User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Coffee’s ‘Sweet Spot’: Daily Consumption and Cardiometabolic Risk
Each and every day, 1 billion people on this planet ingest a particular psychoactive substance. This chemical has fairly profound physiologic effects. It increases levels of nitric oxide in the blood, leads to vasodilation, and, of course, makes you feel more awake. The substance comes in many forms but almost always in a liquid medium. Do you have it yet? That’s right. The substance is caffeine, quite possibly the healthiest recreational drug that has ever been discovered.
This might be my New England upbringing speaking, but when it comes to lifestyle and health, one of the rules I’ve internalized is that things that are pleasurable are generally bad for you. I know, I know — some of you love to exercise. Some of you love doing crosswords. But you know what I mean. I’m talking French fries, smoked meats, drugs, smoking, alcohol, binge-watching Firefly. You’d be suspicious if a study came out suggesting that eating ice cream in bed reduces your risk for heart attack, and so would I. So I’m always on the lookout for those unicorns of lifestyle factors, those rare things that you want to do and are also good for you.
So far, the data are strong for three things: sleeping, (safe) sexual activity, and coffee. You’ll have to stay tuned for articles about the first two. Today, we’re brewing up some deeper insights about the power of java.
I was inspired to write this article because of a paper, “Habitual Coffee, Tea, and Caffeine Consumption, Circulating Metabolites, and the Risk of Cardiometabolic Multimorbidity,” appearing September 17 in The Journal of Clinical Endocrinology and Metabolism (JCEM).
This is not the first study to suggest that coffee intake may be beneficial. A 2013 meta-analysis summarized the results of 36 studies with more than a million participants and found a U-shaped relationship between coffee intake and cardiovascular risk. The sweet spot was at three to five cups a day; people drinking that much coffee had about a 15% reduced risk for cardiovascular disease compared with nondrinkers.
But here’s the thing. Coffee contains caffeine, but it is much more than that. It is a heady brew of various chemicals and compounds, phenols, and chlorogenic acids. And, of course, you can get caffeine from stuff that isn’t coffee — natural things like tea — and decidedly unnatural things like energy drinks. How do you figure out where the benefit really lies?
The JCEM study leveraged the impressive UK Biobank dataset to figure this out. The Biobank recruited more than half a million people from the UK between 2006 and 2010 and collected a wealth of data from each of them: surveys, blood samples, biometrics, medical imaging — the works. And then they followed what would happen to those people medically over time. It’s a pretty amazing resource.
But for the purposes of this study, what you need to know is that just under 200,000 of those participants met the key criteria for this study: being free from cardiovascular disease at baseline; having completed a detailed survey about their coffee, tea, and other caffeinated beverage intake; and having adequate follow-up. A subset of that number, just under 100,000, had metabolomic data — which is where this study really gets interesting.
We’ll dive into the metabolome in a moment, but first let’s just talk about the main finding, the relationship between coffee, tea, or caffeine and cardiovascular disease. But to do that, we need to acknowledge that people who drink a lot of coffee are different from people who don’t, and it might be those differences, not the coffee itself, that are beneficial.
What were those differences? People who drank more coffee tended to be a bit older, were less likely to be female, and were slightly more likely to engage in physical activity. They ate less processed meat but also fewer vegetables. Some of those factors, like being female, are generally protective against cardiovascular disease; but some, like age, are definitely not. The authors adjusted for these and multiple other factors, including alcohol intake, BMI, kidney function, and many others to try to disentangle the effect of being the type of person who drinks a lot of coffee from the drinking a lot of coffee itself.
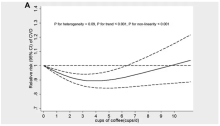
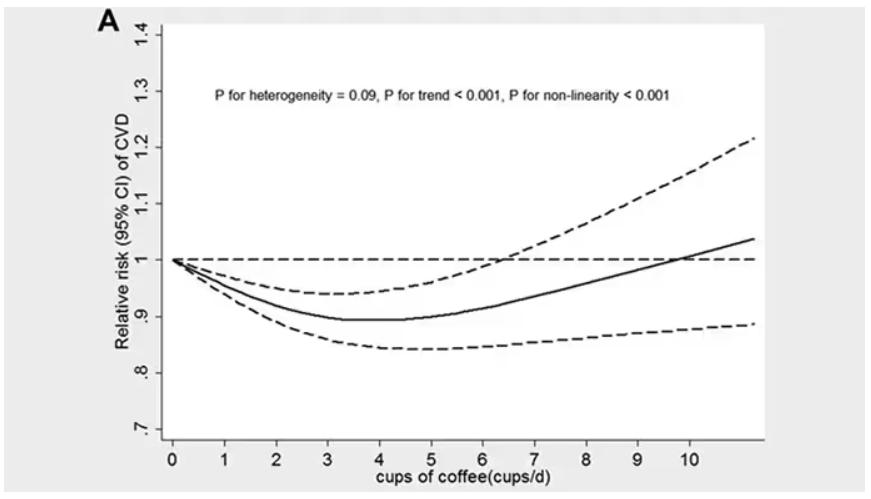
These are the results of the fully adjusted model. Compared with nonconsumers, you can see that people in the higher range of coffee, tea, or just caffeine intake have almost a 40% reduction in cardiovascular disease in follow-up.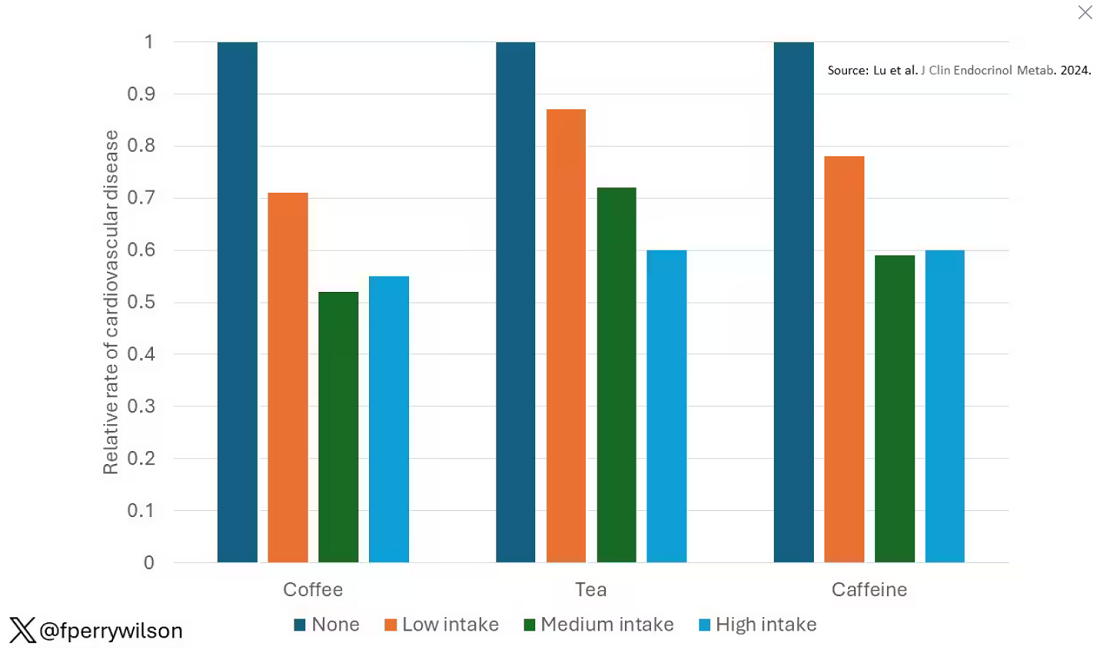
Looking at the benefit across the spectrum of intake, you again see that U-shaped curve, suggesting that a sweet spot for daily consumption can be found around 3 cups of coffee or tea (or 250 mg of caffeine). A standard energy drink contains about 120 mg of caffeine. 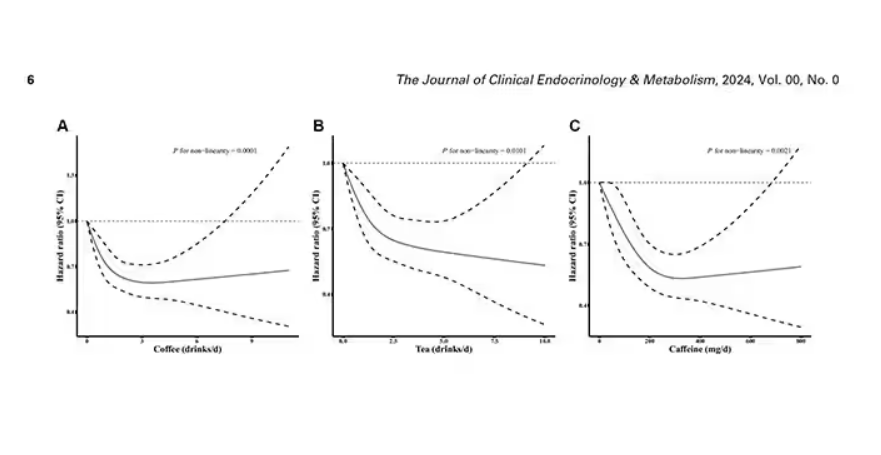
But if this is true, it would be good to know why. To figure that out, the authors turned to the metabolome. The idea here is that your body is constantly breaking stuff down, taking all these proteins and chemicals and compounds that we ingest and turning them into metabolites. Using advanced measurement techniques, researchers can measure hundreds or even thousands of metabolites from a single blood sample. They provide information, obviously, about the food you eat and the drinks you drink, but what is really intriguing is that some metabolites are associated with better health and some with worse
In this study, researchers measured 168 individual metabolites. Eighty of them, nearly half, were significantly altered in people who drank more coffee.
This figure summarizes the findings, and yes, this is way too complicated. 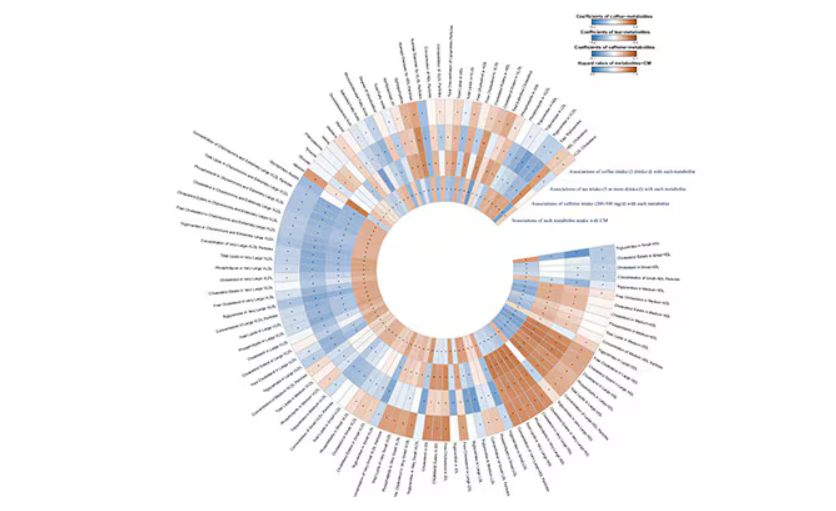
But here’s how to interpret it. The inner ring shows you how certain metabolites are associated with cardiovascular disease. The outer rings show you how those metabolites are associated with coffee, tea, or caffeine. The interesting part is that the sections of the ring (outer rings and inner rings) are very different colors.
Like here.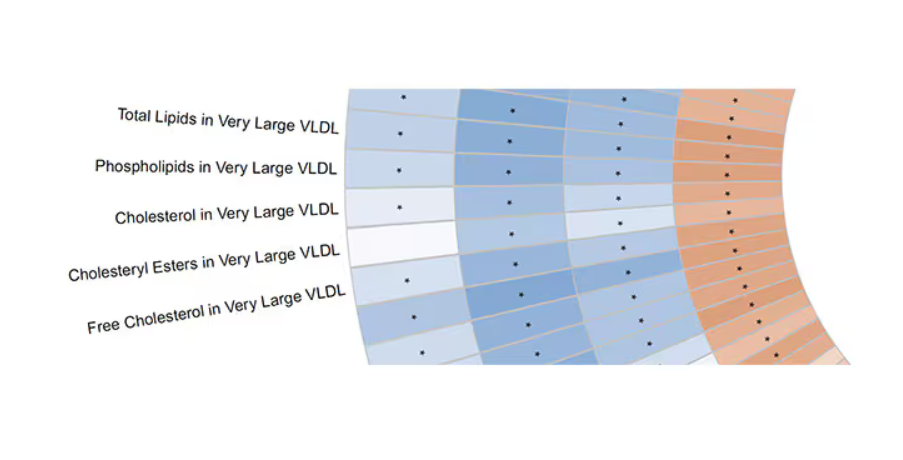
What you see here is a fairly profound effect that coffee, tea, or caffeine intake has on metabolites of VLDL — bad cholesterol. The beverages lower it, and, of course, higher levels lead to cardiovascular disease. This means that this is a potential causal pathway from coffee intake to heart protection.
And that’s not the only one.
You see a similar relationship for saturated fatty acids. Higher levels lead to cardiovascular disease, and coffee intake lowers levels. The reverse works too: Lower levels of histidine (an amino acid) increase cardiovascular risk, and coffee seems to raise those levels.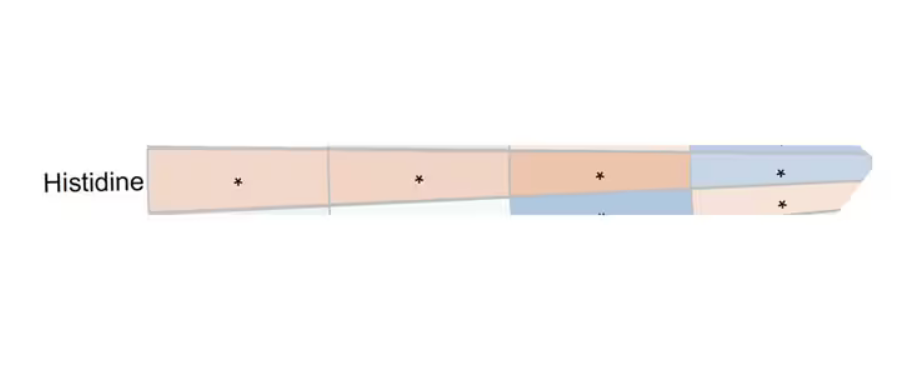
Is this all too good to be true? It’s hard to say. The data on coffee’s benefits have been remarkably consistent. Still, I wouldn’t be a good doctor if I didn’t mention that clearly there is a difference between a cup of black coffee and a venti caramel Frappuccino.
Nevertheless, coffee remains firmly in my holy trinity of enjoyable things that are, for whatever reason, still good for you. So, when you’re having that second, or third, or maybe fourth cup of the day, you can take that to heart.
Dr. Wilson, associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator, reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Each and every day, 1 billion people on this planet ingest a particular psychoactive substance. This chemical has fairly profound physiologic effects. It increases levels of nitric oxide in the blood, leads to vasodilation, and, of course, makes you feel more awake. The substance comes in many forms but almost always in a liquid medium. Do you have it yet? That’s right. The substance is caffeine, quite possibly the healthiest recreational drug that has ever been discovered.
This might be my New England upbringing speaking, but when it comes to lifestyle and health, one of the rules I’ve internalized is that things that are pleasurable are generally bad for you. I know, I know — some of you love to exercise. Some of you love doing crosswords. But you know what I mean. I’m talking French fries, smoked meats, drugs, smoking, alcohol, binge-watching Firefly. You’d be suspicious if a study came out suggesting that eating ice cream in bed reduces your risk for heart attack, and so would I. So I’m always on the lookout for those unicorns of lifestyle factors, those rare things that you want to do and are also good for you.
So far, the data are strong for three things: sleeping, (safe) sexual activity, and coffee. You’ll have to stay tuned for articles about the first two. Today, we’re brewing up some deeper insights about the power of java.
I was inspired to write this article because of a paper, “Habitual Coffee, Tea, and Caffeine Consumption, Circulating Metabolites, and the Risk of Cardiometabolic Multimorbidity,” appearing September 17 in The Journal of Clinical Endocrinology and Metabolism (JCEM).
This is not the first study to suggest that coffee intake may be beneficial. A 2013 meta-analysis summarized the results of 36 studies with more than a million participants and found a U-shaped relationship between coffee intake and cardiovascular risk. The sweet spot was at three to five cups a day; people drinking that much coffee had about a 15% reduced risk for cardiovascular disease compared with nondrinkers.
But here’s the thing. Coffee contains caffeine, but it is much more than that. It is a heady brew of various chemicals and compounds, phenols, and chlorogenic acids. And, of course, you can get caffeine from stuff that isn’t coffee — natural things like tea — and decidedly unnatural things like energy drinks. How do you figure out where the benefit really lies?
The JCEM study leveraged the impressive UK Biobank dataset to figure this out. The Biobank recruited more than half a million people from the UK between 2006 and 2010 and collected a wealth of data from each of them: surveys, blood samples, biometrics, medical imaging — the works. And then they followed what would happen to those people medically over time. It’s a pretty amazing resource.
But for the purposes of this study, what you need to know is that just under 200,000 of those participants met the key criteria for this study: being free from cardiovascular disease at baseline; having completed a detailed survey about their coffee, tea, and other caffeinated beverage intake; and having adequate follow-up. A subset of that number, just under 100,000, had metabolomic data — which is where this study really gets interesting.
We’ll dive into the metabolome in a moment, but first let’s just talk about the main finding, the relationship between coffee, tea, or caffeine and cardiovascular disease. But to do that, we need to acknowledge that people who drink a lot of coffee are different from people who don’t, and it might be those differences, not the coffee itself, that are beneficial.
What were those differences? People who drank more coffee tended to be a bit older, were less likely to be female, and were slightly more likely to engage in physical activity. They ate less processed meat but also fewer vegetables. Some of those factors, like being female, are generally protective against cardiovascular disease; but some, like age, are definitely not. The authors adjusted for these and multiple other factors, including alcohol intake, BMI, kidney function, and many others to try to disentangle the effect of being the type of person who drinks a lot of coffee from the drinking a lot of coffee itself.
These are the results of the fully adjusted model. Compared with nonconsumers, you can see that people in the higher range of coffee, tea, or just caffeine intake have almost a 40% reduction in cardiovascular disease in follow-up.
Looking at the benefit across the spectrum of intake, you again see that U-shaped curve, suggesting that a sweet spot for daily consumption can be found around 3 cups of coffee or tea (or 250 mg of caffeine). A standard energy drink contains about 120 mg of caffeine. 
But if this is true, it would be good to know why. To figure that out, the authors turned to the metabolome. The idea here is that your body is constantly breaking stuff down, taking all these proteins and chemicals and compounds that we ingest and turning them into metabolites. Using advanced measurement techniques, researchers can measure hundreds or even thousands of metabolites from a single blood sample. They provide information, obviously, about the food you eat and the drinks you drink, but what is really intriguing is that some metabolites are associated with better health and some with worse
In this study, researchers measured 168 individual metabolites. Eighty of them, nearly half, were significantly altered in people who drank more coffee.
This figure summarizes the findings, and yes, this is way too complicated. 
But here’s how to interpret it. The inner ring shows you how certain metabolites are associated with cardiovascular disease. The outer rings show you how those metabolites are associated with coffee, tea, or caffeine. The interesting part is that the sections of the ring (outer rings and inner rings) are very different colors.
Like here.
What you see here is a fairly profound effect that coffee, tea, or caffeine intake has on metabolites of VLDL — bad cholesterol. The beverages lower it, and, of course, higher levels lead to cardiovascular disease. This means that this is a potential causal pathway from coffee intake to heart protection.
And that’s not the only one.
You see a similar relationship for saturated fatty acids. Higher levels lead to cardiovascular disease, and coffee intake lowers levels. The reverse works too: Lower levels of histidine (an amino acid) increase cardiovascular risk, and coffee seems to raise those levels.
Is this all too good to be true? It’s hard to say. The data on coffee’s benefits have been remarkably consistent. Still, I wouldn’t be a good doctor if I didn’t mention that clearly there is a difference between a cup of black coffee and a venti caramel Frappuccino.
Nevertheless, coffee remains firmly in my holy trinity of enjoyable things that are, for whatever reason, still good for you. So, when you’re having that second, or third, or maybe fourth cup of the day, you can take that to heart.
Dr. Wilson, associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator, reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Each and every day, 1 billion people on this planet ingest a particular psychoactive substance. This chemical has fairly profound physiologic effects. It increases levels of nitric oxide in the blood, leads to vasodilation, and, of course, makes you feel more awake. The substance comes in many forms but almost always in a liquid medium. Do you have it yet? That’s right. The substance is caffeine, quite possibly the healthiest recreational drug that has ever been discovered.
This might be my New England upbringing speaking, but when it comes to lifestyle and health, one of the rules I’ve internalized is that things that are pleasurable are generally bad for you. I know, I know — some of you love to exercise. Some of you love doing crosswords. But you know what I mean. I’m talking French fries, smoked meats, drugs, smoking, alcohol, binge-watching Firefly. You’d be suspicious if a study came out suggesting that eating ice cream in bed reduces your risk for heart attack, and so would I. So I’m always on the lookout for those unicorns of lifestyle factors, those rare things that you want to do and are also good for you.
So far, the data are strong for three things: sleeping, (safe) sexual activity, and coffee. You’ll have to stay tuned for articles about the first two. Today, we’re brewing up some deeper insights about the power of java.
I was inspired to write this article because of a paper, “Habitual Coffee, Tea, and Caffeine Consumption, Circulating Metabolites, and the Risk of Cardiometabolic Multimorbidity,” appearing September 17 in The Journal of Clinical Endocrinology and Metabolism (JCEM).
This is not the first study to suggest that coffee intake may be beneficial. A 2013 meta-analysis summarized the results of 36 studies with more than a million participants and found a U-shaped relationship between coffee intake and cardiovascular risk. The sweet spot was at three to five cups a day; people drinking that much coffee had about a 15% reduced risk for cardiovascular disease compared with nondrinkers.
But here’s the thing. Coffee contains caffeine, but it is much more than that. It is a heady brew of various chemicals and compounds, phenols, and chlorogenic acids. And, of course, you can get caffeine from stuff that isn’t coffee — natural things like tea — and decidedly unnatural things like energy drinks. How do you figure out where the benefit really lies?
The JCEM study leveraged the impressive UK Biobank dataset to figure this out. The Biobank recruited more than half a million people from the UK between 2006 and 2010 and collected a wealth of data from each of them: surveys, blood samples, biometrics, medical imaging — the works. And then they followed what would happen to those people medically over time. It’s a pretty amazing resource.
But for the purposes of this study, what you need to know is that just under 200,000 of those participants met the key criteria for this study: being free from cardiovascular disease at baseline; having completed a detailed survey about their coffee, tea, and other caffeinated beverage intake; and having adequate follow-up. A subset of that number, just under 100,000, had metabolomic data — which is where this study really gets interesting.
We’ll dive into the metabolome in a moment, but first let’s just talk about the main finding, the relationship between coffee, tea, or caffeine and cardiovascular disease. But to do that, we need to acknowledge that people who drink a lot of coffee are different from people who don’t, and it might be those differences, not the coffee itself, that are beneficial.
What were those differences? People who drank more coffee tended to be a bit older, were less likely to be female, and were slightly more likely to engage in physical activity. They ate less processed meat but also fewer vegetables. Some of those factors, like being female, are generally protective against cardiovascular disease; but some, like age, are definitely not. The authors adjusted for these and multiple other factors, including alcohol intake, BMI, kidney function, and many others to try to disentangle the effect of being the type of person who drinks a lot of coffee from the drinking a lot of coffee itself.
These are the results of the fully adjusted model. Compared with nonconsumers, you can see that people in the higher range of coffee, tea, or just caffeine intake have almost a 40% reduction in cardiovascular disease in follow-up.
Looking at the benefit across the spectrum of intake, you again see that U-shaped curve, suggesting that a sweet spot for daily consumption can be found around 3 cups of coffee or tea (or 250 mg of caffeine). A standard energy drink contains about 120 mg of caffeine. 
But if this is true, it would be good to know why. To figure that out, the authors turned to the metabolome. The idea here is that your body is constantly breaking stuff down, taking all these proteins and chemicals and compounds that we ingest and turning them into metabolites. Using advanced measurement techniques, researchers can measure hundreds or even thousands of metabolites from a single blood sample. They provide information, obviously, about the food you eat and the drinks you drink, but what is really intriguing is that some metabolites are associated with better health and some with worse
In this study, researchers measured 168 individual metabolites. Eighty of them, nearly half, were significantly altered in people who drank more coffee.
This figure summarizes the findings, and yes, this is way too complicated. 
But here’s how to interpret it. The inner ring shows you how certain metabolites are associated with cardiovascular disease. The outer rings show you how those metabolites are associated with coffee, tea, or caffeine. The interesting part is that the sections of the ring (outer rings and inner rings) are very different colors.
Like here.
What you see here is a fairly profound effect that coffee, tea, or caffeine intake has on metabolites of VLDL — bad cholesterol. The beverages lower it, and, of course, higher levels lead to cardiovascular disease. This means that this is a potential causal pathway from coffee intake to heart protection.
And that’s not the only one.
You see a similar relationship for saturated fatty acids. Higher levels lead to cardiovascular disease, and coffee intake lowers levels. The reverse works too: Lower levels of histidine (an amino acid) increase cardiovascular risk, and coffee seems to raise those levels.
Is this all too good to be true? It’s hard to say. The data on coffee’s benefits have been remarkably consistent. Still, I wouldn’t be a good doctor if I didn’t mention that clearly there is a difference between a cup of black coffee and a venti caramel Frappuccino.
Nevertheless, coffee remains firmly in my holy trinity of enjoyable things that are, for whatever reason, still good for you. So, when you’re having that second, or third, or maybe fourth cup of the day, you can take that to heart.
Dr. Wilson, associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator, reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Will New Obesity Drugs Make Bariatric Surgery Obsolete?
MADRID — In spirited presentations at the annual meeting of the European Association for the Study of Diabetes, Louis J. Aronne, MD, of Weill Cornell Medicine in New York City, made a compelling case that the next generation of obesity medications will make bariatric surgery obsolete, and Francesco Rubino, MD, of King’s College London in England, made an equally compelling case that they will not.
In fact, Dr. Rubino predicted that “metabolic” surgery — new nomenclature reflecting the power of surgery to reduce not only obesity, but also other metabolic conditions, over the long term — will continue and could even increase in years to come.
‘Medical Treatment Will Dominate’
“Obesity treatment is the superhero of treating metabolic disease because it can defeat all of the bad guys at once, not just one, like the other treatments,” Dr. Aronne told meeting attendees. “If you treat somebody’s cholesterol, you’re just treating their cholesterol, and you may actually increase their risk of developing type 2 diabetes (T2D). You treat their blood pressure, you don’t treat their glucose and you don’t treat their lipids — the list goes on and on and on. But by treating obesity, if you can get enough weight loss, you can get all those things at once.”
He pointed to the SELECT trial, which showed that treating obesity with a glucagon-like peptide 1 receptor agonist reduced major adverse cardiovascular events as well as death from any cause, results in line with those from other modes of treatment for cardiovascular disease (CVD) or lipid lowering, he said. “But we get much more with these drugs, including positive effects on heart failure, chronic kidney disease, and a 73% reduction in T2D. So, we’re now on the verge of a major change in the way we manage metabolic disease.”
Dr. Aronne drew a parallel between treating obesity and the historic way of treating hypertension. Years ago, he said, “we waited too long to treat people. We waited until they had severe hypertension that in many cases was irreversible. What would you prefer to do now for obesity — have the patient lose weight with a medicine that is proven to reduce complications or wait until they develop diabetes, high blood pressure, heart disease and then have them undergo surgery to treat that?”
Looking ahead, “the trend could be to treat obesity before it gets out of hand,” he suggested. Treatment might start in people with a body mass index (BMI) of 27 kg/m2, who would be treated to a target BMI of 25. “That’s only a 10% or so change, but our goal would be to keep them in the normal range so they never go above that target. In fact, I think we’re going to be looking at people with severe obesity in a few years and saying, ‘I can’t believe someone didn’t treat that guy earlier.’ What’s going to happen to bariatric surgery if no one gets to a higher weight?”
The plethora of current weight-loss drugs and the large group on the horizon mean that if someone doesn’t respond to one drug, there will be plenty of other choices, Dr. Aronne continued. People will be referred for surgery, but possibly only after they’ve not responded to medical treatment — or just the opposite. “In the United States, it’s much cheaper to have surgery, and I bet the insurance companies are going to make people have surgery before they can get the medicines,” he acknowledged.
A recent report from Morgan Stanley suggests that the global market for the newer weight-loss drugs could increase by 15-fold over the next 5 years as their benefits expand beyond weight loss and that as much as 9% of the US population will be taking the drugs by 2035, Dr. Aronne said, adding that he thinks 9% is an underestimate. By contrast, the number of patients treated by his team’s surgical program is down about 20%.
“I think it’s very clear that medical treatment is going to dominate,” he concluded. “But, it’s also possible that surgery could go up because so many people are going to be coming for medical therapy that we may wind up referring more for surgical therapy.”
‘Surgery Is Saving Lives’
Dr. Rubino is convinced that anti-obesity drugs will not make surgery obsolete, “but it will not be business as usual,” he told meeting attendees. “In fact, I think these drugs will expedite a process that is already ongoing — a transformation of bariatric into metabolic surgery.”
“Bariatric surgery will go down in history as one of the biggest missed opportunities that we, as medical professionals, have seen over the past many years,” he said. “It has been shown beyond any doubt to reduce all-cause mortality — in other words, it saves lives,” and it’s also cost effective and improves quality of life. Yet, fewer than 1% of people globally who meet the criteria actually get the surgery.
Many clinicians don’t inform patients about the treatment and don’t refer them for it, he said. “That would be equivalent to having surgery for CVD [cardiovascular disease], cancer, or other important diseases available but not being accessed as they should be.”
A big reason for the dearth of procedures is that people have unrealistic expectations about diet and exercise interventions for weight loss, he said. His team’s survey, presented at the 26th World Congress of the International Federation for the Surgery of Obesity and Metabolic Disorders, showed that 43% of respondents believed diet and exercise was the best treatment for severe obesity (BMI > 35). A more recent survey asked which among several choices was the most effective weight-loss intervention, and again a large proportion “believed wrongly that diet and exercise is most effective — more so than drugs or surgery — despite plenty of evidence that this is not the case.”
In this context, he said, “any surgery, no matter how safe or effective, would never be very popular.” If obesity is viewed as a modifiable risk factor, patients may say they’ll think about it for 6 months. In contrast, “nobody will tell you ‘I will think about it’ if you tell them they need gallbladder surgery to get rid of gallstone pain.”
Although drugs are available to treat obesity, none of them are curative, and if they’re stopped, the weight comes back, Dr. Rubino pointed out. “Efficacy of drugs is measured in weeks or months, whereas efficacy of surgery is measured in decades of durability — in the case of bariatric surgery, 10-20 years. That’s why bariatric surgery will remain an option,” he said. “It’s not just preventing disease, it’s resolving ongoing disease.”
Furthermore, bariatric surgery is showing value for people with established T2D, whereas in the past, it was mainly considered to be a weight-loss intervention for younger, healthier patients, he said. “In my practice, we’re operating more often in people with T2D, even those at higher risk for anesthesia and surgery — eg, patients with heart failure, chronic kidney disease, on dialysis — and we’re still able to maintain the same safety with minimally invasive laparoscopic surgery that we had with healthier patients.”
A vote held at the end of the session revealed that the audience was split about half and half in favor of drugs making bariatric surgery obsolete or not.
“I think we may have to duke it out now,” Dr. Aronne quipped.
Dr. Aronne disclosed being a consultant, speaker, and adviser for and receiving research support from Altimmune, Amgen, AstraZeneca, Eli Lilly, Intellihealth, Janssen, Novo Nordisk, Pfizer, Senda, UnitedHealth Group, Versanis, and others; he has ownership interests in ERX, Intellihealth, Jamieson, Kallyope, Skye Bioscience, Veru, and others; and he is on the board of directors of ERX, Jamieson Wellness, and Intellihealth/FlyteHealth. Dr. Rubino disclosed receiving research and educational grants from Novo Nordisk, Ethicon, and Medtronic; he is on the scientific advisory board/data safety advisory board for Keyron, Morphic Medical, and GT Metabolic Solutions; he receives speaking honoraria from Medtronic, Ethicon, Novo Nordisk, and Eli Lilly; and he is president of the nonprofit Metabolic Health Institute.
A version of this article first appeared on Medscape.com.
MADRID — In spirited presentations at the annual meeting of the European Association for the Study of Diabetes, Louis J. Aronne, MD, of Weill Cornell Medicine in New York City, made a compelling case that the next generation of obesity medications will make bariatric surgery obsolete, and Francesco Rubino, MD, of King’s College London in England, made an equally compelling case that they will not.
In fact, Dr. Rubino predicted that “metabolic” surgery — new nomenclature reflecting the power of surgery to reduce not only obesity, but also other metabolic conditions, over the long term — will continue and could even increase in years to come.
‘Medical Treatment Will Dominate’
“Obesity treatment is the superhero of treating metabolic disease because it can defeat all of the bad guys at once, not just one, like the other treatments,” Dr. Aronne told meeting attendees. “If you treat somebody’s cholesterol, you’re just treating their cholesterol, and you may actually increase their risk of developing type 2 diabetes (T2D). You treat their blood pressure, you don’t treat their glucose and you don’t treat their lipids — the list goes on and on and on. But by treating obesity, if you can get enough weight loss, you can get all those things at once.”
He pointed to the SELECT trial, which showed that treating obesity with a glucagon-like peptide 1 receptor agonist reduced major adverse cardiovascular events as well as death from any cause, results in line with those from other modes of treatment for cardiovascular disease (CVD) or lipid lowering, he said. “But we get much more with these drugs, including positive effects on heart failure, chronic kidney disease, and a 73% reduction in T2D. So, we’re now on the verge of a major change in the way we manage metabolic disease.”
Dr. Aronne drew a parallel between treating obesity and the historic way of treating hypertension. Years ago, he said, “we waited too long to treat people. We waited until they had severe hypertension that in many cases was irreversible. What would you prefer to do now for obesity — have the patient lose weight with a medicine that is proven to reduce complications or wait until they develop diabetes, high blood pressure, heart disease and then have them undergo surgery to treat that?”
Looking ahead, “the trend could be to treat obesity before it gets out of hand,” he suggested. Treatment might start in people with a body mass index (BMI) of 27 kg/m2, who would be treated to a target BMI of 25. “That’s only a 10% or so change, but our goal would be to keep them in the normal range so they never go above that target. In fact, I think we’re going to be looking at people with severe obesity in a few years and saying, ‘I can’t believe someone didn’t treat that guy earlier.’ What’s going to happen to bariatric surgery if no one gets to a higher weight?”
The plethora of current weight-loss drugs and the large group on the horizon mean that if someone doesn’t respond to one drug, there will be plenty of other choices, Dr. Aronne continued. People will be referred for surgery, but possibly only after they’ve not responded to medical treatment — or just the opposite. “In the United States, it’s much cheaper to have surgery, and I bet the insurance companies are going to make people have surgery before they can get the medicines,” he acknowledged.
A recent report from Morgan Stanley suggests that the global market for the newer weight-loss drugs could increase by 15-fold over the next 5 years as their benefits expand beyond weight loss and that as much as 9% of the US population will be taking the drugs by 2035, Dr. Aronne said, adding that he thinks 9% is an underestimate. By contrast, the number of patients treated by his team’s surgical program is down about 20%.
“I think it’s very clear that medical treatment is going to dominate,” he concluded. “But, it’s also possible that surgery could go up because so many people are going to be coming for medical therapy that we may wind up referring more for surgical therapy.”
‘Surgery Is Saving Lives’
Dr. Rubino is convinced that anti-obesity drugs will not make surgery obsolete, “but it will not be business as usual,” he told meeting attendees. “In fact, I think these drugs will expedite a process that is already ongoing — a transformation of bariatric into metabolic surgery.”
“Bariatric surgery will go down in history as one of the biggest missed opportunities that we, as medical professionals, have seen over the past many years,” he said. “It has been shown beyond any doubt to reduce all-cause mortality — in other words, it saves lives,” and it’s also cost effective and improves quality of life. Yet, fewer than 1% of people globally who meet the criteria actually get the surgery.
Many clinicians don’t inform patients about the treatment and don’t refer them for it, he said. “That would be equivalent to having surgery for CVD [cardiovascular disease], cancer, or other important diseases available but not being accessed as they should be.”
A big reason for the dearth of procedures is that people have unrealistic expectations about diet and exercise interventions for weight loss, he said. His team’s survey, presented at the 26th World Congress of the International Federation for the Surgery of Obesity and Metabolic Disorders, showed that 43% of respondents believed diet and exercise was the best treatment for severe obesity (BMI > 35). A more recent survey asked which among several choices was the most effective weight-loss intervention, and again a large proportion “believed wrongly that diet and exercise is most effective — more so than drugs or surgery — despite plenty of evidence that this is not the case.”
In this context, he said, “any surgery, no matter how safe or effective, would never be very popular.” If obesity is viewed as a modifiable risk factor, patients may say they’ll think about it for 6 months. In contrast, “nobody will tell you ‘I will think about it’ if you tell them they need gallbladder surgery to get rid of gallstone pain.”
Although drugs are available to treat obesity, none of them are curative, and if they’re stopped, the weight comes back, Dr. Rubino pointed out. “Efficacy of drugs is measured in weeks or months, whereas efficacy of surgery is measured in decades of durability — in the case of bariatric surgery, 10-20 years. That’s why bariatric surgery will remain an option,” he said. “It’s not just preventing disease, it’s resolving ongoing disease.”
Furthermore, bariatric surgery is showing value for people with established T2D, whereas in the past, it was mainly considered to be a weight-loss intervention for younger, healthier patients, he said. “In my practice, we’re operating more often in people with T2D, even those at higher risk for anesthesia and surgery — eg, patients with heart failure, chronic kidney disease, on dialysis — and we’re still able to maintain the same safety with minimally invasive laparoscopic surgery that we had with healthier patients.”
A vote held at the end of the session revealed that the audience was split about half and half in favor of drugs making bariatric surgery obsolete or not.
“I think we may have to duke it out now,” Dr. Aronne quipped.
Dr. Aronne disclosed being a consultant, speaker, and adviser for and receiving research support from Altimmune, Amgen, AstraZeneca, Eli Lilly, Intellihealth, Janssen, Novo Nordisk, Pfizer, Senda, UnitedHealth Group, Versanis, and others; he has ownership interests in ERX, Intellihealth, Jamieson, Kallyope, Skye Bioscience, Veru, and others; and he is on the board of directors of ERX, Jamieson Wellness, and Intellihealth/FlyteHealth. Dr. Rubino disclosed receiving research and educational grants from Novo Nordisk, Ethicon, and Medtronic; he is on the scientific advisory board/data safety advisory board for Keyron, Morphic Medical, and GT Metabolic Solutions; he receives speaking honoraria from Medtronic, Ethicon, Novo Nordisk, and Eli Lilly; and he is president of the nonprofit Metabolic Health Institute.
A version of this article first appeared on Medscape.com.
MADRID — In spirited presentations at the annual meeting of the European Association for the Study of Diabetes, Louis J. Aronne, MD, of Weill Cornell Medicine in New York City, made a compelling case that the next generation of obesity medications will make bariatric surgery obsolete, and Francesco Rubino, MD, of King’s College London in England, made an equally compelling case that they will not.
In fact, Dr. Rubino predicted that “metabolic” surgery — new nomenclature reflecting the power of surgery to reduce not only obesity, but also other metabolic conditions, over the long term — will continue and could even increase in years to come.
‘Medical Treatment Will Dominate’
“Obesity treatment is the superhero of treating metabolic disease because it can defeat all of the bad guys at once, not just one, like the other treatments,” Dr. Aronne told meeting attendees. “If you treat somebody’s cholesterol, you’re just treating their cholesterol, and you may actually increase their risk of developing type 2 diabetes (T2D). You treat their blood pressure, you don’t treat their glucose and you don’t treat their lipids — the list goes on and on and on. But by treating obesity, if you can get enough weight loss, you can get all those things at once.”
He pointed to the SELECT trial, which showed that treating obesity with a glucagon-like peptide 1 receptor agonist reduced major adverse cardiovascular events as well as death from any cause, results in line with those from other modes of treatment for cardiovascular disease (CVD) or lipid lowering, he said. “But we get much more with these drugs, including positive effects on heart failure, chronic kidney disease, and a 73% reduction in T2D. So, we’re now on the verge of a major change in the way we manage metabolic disease.”
Dr. Aronne drew a parallel between treating obesity and the historic way of treating hypertension. Years ago, he said, “we waited too long to treat people. We waited until they had severe hypertension that in many cases was irreversible. What would you prefer to do now for obesity — have the patient lose weight with a medicine that is proven to reduce complications or wait until they develop diabetes, high blood pressure, heart disease and then have them undergo surgery to treat that?”
Looking ahead, “the trend could be to treat obesity before it gets out of hand,” he suggested. Treatment might start in people with a body mass index (BMI) of 27 kg/m2, who would be treated to a target BMI of 25. “That’s only a 10% or so change, but our goal would be to keep them in the normal range so they never go above that target. In fact, I think we’re going to be looking at people with severe obesity in a few years and saying, ‘I can’t believe someone didn’t treat that guy earlier.’ What’s going to happen to bariatric surgery if no one gets to a higher weight?”
The plethora of current weight-loss drugs and the large group on the horizon mean that if someone doesn’t respond to one drug, there will be plenty of other choices, Dr. Aronne continued. People will be referred for surgery, but possibly only after they’ve not responded to medical treatment — or just the opposite. “In the United States, it’s much cheaper to have surgery, and I bet the insurance companies are going to make people have surgery before they can get the medicines,” he acknowledged.
A recent report from Morgan Stanley suggests that the global market for the newer weight-loss drugs could increase by 15-fold over the next 5 years as their benefits expand beyond weight loss and that as much as 9% of the US population will be taking the drugs by 2035, Dr. Aronne said, adding that he thinks 9% is an underestimate. By contrast, the number of patients treated by his team’s surgical program is down about 20%.
“I think it’s very clear that medical treatment is going to dominate,” he concluded. “But, it’s also possible that surgery could go up because so many people are going to be coming for medical therapy that we may wind up referring more for surgical therapy.”
‘Surgery Is Saving Lives’
Dr. Rubino is convinced that anti-obesity drugs will not make surgery obsolete, “but it will not be business as usual,” he told meeting attendees. “In fact, I think these drugs will expedite a process that is already ongoing — a transformation of bariatric into metabolic surgery.”
“Bariatric surgery will go down in history as one of the biggest missed opportunities that we, as medical professionals, have seen over the past many years,” he said. “It has been shown beyond any doubt to reduce all-cause mortality — in other words, it saves lives,” and it’s also cost effective and improves quality of life. Yet, fewer than 1% of people globally who meet the criteria actually get the surgery.
Many clinicians don’t inform patients about the treatment and don’t refer them for it, he said. “That would be equivalent to having surgery for CVD [cardiovascular disease], cancer, or other important diseases available but not being accessed as they should be.”
A big reason for the dearth of procedures is that people have unrealistic expectations about diet and exercise interventions for weight loss, he said. His team’s survey, presented at the 26th World Congress of the International Federation for the Surgery of Obesity and Metabolic Disorders, showed that 43% of respondents believed diet and exercise was the best treatment for severe obesity (BMI > 35). A more recent survey asked which among several choices was the most effective weight-loss intervention, and again a large proportion “believed wrongly that diet and exercise is most effective — more so than drugs or surgery — despite plenty of evidence that this is not the case.”
In this context, he said, “any surgery, no matter how safe or effective, would never be very popular.” If obesity is viewed as a modifiable risk factor, patients may say they’ll think about it for 6 months. In contrast, “nobody will tell you ‘I will think about it’ if you tell them they need gallbladder surgery to get rid of gallstone pain.”
Although drugs are available to treat obesity, none of them are curative, and if they’re stopped, the weight comes back, Dr. Rubino pointed out. “Efficacy of drugs is measured in weeks or months, whereas efficacy of surgery is measured in decades of durability — in the case of bariatric surgery, 10-20 years. That’s why bariatric surgery will remain an option,” he said. “It’s not just preventing disease, it’s resolving ongoing disease.”
Furthermore, bariatric surgery is showing value for people with established T2D, whereas in the past, it was mainly considered to be a weight-loss intervention for younger, healthier patients, he said. “In my practice, we’re operating more often in people with T2D, even those at higher risk for anesthesia and surgery — eg, patients with heart failure, chronic kidney disease, on dialysis — and we’re still able to maintain the same safety with minimally invasive laparoscopic surgery that we had with healthier patients.”
A vote held at the end of the session revealed that the audience was split about half and half in favor of drugs making bariatric surgery obsolete or not.
“I think we may have to duke it out now,” Dr. Aronne quipped.
Dr. Aronne disclosed being a consultant, speaker, and adviser for and receiving research support from Altimmune, Amgen, AstraZeneca, Eli Lilly, Intellihealth, Janssen, Novo Nordisk, Pfizer, Senda, UnitedHealth Group, Versanis, and others; he has ownership interests in ERX, Intellihealth, Jamieson, Kallyope, Skye Bioscience, Veru, and others; and he is on the board of directors of ERX, Jamieson Wellness, and Intellihealth/FlyteHealth. Dr. Rubino disclosed receiving research and educational grants from Novo Nordisk, Ethicon, and Medtronic; he is on the scientific advisory board/data safety advisory board for Keyron, Morphic Medical, and GT Metabolic Solutions; he receives speaking honoraria from Medtronic, Ethicon, Novo Nordisk, and Eli Lilly; and he is president of the nonprofit Metabolic Health Institute.
A version of this article first appeared on Medscape.com.
FROM EASD 2024
Hot Flashes: Do They Predict CVD and Dementia?
This transcript has been edited for clarity.
I’d like to talk about a recent report in the journal Menopause linking menopausal symptoms to increased risk for cognitive impairment. I’d also like to discuss some of the recent studies that have addressed whether hot flashes are linked to increased risk for heart disease and other forms of cardiovascular disease (CVD).
Given that 75%-80% of perimenopausal and postmenopausal women have hot flashes and vasomotor symptoms, it’s undoubtedly a more complex relationship between hot flashes and these outcomes than a simple one-size-fits-all, yes-or-no question.
Increasing evidence shows that several additional factors are important, including the age at which the symptoms are occurring, the time since menopause, the severity of the symptoms, whether they co-occur with night sweats and sleep disruption, and the cardiovascular status of the woman.
Several studies suggest that women who have more severe hot flashes and vasomotor symptoms are more likely to have prevalent cardiovascular risk factors — hypertension, dyslipidemia, high body mass index, endothelial dysfunction — as measured by flow-mediated vasodilation and other measures.
It is quite plausible that hot flashes could be a marker for increased risk for cognitive impairment. But the question remains, are hot flashes associated with cognitive impairment independent of these other risk factors? It appears that the associations between hot flashes, vasomotor symptoms, and CVD, and other adverse outcomes, may be more likely when hot flashes persist after age 60 or are newly occurring in later menopause. In the Women’s Health Initiative observational study, the presence of hot flashes and vasomotor symptoms in early menopause was not linked to any increased risk for heart attack, stroke, total CVD, or all-cause mortality.
However, the onset of these symptoms, especially new onset of these symptoms after age 60 or in later menopause, was in fact linked to increased risk for CVD and all-cause mortality. With respect to cognitive impairment, if a woman is having hot flashes and night sweats with regular sleep disruption, performance on cognitive testing would not be as favorable as it would be in the absence of these symptoms.
This brings us to the new study in Menopause that included approximately 1300 Latino women in nine Latin American countries, with an average age of 55 years. Looking at the association between severe menopausal symptoms and cognitive impairment, researchers found that women with severe symptoms were more likely to have cognitive impairment.
Conversely, they found that the women who had a favorable CVD risk factor status (physically active, lower BMI, healthier) and were ever users of estrogen were less likely to have cognitive impairment.
Clearly, for estrogen therapy, we need randomized clinical trials of the presence or absence of vasomotor symptoms and cognitive and CVD outcomes. Such analyses are ongoing, and new randomized trials focused specifically on women in early menopause would be very beneficial.
At the present time, it’s important that we not alarm women about the associations seen in some of these studies because often they are not independent associations; they aren’t independent of other risk factors that are commonly linked to hot flashes and night sweats. There are many other complexities in the relationship between hot flashes and cognitive impairment.
We need to appreciate that women who have moderate to severe hot flashes (especially when associated with disrupted sleep) do have impaired quality of life. It’s important to treat these symptoms, especially in early menopause, and very effective hormonal and nonhormonal treatments are available.
For women with symptoms that persist into later menopause or who have new onset of symptoms in later menopause, it’s important to prioritize cardiovascular health. For example, be more vigilant about behavioral lifestyle counseling to lower risk, and be even more aggressive in treating dyslipidemia and diabetes.
JoAnn E. Manson, Professor of Medicine and the Michael and Lee Bell Professor of Women’s Health, Harvard Medical School; Chief, Division of Preventive Medicine, Brigham and Women’s Hospital, Boston, Massachusetts; and Past President, North American Menopause Society, 2011-2012, has disclosed the following relevant financial relationships: Received study pill donation and infrastructure support from Mars Symbioscience (for the COSMOS trial).
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’d like to talk about a recent report in the journal Menopause linking menopausal symptoms to increased risk for cognitive impairment. I’d also like to discuss some of the recent studies that have addressed whether hot flashes are linked to increased risk for heart disease and other forms of cardiovascular disease (CVD).
Given that 75%-80% of perimenopausal and postmenopausal women have hot flashes and vasomotor symptoms, it’s undoubtedly a more complex relationship between hot flashes and these outcomes than a simple one-size-fits-all, yes-or-no question.
Increasing evidence shows that several additional factors are important, including the age at which the symptoms are occurring, the time since menopause, the severity of the symptoms, whether they co-occur with night sweats and sleep disruption, and the cardiovascular status of the woman.
Several studies suggest that women who have more severe hot flashes and vasomotor symptoms are more likely to have prevalent cardiovascular risk factors — hypertension, dyslipidemia, high body mass index, endothelial dysfunction — as measured by flow-mediated vasodilation and other measures.
It is quite plausible that hot flashes could be a marker for increased risk for cognitive impairment. But the question remains, are hot flashes associated with cognitive impairment independent of these other risk factors? It appears that the associations between hot flashes, vasomotor symptoms, and CVD, and other adverse outcomes, may be more likely when hot flashes persist after age 60 or are newly occurring in later menopause. In the Women’s Health Initiative observational study, the presence of hot flashes and vasomotor symptoms in early menopause was not linked to any increased risk for heart attack, stroke, total CVD, or all-cause mortality.
However, the onset of these symptoms, especially new onset of these symptoms after age 60 or in later menopause, was in fact linked to increased risk for CVD and all-cause mortality. With respect to cognitive impairment, if a woman is having hot flashes and night sweats with regular sleep disruption, performance on cognitive testing would not be as favorable as it would be in the absence of these symptoms.
This brings us to the new study in Menopause that included approximately 1300 Latino women in nine Latin American countries, with an average age of 55 years. Looking at the association between severe menopausal symptoms and cognitive impairment, researchers found that women with severe symptoms were more likely to have cognitive impairment.
Conversely, they found that the women who had a favorable CVD risk factor status (physically active, lower BMI, healthier) and were ever users of estrogen were less likely to have cognitive impairment.
Clearly, for estrogen therapy, we need randomized clinical trials of the presence or absence of vasomotor symptoms and cognitive and CVD outcomes. Such analyses are ongoing, and new randomized trials focused specifically on women in early menopause would be very beneficial.
At the present time, it’s important that we not alarm women about the associations seen in some of these studies because often they are not independent associations; they aren’t independent of other risk factors that are commonly linked to hot flashes and night sweats. There are many other complexities in the relationship between hot flashes and cognitive impairment.
We need to appreciate that women who have moderate to severe hot flashes (especially when associated with disrupted sleep) do have impaired quality of life. It’s important to treat these symptoms, especially in early menopause, and very effective hormonal and nonhormonal treatments are available.
For women with symptoms that persist into later menopause or who have new onset of symptoms in later menopause, it’s important to prioritize cardiovascular health. For example, be more vigilant about behavioral lifestyle counseling to lower risk, and be even more aggressive in treating dyslipidemia and diabetes.
JoAnn E. Manson, Professor of Medicine and the Michael and Lee Bell Professor of Women’s Health, Harvard Medical School; Chief, Division of Preventive Medicine, Brigham and Women’s Hospital, Boston, Massachusetts; and Past President, North American Menopause Society, 2011-2012, has disclosed the following relevant financial relationships: Received study pill donation and infrastructure support from Mars Symbioscience (for the COSMOS trial).
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’d like to talk about a recent report in the journal Menopause linking menopausal symptoms to increased risk for cognitive impairment. I’d also like to discuss some of the recent studies that have addressed whether hot flashes are linked to increased risk for heart disease and other forms of cardiovascular disease (CVD).
Given that 75%-80% of perimenopausal and postmenopausal women have hot flashes and vasomotor symptoms, it’s undoubtedly a more complex relationship between hot flashes and these outcomes than a simple one-size-fits-all, yes-or-no question.
Increasing evidence shows that several additional factors are important, including the age at which the symptoms are occurring, the time since menopause, the severity of the symptoms, whether they co-occur with night sweats and sleep disruption, and the cardiovascular status of the woman.
Several studies suggest that women who have more severe hot flashes and vasomotor symptoms are more likely to have prevalent cardiovascular risk factors — hypertension, dyslipidemia, high body mass index, endothelial dysfunction — as measured by flow-mediated vasodilation and other measures.
It is quite plausible that hot flashes could be a marker for increased risk for cognitive impairment. But the question remains, are hot flashes associated with cognitive impairment independent of these other risk factors? It appears that the associations between hot flashes, vasomotor symptoms, and CVD, and other adverse outcomes, may be more likely when hot flashes persist after age 60 or are newly occurring in later menopause. In the Women’s Health Initiative observational study, the presence of hot flashes and vasomotor symptoms in early menopause was not linked to any increased risk for heart attack, stroke, total CVD, or all-cause mortality.
However, the onset of these symptoms, especially new onset of these symptoms after age 60 or in later menopause, was in fact linked to increased risk for CVD and all-cause mortality. With respect to cognitive impairment, if a woman is having hot flashes and night sweats with regular sleep disruption, performance on cognitive testing would not be as favorable as it would be in the absence of these symptoms.
This brings us to the new study in Menopause that included approximately 1300 Latino women in nine Latin American countries, with an average age of 55 years. Looking at the association between severe menopausal symptoms and cognitive impairment, researchers found that women with severe symptoms were more likely to have cognitive impairment.
Conversely, they found that the women who had a favorable CVD risk factor status (physically active, lower BMI, healthier) and were ever users of estrogen were less likely to have cognitive impairment.
Clearly, for estrogen therapy, we need randomized clinical trials of the presence or absence of vasomotor symptoms and cognitive and CVD outcomes. Such analyses are ongoing, and new randomized trials focused specifically on women in early menopause would be very beneficial.
At the present time, it’s important that we not alarm women about the associations seen in some of these studies because often they are not independent associations; they aren’t independent of other risk factors that are commonly linked to hot flashes and night sweats. There are many other complexities in the relationship between hot flashes and cognitive impairment.
We need to appreciate that women who have moderate to severe hot flashes (especially when associated with disrupted sleep) do have impaired quality of life. It’s important to treat these symptoms, especially in early menopause, and very effective hormonal and nonhormonal treatments are available.
For women with symptoms that persist into later menopause or who have new onset of symptoms in later menopause, it’s important to prioritize cardiovascular health. For example, be more vigilant about behavioral lifestyle counseling to lower risk, and be even more aggressive in treating dyslipidemia and diabetes.
JoAnn E. Manson, Professor of Medicine and the Michael and Lee Bell Professor of Women’s Health, Harvard Medical School; Chief, Division of Preventive Medicine, Brigham and Women’s Hospital, Boston, Massachusetts; and Past President, North American Menopause Society, 2011-2012, has disclosed the following relevant financial relationships: Received study pill donation and infrastructure support from Mars Symbioscience (for the COSMOS trial).
A version of this article first appeared on Medscape.com.
‘Reform School’ for Pharmacy Benefit Managers: How Might Legislation Help Patients?
The term “reform school” is a bit outdated. It used to refer to institutions where young offenders were sent instead of prison. Some argue that pharmacy benefit managers (PBMs) should bypass reform school and go straight to prison. “PBM reform” has become a ubiquitous term, encompassing any legislative or regulatory efforts aimed at curbing PBMs’ bad behavior. When discussing PBM reform, it’s crucial to understand the various segments of the healthcare system affected by PBMs. This complexity often makes it challenging to determine what these reform packages would actually achieve and who they would benefit.
Pharmacists have long been vocal critics of PBMs, and while their issues are extremely important, it is essential to remember that the ultimate victims of PBM misconduct, in terms of access to care, are patients. At some point, we will all be patients, making this issue universally relevant. It has been quite challenging to follow federal legislation on this topic as these packages attempt to address a number of bad behaviors by PBMs affecting a variety of victims. This discussion will examine those reforms that would directly improve patient’s access to available and affordable medications.
Policy Categories of PBM Reform
There are five policy categories of PBM reform legislation overall, including three that have the greatest potential to directly address patient needs. The first is patient access to medications (utilization management, copay assistance, prior authorization, etc.), followed by delinking drug list prices from PBM income and pass-through of price concessions from the manufacturer. The remaining two categories involve transparency and pharmacy-facing reform, both of which are very important. However, this discussion will revolve around the first three categories. It should be noted that many of the legislation packages addressing the categories of patient access, delinking, and pass-through also include transparency issues, particularly as they relate to pharmacy-facing issues.
Patient Access to Medications — Step Therapy Legislation
One of the major obstacles to patient access to medications is the use of PBM utilization management tools such as step therapy (“fail first”), prior authorizations, nonmedical switching, and formulary exclusions. These tools dictate when patients can obtain necessary medications and for how long patients who are stable on their current treatments can remain on them.
While many states have enacted step therapy reforms to prevent stable patients from being whip-sawed between medications that maximize PBM profits (often labeled as “savings”), these state protections apply only to state-regulated health plans. These include fully insured health plans and those offered through the Affordable Care Act’s Health Insurance Marketplace. It also includes state employees, state corrections, and, in some cases, state labor unions. State legislation does not extend to patients covered by employer self-insured health plans, called ERISA plans for the federal law that governs employee benefit plans, the Employee Retirement Income Security Act. These ERISA plans include nearly 35 million people nationwide.
This is where the Safe Step Act (S.652/H.R.2630) becomes crucial, as it allows employees to request exceptions to harmful fail-first protocols. The bill has gained significant momentum, having been reported out of the Senate HELP Committee and discussed in House markups. The Safe Step Act would mandate that an exception to a step therapy protocol must be granted if:
- The required treatment has been ineffective
- The treatment is expected to be ineffective, and delaying effective treatment would lead to irreversible consequences
- The treatment will cause or is likely to cause an adverse reaction
- The treatment is expected to prevent the individual from performing daily activities or occupational responsibilities
- The individual is stable on their current prescription drugs
- There are other circumstances as determined by the Employee Benefits Security Administration
This legislation is vital for ensuring that patients have timely access to the medications they need without unnecessary delays or disruptions.
Patient Access to Medications — Prior Authorizations
Another significant issue affecting patient access to medications is prior authorizations (PAs). According to an American Medical Association survey, nearly one in four physicians (24%) report that a PA has led to a serious adverse event for a patient in their care. In rheumatology, PAs often result in delays in care (even for those initially approved) and a significant increase in steroid usage. In particular, PAs in Medicare Advantage (MA) plans are harmful to Medicare beneficiaries.
The Improving Seniors’ Timely Access to Care Act (H.R.8702 / S.4532) aims to reform PAs used in MA plans, making the process more efficient and transparent to improve access to care for seniors. Unfortunately, it does not cover Part D drugs and may only cover Part B drugs depending on the MA plan’s benefit package. Here are the key provisions of the act:
- Electronic PA: Implementing real-time decisions for routinely approved items and services.
- Transparency: Requiring annual publication of PA information, such as the percentage of requests approved and the average response time.
- Quality and Timeliness Standards: The Centers for Medicare & Medicaid Services (CMS) will set standards for the quality and timeliness of PA determinations.
- Streamlining Approvals: Simplifying the approval process and reducing the time allowed for health plans to consider PA requests.
This bill passed the House in September 2022 but stalled in the Senate because of an unfavorable Congressional Budget Office score. CMS has since finalized portions of this bill via regulation, zeroing out the CBO score and increasing the chances of its passage.
Delinking Drug Prices from PBM Income and Pass-Through of Price Concessions
Affordability is a crucial aspect of accessibility, especially when it comes to medications. Over the years, we’ve learned that PBMs often favor placing the highest list price drugs on formularies because the rebates and various fees they receive from manufacturers are based on a percentage of the list price. In other words, the higher the medication’s price, the more money the PBM makes.
This practice is evident in both commercial and government formularies, where brand-name drugs are often preferred, while lower-priced generics are either excluded or placed on higher tiers. As a result, while major PBMs benefit from these rebates and fees, patients continue to pay their cost share based on the list price of the medication.
To improve the affordability of medications, a key aspect of PBM reform should be to disincentivize PBMs from selecting higher-priced medications and/or require the pass-through of manufacturer price concessions to patients.
Several major PBM reform bills are currently being considered that address either the delinking of price concessions from the list price of the drug or some form of pass-through of these concessions. These reforms are essential to ensure that patients can access affordable medications without being burdened by inflated costs.
The legislation includes the Pharmacy Benefit Manager Reform Act (S.1339); the Modernizing & Ensuring PBM Accountability Act (S.2973); the Better Mental Health Care, Lower Cost Drugs, and Extenders Act (S.3430); the Protecting Patients Against PBM Abuses Act (H.R. 2880); the DRUG Act (S.2474 / H.R.6283); and the Share the Savings with Seniors Act (S.2474 / H.R.5376).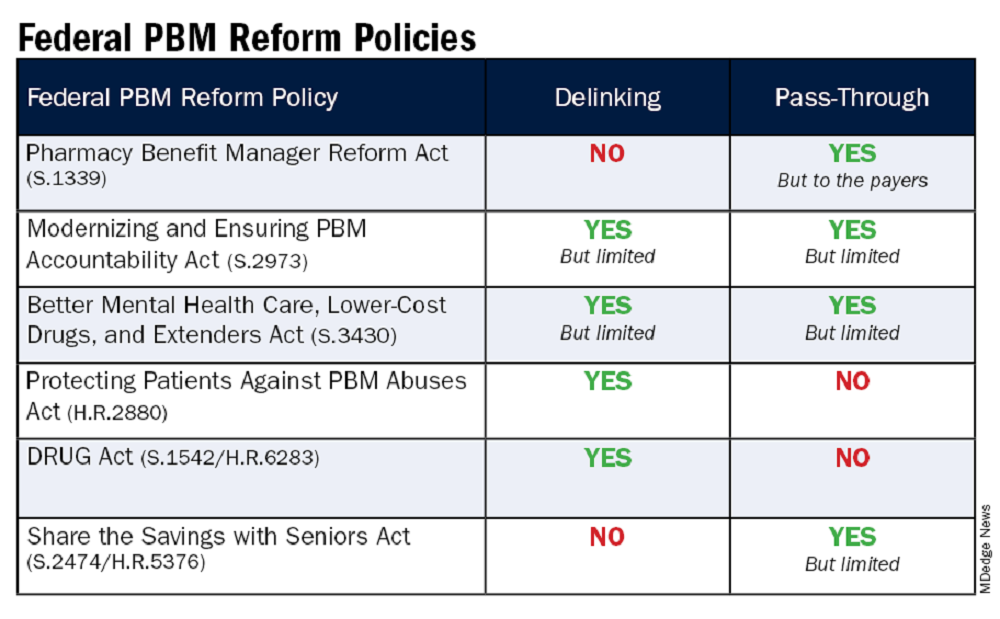
As with all legislation, there are limitations and compromises in each of these. However, these bills are a good first step in addressing PBM remuneration (rebates and fees) based on the list price of the drug and/or passing through to the patient the benefit of manufacturer price concessions. By focusing on key areas like utilization management, delinking drug prices from PBM income, and allowing patients to directly benefit from manufacturer price concessions, we can work toward a more equitable and efficient healthcare system. Reigning in PBM bad behavior is a challenge, but the potential benefits for patient care and access make it a crucial fight worth pursuing.
Please help in efforts to improve patients’ access to available and affordable medications by contacting your representatives in Congress to impart to them the importance of passing legislation. The CSRO’s legislative map tool can help to inform you of the latest information on these and other bills and assist you in engaging with your representatives on them.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s vice president of Advocacy and Government Affairs and its immediate past president, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. She has no relevant conflicts of interest to disclose. You can reach her at [email protected].
The term “reform school” is a bit outdated. It used to refer to institutions where young offenders were sent instead of prison. Some argue that pharmacy benefit managers (PBMs) should bypass reform school and go straight to prison. “PBM reform” has become a ubiquitous term, encompassing any legislative or regulatory efforts aimed at curbing PBMs’ bad behavior. When discussing PBM reform, it’s crucial to understand the various segments of the healthcare system affected by PBMs. This complexity often makes it challenging to determine what these reform packages would actually achieve and who they would benefit.
Pharmacists have long been vocal critics of PBMs, and while their issues are extremely important, it is essential to remember that the ultimate victims of PBM misconduct, in terms of access to care, are patients. At some point, we will all be patients, making this issue universally relevant. It has been quite challenging to follow federal legislation on this topic as these packages attempt to address a number of bad behaviors by PBMs affecting a variety of victims. This discussion will examine those reforms that would directly improve patient’s access to available and affordable medications.
Policy Categories of PBM Reform
There are five policy categories of PBM reform legislation overall, including three that have the greatest potential to directly address patient needs. The first is patient access to medications (utilization management, copay assistance, prior authorization, etc.), followed by delinking drug list prices from PBM income and pass-through of price concessions from the manufacturer. The remaining two categories involve transparency and pharmacy-facing reform, both of which are very important. However, this discussion will revolve around the first three categories. It should be noted that many of the legislation packages addressing the categories of patient access, delinking, and pass-through also include transparency issues, particularly as they relate to pharmacy-facing issues.
Patient Access to Medications — Step Therapy Legislation
One of the major obstacles to patient access to medications is the use of PBM utilization management tools such as step therapy (“fail first”), prior authorizations, nonmedical switching, and formulary exclusions. These tools dictate when patients can obtain necessary medications and for how long patients who are stable on their current treatments can remain on them.
While many states have enacted step therapy reforms to prevent stable patients from being whip-sawed between medications that maximize PBM profits (often labeled as “savings”), these state protections apply only to state-regulated health plans. These include fully insured health plans and those offered through the Affordable Care Act’s Health Insurance Marketplace. It also includes state employees, state corrections, and, in some cases, state labor unions. State legislation does not extend to patients covered by employer self-insured health plans, called ERISA plans for the federal law that governs employee benefit plans, the Employee Retirement Income Security Act. These ERISA plans include nearly 35 million people nationwide.
This is where the Safe Step Act (S.652/H.R.2630) becomes crucial, as it allows employees to request exceptions to harmful fail-first protocols. The bill has gained significant momentum, having been reported out of the Senate HELP Committee and discussed in House markups. The Safe Step Act would mandate that an exception to a step therapy protocol must be granted if:
- The required treatment has been ineffective
- The treatment is expected to be ineffective, and delaying effective treatment would lead to irreversible consequences
- The treatment will cause or is likely to cause an adverse reaction
- The treatment is expected to prevent the individual from performing daily activities or occupational responsibilities
- The individual is stable on their current prescription drugs
- There are other circumstances as determined by the Employee Benefits Security Administration
This legislation is vital for ensuring that patients have timely access to the medications they need without unnecessary delays or disruptions.
Patient Access to Medications — Prior Authorizations
Another significant issue affecting patient access to medications is prior authorizations (PAs). According to an American Medical Association survey, nearly one in four physicians (24%) report that a PA has led to a serious adverse event for a patient in their care. In rheumatology, PAs often result in delays in care (even for those initially approved) and a significant increase in steroid usage. In particular, PAs in Medicare Advantage (MA) plans are harmful to Medicare beneficiaries.
The Improving Seniors’ Timely Access to Care Act (H.R.8702 / S.4532) aims to reform PAs used in MA plans, making the process more efficient and transparent to improve access to care for seniors. Unfortunately, it does not cover Part D drugs and may only cover Part B drugs depending on the MA plan’s benefit package. Here are the key provisions of the act:
- Electronic PA: Implementing real-time decisions for routinely approved items and services.
- Transparency: Requiring annual publication of PA information, such as the percentage of requests approved and the average response time.
- Quality and Timeliness Standards: The Centers for Medicare & Medicaid Services (CMS) will set standards for the quality and timeliness of PA determinations.
- Streamlining Approvals: Simplifying the approval process and reducing the time allowed for health plans to consider PA requests.
This bill passed the House in September 2022 but stalled in the Senate because of an unfavorable Congressional Budget Office score. CMS has since finalized portions of this bill via regulation, zeroing out the CBO score and increasing the chances of its passage.
Delinking Drug Prices from PBM Income and Pass-Through of Price Concessions
Affordability is a crucial aspect of accessibility, especially when it comes to medications. Over the years, we’ve learned that PBMs often favor placing the highest list price drugs on formularies because the rebates and various fees they receive from manufacturers are based on a percentage of the list price. In other words, the higher the medication’s price, the more money the PBM makes.
This practice is evident in both commercial and government formularies, where brand-name drugs are often preferred, while lower-priced generics are either excluded or placed on higher tiers. As a result, while major PBMs benefit from these rebates and fees, patients continue to pay their cost share based on the list price of the medication.
To improve the affordability of medications, a key aspect of PBM reform should be to disincentivize PBMs from selecting higher-priced medications and/or require the pass-through of manufacturer price concessions to patients.
Several major PBM reform bills are currently being considered that address either the delinking of price concessions from the list price of the drug or some form of pass-through of these concessions. These reforms are essential to ensure that patients can access affordable medications without being burdened by inflated costs.
The legislation includes the Pharmacy Benefit Manager Reform Act (S.1339); the Modernizing & Ensuring PBM Accountability Act (S.2973); the Better Mental Health Care, Lower Cost Drugs, and Extenders Act (S.3430); the Protecting Patients Against PBM Abuses Act (H.R. 2880); the DRUG Act (S.2474 / H.R.6283); and the Share the Savings with Seniors Act (S.2474 / H.R.5376).
As with all legislation, there are limitations and compromises in each of these. However, these bills are a good first step in addressing PBM remuneration (rebates and fees) based on the list price of the drug and/or passing through to the patient the benefit of manufacturer price concessions. By focusing on key areas like utilization management, delinking drug prices from PBM income, and allowing patients to directly benefit from manufacturer price concessions, we can work toward a more equitable and efficient healthcare system. Reigning in PBM bad behavior is a challenge, but the potential benefits for patient care and access make it a crucial fight worth pursuing.
Please help in efforts to improve patients’ access to available and affordable medications by contacting your representatives in Congress to impart to them the importance of passing legislation. The CSRO’s legislative map tool can help to inform you of the latest information on these and other bills and assist you in engaging with your representatives on them.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s vice president of Advocacy and Government Affairs and its immediate past president, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. She has no relevant conflicts of interest to disclose. You can reach her at [email protected].
The term “reform school” is a bit outdated. It used to refer to institutions where young offenders were sent instead of prison. Some argue that pharmacy benefit managers (PBMs) should bypass reform school and go straight to prison. “PBM reform” has become a ubiquitous term, encompassing any legislative or regulatory efforts aimed at curbing PBMs’ bad behavior. When discussing PBM reform, it’s crucial to understand the various segments of the healthcare system affected by PBMs. This complexity often makes it challenging to determine what these reform packages would actually achieve and who they would benefit.
Pharmacists have long been vocal critics of PBMs, and while their issues are extremely important, it is essential to remember that the ultimate victims of PBM misconduct, in terms of access to care, are patients. At some point, we will all be patients, making this issue universally relevant. It has been quite challenging to follow federal legislation on this topic as these packages attempt to address a number of bad behaviors by PBMs affecting a variety of victims. This discussion will examine those reforms that would directly improve patient’s access to available and affordable medications.
Policy Categories of PBM Reform
There are five policy categories of PBM reform legislation overall, including three that have the greatest potential to directly address patient needs. The first is patient access to medications (utilization management, copay assistance, prior authorization, etc.), followed by delinking drug list prices from PBM income and pass-through of price concessions from the manufacturer. The remaining two categories involve transparency and pharmacy-facing reform, both of which are very important. However, this discussion will revolve around the first three categories. It should be noted that many of the legislation packages addressing the categories of patient access, delinking, and pass-through also include transparency issues, particularly as they relate to pharmacy-facing issues.
Patient Access to Medications — Step Therapy Legislation
One of the major obstacles to patient access to medications is the use of PBM utilization management tools such as step therapy (“fail first”), prior authorizations, nonmedical switching, and formulary exclusions. These tools dictate when patients can obtain necessary medications and for how long patients who are stable on their current treatments can remain on them.
While many states have enacted step therapy reforms to prevent stable patients from being whip-sawed between medications that maximize PBM profits (often labeled as “savings”), these state protections apply only to state-regulated health plans. These include fully insured health plans and those offered through the Affordable Care Act’s Health Insurance Marketplace. It also includes state employees, state corrections, and, in some cases, state labor unions. State legislation does not extend to patients covered by employer self-insured health plans, called ERISA plans for the federal law that governs employee benefit plans, the Employee Retirement Income Security Act. These ERISA plans include nearly 35 million people nationwide.
This is where the Safe Step Act (S.652/H.R.2630) becomes crucial, as it allows employees to request exceptions to harmful fail-first protocols. The bill has gained significant momentum, having been reported out of the Senate HELP Committee and discussed in House markups. The Safe Step Act would mandate that an exception to a step therapy protocol must be granted if:
- The required treatment has been ineffective
- The treatment is expected to be ineffective, and delaying effective treatment would lead to irreversible consequences
- The treatment will cause or is likely to cause an adverse reaction
- The treatment is expected to prevent the individual from performing daily activities or occupational responsibilities
- The individual is stable on their current prescription drugs
- There are other circumstances as determined by the Employee Benefits Security Administration
This legislation is vital for ensuring that patients have timely access to the medications they need without unnecessary delays or disruptions.
Patient Access to Medications — Prior Authorizations
Another significant issue affecting patient access to medications is prior authorizations (PAs). According to an American Medical Association survey, nearly one in four physicians (24%) report that a PA has led to a serious adverse event for a patient in their care. In rheumatology, PAs often result in delays in care (even for those initially approved) and a significant increase in steroid usage. In particular, PAs in Medicare Advantage (MA) plans are harmful to Medicare beneficiaries.
The Improving Seniors’ Timely Access to Care Act (H.R.8702 / S.4532) aims to reform PAs used in MA plans, making the process more efficient and transparent to improve access to care for seniors. Unfortunately, it does not cover Part D drugs and may only cover Part B drugs depending on the MA plan’s benefit package. Here are the key provisions of the act:
- Electronic PA: Implementing real-time decisions for routinely approved items and services.
- Transparency: Requiring annual publication of PA information, such as the percentage of requests approved and the average response time.
- Quality and Timeliness Standards: The Centers for Medicare & Medicaid Services (CMS) will set standards for the quality and timeliness of PA determinations.
- Streamlining Approvals: Simplifying the approval process and reducing the time allowed for health plans to consider PA requests.
This bill passed the House in September 2022 but stalled in the Senate because of an unfavorable Congressional Budget Office score. CMS has since finalized portions of this bill via regulation, zeroing out the CBO score and increasing the chances of its passage.
Delinking Drug Prices from PBM Income and Pass-Through of Price Concessions
Affordability is a crucial aspect of accessibility, especially when it comes to medications. Over the years, we’ve learned that PBMs often favor placing the highest list price drugs on formularies because the rebates and various fees they receive from manufacturers are based on a percentage of the list price. In other words, the higher the medication’s price, the more money the PBM makes.
This practice is evident in both commercial and government formularies, where brand-name drugs are often preferred, while lower-priced generics are either excluded or placed on higher tiers. As a result, while major PBMs benefit from these rebates and fees, patients continue to pay their cost share based on the list price of the medication.
To improve the affordability of medications, a key aspect of PBM reform should be to disincentivize PBMs from selecting higher-priced medications and/or require the pass-through of manufacturer price concessions to patients.
Several major PBM reform bills are currently being considered that address either the delinking of price concessions from the list price of the drug or some form of pass-through of these concessions. These reforms are essential to ensure that patients can access affordable medications without being burdened by inflated costs.
The legislation includes the Pharmacy Benefit Manager Reform Act (S.1339); the Modernizing & Ensuring PBM Accountability Act (S.2973); the Better Mental Health Care, Lower Cost Drugs, and Extenders Act (S.3430); the Protecting Patients Against PBM Abuses Act (H.R. 2880); the DRUG Act (S.2474 / H.R.6283); and the Share the Savings with Seniors Act (S.2474 / H.R.5376).
As with all legislation, there are limitations and compromises in each of these. However, these bills are a good first step in addressing PBM remuneration (rebates and fees) based on the list price of the drug and/or passing through to the patient the benefit of manufacturer price concessions. By focusing on key areas like utilization management, delinking drug prices from PBM income, and allowing patients to directly benefit from manufacturer price concessions, we can work toward a more equitable and efficient healthcare system. Reigning in PBM bad behavior is a challenge, but the potential benefits for patient care and access make it a crucial fight worth pursuing.
Please help in efforts to improve patients’ access to available and affordable medications by contacting your representatives in Congress to impart to them the importance of passing legislation. The CSRO’s legislative map tool can help to inform you of the latest information on these and other bills and assist you in engaging with your representatives on them.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s vice president of Advocacy and Government Affairs and its immediate past president, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. She has no relevant conflicts of interest to disclose. You can reach her at [email protected].
Stones, Bones, Groans, and Moans: Could This Be Primary Hyperparathyroidism?
This transcript has been edited for clarity.
Matthew F. Watto, MD: Welcome back to The Curbsiders. I’m Dr Matthew Frank Watto, here with my great friend and America’s primary care physician, Dr. Paul Nelson Williams.
Paul, we’re going to talk about our primary hyperparathyroidism podcast with Dr. Lindsay Kuo. It’s a topic that I feel much more clear on now.
Now, Paul, in primary care, you see a lot of calcium that is just slightly high. Can we just blame that on thiazide diuretics?
Paul N. Williams, MD: It’s a place to start. As you’re starting to think about the possible etiologies, primary hyperparathyroidism and malignancy are the two that roll right off the tongue, but it is worth going back to the patient’s medication list and making sure you’re not missing something.
Thiazides famously cause hypercalcemia, but in some of the reading I did for this episode, they may just uncover it a little bit early. Patients who are on thiazides who become hypercalcemic seem to go on to develop primary hyperthyroidism anyway. So I don’t think you can solely blame the thiazide.
Another medication that can be causative is lithium. So a good place to look first after you’ve repeated the labs and confirmed hypercalcemia is the patient’s medication list.
Dr. Watto: We’ve talked before about the basic workup for hypercalcemia, and determining whether it’s PTH dependent or PTH independent. On the podcast, we talk more about the full workup, but I wanted to talk about the classic symptoms. Our expert made the point that we don’t see them as much anymore, although we do see kidney stones. People used to present very late in the disease because they weren’t having labs done routinely.
The classic symptoms include osteoporosis and bone tumors. People can get nephrocalcinosis and kidney stones. I hadn’t really thought of it this way because we’re used to diagnosing it early now. Do you feel the same?
Dr. Williams: As labs have started routinely reporting calcium levels, this is more and more often how it’s picked up. The other aspect is that as we are screening for and finding osteoporosis, part of the workup almost always involves getting a parathyroid hormone and a calcium level. We’re seeing these lab abnormalities before we’re seeing symptoms, which is good.
But it also makes things more diagnostically thorny.
Dr. Watto: Dr. Lindsay Kuo made the point that when she sees patients before and after surgery, she’s aware of these nonclassic symptoms — the stones, bones, groans, and the psychiatric overtones that can be anything from fatigue or irritability to dysphoria.
Some people have a generalized weakness that’s very nonspecific. Dr. Kuo said that sometimes these symptoms will disappear after surgery. The patients may just have gotten used to them, or they thought these symptoms were caused by something else, but after surgery they went away.
There are these nonclassic symptoms that are harder to pin down. I was surprised by that.
Dr. Williams: She mentioned polydipsia and polyuria, which have been reported in other studies. It seems like it can be anything. You have to take a good history, but none of those things in and of themselves is an indication for operating unless the patient has the classic renal or bone manifestations.
Dr. Watto: The other thing we talked about is a normal calcium level in a patient with primary hyperparathyroidism, or the finding of a PTH level in the normal range but with a high calcium level that is inappropriate. Can you talk a little bit about those two situations?
Dr. Williams: They’re hard to say but kind of easy to manage because you treat them the same way as someone who has elevated calcium and PTH levels.
The normocalcemic patient is something we might stumble across with osteoporosis screening. Initially the calcium level is elevated, so you repeat it and it’s normal but with an elevated PTH level. You’re like, shoot. Now what?
It turns out that most endocrine surgeons say that the indications for surgery for the classic form of primary hyperparathyroidism apply to these patients as well, and it probably helps with the bone outcomes, which is one of the things they follow most closely. If you have hypercalcemia, you should have a suppressed PTH level, the so-called normohormonal hyperparathyroidism, which is not normal at all. So even if the PTH is in the normal range, it’s still relatively elevated compared with what it should be. That situation is treated in the same way as the classic elevated PTH and elevated calcium levels.
Dr. Watto: If the calcium is abnormal and the PTH is not quite what you’d expect it to be, you can always ask your friendly neighborhood endocrinologist to help you figure out whether the patient really has one of these conditions. You have to make sure that they don’t have a simple secondary cause like a low vitamin D level. In that case, you fix the vitamin D and then recheck the numbers to see if they’ve normalized. But I have found a bunch of these edge cases in which it has been helpful to confer with an endocrinologist, especially before you send someone to a surgeon to take out their parathyroid gland.
This was a really fantastic conversation. If you want to hear the full podcast episode, click here.
Dr. Watto, Clinical Assistant Professor, Department of Medicine, Perelman School of Medicine at University of Pennsylvania; Internist, Department of Medicine, Hospital Medicine Section, Pennsylvania Hospital, Philadelphia, Pennsylvania, has disclosed no relevant financial relationships. Dr. Williams, Associate Professor of Clinical Medicine, Department of General Internal Medicine, Lewis Katz School of Medicine; Staff Physician, Department of General Internal Medicine, Temple Internal Medicine Associates, Philadelphia, Pennsylvania, served as a director, officer, partner, employee, adviser, consultant, or trustee for The Curbsiders, and has received income in an amount equal to or greater than $250 from The Curbsiders.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Matthew F. Watto, MD: Welcome back to The Curbsiders. I’m Dr Matthew Frank Watto, here with my great friend and America’s primary care physician, Dr. Paul Nelson Williams.
Paul, we’re going to talk about our primary hyperparathyroidism podcast with Dr. Lindsay Kuo. It’s a topic that I feel much more clear on now.
Now, Paul, in primary care, you see a lot of calcium that is just slightly high. Can we just blame that on thiazide diuretics?
Paul N. Williams, MD: It’s a place to start. As you’re starting to think about the possible etiologies, primary hyperparathyroidism and malignancy are the two that roll right off the tongue, but it is worth going back to the patient’s medication list and making sure you’re not missing something.
Thiazides famously cause hypercalcemia, but in some of the reading I did for this episode, they may just uncover it a little bit early. Patients who are on thiazides who become hypercalcemic seem to go on to develop primary hyperthyroidism anyway. So I don’t think you can solely blame the thiazide.
Another medication that can be causative is lithium. So a good place to look first after you’ve repeated the labs and confirmed hypercalcemia is the patient’s medication list.
Dr. Watto: We’ve talked before about the basic workup for hypercalcemia, and determining whether it’s PTH dependent or PTH independent. On the podcast, we talk more about the full workup, but I wanted to talk about the classic symptoms. Our expert made the point that we don’t see them as much anymore, although we do see kidney stones. People used to present very late in the disease because they weren’t having labs done routinely.
The classic symptoms include osteoporosis and bone tumors. People can get nephrocalcinosis and kidney stones. I hadn’t really thought of it this way because we’re used to diagnosing it early now. Do you feel the same?
Dr. Williams: As labs have started routinely reporting calcium levels, this is more and more often how it’s picked up. The other aspect is that as we are screening for and finding osteoporosis, part of the workup almost always involves getting a parathyroid hormone and a calcium level. We’re seeing these lab abnormalities before we’re seeing symptoms, which is good.
But it also makes things more diagnostically thorny.
Dr. Watto: Dr. Lindsay Kuo made the point that when she sees patients before and after surgery, she’s aware of these nonclassic symptoms — the stones, bones, groans, and the psychiatric overtones that can be anything from fatigue or irritability to dysphoria.
Some people have a generalized weakness that’s very nonspecific. Dr. Kuo said that sometimes these symptoms will disappear after surgery. The patients may just have gotten used to them, or they thought these symptoms were caused by something else, but after surgery they went away.
There are these nonclassic symptoms that are harder to pin down. I was surprised by that.
Dr. Williams: She mentioned polydipsia and polyuria, which have been reported in other studies. It seems like it can be anything. You have to take a good history, but none of those things in and of themselves is an indication for operating unless the patient has the classic renal or bone manifestations.
Dr. Watto: The other thing we talked about is a normal calcium level in a patient with primary hyperparathyroidism, or the finding of a PTH level in the normal range but with a high calcium level that is inappropriate. Can you talk a little bit about those two situations?
Dr. Williams: They’re hard to say but kind of easy to manage because you treat them the same way as someone who has elevated calcium and PTH levels.
The normocalcemic patient is something we might stumble across with osteoporosis screening. Initially the calcium level is elevated, so you repeat it and it’s normal but with an elevated PTH level. You’re like, shoot. Now what?
It turns out that most endocrine surgeons say that the indications for surgery for the classic form of primary hyperparathyroidism apply to these patients as well, and it probably helps with the bone outcomes, which is one of the things they follow most closely. If you have hypercalcemia, you should have a suppressed PTH level, the so-called normohormonal hyperparathyroidism, which is not normal at all. So even if the PTH is in the normal range, it’s still relatively elevated compared with what it should be. That situation is treated in the same way as the classic elevated PTH and elevated calcium levels.
Dr. Watto: If the calcium is abnormal and the PTH is not quite what you’d expect it to be, you can always ask your friendly neighborhood endocrinologist to help you figure out whether the patient really has one of these conditions. You have to make sure that they don’t have a simple secondary cause like a low vitamin D level. In that case, you fix the vitamin D and then recheck the numbers to see if they’ve normalized. But I have found a bunch of these edge cases in which it has been helpful to confer with an endocrinologist, especially before you send someone to a surgeon to take out their parathyroid gland.
This was a really fantastic conversation. If you want to hear the full podcast episode, click here.
Dr. Watto, Clinical Assistant Professor, Department of Medicine, Perelman School of Medicine at University of Pennsylvania; Internist, Department of Medicine, Hospital Medicine Section, Pennsylvania Hospital, Philadelphia, Pennsylvania, has disclosed no relevant financial relationships. Dr. Williams, Associate Professor of Clinical Medicine, Department of General Internal Medicine, Lewis Katz School of Medicine; Staff Physician, Department of General Internal Medicine, Temple Internal Medicine Associates, Philadelphia, Pennsylvania, served as a director, officer, partner, employee, adviser, consultant, or trustee for The Curbsiders, and has received income in an amount equal to or greater than $250 from The Curbsiders.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Matthew F. Watto, MD: Welcome back to The Curbsiders. I’m Dr Matthew Frank Watto, here with my great friend and America’s primary care physician, Dr. Paul Nelson Williams.
Paul, we’re going to talk about our primary hyperparathyroidism podcast with Dr. Lindsay Kuo. It’s a topic that I feel much more clear on now.
Now, Paul, in primary care, you see a lot of calcium that is just slightly high. Can we just blame that on thiazide diuretics?
Paul N. Williams, MD: It’s a place to start. As you’re starting to think about the possible etiologies, primary hyperparathyroidism and malignancy are the two that roll right off the tongue, but it is worth going back to the patient’s medication list and making sure you’re not missing something.
Thiazides famously cause hypercalcemia, but in some of the reading I did for this episode, they may just uncover it a little bit early. Patients who are on thiazides who become hypercalcemic seem to go on to develop primary hyperthyroidism anyway. So I don’t think you can solely blame the thiazide.
Another medication that can be causative is lithium. So a good place to look first after you’ve repeated the labs and confirmed hypercalcemia is the patient’s medication list.
Dr. Watto: We’ve talked before about the basic workup for hypercalcemia, and determining whether it’s PTH dependent or PTH independent. On the podcast, we talk more about the full workup, but I wanted to talk about the classic symptoms. Our expert made the point that we don’t see them as much anymore, although we do see kidney stones. People used to present very late in the disease because they weren’t having labs done routinely.
The classic symptoms include osteoporosis and bone tumors. People can get nephrocalcinosis and kidney stones. I hadn’t really thought of it this way because we’re used to diagnosing it early now. Do you feel the same?
Dr. Williams: As labs have started routinely reporting calcium levels, this is more and more often how it’s picked up. The other aspect is that as we are screening for and finding osteoporosis, part of the workup almost always involves getting a parathyroid hormone and a calcium level. We’re seeing these lab abnormalities before we’re seeing symptoms, which is good.
But it also makes things more diagnostically thorny.
Dr. Watto: Dr. Lindsay Kuo made the point that when she sees patients before and after surgery, she’s aware of these nonclassic symptoms — the stones, bones, groans, and the psychiatric overtones that can be anything from fatigue or irritability to dysphoria.
Some people have a generalized weakness that’s very nonspecific. Dr. Kuo said that sometimes these symptoms will disappear after surgery. The patients may just have gotten used to them, or they thought these symptoms were caused by something else, but after surgery they went away.
There are these nonclassic symptoms that are harder to pin down. I was surprised by that.
Dr. Williams: She mentioned polydipsia and polyuria, which have been reported in other studies. It seems like it can be anything. You have to take a good history, but none of those things in and of themselves is an indication for operating unless the patient has the classic renal or bone manifestations.
Dr. Watto: The other thing we talked about is a normal calcium level in a patient with primary hyperparathyroidism, or the finding of a PTH level in the normal range but with a high calcium level that is inappropriate. Can you talk a little bit about those two situations?
Dr. Williams: They’re hard to say but kind of easy to manage because you treat them the same way as someone who has elevated calcium and PTH levels.
The normocalcemic patient is something we might stumble across with osteoporosis screening. Initially the calcium level is elevated, so you repeat it and it’s normal but with an elevated PTH level. You’re like, shoot. Now what?
It turns out that most endocrine surgeons say that the indications for surgery for the classic form of primary hyperparathyroidism apply to these patients as well, and it probably helps with the bone outcomes, which is one of the things they follow most closely. If you have hypercalcemia, you should have a suppressed PTH level, the so-called normohormonal hyperparathyroidism, which is not normal at all. So even if the PTH is in the normal range, it’s still relatively elevated compared with what it should be. That situation is treated in the same way as the classic elevated PTH and elevated calcium levels.
Dr. Watto: If the calcium is abnormal and the PTH is not quite what you’d expect it to be, you can always ask your friendly neighborhood endocrinologist to help you figure out whether the patient really has one of these conditions. You have to make sure that they don’t have a simple secondary cause like a low vitamin D level. In that case, you fix the vitamin D and then recheck the numbers to see if they’ve normalized. But I have found a bunch of these edge cases in which it has been helpful to confer with an endocrinologist, especially before you send someone to a surgeon to take out their parathyroid gland.
This was a really fantastic conversation. If you want to hear the full podcast episode, click here.
Dr. Watto, Clinical Assistant Professor, Department of Medicine, Perelman School of Medicine at University of Pennsylvania; Internist, Department of Medicine, Hospital Medicine Section, Pennsylvania Hospital, Philadelphia, Pennsylvania, has disclosed no relevant financial relationships. Dr. Williams, Associate Professor of Clinical Medicine, Department of General Internal Medicine, Lewis Katz School of Medicine; Staff Physician, Department of General Internal Medicine, Temple Internal Medicine Associates, Philadelphia, Pennsylvania, served as a director, officer, partner, employee, adviser, consultant, or trustee for The Curbsiders, and has received income in an amount equal to or greater than $250 from The Curbsiders.
A version of this article first appeared on Medscape.com.
AI-Powered Clinical Documentation Tool Reduces EHR Time for Clinicians
TOPLINE:
An artificial intelligence (AI)-powered clinical documentation tool helped reduce time spent on electronic health records (EHR) at home for almost 48% physicians, and nearly 45% reported less weekly time spent on EHR tasks outside of normal work hours.
METHODOLOGY:
- Researchers recruited 112 clinicians from family medicine, internal medicine, and general pediatrics in North Carolina and Georgia.
- Patients were divided into an intervention group (n = 85) and control group (n = 55), with the intervention group receiving a 1-hour training program on a commercially available AI tool.
- A seven-question survey was administered to participants before and 5 weeks after the intervention to evaluate their experience.
TAKEAWAY:
- The researchers found 47.1% of clinicians in the intervention group reported spending less time on the EHR at home compared with 14.5% in the control group (P < .001); 44.7% reported decreased weekly time on the EHR outside normal work hours compared with 20% in the control group (P = .003).
- The study revealed 43.5% of physicians who used the AI instrument reported spending less time on documentation after visits compared with 18.2% in the control group (P = .002).
- Further, 44.7% reported less frustration when using the EHR compared with 14.5% in the control group (P < .001).
IN PRACTICE:
“Approximately half of clinicians using the AI-powered clinical documentation tool based on interest reported a positive outcome, potentially reducing burnout. However, a significant subset did not find time-saving benefits or improved EHR experience,” the authors of the study wrote.
SOURCE:
The study was led by Tsai-Ling Liu, PhD, Center for Health System Sciences, Atrium Health in Charlotte, North Carolina. It was published online in JAMA Network Open.
LIMITATIONS:
The researchers reported potential selection and recall bias in both groups. Additional research is needed to find areas of improvement and assess the effects on clinician groups and health systems, they said.
DISCLOSURES:
Andrew McWilliams, MD, MPH, reported receiving grants from the Agency for Healthcare Research Quality, the National Institutes of Health, and the Duke Endowment unrelated to this work. Ajay Dharod, MD, reported his role as an electronic health record consultant for the Association of American Medical College CORE program. Jeffrey Cleveland, MD, disclosed his participation on the Executive Client Council, a noncompensated advisory group, for Nuance/Microsoft.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
An artificial intelligence (AI)-powered clinical documentation tool helped reduce time spent on electronic health records (EHR) at home for almost 48% physicians, and nearly 45% reported less weekly time spent on EHR tasks outside of normal work hours.
METHODOLOGY:
- Researchers recruited 112 clinicians from family medicine, internal medicine, and general pediatrics in North Carolina and Georgia.
- Patients were divided into an intervention group (n = 85) and control group (n = 55), with the intervention group receiving a 1-hour training program on a commercially available AI tool.
- A seven-question survey was administered to participants before and 5 weeks after the intervention to evaluate their experience.
TAKEAWAY:
- The researchers found 47.1% of clinicians in the intervention group reported spending less time on the EHR at home compared with 14.5% in the control group (P < .001); 44.7% reported decreased weekly time on the EHR outside normal work hours compared with 20% in the control group (P = .003).
- The study revealed 43.5% of physicians who used the AI instrument reported spending less time on documentation after visits compared with 18.2% in the control group (P = .002).
- Further, 44.7% reported less frustration when using the EHR compared with 14.5% in the control group (P < .001).
IN PRACTICE:
“Approximately half of clinicians using the AI-powered clinical documentation tool based on interest reported a positive outcome, potentially reducing burnout. However, a significant subset did not find time-saving benefits or improved EHR experience,” the authors of the study wrote.
SOURCE:
The study was led by Tsai-Ling Liu, PhD, Center for Health System Sciences, Atrium Health in Charlotte, North Carolina. It was published online in JAMA Network Open.
LIMITATIONS:
The researchers reported potential selection and recall bias in both groups. Additional research is needed to find areas of improvement and assess the effects on clinician groups and health systems, they said.
DISCLOSURES:
Andrew McWilliams, MD, MPH, reported receiving grants from the Agency for Healthcare Research Quality, the National Institutes of Health, and the Duke Endowment unrelated to this work. Ajay Dharod, MD, reported his role as an electronic health record consultant for the Association of American Medical College CORE program. Jeffrey Cleveland, MD, disclosed his participation on the Executive Client Council, a noncompensated advisory group, for Nuance/Microsoft.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
An artificial intelligence (AI)-powered clinical documentation tool helped reduce time spent on electronic health records (EHR) at home for almost 48% physicians, and nearly 45% reported less weekly time spent on EHR tasks outside of normal work hours.
METHODOLOGY:
- Researchers recruited 112 clinicians from family medicine, internal medicine, and general pediatrics in North Carolina and Georgia.
- Patients were divided into an intervention group (n = 85) and control group (n = 55), with the intervention group receiving a 1-hour training program on a commercially available AI tool.
- A seven-question survey was administered to participants before and 5 weeks after the intervention to evaluate their experience.
TAKEAWAY:
- The researchers found 47.1% of clinicians in the intervention group reported spending less time on the EHR at home compared with 14.5% in the control group (P < .001); 44.7% reported decreased weekly time on the EHR outside normal work hours compared with 20% in the control group (P = .003).
- The study revealed 43.5% of physicians who used the AI instrument reported spending less time on documentation after visits compared with 18.2% in the control group (P = .002).
- Further, 44.7% reported less frustration when using the EHR compared with 14.5% in the control group (P < .001).
IN PRACTICE:
“Approximately half of clinicians using the AI-powered clinical documentation tool based on interest reported a positive outcome, potentially reducing burnout. However, a significant subset did not find time-saving benefits or improved EHR experience,” the authors of the study wrote.
SOURCE:
The study was led by Tsai-Ling Liu, PhD, Center for Health System Sciences, Atrium Health in Charlotte, North Carolina. It was published online in JAMA Network Open.
LIMITATIONS:
The researchers reported potential selection and recall bias in both groups. Additional research is needed to find areas of improvement and assess the effects on clinician groups and health systems, they said.
DISCLOSURES:
Andrew McWilliams, MD, MPH, reported receiving grants from the Agency for Healthcare Research Quality, the National Institutes of Health, and the Duke Endowment unrelated to this work. Ajay Dharod, MD, reported his role as an electronic health record consultant for the Association of American Medical College CORE program. Jeffrey Cleveland, MD, disclosed his participation on the Executive Client Council, a noncompensated advisory group, for Nuance/Microsoft.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
GI-Targeted Bitter Hop Extract Curbs Hunger, Food Cravings
TOPLINE:
METHODOLOGY:
- Researchers conducted a randomized, double-blind, crossover study with 30 normal-weight women ages 18-40 with BMIs of 18.5-25. The trial design was similar to that used previously to test the intervention among men.
- Participants engaged in a water-only fast for 24 hours (6 p.m. to 6 p.m.) on three occasions and were given an ad libitum meal to break each fast.
- Participants were randomly assigned to one of three treatments: placebo, a high-dose (500 mg) bitter hop–based appetite suppressant (Amarasate), or a low dose (250 mg) of the appetite suppressant.
- Treatment capsules of half the total dose were given twice daily (16 hours and 20 hours into the fast).
- Participants recorded their subjective feelings about appetite and food cravings using a visual analog scale at 30-minute intervals starting 16 hours into the fast.
TAKEAWAY:
- Both the high-dose and low-dose treatment groups experienced a significant reduction in appetite (ie, hunger, fullness, satisfaction, thoughts of food) and food cravings compared with the placebo group.
- Energy intake at the ad libitum meal was 14.3% lower in the high-dose group than the placebo group (P < .05). It was 8.1% lower in the low-dose group, but the difference was not statistically significant.
- In the high-dose group, two participants reported loose stools and one reported heartburn. In the low-dose group, one participant reported loose stools.
IN PRACTICE:
“These data suggest that appetite suppressant co-therapy may be useful in reducing hunger during fasting in women and show that gastrointestinal delivery of bitter compounds may also be an effective method of reducing cravings for food,” the authors wrote.
SOURCE:
The study, led by Edward Walker, PhD, of the New Zealand Institute for Plant and Food Research Limited, Auckland, New Zealand, was published online in Obesity Pillars.
LIMITATIONS:
The study has limitations that affect its application for weight management. First, the study included only normal-weight individuals, which limits extension of the results for weight loss treatments. Second, it involved acute fasting and does not assess any accommodation to the intervention that may occur over longer-term use. An additional limitation is the small number of participants.
DISCLOSURES:
The research was funded by Calocurb Limited. The article processing charge was funded by the New Zealand Institute for Plant and Food Research Limited, a New Zealand government–owned Crown Research Institute. The authors declare no competing financial interests. The New Zealand Institute for Plant and Food Research Limited has licensed a hop extract as a dietary supplement to Calocurb to commercialize and currently holds a minor shareholding in this company.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Researchers conducted a randomized, double-blind, crossover study with 30 normal-weight women ages 18-40 with BMIs of 18.5-25. The trial design was similar to that used previously to test the intervention among men.
- Participants engaged in a water-only fast for 24 hours (6 p.m. to 6 p.m.) on three occasions and were given an ad libitum meal to break each fast.
- Participants were randomly assigned to one of three treatments: placebo, a high-dose (500 mg) bitter hop–based appetite suppressant (Amarasate), or a low dose (250 mg) of the appetite suppressant.
- Treatment capsules of half the total dose were given twice daily (16 hours and 20 hours into the fast).
- Participants recorded their subjective feelings about appetite and food cravings using a visual analog scale at 30-minute intervals starting 16 hours into the fast.
TAKEAWAY:
- Both the high-dose and low-dose treatment groups experienced a significant reduction in appetite (ie, hunger, fullness, satisfaction, thoughts of food) and food cravings compared with the placebo group.
- Energy intake at the ad libitum meal was 14.3% lower in the high-dose group than the placebo group (P < .05). It was 8.1% lower in the low-dose group, but the difference was not statistically significant.
- In the high-dose group, two participants reported loose stools and one reported heartburn. In the low-dose group, one participant reported loose stools.
IN PRACTICE:
“These data suggest that appetite suppressant co-therapy may be useful in reducing hunger during fasting in women and show that gastrointestinal delivery of bitter compounds may also be an effective method of reducing cravings for food,” the authors wrote.
SOURCE:
The study, led by Edward Walker, PhD, of the New Zealand Institute for Plant and Food Research Limited, Auckland, New Zealand, was published online in Obesity Pillars.
LIMITATIONS:
The study has limitations that affect its application for weight management. First, the study included only normal-weight individuals, which limits extension of the results for weight loss treatments. Second, it involved acute fasting and does not assess any accommodation to the intervention that may occur over longer-term use. An additional limitation is the small number of participants.
DISCLOSURES:
The research was funded by Calocurb Limited. The article processing charge was funded by the New Zealand Institute for Plant and Food Research Limited, a New Zealand government–owned Crown Research Institute. The authors declare no competing financial interests. The New Zealand Institute for Plant and Food Research Limited has licensed a hop extract as a dietary supplement to Calocurb to commercialize and currently holds a minor shareholding in this company.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Researchers conducted a randomized, double-blind, crossover study with 30 normal-weight women ages 18-40 with BMIs of 18.5-25. The trial design was similar to that used previously to test the intervention among men.
- Participants engaged in a water-only fast for 24 hours (6 p.m. to 6 p.m.) on three occasions and were given an ad libitum meal to break each fast.
- Participants were randomly assigned to one of three treatments: placebo, a high-dose (500 mg) bitter hop–based appetite suppressant (Amarasate), or a low dose (250 mg) of the appetite suppressant.
- Treatment capsules of half the total dose were given twice daily (16 hours and 20 hours into the fast).
- Participants recorded their subjective feelings about appetite and food cravings using a visual analog scale at 30-minute intervals starting 16 hours into the fast.
TAKEAWAY:
- Both the high-dose and low-dose treatment groups experienced a significant reduction in appetite (ie, hunger, fullness, satisfaction, thoughts of food) and food cravings compared with the placebo group.
- Energy intake at the ad libitum meal was 14.3% lower in the high-dose group than the placebo group (P < .05). It was 8.1% lower in the low-dose group, but the difference was not statistically significant.
- In the high-dose group, two participants reported loose stools and one reported heartburn. In the low-dose group, one participant reported loose stools.
IN PRACTICE:
“These data suggest that appetite suppressant co-therapy may be useful in reducing hunger during fasting in women and show that gastrointestinal delivery of bitter compounds may also be an effective method of reducing cravings for food,” the authors wrote.
SOURCE:
The study, led by Edward Walker, PhD, of the New Zealand Institute for Plant and Food Research Limited, Auckland, New Zealand, was published online in Obesity Pillars.
LIMITATIONS:
The study has limitations that affect its application for weight management. First, the study included only normal-weight individuals, which limits extension of the results for weight loss treatments. Second, it involved acute fasting and does not assess any accommodation to the intervention that may occur over longer-term use. An additional limitation is the small number of participants.
DISCLOSURES:
The research was funded by Calocurb Limited. The article processing charge was funded by the New Zealand Institute for Plant and Food Research Limited, a New Zealand government–owned Crown Research Institute. The authors declare no competing financial interests. The New Zealand Institute for Plant and Food Research Limited has licensed a hop extract as a dietary supplement to Calocurb to commercialize and currently holds a minor shareholding in this company.
A version of this article first appeared on Medscape.com.
SGLT2 Inhibitor Reduces Risk for Neurodegenerative Diseases in T2D
MADRID — Patients with type 2 diabetes treated with sodium-glucose cotransporter 2 inhibitors (SGLT2is) show significant reductions in the risk of developing neurodegenerative disorders including Alzheimer’s disease, vascular dementia, and Parkinson’s disease, compared with those treated with other antidiabetic drugs, results from a large population-based cohort show.
“This was the largest nationwide population-based longitudinal cohort study to investigate the association between the use of SGLT2 inhibitors and the incidence of all-cause dementia and Parkinson’s disease,” said first author Hae Kyung Kim, MD, of the Department of Internal Medicine, Yonsei University College of Medicine, Seoul, South Korea, in presenting the findings at the annual meeting of the European Association for the Study of Diabetes.
Type 2 diabetes is known to increase the risk for neurodegenerative diseases such as dementia or Alzheimer’s disease, said Dr. Kim. Key factors attributed to the risk include shared pathophysiological mechanisms such as central nervous system insulin resistance and reduced cerebral glucose metabolism.
While research is lacking on the role of antidiabetic drugs in the treatment of neurodegenerative diseases, the researcher noted that “SGLT2 inhibitors, which have shown significant cardiorenal benefits and enhanced energy metabolism through ketogenesis, offer promise.”
To further investigate, Dr. Kim and her colleagues conducted the retrospective study, evaluating data on more than 1.3 million enrollees in Korea’s National Health Insurance Service Database who were aged 40 years or older, diagnosed with type 2 diabetes, and had initiated antidiabetic drugs between September 2014 and December 2019.
In the propensity score analysis, 358,862 patients were matched 1:1, in groups of 179,431 participants each, based on whether they were treated with SGLT2is or other oral antidiabetic drugs. Patients with a history of neurodegenerative disease, cancer, or use of glucagon-like peptide 1 receptor agonists were excluded.
The patients had a mean age of 57.8 years, 57.9% were men, and 6837 had incident dementia or Parkinson’s disease events reported.
With a mean follow-up of 2.88 years, after adjustment for key variables, those treated with SGLT2is had a 19% reduced risk of developing Alzheimer’s disease (adjusted hazard ratio [aHR], 0.81), a 31% reduced risk for vascular dementia (aHR, 0.69), and a 20% reduced risk for Parkinson’s disease (aHR, 0.80) compared with the non-SGLT2i group.
Furthermore, those receiving SGLT2i treatment had a 21% reduced risk for all-cause dementia (aHR, 0.79) and a 22% reduced risk for all-cause dementia and Parkinson’s disease compared with the oral antidiabetic drug group (aHR, 0.78) with a 6-month drug use lag period.
The association was observed regardless of SGLT2i exposure duration. Subgroup analyses indicated that the reductions in neurodegenerative disorders among those receiving SGLT2is were not associated with factors including age, sex, body mass index, blood pressure, glucose, lipid profiles, kidney function, health behaviors, comorbidities, diabetic complications, or other medication use.
Dr. Kim speculated that mechanisms underlying the reduced dementia risk could include SGLT2i effects of mitigating the common severe risk factors of type 2 diabetes and neurodegenerative diseases, including hypertension, heart failure, and chronic kidney disease, and improving hyperperfusion in the heart and cerebral vascular insufficiency.
Commenting on the study to this news organization, Erik H. Serné, MD, of the VU University Medical Centre, Amsterdam, the Netherlands, who comoderated the session, noted that “people with type 2 diabetes have a 50%-100% increased risk of developing dementia, particularly Alzheimer’s disease and vascular dementia.”
“The increasing prevalence of both conditions poses significant public health challenges, highlighting the need for effective prevention strategies and interventions.”
Currently, treatments for dementia are limited, with most primarily addressing symptoms and not the underlying cause of the neurodegenerative disease, he said.
He noted that, in addition to the effects mentioned by Dr. Kim, SGLT2is are also speculated to provide potential neuroprotective effects through improved glycemic control and insulin sensitivity, reduced inflammation and oxidative stress, enhanced mitochondrial function and energy metabolism, and reduced beta-amyloid and tau pathology.
“These mechanisms collectively may reduce the risk of cognitive decline, particularly in diabetic patients, and warrant further investigation in clinical trials to solidify the neuroprotective role of SGLT2 inhibitors,” said Dr. Serné.
In addition to their benefits in type 2 diabetes, SGLT2is “now offer hope in the prevention of dementia, a disease that has very limited therapeutic options thus far. The current data [presented by Dr. Kim] seem to corroborate this,” he added.
Dr. Kim and Dr. Serné had no disclosures to report.
A version of this article first appeared on Medscape.com.
MADRID — Patients with type 2 diabetes treated with sodium-glucose cotransporter 2 inhibitors (SGLT2is) show significant reductions in the risk of developing neurodegenerative disorders including Alzheimer’s disease, vascular dementia, and Parkinson’s disease, compared with those treated with other antidiabetic drugs, results from a large population-based cohort show.
“This was the largest nationwide population-based longitudinal cohort study to investigate the association between the use of SGLT2 inhibitors and the incidence of all-cause dementia and Parkinson’s disease,” said first author Hae Kyung Kim, MD, of the Department of Internal Medicine, Yonsei University College of Medicine, Seoul, South Korea, in presenting the findings at the annual meeting of the European Association for the Study of Diabetes.
Type 2 diabetes is known to increase the risk for neurodegenerative diseases such as dementia or Alzheimer’s disease, said Dr. Kim. Key factors attributed to the risk include shared pathophysiological mechanisms such as central nervous system insulin resistance and reduced cerebral glucose metabolism.
While research is lacking on the role of antidiabetic drugs in the treatment of neurodegenerative diseases, the researcher noted that “SGLT2 inhibitors, which have shown significant cardiorenal benefits and enhanced energy metabolism through ketogenesis, offer promise.”
To further investigate, Dr. Kim and her colleagues conducted the retrospective study, evaluating data on more than 1.3 million enrollees in Korea’s National Health Insurance Service Database who were aged 40 years or older, diagnosed with type 2 diabetes, and had initiated antidiabetic drugs between September 2014 and December 2019.
In the propensity score analysis, 358,862 patients were matched 1:1, in groups of 179,431 participants each, based on whether they were treated with SGLT2is or other oral antidiabetic drugs. Patients with a history of neurodegenerative disease, cancer, or use of glucagon-like peptide 1 receptor agonists were excluded.
The patients had a mean age of 57.8 years, 57.9% were men, and 6837 had incident dementia or Parkinson’s disease events reported.
With a mean follow-up of 2.88 years, after adjustment for key variables, those treated with SGLT2is had a 19% reduced risk of developing Alzheimer’s disease (adjusted hazard ratio [aHR], 0.81), a 31% reduced risk for vascular dementia (aHR, 0.69), and a 20% reduced risk for Parkinson’s disease (aHR, 0.80) compared with the non-SGLT2i group.
Furthermore, those receiving SGLT2i treatment had a 21% reduced risk for all-cause dementia (aHR, 0.79) and a 22% reduced risk for all-cause dementia and Parkinson’s disease compared with the oral antidiabetic drug group (aHR, 0.78) with a 6-month drug use lag period.
The association was observed regardless of SGLT2i exposure duration. Subgroup analyses indicated that the reductions in neurodegenerative disorders among those receiving SGLT2is were not associated with factors including age, sex, body mass index, blood pressure, glucose, lipid profiles, kidney function, health behaviors, comorbidities, diabetic complications, or other medication use.
Dr. Kim speculated that mechanisms underlying the reduced dementia risk could include SGLT2i effects of mitigating the common severe risk factors of type 2 diabetes and neurodegenerative diseases, including hypertension, heart failure, and chronic kidney disease, and improving hyperperfusion in the heart and cerebral vascular insufficiency.
Commenting on the study to this news organization, Erik H. Serné, MD, of the VU University Medical Centre, Amsterdam, the Netherlands, who comoderated the session, noted that “people with type 2 diabetes have a 50%-100% increased risk of developing dementia, particularly Alzheimer’s disease and vascular dementia.”
“The increasing prevalence of both conditions poses significant public health challenges, highlighting the need for effective prevention strategies and interventions.”
Currently, treatments for dementia are limited, with most primarily addressing symptoms and not the underlying cause of the neurodegenerative disease, he said.
He noted that, in addition to the effects mentioned by Dr. Kim, SGLT2is are also speculated to provide potential neuroprotective effects through improved glycemic control and insulin sensitivity, reduced inflammation and oxidative stress, enhanced mitochondrial function and energy metabolism, and reduced beta-amyloid and tau pathology.
“These mechanisms collectively may reduce the risk of cognitive decline, particularly in diabetic patients, and warrant further investigation in clinical trials to solidify the neuroprotective role of SGLT2 inhibitors,” said Dr. Serné.
In addition to their benefits in type 2 diabetes, SGLT2is “now offer hope in the prevention of dementia, a disease that has very limited therapeutic options thus far. The current data [presented by Dr. Kim] seem to corroborate this,” he added.
Dr. Kim and Dr. Serné had no disclosures to report.
A version of this article first appeared on Medscape.com.
MADRID — Patients with type 2 diabetes treated with sodium-glucose cotransporter 2 inhibitors (SGLT2is) show significant reductions in the risk of developing neurodegenerative disorders including Alzheimer’s disease, vascular dementia, and Parkinson’s disease, compared with those treated with other antidiabetic drugs, results from a large population-based cohort show.
“This was the largest nationwide population-based longitudinal cohort study to investigate the association between the use of SGLT2 inhibitors and the incidence of all-cause dementia and Parkinson’s disease,” said first author Hae Kyung Kim, MD, of the Department of Internal Medicine, Yonsei University College of Medicine, Seoul, South Korea, in presenting the findings at the annual meeting of the European Association for the Study of Diabetes.
Type 2 diabetes is known to increase the risk for neurodegenerative diseases such as dementia or Alzheimer’s disease, said Dr. Kim. Key factors attributed to the risk include shared pathophysiological mechanisms such as central nervous system insulin resistance and reduced cerebral glucose metabolism.
While research is lacking on the role of antidiabetic drugs in the treatment of neurodegenerative diseases, the researcher noted that “SGLT2 inhibitors, which have shown significant cardiorenal benefits and enhanced energy metabolism through ketogenesis, offer promise.”
To further investigate, Dr. Kim and her colleagues conducted the retrospective study, evaluating data on more than 1.3 million enrollees in Korea’s National Health Insurance Service Database who were aged 40 years or older, diagnosed with type 2 diabetes, and had initiated antidiabetic drugs between September 2014 and December 2019.
In the propensity score analysis, 358,862 patients were matched 1:1, in groups of 179,431 participants each, based on whether they were treated with SGLT2is or other oral antidiabetic drugs. Patients with a history of neurodegenerative disease, cancer, or use of glucagon-like peptide 1 receptor agonists were excluded.
The patients had a mean age of 57.8 years, 57.9% were men, and 6837 had incident dementia or Parkinson’s disease events reported.
With a mean follow-up of 2.88 years, after adjustment for key variables, those treated with SGLT2is had a 19% reduced risk of developing Alzheimer’s disease (adjusted hazard ratio [aHR], 0.81), a 31% reduced risk for vascular dementia (aHR, 0.69), and a 20% reduced risk for Parkinson’s disease (aHR, 0.80) compared with the non-SGLT2i group.
Furthermore, those receiving SGLT2i treatment had a 21% reduced risk for all-cause dementia (aHR, 0.79) and a 22% reduced risk for all-cause dementia and Parkinson’s disease compared with the oral antidiabetic drug group (aHR, 0.78) with a 6-month drug use lag period.
The association was observed regardless of SGLT2i exposure duration. Subgroup analyses indicated that the reductions in neurodegenerative disorders among those receiving SGLT2is were not associated with factors including age, sex, body mass index, blood pressure, glucose, lipid profiles, kidney function, health behaviors, comorbidities, diabetic complications, or other medication use.
Dr. Kim speculated that mechanisms underlying the reduced dementia risk could include SGLT2i effects of mitigating the common severe risk factors of type 2 diabetes and neurodegenerative diseases, including hypertension, heart failure, and chronic kidney disease, and improving hyperperfusion in the heart and cerebral vascular insufficiency.
Commenting on the study to this news organization, Erik H. Serné, MD, of the VU University Medical Centre, Amsterdam, the Netherlands, who comoderated the session, noted that “people with type 2 diabetes have a 50%-100% increased risk of developing dementia, particularly Alzheimer’s disease and vascular dementia.”
“The increasing prevalence of both conditions poses significant public health challenges, highlighting the need for effective prevention strategies and interventions.”
Currently, treatments for dementia are limited, with most primarily addressing symptoms and not the underlying cause of the neurodegenerative disease, he said.
He noted that, in addition to the effects mentioned by Dr. Kim, SGLT2is are also speculated to provide potential neuroprotective effects through improved glycemic control and insulin sensitivity, reduced inflammation and oxidative stress, enhanced mitochondrial function and energy metabolism, and reduced beta-amyloid and tau pathology.
“These mechanisms collectively may reduce the risk of cognitive decline, particularly in diabetic patients, and warrant further investigation in clinical trials to solidify the neuroprotective role of SGLT2 inhibitors,” said Dr. Serné.
In addition to their benefits in type 2 diabetes, SGLT2is “now offer hope in the prevention of dementia, a disease that has very limited therapeutic options thus far. The current data [presented by Dr. Kim] seem to corroborate this,” he added.
Dr. Kim and Dr. Serné had no disclosures to report.
A version of this article first appeared on Medscape.com.
FROM EASD 2024
‘Remarkable’ Weight Loss Seen With Safe, Tolerable Novel Oral Combination
MADRID — Amycretin, a dual-pathway, oral weight loss drug, led to up to 13% body weight loss in participants with overweight or obesity according to phase 1, first-in-human study data presented at the European Association for the Study of Diabetes (EASD) 2024 annual meeting.
Body weight loss was “remarkable for an orally delivered biologic,” said Agnes Gasiorek, PhD, senior clinical pharmacology specialist at Novo Nordisk, Måløv, Denmark, who presented the results. And “there was no plateauing of weight loss in the treatment period.”
With respect to the primary endpoint, stepwise dose escalation demonstrated that all tested dose levels up to and including 2 × 50 mg over a 12-week escalation period were safe and tolerable, Dr. Gasiorek reported.
The adverse events were in line with what was expected from targeting these receptors, and no new safety signals appeared during the study, she added.
Dual Pathways
Amycretin is a novel protein-based unimolecular amylin combined with a glucagon-like peptide 1 receptor agonist (GLP-1 RA) and is the first oral formulation of this combination under development.
The two components are both known to reduce appetite and energy intake and increase satiety, said Dr. Gasiorek, but amylin is considered to potentially increase leptin sensitivity and GLP-1 RAs are known to increase insulin secretion and biosynthesis. Together, the two components improve insulin sensitivity, decrease glucagon secretion, and lead to acute delay in gastric emptying.
The single-center, placebo-controlled, double-blind phase 1 study enrolled men and women aged 18-55 years (mean, 38-42 years across groups) with a body mass index of 25.0-39.9, without diabetes, and considered otherwise healthy.
Participants were randomly assigned to receive to receive oral amycretin (n = 95) or placebo (n = 29) once a day for up to 12 weeks. Study arms comprised single-ascending dosing (increasing from 1 mg/d to 25 mg), and multiple-ascending dosing. The latter consisted of multiple ascending doses (from 3 to 12 mg) over 10 days and multiple ascending doses (stepwise dose escalation, from 3 mg up to a final dose of 2 × 50 mg) over 12 weeks.
In her presentation at the EASD meeting, Dr. Gasiorek focused on results of the 12-week multiple ascending dose schedule with amycretin 50 mg (n = 16), amycretin 2 × 50 mg (n = 16), and placebo (n = 12).
The primary endpoint of the study was the number of treatment-emergent adverse events, while the area under the amycretin plasma concentration time curve and the maximum plasma concentration of amycretin were secondary endpoints.
The researchers also added percentage change in body weight after 12 weeks of treatment as an exploratory endpoint.
Safety Findings of Multiple Dosing
A total of 242 treatment-emergent adverse events were reported in the combined active and placebo groups and were of mild to moderate severity.
Treatment-emergent adverse events were found in 75% of the amycretin 50 mg group, 93.8% of the amycretin 2 × 50 mg group, and 33.3% of placebo recipients.
“Most adverse events reported were mild to moderate in severity and related to gastrointestinal discomfort (nausea and vomiting) and occurred in a dose-proportional manner,” reported Dr. Gasiorek.
Gastrointestinal events were experienced by 50%, 87.5%, and 16.7% of participants receiving amycretin 50 mg, amycretin 2 × 50 mg, and placebo, respectively (112 in total).
Decreased appetite was also found in 56.3%, 81.3%, and 16.7% of the amycretin 50 mg, amycretin 2 × 50 mg, and placebo groups, respectively.
Two serious adverse events occurred, one of which was acute cholecystitis and the other diabetic ketoacidosis; “however, the [latter] participant was found to have autoantibodies for beta cells before treatment and was later diagnosed with type 1 diabetes,” Dr. Gasiorek said.
Body Weight Reduction
Participants on 50 mg amycretin lost an average of 10.4% of their body weight (estimated treatment difference vs placebo, –9.2; 95% CI, –12.0 to –6.5), whereas those on 2 × 50 mg amycretin lost 13.1% of their body weight (estimated treatment difference vs placebo, –11.8; 95% CI, –14.6 to –9.0). Placebo group participants lost 1.2% of their body weight over the 12 weeks.
Although no plateauing of weight loss was seen, said Dr. Gasiorek, it is important to consider the relatively short treatment duration and the limited time on the final dose, which could potentially introduce bias.
To date, weight loss medications based on GLP-1 RA technology are injectables. A combination of the injectable amylin analogue cagrilintide and the GLP-1 RA semaglutide is also being explored as a subcutaneous treatment solution.
In a comment, Martin Holst Lange, MD, PhD, executive vice president of development at Novo Nordisk, said that “amycretin is the first treatment to harness the two distinct biological pathways stimulated by amylin and GLP-1 in a single molecule.”
The safety and tolerability profiles and the magnitude of weight loss support further development of amycretin in patients with overweight or obesity, said Dr. Lange, who noted that the company was awaiting data from the ongoing phase 1 trial with subcutaneous amycretin, expected in 2025.
Having heard the presentation, co-moderator Timo Müller, PhD, professor at Ludwig Maximilian University of Munich, Germany, gave a considered response. “The drug was relatively well tolerated, with the typical GLP-1–induced GI [gastrointestinal] adverse effects being the most frequently reported.”
But he pointed out that questions remain. “We still need to know whether, at the given dose, the drug outperforms best-in-class drugs like semaglutide or tirzepatide at the highest approved doses. Furthermore, it warrants clarification if and to what extent the activation of the amylin receptor contributes to the shown effect and if and to what extent the glycemic benefits result from activation of the glucagon receptor (amylin improved glycemia by decreasing the secretion of glucagon). In any way, the current data remain friendly and support phase 2 development.”
Oral Meds Could Bring Down Cost
Commenting on the data, Nerys Astbury, PhD, associate professor of diet and obesity at Nuffield Department of Primary Health Care Sciences, University of Oxford, England, said, “It is important to note that whilst the participants in this trial did lose weight over the 12-week study — and this was statistically more weight than in the placebo group — this study was not designed or powered to detect differences in body weight over longer periods of time.”
If the results are confirmed in future studies, amycretin might widen the treatment options and introduce competition, probably bringing down the costs in the longer-term, said Dr. Astbury, who welcomes the prospect.
“It is possible that some people might find the oral medications more acceptable than the injectable GLP-1 agonists currently available,” she said. And the current options are expensive, “which raises challenges to a taxpayer-funded health system like the NHS [National Health Service].”
“Furthermore, if the growing number of oral obesity medications prove safe, tolerable, and effective ... they are likely to significantly reduce the risks of developing many complications of obesity.”
Naveed Sattar, MD, professor of cardiometabolic medicine and honorary consultant, University of Glasgow, Scotland, agreed. “The more medicines coming forward to treat obesity, the better,” he said. In particular, oral medications would be more easily available, and cheaper, “for the many millions around the world struggling with obesity and its complications.”
Dr. Gasiorek declares she is an employee of and a shareholder in Novo Nordisk. Dr. Astbury declares no financial disclosures. Dr. Sattar declares having consulted for several companies that make diabetes medicines but also contributed to several lifestyle trials. For Novo Nordisk, he has consulted for the company on advisory boards, but not on any of their weight loss drug trial committees, and he is on the steering committee for the ZEUS trial, which is not a weight loss trial product but an anti-inflammatory. He does not have any shares for any product in health etc. He declares consulting fees and/or speaker honoraria from Abbott Laboratories, Afimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceuticals, Janssen, Merck Sharp & Dohme, Novartis, Novo Nordisk, Pfizer, and Sanofi, and grant support paid to his university from AstraZeneca, Boehringer Ingelheim, Novartis, and Roche Diagnostics. Dr. Müller received financial support or an honorarium from Novo Nordisk, Merck, Eli Lilly, Boehringer Ingelheim, and Mercodia; he further holds stocks at Novo Nordisk and Eli Lilly and is cofounder of Bluewater Biosciences.
A version of this article first appeared on Medscape.com.
MADRID — Amycretin, a dual-pathway, oral weight loss drug, led to up to 13% body weight loss in participants with overweight or obesity according to phase 1, first-in-human study data presented at the European Association for the Study of Diabetes (EASD) 2024 annual meeting.
Body weight loss was “remarkable for an orally delivered biologic,” said Agnes Gasiorek, PhD, senior clinical pharmacology specialist at Novo Nordisk, Måløv, Denmark, who presented the results. And “there was no plateauing of weight loss in the treatment period.”
With respect to the primary endpoint, stepwise dose escalation demonstrated that all tested dose levels up to and including 2 × 50 mg over a 12-week escalation period were safe and tolerable, Dr. Gasiorek reported.
The adverse events were in line with what was expected from targeting these receptors, and no new safety signals appeared during the study, she added.
Dual Pathways
Amycretin is a novel protein-based unimolecular amylin combined with a glucagon-like peptide 1 receptor agonist (GLP-1 RA) and is the first oral formulation of this combination under development.
The two components are both known to reduce appetite and energy intake and increase satiety, said Dr. Gasiorek, but amylin is considered to potentially increase leptin sensitivity and GLP-1 RAs are known to increase insulin secretion and biosynthesis. Together, the two components improve insulin sensitivity, decrease glucagon secretion, and lead to acute delay in gastric emptying.
The single-center, placebo-controlled, double-blind phase 1 study enrolled men and women aged 18-55 years (mean, 38-42 years across groups) with a body mass index of 25.0-39.9, without diabetes, and considered otherwise healthy.
Participants were randomly assigned to receive to receive oral amycretin (n = 95) or placebo (n = 29) once a day for up to 12 weeks. Study arms comprised single-ascending dosing (increasing from 1 mg/d to 25 mg), and multiple-ascending dosing. The latter consisted of multiple ascending doses (from 3 to 12 mg) over 10 days and multiple ascending doses (stepwise dose escalation, from 3 mg up to a final dose of 2 × 50 mg) over 12 weeks.
In her presentation at the EASD meeting, Dr. Gasiorek focused on results of the 12-week multiple ascending dose schedule with amycretin 50 mg (n = 16), amycretin 2 × 50 mg (n = 16), and placebo (n = 12).
The primary endpoint of the study was the number of treatment-emergent adverse events, while the area under the amycretin plasma concentration time curve and the maximum plasma concentration of amycretin were secondary endpoints.
The researchers also added percentage change in body weight after 12 weeks of treatment as an exploratory endpoint.
Safety Findings of Multiple Dosing
A total of 242 treatment-emergent adverse events were reported in the combined active and placebo groups and were of mild to moderate severity.
Treatment-emergent adverse events were found in 75% of the amycretin 50 mg group, 93.8% of the amycretin 2 × 50 mg group, and 33.3% of placebo recipients.
“Most adverse events reported were mild to moderate in severity and related to gastrointestinal discomfort (nausea and vomiting) and occurred in a dose-proportional manner,” reported Dr. Gasiorek.
Gastrointestinal events were experienced by 50%, 87.5%, and 16.7% of participants receiving amycretin 50 mg, amycretin 2 × 50 mg, and placebo, respectively (112 in total).
Decreased appetite was also found in 56.3%, 81.3%, and 16.7% of the amycretin 50 mg, amycretin 2 × 50 mg, and placebo groups, respectively.
Two serious adverse events occurred, one of which was acute cholecystitis and the other diabetic ketoacidosis; “however, the [latter] participant was found to have autoantibodies for beta cells before treatment and was later diagnosed with type 1 diabetes,” Dr. Gasiorek said.
Body Weight Reduction
Participants on 50 mg amycretin lost an average of 10.4% of their body weight (estimated treatment difference vs placebo, –9.2; 95% CI, –12.0 to –6.5), whereas those on 2 × 50 mg amycretin lost 13.1% of their body weight (estimated treatment difference vs placebo, –11.8; 95% CI, –14.6 to –9.0). Placebo group participants lost 1.2% of their body weight over the 12 weeks.
Although no plateauing of weight loss was seen, said Dr. Gasiorek, it is important to consider the relatively short treatment duration and the limited time on the final dose, which could potentially introduce bias.
To date, weight loss medications based on GLP-1 RA technology are injectables. A combination of the injectable amylin analogue cagrilintide and the GLP-1 RA semaglutide is also being explored as a subcutaneous treatment solution.
In a comment, Martin Holst Lange, MD, PhD, executive vice president of development at Novo Nordisk, said that “amycretin is the first treatment to harness the two distinct biological pathways stimulated by amylin and GLP-1 in a single molecule.”
The safety and tolerability profiles and the magnitude of weight loss support further development of amycretin in patients with overweight or obesity, said Dr. Lange, who noted that the company was awaiting data from the ongoing phase 1 trial with subcutaneous amycretin, expected in 2025.
Having heard the presentation, co-moderator Timo Müller, PhD, professor at Ludwig Maximilian University of Munich, Germany, gave a considered response. “The drug was relatively well tolerated, with the typical GLP-1–induced GI [gastrointestinal] adverse effects being the most frequently reported.”
But he pointed out that questions remain. “We still need to know whether, at the given dose, the drug outperforms best-in-class drugs like semaglutide or tirzepatide at the highest approved doses. Furthermore, it warrants clarification if and to what extent the activation of the amylin receptor contributes to the shown effect and if and to what extent the glycemic benefits result from activation of the glucagon receptor (amylin improved glycemia by decreasing the secretion of glucagon). In any way, the current data remain friendly and support phase 2 development.”
Oral Meds Could Bring Down Cost
Commenting on the data, Nerys Astbury, PhD, associate professor of diet and obesity at Nuffield Department of Primary Health Care Sciences, University of Oxford, England, said, “It is important to note that whilst the participants in this trial did lose weight over the 12-week study — and this was statistically more weight than in the placebo group — this study was not designed or powered to detect differences in body weight over longer periods of time.”
If the results are confirmed in future studies, amycretin might widen the treatment options and introduce competition, probably bringing down the costs in the longer-term, said Dr. Astbury, who welcomes the prospect.
“It is possible that some people might find the oral medications more acceptable than the injectable GLP-1 agonists currently available,” she said. And the current options are expensive, “which raises challenges to a taxpayer-funded health system like the NHS [National Health Service].”
“Furthermore, if the growing number of oral obesity medications prove safe, tolerable, and effective ... they are likely to significantly reduce the risks of developing many complications of obesity.”
Naveed Sattar, MD, professor of cardiometabolic medicine and honorary consultant, University of Glasgow, Scotland, agreed. “The more medicines coming forward to treat obesity, the better,” he said. In particular, oral medications would be more easily available, and cheaper, “for the many millions around the world struggling with obesity and its complications.”
Dr. Gasiorek declares she is an employee of and a shareholder in Novo Nordisk. Dr. Astbury declares no financial disclosures. Dr. Sattar declares having consulted for several companies that make diabetes medicines but also contributed to several lifestyle trials. For Novo Nordisk, he has consulted for the company on advisory boards, but not on any of their weight loss drug trial committees, and he is on the steering committee for the ZEUS trial, which is not a weight loss trial product but an anti-inflammatory. He does not have any shares for any product in health etc. He declares consulting fees and/or speaker honoraria from Abbott Laboratories, Afimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceuticals, Janssen, Merck Sharp & Dohme, Novartis, Novo Nordisk, Pfizer, and Sanofi, and grant support paid to his university from AstraZeneca, Boehringer Ingelheim, Novartis, and Roche Diagnostics. Dr. Müller received financial support or an honorarium from Novo Nordisk, Merck, Eli Lilly, Boehringer Ingelheim, and Mercodia; he further holds stocks at Novo Nordisk and Eli Lilly and is cofounder of Bluewater Biosciences.
A version of this article first appeared on Medscape.com.
MADRID — Amycretin, a dual-pathway, oral weight loss drug, led to up to 13% body weight loss in participants with overweight or obesity according to phase 1, first-in-human study data presented at the European Association for the Study of Diabetes (EASD) 2024 annual meeting.
Body weight loss was “remarkable for an orally delivered biologic,” said Agnes Gasiorek, PhD, senior clinical pharmacology specialist at Novo Nordisk, Måløv, Denmark, who presented the results. And “there was no plateauing of weight loss in the treatment period.”
With respect to the primary endpoint, stepwise dose escalation demonstrated that all tested dose levels up to and including 2 × 50 mg over a 12-week escalation period were safe and tolerable, Dr. Gasiorek reported.
The adverse events were in line with what was expected from targeting these receptors, and no new safety signals appeared during the study, she added.
Dual Pathways
Amycretin is a novel protein-based unimolecular amylin combined with a glucagon-like peptide 1 receptor agonist (GLP-1 RA) and is the first oral formulation of this combination under development.
The two components are both known to reduce appetite and energy intake and increase satiety, said Dr. Gasiorek, but amylin is considered to potentially increase leptin sensitivity and GLP-1 RAs are known to increase insulin secretion and biosynthesis. Together, the two components improve insulin sensitivity, decrease glucagon secretion, and lead to acute delay in gastric emptying.
The single-center, placebo-controlled, double-blind phase 1 study enrolled men and women aged 18-55 years (mean, 38-42 years across groups) with a body mass index of 25.0-39.9, without diabetes, and considered otherwise healthy.
Participants were randomly assigned to receive to receive oral amycretin (n = 95) or placebo (n = 29) once a day for up to 12 weeks. Study arms comprised single-ascending dosing (increasing from 1 mg/d to 25 mg), and multiple-ascending dosing. The latter consisted of multiple ascending doses (from 3 to 12 mg) over 10 days and multiple ascending doses (stepwise dose escalation, from 3 mg up to a final dose of 2 × 50 mg) over 12 weeks.
In her presentation at the EASD meeting, Dr. Gasiorek focused on results of the 12-week multiple ascending dose schedule with amycretin 50 mg (n = 16), amycretin 2 × 50 mg (n = 16), and placebo (n = 12).
The primary endpoint of the study was the number of treatment-emergent adverse events, while the area under the amycretin plasma concentration time curve and the maximum plasma concentration of amycretin were secondary endpoints.
The researchers also added percentage change in body weight after 12 weeks of treatment as an exploratory endpoint.
Safety Findings of Multiple Dosing
A total of 242 treatment-emergent adverse events were reported in the combined active and placebo groups and were of mild to moderate severity.
Treatment-emergent adverse events were found in 75% of the amycretin 50 mg group, 93.8% of the amycretin 2 × 50 mg group, and 33.3% of placebo recipients.
“Most adverse events reported were mild to moderate in severity and related to gastrointestinal discomfort (nausea and vomiting) and occurred in a dose-proportional manner,” reported Dr. Gasiorek.
Gastrointestinal events were experienced by 50%, 87.5%, and 16.7% of participants receiving amycretin 50 mg, amycretin 2 × 50 mg, and placebo, respectively (112 in total).
Decreased appetite was also found in 56.3%, 81.3%, and 16.7% of the amycretin 50 mg, amycretin 2 × 50 mg, and placebo groups, respectively.
Two serious adverse events occurred, one of which was acute cholecystitis and the other diabetic ketoacidosis; “however, the [latter] participant was found to have autoantibodies for beta cells before treatment and was later diagnosed with type 1 diabetes,” Dr. Gasiorek said.
Body Weight Reduction
Participants on 50 mg amycretin lost an average of 10.4% of their body weight (estimated treatment difference vs placebo, –9.2; 95% CI, –12.0 to –6.5), whereas those on 2 × 50 mg amycretin lost 13.1% of their body weight (estimated treatment difference vs placebo, –11.8; 95% CI, –14.6 to –9.0). Placebo group participants lost 1.2% of their body weight over the 12 weeks.
Although no plateauing of weight loss was seen, said Dr. Gasiorek, it is important to consider the relatively short treatment duration and the limited time on the final dose, which could potentially introduce bias.
To date, weight loss medications based on GLP-1 RA technology are injectables. A combination of the injectable amylin analogue cagrilintide and the GLP-1 RA semaglutide is also being explored as a subcutaneous treatment solution.
In a comment, Martin Holst Lange, MD, PhD, executive vice president of development at Novo Nordisk, said that “amycretin is the first treatment to harness the two distinct biological pathways stimulated by amylin and GLP-1 in a single molecule.”
The safety and tolerability profiles and the magnitude of weight loss support further development of amycretin in patients with overweight or obesity, said Dr. Lange, who noted that the company was awaiting data from the ongoing phase 1 trial with subcutaneous amycretin, expected in 2025.
Having heard the presentation, co-moderator Timo Müller, PhD, professor at Ludwig Maximilian University of Munich, Germany, gave a considered response. “The drug was relatively well tolerated, with the typical GLP-1–induced GI [gastrointestinal] adverse effects being the most frequently reported.”
But he pointed out that questions remain. “We still need to know whether, at the given dose, the drug outperforms best-in-class drugs like semaglutide or tirzepatide at the highest approved doses. Furthermore, it warrants clarification if and to what extent the activation of the amylin receptor contributes to the shown effect and if and to what extent the glycemic benefits result from activation of the glucagon receptor (amylin improved glycemia by decreasing the secretion of glucagon). In any way, the current data remain friendly and support phase 2 development.”
Oral Meds Could Bring Down Cost
Commenting on the data, Nerys Astbury, PhD, associate professor of diet and obesity at Nuffield Department of Primary Health Care Sciences, University of Oxford, England, said, “It is important to note that whilst the participants in this trial did lose weight over the 12-week study — and this was statistically more weight than in the placebo group — this study was not designed or powered to detect differences in body weight over longer periods of time.”
If the results are confirmed in future studies, amycretin might widen the treatment options and introduce competition, probably bringing down the costs in the longer-term, said Dr. Astbury, who welcomes the prospect.
“It is possible that some people might find the oral medications more acceptable than the injectable GLP-1 agonists currently available,” she said. And the current options are expensive, “which raises challenges to a taxpayer-funded health system like the NHS [National Health Service].”
“Furthermore, if the growing number of oral obesity medications prove safe, tolerable, and effective ... they are likely to significantly reduce the risks of developing many complications of obesity.”
Naveed Sattar, MD, professor of cardiometabolic medicine and honorary consultant, University of Glasgow, Scotland, agreed. “The more medicines coming forward to treat obesity, the better,” he said. In particular, oral medications would be more easily available, and cheaper, “for the many millions around the world struggling with obesity and its complications.”
Dr. Gasiorek declares she is an employee of and a shareholder in Novo Nordisk. Dr. Astbury declares no financial disclosures. Dr. Sattar declares having consulted for several companies that make diabetes medicines but also contributed to several lifestyle trials. For Novo Nordisk, he has consulted for the company on advisory boards, but not on any of their weight loss drug trial committees, and he is on the steering committee for the ZEUS trial, which is not a weight loss trial product but an anti-inflammatory. He does not have any shares for any product in health etc. He declares consulting fees and/or speaker honoraria from Abbott Laboratories, Afimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceuticals, Janssen, Merck Sharp & Dohme, Novartis, Novo Nordisk, Pfizer, and Sanofi, and grant support paid to his university from AstraZeneca, Boehringer Ingelheim, Novartis, and Roche Diagnostics. Dr. Müller received financial support or an honorarium from Novo Nordisk, Merck, Eli Lilly, Boehringer Ingelheim, and Mercodia; he further holds stocks at Novo Nordisk and Eli Lilly and is cofounder of Bluewater Biosciences.
A version of this article first appeared on Medscape.com.
FROM EASD 2024
Genitourinary Symptoms in Men: Canaries in the Coal Mine for Underlying Chronic Disease
At age 57, a senior scientific researcher in Santa Barbara, California, complained of chronic erectile dysfunction (ED) in what had been a sexually active marriage. “I just couldn’t get an erection, let alone sustain one. Apart from that, I maybe felt a bit tired but generally okay,” he said. Though seemingly well otherwise, 18 months later he was dead of a hereditary right-sided colon cancer.
While not all cases of ED are associated with a dire outcome, the genitourinary signals of ED and lower urinary tract symptoms (LUTS), especially nocturia, serve as sentinel indicators of the presence of, or risk factors for, serious chronic conditions. These commonly include cardiovascular disease (CVD), diabetes, and metabolic syndrome and are associated with obesity, depression, and obstructive sleep apnea.
Sometimes these serious conditions may stay under the radar until men seek help for ED or LUTS.
“We know that among men who had a heart attack, 50% had some degree of ED within 3 years of their cardiac event,” Sam Tafari, MBBS, of the Endocrine and Metabolic Unit at Royal Adelaide Hospital in Adelaide, South Australia, said in an interview.
That’s the bad news. The good news is that these two problems may specifically incentivize men to seek timely care for serious conditions they might otherwise not get, according to Dr. Tafari. And primary care doctors are ideally positioned to get men early multifaceted care. He recently coauthored a call to action on this issue in a review appearing in the Journal of Men’s Health.
In Dr. Tafari’s experience, most patients seeking urological care are unaware of the multiple conditions linked to ED and LUTS. “Many consider these to be due to issues like low testosterone, which actually make up a very small proportion of cases of ED,” he said. Aging, obesity, inactivity, smoking, alcohol abuse, and prescription and street drugs can also contribute to the development of ED.
In most affected men, ED is of vascular etiology, with endothelial dysfunction of the inner lining of blood vessels and smooth muscle the common denominator.
This dysfunction causes inadequate blood supply to both the coronary and the penile arteries, so ED and CVD are considered different manifestations of the same systemic disorder. Because the tumescence-controlling cavernosal vessels of the penis are considerably smaller, the same level of arteriopathy causes a more severe reduction in blood in the erectile tissue. As a result, ED often precedes CVD and presents an early opportunity to screen men for CVD.
As to the mechanisms behind LUTS, Peter N. Tsambarlis, MD, a urologist at Northwestern Medicine in Chicago, subscribes to the inflammation theory. “Suboptimal health issues such as high [blood] pressure, blood lipids, and blood glucose lead to chronic widespread inflammation, which makes the bladder less flexible as a storage vessel,” he explained. “It’s not able to stretch adequately overnight to hold the urine until morning.”
Ask Early, Ask Often
Jeffrey P. Weiss, MD, PhD, chair of the Department of Urology at SUNY Downstate Health Sciences University in Brooklyn, New York, has done research that uncovered a relationship between structural cardiac disease and nocturia. “So if you had to ask a patient a single question that would point to a global health issue, it would be ‘Do you have frequent nighttime urination,’ ” he said.
It’s never too soon to ask men about these symptoms, said Dr. Tsambarlis. The best time to raise issues of ED and LUTS is when a man enters primary care — regardless of age or absence of symptoms. “That way you have a baseline and can watch for changes and do early intervention as needed. Men don’t usually want to bring up sexual dysfunction or urinary health, but asking doesn’t need to dominate the visit,” he said.
Dr. Tafari recommends that primary care physicians adopt a targeted approach using ED and nocturia as entry points for engaging men in their healthcare. While acknowledging that primary care physicians have an ever-growing checklist of questions to ask patients and hardly need one more thing to screen for, he suggests asking two quick, and easy “before you go” genitourinary queries:
- Are you having trouble with erections or having sex?
- Are you getting up at night to pass urine more than once?
“The men really appreciate being asked,” he said. “But what worries me is all the men we don’t see who have these symptoms but don’t know they’re important, and no one is asking about them.”
Gideon Richards, MD, a urologist at the Northwell Health Physician Partners Smith Institute for Urology at Garden City, and director of Men’s Health, Central Region, for Northwell Health in New Hyde Park, both in New York, said erectile problems should not wait for specialty care. By the time men with ED are referred to urology, they may already have failed treatment with first-line phosphodiesterase 5 inhibitor therapy, he said. “A significant proportion will have arteriogenic erectile dysfunction, a measurable decrease in the amount of blood flow into the erectile bodies.”
Addressing the Issue
Addressing genitourinary-signaled issues has the double benefit of easing ED and LUTS and improving men’s health and longevity and may help narrow the worldwide gender gap in life expectancy. As a recent global analysis found, there’s a 5-year longevity disparity favoring women over men. Biology aside, men do not access healthcare as often as women, who consult their general practitioners regularly throughout their lifespan for multiple reasons, including reproductive care, and more screening programs are aimed at women.
Added Dr. Tsambarlis, “Men should know that losing weight and switching to a healthy lifestyle can improve sexual function about half as much as phosphodiesterase 5 inhibitors such as sildenafil [Viagra] or tadalafil [Cialis].”
“Many, however, would prefer just to take drugs rather than change their lifestyle and lose weight. There are certainly effective options available, but these are not uniformly effective,” said Dr. Weiss.
Dr. Tafari’s group is designing a short, simple, culturally acceptable screening tool for use in primary care practice and will monitor its impact on physician prescribing habits and overall men’s health outcomes.
Dr. Tafari received funding from the Hospital Research Foundation and Freemasons Centre for Male Health and Wellbeing in Adelaide, South Australia. Dr. Tafari, Dr. Tsambarlis, Dr. Weiss, and Dr. Richards had no relevant conflicts of interest to declare.
A version of this article appeared on Medscape.com.
At age 57, a senior scientific researcher in Santa Barbara, California, complained of chronic erectile dysfunction (ED) in what had been a sexually active marriage. “I just couldn’t get an erection, let alone sustain one. Apart from that, I maybe felt a bit tired but generally okay,” he said. Though seemingly well otherwise, 18 months later he was dead of a hereditary right-sided colon cancer.
While not all cases of ED are associated with a dire outcome, the genitourinary signals of ED and lower urinary tract symptoms (LUTS), especially nocturia, serve as sentinel indicators of the presence of, or risk factors for, serious chronic conditions. These commonly include cardiovascular disease (CVD), diabetes, and metabolic syndrome and are associated with obesity, depression, and obstructive sleep apnea.
Sometimes these serious conditions may stay under the radar until men seek help for ED or LUTS.
“We know that among men who had a heart attack, 50% had some degree of ED within 3 years of their cardiac event,” Sam Tafari, MBBS, of the Endocrine and Metabolic Unit at Royal Adelaide Hospital in Adelaide, South Australia, said in an interview.
That’s the bad news. The good news is that these two problems may specifically incentivize men to seek timely care for serious conditions they might otherwise not get, according to Dr. Tafari. And primary care doctors are ideally positioned to get men early multifaceted care. He recently coauthored a call to action on this issue in a review appearing in the Journal of Men’s Health.
In Dr. Tafari’s experience, most patients seeking urological care are unaware of the multiple conditions linked to ED and LUTS. “Many consider these to be due to issues like low testosterone, which actually make up a very small proportion of cases of ED,” he said. Aging, obesity, inactivity, smoking, alcohol abuse, and prescription and street drugs can also contribute to the development of ED.
In most affected men, ED is of vascular etiology, with endothelial dysfunction of the inner lining of blood vessels and smooth muscle the common denominator.
This dysfunction causes inadequate blood supply to both the coronary and the penile arteries, so ED and CVD are considered different manifestations of the same systemic disorder. Because the tumescence-controlling cavernosal vessels of the penis are considerably smaller, the same level of arteriopathy causes a more severe reduction in blood in the erectile tissue. As a result, ED often precedes CVD and presents an early opportunity to screen men for CVD.
As to the mechanisms behind LUTS, Peter N. Tsambarlis, MD, a urologist at Northwestern Medicine in Chicago, subscribes to the inflammation theory. “Suboptimal health issues such as high [blood] pressure, blood lipids, and blood glucose lead to chronic widespread inflammation, which makes the bladder less flexible as a storage vessel,” he explained. “It’s not able to stretch adequately overnight to hold the urine until morning.”
Ask Early, Ask Often
Jeffrey P. Weiss, MD, PhD, chair of the Department of Urology at SUNY Downstate Health Sciences University in Brooklyn, New York, has done research that uncovered a relationship between structural cardiac disease and nocturia. “So if you had to ask a patient a single question that would point to a global health issue, it would be ‘Do you have frequent nighttime urination,’ ” he said.
It’s never too soon to ask men about these symptoms, said Dr. Tsambarlis. The best time to raise issues of ED and LUTS is when a man enters primary care — regardless of age or absence of symptoms. “That way you have a baseline and can watch for changes and do early intervention as needed. Men don’t usually want to bring up sexual dysfunction or urinary health, but asking doesn’t need to dominate the visit,” he said.
Dr. Tafari recommends that primary care physicians adopt a targeted approach using ED and nocturia as entry points for engaging men in their healthcare. While acknowledging that primary care physicians have an ever-growing checklist of questions to ask patients and hardly need one more thing to screen for, he suggests asking two quick, and easy “before you go” genitourinary queries:
- Are you having trouble with erections or having sex?
- Are you getting up at night to pass urine more than once?
“The men really appreciate being asked,” he said. “But what worries me is all the men we don’t see who have these symptoms but don’t know they’re important, and no one is asking about them.”
Gideon Richards, MD, a urologist at the Northwell Health Physician Partners Smith Institute for Urology at Garden City, and director of Men’s Health, Central Region, for Northwell Health in New Hyde Park, both in New York, said erectile problems should not wait for specialty care. By the time men with ED are referred to urology, they may already have failed treatment with first-line phosphodiesterase 5 inhibitor therapy, he said. “A significant proportion will have arteriogenic erectile dysfunction, a measurable decrease in the amount of blood flow into the erectile bodies.”
Addressing the Issue
Addressing genitourinary-signaled issues has the double benefit of easing ED and LUTS and improving men’s health and longevity and may help narrow the worldwide gender gap in life expectancy. As a recent global analysis found, there’s a 5-year longevity disparity favoring women over men. Biology aside, men do not access healthcare as often as women, who consult their general practitioners regularly throughout their lifespan for multiple reasons, including reproductive care, and more screening programs are aimed at women.
Added Dr. Tsambarlis, “Men should know that losing weight and switching to a healthy lifestyle can improve sexual function about half as much as phosphodiesterase 5 inhibitors such as sildenafil [Viagra] or tadalafil [Cialis].”
“Many, however, would prefer just to take drugs rather than change their lifestyle and lose weight. There are certainly effective options available, but these are not uniformly effective,” said Dr. Weiss.
Dr. Tafari’s group is designing a short, simple, culturally acceptable screening tool for use in primary care practice and will monitor its impact on physician prescribing habits and overall men’s health outcomes.
Dr. Tafari received funding from the Hospital Research Foundation and Freemasons Centre for Male Health and Wellbeing in Adelaide, South Australia. Dr. Tafari, Dr. Tsambarlis, Dr. Weiss, and Dr. Richards had no relevant conflicts of interest to declare.
A version of this article appeared on Medscape.com.
At age 57, a senior scientific researcher in Santa Barbara, California, complained of chronic erectile dysfunction (ED) in what had been a sexually active marriage. “I just couldn’t get an erection, let alone sustain one. Apart from that, I maybe felt a bit tired but generally okay,” he said. Though seemingly well otherwise, 18 months later he was dead of a hereditary right-sided colon cancer.
While not all cases of ED are associated with a dire outcome, the genitourinary signals of ED and lower urinary tract symptoms (LUTS), especially nocturia, serve as sentinel indicators of the presence of, or risk factors for, serious chronic conditions. These commonly include cardiovascular disease (CVD), diabetes, and metabolic syndrome and are associated with obesity, depression, and obstructive sleep apnea.
Sometimes these serious conditions may stay under the radar until men seek help for ED or LUTS.
“We know that among men who had a heart attack, 50% had some degree of ED within 3 years of their cardiac event,” Sam Tafari, MBBS, of the Endocrine and Metabolic Unit at Royal Adelaide Hospital in Adelaide, South Australia, said in an interview.
That’s the bad news. The good news is that these two problems may specifically incentivize men to seek timely care for serious conditions they might otherwise not get, according to Dr. Tafari. And primary care doctors are ideally positioned to get men early multifaceted care. He recently coauthored a call to action on this issue in a review appearing in the Journal of Men’s Health.
In Dr. Tafari’s experience, most patients seeking urological care are unaware of the multiple conditions linked to ED and LUTS. “Many consider these to be due to issues like low testosterone, which actually make up a very small proportion of cases of ED,” he said. Aging, obesity, inactivity, smoking, alcohol abuse, and prescription and street drugs can also contribute to the development of ED.
In most affected men, ED is of vascular etiology, with endothelial dysfunction of the inner lining of blood vessels and smooth muscle the common denominator.
This dysfunction causes inadequate blood supply to both the coronary and the penile arteries, so ED and CVD are considered different manifestations of the same systemic disorder. Because the tumescence-controlling cavernosal vessels of the penis are considerably smaller, the same level of arteriopathy causes a more severe reduction in blood in the erectile tissue. As a result, ED often precedes CVD and presents an early opportunity to screen men for CVD.
As to the mechanisms behind LUTS, Peter N. Tsambarlis, MD, a urologist at Northwestern Medicine in Chicago, subscribes to the inflammation theory. “Suboptimal health issues such as high [blood] pressure, blood lipids, and blood glucose lead to chronic widespread inflammation, which makes the bladder less flexible as a storage vessel,” he explained. “It’s not able to stretch adequately overnight to hold the urine until morning.”
Ask Early, Ask Often
Jeffrey P. Weiss, MD, PhD, chair of the Department of Urology at SUNY Downstate Health Sciences University in Brooklyn, New York, has done research that uncovered a relationship between structural cardiac disease and nocturia. “So if you had to ask a patient a single question that would point to a global health issue, it would be ‘Do you have frequent nighttime urination,’ ” he said.
It’s never too soon to ask men about these symptoms, said Dr. Tsambarlis. The best time to raise issues of ED and LUTS is when a man enters primary care — regardless of age or absence of symptoms. “That way you have a baseline and can watch for changes and do early intervention as needed. Men don’t usually want to bring up sexual dysfunction or urinary health, but asking doesn’t need to dominate the visit,” he said.
Dr. Tafari recommends that primary care physicians adopt a targeted approach using ED and nocturia as entry points for engaging men in their healthcare. While acknowledging that primary care physicians have an ever-growing checklist of questions to ask patients and hardly need one more thing to screen for, he suggests asking two quick, and easy “before you go” genitourinary queries:
- Are you having trouble with erections or having sex?
- Are you getting up at night to pass urine more than once?
“The men really appreciate being asked,” he said. “But what worries me is all the men we don’t see who have these symptoms but don’t know they’re important, and no one is asking about them.”
Gideon Richards, MD, a urologist at the Northwell Health Physician Partners Smith Institute for Urology at Garden City, and director of Men’s Health, Central Region, for Northwell Health in New Hyde Park, both in New York, said erectile problems should not wait for specialty care. By the time men with ED are referred to urology, they may already have failed treatment with first-line phosphodiesterase 5 inhibitor therapy, he said. “A significant proportion will have arteriogenic erectile dysfunction, a measurable decrease in the amount of blood flow into the erectile bodies.”
Addressing the Issue
Addressing genitourinary-signaled issues has the double benefit of easing ED and LUTS and improving men’s health and longevity and may help narrow the worldwide gender gap in life expectancy. As a recent global analysis found, there’s a 5-year longevity disparity favoring women over men. Biology aside, men do not access healthcare as often as women, who consult their general practitioners regularly throughout their lifespan for multiple reasons, including reproductive care, and more screening programs are aimed at women.
Added Dr. Tsambarlis, “Men should know that losing weight and switching to a healthy lifestyle can improve sexual function about half as much as phosphodiesterase 5 inhibitors such as sildenafil [Viagra] or tadalafil [Cialis].”
“Many, however, would prefer just to take drugs rather than change their lifestyle and lose weight. There are certainly effective options available, but these are not uniformly effective,” said Dr. Weiss.
Dr. Tafari’s group is designing a short, simple, culturally acceptable screening tool for use in primary care practice and will monitor its impact on physician prescribing habits and overall men’s health outcomes.
Dr. Tafari received funding from the Hospital Research Foundation and Freemasons Centre for Male Health and Wellbeing in Adelaide, South Australia. Dr. Tafari, Dr. Tsambarlis, Dr. Weiss, and Dr. Richards had no relevant conflicts of interest to declare.
A version of this article appeared on Medscape.com.