding was reported. Dr. Haviland and several other authors report no disclosures. One author reports consulting fees from Takeda, Zogenix, Marinus, and FOXG1 Research Foundation. Another author reports research support from the International Foundation for CDKL5 Research.
Physicians at a Boston hospital adjusted medical management for nearly three-quarters of patients with infantile- or childhood-onset epilepsy who were diagnosed with genetic epilepsy, researchers reported at the annual meeting of the American Epilepsy Society. The findings provide new insight into the usefulness of genetic tests in children with epilepsy of unknown cause.
“. Genetic testing should be included as part of the standard evaluation of individuals with unexplained pediatric epilepsy as a means of achieving diagnostic precision and informing clinical management,” study lead author Isabel Haviland, MD, a neurologist with Boston Children’s Hospital/Harvard Medical School, said in an interview.
According to Dr. Haviland, the causes of epilepsy are unexplained in an estimated two-thirds of pediatric epilepsy cases. “Increasingly, when genetic testing is available, previously unexplained cases of pediatric epilepsy are being found to have single-gene etiologies,” she said. “Though a genetic diagnosis in this population has implications for medical care, the direct impact on medical management in a clinical setting has not been measured. We aimed to describe the impact of genetic diagnosis on medical management in a cohort of individuals with pediatric epilepsy.”
Researchers tracked 602 patients at Boston Children’s Hospital who received next-generation gene sequencing testing from 2012 to 2019. Of those, Dr. Haviland said, 152 (25%) had a positive result that indicated genetic epilepsy (46% female, median age of onset = 6 months [2-15 months]). These patients were included in the study.
“We documented an impact on medical management in nearly three-fourths of participants (72%),” Dr. Haviland said. “A genetic diagnosis affected at least one of four categories of medical management, including care coordination (48%), treatment (45%), counseling about a change in prognosis (28%), and change in diagnosis for a few individuals who had a prior established diagnosis (1%).”
As examples, she mentioned three cases:
- Testing revealed that a subject has a disease-causing genetic variant in a gene called PRRT2. “This gene is involved in the release of neurotransmitters in the brain,” Dr. Haviland said. “Thanks to his diagnosis, he was treated with the antiseizure medication oxcarbazepine, which is often effective for epilepsy caused by variants in this gene. He had excellent response to the medication and later became seizure free.”
- A subject had a variation in the SCN1A gene that causes types of epilepsy. “At the time of his diagnosis, there was a trial for a medication called fenfluramine being offered for individuals with SCN1A variants, and his family elected to participate,” she said. “This medication was later approved by the [Food and Drug Administration] for SCN1A-related epilepsy.”
- Testing identified disease-causing variant in the GRIN2A gene in another subject. “This gene is involved in brain cell communication,” Dr. Haviland said. “This individual was treated with memantine, which acts on the specific biological pathway affected by the gene. This treatment would not have been considered without the genetic diagnosis as it is currently only approved for Alzheimer’s disease.”
In addition, Dr. Haviland said, researchers found that “there was impact on medical management both in those with earlier age of epilepsy onset (under 2 years) and those with later age of onset, as well as both in those with developmental disorders (such as autism spectrum disorder, intellectual disability and developmental delay) and those with normal development.
As for the cost of genetic tests, Dr. Haviland pointed to a 2019 study that she said estimated epilepsy panel testing runs from $1,500 to $7,500, and the whole exome sequencing from $4,500 to $7,000. “Insurers sometimes cover testing, but not always,” she said. “In some cases, insurance will only cover testing if it is documented that results will directly alter medical management, which highlights the importance of our findings.”
No study funding was reported. Dr. Haviland and several other authors report no disclosures. One author reports consulting fees from Takeda, Zogenix, Marinus, and FOXG1 Research Foundation. Another author reports research support from the International Foundation for CDKL5 Research.
FROM AES 2021
WPATH draft on gender dysphoria ‘skewed and misses urgent issues’
New draft guidance from the World Professional Association for Transgender Health (WPATH) is raising serious concerns among professionals caring for people with gender dysphoria, prompting claims that WPATH is an organization “captured by activists.”
Experts in adolescent and child psychology, as well as pediatric health, have expressed dismay that the WPATH Standards of Care (SOC) 8 appear to miss some of the most urgent issues in the field of transgender medicine and are considered to express a radical and unreserved leaning towards “gender-affirmation.”
The WPATH SOC 8 document is available for view and comment until Dec. 16 until 11.59 PM EST, after which time revisions will be made and the final version published.
Despite repeated attempts by this news organization to seek clarification on certain aspects of the guidance from members of the WPATH SOC 8 committee, requests were declined “until the guidance is finalized.”
According to the WPATH website, the SOC 8 aims to provide “clinical guidance for health professionals to assist transgender and gender diverse people with safe and effective pathways” to manage their gender dysphoria and potentially transition.
Such pathways may relate to primary care, gynecologic and urologic care, reproductive options, voice and communication therapy, mental health services, and hormonal or surgical treatments, among others.
WPATH adds that it was felt necessary to revise the existing SOC 7 (published in 2012) because of recent “globally unprecedented increase and visibility of transgender and gender-diverse people seeking support and gender-affirming medical treatment.”
Gender-affirming medical treatment means different things at different ages. In the case of kids with gender dysphoria who have not yet entered puberty associated with their birth sex, this might include prescribing so-called “puberty blockers” to delay natural puberty – gonadotrophin-releasing hormone analogs that are licensed for use in precocious puberty in children. Such agents have not been licensed for use in children with gender dysphoria, however, so any use for this purpose is off-label.
Following puberty blockade – or in cases where adolescents have already undergone natural puberty – the next step is to begin cross-sex hormones. So, for a female patient who wants to transition to male (FTM), that would be lifelong testosterone, and for a male who wants to be female (MTF), it involves lifelong estrogen. Again, use of such hormones in transgender individuals is entirely off-label.
Just last month, two of America’s leading experts on transgender medicine, both psychologists – including one who is transgender – told this news organization they were concerned that the quality of the evaluations of youth with gender dysphoria are being stifled by activists who are worried that open discussions will further stigmatize trans individuals.
They subsequently wrote an op-ed on the topic entitled, “The mental health establishment is failing trans kids,” which was finally published in the Washington Post on Nov. 24, after numerous other mainstream U.S. media outlets had rejected it.
New SOC 8 ‘is not evidence based,’ should not be new ‘gold standard’
One expert says the draft SOC 8 lacks balance and does not address certain issues, while paying undue attention to others that detract from real questions facing the field of transgender medicine, both in the United States and around the world.
Julia Mason, MD, is a pediatrician based in Gresham, Oregon, with a special interest in children and adolescents experiencing gender dysphoria. “The SOC 8 shows us that WPATH remains captured by activists,” she asserts.
Dr. Mason questions the integrity of WPATH based on what she has read in the draft SOC 8.
“We need a serious organization to take a sober look at the evidence, and that is why we have established the Society for Evidence-Based Gender Medicine [SEGM],” she noted. “This is what we do – we are looking at all of the evidence.”
Dr. Mason is a clinical advisor to SEGM, an organization set-up to evaluate current interventions and evidence on gender dysphoria.
The pediatrician has particular concerns regarding the child and adolescent chapters in the draft SOC 8. The adolescent chapter states: “Guidelines are meant to provide a gold standard based on the available evidence at this moment of time.”
Dr. Mason disputes this assertion. “This document should not be the new gold standard going forward, primarily because it is not evidence based.”
In an interview, Dr. Mason explained that WPATH say they used the “Delphi consensus process” to determine their recommendations, but “this process is designed for use with a panel of experts when evidence is lacking. I would say they didn’t have a panel of experts. They largely had a panel of activists, with a few experts.”
There is no mention, for example, of England’s National Institute for Health and Care Excellence (NICE) evidence reviews on puberty blockers and cross-sex hormones from earlier this year. These reviews determined that no studies have compared cross-sex hormones or puberty blockers with a control group and all follow-up periods for cross-sex hormones were relatively short.
This disappoints Dr. Mason: “These are significant; they are important documents.”
And much of the evidence quoted comes from the well-known and often-quoted “Dutch-protocol” study of 2011, in which the children studied were much younger at the time of their gender dysphoria, compared with the many adolescents who make up the current surge in presentation at gender clinics worldwide, she adds.
Rapid-onset GD: adolescents presenting late with little history
Dr. Mason also stresses that the SOC 8 does not address the most urgent issues in transgender medicine today, mainly because it does not address rapid-onset gender dysphoria (ROGD): “This is the dilemma of the 21st century; it’s new.”
ROGD – a term first coined in 2018 by researcher Lisa Littman, MD, MPH, now president of the Institute for Comprehensive Gender Dysphoria Research (ICGDR) – refers to the phenomena of adolescents expressing a desire to transition from their birth sex after little or no apparent previous indication.
However, the SOC 8 does make reference to aspects of adolescent development that might impact their decision-making processes around gender identity during teen years. The chapter on adolescents reads: “... adolescence is also often associated with increased risk-taking behaviors. Along with these notable changes ... individuation from parents ... [there is] often a heightened focus on peer relationships, which can be both positive and detrimental.”
The guidance goes on to point out that “it is critical to understand how all of these aspects of development may impact the decision-making for a given young person within their specific cultural context.”
Desistance and detransitioning not adequately addressed
Dr. Mason also says there is little mention “about detransitioning in this SOC [8], and ‘gender dysphoria’ and ‘trans’ are terms that are not defined.”
Likewise, there is no mention of desistance, she highlights, which is when individuals naturally resolve their dysphoria around their birth sex as they grow older.
The most recent published data seen by this news organization relates to a study from March 2021 that showed nearly 88% of boys who struggled with gender identity in childhood (approximate mean age 8 years and follow-up at approximate mean age 20 years) desisted. It reads: “Of the 139 participants, 17 (12.2%) were classified as ‘persisters’ and the remaining 122 (87.8%) were classified as desisters.”
“Most children with gender dysphoria will desist and lose their concept of themselves as being the opposite gender,” Dr. Mason explains. “This is the safest path for a child – desistance.”
“Transition can turn a healthy young person into a lifelong medical patient and has significant health risks,” she emphasizes, stressing that transition has not been shown to decrease the probability of suicide, or attempts at suicide, despite myriad claims saying otherwise.
“Before we were routinely transitioning kids at school, the vast majority of children grew out of their gender dysphoria. This history is not recognized at all in these SOC [8],” she maintains.
Ken Zucker, PhD, CPsych, an author of the study of desistance in boys, says the terms desistence and persistence of gender dysphoria have caused some consternation in certain circles.
An editor of the Archives of Sexual Behavior and professor in the department of psychiatry, University of Toronto, Dr. Zucker has published widely on the topic.
He told this news organization: “The terms persistence and desistance have become verboten among the WPATH cognoscenti. Perhaps the contributors to SOC 8 have come up with alternative descriptors.”
“The term ‘desistance’ is particularly annoying to some of the gender-affirming clinicians, because they don’t believe that desistance is bona fide,” Dr. Zucker points out.
“The desistance resisters are like anti-vaxxers – nothing one can provide as evidence for the efficacy of vaccines is sufficient. There will always be a new objection.”
Other mental health issues, in particular ADHD and autism
It is also widely acknowledged that there is a higher rate of neurodevelopmental and psychiatric diagnoses in individuals with gender dysphoria. For example, one 2020 study found that transgender people were three to six times as likely to be autistic as cisgender people (those whose gender is aligned with their birth sex).
Statement one in the chapter on adolescents in draft WPATH SOC 8 does give a nod to this, pointing out that health professionals working with gender diverse adolescents “should receive training and develop expertise in autism spectrum disorders and other neurodiversity conditions.”
It also notes that in some cases “a more extended assessment process may be useful, such as for youth with more complex presentations (e.g., complicated mental health histories, co-occurring autism spectrum characteristics in particular) and an absence of experienced childhood gender incongruence.”
However, Dr. Mason stresses that underlying mental health issues are central to addressing how to manage a significant number of these patients.
“If a young person has ADHD or autism, they are not ready to make decisions about the rest of their life at age 18. Even a neurotypical young person is still developing their frontal cortex in their early 20s, and it takes longer for those with ADHD or on the autism spectrum.”
She firmly believes that the guidance does not give sufficient consideration to comorbidities in people over the age of 18.
According to their [SOC 8] guidelines, “once someone is 18 they are ready for anything,” says Dr. Mason.
Offering some explanation for the increased prevalence of ADHD and autism in those with gender dysphoria, Dr. Mason notes that children can have “hyperfocus” and those with autism will fixate on a particular area of interest. “If a child is unhappy in their life, and this can be more likely if someone is neuro-atypical, then it is likely that the individual might go online and find this one solution [for example, a transgender identity] that seems to fix everything.”
Perceptions of femininity and masculinity can also be extra challenging for a child with autism, Dr. Mason says. “It is relatively easy for an autistic girl to feel like she should be a boy because the rules of femininity are composed of nonverbal, subtle behaviors that can be difficult to pick up on,” she points out. “An autistic child who isn’t particularly good at nonverbal communication might not pick up on these and thus feel they are not very ‘female.’”
“There’s a whole lot of grass-is-greener-type thinking. Girls think boys have an easier life, and boys think girls have an easier life. I know some detransitioners who have spoken eloquently about realizing their mistake on this,” she adds.
Other parts of the SOC 8 that Dr. Mason disagrees with include the recommendation in the adolescent chapter that 14-year-olds are mature enough to start cross-sex hormones, that is, giving testosterone to a female who wants to transition to male or estrogen to a male who wishes to transition to female. “I think that’s far too young,” she asserts.
And she points out that the document states 17-year-olds are ready for genital reassignment surgery. Again, she believes this is far too young.
“Also, the SOC 8 document does not clarify who is appropriate for surgery. Whenever surgery is discussed, it becomes very vague,” she said.
A version of this article first appeared on Medscape.com.
New draft guidance from the World Professional Association for Transgender Health (WPATH) is raising serious concerns among professionals caring for people with gender dysphoria, prompting claims that WPATH is an organization “captured by activists.”
Experts in adolescent and child psychology, as well as pediatric health, have expressed dismay that the WPATH Standards of Care (SOC) 8 appear to miss some of the most urgent issues in the field of transgender medicine and are considered to express a radical and unreserved leaning towards “gender-affirmation.”
The WPATH SOC 8 document is available for view and comment until Dec. 16 until 11.59 PM EST, after which time revisions will be made and the final version published.
Despite repeated attempts by this news organization to seek clarification on certain aspects of the guidance from members of the WPATH SOC 8 committee, requests were declined “until the guidance is finalized.”
According to the WPATH website, the SOC 8 aims to provide “clinical guidance for health professionals to assist transgender and gender diverse people with safe and effective pathways” to manage their gender dysphoria and potentially transition.
Such pathways may relate to primary care, gynecologic and urologic care, reproductive options, voice and communication therapy, mental health services, and hormonal or surgical treatments, among others.
WPATH adds that it was felt necessary to revise the existing SOC 7 (published in 2012) because of recent “globally unprecedented increase and visibility of transgender and gender-diverse people seeking support and gender-affirming medical treatment.”
Gender-affirming medical treatment means different things at different ages. In the case of kids with gender dysphoria who have not yet entered puberty associated with their birth sex, this might include prescribing so-called “puberty blockers” to delay natural puberty – gonadotrophin-releasing hormone analogs that are licensed for use in precocious puberty in children. Such agents have not been licensed for use in children with gender dysphoria, however, so any use for this purpose is off-label.
Following puberty blockade – or in cases where adolescents have already undergone natural puberty – the next step is to begin cross-sex hormones. So, for a female patient who wants to transition to male (FTM), that would be lifelong testosterone, and for a male who wants to be female (MTF), it involves lifelong estrogen. Again, use of such hormones in transgender individuals is entirely off-label.
Just last month, two of America’s leading experts on transgender medicine, both psychologists – including one who is transgender – told this news organization they were concerned that the quality of the evaluations of youth with gender dysphoria are being stifled by activists who are worried that open discussions will further stigmatize trans individuals.
They subsequently wrote an op-ed on the topic entitled, “The mental health establishment is failing trans kids,” which was finally published in the Washington Post on Nov. 24, after numerous other mainstream U.S. media outlets had rejected it.
New SOC 8 ‘is not evidence based,’ should not be new ‘gold standard’
One expert says the draft SOC 8 lacks balance and does not address certain issues, while paying undue attention to others that detract from real questions facing the field of transgender medicine, both in the United States and around the world.
Julia Mason, MD, is a pediatrician based in Gresham, Oregon, with a special interest in children and adolescents experiencing gender dysphoria. “The SOC 8 shows us that WPATH remains captured by activists,” she asserts.
Dr. Mason questions the integrity of WPATH based on what she has read in the draft SOC 8.
“We need a serious organization to take a sober look at the evidence, and that is why we have established the Society for Evidence-Based Gender Medicine [SEGM],” she noted. “This is what we do – we are looking at all of the evidence.”
Dr. Mason is a clinical advisor to SEGM, an organization set-up to evaluate current interventions and evidence on gender dysphoria.
The pediatrician has particular concerns regarding the child and adolescent chapters in the draft SOC 8. The adolescent chapter states: “Guidelines are meant to provide a gold standard based on the available evidence at this moment of time.”
Dr. Mason disputes this assertion. “This document should not be the new gold standard going forward, primarily because it is not evidence based.”
In an interview, Dr. Mason explained that WPATH say they used the “Delphi consensus process” to determine their recommendations, but “this process is designed for use with a panel of experts when evidence is lacking. I would say they didn’t have a panel of experts. They largely had a panel of activists, with a few experts.”
There is no mention, for example, of England’s National Institute for Health and Care Excellence (NICE) evidence reviews on puberty blockers and cross-sex hormones from earlier this year. These reviews determined that no studies have compared cross-sex hormones or puberty blockers with a control group and all follow-up periods for cross-sex hormones were relatively short.
This disappoints Dr. Mason: “These are significant; they are important documents.”
And much of the evidence quoted comes from the well-known and often-quoted “Dutch-protocol” study of 2011, in which the children studied were much younger at the time of their gender dysphoria, compared with the many adolescents who make up the current surge in presentation at gender clinics worldwide, she adds.
Rapid-onset GD: adolescents presenting late with little history
Dr. Mason also stresses that the SOC 8 does not address the most urgent issues in transgender medicine today, mainly because it does not address rapid-onset gender dysphoria (ROGD): “This is the dilemma of the 21st century; it’s new.”
ROGD – a term first coined in 2018 by researcher Lisa Littman, MD, MPH, now president of the Institute for Comprehensive Gender Dysphoria Research (ICGDR) – refers to the phenomena of adolescents expressing a desire to transition from their birth sex after little or no apparent previous indication.
However, the SOC 8 does make reference to aspects of adolescent development that might impact their decision-making processes around gender identity during teen years. The chapter on adolescents reads: “... adolescence is also often associated with increased risk-taking behaviors. Along with these notable changes ... individuation from parents ... [there is] often a heightened focus on peer relationships, which can be both positive and detrimental.”
The guidance goes on to point out that “it is critical to understand how all of these aspects of development may impact the decision-making for a given young person within their specific cultural context.”
Desistance and detransitioning not adequately addressed
Dr. Mason also says there is little mention “about detransitioning in this SOC [8], and ‘gender dysphoria’ and ‘trans’ are terms that are not defined.”
Likewise, there is no mention of desistance, she highlights, which is when individuals naturally resolve their dysphoria around their birth sex as they grow older.
The most recent published data seen by this news organization relates to a study from March 2021 that showed nearly 88% of boys who struggled with gender identity in childhood (approximate mean age 8 years and follow-up at approximate mean age 20 years) desisted. It reads: “Of the 139 participants, 17 (12.2%) were classified as ‘persisters’ and the remaining 122 (87.8%) were classified as desisters.”
“Most children with gender dysphoria will desist and lose their concept of themselves as being the opposite gender,” Dr. Mason explains. “This is the safest path for a child – desistance.”
“Transition can turn a healthy young person into a lifelong medical patient and has significant health risks,” she emphasizes, stressing that transition has not been shown to decrease the probability of suicide, or attempts at suicide, despite myriad claims saying otherwise.
“Before we were routinely transitioning kids at school, the vast majority of children grew out of their gender dysphoria. This history is not recognized at all in these SOC [8],” she maintains.
Ken Zucker, PhD, CPsych, an author of the study of desistance in boys, says the terms desistence and persistence of gender dysphoria have caused some consternation in certain circles.
An editor of the Archives of Sexual Behavior and professor in the department of psychiatry, University of Toronto, Dr. Zucker has published widely on the topic.
He told this news organization: “The terms persistence and desistance have become verboten among the WPATH cognoscenti. Perhaps the contributors to SOC 8 have come up with alternative descriptors.”
“The term ‘desistance’ is particularly annoying to some of the gender-affirming clinicians, because they don’t believe that desistance is bona fide,” Dr. Zucker points out.
“The desistance resisters are like anti-vaxxers – nothing one can provide as evidence for the efficacy of vaccines is sufficient. There will always be a new objection.”
Other mental health issues, in particular ADHD and autism
It is also widely acknowledged that there is a higher rate of neurodevelopmental and psychiatric diagnoses in individuals with gender dysphoria. For example, one 2020 study found that transgender people were three to six times as likely to be autistic as cisgender people (those whose gender is aligned with their birth sex).
Statement one in the chapter on adolescents in draft WPATH SOC 8 does give a nod to this, pointing out that health professionals working with gender diverse adolescents “should receive training and develop expertise in autism spectrum disorders and other neurodiversity conditions.”
It also notes that in some cases “a more extended assessment process may be useful, such as for youth with more complex presentations (e.g., complicated mental health histories, co-occurring autism spectrum characteristics in particular) and an absence of experienced childhood gender incongruence.”
However, Dr. Mason stresses that underlying mental health issues are central to addressing how to manage a significant number of these patients.
“If a young person has ADHD or autism, they are not ready to make decisions about the rest of their life at age 18. Even a neurotypical young person is still developing their frontal cortex in their early 20s, and it takes longer for those with ADHD or on the autism spectrum.”
She firmly believes that the guidance does not give sufficient consideration to comorbidities in people over the age of 18.
According to their [SOC 8] guidelines, “once someone is 18 they are ready for anything,” says Dr. Mason.
Offering some explanation for the increased prevalence of ADHD and autism in those with gender dysphoria, Dr. Mason notes that children can have “hyperfocus” and those with autism will fixate on a particular area of interest. “If a child is unhappy in their life, and this can be more likely if someone is neuro-atypical, then it is likely that the individual might go online and find this one solution [for example, a transgender identity] that seems to fix everything.”
Perceptions of femininity and masculinity can also be extra challenging for a child with autism, Dr. Mason says. “It is relatively easy for an autistic girl to feel like she should be a boy because the rules of femininity are composed of nonverbal, subtle behaviors that can be difficult to pick up on,” she points out. “An autistic child who isn’t particularly good at nonverbal communication might not pick up on these and thus feel they are not very ‘female.’”
“There’s a whole lot of grass-is-greener-type thinking. Girls think boys have an easier life, and boys think girls have an easier life. I know some detransitioners who have spoken eloquently about realizing their mistake on this,” she adds.
Other parts of the SOC 8 that Dr. Mason disagrees with include the recommendation in the adolescent chapter that 14-year-olds are mature enough to start cross-sex hormones, that is, giving testosterone to a female who wants to transition to male or estrogen to a male who wishes to transition to female. “I think that’s far too young,” she asserts.
And she points out that the document states 17-year-olds are ready for genital reassignment surgery. Again, she believes this is far too young.
“Also, the SOC 8 document does not clarify who is appropriate for surgery. Whenever surgery is discussed, it becomes very vague,” she said.
A version of this article first appeared on Medscape.com.
New draft guidance from the World Professional Association for Transgender Health (WPATH) is raising serious concerns among professionals caring for people with gender dysphoria, prompting claims that WPATH is an organization “captured by activists.”
Experts in adolescent and child psychology, as well as pediatric health, have expressed dismay that the WPATH Standards of Care (SOC) 8 appear to miss some of the most urgent issues in the field of transgender medicine and are considered to express a radical and unreserved leaning towards “gender-affirmation.”
The WPATH SOC 8 document is available for view and comment until Dec. 16 until 11.59 PM EST, after which time revisions will be made and the final version published.
Despite repeated attempts by this news organization to seek clarification on certain aspects of the guidance from members of the WPATH SOC 8 committee, requests were declined “until the guidance is finalized.”
According to the WPATH website, the SOC 8 aims to provide “clinical guidance for health professionals to assist transgender and gender diverse people with safe and effective pathways” to manage their gender dysphoria and potentially transition.
Such pathways may relate to primary care, gynecologic and urologic care, reproductive options, voice and communication therapy, mental health services, and hormonal or surgical treatments, among others.
WPATH adds that it was felt necessary to revise the existing SOC 7 (published in 2012) because of recent “globally unprecedented increase and visibility of transgender and gender-diverse people seeking support and gender-affirming medical treatment.”
Gender-affirming medical treatment means different things at different ages. In the case of kids with gender dysphoria who have not yet entered puberty associated with their birth sex, this might include prescribing so-called “puberty blockers” to delay natural puberty – gonadotrophin-releasing hormone analogs that are licensed for use in precocious puberty in children. Such agents have not been licensed for use in children with gender dysphoria, however, so any use for this purpose is off-label.
Following puberty blockade – or in cases where adolescents have already undergone natural puberty – the next step is to begin cross-sex hormones. So, for a female patient who wants to transition to male (FTM), that would be lifelong testosterone, and for a male who wants to be female (MTF), it involves lifelong estrogen. Again, use of such hormones in transgender individuals is entirely off-label.
Just last month, two of America’s leading experts on transgender medicine, both psychologists – including one who is transgender – told this news organization they were concerned that the quality of the evaluations of youth with gender dysphoria are being stifled by activists who are worried that open discussions will further stigmatize trans individuals.
They subsequently wrote an op-ed on the topic entitled, “The mental health establishment is failing trans kids,” which was finally published in the Washington Post on Nov. 24, after numerous other mainstream U.S. media outlets had rejected it.
New SOC 8 ‘is not evidence based,’ should not be new ‘gold standard’
One expert says the draft SOC 8 lacks balance and does not address certain issues, while paying undue attention to others that detract from real questions facing the field of transgender medicine, both in the United States and around the world.
Julia Mason, MD, is a pediatrician based in Gresham, Oregon, with a special interest in children and adolescents experiencing gender dysphoria. “The SOC 8 shows us that WPATH remains captured by activists,” she asserts.
Dr. Mason questions the integrity of WPATH based on what she has read in the draft SOC 8.
“We need a serious organization to take a sober look at the evidence, and that is why we have established the Society for Evidence-Based Gender Medicine [SEGM],” she noted. “This is what we do – we are looking at all of the evidence.”
Dr. Mason is a clinical advisor to SEGM, an organization set-up to evaluate current interventions and evidence on gender dysphoria.
The pediatrician has particular concerns regarding the child and adolescent chapters in the draft SOC 8. The adolescent chapter states: “Guidelines are meant to provide a gold standard based on the available evidence at this moment of time.”
Dr. Mason disputes this assertion. “This document should not be the new gold standard going forward, primarily because it is not evidence based.”
In an interview, Dr. Mason explained that WPATH say they used the “Delphi consensus process” to determine their recommendations, but “this process is designed for use with a panel of experts when evidence is lacking. I would say they didn’t have a panel of experts. They largely had a panel of activists, with a few experts.”
There is no mention, for example, of England’s National Institute for Health and Care Excellence (NICE) evidence reviews on puberty blockers and cross-sex hormones from earlier this year. These reviews determined that no studies have compared cross-sex hormones or puberty blockers with a control group and all follow-up periods for cross-sex hormones were relatively short.
This disappoints Dr. Mason: “These are significant; they are important documents.”
And much of the evidence quoted comes from the well-known and often-quoted “Dutch-protocol” study of 2011, in which the children studied were much younger at the time of their gender dysphoria, compared with the many adolescents who make up the current surge in presentation at gender clinics worldwide, she adds.
Rapid-onset GD: adolescents presenting late with little history
Dr. Mason also stresses that the SOC 8 does not address the most urgent issues in transgender medicine today, mainly because it does not address rapid-onset gender dysphoria (ROGD): “This is the dilemma of the 21st century; it’s new.”
ROGD – a term first coined in 2018 by researcher Lisa Littman, MD, MPH, now president of the Institute for Comprehensive Gender Dysphoria Research (ICGDR) – refers to the phenomena of adolescents expressing a desire to transition from their birth sex after little or no apparent previous indication.
However, the SOC 8 does make reference to aspects of adolescent development that might impact their decision-making processes around gender identity during teen years. The chapter on adolescents reads: “... adolescence is also often associated with increased risk-taking behaviors. Along with these notable changes ... individuation from parents ... [there is] often a heightened focus on peer relationships, which can be both positive and detrimental.”
The guidance goes on to point out that “it is critical to understand how all of these aspects of development may impact the decision-making for a given young person within their specific cultural context.”
Desistance and detransitioning not adequately addressed
Dr. Mason also says there is little mention “about detransitioning in this SOC [8], and ‘gender dysphoria’ and ‘trans’ are terms that are not defined.”
Likewise, there is no mention of desistance, she highlights, which is when individuals naturally resolve their dysphoria around their birth sex as they grow older.
The most recent published data seen by this news organization relates to a study from March 2021 that showed nearly 88% of boys who struggled with gender identity in childhood (approximate mean age 8 years and follow-up at approximate mean age 20 years) desisted. It reads: “Of the 139 participants, 17 (12.2%) were classified as ‘persisters’ and the remaining 122 (87.8%) were classified as desisters.”
“Most children with gender dysphoria will desist and lose their concept of themselves as being the opposite gender,” Dr. Mason explains. “This is the safest path for a child – desistance.”
“Transition can turn a healthy young person into a lifelong medical patient and has significant health risks,” she emphasizes, stressing that transition has not been shown to decrease the probability of suicide, or attempts at suicide, despite myriad claims saying otherwise.
“Before we were routinely transitioning kids at school, the vast majority of children grew out of their gender dysphoria. This history is not recognized at all in these SOC [8],” she maintains.
Ken Zucker, PhD, CPsych, an author of the study of desistance in boys, says the terms desistence and persistence of gender dysphoria have caused some consternation in certain circles.
An editor of the Archives of Sexual Behavior and professor in the department of psychiatry, University of Toronto, Dr. Zucker has published widely on the topic.
He told this news organization: “The terms persistence and desistance have become verboten among the WPATH cognoscenti. Perhaps the contributors to SOC 8 have come up with alternative descriptors.”
“The term ‘desistance’ is particularly annoying to some of the gender-affirming clinicians, because they don’t believe that desistance is bona fide,” Dr. Zucker points out.
“The desistance resisters are like anti-vaxxers – nothing one can provide as evidence for the efficacy of vaccines is sufficient. There will always be a new objection.”
Other mental health issues, in particular ADHD and autism
It is also widely acknowledged that there is a higher rate of neurodevelopmental and psychiatric diagnoses in individuals with gender dysphoria. For example, one 2020 study found that transgender people were three to six times as likely to be autistic as cisgender people (those whose gender is aligned with their birth sex).
Statement one in the chapter on adolescents in draft WPATH SOC 8 does give a nod to this, pointing out that health professionals working with gender diverse adolescents “should receive training and develop expertise in autism spectrum disorders and other neurodiversity conditions.”
It also notes that in some cases “a more extended assessment process may be useful, such as for youth with more complex presentations (e.g., complicated mental health histories, co-occurring autism spectrum characteristics in particular) and an absence of experienced childhood gender incongruence.”
However, Dr. Mason stresses that underlying mental health issues are central to addressing how to manage a significant number of these patients.
“If a young person has ADHD or autism, they are not ready to make decisions about the rest of their life at age 18. Even a neurotypical young person is still developing their frontal cortex in their early 20s, and it takes longer for those with ADHD or on the autism spectrum.”
She firmly believes that the guidance does not give sufficient consideration to comorbidities in people over the age of 18.
According to their [SOC 8] guidelines, “once someone is 18 they are ready for anything,” says Dr. Mason.
Offering some explanation for the increased prevalence of ADHD and autism in those with gender dysphoria, Dr. Mason notes that children can have “hyperfocus” and those with autism will fixate on a particular area of interest. “If a child is unhappy in their life, and this can be more likely if someone is neuro-atypical, then it is likely that the individual might go online and find this one solution [for example, a transgender identity] that seems to fix everything.”
Perceptions of femininity and masculinity can also be extra challenging for a child with autism, Dr. Mason says. “It is relatively easy for an autistic girl to feel like she should be a boy because the rules of femininity are composed of nonverbal, subtle behaviors that can be difficult to pick up on,” she points out. “An autistic child who isn’t particularly good at nonverbal communication might not pick up on these and thus feel they are not very ‘female.’”
“There’s a whole lot of grass-is-greener-type thinking. Girls think boys have an easier life, and boys think girls have an easier life. I know some detransitioners who have spoken eloquently about realizing their mistake on this,” she adds.
Other parts of the SOC 8 that Dr. Mason disagrees with include the recommendation in the adolescent chapter that 14-year-olds are mature enough to start cross-sex hormones, that is, giving testosterone to a female who wants to transition to male or estrogen to a male who wishes to transition to female. “I think that’s far too young,” she asserts.
And she points out that the document states 17-year-olds are ready for genital reassignment surgery. Again, she believes this is far too young.
“Also, the SOC 8 document does not clarify who is appropriate for surgery. Whenever surgery is discussed, it becomes very vague,” she said.
A version of this article first appeared on Medscape.com.






