User login
Clear Coverage Preference for Humira Over Biosimilars Seen in Most Medicare Part D Plans
Despite the influx of adalimumab biosimilars entering the market in 2023, Humira remains on top.
As of January 2024, both high and low concentrations of Humira, the originator adalimumab product, are nearly universally covered by Medicare Part D plans, while only half of these plans covered adalimumab biosimilars, according to a new research letter published online on June 6, 2024, in JAMA.
Of the plans that covered both, only 1.5% had lower-tier placement for biosimilars.
“This study of formulary coverage helps explain limited uptake of adalimumab biosimilars,” wrote the authors, led by Matthew J. Klebanoff, MD, of the University of Pennsylvania, Philadelphia. “Subpar biosimilar adoption will not only undermine their potential to reduce spending but also may deter investments in biosimilar development.”
The analysis included the formulary and enrollment files for 5609 Medicare Part D plans, representing 44.4 million beneficiaries. Drug list prices and whole acquisition costs (WAC) were pulled from the Red Book database, which provides prices for prescription and over-the-counter drugs as well as medical devices and supplies.
Nearly all (98.9%) of Part D plans covered the high-concentration (100 mg/mL) version of adalimumab with a WAC of $6923. This higher concentration is the most popular formulation of the drug, making up an estimated 85% of prescriptions. By comparison, 26.8% of plans covered the high-concentration version of adalimumab-adaz (Hyrimoz), with a WAC 5% less than the reference product.
The unbranded version of adalimumab-adaz, sold at an 81% discount from the reference product, was covered by 13% of plans. Only 4.6% of plans covered high-concentration adalimumab-bwwd (Hadlima), manufactured by Samsung Bioepis.
In January 2024, no high-concentration adalimumab biosimilar had been granted interchangeability status by the US Food and Drug Administration (FDA). Adalimumab-ryvk (Simlandi) was the first biosimilar to receive this designation and was launched in late May 2024.
Coverage for the lower concentration of adalimumab was nearly universal (98.7% of plans). About half of the plans (50.7%) covered adalimumab-adbm (Cyltezo) at a 5% discount. Adalimumab-adbm (Boehringer Ingelheim) was the first interchangeable Humira biosimilar approved by the FDA, but it is only interchangeable with the less popular, lower concentration formulation of adalimumab.
All other biosimilars were covered by less than 5% of Medicare Part D plans, even with some having a WAC 86% below Humira.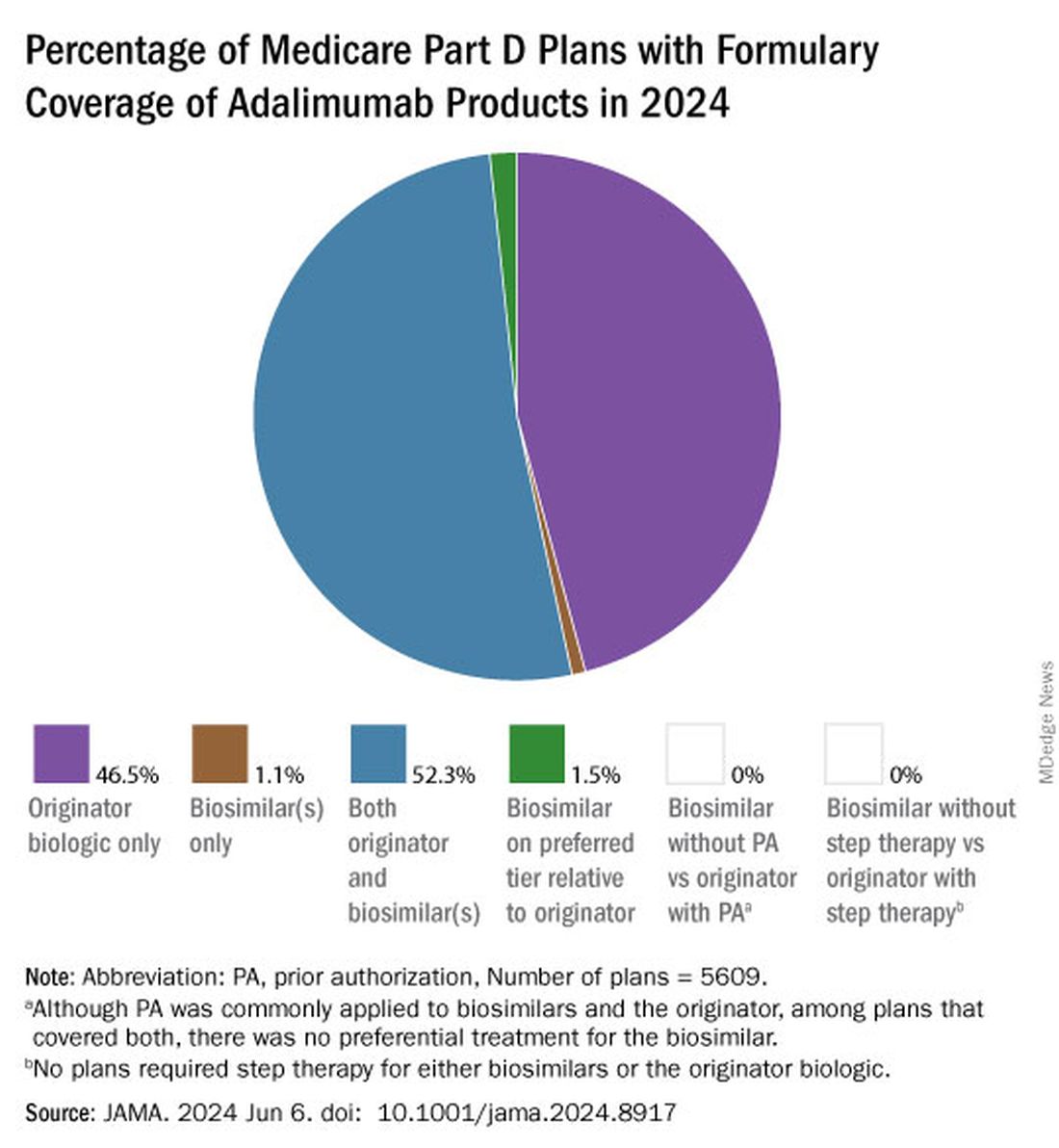
Few plans (1.5%) had biosimilars on preferred tiers compared with the reference product, and no plans used prior authorization to incentivize use of biosimilars. Most plans preferred the higher-priced version of adalimumab biosimilars, which appeals to pharmacy benefit managers who can therefore receive higher rebates, the authors noted.
“Ultimately, biosimilars’ true effect on spending will depend not on their list price but rather on their net price (after rebates) and their influence on originator biologics’ net price,” they wrote. They pointed to the 38% drop in Humira’s annual net price at the end of 2023 compared with the prior year.
“Despite this price decrease, biosimilars offer far greater potential savings: Several adalimumab biosimilars have list prices that are less than half of Humira’s net price,” the authors continued, and encouraged policy makers to mandate coverage for these lower-priced options.
Dr. Klebanoff was supported by a grant from the Health Resources and Services Administration. Two coauthors were supported by a grant from the National Institute on Aging. One author reported receiving consulting fees from AbbVie, which manufactures Humira.
A version of this article appeared on Medscape.com .
Despite the influx of adalimumab biosimilars entering the market in 2023, Humira remains on top.
As of January 2024, both high and low concentrations of Humira, the originator adalimumab product, are nearly universally covered by Medicare Part D plans, while only half of these plans covered adalimumab biosimilars, according to a new research letter published online on June 6, 2024, in JAMA.
Of the plans that covered both, only 1.5% had lower-tier placement for biosimilars.
“This study of formulary coverage helps explain limited uptake of adalimumab biosimilars,” wrote the authors, led by Matthew J. Klebanoff, MD, of the University of Pennsylvania, Philadelphia. “Subpar biosimilar adoption will not only undermine their potential to reduce spending but also may deter investments in biosimilar development.”
The analysis included the formulary and enrollment files for 5609 Medicare Part D plans, representing 44.4 million beneficiaries. Drug list prices and whole acquisition costs (WAC) were pulled from the Red Book database, which provides prices for prescription and over-the-counter drugs as well as medical devices and supplies.
Nearly all (98.9%) of Part D plans covered the high-concentration (100 mg/mL) version of adalimumab with a WAC of $6923. This higher concentration is the most popular formulation of the drug, making up an estimated 85% of prescriptions. By comparison, 26.8% of plans covered the high-concentration version of adalimumab-adaz (Hyrimoz), with a WAC 5% less than the reference product.
The unbranded version of adalimumab-adaz, sold at an 81% discount from the reference product, was covered by 13% of plans. Only 4.6% of plans covered high-concentration adalimumab-bwwd (Hadlima), manufactured by Samsung Bioepis.
In January 2024, no high-concentration adalimumab biosimilar had been granted interchangeability status by the US Food and Drug Administration (FDA). Adalimumab-ryvk (Simlandi) was the first biosimilar to receive this designation and was launched in late May 2024.
Coverage for the lower concentration of adalimumab was nearly universal (98.7% of plans). About half of the plans (50.7%) covered adalimumab-adbm (Cyltezo) at a 5% discount. Adalimumab-adbm (Boehringer Ingelheim) was the first interchangeable Humira biosimilar approved by the FDA, but it is only interchangeable with the less popular, lower concentration formulation of adalimumab.
All other biosimilars were covered by less than 5% of Medicare Part D plans, even with some having a WAC 86% below Humira.
Few plans (1.5%) had biosimilars on preferred tiers compared with the reference product, and no plans used prior authorization to incentivize use of biosimilars. Most plans preferred the higher-priced version of adalimumab biosimilars, which appeals to pharmacy benefit managers who can therefore receive higher rebates, the authors noted.
“Ultimately, biosimilars’ true effect on spending will depend not on their list price but rather on their net price (after rebates) and their influence on originator biologics’ net price,” they wrote. They pointed to the 38% drop in Humira’s annual net price at the end of 2023 compared with the prior year.
“Despite this price decrease, biosimilars offer far greater potential savings: Several adalimumab biosimilars have list prices that are less than half of Humira’s net price,” the authors continued, and encouraged policy makers to mandate coverage for these lower-priced options.
Dr. Klebanoff was supported by a grant from the Health Resources and Services Administration. Two coauthors were supported by a grant from the National Institute on Aging. One author reported receiving consulting fees from AbbVie, which manufactures Humira.
A version of this article appeared on Medscape.com .
Despite the influx of adalimumab biosimilars entering the market in 2023, Humira remains on top.
As of January 2024, both high and low concentrations of Humira, the originator adalimumab product, are nearly universally covered by Medicare Part D plans, while only half of these plans covered adalimumab biosimilars, according to a new research letter published online on June 6, 2024, in JAMA.
Of the plans that covered both, only 1.5% had lower-tier placement for biosimilars.
“This study of formulary coverage helps explain limited uptake of adalimumab biosimilars,” wrote the authors, led by Matthew J. Klebanoff, MD, of the University of Pennsylvania, Philadelphia. “Subpar biosimilar adoption will not only undermine their potential to reduce spending but also may deter investments in biosimilar development.”
The analysis included the formulary and enrollment files for 5609 Medicare Part D plans, representing 44.4 million beneficiaries. Drug list prices and whole acquisition costs (WAC) were pulled from the Red Book database, which provides prices for prescription and over-the-counter drugs as well as medical devices and supplies.
Nearly all (98.9%) of Part D plans covered the high-concentration (100 mg/mL) version of adalimumab with a WAC of $6923. This higher concentration is the most popular formulation of the drug, making up an estimated 85% of prescriptions. By comparison, 26.8% of plans covered the high-concentration version of adalimumab-adaz (Hyrimoz), with a WAC 5% less than the reference product.
The unbranded version of adalimumab-adaz, sold at an 81% discount from the reference product, was covered by 13% of plans. Only 4.6% of plans covered high-concentration adalimumab-bwwd (Hadlima), manufactured by Samsung Bioepis.
In January 2024, no high-concentration adalimumab biosimilar had been granted interchangeability status by the US Food and Drug Administration (FDA). Adalimumab-ryvk (Simlandi) was the first biosimilar to receive this designation and was launched in late May 2024.
Coverage for the lower concentration of adalimumab was nearly universal (98.7% of plans). About half of the plans (50.7%) covered adalimumab-adbm (Cyltezo) at a 5% discount. Adalimumab-adbm (Boehringer Ingelheim) was the first interchangeable Humira biosimilar approved by the FDA, but it is only interchangeable with the less popular, lower concentration formulation of adalimumab.
All other biosimilars were covered by less than 5% of Medicare Part D plans, even with some having a WAC 86% below Humira.
Few plans (1.5%) had biosimilars on preferred tiers compared with the reference product, and no plans used prior authorization to incentivize use of biosimilars. Most plans preferred the higher-priced version of adalimumab biosimilars, which appeals to pharmacy benefit managers who can therefore receive higher rebates, the authors noted.
“Ultimately, biosimilars’ true effect on spending will depend not on their list price but rather on their net price (after rebates) and their influence on originator biologics’ net price,” they wrote. They pointed to the 38% drop in Humira’s annual net price at the end of 2023 compared with the prior year.
“Despite this price decrease, biosimilars offer far greater potential savings: Several adalimumab biosimilars have list prices that are less than half of Humira’s net price,” the authors continued, and encouraged policy makers to mandate coverage for these lower-priced options.
Dr. Klebanoff was supported by a grant from the Health Resources and Services Administration. Two coauthors were supported by a grant from the National Institute on Aging. One author reported receiving consulting fees from AbbVie, which manufactures Humira.
A version of this article appeared on Medscape.com .
FROM JAMA
Over-the-Counter Arthritis Supplements Pose Adrenal Danger
BOSTON —
Patients who have been taking these supplements for prolonged periods must slowly taper off them with corticosteroid replacement, because abruptly stopping the supplement can precipitate AI, Kevin S. Wei, MD, said in a presentation of 12 cases — the largest such series to date of the phenomenon — at the annual meeting of the Endocrine Society.
The specific supplements used were Artri King in eight of the patients, Ardosons in two, and Ajo Rey in one. In April 2022, the US Food and Drug Administration issued a warning that Artri King contains diclofenac and dexamethasone not listed on the product label. In July 2023, the agency issued an expanded warning about that product and others including Ajo Rey.
The supplements are not believed to be sold in the United States, but they are available in Mexico and can be ordered online, said Dr. Wei, a second-year resident at the Keck School of Medicine at the University of California, Los Angeles.
“We found that quite a lot of patients after they’ve been on the Artri King or some other over the counter arthritis supplement, started developing these cushingoid features seen in the physical exam, such as rounded facial features or stretch marks of their abdomen,” he said.
And “when patients are abruptly taken off those supplements … sometimes this can cause them to go into signs or symptoms of adrenal insufficiency. That can occasionally be life-threatening if it’s not addressed in an inpatient setting,” Dr. Wei said.
In an interview, session moderator Sharon L. Wardlaw, MD, professor of medicine at Columbia University Irving Medical Center, New York, explained that when a person takes these drugs containing hidden glucocorticoids, “they won’t be picked up in a cortisol assay, but they’ll suppress the [adrenocorticotropic hormone] and the person’s own cortisol production. They look like they have Cushing, but when you measure their hormone levels, they’re undetectable. And then people wonder what’s going on. Well, their [hypothalamic-pituitary-adrenal] axis is suppressed.”
But if the product is suddenly stopped without cortisol replacement “If they get an infection they can die because they can’t mount a cortisol response.”
The takeaway message, she said, is “always ask patients to show you their supplements and look at them. In many cases, that’s why they work so well for pain relief because they have ingredients that people shouldn’t be taking.”
Twelve Patients Seen During 2022-2023
The 12 patients were seen during 2022-2023 at an endocrinology consult service in an urban safety net hospital. Their median age was 52 years, and one third were women. All had started using the supplements for joint pain, with a median of about 6 months of use prior to cessation.
Presenting symptoms included nausea/vomiting in 42%, fatigue in 42%, abdominal pain in 33%, and dizziness in 17%. Physical exam findings included moon facies in 66%, central adiposity in 66%, abdominal striae in 50%, dorsocervical fat pad in 33%, and bruising in 33%. Three required intensive care admission.
Cortisol testing was performed in 11 of the patients and was normal (≥ 16 mcg/dL) in just one. AI (≤ 3 mcg/dL) was found in three, while the rest had indeterminate results. Of those seven patients, subsequent cosyntropin-stimulation testing suggested AI (cortisol < 16 mcg/dL at 60 minutes post stimulation) in four patients, while the other two showed reduced but normal responses (cortisol 18.2-18.4 mcg/dL).
Ten of the 12 patients were prescribed glucocorticoid tapering replacements to avoid precipitating adrenal crisis, most commonly twice-daily hydrocortisone. Of those ten, eight continued to take the replacement steroids 1-2 years later, Dr. Wei said.
Dr. Wei and Dr. Wardlaw had no disclosures.
A version of this article appeared on Medscape.com.
BOSTON —
Patients who have been taking these supplements for prolonged periods must slowly taper off them with corticosteroid replacement, because abruptly stopping the supplement can precipitate AI, Kevin S. Wei, MD, said in a presentation of 12 cases — the largest such series to date of the phenomenon — at the annual meeting of the Endocrine Society.
The specific supplements used were Artri King in eight of the patients, Ardosons in two, and Ajo Rey in one. In April 2022, the US Food and Drug Administration issued a warning that Artri King contains diclofenac and dexamethasone not listed on the product label. In July 2023, the agency issued an expanded warning about that product and others including Ajo Rey.
The supplements are not believed to be sold in the United States, but they are available in Mexico and can be ordered online, said Dr. Wei, a second-year resident at the Keck School of Medicine at the University of California, Los Angeles.
“We found that quite a lot of patients after they’ve been on the Artri King or some other over the counter arthritis supplement, started developing these cushingoid features seen in the physical exam, such as rounded facial features or stretch marks of their abdomen,” he said.
And “when patients are abruptly taken off those supplements … sometimes this can cause them to go into signs or symptoms of adrenal insufficiency. That can occasionally be life-threatening if it’s not addressed in an inpatient setting,” Dr. Wei said.
In an interview, session moderator Sharon L. Wardlaw, MD, professor of medicine at Columbia University Irving Medical Center, New York, explained that when a person takes these drugs containing hidden glucocorticoids, “they won’t be picked up in a cortisol assay, but they’ll suppress the [adrenocorticotropic hormone] and the person’s own cortisol production. They look like they have Cushing, but when you measure their hormone levels, they’re undetectable. And then people wonder what’s going on. Well, their [hypothalamic-pituitary-adrenal] axis is suppressed.”
But if the product is suddenly stopped without cortisol replacement “If they get an infection they can die because they can’t mount a cortisol response.”
The takeaway message, she said, is “always ask patients to show you their supplements and look at them. In many cases, that’s why they work so well for pain relief because they have ingredients that people shouldn’t be taking.”
Twelve Patients Seen During 2022-2023
The 12 patients were seen during 2022-2023 at an endocrinology consult service in an urban safety net hospital. Their median age was 52 years, and one third were women. All had started using the supplements for joint pain, with a median of about 6 months of use prior to cessation.
Presenting symptoms included nausea/vomiting in 42%, fatigue in 42%, abdominal pain in 33%, and dizziness in 17%. Physical exam findings included moon facies in 66%, central adiposity in 66%, abdominal striae in 50%, dorsocervical fat pad in 33%, and bruising in 33%. Three required intensive care admission.
Cortisol testing was performed in 11 of the patients and was normal (≥ 16 mcg/dL) in just one. AI (≤ 3 mcg/dL) was found in three, while the rest had indeterminate results. Of those seven patients, subsequent cosyntropin-stimulation testing suggested AI (cortisol < 16 mcg/dL at 60 minutes post stimulation) in four patients, while the other two showed reduced but normal responses (cortisol 18.2-18.4 mcg/dL).
Ten of the 12 patients were prescribed glucocorticoid tapering replacements to avoid precipitating adrenal crisis, most commonly twice-daily hydrocortisone. Of those ten, eight continued to take the replacement steroids 1-2 years later, Dr. Wei said.
Dr. Wei and Dr. Wardlaw had no disclosures.
A version of this article appeared on Medscape.com.
BOSTON —
Patients who have been taking these supplements for prolonged periods must slowly taper off them with corticosteroid replacement, because abruptly stopping the supplement can precipitate AI, Kevin S. Wei, MD, said in a presentation of 12 cases — the largest such series to date of the phenomenon — at the annual meeting of the Endocrine Society.
The specific supplements used were Artri King in eight of the patients, Ardosons in two, and Ajo Rey in one. In April 2022, the US Food and Drug Administration issued a warning that Artri King contains diclofenac and dexamethasone not listed on the product label. In July 2023, the agency issued an expanded warning about that product and others including Ajo Rey.
The supplements are not believed to be sold in the United States, but they are available in Mexico and can be ordered online, said Dr. Wei, a second-year resident at the Keck School of Medicine at the University of California, Los Angeles.
“We found that quite a lot of patients after they’ve been on the Artri King or some other over the counter arthritis supplement, started developing these cushingoid features seen in the physical exam, such as rounded facial features or stretch marks of their abdomen,” he said.
And “when patients are abruptly taken off those supplements … sometimes this can cause them to go into signs or symptoms of adrenal insufficiency. That can occasionally be life-threatening if it’s not addressed in an inpatient setting,” Dr. Wei said.
In an interview, session moderator Sharon L. Wardlaw, MD, professor of medicine at Columbia University Irving Medical Center, New York, explained that when a person takes these drugs containing hidden glucocorticoids, “they won’t be picked up in a cortisol assay, but they’ll suppress the [adrenocorticotropic hormone] and the person’s own cortisol production. They look like they have Cushing, but when you measure their hormone levels, they’re undetectable. And then people wonder what’s going on. Well, their [hypothalamic-pituitary-adrenal] axis is suppressed.”
But if the product is suddenly stopped without cortisol replacement “If they get an infection they can die because they can’t mount a cortisol response.”
The takeaway message, she said, is “always ask patients to show you their supplements and look at them. In many cases, that’s why they work so well for pain relief because they have ingredients that people shouldn’t be taking.”
Twelve Patients Seen During 2022-2023
The 12 patients were seen during 2022-2023 at an endocrinology consult service in an urban safety net hospital. Their median age was 52 years, and one third were women. All had started using the supplements for joint pain, with a median of about 6 months of use prior to cessation.
Presenting symptoms included nausea/vomiting in 42%, fatigue in 42%, abdominal pain in 33%, and dizziness in 17%. Physical exam findings included moon facies in 66%, central adiposity in 66%, abdominal striae in 50%, dorsocervical fat pad in 33%, and bruising in 33%. Three required intensive care admission.
Cortisol testing was performed in 11 of the patients and was normal (≥ 16 mcg/dL) in just one. AI (≤ 3 mcg/dL) was found in three, while the rest had indeterminate results. Of those seven patients, subsequent cosyntropin-stimulation testing suggested AI (cortisol < 16 mcg/dL at 60 minutes post stimulation) in four patients, while the other two showed reduced but normal responses (cortisol 18.2-18.4 mcg/dL).
Ten of the 12 patients were prescribed glucocorticoid tapering replacements to avoid precipitating adrenal crisis, most commonly twice-daily hydrocortisone. Of those ten, eight continued to take the replacement steroids 1-2 years later, Dr. Wei said.
Dr. Wei and Dr. Wardlaw had no disclosures.
A version of this article appeared on Medscape.com.
No Increased Risk for Fractures Seen With Frequent Steroid Injections for Musculoskeletal Conditions
TOPLINE:
The cumulative effect of frequent corticosteroid injections (CSIs), a common treatment for musculoskeletal pain, does not appear to increase the risk for fractures.
METHODOLOGY:
- Researchers utilized an institutional electronic health record database to identify adults in Olmsted County, Minnesota, receiving corticosteroid injections from May 1, 2018, to July 1, 2022.
- Corticosteroid equivalents were calculated for medications injected, including methylprednisolone, triamcinolone, betamethasone, and dexamethasone.
- Patients were excluded if they had a prescription for oral prednisone equivalents greater than 2.5 mg/day for more than 30 days.
- Fracture events were identified using ICD-9 and ICD-10 codes and were included only if they occurred after the first corticosteroid injection.
TAKEAWAY:
- A total of 7197 patients were analyzed, with a mean age of 64.4 years, and of these patients, 346 (4.8%) had a new fracture in a mean time of 329 days from the first corticosteroid injection, including 149 (43.1%) in classic osteoporotic locations.
- The study reported no increased fracture risk associated with corticosteroid injections and no significant difference in fracture rates across cumulative corticosteroid injection dose quartiles, regardless of osteoporosis status.
- Factors such as previous fractures, age, and Charlson Comorbidity Index were associated with a higher risk for fractures, not corticosteroid injections.
IN PRACTICE:
“Clinicians should be reassured that frequent CSI is not associated with higher fracture risk and should not withhold these important pain treatments owing to concern for fracture,” wrote the authors of the study.
SOURCE:
The study was led by Terin T. Sytsma, MD, Division of Community Internal Medicine, Geriatrics, and Palliative Care, Mayo Clinic, Rochester, Minnesota. It was published online in JAMA Network Open.
LIMITATIONS:
The study’s retrospective cohort design and its focus on a predominantly White population in a single community may limit the generalizability of the findings. Confounding variables such as smoking status, alcohol intake, and physical activity were acknowledged as potential contributors to fracture risk. Only clinically apparent fractures were considered, excluding silent vertebral fractures, and differences in corticosteroid formulation were not delineated.
DISCLOSURES:
The study was supported by a Mayo Clinic Catalyst Award to Dr. Sytsma. The authors had no conflicts of interest to report.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com.
TOPLINE:
The cumulative effect of frequent corticosteroid injections (CSIs), a common treatment for musculoskeletal pain, does not appear to increase the risk for fractures.
METHODOLOGY:
- Researchers utilized an institutional electronic health record database to identify adults in Olmsted County, Minnesota, receiving corticosteroid injections from May 1, 2018, to July 1, 2022.
- Corticosteroid equivalents were calculated for medications injected, including methylprednisolone, triamcinolone, betamethasone, and dexamethasone.
- Patients were excluded if they had a prescription for oral prednisone equivalents greater than 2.5 mg/day for more than 30 days.
- Fracture events were identified using ICD-9 and ICD-10 codes and were included only if they occurred after the first corticosteroid injection.
TAKEAWAY:
- A total of 7197 patients were analyzed, with a mean age of 64.4 years, and of these patients, 346 (4.8%) had a new fracture in a mean time of 329 days from the first corticosteroid injection, including 149 (43.1%) in classic osteoporotic locations.
- The study reported no increased fracture risk associated with corticosteroid injections and no significant difference in fracture rates across cumulative corticosteroid injection dose quartiles, regardless of osteoporosis status.
- Factors such as previous fractures, age, and Charlson Comorbidity Index were associated with a higher risk for fractures, not corticosteroid injections.
IN PRACTICE:
“Clinicians should be reassured that frequent CSI is not associated with higher fracture risk and should not withhold these important pain treatments owing to concern for fracture,” wrote the authors of the study.
SOURCE:
The study was led by Terin T. Sytsma, MD, Division of Community Internal Medicine, Geriatrics, and Palliative Care, Mayo Clinic, Rochester, Minnesota. It was published online in JAMA Network Open.
LIMITATIONS:
The study’s retrospective cohort design and its focus on a predominantly White population in a single community may limit the generalizability of the findings. Confounding variables such as smoking status, alcohol intake, and physical activity were acknowledged as potential contributors to fracture risk. Only clinically apparent fractures were considered, excluding silent vertebral fractures, and differences in corticosteroid formulation were not delineated.
DISCLOSURES:
The study was supported by a Mayo Clinic Catalyst Award to Dr. Sytsma. The authors had no conflicts of interest to report.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com.
TOPLINE:
The cumulative effect of frequent corticosteroid injections (CSIs), a common treatment for musculoskeletal pain, does not appear to increase the risk for fractures.
METHODOLOGY:
- Researchers utilized an institutional electronic health record database to identify adults in Olmsted County, Minnesota, receiving corticosteroid injections from May 1, 2018, to July 1, 2022.
- Corticosteroid equivalents were calculated for medications injected, including methylprednisolone, triamcinolone, betamethasone, and dexamethasone.
- Patients were excluded if they had a prescription for oral prednisone equivalents greater than 2.5 mg/day for more than 30 days.
- Fracture events were identified using ICD-9 and ICD-10 codes and were included only if they occurred after the first corticosteroid injection.
TAKEAWAY:
- A total of 7197 patients were analyzed, with a mean age of 64.4 years, and of these patients, 346 (4.8%) had a new fracture in a mean time of 329 days from the first corticosteroid injection, including 149 (43.1%) in classic osteoporotic locations.
- The study reported no increased fracture risk associated with corticosteroid injections and no significant difference in fracture rates across cumulative corticosteroid injection dose quartiles, regardless of osteoporosis status.
- Factors such as previous fractures, age, and Charlson Comorbidity Index were associated with a higher risk for fractures, not corticosteroid injections.
IN PRACTICE:
“Clinicians should be reassured that frequent CSI is not associated with higher fracture risk and should not withhold these important pain treatments owing to concern for fracture,” wrote the authors of the study.
SOURCE:
The study was led by Terin T. Sytsma, MD, Division of Community Internal Medicine, Geriatrics, and Palliative Care, Mayo Clinic, Rochester, Minnesota. It was published online in JAMA Network Open.
LIMITATIONS:
The study’s retrospective cohort design and its focus on a predominantly White population in a single community may limit the generalizability of the findings. Confounding variables such as smoking status, alcohol intake, and physical activity were acknowledged as potential contributors to fracture risk. Only clinically apparent fractures were considered, excluding silent vertebral fractures, and differences in corticosteroid formulation were not delineated.
DISCLOSURES:
The study was supported by a Mayo Clinic Catalyst Award to Dr. Sytsma. The authors had no conflicts of interest to report.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com.
Cortisol Test Confirms HPA Axis Recovery from Steroid Use
TOPLINE:
An early serum cortisol concentration of > 237 nmol/L (> 8.6 μg/dL) has been validated as a safe and useful screening test with 100% specificity for predicting recovery of the hypothalamic-pituitary-adrenal (HPA) axis in patients on tapering regimes from long‐term chronic glucocorticoid therapy (CGT).
METHODOLOGY:
- A retrospective review of 250-µg Synacthen test (SST) results performed in patients on tapering CGT doses from a single-center rheumatology department over 12 months.
- A total of 60 SSTs were performed in 58 patients, all in the morning (7-12 AM) after withholding CGT for 48 hours.
- Peripheral blood was sampled for cortisol at baseline, 30 minutes, and 60 minutes.
- Adrenal insufficiency (AI) was defined as a peak serum cortisol concentration.
TAKEAWAY:
- The mean duration of CGT (all prednisolone) was 63 months, prescribed primarily for giant cell arteritis/polymyalgia rheumatica (48%) and inflammatory arthritis (18%), with a mean daily dose of 3.4 mg at the time of SST.
- With the investigators’ previously reported basal serum cortisol concentration of > 237 nmol/L (> 8.6 μg/dL) used to confirm an intact HPA axis, no patient with AI would have been missed, but 37 of 51 (73%) unnecessary SSTs in euadrenal patients would have been avoided.
- A basal serum cortisol concentration of > 227 nmol/L had a specificity of 100% for predicting passing the SST, while a basal serum cortisol concentration of ≤ 55 nmol/L had a 100% sensitivity for predicting failure.
- A mean daily prednisolone dosing at the time of SST in patients with AI was significantly higher than that with normal SSTs (5.7 vs 2.9 mg, respectively; P = .01).
IN PRACTICE:
“This offers a more rapid, convenient, and cost‐effective screening method for patients requiring biochemical assessment of the HPA axis with the potential for significant resource savings without any adverse impact on patient safety,” the authors wrote.
SOURCE:
The study was conducted by Ella Sharma, of the Department of Endocrinology, Royal Victoria Infirmary, Newcastle upon Tyne, UK, and colleagues and published online on May 19, 2024, as a letter in Clinical Endocrinology.
LIMITATIONS:
Not provided.
DISCLOSURES:
Not provided.
A version of this article appeared on Medscape.com.
TOPLINE:
An early serum cortisol concentration of > 237 nmol/L (> 8.6 μg/dL) has been validated as a safe and useful screening test with 100% specificity for predicting recovery of the hypothalamic-pituitary-adrenal (HPA) axis in patients on tapering regimes from long‐term chronic glucocorticoid therapy (CGT).
METHODOLOGY:
- A retrospective review of 250-µg Synacthen test (SST) results performed in patients on tapering CGT doses from a single-center rheumatology department over 12 months.
- A total of 60 SSTs were performed in 58 patients, all in the morning (7-12 AM) after withholding CGT for 48 hours.
- Peripheral blood was sampled for cortisol at baseline, 30 minutes, and 60 minutes.
- Adrenal insufficiency (AI) was defined as a peak serum cortisol concentration.
TAKEAWAY:
- The mean duration of CGT (all prednisolone) was 63 months, prescribed primarily for giant cell arteritis/polymyalgia rheumatica (48%) and inflammatory arthritis (18%), with a mean daily dose of 3.4 mg at the time of SST.
- With the investigators’ previously reported basal serum cortisol concentration of > 237 nmol/L (> 8.6 μg/dL) used to confirm an intact HPA axis, no patient with AI would have been missed, but 37 of 51 (73%) unnecessary SSTs in euadrenal patients would have been avoided.
- A basal serum cortisol concentration of > 227 nmol/L had a specificity of 100% for predicting passing the SST, while a basal serum cortisol concentration of ≤ 55 nmol/L had a 100% sensitivity for predicting failure.
- A mean daily prednisolone dosing at the time of SST in patients with AI was significantly higher than that with normal SSTs (5.7 vs 2.9 mg, respectively; P = .01).
IN PRACTICE:
“This offers a more rapid, convenient, and cost‐effective screening method for patients requiring biochemical assessment of the HPA axis with the potential for significant resource savings without any adverse impact on patient safety,” the authors wrote.
SOURCE:
The study was conducted by Ella Sharma, of the Department of Endocrinology, Royal Victoria Infirmary, Newcastle upon Tyne, UK, and colleagues and published online on May 19, 2024, as a letter in Clinical Endocrinology.
LIMITATIONS:
Not provided.
DISCLOSURES:
Not provided.
A version of this article appeared on Medscape.com.
TOPLINE:
An early serum cortisol concentration of > 237 nmol/L (> 8.6 μg/dL) has been validated as a safe and useful screening test with 100% specificity for predicting recovery of the hypothalamic-pituitary-adrenal (HPA) axis in patients on tapering regimes from long‐term chronic glucocorticoid therapy (CGT).
METHODOLOGY:
- A retrospective review of 250-µg Synacthen test (SST) results performed in patients on tapering CGT doses from a single-center rheumatology department over 12 months.
- A total of 60 SSTs were performed in 58 patients, all in the morning (7-12 AM) after withholding CGT for 48 hours.
- Peripheral blood was sampled for cortisol at baseline, 30 minutes, and 60 minutes.
- Adrenal insufficiency (AI) was defined as a peak serum cortisol concentration.
TAKEAWAY:
- The mean duration of CGT (all prednisolone) was 63 months, prescribed primarily for giant cell arteritis/polymyalgia rheumatica (48%) and inflammatory arthritis (18%), with a mean daily dose of 3.4 mg at the time of SST.
- With the investigators’ previously reported basal serum cortisol concentration of > 237 nmol/L (> 8.6 μg/dL) used to confirm an intact HPA axis, no patient with AI would have been missed, but 37 of 51 (73%) unnecessary SSTs in euadrenal patients would have been avoided.
- A basal serum cortisol concentration of > 227 nmol/L had a specificity of 100% for predicting passing the SST, while a basal serum cortisol concentration of ≤ 55 nmol/L had a 100% sensitivity for predicting failure.
- A mean daily prednisolone dosing at the time of SST in patients with AI was significantly higher than that with normal SSTs (5.7 vs 2.9 mg, respectively; P = .01).
IN PRACTICE:
“This offers a more rapid, convenient, and cost‐effective screening method for patients requiring biochemical assessment of the HPA axis with the potential for significant resource savings without any adverse impact on patient safety,” the authors wrote.
SOURCE:
The study was conducted by Ella Sharma, of the Department of Endocrinology, Royal Victoria Infirmary, Newcastle upon Tyne, UK, and colleagues and published online on May 19, 2024, as a letter in Clinical Endocrinology.
LIMITATIONS:
Not provided.
DISCLOSURES:
Not provided.
A version of this article appeared on Medscape.com.
Key Risk Factors for Hydroxychloroquine Retinopathy Described in Large Study
Older patients prescribed hydroxychloroquine (HCQ) have a higher risk of developing retinal damage from taking the medication, according to a new analysis.
In addition to known risk factors such as a higher weight-based HCQ dose and higher cumulative dose, researchers also found that female sex, chronic kidney disease stage III, and tamoxifen use were associated with HCQ retinopathy.
The findings provide “evidence for other key risk factors for hydroxychloroquine retinopathy beyond hydroxychloroquine exposure itself,” wrote April M. Jorge, MD, of the Division of Rheumatology, Allergy, and Immunology at Massachusetts General Hospital, Boston, and colleagues.
“It is the largest cohort study to date looking specifically at the association of [HCQ] retinopathy with risk factors,” Christina Weng, MD, MBA, professor of ophthalmology at Baylor College of Medicine, Houston, said in an interview. She was not involved with the research. Some of the associations, such as tamoxifen use, “have been suggested before in smaller studies, but never on this scale,” she said.
“It’s provided reinforcement of findings that we have seen from prior research and also some new glimpses into strengthening some associations that were identified, but not yet fully understood, in prior work,” she continued.
Study Details
Researchers identified patients in the Kaiser Permanente Northern California (KPNC), Oakland, California, health system who began taking HCQ between July 1, 1997, and December 14, 2014. To be included, patients needed to have at least 5 years of continuous enrollment in the KPNC system and at least one prescription for HCQ after more than 5 years of starting the drug. Patients were followed from HCQ initiation to their last retinopathy screening study, up to December 31, 2020.
The study was published May 9 in JAMA Network Open.
Of the 4677 users followed for the study, 83% were women, and the average age starting HCQ was 52. Most patients were White (58.1%), while 13.7% were Asian, 10.5% were Black, and 17.7% were Hispanic.
More than 60% of patients had an initial dose > 5 mg/kg/d, though the mean initial dose of HCQ was 4.4 mg/kg/d. After 5 years, only 34.4% of patients were using a daily dose over 5 mg/kg.
Of the entire cohort, 125 patients (2.7%) developed HCQ retinopathy. As expected, cumulative HCQ exposure was associated with a higher retinopathy risk: For every 100 g of HCQ cumulative exposure, risk rose by 64% (hazard ratio [HR], 1.64; 95% CI, 1.44-1.87).
Age was a significant risk factor for retinal damage from HCQ use. Individuals who began taking the drug at 65 years or older were nearly six times more likely to develop retinopathy than those who started HCQ when they were younger than 45. In people aged 55-64 years, this risk was nearly four times higher, and individuals aged 45-54 years when starting the drug were 2.5 times more likely to have retinal damage than those younger than 45.
Other risk factors were female sex (HR, 3.83; 95% CI, 1.86-7.89), chronic kidney disease stage III (HR, 1.95; 95% CI, 1.25-3.04), and tamoxifen use (HR, 3.43; 95% CI, 1.08-10.89), although only 17 patients were taking tamoxifen during the study.
Researchers also found that the type of HCQ retinopathy varied by race. Of the 125 cases in the cohort, 102 had a parafoveal pattern, and 23 had a pericentral pattern. Asian individuals were 15 times more likely, and Black individuals were more than 5 times more likely to develop this pericentral type than were White patients.
This association in Asian patients has also been found in previous studies, Dr. Weng said, and many eye practices now screen their Asian patients with a 30-2 Humphrey visual field — rather than the more commonly used 10-2 — to examine areas farther outside the center.
This study also found this association in Black patients, though only five Black patients developed HCQ retinopathy over the study period.
“More studies and larger studies will be very helpful in strengthening or dispelling some of the associations that have been seen here,” Dr. Weng said.
‘More Room for Personalized Medicine’
The team found a “relatively linear” relationship between HCQ dose and retinopathy risk, with higher daily doses correlating with higher incidence. While 2016 guidelines from the American Academy of Ophthalmology advise using < 5 mg/kg, “what we found is it’s not that straightforward [where there’s] just this one cutoff,” Dr. Jorge told this news organization. “It does seem like the higher the dose of medication per bodyweight and the longer duration of use, there is a higher risk of retinopathy.”
These findings leave “a bit more room for personalized medicine” with patients, she explained. “For an elderly female patient with CKD, aiming for a dose < 5 mg/kg might be more appropriate; however, a young male patient without any additional risk factors may be able to exceed 5 mg/kg and continue to have a low risk for HCQ retinopathy,” she said.
“For anyone, I think it really is more of an individual risk-benefit evaluation,” rather than strict cutoffs, she continued.
“Guidelines are just that: They’re guidelines,” added Dr. Weng, “and treatment plans should be tailored to each individual patient.”
As the study authors also discussed, “the goal is to treat the patient with the lowest dose that is still effective and also be mindful of the duration that a patient is left at higher doses,” Dr. Weng said. “But ultimately, we need to control these diseases, which can cause damage across multiple organ systems in the body. While it’s important to be aware of the potential retinopathy adverse events, we also don’t want physicians to feel restricted in their use of this very effective drug.”
The work of three coauthors on the current study was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS). Dr. Jorge’s work on the study was supported by an award from the Rheumatology Research Foundation and a grant from NIAMS. Dr. Jorge reported clinical trial agreements with Bristol Myers Squibb and Cabaletta Bio outside of this study. Dr. Weng has served as a consultant for Allergan/AbbVie, Alcon, Apellis Pharmaceuticals, Alimera Sciences, DORC, Novartis, Genentech, Regeneron, RegenxBio, Iveric Bio, and EyePoint Pharmaceuticals. Dr. Weng disclosed financial relationships with Springer Publishers (royalties) and DRCR Retina Network, Alimera Sciences, and Applied Genetic Technologies Corporation (research).
A version of this article appeared on Medscape.com.
Older patients prescribed hydroxychloroquine (HCQ) have a higher risk of developing retinal damage from taking the medication, according to a new analysis.
In addition to known risk factors such as a higher weight-based HCQ dose and higher cumulative dose, researchers also found that female sex, chronic kidney disease stage III, and tamoxifen use were associated with HCQ retinopathy.
The findings provide “evidence for other key risk factors for hydroxychloroquine retinopathy beyond hydroxychloroquine exposure itself,” wrote April M. Jorge, MD, of the Division of Rheumatology, Allergy, and Immunology at Massachusetts General Hospital, Boston, and colleagues.
“It is the largest cohort study to date looking specifically at the association of [HCQ] retinopathy with risk factors,” Christina Weng, MD, MBA, professor of ophthalmology at Baylor College of Medicine, Houston, said in an interview. She was not involved with the research. Some of the associations, such as tamoxifen use, “have been suggested before in smaller studies, but never on this scale,” she said.
“It’s provided reinforcement of findings that we have seen from prior research and also some new glimpses into strengthening some associations that were identified, but not yet fully understood, in prior work,” she continued.
Study Details
Researchers identified patients in the Kaiser Permanente Northern California (KPNC), Oakland, California, health system who began taking HCQ between July 1, 1997, and December 14, 2014. To be included, patients needed to have at least 5 years of continuous enrollment in the KPNC system and at least one prescription for HCQ after more than 5 years of starting the drug. Patients were followed from HCQ initiation to their last retinopathy screening study, up to December 31, 2020.
The study was published May 9 in JAMA Network Open.
Of the 4677 users followed for the study, 83% were women, and the average age starting HCQ was 52. Most patients were White (58.1%), while 13.7% were Asian, 10.5% were Black, and 17.7% were Hispanic.
More than 60% of patients had an initial dose > 5 mg/kg/d, though the mean initial dose of HCQ was 4.4 mg/kg/d. After 5 years, only 34.4% of patients were using a daily dose over 5 mg/kg.
Of the entire cohort, 125 patients (2.7%) developed HCQ retinopathy. As expected, cumulative HCQ exposure was associated with a higher retinopathy risk: For every 100 g of HCQ cumulative exposure, risk rose by 64% (hazard ratio [HR], 1.64; 95% CI, 1.44-1.87).
Age was a significant risk factor for retinal damage from HCQ use. Individuals who began taking the drug at 65 years or older were nearly six times more likely to develop retinopathy than those who started HCQ when they were younger than 45. In people aged 55-64 years, this risk was nearly four times higher, and individuals aged 45-54 years when starting the drug were 2.5 times more likely to have retinal damage than those younger than 45.
Other risk factors were female sex (HR, 3.83; 95% CI, 1.86-7.89), chronic kidney disease stage III (HR, 1.95; 95% CI, 1.25-3.04), and tamoxifen use (HR, 3.43; 95% CI, 1.08-10.89), although only 17 patients were taking tamoxifen during the study.
Researchers also found that the type of HCQ retinopathy varied by race. Of the 125 cases in the cohort, 102 had a parafoveal pattern, and 23 had a pericentral pattern. Asian individuals were 15 times more likely, and Black individuals were more than 5 times more likely to develop this pericentral type than were White patients.
This association in Asian patients has also been found in previous studies, Dr. Weng said, and many eye practices now screen their Asian patients with a 30-2 Humphrey visual field — rather than the more commonly used 10-2 — to examine areas farther outside the center.
This study also found this association in Black patients, though only five Black patients developed HCQ retinopathy over the study period.
“More studies and larger studies will be very helpful in strengthening or dispelling some of the associations that have been seen here,” Dr. Weng said.
‘More Room for Personalized Medicine’
The team found a “relatively linear” relationship between HCQ dose and retinopathy risk, with higher daily doses correlating with higher incidence. While 2016 guidelines from the American Academy of Ophthalmology advise using < 5 mg/kg, “what we found is it’s not that straightforward [where there’s] just this one cutoff,” Dr. Jorge told this news organization. “It does seem like the higher the dose of medication per bodyweight and the longer duration of use, there is a higher risk of retinopathy.”
These findings leave “a bit more room for personalized medicine” with patients, she explained. “For an elderly female patient with CKD, aiming for a dose < 5 mg/kg might be more appropriate; however, a young male patient without any additional risk factors may be able to exceed 5 mg/kg and continue to have a low risk for HCQ retinopathy,” she said.
“For anyone, I think it really is more of an individual risk-benefit evaluation,” rather than strict cutoffs, she continued.
“Guidelines are just that: They’re guidelines,” added Dr. Weng, “and treatment plans should be tailored to each individual patient.”
As the study authors also discussed, “the goal is to treat the patient with the lowest dose that is still effective and also be mindful of the duration that a patient is left at higher doses,” Dr. Weng said. “But ultimately, we need to control these diseases, which can cause damage across multiple organ systems in the body. While it’s important to be aware of the potential retinopathy adverse events, we also don’t want physicians to feel restricted in their use of this very effective drug.”
The work of three coauthors on the current study was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS). Dr. Jorge’s work on the study was supported by an award from the Rheumatology Research Foundation and a grant from NIAMS. Dr. Jorge reported clinical trial agreements with Bristol Myers Squibb and Cabaletta Bio outside of this study. Dr. Weng has served as a consultant for Allergan/AbbVie, Alcon, Apellis Pharmaceuticals, Alimera Sciences, DORC, Novartis, Genentech, Regeneron, RegenxBio, Iveric Bio, and EyePoint Pharmaceuticals. Dr. Weng disclosed financial relationships with Springer Publishers (royalties) and DRCR Retina Network, Alimera Sciences, and Applied Genetic Technologies Corporation (research).
A version of this article appeared on Medscape.com.
Older patients prescribed hydroxychloroquine (HCQ) have a higher risk of developing retinal damage from taking the medication, according to a new analysis.
In addition to known risk factors such as a higher weight-based HCQ dose and higher cumulative dose, researchers also found that female sex, chronic kidney disease stage III, and tamoxifen use were associated with HCQ retinopathy.
The findings provide “evidence for other key risk factors for hydroxychloroquine retinopathy beyond hydroxychloroquine exposure itself,” wrote April M. Jorge, MD, of the Division of Rheumatology, Allergy, and Immunology at Massachusetts General Hospital, Boston, and colleagues.
“It is the largest cohort study to date looking specifically at the association of [HCQ] retinopathy with risk factors,” Christina Weng, MD, MBA, professor of ophthalmology at Baylor College of Medicine, Houston, said in an interview. She was not involved with the research. Some of the associations, such as tamoxifen use, “have been suggested before in smaller studies, but never on this scale,” she said.
“It’s provided reinforcement of findings that we have seen from prior research and also some new glimpses into strengthening some associations that were identified, but not yet fully understood, in prior work,” she continued.
Study Details
Researchers identified patients in the Kaiser Permanente Northern California (KPNC), Oakland, California, health system who began taking HCQ between July 1, 1997, and December 14, 2014. To be included, patients needed to have at least 5 years of continuous enrollment in the KPNC system and at least one prescription for HCQ after more than 5 years of starting the drug. Patients were followed from HCQ initiation to their last retinopathy screening study, up to December 31, 2020.
The study was published May 9 in JAMA Network Open.
Of the 4677 users followed for the study, 83% were women, and the average age starting HCQ was 52. Most patients were White (58.1%), while 13.7% were Asian, 10.5% were Black, and 17.7% were Hispanic.
More than 60% of patients had an initial dose > 5 mg/kg/d, though the mean initial dose of HCQ was 4.4 mg/kg/d. After 5 years, only 34.4% of patients were using a daily dose over 5 mg/kg.
Of the entire cohort, 125 patients (2.7%) developed HCQ retinopathy. As expected, cumulative HCQ exposure was associated with a higher retinopathy risk: For every 100 g of HCQ cumulative exposure, risk rose by 64% (hazard ratio [HR], 1.64; 95% CI, 1.44-1.87).
Age was a significant risk factor for retinal damage from HCQ use. Individuals who began taking the drug at 65 years or older were nearly six times more likely to develop retinopathy than those who started HCQ when they were younger than 45. In people aged 55-64 years, this risk was nearly four times higher, and individuals aged 45-54 years when starting the drug were 2.5 times more likely to have retinal damage than those younger than 45.
Other risk factors were female sex (HR, 3.83; 95% CI, 1.86-7.89), chronic kidney disease stage III (HR, 1.95; 95% CI, 1.25-3.04), and tamoxifen use (HR, 3.43; 95% CI, 1.08-10.89), although only 17 patients were taking tamoxifen during the study.
Researchers also found that the type of HCQ retinopathy varied by race. Of the 125 cases in the cohort, 102 had a parafoveal pattern, and 23 had a pericentral pattern. Asian individuals were 15 times more likely, and Black individuals were more than 5 times more likely to develop this pericentral type than were White patients.
This association in Asian patients has also been found in previous studies, Dr. Weng said, and many eye practices now screen their Asian patients with a 30-2 Humphrey visual field — rather than the more commonly used 10-2 — to examine areas farther outside the center.
This study also found this association in Black patients, though only five Black patients developed HCQ retinopathy over the study period.
“More studies and larger studies will be very helpful in strengthening or dispelling some of the associations that have been seen here,” Dr. Weng said.
‘More Room for Personalized Medicine’
The team found a “relatively linear” relationship between HCQ dose and retinopathy risk, with higher daily doses correlating with higher incidence. While 2016 guidelines from the American Academy of Ophthalmology advise using < 5 mg/kg, “what we found is it’s not that straightforward [where there’s] just this one cutoff,” Dr. Jorge told this news organization. “It does seem like the higher the dose of medication per bodyweight and the longer duration of use, there is a higher risk of retinopathy.”
These findings leave “a bit more room for personalized medicine” with patients, she explained. “For an elderly female patient with CKD, aiming for a dose < 5 mg/kg might be more appropriate; however, a young male patient without any additional risk factors may be able to exceed 5 mg/kg and continue to have a low risk for HCQ retinopathy,” she said.
“For anyone, I think it really is more of an individual risk-benefit evaluation,” rather than strict cutoffs, she continued.
“Guidelines are just that: They’re guidelines,” added Dr. Weng, “and treatment plans should be tailored to each individual patient.”
As the study authors also discussed, “the goal is to treat the patient with the lowest dose that is still effective and also be mindful of the duration that a patient is left at higher doses,” Dr. Weng said. “But ultimately, we need to control these diseases, which can cause damage across multiple organ systems in the body. While it’s important to be aware of the potential retinopathy adverse events, we also don’t want physicians to feel restricted in their use of this very effective drug.”
The work of three coauthors on the current study was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS). Dr. Jorge’s work on the study was supported by an award from the Rheumatology Research Foundation and a grant from NIAMS. Dr. Jorge reported clinical trial agreements with Bristol Myers Squibb and Cabaletta Bio outside of this study. Dr. Weng has served as a consultant for Allergan/AbbVie, Alcon, Apellis Pharmaceuticals, Alimera Sciences, DORC, Novartis, Genentech, Regeneron, RegenxBio, Iveric Bio, and EyePoint Pharmaceuticals. Dr. Weng disclosed financial relationships with Springer Publishers (royalties) and DRCR Retina Network, Alimera Sciences, and Applied Genetic Technologies Corporation (research).
A version of this article appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Don’t Leave CVD Risk in RA Undertreated Despite Unresolved Questions
NEW YORK — Patients with rheumatoid arthritis (RA) carry a high risk for cardiovascular events, but mounting clinical evidence suggests they’re being undertreated to manage that risk. Rheumatologists should consider a patient with RA’s cardiovascular disease (CVD) status before deciding on RA treatments, a researcher of cardiometabolic disorders advised.
“The ORAL Surveillance trial suggests that we need to consider cardiovascular risk factors and maybe do additional screening in these patients before we use RA therapies,” Jon T. Giles, MD, PhD, director of the Cedars-Sinai Inflammatory Arthritis Clinical Center at Cedars-Sinai in Los Angeles, told attendees at the 4th Annual Cardiometabolic Risk in Inflammatory Conditions conference.
Underuse of Statins
ORAL Surveillance enrolled 4362 patients with RA aged 50 years and older with at least one cardiovascular risk factor. About 23% of all patients were taking statins, as were about half of patients with a history of atherosclerotic CVD (ASCVD).
“A lot of those people should have been on statins,” Dr. Giles said in an interview. “Not because of their RA but because of their risk factors, and then RA brings it up another notch.” In the population with ASCVD, Dr. Giles added, “It should have been more like 70% and 80%. If we’re talking about a disease that has enhanced cardiovascular risk, then the adoption of standard care that you would do for anybody in the general population should be at that standard and maybe above.”
Multiple studies have documented the underlying risk for CVD events, CV mortality, and subclinical atherosclerosis in people with RA, Dr. Giles noted in his presentation. Physiologically, the RA-specific risk factors most linked to CVD risk are systemic inflammation/cytokine excess and specific circulating T-cell and intermediate monocyte subsets, or both, Dr. Giles said.
Disease-Modifying Antirheumatic Drugs (DMARDs) and CVD Risk
Likewise, research in the past decade has linked methotrexate and tumor necrosis factor (TNF) inhibitors to reduced ASCVD events in RA. Another study showed that abatacept had an effect similar to that of etanercept in patients with RA, and the ENTRACTE trial, for which Dr. Giles was the lead author, demonstrated that tocilizumab matched etanercept in reducing CV events.
The ORAL Surveillance investigators also reported that patients with RA who were receiving the Janus kinase (JAK) inhibitor tofacitinib had a higher risk for major adverse cardiovascular events and cancers than those on TNF therapy, Dr. Giles noted. While statins in combination with JAK inhibitors may have the potential to provide a balance for controlling CV risk in patients with RA, he said later that the potential of JAK inhibitors in reducing CVD risk in RA “is still unsettled.”
The ongoing TARGET trial is further evaluating the impact of DMARDs on vascular inflammation in RA, said Dr. Giles, who’s also a trial principal investigator. TARGET is randomizing 115 patients with RA who didn’t respond to methotrexate to a TNF inhibitor or the addition of sulfasalazine and hydroxychloroquine to their methotrexate. Patients can be on low-intensity but not high-intensity statin therapy, Dr. Giles said.
TARGET results reported last year demonstrated an 8% decrease in arterial fluorodeoxyglucose (FDG) uptake on PET-CT in both treatment arms. Previous studies, Dr. Giles noted, have shown a potential link between FDG and histologic markers of inflammation. “An 8% decrease in vascular FDG is in line with what you would expect from statin treatment,” he said.
TARGET results published in April showed that a measure of a cluster of 12 cytokines and other inflammatory mediators, known as the multibiomarker disease activity (MBDA) score and marketed under the brand name Vectra DA, may help determine arterial FDG uptake. “Those who had a low MBDA score at week 24 actually had the greatest reduction in the arterial FDG,” he said.
Those results were driven entirely by low serum amyloid A (SAA) levels, Dr. Giles said. Those same results didn’t hold for patients in whom SAA and C-reactive protein were correlated.
“So, there’s more to come here,” Dr. Giles said. “We’re looking at other, much larger biomarker panels.”
Nonetheless, he said, sufficient evidence exists to conclude that treating RA to target reduces CV events. “The idea is that at every visit that you see an RA patient, you measure their disease activity, and if they’re not at the target of low disease activity or remission, then you change their therapy to improve that,” he said in an interview.
But an evidence-based guideline is needed to improve coverage of CVD risks in patients with RA, Dr. Giles said. “There is a movement afoot” for a guideline, he said. “If you just did what is supposed to happen for a general population, you would make some improvements. The risk-benefit [ratio] for statins for people with RA has been looked at, and it’s very favorable.”
Unanswered Questions
Dr. Giles noted that the ORAL Surveillance trial has left a number of questions unanswered about the role of JAK inhibitors in managing CVD risk in patients with RA. “The issue that we’re trying to ask is, is it just the TNF inhibitors may be better? Is this a subpopulation issue, or was it just bad luck from the purposes of this one trial? Granted, it was a very large trial, but you can still have luck in terms of getting an effect that’s not accurate.”
Dr. Giles’ “gut feeling” on JAK inhibitors is that they’re not causing harm, but that they’re not as effective as TNF inhibitors in ameliorating CV risks in patients with RA.
Michael S. Garshick, MD, who attended the conference and is head of the cardio-rheumatology program at NYU Langone Health, concurred that a number of unanswered questions persist over the treatment of CVD risk in RA — and autoimmune disease in general.
“I think we’re still trying to prove that DMARDs reduce cardiovascular risk in autoimmune conditions,” he said. “The epidemiologic data would suggest, yes, that inflammation prevention is beneficial for cardiovascular disease, but the TARGET trial suggested that vascular inflammation improved by treating RA, but that biologic therapy wasn’t better than traditional triple therapy.”
Other questions remain unanswered, Dr. Garshick said.
“Is there a specific immunotherapy that is most beneficial to reduce heart disease in patients with an autoimmune condition, whether it’s rheumatoid arthritis, psoriasis, or lupus?”
Dr. Garshick said he’s specifically interested in the residual risk that exists after treating the autoimmunity. “Do you still have a higher risk for heart disease, and if so, why? Is there something else going on that we can’t see?”
The biggest unanswered question, he said, is “How can we do a better job of recognizing heart disease risk in these patients? That’s the low-hanging fruit that people are studying, but across many of those studies, patients have higher rates of blood pressure, cholesterol issues, obesity, diabetes, and many times, we’re not adequately treating these comorbidities.”
That, Dr. Garshick said, may be a result of physician fatigue. “And so [treatment of these comorbidities is] kicked down the road for a year or years,” he added.
Dr. Giles disclosed financial relationships with Pfizer, AbbVie, Eli Lilly, and Novartis. Dr. Garshick disclosed relationships with Kiniksa Pharmaceuticals, Agepha Pharma, Bristol Myers Squibb, and Horizon Therapeutics.
A version of this article appeared on Medscape.com.
NEW YORK — Patients with rheumatoid arthritis (RA) carry a high risk for cardiovascular events, but mounting clinical evidence suggests they’re being undertreated to manage that risk. Rheumatologists should consider a patient with RA’s cardiovascular disease (CVD) status before deciding on RA treatments, a researcher of cardiometabolic disorders advised.
“The ORAL Surveillance trial suggests that we need to consider cardiovascular risk factors and maybe do additional screening in these patients before we use RA therapies,” Jon T. Giles, MD, PhD, director of the Cedars-Sinai Inflammatory Arthritis Clinical Center at Cedars-Sinai in Los Angeles, told attendees at the 4th Annual Cardiometabolic Risk in Inflammatory Conditions conference.
Underuse of Statins
ORAL Surveillance enrolled 4362 patients with RA aged 50 years and older with at least one cardiovascular risk factor. About 23% of all patients were taking statins, as were about half of patients with a history of atherosclerotic CVD (ASCVD).
“A lot of those people should have been on statins,” Dr. Giles said in an interview. “Not because of their RA but because of their risk factors, and then RA brings it up another notch.” In the population with ASCVD, Dr. Giles added, “It should have been more like 70% and 80%. If we’re talking about a disease that has enhanced cardiovascular risk, then the adoption of standard care that you would do for anybody in the general population should be at that standard and maybe above.”
Multiple studies have documented the underlying risk for CVD events, CV mortality, and subclinical atherosclerosis in people with RA, Dr. Giles noted in his presentation. Physiologically, the RA-specific risk factors most linked to CVD risk are systemic inflammation/cytokine excess and specific circulating T-cell and intermediate monocyte subsets, or both, Dr. Giles said.
Disease-Modifying Antirheumatic Drugs (DMARDs) and CVD Risk
Likewise, research in the past decade has linked methotrexate and tumor necrosis factor (TNF) inhibitors to reduced ASCVD events in RA. Another study showed that abatacept had an effect similar to that of etanercept in patients with RA, and the ENTRACTE trial, for which Dr. Giles was the lead author, demonstrated that tocilizumab matched etanercept in reducing CV events.
The ORAL Surveillance investigators also reported that patients with RA who were receiving the Janus kinase (JAK) inhibitor tofacitinib had a higher risk for major adverse cardiovascular events and cancers than those on TNF therapy, Dr. Giles noted. While statins in combination with JAK inhibitors may have the potential to provide a balance for controlling CV risk in patients with RA, he said later that the potential of JAK inhibitors in reducing CVD risk in RA “is still unsettled.”
The ongoing TARGET trial is further evaluating the impact of DMARDs on vascular inflammation in RA, said Dr. Giles, who’s also a trial principal investigator. TARGET is randomizing 115 patients with RA who didn’t respond to methotrexate to a TNF inhibitor or the addition of sulfasalazine and hydroxychloroquine to their methotrexate. Patients can be on low-intensity but not high-intensity statin therapy, Dr. Giles said.
TARGET results reported last year demonstrated an 8% decrease in arterial fluorodeoxyglucose (FDG) uptake on PET-CT in both treatment arms. Previous studies, Dr. Giles noted, have shown a potential link between FDG and histologic markers of inflammation. “An 8% decrease in vascular FDG is in line with what you would expect from statin treatment,” he said.
TARGET results published in April showed that a measure of a cluster of 12 cytokines and other inflammatory mediators, known as the multibiomarker disease activity (MBDA) score and marketed under the brand name Vectra DA, may help determine arterial FDG uptake. “Those who had a low MBDA score at week 24 actually had the greatest reduction in the arterial FDG,” he said.
Those results were driven entirely by low serum amyloid A (SAA) levels, Dr. Giles said. Those same results didn’t hold for patients in whom SAA and C-reactive protein were correlated.
“So, there’s more to come here,” Dr. Giles said. “We’re looking at other, much larger biomarker panels.”
Nonetheless, he said, sufficient evidence exists to conclude that treating RA to target reduces CV events. “The idea is that at every visit that you see an RA patient, you measure their disease activity, and if they’re not at the target of low disease activity or remission, then you change their therapy to improve that,” he said in an interview.
But an evidence-based guideline is needed to improve coverage of CVD risks in patients with RA, Dr. Giles said. “There is a movement afoot” for a guideline, he said. “If you just did what is supposed to happen for a general population, you would make some improvements. The risk-benefit [ratio] for statins for people with RA has been looked at, and it’s very favorable.”
Unanswered Questions
Dr. Giles noted that the ORAL Surveillance trial has left a number of questions unanswered about the role of JAK inhibitors in managing CVD risk in patients with RA. “The issue that we’re trying to ask is, is it just the TNF inhibitors may be better? Is this a subpopulation issue, or was it just bad luck from the purposes of this one trial? Granted, it was a very large trial, but you can still have luck in terms of getting an effect that’s not accurate.”
Dr. Giles’ “gut feeling” on JAK inhibitors is that they’re not causing harm, but that they’re not as effective as TNF inhibitors in ameliorating CV risks in patients with RA.
Michael S. Garshick, MD, who attended the conference and is head of the cardio-rheumatology program at NYU Langone Health, concurred that a number of unanswered questions persist over the treatment of CVD risk in RA — and autoimmune disease in general.
“I think we’re still trying to prove that DMARDs reduce cardiovascular risk in autoimmune conditions,” he said. “The epidemiologic data would suggest, yes, that inflammation prevention is beneficial for cardiovascular disease, but the TARGET trial suggested that vascular inflammation improved by treating RA, but that biologic therapy wasn’t better than traditional triple therapy.”
Other questions remain unanswered, Dr. Garshick said.
“Is there a specific immunotherapy that is most beneficial to reduce heart disease in patients with an autoimmune condition, whether it’s rheumatoid arthritis, psoriasis, or lupus?”
Dr. Garshick said he’s specifically interested in the residual risk that exists after treating the autoimmunity. “Do you still have a higher risk for heart disease, and if so, why? Is there something else going on that we can’t see?”
The biggest unanswered question, he said, is “How can we do a better job of recognizing heart disease risk in these patients? That’s the low-hanging fruit that people are studying, but across many of those studies, patients have higher rates of blood pressure, cholesterol issues, obesity, diabetes, and many times, we’re not adequately treating these comorbidities.”
That, Dr. Garshick said, may be a result of physician fatigue. “And so [treatment of these comorbidities is] kicked down the road for a year or years,” he added.
Dr. Giles disclosed financial relationships with Pfizer, AbbVie, Eli Lilly, and Novartis. Dr. Garshick disclosed relationships with Kiniksa Pharmaceuticals, Agepha Pharma, Bristol Myers Squibb, and Horizon Therapeutics.
A version of this article appeared on Medscape.com.
NEW YORK — Patients with rheumatoid arthritis (RA) carry a high risk for cardiovascular events, but mounting clinical evidence suggests they’re being undertreated to manage that risk. Rheumatologists should consider a patient with RA’s cardiovascular disease (CVD) status before deciding on RA treatments, a researcher of cardiometabolic disorders advised.
“The ORAL Surveillance trial suggests that we need to consider cardiovascular risk factors and maybe do additional screening in these patients before we use RA therapies,” Jon T. Giles, MD, PhD, director of the Cedars-Sinai Inflammatory Arthritis Clinical Center at Cedars-Sinai in Los Angeles, told attendees at the 4th Annual Cardiometabolic Risk in Inflammatory Conditions conference.
Underuse of Statins
ORAL Surveillance enrolled 4362 patients with RA aged 50 years and older with at least one cardiovascular risk factor. About 23% of all patients were taking statins, as were about half of patients with a history of atherosclerotic CVD (ASCVD).
“A lot of those people should have been on statins,” Dr. Giles said in an interview. “Not because of their RA but because of their risk factors, and then RA brings it up another notch.” In the population with ASCVD, Dr. Giles added, “It should have been more like 70% and 80%. If we’re talking about a disease that has enhanced cardiovascular risk, then the adoption of standard care that you would do for anybody in the general population should be at that standard and maybe above.”
Multiple studies have documented the underlying risk for CVD events, CV mortality, and subclinical atherosclerosis in people with RA, Dr. Giles noted in his presentation. Physiologically, the RA-specific risk factors most linked to CVD risk are systemic inflammation/cytokine excess and specific circulating T-cell and intermediate monocyte subsets, or both, Dr. Giles said.
Disease-Modifying Antirheumatic Drugs (DMARDs) and CVD Risk
Likewise, research in the past decade has linked methotrexate and tumor necrosis factor (TNF) inhibitors to reduced ASCVD events in RA. Another study showed that abatacept had an effect similar to that of etanercept in patients with RA, and the ENTRACTE trial, for which Dr. Giles was the lead author, demonstrated that tocilizumab matched etanercept in reducing CV events.
The ORAL Surveillance investigators also reported that patients with RA who were receiving the Janus kinase (JAK) inhibitor tofacitinib had a higher risk for major adverse cardiovascular events and cancers than those on TNF therapy, Dr. Giles noted. While statins in combination with JAK inhibitors may have the potential to provide a balance for controlling CV risk in patients with RA, he said later that the potential of JAK inhibitors in reducing CVD risk in RA “is still unsettled.”
The ongoing TARGET trial is further evaluating the impact of DMARDs on vascular inflammation in RA, said Dr. Giles, who’s also a trial principal investigator. TARGET is randomizing 115 patients with RA who didn’t respond to methotrexate to a TNF inhibitor or the addition of sulfasalazine and hydroxychloroquine to their methotrexate. Patients can be on low-intensity but not high-intensity statin therapy, Dr. Giles said.
TARGET results reported last year demonstrated an 8% decrease in arterial fluorodeoxyglucose (FDG) uptake on PET-CT in both treatment arms. Previous studies, Dr. Giles noted, have shown a potential link between FDG and histologic markers of inflammation. “An 8% decrease in vascular FDG is in line with what you would expect from statin treatment,” he said.
TARGET results published in April showed that a measure of a cluster of 12 cytokines and other inflammatory mediators, known as the multibiomarker disease activity (MBDA) score and marketed under the brand name Vectra DA, may help determine arterial FDG uptake. “Those who had a low MBDA score at week 24 actually had the greatest reduction in the arterial FDG,” he said.
Those results were driven entirely by low serum amyloid A (SAA) levels, Dr. Giles said. Those same results didn’t hold for patients in whom SAA and C-reactive protein were correlated.
“So, there’s more to come here,” Dr. Giles said. “We’re looking at other, much larger biomarker panels.”
Nonetheless, he said, sufficient evidence exists to conclude that treating RA to target reduces CV events. “The idea is that at every visit that you see an RA patient, you measure their disease activity, and if they’re not at the target of low disease activity or remission, then you change their therapy to improve that,” he said in an interview.
But an evidence-based guideline is needed to improve coverage of CVD risks in patients with RA, Dr. Giles said. “There is a movement afoot” for a guideline, he said. “If you just did what is supposed to happen for a general population, you would make some improvements. The risk-benefit [ratio] for statins for people with RA has been looked at, and it’s very favorable.”
Unanswered Questions
Dr. Giles noted that the ORAL Surveillance trial has left a number of questions unanswered about the role of JAK inhibitors in managing CVD risk in patients with RA. “The issue that we’re trying to ask is, is it just the TNF inhibitors may be better? Is this a subpopulation issue, or was it just bad luck from the purposes of this one trial? Granted, it was a very large trial, but you can still have luck in terms of getting an effect that’s not accurate.”
Dr. Giles’ “gut feeling” on JAK inhibitors is that they’re not causing harm, but that they’re not as effective as TNF inhibitors in ameliorating CV risks in patients with RA.
Michael S. Garshick, MD, who attended the conference and is head of the cardio-rheumatology program at NYU Langone Health, concurred that a number of unanswered questions persist over the treatment of CVD risk in RA — and autoimmune disease in general.
“I think we’re still trying to prove that DMARDs reduce cardiovascular risk in autoimmune conditions,” he said. “The epidemiologic data would suggest, yes, that inflammation prevention is beneficial for cardiovascular disease, but the TARGET trial suggested that vascular inflammation improved by treating RA, but that biologic therapy wasn’t better than traditional triple therapy.”
Other questions remain unanswered, Dr. Garshick said.
“Is there a specific immunotherapy that is most beneficial to reduce heart disease in patients with an autoimmune condition, whether it’s rheumatoid arthritis, psoriasis, or lupus?”
Dr. Garshick said he’s specifically interested in the residual risk that exists after treating the autoimmunity. “Do you still have a higher risk for heart disease, and if so, why? Is there something else going on that we can’t see?”
The biggest unanswered question, he said, is “How can we do a better job of recognizing heart disease risk in these patients? That’s the low-hanging fruit that people are studying, but across many of those studies, patients have higher rates of blood pressure, cholesterol issues, obesity, diabetes, and many times, we’re not adequately treating these comorbidities.”
That, Dr. Garshick said, may be a result of physician fatigue. “And so [treatment of these comorbidities is] kicked down the road for a year or years,” he added.
Dr. Giles disclosed financial relationships with Pfizer, AbbVie, Eli Lilly, and Novartis. Dr. Garshick disclosed relationships with Kiniksa Pharmaceuticals, Agepha Pharma, Bristol Myers Squibb, and Horizon Therapeutics.
A version of this article appeared on Medscape.com.
Specialists Are ‘Underwater’ With Some Insurance-Preferred Biosimilars
Editor’s note: This article is adapted from an explanatory statement that Dr. Feldman wrote for the Coalition of State Rheumatology Organizations (CSRO).
According to the Guinness Book of World records, the longest time someone has held their breath underwater voluntarily is 24 minutes and 37.36 seconds. While certainly an amazing feat, UnitedHealthcare, many of the Blues, and other national “payers” are expecting rheumatologists and other specialists to live “underwater” in order to take care of their patients. In other words, these insurance companies are mandating that specialists use certain provider-administered biosimilars whose acquisition cost is higher than what the insurance company is willing to reimburse them. Essentially, the insurance companies expect the rheumatologists to pay them to take care of their patients. Because of the substantial and destabilizing financial losses incurred, many practices and free-standing infusion centers have been forced to cease offering these biosimilars. Most rheumatologists will provide patients with appropriate alternatives when available and permitted by the insurer; otherwise, they must refer patients to hospital-based infusion centers. That results in delayed care and increased costs for patients and the system, because hospital-based infusion typically costs more than twice what office-based infusion costs.
Quantifying the Problem
To help quantify the magnitude of this issue, the Coalition of State Rheumatology Organizations (CSRO) recently conducted a survey of its membership. A shocking 97% of respondents reported that their practice had been affected by reimbursement rates for some biosimilars being lower than acquisition costs, with 91% of respondents stating that this issue is more pronounced for certain biosimilars than others. Across the board, respondents most frequently identified Inflectra (infliximab-dyyb) and Avsola (infliximab-axxq) as being especially affected: Over 88% and over 85% of respondents identified these two products, respectively, as being underwater. These results support the ongoing anecdotal reports CSRO continues to receive from rheumatology practices.
However, the survey results indicated that this issue is by no means confined to those two biosimilars. Truxima (rituximab-abbs) — a biosimilar for Rituxan — was frequently mentioned as well. Notably, respondents almost uniformly identified biosimilars in the infliximab and rituximab families, which illustrates that this issue is no longer confined to one or two early-to-market biosimilars but has almost become a hallmark of this particular biosimilars market. Remarkably, one respondent commented that the brand products are now cheaper to acquire than the biosimilars. Furthermore, the survey included respondents from across the country, indicating that this issue is not confined to a particular region.
How Did This Happen?
Biosimilars held promise for increasing availability and decreasing biologic costs for patients but, thus far, no patients have seen their cost go down. It appears that the only biosimilars that have made it to “preferred” status on the formulary are the ones that have made more money for the middlemen in the drug supply chain, particularly those that construct formularies. Now, we have provider-administered biosimilars whose acquisition cost exceeds the reimbursement for these drugs. This disparity was ultimately created by biosimilar manufacturers “over-rebating” their drugs to health insurance companies to gain “fail-first” status on the formulary.
For example, the manufacturer of Inflectra offered substantial rebates to health insurers for preferred formulary placement. These rebates are factored into the sales price of the medication, which then results in a rapidly declining average sales price (ASP) for the biosimilar. Unfortunately, the acquisition cost for the drug does not experience commensurate reductions, resulting in physicians being reimbursed far less for the drug than it costs to acquire. The financial losses for physicians put them underwater as a result of the acquisition costs for the preferred drugs far surpassing the reimbursement from the health insurance company that constructed the formulary.
While various factors affect ASPs and acquisition costs, this particular consequence of formulary placement based on price concessions is a major driver of the underwater situation in which physicians have found themselves with many biosimilars. Not only does that lead to a lower uptake of biosimilars, but it also results in patients being referred to the hospital outpatient infusion sites to receive this care, as freestanding infusion centers cannot treat these patients either. Hospitals incur higher costs because of facility fees and elevated rates, and this makes private rheumatology in-office infusion centers a much lower-cost option. Similarly, home infusion services, while convenient, are marginally more expensive than private practices and, in cases of biologic infusions, it is important to note that physicians’ offices have a greater safety profile than home infusion of biologics. The overall result of these “fail-first underwater drugs” is delayed and more costly care for the patient and the “system,” particularly self-insured employers.
What Is Being Done to Correct This?
Since ASPs are updated quarterly, it is possible that acquisition costs and reimbursements might stabilize over time, making the drugs affordable again to practices. However, that does not appear to be happening in the near future, so that possibility does not offer immediate relief to struggling practices. It doesn’t promise a favorable outlook for future biosimilar entries of provider-administered medications if formularies continue to prefer the highest-rebated medication.
This dynamic between ASP and acquisition cost does not happen on the pharmacy side because the price concessions on specific drug rebates and fees are proprietary. There appears to be no equivalent to a publicly known ASP on the pharmacy side, which has led to myriad pricing definitions and manipulation on the pharmacy benefit side of medications. In any event, the savings from rebates and other manufacturer price concessions on pharmacy drugs do not influence ASPs of medical benefit drugs.
The Inflation Reduction Act provided a temporary increase in the add-on payment for biosimilars from ASP+6% to ASP+8%, but as long as the biosimilar’s ASP is lower than the reference brand’s ASP, that temporary increase does not appear to make up for the large differential between ASP and acquisition cost. It should be noted that any federal attempt to artificially lower the ASP of a provider-administered drug without a pathway assuring that the acquisition cost for the provider is less than the reimbursement is going to result in loss of access for patients to those medications and/or higher hospital site of care costs.
A Few Partial Fixes, But Most Complaints Go Ignored
Considering the higher costs of hospital-based infusion, insurers should be motivated to keep patients within private practices. Perhaps through insurers’ recognition of that fact, some practices have successfully negotiated exceptions for specific patients by discussing this situation with insurers. From the feedback that CSRO has received from rheumatology practices, it appears that most insurers have been ignoring the complaints from physicians. The few who have responded have resulted in only partial fixes, with some of the biosimilars still left underwater.
Ultimate Solution?
This issue is a direct result of the “rebate game,” whereby price concessions from drug manufacturers drive formulary placement. For provider-administered medications, this results in an artificially lowered ASP, not as a consequence of free-market incentives that benefit the patient, but as a result of misaligned incentives created by Safe Harbor–protected “kickbacks,” distorting the free market and paradoxically reducing access to these medications, delaying care, and increasing prices for patients and the healthcare system.
While federal and state governments are not likely to address this particular situation in the biosimilars market, CSRO is highlighting this issue as a prime example of why the current formulary construction system urgently requires federal reform. At this time, the biosimilars most affected are Inflectra and Avsola, but if nothing changes, more and more biosimilars will fall victim to the short-sighted pricing strategy of aggressive rebating to gain formulary position, with physician purchasers and patients left to navigate the aftermath. The existing system, which necessitates drug companies purchasing formulary access from pharmacy benefit managers, has led to delayed and even denied patient access to certain provider-administered drugs. Moreover, it now appears to be hindering the adoption of biosimilars.
To address this, a multifaceted approach is required. It not only involves reevaluating the rebate system and its impact on formulary construction and ASP, but also ensuring that acquisition costs for providers are aligned with reimbursement rates. Insurers must recognize the economic and clinical value of maintaining infusions within private practices and immediately update their policies to ensure that physician in-office infusion is financially feasible for these “fail-first” biosimilars.
Ultimately, the goal should be to create a sustainable model that promotes the use of affordable biosimilars, enhances patient access to affordable care, and supports the financial viability of medical practices. Concerted efforts to reform the current formulary construction system are required to achieve a healthcare environment that is both cost effective and patient centric.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s vice president of advocacy and government affairs and its immediate past president, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. You can reach her at [email protected].
Editor’s note: This article is adapted from an explanatory statement that Dr. Feldman wrote for the Coalition of State Rheumatology Organizations (CSRO).
According to the Guinness Book of World records, the longest time someone has held their breath underwater voluntarily is 24 minutes and 37.36 seconds. While certainly an amazing feat, UnitedHealthcare, many of the Blues, and other national “payers” are expecting rheumatologists and other specialists to live “underwater” in order to take care of their patients. In other words, these insurance companies are mandating that specialists use certain provider-administered biosimilars whose acquisition cost is higher than what the insurance company is willing to reimburse them. Essentially, the insurance companies expect the rheumatologists to pay them to take care of their patients. Because of the substantial and destabilizing financial losses incurred, many practices and free-standing infusion centers have been forced to cease offering these biosimilars. Most rheumatologists will provide patients with appropriate alternatives when available and permitted by the insurer; otherwise, they must refer patients to hospital-based infusion centers. That results in delayed care and increased costs for patients and the system, because hospital-based infusion typically costs more than twice what office-based infusion costs.
Quantifying the Problem
To help quantify the magnitude of this issue, the Coalition of State Rheumatology Organizations (CSRO) recently conducted a survey of its membership. A shocking 97% of respondents reported that their practice had been affected by reimbursement rates for some biosimilars being lower than acquisition costs, with 91% of respondents stating that this issue is more pronounced for certain biosimilars than others. Across the board, respondents most frequently identified Inflectra (infliximab-dyyb) and Avsola (infliximab-axxq) as being especially affected: Over 88% and over 85% of respondents identified these two products, respectively, as being underwater. These results support the ongoing anecdotal reports CSRO continues to receive from rheumatology practices.
However, the survey results indicated that this issue is by no means confined to those two biosimilars. Truxima (rituximab-abbs) — a biosimilar for Rituxan — was frequently mentioned as well. Notably, respondents almost uniformly identified biosimilars in the infliximab and rituximab families, which illustrates that this issue is no longer confined to one or two early-to-market biosimilars but has almost become a hallmark of this particular biosimilars market. Remarkably, one respondent commented that the brand products are now cheaper to acquire than the biosimilars. Furthermore, the survey included respondents from across the country, indicating that this issue is not confined to a particular region.
How Did This Happen?
Biosimilars held promise for increasing availability and decreasing biologic costs for patients but, thus far, no patients have seen their cost go down. It appears that the only biosimilars that have made it to “preferred” status on the formulary are the ones that have made more money for the middlemen in the drug supply chain, particularly those that construct formularies. Now, we have provider-administered biosimilars whose acquisition cost exceeds the reimbursement for these drugs. This disparity was ultimately created by biosimilar manufacturers “over-rebating” their drugs to health insurance companies to gain “fail-first” status on the formulary.
For example, the manufacturer of Inflectra offered substantial rebates to health insurers for preferred formulary placement. These rebates are factored into the sales price of the medication, which then results in a rapidly declining average sales price (ASP) for the biosimilar. Unfortunately, the acquisition cost for the drug does not experience commensurate reductions, resulting in physicians being reimbursed far less for the drug than it costs to acquire. The financial losses for physicians put them underwater as a result of the acquisition costs for the preferred drugs far surpassing the reimbursement from the health insurance company that constructed the formulary.
While various factors affect ASPs and acquisition costs, this particular consequence of formulary placement based on price concessions is a major driver of the underwater situation in which physicians have found themselves with many biosimilars. Not only does that lead to a lower uptake of biosimilars, but it also results in patients being referred to the hospital outpatient infusion sites to receive this care, as freestanding infusion centers cannot treat these patients either. Hospitals incur higher costs because of facility fees and elevated rates, and this makes private rheumatology in-office infusion centers a much lower-cost option. Similarly, home infusion services, while convenient, are marginally more expensive than private practices and, in cases of biologic infusions, it is important to note that physicians’ offices have a greater safety profile than home infusion of biologics. The overall result of these “fail-first underwater drugs” is delayed and more costly care for the patient and the “system,” particularly self-insured employers.
What Is Being Done to Correct This?
Since ASPs are updated quarterly, it is possible that acquisition costs and reimbursements might stabilize over time, making the drugs affordable again to practices. However, that does not appear to be happening in the near future, so that possibility does not offer immediate relief to struggling practices. It doesn’t promise a favorable outlook for future biosimilar entries of provider-administered medications if formularies continue to prefer the highest-rebated medication.
This dynamic between ASP and acquisition cost does not happen on the pharmacy side because the price concessions on specific drug rebates and fees are proprietary. There appears to be no equivalent to a publicly known ASP on the pharmacy side, which has led to myriad pricing definitions and manipulation on the pharmacy benefit side of medications. In any event, the savings from rebates and other manufacturer price concessions on pharmacy drugs do not influence ASPs of medical benefit drugs.
The Inflation Reduction Act provided a temporary increase in the add-on payment for biosimilars from ASP+6% to ASP+8%, but as long as the biosimilar’s ASP is lower than the reference brand’s ASP, that temporary increase does not appear to make up for the large differential between ASP and acquisition cost. It should be noted that any federal attempt to artificially lower the ASP of a provider-administered drug without a pathway assuring that the acquisition cost for the provider is less than the reimbursement is going to result in loss of access for patients to those medications and/or higher hospital site of care costs.
A Few Partial Fixes, But Most Complaints Go Ignored
Considering the higher costs of hospital-based infusion, insurers should be motivated to keep patients within private practices. Perhaps through insurers’ recognition of that fact, some practices have successfully negotiated exceptions for specific patients by discussing this situation with insurers. From the feedback that CSRO has received from rheumatology practices, it appears that most insurers have been ignoring the complaints from physicians. The few who have responded have resulted in only partial fixes, with some of the biosimilars still left underwater.
Ultimate Solution?
This issue is a direct result of the “rebate game,” whereby price concessions from drug manufacturers drive formulary placement. For provider-administered medications, this results in an artificially lowered ASP, not as a consequence of free-market incentives that benefit the patient, but as a result of misaligned incentives created by Safe Harbor–protected “kickbacks,” distorting the free market and paradoxically reducing access to these medications, delaying care, and increasing prices for patients and the healthcare system.
While federal and state governments are not likely to address this particular situation in the biosimilars market, CSRO is highlighting this issue as a prime example of why the current formulary construction system urgently requires federal reform. At this time, the biosimilars most affected are Inflectra and Avsola, but if nothing changes, more and more biosimilars will fall victim to the short-sighted pricing strategy of aggressive rebating to gain formulary position, with physician purchasers and patients left to navigate the aftermath. The existing system, which necessitates drug companies purchasing formulary access from pharmacy benefit managers, has led to delayed and even denied patient access to certain provider-administered drugs. Moreover, it now appears to be hindering the adoption of biosimilars.
To address this, a multifaceted approach is required. It not only involves reevaluating the rebate system and its impact on formulary construction and ASP, but also ensuring that acquisition costs for providers are aligned with reimbursement rates. Insurers must recognize the economic and clinical value of maintaining infusions within private practices and immediately update their policies to ensure that physician in-office infusion is financially feasible for these “fail-first” biosimilars.
Ultimately, the goal should be to create a sustainable model that promotes the use of affordable biosimilars, enhances patient access to affordable care, and supports the financial viability of medical practices. Concerted efforts to reform the current formulary construction system are required to achieve a healthcare environment that is both cost effective and patient centric.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s vice president of advocacy and government affairs and its immediate past president, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. You can reach her at [email protected].
Editor’s note: This article is adapted from an explanatory statement that Dr. Feldman wrote for the Coalition of State Rheumatology Organizations (CSRO).
According to the Guinness Book of World records, the longest time someone has held their breath underwater voluntarily is 24 minutes and 37.36 seconds. While certainly an amazing feat, UnitedHealthcare, many of the Blues, and other national “payers” are expecting rheumatologists and other specialists to live “underwater” in order to take care of their patients. In other words, these insurance companies are mandating that specialists use certain provider-administered biosimilars whose acquisition cost is higher than what the insurance company is willing to reimburse them. Essentially, the insurance companies expect the rheumatologists to pay them to take care of their patients. Because of the substantial and destabilizing financial losses incurred, many practices and free-standing infusion centers have been forced to cease offering these biosimilars. Most rheumatologists will provide patients with appropriate alternatives when available and permitted by the insurer; otherwise, they must refer patients to hospital-based infusion centers. That results in delayed care and increased costs for patients and the system, because hospital-based infusion typically costs more than twice what office-based infusion costs.
Quantifying the Problem
To help quantify the magnitude of this issue, the Coalition of State Rheumatology Organizations (CSRO) recently conducted a survey of its membership. A shocking 97% of respondents reported that their practice had been affected by reimbursement rates for some biosimilars being lower than acquisition costs, with 91% of respondents stating that this issue is more pronounced for certain biosimilars than others. Across the board, respondents most frequently identified Inflectra (infliximab-dyyb) and Avsola (infliximab-axxq) as being especially affected: Over 88% and over 85% of respondents identified these two products, respectively, as being underwater. These results support the ongoing anecdotal reports CSRO continues to receive from rheumatology practices.
However, the survey results indicated that this issue is by no means confined to those two biosimilars. Truxima (rituximab-abbs) — a biosimilar for Rituxan — was frequently mentioned as well. Notably, respondents almost uniformly identified biosimilars in the infliximab and rituximab families, which illustrates that this issue is no longer confined to one or two early-to-market biosimilars but has almost become a hallmark of this particular biosimilars market. Remarkably, one respondent commented that the brand products are now cheaper to acquire than the biosimilars. Furthermore, the survey included respondents from across the country, indicating that this issue is not confined to a particular region.
How Did This Happen?
Biosimilars held promise for increasing availability and decreasing biologic costs for patients but, thus far, no patients have seen their cost go down. It appears that the only biosimilars that have made it to “preferred” status on the formulary are the ones that have made more money for the middlemen in the drug supply chain, particularly those that construct formularies. Now, we have provider-administered biosimilars whose acquisition cost exceeds the reimbursement for these drugs. This disparity was ultimately created by biosimilar manufacturers “over-rebating” their drugs to health insurance companies to gain “fail-first” status on the formulary.
For example, the manufacturer of Inflectra offered substantial rebates to health insurers for preferred formulary placement. These rebates are factored into the sales price of the medication, which then results in a rapidly declining average sales price (ASP) for the biosimilar. Unfortunately, the acquisition cost for the drug does not experience commensurate reductions, resulting in physicians being reimbursed far less for the drug than it costs to acquire. The financial losses for physicians put them underwater as a result of the acquisition costs for the preferred drugs far surpassing the reimbursement from the health insurance company that constructed the formulary.
While various factors affect ASPs and acquisition costs, this particular consequence of formulary placement based on price concessions is a major driver of the underwater situation in which physicians have found themselves with many biosimilars. Not only does that lead to a lower uptake of biosimilars, but it also results in patients being referred to the hospital outpatient infusion sites to receive this care, as freestanding infusion centers cannot treat these patients either. Hospitals incur higher costs because of facility fees and elevated rates, and this makes private rheumatology in-office infusion centers a much lower-cost option. Similarly, home infusion services, while convenient, are marginally more expensive than private practices and, in cases of biologic infusions, it is important to note that physicians’ offices have a greater safety profile than home infusion of biologics. The overall result of these “fail-first underwater drugs” is delayed and more costly care for the patient and the “system,” particularly self-insured employers.
What Is Being Done to Correct This?
Since ASPs are updated quarterly, it is possible that acquisition costs and reimbursements might stabilize over time, making the drugs affordable again to practices. However, that does not appear to be happening in the near future, so that possibility does not offer immediate relief to struggling practices. It doesn’t promise a favorable outlook for future biosimilar entries of provider-administered medications if formularies continue to prefer the highest-rebated medication.
This dynamic between ASP and acquisition cost does not happen on the pharmacy side because the price concessions on specific drug rebates and fees are proprietary. There appears to be no equivalent to a publicly known ASP on the pharmacy side, which has led to myriad pricing definitions and manipulation on the pharmacy benefit side of medications. In any event, the savings from rebates and other manufacturer price concessions on pharmacy drugs do not influence ASPs of medical benefit drugs.
The Inflation Reduction Act provided a temporary increase in the add-on payment for biosimilars from ASP+6% to ASP+8%, but as long as the biosimilar’s ASP is lower than the reference brand’s ASP, that temporary increase does not appear to make up for the large differential between ASP and acquisition cost. It should be noted that any federal attempt to artificially lower the ASP of a provider-administered drug without a pathway assuring that the acquisition cost for the provider is less than the reimbursement is going to result in loss of access for patients to those medications and/or higher hospital site of care costs.
A Few Partial Fixes, But Most Complaints Go Ignored
Considering the higher costs of hospital-based infusion, insurers should be motivated to keep patients within private practices. Perhaps through insurers’ recognition of that fact, some practices have successfully negotiated exceptions for specific patients by discussing this situation with insurers. From the feedback that CSRO has received from rheumatology practices, it appears that most insurers have been ignoring the complaints from physicians. The few who have responded have resulted in only partial fixes, with some of the biosimilars still left underwater.
Ultimate Solution?
This issue is a direct result of the “rebate game,” whereby price concessions from drug manufacturers drive formulary placement. For provider-administered medications, this results in an artificially lowered ASP, not as a consequence of free-market incentives that benefit the patient, but as a result of misaligned incentives created by Safe Harbor–protected “kickbacks,” distorting the free market and paradoxically reducing access to these medications, delaying care, and increasing prices for patients and the healthcare system.
While federal and state governments are not likely to address this particular situation in the biosimilars market, CSRO is highlighting this issue as a prime example of why the current formulary construction system urgently requires federal reform. At this time, the biosimilars most affected are Inflectra and Avsola, but if nothing changes, more and more biosimilars will fall victim to the short-sighted pricing strategy of aggressive rebating to gain formulary position, with physician purchasers and patients left to navigate the aftermath. The existing system, which necessitates drug companies purchasing formulary access from pharmacy benefit managers, has led to delayed and even denied patient access to certain provider-administered drugs. Moreover, it now appears to be hindering the adoption of biosimilars.
To address this, a multifaceted approach is required. It not only involves reevaluating the rebate system and its impact on formulary construction and ASP, but also ensuring that acquisition costs for providers are aligned with reimbursement rates. Insurers must recognize the economic and clinical value of maintaining infusions within private practices and immediately update their policies to ensure that physician in-office infusion is financially feasible for these “fail-first” biosimilars.
Ultimately, the goal should be to create a sustainable model that promotes the use of affordable biosimilars, enhances patient access to affordable care, and supports the financial viability of medical practices. Concerted efforts to reform the current formulary construction system are required to achieve a healthcare environment that is both cost effective and patient centric.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s vice president of advocacy and government affairs and its immediate past president, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. You can reach her at [email protected].
Cancer Identified Via Serum Metabolites, Lipids in Rheumatic Disease or Paraneoplasia
TOPLINE:
A diagnostic model based on the concentrations of four metabolites and one lipid ratio can reliably predict cancer in patients with rheumatic and musculoskeletal diseases (RMDs) or paraneoplasia, providing high sensitivity and specificity.
METHODOLOGY:
- The metabolome profile can differentiate between nonspecific inflammatory symptoms such as those associated with paraneoplastic conditions or RMDs, which can help accelerate cancer diagnosis and treatment.
- To assess if changes in the serum metabolome profile could indicate cancer in patients with RMD, researchers performed nuclear magnetic resonance analysis of the sera of patients with rheumatoid arthritis (RA) with a history of invasive cancer (n = 56; age, 69.9 years; 76.8% women) or without such history (n = 52; age, 56.1 years; 57.7% women).
- Blinded validation was conducted in a cohort of patients with RA or spondyloarthritis with or without a history of invasive cancer.
- Additionally, the model performance was tested in a cohort of patients having RA or spondyloarthritis with active cancer or cancer treatment, pulmonary and lymphoid type cancers, paraneoplastic syndromes, and facultative solid noninvasive precancerous lesions and nonmelanoma skin cancer; in samples prior to the development of malignancy; and in a cohort of patients with systemic lupus erythematosus (SLE).
- The final model comprised five variables. The goodness of fit of the model was described using the area under the receiver operating characteristic curve (AUC).
TAKEAWAY:
- Based on the concentrations of acetate, creatine, glycine, and formate and the L1/L6 lipid ratio, the diagnostic model yielded an excellent AUC (0.987) and high sensitivity (0.932) and specificity (0.946) for cancer diagnosis in patients with RA.
- The diagnostic model yielded an AUC of 0.937 in the blinded validation cohort of patients with RA and an AUC of 0.927 in the merged RA and spondyloarthritis cohort.
- Although the diagnostic model accurately diagnosed cancer in all the patients with paraneoplasia, it could do so accurately in only 50% of patients with noninvasive or in situ precancerous lesions and nonmelanoma skin cancers.
- The performance of the model was poor in the SLE cohort (AUC, 0.656), and it could not identify patients at risk for later invasive cancer development.
IN PRACTICE:
“This limited-invasive assay has considerable potential of high clinical value to facilitate timely diagnosis of cancer in paraneoplastic rheumatic syndromes as well as become a valuable active surveillance tool in RA and SpA [spondyloarthritis] patients with a high risk of developing cancer,” the authors wrote.
SOURCE:
The study, led by Karolina Gente, MHBA, Heidelberg University Hospital, Heidelberg, Germany, was published online on April 1, 2024, in Annals of the Rheumatic Diseases.
LIMITATIONS:
The limited invasiveness during sampling might account for the model’s inability to identify three early-stage, low-grade tumors and its nonreliability in identifying noninvasive facultative precancerous lesions and nonmelanoma skin cancers. Given its poor performance in the SLE cohort, the model may not be suitable for universal application in more systemic rheumatic diseases.
DISCLOSURES:
This study was supported by an unrestricted investigator-initiated grant from the Foundation Commission of the Medical Faculty, University of Heidelberg, Germany. The authors declared no conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
A diagnostic model based on the concentrations of four metabolites and one lipid ratio can reliably predict cancer in patients with rheumatic and musculoskeletal diseases (RMDs) or paraneoplasia, providing high sensitivity and specificity.
METHODOLOGY:
- The metabolome profile can differentiate between nonspecific inflammatory symptoms such as those associated with paraneoplastic conditions or RMDs, which can help accelerate cancer diagnosis and treatment.
- To assess if changes in the serum metabolome profile could indicate cancer in patients with RMD, researchers performed nuclear magnetic resonance analysis of the sera of patients with rheumatoid arthritis (RA) with a history of invasive cancer (n = 56; age, 69.9 years; 76.8% women) or without such history (n = 52; age, 56.1 years; 57.7% women).
- Blinded validation was conducted in a cohort of patients with RA or spondyloarthritis with or without a history of invasive cancer.
- Additionally, the model performance was tested in a cohort of patients having RA or spondyloarthritis with active cancer or cancer treatment, pulmonary and lymphoid type cancers, paraneoplastic syndromes, and facultative solid noninvasive precancerous lesions and nonmelanoma skin cancer; in samples prior to the development of malignancy; and in a cohort of patients with systemic lupus erythematosus (SLE).
- The final model comprised five variables. The goodness of fit of the model was described using the area under the receiver operating characteristic curve (AUC).
TAKEAWAY:
- Based on the concentrations of acetate, creatine, glycine, and formate and the L1/L6 lipid ratio, the diagnostic model yielded an excellent AUC (0.987) and high sensitivity (0.932) and specificity (0.946) for cancer diagnosis in patients with RA.
- The diagnostic model yielded an AUC of 0.937 in the blinded validation cohort of patients with RA and an AUC of 0.927 in the merged RA and spondyloarthritis cohort.
- Although the diagnostic model accurately diagnosed cancer in all the patients with paraneoplasia, it could do so accurately in only 50% of patients with noninvasive or in situ precancerous lesions and nonmelanoma skin cancers.
- The performance of the model was poor in the SLE cohort (AUC, 0.656), and it could not identify patients at risk for later invasive cancer development.
IN PRACTICE:
“This limited-invasive assay has considerable potential of high clinical value to facilitate timely diagnosis of cancer in paraneoplastic rheumatic syndromes as well as become a valuable active surveillance tool in RA and SpA [spondyloarthritis] patients with a high risk of developing cancer,” the authors wrote.
SOURCE:
The study, led by Karolina Gente, MHBA, Heidelberg University Hospital, Heidelberg, Germany, was published online on April 1, 2024, in Annals of the Rheumatic Diseases.
LIMITATIONS:
The limited invasiveness during sampling might account for the model’s inability to identify three early-stage, low-grade tumors and its nonreliability in identifying noninvasive facultative precancerous lesions and nonmelanoma skin cancers. Given its poor performance in the SLE cohort, the model may not be suitable for universal application in more systemic rheumatic diseases.
DISCLOSURES:
This study was supported by an unrestricted investigator-initiated grant from the Foundation Commission of the Medical Faculty, University of Heidelberg, Germany. The authors declared no conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
A diagnostic model based on the concentrations of four metabolites and one lipid ratio can reliably predict cancer in patients with rheumatic and musculoskeletal diseases (RMDs) or paraneoplasia, providing high sensitivity and specificity.
METHODOLOGY:
- The metabolome profile can differentiate between nonspecific inflammatory symptoms such as those associated with paraneoplastic conditions or RMDs, which can help accelerate cancer diagnosis and treatment.
- To assess if changes in the serum metabolome profile could indicate cancer in patients with RMD, researchers performed nuclear magnetic resonance analysis of the sera of patients with rheumatoid arthritis (RA) with a history of invasive cancer (n = 56; age, 69.9 years; 76.8% women) or without such history (n = 52; age, 56.1 years; 57.7% women).
- Blinded validation was conducted in a cohort of patients with RA or spondyloarthritis with or without a history of invasive cancer.
- Additionally, the model performance was tested in a cohort of patients having RA or spondyloarthritis with active cancer or cancer treatment, pulmonary and lymphoid type cancers, paraneoplastic syndromes, and facultative solid noninvasive precancerous lesions and nonmelanoma skin cancer; in samples prior to the development of malignancy; and in a cohort of patients with systemic lupus erythematosus (SLE).
- The final model comprised five variables. The goodness of fit of the model was described using the area under the receiver operating characteristic curve (AUC).
TAKEAWAY:
- Based on the concentrations of acetate, creatine, glycine, and formate and the L1/L6 lipid ratio, the diagnostic model yielded an excellent AUC (0.987) and high sensitivity (0.932) and specificity (0.946) for cancer diagnosis in patients with RA.
- The diagnostic model yielded an AUC of 0.937 in the blinded validation cohort of patients with RA and an AUC of 0.927 in the merged RA and spondyloarthritis cohort.
- Although the diagnostic model accurately diagnosed cancer in all the patients with paraneoplasia, it could do so accurately in only 50% of patients with noninvasive or in situ precancerous lesions and nonmelanoma skin cancers.
- The performance of the model was poor in the SLE cohort (AUC, 0.656), and it could not identify patients at risk for later invasive cancer development.
IN PRACTICE:
“This limited-invasive assay has considerable potential of high clinical value to facilitate timely diagnosis of cancer in paraneoplastic rheumatic syndromes as well as become a valuable active surveillance tool in RA and SpA [spondyloarthritis] patients with a high risk of developing cancer,” the authors wrote.
SOURCE:
The study, led by Karolina Gente, MHBA, Heidelberg University Hospital, Heidelberg, Germany, was published online on April 1, 2024, in Annals of the Rheumatic Diseases.
LIMITATIONS:
The limited invasiveness during sampling might account for the model’s inability to identify three early-stage, low-grade tumors and its nonreliability in identifying noninvasive facultative precancerous lesions and nonmelanoma skin cancers. Given its poor performance in the SLE cohort, the model may not be suitable for universal application in more systemic rheumatic diseases.
DISCLOSURES:
This study was supported by an unrestricted investigator-initiated grant from the Foundation Commission of the Medical Faculty, University of Heidelberg, Germany. The authors declared no conflicts of interest.
A version of this article appeared on Medscape.com.
bDMARDs Preserve Renal Function in Most Patients With AA Amyloidosis
TOPLINE:
Treatment with biologic disease-modifying antirheumatic drugs (bDMARDs), particularly tocilizumab, can suppress inflammation and preserve renal function in a majority of patients with chronic inflammatory disorders who develop serum amyloid alpha (SAA) amyloidosis.
METHODOLOGY:
- AA amyloidosis, characterized by the misfolding of the SAA protein, is observed in patients with inflammatory diseases and can lead to progressive organ damage, including chronic kidney disease, malabsorption with cachexia, and cardiac failure.
- This monocentric, retrospective analysis assessed the effect of bDMARD therapy on inflammatory biomarker levels and renal outcomes in 83 patients with AA amyloidosis who were followed for a mean period of 4.82 years.
- The patients were stratified into three major subgroups depending on the cause of AA amyloidosis:
- Chronic inflammatory diseases (cid + AA; n = 34) such as rheumatoid arthritis, Crohn’s disease, and chronic infections
- Autoinflammatory syndromes (auto + AA; n = 24) such as familial Mediterranean fever (FMF) and cryopyrin-associated periodic syndrome (CAPS)
- Idiopathic AA (idio + AA; n = 25), wherein the primary disease could not be identified
- Tocilizumab was the most commonly used bDMARD in patients with cid + AA and idio + AA amyloidosis, and interleukin-1 inhibitors were prescribed to patients with auto + AA amyloidosis because tocilizumab has not been approved yet for FMF or CAPS treatment.
- All patients with AA amyloidosis had renal involvement, as confirmed by kidney biopsy.
TAKEAWAY:
- After bDMARD therapy, C-reactive protein levels reduced significantly from baseline to the last-documented visit in all subgroups, while SAA levels declined in the subgroups cid + AA and idio + AA and proteinuria dropped in the subgroups auto + AA and idio + AA.
- bDMARDs prevented progression to end-stage renal disease (ESRD) in 75% of the patients in the overall cohort, with progression to ESRD being prevented in 60% of patients with cid + AA, 88% of patients with auto + AA, and 81% of patients with idio + AA.
- Tocilizumab was more effective than other bDMARDs in preventing renal progression to ESRD (P = .0006), with a similar pattern observed for the subgroups cid + AA (P = .0126) and idio + AA (P = .0259).
- None of the patients receiving tocilizumab died during the nearly 5-year follow-up period.
IN PRACTICE:
“The data suggest preferential use of IL [interleukin]-1 inhibitors and tocilizumab for clinical use in the treatment of AA amyloidosis depending on the respective underlying diseases,” the authors wrote.
SOURCE:
This study, led by Peter Kvacskay, MD, of Heidelberg University Hospital, Heidelberg, Germany, was published online on April 23 in Annals of the Rheumatic Diseases.
LIMITATIONS:
Authors acknowledged the retrospective nature of the analysis and missing data of single patients during the long-term follow-up as major limitations. Furthermore, the cid + AA subgroup was heterogeneous in terms of the pathophysiology of their underlying primary disease.
DISCLOSURES:
This study did not report any source of funding. The authors declared no conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
Treatment with biologic disease-modifying antirheumatic drugs (bDMARDs), particularly tocilizumab, can suppress inflammation and preserve renal function in a majority of patients with chronic inflammatory disorders who develop serum amyloid alpha (SAA) amyloidosis.
METHODOLOGY:
- AA amyloidosis, characterized by the misfolding of the SAA protein, is observed in patients with inflammatory diseases and can lead to progressive organ damage, including chronic kidney disease, malabsorption with cachexia, and cardiac failure.
- This monocentric, retrospective analysis assessed the effect of bDMARD therapy on inflammatory biomarker levels and renal outcomes in 83 patients with AA amyloidosis who were followed for a mean period of 4.82 years.
- The patients were stratified into three major subgroups depending on the cause of AA amyloidosis:
- Chronic inflammatory diseases (cid + AA; n = 34) such as rheumatoid arthritis, Crohn’s disease, and chronic infections
- Autoinflammatory syndromes (auto + AA; n = 24) such as familial Mediterranean fever (FMF) and cryopyrin-associated periodic syndrome (CAPS)
- Idiopathic AA (idio + AA; n = 25), wherein the primary disease could not be identified
- Tocilizumab was the most commonly used bDMARD in patients with cid + AA and idio + AA amyloidosis, and interleukin-1 inhibitors were prescribed to patients with auto + AA amyloidosis because tocilizumab has not been approved yet for FMF or CAPS treatment.
- All patients with AA amyloidosis had renal involvement, as confirmed by kidney biopsy.
TAKEAWAY:
- After bDMARD therapy, C-reactive protein levels reduced significantly from baseline to the last-documented visit in all subgroups, while SAA levels declined in the subgroups cid + AA and idio + AA and proteinuria dropped in the subgroups auto + AA and idio + AA.
- bDMARDs prevented progression to end-stage renal disease (ESRD) in 75% of the patients in the overall cohort, with progression to ESRD being prevented in 60% of patients with cid + AA, 88% of patients with auto + AA, and 81% of patients with idio + AA.
- Tocilizumab was more effective than other bDMARDs in preventing renal progression to ESRD (P = .0006), with a similar pattern observed for the subgroups cid + AA (P = .0126) and idio + AA (P = .0259).
- None of the patients receiving tocilizumab died during the nearly 5-year follow-up period.
IN PRACTICE:
“The data suggest preferential use of IL [interleukin]-1 inhibitors and tocilizumab for clinical use in the treatment of AA amyloidosis depending on the respective underlying diseases,” the authors wrote.
SOURCE:
This study, led by Peter Kvacskay, MD, of Heidelberg University Hospital, Heidelberg, Germany, was published online on April 23 in Annals of the Rheumatic Diseases.
LIMITATIONS:
Authors acknowledged the retrospective nature of the analysis and missing data of single patients during the long-term follow-up as major limitations. Furthermore, the cid + AA subgroup was heterogeneous in terms of the pathophysiology of their underlying primary disease.
DISCLOSURES:
This study did not report any source of funding. The authors declared no conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
Treatment with biologic disease-modifying antirheumatic drugs (bDMARDs), particularly tocilizumab, can suppress inflammation and preserve renal function in a majority of patients with chronic inflammatory disorders who develop serum amyloid alpha (SAA) amyloidosis.
METHODOLOGY:
- AA amyloidosis, characterized by the misfolding of the SAA protein, is observed in patients with inflammatory diseases and can lead to progressive organ damage, including chronic kidney disease, malabsorption with cachexia, and cardiac failure.
- This monocentric, retrospective analysis assessed the effect of bDMARD therapy on inflammatory biomarker levels and renal outcomes in 83 patients with AA amyloidosis who were followed for a mean period of 4.82 years.
- The patients were stratified into three major subgroups depending on the cause of AA amyloidosis:
- Chronic inflammatory diseases (cid + AA; n = 34) such as rheumatoid arthritis, Crohn’s disease, and chronic infections
- Autoinflammatory syndromes (auto + AA; n = 24) such as familial Mediterranean fever (FMF) and cryopyrin-associated periodic syndrome (CAPS)
- Idiopathic AA (idio + AA; n = 25), wherein the primary disease could not be identified
- Tocilizumab was the most commonly used bDMARD in patients with cid + AA and idio + AA amyloidosis, and interleukin-1 inhibitors were prescribed to patients with auto + AA amyloidosis because tocilizumab has not been approved yet for FMF or CAPS treatment.
- All patients with AA amyloidosis had renal involvement, as confirmed by kidney biopsy.
TAKEAWAY:
- After bDMARD therapy, C-reactive protein levels reduced significantly from baseline to the last-documented visit in all subgroups, while SAA levels declined in the subgroups cid + AA and idio + AA and proteinuria dropped in the subgroups auto + AA and idio + AA.
- bDMARDs prevented progression to end-stage renal disease (ESRD) in 75% of the patients in the overall cohort, with progression to ESRD being prevented in 60% of patients with cid + AA, 88% of patients with auto + AA, and 81% of patients with idio + AA.
- Tocilizumab was more effective than other bDMARDs in preventing renal progression to ESRD (P = .0006), with a similar pattern observed for the subgroups cid + AA (P = .0126) and idio + AA (P = .0259).
- None of the patients receiving tocilizumab died during the nearly 5-year follow-up period.
IN PRACTICE:
“The data suggest preferential use of IL [interleukin]-1 inhibitors and tocilizumab for clinical use in the treatment of AA amyloidosis depending on the respective underlying diseases,” the authors wrote.
SOURCE:
This study, led by Peter Kvacskay, MD, of Heidelberg University Hospital, Heidelberg, Germany, was published online on April 23 in Annals of the Rheumatic Diseases.
LIMITATIONS:
Authors acknowledged the retrospective nature of the analysis and missing data of single patients during the long-term follow-up as major limitations. Furthermore, the cid + AA subgroup was heterogeneous in terms of the pathophysiology of their underlying primary disease.
DISCLOSURES:
This study did not report any source of funding. The authors declared no conflicts of interest.
A version of this article appeared on Medscape.com.
Monoclonal Antibody With Unique Mechanism Gets Second Chance in RA
LIVERPOOL, ENGLAND — The IRIS-RA study of the investigational monoclonal antibody drug nipocalimab in patients with rheumatoid arthritis (RA) did not meet its primary endpoint, but there could still be people with moderate to severe RA who might benefit from treatment with the drug, according to information reported at the British Society for Rheumatology annual meeting.
The primary endpoint for the phase 2A trial was the least squares mean change in Disease Activity Score in 28 joints using C-reactive protein (DAS28-CRP) from baseline to 12 weeks of treatment. This was reduced by −1.03 with nipocalimab and by −0.58 with placebo, giving a mean difference of just −0.45 (P = .224).
However, one of the key secondary endpoints was the proportion of patients who had 20% improvement in American College of Rheumatology response criteria (ACR20). Results for this endpoint showed a greater difference in response to nipocalimab vs placebo, with a respective 45.5% and 20.0% (P = .055) of individuals achieving ACR20.
Moreover, an analysis stratifying for anti-citrullinated protein autoantibody (ACPA) levels at baseline found that people with higher levels had a better response to nipocalimab.
Choice of Endpoint
“The way this study was powered was to look at a change between the treatment groups of a DAS28-CRP reduction of 1.0,” said Peter C. Taylor, BMBCh, PhD, the Norman Collisson chair of musculoskeletal medicine at the University of Oxford in Oxford, England.
DAS28-CRP was often chosen as the primary endpoint in small proof-of-concept studies, such as IRIS-RA, because it was a “measure of continuous change [that] theoretically, would allow greater sensitivity to change,” Dr. Taylor added.
“Ironically, it has to be said that had we chosen ACR20, we would have hit the primary endpoint. One lives and learns,” noted Dr. Taylor.
Proof of Concept
IRIS-RA was billed as a “proof-of-concept” study because it was the first time that a monoclonal antibody targeting the neonatal fragment crystallizable receptor (FcRn) was being tested in an RA population.
The study was a randomized double-blind trial in which 33 people with moderate to severe RA who had an inadequate response to tumor necrosis factor (TNF) inhibitors were treated with nipocalimab at a dose of 15 mg/kg given intravenously every 2 weeks, and 20 received a matching placebo. Participants were treated for 10 weeks, and then the primary follow-up was at 12 weeks, with additional follow-up for safety undertaken at 18 weeks.
Nipocalimab is a fully human, immunoglobulin G1 (IgG1) monoclonal antibody that is designed to selectively block the FcRn. By doing so, it essentially stops IgG from being recycled within the immune system, and this in turn lowers IgG levels. That includes potentially harmful ACPAs, among other pathogenic antibodies, Dr. Taylor and fellow investigators explained in their abstract.
“We’ve known for a long time that ACPA have prognostic value, but there’s been controversy about whether or not ACPA are actually pathogenic,” Dr. Taylor said. “So, one of the hypotheses that this study gives rise to is that by blocking FcRn, and thereby reducing, potentially, the concentration of ACPA in the blood, will we actually have efficacy in patients?”
Are ACPA Really Lowered?
Paul Emery, MD, Versus Arthritis professor of rheumatology and director of the Leeds Biomedical Research Centre at the University of Leeds in Leeds, England, questioned the reduction in antibody levels during the discussion that followed.
Although these data had not been presented, Dr. Emery observed that the reduction in IgG was actually greater than that in ACPA, “which is fairly critical. Is it feasible to look to selectively lower normal immunoglobulin over pathogenic autoantibodies?”
Dr. Emery also wanted to know if there “was a floor on the reduction of immunoglobulin” with long-term therapy, “which would be a worry.”
Dr. Taylor responded that total IgG had been reduced by about 65% and ACPA by about 40%. Why this difference exists is not yet clear. It could be because ACPA are part of complexed antibodies.
“Most of these patients are rheumatoid factor [RF]–positive,” said Taylor, pointing out that although IgM “wouldn’t normally be affected by FcRn blockade,” there was a 10% reduction in RF IgM, probably because it was complexed to IgG.
“So, the hypothesis here is that if you look at the clearance of complexes, they’re handled differently in the cytoplasm from the clearance of monomeric IgG. But that’s a hypothesis. It needs further investigation. In vitro, there’s very good, confirmatory evidence to support that. But we’ve yet to explore that more fully in vivo,” Dr. Taylor said.
As for long-term effects, Dr. Taylor responded: “All I can tell you is [that] after the 10-week intervention, that up to an 18-week observation period, immunoglobulin levels recovered very rapidly afterwards. And you mustn’t forget that other isotypes are not affected, unlike rituximab.”
Safety and Other Results
With regard to safety, 27 (82%) of nipocalimab- and 12 (60%) of placebo-treated participants experienced at least one treatment-emergent adverse event (TEAE). The most common, occurring in 10% or more of cases, were RA flares (36.4% for nipocalimab vs 15.0% with placebo), headache (12.1% vs 5.0%), and COVID-19 (12.1% vs 0.0%).
There were three serious TEAEs, all in the nipocalimab-treatment group: One was an infection of a burn that had been present at inclusion, another was a deep vein thrombosis that resolved with apixaban treatment, and the other was an infusion-related reaction that resolved with supportive treatment.
Another notable efficacy finding was the proportion of patients achieving DAS28-CRP remission at 12 weeks in the nipocalimab vs the placebo group was substantially greater if considering only people with high baseline ACPA levels, at a respective 40.0% vs 16.7%, when compared with the total population (21.2% vs 10.0%).
Similar findings were seen for the proportion of patients achieving an ACR50, and there were numerically greater reductions in the components of the ACR response criteria such as tender and swollen joints with nipocalimab vs placebo. All of these were exploratory observations, Dr. Taylor emphasized.
Combination and Further Trials
Further trials of nipocalimab are planned or are already ongoing in systemic lupus erythematosus, active lupus nephritis, Sjögren disease, and five other diseases.
In RA, nipocalimab is now being tested in combination with the TNF inhibitor certolizumab pegol (Cimzia) in the DAISY-RA trial. This is another proof-of-concept, phase 2A trial with a target accrual of 104 patients.
The IRIS-RA study was funded by Janssen Research & Development. Dr. Taylor serves as a consultant to AbbVie, Biogen, Eli Lilly, Fresenius, Galapagos, Gilead Sciences, GlaxoSmithKline, Janssen, Nordic Pharma, Pfizer, Sanofi, Aqtual, and UCB and received research funding from Galapagos, among others. Dr. Emery received research grants paid to his institution from AbbVie, Bristol Myers Squibb (BMS), Pfizer, MSD, and Roche; received consultant fees from BMS, AbbVie, Pfizer, MSD, Novartis, Roche, and UCB; and has undertaken clinical trials and provided expert advice to Pfizer, MSD, AbbVie, BMS, UCB, Roche, Novartis, Samsung, Sandoz, and Lilly.
A version of this article appeared on Medscape.com.
LIVERPOOL, ENGLAND — The IRIS-RA study of the investigational monoclonal antibody drug nipocalimab in patients with rheumatoid arthritis (RA) did not meet its primary endpoint, but there could still be people with moderate to severe RA who might benefit from treatment with the drug, according to information reported at the British Society for Rheumatology annual meeting.
The primary endpoint for the phase 2A trial was the least squares mean change in Disease Activity Score in 28 joints using C-reactive protein (DAS28-CRP) from baseline to 12 weeks of treatment. This was reduced by −1.03 with nipocalimab and by −0.58 with placebo, giving a mean difference of just −0.45 (P = .224).
However, one of the key secondary endpoints was the proportion of patients who had 20% improvement in American College of Rheumatology response criteria (ACR20). Results for this endpoint showed a greater difference in response to nipocalimab vs placebo, with a respective 45.5% and 20.0% (P = .055) of individuals achieving ACR20.
Moreover, an analysis stratifying for anti-citrullinated protein autoantibody (ACPA) levels at baseline found that people with higher levels had a better response to nipocalimab.
Choice of Endpoint
“The way this study was powered was to look at a change between the treatment groups of a DAS28-CRP reduction of 1.0,” said Peter C. Taylor, BMBCh, PhD, the Norman Collisson chair of musculoskeletal medicine at the University of Oxford in Oxford, England.
DAS28-CRP was often chosen as the primary endpoint in small proof-of-concept studies, such as IRIS-RA, because it was a “measure of continuous change [that] theoretically, would allow greater sensitivity to change,” Dr. Taylor added.
“Ironically, it has to be said that had we chosen ACR20, we would have hit the primary endpoint. One lives and learns,” noted Dr. Taylor.
Proof of Concept
IRIS-RA was billed as a “proof-of-concept” study because it was the first time that a monoclonal antibody targeting the neonatal fragment crystallizable receptor (FcRn) was being tested in an RA population.
The study was a randomized double-blind trial in which 33 people with moderate to severe RA who had an inadequate response to tumor necrosis factor (TNF) inhibitors were treated with nipocalimab at a dose of 15 mg/kg given intravenously every 2 weeks, and 20 received a matching placebo. Participants were treated for 10 weeks, and then the primary follow-up was at 12 weeks, with additional follow-up for safety undertaken at 18 weeks.
Nipocalimab is a fully human, immunoglobulin G1 (IgG1) monoclonal antibody that is designed to selectively block the FcRn. By doing so, it essentially stops IgG from being recycled within the immune system, and this in turn lowers IgG levels. That includes potentially harmful ACPAs, among other pathogenic antibodies, Dr. Taylor and fellow investigators explained in their abstract.
“We’ve known for a long time that ACPA have prognostic value, but there’s been controversy about whether or not ACPA are actually pathogenic,” Dr. Taylor said. “So, one of the hypotheses that this study gives rise to is that by blocking FcRn, and thereby reducing, potentially, the concentration of ACPA in the blood, will we actually have efficacy in patients?”
Are ACPA Really Lowered?
Paul Emery, MD, Versus Arthritis professor of rheumatology and director of the Leeds Biomedical Research Centre at the University of Leeds in Leeds, England, questioned the reduction in antibody levels during the discussion that followed.
Although these data had not been presented, Dr. Emery observed that the reduction in IgG was actually greater than that in ACPA, “which is fairly critical. Is it feasible to look to selectively lower normal immunoglobulin over pathogenic autoantibodies?”
Dr. Emery also wanted to know if there “was a floor on the reduction of immunoglobulin” with long-term therapy, “which would be a worry.”
Dr. Taylor responded that total IgG had been reduced by about 65% and ACPA by about 40%. Why this difference exists is not yet clear. It could be because ACPA are part of complexed antibodies.
“Most of these patients are rheumatoid factor [RF]–positive,” said Taylor, pointing out that although IgM “wouldn’t normally be affected by FcRn blockade,” there was a 10% reduction in RF IgM, probably because it was complexed to IgG.
“So, the hypothesis here is that if you look at the clearance of complexes, they’re handled differently in the cytoplasm from the clearance of monomeric IgG. But that’s a hypothesis. It needs further investigation. In vitro, there’s very good, confirmatory evidence to support that. But we’ve yet to explore that more fully in vivo,” Dr. Taylor said.
As for long-term effects, Dr. Taylor responded: “All I can tell you is [that] after the 10-week intervention, that up to an 18-week observation period, immunoglobulin levels recovered very rapidly afterwards. And you mustn’t forget that other isotypes are not affected, unlike rituximab.”
Safety and Other Results
With regard to safety, 27 (82%) of nipocalimab- and 12 (60%) of placebo-treated participants experienced at least one treatment-emergent adverse event (TEAE). The most common, occurring in 10% or more of cases, were RA flares (36.4% for nipocalimab vs 15.0% with placebo), headache (12.1% vs 5.0%), and COVID-19 (12.1% vs 0.0%).
There were three serious TEAEs, all in the nipocalimab-treatment group: One was an infection of a burn that had been present at inclusion, another was a deep vein thrombosis that resolved with apixaban treatment, and the other was an infusion-related reaction that resolved with supportive treatment.
Another notable efficacy finding was the proportion of patients achieving DAS28-CRP remission at 12 weeks in the nipocalimab vs the placebo group was substantially greater if considering only people with high baseline ACPA levels, at a respective 40.0% vs 16.7%, when compared with the total population (21.2% vs 10.0%).
Similar findings were seen for the proportion of patients achieving an ACR50, and there were numerically greater reductions in the components of the ACR response criteria such as tender and swollen joints with nipocalimab vs placebo. All of these were exploratory observations, Dr. Taylor emphasized.
Combination and Further Trials
Further trials of nipocalimab are planned or are already ongoing in systemic lupus erythematosus, active lupus nephritis, Sjögren disease, and five other diseases.
In RA, nipocalimab is now being tested in combination with the TNF inhibitor certolizumab pegol (Cimzia) in the DAISY-RA trial. This is another proof-of-concept, phase 2A trial with a target accrual of 104 patients.
The IRIS-RA study was funded by Janssen Research & Development. Dr. Taylor serves as a consultant to AbbVie, Biogen, Eli Lilly, Fresenius, Galapagos, Gilead Sciences, GlaxoSmithKline, Janssen, Nordic Pharma, Pfizer, Sanofi, Aqtual, and UCB and received research funding from Galapagos, among others. Dr. Emery received research grants paid to his institution from AbbVie, Bristol Myers Squibb (BMS), Pfizer, MSD, and Roche; received consultant fees from BMS, AbbVie, Pfizer, MSD, Novartis, Roche, and UCB; and has undertaken clinical trials and provided expert advice to Pfizer, MSD, AbbVie, BMS, UCB, Roche, Novartis, Samsung, Sandoz, and Lilly.
A version of this article appeared on Medscape.com.
LIVERPOOL, ENGLAND — The IRIS-RA study of the investigational monoclonal antibody drug nipocalimab in patients with rheumatoid arthritis (RA) did not meet its primary endpoint, but there could still be people with moderate to severe RA who might benefit from treatment with the drug, according to information reported at the British Society for Rheumatology annual meeting.
The primary endpoint for the phase 2A trial was the least squares mean change in Disease Activity Score in 28 joints using C-reactive protein (DAS28-CRP) from baseline to 12 weeks of treatment. This was reduced by −1.03 with nipocalimab and by −0.58 with placebo, giving a mean difference of just −0.45 (P = .224).
However, one of the key secondary endpoints was the proportion of patients who had 20% improvement in American College of Rheumatology response criteria (ACR20). Results for this endpoint showed a greater difference in response to nipocalimab vs placebo, with a respective 45.5% and 20.0% (P = .055) of individuals achieving ACR20.
Moreover, an analysis stratifying for anti-citrullinated protein autoantibody (ACPA) levels at baseline found that people with higher levels had a better response to nipocalimab.
Choice of Endpoint
“The way this study was powered was to look at a change between the treatment groups of a DAS28-CRP reduction of 1.0,” said Peter C. Taylor, BMBCh, PhD, the Norman Collisson chair of musculoskeletal medicine at the University of Oxford in Oxford, England.
DAS28-CRP was often chosen as the primary endpoint in small proof-of-concept studies, such as IRIS-RA, because it was a “measure of continuous change [that] theoretically, would allow greater sensitivity to change,” Dr. Taylor added.
“Ironically, it has to be said that had we chosen ACR20, we would have hit the primary endpoint. One lives and learns,” noted Dr. Taylor.
Proof of Concept
IRIS-RA was billed as a “proof-of-concept” study because it was the first time that a monoclonal antibody targeting the neonatal fragment crystallizable receptor (FcRn) was being tested in an RA population.
The study was a randomized double-blind trial in which 33 people with moderate to severe RA who had an inadequate response to tumor necrosis factor (TNF) inhibitors were treated with nipocalimab at a dose of 15 mg/kg given intravenously every 2 weeks, and 20 received a matching placebo. Participants were treated for 10 weeks, and then the primary follow-up was at 12 weeks, with additional follow-up for safety undertaken at 18 weeks.
Nipocalimab is a fully human, immunoglobulin G1 (IgG1) monoclonal antibody that is designed to selectively block the FcRn. By doing so, it essentially stops IgG from being recycled within the immune system, and this in turn lowers IgG levels. That includes potentially harmful ACPAs, among other pathogenic antibodies, Dr. Taylor and fellow investigators explained in their abstract.
“We’ve known for a long time that ACPA have prognostic value, but there’s been controversy about whether or not ACPA are actually pathogenic,” Dr. Taylor said. “So, one of the hypotheses that this study gives rise to is that by blocking FcRn, and thereby reducing, potentially, the concentration of ACPA in the blood, will we actually have efficacy in patients?”
Are ACPA Really Lowered?
Paul Emery, MD, Versus Arthritis professor of rheumatology and director of the Leeds Biomedical Research Centre at the University of Leeds in Leeds, England, questioned the reduction in antibody levels during the discussion that followed.
Although these data had not been presented, Dr. Emery observed that the reduction in IgG was actually greater than that in ACPA, “which is fairly critical. Is it feasible to look to selectively lower normal immunoglobulin over pathogenic autoantibodies?”
Dr. Emery also wanted to know if there “was a floor on the reduction of immunoglobulin” with long-term therapy, “which would be a worry.”
Dr. Taylor responded that total IgG had been reduced by about 65% and ACPA by about 40%. Why this difference exists is not yet clear. It could be because ACPA are part of complexed antibodies.
“Most of these patients are rheumatoid factor [RF]–positive,” said Taylor, pointing out that although IgM “wouldn’t normally be affected by FcRn blockade,” there was a 10% reduction in RF IgM, probably because it was complexed to IgG.
“So, the hypothesis here is that if you look at the clearance of complexes, they’re handled differently in the cytoplasm from the clearance of monomeric IgG. But that’s a hypothesis. It needs further investigation. In vitro, there’s very good, confirmatory evidence to support that. But we’ve yet to explore that more fully in vivo,” Dr. Taylor said.
As for long-term effects, Dr. Taylor responded: “All I can tell you is [that] after the 10-week intervention, that up to an 18-week observation period, immunoglobulin levels recovered very rapidly afterwards. And you mustn’t forget that other isotypes are not affected, unlike rituximab.”
Safety and Other Results
With regard to safety, 27 (82%) of nipocalimab- and 12 (60%) of placebo-treated participants experienced at least one treatment-emergent adverse event (TEAE). The most common, occurring in 10% or more of cases, were RA flares (36.4% for nipocalimab vs 15.0% with placebo), headache (12.1% vs 5.0%), and COVID-19 (12.1% vs 0.0%).
There were three serious TEAEs, all in the nipocalimab-treatment group: One was an infection of a burn that had been present at inclusion, another was a deep vein thrombosis that resolved with apixaban treatment, and the other was an infusion-related reaction that resolved with supportive treatment.
Another notable efficacy finding was the proportion of patients achieving DAS28-CRP remission at 12 weeks in the nipocalimab vs the placebo group was substantially greater if considering only people with high baseline ACPA levels, at a respective 40.0% vs 16.7%, when compared with the total population (21.2% vs 10.0%).
Similar findings were seen for the proportion of patients achieving an ACR50, and there were numerically greater reductions in the components of the ACR response criteria such as tender and swollen joints with nipocalimab vs placebo. All of these were exploratory observations, Dr. Taylor emphasized.
Combination and Further Trials
Further trials of nipocalimab are planned or are already ongoing in systemic lupus erythematosus, active lupus nephritis, Sjögren disease, and five other diseases.
In RA, nipocalimab is now being tested in combination with the TNF inhibitor certolizumab pegol (Cimzia) in the DAISY-RA trial. This is another proof-of-concept, phase 2A trial with a target accrual of 104 patients.
The IRIS-RA study was funded by Janssen Research & Development. Dr. Taylor serves as a consultant to AbbVie, Biogen, Eli Lilly, Fresenius, Galapagos, Gilead Sciences, GlaxoSmithKline, Janssen, Nordic Pharma, Pfizer, Sanofi, Aqtual, and UCB and received research funding from Galapagos, among others. Dr. Emery received research grants paid to his institution from AbbVie, Bristol Myers Squibb (BMS), Pfizer, MSD, and Roche; received consultant fees from BMS, AbbVie, Pfizer, MSD, Novartis, Roche, and UCB; and has undertaken clinical trials and provided expert advice to Pfizer, MSD, AbbVie, BMS, UCB, Roche, Novartis, Samsung, Sandoz, and Lilly.
A version of this article appeared on Medscape.com.
FROM BSR 2024










