User login
Jennifer Smith is the editor of Oncology Practice, part of MDedge Hematology/Oncology. She was previously the editor of Hematology Times, an editor at Principal Investigators Association, and a reporter at The Oneida Daily Dispatch. She has a BS in journalism.
New leaders at SKCC, Mount Sinai, NHF
The Sidney Kimmel Cancer Center–Jefferson Health (SKCC) has a new director of bone marrow transplant and cell-based therapy, Mount Sinai has a new head of clinical cancer research, and the National Hemophilia Foundation (NHF) has a new president and CEO.
Usama Gergis, MD, has joined SKCC in Philadelphia as director of the bone marrow transplant and immune cellular therapy program and as a professor in the department of medical oncology, division of hematological malignancies.
Dr. Gergis came to SKCC from Weill Cornell Medicine in New York, where he helped expand the use of umbilical cord blood transplant and established an immune cellular therapy program. He also established and led one of the largest oncology international medicine programs in the United States.
Dr. Gergis earned his medical degree from Cairo (Egypt) University. He completed an internal medicine residency and a hematology/oncology fellowship at the Brooklyn Hospital of Weill Cornell. He also completed a bone marrow transplant fellowship at the Moffitt Cancer Center in Tampa.
Karyn Aalami Goodman, MD, has been hired to lead clinical cancer research at Mount Sinai, New York. She is now the associate director for clinical research at the Tisch Cancer Institute and a professor and vice-chair for research and quality in the department of radiation oncology. In these roles, she will develop the infrastructure and resources to support clinical trials of cancer patients.
Dr. Goodman’s own research is focused on improving outcomes for patients with gastrointestinal malignancies. She has helped develop protocols combining radiation, chemotherapy, targeted agents, and immunotherapy.
Dr. Goodman earned her medical degree from Stanford (Calif.) University. She completed an internship in internal medicine at Stanford and residency training in radiation oncology at Memorial Sloan-Kettering Cancer Center in New York. She was previously associate director of clinical research at the University of Colorado Cancer Center in Aurora.
Leonard A. Valentino, MD, has assumed the role of president and CEO of the National Hemophilia Foundation in New York. In this role, Dr. Valentino will “work to advance the foundation’s mission of education, advocacy, and research,” according to the NHF.
Dr. Valentino previously held leadership roles at Spark Therapeutics, Shire, Baxalta, and Baxter Healthcare Corporation. Prior to that, he founded and led the Hemophilia and Thrombophilia Center at Rush University Medical Center in Chicago.
Dr. Valentino earned his undergraduate and medical degrees from Creighton University in Omaha, Neb. He completed his residency at the University of Illinois at Chicago and a fellowship in pediatric hematology-oncology at the University of California, Los Angeles .
The Sidney Kimmel Cancer Center–Jefferson Health (SKCC) has a new director of bone marrow transplant and cell-based therapy, Mount Sinai has a new head of clinical cancer research, and the National Hemophilia Foundation (NHF) has a new president and CEO.
Usama Gergis, MD, has joined SKCC in Philadelphia as director of the bone marrow transplant and immune cellular therapy program and as a professor in the department of medical oncology, division of hematological malignancies.
Dr. Gergis came to SKCC from Weill Cornell Medicine in New York, where he helped expand the use of umbilical cord blood transplant and established an immune cellular therapy program. He also established and led one of the largest oncology international medicine programs in the United States.
Dr. Gergis earned his medical degree from Cairo (Egypt) University. He completed an internal medicine residency and a hematology/oncology fellowship at the Brooklyn Hospital of Weill Cornell. He also completed a bone marrow transplant fellowship at the Moffitt Cancer Center in Tampa.
Karyn Aalami Goodman, MD, has been hired to lead clinical cancer research at Mount Sinai, New York. She is now the associate director for clinical research at the Tisch Cancer Institute and a professor and vice-chair for research and quality in the department of radiation oncology. In these roles, she will develop the infrastructure and resources to support clinical trials of cancer patients.
Dr. Goodman’s own research is focused on improving outcomes for patients with gastrointestinal malignancies. She has helped develop protocols combining radiation, chemotherapy, targeted agents, and immunotherapy.
Dr. Goodman earned her medical degree from Stanford (Calif.) University. She completed an internship in internal medicine at Stanford and residency training in radiation oncology at Memorial Sloan-Kettering Cancer Center in New York. She was previously associate director of clinical research at the University of Colorado Cancer Center in Aurora.
Leonard A. Valentino, MD, has assumed the role of president and CEO of the National Hemophilia Foundation in New York. In this role, Dr. Valentino will “work to advance the foundation’s mission of education, advocacy, and research,” according to the NHF.
Dr. Valentino previously held leadership roles at Spark Therapeutics, Shire, Baxalta, and Baxter Healthcare Corporation. Prior to that, he founded and led the Hemophilia and Thrombophilia Center at Rush University Medical Center in Chicago.
Dr. Valentino earned his undergraduate and medical degrees from Creighton University in Omaha, Neb. He completed his residency at the University of Illinois at Chicago and a fellowship in pediatric hematology-oncology at the University of California, Los Angeles .
The Sidney Kimmel Cancer Center–Jefferson Health (SKCC) has a new director of bone marrow transplant and cell-based therapy, Mount Sinai has a new head of clinical cancer research, and the National Hemophilia Foundation (NHF) has a new president and CEO.
Usama Gergis, MD, has joined SKCC in Philadelphia as director of the bone marrow transplant and immune cellular therapy program and as a professor in the department of medical oncology, division of hematological malignancies.
Dr. Gergis came to SKCC from Weill Cornell Medicine in New York, where he helped expand the use of umbilical cord blood transplant and established an immune cellular therapy program. He also established and led one of the largest oncology international medicine programs in the United States.
Dr. Gergis earned his medical degree from Cairo (Egypt) University. He completed an internal medicine residency and a hematology/oncology fellowship at the Brooklyn Hospital of Weill Cornell. He also completed a bone marrow transplant fellowship at the Moffitt Cancer Center in Tampa.
Karyn Aalami Goodman, MD, has been hired to lead clinical cancer research at Mount Sinai, New York. She is now the associate director for clinical research at the Tisch Cancer Institute and a professor and vice-chair for research and quality in the department of radiation oncology. In these roles, she will develop the infrastructure and resources to support clinical trials of cancer patients.
Dr. Goodman’s own research is focused on improving outcomes for patients with gastrointestinal malignancies. She has helped develop protocols combining radiation, chemotherapy, targeted agents, and immunotherapy.
Dr. Goodman earned her medical degree from Stanford (Calif.) University. She completed an internship in internal medicine at Stanford and residency training in radiation oncology at Memorial Sloan-Kettering Cancer Center in New York. She was previously associate director of clinical research at the University of Colorado Cancer Center in Aurora.
Leonard A. Valentino, MD, has assumed the role of president and CEO of the National Hemophilia Foundation in New York. In this role, Dr. Valentino will “work to advance the foundation’s mission of education, advocacy, and research,” according to the NHF.
Dr. Valentino previously held leadership roles at Spark Therapeutics, Shire, Baxalta, and Baxter Healthcare Corporation. Prior to that, he founded and led the Hemophilia and Thrombophilia Center at Rush University Medical Center in Chicago.
Dr. Valentino earned his undergraduate and medical degrees from Creighton University in Omaha, Neb. He completed his residency at the University of Illinois at Chicago and a fellowship in pediatric hematology-oncology at the University of California, Los Angeles .
Gene therapy effective in hemophilia B patients with neutralizing antibodies
The gene therapy etranacogene dezaparvovec (AMT-061) continues to demonstrate safety and efficacy in patients with hemophilia B, according to a 1-year update of a phase 2b trial.
All three patients in this trial experienced sustained increases in factor IX (FIX) activity and were able to stop prophylaxis without suffering any bleeds. Adverse events related to treatment were mild and transient.
These favorable results are particularly noteworthy because all three patients had anti-AAV5 neutralizing antibodies at baseline, according to Steven W. Pipe, MD, of the University of Michigan, Ann Arbor. He noted that studies of etranacogene dezaparvovec and its predecessor, AMT-060, are the only studies that have not excluded hemophilia patients based on preexisting immunity.
Dr. Pipe presented the latest phase 2b results with etranacogene dezaparvovec at the annual congress of the European Association for Haemophilia and Allied Disorders.
Etranacogene dezaparvovec uses an AAV5 serotype with a transgene expression cassette that codes for the hyperactive Padua FIX variant, Dr. Pipe explained. Etranacogene dezaparvovec has a structure that is nearly identical to that of AMT-060, except for two nucleotide substitutions in the coding sequence for FIX.
AMT-060 enabled stable expression of FIX that has persisted for up to 4 years without any late-emergent safety signals (Blood 2019. 134 Supplement 1: 2059). Dr. Pipe said the “enhanced version” of AMT-060, etranacogene dezaparvovec, has produced even higher levels of FIX activity in the phase 2b study (NCT03489291).
The ongoing study enrolled three men with moderate to severe FIX deficiency at baseline. The patients were 43, 50, and 47 years of age, respectively. Two patients are HIV positive, and all had hepatitis C that resolved.
All three patients were receiving FIX prophylaxis and on-demand treatment at baseline. In the year prior to screening, patients had one, three, and five bleeds, respectively. All three patients had anti-AAV5 neutralizing antibodies.
Efficacy
Patients received a single dose of etranacogene dezaparvovec at 2 x 1013 genome copies/kg. All three patients achieved the primary endpoint, which was FIX activity of at least 5% at 6 weeks.
At 52 weeks, the mean FIX activity was 41%. Patients 1 and 3 have maintained FIX activity of 40% or greater, which is in the nonhemophilic range. Patient 2 has maintained FIX activity in the mild range. At 52 weeks, FIX activity levels were 50.2%, 40.8%, and 31.3%, respectively.
All patients remain free of prophylaxis and bleeds. Patient 3 received a single FIX infusion as a precaution in the perioperative setting. There was no evidence of a bleed in this patient.
Safety
Etranacogene dezaparvovec was generally well tolerated, Dr. Pipe said. One patient had two adverse events that were possibly related to etranacogene dezaparvovec. Both events – transient, self-limiting headache and slightly elevated C-reactive protein – resolved without intervention.
There was one serious adverse event, but it was considered unrelated to treatment. Patient 3 required hip surgery for preexisting avascular necrosis.
Dr. Pipe said there was no evidence of transaminitis. There were modest, transient elevations in liver enzymes, but this was not enough to trigger protocol-specified immunosuppression.
Specifically, one patient had ALT elevations at weeks 22 and 44, and one patient had AST elevations at weeks 2, 4, and 31. All of these resolved quickly without treatment or an impact on FIX activity, Dr. Pipe noted.
Next steps
This study is ongoing, and patients will be followed for 5 years. Dr. Pipe said the main focus of follow-up will be to determine if patients maintain durable expression of FIX.
A phase 3 trial of etranacogene dezaparvovec is ongoing as well. The trial, HOPE-B (NCT03569891), is fully enrolled, and dosing is planned for 55 patients.
“We’re looking forward to data analysis later this year,” Dr. Pipe said. “This will be the only phase 3 study, and really the only platform so far, that is not planning to exclude patients based on preexisting immunity.”
If all goes well in the phase 3 study, etranacogene dezaparvovec could be approved by the Food and Drug Administration very soon, Dr. Pipe added.
UniQure, the company developing etranacogene dezaparvovec, is planning to submit the biologics license application to the FDA next year. Etranacogene dezaparvovec was granted breakthrough designation from the FDA and is therefore eligible for priority review, so the gene therapy could be approved as early as 2021.
The phase 2b trial of etranacogene dezaparvovec is sponsored by uniQure. Dr. Pipe disclosed relationships with uniQure and other companies.
SOURCE: Pipe SW et al. EAHAD 2020, Abstract OR10.
The gene therapy etranacogene dezaparvovec (AMT-061) continues to demonstrate safety and efficacy in patients with hemophilia B, according to a 1-year update of a phase 2b trial.
All three patients in this trial experienced sustained increases in factor IX (FIX) activity and were able to stop prophylaxis without suffering any bleeds. Adverse events related to treatment were mild and transient.
These favorable results are particularly noteworthy because all three patients had anti-AAV5 neutralizing antibodies at baseline, according to Steven W. Pipe, MD, of the University of Michigan, Ann Arbor. He noted that studies of etranacogene dezaparvovec and its predecessor, AMT-060, are the only studies that have not excluded hemophilia patients based on preexisting immunity.
Dr. Pipe presented the latest phase 2b results with etranacogene dezaparvovec at the annual congress of the European Association for Haemophilia and Allied Disorders.
Etranacogene dezaparvovec uses an AAV5 serotype with a transgene expression cassette that codes for the hyperactive Padua FIX variant, Dr. Pipe explained. Etranacogene dezaparvovec has a structure that is nearly identical to that of AMT-060, except for two nucleotide substitutions in the coding sequence for FIX.
AMT-060 enabled stable expression of FIX that has persisted for up to 4 years without any late-emergent safety signals (Blood 2019. 134 Supplement 1: 2059). Dr. Pipe said the “enhanced version” of AMT-060, etranacogene dezaparvovec, has produced even higher levels of FIX activity in the phase 2b study (NCT03489291).
The ongoing study enrolled three men with moderate to severe FIX deficiency at baseline. The patients were 43, 50, and 47 years of age, respectively. Two patients are HIV positive, and all had hepatitis C that resolved.
All three patients were receiving FIX prophylaxis and on-demand treatment at baseline. In the year prior to screening, patients had one, three, and five bleeds, respectively. All three patients had anti-AAV5 neutralizing antibodies.
Efficacy
Patients received a single dose of etranacogene dezaparvovec at 2 x 1013 genome copies/kg. All three patients achieved the primary endpoint, which was FIX activity of at least 5% at 6 weeks.
At 52 weeks, the mean FIX activity was 41%. Patients 1 and 3 have maintained FIX activity of 40% or greater, which is in the nonhemophilic range. Patient 2 has maintained FIX activity in the mild range. At 52 weeks, FIX activity levels were 50.2%, 40.8%, and 31.3%, respectively.
All patients remain free of prophylaxis and bleeds. Patient 3 received a single FIX infusion as a precaution in the perioperative setting. There was no evidence of a bleed in this patient.
Safety
Etranacogene dezaparvovec was generally well tolerated, Dr. Pipe said. One patient had two adverse events that were possibly related to etranacogene dezaparvovec. Both events – transient, self-limiting headache and slightly elevated C-reactive protein – resolved without intervention.
There was one serious adverse event, but it was considered unrelated to treatment. Patient 3 required hip surgery for preexisting avascular necrosis.
Dr. Pipe said there was no evidence of transaminitis. There were modest, transient elevations in liver enzymes, but this was not enough to trigger protocol-specified immunosuppression.
Specifically, one patient had ALT elevations at weeks 22 and 44, and one patient had AST elevations at weeks 2, 4, and 31. All of these resolved quickly without treatment or an impact on FIX activity, Dr. Pipe noted.
Next steps
This study is ongoing, and patients will be followed for 5 years. Dr. Pipe said the main focus of follow-up will be to determine if patients maintain durable expression of FIX.
A phase 3 trial of etranacogene dezaparvovec is ongoing as well. The trial, HOPE-B (NCT03569891), is fully enrolled, and dosing is planned for 55 patients.
“We’re looking forward to data analysis later this year,” Dr. Pipe said. “This will be the only phase 3 study, and really the only platform so far, that is not planning to exclude patients based on preexisting immunity.”
If all goes well in the phase 3 study, etranacogene dezaparvovec could be approved by the Food and Drug Administration very soon, Dr. Pipe added.
UniQure, the company developing etranacogene dezaparvovec, is planning to submit the biologics license application to the FDA next year. Etranacogene dezaparvovec was granted breakthrough designation from the FDA and is therefore eligible for priority review, so the gene therapy could be approved as early as 2021.
The phase 2b trial of etranacogene dezaparvovec is sponsored by uniQure. Dr. Pipe disclosed relationships with uniQure and other companies.
SOURCE: Pipe SW et al. EAHAD 2020, Abstract OR10.
The gene therapy etranacogene dezaparvovec (AMT-061) continues to demonstrate safety and efficacy in patients with hemophilia B, according to a 1-year update of a phase 2b trial.
All three patients in this trial experienced sustained increases in factor IX (FIX) activity and were able to stop prophylaxis without suffering any bleeds. Adverse events related to treatment were mild and transient.
These favorable results are particularly noteworthy because all three patients had anti-AAV5 neutralizing antibodies at baseline, according to Steven W. Pipe, MD, of the University of Michigan, Ann Arbor. He noted that studies of etranacogene dezaparvovec and its predecessor, AMT-060, are the only studies that have not excluded hemophilia patients based on preexisting immunity.
Dr. Pipe presented the latest phase 2b results with etranacogene dezaparvovec at the annual congress of the European Association for Haemophilia and Allied Disorders.
Etranacogene dezaparvovec uses an AAV5 serotype with a transgene expression cassette that codes for the hyperactive Padua FIX variant, Dr. Pipe explained. Etranacogene dezaparvovec has a structure that is nearly identical to that of AMT-060, except for two nucleotide substitutions in the coding sequence for FIX.
AMT-060 enabled stable expression of FIX that has persisted for up to 4 years without any late-emergent safety signals (Blood 2019. 134 Supplement 1: 2059). Dr. Pipe said the “enhanced version” of AMT-060, etranacogene dezaparvovec, has produced even higher levels of FIX activity in the phase 2b study (NCT03489291).
The ongoing study enrolled three men with moderate to severe FIX deficiency at baseline. The patients were 43, 50, and 47 years of age, respectively. Two patients are HIV positive, and all had hepatitis C that resolved.
All three patients were receiving FIX prophylaxis and on-demand treatment at baseline. In the year prior to screening, patients had one, three, and five bleeds, respectively. All three patients had anti-AAV5 neutralizing antibodies.
Efficacy
Patients received a single dose of etranacogene dezaparvovec at 2 x 1013 genome copies/kg. All three patients achieved the primary endpoint, which was FIX activity of at least 5% at 6 weeks.
At 52 weeks, the mean FIX activity was 41%. Patients 1 and 3 have maintained FIX activity of 40% or greater, which is in the nonhemophilic range. Patient 2 has maintained FIX activity in the mild range. At 52 weeks, FIX activity levels were 50.2%, 40.8%, and 31.3%, respectively.
All patients remain free of prophylaxis and bleeds. Patient 3 received a single FIX infusion as a precaution in the perioperative setting. There was no evidence of a bleed in this patient.
Safety
Etranacogene dezaparvovec was generally well tolerated, Dr. Pipe said. One patient had two adverse events that were possibly related to etranacogene dezaparvovec. Both events – transient, self-limiting headache and slightly elevated C-reactive protein – resolved without intervention.
There was one serious adverse event, but it was considered unrelated to treatment. Patient 3 required hip surgery for preexisting avascular necrosis.
Dr. Pipe said there was no evidence of transaminitis. There were modest, transient elevations in liver enzymes, but this was not enough to trigger protocol-specified immunosuppression.
Specifically, one patient had ALT elevations at weeks 22 and 44, and one patient had AST elevations at weeks 2, 4, and 31. All of these resolved quickly without treatment or an impact on FIX activity, Dr. Pipe noted.
Next steps
This study is ongoing, and patients will be followed for 5 years. Dr. Pipe said the main focus of follow-up will be to determine if patients maintain durable expression of FIX.
A phase 3 trial of etranacogene dezaparvovec is ongoing as well. The trial, HOPE-B (NCT03569891), is fully enrolled, and dosing is planned for 55 patients.
“We’re looking forward to data analysis later this year,” Dr. Pipe said. “This will be the only phase 3 study, and really the only platform so far, that is not planning to exclude patients based on preexisting immunity.”
If all goes well in the phase 3 study, etranacogene dezaparvovec could be approved by the Food and Drug Administration very soon, Dr. Pipe added.
UniQure, the company developing etranacogene dezaparvovec, is planning to submit the biologics license application to the FDA next year. Etranacogene dezaparvovec was granted breakthrough designation from the FDA and is therefore eligible for priority review, so the gene therapy could be approved as early as 2021.
The phase 2b trial of etranacogene dezaparvovec is sponsored by uniQure. Dr. Pipe disclosed relationships with uniQure and other companies.
SOURCE: Pipe SW et al. EAHAD 2020, Abstract OR10.
REPORTING FROM EAHAD 2020
Fear drives activity changes in hemophilia patients
Fear of negative events can drive changes in activity levels among patients with hemophilia A, results of the HemACTIVE study suggest.
Patients were more likely to adjust their level of physical activity due to fear of bleeding and joint damage rather than previously experienced bleeding or joint damage.
However, past experience was more likely than fear to make patients stop physical activities altogether.
Mark Skinner, of the Institute for Policy Advancement in Washington, D.C., and colleagues presented these findings in a poster from the annual congress of the European Association for Haemophilia and Allied Disorders.
Mr. Skinner, who is a hemophilia patient himself, said the goal of the HemACTIVE study is to better understand how hemophilia affects patients’ lives.
“We wanted to understand the limitations, challenges, and compromises of individuals living with hemophilia,” Mr. Skinner said. “What has motivated them or prevented them from living more full, active lives doing the kind of work, leisure, and social activities that those without hemophilia do? Is it treatment choice, is it satisfaction with treatment, is it fear?
“We wanted to do a comprehensive study that really looked at the intersection of treatment adherence and satisfaction, the emotional components that relate to those decisions, and the challenges and compromises so that we could better identify what we need to consider as patients think about either changing their therapy or changing their treatment regimen on existing therapy.”
Previous results from the HemACTIVE study showed that, although activity levels differed among hemophilia patients, all patients surveyed wanted greater activity levels, better protection from bleeding, better pain relief, and less-frequent infusions (EAHAD 2019, Abstract P084). In addition, patients who used factor VIII products with an extended half-life were more active and more likely to adhere to their prescribed treatment (ISTH 2019, Abstract PB0210).
The results reported at EAHAD 2020 focus on patients’ reasons for modifying physical activity. Patients and caregivers completed a screening phone interview, followed by a 25-minute, web-based questionnaire on patient activity.
There were 275 respondents – 194 patients with hemophilia A and 81 caregivers – from Canada, France, Germany, Italy, and the United States. Patients had severe (61%) or moderate (39%) hemophilia A, and most (67%) were receiving prophylaxis.
Most patients (70%) were “active” or “extremely/very active,” 77% of patients adjusted their activities because of their hemophilia, and nearly half of patients stopped activities because of their disease.
Fear drives adjustments in activity
Patients were sometimes more likely to adjust their activities based on fear of experiencing an event, as opposed to previously experiencing that event.
Specifically, 44% of patients adjusted their activities due to fear of joint damage, compared with 36% of patients who made adjustments because of past significant joint damage.
Similarly, 41% of patients adjusted activities due to fear of breakthrough bleeds, compared with 36% of patients who made adjustments because of past experience with bleeds and 25% who made adjustments because of significant past bleeds.
On the other hand, a similar percentage of patients adjusted activities because of past experience with pain (43%) and fear of pain (41%). And a similar percentage of patients adjusted activities because of existing joint damage restrictions (35%) and fear of joint deterioration (32%).
Past experience prompts discontinuation of activity
Overall, 47% of patients said anxiety was the most common emotional reason for stopping physical activities. However, patients were consistently more likely to stop activities because of past experience rather than fear or anxiety.
Specifically, 50% of patients stopped activities because of significant past joint damage, 46% stopped because of developing joint problems, and 38% stopped due to fear of joint damage.
More patients stopped activities because of significant past bleeds (41%) rather than fear of breakthrough bleeds (26%). More patients stopped activities because they developed chronic pain (38%) rather than fear of pain (less than 15%). And more patients stopped activities because of existing joint damage restrictions (62%) rather than fear of joint damage (34%).
Applying results to practice: Changing the conversation
Ideally, these findings would be used to promote individualized treatment of hemophilia driven by patients’ goals, Mr. Skinner said. By better understanding patients’ feelings and motivations, clinicians may devise more personalized treatment regimens that align with patients’ goals and improve their quality of life.
Rather than adjusting treatment based only on “hard metrics” such as bleeding events, “we need to take a more holistic approach to looking at outcomes that are more important to patients,” Mr. Skinner said. This type of approach is particularly important to Mr. Skinner as someone who has severe hemophilia A.
“Because hemophilia is a life-long disease, and you’re born with it, you make conscious or unconscious adaptations throughout your life,” he explained. “Your expectations or aspirations adjust to what you’ve been told you can or cannot do because of your hemophilia. The choices I made for my career, where I live, the type of vacations I go on, the type of sports I participate in have all been limited over the course of time, which has meant that I’ve made compromises. There are a lot of individuals with hemophilia who are making decisions that are not what their life goals are.
“What this research helps me understand is that we can change the conversation and build it around an individual patient and understand what their aspirations are. If a clinician understands what I’m wanting to achieve in life … we can build a treatment regime around helping me achieve those goals. That is known to improve adherence.
“The goal, really, is to have hemophilia as a secondary consideration. Instead of saying: ‘You have hemophilia, so these are the options available to you,’ you can say, ‘what is it that you would like to achieve, and then we’ll figure out how your treatment for hemophilia can be adjusted to help you achieve those goals.’ It may sound like a nuance, but it really is reversing the conversation. The goal setting first versus your disease comes first.”
The HemACTIVE study was supported by Bayer. Mr. Skinner disclosed relationships with Bayer and other pharmaceutical companies.
SOURCE: Skinner M et al. EAHAD 2020, Abstract P304.
Fear of negative events can drive changes in activity levels among patients with hemophilia A, results of the HemACTIVE study suggest.
Patients were more likely to adjust their level of physical activity due to fear of bleeding and joint damage rather than previously experienced bleeding or joint damage.
However, past experience was more likely than fear to make patients stop physical activities altogether.
Mark Skinner, of the Institute for Policy Advancement in Washington, D.C., and colleagues presented these findings in a poster from the annual congress of the European Association for Haemophilia and Allied Disorders.
Mr. Skinner, who is a hemophilia patient himself, said the goal of the HemACTIVE study is to better understand how hemophilia affects patients’ lives.
“We wanted to understand the limitations, challenges, and compromises of individuals living with hemophilia,” Mr. Skinner said. “What has motivated them or prevented them from living more full, active lives doing the kind of work, leisure, and social activities that those without hemophilia do? Is it treatment choice, is it satisfaction with treatment, is it fear?
“We wanted to do a comprehensive study that really looked at the intersection of treatment adherence and satisfaction, the emotional components that relate to those decisions, and the challenges and compromises so that we could better identify what we need to consider as patients think about either changing their therapy or changing their treatment regimen on existing therapy.”
Previous results from the HemACTIVE study showed that, although activity levels differed among hemophilia patients, all patients surveyed wanted greater activity levels, better protection from bleeding, better pain relief, and less-frequent infusions (EAHAD 2019, Abstract P084). In addition, patients who used factor VIII products with an extended half-life were more active and more likely to adhere to their prescribed treatment (ISTH 2019, Abstract PB0210).
The results reported at EAHAD 2020 focus on patients’ reasons for modifying physical activity. Patients and caregivers completed a screening phone interview, followed by a 25-minute, web-based questionnaire on patient activity.
There were 275 respondents – 194 patients with hemophilia A and 81 caregivers – from Canada, France, Germany, Italy, and the United States. Patients had severe (61%) or moderate (39%) hemophilia A, and most (67%) were receiving prophylaxis.
Most patients (70%) were “active” or “extremely/very active,” 77% of patients adjusted their activities because of their hemophilia, and nearly half of patients stopped activities because of their disease.
Fear drives adjustments in activity
Patients were sometimes more likely to adjust their activities based on fear of experiencing an event, as opposed to previously experiencing that event.
Specifically, 44% of patients adjusted their activities due to fear of joint damage, compared with 36% of patients who made adjustments because of past significant joint damage.
Similarly, 41% of patients adjusted activities due to fear of breakthrough bleeds, compared with 36% of patients who made adjustments because of past experience with bleeds and 25% who made adjustments because of significant past bleeds.
On the other hand, a similar percentage of patients adjusted activities because of past experience with pain (43%) and fear of pain (41%). And a similar percentage of patients adjusted activities because of existing joint damage restrictions (35%) and fear of joint deterioration (32%).
Past experience prompts discontinuation of activity
Overall, 47% of patients said anxiety was the most common emotional reason for stopping physical activities. However, patients were consistently more likely to stop activities because of past experience rather than fear or anxiety.
Specifically, 50% of patients stopped activities because of significant past joint damage, 46% stopped because of developing joint problems, and 38% stopped due to fear of joint damage.
More patients stopped activities because of significant past bleeds (41%) rather than fear of breakthrough bleeds (26%). More patients stopped activities because they developed chronic pain (38%) rather than fear of pain (less than 15%). And more patients stopped activities because of existing joint damage restrictions (62%) rather than fear of joint damage (34%).
Applying results to practice: Changing the conversation
Ideally, these findings would be used to promote individualized treatment of hemophilia driven by patients’ goals, Mr. Skinner said. By better understanding patients’ feelings and motivations, clinicians may devise more personalized treatment regimens that align with patients’ goals and improve their quality of life.
Rather than adjusting treatment based only on “hard metrics” such as bleeding events, “we need to take a more holistic approach to looking at outcomes that are more important to patients,” Mr. Skinner said. This type of approach is particularly important to Mr. Skinner as someone who has severe hemophilia A.
“Because hemophilia is a life-long disease, and you’re born with it, you make conscious or unconscious adaptations throughout your life,” he explained. “Your expectations or aspirations adjust to what you’ve been told you can or cannot do because of your hemophilia. The choices I made for my career, where I live, the type of vacations I go on, the type of sports I participate in have all been limited over the course of time, which has meant that I’ve made compromises. There are a lot of individuals with hemophilia who are making decisions that are not what their life goals are.
“What this research helps me understand is that we can change the conversation and build it around an individual patient and understand what their aspirations are. If a clinician understands what I’m wanting to achieve in life … we can build a treatment regime around helping me achieve those goals. That is known to improve adherence.
“The goal, really, is to have hemophilia as a secondary consideration. Instead of saying: ‘You have hemophilia, so these are the options available to you,’ you can say, ‘what is it that you would like to achieve, and then we’ll figure out how your treatment for hemophilia can be adjusted to help you achieve those goals.’ It may sound like a nuance, but it really is reversing the conversation. The goal setting first versus your disease comes first.”
The HemACTIVE study was supported by Bayer. Mr. Skinner disclosed relationships with Bayer and other pharmaceutical companies.
SOURCE: Skinner M et al. EAHAD 2020, Abstract P304.
Fear of negative events can drive changes in activity levels among patients with hemophilia A, results of the HemACTIVE study suggest.
Patients were more likely to adjust their level of physical activity due to fear of bleeding and joint damage rather than previously experienced bleeding or joint damage.
However, past experience was more likely than fear to make patients stop physical activities altogether.
Mark Skinner, of the Institute for Policy Advancement in Washington, D.C., and colleagues presented these findings in a poster from the annual congress of the European Association for Haemophilia and Allied Disorders.
Mr. Skinner, who is a hemophilia patient himself, said the goal of the HemACTIVE study is to better understand how hemophilia affects patients’ lives.
“We wanted to understand the limitations, challenges, and compromises of individuals living with hemophilia,” Mr. Skinner said. “What has motivated them or prevented them from living more full, active lives doing the kind of work, leisure, and social activities that those without hemophilia do? Is it treatment choice, is it satisfaction with treatment, is it fear?
“We wanted to do a comprehensive study that really looked at the intersection of treatment adherence and satisfaction, the emotional components that relate to those decisions, and the challenges and compromises so that we could better identify what we need to consider as patients think about either changing their therapy or changing their treatment regimen on existing therapy.”
Previous results from the HemACTIVE study showed that, although activity levels differed among hemophilia patients, all patients surveyed wanted greater activity levels, better protection from bleeding, better pain relief, and less-frequent infusions (EAHAD 2019, Abstract P084). In addition, patients who used factor VIII products with an extended half-life were more active and more likely to adhere to their prescribed treatment (ISTH 2019, Abstract PB0210).
The results reported at EAHAD 2020 focus on patients’ reasons for modifying physical activity. Patients and caregivers completed a screening phone interview, followed by a 25-minute, web-based questionnaire on patient activity.
There were 275 respondents – 194 patients with hemophilia A and 81 caregivers – from Canada, France, Germany, Italy, and the United States. Patients had severe (61%) or moderate (39%) hemophilia A, and most (67%) were receiving prophylaxis.
Most patients (70%) were “active” or “extremely/very active,” 77% of patients adjusted their activities because of their hemophilia, and nearly half of patients stopped activities because of their disease.
Fear drives adjustments in activity
Patients were sometimes more likely to adjust their activities based on fear of experiencing an event, as opposed to previously experiencing that event.
Specifically, 44% of patients adjusted their activities due to fear of joint damage, compared with 36% of patients who made adjustments because of past significant joint damage.
Similarly, 41% of patients adjusted activities due to fear of breakthrough bleeds, compared with 36% of patients who made adjustments because of past experience with bleeds and 25% who made adjustments because of significant past bleeds.
On the other hand, a similar percentage of patients adjusted activities because of past experience with pain (43%) and fear of pain (41%). And a similar percentage of patients adjusted activities because of existing joint damage restrictions (35%) and fear of joint deterioration (32%).
Past experience prompts discontinuation of activity
Overall, 47% of patients said anxiety was the most common emotional reason for stopping physical activities. However, patients were consistently more likely to stop activities because of past experience rather than fear or anxiety.
Specifically, 50% of patients stopped activities because of significant past joint damage, 46% stopped because of developing joint problems, and 38% stopped due to fear of joint damage.
More patients stopped activities because of significant past bleeds (41%) rather than fear of breakthrough bleeds (26%). More patients stopped activities because they developed chronic pain (38%) rather than fear of pain (less than 15%). And more patients stopped activities because of existing joint damage restrictions (62%) rather than fear of joint damage (34%).
Applying results to practice: Changing the conversation
Ideally, these findings would be used to promote individualized treatment of hemophilia driven by patients’ goals, Mr. Skinner said. By better understanding patients’ feelings and motivations, clinicians may devise more personalized treatment regimens that align with patients’ goals and improve their quality of life.
Rather than adjusting treatment based only on “hard metrics” such as bleeding events, “we need to take a more holistic approach to looking at outcomes that are more important to patients,” Mr. Skinner said. This type of approach is particularly important to Mr. Skinner as someone who has severe hemophilia A.
“Because hemophilia is a life-long disease, and you’re born with it, you make conscious or unconscious adaptations throughout your life,” he explained. “Your expectations or aspirations adjust to what you’ve been told you can or cannot do because of your hemophilia. The choices I made for my career, where I live, the type of vacations I go on, the type of sports I participate in have all been limited over the course of time, which has meant that I’ve made compromises. There are a lot of individuals with hemophilia who are making decisions that are not what their life goals are.
“What this research helps me understand is that we can change the conversation and build it around an individual patient and understand what their aspirations are. If a clinician understands what I’m wanting to achieve in life … we can build a treatment regime around helping me achieve those goals. That is known to improve adherence.
“The goal, really, is to have hemophilia as a secondary consideration. Instead of saying: ‘You have hemophilia, so these are the options available to you,’ you can say, ‘what is it that you would like to achieve, and then we’ll figure out how your treatment for hemophilia can be adjusted to help you achieve those goals.’ It may sound like a nuance, but it really is reversing the conversation. The goal setting first versus your disease comes first.”
The HemACTIVE study was supported by Bayer. Mr. Skinner disclosed relationships with Bayer and other pharmaceutical companies.
SOURCE: Skinner M et al. EAHAD 2020, Abstract P304.
REPORTING FROM EAHAD 2020
Cancer research foundation awards ‘breakthrough scientists’
The Damon Runyon Cancer Research Foundation has awarded $100,000 each to six “breakthrough scientists.”
The Damon Runyon–Dale F. Frey Award for Breakthrough Scientists provides additional funding to scientists completing a Damon Runyon Fellowship Award who “are most likely to make paradigm-shifting breakthroughs that transform the way we prevent, diagnose, and treat cancer,” according to the foundation announcement.
Lindsay B. Case, PhD, of the University of Texas Southwestern Medical Center, Dallas, received the award for her research investigating the molecular interactions that contribute to integrin signaling and focal adhesion function. This work could reveal new strategies for disrupting integrin signaling in cancer.
Ivana Gasic, DrSc, of Harvard Medical School, Boston, was awarded for her research on tubulin autoregulation and the microtubule integrity response. Her research could provide insight into the workings of microtubule-targeting chemotherapeutic drugs and reveal new pathways to target cancer cells.
Natasha M. O’Brown, PhD, of Harvard Medical School, Boston, was awarded for her research using CRISPR technology and zebrafish to investigate the molecular mechanisms that regulate the blood-brain barrier. This work could reveal new approaches to drug delivery for patients with brain tumors.
Benjamin M. Stinson, PhD, of Harvard Medical School, Boston, was awarded for his work investigating the mechanisms of two main DNA double-strand break repair pathways. This research could shed light on the causes of cancers and have applications for cancer treatment.
Iva A. Tchasovnikarova, PhD, of Massachusetts General Hospital, Boston, was awarded for epigenetics research that may reveal targets for cancer therapy. She developed a method to identify cell-based reporters of any epigenetic process inside the nucleus and aims to use this method to better understand the biology underlying epigenetic mechanisms.
Yi Yin, PhD, of the University of California, Los Angeles, was awarded for work that may help inform the treatment of many cancers. Dr. Yin developed single-cell assays that she will combine with statistical modeling to better understand homologous recombination.
The Damon Runyon Cancer Research Foundation has awarded $100,000 each to six “breakthrough scientists.”
The Damon Runyon–Dale F. Frey Award for Breakthrough Scientists provides additional funding to scientists completing a Damon Runyon Fellowship Award who “are most likely to make paradigm-shifting breakthroughs that transform the way we prevent, diagnose, and treat cancer,” according to the foundation announcement.
Lindsay B. Case, PhD, of the University of Texas Southwestern Medical Center, Dallas, received the award for her research investigating the molecular interactions that contribute to integrin signaling and focal adhesion function. This work could reveal new strategies for disrupting integrin signaling in cancer.
Ivana Gasic, DrSc, of Harvard Medical School, Boston, was awarded for her research on tubulin autoregulation and the microtubule integrity response. Her research could provide insight into the workings of microtubule-targeting chemotherapeutic drugs and reveal new pathways to target cancer cells.
Natasha M. O’Brown, PhD, of Harvard Medical School, Boston, was awarded for her research using CRISPR technology and zebrafish to investigate the molecular mechanisms that regulate the blood-brain barrier. This work could reveal new approaches to drug delivery for patients with brain tumors.
Benjamin M. Stinson, PhD, of Harvard Medical School, Boston, was awarded for his work investigating the mechanisms of two main DNA double-strand break repair pathways. This research could shed light on the causes of cancers and have applications for cancer treatment.
Iva A. Tchasovnikarova, PhD, of Massachusetts General Hospital, Boston, was awarded for epigenetics research that may reveal targets for cancer therapy. She developed a method to identify cell-based reporters of any epigenetic process inside the nucleus and aims to use this method to better understand the biology underlying epigenetic mechanisms.
Yi Yin, PhD, of the University of California, Los Angeles, was awarded for work that may help inform the treatment of many cancers. Dr. Yin developed single-cell assays that she will combine with statistical modeling to better understand homologous recombination.
The Damon Runyon Cancer Research Foundation has awarded $100,000 each to six “breakthrough scientists.”
The Damon Runyon–Dale F. Frey Award for Breakthrough Scientists provides additional funding to scientists completing a Damon Runyon Fellowship Award who “are most likely to make paradigm-shifting breakthroughs that transform the way we prevent, diagnose, and treat cancer,” according to the foundation announcement.
Lindsay B. Case, PhD, of the University of Texas Southwestern Medical Center, Dallas, received the award for her research investigating the molecular interactions that contribute to integrin signaling and focal adhesion function. This work could reveal new strategies for disrupting integrin signaling in cancer.
Ivana Gasic, DrSc, of Harvard Medical School, Boston, was awarded for her research on tubulin autoregulation and the microtubule integrity response. Her research could provide insight into the workings of microtubule-targeting chemotherapeutic drugs and reveal new pathways to target cancer cells.
Natasha M. O’Brown, PhD, of Harvard Medical School, Boston, was awarded for her research using CRISPR technology and zebrafish to investigate the molecular mechanisms that regulate the blood-brain barrier. This work could reveal new approaches to drug delivery for patients with brain tumors.
Benjamin M. Stinson, PhD, of Harvard Medical School, Boston, was awarded for his work investigating the mechanisms of two main DNA double-strand break repair pathways. This research could shed light on the causes of cancers and have applications for cancer treatment.
Iva A. Tchasovnikarova, PhD, of Massachusetts General Hospital, Boston, was awarded for epigenetics research that may reveal targets for cancer therapy. She developed a method to identify cell-based reporters of any epigenetic process inside the nucleus and aims to use this method to better understand the biology underlying epigenetic mechanisms.
Yi Yin, PhD, of the University of California, Los Angeles, was awarded for work that may help inform the treatment of many cancers. Dr. Yin developed single-cell assays that she will combine with statistical modeling to better understand homologous recombination.
Social media may negatively influence acne treatment
A small survey suggests many patients consult social media for advice on acne treatment and follow recommendations that don’t align with clinical guidelines.
Of the 130 patients surveyed, 45% consulted social media for advice on acne treatment, and 52% of those patients followed recommendations that don’t correspond to American Academy of Dermatology (AAD) guidelines. Most patients reported no improvement (40%) or minimal improvement (53%) in their acne after following advice from social media.
“These results suggest that dermatologists should inquire about social media acne treatment advice and directly address misinformation,” wrote Ahmed Yousaf, of West Virginia University, Morgantown, W.Va., and colleagues. Their report is in Pediatric Dermatology.
They conducted the survey of 130 patients treated for acne at West Virginia University. Most patients were female (60%), and a majority were adolescents (54%) or adults (44%). About half of the patients (51%) said their acne was moderate, 38% said it was severe, and 11% said it was mild.
Most patients said they consulted a medical professional for their first acne treatment (58%). However, 16% of patients said they first went to social media for advice, 26% said they consulted family or friends, and 10% took “other” steps as their first approach to acne treatment.
In all, 45% of patients consulted social media for acne treatment advice at some point. This includes 54% of women, 31% of men, 41% of adolescents, and 51% of adults. Social media consultation was more common among patients with severe acne (54%) than among those with mild (36%) or moderate (39%) acne.
The most common social media platforms used were YouTube and Instagram (58% each), followed by Pinterest (31%), Facebook (19%), Twitter (9%), Snapchat (7%), and Tumblr (3%). (Patients could select more than one social media platform.)
Roughly half (52%) of patients who consulted social media followed advice that does not align with AAD guidelines, 31% made changes that are recommended by the AAD, and 17% did not provide information on recommendations they followed.
The social media advice patients followed included using over-the-counter products (81%), making dietary changes (40%), using self-made products (19%), taking supplements (16%), and making changes in exercise routines (7%). (Patients could select more than one treatment approach.)
Among the patients who followed social media advice, 40% said they saw no change in their acne, and 53% reported minimal improvement.
“Only 7% of social media users reported significant improvement in their acne,” Mr. Yousaf and colleagues wrote. “This may be due to less accurate content found on social media compared to other health care sources.”
The authors acknowledged that the patients surveyed were recruited from a dermatology clinic. Therefore, these results “likely underestimate the percentage of patients who improve from social media acne treatment advice and do not consult a medical professional.”
Mr. Yousaf and colleagues did not disclose any conflicts of interest.
SOURCE: Yousaf A et al. Pediatr Dermatol. 2020 Jan 15. doi: 10.1111/pde.14091.
A small survey suggests many patients consult social media for advice on acne treatment and follow recommendations that don’t align with clinical guidelines.
Of the 130 patients surveyed, 45% consulted social media for advice on acne treatment, and 52% of those patients followed recommendations that don’t correspond to American Academy of Dermatology (AAD) guidelines. Most patients reported no improvement (40%) or minimal improvement (53%) in their acne after following advice from social media.
“These results suggest that dermatologists should inquire about social media acne treatment advice and directly address misinformation,” wrote Ahmed Yousaf, of West Virginia University, Morgantown, W.Va., and colleagues. Their report is in Pediatric Dermatology.
They conducted the survey of 130 patients treated for acne at West Virginia University. Most patients were female (60%), and a majority were adolescents (54%) or adults (44%). About half of the patients (51%) said their acne was moderate, 38% said it was severe, and 11% said it was mild.
Most patients said they consulted a medical professional for their first acne treatment (58%). However, 16% of patients said they first went to social media for advice, 26% said they consulted family or friends, and 10% took “other” steps as their first approach to acne treatment.
In all, 45% of patients consulted social media for acne treatment advice at some point. This includes 54% of women, 31% of men, 41% of adolescents, and 51% of adults. Social media consultation was more common among patients with severe acne (54%) than among those with mild (36%) or moderate (39%) acne.
The most common social media platforms used were YouTube and Instagram (58% each), followed by Pinterest (31%), Facebook (19%), Twitter (9%), Snapchat (7%), and Tumblr (3%). (Patients could select more than one social media platform.)
Roughly half (52%) of patients who consulted social media followed advice that does not align with AAD guidelines, 31% made changes that are recommended by the AAD, and 17% did not provide information on recommendations they followed.
The social media advice patients followed included using over-the-counter products (81%), making dietary changes (40%), using self-made products (19%), taking supplements (16%), and making changes in exercise routines (7%). (Patients could select more than one treatment approach.)
Among the patients who followed social media advice, 40% said they saw no change in their acne, and 53% reported minimal improvement.
“Only 7% of social media users reported significant improvement in their acne,” Mr. Yousaf and colleagues wrote. “This may be due to less accurate content found on social media compared to other health care sources.”
The authors acknowledged that the patients surveyed were recruited from a dermatology clinic. Therefore, these results “likely underestimate the percentage of patients who improve from social media acne treatment advice and do not consult a medical professional.”
Mr. Yousaf and colleagues did not disclose any conflicts of interest.
SOURCE: Yousaf A et al. Pediatr Dermatol. 2020 Jan 15. doi: 10.1111/pde.14091.
A small survey suggests many patients consult social media for advice on acne treatment and follow recommendations that don’t align with clinical guidelines.
Of the 130 patients surveyed, 45% consulted social media for advice on acne treatment, and 52% of those patients followed recommendations that don’t correspond to American Academy of Dermatology (AAD) guidelines. Most patients reported no improvement (40%) or minimal improvement (53%) in their acne after following advice from social media.
“These results suggest that dermatologists should inquire about social media acne treatment advice and directly address misinformation,” wrote Ahmed Yousaf, of West Virginia University, Morgantown, W.Va., and colleagues. Their report is in Pediatric Dermatology.
They conducted the survey of 130 patients treated for acne at West Virginia University. Most patients were female (60%), and a majority were adolescents (54%) or adults (44%). About half of the patients (51%) said their acne was moderate, 38% said it was severe, and 11% said it was mild.
Most patients said they consulted a medical professional for their first acne treatment (58%). However, 16% of patients said they first went to social media for advice, 26% said they consulted family or friends, and 10% took “other” steps as their first approach to acne treatment.
In all, 45% of patients consulted social media for acne treatment advice at some point. This includes 54% of women, 31% of men, 41% of adolescents, and 51% of adults. Social media consultation was more common among patients with severe acne (54%) than among those with mild (36%) or moderate (39%) acne.
The most common social media platforms used were YouTube and Instagram (58% each), followed by Pinterest (31%), Facebook (19%), Twitter (9%), Snapchat (7%), and Tumblr (3%). (Patients could select more than one social media platform.)
Roughly half (52%) of patients who consulted social media followed advice that does not align with AAD guidelines, 31% made changes that are recommended by the AAD, and 17% did not provide information on recommendations they followed.
The social media advice patients followed included using over-the-counter products (81%), making dietary changes (40%), using self-made products (19%), taking supplements (16%), and making changes in exercise routines (7%). (Patients could select more than one treatment approach.)
Among the patients who followed social media advice, 40% said they saw no change in their acne, and 53% reported minimal improvement.
“Only 7% of social media users reported significant improvement in their acne,” Mr. Yousaf and colleagues wrote. “This may be due to less accurate content found on social media compared to other health care sources.”
The authors acknowledged that the patients surveyed were recruited from a dermatology clinic. Therefore, these results “likely underestimate the percentage of patients who improve from social media acne treatment advice and do not consult a medical professional.”
Mr. Yousaf and colleagues did not disclose any conflicts of interest.
SOURCE: Yousaf A et al. Pediatr Dermatol. 2020 Jan 15. doi: 10.1111/pde.14091.
FROM PEDIATRIC DERMATOLOGY
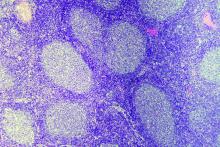
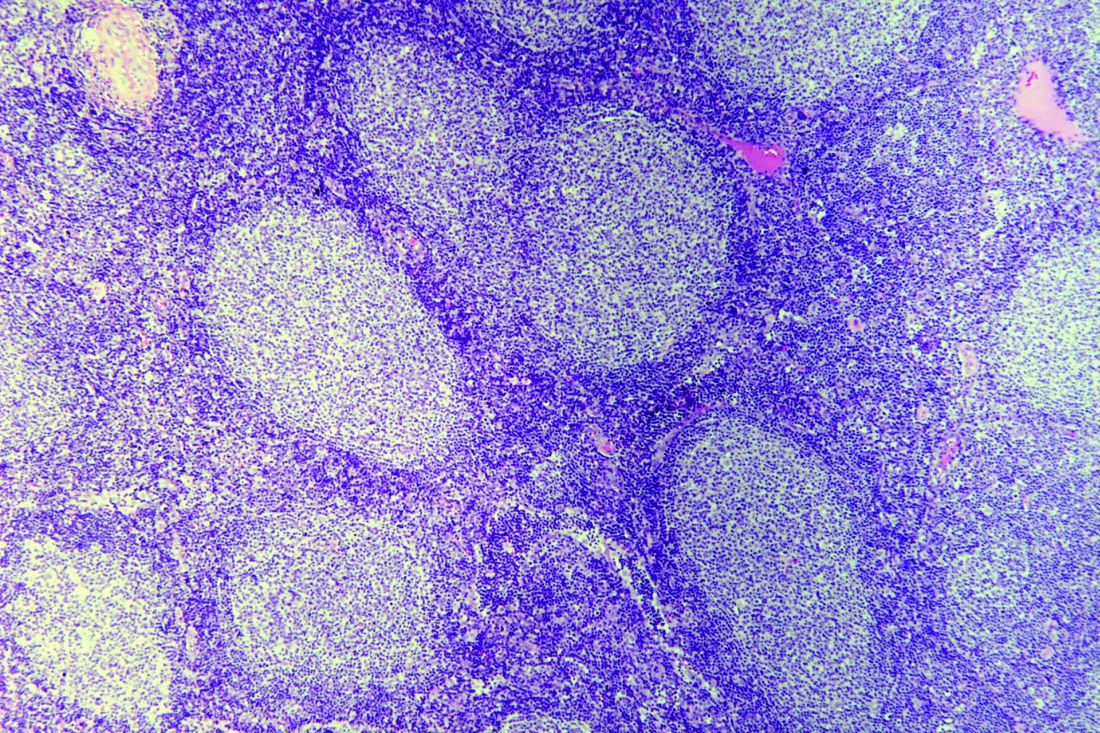
ECHELON-1 update: A+AVD bests ABVD in Hodgkin lymphoma
Brentuximab vedotin plus doxorubicin, vinblastine, and dacarbazine (A+AVD) provides “robust, sustained efficacy” in patients with Hodgkin lymphoma, according to investigators.
In the ECHELON-1 trial, investigators compared A+AVD to doxorubicin, bleomycin, vinblastine, and dacarbazine (ABVD) as frontline treatment for stage III or IV Hodgkin lymphoma. The 3-year progression-free survival (PFS) was superior in patients who received A+AVD, and this benefit was seen across most subgroups.
David J. Straus, MD, of Memorial Sloan Kettering Cancer Center in New York and his colleagues detailed these findings in Blood.
The phase 3 trial (NCT01712490) enrolled 1,334 patients with stage III or IV classical Hodgkin lymphoma. They were randomized to receive A+AVD (n = 664) or ABVD (n = 670). Baseline characteristics were similar between the treatment arms.
Positron emission tomography status after cycle 2 (PET2) was similar between the treatment arms as well. Most patients – 89% of the A+AVD arm and 86% of the ABVD arm – were PET2 negative. Treating physicians used PET2 status as a guide to potentially switch patients to an alternative regimen (radiotherapy or chemotherapy with or without transplant).
In a prior analysis, the study’s primary endpoint was modified PFS (time to progression, death, or noncomplete response after frontline therapy) per an independent review committee (N Engl J Med. 2018;378:331-44). The 2-year modified PFS rate was 82.1% in the A+AVD arm and 77.2% in the ABVD arm (hazard ratio, 0.77; P = .04).
PFS update
In the current analysis, the main exploratory endpoint was PFS per investigator. The 3-year PFS rate was significantly higher in the A+AVD arm than in the ABVD arm – 83.1% and 76.0%, respectively (HR, 0.704; P = .005).
The investigators observed a “consistent improvement in PFS” in the A+AVD arm, regardless of disease stage, International Prognostic score, Eastern Cooperative Oncology Group status, sex, or age. There was a significant improvement in PFS with A+AVD in PET2-negative patients and a trend toward improvement in PET2-positive patients. In the PET2-negative patients, the 3-year PFS was 85.8% in the A+AVD arm and 79.5% in the ABVD arm (HR, 0.69; P = .009). In PET2-positive patients, the 3-year PFS was 67.7% and 51.5%, respectively (HR, 0.59; P = .077).
“These data highlight that A+AVD provides a durable efficacy benefit, compared with ABVD, for frontline stage III/IV cHL [classical Hodgkin lymphoma], which is consistent across key subgroups regardless of patient status at PET2,” Dr. Straus and his colleagues wrote.
Safety update
In both treatment arms, peripheral neuropathy continued to improve or resolve with longer follow-up. Among patients who developed peripheral neuropathy, 78% in the A+AVD arm and 83% in the ABVD arm had improvement or resolution of the condition at 3 years.
Most patients had complete resolution of peripheral neuropathy; 62% in the A+AVD arm and 73% in the ABVD arm. The median time to complete resolution was 28 weeks (range, 0-167 weeks) after stopping A+AVD and 14 weeks (range, 0-188 weeks) after stopping ABVD.
The incidence of secondary malignancies was similar between the treatment arms. There were 14 secondary malignancies in the A+AVD arm (6 solid tumors, 8 hematologic malignancies) and 20 in the ABVD arm (9 solid tumors, 11 hematologic malignancies).
“A+AVD provided a sustained PFS benefit with a predictable and manageable safety profile,” Dr. Straus and colleagues wrote. “These data further support the advantages of A+AVD versus ABVD as frontline treatment of patients with advanced stage III or IV cHL [classical Hodgkin lymphoma].”
The ECHELON-1 trial was sponsored by Millennium Pharmaceuticals (a subsidiary of Takeda) and Seattle Genetics. The investigators disclosed relationships with Millennium, Takeda, Seattle Genetics, and a range of other companies.
SOURCE: Straus DJ et al. Blood. 2020 Jan 16. pii: blood.2019003127. doi: 10.1182/blood.2019003127.
Brentuximab vedotin plus doxorubicin, vinblastine, and dacarbazine (A+AVD) provides “robust, sustained efficacy” in patients with Hodgkin lymphoma, according to investigators.
In the ECHELON-1 trial, investigators compared A+AVD to doxorubicin, bleomycin, vinblastine, and dacarbazine (ABVD) as frontline treatment for stage III or IV Hodgkin lymphoma. The 3-year progression-free survival (PFS) was superior in patients who received A+AVD, and this benefit was seen across most subgroups.
David J. Straus, MD, of Memorial Sloan Kettering Cancer Center in New York and his colleagues detailed these findings in Blood.
The phase 3 trial (NCT01712490) enrolled 1,334 patients with stage III or IV classical Hodgkin lymphoma. They were randomized to receive A+AVD (n = 664) or ABVD (n = 670). Baseline characteristics were similar between the treatment arms.
Positron emission tomography status after cycle 2 (PET2) was similar between the treatment arms as well. Most patients – 89% of the A+AVD arm and 86% of the ABVD arm – were PET2 negative. Treating physicians used PET2 status as a guide to potentially switch patients to an alternative regimen (radiotherapy or chemotherapy with or without transplant).
In a prior analysis, the study’s primary endpoint was modified PFS (time to progression, death, or noncomplete response after frontline therapy) per an independent review committee (N Engl J Med. 2018;378:331-44). The 2-year modified PFS rate was 82.1% in the A+AVD arm and 77.2% in the ABVD arm (hazard ratio, 0.77; P = .04).
PFS update
In the current analysis, the main exploratory endpoint was PFS per investigator. The 3-year PFS rate was significantly higher in the A+AVD arm than in the ABVD arm – 83.1% and 76.0%, respectively (HR, 0.704; P = .005).
The investigators observed a “consistent improvement in PFS” in the A+AVD arm, regardless of disease stage, International Prognostic score, Eastern Cooperative Oncology Group status, sex, or age. There was a significant improvement in PFS with A+AVD in PET2-negative patients and a trend toward improvement in PET2-positive patients. In the PET2-negative patients, the 3-year PFS was 85.8% in the A+AVD arm and 79.5% in the ABVD arm (HR, 0.69; P = .009). In PET2-positive patients, the 3-year PFS was 67.7% and 51.5%, respectively (HR, 0.59; P = .077).
“These data highlight that A+AVD provides a durable efficacy benefit, compared with ABVD, for frontline stage III/IV cHL [classical Hodgkin lymphoma], which is consistent across key subgroups regardless of patient status at PET2,” Dr. Straus and his colleagues wrote.
Safety update
In both treatment arms, peripheral neuropathy continued to improve or resolve with longer follow-up. Among patients who developed peripheral neuropathy, 78% in the A+AVD arm and 83% in the ABVD arm had improvement or resolution of the condition at 3 years.
Most patients had complete resolution of peripheral neuropathy; 62% in the A+AVD arm and 73% in the ABVD arm. The median time to complete resolution was 28 weeks (range, 0-167 weeks) after stopping A+AVD and 14 weeks (range, 0-188 weeks) after stopping ABVD.
The incidence of secondary malignancies was similar between the treatment arms. There were 14 secondary malignancies in the A+AVD arm (6 solid tumors, 8 hematologic malignancies) and 20 in the ABVD arm (9 solid tumors, 11 hematologic malignancies).
“A+AVD provided a sustained PFS benefit with a predictable and manageable safety profile,” Dr. Straus and colleagues wrote. “These data further support the advantages of A+AVD versus ABVD as frontline treatment of patients with advanced stage III or IV cHL [classical Hodgkin lymphoma].”
The ECHELON-1 trial was sponsored by Millennium Pharmaceuticals (a subsidiary of Takeda) and Seattle Genetics. The investigators disclosed relationships with Millennium, Takeda, Seattle Genetics, and a range of other companies.
SOURCE: Straus DJ et al. Blood. 2020 Jan 16. pii: blood.2019003127. doi: 10.1182/blood.2019003127.
Brentuximab vedotin plus doxorubicin, vinblastine, and dacarbazine (A+AVD) provides “robust, sustained efficacy” in patients with Hodgkin lymphoma, according to investigators.
In the ECHELON-1 trial, investigators compared A+AVD to doxorubicin, bleomycin, vinblastine, and dacarbazine (ABVD) as frontline treatment for stage III or IV Hodgkin lymphoma. The 3-year progression-free survival (PFS) was superior in patients who received A+AVD, and this benefit was seen across most subgroups.
David J. Straus, MD, of Memorial Sloan Kettering Cancer Center in New York and his colleagues detailed these findings in Blood.
The phase 3 trial (NCT01712490) enrolled 1,334 patients with stage III or IV classical Hodgkin lymphoma. They were randomized to receive A+AVD (n = 664) or ABVD (n = 670). Baseline characteristics were similar between the treatment arms.
Positron emission tomography status after cycle 2 (PET2) was similar between the treatment arms as well. Most patients – 89% of the A+AVD arm and 86% of the ABVD arm – were PET2 negative. Treating physicians used PET2 status as a guide to potentially switch patients to an alternative regimen (radiotherapy or chemotherapy with or without transplant).
In a prior analysis, the study’s primary endpoint was modified PFS (time to progression, death, or noncomplete response after frontline therapy) per an independent review committee (N Engl J Med. 2018;378:331-44). The 2-year modified PFS rate was 82.1% in the A+AVD arm and 77.2% in the ABVD arm (hazard ratio, 0.77; P = .04).
PFS update
In the current analysis, the main exploratory endpoint was PFS per investigator. The 3-year PFS rate was significantly higher in the A+AVD arm than in the ABVD arm – 83.1% and 76.0%, respectively (HR, 0.704; P = .005).
The investigators observed a “consistent improvement in PFS” in the A+AVD arm, regardless of disease stage, International Prognostic score, Eastern Cooperative Oncology Group status, sex, or age. There was a significant improvement in PFS with A+AVD in PET2-negative patients and a trend toward improvement in PET2-positive patients. In the PET2-negative patients, the 3-year PFS was 85.8% in the A+AVD arm and 79.5% in the ABVD arm (HR, 0.69; P = .009). In PET2-positive patients, the 3-year PFS was 67.7% and 51.5%, respectively (HR, 0.59; P = .077).
“These data highlight that A+AVD provides a durable efficacy benefit, compared with ABVD, for frontline stage III/IV cHL [classical Hodgkin lymphoma], which is consistent across key subgroups regardless of patient status at PET2,” Dr. Straus and his colleagues wrote.
Safety update
In both treatment arms, peripheral neuropathy continued to improve or resolve with longer follow-up. Among patients who developed peripheral neuropathy, 78% in the A+AVD arm and 83% in the ABVD arm had improvement or resolution of the condition at 3 years.
Most patients had complete resolution of peripheral neuropathy; 62% in the A+AVD arm and 73% in the ABVD arm. The median time to complete resolution was 28 weeks (range, 0-167 weeks) after stopping A+AVD and 14 weeks (range, 0-188 weeks) after stopping ABVD.
The incidence of secondary malignancies was similar between the treatment arms. There were 14 secondary malignancies in the A+AVD arm (6 solid tumors, 8 hematologic malignancies) and 20 in the ABVD arm (9 solid tumors, 11 hematologic malignancies).
“A+AVD provided a sustained PFS benefit with a predictable and manageable safety profile,” Dr. Straus and colleagues wrote. “These data further support the advantages of A+AVD versus ABVD as frontline treatment of patients with advanced stage III or IV cHL [classical Hodgkin lymphoma].”
The ECHELON-1 trial was sponsored by Millennium Pharmaceuticals (a subsidiary of Takeda) and Seattle Genetics. The investigators disclosed relationships with Millennium, Takeda, Seattle Genetics, and a range of other companies.
SOURCE: Straus DJ et al. Blood. 2020 Jan 16. pii: blood.2019003127. doi: 10.1182/blood.2019003127.
FROM BLOOD
ASCO issues guidelines on genetic testing in epithelial ovarian cancer
In new guidelines, the American Society of Clinical Oncology recommends offering germline genetic testing for BRCA1, BRCA2, and other ovarian cancer susceptibility genes to all women diagnosed with epithelial ovarian cancer, regardless of their clinical features or family history.
Testing should be offered at diagnosis or as soon as possible after that, Panagiotis A. Konstantinopoulos, MD, PhD, of the Dana-Farber Cancer Institute in Boston, and colleagues wrote in the Journal of Clinical Oncology.
For patients who do not carry a germline pathogenic or likely pathogenic BRCA1/2 variant, the guidelines recommend offering somatic tumor testing for BRCA1/2 pathogenic or likely pathogenic variants. This testing can be offered at the time of disease recurrence after up-front therapy.
The guidelines also recommend somatic tumor testing for mismatch repair deficiency in patients diagnosed with clear cell, endometrioid, or mucinous ovarian cancer. This testing may be offered to patients with other histologic types of epithelial ovarian cancer as well.
Genetic testing, as well as genetic risk evaluation and counseling, should be offered to first- or second-degree blood relatives of a patient with ovarian cancer and a known germline pathogenic cancer susceptibility gene variant, according to the guidelines.
According to the guidelines, genetic evaluations should be conducted in cooperation with other health care providers who are “familiar with the diagnosis and management of hereditary cancer syndromes to determine the most appropriate testing strategy and discuss implications of the findings.”
Patients with identified germline or somatic pathogenic or likely pathogenic BRCA1/2 variants should receive treatments approved for them by the Food and Drug Administration, according to the guidelines. The authors note that patients with these variants have responded well to FDA-approved poly (ADP-ribose) polymerase inhibitors, including niraparib (Zejula), olaparib (Lynparza), and rucaparib (Rubraca).
The guidelines also state that mismatch repair deficiency qualifies for FDA-approved treatment, so patients with recurrent epithelial ovarian cancer and mismatch repair deficiency should receive FDA-approved treatments under their labeled indications.
The guidelines note that clinical decisions should not be based on a variant of uncertain significance. When a patient has such a variant, “clinical features and family history should inform clinical decision making,” according to the guidelines.
Dr. Konstantinopoulos and colleagues formulated the guidelines after reviewing data from 19 studies, including 6 meta-analyses; 11 randomized, controlled trials; and 2 observational studies.
The authors reported relationships with a range of pharmaceutical companies, including those that market drugs for epithelial ovarian cancer.
SOURCE: Konstantinopoulos PA et al. J Clin Oncol. 2020 Jan 27. doi: 10.1200/JCO.19.02960.
In new guidelines, the American Society of Clinical Oncology recommends offering germline genetic testing for BRCA1, BRCA2, and other ovarian cancer susceptibility genes to all women diagnosed with epithelial ovarian cancer, regardless of their clinical features or family history.
Testing should be offered at diagnosis or as soon as possible after that, Panagiotis A. Konstantinopoulos, MD, PhD, of the Dana-Farber Cancer Institute in Boston, and colleagues wrote in the Journal of Clinical Oncology.
For patients who do not carry a germline pathogenic or likely pathogenic BRCA1/2 variant, the guidelines recommend offering somatic tumor testing for BRCA1/2 pathogenic or likely pathogenic variants. This testing can be offered at the time of disease recurrence after up-front therapy.
The guidelines also recommend somatic tumor testing for mismatch repair deficiency in patients diagnosed with clear cell, endometrioid, or mucinous ovarian cancer. This testing may be offered to patients with other histologic types of epithelial ovarian cancer as well.
Genetic testing, as well as genetic risk evaluation and counseling, should be offered to first- or second-degree blood relatives of a patient with ovarian cancer and a known germline pathogenic cancer susceptibility gene variant, according to the guidelines.
According to the guidelines, genetic evaluations should be conducted in cooperation with other health care providers who are “familiar with the diagnosis and management of hereditary cancer syndromes to determine the most appropriate testing strategy and discuss implications of the findings.”
Patients with identified germline or somatic pathogenic or likely pathogenic BRCA1/2 variants should receive treatments approved for them by the Food and Drug Administration, according to the guidelines. The authors note that patients with these variants have responded well to FDA-approved poly (ADP-ribose) polymerase inhibitors, including niraparib (Zejula), olaparib (Lynparza), and rucaparib (Rubraca).
The guidelines also state that mismatch repair deficiency qualifies for FDA-approved treatment, so patients with recurrent epithelial ovarian cancer and mismatch repair deficiency should receive FDA-approved treatments under their labeled indications.
The guidelines note that clinical decisions should not be based on a variant of uncertain significance. When a patient has such a variant, “clinical features and family history should inform clinical decision making,” according to the guidelines.
Dr. Konstantinopoulos and colleagues formulated the guidelines after reviewing data from 19 studies, including 6 meta-analyses; 11 randomized, controlled trials; and 2 observational studies.
The authors reported relationships with a range of pharmaceutical companies, including those that market drugs for epithelial ovarian cancer.
SOURCE: Konstantinopoulos PA et al. J Clin Oncol. 2020 Jan 27. doi: 10.1200/JCO.19.02960.
In new guidelines, the American Society of Clinical Oncology recommends offering germline genetic testing for BRCA1, BRCA2, and other ovarian cancer susceptibility genes to all women diagnosed with epithelial ovarian cancer, regardless of their clinical features or family history.
Testing should be offered at diagnosis or as soon as possible after that, Panagiotis A. Konstantinopoulos, MD, PhD, of the Dana-Farber Cancer Institute in Boston, and colleagues wrote in the Journal of Clinical Oncology.
For patients who do not carry a germline pathogenic or likely pathogenic BRCA1/2 variant, the guidelines recommend offering somatic tumor testing for BRCA1/2 pathogenic or likely pathogenic variants. This testing can be offered at the time of disease recurrence after up-front therapy.
The guidelines also recommend somatic tumor testing for mismatch repair deficiency in patients diagnosed with clear cell, endometrioid, or mucinous ovarian cancer. This testing may be offered to patients with other histologic types of epithelial ovarian cancer as well.
Genetic testing, as well as genetic risk evaluation and counseling, should be offered to first- or second-degree blood relatives of a patient with ovarian cancer and a known germline pathogenic cancer susceptibility gene variant, according to the guidelines.
According to the guidelines, genetic evaluations should be conducted in cooperation with other health care providers who are “familiar with the diagnosis and management of hereditary cancer syndromes to determine the most appropriate testing strategy and discuss implications of the findings.”
Patients with identified germline or somatic pathogenic or likely pathogenic BRCA1/2 variants should receive treatments approved for them by the Food and Drug Administration, according to the guidelines. The authors note that patients with these variants have responded well to FDA-approved poly (ADP-ribose) polymerase inhibitors, including niraparib (Zejula), olaparib (Lynparza), and rucaparib (Rubraca).
The guidelines also state that mismatch repair deficiency qualifies for FDA-approved treatment, so patients with recurrent epithelial ovarian cancer and mismatch repair deficiency should receive FDA-approved treatments under their labeled indications.
The guidelines note that clinical decisions should not be based on a variant of uncertain significance. When a patient has such a variant, “clinical features and family history should inform clinical decision making,” according to the guidelines.
Dr. Konstantinopoulos and colleagues formulated the guidelines after reviewing data from 19 studies, including 6 meta-analyses; 11 randomized, controlled trials; and 2 observational studies.
The authors reported relationships with a range of pharmaceutical companies, including those that market drugs for epithelial ovarian cancer.
SOURCE: Konstantinopoulos PA et al. J Clin Oncol. 2020 Jan 27. doi: 10.1200/JCO.19.02960.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Antimalarial adherence is important for diabetes prevention in lupus
Adhering to antimalarial treatment offers some protection to patients with systemic lupus erythematosus (SLE) from developing type 2 diabetes mellitus (T2DM), according to new research.
Patients who took at least 90% of their prescribed antimalarial doses were 39% less likely to develop T2DM than patients who discontinued antimalarial therapy. Patients who took less than 90% of their prescribed doses but didn’t discontinue treatment were 22% less likely to develop T2DM.
“[O]ur study provides further support for the importance of adherence to antimalarials in SLE by demonstrating protective impacts on T2DM,” Shahrzad Salmasi, PhD, of the University of British Columbia, Vancouver, and colleagues wrote in Arthritis Care & Research.
Dr. Salmasi and colleagues conducted this retrospective study using administrative health data on patients in British Columbia. The researchers analyzed 1,498 patients with SLE. Their mean age was about 44 years, and 91% were women.
The researchers used data on prescription dates and days’ supply to establish antimalarial drug courses and gaps in treatment. A new treatment course occurred when a 90-day gap was exceeded between refills. The researchers calculated the proportion of days covered (PDC) – the total number of days with antimalarials divided by the length of the course – and separated patients into three categories:
- Adherent to treatment – PDC of 0.90 or greater
- Nonadherent – PDC greater than 0 but less than 0.90
- Discontinuer – PDC of 0
The patients had a mean of about 23 antimalarial prescriptions and a mean of about two courses. The mean course duration was 554 days.
At a median follow-up of 4.6 years, there were 140 incident cases of T2DM. The researchers calculated the risk of T2DM among adherent and nonadherent patients, comparing these groups with the discontinuers and adjusting for age, sex, comorbidities, and concomitant medications.
The adjusted hazard ratio for developing T2DM was 0.61 among adherent patients and 0.78 among nonadherent patients.
“This population-based study highlighted that taking less than 90% of the prescribed antimalarials compromises their effect in preventing T2DM in SLE patients,” Dr. Salmasi and colleagues wrote. “Our findings should be used to emphasize the importance of medication adherence in not only treating SLE but also preventing its complications.”
The researchers reported having no conflicts of interest.
SOURCE: Salmasi S et al. Arthritis Care Res. 2020 Jan 21. doi: 10.1002/acr.24147.
Adhering to antimalarial treatment offers some protection to patients with systemic lupus erythematosus (SLE) from developing type 2 diabetes mellitus (T2DM), according to new research.
Patients who took at least 90% of their prescribed antimalarial doses were 39% less likely to develop T2DM than patients who discontinued antimalarial therapy. Patients who took less than 90% of their prescribed doses but didn’t discontinue treatment were 22% less likely to develop T2DM.
“[O]ur study provides further support for the importance of adherence to antimalarials in SLE by demonstrating protective impacts on T2DM,” Shahrzad Salmasi, PhD, of the University of British Columbia, Vancouver, and colleagues wrote in Arthritis Care & Research.
Dr. Salmasi and colleagues conducted this retrospective study using administrative health data on patients in British Columbia. The researchers analyzed 1,498 patients with SLE. Their mean age was about 44 years, and 91% were women.
The researchers used data on prescription dates and days’ supply to establish antimalarial drug courses and gaps in treatment. A new treatment course occurred when a 90-day gap was exceeded between refills. The researchers calculated the proportion of days covered (PDC) – the total number of days with antimalarials divided by the length of the course – and separated patients into three categories:
- Adherent to treatment – PDC of 0.90 or greater
- Nonadherent – PDC greater than 0 but less than 0.90
- Discontinuer – PDC of 0
The patients had a mean of about 23 antimalarial prescriptions and a mean of about two courses. The mean course duration was 554 days.
At a median follow-up of 4.6 years, there were 140 incident cases of T2DM. The researchers calculated the risk of T2DM among adherent and nonadherent patients, comparing these groups with the discontinuers and adjusting for age, sex, comorbidities, and concomitant medications.
The adjusted hazard ratio for developing T2DM was 0.61 among adherent patients and 0.78 among nonadherent patients.
“This population-based study highlighted that taking less than 90% of the prescribed antimalarials compromises their effect in preventing T2DM in SLE patients,” Dr. Salmasi and colleagues wrote. “Our findings should be used to emphasize the importance of medication adherence in not only treating SLE but also preventing its complications.”
The researchers reported having no conflicts of interest.
SOURCE: Salmasi S et al. Arthritis Care Res. 2020 Jan 21. doi: 10.1002/acr.24147.
Adhering to antimalarial treatment offers some protection to patients with systemic lupus erythematosus (SLE) from developing type 2 diabetes mellitus (T2DM), according to new research.
Patients who took at least 90% of their prescribed antimalarial doses were 39% less likely to develop T2DM than patients who discontinued antimalarial therapy. Patients who took less than 90% of their prescribed doses but didn’t discontinue treatment were 22% less likely to develop T2DM.
“[O]ur study provides further support for the importance of adherence to antimalarials in SLE by demonstrating protective impacts on T2DM,” Shahrzad Salmasi, PhD, of the University of British Columbia, Vancouver, and colleagues wrote in Arthritis Care & Research.
Dr. Salmasi and colleagues conducted this retrospective study using administrative health data on patients in British Columbia. The researchers analyzed 1,498 patients with SLE. Their mean age was about 44 years, and 91% were women.
The researchers used data on prescription dates and days’ supply to establish antimalarial drug courses and gaps in treatment. A new treatment course occurred when a 90-day gap was exceeded between refills. The researchers calculated the proportion of days covered (PDC) – the total number of days with antimalarials divided by the length of the course – and separated patients into three categories:
- Adherent to treatment – PDC of 0.90 or greater
- Nonadherent – PDC greater than 0 but less than 0.90
- Discontinuer – PDC of 0
The patients had a mean of about 23 antimalarial prescriptions and a mean of about two courses. The mean course duration was 554 days.
At a median follow-up of 4.6 years, there were 140 incident cases of T2DM. The researchers calculated the risk of T2DM among adherent and nonadherent patients, comparing these groups with the discontinuers and adjusting for age, sex, comorbidities, and concomitant medications.
The adjusted hazard ratio for developing T2DM was 0.61 among adherent patients and 0.78 among nonadherent patients.
“This population-based study highlighted that taking less than 90% of the prescribed antimalarials compromises their effect in preventing T2DM in SLE patients,” Dr. Salmasi and colleagues wrote. “Our findings should be used to emphasize the importance of medication adherence in not only treating SLE but also preventing its complications.”
The researchers reported having no conflicts of interest.
SOURCE: Salmasi S et al. Arthritis Care Res. 2020 Jan 21. doi: 10.1002/acr.24147.
FROM ARTHRITIS CARE & RESEARCH
New appointments at Cleveland Clinic, Fox Chase
Melissa Walt, PsyD, a clinical psychologist, recently joined the Taussig Cancer Institute at Cleveland Clinic. In this role, Dr. Walt focuses on the psychological treatment of cancer patients, helping them adjust to their diagnosis and navigate life with cancer.
Dr. Walt was previously a staff psychologist at the outpatient behavioral health/mental health clinic at the VA Southern Nevada Healthcare System in North Las Vegas. She obtained her doctorate degree from Adler University in Chicago, completed a residency at the VA Southern Nevada Healthcare System, and completed a fellowship at Memphis VA Medical Center.
Sheela Ahmed, MD, has joined Fox Chase Cancer Center in Philadelphia and works in the department of medicine as an associate clinical professor in the division of pulmonary, sleep, and critical care medicine. She is board certified in internal, pulmonary, critical care, and sleep medicine.
Dr. Ahmed was previously a pulmonary, critical care, and sleep physician at WellSpan Health in York, Pa. She received her medical degree from Patna (India) Medical College and Hospital. Dr. Ahmed completed a residency at the Postgraduate Institute of Medical Education and Research in Chandigarh, India, and a fellowship at St. Thomas Hospital in London and North Manchester (England) General Hospital. She also completed a residency and fellowship at Oregon Health and Science University in Portland.
Usman Ali, MD, has joined Fox Chase as an assistant professor in the division of hospital medicine within the department of medicine. Dr. Ali is board certified to practice medicine in Pennsylvania and New Jersey, and he is certified by the Educational Commission for Foreign Medical Graduates.
Dr. Ali previously worked as a contracted hospitalist at Fox Chase. He obtained his medical degree from Jawaharlal Nehru Medical College in Aligarh, India, and completed a residency at the Institute of Ophthalmology in Aligarh. Dr. Ali also completed a residency at Capital Health System in Pennington, N.J., and a fellowship at Mount Sinai Beth Israel Medical Center in New York City.
Dylan R. Sherry, MD, has joined the pain and palliative care program in the department of hematology/oncology at Fox Chase as an assistant professor on the academic clinician track. Dr. Sherry is board certified in internal medicine and board eligible in hospice and palliative medicine.
Dr. Sherry was previously a hospitalist at Abington Memorial Hospital, now Abington Hospital–Jefferson Health, in Abington Township, Pa. He received his medical degree from Case Western Reserve University in Cleveland. He completed his residency and fellowship at Beth Israel Deaconess Medical Center in Boston.
Melissa Walt, PsyD, a clinical psychologist, recently joined the Taussig Cancer Institute at Cleveland Clinic. In this role, Dr. Walt focuses on the psychological treatment of cancer patients, helping them adjust to their diagnosis and navigate life with cancer.
Dr. Walt was previously a staff psychologist at the outpatient behavioral health/mental health clinic at the VA Southern Nevada Healthcare System in North Las Vegas. She obtained her doctorate degree from Adler University in Chicago, completed a residency at the VA Southern Nevada Healthcare System, and completed a fellowship at Memphis VA Medical Center.
Sheela Ahmed, MD, has joined Fox Chase Cancer Center in Philadelphia and works in the department of medicine as an associate clinical professor in the division of pulmonary, sleep, and critical care medicine. She is board certified in internal, pulmonary, critical care, and sleep medicine.
Dr. Ahmed was previously a pulmonary, critical care, and sleep physician at WellSpan Health in York, Pa. She received her medical degree from Patna (India) Medical College and Hospital. Dr. Ahmed completed a residency at the Postgraduate Institute of Medical Education and Research in Chandigarh, India, and a fellowship at St. Thomas Hospital in London and North Manchester (England) General Hospital. She also completed a residency and fellowship at Oregon Health and Science University in Portland.
Usman Ali, MD, has joined Fox Chase as an assistant professor in the division of hospital medicine within the department of medicine. Dr. Ali is board certified to practice medicine in Pennsylvania and New Jersey, and he is certified by the Educational Commission for Foreign Medical Graduates.
Dr. Ali previously worked as a contracted hospitalist at Fox Chase. He obtained his medical degree from Jawaharlal Nehru Medical College in Aligarh, India, and completed a residency at the Institute of Ophthalmology in Aligarh. Dr. Ali also completed a residency at Capital Health System in Pennington, N.J., and a fellowship at Mount Sinai Beth Israel Medical Center in New York City.
Dylan R. Sherry, MD, has joined the pain and palliative care program in the department of hematology/oncology at Fox Chase as an assistant professor on the academic clinician track. Dr. Sherry is board certified in internal medicine and board eligible in hospice and palliative medicine.
Dr. Sherry was previously a hospitalist at Abington Memorial Hospital, now Abington Hospital–Jefferson Health, in Abington Township, Pa. He received his medical degree from Case Western Reserve University in Cleveland. He completed his residency and fellowship at Beth Israel Deaconess Medical Center in Boston.
Melissa Walt, PsyD, a clinical psychologist, recently joined the Taussig Cancer Institute at Cleveland Clinic. In this role, Dr. Walt focuses on the psychological treatment of cancer patients, helping them adjust to their diagnosis and navigate life with cancer.
Dr. Walt was previously a staff psychologist at the outpatient behavioral health/mental health clinic at the VA Southern Nevada Healthcare System in North Las Vegas. She obtained her doctorate degree from Adler University in Chicago, completed a residency at the VA Southern Nevada Healthcare System, and completed a fellowship at Memphis VA Medical Center.
Sheela Ahmed, MD, has joined Fox Chase Cancer Center in Philadelphia and works in the department of medicine as an associate clinical professor in the division of pulmonary, sleep, and critical care medicine. She is board certified in internal, pulmonary, critical care, and sleep medicine.
Dr. Ahmed was previously a pulmonary, critical care, and sleep physician at WellSpan Health in York, Pa. She received her medical degree from Patna (India) Medical College and Hospital. Dr. Ahmed completed a residency at the Postgraduate Institute of Medical Education and Research in Chandigarh, India, and a fellowship at St. Thomas Hospital in London and North Manchester (England) General Hospital. She also completed a residency and fellowship at Oregon Health and Science University in Portland.
Usman Ali, MD, has joined Fox Chase as an assistant professor in the division of hospital medicine within the department of medicine. Dr. Ali is board certified to practice medicine in Pennsylvania and New Jersey, and he is certified by the Educational Commission for Foreign Medical Graduates.
Dr. Ali previously worked as a contracted hospitalist at Fox Chase. He obtained his medical degree from Jawaharlal Nehru Medical College in Aligarh, India, and completed a residency at the Institute of Ophthalmology in Aligarh. Dr. Ali also completed a residency at Capital Health System in Pennington, N.J., and a fellowship at Mount Sinai Beth Israel Medical Center in New York City.
Dylan R. Sherry, MD, has joined the pain and palliative care program in the department of hematology/oncology at Fox Chase as an assistant professor on the academic clinician track. Dr. Sherry is board certified in internal medicine and board eligible in hospice and palliative medicine.
Dr. Sherry was previously a hospitalist at Abington Memorial Hospital, now Abington Hospital–Jefferson Health, in Abington Township, Pa. He received his medical degree from Case Western Reserve University in Cleveland. He completed his residency and fellowship at Beth Israel Deaconess Medical Center in Boston.
Adding lymphopenia component ‘improves’ FLIPI
Incorporating lymphopenia into the Follicular Lymphoma International Prognostic Index (FLIPI) can improve prognostication, according to researchers.
The team added lymphopenia as a point in a revised FLIPI scoring system, called FLIPI-L, and found the new system could better predict overall survival (OS), progression-free survival, and histologic transformation in patients with follicular lymphoma.
George Yang, MD, of Moffitt Cancer Center in Tampa, Fla., and his colleagues described results with the FLIPI-L in a letter published in Blood Cancer Journal.
“Prior studies have demonstrated that lymphopenia was associated with worsened OS in [follicular lymphoma],” Dr. Yang and his colleagues wrote. “Therefore, we hypothesized that lymphopenia may be integrated with existing FLIPI to better stratify long-term survival outcomes and predict for transformation.”
The researchers tested this theory in 736 follicular lymphoma patients who were followed for a median of 72 months (range, 2-211 months). The 5-year OS in this cohort was 81.3%, the 10-year OS was 67.3%, and 18% of patients experienced transformation to high-grade lymphoma.
The researchers defined absolute lymphopenia as less than 1.0 × 109 lymphocytes per liter. In multivariate analyses, lymphopenia was an independent predictor of OS (hazard ratio, 1.74; P less than .01) and transformation (odds ratio, 2.1; P less than .01).
To incorporate lymphopenia into the FLIPI, the researchers created a model in which 1 point was given for each of the standard FLIPI components (age, Ann Arbor stage, number of nodal areas, lactate dehydrogenase, and hemoglobin level), and one point was given for the presence of lymphopenia. Patients in the low-risk FLIPI-L category had 0-1 points, those in the intermediate-risk category had 2-3 points, and patients in the high-risk FLIPI-L category had 4-6 points.
Using the original FLIPI, the 5-year OS was 91% in the low-risk group (0-1), 82.7% in the intermediate-risk group (2), and 66% in the high-risk group (3-5). The 10-year OS was 80.4%, 66%, and 45.8%, respectively.
Using the FLIPI-L, the 5-year OS was 94.5% in the low-risk group (0-1), 89% in the intermediate-risk group (2-3), and 61% in the high-risk group (4-6). The 10-year OS was 83.9%, 68.5%, and 34.5%, respectively.
In a univariate Cox regression analysis of OS, each point increase in FLIPI-L score was associated with a significant increase in hazard ratio. For example, the hazard ratio was 3.4 for patients with a FLIPI-L score of 1 and 30.9 for those with a FLIPI-L score of 6 (P less than .02 for all FLIPI-L scores). Conversely, increases in hazard ratio were not significant with the original FLIPI (P greater than .05 for all FLIPI scores).
The FLIPI-L was prognostic for OS in different treatment groups. In patients who received rituximab alone, radiation alone, or rituximab plus chemotherapy, the scoring system differentiated low-, intermediate-, and high-risk groups (P less than .04). In patients under observation, the FLIPI-L distinguished low/intermediate-risk and high-risk groups (P less than .01).
For patients who progressed within 24 months, the FLIPI-L was more predictive of progression-free survival (P = .05) than was the original FLIPI (P = .11).
Increasing FLIPI-L was an independent predictor of transformation, both when assessed as a continuous variable (P less than .01) and stepwise for FLIPI-L 3-5 (P = .004-.01). The original FLIPI, on the other hand, was not an independent predictor of transformation.
“Our analysis of a lymphopenia cutoff as an addition to the original FLIPI is simple yet improves risk stratification to differentiate between prognostic groups and, importantly, to predict transformation,” Dr. Yang and his colleagues wrote.
The authors reported having no conflicts of interest.
SOURCE: Yang G et al. Blood Cancer J. 2020 Jan 2;9(12):104. doi: 10.1038/s41408-019-0269-6.
Incorporating lymphopenia into the Follicular Lymphoma International Prognostic Index (FLIPI) can improve prognostication, according to researchers.
The team added lymphopenia as a point in a revised FLIPI scoring system, called FLIPI-L, and found the new system could better predict overall survival (OS), progression-free survival, and histologic transformation in patients with follicular lymphoma.
George Yang, MD, of Moffitt Cancer Center in Tampa, Fla., and his colleagues described results with the FLIPI-L in a letter published in Blood Cancer Journal.
“Prior studies have demonstrated that lymphopenia was associated with worsened OS in [follicular lymphoma],” Dr. Yang and his colleagues wrote. “Therefore, we hypothesized that lymphopenia may be integrated with existing FLIPI to better stratify long-term survival outcomes and predict for transformation.”
The researchers tested this theory in 736 follicular lymphoma patients who were followed for a median of 72 months (range, 2-211 months). The 5-year OS in this cohort was 81.3%, the 10-year OS was 67.3%, and 18% of patients experienced transformation to high-grade lymphoma.
The researchers defined absolute lymphopenia as less than 1.0 × 109 lymphocytes per liter. In multivariate analyses, lymphopenia was an independent predictor of OS (hazard ratio, 1.74; P less than .01) and transformation (odds ratio, 2.1; P less than .01).
To incorporate lymphopenia into the FLIPI, the researchers created a model in which 1 point was given for each of the standard FLIPI components (age, Ann Arbor stage, number of nodal areas, lactate dehydrogenase, and hemoglobin level), and one point was given for the presence of lymphopenia. Patients in the low-risk FLIPI-L category had 0-1 points, those in the intermediate-risk category had 2-3 points, and patients in the high-risk FLIPI-L category had 4-6 points.
Using the original FLIPI, the 5-year OS was 91% in the low-risk group (0-1), 82.7% in the intermediate-risk group (2), and 66% in the high-risk group (3-5). The 10-year OS was 80.4%, 66%, and 45.8%, respectively.
Using the FLIPI-L, the 5-year OS was 94.5% in the low-risk group (0-1), 89% in the intermediate-risk group (2-3), and 61% in the high-risk group (4-6). The 10-year OS was 83.9%, 68.5%, and 34.5%, respectively.
In a univariate Cox regression analysis of OS, each point increase in FLIPI-L score was associated with a significant increase in hazard ratio. For example, the hazard ratio was 3.4 for patients with a FLIPI-L score of 1 and 30.9 for those with a FLIPI-L score of 6 (P less than .02 for all FLIPI-L scores). Conversely, increases in hazard ratio were not significant with the original FLIPI (P greater than .05 for all FLIPI scores).
The FLIPI-L was prognostic for OS in different treatment groups. In patients who received rituximab alone, radiation alone, or rituximab plus chemotherapy, the scoring system differentiated low-, intermediate-, and high-risk groups (P less than .04). In patients under observation, the FLIPI-L distinguished low/intermediate-risk and high-risk groups (P less than .01).
For patients who progressed within 24 months, the FLIPI-L was more predictive of progression-free survival (P = .05) than was the original FLIPI (P = .11).
Increasing FLIPI-L was an independent predictor of transformation, both when assessed as a continuous variable (P less than .01) and stepwise for FLIPI-L 3-5 (P = .004-.01). The original FLIPI, on the other hand, was not an independent predictor of transformation.
“Our analysis of a lymphopenia cutoff as an addition to the original FLIPI is simple yet improves risk stratification to differentiate between prognostic groups and, importantly, to predict transformation,” Dr. Yang and his colleagues wrote.
The authors reported having no conflicts of interest.
SOURCE: Yang G et al. Blood Cancer J. 2020 Jan 2;9(12):104. doi: 10.1038/s41408-019-0269-6.
Incorporating lymphopenia into the Follicular Lymphoma International Prognostic Index (FLIPI) can improve prognostication, according to researchers.
The team added lymphopenia as a point in a revised FLIPI scoring system, called FLIPI-L, and found the new system could better predict overall survival (OS), progression-free survival, and histologic transformation in patients with follicular lymphoma.
George Yang, MD, of Moffitt Cancer Center in Tampa, Fla., and his colleagues described results with the FLIPI-L in a letter published in Blood Cancer Journal.
“Prior studies have demonstrated that lymphopenia was associated with worsened OS in [follicular lymphoma],” Dr. Yang and his colleagues wrote. “Therefore, we hypothesized that lymphopenia may be integrated with existing FLIPI to better stratify long-term survival outcomes and predict for transformation.”
The researchers tested this theory in 736 follicular lymphoma patients who were followed for a median of 72 months (range, 2-211 months). The 5-year OS in this cohort was 81.3%, the 10-year OS was 67.3%, and 18% of patients experienced transformation to high-grade lymphoma.
The researchers defined absolute lymphopenia as less than 1.0 × 109 lymphocytes per liter. In multivariate analyses, lymphopenia was an independent predictor of OS (hazard ratio, 1.74; P less than .01) and transformation (odds ratio, 2.1; P less than .01).
To incorporate lymphopenia into the FLIPI, the researchers created a model in which 1 point was given for each of the standard FLIPI components (age, Ann Arbor stage, number of nodal areas, lactate dehydrogenase, and hemoglobin level), and one point was given for the presence of lymphopenia. Patients in the low-risk FLIPI-L category had 0-1 points, those in the intermediate-risk category had 2-3 points, and patients in the high-risk FLIPI-L category had 4-6 points.
Using the original FLIPI, the 5-year OS was 91% in the low-risk group (0-1), 82.7% in the intermediate-risk group (2), and 66% in the high-risk group (3-5). The 10-year OS was 80.4%, 66%, and 45.8%, respectively.
Using the FLIPI-L, the 5-year OS was 94.5% in the low-risk group (0-1), 89% in the intermediate-risk group (2-3), and 61% in the high-risk group (4-6). The 10-year OS was 83.9%, 68.5%, and 34.5%, respectively.
In a univariate Cox regression analysis of OS, each point increase in FLIPI-L score was associated with a significant increase in hazard ratio. For example, the hazard ratio was 3.4 for patients with a FLIPI-L score of 1 and 30.9 for those with a FLIPI-L score of 6 (P less than .02 for all FLIPI-L scores). Conversely, increases in hazard ratio were not significant with the original FLIPI (P greater than .05 for all FLIPI scores).
The FLIPI-L was prognostic for OS in different treatment groups. In patients who received rituximab alone, radiation alone, or rituximab plus chemotherapy, the scoring system differentiated low-, intermediate-, and high-risk groups (P less than .04). In patients under observation, the FLIPI-L distinguished low/intermediate-risk and high-risk groups (P less than .01).
For patients who progressed within 24 months, the FLIPI-L was more predictive of progression-free survival (P = .05) than was the original FLIPI (P = .11).
Increasing FLIPI-L was an independent predictor of transformation, both when assessed as a continuous variable (P less than .01) and stepwise for FLIPI-L 3-5 (P = .004-.01). The original FLIPI, on the other hand, was not an independent predictor of transformation.
“Our analysis of a lymphopenia cutoff as an addition to the original FLIPI is simple yet improves risk stratification to differentiate between prognostic groups and, importantly, to predict transformation,” Dr. Yang and his colleagues wrote.
The authors reported having no conflicts of interest.
SOURCE: Yang G et al. Blood Cancer J. 2020 Jan 2;9(12):104. doi: 10.1038/s41408-019-0269-6.
FROM BLOOD CANCER JOURNAL