User login
FDA grants emergency use authorization to Lilly’s antibody COVID-19 therapy
The monoclonal antibody therapy has emergency authorization for treating patients who have tested positive for SARS-CoV-2 infection and who are considered to be at high risk for progression to severe COVID-19 or hospitalization. To be eligible for treatment with bamlanivimab, patients must be at least 12 years of age and weigh at least 40 kg (approximately 88 lb). The agency notes that this includes patients aged 65 years and older or people with certain chronic conditions.
Bamlanivimab is not authorized for use in patients who are hospitalized or who require oxygen therapy because of COVID-19. The FDA’s action comes less than 2 weeks after Eli Lilly halted the ACTIV-3 study of the therapy for severe, hospitalized COVID-19 patients after evidence showed that adding the antibody therapy to standard care did not improve outcomes over standard care alone for patients with advanced COVID-19.
The government contract with Eli Lilly involves the purchase of 300,000 doses through December, with the option to procure another 650,000 doses through June 2021.
Because of Operation Warp Speed, “we have supplies to distribute now. Product distribution will begin this week,” US Health & Human Services (HHS) Secretary Alex Azar said at a news conference today.
“We talked about building the bridge to safe and effective vaccines” for COVID-19, Azar added. “With this therapeutic, the bridge is taking shape.”
Bamlanivimab 700 mg will be administered as a 1-hour infusion followed by a 1-hour observation period for detecting any infusion-related side effects. The authorized dose is 700 mg, which was on the lower end of the dose range evaluated in studies.
During the press conference, a reporter asked whether the lower dose was chosen in order that more doses of the antibody could be made available. “The lower dose is a rational choice in this situation because we don’t want to give more of a drug than you need,” said Janet Woodcock, MD, the therapeutics lead for Operation Warp Speed. “I think we could probably go lower.”
Bamlanivimab works by attaching to the virus and blocking its entry into the cells and possibly by helping the patients’ immune system clear the virus, said Woodcock, who is also director of the FDA’s Center for Drug Evaluation and Research.
“The goal is to treat high-risk people as soon as possible after they show symptoms and are diagnosed,” she added.
Infusions an initial challenge?
There could be some logistic challenges at first because the antibody is administered via infusion. “We expect there will initially be a challenge in administering ... these infusions and setting up infusion centers,” Woodcock said.
Outpatient intravenous infusions are normally performed at infusion centers for patients with cancer and immune disorders, she noted. “You really don’t want them mixing with people who have COVID-19 disease, so we will need to set up separate sites.”
Bamlanivimab will be provided free of cost to patients, Azar said. Patients should be aware that coinsurance may be required for the infusion.
“Fair and equitable” distribution planned
During phase 1 of distribution, the agent will first be allocated to hospitals and hospital-affiliated locations only, John Redd, MD, MPH, chief medical officer, Office of the Assistant Secretary for Preparedness and Response at HHS, said at the press conference.
During phase 2, “there will be expanded distribution to outpatient sites,” he said. In an effort to keep the process transparent, a new website features the latest updates on the distribution of bamlanivimab.
Allocation will be based on two factors: the number of new cases reported in a state or territory in the prior 7 days, and rates of COVID-19 hospitalization during the same period.
Asked why the government would determine distribution of the antibody on the basis of the number of hospitalized patients when the indication includes prevention of admission, Woodcock replied that hospitalization is a surrogate measure that can reflect risk factors in a particular state population, such as obesity, diabetes, or the proportion of older people.
Furthermore, the confirmed cases are a “leading indicator,” she said, that can help identify a steep rise in COVID-19 cases that could indicate more hospitalizations are likely soon. “We don’t want to miss that.”
Data underlying the EUA decision
A decrease in hospitalizations or emergency department visits within 28 days of treatment in preclinical studies was “the most important evidence that bamlanivimab may be effective,” the agency noted in the press release announcing the EUA. Among patients at high risk for progression, 3% required such interventions, compared with 10% of placebo-treated patients.
Potential side effects of bamlanivimab include anaphylaxis, infusion-related reactions, nausea, diarrhea, dizziness, headache, itching, and vomiting.
“As illustrated by today’s action, the FDA remains committed to expediting the development and availability of potential COVID-19 treatments and providing sick patients timely access to new therapies where appropriate,” FDA Commissioner Stephen M. Hahn, MD, said in the news release.
Healthcare providers can download a detailed FDA fact sheet on the EUA for bamlanivimab, which includes dosing instructions.
This article first appeared on Medscape.com.
The monoclonal antibody therapy has emergency authorization for treating patients who have tested positive for SARS-CoV-2 infection and who are considered to be at high risk for progression to severe COVID-19 or hospitalization. To be eligible for treatment with bamlanivimab, patients must be at least 12 years of age and weigh at least 40 kg (approximately 88 lb). The agency notes that this includes patients aged 65 years and older or people with certain chronic conditions.
Bamlanivimab is not authorized for use in patients who are hospitalized or who require oxygen therapy because of COVID-19. The FDA’s action comes less than 2 weeks after Eli Lilly halted the ACTIV-3 study of the therapy for severe, hospitalized COVID-19 patients after evidence showed that adding the antibody therapy to standard care did not improve outcomes over standard care alone for patients with advanced COVID-19.
The government contract with Eli Lilly involves the purchase of 300,000 doses through December, with the option to procure another 650,000 doses through June 2021.
Because of Operation Warp Speed, “we have supplies to distribute now. Product distribution will begin this week,” US Health & Human Services (HHS) Secretary Alex Azar said at a news conference today.
“We talked about building the bridge to safe and effective vaccines” for COVID-19, Azar added. “With this therapeutic, the bridge is taking shape.”
Bamlanivimab 700 mg will be administered as a 1-hour infusion followed by a 1-hour observation period for detecting any infusion-related side effects. The authorized dose is 700 mg, which was on the lower end of the dose range evaluated in studies.
During the press conference, a reporter asked whether the lower dose was chosen in order that more doses of the antibody could be made available. “The lower dose is a rational choice in this situation because we don’t want to give more of a drug than you need,” said Janet Woodcock, MD, the therapeutics lead for Operation Warp Speed. “I think we could probably go lower.”
Bamlanivimab works by attaching to the virus and blocking its entry into the cells and possibly by helping the patients’ immune system clear the virus, said Woodcock, who is also director of the FDA’s Center for Drug Evaluation and Research.
“The goal is to treat high-risk people as soon as possible after they show symptoms and are diagnosed,” she added.
Infusions an initial challenge?
There could be some logistic challenges at first because the antibody is administered via infusion. “We expect there will initially be a challenge in administering ... these infusions and setting up infusion centers,” Woodcock said.
Outpatient intravenous infusions are normally performed at infusion centers for patients with cancer and immune disorders, she noted. “You really don’t want them mixing with people who have COVID-19 disease, so we will need to set up separate sites.”
Bamlanivimab will be provided free of cost to patients, Azar said. Patients should be aware that coinsurance may be required for the infusion.
“Fair and equitable” distribution planned
During phase 1 of distribution, the agent will first be allocated to hospitals and hospital-affiliated locations only, John Redd, MD, MPH, chief medical officer, Office of the Assistant Secretary for Preparedness and Response at HHS, said at the press conference.
During phase 2, “there will be expanded distribution to outpatient sites,” he said. In an effort to keep the process transparent, a new website features the latest updates on the distribution of bamlanivimab.
Allocation will be based on two factors: the number of new cases reported in a state or territory in the prior 7 days, and rates of COVID-19 hospitalization during the same period.
Asked why the government would determine distribution of the antibody on the basis of the number of hospitalized patients when the indication includes prevention of admission, Woodcock replied that hospitalization is a surrogate measure that can reflect risk factors in a particular state population, such as obesity, diabetes, or the proportion of older people.
Furthermore, the confirmed cases are a “leading indicator,” she said, that can help identify a steep rise in COVID-19 cases that could indicate more hospitalizations are likely soon. “We don’t want to miss that.”
Data underlying the EUA decision
A decrease in hospitalizations or emergency department visits within 28 days of treatment in preclinical studies was “the most important evidence that bamlanivimab may be effective,” the agency noted in the press release announcing the EUA. Among patients at high risk for progression, 3% required such interventions, compared with 10% of placebo-treated patients.
Potential side effects of bamlanivimab include anaphylaxis, infusion-related reactions, nausea, diarrhea, dizziness, headache, itching, and vomiting.
“As illustrated by today’s action, the FDA remains committed to expediting the development and availability of potential COVID-19 treatments and providing sick patients timely access to new therapies where appropriate,” FDA Commissioner Stephen M. Hahn, MD, said in the news release.
Healthcare providers can download a detailed FDA fact sheet on the EUA for bamlanivimab, which includes dosing instructions.
This article first appeared on Medscape.com.
The monoclonal antibody therapy has emergency authorization for treating patients who have tested positive for SARS-CoV-2 infection and who are considered to be at high risk for progression to severe COVID-19 or hospitalization. To be eligible for treatment with bamlanivimab, patients must be at least 12 years of age and weigh at least 40 kg (approximately 88 lb). The agency notes that this includes patients aged 65 years and older or people with certain chronic conditions.
Bamlanivimab is not authorized for use in patients who are hospitalized or who require oxygen therapy because of COVID-19. The FDA’s action comes less than 2 weeks after Eli Lilly halted the ACTIV-3 study of the therapy for severe, hospitalized COVID-19 patients after evidence showed that adding the antibody therapy to standard care did not improve outcomes over standard care alone for patients with advanced COVID-19.
The government contract with Eli Lilly involves the purchase of 300,000 doses through December, with the option to procure another 650,000 doses through June 2021.
Because of Operation Warp Speed, “we have supplies to distribute now. Product distribution will begin this week,” US Health & Human Services (HHS) Secretary Alex Azar said at a news conference today.
“We talked about building the bridge to safe and effective vaccines” for COVID-19, Azar added. “With this therapeutic, the bridge is taking shape.”
Bamlanivimab 700 mg will be administered as a 1-hour infusion followed by a 1-hour observation period for detecting any infusion-related side effects. The authorized dose is 700 mg, which was on the lower end of the dose range evaluated in studies.
During the press conference, a reporter asked whether the lower dose was chosen in order that more doses of the antibody could be made available. “The lower dose is a rational choice in this situation because we don’t want to give more of a drug than you need,” said Janet Woodcock, MD, the therapeutics lead for Operation Warp Speed. “I think we could probably go lower.”
Bamlanivimab works by attaching to the virus and blocking its entry into the cells and possibly by helping the patients’ immune system clear the virus, said Woodcock, who is also director of the FDA’s Center for Drug Evaluation and Research.
“The goal is to treat high-risk people as soon as possible after they show symptoms and are diagnosed,” she added.
Infusions an initial challenge?
There could be some logistic challenges at first because the antibody is administered via infusion. “We expect there will initially be a challenge in administering ... these infusions and setting up infusion centers,” Woodcock said.
Outpatient intravenous infusions are normally performed at infusion centers for patients with cancer and immune disorders, she noted. “You really don’t want them mixing with people who have COVID-19 disease, so we will need to set up separate sites.”
Bamlanivimab will be provided free of cost to patients, Azar said. Patients should be aware that coinsurance may be required for the infusion.
“Fair and equitable” distribution planned
During phase 1 of distribution, the agent will first be allocated to hospitals and hospital-affiliated locations only, John Redd, MD, MPH, chief medical officer, Office of the Assistant Secretary for Preparedness and Response at HHS, said at the press conference.
During phase 2, “there will be expanded distribution to outpatient sites,” he said. In an effort to keep the process transparent, a new website features the latest updates on the distribution of bamlanivimab.
Allocation will be based on two factors: the number of new cases reported in a state or territory in the prior 7 days, and rates of COVID-19 hospitalization during the same period.
Asked why the government would determine distribution of the antibody on the basis of the number of hospitalized patients when the indication includes prevention of admission, Woodcock replied that hospitalization is a surrogate measure that can reflect risk factors in a particular state population, such as obesity, diabetes, or the proportion of older people.
Furthermore, the confirmed cases are a “leading indicator,” she said, that can help identify a steep rise in COVID-19 cases that could indicate more hospitalizations are likely soon. “We don’t want to miss that.”
Data underlying the EUA decision
A decrease in hospitalizations or emergency department visits within 28 days of treatment in preclinical studies was “the most important evidence that bamlanivimab may be effective,” the agency noted in the press release announcing the EUA. Among patients at high risk for progression, 3% required such interventions, compared with 10% of placebo-treated patients.
Potential side effects of bamlanivimab include anaphylaxis, infusion-related reactions, nausea, diarrhea, dizziness, headache, itching, and vomiting.
“As illustrated by today’s action, the FDA remains committed to expediting the development and availability of potential COVID-19 treatments and providing sick patients timely access to new therapies where appropriate,” FDA Commissioner Stephen M. Hahn, MD, said in the news release.
Healthcare providers can download a detailed FDA fact sheet on the EUA for bamlanivimab, which includes dosing instructions.
This article first appeared on Medscape.com.
Pfizer vaccine data show 90% efficacy in early results
A vaccine candidate against SARS-CoV-2 has been found to be 90% effective in preventing COVID-19 in trial volunteers who were without evidence of prior infection of the virus, results from an interim analysis of a phase 3 study demonstrated.
BTN162b2, a messenger RNA–based vaccine candidate that requires two doses, is being developed by Pfizer and BioNTech SE independently of the Trump administration’s Operation Warp Speed. A global phase 3 clinical trial of BTN162b2 began on July 27 and has enrolled 43,538 participants to date; 42% of enrollees have racially and ethnically diverse backgrounds.
According to a press release issued by the two companies, 38,955 trial volunteers had received a second dose of either vaccine or placebo as of Nov. 8. An interim analysis of 94 individuals conducted by an independent data monitoring committee (DMC) found that the vaccine efficacy rate was above 90% 7 days after the second dose. This means that protection was achieved 28 days after the first vaccine dose.
“It’s promising in that it validates the genetic strategy – whether it’s mRNA vaccines or DNA vaccines,” Paul A. Offit, MD, told Medscape Medical News. Offit is a member of the US Food and Drug Administraiton’s COVID-19 Vaccine Advisory Committee. “All of them have the same approach, which is that they introduce the gene that codes for the coronavirus spike protein into the cell. Your cell makes the spike protein, and your immune system makes antibodies to the spike protein. At least in these preliminary data, which involved 94 people getting sick, it looks like it’s effective. That’s good. We knew that it seemed to work in experimental animals, but you never know until you put it into people.”
According to Pfizer and BioNTech SE, a final data analysis is planned once 164 confirmed COVID-19 cases have accrued. So far, the DMC has not reported any serious safety concerns. It recommends that the study continue to collect safety and efficacy data as planned. The companies plan to apply to the FDA for emergency use authorization soon after the required safety milestone is achieved.
Pfizer CEO Albert Bourla, DVM, PhD, added in a separate press release, “It’s important to note that we cannot apply for FDA Emergency Use Authorization based on these efficacy results alone. More data on safety is also needed, and we are continuing to accumulate that safety data as part of our ongoing clinical study.
“We estimate that a median of two months of safety data following the second and final dose of the vaccine candidate – required by FDA’s guidance for potential Emergency Use Authorization – will be available by the third week of November.”
Offit, professor of pediatrics in the Division of Infectious Diseases at the Children’s Hospital of Philadelphia, said that, if BTN162b2 is approved, administering it will be tricky. “This particular vaccine has to be shipped and stored at –70° C or –80° C, which we’ve never done before in this country,” he said. “That means maintaining the product on dry ice. That’s going to be a challenge for distribution, I think.”
Good news, but…
In the press release, BioNTech SE’s cofounder and CEO, Ugur Sahin, MD, characterized the findings as “a victory for innovation, science and a global collaborative effort. When we embarked on this journey 10 months ago this is what we aspired to achieve. Especially today, while we are all in the midst of a second wave and many of us in lockdown, we appreciate even more how important this milestone is on our path towards ending this pandemic and for all of us to regain a sense of normality.”
President-elect Joe Biden also weighed in, calling the results “excellent news” in a news release.
“At the same time, it is also important to understand that the end of the battle against COVID-19 is still months away,” he said. “This news follows a previously announced timeline by industry officials that forecast vaccine approval by late November. Even if that is achieved, and some Americans are vaccinated later this year, it will be many more months before there is widespread vaccination in this country.
“Today’s news does not change this urgent reality. Americans will have to rely on masking, distancing, contact tracing, hand washing, and other measures to keep themselves safe well into next year,” Biden added.
This article first appeared on Medscape.com.
A vaccine candidate against SARS-CoV-2 has been found to be 90% effective in preventing COVID-19 in trial volunteers who were without evidence of prior infection of the virus, results from an interim analysis of a phase 3 study demonstrated.
BTN162b2, a messenger RNA–based vaccine candidate that requires two doses, is being developed by Pfizer and BioNTech SE independently of the Trump administration’s Operation Warp Speed. A global phase 3 clinical trial of BTN162b2 began on July 27 and has enrolled 43,538 participants to date; 42% of enrollees have racially and ethnically diverse backgrounds.
According to a press release issued by the two companies, 38,955 trial volunteers had received a second dose of either vaccine or placebo as of Nov. 8. An interim analysis of 94 individuals conducted by an independent data monitoring committee (DMC) found that the vaccine efficacy rate was above 90% 7 days after the second dose. This means that protection was achieved 28 days after the first vaccine dose.
“It’s promising in that it validates the genetic strategy – whether it’s mRNA vaccines or DNA vaccines,” Paul A. Offit, MD, told Medscape Medical News. Offit is a member of the US Food and Drug Administraiton’s COVID-19 Vaccine Advisory Committee. “All of them have the same approach, which is that they introduce the gene that codes for the coronavirus spike protein into the cell. Your cell makes the spike protein, and your immune system makes antibodies to the spike protein. At least in these preliminary data, which involved 94 people getting sick, it looks like it’s effective. That’s good. We knew that it seemed to work in experimental animals, but you never know until you put it into people.”
According to Pfizer and BioNTech SE, a final data analysis is planned once 164 confirmed COVID-19 cases have accrued. So far, the DMC has not reported any serious safety concerns. It recommends that the study continue to collect safety and efficacy data as planned. The companies plan to apply to the FDA for emergency use authorization soon after the required safety milestone is achieved.
Pfizer CEO Albert Bourla, DVM, PhD, added in a separate press release, “It’s important to note that we cannot apply for FDA Emergency Use Authorization based on these efficacy results alone. More data on safety is also needed, and we are continuing to accumulate that safety data as part of our ongoing clinical study.
“We estimate that a median of two months of safety data following the second and final dose of the vaccine candidate – required by FDA’s guidance for potential Emergency Use Authorization – will be available by the third week of November.”
Offit, professor of pediatrics in the Division of Infectious Diseases at the Children’s Hospital of Philadelphia, said that, if BTN162b2 is approved, administering it will be tricky. “This particular vaccine has to be shipped and stored at –70° C or –80° C, which we’ve never done before in this country,” he said. “That means maintaining the product on dry ice. That’s going to be a challenge for distribution, I think.”
Good news, but…
In the press release, BioNTech SE’s cofounder and CEO, Ugur Sahin, MD, characterized the findings as “a victory for innovation, science and a global collaborative effort. When we embarked on this journey 10 months ago this is what we aspired to achieve. Especially today, while we are all in the midst of a second wave and many of us in lockdown, we appreciate even more how important this milestone is on our path towards ending this pandemic and for all of us to regain a sense of normality.”
President-elect Joe Biden also weighed in, calling the results “excellent news” in a news release.
“At the same time, it is also important to understand that the end of the battle against COVID-19 is still months away,” he said. “This news follows a previously announced timeline by industry officials that forecast vaccine approval by late November. Even if that is achieved, and some Americans are vaccinated later this year, it will be many more months before there is widespread vaccination in this country.
“Today’s news does not change this urgent reality. Americans will have to rely on masking, distancing, contact tracing, hand washing, and other measures to keep themselves safe well into next year,” Biden added.
This article first appeared on Medscape.com.
A vaccine candidate against SARS-CoV-2 has been found to be 90% effective in preventing COVID-19 in trial volunteers who were without evidence of prior infection of the virus, results from an interim analysis of a phase 3 study demonstrated.
BTN162b2, a messenger RNA–based vaccine candidate that requires two doses, is being developed by Pfizer and BioNTech SE independently of the Trump administration’s Operation Warp Speed. A global phase 3 clinical trial of BTN162b2 began on July 27 and has enrolled 43,538 participants to date; 42% of enrollees have racially and ethnically diverse backgrounds.
According to a press release issued by the two companies, 38,955 trial volunteers had received a second dose of either vaccine or placebo as of Nov. 8. An interim analysis of 94 individuals conducted by an independent data monitoring committee (DMC) found that the vaccine efficacy rate was above 90% 7 days after the second dose. This means that protection was achieved 28 days after the first vaccine dose.
“It’s promising in that it validates the genetic strategy – whether it’s mRNA vaccines or DNA vaccines,” Paul A. Offit, MD, told Medscape Medical News. Offit is a member of the US Food and Drug Administraiton’s COVID-19 Vaccine Advisory Committee. “All of them have the same approach, which is that they introduce the gene that codes for the coronavirus spike protein into the cell. Your cell makes the spike protein, and your immune system makes antibodies to the spike protein. At least in these preliminary data, which involved 94 people getting sick, it looks like it’s effective. That’s good. We knew that it seemed to work in experimental animals, but you never know until you put it into people.”
According to Pfizer and BioNTech SE, a final data analysis is planned once 164 confirmed COVID-19 cases have accrued. So far, the DMC has not reported any serious safety concerns. It recommends that the study continue to collect safety and efficacy data as planned. The companies plan to apply to the FDA for emergency use authorization soon after the required safety milestone is achieved.
Pfizer CEO Albert Bourla, DVM, PhD, added in a separate press release, “It’s important to note that we cannot apply for FDA Emergency Use Authorization based on these efficacy results alone. More data on safety is also needed, and we are continuing to accumulate that safety data as part of our ongoing clinical study.
“We estimate that a median of two months of safety data following the second and final dose of the vaccine candidate – required by FDA’s guidance for potential Emergency Use Authorization – will be available by the third week of November.”
Offit, professor of pediatrics in the Division of Infectious Diseases at the Children’s Hospital of Philadelphia, said that, if BTN162b2 is approved, administering it will be tricky. “This particular vaccine has to be shipped and stored at –70° C or –80° C, which we’ve never done before in this country,” he said. “That means maintaining the product on dry ice. That’s going to be a challenge for distribution, I think.”
Good news, but…
In the press release, BioNTech SE’s cofounder and CEO, Ugur Sahin, MD, characterized the findings as “a victory for innovation, science and a global collaborative effort. When we embarked on this journey 10 months ago this is what we aspired to achieve. Especially today, while we are all in the midst of a second wave and many of us in lockdown, we appreciate even more how important this milestone is on our path towards ending this pandemic and for all of us to regain a sense of normality.”
President-elect Joe Biden also weighed in, calling the results “excellent news” in a news release.
“At the same time, it is also important to understand that the end of the battle against COVID-19 is still months away,” he said. “This news follows a previously announced timeline by industry officials that forecast vaccine approval by late November. Even if that is achieved, and some Americans are vaccinated later this year, it will be many more months before there is widespread vaccination in this country.
“Today’s news does not change this urgent reality. Americans will have to rely on masking, distancing, contact tracing, hand washing, and other measures to keep themselves safe well into next year,” Biden added.
This article first appeared on Medscape.com.
Few women hospitalized for influenza have been vaccinated
Researchers analyzed data from 9,652 women ages 15-44 who were hospitalized with laboratory-confirmed influenza from October through April during the 2010-2019 influenza seasons. Data were pulled from the U.S. Influenza Hospitalization Surveillance Network (FluSurv-NET).
Of those women, 2,697 (28%) were pregnant. Median age was 28 and median gestational age was 32 weeks. Those studied included 36% who were non-Hispanic White; 29% non-Hispanic Black; and 20% Hispanic women.
Some 89% of the women, pregnant and nonpregnant, received antivirals while in the hospital but only 31% reported they had received the flu vaccine in the current season, despite guideline recommendations citing clear evidence that vaccination is safe for mother and baby.
Rachel Holstein, MPH, an epidemiology and information science fellow at the Centers for Disease Control and Prevention, who presented her team’s work as part of IDWeek 2020, explained that the mother’s vaccination can help protect the baby from flu infection for several months after birth, before the baby can be vaccinated.
She noted that pregnant women are at high risk for influenza-associated hospitalization.
“Changes in the immune system, heart, and lungs during pregnancy make pregnant women, and women up to 2 weeks post partum, more prone to severe illness from flu, including illness resulting in hospitalization,” she said in an interview
“Vaccination has been shown to reduce the risk of flu-associated acute respiratory infection in pregnant women by up to one-half,” she said. “A 2018 study showed that getting a flu shot reduced a pregnant woman’s risk of being hospitalized with flu by an average of 40%.»
FluSurv-NET data show hospitalizations were more common in the third trimester of pregnancy compared with the first and second, Holstein said. The most common underlying conditions among these women were asthma (23%) and obesity (10%), and 12% were current tobacco smokers. Overall, 5% of pregnant women with flu required ICU admission, 2% needed mechanical ventilation, and 6% developed pneumonia.
Vaccine uptake lowest in first two trimesters
Holstein said vaccine coverage was lowest among women in their first or second trimesters for all 9 seasons, and overall vaccination coverage increased significantly over time.
Uptake also differed by age. The data showed coverage was lower among women aged 15-34 years, compared with women 35 years and older (34% vs. 50%).
“It was as low as 15% among pregnant women aged 15-34 years in the 2011-12 season,” she added.
Jeanne Sheffield, MD, director of the division of maternal-fetal medicine at Johns Hopkins Medicine, Baltimore, said in an interview the low uptake of vaccine shown in this study is both familiar and frustrating.
She said education from health care providers has improved, but women are nonetheless frequently fearful. She pointed out the widespread phenomenon of vaccine hesitancy in the general population.
Coverage was 45.3% among adults in the 2018-2019 flu season, 8.2 percentage points higher than coverage during the 2017-18 season (37.1%) according to CDC estimates.
Added to that, she said, is further hesitancy when women believe vaccination could harm the unborn baby, despite “very good data that flu vaccine is safe in pregnancy, acceptable in pregnancy in all trimesters, and is optimal standard of care.”
Holstein added, “We know from past research that a range of factors – including negative attitudes and beliefs about vaccines, less knowledge about and access to vaccines, and a lack of trust in healthcare providers and vaccines – can contribute to lower vaccination rates.”
Healthcare providers play a key role in increasing flu vaccinations among pregnant women, she said.
“A provider recommendation, combined with an offer to administer a flu vaccine at the time of visit, remains one of the best ways to accomplish this,” Holstein said.
Holstein and Sheffield have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Researchers analyzed data from 9,652 women ages 15-44 who were hospitalized with laboratory-confirmed influenza from October through April during the 2010-2019 influenza seasons. Data were pulled from the U.S. Influenza Hospitalization Surveillance Network (FluSurv-NET).
Of those women, 2,697 (28%) were pregnant. Median age was 28 and median gestational age was 32 weeks. Those studied included 36% who were non-Hispanic White; 29% non-Hispanic Black; and 20% Hispanic women.
Some 89% of the women, pregnant and nonpregnant, received antivirals while in the hospital but only 31% reported they had received the flu vaccine in the current season, despite guideline recommendations citing clear evidence that vaccination is safe for mother and baby.
Rachel Holstein, MPH, an epidemiology and information science fellow at the Centers for Disease Control and Prevention, who presented her team’s work as part of IDWeek 2020, explained that the mother’s vaccination can help protect the baby from flu infection for several months after birth, before the baby can be vaccinated.
She noted that pregnant women are at high risk for influenza-associated hospitalization.
“Changes in the immune system, heart, and lungs during pregnancy make pregnant women, and women up to 2 weeks post partum, more prone to severe illness from flu, including illness resulting in hospitalization,” she said in an interview
“Vaccination has been shown to reduce the risk of flu-associated acute respiratory infection in pregnant women by up to one-half,” she said. “A 2018 study showed that getting a flu shot reduced a pregnant woman’s risk of being hospitalized with flu by an average of 40%.»
FluSurv-NET data show hospitalizations were more common in the third trimester of pregnancy compared with the first and second, Holstein said. The most common underlying conditions among these women were asthma (23%) and obesity (10%), and 12% were current tobacco smokers. Overall, 5% of pregnant women with flu required ICU admission, 2% needed mechanical ventilation, and 6% developed pneumonia.
Vaccine uptake lowest in first two trimesters
Holstein said vaccine coverage was lowest among women in their first or second trimesters for all 9 seasons, and overall vaccination coverage increased significantly over time.
Uptake also differed by age. The data showed coverage was lower among women aged 15-34 years, compared with women 35 years and older (34% vs. 50%).
“It was as low as 15% among pregnant women aged 15-34 years in the 2011-12 season,” she added.
Jeanne Sheffield, MD, director of the division of maternal-fetal medicine at Johns Hopkins Medicine, Baltimore, said in an interview the low uptake of vaccine shown in this study is both familiar and frustrating.
She said education from health care providers has improved, but women are nonetheless frequently fearful. She pointed out the widespread phenomenon of vaccine hesitancy in the general population.
Coverage was 45.3% among adults in the 2018-2019 flu season, 8.2 percentage points higher than coverage during the 2017-18 season (37.1%) according to CDC estimates.
Added to that, she said, is further hesitancy when women believe vaccination could harm the unborn baby, despite “very good data that flu vaccine is safe in pregnancy, acceptable in pregnancy in all trimesters, and is optimal standard of care.”
Holstein added, “We know from past research that a range of factors – including negative attitudes and beliefs about vaccines, less knowledge about and access to vaccines, and a lack of trust in healthcare providers and vaccines – can contribute to lower vaccination rates.”
Healthcare providers play a key role in increasing flu vaccinations among pregnant women, she said.
“A provider recommendation, combined with an offer to administer a flu vaccine at the time of visit, remains one of the best ways to accomplish this,” Holstein said.
Holstein and Sheffield have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Researchers analyzed data from 9,652 women ages 15-44 who were hospitalized with laboratory-confirmed influenza from October through April during the 2010-2019 influenza seasons. Data were pulled from the U.S. Influenza Hospitalization Surveillance Network (FluSurv-NET).
Of those women, 2,697 (28%) were pregnant. Median age was 28 and median gestational age was 32 weeks. Those studied included 36% who were non-Hispanic White; 29% non-Hispanic Black; and 20% Hispanic women.
Some 89% of the women, pregnant and nonpregnant, received antivirals while in the hospital but only 31% reported they had received the flu vaccine in the current season, despite guideline recommendations citing clear evidence that vaccination is safe for mother and baby.
Rachel Holstein, MPH, an epidemiology and information science fellow at the Centers for Disease Control and Prevention, who presented her team’s work as part of IDWeek 2020, explained that the mother’s vaccination can help protect the baby from flu infection for several months after birth, before the baby can be vaccinated.
She noted that pregnant women are at high risk for influenza-associated hospitalization.
“Changes in the immune system, heart, and lungs during pregnancy make pregnant women, and women up to 2 weeks post partum, more prone to severe illness from flu, including illness resulting in hospitalization,” she said in an interview
“Vaccination has been shown to reduce the risk of flu-associated acute respiratory infection in pregnant women by up to one-half,” she said. “A 2018 study showed that getting a flu shot reduced a pregnant woman’s risk of being hospitalized with flu by an average of 40%.»
FluSurv-NET data show hospitalizations were more common in the third trimester of pregnancy compared with the first and second, Holstein said. The most common underlying conditions among these women were asthma (23%) and obesity (10%), and 12% were current tobacco smokers. Overall, 5% of pregnant women with flu required ICU admission, 2% needed mechanical ventilation, and 6% developed pneumonia.
Vaccine uptake lowest in first two trimesters
Holstein said vaccine coverage was lowest among women in their first or second trimesters for all 9 seasons, and overall vaccination coverage increased significantly over time.
Uptake also differed by age. The data showed coverage was lower among women aged 15-34 years, compared with women 35 years and older (34% vs. 50%).
“It was as low as 15% among pregnant women aged 15-34 years in the 2011-12 season,” she added.
Jeanne Sheffield, MD, director of the division of maternal-fetal medicine at Johns Hopkins Medicine, Baltimore, said in an interview the low uptake of vaccine shown in this study is both familiar and frustrating.
She said education from health care providers has improved, but women are nonetheless frequently fearful. She pointed out the widespread phenomenon of vaccine hesitancy in the general population.
Coverage was 45.3% among adults in the 2018-2019 flu season, 8.2 percentage points higher than coverage during the 2017-18 season (37.1%) according to CDC estimates.
Added to that, she said, is further hesitancy when women believe vaccination could harm the unborn baby, despite “very good data that flu vaccine is safe in pregnancy, acceptable in pregnancy in all trimesters, and is optimal standard of care.”
Holstein added, “We know from past research that a range of factors – including negative attitudes and beliefs about vaccines, less knowledge about and access to vaccines, and a lack of trust in healthcare providers and vaccines – can contribute to lower vaccination rates.”
Healthcare providers play a key role in increasing flu vaccinations among pregnant women, she said.
“A provider recommendation, combined with an offer to administer a flu vaccine at the time of visit, remains one of the best ways to accomplish this,” Holstein said.
Holstein and Sheffield have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Two-thirds of U.S. teens fail to get needed vaccines
Only 30.6% of American adolescents complete three routinely recommended vaccinations, new research has found, but that number varies widely by state.
The Advisory Committee on Immunization Practices recommends that, by age 17 years, adolescents complete three key immunizations: human papillomavirus (HPV), quadrivalent meningococcal conjugate (MenACWY), and Tdap.
Sara Poston, PharmD, senior director for health outcomes research at GlaxoSmithKline, said at a press conference during IDWeek, an annual scientific meeting on infectious diseases held virtually this year, that her team set out to determine how many teens were completing the vaccinations and how the number varied by state and by behavioral factors.
Completion of the vaccines means getting the HPV series (two doses for people aged 9-14 years at first vaccination or three doses for those aged 15 years or older at first vaccination), completion of the MenACWY series (two doses), and getting a Tdap vaccine (one dose).
Rhode Island has the highest rates
Some states are clearly doing better than others. Idaho had the lowest completion rate (11.3%; 95% confidence interval, 6.9%-18.0%), and Rhode Island had the highest (56.4%; 95% CI, 49.8%-62.8%).
In the 2018 National Immunization Survey–Teen (NIS-Teen), Rhode Island had the highest vaccination coverage rate in the nation for meningococcal vaccine (98.7%) and the second-highest coverage rate for Tdap (96.3%) for adolescents aged 13-17 years. Also in 2018, the state had the highest vaccination rates in the nation for the HPV series for both male and female adolescents 13-17 years of age (78.1%), well above the national average of 51.1%.
Researchers used information from the Centers for Disease Control and Prevention as well as 2015-2018 NIS-Teen data to estimate national and state-level completion rates by age 17. They then combined NIS-Teen data with public state-level data to evaluate what was driving or discouraging completion.
“The good news is, we found some variables that we consider actionable and can be used by states and local health departments to improve the rates,” Dr. Poston said.
Those include encouraging a health care visit at age 16 or 17, provider recommendations to families to get the HPV vaccine, and state-level mandates for the MenACWY vaccine.
Those who had a health care visit at 16 or 17 were more than twice as likely to complete their vaccines (odds ratio, 2.35; 95% CI, 1.80-3.07). Those for whom HPV vaccination had ever been recommended by a health care provider were more than three times as likely to complete their vaccinations (OR, 3.24; 95% CI, 2.76-3.80).
Other factors predictive of completing the vaccines included being Black or Hispanic and having Medicaid insurance.
At the state level, “living in a state with a mandate for the meningococcal ACWY vaccine in elementary or secondary school was also associated with likelihood of vaccination,” Dr. Poston said. Teens in states with mandates were 60% more likely to complete the vaccines than those in states without mandates. (OR, 1.6; 95% CI, 1.2-2.3)
Marielle Fricchione, MD, assistant professor of pediatric infectious diseases at Rush Medical College, Chicago said in an interview, “Teen vaccines are notoriously hard to get into kids because it’s hard to get them back into the office for second doses.”
She said that Illinois is one of the states with a two-dose mandate for MenACWY before entering 6th grade and 12th grade, which has kept vaccination coverage high.
Educating providers on how to recommend HPV vaccination is the biggest vaccine focus, she added.
Schedule next dose at first visit
One thing her department has found successful in HPV completion is scheduling the second dose while the teen is in the office for the first dose.
“Also, you have to recommend it just as strongly for boys as you do for girls, because oropharyngeal cancer is like an epidemic right now for men, and HPV-related oropharyngeal cancer is on an exponential rise,” Dr. Fricchione said.
According to the CDC, HPV is thought to cause 70% of oropharyngeal cancers in the United States.
Equipping providers with statistics on the effectiveness of HPV vaccination in preventing cancer can take away the uneasiness in talking about sexual transmission.
“That really seems to help them give a strong recommendation. It puts them in a data-driven position to talk about the vaccine,” she said. “Once you put that data in front of the providers, they’re floored.”
Research was funded by GlaxoSmithKline. Dr. Poston is employed by GlaxoSmithKline. Dr. Fricchione disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Only 30.6% of American adolescents complete three routinely recommended vaccinations, new research has found, but that number varies widely by state.
The Advisory Committee on Immunization Practices recommends that, by age 17 years, adolescents complete three key immunizations: human papillomavirus (HPV), quadrivalent meningococcal conjugate (MenACWY), and Tdap.
Sara Poston, PharmD, senior director for health outcomes research at GlaxoSmithKline, said at a press conference during IDWeek, an annual scientific meeting on infectious diseases held virtually this year, that her team set out to determine how many teens were completing the vaccinations and how the number varied by state and by behavioral factors.
Completion of the vaccines means getting the HPV series (two doses for people aged 9-14 years at first vaccination or three doses for those aged 15 years or older at first vaccination), completion of the MenACWY series (two doses), and getting a Tdap vaccine (one dose).
Rhode Island has the highest rates
Some states are clearly doing better than others. Idaho had the lowest completion rate (11.3%; 95% confidence interval, 6.9%-18.0%), and Rhode Island had the highest (56.4%; 95% CI, 49.8%-62.8%).
In the 2018 National Immunization Survey–Teen (NIS-Teen), Rhode Island had the highest vaccination coverage rate in the nation for meningococcal vaccine (98.7%) and the second-highest coverage rate for Tdap (96.3%) for adolescents aged 13-17 years. Also in 2018, the state had the highest vaccination rates in the nation for the HPV series for both male and female adolescents 13-17 years of age (78.1%), well above the national average of 51.1%.
Researchers used information from the Centers for Disease Control and Prevention as well as 2015-2018 NIS-Teen data to estimate national and state-level completion rates by age 17. They then combined NIS-Teen data with public state-level data to evaluate what was driving or discouraging completion.
“The good news is, we found some variables that we consider actionable and can be used by states and local health departments to improve the rates,” Dr. Poston said.
Those include encouraging a health care visit at age 16 or 17, provider recommendations to families to get the HPV vaccine, and state-level mandates for the MenACWY vaccine.
Those who had a health care visit at 16 or 17 were more than twice as likely to complete their vaccines (odds ratio, 2.35; 95% CI, 1.80-3.07). Those for whom HPV vaccination had ever been recommended by a health care provider were more than three times as likely to complete their vaccinations (OR, 3.24; 95% CI, 2.76-3.80).
Other factors predictive of completing the vaccines included being Black or Hispanic and having Medicaid insurance.
At the state level, “living in a state with a mandate for the meningococcal ACWY vaccine in elementary or secondary school was also associated with likelihood of vaccination,” Dr. Poston said. Teens in states with mandates were 60% more likely to complete the vaccines than those in states without mandates. (OR, 1.6; 95% CI, 1.2-2.3)
Marielle Fricchione, MD, assistant professor of pediatric infectious diseases at Rush Medical College, Chicago said in an interview, “Teen vaccines are notoriously hard to get into kids because it’s hard to get them back into the office for second doses.”
She said that Illinois is one of the states with a two-dose mandate for MenACWY before entering 6th grade and 12th grade, which has kept vaccination coverage high.
Educating providers on how to recommend HPV vaccination is the biggest vaccine focus, she added.
Schedule next dose at first visit
One thing her department has found successful in HPV completion is scheduling the second dose while the teen is in the office for the first dose.
“Also, you have to recommend it just as strongly for boys as you do for girls, because oropharyngeal cancer is like an epidemic right now for men, and HPV-related oropharyngeal cancer is on an exponential rise,” Dr. Fricchione said.
According to the CDC, HPV is thought to cause 70% of oropharyngeal cancers in the United States.
Equipping providers with statistics on the effectiveness of HPV vaccination in preventing cancer can take away the uneasiness in talking about sexual transmission.
“That really seems to help them give a strong recommendation. It puts them in a data-driven position to talk about the vaccine,” she said. “Once you put that data in front of the providers, they’re floored.”
Research was funded by GlaxoSmithKline. Dr. Poston is employed by GlaxoSmithKline. Dr. Fricchione disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Only 30.6% of American adolescents complete three routinely recommended vaccinations, new research has found, but that number varies widely by state.
The Advisory Committee on Immunization Practices recommends that, by age 17 years, adolescents complete three key immunizations: human papillomavirus (HPV), quadrivalent meningococcal conjugate (MenACWY), and Tdap.
Sara Poston, PharmD, senior director for health outcomes research at GlaxoSmithKline, said at a press conference during IDWeek, an annual scientific meeting on infectious diseases held virtually this year, that her team set out to determine how many teens were completing the vaccinations and how the number varied by state and by behavioral factors.
Completion of the vaccines means getting the HPV series (two doses for people aged 9-14 years at first vaccination or three doses for those aged 15 years or older at first vaccination), completion of the MenACWY series (two doses), and getting a Tdap vaccine (one dose).
Rhode Island has the highest rates
Some states are clearly doing better than others. Idaho had the lowest completion rate (11.3%; 95% confidence interval, 6.9%-18.0%), and Rhode Island had the highest (56.4%; 95% CI, 49.8%-62.8%).
In the 2018 National Immunization Survey–Teen (NIS-Teen), Rhode Island had the highest vaccination coverage rate in the nation for meningococcal vaccine (98.7%) and the second-highest coverage rate for Tdap (96.3%) for adolescents aged 13-17 years. Also in 2018, the state had the highest vaccination rates in the nation for the HPV series for both male and female adolescents 13-17 years of age (78.1%), well above the national average of 51.1%.
Researchers used information from the Centers for Disease Control and Prevention as well as 2015-2018 NIS-Teen data to estimate national and state-level completion rates by age 17. They then combined NIS-Teen data with public state-level data to evaluate what was driving or discouraging completion.
“The good news is, we found some variables that we consider actionable and can be used by states and local health departments to improve the rates,” Dr. Poston said.
Those include encouraging a health care visit at age 16 or 17, provider recommendations to families to get the HPV vaccine, and state-level mandates for the MenACWY vaccine.
Those who had a health care visit at 16 or 17 were more than twice as likely to complete their vaccines (odds ratio, 2.35; 95% CI, 1.80-3.07). Those for whom HPV vaccination had ever been recommended by a health care provider were more than three times as likely to complete their vaccinations (OR, 3.24; 95% CI, 2.76-3.80).
Other factors predictive of completing the vaccines included being Black or Hispanic and having Medicaid insurance.
At the state level, “living in a state with a mandate for the meningococcal ACWY vaccine in elementary or secondary school was also associated with likelihood of vaccination,” Dr. Poston said. Teens in states with mandates were 60% more likely to complete the vaccines than those in states without mandates. (OR, 1.6; 95% CI, 1.2-2.3)
Marielle Fricchione, MD, assistant professor of pediatric infectious diseases at Rush Medical College, Chicago said in an interview, “Teen vaccines are notoriously hard to get into kids because it’s hard to get them back into the office for second doses.”
She said that Illinois is one of the states with a two-dose mandate for MenACWY before entering 6th grade and 12th grade, which has kept vaccination coverage high.
Educating providers on how to recommend HPV vaccination is the biggest vaccine focus, she added.
Schedule next dose at first visit
One thing her department has found successful in HPV completion is scheduling the second dose while the teen is in the office for the first dose.
“Also, you have to recommend it just as strongly for boys as you do for girls, because oropharyngeal cancer is like an epidemic right now for men, and HPV-related oropharyngeal cancer is on an exponential rise,” Dr. Fricchione said.
According to the CDC, HPV is thought to cause 70% of oropharyngeal cancers in the United States.
Equipping providers with statistics on the effectiveness of HPV vaccination in preventing cancer can take away the uneasiness in talking about sexual transmission.
“That really seems to help them give a strong recommendation. It puts them in a data-driven position to talk about the vaccine,” she said. “Once you put that data in front of the providers, they’re floored.”
Research was funded by GlaxoSmithKline. Dr. Poston is employed by GlaxoSmithKline. Dr. Fricchione disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM IDWEEK 2020
Survey: Acceptance of COVID-19 vaccine dips below 50%
Less than half of Americans now say that they would get a coronavirus vaccine if one became available, according to a survey conducted Oct. 8-10.
the lowest number since the weekly survey began at the end of February, digital media company Morning Consult reported.
Americans’ willingness to receive such a vaccine reached its high point, 72%, in early April but has been steadily dropping. “Overall willingness has hovered around 50% throughout September, fueled primarily by a sharp drop among Democrats since mid-August, around the time reports of White House interference at the Food and Drug Administration and other federal health agencies began to command more public attention,” Morning Consult noted.
Despite that drop, a majority of Democrats (55%) are still willing to get a COVID-19 vaccine, compared with 48% of Republicans and just 41% of independents. The willingness gap between the two parties was quite a bit wider in the previous poll, conducted Oct. 1-4: 60% of Democrats versus 48% for Republicans, the company said.
“Keeping with longstanding trends, the survey also shows women were less likely to say they’d seek a vaccine than men (42% to 55%), as were people with lower education levels and those who live in rural areas,” the news outlet added.
The latest poll results also show that 33% of respondents (43% of Republicans/25% of Democrats) are socializing in public places. The overall number was just 8% in mid-April but was up to 27% by mid-June. The proportion of all adults who believe in the effectiveness of face masks has been around 80% since April, but there is a significant gap between those who strongly approve of President Trump (66%) and those who strongly disapprove (95%), Morning Consult said.
Less than half of Americans now say that they would get a coronavirus vaccine if one became available, according to a survey conducted Oct. 8-10.
the lowest number since the weekly survey began at the end of February, digital media company Morning Consult reported.
Americans’ willingness to receive such a vaccine reached its high point, 72%, in early April but has been steadily dropping. “Overall willingness has hovered around 50% throughout September, fueled primarily by a sharp drop among Democrats since mid-August, around the time reports of White House interference at the Food and Drug Administration and other federal health agencies began to command more public attention,” Morning Consult noted.
Despite that drop, a majority of Democrats (55%) are still willing to get a COVID-19 vaccine, compared with 48% of Republicans and just 41% of independents. The willingness gap between the two parties was quite a bit wider in the previous poll, conducted Oct. 1-4: 60% of Democrats versus 48% for Republicans, the company said.
“Keeping with longstanding trends, the survey also shows women were less likely to say they’d seek a vaccine than men (42% to 55%), as were people with lower education levels and those who live in rural areas,” the news outlet added.
The latest poll results also show that 33% of respondents (43% of Republicans/25% of Democrats) are socializing in public places. The overall number was just 8% in mid-April but was up to 27% by mid-June. The proportion of all adults who believe in the effectiveness of face masks has been around 80% since April, but there is a significant gap between those who strongly approve of President Trump (66%) and those who strongly disapprove (95%), Morning Consult said.
Less than half of Americans now say that they would get a coronavirus vaccine if one became available, according to a survey conducted Oct. 8-10.
the lowest number since the weekly survey began at the end of February, digital media company Morning Consult reported.
Americans’ willingness to receive such a vaccine reached its high point, 72%, in early April but has been steadily dropping. “Overall willingness has hovered around 50% throughout September, fueled primarily by a sharp drop among Democrats since mid-August, around the time reports of White House interference at the Food and Drug Administration and other federal health agencies began to command more public attention,” Morning Consult noted.
Despite that drop, a majority of Democrats (55%) are still willing to get a COVID-19 vaccine, compared with 48% of Republicans and just 41% of independents. The willingness gap between the two parties was quite a bit wider in the previous poll, conducted Oct. 1-4: 60% of Democrats versus 48% for Republicans, the company said.
“Keeping with longstanding trends, the survey also shows women were less likely to say they’d seek a vaccine than men (42% to 55%), as were people with lower education levels and those who live in rural areas,” the news outlet added.
The latest poll results also show that 33% of respondents (43% of Republicans/25% of Democrats) are socializing in public places. The overall number was just 8% in mid-April but was up to 27% by mid-June. The proportion of all adults who believe in the effectiveness of face masks has been around 80% since April, but there is a significant gap between those who strongly approve of President Trump (66%) and those who strongly disapprove (95%), Morning Consult said.
Worldwide measles vaccination is flagging
After almost 2 decades of progress, the global state of measles vaccination and measles mortality is deteriorating.
One of the most serious concerns of measles infection is its long-term neurological complications, including the fatal subacute sclerosing panencephalitis (SSPE) and measles inclusion-body encephalitis (MIBE), which is usually seen in immune deficient children. Although some efforts are being made to determine which patients might be most vulnerable to these outcomes, and to treat them, the best approach is still prevention and vaccination, according to Banu Anlar, MD, of Hacettepe University, Ankara, Turkey, who spoke during a session at the 2020 CNS-ICNA Conjoint Meeting, held virtually this year.
Worldwide vaccination strategies have slipped in recent years, leading to upticks in measles cases and vaccination rates. As a result, in 2018 the World Health Organization postponed its goal of eliminating measles by 2020. Future eradication goals will likely need to be modified, according to Anaita Udwadia Hegde MD, a pediatric neurologist in Mumbai, India, who also presented at the session.
After measles deaths dropped 74% between 2000 and 2010, coinciding with widespread increases in vaccination, the WHO felt emboldened to deal the disease a knockout blow. In 2010, it held a Global Technical Consultation to determine the feasibility of an eradication campaign, which concluded it should be possible by 2020. Several characteristics of measles made that a reasonable goal: It is passed only among humans, with no known animal reservoir; natural infection grants lifelong immunity; there is only one serotype; the virus is genetically stable; the vaccine is safe and leads to 95%-97% seroconversion after two doses, which provides long-term protection against known genotypes; the disease is easily recognized and tested for; and it had been successfully eliminated already in some regions of the world.
As of 2017, analyses showed that the vaccination program saved the lives of about 1.5 million children. That was a cause for celebration, but the goal of eradication has remained elusive. Vaccination rates have trailed targets. In 2018, UNICEF and WHO estimated that 86% of children globally received the first measles vaccine, unchanged from 2010 and below the goal of 95%. Only 69% of children received the second dose, below the goal of 80%. Four countries in Europe lost their measles elimination status in 2018.
Other attempts to eradicate diseases have met with mixed results. The only full success was smallpox, eliminated in 1977. Similar efforts with polio, malaria, guinea worm, and now measles have all come up short. Those failures could complicate future efforts because global agencies and donors may be leery of past failures because of potential harm to their reputations, according to Dr. Hegde.
Such programs require sustained financial commitment and political support as well as local trust. Nevertheless, they must continue for ethical reasons, said Dr. Hegde, but also for economic ones: Every $1 spent on vaccination programs saves $58 in future costs in low- and middle-income countries. Missed childhood vaccination also results in future vulnerable teenagers and young adults, and these populations are much harder to reach and can drive large outbreaks.
Several factors are contributing to the global regression in vaccine coverage, according to Kristen Feemster, MD, MPH, a pediatric infectious disease physician and the global director of medical affairs at Merck. Globalization has enabled the spread of the disease. Most cases in the United States are imported by travelers to countries where the disease is endemic. “Measles can happen anywhere in the world, and when it does it can travel and spread. If you have an unvaccinated traveler who is exposed to measles abroad, they can return home and spread it to anyone else who is unvaccinated or not otherwise immune. When we see cases they’ve been sporadic, but if you return to a community where immunization rates are low, you have the potential for more sustained spread,” Dr. Feemster said during her presentation.
Why are so many travelers unvaccinated? A key reason is that vaccine hesitance is growing. Most affected individuals involved in outbreaks are unvaccinated, usually by choice rather than for medical reasons. Concerns continue over the measles vaccine and autism, growing out of the debunked studies of Andrew Wakefield. In one example, a Somali community in Minnesota experienced a higher than usual number of autism cases and parents sought reasons to explain it. They discovered the supposed connection between vaccination and autism, and Wakefield himself met with a group of them. The result was a drop in vaccination and, in 2011 and 2017, sizable measles outbreaks.
2020 has of course brought a fresh challenge to measles vaccine with the COVID-19 pandemic, which has reduced access to health care and shifted scientific and health care interest away from measles and other vaccine-preventable diseases. On the positive side, social distancing, mask wearing, and restricted movement are likely reducing exposure to measles, but reduced vaccination rates are likely to result in future outbreaks. “There’s been a significant decrease in rates for routine immunizations globally, so there’s a potential for yet another resurgence of measles and other vaccine-preventable diseases,” said Dr. Feemster.
Dr. Feemster is an employee of Merck. Dr. Anlar and Dr. Hegde did not disclose any relevant financial relationships.
After almost 2 decades of progress, the global state of measles vaccination and measles mortality is deteriorating.
One of the most serious concerns of measles infection is its long-term neurological complications, including the fatal subacute sclerosing panencephalitis (SSPE) and measles inclusion-body encephalitis (MIBE), which is usually seen in immune deficient children. Although some efforts are being made to determine which patients might be most vulnerable to these outcomes, and to treat them, the best approach is still prevention and vaccination, according to Banu Anlar, MD, of Hacettepe University, Ankara, Turkey, who spoke during a session at the 2020 CNS-ICNA Conjoint Meeting, held virtually this year.
Worldwide vaccination strategies have slipped in recent years, leading to upticks in measles cases and vaccination rates. As a result, in 2018 the World Health Organization postponed its goal of eliminating measles by 2020. Future eradication goals will likely need to be modified, according to Anaita Udwadia Hegde MD, a pediatric neurologist in Mumbai, India, who also presented at the session.
After measles deaths dropped 74% between 2000 and 2010, coinciding with widespread increases in vaccination, the WHO felt emboldened to deal the disease a knockout blow. In 2010, it held a Global Technical Consultation to determine the feasibility of an eradication campaign, which concluded it should be possible by 2020. Several characteristics of measles made that a reasonable goal: It is passed only among humans, with no known animal reservoir; natural infection grants lifelong immunity; there is only one serotype; the virus is genetically stable; the vaccine is safe and leads to 95%-97% seroconversion after two doses, which provides long-term protection against known genotypes; the disease is easily recognized and tested for; and it had been successfully eliminated already in some regions of the world.
As of 2017, analyses showed that the vaccination program saved the lives of about 1.5 million children. That was a cause for celebration, but the goal of eradication has remained elusive. Vaccination rates have trailed targets. In 2018, UNICEF and WHO estimated that 86% of children globally received the first measles vaccine, unchanged from 2010 and below the goal of 95%. Only 69% of children received the second dose, below the goal of 80%. Four countries in Europe lost their measles elimination status in 2018.
Other attempts to eradicate diseases have met with mixed results. The only full success was smallpox, eliminated in 1977. Similar efforts with polio, malaria, guinea worm, and now measles have all come up short. Those failures could complicate future efforts because global agencies and donors may be leery of past failures because of potential harm to their reputations, according to Dr. Hegde.
Such programs require sustained financial commitment and political support as well as local trust. Nevertheless, they must continue for ethical reasons, said Dr. Hegde, but also for economic ones: Every $1 spent on vaccination programs saves $58 in future costs in low- and middle-income countries. Missed childhood vaccination also results in future vulnerable teenagers and young adults, and these populations are much harder to reach and can drive large outbreaks.
Several factors are contributing to the global regression in vaccine coverage, according to Kristen Feemster, MD, MPH, a pediatric infectious disease physician and the global director of medical affairs at Merck. Globalization has enabled the spread of the disease. Most cases in the United States are imported by travelers to countries where the disease is endemic. “Measles can happen anywhere in the world, and when it does it can travel and spread. If you have an unvaccinated traveler who is exposed to measles abroad, they can return home and spread it to anyone else who is unvaccinated or not otherwise immune. When we see cases they’ve been sporadic, but if you return to a community where immunization rates are low, you have the potential for more sustained spread,” Dr. Feemster said during her presentation.
Why are so many travelers unvaccinated? A key reason is that vaccine hesitance is growing. Most affected individuals involved in outbreaks are unvaccinated, usually by choice rather than for medical reasons. Concerns continue over the measles vaccine and autism, growing out of the debunked studies of Andrew Wakefield. In one example, a Somali community in Minnesota experienced a higher than usual number of autism cases and parents sought reasons to explain it. They discovered the supposed connection between vaccination and autism, and Wakefield himself met with a group of them. The result was a drop in vaccination and, in 2011 and 2017, sizable measles outbreaks.
2020 has of course brought a fresh challenge to measles vaccine with the COVID-19 pandemic, which has reduced access to health care and shifted scientific and health care interest away from measles and other vaccine-preventable diseases. On the positive side, social distancing, mask wearing, and restricted movement are likely reducing exposure to measles, but reduced vaccination rates are likely to result in future outbreaks. “There’s been a significant decrease in rates for routine immunizations globally, so there’s a potential for yet another resurgence of measles and other vaccine-preventable diseases,” said Dr. Feemster.
Dr. Feemster is an employee of Merck. Dr. Anlar and Dr. Hegde did not disclose any relevant financial relationships.
After almost 2 decades of progress, the global state of measles vaccination and measles mortality is deteriorating.
One of the most serious concerns of measles infection is its long-term neurological complications, including the fatal subacute sclerosing panencephalitis (SSPE) and measles inclusion-body encephalitis (MIBE), which is usually seen in immune deficient children. Although some efforts are being made to determine which patients might be most vulnerable to these outcomes, and to treat them, the best approach is still prevention and vaccination, according to Banu Anlar, MD, of Hacettepe University, Ankara, Turkey, who spoke during a session at the 2020 CNS-ICNA Conjoint Meeting, held virtually this year.
Worldwide vaccination strategies have slipped in recent years, leading to upticks in measles cases and vaccination rates. As a result, in 2018 the World Health Organization postponed its goal of eliminating measles by 2020. Future eradication goals will likely need to be modified, according to Anaita Udwadia Hegde MD, a pediatric neurologist in Mumbai, India, who also presented at the session.
After measles deaths dropped 74% between 2000 and 2010, coinciding with widespread increases in vaccination, the WHO felt emboldened to deal the disease a knockout blow. In 2010, it held a Global Technical Consultation to determine the feasibility of an eradication campaign, which concluded it should be possible by 2020. Several characteristics of measles made that a reasonable goal: It is passed only among humans, with no known animal reservoir; natural infection grants lifelong immunity; there is only one serotype; the virus is genetically stable; the vaccine is safe and leads to 95%-97% seroconversion after two doses, which provides long-term protection against known genotypes; the disease is easily recognized and tested for; and it had been successfully eliminated already in some regions of the world.
As of 2017, analyses showed that the vaccination program saved the lives of about 1.5 million children. That was a cause for celebration, but the goal of eradication has remained elusive. Vaccination rates have trailed targets. In 2018, UNICEF and WHO estimated that 86% of children globally received the first measles vaccine, unchanged from 2010 and below the goal of 95%. Only 69% of children received the second dose, below the goal of 80%. Four countries in Europe lost their measles elimination status in 2018.
Other attempts to eradicate diseases have met with mixed results. The only full success was smallpox, eliminated in 1977. Similar efforts with polio, malaria, guinea worm, and now measles have all come up short. Those failures could complicate future efforts because global agencies and donors may be leery of past failures because of potential harm to their reputations, according to Dr. Hegde.
Such programs require sustained financial commitment and political support as well as local trust. Nevertheless, they must continue for ethical reasons, said Dr. Hegde, but also for economic ones: Every $1 spent on vaccination programs saves $58 in future costs in low- and middle-income countries. Missed childhood vaccination also results in future vulnerable teenagers and young adults, and these populations are much harder to reach and can drive large outbreaks.
Several factors are contributing to the global regression in vaccine coverage, according to Kristen Feemster, MD, MPH, a pediatric infectious disease physician and the global director of medical affairs at Merck. Globalization has enabled the spread of the disease. Most cases in the United States are imported by travelers to countries where the disease is endemic. “Measles can happen anywhere in the world, and when it does it can travel and spread. If you have an unvaccinated traveler who is exposed to measles abroad, they can return home and spread it to anyone else who is unvaccinated or not otherwise immune. When we see cases they’ve been sporadic, but if you return to a community where immunization rates are low, you have the potential for more sustained spread,” Dr. Feemster said during her presentation.
Why are so many travelers unvaccinated? A key reason is that vaccine hesitance is growing. Most affected individuals involved in outbreaks are unvaccinated, usually by choice rather than for medical reasons. Concerns continue over the measles vaccine and autism, growing out of the debunked studies of Andrew Wakefield. In one example, a Somali community in Minnesota experienced a higher than usual number of autism cases and parents sought reasons to explain it. They discovered the supposed connection between vaccination and autism, and Wakefield himself met with a group of them. The result was a drop in vaccination and, in 2011 and 2017, sizable measles outbreaks.
2020 has of course brought a fresh challenge to measles vaccine with the COVID-19 pandemic, which has reduced access to health care and shifted scientific and health care interest away from measles and other vaccine-preventable diseases. On the positive side, social distancing, mask wearing, and restricted movement are likely reducing exposure to measles, but reduced vaccination rates are likely to result in future outbreaks. “There’s been a significant decrease in rates for routine immunizations globally, so there’s a potential for yet another resurgence of measles and other vaccine-preventable diseases,” said Dr. Feemster.
Dr. Feemster is an employee of Merck. Dr. Anlar and Dr. Hegde did not disclose any relevant financial relationships.
FROM CNS-ICNA 2020
Fauci: Cautious optimism for COVID-19 vaccine by end of 2020
with distribution of first doses possible before the end of the year, according to Anthony S. Fauci, MD, director, National Institute of Allergy and Infectious Diseases, Bethesda, Md.
“Given the rate of infection that’s going on in this country, and the distribution of the clinical trial sites involving tens of thousands of volunteers, we project that we will have an answer as to whether or not we have a safe and effective vaccine by November or December,” Dr. Fauci said today in his virtual keynote address during the annual meeting of the American College of Chest Physicians.
“It may come earlier -- this month, in October,” he added in his remarks. “That is unlikely – it is more likely that we’ll have an answer in November and December.”
If that timing does come to pass, Dr. Fauci said, it’s possible that distribution of doses could start at the end of the year, continuing throughout the beginning and middle of 2021.
Although there are no guarantees, Dr. Fauci said he is “cautiously optimistic” regarding the timeline.
He said that his optimism is based in part on animal studies and phase 1 data that demonstrate robust neutralizing antibody responses to a vaccine that are equivalent to, if not greater than, natural infection with the SARS-CoV-2 virus that causes COVID-19.
Rapid development gives reason for hope
Ryan C. Maves, MD, FCCP, a critical care and infectious disease specialist at Naval Medical Center San Diego, said there is reason to be hopeful that a vaccine will be available by the end of the calendar year. He cautioned, however, that this timing is based on the assumption that one of the vaccines will be proven safe and effective very soon.
“We’re lucky to have multiple phase 3 trials using multiple vaccine technologies in different platforms,” Dr. Maves said in a panel discussion following Dr. Fauci’s remarks. “I think the odds are very high that one of them will be effective.”
“I’m hoping that multiple vaccines will be effective,” Dr. Maves added. “Then we’ll be in a good position of determining which is the best of several good options, as a society and as a world.”
COVID-19 vaccine development over the past year has been remarkably fast, especially given the previous record set by the mumps vaccine, which took about four years to go from initial steps to rollout, Dr. Maves noted.
Dr. Fauci said the federal government has taken a “strategic approach” to the COVID-19 vaccine that includes direct involvement in the research and development of six different vaccine candidates, five of which are now in phase 3 trials.
As part of that strategic approach, the study protocols are harmonized to have a common data and safety monitoring board, common primary and secondary endpoints, and an independent statistical group to determine correlates of protection, Dr. Fauci said.
Prioritizing COVID-19 vaccine distribution
Who gets COVID-19 vaccine first will be a challenge for governmental organizations as well as bioethicists, who have proposed different strategies for fairly prioritizing different groups for access.
Reaching communities of color will be an important consideration for prioritization, according to Dr. Maves, given the disproportionate burden of disease on Black and Hispanic individuals, among other such populations.
COVID-19–related hospitalization rates have been substantially higher in communities of color, Dr. Fauci said in his keynote address. Age-adjusted hospitalization rates for Hispanic/Latinx and Black populations are 375 to 368 per 100,000, respectively, compared with just 82 per 100,000 for White non-Hispanics, according to data from the Centers for Disease Control and Prevention.
Outreach to those communities should include building trust in those populations that they will benefit from a safe and effective vaccine, and making sure that the vaccine is available to those communities as quickly as possible, Dr. Maves said.
Dr. Fauci and Dr. Maves provided no disclosures related to their presentations.
with distribution of first doses possible before the end of the year, according to Anthony S. Fauci, MD, director, National Institute of Allergy and Infectious Diseases, Bethesda, Md.
“Given the rate of infection that’s going on in this country, and the distribution of the clinical trial sites involving tens of thousands of volunteers, we project that we will have an answer as to whether or not we have a safe and effective vaccine by November or December,” Dr. Fauci said today in his virtual keynote address during the annual meeting of the American College of Chest Physicians.
“It may come earlier -- this month, in October,” he added in his remarks. “That is unlikely – it is more likely that we’ll have an answer in November and December.”
If that timing does come to pass, Dr. Fauci said, it’s possible that distribution of doses could start at the end of the year, continuing throughout the beginning and middle of 2021.
Although there are no guarantees, Dr. Fauci said he is “cautiously optimistic” regarding the timeline.
He said that his optimism is based in part on animal studies and phase 1 data that demonstrate robust neutralizing antibody responses to a vaccine that are equivalent to, if not greater than, natural infection with the SARS-CoV-2 virus that causes COVID-19.
Rapid development gives reason for hope
Ryan C. Maves, MD, FCCP, a critical care and infectious disease specialist at Naval Medical Center San Diego, said there is reason to be hopeful that a vaccine will be available by the end of the calendar year. He cautioned, however, that this timing is based on the assumption that one of the vaccines will be proven safe and effective very soon.
“We’re lucky to have multiple phase 3 trials using multiple vaccine technologies in different platforms,” Dr. Maves said in a panel discussion following Dr. Fauci’s remarks. “I think the odds are very high that one of them will be effective.”
“I’m hoping that multiple vaccines will be effective,” Dr. Maves added. “Then we’ll be in a good position of determining which is the best of several good options, as a society and as a world.”
COVID-19 vaccine development over the past year has been remarkably fast, especially given the previous record set by the mumps vaccine, which took about four years to go from initial steps to rollout, Dr. Maves noted.
Dr. Fauci said the federal government has taken a “strategic approach” to the COVID-19 vaccine that includes direct involvement in the research and development of six different vaccine candidates, five of which are now in phase 3 trials.
As part of that strategic approach, the study protocols are harmonized to have a common data and safety monitoring board, common primary and secondary endpoints, and an independent statistical group to determine correlates of protection, Dr. Fauci said.
Prioritizing COVID-19 vaccine distribution
Who gets COVID-19 vaccine first will be a challenge for governmental organizations as well as bioethicists, who have proposed different strategies for fairly prioritizing different groups for access.
Reaching communities of color will be an important consideration for prioritization, according to Dr. Maves, given the disproportionate burden of disease on Black and Hispanic individuals, among other such populations.
COVID-19–related hospitalization rates have been substantially higher in communities of color, Dr. Fauci said in his keynote address. Age-adjusted hospitalization rates for Hispanic/Latinx and Black populations are 375 to 368 per 100,000, respectively, compared with just 82 per 100,000 for White non-Hispanics, according to data from the Centers for Disease Control and Prevention.
Outreach to those communities should include building trust in those populations that they will benefit from a safe and effective vaccine, and making sure that the vaccine is available to those communities as quickly as possible, Dr. Maves said.
Dr. Fauci and Dr. Maves provided no disclosures related to their presentations.
with distribution of first doses possible before the end of the year, according to Anthony S. Fauci, MD, director, National Institute of Allergy and Infectious Diseases, Bethesda, Md.
“Given the rate of infection that’s going on in this country, and the distribution of the clinical trial sites involving tens of thousands of volunteers, we project that we will have an answer as to whether or not we have a safe and effective vaccine by November or December,” Dr. Fauci said today in his virtual keynote address during the annual meeting of the American College of Chest Physicians.
“It may come earlier -- this month, in October,” he added in his remarks. “That is unlikely – it is more likely that we’ll have an answer in November and December.”
If that timing does come to pass, Dr. Fauci said, it’s possible that distribution of doses could start at the end of the year, continuing throughout the beginning and middle of 2021.
Although there are no guarantees, Dr. Fauci said he is “cautiously optimistic” regarding the timeline.
He said that his optimism is based in part on animal studies and phase 1 data that demonstrate robust neutralizing antibody responses to a vaccine that are equivalent to, if not greater than, natural infection with the SARS-CoV-2 virus that causes COVID-19.
Rapid development gives reason for hope
Ryan C. Maves, MD, FCCP, a critical care and infectious disease specialist at Naval Medical Center San Diego, said there is reason to be hopeful that a vaccine will be available by the end of the calendar year. He cautioned, however, that this timing is based on the assumption that one of the vaccines will be proven safe and effective very soon.
“We’re lucky to have multiple phase 3 trials using multiple vaccine technologies in different platforms,” Dr. Maves said in a panel discussion following Dr. Fauci’s remarks. “I think the odds are very high that one of them will be effective.”
“I’m hoping that multiple vaccines will be effective,” Dr. Maves added. “Then we’ll be in a good position of determining which is the best of several good options, as a society and as a world.”
COVID-19 vaccine development over the past year has been remarkably fast, especially given the previous record set by the mumps vaccine, which took about four years to go from initial steps to rollout, Dr. Maves noted.
Dr. Fauci said the federal government has taken a “strategic approach” to the COVID-19 vaccine that includes direct involvement in the research and development of six different vaccine candidates, five of which are now in phase 3 trials.
As part of that strategic approach, the study protocols are harmonized to have a common data and safety monitoring board, common primary and secondary endpoints, and an independent statistical group to determine correlates of protection, Dr. Fauci said.
Prioritizing COVID-19 vaccine distribution
Who gets COVID-19 vaccine first will be a challenge for governmental organizations as well as bioethicists, who have proposed different strategies for fairly prioritizing different groups for access.
Reaching communities of color will be an important consideration for prioritization, according to Dr. Maves, given the disproportionate burden of disease on Black and Hispanic individuals, among other such populations.
COVID-19–related hospitalization rates have been substantially higher in communities of color, Dr. Fauci said in his keynote address. Age-adjusted hospitalization rates for Hispanic/Latinx and Black populations are 375 to 368 per 100,000, respectively, compared with just 82 per 100,000 for White non-Hispanics, according to data from the Centers for Disease Control and Prevention.
Outreach to those communities should include building trust in those populations that they will benefit from a safe and effective vaccine, and making sure that the vaccine is available to those communities as quickly as possible, Dr. Maves said.
Dr. Fauci and Dr. Maves provided no disclosures related to their presentations.
FROM CHEST 2020
Flu vaccine significantly cuts pediatric hospitalizations
Unlike previous studies focused on vaccine effectiveness (VE) in ambulatory care office visits, Angela P. Campbell, MD, MPH, and associates have uncovered evidence of the overall benefit influenza vaccines play in reducing hospitalizations and emergency department visits in pediatric influenza patients.
“Our data provide important VE estimates against severe influenza in children,” the researchers noted in Pediatrics, adding that the findings “provide important evidence supporting the annual recommendation that all children 6 months and older should receive influenza vaccination.”
Dr. Campbell and colleagues collected ongoing surveillance data from the New Vaccine Surveillance Network (NVSN), which is a network of pediatric hospitals across seven cities, including Kansas City, Mo.; Rochester, N.Y.; Cincinnati; Pittsburgh; Nashville, Tenn.; Houston; and Seattle. The influenza season encompassed the period Nov. 7, 2018 to June 21, 2019.
A total of 2,748 hospitalized children and 2,676 children who had completed ED visits that did not lead to hospitalization were included. Once those under 6 months were excluded, 1,792 hospitalized children were included in the VE analysis; of these, 226 (13%) tested positive for influenza infection, including 211 (93%) with influenza A viruses and 15 (7%) with influenza B viruses. Fully 1,611 of the patients (90%), had verified vaccine status, while 181 (10%) had solely parental reported vaccine status. The researchers reported 88 (5%) of the patients received mechanical ventilation and 7 (<1%) died.
Most noteworthy, They further estimated a significant reduction in hospitalizations linked to A(H3N2) and A(H1N1)pdm09 viruses, even in the presence of circulating A(H3N2) viruses that differed from the A(H3N2) vaccine component.
Studies from other countries during the same time period showed that while “significant protection against influenza-associated ambulatory care visits and hospitalizations among children infected with A(H1N1)pdm09 viruses” was observed, the same could not be said for protection against A(H3N2) viruses, which varied among pediatric outpatients in the United States (24%), in England (17% outpatient; 31% inpatient), Europe (46%), and Canada (48%). They explained that such variation in vaccine protection is multifactorial, and includes virus-, host-, and environment-related factors. They also noted that regional variations in circulating viruses, host factors including age, imprinting, and previous vaccination could explain the study’s finding of vaccine protection against both A(H1N1)pdm09 and A(H3N2) viruses.
When comparing VE estimates between ED visits and hospitalizations, the researchers observed one significant difference, that “hospitalized children likely represent more medically complex patients, with 58% having underlying medical conditions and 38% reporting at lease one hospitalization in the past year, compared with 28% and 14% respectively, among ED participants.”
Strengths of the study included the prospective multisite enrollment that provided data across diverse locations and representation from pediatric hospitalizations and ED care, which were not previously strongly represented in the literature. The single-season study with small sample size was considered a limitation, as was the inability to evaluate full and partial vaccine status. Vaccine data also were limited for many of the ED patients observed.
Dr. Campbell and colleagues did caution that while they consider their test-negative design optimal for evaluating both hospitalized and ED patients, they feel their results should not be “interpreted as VE against influenza-associated ambulatory care visits or infections that are not medically attended.”
In a separate interview, Michael E. Pichichero, MD, director of the Rochester General Hospital Research Institute and a clinical professor of pediatrics at the University of Rochester (N.Y.), observed: “There are really no surprises here. A well done contemporary study confirms again the benefits of annual influenza vaccinations for children. Viral coinfections involving SARS-CoV-2 and influenza have been reported from Australia to cause heightened illnesses. That observation provides further impetus for parents to have their children receive influenza vaccinations.”
The researchers cited multiple sources of financial support for their ongoing work, including Sanofi, Quidel, Moderna, Karius, GlaxoSmithKline, Merck, AstraZeneca, and Pfizer. Funding for this study was supported by the Centers for Disease Control and Prevention. Dr. Pichichero said he had no relevant financial disclosures.
SOURCE: Campbell AP et al. Pediatrics. 2020. doi: 10.1542/peds.2020-1368.
Unlike previous studies focused on vaccine effectiveness (VE) in ambulatory care office visits, Angela P. Campbell, MD, MPH, and associates have uncovered evidence of the overall benefit influenza vaccines play in reducing hospitalizations and emergency department visits in pediatric influenza patients.
“Our data provide important VE estimates against severe influenza in children,” the researchers noted in Pediatrics, adding that the findings “provide important evidence supporting the annual recommendation that all children 6 months and older should receive influenza vaccination.”
Dr. Campbell and colleagues collected ongoing surveillance data from the New Vaccine Surveillance Network (NVSN), which is a network of pediatric hospitals across seven cities, including Kansas City, Mo.; Rochester, N.Y.; Cincinnati; Pittsburgh; Nashville, Tenn.; Houston; and Seattle. The influenza season encompassed the period Nov. 7, 2018 to June 21, 2019.
A total of 2,748 hospitalized children and 2,676 children who had completed ED visits that did not lead to hospitalization were included. Once those under 6 months were excluded, 1,792 hospitalized children were included in the VE analysis; of these, 226 (13%) tested positive for influenza infection, including 211 (93%) with influenza A viruses and 15 (7%) with influenza B viruses. Fully 1,611 of the patients (90%), had verified vaccine status, while 181 (10%) had solely parental reported vaccine status. The researchers reported 88 (5%) of the patients received mechanical ventilation and 7 (<1%) died.
Most noteworthy, They further estimated a significant reduction in hospitalizations linked to A(H3N2) and A(H1N1)pdm09 viruses, even in the presence of circulating A(H3N2) viruses that differed from the A(H3N2) vaccine component.
Studies from other countries during the same time period showed that while “significant protection against influenza-associated ambulatory care visits and hospitalizations among children infected with A(H1N1)pdm09 viruses” was observed, the same could not be said for protection against A(H3N2) viruses, which varied among pediatric outpatients in the United States (24%), in England (17% outpatient; 31% inpatient), Europe (46%), and Canada (48%). They explained that such variation in vaccine protection is multifactorial, and includes virus-, host-, and environment-related factors. They also noted that regional variations in circulating viruses, host factors including age, imprinting, and previous vaccination could explain the study’s finding of vaccine protection against both A(H1N1)pdm09 and A(H3N2) viruses.
When comparing VE estimates between ED visits and hospitalizations, the researchers observed one significant difference, that “hospitalized children likely represent more medically complex patients, with 58% having underlying medical conditions and 38% reporting at lease one hospitalization in the past year, compared with 28% and 14% respectively, among ED participants.”
Strengths of the study included the prospective multisite enrollment that provided data across diverse locations and representation from pediatric hospitalizations and ED care, which were not previously strongly represented in the literature. The single-season study with small sample size was considered a limitation, as was the inability to evaluate full and partial vaccine status. Vaccine data also were limited for many of the ED patients observed.
Dr. Campbell and colleagues did caution that while they consider their test-negative design optimal for evaluating both hospitalized and ED patients, they feel their results should not be “interpreted as VE against influenza-associated ambulatory care visits or infections that are not medically attended.”
In a separate interview, Michael E. Pichichero, MD, director of the Rochester General Hospital Research Institute and a clinical professor of pediatrics at the University of Rochester (N.Y.), observed: “There are really no surprises here. A well done contemporary study confirms again the benefits of annual influenza vaccinations for children. Viral coinfections involving SARS-CoV-2 and influenza have been reported from Australia to cause heightened illnesses. That observation provides further impetus for parents to have their children receive influenza vaccinations.”
The researchers cited multiple sources of financial support for their ongoing work, including Sanofi, Quidel, Moderna, Karius, GlaxoSmithKline, Merck, AstraZeneca, and Pfizer. Funding for this study was supported by the Centers for Disease Control and Prevention. Dr. Pichichero said he had no relevant financial disclosures.
SOURCE: Campbell AP et al. Pediatrics. 2020. doi: 10.1542/peds.2020-1368.
Unlike previous studies focused on vaccine effectiveness (VE) in ambulatory care office visits, Angela P. Campbell, MD, MPH, and associates have uncovered evidence of the overall benefit influenza vaccines play in reducing hospitalizations and emergency department visits in pediatric influenza patients.
“Our data provide important VE estimates against severe influenza in children,” the researchers noted in Pediatrics, adding that the findings “provide important evidence supporting the annual recommendation that all children 6 months and older should receive influenza vaccination.”
Dr. Campbell and colleagues collected ongoing surveillance data from the New Vaccine Surveillance Network (NVSN), which is a network of pediatric hospitals across seven cities, including Kansas City, Mo.; Rochester, N.Y.; Cincinnati; Pittsburgh; Nashville, Tenn.; Houston; and Seattle. The influenza season encompassed the period Nov. 7, 2018 to June 21, 2019.
A total of 2,748 hospitalized children and 2,676 children who had completed ED visits that did not lead to hospitalization were included. Once those under 6 months were excluded, 1,792 hospitalized children were included in the VE analysis; of these, 226 (13%) tested positive for influenza infection, including 211 (93%) with influenza A viruses and 15 (7%) with influenza B viruses. Fully 1,611 of the patients (90%), had verified vaccine status, while 181 (10%) had solely parental reported vaccine status. The researchers reported 88 (5%) of the patients received mechanical ventilation and 7 (<1%) died.
Most noteworthy, They further estimated a significant reduction in hospitalizations linked to A(H3N2) and A(H1N1)pdm09 viruses, even in the presence of circulating A(H3N2) viruses that differed from the A(H3N2) vaccine component.
Studies from other countries during the same time period showed that while “significant protection against influenza-associated ambulatory care visits and hospitalizations among children infected with A(H1N1)pdm09 viruses” was observed, the same could not be said for protection against A(H3N2) viruses, which varied among pediatric outpatients in the United States (24%), in England (17% outpatient; 31% inpatient), Europe (46%), and Canada (48%). They explained that such variation in vaccine protection is multifactorial, and includes virus-, host-, and environment-related factors. They also noted that regional variations in circulating viruses, host factors including age, imprinting, and previous vaccination could explain the study’s finding of vaccine protection against both A(H1N1)pdm09 and A(H3N2) viruses.
When comparing VE estimates between ED visits and hospitalizations, the researchers observed one significant difference, that “hospitalized children likely represent more medically complex patients, with 58% having underlying medical conditions and 38% reporting at lease one hospitalization in the past year, compared with 28% and 14% respectively, among ED participants.”
Strengths of the study included the prospective multisite enrollment that provided data across diverse locations and representation from pediatric hospitalizations and ED care, which were not previously strongly represented in the literature. The single-season study with small sample size was considered a limitation, as was the inability to evaluate full and partial vaccine status. Vaccine data also were limited for many of the ED patients observed.
Dr. Campbell and colleagues did caution that while they consider their test-negative design optimal for evaluating both hospitalized and ED patients, they feel their results should not be “interpreted as VE against influenza-associated ambulatory care visits or infections that are not medically attended.”
In a separate interview, Michael E. Pichichero, MD, director of the Rochester General Hospital Research Institute and a clinical professor of pediatrics at the University of Rochester (N.Y.), observed: “There are really no surprises here. A well done contemporary study confirms again the benefits of annual influenza vaccinations for children. Viral coinfections involving SARS-CoV-2 and influenza have been reported from Australia to cause heightened illnesses. That observation provides further impetus for parents to have their children receive influenza vaccinations.”
The researchers cited multiple sources of financial support for their ongoing work, including Sanofi, Quidel, Moderna, Karius, GlaxoSmithKline, Merck, AstraZeneca, and Pfizer. Funding for this study was supported by the Centers for Disease Control and Prevention. Dr. Pichichero said he had no relevant financial disclosures.
SOURCE: Campbell AP et al. Pediatrics. 2020. doi: 10.1542/peds.2020-1368.
FROM PEDIATRICS
HPV vaccination remains below Healthy People goals despite increases
and vary widely across states based on data from a nested cohort study including more than 7 million children.
“Understanding regional and temporal variations in HPV vaccination coverage may help improve HPV vaccination uptake by informing public health policy,” Szu-Ta Chen, MD, of Harvard University, Boston, and colleagues wrote in Pediatrics.
To identify trends in one-dose and two-dose human papillomavirus (HPV) vaccination coverage, the researchers reviewed data from the MarketScan health care database between January 2003 and December 2017 that included 7,837,480 children and 19,843,737 person-years. The children were followed starting at age 9, when HPV vaccination could begin, and ending at one of the following: the first or second vaccination, insurance disenrollment, December 2017, or the end of the year in which they turned 17.
Overall, the proportion of 15-year-old girls and boys with at least a one-dose HPV vaccination increased from 38% and 5%, respectively, in 2011 to 57% and 51%, respectively, in 2017. The comparable proportions of girls and boys with at least a two-dose vaccination increased from 30% and 2%, respectively, in 2011 to 46% and 39%, respectively, in 2017.
Coverage lacks consistency across states
However, the vaccination coverage varied widely across states; two-dose HPV vaccination coverage ranged from 80% of girls in the District of Columbia to 15% of boys in Mississippi. In general, states with more HPV vaccine interventions had higher levels of vaccination, the researchers noted.
Legislation to improve vaccination education showed the strongest association with coverage; an 8.8% increase in coverage for girls and an 8.7% increase for boys. Pediatrician availability also was a factor associated with a 1.1% increase in coverage estimated for every pediatrician per 10,000 children.
Cumulative HPV vaccinations seen among children continuously enrolled in the study were similar to the primary analysis, the Dr. Chen and associates said. “After the initial HPV vaccination, 87% of girls and 82% of boys received a second dose by age 17 in the most recent cohorts.”
However, the HPV vaccination coverage remains below the Healthy People 2020 goal of 80% of children vaccinated by age 15 years, the researchers said. Barriers to vaccination may include a lack of routine clinical encounters in adolescents aged 11-17 years. HPV vaccination coverage was higher in urban populations, compared with rural, which may be related to a lack of providers in rural areas.
“Thus, measures beyond recommending routine vaccination at annual check-ups might be necessary to attain sufficient HPV vaccine coverage, and the optimal strategy may differ by state characteristics,” they wrote.
The study findings were limited by several factors including the use of data from only commercially-insured children and lack of data on vaccines received outside of insurance, the researchers noted.
However, the results were strengthened by the large, population-based sample, and support the need for increased efforts in HPV vaccination. “Most states will not achieve the Healthy People 2020 goal of 80% coverage with at least two HPV vaccine doses by 2020,” Dr. Chen and associates concluded.
Vaccination goals are possible with effort in the right places
The fact of below-target vaccination for HPV in the United States may be old news, but the current study offers new insights on HPV uptake, Amanda F. Dempsey, MD, PhD, of the University of Colorado at Denver, in Aurora, wrote in an accompanying editorial.
“A unique feature of this study is the ability of its researchers to study individuals over time, particularly at a national scope,” which yielded two key messages, she said.
The longitudinal examination of vaccination levels among birth cohorts showed that similar vaccination levels were achieved more quickly each year.
“For example, among the birth cohort from the year 2000, representing 17-year-olds at the time data were abstracted for the study, 40% vaccination coverage was achieved when this group was 14 years old. In contrast, among the birth cohort from the year 2005, representing 12-year-olds at the time of data abstraction, 40% vaccination coverage was reached at the age of 12,” Dr. Dempsey explained.
In addition, the study design allowed the researchers to model future vaccine coverage based on current trends, said Dr. Dempsey. “The authors estimate that, by the year 2022, the 2012 birth cohort will have reached 80% coverage for the first dose in the HPV vaccine series.”
Dr. Dempsey said she was surprised that the models did not support the hypothesis that school mandates for vaccination would increase coverage; however, there were few states in this category.
Although the findings were limited by the lack of data on uninsured children and those insured by Medicaid, the state-by-state results show that the achievement of national vaccination goals is possible, Dr. Dempsey said. In addition, the findings “warrant close consideration by policy makers and the medical community at large regarding vaccination policies and workforce,” she emphasized.The study received no outside funding. Dr. Chen had no financial conflicts to disclose. Several coauthors reported research grants to their institutions from pharmaceutical companies or being consultants to such companies. Dr. Dempsey disclosed serving on the advisory boards for Merck, Pfizer, and Sanofi Pasteur.
SOURCE: Chen S-T et al. Pediatrics. 2020 Sep 14. doi: 10.1542/peds.2019-3557.
and vary widely across states based on data from a nested cohort study including more than 7 million children.
“Understanding regional and temporal variations in HPV vaccination coverage may help improve HPV vaccination uptake by informing public health policy,” Szu-Ta Chen, MD, of Harvard University, Boston, and colleagues wrote in Pediatrics.
To identify trends in one-dose and two-dose human papillomavirus (HPV) vaccination coverage, the researchers reviewed data from the MarketScan health care database between January 2003 and December 2017 that included 7,837,480 children and 19,843,737 person-years. The children were followed starting at age 9, when HPV vaccination could begin, and ending at one of the following: the first or second vaccination, insurance disenrollment, December 2017, or the end of the year in which they turned 17.
Overall, the proportion of 15-year-old girls and boys with at least a one-dose HPV vaccination increased from 38% and 5%, respectively, in 2011 to 57% and 51%, respectively, in 2017. The comparable proportions of girls and boys with at least a two-dose vaccination increased from 30% and 2%, respectively, in 2011 to 46% and 39%, respectively, in 2017.
Coverage lacks consistency across states
However, the vaccination coverage varied widely across states; two-dose HPV vaccination coverage ranged from 80% of girls in the District of Columbia to 15% of boys in Mississippi. In general, states with more HPV vaccine interventions had higher levels of vaccination, the researchers noted.
Legislation to improve vaccination education showed the strongest association with coverage; an 8.8% increase in coverage for girls and an 8.7% increase for boys. Pediatrician availability also was a factor associated with a 1.1% increase in coverage estimated for every pediatrician per 10,000 children.
Cumulative HPV vaccinations seen among children continuously enrolled in the study were similar to the primary analysis, the Dr. Chen and associates said. “After the initial HPV vaccination, 87% of girls and 82% of boys received a second dose by age 17 in the most recent cohorts.”
However, the HPV vaccination coverage remains below the Healthy People 2020 goal of 80% of children vaccinated by age 15 years, the researchers said. Barriers to vaccination may include a lack of routine clinical encounters in adolescents aged 11-17 years. HPV vaccination coverage was higher in urban populations, compared with rural, which may be related to a lack of providers in rural areas.
“Thus, measures beyond recommending routine vaccination at annual check-ups might be necessary to attain sufficient HPV vaccine coverage, and the optimal strategy may differ by state characteristics,” they wrote.
The study findings were limited by several factors including the use of data from only commercially-insured children and lack of data on vaccines received outside of insurance, the researchers noted.
However, the results were strengthened by the large, population-based sample, and support the need for increased efforts in HPV vaccination. “Most states will not achieve the Healthy People 2020 goal of 80% coverage with at least two HPV vaccine doses by 2020,” Dr. Chen and associates concluded.
Vaccination goals are possible with effort in the right places
The fact of below-target vaccination for HPV in the United States may be old news, but the current study offers new insights on HPV uptake, Amanda F. Dempsey, MD, PhD, of the University of Colorado at Denver, in Aurora, wrote in an accompanying editorial.
“A unique feature of this study is the ability of its researchers to study individuals over time, particularly at a national scope,” which yielded two key messages, she said.
The longitudinal examination of vaccination levels among birth cohorts showed that similar vaccination levels were achieved more quickly each year.
“For example, among the birth cohort from the year 2000, representing 17-year-olds at the time data were abstracted for the study, 40% vaccination coverage was achieved when this group was 14 years old. In contrast, among the birth cohort from the year 2005, representing 12-year-olds at the time of data abstraction, 40% vaccination coverage was reached at the age of 12,” Dr. Dempsey explained.
In addition, the study design allowed the researchers to model future vaccine coverage based on current trends, said Dr. Dempsey. “The authors estimate that, by the year 2022, the 2012 birth cohort will have reached 80% coverage for the first dose in the HPV vaccine series.”
Dr. Dempsey said she was surprised that the models did not support the hypothesis that school mandates for vaccination would increase coverage; however, there were few states in this category.
Although the findings were limited by the lack of data on uninsured children and those insured by Medicaid, the state-by-state results show that the achievement of national vaccination goals is possible, Dr. Dempsey said. In addition, the findings “warrant close consideration by policy makers and the medical community at large regarding vaccination policies and workforce,” she emphasized.The study received no outside funding. Dr. Chen had no financial conflicts to disclose. Several coauthors reported research grants to their institutions from pharmaceutical companies or being consultants to such companies. Dr. Dempsey disclosed serving on the advisory boards for Merck, Pfizer, and Sanofi Pasteur.
SOURCE: Chen S-T et al. Pediatrics. 2020 Sep 14. doi: 10.1542/peds.2019-3557.
and vary widely across states based on data from a nested cohort study including more than 7 million children.
“Understanding regional and temporal variations in HPV vaccination coverage may help improve HPV vaccination uptake by informing public health policy,” Szu-Ta Chen, MD, of Harvard University, Boston, and colleagues wrote in Pediatrics.
To identify trends in one-dose and two-dose human papillomavirus (HPV) vaccination coverage, the researchers reviewed data from the MarketScan health care database between January 2003 and December 2017 that included 7,837,480 children and 19,843,737 person-years. The children were followed starting at age 9, when HPV vaccination could begin, and ending at one of the following: the first or second vaccination, insurance disenrollment, December 2017, or the end of the year in which they turned 17.
Overall, the proportion of 15-year-old girls and boys with at least a one-dose HPV vaccination increased from 38% and 5%, respectively, in 2011 to 57% and 51%, respectively, in 2017. The comparable proportions of girls and boys with at least a two-dose vaccination increased from 30% and 2%, respectively, in 2011 to 46% and 39%, respectively, in 2017.
Coverage lacks consistency across states
However, the vaccination coverage varied widely across states; two-dose HPV vaccination coverage ranged from 80% of girls in the District of Columbia to 15% of boys in Mississippi. In general, states with more HPV vaccine interventions had higher levels of vaccination, the researchers noted.
Legislation to improve vaccination education showed the strongest association with coverage; an 8.8% increase in coverage for girls and an 8.7% increase for boys. Pediatrician availability also was a factor associated with a 1.1% increase in coverage estimated for every pediatrician per 10,000 children.
Cumulative HPV vaccinations seen among children continuously enrolled in the study were similar to the primary analysis, the Dr. Chen and associates said. “After the initial HPV vaccination, 87% of girls and 82% of boys received a second dose by age 17 in the most recent cohorts.”
However, the HPV vaccination coverage remains below the Healthy People 2020 goal of 80% of children vaccinated by age 15 years, the researchers said. Barriers to vaccination may include a lack of routine clinical encounters in adolescents aged 11-17 years. HPV vaccination coverage was higher in urban populations, compared with rural, which may be related to a lack of providers in rural areas.
“Thus, measures beyond recommending routine vaccination at annual check-ups might be necessary to attain sufficient HPV vaccine coverage, and the optimal strategy may differ by state characteristics,” they wrote.
The study findings were limited by several factors including the use of data from only commercially-insured children and lack of data on vaccines received outside of insurance, the researchers noted.
However, the results were strengthened by the large, population-based sample, and support the need for increased efforts in HPV vaccination. “Most states will not achieve the Healthy People 2020 goal of 80% coverage with at least two HPV vaccine doses by 2020,” Dr. Chen and associates concluded.
Vaccination goals are possible with effort in the right places
The fact of below-target vaccination for HPV in the United States may be old news, but the current study offers new insights on HPV uptake, Amanda F. Dempsey, MD, PhD, of the University of Colorado at Denver, in Aurora, wrote in an accompanying editorial.
“A unique feature of this study is the ability of its researchers to study individuals over time, particularly at a national scope,” which yielded two key messages, she said.
The longitudinal examination of vaccination levels among birth cohorts showed that similar vaccination levels were achieved more quickly each year.
“For example, among the birth cohort from the year 2000, representing 17-year-olds at the time data were abstracted for the study, 40% vaccination coverage was achieved when this group was 14 years old. In contrast, among the birth cohort from the year 2005, representing 12-year-olds at the time of data abstraction, 40% vaccination coverage was reached at the age of 12,” Dr. Dempsey explained.
In addition, the study design allowed the researchers to model future vaccine coverage based on current trends, said Dr. Dempsey. “The authors estimate that, by the year 2022, the 2012 birth cohort will have reached 80% coverage for the first dose in the HPV vaccine series.”
Dr. Dempsey said she was surprised that the models did not support the hypothesis that school mandates for vaccination would increase coverage; however, there were few states in this category.
Although the findings were limited by the lack of data on uninsured children and those insured by Medicaid, the state-by-state results show that the achievement of national vaccination goals is possible, Dr. Dempsey said. In addition, the findings “warrant close consideration by policy makers and the medical community at large regarding vaccination policies and workforce,” she emphasized.The study received no outside funding. Dr. Chen had no financial conflicts to disclose. Several coauthors reported research grants to their institutions from pharmaceutical companies or being consultants to such companies. Dr. Dempsey disclosed serving on the advisory boards for Merck, Pfizer, and Sanofi Pasteur.
SOURCE: Chen S-T et al. Pediatrics. 2020 Sep 14. doi: 10.1542/peds.2019-3557.
FROM PEDIATRICS
Prospects and challenges for the upcoming influenza season
The 2020-2021 influenza season is shaping up to be challenging. Its likely concurrence with the ongoing severe acute respiratory syndrome-coronavirus 2 (SARS-coV-2) pandemic (COVID-19) will pose diagnostic and therapeutic dilemmas and could overload the hospital system. But there could also be potential synergies in preventing morbidity and mortality from each disease.
A consistent pattern overthe past few influenza seasons
During the 2019-2020 flu season, there were an estimated 410,000 to 740,000 hospitalizations and 24,000 to 62,000 deaths attributed to influenza.1 As seen in FIGURE 1, office visits for influenza-like illness (ILI) began to increase in late November and early December in each of the last 3 years (2017-2018, 2018-2019, 2019-2020) and stayed elevated above baseline for about 4 months each season.1
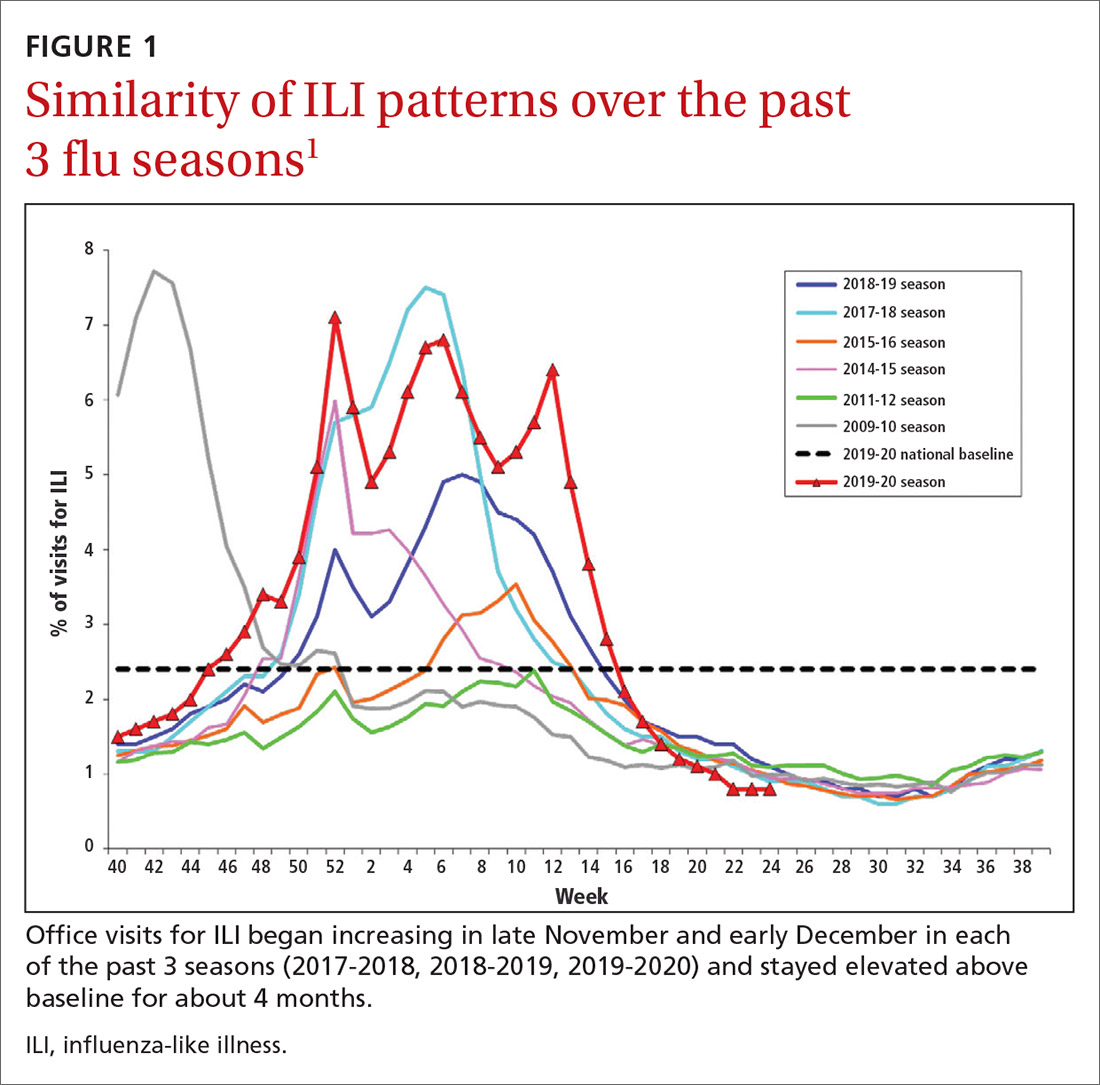
The effectiveness of influenza vaccine during the 2019-2020 season is being estimated using the US Flu Vaccine Effectiveness Network, which has close to 9000 enrollees. Overall, it appears the vaccine was 39% effective against medically attended influenza, with a higher effectiveness against influenza B (44%) than against A/H1N1 (31%). Effectiveness against influenza B was similar in all age groups, but effectiveness against A/H1N1 was highest for those ages 50 to 64 years (45%) and lowest for those ages 6 months through 8 years (22%), although 95% confidence intervals overlapped for all age groups (FIGURE 2). These preliminary effectiveness rates were presented at the summer meeting of the Advisory Committee on Immunization Practices (ACIP).1
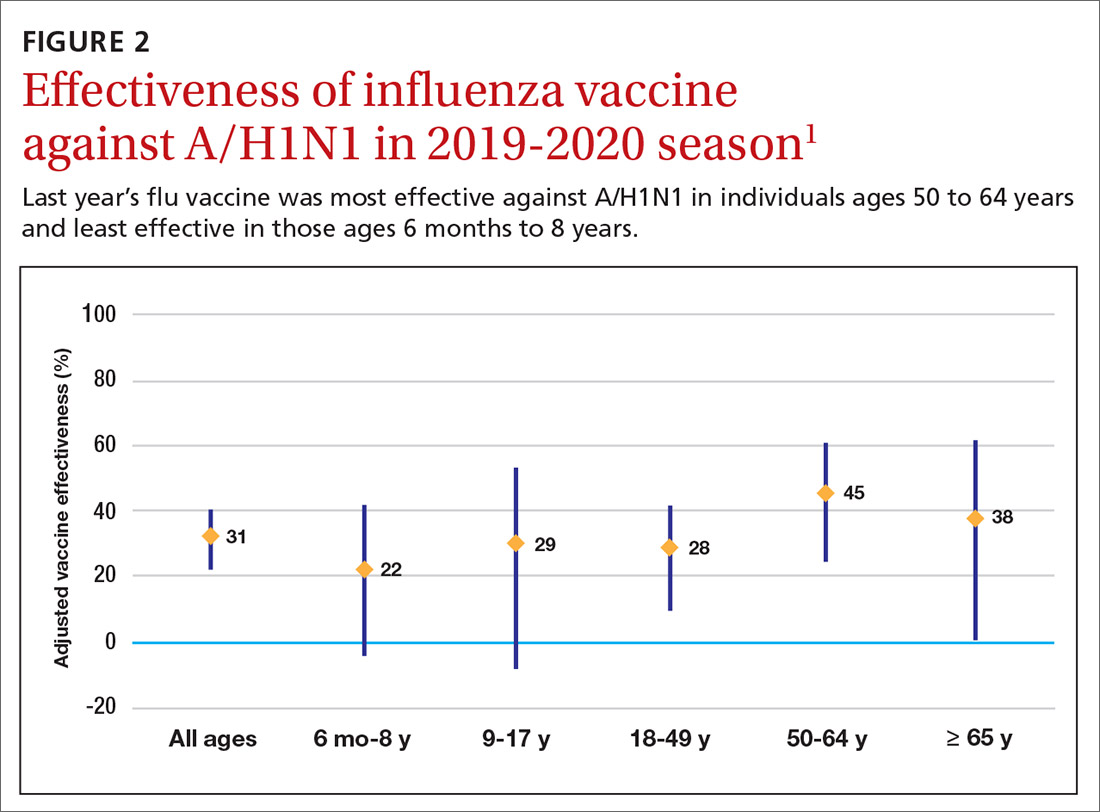
Influenza vaccine safety data for 2019-2020 were based on the Vaccine Adverse Event Reporting System (VAERS), a passive surveillance system, and on the Vaccine Safety Datalink (VSD) system, an active surveillance system involving close to 6 million doses administered at VSD sites. No safety concerns were identified for any of the different vaccine types. Both the VAERS and VSD surveillance systems have been described in more detail in a previous Practice Alert.2
Recommendations for 2020-2021
The composition of the influenza vaccines for this year’s flu season will be different for 3 of the 4 antigens: A/H1N1, A/H2N2 and B/Victoria.3 The antigens included in the influenza vaccines each year are decided on in the spring, based on surveillance of circulating strains around the world. The effectiveness of the vaccine each year largely depends on how well the strains included in the vaccine match those circulating in the United States during the influenza season.
The main immunization recommendation for preventing morbidity and mortality from influenza has not changed: All individuals ages 6 months and older without a contraindication should receive an influenza vaccine.4 The Centers for Disease Control and Prevention (CDC) recommends that patients receive the vaccine by the end of October.4 This includes the second dose for those children younger than 9 years who need 2 doses—ie, those who have received fewer than 2 doses of influenza vaccine prior to July 2020. Vaccination should continue through the end of the season for anyone who has not received a 2020-2021 influenza vaccine.
Two new influenza vaccine products are available for use in those ages 65 years and older: Fluzone high-dose quadrivalent and Fluad Quadrivalent (adjuvanted).4 Both of these products were available last year as trivalent options. Currently no specific vaccine product is listed as preferred by ACIP for those ages 65 and older.
Continue to: New vaccine contraindications
New vaccine contraindications. Four medical conditions have been added to the list of contraindications for quadrivalent live, attenuated influenza vaccine (LAIV4): cochlear implant, cerebrospinal fluid leak, asplenia (anatomic and functional), and sickle cell anemia.4 In addition, those who receive LAIV4 should not be prescribed an influenza antiviral until 2 weeks after receiving the vaccine. And the vaccine should not be administered for 48 hours after receipt of oseltamivir or zanamivir, 5 days after peramivir, and 17 days after baloxavir marboxil.4 This is to prevent possible antiviral inactivation of the live attenuated influenza viruses in the vaccine.
For those who have a history of severe allergic reaction to eggs, there are now 2 egg-free options: cell-culture-based inactivated vaccine (ccIIV4) and recombinant influenza vaccine (RIV4).3,4 Urticaria alone is not considered a severe reaction. If neither of these egg-free options is available, a vaccine may still be administered in a medical setting supervised by a provider who is able to manage a severe allergic reaction (which rarely occurs).
All vaccine products available for the upcoming influenza season are listed and described on the CDC Web site, as is a summary of related recommendations.4 Particular attention should be paid to the dose of vaccine administered, as it differs by product for those ages 6 through 35 months of age and those ages 65 years and older.
Use of antiviral medications
Four antiviral medications are now available for treating influenza (3 neuraminidase inhibitors and 1 endonuclease inhibitor), and there are 2 agents for preventing influenza, both neuraminidase inhibitors (TABLE 1).5 The CDC recommends treating with antivirals as soon as possible if individuals with confirmed or suspected influenza require hospitalization; have severe, complicated, or progressive illness; or are at high risk for complications. Use antivirals based on clinical judgment if previously healthy individuals do not have severe complications and are not at increased risk for complications, and only if the medication can be started within 48 hours of symptom onset.
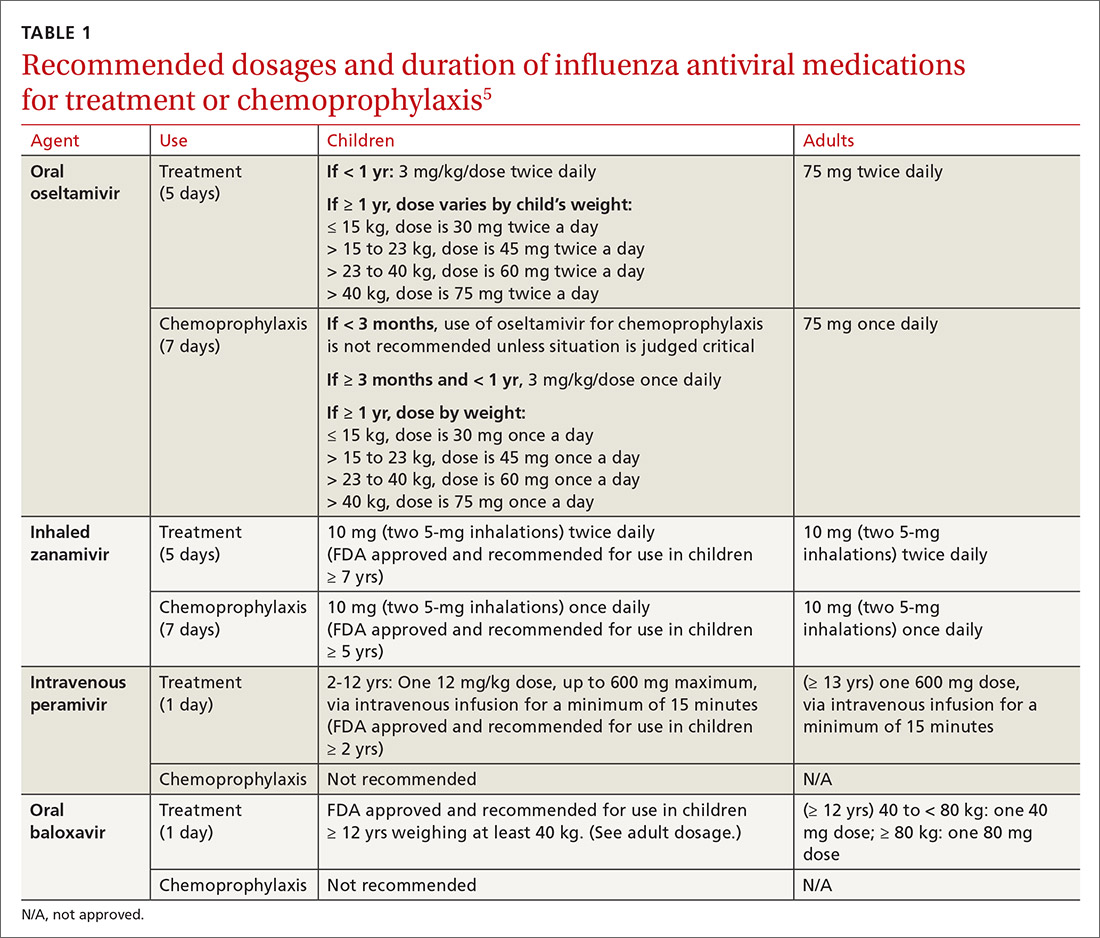
The CDC discourages widespread use of antivirals to prevent influenza, either pre- or postexposure, although it specifies certain situations in which usage would be acceptable (TABLE 2).5 There is some concern that widespread use could lead to the emergence of drug-resistant strains and that using postexposure dosing could lead to suboptimal treatment if influenza infection occurred before the start of prophylaxis. If postexposure antivirals are prescribed, they should be started within 48 hours of exposure and continued for 7 days after the last exposure.

Continue to: A potential perfect storm
A potential perfect storm: Concurrence of influenza and SARS-coV-19
While we have vaccines and antivirals to prevent influenza, and have effective antivirals for treatment, no prevention or treatment options exist for COVID-19, except, possibly, dexamethasone to reduce mortality among those seriously ill.6 The concurrence of influenza and COVID-19 will present unique challenges for the health care system.
Action steps. Keep abreast of the incidences of circulating SARS-coV-19 and influenza viruses in your community. The similar signs and symptoms of these 2 infectious agents will complicate diagnosis. Rapid, or point-of-care, tests for influenza are widely available, but their accuracy varies and not all tests detect both influenza A and B. The CDC lists approved point-of-care tests at www.cdc.gov/flu/professionals/diagnosis/table-ridt.html and advises on how to interpret these test results when influenza is and is not circulating in the community, at www.cdc.gov/flu/professionals/diagnosis/clinician_guidance_ridt.htm.
Clinical practice advice for both conditions should be implemented when any patient presents with ILI:7
- Most patients who are not seriously ill and have no conditions that place them at high risk for adverse outcomes can be treated symptomatically at home.
- Those with ILI should be tested for both influenza virus and SARS-CoV-2 if testing is available. It is possible to be co-infected.
- Sick patients should self-isolate at home for the duration of their symptoms.
- If others live in the house, the sick person should stay in a separate room and wear a mask. Everyone in the house should cover coughs and sneezes (if not wearing a mask), dispose of used tissues in a trash can (rather than leaving them on night stands and countertops), and wash hands frequently.
- All household members should be vaccinated against influenza. Those who are unvaccinated, and those at high risk who have been recently vaccinated, can consider influenza antiviral prophylaxis. If the sick family member is confirmed to have COVID-19, with no co-existing influenza, anti-influenza antiviral prophylaxis may be discontinued.
- Clinical infection control practices should be the same for anyone presenting with ILI.7 Enhanced clinic-based infection control practices to prevent spread of SARS-CoV-2 are listed in TABLE 3.8
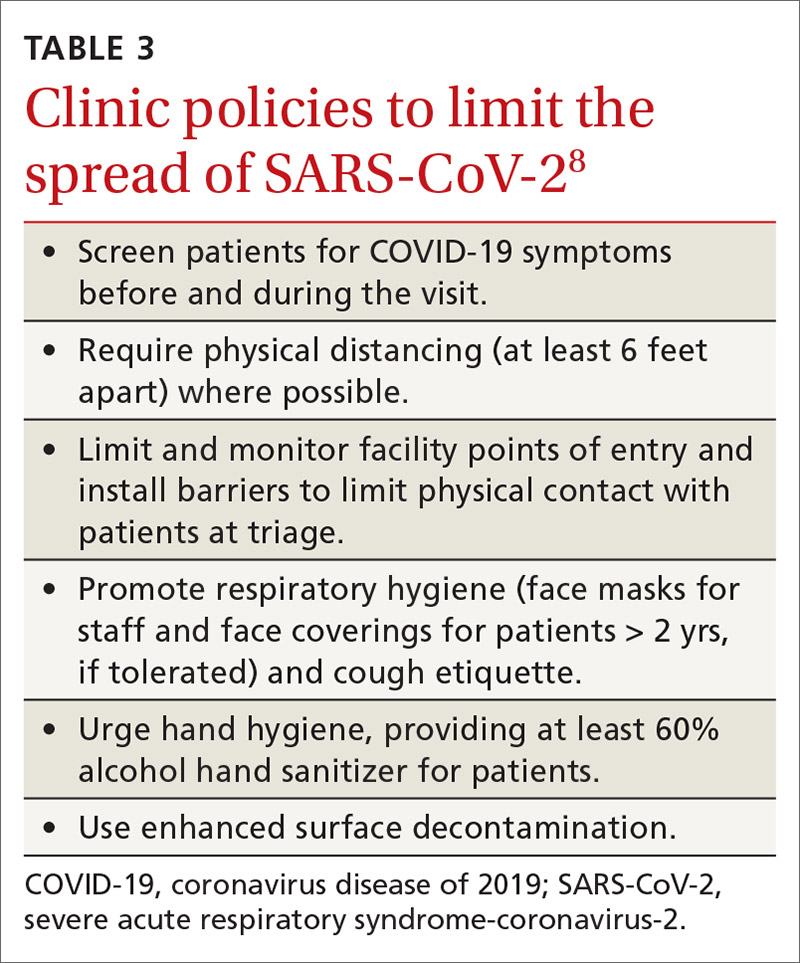
Since there currently are no preventive medications proven to work for COVID-19, the main clinical decision physicians will have to make when a patient presents with ILI is whether to use antivirals to treat those who are at risk for complications based on the result of rapid, on-site influenza testing, or clinical presentation, or both. In this situation, knowledge of which viruses are circulating at high rates in the community could be valuable.
Milder season or perfect storm? The society-wide interventions that have been encouraged (although not mandated everywhere) to prevent community spread of SARS-CoV-2 should help prevent the community spread of influenza as well, and, if adhered to, may lead to a milder influenza season than would otherwise have occurred. However, given the uncertainties, the combination of influenza and coronavirus could present a perfect storm for the health care system and result in higher-than-normal morbidity and mortality from ILI and pneumonia overall.
Continue to: The possibility that one or more vaccines...
The possibility that one or more vaccines to prevent COVID-19 may be available in late 2020 or early 2021 offers hope. However, in current testing, the vaccine is not being given simultaneously with the influenza vaccine. If the potential for adverse interaction exists between the vaccines, it is important that influenza vaccine be given by mid- to late-October to avoid such an interaction if and when the new SARS-CoV-2 vaccine becomes available. Individuals who have symptoms of COVID-19 should not be vaccinated with influenza vaccine until they are considered noninfectious.
Encourage influenza vaccination. The COVID-19 pandemic may make it difficult to achieve desired community influenza vaccine levels because of decreased visits to medical facilities for preventive care, possible lower insurance coverage due to loss of employment, and a decrease in worksite mass vaccination programs. This makes it important for family physicians to encourage and offer influenza vaccines at their clinical sites.
Several evidence-based practices have been shown to improve vaccine uptake. Examples of such practices include patient reminder and recall systems that provide feedback to clinicians about rates of vaccination among patients, and establishing standing orders for vaccine administration that allow other health care providers to assess a patient’s immunization status and administer vaccinations according to a protocol.9 Finally, the CDC provides a video on how to recommend influenza vaccine to those who may be resistant (www.cdc.gov/vaccines/howirecommend/adult-vacc-videos.html).
SIDEBAR
CDC influenza resources
Point-of-care tests that detect both influenza A and B viruses approved by the CDC
www.cdc.gov/flu/professionals/diagnosis/table-ridt.html
Advice on how to interpret the test results
www.cdc.gov/flu/professionals/diagnosis/clinician_guidance_ridt.htm
How to recommend influenza vaccine to reluctant patients
www.cdc.gov/vaccines/howirecommend/adult-vacc-videos.html
CDC, Centers for Disease Control and Prevention.
1. Grohskopf L. Influenza work groups: updates, considerations, and proposed recommendations for the 2020-2021 season. Presented at the ACIP meeting June 24, 2020. www.youtube.com/watch?v=W1SV2DSJsaQ&list=PLvrp9iOILTQb6D9e1YZWpbUvzfptNMKx2&index=8&t=0s. [Time stamp: 1:26:48] Accessed Septemeber 29, 2020.
2. Campos-Outcalt D. Facts to help you keep pace with the vaccine conversation. J Fam Pract. 2019;68:341-346.
3. Grohskopf L, Alyanak E, Broder KR, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2020-21 Influenza Season. MMWR Recomm Rep. 2020;69:1-24.
4. Prevention and Control of Seasonal Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices (ACIP)—United States, 2020-21 Summary of Recommendations. www.cdc.gov/flu/pdf/professionals/acip/acip-2020-21-summary-of-recommendations.pdf. Accessed September 29, 2020.
5. CDC. Influenza antiviral medications: summary for clinicians. www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm. Accessed September 29, 2020.
6. NIH. COVID-19 treatment guidelines. Corticosteroids. www.covid19treatmentguidelines.nih.gov/immune-based-therapy/immunomodulators/corticosteroids/. Accessed September 29, 2020.
7. CDC. Infection control. www.cdc.gov/infectioncontrol/. Accessed September 29, 2020.
8. CDC. Interim infection prevention and control recommendations for healthcare personnel during the coronavirus disease 2019 (COVID-19) pandemic. www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control.html. Accessed September 29, 2020.
9. HHS. CPSTF findings for increasing vaccination. www.thecommunityguide.org/content/task-force-findings-increasing-vaccination. Accessed September 29, 2020.
The 2020-2021 influenza season is shaping up to be challenging. Its likely concurrence with the ongoing severe acute respiratory syndrome-coronavirus 2 (SARS-coV-2) pandemic (COVID-19) will pose diagnostic and therapeutic dilemmas and could overload the hospital system. But there could also be potential synergies in preventing morbidity and mortality from each disease.
A consistent pattern overthe past few influenza seasons
During the 2019-2020 flu season, there were an estimated 410,000 to 740,000 hospitalizations and 24,000 to 62,000 deaths attributed to influenza.1 As seen in FIGURE 1, office visits for influenza-like illness (ILI) began to increase in late November and early December in each of the last 3 years (2017-2018, 2018-2019, 2019-2020) and stayed elevated above baseline for about 4 months each season.1

The effectiveness of influenza vaccine during the 2019-2020 season is being estimated using the US Flu Vaccine Effectiveness Network, which has close to 9000 enrollees. Overall, it appears the vaccine was 39% effective against medically attended influenza, with a higher effectiveness against influenza B (44%) than against A/H1N1 (31%). Effectiveness against influenza B was similar in all age groups, but effectiveness against A/H1N1 was highest for those ages 50 to 64 years (45%) and lowest for those ages 6 months through 8 years (22%), although 95% confidence intervals overlapped for all age groups (FIGURE 2). These preliminary effectiveness rates were presented at the summer meeting of the Advisory Committee on Immunization Practices (ACIP).1

Influenza vaccine safety data for 2019-2020 were based on the Vaccine Adverse Event Reporting System (VAERS), a passive surveillance system, and on the Vaccine Safety Datalink (VSD) system, an active surveillance system involving close to 6 million doses administered at VSD sites. No safety concerns were identified for any of the different vaccine types. Both the VAERS and VSD surveillance systems have been described in more detail in a previous Practice Alert.2
Recommendations for 2020-2021
The composition of the influenza vaccines for this year’s flu season will be different for 3 of the 4 antigens: A/H1N1, A/H2N2 and B/Victoria.3 The antigens included in the influenza vaccines each year are decided on in the spring, based on surveillance of circulating strains around the world. The effectiveness of the vaccine each year largely depends on how well the strains included in the vaccine match those circulating in the United States during the influenza season.
The main immunization recommendation for preventing morbidity and mortality from influenza has not changed: All individuals ages 6 months and older without a contraindication should receive an influenza vaccine.4 The Centers for Disease Control and Prevention (CDC) recommends that patients receive the vaccine by the end of October.4 This includes the second dose for those children younger than 9 years who need 2 doses—ie, those who have received fewer than 2 doses of influenza vaccine prior to July 2020. Vaccination should continue through the end of the season for anyone who has not received a 2020-2021 influenza vaccine.
Two new influenza vaccine products are available for use in those ages 65 years and older: Fluzone high-dose quadrivalent and Fluad Quadrivalent (adjuvanted).4 Both of these products were available last year as trivalent options. Currently no specific vaccine product is listed as preferred by ACIP for those ages 65 and older.
Continue to: New vaccine contraindications
New vaccine contraindications. Four medical conditions have been added to the list of contraindications for quadrivalent live, attenuated influenza vaccine (LAIV4): cochlear implant, cerebrospinal fluid leak, asplenia (anatomic and functional), and sickle cell anemia.4 In addition, those who receive LAIV4 should not be prescribed an influenza antiviral until 2 weeks after receiving the vaccine. And the vaccine should not be administered for 48 hours after receipt of oseltamivir or zanamivir, 5 days after peramivir, and 17 days after baloxavir marboxil.4 This is to prevent possible antiviral inactivation of the live attenuated influenza viruses in the vaccine.
For those who have a history of severe allergic reaction to eggs, there are now 2 egg-free options: cell-culture-based inactivated vaccine (ccIIV4) and recombinant influenza vaccine (RIV4).3,4 Urticaria alone is not considered a severe reaction. If neither of these egg-free options is available, a vaccine may still be administered in a medical setting supervised by a provider who is able to manage a severe allergic reaction (which rarely occurs).
All vaccine products available for the upcoming influenza season are listed and described on the CDC Web site, as is a summary of related recommendations.4 Particular attention should be paid to the dose of vaccine administered, as it differs by product for those ages 6 through 35 months of age and those ages 65 years and older.
Use of antiviral medications
Four antiviral medications are now available for treating influenza (3 neuraminidase inhibitors and 1 endonuclease inhibitor), and there are 2 agents for preventing influenza, both neuraminidase inhibitors (TABLE 1).5 The CDC recommends treating with antivirals as soon as possible if individuals with confirmed or suspected influenza require hospitalization; have severe, complicated, or progressive illness; or are at high risk for complications. Use antivirals based on clinical judgment if previously healthy individuals do not have severe complications and are not at increased risk for complications, and only if the medication can be started within 48 hours of symptom onset.

The CDC discourages widespread use of antivirals to prevent influenza, either pre- or postexposure, although it specifies certain situations in which usage would be acceptable (TABLE 2).5 There is some concern that widespread use could lead to the emergence of drug-resistant strains and that using postexposure dosing could lead to suboptimal treatment if influenza infection occurred before the start of prophylaxis. If postexposure antivirals are prescribed, they should be started within 48 hours of exposure and continued for 7 days after the last exposure.

Continue to: A potential perfect storm
A potential perfect storm: Concurrence of influenza and SARS-coV-19
While we have vaccines and antivirals to prevent influenza, and have effective antivirals for treatment, no prevention or treatment options exist for COVID-19, except, possibly, dexamethasone to reduce mortality among those seriously ill.6 The concurrence of influenza and COVID-19 will present unique challenges for the health care system.
Action steps. Keep abreast of the incidences of circulating SARS-coV-19 and influenza viruses in your community. The similar signs and symptoms of these 2 infectious agents will complicate diagnosis. Rapid, or point-of-care, tests for influenza are widely available, but their accuracy varies and not all tests detect both influenza A and B. The CDC lists approved point-of-care tests at www.cdc.gov/flu/professionals/diagnosis/table-ridt.html and advises on how to interpret these test results when influenza is and is not circulating in the community, at www.cdc.gov/flu/professionals/diagnosis/clinician_guidance_ridt.htm.
Clinical practice advice for both conditions should be implemented when any patient presents with ILI:7
- Most patients who are not seriously ill and have no conditions that place them at high risk for adverse outcomes can be treated symptomatically at home.
- Those with ILI should be tested for both influenza virus and SARS-CoV-2 if testing is available. It is possible to be co-infected.
- Sick patients should self-isolate at home for the duration of their symptoms.
- If others live in the house, the sick person should stay in a separate room and wear a mask. Everyone in the house should cover coughs and sneezes (if not wearing a mask), dispose of used tissues in a trash can (rather than leaving them on night stands and countertops), and wash hands frequently.
- All household members should be vaccinated against influenza. Those who are unvaccinated, and those at high risk who have been recently vaccinated, can consider influenza antiviral prophylaxis. If the sick family member is confirmed to have COVID-19, with no co-existing influenza, anti-influenza antiviral prophylaxis may be discontinued.
- Clinical infection control practices should be the same for anyone presenting with ILI.7 Enhanced clinic-based infection control practices to prevent spread of SARS-CoV-2 are listed in TABLE 3.8

Since there currently are no preventive medications proven to work for COVID-19, the main clinical decision physicians will have to make when a patient presents with ILI is whether to use antivirals to treat those who are at risk for complications based on the result of rapid, on-site influenza testing, or clinical presentation, or both. In this situation, knowledge of which viruses are circulating at high rates in the community could be valuable.
Milder season or perfect storm? The society-wide interventions that have been encouraged (although not mandated everywhere) to prevent community spread of SARS-CoV-2 should help prevent the community spread of influenza as well, and, if adhered to, may lead to a milder influenza season than would otherwise have occurred. However, given the uncertainties, the combination of influenza and coronavirus could present a perfect storm for the health care system and result in higher-than-normal morbidity and mortality from ILI and pneumonia overall.
Continue to: The possibility that one or more vaccines...
The possibility that one or more vaccines to prevent COVID-19 may be available in late 2020 or early 2021 offers hope. However, in current testing, the vaccine is not being given simultaneously with the influenza vaccine. If the potential for adverse interaction exists between the vaccines, it is important that influenza vaccine be given by mid- to late-October to avoid such an interaction if and when the new SARS-CoV-2 vaccine becomes available. Individuals who have symptoms of COVID-19 should not be vaccinated with influenza vaccine until they are considered noninfectious.
Encourage influenza vaccination. The COVID-19 pandemic may make it difficult to achieve desired community influenza vaccine levels because of decreased visits to medical facilities for preventive care, possible lower insurance coverage due to loss of employment, and a decrease in worksite mass vaccination programs. This makes it important for family physicians to encourage and offer influenza vaccines at their clinical sites.
Several evidence-based practices have been shown to improve vaccine uptake. Examples of such practices include patient reminder and recall systems that provide feedback to clinicians about rates of vaccination among patients, and establishing standing orders for vaccine administration that allow other health care providers to assess a patient’s immunization status and administer vaccinations according to a protocol.9 Finally, the CDC provides a video on how to recommend influenza vaccine to those who may be resistant (www.cdc.gov/vaccines/howirecommend/adult-vacc-videos.html).
SIDEBAR
CDC influenza resources
Point-of-care tests that detect both influenza A and B viruses approved by the CDC
www.cdc.gov/flu/professionals/diagnosis/table-ridt.html
Advice on how to interpret the test results
www.cdc.gov/flu/professionals/diagnosis/clinician_guidance_ridt.htm
How to recommend influenza vaccine to reluctant patients
www.cdc.gov/vaccines/howirecommend/adult-vacc-videos.html
CDC, Centers for Disease Control and Prevention.
The 2020-2021 influenza season is shaping up to be challenging. Its likely concurrence with the ongoing severe acute respiratory syndrome-coronavirus 2 (SARS-coV-2) pandemic (COVID-19) will pose diagnostic and therapeutic dilemmas and could overload the hospital system. But there could also be potential synergies in preventing morbidity and mortality from each disease.
A consistent pattern overthe past few influenza seasons
During the 2019-2020 flu season, there were an estimated 410,000 to 740,000 hospitalizations and 24,000 to 62,000 deaths attributed to influenza.1 As seen in FIGURE 1, office visits for influenza-like illness (ILI) began to increase in late November and early December in each of the last 3 years (2017-2018, 2018-2019, 2019-2020) and stayed elevated above baseline for about 4 months each season.1

The effectiveness of influenza vaccine during the 2019-2020 season is being estimated using the US Flu Vaccine Effectiveness Network, which has close to 9000 enrollees. Overall, it appears the vaccine was 39% effective against medically attended influenza, with a higher effectiveness against influenza B (44%) than against A/H1N1 (31%). Effectiveness against influenza B was similar in all age groups, but effectiveness against A/H1N1 was highest for those ages 50 to 64 years (45%) and lowest for those ages 6 months through 8 years (22%), although 95% confidence intervals overlapped for all age groups (FIGURE 2). These preliminary effectiveness rates were presented at the summer meeting of the Advisory Committee on Immunization Practices (ACIP).1

Influenza vaccine safety data for 2019-2020 were based on the Vaccine Adverse Event Reporting System (VAERS), a passive surveillance system, and on the Vaccine Safety Datalink (VSD) system, an active surveillance system involving close to 6 million doses administered at VSD sites. No safety concerns were identified for any of the different vaccine types. Both the VAERS and VSD surveillance systems have been described in more detail in a previous Practice Alert.2
Recommendations for 2020-2021
The composition of the influenza vaccines for this year’s flu season will be different for 3 of the 4 antigens: A/H1N1, A/H2N2 and B/Victoria.3 The antigens included in the influenza vaccines each year are decided on in the spring, based on surveillance of circulating strains around the world. The effectiveness of the vaccine each year largely depends on how well the strains included in the vaccine match those circulating in the United States during the influenza season.
The main immunization recommendation for preventing morbidity and mortality from influenza has not changed: All individuals ages 6 months and older without a contraindication should receive an influenza vaccine.4 The Centers for Disease Control and Prevention (CDC) recommends that patients receive the vaccine by the end of October.4 This includes the second dose for those children younger than 9 years who need 2 doses—ie, those who have received fewer than 2 doses of influenza vaccine prior to July 2020. Vaccination should continue through the end of the season for anyone who has not received a 2020-2021 influenza vaccine.
Two new influenza vaccine products are available for use in those ages 65 years and older: Fluzone high-dose quadrivalent and Fluad Quadrivalent (adjuvanted).4 Both of these products were available last year as trivalent options. Currently no specific vaccine product is listed as preferred by ACIP for those ages 65 and older.
Continue to: New vaccine contraindications
New vaccine contraindications. Four medical conditions have been added to the list of contraindications for quadrivalent live, attenuated influenza vaccine (LAIV4): cochlear implant, cerebrospinal fluid leak, asplenia (anatomic and functional), and sickle cell anemia.4 In addition, those who receive LAIV4 should not be prescribed an influenza antiviral until 2 weeks after receiving the vaccine. And the vaccine should not be administered for 48 hours after receipt of oseltamivir or zanamivir, 5 days after peramivir, and 17 days after baloxavir marboxil.4 This is to prevent possible antiviral inactivation of the live attenuated influenza viruses in the vaccine.
For those who have a history of severe allergic reaction to eggs, there are now 2 egg-free options: cell-culture-based inactivated vaccine (ccIIV4) and recombinant influenza vaccine (RIV4).3,4 Urticaria alone is not considered a severe reaction. If neither of these egg-free options is available, a vaccine may still be administered in a medical setting supervised by a provider who is able to manage a severe allergic reaction (which rarely occurs).
All vaccine products available for the upcoming influenza season are listed and described on the CDC Web site, as is a summary of related recommendations.4 Particular attention should be paid to the dose of vaccine administered, as it differs by product for those ages 6 through 35 months of age and those ages 65 years and older.
Use of antiviral medications
Four antiviral medications are now available for treating influenza (3 neuraminidase inhibitors and 1 endonuclease inhibitor), and there are 2 agents for preventing influenza, both neuraminidase inhibitors (TABLE 1).5 The CDC recommends treating with antivirals as soon as possible if individuals with confirmed or suspected influenza require hospitalization; have severe, complicated, or progressive illness; or are at high risk for complications. Use antivirals based on clinical judgment if previously healthy individuals do not have severe complications and are not at increased risk for complications, and only if the medication can be started within 48 hours of symptom onset.

The CDC discourages widespread use of antivirals to prevent influenza, either pre- or postexposure, although it specifies certain situations in which usage would be acceptable (TABLE 2).5 There is some concern that widespread use could lead to the emergence of drug-resistant strains and that using postexposure dosing could lead to suboptimal treatment if influenza infection occurred before the start of prophylaxis. If postexposure antivirals are prescribed, they should be started within 48 hours of exposure and continued for 7 days after the last exposure.

Continue to: A potential perfect storm
A potential perfect storm: Concurrence of influenza and SARS-coV-19
While we have vaccines and antivirals to prevent influenza, and have effective antivirals for treatment, no prevention or treatment options exist for COVID-19, except, possibly, dexamethasone to reduce mortality among those seriously ill.6 The concurrence of influenza and COVID-19 will present unique challenges for the health care system.
Action steps. Keep abreast of the incidences of circulating SARS-coV-19 and influenza viruses in your community. The similar signs and symptoms of these 2 infectious agents will complicate diagnosis. Rapid, or point-of-care, tests for influenza are widely available, but their accuracy varies and not all tests detect both influenza A and B. The CDC lists approved point-of-care tests at www.cdc.gov/flu/professionals/diagnosis/table-ridt.html and advises on how to interpret these test results when influenza is and is not circulating in the community, at www.cdc.gov/flu/professionals/diagnosis/clinician_guidance_ridt.htm.
Clinical practice advice for both conditions should be implemented when any patient presents with ILI:7
- Most patients who are not seriously ill and have no conditions that place them at high risk for adverse outcomes can be treated symptomatically at home.
- Those with ILI should be tested for both influenza virus and SARS-CoV-2 if testing is available. It is possible to be co-infected.
- Sick patients should self-isolate at home for the duration of their symptoms.
- If others live in the house, the sick person should stay in a separate room and wear a mask. Everyone in the house should cover coughs and sneezes (if not wearing a mask), dispose of used tissues in a trash can (rather than leaving them on night stands and countertops), and wash hands frequently.
- All household members should be vaccinated against influenza. Those who are unvaccinated, and those at high risk who have been recently vaccinated, can consider influenza antiviral prophylaxis. If the sick family member is confirmed to have COVID-19, with no co-existing influenza, anti-influenza antiviral prophylaxis may be discontinued.
- Clinical infection control practices should be the same for anyone presenting with ILI.7 Enhanced clinic-based infection control practices to prevent spread of SARS-CoV-2 are listed in TABLE 3.8

Since there currently are no preventive medications proven to work for COVID-19, the main clinical decision physicians will have to make when a patient presents with ILI is whether to use antivirals to treat those who are at risk for complications based on the result of rapid, on-site influenza testing, or clinical presentation, or both. In this situation, knowledge of which viruses are circulating at high rates in the community could be valuable.
Milder season or perfect storm? The society-wide interventions that have been encouraged (although not mandated everywhere) to prevent community spread of SARS-CoV-2 should help prevent the community spread of influenza as well, and, if adhered to, may lead to a milder influenza season than would otherwise have occurred. However, given the uncertainties, the combination of influenza and coronavirus could present a perfect storm for the health care system and result in higher-than-normal morbidity and mortality from ILI and pneumonia overall.
Continue to: The possibility that one or more vaccines...
The possibility that one or more vaccines to prevent COVID-19 may be available in late 2020 or early 2021 offers hope. However, in current testing, the vaccine is not being given simultaneously with the influenza vaccine. If the potential for adverse interaction exists between the vaccines, it is important that influenza vaccine be given by mid- to late-October to avoid such an interaction if and when the new SARS-CoV-2 vaccine becomes available. Individuals who have symptoms of COVID-19 should not be vaccinated with influenza vaccine until they are considered noninfectious.
Encourage influenza vaccination. The COVID-19 pandemic may make it difficult to achieve desired community influenza vaccine levels because of decreased visits to medical facilities for preventive care, possible lower insurance coverage due to loss of employment, and a decrease in worksite mass vaccination programs. This makes it important for family physicians to encourage and offer influenza vaccines at their clinical sites.
Several evidence-based practices have been shown to improve vaccine uptake. Examples of such practices include patient reminder and recall systems that provide feedback to clinicians about rates of vaccination among patients, and establishing standing orders for vaccine administration that allow other health care providers to assess a patient’s immunization status and administer vaccinations according to a protocol.9 Finally, the CDC provides a video on how to recommend influenza vaccine to those who may be resistant (www.cdc.gov/vaccines/howirecommend/adult-vacc-videos.html).
SIDEBAR
CDC influenza resources
Point-of-care tests that detect both influenza A and B viruses approved by the CDC
www.cdc.gov/flu/professionals/diagnosis/table-ridt.html
Advice on how to interpret the test results
www.cdc.gov/flu/professionals/diagnosis/clinician_guidance_ridt.htm
How to recommend influenza vaccine to reluctant patients
www.cdc.gov/vaccines/howirecommend/adult-vacc-videos.html
CDC, Centers for Disease Control and Prevention.
1. Grohskopf L. Influenza work groups: updates, considerations, and proposed recommendations for the 2020-2021 season. Presented at the ACIP meeting June 24, 2020. www.youtube.com/watch?v=W1SV2DSJsaQ&list=PLvrp9iOILTQb6D9e1YZWpbUvzfptNMKx2&index=8&t=0s. [Time stamp: 1:26:48] Accessed Septemeber 29, 2020.
2. Campos-Outcalt D. Facts to help you keep pace with the vaccine conversation. J Fam Pract. 2019;68:341-346.
3. Grohskopf L, Alyanak E, Broder KR, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2020-21 Influenza Season. MMWR Recomm Rep. 2020;69:1-24.
4. Prevention and Control of Seasonal Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices (ACIP)—United States, 2020-21 Summary of Recommendations. www.cdc.gov/flu/pdf/professionals/acip/acip-2020-21-summary-of-recommendations.pdf. Accessed September 29, 2020.
5. CDC. Influenza antiviral medications: summary for clinicians. www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm. Accessed September 29, 2020.
6. NIH. COVID-19 treatment guidelines. Corticosteroids. www.covid19treatmentguidelines.nih.gov/immune-based-therapy/immunomodulators/corticosteroids/. Accessed September 29, 2020.
7. CDC. Infection control. www.cdc.gov/infectioncontrol/. Accessed September 29, 2020.
8. CDC. Interim infection prevention and control recommendations for healthcare personnel during the coronavirus disease 2019 (COVID-19) pandemic. www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control.html. Accessed September 29, 2020.
9. HHS. CPSTF findings for increasing vaccination. www.thecommunityguide.org/content/task-force-findings-increasing-vaccination. Accessed September 29, 2020.
1. Grohskopf L. Influenza work groups: updates, considerations, and proposed recommendations for the 2020-2021 season. Presented at the ACIP meeting June 24, 2020. www.youtube.com/watch?v=W1SV2DSJsaQ&list=PLvrp9iOILTQb6D9e1YZWpbUvzfptNMKx2&index=8&t=0s. [Time stamp: 1:26:48] Accessed Septemeber 29, 2020.
2. Campos-Outcalt D. Facts to help you keep pace with the vaccine conversation. J Fam Pract. 2019;68:341-346.
3. Grohskopf L, Alyanak E, Broder KR, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2020-21 Influenza Season. MMWR Recomm Rep. 2020;69:1-24.
4. Prevention and Control of Seasonal Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices (ACIP)—United States, 2020-21 Summary of Recommendations. www.cdc.gov/flu/pdf/professionals/acip/acip-2020-21-summary-of-recommendations.pdf. Accessed September 29, 2020.
5. CDC. Influenza antiviral medications: summary for clinicians. www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm. Accessed September 29, 2020.
6. NIH. COVID-19 treatment guidelines. Corticosteroids. www.covid19treatmentguidelines.nih.gov/immune-based-therapy/immunomodulators/corticosteroids/. Accessed September 29, 2020.
7. CDC. Infection control. www.cdc.gov/infectioncontrol/. Accessed September 29, 2020.
8. CDC. Interim infection prevention and control recommendations for healthcare personnel during the coronavirus disease 2019 (COVID-19) pandemic. www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control.html. Accessed September 29, 2020.
9. HHS. CPSTF findings for increasing vaccination. www.thecommunityguide.org/content/task-force-findings-increasing-vaccination. Accessed September 29, 2020.