User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Keeping the Differential at Hand
ANSWER
The correct answer is granuloma annulare (GA; choice “a”).
DISCUSSION
One of the most difficult concepts to grasp in dermatologic diagnosis is that almost all lesions and conditions—even the most common—have a broad range of morphologic presentations. These will often differ from the textbook photos. In this digital age, a simple Internet search will provide many results showing a diverse morphologic spectrum for many diseases, conditions, and lesions.
GA is a good example of how the presentation can vary. It has raised rolled margins and delled (gently concave) centers. The brownish red color is typical, but this patient showed a deeper red than most cases of GA. Occasionally, the red color is even deeper, with large patches of darkened skin and no palpable component.
Unfortunately, the misdiagnosis of fungal infection in this patient is typical. But dermatophytosis (otherwise known as “ringworm”)—the most common fungal skin infection—involves the epidermis. This means the patient would have scaly skin, probably with a well-defined margin—factors missing in this case. In addition, this patient reported no contact with sources of infection: animals, children, or immunosuppressive agents.
Ultimately, the most basic information that this patient's past providers neglected was a differential diagnosis. By establishing a differential, there was a clear decision to biopsy, which not only revealed the correct diagnosis but effectively ruled out the other options.
In defense of the patient’s other providers, I must admit that as a young primary care provider, I made this same mistake for exactly the same reasons.
TREATMENT
Most cases of GA are mild and self-limited, requiring no treatment. This is fortunate because no effective treatment exists. Topical steroids and cryotherapy will lighten the lesion, but GA nearly always resolves on its own.
ANSWER
The correct answer is granuloma annulare (GA; choice “a”).
DISCUSSION
One of the most difficult concepts to grasp in dermatologic diagnosis is that almost all lesions and conditions—even the most common—have a broad range of morphologic presentations. These will often differ from the textbook photos. In this digital age, a simple Internet search will provide many results showing a diverse morphologic spectrum for many diseases, conditions, and lesions.
GA is a good example of how the presentation can vary. It has raised rolled margins and delled (gently concave) centers. The brownish red color is typical, but this patient showed a deeper red than most cases of GA. Occasionally, the red color is even deeper, with large patches of darkened skin and no palpable component.
Unfortunately, the misdiagnosis of fungal infection in this patient is typical. But dermatophytosis (otherwise known as “ringworm”)—the most common fungal skin infection—involves the epidermis. This means the patient would have scaly skin, probably with a well-defined margin—factors missing in this case. In addition, this patient reported no contact with sources of infection: animals, children, or immunosuppressive agents.
Ultimately, the most basic information that this patient's past providers neglected was a differential diagnosis. By establishing a differential, there was a clear decision to biopsy, which not only revealed the correct diagnosis but effectively ruled out the other options.
In defense of the patient’s other providers, I must admit that as a young primary care provider, I made this same mistake for exactly the same reasons.
TREATMENT
Most cases of GA are mild and self-limited, requiring no treatment. This is fortunate because no effective treatment exists. Topical steroids and cryotherapy will lighten the lesion, but GA nearly always resolves on its own.
ANSWER
The correct answer is granuloma annulare (GA; choice “a”).
DISCUSSION
One of the most difficult concepts to grasp in dermatologic diagnosis is that almost all lesions and conditions—even the most common—have a broad range of morphologic presentations. These will often differ from the textbook photos. In this digital age, a simple Internet search will provide many results showing a diverse morphologic spectrum for many diseases, conditions, and lesions.
GA is a good example of how the presentation can vary. It has raised rolled margins and delled (gently concave) centers. The brownish red color is typical, but this patient showed a deeper red than most cases of GA. Occasionally, the red color is even deeper, with large patches of darkened skin and no palpable component.
Unfortunately, the misdiagnosis of fungal infection in this patient is typical. But dermatophytosis (otherwise known as “ringworm”)—the most common fungal skin infection—involves the epidermis. This means the patient would have scaly skin, probably with a well-defined margin—factors missing in this case. In addition, this patient reported no contact with sources of infection: animals, children, or immunosuppressive agents.
Ultimately, the most basic information that this patient's past providers neglected was a differential diagnosis. By establishing a differential, there was a clear decision to biopsy, which not only revealed the correct diagnosis but effectively ruled out the other options.
In defense of the patient’s other providers, I must admit that as a young primary care provider, I made this same mistake for exactly the same reasons.
TREATMENT
Most cases of GA are mild and self-limited, requiring no treatment. This is fortunate because no effective treatment exists. Topical steroids and cryotherapy will lighten the lesion, but GA nearly always resolves on its own.
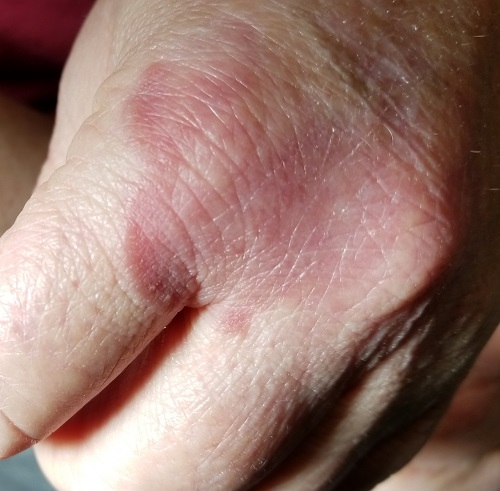
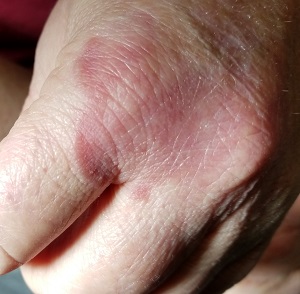
Several months ago, an asymptomatic rash slowly manifested on a 60-year-old woman’s hand. The rash—diagnosed previously as a fungal infection—continues to grow despite application of multiple antifungal creams, including tolnaftate, clotrimazole, and terbinafine. In addition, she was treated with a 1-month course of oral terbinafine 250 mg/d. Unfortunately, no treatment has provided her relief.
The patient is in otherwise good health. She denies any injury to the area. She also reports no exposure to children or animals.
The rash is a reddish brown, annular patch of skin that covers most of the dorsum of her left hand. The borders are slightly raised and thickened. It is nontender and readily blanchable. It is intradermal, with no surface disturbance such as scaling.
Punch biopsy shows palisaded granulomatous features, with no epidermal changes. Stains for fungi and bacteria fail to demonstrate any organisms.
Studies gauge toll of pausing fertility treatment during pandemic
More than 60% of patients at a center for reproductive medicine in Utah who had fertility treatments canceled because of the COVID-19 pandemic opted to resume treatment once the suspension was lifted about 7 weeks later.
At another fertility center in New York, a survey found that 96% of respondents who had a cycle canceled because of the pandemic found it upsetting, and 22% found it extremely upsetting, with extremely upsetting defined as equivalent to the loss of a child.
The indefinite time frame for resuming treatment when the New York survey was conducted may have been a major source of distress for patients, one of the researchers said at the American Society for Reproductive Medicine’s 2020 annual meeting, held virtually this year.
“They don’t know when they might have that chance again,” said Jenna M. Turocy, MD, of Columbia University Fertility Center, New York.
COVID-19 guidelines published by ASRM on March 17 recommended the suspension of new treatment cycles, including ovulation induction, intrauterine inseminations, and in vitro fertilization (IVF).
An ASRM COVID-19 task force has since supported “the measured resumption of fertility care following the easing of restrictions,” said Paul C. Lin, MD, president of the Society for Assisted Reproductive Technology and a member of the task force.
“Over the past several months, significant knowledge has been gained regarding the COVID-19 virus and its impact on patients and the medical system,” he said in a news release about the two studies that assessed the pandemic’s effects.
Certain precautions remain. “It has become clear that we will need to be practicing COVID-19 protocols at least until an effective and safe vaccine or broadly effective treatment becomes widely available,” Dr. Lin said.
Desire to proceed during a pandemic
The center continued to offer IVF cycles for oncofertility patients on an urgent basis.
In early May, patients whose cycles had been suspended had the option to receive treatment.
“Upon reopening, every patient received standardized counseling from their primary IVF physician,” Lauren Verrilli, MD, a reproductive endocrinology and infertility fellow at the University of Utah, Salt Lake City, said at the virtual meeting.
Doctors explained that much remained unknown about COVID-19 in pregnancy, and that it was unclear whether the clinic would need to shut down again. In addition, patients had to undergo COVID-19 testing.
To identify factors associated with proceeding with treatment after the suspension, the researchers compared patients who resumed treatment with patients who did not.
Their analysis included 278 patients who had planned an IVF cycle or frozen embryo transfer (FET) prior to the shutdown. The researchers examined factors such as age, parity, anti-Müllerian hormone, antral follicle count, history of prior IVF cycles or FET, number of frozen blastocysts, gamete source, and use of a gestational carrier.
In all, 62% of patients opted to receive treatment once restrictions were lifted, including 69 of the 133 (52%) patients with planned fresh cycles and 104 of the 145 (72%) patients with planned FET cycles.
Among those with planned fresh cycles, those who opted to resume treatment tended to be older than those who did not resume treatment, with a median age of 37 years versus 35 years, but the difference was not statistically significant.
Among patients with planned FET cycles, those who did not resume treatment were more likely to have a gestational carrier, compared with those who resumed treatment (7% vs. 1%). In some cases, gestational carriers lived in another state and the pandemic complicated travel arrangements, which contributed to delays, Dr. Verrilli said.
The analysis did not include information about income or socioeconomic status, which may play a role in patients’ decisions, Dr. Verrilli said.
Emotional impact of indefinite delay
Fertility treatment is often time sensitive, particularly for patients with advanced reproductive age or diminished ovarian reserve, and indefinite postponement of fertility treatment potentially could lead some patients to lose the ability to conceive with their own gametes, Dr. Turocy said.
In early April, Dr. Turocy and colleagues surveyed patients at their academic fertility center in New York City to assess patients’ reactions to the ASRM recommendations. They decided to conduct the study after they realized that treatment cancellations were having a significant emotional effect.
Investigators emailed an 18-item survey to more than 3,000 patients.
In all, 518 patients completed the survey, a response rate of 17%. Patients had an average age of 37 years (range, 23-52 years), and 92% were female. About 24% had children, and 66% had received at least one fertility treatment.
Half had a cycle canceled because of the COVID-19 pandemic, including timed intercourse cycles (5%), intrauterine insemination cycles (23%), IVF cycles with a planned fresh embryo transfer (10%), IVF with all frozen embryos (27%), egg freeze cycles (3%), and FET cycles (30%).
In response to survey questions about whether they agreed with ASRM recommendations, “the reactions were mixed,” Dr. Turocy said.
About 36% of patients agreed all fertility cycles should be canceled, 22% were unsure, and 43% disagreed with the recommendation. Patients who had a cycle canceled were slightly more likely to agree with the cancellations (40% agreed) than those who did not have a cycle canceled (30% agreed), Dr. Turocy said.
Most respondents would have preferred an option to start a treatment cycle in consultation with their doctor. Half “would have chosen to start a new cycle during the height of the pandemic in New York City,” Dr. Turocy said.
Patient opinions may vary by region and depend on the severity of COVID-19 outbreaks there, and they also might change over time, Dr. Turocy suggested. In addition, the opinions and characteristics of patients who responded to the anonymous survey may differ from those of patients who did not respond.
Dr. Verrilli, Dr. Turocy, and Dr. Lin had no relevant financial disclosures.
More than 60% of patients at a center for reproductive medicine in Utah who had fertility treatments canceled because of the COVID-19 pandemic opted to resume treatment once the suspension was lifted about 7 weeks later.
At another fertility center in New York, a survey found that 96% of respondents who had a cycle canceled because of the pandemic found it upsetting, and 22% found it extremely upsetting, with extremely upsetting defined as equivalent to the loss of a child.
The indefinite time frame for resuming treatment when the New York survey was conducted may have been a major source of distress for patients, one of the researchers said at the American Society for Reproductive Medicine’s 2020 annual meeting, held virtually this year.
“They don’t know when they might have that chance again,” said Jenna M. Turocy, MD, of Columbia University Fertility Center, New York.
COVID-19 guidelines published by ASRM on March 17 recommended the suspension of new treatment cycles, including ovulation induction, intrauterine inseminations, and in vitro fertilization (IVF).
An ASRM COVID-19 task force has since supported “the measured resumption of fertility care following the easing of restrictions,” said Paul C. Lin, MD, president of the Society for Assisted Reproductive Technology and a member of the task force.
“Over the past several months, significant knowledge has been gained regarding the COVID-19 virus and its impact on patients and the medical system,” he said in a news release about the two studies that assessed the pandemic’s effects.
Certain precautions remain. “It has become clear that we will need to be practicing COVID-19 protocols at least until an effective and safe vaccine or broadly effective treatment becomes widely available,” Dr. Lin said.
Desire to proceed during a pandemic
The center continued to offer IVF cycles for oncofertility patients on an urgent basis.
In early May, patients whose cycles had been suspended had the option to receive treatment.
“Upon reopening, every patient received standardized counseling from their primary IVF physician,” Lauren Verrilli, MD, a reproductive endocrinology and infertility fellow at the University of Utah, Salt Lake City, said at the virtual meeting.
Doctors explained that much remained unknown about COVID-19 in pregnancy, and that it was unclear whether the clinic would need to shut down again. In addition, patients had to undergo COVID-19 testing.
To identify factors associated with proceeding with treatment after the suspension, the researchers compared patients who resumed treatment with patients who did not.
Their analysis included 278 patients who had planned an IVF cycle or frozen embryo transfer (FET) prior to the shutdown. The researchers examined factors such as age, parity, anti-Müllerian hormone, antral follicle count, history of prior IVF cycles or FET, number of frozen blastocysts, gamete source, and use of a gestational carrier.
In all, 62% of patients opted to receive treatment once restrictions were lifted, including 69 of the 133 (52%) patients with planned fresh cycles and 104 of the 145 (72%) patients with planned FET cycles.
Among those with planned fresh cycles, those who opted to resume treatment tended to be older than those who did not resume treatment, with a median age of 37 years versus 35 years, but the difference was not statistically significant.
Among patients with planned FET cycles, those who did not resume treatment were more likely to have a gestational carrier, compared with those who resumed treatment (7% vs. 1%). In some cases, gestational carriers lived in another state and the pandemic complicated travel arrangements, which contributed to delays, Dr. Verrilli said.
The analysis did not include information about income or socioeconomic status, which may play a role in patients’ decisions, Dr. Verrilli said.
Emotional impact of indefinite delay
Fertility treatment is often time sensitive, particularly for patients with advanced reproductive age or diminished ovarian reserve, and indefinite postponement of fertility treatment potentially could lead some patients to lose the ability to conceive with their own gametes, Dr. Turocy said.
In early April, Dr. Turocy and colleagues surveyed patients at their academic fertility center in New York City to assess patients’ reactions to the ASRM recommendations. They decided to conduct the study after they realized that treatment cancellations were having a significant emotional effect.
Investigators emailed an 18-item survey to more than 3,000 patients.
In all, 518 patients completed the survey, a response rate of 17%. Patients had an average age of 37 years (range, 23-52 years), and 92% were female. About 24% had children, and 66% had received at least one fertility treatment.
Half had a cycle canceled because of the COVID-19 pandemic, including timed intercourse cycles (5%), intrauterine insemination cycles (23%), IVF cycles with a planned fresh embryo transfer (10%), IVF with all frozen embryos (27%), egg freeze cycles (3%), and FET cycles (30%).
In response to survey questions about whether they agreed with ASRM recommendations, “the reactions were mixed,” Dr. Turocy said.
About 36% of patients agreed all fertility cycles should be canceled, 22% were unsure, and 43% disagreed with the recommendation. Patients who had a cycle canceled were slightly more likely to agree with the cancellations (40% agreed) than those who did not have a cycle canceled (30% agreed), Dr. Turocy said.
Most respondents would have preferred an option to start a treatment cycle in consultation with their doctor. Half “would have chosen to start a new cycle during the height of the pandemic in New York City,” Dr. Turocy said.
Patient opinions may vary by region and depend on the severity of COVID-19 outbreaks there, and they also might change over time, Dr. Turocy suggested. In addition, the opinions and characteristics of patients who responded to the anonymous survey may differ from those of patients who did not respond.
Dr. Verrilli, Dr. Turocy, and Dr. Lin had no relevant financial disclosures.
More than 60% of patients at a center for reproductive medicine in Utah who had fertility treatments canceled because of the COVID-19 pandemic opted to resume treatment once the suspension was lifted about 7 weeks later.
At another fertility center in New York, a survey found that 96% of respondents who had a cycle canceled because of the pandemic found it upsetting, and 22% found it extremely upsetting, with extremely upsetting defined as equivalent to the loss of a child.
The indefinite time frame for resuming treatment when the New York survey was conducted may have been a major source of distress for patients, one of the researchers said at the American Society for Reproductive Medicine’s 2020 annual meeting, held virtually this year.
“They don’t know when they might have that chance again,” said Jenna M. Turocy, MD, of Columbia University Fertility Center, New York.
COVID-19 guidelines published by ASRM on March 17 recommended the suspension of new treatment cycles, including ovulation induction, intrauterine inseminations, and in vitro fertilization (IVF).
An ASRM COVID-19 task force has since supported “the measured resumption of fertility care following the easing of restrictions,” said Paul C. Lin, MD, president of the Society for Assisted Reproductive Technology and a member of the task force.
“Over the past several months, significant knowledge has been gained regarding the COVID-19 virus and its impact on patients and the medical system,” he said in a news release about the two studies that assessed the pandemic’s effects.
Certain precautions remain. “It has become clear that we will need to be practicing COVID-19 protocols at least until an effective and safe vaccine or broadly effective treatment becomes widely available,” Dr. Lin said.
Desire to proceed during a pandemic
The center continued to offer IVF cycles for oncofertility patients on an urgent basis.
In early May, patients whose cycles had been suspended had the option to receive treatment.
“Upon reopening, every patient received standardized counseling from their primary IVF physician,” Lauren Verrilli, MD, a reproductive endocrinology and infertility fellow at the University of Utah, Salt Lake City, said at the virtual meeting.
Doctors explained that much remained unknown about COVID-19 in pregnancy, and that it was unclear whether the clinic would need to shut down again. In addition, patients had to undergo COVID-19 testing.
To identify factors associated with proceeding with treatment after the suspension, the researchers compared patients who resumed treatment with patients who did not.
Their analysis included 278 patients who had planned an IVF cycle or frozen embryo transfer (FET) prior to the shutdown. The researchers examined factors such as age, parity, anti-Müllerian hormone, antral follicle count, history of prior IVF cycles or FET, number of frozen blastocysts, gamete source, and use of a gestational carrier.
In all, 62% of patients opted to receive treatment once restrictions were lifted, including 69 of the 133 (52%) patients with planned fresh cycles and 104 of the 145 (72%) patients with planned FET cycles.
Among those with planned fresh cycles, those who opted to resume treatment tended to be older than those who did not resume treatment, with a median age of 37 years versus 35 years, but the difference was not statistically significant.
Among patients with planned FET cycles, those who did not resume treatment were more likely to have a gestational carrier, compared with those who resumed treatment (7% vs. 1%). In some cases, gestational carriers lived in another state and the pandemic complicated travel arrangements, which contributed to delays, Dr. Verrilli said.
The analysis did not include information about income or socioeconomic status, which may play a role in patients’ decisions, Dr. Verrilli said.
Emotional impact of indefinite delay
Fertility treatment is often time sensitive, particularly for patients with advanced reproductive age or diminished ovarian reserve, and indefinite postponement of fertility treatment potentially could lead some patients to lose the ability to conceive with their own gametes, Dr. Turocy said.
In early April, Dr. Turocy and colleagues surveyed patients at their academic fertility center in New York City to assess patients’ reactions to the ASRM recommendations. They decided to conduct the study after they realized that treatment cancellations were having a significant emotional effect.
Investigators emailed an 18-item survey to more than 3,000 patients.
In all, 518 patients completed the survey, a response rate of 17%. Patients had an average age of 37 years (range, 23-52 years), and 92% were female. About 24% had children, and 66% had received at least one fertility treatment.
Half had a cycle canceled because of the COVID-19 pandemic, including timed intercourse cycles (5%), intrauterine insemination cycles (23%), IVF cycles with a planned fresh embryo transfer (10%), IVF with all frozen embryos (27%), egg freeze cycles (3%), and FET cycles (30%).
In response to survey questions about whether they agreed with ASRM recommendations, “the reactions were mixed,” Dr. Turocy said.
About 36% of patients agreed all fertility cycles should be canceled, 22% were unsure, and 43% disagreed with the recommendation. Patients who had a cycle canceled were slightly more likely to agree with the cancellations (40% agreed) than those who did not have a cycle canceled (30% agreed), Dr. Turocy said.
Most respondents would have preferred an option to start a treatment cycle in consultation with their doctor. Half “would have chosen to start a new cycle during the height of the pandemic in New York City,” Dr. Turocy said.
Patient opinions may vary by region and depend on the severity of COVID-19 outbreaks there, and they also might change over time, Dr. Turocy suggested. In addition, the opinions and characteristics of patients who responded to the anonymous survey may differ from those of patients who did not respond.
Dr. Verrilli, Dr. Turocy, and Dr. Lin had no relevant financial disclosures.
FROM ASRM 2020
Painful pustules on hands
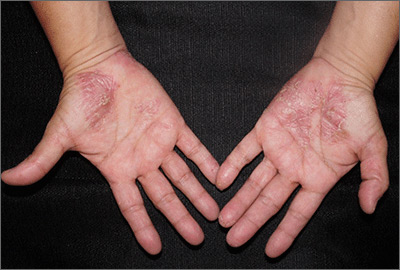
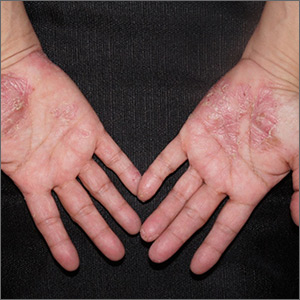
Close inspection of the plaques revealed multiple small pustules and mahogany colored macules on a broad, well-demarcated scaly red plaque. This pattern was consistent with palmoplantar pustular psoriasis.
Palmoplantar psoriasis is a chronic disease that may manifest at any age and in both sexes. The diagnosis may be clinical, as it was in this case. Bacterial culture of a pustule may demonstrate staphylococcus superinfection—a problem more likely to occur in smokers. More widespread disease, spiking fevers, or tachycardia could indicate a need for hospitalization for further work-up and management.
With disease localized to this patient’s hands and feet, the physician considered dyshidrotic eczema as part of the differential diagnosis. However dyshidrotic eczema, which presents with clear tapioca-like vesicles, is profoundly itchy; pustular psoriasis is often painful. Other differences: The vesicles in dyshidrotic eczema do not uniformly form pustules nor do they form brown macules upon healing, as occurs in palmoplantar pustular psoriasis.
Treatment for very limited disease includes topical ultrapotent steroids, topical retinoids, or phototherapy—either alone or in combination. However, more extensive disease, even if limited to hands and feet, merits systemic therapy with acitretin, methotrexate, or biologic agents. (Acitretin and methotrexate are reliable teratogens and should not be used in women who are, or may become, pregnant.) Individuals with known superinfection often benefit from antibiotic therapy, as well.
In this case, the patient had already undergone a tubal ligation for contraception. Therefore, she was started on acitretin 25 mg PO daily. Patients on systemic retinoids undergo laboratory surveillance for transaminitis and hypertriglyceridemia regularly. Adverse effects are generally well tolerated, but include photosensitivity and dry skin, lips, and eyes. The patient improved significantly on 25 mg/d and the dosage was reduced to 10 mg/d after 3 months. She currently remains clear on this regimen.
Text and photos courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. (Photo copyright retained.)
Misiak-Galazka M, Zozula J, Rudnicka L. Palmoplantar pustulosis: recent advances in etiopathogenesis and emerging treatments. Am J Clin Dermatol. 2020;21:355-370.

Close inspection of the plaques revealed multiple small pustules and mahogany colored macules on a broad, well-demarcated scaly red plaque. This pattern was consistent with palmoplantar pustular psoriasis.
Palmoplantar psoriasis is a chronic disease that may manifest at any age and in both sexes. The diagnosis may be clinical, as it was in this case. Bacterial culture of a pustule may demonstrate staphylococcus superinfection—a problem more likely to occur in smokers. More widespread disease, spiking fevers, or tachycardia could indicate a need for hospitalization for further work-up and management.
With disease localized to this patient’s hands and feet, the physician considered dyshidrotic eczema as part of the differential diagnosis. However dyshidrotic eczema, which presents with clear tapioca-like vesicles, is profoundly itchy; pustular psoriasis is often painful. Other differences: The vesicles in dyshidrotic eczema do not uniformly form pustules nor do they form brown macules upon healing, as occurs in palmoplantar pustular psoriasis.
Treatment for very limited disease includes topical ultrapotent steroids, topical retinoids, or phototherapy—either alone or in combination. However, more extensive disease, even if limited to hands and feet, merits systemic therapy with acitretin, methotrexate, or biologic agents. (Acitretin and methotrexate are reliable teratogens and should not be used in women who are, or may become, pregnant.) Individuals with known superinfection often benefit from antibiotic therapy, as well.
In this case, the patient had already undergone a tubal ligation for contraception. Therefore, she was started on acitretin 25 mg PO daily. Patients on systemic retinoids undergo laboratory surveillance for transaminitis and hypertriglyceridemia regularly. Adverse effects are generally well tolerated, but include photosensitivity and dry skin, lips, and eyes. The patient improved significantly on 25 mg/d and the dosage was reduced to 10 mg/d after 3 months. She currently remains clear on this regimen.
Text and photos courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. (Photo copyright retained.)

Close inspection of the plaques revealed multiple small pustules and mahogany colored macules on a broad, well-demarcated scaly red plaque. This pattern was consistent with palmoplantar pustular psoriasis.
Palmoplantar psoriasis is a chronic disease that may manifest at any age and in both sexes. The diagnosis may be clinical, as it was in this case. Bacterial culture of a pustule may demonstrate staphylococcus superinfection—a problem more likely to occur in smokers. More widespread disease, spiking fevers, or tachycardia could indicate a need for hospitalization for further work-up and management.
With disease localized to this patient’s hands and feet, the physician considered dyshidrotic eczema as part of the differential diagnosis. However dyshidrotic eczema, which presents with clear tapioca-like vesicles, is profoundly itchy; pustular psoriasis is often painful. Other differences: The vesicles in dyshidrotic eczema do not uniformly form pustules nor do they form brown macules upon healing, as occurs in palmoplantar pustular psoriasis.
Treatment for very limited disease includes topical ultrapotent steroids, topical retinoids, or phototherapy—either alone or in combination. However, more extensive disease, even if limited to hands and feet, merits systemic therapy with acitretin, methotrexate, or biologic agents. (Acitretin and methotrexate are reliable teratogens and should not be used in women who are, or may become, pregnant.) Individuals with known superinfection often benefit from antibiotic therapy, as well.
In this case, the patient had already undergone a tubal ligation for contraception. Therefore, she was started on acitretin 25 mg PO daily. Patients on systemic retinoids undergo laboratory surveillance for transaminitis and hypertriglyceridemia regularly. Adverse effects are generally well tolerated, but include photosensitivity and dry skin, lips, and eyes. The patient improved significantly on 25 mg/d and the dosage was reduced to 10 mg/d after 3 months. She currently remains clear on this regimen.
Text and photos courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. (Photo copyright retained.)
Misiak-Galazka M, Zozula J, Rudnicka L. Palmoplantar pustulosis: recent advances in etiopathogenesis and emerging treatments. Am J Clin Dermatol. 2020;21:355-370.
Misiak-Galazka M, Zozula J, Rudnicka L. Palmoplantar pustulosis: recent advances in etiopathogenesis and emerging treatments. Am J Clin Dermatol. 2020;21:355-370.
Experts disagree with USPSTF’s take on pediatric blood pressure screening
Current evidence is insufficient to assess the balance of benefits and harms of screening for high blood pressure in children and adolescents, the U.S. Preventive Services Task Force reported in JAMA.
However, two experts in this area suggested there is evidence if you know where to look, and pediatric BP testing is crucial now.
In this update to the 2013 statement, the USPSTF’s systematic review focused on evidence surrounding the benefits of screening, test accuracy, treatment effectiveness and harms, and links between hypertension and cardiovascular disease (CVD) markers in childhood and adulthood.
Limited information was available on the accuracy of screening tests. No studies were found that directly evaluated screening for pediatric high BP or reported effectiveness in delayed onset or risk reduction for cardiovascular outcomes related to hypertension. Additionally, no studies were found that addressed screening for secondary hypertension in asymptomatic pediatric patients. No studies were found that evaluated the treatment of primary childhood hypertension and BP reduction or other outcomes in adulthood. The panel also was unable to identify any studies that reported on harms of screening and treatment.
When the adult framework for cardiovascular risk reduction is extended in pediatric patients, there are methodological challenges that make it harder to determine how much of the potential burden can actually be prevented, the panel said. The clinical and epidemiologic significance of percentile thresholds that are used to determine their ties to adult CVD has limited supporting evidence. Inconsistent performance characteristics of current diagnostic methods, of which there are few, tend to yield unfavorable high false-positive rates. Such false positives are potentially harmful, because they lead to “unnecessary secondary evaluations or treatments.” Because pharmacologic management of pediatric hypertension is continued for a much longer period, it is the increased likelihood of adverse events that should be cause for concern.
Should the focus for screening be shifted to significant risk factors?
In an accompanying editorial, Joseph T. Flynn, MD, MS, of Seattle Children’s Hospital, said that the outcome of the latest statement is expected, “given how the key questions were framed and the analysis performed.” To begin, he suggested restating the question: “What is the best approach to assess whether childhood BP measurement is associated with adult CVD or whether treatment of high BP in childhood is associated with reducing the burden of adult CVD?” The answer is to tackle these questions with randomized clinical trials that compare screening to no screening and treatment to no treatment. But such studies are likely infeasible, partly because of the required length of follow-up of 5-6 decades.
Perhaps a better question would be: “Does BP measurement in childhood identify children and adolescents who already have markers of CVD or who are at risk of developing them as adults?” Were these youth to be identified, they would become candidates for approaches that seek to prevent disease progression. Reframing the question in this manner better positions physicians to focus on prevention and sidestep “the requirement that the only acceptable outcome is prevention of CVD events in adulthood,” he explained.
The next step would be to identify data already available to address the reframed question. Cross-sectional studies could be used to make the association between BP levels and cardiovascular risk markers already present. For example, several publications from the multicenter Study of High Blood Pressure in Pediatrics: Adult Hypertension Onset in Youth (SHIP-AHOY), which enrolled roughly 400 youth, provided data that reinforce prior single-center studies that essentially proved there are adverse consequences for youth with high BP, and they “set the stage for the institution of measures designed to reverse target-organ damage and reduce cardiovascular risk in youth,” said Dr. Flynn.
More specifically, results from SHIP-AHOY “have demonstrated that increased left ventricular mass can be demonstrated at BP levels currently classified as normotensive and that abnormal left ventricular function can be seen at similar BP levels,” Dr. Flynn noted. In addition, “they have established a substantial association between an abnormal metabolic phenotype and several forms of target-organ damage associated with high BP.”
One approach is to analyze longitudinal cohort studies
Because there is a paucity of prospective clinical trials, Dr. Flynn suggested that analyzing longitudinal cohort studies would be the most effective approach for evaluating the potential link between current BP levels and future CVD. Such studies already have “data that address an important point raised in the USPSTF statement, namely whether the pediatric percentile-based BP cut points, such as those in the 2017 AAP [American Academy of Pediatrics] guideline, are associated with adult hypertension and CVD,” noted Dr. Flynn. “In the International Childhood Cardiovascular Cohort Consortium study, the specific childhood BP levels that were associated with increased adult carotid intima-medial thickness were remarkably similar to the BP percentile cut points in the AAP guideline for children of similar ages.”
Analysis of data from the Bogalusa Heart Study found looking at children classified as having high BP by the 2017 AAP guideline had “increased relative risks of having hypertension, left ventricular hypertrophy, or metabolic syndrome as adults 36 years later.”
“The conclusions of the USPSTF statement underscore the need for additional research on childhood high BP and its association with adult CVD. The starting points for such research can be deduced from currently available cross-sectional and longitudinal data, which demonstrate the detrimental outcomes associated with high BP in youth. Using these data to reframe and answer the questions raised by the USPSTF should point the way toward effective prevention of adult CVD,” concluded Dr. Flynn.
In a separate interview, Kristen Sexson Tejtel, MD, PhD, MPH, medical director of the preventive cardiology clinic at Texas Children’s Hospital and Baylor College of Medicine, both in Houston, noted that in spite of USPSTF’s findings, there is actually an association between children with high blood pressure and intermediate outcomes in adults.
“Dr. Flynn suggests reframing the question. In fact, evidence exists that children with high blood pressure are at higher risk of left ventricular hypertrophy, increased arterial stiffness, and changes in retinal arteries,” noted Dr. Sexson Tejtel.
Evidence of pediatric heart damage has been documented in autopsies
“It is imperative that children have blood pressure evaluation,” she urged. “There is evidence that there are changes similar to those seen in adults with cardiovascular compromise. It has been shown that children dying of other causes [accidents] who have these problems also have more plaque on autopsy, indicating that those with high blood pressure are more likely to have markers of CVD already present in childhood.
“One of the keys of pediatric medicine is prevention and the counseling for prevention of adult diseases. The duration of study necessary to objectively determine whether treatment of hypertension in childhood reduces the risk of adult cardiac problems is extensive. If nothing is done now, we are putting more future generations in danger. We must provide appropriate counseling for children and their families regarding lifestyle improvements, to have a chance to improve cardiovascular risk factors in adults, including hypertension, hyperlipidemia and/or obesity,” urged Dr. Sexson Tejtel.
All members of the USPSTF received travel reimbursement and honoraria. Dr. Barry received grants and personal fees from Healthwise. The U.S. Congress mandates that the Agency for Healthcare Research and Quality support the operations of the USPSTF. Dr. Flynn reported receiving grants from the National Institutes of Health and royalties from UpToDate and Springer outside the submitted work. Dr. Sexson Tejtel said she had no relevant financial disclosures or conflicts of interest.
SOURCE: USPSTF. JAMA. 2020 Nov 10. doi: 10.1001/jama.2020.20122.
Current evidence is insufficient to assess the balance of benefits and harms of screening for high blood pressure in children and adolescents, the U.S. Preventive Services Task Force reported in JAMA.
However, two experts in this area suggested there is evidence if you know where to look, and pediatric BP testing is crucial now.
In this update to the 2013 statement, the USPSTF’s systematic review focused on evidence surrounding the benefits of screening, test accuracy, treatment effectiveness and harms, and links between hypertension and cardiovascular disease (CVD) markers in childhood and adulthood.
Limited information was available on the accuracy of screening tests. No studies were found that directly evaluated screening for pediatric high BP or reported effectiveness in delayed onset or risk reduction for cardiovascular outcomes related to hypertension. Additionally, no studies were found that addressed screening for secondary hypertension in asymptomatic pediatric patients. No studies were found that evaluated the treatment of primary childhood hypertension and BP reduction or other outcomes in adulthood. The panel also was unable to identify any studies that reported on harms of screening and treatment.
When the adult framework for cardiovascular risk reduction is extended in pediatric patients, there are methodological challenges that make it harder to determine how much of the potential burden can actually be prevented, the panel said. The clinical and epidemiologic significance of percentile thresholds that are used to determine their ties to adult CVD has limited supporting evidence. Inconsistent performance characteristics of current diagnostic methods, of which there are few, tend to yield unfavorable high false-positive rates. Such false positives are potentially harmful, because they lead to “unnecessary secondary evaluations or treatments.” Because pharmacologic management of pediatric hypertension is continued for a much longer period, it is the increased likelihood of adverse events that should be cause for concern.
Should the focus for screening be shifted to significant risk factors?
In an accompanying editorial, Joseph T. Flynn, MD, MS, of Seattle Children’s Hospital, said that the outcome of the latest statement is expected, “given how the key questions were framed and the analysis performed.” To begin, he suggested restating the question: “What is the best approach to assess whether childhood BP measurement is associated with adult CVD or whether treatment of high BP in childhood is associated with reducing the burden of adult CVD?” The answer is to tackle these questions with randomized clinical trials that compare screening to no screening and treatment to no treatment. But such studies are likely infeasible, partly because of the required length of follow-up of 5-6 decades.
Perhaps a better question would be: “Does BP measurement in childhood identify children and adolescents who already have markers of CVD or who are at risk of developing them as adults?” Were these youth to be identified, they would become candidates for approaches that seek to prevent disease progression. Reframing the question in this manner better positions physicians to focus on prevention and sidestep “the requirement that the only acceptable outcome is prevention of CVD events in adulthood,” he explained.
The next step would be to identify data already available to address the reframed question. Cross-sectional studies could be used to make the association between BP levels and cardiovascular risk markers already present. For example, several publications from the multicenter Study of High Blood Pressure in Pediatrics: Adult Hypertension Onset in Youth (SHIP-AHOY), which enrolled roughly 400 youth, provided data that reinforce prior single-center studies that essentially proved there are adverse consequences for youth with high BP, and they “set the stage for the institution of measures designed to reverse target-organ damage and reduce cardiovascular risk in youth,” said Dr. Flynn.
More specifically, results from SHIP-AHOY “have demonstrated that increased left ventricular mass can be demonstrated at BP levels currently classified as normotensive and that abnormal left ventricular function can be seen at similar BP levels,” Dr. Flynn noted. In addition, “they have established a substantial association between an abnormal metabolic phenotype and several forms of target-organ damage associated with high BP.”
One approach is to analyze longitudinal cohort studies
Because there is a paucity of prospective clinical trials, Dr. Flynn suggested that analyzing longitudinal cohort studies would be the most effective approach for evaluating the potential link between current BP levels and future CVD. Such studies already have “data that address an important point raised in the USPSTF statement, namely whether the pediatric percentile-based BP cut points, such as those in the 2017 AAP [American Academy of Pediatrics] guideline, are associated with adult hypertension and CVD,” noted Dr. Flynn. “In the International Childhood Cardiovascular Cohort Consortium study, the specific childhood BP levels that were associated with increased adult carotid intima-medial thickness were remarkably similar to the BP percentile cut points in the AAP guideline for children of similar ages.”
Analysis of data from the Bogalusa Heart Study found looking at children classified as having high BP by the 2017 AAP guideline had “increased relative risks of having hypertension, left ventricular hypertrophy, or metabolic syndrome as adults 36 years later.”
“The conclusions of the USPSTF statement underscore the need for additional research on childhood high BP and its association with adult CVD. The starting points for such research can be deduced from currently available cross-sectional and longitudinal data, which demonstrate the detrimental outcomes associated with high BP in youth. Using these data to reframe and answer the questions raised by the USPSTF should point the way toward effective prevention of adult CVD,” concluded Dr. Flynn.
In a separate interview, Kristen Sexson Tejtel, MD, PhD, MPH, medical director of the preventive cardiology clinic at Texas Children’s Hospital and Baylor College of Medicine, both in Houston, noted that in spite of USPSTF’s findings, there is actually an association between children with high blood pressure and intermediate outcomes in adults.
“Dr. Flynn suggests reframing the question. In fact, evidence exists that children with high blood pressure are at higher risk of left ventricular hypertrophy, increased arterial stiffness, and changes in retinal arteries,” noted Dr. Sexson Tejtel.
Evidence of pediatric heart damage has been documented in autopsies
“It is imperative that children have blood pressure evaluation,” she urged. “There is evidence that there are changes similar to those seen in adults with cardiovascular compromise. It has been shown that children dying of other causes [accidents] who have these problems also have more plaque on autopsy, indicating that those with high blood pressure are more likely to have markers of CVD already present in childhood.
“One of the keys of pediatric medicine is prevention and the counseling for prevention of adult diseases. The duration of study necessary to objectively determine whether treatment of hypertension in childhood reduces the risk of adult cardiac problems is extensive. If nothing is done now, we are putting more future generations in danger. We must provide appropriate counseling for children and their families regarding lifestyle improvements, to have a chance to improve cardiovascular risk factors in adults, including hypertension, hyperlipidemia and/or obesity,” urged Dr. Sexson Tejtel.
All members of the USPSTF received travel reimbursement and honoraria. Dr. Barry received grants and personal fees from Healthwise. The U.S. Congress mandates that the Agency for Healthcare Research and Quality support the operations of the USPSTF. Dr. Flynn reported receiving grants from the National Institutes of Health and royalties from UpToDate and Springer outside the submitted work. Dr. Sexson Tejtel said she had no relevant financial disclosures or conflicts of interest.
SOURCE: USPSTF. JAMA. 2020 Nov 10. doi: 10.1001/jama.2020.20122.
Current evidence is insufficient to assess the balance of benefits and harms of screening for high blood pressure in children and adolescents, the U.S. Preventive Services Task Force reported in JAMA.
However, two experts in this area suggested there is evidence if you know where to look, and pediatric BP testing is crucial now.
In this update to the 2013 statement, the USPSTF’s systematic review focused on evidence surrounding the benefits of screening, test accuracy, treatment effectiveness and harms, and links between hypertension and cardiovascular disease (CVD) markers in childhood and adulthood.
Limited information was available on the accuracy of screening tests. No studies were found that directly evaluated screening for pediatric high BP or reported effectiveness in delayed onset or risk reduction for cardiovascular outcomes related to hypertension. Additionally, no studies were found that addressed screening for secondary hypertension in asymptomatic pediatric patients. No studies were found that evaluated the treatment of primary childhood hypertension and BP reduction or other outcomes in adulthood. The panel also was unable to identify any studies that reported on harms of screening and treatment.
When the adult framework for cardiovascular risk reduction is extended in pediatric patients, there are methodological challenges that make it harder to determine how much of the potential burden can actually be prevented, the panel said. The clinical and epidemiologic significance of percentile thresholds that are used to determine their ties to adult CVD has limited supporting evidence. Inconsistent performance characteristics of current diagnostic methods, of which there are few, tend to yield unfavorable high false-positive rates. Such false positives are potentially harmful, because they lead to “unnecessary secondary evaluations or treatments.” Because pharmacologic management of pediatric hypertension is continued for a much longer period, it is the increased likelihood of adverse events that should be cause for concern.
Should the focus for screening be shifted to significant risk factors?
In an accompanying editorial, Joseph T. Flynn, MD, MS, of Seattle Children’s Hospital, said that the outcome of the latest statement is expected, “given how the key questions were framed and the analysis performed.” To begin, he suggested restating the question: “What is the best approach to assess whether childhood BP measurement is associated with adult CVD or whether treatment of high BP in childhood is associated with reducing the burden of adult CVD?” The answer is to tackle these questions with randomized clinical trials that compare screening to no screening and treatment to no treatment. But such studies are likely infeasible, partly because of the required length of follow-up of 5-6 decades.
Perhaps a better question would be: “Does BP measurement in childhood identify children and adolescents who already have markers of CVD or who are at risk of developing them as adults?” Were these youth to be identified, they would become candidates for approaches that seek to prevent disease progression. Reframing the question in this manner better positions physicians to focus on prevention and sidestep “the requirement that the only acceptable outcome is prevention of CVD events in adulthood,” he explained.
The next step would be to identify data already available to address the reframed question. Cross-sectional studies could be used to make the association between BP levels and cardiovascular risk markers already present. For example, several publications from the multicenter Study of High Blood Pressure in Pediatrics: Adult Hypertension Onset in Youth (SHIP-AHOY), which enrolled roughly 400 youth, provided data that reinforce prior single-center studies that essentially proved there are adverse consequences for youth with high BP, and they “set the stage for the institution of measures designed to reverse target-organ damage and reduce cardiovascular risk in youth,” said Dr. Flynn.
More specifically, results from SHIP-AHOY “have demonstrated that increased left ventricular mass can be demonstrated at BP levels currently classified as normotensive and that abnormal left ventricular function can be seen at similar BP levels,” Dr. Flynn noted. In addition, “they have established a substantial association between an abnormal metabolic phenotype and several forms of target-organ damage associated with high BP.”
One approach is to analyze longitudinal cohort studies
Because there is a paucity of prospective clinical trials, Dr. Flynn suggested that analyzing longitudinal cohort studies would be the most effective approach for evaluating the potential link between current BP levels and future CVD. Such studies already have “data that address an important point raised in the USPSTF statement, namely whether the pediatric percentile-based BP cut points, such as those in the 2017 AAP [American Academy of Pediatrics] guideline, are associated with adult hypertension and CVD,” noted Dr. Flynn. “In the International Childhood Cardiovascular Cohort Consortium study, the specific childhood BP levels that were associated with increased adult carotid intima-medial thickness were remarkably similar to the BP percentile cut points in the AAP guideline for children of similar ages.”
Analysis of data from the Bogalusa Heart Study found looking at children classified as having high BP by the 2017 AAP guideline had “increased relative risks of having hypertension, left ventricular hypertrophy, or metabolic syndrome as adults 36 years later.”
“The conclusions of the USPSTF statement underscore the need for additional research on childhood high BP and its association with adult CVD. The starting points for such research can be deduced from currently available cross-sectional and longitudinal data, which demonstrate the detrimental outcomes associated with high BP in youth. Using these data to reframe and answer the questions raised by the USPSTF should point the way toward effective prevention of adult CVD,” concluded Dr. Flynn.
In a separate interview, Kristen Sexson Tejtel, MD, PhD, MPH, medical director of the preventive cardiology clinic at Texas Children’s Hospital and Baylor College of Medicine, both in Houston, noted that in spite of USPSTF’s findings, there is actually an association between children with high blood pressure and intermediate outcomes in adults.
“Dr. Flynn suggests reframing the question. In fact, evidence exists that children with high blood pressure are at higher risk of left ventricular hypertrophy, increased arterial stiffness, and changes in retinal arteries,” noted Dr. Sexson Tejtel.
Evidence of pediatric heart damage has been documented in autopsies
“It is imperative that children have blood pressure evaluation,” she urged. “There is evidence that there are changes similar to those seen in adults with cardiovascular compromise. It has been shown that children dying of other causes [accidents] who have these problems also have more plaque on autopsy, indicating that those with high blood pressure are more likely to have markers of CVD already present in childhood.
“One of the keys of pediatric medicine is prevention and the counseling for prevention of adult diseases. The duration of study necessary to objectively determine whether treatment of hypertension in childhood reduces the risk of adult cardiac problems is extensive. If nothing is done now, we are putting more future generations in danger. We must provide appropriate counseling for children and their families regarding lifestyle improvements, to have a chance to improve cardiovascular risk factors in adults, including hypertension, hyperlipidemia and/or obesity,” urged Dr. Sexson Tejtel.
All members of the USPSTF received travel reimbursement and honoraria. Dr. Barry received grants and personal fees from Healthwise. The U.S. Congress mandates that the Agency for Healthcare Research and Quality support the operations of the USPSTF. Dr. Flynn reported receiving grants from the National Institutes of Health and royalties from UpToDate and Springer outside the submitted work. Dr. Sexson Tejtel said she had no relevant financial disclosures or conflicts of interest.
SOURCE: USPSTF. JAMA. 2020 Nov 10. doi: 10.1001/jama.2020.20122.
FROM JAMA
Liver-related deaths decline after Medicaid expansion under ACA
Liver-related deaths declined and liver transplant waitlist inequities decreased in states that implemented Medicaid expansion under the Affordable Care Act (ACA), results of an innovative study showed.
About 1 year after Medicaid expansion began on Jan. 1, 2014, the rate of liver-related mortality in 18 states that took advantage of expanded coverage began to decline, whereas the rate of liver-related deaths in 14 states that did not expand Medicaid continued to climb, reported Nabeel Wahid, MD, a resident at Weill Cornell Medicine, New York.
The differences in liver-related mortality between Medicaid expansion and nonexpansion states was particularly pronounced in people of Asian background, in Whites, and in African Americans, he said in an oral abstract presentation during the virtual annual meeting of the American Association for the Study of Liver Diseases.
“The expansion of government health care programs such as Medicaid may improve liver-related mortality and liver transplant waitlist placement,” he said.
The implementation of the ACA, also known as Obamacare, resulted in “a pretty dramatic increase in the number of Americans with health insurance today, and there’s a lot of literature out there looking at a variety of domains throughout health care that have found that Medicaid expansion increased access and decreased disparities in care,” he added.
For example, a report published in 2019 by the nonpartisan National Bureau of Economic Research Priorities showed a significant reduction in disease-related deaths in Americans aged 55-64 in Medicaid expansion states compared with nonexpansion states.
In addition as previously reported, before Medicaid expansion African Americans were 4.8% less likely than were Whites to receive timely cancer treatment, defined as treatment starting within 30 days of diagnosis of an advanced or metastatic solid tumor. After Medicaid expansion, however, the difference between the racial groups had dwindled to just 0.8% and was no longer statistically significant.
“However, specifically in the realm of liver transplantation and liver disease, there’s very limited literature showing any sort of significant impact on care resulting from Medicaid expansion,” Dr. Wahid said.
Listing-to-death ratio
To test their hypothesis that Medicaid expansion decreased racial disparities and improved liver-related deaths and transplant waitlist placement, Dr. Wahid and colleagues compared liver-related deaths and liver transplants listings between Medicaid expansion and nonexpansion states in the 5 years before expansion (2009-2013) and in the 5 years after expansion (2014-2018). They excluded all states without transplant centers as well as patients younger than 25 or older than 64, who were likely to be covered by other types of insurance.
They obtained data for listing from the United Network for Organ Sharing (UNOS) and on end-stage liver disease from the Centers for Disease Control and Prevention’s WONDER database.
They also used a novel measure called the listing-to-death ratio (LDR), a surrogate endpoint for waitlist placement calculated as the ratio of listings for liver transplantation relative to the number of deaths from liver disease, with a higher LDR score corresponding to improved waitlist placement.
They found that throughout the entire study period, Medicaid expansion states had lower liver-related deaths, higher liver transplant listings, and higher LDR.
Using joinpoint regression to examine changes at a specific time point, the investigators determined that the annual percentage change in liver-related deaths increased in both nonexpansion states (mean 4.3%) and expansion states (3.0%) before 2014. However, beginning around 2015, liver-related deaths began to decline in expansion states by a mean of –0.6%, while they continued on an upward trajectory in the nonexpansion states, albeit at a somewhat slower pace (mean APC 2% from 2014 through 2018).
Among all racial and ethnic groups (Whites, African Americans, Hispanics, and Asians) liver-related deaths increased from 2014 to 2018 in nonexpansion states, with the highest annual percentage change in Asians, at slightly more than 8%.
In contrast, among Asians in the expansion states, liver-related deaths over the same period increased by less than 1%, and in both Whites and African Americans liver-related deaths declined.
In addition, starting in 2015, the annual percent change in LDR increased only in expansion states primarily because of fewer end-stage liver disease deaths (the denominator in the LDR equation) rather than increased listings (the numerator).
‘No-brainer’
A gastroenterologist who was not involved in the study said that there are two key points in favor of the study.
“The obvious message is that transplant is costly, and patients need to be insured to get a liver transplant, because nobody is paying for this out of pocket. So if more candidates are insured that will reduce disparities and improve access to liver transplant for those who need it,” said Elliot Benjamin Tapper, MD, of the University of Michigan, Ann Arbor.
“It’s a no-brainer that the lack of insurance accessibility for the most vulnerable people in the United States meant that they were dying of cirrhosis instead of being transplanted,” he said in an interview.
He also commended the authors on their use of population-based data to identify outcomes.
“We know how many people are listed for liver transplant, but we don’t know how many people could have been listed. We know how many people are transplanted, but we don’t know how many people with decompensated cirrhosis should have been given that chance. We lacked that denominator,” he said.
“The innovation that makes this particular paper worthwhile is that in the absence of that denominator, they were able to construct it, so we can know from other data sources distinct from the waitlist rolls how many people are dying of cirrhosis,” he added.
No study funding source was reported. Dr. Wahid and Dr. Tapper reported no conflicts of interest.
SOURCE: Wahid N et al. AASLD 2020. Abstract 153.
Liver-related deaths declined and liver transplant waitlist inequities decreased in states that implemented Medicaid expansion under the Affordable Care Act (ACA), results of an innovative study showed.
About 1 year after Medicaid expansion began on Jan. 1, 2014, the rate of liver-related mortality in 18 states that took advantage of expanded coverage began to decline, whereas the rate of liver-related deaths in 14 states that did not expand Medicaid continued to climb, reported Nabeel Wahid, MD, a resident at Weill Cornell Medicine, New York.
The differences in liver-related mortality between Medicaid expansion and nonexpansion states was particularly pronounced in people of Asian background, in Whites, and in African Americans, he said in an oral abstract presentation during the virtual annual meeting of the American Association for the Study of Liver Diseases.
“The expansion of government health care programs such as Medicaid may improve liver-related mortality and liver transplant waitlist placement,” he said.
The implementation of the ACA, also known as Obamacare, resulted in “a pretty dramatic increase in the number of Americans with health insurance today, and there’s a lot of literature out there looking at a variety of domains throughout health care that have found that Medicaid expansion increased access and decreased disparities in care,” he added.
For example, a report published in 2019 by the nonpartisan National Bureau of Economic Research Priorities showed a significant reduction in disease-related deaths in Americans aged 55-64 in Medicaid expansion states compared with nonexpansion states.
In addition as previously reported, before Medicaid expansion African Americans were 4.8% less likely than were Whites to receive timely cancer treatment, defined as treatment starting within 30 days of diagnosis of an advanced or metastatic solid tumor. After Medicaid expansion, however, the difference between the racial groups had dwindled to just 0.8% and was no longer statistically significant.
“However, specifically in the realm of liver transplantation and liver disease, there’s very limited literature showing any sort of significant impact on care resulting from Medicaid expansion,” Dr. Wahid said.
Listing-to-death ratio
To test their hypothesis that Medicaid expansion decreased racial disparities and improved liver-related deaths and transplant waitlist placement, Dr. Wahid and colleagues compared liver-related deaths and liver transplants listings between Medicaid expansion and nonexpansion states in the 5 years before expansion (2009-2013) and in the 5 years after expansion (2014-2018). They excluded all states without transplant centers as well as patients younger than 25 or older than 64, who were likely to be covered by other types of insurance.
They obtained data for listing from the United Network for Organ Sharing (UNOS) and on end-stage liver disease from the Centers for Disease Control and Prevention’s WONDER database.
They also used a novel measure called the listing-to-death ratio (LDR), a surrogate endpoint for waitlist placement calculated as the ratio of listings for liver transplantation relative to the number of deaths from liver disease, with a higher LDR score corresponding to improved waitlist placement.
They found that throughout the entire study period, Medicaid expansion states had lower liver-related deaths, higher liver transplant listings, and higher LDR.
Using joinpoint regression to examine changes at a specific time point, the investigators determined that the annual percentage change in liver-related deaths increased in both nonexpansion states (mean 4.3%) and expansion states (3.0%) before 2014. However, beginning around 2015, liver-related deaths began to decline in expansion states by a mean of –0.6%, while they continued on an upward trajectory in the nonexpansion states, albeit at a somewhat slower pace (mean APC 2% from 2014 through 2018).
Among all racial and ethnic groups (Whites, African Americans, Hispanics, and Asians) liver-related deaths increased from 2014 to 2018 in nonexpansion states, with the highest annual percentage change in Asians, at slightly more than 8%.
In contrast, among Asians in the expansion states, liver-related deaths over the same period increased by less than 1%, and in both Whites and African Americans liver-related deaths declined.
In addition, starting in 2015, the annual percent change in LDR increased only in expansion states primarily because of fewer end-stage liver disease deaths (the denominator in the LDR equation) rather than increased listings (the numerator).
‘No-brainer’
A gastroenterologist who was not involved in the study said that there are two key points in favor of the study.
“The obvious message is that transplant is costly, and patients need to be insured to get a liver transplant, because nobody is paying for this out of pocket. So if more candidates are insured that will reduce disparities and improve access to liver transplant for those who need it,” said Elliot Benjamin Tapper, MD, of the University of Michigan, Ann Arbor.
“It’s a no-brainer that the lack of insurance accessibility for the most vulnerable people in the United States meant that they were dying of cirrhosis instead of being transplanted,” he said in an interview.
He also commended the authors on their use of population-based data to identify outcomes.
“We know how many people are listed for liver transplant, but we don’t know how many people could have been listed. We know how many people are transplanted, but we don’t know how many people with decompensated cirrhosis should have been given that chance. We lacked that denominator,” he said.
“The innovation that makes this particular paper worthwhile is that in the absence of that denominator, they were able to construct it, so we can know from other data sources distinct from the waitlist rolls how many people are dying of cirrhosis,” he added.
No study funding source was reported. Dr. Wahid and Dr. Tapper reported no conflicts of interest.
SOURCE: Wahid N et al. AASLD 2020. Abstract 153.
Liver-related deaths declined and liver transplant waitlist inequities decreased in states that implemented Medicaid expansion under the Affordable Care Act (ACA), results of an innovative study showed.
About 1 year after Medicaid expansion began on Jan. 1, 2014, the rate of liver-related mortality in 18 states that took advantage of expanded coverage began to decline, whereas the rate of liver-related deaths in 14 states that did not expand Medicaid continued to climb, reported Nabeel Wahid, MD, a resident at Weill Cornell Medicine, New York.
The differences in liver-related mortality between Medicaid expansion and nonexpansion states was particularly pronounced in people of Asian background, in Whites, and in African Americans, he said in an oral abstract presentation during the virtual annual meeting of the American Association for the Study of Liver Diseases.
“The expansion of government health care programs such as Medicaid may improve liver-related mortality and liver transplant waitlist placement,” he said.
The implementation of the ACA, also known as Obamacare, resulted in “a pretty dramatic increase in the number of Americans with health insurance today, and there’s a lot of literature out there looking at a variety of domains throughout health care that have found that Medicaid expansion increased access and decreased disparities in care,” he added.
For example, a report published in 2019 by the nonpartisan National Bureau of Economic Research Priorities showed a significant reduction in disease-related deaths in Americans aged 55-64 in Medicaid expansion states compared with nonexpansion states.
In addition as previously reported, before Medicaid expansion African Americans were 4.8% less likely than were Whites to receive timely cancer treatment, defined as treatment starting within 30 days of diagnosis of an advanced or metastatic solid tumor. After Medicaid expansion, however, the difference between the racial groups had dwindled to just 0.8% and was no longer statistically significant.
“However, specifically in the realm of liver transplantation and liver disease, there’s very limited literature showing any sort of significant impact on care resulting from Medicaid expansion,” Dr. Wahid said.
Listing-to-death ratio
To test their hypothesis that Medicaid expansion decreased racial disparities and improved liver-related deaths and transplant waitlist placement, Dr. Wahid and colleagues compared liver-related deaths and liver transplants listings between Medicaid expansion and nonexpansion states in the 5 years before expansion (2009-2013) and in the 5 years after expansion (2014-2018). They excluded all states without transplant centers as well as patients younger than 25 or older than 64, who were likely to be covered by other types of insurance.
They obtained data for listing from the United Network for Organ Sharing (UNOS) and on end-stage liver disease from the Centers for Disease Control and Prevention’s WONDER database.
They also used a novel measure called the listing-to-death ratio (LDR), a surrogate endpoint for waitlist placement calculated as the ratio of listings for liver transplantation relative to the number of deaths from liver disease, with a higher LDR score corresponding to improved waitlist placement.
They found that throughout the entire study period, Medicaid expansion states had lower liver-related deaths, higher liver transplant listings, and higher LDR.
Using joinpoint regression to examine changes at a specific time point, the investigators determined that the annual percentage change in liver-related deaths increased in both nonexpansion states (mean 4.3%) and expansion states (3.0%) before 2014. However, beginning around 2015, liver-related deaths began to decline in expansion states by a mean of –0.6%, while they continued on an upward trajectory in the nonexpansion states, albeit at a somewhat slower pace (mean APC 2% from 2014 through 2018).
Among all racial and ethnic groups (Whites, African Americans, Hispanics, and Asians) liver-related deaths increased from 2014 to 2018 in nonexpansion states, with the highest annual percentage change in Asians, at slightly more than 8%.
In contrast, among Asians in the expansion states, liver-related deaths over the same period increased by less than 1%, and in both Whites and African Americans liver-related deaths declined.
In addition, starting in 2015, the annual percent change in LDR increased only in expansion states primarily because of fewer end-stage liver disease deaths (the denominator in the LDR equation) rather than increased listings (the numerator).
‘No-brainer’
A gastroenterologist who was not involved in the study said that there are two key points in favor of the study.
“The obvious message is that transplant is costly, and patients need to be insured to get a liver transplant, because nobody is paying for this out of pocket. So if more candidates are insured that will reduce disparities and improve access to liver transplant for those who need it,” said Elliot Benjamin Tapper, MD, of the University of Michigan, Ann Arbor.
“It’s a no-brainer that the lack of insurance accessibility for the most vulnerable people in the United States meant that they were dying of cirrhosis instead of being transplanted,” he said in an interview.
He also commended the authors on their use of population-based data to identify outcomes.
“We know how many people are listed for liver transplant, but we don’t know how many people could have been listed. We know how many people are transplanted, but we don’t know how many people with decompensated cirrhosis should have been given that chance. We lacked that denominator,” he said.
“The innovation that makes this particular paper worthwhile is that in the absence of that denominator, they were able to construct it, so we can know from other data sources distinct from the waitlist rolls how many people are dying of cirrhosis,” he added.
No study funding source was reported. Dr. Wahid and Dr. Tapper reported no conflicts of interest.
SOURCE: Wahid N et al. AASLD 2020. Abstract 153.
FROM THE LIVER MEETING DIGITAL EXPERIENCE
Liver injury linked to COVID-19–related coagulopathy
There is a link between liver injury and a tendency toward excessive clotting in patients with COVID-19, and the organ’s own blood vessels could be responsible, new research shows.
Cells that line the liver’s blood vessels produce high levels of factor VIII, a coagulation factor, when they are exposed to interleukin-6, an inflammatory molecule associated with COVID-19.
These findings “center the liver in global coagulopathy of COVID-19 and define a mechanism for increased coagulation factor levels that may be treatment targets,” said investigator Matthew McConnell, MD, from the Yale University, New Haven, Conn.
The effect of IL-6 on the liver sinusoidal endothelial cells lining the liver blood vessels creates a prothrombotic environment that includes the release of factor VIII, said Dr. McConnell, who presented the results at the virtual annual meeting of the American Association for the Study of Liver Diseases.
These associations offer insights into why COVID-19 patients with underlying liver disease can experience “devastating complications” related to improper blood vessel function in the organ, he added.
For their study, Dr. McConnell and colleagues analyzed data on ALT and hypercoagulability from 68 adults treated at the Yale–New Haven Hospital. The liver and coagulation tests were administered within 5 days of each other.
The team set the ALT cutoff for liver injury at three times the upper limit of normal. Patients with two or more parameters indicating excessive clotting were considered to have a hypercoagulable profile, which Dr. McConnell called “a signature clinical finding of COVID-19 infection.”
Patients with high levels of ALT also experienced elevations in clotting-related factors, such as fibrinogen levels and the activity of factor VIII and factor II. Furthermore, liver injury was significantly associated with hypercoagulability (P < .05).
Because COVID-19 is linked to the proinflammatory IL-6, the investigators examined how this cytokine and its receptor affect human liver sinusoidal cells. Cells exposed to IL-6 and its receptor pumped out factor VIII at levels that were significantly higher than in unexposed cells (P < .01). Exposed cells also produced significantly more von Willebrand factor (P < .05), another prothrombotic molecule, and showed increased expression of genes that induce the expression of factor VIII.
“As we learn more about COVID-19, we find that it is as much a coagulatory as a respiratory disease,” said Tien Dong, MD, PhD, from the University of California, Los Angeles, who was not involved in the study.
These findings are in line with a lot of other COVID-19-related research that suggests a link between hepatocyte injury and clotting disorders, he added.
One important factor is existing liver disease, said Dr. Dong. “If you have COVID-19 on top of that, you’re probably at risk of developing acute liver injury from the infection itself.”
That said, it’s still a good idea to check liver function in patients with COVID-19 and no known liver disease, he advised. Staying on top of these measures will keep the odds of long-term problems “a lot lower.”
There is utility in the findings beyond COVID-19, said Dr. McConnell. They provide “insights into complications of critical illness, in general, in the liver blood vessels” of patients with underlying liver disease.
Dr. McConnell and Dr. Dong have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
There is a link between liver injury and a tendency toward excessive clotting in patients with COVID-19, and the organ’s own blood vessels could be responsible, new research shows.
Cells that line the liver’s blood vessels produce high levels of factor VIII, a coagulation factor, when they are exposed to interleukin-6, an inflammatory molecule associated with COVID-19.
These findings “center the liver in global coagulopathy of COVID-19 and define a mechanism for increased coagulation factor levels that may be treatment targets,” said investigator Matthew McConnell, MD, from the Yale University, New Haven, Conn.
The effect of IL-6 on the liver sinusoidal endothelial cells lining the liver blood vessels creates a prothrombotic environment that includes the release of factor VIII, said Dr. McConnell, who presented the results at the virtual annual meeting of the American Association for the Study of Liver Diseases.
These associations offer insights into why COVID-19 patients with underlying liver disease can experience “devastating complications” related to improper blood vessel function in the organ, he added.
For their study, Dr. McConnell and colleagues analyzed data on ALT and hypercoagulability from 68 adults treated at the Yale–New Haven Hospital. The liver and coagulation tests were administered within 5 days of each other.
The team set the ALT cutoff for liver injury at three times the upper limit of normal. Patients with two or more parameters indicating excessive clotting were considered to have a hypercoagulable profile, which Dr. McConnell called “a signature clinical finding of COVID-19 infection.”
Patients with high levels of ALT also experienced elevations in clotting-related factors, such as fibrinogen levels and the activity of factor VIII and factor II. Furthermore, liver injury was significantly associated with hypercoagulability (P < .05).
Because COVID-19 is linked to the proinflammatory IL-6, the investigators examined how this cytokine and its receptor affect human liver sinusoidal cells. Cells exposed to IL-6 and its receptor pumped out factor VIII at levels that were significantly higher than in unexposed cells (P < .01). Exposed cells also produced significantly more von Willebrand factor (P < .05), another prothrombotic molecule, and showed increased expression of genes that induce the expression of factor VIII.
“As we learn more about COVID-19, we find that it is as much a coagulatory as a respiratory disease,” said Tien Dong, MD, PhD, from the University of California, Los Angeles, who was not involved in the study.
These findings are in line with a lot of other COVID-19-related research that suggests a link between hepatocyte injury and clotting disorders, he added.
One important factor is existing liver disease, said Dr. Dong. “If you have COVID-19 on top of that, you’re probably at risk of developing acute liver injury from the infection itself.”
That said, it’s still a good idea to check liver function in patients with COVID-19 and no known liver disease, he advised. Staying on top of these measures will keep the odds of long-term problems “a lot lower.”
There is utility in the findings beyond COVID-19, said Dr. McConnell. They provide “insights into complications of critical illness, in general, in the liver blood vessels” of patients with underlying liver disease.
Dr. McConnell and Dr. Dong have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
There is a link between liver injury and a tendency toward excessive clotting in patients with COVID-19, and the organ’s own blood vessels could be responsible, new research shows.
Cells that line the liver’s blood vessels produce high levels of factor VIII, a coagulation factor, when they are exposed to interleukin-6, an inflammatory molecule associated with COVID-19.
These findings “center the liver in global coagulopathy of COVID-19 and define a mechanism for increased coagulation factor levels that may be treatment targets,” said investigator Matthew McConnell, MD, from the Yale University, New Haven, Conn.
The effect of IL-6 on the liver sinusoidal endothelial cells lining the liver blood vessels creates a prothrombotic environment that includes the release of factor VIII, said Dr. McConnell, who presented the results at the virtual annual meeting of the American Association for the Study of Liver Diseases.
These associations offer insights into why COVID-19 patients with underlying liver disease can experience “devastating complications” related to improper blood vessel function in the organ, he added.
For their study, Dr. McConnell and colleagues analyzed data on ALT and hypercoagulability from 68 adults treated at the Yale–New Haven Hospital. The liver and coagulation tests were administered within 5 days of each other.
The team set the ALT cutoff for liver injury at three times the upper limit of normal. Patients with two or more parameters indicating excessive clotting were considered to have a hypercoagulable profile, which Dr. McConnell called “a signature clinical finding of COVID-19 infection.”
Patients with high levels of ALT also experienced elevations in clotting-related factors, such as fibrinogen levels and the activity of factor VIII and factor II. Furthermore, liver injury was significantly associated with hypercoagulability (P < .05).
Because COVID-19 is linked to the proinflammatory IL-6, the investigators examined how this cytokine and its receptor affect human liver sinusoidal cells. Cells exposed to IL-6 and its receptor pumped out factor VIII at levels that were significantly higher than in unexposed cells (P < .01). Exposed cells also produced significantly more von Willebrand factor (P < .05), another prothrombotic molecule, and showed increased expression of genes that induce the expression of factor VIII.
“As we learn more about COVID-19, we find that it is as much a coagulatory as a respiratory disease,” said Tien Dong, MD, PhD, from the University of California, Los Angeles, who was not involved in the study.
These findings are in line with a lot of other COVID-19-related research that suggests a link between hepatocyte injury and clotting disorders, he added.
One important factor is existing liver disease, said Dr. Dong. “If you have COVID-19 on top of that, you’re probably at risk of developing acute liver injury from the infection itself.”
That said, it’s still a good idea to check liver function in patients with COVID-19 and no known liver disease, he advised. Staying on top of these measures will keep the odds of long-term problems “a lot lower.”
There is utility in the findings beyond COVID-19, said Dr. McConnell. They provide “insights into complications of critical illness, in general, in the liver blood vessels” of patients with underlying liver disease.
Dr. McConnell and Dr. Dong have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Cardiac arrest in COVID-19 pandemic: ‘Survival is possible’
In the early weeks of the COVID-19 pandemic in the United States, rates of sustained return of spontaneous circulation after out-of-hospital cardiac arrest were lower throughout the country, compared with a year earlier, in one study.
A second study of that period showed that patients with COVID-19 had rates that were better than previously reported of surviving in-hospital cardiac arrest.
Paul S. Chan, MD, presented the out-of-hospital cardiac arrest research, and Oscar J. Mitchell, MD, presented the in-hospital cardiac arrest findings in a late-breaking resuscitation science session at the American Heart Association scientific sessions. The former study was also simultaneously published online Nov. 14 in JAMA Cardiology.
Importantly, “the survival rates were not zero in either setting,” said Dr. Chan, commenting on the implications of both studies taken together.
“The survival rates – either return of circulation or survival to discharge – were not futile,” Dr. Chan, from Saint Luke’s Mid America Heart Institute, Kansas City, Missouri, said in an interview.
“And I think that’s an overall important message – that we can’t write off patients who have a cardiac arrest at this point,” he stressed. “They deserve a response. Although the outcomes might not be as good as we had seen in years prior, we are seeing patients making it out of the hospital and surviving.”
Dr. Mitchell, from the University of Pennsylvania in Philadelphia, echoed this message in an interview.
“I think that the key finding here is that survival is possible after patients with COVID-19 suffer an in-hospital cardiac arrest,” Dr. Mitchell said. “We hope that the information from our study will be of use to frontline providers who are treating patients with COVID-19.”
“In coming weeks, there will likely be increased hospital strain and enormous challenges to providing COVID-19 care,” added Benjamin S. Abella, MD, the senior author of the in-hospital study. Dr. Abella is also from the University of Pennsylvania and was cochair of the Resuscitation Science symposium during the AHA meeting.
“It is crucial that hospital leaders prepare now for how they will manage COVID-19 resuscitation efforts,” Dr. Abella said. “Emergency medicine and critical care leaders must be mindful that many COVID-19 patients with arrest could survive to return to their families.”
“It is important to note both studies demonstrated variations in outcome and that those differences were associated with the differential COVID prevalence and mortality,” session comoderator Cindy H. Hsu, MD, PhD, University of Michigan, said in an interview.
“Future studies,” she said, “should address knowledge gaps including associated comorbidities and affected resuscitation process variables during the COVID-19 pandemic.”
Out-of-hospital cardiac arrest, March 2019 vs. March 2020
Compared with 2019, in 2020, the reported rates of return of spontaneous circulation after out-of-hospital cardiac arrest fell from 25% to 10.6% in New York and from 13.5% to 5.0% in northern Italy – two areas that were severely affected, Dr. Chan noted.
In this study, the researchers aimed to examine whether out-of-hospital cardiac arrest outcomes would be similar throughout the United States, including areas that were less severely affected, in the first weeks of the pandemic.
They linked data from the Cardiac Arrest Registry to Enhance Survival (CARES), which covers an area with about 152 million U.S. residents, with COVID-19 disease mortality data.
There were 9,863 out-of-hospital arrests from March 16 to April 30, 2020, compared with 9,440 cases during this time in 2019.
The patients in both years had a similar age (mean, 62 years) and sex (62% male), but there were more Black patients in 2020 (28% vs. 23%).
Overall, in communities with low to high rates of death from COVID-19, the rate of return of spontaneous circulation was 18% lower in that early pandemic period than in the same time in the previous year (23% vs. 29.8%; adjusted rate ratio, 0.82).
The rates of return of spontaneous circulation were also lower in communities with a low rate of COVID-19 mortality, but to a lesser extent (11%-15% lower in 2020 vs. 2019).
In the subset of emergency medical agencies with complete data on hospital survival, overall rates of survival to discharge were 17% lower during the studied pandemic period versus the same time a year earlier (6.6% vs. 9.8%; adjusted RR, 0.83).
This drop in survival was greater in communities with moderate to high COVID-19 mortality.
These outcomes were not explained by differences in emergency medical services arrival or treatment times, rates of bystander CPR, or initial out-of-hospital cardiac arrest rhythm.
Dr. Chan was a coauthor of an interim guidance issued April 9, 2020, by the AHA and several other medical societies for ways to protect frontline workers from contracting COVID-19 while they were performing CPR.
Communities that were not heavily affected by COVID-19 could have also been following the recommendations, which might have affected outcomes, he speculated.
For example, “when we pause chest compressions it can potentially worsen survival even if it’s for a short period of time. That might explain the lower rates of return of circulation.”
“That guidance was really meant for heavily affected communities,” Dr. Chan added. “Of course, as we speak, the pandemic is pretty much everywhere in the United States. It’s not just in the northeast; it’s not just in Arizona, Florida, California, Texas like it was in the summer. You are seeing surges in 46 of the 50 states.
“If your community is heavily affected by COVID-19 in terms of deaths at this time, paramedics will need to take caution to also help protect themselves, and the guidance may apply at that point,” he said.
In-hospital cardiac arrest, March Through May 2020
The early studies of in-hospital cardiac arrest in patients with COVID-19 showed “concerningly low rates” of return of spontaneous circulation and survival, said Dr. Mitchell.
“The first was a study from Wuhan, which demonstrated a 2.9% 30-day survival and the second was a small cohort from NYC with 0% survival to hospital discharge,” he said. “This raised concerns that offering CPR to patients who had a cardiac arrest from COVID-19 might only hold a low probability of success.”
To investigate this, the researchers formed a COVID study group comprising two hospitals in New York and nine hospitals in the Northeast and West Coast.
They identified 260 hospitalized adult patients with COVID-19 who had in-hospital cardiac arrest between March 1 and May 31, 2020. The patients had a median age of 69 years, and 72% were male. Most had preexisting comorbidities. Most of the cardiac arrests were in the ICU (64%), and almost all were witnessed (91%).
Return of spontaneous circulation occurred in 22% of the patients, and 12% had survived 30 days later. Of the 260 cardiac arrests, most (204) occurred in the New York hospitals.
There was a huge variation in outcomes. The rate of sustained return of spontaneous circulation was much lower in the two hospitals in New York compared with elsewhere (11% vs. 64%), as was 30-day survival (6% vs. 36%).
“Variation in outcomes from [in-hospital cardiac arrest] has been well described prior to the COVID-19 pandemic,” said Dr. Mitchell, “and is felt to be due to a range of factors, including variation in detection and prevention of cardiac arrest, management of patients during the cardiac arrest, and differences in postarrest care – including targeted temperature management and neuroprognostication.”
“We hypothesize that the strains of the COVID-19 pandemic may have amplified these variations (although we were unable to compare hospital performance before and after the pandemic),” he said.
Nevertheless, “in contrast to [earlier] studies, we have found that survival with a good neurological status is possible after in-hospital cardiac arrest in patients with COVID-19, which is certainly reassuring for those of us on the front line.”
Dr. Chan has received research support from the American Heart Association (which helps fund CARES); the National Heart, Lung, and Blood Institute; and Optum Rx. Dr. Abella has received honoraria from NeuroproteXeon, Becton Dickinson, and Physio-Control, and research grants from Medtronic, PCORI, Physio-Control, Stryker, and TerSera. Dr. Mitchell has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In the early weeks of the COVID-19 pandemic in the United States, rates of sustained return of spontaneous circulation after out-of-hospital cardiac arrest were lower throughout the country, compared with a year earlier, in one study.
A second study of that period showed that patients with COVID-19 had rates that were better than previously reported of surviving in-hospital cardiac arrest.
Paul S. Chan, MD, presented the out-of-hospital cardiac arrest research, and Oscar J. Mitchell, MD, presented the in-hospital cardiac arrest findings in a late-breaking resuscitation science session at the American Heart Association scientific sessions. The former study was also simultaneously published online Nov. 14 in JAMA Cardiology.
Importantly, “the survival rates were not zero in either setting,” said Dr. Chan, commenting on the implications of both studies taken together.
“The survival rates – either return of circulation or survival to discharge – were not futile,” Dr. Chan, from Saint Luke’s Mid America Heart Institute, Kansas City, Missouri, said in an interview.
“And I think that’s an overall important message – that we can’t write off patients who have a cardiac arrest at this point,” he stressed. “They deserve a response. Although the outcomes might not be as good as we had seen in years prior, we are seeing patients making it out of the hospital and surviving.”
Dr. Mitchell, from the University of Pennsylvania in Philadelphia, echoed this message in an interview.
“I think that the key finding here is that survival is possible after patients with COVID-19 suffer an in-hospital cardiac arrest,” Dr. Mitchell said. “We hope that the information from our study will be of use to frontline providers who are treating patients with COVID-19.”
“In coming weeks, there will likely be increased hospital strain and enormous challenges to providing COVID-19 care,” added Benjamin S. Abella, MD, the senior author of the in-hospital study. Dr. Abella is also from the University of Pennsylvania and was cochair of the Resuscitation Science symposium during the AHA meeting.
“It is crucial that hospital leaders prepare now for how they will manage COVID-19 resuscitation efforts,” Dr. Abella said. “Emergency medicine and critical care leaders must be mindful that many COVID-19 patients with arrest could survive to return to their families.”
“It is important to note both studies demonstrated variations in outcome and that those differences were associated with the differential COVID prevalence and mortality,” session comoderator Cindy H. Hsu, MD, PhD, University of Michigan, said in an interview.
“Future studies,” she said, “should address knowledge gaps including associated comorbidities and affected resuscitation process variables during the COVID-19 pandemic.”
Out-of-hospital cardiac arrest, March 2019 vs. March 2020
Compared with 2019, in 2020, the reported rates of return of spontaneous circulation after out-of-hospital cardiac arrest fell from 25% to 10.6% in New York and from 13.5% to 5.0% in northern Italy – two areas that were severely affected, Dr. Chan noted.
In this study, the researchers aimed to examine whether out-of-hospital cardiac arrest outcomes would be similar throughout the United States, including areas that were less severely affected, in the first weeks of the pandemic.
They linked data from the Cardiac Arrest Registry to Enhance Survival (CARES), which covers an area with about 152 million U.S. residents, with COVID-19 disease mortality data.
There were 9,863 out-of-hospital arrests from March 16 to April 30, 2020, compared with 9,440 cases during this time in 2019.
The patients in both years had a similar age (mean, 62 years) and sex (62% male), but there were more Black patients in 2020 (28% vs. 23%).
Overall, in communities with low to high rates of death from COVID-19, the rate of return of spontaneous circulation was 18% lower in that early pandemic period than in the same time in the previous year (23% vs. 29.8%; adjusted rate ratio, 0.82).
The rates of return of spontaneous circulation were also lower in communities with a low rate of COVID-19 mortality, but to a lesser extent (11%-15% lower in 2020 vs. 2019).
In the subset of emergency medical agencies with complete data on hospital survival, overall rates of survival to discharge were 17% lower during the studied pandemic period versus the same time a year earlier (6.6% vs. 9.8%; adjusted RR, 0.83).
This drop in survival was greater in communities with moderate to high COVID-19 mortality.
These outcomes were not explained by differences in emergency medical services arrival or treatment times, rates of bystander CPR, or initial out-of-hospital cardiac arrest rhythm.
Dr. Chan was a coauthor of an interim guidance issued April 9, 2020, by the AHA and several other medical societies for ways to protect frontline workers from contracting COVID-19 while they were performing CPR.
Communities that were not heavily affected by COVID-19 could have also been following the recommendations, which might have affected outcomes, he speculated.
For example, “when we pause chest compressions it can potentially worsen survival even if it’s for a short period of time. That might explain the lower rates of return of circulation.”
“That guidance was really meant for heavily affected communities,” Dr. Chan added. “Of course, as we speak, the pandemic is pretty much everywhere in the United States. It’s not just in the northeast; it’s not just in Arizona, Florida, California, Texas like it was in the summer. You are seeing surges in 46 of the 50 states.
“If your community is heavily affected by COVID-19 in terms of deaths at this time, paramedics will need to take caution to also help protect themselves, and the guidance may apply at that point,” he said.
In-hospital cardiac arrest, March Through May 2020
The early studies of in-hospital cardiac arrest in patients with COVID-19 showed “concerningly low rates” of return of spontaneous circulation and survival, said Dr. Mitchell.
“The first was a study from Wuhan, which demonstrated a 2.9% 30-day survival and the second was a small cohort from NYC with 0% survival to hospital discharge,” he said. “This raised concerns that offering CPR to patients who had a cardiac arrest from COVID-19 might only hold a low probability of success.”
To investigate this, the researchers formed a COVID study group comprising two hospitals in New York and nine hospitals in the Northeast and West Coast.
They identified 260 hospitalized adult patients with COVID-19 who had in-hospital cardiac arrest between March 1 and May 31, 2020. The patients had a median age of 69 years, and 72% were male. Most had preexisting comorbidities. Most of the cardiac arrests were in the ICU (64%), and almost all were witnessed (91%).
Return of spontaneous circulation occurred in 22% of the patients, and 12% had survived 30 days later. Of the 260 cardiac arrests, most (204) occurred in the New York hospitals.
There was a huge variation in outcomes. The rate of sustained return of spontaneous circulation was much lower in the two hospitals in New York compared with elsewhere (11% vs. 64%), as was 30-day survival (6% vs. 36%).
“Variation in outcomes from [in-hospital cardiac arrest] has been well described prior to the COVID-19 pandemic,” said Dr. Mitchell, “and is felt to be due to a range of factors, including variation in detection and prevention of cardiac arrest, management of patients during the cardiac arrest, and differences in postarrest care – including targeted temperature management and neuroprognostication.”
“We hypothesize that the strains of the COVID-19 pandemic may have amplified these variations (although we were unable to compare hospital performance before and after the pandemic),” he said.
Nevertheless, “in contrast to [earlier] studies, we have found that survival with a good neurological status is possible after in-hospital cardiac arrest in patients with COVID-19, which is certainly reassuring for those of us on the front line.”
Dr. Chan has received research support from the American Heart Association (which helps fund CARES); the National Heart, Lung, and Blood Institute; and Optum Rx. Dr. Abella has received honoraria from NeuroproteXeon, Becton Dickinson, and Physio-Control, and research grants from Medtronic, PCORI, Physio-Control, Stryker, and TerSera. Dr. Mitchell has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In the early weeks of the COVID-19 pandemic in the United States, rates of sustained return of spontaneous circulation after out-of-hospital cardiac arrest were lower throughout the country, compared with a year earlier, in one study.
A second study of that period showed that patients with COVID-19 had rates that were better than previously reported of surviving in-hospital cardiac arrest.
Paul S. Chan, MD, presented the out-of-hospital cardiac arrest research, and Oscar J. Mitchell, MD, presented the in-hospital cardiac arrest findings in a late-breaking resuscitation science session at the American Heart Association scientific sessions. The former study was also simultaneously published online Nov. 14 in JAMA Cardiology.
Importantly, “the survival rates were not zero in either setting,” said Dr. Chan, commenting on the implications of both studies taken together.
“The survival rates – either return of circulation or survival to discharge – were not futile,” Dr. Chan, from Saint Luke’s Mid America Heart Institute, Kansas City, Missouri, said in an interview.
“And I think that’s an overall important message – that we can’t write off patients who have a cardiac arrest at this point,” he stressed. “They deserve a response. Although the outcomes might not be as good as we had seen in years prior, we are seeing patients making it out of the hospital and surviving.”
Dr. Mitchell, from the University of Pennsylvania in Philadelphia, echoed this message in an interview.
“I think that the key finding here is that survival is possible after patients with COVID-19 suffer an in-hospital cardiac arrest,” Dr. Mitchell said. “We hope that the information from our study will be of use to frontline providers who are treating patients with COVID-19.”
“In coming weeks, there will likely be increased hospital strain and enormous challenges to providing COVID-19 care,” added Benjamin S. Abella, MD, the senior author of the in-hospital study. Dr. Abella is also from the University of Pennsylvania and was cochair of the Resuscitation Science symposium during the AHA meeting.
“It is crucial that hospital leaders prepare now for how they will manage COVID-19 resuscitation efforts,” Dr. Abella said. “Emergency medicine and critical care leaders must be mindful that many COVID-19 patients with arrest could survive to return to their families.”
“It is important to note both studies demonstrated variations in outcome and that those differences were associated with the differential COVID prevalence and mortality,” session comoderator Cindy H. Hsu, MD, PhD, University of Michigan, said in an interview.
“Future studies,” she said, “should address knowledge gaps including associated comorbidities and affected resuscitation process variables during the COVID-19 pandemic.”
Out-of-hospital cardiac arrest, March 2019 vs. March 2020
Compared with 2019, in 2020, the reported rates of return of spontaneous circulation after out-of-hospital cardiac arrest fell from 25% to 10.6% in New York and from 13.5% to 5.0% in northern Italy – two areas that were severely affected, Dr. Chan noted.
In this study, the researchers aimed to examine whether out-of-hospital cardiac arrest outcomes would be similar throughout the United States, including areas that were less severely affected, in the first weeks of the pandemic.
They linked data from the Cardiac Arrest Registry to Enhance Survival (CARES), which covers an area with about 152 million U.S. residents, with COVID-19 disease mortality data.
There were 9,863 out-of-hospital arrests from March 16 to April 30, 2020, compared with 9,440 cases during this time in 2019.
The patients in both years had a similar age (mean, 62 years) and sex (62% male), but there were more Black patients in 2020 (28% vs. 23%).
Overall, in communities with low to high rates of death from COVID-19, the rate of return of spontaneous circulation was 18% lower in that early pandemic period than in the same time in the previous year (23% vs. 29.8%; adjusted rate ratio, 0.82).
The rates of return of spontaneous circulation were also lower in communities with a low rate of COVID-19 mortality, but to a lesser extent (11%-15% lower in 2020 vs. 2019).
In the subset of emergency medical agencies with complete data on hospital survival, overall rates of survival to discharge were 17% lower during the studied pandemic period versus the same time a year earlier (6.6% vs. 9.8%; adjusted RR, 0.83).
This drop in survival was greater in communities with moderate to high COVID-19 mortality.
These outcomes were not explained by differences in emergency medical services arrival or treatment times, rates of bystander CPR, or initial out-of-hospital cardiac arrest rhythm.
Dr. Chan was a coauthor of an interim guidance issued April 9, 2020, by the AHA and several other medical societies for ways to protect frontline workers from contracting COVID-19 while they were performing CPR.
Communities that were not heavily affected by COVID-19 could have also been following the recommendations, which might have affected outcomes, he speculated.
For example, “when we pause chest compressions it can potentially worsen survival even if it’s for a short period of time. That might explain the lower rates of return of circulation.”
“That guidance was really meant for heavily affected communities,” Dr. Chan added. “Of course, as we speak, the pandemic is pretty much everywhere in the United States. It’s not just in the northeast; it’s not just in Arizona, Florida, California, Texas like it was in the summer. You are seeing surges in 46 of the 50 states.
“If your community is heavily affected by COVID-19 in terms of deaths at this time, paramedics will need to take caution to also help protect themselves, and the guidance may apply at that point,” he said.
In-hospital cardiac arrest, March Through May 2020
The early studies of in-hospital cardiac arrest in patients with COVID-19 showed “concerningly low rates” of return of spontaneous circulation and survival, said Dr. Mitchell.
“The first was a study from Wuhan, which demonstrated a 2.9% 30-day survival and the second was a small cohort from NYC with 0% survival to hospital discharge,” he said. “This raised concerns that offering CPR to patients who had a cardiac arrest from COVID-19 might only hold a low probability of success.”
To investigate this, the researchers formed a COVID study group comprising two hospitals in New York and nine hospitals in the Northeast and West Coast.
They identified 260 hospitalized adult patients with COVID-19 who had in-hospital cardiac arrest between March 1 and May 31, 2020. The patients had a median age of 69 years, and 72% were male. Most had preexisting comorbidities. Most of the cardiac arrests were in the ICU (64%), and almost all were witnessed (91%).
Return of spontaneous circulation occurred in 22% of the patients, and 12% had survived 30 days later. Of the 260 cardiac arrests, most (204) occurred in the New York hospitals.
There was a huge variation in outcomes. The rate of sustained return of spontaneous circulation was much lower in the two hospitals in New York compared with elsewhere (11% vs. 64%), as was 30-day survival (6% vs. 36%).
“Variation in outcomes from [in-hospital cardiac arrest] has been well described prior to the COVID-19 pandemic,” said Dr. Mitchell, “and is felt to be due to a range of factors, including variation in detection and prevention of cardiac arrest, management of patients during the cardiac arrest, and differences in postarrest care – including targeted temperature management and neuroprognostication.”
“We hypothesize that the strains of the COVID-19 pandemic may have amplified these variations (although we were unable to compare hospital performance before and after the pandemic),” he said.
Nevertheless, “in contrast to [earlier] studies, we have found that survival with a good neurological status is possible after in-hospital cardiac arrest in patients with COVID-19, which is certainly reassuring for those of us on the front line.”
Dr. Chan has received research support from the American Heart Association (which helps fund CARES); the National Heart, Lung, and Blood Institute; and Optum Rx. Dr. Abella has received honoraria from NeuroproteXeon, Becton Dickinson, and Physio-Control, and research grants from Medtronic, PCORI, Physio-Control, Stryker, and TerSera. Dr. Mitchell has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM AHA 2020
'Tragic' milestone: 1 million children with COVID-19
The number of new cases soared in the past week as the United States exceeded 1 million children infected with the coronavirus, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

For the first time, the number of cases in children for the week ending Nov. 12 passed 100,000, and it didn’t stop until it reached 111,946, bringing the total for the pandemic to 1,039,464 reported cases in 49 states (New York is not reporting ages), the District of Columbia, New York City, and Guam, the AAP and the CHA said in their weekly COVID-19 update.
“As a pediatrician who has practiced medicine for over 3 decades, I find this number staggering and tragic. We haven’t seen a virus flash through our communities in this way since before we had vaccines for measles and polio,” AAP President Sally Goza, MD, said in a written statement.
The previous 1-week high of almost 74,000 cases came just last week, and that number had surpassed the previous week’s new high of 61,000. The number of cumulative child cases, meanwhile, has doubled since Sept. 3, when it was just over 513,000. Children now represent 11.5% of all COVID-19 cases since the start of the pandemic in the jurisdictions reporting age distribution, the AAP and CHA said.
For the week ending Nov. 12, COVID-19 cases children made up 14% of cases nationally, rising from 13% the week before and reversing a decline that started in mid-October, the AAP/CHA data show.
The two groups continue to note the rarity of severe illness in children, but the number of deaths nationally had its biggest 1-week increase since late July, as the total rose from 123 to 133 in the 42 states reporting such data by age, as well as New York City. The cumulative hospitalization rate for children decreased slightly in the past week and is now down to 1.6% in the 23 states (and NYC) with available data, the AAP and CHA said.
The AAP called on elected leaders to enact a national strategy to combat the spread of the virus and urged health authorities to do more to collect data on longer-term impacts on children.
We’re very concerned about how this will impact all children, including toddlers who are missing key educational opportunities, as well as adolescents who may be at higher risk for anxiety and depression,” Dr. Goza said.
The number of new cases soared in the past week as the United States exceeded 1 million children infected with the coronavirus, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

For the first time, the number of cases in children for the week ending Nov. 12 passed 100,000, and it didn’t stop until it reached 111,946, bringing the total for the pandemic to 1,039,464 reported cases in 49 states (New York is not reporting ages), the District of Columbia, New York City, and Guam, the AAP and the CHA said in their weekly COVID-19 update.
“As a pediatrician who has practiced medicine for over 3 decades, I find this number staggering and tragic. We haven’t seen a virus flash through our communities in this way since before we had vaccines for measles and polio,” AAP President Sally Goza, MD, said in a written statement.
The previous 1-week high of almost 74,000 cases came just last week, and that number had surpassed the previous week’s new high of 61,000. The number of cumulative child cases, meanwhile, has doubled since Sept. 3, when it was just over 513,000. Children now represent 11.5% of all COVID-19 cases since the start of the pandemic in the jurisdictions reporting age distribution, the AAP and CHA said.
For the week ending Nov. 12, COVID-19 cases children made up 14% of cases nationally, rising from 13% the week before and reversing a decline that started in mid-October, the AAP/CHA data show.
The two groups continue to note the rarity of severe illness in children, but the number of deaths nationally had its biggest 1-week increase since late July, as the total rose from 123 to 133 in the 42 states reporting such data by age, as well as New York City. The cumulative hospitalization rate for children decreased slightly in the past week and is now down to 1.6% in the 23 states (and NYC) with available data, the AAP and CHA said.
The AAP called on elected leaders to enact a national strategy to combat the spread of the virus and urged health authorities to do more to collect data on longer-term impacts on children.
We’re very concerned about how this will impact all children, including toddlers who are missing key educational opportunities, as well as adolescents who may be at higher risk for anxiety and depression,” Dr. Goza said.
The number of new cases soared in the past week as the United States exceeded 1 million children infected with the coronavirus, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

For the first time, the number of cases in children for the week ending Nov. 12 passed 100,000, and it didn’t stop until it reached 111,946, bringing the total for the pandemic to 1,039,464 reported cases in 49 states (New York is not reporting ages), the District of Columbia, New York City, and Guam, the AAP and the CHA said in their weekly COVID-19 update.
“As a pediatrician who has practiced medicine for over 3 decades, I find this number staggering and tragic. We haven’t seen a virus flash through our communities in this way since before we had vaccines for measles and polio,” AAP President Sally Goza, MD, said in a written statement.
The previous 1-week high of almost 74,000 cases came just last week, and that number had surpassed the previous week’s new high of 61,000. The number of cumulative child cases, meanwhile, has doubled since Sept. 3, when it was just over 513,000. Children now represent 11.5% of all COVID-19 cases since the start of the pandemic in the jurisdictions reporting age distribution, the AAP and CHA said.
For the week ending Nov. 12, COVID-19 cases children made up 14% of cases nationally, rising from 13% the week before and reversing a decline that started in mid-October, the AAP/CHA data show.
The two groups continue to note the rarity of severe illness in children, but the number of deaths nationally had its biggest 1-week increase since late July, as the total rose from 123 to 133 in the 42 states reporting such data by age, as well as New York City. The cumulative hospitalization rate for children decreased slightly in the past week and is now down to 1.6% in the 23 states (and NYC) with available data, the AAP and CHA said.
The AAP called on elected leaders to enact a national strategy to combat the spread of the virus and urged health authorities to do more to collect data on longer-term impacts on children.
We’re very concerned about how this will impact all children, including toddlers who are missing key educational opportunities, as well as adolescents who may be at higher risk for anxiety and depression,” Dr. Goza said.
New-onset AFib common but unrecognized in the month after cardiac surgery
One in five patients at elevated stroke risk who underwent cardiac surgery with no history of atrial fibrillation preoperatively or at discharge developed postoperative AFib documented on a continuous cardiac rhythm monitoring device within the first 30 days after leaving the hospital in the randomized SEARCH-AF trial.
“Postoperative atrial fibrillation after cardiac surgery is not confined to the hospitalization period per se. We believe that these data should help inform on clinical practice guidelines on monitoring for postoperative atrial fibrillation in such patients,” said Subodh Verma, MD, PhD, reporting the results at the virtual American Heart Association scientific sessions.
“Guidelines provide little or no direction on optimal monitoring post cardiac surgery, particularly if patients are in sinus rhythm at discharge,” the surgeon noted.
SEARCH-AF was an open-label, multicenter study that included 336 patients at elevated stroke risk with an average CHA2DS2-VASc score of 4, no history of preoperative AFib, and none more than briefly with resolution during hospitalization. They were randomized to 30 days of postdischarge continuous cardiac rhythm monitoring with Medtronic’s SEEQ device, to Icentia’s CardioSTAT device, or to usual care, with Holter monitoring at the discretion of the treating physicians.
The primary result was a cumulative duration of AFib or atrial flutter of 6 minutes or longer during that 30-day period. This outcome occurred in 19.6% of the enhanced cardiac monitoring group and 1.7% of usual-care controls. Thus, there is an ongoing persistent occult risk of AFib that typically goes unrecognized. This 10-fold difference in the incidence of postoperative AFib translated into an absolute 17.9% between-group difference and a number-needed-to-treat of 6.
The secondary outcome of a cumulative atrial fib/flutter burden of 6 hours or more during 30 days occurred in 8.6% of the continuously monitored group and none of the controls. A cumulative AFib/flutter burden of 24 hours or greater occurred in 3.1% of the enhanced cardiac monitoring group and zero controls. These are AFib burdens that in other studies have been linked to increased risks of stroke and death, said Dr. Verma, professor of cardiovascular surgery at the University of Toronto.
“From a clinical standpoint, what this trial tells me is for my patients being discharged home tomorrow from the hospital, where they haven’t had AFib and I haven’t initiated anticoagulation, I have a low threshold to monitor these patients and to watch for periods of sustained unrecognized atrial fibrillation,” the surgeon added.
Experts: Results won’t change guidelines
Discussant Ben Freedman, MBBS, PhD, noted that the U.S. Preventive Services Task Force has stated that there are insufficient data available to recommend ECG screening for AFib to prevent stroke. Before the task force can be convinced to recommend it and for payers to cover it, a number of key questions need to be answered. And the SEARCH-AF trial doesn’t provide those answers, said Dr. Freedman, professor of cardiology and deputy director of the Heart Research Institute at the University of Sydney.
First off, it’ll be necessary to know if the risk posed by screen-detected AFib, including postoperative AFib, is similar to that of clinical AFib. Next, it must be shown that this screen-detected postoperative AFib is actionable; that is, that a screening strategy to detect postoperative AFib arising after discharge and then treat with oral anticoagulants will actually prevent more strokes than with usual care. There are large studies underway addressing that question, including HEARTLINE, STROKESTOP, and SAFERGUARD-AF, he observed.
In an interview, Rod S. Passman, MD, who gave a state-of-the-art talk on AFib detection at the meeting and wasn’t involved in SEARCH-AF, said he doesn’t consider the results practice-changing.
“It’s not guideline-changing because you’ve only shown that more intensive monitoring finds more AFib. Guideline-changing would be that finding that AFib and doing something about it impacts hard outcomes, and we don’t have that data yet,” said Dr. Passman, an electrophysiologist who is director of the Center for Arrhythmia Research and professor of medicine and preventive medicine at Northwestern University, Chicago.
The SEARCH-AF trial was funded by the Heart and Stroke Foundation of Canada, Bristol Myers Squibb, Pfizer, and Boehringer Ingelheim. Dr. Verma reported having received speaker’s fees and/or research support from those and other pharmaceutical companies. Dr. Freedman disclosed having no financial conflicts.
One in five patients at elevated stroke risk who underwent cardiac surgery with no history of atrial fibrillation preoperatively or at discharge developed postoperative AFib documented on a continuous cardiac rhythm monitoring device within the first 30 days after leaving the hospital in the randomized SEARCH-AF trial.
“Postoperative atrial fibrillation after cardiac surgery is not confined to the hospitalization period per se. We believe that these data should help inform on clinical practice guidelines on monitoring for postoperative atrial fibrillation in such patients,” said Subodh Verma, MD, PhD, reporting the results at the virtual American Heart Association scientific sessions.
“Guidelines provide little or no direction on optimal monitoring post cardiac surgery, particularly if patients are in sinus rhythm at discharge,” the surgeon noted.
SEARCH-AF was an open-label, multicenter study that included 336 patients at elevated stroke risk with an average CHA2DS2-VASc score of 4, no history of preoperative AFib, and none more than briefly with resolution during hospitalization. They were randomized to 30 days of postdischarge continuous cardiac rhythm monitoring with Medtronic’s SEEQ device, to Icentia’s CardioSTAT device, or to usual care, with Holter monitoring at the discretion of the treating physicians.
The primary result was a cumulative duration of AFib or atrial flutter of 6 minutes or longer during that 30-day period. This outcome occurred in 19.6% of the enhanced cardiac monitoring group and 1.7% of usual-care controls. Thus, there is an ongoing persistent occult risk of AFib that typically goes unrecognized. This 10-fold difference in the incidence of postoperative AFib translated into an absolute 17.9% between-group difference and a number-needed-to-treat of 6.
The secondary outcome of a cumulative atrial fib/flutter burden of 6 hours or more during 30 days occurred in 8.6% of the continuously monitored group and none of the controls. A cumulative AFib/flutter burden of 24 hours or greater occurred in 3.1% of the enhanced cardiac monitoring group and zero controls. These are AFib burdens that in other studies have been linked to increased risks of stroke and death, said Dr. Verma, professor of cardiovascular surgery at the University of Toronto.
“From a clinical standpoint, what this trial tells me is for my patients being discharged home tomorrow from the hospital, where they haven’t had AFib and I haven’t initiated anticoagulation, I have a low threshold to monitor these patients and to watch for periods of sustained unrecognized atrial fibrillation,” the surgeon added.
Experts: Results won’t change guidelines
Discussant Ben Freedman, MBBS, PhD, noted that the U.S. Preventive Services Task Force has stated that there are insufficient data available to recommend ECG screening for AFib to prevent stroke. Before the task force can be convinced to recommend it and for payers to cover it, a number of key questions need to be answered. And the SEARCH-AF trial doesn’t provide those answers, said Dr. Freedman, professor of cardiology and deputy director of the Heart Research Institute at the University of Sydney.
First off, it’ll be necessary to know if the risk posed by screen-detected AFib, including postoperative AFib, is similar to that of clinical AFib. Next, it must be shown that this screen-detected postoperative AFib is actionable; that is, that a screening strategy to detect postoperative AFib arising after discharge and then treat with oral anticoagulants will actually prevent more strokes than with usual care. There are large studies underway addressing that question, including HEARTLINE, STROKESTOP, and SAFERGUARD-AF, he observed.
In an interview, Rod S. Passman, MD, who gave a state-of-the-art talk on AFib detection at the meeting and wasn’t involved in SEARCH-AF, said he doesn’t consider the results practice-changing.
“It’s not guideline-changing because you’ve only shown that more intensive monitoring finds more AFib. Guideline-changing would be that finding that AFib and doing something about it impacts hard outcomes, and we don’t have that data yet,” said Dr. Passman, an electrophysiologist who is director of the Center for Arrhythmia Research and professor of medicine and preventive medicine at Northwestern University, Chicago.
The SEARCH-AF trial was funded by the Heart and Stroke Foundation of Canada, Bristol Myers Squibb, Pfizer, and Boehringer Ingelheim. Dr. Verma reported having received speaker’s fees and/or research support from those and other pharmaceutical companies. Dr. Freedman disclosed having no financial conflicts.
One in five patients at elevated stroke risk who underwent cardiac surgery with no history of atrial fibrillation preoperatively or at discharge developed postoperative AFib documented on a continuous cardiac rhythm monitoring device within the first 30 days after leaving the hospital in the randomized SEARCH-AF trial.
“Postoperative atrial fibrillation after cardiac surgery is not confined to the hospitalization period per se. We believe that these data should help inform on clinical practice guidelines on monitoring for postoperative atrial fibrillation in such patients,” said Subodh Verma, MD, PhD, reporting the results at the virtual American Heart Association scientific sessions.
“Guidelines provide little or no direction on optimal monitoring post cardiac surgery, particularly if patients are in sinus rhythm at discharge,” the surgeon noted.
SEARCH-AF was an open-label, multicenter study that included 336 patients at elevated stroke risk with an average CHA2DS2-VASc score of 4, no history of preoperative AFib, and none more than briefly with resolution during hospitalization. They were randomized to 30 days of postdischarge continuous cardiac rhythm monitoring with Medtronic’s SEEQ device, to Icentia’s CardioSTAT device, or to usual care, with Holter monitoring at the discretion of the treating physicians.
The primary result was a cumulative duration of AFib or atrial flutter of 6 minutes or longer during that 30-day period. This outcome occurred in 19.6% of the enhanced cardiac monitoring group and 1.7% of usual-care controls. Thus, there is an ongoing persistent occult risk of AFib that typically goes unrecognized. This 10-fold difference in the incidence of postoperative AFib translated into an absolute 17.9% between-group difference and a number-needed-to-treat of 6.
The secondary outcome of a cumulative atrial fib/flutter burden of 6 hours or more during 30 days occurred in 8.6% of the continuously monitored group and none of the controls. A cumulative AFib/flutter burden of 24 hours or greater occurred in 3.1% of the enhanced cardiac monitoring group and zero controls. These are AFib burdens that in other studies have been linked to increased risks of stroke and death, said Dr. Verma, professor of cardiovascular surgery at the University of Toronto.
“From a clinical standpoint, what this trial tells me is for my patients being discharged home tomorrow from the hospital, where they haven’t had AFib and I haven’t initiated anticoagulation, I have a low threshold to monitor these patients and to watch for periods of sustained unrecognized atrial fibrillation,” the surgeon added.
Experts: Results won’t change guidelines
Discussant Ben Freedman, MBBS, PhD, noted that the U.S. Preventive Services Task Force has stated that there are insufficient data available to recommend ECG screening for AFib to prevent stroke. Before the task force can be convinced to recommend it and for payers to cover it, a number of key questions need to be answered. And the SEARCH-AF trial doesn’t provide those answers, said Dr. Freedman, professor of cardiology and deputy director of the Heart Research Institute at the University of Sydney.
First off, it’ll be necessary to know if the risk posed by screen-detected AFib, including postoperative AFib, is similar to that of clinical AFib. Next, it must be shown that this screen-detected postoperative AFib is actionable; that is, that a screening strategy to detect postoperative AFib arising after discharge and then treat with oral anticoagulants will actually prevent more strokes than with usual care. There are large studies underway addressing that question, including HEARTLINE, STROKESTOP, and SAFERGUARD-AF, he observed.
In an interview, Rod S. Passman, MD, who gave a state-of-the-art talk on AFib detection at the meeting and wasn’t involved in SEARCH-AF, said he doesn’t consider the results practice-changing.
“It’s not guideline-changing because you’ve only shown that more intensive monitoring finds more AFib. Guideline-changing would be that finding that AFib and doing something about it impacts hard outcomes, and we don’t have that data yet,” said Dr. Passman, an electrophysiologist who is director of the Center for Arrhythmia Research and professor of medicine and preventive medicine at Northwestern University, Chicago.
The SEARCH-AF trial was funded by the Heart and Stroke Foundation of Canada, Bristol Myers Squibb, Pfizer, and Boehringer Ingelheim. Dr. Verma reported having received speaker’s fees and/or research support from those and other pharmaceutical companies. Dr. Freedman disclosed having no financial conflicts.
FROM AHA 2020
FDA-approved peanut immunotherapy protocol comes with a cost
Peanut allergy immunotherapy now comes with approval from the US Food and Drug Administration (FDA), but it also comes with protocols, standards, and paperwork. Whether it will be widely adopted has yet to be determined.
A few dozen allergists around the world have been offering food allergy immunotherapy for many years, having developed their own measuring techniques using store-bought food.
But the vast majority of allergists are not interested in developing home-grown treatments, not only because it involves research and development, but also because it comes with legal risks.
“Finally we have another treatment option,” said Edwin Kim, MD, from the UNC Allergy and Immunology Clinic in Chapel Hill, N.C. “This is what we were waiting for. It’s not cowboy stuff; this works.”
In January, the FDA approved peanut allergen powder (Palforzia) for patients 4-17 years of age, as reported by Medscape Medical News.
The pill contains measured doses of peanut flour and comes with a protocol that will allow allergists to bring patients to a peanut tolerance of 300 mg (about one peanut) and a black-box warning about anaphylaxis risk.
And before allergists can prescribe it, they must take a Risk Evaluation and Mitigation Strategy course to learn about dosing and the allergic reaction protocol.
“That may scare some away,” said Dr. Kim, who discussed the FDA-approved option during his presentation at the American College of Allergy, Asthma & Immunology 2020 Annual Scientific Meeting.
Allergic reaction, including the potential for anaphylaxis, has always been an issue with immunotherapy.
“People make the argument that there is a difference” between an expected allergic reaction – such as one that occurs after the administration of immunotherapy – and an unexpected reaction, he said. Because an expected reaction can be treated quickly, “some feel these expected reactions don’t matter so much.”
“Others say a reaction is a reaction” and argue that if, a treatment causes reaction, then it doesn’t make sense, he explained.
It comes down to patients – they must be willing to take a risk to develop tolerance and improve their quality of life – and the allergists willing to treat them.
The peanut powder involves paperwork, preauthorization forms, denials of care, a higher price tag, regimented procedures, and a prerequisite number of visits with patients. “Not everyone will want to do this,” said Dr. Kim.
The regimen involves three phases. During initial dose escalation, five doses are administered in the office on day 1. Then, over the next 6 months, updoses are administered during 11 in-office sessions and a 300-mg tolerance is achieved. Finally, to maintain tolerance to one peanut, daily doses are administered at home.
The drug cost alone is about $4,200 a year, according to Institute for Clinical and Economic Review. Peanut flour from the grocery store is cheaper, but comes with the risk of bacteria or other contamination.
“This product offers some reassurance, and that matters,” Dr. Kim said.
It’s good to have more options for food allergy treatment. “We need a more proactive way to treat food allergy; avoidance is not good enough,” he explained. “And presumably, at some point, the patient will be able to eat a grocery-store peanut instead of buying the pills.”
The art of medicine
But not all allergists will be able to make the protocol work. And it’s not clear whether there is room to alter treatment and offer patients with a higher tolerance a higher starting dose. What we do know, though, is that “the product leaves little room for ‘the art of medicine,’ ” Kim said.
That art is practiced by Arnon Elizur, MD, from the Shamir Medical Center in Tzrifin, Israel, but it’s backed by a rigid home-grown protocol.
Since 2010, he has treated 1,800 patients for peanut allergy, updosing slowly to a tolerance of 3,000 mg of peanut, the equivalent of 10 peanuts. He keeps the maintenance dose at four peanuts (1,200 mg). His center takes a personalized approach, starting patients on the highest dose they can tolerate and working up, with daily patient check-ins from home and a staff available around the clock to answer questions and deal with reactions.
“We aim for full sensitization,” Dr. Elizur said in an interview.
The peanut pill is “a big step forward” for immunotherapy, he said. It is “a standardized product, checked for bacteria and allergen content, which is available to a wide community of physicians.”
But, he pointed out, “it’s expensive.” And it’s only for peanut. “There are millions of food-allergic patients around the world dying from adverse reactions to many different kinds of food. We don’t want to wait for years for a product for all of them. We can use the actual food.”
He questions the lifelong maintenance protocol with a daily 300-mg pill. “If you can’t eat a peanut, why would you buy a drug that’s a peanut?” he asked.
He also said he’s disappointed that the product is not indicated for adults.
At the Shamir clinic, reactions are closely monitored. “Some are mild, others we treat with autoinjectors, epinephrine,” he reported. “Those are the most undesirable.”
Data from his center show that reactions occur in about 15% of patients. But his treatment success rates are good. In an average of 8 months, he is able to get 80% of his adult patients to full sensitization.
But it’s not for all patients or for all clinics, he acknowledged. “We continue to look at this balance in quality of life throughout the process. Our goal is to improve the quality of life threshold.”
Treatment that involves “native food” is “a lot of work” and requires “a lot of investment,” Dr. Elizur said. His center uses a web reporting system to maintain a 24/7 dialogue with patients, “and we look at the reports every day.” They also have a physician on call at all times. “Not everyone can commit to providing care throughout the day and night.”
His center charges the equivalent of $US3,000 per food allergy treated. “That’s whether it takes 6 months or 2 years,” he said.
There are more than 1,000 people on his 3-year waiting list.
“This is the first year that the American College of Allergy, Asthma, and Immunology is not hosting a pro–con debate on oral immunotherapy,” Dr. Kim pointed out. “We have a therapy now.”
However, the pandemic has slowed treatment uptake. “Immunotherapy is not easy to do, whether it’s FDA approved or not,” he explained. With at least 11 doctor visits in the first 6 months – each visit is between 30 minutes and 2-3 hours long – it hasn’t been possible to set up this year. “It’s not ideal.”
It will be interesting to see “how this will roll out and how it will be adopted,” Dr. Kim said. “From a food allergy point of view, the next 12 months are going to be very interesting.”
Dr. Kim reports receiving consulting honorarium from Aimmune, the maker of Palforzia; being on the clinical medical advisory board for DBV Technologies; and consulting for Aimmune, Allakos, Allergenis, DBV, Duke Clinical Research Institute, Ukko Incorporated, Vibrant America, and Kenota Health. Dr. Elizur has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Peanut allergy immunotherapy now comes with approval from the US Food and Drug Administration (FDA), but it also comes with protocols, standards, and paperwork. Whether it will be widely adopted has yet to be determined.
A few dozen allergists around the world have been offering food allergy immunotherapy for many years, having developed their own measuring techniques using store-bought food.
But the vast majority of allergists are not interested in developing home-grown treatments, not only because it involves research and development, but also because it comes with legal risks.
“Finally we have another treatment option,” said Edwin Kim, MD, from the UNC Allergy and Immunology Clinic in Chapel Hill, N.C. “This is what we were waiting for. It’s not cowboy stuff; this works.”
In January, the FDA approved peanut allergen powder (Palforzia) for patients 4-17 years of age, as reported by Medscape Medical News.
The pill contains measured doses of peanut flour and comes with a protocol that will allow allergists to bring patients to a peanut tolerance of 300 mg (about one peanut) and a black-box warning about anaphylaxis risk.
And before allergists can prescribe it, they must take a Risk Evaluation and Mitigation Strategy course to learn about dosing and the allergic reaction protocol.
“That may scare some away,” said Dr. Kim, who discussed the FDA-approved option during his presentation at the American College of Allergy, Asthma & Immunology 2020 Annual Scientific Meeting.
Allergic reaction, including the potential for anaphylaxis, has always been an issue with immunotherapy.
“People make the argument that there is a difference” between an expected allergic reaction – such as one that occurs after the administration of immunotherapy – and an unexpected reaction, he said. Because an expected reaction can be treated quickly, “some feel these expected reactions don’t matter so much.”
“Others say a reaction is a reaction” and argue that if, a treatment causes reaction, then it doesn’t make sense, he explained.
It comes down to patients – they must be willing to take a risk to develop tolerance and improve their quality of life – and the allergists willing to treat them.
The peanut powder involves paperwork, preauthorization forms, denials of care, a higher price tag, regimented procedures, and a prerequisite number of visits with patients. “Not everyone will want to do this,” said Dr. Kim.
The regimen involves three phases. During initial dose escalation, five doses are administered in the office on day 1. Then, over the next 6 months, updoses are administered during 11 in-office sessions and a 300-mg tolerance is achieved. Finally, to maintain tolerance to one peanut, daily doses are administered at home.
The drug cost alone is about $4,200 a year, according to Institute for Clinical and Economic Review. Peanut flour from the grocery store is cheaper, but comes with the risk of bacteria or other contamination.
“This product offers some reassurance, and that matters,” Dr. Kim said.
It’s good to have more options for food allergy treatment. “We need a more proactive way to treat food allergy; avoidance is not good enough,” he explained. “And presumably, at some point, the patient will be able to eat a grocery-store peanut instead of buying the pills.”
The art of medicine
But not all allergists will be able to make the protocol work. And it’s not clear whether there is room to alter treatment and offer patients with a higher tolerance a higher starting dose. What we do know, though, is that “the product leaves little room for ‘the art of medicine,’ ” Kim said.
That art is practiced by Arnon Elizur, MD, from the Shamir Medical Center in Tzrifin, Israel, but it’s backed by a rigid home-grown protocol.
Since 2010, he has treated 1,800 patients for peanut allergy, updosing slowly to a tolerance of 3,000 mg of peanut, the equivalent of 10 peanuts. He keeps the maintenance dose at four peanuts (1,200 mg). His center takes a personalized approach, starting patients on the highest dose they can tolerate and working up, with daily patient check-ins from home and a staff available around the clock to answer questions and deal with reactions.
“We aim for full sensitization,” Dr. Elizur said in an interview.
The peanut pill is “a big step forward” for immunotherapy, he said. It is “a standardized product, checked for bacteria and allergen content, which is available to a wide community of physicians.”
But, he pointed out, “it’s expensive.” And it’s only for peanut. “There are millions of food-allergic patients around the world dying from adverse reactions to many different kinds of food. We don’t want to wait for years for a product for all of them. We can use the actual food.”
He questions the lifelong maintenance protocol with a daily 300-mg pill. “If you can’t eat a peanut, why would you buy a drug that’s a peanut?” he asked.
He also said he’s disappointed that the product is not indicated for adults.
At the Shamir clinic, reactions are closely monitored. “Some are mild, others we treat with autoinjectors, epinephrine,” he reported. “Those are the most undesirable.”
Data from his center show that reactions occur in about 15% of patients. But his treatment success rates are good. In an average of 8 months, he is able to get 80% of his adult patients to full sensitization.
But it’s not for all patients or for all clinics, he acknowledged. “We continue to look at this balance in quality of life throughout the process. Our goal is to improve the quality of life threshold.”
Treatment that involves “native food” is “a lot of work” and requires “a lot of investment,” Dr. Elizur said. His center uses a web reporting system to maintain a 24/7 dialogue with patients, “and we look at the reports every day.” They also have a physician on call at all times. “Not everyone can commit to providing care throughout the day and night.”
His center charges the equivalent of $US3,000 per food allergy treated. “That’s whether it takes 6 months or 2 years,” he said.
There are more than 1,000 people on his 3-year waiting list.
“This is the first year that the American College of Allergy, Asthma, and Immunology is not hosting a pro–con debate on oral immunotherapy,” Dr. Kim pointed out. “We have a therapy now.”
However, the pandemic has slowed treatment uptake. “Immunotherapy is not easy to do, whether it’s FDA approved or not,” he explained. With at least 11 doctor visits in the first 6 months – each visit is between 30 minutes and 2-3 hours long – it hasn’t been possible to set up this year. “It’s not ideal.”
It will be interesting to see “how this will roll out and how it will be adopted,” Dr. Kim said. “From a food allergy point of view, the next 12 months are going to be very interesting.”
Dr. Kim reports receiving consulting honorarium from Aimmune, the maker of Palforzia; being on the clinical medical advisory board for DBV Technologies; and consulting for Aimmune, Allakos, Allergenis, DBV, Duke Clinical Research Institute, Ukko Incorporated, Vibrant America, and Kenota Health. Dr. Elizur has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Peanut allergy immunotherapy now comes with approval from the US Food and Drug Administration (FDA), but it also comes with protocols, standards, and paperwork. Whether it will be widely adopted has yet to be determined.
A few dozen allergists around the world have been offering food allergy immunotherapy for many years, having developed their own measuring techniques using store-bought food.
But the vast majority of allergists are not interested in developing home-grown treatments, not only because it involves research and development, but also because it comes with legal risks.
“Finally we have another treatment option,” said Edwin Kim, MD, from the UNC Allergy and Immunology Clinic in Chapel Hill, N.C. “This is what we were waiting for. It’s not cowboy stuff; this works.”
In January, the FDA approved peanut allergen powder (Palforzia) for patients 4-17 years of age, as reported by Medscape Medical News.
The pill contains measured doses of peanut flour and comes with a protocol that will allow allergists to bring patients to a peanut tolerance of 300 mg (about one peanut) and a black-box warning about anaphylaxis risk.
And before allergists can prescribe it, they must take a Risk Evaluation and Mitigation Strategy course to learn about dosing and the allergic reaction protocol.
“That may scare some away,” said Dr. Kim, who discussed the FDA-approved option during his presentation at the American College of Allergy, Asthma & Immunology 2020 Annual Scientific Meeting.
Allergic reaction, including the potential for anaphylaxis, has always been an issue with immunotherapy.
“People make the argument that there is a difference” between an expected allergic reaction – such as one that occurs after the administration of immunotherapy – and an unexpected reaction, he said. Because an expected reaction can be treated quickly, “some feel these expected reactions don’t matter so much.”
“Others say a reaction is a reaction” and argue that if, a treatment causes reaction, then it doesn’t make sense, he explained.
It comes down to patients – they must be willing to take a risk to develop tolerance and improve their quality of life – and the allergists willing to treat them.
The peanut powder involves paperwork, preauthorization forms, denials of care, a higher price tag, regimented procedures, and a prerequisite number of visits with patients. “Not everyone will want to do this,” said Dr. Kim.
The regimen involves three phases. During initial dose escalation, five doses are administered in the office on day 1. Then, over the next 6 months, updoses are administered during 11 in-office sessions and a 300-mg tolerance is achieved. Finally, to maintain tolerance to one peanut, daily doses are administered at home.
The drug cost alone is about $4,200 a year, according to Institute for Clinical and Economic Review. Peanut flour from the grocery store is cheaper, but comes with the risk of bacteria or other contamination.
“This product offers some reassurance, and that matters,” Dr. Kim said.
It’s good to have more options for food allergy treatment. “We need a more proactive way to treat food allergy; avoidance is not good enough,” he explained. “And presumably, at some point, the patient will be able to eat a grocery-store peanut instead of buying the pills.”
The art of medicine
But not all allergists will be able to make the protocol work. And it’s not clear whether there is room to alter treatment and offer patients with a higher tolerance a higher starting dose. What we do know, though, is that “the product leaves little room for ‘the art of medicine,’ ” Kim said.
That art is practiced by Arnon Elizur, MD, from the Shamir Medical Center in Tzrifin, Israel, but it’s backed by a rigid home-grown protocol.
Since 2010, he has treated 1,800 patients for peanut allergy, updosing slowly to a tolerance of 3,000 mg of peanut, the equivalent of 10 peanuts. He keeps the maintenance dose at four peanuts (1,200 mg). His center takes a personalized approach, starting patients on the highest dose they can tolerate and working up, with daily patient check-ins from home and a staff available around the clock to answer questions and deal with reactions.
“We aim for full sensitization,” Dr. Elizur said in an interview.
The peanut pill is “a big step forward” for immunotherapy, he said. It is “a standardized product, checked for bacteria and allergen content, which is available to a wide community of physicians.”
But, he pointed out, “it’s expensive.” And it’s only for peanut. “There are millions of food-allergic patients around the world dying from adverse reactions to many different kinds of food. We don’t want to wait for years for a product for all of them. We can use the actual food.”
He questions the lifelong maintenance protocol with a daily 300-mg pill. “If you can’t eat a peanut, why would you buy a drug that’s a peanut?” he asked.
He also said he’s disappointed that the product is not indicated for adults.
At the Shamir clinic, reactions are closely monitored. “Some are mild, others we treat with autoinjectors, epinephrine,” he reported. “Those are the most undesirable.”
Data from his center show that reactions occur in about 15% of patients. But his treatment success rates are good. In an average of 8 months, he is able to get 80% of his adult patients to full sensitization.
But it’s not for all patients or for all clinics, he acknowledged. “We continue to look at this balance in quality of life throughout the process. Our goal is to improve the quality of life threshold.”
Treatment that involves “native food” is “a lot of work” and requires “a lot of investment,” Dr. Elizur said. His center uses a web reporting system to maintain a 24/7 dialogue with patients, “and we look at the reports every day.” They also have a physician on call at all times. “Not everyone can commit to providing care throughout the day and night.”
His center charges the equivalent of $US3,000 per food allergy treated. “That’s whether it takes 6 months or 2 years,” he said.
There are more than 1,000 people on his 3-year waiting list.
“This is the first year that the American College of Allergy, Asthma, and Immunology is not hosting a pro–con debate on oral immunotherapy,” Dr. Kim pointed out. “We have a therapy now.”
However, the pandemic has slowed treatment uptake. “Immunotherapy is not easy to do, whether it’s FDA approved or not,” he explained. With at least 11 doctor visits in the first 6 months – each visit is between 30 minutes and 2-3 hours long – it hasn’t been possible to set up this year. “It’s not ideal.”
It will be interesting to see “how this will roll out and how it will be adopted,” Dr. Kim said. “From a food allergy point of view, the next 12 months are going to be very interesting.”
Dr. Kim reports receiving consulting honorarium from Aimmune, the maker of Palforzia; being on the clinical medical advisory board for DBV Technologies; and consulting for Aimmune, Allakos, Allergenis, DBV, Duke Clinical Research Institute, Ukko Incorporated, Vibrant America, and Kenota Health. Dr. Elizur has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.