User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Moisturizers and skin barrier repair
There are dozens of skin care products that claim to repair the barrier that do not have the science or ingredient content to back them up.
Does a skin barrier repair moisturizer really repair?
First, let’s briefly review what the skin barrier is. The stratum corneum (SC), the most superficial layer of the epidermis, averages approximately 15-cell layers in thickness.1,2 The keratinocytes reside there in a pattern resembling a brick wall. The “mortar” is composed of the lipid contents extruded from the lamellar granules. This protective barrier functions to prevent transepidermal water loss (TEWL) and entry of allergens, irritants, and pathogens into deeper layers of the skin. This column will focus briefly on the structure and function of the skin barrier and the barrier repair technologies that use synthetic lipids such as myristoyl-palmitoyl and myristyl/palmityl-oxo-stearamide/arachamide MEA.
Structure of the skin barrier
SC keratinocytes are surrounded by lamella made from lipid bilayers. The lipids have hydrophilic heads and hydrophobic tails; the bilayer arises when the hydrophobic tails face the center and the hydrophilic heads face out of the bilayer. This formation yields a disc-shaped hydrophobic lamellar center. There are actually several of these lamellar layers between keratinocytes.
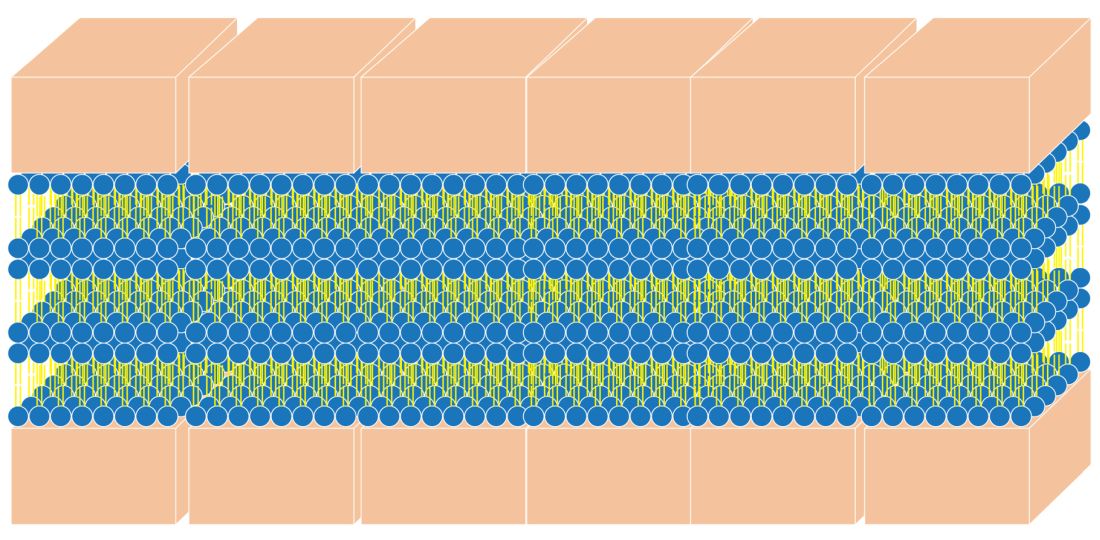
The naturally occurring primary lipids of the bilayer lamellae are made up of an equal ratio of ceramides, cholesterol, and free fatty acid. Arranged in a 1:1:1 ratio, they fit together like pieces of a puzzle to achieve skin barrier homeostasis. The shape and size of these puzzle pieces is critical. An incorrect shape results in a hole in the skin barrier resulting in dehydration, inflammation, and sensitivity.
Ceramides
Ceramides are a complex family of lipids (sphingolipids – a sphingoid base and a fatty acid) involved in cell, as well as barrier, homeostasis and water-holding capacity. In fact, they are known to play a crucial role in cell proliferation, differentiation, and apoptosis.3 There are at least 16 types of naturally occurring ceramides. For years, they have been included in barrier repair moisturizers. They are difficult to work with in moisturizers for several reasons:
- Ceramides are abundant in brain tissue and the ceramides used in moisturizers in the past were derived from bovine brain tissue. Prior to the emergence of bovine spongiform encephalopathy (mad cow disease), many ceramides in skin-care products were animal derived, which made them expensive and undesirable.
- Ceramides in skin care that are made from plant sources are referred to as phyto-derived ceramides. Although they share a similar structure with ceramides that occur in human skin, there are differences in chain length, hydroxylation pattern, and the degree of unsaturation that lead to structural diversity.4 The shape of ceramides is critical for a strong skin barrier because the lipids in the skin barrier must fit together like puzzle pieces to form a water-tight barrier. Natural sources of ceramides include rice, wheat, potato, konjac, and maize. Standardization of ceramide shape and structure makes using phyto-derived ceramides in skin care products challenging.
- Ceramides, because of their waxy consistency, require heat during the mixing process of skin care product manufacturing. This heat can make other ingredients inactive in the skin care formulation. (Ceramides are typically added early in the formulation process, and the heat-sensitive ones are added later.)
- Many forms of ceramides are unstable in the product manufacturing and bottling processes.
- Skin penetration of ceramides depends on the shape and size of ceramides.
Synthetic ceramides have been developed to make ceramides safe, affordable, and more easily formulated into moisturizers. These formulations synthesized in the lab are sometimes called pseudoceramides because they are structurally different compounds that mimic the activity of ceramides. They are developed to be less expensive to manufacture, safer than those derived from animals, and easier to formulate, and they can be made into the specific shape of the ceramide puzzle piece.
Ceramides in skin care
The naturally occurring intercellular lipids of the SC are composed of approximately equal proportions of ceramides, cholesterol, and fatty acids (referred to in this article as the “three barrier lipids” for simplicity).5-9 Alterations in any of these three barrier lipids or their regulatory enzymes result in impairments in the function of the epidermal barrier. Therefore, any synthetic ceramide must mimic the shape of natural ceramides, or the three barrier lipids in the moisturizer must mimic the shape of the entire bilayer lamella. Unfortunately, most barrier repair moisturizers do not meet these criteria and are not true barrier repair moisturizers.
How do you know if a moisturizer repairs the skin barrier?
Clinical tests such as measuring transepidermal water loss (TEWL) with a Tewameter are usually done to support the barrier repair claim. However, occlusive ingredients like oils can lower TEWL without affecting the barrier. In fact, we believe that sebum on the skin can make an impaired barrier and result in normal TEWL even when the barrier is impaired. So, just because a product improved TEWL does not necessarily mean that it repairs the barrier.
One way to test the ability of a moisturizer to repair the barrier is to look at a structural analysis of the moisturizer to see if it forms the requisite bilayer lamellar shape. An easy way to do this testing is to look for the cross pattern under a cross polarized microscope. The cross pattern is known as optical anisotropy. 8
The best barrier repair creams
Optimal barrier repair creams either feature a 1:1:1 ratio of epidermal lipids or form a cross structure when viewed with a cross-polarized microscope.8 There are several categories of barrier repair moisturizers that meet these criteria.
Barrier repair creams with a 1:1:1 ratio of lipids:
Peter Elias, MD, holds the patent on barrier repair moisturizer technology that has a 1:1:1 ratio. His well-established technology is used in a prescription barrier repair cream called EpiCeram® which is approved by the Food and Drug Administration to treat eczema. There are no other moisturizers that I know of that contain this 1:1:1 lipid ratio.
There is a barrier repair cream on the market that contains a 2:4:2 ratio of lipids based on a study that showed that this ratio is effective in older skin with an impaired barrier. It is unknown if this moisturizer forms a cross pattern.
Barrier repair creams that demonstrate a cross pattern:
Multilamellar emulsion (MLE) technology: This barrier repair technology, invented in South Korea, contains the synthetic pseudoceramide called myristyl/palmityl-oxo-stearamide/arachamide MEA (C34H67NO3/C36H71NO3/C38H75NO3), or the pseudoceramide myristoyl-palmitoyl-oxostearamide-arachamide MEA.
In a 2019 pilot study by Ye and colleagues, the investigators treated 33 older volunteers twice daily for 30 days with approximately 3 mL of an emollient containing MLE technology. In addition, 30 untreated older subjects and 11 young volunteers served as controls. The investigators found that the topically applied barrier repair emollient significantly improved barrier function, as well as stratum corneum hydration. Circulating levels of the important, age-related plasma cytokines interleukin-1 beta and IL-6 were found to have normalized, while tumor necrosis factor–alpha decreased markedly. The investigators suggested that repair of the skin barrier might diminish circulating proinflammatory cytokine levels (such as amyloid A) in aged humans, potentially mitigating the development of chronic inflammatory conditions.10
MLE technology has also been shown to improve childhood atopic dermatitis and prevent steroid atrophy.11,12 The consistent use of MLE technology in moisturizers has been shown to alleviate inflammatory factors in the blood and is believed to lessen systemic inflammation.10
Physiologic (PSL) lipid repair technology: This technology was invented by one of the South Korean researchers who helped develop MLE technology. It contains pseudoceramides, fatty acids, and cholesterol. The figure of the cross pattern above, as seen under the cross polarized microscope, is an image taken of this PSL lipid repair technology.
Conclusion
Do not believe that a moisturizer repairs the barrier just because it says so on the label. Three of the most popular body moisturizes used to treat eczema do not actually have the proper formula to repair the barrier. Unfortunately, there are dozens of skin care products that claim to repair the barrier that do not have the science or ingredient content to back them up. To restore the skin barrier to a healthy condition, it is imperative that the barrier repair moisturizers that you are recommending for patients have the correct 1:1:1 ratio of epidermal lipids or contain bilayer lamella that mimic the natural multilamellar layers and display the cross pattern under a cross-polarized microscope.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann has written two textbooks and a New York Times Best Sellers book for consumers. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Revance, Evolus, and Burt’s Bees. She is the CEO of Skin Type Solutions, a company that independently tests skin care products and makes recommendations to physicians on which skin care technologies are best. Write to her at [email protected].
References
1. Christophers E and Kligman AM. J Invest Dermatol. 1964;42:407-9.
2. Blair C. Br J Dermatol. 1968;80(7):430-6.
3. Morita O et al. Food Chem Toxicol. 2009 Apr;47(4):681-6.
4. Tessema E N et al. Skin pharmacology and physiology. 2017;30(3):115-38.
5. Coderch L et al. Am J Clin Dermatol. 2003;4(2):107-29.
6. Man MQ et al. Arch Dermatol. 1993;129(6):728-38.
7. Man MQ M et al. J Invest Dermatol. 1996 May;106(5):1096-101.
8. Park BD et al. J Invest Dermatol. 2003;121(4):794-801.
9. Proksch E and Jensen J. Skin as an organ of protection, in “Fitzpatrick’s Dermatology in General Medicine,” 7th ed. New York: McGraw-Hill, 2008, pp. 383-95.
10. Ye L et al. J Eur Acad Dermatol Venereol. 2019;33(11):2197-201.
11. Lee EJ et al. Ann Dermatol. 2003;15(4):133-8.
12. Ahn SK et al. J Dermatol. 2006;33(2):80-90.
There are dozens of skin care products that claim to repair the barrier that do not have the science or ingredient content to back them up.
Does a skin barrier repair moisturizer really repair?
First, let’s briefly review what the skin barrier is. The stratum corneum (SC), the most superficial layer of the epidermis, averages approximately 15-cell layers in thickness.1,2 The keratinocytes reside there in a pattern resembling a brick wall. The “mortar” is composed of the lipid contents extruded from the lamellar granules. This protective barrier functions to prevent transepidermal water loss (TEWL) and entry of allergens, irritants, and pathogens into deeper layers of the skin. This column will focus briefly on the structure and function of the skin barrier and the barrier repair technologies that use synthetic lipids such as myristoyl-palmitoyl and myristyl/palmityl-oxo-stearamide/arachamide MEA.
Structure of the skin barrier
SC keratinocytes are surrounded by lamella made from lipid bilayers. The lipids have hydrophilic heads and hydrophobic tails; the bilayer arises when the hydrophobic tails face the center and the hydrophilic heads face out of the bilayer. This formation yields a disc-shaped hydrophobic lamellar center. There are actually several of these lamellar layers between keratinocytes.

The naturally occurring primary lipids of the bilayer lamellae are made up of an equal ratio of ceramides, cholesterol, and free fatty acid. Arranged in a 1:1:1 ratio, they fit together like pieces of a puzzle to achieve skin barrier homeostasis. The shape and size of these puzzle pieces is critical. An incorrect shape results in a hole in the skin barrier resulting in dehydration, inflammation, and sensitivity.
Ceramides
Ceramides are a complex family of lipids (sphingolipids – a sphingoid base and a fatty acid) involved in cell, as well as barrier, homeostasis and water-holding capacity. In fact, they are known to play a crucial role in cell proliferation, differentiation, and apoptosis.3 There are at least 16 types of naturally occurring ceramides. For years, they have been included in barrier repair moisturizers. They are difficult to work with in moisturizers for several reasons:
- Ceramides are abundant in brain tissue and the ceramides used in moisturizers in the past were derived from bovine brain tissue. Prior to the emergence of bovine spongiform encephalopathy (mad cow disease), many ceramides in skin-care products were animal derived, which made them expensive and undesirable.
- Ceramides in skin care that are made from plant sources are referred to as phyto-derived ceramides. Although they share a similar structure with ceramides that occur in human skin, there are differences in chain length, hydroxylation pattern, and the degree of unsaturation that lead to structural diversity.4 The shape of ceramides is critical for a strong skin barrier because the lipids in the skin barrier must fit together like puzzle pieces to form a water-tight barrier. Natural sources of ceramides include rice, wheat, potato, konjac, and maize. Standardization of ceramide shape and structure makes using phyto-derived ceramides in skin care products challenging.
- Ceramides, because of their waxy consistency, require heat during the mixing process of skin care product manufacturing. This heat can make other ingredients inactive in the skin care formulation. (Ceramides are typically added early in the formulation process, and the heat-sensitive ones are added later.)
- Many forms of ceramides are unstable in the product manufacturing and bottling processes.
- Skin penetration of ceramides depends on the shape and size of ceramides.
Synthetic ceramides have been developed to make ceramides safe, affordable, and more easily formulated into moisturizers. These formulations synthesized in the lab are sometimes called pseudoceramides because they are structurally different compounds that mimic the activity of ceramides. They are developed to be less expensive to manufacture, safer than those derived from animals, and easier to formulate, and they can be made into the specific shape of the ceramide puzzle piece.
Ceramides in skin care
The naturally occurring intercellular lipids of the SC are composed of approximately equal proportions of ceramides, cholesterol, and fatty acids (referred to in this article as the “three barrier lipids” for simplicity).5-9 Alterations in any of these three barrier lipids or their regulatory enzymes result in impairments in the function of the epidermal barrier. Therefore, any synthetic ceramide must mimic the shape of natural ceramides, or the three barrier lipids in the moisturizer must mimic the shape of the entire bilayer lamella. Unfortunately, most barrier repair moisturizers do not meet these criteria and are not true barrier repair moisturizers.
How do you know if a moisturizer repairs the skin barrier?
Clinical tests such as measuring transepidermal water loss (TEWL) with a Tewameter are usually done to support the barrier repair claim. However, occlusive ingredients like oils can lower TEWL without affecting the barrier. In fact, we believe that sebum on the skin can make an impaired barrier and result in normal TEWL even when the barrier is impaired. So, just because a product improved TEWL does not necessarily mean that it repairs the barrier.
One way to test the ability of a moisturizer to repair the barrier is to look at a structural analysis of the moisturizer to see if it forms the requisite bilayer lamellar shape. An easy way to do this testing is to look for the cross pattern under a cross polarized microscope. The cross pattern is known as optical anisotropy. 8
The best barrier repair creams
Optimal barrier repair creams either feature a 1:1:1 ratio of epidermal lipids or form a cross structure when viewed with a cross-polarized microscope.8 There are several categories of barrier repair moisturizers that meet these criteria.
Barrier repair creams with a 1:1:1 ratio of lipids:
Peter Elias, MD, holds the patent on barrier repair moisturizer technology that has a 1:1:1 ratio. His well-established technology is used in a prescription barrier repair cream called EpiCeram® which is approved by the Food and Drug Administration to treat eczema. There are no other moisturizers that I know of that contain this 1:1:1 lipid ratio.
There is a barrier repair cream on the market that contains a 2:4:2 ratio of lipids based on a study that showed that this ratio is effective in older skin with an impaired barrier. It is unknown if this moisturizer forms a cross pattern.
Barrier repair creams that demonstrate a cross pattern:
Multilamellar emulsion (MLE) technology: This barrier repair technology, invented in South Korea, contains the synthetic pseudoceramide called myristyl/palmityl-oxo-stearamide/arachamide MEA (C34H67NO3/C36H71NO3/C38H75NO3), or the pseudoceramide myristoyl-palmitoyl-oxostearamide-arachamide MEA.
In a 2019 pilot study by Ye and colleagues, the investigators treated 33 older volunteers twice daily for 30 days with approximately 3 mL of an emollient containing MLE technology. In addition, 30 untreated older subjects and 11 young volunteers served as controls. The investigators found that the topically applied barrier repair emollient significantly improved barrier function, as well as stratum corneum hydration. Circulating levels of the important, age-related plasma cytokines interleukin-1 beta and IL-6 were found to have normalized, while tumor necrosis factor–alpha decreased markedly. The investigators suggested that repair of the skin barrier might diminish circulating proinflammatory cytokine levels (such as amyloid A) in aged humans, potentially mitigating the development of chronic inflammatory conditions.10
MLE technology has also been shown to improve childhood atopic dermatitis and prevent steroid atrophy.11,12 The consistent use of MLE technology in moisturizers has been shown to alleviate inflammatory factors in the blood and is believed to lessen systemic inflammation.10
Physiologic (PSL) lipid repair technology: This technology was invented by one of the South Korean researchers who helped develop MLE technology. It contains pseudoceramides, fatty acids, and cholesterol. The figure of the cross pattern above, as seen under the cross polarized microscope, is an image taken of this PSL lipid repair technology.
Conclusion
Do not believe that a moisturizer repairs the barrier just because it says so on the label. Three of the most popular body moisturizes used to treat eczema do not actually have the proper formula to repair the barrier. Unfortunately, there are dozens of skin care products that claim to repair the barrier that do not have the science or ingredient content to back them up. To restore the skin barrier to a healthy condition, it is imperative that the barrier repair moisturizers that you are recommending for patients have the correct 1:1:1 ratio of epidermal lipids or contain bilayer lamella that mimic the natural multilamellar layers and display the cross pattern under a cross-polarized microscope.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann has written two textbooks and a New York Times Best Sellers book for consumers. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Revance, Evolus, and Burt’s Bees. She is the CEO of Skin Type Solutions, a company that independently tests skin care products and makes recommendations to physicians on which skin care technologies are best. Write to her at [email protected].
References
1. Christophers E and Kligman AM. J Invest Dermatol. 1964;42:407-9.
2. Blair C. Br J Dermatol. 1968;80(7):430-6.
3. Morita O et al. Food Chem Toxicol. 2009 Apr;47(4):681-6.
4. Tessema E N et al. Skin pharmacology and physiology. 2017;30(3):115-38.
5. Coderch L et al. Am J Clin Dermatol. 2003;4(2):107-29.
6. Man MQ et al. Arch Dermatol. 1993;129(6):728-38.
7. Man MQ M et al. J Invest Dermatol. 1996 May;106(5):1096-101.
8. Park BD et al. J Invest Dermatol. 2003;121(4):794-801.
9. Proksch E and Jensen J. Skin as an organ of protection, in “Fitzpatrick’s Dermatology in General Medicine,” 7th ed. New York: McGraw-Hill, 2008, pp. 383-95.
10. Ye L et al. J Eur Acad Dermatol Venereol. 2019;33(11):2197-201.
11. Lee EJ et al. Ann Dermatol. 2003;15(4):133-8.
12. Ahn SK et al. J Dermatol. 2006;33(2):80-90.
There are dozens of skin care products that claim to repair the barrier that do not have the science or ingredient content to back them up.
Does a skin barrier repair moisturizer really repair?
First, let’s briefly review what the skin barrier is. The stratum corneum (SC), the most superficial layer of the epidermis, averages approximately 15-cell layers in thickness.1,2 The keratinocytes reside there in a pattern resembling a brick wall. The “mortar” is composed of the lipid contents extruded from the lamellar granules. This protective barrier functions to prevent transepidermal water loss (TEWL) and entry of allergens, irritants, and pathogens into deeper layers of the skin. This column will focus briefly on the structure and function of the skin barrier and the barrier repair technologies that use synthetic lipids such as myristoyl-palmitoyl and myristyl/palmityl-oxo-stearamide/arachamide MEA.
Structure of the skin barrier
SC keratinocytes are surrounded by lamella made from lipid bilayers. The lipids have hydrophilic heads and hydrophobic tails; the bilayer arises when the hydrophobic tails face the center and the hydrophilic heads face out of the bilayer. This formation yields a disc-shaped hydrophobic lamellar center. There are actually several of these lamellar layers between keratinocytes.

The naturally occurring primary lipids of the bilayer lamellae are made up of an equal ratio of ceramides, cholesterol, and free fatty acid. Arranged in a 1:1:1 ratio, they fit together like pieces of a puzzle to achieve skin barrier homeostasis. The shape and size of these puzzle pieces is critical. An incorrect shape results in a hole in the skin barrier resulting in dehydration, inflammation, and sensitivity.
Ceramides
Ceramides are a complex family of lipids (sphingolipids – a sphingoid base and a fatty acid) involved in cell, as well as barrier, homeostasis and water-holding capacity. In fact, they are known to play a crucial role in cell proliferation, differentiation, and apoptosis.3 There are at least 16 types of naturally occurring ceramides. For years, they have been included in barrier repair moisturizers. They are difficult to work with in moisturizers for several reasons:
- Ceramides are abundant in brain tissue and the ceramides used in moisturizers in the past were derived from bovine brain tissue. Prior to the emergence of bovine spongiform encephalopathy (mad cow disease), many ceramides in skin-care products were animal derived, which made them expensive and undesirable.
- Ceramides in skin care that are made from plant sources are referred to as phyto-derived ceramides. Although they share a similar structure with ceramides that occur in human skin, there are differences in chain length, hydroxylation pattern, and the degree of unsaturation that lead to structural diversity.4 The shape of ceramides is critical for a strong skin barrier because the lipids in the skin barrier must fit together like puzzle pieces to form a water-tight barrier. Natural sources of ceramides include rice, wheat, potato, konjac, and maize. Standardization of ceramide shape and structure makes using phyto-derived ceramides in skin care products challenging.
- Ceramides, because of their waxy consistency, require heat during the mixing process of skin care product manufacturing. This heat can make other ingredients inactive in the skin care formulation. (Ceramides are typically added early in the formulation process, and the heat-sensitive ones are added later.)
- Many forms of ceramides are unstable in the product manufacturing and bottling processes.
- Skin penetration of ceramides depends on the shape and size of ceramides.
Synthetic ceramides have been developed to make ceramides safe, affordable, and more easily formulated into moisturizers. These formulations synthesized in the lab are sometimes called pseudoceramides because they are structurally different compounds that mimic the activity of ceramides. They are developed to be less expensive to manufacture, safer than those derived from animals, and easier to formulate, and they can be made into the specific shape of the ceramide puzzle piece.
Ceramides in skin care
The naturally occurring intercellular lipids of the SC are composed of approximately equal proportions of ceramides, cholesterol, and fatty acids (referred to in this article as the “three barrier lipids” for simplicity).5-9 Alterations in any of these three barrier lipids or their regulatory enzymes result in impairments in the function of the epidermal barrier. Therefore, any synthetic ceramide must mimic the shape of natural ceramides, or the three barrier lipids in the moisturizer must mimic the shape of the entire bilayer lamella. Unfortunately, most barrier repair moisturizers do not meet these criteria and are not true barrier repair moisturizers.
How do you know if a moisturizer repairs the skin barrier?
Clinical tests such as measuring transepidermal water loss (TEWL) with a Tewameter are usually done to support the barrier repair claim. However, occlusive ingredients like oils can lower TEWL without affecting the barrier. In fact, we believe that sebum on the skin can make an impaired barrier and result in normal TEWL even when the barrier is impaired. So, just because a product improved TEWL does not necessarily mean that it repairs the barrier.
One way to test the ability of a moisturizer to repair the barrier is to look at a structural analysis of the moisturizer to see if it forms the requisite bilayer lamellar shape. An easy way to do this testing is to look for the cross pattern under a cross polarized microscope. The cross pattern is known as optical anisotropy. 8
The best barrier repair creams
Optimal barrier repair creams either feature a 1:1:1 ratio of epidermal lipids or form a cross structure when viewed with a cross-polarized microscope.8 There are several categories of barrier repair moisturizers that meet these criteria.
Barrier repair creams with a 1:1:1 ratio of lipids:
Peter Elias, MD, holds the patent on barrier repair moisturizer technology that has a 1:1:1 ratio. His well-established technology is used in a prescription barrier repair cream called EpiCeram® which is approved by the Food and Drug Administration to treat eczema. There are no other moisturizers that I know of that contain this 1:1:1 lipid ratio.
There is a barrier repair cream on the market that contains a 2:4:2 ratio of lipids based on a study that showed that this ratio is effective in older skin with an impaired barrier. It is unknown if this moisturizer forms a cross pattern.
Barrier repair creams that demonstrate a cross pattern:
Multilamellar emulsion (MLE) technology: This barrier repair technology, invented in South Korea, contains the synthetic pseudoceramide called myristyl/palmityl-oxo-stearamide/arachamide MEA (C34H67NO3/C36H71NO3/C38H75NO3), or the pseudoceramide myristoyl-palmitoyl-oxostearamide-arachamide MEA.
In a 2019 pilot study by Ye and colleagues, the investigators treated 33 older volunteers twice daily for 30 days with approximately 3 mL of an emollient containing MLE technology. In addition, 30 untreated older subjects and 11 young volunteers served as controls. The investigators found that the topically applied barrier repair emollient significantly improved barrier function, as well as stratum corneum hydration. Circulating levels of the important, age-related plasma cytokines interleukin-1 beta and IL-6 were found to have normalized, while tumor necrosis factor–alpha decreased markedly. The investigators suggested that repair of the skin barrier might diminish circulating proinflammatory cytokine levels (such as amyloid A) in aged humans, potentially mitigating the development of chronic inflammatory conditions.10
MLE technology has also been shown to improve childhood atopic dermatitis and prevent steroid atrophy.11,12 The consistent use of MLE technology in moisturizers has been shown to alleviate inflammatory factors in the blood and is believed to lessen systemic inflammation.10
Physiologic (PSL) lipid repair technology: This technology was invented by one of the South Korean researchers who helped develop MLE technology. It contains pseudoceramides, fatty acids, and cholesterol. The figure of the cross pattern above, as seen under the cross polarized microscope, is an image taken of this PSL lipid repair technology.
Conclusion
Do not believe that a moisturizer repairs the barrier just because it says so on the label. Three of the most popular body moisturizes used to treat eczema do not actually have the proper formula to repair the barrier. Unfortunately, there are dozens of skin care products that claim to repair the barrier that do not have the science or ingredient content to back them up. To restore the skin barrier to a healthy condition, it is imperative that the barrier repair moisturizers that you are recommending for patients have the correct 1:1:1 ratio of epidermal lipids or contain bilayer lamella that mimic the natural multilamellar layers and display the cross pattern under a cross-polarized microscope.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur who practices in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann has written two textbooks and a New York Times Best Sellers book for consumers. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Revance, Evolus, and Burt’s Bees. She is the CEO of Skin Type Solutions, a company that independently tests skin care products and makes recommendations to physicians on which skin care technologies are best. Write to her at [email protected].
References
1. Christophers E and Kligman AM. J Invest Dermatol. 1964;42:407-9.
2. Blair C. Br J Dermatol. 1968;80(7):430-6.
3. Morita O et al. Food Chem Toxicol. 2009 Apr;47(4):681-6.
4. Tessema E N et al. Skin pharmacology and physiology. 2017;30(3):115-38.
5. Coderch L et al. Am J Clin Dermatol. 2003;4(2):107-29.
6. Man MQ et al. Arch Dermatol. 1993;129(6):728-38.
7. Man MQ M et al. J Invest Dermatol. 1996 May;106(5):1096-101.
8. Park BD et al. J Invest Dermatol. 2003;121(4):794-801.
9. Proksch E and Jensen J. Skin as an organ of protection, in “Fitzpatrick’s Dermatology in General Medicine,” 7th ed. New York: McGraw-Hill, 2008, pp. 383-95.
10. Ye L et al. J Eur Acad Dermatol Venereol. 2019;33(11):2197-201.
11. Lee EJ et al. Ann Dermatol. 2003;15(4):133-8.
12. Ahn SK et al. J Dermatol. 2006;33(2):80-90.
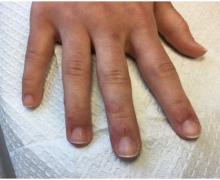
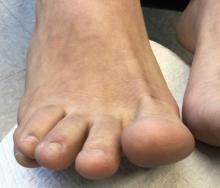
A 22-year-old presented with erythematous papules on her fingers and toes
than men. Clinically, distal extremities such as toes, fingertips and heels, as well as the rims of the ears or nose develop erythematous to purple plaques. Lesions may be painful or pruritic. Over time, lesions may develop atrophy and resemble those of discoid lupus. While the pathogenesis is unknown, exposure to cold or wet environments can precipitate lesions.
Histopathology reveals a deep and superficial lymphocytic infiltrate with perieccrine involvement and fibrin deposition in vessels. Dermal edema is often present. Direct immunofluorescence shows an interface dermatitis positive for IgM, IgA, and C3.
The Mayo Clinic developed diagnostic criteria for diagnosing chilblains lupus. Two major criteria are acral skin lesions induced by cold exposure and evidence of lupus erythematosus in skin lesions (histopathologically or by direct immunofluorescence). Three minor criteria are the coexistence of systemic lupus erythematosus or discoid lupus erythematosus, response to antilupus treatment, and negative cryoglobulin and cold agglutinin studies.
Chilblains, or perniosis, has a similar clinical presentation to chilblain lupus erythematosus. However, serologic evidence of lupus, such as a positive antinuclear antibody (ANA), will be absent. Lupus pernio (Besnier-Tenneson syndrome) is a form of sarcoidosis that tends to favor the nose. These lesions are not precipitated by cold. It can be differentiated on histology. “COVID toes” is an entity described during the coronavirus pandemic, during which dermatologists noted pernio-like lesions in patients testing positive for coronavirus.
The patient’s labs revealed a positive ANA at 1:320 in a nucleolar speckled pattern, elevated double-stranded DNA, low C3 and C4 levels, elevated cardiolipin IgM Ab, and elevated sedimentation rate. COVID-19 antigen testing and COVID-19 antibodies were negative. A serum protein electrophoresis was negative. Cryoglobulins were negative.
Treatment includes protection from cold. Smoking cessation should be discussed. Topical steroids and topical calcineurin inhibitors are first-line treatments for mild disease. Antimalarials, such as hydroxychloroquine can be helpful. Systemic calcium channel blockers, systemic steroids, mycophenolate mofetil, and tacrolimus have all been reported as treatments. This patient responded well to hydroxychloroquine and topical steroids with full resolution of lesions.
This case was submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Su WP et al. Cutis. 1994 Dec;54(6):395-9.
Werth V and Newman S. Chilblain lupus (SLE pernio). Dermatology Advisor. 2017.
than men. Clinically, distal extremities such as toes, fingertips and heels, as well as the rims of the ears or nose develop erythematous to purple plaques. Lesions may be painful or pruritic. Over time, lesions may develop atrophy and resemble those of discoid lupus. While the pathogenesis is unknown, exposure to cold or wet environments can precipitate lesions.
Histopathology reveals a deep and superficial lymphocytic infiltrate with perieccrine involvement and fibrin deposition in vessels. Dermal edema is often present. Direct immunofluorescence shows an interface dermatitis positive for IgM, IgA, and C3.
The Mayo Clinic developed diagnostic criteria for diagnosing chilblains lupus. Two major criteria are acral skin lesions induced by cold exposure and evidence of lupus erythematosus in skin lesions (histopathologically or by direct immunofluorescence). Three minor criteria are the coexistence of systemic lupus erythematosus or discoid lupus erythematosus, response to antilupus treatment, and negative cryoglobulin and cold agglutinin studies.
Chilblains, or perniosis, has a similar clinical presentation to chilblain lupus erythematosus. However, serologic evidence of lupus, such as a positive antinuclear antibody (ANA), will be absent. Lupus pernio (Besnier-Tenneson syndrome) is a form of sarcoidosis that tends to favor the nose. These lesions are not precipitated by cold. It can be differentiated on histology. “COVID toes” is an entity described during the coronavirus pandemic, during which dermatologists noted pernio-like lesions in patients testing positive for coronavirus.
The patient’s labs revealed a positive ANA at 1:320 in a nucleolar speckled pattern, elevated double-stranded DNA, low C3 and C4 levels, elevated cardiolipin IgM Ab, and elevated sedimentation rate. COVID-19 antigen testing and COVID-19 antibodies were negative. A serum protein electrophoresis was negative. Cryoglobulins were negative.
Treatment includes protection from cold. Smoking cessation should be discussed. Topical steroids and topical calcineurin inhibitors are first-line treatments for mild disease. Antimalarials, such as hydroxychloroquine can be helpful. Systemic calcium channel blockers, systemic steroids, mycophenolate mofetil, and tacrolimus have all been reported as treatments. This patient responded well to hydroxychloroquine and topical steroids with full resolution of lesions.
This case was submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Su WP et al. Cutis. 1994 Dec;54(6):395-9.
Werth V and Newman S. Chilblain lupus (SLE pernio). Dermatology Advisor. 2017.
than men. Clinically, distal extremities such as toes, fingertips and heels, as well as the rims of the ears or nose develop erythematous to purple plaques. Lesions may be painful or pruritic. Over time, lesions may develop atrophy and resemble those of discoid lupus. While the pathogenesis is unknown, exposure to cold or wet environments can precipitate lesions.
Histopathology reveals a deep and superficial lymphocytic infiltrate with perieccrine involvement and fibrin deposition in vessels. Dermal edema is often present. Direct immunofluorescence shows an interface dermatitis positive for IgM, IgA, and C3.
The Mayo Clinic developed diagnostic criteria for diagnosing chilblains lupus. Two major criteria are acral skin lesions induced by cold exposure and evidence of lupus erythematosus in skin lesions (histopathologically or by direct immunofluorescence). Three minor criteria are the coexistence of systemic lupus erythematosus or discoid lupus erythematosus, response to antilupus treatment, and negative cryoglobulin and cold agglutinin studies.
Chilblains, or perniosis, has a similar clinical presentation to chilblain lupus erythematosus. However, serologic evidence of lupus, such as a positive antinuclear antibody (ANA), will be absent. Lupus pernio (Besnier-Tenneson syndrome) is a form of sarcoidosis that tends to favor the nose. These lesions are not precipitated by cold. It can be differentiated on histology. “COVID toes” is an entity described during the coronavirus pandemic, during which dermatologists noted pernio-like lesions in patients testing positive for coronavirus.
The patient’s labs revealed a positive ANA at 1:320 in a nucleolar speckled pattern, elevated double-stranded DNA, low C3 and C4 levels, elevated cardiolipin IgM Ab, and elevated sedimentation rate. COVID-19 antigen testing and COVID-19 antibodies were negative. A serum protein electrophoresis was negative. Cryoglobulins were negative.
Treatment includes protection from cold. Smoking cessation should be discussed. Topical steroids and topical calcineurin inhibitors are first-line treatments for mild disease. Antimalarials, such as hydroxychloroquine can be helpful. Systemic calcium channel blockers, systemic steroids, mycophenolate mofetil, and tacrolimus have all been reported as treatments. This patient responded well to hydroxychloroquine and topical steroids with full resolution of lesions.
This case was submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Su WP et al. Cutis. 1994 Dec;54(6):395-9.
Werth V and Newman S. Chilblain lupus (SLE pernio). Dermatology Advisor. 2017.
12 state boards have disciplined docs for COVID misinformation
, according to a new survey from the Federation of State Medical Boards (FSMB).
The FSMB reports that in its 2021 annual survey two-thirds of its 71 member boards (which includes the United States, its territories, and Washington, DC) reported an increase in complaints about doctors spreading false or misleading information.
“The staggering number of state medical boards that have seen an increase in COVID-19 disinformation complaints is a sign of how widespread the issue has become,” said Humayun J. Chaudhry, DO, MACP, president and CEO of the FSMB, in a statement.
The FSMB board of directors warned physicians in July that they risked disciplinary action if they spread COVID-19 vaccine misinformation or disinformation.
The organization said 15 state boards have now adopted similar statements.
Dr. Chaudhry said the FSMB was “encouraged by the number of boards that have already taken action to combat COVID-19 disinformation by disciplining physicians who engage in that behavior and by reminding all physicians that their words and actions matter, and they should think twice before spreading disinformation that may harm patients.”
This news organization asked the FSMB for further comment on why more physicians have not been disciplined, but did not receive a response before publication.
Misinformation policies a new battleground
The FSMB and member board policies on COVID-19 around the country have become a new front in the war against mandates and restrictions.
The Tennessee Board of Medical Examiners voted just recently to remove its statement of policy against the spread of misinformation from its website after a Republican lawmaker allegedly threatened to dissolve the board.
The vote came just a few months after the board had approved the policy. The board did not rescind the policy, however, according to a report by the Associated Press.
In California, the president of the state’s medical board tweeted on December 8 about what she said was an incident of harassment by a group that has promoted “fake COVID-19 treatments.”Ms. Kristina Lawson said she observed four men sitting in front of her house in a truck. They flew a drone over her residence, and then followed her to work, parking nose-to-nose with her vehicle.
Ms. Lawson claimed that when she went to drive home the four men ambushed her in what was by then a dark parking garage. She said her “concern turned to terror” as they jumped out, cameras and recording equipment in hand.
The men told law enforcement called to the scene that they were just trying to interview her, according to a statement emailed by Ms. Lawson.
They had not made such a request to the California Medical Board.
Ms. Lawson tweeted that she would continue to volunteer for the board. “That means protecting Californians from bad doctors, and ensuring disinformation and misinformation do not detract from our work to protect patients and consumers,” she wrote.
The men who ambushed Ms. Larson allegedly identified themselves and were wearing clothing emblazoned with the logo of “America’s Frontline Doctors,” an organization that has trafficked in COVID-19 conspiracy theories and promoted unproven treatments like hydroxychloroquine and ivermectin, according to Time. It is led by Simone Gold, MD, who was arrested for breaching the U.S. Capitol on January 6.
Despite her activities, on November 30, the California Medical Board renewed Ms. Gold’s 2-year license to practice.
Who’s being disciplined, who’s not
Dr. Gold is not alone. An investigation by NPRin September found that 15 of 16 physicians who have spread false information in a high-profile manner have medical licenses in good standing.
Sherri Tenpenny, DO, who has claimed that COVID-19 vaccines magnetize people and “interface” with 5G cell phone towers, was able to renew her license with the Ohio State Medical Board on October 1, according to the Cincinnati Enquirer.
Some boards have acted. The Oregon Medical Board revoked the license of Steven LaTulippe, MD, and fined him $10,000 for spreading misinformation about masks and overprescribing opioids.
In August, Rhode Island’s Board of Medical Licensure suspended Mark Brody’s license for 5 years after finding that the doctor spread falsehoods about COVID-19 vaccines, according to board documents.
Maine physician Paul Gosselin, DO, is on temporary suspension until a February hearing, while the osteopathic board investigates his issuance of vaccine exemption letters and the promotion of unproven COVID-19 therapies.
The board found that Gosselin had “engaged in conduct that constitutes fraud or deceit,” according to official documents.
The Washington State Medical Board has opened an investigation into Ryan N. Cole, MD, a physician who has claimed that COVID vaccines are “fake,” and was appointed to a regional health board in Idaho in September, according to the Washington Post.
The Idaho Capital Sun reported that Dr. Cole claims he is licensed in 11 states, including Washington. The Idaho Medical Association has also filed a complaint about Dr. Cole with the Idaho Board of Medicine, according to the paper.
New FSMB guidance coming
The FSMB said it expects more disciplinary actions as investigations continue to unfold.
The organization is drafting a new policy document that will include further guidelines and recommendations for state medical boards “to help address the spread of disinformation,” it said. The final document would be released in April 2022.
In the meantime, some states, like Tennessee and others, are trying to find ways to counter the current policy — a development the FSMB called “troubling.”
“The FSMB strongly opposes any effort to restrict a board’s authority to evaluate the standard of care and assess risk for patient harm,” the organization said in its statement.
A version of this article was first published on Medscape.com.
, according to a new survey from the Federation of State Medical Boards (FSMB).
The FSMB reports that in its 2021 annual survey two-thirds of its 71 member boards (which includes the United States, its territories, and Washington, DC) reported an increase in complaints about doctors spreading false or misleading information.
“The staggering number of state medical boards that have seen an increase in COVID-19 disinformation complaints is a sign of how widespread the issue has become,” said Humayun J. Chaudhry, DO, MACP, president and CEO of the FSMB, in a statement.
The FSMB board of directors warned physicians in July that they risked disciplinary action if they spread COVID-19 vaccine misinformation or disinformation.
The organization said 15 state boards have now adopted similar statements.
Dr. Chaudhry said the FSMB was “encouraged by the number of boards that have already taken action to combat COVID-19 disinformation by disciplining physicians who engage in that behavior and by reminding all physicians that their words and actions matter, and they should think twice before spreading disinformation that may harm patients.”
This news organization asked the FSMB for further comment on why more physicians have not been disciplined, but did not receive a response before publication.
Misinformation policies a new battleground
The FSMB and member board policies on COVID-19 around the country have become a new front in the war against mandates and restrictions.
The Tennessee Board of Medical Examiners voted just recently to remove its statement of policy against the spread of misinformation from its website after a Republican lawmaker allegedly threatened to dissolve the board.
The vote came just a few months after the board had approved the policy. The board did not rescind the policy, however, according to a report by the Associated Press.
In California, the president of the state’s medical board tweeted on December 8 about what she said was an incident of harassment by a group that has promoted “fake COVID-19 treatments.”Ms. Kristina Lawson said she observed four men sitting in front of her house in a truck. They flew a drone over her residence, and then followed her to work, parking nose-to-nose with her vehicle.
Ms. Lawson claimed that when she went to drive home the four men ambushed her in what was by then a dark parking garage. She said her “concern turned to terror” as they jumped out, cameras and recording equipment in hand.
The men told law enforcement called to the scene that they were just trying to interview her, according to a statement emailed by Ms. Lawson.
They had not made such a request to the California Medical Board.
Ms. Lawson tweeted that she would continue to volunteer for the board. “That means protecting Californians from bad doctors, and ensuring disinformation and misinformation do not detract from our work to protect patients and consumers,” she wrote.
The men who ambushed Ms. Larson allegedly identified themselves and were wearing clothing emblazoned with the logo of “America’s Frontline Doctors,” an organization that has trafficked in COVID-19 conspiracy theories and promoted unproven treatments like hydroxychloroquine and ivermectin, according to Time. It is led by Simone Gold, MD, who was arrested for breaching the U.S. Capitol on January 6.
Despite her activities, on November 30, the California Medical Board renewed Ms. Gold’s 2-year license to practice.
Who’s being disciplined, who’s not
Dr. Gold is not alone. An investigation by NPRin September found that 15 of 16 physicians who have spread false information in a high-profile manner have medical licenses in good standing.
Sherri Tenpenny, DO, who has claimed that COVID-19 vaccines magnetize people and “interface” with 5G cell phone towers, was able to renew her license with the Ohio State Medical Board on October 1, according to the Cincinnati Enquirer.
Some boards have acted. The Oregon Medical Board revoked the license of Steven LaTulippe, MD, and fined him $10,000 for spreading misinformation about masks and overprescribing opioids.
In August, Rhode Island’s Board of Medical Licensure suspended Mark Brody’s license for 5 years after finding that the doctor spread falsehoods about COVID-19 vaccines, according to board documents.
Maine physician Paul Gosselin, DO, is on temporary suspension until a February hearing, while the osteopathic board investigates his issuance of vaccine exemption letters and the promotion of unproven COVID-19 therapies.
The board found that Gosselin had “engaged in conduct that constitutes fraud or deceit,” according to official documents.
The Washington State Medical Board has opened an investigation into Ryan N. Cole, MD, a physician who has claimed that COVID vaccines are “fake,” and was appointed to a regional health board in Idaho in September, according to the Washington Post.
The Idaho Capital Sun reported that Dr. Cole claims he is licensed in 11 states, including Washington. The Idaho Medical Association has also filed a complaint about Dr. Cole with the Idaho Board of Medicine, according to the paper.
New FSMB guidance coming
The FSMB said it expects more disciplinary actions as investigations continue to unfold.
The organization is drafting a new policy document that will include further guidelines and recommendations for state medical boards “to help address the spread of disinformation,” it said. The final document would be released in April 2022.
In the meantime, some states, like Tennessee and others, are trying to find ways to counter the current policy — a development the FSMB called “troubling.”
“The FSMB strongly opposes any effort to restrict a board’s authority to evaluate the standard of care and assess risk for patient harm,” the organization said in its statement.
A version of this article was first published on Medscape.com.
, according to a new survey from the Federation of State Medical Boards (FSMB).
The FSMB reports that in its 2021 annual survey two-thirds of its 71 member boards (which includes the United States, its territories, and Washington, DC) reported an increase in complaints about doctors spreading false or misleading information.
“The staggering number of state medical boards that have seen an increase in COVID-19 disinformation complaints is a sign of how widespread the issue has become,” said Humayun J. Chaudhry, DO, MACP, president and CEO of the FSMB, in a statement.
The FSMB board of directors warned physicians in July that they risked disciplinary action if they spread COVID-19 vaccine misinformation or disinformation.
The organization said 15 state boards have now adopted similar statements.
Dr. Chaudhry said the FSMB was “encouraged by the number of boards that have already taken action to combat COVID-19 disinformation by disciplining physicians who engage in that behavior and by reminding all physicians that their words and actions matter, and they should think twice before spreading disinformation that may harm patients.”
This news organization asked the FSMB for further comment on why more physicians have not been disciplined, but did not receive a response before publication.
Misinformation policies a new battleground
The FSMB and member board policies on COVID-19 around the country have become a new front in the war against mandates and restrictions.
The Tennessee Board of Medical Examiners voted just recently to remove its statement of policy against the spread of misinformation from its website after a Republican lawmaker allegedly threatened to dissolve the board.
The vote came just a few months after the board had approved the policy. The board did not rescind the policy, however, according to a report by the Associated Press.
In California, the president of the state’s medical board tweeted on December 8 about what she said was an incident of harassment by a group that has promoted “fake COVID-19 treatments.”Ms. Kristina Lawson said she observed four men sitting in front of her house in a truck. They flew a drone over her residence, and then followed her to work, parking nose-to-nose with her vehicle.
Ms. Lawson claimed that when she went to drive home the four men ambushed her in what was by then a dark parking garage. She said her “concern turned to terror” as they jumped out, cameras and recording equipment in hand.
The men told law enforcement called to the scene that they were just trying to interview her, according to a statement emailed by Ms. Lawson.
They had not made such a request to the California Medical Board.
Ms. Lawson tweeted that she would continue to volunteer for the board. “That means protecting Californians from bad doctors, and ensuring disinformation and misinformation do not detract from our work to protect patients and consumers,” she wrote.
The men who ambushed Ms. Larson allegedly identified themselves and were wearing clothing emblazoned with the logo of “America’s Frontline Doctors,” an organization that has trafficked in COVID-19 conspiracy theories and promoted unproven treatments like hydroxychloroquine and ivermectin, according to Time. It is led by Simone Gold, MD, who was arrested for breaching the U.S. Capitol on January 6.
Despite her activities, on November 30, the California Medical Board renewed Ms. Gold’s 2-year license to practice.
Who’s being disciplined, who’s not
Dr. Gold is not alone. An investigation by NPRin September found that 15 of 16 physicians who have spread false information in a high-profile manner have medical licenses in good standing.
Sherri Tenpenny, DO, who has claimed that COVID-19 vaccines magnetize people and “interface” with 5G cell phone towers, was able to renew her license with the Ohio State Medical Board on October 1, according to the Cincinnati Enquirer.
Some boards have acted. The Oregon Medical Board revoked the license of Steven LaTulippe, MD, and fined him $10,000 for spreading misinformation about masks and overprescribing opioids.
In August, Rhode Island’s Board of Medical Licensure suspended Mark Brody’s license for 5 years after finding that the doctor spread falsehoods about COVID-19 vaccines, according to board documents.
Maine physician Paul Gosselin, DO, is on temporary suspension until a February hearing, while the osteopathic board investigates his issuance of vaccine exemption letters and the promotion of unproven COVID-19 therapies.
The board found that Gosselin had “engaged in conduct that constitutes fraud or deceit,” according to official documents.
The Washington State Medical Board has opened an investigation into Ryan N. Cole, MD, a physician who has claimed that COVID vaccines are “fake,” and was appointed to a regional health board in Idaho in September, according to the Washington Post.
The Idaho Capital Sun reported that Dr. Cole claims he is licensed in 11 states, including Washington. The Idaho Medical Association has also filed a complaint about Dr. Cole with the Idaho Board of Medicine, according to the paper.
New FSMB guidance coming
The FSMB said it expects more disciplinary actions as investigations continue to unfold.
The organization is drafting a new policy document that will include further guidelines and recommendations for state medical boards “to help address the spread of disinformation,” it said. The final document would be released in April 2022.
In the meantime, some states, like Tennessee and others, are trying to find ways to counter the current policy — a development the FSMB called “troubling.”
“The FSMB strongly opposes any effort to restrict a board’s authority to evaluate the standard of care and assess risk for patient harm,” the organization said in its statement.
A version of this article was first published on Medscape.com.
Major COVID-19 case growth expected in coming weeks
by the PolicyLab at Children’s Hospital of Philadelphia.
Large metropolitan areas, especially those in the Northeast, are already seeing a major increase in cases following Thanksgiving, and that trend is expected to continue.
“Why? Simply stated, the large amount of Thanksgiving travel and gatherings undermined the nation’s pandemic footing and has elevated disease burden in areas of the country that were fortunate to have lower case rates before the holidays,” the forecasters wrote.
Case numbers in New York City are expected to double throughout December, the forecasters said. Similar growth could happen across Boston, Philadelphia, and Baltimore.
Overall, COVID-19 cases, hospitalizations, and deaths are rising across the United States but remain below levels seen during the summer and last winter’s surges, according to the New York Times. The increase is still being driven by the Delta variant, though it remains unclear how the Omicron variant, which has been detected in 27 states, could affect the trends in the coming weeks.
During the past week, the United States has reported an average of more than 120,000 new cases each day, the newspaper reported, which is an increase of 38% from two weeks ago.
The daily average of COVID-19 hospitalizations is around 64,000, which marks an increase of 22% from two weeks ago. More than 1,300 deaths are being reported each day, which is up 26%.
Numerous states are reporting double the cases from two weeks ago, stretching across the country from states in the Northeast such as Connecticut and Rhode Island to southern states such as North Carolina and Texas and western states such as California.
The Great Lakes region and the Northeast are seeing some of the most severe increases, the newspaper reported. New Hampshire leads the United States in recent cases per capita, and Maine has reported more cases in the past week than in any other seven-day period during the pandemic.
Michigan has the country’s highest hospitalization rate, and federal medical teams have been sent to the state to help with the surge in patients, according to The Detroit News. Michigan’s top public health officials described the surge as a “critical” and “deeply concerning” situation on Dec. 10, and they requested 200 more ventilators from the Strategic National Stockpile.
Indiana, Maine, and New York have also requested aid from the National Guard, according to USA Today. Health officials in those states urged residents to get vaccines or booster shots and wear masks in indoor public settings.
The Omicron variant can evade some vaccine protection, but booster shots can increase efficacy and offer more coverage, Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said Dec. 12.
“If you want to be optimally protected, absolutely get a booster,” he said on ABC’s “This Week.”
In addition, New York Gov. Kathy Hochul has announced a statewide mask mandate, which will take effect Dec. 13. Masks will be required in all indoor public spaces and businesses, unless the location implements a vaccine requirement instead, the news outlet reported.
A version of this article first appeared on WebMD.com.
by the PolicyLab at Children’s Hospital of Philadelphia.
Large metropolitan areas, especially those in the Northeast, are already seeing a major increase in cases following Thanksgiving, and that trend is expected to continue.
“Why? Simply stated, the large amount of Thanksgiving travel and gatherings undermined the nation’s pandemic footing and has elevated disease burden in areas of the country that were fortunate to have lower case rates before the holidays,” the forecasters wrote.
Case numbers in New York City are expected to double throughout December, the forecasters said. Similar growth could happen across Boston, Philadelphia, and Baltimore.
Overall, COVID-19 cases, hospitalizations, and deaths are rising across the United States but remain below levels seen during the summer and last winter’s surges, according to the New York Times. The increase is still being driven by the Delta variant, though it remains unclear how the Omicron variant, which has been detected in 27 states, could affect the trends in the coming weeks.
During the past week, the United States has reported an average of more than 120,000 new cases each day, the newspaper reported, which is an increase of 38% from two weeks ago.
The daily average of COVID-19 hospitalizations is around 64,000, which marks an increase of 22% from two weeks ago. More than 1,300 deaths are being reported each day, which is up 26%.
Numerous states are reporting double the cases from two weeks ago, stretching across the country from states in the Northeast such as Connecticut and Rhode Island to southern states such as North Carolina and Texas and western states such as California.
The Great Lakes region and the Northeast are seeing some of the most severe increases, the newspaper reported. New Hampshire leads the United States in recent cases per capita, and Maine has reported more cases in the past week than in any other seven-day period during the pandemic.
Michigan has the country’s highest hospitalization rate, and federal medical teams have been sent to the state to help with the surge in patients, according to The Detroit News. Michigan’s top public health officials described the surge as a “critical” and “deeply concerning” situation on Dec. 10, and they requested 200 more ventilators from the Strategic National Stockpile.
Indiana, Maine, and New York have also requested aid from the National Guard, according to USA Today. Health officials in those states urged residents to get vaccines or booster shots and wear masks in indoor public settings.
The Omicron variant can evade some vaccine protection, but booster shots can increase efficacy and offer more coverage, Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said Dec. 12.
“If you want to be optimally protected, absolutely get a booster,” he said on ABC’s “This Week.”
In addition, New York Gov. Kathy Hochul has announced a statewide mask mandate, which will take effect Dec. 13. Masks will be required in all indoor public spaces and businesses, unless the location implements a vaccine requirement instead, the news outlet reported.
A version of this article first appeared on WebMD.com.
by the PolicyLab at Children’s Hospital of Philadelphia.
Large metropolitan areas, especially those in the Northeast, are already seeing a major increase in cases following Thanksgiving, and that trend is expected to continue.
“Why? Simply stated, the large amount of Thanksgiving travel and gatherings undermined the nation’s pandemic footing and has elevated disease burden in areas of the country that were fortunate to have lower case rates before the holidays,” the forecasters wrote.
Case numbers in New York City are expected to double throughout December, the forecasters said. Similar growth could happen across Boston, Philadelphia, and Baltimore.
Overall, COVID-19 cases, hospitalizations, and deaths are rising across the United States but remain below levels seen during the summer and last winter’s surges, according to the New York Times. The increase is still being driven by the Delta variant, though it remains unclear how the Omicron variant, which has been detected in 27 states, could affect the trends in the coming weeks.
During the past week, the United States has reported an average of more than 120,000 new cases each day, the newspaper reported, which is an increase of 38% from two weeks ago.
The daily average of COVID-19 hospitalizations is around 64,000, which marks an increase of 22% from two weeks ago. More than 1,300 deaths are being reported each day, which is up 26%.
Numerous states are reporting double the cases from two weeks ago, stretching across the country from states in the Northeast such as Connecticut and Rhode Island to southern states such as North Carolina and Texas and western states such as California.
The Great Lakes region and the Northeast are seeing some of the most severe increases, the newspaper reported. New Hampshire leads the United States in recent cases per capita, and Maine has reported more cases in the past week than in any other seven-day period during the pandemic.
Michigan has the country’s highest hospitalization rate, and federal medical teams have been sent to the state to help with the surge in patients, according to The Detroit News. Michigan’s top public health officials described the surge as a “critical” and “deeply concerning” situation on Dec. 10, and they requested 200 more ventilators from the Strategic National Stockpile.
Indiana, Maine, and New York have also requested aid from the National Guard, according to USA Today. Health officials in those states urged residents to get vaccines or booster shots and wear masks in indoor public settings.
The Omicron variant can evade some vaccine protection, but booster shots can increase efficacy and offer more coverage, Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said Dec. 12.
“If you want to be optimally protected, absolutely get a booster,” he said on ABC’s “This Week.”
In addition, New York Gov. Kathy Hochul has announced a statewide mask mandate, which will take effect Dec. 13. Masks will be required in all indoor public spaces and businesses, unless the location implements a vaccine requirement instead, the news outlet reported.
A version of this article first appeared on WebMD.com.
Abrocitinib approved for atopic dermatitis in Europe
who are candidates for systemic therapy, the manufacturer announced.
Approval by the European Commission was based on the results of studies that include four phase 3 clinical trials (JADE MONO-1, JADE-MONO-2, JADE COMPARE, JADE REGIMEN) and an ongoing open-label extension study (JADE EXTEND) in over 2,800 patients, according to the Pfizer press release announcing the approval. The approved doses are 100 and 200 mg a day; a 50-mg dose was approved for patients with moderate and severe renal impairment and “ certain patients receiving treatment with inhibitors of cytochrome P450 (CYP) 2C19,” the release said.
The approval follows a positive opinion by the Committee for Medicinal Products for Human Use of the European Medicines Agency supporting marketing authorization for treating AD, issued in October. It will be marketed as Cibinqo.
Abrocitinib is under review at the Food and Drug Administration. It was approved earlier in 2021 for treating AD in the United Kingdom, Japan, and Korea.
[email protected]
who are candidates for systemic therapy, the manufacturer announced.
Approval by the European Commission was based on the results of studies that include four phase 3 clinical trials (JADE MONO-1, JADE-MONO-2, JADE COMPARE, JADE REGIMEN) and an ongoing open-label extension study (JADE EXTEND) in over 2,800 patients, according to the Pfizer press release announcing the approval. The approved doses are 100 and 200 mg a day; a 50-mg dose was approved for patients with moderate and severe renal impairment and “ certain patients receiving treatment with inhibitors of cytochrome P450 (CYP) 2C19,” the release said.
The approval follows a positive opinion by the Committee for Medicinal Products for Human Use of the European Medicines Agency supporting marketing authorization for treating AD, issued in October. It will be marketed as Cibinqo.
Abrocitinib is under review at the Food and Drug Administration. It was approved earlier in 2021 for treating AD in the United Kingdom, Japan, and Korea.
[email protected]
who are candidates for systemic therapy, the manufacturer announced.
Approval by the European Commission was based on the results of studies that include four phase 3 clinical trials (JADE MONO-1, JADE-MONO-2, JADE COMPARE, JADE REGIMEN) and an ongoing open-label extension study (JADE EXTEND) in over 2,800 patients, according to the Pfizer press release announcing the approval. The approved doses are 100 and 200 mg a day; a 50-mg dose was approved for patients with moderate and severe renal impairment and “ certain patients receiving treatment with inhibitors of cytochrome P450 (CYP) 2C19,” the release said.
The approval follows a positive opinion by the Committee for Medicinal Products for Human Use of the European Medicines Agency supporting marketing authorization for treating AD, issued in October. It will be marketed as Cibinqo.
Abrocitinib is under review at the Food and Drug Administration. It was approved earlier in 2021 for treating AD in the United Kingdom, Japan, and Korea.
[email protected]
What is the diagnosis?
As the lesion was growing, getting more violaceous and indurated, a biopsy was performed. The biopsy showed multiple discrete lobules of dermal capillaries with slight extension into the superficial subcutis. Capillary lobules demonstrate the “cannonball-like” architecture often associated with tufted angioma, and some lobules showed bulging into adjacent thin-walled vessels. Spindled endothelial cells lining slit-like vessels were present in the mid dermis, although this comprises a minority of the lesion. The majority of the subcutis was uninvolved. The findings are overall most consistent with a tufted angioma.
Kaposiform hemangioendothelioma (KHE) has been considered given the presence of occasional slit-like vascular spaces; however, the lesion is predominantly superficial and therefore the lesion is best classified as tufted angioma. GLUT–1 staining was negative.
At the time of biopsy, blood work was ordered, which showed a normal complete blood count with normal number of platelets, slightly elevated D-dimer, and slightly low fibrinogen. Several repeat blood counts and coagulation tests once a week for a few weeks revealed no changes.
The patient was started on aspirin at a dose of 5 mg/kg per day. After a week on the medication the lesion was starting to get smaller and less red.
Tufted angiomas are a rare type of vascular tumor within the spectrum of kaposiform hemangioendotheliomas. Most cases present within the first year of life; some occur at birth. They usually present as papules, plaques, or erythematous, violaceous indurated nodules on the face, neck, trunk, and extremities. The lesions can also be present with hyperhidrosis and hypertrichosis. Clinically, the lesions will have to be differentiated from other vascular tumors such as infantile hemangiomas, congenital hemangiomas, and Kaposi’s sarcoma, as well as subcutaneous fat necrosis of the newborn, cellulitis, and nonaccidental trauma.
Pathogenesis of tufted angiomas is poorly understood. A recent case report found a somatic mutation on GNA14.This protein regulates Ras activity and modulates endothelial cell permeability and migration in response to FGF2 and VEGFA. The p.205L mutation causes activation of GNA14, which upregulates pERK-MAPK pathway, suggesting MAPK inhibition as a potential target for therapy. Clinically, tufted angioma can present in three patterns: uncomplicated tufted angioma (most common type); tufted angioma without thrombocytopenia but with chronic coagulopathy, as it was seen in our patient; and tufted angioma associated with Kasabach-Merritt phenomenon (KMP). KMP is characterized by thrombocytopenia in association with microangiopathic hemolytic anemia, consumptive coagulopathy, and enlarging vascular tumor. Treatment of uncomplicated tufted angioma will depend on symptomatology, size, and location of the lesion. Smaller lesions in noncosmetically sensitive areas can be treated with surgical excision. Cases that are not amenable to excision can be treated with aspirin. There are also reports of response to topical modalities including tacrolimus and timolol. For complicated cases associated with KMP, sirolimus, systemic corticosteroids, ticlopidine, interferon, or vincristine are recommended. Some lesions may demonstrate spontaneous regression.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Cohen S et al. Dermatol Online J. 2019 Sep 15;25(9):13030/qt6pv254mc.
Lim YH et al. Pediatr Dermatol. 2019 Nov;36(6):963-4.
Prasuna A, Rao PN. Indian Dermatol Online J. 2015;6:266-8.
As the lesion was growing, getting more violaceous and indurated, a biopsy was performed. The biopsy showed multiple discrete lobules of dermal capillaries with slight extension into the superficial subcutis. Capillary lobules demonstrate the “cannonball-like” architecture often associated with tufted angioma, and some lobules showed bulging into adjacent thin-walled vessels. Spindled endothelial cells lining slit-like vessels were present in the mid dermis, although this comprises a minority of the lesion. The majority of the subcutis was uninvolved. The findings are overall most consistent with a tufted angioma.
Kaposiform hemangioendothelioma (KHE) has been considered given the presence of occasional slit-like vascular spaces; however, the lesion is predominantly superficial and therefore the lesion is best classified as tufted angioma. GLUT–1 staining was negative.
At the time of biopsy, blood work was ordered, which showed a normal complete blood count with normal number of platelets, slightly elevated D-dimer, and slightly low fibrinogen. Several repeat blood counts and coagulation tests once a week for a few weeks revealed no changes.
The patient was started on aspirin at a dose of 5 mg/kg per day. After a week on the medication the lesion was starting to get smaller and less red.
Tufted angiomas are a rare type of vascular tumor within the spectrum of kaposiform hemangioendotheliomas. Most cases present within the first year of life; some occur at birth. They usually present as papules, plaques, or erythematous, violaceous indurated nodules on the face, neck, trunk, and extremities. The lesions can also be present with hyperhidrosis and hypertrichosis. Clinically, the lesions will have to be differentiated from other vascular tumors such as infantile hemangiomas, congenital hemangiomas, and Kaposi’s sarcoma, as well as subcutaneous fat necrosis of the newborn, cellulitis, and nonaccidental trauma.
Pathogenesis of tufted angiomas is poorly understood. A recent case report found a somatic mutation on GNA14.This protein regulates Ras activity and modulates endothelial cell permeability and migration in response to FGF2 and VEGFA. The p.205L mutation causes activation of GNA14, which upregulates pERK-MAPK pathway, suggesting MAPK inhibition as a potential target for therapy. Clinically, tufted angioma can present in three patterns: uncomplicated tufted angioma (most common type); tufted angioma without thrombocytopenia but with chronic coagulopathy, as it was seen in our patient; and tufted angioma associated with Kasabach-Merritt phenomenon (KMP). KMP is characterized by thrombocytopenia in association with microangiopathic hemolytic anemia, consumptive coagulopathy, and enlarging vascular tumor. Treatment of uncomplicated tufted angioma will depend on symptomatology, size, and location of the lesion. Smaller lesions in noncosmetically sensitive areas can be treated with surgical excision. Cases that are not amenable to excision can be treated with aspirin. There are also reports of response to topical modalities including tacrolimus and timolol. For complicated cases associated with KMP, sirolimus, systemic corticosteroids, ticlopidine, interferon, or vincristine are recommended. Some lesions may demonstrate spontaneous regression.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Cohen S et al. Dermatol Online J. 2019 Sep 15;25(9):13030/qt6pv254mc.
Lim YH et al. Pediatr Dermatol. 2019 Nov;36(6):963-4.
Prasuna A, Rao PN. Indian Dermatol Online J. 2015;6:266-8.
As the lesion was growing, getting more violaceous and indurated, a biopsy was performed. The biopsy showed multiple discrete lobules of dermal capillaries with slight extension into the superficial subcutis. Capillary lobules demonstrate the “cannonball-like” architecture often associated with tufted angioma, and some lobules showed bulging into adjacent thin-walled vessels. Spindled endothelial cells lining slit-like vessels were present in the mid dermis, although this comprises a minority of the lesion. The majority of the subcutis was uninvolved. The findings are overall most consistent with a tufted angioma.
Kaposiform hemangioendothelioma (KHE) has been considered given the presence of occasional slit-like vascular spaces; however, the lesion is predominantly superficial and therefore the lesion is best classified as tufted angioma. GLUT–1 staining was negative.
At the time of biopsy, blood work was ordered, which showed a normal complete blood count with normal number of platelets, slightly elevated D-dimer, and slightly low fibrinogen. Several repeat blood counts and coagulation tests once a week for a few weeks revealed no changes.
The patient was started on aspirin at a dose of 5 mg/kg per day. After a week on the medication the lesion was starting to get smaller and less red.
Tufted angiomas are a rare type of vascular tumor within the spectrum of kaposiform hemangioendotheliomas. Most cases present within the first year of life; some occur at birth. They usually present as papules, plaques, or erythematous, violaceous indurated nodules on the face, neck, trunk, and extremities. The lesions can also be present with hyperhidrosis and hypertrichosis. Clinically, the lesions will have to be differentiated from other vascular tumors such as infantile hemangiomas, congenital hemangiomas, and Kaposi’s sarcoma, as well as subcutaneous fat necrosis of the newborn, cellulitis, and nonaccidental trauma.
Pathogenesis of tufted angiomas is poorly understood. A recent case report found a somatic mutation on GNA14.This protein regulates Ras activity and modulates endothelial cell permeability and migration in response to FGF2 and VEGFA. The p.205L mutation causes activation of GNA14, which upregulates pERK-MAPK pathway, suggesting MAPK inhibition as a potential target for therapy. Clinically, tufted angioma can present in three patterns: uncomplicated tufted angioma (most common type); tufted angioma without thrombocytopenia but with chronic coagulopathy, as it was seen in our patient; and tufted angioma associated with Kasabach-Merritt phenomenon (KMP). KMP is characterized by thrombocytopenia in association with microangiopathic hemolytic anemia, consumptive coagulopathy, and enlarging vascular tumor. Treatment of uncomplicated tufted angioma will depend on symptomatology, size, and location of the lesion. Smaller lesions in noncosmetically sensitive areas can be treated with surgical excision. Cases that are not amenable to excision can be treated with aspirin. There are also reports of response to topical modalities including tacrolimus and timolol. For complicated cases associated with KMP, sirolimus, systemic corticosteroids, ticlopidine, interferon, or vincristine are recommended. Some lesions may demonstrate spontaneous regression.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Cohen S et al. Dermatol Online J. 2019 Sep 15;25(9):13030/qt6pv254mc.
Lim YH et al. Pediatr Dermatol. 2019 Nov;36(6):963-4.
Prasuna A, Rao PN. Indian Dermatol Online J. 2015;6:266-8.
A 35-day-old female was referred to our pediatric dermatology clinic for evaluation of a red lesion on the right arm. The lesion presented at about 4 days of life as a red plaque (image 1 at 8 days of life).
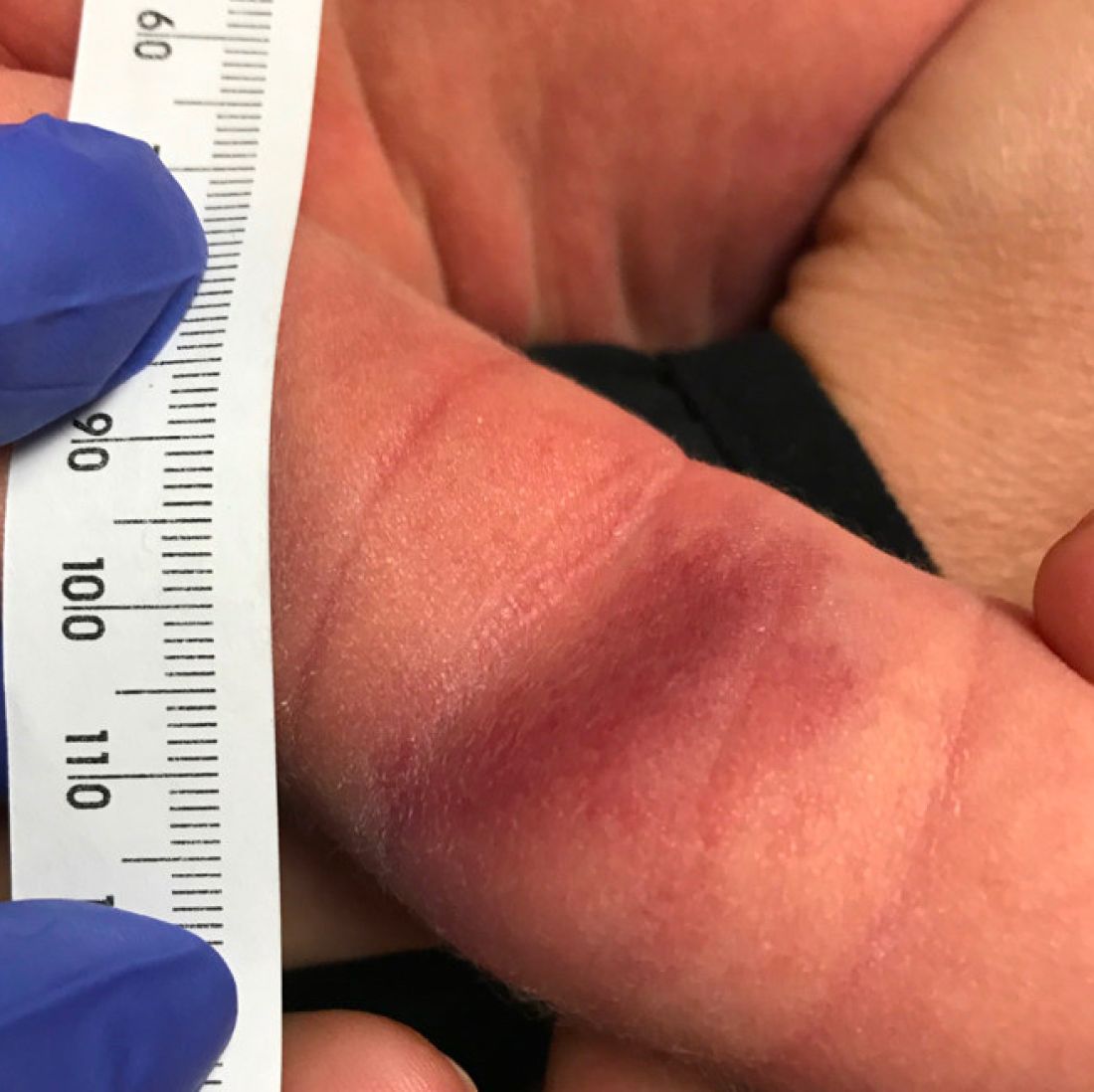
On the following days, the lesion started growing but it didn't seem to be tender or bothersome to the patient (image 2, at 35 days of life).
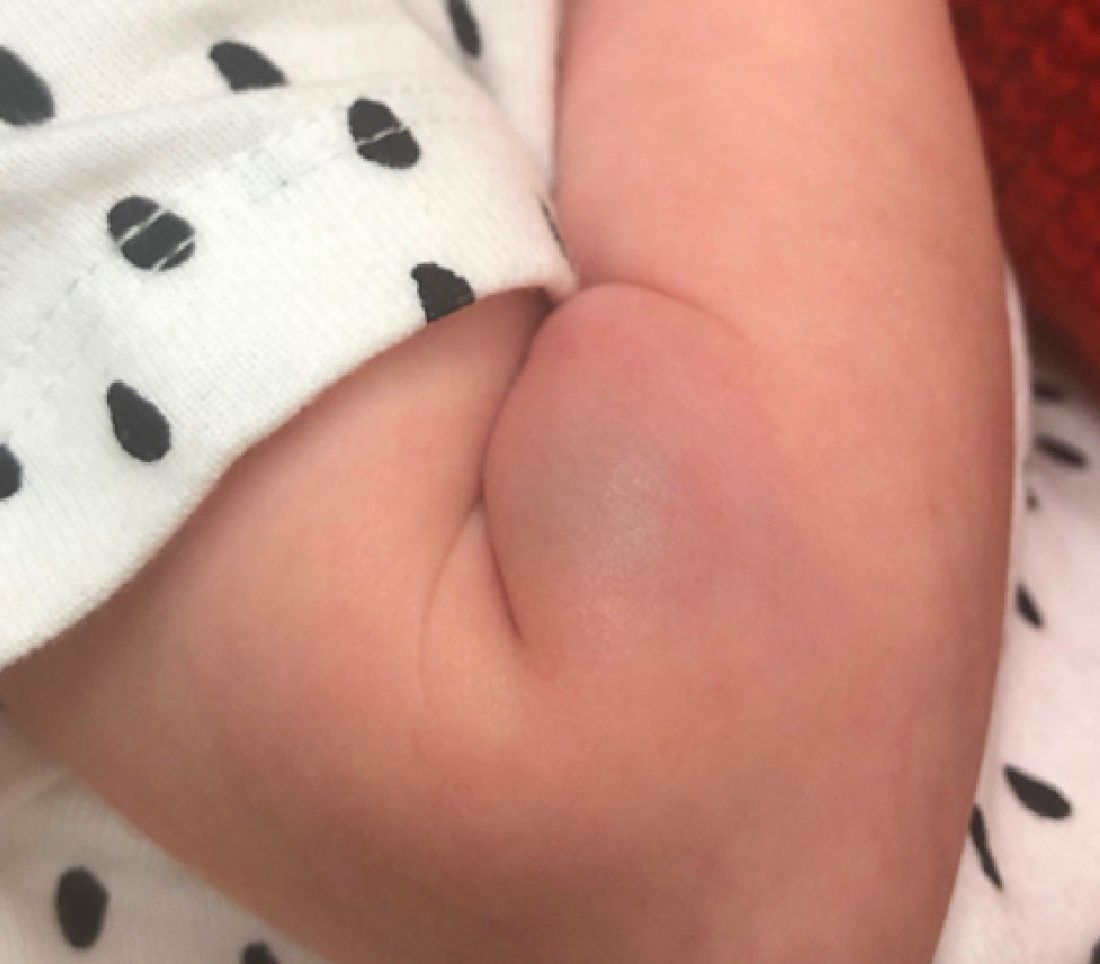
At a 2-week follow up the lesion was getting fuller and more violaceous. There was no history of fever and the lesion didn't appear tender to the touch.
She was born via normal spontaneous vaginal delivery. There were no complications and the mother received prenatal care.
On exam she had a red to violaceous nodule on the right arm (image 3 at 45 days of life).
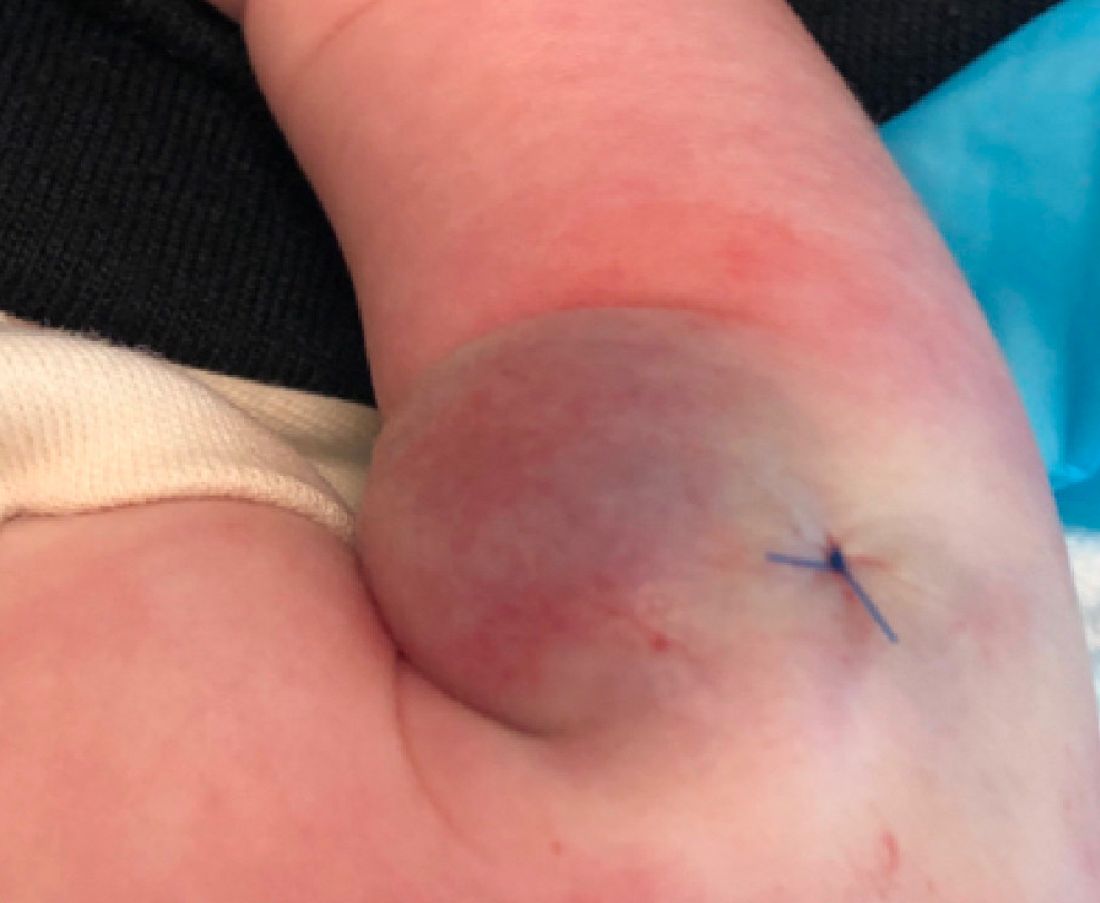
Filler complications involving vascular necrosis, vision changes on the rise
analysis showed.
“The ASDS estimates that 1.6 million soft tissue filler procedures were performed in 2019, a 78% increase from 2012,” presenting author Michelle Xiong, a 4th-year student at Brown University, Providence, R.I., said during a virtual abstract session at the annual meeting of the American Society for Dermatologic Surgery. “The popularity of dermal fillers continues to increase. With that, there is increasing concern of possible associated adverse events. Most concerning are those related to vascular occlusion.”
Under the supervision of senior author Kachiu C. Lee, MD, MPH, of Main Line Center for Laser Surgery in Ardmore, Pa., Ms. Xiong and colleagues analyzed the Food and Drug Administration’s Manufacturer and User Facility Device Experience (MAUDE) database of medical device–related adverse event reports, to better understand and characterize dermal filler-related complications. They limited the analysis to adverse events involving injectable fillers from January 2014 to December 2020 and determined the number of complications by type per year and reviewed reports to identify injection site locations. Next, they used the binomial test to compare the proportion of complication categories from 2014 through 2016 and from 2017 through 2020.
In all, 5,994 reports were identified during the 7-year study period. To evaluate trends over time, the researchers estimated the rate of complications per 100 reports each year. While the absolute number of reports increased over time, the rate of adverse events per 100 reports decreased, suggesting an overall improvement in safety.
When the researchers focused on complications involving vascular occlusion, they found that vascular necrosis accounted for 3.5% of all complications, compared with vision changes (1.5% of all complications), and stroke (0.3% of all complications). When comparing the years 2014-2016 with 2017-2020, there was a significant increase in adverse events involving vascular necrosis (0.9%; P = .018) and vision changes (0.94%; P = .001), but no significant difference in the number of reports of stroke (-0.1%; P = .409). “This highlights that serious complications like necrosis and vision changes have increased over time,” Ms. Xiong said.
Overall, the three most common injection sites involving necrosis and vision changes were the cheek, the nose, and the nasolabial fold. The cheek was the most common site associated with stroke. “These findings are similar to those of previous studies, further emphasizing that the nose, nasolabial fold, and cheek are possibly challenging injection sites,” she said.
“In general, as the face is a highly vascular area with many anastomoses, it’s especially important to be aware of facial anatomy when injecting. In addition to awareness of anatomy, injection techniques can influence vascular complications. Unfortunately, the event narratives in the MAUDE database did not go into detail about the procedural technique.”
Ms. Xiong said that as the popularity of dermal fillers continues to grow, “it’s important for providers to understand the possible adverse events, both to better counsel patients and to improve safety management. The proportion of serious complications such as vascular necrosis and vision changes have increased from 2014 to 2020. This highlights an increased need for training to better understand facial anatomy and to emphasize practice techniques to minimize risk.”
Dr. Lee acknowledged certain limitations of the study, including that “submission of adverse events to the MAUDE database are not verified or standardized,” she told this news organization.
“With the ever-increasing popularity of fillers, it is not surprising that the absolute number of complications is rising, but it is also reassuring to see that the overall ratio of complications per hundred reports is down,” said Lawrence J. Green, MD, clinical professor of dermatology at George Washington University, Washington, who was asked to comment on the study. “I would be curious to know what proportion of filler complications are due to non–core practitioners compared to dermatologists and plastic surgeons.”
The researchers reported having no financial disclosures.
Dr. Green disclosed that he is a speaker, consultant, or investigator for numerous pharmaceutical companies.
analysis showed.
“The ASDS estimates that 1.6 million soft tissue filler procedures were performed in 2019, a 78% increase from 2012,” presenting author Michelle Xiong, a 4th-year student at Brown University, Providence, R.I., said during a virtual abstract session at the annual meeting of the American Society for Dermatologic Surgery. “The popularity of dermal fillers continues to increase. With that, there is increasing concern of possible associated adverse events. Most concerning are those related to vascular occlusion.”
Under the supervision of senior author Kachiu C. Lee, MD, MPH, of Main Line Center for Laser Surgery in Ardmore, Pa., Ms. Xiong and colleagues analyzed the Food and Drug Administration’s Manufacturer and User Facility Device Experience (MAUDE) database of medical device–related adverse event reports, to better understand and characterize dermal filler-related complications. They limited the analysis to adverse events involving injectable fillers from January 2014 to December 2020 and determined the number of complications by type per year and reviewed reports to identify injection site locations. Next, they used the binomial test to compare the proportion of complication categories from 2014 through 2016 and from 2017 through 2020.
In all, 5,994 reports were identified during the 7-year study period. To evaluate trends over time, the researchers estimated the rate of complications per 100 reports each year. While the absolute number of reports increased over time, the rate of adverse events per 100 reports decreased, suggesting an overall improvement in safety.
When the researchers focused on complications involving vascular occlusion, they found that vascular necrosis accounted for 3.5% of all complications, compared with vision changes (1.5% of all complications), and stroke (0.3% of all complications). When comparing the years 2014-2016 with 2017-2020, there was a significant increase in adverse events involving vascular necrosis (0.9%; P = .018) and vision changes (0.94%; P = .001), but no significant difference in the number of reports of stroke (-0.1%; P = .409). “This highlights that serious complications like necrosis and vision changes have increased over time,” Ms. Xiong said.
Overall, the three most common injection sites involving necrosis and vision changes were the cheek, the nose, and the nasolabial fold. The cheek was the most common site associated with stroke. “These findings are similar to those of previous studies, further emphasizing that the nose, nasolabial fold, and cheek are possibly challenging injection sites,” she said.
“In general, as the face is a highly vascular area with many anastomoses, it’s especially important to be aware of facial anatomy when injecting. In addition to awareness of anatomy, injection techniques can influence vascular complications. Unfortunately, the event narratives in the MAUDE database did not go into detail about the procedural technique.”
Ms. Xiong said that as the popularity of dermal fillers continues to grow, “it’s important for providers to understand the possible adverse events, both to better counsel patients and to improve safety management. The proportion of serious complications such as vascular necrosis and vision changes have increased from 2014 to 2020. This highlights an increased need for training to better understand facial anatomy and to emphasize practice techniques to minimize risk.”
Dr. Lee acknowledged certain limitations of the study, including that “submission of adverse events to the MAUDE database are not verified or standardized,” she told this news organization.
“With the ever-increasing popularity of fillers, it is not surprising that the absolute number of complications is rising, but it is also reassuring to see that the overall ratio of complications per hundred reports is down,” said Lawrence J. Green, MD, clinical professor of dermatology at George Washington University, Washington, who was asked to comment on the study. “I would be curious to know what proportion of filler complications are due to non–core practitioners compared to dermatologists and plastic surgeons.”
The researchers reported having no financial disclosures.
Dr. Green disclosed that he is a speaker, consultant, or investigator for numerous pharmaceutical companies.
analysis showed.
“The ASDS estimates that 1.6 million soft tissue filler procedures were performed in 2019, a 78% increase from 2012,” presenting author Michelle Xiong, a 4th-year student at Brown University, Providence, R.I., said during a virtual abstract session at the annual meeting of the American Society for Dermatologic Surgery. “The popularity of dermal fillers continues to increase. With that, there is increasing concern of possible associated adverse events. Most concerning are those related to vascular occlusion.”
Under the supervision of senior author Kachiu C. Lee, MD, MPH, of Main Line Center for Laser Surgery in Ardmore, Pa., Ms. Xiong and colleagues analyzed the Food and Drug Administration’s Manufacturer and User Facility Device Experience (MAUDE) database of medical device–related adverse event reports, to better understand and characterize dermal filler-related complications. They limited the analysis to adverse events involving injectable fillers from January 2014 to December 2020 and determined the number of complications by type per year and reviewed reports to identify injection site locations. Next, they used the binomial test to compare the proportion of complication categories from 2014 through 2016 and from 2017 through 2020.
In all, 5,994 reports were identified during the 7-year study period. To evaluate trends over time, the researchers estimated the rate of complications per 100 reports each year. While the absolute number of reports increased over time, the rate of adverse events per 100 reports decreased, suggesting an overall improvement in safety.
When the researchers focused on complications involving vascular occlusion, they found that vascular necrosis accounted for 3.5% of all complications, compared with vision changes (1.5% of all complications), and stroke (0.3% of all complications). When comparing the years 2014-2016 with 2017-2020, there was a significant increase in adverse events involving vascular necrosis (0.9%; P = .018) and vision changes (0.94%; P = .001), but no significant difference in the number of reports of stroke (-0.1%; P = .409). “This highlights that serious complications like necrosis and vision changes have increased over time,” Ms. Xiong said.
Overall, the three most common injection sites involving necrosis and vision changes were the cheek, the nose, and the nasolabial fold. The cheek was the most common site associated with stroke. “These findings are similar to those of previous studies, further emphasizing that the nose, nasolabial fold, and cheek are possibly challenging injection sites,” she said.
“In general, as the face is a highly vascular area with many anastomoses, it’s especially important to be aware of facial anatomy when injecting. In addition to awareness of anatomy, injection techniques can influence vascular complications. Unfortunately, the event narratives in the MAUDE database did not go into detail about the procedural technique.”
Ms. Xiong said that as the popularity of dermal fillers continues to grow, “it’s important for providers to understand the possible adverse events, both to better counsel patients and to improve safety management. The proportion of serious complications such as vascular necrosis and vision changes have increased from 2014 to 2020. This highlights an increased need for training to better understand facial anatomy and to emphasize practice techniques to minimize risk.”
Dr. Lee acknowledged certain limitations of the study, including that “submission of adverse events to the MAUDE database are not verified or standardized,” she told this news organization.
“With the ever-increasing popularity of fillers, it is not surprising that the absolute number of complications is rising, but it is also reassuring to see that the overall ratio of complications per hundred reports is down,” said Lawrence J. Green, MD, clinical professor of dermatology at George Washington University, Washington, who was asked to comment on the study. “I would be curious to know what proportion of filler complications are due to non–core practitioners compared to dermatologists and plastic surgeons.”
The researchers reported having no financial disclosures.
Dr. Green disclosed that he is a speaker, consultant, or investigator for numerous pharmaceutical companies.
FROM ASDS 2021
Omicron may require fourth vaccine dose, Pfizer says
, Pfizer officials said on Dec. 8.
The standard two doses may be less effective against the variant, the company announced earlier in the day, and a booster dose increases neutralizing antibodies.
But the timeline might need to be moved up for a fourth dose. Previously, Pfizer CEO Albert Bourla, PhD, said another dose might be needed about a year after a third shot. Now the company’s scientists believe that a fourth shot, which targets the Omicron variant, could be required sooner.
“With Omicron, we need to wait and see because we have very little information. We may need it faster,” Dr. Bourla said on CNBC’s Squawk Box.
“But for right now, the most important thing is that we have winter in front of us,” he said. “From a healthcare perspective, it is important to understand that we need to be well-protected to go through the winter.”
A third dose should provide protection throughout the winter, Dr. Bourla said. That may buy time until the early spring to develop new shots that target Omicron, which Pfizer could have ready by March, according to Bloomberg News.
As of the afternoon of Dec. 8, 43 people in 19 states had tested positive for the Omicron variant, according to The Associated Press. More than 75% had been vaccinated, and a third had had booster shots. About a third had traveled internationally.
Nearly all of them have had mild symptoms so far, the AP reported, with the most common symptoms being a cough, congestion, and fatigue. One person has been hospitalized, but no deaths have been reported so far.
The CDC is still trying to determine how the Omicron variant may affect the course of the pandemic and whether the strain is more contagious or causes more severe disease.
“What we generally know is the more mutations a variant has, the higher level you need your immunity to be,” Rochelle Walensky, MD, director of the CDC, told the AP.
“We want to make sure we bolster everybody’s immunity,” she said. “And that’s really what motivated the decision to expand our guidance [on boosters for all adults].”
The Omicron variant has been reported in 57 countries so far, World Health Organization officials reported Dec. 8, and they expect that number to continue growing.
“Certain features of Omicron, including its global spread and large number of mutations, suggest it could have a major impact on the course of the pandemic. Exactly what that impact will be is still difficult to know,” Tedros Adhanom Ghebreyesus, PhD, the World Health Organization’s director-general, said during a media briefing.
Several studies suggest that Omicron leads to a rapid increase in transmission, he said, though scientists are still trying to understand whether it can “outcompete Delta.” Data from South Africa also suggests a higher risk of reinfection with Omicron, though it appears to cause milder disease than Delta, he noted.
“Even though we still need answers to some crucial questions, we are not defenseless against Omicron or Delta,” he said. “The steps countries take today, and in the coming days and weeks, will determine how Omicron unfolds.”
A version of this article first appeared on WebMD.com.
, Pfizer officials said on Dec. 8.
The standard two doses may be less effective against the variant, the company announced earlier in the day, and a booster dose increases neutralizing antibodies.
But the timeline might need to be moved up for a fourth dose. Previously, Pfizer CEO Albert Bourla, PhD, said another dose might be needed about a year after a third shot. Now the company’s scientists believe that a fourth shot, which targets the Omicron variant, could be required sooner.
“With Omicron, we need to wait and see because we have very little information. We may need it faster,” Dr. Bourla said on CNBC’s Squawk Box.
“But for right now, the most important thing is that we have winter in front of us,” he said. “From a healthcare perspective, it is important to understand that we need to be well-protected to go through the winter.”
A third dose should provide protection throughout the winter, Dr. Bourla said. That may buy time until the early spring to develop new shots that target Omicron, which Pfizer could have ready by March, according to Bloomberg News.
As of the afternoon of Dec. 8, 43 people in 19 states had tested positive for the Omicron variant, according to The Associated Press. More than 75% had been vaccinated, and a third had had booster shots. About a third had traveled internationally.
Nearly all of them have had mild symptoms so far, the AP reported, with the most common symptoms being a cough, congestion, and fatigue. One person has been hospitalized, but no deaths have been reported so far.
The CDC is still trying to determine how the Omicron variant may affect the course of the pandemic and whether the strain is more contagious or causes more severe disease.
“What we generally know is the more mutations a variant has, the higher level you need your immunity to be,” Rochelle Walensky, MD, director of the CDC, told the AP.
“We want to make sure we bolster everybody’s immunity,” she said. “And that’s really what motivated the decision to expand our guidance [on boosters for all adults].”
The Omicron variant has been reported in 57 countries so far, World Health Organization officials reported Dec. 8, and they expect that number to continue growing.
“Certain features of Omicron, including its global spread and large number of mutations, suggest it could have a major impact on the course of the pandemic. Exactly what that impact will be is still difficult to know,” Tedros Adhanom Ghebreyesus, PhD, the World Health Organization’s director-general, said during a media briefing.
Several studies suggest that Omicron leads to a rapid increase in transmission, he said, though scientists are still trying to understand whether it can “outcompete Delta.” Data from South Africa also suggests a higher risk of reinfection with Omicron, though it appears to cause milder disease than Delta, he noted.
“Even though we still need answers to some crucial questions, we are not defenseless against Omicron or Delta,” he said. “The steps countries take today, and in the coming days and weeks, will determine how Omicron unfolds.”
A version of this article first appeared on WebMD.com.
, Pfizer officials said on Dec. 8.
The standard two doses may be less effective against the variant, the company announced earlier in the day, and a booster dose increases neutralizing antibodies.
But the timeline might need to be moved up for a fourth dose. Previously, Pfizer CEO Albert Bourla, PhD, said another dose might be needed about a year after a third shot. Now the company’s scientists believe that a fourth shot, which targets the Omicron variant, could be required sooner.
“With Omicron, we need to wait and see because we have very little information. We may need it faster,” Dr. Bourla said on CNBC’s Squawk Box.
“But for right now, the most important thing is that we have winter in front of us,” he said. “From a healthcare perspective, it is important to understand that we need to be well-protected to go through the winter.”
A third dose should provide protection throughout the winter, Dr. Bourla said. That may buy time until the early spring to develop new shots that target Omicron, which Pfizer could have ready by March, according to Bloomberg News.
As of the afternoon of Dec. 8, 43 people in 19 states had tested positive for the Omicron variant, according to The Associated Press. More than 75% had been vaccinated, and a third had had booster shots. About a third had traveled internationally.
Nearly all of them have had mild symptoms so far, the AP reported, with the most common symptoms being a cough, congestion, and fatigue. One person has been hospitalized, but no deaths have been reported so far.
The CDC is still trying to determine how the Omicron variant may affect the course of the pandemic and whether the strain is more contagious or causes more severe disease.
“What we generally know is the more mutations a variant has, the higher level you need your immunity to be,” Rochelle Walensky, MD, director of the CDC, told the AP.
“We want to make sure we bolster everybody’s immunity,” she said. “And that’s really what motivated the decision to expand our guidance [on boosters for all adults].”
The Omicron variant has been reported in 57 countries so far, World Health Organization officials reported Dec. 8, and they expect that number to continue growing.
“Certain features of Omicron, including its global spread and large number of mutations, suggest it could have a major impact on the course of the pandemic. Exactly what that impact will be is still difficult to know,” Tedros Adhanom Ghebreyesus, PhD, the World Health Organization’s director-general, said during a media briefing.
Several studies suggest that Omicron leads to a rapid increase in transmission, he said, though scientists are still trying to understand whether it can “outcompete Delta.” Data from South Africa also suggests a higher risk of reinfection with Omicron, though it appears to cause milder disease than Delta, he noted.
“Even though we still need answers to some crucial questions, we are not defenseless against Omicron or Delta,” he said. “The steps countries take today, and in the coming days and weeks, will determine how Omicron unfolds.”
A version of this article first appeared on WebMD.com.
Medical board stops warning docs against giving false COVID information
Under pressure from Republican state lawmakers, t
The board’s 7-3 vote on December 7 to delete the statement followed repeated threats by a powerful state House Republican to dissolve the board and appoint all new members if it did not immediately take it down.
The Tennessee board’s statement was a verbatim restatement of a warning to physicians issued by the Federation of State Medical Boards in July. The federation cited a “dramatic increase” in dissemination of misinformation and disinformation about the COVID-19 vaccine by physicians. It said that’s dangerous because physicians enjoy a high degree of public credibility.
Across the country, state medical licensing boards and state and national medical associations and specialty boards are struggling with how to respond to scientifically baseless public statements about COVID-19 by some physicians, which they say are increasing public confusion, political conflict, and preventable illnesses and deaths.
There have been only a small number of disciplinary actions by medical boards against physicians for spreading false COVID-19 information. Critics say the boards have been weak in responding to these dangerous violations of medical standards. As an example, they cite the State Medical Board of Ohio’s September renewal of the medical license of Sherri Tenpenny, DO, who had previously testified before Ohio lawmakers that COVID-19 vaccines magnetize their recipients and “interface” with cell phone towers.
“I’m not satisfied with what medical boards have done, and we are ramping up our efforts to press the boards to hold these physicians accountable,” said Nick Sawyer, MD, an emergency physician in Sacramento, Calif., who heads a group of healthcare professionals called No License for Disinformation.
Still, Tennessee board members insisted that the board’s policy of disciplining physicians who disseminate false information about COVID-19 vaccinations remains in effect, because state law empowers the board to take action against doctors whose unprofessional behavior endangers the public.
“COVID misinformation and disinformation has caused undue loss of life and jobs and other incalculable loss in our society,” said Melanie Blake, MD, MBA, a Chattanooga internist who’s president of the board. “Physicians have a responsibility to uphold their oath and put forward consensus-driven medical principles.”
But state Rep. John Ragan, the Republican co-chairman of the Joint Government Operations Committee, told the Tennessean newspaper that deleting the statement from the board’s website was equivalent to rescinding the policy. Ragan, who identifies himself as a business consultant and retired Air Force pilot, did not respond to a request for comment for this article.
Blake acknowledged that removing the statement from the board’s website has the potential to confuse Tennessee physicians. And the pressure from GOP lawmakers, who overwhelmingly control the Tennessee legislature, could discourage investigations and disciplinary actions against physicians who allegedly spread COVID-19 misinformation, she added. “It’s hard for me to answer whether this puts a chill on us,” she said.
In September, the Tennessee board, besides approving the general statement that physicians who spread COVID-19 disinformation could face licensure action, also directed the State Department of Health to prioritize investigations of physicians who spread outrageous claims. The board cited statements such as the vaccines are poisonous, cause infertility, contain microchips, or magnetize the body.
In response, the Tennessee General Assembly passed a bill in late October prohibiting the board from implementing any disciplinary process regarding the prescribing of “medication for COVID-19” without review and approval by Ragan’s committee. It’s not clear whether that language covers vaccines.
Last summer, in a similar move, Ragan threatened to dissolve the State Department of Health because its top vaccination official wrote a letter to medical providers explaining that state law allowed them to give COVID-19 vaccinations to minors older than 14 without parental consent. That official, Michelle Fiscus, MD, was fired in July.
Republican Sen. Richard Briggs, MD, a cardiothoracic surgeon who voted against the October legislation affecting COVID-related disciplinary actions, criticized his GOP colleagues’ interference in the medical board’s licensure decisions. “The mission of the board is to protect the health and safety of Tennessee citizens, and this was in complete conflict with that mission,” he said.
The Federation of State Medical Boards similarly condemned the Tennessee lawmakers’ moves. “The FSMB strongly opposes restricting a board’s authority to evaluate the standard of care and assess potential risk for patient harm,” a spokesman said. “Any interference, politically motivated or otherwise, is unhelpful and dangerous.”
But Arthur Caplan, PhD, a professor of bioethics at NYU School of Medicine, doubts that state medical boards are up to the task of policing disinformation spread by physicians. That’s because they ultimately are under the control of elected state officials, who may force the boards to base policy on ideology rather than science.
He said medical board members in Florida and another GOP-controlled state have told him they do not want to pursue disciplinary actions against physicians for COVID-19 misinformation for fear of political backlash.
Michele Heisler, MD, medical director of Physicians for Human Rights, agreed that the Tennessee situation highlights the looming political threat to the independence of state medical boards. She urged other medical organizations, particularly medical specialty boards, to step in.
“As a profession, we need to take a stance against this,” said Heisler, who’s a professor of internal medicine and public health at the University of Michigan. “Our credibility as physicians is at stake.”
A version of this article first appeared on Medscape.com.
Under pressure from Republican state lawmakers, t
The board’s 7-3 vote on December 7 to delete the statement followed repeated threats by a powerful state House Republican to dissolve the board and appoint all new members if it did not immediately take it down.
The Tennessee board’s statement was a verbatim restatement of a warning to physicians issued by the Federation of State Medical Boards in July. The federation cited a “dramatic increase” in dissemination of misinformation and disinformation about the COVID-19 vaccine by physicians. It said that’s dangerous because physicians enjoy a high degree of public credibility.
Across the country, state medical licensing boards and state and national medical associations and specialty boards are struggling with how to respond to scientifically baseless public statements about COVID-19 by some physicians, which they say are increasing public confusion, political conflict, and preventable illnesses and deaths.
There have been only a small number of disciplinary actions by medical boards against physicians for spreading false COVID-19 information. Critics say the boards have been weak in responding to these dangerous violations of medical standards. As an example, they cite the State Medical Board of Ohio’s September renewal of the medical license of Sherri Tenpenny, DO, who had previously testified before Ohio lawmakers that COVID-19 vaccines magnetize their recipients and “interface” with cell phone towers.
“I’m not satisfied with what medical boards have done, and we are ramping up our efforts to press the boards to hold these physicians accountable,” said Nick Sawyer, MD, an emergency physician in Sacramento, Calif., who heads a group of healthcare professionals called No License for Disinformation.
Still, Tennessee board members insisted that the board’s policy of disciplining physicians who disseminate false information about COVID-19 vaccinations remains in effect, because state law empowers the board to take action against doctors whose unprofessional behavior endangers the public.
“COVID misinformation and disinformation has caused undue loss of life and jobs and other incalculable loss in our society,” said Melanie Blake, MD, MBA, a Chattanooga internist who’s president of the board. “Physicians have a responsibility to uphold their oath and put forward consensus-driven medical principles.”
But state Rep. John Ragan, the Republican co-chairman of the Joint Government Operations Committee, told the Tennessean newspaper that deleting the statement from the board’s website was equivalent to rescinding the policy. Ragan, who identifies himself as a business consultant and retired Air Force pilot, did not respond to a request for comment for this article.
Blake acknowledged that removing the statement from the board’s website has the potential to confuse Tennessee physicians. And the pressure from GOP lawmakers, who overwhelmingly control the Tennessee legislature, could discourage investigations and disciplinary actions against physicians who allegedly spread COVID-19 misinformation, she added. “It’s hard for me to answer whether this puts a chill on us,” she said.
In September, the Tennessee board, besides approving the general statement that physicians who spread COVID-19 disinformation could face licensure action, also directed the State Department of Health to prioritize investigations of physicians who spread outrageous claims. The board cited statements such as the vaccines are poisonous, cause infertility, contain microchips, or magnetize the body.
In response, the Tennessee General Assembly passed a bill in late October prohibiting the board from implementing any disciplinary process regarding the prescribing of “medication for COVID-19” without review and approval by Ragan’s committee. It’s not clear whether that language covers vaccines.
Last summer, in a similar move, Ragan threatened to dissolve the State Department of Health because its top vaccination official wrote a letter to medical providers explaining that state law allowed them to give COVID-19 vaccinations to minors older than 14 without parental consent. That official, Michelle Fiscus, MD, was fired in July.
Republican Sen. Richard Briggs, MD, a cardiothoracic surgeon who voted against the October legislation affecting COVID-related disciplinary actions, criticized his GOP colleagues’ interference in the medical board’s licensure decisions. “The mission of the board is to protect the health and safety of Tennessee citizens, and this was in complete conflict with that mission,” he said.
The Federation of State Medical Boards similarly condemned the Tennessee lawmakers’ moves. “The FSMB strongly opposes restricting a board’s authority to evaluate the standard of care and assess potential risk for patient harm,” a spokesman said. “Any interference, politically motivated or otherwise, is unhelpful and dangerous.”
But Arthur Caplan, PhD, a professor of bioethics at NYU School of Medicine, doubts that state medical boards are up to the task of policing disinformation spread by physicians. That’s because they ultimately are under the control of elected state officials, who may force the boards to base policy on ideology rather than science.
He said medical board members in Florida and another GOP-controlled state have told him they do not want to pursue disciplinary actions against physicians for COVID-19 misinformation for fear of political backlash.
Michele Heisler, MD, medical director of Physicians for Human Rights, agreed that the Tennessee situation highlights the looming political threat to the independence of state medical boards. She urged other medical organizations, particularly medical specialty boards, to step in.
“As a profession, we need to take a stance against this,” said Heisler, who’s a professor of internal medicine and public health at the University of Michigan. “Our credibility as physicians is at stake.”
A version of this article first appeared on Medscape.com.
Under pressure from Republican state lawmakers, t
The board’s 7-3 vote on December 7 to delete the statement followed repeated threats by a powerful state House Republican to dissolve the board and appoint all new members if it did not immediately take it down.
The Tennessee board’s statement was a verbatim restatement of a warning to physicians issued by the Federation of State Medical Boards in July. The federation cited a “dramatic increase” in dissemination of misinformation and disinformation about the COVID-19 vaccine by physicians. It said that’s dangerous because physicians enjoy a high degree of public credibility.
Across the country, state medical licensing boards and state and national medical associations and specialty boards are struggling with how to respond to scientifically baseless public statements about COVID-19 by some physicians, which they say are increasing public confusion, political conflict, and preventable illnesses and deaths.
There have been only a small number of disciplinary actions by medical boards against physicians for spreading false COVID-19 information. Critics say the boards have been weak in responding to these dangerous violations of medical standards. As an example, they cite the State Medical Board of Ohio’s September renewal of the medical license of Sherri Tenpenny, DO, who had previously testified before Ohio lawmakers that COVID-19 vaccines magnetize their recipients and “interface” with cell phone towers.
“I’m not satisfied with what medical boards have done, and we are ramping up our efforts to press the boards to hold these physicians accountable,” said Nick Sawyer, MD, an emergency physician in Sacramento, Calif., who heads a group of healthcare professionals called No License for Disinformation.
Still, Tennessee board members insisted that the board’s policy of disciplining physicians who disseminate false information about COVID-19 vaccinations remains in effect, because state law empowers the board to take action against doctors whose unprofessional behavior endangers the public.
“COVID misinformation and disinformation has caused undue loss of life and jobs and other incalculable loss in our society,” said Melanie Blake, MD, MBA, a Chattanooga internist who’s president of the board. “Physicians have a responsibility to uphold their oath and put forward consensus-driven medical principles.”
But state Rep. John Ragan, the Republican co-chairman of the Joint Government Operations Committee, told the Tennessean newspaper that deleting the statement from the board’s website was equivalent to rescinding the policy. Ragan, who identifies himself as a business consultant and retired Air Force pilot, did not respond to a request for comment for this article.
Blake acknowledged that removing the statement from the board’s website has the potential to confuse Tennessee physicians. And the pressure from GOP lawmakers, who overwhelmingly control the Tennessee legislature, could discourage investigations and disciplinary actions against physicians who allegedly spread COVID-19 misinformation, she added. “It’s hard for me to answer whether this puts a chill on us,” she said.
In September, the Tennessee board, besides approving the general statement that physicians who spread COVID-19 disinformation could face licensure action, also directed the State Department of Health to prioritize investigations of physicians who spread outrageous claims. The board cited statements such as the vaccines are poisonous, cause infertility, contain microchips, or magnetize the body.
In response, the Tennessee General Assembly passed a bill in late October prohibiting the board from implementing any disciplinary process regarding the prescribing of “medication for COVID-19” without review and approval by Ragan’s committee. It’s not clear whether that language covers vaccines.
Last summer, in a similar move, Ragan threatened to dissolve the State Department of Health because its top vaccination official wrote a letter to medical providers explaining that state law allowed them to give COVID-19 vaccinations to minors older than 14 without parental consent. That official, Michelle Fiscus, MD, was fired in July.
Republican Sen. Richard Briggs, MD, a cardiothoracic surgeon who voted against the October legislation affecting COVID-related disciplinary actions, criticized his GOP colleagues’ interference in the medical board’s licensure decisions. “The mission of the board is to protect the health and safety of Tennessee citizens, and this was in complete conflict with that mission,” he said.
The Federation of State Medical Boards similarly condemned the Tennessee lawmakers’ moves. “The FSMB strongly opposes restricting a board’s authority to evaluate the standard of care and assess potential risk for patient harm,” a spokesman said. “Any interference, politically motivated or otherwise, is unhelpful and dangerous.”
But Arthur Caplan, PhD, a professor of bioethics at NYU School of Medicine, doubts that state medical boards are up to the task of policing disinformation spread by physicians. That’s because they ultimately are under the control of elected state officials, who may force the boards to base policy on ideology rather than science.
He said medical board members in Florida and another GOP-controlled state have told him they do not want to pursue disciplinary actions against physicians for COVID-19 misinformation for fear of political backlash.
Michele Heisler, MD, medical director of Physicians for Human Rights, agreed that the Tennessee situation highlights the looming political threat to the independence of state medical boards. She urged other medical organizations, particularly medical specialty boards, to step in.
“As a profession, we need to take a stance against this,” said Heisler, who’s a professor of internal medicine and public health at the University of Michigan. “Our credibility as physicians is at stake.”
A version of this article first appeared on Medscape.com.
AMA, hospital group sue federal government over surprise billing law
which tilts toward using prevailing rates paid for services.
The American Hospital Association and American Medical Association said they will ask the U.S. District Court for the District of Columbia to try to prevent implementation of certain provisions of new federal rules on surprise bills. This court is often a venue for fights over federal rules. Also joining the suit are Nevada-based Renown Health, UMass Memorial Health, and two physicians based in North Carolina, AHA and AMA said.
Federal agencies, including the Department of Health & Human Services, in September had unveiled the rule on surprise medical bills that will take effect Jan. 1.
Under this rule, a key benchmark for payment disputes would be the qualifying payment amount (QPA), which is pegged to median contracted rates. In the dispute-resolution process outlined in the rule, there is a presumption that the QPA is the appropriate out-of-network rate.
The rule allows for exceptions in which the independent mediating organization handling the payment dispute resolution has “credible information” as to why the QPA is materially different from the appropriate out-of-network rate.
In the view of the federal agencies that issued the rule, this approach “encourages predictable outcomes,” which likely would reduce the number of disputes that go through the resolution process while also “providing equitable and clear standards” for cases to appropriately deviate from QPA. HHS was joined in issuing the rule by the Treasury and Labor Departments and the Office of Personnel Management.
AMA and AHA disagree with their view, seeing this approach as a boon for insurers at the expense of physicians and hospitals.
In a press release, they said the rule’s approach to surprise billing would “all but ensure that hospitals, physicians, and other providers will routinely be undercompensated by commercial insurers, and patients will have fewer choices for access to in-network services.”
The rule is part of the implementation of a federal law passed in December 2020, known as the No Surprises Act. In their statement, AHA and AMA said their legal challenge would not prevent “core patient protections’’ of that law from moving forward.
“No patient should fear receiving a surprise medical bill,” Rick Pollack, AHA president and chief executive, said in the statement. “That is why hospitals and health systems supported the No Surprises Act to protect patients and keep them out of the middle of disputes between providers and insurers. Congress carefully crafted the law with a balanced, patient-friendly approach and it should be implemented as intended.”
AMA President Gerald E. Harmon, MD, added the approach used in the rule on surprise billing could create “an unsustainable situation for physicians.”
“Our legal challenge urges regulators to ensure there is a fair and meaningful process to resolve disputes between health care providers and insurance companies,” Dr. Harmon said.
AHA and AMA included with their statement a link to a November letter from more than 150 members of Congress, who also objected to the approach taken in designing the independent dispute-resolution (IDR) process.
“This directive establishes a de facto benchmark rate, making the median in-network rate the default factor considered in the IDR process. This approach is contrary to statute and could incentivize insurance companies to set artificially low payment rates, which would narrow provider networks and jeopardize patient access to care – the exact opposite of the goal of the law,” wrote the members of Congress, including Rep. Raul Ruiz, MD, a California Democrat, and Rep. Larry Bucshon, MD, an Indiana Republican.
A version of this article first appeared on Medscape.com.
which tilts toward using prevailing rates paid for services.
The American Hospital Association and American Medical Association said they will ask the U.S. District Court for the District of Columbia to try to prevent implementation of certain provisions of new federal rules on surprise bills. This court is often a venue for fights over federal rules. Also joining the suit are Nevada-based Renown Health, UMass Memorial Health, and two physicians based in North Carolina, AHA and AMA said.
Federal agencies, including the Department of Health & Human Services, in September had unveiled the rule on surprise medical bills that will take effect Jan. 1.
Under this rule, a key benchmark for payment disputes would be the qualifying payment amount (QPA), which is pegged to median contracted rates. In the dispute-resolution process outlined in the rule, there is a presumption that the QPA is the appropriate out-of-network rate.
The rule allows for exceptions in which the independent mediating organization handling the payment dispute resolution has “credible information” as to why the QPA is materially different from the appropriate out-of-network rate.
In the view of the federal agencies that issued the rule, this approach “encourages predictable outcomes,” which likely would reduce the number of disputes that go through the resolution process while also “providing equitable and clear standards” for cases to appropriately deviate from QPA. HHS was joined in issuing the rule by the Treasury and Labor Departments and the Office of Personnel Management.
AMA and AHA disagree with their view, seeing this approach as a boon for insurers at the expense of physicians and hospitals.
In a press release, they said the rule’s approach to surprise billing would “all but ensure that hospitals, physicians, and other providers will routinely be undercompensated by commercial insurers, and patients will have fewer choices for access to in-network services.”
The rule is part of the implementation of a federal law passed in December 2020, known as the No Surprises Act. In their statement, AHA and AMA said their legal challenge would not prevent “core patient protections’’ of that law from moving forward.
“No patient should fear receiving a surprise medical bill,” Rick Pollack, AHA president and chief executive, said in the statement. “That is why hospitals and health systems supported the No Surprises Act to protect patients and keep them out of the middle of disputes between providers and insurers. Congress carefully crafted the law with a balanced, patient-friendly approach and it should be implemented as intended.”
AMA President Gerald E. Harmon, MD, added the approach used in the rule on surprise billing could create “an unsustainable situation for physicians.”
“Our legal challenge urges regulators to ensure there is a fair and meaningful process to resolve disputes between health care providers and insurance companies,” Dr. Harmon said.
AHA and AMA included with their statement a link to a November letter from more than 150 members of Congress, who also objected to the approach taken in designing the independent dispute-resolution (IDR) process.
“This directive establishes a de facto benchmark rate, making the median in-network rate the default factor considered in the IDR process. This approach is contrary to statute and could incentivize insurance companies to set artificially low payment rates, which would narrow provider networks and jeopardize patient access to care – the exact opposite of the goal of the law,” wrote the members of Congress, including Rep. Raul Ruiz, MD, a California Democrat, and Rep. Larry Bucshon, MD, an Indiana Republican.
A version of this article first appeared on Medscape.com.
which tilts toward using prevailing rates paid for services.
The American Hospital Association and American Medical Association said they will ask the U.S. District Court for the District of Columbia to try to prevent implementation of certain provisions of new federal rules on surprise bills. This court is often a venue for fights over federal rules. Also joining the suit are Nevada-based Renown Health, UMass Memorial Health, and two physicians based in North Carolina, AHA and AMA said.
Federal agencies, including the Department of Health & Human Services, in September had unveiled the rule on surprise medical bills that will take effect Jan. 1.
Under this rule, a key benchmark for payment disputes would be the qualifying payment amount (QPA), which is pegged to median contracted rates. In the dispute-resolution process outlined in the rule, there is a presumption that the QPA is the appropriate out-of-network rate.
The rule allows for exceptions in which the independent mediating organization handling the payment dispute resolution has “credible information” as to why the QPA is materially different from the appropriate out-of-network rate.
In the view of the federal agencies that issued the rule, this approach “encourages predictable outcomes,” which likely would reduce the number of disputes that go through the resolution process while also “providing equitable and clear standards” for cases to appropriately deviate from QPA. HHS was joined in issuing the rule by the Treasury and Labor Departments and the Office of Personnel Management.
AMA and AHA disagree with their view, seeing this approach as a boon for insurers at the expense of physicians and hospitals.
In a press release, they said the rule’s approach to surprise billing would “all but ensure that hospitals, physicians, and other providers will routinely be undercompensated by commercial insurers, and patients will have fewer choices for access to in-network services.”
The rule is part of the implementation of a federal law passed in December 2020, known as the No Surprises Act. In their statement, AHA and AMA said their legal challenge would not prevent “core patient protections’’ of that law from moving forward.
“No patient should fear receiving a surprise medical bill,” Rick Pollack, AHA president and chief executive, said in the statement. “That is why hospitals and health systems supported the No Surprises Act to protect patients and keep them out of the middle of disputes between providers and insurers. Congress carefully crafted the law with a balanced, patient-friendly approach and it should be implemented as intended.”
AMA President Gerald E. Harmon, MD, added the approach used in the rule on surprise billing could create “an unsustainable situation for physicians.”
“Our legal challenge urges regulators to ensure there is a fair and meaningful process to resolve disputes between health care providers and insurance companies,” Dr. Harmon said.
AHA and AMA included with their statement a link to a November letter from more than 150 members of Congress, who also objected to the approach taken in designing the independent dispute-resolution (IDR) process.
“This directive establishes a de facto benchmark rate, making the median in-network rate the default factor considered in the IDR process. This approach is contrary to statute and could incentivize insurance companies to set artificially low payment rates, which would narrow provider networks and jeopardize patient access to care – the exact opposite of the goal of the law,” wrote the members of Congress, including Rep. Raul Ruiz, MD, a California Democrat, and Rep. Larry Bucshon, MD, an Indiana Republican.
A version of this article first appeared on Medscape.com.