User login
Serum brodalumab levels linked with treatment outcomes in patients with psoriasis
In a study of patients with psoriasis who had previously failed treatment with interleukin-17 receptor A inhibitor therapy, “all patients with quantifiable levels of brodalumab after 12 weeks of therapy experienced PASI reductions” and subquantifiable brodalumab levels were associated with a lack of response after 12 weeks, they wrote in JAMA Dermatology.
Lead study author Christian Enevold, PhD, a researcher at the Institute for Inflammation Research at Copenhagen University Hospital, and colleagues monitored patients with plaque psoriasis who had not improved with previous IL-17A inhibitor therapy, to evaluate whether trough levels and antidrug antibodies were associated with clinical response in this group of patients.
The 20 consecutive adult patients were treated at two academic hospital dermatology clinics between 2018 and 2020 and ranged in age from 19 to 66 years; 13 were male. At baseline, their weight ranged from 59 to 182 kg (median, 103 kg), their body mass index (BMI) ranged from 20 to 50 (median, 32), and their Psoriasis Area and Severity Index (PASI) scores ranged from 7 to 26 (median, 13). All had failed treatment with at least one IL-17A inhibitor, and 90% had failed treatment with at least one tumor necrosis factor–alpha or IL-12/-23 inhibitor.
Patients stopped taking systemic psoriasis therapies for 4 weeks before entering the study, then received subcutaneous injections of 210 mg of the IL-17A inhibitor brodalumab (Siliq) at weeks 0, 1, 2, and every 2 weeks thereafter. Patients whose PASI scores did not improve at least 75% from baseline (PASI 75) after 12 weeks of brodalumab discontinued treatment and left the study, while those who maintained PASI 75 were monitored for up to 52 weeks.
The researchers used assays to compare decreases in PASI score with brodalumab levels as well as with antibrodalumab antibodies at 12 weeks, and determined the following:
- Participants with quantifiable brodalumab levels (≥ 0.05 mcg/mL) showed a greater drop in PASI scores (median, 93%; range, 61%-100%) than those without quantifiable brodalumab levels (median, −3; range, −49% to 94%) (P = .006).
- Four of 5 patients (80%) who did not achieve a PASI 75, compared with 3 of 14 PASI 75 responders (21%), had drug levels too low to be measured (< 0.05 mcg/mL).
- The eight patients who did not have obesity (BMI < 30) had PASI reductions of at least 77%, and seven of the eight patients (88%) had quantifiable brodalumab levels.
- Six of the 12 patients with obesity (BMI ≥ 30) had brodalumab levels too low to be measured. Of those, four had increased PASI after 12 weeks of treatment. For all patients with obesity with quantifiable brodalumab levels, PASI scores dropped by at least 61% after 12 weeks.
- Five of the 12 (42%) patients with obesity versus 7 of the 8 (88%) patients without obesity had quantifiable brodalumab levels.
- None of the seven patients (35%) with subquantifiable drug levels after 12 weeks remained PASI responders.
- No antibrodalumab antibodies were detected in any serum samples.
The authors acknowledged that there were limitations of the study, including its retrospective design and restriction to the few available participants with a history of treatment failure.
George Han, MD, PhD, associate professor of dermatology at Hofstra University, Hempstead, N.Y., said in an interview that he found the study interesting. “The authors did an admirable job looking at many factors to try to understand response to treatment in a challenging population of patients who had failed at least one, and in many cases, numerous, biologics from different classes.”
“The most interesting finding is that patients with higher BMIs had much higher rates of low-to-undetectable drug concentration,” said Dr. Han, who was not involved in the study. “This very practical finding could help patient care immediately. While it’s impractical to start performing assays of drug concentration in clinical practice, this finding certainly would guide my conversations with my heavier-set patients who have had multiple failures on previous biologics.
“I’m looking forward to further studies that explore this issue and provide better evidence-based guidance for treating patients who have experienced multi-biologic failure,” he added.
Robert A. Dorschner, MD, assistant professor of dermatology at the UC San Diego Health System, also welcomed the study’s results.
“Current psoriasis treatment is based on trial-and-error application of various biologics targeting different pathways, with initial selection frequently based on insurance preference, not patient characteristics,” he said in an interview.
“Studies like this help clinicians make more informed decisions about whether a patient may benefit from a different dose or may require a different drug, and make those decisions earlier in therapy,” he said. “This can improve patient care and decrease costs associated with prolonged treatments with ineffective drugs.”
But Dr. Dorschner, who also was not involved in the study, cautions clinicians to not draw conclusions about dose adjustments from these results. “These findings need to be verified in a larger cohort,” he advised, “and they should drive future studies with larger cohorts and prospective designs.”
“The last couple of decades have seen an explosion in the availability of biologics targeting different cytokines, with significant benefits to patients,” Dr. Dorschner explained. “However, there is a dearth of information on how to choose the right biologic for a particular patient and how to assess the benefit of dose alteration versus changing the drug target. Medicine needs more studies like this one.”
Several authors of the study report financial relationships with LEO Pharma and other pharmaceutical companies. Most authors, including Dr. Enevold, reported no relevant financial relationships. Dr. Dorschner reported no relevant financial relationships. Dr. Han reported financial relationships with pharmaceutical companies not involved in the study. The study was funded by LEO Pharma and the Danish Biotechnology Program.
In a study of patients with psoriasis who had previously failed treatment with interleukin-17 receptor A inhibitor therapy, “all patients with quantifiable levels of brodalumab after 12 weeks of therapy experienced PASI reductions” and subquantifiable brodalumab levels were associated with a lack of response after 12 weeks, they wrote in JAMA Dermatology.
Lead study author Christian Enevold, PhD, a researcher at the Institute for Inflammation Research at Copenhagen University Hospital, and colleagues monitored patients with plaque psoriasis who had not improved with previous IL-17A inhibitor therapy, to evaluate whether trough levels and antidrug antibodies were associated with clinical response in this group of patients.
The 20 consecutive adult patients were treated at two academic hospital dermatology clinics between 2018 and 2020 and ranged in age from 19 to 66 years; 13 were male. At baseline, their weight ranged from 59 to 182 kg (median, 103 kg), their body mass index (BMI) ranged from 20 to 50 (median, 32), and their Psoriasis Area and Severity Index (PASI) scores ranged from 7 to 26 (median, 13). All had failed treatment with at least one IL-17A inhibitor, and 90% had failed treatment with at least one tumor necrosis factor–alpha or IL-12/-23 inhibitor.
Patients stopped taking systemic psoriasis therapies for 4 weeks before entering the study, then received subcutaneous injections of 210 mg of the IL-17A inhibitor brodalumab (Siliq) at weeks 0, 1, 2, and every 2 weeks thereafter. Patients whose PASI scores did not improve at least 75% from baseline (PASI 75) after 12 weeks of brodalumab discontinued treatment and left the study, while those who maintained PASI 75 were monitored for up to 52 weeks.
The researchers used assays to compare decreases in PASI score with brodalumab levels as well as with antibrodalumab antibodies at 12 weeks, and determined the following:
- Participants with quantifiable brodalumab levels (≥ 0.05 mcg/mL) showed a greater drop in PASI scores (median, 93%; range, 61%-100%) than those without quantifiable brodalumab levels (median, −3; range, −49% to 94%) (P = .006).
- Four of 5 patients (80%) who did not achieve a PASI 75, compared with 3 of 14 PASI 75 responders (21%), had drug levels too low to be measured (< 0.05 mcg/mL).
- The eight patients who did not have obesity (BMI < 30) had PASI reductions of at least 77%, and seven of the eight patients (88%) had quantifiable brodalumab levels.
- Six of the 12 patients with obesity (BMI ≥ 30) had brodalumab levels too low to be measured. Of those, four had increased PASI after 12 weeks of treatment. For all patients with obesity with quantifiable brodalumab levels, PASI scores dropped by at least 61% after 12 weeks.
- Five of the 12 (42%) patients with obesity versus 7 of the 8 (88%) patients without obesity had quantifiable brodalumab levels.
- None of the seven patients (35%) with subquantifiable drug levels after 12 weeks remained PASI responders.
- No antibrodalumab antibodies were detected in any serum samples.
The authors acknowledged that there were limitations of the study, including its retrospective design and restriction to the few available participants with a history of treatment failure.
George Han, MD, PhD, associate professor of dermatology at Hofstra University, Hempstead, N.Y., said in an interview that he found the study interesting. “The authors did an admirable job looking at many factors to try to understand response to treatment in a challenging population of patients who had failed at least one, and in many cases, numerous, biologics from different classes.”
“The most interesting finding is that patients with higher BMIs had much higher rates of low-to-undetectable drug concentration,” said Dr. Han, who was not involved in the study. “This very practical finding could help patient care immediately. While it’s impractical to start performing assays of drug concentration in clinical practice, this finding certainly would guide my conversations with my heavier-set patients who have had multiple failures on previous biologics.
“I’m looking forward to further studies that explore this issue and provide better evidence-based guidance for treating patients who have experienced multi-biologic failure,” he added.
Robert A. Dorschner, MD, assistant professor of dermatology at the UC San Diego Health System, also welcomed the study’s results.
“Current psoriasis treatment is based on trial-and-error application of various biologics targeting different pathways, with initial selection frequently based on insurance preference, not patient characteristics,” he said in an interview.
“Studies like this help clinicians make more informed decisions about whether a patient may benefit from a different dose or may require a different drug, and make those decisions earlier in therapy,” he said. “This can improve patient care and decrease costs associated with prolonged treatments with ineffective drugs.”
But Dr. Dorschner, who also was not involved in the study, cautions clinicians to not draw conclusions about dose adjustments from these results. “These findings need to be verified in a larger cohort,” he advised, “and they should drive future studies with larger cohorts and prospective designs.”
“The last couple of decades have seen an explosion in the availability of biologics targeting different cytokines, with significant benefits to patients,” Dr. Dorschner explained. “However, there is a dearth of information on how to choose the right biologic for a particular patient and how to assess the benefit of dose alteration versus changing the drug target. Medicine needs more studies like this one.”
Several authors of the study report financial relationships with LEO Pharma and other pharmaceutical companies. Most authors, including Dr. Enevold, reported no relevant financial relationships. Dr. Dorschner reported no relevant financial relationships. Dr. Han reported financial relationships with pharmaceutical companies not involved in the study. The study was funded by LEO Pharma and the Danish Biotechnology Program.
In a study of patients with psoriasis who had previously failed treatment with interleukin-17 receptor A inhibitor therapy, “all patients with quantifiable levels of brodalumab after 12 weeks of therapy experienced PASI reductions” and subquantifiable brodalumab levels were associated with a lack of response after 12 weeks, they wrote in JAMA Dermatology.
Lead study author Christian Enevold, PhD, a researcher at the Institute for Inflammation Research at Copenhagen University Hospital, and colleagues monitored patients with plaque psoriasis who had not improved with previous IL-17A inhibitor therapy, to evaluate whether trough levels and antidrug antibodies were associated with clinical response in this group of patients.
The 20 consecutive adult patients were treated at two academic hospital dermatology clinics between 2018 and 2020 and ranged in age from 19 to 66 years; 13 were male. At baseline, their weight ranged from 59 to 182 kg (median, 103 kg), their body mass index (BMI) ranged from 20 to 50 (median, 32), and their Psoriasis Area and Severity Index (PASI) scores ranged from 7 to 26 (median, 13). All had failed treatment with at least one IL-17A inhibitor, and 90% had failed treatment with at least one tumor necrosis factor–alpha or IL-12/-23 inhibitor.
Patients stopped taking systemic psoriasis therapies for 4 weeks before entering the study, then received subcutaneous injections of 210 mg of the IL-17A inhibitor brodalumab (Siliq) at weeks 0, 1, 2, and every 2 weeks thereafter. Patients whose PASI scores did not improve at least 75% from baseline (PASI 75) after 12 weeks of brodalumab discontinued treatment and left the study, while those who maintained PASI 75 were monitored for up to 52 weeks.
The researchers used assays to compare decreases in PASI score with brodalumab levels as well as with antibrodalumab antibodies at 12 weeks, and determined the following:
- Participants with quantifiable brodalumab levels (≥ 0.05 mcg/mL) showed a greater drop in PASI scores (median, 93%; range, 61%-100%) than those without quantifiable brodalumab levels (median, −3; range, −49% to 94%) (P = .006).
- Four of 5 patients (80%) who did not achieve a PASI 75, compared with 3 of 14 PASI 75 responders (21%), had drug levels too low to be measured (< 0.05 mcg/mL).
- The eight patients who did not have obesity (BMI < 30) had PASI reductions of at least 77%, and seven of the eight patients (88%) had quantifiable brodalumab levels.
- Six of the 12 patients with obesity (BMI ≥ 30) had brodalumab levels too low to be measured. Of those, four had increased PASI after 12 weeks of treatment. For all patients with obesity with quantifiable brodalumab levels, PASI scores dropped by at least 61% after 12 weeks.
- Five of the 12 (42%) patients with obesity versus 7 of the 8 (88%) patients without obesity had quantifiable brodalumab levels.
- None of the seven patients (35%) with subquantifiable drug levels after 12 weeks remained PASI responders.
- No antibrodalumab antibodies were detected in any serum samples.
The authors acknowledged that there were limitations of the study, including its retrospective design and restriction to the few available participants with a history of treatment failure.
George Han, MD, PhD, associate professor of dermatology at Hofstra University, Hempstead, N.Y., said in an interview that he found the study interesting. “The authors did an admirable job looking at many factors to try to understand response to treatment in a challenging population of patients who had failed at least one, and in many cases, numerous, biologics from different classes.”
“The most interesting finding is that patients with higher BMIs had much higher rates of low-to-undetectable drug concentration,” said Dr. Han, who was not involved in the study. “This very practical finding could help patient care immediately. While it’s impractical to start performing assays of drug concentration in clinical practice, this finding certainly would guide my conversations with my heavier-set patients who have had multiple failures on previous biologics.
“I’m looking forward to further studies that explore this issue and provide better evidence-based guidance for treating patients who have experienced multi-biologic failure,” he added.
Robert A. Dorschner, MD, assistant professor of dermatology at the UC San Diego Health System, also welcomed the study’s results.
“Current psoriasis treatment is based on trial-and-error application of various biologics targeting different pathways, with initial selection frequently based on insurance preference, not patient characteristics,” he said in an interview.
“Studies like this help clinicians make more informed decisions about whether a patient may benefit from a different dose or may require a different drug, and make those decisions earlier in therapy,” he said. “This can improve patient care and decrease costs associated with prolonged treatments with ineffective drugs.”
But Dr. Dorschner, who also was not involved in the study, cautions clinicians to not draw conclusions about dose adjustments from these results. “These findings need to be verified in a larger cohort,” he advised, “and they should drive future studies with larger cohorts and prospective designs.”
“The last couple of decades have seen an explosion in the availability of biologics targeting different cytokines, with significant benefits to patients,” Dr. Dorschner explained. “However, there is a dearth of information on how to choose the right biologic for a particular patient and how to assess the benefit of dose alteration versus changing the drug target. Medicine needs more studies like this one.”
Several authors of the study report financial relationships with LEO Pharma and other pharmaceutical companies. Most authors, including Dr. Enevold, reported no relevant financial relationships. Dr. Dorschner reported no relevant financial relationships. Dr. Han reported financial relationships with pharmaceutical companies not involved in the study. The study was funded by LEO Pharma and the Danish Biotechnology Program.
FROM JAMA DERMATOLOGY
FDA approves topical tapinarof for plaque psoriasis
The the manufacturer announced.
Tapinarof is an aryl hydrocarbon receptor agonist and is the first FDA-approved steroid-free topical medication in this class, according to a press release from the manufacturer, Dermavant.
Approval was based on results of three studies in a phase 3 clinical trial program (PSOARING 1, PSOARING 2), and an open-label extension study, (PSOARING 3), the company release said. In PSOARING 1 and 2, approximately 1,000 adults aged 18-75 years (median age, 51 years) with plaque psoriasis were randomized to once-daily topical tapinarof or placebo for up to 12 weeks; 85% were White and 57% were men. The study findings were published in the New England Journal of Medicine in December 2021.
The primary endpoint for both trials was the proportion of patients who achieved Physician Global Assessment (PGA) scores score of “clear” (0) or “almost clear” (1) and improvement of at least two grades from baseline.
After 12 weeks, 36% of the patients in PSOARING 1 and 40% in PSOARING 2 who received tapinarof met the primary outcome, compared with 6% of patients on placebo (P < .001 for both studies). Of these, a total of 73 patients from both studies who achieved PGA scores of 0 were entered in PSOARING 3, a 40-week open-label extension study, in which they stopped tapinarof treatment and retained PGA scores of 0 or 1 for approximately 4 months off treatment. An additional 312 patients who were enrolled in the PSOARING 3 extension study achieved PGA scores of 0 at least once during the study period, with “remittive” effects lasting a mean of 130 days off of treatment.
In addition, patients who received tapinarof in the PSOARING 1 and 2 studies showed significant improvement from baseline, compared with patients on placebo, across a range of secondary endpoints including a 75% or greater improvement in Psoriasis Area and Severity Index score (PASI 75).
In PSOARING 1, and 2, respectively, 36.1% and 47.6% of those on tapinarof achieved a PASI 75 response at week 12, compared with 10.2% and 6.9% of those on the vehicle (P < .001 for both).
Across all three studies, the majority adverse events were mild to moderate, and limited to the application site.
The most common adverse events reported by patients in the tapinarof groups were folliculitis, nasopharyngitis, and contact dermatitis. Headaches were more common among those treated with tapinarof than those on vehicle in the studies (3.8% vs. 2.4% in PSOARING 1, and 3.8% vs. 0.6% in PSOARING 2), leading to only three treatment discontinuations.
At the end of the PSOARING 3 study (at either week 40 or early termination), 599 participants responded to satisfaction questionnaires. Of these, 83.6% said they were satisfied with the results of tapinarof treatment, and 81.7% said it was more effective than previous topical treatments they had used, according to the company’s release.
Tapinarof cream can be used on all areas of the body, including the face, skin folds, neck, genitalia, anal crux, inflammatory areas, and axillae, according to the company release.
Full prescribing information is available here.
The the manufacturer announced.
Tapinarof is an aryl hydrocarbon receptor agonist and is the first FDA-approved steroid-free topical medication in this class, according to a press release from the manufacturer, Dermavant.
Approval was based on results of three studies in a phase 3 clinical trial program (PSOARING 1, PSOARING 2), and an open-label extension study, (PSOARING 3), the company release said. In PSOARING 1 and 2, approximately 1,000 adults aged 18-75 years (median age, 51 years) with plaque psoriasis were randomized to once-daily topical tapinarof or placebo for up to 12 weeks; 85% were White and 57% were men. The study findings were published in the New England Journal of Medicine in December 2021.
The primary endpoint for both trials was the proportion of patients who achieved Physician Global Assessment (PGA) scores score of “clear” (0) or “almost clear” (1) and improvement of at least two grades from baseline.
After 12 weeks, 36% of the patients in PSOARING 1 and 40% in PSOARING 2 who received tapinarof met the primary outcome, compared with 6% of patients on placebo (P < .001 for both studies). Of these, a total of 73 patients from both studies who achieved PGA scores of 0 were entered in PSOARING 3, a 40-week open-label extension study, in which they stopped tapinarof treatment and retained PGA scores of 0 or 1 for approximately 4 months off treatment. An additional 312 patients who were enrolled in the PSOARING 3 extension study achieved PGA scores of 0 at least once during the study period, with “remittive” effects lasting a mean of 130 days off of treatment.
In addition, patients who received tapinarof in the PSOARING 1 and 2 studies showed significant improvement from baseline, compared with patients on placebo, across a range of secondary endpoints including a 75% or greater improvement in Psoriasis Area and Severity Index score (PASI 75).
In PSOARING 1, and 2, respectively, 36.1% and 47.6% of those on tapinarof achieved a PASI 75 response at week 12, compared with 10.2% and 6.9% of those on the vehicle (P < .001 for both).
Across all three studies, the majority adverse events were mild to moderate, and limited to the application site.
The most common adverse events reported by patients in the tapinarof groups were folliculitis, nasopharyngitis, and contact dermatitis. Headaches were more common among those treated with tapinarof than those on vehicle in the studies (3.8% vs. 2.4% in PSOARING 1, and 3.8% vs. 0.6% in PSOARING 2), leading to only three treatment discontinuations.
At the end of the PSOARING 3 study (at either week 40 or early termination), 599 participants responded to satisfaction questionnaires. Of these, 83.6% said they were satisfied with the results of tapinarof treatment, and 81.7% said it was more effective than previous topical treatments they had used, according to the company’s release.
Tapinarof cream can be used on all areas of the body, including the face, skin folds, neck, genitalia, anal crux, inflammatory areas, and axillae, according to the company release.
Full prescribing information is available here.
The the manufacturer announced.
Tapinarof is an aryl hydrocarbon receptor agonist and is the first FDA-approved steroid-free topical medication in this class, according to a press release from the manufacturer, Dermavant.
Approval was based on results of three studies in a phase 3 clinical trial program (PSOARING 1, PSOARING 2), and an open-label extension study, (PSOARING 3), the company release said. In PSOARING 1 and 2, approximately 1,000 adults aged 18-75 years (median age, 51 years) with plaque psoriasis were randomized to once-daily topical tapinarof or placebo for up to 12 weeks; 85% were White and 57% were men. The study findings were published in the New England Journal of Medicine in December 2021.
The primary endpoint for both trials was the proportion of patients who achieved Physician Global Assessment (PGA) scores score of “clear” (0) or “almost clear” (1) and improvement of at least two grades from baseline.
After 12 weeks, 36% of the patients in PSOARING 1 and 40% in PSOARING 2 who received tapinarof met the primary outcome, compared with 6% of patients on placebo (P < .001 for both studies). Of these, a total of 73 patients from both studies who achieved PGA scores of 0 were entered in PSOARING 3, a 40-week open-label extension study, in which they stopped tapinarof treatment and retained PGA scores of 0 or 1 for approximately 4 months off treatment. An additional 312 patients who were enrolled in the PSOARING 3 extension study achieved PGA scores of 0 at least once during the study period, with “remittive” effects lasting a mean of 130 days off of treatment.
In addition, patients who received tapinarof in the PSOARING 1 and 2 studies showed significant improvement from baseline, compared with patients on placebo, across a range of secondary endpoints including a 75% or greater improvement in Psoriasis Area and Severity Index score (PASI 75).
In PSOARING 1, and 2, respectively, 36.1% and 47.6% of those on tapinarof achieved a PASI 75 response at week 12, compared with 10.2% and 6.9% of those on the vehicle (P < .001 for both).
Across all three studies, the majority adverse events were mild to moderate, and limited to the application site.
The most common adverse events reported by patients in the tapinarof groups were folliculitis, nasopharyngitis, and contact dermatitis. Headaches were more common among those treated with tapinarof than those on vehicle in the studies (3.8% vs. 2.4% in PSOARING 1, and 3.8% vs. 0.6% in PSOARING 2), leading to only three treatment discontinuations.
At the end of the PSOARING 3 study (at either week 40 or early termination), 599 participants responded to satisfaction questionnaires. Of these, 83.6% said they were satisfied with the results of tapinarof treatment, and 81.7% said it was more effective than previous topical treatments they had used, according to the company’s release.
Tapinarof cream can be used on all areas of the body, including the face, skin folds, neck, genitalia, anal crux, inflammatory areas, and axillae, according to the company release.
Full prescribing information is available here.
Severe infections often accompany severe psoriasis
of nearly 95,000 patients.
Although previous studies have shown a higher risk for comorbid conditions in people with psoriasis, compared with those without psoriasis, data on the occurrence of severe and rare infections in patients with psoriasis are limited, wrote Nikolai Loft, MD, of the department of dermatology and allergy, Copenhagen University Hospital, Gentofte, and colleagues.
Psoriasis patients are often treated with immunosuppressive therapies that may promote or aggravate infections; therefore, a better understanding of psoriasis and risk of infections is needed, they said. In a study published in the British Journal of Dermatology, Dr. Loft and his coinvestigators reviewed data on adults aged 18 years and older from the Danish National Patient Register between Jan. 1, 1997 and Dec. 31, 2018. The study population included 94,450 adults with psoriasis and 566,700 matched controls. Patients with any type of psoriasis and any degree of severity were included.
The primary outcome was the occurrence of severe infections, defined as those requiring assessment at a hospital, and rare infections, defined as HIV, TB, HBV, and HCV. The median age of the participants was 52.3 years, and slightly more than half were women.
Overall, the incidence rate of severe and rare infections among patients with any type of psoriasis was 3,104.9 per 100,000 person-years, compared with 2,381.1 for controls, with a hazard ratio, adjusted for gender, age, ethnicity, socioeconomic status, alcohol-related conditions, and Charlson comorbidity index (aHR) of 1.29.
For any infections resulting in hospitalization, the incidence rate was 2,005.1 vs. 1,531.8 per 100,000 person-years for patients with any type of psoriasis and controls, respectively.
The results were similar when severe infections and rare infections were analyzed separately. The incidence rate of severe infections was 3,080.6 and 2,364.4 per 100,000 person-years for patients with any psoriasis, compared with controls; the incidence rate for rare infections was 42.9 and 31.8 for all psoriasis patients and controls, respectively.
When the data were examined by psoriasis severity, the incidence rate of severe and rare infections among patients with severe psoriasis was 3,847.7 per 100,000 person-years, compared with 2,351.9 per 100,000 person years among controls (aHR, 1.58) and also higher than in patients with mild psoriasis. The incidence rate of severe and rare infections in patients with mild psoriasis (2,979.1 per 100,000 person-years) also was higher than in controls (aHR, 1.26).
Factors that might explain the increased infection risk with severe psoriasis include the altered immune environment in these patients, the researchers wrote in their discussion of the findings. Also, “patients with severe psoriasis are defined by their eligibility for systemics, either conventional or biologic,” and their increased infection risk may stem from these treatments, rather than disease severity itself, they noted.
The study findings were limited by several factors including the lack of data on such confounders as weight, body mass index, and smoking status, they added. Other limitations included potential surveillance bias because of greater TB screening, and the use of prescriptions, rather than the Psoriasis Area Severity Index, to define severity. However, the results were strengthened by the large sample size, and suggest that patients with any type of psoriasis have higher rates of any infection, severe or rare, than the general population, the researchers concluded.
Data show need for clinician vigilance
Based on the 2020 Census data, an estimated 7.55 million adults in the United States have psoriasis, David Robles, MD, said in an interview. “Patients with psoriasis have a high risk for multiple comorbid conditions including metabolic syndrome, which is characterized by obesity, hypertension, and dyslipidemia,” said Dr. Robles, a dermatologist in private practice in Pomona, Calif., who was not involved in the study. “Although these complications were previously attributed to diet and obesity, it has become clear that the proinflammatory cytokines associated with psoriasis may be playing an important role underlying the pathologic basis of these other comorbidities.”
There is an emerging body of literature “indicating that psoriasis is associated with an increased risk of infections,” he added. Research in this area is particularly important because of the increased risk of infections associated with many biologic and immune-modulating treatments for psoriasis, Dr. Robles noted.
The study findings “indicate that, as the severity of psoriasis increases, so does the risk of severe and rare infections,” he said. “This makes it imperative for clinicians to be alert to the possibility of severe or rare infections in patients with psoriasis, especially those with severe psoriasis, so that early intervention can be initiated.”
As for additional research, “as an immunologist and dermatologist, I cannot help but think about the possible role the genetic and cytokine pathways involved in psoriasis may be playing in modulating the immune system and/or microbiome, and whether this contributes to a higher risk of infections,” Dr. Robles said. “Just as it was discovered that patients with atopic dermatitis have decreased levels of antimicrobial peptides in their skin, making them susceptible to recurrent bacterial skin infections, we may find that the genetic and immunological changes associated with psoriasis may independently contribute to infection susceptibility,” he noted. “More basic immunology and virology research may one day shed light on this observation.”
The study was supported by Novartis. Lead author Dr. Loft disclosed serving as a speaker for Eli Lilly and Janssen Cilag, other authors disclosed relationships with multiple companies including Novartis, and two authors are Novartis employees. Dr. Robles had no relevant financial disclosures.
of nearly 95,000 patients.
Although previous studies have shown a higher risk for comorbid conditions in people with psoriasis, compared with those without psoriasis, data on the occurrence of severe and rare infections in patients with psoriasis are limited, wrote Nikolai Loft, MD, of the department of dermatology and allergy, Copenhagen University Hospital, Gentofte, and colleagues.
Psoriasis patients are often treated with immunosuppressive therapies that may promote or aggravate infections; therefore, a better understanding of psoriasis and risk of infections is needed, they said. In a study published in the British Journal of Dermatology, Dr. Loft and his coinvestigators reviewed data on adults aged 18 years and older from the Danish National Patient Register between Jan. 1, 1997 and Dec. 31, 2018. The study population included 94,450 adults with psoriasis and 566,700 matched controls. Patients with any type of psoriasis and any degree of severity were included.
The primary outcome was the occurrence of severe infections, defined as those requiring assessment at a hospital, and rare infections, defined as HIV, TB, HBV, and HCV. The median age of the participants was 52.3 years, and slightly more than half were women.
Overall, the incidence rate of severe and rare infections among patients with any type of psoriasis was 3,104.9 per 100,000 person-years, compared with 2,381.1 for controls, with a hazard ratio, adjusted for gender, age, ethnicity, socioeconomic status, alcohol-related conditions, and Charlson comorbidity index (aHR) of 1.29.
For any infections resulting in hospitalization, the incidence rate was 2,005.1 vs. 1,531.8 per 100,000 person-years for patients with any type of psoriasis and controls, respectively.
The results were similar when severe infections and rare infections were analyzed separately. The incidence rate of severe infections was 3,080.6 and 2,364.4 per 100,000 person-years for patients with any psoriasis, compared with controls; the incidence rate for rare infections was 42.9 and 31.8 for all psoriasis patients and controls, respectively.
When the data were examined by psoriasis severity, the incidence rate of severe and rare infections among patients with severe psoriasis was 3,847.7 per 100,000 person-years, compared with 2,351.9 per 100,000 person years among controls (aHR, 1.58) and also higher than in patients with mild psoriasis. The incidence rate of severe and rare infections in patients with mild psoriasis (2,979.1 per 100,000 person-years) also was higher than in controls (aHR, 1.26).
Factors that might explain the increased infection risk with severe psoriasis include the altered immune environment in these patients, the researchers wrote in their discussion of the findings. Also, “patients with severe psoriasis are defined by their eligibility for systemics, either conventional or biologic,” and their increased infection risk may stem from these treatments, rather than disease severity itself, they noted.
The study findings were limited by several factors including the lack of data on such confounders as weight, body mass index, and smoking status, they added. Other limitations included potential surveillance bias because of greater TB screening, and the use of prescriptions, rather than the Psoriasis Area Severity Index, to define severity. However, the results were strengthened by the large sample size, and suggest that patients with any type of psoriasis have higher rates of any infection, severe or rare, than the general population, the researchers concluded.
Data show need for clinician vigilance
Based on the 2020 Census data, an estimated 7.55 million adults in the United States have psoriasis, David Robles, MD, said in an interview. “Patients with psoriasis have a high risk for multiple comorbid conditions including metabolic syndrome, which is characterized by obesity, hypertension, and dyslipidemia,” said Dr. Robles, a dermatologist in private practice in Pomona, Calif., who was not involved in the study. “Although these complications were previously attributed to diet and obesity, it has become clear that the proinflammatory cytokines associated with psoriasis may be playing an important role underlying the pathologic basis of these other comorbidities.”
There is an emerging body of literature “indicating that psoriasis is associated with an increased risk of infections,” he added. Research in this area is particularly important because of the increased risk of infections associated with many biologic and immune-modulating treatments for psoriasis, Dr. Robles noted.
The study findings “indicate that, as the severity of psoriasis increases, so does the risk of severe and rare infections,” he said. “This makes it imperative for clinicians to be alert to the possibility of severe or rare infections in patients with psoriasis, especially those with severe psoriasis, so that early intervention can be initiated.”
As for additional research, “as an immunologist and dermatologist, I cannot help but think about the possible role the genetic and cytokine pathways involved in psoriasis may be playing in modulating the immune system and/or microbiome, and whether this contributes to a higher risk of infections,” Dr. Robles said. “Just as it was discovered that patients with atopic dermatitis have decreased levels of antimicrobial peptides in their skin, making them susceptible to recurrent bacterial skin infections, we may find that the genetic and immunological changes associated with psoriasis may independently contribute to infection susceptibility,” he noted. “More basic immunology and virology research may one day shed light on this observation.”
The study was supported by Novartis. Lead author Dr. Loft disclosed serving as a speaker for Eli Lilly and Janssen Cilag, other authors disclosed relationships with multiple companies including Novartis, and two authors are Novartis employees. Dr. Robles had no relevant financial disclosures.
of nearly 95,000 patients.
Although previous studies have shown a higher risk for comorbid conditions in people with psoriasis, compared with those without psoriasis, data on the occurrence of severe and rare infections in patients with psoriasis are limited, wrote Nikolai Loft, MD, of the department of dermatology and allergy, Copenhagen University Hospital, Gentofte, and colleagues.
Psoriasis patients are often treated with immunosuppressive therapies that may promote or aggravate infections; therefore, a better understanding of psoriasis and risk of infections is needed, they said. In a study published in the British Journal of Dermatology, Dr. Loft and his coinvestigators reviewed data on adults aged 18 years and older from the Danish National Patient Register between Jan. 1, 1997 and Dec. 31, 2018. The study population included 94,450 adults with psoriasis and 566,700 matched controls. Patients with any type of psoriasis and any degree of severity were included.
The primary outcome was the occurrence of severe infections, defined as those requiring assessment at a hospital, and rare infections, defined as HIV, TB, HBV, and HCV. The median age of the participants was 52.3 years, and slightly more than half were women.
Overall, the incidence rate of severe and rare infections among patients with any type of psoriasis was 3,104.9 per 100,000 person-years, compared with 2,381.1 for controls, with a hazard ratio, adjusted for gender, age, ethnicity, socioeconomic status, alcohol-related conditions, and Charlson comorbidity index (aHR) of 1.29.
For any infections resulting in hospitalization, the incidence rate was 2,005.1 vs. 1,531.8 per 100,000 person-years for patients with any type of psoriasis and controls, respectively.
The results were similar when severe infections and rare infections were analyzed separately. The incidence rate of severe infections was 3,080.6 and 2,364.4 per 100,000 person-years for patients with any psoriasis, compared with controls; the incidence rate for rare infections was 42.9 and 31.8 for all psoriasis patients and controls, respectively.
When the data were examined by psoriasis severity, the incidence rate of severe and rare infections among patients with severe psoriasis was 3,847.7 per 100,000 person-years, compared with 2,351.9 per 100,000 person years among controls (aHR, 1.58) and also higher than in patients with mild psoriasis. The incidence rate of severe and rare infections in patients with mild psoriasis (2,979.1 per 100,000 person-years) also was higher than in controls (aHR, 1.26).
Factors that might explain the increased infection risk with severe psoriasis include the altered immune environment in these patients, the researchers wrote in their discussion of the findings. Also, “patients with severe psoriasis are defined by their eligibility for systemics, either conventional or biologic,” and their increased infection risk may stem from these treatments, rather than disease severity itself, they noted.
The study findings were limited by several factors including the lack of data on such confounders as weight, body mass index, and smoking status, they added. Other limitations included potential surveillance bias because of greater TB screening, and the use of prescriptions, rather than the Psoriasis Area Severity Index, to define severity. However, the results were strengthened by the large sample size, and suggest that patients with any type of psoriasis have higher rates of any infection, severe or rare, than the general population, the researchers concluded.
Data show need for clinician vigilance
Based on the 2020 Census data, an estimated 7.55 million adults in the United States have psoriasis, David Robles, MD, said in an interview. “Patients with psoriasis have a high risk for multiple comorbid conditions including metabolic syndrome, which is characterized by obesity, hypertension, and dyslipidemia,” said Dr. Robles, a dermatologist in private practice in Pomona, Calif., who was not involved in the study. “Although these complications were previously attributed to diet and obesity, it has become clear that the proinflammatory cytokines associated with psoriasis may be playing an important role underlying the pathologic basis of these other comorbidities.”
There is an emerging body of literature “indicating that psoriasis is associated with an increased risk of infections,” he added. Research in this area is particularly important because of the increased risk of infections associated with many biologic and immune-modulating treatments for psoriasis, Dr. Robles noted.
The study findings “indicate that, as the severity of psoriasis increases, so does the risk of severe and rare infections,” he said. “This makes it imperative for clinicians to be alert to the possibility of severe or rare infections in patients with psoriasis, especially those with severe psoriasis, so that early intervention can be initiated.”
As for additional research, “as an immunologist and dermatologist, I cannot help but think about the possible role the genetic and cytokine pathways involved in psoriasis may be playing in modulating the immune system and/or microbiome, and whether this contributes to a higher risk of infections,” Dr. Robles said. “Just as it was discovered that patients with atopic dermatitis have decreased levels of antimicrobial peptides in their skin, making them susceptible to recurrent bacterial skin infections, we may find that the genetic and immunological changes associated with psoriasis may independently contribute to infection susceptibility,” he noted. “More basic immunology and virology research may one day shed light on this observation.”
The study was supported by Novartis. Lead author Dr. Loft disclosed serving as a speaker for Eli Lilly and Janssen Cilag, other authors disclosed relationships with multiple companies including Novartis, and two authors are Novartis employees. Dr. Robles had no relevant financial disclosures.
FROM BRITISH JOURNAL OF DERMATOLOGY
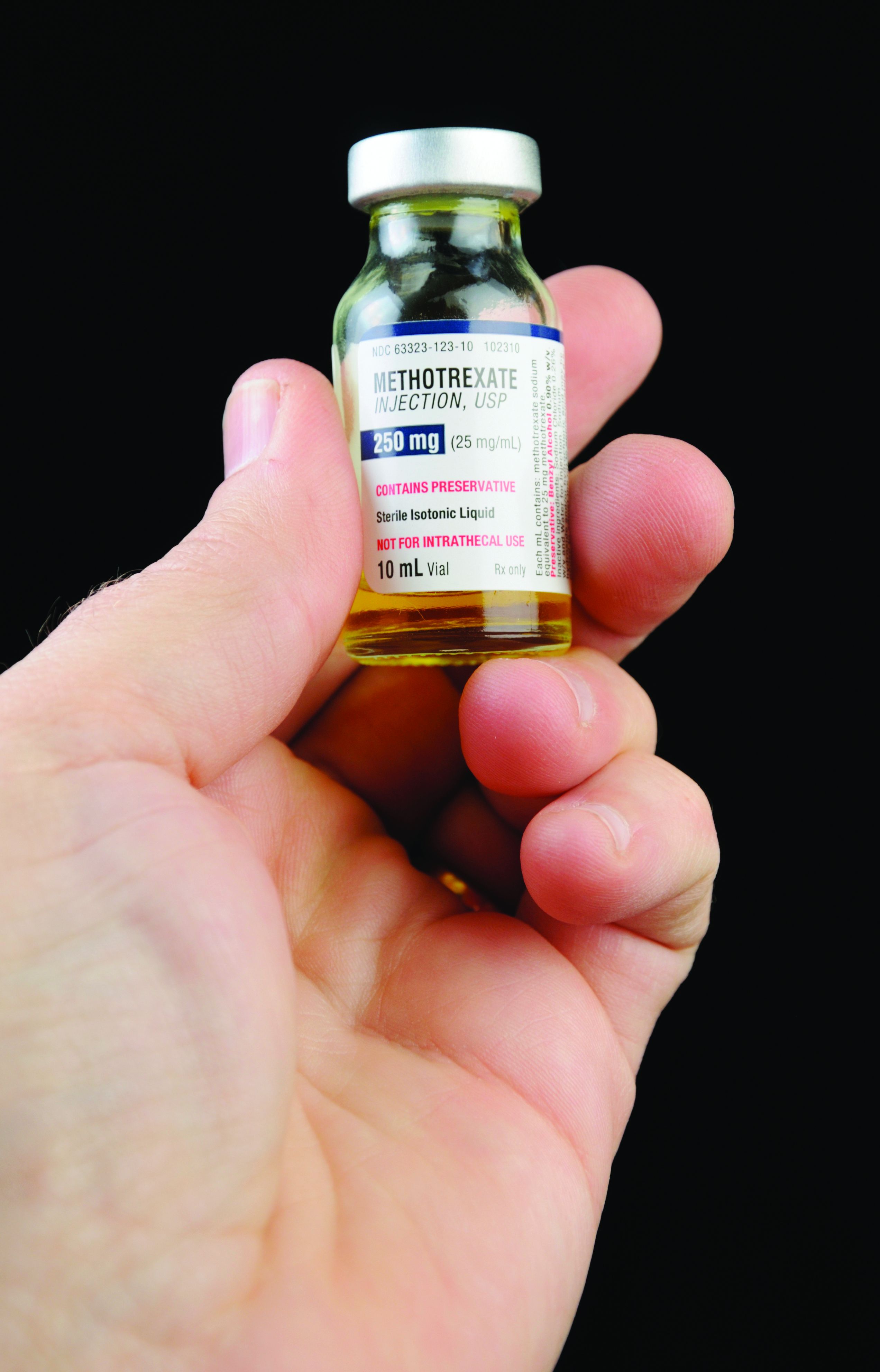
Reduced-frequency methotrexate monitoring causes no harm
Reducing the frequency of routine blood monitoring for methotrexate in patients with rheumatoid arthritis during the COVID-19 pandemic was associated with no adverse outcomes for patients, British researchers have found.
Similar laboratory results were recorded in patients who were switched from testing once per month to once every 3 or 5 months, Natasha Wood, a general practice trainee at North Devon District Hospital in Barnstaple, England, reported at the annual meeting of the British Society for Rheumatology.
“Less frequent monitoring did not result in patient harm,” she said.
“There’s an increasing evidence base; we wonder whether now’s the time to reconsider our DMARD-monitoring strategy,” Ms. Wood said.
Changes in monitoring because of pandemic
Methotrexate monitoring is important to minimize the risk of harm to patients, and it is recommended that standard laboratory tests, such as a complete blood count, creatinine, and liver enzymes are measured regularly. Indeed, both the BSR and the American College of Rheumatology have specific recommendations on the monitoring of methotrexate and other conventional synthetic disease-modifying antirheumatic drugs (csDMARDS).
“The BSR used to advise for monthly blood tests in patients taking methotrexate,” Ms. Wood said, but the BSR moved to recommend testing patients on a stable dose every 3 months in 2017.
“Things of course changed again rapidly with COVID, with the BSR quickly updating their guidelines advising for less frequent monitoring in this patient group,” Ms. Wood said.
As a result, the North Devon Clinical Commissioning Group, which covers the hospital where Ms. Wood works, agreed to allow testing every 6 months for patients on a stable methotrexate dose. “This was across specialties, so not just rheumatology, but dermatology and gastroenterology as well,” she said.
“This provided us with a really exciting and unique opportunity to look at this patient group and see what happened,” Ms. Wood explained.
Effect of less frequent monitoring
At the meeting, Ms. Wood presented the results of an audit of 854 patients found via a search of hospital pathology records who were stable on methotrexate monotherapy for at least 12 months.
Two subanalyses were performed: One looked at patients who had changed from blood testing once every month to once every 3 months (n = 229) and the other looking at a group of 120 patients who had gone from testing once every 3 months to approximately every 5 months.
The mean age of patients was 67 for monthly testing, 69 for testing every 3 months, and 66 for testing about every 5 months, with around two-thirds of patients being of female sex.
A comparison of the number of blood tests performed to the end of April 2020 with the number performed to the end of April 2021 showed that there had mainly been a shift from testing once per month to once every 3 months, with some patients being tested in line with the revised BSR guidelines at around 5 months.
“Interestingly, a third of this group had no changed monitoring frequency despite the change in guidelines,” Ms. Wood said.
“Prepandemic, most patients [were] having monthly bloods despite BSR advice from 2017, and despite the pandemic with the updated shared care guidelines,” patients were still having blood drawn every 3 months, Ms. Wood noted. This perhaps needs further investigation and consideration to understand why recommended changes to the frequency of testing are not being adhered to.
The overall distribution of laboratory findings was similar among those who went from testing once per month to once every 3 months and from every 3 months to every 5 months. This included the distribution of neutrophils, whole blood counts, and alanine aminotransferase. There were some changes for platelets, mean cell volume, and the estimated glomerular filtration rate, but these were not clinically significant.
“Abnormal blood results aren’t common in stable methotrexate monotherapy patients,” Ms. Wood reported. “Where abnormalities did occur, it was in the context of patients being concurrently unwell and symptomatic.”
Time for patient-initiated testing?
There are several advantages of less frequent methotrexate monitoring, Ms. Wood said. One is the practicalities of getting to and from appointments, particularly in remote locations, such as where she works.
In addition to reducing workloads and pressure on already busy hospitals and primary care, this could have a huge environmental impact, she suggested.
Moreover, “moderate-quality evidence” supports the current monitoring frequency recommendation.
“We know that our numbers are small – we’re a small center – but our findings are consistent with much larger studies across the U.K.,” Ms. Wood said.
“We wonder whether there’s the possibility of moving towards annual monitoring with good safety netting and patient education for additional blood tests if they are unwell,” she said, adding that “now may be the time for patient-initiated methotrexate monitoring.”
Ms. Wood disclosed Janssen sponsorship for attending the BSR 2022 annual meeting.
Reducing the frequency of routine blood monitoring for methotrexate in patients with rheumatoid arthritis during the COVID-19 pandemic was associated with no adverse outcomes for patients, British researchers have found.
Similar laboratory results were recorded in patients who were switched from testing once per month to once every 3 or 5 months, Natasha Wood, a general practice trainee at North Devon District Hospital in Barnstaple, England, reported at the annual meeting of the British Society for Rheumatology.
“Less frequent monitoring did not result in patient harm,” she said.
“There’s an increasing evidence base; we wonder whether now’s the time to reconsider our DMARD-monitoring strategy,” Ms. Wood said.
Changes in monitoring because of pandemic
Methotrexate monitoring is important to minimize the risk of harm to patients, and it is recommended that standard laboratory tests, such as a complete blood count, creatinine, and liver enzymes are measured regularly. Indeed, both the BSR and the American College of Rheumatology have specific recommendations on the monitoring of methotrexate and other conventional synthetic disease-modifying antirheumatic drugs (csDMARDS).
“The BSR used to advise for monthly blood tests in patients taking methotrexate,” Ms. Wood said, but the BSR moved to recommend testing patients on a stable dose every 3 months in 2017.
“Things of course changed again rapidly with COVID, with the BSR quickly updating their guidelines advising for less frequent monitoring in this patient group,” Ms. Wood said.
As a result, the North Devon Clinical Commissioning Group, which covers the hospital where Ms. Wood works, agreed to allow testing every 6 months for patients on a stable methotrexate dose. “This was across specialties, so not just rheumatology, but dermatology and gastroenterology as well,” she said.
“This provided us with a really exciting and unique opportunity to look at this patient group and see what happened,” Ms. Wood explained.
Effect of less frequent monitoring
At the meeting, Ms. Wood presented the results of an audit of 854 patients found via a search of hospital pathology records who were stable on methotrexate monotherapy for at least 12 months.
Two subanalyses were performed: One looked at patients who had changed from blood testing once every month to once every 3 months (n = 229) and the other looking at a group of 120 patients who had gone from testing once every 3 months to approximately every 5 months.
The mean age of patients was 67 for monthly testing, 69 for testing every 3 months, and 66 for testing about every 5 months, with around two-thirds of patients being of female sex.
A comparison of the number of blood tests performed to the end of April 2020 with the number performed to the end of April 2021 showed that there had mainly been a shift from testing once per month to once every 3 months, with some patients being tested in line with the revised BSR guidelines at around 5 months.
“Interestingly, a third of this group had no changed monitoring frequency despite the change in guidelines,” Ms. Wood said.
“Prepandemic, most patients [were] having monthly bloods despite BSR advice from 2017, and despite the pandemic with the updated shared care guidelines,” patients were still having blood drawn every 3 months, Ms. Wood noted. This perhaps needs further investigation and consideration to understand why recommended changes to the frequency of testing are not being adhered to.
The overall distribution of laboratory findings was similar among those who went from testing once per month to once every 3 months and from every 3 months to every 5 months. This included the distribution of neutrophils, whole blood counts, and alanine aminotransferase. There were some changes for platelets, mean cell volume, and the estimated glomerular filtration rate, but these were not clinically significant.
“Abnormal blood results aren’t common in stable methotrexate monotherapy patients,” Ms. Wood reported. “Where abnormalities did occur, it was in the context of patients being concurrently unwell and symptomatic.”
Time for patient-initiated testing?
There are several advantages of less frequent methotrexate monitoring, Ms. Wood said. One is the practicalities of getting to and from appointments, particularly in remote locations, such as where she works.
In addition to reducing workloads and pressure on already busy hospitals and primary care, this could have a huge environmental impact, she suggested.
Moreover, “moderate-quality evidence” supports the current monitoring frequency recommendation.
“We know that our numbers are small – we’re a small center – but our findings are consistent with much larger studies across the U.K.,” Ms. Wood said.
“We wonder whether there’s the possibility of moving towards annual monitoring with good safety netting and patient education for additional blood tests if they are unwell,” she said, adding that “now may be the time for patient-initiated methotrexate monitoring.”
Ms. Wood disclosed Janssen sponsorship for attending the BSR 2022 annual meeting.
Reducing the frequency of routine blood monitoring for methotrexate in patients with rheumatoid arthritis during the COVID-19 pandemic was associated with no adverse outcomes for patients, British researchers have found.
Similar laboratory results were recorded in patients who were switched from testing once per month to once every 3 or 5 months, Natasha Wood, a general practice trainee at North Devon District Hospital in Barnstaple, England, reported at the annual meeting of the British Society for Rheumatology.
“Less frequent monitoring did not result in patient harm,” she said.
“There’s an increasing evidence base; we wonder whether now’s the time to reconsider our DMARD-monitoring strategy,” Ms. Wood said.
Changes in monitoring because of pandemic
Methotrexate monitoring is important to minimize the risk of harm to patients, and it is recommended that standard laboratory tests, such as a complete blood count, creatinine, and liver enzymes are measured regularly. Indeed, both the BSR and the American College of Rheumatology have specific recommendations on the monitoring of methotrexate and other conventional synthetic disease-modifying antirheumatic drugs (csDMARDS).
“The BSR used to advise for monthly blood tests in patients taking methotrexate,” Ms. Wood said, but the BSR moved to recommend testing patients on a stable dose every 3 months in 2017.
“Things of course changed again rapidly with COVID, with the BSR quickly updating their guidelines advising for less frequent monitoring in this patient group,” Ms. Wood said.
As a result, the North Devon Clinical Commissioning Group, which covers the hospital where Ms. Wood works, agreed to allow testing every 6 months for patients on a stable methotrexate dose. “This was across specialties, so not just rheumatology, but dermatology and gastroenterology as well,” she said.
“This provided us with a really exciting and unique opportunity to look at this patient group and see what happened,” Ms. Wood explained.
Effect of less frequent monitoring
At the meeting, Ms. Wood presented the results of an audit of 854 patients found via a search of hospital pathology records who were stable on methotrexate monotherapy for at least 12 months.
Two subanalyses were performed: One looked at patients who had changed from blood testing once every month to once every 3 months (n = 229) and the other looking at a group of 120 patients who had gone from testing once every 3 months to approximately every 5 months.
The mean age of patients was 67 for monthly testing, 69 for testing every 3 months, and 66 for testing about every 5 months, with around two-thirds of patients being of female sex.
A comparison of the number of blood tests performed to the end of April 2020 with the number performed to the end of April 2021 showed that there had mainly been a shift from testing once per month to once every 3 months, with some patients being tested in line with the revised BSR guidelines at around 5 months.
“Interestingly, a third of this group had no changed monitoring frequency despite the change in guidelines,” Ms. Wood said.
“Prepandemic, most patients [were] having monthly bloods despite BSR advice from 2017, and despite the pandemic with the updated shared care guidelines,” patients were still having blood drawn every 3 months, Ms. Wood noted. This perhaps needs further investigation and consideration to understand why recommended changes to the frequency of testing are not being adhered to.
The overall distribution of laboratory findings was similar among those who went from testing once per month to once every 3 months and from every 3 months to every 5 months. This included the distribution of neutrophils, whole blood counts, and alanine aminotransferase. There were some changes for platelets, mean cell volume, and the estimated glomerular filtration rate, but these were not clinically significant.
“Abnormal blood results aren’t common in stable methotrexate monotherapy patients,” Ms. Wood reported. “Where abnormalities did occur, it was in the context of patients being concurrently unwell and symptomatic.”
Time for patient-initiated testing?
There are several advantages of less frequent methotrexate monitoring, Ms. Wood said. One is the practicalities of getting to and from appointments, particularly in remote locations, such as where she works.
In addition to reducing workloads and pressure on already busy hospitals and primary care, this could have a huge environmental impact, she suggested.
Moreover, “moderate-quality evidence” supports the current monitoring frequency recommendation.
“We know that our numbers are small – we’re a small center – but our findings are consistent with much larger studies across the U.K.,” Ms. Wood said.
“We wonder whether there’s the possibility of moving towards annual monitoring with good safety netting and patient education for additional blood tests if they are unwell,” she said, adding that “now may be the time for patient-initiated methotrexate monitoring.”
Ms. Wood disclosed Janssen sponsorship for attending the BSR 2022 annual meeting.
FROM BSR 2022
Parents’ autoimmune diseases may affect children’s development
Results of a meta-analysis carried out by a French team indicate that there is a link between a father’s or mother’s autoimmune disease and their children’s risk of developing certain neurodevelopmental disorders (autism spectrum disorder [ASD] and attention-deficit/hyperactivity disorder). This meta-analysis is the first to separately explore the link between a father’s or mother’s autoimmune disease and the onset of neurodevelopmental disorders in their children.
According to its authors, these associations may result from exposure to environmental factors that contribute to autoimmune disorders, such as exposure to pollutants or cigarette smoke, and/or genetic predisposition, including genes relating to cytokines or to the HLA system.
Research is needed to determine the pathophysiologic links between these associations. This study suggests that there could be a shared mechanism between both parents, even though the maternal route seems to constitute an additional excess risk.
Why is this important?
Neurodevelopmental disorders are said to occur because of a close interrelationship between a person’s genes and environment. Immune-mediated adverse reactions may play an important role in triggering such disorders, as has been shown in associated epidemiologic studies and in animal studies. Autoimmune and autoinflammatory disorders are effectively characterized by the activation of the immune system, the circulation of autoantibodies, and the secretion of cytokines that are harmful to certain tissues.
Some relevant studies suggest a link between autoimmune disorders in the family or in the mother and the onset of neurodevelopmental disorders in their children. However, none of the studies have distinguished the influence of each of the parents so as to provide data that can be used to assess whether this association is more likely to be direct, and thus established during pregnancy, or rather genetic or environmental.
Main findings
Overall, the meta-analysis involved 14 studies that included 845,411 mothers and 601,148 fathers with an autoimmune disease, 4,984,965 control mothers and 4,992,854 control fathers. There were 182,927 children with neurodevelopmental disorders and 14,168,474 with no such diagnosis.
Globally, autoimmune diseases in mothers (adjusted odds ratio, 1.27 [1.03-1.57]; P = .02; I2 = 65%) and in fathers (AOR, 1.18 [1.07-1.30]; P = .01; I2 = 15.5%) are associated with a diagnosis of ASD in children. Similarly, they are associated with an increased risk of ADHD in children (AOR, 1.31 [1.11-1.55]; P = .001; I2 = 93% and AOR, 1.14 [1.10-1.17]; P < .0001; I2 = 0%, respectively, for mothers and fathers).
In mothers, type 1 diabetes (AOR, 1.60 [1.18-2.18]; P = .002; I2 = 0%), psoriasis (AOR, 1.45 [1.14-1.85]; P = .002; I2 = 0%), and rheumatoid arthritis (AOR, 1.38 [1.14-1.68]; P = .001; I2 = 0.8%) were associated with a risk of ASD in children. These three conditions also predisposed children to the risk of ADHD (AOR, 1.36 [1.24-1.52]; 1.41 [1.29-1.54]; and 1.32 [1.25-1.40], respectively, all P < .0001).
In fathers, type 1 diabetes considered in isolation was associated with a risk of ASD and ADHD in children (AOR, 1.42 [1.10-1.83] and 1.19 [1.08-1.31], respectively), while psoriasis (AOR, 1.18 [1.12-1.24]; P < .0001) is associated with a risk of ADHD in children.
A version of this article first appeared on Medscape.com.
Results of a meta-analysis carried out by a French team indicate that there is a link between a father’s or mother’s autoimmune disease and their children’s risk of developing certain neurodevelopmental disorders (autism spectrum disorder [ASD] and attention-deficit/hyperactivity disorder). This meta-analysis is the first to separately explore the link between a father’s or mother’s autoimmune disease and the onset of neurodevelopmental disorders in their children.
According to its authors, these associations may result from exposure to environmental factors that contribute to autoimmune disorders, such as exposure to pollutants or cigarette smoke, and/or genetic predisposition, including genes relating to cytokines or to the HLA system.
Research is needed to determine the pathophysiologic links between these associations. This study suggests that there could be a shared mechanism between both parents, even though the maternal route seems to constitute an additional excess risk.
Why is this important?
Neurodevelopmental disorders are said to occur because of a close interrelationship between a person’s genes and environment. Immune-mediated adverse reactions may play an important role in triggering such disorders, as has been shown in associated epidemiologic studies and in animal studies. Autoimmune and autoinflammatory disorders are effectively characterized by the activation of the immune system, the circulation of autoantibodies, and the secretion of cytokines that are harmful to certain tissues.
Some relevant studies suggest a link between autoimmune disorders in the family or in the mother and the onset of neurodevelopmental disorders in their children. However, none of the studies have distinguished the influence of each of the parents so as to provide data that can be used to assess whether this association is more likely to be direct, and thus established during pregnancy, or rather genetic or environmental.
Main findings
Overall, the meta-analysis involved 14 studies that included 845,411 mothers and 601,148 fathers with an autoimmune disease, 4,984,965 control mothers and 4,992,854 control fathers. There were 182,927 children with neurodevelopmental disorders and 14,168,474 with no such diagnosis.
Globally, autoimmune diseases in mothers (adjusted odds ratio, 1.27 [1.03-1.57]; P = .02; I2 = 65%) and in fathers (AOR, 1.18 [1.07-1.30]; P = .01; I2 = 15.5%) are associated with a diagnosis of ASD in children. Similarly, they are associated with an increased risk of ADHD in children (AOR, 1.31 [1.11-1.55]; P = .001; I2 = 93% and AOR, 1.14 [1.10-1.17]; P < .0001; I2 = 0%, respectively, for mothers and fathers).
In mothers, type 1 diabetes (AOR, 1.60 [1.18-2.18]; P = .002; I2 = 0%), psoriasis (AOR, 1.45 [1.14-1.85]; P = .002; I2 = 0%), and rheumatoid arthritis (AOR, 1.38 [1.14-1.68]; P = .001; I2 = 0.8%) were associated with a risk of ASD in children. These three conditions also predisposed children to the risk of ADHD (AOR, 1.36 [1.24-1.52]; 1.41 [1.29-1.54]; and 1.32 [1.25-1.40], respectively, all P < .0001).
In fathers, type 1 diabetes considered in isolation was associated with a risk of ASD and ADHD in children (AOR, 1.42 [1.10-1.83] and 1.19 [1.08-1.31], respectively), while psoriasis (AOR, 1.18 [1.12-1.24]; P < .0001) is associated with a risk of ADHD in children.
A version of this article first appeared on Medscape.com.
Results of a meta-analysis carried out by a French team indicate that there is a link between a father’s or mother’s autoimmune disease and their children’s risk of developing certain neurodevelopmental disorders (autism spectrum disorder [ASD] and attention-deficit/hyperactivity disorder). This meta-analysis is the first to separately explore the link between a father’s or mother’s autoimmune disease and the onset of neurodevelopmental disorders in their children.
According to its authors, these associations may result from exposure to environmental factors that contribute to autoimmune disorders, such as exposure to pollutants or cigarette smoke, and/or genetic predisposition, including genes relating to cytokines or to the HLA system.
Research is needed to determine the pathophysiologic links between these associations. This study suggests that there could be a shared mechanism between both parents, even though the maternal route seems to constitute an additional excess risk.
Why is this important?
Neurodevelopmental disorders are said to occur because of a close interrelationship between a person’s genes and environment. Immune-mediated adverse reactions may play an important role in triggering such disorders, as has been shown in associated epidemiologic studies and in animal studies. Autoimmune and autoinflammatory disorders are effectively characterized by the activation of the immune system, the circulation of autoantibodies, and the secretion of cytokines that are harmful to certain tissues.
Some relevant studies suggest a link between autoimmune disorders in the family or in the mother and the onset of neurodevelopmental disorders in their children. However, none of the studies have distinguished the influence of each of the parents so as to provide data that can be used to assess whether this association is more likely to be direct, and thus established during pregnancy, or rather genetic or environmental.
Main findings
Overall, the meta-analysis involved 14 studies that included 845,411 mothers and 601,148 fathers with an autoimmune disease, 4,984,965 control mothers and 4,992,854 control fathers. There were 182,927 children with neurodevelopmental disorders and 14,168,474 with no such diagnosis.
Globally, autoimmune diseases in mothers (adjusted odds ratio, 1.27 [1.03-1.57]; P = .02; I2 = 65%) and in fathers (AOR, 1.18 [1.07-1.30]; P = .01; I2 = 15.5%) are associated with a diagnosis of ASD in children. Similarly, they are associated with an increased risk of ADHD in children (AOR, 1.31 [1.11-1.55]; P = .001; I2 = 93% and AOR, 1.14 [1.10-1.17]; P < .0001; I2 = 0%, respectively, for mothers and fathers).
In mothers, type 1 diabetes (AOR, 1.60 [1.18-2.18]; P = .002; I2 = 0%), psoriasis (AOR, 1.45 [1.14-1.85]; P = .002; I2 = 0%), and rheumatoid arthritis (AOR, 1.38 [1.14-1.68]; P = .001; I2 = 0.8%) were associated with a risk of ASD in children. These three conditions also predisposed children to the risk of ADHD (AOR, 1.36 [1.24-1.52]; 1.41 [1.29-1.54]; and 1.32 [1.25-1.40], respectively, all P < .0001).
In fathers, type 1 diabetes considered in isolation was associated with a risk of ASD and ADHD in children (AOR, 1.42 [1.10-1.83] and 1.19 [1.08-1.31], respectively), while psoriasis (AOR, 1.18 [1.12-1.24]; P < .0001) is associated with a risk of ADHD in children.
A version of this article first appeared on Medscape.com.
FROM TRANSLATIONAL PSYCHIATRY
NB-UVB phototherapy plays a key role in psoriasis treatment, expert says
BOSTON – In 2012, about 50% of patients receiving phototherapy at Brigham and Women’s Hospital in Boston were being treated for psoriasis. A decade later, that proportion has dropped to 20%.
Several factors have contributed to this trend, namely, the development of biologics, the COVID-19 pandemic, “and the rise of home phototherapy options,” Elizabeth A. Buzney, MD, codirector of the phototherapy center at Brigham and Women’s department of dermatology, said at the annual meeting of the American Academy of Dermatology. In her clinical opinion, phototherapy plays an essential role in the treatment of psoriasis.
“It is medically and financially responsible to review the option of phototherapy with every psoriasis patient,” Dr. Buzney said. “Many patients are not medical or financial candidates for biologic/apremilast therapy, or just would prefer nonsystemic therapy.”
In one meta-analysis, the proportion of patients achieving Psoriasis Area and Severity Index (PASI) 75 with NB-UVB therapy was 70% after 20-40 sessions, just below the efficacy of newer biologics – but better than ustekinumab and adalimumab.
“Phototherapy is not so far out of range as you might think it is,” she said, noting that other studies of NB-UVB therapy show PASI 75 responses of 62% and PASI 90 responses of 40%.
Phototherapy can also be an appealing option because biologics aren’t the best option for all patients with psoriasis. They are expensive for the health care system and potentially for patients, require initial and potentially continued lab testing and monitoring, and require injections, “which some patients don’t like,” said Dr. Buzney, who is also vice-chair of clinical affairs at the Brigham and Women’s Hospital department of dermatology. “There’s an infrequent risk of serious infection and there is risk in patients with HIV, TB, and hepatitis that you have to address. There is also concern for the impact of biologics on patients with a recent cancer.”
On the other hand, few contraindications to NB-UVB exist. According to joint American Academy of Dermatology-National Psoriasis Foundation guidelines on the management and treatment of psoriasis with phototherapy, published in 2019, NB-UVB therapy is only contraindicated in patients with xeroderma pigmentosa and other photosensitive disorders. Concurrent use of cyclosporine and NB-UVB treatment is also contraindicated because of the calculated increase in risk of skin cancer, extrapolated from data on risk with cyclosporine and PUVA (psoralen and ultraviolet A therapy).
The guidelines state that NB-UVB can be used with caution in lupus patients with no history of photosensitivity and who are SS-A negative, as well as patients with a history of melanoma or multiple nonmelanoma skin cancers, a history of recurrent oral herpes simplex virus infection, a history of arsenic intake, prior exposure to ionizing radiation, and those taking photosensitizing medications (since NB-UVB lamps emit “negligible” UVA).
It’s also safe to use during pregnancy and in children. “It’s safe and effective for the right patient,” Dr. Buzney said, discussing how phototherapy can be modified to accommodate children. “You can consider a slower dose-increased regimen. Will children keep the eye protection on? That’s a tricky one. How are you going to manage their anxiety during treatment and involve their family?”
Subgroups of patients who demonstrate a better response to NB-UVB treatment include those with guttate psoriasis, compared with plaque psoriasis, nonsmokers, those with a lower BMI, those with a higher baseline PASI, and those who demonstrate a faster trajectory of clinical response over the first 2-3 weeks of treatment.
Why would one not use phototherapy for psoriasis? “Cost and convenience,” Dr. Buzney said. “There is lost time/revenue to commute to treatment, which may involve multiple times per week. Coming to a public space when COVID-19 is still lingering is another concern, as are the out-of-pocket costs for copays and parking.”
For these reasons, she considers home phototherapy as a transformative option for many patients. Home phototherapy booths provide a safe and effective way to use NB-UVB phototherapy while minimizing copays and commuting costs. The one-time price tag of home NB-UVB booths runs between $5,000 and $7,000, but that is “much less expensive than the biologics,” which can cost $40,000-$50,000 per year, she said.
A small cross-sectional study of office- versus home-based NB-UVB in patients with vitiligo found a cost savings for home-based NB-UVB after 3 months.
One of the challenges with home phototherapy is the lack of long-term studies on patient use. In a small study Dr. Buzney conducted of 30 patients who were prescribed home phototherapy in the last 5 years, 65% practiced (or had practiced) conservative dosing, 83% had continued care with a dermatologist, 19% reported sunburns (5 mild and 1 severe), and 50% had discontinued the therapy at the time of survey because of a perceived lack of efficacy and inconvenience. But 30% of those who had stopped had done so within one month of getting their home booth.
“This tells me that we have to educate our patients better about what expectations should be and make sure they understand how to use their booths,” she said. “Home phototherapy has changed my practice, but not everyone is a candidate for it. Some patients are not dependable. Others are unable to understand instructions.”
Cost to purchase a NB-UVB booth is also an issue, she noted. “Typically, a percentage of cost is covered by insurance, but it’s problematic to purchase a booth if patients don’t know it’s going to work for them or not. Then you have college students who don’t have the space in their apartment or dorm room for a booth.”
Dr. Buzney reported having no relevant financial conflicts.
BOSTON – In 2012, about 50% of patients receiving phototherapy at Brigham and Women’s Hospital in Boston were being treated for psoriasis. A decade later, that proportion has dropped to 20%.
Several factors have contributed to this trend, namely, the development of biologics, the COVID-19 pandemic, “and the rise of home phototherapy options,” Elizabeth A. Buzney, MD, codirector of the phototherapy center at Brigham and Women’s department of dermatology, said at the annual meeting of the American Academy of Dermatology. In her clinical opinion, phototherapy plays an essential role in the treatment of psoriasis.
“It is medically and financially responsible to review the option of phototherapy with every psoriasis patient,” Dr. Buzney said. “Many patients are not medical or financial candidates for biologic/apremilast therapy, or just would prefer nonsystemic therapy.”
In one meta-analysis, the proportion of patients achieving Psoriasis Area and Severity Index (PASI) 75 with NB-UVB therapy was 70% after 20-40 sessions, just below the efficacy of newer biologics – but better than ustekinumab and adalimumab.
“Phototherapy is not so far out of range as you might think it is,” she said, noting that other studies of NB-UVB therapy show PASI 75 responses of 62% and PASI 90 responses of 40%.
Phototherapy can also be an appealing option because biologics aren’t the best option for all patients with psoriasis. They are expensive for the health care system and potentially for patients, require initial and potentially continued lab testing and monitoring, and require injections, “which some patients don’t like,” said Dr. Buzney, who is also vice-chair of clinical affairs at the Brigham and Women’s Hospital department of dermatology. “There’s an infrequent risk of serious infection and there is risk in patients with HIV, TB, and hepatitis that you have to address. There is also concern for the impact of biologics on patients with a recent cancer.”
On the other hand, few contraindications to NB-UVB exist. According to joint American Academy of Dermatology-National Psoriasis Foundation guidelines on the management and treatment of psoriasis with phototherapy, published in 2019, NB-UVB therapy is only contraindicated in patients with xeroderma pigmentosa and other photosensitive disorders. Concurrent use of cyclosporine and NB-UVB treatment is also contraindicated because of the calculated increase in risk of skin cancer, extrapolated from data on risk with cyclosporine and PUVA (psoralen and ultraviolet A therapy).
The guidelines state that NB-UVB can be used with caution in lupus patients with no history of photosensitivity and who are SS-A negative, as well as patients with a history of melanoma or multiple nonmelanoma skin cancers, a history of recurrent oral herpes simplex virus infection, a history of arsenic intake, prior exposure to ionizing radiation, and those taking photosensitizing medications (since NB-UVB lamps emit “negligible” UVA).
It’s also safe to use during pregnancy and in children. “It’s safe and effective for the right patient,” Dr. Buzney said, discussing how phototherapy can be modified to accommodate children. “You can consider a slower dose-increased regimen. Will children keep the eye protection on? That’s a tricky one. How are you going to manage their anxiety during treatment and involve their family?”
Subgroups of patients who demonstrate a better response to NB-UVB treatment include those with guttate psoriasis, compared with plaque psoriasis, nonsmokers, those with a lower BMI, those with a higher baseline PASI, and those who demonstrate a faster trajectory of clinical response over the first 2-3 weeks of treatment.
Why would one not use phototherapy for psoriasis? “Cost and convenience,” Dr. Buzney said. “There is lost time/revenue to commute to treatment, which may involve multiple times per week. Coming to a public space when COVID-19 is still lingering is another concern, as are the out-of-pocket costs for copays and parking.”
For these reasons, she considers home phototherapy as a transformative option for many patients. Home phototherapy booths provide a safe and effective way to use NB-UVB phototherapy while minimizing copays and commuting costs. The one-time price tag of home NB-UVB booths runs between $5,000 and $7,000, but that is “much less expensive than the biologics,” which can cost $40,000-$50,000 per year, she said.
A small cross-sectional study of office- versus home-based NB-UVB in patients with vitiligo found a cost savings for home-based NB-UVB after 3 months.
One of the challenges with home phototherapy is the lack of long-term studies on patient use. In a small study Dr. Buzney conducted of 30 patients who were prescribed home phototherapy in the last 5 years, 65% practiced (or had practiced) conservative dosing, 83% had continued care with a dermatologist, 19% reported sunburns (5 mild and 1 severe), and 50% had discontinued the therapy at the time of survey because of a perceived lack of efficacy and inconvenience. But 30% of those who had stopped had done so within one month of getting their home booth.
“This tells me that we have to educate our patients better about what expectations should be and make sure they understand how to use their booths,” she said. “Home phototherapy has changed my practice, but not everyone is a candidate for it. Some patients are not dependable. Others are unable to understand instructions.”
Cost to purchase a NB-UVB booth is also an issue, she noted. “Typically, a percentage of cost is covered by insurance, but it’s problematic to purchase a booth if patients don’t know it’s going to work for them or not. Then you have college students who don’t have the space in their apartment or dorm room for a booth.”
Dr. Buzney reported having no relevant financial conflicts.
BOSTON – In 2012, about 50% of patients receiving phototherapy at Brigham and Women’s Hospital in Boston were being treated for psoriasis. A decade later, that proportion has dropped to 20%.
Several factors have contributed to this trend, namely, the development of biologics, the COVID-19 pandemic, “and the rise of home phototherapy options,” Elizabeth A. Buzney, MD, codirector of the phototherapy center at Brigham and Women’s department of dermatology, said at the annual meeting of the American Academy of Dermatology. In her clinical opinion, phototherapy plays an essential role in the treatment of psoriasis.
“It is medically and financially responsible to review the option of phototherapy with every psoriasis patient,” Dr. Buzney said. “Many patients are not medical or financial candidates for biologic/apremilast therapy, or just would prefer nonsystemic therapy.”
In one meta-analysis, the proportion of patients achieving Psoriasis Area and Severity Index (PASI) 75 with NB-UVB therapy was 70% after 20-40 sessions, just below the efficacy of newer biologics – but better than ustekinumab and adalimumab.
“Phototherapy is not so far out of range as you might think it is,” she said, noting that other studies of NB-UVB therapy show PASI 75 responses of 62% and PASI 90 responses of 40%.
Phototherapy can also be an appealing option because biologics aren’t the best option for all patients with psoriasis. They are expensive for the health care system and potentially for patients, require initial and potentially continued lab testing and monitoring, and require injections, “which some patients don’t like,” said Dr. Buzney, who is also vice-chair of clinical affairs at the Brigham and Women’s Hospital department of dermatology. “There’s an infrequent risk of serious infection and there is risk in patients with HIV, TB, and hepatitis that you have to address. There is also concern for the impact of biologics on patients with a recent cancer.”
On the other hand, few contraindications to NB-UVB exist. According to joint American Academy of Dermatology-National Psoriasis Foundation guidelines on the management and treatment of psoriasis with phototherapy, published in 2019, NB-UVB therapy is only contraindicated in patients with xeroderma pigmentosa and other photosensitive disorders. Concurrent use of cyclosporine and NB-UVB treatment is also contraindicated because of the calculated increase in risk of skin cancer, extrapolated from data on risk with cyclosporine and PUVA (psoralen and ultraviolet A therapy).
The guidelines state that NB-UVB can be used with caution in lupus patients with no history of photosensitivity and who are SS-A negative, as well as patients with a history of melanoma or multiple nonmelanoma skin cancers, a history of recurrent oral herpes simplex virus infection, a history of arsenic intake, prior exposure to ionizing radiation, and those taking photosensitizing medications (since NB-UVB lamps emit “negligible” UVA).
It’s also safe to use during pregnancy and in children. “It’s safe and effective for the right patient,” Dr. Buzney said, discussing how phototherapy can be modified to accommodate children. “You can consider a slower dose-increased regimen. Will children keep the eye protection on? That’s a tricky one. How are you going to manage their anxiety during treatment and involve their family?”
Subgroups of patients who demonstrate a better response to NB-UVB treatment include those with guttate psoriasis, compared with plaque psoriasis, nonsmokers, those with a lower BMI, those with a higher baseline PASI, and those who demonstrate a faster trajectory of clinical response over the first 2-3 weeks of treatment.
Why would one not use phototherapy for psoriasis? “Cost and convenience,” Dr. Buzney said. “There is lost time/revenue to commute to treatment, which may involve multiple times per week. Coming to a public space when COVID-19 is still lingering is another concern, as are the out-of-pocket costs for copays and parking.”
For these reasons, she considers home phototherapy as a transformative option for many patients. Home phototherapy booths provide a safe and effective way to use NB-UVB phototherapy while minimizing copays and commuting costs. The one-time price tag of home NB-UVB booths runs between $5,000 and $7,000, but that is “much less expensive than the biologics,” which can cost $40,000-$50,000 per year, she said.
A small cross-sectional study of office- versus home-based NB-UVB in patients with vitiligo found a cost savings for home-based NB-UVB after 3 months.
One of the challenges with home phototherapy is the lack of long-term studies on patient use. In a small study Dr. Buzney conducted of 30 patients who were prescribed home phototherapy in the last 5 years, 65% practiced (or had practiced) conservative dosing, 83% had continued care with a dermatologist, 19% reported sunburns (5 mild and 1 severe), and 50% had discontinued the therapy at the time of survey because of a perceived lack of efficacy and inconvenience. But 30% of those who had stopped had done so within one month of getting their home booth.
“This tells me that we have to educate our patients better about what expectations should be and make sure they understand how to use their booths,” she said. “Home phototherapy has changed my practice, but not everyone is a candidate for it. Some patients are not dependable. Others are unable to understand instructions.”
Cost to purchase a NB-UVB booth is also an issue, she noted. “Typically, a percentage of cost is covered by insurance, but it’s problematic to purchase a booth if patients don’t know it’s going to work for them or not. Then you have college students who don’t have the space in their apartment or dorm room for a booth.”
Dr. Buzney reported having no relevant financial conflicts.
AT AAD 22
Long-term efficacy, safety data for ixekizumab in pediatric psoriasis reported
with the interleukin (IL)-17 inhibitor, investigators reported.
In addition, findings of a substudy, which evaluated randomized withdrawal of treatment after 60 weeks, suggest patients were able to regain benefit after not being treated for a period.
Ixekizumab (Taltz) was approved by the U.S. Food and Drug Administration for treating pediatric psoriasis in March 2020 for patients aged 6 years and older with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy.
The trial (IXORA-PEDS) involved 171 patients aged 6-17 years (mean age, 13.5 years; 99 females and 72 males), who were randomly assigned to receive ixekizumab via subcutaneous administration every 4 weeks (115) or placebo for 12 weeks (56). Thereafter, 166 patients continued in an open-label maintenance period in which they were treated every 4 weeks for 12-60 weeks. This was followed by an extension period of up to 108 weeks, which was completed by 139 patients (83.7%). At baseline, the patients’ Psoriasis Area and Severity Index (PASI) score was 12 or higher, the static Physician’s Global Assessment (sPGA) score was 3 or higher, and 10% or more of body surface area was affected.
In the study, at 12 weeks, treatment with ixekizumab was superior to placebo, with sustained responses through 48 weeks. In the follow-up phase, primary and secondary endpoints were sustained through week 108, with patients achieving or maintaining PASI 75 (91.7%), PASI 90 (79%), PASI 100 (55.1%), sPGA 0 or 1 (78.3%), and sPGA 0 (52.4%). Significant improvements in itch were seen at 12 weeks and were sustained with “meaningful improvements in itch for 78.5% of these patients at week 108,” the investigators report.
Among the patients who received ixekizumab, clearance rates in areas that are difficult to treat increased from week 12 to week 108 among those affected. During this time, clearance of nail psoriasis increased from 22.8% to 68.1%, clearance of palmoplantar psoriasis increased from 46.2% to 90%, clearance of scalp psoriasis increased from 70.7% to 76.2%, and clearance of genital psoriasis increased from 83.3% to 87.5%.
No new safety findings during weeks 48-108 of the trial were reported, including no new cases of inflammatory bowel disease (IBD) or Candida infections. The results were reported in JAMA Dermatology.
“Safety is really what we think of most when we are talking about pediatric patients, especially since they may be on these for decades and ... since they most commonly start these therapies in adolescence,” said Amy Paller, MD, the study’s lead author, in an interview. “To be able to take this out 108 weeks, 2 years, is starting to get to a point where we are getting more comfortable with safety. Clearly, no new signals arose.” Dr. Paller is chair of the department of dermatology and professor of dermatology and pediatrics, Northwestern University, Chicago.
One of the biggest concerns with using IL-17 inhibitors such as ixekizumab to manage psoriasis is the development of IBD, said Dr. Paller. She noted that four cases of IBD were reported before the extension phase of the trial but that no new IBD cases were reported after week 48.
“We would not start this as a treatment of choice in someone with Crohn’s disease, or perhaps we would think twice about using it in someone with a strong family history [of Crohn’s disease],” said Dr. Paller, who is also the director of the Skin Biology and Diseases Resource-Based Center at Northwestern. “Otherwise, it does not make me concerned about its use.”
Commenting on the study, Kelly M. Cordoro, MD, professor of dermatology and pediatrics at the University of California, San Francisco, said that the trial’s results provide additional evidence regarding the optimal management of pediatric psoriasis.
“The landscape has shifted toward involving more pediatric patients in clinical trials, thereby providing dermatologists with data to select safe and effective therapies to manage children with psoriasis,” Dr. Cordoro said in an interview. “We have data showing that children with psoriasis have been undertreated, likely because of concerns about safety. The more evidence available from trials such as this, the more likely children are to receive necessary treatment.”
The efficacy data from the study on difficult-to-treat areas of psoriasis, in addition to improvements in BSA and PASI measures, are significant for clinicians deciding on a therapy for patients with psoriasis concentrated in specific body sites. “It was very valuable that the efficacy data was provided by site, such as scalp, palmoplantar, nails, and genital psoriasis, as these are low-BSA but high-impact areas for patients,” said Dr. Cordoro.
The trial data on Crohn’s disease buttress her decision to continue to refrain from initiating ixekizumab in a child with IBD or who is at high risk for IBD. “I was happy to see that there was not a signal for Candida infection,” she added.
Interestingly, in the substudy in the European population, in which there was a double-blind, randomized withdrawal period, fewer patients who were reassigned to receive ixekizumab experienced relapse, compared with those who were reassigned to receive placebo. A total of 90.9% of patients who received placebo experienced relapse, compared with 17.6% of patients treated with ixekizumab. The median time to relapse in the placebo group was 149 days.
“There are data in the adult population that suggest intermittent treatment does allow for recapture of clinical response,” said Dr. Cordoro. “While it is not a large enough dataset to know definitively, this substudy of patients suggests the possibility of intermittent treatment and the ability to regain control [of psoriasis] after a period off drug.”
The study was funded by Eli Lilly. Dr. Paller is an investigator and consultant for Eli Lilly. Several other authors have received grants, personal fees, and/or were a consultant to Eli Lilly, and two authors are Eli Lilly employees. Dr. Cordoro reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
with the interleukin (IL)-17 inhibitor, investigators reported.
In addition, findings of a substudy, which evaluated randomized withdrawal of treatment after 60 weeks, suggest patients were able to regain benefit after not being treated for a period.
Ixekizumab (Taltz) was approved by the U.S. Food and Drug Administration for treating pediatric psoriasis in March 2020 for patients aged 6 years and older with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy.
The trial (IXORA-PEDS) involved 171 patients aged 6-17 years (mean age, 13.5 years; 99 females and 72 males), who were randomly assigned to receive ixekizumab via subcutaneous administration every 4 weeks (115) or placebo for 12 weeks (56). Thereafter, 166 patients continued in an open-label maintenance period in which they were treated every 4 weeks for 12-60 weeks. This was followed by an extension period of up to 108 weeks, which was completed by 139 patients (83.7%). At baseline, the patients’ Psoriasis Area and Severity Index (PASI) score was 12 or higher, the static Physician’s Global Assessment (sPGA) score was 3 or higher, and 10% or more of body surface area was affected.
In the study, at 12 weeks, treatment with ixekizumab was superior to placebo, with sustained responses through 48 weeks. In the follow-up phase, primary and secondary endpoints were sustained through week 108, with patients achieving or maintaining PASI 75 (91.7%), PASI 90 (79%), PASI 100 (55.1%), sPGA 0 or 1 (78.3%), and sPGA 0 (52.4%). Significant improvements in itch were seen at 12 weeks and were sustained with “meaningful improvements in itch for 78.5% of these patients at week 108,” the investigators report.
Among the patients who received ixekizumab, clearance rates in areas that are difficult to treat increased from week 12 to week 108 among those affected. During this time, clearance of nail psoriasis increased from 22.8% to 68.1%, clearance of palmoplantar psoriasis increased from 46.2% to 90%, clearance of scalp psoriasis increased from 70.7% to 76.2%, and clearance of genital psoriasis increased from 83.3% to 87.5%.
No new safety findings during weeks 48-108 of the trial were reported, including no new cases of inflammatory bowel disease (IBD) or Candida infections. The results were reported in JAMA Dermatology.
“Safety is really what we think of most when we are talking about pediatric patients, especially since they may be on these for decades and ... since they most commonly start these therapies in adolescence,” said Amy Paller, MD, the study’s lead author, in an interview. “To be able to take this out 108 weeks, 2 years, is starting to get to a point where we are getting more comfortable with safety. Clearly, no new signals arose.” Dr. Paller is chair of the department of dermatology and professor of dermatology and pediatrics, Northwestern University, Chicago.
One of the biggest concerns with using IL-17 inhibitors such as ixekizumab to manage psoriasis is the development of IBD, said Dr. Paller. She noted that four cases of IBD were reported before the extension phase of the trial but that no new IBD cases were reported after week 48.
“We would not start this as a treatment of choice in someone with Crohn’s disease, or perhaps we would think twice about using it in someone with a strong family history [of Crohn’s disease],” said Dr. Paller, who is also the director of the Skin Biology and Diseases Resource-Based Center at Northwestern. “Otherwise, it does not make me concerned about its use.”
Commenting on the study, Kelly M. Cordoro, MD, professor of dermatology and pediatrics at the University of California, San Francisco, said that the trial’s results provide additional evidence regarding the optimal management of pediatric psoriasis.
“The landscape has shifted toward involving more pediatric patients in clinical trials, thereby providing dermatologists with data to select safe and effective therapies to manage children with psoriasis,” Dr. Cordoro said in an interview. “We have data showing that children with psoriasis have been undertreated, likely because of concerns about safety. The more evidence available from trials such as this, the more likely children are to receive necessary treatment.”
The efficacy data from the study on difficult-to-treat areas of psoriasis, in addition to improvements in BSA and PASI measures, are significant for clinicians deciding on a therapy for patients with psoriasis concentrated in specific body sites. “It was very valuable that the efficacy data was provided by site, such as scalp, palmoplantar, nails, and genital psoriasis, as these are low-BSA but high-impact areas for patients,” said Dr. Cordoro.
The trial data on Crohn’s disease buttress her decision to continue to refrain from initiating ixekizumab in a child with IBD or who is at high risk for IBD. “I was happy to see that there was not a signal for Candida infection,” she added.
Interestingly, in the substudy in the European population, in which there was a double-blind, randomized withdrawal period, fewer patients who were reassigned to receive ixekizumab experienced relapse, compared with those who were reassigned to receive placebo. A total of 90.9% of patients who received placebo experienced relapse, compared with 17.6% of patients treated with ixekizumab. The median time to relapse in the placebo group was 149 days.
“There are data in the adult population that suggest intermittent treatment does allow for recapture of clinical response,” said Dr. Cordoro. “While it is not a large enough dataset to know definitively, this substudy of patients suggests the possibility of intermittent treatment and the ability to regain control [of psoriasis] after a period off drug.”
The study was funded by Eli Lilly. Dr. Paller is an investigator and consultant for Eli Lilly. Several other authors have received grants, personal fees, and/or were a consultant to Eli Lilly, and two authors are Eli Lilly employees. Dr. Cordoro reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
with the interleukin (IL)-17 inhibitor, investigators reported.
In addition, findings of a substudy, which evaluated randomized withdrawal of treatment after 60 weeks, suggest patients were able to regain benefit after not being treated for a period.
Ixekizumab (Taltz) was approved by the U.S. Food and Drug Administration for treating pediatric psoriasis in March 2020 for patients aged 6 years and older with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy.
The trial (IXORA-PEDS) involved 171 patients aged 6-17 years (mean age, 13.5 years; 99 females and 72 males), who were randomly assigned to receive ixekizumab via subcutaneous administration every 4 weeks (115) or placebo for 12 weeks (56). Thereafter, 166 patients continued in an open-label maintenance period in which they were treated every 4 weeks for 12-60 weeks. This was followed by an extension period of up to 108 weeks, which was completed by 139 patients (83.7%). At baseline, the patients’ Psoriasis Area and Severity Index (PASI) score was 12 or higher, the static Physician’s Global Assessment (sPGA) score was 3 or higher, and 10% or more of body surface area was affected.
In the study, at 12 weeks, treatment with ixekizumab was superior to placebo, with sustained responses through 48 weeks. In the follow-up phase, primary and secondary endpoints were sustained through week 108, with patients achieving or maintaining PASI 75 (91.7%), PASI 90 (79%), PASI 100 (55.1%), sPGA 0 or 1 (78.3%), and sPGA 0 (52.4%). Significant improvements in itch were seen at 12 weeks and were sustained with “meaningful improvements in itch for 78.5% of these patients at week 108,” the investigators report.
Among the patients who received ixekizumab, clearance rates in areas that are difficult to treat increased from week 12 to week 108 among those affected. During this time, clearance of nail psoriasis increased from 22.8% to 68.1%, clearance of palmoplantar psoriasis increased from 46.2% to 90%, clearance of scalp psoriasis increased from 70.7% to 76.2%, and clearance of genital psoriasis increased from 83.3% to 87.5%.
No new safety findings during weeks 48-108 of the trial were reported, including no new cases of inflammatory bowel disease (IBD) or Candida infections. The results were reported in JAMA Dermatology.
“Safety is really what we think of most when we are talking about pediatric patients, especially since they may be on these for decades and ... since they most commonly start these therapies in adolescence,” said Amy Paller, MD, the study’s lead author, in an interview. “To be able to take this out 108 weeks, 2 years, is starting to get to a point where we are getting more comfortable with safety. Clearly, no new signals arose.” Dr. Paller is chair of the department of dermatology and professor of dermatology and pediatrics, Northwestern University, Chicago.
One of the biggest concerns with using IL-17 inhibitors such as ixekizumab to manage psoriasis is the development of IBD, said Dr. Paller. She noted that four cases of IBD were reported before the extension phase of the trial but that no new IBD cases were reported after week 48.
“We would not start this as a treatment of choice in someone with Crohn’s disease, or perhaps we would think twice about using it in someone with a strong family history [of Crohn’s disease],” said Dr. Paller, who is also the director of the Skin Biology and Diseases Resource-Based Center at Northwestern. “Otherwise, it does not make me concerned about its use.”
Commenting on the study, Kelly M. Cordoro, MD, professor of dermatology and pediatrics at the University of California, San Francisco, said that the trial’s results provide additional evidence regarding the optimal management of pediatric psoriasis.
“The landscape has shifted toward involving more pediatric patients in clinical trials, thereby providing dermatologists with data to select safe and effective therapies to manage children with psoriasis,” Dr. Cordoro said in an interview. “We have data showing that children with psoriasis have been undertreated, likely because of concerns about safety. The more evidence available from trials such as this, the more likely children are to receive necessary treatment.”
The efficacy data from the study on difficult-to-treat areas of psoriasis, in addition to improvements in BSA and PASI measures, are significant for clinicians deciding on a therapy for patients with psoriasis concentrated in specific body sites. “It was very valuable that the efficacy data was provided by site, such as scalp, palmoplantar, nails, and genital psoriasis, as these are low-BSA but high-impact areas for patients,” said Dr. Cordoro.
The trial data on Crohn’s disease buttress her decision to continue to refrain from initiating ixekizumab in a child with IBD or who is at high risk for IBD. “I was happy to see that there was not a signal for Candida infection,” she added.
Interestingly, in the substudy in the European population, in which there was a double-blind, randomized withdrawal period, fewer patients who were reassigned to receive ixekizumab experienced relapse, compared with those who were reassigned to receive placebo. A total of 90.9% of patients who received placebo experienced relapse, compared with 17.6% of patients treated with ixekizumab. The median time to relapse in the placebo group was 149 days.
“There are data in the adult population that suggest intermittent treatment does allow for recapture of clinical response,” said Dr. Cordoro. “While it is not a large enough dataset to know definitively, this substudy of patients suggests the possibility of intermittent treatment and the ability to regain control [of psoriasis] after a period off drug.”
The study was funded by Eli Lilly. Dr. Paller is an investigator and consultant for Eli Lilly. Several other authors have received grants, personal fees, and/or were a consultant to Eli Lilly, and two authors are Eli Lilly employees. Dr. Cordoro reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Secukinumab’s antipsoriatic effects confirmed in U.S. patient population
and those who up-titrated to 300 mg from the lower approved dose of 150 mg also saw benefits obtained at that level.
Researchers conducted a postmarketing trial of secukinumab in patients at U.S. centers, called CHOICE, after it was approved for psoriasis and PsA in 2015 and 2016 based on trials mainly conducted outside of the United States. The American patients in those studies “had a baseline clinical profile indicating harder-to-treat disease than the total study population, including higher body mass index (BMI), higher tender and swollen joint counts, increased prevalence of enthesitis and dactylitis, and more tumor necrosis factor inhibitor (TNFi) experience,” Tien Q. Nguyen, MD, a dermatologist in private practice in Irvine, Calif., and colleagues wrote in the Journal of Rheumatology.
In order to get a better sense of how secukinumab performs in U.S. patients who have not been treated with biologics, the researchers conducted the multicenter, randomized, double-blind, placebo-controlled, parallel-group, phase 4 CHOICE trial. It recruited patients for about 26 months at 67 U.S. centers during 2016-2018. The 258 patients randomized in the study to 300 mg (n = 103), 150 mg secukinumab (n = 103), or placebo (n = 52) had a mean time since PsA diagnosis of 3.0-3.9 years and all had a mean BMI of greater than 30 kg/m2, with dactylitis present in 48% and enthesitis in 73%. About one-third were taking methotrexate at baseline.
At week 16, patients taking secukinumab 300 mg were about 3.5 times more likely to have 20% improvement in American College of Rheumatology response criteria than with placebo (51.5% vs. 23.1%), whereas the response rate with 150 mg was not significantly different from placebo (36.9%). Rates of achieving ACR50 were significantly greater for both 300- and 150-mg doses versus placebo (28.2% and 24.3% vs. 5.8%), but only 300 mg led to a statistically significant difference in the rate of ACR70 responses, compared with placebo (17.5% vs. 1.9%).
In general, efficacy based on ACR20/50/70 responses and either remission or low disease activity on the Disease Activity in Psoriatic Arthritis index was lower among patients with less than 10 tender joints and less than 10 swollen joints at baseline. Methotrexate use at baseline did not affect ACR20 rates at week 16 in patients taking secukinumab, but the effect of methotrexate on ACR20 rates was noticeable among placebo-treated patients (38.9% vs. 14.7%). Enthesitis appeared to resolve significantly more often among patients on secukinumab, and more patients on secukinumab also had their dactylitis resolve, but the difference was not statistically significant.
Patients with psoriasis affecting more than 3% of their body surface area experienced higher response rates on the Psoriasis Area Severity Index (PASI) for 75%, 90%, and 100% skin lesion clearance than did patients taking placebo.
Patients who switched from 150 mg to 300 mg secukinumab after week 16 in the second treatment period of the trial more often achieved ACR20/50/70 responses by week 52, going from 2.4% to 65.9% of the up-titration subset for ACR20 and from 0% to 34.1% for ACR50 and to 12.2% for ACR70. Patients on placebo who switched also experienced increases in these response rates out to week 52. However, BMI above 30 kg/m2 led to numerically lower ACR50, ACR70, and PASI response rates at week 52.
The researchers noted that the response rates observed in CHOICE were lower than for the pivotal trials used for Food and Drug Administration approval for PsA, which “may have been due to patients in CHOICE having higher disease activity scores at baseline, compared with TNFi-naive patients” in the pivotal trials.
The safety profile of secukinumab appeared to be no different from what has been reported previously. The researchers said that, throughout the 52-week study, the most common adverse events in patients receiving secukinumab were upper respiratory tract infection in about 13% and diarrhea in about 7%. Most adverse events were mild or moderate, with serious adverse events occurring in 9.6% of patients taking secukinumab 300 mg and in 7.8% of patients taking secukinumab 150 mg over the 52 weeks.
“Overall, the findings from CHOICE were consistent with previous studies and demonstrated that secukinumab provides significant and sustained improvements in signs and symptoms of psoriatic arthritis. Our findings suggest that secukinumab 300 mg is safe and efficacious as a first-line biologic treatment for patients with PsA. Further studies will also help determine the optimal dose of secukinumab for treating overweight patients or those with high disease activity at treatment initiation,” the authors wrote.
The study was funded by Novartis, which manufactures secukinumab. Dr. Nguyen and some coauthors reported serving as a consultant, investigator, and/or speaker for numerous pharmaceutical companies, including Novartis.
and those who up-titrated to 300 mg from the lower approved dose of 150 mg also saw benefits obtained at that level.
Researchers conducted a postmarketing trial of secukinumab in patients at U.S. centers, called CHOICE, after it was approved for psoriasis and PsA in 2015 and 2016 based on trials mainly conducted outside of the United States. The American patients in those studies “had a baseline clinical profile indicating harder-to-treat disease than the total study population, including higher body mass index (BMI), higher tender and swollen joint counts, increased prevalence of enthesitis and dactylitis, and more tumor necrosis factor inhibitor (TNFi) experience,” Tien Q. Nguyen, MD, a dermatologist in private practice in Irvine, Calif., and colleagues wrote in the Journal of Rheumatology.
In order to get a better sense of how secukinumab performs in U.S. patients who have not been treated with biologics, the researchers conducted the multicenter, randomized, double-blind, placebo-controlled, parallel-group, phase 4 CHOICE trial. It recruited patients for about 26 months at 67 U.S. centers during 2016-2018. The 258 patients randomized in the study to 300 mg (n = 103), 150 mg secukinumab (n = 103), or placebo (n = 52) had a mean time since PsA diagnosis of 3.0-3.9 years and all had a mean BMI of greater than 30 kg/m2, with dactylitis present in 48% and enthesitis in 73%. About one-third were taking methotrexate at baseline.
At week 16, patients taking secukinumab 300 mg were about 3.5 times more likely to have 20% improvement in American College of Rheumatology response criteria than with placebo (51.5% vs. 23.1%), whereas the response rate with 150 mg was not significantly different from placebo (36.9%). Rates of achieving ACR50 were significantly greater for both 300- and 150-mg doses versus placebo (28.2% and 24.3% vs. 5.8%), but only 300 mg led to a statistically significant difference in the rate of ACR70 responses, compared with placebo (17.5% vs. 1.9%).
In general, efficacy based on ACR20/50/70 responses and either remission or low disease activity on the Disease Activity in Psoriatic Arthritis index was lower among patients with less than 10 tender joints and less than 10 swollen joints at baseline. Methotrexate use at baseline did not affect ACR20 rates at week 16 in patients taking secukinumab, but the effect of methotrexate on ACR20 rates was noticeable among placebo-treated patients (38.9% vs. 14.7%). Enthesitis appeared to resolve significantly more often among patients on secukinumab, and more patients on secukinumab also had their dactylitis resolve, but the difference was not statistically significant.
Patients with psoriasis affecting more than 3% of their body surface area experienced higher response rates on the Psoriasis Area Severity Index (PASI) for 75%, 90%, and 100% skin lesion clearance than did patients taking placebo.
Patients who switched from 150 mg to 300 mg secukinumab after week 16 in the second treatment period of the trial more often achieved ACR20/50/70 responses by week 52, going from 2.4% to 65.9% of the up-titration subset for ACR20 and from 0% to 34.1% for ACR50 and to 12.2% for ACR70. Patients on placebo who switched also experienced increases in these response rates out to week 52. However, BMI above 30 kg/m2 led to numerically lower ACR50, ACR70, and PASI response rates at week 52.
The researchers noted that the response rates observed in CHOICE were lower than for the pivotal trials used for Food and Drug Administration approval for PsA, which “may have been due to patients in CHOICE having higher disease activity scores at baseline, compared with TNFi-naive patients” in the pivotal trials.
The safety profile of secukinumab appeared to be no different from what has been reported previously. The researchers said that, throughout the 52-week study, the most common adverse events in patients receiving secukinumab were upper respiratory tract infection in about 13% and diarrhea in about 7%. Most adverse events were mild or moderate, with serious adverse events occurring in 9.6% of patients taking secukinumab 300 mg and in 7.8% of patients taking secukinumab 150 mg over the 52 weeks.
“Overall, the findings from CHOICE were consistent with previous studies and demonstrated that secukinumab provides significant and sustained improvements in signs and symptoms of psoriatic arthritis. Our findings suggest that secukinumab 300 mg is safe and efficacious as a first-line biologic treatment for patients with PsA. Further studies will also help determine the optimal dose of secukinumab for treating overweight patients or those with high disease activity at treatment initiation,” the authors wrote.
The study was funded by Novartis, which manufactures secukinumab. Dr. Nguyen and some coauthors reported serving as a consultant, investigator, and/or speaker for numerous pharmaceutical companies, including Novartis.
and those who up-titrated to 300 mg from the lower approved dose of 150 mg also saw benefits obtained at that level.
Researchers conducted a postmarketing trial of secukinumab in patients at U.S. centers, called CHOICE, after it was approved for psoriasis and PsA in 2015 and 2016 based on trials mainly conducted outside of the United States. The American patients in those studies “had a baseline clinical profile indicating harder-to-treat disease than the total study population, including higher body mass index (BMI), higher tender and swollen joint counts, increased prevalence of enthesitis and dactylitis, and more tumor necrosis factor inhibitor (TNFi) experience,” Tien Q. Nguyen, MD, a dermatologist in private practice in Irvine, Calif., and colleagues wrote in the Journal of Rheumatology.
In order to get a better sense of how secukinumab performs in U.S. patients who have not been treated with biologics, the researchers conducted the multicenter, randomized, double-blind, placebo-controlled, parallel-group, phase 4 CHOICE trial. It recruited patients for about 26 months at 67 U.S. centers during 2016-2018. The 258 patients randomized in the study to 300 mg (n = 103), 150 mg secukinumab (n = 103), or placebo (n = 52) had a mean time since PsA diagnosis of 3.0-3.9 years and all had a mean BMI of greater than 30 kg/m2, with dactylitis present in 48% and enthesitis in 73%. About one-third were taking methotrexate at baseline.
At week 16, patients taking secukinumab 300 mg were about 3.5 times more likely to have 20% improvement in American College of Rheumatology response criteria than with placebo (51.5% vs. 23.1%), whereas the response rate with 150 mg was not significantly different from placebo (36.9%). Rates of achieving ACR50 were significantly greater for both 300- and 150-mg doses versus placebo (28.2% and 24.3% vs. 5.8%), but only 300 mg led to a statistically significant difference in the rate of ACR70 responses, compared with placebo (17.5% vs. 1.9%).
In general, efficacy based on ACR20/50/70 responses and either remission or low disease activity on the Disease Activity in Psoriatic Arthritis index was lower among patients with less than 10 tender joints and less than 10 swollen joints at baseline. Methotrexate use at baseline did not affect ACR20 rates at week 16 in patients taking secukinumab, but the effect of methotrexate on ACR20 rates was noticeable among placebo-treated patients (38.9% vs. 14.7%). Enthesitis appeared to resolve significantly more often among patients on secukinumab, and more patients on secukinumab also had their dactylitis resolve, but the difference was not statistically significant.
Patients with psoriasis affecting more than 3% of their body surface area experienced higher response rates on the Psoriasis Area Severity Index (PASI) for 75%, 90%, and 100% skin lesion clearance than did patients taking placebo.
Patients who switched from 150 mg to 300 mg secukinumab after week 16 in the second treatment period of the trial more often achieved ACR20/50/70 responses by week 52, going from 2.4% to 65.9% of the up-titration subset for ACR20 and from 0% to 34.1% for ACR50 and to 12.2% for ACR70. Patients on placebo who switched also experienced increases in these response rates out to week 52. However, BMI above 30 kg/m2 led to numerically lower ACR50, ACR70, and PASI response rates at week 52.
The researchers noted that the response rates observed in CHOICE were lower than for the pivotal trials used for Food and Drug Administration approval for PsA, which “may have been due to patients in CHOICE having higher disease activity scores at baseline, compared with TNFi-naive patients” in the pivotal trials.
The safety profile of secukinumab appeared to be no different from what has been reported previously. The researchers said that, throughout the 52-week study, the most common adverse events in patients receiving secukinumab were upper respiratory tract infection in about 13% and diarrhea in about 7%. Most adverse events were mild or moderate, with serious adverse events occurring in 9.6% of patients taking secukinumab 300 mg and in 7.8% of patients taking secukinumab 150 mg over the 52 weeks.
“Overall, the findings from CHOICE were consistent with previous studies and demonstrated that secukinumab provides significant and sustained improvements in signs and symptoms of psoriatic arthritis. Our findings suggest that secukinumab 300 mg is safe and efficacious as a first-line biologic treatment for patients with PsA. Further studies will also help determine the optimal dose of secukinumab for treating overweight patients or those with high disease activity at treatment initiation,” the authors wrote.
The study was funded by Novartis, which manufactures secukinumab. Dr. Nguyen and some coauthors reported serving as a consultant, investigator, and/or speaker for numerous pharmaceutical companies, including Novartis.
FROM THE JOURNAL OF RHEUMATOLOGY
Cupping in dermatology
My inspiration to write about cupping this month stems from the perception that everyone seems to be talking about it, from a facialist who suggested it for me to a coworker who swears by cupping to treat her allergies. Cupping is by no means a novel procedure. Its use as a health therapy dates back thousands of years to ancient Egypt (1500 BCE), ancient Greece (described by Hippocrates), ancient Rome (described by the Greek physician Galen), China (during the Han dynasty, 206 BCE to 220 CE) and traditional Islamic culture.1 Over the past decade, the popularity of this ancient procedure has been increasing in the United States.1 Cupping has been applied as a remedy for various dermatologic and medical conditions, including herpes zoster, headaches, diminished appetite, maldigestion, abscess evacuation, narcolepsy, pain, fever, dysmenorrhea, and gout.1,2
Theories on the mechanism(s) of action
The practice of cupping is differentiated into dry and wet cupping.1,2 Traditionally, with dry cupping, a flame is applied to heat the air inside a thick glass cup (rather than the cup itself).1 The cup is placed on the skin surface, and negative pressure suctions the skin into the cup. Wet cupping differs mainly from dry cupping in that it involves blood-letting. Cups made of either silicone or glass of varying size and shapes are used. Modern adaptations to cupping include needle, herbal, and pulsatile cupping, as well as a “moving cupping” technique (vs. traditionally stationary cups).1
There are several theories, many of which are derived from the nondermatologic literature (that is, pain management), as to how cupping may deliver a clinical benefit. Some theories are based in scientific and medical principles, whereas other theories are more whimsical – specifically, that cupping draws out evil spirits.2 Studies of dry cupping have suggested that the procedure results in increased oxygenation of muscles via a local increase in oxygenated hemoglobin, which may help improve muscular activity and reduce pain.1 As theorized by Lowe in 2017, negative pressure exerted by dry cupping leads to stretching and dilation of capillaries, which increases blood flow.3 Wet cupping has been shown to increase heat shock protein 70 (HSP70) and beta-endorphin expression in rat models, which is thought to facilitate pain management.1 Removal of oxidants and reduction of reactive oxygen species in the blood is believed to be among the benefits of wet cupping.1
Cupping in general dermatology
While , as well as various inflammatory conditions.
Herpes zoster
In 2010, Cao et al. reported on their systematic review of wet cupping after completing searches of multiple databases (that is, PubMed, the Cochrane Library [Issue 3, 2008], China Network Knowledge Infrastructure, Chinese Scientific Journal Database, and Wan Fang Database). They identified eight randomized controlled trials involving 651 patients, with meta-analyses revealing that wet cupping performed better than medications in terms of the number of “cured” patients, number of patients with improved symptoms, and a lower incidence of postherpetic neuralgia. Wet cupping, in addition to medication, was also found to be superior to medication alone in multiple patients. The researchers concluded that wet cupping appears to effectively treat herpes zoster.4 However, the study failed to identify which medications were used to treat herpes zoster. In the United States, common medications for herpes zoster include acyclovir, valacyclovir, steroids, gabapentin, and other neuromodulators. Without knowing which medications were used, it is difficult to compare cupping to medication in terms of efficacy in treating herpes zoster.
Urticaria
Urticaria (hives) is an inflammatory skin condition that can be very uncomfortable for patients but often resolves without intervention within several months after onset. In 2001, Li and Ding reported on the treatment with cupping of 40 patients with urticaria. The cure rate among the treatment group was cited as 55%, compared with 30% in the control group, who were treated with a traditional Chinese remedy and an unidentified first-generation antihistamine.1,5 In 2020, Xiao et al. conducted a systematic review and meta-analysis of cupping therapy for patients with chronic urticaria. They identified 13 comparisons from 12 randomized controlled trials involving 842 subjects. The investigators found no significant differences between wet cupping and medication usage. They also found that cupping combined with antihistamine treatment was superior to antihistamines alone, and cupping therapy with acupuncture was more effective than acupuncture alone. The investigators did call for caution, citing the poor quality of the studies reviewed.6
It is important to note that it is difficult to attribute resolution of urticaria to the use of cupping given the self-resolution often associated with this condition. Antihistamines are the mainstay of therapy for urticaria, but in my personal experience, patients are not entirely satisfied with the level of symptom control with antihistamines alone and often search for alternative therapies to control the pesky hives and associated itch. In 2014, omalizumab (Xolair) was approved for treating chronic idiopathic urticaria, which has helped patients control symptoms of chronic idiopathic urticaria without needing to take antihistamines. There was no indication that the studies reviewed by Xiao et al. compared cupping against this new effective treatment. Therefore, these studies comparing cupping to medical management are outdated.
Acne, eczema, and psoriasis
Soliman’s 2018 review of cupping in dermatology included a few studies on these common cutaneous conditions. For instance, a 2013 single-blind prospective study by Xu et al. reported on the results of patients with moderate acne who received wet cupping (in the form of prickling bloodletting) twice weekly for 6 weeks.7 They reported that patients demonstrated improvement in the global acne grading system (GAGS) score by the end of the trial.1,7 Unfortunately, cupping was not compared with standard acne treatments (that is, benzoyl peroxide, topical and oral antibiotics, isotretinoin, topical retinoids, spironolactone).
In evaluating cupping for acute eczema, wet cupping was compared with oral loratadine and topical ointments in a 2007 study by Yao and Li. They divided 88 cases into treatment and control groups, with the former group (n = 46) receiving bloodletting puncturing and cupping and the control group (n = 42) receiving oral loratadine and topical Pairuisong (an herbal ointment used in Chinese medicine). The investigators observed no significant difference in total effective rates but a superior difference in the rates of responses that were considered “cured” and “markedly effective” in favor of the cupping treatment.1,8 However, a case report by Hon et al. has indicated that cupping therapy may be associated with more harm than benefit when used as an eczema treatment.1,9
In addition, it is important to note that the past 5 years have been gamechanging in the management of chronic eczema in terms of the array of novel and effective therapies (e.g., dupilumab and JAK inhibitors) and chronic moderate-to-severe eczema has become very treatable. Similarly, acute eczema is often successfully managed with topical steroids, calcineurin inhibitors, and emollients. As such, there is no compelling reason to consider an unproven treatment such as cupping.
In 2020, Xing et al. reviewed 16 randomized controlled trials assessing the use of “moving cupping” for plaque psoriasis, with 1,164 patients meeting inclusion criteria. Moving cupping was found to be significantly more effective than “no-moving” cupping therapy, and moving cupping, combined with medications, performed better than medications alone.10 None of the trials evaluated in this study included randomized controlled trials that compared patients using any of the more modern psoriasis medications, specifically biologics. And, again, the studies evaluated were not of the highest quality.
The data that support cupping, as summarized above, are based mostly on case reports, and strong double-blind prospective studies are lacking. Additionally, most of the studies cited gauged the efficacy of cupping using qualitative endpoints, rather than standardized quantitative endpoints and scales. Moreover, spontaneous remission of various dermatoses can occur, or they can improve over time, including acute eczema, psoriasis, and, especially, urticaria.
Adverse effects of cupping
Often alternative therapies are seen as “benign” and without adverse effects. However, complications can result from cupping. Trauma can be induced from the cupping itself by damaging superficial blood vessels and causing bruising.1,11 Blistering can also occur secondary to the suction effect, and the epidermal and dermal layers of the skin can be separated.1,11 Further, burns and discoloration have also been noted secondary to heat, trauma, and post inflammatory pigmentary changes.1,11 Another risk of cupping is the Koebner phenomenon, which occurs with psoriasis, with new lesions appearing in traumatized skin.12 Other adverse outcomes that have been reported with cupping include reactivation of herpes simplex virus secondary to skin trauma, iron deficiency anemia (secondary to blood loss), panniculitis, infections, and residual marks mistaken for signs of child abuse.1,11
Cupping in aesthetic dermatology
Facial cupping, a distinct practice from body cupping used to treat general dermatology conditions described previously, is also increasing in popularity. This practice is usually conducted in association with a facial or facial acupuncture by an aesthetician or other licensed professional. It can also be performed using at-home kits. The marketing claims for facial cupping cite improved tightening and contouring of facial skin, increased facial microcirculation and collagen synthesis, and enhanced lymphatic flow to aid with facial puffiness or swelling. One supposed mechanism for these benefits is that cupping increases blood flow. Interestingly, there was a 2020 animal study in which photoacoustic imaging of a mouse ear revealed increased temporary blood flow in the cupping microenvironment.13 Currently, however, there is no evidence in the English scientific literature that supports facial cupping. The benefits attributed to facial cupping for aesthetic purposes have emerged only in personal anecdotes. The temporary increase in blood flow may induce inflammation and swelling that adds volume to the face and temporarily diminishes wrinkles. However, this temporary plumpness may be associated with adverse effects, such as local trauma, irritation, bruising, postinflammatory pigmentary alteration, or even herpes reactivation. In my opinion, the possible adverse effects of cupping outweigh any potential benefit, especially given the insufficient evidence supporting the utility of cupping for cosmetic enhancement.
Summary
There is increasing interest among patients to incorporate complementary and alternative medicine – including the ancient tradition of cupping – in managing medical dermatologic conditions. However, current evidence supporting cupping as an effective therapeutic strategy is not strong, with most studies to date appearing to be of poor quality or not sufficiently convincing to displace standard therapies. Our medical strategies for managing chronic dermatologic conditions, particularly inflammatory disorders, continue to improve from both a safety and a proven efficacy standpoint. Therefore, I would not forgo medical management in favor of cupping. While cupping can be used as an adjunct therapy, I would caution patients about possible adverse side effects. In the aesthetic world, cupping is also gaining popularity, but this trend is also not supported by current evidence or studies, at least in the Western literature.
Dr. Goldman is a dermatologist in private practice in Miami and specializes in cosmetic and general dermatology. She practices at Baumann Cosmetic & Research Institute and is also opening a general dermatology practice. Write to her at [email protected] or message her on Instragram @DrChloeGoldman. Dr. Goldman receives compensation to create social media content for Replenix, a skin care company. She has no other disclosures.
References
1. Soliman Y et al. Acta Dermatovenerol Alp Pannonica Adriat. 2018 Jun;27(2):103-7.
2. França K and Lotti T. Advances in Integrative Dermatology. John Wiley & Sons, 2019.
3. Lowe DT. Complement Ther Clin Pract. 2017 Nov;29:162-8.
4.Cao H et al. Altern Ther Health Med. 2010 Nov-Dec;16(6):48-54.
5. Li L and Ding J. J Tradit Chin Med. 2001 Mar;21(1):37-8.
6. Xiao XJ et al. J Integr Med. 2020 Jul;18(4):303-12.
7. Xu J et al. J Tradit Chin Med. 2013 Dec;33(6):752-6.
8. Yao J et al. Zhongguo Zhen Jiu. 2007; Jun;27(6):424-6.
9. Hon KL et al. Case Rep Pediatr. 2013;2013:605829.
10. Xing M et al. Medicine (Baltimore). 2020 Oct 9;99(41):e22539.
11. Kim TH et al. Eur J Integr Med. 2014 Aug 1;6(4):434-40.
12. Vender R and Vender R. J Cutan Med Surg. 2015 May-Jun;19(3):320-2.
13. Zhou Y et al. Biomed Opt Express. 2020 Apr 6;11(5):2394-401.
This article was updated 4/25/22.
My inspiration to write about cupping this month stems from the perception that everyone seems to be talking about it, from a facialist who suggested it for me to a coworker who swears by cupping to treat her allergies. Cupping is by no means a novel procedure. Its use as a health therapy dates back thousands of years to ancient Egypt (1500 BCE), ancient Greece (described by Hippocrates), ancient Rome (described by the Greek physician Galen), China (during the Han dynasty, 206 BCE to 220 CE) and traditional Islamic culture.1 Over the past decade, the popularity of this ancient procedure has been increasing in the United States.1 Cupping has been applied as a remedy for various dermatologic and medical conditions, including herpes zoster, headaches, diminished appetite, maldigestion, abscess evacuation, narcolepsy, pain, fever, dysmenorrhea, and gout.1,2
Theories on the mechanism(s) of action
The practice of cupping is differentiated into dry and wet cupping.1,2 Traditionally, with dry cupping, a flame is applied to heat the air inside a thick glass cup (rather than the cup itself).1 The cup is placed on the skin surface, and negative pressure suctions the skin into the cup. Wet cupping differs mainly from dry cupping in that it involves blood-letting. Cups made of either silicone or glass of varying size and shapes are used. Modern adaptations to cupping include needle, herbal, and pulsatile cupping, as well as a “moving cupping” technique (vs. traditionally stationary cups).1
There are several theories, many of which are derived from the nondermatologic literature (that is, pain management), as to how cupping may deliver a clinical benefit. Some theories are based in scientific and medical principles, whereas other theories are more whimsical – specifically, that cupping draws out evil spirits.2 Studies of dry cupping have suggested that the procedure results in increased oxygenation of muscles via a local increase in oxygenated hemoglobin, which may help improve muscular activity and reduce pain.1 As theorized by Lowe in 2017, negative pressure exerted by dry cupping leads to stretching and dilation of capillaries, which increases blood flow.3 Wet cupping has been shown to increase heat shock protein 70 (HSP70) and beta-endorphin expression in rat models, which is thought to facilitate pain management.1 Removal of oxidants and reduction of reactive oxygen species in the blood is believed to be among the benefits of wet cupping.1
Cupping in general dermatology
While , as well as various inflammatory conditions.
Herpes zoster
In 2010, Cao et al. reported on their systematic review of wet cupping after completing searches of multiple databases (that is, PubMed, the Cochrane Library [Issue 3, 2008], China Network Knowledge Infrastructure, Chinese Scientific Journal Database, and Wan Fang Database). They identified eight randomized controlled trials involving 651 patients, with meta-analyses revealing that wet cupping performed better than medications in terms of the number of “cured” patients, number of patients with improved symptoms, and a lower incidence of postherpetic neuralgia. Wet cupping, in addition to medication, was also found to be superior to medication alone in multiple patients. The researchers concluded that wet cupping appears to effectively treat herpes zoster.4 However, the study failed to identify which medications were used to treat herpes zoster. In the United States, common medications for herpes zoster include acyclovir, valacyclovir, steroids, gabapentin, and other neuromodulators. Without knowing which medications were used, it is difficult to compare cupping to medication in terms of efficacy in treating herpes zoster.
Urticaria
Urticaria (hives) is an inflammatory skin condition that can be very uncomfortable for patients but often resolves without intervention within several months after onset. In 2001, Li and Ding reported on the treatment with cupping of 40 patients with urticaria. The cure rate among the treatment group was cited as 55%, compared with 30% in the control group, who were treated with a traditional Chinese remedy and an unidentified first-generation antihistamine.1,5 In 2020, Xiao et al. conducted a systematic review and meta-analysis of cupping therapy for patients with chronic urticaria. They identified 13 comparisons from 12 randomized controlled trials involving 842 subjects. The investigators found no significant differences between wet cupping and medication usage. They also found that cupping combined with antihistamine treatment was superior to antihistamines alone, and cupping therapy with acupuncture was more effective than acupuncture alone. The investigators did call for caution, citing the poor quality of the studies reviewed.6
It is important to note that it is difficult to attribute resolution of urticaria to the use of cupping given the self-resolution often associated with this condition. Antihistamines are the mainstay of therapy for urticaria, but in my personal experience, patients are not entirely satisfied with the level of symptom control with antihistamines alone and often search for alternative therapies to control the pesky hives and associated itch. In 2014, omalizumab (Xolair) was approved for treating chronic idiopathic urticaria, which has helped patients control symptoms of chronic idiopathic urticaria without needing to take antihistamines. There was no indication that the studies reviewed by Xiao et al. compared cupping against this new effective treatment. Therefore, these studies comparing cupping to medical management are outdated.
Acne, eczema, and psoriasis
Soliman’s 2018 review of cupping in dermatology included a few studies on these common cutaneous conditions. For instance, a 2013 single-blind prospective study by Xu et al. reported on the results of patients with moderate acne who received wet cupping (in the form of prickling bloodletting) twice weekly for 6 weeks.7 They reported that patients demonstrated improvement in the global acne grading system (GAGS) score by the end of the trial.1,7 Unfortunately, cupping was not compared with standard acne treatments (that is, benzoyl peroxide, topical and oral antibiotics, isotretinoin, topical retinoids, spironolactone).
In evaluating cupping for acute eczema, wet cupping was compared with oral loratadine and topical ointments in a 2007 study by Yao and Li. They divided 88 cases into treatment and control groups, with the former group (n = 46) receiving bloodletting puncturing and cupping and the control group (n = 42) receiving oral loratadine and topical Pairuisong (an herbal ointment used in Chinese medicine). The investigators observed no significant difference in total effective rates but a superior difference in the rates of responses that were considered “cured” and “markedly effective” in favor of the cupping treatment.1,8 However, a case report by Hon et al. has indicated that cupping therapy may be associated with more harm than benefit when used as an eczema treatment.1,9
In addition, it is important to note that the past 5 years have been gamechanging in the management of chronic eczema in terms of the array of novel and effective therapies (e.g., dupilumab and JAK inhibitors) and chronic moderate-to-severe eczema has become very treatable. Similarly, acute eczema is often successfully managed with topical steroids, calcineurin inhibitors, and emollients. As such, there is no compelling reason to consider an unproven treatment such as cupping.
In 2020, Xing et al. reviewed 16 randomized controlled trials assessing the use of “moving cupping” for plaque psoriasis, with 1,164 patients meeting inclusion criteria. Moving cupping was found to be significantly more effective than “no-moving” cupping therapy, and moving cupping, combined with medications, performed better than medications alone.10 None of the trials evaluated in this study included randomized controlled trials that compared patients using any of the more modern psoriasis medications, specifically biologics. And, again, the studies evaluated were not of the highest quality.
The data that support cupping, as summarized above, are based mostly on case reports, and strong double-blind prospective studies are lacking. Additionally, most of the studies cited gauged the efficacy of cupping using qualitative endpoints, rather than standardized quantitative endpoints and scales. Moreover, spontaneous remission of various dermatoses can occur, or they can improve over time, including acute eczema, psoriasis, and, especially, urticaria.
Adverse effects of cupping
Often alternative therapies are seen as “benign” and without adverse effects. However, complications can result from cupping. Trauma can be induced from the cupping itself by damaging superficial blood vessels and causing bruising.1,11 Blistering can also occur secondary to the suction effect, and the epidermal and dermal layers of the skin can be separated.1,11 Further, burns and discoloration have also been noted secondary to heat, trauma, and post inflammatory pigmentary changes.1,11 Another risk of cupping is the Koebner phenomenon, which occurs with psoriasis, with new lesions appearing in traumatized skin.12 Other adverse outcomes that have been reported with cupping include reactivation of herpes simplex virus secondary to skin trauma, iron deficiency anemia (secondary to blood loss), panniculitis, infections, and residual marks mistaken for signs of child abuse.1,11
Cupping in aesthetic dermatology
Facial cupping, a distinct practice from body cupping used to treat general dermatology conditions described previously, is also increasing in popularity. This practice is usually conducted in association with a facial or facial acupuncture by an aesthetician or other licensed professional. It can also be performed using at-home kits. The marketing claims for facial cupping cite improved tightening and contouring of facial skin, increased facial microcirculation and collagen synthesis, and enhanced lymphatic flow to aid with facial puffiness or swelling. One supposed mechanism for these benefits is that cupping increases blood flow. Interestingly, there was a 2020 animal study in which photoacoustic imaging of a mouse ear revealed increased temporary blood flow in the cupping microenvironment.13 Currently, however, there is no evidence in the English scientific literature that supports facial cupping. The benefits attributed to facial cupping for aesthetic purposes have emerged only in personal anecdotes. The temporary increase in blood flow may induce inflammation and swelling that adds volume to the face and temporarily diminishes wrinkles. However, this temporary plumpness may be associated with adverse effects, such as local trauma, irritation, bruising, postinflammatory pigmentary alteration, or even herpes reactivation. In my opinion, the possible adverse effects of cupping outweigh any potential benefit, especially given the insufficient evidence supporting the utility of cupping for cosmetic enhancement.
Summary
There is increasing interest among patients to incorporate complementary and alternative medicine – including the ancient tradition of cupping – in managing medical dermatologic conditions. However, current evidence supporting cupping as an effective therapeutic strategy is not strong, with most studies to date appearing to be of poor quality or not sufficiently convincing to displace standard therapies. Our medical strategies for managing chronic dermatologic conditions, particularly inflammatory disorders, continue to improve from both a safety and a proven efficacy standpoint. Therefore, I would not forgo medical management in favor of cupping. While cupping can be used as an adjunct therapy, I would caution patients about possible adverse side effects. In the aesthetic world, cupping is also gaining popularity, but this trend is also not supported by current evidence or studies, at least in the Western literature.
Dr. Goldman is a dermatologist in private practice in Miami and specializes in cosmetic and general dermatology. She practices at Baumann Cosmetic & Research Institute and is also opening a general dermatology practice. Write to her at [email protected] or message her on Instragram @DrChloeGoldman. Dr. Goldman receives compensation to create social media content for Replenix, a skin care company. She has no other disclosures.
References
1. Soliman Y et al. Acta Dermatovenerol Alp Pannonica Adriat. 2018 Jun;27(2):103-7.
2. França K and Lotti T. Advances in Integrative Dermatology. John Wiley & Sons, 2019.
3. Lowe DT. Complement Ther Clin Pract. 2017 Nov;29:162-8.
4.Cao H et al. Altern Ther Health Med. 2010 Nov-Dec;16(6):48-54.
5. Li L and Ding J. J Tradit Chin Med. 2001 Mar;21(1):37-8.
6. Xiao XJ et al. J Integr Med. 2020 Jul;18(4):303-12.
7. Xu J et al. J Tradit Chin Med. 2013 Dec;33(6):752-6.
8. Yao J et al. Zhongguo Zhen Jiu. 2007; Jun;27(6):424-6.
9. Hon KL et al. Case Rep Pediatr. 2013;2013:605829.
10. Xing M et al. Medicine (Baltimore). 2020 Oct 9;99(41):e22539.
11. Kim TH et al. Eur J Integr Med. 2014 Aug 1;6(4):434-40.
12. Vender R and Vender R. J Cutan Med Surg. 2015 May-Jun;19(3):320-2.
13. Zhou Y et al. Biomed Opt Express. 2020 Apr 6;11(5):2394-401.
This article was updated 4/25/22.
My inspiration to write about cupping this month stems from the perception that everyone seems to be talking about it, from a facialist who suggested it for me to a coworker who swears by cupping to treat her allergies. Cupping is by no means a novel procedure. Its use as a health therapy dates back thousands of years to ancient Egypt (1500 BCE), ancient Greece (described by Hippocrates), ancient Rome (described by the Greek physician Galen), China (during the Han dynasty, 206 BCE to 220 CE) and traditional Islamic culture.1 Over the past decade, the popularity of this ancient procedure has been increasing in the United States.1 Cupping has been applied as a remedy for various dermatologic and medical conditions, including herpes zoster, headaches, diminished appetite, maldigestion, abscess evacuation, narcolepsy, pain, fever, dysmenorrhea, and gout.1,2
Theories on the mechanism(s) of action
The practice of cupping is differentiated into dry and wet cupping.1,2 Traditionally, with dry cupping, a flame is applied to heat the air inside a thick glass cup (rather than the cup itself).1 The cup is placed on the skin surface, and negative pressure suctions the skin into the cup. Wet cupping differs mainly from dry cupping in that it involves blood-letting. Cups made of either silicone or glass of varying size and shapes are used. Modern adaptations to cupping include needle, herbal, and pulsatile cupping, as well as a “moving cupping” technique (vs. traditionally stationary cups).1
There are several theories, many of which are derived from the nondermatologic literature (that is, pain management), as to how cupping may deliver a clinical benefit. Some theories are based in scientific and medical principles, whereas other theories are more whimsical – specifically, that cupping draws out evil spirits.2 Studies of dry cupping have suggested that the procedure results in increased oxygenation of muscles via a local increase in oxygenated hemoglobin, which may help improve muscular activity and reduce pain.1 As theorized by Lowe in 2017, negative pressure exerted by dry cupping leads to stretching and dilation of capillaries, which increases blood flow.3 Wet cupping has been shown to increase heat shock protein 70 (HSP70) and beta-endorphin expression in rat models, which is thought to facilitate pain management.1 Removal of oxidants and reduction of reactive oxygen species in the blood is believed to be among the benefits of wet cupping.1
Cupping in general dermatology
While , as well as various inflammatory conditions.
Herpes zoster
In 2010, Cao et al. reported on their systematic review of wet cupping after completing searches of multiple databases (that is, PubMed, the Cochrane Library [Issue 3, 2008], China Network Knowledge Infrastructure, Chinese Scientific Journal Database, and Wan Fang Database). They identified eight randomized controlled trials involving 651 patients, with meta-analyses revealing that wet cupping performed better than medications in terms of the number of “cured” patients, number of patients with improved symptoms, and a lower incidence of postherpetic neuralgia. Wet cupping, in addition to medication, was also found to be superior to medication alone in multiple patients. The researchers concluded that wet cupping appears to effectively treat herpes zoster.4 However, the study failed to identify which medications were used to treat herpes zoster. In the United States, common medications for herpes zoster include acyclovir, valacyclovir, steroids, gabapentin, and other neuromodulators. Without knowing which medications were used, it is difficult to compare cupping to medication in terms of efficacy in treating herpes zoster.
Urticaria
Urticaria (hives) is an inflammatory skin condition that can be very uncomfortable for patients but often resolves without intervention within several months after onset. In 2001, Li and Ding reported on the treatment with cupping of 40 patients with urticaria. The cure rate among the treatment group was cited as 55%, compared with 30% in the control group, who were treated with a traditional Chinese remedy and an unidentified first-generation antihistamine.1,5 In 2020, Xiao et al. conducted a systematic review and meta-analysis of cupping therapy for patients with chronic urticaria. They identified 13 comparisons from 12 randomized controlled trials involving 842 subjects. The investigators found no significant differences between wet cupping and medication usage. They also found that cupping combined with antihistamine treatment was superior to antihistamines alone, and cupping therapy with acupuncture was more effective than acupuncture alone. The investigators did call for caution, citing the poor quality of the studies reviewed.6
It is important to note that it is difficult to attribute resolution of urticaria to the use of cupping given the self-resolution often associated with this condition. Antihistamines are the mainstay of therapy for urticaria, but in my personal experience, patients are not entirely satisfied with the level of symptom control with antihistamines alone and often search for alternative therapies to control the pesky hives and associated itch. In 2014, omalizumab (Xolair) was approved for treating chronic idiopathic urticaria, which has helped patients control symptoms of chronic idiopathic urticaria without needing to take antihistamines. There was no indication that the studies reviewed by Xiao et al. compared cupping against this new effective treatment. Therefore, these studies comparing cupping to medical management are outdated.
Acne, eczema, and psoriasis
Soliman’s 2018 review of cupping in dermatology included a few studies on these common cutaneous conditions. For instance, a 2013 single-blind prospective study by Xu et al. reported on the results of patients with moderate acne who received wet cupping (in the form of prickling bloodletting) twice weekly for 6 weeks.7 They reported that patients demonstrated improvement in the global acne grading system (GAGS) score by the end of the trial.1,7 Unfortunately, cupping was not compared with standard acne treatments (that is, benzoyl peroxide, topical and oral antibiotics, isotretinoin, topical retinoids, spironolactone).
In evaluating cupping for acute eczema, wet cupping was compared with oral loratadine and topical ointments in a 2007 study by Yao and Li. They divided 88 cases into treatment and control groups, with the former group (n = 46) receiving bloodletting puncturing and cupping and the control group (n = 42) receiving oral loratadine and topical Pairuisong (an herbal ointment used in Chinese medicine). The investigators observed no significant difference in total effective rates but a superior difference in the rates of responses that were considered “cured” and “markedly effective” in favor of the cupping treatment.1,8 However, a case report by Hon et al. has indicated that cupping therapy may be associated with more harm than benefit when used as an eczema treatment.1,9
In addition, it is important to note that the past 5 years have been gamechanging in the management of chronic eczema in terms of the array of novel and effective therapies (e.g., dupilumab and JAK inhibitors) and chronic moderate-to-severe eczema has become very treatable. Similarly, acute eczema is often successfully managed with topical steroids, calcineurin inhibitors, and emollients. As such, there is no compelling reason to consider an unproven treatment such as cupping.
In 2020, Xing et al. reviewed 16 randomized controlled trials assessing the use of “moving cupping” for plaque psoriasis, with 1,164 patients meeting inclusion criteria. Moving cupping was found to be significantly more effective than “no-moving” cupping therapy, and moving cupping, combined with medications, performed better than medications alone.10 None of the trials evaluated in this study included randomized controlled trials that compared patients using any of the more modern psoriasis medications, specifically biologics. And, again, the studies evaluated were not of the highest quality.
The data that support cupping, as summarized above, are based mostly on case reports, and strong double-blind prospective studies are lacking. Additionally, most of the studies cited gauged the efficacy of cupping using qualitative endpoints, rather than standardized quantitative endpoints and scales. Moreover, spontaneous remission of various dermatoses can occur, or they can improve over time, including acute eczema, psoriasis, and, especially, urticaria.
Adverse effects of cupping
Often alternative therapies are seen as “benign” and without adverse effects. However, complications can result from cupping. Trauma can be induced from the cupping itself by damaging superficial blood vessels and causing bruising.1,11 Blistering can also occur secondary to the suction effect, and the epidermal and dermal layers of the skin can be separated.1,11 Further, burns and discoloration have also been noted secondary to heat, trauma, and post inflammatory pigmentary changes.1,11 Another risk of cupping is the Koebner phenomenon, which occurs with psoriasis, with new lesions appearing in traumatized skin.12 Other adverse outcomes that have been reported with cupping include reactivation of herpes simplex virus secondary to skin trauma, iron deficiency anemia (secondary to blood loss), panniculitis, infections, and residual marks mistaken for signs of child abuse.1,11
Cupping in aesthetic dermatology
Facial cupping, a distinct practice from body cupping used to treat general dermatology conditions described previously, is also increasing in popularity. This practice is usually conducted in association with a facial or facial acupuncture by an aesthetician or other licensed professional. It can also be performed using at-home kits. The marketing claims for facial cupping cite improved tightening and contouring of facial skin, increased facial microcirculation and collagen synthesis, and enhanced lymphatic flow to aid with facial puffiness or swelling. One supposed mechanism for these benefits is that cupping increases blood flow. Interestingly, there was a 2020 animal study in which photoacoustic imaging of a mouse ear revealed increased temporary blood flow in the cupping microenvironment.13 Currently, however, there is no evidence in the English scientific literature that supports facial cupping. The benefits attributed to facial cupping for aesthetic purposes have emerged only in personal anecdotes. The temporary increase in blood flow may induce inflammation and swelling that adds volume to the face and temporarily diminishes wrinkles. However, this temporary plumpness may be associated with adverse effects, such as local trauma, irritation, bruising, postinflammatory pigmentary alteration, or even herpes reactivation. In my opinion, the possible adverse effects of cupping outweigh any potential benefit, especially given the insufficient evidence supporting the utility of cupping for cosmetic enhancement.
Summary
There is increasing interest among patients to incorporate complementary and alternative medicine – including the ancient tradition of cupping – in managing medical dermatologic conditions. However, current evidence supporting cupping as an effective therapeutic strategy is not strong, with most studies to date appearing to be of poor quality or not sufficiently convincing to displace standard therapies. Our medical strategies for managing chronic dermatologic conditions, particularly inflammatory disorders, continue to improve from both a safety and a proven efficacy standpoint. Therefore, I would not forgo medical management in favor of cupping. While cupping can be used as an adjunct therapy, I would caution patients about possible adverse side effects. In the aesthetic world, cupping is also gaining popularity, but this trend is also not supported by current evidence or studies, at least in the Western literature.
Dr. Goldman is a dermatologist in private practice in Miami and specializes in cosmetic and general dermatology. She practices at Baumann Cosmetic & Research Institute and is also opening a general dermatology practice. Write to her at [email protected] or message her on Instragram @DrChloeGoldman. Dr. Goldman receives compensation to create social media content for Replenix, a skin care company. She has no other disclosures.
References
1. Soliman Y et al. Acta Dermatovenerol Alp Pannonica Adriat. 2018 Jun;27(2):103-7.
2. França K and Lotti T. Advances in Integrative Dermatology. John Wiley & Sons, 2019.
3. Lowe DT. Complement Ther Clin Pract. 2017 Nov;29:162-8.
4.Cao H et al. Altern Ther Health Med. 2010 Nov-Dec;16(6):48-54.
5. Li L and Ding J. J Tradit Chin Med. 2001 Mar;21(1):37-8.
6. Xiao XJ et al. J Integr Med. 2020 Jul;18(4):303-12.
7. Xu J et al. J Tradit Chin Med. 2013 Dec;33(6):752-6.
8. Yao J et al. Zhongguo Zhen Jiu. 2007; Jun;27(6):424-6.
9. Hon KL et al. Case Rep Pediatr. 2013;2013:605829.
10. Xing M et al. Medicine (Baltimore). 2020 Oct 9;99(41):e22539.
11. Kim TH et al. Eur J Integr Med. 2014 Aug 1;6(4):434-40.
12. Vender R and Vender R. J Cutan Med Surg. 2015 May-Jun;19(3):320-2.
13. Zhou Y et al. Biomed Opt Express. 2020 Apr 6;11(5):2394-401.
This article was updated 4/25/22.
Apremilast has neutral effect on vascular inflammation in psoriasis study
BOSTON – Treatment with , and glucose metabolism, in a study presented at the 2022 American Academy of Dermatology annual meeting.
In the phase 4, open-label, single arm trial, participants also lost subcutaneous and visceral fat after 16 weeks on the oral medication, a phosphodiesterase 4 (PDE4) inhibitor, and maintained that loss at 52 weeks.
People with psoriasis have an increased risk of obesity, type 2 diabetes, and cardiovascular events. Patients with more significant psoriasis “tend to die about 5 years younger than they should, based on their risk factors for mortality,” Joel Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania Perelman School of Medicine, Philadelphia, told this news organization.
He led the research and presented the findings at the AAD meeting March 26. “As a result, there has been a keen interest in understanding how psoriasis therapies impact cardiovascular risk, the idea being that by controlling inflammation, you may lower the risk of these patients developing cardiovascular disease over time,” he said.
Previous trials looking at the effect of psoriasis therapies on vascular inflammation “have been, for the most part, inconclusive,” Michael Garshick, MD, a cardiologist at NYU Langone Health, told this news organization. Dr. Garshick was not involved with the research. A 2021 systematic review of psoriasis clinicals trials reported that the tumor necrosis factor (TNF) blocker adalimumab (Humira) and phototherapy had the greatest effect on cardiometabolic markers, while ustekinumab (Stelara), an interleukin (IL)-12 and IL-23 antagonist, was the only treatment that improved vascular inflammation. These variable findings make this area “ripe for study,” noted Dr. Garshick.
To observe how apremilast, which is approved by the FDA for treating psoriasis and psoriatic arthritis, affected vascular inflammation, adiposity, and blood-based cardiometabolic markers, Dr. Gelfand organized an open-label study in adults with moderate-to-severe psoriasis. All participants were 18 years or older, had psoriasis for at least 6 months, and were candidates for systemic therapy. All patients underwent FDG PET/CT scans to assess aortic vascular inflammation and had blood work at baseline. Of the 70 patients originally enrolled in the study, 60 remained in the study at week 16, including 57 who underwent imaging for the second time. Thirty-nine participants remained in the study until week 52, and all except one had another scan.
The average age of participants was 47 years, and their mean BMI was 30. More than 80% of participants were White (83%) and 77% were male. The study population had lived with psoriasis for an average of 16 years and 8 patients also had psoriatic arthritis. At baseline, on average, participants had a Psoriasis Area and Severity Index (PASI) score of 18.62, a dermatology life quality index (DLQI) score of 11.60, and 22% of participants’ BSA (body surface area) were affected. The mean TBRmax, the marker for vascular inflammation, was 1.61.
Treatment responses were as expected for apremilast, with 35% of patients achieving PASI 75 and 65% of participants reporting DLQI scores of 5 or less by 16 weeks. At 52 weeks, 31% of the cohort had achieved PASI 75, and 67% reported DLQI score of 5 or higher. All psoriasis endpoints had improved since baseline (P = .001).
Throughout the study period, there was no significant change in TBRmax. However, in a sensitivity analysis, the 16 patients with a baseline TBRmax of 1.6 or higher had an absolute reduction of 0.21 in TBR by week 52. “That suggests that maybe a subset of people who have higher levels of aortic inflammation at baseline may experience some reduction that portend, potentially, some health benefits over time,” Dr. Gelfand said. “Ultimately, I wouldn’t hang my hat on the finding,” he said, noting that additional research comparing the treatment to placebo is necessary.
Both visceral and subcutaneous adipose tissue (VAT and SAT) decreased by week 16, and this reduction was maintained through week 52. In the first 16 weeks of the study, VAT decreased by 5.32% (P = .0009), and SAT decreased by 5.53% (P = .0005). From baseline to 52 weeks, VAT decreased by 5.52% (P = .0148), and SAT decreased by 5.50% (P = .0096). There were no significant differences between week 16 and week 52 in VAT or SAT.
Of the 68 blood biomarkers analyzed, there were significant decreases in the inflammatory markers ferritin (P = .015) and IL-beta (P = .006), the lipid metabolism biomarker HDL-cholesterol efflux (P = .008), and ketone bodies (P = .006). There were also increases in the inflammatory marker IL-8 (P = .003), the lipid metabolism marker ApoA (P = .05), and insulin (P = .05). Ferritin was the only biomarker that was reduced on both week 16 and week 52.
“If you want to be a purist, this was a negative trial,” said Dr. Garshick, because apremilast was not found to decrease vascular inflammation; however, he noted that the biomarker changes “were hopeful secondary endpoints.” It could be, he said, that another outcome measure may be better able to show changes in vascular inflammation compared with FDG. “It’s always hard to figure out what a good surrogate endpoint is in cardiovascular trials,” he noted, “so it may be that FDG/PET is too noisy or not reliable enough to see the outcome that we want to see.”
Dr. Gelfand reports consulting fees/grants from Amgen, AbbVie, BMS, Boehringer Ingelheim, Janssen Biologics, Novartis Corp, Pfizer, and UCB (DSMB). He serves as the Deputy Editor for the Journal of Investigative Dermatology and the Chief Medical Editor at Healio Psoriatic Disease and receives honoraria for both roles. Dr. Garshick has received consulting fees from AbbVie.
A version of this article first appeared on Medscape.com.
BOSTON – Treatment with , and glucose metabolism, in a study presented at the 2022 American Academy of Dermatology annual meeting.
In the phase 4, open-label, single arm trial, participants also lost subcutaneous and visceral fat after 16 weeks on the oral medication, a phosphodiesterase 4 (PDE4) inhibitor, and maintained that loss at 52 weeks.
People with psoriasis have an increased risk of obesity, type 2 diabetes, and cardiovascular events. Patients with more significant psoriasis “tend to die about 5 years younger than they should, based on their risk factors for mortality,” Joel Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania Perelman School of Medicine, Philadelphia, told this news organization.
He led the research and presented the findings at the AAD meeting March 26. “As a result, there has been a keen interest in understanding how psoriasis therapies impact cardiovascular risk, the idea being that by controlling inflammation, you may lower the risk of these patients developing cardiovascular disease over time,” he said.
Previous trials looking at the effect of psoriasis therapies on vascular inflammation “have been, for the most part, inconclusive,” Michael Garshick, MD, a cardiologist at NYU Langone Health, told this news organization. Dr. Garshick was not involved with the research. A 2021 systematic review of psoriasis clinicals trials reported that the tumor necrosis factor (TNF) blocker adalimumab (Humira) and phototherapy had the greatest effect on cardiometabolic markers, while ustekinumab (Stelara), an interleukin (IL)-12 and IL-23 antagonist, was the only treatment that improved vascular inflammation. These variable findings make this area “ripe for study,” noted Dr. Garshick.
To observe how apremilast, which is approved by the FDA for treating psoriasis and psoriatic arthritis, affected vascular inflammation, adiposity, and blood-based cardiometabolic markers, Dr. Gelfand organized an open-label study in adults with moderate-to-severe psoriasis. All participants were 18 years or older, had psoriasis for at least 6 months, and were candidates for systemic therapy. All patients underwent FDG PET/CT scans to assess aortic vascular inflammation and had blood work at baseline. Of the 70 patients originally enrolled in the study, 60 remained in the study at week 16, including 57 who underwent imaging for the second time. Thirty-nine participants remained in the study until week 52, and all except one had another scan.
The average age of participants was 47 years, and their mean BMI was 30. More than 80% of participants were White (83%) and 77% were male. The study population had lived with psoriasis for an average of 16 years and 8 patients also had psoriatic arthritis. At baseline, on average, participants had a Psoriasis Area and Severity Index (PASI) score of 18.62, a dermatology life quality index (DLQI) score of 11.60, and 22% of participants’ BSA (body surface area) were affected. The mean TBRmax, the marker for vascular inflammation, was 1.61.
Treatment responses were as expected for apremilast, with 35% of patients achieving PASI 75 and 65% of participants reporting DLQI scores of 5 or less by 16 weeks. At 52 weeks, 31% of the cohort had achieved PASI 75, and 67% reported DLQI score of 5 or higher. All psoriasis endpoints had improved since baseline (P = .001).
Throughout the study period, there was no significant change in TBRmax. However, in a sensitivity analysis, the 16 patients with a baseline TBRmax of 1.6 or higher had an absolute reduction of 0.21 in TBR by week 52. “That suggests that maybe a subset of people who have higher levels of aortic inflammation at baseline may experience some reduction that portend, potentially, some health benefits over time,” Dr. Gelfand said. “Ultimately, I wouldn’t hang my hat on the finding,” he said, noting that additional research comparing the treatment to placebo is necessary.
Both visceral and subcutaneous adipose tissue (VAT and SAT) decreased by week 16, and this reduction was maintained through week 52. In the first 16 weeks of the study, VAT decreased by 5.32% (P = .0009), and SAT decreased by 5.53% (P = .0005). From baseline to 52 weeks, VAT decreased by 5.52% (P = .0148), and SAT decreased by 5.50% (P = .0096). There were no significant differences between week 16 and week 52 in VAT or SAT.
Of the 68 blood biomarkers analyzed, there were significant decreases in the inflammatory markers ferritin (P = .015) and IL-beta (P = .006), the lipid metabolism biomarker HDL-cholesterol efflux (P = .008), and ketone bodies (P = .006). There were also increases in the inflammatory marker IL-8 (P = .003), the lipid metabolism marker ApoA (P = .05), and insulin (P = .05). Ferritin was the only biomarker that was reduced on both week 16 and week 52.
“If you want to be a purist, this was a negative trial,” said Dr. Garshick, because apremilast was not found to decrease vascular inflammation; however, he noted that the biomarker changes “were hopeful secondary endpoints.” It could be, he said, that another outcome measure may be better able to show changes in vascular inflammation compared with FDG. “It’s always hard to figure out what a good surrogate endpoint is in cardiovascular trials,” he noted, “so it may be that FDG/PET is too noisy or not reliable enough to see the outcome that we want to see.”
Dr. Gelfand reports consulting fees/grants from Amgen, AbbVie, BMS, Boehringer Ingelheim, Janssen Biologics, Novartis Corp, Pfizer, and UCB (DSMB). He serves as the Deputy Editor for the Journal of Investigative Dermatology and the Chief Medical Editor at Healio Psoriatic Disease and receives honoraria for both roles. Dr. Garshick has received consulting fees from AbbVie.
A version of this article first appeared on Medscape.com.
BOSTON – Treatment with , and glucose metabolism, in a study presented at the 2022 American Academy of Dermatology annual meeting.
In the phase 4, open-label, single arm trial, participants also lost subcutaneous and visceral fat after 16 weeks on the oral medication, a phosphodiesterase 4 (PDE4) inhibitor, and maintained that loss at 52 weeks.
People with psoriasis have an increased risk of obesity, type 2 diabetes, and cardiovascular events. Patients with more significant psoriasis “tend to die about 5 years younger than they should, based on their risk factors for mortality,” Joel Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania Perelman School of Medicine, Philadelphia, told this news organization.
He led the research and presented the findings at the AAD meeting March 26. “As a result, there has been a keen interest in understanding how psoriasis therapies impact cardiovascular risk, the idea being that by controlling inflammation, you may lower the risk of these patients developing cardiovascular disease over time,” he said.
Previous trials looking at the effect of psoriasis therapies on vascular inflammation “have been, for the most part, inconclusive,” Michael Garshick, MD, a cardiologist at NYU Langone Health, told this news organization. Dr. Garshick was not involved with the research. A 2021 systematic review of psoriasis clinicals trials reported that the tumor necrosis factor (TNF) blocker adalimumab (Humira) and phototherapy had the greatest effect on cardiometabolic markers, while ustekinumab (Stelara), an interleukin (IL)-12 and IL-23 antagonist, was the only treatment that improved vascular inflammation. These variable findings make this area “ripe for study,” noted Dr. Garshick.
To observe how apremilast, which is approved by the FDA for treating psoriasis and psoriatic arthritis, affected vascular inflammation, adiposity, and blood-based cardiometabolic markers, Dr. Gelfand organized an open-label study in adults with moderate-to-severe psoriasis. All participants were 18 years or older, had psoriasis for at least 6 months, and were candidates for systemic therapy. All patients underwent FDG PET/CT scans to assess aortic vascular inflammation and had blood work at baseline. Of the 70 patients originally enrolled in the study, 60 remained in the study at week 16, including 57 who underwent imaging for the second time. Thirty-nine participants remained in the study until week 52, and all except one had another scan.
The average age of participants was 47 years, and their mean BMI was 30. More than 80% of participants were White (83%) and 77% were male. The study population had lived with psoriasis for an average of 16 years and 8 patients also had psoriatic arthritis. At baseline, on average, participants had a Psoriasis Area and Severity Index (PASI) score of 18.62, a dermatology life quality index (DLQI) score of 11.60, and 22% of participants’ BSA (body surface area) were affected. The mean TBRmax, the marker for vascular inflammation, was 1.61.
Treatment responses were as expected for apremilast, with 35% of patients achieving PASI 75 and 65% of participants reporting DLQI scores of 5 or less by 16 weeks. At 52 weeks, 31% of the cohort had achieved PASI 75, and 67% reported DLQI score of 5 or higher. All psoriasis endpoints had improved since baseline (P = .001).
Throughout the study period, there was no significant change in TBRmax. However, in a sensitivity analysis, the 16 patients with a baseline TBRmax of 1.6 or higher had an absolute reduction of 0.21 in TBR by week 52. “That suggests that maybe a subset of people who have higher levels of aortic inflammation at baseline may experience some reduction that portend, potentially, some health benefits over time,” Dr. Gelfand said. “Ultimately, I wouldn’t hang my hat on the finding,” he said, noting that additional research comparing the treatment to placebo is necessary.
Both visceral and subcutaneous adipose tissue (VAT and SAT) decreased by week 16, and this reduction was maintained through week 52. In the first 16 weeks of the study, VAT decreased by 5.32% (P = .0009), and SAT decreased by 5.53% (P = .0005). From baseline to 52 weeks, VAT decreased by 5.52% (P = .0148), and SAT decreased by 5.50% (P = .0096). There were no significant differences between week 16 and week 52 in VAT or SAT.
Of the 68 blood biomarkers analyzed, there were significant decreases in the inflammatory markers ferritin (P = .015) and IL-beta (P = .006), the lipid metabolism biomarker HDL-cholesterol efflux (P = .008), and ketone bodies (P = .006). There were also increases in the inflammatory marker IL-8 (P = .003), the lipid metabolism marker ApoA (P = .05), and insulin (P = .05). Ferritin was the only biomarker that was reduced on both week 16 and week 52.
“If you want to be a purist, this was a negative trial,” said Dr. Garshick, because apremilast was not found to decrease vascular inflammation; however, he noted that the biomarker changes “were hopeful secondary endpoints.” It could be, he said, that another outcome measure may be better able to show changes in vascular inflammation compared with FDG. “It’s always hard to figure out what a good surrogate endpoint is in cardiovascular trials,” he noted, “so it may be that FDG/PET is too noisy or not reliable enough to see the outcome that we want to see.”
Dr. Gelfand reports consulting fees/grants from Amgen, AbbVie, BMS, Boehringer Ingelheim, Janssen Biologics, Novartis Corp, Pfizer, and UCB (DSMB). He serves as the Deputy Editor for the Journal of Investigative Dermatology and the Chief Medical Editor at Healio Psoriatic Disease and receives honoraria for both roles. Dr. Garshick has received consulting fees from AbbVie.
A version of this article first appeared on Medscape.com.
AT AAD 2022