User login
Official Newspaper of the American College of Surgeons
High TAVR rates linked to fewer readmissions
WASHINGTON – Hospitals with a higher volume of transcatheter aortic valve replacements (TAVRs) have significantly lower 30-day readmission rates, according to an observational study.
In a study of 129 hospitals, those that performed more than 100 TAVR procedures had a 24% and 25% lower readmission rate compared with hospitals that performed 50 to100 TAVRs (P less than .001) and hospitals that performed fewer that 50 TAVRs (P = .007) respectively (JAMA Cardiol. 2017 May 11. doi: 10.1001/jamacardio.2017.1630).
This finding could have serious financial and medical implications for hospitals that are deciding whether or not to focus on this minimally invasive procedure, according to Sahil Khera, MD, MPH, chief resident and cardiology fellow at New York Medical College, Valhalla, and his colleagues.
“Lower readmission rates at high-volume hospitals substantially reduce health care expenditure,” said Dr. Khera and colleagues. “As new TAVR programs open across the country, these data will guide policymakers to identify targets for optimizing and standardizing TAVR outcomes across hospitals.”
To study the correlation between TAVR procedures and readmission rates, the investigators gathered records on hospitals that performed at least five TAVRs in 2014, which were then categorized into high-, medium-, or low-volume categories, and cross-referenced with the 2014 Nationwide Readmissions Database.
Of the 16,252 TAVR procedures conducted in 2014, 663 (4%), 3,067 (19%), and 12,522 (77%) were performed at low-, medium-, and high-volume hospitals, respectively, according to the investigators.
Patients undergoing these procedures were on average 81 years of age, with an average of four Elixhauser comorbidities, most commonly dyslipidemia (64%), hypertension (80%), heart failure (75%), and known coronary artery disease (69%), with a majority having undergone an endovascular procedure (83%).
However, the researchers found the population of TAVR patients of high volume hospitals were slightly younger, had fewer women, were more likely to be in a higher income household, and were less likely to undergo a transapical procedure than in low volume hospitals, which Dr. Khera and fellow researchers believe may have some impact on their findings.
“Low-volume hospitals were more likely to operate on patients with a higher number of comorbidities compared with high-volume hospitals and were more likely to use the TA approach,” according to investigators, “Transapical TAVR is associated with poorer short- and intermediate-term mortality, increased use of skilled nursing care facilities, longer hospital stays, and readmissions when compared with transfemoral TAVR.”
Overall, there were 2,667 readmissions reported, among which high volume hospitals reported a 30-day readmission rate of 15.6%, while low- and medium-volume hospitals reported similarly higher rates of 19.5% and 19%.
When looking into the causes for these readmissions, the investigators found that 1,619 (61%) were due to noncardiac causes, which appeared in all three hospitals, despite a larger proportion present in low-volume hospitals as opposed to medium and high-volume hospitals (65.6% vs. 60.1% and 60.6%, respectively).
“Infection, respiratory, endocrine/metabolic, renal, and trauma problems were more common in low-volume hospitals,” according to the researchers. “Whereas gastrointestinal and TIA/stroke issues were more common in medium and high volume hospitals.”
While price and length of stay did not differ among the volume categories, the investigators estimate the lower rate of readmissions saved high-volume hospitals approximately $6.5 million.
They found that while the difference of readmissions between hospital classifications narrowed when controlling for experience, the margin was still significant. They admitted, however, that the possibility of greater access to more technologically advanced TAVR in high-volume hospitals may affect the findings.
This study was limited by administrative nature of the database used, which does not make available information such as valve type, patient risk scores, or medications.
One investigator has received personal fees from Edwards Lifesciences and Medtronic; another has received grants and personal fees from various pharmaceutical companies, educational institutions, and publications; and a third has consulted for Medtronic.
[email protected]
On Twitter @eaztweets
Considering the idea of using readmissions in comparison to rate of TAVR procedures is interesting, the number of confounds are too great to give any kind of accurate representation of medical practice. While the authors of this study do address its limitations, including a learning curve as it relates to the risk of inpatient mortality, the number of adjustments that must be made to account for the additional confounding factors are simply too insurmountable to give an accurate estimate of statistical and clinical importance.
For example, researchers found TAVR readmissions were associated with certain baseline comorbidities, access sites, and complications. However, association does not mean causation and so the categorization of cardiac-related vs. noncardiac-related readmissions must be approached with some caution.
If one were to try to use readmission rates after TAVR to argue for reimbursement of the procedure, one would need to determine a well established, validated reimbursement rate for TAVR readmissions, which has not been done.
Also, the advancing nature of this procedure, combined with a constant focus from hospitals to reduce readmission rates means any baseline for readmissions used would most likely be out of date.
It would be unlikely for investigators to factor in the cause of reduced readmission rates, which could be a factor of increased technology, more experienced physicians, lower-risk patients, or any combination thereof.
Holding TAVR sites accountable for quality of care is of course important, but using readmission rates to determine something like funding is not appropriate when the measurement being used is so complex.
Perhaps a better approach would be to widen access for low volume hospitals to resources that would improve the TAVR processes and encourage using financial incentives.
Evaluations by physical therapists or a similar procedure should be put into place before discharge to assess a patient’s risk of readmission.
Overall, this is a multifaceted issue that would be better helped by promoting TAVR best practices and encouraging hospitals to compare themselves against each other to reduce unnecessary readmissions.
John D. Carroll, MD, is professor of medicine and director of the Cardiac and Vascular Center at the University of Colorado, Denver. He made his remarks in an editorial in JAMA Cardiology ( doi: 10.1001/jamacardio.2017.1650 ).
Considering the idea of using readmissions in comparison to rate of TAVR procedures is interesting, the number of confounds are too great to give any kind of accurate representation of medical practice. While the authors of this study do address its limitations, including a learning curve as it relates to the risk of inpatient mortality, the number of adjustments that must be made to account for the additional confounding factors are simply too insurmountable to give an accurate estimate of statistical and clinical importance.
For example, researchers found TAVR readmissions were associated with certain baseline comorbidities, access sites, and complications. However, association does not mean causation and so the categorization of cardiac-related vs. noncardiac-related readmissions must be approached with some caution.
If one were to try to use readmission rates after TAVR to argue for reimbursement of the procedure, one would need to determine a well established, validated reimbursement rate for TAVR readmissions, which has not been done.
Also, the advancing nature of this procedure, combined with a constant focus from hospitals to reduce readmission rates means any baseline for readmissions used would most likely be out of date.
It would be unlikely for investigators to factor in the cause of reduced readmission rates, which could be a factor of increased technology, more experienced physicians, lower-risk patients, or any combination thereof.
Holding TAVR sites accountable for quality of care is of course important, but using readmission rates to determine something like funding is not appropriate when the measurement being used is so complex.
Perhaps a better approach would be to widen access for low volume hospitals to resources that would improve the TAVR processes and encourage using financial incentives.
Evaluations by physical therapists or a similar procedure should be put into place before discharge to assess a patient’s risk of readmission.
Overall, this is a multifaceted issue that would be better helped by promoting TAVR best practices and encouraging hospitals to compare themselves against each other to reduce unnecessary readmissions.
John D. Carroll, MD, is professor of medicine and director of the Cardiac and Vascular Center at the University of Colorado, Denver. He made his remarks in an editorial in JAMA Cardiology ( doi: 10.1001/jamacardio.2017.1650 ).
Considering the idea of using readmissions in comparison to rate of TAVR procedures is interesting, the number of confounds are too great to give any kind of accurate representation of medical practice. While the authors of this study do address its limitations, including a learning curve as it relates to the risk of inpatient mortality, the number of adjustments that must be made to account for the additional confounding factors are simply too insurmountable to give an accurate estimate of statistical and clinical importance.
For example, researchers found TAVR readmissions were associated with certain baseline comorbidities, access sites, and complications. However, association does not mean causation and so the categorization of cardiac-related vs. noncardiac-related readmissions must be approached with some caution.
If one were to try to use readmission rates after TAVR to argue for reimbursement of the procedure, one would need to determine a well established, validated reimbursement rate for TAVR readmissions, which has not been done.
Also, the advancing nature of this procedure, combined with a constant focus from hospitals to reduce readmission rates means any baseline for readmissions used would most likely be out of date.
It would be unlikely for investigators to factor in the cause of reduced readmission rates, which could be a factor of increased technology, more experienced physicians, lower-risk patients, or any combination thereof.
Holding TAVR sites accountable for quality of care is of course important, but using readmission rates to determine something like funding is not appropriate when the measurement being used is so complex.
Perhaps a better approach would be to widen access for low volume hospitals to resources that would improve the TAVR processes and encourage using financial incentives.
Evaluations by physical therapists or a similar procedure should be put into place before discharge to assess a patient’s risk of readmission.
Overall, this is a multifaceted issue that would be better helped by promoting TAVR best practices and encouraging hospitals to compare themselves against each other to reduce unnecessary readmissions.
John D. Carroll, MD, is professor of medicine and director of the Cardiac and Vascular Center at the University of Colorado, Denver. He made his remarks in an editorial in JAMA Cardiology ( doi: 10.1001/jamacardio.2017.1650 ).
WASHINGTON – Hospitals with a higher volume of transcatheter aortic valve replacements (TAVRs) have significantly lower 30-day readmission rates, according to an observational study.
In a study of 129 hospitals, those that performed more than 100 TAVR procedures had a 24% and 25% lower readmission rate compared with hospitals that performed 50 to100 TAVRs (P less than .001) and hospitals that performed fewer that 50 TAVRs (P = .007) respectively (JAMA Cardiol. 2017 May 11. doi: 10.1001/jamacardio.2017.1630).
This finding could have serious financial and medical implications for hospitals that are deciding whether or not to focus on this minimally invasive procedure, according to Sahil Khera, MD, MPH, chief resident and cardiology fellow at New York Medical College, Valhalla, and his colleagues.
“Lower readmission rates at high-volume hospitals substantially reduce health care expenditure,” said Dr. Khera and colleagues. “As new TAVR programs open across the country, these data will guide policymakers to identify targets for optimizing and standardizing TAVR outcomes across hospitals.”
To study the correlation between TAVR procedures and readmission rates, the investigators gathered records on hospitals that performed at least five TAVRs in 2014, which were then categorized into high-, medium-, or low-volume categories, and cross-referenced with the 2014 Nationwide Readmissions Database.
Of the 16,252 TAVR procedures conducted in 2014, 663 (4%), 3,067 (19%), and 12,522 (77%) were performed at low-, medium-, and high-volume hospitals, respectively, according to the investigators.
Patients undergoing these procedures were on average 81 years of age, with an average of four Elixhauser comorbidities, most commonly dyslipidemia (64%), hypertension (80%), heart failure (75%), and known coronary artery disease (69%), with a majority having undergone an endovascular procedure (83%).
However, the researchers found the population of TAVR patients of high volume hospitals were slightly younger, had fewer women, were more likely to be in a higher income household, and were less likely to undergo a transapical procedure than in low volume hospitals, which Dr. Khera and fellow researchers believe may have some impact on their findings.
“Low-volume hospitals were more likely to operate on patients with a higher number of comorbidities compared with high-volume hospitals and were more likely to use the TA approach,” according to investigators, “Transapical TAVR is associated with poorer short- and intermediate-term mortality, increased use of skilled nursing care facilities, longer hospital stays, and readmissions when compared with transfemoral TAVR.”
Overall, there were 2,667 readmissions reported, among which high volume hospitals reported a 30-day readmission rate of 15.6%, while low- and medium-volume hospitals reported similarly higher rates of 19.5% and 19%.
When looking into the causes for these readmissions, the investigators found that 1,619 (61%) were due to noncardiac causes, which appeared in all three hospitals, despite a larger proportion present in low-volume hospitals as opposed to medium and high-volume hospitals (65.6% vs. 60.1% and 60.6%, respectively).
“Infection, respiratory, endocrine/metabolic, renal, and trauma problems were more common in low-volume hospitals,” according to the researchers. “Whereas gastrointestinal and TIA/stroke issues were more common in medium and high volume hospitals.”
While price and length of stay did not differ among the volume categories, the investigators estimate the lower rate of readmissions saved high-volume hospitals approximately $6.5 million.
They found that while the difference of readmissions between hospital classifications narrowed when controlling for experience, the margin was still significant. They admitted, however, that the possibility of greater access to more technologically advanced TAVR in high-volume hospitals may affect the findings.
This study was limited by administrative nature of the database used, which does not make available information such as valve type, patient risk scores, or medications.
One investigator has received personal fees from Edwards Lifesciences and Medtronic; another has received grants and personal fees from various pharmaceutical companies, educational institutions, and publications; and a third has consulted for Medtronic.
[email protected]
On Twitter @eaztweets
WASHINGTON – Hospitals with a higher volume of transcatheter aortic valve replacements (TAVRs) have significantly lower 30-day readmission rates, according to an observational study.
In a study of 129 hospitals, those that performed more than 100 TAVR procedures had a 24% and 25% lower readmission rate compared with hospitals that performed 50 to100 TAVRs (P less than .001) and hospitals that performed fewer that 50 TAVRs (P = .007) respectively (JAMA Cardiol. 2017 May 11. doi: 10.1001/jamacardio.2017.1630).
This finding could have serious financial and medical implications for hospitals that are deciding whether or not to focus on this minimally invasive procedure, according to Sahil Khera, MD, MPH, chief resident and cardiology fellow at New York Medical College, Valhalla, and his colleagues.
“Lower readmission rates at high-volume hospitals substantially reduce health care expenditure,” said Dr. Khera and colleagues. “As new TAVR programs open across the country, these data will guide policymakers to identify targets for optimizing and standardizing TAVR outcomes across hospitals.”
To study the correlation between TAVR procedures and readmission rates, the investigators gathered records on hospitals that performed at least five TAVRs in 2014, which were then categorized into high-, medium-, or low-volume categories, and cross-referenced with the 2014 Nationwide Readmissions Database.
Of the 16,252 TAVR procedures conducted in 2014, 663 (4%), 3,067 (19%), and 12,522 (77%) were performed at low-, medium-, and high-volume hospitals, respectively, according to the investigators.
Patients undergoing these procedures were on average 81 years of age, with an average of four Elixhauser comorbidities, most commonly dyslipidemia (64%), hypertension (80%), heart failure (75%), and known coronary artery disease (69%), with a majority having undergone an endovascular procedure (83%).
However, the researchers found the population of TAVR patients of high volume hospitals were slightly younger, had fewer women, were more likely to be in a higher income household, and were less likely to undergo a transapical procedure than in low volume hospitals, which Dr. Khera and fellow researchers believe may have some impact on their findings.
“Low-volume hospitals were more likely to operate on patients with a higher number of comorbidities compared with high-volume hospitals and were more likely to use the TA approach,” according to investigators, “Transapical TAVR is associated with poorer short- and intermediate-term mortality, increased use of skilled nursing care facilities, longer hospital stays, and readmissions when compared with transfemoral TAVR.”
Overall, there were 2,667 readmissions reported, among which high volume hospitals reported a 30-day readmission rate of 15.6%, while low- and medium-volume hospitals reported similarly higher rates of 19.5% and 19%.
When looking into the causes for these readmissions, the investigators found that 1,619 (61%) were due to noncardiac causes, which appeared in all three hospitals, despite a larger proportion present in low-volume hospitals as opposed to medium and high-volume hospitals (65.6% vs. 60.1% and 60.6%, respectively).
“Infection, respiratory, endocrine/metabolic, renal, and trauma problems were more common in low-volume hospitals,” according to the researchers. “Whereas gastrointestinal and TIA/stroke issues were more common in medium and high volume hospitals.”
While price and length of stay did not differ among the volume categories, the investigators estimate the lower rate of readmissions saved high-volume hospitals approximately $6.5 million.
They found that while the difference of readmissions between hospital classifications narrowed when controlling for experience, the margin was still significant. They admitted, however, that the possibility of greater access to more technologically advanced TAVR in high-volume hospitals may affect the findings.
This study was limited by administrative nature of the database used, which does not make available information such as valve type, patient risk scores, or medications.
One investigator has received personal fees from Edwards Lifesciences and Medtronic; another has received grants and personal fees from various pharmaceutical companies, educational institutions, and publications; and a third has consulted for Medtronic.
[email protected]
On Twitter @eaztweets
FROM JAMA CARDIOLOGY
Key clinical point:
Major finding: Hospitals that performed more than 100 TAVR procedures reported significantly lower 30-day readmission rates, compared with those that performed 50-100 procedures and hospitals that performed less than 50.
Data source: Observational study of 129 hospitals that performed at least five TAVRs in 2015, and documents gathered from the 2014 Nationwide Readmissions Database.
Disclosures: One investigator has received personal fees from Edwards Lifesciences and Medtronic; another has received grants and personal fees from various pharmaceutical companies, educational institutions, and publications; and a third has consulted for Medtronic.
Follow five tips to mitigate opioid prescribing risks
CHICAGO – As the epidemic of opioid addiction and overdose deaths continues to surge, state and federal authorities are keeping a close eye on physicians who prescribe controlled substances.
Experts offer the following guidance on how well-meaning doctors can avoid coming under scrutiny for prescribing opioids and successfully manage investigations and audits.
1. Know who’s on the radar: The Drug Enforcement Agency (DEA) compiles a “black list” yearly of physicians and health care providers they plan to target for audits, said Natalia Mazina, a San Francisco–based attorney who specializes in health and pharmacy law. For 2017, the list includes physicians who have prior noncompliance records, providers who specialize in pain management, and those who dispense or administer large quantities of controlled substances.
In addition, family physicians, psychiatrists, and other specialists who come under investigation by a state medical board because of suspected inappropriate prescribing or reporting violations may also come under the purview of federal authorities, Ms. Mazina said.
“If they come on the medical board radar, they may come on the [DEA’s] radar as well,” she said during the interview. “They just have to watch how many prescriptions they write for controlled substances and make sure they are legitimate prescriptions.”
2. Maintain proper records: Poor record keeping is a top reason that the DEA investigates health care providers for potential prescribing violations, said Dennis A. Wichern, a DEA agent with the Chicago Field Division. Federal law requires that registered practitioners who store or dispense controlled substances keep records of controlled substances coming in and out of the practice. That includes physicians who hand out samples of controlled substances to patients and also pertains to samples provided to doctors by pharmaceutical companies.
Records should include whether the inventory was taken at the beginning or close of business, names of controlled substances, each finished form of the substances, the number of dosage units of each finished form in the commercial container, the number of commercial containers of each finished form, and disposition of the controlled substances.
Law requires that physicians take a new inventory of all controlled substances on hand every 2 years. Doctors are not required to keep records of controlled substances that are merely prescribed, unless such substances are prescribed in the course of maintenance or detoxification treatment.
Ms. Mazina notes that there are many software platforms that can assist practices with proper inventory and record keeping for opioids and other drugs.
3. Check the state database: Before prescribing opioids, check your state’s prescription drug monitoring program (PDMP) database, advises Ms. Mazina. At least 37 states have operational PDMPs that receive and distribute controlled substance prescription information to authorized users. About 11 states have enacted legislation to establish a PDMP, but some databases are not fully operational.
A state’s PDMP can reveal whether patients may be obtaining multiple controlled substance prescriptions from different doctors or doctor-shopping, Ms. Mazina said. Such due diligence helps inform treatment decisions and can assist a doctor’s case if a medical board or DEA investigation later arises.
“Even if your state law does not require you to check a patient’s history prior to prescribing, you have to check it to protect yourself,” she said. “If you want to avoid controlled substances problems, PDMP is the way to go.”
4. Establish an audit response plan: Have an audit response plan ready to roll should an inquiry arise, experts advise. The policies ensure that only approved information is released to authorities, and that all staff members are on the same page about how to react to audits, Ms. Mazina said.
Plans should clearly state what information can be collected and what data should be kept confidential. Financial information, for example, should be off limits, she said. Government agents are entitled to inventory, dispensary data, and records of receipts.
“Agents very often do the mirror image of the database, and they get too much information,” she said. “You don’t want to [allow] that.”
Train staff members how to respond to government authorities seeking audit information, and explain they have the right to refuse being interviewed, Ms. Mazina said.
“Train your employees on what’s going to happen if the DEA comes in,” she said. “If I don’t have clear policies and procedures, and I’m not trained, I might disclose everything and blame someone. That puts everyone in a [bad] position, because [authorities] will record everything and use it against [the practice].”
5. Confer with the experts: It doesn’t hurt to consult with other medical professionals, such as emergency physicians or pain management specialists, for practical advice on inventory policies or software suggestions. But when it comes to staying updated on new drug laws and regulations, confer with a health law attorney or compliance officer, Ms. Mazina said. The DEA website also includes useful information about recent laws and rules pertaining to prescription drugs, as does the Centers for Disease Control and Prevention website.
If an investigation or audit emerges, work with an attorney as early as possible. Often, practices wait until too late after an investigation begins to contact legal counsel, Ms. Mazina noted. The earlier an attorney gets involved, the sooner that person can build a strong case for the practice and work toward the best resolution.
“Very often, the physician thinks they are right, and there’s nothing for them to fear,” she said. “There is something for you to fear. There’s a lot at stake.”
[email protected]
On Twitter @legal_med
CHICAGO – As the epidemic of opioid addiction and overdose deaths continues to surge, state and federal authorities are keeping a close eye on physicians who prescribe controlled substances.
Experts offer the following guidance on how well-meaning doctors can avoid coming under scrutiny for prescribing opioids and successfully manage investigations and audits.
1. Know who’s on the radar: The Drug Enforcement Agency (DEA) compiles a “black list” yearly of physicians and health care providers they plan to target for audits, said Natalia Mazina, a San Francisco–based attorney who specializes in health and pharmacy law. For 2017, the list includes physicians who have prior noncompliance records, providers who specialize in pain management, and those who dispense or administer large quantities of controlled substances.
In addition, family physicians, psychiatrists, and other specialists who come under investigation by a state medical board because of suspected inappropriate prescribing or reporting violations may also come under the purview of federal authorities, Ms. Mazina said.
“If they come on the medical board radar, they may come on the [DEA’s] radar as well,” she said during the interview. “They just have to watch how many prescriptions they write for controlled substances and make sure they are legitimate prescriptions.”
2. Maintain proper records: Poor record keeping is a top reason that the DEA investigates health care providers for potential prescribing violations, said Dennis A. Wichern, a DEA agent with the Chicago Field Division. Federal law requires that registered practitioners who store or dispense controlled substances keep records of controlled substances coming in and out of the practice. That includes physicians who hand out samples of controlled substances to patients and also pertains to samples provided to doctors by pharmaceutical companies.
Records should include whether the inventory was taken at the beginning or close of business, names of controlled substances, each finished form of the substances, the number of dosage units of each finished form in the commercial container, the number of commercial containers of each finished form, and disposition of the controlled substances.
Law requires that physicians take a new inventory of all controlled substances on hand every 2 years. Doctors are not required to keep records of controlled substances that are merely prescribed, unless such substances are prescribed in the course of maintenance or detoxification treatment.
Ms. Mazina notes that there are many software platforms that can assist practices with proper inventory and record keeping for opioids and other drugs.
3. Check the state database: Before prescribing opioids, check your state’s prescription drug monitoring program (PDMP) database, advises Ms. Mazina. At least 37 states have operational PDMPs that receive and distribute controlled substance prescription information to authorized users. About 11 states have enacted legislation to establish a PDMP, but some databases are not fully operational.
A state’s PDMP can reveal whether patients may be obtaining multiple controlled substance prescriptions from different doctors or doctor-shopping, Ms. Mazina said. Such due diligence helps inform treatment decisions and can assist a doctor’s case if a medical board or DEA investigation later arises.
“Even if your state law does not require you to check a patient’s history prior to prescribing, you have to check it to protect yourself,” she said. “If you want to avoid controlled substances problems, PDMP is the way to go.”
4. Establish an audit response plan: Have an audit response plan ready to roll should an inquiry arise, experts advise. The policies ensure that only approved information is released to authorities, and that all staff members are on the same page about how to react to audits, Ms. Mazina said.
Plans should clearly state what information can be collected and what data should be kept confidential. Financial information, for example, should be off limits, she said. Government agents are entitled to inventory, dispensary data, and records of receipts.
“Agents very often do the mirror image of the database, and they get too much information,” she said. “You don’t want to [allow] that.”
Train staff members how to respond to government authorities seeking audit information, and explain they have the right to refuse being interviewed, Ms. Mazina said.
“Train your employees on what’s going to happen if the DEA comes in,” she said. “If I don’t have clear policies and procedures, and I’m not trained, I might disclose everything and blame someone. That puts everyone in a [bad] position, because [authorities] will record everything and use it against [the practice].”
5. Confer with the experts: It doesn’t hurt to consult with other medical professionals, such as emergency physicians or pain management specialists, for practical advice on inventory policies or software suggestions. But when it comes to staying updated on new drug laws and regulations, confer with a health law attorney or compliance officer, Ms. Mazina said. The DEA website also includes useful information about recent laws and rules pertaining to prescription drugs, as does the Centers for Disease Control and Prevention website.
If an investigation or audit emerges, work with an attorney as early as possible. Often, practices wait until too late after an investigation begins to contact legal counsel, Ms. Mazina noted. The earlier an attorney gets involved, the sooner that person can build a strong case for the practice and work toward the best resolution.
“Very often, the physician thinks they are right, and there’s nothing for them to fear,” she said. “There is something for you to fear. There’s a lot at stake.”
[email protected]
On Twitter @legal_med
CHICAGO – As the epidemic of opioid addiction and overdose deaths continues to surge, state and federal authorities are keeping a close eye on physicians who prescribe controlled substances.
Experts offer the following guidance on how well-meaning doctors can avoid coming under scrutiny for prescribing opioids and successfully manage investigations and audits.
1. Know who’s on the radar: The Drug Enforcement Agency (DEA) compiles a “black list” yearly of physicians and health care providers they plan to target for audits, said Natalia Mazina, a San Francisco–based attorney who specializes in health and pharmacy law. For 2017, the list includes physicians who have prior noncompliance records, providers who specialize in pain management, and those who dispense or administer large quantities of controlled substances.
In addition, family physicians, psychiatrists, and other specialists who come under investigation by a state medical board because of suspected inappropriate prescribing or reporting violations may also come under the purview of federal authorities, Ms. Mazina said.
“If they come on the medical board radar, they may come on the [DEA’s] radar as well,” she said during the interview. “They just have to watch how many prescriptions they write for controlled substances and make sure they are legitimate prescriptions.”
2. Maintain proper records: Poor record keeping is a top reason that the DEA investigates health care providers for potential prescribing violations, said Dennis A. Wichern, a DEA agent with the Chicago Field Division. Federal law requires that registered practitioners who store or dispense controlled substances keep records of controlled substances coming in and out of the practice. That includes physicians who hand out samples of controlled substances to patients and also pertains to samples provided to doctors by pharmaceutical companies.
Records should include whether the inventory was taken at the beginning or close of business, names of controlled substances, each finished form of the substances, the number of dosage units of each finished form in the commercial container, the number of commercial containers of each finished form, and disposition of the controlled substances.
Law requires that physicians take a new inventory of all controlled substances on hand every 2 years. Doctors are not required to keep records of controlled substances that are merely prescribed, unless such substances are prescribed in the course of maintenance or detoxification treatment.
Ms. Mazina notes that there are many software platforms that can assist practices with proper inventory and record keeping for opioids and other drugs.
3. Check the state database: Before prescribing opioids, check your state’s prescription drug monitoring program (PDMP) database, advises Ms. Mazina. At least 37 states have operational PDMPs that receive and distribute controlled substance prescription information to authorized users. About 11 states have enacted legislation to establish a PDMP, but some databases are not fully operational.
A state’s PDMP can reveal whether patients may be obtaining multiple controlled substance prescriptions from different doctors or doctor-shopping, Ms. Mazina said. Such due diligence helps inform treatment decisions and can assist a doctor’s case if a medical board or DEA investigation later arises.
“Even if your state law does not require you to check a patient’s history prior to prescribing, you have to check it to protect yourself,” she said. “If you want to avoid controlled substances problems, PDMP is the way to go.”
4. Establish an audit response plan: Have an audit response plan ready to roll should an inquiry arise, experts advise. The policies ensure that only approved information is released to authorities, and that all staff members are on the same page about how to react to audits, Ms. Mazina said.
Plans should clearly state what information can be collected and what data should be kept confidential. Financial information, for example, should be off limits, she said. Government agents are entitled to inventory, dispensary data, and records of receipts.
“Agents very often do the mirror image of the database, and they get too much information,” she said. “You don’t want to [allow] that.”
Train staff members how to respond to government authorities seeking audit information, and explain they have the right to refuse being interviewed, Ms. Mazina said.
“Train your employees on what’s going to happen if the DEA comes in,” she said. “If I don’t have clear policies and procedures, and I’m not trained, I might disclose everything and blame someone. That puts everyone in a [bad] position, because [authorities] will record everything and use it against [the practice].”
5. Confer with the experts: It doesn’t hurt to consult with other medical professionals, such as emergency physicians or pain management specialists, for practical advice on inventory policies or software suggestions. But when it comes to staying updated on new drug laws and regulations, confer with a health law attorney or compliance officer, Ms. Mazina said. The DEA website also includes useful information about recent laws and rules pertaining to prescription drugs, as does the Centers for Disease Control and Prevention website.
If an investigation or audit emerges, work with an attorney as early as possible. Often, practices wait until too late after an investigation begins to contact legal counsel, Ms. Mazina noted. The earlier an attorney gets involved, the sooner that person can build a strong case for the practice and work toward the best resolution.
“Very often, the physician thinks they are right, and there’s nothing for them to fear,” she said. “There is something for you to fear. There’s a lot at stake.”
[email protected]
On Twitter @legal_med
AT THE PHYSICIANS LEGAL ISSUES CONFERENCE
Senate health care proposal already facing uphill battle
Senate Republican leaders are facing pushback from almost every side on their Affordable Care Act repeal/replace proposal – so much so that the current plan is unlikely to gain enough support to pass.
“For a variety of reasons, we are not ready to vote for this bill, but we are open to negotiation and obtaining more information before it is brought to the floor,” Sen. Rand Paul (R-Ky.), Sen. Ted Cruz (R-Texas), Sen. Ron Johnson (R-Wis.), and Sen. Mike Lee (R-Utah) said in a joint statement issued June 22, the day the plan was published. “There are provisions in this draft that represent an improvement to our current health care system, but it does not appear this draft as written will accomplish the most important promise that we made to Americans to repeal Obamacare and lower their health care costs.”
Like the House-passed American Health Care Act (H.R. 1628), the proposed BCRA would reduce Medicaid spending and would address rising premiums in the individual health insurance marketplace; however, BCRA would take a slightly different path to the same destination.
Like the House bill, BCRA also targets funding for Planned Parenthood, although because of Senate procedural rules, it is a more indirect funding ban.
A key difference between BCRA and the House bill is how insurance premium support is calculated. The AHCA would base tax credits on age, providing a lesser benefit for older, but pre–Medicare-age adults. In contrast, BCRA would base tax credits on income while limiting eligibility to households at 350% of the federal poverty line. Further, credits would cover only 58% of the actuarial value of health insurance under BCRA.
The draft Senate plan would not allow states to request a waiver from the ACA’s so-called community waiver provisions – the portion of the law that requires health insurance premiums to be the same regardless of age or preexisting condition; the House-passed AHCA would allow those waivers.
Medicaid expansion would be rolled back under the Senate plan, but at a slower pace than the AHCA would require – by 2023 under BCRA vs. 2020 under AHCA.
The BCRA would establish a per capita funding mechanism for Medicaid going forward, which would base funding on historic Medicaid expenditures and uses an economic index to track inflation and adjust payments accordingly.
To address the needs of people with greater health care needs, the Senate proposal would provide $57 billion over the first 4 years, then another $57 billion over the next 8. The funds would be available for programs such as premium support or high-risk pools to help individuals who are expected to be high users of health care. States would be required to match funds starting in 2022.
Experts were quick to weigh in on the Senate plan.
The BCRA needs to do three things, according to Grace-Marie Turner, president of the Galen Institute: Provide a safety net for those covered through the ACA so that they do not lose coverage in the transition, modernize Medicaid, and give states more authority and options to reform their own health insurance markets.
“We have learned that the federal government is not able to regulate something as local as health insurance,” Ms. Turner said. “They cannot create policies and legislation that works for people in downtown Manhattan and rural Montana and southern New Mexico and the panhandle of Florida. There are too many different populations. The states need to do that, and this bill also would give the states more authority to begin to oversee their health insurance markets but with new funding to provide extra help for the people who have difficulty buying progress.”
She said it could be much better if the Senate did not use the reconciliation process, “but within the confines of that, both the House and Senate bills do the same thing.”
Ms. Turner also stressed that there are more reforms coming later, as the Senate and House address other portions of ACA repeal/replacement.
“I hope that [senators] would see moving this forward as beneficial so that then they can move additional pieces of legislation, hopefully, with 60 votes to go through the regular process, to have additional follow-up bills. This is not the end of the story. This is just rescuing us from Obamacare,” she said. “Then we need to go forward and think about what do we need to do to make our health sector work better in the future by putting doctors and patients, rather than government, in charge of choices.”
Doctors, however, did not agree.
“This bill significantly decreases patients’ ability to access high-quality health care, and affordable coverage for millions of Americans will be in jeopardy if the legislation is passed,” Boyd Buser, MD, a doctor of osteopathy and president of the American Osteopathic Association, said in a statement.
He noted that the Medicaid cuts will have a “devastating impact, especially in areas of our country hardest hit by the ongoing opioid epidemic. ... The Senate bill should have prioritized prevention and care coordination, two measures proven to reduce overall health costs by eliminating waste and addressing health problems at the most treatable stage. Decreasing the number of Americans with coverage as it intends does will not lower costs.”
A vote on the proposal could come as early as June 29 before the Senate breaks for the 4th of July recess.
Republican lawmakers and the Trump administration have vowed to address the ACA in other ways as well, by reviewing and possibly changing all relevant regulations, then using the regular legislative process, which would need 60 votes, to address issues that cannot be handled by the budget reconciliation process.
Senate Republican leaders are facing pushback from almost every side on their Affordable Care Act repeal/replace proposal – so much so that the current plan is unlikely to gain enough support to pass.
“For a variety of reasons, we are not ready to vote for this bill, but we are open to negotiation and obtaining more information before it is brought to the floor,” Sen. Rand Paul (R-Ky.), Sen. Ted Cruz (R-Texas), Sen. Ron Johnson (R-Wis.), and Sen. Mike Lee (R-Utah) said in a joint statement issued June 22, the day the plan was published. “There are provisions in this draft that represent an improvement to our current health care system, but it does not appear this draft as written will accomplish the most important promise that we made to Americans to repeal Obamacare and lower their health care costs.”
Like the House-passed American Health Care Act (H.R. 1628), the proposed BCRA would reduce Medicaid spending and would address rising premiums in the individual health insurance marketplace; however, BCRA would take a slightly different path to the same destination.
Like the House bill, BCRA also targets funding for Planned Parenthood, although because of Senate procedural rules, it is a more indirect funding ban.
A key difference between BCRA and the House bill is how insurance premium support is calculated. The AHCA would base tax credits on age, providing a lesser benefit for older, but pre–Medicare-age adults. In contrast, BCRA would base tax credits on income while limiting eligibility to households at 350% of the federal poverty line. Further, credits would cover only 58% of the actuarial value of health insurance under BCRA.
The draft Senate plan would not allow states to request a waiver from the ACA’s so-called community waiver provisions – the portion of the law that requires health insurance premiums to be the same regardless of age or preexisting condition; the House-passed AHCA would allow those waivers.
Medicaid expansion would be rolled back under the Senate plan, but at a slower pace than the AHCA would require – by 2023 under BCRA vs. 2020 under AHCA.
The BCRA would establish a per capita funding mechanism for Medicaid going forward, which would base funding on historic Medicaid expenditures and uses an economic index to track inflation and adjust payments accordingly.
To address the needs of people with greater health care needs, the Senate proposal would provide $57 billion over the first 4 years, then another $57 billion over the next 8. The funds would be available for programs such as premium support or high-risk pools to help individuals who are expected to be high users of health care. States would be required to match funds starting in 2022.
Experts were quick to weigh in on the Senate plan.
The BCRA needs to do three things, according to Grace-Marie Turner, president of the Galen Institute: Provide a safety net for those covered through the ACA so that they do not lose coverage in the transition, modernize Medicaid, and give states more authority and options to reform their own health insurance markets.
“We have learned that the federal government is not able to regulate something as local as health insurance,” Ms. Turner said. “They cannot create policies and legislation that works for people in downtown Manhattan and rural Montana and southern New Mexico and the panhandle of Florida. There are too many different populations. The states need to do that, and this bill also would give the states more authority to begin to oversee their health insurance markets but with new funding to provide extra help for the people who have difficulty buying progress.”
She said it could be much better if the Senate did not use the reconciliation process, “but within the confines of that, both the House and Senate bills do the same thing.”
Ms. Turner also stressed that there are more reforms coming later, as the Senate and House address other portions of ACA repeal/replacement.
“I hope that [senators] would see moving this forward as beneficial so that then they can move additional pieces of legislation, hopefully, with 60 votes to go through the regular process, to have additional follow-up bills. This is not the end of the story. This is just rescuing us from Obamacare,” she said. “Then we need to go forward and think about what do we need to do to make our health sector work better in the future by putting doctors and patients, rather than government, in charge of choices.”
Doctors, however, did not agree.
“This bill significantly decreases patients’ ability to access high-quality health care, and affordable coverage for millions of Americans will be in jeopardy if the legislation is passed,” Boyd Buser, MD, a doctor of osteopathy and president of the American Osteopathic Association, said in a statement.
He noted that the Medicaid cuts will have a “devastating impact, especially in areas of our country hardest hit by the ongoing opioid epidemic. ... The Senate bill should have prioritized prevention and care coordination, two measures proven to reduce overall health costs by eliminating waste and addressing health problems at the most treatable stage. Decreasing the number of Americans with coverage as it intends does will not lower costs.”
A vote on the proposal could come as early as June 29 before the Senate breaks for the 4th of July recess.
Republican lawmakers and the Trump administration have vowed to address the ACA in other ways as well, by reviewing and possibly changing all relevant regulations, then using the regular legislative process, which would need 60 votes, to address issues that cannot be handled by the budget reconciliation process.
Senate Republican leaders are facing pushback from almost every side on their Affordable Care Act repeal/replace proposal – so much so that the current plan is unlikely to gain enough support to pass.
“For a variety of reasons, we are not ready to vote for this bill, but we are open to negotiation and obtaining more information before it is brought to the floor,” Sen. Rand Paul (R-Ky.), Sen. Ted Cruz (R-Texas), Sen. Ron Johnson (R-Wis.), and Sen. Mike Lee (R-Utah) said in a joint statement issued June 22, the day the plan was published. “There are provisions in this draft that represent an improvement to our current health care system, but it does not appear this draft as written will accomplish the most important promise that we made to Americans to repeal Obamacare and lower their health care costs.”
Like the House-passed American Health Care Act (H.R. 1628), the proposed BCRA would reduce Medicaid spending and would address rising premiums in the individual health insurance marketplace; however, BCRA would take a slightly different path to the same destination.
Like the House bill, BCRA also targets funding for Planned Parenthood, although because of Senate procedural rules, it is a more indirect funding ban.
A key difference between BCRA and the House bill is how insurance premium support is calculated. The AHCA would base tax credits on age, providing a lesser benefit for older, but pre–Medicare-age adults. In contrast, BCRA would base tax credits on income while limiting eligibility to households at 350% of the federal poverty line. Further, credits would cover only 58% of the actuarial value of health insurance under BCRA.
The draft Senate plan would not allow states to request a waiver from the ACA’s so-called community waiver provisions – the portion of the law that requires health insurance premiums to be the same regardless of age or preexisting condition; the House-passed AHCA would allow those waivers.
Medicaid expansion would be rolled back under the Senate plan, but at a slower pace than the AHCA would require – by 2023 under BCRA vs. 2020 under AHCA.
The BCRA would establish a per capita funding mechanism for Medicaid going forward, which would base funding on historic Medicaid expenditures and uses an economic index to track inflation and adjust payments accordingly.
To address the needs of people with greater health care needs, the Senate proposal would provide $57 billion over the first 4 years, then another $57 billion over the next 8. The funds would be available for programs such as premium support or high-risk pools to help individuals who are expected to be high users of health care. States would be required to match funds starting in 2022.
Experts were quick to weigh in on the Senate plan.
The BCRA needs to do three things, according to Grace-Marie Turner, president of the Galen Institute: Provide a safety net for those covered through the ACA so that they do not lose coverage in the transition, modernize Medicaid, and give states more authority and options to reform their own health insurance markets.
“We have learned that the federal government is not able to regulate something as local as health insurance,” Ms. Turner said. “They cannot create policies and legislation that works for people in downtown Manhattan and rural Montana and southern New Mexico and the panhandle of Florida. There are too many different populations. The states need to do that, and this bill also would give the states more authority to begin to oversee their health insurance markets but with new funding to provide extra help for the people who have difficulty buying progress.”
She said it could be much better if the Senate did not use the reconciliation process, “but within the confines of that, both the House and Senate bills do the same thing.”
Ms. Turner also stressed that there are more reforms coming later, as the Senate and House address other portions of ACA repeal/replacement.
“I hope that [senators] would see moving this forward as beneficial so that then they can move additional pieces of legislation, hopefully, with 60 votes to go through the regular process, to have additional follow-up bills. This is not the end of the story. This is just rescuing us from Obamacare,” she said. “Then we need to go forward and think about what do we need to do to make our health sector work better in the future by putting doctors and patients, rather than government, in charge of choices.”
Doctors, however, did not agree.
“This bill significantly decreases patients’ ability to access high-quality health care, and affordable coverage for millions of Americans will be in jeopardy if the legislation is passed,” Boyd Buser, MD, a doctor of osteopathy and president of the American Osteopathic Association, said in a statement.
He noted that the Medicaid cuts will have a “devastating impact, especially in areas of our country hardest hit by the ongoing opioid epidemic. ... The Senate bill should have prioritized prevention and care coordination, two measures proven to reduce overall health costs by eliminating waste and addressing health problems at the most treatable stage. Decreasing the number of Americans with coverage as it intends does will not lower costs.”
A vote on the proposal could come as early as June 29 before the Senate breaks for the 4th of July recess.
Republican lawmakers and the Trump administration have vowed to address the ACA in other ways as well, by reviewing and possibly changing all relevant regulations, then using the regular legislative process, which would need 60 votes, to address issues that cannot be handled by the budget reconciliation process.
For opioid-related hospitalizations, men and women are equal
Equality is not always a good thing, particularly with opioids.
In 2005, the rate of opioid-related inpatient hospital stays was 145.6 per 100,000 population for males of all ages and 127.8 for females of all ages. By 2014, however, equality had arrived: Females had a rate of 224.1 per 100,000, compared with 225 for males, according to the Agency for Healthcare Research and Quality. Those increases in hospital admissions work out to 75% for females and 55% for males.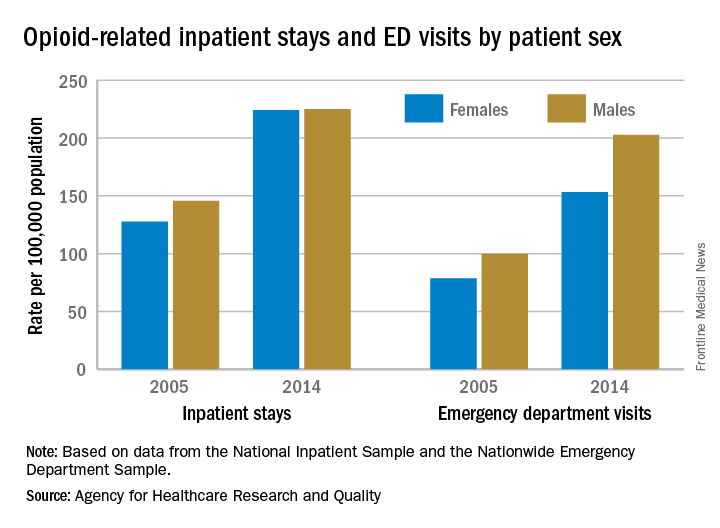
The states tell a similarly unequal story for opioid-related ED visits. In 2014, males had the higher rate in 23 states, and females had the higher rate in 7 states. (Washington, D.C., and 20 states do not participate in the State Emergency Department Databases and were not included in this analysis.)
Among the 30 participating states, Massachusetts had the highest visit rates for both males (598.8) and females (310.4), and Iowa had the lowest at 37 for males and 53.1 for females, AHRQ said.
The roles were reversed for opioid-related hospital admissions in the states in 2014: Females had the higher rate in 33 of the states participating in the State Inpatient Databases, compared with 11 states and the District of Columbia for males.
West Virginia had the highest rate for females at 371.2, and Washington, D.C., had the highest rate for males at 472. The lowest rates for both females (82.3) and males (63) were found in Iowa, according to the report.
Equality is not always a good thing, particularly with opioids.
In 2005, the rate of opioid-related inpatient hospital stays was 145.6 per 100,000 population for males of all ages and 127.8 for females of all ages. By 2014, however, equality had arrived: Females had a rate of 224.1 per 100,000, compared with 225 for males, according to the Agency for Healthcare Research and Quality. Those increases in hospital admissions work out to 75% for females and 55% for males.
The states tell a similarly unequal story for opioid-related ED visits. In 2014, males had the higher rate in 23 states, and females had the higher rate in 7 states. (Washington, D.C., and 20 states do not participate in the State Emergency Department Databases and were not included in this analysis.)
Among the 30 participating states, Massachusetts had the highest visit rates for both males (598.8) and females (310.4), and Iowa had the lowest at 37 for males and 53.1 for females, AHRQ said.
The roles were reversed for opioid-related hospital admissions in the states in 2014: Females had the higher rate in 33 of the states participating in the State Inpatient Databases, compared with 11 states and the District of Columbia for males.
West Virginia had the highest rate for females at 371.2, and Washington, D.C., had the highest rate for males at 472. The lowest rates for both females (82.3) and males (63) were found in Iowa, according to the report.
Equality is not always a good thing, particularly with opioids.
In 2005, the rate of opioid-related inpatient hospital stays was 145.6 per 100,000 population for males of all ages and 127.8 for females of all ages. By 2014, however, equality had arrived: Females had a rate of 224.1 per 100,000, compared with 225 for males, according to the Agency for Healthcare Research and Quality. Those increases in hospital admissions work out to 75% for females and 55% for males.
The states tell a similarly unequal story for opioid-related ED visits. In 2014, males had the higher rate in 23 states, and females had the higher rate in 7 states. (Washington, D.C., and 20 states do not participate in the State Emergency Department Databases and were not included in this analysis.)
Among the 30 participating states, Massachusetts had the highest visit rates for both males (598.8) and females (310.4), and Iowa had the lowest at 37 for males and 53.1 for females, AHRQ said.
The roles were reversed for opioid-related hospital admissions in the states in 2014: Females had the higher rate in 33 of the states participating in the State Inpatient Databases, compared with 11 states and the District of Columbia for males.
West Virginia had the highest rate for females at 371.2, and Washington, D.C., had the highest rate for males at 472. The lowest rates for both females (82.3) and males (63) were found in Iowa, according to the report.
Proper catheter removal promotes colorectal surgery recovery
Adherence to guidelines for urinary catheter removal significantly reduced rates of urinary tract infection and length of hospital stay, based on data from almost 3,000 patients.
UTIs occurred in 0.8% of colonic surgery patients who were guideline compliant, compared with 4.1% of noncompliant patients. Similarly, UTIs were significantly less likely in rectal surgery patients who complied with guidelines, compared with those who did not (3.5% vs. 9.6%).
The Implementation of the Enhanced Recovery After Surgery (iERAS) program involved 15 academic hospitals in Ontario, Canada. The guidelines include prompt removal of catheters from colonic and rectal surgery patients. Recommended removal times ranged from 24 hours to 72 hours, depending on the procedure.
“Although numerous reports have documented the overall effectiveness of bundled ERAS interventions improving outcome, the contribution of individual components of the pathway in the context of an ERAS implementation program is uncertain,” wrote Allan Okrainec, MD, of the University of Toronto, and his colleagues.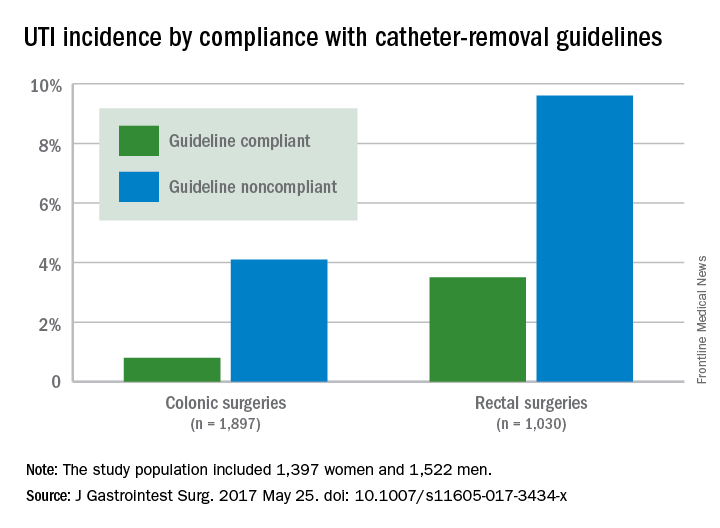
Overall, 53% of patients had catheters removed in compliance with the guidelines, including 47.2% of those with colonic resections and 69.5% of those with rectal resections.
The average length of hospital stay for colonic surgery patients who complied with the urinary catheter guideline was 4 days, compared with 5 days for noncompliant patients, a statistically significant difference. Similarly, the average length of stay was significantly shorter for compliant rectal surgery patients, compared with noncompliant patients (5 days vs. 8 days, respectively).
Reinsertion of the urinary catheter was needed for 6% of the patients, including 122 patients with suspected or confirmed urinary retention and 36 patients who had other indications for reinsertion.
“One of the concerns about early removal of catheters is that it might lead to an increased need for reinsertion,” the researchers noted. “In our study, guideline compliance was associated with an increased rate of catheter reinsertion, but [it] only increased the risk by 2%-3% in patients having rectal or colonic procedures,” the investigators said.
The study results were limited by a lack of information about reasons for noncompliance with the guidelines, the researchers added. However, the data support the value of early catheter removal for surgical patients. “It is noteworthy that, in our study, as well as others, urinary retention rates are relatively low,” they said.
The Council of Academic Hospitals of Ontario funded the study. The researchers had no financial conflicts to disclose.
Adherence to guidelines for urinary catheter removal significantly reduced rates of urinary tract infection and length of hospital stay, based on data from almost 3,000 patients.
UTIs occurred in 0.8% of colonic surgery patients who were guideline compliant, compared with 4.1% of noncompliant patients. Similarly, UTIs were significantly less likely in rectal surgery patients who complied with guidelines, compared with those who did not (3.5% vs. 9.6%).
The Implementation of the Enhanced Recovery After Surgery (iERAS) program involved 15 academic hospitals in Ontario, Canada. The guidelines include prompt removal of catheters from colonic and rectal surgery patients. Recommended removal times ranged from 24 hours to 72 hours, depending on the procedure.
“Although numerous reports have documented the overall effectiveness of bundled ERAS interventions improving outcome, the contribution of individual components of the pathway in the context of an ERAS implementation program is uncertain,” wrote Allan Okrainec, MD, of the University of Toronto, and his colleagues.
Overall, 53% of patients had catheters removed in compliance with the guidelines, including 47.2% of those with colonic resections and 69.5% of those with rectal resections.
The average length of hospital stay for colonic surgery patients who complied with the urinary catheter guideline was 4 days, compared with 5 days for noncompliant patients, a statistically significant difference. Similarly, the average length of stay was significantly shorter for compliant rectal surgery patients, compared with noncompliant patients (5 days vs. 8 days, respectively).
Reinsertion of the urinary catheter was needed for 6% of the patients, including 122 patients with suspected or confirmed urinary retention and 36 patients who had other indications for reinsertion.
“One of the concerns about early removal of catheters is that it might lead to an increased need for reinsertion,” the researchers noted. “In our study, guideline compliance was associated with an increased rate of catheter reinsertion, but [it] only increased the risk by 2%-3% in patients having rectal or colonic procedures,” the investigators said.
The study results were limited by a lack of information about reasons for noncompliance with the guidelines, the researchers added. However, the data support the value of early catheter removal for surgical patients. “It is noteworthy that, in our study, as well as others, urinary retention rates are relatively low,” they said.
The Council of Academic Hospitals of Ontario funded the study. The researchers had no financial conflicts to disclose.
Adherence to guidelines for urinary catheter removal significantly reduced rates of urinary tract infection and length of hospital stay, based on data from almost 3,000 patients.
UTIs occurred in 0.8% of colonic surgery patients who were guideline compliant, compared with 4.1% of noncompliant patients. Similarly, UTIs were significantly less likely in rectal surgery patients who complied with guidelines, compared with those who did not (3.5% vs. 9.6%).
The Implementation of the Enhanced Recovery After Surgery (iERAS) program involved 15 academic hospitals in Ontario, Canada. The guidelines include prompt removal of catheters from colonic and rectal surgery patients. Recommended removal times ranged from 24 hours to 72 hours, depending on the procedure.
“Although numerous reports have documented the overall effectiveness of bundled ERAS interventions improving outcome, the contribution of individual components of the pathway in the context of an ERAS implementation program is uncertain,” wrote Allan Okrainec, MD, of the University of Toronto, and his colleagues.
Overall, 53% of patients had catheters removed in compliance with the guidelines, including 47.2% of those with colonic resections and 69.5% of those with rectal resections.
The average length of hospital stay for colonic surgery patients who complied with the urinary catheter guideline was 4 days, compared with 5 days for noncompliant patients, a statistically significant difference. Similarly, the average length of stay was significantly shorter for compliant rectal surgery patients, compared with noncompliant patients (5 days vs. 8 days, respectively).
Reinsertion of the urinary catheter was needed for 6% of the patients, including 122 patients with suspected or confirmed urinary retention and 36 patients who had other indications for reinsertion.
“One of the concerns about early removal of catheters is that it might lead to an increased need for reinsertion,” the researchers noted. “In our study, guideline compliance was associated with an increased rate of catheter reinsertion, but [it] only increased the risk by 2%-3% in patients having rectal or colonic procedures,” the investigators said.
The study results were limited by a lack of information about reasons for noncompliance with the guidelines, the researchers added. However, the data support the value of early catheter removal for surgical patients. “It is noteworthy that, in our study, as well as others, urinary retention rates are relatively low,” they said.
The Council of Academic Hospitals of Ontario funded the study. The researchers had no financial conflicts to disclose.
FROM THE JOURNAL OF GASTROINTESTINAL SURGERY
Key clinical point: Compliance with guidelines for urinary catheter removal significantly reduced hospital stay and UTI rates in colon and rectal surgery patients.
Major finding: UTI rates in colonic surgery patients were 0.8% and 4.1%, respectively, for those who were compliant and noncompliant with the guidelines. Rates were 3.5% and 9.6%, respectively, for compliant and noncompliant rectal surgery patients.
Data source: A prospective study of 2,927 adults who underwent colonic or rectal surgery between September 2012 and April 2015.
Disclosures: The Council of Academic Hospitals of Ontario funded the study. The researchers had no financial conflicts to disclose.
How to prevent secondary posttraumatic knee osteoarthritis
LAS VEGAS – A variety of evidence-based strategies are available for preventing posttraumatic knee osteoarthritis (KOA) in patients who have already sustained an anterior cruciate ligament (ACL) injury. And they’re generally ignored, according to May Arna Risberg, PhD.
“We have a lot of knowledge. We can use secondary prevention strategies. And here I think we, as physical therapists, physicians, and orthopedic surgeons, are doing a lousy job because we are sending these ACL-injured patients back to sports before they have normalized knee function and quadriceps strength,” said Dr. Risberg, professor of sports medicine at the Norwegian School of Sport Sciences in Oslo.
With no proven disease-modifying therapy for KOA available to date, secondary prevention of posttraumatic KOA is worthy of high-priority status, she said at the World Congress on Osteoarthritis. An estimated 250,00 ACL injuries occur annually in the United States, and up to one-half of affected patients, most of whom are young, active people, will experience a second ACL rupture within the first few years after undergoing their initial reconstruction. This second ACL injury greatly increases their risk of developing posttraumatic KOA within 15-20 years, while they are still relatively young, she said.
Moreover, if the second ACL injury involves meniscus surgery, the 5-year risk of posttraumatic KOA roughly triples to up to 48%.
She highlighted a few effective strategies for preventing posttraumatic KOA in patients who already have an ACL injury.
Avoid reinjury
Dr. Risberg was senior author of a recent report from the prospective Delaware-Oslo Cohort Study involving 106 athletes who underwent ACL reconstruction following injury in what she termed level I sports. These are sports that entail lots of pivoting, jumping, and hard cutting, such as basketball, soccer, and handball.
In the first 2 years after ACL repair, 30% of patients who returned to participation in a level 1 sport experienced an ACL reinjury, compared with just 8% who opted for a lower-level sport. Athletes who returned to a level 1 sport had an adjusted 4.3 times greater ACL reinjury rate than those who didn’t, Dr. Risberg noted at the congress sponsored by the Osteoarthritis Research Society International.
The good news is that this sharply increased reinjury risk was mitigated if return to a level 1 sport was delayed for at least 9 months post surgery and if the patient had regained quadriceps strength comparable to the uninjured side. For every month that return to sport was delayed out until 9 months post ACL reconstruction, the knee reinjury rate was reduced by 51% (Br J Sports Med. 2016;50:804-8).
In a meta-analysis by other investigators of 12 studies including 5,707 participants, weakness of the knee extensor muscles was independently associated with a 1.65 times increased risk of developing KOA (Osteoarthritis Cartilage. 2015 Feb;23[2]:171-7).
Attend to BMI
A discussion of the importance of maintaining a healthy body weight is an important aspect of patient education for athletes with knee injuries. In a cohort study of 988 patients who underwent primary ACL reconstruction, being overweight or obese was associated with a significantly increased risk of subsequent meniscal tears and chondral lesions (Am J Sports Med. 2015 Dec;43[12]:2966-73).
Also, it’s well established that obesity is a risk factor for knee OA, and Canadian investigators have shown that young athletes with a sports-related intra-articular knee injury were 3.75 times more likely to be overweight or obese 3-10 years post injury, compared with matched uninjured controls (Osteoarthritis Cartilage. 2015 Jul;23[7]:1122-9).
Consider prehabilitative exercise training
Dr. Risberg and coinvestigators have reported that preoperative quadriceps muscle strength deficits are predictive of impaired knee function, as measured by the Cincinnati Knee Score 2 years post surgery. She said she believes ACL reconstruction shouldn’t be done until quadriceps muscle strength is at least 80% of that in the uninjured limb (Br J Sports Med. 2009 May;43[5]:371-6). She and her coinvestigators have published the details of a 5-week progressive exercise therapy program in which they have shown results in significantly improved early postoperative knee function (J Orthop Sports Phys Ther. 2010 Nov;40[11]:705-21). They now try to have patients complete the twice-weekly, 5-week program before final decisions are reached regarding whether to have ACL reconstruction.
Test all before okaying return to sport
It’s important to know if patients who have undergone ACL reconstruction have gotten full knee function back before determining if they’re ready for full-on sports participation. In the Delaware-Oslo Cohort Study, patients who delayed their return until at least 9 months after surgery and passed the return-to-sports test had a 5.6% reinjury rate within 2 years, while those who failed the return-to-sports criteria had a 38.2% ACL reinjury rate.
The return-to-sports testing utilized in this study entailed isokinetic quadriceps strength testing, the single hop leg test, the 14-item self-rated Knee Outcome Survey–Activities of Daily Living Scale, and a self-rated Global Rating Scale of perceived function on a 0-100 scale. To be cleared for return to sports, a patient had to demonstrate having regained at least 90% of quadriceps muscle strength and hop performance along with scoring in the normative range on both of the self-rating instruments.
Surgical vs. nonsurgical treatment of ACL rupture
The evidence on this score is conflicting, according to Dr. Risberg. While most physical therapists believe ACL reconstruction doesn’t protect against later development of KOA, as reflected in a meta-analysis of published studies (J Bone Joint Surg Am. 2014 Feb 19;96[4]:292-300), a more recent retrospective comparison of 964 patients with an isolated ACL tear and an equal number of matched controls concluded that patients treated nonoperatively were six times more likely to have been diagnosed with KOA and 16.7 times more likely to have undergone total knee replacement at a mean follow-up of 13.7 years than were those treated with ACL reconstruction (Am J Sports Med. 2016 Jul;44[7]:1699-707).
Dr. Risberg’s fellow panelist Jackie Whittaker, PhD, said that, as long as quadriceps muscle strengthening is a priority, it makes sense to strengthen the hamstring as well, particularly if the ACL reconstruction utilized the hamstring tendon.
“Also, I would add that it’s important to develop a relationship with these ACL-injured people, who are often very young. Preventing a disease that they’re going to get 20 years later isn’t a priority for them. You need to develop that relationship and build it up over time. Helping them set realistic expectations is very important. And we need to do what we can to help them find some sort of competitive outlet. A lot of these kids were very competitive, and now they’ve had an injury and can’t compete. They don’t want to go back to playing just any sport. They want to be able to be competitive, and if you don’t help them find another way to express that, they sort of give up on physical activity altogether,” according to Dr. Whittaker of the University of Alberta in Edmonton.
Dr. Risberg and Dr. Whittaker reported having no financial conflicts of interest.
LAS VEGAS – A variety of evidence-based strategies are available for preventing posttraumatic knee osteoarthritis (KOA) in patients who have already sustained an anterior cruciate ligament (ACL) injury. And they’re generally ignored, according to May Arna Risberg, PhD.
“We have a lot of knowledge. We can use secondary prevention strategies. And here I think we, as physical therapists, physicians, and orthopedic surgeons, are doing a lousy job because we are sending these ACL-injured patients back to sports before they have normalized knee function and quadriceps strength,” said Dr. Risberg, professor of sports medicine at the Norwegian School of Sport Sciences in Oslo.
With no proven disease-modifying therapy for KOA available to date, secondary prevention of posttraumatic KOA is worthy of high-priority status, she said at the World Congress on Osteoarthritis. An estimated 250,00 ACL injuries occur annually in the United States, and up to one-half of affected patients, most of whom are young, active people, will experience a second ACL rupture within the first few years after undergoing their initial reconstruction. This second ACL injury greatly increases their risk of developing posttraumatic KOA within 15-20 years, while they are still relatively young, she said.
Moreover, if the second ACL injury involves meniscus surgery, the 5-year risk of posttraumatic KOA roughly triples to up to 48%.
She highlighted a few effective strategies for preventing posttraumatic KOA in patients who already have an ACL injury.
Avoid reinjury
Dr. Risberg was senior author of a recent report from the prospective Delaware-Oslo Cohort Study involving 106 athletes who underwent ACL reconstruction following injury in what she termed level I sports. These are sports that entail lots of pivoting, jumping, and hard cutting, such as basketball, soccer, and handball.
In the first 2 years after ACL repair, 30% of patients who returned to participation in a level 1 sport experienced an ACL reinjury, compared with just 8% who opted for a lower-level sport. Athletes who returned to a level 1 sport had an adjusted 4.3 times greater ACL reinjury rate than those who didn’t, Dr. Risberg noted at the congress sponsored by the Osteoarthritis Research Society International.
The good news is that this sharply increased reinjury risk was mitigated if return to a level 1 sport was delayed for at least 9 months post surgery and if the patient had regained quadriceps strength comparable to the uninjured side. For every month that return to sport was delayed out until 9 months post ACL reconstruction, the knee reinjury rate was reduced by 51% (Br J Sports Med. 2016;50:804-8).
In a meta-analysis by other investigators of 12 studies including 5,707 participants, weakness of the knee extensor muscles was independently associated with a 1.65 times increased risk of developing KOA (Osteoarthritis Cartilage. 2015 Feb;23[2]:171-7).
Attend to BMI
A discussion of the importance of maintaining a healthy body weight is an important aspect of patient education for athletes with knee injuries. In a cohort study of 988 patients who underwent primary ACL reconstruction, being overweight or obese was associated with a significantly increased risk of subsequent meniscal tears and chondral lesions (Am J Sports Med. 2015 Dec;43[12]:2966-73).
Also, it’s well established that obesity is a risk factor for knee OA, and Canadian investigators have shown that young athletes with a sports-related intra-articular knee injury were 3.75 times more likely to be overweight or obese 3-10 years post injury, compared with matched uninjured controls (Osteoarthritis Cartilage. 2015 Jul;23[7]:1122-9).
Consider prehabilitative exercise training
Dr. Risberg and coinvestigators have reported that preoperative quadriceps muscle strength deficits are predictive of impaired knee function, as measured by the Cincinnati Knee Score 2 years post surgery. She said she believes ACL reconstruction shouldn’t be done until quadriceps muscle strength is at least 80% of that in the uninjured limb (Br J Sports Med. 2009 May;43[5]:371-6). She and her coinvestigators have published the details of a 5-week progressive exercise therapy program in which they have shown results in significantly improved early postoperative knee function (J Orthop Sports Phys Ther. 2010 Nov;40[11]:705-21). They now try to have patients complete the twice-weekly, 5-week program before final decisions are reached regarding whether to have ACL reconstruction.
Test all before okaying return to sport
It’s important to know if patients who have undergone ACL reconstruction have gotten full knee function back before determining if they’re ready for full-on sports participation. In the Delaware-Oslo Cohort Study, patients who delayed their return until at least 9 months after surgery and passed the return-to-sports test had a 5.6% reinjury rate within 2 years, while those who failed the return-to-sports criteria had a 38.2% ACL reinjury rate.
The return-to-sports testing utilized in this study entailed isokinetic quadriceps strength testing, the single hop leg test, the 14-item self-rated Knee Outcome Survey–Activities of Daily Living Scale, and a self-rated Global Rating Scale of perceived function on a 0-100 scale. To be cleared for return to sports, a patient had to demonstrate having regained at least 90% of quadriceps muscle strength and hop performance along with scoring in the normative range on both of the self-rating instruments.
Surgical vs. nonsurgical treatment of ACL rupture
The evidence on this score is conflicting, according to Dr. Risberg. While most physical therapists believe ACL reconstruction doesn’t protect against later development of KOA, as reflected in a meta-analysis of published studies (J Bone Joint Surg Am. 2014 Feb 19;96[4]:292-300), a more recent retrospective comparison of 964 patients with an isolated ACL tear and an equal number of matched controls concluded that patients treated nonoperatively were six times more likely to have been diagnosed with KOA and 16.7 times more likely to have undergone total knee replacement at a mean follow-up of 13.7 years than were those treated with ACL reconstruction (Am J Sports Med. 2016 Jul;44[7]:1699-707).
Dr. Risberg’s fellow panelist Jackie Whittaker, PhD, said that, as long as quadriceps muscle strengthening is a priority, it makes sense to strengthen the hamstring as well, particularly if the ACL reconstruction utilized the hamstring tendon.
“Also, I would add that it’s important to develop a relationship with these ACL-injured people, who are often very young. Preventing a disease that they’re going to get 20 years later isn’t a priority for them. You need to develop that relationship and build it up over time. Helping them set realistic expectations is very important. And we need to do what we can to help them find some sort of competitive outlet. A lot of these kids were very competitive, and now they’ve had an injury and can’t compete. They don’t want to go back to playing just any sport. They want to be able to be competitive, and if you don’t help them find another way to express that, they sort of give up on physical activity altogether,” according to Dr. Whittaker of the University of Alberta in Edmonton.
Dr. Risberg and Dr. Whittaker reported having no financial conflicts of interest.
LAS VEGAS – A variety of evidence-based strategies are available for preventing posttraumatic knee osteoarthritis (KOA) in patients who have already sustained an anterior cruciate ligament (ACL) injury. And they’re generally ignored, according to May Arna Risberg, PhD.
“We have a lot of knowledge. We can use secondary prevention strategies. And here I think we, as physical therapists, physicians, and orthopedic surgeons, are doing a lousy job because we are sending these ACL-injured patients back to sports before they have normalized knee function and quadriceps strength,” said Dr. Risberg, professor of sports medicine at the Norwegian School of Sport Sciences in Oslo.
With no proven disease-modifying therapy for KOA available to date, secondary prevention of posttraumatic KOA is worthy of high-priority status, she said at the World Congress on Osteoarthritis. An estimated 250,00 ACL injuries occur annually in the United States, and up to one-half of affected patients, most of whom are young, active people, will experience a second ACL rupture within the first few years after undergoing their initial reconstruction. This second ACL injury greatly increases their risk of developing posttraumatic KOA within 15-20 years, while they are still relatively young, she said.
Moreover, if the second ACL injury involves meniscus surgery, the 5-year risk of posttraumatic KOA roughly triples to up to 48%.
She highlighted a few effective strategies for preventing posttraumatic KOA in patients who already have an ACL injury.
Avoid reinjury
Dr. Risberg was senior author of a recent report from the prospective Delaware-Oslo Cohort Study involving 106 athletes who underwent ACL reconstruction following injury in what she termed level I sports. These are sports that entail lots of pivoting, jumping, and hard cutting, such as basketball, soccer, and handball.
In the first 2 years after ACL repair, 30% of patients who returned to participation in a level 1 sport experienced an ACL reinjury, compared with just 8% who opted for a lower-level sport. Athletes who returned to a level 1 sport had an adjusted 4.3 times greater ACL reinjury rate than those who didn’t, Dr. Risberg noted at the congress sponsored by the Osteoarthritis Research Society International.
The good news is that this sharply increased reinjury risk was mitigated if return to a level 1 sport was delayed for at least 9 months post surgery and if the patient had regained quadriceps strength comparable to the uninjured side. For every month that return to sport was delayed out until 9 months post ACL reconstruction, the knee reinjury rate was reduced by 51% (Br J Sports Med. 2016;50:804-8).
In a meta-analysis by other investigators of 12 studies including 5,707 participants, weakness of the knee extensor muscles was independently associated with a 1.65 times increased risk of developing KOA (Osteoarthritis Cartilage. 2015 Feb;23[2]:171-7).
Attend to BMI
A discussion of the importance of maintaining a healthy body weight is an important aspect of patient education for athletes with knee injuries. In a cohort study of 988 patients who underwent primary ACL reconstruction, being overweight or obese was associated with a significantly increased risk of subsequent meniscal tears and chondral lesions (Am J Sports Med. 2015 Dec;43[12]:2966-73).
Also, it’s well established that obesity is a risk factor for knee OA, and Canadian investigators have shown that young athletes with a sports-related intra-articular knee injury were 3.75 times more likely to be overweight or obese 3-10 years post injury, compared with matched uninjured controls (Osteoarthritis Cartilage. 2015 Jul;23[7]:1122-9).
Consider prehabilitative exercise training
Dr. Risberg and coinvestigators have reported that preoperative quadriceps muscle strength deficits are predictive of impaired knee function, as measured by the Cincinnati Knee Score 2 years post surgery. She said she believes ACL reconstruction shouldn’t be done until quadriceps muscle strength is at least 80% of that in the uninjured limb (Br J Sports Med. 2009 May;43[5]:371-6). She and her coinvestigators have published the details of a 5-week progressive exercise therapy program in which they have shown results in significantly improved early postoperative knee function (J Orthop Sports Phys Ther. 2010 Nov;40[11]:705-21). They now try to have patients complete the twice-weekly, 5-week program before final decisions are reached regarding whether to have ACL reconstruction.
Test all before okaying return to sport
It’s important to know if patients who have undergone ACL reconstruction have gotten full knee function back before determining if they’re ready for full-on sports participation. In the Delaware-Oslo Cohort Study, patients who delayed their return until at least 9 months after surgery and passed the return-to-sports test had a 5.6% reinjury rate within 2 years, while those who failed the return-to-sports criteria had a 38.2% ACL reinjury rate.
The return-to-sports testing utilized in this study entailed isokinetic quadriceps strength testing, the single hop leg test, the 14-item self-rated Knee Outcome Survey–Activities of Daily Living Scale, and a self-rated Global Rating Scale of perceived function on a 0-100 scale. To be cleared for return to sports, a patient had to demonstrate having regained at least 90% of quadriceps muscle strength and hop performance along with scoring in the normative range on both of the self-rating instruments.
Surgical vs. nonsurgical treatment of ACL rupture
The evidence on this score is conflicting, according to Dr. Risberg. While most physical therapists believe ACL reconstruction doesn’t protect against later development of KOA, as reflected in a meta-analysis of published studies (J Bone Joint Surg Am. 2014 Feb 19;96[4]:292-300), a more recent retrospective comparison of 964 patients with an isolated ACL tear and an equal number of matched controls concluded that patients treated nonoperatively were six times more likely to have been diagnosed with KOA and 16.7 times more likely to have undergone total knee replacement at a mean follow-up of 13.7 years than were those treated with ACL reconstruction (Am J Sports Med. 2016 Jul;44[7]:1699-707).
Dr. Risberg’s fellow panelist Jackie Whittaker, PhD, said that, as long as quadriceps muscle strengthening is a priority, it makes sense to strengthen the hamstring as well, particularly if the ACL reconstruction utilized the hamstring tendon.
“Also, I would add that it’s important to develop a relationship with these ACL-injured people, who are often very young. Preventing a disease that they’re going to get 20 years later isn’t a priority for them. You need to develop that relationship and build it up over time. Helping them set realistic expectations is very important. And we need to do what we can to help them find some sort of competitive outlet. A lot of these kids were very competitive, and now they’ve had an injury and can’t compete. They don’t want to go back to playing just any sport. They want to be able to be competitive, and if you don’t help them find another way to express that, they sort of give up on physical activity altogether,” according to Dr. Whittaker of the University of Alberta in Edmonton.
Dr. Risberg and Dr. Whittaker reported having no financial conflicts of interest.
EXPERT ANALYSIS FROM OARSI 2017
Surgeon volume tied to mitral valve surgery outcomes
CHICAGO – A total annual surgeon volume of fewer than 25 operations was associated with increased 1-year mortality and reoperation rates, according to a study presented at the 2017 American Association for Thoracic Surgery Centennial.
Improvements in repair rates, survival, and freedom from reoperation increased with increasing surgeon case volumes, Joanna Chikwe, MD, and her colleagues at the Icahn School of Medicine at Mount Sinai, New York, stated.
The researchers compared repair rates, long-term survival, and risk of postrepair reoperation in patients with degenerative disease according to total annual surgeon volume, which was defined as any mitral valve operation for any cause during the study period. The study was simultaneously published in the Journal of the American College of Cardiology (2017 May 16;69[19]:2397-406).
Mitral valve repair is the preferred treatment, compared with valve replacement, for the treatment of severe mitral valve regurgitation in patients who have degenerative valve disease with mitral valve prolapse, and both U.S. and European guidelines strongly recommend valve repair whenever possible, according to Dr. Chikwe and her colleagues.
But, “mitral valve replacement unfortunately remains relatively common in patients with degenerative valve disease,” they stated.
A total of 313 surgeons from 41 institutions met the study eligibility criteria, according to the researchers. They performed a median of 10 mitral valve operations per year (range, 1-230). The median annual institutional mitral valve volume was 59 mitral valve operations, ranging from a minimum of 6 to a maximum of 310 operations. Repair rates for primary mitral valve operations for any cause at all 41 institutions varied from 15% to 83%, and repair rates for degenerative mitral valve operations varied from 25% to 100%.
In the study cohort, surgeons with a total annual volume of less than 25 operations carried out 25% of procedures.
After multivariable adjustment, total annual surgeon volume was independently associated with the probability of mitral valve repair. The chance of repair increased significantly by 13% for every 10-case increment in total annual surgeon volume (P less than .001).
In addition, compared with patients operated on by surgeons with a total annual surgeon volume of 10 or fewer operations, patients operated on by surgeons with a total annual surgeon volume of greater than 50 operations were more than three times as likely to undergo mitral valve repair rather than replacement.
A total annual surgeon volume of less than 25 operations was associated with lower mitral valve repair rates and with increased 1-year mortality and mitral valve reoperation rates. Improvements in repair rates, survival, and freedom from reoperation increased with increasing surgeon case volumes.
After 1 year of repair or replacement, the actuarial survival of patients with degenerative mitral valve disease who were operated on by surgeons performing greater than 50 operations per year was 97.8%, compared with 94.1% for patients operated on by surgeons performing less than or equal to 10 operations a year.
Compared with replacement, mitral repair was significantly associated with better survival, but total annual surgeon volume still remained a significant independent predictor (P less than .001). In addition, for those patients who underwent mitral valve repair, total annual surgeon volume was a significant independent predictor of late death.
The results are important, the investigators noted, given that the median number of mitral valve operations performed annually by individual surgeons in the United States was five, according to an analysis of the Society of Thoracic Surgeons database – and that, in New York state, most surgeons actually performed less than one mitral operation per month.
There were significant differences seen in the patient characteristics across surgeons’ case volume groups. The prevalence of congestive heart failure was significantly higher in patients operated on by surgeons with lower volumes, and the proportion of patients undergoing urgent surgery was also significantly higher for lower-volume surgeons.
“This leads to a double jeopardy, where sicker patients are adversely affected by the lower repair rates and poorer outcomes seen with lower-volume surgeons, and it underscores the need to refer the highest-risk patients to high-volume surgeons,” said Dr. Chickwe and her colleagues.
However, “even among high-volume surgeons, there was an observed variability of degenerative disease repair rates, ranging from 19% to nearly 100%,” they added. “This finding reflects that surgeon volume is not the only factor for better outcomes, and it emphasizes the need for more transparency of surgeon-related factors and outcomes of degenerative mitral valve surgery for patients and referring cardiologists.
“Considering that there was an incremental improvement in survival and probability of repair with increasing volume over 25 operations, one could make the argument that a minimum volume target of 50, or even more, operations would be optimal,” the researchers noted. “Developing more very high-volume surgeons experienced in mitral valve repair would likely be particularly beneficial for patients with complex but repairable mitral valve disease, and for asymptomatic patients whose repair feasibility would optimally approach 100%.”
Dr. David Adams is the national coprincipal investigator of the Core Valve United States Pivotal Trial, supported by Medtronic. Dr. Chikwe received speaker honoraria from Edwards Lifesciences. The other coauthors had no disclosures to report.
CHICAGO – A total annual surgeon volume of fewer than 25 operations was associated with increased 1-year mortality and reoperation rates, according to a study presented at the 2017 American Association for Thoracic Surgery Centennial.
Improvements in repair rates, survival, and freedom from reoperation increased with increasing surgeon case volumes, Joanna Chikwe, MD, and her colleagues at the Icahn School of Medicine at Mount Sinai, New York, stated.
The researchers compared repair rates, long-term survival, and risk of postrepair reoperation in patients with degenerative disease according to total annual surgeon volume, which was defined as any mitral valve operation for any cause during the study period. The study was simultaneously published in the Journal of the American College of Cardiology (2017 May 16;69[19]:2397-406).
Mitral valve repair is the preferred treatment, compared with valve replacement, for the treatment of severe mitral valve regurgitation in patients who have degenerative valve disease with mitral valve prolapse, and both U.S. and European guidelines strongly recommend valve repair whenever possible, according to Dr. Chikwe and her colleagues.
But, “mitral valve replacement unfortunately remains relatively common in patients with degenerative valve disease,” they stated.
A total of 313 surgeons from 41 institutions met the study eligibility criteria, according to the researchers. They performed a median of 10 mitral valve operations per year (range, 1-230). The median annual institutional mitral valve volume was 59 mitral valve operations, ranging from a minimum of 6 to a maximum of 310 operations. Repair rates for primary mitral valve operations for any cause at all 41 institutions varied from 15% to 83%, and repair rates for degenerative mitral valve operations varied from 25% to 100%.
In the study cohort, surgeons with a total annual volume of less than 25 operations carried out 25% of procedures.
After multivariable adjustment, total annual surgeon volume was independently associated with the probability of mitral valve repair. The chance of repair increased significantly by 13% for every 10-case increment in total annual surgeon volume (P less than .001).
In addition, compared with patients operated on by surgeons with a total annual surgeon volume of 10 or fewer operations, patients operated on by surgeons with a total annual surgeon volume of greater than 50 operations were more than three times as likely to undergo mitral valve repair rather than replacement.
A total annual surgeon volume of less than 25 operations was associated with lower mitral valve repair rates and with increased 1-year mortality and mitral valve reoperation rates. Improvements in repair rates, survival, and freedom from reoperation increased with increasing surgeon case volumes.
After 1 year of repair or replacement, the actuarial survival of patients with degenerative mitral valve disease who were operated on by surgeons performing greater than 50 operations per year was 97.8%, compared with 94.1% for patients operated on by surgeons performing less than or equal to 10 operations a year.
Compared with replacement, mitral repair was significantly associated with better survival, but total annual surgeon volume still remained a significant independent predictor (P less than .001). In addition, for those patients who underwent mitral valve repair, total annual surgeon volume was a significant independent predictor of late death.
The results are important, the investigators noted, given that the median number of mitral valve operations performed annually by individual surgeons in the United States was five, according to an analysis of the Society of Thoracic Surgeons database – and that, in New York state, most surgeons actually performed less than one mitral operation per month.
There were significant differences seen in the patient characteristics across surgeons’ case volume groups. The prevalence of congestive heart failure was significantly higher in patients operated on by surgeons with lower volumes, and the proportion of patients undergoing urgent surgery was also significantly higher for lower-volume surgeons.
“This leads to a double jeopardy, where sicker patients are adversely affected by the lower repair rates and poorer outcomes seen with lower-volume surgeons, and it underscores the need to refer the highest-risk patients to high-volume surgeons,” said Dr. Chickwe and her colleagues.
However, “even among high-volume surgeons, there was an observed variability of degenerative disease repair rates, ranging from 19% to nearly 100%,” they added. “This finding reflects that surgeon volume is not the only factor for better outcomes, and it emphasizes the need for more transparency of surgeon-related factors and outcomes of degenerative mitral valve surgery for patients and referring cardiologists.
“Considering that there was an incremental improvement in survival and probability of repair with increasing volume over 25 operations, one could make the argument that a minimum volume target of 50, or even more, operations would be optimal,” the researchers noted. “Developing more very high-volume surgeons experienced in mitral valve repair would likely be particularly beneficial for patients with complex but repairable mitral valve disease, and for asymptomatic patients whose repair feasibility would optimally approach 100%.”
Dr. David Adams is the national coprincipal investigator of the Core Valve United States Pivotal Trial, supported by Medtronic. Dr. Chikwe received speaker honoraria from Edwards Lifesciences. The other coauthors had no disclosures to report.
CHICAGO – A total annual surgeon volume of fewer than 25 operations was associated with increased 1-year mortality and reoperation rates, according to a study presented at the 2017 American Association for Thoracic Surgery Centennial.
Improvements in repair rates, survival, and freedom from reoperation increased with increasing surgeon case volumes, Joanna Chikwe, MD, and her colleagues at the Icahn School of Medicine at Mount Sinai, New York, stated.
The researchers compared repair rates, long-term survival, and risk of postrepair reoperation in patients with degenerative disease according to total annual surgeon volume, which was defined as any mitral valve operation for any cause during the study period. The study was simultaneously published in the Journal of the American College of Cardiology (2017 May 16;69[19]:2397-406).
Mitral valve repair is the preferred treatment, compared with valve replacement, for the treatment of severe mitral valve regurgitation in patients who have degenerative valve disease with mitral valve prolapse, and both U.S. and European guidelines strongly recommend valve repair whenever possible, according to Dr. Chikwe and her colleagues.
But, “mitral valve replacement unfortunately remains relatively common in patients with degenerative valve disease,” they stated.
A total of 313 surgeons from 41 institutions met the study eligibility criteria, according to the researchers. They performed a median of 10 mitral valve operations per year (range, 1-230). The median annual institutional mitral valve volume was 59 mitral valve operations, ranging from a minimum of 6 to a maximum of 310 operations. Repair rates for primary mitral valve operations for any cause at all 41 institutions varied from 15% to 83%, and repair rates for degenerative mitral valve operations varied from 25% to 100%.
In the study cohort, surgeons with a total annual volume of less than 25 operations carried out 25% of procedures.
After multivariable adjustment, total annual surgeon volume was independently associated with the probability of mitral valve repair. The chance of repair increased significantly by 13% for every 10-case increment in total annual surgeon volume (P less than .001).
In addition, compared with patients operated on by surgeons with a total annual surgeon volume of 10 or fewer operations, patients operated on by surgeons with a total annual surgeon volume of greater than 50 operations were more than three times as likely to undergo mitral valve repair rather than replacement.
A total annual surgeon volume of less than 25 operations was associated with lower mitral valve repair rates and with increased 1-year mortality and mitral valve reoperation rates. Improvements in repair rates, survival, and freedom from reoperation increased with increasing surgeon case volumes.
After 1 year of repair or replacement, the actuarial survival of patients with degenerative mitral valve disease who were operated on by surgeons performing greater than 50 operations per year was 97.8%, compared with 94.1% for patients operated on by surgeons performing less than or equal to 10 operations a year.
Compared with replacement, mitral repair was significantly associated with better survival, but total annual surgeon volume still remained a significant independent predictor (P less than .001). In addition, for those patients who underwent mitral valve repair, total annual surgeon volume was a significant independent predictor of late death.
The results are important, the investigators noted, given that the median number of mitral valve operations performed annually by individual surgeons in the United States was five, according to an analysis of the Society of Thoracic Surgeons database – and that, in New York state, most surgeons actually performed less than one mitral operation per month.
There were significant differences seen in the patient characteristics across surgeons’ case volume groups. The prevalence of congestive heart failure was significantly higher in patients operated on by surgeons with lower volumes, and the proportion of patients undergoing urgent surgery was also significantly higher for lower-volume surgeons.
“This leads to a double jeopardy, where sicker patients are adversely affected by the lower repair rates and poorer outcomes seen with lower-volume surgeons, and it underscores the need to refer the highest-risk patients to high-volume surgeons,” said Dr. Chickwe and her colleagues.
However, “even among high-volume surgeons, there was an observed variability of degenerative disease repair rates, ranging from 19% to nearly 100%,” they added. “This finding reflects that surgeon volume is not the only factor for better outcomes, and it emphasizes the need for more transparency of surgeon-related factors and outcomes of degenerative mitral valve surgery for patients and referring cardiologists.
“Considering that there was an incremental improvement in survival and probability of repair with increasing volume over 25 operations, one could make the argument that a minimum volume target of 50, or even more, operations would be optimal,” the researchers noted. “Developing more very high-volume surgeons experienced in mitral valve repair would likely be particularly beneficial for patients with complex but repairable mitral valve disease, and for asymptomatic patients whose repair feasibility would optimally approach 100%.”
Dr. David Adams is the national coprincipal investigator of the Core Valve United States Pivotal Trial, supported by Medtronic. Dr. Chikwe received speaker honoraria from Edwards Lifesciences. The other coauthors had no disclosures to report.
FROM THE AATS ANNUAL MEETING AND THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Key clinical point:
Major finding: Mitral valve reoperation rates steadily decreased with increasing surgeon volume until 25 operations per year, coupled to an improved 1-year survival for every 10 additional operations more than that.
Data source: A mandatory New York state database containing 5,475 patients who underwent mitral valve repair between 2002 and 2013.
Disclosures: Dr. David Adams is the national coprincipal investigator of the Core Valve United States Pivotal Trial, supported by Medtronic. Dr. Joanna Chikwe received speaker honoraria from Edwards Lifesciences. The other coauthors had no disclosures to report.
CMS proposes exempting more practices from MACRA
The Centers for Medicare & Medicaid Services seeks to exempt more practices participating in Medicare’s Quality Payment Program, the value-based payment program created by Medicare Access and CHIP Reauthorization Act of 2015 (MACRA).
Currently, physicians who receive $30,000 or less in Medicare Part B payments or have 100 or fewer Medicare patients are exempt from QPP but may choose to participate. The proposed rule for the second year of QPP (calendar year 2018) would raise the threshold to $90,000 or less in Part B payments or 200 or fewer Medicare patients. The proposed rule was released June 20.
Under QPP, physicians and practices that are not exempt will choose one of two tracks – the Merit-Based Incentive Payment System (MIPS), which will pay bonuses based on meeting certain quality thresholds, or the advanced alternative payment models (APMs), under which physicians will assume more risk in exchange for greater potential rewards for quality improvement.
“We’ve heard the concerns that too many quality programs, technology requirements, and measures get between the doctor and the patient,” CMS Administrator Seema Verma said in a statement. “That’s why we’re taking a hard look at reducing burdens. By proposing this rule, we aim to improve Medicare by helping doctors and clinicians concentrate on caring for their patients rather than filling out paperwork.”
The proposed rule also introduces the framework for the development of “virtual groups,” a mechanism by which small and solo practices who meet the eligibility requirements for participating in MIPS can pool their reporting into the program to make it easier to share in the bonuses for meeting quality thresholds.
The Centers for Medicare & Medicaid Services seeks to exempt more practices participating in Medicare’s Quality Payment Program, the value-based payment program created by Medicare Access and CHIP Reauthorization Act of 2015 (MACRA).
Currently, physicians who receive $30,000 or less in Medicare Part B payments or have 100 or fewer Medicare patients are exempt from QPP but may choose to participate. The proposed rule for the second year of QPP (calendar year 2018) would raise the threshold to $90,000 or less in Part B payments or 200 or fewer Medicare patients. The proposed rule was released June 20.
Under QPP, physicians and practices that are not exempt will choose one of two tracks – the Merit-Based Incentive Payment System (MIPS), which will pay bonuses based on meeting certain quality thresholds, or the advanced alternative payment models (APMs), under which physicians will assume more risk in exchange for greater potential rewards for quality improvement.
“We’ve heard the concerns that too many quality programs, technology requirements, and measures get between the doctor and the patient,” CMS Administrator Seema Verma said in a statement. “That’s why we’re taking a hard look at reducing burdens. By proposing this rule, we aim to improve Medicare by helping doctors and clinicians concentrate on caring for their patients rather than filling out paperwork.”
The proposed rule also introduces the framework for the development of “virtual groups,” a mechanism by which small and solo practices who meet the eligibility requirements for participating in MIPS can pool their reporting into the program to make it easier to share in the bonuses for meeting quality thresholds.
The Centers for Medicare & Medicaid Services seeks to exempt more practices participating in Medicare’s Quality Payment Program, the value-based payment program created by Medicare Access and CHIP Reauthorization Act of 2015 (MACRA).
Currently, physicians who receive $30,000 or less in Medicare Part B payments or have 100 or fewer Medicare patients are exempt from QPP but may choose to participate. The proposed rule for the second year of QPP (calendar year 2018) would raise the threshold to $90,000 or less in Part B payments or 200 or fewer Medicare patients. The proposed rule was released June 20.
Under QPP, physicians and practices that are not exempt will choose one of two tracks – the Merit-Based Incentive Payment System (MIPS), which will pay bonuses based on meeting certain quality thresholds, or the advanced alternative payment models (APMs), under which physicians will assume more risk in exchange for greater potential rewards for quality improvement.
“We’ve heard the concerns that too many quality programs, technology requirements, and measures get between the doctor and the patient,” CMS Administrator Seema Verma said in a statement. “That’s why we’re taking a hard look at reducing burdens. By proposing this rule, we aim to improve Medicare by helping doctors and clinicians concentrate on caring for their patients rather than filling out paperwork.”
The proposed rule also introduces the framework for the development of “virtual groups,” a mechanism by which small and solo practices who meet the eligibility requirements for participating in MIPS can pool their reporting into the program to make it easier to share in the bonuses for meeting quality thresholds.
Points/Counterpoint: Should surgeons operate on functional tricuspid regurgitation?
Yes, functional TR is worth repairing (David H. Adams, MD)
Functional tricuspid regurgitation is a common finding in patients undergoing degenerative mitral valve repair. Severe tricuspid regurgitation is unusual, and clearly there is little debate on the merits of concomitant tricuspid repair for these patients. Moderate tricuspid regurgitation is identified preoperatively in around 15% of patients undergoing degenerative mitral repair (J Thorac Cardiovasc Surg. 2011;142:608-13), and concomitant tricuspid repair in these patients is certainly supported by both the American and European guidelines (J Am Coll Cardiol. 2017. doi: 10.1016/j.jacc.2017.03.011; Eur Heart J. 2012;33:2451-96).
What experience and evidence has led us to a more aggressive approach? One of the most important influences on our early adoption of tricuspid repair at the time of mitral surgery was linked to observations that tricuspid regurgitation (TR) sometimes progressed after isolated mitral valve repair (MVR), with some patients developing moderate or worse insufficiency. Certainly, the impact of significant tricuspid regurgitation on the quality and length of patients’ lives and the challenges of reoperation for isolated tricuspid regurgitation are well known to all surgeons.
Consequently, the importance of treating significant annular dilatation, even without significant tricuspid regurgitation, is supported by the guidelines. Our own experience with an aggressive approach to functional tricuspid regurgitation (FTR) at the time of mitral surgery put an exclamation point on this (J Am Coll Cardiol. 2015;65:1931-8). We found that concomitant tricuspid repair in patients who were worse off before surgery with more TR and higher rates of atrial fibrillation and right-sided dysfunction, actually did better during 5 years of follow-up than the isolated mitral repair patients who started with completely normal tricuspid valve anatomy and ventricular function.
Benign neglect is always an option (J Thorac Cardiovasc Surg. 2017;154:125-6), but we agree with Roberto Dion, MD – despite our friends’ opinions in Toronto and Rochester – we would much prefer to have minimal TR and a normal sized tricuspid valve after MVR. Ask yourself: would you rather have no TR and a normal sized tricuspid valve after you undergo a mitral operation, or a very dilated annulus and perhaps moderate FTR? I am pretty sure I know the answer, but if you are not sure, read our paper.
Dr. Adams is cardiac surgeon-in-chief, Mount Sinai Health System, and Marie-Josée and Henry R. Kravis Professor and Chairman, department of cardiovascular surgery, Icahn School of Medicine at Mount Sinai and The Mount Sinai Hospital, and president-elect of the American Association for Thoracic Surgery. He disclosed he is the national co-principal investigator for the Medtronic NeoChord trial, and receives royalties from Medtronic and Edwards Lifesciences. The Icahn School of Medicine at Mount Sinai receives royalty payments from Edwards Lifesciences and Medtronic for intellectual property related to Dr. Adams’ involvement in the development of 2 mitral valve repair rings and 1 tricuspid valve repair ring.
No, a patient with FTR does not necessarily need repair (Tirone David, MD)
In our clinic, a patient who undergoes MVR and has FTR generally goes home without an annuloplasty. We now have 12 years or more of follow-up in these patients, and they do not develop TR if their MVR is competent. We have reported that preoperative TR in patients who had MVR is associated with mitral valve disease and often improves after the operation (J Thorac Cardiovasc Surg. 2017;154:110-22). New postoperative TR is uncommon.
Ninety percent of my mitral valve repair patients today have no symptoms. Of those patients, a small proportion have moderate TR.
Dr. David is a professor of surgery at Toronto General Hospital. He reported no financial relationships.
Yes, but repair of FTR requires caution (Gilles Dreyfus, MD)
The controversy surrounding the legitimacy of concomitant tricuspid annuloplasty for functional TR during MVR begs for a clinical trial, but before we can conduct a clinical trial, we must define the primary and secondary endpoints. We’ve seen recent prospective, randomized trials that have reported faulty conclusions because the primary endpoints were wrong.
We need a strong debate to agree on those endpoints. Mortality as an endpoint will probably take a very long time to arrive at.
We’re mixing up many different factors. We’re mixing up TR grading, and we know that grading is unreliable. We have all seen patients with full-fledged TR, and after we put them on Lasix (furosemide, Sanofi), 3 days later they have mild TR. So the same patient with no treatment becomes let’s say a “Dreyfus indication,” and then suddenly in 3 days the patient doesn’t need surgery. At any further stage of his life this patient can experience severe TR again; tricuspid annuloplasty will prevent that from happening.
Moderate TR according to the common definition does not exist. If you look at the reports in echocardiography and if you ask any cardiologist, everything between no TR and severe TR is considered moderate. We have proposed a new staging system for evaluating FTR that uses three factors: TR severity; annular dilatation; and extent of tethering, or mode of leaflet coaptation (J Am Coll Cardiol. 2015;65:2331-6).
Dr. Dreyfus is director of the medical and surgical team at the Cardiothoracic Centre of Monaco in Monte Carlo and professor of cardiothoracic surgery at Paris V University and the Imperial College of London. He disclosed receiving speaker fees from Edwards Lifesciences, LivaNova, and Medtronic.
No, few FTR patients at risk after MVR (Hartzell Schaff, MD)
Echocardiography can provide a great deal of information about when concomitant repair for TR is indicated during MVR. We’ve found that if the patient has no right-sided signs, has normal right atrial pressure, and has mild or mild/moderate TR at the time of repair to the journey mitral valve, the chance of him returning for a tricuspid valve procedure is near zero.
A few patients do return after MVR. They develop atrial fibrillation and may need a pacemaker, but we see very few patients return for tricuspid surgery.
It’s fair to say we cannot demonstrate any benefit of correcting functional mitral regurgitation. Why would we think there’s a benefit of correcting FTR? We can say we’re going to look at grade of TR down the road, or we could look at some other endpoint, but think about it this way: If we cannot prove that correcting functional mitral regurgitation is helpful, why is correcting FTR going to help?
Dr. Schaff is a cardiothoracic surgeon at Mayo Clinic Foundation, Rochester, Minn. He reported no financial relationships.
Yes, functional TR is worth repairing (David H. Adams, MD)
Functional tricuspid regurgitation is a common finding in patients undergoing degenerative mitral valve repair. Severe tricuspid regurgitation is unusual, and clearly there is little debate on the merits of concomitant tricuspid repair for these patients. Moderate tricuspid regurgitation is identified preoperatively in around 15% of patients undergoing degenerative mitral repair (J Thorac Cardiovasc Surg. 2011;142:608-13), and concomitant tricuspid repair in these patients is certainly supported by both the American and European guidelines (J Am Coll Cardiol. 2017. doi: 10.1016/j.jacc.2017.03.011; Eur Heart J. 2012;33:2451-96).
What experience and evidence has led us to a more aggressive approach? One of the most important influences on our early adoption of tricuspid repair at the time of mitral surgery was linked to observations that tricuspid regurgitation (TR) sometimes progressed after isolated mitral valve repair (MVR), with some patients developing moderate or worse insufficiency. Certainly, the impact of significant tricuspid regurgitation on the quality and length of patients’ lives and the challenges of reoperation for isolated tricuspid regurgitation are well known to all surgeons.
Consequently, the importance of treating significant annular dilatation, even without significant tricuspid regurgitation, is supported by the guidelines. Our own experience with an aggressive approach to functional tricuspid regurgitation (FTR) at the time of mitral surgery put an exclamation point on this (J Am Coll Cardiol. 2015;65:1931-8). We found that concomitant tricuspid repair in patients who were worse off before surgery with more TR and higher rates of atrial fibrillation and right-sided dysfunction, actually did better during 5 years of follow-up than the isolated mitral repair patients who started with completely normal tricuspid valve anatomy and ventricular function.
Benign neglect is always an option (J Thorac Cardiovasc Surg. 2017;154:125-6), but we agree with Roberto Dion, MD – despite our friends’ opinions in Toronto and Rochester – we would much prefer to have minimal TR and a normal sized tricuspid valve after MVR. Ask yourself: would you rather have no TR and a normal sized tricuspid valve after you undergo a mitral operation, or a very dilated annulus and perhaps moderate FTR? I am pretty sure I know the answer, but if you are not sure, read our paper.
Dr. Adams is cardiac surgeon-in-chief, Mount Sinai Health System, and Marie-Josée and Henry R. Kravis Professor and Chairman, department of cardiovascular surgery, Icahn School of Medicine at Mount Sinai and The Mount Sinai Hospital, and president-elect of the American Association for Thoracic Surgery. He disclosed he is the national co-principal investigator for the Medtronic NeoChord trial, and receives royalties from Medtronic and Edwards Lifesciences. The Icahn School of Medicine at Mount Sinai receives royalty payments from Edwards Lifesciences and Medtronic for intellectual property related to Dr. Adams’ involvement in the development of 2 mitral valve repair rings and 1 tricuspid valve repair ring.
No, a patient with FTR does not necessarily need repair (Tirone David, MD)
In our clinic, a patient who undergoes MVR and has FTR generally goes home without an annuloplasty. We now have 12 years or more of follow-up in these patients, and they do not develop TR if their MVR is competent. We have reported that preoperative TR in patients who had MVR is associated with mitral valve disease and often improves after the operation (J Thorac Cardiovasc Surg. 2017;154:110-22). New postoperative TR is uncommon.
Ninety percent of my mitral valve repair patients today have no symptoms. Of those patients, a small proportion have moderate TR.
Dr. David is a professor of surgery at Toronto General Hospital. He reported no financial relationships.
Yes, but repair of FTR requires caution (Gilles Dreyfus, MD)
The controversy surrounding the legitimacy of concomitant tricuspid annuloplasty for functional TR during MVR begs for a clinical trial, but before we can conduct a clinical trial, we must define the primary and secondary endpoints. We’ve seen recent prospective, randomized trials that have reported faulty conclusions because the primary endpoints were wrong.
We need a strong debate to agree on those endpoints. Mortality as an endpoint will probably take a very long time to arrive at.
We’re mixing up many different factors. We’re mixing up TR grading, and we know that grading is unreliable. We have all seen patients with full-fledged TR, and after we put them on Lasix (furosemide, Sanofi), 3 days later they have mild TR. So the same patient with no treatment becomes let’s say a “Dreyfus indication,” and then suddenly in 3 days the patient doesn’t need surgery. At any further stage of his life this patient can experience severe TR again; tricuspid annuloplasty will prevent that from happening.
Moderate TR according to the common definition does not exist. If you look at the reports in echocardiography and if you ask any cardiologist, everything between no TR and severe TR is considered moderate. We have proposed a new staging system for evaluating FTR that uses three factors: TR severity; annular dilatation; and extent of tethering, or mode of leaflet coaptation (J Am Coll Cardiol. 2015;65:2331-6).
Dr. Dreyfus is director of the medical and surgical team at the Cardiothoracic Centre of Monaco in Monte Carlo and professor of cardiothoracic surgery at Paris V University and the Imperial College of London. He disclosed receiving speaker fees from Edwards Lifesciences, LivaNova, and Medtronic.
No, few FTR patients at risk after MVR (Hartzell Schaff, MD)
Echocardiography can provide a great deal of information about when concomitant repair for TR is indicated during MVR. We’ve found that if the patient has no right-sided signs, has normal right atrial pressure, and has mild or mild/moderate TR at the time of repair to the journey mitral valve, the chance of him returning for a tricuspid valve procedure is near zero.
A few patients do return after MVR. They develop atrial fibrillation and may need a pacemaker, but we see very few patients return for tricuspid surgery.
It’s fair to say we cannot demonstrate any benefit of correcting functional mitral regurgitation. Why would we think there’s a benefit of correcting FTR? We can say we’re going to look at grade of TR down the road, or we could look at some other endpoint, but think about it this way: If we cannot prove that correcting functional mitral regurgitation is helpful, why is correcting FTR going to help?
Dr. Schaff is a cardiothoracic surgeon at Mayo Clinic Foundation, Rochester, Minn. He reported no financial relationships.
Yes, functional TR is worth repairing (David H. Adams, MD)
Functional tricuspid regurgitation is a common finding in patients undergoing degenerative mitral valve repair. Severe tricuspid regurgitation is unusual, and clearly there is little debate on the merits of concomitant tricuspid repair for these patients. Moderate tricuspid regurgitation is identified preoperatively in around 15% of patients undergoing degenerative mitral repair (J Thorac Cardiovasc Surg. 2011;142:608-13), and concomitant tricuspid repair in these patients is certainly supported by both the American and European guidelines (J Am Coll Cardiol. 2017. doi: 10.1016/j.jacc.2017.03.011; Eur Heart J. 2012;33:2451-96).
What experience and evidence has led us to a more aggressive approach? One of the most important influences on our early adoption of tricuspid repair at the time of mitral surgery was linked to observations that tricuspid regurgitation (TR) sometimes progressed after isolated mitral valve repair (MVR), with some patients developing moderate or worse insufficiency. Certainly, the impact of significant tricuspid regurgitation on the quality and length of patients’ lives and the challenges of reoperation for isolated tricuspid regurgitation are well known to all surgeons.
Consequently, the importance of treating significant annular dilatation, even without significant tricuspid regurgitation, is supported by the guidelines. Our own experience with an aggressive approach to functional tricuspid regurgitation (FTR) at the time of mitral surgery put an exclamation point on this (J Am Coll Cardiol. 2015;65:1931-8). We found that concomitant tricuspid repair in patients who were worse off before surgery with more TR and higher rates of atrial fibrillation and right-sided dysfunction, actually did better during 5 years of follow-up than the isolated mitral repair patients who started with completely normal tricuspid valve anatomy and ventricular function.
Benign neglect is always an option (J Thorac Cardiovasc Surg. 2017;154:125-6), but we agree with Roberto Dion, MD – despite our friends’ opinions in Toronto and Rochester – we would much prefer to have minimal TR and a normal sized tricuspid valve after MVR. Ask yourself: would you rather have no TR and a normal sized tricuspid valve after you undergo a mitral operation, or a very dilated annulus and perhaps moderate FTR? I am pretty sure I know the answer, but if you are not sure, read our paper.
Dr. Adams is cardiac surgeon-in-chief, Mount Sinai Health System, and Marie-Josée and Henry R. Kravis Professor and Chairman, department of cardiovascular surgery, Icahn School of Medicine at Mount Sinai and The Mount Sinai Hospital, and president-elect of the American Association for Thoracic Surgery. He disclosed he is the national co-principal investigator for the Medtronic NeoChord trial, and receives royalties from Medtronic and Edwards Lifesciences. The Icahn School of Medicine at Mount Sinai receives royalty payments from Edwards Lifesciences and Medtronic for intellectual property related to Dr. Adams’ involvement in the development of 2 mitral valve repair rings and 1 tricuspid valve repair ring.
No, a patient with FTR does not necessarily need repair (Tirone David, MD)
In our clinic, a patient who undergoes MVR and has FTR generally goes home without an annuloplasty. We now have 12 years or more of follow-up in these patients, and they do not develop TR if their MVR is competent. We have reported that preoperative TR in patients who had MVR is associated with mitral valve disease and often improves after the operation (J Thorac Cardiovasc Surg. 2017;154:110-22). New postoperative TR is uncommon.
Ninety percent of my mitral valve repair patients today have no symptoms. Of those patients, a small proportion have moderate TR.
Dr. David is a professor of surgery at Toronto General Hospital. He reported no financial relationships.
Yes, but repair of FTR requires caution (Gilles Dreyfus, MD)
The controversy surrounding the legitimacy of concomitant tricuspid annuloplasty for functional TR during MVR begs for a clinical trial, but before we can conduct a clinical trial, we must define the primary and secondary endpoints. We’ve seen recent prospective, randomized trials that have reported faulty conclusions because the primary endpoints were wrong.
We need a strong debate to agree on those endpoints. Mortality as an endpoint will probably take a very long time to arrive at.
We’re mixing up many different factors. We’re mixing up TR grading, and we know that grading is unreliable. We have all seen patients with full-fledged TR, and after we put them on Lasix (furosemide, Sanofi), 3 days later they have mild TR. So the same patient with no treatment becomes let’s say a “Dreyfus indication,” and then suddenly in 3 days the patient doesn’t need surgery. At any further stage of his life this patient can experience severe TR again; tricuspid annuloplasty will prevent that from happening.
Moderate TR according to the common definition does not exist. If you look at the reports in echocardiography and if you ask any cardiologist, everything between no TR and severe TR is considered moderate. We have proposed a new staging system for evaluating FTR that uses three factors: TR severity; annular dilatation; and extent of tethering, or mode of leaflet coaptation (J Am Coll Cardiol. 2015;65:2331-6).
Dr. Dreyfus is director of the medical and surgical team at the Cardiothoracic Centre of Monaco in Monte Carlo and professor of cardiothoracic surgery at Paris V University and the Imperial College of London. He disclosed receiving speaker fees from Edwards Lifesciences, LivaNova, and Medtronic.
No, few FTR patients at risk after MVR (Hartzell Schaff, MD)
Echocardiography can provide a great deal of information about when concomitant repair for TR is indicated during MVR. We’ve found that if the patient has no right-sided signs, has normal right atrial pressure, and has mild or mild/moderate TR at the time of repair to the journey mitral valve, the chance of him returning for a tricuspid valve procedure is near zero.
A few patients do return after MVR. They develop atrial fibrillation and may need a pacemaker, but we see very few patients return for tricuspid surgery.
It’s fair to say we cannot demonstrate any benefit of correcting functional mitral regurgitation. Why would we think there’s a benefit of correcting FTR? We can say we’re going to look at grade of TR down the road, or we could look at some other endpoint, but think about it this way: If we cannot prove that correcting functional mitral regurgitation is helpful, why is correcting FTR going to help?
Dr. Schaff is a cardiothoracic surgeon at Mayo Clinic Foundation, Rochester, Minn. He reported no financial relationships.
AT THE AATS MITRAL CONCLAVE 2017
Massive blood transfusions increase risk with CRS/HIPEC
Massive allogenic blood transfusion during cytoreductive surgery with hyperthermic intraperitoneal chemotherapy (CRS/HIPEC) increased the risk of major complications and reduced overall survival in a review of 936 cases at St. George Hospital near Sydney, Australia.
CRS/HIPEC is a long, complex procedure for peritoneal carcinomatosis, pseudomyxoma peritonei, peritoneal mesothelioma, and other abdominal cancers. The abdomen is opened, the cancer is debulked as much as possible, and the cavity is filled with heated chemotherapy drugs. Because CRS/HIPEC often requires multivisceral resection and dissection in multiple abdominal regions, up to 77% of patients require intraoperative transfusions, and up to 37% require massive allogenic blood transfusions (MABT) with five or more units.
Blood transfusions are known to be associated with poorer cancer surgery outcomes, but their effect in CRS/HIPEC hasn’t been much studied, which is “surprising given the extent to which blood products are used in” the procedure, said investigators led by Akshat Saxena, MD, a surgeon at St. George Hospital (J Gastrointest Surg. 2017 May 30. doi: 10.1007/s11605-017-3444-8).
Based on their findings, the researchers concluded that “there is a real need to evaluate new strategies to reduce the rate of MABT during CRS/HIPEC.”
The procedures in the study were performed from 1996 to 2016. The in-hospital mortality rate was 0.3% in patients who did not have MABT but 4.4% among the 337 patients (36%) who did. Even after adjusting for confounders on multivariate analysis, including the fact that MABT patients had more extensive disease and longer surgeries, MABT significantly increased the risk of in-hospital mortality (relative risk, 7.72; P = .021). In patients requiring MABT had a 5-year survival of 5%. In patients not requiring MABT, 5-year survival was at 36%. The difference remained significant on multivariate analysis.
MABT patients also had twice the risk of life-threatening complications and complications requiring surgical, endoscopic, or radiological intervention (62% versus 30%; RR, 2.05; P less than .001). MABT patients were more likely to stay in the ICU for 4 or more days and in the hospital for 28 or more days.
Worse overall survival with MABT was driven at least in part by patients who had CRS/HIPEC for colorectal cancer peritoneal carcinomatosis and pseudomyxoma peritonei. MABT did not seem to contribute to lower survival in patients who had the procedure for appendiceal or ovarian cancer. “It seems that the impact of long-term immunomodulation induced by blood transfusion” – the suspected mechanism through which transfusions cause problems – “varies according to the disease subtype. This warrants further investigation,” the investigators said.
Several strategies have been tried to reduce the need for transfusions during CRS/HIPEC. The study team previously reported that preemptive clotting factor replacement helps. Others have had success with preemptive tranexamic acid and cryoprecipitate to address low serum fibrinogen levels during CRS/HIPEC. “Further evaluation of both these strategies is warranted,” the researchers said.
Funding source and disclosure information were not included in the study report.
Massive allogenic blood transfusion during cytoreductive surgery with hyperthermic intraperitoneal chemotherapy (CRS/HIPEC) increased the risk of major complications and reduced overall survival in a review of 936 cases at St. George Hospital near Sydney, Australia.
CRS/HIPEC is a long, complex procedure for peritoneal carcinomatosis, pseudomyxoma peritonei, peritoneal mesothelioma, and other abdominal cancers. The abdomen is opened, the cancer is debulked as much as possible, and the cavity is filled with heated chemotherapy drugs. Because CRS/HIPEC often requires multivisceral resection and dissection in multiple abdominal regions, up to 77% of patients require intraoperative transfusions, and up to 37% require massive allogenic blood transfusions (MABT) with five or more units.
Blood transfusions are known to be associated with poorer cancer surgery outcomes, but their effect in CRS/HIPEC hasn’t been much studied, which is “surprising given the extent to which blood products are used in” the procedure, said investigators led by Akshat Saxena, MD, a surgeon at St. George Hospital (J Gastrointest Surg. 2017 May 30. doi: 10.1007/s11605-017-3444-8).
Based on their findings, the researchers concluded that “there is a real need to evaluate new strategies to reduce the rate of MABT during CRS/HIPEC.”
The procedures in the study were performed from 1996 to 2016. The in-hospital mortality rate was 0.3% in patients who did not have MABT but 4.4% among the 337 patients (36%) who did. Even after adjusting for confounders on multivariate analysis, including the fact that MABT patients had more extensive disease and longer surgeries, MABT significantly increased the risk of in-hospital mortality (relative risk, 7.72; P = .021). In patients requiring MABT had a 5-year survival of 5%. In patients not requiring MABT, 5-year survival was at 36%. The difference remained significant on multivariate analysis.
MABT patients also had twice the risk of life-threatening complications and complications requiring surgical, endoscopic, or radiological intervention (62% versus 30%; RR, 2.05; P less than .001). MABT patients were more likely to stay in the ICU for 4 or more days and in the hospital for 28 or more days.
Worse overall survival with MABT was driven at least in part by patients who had CRS/HIPEC for colorectal cancer peritoneal carcinomatosis and pseudomyxoma peritonei. MABT did not seem to contribute to lower survival in patients who had the procedure for appendiceal or ovarian cancer. “It seems that the impact of long-term immunomodulation induced by blood transfusion” – the suspected mechanism through which transfusions cause problems – “varies according to the disease subtype. This warrants further investigation,” the investigators said.
Several strategies have been tried to reduce the need for transfusions during CRS/HIPEC. The study team previously reported that preemptive clotting factor replacement helps. Others have had success with preemptive tranexamic acid and cryoprecipitate to address low serum fibrinogen levels during CRS/HIPEC. “Further evaluation of both these strategies is warranted,” the researchers said.
Funding source and disclosure information were not included in the study report.
Massive allogenic blood transfusion during cytoreductive surgery with hyperthermic intraperitoneal chemotherapy (CRS/HIPEC) increased the risk of major complications and reduced overall survival in a review of 936 cases at St. George Hospital near Sydney, Australia.
CRS/HIPEC is a long, complex procedure for peritoneal carcinomatosis, pseudomyxoma peritonei, peritoneal mesothelioma, and other abdominal cancers. The abdomen is opened, the cancer is debulked as much as possible, and the cavity is filled with heated chemotherapy drugs. Because CRS/HIPEC often requires multivisceral resection and dissection in multiple abdominal regions, up to 77% of patients require intraoperative transfusions, and up to 37% require massive allogenic blood transfusions (MABT) with five or more units.
Blood transfusions are known to be associated with poorer cancer surgery outcomes, but their effect in CRS/HIPEC hasn’t been much studied, which is “surprising given the extent to which blood products are used in” the procedure, said investigators led by Akshat Saxena, MD, a surgeon at St. George Hospital (J Gastrointest Surg. 2017 May 30. doi: 10.1007/s11605-017-3444-8).
Based on their findings, the researchers concluded that “there is a real need to evaluate new strategies to reduce the rate of MABT during CRS/HIPEC.”
The procedures in the study were performed from 1996 to 2016. The in-hospital mortality rate was 0.3% in patients who did not have MABT but 4.4% among the 337 patients (36%) who did. Even after adjusting for confounders on multivariate analysis, including the fact that MABT patients had more extensive disease and longer surgeries, MABT significantly increased the risk of in-hospital mortality (relative risk, 7.72; P = .021). In patients requiring MABT had a 5-year survival of 5%. In patients not requiring MABT, 5-year survival was at 36%. The difference remained significant on multivariate analysis.
MABT patients also had twice the risk of life-threatening complications and complications requiring surgical, endoscopic, or radiological intervention (62% versus 30%; RR, 2.05; P less than .001). MABT patients were more likely to stay in the ICU for 4 or more days and in the hospital for 28 or more days.
Worse overall survival with MABT was driven at least in part by patients who had CRS/HIPEC for colorectal cancer peritoneal carcinomatosis and pseudomyxoma peritonei. MABT did not seem to contribute to lower survival in patients who had the procedure for appendiceal or ovarian cancer. “It seems that the impact of long-term immunomodulation induced by blood transfusion” – the suspected mechanism through which transfusions cause problems – “varies according to the disease subtype. This warrants further investigation,” the investigators said.
Several strategies have been tried to reduce the need for transfusions during CRS/HIPEC. The study team previously reported that preemptive clotting factor replacement helps. Others have had success with preemptive tranexamic acid and cryoprecipitate to address low serum fibrinogen levels during CRS/HIPEC. “Further evaluation of both these strategies is warranted,” the researchers said.
Funding source and disclosure information were not included in the study report.
FROM THE JOURNAL OF GASTROINTESTINAL SURGERY
Key clinical point:
Major finding: Even after adjusting for confounders, MABT significantly increased the risk of in-hospital mortality (RR, 7.72; P = .021).
Data source: A single institution review of 936 cases.
Disclosures: Funding source and disclosure information were not included in the study report.