User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Indiana cardiologist faces multiple malpractice complaints
alleging he performed unnecessary cardiac procedures that led to physical and emotional harm.
The medical records for one patient, 70-year-old John Pflum, of Noblesville, Ind., show that Edward Harlamert, MD, performed 44 heart catheterizations and inserted at least 41 stents between 2004 and 2013, according to an investigation by WTHR 13News in Indianapolis that was published Dec. 14.
The news outlet asked four cardiologists to review and comment on John Pflum’s medical records.
“There is not a single scenario I can think of where doing this level of stents and angiograms would be justified or make sense. I have never seen this happen in the course of my medical training or my medical career,” Payal Kohli, MD, cardiologist and medical director of Cherry Creek Heart in Denver, told 13News.
Sunil Rao, MD, director of interventional cardiology at NYU Langone Health and president of the Society for Cardiovascular Angioplasty and Interventions, who also reviewed Mr. Pflum’s medical records for 13News, said he’s “never seen a patient who has gotten this many procedures.”
Dr. Rao said that on the basis of what he saw in the records and in the images, there were several deviations from the standard of care.
Two other independent cardiologists who spoke with 13News voiced similar opinions.
Mr. Pflum was “getting cathed almost every month. That’s not how it’s done,” Morton Rinder, MD, an interventional cardiologist at St. Luke’s Hospital near St. Louis, told 13News.
Dr. Rinder has been hired as a medical consultant for the attorneys who filed Mr. Pflum’s malpractice complaint against Dr. Harlamert.
Cardiologists who reviewed the catheterization films for 13News said some of Mr. Pflum’s heart blockages met the 70% threshold to warrant consideration of a stent, while others clearly did not. In-stent restenosis occurred in several of the implanted stents, requiring a second open heart surgery.
In a statement, Dr. Harlamert’s attorneys told 13News that Dr. Harlamert has “always been committed to providing quality care to patients” and that he treated his cardiology patients “based on their unique circumstances, his expertise, and the tools available.
“Because of stringent privacy laws and pending litigation, a response to a local news story is not the proper forum to present a picture of any particular treatment decision, especially when that picture may be incomplete at this time,” the statement reads.
A version of this article first appeared on Medscape.com.
alleging he performed unnecessary cardiac procedures that led to physical and emotional harm.
The medical records for one patient, 70-year-old John Pflum, of Noblesville, Ind., show that Edward Harlamert, MD, performed 44 heart catheterizations and inserted at least 41 stents between 2004 and 2013, according to an investigation by WTHR 13News in Indianapolis that was published Dec. 14.
The news outlet asked four cardiologists to review and comment on John Pflum’s medical records.
“There is not a single scenario I can think of where doing this level of stents and angiograms would be justified or make sense. I have never seen this happen in the course of my medical training or my medical career,” Payal Kohli, MD, cardiologist and medical director of Cherry Creek Heart in Denver, told 13News.
Sunil Rao, MD, director of interventional cardiology at NYU Langone Health and president of the Society for Cardiovascular Angioplasty and Interventions, who also reviewed Mr. Pflum’s medical records for 13News, said he’s “never seen a patient who has gotten this many procedures.”
Dr. Rao said that on the basis of what he saw in the records and in the images, there were several deviations from the standard of care.
Two other independent cardiologists who spoke with 13News voiced similar opinions.
Mr. Pflum was “getting cathed almost every month. That’s not how it’s done,” Morton Rinder, MD, an interventional cardiologist at St. Luke’s Hospital near St. Louis, told 13News.
Dr. Rinder has been hired as a medical consultant for the attorneys who filed Mr. Pflum’s malpractice complaint against Dr. Harlamert.
Cardiologists who reviewed the catheterization films for 13News said some of Mr. Pflum’s heart blockages met the 70% threshold to warrant consideration of a stent, while others clearly did not. In-stent restenosis occurred in several of the implanted stents, requiring a second open heart surgery.
In a statement, Dr. Harlamert’s attorneys told 13News that Dr. Harlamert has “always been committed to providing quality care to patients” and that he treated his cardiology patients “based on their unique circumstances, his expertise, and the tools available.
“Because of stringent privacy laws and pending litigation, a response to a local news story is not the proper forum to present a picture of any particular treatment decision, especially when that picture may be incomplete at this time,” the statement reads.
A version of this article first appeared on Medscape.com.
alleging he performed unnecessary cardiac procedures that led to physical and emotional harm.
The medical records for one patient, 70-year-old John Pflum, of Noblesville, Ind., show that Edward Harlamert, MD, performed 44 heart catheterizations and inserted at least 41 stents between 2004 and 2013, according to an investigation by WTHR 13News in Indianapolis that was published Dec. 14.
The news outlet asked four cardiologists to review and comment on John Pflum’s medical records.
“There is not a single scenario I can think of where doing this level of stents and angiograms would be justified or make sense. I have never seen this happen in the course of my medical training or my medical career,” Payal Kohli, MD, cardiologist and medical director of Cherry Creek Heart in Denver, told 13News.
Sunil Rao, MD, director of interventional cardiology at NYU Langone Health and president of the Society for Cardiovascular Angioplasty and Interventions, who also reviewed Mr. Pflum’s medical records for 13News, said he’s “never seen a patient who has gotten this many procedures.”
Dr. Rao said that on the basis of what he saw in the records and in the images, there were several deviations from the standard of care.
Two other independent cardiologists who spoke with 13News voiced similar opinions.
Mr. Pflum was “getting cathed almost every month. That’s not how it’s done,” Morton Rinder, MD, an interventional cardiologist at St. Luke’s Hospital near St. Louis, told 13News.
Dr. Rinder has been hired as a medical consultant for the attorneys who filed Mr. Pflum’s malpractice complaint against Dr. Harlamert.
Cardiologists who reviewed the catheterization films for 13News said some of Mr. Pflum’s heart blockages met the 70% threshold to warrant consideration of a stent, while others clearly did not. In-stent restenosis occurred in several of the implanted stents, requiring a second open heart surgery.
In a statement, Dr. Harlamert’s attorneys told 13News that Dr. Harlamert has “always been committed to providing quality care to patients” and that he treated his cardiology patients “based on their unique circumstances, his expertise, and the tools available.
“Because of stringent privacy laws and pending litigation, a response to a local news story is not the proper forum to present a picture of any particular treatment decision, especially when that picture may be incomplete at this time,” the statement reads.
A version of this article first appeared on Medscape.com.
Children and COVID: New-case counts offer dueling narratives
New COVID-19 cases in children jumped by 66% during the first 2 weeks of December after an 8-week steady period lasting through October and November, according to the American Academy of Pediatrics and the Children’s Hospital Association.
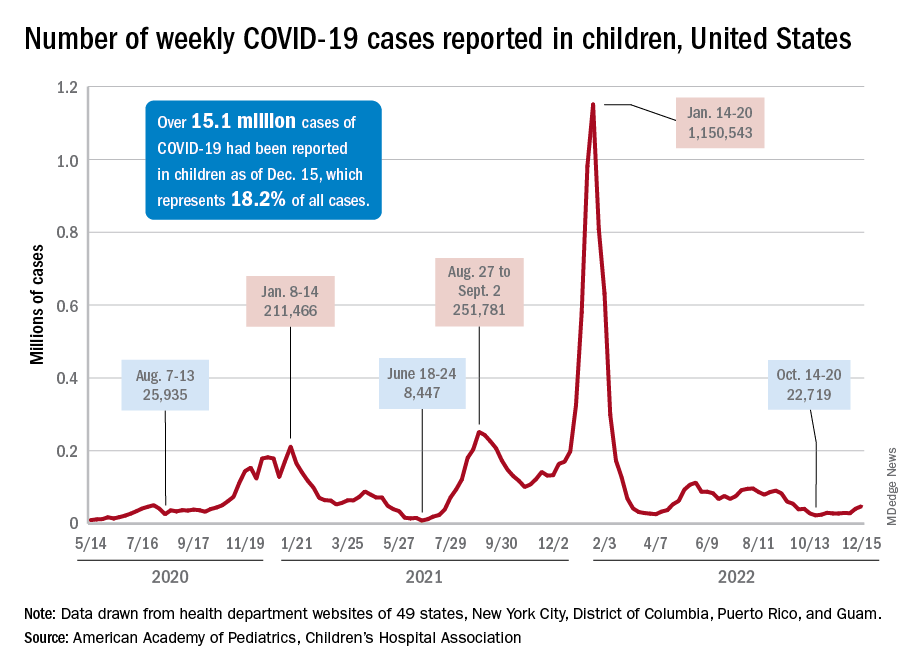
and totaling less than 29,000 for the week of Nov. 25 to Dec. 1. That increase of almost 19,000 cases is the largest over a 2-week period since late July, the AAP and CHA said in their weekly COVID report based on data collected from state and territorial health department websites.
[This publication has been following the AAP/CHA report since the summer of 2020 and continues to share the data for the sake of consistency, but it must be noted that a number of states are no longer updating their public COVID dashboards. As a result, there is now a considerable discrepancy between the AAP/CHA weekly figures and those reported by the Centers for Disease Control and Prevention, which has no such limitations on state data.]
The situation involving new cases over the last 2 weeks is quite different from the CDC’s perspective. The agency does not publish a weekly count, instead offering cumulative cases, which stood at almost 16.1 million as of Dec. 14. Calculating a 2-week total puts the new-case count for Dec. 1-14 at 113,572 among children aged 0-17 years. That is higher than the AAP/CHA count (88,629) for roughly the same period, but it is actually lower than the CDC’s figure (161,832) for the last 2 weeks of November.
The CDC data, in other words, suggest that new cases have gone down in the last 2 weeks, while the AAP and CHA, with their somewhat limited perspective, announced that new cases have gone up.
One COVID-related measure from the CDC that is not contradicted by other sources is hospitalization rates, which had climbed from 0.16 new admissions in children aged 0-17 years with confirmed COVID per 100,000 population on Oct. 22 to 0.29 per 100,000 on Dec. 9. Visits to the emergency department with diagnosed COVID, meanwhile, have been fairly steady so far through December in children, according to the CDC.
New COVID-19 cases in children jumped by 66% during the first 2 weeks of December after an 8-week steady period lasting through October and November, according to the American Academy of Pediatrics and the Children’s Hospital Association.

and totaling less than 29,000 for the week of Nov. 25 to Dec. 1. That increase of almost 19,000 cases is the largest over a 2-week period since late July, the AAP and CHA said in their weekly COVID report based on data collected from state and territorial health department websites.
[This publication has been following the AAP/CHA report since the summer of 2020 and continues to share the data for the sake of consistency, but it must be noted that a number of states are no longer updating their public COVID dashboards. As a result, there is now a considerable discrepancy between the AAP/CHA weekly figures and those reported by the Centers for Disease Control and Prevention, which has no such limitations on state data.]
The situation involving new cases over the last 2 weeks is quite different from the CDC’s perspective. The agency does not publish a weekly count, instead offering cumulative cases, which stood at almost 16.1 million as of Dec. 14. Calculating a 2-week total puts the new-case count for Dec. 1-14 at 113,572 among children aged 0-17 years. That is higher than the AAP/CHA count (88,629) for roughly the same period, but it is actually lower than the CDC’s figure (161,832) for the last 2 weeks of November.
The CDC data, in other words, suggest that new cases have gone down in the last 2 weeks, while the AAP and CHA, with their somewhat limited perspective, announced that new cases have gone up.
One COVID-related measure from the CDC that is not contradicted by other sources is hospitalization rates, which had climbed from 0.16 new admissions in children aged 0-17 years with confirmed COVID per 100,000 population on Oct. 22 to 0.29 per 100,000 on Dec. 9. Visits to the emergency department with diagnosed COVID, meanwhile, have been fairly steady so far through December in children, according to the CDC.
New COVID-19 cases in children jumped by 66% during the first 2 weeks of December after an 8-week steady period lasting through October and November, according to the American Academy of Pediatrics and the Children’s Hospital Association.

and totaling less than 29,000 for the week of Nov. 25 to Dec. 1. That increase of almost 19,000 cases is the largest over a 2-week period since late July, the AAP and CHA said in their weekly COVID report based on data collected from state and territorial health department websites.
[This publication has been following the AAP/CHA report since the summer of 2020 and continues to share the data for the sake of consistency, but it must be noted that a number of states are no longer updating their public COVID dashboards. As a result, there is now a considerable discrepancy between the AAP/CHA weekly figures and those reported by the Centers for Disease Control and Prevention, which has no such limitations on state data.]
The situation involving new cases over the last 2 weeks is quite different from the CDC’s perspective. The agency does not publish a weekly count, instead offering cumulative cases, which stood at almost 16.1 million as of Dec. 14. Calculating a 2-week total puts the new-case count for Dec. 1-14 at 113,572 among children aged 0-17 years. That is higher than the AAP/CHA count (88,629) for roughly the same period, but it is actually lower than the CDC’s figure (161,832) for the last 2 weeks of November.
The CDC data, in other words, suggest that new cases have gone down in the last 2 weeks, while the AAP and CHA, with their somewhat limited perspective, announced that new cases have gone up.
One COVID-related measure from the CDC that is not contradicted by other sources is hospitalization rates, which had climbed from 0.16 new admissions in children aged 0-17 years with confirmed COVID per 100,000 population on Oct. 22 to 0.29 per 100,000 on Dec. 9. Visits to the emergency department with diagnosed COVID, meanwhile, have been fairly steady so far through December in children, according to the CDC.
Researchers probe ‘systematic error’ in gun injury data
These coding inaccuracies could distort our understanding of gun violence in the United States and make it seem like accidental shootings are more common than they really are, researchers reported in JAMA Network Open.
“The systematic error in intent classification is not widely known or acknowledged by researchers in this field,” Philip J. Cook, PhD, of Duke University, Durham, N.C., and Susan T. Parker, of the University of Michigan, Ann Arbor, wrote in an invited commentary about the new findings. “The bulk of all shootings, nonfatal and fatal together, are assaults, which is to say the result of one person intentionally shooting another. An accurate statistical portrait thus suggests that gun violence is predominantly a crime problem.”
In 2020, 79% of all homicides and 53% of all suicides involved firearms, the CDC reported. Gun violence is now the leading cause of death for children in the United States, government data show.
For the new study, Matthew Miller, MD, ScD, of Northeastern University and the Harvard Injury Control Research Center in Boston, and his colleagues examined how International Classification of Diseases (ICD) codes may misclassify the intent behind gunshot injuries.
Dr. Miller’s group looked at 1,227 incidents between 2008 and 2019 at three major trauma centers – Brigham and Women’s Hospital and Massachusetts General Hospital, both in Boston, and Harborview Medical Center in Seattle.
Of those shootings, 837 (68.2%) involved assaults, 168 (13.5%) were unintentional, 124 (9.9%) were deliberate self-harm, and 43 (3.4%) were instances of legal intervention, based on the researchers’ review of medical records.
ICD codes at discharge, however, labeled 581 cases (47.4%) as assaults and 432 (35.2%) as unintentional.
The researchers found that 234 of the 837 assaults (28%) and 9 of the 43 legal interventions (20.9%) were miscoded as unintentional. This problem occurred even when the “medical narrative explicitly indicated that the shooting was an act of interpersonal violence,” such as a drive-by shooting or an act of domestic violence, the researchers reported.
Hospital trauma registrars, who detail the circumstances surrounding injuries, were mostly in agreement with the researchers.
Medical coders “would likely have little trouble characterizing firearm injury intent accurately if incentives were created for them to do so,” the authors wrote.
Trends and interventions
Separately, researchers published studies showing that gun violence tends to affect various demographics differently, and that remediating abandoned houses could help reduce gun crime.
Lindsay Young, of the University of Cincinnati, and Henry Xiang, MD, PhD, director of the Center for Pediatric Trauma Research at Nationwide Children’s Hospital in Columbus, Ohio, analyzed rates of firearm deaths from 1981 to 2020.
They found that the rate of firearm-related homicide was five times higher among males than females, and the rate of suicide involving firearms was nearly seven times higher for men, they reported in PLOS ONE.
Black men were the group most affected by homicide, whereas White men were most affected by suicide, they found.
To see if fixing abandoned properties would improve health and reduce gun violence in low-income, Black neighborhoods in Philadelphia, Eugenia C. South, MD, of the University of Pennsylvania, Philadelphia, and colleagues conducted a randomized trial.
They randomly assigned abandoned properties in some areas to undergo full remediation (installing working windows and doors, cleaning trash, and weeding); trash cleanup and weeding only; or no intervention.
“Abandoned houses that were remediated showed substantial drops in nearby weapons violations (−8.43%), gun assaults (−13.12%), and to a lesser extent shootings (−6.96%),” the researchers reported.
The intervention targets effects of segregation that have resulted from “historical and ongoing government and private-sector policies” that lead to disinvestment in Black, urban communities, they wrote. Abandoned houses can be used to store firearms and for other illegal activity. They also can engender feelings of fear, neglect, and stress in communities, the researchers noted.
Dr. Miller’s study was funded by the National Collaborative on Gun Violence Research; coauthors disclosed corporate, government, and university grants. The full list of disclosures can be found with the original article. Editorialists Dr. Cook and Dr. Parker report no relevant financial relationships. Dr. South’s study was funded by the National Institutes of Health. Dr. South and some coauthors disclosed government grants.
A version of this article first appeared on Medscape.com.
These coding inaccuracies could distort our understanding of gun violence in the United States and make it seem like accidental shootings are more common than they really are, researchers reported in JAMA Network Open.
“The systematic error in intent classification is not widely known or acknowledged by researchers in this field,” Philip J. Cook, PhD, of Duke University, Durham, N.C., and Susan T. Parker, of the University of Michigan, Ann Arbor, wrote in an invited commentary about the new findings. “The bulk of all shootings, nonfatal and fatal together, are assaults, which is to say the result of one person intentionally shooting another. An accurate statistical portrait thus suggests that gun violence is predominantly a crime problem.”
In 2020, 79% of all homicides and 53% of all suicides involved firearms, the CDC reported. Gun violence is now the leading cause of death for children in the United States, government data show.
For the new study, Matthew Miller, MD, ScD, of Northeastern University and the Harvard Injury Control Research Center in Boston, and his colleagues examined how International Classification of Diseases (ICD) codes may misclassify the intent behind gunshot injuries.
Dr. Miller’s group looked at 1,227 incidents between 2008 and 2019 at three major trauma centers – Brigham and Women’s Hospital and Massachusetts General Hospital, both in Boston, and Harborview Medical Center in Seattle.
Of those shootings, 837 (68.2%) involved assaults, 168 (13.5%) were unintentional, 124 (9.9%) were deliberate self-harm, and 43 (3.4%) were instances of legal intervention, based on the researchers’ review of medical records.
ICD codes at discharge, however, labeled 581 cases (47.4%) as assaults and 432 (35.2%) as unintentional.
The researchers found that 234 of the 837 assaults (28%) and 9 of the 43 legal interventions (20.9%) were miscoded as unintentional. This problem occurred even when the “medical narrative explicitly indicated that the shooting was an act of interpersonal violence,” such as a drive-by shooting or an act of domestic violence, the researchers reported.
Hospital trauma registrars, who detail the circumstances surrounding injuries, were mostly in agreement with the researchers.
Medical coders “would likely have little trouble characterizing firearm injury intent accurately if incentives were created for them to do so,” the authors wrote.
Trends and interventions
Separately, researchers published studies showing that gun violence tends to affect various demographics differently, and that remediating abandoned houses could help reduce gun crime.
Lindsay Young, of the University of Cincinnati, and Henry Xiang, MD, PhD, director of the Center for Pediatric Trauma Research at Nationwide Children’s Hospital in Columbus, Ohio, analyzed rates of firearm deaths from 1981 to 2020.
They found that the rate of firearm-related homicide was five times higher among males than females, and the rate of suicide involving firearms was nearly seven times higher for men, they reported in PLOS ONE.
Black men were the group most affected by homicide, whereas White men were most affected by suicide, they found.
To see if fixing abandoned properties would improve health and reduce gun violence in low-income, Black neighborhoods in Philadelphia, Eugenia C. South, MD, of the University of Pennsylvania, Philadelphia, and colleagues conducted a randomized trial.
They randomly assigned abandoned properties in some areas to undergo full remediation (installing working windows and doors, cleaning trash, and weeding); trash cleanup and weeding only; or no intervention.
“Abandoned houses that were remediated showed substantial drops in nearby weapons violations (−8.43%), gun assaults (−13.12%), and to a lesser extent shootings (−6.96%),” the researchers reported.
The intervention targets effects of segregation that have resulted from “historical and ongoing government and private-sector policies” that lead to disinvestment in Black, urban communities, they wrote. Abandoned houses can be used to store firearms and for other illegal activity. They also can engender feelings of fear, neglect, and stress in communities, the researchers noted.
Dr. Miller’s study was funded by the National Collaborative on Gun Violence Research; coauthors disclosed corporate, government, and university grants. The full list of disclosures can be found with the original article. Editorialists Dr. Cook and Dr. Parker report no relevant financial relationships. Dr. South’s study was funded by the National Institutes of Health. Dr. South and some coauthors disclosed government grants.
A version of this article first appeared on Medscape.com.
These coding inaccuracies could distort our understanding of gun violence in the United States and make it seem like accidental shootings are more common than they really are, researchers reported in JAMA Network Open.
“The systematic error in intent classification is not widely known or acknowledged by researchers in this field,” Philip J. Cook, PhD, of Duke University, Durham, N.C., and Susan T. Parker, of the University of Michigan, Ann Arbor, wrote in an invited commentary about the new findings. “The bulk of all shootings, nonfatal and fatal together, are assaults, which is to say the result of one person intentionally shooting another. An accurate statistical portrait thus suggests that gun violence is predominantly a crime problem.”
In 2020, 79% of all homicides and 53% of all suicides involved firearms, the CDC reported. Gun violence is now the leading cause of death for children in the United States, government data show.
For the new study, Matthew Miller, MD, ScD, of Northeastern University and the Harvard Injury Control Research Center in Boston, and his colleagues examined how International Classification of Diseases (ICD) codes may misclassify the intent behind gunshot injuries.
Dr. Miller’s group looked at 1,227 incidents between 2008 and 2019 at three major trauma centers – Brigham and Women’s Hospital and Massachusetts General Hospital, both in Boston, and Harborview Medical Center in Seattle.
Of those shootings, 837 (68.2%) involved assaults, 168 (13.5%) were unintentional, 124 (9.9%) were deliberate self-harm, and 43 (3.4%) were instances of legal intervention, based on the researchers’ review of medical records.
ICD codes at discharge, however, labeled 581 cases (47.4%) as assaults and 432 (35.2%) as unintentional.
The researchers found that 234 of the 837 assaults (28%) and 9 of the 43 legal interventions (20.9%) were miscoded as unintentional. This problem occurred even when the “medical narrative explicitly indicated that the shooting was an act of interpersonal violence,” such as a drive-by shooting or an act of domestic violence, the researchers reported.
Hospital trauma registrars, who detail the circumstances surrounding injuries, were mostly in agreement with the researchers.
Medical coders “would likely have little trouble characterizing firearm injury intent accurately if incentives were created for them to do so,” the authors wrote.
Trends and interventions
Separately, researchers published studies showing that gun violence tends to affect various demographics differently, and that remediating abandoned houses could help reduce gun crime.
Lindsay Young, of the University of Cincinnati, and Henry Xiang, MD, PhD, director of the Center for Pediatric Trauma Research at Nationwide Children’s Hospital in Columbus, Ohio, analyzed rates of firearm deaths from 1981 to 2020.
They found that the rate of firearm-related homicide was five times higher among males than females, and the rate of suicide involving firearms was nearly seven times higher for men, they reported in PLOS ONE.
Black men were the group most affected by homicide, whereas White men were most affected by suicide, they found.
To see if fixing abandoned properties would improve health and reduce gun violence in low-income, Black neighborhoods in Philadelphia, Eugenia C. South, MD, of the University of Pennsylvania, Philadelphia, and colleagues conducted a randomized trial.
They randomly assigned abandoned properties in some areas to undergo full remediation (installing working windows and doors, cleaning trash, and weeding); trash cleanup and weeding only; or no intervention.
“Abandoned houses that were remediated showed substantial drops in nearby weapons violations (−8.43%), gun assaults (−13.12%), and to a lesser extent shootings (−6.96%),” the researchers reported.
The intervention targets effects of segregation that have resulted from “historical and ongoing government and private-sector policies” that lead to disinvestment in Black, urban communities, they wrote. Abandoned houses can be used to store firearms and for other illegal activity. They also can engender feelings of fear, neglect, and stress in communities, the researchers noted.
Dr. Miller’s study was funded by the National Collaborative on Gun Violence Research; coauthors disclosed corporate, government, and university grants. The full list of disclosures can be found with the original article. Editorialists Dr. Cook and Dr. Parker report no relevant financial relationships. Dr. South’s study was funded by the National Institutes of Health. Dr. South and some coauthors disclosed government grants.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
COVID booster shot poll: People ‘don’t think they need one’
Now, a new poll shows why so few people are willing to roll up their sleeves again.
The most common reasons people give for not getting the latest booster shot is that they “don’t think they need one” (44%) and they “don’t think the benefits are worth it” (37%), according to poll results from the Kaiser Family Foundation.
The data comes amid announcements by the Centers for Disease Control and Prevention that boosters reduced COVID-19 hospitalizations by up to 57% for U.S. adults and by up to 84% for people age 65 and older. Those figures are just the latest in a mountain of research reporting the public health benefits of COVID-19 vaccines.
Despite all of the statistical data, health officials’ recent vaccination campaigns have proven far from compelling.
So far, just 15% of people age 12 and older have gotten the latest booster, and 36% of people age 65 and older have gotten it, the CDC’s vaccination trackershows.
Since the start of the pandemic, 1.1 million people in the U.S. have died from COVID-19, with the number of deaths currently rising by 400 per day, The New York Times COVID tracker shows.
Many experts continue to note the need for everyone to get booster shots regularly, but some advocate that perhaps a change in strategy is in order.
“What the administration should do is push for vaccinating people in high-risk groups, including those who are older, those who are immunocompromised and those who have comorbidities,” Paul Offitt, MD, director of the Vaccine Education Center at Children’s Hospital of Philadelphia, told CNN.
Federal regulators have announced they will meet Jan. 26 with a panel of vaccine advisors to examine the current recommended vaccination schedule as well as look at the effectiveness and composition of current vaccines and boosters, with an eye toward the make-up of next-generation shots.
Vaccines are the “best available protection” against hospitalization and death caused by COVID-19, said Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, in a statement announcing the planned meeting.
“Since the initial authorizations of these vaccines, we have learned that protection wanes over time, especially as the virus rapidly mutates and new variants and subvariants emerge,” he said. “Therefore, it’s important to continue discussions about the optimal composition of COVID-19 vaccines for primary and booster vaccination, as well as the optimal interval for booster vaccination.”
A version of this article first appeared on WebMD.com.
Now, a new poll shows why so few people are willing to roll up their sleeves again.
The most common reasons people give for not getting the latest booster shot is that they “don’t think they need one” (44%) and they “don’t think the benefits are worth it” (37%), according to poll results from the Kaiser Family Foundation.
The data comes amid announcements by the Centers for Disease Control and Prevention that boosters reduced COVID-19 hospitalizations by up to 57% for U.S. adults and by up to 84% for people age 65 and older. Those figures are just the latest in a mountain of research reporting the public health benefits of COVID-19 vaccines.
Despite all of the statistical data, health officials’ recent vaccination campaigns have proven far from compelling.
So far, just 15% of people age 12 and older have gotten the latest booster, and 36% of people age 65 and older have gotten it, the CDC’s vaccination trackershows.
Since the start of the pandemic, 1.1 million people in the U.S. have died from COVID-19, with the number of deaths currently rising by 400 per day, The New York Times COVID tracker shows.
Many experts continue to note the need for everyone to get booster shots regularly, but some advocate that perhaps a change in strategy is in order.
“What the administration should do is push for vaccinating people in high-risk groups, including those who are older, those who are immunocompromised and those who have comorbidities,” Paul Offitt, MD, director of the Vaccine Education Center at Children’s Hospital of Philadelphia, told CNN.
Federal regulators have announced they will meet Jan. 26 with a panel of vaccine advisors to examine the current recommended vaccination schedule as well as look at the effectiveness and composition of current vaccines and boosters, with an eye toward the make-up of next-generation shots.
Vaccines are the “best available protection” against hospitalization and death caused by COVID-19, said Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, in a statement announcing the planned meeting.
“Since the initial authorizations of these vaccines, we have learned that protection wanes over time, especially as the virus rapidly mutates and new variants and subvariants emerge,” he said. “Therefore, it’s important to continue discussions about the optimal composition of COVID-19 vaccines for primary and booster vaccination, as well as the optimal interval for booster vaccination.”
A version of this article first appeared on WebMD.com.
Now, a new poll shows why so few people are willing to roll up their sleeves again.
The most common reasons people give for not getting the latest booster shot is that they “don’t think they need one” (44%) and they “don’t think the benefits are worth it” (37%), according to poll results from the Kaiser Family Foundation.
The data comes amid announcements by the Centers for Disease Control and Prevention that boosters reduced COVID-19 hospitalizations by up to 57% for U.S. adults and by up to 84% for people age 65 and older. Those figures are just the latest in a mountain of research reporting the public health benefits of COVID-19 vaccines.
Despite all of the statistical data, health officials’ recent vaccination campaigns have proven far from compelling.
So far, just 15% of people age 12 and older have gotten the latest booster, and 36% of people age 65 and older have gotten it, the CDC’s vaccination trackershows.
Since the start of the pandemic, 1.1 million people in the U.S. have died from COVID-19, with the number of deaths currently rising by 400 per day, The New York Times COVID tracker shows.
Many experts continue to note the need for everyone to get booster shots regularly, but some advocate that perhaps a change in strategy is in order.
“What the administration should do is push for vaccinating people in high-risk groups, including those who are older, those who are immunocompromised and those who have comorbidities,” Paul Offitt, MD, director of the Vaccine Education Center at Children’s Hospital of Philadelphia, told CNN.
Federal regulators have announced they will meet Jan. 26 with a panel of vaccine advisors to examine the current recommended vaccination schedule as well as look at the effectiveness and composition of current vaccines and boosters, with an eye toward the make-up of next-generation shots.
Vaccines are the “best available protection” against hospitalization and death caused by COVID-19, said Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, in a statement announcing the planned meeting.
“Since the initial authorizations of these vaccines, we have learned that protection wanes over time, especially as the virus rapidly mutates and new variants and subvariants emerge,” he said. “Therefore, it’s important to continue discussions about the optimal composition of COVID-19 vaccines for primary and booster vaccination, as well as the optimal interval for booster vaccination.”
A version of this article first appeared on WebMD.com.
Hospitals with more diverse and uninsured patients more likely to provide delayed fracture care
Regardless of individual patient-level characteristics such as race, ethnicity, or insurance status, these patients were more likely to miss the recommended 24-hour benchmark for surgery.
“Institutions that treat a less diverse patient population appeared to be more resilient to the mix of insurance status in their patient population and were more likely to meet time-to-surgery benchmarks, regardless of patient insurance status or population-based insurance mix,” write study author Ida Leah Gitajn, MD, an orthopedic trauma surgeon at Dartmouth-Hitchcock Medical Center, Lebanon, N.H., and colleagues.
“While it is unsurprising that increased delays were associated with underfunded institutions, the association between institutional-level racial disparity and surgical delays implies structural health systems bias,” the authors wrote.
The study was published online in JAMA Network Open.
Site performance varied
Racial inequalities in health care utilization and outcomes have been documented in many medical specialties, including orthopedic trauma, the study authors write. However, previous studies evaluating racial disparities in fracture care have been limited to patient-level associations rather than hospital-level factors.
The investigators conducted a secondary analysis of prospectively collected multicenter data for 2,565 patients with hip and femur fractures enrolled in two randomized trials at 23 sites in the United States and Canada. The researchers assessed whether disparities in meeting 24-hour time-to-surgery benchmarks exist at the patient level or at the institutional level, evaluating the association of race, ethnicity, and insurance status.
The cohort study used data from the Program of Randomized Trials to Evaluate Preoperative Antiseptic Skin Solutions in Orthopaedic Trauma (PREP-IT), which enrolled patients from 2018-2021 and followed them for 1 year. All patients with hip and femur fractures enrolled in the PREP-IT program were included in the analysis, which was conducted from April to September of this year.
The cohort included 2,565 patients with an average age of about 65 years. About 82% of patients were White, 13.4% were Black, 3.2% were Asian, and 1.1% were classified as another race or ethnicity. Among the study population, 32.5% of participants were employed, and 92.2% had health insurance. Nearly 40% had a femur fracture with an average injury severity score of 10.4.
Overall, 596 patients (23.2%) didn’t meet the 24-hour time-to-operating-room benchmark. Patients who didn’t meet the 24-hour surgical window were more likely to be older, women, and have a femur fracture. They were less likely to be employed.
The 23 sites had variability in meeting the 24-hour benchmark, race and ethnicity distribution, and population-based health insurance. Institutions met benchmarks at frequencies ranging from 45.2% (for 196 of 433 procedures) to 97.4% (37 of 38 procedures). Minority race and ethnicity distribution ranged from 0% (in 99 procedures) to 58.2% (in 53 of 91 procedures). The proportion of uninsured patients ranged from 0% (in 64 procedures) to 34.2% (in 13 of 38 procedures).
At the patient level, there was no association between missing the 24-hour benchmark and race or ethnicity, and there was no independent association between hospital population racial composition and surgical delay. In an analysis that controlled for patient-level characteristics, there was no association between missing the 24-hour benchmark and patient-level insurance status.
There was an independent association, however, between the hospital population insurance coverage and hospital population racial composition as an interaction term, suggesting a moderating effect (P = .03), the study authors write.
At low rates of uninsured patients, the probability of missing the 24-hour benchmark was 12.5%-14.6% when racial composition varied from 0%-50% minority patients. In contrast, at higher rates of uninsured patients, the risk of missing the 24-hour window was higher among more diverse populations. For instance, at 30% uninsured, the risk of missing the benchmark was 0.5% when the racial composition was low and 17.6% at 50% minority patients.
Additional studies are needed to understand the findings and how health system programs or structures play a role, the authors write. For instance, well-funded health systems that care for a higher proportion of insured patients likely have quality improvement programs and other support structures, such as operating room access, that ensure appropriate time-to-surgery benchmarks for time-sensitive fractures, they say.
Addressing inequalities
Troy Amen, MD, MBA, an orthopedic surgery resident at the Hospital for Special Surgery, New York, said, “Despite these disparities being reported and well documented in recent years, unfortunately, not enough has been done to address them or understand their fundamental root causes.”
Dr. Amen, who wasn’t involved with this study, has researched racial and ethnic disparities in hip fracture surgery care across the United States. He and his colleagues found disparities in delayed time-to-surgery, particularly for Black patients.
“We live in a country and society where we want and strive for equality of care for patients regardless of race, ethnicity, gender, sexual orientation, or background,” he said. “We have a moral imperative to address these disparities as health care providers, not only among ourselves, but also in conjunction with lawmakers, hospital administrators, and health policy specialists.”
Uma Srikumaran, MD, an associate professor of orthopedic surgery at Johns Hopkins University, Baltimore, wasn’t involved with this study but has researched racial disparities in the timing of radiographic assessment and surgical treatment of hip fractures.
“Though we understand that racial disparities are pervasive in health care, we have a great deal left to understand about the extent of those disparities and all the various factors that contribute to them,” Dr. Srikumaran told this news organization.
Dr. Srikumaran and colleagues have found that Black patients had longer wait times for evaluation and surgery than White patients.
“We all want to get to the solutions, but those can be difficult to execute without an intricate understanding of the problem,” he said. “We should encourage this type of research all throughout health care in general but also very locally, as solutions are not likely to be one-size-fits-all.”
Dr. Srikumaran pointed to the need to measure the problem in specific pathologies, populations, geographies, hospital types, and other factors.
“Studying the trends of this issue will help us determine whether our national or local initiatives are making a difference and which interventions are most effective for a particular hospital, geographic location, or particular pathology,” he said. “Accordingly, if a particular hospital or health system isn’t looking at differences in the delivery of care by race, they are missing an opportunity to ensure equity and raise overall quality.”
The study was supported by funding from the Patient Centered Outcomes Research Institute. Dr. Gitajn reported receiving personal fees for consulting and teaching work from Stryker outside the submitted work. Dr. Amen and Dr. Srikumaran reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Regardless of individual patient-level characteristics such as race, ethnicity, or insurance status, these patients were more likely to miss the recommended 24-hour benchmark for surgery.
“Institutions that treat a less diverse patient population appeared to be more resilient to the mix of insurance status in their patient population and were more likely to meet time-to-surgery benchmarks, regardless of patient insurance status or population-based insurance mix,” write study author Ida Leah Gitajn, MD, an orthopedic trauma surgeon at Dartmouth-Hitchcock Medical Center, Lebanon, N.H., and colleagues.
“While it is unsurprising that increased delays were associated with underfunded institutions, the association between institutional-level racial disparity and surgical delays implies structural health systems bias,” the authors wrote.
The study was published online in JAMA Network Open.
Site performance varied
Racial inequalities in health care utilization and outcomes have been documented in many medical specialties, including orthopedic trauma, the study authors write. However, previous studies evaluating racial disparities in fracture care have been limited to patient-level associations rather than hospital-level factors.
The investigators conducted a secondary analysis of prospectively collected multicenter data for 2,565 patients with hip and femur fractures enrolled in two randomized trials at 23 sites in the United States and Canada. The researchers assessed whether disparities in meeting 24-hour time-to-surgery benchmarks exist at the patient level or at the institutional level, evaluating the association of race, ethnicity, and insurance status.
The cohort study used data from the Program of Randomized Trials to Evaluate Preoperative Antiseptic Skin Solutions in Orthopaedic Trauma (PREP-IT), which enrolled patients from 2018-2021 and followed them for 1 year. All patients with hip and femur fractures enrolled in the PREP-IT program were included in the analysis, which was conducted from April to September of this year.
The cohort included 2,565 patients with an average age of about 65 years. About 82% of patients were White, 13.4% were Black, 3.2% were Asian, and 1.1% were classified as another race or ethnicity. Among the study population, 32.5% of participants were employed, and 92.2% had health insurance. Nearly 40% had a femur fracture with an average injury severity score of 10.4.
Overall, 596 patients (23.2%) didn’t meet the 24-hour time-to-operating-room benchmark. Patients who didn’t meet the 24-hour surgical window were more likely to be older, women, and have a femur fracture. They were less likely to be employed.
The 23 sites had variability in meeting the 24-hour benchmark, race and ethnicity distribution, and population-based health insurance. Institutions met benchmarks at frequencies ranging from 45.2% (for 196 of 433 procedures) to 97.4% (37 of 38 procedures). Minority race and ethnicity distribution ranged from 0% (in 99 procedures) to 58.2% (in 53 of 91 procedures). The proportion of uninsured patients ranged from 0% (in 64 procedures) to 34.2% (in 13 of 38 procedures).
At the patient level, there was no association between missing the 24-hour benchmark and race or ethnicity, and there was no independent association between hospital population racial composition and surgical delay. In an analysis that controlled for patient-level characteristics, there was no association between missing the 24-hour benchmark and patient-level insurance status.
There was an independent association, however, between the hospital population insurance coverage and hospital population racial composition as an interaction term, suggesting a moderating effect (P = .03), the study authors write.
At low rates of uninsured patients, the probability of missing the 24-hour benchmark was 12.5%-14.6% when racial composition varied from 0%-50% minority patients. In contrast, at higher rates of uninsured patients, the risk of missing the 24-hour window was higher among more diverse populations. For instance, at 30% uninsured, the risk of missing the benchmark was 0.5% when the racial composition was low and 17.6% at 50% minority patients.
Additional studies are needed to understand the findings and how health system programs or structures play a role, the authors write. For instance, well-funded health systems that care for a higher proportion of insured patients likely have quality improvement programs and other support structures, such as operating room access, that ensure appropriate time-to-surgery benchmarks for time-sensitive fractures, they say.
Addressing inequalities
Troy Amen, MD, MBA, an orthopedic surgery resident at the Hospital for Special Surgery, New York, said, “Despite these disparities being reported and well documented in recent years, unfortunately, not enough has been done to address them or understand their fundamental root causes.”
Dr. Amen, who wasn’t involved with this study, has researched racial and ethnic disparities in hip fracture surgery care across the United States. He and his colleagues found disparities in delayed time-to-surgery, particularly for Black patients.
“We live in a country and society where we want and strive for equality of care for patients regardless of race, ethnicity, gender, sexual orientation, or background,” he said. “We have a moral imperative to address these disparities as health care providers, not only among ourselves, but also in conjunction with lawmakers, hospital administrators, and health policy specialists.”
Uma Srikumaran, MD, an associate professor of orthopedic surgery at Johns Hopkins University, Baltimore, wasn’t involved with this study but has researched racial disparities in the timing of radiographic assessment and surgical treatment of hip fractures.
“Though we understand that racial disparities are pervasive in health care, we have a great deal left to understand about the extent of those disparities and all the various factors that contribute to them,” Dr. Srikumaran told this news organization.
Dr. Srikumaran and colleagues have found that Black patients had longer wait times for evaluation and surgery than White patients.
“We all want to get to the solutions, but those can be difficult to execute without an intricate understanding of the problem,” he said. “We should encourage this type of research all throughout health care in general but also very locally, as solutions are not likely to be one-size-fits-all.”
Dr. Srikumaran pointed to the need to measure the problem in specific pathologies, populations, geographies, hospital types, and other factors.
“Studying the trends of this issue will help us determine whether our national or local initiatives are making a difference and which interventions are most effective for a particular hospital, geographic location, or particular pathology,” he said. “Accordingly, if a particular hospital or health system isn’t looking at differences in the delivery of care by race, they are missing an opportunity to ensure equity and raise overall quality.”
The study was supported by funding from the Patient Centered Outcomes Research Institute. Dr. Gitajn reported receiving personal fees for consulting and teaching work from Stryker outside the submitted work. Dr. Amen and Dr. Srikumaran reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Regardless of individual patient-level characteristics such as race, ethnicity, or insurance status, these patients were more likely to miss the recommended 24-hour benchmark for surgery.
“Institutions that treat a less diverse patient population appeared to be more resilient to the mix of insurance status in their patient population and were more likely to meet time-to-surgery benchmarks, regardless of patient insurance status or population-based insurance mix,” write study author Ida Leah Gitajn, MD, an orthopedic trauma surgeon at Dartmouth-Hitchcock Medical Center, Lebanon, N.H., and colleagues.
“While it is unsurprising that increased delays were associated with underfunded institutions, the association between institutional-level racial disparity and surgical delays implies structural health systems bias,” the authors wrote.
The study was published online in JAMA Network Open.
Site performance varied
Racial inequalities in health care utilization and outcomes have been documented in many medical specialties, including orthopedic trauma, the study authors write. However, previous studies evaluating racial disparities in fracture care have been limited to patient-level associations rather than hospital-level factors.
The investigators conducted a secondary analysis of prospectively collected multicenter data for 2,565 patients with hip and femur fractures enrolled in two randomized trials at 23 sites in the United States and Canada. The researchers assessed whether disparities in meeting 24-hour time-to-surgery benchmarks exist at the patient level or at the institutional level, evaluating the association of race, ethnicity, and insurance status.
The cohort study used data from the Program of Randomized Trials to Evaluate Preoperative Antiseptic Skin Solutions in Orthopaedic Trauma (PREP-IT), which enrolled patients from 2018-2021 and followed them for 1 year. All patients with hip and femur fractures enrolled in the PREP-IT program were included in the analysis, which was conducted from April to September of this year.
The cohort included 2,565 patients with an average age of about 65 years. About 82% of patients were White, 13.4% were Black, 3.2% were Asian, and 1.1% were classified as another race or ethnicity. Among the study population, 32.5% of participants were employed, and 92.2% had health insurance. Nearly 40% had a femur fracture with an average injury severity score of 10.4.
Overall, 596 patients (23.2%) didn’t meet the 24-hour time-to-operating-room benchmark. Patients who didn’t meet the 24-hour surgical window were more likely to be older, women, and have a femur fracture. They were less likely to be employed.
The 23 sites had variability in meeting the 24-hour benchmark, race and ethnicity distribution, and population-based health insurance. Institutions met benchmarks at frequencies ranging from 45.2% (for 196 of 433 procedures) to 97.4% (37 of 38 procedures). Minority race and ethnicity distribution ranged from 0% (in 99 procedures) to 58.2% (in 53 of 91 procedures). The proportion of uninsured patients ranged from 0% (in 64 procedures) to 34.2% (in 13 of 38 procedures).
At the patient level, there was no association between missing the 24-hour benchmark and race or ethnicity, and there was no independent association between hospital population racial composition and surgical delay. In an analysis that controlled for patient-level characteristics, there was no association between missing the 24-hour benchmark and patient-level insurance status.
There was an independent association, however, between the hospital population insurance coverage and hospital population racial composition as an interaction term, suggesting a moderating effect (P = .03), the study authors write.
At low rates of uninsured patients, the probability of missing the 24-hour benchmark was 12.5%-14.6% when racial composition varied from 0%-50% minority patients. In contrast, at higher rates of uninsured patients, the risk of missing the 24-hour window was higher among more diverse populations. For instance, at 30% uninsured, the risk of missing the benchmark was 0.5% when the racial composition was low and 17.6% at 50% minority patients.
Additional studies are needed to understand the findings and how health system programs or structures play a role, the authors write. For instance, well-funded health systems that care for a higher proportion of insured patients likely have quality improvement programs and other support structures, such as operating room access, that ensure appropriate time-to-surgery benchmarks for time-sensitive fractures, they say.
Addressing inequalities
Troy Amen, MD, MBA, an orthopedic surgery resident at the Hospital for Special Surgery, New York, said, “Despite these disparities being reported and well documented in recent years, unfortunately, not enough has been done to address them or understand their fundamental root causes.”
Dr. Amen, who wasn’t involved with this study, has researched racial and ethnic disparities in hip fracture surgery care across the United States. He and his colleagues found disparities in delayed time-to-surgery, particularly for Black patients.
“We live in a country and society where we want and strive for equality of care for patients regardless of race, ethnicity, gender, sexual orientation, or background,” he said. “We have a moral imperative to address these disparities as health care providers, not only among ourselves, but also in conjunction with lawmakers, hospital administrators, and health policy specialists.”
Uma Srikumaran, MD, an associate professor of orthopedic surgery at Johns Hopkins University, Baltimore, wasn’t involved with this study but has researched racial disparities in the timing of radiographic assessment and surgical treatment of hip fractures.
“Though we understand that racial disparities are pervasive in health care, we have a great deal left to understand about the extent of those disparities and all the various factors that contribute to them,” Dr. Srikumaran told this news organization.
Dr. Srikumaran and colleagues have found that Black patients had longer wait times for evaluation and surgery than White patients.
“We all want to get to the solutions, but those can be difficult to execute without an intricate understanding of the problem,” he said. “We should encourage this type of research all throughout health care in general but also very locally, as solutions are not likely to be one-size-fits-all.”
Dr. Srikumaran pointed to the need to measure the problem in specific pathologies, populations, geographies, hospital types, and other factors.
“Studying the trends of this issue will help us determine whether our national or local initiatives are making a difference and which interventions are most effective for a particular hospital, geographic location, or particular pathology,” he said. “Accordingly, if a particular hospital or health system isn’t looking at differences in the delivery of care by race, they are missing an opportunity to ensure equity and raise overall quality.”
The study was supported by funding from the Patient Centered Outcomes Research Institute. Dr. Gitajn reported receiving personal fees for consulting and teaching work from Stryker outside the submitted work. Dr. Amen and Dr. Srikumaran reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
The 'Plaque Hypothesis': Focus on vulnerable lesions to cut events
A new strategy for the management of atherosclerotic plaque as a source of major adverse cardiac events is needed with the focus shifting from the flow-limiting coronary artery luminal lesions to the overall atherosclerotic burden, be it obstructive or nonobstructive, according to a review article.
The article, by Peter H. Stone, MD, and Peter Libby, MD, Brigham and Women’s Hospital, Boston, and William E. Boden, MD, Boston University School of Medicine, was published online in JAMA Cardiology.
The review explored new data from vascular biology, atherosclerosis imaging, natural history outcome studies, and large-scale clinical trials that support what the authors refer to as “The Plaque Hypothesis” – the idea that major adverse cardiac events such as myocardial infarction and cardiac death are triggered by destabilization of vulnerable plaque, which may be obstructive or nonobstructive.
“We need to consider embracing a new management strategy that directs our diagnostic and management focus to evaluate the entire length of the atheromatous coronary artery and broaden the target of our therapeutic intervention to include all regions of the plaque (both flow-limiting and non–flow-limiting), even those that are distant from the presumed ischemia-producing obstruction,” the authors concluded.
Dr. Stone explained to this news organization that, for several decades, the medical community has focused on plaques causing severe obstruction of coronary arteries as being responsible for major adverse cardiac events. This approach – known as the Ischemia Hypothesis – has been the accepted strategy for many years, with all guidelines advising the identification of the stenoses that cause the most obstruction for treatment with stenting.
However, the authors pointed out that a number of studies have now suggested that, while these severe obstructive stenoses cause angina, they do not seem to be responsible for the hard events of MI, acute coronary syndrome (ACS), and cardiac death.
Several studies including the COURAGE trial and BARI-2D, and the recent ISCHEMIA trial have all failed to show a reduction in these hard endpoints by intervening on these severe obstructive lesions, Dr. Stone noted.
“We present evidence for a new approach – that it is the composition and vascular biology of the atherosclerotic plaques that cause MI, ACS, and cardiac death, rather than simply how obstructive they are,” he said.
Dr. Stone pointed out that plaque seen on a coronary angiogram looks at only the lumen of the artery, but plaque is primarily based in the wall of the artery, and if that plaque is inflamed it can easily be the culprit responsible for adverse events even without encroaching into the lumen.
“Our paper describes many factors which can cause plaques to destabilize and cause an ACS. These include anatomical, biochemical, and biomechanical features that together cause plaque rupture or erosion and precipitate a clinical event. It is not sufficient to just look for obstructive plaques on a coronary angiogram,” he said. “We are barking up the wrong tree. We need to look for inflamed plaque in the whole wall of the coronary arteries.”
The authors described different factors that identify a plaque at high risk of destabilization. These include a large area of vulnerable plaque, a thin-cap atheroma, a severe inflamed core, macrocalcifications, a large plaque burden, and a physical profile that would encourage a thrombus to become trapped.
“Atherosclerotic plaques are very heterogeneous and complex structures and it is not just the mountain peaks but also the lower foothills that can precipitate a flow-limiting obstruction,” Dr. Stone noted.
“The slope of the mountain is probably very important in the ability for a thrombus to form. If the slope is gradual there isn’t a problem. But if the slope is jagged with sharp edges this can cause a thrombus to become trapped. We need to look at the entirety of plaque and all its risk features to identify the culprit areas that could cause MI or cardiac death. These are typically not the obstructive plaques we have all been fixated on for many years,” he added.
“We need to focus on plaque heterogeneity. Once plaque is old and just made up of scar tissue which is not inflamed it does not cause much [of] a problem – we can probably just leave it alone. Some of these obstructive plaques may cause some angina but many do not cause major cardiac events unless they have other high-risk features,” he said.
“Cardiac events are still caused by obstruction of blood flow but that can be an abrupt process where a thrombus attaches itself to an area of destabilized plaque. These areas of plaque were not necessarily obstructing to start with. We believe that this is the explanation behind the observation that 50% of all people who have an MI (half of which are fatal) do not have symptoms beforehand,” Dr. Stone commented.
Because these areas of destabilized plaque do not cause symptoms, he believes that vast populations of people with established cardiovascular risk factors should undergo screening. “At the moment we wait for people to experience chest pain or to have an MI – that is far too little too late.”
To identify these areas of high-risk plaques, imaging techniques looking inside the artery wall are needed such as intravascular ultrasound. However, this is an invasive procedure, and the noninvasive coronary CT angiography also gives a good picture, so it is probably the best way to begin as a wider screening modality, with more invasive screening methods then used in those found to be at risk, Dr. Stone suggested.
Plaques that are identified as likely to destabilize can be treated with percutaneous coronary intervention and stenting.
While systemic therapies are useful, those currently available are not sufficient, Dr. Stone noted. For example, there are still high levels of major cardiac events in patients treated with the PCSK9 inhibitors, which bring about very large reductions in LDL cholesterol. “These therapies are beneficial, but they are not enough on their own. So, these areas of unstable plaque would need to be treated with stenting or something similar. We believe that the intervention of stenting is good but at present it is targeted at the wrong areas,” he stated.
“Clearly what we’ve been doing – stenting only obstructive lesions – does not reduce hard clinical events. Imaging methods have improved so much in recent years that we can now identify high-risk areas of plaque. This whole field of studying the vulnerable plaque has been ongoing for many years, but it is only recently that imaging methods have become good enough to identify plaques at risk. This field is now coming of age,” he added.
The next steps are to start identifying these plaques in larger populations, more accurately characterizing those at the highest risk, and then performing randomized trials of preemptive intervention in those believed to be at highest risk, and follow up for clinical events, Dr. Stone explained.
Advances in detecting unstable plaque may also permit early evaluation of novel therapeutics and gauge the intensity of lifestyle and disease-modifying pharmacotherapy, the authors suggested.
This work was supported in part by the National Heart, Lung, and Blood Institute, the American Heart Association, the RRM Charitable Fund, the Simard Fund, and the Schaubert Family. Dr. Libby is an unpaid consultant to or involved in clinical trials with Amgen, AstraZeneca, Baim Institute, Beren Therapeutics, Esperion Therapeutics, Genentech, Kancera, Kowa Pharmaceuticals, MedImmune, Merck, Norvo Nordisk, Novartis, Pfizer, and Sanofi-Regeneron; and is a member of the scientific advisory board for Amgen, Caristo Diagnostics, Cartesian Therapeutics, CSL Behring, DalCor Pharmaceuticals, Dewpoint Therapeutics, Elucid Bioimaging, Kancera, Kowa Pharmaceuticals, Olatec Therapeutics, MedImmune, Moderna, Novartis, PlaqueTec, TenSixteen Bio, Soley Thereapeutics, and XBiotech.
A version of this article first appeared on Medscape.com.
A new strategy for the management of atherosclerotic plaque as a source of major adverse cardiac events is needed with the focus shifting from the flow-limiting coronary artery luminal lesions to the overall atherosclerotic burden, be it obstructive or nonobstructive, according to a review article.
The article, by Peter H. Stone, MD, and Peter Libby, MD, Brigham and Women’s Hospital, Boston, and William E. Boden, MD, Boston University School of Medicine, was published online in JAMA Cardiology.
The review explored new data from vascular biology, atherosclerosis imaging, natural history outcome studies, and large-scale clinical trials that support what the authors refer to as “The Plaque Hypothesis” – the idea that major adverse cardiac events such as myocardial infarction and cardiac death are triggered by destabilization of vulnerable plaque, which may be obstructive or nonobstructive.
“We need to consider embracing a new management strategy that directs our diagnostic and management focus to evaluate the entire length of the atheromatous coronary artery and broaden the target of our therapeutic intervention to include all regions of the plaque (both flow-limiting and non–flow-limiting), even those that are distant from the presumed ischemia-producing obstruction,” the authors concluded.
Dr. Stone explained to this news organization that, for several decades, the medical community has focused on plaques causing severe obstruction of coronary arteries as being responsible for major adverse cardiac events. This approach – known as the Ischemia Hypothesis – has been the accepted strategy for many years, with all guidelines advising the identification of the stenoses that cause the most obstruction for treatment with stenting.
However, the authors pointed out that a number of studies have now suggested that, while these severe obstructive stenoses cause angina, they do not seem to be responsible for the hard events of MI, acute coronary syndrome (ACS), and cardiac death.
Several studies including the COURAGE trial and BARI-2D, and the recent ISCHEMIA trial have all failed to show a reduction in these hard endpoints by intervening on these severe obstructive lesions, Dr. Stone noted.
“We present evidence for a new approach – that it is the composition and vascular biology of the atherosclerotic plaques that cause MI, ACS, and cardiac death, rather than simply how obstructive they are,” he said.
Dr. Stone pointed out that plaque seen on a coronary angiogram looks at only the lumen of the artery, but plaque is primarily based in the wall of the artery, and if that plaque is inflamed it can easily be the culprit responsible for adverse events even without encroaching into the lumen.
“Our paper describes many factors which can cause plaques to destabilize and cause an ACS. These include anatomical, biochemical, and biomechanical features that together cause plaque rupture or erosion and precipitate a clinical event. It is not sufficient to just look for obstructive plaques on a coronary angiogram,” he said. “We are barking up the wrong tree. We need to look for inflamed plaque in the whole wall of the coronary arteries.”
The authors described different factors that identify a plaque at high risk of destabilization. These include a large area of vulnerable plaque, a thin-cap atheroma, a severe inflamed core, macrocalcifications, a large plaque burden, and a physical profile that would encourage a thrombus to become trapped.
“Atherosclerotic plaques are very heterogeneous and complex structures and it is not just the mountain peaks but also the lower foothills that can precipitate a flow-limiting obstruction,” Dr. Stone noted.
“The slope of the mountain is probably very important in the ability for a thrombus to form. If the slope is gradual there isn’t a problem. But if the slope is jagged with sharp edges this can cause a thrombus to become trapped. We need to look at the entirety of plaque and all its risk features to identify the culprit areas that could cause MI or cardiac death. These are typically not the obstructive plaques we have all been fixated on for many years,” he added.
“We need to focus on plaque heterogeneity. Once plaque is old and just made up of scar tissue which is not inflamed it does not cause much [of] a problem – we can probably just leave it alone. Some of these obstructive plaques may cause some angina but many do not cause major cardiac events unless they have other high-risk features,” he said.
“Cardiac events are still caused by obstruction of blood flow but that can be an abrupt process where a thrombus attaches itself to an area of destabilized plaque. These areas of plaque were not necessarily obstructing to start with. We believe that this is the explanation behind the observation that 50% of all people who have an MI (half of which are fatal) do not have symptoms beforehand,” Dr. Stone commented.
Because these areas of destabilized plaque do not cause symptoms, he believes that vast populations of people with established cardiovascular risk factors should undergo screening. “At the moment we wait for people to experience chest pain or to have an MI – that is far too little too late.”
To identify these areas of high-risk plaques, imaging techniques looking inside the artery wall are needed such as intravascular ultrasound. However, this is an invasive procedure, and the noninvasive coronary CT angiography also gives a good picture, so it is probably the best way to begin as a wider screening modality, with more invasive screening methods then used in those found to be at risk, Dr. Stone suggested.
Plaques that are identified as likely to destabilize can be treated with percutaneous coronary intervention and stenting.
While systemic therapies are useful, those currently available are not sufficient, Dr. Stone noted. For example, there are still high levels of major cardiac events in patients treated with the PCSK9 inhibitors, which bring about very large reductions in LDL cholesterol. “These therapies are beneficial, but they are not enough on their own. So, these areas of unstable plaque would need to be treated with stenting or something similar. We believe that the intervention of stenting is good but at present it is targeted at the wrong areas,” he stated.
“Clearly what we’ve been doing – stenting only obstructive lesions – does not reduce hard clinical events. Imaging methods have improved so much in recent years that we can now identify high-risk areas of plaque. This whole field of studying the vulnerable plaque has been ongoing for many years, but it is only recently that imaging methods have become good enough to identify plaques at risk. This field is now coming of age,” he added.
The next steps are to start identifying these plaques in larger populations, more accurately characterizing those at the highest risk, and then performing randomized trials of preemptive intervention in those believed to be at highest risk, and follow up for clinical events, Dr. Stone explained.
Advances in detecting unstable plaque may also permit early evaluation of novel therapeutics and gauge the intensity of lifestyle and disease-modifying pharmacotherapy, the authors suggested.
This work was supported in part by the National Heart, Lung, and Blood Institute, the American Heart Association, the RRM Charitable Fund, the Simard Fund, and the Schaubert Family. Dr. Libby is an unpaid consultant to or involved in clinical trials with Amgen, AstraZeneca, Baim Institute, Beren Therapeutics, Esperion Therapeutics, Genentech, Kancera, Kowa Pharmaceuticals, MedImmune, Merck, Norvo Nordisk, Novartis, Pfizer, and Sanofi-Regeneron; and is a member of the scientific advisory board for Amgen, Caristo Diagnostics, Cartesian Therapeutics, CSL Behring, DalCor Pharmaceuticals, Dewpoint Therapeutics, Elucid Bioimaging, Kancera, Kowa Pharmaceuticals, Olatec Therapeutics, MedImmune, Moderna, Novartis, PlaqueTec, TenSixteen Bio, Soley Thereapeutics, and XBiotech.
A version of this article first appeared on Medscape.com.
A new strategy for the management of atherosclerotic plaque as a source of major adverse cardiac events is needed with the focus shifting from the flow-limiting coronary artery luminal lesions to the overall atherosclerotic burden, be it obstructive or nonobstructive, according to a review article.
The article, by Peter H. Stone, MD, and Peter Libby, MD, Brigham and Women’s Hospital, Boston, and William E. Boden, MD, Boston University School of Medicine, was published online in JAMA Cardiology.
The review explored new data from vascular biology, atherosclerosis imaging, natural history outcome studies, and large-scale clinical trials that support what the authors refer to as “The Plaque Hypothesis” – the idea that major adverse cardiac events such as myocardial infarction and cardiac death are triggered by destabilization of vulnerable plaque, which may be obstructive or nonobstructive.
“We need to consider embracing a new management strategy that directs our diagnostic and management focus to evaluate the entire length of the atheromatous coronary artery and broaden the target of our therapeutic intervention to include all regions of the plaque (both flow-limiting and non–flow-limiting), even those that are distant from the presumed ischemia-producing obstruction,” the authors concluded.
Dr. Stone explained to this news organization that, for several decades, the medical community has focused on plaques causing severe obstruction of coronary arteries as being responsible for major adverse cardiac events. This approach – known as the Ischemia Hypothesis – has been the accepted strategy for many years, with all guidelines advising the identification of the stenoses that cause the most obstruction for treatment with stenting.
However, the authors pointed out that a number of studies have now suggested that, while these severe obstructive stenoses cause angina, they do not seem to be responsible for the hard events of MI, acute coronary syndrome (ACS), and cardiac death.
Several studies including the COURAGE trial and BARI-2D, and the recent ISCHEMIA trial have all failed to show a reduction in these hard endpoints by intervening on these severe obstructive lesions, Dr. Stone noted.
“We present evidence for a new approach – that it is the composition and vascular biology of the atherosclerotic plaques that cause MI, ACS, and cardiac death, rather than simply how obstructive they are,” he said.
Dr. Stone pointed out that plaque seen on a coronary angiogram looks at only the lumen of the artery, but plaque is primarily based in the wall of the artery, and if that plaque is inflamed it can easily be the culprit responsible for adverse events even without encroaching into the lumen.
“Our paper describes many factors which can cause plaques to destabilize and cause an ACS. These include anatomical, biochemical, and biomechanical features that together cause plaque rupture or erosion and precipitate a clinical event. It is not sufficient to just look for obstructive plaques on a coronary angiogram,” he said. “We are barking up the wrong tree. We need to look for inflamed plaque in the whole wall of the coronary arteries.”
The authors described different factors that identify a plaque at high risk of destabilization. These include a large area of vulnerable plaque, a thin-cap atheroma, a severe inflamed core, macrocalcifications, a large plaque burden, and a physical profile that would encourage a thrombus to become trapped.
“Atherosclerotic plaques are very heterogeneous and complex structures and it is not just the mountain peaks but also the lower foothills that can precipitate a flow-limiting obstruction,” Dr. Stone noted.
“The slope of the mountain is probably very important in the ability for a thrombus to form. If the slope is gradual there isn’t a problem. But if the slope is jagged with sharp edges this can cause a thrombus to become trapped. We need to look at the entirety of plaque and all its risk features to identify the culprit areas that could cause MI or cardiac death. These are typically not the obstructive plaques we have all been fixated on for many years,” he added.
“We need to focus on plaque heterogeneity. Once plaque is old and just made up of scar tissue which is not inflamed it does not cause much [of] a problem – we can probably just leave it alone. Some of these obstructive plaques may cause some angina but many do not cause major cardiac events unless they have other high-risk features,” he said.
“Cardiac events are still caused by obstruction of blood flow but that can be an abrupt process where a thrombus attaches itself to an area of destabilized plaque. These areas of plaque were not necessarily obstructing to start with. We believe that this is the explanation behind the observation that 50% of all people who have an MI (half of which are fatal) do not have symptoms beforehand,” Dr. Stone commented.
Because these areas of destabilized plaque do not cause symptoms, he believes that vast populations of people with established cardiovascular risk factors should undergo screening. “At the moment we wait for people to experience chest pain or to have an MI – that is far too little too late.”
To identify these areas of high-risk plaques, imaging techniques looking inside the artery wall are needed such as intravascular ultrasound. However, this is an invasive procedure, and the noninvasive coronary CT angiography also gives a good picture, so it is probably the best way to begin as a wider screening modality, with more invasive screening methods then used in those found to be at risk, Dr. Stone suggested.
Plaques that are identified as likely to destabilize can be treated with percutaneous coronary intervention and stenting.
While systemic therapies are useful, those currently available are not sufficient, Dr. Stone noted. For example, there are still high levels of major cardiac events in patients treated with the PCSK9 inhibitors, which bring about very large reductions in LDL cholesterol. “These therapies are beneficial, but they are not enough on their own. So, these areas of unstable plaque would need to be treated with stenting or something similar. We believe that the intervention of stenting is good but at present it is targeted at the wrong areas,” he stated.
“Clearly what we’ve been doing – stenting only obstructive lesions – does not reduce hard clinical events. Imaging methods have improved so much in recent years that we can now identify high-risk areas of plaque. This whole field of studying the vulnerable plaque has been ongoing for many years, but it is only recently that imaging methods have become good enough to identify plaques at risk. This field is now coming of age,” he added.
The next steps are to start identifying these plaques in larger populations, more accurately characterizing those at the highest risk, and then performing randomized trials of preemptive intervention in those believed to be at highest risk, and follow up for clinical events, Dr. Stone explained.
Advances in detecting unstable plaque may also permit early evaluation of novel therapeutics and gauge the intensity of lifestyle and disease-modifying pharmacotherapy, the authors suggested.
This work was supported in part by the National Heart, Lung, and Blood Institute, the American Heart Association, the RRM Charitable Fund, the Simard Fund, and the Schaubert Family. Dr. Libby is an unpaid consultant to or involved in clinical trials with Amgen, AstraZeneca, Baim Institute, Beren Therapeutics, Esperion Therapeutics, Genentech, Kancera, Kowa Pharmaceuticals, MedImmune, Merck, Norvo Nordisk, Novartis, Pfizer, and Sanofi-Regeneron; and is a member of the scientific advisory board for Amgen, Caristo Diagnostics, Cartesian Therapeutics, CSL Behring, DalCor Pharmaceuticals, Dewpoint Therapeutics, Elucid Bioimaging, Kancera, Kowa Pharmaceuticals, Olatec Therapeutics, MedImmune, Moderna, Novartis, PlaqueTec, TenSixteen Bio, Soley Thereapeutics, and XBiotech.
A version of this article first appeared on Medscape.com.
New AHA statement on managing ACS in older adults
Age-related changes in general and cardiovascular health likely require modifications in how acute coronary syndrome (ACS) is diagnosed and managed in adults aged 75 and older, the American Heart Association says in a new scientific statement.
The statement outlines a framework to integrate geriatric risks into the management of ACS, including the diagnostic approach, pharmacotherapy, revascularization strategies, prevention of adverse events, and transition care planning.
The 31-page statement was published online in the AHA journal Circulation (2022 Dec 12. doi: 10.1161/CIR.0000000000001112). It updates a 2007 AHA statement on treatment of ACS in the elderly.
Complex patient group
Adults aged 75 and older make up roughly 30%-40% of all hospitalized patients with ACS and the majority of ACS-related deaths occur in this group, the writing group notes.
“Older patients have more pronounced anatomical changes and more severe functional impairment, and they are more likely to have additional health conditions,” writing group chair Abdulla A. Damluji, MD, PhD, director of the Inova Center of Outcomes Research in Fairfax, Va., notes in a news release.
“These include frailty, other chronic disorders (treated with multiple medications), physical dysfunction, cognitive decline and/or urinary incontinence – and these are not regularly studied in the context of ACS,” Dr. Damluji explained.
The writing group notes that the presence of one or more geriatric syndromes may substantially affect ACS clinical presentation, clinical course and prognosis, therapeutic decision-making, and response to treatment.
“It is therefore fundamental that clinicians caring for older patients with ACS be alert to the presence of geriatric syndromes and be able to integrate them into the care plan when appropriate,” they say.
They recommend a holistic, individualized, and patient-centered approach to ACS care in the elderly, taking into consideration coexisting and overlapping health issues.
Considerations for clinical care
The AHA statement offers several “considerations for clinical practice” with regard to ACS diagnosis and management in elderly adults. They include:
- ACS presentations without chest pain, such as shortness of breath, syncope, or sudden confusion, are more common in older adults.
- Many older adults have persistent elevations in cardiac troponin levels from myocardial fibrosis and kidney disease that diminish the positive predictive value of high-sensitivity cardiac troponin (hs-cTn) assays for identifying acute and chronic myocardial injury. For this reason, evaluating patterns of rise and fall is essential.
- Age-related changes in metabolism, weight, and muscle mass may require different choices in anticoagulant medications to lower bleeding risk.
- Clopidogrel (Plavix) is the preferred P2Y12 inhibitor because of a significantly lower bleeding profile than ticagrelor (Brilinta) or prasugrel (Effient). For patients with ST-segment myocardial infarction (STEMI) or complex anatomy, the use of ticagrelor is “reasonable.”
- Poor kidney function can increase the risk for contrast-induced acute kidney injury.
- Although the risks are greater, percutaneous coronary intervention or bypass surgery are beneficial in select older adults with ACS.
- Post-MI care should include cardiac rehabilitation tailored to address each patient’s circumstances and personal goals of care.
- For patients with cognitive difficulties and limited mobility, consider simplified medication plans with fewer doses per day and 90-day supplies to prevent the need for frequent refills.
- Patient care plans should be individualized, with input from a multidisciplinary team that may include cardiologists, surgeons, geriatricians, primary care clinicians, nutritionists, social workers, and family members.
- Determine a priori goals of care in older patients to help avoid an unwanted or futile intervention.
This scientific statement was prepared by the volunteer writing group on behalf of the AHA Cardiovascular Diseases in Older Populations Committee of the Council on Clinical Cardiology; the Council on Cardiovascular and Stroke Nursing; the Council on Cardiovascular Radiology and Intervention; and the Council on Lifestyle and Cardiometabolic Health.
A version of this article first appeared on Medscape.com.
Age-related changes in general and cardiovascular health likely require modifications in how acute coronary syndrome (ACS) is diagnosed and managed in adults aged 75 and older, the American Heart Association says in a new scientific statement.
The statement outlines a framework to integrate geriatric risks into the management of ACS, including the diagnostic approach, pharmacotherapy, revascularization strategies, prevention of adverse events, and transition care planning.
The 31-page statement was published online in the AHA journal Circulation (2022 Dec 12. doi: 10.1161/CIR.0000000000001112). It updates a 2007 AHA statement on treatment of ACS in the elderly.
Complex patient group
Adults aged 75 and older make up roughly 30%-40% of all hospitalized patients with ACS and the majority of ACS-related deaths occur in this group, the writing group notes.
“Older patients have more pronounced anatomical changes and more severe functional impairment, and they are more likely to have additional health conditions,” writing group chair Abdulla A. Damluji, MD, PhD, director of the Inova Center of Outcomes Research in Fairfax, Va., notes in a news release.
“These include frailty, other chronic disorders (treated with multiple medications), physical dysfunction, cognitive decline and/or urinary incontinence – and these are not regularly studied in the context of ACS,” Dr. Damluji explained.
The writing group notes that the presence of one or more geriatric syndromes may substantially affect ACS clinical presentation, clinical course and prognosis, therapeutic decision-making, and response to treatment.
“It is therefore fundamental that clinicians caring for older patients with ACS be alert to the presence of geriatric syndromes and be able to integrate them into the care plan when appropriate,” they say.
They recommend a holistic, individualized, and patient-centered approach to ACS care in the elderly, taking into consideration coexisting and overlapping health issues.
Considerations for clinical care
The AHA statement offers several “considerations for clinical practice” with regard to ACS diagnosis and management in elderly adults. They include:
- ACS presentations without chest pain, such as shortness of breath, syncope, or sudden confusion, are more common in older adults.
- Many older adults have persistent elevations in cardiac troponin levels from myocardial fibrosis and kidney disease that diminish the positive predictive value of high-sensitivity cardiac troponin (hs-cTn) assays for identifying acute and chronic myocardial injury. For this reason, evaluating patterns of rise and fall is essential.
- Age-related changes in metabolism, weight, and muscle mass may require different choices in anticoagulant medications to lower bleeding risk.
- Clopidogrel (Plavix) is the preferred P2Y12 inhibitor because of a significantly lower bleeding profile than ticagrelor (Brilinta) or prasugrel (Effient). For patients with ST-segment myocardial infarction (STEMI) or complex anatomy, the use of ticagrelor is “reasonable.”
- Poor kidney function can increase the risk for contrast-induced acute kidney injury.
- Although the risks are greater, percutaneous coronary intervention or bypass surgery are beneficial in select older adults with ACS.
- Post-MI care should include cardiac rehabilitation tailored to address each patient’s circumstances and personal goals of care.
- For patients with cognitive difficulties and limited mobility, consider simplified medication plans with fewer doses per day and 90-day supplies to prevent the need for frequent refills.
- Patient care plans should be individualized, with input from a multidisciplinary team that may include cardiologists, surgeons, geriatricians, primary care clinicians, nutritionists, social workers, and family members.
- Determine a priori goals of care in older patients to help avoid an unwanted or futile intervention.
This scientific statement was prepared by the volunteer writing group on behalf of the AHA Cardiovascular Diseases in Older Populations Committee of the Council on Clinical Cardiology; the Council on Cardiovascular and Stroke Nursing; the Council on Cardiovascular Radiology and Intervention; and the Council on Lifestyle and Cardiometabolic Health.
A version of this article first appeared on Medscape.com.
Age-related changes in general and cardiovascular health likely require modifications in how acute coronary syndrome (ACS) is diagnosed and managed in adults aged 75 and older, the American Heart Association says in a new scientific statement.
The statement outlines a framework to integrate geriatric risks into the management of ACS, including the diagnostic approach, pharmacotherapy, revascularization strategies, prevention of adverse events, and transition care planning.
The 31-page statement was published online in the AHA journal Circulation (2022 Dec 12. doi: 10.1161/CIR.0000000000001112). It updates a 2007 AHA statement on treatment of ACS in the elderly.
Complex patient group
Adults aged 75 and older make up roughly 30%-40% of all hospitalized patients with ACS and the majority of ACS-related deaths occur in this group, the writing group notes.
“Older patients have more pronounced anatomical changes and more severe functional impairment, and they are more likely to have additional health conditions,” writing group chair Abdulla A. Damluji, MD, PhD, director of the Inova Center of Outcomes Research in Fairfax, Va., notes in a news release.
“These include frailty, other chronic disorders (treated with multiple medications), physical dysfunction, cognitive decline and/or urinary incontinence – and these are not regularly studied in the context of ACS,” Dr. Damluji explained.
The writing group notes that the presence of one or more geriatric syndromes may substantially affect ACS clinical presentation, clinical course and prognosis, therapeutic decision-making, and response to treatment.
“It is therefore fundamental that clinicians caring for older patients with ACS be alert to the presence of geriatric syndromes and be able to integrate them into the care plan when appropriate,” they say.
They recommend a holistic, individualized, and patient-centered approach to ACS care in the elderly, taking into consideration coexisting and overlapping health issues.
Considerations for clinical care
The AHA statement offers several “considerations for clinical practice” with regard to ACS diagnosis and management in elderly adults. They include:
- ACS presentations without chest pain, such as shortness of breath, syncope, or sudden confusion, are more common in older adults.
- Many older adults have persistent elevations in cardiac troponin levels from myocardial fibrosis and kidney disease that diminish the positive predictive value of high-sensitivity cardiac troponin (hs-cTn) assays for identifying acute and chronic myocardial injury. For this reason, evaluating patterns of rise and fall is essential.
- Age-related changes in metabolism, weight, and muscle mass may require different choices in anticoagulant medications to lower bleeding risk.
- Clopidogrel (Plavix) is the preferred P2Y12 inhibitor because of a significantly lower bleeding profile than ticagrelor (Brilinta) or prasugrel (Effient). For patients with ST-segment myocardial infarction (STEMI) or complex anatomy, the use of ticagrelor is “reasonable.”
- Poor kidney function can increase the risk for contrast-induced acute kidney injury.
- Although the risks are greater, percutaneous coronary intervention or bypass surgery are beneficial in select older adults with ACS.
- Post-MI care should include cardiac rehabilitation tailored to address each patient’s circumstances and personal goals of care.
- For patients with cognitive difficulties and limited mobility, consider simplified medication plans with fewer doses per day and 90-day supplies to prevent the need for frequent refills.
- Patient care plans should be individualized, with input from a multidisciplinary team that may include cardiologists, surgeons, geriatricians, primary care clinicians, nutritionists, social workers, and family members.
- Determine a priori goals of care in older patients to help avoid an unwanted or futile intervention.
This scientific statement was prepared by the volunteer writing group on behalf of the AHA Cardiovascular Diseases in Older Populations Committee of the Council on Clinical Cardiology; the Council on Cardiovascular and Stroke Nursing; the Council on Cardiovascular Radiology and Intervention; and the Council on Lifestyle and Cardiometabolic Health.
A version of this article first appeared on Medscape.com.
Severe OSA tied to poor prognoses in stroke patients
Patients with acute ischemic stroke had a worse prognosis if they had also experienced severe obstructive sleep apnea (OSA), based on data from 125 individuals.
OSA is on the rise, and is associated with pathophysiological changes, and data from previous studies suggest that severe OSA doubles the risk of stroke and increases risk of stroke recurrence, according to Juan Xu, PhD, of Soochow University, Suzhou, China, and colleagues.
“There is a high comorbidity between stroke and OSA,” and effective sleep is important to cerebral function recovery, the researchers wrote. Early prediction of stroke prognosis may inform treatment in stroke patients, but the value of OSA as a predictor of functional prognosis has not been explored.
In a study published in Sleep Medicine, the researchers analyzed data from 125 adults with mild to moderate ischemic stroke and OSA. The participants underwent polysomnography within a week of stroke onset between January 2015 and June 2020 and were grouped by severity according to apnea-hypopnea index (AHI) of either less than 30/h (not severe) or 30/h or higher (severe). The mean age of the patients was 58 years, and 87% were men. Approximately one-third of the participants met the criteria for severe OSA.
The researchers assessed the impact of OSA on functional prognosis in the acute phase of stroke, and reviewed quantitative electroencephalography (EEG) markers in stroke patients during sleep.
Overall, individuals with severe OSA were significantly more likely than those with less severe OSA to have comorbid hypertension (85.4% vs. 56%; P = .002) and a higher body mass index (28 vs. 24; P < .001). Other factors including blood pressure, smoking history, alcohol use, and comorbid diabetes were similar between the groups.
Quantitative EEG among patients with severe OSA showed lower relative power of high-frequency bands (alpha, beta, and sigma). The EEG also showed higher delta/alpha power ratio and slowing ratio, and higher delta relative power (delta RP) in severe OSA (P < .05 for all).
In addition, severe OSA was associated with more than triple the risk (3.6-fold increase) of poor prognosis, defined as a Modified Rankin Scale score of 3 or higher (24.4% for severe OSA vs. 8.3% for nonsevere OSA; P = .03).
the researchers wrote. “Integrating the alteration of quantitative EEG parameters may improve the accuracy of early predictions of functional prognosis in patients with stroke.”
The findings were limited by several factors including the retrospective design and the lack of a sizable non-OSA control group, the researchers noted. Other limitations included the use of an AHI of 30/h or higher to define severity and the use of data from medical histories, with the potential for information bias, and the use of only 30-second continuous polysomnography segments.
However, the results suggest that increased delta RP and TSR, and decreased alpha, beta, and sigma RP, may be independent predictors of a poor functional prognosis in stroke patients with OSA, and that the prognosis could be improved by treating the OSA, they concluded.
The study was supported by the Natural Science Foundation of China and the Discipline Construction Program of the Second Affiliated Hospital of Soochow University. The researchers reported no financial conflicts.
A version of this article first appeared on Medscape.com.
Patients with acute ischemic stroke had a worse prognosis if they had also experienced severe obstructive sleep apnea (OSA), based on data from 125 individuals.
OSA is on the rise, and is associated with pathophysiological changes, and data from previous studies suggest that severe OSA doubles the risk of stroke and increases risk of stroke recurrence, according to Juan Xu, PhD, of Soochow University, Suzhou, China, and colleagues.
“There is a high comorbidity between stroke and OSA,” and effective sleep is important to cerebral function recovery, the researchers wrote. Early prediction of stroke prognosis may inform treatment in stroke patients, but the value of OSA as a predictor of functional prognosis has not been explored.
In a study published in Sleep Medicine, the researchers analyzed data from 125 adults with mild to moderate ischemic stroke and OSA. The participants underwent polysomnography within a week of stroke onset between January 2015 and June 2020 and were grouped by severity according to apnea-hypopnea index (AHI) of either less than 30/h (not severe) or 30/h or higher (severe). The mean age of the patients was 58 years, and 87% were men. Approximately one-third of the participants met the criteria for severe OSA.
The researchers assessed the impact of OSA on functional prognosis in the acute phase of stroke, and reviewed quantitative electroencephalography (EEG) markers in stroke patients during sleep.
Overall, individuals with severe OSA were significantly more likely than those with less severe OSA to have comorbid hypertension (85.4% vs. 56%; P = .002) and a higher body mass index (28 vs. 24; P < .001). Other factors including blood pressure, smoking history, alcohol use, and comorbid diabetes were similar between the groups.
Quantitative EEG among patients with severe OSA showed lower relative power of high-frequency bands (alpha, beta, and sigma). The EEG also showed higher delta/alpha power ratio and slowing ratio, and higher delta relative power (delta RP) in severe OSA (P < .05 for all).
In addition, severe OSA was associated with more than triple the risk (3.6-fold increase) of poor prognosis, defined as a Modified Rankin Scale score of 3 or higher (24.4% for severe OSA vs. 8.3% for nonsevere OSA; P = .03).
the researchers wrote. “Integrating the alteration of quantitative EEG parameters may improve the accuracy of early predictions of functional prognosis in patients with stroke.”
The findings were limited by several factors including the retrospective design and the lack of a sizable non-OSA control group, the researchers noted. Other limitations included the use of an AHI of 30/h or higher to define severity and the use of data from medical histories, with the potential for information bias, and the use of only 30-second continuous polysomnography segments.
However, the results suggest that increased delta RP and TSR, and decreased alpha, beta, and sigma RP, may be independent predictors of a poor functional prognosis in stroke patients with OSA, and that the prognosis could be improved by treating the OSA, they concluded.
The study was supported by the Natural Science Foundation of China and the Discipline Construction Program of the Second Affiliated Hospital of Soochow University. The researchers reported no financial conflicts.
A version of this article first appeared on Medscape.com.
Patients with acute ischemic stroke had a worse prognosis if they had also experienced severe obstructive sleep apnea (OSA), based on data from 125 individuals.
OSA is on the rise, and is associated with pathophysiological changes, and data from previous studies suggest that severe OSA doubles the risk of stroke and increases risk of stroke recurrence, according to Juan Xu, PhD, of Soochow University, Suzhou, China, and colleagues.
“There is a high comorbidity between stroke and OSA,” and effective sleep is important to cerebral function recovery, the researchers wrote. Early prediction of stroke prognosis may inform treatment in stroke patients, but the value of OSA as a predictor of functional prognosis has not been explored.
In a study published in Sleep Medicine, the researchers analyzed data from 125 adults with mild to moderate ischemic stroke and OSA. The participants underwent polysomnography within a week of stroke onset between January 2015 and June 2020 and were grouped by severity according to apnea-hypopnea index (AHI) of either less than 30/h (not severe) or 30/h or higher (severe). The mean age of the patients was 58 years, and 87% were men. Approximately one-third of the participants met the criteria for severe OSA.
The researchers assessed the impact of OSA on functional prognosis in the acute phase of stroke, and reviewed quantitative electroencephalography (EEG) markers in stroke patients during sleep.
Overall, individuals with severe OSA were significantly more likely than those with less severe OSA to have comorbid hypertension (85.4% vs. 56%; P = .002) and a higher body mass index (28 vs. 24; P < .001). Other factors including blood pressure, smoking history, alcohol use, and comorbid diabetes were similar between the groups.
Quantitative EEG among patients with severe OSA showed lower relative power of high-frequency bands (alpha, beta, and sigma). The EEG also showed higher delta/alpha power ratio and slowing ratio, and higher delta relative power (delta RP) in severe OSA (P < .05 for all).
In addition, severe OSA was associated with more than triple the risk (3.6-fold increase) of poor prognosis, defined as a Modified Rankin Scale score of 3 or higher (24.4% for severe OSA vs. 8.3% for nonsevere OSA; P = .03).
the researchers wrote. “Integrating the alteration of quantitative EEG parameters may improve the accuracy of early predictions of functional prognosis in patients with stroke.”
The findings were limited by several factors including the retrospective design and the lack of a sizable non-OSA control group, the researchers noted. Other limitations included the use of an AHI of 30/h or higher to define severity and the use of data from medical histories, with the potential for information bias, and the use of only 30-second continuous polysomnography segments.
However, the results suggest that increased delta RP and TSR, and decreased alpha, beta, and sigma RP, may be independent predictors of a poor functional prognosis in stroke patients with OSA, and that the prognosis could be improved by treating the OSA, they concluded.
The study was supported by the Natural Science Foundation of China and the Discipline Construction Program of the Second Affiliated Hospital of Soochow University. The researchers reported no financial conflicts.
A version of this article first appeared on Medscape.com.
FROM SLEEP MEDICINE
Multiple myeloma diagnosed more via emergency care during COVID
The study covered in this summary was published on Research Square as a preprint and has not yet been peer reviewed.
Key takeaway
Why this matters
While trying to avoid COVID-19 infection, patients ultimately diagnosed with multiple myeloma may have delayed interactions with healthcare professionals and consequently delayed their cancer diagnosis.
Study design
Researchers collected data on newly diagnosed patients with multiple myeloma from January 2019 until July 2021 across five institutions (three universities and two hospitals) in England. In total, 323 patients with multiple myeloma were identified.
Patients were divided into two groups: those diagnosed between Jan. 1, 2019, until Jan. 31, 2020, or pre-COVID, and those diagnosed from Feb. 1, 2020, to July 31, 2021, or post COVID.
Key results
Among all patients, 80 (24.8%) were diagnosed with smoldering multiple myeloma and 243 (75.2%) were diagnosed with multiple myeloma requiring treatment.
Significantly more patients in the post-COVID group were diagnosed with myeloma through the emergency route (45.5% post COVID vs. 32.7% pre-COVID; P = .03).
Clinical complications leading to emergency admission prior to a myeloma diagnosis also differed between the two cohorts: Acute kidney injury accounted for most emergency admissions in the pre-COVID cohort while skeletal-related events, including spinal cord compression, were the major causes for diagnosis through the emergency route in the post-COVID cohort.
Patients who were diagnosed with symptomatic myeloma pre-COVID were more likely to be treated with a triplet rather than doublet combination compared with those diagnosed in the post-COVID period (triplet pre-COVID 79.1%, post COVID 63.75%; P = .014).
Overall survival at 1 year was not significantly different between the pre-COVID and post-COVID groups: 88.2% pre-COVID, compared with 87.8% post COVID.
Overall, the authors concluded that the COVID pandemic “resulted in a shift in the symptomatology, disease burden, and routes of diagnosis of patients presenting with myeloma” and “this may have significant consequences” over the long term.
Limitations
The study does not provide a clear time frame of delays in diagnosis.
Disclosures
The study authors did not report any conflicts of interest.
A version of this article first appeared on Medscape.com .
The study covered in this summary was published on Research Square as a preprint and has not yet been peer reviewed.
Key takeaway
Why this matters
While trying to avoid COVID-19 infection, patients ultimately diagnosed with multiple myeloma may have delayed interactions with healthcare professionals and consequently delayed their cancer diagnosis.
Study design
Researchers collected data on newly diagnosed patients with multiple myeloma from January 2019 until July 2021 across five institutions (three universities and two hospitals) in England. In total, 323 patients with multiple myeloma were identified.
Patients were divided into two groups: those diagnosed between Jan. 1, 2019, until Jan. 31, 2020, or pre-COVID, and those diagnosed from Feb. 1, 2020, to July 31, 2021, or post COVID.
Key results
Among all patients, 80 (24.8%) were diagnosed with smoldering multiple myeloma and 243 (75.2%) were diagnosed with multiple myeloma requiring treatment.
Significantly more patients in the post-COVID group were diagnosed with myeloma through the emergency route (45.5% post COVID vs. 32.7% pre-COVID; P = .03).
Clinical complications leading to emergency admission prior to a myeloma diagnosis also differed between the two cohorts: Acute kidney injury accounted for most emergency admissions in the pre-COVID cohort while skeletal-related events, including spinal cord compression, were the major causes for diagnosis through the emergency route in the post-COVID cohort.
Patients who were diagnosed with symptomatic myeloma pre-COVID were more likely to be treated with a triplet rather than doublet combination compared with those diagnosed in the post-COVID period (triplet pre-COVID 79.1%, post COVID 63.75%; P = .014).
Overall survival at 1 year was not significantly different between the pre-COVID and post-COVID groups: 88.2% pre-COVID, compared with 87.8% post COVID.
Overall, the authors concluded that the COVID pandemic “resulted in a shift in the symptomatology, disease burden, and routes of diagnosis of patients presenting with myeloma” and “this may have significant consequences” over the long term.
Limitations
The study does not provide a clear time frame of delays in diagnosis.
Disclosures
The study authors did not report any conflicts of interest.
A version of this article first appeared on Medscape.com .
The study covered in this summary was published on Research Square as a preprint and has not yet been peer reviewed.
Key takeaway
Why this matters
While trying to avoid COVID-19 infection, patients ultimately diagnosed with multiple myeloma may have delayed interactions with healthcare professionals and consequently delayed their cancer diagnosis.
Study design
Researchers collected data on newly diagnosed patients with multiple myeloma from January 2019 until July 2021 across five institutions (three universities and two hospitals) in England. In total, 323 patients with multiple myeloma were identified.
Patients were divided into two groups: those diagnosed between Jan. 1, 2019, until Jan. 31, 2020, or pre-COVID, and those diagnosed from Feb. 1, 2020, to July 31, 2021, or post COVID.
Key results
Among all patients, 80 (24.8%) were diagnosed with smoldering multiple myeloma and 243 (75.2%) were diagnosed with multiple myeloma requiring treatment.
Significantly more patients in the post-COVID group were diagnosed with myeloma through the emergency route (45.5% post COVID vs. 32.7% pre-COVID; P = .03).
Clinical complications leading to emergency admission prior to a myeloma diagnosis also differed between the two cohorts: Acute kidney injury accounted for most emergency admissions in the pre-COVID cohort while skeletal-related events, including spinal cord compression, were the major causes for diagnosis through the emergency route in the post-COVID cohort.
Patients who were diagnosed with symptomatic myeloma pre-COVID were more likely to be treated with a triplet rather than doublet combination compared with those diagnosed in the post-COVID period (triplet pre-COVID 79.1%, post COVID 63.75%; P = .014).
Overall survival at 1 year was not significantly different between the pre-COVID and post-COVID groups: 88.2% pre-COVID, compared with 87.8% post COVID.
Overall, the authors concluded that the COVID pandemic “resulted in a shift in the symptomatology, disease burden, and routes of diagnosis of patients presenting with myeloma” and “this may have significant consequences” over the long term.
Limitations
The study does not provide a clear time frame of delays in diagnosis.
Disclosures
The study authors did not report any conflicts of interest.
A version of this article first appeared on Medscape.com .
Rise of ‘alarming’ subvariants of COVID ‘worrisome’ for winter
It’s a story perhaps more appropriate for Halloween than for the festive holiday season, given its scary implications.
Not too dire so far, until the researchers’ other findings are considered.
The BQ.1, BQ1.1, XBB, and XBB.1 subvariants are the most resistant to neutralizing antibodies, researcher Qian Wang, PhD, and colleagues wrote in a study published online in the journal Cell. This means people have no or “markedly reduced” protection against infection from these four strains, even if they’ve already had COVID-19 or are vaccinated and boosted multiple times, including with a bivalent vaccine.
On top of that, all available monoclonal antibody treatments are mostly or completely ineffective against these subvariants.
What does that mean for the immediate future? The findings are definitely “worrisome,” said Eric Topol, MD, founder and director of the Scripps Translational Research Institute in La Jolla, Calif.
But evidence from other countries, specifically Singapore and France, show that at least two of these variants turned out not to be as damaging as expected, likely because of high numbers of people vaccinated or who survived previous infections, he said.
Still, there is little to celebrate in the new findings, except that COVID-19 vaccinations and prior infections can still reduce the risk for serious outcomes such as hospitalization and death, the researchers wrote.
In fact, Centers for Disease Control and Prevention data released on Dec. 16 shows that people who have received four shots of the original COVID-19 vaccines as well as the bivalent booster were 57% less likely to visit an urgent care clinic or emergency room, regardless of age.
It comes at a time when BQ.1 and BQ.1.1 account for about 70% of the circulating variants, data show. In addition, hospitalizations are up 18% over the past 2 weeks and COVID-19 deaths are up 50% nationwide, The New York Times reported.
Globally, in many places, an “immunity wall” that has been built, Dr. Topol said. That may not be the case in the United States.
“The problem in the United States, making it harder to predict, is that we have a very low rate of recent boosters, in the past 6 months, especially in seniors,” he said. For example, only 36% of Americans aged 65 years and older, the group with highest risk, have received an updated bivalent booster.
An evolving virus
The subvariants are successfully replacing BA.5, which reigned as one of the most common Omicron variants over the past year. The latest CDC data show that BA.5 now accounts for only about 10% of the circulating virus. The researchers wrote: “This rapid replacement of virus strains is raising the specter of yet another wave of infections in the coming months.”
BQ.1 and BQ.1.1 evolved directly from BA.5 – adding more and some novel mutations to the SARS-CoV-2 virus. XBB and XBB.1 are the “offspring” of a combination of two other strains, known as BJ.1 and BA.2.75.
The story sounds familiar to the researchers. “The rapid rise of these subvariants and their extensive array of spike mutations are reminiscent of the appearance of the first Omicron variant last year, thus raising concerns that they may further compromise the efficacy of current COVID-19 vaccines and monoclonal antibody therapeutics,” they wrote. “We now report findings that indicate that such concerns are, sadly, justified, especially so for the XBB and XBB.1 subvariants.”
To figure out how effective existing antibodies could be against these newer subvariants, Dr. Wang and colleagues used blood samples from five groups of people. They tested serum from people who had three doses of the original COVID-19 vaccine, four doses of the original vaccine, those who received a bivalent booster, people who experienced a breakthrough infection with the BA.2 Omicron variant, and those who had a breakthrough with a BA.4 or BA.5 variant.
Adding the new subvariants to these serum samples revealed that the existing antibodies in the blood were ineffective at wiping out or neutralizing BQ.1, BQ.1.1, XBB, and XBB.1.
The BQ.1 subvariant was six times more resistant to antibodies than BA.5, its parent strain, and XBB.1 was 63 times more resistant compared with its predecessor, BA.2.
This shift in the ability of vaccines to stop the subvariants “is particularly concerning,” the researchers wrote.
Wiping out treatments too
Dr. Wang and colleagues also tested how well a panel of 23 different monoclonal antibody drugs might work against the four subvariants. The therapies all worked well against the original Omicron variant and included some approved for use through the Food and Drug Administration emergency use authorization (EUA) program at the time of the study.
They found that 19 of these 23 monoclonal antibodies lost effectiveness “greatly or completely” against XBB and XBB.1, for example.
This is not the first time that monoclonal antibody therapies have gone from effective to ineffective. Previous variants have come out that no longer responded to treatment with bamlanivimab, etesevimab, imdevimab, casirivimab, tixagevimab, cilgavimab, and sotrovimab. Bebtelovimab now joins this list and is no longer available from Eli Lilly under EUA because of this lack of effectiveness.
The lack of an effective monoclonal antibody treatment “poses a serious problem for millions of immunocompromised individuals who do not respond robustly to COVID-19 vaccines,” the researchers wrote, adding that “the urgent need to develop active monoclonal antibodies for clinical use is obvious.”
A limitation of the study is that the work is done in blood samples. The effectiveness of COVID-19 vaccination against the BQ and XBB subvariants should be evaluated in people in clinical studies, the authors noted.
Also, the current study looked at how well antibodies could neutralize the viral strains, but future research, they added, should look at how well “cellular immunity” or other aspects of the immune system might protect people.
Going forward, the challenge remains to develop vaccines and treatments that offer broad protection as the coronavirus continues to evolve.
In an alarming ending, the researchers wrote: “We have collectively chased after SARS-CoV-2 variants for over 2 years, and yet, the virus continues to evolve and evade.”
A version of this article first appeared on Medscape.com.
It’s a story perhaps more appropriate for Halloween than for the festive holiday season, given its scary implications.
Not too dire so far, until the researchers’ other findings are considered.
The BQ.1, BQ1.1, XBB, and XBB.1 subvariants are the most resistant to neutralizing antibodies, researcher Qian Wang, PhD, and colleagues wrote in a study published online in the journal Cell. This means people have no or “markedly reduced” protection against infection from these four strains, even if they’ve already had COVID-19 or are vaccinated and boosted multiple times, including with a bivalent vaccine.
On top of that, all available monoclonal antibody treatments are mostly or completely ineffective against these subvariants.
What does that mean for the immediate future? The findings are definitely “worrisome,” said Eric Topol, MD, founder and director of the Scripps Translational Research Institute in La Jolla, Calif.
But evidence from other countries, specifically Singapore and France, show that at least two of these variants turned out not to be as damaging as expected, likely because of high numbers of people vaccinated or who survived previous infections, he said.
Still, there is little to celebrate in the new findings, except that COVID-19 vaccinations and prior infections can still reduce the risk for serious outcomes such as hospitalization and death, the researchers wrote.
In fact, Centers for Disease Control and Prevention data released on Dec. 16 shows that people who have received four shots of the original COVID-19 vaccines as well as the bivalent booster were 57% less likely to visit an urgent care clinic or emergency room, regardless of age.
It comes at a time when BQ.1 and BQ.1.1 account for about 70% of the circulating variants, data show. In addition, hospitalizations are up 18% over the past 2 weeks and COVID-19 deaths are up 50% nationwide, The New York Times reported.
Globally, in many places, an “immunity wall” that has been built, Dr. Topol said. That may not be the case in the United States.
“The problem in the United States, making it harder to predict, is that we have a very low rate of recent boosters, in the past 6 months, especially in seniors,” he said. For example, only 36% of Americans aged 65 years and older, the group with highest risk, have received an updated bivalent booster.
An evolving virus
The subvariants are successfully replacing BA.5, which reigned as one of the most common Omicron variants over the past year. The latest CDC data show that BA.5 now accounts for only about 10% of the circulating virus. The researchers wrote: “This rapid replacement of virus strains is raising the specter of yet another wave of infections in the coming months.”
BQ.1 and BQ.1.1 evolved directly from BA.5 – adding more and some novel mutations to the SARS-CoV-2 virus. XBB and XBB.1 are the “offspring” of a combination of two other strains, known as BJ.1 and BA.2.75.
The story sounds familiar to the researchers. “The rapid rise of these subvariants and their extensive array of spike mutations are reminiscent of the appearance of the first Omicron variant last year, thus raising concerns that they may further compromise the efficacy of current COVID-19 vaccines and monoclonal antibody therapeutics,” they wrote. “We now report findings that indicate that such concerns are, sadly, justified, especially so for the XBB and XBB.1 subvariants.”
To figure out how effective existing antibodies could be against these newer subvariants, Dr. Wang and colleagues used blood samples from five groups of people. They tested serum from people who had three doses of the original COVID-19 vaccine, four doses of the original vaccine, those who received a bivalent booster, people who experienced a breakthrough infection with the BA.2 Omicron variant, and those who had a breakthrough with a BA.4 or BA.5 variant.
Adding the new subvariants to these serum samples revealed that the existing antibodies in the blood were ineffective at wiping out or neutralizing BQ.1, BQ.1.1, XBB, and XBB.1.
The BQ.1 subvariant was six times more resistant to antibodies than BA.5, its parent strain, and XBB.1 was 63 times more resistant compared with its predecessor, BA.2.
This shift in the ability of vaccines to stop the subvariants “is particularly concerning,” the researchers wrote.
Wiping out treatments too
Dr. Wang and colleagues also tested how well a panel of 23 different monoclonal antibody drugs might work against the four subvariants. The therapies all worked well against the original Omicron variant and included some approved for use through the Food and Drug Administration emergency use authorization (EUA) program at the time of the study.
They found that 19 of these 23 monoclonal antibodies lost effectiveness “greatly or completely” against XBB and XBB.1, for example.
This is not the first time that monoclonal antibody therapies have gone from effective to ineffective. Previous variants have come out that no longer responded to treatment with bamlanivimab, etesevimab, imdevimab, casirivimab, tixagevimab, cilgavimab, and sotrovimab. Bebtelovimab now joins this list and is no longer available from Eli Lilly under EUA because of this lack of effectiveness.
The lack of an effective monoclonal antibody treatment “poses a serious problem for millions of immunocompromised individuals who do not respond robustly to COVID-19 vaccines,” the researchers wrote, adding that “the urgent need to develop active monoclonal antibodies for clinical use is obvious.”
A limitation of the study is that the work is done in blood samples. The effectiveness of COVID-19 vaccination against the BQ and XBB subvariants should be evaluated in people in clinical studies, the authors noted.
Also, the current study looked at how well antibodies could neutralize the viral strains, but future research, they added, should look at how well “cellular immunity” or other aspects of the immune system might protect people.
Going forward, the challenge remains to develop vaccines and treatments that offer broad protection as the coronavirus continues to evolve.
In an alarming ending, the researchers wrote: “We have collectively chased after SARS-CoV-2 variants for over 2 years, and yet, the virus continues to evolve and evade.”
A version of this article first appeared on Medscape.com.
It’s a story perhaps more appropriate for Halloween than for the festive holiday season, given its scary implications.
Not too dire so far, until the researchers’ other findings are considered.
The BQ.1, BQ1.1, XBB, and XBB.1 subvariants are the most resistant to neutralizing antibodies, researcher Qian Wang, PhD, and colleagues wrote in a study published online in the journal Cell. This means people have no or “markedly reduced” protection against infection from these four strains, even if they’ve already had COVID-19 or are vaccinated and boosted multiple times, including with a bivalent vaccine.
On top of that, all available monoclonal antibody treatments are mostly or completely ineffective against these subvariants.
What does that mean for the immediate future? The findings are definitely “worrisome,” said Eric Topol, MD, founder and director of the Scripps Translational Research Institute in La Jolla, Calif.
But evidence from other countries, specifically Singapore and France, show that at least two of these variants turned out not to be as damaging as expected, likely because of high numbers of people vaccinated or who survived previous infections, he said.
Still, there is little to celebrate in the new findings, except that COVID-19 vaccinations and prior infections can still reduce the risk for serious outcomes such as hospitalization and death, the researchers wrote.
In fact, Centers for Disease Control and Prevention data released on Dec. 16 shows that people who have received four shots of the original COVID-19 vaccines as well as the bivalent booster were 57% less likely to visit an urgent care clinic or emergency room, regardless of age.
It comes at a time when BQ.1 and BQ.1.1 account for about 70% of the circulating variants, data show. In addition, hospitalizations are up 18% over the past 2 weeks and COVID-19 deaths are up 50% nationwide, The New York Times reported.
Globally, in many places, an “immunity wall” that has been built, Dr. Topol said. That may not be the case in the United States.
“The problem in the United States, making it harder to predict, is that we have a very low rate of recent boosters, in the past 6 months, especially in seniors,” he said. For example, only 36% of Americans aged 65 years and older, the group with highest risk, have received an updated bivalent booster.
An evolving virus
The subvariants are successfully replacing BA.5, which reigned as one of the most common Omicron variants over the past year. The latest CDC data show that BA.5 now accounts for only about 10% of the circulating virus. The researchers wrote: “This rapid replacement of virus strains is raising the specter of yet another wave of infections in the coming months.”
BQ.1 and BQ.1.1 evolved directly from BA.5 – adding more and some novel mutations to the SARS-CoV-2 virus. XBB and XBB.1 are the “offspring” of a combination of two other strains, known as BJ.1 and BA.2.75.
The story sounds familiar to the researchers. “The rapid rise of these subvariants and their extensive array of spike mutations are reminiscent of the appearance of the first Omicron variant last year, thus raising concerns that they may further compromise the efficacy of current COVID-19 vaccines and monoclonal antibody therapeutics,” they wrote. “We now report findings that indicate that such concerns are, sadly, justified, especially so for the XBB and XBB.1 subvariants.”
To figure out how effective existing antibodies could be against these newer subvariants, Dr. Wang and colleagues used blood samples from five groups of people. They tested serum from people who had three doses of the original COVID-19 vaccine, four doses of the original vaccine, those who received a bivalent booster, people who experienced a breakthrough infection with the BA.2 Omicron variant, and those who had a breakthrough with a BA.4 or BA.5 variant.
Adding the new subvariants to these serum samples revealed that the existing antibodies in the blood were ineffective at wiping out or neutralizing BQ.1, BQ.1.1, XBB, and XBB.1.
The BQ.1 subvariant was six times more resistant to antibodies than BA.5, its parent strain, and XBB.1 was 63 times more resistant compared with its predecessor, BA.2.
This shift in the ability of vaccines to stop the subvariants “is particularly concerning,” the researchers wrote.
Wiping out treatments too
Dr. Wang and colleagues also tested how well a panel of 23 different monoclonal antibody drugs might work against the four subvariants. The therapies all worked well against the original Omicron variant and included some approved for use through the Food and Drug Administration emergency use authorization (EUA) program at the time of the study.
They found that 19 of these 23 monoclonal antibodies lost effectiveness “greatly or completely” against XBB and XBB.1, for example.
This is not the first time that monoclonal antibody therapies have gone from effective to ineffective. Previous variants have come out that no longer responded to treatment with bamlanivimab, etesevimab, imdevimab, casirivimab, tixagevimab, cilgavimab, and sotrovimab. Bebtelovimab now joins this list and is no longer available from Eli Lilly under EUA because of this lack of effectiveness.
The lack of an effective monoclonal antibody treatment “poses a serious problem for millions of immunocompromised individuals who do not respond robustly to COVID-19 vaccines,” the researchers wrote, adding that “the urgent need to develop active monoclonal antibodies for clinical use is obvious.”
A limitation of the study is that the work is done in blood samples. The effectiveness of COVID-19 vaccination against the BQ and XBB subvariants should be evaluated in people in clinical studies, the authors noted.
Also, the current study looked at how well antibodies could neutralize the viral strains, but future research, they added, should look at how well “cellular immunity” or other aspects of the immune system might protect people.
Going forward, the challenge remains to develop vaccines and treatments that offer broad protection as the coronavirus continues to evolve.
In an alarming ending, the researchers wrote: “We have collectively chased after SARS-CoV-2 variants for over 2 years, and yet, the virus continues to evolve and evade.”
A version of this article first appeared on Medscape.com.
FROM CELL

