User login
New research in otitis media
New research was presented at the International Society for Otitis Media meeting in June 2019, which I attended. I would like to share a selection of new findings from the many presentations.
Transtympanic antibiotic delivery
Topical therapy has been used to treat only otitis externa and acute otitis media (AOM) with ear discharge. Giving antibiotics through the tympanic membrane could mitigate many of the concerns about antibiotic use driving antibiotic resistance of bacteria among children. Up to now, using antibiotics in the ear canal to treat AOM has not been considered because the tympanic membrane is highly impermeable to the transtympanic diffusion of any drugs. However, in recent years, a number of different drug delivery systems have been developed, and in some cases, animal studies have shown that noninvasive transtympanic delivery is possible so that drugs can reach high concentrations in the middle ear without damage. Nanovesicles and nanoliposomes that contain antibiotics and are small enough to pass through the eardrum have been developed and tested in animal models; these show promise. Ototopical administration of a drug called vinpocetine that was repurposed has been tested in mice and shown to reduce inflammation and mucus production in the middle ear during otitis media.
Biofilms
Antibiotic treatment failure can occur in AOM for several reasons. The treatment of choice, amoxicillin, for example may fail to achieve an adequate concentration because of poor absorption in the gastrointestinal tract or poor penetration into the middle ear. Or, the antibiotic chosen may not be effective because of resistance of the strain causing the infection. Another explanation, especially in recurrent AOM and chronic AOM, could be the presence of biofilms. Biofilms are multicellular bacterial communities incorporated in a polymeric, plasticlike matrix in which pathogens are protected from antibiotic activity. The biofilm provides a physical barrier to antibiotic penetration, and bacteria can persist in the middle ear and periodically cause a new AOM. If AOM persists or becomes a more chronic otitis media with effusion, the “glue ear” causes an environment in the middle ear that is low in oxygen. A low-oxygen environment is favorable to biofilms. Also one might expect that middle ear pus would have a low pH, but actual measurements show the pH is highly alkaline. Species of Haemophilus influenzae have been identified as more virulent when in an alkaline pH or the alkaline pH makes the H. influenzae persist better in the middle ear, perhaps in a biofilm. To eliminate biofilms and improve antibiotic efficacy, a vaccine against a protein expressed by H. influenzae has been developed. Antibodies against this protein have been shown to disrupt and prevent the formation of biofilms in an animal model.
Probiotics
The normal bacteria that live in the nasopharynx of children with recurrent AOM is now known to differ from that of children who experience infrequent AOM or remain AOM-free throughout childhood. The use of oral pre- and probiotics for AOM prophylaxis remains debated because the results of studies are conflicting and frequently show no effect. So the idea of using prebiotics or probiotics to create a favorable “microbiome” of the nose is under investigation. Two species of bacteria that are gathering the most attention are Corynebacterium species (a few types in particular) and a bacteria called Dolosigranulum pigrum. Delivery of the commensal species would be as a nose spray.
Vaccines
The use of pneumococcal conjugate vaccines (PCVs) has reduced the frequency of AOM caused by Streptococcus pneumoniae. PCVs are not as effective against AOM as they are against invasive pneumococcal disease, but they still help a lot. However, because there are now at least 96 different serotypes of the pneumococcus based on different capsular types, we see a pattern of replacement of disease-causing strains by new strains within a few years of introduction of a new formulation. We started with 7 serotypes (Prevnar 7) in year 2000, and it was replaced by the current formulation with 13 serotypes (Prevnar 13) in 2010. Replacements have occurred again so vaccine companies are making new formulations for the future that include more serotypes, up to 20 serotypes. But, technically and feasibility-wise there is a limit to making such vaccines. A vaccine based on killed unencapsulated bacteria has been tested for safety and immunogenicity in young children. There is no test so far for prevention of AOM. Another type of vaccine based on proteins expressed by the pneumococcus that could be vaccine targets was tested in American Navajo children, and it failed to be as efficacious as hoped.
Biomarkers.
Due to recurrent AOM or persistent otitis media with effusion, about 15% of children in the United States receive tympanostomy tubes. Among those who receive tubes, about 20% go on to receive a second set of tubes, often with adenotonsillectomy. To find a biomarker that could identify children likely to require a second set of tubes, the fluid in the middle ear was tested when a first set of tubes were inserted. If bacteria were detected by polymerase chain reaction (PCR) testing or if a profile of specific inflammatory cytokines was measured, those results could be used to predict a high likelihood for a second set of tubes.
Overdiagnosis
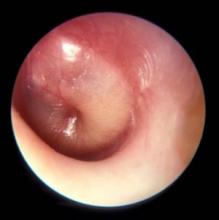
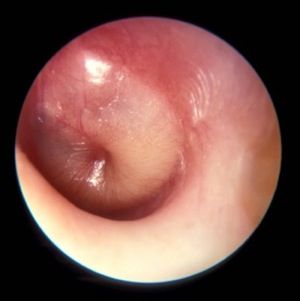
Diagnosis of AOM is challenging in young children, in whom it most frequently occurs. The ear canal is typically about 3 mm wide, the child struggles during the examination, and diagnostic skills are not taught in training, resulting in a high overdiagnosis rate. I presented data that suggest too many children who are not truly otitis prone have been classified as otitis prone based on incorrect clinical diagnosis. My colleagues and I found that 30% of children reach the threshold of three episodes of AOM in 6 months or four within a year when diagnosed by community pediatricians, similar to many other studies. Validated otoscopists (trained by experts with diagnosis definitively proven as at least 85% accurate using tympanocentesis) classify 15% of children as otitis prone – half as many. If tympanocentesis is used to prove middle ear fluid has bacterial pathogens (about 95% yield a bacterial otopathogen using culture and PCR), then about 10% of children are classified as otitis prone – one-third as many. This suggests that children clinically diagnosed by community-based pediatricians are overdiagnosed with AOM, perhaps three times more often than true. And that leads to overuse of antibiotics and referrals for tympanostomy tube surgery more often than should occur. So we need to improve diagnostic methods beyond otoscopy. New types of imaging for the eardrum and middle ear using novel technologies are in early clinical trials.
Immunity
The notion that young children get AOM because of Eustachian tube dysfunction in their early years of life (horizontal anatomy) may be true, but there is more to the story. After 10 years of work, the scientists in my research group have shown that children in the first 3 years of life can have an immune system that is suppressed – it is poorly responsive to pathogens and routine pediatric vaccines. Many features resemble a neonatal immune system, beginning life with a suppressed immune system or being in cytokine storm from birth. We introduced the term “prolonged neonatal-like immune profile (PNIP)” to give a general description of the immune responses we have found in otitis-prone children. They outgrow this. So the immune maturation is delayed but not permanent. It is mostly resolved by age 3 years. We found problems in both innate and adaptive immunity. It may be that the main explanation for recurrent AOM in the first years of life is PNIP. Scientists from Australia also reported immunity problems in Aboriginal children and they are very otitis prone, often progressing to chronic suppurative otitis media. Animal model studies of AOM show inadequate innate and adaptive immunity importantly contribute to the infection as well.
Dr. Pichichero is a specialist in pediatric infectious diseases and director of the Research Institute at Rochester (N.Y.) General Hospital. He has no conflicts to declare. Email him at [email protected].
New research was presented at the International Society for Otitis Media meeting in June 2019, which I attended. I would like to share a selection of new findings from the many presentations.
Transtympanic antibiotic delivery
Topical therapy has been used to treat only otitis externa and acute otitis media (AOM) with ear discharge. Giving antibiotics through the tympanic membrane could mitigate many of the concerns about antibiotic use driving antibiotic resistance of bacteria among children. Up to now, using antibiotics in the ear canal to treat AOM has not been considered because the tympanic membrane is highly impermeable to the transtympanic diffusion of any drugs. However, in recent years, a number of different drug delivery systems have been developed, and in some cases, animal studies have shown that noninvasive transtympanic delivery is possible so that drugs can reach high concentrations in the middle ear without damage. Nanovesicles and nanoliposomes that contain antibiotics and are small enough to pass through the eardrum have been developed and tested in animal models; these show promise. Ototopical administration of a drug called vinpocetine that was repurposed has been tested in mice and shown to reduce inflammation and mucus production in the middle ear during otitis media.
Biofilms
Antibiotic treatment failure can occur in AOM for several reasons. The treatment of choice, amoxicillin, for example may fail to achieve an adequate concentration because of poor absorption in the gastrointestinal tract or poor penetration into the middle ear. Or, the antibiotic chosen may not be effective because of resistance of the strain causing the infection. Another explanation, especially in recurrent AOM and chronic AOM, could be the presence of biofilms. Biofilms are multicellular bacterial communities incorporated in a polymeric, plasticlike matrix in which pathogens are protected from antibiotic activity. The biofilm provides a physical barrier to antibiotic penetration, and bacteria can persist in the middle ear and periodically cause a new AOM. If AOM persists or becomes a more chronic otitis media with effusion, the “glue ear” causes an environment in the middle ear that is low in oxygen. A low-oxygen environment is favorable to biofilms. Also one might expect that middle ear pus would have a low pH, but actual measurements show the pH is highly alkaline. Species of Haemophilus influenzae have been identified as more virulent when in an alkaline pH or the alkaline pH makes the H. influenzae persist better in the middle ear, perhaps in a biofilm. To eliminate biofilms and improve antibiotic efficacy, a vaccine against a protein expressed by H. influenzae has been developed. Antibodies against this protein have been shown to disrupt and prevent the formation of biofilms in an animal model.
Probiotics
The normal bacteria that live in the nasopharynx of children with recurrent AOM is now known to differ from that of children who experience infrequent AOM or remain AOM-free throughout childhood. The use of oral pre- and probiotics for AOM prophylaxis remains debated because the results of studies are conflicting and frequently show no effect. So the idea of using prebiotics or probiotics to create a favorable “microbiome” of the nose is under investigation. Two species of bacteria that are gathering the most attention are Corynebacterium species (a few types in particular) and a bacteria called Dolosigranulum pigrum. Delivery of the commensal species would be as a nose spray.
Vaccines
The use of pneumococcal conjugate vaccines (PCVs) has reduced the frequency of AOM caused by Streptococcus pneumoniae. PCVs are not as effective against AOM as they are against invasive pneumococcal disease, but they still help a lot. However, because there are now at least 96 different serotypes of the pneumococcus based on different capsular types, we see a pattern of replacement of disease-causing strains by new strains within a few years of introduction of a new formulation. We started with 7 serotypes (Prevnar 7) in year 2000, and it was replaced by the current formulation with 13 serotypes (Prevnar 13) in 2010. Replacements have occurred again so vaccine companies are making new formulations for the future that include more serotypes, up to 20 serotypes. But, technically and feasibility-wise there is a limit to making such vaccines. A vaccine based on killed unencapsulated bacteria has been tested for safety and immunogenicity in young children. There is no test so far for prevention of AOM. Another type of vaccine based on proteins expressed by the pneumococcus that could be vaccine targets was tested in American Navajo children, and it failed to be as efficacious as hoped.
Biomarkers.
Due to recurrent AOM or persistent otitis media with effusion, about 15% of children in the United States receive tympanostomy tubes. Among those who receive tubes, about 20% go on to receive a second set of tubes, often with adenotonsillectomy. To find a biomarker that could identify children likely to require a second set of tubes, the fluid in the middle ear was tested when a first set of tubes were inserted. If bacteria were detected by polymerase chain reaction (PCR) testing or if a profile of specific inflammatory cytokines was measured, those results could be used to predict a high likelihood for a second set of tubes.
Overdiagnosis
Diagnosis of AOM is challenging in young children, in whom it most frequently occurs. The ear canal is typically about 3 mm wide, the child struggles during the examination, and diagnostic skills are not taught in training, resulting in a high overdiagnosis rate. I presented data that suggest too many children who are not truly otitis prone have been classified as otitis prone based on incorrect clinical diagnosis. My colleagues and I found that 30% of children reach the threshold of three episodes of AOM in 6 months or four within a year when diagnosed by community pediatricians, similar to many other studies. Validated otoscopists (trained by experts with diagnosis definitively proven as at least 85% accurate using tympanocentesis) classify 15% of children as otitis prone – half as many. If tympanocentesis is used to prove middle ear fluid has bacterial pathogens (about 95% yield a bacterial otopathogen using culture and PCR), then about 10% of children are classified as otitis prone – one-third as many. This suggests that children clinically diagnosed by community-based pediatricians are overdiagnosed with AOM, perhaps three times more often than true. And that leads to overuse of antibiotics and referrals for tympanostomy tube surgery more often than should occur. So we need to improve diagnostic methods beyond otoscopy. New types of imaging for the eardrum and middle ear using novel technologies are in early clinical trials.
Immunity
The notion that young children get AOM because of Eustachian tube dysfunction in their early years of life (horizontal anatomy) may be true, but there is more to the story. After 10 years of work, the scientists in my research group have shown that children in the first 3 years of life can have an immune system that is suppressed – it is poorly responsive to pathogens and routine pediatric vaccines. Many features resemble a neonatal immune system, beginning life with a suppressed immune system or being in cytokine storm from birth. We introduced the term “prolonged neonatal-like immune profile (PNIP)” to give a general description of the immune responses we have found in otitis-prone children. They outgrow this. So the immune maturation is delayed but not permanent. It is mostly resolved by age 3 years. We found problems in both innate and adaptive immunity. It may be that the main explanation for recurrent AOM in the first years of life is PNIP. Scientists from Australia also reported immunity problems in Aboriginal children and they are very otitis prone, often progressing to chronic suppurative otitis media. Animal model studies of AOM show inadequate innate and adaptive immunity importantly contribute to the infection as well.
Dr. Pichichero is a specialist in pediatric infectious diseases and director of the Research Institute at Rochester (N.Y.) General Hospital. He has no conflicts to declare. Email him at [email protected].
New research was presented at the International Society for Otitis Media meeting in June 2019, which I attended. I would like to share a selection of new findings from the many presentations.
Transtympanic antibiotic delivery
Topical therapy has been used to treat only otitis externa and acute otitis media (AOM) with ear discharge. Giving antibiotics through the tympanic membrane could mitigate many of the concerns about antibiotic use driving antibiotic resistance of bacteria among children. Up to now, using antibiotics in the ear canal to treat AOM has not been considered because the tympanic membrane is highly impermeable to the transtympanic diffusion of any drugs. However, in recent years, a number of different drug delivery systems have been developed, and in some cases, animal studies have shown that noninvasive transtympanic delivery is possible so that drugs can reach high concentrations in the middle ear without damage. Nanovesicles and nanoliposomes that contain antibiotics and are small enough to pass through the eardrum have been developed and tested in animal models; these show promise. Ototopical administration of a drug called vinpocetine that was repurposed has been tested in mice and shown to reduce inflammation and mucus production in the middle ear during otitis media.
Biofilms
Antibiotic treatment failure can occur in AOM for several reasons. The treatment of choice, amoxicillin, for example may fail to achieve an adequate concentration because of poor absorption in the gastrointestinal tract or poor penetration into the middle ear. Or, the antibiotic chosen may not be effective because of resistance of the strain causing the infection. Another explanation, especially in recurrent AOM and chronic AOM, could be the presence of biofilms. Biofilms are multicellular bacterial communities incorporated in a polymeric, plasticlike matrix in which pathogens are protected from antibiotic activity. The biofilm provides a physical barrier to antibiotic penetration, and bacteria can persist in the middle ear and periodically cause a new AOM. If AOM persists or becomes a more chronic otitis media with effusion, the “glue ear” causes an environment in the middle ear that is low in oxygen. A low-oxygen environment is favorable to biofilms. Also one might expect that middle ear pus would have a low pH, but actual measurements show the pH is highly alkaline. Species of Haemophilus influenzae have been identified as more virulent when in an alkaline pH or the alkaline pH makes the H. influenzae persist better in the middle ear, perhaps in a biofilm. To eliminate biofilms and improve antibiotic efficacy, a vaccine against a protein expressed by H. influenzae has been developed. Antibodies against this protein have been shown to disrupt and prevent the formation of biofilms in an animal model.
Probiotics
The normal bacteria that live in the nasopharynx of children with recurrent AOM is now known to differ from that of children who experience infrequent AOM or remain AOM-free throughout childhood. The use of oral pre- and probiotics for AOM prophylaxis remains debated because the results of studies are conflicting and frequently show no effect. So the idea of using prebiotics or probiotics to create a favorable “microbiome” of the nose is under investigation. Two species of bacteria that are gathering the most attention are Corynebacterium species (a few types in particular) and a bacteria called Dolosigranulum pigrum. Delivery of the commensal species would be as a nose spray.
Vaccines
The use of pneumococcal conjugate vaccines (PCVs) has reduced the frequency of AOM caused by Streptococcus pneumoniae. PCVs are not as effective against AOM as they are against invasive pneumococcal disease, but they still help a lot. However, because there are now at least 96 different serotypes of the pneumococcus based on different capsular types, we see a pattern of replacement of disease-causing strains by new strains within a few years of introduction of a new formulation. We started with 7 serotypes (Prevnar 7) in year 2000, and it was replaced by the current formulation with 13 serotypes (Prevnar 13) in 2010. Replacements have occurred again so vaccine companies are making new formulations for the future that include more serotypes, up to 20 serotypes. But, technically and feasibility-wise there is a limit to making such vaccines. A vaccine based on killed unencapsulated bacteria has been tested for safety and immunogenicity in young children. There is no test so far for prevention of AOM. Another type of vaccine based on proteins expressed by the pneumococcus that could be vaccine targets was tested in American Navajo children, and it failed to be as efficacious as hoped.
Biomarkers.
Due to recurrent AOM or persistent otitis media with effusion, about 15% of children in the United States receive tympanostomy tubes. Among those who receive tubes, about 20% go on to receive a second set of tubes, often with adenotonsillectomy. To find a biomarker that could identify children likely to require a second set of tubes, the fluid in the middle ear was tested when a first set of tubes were inserted. If bacteria were detected by polymerase chain reaction (PCR) testing or if a profile of specific inflammatory cytokines was measured, those results could be used to predict a high likelihood for a second set of tubes.
Overdiagnosis
Diagnosis of AOM is challenging in young children, in whom it most frequently occurs. The ear canal is typically about 3 mm wide, the child struggles during the examination, and diagnostic skills are not taught in training, resulting in a high overdiagnosis rate. I presented data that suggest too many children who are not truly otitis prone have been classified as otitis prone based on incorrect clinical diagnosis. My colleagues and I found that 30% of children reach the threshold of three episodes of AOM in 6 months or four within a year when diagnosed by community pediatricians, similar to many other studies. Validated otoscopists (trained by experts with diagnosis definitively proven as at least 85% accurate using tympanocentesis) classify 15% of children as otitis prone – half as many. If tympanocentesis is used to prove middle ear fluid has bacterial pathogens (about 95% yield a bacterial otopathogen using culture and PCR), then about 10% of children are classified as otitis prone – one-third as many. This suggests that children clinically diagnosed by community-based pediatricians are overdiagnosed with AOM, perhaps three times more often than true. And that leads to overuse of antibiotics and referrals for tympanostomy tube surgery more often than should occur. So we need to improve diagnostic methods beyond otoscopy. New types of imaging for the eardrum and middle ear using novel technologies are in early clinical trials.
Immunity
The notion that young children get AOM because of Eustachian tube dysfunction in their early years of life (horizontal anatomy) may be true, but there is more to the story. After 10 years of work, the scientists in my research group have shown that children in the first 3 years of life can have an immune system that is suppressed – it is poorly responsive to pathogens and routine pediatric vaccines. Many features resemble a neonatal immune system, beginning life with a suppressed immune system or being in cytokine storm from birth. We introduced the term “prolonged neonatal-like immune profile (PNIP)” to give a general description of the immune responses we have found in otitis-prone children. They outgrow this. So the immune maturation is delayed but not permanent. It is mostly resolved by age 3 years. We found problems in both innate and adaptive immunity. It may be that the main explanation for recurrent AOM in the first years of life is PNIP. Scientists from Australia also reported immunity problems in Aboriginal children and they are very otitis prone, often progressing to chronic suppurative otitis media. Animal model studies of AOM show inadequate innate and adaptive immunity importantly contribute to the infection as well.
Dr. Pichichero is a specialist in pediatric infectious diseases and director of the Research Institute at Rochester (N.Y.) General Hospital. He has no conflicts to declare. Email him at [email protected].
Click for Credit: Roux-en-Y for diabetes; Exercise & fall prevention; more
Here are 5 articles from the July issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Cloud of inconsistency hangs over cannabis data
To take the posttest, go to: https://bit.ly/2NfjaDS
Expires February 6, 2020
2. Roux-en-Y achieves diabetes remission in majority of patients
To take the posttest, go to: https://bit.ly/2x9hLnE
Expires February 6, 2020
3. Socioeconomic status, race found to impact CPAP compliance
To take the posttest, go to: https://bit.ly/2RBpLa9
Expires February 8, 2020
4. Exercise type matters for fall prevention among elderly
To take the posttest, go to: https://bit.ly/2X26OUh
Expires February 12, 2020
5. Adult HIV patients should receive standard vaccinations, with caveats
To take the posttest, go to: https://bit.ly/2X1S7LV
Expires February 12, 2020
Here are 5 articles from the July issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Cloud of inconsistency hangs over cannabis data
To take the posttest, go to: https://bit.ly/2NfjaDS
Expires February 6, 2020
2. Roux-en-Y achieves diabetes remission in majority of patients
To take the posttest, go to: https://bit.ly/2x9hLnE
Expires February 6, 2020
3. Socioeconomic status, race found to impact CPAP compliance
To take the posttest, go to: https://bit.ly/2RBpLa9
Expires February 8, 2020
4. Exercise type matters for fall prevention among elderly
To take the posttest, go to: https://bit.ly/2X26OUh
Expires February 12, 2020
5. Adult HIV patients should receive standard vaccinations, with caveats
To take the posttest, go to: https://bit.ly/2X1S7LV
Expires February 12, 2020
Here are 5 articles from the July issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Cloud of inconsistency hangs over cannabis data
To take the posttest, go to: https://bit.ly/2NfjaDS
Expires February 6, 2020
2. Roux-en-Y achieves diabetes remission in majority of patients
To take the posttest, go to: https://bit.ly/2x9hLnE
Expires February 6, 2020
3. Socioeconomic status, race found to impact CPAP compliance
To take the posttest, go to: https://bit.ly/2RBpLa9
Expires February 8, 2020
4. Exercise type matters for fall prevention among elderly
To take the posttest, go to: https://bit.ly/2X26OUh
Expires February 12, 2020
5. Adult HIV patients should receive standard vaccinations, with caveats
To take the posttest, go to: https://bit.ly/2X1S7LV
Expires February 12, 2020
ACIP approves meningococcal booster for persons at increased risk
according to the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The committee voted unanimously in favor of a booster dose of MenB vaccine 1 year after completion of the primary series, with additional boosters every 2-3 years “for as long as risk remains” for high-risk persons, including microbiologists and persons with complement deficiency, complement inhibitor use, or asplenia.
The committee also voted unanimously in favor of a one-time MenB booster for individuals aged 10 years and older who are at least a year beyond completion of a MenB primary series and deemed at increased risk by public health officials in an outbreak situation.
In addition, “a booster dose interval of 6 months or more may be considered by public health officials depending on the specific outbreak, vaccine strategy, and projected duration of elevated risk” according to the language, which was included in the unanimously approved statement “Meningococcal Vaccination: Recommendations of The Advisory Committee on Immunization Practices.”
The updated statement on meningococcal vaccination was developed in 2019 “to consolidate all existing ACIP recommendations for MenACWY and MenB vaccines in a single document,” said Sarah Mbaeyi, MD, of the CDC’s National Center for Immunization and Respiratory Diseases, who presented immunogenicity data and the proposed recommendations.
The statement includes the recommendation of a MenB primary series for individuals aged 16-23 years based on shared clinical decision making. Kelly Moore, MD, of Vanderbilt University, Nashville, Tenn., noted the importance of ongoing data collection, and said clinicians must make clear to patients that, “if they want protection, they need the booster.”
Approximately 7% of serogroup B cases in the United States are related to disease outbreaks, mainly among college students, Dr. Mbaeyi said. All 13 universities that experienced outbreaks between 2013 and 2019 have implemented a MenB primary series, and one university has implemented an off-label booster program.
The work group concluded that a MenB booster dose is necessary to sustain protection against serogroup B disease in persons at increased risk during an outbreak, and that the potential benefits outweighed the harms given the seriousness of meningococcal disease.
Paul Hunter, MD, of the City of Milwaukee Health Department, noted that “the booster recommendation gives more flexibility” in an outbreak response.
The committee also voted unanimously to approve the Vaccines for Children resolution for the meningococcal vaccine that updates language to align with the new recommendations.
The ACIP members had no financial conflicts to disclose.
according to the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The committee voted unanimously in favor of a booster dose of MenB vaccine 1 year after completion of the primary series, with additional boosters every 2-3 years “for as long as risk remains” for high-risk persons, including microbiologists and persons with complement deficiency, complement inhibitor use, or asplenia.
The committee also voted unanimously in favor of a one-time MenB booster for individuals aged 10 years and older who are at least a year beyond completion of a MenB primary series and deemed at increased risk by public health officials in an outbreak situation.
In addition, “a booster dose interval of 6 months or more may be considered by public health officials depending on the specific outbreak, vaccine strategy, and projected duration of elevated risk” according to the language, which was included in the unanimously approved statement “Meningococcal Vaccination: Recommendations of The Advisory Committee on Immunization Practices.”
The updated statement on meningococcal vaccination was developed in 2019 “to consolidate all existing ACIP recommendations for MenACWY and MenB vaccines in a single document,” said Sarah Mbaeyi, MD, of the CDC’s National Center for Immunization and Respiratory Diseases, who presented immunogenicity data and the proposed recommendations.
The statement includes the recommendation of a MenB primary series for individuals aged 16-23 years based on shared clinical decision making. Kelly Moore, MD, of Vanderbilt University, Nashville, Tenn., noted the importance of ongoing data collection, and said clinicians must make clear to patients that, “if they want protection, they need the booster.”
Approximately 7% of serogroup B cases in the United States are related to disease outbreaks, mainly among college students, Dr. Mbaeyi said. All 13 universities that experienced outbreaks between 2013 and 2019 have implemented a MenB primary series, and one university has implemented an off-label booster program.
The work group concluded that a MenB booster dose is necessary to sustain protection against serogroup B disease in persons at increased risk during an outbreak, and that the potential benefits outweighed the harms given the seriousness of meningococcal disease.
Paul Hunter, MD, of the City of Milwaukee Health Department, noted that “the booster recommendation gives more flexibility” in an outbreak response.
The committee also voted unanimously to approve the Vaccines for Children resolution for the meningococcal vaccine that updates language to align with the new recommendations.
The ACIP members had no financial conflicts to disclose.
according to the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The committee voted unanimously in favor of a booster dose of MenB vaccine 1 year after completion of the primary series, with additional boosters every 2-3 years “for as long as risk remains” for high-risk persons, including microbiologists and persons with complement deficiency, complement inhibitor use, or asplenia.
The committee also voted unanimously in favor of a one-time MenB booster for individuals aged 10 years and older who are at least a year beyond completion of a MenB primary series and deemed at increased risk by public health officials in an outbreak situation.
In addition, “a booster dose interval of 6 months or more may be considered by public health officials depending on the specific outbreak, vaccine strategy, and projected duration of elevated risk” according to the language, which was included in the unanimously approved statement “Meningococcal Vaccination: Recommendations of The Advisory Committee on Immunization Practices.”
The updated statement on meningococcal vaccination was developed in 2019 “to consolidate all existing ACIP recommendations for MenACWY and MenB vaccines in a single document,” said Sarah Mbaeyi, MD, of the CDC’s National Center for Immunization and Respiratory Diseases, who presented immunogenicity data and the proposed recommendations.
The statement includes the recommendation of a MenB primary series for individuals aged 16-23 years based on shared clinical decision making. Kelly Moore, MD, of Vanderbilt University, Nashville, Tenn., noted the importance of ongoing data collection, and said clinicians must make clear to patients that, “if they want protection, they need the booster.”
Approximately 7% of serogroup B cases in the United States are related to disease outbreaks, mainly among college students, Dr. Mbaeyi said. All 13 universities that experienced outbreaks between 2013 and 2019 have implemented a MenB primary series, and one university has implemented an off-label booster program.
The work group concluded that a MenB booster dose is necessary to sustain protection against serogroup B disease in persons at increased risk during an outbreak, and that the potential benefits outweighed the harms given the seriousness of meningococcal disease.
Paul Hunter, MD, of the City of Milwaukee Health Department, noted that “the booster recommendation gives more flexibility” in an outbreak response.
The committee also voted unanimously to approve the Vaccines for Children resolution for the meningococcal vaccine that updates language to align with the new recommendations.
The ACIP members had no financial conflicts to disclose.
REPORTING FROM AN ACIP MEETING
ACIP approves flu vaccine recommendations for 2019-2020 season
All individuals aged 6 months and older should receive the influenza vaccine by the end of October next season, according to the Centers for Disease Control and Prevention’s Committee on Immunization Practices. The committee voted unanimously to accept minor updates to the ACIP flu recommendations for the 2019-2020 season, but no major changes were made from recent years.
The past flu season was moderate overall, but notable for two waves of viral infections of similar magnitude, one with H1N1 and another with H3N2, said Lynette Brewer of the CDC’s National Center for Immunization and Respiratory Diseases, who presented data on last year’s flu activity.
Last year’s vaccine likely prevented between 40,000 and 90,000 hospitalizations, but mostly reduced the burden of H1N1 disease and provided no real protection against H3N2, she said.
The recommended H3N2 component for next season is A/Kansas/14/2017–like virus, which is genetically similar to the H3N2 that circulated last year.
Lisa Grohskopf, MD, of the CDC’s influenza division, presented the minor adjustments that included the changes in vaccine composition for next year, some licensure changes, and a new table summarizing dose volumes. Also, language was changed to advise vaccination for all eligible individuals by the end of October, and individuals who need two doses should have the first one as soon as it becomes available, in July or August if possible. The updated language also clarified that 8 year olds who need two doses should receive the second dose, even if they turn 9 between the two doses.
Additional guidance updates approved by the committee included harmonizing language on groups that should be the focus of vaccination in the event of limited supply to be more consistent with the 2011 ACIP Recommendations for the Immunization of Health Care Personnel.
The committee also voted unanimously to accept the proposed influenza vaccine in the Vaccines for Children program; there were no changes in recommended dosing intervals, dosages, contraindications, or precautions, according to Frank Whitlach of the National Center for Immunization and Respiratory Diseases, who presented the Vaccines for Children information.
The ACIP members had no financial conflicts to disclose.
All individuals aged 6 months and older should receive the influenza vaccine by the end of October next season, according to the Centers for Disease Control and Prevention’s Committee on Immunization Practices. The committee voted unanimously to accept minor updates to the ACIP flu recommendations for the 2019-2020 season, but no major changes were made from recent years.
The past flu season was moderate overall, but notable for two waves of viral infections of similar magnitude, one with H1N1 and another with H3N2, said Lynette Brewer of the CDC’s National Center for Immunization and Respiratory Diseases, who presented data on last year’s flu activity.
Last year’s vaccine likely prevented between 40,000 and 90,000 hospitalizations, but mostly reduced the burden of H1N1 disease and provided no real protection against H3N2, she said.
The recommended H3N2 component for next season is A/Kansas/14/2017–like virus, which is genetically similar to the H3N2 that circulated last year.
Lisa Grohskopf, MD, of the CDC’s influenza division, presented the minor adjustments that included the changes in vaccine composition for next year, some licensure changes, and a new table summarizing dose volumes. Also, language was changed to advise vaccination for all eligible individuals by the end of October, and individuals who need two doses should have the first one as soon as it becomes available, in July or August if possible. The updated language also clarified that 8 year olds who need two doses should receive the second dose, even if they turn 9 between the two doses.
Additional guidance updates approved by the committee included harmonizing language on groups that should be the focus of vaccination in the event of limited supply to be more consistent with the 2011 ACIP Recommendations for the Immunization of Health Care Personnel.
The committee also voted unanimously to accept the proposed influenza vaccine in the Vaccines for Children program; there were no changes in recommended dosing intervals, dosages, contraindications, or precautions, according to Frank Whitlach of the National Center for Immunization and Respiratory Diseases, who presented the Vaccines for Children information.
The ACIP members had no financial conflicts to disclose.
All individuals aged 6 months and older should receive the influenza vaccine by the end of October next season, according to the Centers for Disease Control and Prevention’s Committee on Immunization Practices. The committee voted unanimously to accept minor updates to the ACIP flu recommendations for the 2019-2020 season, but no major changes were made from recent years.
The past flu season was moderate overall, but notable for two waves of viral infections of similar magnitude, one with H1N1 and another with H3N2, said Lynette Brewer of the CDC’s National Center for Immunization and Respiratory Diseases, who presented data on last year’s flu activity.
Last year’s vaccine likely prevented between 40,000 and 90,000 hospitalizations, but mostly reduced the burden of H1N1 disease and provided no real protection against H3N2, she said.
The recommended H3N2 component for next season is A/Kansas/14/2017–like virus, which is genetically similar to the H3N2 that circulated last year.
Lisa Grohskopf, MD, of the CDC’s influenza division, presented the minor adjustments that included the changes in vaccine composition for next year, some licensure changes, and a new table summarizing dose volumes. Also, language was changed to advise vaccination for all eligible individuals by the end of October, and individuals who need two doses should have the first one as soon as it becomes available, in July or August if possible. The updated language also clarified that 8 year olds who need two doses should receive the second dose, even if they turn 9 between the two doses.
Additional guidance updates approved by the committee included harmonizing language on groups that should be the focus of vaccination in the event of limited supply to be more consistent with the 2011 ACIP Recommendations for the Immunization of Health Care Personnel.
The committee also voted unanimously to accept the proposed influenza vaccine in the Vaccines for Children program; there were no changes in recommended dosing intervals, dosages, contraindications, or precautions, according to Frank Whitlach of the National Center for Immunization and Respiratory Diseases, who presented the Vaccines for Children information.
The ACIP members had no financial conflicts to disclose.
REPORTING FROM AN ACIP MEETING
ACIP endorses catch-up hepatitis A vaccinations
The Centers for Disease Control and Prevention’s Committee on Immunization Practices voted unanimously in support of three recommendations for the use of hepatitis A vaccines.
The committee recommended catch-up vaccination at any age for all children aged 2-18 years who had not previously received hepatitis A vaccination, recommended that all persons with HIV aged 1 year and older should be vaccinated with the hepatitis A vaccine, and approved updating the language in the full hepatitis A vaccine statement, “Prevention of Hepatitis A Virus Infection in The United States: Recommendations of The Advisory Committee on Immunization Practices.”
Catch-up vaccination will expand coverage to adolescents who might have missed it, and data show that the vaccine effectiveness is high, and the rates of adverse events are low in the child and adolescent population, said Noele Nelson, MD, of the CDC’s National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, who presented the recommendations to the committee. “Recent outbreaks are occurring primarily among adults,” and many cases are among persons who use drugs or are homeless, she added.
Several committee members noted that the specific recommendations for catch-up in children and teens and for vaccination of HIV patients offer more opportunities for protection than risk-based recommendations. Catching up with vaccinating adolescents is “more effective than tracking down high-risk adults later in life,” noted Grace Lee, MD, of Lucile Packard Children’s Hospital at Stanford, Calif.
The committee also recommended that all persons with HIV aged 1 year and older should be vaccinated with the hepatitis A vaccine. Data on persons with HIV show that approximately 60% have at least one risk factor for hepatitis A, such as men who have sex with men or individuals engaged in intravenous drug use, said Dr. Nelson. Data also show that individuals with HIV are at increased risk for complications if they get hepatitis A.
The committee’s approval of the full hepatitis A vaccine statement included one notable change – the removal of clotting factor disorders as a high-risk group. The risk has decreased over time based on improvements such as better screening of source plasma, and this group is now at no greater risk than the general population, according to work group chair Kelly Moore, MD, of Vanderbilt University, Nashville, Tenn.
The ACIP members had no financial conflicts to disclose.
The Centers for Disease Control and Prevention’s Committee on Immunization Practices voted unanimously in support of three recommendations for the use of hepatitis A vaccines.
The committee recommended catch-up vaccination at any age for all children aged 2-18 years who had not previously received hepatitis A vaccination, recommended that all persons with HIV aged 1 year and older should be vaccinated with the hepatitis A vaccine, and approved updating the language in the full hepatitis A vaccine statement, “Prevention of Hepatitis A Virus Infection in The United States: Recommendations of The Advisory Committee on Immunization Practices.”
Catch-up vaccination will expand coverage to adolescents who might have missed it, and data show that the vaccine effectiveness is high, and the rates of adverse events are low in the child and adolescent population, said Noele Nelson, MD, of the CDC’s National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, who presented the recommendations to the committee. “Recent outbreaks are occurring primarily among adults,” and many cases are among persons who use drugs or are homeless, she added.
Several committee members noted that the specific recommendations for catch-up in children and teens and for vaccination of HIV patients offer more opportunities for protection than risk-based recommendations. Catching up with vaccinating adolescents is “more effective than tracking down high-risk adults later in life,” noted Grace Lee, MD, of Lucile Packard Children’s Hospital at Stanford, Calif.
The committee also recommended that all persons with HIV aged 1 year and older should be vaccinated with the hepatitis A vaccine. Data on persons with HIV show that approximately 60% have at least one risk factor for hepatitis A, such as men who have sex with men or individuals engaged in intravenous drug use, said Dr. Nelson. Data also show that individuals with HIV are at increased risk for complications if they get hepatitis A.
The committee’s approval of the full hepatitis A vaccine statement included one notable change – the removal of clotting factor disorders as a high-risk group. The risk has decreased over time based on improvements such as better screening of source plasma, and this group is now at no greater risk than the general population, according to work group chair Kelly Moore, MD, of Vanderbilt University, Nashville, Tenn.
The ACIP members had no financial conflicts to disclose.
The Centers for Disease Control and Prevention’s Committee on Immunization Practices voted unanimously in support of three recommendations for the use of hepatitis A vaccines.
The committee recommended catch-up vaccination at any age for all children aged 2-18 years who had not previously received hepatitis A vaccination, recommended that all persons with HIV aged 1 year and older should be vaccinated with the hepatitis A vaccine, and approved updating the language in the full hepatitis A vaccine statement, “Prevention of Hepatitis A Virus Infection in The United States: Recommendations of The Advisory Committee on Immunization Practices.”
Catch-up vaccination will expand coverage to adolescents who might have missed it, and data show that the vaccine effectiveness is high, and the rates of adverse events are low in the child and adolescent population, said Noele Nelson, MD, of the CDC’s National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, who presented the recommendations to the committee. “Recent outbreaks are occurring primarily among adults,” and many cases are among persons who use drugs or are homeless, she added.
Several committee members noted that the specific recommendations for catch-up in children and teens and for vaccination of HIV patients offer more opportunities for protection than risk-based recommendations. Catching up with vaccinating adolescents is “more effective than tracking down high-risk adults later in life,” noted Grace Lee, MD, of Lucile Packard Children’s Hospital at Stanford, Calif.
The committee also recommended that all persons with HIV aged 1 year and older should be vaccinated with the hepatitis A vaccine. Data on persons with HIV show that approximately 60% have at least one risk factor for hepatitis A, such as men who have sex with men or individuals engaged in intravenous drug use, said Dr. Nelson. Data also show that individuals with HIV are at increased risk for complications if they get hepatitis A.
The committee’s approval of the full hepatitis A vaccine statement included one notable change – the removal of clotting factor disorders as a high-risk group. The risk has decreased over time based on improvements such as better screening of source plasma, and this group is now at no greater risk than the general population, according to work group chair Kelly Moore, MD, of Vanderbilt University, Nashville, Tenn.
The ACIP members had no financial conflicts to disclose.
REPORTING FROM AN ACIP MEETING
ACIP adds hexavalent vaccine to VFC program
The pediatric hexavalent vaccine (DTaP-[inactivated poliovirus] IPV-[hepatitis B] HepB-[Haemophilis influenzae type b] Hib) should be included as an option in the Vaccines for Children (VFC) program for the infant series at ages 2, 4, and 6 months, according to unanimous votes at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The addition of the vaccine to the VFC program required no motions on the part of the committee, but involved separate votes on each component of the vaccine.
Combination vaccination has been associated with increased coverage and more likely completion of the full infant vaccine series, said Sara Oliver, MD, of the CDC’s National Center for Immunization and Respiratory Diseases.
The new vaccine is being developed jointly by Sanofi and Merck, and has been approved by the Food and Drug Administration for use in children through age 4 years.
Dr. Oliver presented evidence that the safety profile of the combination vaccine is consistent with that of the component vaccines. In addition, “use of combination vaccines can reduce the number of injections patient receive and alleviate concern associated with the number of injections,” she said. However, “considerations should include provider assessment, patient preference, and the potential for adverse events.”
although it will not be available until 2021 in order to ensure sufficient supply, Dr. Oliver noted.
The combination vaccination work group considered whether the new vaccine should be preferentially recommended for American Indian and Alaskan Native populations, but they concluded that post–dose one immunogenicity data are needed before such a preferential recommendation can be made.
The ACIP members had no financial conflicts to disclose.
The pediatric hexavalent vaccine (DTaP-[inactivated poliovirus] IPV-[hepatitis B] HepB-[Haemophilis influenzae type b] Hib) should be included as an option in the Vaccines for Children (VFC) program for the infant series at ages 2, 4, and 6 months, according to unanimous votes at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The addition of the vaccine to the VFC program required no motions on the part of the committee, but involved separate votes on each component of the vaccine.
Combination vaccination has been associated with increased coverage and more likely completion of the full infant vaccine series, said Sara Oliver, MD, of the CDC’s National Center for Immunization and Respiratory Diseases.
The new vaccine is being developed jointly by Sanofi and Merck, and has been approved by the Food and Drug Administration for use in children through age 4 years.
Dr. Oliver presented evidence that the safety profile of the combination vaccine is consistent with that of the component vaccines. In addition, “use of combination vaccines can reduce the number of injections patient receive and alleviate concern associated with the number of injections,” she said. However, “considerations should include provider assessment, patient preference, and the potential for adverse events.”
although it will not be available until 2021 in order to ensure sufficient supply, Dr. Oliver noted.
The combination vaccination work group considered whether the new vaccine should be preferentially recommended for American Indian and Alaskan Native populations, but they concluded that post–dose one immunogenicity data are needed before such a preferential recommendation can be made.
The ACIP members had no financial conflicts to disclose.
The pediatric hexavalent vaccine (DTaP-[inactivated poliovirus] IPV-[hepatitis B] HepB-[Haemophilis influenzae type b] Hib) should be included as an option in the Vaccines for Children (VFC) program for the infant series at ages 2, 4, and 6 months, according to unanimous votes at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The addition of the vaccine to the VFC program required no motions on the part of the committee, but involved separate votes on each component of the vaccine.
Combination vaccination has been associated with increased coverage and more likely completion of the full infant vaccine series, said Sara Oliver, MD, of the CDC’s National Center for Immunization and Respiratory Diseases.
The new vaccine is being developed jointly by Sanofi and Merck, and has been approved by the Food and Drug Administration for use in children through age 4 years.
Dr. Oliver presented evidence that the safety profile of the combination vaccine is consistent with that of the component vaccines. In addition, “use of combination vaccines can reduce the number of injections patient receive and alleviate concern associated with the number of injections,” she said. However, “considerations should include provider assessment, patient preference, and the potential for adverse events.”
although it will not be available until 2021 in order to ensure sufficient supply, Dr. Oliver noted.
The combination vaccination work group considered whether the new vaccine should be preferentially recommended for American Indian and Alaskan Native populations, but they concluded that post–dose one immunogenicity data are needed before such a preferential recommendation can be made.
The ACIP members had no financial conflicts to disclose.
REPORTING FROM AN ACIP MEETING
ACIP favors shared decision on pneumococcal vaccine for older adults
Pneumococcal vaccination with the 13-valent pneumococcal conjugate vaccine (PCV13) based on shared clinical decision making is recommended for immunocompetent adults aged 65 years and older who have not previously received PCV13, and all adults aged 65 years and older should continue to receive the pneumococcal polysaccharide vaccine (PPSV23), according to a vote at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The motion passed with an 11-1 vote after members voted down two other options to either discontinue or continue the current recommendation of PCV13 for all immunocompetent adults aged 65 years and older. The current recommendation for PCV13 for adults aged 65 years and older has been in place since 2014.
The pneumococcal work group assessed indirect effects of the pediatric PCV vaccination on older adults prior to 2014 and since 2014, and what additional benefits might be expected if routine vaccination of older adults continued.
“Indirect effects have been observed in all age groups” said Almea Matanock, MD, of the CDC’s National Center for Immunization and Respiratory Diseases. Although there were no safety concerns, the public health impact of continued vaccination of adults was minimal.
Although PCV13 resulted in a 75% reduction in vaccine-type invasive pneumococcal disease and a 45% reduction in vaccine-type nonbacteremic pneumonia in 2014, the annual number needed to vaccinate to prevent a single case of outpatient pneumonia was 2,600, said Dr. Matanock.
Dr. Matanock presented key issues from the Evidence to Recommendations Framework for and against the recommendation for PCV13 in older adults. Work group comments in favor of continuing the recommendation for PCV13 in older adults included effective disease prevention and the potential negative impact on the importance of adult vaccines if the vaccine was no longer recommended. However, some work group members and committee members expressed concern about resource allocation and steering vaccines away from younger age groups in whom they have been more consistently effective.
Paul Hunter, MD, of the City of Milwaukee Health Department, voted against the shared clinical decision making, and instead favored discontinuing the recommendation for PCV13 for older adults. “I think clinicians need a clear message,” he said, adding that “the public health bang for the buck is with the kids.”
“I think there was a recognition that the population level benefit is minimal,” said work group chair Grace Lee, MD.
Although the work group recognized some benefit for older adults, the burden of disease for PCV-specific disease is low, compared with all-cause pneumonia, said Dr. Lee of Lucile Packard Children’s Hospital at Stanford, Calif. However, the recommendation for shared clinical decision making allows for potential insurance coverage of the vaccine for adults who decide after discussion with their health care provider that they would benefit.
“We are still unpacking this construct” of shared clinical decision making, which in this case applies to adults without immunocompromising conditions, and is more of a provider assessment than a risk assessment, she said.
The ACIP members had no financial conflicts to disclose.
Pneumococcal vaccination with the 13-valent pneumococcal conjugate vaccine (PCV13) based on shared clinical decision making is recommended for immunocompetent adults aged 65 years and older who have not previously received PCV13, and all adults aged 65 years and older should continue to receive the pneumococcal polysaccharide vaccine (PPSV23), according to a vote at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The motion passed with an 11-1 vote after members voted down two other options to either discontinue or continue the current recommendation of PCV13 for all immunocompetent adults aged 65 years and older. The current recommendation for PCV13 for adults aged 65 years and older has been in place since 2014.
The pneumococcal work group assessed indirect effects of the pediatric PCV vaccination on older adults prior to 2014 and since 2014, and what additional benefits might be expected if routine vaccination of older adults continued.
“Indirect effects have been observed in all age groups” said Almea Matanock, MD, of the CDC’s National Center for Immunization and Respiratory Diseases. Although there were no safety concerns, the public health impact of continued vaccination of adults was minimal.
Although PCV13 resulted in a 75% reduction in vaccine-type invasive pneumococcal disease and a 45% reduction in vaccine-type nonbacteremic pneumonia in 2014, the annual number needed to vaccinate to prevent a single case of outpatient pneumonia was 2,600, said Dr. Matanock.
Dr. Matanock presented key issues from the Evidence to Recommendations Framework for and against the recommendation for PCV13 in older adults. Work group comments in favor of continuing the recommendation for PCV13 in older adults included effective disease prevention and the potential negative impact on the importance of adult vaccines if the vaccine was no longer recommended. However, some work group members and committee members expressed concern about resource allocation and steering vaccines away from younger age groups in whom they have been more consistently effective.
Paul Hunter, MD, of the City of Milwaukee Health Department, voted against the shared clinical decision making, and instead favored discontinuing the recommendation for PCV13 for older adults. “I think clinicians need a clear message,” he said, adding that “the public health bang for the buck is with the kids.”
“I think there was a recognition that the population level benefit is minimal,” said work group chair Grace Lee, MD.
Although the work group recognized some benefit for older adults, the burden of disease for PCV-specific disease is low, compared with all-cause pneumonia, said Dr. Lee of Lucile Packard Children’s Hospital at Stanford, Calif. However, the recommendation for shared clinical decision making allows for potential insurance coverage of the vaccine for adults who decide after discussion with their health care provider that they would benefit.
“We are still unpacking this construct” of shared clinical decision making, which in this case applies to adults without immunocompromising conditions, and is more of a provider assessment than a risk assessment, she said.
The ACIP members had no financial conflicts to disclose.
Pneumococcal vaccination with the 13-valent pneumococcal conjugate vaccine (PCV13) based on shared clinical decision making is recommended for immunocompetent adults aged 65 years and older who have not previously received PCV13, and all adults aged 65 years and older should continue to receive the pneumococcal polysaccharide vaccine (PPSV23), according to a vote at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
The motion passed with an 11-1 vote after members voted down two other options to either discontinue or continue the current recommendation of PCV13 for all immunocompetent adults aged 65 years and older. The current recommendation for PCV13 for adults aged 65 years and older has been in place since 2014.
The pneumococcal work group assessed indirect effects of the pediatric PCV vaccination on older adults prior to 2014 and since 2014, and what additional benefits might be expected if routine vaccination of older adults continued.
“Indirect effects have been observed in all age groups” said Almea Matanock, MD, of the CDC’s National Center for Immunization and Respiratory Diseases. Although there were no safety concerns, the public health impact of continued vaccination of adults was minimal.
Although PCV13 resulted in a 75% reduction in vaccine-type invasive pneumococcal disease and a 45% reduction in vaccine-type nonbacteremic pneumonia in 2014, the annual number needed to vaccinate to prevent a single case of outpatient pneumonia was 2,600, said Dr. Matanock.
Dr. Matanock presented key issues from the Evidence to Recommendations Framework for and against the recommendation for PCV13 in older adults. Work group comments in favor of continuing the recommendation for PCV13 in older adults included effective disease prevention and the potential negative impact on the importance of adult vaccines if the vaccine was no longer recommended. However, some work group members and committee members expressed concern about resource allocation and steering vaccines away from younger age groups in whom they have been more consistently effective.
Paul Hunter, MD, of the City of Milwaukee Health Department, voted against the shared clinical decision making, and instead favored discontinuing the recommendation for PCV13 for older adults. “I think clinicians need a clear message,” he said, adding that “the public health bang for the buck is with the kids.”
“I think there was a recognition that the population level benefit is minimal,” said work group chair Grace Lee, MD.
Although the work group recognized some benefit for older adults, the burden of disease for PCV-specific disease is low, compared with all-cause pneumonia, said Dr. Lee of Lucile Packard Children’s Hospital at Stanford, Calif. However, the recommendation for shared clinical decision making allows for potential insurance coverage of the vaccine for adults who decide after discussion with their health care provider that they would benefit.
“We are still unpacking this construct” of shared clinical decision making, which in this case applies to adults without immunocompromising conditions, and is more of a provider assessment than a risk assessment, she said.
The ACIP members had no financial conflicts to disclose.
REPORTING FROM AN ACIP MEETING
ACIP extends HPV vaccine coverage
according to a unanimous vote at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
This change affects males aged 22 through 26 years; the HPV vaccine is currently recommended for males and females aged 11 or 12 years, with catch-up vaccination through age 21 for males and age 26 for females.
The change was supported in part by increased interest in simplifying and harmonizing the vaccine schedule, said Lauri Markowitz, MD, of the CDC’s National Center for Immunization and Respiratory Diseases (NCIRD), who presented the HPV work group’s considerations.
In addition, the committee voted 10-4 in favor of catch-up HPV vaccination, based on shared clinical decision making, for all adults aged 27 through 45 years.
Although the current program of HPV vaccination for youth has demonstrated effectiveness, data from multiple models suggest that widespread HPV vaccination for adults older than 26 years is much less cost effective, and would yield relatively small additional health benefits, Dr. Markowitz said.
The HPV work group reviewed data from a range of clinical trials, epidemiology, and natural history, as well as results from five different health economic models. They concluded that an assessment of benefits and harms favors expanding the catch-up vaccination to all individuals through 26 years, said Elissa Meites, MD, of the CDC, who presented the official work group opinion. The group’s opinion on the second question was that the additional population level benefit of expanding HPV vaccination to all adults would be minimal and not a reasonable and effective allocation of resources, but that shared clinical decision making would allow flexibility.
The committee expressed strong opinions about the potential for shared clinical decision making as a policy for vaccination for adults older than 26 years. Some felt that this option was a way to include adults at risk for HPV, such as divorced women with new partners, or women getting married for the first time later in life who might not have been exposed to HPV through other relationships. In addition, supporters noted that the shared clinical decision-making option would allow for potential insurance coverage, and would involve discussion between doctors and patients to assess risk.
However, other committee members felt that any recommendation for older adult vaccination would distract clinicians from the importance and value of HPV vaccination for the target age group of 11- and 12-year-olds, and might divert resources from the younger age group in whom it has shown the most benefit.
Resource allocation was a concern voiced by many committee members. Kelly Moore, MD, MPH, of Vanderbilt University, Nashville, Tenn., said she voted no on expanding vaccination to older adults because “we didn’t have details on shared clinical decision making, in the absence of information on what that meant, and in the presence of supply questions, I didn’t feel comfortable expanding vaccination to a huge population,” she said.
Paul Hunter, MD, of the City of Milwaukee Health Department, also voted no, and expressed concern that expanding the HPV vaccination recommendations to older adults would send the message that vaccination for children and teens is not effective or important.
The text of the new recommendations for routine and catch-up vaccination states that the recommendations “also apply to MSM [men who have sex with men], transgender people, and people with immunocompromising conditions.”
The ACIP members had no financial conflicts to disclose.
according to a unanimous vote at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
This change affects males aged 22 through 26 years; the HPV vaccine is currently recommended for males and females aged 11 or 12 years, with catch-up vaccination through age 21 for males and age 26 for females.
The change was supported in part by increased interest in simplifying and harmonizing the vaccine schedule, said Lauri Markowitz, MD, of the CDC’s National Center for Immunization and Respiratory Diseases (NCIRD), who presented the HPV work group’s considerations.
In addition, the committee voted 10-4 in favor of catch-up HPV vaccination, based on shared clinical decision making, for all adults aged 27 through 45 years.
Although the current program of HPV vaccination for youth has demonstrated effectiveness, data from multiple models suggest that widespread HPV vaccination for adults older than 26 years is much less cost effective, and would yield relatively small additional health benefits, Dr. Markowitz said.
The HPV work group reviewed data from a range of clinical trials, epidemiology, and natural history, as well as results from five different health economic models. They concluded that an assessment of benefits and harms favors expanding the catch-up vaccination to all individuals through 26 years, said Elissa Meites, MD, of the CDC, who presented the official work group opinion. The group’s opinion on the second question was that the additional population level benefit of expanding HPV vaccination to all adults would be minimal and not a reasonable and effective allocation of resources, but that shared clinical decision making would allow flexibility.
The committee expressed strong opinions about the potential for shared clinical decision making as a policy for vaccination for adults older than 26 years. Some felt that this option was a way to include adults at risk for HPV, such as divorced women with new partners, or women getting married for the first time later in life who might not have been exposed to HPV through other relationships. In addition, supporters noted that the shared clinical decision-making option would allow for potential insurance coverage, and would involve discussion between doctors and patients to assess risk.
However, other committee members felt that any recommendation for older adult vaccination would distract clinicians from the importance and value of HPV vaccination for the target age group of 11- and 12-year-olds, and might divert resources from the younger age group in whom it has shown the most benefit.
Resource allocation was a concern voiced by many committee members. Kelly Moore, MD, MPH, of Vanderbilt University, Nashville, Tenn., said she voted no on expanding vaccination to older adults because “we didn’t have details on shared clinical decision making, in the absence of information on what that meant, and in the presence of supply questions, I didn’t feel comfortable expanding vaccination to a huge population,” she said.
Paul Hunter, MD, of the City of Milwaukee Health Department, also voted no, and expressed concern that expanding the HPV vaccination recommendations to older adults would send the message that vaccination for children and teens is not effective or important.
The text of the new recommendations for routine and catch-up vaccination states that the recommendations “also apply to MSM [men who have sex with men], transgender people, and people with immunocompromising conditions.”
The ACIP members had no financial conflicts to disclose.
according to a unanimous vote at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
This change affects males aged 22 through 26 years; the HPV vaccine is currently recommended for males and females aged 11 or 12 years, with catch-up vaccination through age 21 for males and age 26 for females.
The change was supported in part by increased interest in simplifying and harmonizing the vaccine schedule, said Lauri Markowitz, MD, of the CDC’s National Center for Immunization and Respiratory Diseases (NCIRD), who presented the HPV work group’s considerations.
In addition, the committee voted 10-4 in favor of catch-up HPV vaccination, based on shared clinical decision making, for all adults aged 27 through 45 years.
Although the current program of HPV vaccination for youth has demonstrated effectiveness, data from multiple models suggest that widespread HPV vaccination for adults older than 26 years is much less cost effective, and would yield relatively small additional health benefits, Dr. Markowitz said.
The HPV work group reviewed data from a range of clinical trials, epidemiology, and natural history, as well as results from five different health economic models. They concluded that an assessment of benefits and harms favors expanding the catch-up vaccination to all individuals through 26 years, said Elissa Meites, MD, of the CDC, who presented the official work group opinion. The group’s opinion on the second question was that the additional population level benefit of expanding HPV vaccination to all adults would be minimal and not a reasonable and effective allocation of resources, but that shared clinical decision making would allow flexibility.
The committee expressed strong opinions about the potential for shared clinical decision making as a policy for vaccination for adults older than 26 years. Some felt that this option was a way to include adults at risk for HPV, such as divorced women with new partners, or women getting married for the first time later in life who might not have been exposed to HPV through other relationships. In addition, supporters noted that the shared clinical decision-making option would allow for potential insurance coverage, and would involve discussion between doctors and patients to assess risk.
However, other committee members felt that any recommendation for older adult vaccination would distract clinicians from the importance and value of HPV vaccination for the target age group of 11- and 12-year-olds, and might divert resources from the younger age group in whom it has shown the most benefit.
Resource allocation was a concern voiced by many committee members. Kelly Moore, MD, MPH, of Vanderbilt University, Nashville, Tenn., said she voted no on expanding vaccination to older adults because “we didn’t have details on shared clinical decision making, in the absence of information on what that meant, and in the presence of supply questions, I didn’t feel comfortable expanding vaccination to a huge population,” she said.
Paul Hunter, MD, of the City of Milwaukee Health Department, also voted no, and expressed concern that expanding the HPV vaccination recommendations to older adults would send the message that vaccination for children and teens is not effective or important.
The text of the new recommendations for routine and catch-up vaccination states that the recommendations “also apply to MSM [men who have sex with men], transgender people, and people with immunocompromising conditions.”
The ACIP members had no financial conflicts to disclose.
REPORTING FROM AN ACIP MEETING
Substantial reductions in HPV infections, CIN2+ after vaccination
The introduction of the human papillomavirus according to a meta-analysis of data from more than 60 million individuals worldwide.
Mélanie Drolet, PhD, from the Centre de recherche du CHU de Québec–Université Laval, and coauthors of the HPV Vaccination Impact Study Group reported the results of a systematic review and meta-analysis of 65 studies showing pre- and postvaccination frequency of at least one HPV-related endpoint published in the Lancet. The studies were conducted in 14 high-income countries, 12 of which were vaccinating only women and girls, with the results at 5-8 years published in the Lancet.
At 5-8 years after a vaccination program was implemented, there was a significant 83% reduction in the prevalence of HPV 16 and 18, both of which are targeted by the vaccine, among girls aged 13-19 years; a 66% reduction among women aged 20-24 years; and a 37% reduction in women aged 25-29 years, even though most of these women were unvaccinated.
There also were significant decreases at 5-8 years in the prevalence of HPV subtypes 31, 33, and 45, which are not included in the vaccine but against which the vaccine appears to offer cross-protection. Among girls aged 13-19 years, there was a significant 54% reduction in the prevalence of these subtypes, among women aged 20-24 years there was a nonsignificant 28% decrease, but among women aged 25-29 years, there was no significant decrease.
The analysis also found significant declines in the prevalence of cervical intraepithelial neoplasias (CINs) of grade 2 or above. At 5-9 years after vaccination was introduced, CIN2+ decreased by 51% among girls aged 15-19 years who also were screened for cervical cancer, and by 31% among women aged 20-24 years.
However, over the same time period, the rates of CIN2+ increased by a significant 19% among mostly unvaccinated women aged 25-29 years and 23% among mostly unvaccinated women aged 30-39 years, despite both groups being screened for cervical abnormalities.
While most of the countries in the study vaccinated only girls and women, two studies did find nonsignificant decreases in the prevalence of HPV 16, 18, 31, 33, and 45 among boys aged 16-19 years, but not among men aged 20-24 years.
HPV vaccination also was associated with significant declines in the incidence of anogenital warts among both males and females. In the first 4 years alone, vaccination was associated with significant reductions in anogenital wart diagnoses among females aged 15-29 years, as well as nonsignificant but “substantial” reductions in unvaccinated boys aged 15-19 years.
After 5-8 years, anogenital wart diagnoses decreased by 67% among girls aged 15-19 years, significantly by 54% among women aged 20-24 years, and 31% among women aged 25-29 years – all significant changes. Among boys aged 15-19 years, anogenital wart diagnoses decreased by a significant 48%, and among men aged 20-24 years they decreased by a significant 32%.
The decreases in anogenital wart diagnoses were even greater in countries that implemented vaccination among multiple cohorts simultaneously and achieved high vaccination coverage, compared with countries that vaccinated only one cohort at a time or had low routine vaccination coverage.
“Our study is the first to show the real-world additional benefit of multicohort HPV vaccination and high routine vaccination coverage, and the fast and substantial herd effects of vaccination in countries which implement these measures,” wrote Dr. Drolet and coauthors. “The greater impact of multicohort vaccination was similar when restricting the analyses to countries with high routine vaccination coverage.”
They pointed to the World Health Organization’s recently revised position on HPV vaccination, which now recommends vaccination of multiple cohorts of girls aged 9-14 years, although they raised the question of what might be the optimal number of age cohorts. “Number needed to vaccinate and cost-effectiveness analyses in high-income countries suggest that vaccinating multiple cohorts of individuals up to 18 years of age is highly efficient and cost effective.”
This analysis by Drolet et al. “provides compelling evidence for HPV vaccine efficacy on all outcomes explored and for almost all age strata,” Dr. Silvia de Sanjose, of PATH in Seattle, and Dr. Sinead Delany-Moretlwe of the Wits Reproductive Health and HIV Institute at the University of Witwatersrand in Johannesburg, said in an accompanying editorial (Lancet. 2019 Jun 26. doi: 10.1016/ S0140-6736[19]30549-5). This study shows just how effective HPV vaccination can be across a range of outcomes and ages, and also demonstrates the herd immunity benefits, particularly when multiple cohorts are vaccinated and there is high vaccination coverage.
One key limitation of this analysis is the lack of data from low- and middle-income countries. The data by Drolet et al. “emphasise the importance of redoubling our efforts to tackle the fiscal, supply, and programmatic barriers that currently limit HPV vaccine programmes; with these efforts, HPV vaccination could become a hallmark investment of cancer prevention in the 21st century,” Dr. de Sanjose and Dr. Delany-Moretlwe concluded.
The study was funded by WHO, Canadian Institutes of Health Research, and Fonds de recherche du Québec–Santé. No conflicts of interest were declared.
Dr. de Sanjose declared previous institutional support from Merck.
SOURCE: Drolet M et al. Lancet 2019 Jun 26. doi: 10.1016/ S0140-6736(19)30298-3.
The introduction of the human papillomavirus according to a meta-analysis of data from more than 60 million individuals worldwide.
Mélanie Drolet, PhD, from the Centre de recherche du CHU de Québec–Université Laval, and coauthors of the HPV Vaccination Impact Study Group reported the results of a systematic review and meta-analysis of 65 studies showing pre- and postvaccination frequency of at least one HPV-related endpoint published in the Lancet. The studies were conducted in 14 high-income countries, 12 of which were vaccinating only women and girls, with the results at 5-8 years published in the Lancet.
At 5-8 years after a vaccination program was implemented, there was a significant 83% reduction in the prevalence of HPV 16 and 18, both of which are targeted by the vaccine, among girls aged 13-19 years; a 66% reduction among women aged 20-24 years; and a 37% reduction in women aged 25-29 years, even though most of these women were unvaccinated.
There also were significant decreases at 5-8 years in the prevalence of HPV subtypes 31, 33, and 45, which are not included in the vaccine but against which the vaccine appears to offer cross-protection. Among girls aged 13-19 years, there was a significant 54% reduction in the prevalence of these subtypes, among women aged 20-24 years there was a nonsignificant 28% decrease, but among women aged 25-29 years, there was no significant decrease.
The analysis also found significant declines in the prevalence of cervical intraepithelial neoplasias (CINs) of grade 2 or above. At 5-9 years after vaccination was introduced, CIN2+ decreased by 51% among girls aged 15-19 years who also were screened for cervical cancer, and by 31% among women aged 20-24 years.
However, over the same time period, the rates of CIN2+ increased by a significant 19% among mostly unvaccinated women aged 25-29 years and 23% among mostly unvaccinated women aged 30-39 years, despite both groups being screened for cervical abnormalities.
While most of the countries in the study vaccinated only girls and women, two studies did find nonsignificant decreases in the prevalence of HPV 16, 18, 31, 33, and 45 among boys aged 16-19 years, but not among men aged 20-24 years.
HPV vaccination also was associated with significant declines in the incidence of anogenital warts among both males and females. In the first 4 years alone, vaccination was associated with significant reductions in anogenital wart diagnoses among females aged 15-29 years, as well as nonsignificant but “substantial” reductions in unvaccinated boys aged 15-19 years.
After 5-8 years, anogenital wart diagnoses decreased by 67% among girls aged 15-19 years, significantly by 54% among women aged 20-24 years, and 31% among women aged 25-29 years – all significant changes. Among boys aged 15-19 years, anogenital wart diagnoses decreased by a significant 48%, and among men aged 20-24 years they decreased by a significant 32%.
The decreases in anogenital wart diagnoses were even greater in countries that implemented vaccination among multiple cohorts simultaneously and achieved high vaccination coverage, compared with countries that vaccinated only one cohort at a time or had low routine vaccination coverage.
“Our study is the first to show the real-world additional benefit of multicohort HPV vaccination and high routine vaccination coverage, and the fast and substantial herd effects of vaccination in countries which implement these measures,” wrote Dr. Drolet and coauthors. “The greater impact of multicohort vaccination was similar when restricting the analyses to countries with high routine vaccination coverage.”
They pointed to the World Health Organization’s recently revised position on HPV vaccination, which now recommends vaccination of multiple cohorts of girls aged 9-14 years, although they raised the question of what might be the optimal number of age cohorts. “Number needed to vaccinate and cost-effectiveness analyses in high-income countries suggest that vaccinating multiple cohorts of individuals up to 18 years of age is highly efficient and cost effective.”
This analysis by Drolet et al. “provides compelling evidence for HPV vaccine efficacy on all outcomes explored and for almost all age strata,” Dr. Silvia de Sanjose, of PATH in Seattle, and Dr. Sinead Delany-Moretlwe of the Wits Reproductive Health and HIV Institute at the University of Witwatersrand in Johannesburg, said in an accompanying editorial (Lancet. 2019 Jun 26. doi: 10.1016/ S0140-6736[19]30549-5). This study shows just how effective HPV vaccination can be across a range of outcomes and ages, and also demonstrates the herd immunity benefits, particularly when multiple cohorts are vaccinated and there is high vaccination coverage.
One key limitation of this analysis is the lack of data from low- and middle-income countries. The data by Drolet et al. “emphasise the importance of redoubling our efforts to tackle the fiscal, supply, and programmatic barriers that currently limit HPV vaccine programmes; with these efforts, HPV vaccination could become a hallmark investment of cancer prevention in the 21st century,” Dr. de Sanjose and Dr. Delany-Moretlwe concluded.
The study was funded by WHO, Canadian Institutes of Health Research, and Fonds de recherche du Québec–Santé. No conflicts of interest were declared.
Dr. de Sanjose declared previous institutional support from Merck.
SOURCE: Drolet M et al. Lancet 2019 Jun 26. doi: 10.1016/ S0140-6736(19)30298-3.
The introduction of the human papillomavirus according to a meta-analysis of data from more than 60 million individuals worldwide.
Mélanie Drolet, PhD, from the Centre de recherche du CHU de Québec–Université Laval, and coauthors of the HPV Vaccination Impact Study Group reported the results of a systematic review and meta-analysis of 65 studies showing pre- and postvaccination frequency of at least one HPV-related endpoint published in the Lancet. The studies were conducted in 14 high-income countries, 12 of which were vaccinating only women and girls, with the results at 5-8 years published in the Lancet.
At 5-8 years after a vaccination program was implemented, there was a significant 83% reduction in the prevalence of HPV 16 and 18, both of which are targeted by the vaccine, among girls aged 13-19 years; a 66% reduction among women aged 20-24 years; and a 37% reduction in women aged 25-29 years, even though most of these women were unvaccinated.
There also were significant decreases at 5-8 years in the prevalence of HPV subtypes 31, 33, and 45, which are not included in the vaccine but against which the vaccine appears to offer cross-protection. Among girls aged 13-19 years, there was a significant 54% reduction in the prevalence of these subtypes, among women aged 20-24 years there was a nonsignificant 28% decrease, but among women aged 25-29 years, there was no significant decrease.
The analysis also found significant declines in the prevalence of cervical intraepithelial neoplasias (CINs) of grade 2 or above. At 5-9 years after vaccination was introduced, CIN2+ decreased by 51% among girls aged 15-19 years who also were screened for cervical cancer, and by 31% among women aged 20-24 years.
However, over the same time period, the rates of CIN2+ increased by a significant 19% among mostly unvaccinated women aged 25-29 years and 23% among mostly unvaccinated women aged 30-39 years, despite both groups being screened for cervical abnormalities.
While most of the countries in the study vaccinated only girls and women, two studies did find nonsignificant decreases in the prevalence of HPV 16, 18, 31, 33, and 45 among boys aged 16-19 years, but not among men aged 20-24 years.
HPV vaccination also was associated with significant declines in the incidence of anogenital warts among both males and females. In the first 4 years alone, vaccination was associated with significant reductions in anogenital wart diagnoses among females aged 15-29 years, as well as nonsignificant but “substantial” reductions in unvaccinated boys aged 15-19 years.
After 5-8 years, anogenital wart diagnoses decreased by 67% among girls aged 15-19 years, significantly by 54% among women aged 20-24 years, and 31% among women aged 25-29 years – all significant changes. Among boys aged 15-19 years, anogenital wart diagnoses decreased by a significant 48%, and among men aged 20-24 years they decreased by a significant 32%.
The decreases in anogenital wart diagnoses were even greater in countries that implemented vaccination among multiple cohorts simultaneously and achieved high vaccination coverage, compared with countries that vaccinated only one cohort at a time or had low routine vaccination coverage.
“Our study is the first to show the real-world additional benefit of multicohort HPV vaccination and high routine vaccination coverage, and the fast and substantial herd effects of vaccination in countries which implement these measures,” wrote Dr. Drolet and coauthors. “The greater impact of multicohort vaccination was similar when restricting the analyses to countries with high routine vaccination coverage.”
They pointed to the World Health Organization’s recently revised position on HPV vaccination, which now recommends vaccination of multiple cohorts of girls aged 9-14 years, although they raised the question of what might be the optimal number of age cohorts. “Number needed to vaccinate and cost-effectiveness analyses in high-income countries suggest that vaccinating multiple cohorts of individuals up to 18 years of age is highly efficient and cost effective.”
This analysis by Drolet et al. “provides compelling evidence for HPV vaccine efficacy on all outcomes explored and for almost all age strata,” Dr. Silvia de Sanjose, of PATH in Seattle, and Dr. Sinead Delany-Moretlwe of the Wits Reproductive Health and HIV Institute at the University of Witwatersrand in Johannesburg, said in an accompanying editorial (Lancet. 2019 Jun 26. doi: 10.1016/ S0140-6736[19]30549-5). This study shows just how effective HPV vaccination can be across a range of outcomes and ages, and also demonstrates the herd immunity benefits, particularly when multiple cohorts are vaccinated and there is high vaccination coverage.
One key limitation of this analysis is the lack of data from low- and middle-income countries. The data by Drolet et al. “emphasise the importance of redoubling our efforts to tackle the fiscal, supply, and programmatic barriers that currently limit HPV vaccine programmes; with these efforts, HPV vaccination could become a hallmark investment of cancer prevention in the 21st century,” Dr. de Sanjose and Dr. Delany-Moretlwe concluded.
The study was funded by WHO, Canadian Institutes of Health Research, and Fonds de recherche du Québec–Santé. No conflicts of interest were declared.
Dr. de Sanjose declared previous institutional support from Merck.
SOURCE: Drolet M et al. Lancet 2019 Jun 26. doi: 10.1016/ S0140-6736(19)30298-3.
FROM THE LANCET
Key clinical point: Significant declines in HPV infections, CIN2+, and anogenital warts have occurred after the introduction of HPV vaccine programs, some because of herd effects.
Major finding: The HPV vaccination program is associated with a significant 83% reduction in the prevalence of HPV 16 and 18 among girls aged 13-19 years in 14 high-income countries.
Study details: Systematic review and meta-analysis of 65 studies involving more than 60 million individuals in 14 countries.
Disclosures: The study was funded by World Health Organization, Canadian Institutes of Health Research, and Fonds de recherche du Québec – Santé. No conflicts of interest were declared.
Source: Drolet M et al. Lancet 2019 Jun 26. doi: 10.1016/ S0140-6736(19)30298-3.
Almost half of Americans express doubts about vaccines
according to the American Osteopathic Association.
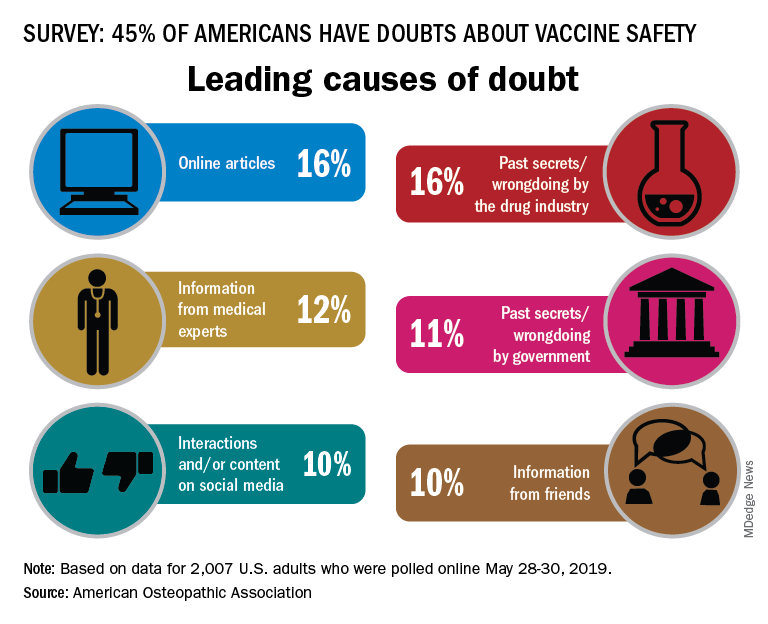
In a survey conducted by the Harris Poll on behalf of the AOA, 45% of the 2,007 respondents expressed a negative attitude towards vaccine safety, with online articles (16%) and past secrets/wrongdoings by the pharmaceutical industry (16%) cited as the leading causes, the AOA said.
There was no difference in negative attitude between men and women, but age, region, and parental status each had a notable effect. Doubts of vaccine safety were highest in those aged 18-34 years (55%) and lowest in those aged 65 and older (29%). Those living in the West had the highest rate at 50%, while residents of the Midwest were lowest at 39%, and the negative attitude rate was 55% for adults who had children under age 18 years and 40% for those who did not, the AOA reported.
Respondents to the survey, conducted May 28-30, 2019, also were asked to choose one of five statements that best expressed their view of vaccines, and those data paint a somewhat different picture:
- 2% said vaccines are unsafe and ineffective.
- 6% said that the side-effect risks outweigh the benefits.
- 9% said they were not sure if vaccines are safe and effective.
- 31% said that the benefits outweigh the risks.
- 51% said that vaccines are safe and effective.
Social media were another important source of doubt among respondents, but they have not been effective at countering the spread of vaccine misinformation, said psychiatrist Rachel Shmuts, DO, of Cherry Hill, N.J.
Confirmation bias makes it difficult to convince someone vaccines are safe, effective, and necessary once they believe they are not. “The number of people who believe vaccines are dangerous and refuse to get them is still relatively small. However, online support groups seem to solidify their beliefs, making them less susceptible to influence from their neighbors and real-world communities,” she said in the AOA statement.
Osteopathic family physician Paul Ehrmann, DO, said in the statement, “People know that a lot of practices won’t accept patients who don’t vaccinate, so when they find one that will, they spread the word to their community that it’s a safe place. Whether intentional or not, those doctors are often seen as endorsing anti-vaxxer beliefs.”
In 2017, his home state of Michigan, with other partners, put on a public information campaign. It has “significantly improved vaccination rates across demographics,” according to the statement.
“Beliefs are hard to change especially when they’re based in fear,” Dr. Ehrmann, of Royal Oak, Mich., said in the statement. “But, being responsible for our patients’ health and the public’s health, we can’t afford to give in to those fears. We must insist on evidence-based medicine.”
according to the American Osteopathic Association.

In a survey conducted by the Harris Poll on behalf of the AOA, 45% of the 2,007 respondents expressed a negative attitude towards vaccine safety, with online articles (16%) and past secrets/wrongdoings by the pharmaceutical industry (16%) cited as the leading causes, the AOA said.
There was no difference in negative attitude between men and women, but age, region, and parental status each had a notable effect. Doubts of vaccine safety were highest in those aged 18-34 years (55%) and lowest in those aged 65 and older (29%). Those living in the West had the highest rate at 50%, while residents of the Midwest were lowest at 39%, and the negative attitude rate was 55% for adults who had children under age 18 years and 40% for those who did not, the AOA reported.
Respondents to the survey, conducted May 28-30, 2019, also were asked to choose one of five statements that best expressed their view of vaccines, and those data paint a somewhat different picture:
- 2% said vaccines are unsafe and ineffective.
- 6% said that the side-effect risks outweigh the benefits.
- 9% said they were not sure if vaccines are safe and effective.
- 31% said that the benefits outweigh the risks.
- 51% said that vaccines are safe and effective.
Social media were another important source of doubt among respondents, but they have not been effective at countering the spread of vaccine misinformation, said psychiatrist Rachel Shmuts, DO, of Cherry Hill, N.J.
Confirmation bias makes it difficult to convince someone vaccines are safe, effective, and necessary once they believe they are not. “The number of people who believe vaccines are dangerous and refuse to get them is still relatively small. However, online support groups seem to solidify their beliefs, making them less susceptible to influence from their neighbors and real-world communities,” she said in the AOA statement.
Osteopathic family physician Paul Ehrmann, DO, said in the statement, “People know that a lot of practices won’t accept patients who don’t vaccinate, so when they find one that will, they spread the word to their community that it’s a safe place. Whether intentional or not, those doctors are often seen as endorsing anti-vaxxer beliefs.”
In 2017, his home state of Michigan, with other partners, put on a public information campaign. It has “significantly improved vaccination rates across demographics,” according to the statement.
“Beliefs are hard to change especially when they’re based in fear,” Dr. Ehrmann, of Royal Oak, Mich., said in the statement. “But, being responsible for our patients’ health and the public’s health, we can’t afford to give in to those fears. We must insist on evidence-based medicine.”
according to the American Osteopathic Association.

In a survey conducted by the Harris Poll on behalf of the AOA, 45% of the 2,007 respondents expressed a negative attitude towards vaccine safety, with online articles (16%) and past secrets/wrongdoings by the pharmaceutical industry (16%) cited as the leading causes, the AOA said.
There was no difference in negative attitude between men and women, but age, region, and parental status each had a notable effect. Doubts of vaccine safety were highest in those aged 18-34 years (55%) and lowest in those aged 65 and older (29%). Those living in the West had the highest rate at 50%, while residents of the Midwest were lowest at 39%, and the negative attitude rate was 55% for adults who had children under age 18 years and 40% for those who did not, the AOA reported.
Respondents to the survey, conducted May 28-30, 2019, also were asked to choose one of five statements that best expressed their view of vaccines, and those data paint a somewhat different picture:
- 2% said vaccines are unsafe and ineffective.
- 6% said that the side-effect risks outweigh the benefits.
- 9% said they were not sure if vaccines are safe and effective.
- 31% said that the benefits outweigh the risks.
- 51% said that vaccines are safe and effective.
Social media were another important source of doubt among respondents, but they have not been effective at countering the spread of vaccine misinformation, said psychiatrist Rachel Shmuts, DO, of Cherry Hill, N.J.
Confirmation bias makes it difficult to convince someone vaccines are safe, effective, and necessary once they believe they are not. “The number of people who believe vaccines are dangerous and refuse to get them is still relatively small. However, online support groups seem to solidify their beliefs, making them less susceptible to influence from their neighbors and real-world communities,” she said in the AOA statement.
Osteopathic family physician Paul Ehrmann, DO, said in the statement, “People know that a lot of practices won’t accept patients who don’t vaccinate, so when they find one that will, they spread the word to their community that it’s a safe place. Whether intentional or not, those doctors are often seen as endorsing anti-vaxxer beliefs.”
In 2017, his home state of Michigan, with other partners, put on a public information campaign. It has “significantly improved vaccination rates across demographics,” according to the statement.
“Beliefs are hard to change especially when they’re based in fear,” Dr. Ehrmann, of Royal Oak, Mich., said in the statement. “But, being responsible for our patients’ health and the public’s health, we can’t afford to give in to those fears. We must insist on evidence-based medicine.”