User login
Official news magazine of the Society of Hospital Medicine
Copyright by Society of Hospital Medicine or related companies. All rights reserved. ISSN 1553-085X
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'pane-pub-article-hospitalist')]


The hospitalist role in treating opioid use disorder
Screen patients at the time of admission
Let’s begin with a brief case. A 25-year-old patient with a history of injection heroin use is in your care. He is admitted for treatment of endocarditis and will remain in the hospital for intravenous antibiotics for several weeks. Over the first few days of hospitalization, he frequently asks for pain medicine, stating that he is in severe pain, withdrawal, and having opioid cravings. On day 3, he leaves the hospital against medical advice. After 2 weeks, he presents to the ED in septic shock and spends several weeks in the ICU. Or, alternatively, he is found down in the community and pronounced dead from a heroin overdose.
These cases occur all too often, and hospitalists across the nation are actively building knowledge and programs to improve care for patients with opioid use disorder (OUD). It is evident that opioid misuse is the public health crisis of our time. In 2017, over 70,000 patients died from an overdose, and over 2 million patients in the United States have a diagnosis of OUD.1,2 Many of these patients interact with the hospital at some point during the course of their illness for management of overdose, withdrawal, and other complications of OUD, including endocarditis, osteomyelitis, and skin and soft tissue infections. Moreover, just 20% of the 580,000 patients hospitalized with OUD in 2015 presented as a direct sequelae of the disease.3 Patients with OUD are often admitted for unrelated reasons, but their addiction goes unaddressed.
Opioid use disorder, like many of the other conditions we see, is a chronic relapsing remitting medical disease and a risk factor for premature mortality. When a patient with diabetes is admitted with cellulitis, we might check an A1C, provide diabetic counseling, and offer evidence-based diabetes treatment, including medications like insulin. We rarely build similar systems of care within the walls of our hospitals to treat OUD like we do for diabetes or other commonly encountered diseases like heart failure and chronic obstructive pulmonary disease.
We should be intentional about separating prevention from treatment. Significant work has gone into reducing the availability of prescription opioids and increasing utilization of prescription drug monitoring programs. As a result, the average morphine milligram equivalent per opioid prescription has decreased since 2010.4 An unintended consequence of restricting legal opioids is potentially pushing patients with opioid addiction towards heroin and fentanyl. Limiting opioid prescriptions alone will only decrease opioid overdose mortality by 5% through 2025.5 Thus, treatment of OUD is critical and something that hospitalists should be trained and engaged in.
Food and Drug Administration–approved OUD treatment includes buprenorphine, methadone, and extended-release naltrexone. Buprenorphine is a partial opioid agonist that treats withdrawal and cravings. Buprenorphine started in the hospital reduces mortality, increases time spent in outpatient treatment after discharge, and reduces opioid-related 30-day readmissions by over 50%.6-8 The number needed to treat with buprenorphine to prevent return to illicit opioid use is two.9 While physicians require an 8-hour “x-waiver” training (physician assistants and nurse practitioners require a 24-hour training) to prescribe buprenorphine for the outpatient treatment of OUD, such certification is not required to order the medication as part of an acute hospitalization.
Hospitalization represents a reachable moment and unique opportunity to start treatment for OUD. Patients are away from triggering environments and surrounded by supportive staff. Unfortunately, up to 30% of these patients leave the hospital against medical advice because of inadequately treated withdrawal, unaddressed cravings, and fear of mistreatment.10 Buprenorphine therapy may help tackle the physiological piece of hospital-based treatment, but we also must work on shifting the culture of our institutions. Importantly, OUD is a medical diagnosis. These patients must receive the same dignity, autonomy, and meaningful care afforded to patients with other medical diagnoses. Patients with OUD are not “addicts,” “abusers,” or “frequent fliers.”
Hospitalists have a clear and compelling role in treating OUD. The National Academy of Medicine recently held a workshop where they compared similarities of the HIV crisis with today’s opioid epidemic. The Academy advocated for the development of hospital-based protocols that empower physicians, physician assistants, and nurse practitioners to integrate the treatment of OUD into their practice.11 Some in our field may feel that treating underlying addiction is a role for behavioral health practitioners. This is akin to having said that HIV specialists should be the only providers to treat patients with HIV during its peak. There are simply not enough psychiatrists or addiction medicine specialists to treat all of the patients who need us during this time of national urgency.
There are several examples of institutions that are laying the groundwork for this important work. The University of California, San Francisco; Oregon Health and Science University, Portland; the University of Colorado at Denver, Aurora; Rush Medical College, Boston; Boston Medical Center; the Icahn School of Medicine at Mount Sinai, New York; and the University of Texas at Austin – to name a few. Offering OUD treatment in the hospital setting must be our new and only acceptable standard of care.
What is next? We can start by screening patients for OUD at the time of admission. This can be accomplished by asking two questions: Does the patient misuse prescription or nonprescription opioids? And if so, does the patient become sick if they abruptly stop? If the patient says yes to both, steps should be taken to provide direct and purposeful care related to OUD. Hospitalists should become familiar with buprenorphine therapy and work to reduce stigma by using people-first language with patients, staff, and in medical documentation.
As a society, we should balance our past focus on optimizing opioid prescribing with current efforts to bolster treatment. To that end, a group of SHM members applied to establish a Substance Use Disorder Special Interest Group, which was recently approved by the SHM board of directors. Details on its rollout will be announced shortly. The intention is that this group will serve as a resource to SHM membership and leadership
As practitioners of hospital medicine, we may not have anticipated playing a direct role in treating patients’ underlying addiction. By empowering hospitalists and wisely using medical hospitalization as a time to treat OUD, we can all have an incredible impact on our patients. Let’s get to work.
Mr. Bottner is a hospitalist at Dell Seton Medical Center, Austin, Texas, and clinical assistant professor at the University of Texas at Austin.
References
1. Katz J. You draw it: Just how bad is the drug overdose epidemic? New York Times. https://www.nytimes.com/interactive/2017/04/14/upshot/drug-overdose-epidemic-you-draw-it.html. Published Oct 26, 2017.
2. National Institute on Drug Abuse. Ohio – Opioid summaries by state. 2018. https://d14rmgtrwzf5a.cloudfront.net/sites/default/files/ohio_2018.pdf.
3. Peterson C et al. U.S. hospital discharges documenting patient opioid use disorder without opioid overdose or treatment services, 2011-2015. J Subst Abuse Treat. 2018;92:35-39. doi: 10.1016/j.jsat.2018.06.008.
4. Guy GP. Vital Signs: Changes in opioid prescribing in the United States, 2006-2015. Morb Mortal Wkly Rep. 2017;66. doi: 10.15585/mmwr.mm6626a4.
5. Chen Q et al. Prevention of prescription opioid misuse and projected overdose deaths in the United States. JAMA Netw Open. doi: 10.1001/jamanetworkopen.2018.7621.
6. Liebschutz J et al. Buprenorphine treatment for hospitalized, opioid-dependent patients: A randomized clinical trial. JAMA Intern Med. 2014;174(8):1369-76.
7. Moreno JL et al. Predictors for 30-day and 90-day hospital readmission among patients with opioid use disorder. J Addict Med. 2019. doi: 10.1097/ADM.0000000000000499.
8. Larochelle MR et al. Medication for opioid use disorder after nonfatal opioid overdose and association with mortality: A cohort study. Ann Intern Med. June 2018. doi: 10.7326/M17-3107.
9. Raleigh MF. Buprenorphine maintenance vs. placebo for opioid dependence. Am Fam Physician. 2017;95(5). https://www.aafp.org/afp/2017/0301/od1.html. Accessed May 12, 2019.
10. Ti L et al. Leaving the hospital against medical advice among people who use illicit drugs: A systematic review. Am J Public Health. 2015;105(12):2587. doi: 10.2105/AJPH.2015.302885a.
11. Springer SAM et al. Integrating treatment at the intersection of opioid use disorder and infectious disease epidemics in medical settings: A call for action after a National Academies of Sciences, Engineering, and Medicine workshop. Ann Intern Med. 2018;169(5):335-6. doi: 10.7326/M18-1203.
Screen patients at the time of admission
Screen patients at the time of admission
Let’s begin with a brief case. A 25-year-old patient with a history of injection heroin use is in your care. He is admitted for treatment of endocarditis and will remain in the hospital for intravenous antibiotics for several weeks. Over the first few days of hospitalization, he frequently asks for pain medicine, stating that he is in severe pain, withdrawal, and having opioid cravings. On day 3, he leaves the hospital against medical advice. After 2 weeks, he presents to the ED in septic shock and spends several weeks in the ICU. Or, alternatively, he is found down in the community and pronounced dead from a heroin overdose.
These cases occur all too often, and hospitalists across the nation are actively building knowledge and programs to improve care for patients with opioid use disorder (OUD). It is evident that opioid misuse is the public health crisis of our time. In 2017, over 70,000 patients died from an overdose, and over 2 million patients in the United States have a diagnosis of OUD.1,2 Many of these patients interact with the hospital at some point during the course of their illness for management of overdose, withdrawal, and other complications of OUD, including endocarditis, osteomyelitis, and skin and soft tissue infections. Moreover, just 20% of the 580,000 patients hospitalized with OUD in 2015 presented as a direct sequelae of the disease.3 Patients with OUD are often admitted for unrelated reasons, but their addiction goes unaddressed.
Opioid use disorder, like many of the other conditions we see, is a chronic relapsing remitting medical disease and a risk factor for premature mortality. When a patient with diabetes is admitted with cellulitis, we might check an A1C, provide diabetic counseling, and offer evidence-based diabetes treatment, including medications like insulin. We rarely build similar systems of care within the walls of our hospitals to treat OUD like we do for diabetes or other commonly encountered diseases like heart failure and chronic obstructive pulmonary disease.
We should be intentional about separating prevention from treatment. Significant work has gone into reducing the availability of prescription opioids and increasing utilization of prescription drug monitoring programs. As a result, the average morphine milligram equivalent per opioid prescription has decreased since 2010.4 An unintended consequence of restricting legal opioids is potentially pushing patients with opioid addiction towards heroin and fentanyl. Limiting opioid prescriptions alone will only decrease opioid overdose mortality by 5% through 2025.5 Thus, treatment of OUD is critical and something that hospitalists should be trained and engaged in.
Food and Drug Administration–approved OUD treatment includes buprenorphine, methadone, and extended-release naltrexone. Buprenorphine is a partial opioid agonist that treats withdrawal and cravings. Buprenorphine started in the hospital reduces mortality, increases time spent in outpatient treatment after discharge, and reduces opioid-related 30-day readmissions by over 50%.6-8 The number needed to treat with buprenorphine to prevent return to illicit opioid use is two.9 While physicians require an 8-hour “x-waiver” training (physician assistants and nurse practitioners require a 24-hour training) to prescribe buprenorphine for the outpatient treatment of OUD, such certification is not required to order the medication as part of an acute hospitalization.
Hospitalization represents a reachable moment and unique opportunity to start treatment for OUD. Patients are away from triggering environments and surrounded by supportive staff. Unfortunately, up to 30% of these patients leave the hospital against medical advice because of inadequately treated withdrawal, unaddressed cravings, and fear of mistreatment.10 Buprenorphine therapy may help tackle the physiological piece of hospital-based treatment, but we also must work on shifting the culture of our institutions. Importantly, OUD is a medical diagnosis. These patients must receive the same dignity, autonomy, and meaningful care afforded to patients with other medical diagnoses. Patients with OUD are not “addicts,” “abusers,” or “frequent fliers.”
Hospitalists have a clear and compelling role in treating OUD. The National Academy of Medicine recently held a workshop where they compared similarities of the HIV crisis with today’s opioid epidemic. The Academy advocated for the development of hospital-based protocols that empower physicians, physician assistants, and nurse practitioners to integrate the treatment of OUD into their practice.11 Some in our field may feel that treating underlying addiction is a role for behavioral health practitioners. This is akin to having said that HIV specialists should be the only providers to treat patients with HIV during its peak. There are simply not enough psychiatrists or addiction medicine specialists to treat all of the patients who need us during this time of national urgency.
There are several examples of institutions that are laying the groundwork for this important work. The University of California, San Francisco; Oregon Health and Science University, Portland; the University of Colorado at Denver, Aurora; Rush Medical College, Boston; Boston Medical Center; the Icahn School of Medicine at Mount Sinai, New York; and the University of Texas at Austin – to name a few. Offering OUD treatment in the hospital setting must be our new and only acceptable standard of care.
What is next? We can start by screening patients for OUD at the time of admission. This can be accomplished by asking two questions: Does the patient misuse prescription or nonprescription opioids? And if so, does the patient become sick if they abruptly stop? If the patient says yes to both, steps should be taken to provide direct and purposeful care related to OUD. Hospitalists should become familiar with buprenorphine therapy and work to reduce stigma by using people-first language with patients, staff, and in medical documentation.
As a society, we should balance our past focus on optimizing opioid prescribing with current efforts to bolster treatment. To that end, a group of SHM members applied to establish a Substance Use Disorder Special Interest Group, which was recently approved by the SHM board of directors. Details on its rollout will be announced shortly. The intention is that this group will serve as a resource to SHM membership and leadership
As practitioners of hospital medicine, we may not have anticipated playing a direct role in treating patients’ underlying addiction. By empowering hospitalists and wisely using medical hospitalization as a time to treat OUD, we can all have an incredible impact on our patients. Let’s get to work.
Mr. Bottner is a hospitalist at Dell Seton Medical Center, Austin, Texas, and clinical assistant professor at the University of Texas at Austin.
References
1. Katz J. You draw it: Just how bad is the drug overdose epidemic? New York Times. https://www.nytimes.com/interactive/2017/04/14/upshot/drug-overdose-epidemic-you-draw-it.html. Published Oct 26, 2017.
2. National Institute on Drug Abuse. Ohio – Opioid summaries by state. 2018. https://d14rmgtrwzf5a.cloudfront.net/sites/default/files/ohio_2018.pdf.
3. Peterson C et al. U.S. hospital discharges documenting patient opioid use disorder without opioid overdose or treatment services, 2011-2015. J Subst Abuse Treat. 2018;92:35-39. doi: 10.1016/j.jsat.2018.06.008.
4. Guy GP. Vital Signs: Changes in opioid prescribing in the United States, 2006-2015. Morb Mortal Wkly Rep. 2017;66. doi: 10.15585/mmwr.mm6626a4.
5. Chen Q et al. Prevention of prescription opioid misuse and projected overdose deaths in the United States. JAMA Netw Open. doi: 10.1001/jamanetworkopen.2018.7621.
6. Liebschutz J et al. Buprenorphine treatment for hospitalized, opioid-dependent patients: A randomized clinical trial. JAMA Intern Med. 2014;174(8):1369-76.
7. Moreno JL et al. Predictors for 30-day and 90-day hospital readmission among patients with opioid use disorder. J Addict Med. 2019. doi: 10.1097/ADM.0000000000000499.
8. Larochelle MR et al. Medication for opioid use disorder after nonfatal opioid overdose and association with mortality: A cohort study. Ann Intern Med. June 2018. doi: 10.7326/M17-3107.
9. Raleigh MF. Buprenorphine maintenance vs. placebo for opioid dependence. Am Fam Physician. 2017;95(5). https://www.aafp.org/afp/2017/0301/od1.html. Accessed May 12, 2019.
10. Ti L et al. Leaving the hospital against medical advice among people who use illicit drugs: A systematic review. Am J Public Health. 2015;105(12):2587. doi: 10.2105/AJPH.2015.302885a.
11. Springer SAM et al. Integrating treatment at the intersection of opioid use disorder and infectious disease epidemics in medical settings: A call for action after a National Academies of Sciences, Engineering, and Medicine workshop. Ann Intern Med. 2018;169(5):335-6. doi: 10.7326/M18-1203.
Let’s begin with a brief case. A 25-year-old patient with a history of injection heroin use is in your care. He is admitted for treatment of endocarditis and will remain in the hospital for intravenous antibiotics for several weeks. Over the first few days of hospitalization, he frequently asks for pain medicine, stating that he is in severe pain, withdrawal, and having opioid cravings. On day 3, he leaves the hospital against medical advice. After 2 weeks, he presents to the ED in septic shock and spends several weeks in the ICU. Or, alternatively, he is found down in the community and pronounced dead from a heroin overdose.
These cases occur all too often, and hospitalists across the nation are actively building knowledge and programs to improve care for patients with opioid use disorder (OUD). It is evident that opioid misuse is the public health crisis of our time. In 2017, over 70,000 patients died from an overdose, and over 2 million patients in the United States have a diagnosis of OUD.1,2 Many of these patients interact with the hospital at some point during the course of their illness for management of overdose, withdrawal, and other complications of OUD, including endocarditis, osteomyelitis, and skin and soft tissue infections. Moreover, just 20% of the 580,000 patients hospitalized with OUD in 2015 presented as a direct sequelae of the disease.3 Patients with OUD are often admitted for unrelated reasons, but their addiction goes unaddressed.
Opioid use disorder, like many of the other conditions we see, is a chronic relapsing remitting medical disease and a risk factor for premature mortality. When a patient with diabetes is admitted with cellulitis, we might check an A1C, provide diabetic counseling, and offer evidence-based diabetes treatment, including medications like insulin. We rarely build similar systems of care within the walls of our hospitals to treat OUD like we do for diabetes or other commonly encountered diseases like heart failure and chronic obstructive pulmonary disease.
We should be intentional about separating prevention from treatment. Significant work has gone into reducing the availability of prescription opioids and increasing utilization of prescription drug monitoring programs. As a result, the average morphine milligram equivalent per opioid prescription has decreased since 2010.4 An unintended consequence of restricting legal opioids is potentially pushing patients with opioid addiction towards heroin and fentanyl. Limiting opioid prescriptions alone will only decrease opioid overdose mortality by 5% through 2025.5 Thus, treatment of OUD is critical and something that hospitalists should be trained and engaged in.
Food and Drug Administration–approved OUD treatment includes buprenorphine, methadone, and extended-release naltrexone. Buprenorphine is a partial opioid agonist that treats withdrawal and cravings. Buprenorphine started in the hospital reduces mortality, increases time spent in outpatient treatment after discharge, and reduces opioid-related 30-day readmissions by over 50%.6-8 The number needed to treat with buprenorphine to prevent return to illicit opioid use is two.9 While physicians require an 8-hour “x-waiver” training (physician assistants and nurse practitioners require a 24-hour training) to prescribe buprenorphine for the outpatient treatment of OUD, such certification is not required to order the medication as part of an acute hospitalization.
Hospitalization represents a reachable moment and unique opportunity to start treatment for OUD. Patients are away from triggering environments and surrounded by supportive staff. Unfortunately, up to 30% of these patients leave the hospital against medical advice because of inadequately treated withdrawal, unaddressed cravings, and fear of mistreatment.10 Buprenorphine therapy may help tackle the physiological piece of hospital-based treatment, but we also must work on shifting the culture of our institutions. Importantly, OUD is a medical diagnosis. These patients must receive the same dignity, autonomy, and meaningful care afforded to patients with other medical diagnoses. Patients with OUD are not “addicts,” “abusers,” or “frequent fliers.”
Hospitalists have a clear and compelling role in treating OUD. The National Academy of Medicine recently held a workshop where they compared similarities of the HIV crisis with today’s opioid epidemic. The Academy advocated for the development of hospital-based protocols that empower physicians, physician assistants, and nurse practitioners to integrate the treatment of OUD into their practice.11 Some in our field may feel that treating underlying addiction is a role for behavioral health practitioners. This is akin to having said that HIV specialists should be the only providers to treat patients with HIV during its peak. There are simply not enough psychiatrists or addiction medicine specialists to treat all of the patients who need us during this time of national urgency.
There are several examples of institutions that are laying the groundwork for this important work. The University of California, San Francisco; Oregon Health and Science University, Portland; the University of Colorado at Denver, Aurora; Rush Medical College, Boston; Boston Medical Center; the Icahn School of Medicine at Mount Sinai, New York; and the University of Texas at Austin – to name a few. Offering OUD treatment in the hospital setting must be our new and only acceptable standard of care.
What is next? We can start by screening patients for OUD at the time of admission. This can be accomplished by asking two questions: Does the patient misuse prescription or nonprescription opioids? And if so, does the patient become sick if they abruptly stop? If the patient says yes to both, steps should be taken to provide direct and purposeful care related to OUD. Hospitalists should become familiar with buprenorphine therapy and work to reduce stigma by using people-first language with patients, staff, and in medical documentation.
As a society, we should balance our past focus on optimizing opioid prescribing with current efforts to bolster treatment. To that end, a group of SHM members applied to establish a Substance Use Disorder Special Interest Group, which was recently approved by the SHM board of directors. Details on its rollout will be announced shortly. The intention is that this group will serve as a resource to SHM membership and leadership
As practitioners of hospital medicine, we may not have anticipated playing a direct role in treating patients’ underlying addiction. By empowering hospitalists and wisely using medical hospitalization as a time to treat OUD, we can all have an incredible impact on our patients. Let’s get to work.
Mr. Bottner is a hospitalist at Dell Seton Medical Center, Austin, Texas, and clinical assistant professor at the University of Texas at Austin.
References
1. Katz J. You draw it: Just how bad is the drug overdose epidemic? New York Times. https://www.nytimes.com/interactive/2017/04/14/upshot/drug-overdose-epidemic-you-draw-it.html. Published Oct 26, 2017.
2. National Institute on Drug Abuse. Ohio – Opioid summaries by state. 2018. https://d14rmgtrwzf5a.cloudfront.net/sites/default/files/ohio_2018.pdf.
3. Peterson C et al. U.S. hospital discharges documenting patient opioid use disorder without opioid overdose or treatment services, 2011-2015. J Subst Abuse Treat. 2018;92:35-39. doi: 10.1016/j.jsat.2018.06.008.
4. Guy GP. Vital Signs: Changes in opioid prescribing in the United States, 2006-2015. Morb Mortal Wkly Rep. 2017;66. doi: 10.15585/mmwr.mm6626a4.
5. Chen Q et al. Prevention of prescription opioid misuse and projected overdose deaths in the United States. JAMA Netw Open. doi: 10.1001/jamanetworkopen.2018.7621.
6. Liebschutz J et al. Buprenorphine treatment for hospitalized, opioid-dependent patients: A randomized clinical trial. JAMA Intern Med. 2014;174(8):1369-76.
7. Moreno JL et al. Predictors for 30-day and 90-day hospital readmission among patients with opioid use disorder. J Addict Med. 2019. doi: 10.1097/ADM.0000000000000499.
8. Larochelle MR et al. Medication for opioid use disorder after nonfatal opioid overdose and association with mortality: A cohort study. Ann Intern Med. June 2018. doi: 10.7326/M17-3107.
9. Raleigh MF. Buprenorphine maintenance vs. placebo for opioid dependence. Am Fam Physician. 2017;95(5). https://www.aafp.org/afp/2017/0301/od1.html. Accessed May 12, 2019.
10. Ti L et al. Leaving the hospital against medical advice among people who use illicit drugs: A systematic review. Am J Public Health. 2015;105(12):2587. doi: 10.2105/AJPH.2015.302885a.
11. Springer SAM et al. Integrating treatment at the intersection of opioid use disorder and infectious disease epidemics in medical settings: A call for action after a National Academies of Sciences, Engineering, and Medicine workshop. Ann Intern Med. 2018;169(5):335-6. doi: 10.7326/M18-1203.
Same-day discharge after elective PCI has increased value and patient satisfaction
Background: SDDs are as safe as non-SDDs (NSDDs) in patients after elective PCI, yet there has been only a modest increase in SDD.
Study design: Observational cross-sectional cohort study.
Setting: 493 hospitals in the United States.
Synopsis: With use of the national Premier Healthcare Database, 672,470 elective PCIs from January 2006 to December 2015 with 1-year follow-up showed a wide variation in SDD from 0% to 83% among hospitals with the overall corrected rate of 3.5%. Low-volume PCI hospitals did not increase the rate. Additionally, the cost of SDD patients was $5,128 less than NSDD patients. There was cost saving even with higher-risk transfemoral approaches and patients needing periprocedural hemodynamic or ventilatory support. Complications (death, bleeding, acute kidney injury, or acute MI at 30, 90, and 365 days) were not higher for SDD than for NSDD patients.
Limitations include that 2015 data may not reflect current practices. ICD 9 codes used for obtaining complications data can be misclassified. Cost savings are variable. Patients with periprocedural complications were not candidates for SDD but were included in the data. The study does not account for variation in technique, PCI characteristics, or SDD criteria of hospitals.
Bottom line: Prevalence of SDDs for elective PCI patients varies by institution and is an underutilized opportunity to significantly reduce hospital costs and increase patient satisfaction while maintaining the safety of patients.
Citation: Amin AP et al. Association of same-day discharge after elective percutaneous coronary intervention in the United States with costs and outcomes. JAMA Cardiol. Published online 2018 Sep 26. doi: 10.1001/jamacardio.2018.3029.
Dr. Kochar is an assistant professor of medicine in the division of hospital medicine at Mount Sinai Hospital, New York.
Background: SDDs are as safe as non-SDDs (NSDDs) in patients after elective PCI, yet there has been only a modest increase in SDD.
Study design: Observational cross-sectional cohort study.
Setting: 493 hospitals in the United States.
Synopsis: With use of the national Premier Healthcare Database, 672,470 elective PCIs from January 2006 to December 2015 with 1-year follow-up showed a wide variation in SDD from 0% to 83% among hospitals with the overall corrected rate of 3.5%. Low-volume PCI hospitals did not increase the rate. Additionally, the cost of SDD patients was $5,128 less than NSDD patients. There was cost saving even with higher-risk transfemoral approaches and patients needing periprocedural hemodynamic or ventilatory support. Complications (death, bleeding, acute kidney injury, or acute MI at 30, 90, and 365 days) were not higher for SDD than for NSDD patients.
Limitations include that 2015 data may not reflect current practices. ICD 9 codes used for obtaining complications data can be misclassified. Cost savings are variable. Patients with periprocedural complications were not candidates for SDD but were included in the data. The study does not account for variation in technique, PCI characteristics, or SDD criteria of hospitals.
Bottom line: Prevalence of SDDs for elective PCI patients varies by institution and is an underutilized opportunity to significantly reduce hospital costs and increase patient satisfaction while maintaining the safety of patients.
Citation: Amin AP et al. Association of same-day discharge after elective percutaneous coronary intervention in the United States with costs and outcomes. JAMA Cardiol. Published online 2018 Sep 26. doi: 10.1001/jamacardio.2018.3029.
Dr. Kochar is an assistant professor of medicine in the division of hospital medicine at Mount Sinai Hospital, New York.
Background: SDDs are as safe as non-SDDs (NSDDs) in patients after elective PCI, yet there has been only a modest increase in SDD.
Study design: Observational cross-sectional cohort study.
Setting: 493 hospitals in the United States.
Synopsis: With use of the national Premier Healthcare Database, 672,470 elective PCIs from January 2006 to December 2015 with 1-year follow-up showed a wide variation in SDD from 0% to 83% among hospitals with the overall corrected rate of 3.5%. Low-volume PCI hospitals did not increase the rate. Additionally, the cost of SDD patients was $5,128 less than NSDD patients. There was cost saving even with higher-risk transfemoral approaches and patients needing periprocedural hemodynamic or ventilatory support. Complications (death, bleeding, acute kidney injury, or acute MI at 30, 90, and 365 days) were not higher for SDD than for NSDD patients.
Limitations include that 2015 data may not reflect current practices. ICD 9 codes used for obtaining complications data can be misclassified. Cost savings are variable. Patients with periprocedural complications were not candidates for SDD but were included in the data. The study does not account for variation in technique, PCI characteristics, or SDD criteria of hospitals.
Bottom line: Prevalence of SDDs for elective PCI patients varies by institution and is an underutilized opportunity to significantly reduce hospital costs and increase patient satisfaction while maintaining the safety of patients.
Citation: Amin AP et al. Association of same-day discharge after elective percutaneous coronary intervention in the United States with costs and outcomes. JAMA Cardiol. Published online 2018 Sep 26. doi: 10.1001/jamacardio.2018.3029.
Dr. Kochar is an assistant professor of medicine in the division of hospital medicine at Mount Sinai Hospital, New York.
Risk of cardiac events jumps after COPD exacerbation
particularly in older individuals, new research has found.
In Respirology, researchers report the outcomes of a nationwide, register-based study involving 118,807 patients with chronic obstructive pulmonary disease (COPD) who experienced a major adverse cardiac event after an exacerbation.
They found that the risk of any major cardiac adverse event increased 270% in the 4 weeks after the onset of an exacerbation (95% confidence interval, 3.60-3.80). The strongest association was seen for cardiovascular death, for which there was a 333% increase in risk, but there was also a 257% increase in the risk of acute MI and 178% increase in the risk of stroke.
The risk of major adverse cardiac events was even higher among individuals who were hospitalized because of their COPD exacerbation (odds ratio, 5.92), compared with a 150% increase in risk among those who weren’t hospitalized but were treated with oral corticosteroids and 108% increase among those treated with amoxicillin with enzyme inhibitors.
The risk of a major cardiac event after a COPD exacerbation also increased with age. Among individuals younger than 55 years, there was a 131% increase in risk, but among those aged 55-69 years there was a 234% increase, among those aged 70-79 years the risk increased 282%, and among those aged 80 years and older it increased 318%.
Mette Reilev, from the department of public health at the University of Southern Denmark, Odense, and coauthors suggested that acute exacerbations were associated with elevated levels of systemic inflammatory markers such as fibrinogen and interleukin-6, which were potently prothrombotic and could potentially trigger cardiovascular events.
“Additionally, exacerbations may trigger type II myocardial infarctions secondary to an imbalance in oxygen supply and demand,” they wrote.
The authors raised the question of whether cardiovascular prevention strategies should be part of treatment recommendations for people with COPD, and suggested that prevention of COPD exacerbations could be justified even on cardiovascular grounds alone.
“Studies investigating the effect of cardiovascular treatment on the course of disease among COPD exacerbators are extremely scarce,” they wrote. “Thus, it is currently unknown how to optimize treatment and mitigate the increased risk of [major adverse cardiovascular events] following the onset of exacerbations.”
However, they noted that prednisolone treatment for more severe exacerbations may have a confounding effect, as oral corticosteroids could induce dyslipidemia, hypertension, and hyperglycemia, and increase long-term cardiovascular risk.
Six authors declared funding from the pharmaceutical industry – three of which were institutional support – unrelated to the study.
SOURCE: Reilev M et al. Respirology. 2019 Jun 21. doi: 10.1111/resp.13620.
particularly in older individuals, new research has found.
In Respirology, researchers report the outcomes of a nationwide, register-based study involving 118,807 patients with chronic obstructive pulmonary disease (COPD) who experienced a major adverse cardiac event after an exacerbation.
They found that the risk of any major cardiac adverse event increased 270% in the 4 weeks after the onset of an exacerbation (95% confidence interval, 3.60-3.80). The strongest association was seen for cardiovascular death, for which there was a 333% increase in risk, but there was also a 257% increase in the risk of acute MI and 178% increase in the risk of stroke.
The risk of major adverse cardiac events was even higher among individuals who were hospitalized because of their COPD exacerbation (odds ratio, 5.92), compared with a 150% increase in risk among those who weren’t hospitalized but were treated with oral corticosteroids and 108% increase among those treated with amoxicillin with enzyme inhibitors.
The risk of a major cardiac event after a COPD exacerbation also increased with age. Among individuals younger than 55 years, there was a 131% increase in risk, but among those aged 55-69 years there was a 234% increase, among those aged 70-79 years the risk increased 282%, and among those aged 80 years and older it increased 318%.
Mette Reilev, from the department of public health at the University of Southern Denmark, Odense, and coauthors suggested that acute exacerbations were associated with elevated levels of systemic inflammatory markers such as fibrinogen and interleukin-6, which were potently prothrombotic and could potentially trigger cardiovascular events.
“Additionally, exacerbations may trigger type II myocardial infarctions secondary to an imbalance in oxygen supply and demand,” they wrote.
The authors raised the question of whether cardiovascular prevention strategies should be part of treatment recommendations for people with COPD, and suggested that prevention of COPD exacerbations could be justified even on cardiovascular grounds alone.
“Studies investigating the effect of cardiovascular treatment on the course of disease among COPD exacerbators are extremely scarce,” they wrote. “Thus, it is currently unknown how to optimize treatment and mitigate the increased risk of [major adverse cardiovascular events] following the onset of exacerbations.”
However, they noted that prednisolone treatment for more severe exacerbations may have a confounding effect, as oral corticosteroids could induce dyslipidemia, hypertension, and hyperglycemia, and increase long-term cardiovascular risk.
Six authors declared funding from the pharmaceutical industry – three of which were institutional support – unrelated to the study.
SOURCE: Reilev M et al. Respirology. 2019 Jun 21. doi: 10.1111/resp.13620.
particularly in older individuals, new research has found.
In Respirology, researchers report the outcomes of a nationwide, register-based study involving 118,807 patients with chronic obstructive pulmonary disease (COPD) who experienced a major adverse cardiac event after an exacerbation.
They found that the risk of any major cardiac adverse event increased 270% in the 4 weeks after the onset of an exacerbation (95% confidence interval, 3.60-3.80). The strongest association was seen for cardiovascular death, for which there was a 333% increase in risk, but there was also a 257% increase in the risk of acute MI and 178% increase in the risk of stroke.
The risk of major adverse cardiac events was even higher among individuals who were hospitalized because of their COPD exacerbation (odds ratio, 5.92), compared with a 150% increase in risk among those who weren’t hospitalized but were treated with oral corticosteroids and 108% increase among those treated with amoxicillin with enzyme inhibitors.
The risk of a major cardiac event after a COPD exacerbation also increased with age. Among individuals younger than 55 years, there was a 131% increase in risk, but among those aged 55-69 years there was a 234% increase, among those aged 70-79 years the risk increased 282%, and among those aged 80 years and older it increased 318%.
Mette Reilev, from the department of public health at the University of Southern Denmark, Odense, and coauthors suggested that acute exacerbations were associated with elevated levels of systemic inflammatory markers such as fibrinogen and interleukin-6, which were potently prothrombotic and could potentially trigger cardiovascular events.
“Additionally, exacerbations may trigger type II myocardial infarctions secondary to an imbalance in oxygen supply and demand,” they wrote.
The authors raised the question of whether cardiovascular prevention strategies should be part of treatment recommendations for people with COPD, and suggested that prevention of COPD exacerbations could be justified even on cardiovascular grounds alone.
“Studies investigating the effect of cardiovascular treatment on the course of disease among COPD exacerbators are extremely scarce,” they wrote. “Thus, it is currently unknown how to optimize treatment and mitigate the increased risk of [major adverse cardiovascular events] following the onset of exacerbations.”
However, they noted that prednisolone treatment for more severe exacerbations may have a confounding effect, as oral corticosteroids could induce dyslipidemia, hypertension, and hyperglycemia, and increase long-term cardiovascular risk.
Six authors declared funding from the pharmaceutical industry – three of which were institutional support – unrelated to the study.
SOURCE: Reilev M et al. Respirology. 2019 Jun 21. doi: 10.1111/resp.13620.
FROM RESPIROLOGY
Evidence supports accuracy of COPD diagnosis tool
The ratio of the forced expiratory volume in 1 second to the forced vital capacity (FEV1:FVC) at the recommended threshold of 0.70 effectively diagnosed individuals at risk for clinically significant COPD, a longitudinal study of more than 24,000 individuals has found.
Guidelines from respiratory societies have long recommended a diagnosis of airflow obstruction when the FEV1:FVC is less than 0.70, but no rigorous, population-based studies have been conducted to support this recommendation, wrote Surya P. Bhatt, MD, of the University of Alabama at Birmingham, and colleagues.
“The selection of a threshold for defining airflow obstruction has major implications for patient care and public health as the prevalence of airflow obstruction can vary by as much as 33% depending on which threshold is selected,” they said.
In a study published in JAMA, the researchers reviewed data from 24,207 participants in the National Heart, Lung, and Blood Institute Pooled Cohorts Study to assess the accuracy of different thresholds in predicting COPD events in a large, multiethnic, U.S. population. All participants underwent spirometry; the average age at spirometry was 63 years, and 54% of the patients were women. Patients were enrolled during 1987-2000 and received follow-up longitudinally through 2016.
Overall, 3,925 participants experienced COPD-related events during an average of 15 years of follow-up (more than 340,757 person-years). These events included 3,563 hospitalizations and 447 deaths related to COPD.
The researchers compared three thresholds for FEV1:FVC ratios: a fixed optimal threshold of 0.71, a lower limit of normal (LLN) defined as 0.034, and the currently recommended 0.70.
The optimal 0.71 was not significantly different from the recommended 0.70 but was significantly more accurate than the LLN of 0.034. In addition, the 0.70 value was the optimal predictor in a subgroup analysis of ever-smokers and in multivariate analysis.
The findings were limited by several factors including the use of prebronchodilator spirometry, lack of adjustment for medication use, and limitation of outcomes to COPD mortality or clinical events mainly caused by COPD, which might exclude patients with mild to moderate disease, the researchers noted.
However, ” to help clinicians identify patients at increased risk for significant COPD, they said.
Lead author Dr. Bhatt disclosed a National Institutes of Health grant, consulting fees from Sunovion and research funds from Proterix Bio. The study was supported by grants from multiple agencies of the National Institutes of Health, including the National Heart, Lung, and Blood Institute, the National Institute of Neurological Disorders and Stroke, and the National Institute on Aging.
The ratio of the forced expiratory volume in 1 second to the forced vital capacity (FEV1:FVC) at the recommended threshold of 0.70 effectively diagnosed individuals at risk for clinically significant COPD, a longitudinal study of more than 24,000 individuals has found.
Guidelines from respiratory societies have long recommended a diagnosis of airflow obstruction when the FEV1:FVC is less than 0.70, but no rigorous, population-based studies have been conducted to support this recommendation, wrote Surya P. Bhatt, MD, of the University of Alabama at Birmingham, and colleagues.
“The selection of a threshold for defining airflow obstruction has major implications for patient care and public health as the prevalence of airflow obstruction can vary by as much as 33% depending on which threshold is selected,” they said.
In a study published in JAMA, the researchers reviewed data from 24,207 participants in the National Heart, Lung, and Blood Institute Pooled Cohorts Study to assess the accuracy of different thresholds in predicting COPD events in a large, multiethnic, U.S. population. All participants underwent spirometry; the average age at spirometry was 63 years, and 54% of the patients were women. Patients were enrolled during 1987-2000 and received follow-up longitudinally through 2016.
Overall, 3,925 participants experienced COPD-related events during an average of 15 years of follow-up (more than 340,757 person-years). These events included 3,563 hospitalizations and 447 deaths related to COPD.
The researchers compared three thresholds for FEV1:FVC ratios: a fixed optimal threshold of 0.71, a lower limit of normal (LLN) defined as 0.034, and the currently recommended 0.70.
The optimal 0.71 was not significantly different from the recommended 0.70 but was significantly more accurate than the LLN of 0.034. In addition, the 0.70 value was the optimal predictor in a subgroup analysis of ever-smokers and in multivariate analysis.
The findings were limited by several factors including the use of prebronchodilator spirometry, lack of adjustment for medication use, and limitation of outcomes to COPD mortality or clinical events mainly caused by COPD, which might exclude patients with mild to moderate disease, the researchers noted.
However, ” to help clinicians identify patients at increased risk for significant COPD, they said.
Lead author Dr. Bhatt disclosed a National Institutes of Health grant, consulting fees from Sunovion and research funds from Proterix Bio. The study was supported by grants from multiple agencies of the National Institutes of Health, including the National Heart, Lung, and Blood Institute, the National Institute of Neurological Disorders and Stroke, and the National Institute on Aging.
The ratio of the forced expiratory volume in 1 second to the forced vital capacity (FEV1:FVC) at the recommended threshold of 0.70 effectively diagnosed individuals at risk for clinically significant COPD, a longitudinal study of more than 24,000 individuals has found.
Guidelines from respiratory societies have long recommended a diagnosis of airflow obstruction when the FEV1:FVC is less than 0.70, but no rigorous, population-based studies have been conducted to support this recommendation, wrote Surya P. Bhatt, MD, of the University of Alabama at Birmingham, and colleagues.
“The selection of a threshold for defining airflow obstruction has major implications for patient care and public health as the prevalence of airflow obstruction can vary by as much as 33% depending on which threshold is selected,” they said.
In a study published in JAMA, the researchers reviewed data from 24,207 participants in the National Heart, Lung, and Blood Institute Pooled Cohorts Study to assess the accuracy of different thresholds in predicting COPD events in a large, multiethnic, U.S. population. All participants underwent spirometry; the average age at spirometry was 63 years, and 54% of the patients were women. Patients were enrolled during 1987-2000 and received follow-up longitudinally through 2016.
Overall, 3,925 participants experienced COPD-related events during an average of 15 years of follow-up (more than 340,757 person-years). These events included 3,563 hospitalizations and 447 deaths related to COPD.
The researchers compared three thresholds for FEV1:FVC ratios: a fixed optimal threshold of 0.71, a lower limit of normal (LLN) defined as 0.034, and the currently recommended 0.70.
The optimal 0.71 was not significantly different from the recommended 0.70 but was significantly more accurate than the LLN of 0.034. In addition, the 0.70 value was the optimal predictor in a subgroup analysis of ever-smokers and in multivariate analysis.
The findings were limited by several factors including the use of prebronchodilator spirometry, lack of adjustment for medication use, and limitation of outcomes to COPD mortality or clinical events mainly caused by COPD, which might exclude patients with mild to moderate disease, the researchers noted.
However, ” to help clinicians identify patients at increased risk for significant COPD, they said.
Lead author Dr. Bhatt disclosed a National Institutes of Health grant, consulting fees from Sunovion and research funds from Proterix Bio. The study was supported by grants from multiple agencies of the National Institutes of Health, including the National Heart, Lung, and Blood Institute, the National Institute of Neurological Disorders and Stroke, and the National Institute on Aging.
FROM JAMA
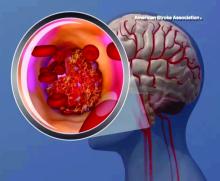
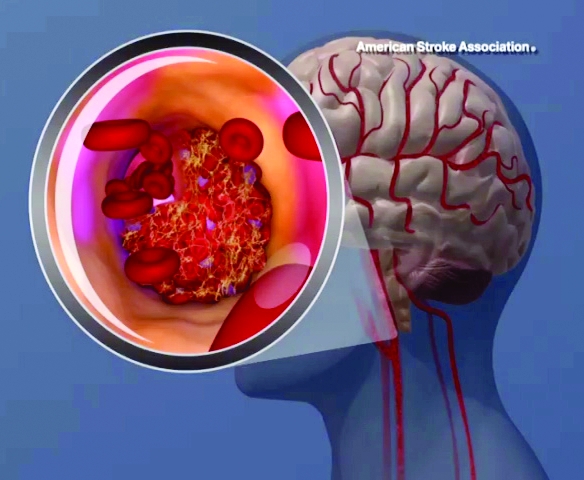
Lipoprotein(a) levels tied to higher ischemic stroke risk
High levels of lipoprotein(a) [Lp(a)] and LPA genotypes were linked to increased ischemic stroke risk in a recent large, contemporary general population study, investigators are reporting in the Journal of the American College of Cardiology.
Anne Langsted, MD, with Copenhagen University Hospital and the University of Copenhagen in Denmark, and her co-researchers evaluated the impact of high Lp(a) levels in a large contemporary cohort of 49,699 individuals in the Copenhagen General Population Study, and another 10,813 individuals in the Copenhagen City Heart Study.
Measurements assessed included plasma lipoprotein(a) levels and carrier or noncarrier status for LPA rs10455872. The endpoint of ischemic stroke was ascertained from Danish national health registries and confirmed by physicians.
Although risk estimates were less pronounced than what was reported before regarding the link between Lp(a) for ischemic heart disease and aortic valve stenosis, the risk of stroke was increased by a factor of 1.6 among individuals with high Lp(a) levels as compared to those with lower levels, the investigators said.
Compared with noncarriers of LPA rs1045572, the hazard ratio for ischemic stroke was 1.23 for carriers of LPA rs1045572, which was associated with high levels plasma lipoprotein(a) levels, according to the researchers.
“Our results indicate a causal association of Lp(a) with risk of ischemic stroke, and emphasize the need for randomized, controlled clinical trials on the effect of Lp(a)-lowering to prevent cardiovascular disease including ischemic stroke,” About 20% of the general population have high Lp(a) levels, and some individuals have extremely high levels, Dr. Langsted and co-authors said in their report.
Interest in Lp(a) as a risk factor for cardiovascular disease has been reignited following large studies showing that high Lp(a) levels were linked to increased risk of myocardial infarction and aortic valve stenosis, according to the investigators.
However, results of various studies are conflicting as to whether high Lp(a) levels increase risk of hemorrhagic or ischemic stroke, they said.
Both cohort studies used in the analysis were supported by sources in Denmark including the Danish Medical Research Council and Copenhagen University Hospital. Dr. Langsted had no disclosures. One co-author reported disclosures related to Akcea, Amgen, Sanofi, Regeneron, and AstraZeneca.
SOURCE: Langsted A, et al. JACC 2019;74[1]: 54-66. doi: 10.1016/j.jacc.2019.03.524
This study linking high lipoprotein(a) [Lp(a)] levels to stroke risk, taken together with previous research, provide a sound basis to routinely perform one-time screening so that individuals with inherited high levels can try to avoid adverse cardiovascular outcomes, according to Christie M. Ballantyne, MD.
“As someone in the dual role of preventive cardiologist and patient with a strong family history of cardiovascular disease, I think that we have sufficient evidence that high Lp(a) is strongly associated with an increased risk of myocardial infarction, stroke, and aortic valve stenosis,” Dr. Ballantyne wrote in an editorial comment on the study.
Evidence is now “overwhelming” that high Lp(a) is linked to myocardial infarction and stroke, and it’s known that statins and aspirin reduce risk of these outcomes, he said in the commentary.
Despite that, scientific statements do not recommend routine Lp(a) testing due to a lack of clinical trials evidence; as a result, clinical trials are not including Lp(a) as a routine measurement: “We thus have a loop of futility—lack of routine measurement leads to lack of data,” he said.
This most recent study from Langsted and colleagues demonstrates that high Lp(a) levels, and genetic variants associated with Lp(a), are associated with increased ischemic stroke risk. “The genetics strongly supported that high Lp(a) levels were in the causal pathway for ischemic stroke and coronary heart disease,” Dr. Ballantyne said.
One major strength and weakness of the study is its large and relatively homogeneous European population—that bolstered the genetic analyses, but also means the data can’t be extrapolated to other populations, such as Africans and East Asians, who have higher stroke rates compared with Europeans, Dr. Ballantyne said.
Dr. Ballantyne is with the Department of Medicine and Center for Cardiometabolic Disease Prevention, Baylor College of Medicine, Houston, Tex. His editorial comment appears in the Journal of the American College of Cardiology (2019;74[1]:67-9. doi:10.1016/j.jacc.2019.05.029 . Dr. Ballantyne reported disclosures related to Akcea, Amgen, and Novartis.
This study linking high lipoprotein(a) [Lp(a)] levels to stroke risk, taken together with previous research, provide a sound basis to routinely perform one-time screening so that individuals with inherited high levels can try to avoid adverse cardiovascular outcomes, according to Christie M. Ballantyne, MD.
“As someone in the dual role of preventive cardiologist and patient with a strong family history of cardiovascular disease, I think that we have sufficient evidence that high Lp(a) is strongly associated with an increased risk of myocardial infarction, stroke, and aortic valve stenosis,” Dr. Ballantyne wrote in an editorial comment on the study.
Evidence is now “overwhelming” that high Lp(a) is linked to myocardial infarction and stroke, and it’s known that statins and aspirin reduce risk of these outcomes, he said in the commentary.
Despite that, scientific statements do not recommend routine Lp(a) testing due to a lack of clinical trials evidence; as a result, clinical trials are not including Lp(a) as a routine measurement: “We thus have a loop of futility—lack of routine measurement leads to lack of data,” he said.
This most recent study from Langsted and colleagues demonstrates that high Lp(a) levels, and genetic variants associated with Lp(a), are associated with increased ischemic stroke risk. “The genetics strongly supported that high Lp(a) levels were in the causal pathway for ischemic stroke and coronary heart disease,” Dr. Ballantyne said.
One major strength and weakness of the study is its large and relatively homogeneous European population—that bolstered the genetic analyses, but also means the data can’t be extrapolated to other populations, such as Africans and East Asians, who have higher stroke rates compared with Europeans, Dr. Ballantyne said.
Dr. Ballantyne is with the Department of Medicine and Center for Cardiometabolic Disease Prevention, Baylor College of Medicine, Houston, Tex. His editorial comment appears in the Journal of the American College of Cardiology (2019;74[1]:67-9. doi:10.1016/j.jacc.2019.05.029 . Dr. Ballantyne reported disclosures related to Akcea, Amgen, and Novartis.
This study linking high lipoprotein(a) [Lp(a)] levels to stroke risk, taken together with previous research, provide a sound basis to routinely perform one-time screening so that individuals with inherited high levels can try to avoid adverse cardiovascular outcomes, according to Christie M. Ballantyne, MD.
“As someone in the dual role of preventive cardiologist and patient with a strong family history of cardiovascular disease, I think that we have sufficient evidence that high Lp(a) is strongly associated with an increased risk of myocardial infarction, stroke, and aortic valve stenosis,” Dr. Ballantyne wrote in an editorial comment on the study.
Evidence is now “overwhelming” that high Lp(a) is linked to myocardial infarction and stroke, and it’s known that statins and aspirin reduce risk of these outcomes, he said in the commentary.
Despite that, scientific statements do not recommend routine Lp(a) testing due to a lack of clinical trials evidence; as a result, clinical trials are not including Lp(a) as a routine measurement: “We thus have a loop of futility—lack of routine measurement leads to lack of data,” he said.
This most recent study from Langsted and colleagues demonstrates that high Lp(a) levels, and genetic variants associated with Lp(a), are associated with increased ischemic stroke risk. “The genetics strongly supported that high Lp(a) levels were in the causal pathway for ischemic stroke and coronary heart disease,” Dr. Ballantyne said.
One major strength and weakness of the study is its large and relatively homogeneous European population—that bolstered the genetic analyses, but also means the data can’t be extrapolated to other populations, such as Africans and East Asians, who have higher stroke rates compared with Europeans, Dr. Ballantyne said.
Dr. Ballantyne is with the Department of Medicine and Center for Cardiometabolic Disease Prevention, Baylor College of Medicine, Houston, Tex. His editorial comment appears in the Journal of the American College of Cardiology (2019;74[1]:67-9. doi:10.1016/j.jacc.2019.05.029 . Dr. Ballantyne reported disclosures related to Akcea, Amgen, and Novartis.
High levels of lipoprotein(a) [Lp(a)] and LPA genotypes were linked to increased ischemic stroke risk in a recent large, contemporary general population study, investigators are reporting in the Journal of the American College of Cardiology.
Anne Langsted, MD, with Copenhagen University Hospital and the University of Copenhagen in Denmark, and her co-researchers evaluated the impact of high Lp(a) levels in a large contemporary cohort of 49,699 individuals in the Copenhagen General Population Study, and another 10,813 individuals in the Copenhagen City Heart Study.
Measurements assessed included plasma lipoprotein(a) levels and carrier or noncarrier status for LPA rs10455872. The endpoint of ischemic stroke was ascertained from Danish national health registries and confirmed by physicians.
Although risk estimates were less pronounced than what was reported before regarding the link between Lp(a) for ischemic heart disease and aortic valve stenosis, the risk of stroke was increased by a factor of 1.6 among individuals with high Lp(a) levels as compared to those with lower levels, the investigators said.
Compared with noncarriers of LPA rs1045572, the hazard ratio for ischemic stroke was 1.23 for carriers of LPA rs1045572, which was associated with high levels plasma lipoprotein(a) levels, according to the researchers.
“Our results indicate a causal association of Lp(a) with risk of ischemic stroke, and emphasize the need for randomized, controlled clinical trials on the effect of Lp(a)-lowering to prevent cardiovascular disease including ischemic stroke,” About 20% of the general population have high Lp(a) levels, and some individuals have extremely high levels, Dr. Langsted and co-authors said in their report.
Interest in Lp(a) as a risk factor for cardiovascular disease has been reignited following large studies showing that high Lp(a) levels were linked to increased risk of myocardial infarction and aortic valve stenosis, according to the investigators.
However, results of various studies are conflicting as to whether high Lp(a) levels increase risk of hemorrhagic or ischemic stroke, they said.
Both cohort studies used in the analysis were supported by sources in Denmark including the Danish Medical Research Council and Copenhagen University Hospital. Dr. Langsted had no disclosures. One co-author reported disclosures related to Akcea, Amgen, Sanofi, Regeneron, and AstraZeneca.
SOURCE: Langsted A, et al. JACC 2019;74[1]: 54-66. doi: 10.1016/j.jacc.2019.03.524
High levels of lipoprotein(a) [Lp(a)] and LPA genotypes were linked to increased ischemic stroke risk in a recent large, contemporary general population study, investigators are reporting in the Journal of the American College of Cardiology.
Anne Langsted, MD, with Copenhagen University Hospital and the University of Copenhagen in Denmark, and her co-researchers evaluated the impact of high Lp(a) levels in a large contemporary cohort of 49,699 individuals in the Copenhagen General Population Study, and another 10,813 individuals in the Copenhagen City Heart Study.
Measurements assessed included plasma lipoprotein(a) levels and carrier or noncarrier status for LPA rs10455872. The endpoint of ischemic stroke was ascertained from Danish national health registries and confirmed by physicians.
Although risk estimates were less pronounced than what was reported before regarding the link between Lp(a) for ischemic heart disease and aortic valve stenosis, the risk of stroke was increased by a factor of 1.6 among individuals with high Lp(a) levels as compared to those with lower levels, the investigators said.
Compared with noncarriers of LPA rs1045572, the hazard ratio for ischemic stroke was 1.23 for carriers of LPA rs1045572, which was associated with high levels plasma lipoprotein(a) levels, according to the researchers.
“Our results indicate a causal association of Lp(a) with risk of ischemic stroke, and emphasize the need for randomized, controlled clinical trials on the effect of Lp(a)-lowering to prevent cardiovascular disease including ischemic stroke,” About 20% of the general population have high Lp(a) levels, and some individuals have extremely high levels, Dr. Langsted and co-authors said in their report.
Interest in Lp(a) as a risk factor for cardiovascular disease has been reignited following large studies showing that high Lp(a) levels were linked to increased risk of myocardial infarction and aortic valve stenosis, according to the investigators.
However, results of various studies are conflicting as to whether high Lp(a) levels increase risk of hemorrhagic or ischemic stroke, they said.
Both cohort studies used in the analysis were supported by sources in Denmark including the Danish Medical Research Council and Copenhagen University Hospital. Dr. Langsted had no disclosures. One co-author reported disclosures related to Akcea, Amgen, Sanofi, Regeneron, and AstraZeneca.
SOURCE: Langsted A, et al. JACC 2019;74[1]: 54-66. doi: 10.1016/j.jacc.2019.03.524
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Key clinical point:
Major finding: Stroke risk was 1.6X higher with high Lp(a) levels.
Study details: Analysis of 49,699 individuals in the Copenhagen General Population Study, and 10,813 individuals in the Copenhagen City Heart Study.
Disclosures: Both studies were supported by the sources in Denmark including the Danish Medical Research Council and Copenhagen University Hospital. Dr. Langsted had no disclosures.
Source: Langsted A, et al. JACC 2019;74[1]: 54-66. doi: 10.1016/j.jacc.2019.03.524
Mitral valve repair improves prognosis in heart failure patients with secondary MR
Background: In patients with primary degenerative MR, MVR is curative, with the transcatheter approach being safer than surgical repair. However, it is unknown whether patients with secondary MR from left ventricular dilatation would confer the same benefit of MVR.
Study design: Multicenter, randomized, controlled, parallel-group, open-label trial.
Setting: 78 sites in the United States and Canada.
Synopsis: From December 2012 to June 2017, 614 patients from 78 centers in the United States and Canada with symptomatic MR were enrolled with 302 patients assigned to the device group (transcatheter MVR and medical treatment) and 312 to the control group (medical therapy). Over 2 years, the device group’s annual rate for heart failure hospitalizations was significantly lower (35.8%/patient-year versus 67.9%/patient-year in the control group), as was all-cause mortality (29.1% for the device group versus 46.1%). The rate of freedom from device-related complications was 96.6%, better than the goal of 88%. There was improvement in quality of life, functional capacity, severity of MR, and left ventricular remodeling.
Limitations include that investigators were not blinded because the device was visible on imaging. Longer follow-up in the device group may have contributed to the observed decreased mortality. It is unknown whether less-symptomatic patients would attain the same benefit.
Bottom line: In patients with symptomatic, moderate to severe, and severe secondary MR, MVR lowers rates of hospitalization, decreases mortality, and improves quality of life.
Citation: Stone GW et al. Transcatheter mitral-valve repair in patients with heart failure. N Engl J Med. 2018 Sep 23. doi: 10.1056/NEJMoa1806640.
Dr. Kochar is an assistant professor of medicine in the division of hospital medicine at Mount Sinai Hospital, New York.
Background: In patients with primary degenerative MR, MVR is curative, with the transcatheter approach being safer than surgical repair. However, it is unknown whether patients with secondary MR from left ventricular dilatation would confer the same benefit of MVR.
Study design: Multicenter, randomized, controlled, parallel-group, open-label trial.
Setting: 78 sites in the United States and Canada.
Synopsis: From December 2012 to June 2017, 614 patients from 78 centers in the United States and Canada with symptomatic MR were enrolled with 302 patients assigned to the device group (transcatheter MVR and medical treatment) and 312 to the control group (medical therapy). Over 2 years, the device group’s annual rate for heart failure hospitalizations was significantly lower (35.8%/patient-year versus 67.9%/patient-year in the control group), as was all-cause mortality (29.1% for the device group versus 46.1%). The rate of freedom from device-related complications was 96.6%, better than the goal of 88%. There was improvement in quality of life, functional capacity, severity of MR, and left ventricular remodeling.
Limitations include that investigators were not blinded because the device was visible on imaging. Longer follow-up in the device group may have contributed to the observed decreased mortality. It is unknown whether less-symptomatic patients would attain the same benefit.
Bottom line: In patients with symptomatic, moderate to severe, and severe secondary MR, MVR lowers rates of hospitalization, decreases mortality, and improves quality of life.
Citation: Stone GW et al. Transcatheter mitral-valve repair in patients with heart failure. N Engl J Med. 2018 Sep 23. doi: 10.1056/NEJMoa1806640.
Dr. Kochar is an assistant professor of medicine in the division of hospital medicine at Mount Sinai Hospital, New York.
Background: In patients with primary degenerative MR, MVR is curative, with the transcatheter approach being safer than surgical repair. However, it is unknown whether patients with secondary MR from left ventricular dilatation would confer the same benefit of MVR.
Study design: Multicenter, randomized, controlled, parallel-group, open-label trial.
Setting: 78 sites in the United States and Canada.
Synopsis: From December 2012 to June 2017, 614 patients from 78 centers in the United States and Canada with symptomatic MR were enrolled with 302 patients assigned to the device group (transcatheter MVR and medical treatment) and 312 to the control group (medical therapy). Over 2 years, the device group’s annual rate for heart failure hospitalizations was significantly lower (35.8%/patient-year versus 67.9%/patient-year in the control group), as was all-cause mortality (29.1% for the device group versus 46.1%). The rate of freedom from device-related complications was 96.6%, better than the goal of 88%. There was improvement in quality of life, functional capacity, severity of MR, and left ventricular remodeling.
Limitations include that investigators were not blinded because the device was visible on imaging. Longer follow-up in the device group may have contributed to the observed decreased mortality. It is unknown whether less-symptomatic patients would attain the same benefit.
Bottom line: In patients with symptomatic, moderate to severe, and severe secondary MR, MVR lowers rates of hospitalization, decreases mortality, and improves quality of life.
Citation: Stone GW et al. Transcatheter mitral-valve repair in patients with heart failure. N Engl J Med. 2018 Sep 23. doi: 10.1056/NEJMoa1806640.
Dr. Kochar is an assistant professor of medicine in the division of hospital medicine at Mount Sinai Hospital, New York.
Daily aspirin use may not improve CV outcomes in healthy elderly
Clinical question: What are the benefits and risks of daily aspirin use for primary prevention in healthy elderly adults?
Background: Prior studies have shown the efficacy of aspirin for secondary prevention of cardiovascular disease and stroke, but the evidence supporting the use of aspirin for primary prevention is less certain.
Study design: Randomized, double-blind, placebo-controlled prospective study with a 5-year study period.
Setting: Australia and the United States.
Synopsis: The Aspirin in Reducing Events in the Elderly (ASPREE) trial included 19,114 community-dwelling healthy people (aged 70 years and older overall and aged 65 years and older if black or Hispanic), without cardiovascular disease, dementia or disability. The goal was to investigate the effect of daily low-dose aspirin (100 mg, enteric coated) on healthy life span (without dementia or disability), with prespecified secondary outcomes (cardiovascular events and major hemorrhage).
Analysis was by intention-to-treat. Participants were predominantly Caucasian, approximately 10% of patients had diabetes, 74% had hypertension, and 65% had dyslipidemia. There was high adherence to the intervention. There was no significant difference in the primary outcome (disability-free survival) or in the secondary outcome of cardiovascular event (fatal or nonfatal MI or stroke, or hospitalization for heart failure.) The rate of major hemorrhage (hemorrhagic stroke, symptomatic intracranial bleeding, clinically significant extracranial bleeding) was higher in the aspirin group (P less than .001). In contrast to prior studies, subgroup analysis showed higher mortality in the aspirin group (attributed to an increase in the risk of cancer-related death.) The authors warn that this finding should be interpreted with caution.
Bottom line: Aspirin use for primary prevention in healthy elderly persons over a 5-year period did not change disability-free survival, did not decrease cardiovascular risk, and increased the rate of major hemorrhage.
Citation: McNeil JJ et al. Effect of aspirin on all-cause mortality in the healthy elderly. N Engl J Med. 2018;379:1519-28.
Dr. Linker is an assistant professor of medicine in the division of hospital medicine at Mount Sinai Hospital, New York.
Clinical question: What are the benefits and risks of daily aspirin use for primary prevention in healthy elderly adults?
Background: Prior studies have shown the efficacy of aspirin for secondary prevention of cardiovascular disease and stroke, but the evidence supporting the use of aspirin for primary prevention is less certain.
Study design: Randomized, double-blind, placebo-controlled prospective study with a 5-year study period.
Setting: Australia and the United States.
Synopsis: The Aspirin in Reducing Events in the Elderly (ASPREE) trial included 19,114 community-dwelling healthy people (aged 70 years and older overall and aged 65 years and older if black or Hispanic), without cardiovascular disease, dementia or disability. The goal was to investigate the effect of daily low-dose aspirin (100 mg, enteric coated) on healthy life span (without dementia or disability), with prespecified secondary outcomes (cardiovascular events and major hemorrhage).
Analysis was by intention-to-treat. Participants were predominantly Caucasian, approximately 10% of patients had diabetes, 74% had hypertension, and 65% had dyslipidemia. There was high adherence to the intervention. There was no significant difference in the primary outcome (disability-free survival) or in the secondary outcome of cardiovascular event (fatal or nonfatal MI or stroke, or hospitalization for heart failure.) The rate of major hemorrhage (hemorrhagic stroke, symptomatic intracranial bleeding, clinically significant extracranial bleeding) was higher in the aspirin group (P less than .001). In contrast to prior studies, subgroup analysis showed higher mortality in the aspirin group (attributed to an increase in the risk of cancer-related death.) The authors warn that this finding should be interpreted with caution.
Bottom line: Aspirin use for primary prevention in healthy elderly persons over a 5-year period did not change disability-free survival, did not decrease cardiovascular risk, and increased the rate of major hemorrhage.
Citation: McNeil JJ et al. Effect of aspirin on all-cause mortality in the healthy elderly. N Engl J Med. 2018;379:1519-28.
Dr. Linker is an assistant professor of medicine in the division of hospital medicine at Mount Sinai Hospital, New York.
Clinical question: What are the benefits and risks of daily aspirin use for primary prevention in healthy elderly adults?
Background: Prior studies have shown the efficacy of aspirin for secondary prevention of cardiovascular disease and stroke, but the evidence supporting the use of aspirin for primary prevention is less certain.
Study design: Randomized, double-blind, placebo-controlled prospective study with a 5-year study period.
Setting: Australia and the United States.
Synopsis: The Aspirin in Reducing Events in the Elderly (ASPREE) trial included 19,114 community-dwelling healthy people (aged 70 years and older overall and aged 65 years and older if black or Hispanic), without cardiovascular disease, dementia or disability. The goal was to investigate the effect of daily low-dose aspirin (100 mg, enteric coated) on healthy life span (without dementia or disability), with prespecified secondary outcomes (cardiovascular events and major hemorrhage).
Analysis was by intention-to-treat. Participants were predominantly Caucasian, approximately 10% of patients had diabetes, 74% had hypertension, and 65% had dyslipidemia. There was high adherence to the intervention. There was no significant difference in the primary outcome (disability-free survival) or in the secondary outcome of cardiovascular event (fatal or nonfatal MI or stroke, or hospitalization for heart failure.) The rate of major hemorrhage (hemorrhagic stroke, symptomatic intracranial bleeding, clinically significant extracranial bleeding) was higher in the aspirin group (P less than .001). In contrast to prior studies, subgroup analysis showed higher mortality in the aspirin group (attributed to an increase in the risk of cancer-related death.) The authors warn that this finding should be interpreted with caution.
Bottom line: Aspirin use for primary prevention in healthy elderly persons over a 5-year period did not change disability-free survival, did not decrease cardiovascular risk, and increased the rate of major hemorrhage.
Citation: McNeil JJ et al. Effect of aspirin on all-cause mortality in the healthy elderly. N Engl J Med. 2018;379:1519-28.
Dr. Linker is an assistant professor of medicine in the division of hospital medicine at Mount Sinai Hospital, New York.
FDA issues warning on insulin pump cybersecurity weakness
The Food and Drug Administration has issued a warning to patients and health care providers that a pair of Medtronic insulin pumps are being recalled because of potential cybersecurity risks, according to a press release.
The affected devices are the MiniMed 508 and MiniMed Paradigm series insulin pumps, which wirelessly connect to both the patient’s blood glucose meter and continuous glucose monitoring system. A remote controller and CareLink USB – a thumb-sized wireless device that plugs into a computer – are used to operate the devices; the remote controller sends insulin dosing commands to the pump and the CareLink USB can be used to download and share data with the patient’s health care provider.
The potential risk involves the wireless communication between the pumps and related devices such as the blood glucose meter and remote controller. The FDA has identified a cybersecurity vulnerability within the insulin pumps, and is concerned that a third party could connect to the device and change the pump’s settings. Insulin could be given in excess, causing hypoglycemia, or stopped, causing hyperglycemia or diabetic ketoacidosis.
Medtronic has identified 4,000 patients in the United States who are affected by the security weakness. Because the company is unable to adequately update or patch the device to remove the weakness, the FDA is working to ensure that Medtronic addresses the issue in any way possible, including helping patients with affected pumps switch to newer models.
“While we are not aware of patients who may have been harmed by this particular cybersecurity vulnerability, the risk of patient harm if such a vulnerability were left unaddressed is significant. The safety communication issued today contains recommendations for what actions patients and health care providers should take to avoid the risk this vulnerability could pose,” said Suzanne Schwartz, MD, MBA, deputy director of the Office of Strategic Partnerships and Technology Innovation.
Find the full press release on the FDA website.
The Food and Drug Administration has issued a warning to patients and health care providers that a pair of Medtronic insulin pumps are being recalled because of potential cybersecurity risks, according to a press release.
The affected devices are the MiniMed 508 and MiniMed Paradigm series insulin pumps, which wirelessly connect to both the patient’s blood glucose meter and continuous glucose monitoring system. A remote controller and CareLink USB – a thumb-sized wireless device that plugs into a computer – are used to operate the devices; the remote controller sends insulin dosing commands to the pump and the CareLink USB can be used to download and share data with the patient’s health care provider.
The potential risk involves the wireless communication between the pumps and related devices such as the blood glucose meter and remote controller. The FDA has identified a cybersecurity vulnerability within the insulin pumps, and is concerned that a third party could connect to the device and change the pump’s settings. Insulin could be given in excess, causing hypoglycemia, or stopped, causing hyperglycemia or diabetic ketoacidosis.
Medtronic has identified 4,000 patients in the United States who are affected by the security weakness. Because the company is unable to adequately update or patch the device to remove the weakness, the FDA is working to ensure that Medtronic addresses the issue in any way possible, including helping patients with affected pumps switch to newer models.
“While we are not aware of patients who may have been harmed by this particular cybersecurity vulnerability, the risk of patient harm if such a vulnerability were left unaddressed is significant. The safety communication issued today contains recommendations for what actions patients and health care providers should take to avoid the risk this vulnerability could pose,” said Suzanne Schwartz, MD, MBA, deputy director of the Office of Strategic Partnerships and Technology Innovation.
Find the full press release on the FDA website.
The Food and Drug Administration has issued a warning to patients and health care providers that a pair of Medtronic insulin pumps are being recalled because of potential cybersecurity risks, according to a press release.
The affected devices are the MiniMed 508 and MiniMed Paradigm series insulin pumps, which wirelessly connect to both the patient’s blood glucose meter and continuous glucose monitoring system. A remote controller and CareLink USB – a thumb-sized wireless device that plugs into a computer – are used to operate the devices; the remote controller sends insulin dosing commands to the pump and the CareLink USB can be used to download and share data with the patient’s health care provider.
The potential risk involves the wireless communication between the pumps and related devices such as the blood glucose meter and remote controller. The FDA has identified a cybersecurity vulnerability within the insulin pumps, and is concerned that a third party could connect to the device and change the pump’s settings. Insulin could be given in excess, causing hypoglycemia, or stopped, causing hyperglycemia or diabetic ketoacidosis.
Medtronic has identified 4,000 patients in the United States who are affected by the security weakness. Because the company is unable to adequately update or patch the device to remove the weakness, the FDA is working to ensure that Medtronic addresses the issue in any way possible, including helping patients with affected pumps switch to newer models.
“While we are not aware of patients who may have been harmed by this particular cybersecurity vulnerability, the risk of patient harm if such a vulnerability were left unaddressed is significant. The safety communication issued today contains recommendations for what actions patients and health care providers should take to avoid the risk this vulnerability could pose,” said Suzanne Schwartz, MD, MBA, deputy director of the Office of Strategic Partnerships and Technology Innovation.
Find the full press release on the FDA website.
Short Takes
Restrictive transfusion strategy for cardiac surgery patients remains noninferior at 6 months post op
The authors previously reported that, in moderate- to high-risk cardiac surgery patients, a restrictive transfusion strategy was noninferior to a liberal strategy based on the clinical outcomes of all-cause mortality, MI, stroke, or new renal failure with dialysis. The groups continued to show no significant difference in outcomes at 6 months post op.
Citation: Mazer CD et al. Six-month outcomes after restrictive or liberal transfusion for cardiac surgery. N Engl J Med. 2018;379:1224-33.
Restrictive transfusion strategy for cardiac surgery patients remains noninferior at 6 months post op
The authors previously reported that, in moderate- to high-risk cardiac surgery patients, a restrictive transfusion strategy was noninferior to a liberal strategy based on the clinical outcomes of all-cause mortality, MI, stroke, or new renal failure with dialysis. The groups continued to show no significant difference in outcomes at 6 months post op.
Citation: Mazer CD et al. Six-month outcomes after restrictive or liberal transfusion for cardiac surgery. N Engl J Med. 2018;379:1224-33.
Restrictive transfusion strategy for cardiac surgery patients remains noninferior at 6 months post op
The authors previously reported that, in moderate- to high-risk cardiac surgery patients, a restrictive transfusion strategy was noninferior to a liberal strategy based on the clinical outcomes of all-cause mortality, MI, stroke, or new renal failure with dialysis. The groups continued to show no significant difference in outcomes at 6 months post op.
Citation: Mazer CD et al. Six-month outcomes after restrictive or liberal transfusion for cardiac surgery. N Engl J Med. 2018;379:1224-33.
Subset of patients benefits from in-hospital sleep apnea screening
SAN ANTONIO – In the clinical opinion of Richard J. Schwab, MD,
“Many diseases are adversely affected by sleep apnea, including myocardial infarction, hypertension, a cerebrovascular accident, pulmonary hypertension, atrial fibrillation, diabetes, and congestive heart failure,” Dr. Schwab, interim chief of the University of Pennsylvania Perelman School of Medicine’s Division of Sleep Medicine, said at the annual meeting of the Associated Professional Sleep Societies.
“Continuous positive airway pressure [CPAP] may help heart failure patients and reduce 30-day readmission rates, which has important financial implications in the University of Pennsylvania Health system. CPAP may also decrease the rapid responses and cardiac arrests at night,” he said.
A few years ago, Dr. Schwab and his associates set out to determine whether PAP adherence in cardiac patients with sleep-disordered breathing reduced readmission rates 30 days after discharge (J Clin Sleep Med. 2014;10:1051-59). They evaluated 104 consecutive cardiovascular hospitalized patients reporting symptoms of sleep-disordered breathing (SDB) between January of 2012 and March of 2013, and collected demographic data, SDB type, PAP adherence, and data regarding 30-day hospital readmission/ED visits. Apnea was scored when there was a 90% or greater cessation of airflow detected through the nasal pressure sensor. Hypopnea was scored when there was at least a 50% reduction in airflow with an associated 3% or greater oxyhemoglobin desaturation. Central apnea (CSA) was scored when there was a 90% or greater cessation of airflow detected through the nasal pressure sensor and no effort in the thorax and abdomen. If more than 50% of the apneas were central, the SDB was classified as CSA. If more than 50% of apneas were obstructive in nature, it was considered obstructive sleep apnea (OSA).
The mean age of the patients was 59 years, 63% were male, their mean body mass index was 34 kg/m2, 87% had heart failure, and 82% had hypertension. Of the 104 patients, 81 had SDB and 23 did not. The 30-day readmission rate was 29% in patients who did not use PAP, 30% in partial users, and 0% in full users (P = .0246).
The researchers found that 81 patients (78%) had sleep disordered breathing. Of these, 65 (80%) had OSA while 16 (20%) had CSA. The study demonstrated that performing inpatient sleep studies was feasible. “Our study indicated that SDB is common in hospitalized cardiac patients, with the majority of patients manifesting OSA,” said Dr. Schwab, medical director of the Penn Sleep Centers. “The data suggest that hospital readmission and ED visits 30 days after discharge were significantly lower in patients with cardiac disease and SDB who adhere to PAP treatment than those who are not adherent.”
Dr. Schwab is part of a research team conducting a longer study with ResMed to examine 30-, 60-, and 90-day readmission rates in cardiac inpatients newly diagnosed with OSA and started on auto-PAP (APAP). They plan to evaluate the ejection fraction during hospitalization and in follow-up, as well as the effect of an in-laboratory sleep study at 1 month. The long-term follow-up is planned for 3 years.
Launching an inpatient sleep apnea consult service in the hospital makes sense, Dr. Schwab continued, because home sleep studies are approved for the diagnosis of sleep apnea, APAP can determine optimal CPAP settings, insurance will cover CPAP with a home or inpatient sleep study, and patients can get CPAP/APAP at or before discharge. “Sleep techs or respiratory therapists can perform these sleep studies,” he said. At Penn, a nurse practitioner (NP) runs this service using the Alice NightOne home sleep testing device and the WatchPAT portable sleep apnea diagnostic device.
The notion of performing in-hospital sleep studies should be an easy sell to cardiologists and hospital administrators, Dr. Schwab said, because the program will decrease hospital readmissions, “which is going to save the hospital a lot of money. In addition, these patients can come back for in-laboratory sleep studies. There is also increased revenue from the consults and progress notes, and the professional fee for sleep study interpretation. The most challenging part of the inpatient sleep consult service is trying to get these patients to follow up in the sleep center with the NP.”
Dr. Schwab is an investigator for the recently launched Penn Medicine Nudge Unit Project, which is funded by the National Institutes of Health. The project includes a multidisciplinary team of providers from the Hospital of the University of Pennsylvania, Penn Presbyterian Medical Center, and Penn Medicine Risk Management. If an inpatient has a BMI of 35 kg/m2 or greater, the clinician will be “nudged” via an enterprise messaging system (EMS) prompt to order an inpatient sleep oximetry. “They have to respond to that nudge,” Dr. Schwab said. “If the oximetry is consistent for sleep apnea, there will be another nudge to consult with the sleep medicine team. If the oximetry is negative, they will be nudged to get an outpatient consult with the sleep medicine team.” For patients undergoing preadmission testing for any type of surgery who score 4 or more on the STOP-Bang questionnaire (Chest 2016;149:631-38), the clinician is “nudged” to order an outpatient sleep consultation.
Benefits to such an approach, he said, include a decrease in resource allocation, shorter hospital stays, patient perceived improvement in quality of sleep, improved patient survey scores, and the fact that apnea treatment may decrease the need for rapid response. “It also reduces medical-legal concerns, improves patient outcomes, decreases readmissions, and generates revenue from inpatient and outpatient sleep studies,” Dr. Schwab said. Barriers to such an approach include the fact that there is no defined pathway at many institutions for recognizing and referring suspected OSA patients. “There is often a lack of care coordination between primary providers and sleep medicine, and sleep is viewed as ambulatory care, not as a part of inpatient care,” he said.
Last year, Dr. Schwab and his colleagues at UPenn conducted a pilot study to develop and test a pathway for identifying OSA in high-risk inpatient and preadmission patient populations. Of 389 patients admitted between Aug. 20 and Sept. 20 of 2018, 43 had a BMI of 35 kg/m2 or greater. Of these, 10 were screened with oximetry and 8 were positive for severe apnea. Of these eight cases, five inpatient consults were ordered, one outpatient consult was ordered, one patient had no consult ordered, and one patient was discharged before the consult was ordered.
Dr. Schwab also performed a pilot study in patients undergoing preoperative testing with the STOP-Bang questionnaire. “When we piloted this, there were over 200 patients who could have been sent to the outpatient sleep consult service, and we referred none,” Dr. Schwab said. “We are just starting to implement a program to screen them. We can treat these people for their sleep apnea and prevent chronic adverse sequelae associated with this disease.”
Both the inpatient and outpatient screening programs for sleep apnea are built within their electronic medical record. “Building this within your EMR requires effort, but it’s doable,” he said.
Dr. Schwab disclosed that he has received grants from the National Institutes of Health, ResMed, and Inspire Medical Systems.
SAN ANTONIO – In the clinical opinion of Richard J. Schwab, MD,
“Many diseases are adversely affected by sleep apnea, including myocardial infarction, hypertension, a cerebrovascular accident, pulmonary hypertension, atrial fibrillation, diabetes, and congestive heart failure,” Dr. Schwab, interim chief of the University of Pennsylvania Perelman School of Medicine’s Division of Sleep Medicine, said at the annual meeting of the Associated Professional Sleep Societies.
“Continuous positive airway pressure [CPAP] may help heart failure patients and reduce 30-day readmission rates, which has important financial implications in the University of Pennsylvania Health system. CPAP may also decrease the rapid responses and cardiac arrests at night,” he said.
A few years ago, Dr. Schwab and his associates set out to determine whether PAP adherence in cardiac patients with sleep-disordered breathing reduced readmission rates 30 days after discharge (J Clin Sleep Med. 2014;10:1051-59). They evaluated 104 consecutive cardiovascular hospitalized patients reporting symptoms of sleep-disordered breathing (SDB) between January of 2012 and March of 2013, and collected demographic data, SDB type, PAP adherence, and data regarding 30-day hospital readmission/ED visits. Apnea was scored when there was a 90% or greater cessation of airflow detected through the nasal pressure sensor. Hypopnea was scored when there was at least a 50% reduction in airflow with an associated 3% or greater oxyhemoglobin desaturation. Central apnea (CSA) was scored when there was a 90% or greater cessation of airflow detected through the nasal pressure sensor and no effort in the thorax and abdomen. If more than 50% of the apneas were central, the SDB was classified as CSA. If more than 50% of apneas were obstructive in nature, it was considered obstructive sleep apnea (OSA).
The mean age of the patients was 59 years, 63% were male, their mean body mass index was 34 kg/m2, 87% had heart failure, and 82% had hypertension. Of the 104 patients, 81 had SDB and 23 did not. The 30-day readmission rate was 29% in patients who did not use PAP, 30% in partial users, and 0% in full users (P = .0246).
The researchers found that 81 patients (78%) had sleep disordered breathing. Of these, 65 (80%) had OSA while 16 (20%) had CSA. The study demonstrated that performing inpatient sleep studies was feasible. “Our study indicated that SDB is common in hospitalized cardiac patients, with the majority of patients manifesting OSA,” said Dr. Schwab, medical director of the Penn Sleep Centers. “The data suggest that hospital readmission and ED visits 30 days after discharge were significantly lower in patients with cardiac disease and SDB who adhere to PAP treatment than those who are not adherent.”
Dr. Schwab is part of a research team conducting a longer study with ResMed to examine 30-, 60-, and 90-day readmission rates in cardiac inpatients newly diagnosed with OSA and started on auto-PAP (APAP). They plan to evaluate the ejection fraction during hospitalization and in follow-up, as well as the effect of an in-laboratory sleep study at 1 month. The long-term follow-up is planned for 3 years.
Launching an inpatient sleep apnea consult service in the hospital makes sense, Dr. Schwab continued, because home sleep studies are approved for the diagnosis of sleep apnea, APAP can determine optimal CPAP settings, insurance will cover CPAP with a home or inpatient sleep study, and patients can get CPAP/APAP at or before discharge. “Sleep techs or respiratory therapists can perform these sleep studies,” he said. At Penn, a nurse practitioner (NP) runs this service using the Alice NightOne home sleep testing device and the WatchPAT portable sleep apnea diagnostic device.
The notion of performing in-hospital sleep studies should be an easy sell to cardiologists and hospital administrators, Dr. Schwab said, because the program will decrease hospital readmissions, “which is going to save the hospital a lot of money. In addition, these patients can come back for in-laboratory sleep studies. There is also increased revenue from the consults and progress notes, and the professional fee for sleep study interpretation. The most challenging part of the inpatient sleep consult service is trying to get these patients to follow up in the sleep center with the NP.”
Dr. Schwab is an investigator for the recently launched Penn Medicine Nudge Unit Project, which is funded by the National Institutes of Health. The project includes a multidisciplinary team of providers from the Hospital of the University of Pennsylvania, Penn Presbyterian Medical Center, and Penn Medicine Risk Management. If an inpatient has a BMI of 35 kg/m2 or greater, the clinician will be “nudged” via an enterprise messaging system (EMS) prompt to order an inpatient sleep oximetry. “They have to respond to that nudge,” Dr. Schwab said. “If the oximetry is consistent for sleep apnea, there will be another nudge to consult with the sleep medicine team. If the oximetry is negative, they will be nudged to get an outpatient consult with the sleep medicine team.” For patients undergoing preadmission testing for any type of surgery who score 4 or more on the STOP-Bang questionnaire (Chest 2016;149:631-38), the clinician is “nudged” to order an outpatient sleep consultation.
Benefits to such an approach, he said, include a decrease in resource allocation, shorter hospital stays, patient perceived improvement in quality of sleep, improved patient survey scores, and the fact that apnea treatment may decrease the need for rapid response. “It also reduces medical-legal concerns, improves patient outcomes, decreases readmissions, and generates revenue from inpatient and outpatient sleep studies,” Dr. Schwab said. Barriers to such an approach include the fact that there is no defined pathway at many institutions for recognizing and referring suspected OSA patients. “There is often a lack of care coordination between primary providers and sleep medicine, and sleep is viewed as ambulatory care, not as a part of inpatient care,” he said.
Last year, Dr. Schwab and his colleagues at UPenn conducted a pilot study to develop and test a pathway for identifying OSA in high-risk inpatient and preadmission patient populations. Of 389 patients admitted between Aug. 20 and Sept. 20 of 2018, 43 had a BMI of 35 kg/m2 or greater. Of these, 10 were screened with oximetry and 8 were positive for severe apnea. Of these eight cases, five inpatient consults were ordered, one outpatient consult was ordered, one patient had no consult ordered, and one patient was discharged before the consult was ordered.
Dr. Schwab also performed a pilot study in patients undergoing preoperative testing with the STOP-Bang questionnaire. “When we piloted this, there were over 200 patients who could have been sent to the outpatient sleep consult service, and we referred none,” Dr. Schwab said. “We are just starting to implement a program to screen them. We can treat these people for their sleep apnea and prevent chronic adverse sequelae associated with this disease.”
Both the inpatient and outpatient screening programs for sleep apnea are built within their electronic medical record. “Building this within your EMR requires effort, but it’s doable,” he said.
Dr. Schwab disclosed that he has received grants from the National Institutes of Health, ResMed, and Inspire Medical Systems.
SAN ANTONIO – In the clinical opinion of Richard J. Schwab, MD,
“Many diseases are adversely affected by sleep apnea, including myocardial infarction, hypertension, a cerebrovascular accident, pulmonary hypertension, atrial fibrillation, diabetes, and congestive heart failure,” Dr. Schwab, interim chief of the University of Pennsylvania Perelman School of Medicine’s Division of Sleep Medicine, said at the annual meeting of the Associated Professional Sleep Societies.
“Continuous positive airway pressure [CPAP] may help heart failure patients and reduce 30-day readmission rates, which has important financial implications in the University of Pennsylvania Health system. CPAP may also decrease the rapid responses and cardiac arrests at night,” he said.
A few years ago, Dr. Schwab and his associates set out to determine whether PAP adherence in cardiac patients with sleep-disordered breathing reduced readmission rates 30 days after discharge (J Clin Sleep Med. 2014;10:1051-59). They evaluated 104 consecutive cardiovascular hospitalized patients reporting symptoms of sleep-disordered breathing (SDB) between January of 2012 and March of 2013, and collected demographic data, SDB type, PAP adherence, and data regarding 30-day hospital readmission/ED visits. Apnea was scored when there was a 90% or greater cessation of airflow detected through the nasal pressure sensor. Hypopnea was scored when there was at least a 50% reduction in airflow with an associated 3% or greater oxyhemoglobin desaturation. Central apnea (CSA) was scored when there was a 90% or greater cessation of airflow detected through the nasal pressure sensor and no effort in the thorax and abdomen. If more than 50% of the apneas were central, the SDB was classified as CSA. If more than 50% of apneas were obstructive in nature, it was considered obstructive sleep apnea (OSA).
The mean age of the patients was 59 years, 63% were male, their mean body mass index was 34 kg/m2, 87% had heart failure, and 82% had hypertension. Of the 104 patients, 81 had SDB and 23 did not. The 30-day readmission rate was 29% in patients who did not use PAP, 30% in partial users, and 0% in full users (P = .0246).
The researchers found that 81 patients (78%) had sleep disordered breathing. Of these, 65 (80%) had OSA while 16 (20%) had CSA. The study demonstrated that performing inpatient sleep studies was feasible. “Our study indicated that SDB is common in hospitalized cardiac patients, with the majority of patients manifesting OSA,” said Dr. Schwab, medical director of the Penn Sleep Centers. “The data suggest that hospital readmission and ED visits 30 days after discharge were significantly lower in patients with cardiac disease and SDB who adhere to PAP treatment than those who are not adherent.”
Dr. Schwab is part of a research team conducting a longer study with ResMed to examine 30-, 60-, and 90-day readmission rates in cardiac inpatients newly diagnosed with OSA and started on auto-PAP (APAP). They plan to evaluate the ejection fraction during hospitalization and in follow-up, as well as the effect of an in-laboratory sleep study at 1 month. The long-term follow-up is planned for 3 years.
Launching an inpatient sleep apnea consult service in the hospital makes sense, Dr. Schwab continued, because home sleep studies are approved for the diagnosis of sleep apnea, APAP can determine optimal CPAP settings, insurance will cover CPAP with a home or inpatient sleep study, and patients can get CPAP/APAP at or before discharge. “Sleep techs or respiratory therapists can perform these sleep studies,” he said. At Penn, a nurse practitioner (NP) runs this service using the Alice NightOne home sleep testing device and the WatchPAT portable sleep apnea diagnostic device.
The notion of performing in-hospital sleep studies should be an easy sell to cardiologists and hospital administrators, Dr. Schwab said, because the program will decrease hospital readmissions, “which is going to save the hospital a lot of money. In addition, these patients can come back for in-laboratory sleep studies. There is also increased revenue from the consults and progress notes, and the professional fee for sleep study interpretation. The most challenging part of the inpatient sleep consult service is trying to get these patients to follow up in the sleep center with the NP.”
Dr. Schwab is an investigator for the recently launched Penn Medicine Nudge Unit Project, which is funded by the National Institutes of Health. The project includes a multidisciplinary team of providers from the Hospital of the University of Pennsylvania, Penn Presbyterian Medical Center, and Penn Medicine Risk Management. If an inpatient has a BMI of 35 kg/m2 or greater, the clinician will be “nudged” via an enterprise messaging system (EMS) prompt to order an inpatient sleep oximetry. “They have to respond to that nudge,” Dr. Schwab said. “If the oximetry is consistent for sleep apnea, there will be another nudge to consult with the sleep medicine team. If the oximetry is negative, they will be nudged to get an outpatient consult with the sleep medicine team.” For patients undergoing preadmission testing for any type of surgery who score 4 or more on the STOP-Bang questionnaire (Chest 2016;149:631-38), the clinician is “nudged” to order an outpatient sleep consultation.
Benefits to such an approach, he said, include a decrease in resource allocation, shorter hospital stays, patient perceived improvement in quality of sleep, improved patient survey scores, and the fact that apnea treatment may decrease the need for rapid response. “It also reduces medical-legal concerns, improves patient outcomes, decreases readmissions, and generates revenue from inpatient and outpatient sleep studies,” Dr. Schwab said. Barriers to such an approach include the fact that there is no defined pathway at many institutions for recognizing and referring suspected OSA patients. “There is often a lack of care coordination between primary providers and sleep medicine, and sleep is viewed as ambulatory care, not as a part of inpatient care,” he said.
Last year, Dr. Schwab and his colleagues at UPenn conducted a pilot study to develop and test a pathway for identifying OSA in high-risk inpatient and preadmission patient populations. Of 389 patients admitted between Aug. 20 and Sept. 20 of 2018, 43 had a BMI of 35 kg/m2 or greater. Of these, 10 were screened with oximetry and 8 were positive for severe apnea. Of these eight cases, five inpatient consults were ordered, one outpatient consult was ordered, one patient had no consult ordered, and one patient was discharged before the consult was ordered.
Dr. Schwab also performed a pilot study in patients undergoing preoperative testing with the STOP-Bang questionnaire. “When we piloted this, there were over 200 patients who could have been sent to the outpatient sleep consult service, and we referred none,” Dr. Schwab said. “We are just starting to implement a program to screen them. We can treat these people for their sleep apnea and prevent chronic adverse sequelae associated with this disease.”
Both the inpatient and outpatient screening programs for sleep apnea are built within their electronic medical record. “Building this within your EMR requires effort, but it’s doable,” he said.
Dr. Schwab disclosed that he has received grants from the National Institutes of Health, ResMed, and Inspire Medical Systems.
EXPERT ANALYSIS FROM SLEEP 2019