User login
Peripheral manifestations make mark on spondyloarthritis trajectory
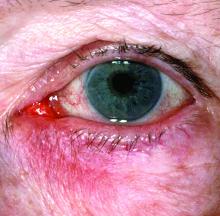
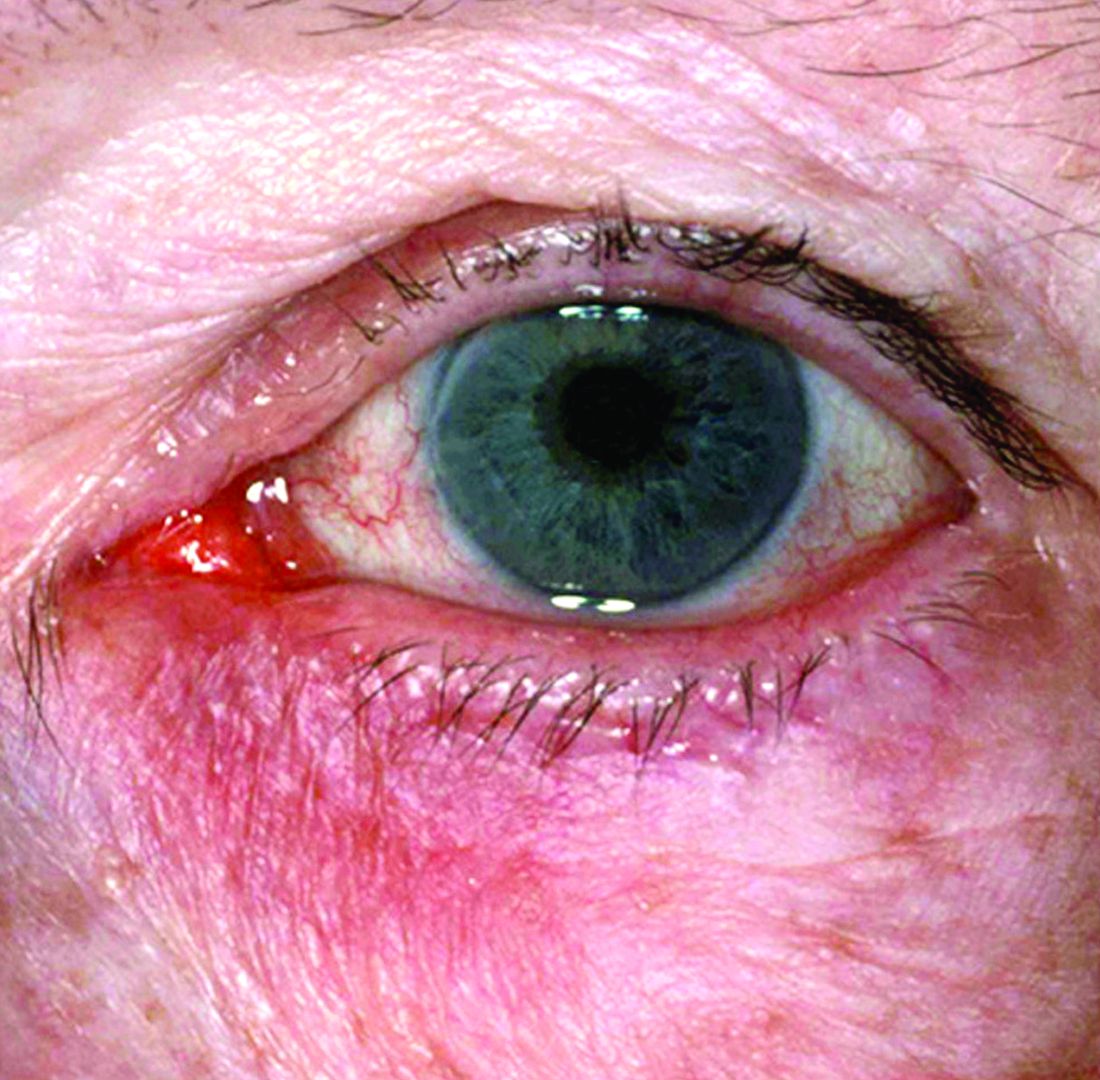
Peripheral manifestations contribute significantly to disease activity in adults with spondyloarthritis (SpA), point toward a generally worse prognosis, and play a big role in defining the phenotypic clustering of the heterogenous disease, according to findings from what researchers called the first prospective study “to comprehensively describe the prevalence, clinical patterns, and prognostic implications of peripheral manifestations across the entire SpA spectrum.”
The stratification of patients in the study based on the presence of peripheral manifestations (arthritis, enthesitis, and/or dactylitis) led to the identification of an endotype with unfavorable outcomes, which not only has prognostic value but supports the need for an endotype-based treatment approach rather than one centered on C-reactive protein (CRP) and the Ankylosing Spondylitis Disease Activity Score (ASDAS).
The findings “advocate strongly for the presence of a distinct SpA endotype, based on potentially different immunopathological mechanisms and characterized by high disease activity at initial presentation with lack of substantial improvement upon follow-up,” first author Ann-Sophie De Craemer, MD, of Ghent, Belgium, and colleagues wrote.
Because the diagnostic and prognostic value of peripheral manifestations has not been well studied in SpA in general and in newly diagnosed patients in particular, Dr. De Craemer and associates decided to analyze their impact in 367 patients in the Be-Giant (BelGian Inflammatory Arthritis and spoNdylitis cohorT) cohort, a multicenter, prospective, observational cohort of newly diagnosed patients with SpA in Belgium. The study was published in Rheumatology.
The study population included 257 (70%) patients with axial-predominant SpA (axSpA) as classified by Assessment of Spondyloarthritis International Society criteria and 110 (30%) with peripheral-predominant SpA (pSpA) as defined by ASAS criteria. A total of 52 patients with axSpA had peripheral manifestations at baseline. The mean age of the patients was 34 years, and 52% were male.
The 162 patients with peripheral manifestations included 143 with arthritis, 52 with enthesitis, and 55 with dactylitis.
Two patient clusters emerge from data
In a cluster analysis that used baseline clinical features, the researchers divided the patients into cluster A (of which 242 of 248 were patients with axSpA) and cluster B (of which 104 of 119 were patients with pSpA). Most of the patients with peripheral manifestations were in cluster B (117 of 162 [72%]), compared with cluster A (45 of 162 [28%]).
A longitudinal analysis included 195 patients who completed a minimum 2-year follow-up. The longitudinal analysis identified high- and low-disease activity trajectories in each cluster.
In axSpA-predominant cluster A, patients with “high” trajectory had high disease activity levels at baseline (mean ASDAS-CRP, 3.2) that remained relatively stable, while those in the low-trajectory group (62%) had less disease activity at baseline (mean ASDAS-CRP, 2.0), which then further declined during follow-up.
Patients in the high trajectory in cluster A were more often affected by peripheral manifestations, “which remained a significant predictor in multivariate analysis,” with an odds ratio of 2.4, the researchers noted. In addition, patients with peripheral manifestations were significantly more likely to have persistent high disease activity despite starting biologics earlier than patients without peripheral manifestations (hazard ratio, 2.1).
Patients in pSpA-predominant cluster B showed differences that were similar to those seen in cluster A in terms of high– and low–disease activity trajectories (mean ASDAS-CRP of 3.6 and 2.8, respectively), but among these patients, a high level of disease activity was significantly associated with elevated CRP, rather than with peripheral disease, the researchers said.
The study findings were limited by several factors, including the exclusion of patients who did not complete the follow-up, which reduced the sample size for longitudinal analysis. However, the results were strengthened by the inclusion of patients from the full SpA spectrum, a geographically spread-out patient population, and a study design that mirrored clinical practice, the researchers noted.
The Be-Giant cohort was supported by an unrestricted grant from AbbVie. Several authors reported financial relationships with AbbVie and other pharmaceutical companies.
Peripheral manifestations contribute significantly to disease activity in adults with spondyloarthritis (SpA), point toward a generally worse prognosis, and play a big role in defining the phenotypic clustering of the heterogenous disease, according to findings from what researchers called the first prospective study “to comprehensively describe the prevalence, clinical patterns, and prognostic implications of peripheral manifestations across the entire SpA spectrum.”
The stratification of patients in the study based on the presence of peripheral manifestations (arthritis, enthesitis, and/or dactylitis) led to the identification of an endotype with unfavorable outcomes, which not only has prognostic value but supports the need for an endotype-based treatment approach rather than one centered on C-reactive protein (CRP) and the Ankylosing Spondylitis Disease Activity Score (ASDAS).
The findings “advocate strongly for the presence of a distinct SpA endotype, based on potentially different immunopathological mechanisms and characterized by high disease activity at initial presentation with lack of substantial improvement upon follow-up,” first author Ann-Sophie De Craemer, MD, of Ghent, Belgium, and colleagues wrote.
Because the diagnostic and prognostic value of peripheral manifestations has not been well studied in SpA in general and in newly diagnosed patients in particular, Dr. De Craemer and associates decided to analyze their impact in 367 patients in the Be-Giant (BelGian Inflammatory Arthritis and spoNdylitis cohorT) cohort, a multicenter, prospective, observational cohort of newly diagnosed patients with SpA in Belgium. The study was published in Rheumatology.
The study population included 257 (70%) patients with axial-predominant SpA (axSpA) as classified by Assessment of Spondyloarthritis International Society criteria and 110 (30%) with peripheral-predominant SpA (pSpA) as defined by ASAS criteria. A total of 52 patients with axSpA had peripheral manifestations at baseline. The mean age of the patients was 34 years, and 52% were male.
The 162 patients with peripheral manifestations included 143 with arthritis, 52 with enthesitis, and 55 with dactylitis.
Two patient clusters emerge from data
In a cluster analysis that used baseline clinical features, the researchers divided the patients into cluster A (of which 242 of 248 were patients with axSpA) and cluster B (of which 104 of 119 were patients with pSpA). Most of the patients with peripheral manifestations were in cluster B (117 of 162 [72%]), compared with cluster A (45 of 162 [28%]).
A longitudinal analysis included 195 patients who completed a minimum 2-year follow-up. The longitudinal analysis identified high- and low-disease activity trajectories in each cluster.
In axSpA-predominant cluster A, patients with “high” trajectory had high disease activity levels at baseline (mean ASDAS-CRP, 3.2) that remained relatively stable, while those in the low-trajectory group (62%) had less disease activity at baseline (mean ASDAS-CRP, 2.0), which then further declined during follow-up.
Patients in the high trajectory in cluster A were more often affected by peripheral manifestations, “which remained a significant predictor in multivariate analysis,” with an odds ratio of 2.4, the researchers noted. In addition, patients with peripheral manifestations were significantly more likely to have persistent high disease activity despite starting biologics earlier than patients without peripheral manifestations (hazard ratio, 2.1).
Patients in pSpA-predominant cluster B showed differences that were similar to those seen in cluster A in terms of high– and low–disease activity trajectories (mean ASDAS-CRP of 3.6 and 2.8, respectively), but among these patients, a high level of disease activity was significantly associated with elevated CRP, rather than with peripheral disease, the researchers said.
The study findings were limited by several factors, including the exclusion of patients who did not complete the follow-up, which reduced the sample size for longitudinal analysis. However, the results were strengthened by the inclusion of patients from the full SpA spectrum, a geographically spread-out patient population, and a study design that mirrored clinical practice, the researchers noted.
The Be-Giant cohort was supported by an unrestricted grant from AbbVie. Several authors reported financial relationships with AbbVie and other pharmaceutical companies.
Peripheral manifestations contribute significantly to disease activity in adults with spondyloarthritis (SpA), point toward a generally worse prognosis, and play a big role in defining the phenotypic clustering of the heterogenous disease, according to findings from what researchers called the first prospective study “to comprehensively describe the prevalence, clinical patterns, and prognostic implications of peripheral manifestations across the entire SpA spectrum.”
The stratification of patients in the study based on the presence of peripheral manifestations (arthritis, enthesitis, and/or dactylitis) led to the identification of an endotype with unfavorable outcomes, which not only has prognostic value but supports the need for an endotype-based treatment approach rather than one centered on C-reactive protein (CRP) and the Ankylosing Spondylitis Disease Activity Score (ASDAS).
The findings “advocate strongly for the presence of a distinct SpA endotype, based on potentially different immunopathological mechanisms and characterized by high disease activity at initial presentation with lack of substantial improvement upon follow-up,” first author Ann-Sophie De Craemer, MD, of Ghent, Belgium, and colleagues wrote.
Because the diagnostic and prognostic value of peripheral manifestations has not been well studied in SpA in general and in newly diagnosed patients in particular, Dr. De Craemer and associates decided to analyze their impact in 367 patients in the Be-Giant (BelGian Inflammatory Arthritis and spoNdylitis cohorT) cohort, a multicenter, prospective, observational cohort of newly diagnosed patients with SpA in Belgium. The study was published in Rheumatology.
The study population included 257 (70%) patients with axial-predominant SpA (axSpA) as classified by Assessment of Spondyloarthritis International Society criteria and 110 (30%) with peripheral-predominant SpA (pSpA) as defined by ASAS criteria. A total of 52 patients with axSpA had peripheral manifestations at baseline. The mean age of the patients was 34 years, and 52% were male.
The 162 patients with peripheral manifestations included 143 with arthritis, 52 with enthesitis, and 55 with dactylitis.
Two patient clusters emerge from data
In a cluster analysis that used baseline clinical features, the researchers divided the patients into cluster A (of which 242 of 248 were patients with axSpA) and cluster B (of which 104 of 119 were patients with pSpA). Most of the patients with peripheral manifestations were in cluster B (117 of 162 [72%]), compared with cluster A (45 of 162 [28%]).
A longitudinal analysis included 195 patients who completed a minimum 2-year follow-up. The longitudinal analysis identified high- and low-disease activity trajectories in each cluster.
In axSpA-predominant cluster A, patients with “high” trajectory had high disease activity levels at baseline (mean ASDAS-CRP, 3.2) that remained relatively stable, while those in the low-trajectory group (62%) had less disease activity at baseline (mean ASDAS-CRP, 2.0), which then further declined during follow-up.
Patients in the high trajectory in cluster A were more often affected by peripheral manifestations, “which remained a significant predictor in multivariate analysis,” with an odds ratio of 2.4, the researchers noted. In addition, patients with peripheral manifestations were significantly more likely to have persistent high disease activity despite starting biologics earlier than patients without peripheral manifestations (hazard ratio, 2.1).
Patients in pSpA-predominant cluster B showed differences that were similar to those seen in cluster A in terms of high– and low–disease activity trajectories (mean ASDAS-CRP of 3.6 and 2.8, respectively), but among these patients, a high level of disease activity was significantly associated with elevated CRP, rather than with peripheral disease, the researchers said.
The study findings were limited by several factors, including the exclusion of patients who did not complete the follow-up, which reduced the sample size for longitudinal analysis. However, the results were strengthened by the inclusion of patients from the full SpA spectrum, a geographically spread-out patient population, and a study design that mirrored clinical practice, the researchers noted.
The Be-Giant cohort was supported by an unrestricted grant from AbbVie. Several authors reported financial relationships with AbbVie and other pharmaceutical companies.
FROM RHEUMATOLOGY
Poorly controlled asthma predicts COVID-19 hospitalization in children
Children and adolescents with poorly controlled asthma were three to six times more likely to be hospitalized with COVID-19 infections, based on data from a national study of more than 750,000 children in Scotland.
Although the majority of COVID-19 cases in children have been mild, some children require hospitalization, wrote Ting Shi, PhD, of the University of Edinburgh (Scotland) and colleagues.
Vaccination policies to potentially reduce infection and hospitalization of children remain inconsistent, the researchers said. Identifying which school-age children would derive the greatest benefit from vaccination “could help to reduce the risk of infection and consequently the need for children to have time off school; and might also reduce the risk of spread of SARS-CoV-2 within schools and households,” but the potential benefits of vaccination for children with asthma in particular have not been well studied, they wrote.
The United Kingdom’s Joint Commission on Vaccination and Immunisation commissioned research on the rates of hospitalization among children with poorly controlled asthma. In a national incidence cohort study published in The Lancet Respiratory Medicine, the researchers reviewed data from all children aged 5-17 years in Scotland who were enrolled in the linked dataset of Early Pandemic Evaluation and Enhanced Surveillance of COVID-19 (EAVE II). The total number of children in the dataset was 752,867, and 63,463 (8.4%) of these had diagnosed asthma. Among the children with asthma, 4,339 (6.8%) had confirmed COVID-19 infections between March 1, 2020, and July 27, 2021. A total of 67 infected children were hospitalized. Of the 689,404 children without asthma, 40,231 (5.8%) had confirmed COVID-19 infections, and 382 (0.9%) of these children were hospitalized.
Overall, hospital admission rates for COVID-19 were significantly higher among children with asthma, compared to those without asthma (adjusted hazard ratio, 1.49), and the rates increased among children with poorly controlled asthma.
The researchers used previous hospital admission for asthma as a measure of uncontrolled asthma, and found that hospitalization was at least six times as likely for children with poorly controlled asthma, compared with those with no asthma (aHR, 6.40), although children with well-controlled asthma also had an increased risk of hospitalization, compared with those with no asthma (aHR, 1.36).
When the researchers used oral corticosteroid prescriptions as an indicator of uncontrolled asthma, the adjusted hazard ratios were 3.38, 3.53, 1.52, and 1.34 for children with prescribed corticosteroid courses of three or more, two, one, and none, respectively, compared with children with no asthma.
These hazard ratios remained significant after controlling for factors including age, sex, socioeconomic status, comorbidity, and previous hospital admission, the researchers wrote.
In an age-based analysis, results were similar for children aged 12-17 years, but in children aged 5-11 years, the hospitalization risk decreased for those with one course of corticosteroids and reached the highest rate for those with three or more courses, rather than two courses.
The study findings were limited by several factors including the relatively small numbers of COVID-19 hospitalizations, ICU admissions, and deaths in children with asthma, the researchers noted. Other limitations include potential changes in asthma control over the study period, and lack of data on certain confounders such as tobacco use, unsuitable housing, and ethnicity, they noted. However, the results were strengthened by the use of a large, national dataset, and access to electronic health records, they said.
The findings reflect data from previous studies suggesting increased risk of hospitalization for patients with respiratory illness who develop COVID-19 infections, the researchers wrote.
The results emphasize the importance of good asthma control to protect children from severe COVID-19, and careful monitoring of children with poorly controlled asthma who do become infected, they added.
“The findings from this linkage of multiple data sources have helped inform the prioritisation of school-aged children with poorly controlled asthma for vaccines,” they concluded.
Findings support value of vaccination for children with asthma
“Pediatricians see many children who suffer from asthma, and although one could assume that these children would have more serious consequences from contracting COVID-19, the current study examines a large database in a way not possible in the United States to address the severity question,” said Suzanne C. Boulter, MD, of the Geisel School of Medicine at Dartmouth, Hanover, N.H. “The authors used prior hospitalization rate or two prescriptions for oral corticosteroids as markers of asthma severity prior to the onset of COVID-19 in Scotland, and they collected retrospective data for 16 months of the pandemic through July of 2021, showing a significant increase in hospitalization for those children,” she said. Dr. Boulter said she was not surprised by this finding, given the impact of COVID-19 on the respiratory system.
“Pediatricians have found significant challenges from some groups of parents when discussing the indications and need for vaccination in their patients,” said Dr. Boulter. “Having this data on the increased risk of morbidity and mortality in children with asthma might help parents who are uncertain about the risk/benefit ratio of the vaccine make their decision,” she said.
Dr. Boulter said she hoped that additional studies will yield ongoing information about hospitalization rates for COVID-19 not only about asthma, but also other diagnoses affecting children in the United States and worldwide.
“It would also be important to see a breakdown of ethnic factors and adverse childhood experiences and how they relate to hospitalization and death from COVID-19,” Dr. Boulter said.
“The results of this study are not surprising, as we have known for a long time that children with severe asthma are more susceptible to severe respiratory viruses,” Francis E. Rushton, MD, a pediatrician in Beaufort, S.C., said in an interview. “But the study is still important, as it helps us determine which children are most urgently in need of protection from COVID-19 in any of its forms,” he emphasized. In particular, the current study underlines the importance of vaccinating children with unstable asthma, Dr. Rushton said.
Going forward, “it would be interesting to do additional studies looking at other markers for poor asthma control that could guide our vaccine efforts so that they are focused on those most at risk,” he added.
The study was supported by the UK Research and Innovation (Medical Research Council), Research and Innovation Industrial Strategy Challenge Fund, Health Data Research UK, and the Scottish Government. Lead author Dr. Shi had no financial conflicts to disclose. Dr. Rushton and Dr. Boulter had no financial conflicts to disclose, but each serves on the Editorial Advisory Board of Pediatric News.
Children and adolescents with poorly controlled asthma were three to six times more likely to be hospitalized with COVID-19 infections, based on data from a national study of more than 750,000 children in Scotland.
Although the majority of COVID-19 cases in children have been mild, some children require hospitalization, wrote Ting Shi, PhD, of the University of Edinburgh (Scotland) and colleagues.
Vaccination policies to potentially reduce infection and hospitalization of children remain inconsistent, the researchers said. Identifying which school-age children would derive the greatest benefit from vaccination “could help to reduce the risk of infection and consequently the need for children to have time off school; and might also reduce the risk of spread of SARS-CoV-2 within schools and households,” but the potential benefits of vaccination for children with asthma in particular have not been well studied, they wrote.
The United Kingdom’s Joint Commission on Vaccination and Immunisation commissioned research on the rates of hospitalization among children with poorly controlled asthma. In a national incidence cohort study published in The Lancet Respiratory Medicine, the researchers reviewed data from all children aged 5-17 years in Scotland who were enrolled in the linked dataset of Early Pandemic Evaluation and Enhanced Surveillance of COVID-19 (EAVE II). The total number of children in the dataset was 752,867, and 63,463 (8.4%) of these had diagnosed asthma. Among the children with asthma, 4,339 (6.8%) had confirmed COVID-19 infections between March 1, 2020, and July 27, 2021. A total of 67 infected children were hospitalized. Of the 689,404 children without asthma, 40,231 (5.8%) had confirmed COVID-19 infections, and 382 (0.9%) of these children were hospitalized.
Overall, hospital admission rates for COVID-19 were significantly higher among children with asthma, compared to those without asthma (adjusted hazard ratio, 1.49), and the rates increased among children with poorly controlled asthma.
The researchers used previous hospital admission for asthma as a measure of uncontrolled asthma, and found that hospitalization was at least six times as likely for children with poorly controlled asthma, compared with those with no asthma (aHR, 6.40), although children with well-controlled asthma also had an increased risk of hospitalization, compared with those with no asthma (aHR, 1.36).
When the researchers used oral corticosteroid prescriptions as an indicator of uncontrolled asthma, the adjusted hazard ratios were 3.38, 3.53, 1.52, and 1.34 for children with prescribed corticosteroid courses of three or more, two, one, and none, respectively, compared with children with no asthma.
These hazard ratios remained significant after controlling for factors including age, sex, socioeconomic status, comorbidity, and previous hospital admission, the researchers wrote.
In an age-based analysis, results were similar for children aged 12-17 years, but in children aged 5-11 years, the hospitalization risk decreased for those with one course of corticosteroids and reached the highest rate for those with three or more courses, rather than two courses.
The study findings were limited by several factors including the relatively small numbers of COVID-19 hospitalizations, ICU admissions, and deaths in children with asthma, the researchers noted. Other limitations include potential changes in asthma control over the study period, and lack of data on certain confounders such as tobacco use, unsuitable housing, and ethnicity, they noted. However, the results were strengthened by the use of a large, national dataset, and access to electronic health records, they said.
The findings reflect data from previous studies suggesting increased risk of hospitalization for patients with respiratory illness who develop COVID-19 infections, the researchers wrote.
The results emphasize the importance of good asthma control to protect children from severe COVID-19, and careful monitoring of children with poorly controlled asthma who do become infected, they added.
“The findings from this linkage of multiple data sources have helped inform the prioritisation of school-aged children with poorly controlled asthma for vaccines,” they concluded.
Findings support value of vaccination for children with asthma
“Pediatricians see many children who suffer from asthma, and although one could assume that these children would have more serious consequences from contracting COVID-19, the current study examines a large database in a way not possible in the United States to address the severity question,” said Suzanne C. Boulter, MD, of the Geisel School of Medicine at Dartmouth, Hanover, N.H. “The authors used prior hospitalization rate or two prescriptions for oral corticosteroids as markers of asthma severity prior to the onset of COVID-19 in Scotland, and they collected retrospective data for 16 months of the pandemic through July of 2021, showing a significant increase in hospitalization for those children,” she said. Dr. Boulter said she was not surprised by this finding, given the impact of COVID-19 on the respiratory system.
“Pediatricians have found significant challenges from some groups of parents when discussing the indications and need for vaccination in their patients,” said Dr. Boulter. “Having this data on the increased risk of morbidity and mortality in children with asthma might help parents who are uncertain about the risk/benefit ratio of the vaccine make their decision,” she said.
Dr. Boulter said she hoped that additional studies will yield ongoing information about hospitalization rates for COVID-19 not only about asthma, but also other diagnoses affecting children in the United States and worldwide.
“It would also be important to see a breakdown of ethnic factors and adverse childhood experiences and how they relate to hospitalization and death from COVID-19,” Dr. Boulter said.
“The results of this study are not surprising, as we have known for a long time that children with severe asthma are more susceptible to severe respiratory viruses,” Francis E. Rushton, MD, a pediatrician in Beaufort, S.C., said in an interview. “But the study is still important, as it helps us determine which children are most urgently in need of protection from COVID-19 in any of its forms,” he emphasized. In particular, the current study underlines the importance of vaccinating children with unstable asthma, Dr. Rushton said.
Going forward, “it would be interesting to do additional studies looking at other markers for poor asthma control that could guide our vaccine efforts so that they are focused on those most at risk,” he added.
The study was supported by the UK Research and Innovation (Medical Research Council), Research and Innovation Industrial Strategy Challenge Fund, Health Data Research UK, and the Scottish Government. Lead author Dr. Shi had no financial conflicts to disclose. Dr. Rushton and Dr. Boulter had no financial conflicts to disclose, but each serves on the Editorial Advisory Board of Pediatric News.
Children and adolescents with poorly controlled asthma were three to six times more likely to be hospitalized with COVID-19 infections, based on data from a national study of more than 750,000 children in Scotland.
Although the majority of COVID-19 cases in children have been mild, some children require hospitalization, wrote Ting Shi, PhD, of the University of Edinburgh (Scotland) and colleagues.
Vaccination policies to potentially reduce infection and hospitalization of children remain inconsistent, the researchers said. Identifying which school-age children would derive the greatest benefit from vaccination “could help to reduce the risk of infection and consequently the need for children to have time off school; and might also reduce the risk of spread of SARS-CoV-2 within schools and households,” but the potential benefits of vaccination for children with asthma in particular have not been well studied, they wrote.
The United Kingdom’s Joint Commission on Vaccination and Immunisation commissioned research on the rates of hospitalization among children with poorly controlled asthma. In a national incidence cohort study published in The Lancet Respiratory Medicine, the researchers reviewed data from all children aged 5-17 years in Scotland who were enrolled in the linked dataset of Early Pandemic Evaluation and Enhanced Surveillance of COVID-19 (EAVE II). The total number of children in the dataset was 752,867, and 63,463 (8.4%) of these had diagnosed asthma. Among the children with asthma, 4,339 (6.8%) had confirmed COVID-19 infections between March 1, 2020, and July 27, 2021. A total of 67 infected children were hospitalized. Of the 689,404 children without asthma, 40,231 (5.8%) had confirmed COVID-19 infections, and 382 (0.9%) of these children were hospitalized.
Overall, hospital admission rates for COVID-19 were significantly higher among children with asthma, compared to those without asthma (adjusted hazard ratio, 1.49), and the rates increased among children with poorly controlled asthma.
The researchers used previous hospital admission for asthma as a measure of uncontrolled asthma, and found that hospitalization was at least six times as likely for children with poorly controlled asthma, compared with those with no asthma (aHR, 6.40), although children with well-controlled asthma also had an increased risk of hospitalization, compared with those with no asthma (aHR, 1.36).
When the researchers used oral corticosteroid prescriptions as an indicator of uncontrolled asthma, the adjusted hazard ratios were 3.38, 3.53, 1.52, and 1.34 for children with prescribed corticosteroid courses of three or more, two, one, and none, respectively, compared with children with no asthma.
These hazard ratios remained significant after controlling for factors including age, sex, socioeconomic status, comorbidity, and previous hospital admission, the researchers wrote.
In an age-based analysis, results were similar for children aged 12-17 years, but in children aged 5-11 years, the hospitalization risk decreased for those with one course of corticosteroids and reached the highest rate for those with three or more courses, rather than two courses.
The study findings were limited by several factors including the relatively small numbers of COVID-19 hospitalizations, ICU admissions, and deaths in children with asthma, the researchers noted. Other limitations include potential changes in asthma control over the study period, and lack of data on certain confounders such as tobacco use, unsuitable housing, and ethnicity, they noted. However, the results were strengthened by the use of a large, national dataset, and access to electronic health records, they said.
The findings reflect data from previous studies suggesting increased risk of hospitalization for patients with respiratory illness who develop COVID-19 infections, the researchers wrote.
The results emphasize the importance of good asthma control to protect children from severe COVID-19, and careful monitoring of children with poorly controlled asthma who do become infected, they added.
“The findings from this linkage of multiple data sources have helped inform the prioritisation of school-aged children with poorly controlled asthma for vaccines,” they concluded.
Findings support value of vaccination for children with asthma
“Pediatricians see many children who suffer from asthma, and although one could assume that these children would have more serious consequences from contracting COVID-19, the current study examines a large database in a way not possible in the United States to address the severity question,” said Suzanne C. Boulter, MD, of the Geisel School of Medicine at Dartmouth, Hanover, N.H. “The authors used prior hospitalization rate or two prescriptions for oral corticosteroids as markers of asthma severity prior to the onset of COVID-19 in Scotland, and they collected retrospective data for 16 months of the pandemic through July of 2021, showing a significant increase in hospitalization for those children,” she said. Dr. Boulter said she was not surprised by this finding, given the impact of COVID-19 on the respiratory system.
“Pediatricians have found significant challenges from some groups of parents when discussing the indications and need for vaccination in their patients,” said Dr. Boulter. “Having this data on the increased risk of morbidity and mortality in children with asthma might help parents who are uncertain about the risk/benefit ratio of the vaccine make their decision,” she said.
Dr. Boulter said she hoped that additional studies will yield ongoing information about hospitalization rates for COVID-19 not only about asthma, but also other diagnoses affecting children in the United States and worldwide.
“It would also be important to see a breakdown of ethnic factors and adverse childhood experiences and how they relate to hospitalization and death from COVID-19,” Dr. Boulter said.
“The results of this study are not surprising, as we have known for a long time that children with severe asthma are more susceptible to severe respiratory viruses,” Francis E. Rushton, MD, a pediatrician in Beaufort, S.C., said in an interview. “But the study is still important, as it helps us determine which children are most urgently in need of protection from COVID-19 in any of its forms,” he emphasized. In particular, the current study underlines the importance of vaccinating children with unstable asthma, Dr. Rushton said.
Going forward, “it would be interesting to do additional studies looking at other markers for poor asthma control that could guide our vaccine efforts so that they are focused on those most at risk,” he added.
The study was supported by the UK Research and Innovation (Medical Research Council), Research and Innovation Industrial Strategy Challenge Fund, Health Data Research UK, and the Scottish Government. Lead author Dr. Shi had no financial conflicts to disclose. Dr. Rushton and Dr. Boulter had no financial conflicts to disclose, but each serves on the Editorial Advisory Board of Pediatric News.
FROM THE LANCET
Sickle cell raises risk for stillbirth
Both sickle cell trait and sickle cell disease were significantly associated with an increased risk of stillbirth, based on data from more than 50,000 women.
Pregnant women with sickle cell disease (SCD) are at increased risk of complications, including stillbirth, but many women with the disease in the United States lack access to specialty care, Silvia P. Canelón, PhD, of the University of Pennsylvania, Philadelphia, and colleagues wrote. Sickle cell trait (SCT), defined as one abnormal allele of the hemoglobin gene, is not considered a disease state because many carriers are asymptomatic, and therefore even less likely to be assessed for potential complications. “However, it is possible for people with SCT to experience sickling of red blood cells under severe hypoxia, dehydration, and hyperthermia. This condition can lead to severe medical complications for sickle cell carriers, including fetal loss, splenic infarction, exercise-related sudden death, and others,” they noted.
In a study published in JAMA Network Open, the researchers reviewed data from 63,334 deliveries in 50,560 women between Jan. 1, 2010, and Aug. 15, 2017, at four quaternary academic medical centers in Pennsylvania. Of these, 1,904 had SCT but not SCD, and 164 had SCD. The mean age of the women was 29.5 years, and approximately 56% were single at the time of delivery. A majority (87%) of the study population was Rhesus-factor positive, 47.0% were Black or African American, 33.7% were White, and 45.2% had ABO blood type O.
Risk factors for stillbirth used in the analysis included SCD, numbers of pain crises and blood transfusions before delivery, delivery episode (to represent parity), history of cesarean delivery, multiple gestation, age, marital status, race and ethnicity, ABO blood type, Rhesus factor, and year of delivery.
Overall, the prevalence of stillbirth in women with SCT was 1.1%, compared with 0.8% in the general study population, and was significantly associated with increased risk of stillbirth after controlling for multiple risk factors. The adjusted odds ratio was 8.94 for stillbirth risk in women with SCT, compared with women without SCT (P = .045), although the risk was greater among women with SCD, compared with those without SCD (aOR, 26.40).
“In addition, the stratified analysis found Black or African American patients with SCD to be at higher risk of stillbirth, compared with Black or African American patients without SCD (aOR, 3.59),” but no significant association was noted between stillbirth and SCT, the researchers wrote. Stillbirth rates were 1.1% in Black or African American women overall, 2.7% in those with SCD, and 1.0% in those with SCT. Overall, multiple gestation was associated with an increased risk of stillbirth (aOR, 4.68), while a history of cesarean delivery and being married at the time of delivery were associated with decreased risk (aOR, 0.44 and 0.72, respectively).
The lack of association between stillbirth and SCT in Black or African American patients supports some previous research, but contradicts other studies, the researchers wrote. “Ultimately, it may be impossible to disentangle the risks due to the disease and those due to disparities associated with the disease that have resulted from longstanding inequity and stigma,” they said. The findings also suggest that biological mechanisms of SCT may contribute to severe clinical complications, and therefore “invite a more critical examination of the assumption that SCT is not a disease state.”
The study findings were limited by several factors including the lack of assessment of SCT independent of other comorbidities, such as hypertension, preeclampsia, diabetes, and obesity, and by the use of billing codes that could misclassify patients, the researchers noted.
However, the results support some findings from previous studies of the potential health complications for pregnant SCT patients. The large study population highlights the need to identify women’s SCT status during obstetric care, and to provide both pregnancy guidance for SCT patients and systemic support of comprehensive care for SCD and SCT patients, they concluded.
Disparities may drive stillbirth in sickle cell trait women
“There is a paucity of research evaluating sickle cell trait and the risk of adverse pregnancy outcomes such as stillbirth,” Iris Krishna, MD, of Emory University, Atlanta, said in an interview. “Prior studies evaluating the risk of stillbirth have yielded mixed results, and an increased risk of stillbirth in women with sickle cell trait has not been established. This study is unique in that it attempts to address how racial inequities and health disparities may contribute to risk of stillbirth in women with sickle cell trait.”
Although the study findings suggest an increased risk of stillbirth in women with sickle cell trait, an analysis stratified for Black or African American patients showed no association, Dr. Krishna said. “The prevalence of stillbirth was noted to be 1% among Black or African American patients with sickle cell trait compared to the prevalence of stillbirth of 1.1% among Black or African American women with no sickle cell trait or disease. Although, sickle cell trait or sickle cell disease can be found in any racial or ethnic group, it disproportionately affects Black or African Americans, with a sickle cell trait carrier rate of approximately 1 in 10. The mixed findings in this study amongst racial/ethnic groups further suggest that there is more research needed before an association between stillbirth and sickle cell trait can be supported.”
As for clinical implications, “it is well established that for women with sickle cell trait there is an increased risk of urinary tract infections in pregnancy,” said Dr. Krishna. “Women with sickle cell trait should have a urine culture performed at their first prenatal visit and each trimester. At this time, studies evaluating risk of stillbirth in women with sickle cell trait have yielded conflicting results, and current consensus is that women with sickle cell trait are not at increased risk. In comparison, women with sickle cell disease are at increased risk for stillbirth and adverse pregnancy outcomes. Women with sickle cell disease should be followed closely during pregnancy and fetal surveillance implemented at 32 weeks, if not sooner, to reduce risk of stillbirth.
“Prior studies evaluating risk of stillbirth in women with sickle cell trait consist of retrospective cohorts with small study populations,” Dr. Krishna added. Notably, the current study was limited by the inability to adjust for comorbidities including diabetes, hypertension, and obesity, that are not only associated with an increased risk for stillbirth, but also disproportionately common among Black women.
“More studies are needed evaluating the relationship between these comorbidities as well as studies specifically evaluating how race affects care and pregnancy outcomes,” Dr. Krisha emphasized.
The study was funded by the University of Pennsylvania department of biostatistics, epidemiology, and informatics. Lead author Dr. Canelón disclosed grants from the Centers for Disease Control and Prevention, Clinical and Translational Science Awards, and grants from the National Institutes of Health outside the submitted work. Dr. Krishna had no financial conflicts to disclose, but serves on the editorial advisory board of Ob.Gyn News.
Both sickle cell trait and sickle cell disease were significantly associated with an increased risk of stillbirth, based on data from more than 50,000 women.
Pregnant women with sickle cell disease (SCD) are at increased risk of complications, including stillbirth, but many women with the disease in the United States lack access to specialty care, Silvia P. Canelón, PhD, of the University of Pennsylvania, Philadelphia, and colleagues wrote. Sickle cell trait (SCT), defined as one abnormal allele of the hemoglobin gene, is not considered a disease state because many carriers are asymptomatic, and therefore even less likely to be assessed for potential complications. “However, it is possible for people with SCT to experience sickling of red blood cells under severe hypoxia, dehydration, and hyperthermia. This condition can lead to severe medical complications for sickle cell carriers, including fetal loss, splenic infarction, exercise-related sudden death, and others,” they noted.
In a study published in JAMA Network Open, the researchers reviewed data from 63,334 deliveries in 50,560 women between Jan. 1, 2010, and Aug. 15, 2017, at four quaternary academic medical centers in Pennsylvania. Of these, 1,904 had SCT but not SCD, and 164 had SCD. The mean age of the women was 29.5 years, and approximately 56% were single at the time of delivery. A majority (87%) of the study population was Rhesus-factor positive, 47.0% were Black or African American, 33.7% were White, and 45.2% had ABO blood type O.
Risk factors for stillbirth used in the analysis included SCD, numbers of pain crises and blood transfusions before delivery, delivery episode (to represent parity), history of cesarean delivery, multiple gestation, age, marital status, race and ethnicity, ABO blood type, Rhesus factor, and year of delivery.
Overall, the prevalence of stillbirth in women with SCT was 1.1%, compared with 0.8% in the general study population, and was significantly associated with increased risk of stillbirth after controlling for multiple risk factors. The adjusted odds ratio was 8.94 for stillbirth risk in women with SCT, compared with women without SCT (P = .045), although the risk was greater among women with SCD, compared with those without SCD (aOR, 26.40).
“In addition, the stratified analysis found Black or African American patients with SCD to be at higher risk of stillbirth, compared with Black or African American patients without SCD (aOR, 3.59),” but no significant association was noted between stillbirth and SCT, the researchers wrote. Stillbirth rates were 1.1% in Black or African American women overall, 2.7% in those with SCD, and 1.0% in those with SCT. Overall, multiple gestation was associated with an increased risk of stillbirth (aOR, 4.68), while a history of cesarean delivery and being married at the time of delivery were associated with decreased risk (aOR, 0.44 and 0.72, respectively).
The lack of association between stillbirth and SCT in Black or African American patients supports some previous research, but contradicts other studies, the researchers wrote. “Ultimately, it may be impossible to disentangle the risks due to the disease and those due to disparities associated with the disease that have resulted from longstanding inequity and stigma,” they said. The findings also suggest that biological mechanisms of SCT may contribute to severe clinical complications, and therefore “invite a more critical examination of the assumption that SCT is not a disease state.”
The study findings were limited by several factors including the lack of assessment of SCT independent of other comorbidities, such as hypertension, preeclampsia, diabetes, and obesity, and by the use of billing codes that could misclassify patients, the researchers noted.
However, the results support some findings from previous studies of the potential health complications for pregnant SCT patients. The large study population highlights the need to identify women’s SCT status during obstetric care, and to provide both pregnancy guidance for SCT patients and systemic support of comprehensive care for SCD and SCT patients, they concluded.
Disparities may drive stillbirth in sickle cell trait women
“There is a paucity of research evaluating sickle cell trait and the risk of adverse pregnancy outcomes such as stillbirth,” Iris Krishna, MD, of Emory University, Atlanta, said in an interview. “Prior studies evaluating the risk of stillbirth have yielded mixed results, and an increased risk of stillbirth in women with sickle cell trait has not been established. This study is unique in that it attempts to address how racial inequities and health disparities may contribute to risk of stillbirth in women with sickle cell trait.”
Although the study findings suggest an increased risk of stillbirth in women with sickle cell trait, an analysis stratified for Black or African American patients showed no association, Dr. Krishna said. “The prevalence of stillbirth was noted to be 1% among Black or African American patients with sickle cell trait compared to the prevalence of stillbirth of 1.1% among Black or African American women with no sickle cell trait or disease. Although, sickle cell trait or sickle cell disease can be found in any racial or ethnic group, it disproportionately affects Black or African Americans, with a sickle cell trait carrier rate of approximately 1 in 10. The mixed findings in this study amongst racial/ethnic groups further suggest that there is more research needed before an association between stillbirth and sickle cell trait can be supported.”
As for clinical implications, “it is well established that for women with sickle cell trait there is an increased risk of urinary tract infections in pregnancy,” said Dr. Krishna. “Women with sickle cell trait should have a urine culture performed at their first prenatal visit and each trimester. At this time, studies evaluating risk of stillbirth in women with sickle cell trait have yielded conflicting results, and current consensus is that women with sickle cell trait are not at increased risk. In comparison, women with sickle cell disease are at increased risk for stillbirth and adverse pregnancy outcomes. Women with sickle cell disease should be followed closely during pregnancy and fetal surveillance implemented at 32 weeks, if not sooner, to reduce risk of stillbirth.
“Prior studies evaluating risk of stillbirth in women with sickle cell trait consist of retrospective cohorts with small study populations,” Dr. Krishna added. Notably, the current study was limited by the inability to adjust for comorbidities including diabetes, hypertension, and obesity, that are not only associated with an increased risk for stillbirth, but also disproportionately common among Black women.
“More studies are needed evaluating the relationship between these comorbidities as well as studies specifically evaluating how race affects care and pregnancy outcomes,” Dr. Krisha emphasized.
The study was funded by the University of Pennsylvania department of biostatistics, epidemiology, and informatics. Lead author Dr. Canelón disclosed grants from the Centers for Disease Control and Prevention, Clinical and Translational Science Awards, and grants from the National Institutes of Health outside the submitted work. Dr. Krishna had no financial conflicts to disclose, but serves on the editorial advisory board of Ob.Gyn News.
Both sickle cell trait and sickle cell disease were significantly associated with an increased risk of stillbirth, based on data from more than 50,000 women.
Pregnant women with sickle cell disease (SCD) are at increased risk of complications, including stillbirth, but many women with the disease in the United States lack access to specialty care, Silvia P. Canelón, PhD, of the University of Pennsylvania, Philadelphia, and colleagues wrote. Sickle cell trait (SCT), defined as one abnormal allele of the hemoglobin gene, is not considered a disease state because many carriers are asymptomatic, and therefore even less likely to be assessed for potential complications. “However, it is possible for people with SCT to experience sickling of red blood cells under severe hypoxia, dehydration, and hyperthermia. This condition can lead to severe medical complications for sickle cell carriers, including fetal loss, splenic infarction, exercise-related sudden death, and others,” they noted.
In a study published in JAMA Network Open, the researchers reviewed data from 63,334 deliveries in 50,560 women between Jan. 1, 2010, and Aug. 15, 2017, at four quaternary academic medical centers in Pennsylvania. Of these, 1,904 had SCT but not SCD, and 164 had SCD. The mean age of the women was 29.5 years, and approximately 56% were single at the time of delivery. A majority (87%) of the study population was Rhesus-factor positive, 47.0% were Black or African American, 33.7% were White, and 45.2% had ABO blood type O.
Risk factors for stillbirth used in the analysis included SCD, numbers of pain crises and blood transfusions before delivery, delivery episode (to represent parity), history of cesarean delivery, multiple gestation, age, marital status, race and ethnicity, ABO blood type, Rhesus factor, and year of delivery.
Overall, the prevalence of stillbirth in women with SCT was 1.1%, compared with 0.8% in the general study population, and was significantly associated with increased risk of stillbirth after controlling for multiple risk factors. The adjusted odds ratio was 8.94 for stillbirth risk in women with SCT, compared with women without SCT (P = .045), although the risk was greater among women with SCD, compared with those without SCD (aOR, 26.40).
“In addition, the stratified analysis found Black or African American patients with SCD to be at higher risk of stillbirth, compared with Black or African American patients without SCD (aOR, 3.59),” but no significant association was noted between stillbirth and SCT, the researchers wrote. Stillbirth rates were 1.1% in Black or African American women overall, 2.7% in those with SCD, and 1.0% in those with SCT. Overall, multiple gestation was associated with an increased risk of stillbirth (aOR, 4.68), while a history of cesarean delivery and being married at the time of delivery were associated with decreased risk (aOR, 0.44 and 0.72, respectively).
The lack of association between stillbirth and SCT in Black or African American patients supports some previous research, but contradicts other studies, the researchers wrote. “Ultimately, it may be impossible to disentangle the risks due to the disease and those due to disparities associated with the disease that have resulted from longstanding inequity and stigma,” they said. The findings also suggest that biological mechanisms of SCT may contribute to severe clinical complications, and therefore “invite a more critical examination of the assumption that SCT is not a disease state.”
The study findings were limited by several factors including the lack of assessment of SCT independent of other comorbidities, such as hypertension, preeclampsia, diabetes, and obesity, and by the use of billing codes that could misclassify patients, the researchers noted.
However, the results support some findings from previous studies of the potential health complications for pregnant SCT patients. The large study population highlights the need to identify women’s SCT status during obstetric care, and to provide both pregnancy guidance for SCT patients and systemic support of comprehensive care for SCD and SCT patients, they concluded.
Disparities may drive stillbirth in sickle cell trait women
“There is a paucity of research evaluating sickle cell trait and the risk of adverse pregnancy outcomes such as stillbirth,” Iris Krishna, MD, of Emory University, Atlanta, said in an interview. “Prior studies evaluating the risk of stillbirth have yielded mixed results, and an increased risk of stillbirth in women with sickle cell trait has not been established. This study is unique in that it attempts to address how racial inequities and health disparities may contribute to risk of stillbirth in women with sickle cell trait.”
Although the study findings suggest an increased risk of stillbirth in women with sickle cell trait, an analysis stratified for Black or African American patients showed no association, Dr. Krishna said. “The prevalence of stillbirth was noted to be 1% among Black or African American patients with sickle cell trait compared to the prevalence of stillbirth of 1.1% among Black or African American women with no sickle cell trait or disease. Although, sickle cell trait or sickle cell disease can be found in any racial or ethnic group, it disproportionately affects Black or African Americans, with a sickle cell trait carrier rate of approximately 1 in 10. The mixed findings in this study amongst racial/ethnic groups further suggest that there is more research needed before an association between stillbirth and sickle cell trait can be supported.”
As for clinical implications, “it is well established that for women with sickle cell trait there is an increased risk of urinary tract infections in pregnancy,” said Dr. Krishna. “Women with sickle cell trait should have a urine culture performed at their first prenatal visit and each trimester. At this time, studies evaluating risk of stillbirth in women with sickle cell trait have yielded conflicting results, and current consensus is that women with sickle cell trait are not at increased risk. In comparison, women with sickle cell disease are at increased risk for stillbirth and adverse pregnancy outcomes. Women with sickle cell disease should be followed closely during pregnancy and fetal surveillance implemented at 32 weeks, if not sooner, to reduce risk of stillbirth.
“Prior studies evaluating risk of stillbirth in women with sickle cell trait consist of retrospective cohorts with small study populations,” Dr. Krishna added. Notably, the current study was limited by the inability to adjust for comorbidities including diabetes, hypertension, and obesity, that are not only associated with an increased risk for stillbirth, but also disproportionately common among Black women.
“More studies are needed evaluating the relationship between these comorbidities as well as studies specifically evaluating how race affects care and pregnancy outcomes,” Dr. Krisha emphasized.
The study was funded by the University of Pennsylvania department of biostatistics, epidemiology, and informatics. Lead author Dr. Canelón disclosed grants from the Centers for Disease Control and Prevention, Clinical and Translational Science Awards, and grants from the National Institutes of Health outside the submitted work. Dr. Krishna had no financial conflicts to disclose, but serves on the editorial advisory board of Ob.Gyn News.
FROM JAMA NETWORK OPEN
Ulcerative colitis: Donor-derived strains predict response in FMT
The Odoribacter splanchnicus strain of human donor-derived bacteria correlated with clinical response to ulcerative colitis in study in which mouse models were colonized with patient-derived strains.
Although some recent trials have shown the effectiveness of fecal microbiota transplantation (FMT) for patients with ulcerative colitis (UC), the current process is limited by the used of crude donor fecal material, which increases the risk of infection and decreases potential effectiveness, Svetlana Lima, MD, of Weill Cornell Medicine, New York, and colleagues wrote.
“Rational selection and production of specific microbial strains or communities could improve efficacy, minimize the risk of adverse reactions as well as increase the acceptance of microbiome-based therapies,” the researchers wrote.
In a study published in Gastroenterology, the researchers used metagenomic analysis and IgA sequencing (for sorting and sequencing IgA-coated microbiota) to identify a core of transferable and IgA-coated microbiota. They conducted metagenomic sequencing on 60 stool samples, including 20 recipient-participants with active UC who were treated with FMT, and another 20 FMT recipients with data from 4 weeks after FMT from a previously reported trial.
The core transferable microbiota (CTM) included 22 species of bacteria at 4 weeks after FMT. To determine a relationship between CTM and clinical response to FMT, the researchers defined clinical response as a decrease in Mayo score of 3 or greater with a rectal bleeding score of 1 or less by 4 weeks after FMT; 35% of study participants met this endpoint.* A total of 20 species were unique to the responders. “Of the donor-derived genera, only the relative abundance of Odoribacter at [week 4] post FMT and its increase post FMT was found to significantly correlate with decrease in Mayo score,” the researchers noted.
The researchers then colonized germ-free or genetically engineered mice with patient-derived bacterial strains.
O. splanchnicus also increased induction of interleukin-10, and increased the production of short-chain fatty acids. Taken together, these factors allowed for O. splanchnicus to limit colitis in the mice.
The study findings represent the first strain-level analysis of FMT in UC participants, and define a transferable microbiota associated with clinical response that could serve as a prognostic biomarker, the researchers noted in their discussion section. Although analysis revealed 12 donor-derived bacterial species that predicted clinical response, further IgA analysis identified O. splanchnicus as “the only microbe within the responders core that correlates with clinical response and highlights the potential impact of this taxa seen in independent cohorts, as well as mouse models of colitis and colorectal cancer,” the researchers emphasized.
The study findings were limited by the small sample size and the lack of prospective data. However, “collectively, this work provides the first evidence of transferable, donor-derived strains that correlate with clinical response to FMT in UC and reveals O. splanchnicus as a key component, which mechanistically promotes protection through both cellular and metabolic function,” the researchers said. “These mechanistic features will help enable desperately needed strategies to enhance therapeutic efficacy of microbial therapy for UC.”
Study strains improve effectiveness
“There is an accumulating body of evidence that suggests that gut dysbiosis, or the imbalance between good and bad microbes, plays an important role in the pathogenesis and progression of ulcerative colitis,” Jeffrey Berinstein, MD, of the University of Michigan, Ann Arbor, said in an interview. “It is for this reason that therapeutic manipulation of gut microbiota with fecal microbiota transplant is being explored as a potential treatment option for UC. FMT has demonstrated promise for ulcerative colitis, however little is known about the specific microbiota strains contributing to this observed improvement. In this study, the authors aimed to better understand the mechanisms and the specific strains in FMT contributing to this observed improvement, which is an important step toward improving efficacy and minimizing the risk of adverse events related to FMT in the future.”
Dr. Berinstein was surprised that O. splanchnicus was the only microbe identified that correlated with clinical response. “Previous studies have suggested that microbial diversity is a key factor in successful response to FMT,” he noted. “FMT remains an important potential nonpharmacologic treatment strategy for ulcerative colitis, however more research is needed to understand the mechanism and to develop safer and more efficacious methods for delivering FMT.” Specifically, prospective studies are needed to explore the efficacy and safety of FMT enriched in strains of O. splanchnicus to confirm the current study findings.
The current study is important at this time because, although microbial transferability has emerged as a potential to treat IBD, “the mechanistic understanding of microbial transferability and engraftment has been lacking,” Atsushi Sakuraba, MD, PhD of the University of Chicago, said in an interview. “I was surprised that the effectiveness of FMT in UC could be narrowed down to O. splanchnicus.” The current take-home message for clinicians is that, although FMT currently uses crude donor fecal material, it may soon use more selected microbial strains. However, “whether transfer of O. splanchnicus alone or enriched fecal material provide improved efficacy and safety need to be analyzed,” he added.
The study was supported by Boehringer Ingelheim, the National Institutes of Health, the Kenneth Rainin Foundation, and the Charina Foundation. One coauthor disclosed grant support from Boehringer Ingelheim for this study, and several coauthors are employees of Boehringer Ingelheim. Neither Dr. Berinstein nor Dr. Sakuraba had no financial conflicts to disclose.
This article was updated Dec. 1, 2021.
*Correction, 4/11/22: An earlier version of this article misstated the definition of clinical response.
The Odoribacter splanchnicus strain of human donor-derived bacteria correlated with clinical response to ulcerative colitis in study in which mouse models were colonized with patient-derived strains.
Although some recent trials have shown the effectiveness of fecal microbiota transplantation (FMT) for patients with ulcerative colitis (UC), the current process is limited by the used of crude donor fecal material, which increases the risk of infection and decreases potential effectiveness, Svetlana Lima, MD, of Weill Cornell Medicine, New York, and colleagues wrote.
“Rational selection and production of specific microbial strains or communities could improve efficacy, minimize the risk of adverse reactions as well as increase the acceptance of microbiome-based therapies,” the researchers wrote.
In a study published in Gastroenterology, the researchers used metagenomic analysis and IgA sequencing (for sorting and sequencing IgA-coated microbiota) to identify a core of transferable and IgA-coated microbiota. They conducted metagenomic sequencing on 60 stool samples, including 20 recipient-participants with active UC who were treated with FMT, and another 20 FMT recipients with data from 4 weeks after FMT from a previously reported trial.
The core transferable microbiota (CTM) included 22 species of bacteria at 4 weeks after FMT. To determine a relationship between CTM and clinical response to FMT, the researchers defined clinical response as a decrease in Mayo score of 3 or greater with a rectal bleeding score of 1 or less by 4 weeks after FMT; 35% of study participants met this endpoint.* A total of 20 species were unique to the responders. “Of the donor-derived genera, only the relative abundance of Odoribacter at [week 4] post FMT and its increase post FMT was found to significantly correlate with decrease in Mayo score,” the researchers noted.
The researchers then colonized germ-free or genetically engineered mice with patient-derived bacterial strains.
O. splanchnicus also increased induction of interleukin-10, and increased the production of short-chain fatty acids. Taken together, these factors allowed for O. splanchnicus to limit colitis in the mice.
The study findings represent the first strain-level analysis of FMT in UC participants, and define a transferable microbiota associated with clinical response that could serve as a prognostic biomarker, the researchers noted in their discussion section. Although analysis revealed 12 donor-derived bacterial species that predicted clinical response, further IgA analysis identified O. splanchnicus as “the only microbe within the responders core that correlates with clinical response and highlights the potential impact of this taxa seen in independent cohorts, as well as mouse models of colitis and colorectal cancer,” the researchers emphasized.
The study findings were limited by the small sample size and the lack of prospective data. However, “collectively, this work provides the first evidence of transferable, donor-derived strains that correlate with clinical response to FMT in UC and reveals O. splanchnicus as a key component, which mechanistically promotes protection through both cellular and metabolic function,” the researchers said. “These mechanistic features will help enable desperately needed strategies to enhance therapeutic efficacy of microbial therapy for UC.”
Study strains improve effectiveness
“There is an accumulating body of evidence that suggests that gut dysbiosis, or the imbalance between good and bad microbes, plays an important role in the pathogenesis and progression of ulcerative colitis,” Jeffrey Berinstein, MD, of the University of Michigan, Ann Arbor, said in an interview. “It is for this reason that therapeutic manipulation of gut microbiota with fecal microbiota transplant is being explored as a potential treatment option for UC. FMT has demonstrated promise for ulcerative colitis, however little is known about the specific microbiota strains contributing to this observed improvement. In this study, the authors aimed to better understand the mechanisms and the specific strains in FMT contributing to this observed improvement, which is an important step toward improving efficacy and minimizing the risk of adverse events related to FMT in the future.”
Dr. Berinstein was surprised that O. splanchnicus was the only microbe identified that correlated with clinical response. “Previous studies have suggested that microbial diversity is a key factor in successful response to FMT,” he noted. “FMT remains an important potential nonpharmacologic treatment strategy for ulcerative colitis, however more research is needed to understand the mechanism and to develop safer and more efficacious methods for delivering FMT.” Specifically, prospective studies are needed to explore the efficacy and safety of FMT enriched in strains of O. splanchnicus to confirm the current study findings.
The current study is important at this time because, although microbial transferability has emerged as a potential to treat IBD, “the mechanistic understanding of microbial transferability and engraftment has been lacking,” Atsushi Sakuraba, MD, PhD of the University of Chicago, said in an interview. “I was surprised that the effectiveness of FMT in UC could be narrowed down to O. splanchnicus.” The current take-home message for clinicians is that, although FMT currently uses crude donor fecal material, it may soon use more selected microbial strains. However, “whether transfer of O. splanchnicus alone or enriched fecal material provide improved efficacy and safety need to be analyzed,” he added.
The study was supported by Boehringer Ingelheim, the National Institutes of Health, the Kenneth Rainin Foundation, and the Charina Foundation. One coauthor disclosed grant support from Boehringer Ingelheim for this study, and several coauthors are employees of Boehringer Ingelheim. Neither Dr. Berinstein nor Dr. Sakuraba had no financial conflicts to disclose.
This article was updated Dec. 1, 2021.
*Correction, 4/11/22: An earlier version of this article misstated the definition of clinical response.
The Odoribacter splanchnicus strain of human donor-derived bacteria correlated with clinical response to ulcerative colitis in study in which mouse models were colonized with patient-derived strains.
Although some recent trials have shown the effectiveness of fecal microbiota transplantation (FMT) for patients with ulcerative colitis (UC), the current process is limited by the used of crude donor fecal material, which increases the risk of infection and decreases potential effectiveness, Svetlana Lima, MD, of Weill Cornell Medicine, New York, and colleagues wrote.
“Rational selection and production of specific microbial strains or communities could improve efficacy, minimize the risk of adverse reactions as well as increase the acceptance of microbiome-based therapies,” the researchers wrote.
In a study published in Gastroenterology, the researchers used metagenomic analysis and IgA sequencing (for sorting and sequencing IgA-coated microbiota) to identify a core of transferable and IgA-coated microbiota. They conducted metagenomic sequencing on 60 stool samples, including 20 recipient-participants with active UC who were treated with FMT, and another 20 FMT recipients with data from 4 weeks after FMT from a previously reported trial.
The core transferable microbiota (CTM) included 22 species of bacteria at 4 weeks after FMT. To determine a relationship between CTM and clinical response to FMT, the researchers defined clinical response as a decrease in Mayo score of 3 or greater with a rectal bleeding score of 1 or less by 4 weeks after FMT; 35% of study participants met this endpoint.* A total of 20 species were unique to the responders. “Of the donor-derived genera, only the relative abundance of Odoribacter at [week 4] post FMT and its increase post FMT was found to significantly correlate with decrease in Mayo score,” the researchers noted.
The researchers then colonized germ-free or genetically engineered mice with patient-derived bacterial strains.
O. splanchnicus also increased induction of interleukin-10, and increased the production of short-chain fatty acids. Taken together, these factors allowed for O. splanchnicus to limit colitis in the mice.
The study findings represent the first strain-level analysis of FMT in UC participants, and define a transferable microbiota associated with clinical response that could serve as a prognostic biomarker, the researchers noted in their discussion section. Although analysis revealed 12 donor-derived bacterial species that predicted clinical response, further IgA analysis identified O. splanchnicus as “the only microbe within the responders core that correlates with clinical response and highlights the potential impact of this taxa seen in independent cohorts, as well as mouse models of colitis and colorectal cancer,” the researchers emphasized.
The study findings were limited by the small sample size and the lack of prospective data. However, “collectively, this work provides the first evidence of transferable, donor-derived strains that correlate with clinical response to FMT in UC and reveals O. splanchnicus as a key component, which mechanistically promotes protection through both cellular and metabolic function,” the researchers said. “These mechanistic features will help enable desperately needed strategies to enhance therapeutic efficacy of microbial therapy for UC.”
Study strains improve effectiveness
“There is an accumulating body of evidence that suggests that gut dysbiosis, or the imbalance between good and bad microbes, plays an important role in the pathogenesis and progression of ulcerative colitis,” Jeffrey Berinstein, MD, of the University of Michigan, Ann Arbor, said in an interview. “It is for this reason that therapeutic manipulation of gut microbiota with fecal microbiota transplant is being explored as a potential treatment option for UC. FMT has demonstrated promise for ulcerative colitis, however little is known about the specific microbiota strains contributing to this observed improvement. In this study, the authors aimed to better understand the mechanisms and the specific strains in FMT contributing to this observed improvement, which is an important step toward improving efficacy and minimizing the risk of adverse events related to FMT in the future.”
Dr. Berinstein was surprised that O. splanchnicus was the only microbe identified that correlated with clinical response. “Previous studies have suggested that microbial diversity is a key factor in successful response to FMT,” he noted. “FMT remains an important potential nonpharmacologic treatment strategy for ulcerative colitis, however more research is needed to understand the mechanism and to develop safer and more efficacious methods for delivering FMT.” Specifically, prospective studies are needed to explore the efficacy and safety of FMT enriched in strains of O. splanchnicus to confirm the current study findings.
The current study is important at this time because, although microbial transferability has emerged as a potential to treat IBD, “the mechanistic understanding of microbial transferability and engraftment has been lacking,” Atsushi Sakuraba, MD, PhD of the University of Chicago, said in an interview. “I was surprised that the effectiveness of FMT in UC could be narrowed down to O. splanchnicus.” The current take-home message for clinicians is that, although FMT currently uses crude donor fecal material, it may soon use more selected microbial strains. However, “whether transfer of O. splanchnicus alone or enriched fecal material provide improved efficacy and safety need to be analyzed,” he added.
The study was supported by Boehringer Ingelheim, the National Institutes of Health, the Kenneth Rainin Foundation, and the Charina Foundation. One coauthor disclosed grant support from Boehringer Ingelheim for this study, and several coauthors are employees of Boehringer Ingelheim. Neither Dr. Berinstein nor Dr. Sakuraba had no financial conflicts to disclose.
This article was updated Dec. 1, 2021.
*Correction, 4/11/22: An earlier version of this article misstated the definition of clinical response.
FROM GASTROENTEROLOGY
High-poverty areas host more firearm-related youth deaths
Higher poverty concentration at the county level significantly increased the risk of firearm-related deaths in children and youth aged 5-24 years in the United States, based on a review of approximately 67,000 fatalities.
Firearms are the second-leading cause of death in children and young adults in the United States, according to data from the Centers for Disease Control and Prevention, wrote Jefferson T. Barrett, MD, of The Children’s Hospital at Montefiore, New York, and colleagues. County-level poverty has been associated with increased injury mortality in children, but the association between county-level poverty and firearm-related mortality in particular has not been well studied.
In a cross-sectional study published in JAMA Pediatrics, 67,905 firearm-related deaths in children and youth aged 5-24 years that occurred between Jan. 1, 2007, and Dec. 31, 2016 were analyzed. The deaths included 42,512 homicides (62.6%), 23,034 suicides (33.9%), and 1,627 unintentional deaths (2.4%).
County poverty data were acquired from the U.S. Census Bureau. County-level poverty was divided into five categories based on percentage of the population living below the federal poverty level: 0%-4.9%, 5%-9.9%, 10%-14.9%, 15%-19.9%, and 20% or more.
Overall, 88.6% of the total deaths were in males. Notably, 44.8% of total firearm-related deaths and 63.9% of homicides occurred in non-Hispanic Blacks, who make up only 14% of the youth population in the United States, the researchers wrote.
The total number of firearm-related deaths was 248 in the lowest quintile of poverty concentration, followed by 6,841, 18,551, 27,305, and 14,960 in the remaining quintiles.
In a multivariate regression model that included demographics, urban versus rural, and statewide firearm prevalence, youth in counties with the highest quintile of poverty concentration had an increased rate of total firearm-related deaths (adjusted incidence rate ratio, 2.29), as well as increased rates of homicides, suicides, and unintentional deaths (aIRR, 3.55, 1.45, and 9.32, respectively), compared with those living in the lowest quintile of poverty concentration. Individuals in the highest poverty quintile accounted for 22.0% of total firearm-related deaths, 25.5% of homicides, 15.3% of suicides, and 25.1% of unintentional deaths.
The researchers also calculated the population-attributable fraction (PAF) and years of potential life lost. “The PAF represents the proportion of deaths associated with a particular exposure, which was concentrated county poverty in this study,” they explained. The PAF for all firearm-related deaths was 0.51, PAFs for homicides, suicides, and unintentional deaths were 0.66, 0.30, and 0.86, respectively. The PAF calculation translated to 34,292 firearm-related deaths that may not have occurred if youth in all counties had the same risk as those in counties with the lowest poverty concentration.
“Over the 10-year study period, we observed 3,833,105 years of potential life lost in youth aged 5-24 years from firearm-related deaths,” the researchers wrote.
The study findings were limited by several factors including the potential bias of a cross-section design, and inability to account for all the ways that county-level poverty might increase the risk of firearm-related death in children and teens, the researchers noted. Other potential limitations include possible misclassification of death, lack of data on individual family incomes, shifts in counties in the poverty categories over time, and the use of statewide, rather than countywide, estimates of firearm ownership.
However, the results are consistent with those of previous studies, and add that “mortality rates were consistent even after controlling for demographic variables, county urbanicity, and statewide firearm prevalence,” the researchers concluded.
Address structural racism to reduce disparities
“Firearm-related homicides among youth aged 5-24 years are among the causes of death with the greatest disparities,” based on CDC fatal injury reports, wrote Alice M. Ellyson, PhD, Frederick P. Rivara, MD, and Ali Rowhani-Rahbar, MD, all of the University of Washington, Seattle, in an accompanying editorial.
The current study builds on previous research, including studies showing an association between income inequality and firearm-related homicide, they said. More research is needed to determine how to intervene in the pathways between poverty and firearm-related death. For example, if access to high-quality health care is a factor, programs to increase access to health insurance, such as the Affordable Care Act and Children’s Health Insurance Program, or to increase access to high-quality trauma care may help reduce firearm-related death in youth.
“The study of where, how, and why racism operates as a factor in both poverty and firearm-related death must continue, especially considering the disparities consistently documented in Alaska Native or American Indian, Black, and Hispanic communities,” the editorialists wrote.
“Key potential mechanisms for reducing the consequences of poverty for firearm-related death are often denied to racial and ethnic minority groups through a variety of structures, policies, and systems in health care, employment, housing, transportation, and education,” they emphasized, and the impact of racism, not only on the pathways to poverty, but also on mediators between poverty and firearm-related death, must be explored.
Findings spotlight need to for poverty programs
The study was an interesting look at the specific relationship between poverty and firearm-related deaths in people aged younger than 25 years in the United States, Tim Joos, MD, of Seattle said in an interview.
“Although America is not a poor country, the combination of poverty within America and its unique gun culture seems to prove deadly for its youth,” Dr. Joos said. “The strongest relationship is between firearm-related homicide and poverty, but unintentional firearm deaths and poverty also are clearly linked, whereas the link between firearm-related suicide and poverty appears to be present, but small.”.
In the current study, “the authors note that firearm deaths are the second-leading cause of death among all people ages 15-24 years,” said Dr. Joos. “Many of us have followed children from infancy just to have them meet this untimely end as adolescents, wishing we had a vaccine or other remedy in our toolbelt for this particular scourge.
“As our country currently debates the size of the social safety net, this study is one of many that suggests government programs aimed at poverty alleviation would substantially contribute to the health of American youth,” Dr. Joos added.
The study received no outside funding. Lead author Dr. Barrett had no financial conflicts to disclose. Dr. Ellyson disclosed funds from the CDC, the state of Washington, and the Grandmothers Against Gun Violence Foundation for research outside the submitted work. Dr. Rivara disclosed funds from the National Institutes of Health, the State of Washington, and the National Collaborative on Gun Violence Research for research outside the submitted work. Dr. Rowhani-Rahbar disclosed funds from the CDC, National Institutes of Health, National Collaborative on Gun Violence Research, Fund for a Safer Future, and state of Washington for research outside the submitted work. Dr. Joos had no financial conflicts to disclose, but serves on the editorial advisory board of Pediatric News.
Higher poverty concentration at the county level significantly increased the risk of firearm-related deaths in children and youth aged 5-24 years in the United States, based on a review of approximately 67,000 fatalities.
Firearms are the second-leading cause of death in children and young adults in the United States, according to data from the Centers for Disease Control and Prevention, wrote Jefferson T. Barrett, MD, of The Children’s Hospital at Montefiore, New York, and colleagues. County-level poverty has been associated with increased injury mortality in children, but the association between county-level poverty and firearm-related mortality in particular has not been well studied.
In a cross-sectional study published in JAMA Pediatrics, 67,905 firearm-related deaths in children and youth aged 5-24 years that occurred between Jan. 1, 2007, and Dec. 31, 2016 were analyzed. The deaths included 42,512 homicides (62.6%), 23,034 suicides (33.9%), and 1,627 unintentional deaths (2.4%).
County poverty data were acquired from the U.S. Census Bureau. County-level poverty was divided into five categories based on percentage of the population living below the federal poverty level: 0%-4.9%, 5%-9.9%, 10%-14.9%, 15%-19.9%, and 20% or more.
Overall, 88.6% of the total deaths were in males. Notably, 44.8% of total firearm-related deaths and 63.9% of homicides occurred in non-Hispanic Blacks, who make up only 14% of the youth population in the United States, the researchers wrote.
The total number of firearm-related deaths was 248 in the lowest quintile of poverty concentration, followed by 6,841, 18,551, 27,305, and 14,960 in the remaining quintiles.
In a multivariate regression model that included demographics, urban versus rural, and statewide firearm prevalence, youth in counties with the highest quintile of poverty concentration had an increased rate of total firearm-related deaths (adjusted incidence rate ratio, 2.29), as well as increased rates of homicides, suicides, and unintentional deaths (aIRR, 3.55, 1.45, and 9.32, respectively), compared with those living in the lowest quintile of poverty concentration. Individuals in the highest poverty quintile accounted for 22.0% of total firearm-related deaths, 25.5% of homicides, 15.3% of suicides, and 25.1% of unintentional deaths.
The researchers also calculated the population-attributable fraction (PAF) and years of potential life lost. “The PAF represents the proportion of deaths associated with a particular exposure, which was concentrated county poverty in this study,” they explained. The PAF for all firearm-related deaths was 0.51, PAFs for homicides, suicides, and unintentional deaths were 0.66, 0.30, and 0.86, respectively. The PAF calculation translated to 34,292 firearm-related deaths that may not have occurred if youth in all counties had the same risk as those in counties with the lowest poverty concentration.
“Over the 10-year study period, we observed 3,833,105 years of potential life lost in youth aged 5-24 years from firearm-related deaths,” the researchers wrote.
The study findings were limited by several factors including the potential bias of a cross-section design, and inability to account for all the ways that county-level poverty might increase the risk of firearm-related death in children and teens, the researchers noted. Other potential limitations include possible misclassification of death, lack of data on individual family incomes, shifts in counties in the poverty categories over time, and the use of statewide, rather than countywide, estimates of firearm ownership.
However, the results are consistent with those of previous studies, and add that “mortality rates were consistent even after controlling for demographic variables, county urbanicity, and statewide firearm prevalence,” the researchers concluded.
Address structural racism to reduce disparities
“Firearm-related homicides among youth aged 5-24 years are among the causes of death with the greatest disparities,” based on CDC fatal injury reports, wrote Alice M. Ellyson, PhD, Frederick P. Rivara, MD, and Ali Rowhani-Rahbar, MD, all of the University of Washington, Seattle, in an accompanying editorial.
The current study builds on previous research, including studies showing an association between income inequality and firearm-related homicide, they said. More research is needed to determine how to intervene in the pathways between poverty and firearm-related death. For example, if access to high-quality health care is a factor, programs to increase access to health insurance, such as the Affordable Care Act and Children’s Health Insurance Program, or to increase access to high-quality trauma care may help reduce firearm-related death in youth.
“The study of where, how, and why racism operates as a factor in both poverty and firearm-related death must continue, especially considering the disparities consistently documented in Alaska Native or American Indian, Black, and Hispanic communities,” the editorialists wrote.
“Key potential mechanisms for reducing the consequences of poverty for firearm-related death are often denied to racial and ethnic minority groups through a variety of structures, policies, and systems in health care, employment, housing, transportation, and education,” they emphasized, and the impact of racism, not only on the pathways to poverty, but also on mediators between poverty and firearm-related death, must be explored.
Findings spotlight need to for poverty programs
The study was an interesting look at the specific relationship between poverty and firearm-related deaths in people aged younger than 25 years in the United States, Tim Joos, MD, of Seattle said in an interview.
“Although America is not a poor country, the combination of poverty within America and its unique gun culture seems to prove deadly for its youth,” Dr. Joos said. “The strongest relationship is between firearm-related homicide and poverty, but unintentional firearm deaths and poverty also are clearly linked, whereas the link between firearm-related suicide and poverty appears to be present, but small.”.
In the current study, “the authors note that firearm deaths are the second-leading cause of death among all people ages 15-24 years,” said Dr. Joos. “Many of us have followed children from infancy just to have them meet this untimely end as adolescents, wishing we had a vaccine or other remedy in our toolbelt for this particular scourge.
“As our country currently debates the size of the social safety net, this study is one of many that suggests government programs aimed at poverty alleviation would substantially contribute to the health of American youth,” Dr. Joos added.
The study received no outside funding. Lead author Dr. Barrett had no financial conflicts to disclose. Dr. Ellyson disclosed funds from the CDC, the state of Washington, and the Grandmothers Against Gun Violence Foundation for research outside the submitted work. Dr. Rivara disclosed funds from the National Institutes of Health, the State of Washington, and the National Collaborative on Gun Violence Research for research outside the submitted work. Dr. Rowhani-Rahbar disclosed funds from the CDC, National Institutes of Health, National Collaborative on Gun Violence Research, Fund for a Safer Future, and state of Washington for research outside the submitted work. Dr. Joos had no financial conflicts to disclose, but serves on the editorial advisory board of Pediatric News.
Higher poverty concentration at the county level significantly increased the risk of firearm-related deaths in children and youth aged 5-24 years in the United States, based on a review of approximately 67,000 fatalities.
Firearms are the second-leading cause of death in children and young adults in the United States, according to data from the Centers for Disease Control and Prevention, wrote Jefferson T. Barrett, MD, of The Children’s Hospital at Montefiore, New York, and colleagues. County-level poverty has been associated with increased injury mortality in children, but the association between county-level poverty and firearm-related mortality in particular has not been well studied.
In a cross-sectional study published in JAMA Pediatrics, 67,905 firearm-related deaths in children and youth aged 5-24 years that occurred between Jan. 1, 2007, and Dec. 31, 2016 were analyzed. The deaths included 42,512 homicides (62.6%), 23,034 suicides (33.9%), and 1,627 unintentional deaths (2.4%).
County poverty data were acquired from the U.S. Census Bureau. County-level poverty was divided into five categories based on percentage of the population living below the federal poverty level: 0%-4.9%, 5%-9.9%, 10%-14.9%, 15%-19.9%, and 20% or more.
Overall, 88.6% of the total deaths were in males. Notably, 44.8% of total firearm-related deaths and 63.9% of homicides occurred in non-Hispanic Blacks, who make up only 14% of the youth population in the United States, the researchers wrote.
The total number of firearm-related deaths was 248 in the lowest quintile of poverty concentration, followed by 6,841, 18,551, 27,305, and 14,960 in the remaining quintiles.
In a multivariate regression model that included demographics, urban versus rural, and statewide firearm prevalence, youth in counties with the highest quintile of poverty concentration had an increased rate of total firearm-related deaths (adjusted incidence rate ratio, 2.29), as well as increased rates of homicides, suicides, and unintentional deaths (aIRR, 3.55, 1.45, and 9.32, respectively), compared with those living in the lowest quintile of poverty concentration. Individuals in the highest poverty quintile accounted for 22.0% of total firearm-related deaths, 25.5% of homicides, 15.3% of suicides, and 25.1% of unintentional deaths.
The researchers also calculated the population-attributable fraction (PAF) and years of potential life lost. “The PAF represents the proportion of deaths associated with a particular exposure, which was concentrated county poverty in this study,” they explained. The PAF for all firearm-related deaths was 0.51, PAFs for homicides, suicides, and unintentional deaths were 0.66, 0.30, and 0.86, respectively. The PAF calculation translated to 34,292 firearm-related deaths that may not have occurred if youth in all counties had the same risk as those in counties with the lowest poverty concentration.
“Over the 10-year study period, we observed 3,833,105 years of potential life lost in youth aged 5-24 years from firearm-related deaths,” the researchers wrote.
The study findings were limited by several factors including the potential bias of a cross-section design, and inability to account for all the ways that county-level poverty might increase the risk of firearm-related death in children and teens, the researchers noted. Other potential limitations include possible misclassification of death, lack of data on individual family incomes, shifts in counties in the poverty categories over time, and the use of statewide, rather than countywide, estimates of firearm ownership.
However, the results are consistent with those of previous studies, and add that “mortality rates were consistent even after controlling for demographic variables, county urbanicity, and statewide firearm prevalence,” the researchers concluded.
Address structural racism to reduce disparities
“Firearm-related homicides among youth aged 5-24 years are among the causes of death with the greatest disparities,” based on CDC fatal injury reports, wrote Alice M. Ellyson, PhD, Frederick P. Rivara, MD, and Ali Rowhani-Rahbar, MD, all of the University of Washington, Seattle, in an accompanying editorial.
The current study builds on previous research, including studies showing an association between income inequality and firearm-related homicide, they said. More research is needed to determine how to intervene in the pathways between poverty and firearm-related death. For example, if access to high-quality health care is a factor, programs to increase access to health insurance, such as the Affordable Care Act and Children’s Health Insurance Program, or to increase access to high-quality trauma care may help reduce firearm-related death in youth.
“The study of where, how, and why racism operates as a factor in both poverty and firearm-related death must continue, especially considering the disparities consistently documented in Alaska Native or American Indian, Black, and Hispanic communities,” the editorialists wrote.
“Key potential mechanisms for reducing the consequences of poverty for firearm-related death are often denied to racial and ethnic minority groups through a variety of structures, policies, and systems in health care, employment, housing, transportation, and education,” they emphasized, and the impact of racism, not only on the pathways to poverty, but also on mediators between poverty and firearm-related death, must be explored.
Findings spotlight need to for poverty programs
The study was an interesting look at the specific relationship between poverty and firearm-related deaths in people aged younger than 25 years in the United States, Tim Joos, MD, of Seattle said in an interview.
“Although America is not a poor country, the combination of poverty within America and its unique gun culture seems to prove deadly for its youth,” Dr. Joos said. “The strongest relationship is between firearm-related homicide and poverty, but unintentional firearm deaths and poverty also are clearly linked, whereas the link between firearm-related suicide and poverty appears to be present, but small.”.
In the current study, “the authors note that firearm deaths are the second-leading cause of death among all people ages 15-24 years,” said Dr. Joos. “Many of us have followed children from infancy just to have them meet this untimely end as adolescents, wishing we had a vaccine or other remedy in our toolbelt for this particular scourge.
“As our country currently debates the size of the social safety net, this study is one of many that suggests government programs aimed at poverty alleviation would substantially contribute to the health of American youth,” Dr. Joos added.
The study received no outside funding. Lead author Dr. Barrett had no financial conflicts to disclose. Dr. Ellyson disclosed funds from the CDC, the state of Washington, and the Grandmothers Against Gun Violence Foundation for research outside the submitted work. Dr. Rivara disclosed funds from the National Institutes of Health, the State of Washington, and the National Collaborative on Gun Violence Research for research outside the submitted work. Dr. Rowhani-Rahbar disclosed funds from the CDC, National Institutes of Health, National Collaborative on Gun Violence Research, Fund for a Safer Future, and state of Washington for research outside the submitted work. Dr. Joos had no financial conflicts to disclose, but serves on the editorial advisory board of Pediatric News.
FROM JAMA PEDIATRICS
Social media use associated with depression in adults
Use of social media has been linked to increased anxiety and depression, as well as reduced well-being in adolescents and young adults, but similar associations in older adults have not been well studied, and longitudinal data are lacking, Ron H. Perlis, MD, of Massachusetts General Hospital, Boston, and colleagues wrote in their paper, which was published in JAMA Network Open.
To examine the association between social media use and depressive symptoms in older adults, the researchers reviewed data from 13 waves of an internet survey conducted each month between May 2020 and May 2021. The survey respondents included individuals aged 18 years and older, with a mean age of 56 years.
In the study the researchers analyzed responses from 5,395 individuals aged 18 years and older, with a mean age of 56 years. The study participants had minimal or no depressive symptoms at baseline, according to scores on the nine-item Patient Health Questionnaire (PHQ-9).
Overall, 8.9% of the respondents reported a worsening of 5 points or more on the PHQ-9 score on a follow-up survey, which was the primary outcome. Participants who reported using social media platforms Snapchat, Facebook, or TikTok were significantly more likely to report increased depressive symptoms, compared with those who did not report use of social media. The fully adjusted odds ratio was largest for Snapchat (aOR, 1.53), followed by Facebook (aOR, 1.42), and TikTok (aOR, 1.39).
Incorporating recent television and internet news terms, such as COVID-19, changed the association for Snapchat, for which the aOR decreased from 1.53 to 1.12 when news source terms were included in the survey. TikTok and Facebook associations remained similar.
When the results were further stratified by age, use of TikTok and Snapchat was associated with depressive symptoms in those aged 35 years and older, but not in those younger than 35 years. However, the opposite pattern emerged for Facebook; use was associated with depressive symptoms for individuals younger than 35 years, but not in those aged 35 years and older (aOR, 2.60 vs. aOR, 1.12).
The association between increased self-reported depressive symptoms and use of certain social media platforms was not impacted by baseline social support or face-to-face interactions, the researchers noted.
Family physician was surprised results weren’t more significant
In the current study, “I was honestly surprised the results weren’t more significant,” Mary Ann Dakkak, MD, of Boston University said in an interview. “That said, social media uses during the COVID pandemic may have been a necessary social outlet and form of connection for many people who were otherwise isolated.”
To still see a significant increase in depression when social media could have been a positive force may suggest a heavier impact during “normal” times, she added.
“It is not surprising that what we see in youth is shown among adults,” noted Dr. Dakkak, who was not involved with this study. “I always tell my patients that what is good for their children is good for the adults too, and vice versa.
“We expect to see outcomes of this on youth and adults who have been more isolated, who have used more screen time for learning, work, connection and boredom, in the near future,” she said. “The complex nature of why social media may have been used more heavily for connection during a time when in-person meetings were not possible may be a heavy confounder as the typical profile of heavy social media users may have differed during the COVID shutdowns.”
Psychiatrist: Balance benefits of social media with mental health risks
The current study was likely conducted before the recent news on “hidden” Facebook data and the implications that Facebook knew it was contributing to worsened mental health in teens, particularly around self-esteem, Jessica “Jessi” Gold, MD, a psychiatrist at Washington University, St. Louis, said in an interview.
“If you look more specifically at other studies, however, the data around social media and mental health is constantly varied, with some showing benefits and some showing negatives, and none conclusively suggesting either way,” said Dr. Gold, who also was not involved with the new research. “More data are needed, especially longitudinally and on a broader age group, to understand social media’s impact on mental health over time.
“It is also even more important in the wake of COVID-19, as so many people have turned to social media as a primary source of social support and connection, and are using it even more than before,” she emphasized.
In the current study, “I think the most interesting information is that, for TikTok and Snapchat, the effects seemed to be more pronounced in those older than 35 years who used social media,” said Dr. Gold.
What this study leaves unanswered is “whether people who might develop depression are simply more prone to use social media in the first place, such as to seek out social support,” Dr. Gold said. “Also, we don’t know anything about how long they are using social media or what they are using it for, which to me is important for understanding more about the nuance of the relationship with mental health and social media.”
Experts advise clinicians to discuss social media with patients
This new research suggests that clinicians should be talking to their patients about how social media impacts their emotional reactions, as well as their sleep, Dr. Gold said.
“Patients should be asking themselves how they are feeling when they are on social media and not using it before sleep. They should also be considering time limits and how to effectively use social media while taking care of their mental health,” she said. This conversation between clinician and patient should be had with any patient of any age, who uses social media, not only with teenagers.
“This is also a conversation about moderation, and knowing that individuals may feel they benefit from social media, that they should balance these benefits with potential mental health risks,” she said.
“Studies such as this one shed light onto why social media consumption should be at least a point of discussion with our patients,” said Dr. Dakkak.
She advised clinicians to ask and listen to patients and their families when it comes to screen time habits. “Whenever I see a patient with mood symptoms, I ask about their habits – eating, sleeping, socializing, screen time – including phone time. I ask about the family dynamics around screen time.
“I’ve added screen time to my adolescent assessment. Discussing safe use of cell phones and social media can have a significant impact on adolescent behavior and wellbeing, and parents are very thankful for the help,” she said. “This study encourages us to add screen time to the assessments we do at all adult ages, especially if mood symptoms exist,” Dr. Dakkak emphasized.
Suggestions for future research
Dr. Dakkak added that more areas for research include the differences in the impact of social media use on content creators versus content consumers. Also, “I would like to see research using the real data of use, the times of use, interruptions in sleep and use, possible confounding variables to include exercise, presence of intimate relationship and school/job performance.”
Given the many confounding variables, more controlled studies are needed to examine mental health outcomes in use, how long people use social media, and the impact of interventions such as time limits, Dr. Gold said.
“We can’t ignore the benefits of social media, such as helping those with social anxiety, finding peer support, and normalizing mental health, and those factors need to be studied and measured more effectively as well, she said.
Take-home message
It is important to recognize that the current study represents a correlation, not causality, said Dr. Gold. In addressing the issues of how social media impact mental health, “as always, the hardest thing is that many people get their news from social media, and often get social support from social media, so there has to be a balance of not removing social media completely, but of helping people see how it affects their mental health and how to find balance.”
The study findings were limited by several factors, including the inability to control for all potential confounders, the inability to assess the nature of social media use, and the lack of dose-response data, the researchers noted. Although the surveys in the current study were not specific to COVID-19, the effects of social media on depression may be specific to the content, and the findings may not generalize beyond the COVID-19 pandemic period.
Approximately two-thirds (66%) of the study participants identified as female, and 76% as White; 11% as Black; 6% as Asian; 5% as Hispanic; and 2% as American Indian or Alaska Native, Pacific Islander or Native Hawaiian, or other.
The National Institute of Mental Health provided a grant for the study to Dr. Pelis, who disclosed consulting fees from various companies and equity in Psy Therapeutics. The study’s lead author also serves as associate editor for JAMA Network Open, but was not involved in the decision process for publication of this study. Dr. Gold disclosed conducting a conference for Johnson & Johnson about social media and health care workers, and was on the advisory council.
Use of social media has been linked to increased anxiety and depression, as well as reduced well-being in adolescents and young adults, but similar associations in older adults have not been well studied, and longitudinal data are lacking, Ron H. Perlis, MD, of Massachusetts General Hospital, Boston, and colleagues wrote in their paper, which was published in JAMA Network Open.
To examine the association between social media use and depressive symptoms in older adults, the researchers reviewed data from 13 waves of an internet survey conducted each month between May 2020 and May 2021. The survey respondents included individuals aged 18 years and older, with a mean age of 56 years.
In the study the researchers analyzed responses from 5,395 individuals aged 18 years and older, with a mean age of 56 years. The study participants had minimal or no depressive symptoms at baseline, according to scores on the nine-item Patient Health Questionnaire (PHQ-9).
Overall, 8.9% of the respondents reported a worsening of 5 points or more on the PHQ-9 score on a follow-up survey, which was the primary outcome. Participants who reported using social media platforms Snapchat, Facebook, or TikTok were significantly more likely to report increased depressive symptoms, compared with those who did not report use of social media. The fully adjusted odds ratio was largest for Snapchat (aOR, 1.53), followed by Facebook (aOR, 1.42), and TikTok (aOR, 1.39).
Incorporating recent television and internet news terms, such as COVID-19, changed the association for Snapchat, for which the aOR decreased from 1.53 to 1.12 when news source terms were included in the survey. TikTok and Facebook associations remained similar.
When the results were further stratified by age, use of TikTok and Snapchat was associated with depressive symptoms in those aged 35 years and older, but not in those younger than 35 years. However, the opposite pattern emerged for Facebook; use was associated with depressive symptoms for individuals younger than 35 years, but not in those aged 35 years and older (aOR, 2.60 vs. aOR, 1.12).
The association between increased self-reported depressive symptoms and use of certain social media platforms was not impacted by baseline social support or face-to-face interactions, the researchers noted.
Family physician was surprised results weren’t more significant
In the current study, “I was honestly surprised the results weren’t more significant,” Mary Ann Dakkak, MD, of Boston University said in an interview. “That said, social media uses during the COVID pandemic may have been a necessary social outlet and form of connection for many people who were otherwise isolated.”
To still see a significant increase in depression when social media could have been a positive force may suggest a heavier impact during “normal” times, she added.
“It is not surprising that what we see in youth is shown among adults,” noted Dr. Dakkak, who was not involved with this study. “I always tell my patients that what is good for their children is good for the adults too, and vice versa.
“We expect to see outcomes of this on youth and adults who have been more isolated, who have used more screen time for learning, work, connection and boredom, in the near future,” she said. “The complex nature of why social media may have been used more heavily for connection during a time when in-person meetings were not possible may be a heavy confounder as the typical profile of heavy social media users may have differed during the COVID shutdowns.”
Psychiatrist: Balance benefits of social media with mental health risks
The current study was likely conducted before the recent news on “hidden” Facebook data and the implications that Facebook knew it was contributing to worsened mental health in teens, particularly around self-esteem, Jessica “Jessi” Gold, MD, a psychiatrist at Washington University, St. Louis, said in an interview.
“If you look more specifically at other studies, however, the data around social media and mental health is constantly varied, with some showing benefits and some showing negatives, and none conclusively suggesting either way,” said Dr. Gold, who also was not involved with the new research. “More data are needed, especially longitudinally and on a broader age group, to understand social media’s impact on mental health over time.
“It is also even more important in the wake of COVID-19, as so many people have turned to social media as a primary source of social support and connection, and are using it even more than before,” she emphasized.
In the current study, “I think the most interesting information is that, for TikTok and Snapchat, the effects seemed to be more pronounced in those older than 35 years who used social media,” said Dr. Gold.
What this study leaves unanswered is “whether people who might develop depression are simply more prone to use social media in the first place, such as to seek out social support,” Dr. Gold said. “Also, we don’t know anything about how long they are using social media or what they are using it for, which to me is important for understanding more about the nuance of the relationship with mental health and social media.”
Experts advise clinicians to discuss social media with patients
This new research suggests that clinicians should be talking to their patients about how social media impacts their emotional reactions, as well as their sleep, Dr. Gold said.
“Patients should be asking themselves how they are feeling when they are on social media and not using it before sleep. They should also be considering time limits and how to effectively use social media while taking care of their mental health,” she said. This conversation between clinician and patient should be had with any patient of any age, who uses social media, not only with teenagers.
“This is also a conversation about moderation, and knowing that individuals may feel they benefit from social media, that they should balance these benefits with potential mental health risks,” she said.
“Studies such as this one shed light onto why social media consumption should be at least a point of discussion with our patients,” said Dr. Dakkak.
She advised clinicians to ask and listen to patients and their families when it comes to screen time habits. “Whenever I see a patient with mood symptoms, I ask about their habits – eating, sleeping, socializing, screen time – including phone time. I ask about the family dynamics around screen time.
“I’ve added screen time to my adolescent assessment. Discussing safe use of cell phones and social media can have a significant impact on adolescent behavior and wellbeing, and parents are very thankful for the help,” she said. “This study encourages us to add screen time to the assessments we do at all adult ages, especially if mood symptoms exist,” Dr. Dakkak emphasized.
Suggestions for future research
Dr. Dakkak added that more areas for research include the differences in the impact of social media use on content creators versus content consumers. Also, “I would like to see research using the real data of use, the times of use, interruptions in sleep and use, possible confounding variables to include exercise, presence of intimate relationship and school/job performance.”
Given the many confounding variables, more controlled studies are needed to examine mental health outcomes in use, how long people use social media, and the impact of interventions such as time limits, Dr. Gold said.
“We can’t ignore the benefits of social media, such as helping those with social anxiety, finding peer support, and normalizing mental health, and those factors need to be studied and measured more effectively as well, she said.
Take-home message
It is important to recognize that the current study represents a correlation, not causality, said Dr. Gold. In addressing the issues of how social media impact mental health, “as always, the hardest thing is that many people get their news from social media, and often get social support from social media, so there has to be a balance of not removing social media completely, but of helping people see how it affects their mental health and how to find balance.”
The study findings were limited by several factors, including the inability to control for all potential confounders, the inability to assess the nature of social media use, and the lack of dose-response data, the researchers noted. Although the surveys in the current study were not specific to COVID-19, the effects of social media on depression may be specific to the content, and the findings may not generalize beyond the COVID-19 pandemic period.
Approximately two-thirds (66%) of the study participants identified as female, and 76% as White; 11% as Black; 6% as Asian; 5% as Hispanic; and 2% as American Indian or Alaska Native, Pacific Islander or Native Hawaiian, or other.
The National Institute of Mental Health provided a grant for the study to Dr. Pelis, who disclosed consulting fees from various companies and equity in Psy Therapeutics. The study’s lead author also serves as associate editor for JAMA Network Open, but was not involved in the decision process for publication of this study. Dr. Gold disclosed conducting a conference for Johnson & Johnson about social media and health care workers, and was on the advisory council.
Use of social media has been linked to increased anxiety and depression, as well as reduced well-being in adolescents and young adults, but similar associations in older adults have not been well studied, and longitudinal data are lacking, Ron H. Perlis, MD, of Massachusetts General Hospital, Boston, and colleagues wrote in their paper, which was published in JAMA Network Open.
To examine the association between social media use and depressive symptoms in older adults, the researchers reviewed data from 13 waves of an internet survey conducted each month between May 2020 and May 2021. The survey respondents included individuals aged 18 years and older, with a mean age of 56 years.
In the study the researchers analyzed responses from 5,395 individuals aged 18 years and older, with a mean age of 56 years. The study participants had minimal or no depressive symptoms at baseline, according to scores on the nine-item Patient Health Questionnaire (PHQ-9).
Overall, 8.9% of the respondents reported a worsening of 5 points or more on the PHQ-9 score on a follow-up survey, which was the primary outcome. Participants who reported using social media platforms Snapchat, Facebook, or TikTok were significantly more likely to report increased depressive symptoms, compared with those who did not report use of social media. The fully adjusted odds ratio was largest for Snapchat (aOR, 1.53), followed by Facebook (aOR, 1.42), and TikTok (aOR, 1.39).
Incorporating recent television and internet news terms, such as COVID-19, changed the association for Snapchat, for which the aOR decreased from 1.53 to 1.12 when news source terms were included in the survey. TikTok and Facebook associations remained similar.
When the results were further stratified by age, use of TikTok and Snapchat was associated with depressive symptoms in those aged 35 years and older, but not in those younger than 35 years. However, the opposite pattern emerged for Facebook; use was associated with depressive symptoms for individuals younger than 35 years, but not in those aged 35 years and older (aOR, 2.60 vs. aOR, 1.12).
The association between increased self-reported depressive symptoms and use of certain social media platforms was not impacted by baseline social support or face-to-face interactions, the researchers noted.
Family physician was surprised results weren’t more significant
In the current study, “I was honestly surprised the results weren’t more significant,” Mary Ann Dakkak, MD, of Boston University said in an interview. “That said, social media uses during the COVID pandemic may have been a necessary social outlet and form of connection for many people who were otherwise isolated.”
To still see a significant increase in depression when social media could have been a positive force may suggest a heavier impact during “normal” times, she added.
“It is not surprising that what we see in youth is shown among adults,” noted Dr. Dakkak, who was not involved with this study. “I always tell my patients that what is good for their children is good for the adults too, and vice versa.
“We expect to see outcomes of this on youth and adults who have been more isolated, who have used more screen time for learning, work, connection and boredom, in the near future,” she said. “The complex nature of why social media may have been used more heavily for connection during a time when in-person meetings were not possible may be a heavy confounder as the typical profile of heavy social media users may have differed during the COVID shutdowns.”
Psychiatrist: Balance benefits of social media with mental health risks
The current study was likely conducted before the recent news on “hidden” Facebook data and the implications that Facebook knew it was contributing to worsened mental health in teens, particularly around self-esteem, Jessica “Jessi” Gold, MD, a psychiatrist at Washington University, St. Louis, said in an interview.
“If you look more specifically at other studies, however, the data around social media and mental health is constantly varied, with some showing benefits and some showing negatives, and none conclusively suggesting either way,” said Dr. Gold, who also was not involved with the new research. “More data are needed, especially longitudinally and on a broader age group, to understand social media’s impact on mental health over time.
“It is also even more important in the wake of COVID-19, as so many people have turned to social media as a primary source of social support and connection, and are using it even more than before,” she emphasized.
In the current study, “I think the most interesting information is that, for TikTok and Snapchat, the effects seemed to be more pronounced in those older than 35 years who used social media,” said Dr. Gold.
What this study leaves unanswered is “whether people who might develop depression are simply more prone to use social media in the first place, such as to seek out social support,” Dr. Gold said. “Also, we don’t know anything about how long they are using social media or what they are using it for, which to me is important for understanding more about the nuance of the relationship with mental health and social media.”
Experts advise clinicians to discuss social media with patients
This new research suggests that clinicians should be talking to their patients about how social media impacts their emotional reactions, as well as their sleep, Dr. Gold said.
“Patients should be asking themselves how they are feeling when they are on social media and not using it before sleep. They should also be considering time limits and how to effectively use social media while taking care of their mental health,” she said. This conversation between clinician and patient should be had with any patient of any age, who uses social media, not only with teenagers.
“This is also a conversation about moderation, and knowing that individuals may feel they benefit from social media, that they should balance these benefits with potential mental health risks,” she said.
“Studies such as this one shed light onto why social media consumption should be at least a point of discussion with our patients,” said Dr. Dakkak.
She advised clinicians to ask and listen to patients and their families when it comes to screen time habits. “Whenever I see a patient with mood symptoms, I ask about their habits – eating, sleeping, socializing, screen time – including phone time. I ask about the family dynamics around screen time.
“I’ve added screen time to my adolescent assessment. Discussing safe use of cell phones and social media can have a significant impact on adolescent behavior and wellbeing, and parents are very thankful for the help,” she said. “This study encourages us to add screen time to the assessments we do at all adult ages, especially if mood symptoms exist,” Dr. Dakkak emphasized.
Suggestions for future research
Dr. Dakkak added that more areas for research include the differences in the impact of social media use on content creators versus content consumers. Also, “I would like to see research using the real data of use, the times of use, interruptions in sleep and use, possible confounding variables to include exercise, presence of intimate relationship and school/job performance.”
Given the many confounding variables, more controlled studies are needed to examine mental health outcomes in use, how long people use social media, and the impact of interventions such as time limits, Dr. Gold said.
“We can’t ignore the benefits of social media, such as helping those with social anxiety, finding peer support, and normalizing mental health, and those factors need to be studied and measured more effectively as well, she said.
Take-home message
It is important to recognize that the current study represents a correlation, not causality, said Dr. Gold. In addressing the issues of how social media impact mental health, “as always, the hardest thing is that many people get their news from social media, and often get social support from social media, so there has to be a balance of not removing social media completely, but of helping people see how it affects their mental health and how to find balance.”
The study findings were limited by several factors, including the inability to control for all potential confounders, the inability to assess the nature of social media use, and the lack of dose-response data, the researchers noted. Although the surveys in the current study were not specific to COVID-19, the effects of social media on depression may be specific to the content, and the findings may not generalize beyond the COVID-19 pandemic period.
Approximately two-thirds (66%) of the study participants identified as female, and 76% as White; 11% as Black; 6% as Asian; 5% as Hispanic; and 2% as American Indian or Alaska Native, Pacific Islander or Native Hawaiian, or other.
The National Institute of Mental Health provided a grant for the study to Dr. Pelis, who disclosed consulting fees from various companies and equity in Psy Therapeutics. The study’s lead author also serves as associate editor for JAMA Network Open, but was not involved in the decision process for publication of this study. Dr. Gold disclosed conducting a conference for Johnson & Johnson about social media and health care workers, and was on the advisory council.
FROM JAMA NETWORK OPEN
Microbiome studies among those awarded National Rosacea Society grants
A study on this year, as part of the organization’s research grants program.
The NRS research grants program was created to increase knowledge and understanding of not only the potential causes of rosacea, but other aspects of the disease that may inform prevention, treatment, or a potential cure, according to the press release announcing the recipients.
New research grant recipient Sezen Karakus, MD, of the Johns Hopkins Wilmer Eye Institute, Baltimore, received $15,000 for a study on the contribution of the ocular surface microbiome to the development of rosacea. Ocular rosacea can result in corneal complications severe enough to affect vision, and identifying the microorganisms on the ocular surface may lead to new treatment strategies, Dr. Karakus said in the release. He will collaborate on this research with dermatologist Noori Kim, MD, of Johns Hopkins University, Baltimore.
A second new research grant went to Emmanuel Contassot, MD, project leader in the dermatology department at of the University Hospital of Basel, Switzerland, who received $5,000 to investigate whether certain elevated intracellular signals in rosacea lesions may promote the skin inflammation that may be a root cause of the condition.
The NRS also renewed its support of a pair of ongoing studies. Michelle Trautwein, MD, of the Institute for Biodiversity Science and Sustainability at the California Academy of Sciences, continues her work on the first study to sequence the genome of Demodex mites; the study also identifies associated bacteria that may play a role in rosacea.
A second ongoing study by Tissa Hata, MD, of the University of California, San Diego, focuses on the normalization of the microbiome in people with rosacea. Dr. Hata’s work identifies types of bacteria associated with rosacea, as well as bacteria that may be associated with healthy skin after successful treatment of rosacea, including Cutibacterium acnes and Staphylococcus epidermidis.
The deadline to submit research proposals for next year’s grants is June 17, 2022. Researchers can find forms and instructions at the research grants section of the NRS website or by contacting the National Rosacea Society at 111 Lions Dr., Suite 216, Barrington, Ill., 60010, by telephone at 1-888-662-5874, or by email at [email protected].
A study on this year, as part of the organization’s research grants program.
The NRS research grants program was created to increase knowledge and understanding of not only the potential causes of rosacea, but other aspects of the disease that may inform prevention, treatment, or a potential cure, according to the press release announcing the recipients.
New research grant recipient Sezen Karakus, MD, of the Johns Hopkins Wilmer Eye Institute, Baltimore, received $15,000 for a study on the contribution of the ocular surface microbiome to the development of rosacea. Ocular rosacea can result in corneal complications severe enough to affect vision, and identifying the microorganisms on the ocular surface may lead to new treatment strategies, Dr. Karakus said in the release. He will collaborate on this research with dermatologist Noori Kim, MD, of Johns Hopkins University, Baltimore.
A second new research grant went to Emmanuel Contassot, MD, project leader in the dermatology department at of the University Hospital of Basel, Switzerland, who received $5,000 to investigate whether certain elevated intracellular signals in rosacea lesions may promote the skin inflammation that may be a root cause of the condition.
The NRS also renewed its support of a pair of ongoing studies. Michelle Trautwein, MD, of the Institute for Biodiversity Science and Sustainability at the California Academy of Sciences, continues her work on the first study to sequence the genome of Demodex mites; the study also identifies associated bacteria that may play a role in rosacea.
A second ongoing study by Tissa Hata, MD, of the University of California, San Diego, focuses on the normalization of the microbiome in people with rosacea. Dr. Hata’s work identifies types of bacteria associated with rosacea, as well as bacteria that may be associated with healthy skin after successful treatment of rosacea, including Cutibacterium acnes and Staphylococcus epidermidis.
The deadline to submit research proposals for next year’s grants is June 17, 2022. Researchers can find forms and instructions at the research grants section of the NRS website or by contacting the National Rosacea Society at 111 Lions Dr., Suite 216, Barrington, Ill., 60010, by telephone at 1-888-662-5874, or by email at [email protected].
A study on this year, as part of the organization’s research grants program.
The NRS research grants program was created to increase knowledge and understanding of not only the potential causes of rosacea, but other aspects of the disease that may inform prevention, treatment, or a potential cure, according to the press release announcing the recipients.
New research grant recipient Sezen Karakus, MD, of the Johns Hopkins Wilmer Eye Institute, Baltimore, received $15,000 for a study on the contribution of the ocular surface microbiome to the development of rosacea. Ocular rosacea can result in corneal complications severe enough to affect vision, and identifying the microorganisms on the ocular surface may lead to new treatment strategies, Dr. Karakus said in the release. He will collaborate on this research with dermatologist Noori Kim, MD, of Johns Hopkins University, Baltimore.
A second new research grant went to Emmanuel Contassot, MD, project leader in the dermatology department at of the University Hospital of Basel, Switzerland, who received $5,000 to investigate whether certain elevated intracellular signals in rosacea lesions may promote the skin inflammation that may be a root cause of the condition.
The NRS also renewed its support of a pair of ongoing studies. Michelle Trautwein, MD, of the Institute for Biodiversity Science and Sustainability at the California Academy of Sciences, continues her work on the first study to sequence the genome of Demodex mites; the study also identifies associated bacteria that may play a role in rosacea.
A second ongoing study by Tissa Hata, MD, of the University of California, San Diego, focuses on the normalization of the microbiome in people with rosacea. Dr. Hata’s work identifies types of bacteria associated with rosacea, as well as bacteria that may be associated with healthy skin after successful treatment of rosacea, including Cutibacterium acnes and Staphylococcus epidermidis.
The deadline to submit research proposals for next year’s grants is June 17, 2022. Researchers can find forms and instructions at the research grants section of the NRS website or by contacting the National Rosacea Society at 111 Lions Dr., Suite 216, Barrington, Ill., 60010, by telephone at 1-888-662-5874, or by email at [email protected].
Ferric carboxymaltose calms restless legs
Treatment with intravenous ferric carboxymaltose significantly improved symptoms in restless legs syndrome (RLS) patients with iron-deficiency anemia (IDA), data from 29 adults show.
RLS occurs among individuals with normal iron but is at least six times higher among individuals with IDA, Hyoeun Bae, MD, of Keimyung University, Daegu, South Korea, and colleagues wrote. Previous studies have explored iron treatments for RLS patients with IDA, however, guidelines for treatment have not yet been published.
In a study published in Sleep Medicine, the researchers randomized 29 RLS patients with IDA to either 1,500 mg IV ferric carboxymaltose (FCM) or placebo for a short-term period of 6 weeks, followed by a phase 2 study for responders that lasted for 52 weeks. Baseline characteristics, including age, gender, iron parameters, and sleep and mood scales were similar between the groups.
At 6 weeks, patients in the FCM group showed significant improvement in RLS symptom severity based on changes from baseline International Restless Legs Syndrome Study Group scale (IRLS) scores, compared with placebo patients (–13.47 vs. 1.36, P < .001). A secondary outcome of sleep quality also improved significantly in the FCM group, compared with the placebo group.
After 6 weeks, 11 of the 14 patients in the placebo group also received 1,500 mg FCM for an open-label study. These patients also showed significant improvement in IRLS scores from baseline to 6 weeks.
All 23 responders from the short-term studies (13 who received FCM initially and 10 from the postplacebo group) enrolled in a phase 2 long-term study that lasted for 52 weeks; 14 of these completed the full 52-week study period.
Overall, 61% of participants in phase 2 of the study remained off their RLS medications at 52 weeks, and no serious adverse events were reported during the study period. Of these, 10 received one additional dose of FCM and 4 received more than one additional dose. The median change in IRLS score at 4 weeks after treatment was –4.00, compared with the score prior to treatment.
The study is the first of its design to show benefits of intravenous iron therapy for RLS in patients with IDA, the researchers said, noting that the findings of improved, but not cured, RLS symptoms might suggest that more than 1,500 mg of iron is needed to fully treat RLS in this patient population. “A second interpretation is that the RLS and IDA were separate events: a patient with idiopathic RLS who subsequently developed anemia,” they said. “Treating the IDA might improve symptoms but may not eliminate the symptoms.”
The study findings were limited by several factors, including the relatively small study population and inability to know the time frame for the development of IDA, the researchers noted. However, the results support the use of intravenous iron therapy for relief of RLS in IDA patients.
“Since IDA could result in epigenetic changes leading to irreversible state of RLS, then urgent and adequate management of the IDA in RLS patients would seem a very prudent and important clinical approach to this specific clinical condition,” they concluded.
The study received no outside funding. The researchers had no disclosures.
Treatment with intravenous ferric carboxymaltose significantly improved symptoms in restless legs syndrome (RLS) patients with iron-deficiency anemia (IDA), data from 29 adults show.
RLS occurs among individuals with normal iron but is at least six times higher among individuals with IDA, Hyoeun Bae, MD, of Keimyung University, Daegu, South Korea, and colleagues wrote. Previous studies have explored iron treatments for RLS patients with IDA, however, guidelines for treatment have not yet been published.
In a study published in Sleep Medicine, the researchers randomized 29 RLS patients with IDA to either 1,500 mg IV ferric carboxymaltose (FCM) or placebo for a short-term period of 6 weeks, followed by a phase 2 study for responders that lasted for 52 weeks. Baseline characteristics, including age, gender, iron parameters, and sleep and mood scales were similar between the groups.
At 6 weeks, patients in the FCM group showed significant improvement in RLS symptom severity based on changes from baseline International Restless Legs Syndrome Study Group scale (IRLS) scores, compared with placebo patients (–13.47 vs. 1.36, P < .001). A secondary outcome of sleep quality also improved significantly in the FCM group, compared with the placebo group.
After 6 weeks, 11 of the 14 patients in the placebo group also received 1,500 mg FCM for an open-label study. These patients also showed significant improvement in IRLS scores from baseline to 6 weeks.
All 23 responders from the short-term studies (13 who received FCM initially and 10 from the postplacebo group) enrolled in a phase 2 long-term study that lasted for 52 weeks; 14 of these completed the full 52-week study period.
Overall, 61% of participants in phase 2 of the study remained off their RLS medications at 52 weeks, and no serious adverse events were reported during the study period. Of these, 10 received one additional dose of FCM and 4 received more than one additional dose. The median change in IRLS score at 4 weeks after treatment was –4.00, compared with the score prior to treatment.
The study is the first of its design to show benefits of intravenous iron therapy for RLS in patients with IDA, the researchers said, noting that the findings of improved, but not cured, RLS symptoms might suggest that more than 1,500 mg of iron is needed to fully treat RLS in this patient population. “A second interpretation is that the RLS and IDA were separate events: a patient with idiopathic RLS who subsequently developed anemia,” they said. “Treating the IDA might improve symptoms but may not eliminate the symptoms.”
The study findings were limited by several factors, including the relatively small study population and inability to know the time frame for the development of IDA, the researchers noted. However, the results support the use of intravenous iron therapy for relief of RLS in IDA patients.
“Since IDA could result in epigenetic changes leading to irreversible state of RLS, then urgent and adequate management of the IDA in RLS patients would seem a very prudent and important clinical approach to this specific clinical condition,” they concluded.
The study received no outside funding. The researchers had no disclosures.
Treatment with intravenous ferric carboxymaltose significantly improved symptoms in restless legs syndrome (RLS) patients with iron-deficiency anemia (IDA), data from 29 adults show.
RLS occurs among individuals with normal iron but is at least six times higher among individuals with IDA, Hyoeun Bae, MD, of Keimyung University, Daegu, South Korea, and colleagues wrote. Previous studies have explored iron treatments for RLS patients with IDA, however, guidelines for treatment have not yet been published.
In a study published in Sleep Medicine, the researchers randomized 29 RLS patients with IDA to either 1,500 mg IV ferric carboxymaltose (FCM) or placebo for a short-term period of 6 weeks, followed by a phase 2 study for responders that lasted for 52 weeks. Baseline characteristics, including age, gender, iron parameters, and sleep and mood scales were similar between the groups.
At 6 weeks, patients in the FCM group showed significant improvement in RLS symptom severity based on changes from baseline International Restless Legs Syndrome Study Group scale (IRLS) scores, compared with placebo patients (–13.47 vs. 1.36, P < .001). A secondary outcome of sleep quality also improved significantly in the FCM group, compared with the placebo group.
After 6 weeks, 11 of the 14 patients in the placebo group also received 1,500 mg FCM for an open-label study. These patients also showed significant improvement in IRLS scores from baseline to 6 weeks.
All 23 responders from the short-term studies (13 who received FCM initially and 10 from the postplacebo group) enrolled in a phase 2 long-term study that lasted for 52 weeks; 14 of these completed the full 52-week study period.
Overall, 61% of participants in phase 2 of the study remained off their RLS medications at 52 weeks, and no serious adverse events were reported during the study period. Of these, 10 received one additional dose of FCM and 4 received more than one additional dose. The median change in IRLS score at 4 weeks after treatment was –4.00, compared with the score prior to treatment.
The study is the first of its design to show benefits of intravenous iron therapy for RLS in patients with IDA, the researchers said, noting that the findings of improved, but not cured, RLS symptoms might suggest that more than 1,500 mg of iron is needed to fully treat RLS in this patient population. “A second interpretation is that the RLS and IDA were separate events: a patient with idiopathic RLS who subsequently developed anemia,” they said. “Treating the IDA might improve symptoms but may not eliminate the symptoms.”
The study findings were limited by several factors, including the relatively small study population and inability to know the time frame for the development of IDA, the researchers noted. However, the results support the use of intravenous iron therapy for relief of RLS in IDA patients.
“Since IDA could result in epigenetic changes leading to irreversible state of RLS, then urgent and adequate management of the IDA in RLS patients would seem a very prudent and important clinical approach to this specific clinical condition,” they concluded.
The study received no outside funding. The researchers had no disclosures.
FROM SLEEP MEDICINE
Transdermal patches ease extrapyramidal symptoms in schizophrenia
Use of a transdermal blonanserin patch significantly improved extrapyramidal symptoms (EPS), compared with oral blonanserin tablets in patients with schizophrenia, according to results of an open-label study of 155 adults.
Blonanserin, a second-generation antipsychotic, has been shown to reduce extrapyramidal symptoms when used to treat schizophrenia, but the impact of switching to a patch on extrapyramidal symptoms and on the use of antiparkinson drugs has not been well studied, Kazutaka Ohi, MD, of Gifu University Graduate School of Medicine, Seki, Japan, and colleagues wrote. Advantages of the patch include the ability to provide stable blood concentrations and the ability to be concealed under clothing to avoid patients’ embarrassment at taking oral medications.
In a study published in Progress in Neuropsychopharmacology & Biological Psychiatry, the researchers identified 155 adults aged 18 years and older diagnosed with schizophrenia who were treated at 37 medical institutions in Japan between February 2015 and May 2017.
The first cohort of 97 patients received blonanserin tablets (8-16 mg/day) for 6 weeks, followed by blonanserin transdermal patches (40-80 mg/day) once daily for 1 year. The second cohort of 58 patients received continuous blonanserin patch therapy. Extrapyramidal symptoms were assessed using the Drug-Induced Extrapyramidal Symptoms Scale (DIEPSS); individual scores ranged from a 0 for normal to a 4 for severe.
Overall, DIEPSS scores decreased significantly in both cohorts after switching from blonanserin tablets or powders to transdermal patches. The average DIEPSS change from baseline at 3, 6, and 12 months was –0.44, –0.07, and –0.14, respectively, in cohort 1, and –0.16, –0.74, and –0.81, respectively, in cohort 2.
The researchers also assessed the impact of transition to transdermal patches on the use of antiparkinsonism drugs using the biperiden equivalents of total antiparkinsonian drugs (BPD-eq) measure. At baseline, about 22% of patients used concomitant antiparkinsonism drugs, compared with 25.8% at 1 year after starting patch treatment. The dose of antiparkinson drugs was not significantly decreased after switching to transdermal patches, in part because of psychiatrists’ prescribing behaviors, Dr. Ohi and colleagues noted.
As a secondary outcome, the researchers examined psychotic symptoms and found that Positive and Negative Syndrome Scale (PANSS) negative symptom scores decreased significantly in patients in cohort 1 who switched from tablets or powders to patches. Changes in scores from baseline to 3, 6, and 12 months were –0.7, –1.0, and –1.3, respectively. Positive PANSS scores did not change significantly in cohort 1. In cohort 2, both positive and negative PANSS scores decreased significantly over 12 months after switching from blonanserin tablets/powders to patches. The mean changes in scores from baseline to 3, 6, and 12 months were –1.6, –2.3, and –2.4, respectively, for PANSS positive symptom scores, and –1.4, –2.7, and –2.8, respectively, for negative symptom scores.
A total of 41.2% of cohort 1 patients and 44.8% of cohort 2 patients discontinued patch treatments by 1 year. Four patients discontinued the patch because of EPS during the treatment period in cohort 1; no patients in cohort 2 discontinued because of EPS.
The study findings were limited by several factors, including the open-label design and lack of controls; also, the study did not examine crossover changes in patients who switched from tablets or powders to patches, the researchers noted.
However, the results indicate that direct switching from blonanserin tablets or powders to transdermal patches reduced EPS and psychotic symptoms in schizophrenia and may be more acceptable to patients, compared with oral medications, as well as more effective, they concluded.
The study received no outside funding, and Dr. Ohi and colleagues had no disclosures.
Use of a transdermal blonanserin patch significantly improved extrapyramidal symptoms (EPS), compared with oral blonanserin tablets in patients with schizophrenia, according to results of an open-label study of 155 adults.
Blonanserin, a second-generation antipsychotic, has been shown to reduce extrapyramidal symptoms when used to treat schizophrenia, but the impact of switching to a patch on extrapyramidal symptoms and on the use of antiparkinson drugs has not been well studied, Kazutaka Ohi, MD, of Gifu University Graduate School of Medicine, Seki, Japan, and colleagues wrote. Advantages of the patch include the ability to provide stable blood concentrations and the ability to be concealed under clothing to avoid patients’ embarrassment at taking oral medications.
In a study published in Progress in Neuropsychopharmacology & Biological Psychiatry, the researchers identified 155 adults aged 18 years and older diagnosed with schizophrenia who were treated at 37 medical institutions in Japan between February 2015 and May 2017.
The first cohort of 97 patients received blonanserin tablets (8-16 mg/day) for 6 weeks, followed by blonanserin transdermal patches (40-80 mg/day) once daily for 1 year. The second cohort of 58 patients received continuous blonanserin patch therapy. Extrapyramidal symptoms were assessed using the Drug-Induced Extrapyramidal Symptoms Scale (DIEPSS); individual scores ranged from a 0 for normal to a 4 for severe.
Overall, DIEPSS scores decreased significantly in both cohorts after switching from blonanserin tablets or powders to transdermal patches. The average DIEPSS change from baseline at 3, 6, and 12 months was –0.44, –0.07, and –0.14, respectively, in cohort 1, and –0.16, –0.74, and –0.81, respectively, in cohort 2.
The researchers also assessed the impact of transition to transdermal patches on the use of antiparkinsonism drugs using the biperiden equivalents of total antiparkinsonian drugs (BPD-eq) measure. At baseline, about 22% of patients used concomitant antiparkinsonism drugs, compared with 25.8% at 1 year after starting patch treatment. The dose of antiparkinson drugs was not significantly decreased after switching to transdermal patches, in part because of psychiatrists’ prescribing behaviors, Dr. Ohi and colleagues noted.
As a secondary outcome, the researchers examined psychotic symptoms and found that Positive and Negative Syndrome Scale (PANSS) negative symptom scores decreased significantly in patients in cohort 1 who switched from tablets or powders to patches. Changes in scores from baseline to 3, 6, and 12 months were –0.7, –1.0, and –1.3, respectively. Positive PANSS scores did not change significantly in cohort 1. In cohort 2, both positive and negative PANSS scores decreased significantly over 12 months after switching from blonanserin tablets/powders to patches. The mean changes in scores from baseline to 3, 6, and 12 months were –1.6, –2.3, and –2.4, respectively, for PANSS positive symptom scores, and –1.4, –2.7, and –2.8, respectively, for negative symptom scores.
A total of 41.2% of cohort 1 patients and 44.8% of cohort 2 patients discontinued patch treatments by 1 year. Four patients discontinued the patch because of EPS during the treatment period in cohort 1; no patients in cohort 2 discontinued because of EPS.
The study findings were limited by several factors, including the open-label design and lack of controls; also, the study did not examine crossover changes in patients who switched from tablets or powders to patches, the researchers noted.
However, the results indicate that direct switching from blonanserin tablets or powders to transdermal patches reduced EPS and psychotic symptoms in schizophrenia and may be more acceptable to patients, compared with oral medications, as well as more effective, they concluded.
The study received no outside funding, and Dr. Ohi and colleagues had no disclosures.
Use of a transdermal blonanserin patch significantly improved extrapyramidal symptoms (EPS), compared with oral blonanserin tablets in patients with schizophrenia, according to results of an open-label study of 155 adults.
Blonanserin, a second-generation antipsychotic, has been shown to reduce extrapyramidal symptoms when used to treat schizophrenia, but the impact of switching to a patch on extrapyramidal symptoms and on the use of antiparkinson drugs has not been well studied, Kazutaka Ohi, MD, of Gifu University Graduate School of Medicine, Seki, Japan, and colleagues wrote. Advantages of the patch include the ability to provide stable blood concentrations and the ability to be concealed under clothing to avoid patients’ embarrassment at taking oral medications.
In a study published in Progress in Neuropsychopharmacology & Biological Psychiatry, the researchers identified 155 adults aged 18 years and older diagnosed with schizophrenia who were treated at 37 medical institutions in Japan between February 2015 and May 2017.
The first cohort of 97 patients received blonanserin tablets (8-16 mg/day) for 6 weeks, followed by blonanserin transdermal patches (40-80 mg/day) once daily for 1 year. The second cohort of 58 patients received continuous blonanserin patch therapy. Extrapyramidal symptoms were assessed using the Drug-Induced Extrapyramidal Symptoms Scale (DIEPSS); individual scores ranged from a 0 for normal to a 4 for severe.
Overall, DIEPSS scores decreased significantly in both cohorts after switching from blonanserin tablets or powders to transdermal patches. The average DIEPSS change from baseline at 3, 6, and 12 months was –0.44, –0.07, and –0.14, respectively, in cohort 1, and –0.16, –0.74, and –0.81, respectively, in cohort 2.
The researchers also assessed the impact of transition to transdermal patches on the use of antiparkinsonism drugs using the biperiden equivalents of total antiparkinsonian drugs (BPD-eq) measure. At baseline, about 22% of patients used concomitant antiparkinsonism drugs, compared with 25.8% at 1 year after starting patch treatment. The dose of antiparkinson drugs was not significantly decreased after switching to transdermal patches, in part because of psychiatrists’ prescribing behaviors, Dr. Ohi and colleagues noted.
As a secondary outcome, the researchers examined psychotic symptoms and found that Positive and Negative Syndrome Scale (PANSS) negative symptom scores decreased significantly in patients in cohort 1 who switched from tablets or powders to patches. Changes in scores from baseline to 3, 6, and 12 months were –0.7, –1.0, and –1.3, respectively. Positive PANSS scores did not change significantly in cohort 1. In cohort 2, both positive and negative PANSS scores decreased significantly over 12 months after switching from blonanserin tablets/powders to patches. The mean changes in scores from baseline to 3, 6, and 12 months were –1.6, –2.3, and –2.4, respectively, for PANSS positive symptom scores, and –1.4, –2.7, and –2.8, respectively, for negative symptom scores.
A total of 41.2% of cohort 1 patients and 44.8% of cohort 2 patients discontinued patch treatments by 1 year. Four patients discontinued the patch because of EPS during the treatment period in cohort 1; no patients in cohort 2 discontinued because of EPS.
The study findings were limited by several factors, including the open-label design and lack of controls; also, the study did not examine crossover changes in patients who switched from tablets or powders to patches, the researchers noted.
However, the results indicate that direct switching from blonanserin tablets or powders to transdermal patches reduced EPS and psychotic symptoms in schizophrenia and may be more acceptable to patients, compared with oral medications, as well as more effective, they concluded.
The study received no outside funding, and Dr. Ohi and colleagues had no disclosures.
FROM PROGRESS IN NEUROPSYCHOPHARMACOLOGY & BIOLOGICAL PSYCHIATRY
Breast cancer history promotes vertebral fracture risk
Women with a history of stage III to stage IV breast cancer had significantly more pathologic vertebral fractures compared to those with stage I and stage II disease, based on data from approximately 5,000 adult women.
Breast cancer remains associated with increased fracture risk in part because of estrogen deficiency, aromatase inhibitors, frailty, and skeletal metastases, wrote Joan C. Lo, MD, of Kaiser Permanente Northern California, Oakland, and colleagues. Fractures associated with these factors have been studied, but many of the existing epidemiologic studies lack detail on fractures related to cancer, they noted. The researchers examined the association between pathologic fractures and major osteoporotic fractures in women with invasive breast cancer who received endocrine therapy.
In a study published in JAMA Network Open (2021 Nov 18. doi: 10.1001/jamanetworkopen.2021.33861), the researchers reviewed data from 5,010 women enrolled in the Pathways Study (3,312 women) or Research Program on Genes, Environment, and Health (RPGEH) study (1,698 women) with newly diagnosed invasive breast cancer who received endocrine therapy. The women were followed for up to 10 years for incident fracture, with a median follow-up period of 6.7 years.
The average age of the women was 60.2 years; 73.3% were non-Hispanic White, 4.9% were Black, 9.4% were Hispanic, and 1.6% were women whose ethnicity was unknown. Approximately 90% of the women were at stage I to stage II at initial diagnosis.
Overall, 340 (6.8%) had incident fractures during the follow-up period. The incident fractures included 46 hip, 104 vertebral, 78 humerus, and 137 wrist fractures. Significantly more women with hip fracture (43.5%) were age 80 years or older, compared with less than 25% of women with vertebral fractures (22.1%), humerus (19.2%), or wrist fracture (15.3%).
Pathologic fractures accounted for 22 of 104 incident vertebral fractures (21.2%) and fewer than 5 of 46 incident hip fractures (8.7%); few wrist and humerus fractures were pathologic. According to tumor stage, 15 of 87 (17.2%) vertebral fractures in women with initial stage I and II were pathologic, compared to 7 of 17 (41.2%) in women with initial stage III to stage IV breast cancer (P < .05).
The results emphasized the need to consider vertebral fracture risk in women with breast cancer, notably advanced stage cancer, as approximately one-third of the incident vertebral fractures in this subset of patients was deemed cancer-related, the researchers noted.
“As the axial skeleton is a common site for breast cancer metastasis and vertebrae a common site for pathologic fracture, primary care physicians should consider the possibility of pathologic fracture in women with higher risk based on advanced-stage cancer history,” the researchers wrote.
The study findings were limited by several factors, including the lack of data on fracture risk factors, treatment, and chemotherapy, and the inclusion only of clinically diagnosed fractures and not asymptomatic vertebral fractures, the researchers noted. However, the results were strengthened by the large sample size and comprehensive fracture assessment, they said. Additional studies to examine nonpathologic fracture risk according to breast cancer treatment, such as the use of aromatase inhibitors versus cytotoxic chemotherapy, may inform which women would benefit from more aggressive osteoporotic fracture prevention, they concluded.
Findings inform shared decision-making
“This study highlights the apparent association between an initial diagnosis of stage III or IV breast cancer and an increased risk for pathologic vertebral fracture,” said Constance Bohon, MD, a gynecologist in private practice in Washington, D.C., in an interview. “Most likely this finding is secondary to breast cancer metastases,” Dr. Bohon noted. However, she questioned whether there is a difference in fracture rates between women who received only aromatase inhibitors, those who received tamoxifen, and those who received both treatments.
“Additional data to determine the age of menopause, exercise frequency, current weight, and family history of osteoporosis may serve to identify those at highest risk for pathologic vertebral fracture,” said Dr. Bohon. “Until further data are available, clinicians should review this study and counsel their patients regarding options to potentially mitigate their apparent increased risk for pathologic vertebral fracture,” she emphasized.
The study was supported by the National Cancer Institute, National Institutes of Health, and the Research Program on Genes, Environment, and Health of Kaiser Permanente Northern California. The researchers had no financial conflicts to disclose. Dr. Bohon had no financial conflicts to disclose but serves on the Editorial Advisory Board of Ob.Gyn. News.
Women with a history of stage III to stage IV breast cancer had significantly more pathologic vertebral fractures compared to those with stage I and stage II disease, based on data from approximately 5,000 adult women.
Breast cancer remains associated with increased fracture risk in part because of estrogen deficiency, aromatase inhibitors, frailty, and skeletal metastases, wrote Joan C. Lo, MD, of Kaiser Permanente Northern California, Oakland, and colleagues. Fractures associated with these factors have been studied, but many of the existing epidemiologic studies lack detail on fractures related to cancer, they noted. The researchers examined the association between pathologic fractures and major osteoporotic fractures in women with invasive breast cancer who received endocrine therapy.
In a study published in JAMA Network Open (2021 Nov 18. doi: 10.1001/jamanetworkopen.2021.33861), the researchers reviewed data from 5,010 women enrolled in the Pathways Study (3,312 women) or Research Program on Genes, Environment, and Health (RPGEH) study (1,698 women) with newly diagnosed invasive breast cancer who received endocrine therapy. The women were followed for up to 10 years for incident fracture, with a median follow-up period of 6.7 years.
The average age of the women was 60.2 years; 73.3% were non-Hispanic White, 4.9% were Black, 9.4% were Hispanic, and 1.6% were women whose ethnicity was unknown. Approximately 90% of the women were at stage I to stage II at initial diagnosis.
Overall, 340 (6.8%) had incident fractures during the follow-up period. The incident fractures included 46 hip, 104 vertebral, 78 humerus, and 137 wrist fractures. Significantly more women with hip fracture (43.5%) were age 80 years or older, compared with less than 25% of women with vertebral fractures (22.1%), humerus (19.2%), or wrist fracture (15.3%).
Pathologic fractures accounted for 22 of 104 incident vertebral fractures (21.2%) and fewer than 5 of 46 incident hip fractures (8.7%); few wrist and humerus fractures were pathologic. According to tumor stage, 15 of 87 (17.2%) vertebral fractures in women with initial stage I and II were pathologic, compared to 7 of 17 (41.2%) in women with initial stage III to stage IV breast cancer (P < .05).
The results emphasized the need to consider vertebral fracture risk in women with breast cancer, notably advanced stage cancer, as approximately one-third of the incident vertebral fractures in this subset of patients was deemed cancer-related, the researchers noted.
“As the axial skeleton is a common site for breast cancer metastasis and vertebrae a common site for pathologic fracture, primary care physicians should consider the possibility of pathologic fracture in women with higher risk based on advanced-stage cancer history,” the researchers wrote.
The study findings were limited by several factors, including the lack of data on fracture risk factors, treatment, and chemotherapy, and the inclusion only of clinically diagnosed fractures and not asymptomatic vertebral fractures, the researchers noted. However, the results were strengthened by the large sample size and comprehensive fracture assessment, they said. Additional studies to examine nonpathologic fracture risk according to breast cancer treatment, such as the use of aromatase inhibitors versus cytotoxic chemotherapy, may inform which women would benefit from more aggressive osteoporotic fracture prevention, they concluded.
Findings inform shared decision-making
“This study highlights the apparent association between an initial diagnosis of stage III or IV breast cancer and an increased risk for pathologic vertebral fracture,” said Constance Bohon, MD, a gynecologist in private practice in Washington, D.C., in an interview. “Most likely this finding is secondary to breast cancer metastases,” Dr. Bohon noted. However, she questioned whether there is a difference in fracture rates between women who received only aromatase inhibitors, those who received tamoxifen, and those who received both treatments.
“Additional data to determine the age of menopause, exercise frequency, current weight, and family history of osteoporosis may serve to identify those at highest risk for pathologic vertebral fracture,” said Dr. Bohon. “Until further data are available, clinicians should review this study and counsel their patients regarding options to potentially mitigate their apparent increased risk for pathologic vertebral fracture,” she emphasized.
The study was supported by the National Cancer Institute, National Institutes of Health, and the Research Program on Genes, Environment, and Health of Kaiser Permanente Northern California. The researchers had no financial conflicts to disclose. Dr. Bohon had no financial conflicts to disclose but serves on the Editorial Advisory Board of Ob.Gyn. News.
Women with a history of stage III to stage IV breast cancer had significantly more pathologic vertebral fractures compared to those with stage I and stage II disease, based on data from approximately 5,000 adult women.
Breast cancer remains associated with increased fracture risk in part because of estrogen deficiency, aromatase inhibitors, frailty, and skeletal metastases, wrote Joan C. Lo, MD, of Kaiser Permanente Northern California, Oakland, and colleagues. Fractures associated with these factors have been studied, but many of the existing epidemiologic studies lack detail on fractures related to cancer, they noted. The researchers examined the association between pathologic fractures and major osteoporotic fractures in women with invasive breast cancer who received endocrine therapy.
In a study published in JAMA Network Open (2021 Nov 18. doi: 10.1001/jamanetworkopen.2021.33861), the researchers reviewed data from 5,010 women enrolled in the Pathways Study (3,312 women) or Research Program on Genes, Environment, and Health (RPGEH) study (1,698 women) with newly diagnosed invasive breast cancer who received endocrine therapy. The women were followed for up to 10 years for incident fracture, with a median follow-up period of 6.7 years.
The average age of the women was 60.2 years; 73.3% were non-Hispanic White, 4.9% were Black, 9.4% were Hispanic, and 1.6% were women whose ethnicity was unknown. Approximately 90% of the women were at stage I to stage II at initial diagnosis.
Overall, 340 (6.8%) had incident fractures during the follow-up period. The incident fractures included 46 hip, 104 vertebral, 78 humerus, and 137 wrist fractures. Significantly more women with hip fracture (43.5%) were age 80 years or older, compared with less than 25% of women with vertebral fractures (22.1%), humerus (19.2%), or wrist fracture (15.3%).
Pathologic fractures accounted for 22 of 104 incident vertebral fractures (21.2%) and fewer than 5 of 46 incident hip fractures (8.7%); few wrist and humerus fractures were pathologic. According to tumor stage, 15 of 87 (17.2%) vertebral fractures in women with initial stage I and II were pathologic, compared to 7 of 17 (41.2%) in women with initial stage III to stage IV breast cancer (P < .05).
The results emphasized the need to consider vertebral fracture risk in women with breast cancer, notably advanced stage cancer, as approximately one-third of the incident vertebral fractures in this subset of patients was deemed cancer-related, the researchers noted.
“As the axial skeleton is a common site for breast cancer metastasis and vertebrae a common site for pathologic fracture, primary care physicians should consider the possibility of pathologic fracture in women with higher risk based on advanced-stage cancer history,” the researchers wrote.
The study findings were limited by several factors, including the lack of data on fracture risk factors, treatment, and chemotherapy, and the inclusion only of clinically diagnosed fractures and not asymptomatic vertebral fractures, the researchers noted. However, the results were strengthened by the large sample size and comprehensive fracture assessment, they said. Additional studies to examine nonpathologic fracture risk according to breast cancer treatment, such as the use of aromatase inhibitors versus cytotoxic chemotherapy, may inform which women would benefit from more aggressive osteoporotic fracture prevention, they concluded.
Findings inform shared decision-making
“This study highlights the apparent association between an initial diagnosis of stage III or IV breast cancer and an increased risk for pathologic vertebral fracture,” said Constance Bohon, MD, a gynecologist in private practice in Washington, D.C., in an interview. “Most likely this finding is secondary to breast cancer metastases,” Dr. Bohon noted. However, she questioned whether there is a difference in fracture rates between women who received only aromatase inhibitors, those who received tamoxifen, and those who received both treatments.
“Additional data to determine the age of menopause, exercise frequency, current weight, and family history of osteoporosis may serve to identify those at highest risk for pathologic vertebral fracture,” said Dr. Bohon. “Until further data are available, clinicians should review this study and counsel their patients regarding options to potentially mitigate their apparent increased risk for pathologic vertebral fracture,” she emphasized.
The study was supported by the National Cancer Institute, National Institutes of Health, and the Research Program on Genes, Environment, and Health of Kaiser Permanente Northern California. The researchers had no financial conflicts to disclose. Dr. Bohon had no financial conflicts to disclose but serves on the Editorial Advisory Board of Ob.Gyn. News.
FROM JAMA NETWORK OPEN