User login
Leukocytoclastic vasculitis incidence underestimated
ASHEVILLE, N.C. – The incidence of leukocytoclastic vasculitis in the general population is 1.5 to 3 times higher than previously reported, based on data from a large, population-based study.
Based on these findings, leukocytoclastic vasculitis (LCV) is “more common than cutaneous lupus,” said Dr. David A. Wetter of the Mayo Clinic, Rochester, Minn.
Dr. Wetter and his associates reviewed data from all residents of Olmstead County, Minn., with a biopsy-proven diagnosis of LCV from January 1, 1996, through December 31, 2010.
A total of 84 patients were identified, for an overall incidence of 4.5 per 100,000 person-years. The mean age was 48 years, and approximately half were women.
The researchers divided the patients into five subtypes: cutaneous small vessel vasculitis (CSVV), IgA vasculitis, urticarial vasculitis, cryoglobulinemic vasculitis, and antineutrophil cytoplasmic antibody–associated vasculitis.
Systemic involvement occurred in 46% of the patients, with the kidneys involved in 44% of these cases. Recurrent disease was observed in 30% of the 80 patients for whom follow-up data were available.
In addition, LCV was idiopathic in 76% of CSVV patients, significantly higher than the 45% rate reported in the current textbooks, Dr. Wetter noted. Idiopathic LCV also occurred in 96% of patients with IgA vasculitis.
Overall survival was significantly decreased in the LCV patients compared to the general Minnesota population, but the reasons for this difference remain unclear and are worthy of additional study, Dr. Wetter noted. However, survival was not significantly decreased in the subset of patients with IgA vasculitis, despite the high rate of systemic involvement, he added.
Although the study was limited by its retrospective nature and relatively homogeneous white population, it is the first known population-based study of histopathologically-defined LCV, and the data support the evaluation of all patients with LCV “for extracutaneous disease and systemic etiologies for their disease,” Dr. Wetter said.
Dr. Wetter presented the study findings at the annual meeting of the Noah Worcester Dermatological Society. The data also were published in the Mayo Clinic Proceedings in 2014 (Mayo Clin. Proc. 2014;89:1515-24).
Dr. Wetter had no financial conflicts to disclose.
ASHEVILLE, N.C. – The incidence of leukocytoclastic vasculitis in the general population is 1.5 to 3 times higher than previously reported, based on data from a large, population-based study.
Based on these findings, leukocytoclastic vasculitis (LCV) is “more common than cutaneous lupus,” said Dr. David A. Wetter of the Mayo Clinic, Rochester, Minn.
Dr. Wetter and his associates reviewed data from all residents of Olmstead County, Minn., with a biopsy-proven diagnosis of LCV from January 1, 1996, through December 31, 2010.
A total of 84 patients were identified, for an overall incidence of 4.5 per 100,000 person-years. The mean age was 48 years, and approximately half were women.
The researchers divided the patients into five subtypes: cutaneous small vessel vasculitis (CSVV), IgA vasculitis, urticarial vasculitis, cryoglobulinemic vasculitis, and antineutrophil cytoplasmic antibody–associated vasculitis.
Systemic involvement occurred in 46% of the patients, with the kidneys involved in 44% of these cases. Recurrent disease was observed in 30% of the 80 patients for whom follow-up data were available.
In addition, LCV was idiopathic in 76% of CSVV patients, significantly higher than the 45% rate reported in the current textbooks, Dr. Wetter noted. Idiopathic LCV also occurred in 96% of patients with IgA vasculitis.
Overall survival was significantly decreased in the LCV patients compared to the general Minnesota population, but the reasons for this difference remain unclear and are worthy of additional study, Dr. Wetter noted. However, survival was not significantly decreased in the subset of patients with IgA vasculitis, despite the high rate of systemic involvement, he added.
Although the study was limited by its retrospective nature and relatively homogeneous white population, it is the first known population-based study of histopathologically-defined LCV, and the data support the evaluation of all patients with LCV “for extracutaneous disease and systemic etiologies for their disease,” Dr. Wetter said.
Dr. Wetter presented the study findings at the annual meeting of the Noah Worcester Dermatological Society. The data also were published in the Mayo Clinic Proceedings in 2014 (Mayo Clin. Proc. 2014;89:1515-24).
Dr. Wetter had no financial conflicts to disclose.
ASHEVILLE, N.C. – The incidence of leukocytoclastic vasculitis in the general population is 1.5 to 3 times higher than previously reported, based on data from a large, population-based study.
Based on these findings, leukocytoclastic vasculitis (LCV) is “more common than cutaneous lupus,” said Dr. David A. Wetter of the Mayo Clinic, Rochester, Minn.
Dr. Wetter and his associates reviewed data from all residents of Olmstead County, Minn., with a biopsy-proven diagnosis of LCV from January 1, 1996, through December 31, 2010.
A total of 84 patients were identified, for an overall incidence of 4.5 per 100,000 person-years. The mean age was 48 years, and approximately half were women.
The researchers divided the patients into five subtypes: cutaneous small vessel vasculitis (CSVV), IgA vasculitis, urticarial vasculitis, cryoglobulinemic vasculitis, and antineutrophil cytoplasmic antibody–associated vasculitis.
Systemic involvement occurred in 46% of the patients, with the kidneys involved in 44% of these cases. Recurrent disease was observed in 30% of the 80 patients for whom follow-up data were available.
In addition, LCV was idiopathic in 76% of CSVV patients, significantly higher than the 45% rate reported in the current textbooks, Dr. Wetter noted. Idiopathic LCV also occurred in 96% of patients with IgA vasculitis.
Overall survival was significantly decreased in the LCV patients compared to the general Minnesota population, but the reasons for this difference remain unclear and are worthy of additional study, Dr. Wetter noted. However, survival was not significantly decreased in the subset of patients with IgA vasculitis, despite the high rate of systemic involvement, he added.
Although the study was limited by its retrospective nature and relatively homogeneous white population, it is the first known population-based study of histopathologically-defined LCV, and the data support the evaluation of all patients with LCV “for extracutaneous disease and systemic etiologies for their disease,” Dr. Wetter said.
Dr. Wetter presented the study findings at the annual meeting of the Noah Worcester Dermatological Society. The data also were published in the Mayo Clinic Proceedings in 2014 (Mayo Clin. Proc. 2014;89:1515-24).
Dr. Wetter had no financial conflicts to disclose.
AT NOAH 57
Key clinical point: The data support the evaluation of all patients with LCV for extracutaneous disease and systemic etiologies.
Major finding: The incidence of leukocytoclastic vasculitis in the general population is 1.5 to 3 times higher than previously reported.
Data source: A population-based study of all residents of Olmstead County, Minn., with a biopsy-proven diagnosis of LCV from January 1, 1996, through December 31, 2010.
Disclosures: Dr. Wetter had no financial conflicts to disclose.
VIDEO: Larger lentigo maligna lesions increase risk
ASHEVILLE, N.C. – What are the risk factors for invasive melanoma in patients with lentigo maligna? Size, for one thing, according to Dr. Suzanne M. Olbricht.
In an interview at the annual meeting of the Noah Worcester Dermatological Society, Dr. Olbricht of the Lahey Hospital and Medical Center in Burlington, Mass., reviewed evidence suggesting that the recurrence rate is highest for large lesions. “This is important information that helps us think about the treatments we can use,” she said.
Dr. Olbricht had no financial conflicts to disclose.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ASHEVILLE, N.C. – What are the risk factors for invasive melanoma in patients with lentigo maligna? Size, for one thing, according to Dr. Suzanne M. Olbricht.
In an interview at the annual meeting of the Noah Worcester Dermatological Society, Dr. Olbricht of the Lahey Hospital and Medical Center in Burlington, Mass., reviewed evidence suggesting that the recurrence rate is highest for large lesions. “This is important information that helps us think about the treatments we can use,” she said.
Dr. Olbricht had no financial conflicts to disclose.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ASHEVILLE, N.C. – What are the risk factors for invasive melanoma in patients with lentigo maligna? Size, for one thing, according to Dr. Suzanne M. Olbricht.
In an interview at the annual meeting of the Noah Worcester Dermatological Society, Dr. Olbricht of the Lahey Hospital and Medical Center in Burlington, Mass., reviewed evidence suggesting that the recurrence rate is highest for large lesions. “This is important information that helps us think about the treatments we can use,” she said.
Dr. Olbricht had no financial conflicts to disclose.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT NOAH 57
Contamination prompts voluntary injectables recall
Mylan N.V. has announced a voluntary recall of specific lots of injectable products for conditions including rheumatoid arthritis, severe psoriasis, lung cancer, breast cancer, ovarian cancer, and acute nonlymphocytic leukemia because foreign particles were observed during testing of retention samples, according to a statement issued by the company on April 23.
The specific lot numbers can be found on the company’s website.
Affected products include methotrexate injection, USP 25 mg/mL, which is indicated for adult rheumatoid arthritis, severe psoriasis, and some neoplastic diseases. According to Mylan, the contaminated lot was distributed in the United States between Jan. 16, 2014, and March 25, 2014.
Other recalled injectable products include gemcitabine, USP 200 mg, an intravenous product for ovarian cancer, breast cancer, non–small cell lung cancer, and pancreatic cancer; carboplatin 10 mg/mL, indicated for advanced ovarian cancer; and cytarabine, indicated for remission induction in acute nonlymphocytic leukemia in adults and children. The lots for these products were distributed in the United States in various periods of several months during 2014.
The recall is being conducted with the knowledge of the Food and Drug Administration, and any adverse events or quality problems should be reported to the FDA’s MedWatch adverse event reporting program.
Mylan N.V. has announced a voluntary recall of specific lots of injectable products for conditions including rheumatoid arthritis, severe psoriasis, lung cancer, breast cancer, ovarian cancer, and acute nonlymphocytic leukemia because foreign particles were observed during testing of retention samples, according to a statement issued by the company on April 23.
The specific lot numbers can be found on the company’s website.
Affected products include methotrexate injection, USP 25 mg/mL, which is indicated for adult rheumatoid arthritis, severe psoriasis, and some neoplastic diseases. According to Mylan, the contaminated lot was distributed in the United States between Jan. 16, 2014, and March 25, 2014.
Other recalled injectable products include gemcitabine, USP 200 mg, an intravenous product for ovarian cancer, breast cancer, non–small cell lung cancer, and pancreatic cancer; carboplatin 10 mg/mL, indicated for advanced ovarian cancer; and cytarabine, indicated for remission induction in acute nonlymphocytic leukemia in adults and children. The lots for these products were distributed in the United States in various periods of several months during 2014.
The recall is being conducted with the knowledge of the Food and Drug Administration, and any adverse events or quality problems should be reported to the FDA’s MedWatch adverse event reporting program.
Mylan N.V. has announced a voluntary recall of specific lots of injectable products for conditions including rheumatoid arthritis, severe psoriasis, lung cancer, breast cancer, ovarian cancer, and acute nonlymphocytic leukemia because foreign particles were observed during testing of retention samples, according to a statement issued by the company on April 23.
The specific lot numbers can be found on the company’s website.
Affected products include methotrexate injection, USP 25 mg/mL, which is indicated for adult rheumatoid arthritis, severe psoriasis, and some neoplastic diseases. According to Mylan, the contaminated lot was distributed in the United States between Jan. 16, 2014, and March 25, 2014.
Other recalled injectable products include gemcitabine, USP 200 mg, an intravenous product for ovarian cancer, breast cancer, non–small cell lung cancer, and pancreatic cancer; carboplatin 10 mg/mL, indicated for advanced ovarian cancer; and cytarabine, indicated for remission induction in acute nonlymphocytic leukemia in adults and children. The lots for these products were distributed in the United States in various periods of several months during 2014.
The recall is being conducted with the knowledge of the Food and Drug Administration, and any adverse events or quality problems should be reported to the FDA’s MedWatch adverse event reporting program.
VIDEO: Nanodermatology Society roundtable tackles tough questions on photoprotection
SAN FRANCISCO – What role does nanotechnology play in photoprotection? How is the current regulatory climate helping and hurting innovation and product development? What are the real and perceived safety issues? These and other questions were addressed in a roundtable discussion hosted by the Nanodermatology Society at the annual meeting of the American Academy of Dermatology. Participants included dermatology practitioners and researchers, along with representatives of industry and the media.
“We have good data on a lot of the filters that are out there,” emphasized Dr. Adnan Nasir, president of the Nanodermatology Society and moderator of the roundtable.
Given the epidemic of skin cancer in the United States, “I hope that we can target our message as a public health message” and increase the confidence of physicians in discussing photoprotection and the confidence of patients in their doctors’ opinions on this important topic, Dr. Nasir said.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN FRANCISCO – What role does nanotechnology play in photoprotection? How is the current regulatory climate helping and hurting innovation and product development? What are the real and perceived safety issues? These and other questions were addressed in a roundtable discussion hosted by the Nanodermatology Society at the annual meeting of the American Academy of Dermatology. Participants included dermatology practitioners and researchers, along with representatives of industry and the media.
“We have good data on a lot of the filters that are out there,” emphasized Dr. Adnan Nasir, president of the Nanodermatology Society and moderator of the roundtable.
Given the epidemic of skin cancer in the United States, “I hope that we can target our message as a public health message” and increase the confidence of physicians in discussing photoprotection and the confidence of patients in their doctors’ opinions on this important topic, Dr. Nasir said.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN FRANCISCO – What role does nanotechnology play in photoprotection? How is the current regulatory climate helping and hurting innovation and product development? What are the real and perceived safety issues? These and other questions were addressed in a roundtable discussion hosted by the Nanodermatology Society at the annual meeting of the American Academy of Dermatology. Participants included dermatology practitioners and researchers, along with representatives of industry and the media.
“We have good data on a lot of the filters that are out there,” emphasized Dr. Adnan Nasir, president of the Nanodermatology Society and moderator of the roundtable.
Given the epidemic of skin cancer in the United States, “I hope that we can target our message as a public health message” and increase the confidence of physicians in discussing photoprotection and the confidence of patients in their doctors’ opinions on this important topic, Dr. Nasir said.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE AAD ANNUAL MEETING
Contact allergen of 2015: Formaldehyde
SAN FRANCISCO – Formaldehyde has been named the American Contact Dermatitis Society Contact Allergen of the Year for 2015, Dr. David E. Cohen said in the hot topics session at the annual meeting of the American Academy of Dermatology.
Formaldehyde has been a factor in contact dermatitis for a long time, and exposure to it is widespread, said Dr. Cohen of New York University.
However, recent studies have reassessed the clinical relevance of formaldehyde allergy, and a patch test concentration of 2.0% has been recommended by the European Environmental Contact Dermatitis Research group, according to a report published in the journal Dermatitis (Dermatitis 2015;26:3-6).
“Without causing any more irritant reactions, the patch test concentration of 2.0% detects twice as many contact allergies and enables the diagnosis of formaldehyde-allergic patients who otherwise would have been missed,” the authors wrote.
SAN FRANCISCO – Formaldehyde has been named the American Contact Dermatitis Society Contact Allergen of the Year for 2015, Dr. David E. Cohen said in the hot topics session at the annual meeting of the American Academy of Dermatology.
Formaldehyde has been a factor in contact dermatitis for a long time, and exposure to it is widespread, said Dr. Cohen of New York University.
However, recent studies have reassessed the clinical relevance of formaldehyde allergy, and a patch test concentration of 2.0% has been recommended by the European Environmental Contact Dermatitis Research group, according to a report published in the journal Dermatitis (Dermatitis 2015;26:3-6).
“Without causing any more irritant reactions, the patch test concentration of 2.0% detects twice as many contact allergies and enables the diagnosis of formaldehyde-allergic patients who otherwise would have been missed,” the authors wrote.
SAN FRANCISCO – Formaldehyde has been named the American Contact Dermatitis Society Contact Allergen of the Year for 2015, Dr. David E. Cohen said in the hot topics session at the annual meeting of the American Academy of Dermatology.
Formaldehyde has been a factor in contact dermatitis for a long time, and exposure to it is widespread, said Dr. Cohen of New York University.
However, recent studies have reassessed the clinical relevance of formaldehyde allergy, and a patch test concentration of 2.0% has been recommended by the European Environmental Contact Dermatitis Research group, according to a report published in the journal Dermatitis (Dermatitis 2015;26:3-6).
“Without causing any more irritant reactions, the patch test concentration of 2.0% detects twice as many contact allergies and enables the diagnosis of formaldehyde-allergic patients who otherwise would have been missed,” the authors wrote.
AT THE AAD ANNUAL MEETING
Customizable features enhance the latest fillers
Be prepared for more dermal fillers featuring a mix of low- and high-molecular-weight hyaluronic acid, as well as options for customization, said Dr. Nowell Solish.
Such a combination results in a highly crosslinked product that promotes greater duration with less swelling of the product and improved spreadability, he said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
“There is a role for dermal fillers with these characteristics,” said Dr. Solish of the University of Toronto. He compared the uses of several combination products, including Voluma, Volbella, and Volift.
Dr. Solish’s take on Voluma: “Great for volume,” particularly for the chest and midface, with a low and manageable rate of adverse events.
By contrast, Volbella, which is not yet approved for use in the United States, is a fast-flowing filler suited for lip hydration and perioral lines, rather than volume, he said. In a 2012 study, 48% of 58 adult patients showed an improvement of 1 point or higher on the Lip Fullness Scale at 1 year, based on physician assessment, he said (Clin. Cosmet. Investig. Dermatol. 2012;5;167-72).
Volift, another combination high/low-molecular-weight hyaluronic acid product, is “likely last to get approved” in the United States, Dr. Solish said. The product, however, is “great for marionette lines and smile lines,” as well as for improving the appearance of mild to moderate lines around the face.
Emervel is an example of a product in which the varying particle size allows for customization. The hyaluronic acid concentration of Emervel is consistent, but the product is available in three particle sizes for three depths of injection and varying lifting capacity, he explained.
Finally, the use of a filler containing polymethylmethacrylate (PMMA) microspheres was associated with significant improvements in atrophic facial acne scars, Dr. Solish said, citing a recent randomized controlled trial of 147 subjects (J. Am. Acad. Dermatol. 2014;71:77-83). The microspheres are designed to be consistent is size (30-50 mcm). Treatment-related adverse events were generally mild and transient.
The calcium microsphere filler Radiesse with lidocaine was recently approved by the Food and Drug Administration and should be available soon, he added.
Dr. Solish disclosed serving as an investigator and consultant for Allergan, Medicis, Revance Therapeutics, Marz, and Indeed. SDEF and this news organization are owned by the same parent company.
Be prepared for more dermal fillers featuring a mix of low- and high-molecular-weight hyaluronic acid, as well as options for customization, said Dr. Nowell Solish.
Such a combination results in a highly crosslinked product that promotes greater duration with less swelling of the product and improved spreadability, he said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
“There is a role for dermal fillers with these characteristics,” said Dr. Solish of the University of Toronto. He compared the uses of several combination products, including Voluma, Volbella, and Volift.
Dr. Solish’s take on Voluma: “Great for volume,” particularly for the chest and midface, with a low and manageable rate of adverse events.
By contrast, Volbella, which is not yet approved for use in the United States, is a fast-flowing filler suited for lip hydration and perioral lines, rather than volume, he said. In a 2012 study, 48% of 58 adult patients showed an improvement of 1 point or higher on the Lip Fullness Scale at 1 year, based on physician assessment, he said (Clin. Cosmet. Investig. Dermatol. 2012;5;167-72).
Volift, another combination high/low-molecular-weight hyaluronic acid product, is “likely last to get approved” in the United States, Dr. Solish said. The product, however, is “great for marionette lines and smile lines,” as well as for improving the appearance of mild to moderate lines around the face.
Emervel is an example of a product in which the varying particle size allows for customization. The hyaluronic acid concentration of Emervel is consistent, but the product is available in three particle sizes for three depths of injection and varying lifting capacity, he explained.
Finally, the use of a filler containing polymethylmethacrylate (PMMA) microspheres was associated with significant improvements in atrophic facial acne scars, Dr. Solish said, citing a recent randomized controlled trial of 147 subjects (J. Am. Acad. Dermatol. 2014;71:77-83). The microspheres are designed to be consistent is size (30-50 mcm). Treatment-related adverse events were generally mild and transient.
The calcium microsphere filler Radiesse with lidocaine was recently approved by the Food and Drug Administration and should be available soon, he added.
Dr. Solish disclosed serving as an investigator and consultant for Allergan, Medicis, Revance Therapeutics, Marz, and Indeed. SDEF and this news organization are owned by the same parent company.
Be prepared for more dermal fillers featuring a mix of low- and high-molecular-weight hyaluronic acid, as well as options for customization, said Dr. Nowell Solish.
Such a combination results in a highly crosslinked product that promotes greater duration with less swelling of the product and improved spreadability, he said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
“There is a role for dermal fillers with these characteristics,” said Dr. Solish of the University of Toronto. He compared the uses of several combination products, including Voluma, Volbella, and Volift.
Dr. Solish’s take on Voluma: “Great for volume,” particularly for the chest and midface, with a low and manageable rate of adverse events.
By contrast, Volbella, which is not yet approved for use in the United States, is a fast-flowing filler suited for lip hydration and perioral lines, rather than volume, he said. In a 2012 study, 48% of 58 adult patients showed an improvement of 1 point or higher on the Lip Fullness Scale at 1 year, based on physician assessment, he said (Clin. Cosmet. Investig. Dermatol. 2012;5;167-72).
Volift, another combination high/low-molecular-weight hyaluronic acid product, is “likely last to get approved” in the United States, Dr. Solish said. The product, however, is “great for marionette lines and smile lines,” as well as for improving the appearance of mild to moderate lines around the face.
Emervel is an example of a product in which the varying particle size allows for customization. The hyaluronic acid concentration of Emervel is consistent, but the product is available in three particle sizes for three depths of injection and varying lifting capacity, he explained.
Finally, the use of a filler containing polymethylmethacrylate (PMMA) microspheres was associated with significant improvements in atrophic facial acne scars, Dr. Solish said, citing a recent randomized controlled trial of 147 subjects (J. Am. Acad. Dermatol. 2014;71:77-83). The microspheres are designed to be consistent is size (30-50 mcm). Treatment-related adverse events were generally mild and transient.
The calcium microsphere filler Radiesse with lidocaine was recently approved by the Food and Drug Administration and should be available soon, he added.
Dr. Solish disclosed serving as an investigator and consultant for Allergan, Medicis, Revance Therapeutics, Marz, and Indeed. SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Secukinumab Earns FDA Approval for Plaque Psoriasis
The U.S. Food and Drug Administration has approved secukinumab for the treatment of moderate to severe plaque psoriasis in adults.
The injectable interleukin-17A inhibitor is indicated for psoriasis patients who are candidates for systemic therapy and/or phototherapy, according to a press release from the FDA. Novartis Pharmaceuticals will market secukinumab as Cosentyx.
The Jan. 21 approval was based on data from randomized, placebo-controlled clinical trials involving 2,403 patients. The most common side effects reported in the trials included upper respiratory infections and diarrhea.
The biologic is being approved with a medication guide “to inform patients that, because Cosentyx is a medicine that affects the immune system, patients may have a greater risk of getting an infection,” according to the FDA release. Consequently, the FDA advised clinicians to be cautious when prescribing secukinumab to patients with chronic or recurrent infections, and to those with active Crohn’s disease.
The FDA approval followed an advisory committee meeting in October 2014.
The U.S. Food and Drug Administration has approved secukinumab for the treatment of moderate to severe plaque psoriasis in adults.
The injectable interleukin-17A inhibitor is indicated for psoriasis patients who are candidates for systemic therapy and/or phototherapy, according to a press release from the FDA. Novartis Pharmaceuticals will market secukinumab as Cosentyx.
The Jan. 21 approval was based on data from randomized, placebo-controlled clinical trials involving 2,403 patients. The most common side effects reported in the trials included upper respiratory infections and diarrhea.
The biologic is being approved with a medication guide “to inform patients that, because Cosentyx is a medicine that affects the immune system, patients may have a greater risk of getting an infection,” according to the FDA release. Consequently, the FDA advised clinicians to be cautious when prescribing secukinumab to patients with chronic or recurrent infections, and to those with active Crohn’s disease.
The FDA approval followed an advisory committee meeting in October 2014.
The U.S. Food and Drug Administration has approved secukinumab for the treatment of moderate to severe plaque psoriasis in adults.
The injectable interleukin-17A inhibitor is indicated for psoriasis patients who are candidates for systemic therapy and/or phototherapy, according to a press release from the FDA. Novartis Pharmaceuticals will market secukinumab as Cosentyx.
The Jan. 21 approval was based on data from randomized, placebo-controlled clinical trials involving 2,403 patients. The most common side effects reported in the trials included upper respiratory infections and diarrhea.
The biologic is being approved with a medication guide “to inform patients that, because Cosentyx is a medicine that affects the immune system, patients may have a greater risk of getting an infection,” according to the FDA release. Consequently, the FDA advised clinicians to be cautious when prescribing secukinumab to patients with chronic or recurrent infections, and to those with active Crohn’s disease.
The FDA approval followed an advisory committee meeting in October 2014.
Secukinumab earns FDA approval for plaque psoriasis
The U.S. Food and Drug Administration has approved secukinumab for the treatment of moderate to severe plaque psoriasis in adults.
The injectable interleukin-17A inhibitor is indicated for psoriasis patients who are candidates for systemic therapy and/or phototherapy, according to a press release from the FDA. Novartis Pharmaceuticals will market secukinumab as Cosentyx.
The Jan. 21 approval was based on data from randomized, placebo-controlled clinical trials involving 2,403 patients. The most common side effects reported in the trials included upper respiratory infections and diarrhea.
The biologic is being approved with a medication guide “to inform patients that, because Cosentyx is a medicine that affects the immune system, patients may have a greater risk of getting an infection,” according to the FDA release. Consequently, the FDA advised clinicians to be cautious when prescribing secukinumab to patients with chronic or recurrent infections, and to those with active Crohn’s disease.
The FDA approval followed an advisory committee meeting in October 2014.
The U.S. Food and Drug Administration has approved secukinumab for the treatment of moderate to severe plaque psoriasis in adults.
The injectable interleukin-17A inhibitor is indicated for psoriasis patients who are candidates for systemic therapy and/or phototherapy, according to a press release from the FDA. Novartis Pharmaceuticals will market secukinumab as Cosentyx.
The Jan. 21 approval was based on data from randomized, placebo-controlled clinical trials involving 2,403 patients. The most common side effects reported in the trials included upper respiratory infections and diarrhea.
The biologic is being approved with a medication guide “to inform patients that, because Cosentyx is a medicine that affects the immune system, patients may have a greater risk of getting an infection,” according to the FDA release. Consequently, the FDA advised clinicians to be cautious when prescribing secukinumab to patients with chronic or recurrent infections, and to those with active Crohn’s disease.
The FDA approval followed an advisory committee meeting in October 2014.
The U.S. Food and Drug Administration has approved secukinumab for the treatment of moderate to severe plaque psoriasis in adults.
The injectable interleukin-17A inhibitor is indicated for psoriasis patients who are candidates for systemic therapy and/or phototherapy, according to a press release from the FDA. Novartis Pharmaceuticals will market secukinumab as Cosentyx.
The Jan. 21 approval was based on data from randomized, placebo-controlled clinical trials involving 2,403 patients. The most common side effects reported in the trials included upper respiratory infections and diarrhea.
The biologic is being approved with a medication guide “to inform patients that, because Cosentyx is a medicine that affects the immune system, patients may have a greater risk of getting an infection,” according to the FDA release. Consequently, the FDA advised clinicians to be cautious when prescribing secukinumab to patients with chronic or recurrent infections, and to those with active Crohn’s disease.
The FDA approval followed an advisory committee meeting in October 2014.
Consider phototherapy for certain psoriasis patients
LAS VEGAS – Psoriasis patients who are unresponsive to topical therapy, do not have psoriatic arthritis, and can make regular office visits may be candidates for successful treatment with phototherapy.
A patient with type II skin and extensive plaque psoriasis could be treated with a targeted NB-UVB laser, Dr. Kenneth B. Gordon said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
However, it’s important to evaluate each patient individually on phototherapy decisions, rather than relying on published protocols, noted Dr. Gordon of Northwestern University Feinberg School of Medicine in Chicago.
The advantages of the NB-UVB laser for psoriasis include a high rate of response (data show more than 75% of patients achieve PASI 75), the ability to avoid treating uninvolved skin, and potential long-term clearing of lesions, he said.
For someone with extensive plaque psoriasis, Dr. Gordon said he would start with 3 treatments per week, at 200 mJ, and increase the fluence by 25 mJ if necessary. “A good response can be expected in 4-6 weeks,” he said.
As for long-term treatment, “I leave it up to the patient but offer long-term maintenance,” said Dr. Gordon. Long-term efficacy of NB-UVB for plaque psoriasis has not been well studied, and safety data are unknown. “It is unclear whether there is a skin cancer risk,” he said. Reducing the number of exposures for a patient on maintenance therapy may be an option, he added.
Short-term treatment with NB-UVB can be effective in some psoriasis patients, such as in cases of an acute flare of guttate psoriasis, Dr. Gordon noted. Psoralen and UV light therapy (PUVA) is another option, he said.
Phototherapy also has a role in managing psoriasis in conjunction with retinoids, Dr. Gordon said. He cited an example of a 59-year-old woman with palmar psoriasis. For this patient, he said he would recommend starting with acitretin for approximately 1 month, if the patient tolerates it. “If the acitretin works by itself, no need to add phototherapy,” he said. However, if UVB is added, account for the photosensitizing agent, he emphasized. “Decrease the starting dose by 25-50 mJ or decrease the skin type by one,” he said.
Some psoriasis patients will not benefit from phototherapy, particularly those with erythroderma, said Dr. Gordon. “If skin is highly inflamed, phototherapy could induce easy burning, Koebnerization,” or other complications, he noted.
Dr. Gordon disclosed that his department at Northwestern derives income from phototherapy, but he personally does not. He also disclosed receiving research grants from and/or serving as a consultant to multiple companies including AbbVie, Amgen, Celgene, Eli Lilly, and Janssen.
LAS VEGAS – Psoriasis patients who are unresponsive to topical therapy, do not have psoriatic arthritis, and can make regular office visits may be candidates for successful treatment with phototherapy.
A patient with type II skin and extensive plaque psoriasis could be treated with a targeted NB-UVB laser, Dr. Kenneth B. Gordon said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
However, it’s important to evaluate each patient individually on phototherapy decisions, rather than relying on published protocols, noted Dr. Gordon of Northwestern University Feinberg School of Medicine in Chicago.
The advantages of the NB-UVB laser for psoriasis include a high rate of response (data show more than 75% of patients achieve PASI 75), the ability to avoid treating uninvolved skin, and potential long-term clearing of lesions, he said.
For someone with extensive plaque psoriasis, Dr. Gordon said he would start with 3 treatments per week, at 200 mJ, and increase the fluence by 25 mJ if necessary. “A good response can be expected in 4-6 weeks,” he said.
As for long-term treatment, “I leave it up to the patient but offer long-term maintenance,” said Dr. Gordon. Long-term efficacy of NB-UVB for plaque psoriasis has not been well studied, and safety data are unknown. “It is unclear whether there is a skin cancer risk,” he said. Reducing the number of exposures for a patient on maintenance therapy may be an option, he added.
Short-term treatment with NB-UVB can be effective in some psoriasis patients, such as in cases of an acute flare of guttate psoriasis, Dr. Gordon noted. Psoralen and UV light therapy (PUVA) is another option, he said.
Phototherapy also has a role in managing psoriasis in conjunction with retinoids, Dr. Gordon said. He cited an example of a 59-year-old woman with palmar psoriasis. For this patient, he said he would recommend starting with acitretin for approximately 1 month, if the patient tolerates it. “If the acitretin works by itself, no need to add phototherapy,” he said. However, if UVB is added, account for the photosensitizing agent, he emphasized. “Decrease the starting dose by 25-50 mJ or decrease the skin type by one,” he said.
Some psoriasis patients will not benefit from phototherapy, particularly those with erythroderma, said Dr. Gordon. “If skin is highly inflamed, phototherapy could induce easy burning, Koebnerization,” or other complications, he noted.
Dr. Gordon disclosed that his department at Northwestern derives income from phototherapy, but he personally does not. He also disclosed receiving research grants from and/or serving as a consultant to multiple companies including AbbVie, Amgen, Celgene, Eli Lilly, and Janssen.
LAS VEGAS – Psoriasis patients who are unresponsive to topical therapy, do not have psoriatic arthritis, and can make regular office visits may be candidates for successful treatment with phototherapy.
A patient with type II skin and extensive plaque psoriasis could be treated with a targeted NB-UVB laser, Dr. Kenneth B. Gordon said at Skin Disease Education Foundation’s annual Las Vegas Dermatology Seminar.
However, it’s important to evaluate each patient individually on phototherapy decisions, rather than relying on published protocols, noted Dr. Gordon of Northwestern University Feinberg School of Medicine in Chicago.
The advantages of the NB-UVB laser for psoriasis include a high rate of response (data show more than 75% of patients achieve PASI 75), the ability to avoid treating uninvolved skin, and potential long-term clearing of lesions, he said.
For someone with extensive plaque psoriasis, Dr. Gordon said he would start with 3 treatments per week, at 200 mJ, and increase the fluence by 25 mJ if necessary. “A good response can be expected in 4-6 weeks,” he said.
As for long-term treatment, “I leave it up to the patient but offer long-term maintenance,” said Dr. Gordon. Long-term efficacy of NB-UVB for plaque psoriasis has not been well studied, and safety data are unknown. “It is unclear whether there is a skin cancer risk,” he said. Reducing the number of exposures for a patient on maintenance therapy may be an option, he added.
Short-term treatment with NB-UVB can be effective in some psoriasis patients, such as in cases of an acute flare of guttate psoriasis, Dr. Gordon noted. Psoralen and UV light therapy (PUVA) is another option, he said.
Phototherapy also has a role in managing psoriasis in conjunction with retinoids, Dr. Gordon said. He cited an example of a 59-year-old woman with palmar psoriasis. For this patient, he said he would recommend starting with acitretin for approximately 1 month, if the patient tolerates it. “If the acitretin works by itself, no need to add phototherapy,” he said. However, if UVB is added, account for the photosensitizing agent, he emphasized. “Decrease the starting dose by 25-50 mJ or decrease the skin type by one,” he said.
Some psoriasis patients will not benefit from phototherapy, particularly those with erythroderma, said Dr. Gordon. “If skin is highly inflamed, phototherapy could induce easy burning, Koebnerization,” or other complications, he noted.
Dr. Gordon disclosed that his department at Northwestern derives income from phototherapy, but he personally does not. He also disclosed receiving research grants from and/or serving as a consultant to multiple companies including AbbVie, Amgen, Celgene, Eli Lilly, and Janssen.
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
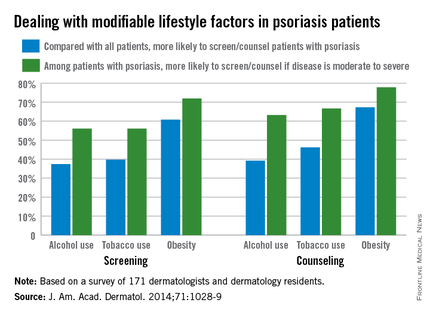
Dermatologists counsel psoriasis patients on obesity, less on alcohol and tobacco
Less than half of dermatologists screen or counsel psoriasis patients about alcohol or tobacco use, compared with all patients, but approximately 60% screen or counsel psoriasis patients about obesity, based on data from an online survey of 171 dermatologists and dermatology residents.
The findings were published in a letter to the editor in the November issue of the Journal of the American Academy of Dermatology (2014;71:1028-9).
However, among patients with psoriasis, more than two-thirds of the respondents said they were more likely to both screen and counsel patients with moderate to severe psoriasis about alcohol, tobacco, and obesity, compared with psoriasis patients overall.
Previous research suggests that if psoriasis patients’ issues with alcohol, tobacco, and obesity are addressed, “patients may experience disease improvement, emphasizing the importance of screening and counseling,” wrote Brandon L. Adler and Aimee E. Krausz, medical students at Albert Einstein College of Medicine, Bronx, N.Y., and their colleagues.
Overall, 87% of respondents believed themselves responsible for screening for the three risk factors, but 56% believed themselves responsible for counseling.
Although the results were limited by the focus on academic dermatologists, the findings suggest a need to improve dermatologists’ confidence in counseling patients, the researchers noted. “Systematic training and effective counseling instruments would empower practitioners to translate this knowledge into clinical practice.”
The study was supported by the Dermatology Foundation Career Development Awards Program.
The researchers had no financial conflicts to disclose.
|
Less than half of dermatologists screen or counsel psoriasis patients about alcohol or tobacco use, compared with all patients, but approximately 60% screen or counsel psoriasis patients about obesity, based on data from an online survey of 171 dermatologists and dermatology residents.
The findings were published in a letter to the editor in the November issue of the Journal of the American Academy of Dermatology (2014;71:1028-9).
However, among patients with psoriasis, more than two-thirds of the respondents said they were more likely to both screen and counsel patients with moderate to severe psoriasis about alcohol, tobacco, and obesity, compared with psoriasis patients overall.
Previous research suggests that if psoriasis patients’ issues with alcohol, tobacco, and obesity are addressed, “patients may experience disease improvement, emphasizing the importance of screening and counseling,” wrote Brandon L. Adler and Aimee E. Krausz, medical students at Albert Einstein College of Medicine, Bronx, N.Y., and their colleagues.
Overall, 87% of respondents believed themselves responsible for screening for the three risk factors, but 56% believed themselves responsible for counseling.
Although the results were limited by the focus on academic dermatologists, the findings suggest a need to improve dermatologists’ confidence in counseling patients, the researchers noted. “Systematic training and effective counseling instruments would empower practitioners to translate this knowledge into clinical practice.”
The study was supported by the Dermatology Foundation Career Development Awards Program.
The researchers had no financial conflicts to disclose.
|

Less than half of dermatologists screen or counsel psoriasis patients about alcohol or tobacco use, compared with all patients, but approximately 60% screen or counsel psoriasis patients about obesity, based on data from an online survey of 171 dermatologists and dermatology residents.
The findings were published in a letter to the editor in the November issue of the Journal of the American Academy of Dermatology (2014;71:1028-9).
However, among patients with psoriasis, more than two-thirds of the respondents said they were more likely to both screen and counsel patients with moderate to severe psoriasis about alcohol, tobacco, and obesity, compared with psoriasis patients overall.
Previous research suggests that if psoriasis patients’ issues with alcohol, tobacco, and obesity are addressed, “patients may experience disease improvement, emphasizing the importance of screening and counseling,” wrote Brandon L. Adler and Aimee E. Krausz, medical students at Albert Einstein College of Medicine, Bronx, N.Y., and their colleagues.
Overall, 87% of respondents believed themselves responsible for screening for the three risk factors, but 56% believed themselves responsible for counseling.
Although the results were limited by the focus on academic dermatologists, the findings suggest a need to improve dermatologists’ confidence in counseling patients, the researchers noted. “Systematic training and effective counseling instruments would empower practitioners to translate this knowledge into clinical practice.”
The study was supported by the Dermatology Foundation Career Development Awards Program.
The researchers had no financial conflicts to disclose.
|

FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY







