User login
ID Practitioner is an independent news source that provides infectious disease specialists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the infectious disease specialist’s practice. Specialty focus topics include antimicrobial resistance, emerging infections, global ID, hepatitis, HIV, hospital-acquired infections, immunizations and vaccines, influenza, mycoses, pediatric infections, and STIs. Infectious Diseases News is owned by Frontline Medical Communications.
sofosbuvir
ritonavir with dasabuvir
discount
support path
program
ritonavir
greedy
ledipasvir
assistance
viekira pak
vpak
advocacy
needy
protest
abbvie
paritaprevir
ombitasvir
direct-acting antivirals
dasabuvir
gilead
fake-ovir
support
v pak
oasis
harvoni
section[contains(@class, 'footer-nav-section-wrapper')]
div[contains(@class, 'pane-pub-article-idp')]
div[contains(@class, 'pane-medstat-latest-articles-articles-section')]
div[contains(@class, 'pane-pub-home-idp')]
div[contains(@class, 'pane-pub-topic-idp')]
Hospitalist well-being during the pandemic
Navigating COVID-19 requires self-care
The global COVID-19 pandemic has escalated everyone’s stress levels, especially clinicians caring for hospitalized patients. New pressures have added to everyday stress, new studies have revised prior patient care recommendations, and the world generally seems upside down. What can a busy hospitalist do to maintain a modicum of sanity in all the craziness?
The stressors facing hospitalists
Uncertainty
Of all the burdens COVID-19 has unleashed, the biggest may be uncertainty. Not only is there unease about the virus itself, there also is legitimate concern about the future of medicine, said Elizabeth Harry, MD, SFHM, a hospitalist and senior director of clinical affairs at the University of Colorado Hospital in Aurora.
“What does it look like after an event like this, particularly in areas like academic medicine and teaching our next generation and getting funding for research? And how do we continue to produce physicians that can provide excellent care?” she asked.
There is also uncertainty in the best way to care for patients, said Eileen Barrett, MD, MPH, SFHM, a hospitalist at the University of New Mexico, Albuquerque.
“There are some models that are emerging to predict who will have a worse outcome, but they’re still not great models, so we have uncertainty for a given patient.” And, she noted, as the science continues to evolve, there exists a constant worry that “you might have inadvertently caused someone harm.”
The financial implications of the pandemic are creating uncertainty too. “When you fund a health care system with elective procedures and you can’t do those, and instead have to shift to the most essential services, a lot of places are seeing a massive deficit, which is going to affect staff morale and some physician offices are going to close,” said Elisabeth Poorman, MD, MPH, a primary care and internal medicine physician and chair of the King County Medical Society Physician Wellness Committee in Seattle.
Fear
When the pandemic began in the United States, “fear of the unknown was perhaps the scariest part, particularly as it pertained to personal protective equipment,” said Mark Rudolph, MD, SFHM, chief experience officer and vice president of patient experience and physician development at Sound Physicians in Tacoma, Wash. “For most clinicians, this is the first time that they are themselves in harm’s way while they do their jobs. And worse, they risk bringing the virus home to their families. That is the concern I hear most.”
Anxiety
Worrying about being able to provide excellent patient care is a big stressor, especially since this is the heart and soul of why most hospitalists have gone into their line of work.
“Part of providing excellent care to your patients is providing excellent supportive care to their families,” Dr. Harry said. “There’s some dissonance there in not being able to allow the family to come visit, but wanting to keep them safe, and it feels really hard to support your patients and support their families in the best way. It can feel like you’re just watching and waiting to see what will happen, and that we don’t have a lot of agency over which direction things take.”
There is concern for health care team members as well, Dr. Harry added. “Physicians care a lot about their teams and how they’re doing. I think there’s a sense of esprit de corps among folks and worry for each other there.”
Guilt
Although you may be at the hospital all day, you may feel guilty when you are not providing direct patient care. Or maybe you or someone on your team has an immunodeficiency and can’t be on the front line. Perhaps one of your team members contracted COVID-19 and you did not. Whatever the case, guilt is another emotion that is rampant among hospitalists right now, Dr. Barrett said.
Burnout
Unfortunately, burnout is a potential reality in times of high stress. “Burnout is dynamic,” said Dr. Poorman. “It’s a process by which your emotional and cognitive reserves are exhausted. The people with the highest burnout are the ones who are still trying to provide the standard of care, or above the standard of care in dysfunctional systems.”
Dr. Harry noted that burnout presents in different ways for different people, but Dr. Rudolph added that it’s crucial for hospitalist team members to watch for signs of burnout so they can intervene and/or get help for their colleagues.
Warning signs in yourself or others that burnout could be on the horizon include:
- Fatigue/exhaustion – Whether emotional or physical (or both), this can become a problem if it “just doesn’t seem to go away despite rest and time away from work,” said Dr. Rudolph.
- Behavioral changes – Any behavior that’s out of the ordinary may be a red flag, like lashing out at someone at work.
- Overwork – Working too much can be caused by an inability to let go of patient care, Dr. Barrett said.
- Not working enough – This may include avoiding tasks and having difficulty meeting deadlines.
- Maladaptive coping behaviors – Excessive consumption of alcohol or drugs is a common coping mechanism. “Even excessive consumption of news is something that people are using to numb out a little bit,” said Dr. Harry.
- Depersonalization – “This is where you start to look at patients, colleagues, or administrators as ‘them’ and you can’t connect as deeply,” Dr. Harry said. “Part of that’s protective and a normal thing to do during a big trauma like this, but it’s also incredibly distancing. Any language that people start using that feels like ‘us’ or ‘them’ is a warning sign.”
- Disengagement – Many people disengage from their work, but Dr. Poorman said physicians tend to disengage from other parts of their lives, such as exercise and family interaction.
Protecting yourself while supporting others
Like the illustration of putting the oxygen mask on yourself first so you can help others, it’s important to protect your own mental and physical health as you support your fellow physicians. Here’s what the experts suggest.
Focus on basic needs
“When you’re in the midst of a trauma, which we are, you don’t want to open all of that up and go to the depths of your thoughts about the grief of all of it because it can actually make the trauma worse,” said Dr. Harry. “There’s a lot of literature that debriefing is really helpful after the event, but if you do it during the event, it can be really dangerous.”
Instead, she said, the goal should be focusing on your basic needs and what you need to do to get through each day, like keeping you and your family in good health. “What is your purpose? Staying connected to why you do this and staying focused on the present is really important,” Dr. Harry noted.
Do your best to get a good night’s sleep, exercise as much as you can, talk to others, and see a mental health provider if your anxiety is too high, advises Dr. Barrett. “Even avoiding blue light from phones and screens within 2 hours of bedtime, parking further away from the hospital and walking, and taking the stairs are things that add up in a big way.”
Keep up your normal routine
“Right now, it’s really critical for clinicians to keep up components of their routine that feel ‘normal,’ ” Dr. Rudolph said. “Whether it’s exercise, playing board games with their kids, or spending time on a hobby, it’s critical to allow yourself these comfortable, predictable, and rewarding detours.”
Set limits
People under stress tend to find unhealthy ways to cope. Instead, try being intentional about what you are consuming by putting limits on things like your news, alcohol consumption, and the number of hours you work, said Dr. Harry.
Implement a culture of wellness
Dr. Barrett believes in creating the work culture we want to be in, one that ensures people have psychological safety, allows them to ask for help, encourages them to disconnect completely from work, and makes them feel valued and listened to. She likes the example of “the pause,” which is called by a team member right after a patient expires.
“It’s a 30-second moment of silence where we reflect on the patient, their loved ones, and every member of the health care team who helped support and treat them,” said Dr. Barrett. “At the conclusion, you say: ‘Thank you. Is there anything you need to be able to go back to the care of other patients?’ Because it’s unnatural to have this terrible thing that happened and then just act like nothing happened.”
Target resources
Be proactive and know where to find resources before you need them, advised Dr. Harry. “Most institutions have free mental health resources, either through their employee assistance programs or HR, plus there’s lots of national organizations that are offering free resources to health care providers.”
Focus on what you can control
Separating what is under your control from what is not is a struggle for everyone, Dr. Poorman said, but it’s helpful to think about the ways you can have an impact and what you’re able to control.
“There was a woman who was diagnosed with early-onset Parkinson’s that I heard giving an interview at the beginning of this pandemic,” she said. “It was the most helpful advice I got, which was: ‘Think of the next good thing you can do.’ You can’t fix everything, so what’s the next good thing you can do?”
Maintain connectivity
Make sure you are utilizing your support circle and staying connected. “That sense of connection is incredibly protective on multiple fronts for depression, for burnout, for suicide ideation, etc.,” Dr. Harry said.
“It doesn’t matter if it’s your teammates at work, your family at home, your best friend from medical school – whomever you can debrief with, vent with, and just share your thoughts and feelings with, these outlets are critical for all of us to process our emotions and diffuse stress and anxiety,” said Dr. Rudolph.
Dr. Poorman is concerned that there could be a spike in physician suicides caused by increased stress, so she also encourages talking openly about what is going on and about getting help when it’s necessary. “Many of us are afraid to seek care because we can actually have our ability to practice medicine questioned, but now is not the time for heroes. Now is the time for people who are willing to recognize their own strengths and limitations to take care of one another.”
Be compassionate toward others
Keep in mind that everyone is stressed out and offer empathy and compassion. “I think everybody’s struggling to try to figure this out and the more that we can give each other the benefit of the doubt and a little grace, the more protective that is,” said Dr. Harry.
Listening is meaningful too. “Recognizing opportunities to validate and acknowledge the feelings that are being shared with you by your colleagues is critical,” Dr. Rudolph said. “We all need to know that we’re not alone, that our thoughts and feelings are okay, and when we share a difficult story, the value of someone saying something as simple as, ‘wow, that sounds like it was really hard,’ is immense.”
Be compassionate toward yourself
Try to give yourself a break and be as compassionate with yourself as you would with others. It’s okay that you’re not getting in shape, publishing prolifically, or redesigning your house right now.
“There’s a lot of data linking lack of self-compassion to burnout,” said Dr. Harry. She says there are courses on self-compassion available that help you work on being kinder to yourself.
Get a “battle buddy”
The American Medical Association has a free “buddy system” program called PeerRx to help physicians cope during the pandemic. Dr. Rudolph said that now is a great time to use this military-developed intervention in which each team member checks in with a chosen partner at agreed-upon intervals.
For example, “You can tell that person: ‘If I don’t call my family for a week that’s a red flag for me.’ And then you hold each other accountable to those things,” Dr. Harry said.
The buddy system is another way to harness that sense of connection that is so vital to our health and well-being.
“The simple act of showing that you care … can make all the difference when you’re doing this kind of work that is both challenging and dangerous,” said Dr. Rudolph.
Navigating COVID-19 requires self-care
Navigating COVID-19 requires self-care
The global COVID-19 pandemic has escalated everyone’s stress levels, especially clinicians caring for hospitalized patients. New pressures have added to everyday stress, new studies have revised prior patient care recommendations, and the world generally seems upside down. What can a busy hospitalist do to maintain a modicum of sanity in all the craziness?
The stressors facing hospitalists
Uncertainty
Of all the burdens COVID-19 has unleashed, the biggest may be uncertainty. Not only is there unease about the virus itself, there also is legitimate concern about the future of medicine, said Elizabeth Harry, MD, SFHM, a hospitalist and senior director of clinical affairs at the University of Colorado Hospital in Aurora.
“What does it look like after an event like this, particularly in areas like academic medicine and teaching our next generation and getting funding for research? And how do we continue to produce physicians that can provide excellent care?” she asked.
There is also uncertainty in the best way to care for patients, said Eileen Barrett, MD, MPH, SFHM, a hospitalist at the University of New Mexico, Albuquerque.
“There are some models that are emerging to predict who will have a worse outcome, but they’re still not great models, so we have uncertainty for a given patient.” And, she noted, as the science continues to evolve, there exists a constant worry that “you might have inadvertently caused someone harm.”
The financial implications of the pandemic are creating uncertainty too. “When you fund a health care system with elective procedures and you can’t do those, and instead have to shift to the most essential services, a lot of places are seeing a massive deficit, which is going to affect staff morale and some physician offices are going to close,” said Elisabeth Poorman, MD, MPH, a primary care and internal medicine physician and chair of the King County Medical Society Physician Wellness Committee in Seattle.
Fear
When the pandemic began in the United States, “fear of the unknown was perhaps the scariest part, particularly as it pertained to personal protective equipment,” said Mark Rudolph, MD, SFHM, chief experience officer and vice president of patient experience and physician development at Sound Physicians in Tacoma, Wash. “For most clinicians, this is the first time that they are themselves in harm’s way while they do their jobs. And worse, they risk bringing the virus home to their families. That is the concern I hear most.”
Anxiety
Worrying about being able to provide excellent patient care is a big stressor, especially since this is the heart and soul of why most hospitalists have gone into their line of work.
“Part of providing excellent care to your patients is providing excellent supportive care to their families,” Dr. Harry said. “There’s some dissonance there in not being able to allow the family to come visit, but wanting to keep them safe, and it feels really hard to support your patients and support their families in the best way. It can feel like you’re just watching and waiting to see what will happen, and that we don’t have a lot of agency over which direction things take.”
There is concern for health care team members as well, Dr. Harry added. “Physicians care a lot about their teams and how they’re doing. I think there’s a sense of esprit de corps among folks and worry for each other there.”
Guilt
Although you may be at the hospital all day, you may feel guilty when you are not providing direct patient care. Or maybe you or someone on your team has an immunodeficiency and can’t be on the front line. Perhaps one of your team members contracted COVID-19 and you did not. Whatever the case, guilt is another emotion that is rampant among hospitalists right now, Dr. Barrett said.
Burnout
Unfortunately, burnout is a potential reality in times of high stress. “Burnout is dynamic,” said Dr. Poorman. “It’s a process by which your emotional and cognitive reserves are exhausted. The people with the highest burnout are the ones who are still trying to provide the standard of care, or above the standard of care in dysfunctional systems.”
Dr. Harry noted that burnout presents in different ways for different people, but Dr. Rudolph added that it’s crucial for hospitalist team members to watch for signs of burnout so they can intervene and/or get help for their colleagues.
Warning signs in yourself or others that burnout could be on the horizon include:
- Fatigue/exhaustion – Whether emotional or physical (or both), this can become a problem if it “just doesn’t seem to go away despite rest and time away from work,” said Dr. Rudolph.
- Behavioral changes – Any behavior that’s out of the ordinary may be a red flag, like lashing out at someone at work.
- Overwork – Working too much can be caused by an inability to let go of patient care, Dr. Barrett said.
- Not working enough – This may include avoiding tasks and having difficulty meeting deadlines.
- Maladaptive coping behaviors – Excessive consumption of alcohol or drugs is a common coping mechanism. “Even excessive consumption of news is something that people are using to numb out a little bit,” said Dr. Harry.
- Depersonalization – “This is where you start to look at patients, colleagues, or administrators as ‘them’ and you can’t connect as deeply,” Dr. Harry said. “Part of that’s protective and a normal thing to do during a big trauma like this, but it’s also incredibly distancing. Any language that people start using that feels like ‘us’ or ‘them’ is a warning sign.”
- Disengagement – Many people disengage from their work, but Dr. Poorman said physicians tend to disengage from other parts of their lives, such as exercise and family interaction.
Protecting yourself while supporting others
Like the illustration of putting the oxygen mask on yourself first so you can help others, it’s important to protect your own mental and physical health as you support your fellow physicians. Here’s what the experts suggest.
Focus on basic needs
“When you’re in the midst of a trauma, which we are, you don’t want to open all of that up and go to the depths of your thoughts about the grief of all of it because it can actually make the trauma worse,” said Dr. Harry. “There’s a lot of literature that debriefing is really helpful after the event, but if you do it during the event, it can be really dangerous.”
Instead, she said, the goal should be focusing on your basic needs and what you need to do to get through each day, like keeping you and your family in good health. “What is your purpose? Staying connected to why you do this and staying focused on the present is really important,” Dr. Harry noted.
Do your best to get a good night’s sleep, exercise as much as you can, talk to others, and see a mental health provider if your anxiety is too high, advises Dr. Barrett. “Even avoiding blue light from phones and screens within 2 hours of bedtime, parking further away from the hospital and walking, and taking the stairs are things that add up in a big way.”
Keep up your normal routine
“Right now, it’s really critical for clinicians to keep up components of their routine that feel ‘normal,’ ” Dr. Rudolph said. “Whether it’s exercise, playing board games with their kids, or spending time on a hobby, it’s critical to allow yourself these comfortable, predictable, and rewarding detours.”
Set limits
People under stress tend to find unhealthy ways to cope. Instead, try being intentional about what you are consuming by putting limits on things like your news, alcohol consumption, and the number of hours you work, said Dr. Harry.
Implement a culture of wellness
Dr. Barrett believes in creating the work culture we want to be in, one that ensures people have psychological safety, allows them to ask for help, encourages them to disconnect completely from work, and makes them feel valued and listened to. She likes the example of “the pause,” which is called by a team member right after a patient expires.
“It’s a 30-second moment of silence where we reflect on the patient, their loved ones, and every member of the health care team who helped support and treat them,” said Dr. Barrett. “At the conclusion, you say: ‘Thank you. Is there anything you need to be able to go back to the care of other patients?’ Because it’s unnatural to have this terrible thing that happened and then just act like nothing happened.”
Target resources
Be proactive and know where to find resources before you need them, advised Dr. Harry. “Most institutions have free mental health resources, either through their employee assistance programs or HR, plus there’s lots of national organizations that are offering free resources to health care providers.”
Focus on what you can control
Separating what is under your control from what is not is a struggle for everyone, Dr. Poorman said, but it’s helpful to think about the ways you can have an impact and what you’re able to control.
“There was a woman who was diagnosed with early-onset Parkinson’s that I heard giving an interview at the beginning of this pandemic,” she said. “It was the most helpful advice I got, which was: ‘Think of the next good thing you can do.’ You can’t fix everything, so what’s the next good thing you can do?”
Maintain connectivity
Make sure you are utilizing your support circle and staying connected. “That sense of connection is incredibly protective on multiple fronts for depression, for burnout, for suicide ideation, etc.,” Dr. Harry said.
“It doesn’t matter if it’s your teammates at work, your family at home, your best friend from medical school – whomever you can debrief with, vent with, and just share your thoughts and feelings with, these outlets are critical for all of us to process our emotions and diffuse stress and anxiety,” said Dr. Rudolph.
Dr. Poorman is concerned that there could be a spike in physician suicides caused by increased stress, so she also encourages talking openly about what is going on and about getting help when it’s necessary. “Many of us are afraid to seek care because we can actually have our ability to practice medicine questioned, but now is not the time for heroes. Now is the time for people who are willing to recognize their own strengths and limitations to take care of one another.”
Be compassionate toward others
Keep in mind that everyone is stressed out and offer empathy and compassion. “I think everybody’s struggling to try to figure this out and the more that we can give each other the benefit of the doubt and a little grace, the more protective that is,” said Dr. Harry.
Listening is meaningful too. “Recognizing opportunities to validate and acknowledge the feelings that are being shared with you by your colleagues is critical,” Dr. Rudolph said. “We all need to know that we’re not alone, that our thoughts and feelings are okay, and when we share a difficult story, the value of someone saying something as simple as, ‘wow, that sounds like it was really hard,’ is immense.”
Be compassionate toward yourself
Try to give yourself a break and be as compassionate with yourself as you would with others. It’s okay that you’re not getting in shape, publishing prolifically, or redesigning your house right now.
“There’s a lot of data linking lack of self-compassion to burnout,” said Dr. Harry. She says there are courses on self-compassion available that help you work on being kinder to yourself.
Get a “battle buddy”
The American Medical Association has a free “buddy system” program called PeerRx to help physicians cope during the pandemic. Dr. Rudolph said that now is a great time to use this military-developed intervention in which each team member checks in with a chosen partner at agreed-upon intervals.
For example, “You can tell that person: ‘If I don’t call my family for a week that’s a red flag for me.’ And then you hold each other accountable to those things,” Dr. Harry said.
The buddy system is another way to harness that sense of connection that is so vital to our health and well-being.
“The simple act of showing that you care … can make all the difference when you’re doing this kind of work that is both challenging and dangerous,” said Dr. Rudolph.
The global COVID-19 pandemic has escalated everyone’s stress levels, especially clinicians caring for hospitalized patients. New pressures have added to everyday stress, new studies have revised prior patient care recommendations, and the world generally seems upside down. What can a busy hospitalist do to maintain a modicum of sanity in all the craziness?
The stressors facing hospitalists
Uncertainty
Of all the burdens COVID-19 has unleashed, the biggest may be uncertainty. Not only is there unease about the virus itself, there also is legitimate concern about the future of medicine, said Elizabeth Harry, MD, SFHM, a hospitalist and senior director of clinical affairs at the University of Colorado Hospital in Aurora.
“What does it look like after an event like this, particularly in areas like academic medicine and teaching our next generation and getting funding for research? And how do we continue to produce physicians that can provide excellent care?” she asked.
There is also uncertainty in the best way to care for patients, said Eileen Barrett, MD, MPH, SFHM, a hospitalist at the University of New Mexico, Albuquerque.
“There are some models that are emerging to predict who will have a worse outcome, but they’re still not great models, so we have uncertainty for a given patient.” And, she noted, as the science continues to evolve, there exists a constant worry that “you might have inadvertently caused someone harm.”
The financial implications of the pandemic are creating uncertainty too. “When you fund a health care system with elective procedures and you can’t do those, and instead have to shift to the most essential services, a lot of places are seeing a massive deficit, which is going to affect staff morale and some physician offices are going to close,” said Elisabeth Poorman, MD, MPH, a primary care and internal medicine physician and chair of the King County Medical Society Physician Wellness Committee in Seattle.
Fear
When the pandemic began in the United States, “fear of the unknown was perhaps the scariest part, particularly as it pertained to personal protective equipment,” said Mark Rudolph, MD, SFHM, chief experience officer and vice president of patient experience and physician development at Sound Physicians in Tacoma, Wash. “For most clinicians, this is the first time that they are themselves in harm’s way while they do their jobs. And worse, they risk bringing the virus home to their families. That is the concern I hear most.”
Anxiety
Worrying about being able to provide excellent patient care is a big stressor, especially since this is the heart and soul of why most hospitalists have gone into their line of work.
“Part of providing excellent care to your patients is providing excellent supportive care to their families,” Dr. Harry said. “There’s some dissonance there in not being able to allow the family to come visit, but wanting to keep them safe, and it feels really hard to support your patients and support their families in the best way. It can feel like you’re just watching and waiting to see what will happen, and that we don’t have a lot of agency over which direction things take.”
There is concern for health care team members as well, Dr. Harry added. “Physicians care a lot about their teams and how they’re doing. I think there’s a sense of esprit de corps among folks and worry for each other there.”
Guilt
Although you may be at the hospital all day, you may feel guilty when you are not providing direct patient care. Or maybe you or someone on your team has an immunodeficiency and can’t be on the front line. Perhaps one of your team members contracted COVID-19 and you did not. Whatever the case, guilt is another emotion that is rampant among hospitalists right now, Dr. Barrett said.
Burnout
Unfortunately, burnout is a potential reality in times of high stress. “Burnout is dynamic,” said Dr. Poorman. “It’s a process by which your emotional and cognitive reserves are exhausted. The people with the highest burnout are the ones who are still trying to provide the standard of care, or above the standard of care in dysfunctional systems.”
Dr. Harry noted that burnout presents in different ways for different people, but Dr. Rudolph added that it’s crucial for hospitalist team members to watch for signs of burnout so they can intervene and/or get help for their colleagues.
Warning signs in yourself or others that burnout could be on the horizon include:
- Fatigue/exhaustion – Whether emotional or physical (or both), this can become a problem if it “just doesn’t seem to go away despite rest and time away from work,” said Dr. Rudolph.
- Behavioral changes – Any behavior that’s out of the ordinary may be a red flag, like lashing out at someone at work.
- Overwork – Working too much can be caused by an inability to let go of patient care, Dr. Barrett said.
- Not working enough – This may include avoiding tasks and having difficulty meeting deadlines.
- Maladaptive coping behaviors – Excessive consumption of alcohol or drugs is a common coping mechanism. “Even excessive consumption of news is something that people are using to numb out a little bit,” said Dr. Harry.
- Depersonalization – “This is where you start to look at patients, colleagues, or administrators as ‘them’ and you can’t connect as deeply,” Dr. Harry said. “Part of that’s protective and a normal thing to do during a big trauma like this, but it’s also incredibly distancing. Any language that people start using that feels like ‘us’ or ‘them’ is a warning sign.”
- Disengagement – Many people disengage from their work, but Dr. Poorman said physicians tend to disengage from other parts of their lives, such as exercise and family interaction.
Protecting yourself while supporting others
Like the illustration of putting the oxygen mask on yourself first so you can help others, it’s important to protect your own mental and physical health as you support your fellow physicians. Here’s what the experts suggest.
Focus on basic needs
“When you’re in the midst of a trauma, which we are, you don’t want to open all of that up and go to the depths of your thoughts about the grief of all of it because it can actually make the trauma worse,” said Dr. Harry. “There’s a lot of literature that debriefing is really helpful after the event, but if you do it during the event, it can be really dangerous.”
Instead, she said, the goal should be focusing on your basic needs and what you need to do to get through each day, like keeping you and your family in good health. “What is your purpose? Staying connected to why you do this and staying focused on the present is really important,” Dr. Harry noted.
Do your best to get a good night’s sleep, exercise as much as you can, talk to others, and see a mental health provider if your anxiety is too high, advises Dr. Barrett. “Even avoiding blue light from phones and screens within 2 hours of bedtime, parking further away from the hospital and walking, and taking the stairs are things that add up in a big way.”
Keep up your normal routine
“Right now, it’s really critical for clinicians to keep up components of their routine that feel ‘normal,’ ” Dr. Rudolph said. “Whether it’s exercise, playing board games with their kids, or spending time on a hobby, it’s critical to allow yourself these comfortable, predictable, and rewarding detours.”
Set limits
People under stress tend to find unhealthy ways to cope. Instead, try being intentional about what you are consuming by putting limits on things like your news, alcohol consumption, and the number of hours you work, said Dr. Harry.
Implement a culture of wellness
Dr. Barrett believes in creating the work culture we want to be in, one that ensures people have psychological safety, allows them to ask for help, encourages them to disconnect completely from work, and makes them feel valued and listened to. She likes the example of “the pause,” which is called by a team member right after a patient expires.
“It’s a 30-second moment of silence where we reflect on the patient, their loved ones, and every member of the health care team who helped support and treat them,” said Dr. Barrett. “At the conclusion, you say: ‘Thank you. Is there anything you need to be able to go back to the care of other patients?’ Because it’s unnatural to have this terrible thing that happened and then just act like nothing happened.”
Target resources
Be proactive and know where to find resources before you need them, advised Dr. Harry. “Most institutions have free mental health resources, either through their employee assistance programs or HR, plus there’s lots of national organizations that are offering free resources to health care providers.”
Focus on what you can control
Separating what is under your control from what is not is a struggle for everyone, Dr. Poorman said, but it’s helpful to think about the ways you can have an impact and what you’re able to control.
“There was a woman who was diagnosed with early-onset Parkinson’s that I heard giving an interview at the beginning of this pandemic,” she said. “It was the most helpful advice I got, which was: ‘Think of the next good thing you can do.’ You can’t fix everything, so what’s the next good thing you can do?”
Maintain connectivity
Make sure you are utilizing your support circle and staying connected. “That sense of connection is incredibly protective on multiple fronts for depression, for burnout, for suicide ideation, etc.,” Dr. Harry said.
“It doesn’t matter if it’s your teammates at work, your family at home, your best friend from medical school – whomever you can debrief with, vent with, and just share your thoughts and feelings with, these outlets are critical for all of us to process our emotions and diffuse stress and anxiety,” said Dr. Rudolph.
Dr. Poorman is concerned that there could be a spike in physician suicides caused by increased stress, so she also encourages talking openly about what is going on and about getting help when it’s necessary. “Many of us are afraid to seek care because we can actually have our ability to practice medicine questioned, but now is not the time for heroes. Now is the time for people who are willing to recognize their own strengths and limitations to take care of one another.”
Be compassionate toward others
Keep in mind that everyone is stressed out and offer empathy and compassion. “I think everybody’s struggling to try to figure this out and the more that we can give each other the benefit of the doubt and a little grace, the more protective that is,” said Dr. Harry.
Listening is meaningful too. “Recognizing opportunities to validate and acknowledge the feelings that are being shared with you by your colleagues is critical,” Dr. Rudolph said. “We all need to know that we’re not alone, that our thoughts and feelings are okay, and when we share a difficult story, the value of someone saying something as simple as, ‘wow, that sounds like it was really hard,’ is immense.”
Be compassionate toward yourself
Try to give yourself a break and be as compassionate with yourself as you would with others. It’s okay that you’re not getting in shape, publishing prolifically, or redesigning your house right now.
“There’s a lot of data linking lack of self-compassion to burnout,” said Dr. Harry. She says there are courses on self-compassion available that help you work on being kinder to yourself.
Get a “battle buddy”
The American Medical Association has a free “buddy system” program called PeerRx to help physicians cope during the pandemic. Dr. Rudolph said that now is a great time to use this military-developed intervention in which each team member checks in with a chosen partner at agreed-upon intervals.
For example, “You can tell that person: ‘If I don’t call my family for a week that’s a red flag for me.’ And then you hold each other accountable to those things,” Dr. Harry said.
The buddy system is another way to harness that sense of connection that is so vital to our health and well-being.
“The simple act of showing that you care … can make all the difference when you’re doing this kind of work that is both challenging and dangerous,” said Dr. Rudolph.
Comorbidities increase COVID-19 deaths by factor of 12
, compared with those who have no such condition, according to the Centers for Disease Control and Prevention.
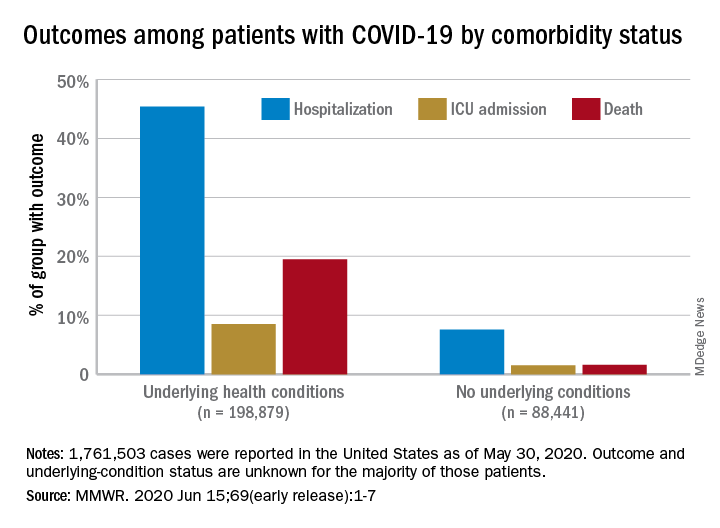
Among those with underlying conditions such as cardiovascular disease or diabetes, 45.4% of patients with COVID-19 were hospitalized, versus 7.6% of patients without an underlying condition, said Erin K. Stokes, MPH, and associates of the CDC COVID-19 Emergency Response team.
The difference in deaths was even greater over the study period of Jan. 22–May 30, 2020: 19.5% of COVID-19 patients with underlying conditions died, compared with 1.6% of those with no underlying condition. The gap narrowed, however, for ICU admissions, with corresponding rates of 8.5% and 1.5%, the investigators reported June 15 in the Morbidity and Mortality Weekly Report.
“The COVID-19 pandemic continues to be severe, particularly in certain population groups,” they said.
The cumulative incidence of laboratory-confirmed cases up to May 30, for instance, was nearly twice as high for those aged 80 years and over (902 per 100,000 population) than for those aged 70-79 years (464.2 per 100,000). Those aged 50-59 years had the second-highest incidence, 550.5 per 100,000, Ms. Stokes and associates said.
“Among cases with known race and ethnicity, 33% of persons were Hispanic, 22% were black, and 1.3% were [American Indian/Alaska Native]. These findings suggest that persons in these groups, who account for 18%, 13%, and 0.7% of the U.S. population, respectively, are disproportionately affected by the COVID-19 pandemic,” they wrote.
Another source of disparity: “Incidence among males and females was similar overall, [but] severe outcomes were more commonly reported among males,” the investigators noted. Cumulative incidence was 401.1 per 100,000 for males and 406.0 for females, but 6.0% of male patients died, compared with 4.8% of females.
As of May 30, a total of 1,761,503 cases and 103,700 deaths had been reported to the CDC. Of those cases, approximately 1.3 million were included in the analysis, with data on individual underlying health conditions available for 287,320 (22%). The split on those cases was 198,879 with health conditions and 88,411 without, the CDC data show.
The most frequently reported underlying conditions were cardiovascular disease (32%), diabetes (30%), chronic lung disease (18%), and renal disease (7.6%), and there were no significant differences between males and females, Ms. Stokes and associates said.
The pandemic “is an ongoing public health crisis in the United States that continues to affect all populations and result in severe outcomes including death,” they said, emphasizing “the continued need for community mitigation strategies, especially for vulnerable populations, to slow COVID-19 transmission.”
SOURCE: Stokes EK et al. MMWR. 2020 Jun 15;69(early release):1-7.
, compared with those who have no such condition, according to the Centers for Disease Control and Prevention.

Among those with underlying conditions such as cardiovascular disease or diabetes, 45.4% of patients with COVID-19 were hospitalized, versus 7.6% of patients without an underlying condition, said Erin K. Stokes, MPH, and associates of the CDC COVID-19 Emergency Response team.
The difference in deaths was even greater over the study period of Jan. 22–May 30, 2020: 19.5% of COVID-19 patients with underlying conditions died, compared with 1.6% of those with no underlying condition. The gap narrowed, however, for ICU admissions, with corresponding rates of 8.5% and 1.5%, the investigators reported June 15 in the Morbidity and Mortality Weekly Report.
“The COVID-19 pandemic continues to be severe, particularly in certain population groups,” they said.
The cumulative incidence of laboratory-confirmed cases up to May 30, for instance, was nearly twice as high for those aged 80 years and over (902 per 100,000 population) than for those aged 70-79 years (464.2 per 100,000). Those aged 50-59 years had the second-highest incidence, 550.5 per 100,000, Ms. Stokes and associates said.
“Among cases with known race and ethnicity, 33% of persons were Hispanic, 22% were black, and 1.3% were [American Indian/Alaska Native]. These findings suggest that persons in these groups, who account for 18%, 13%, and 0.7% of the U.S. population, respectively, are disproportionately affected by the COVID-19 pandemic,” they wrote.
Another source of disparity: “Incidence among males and females was similar overall, [but] severe outcomes were more commonly reported among males,” the investigators noted. Cumulative incidence was 401.1 per 100,000 for males and 406.0 for females, but 6.0% of male patients died, compared with 4.8% of females.
As of May 30, a total of 1,761,503 cases and 103,700 deaths had been reported to the CDC. Of those cases, approximately 1.3 million were included in the analysis, with data on individual underlying health conditions available for 287,320 (22%). The split on those cases was 198,879 with health conditions and 88,411 without, the CDC data show.
The most frequently reported underlying conditions were cardiovascular disease (32%), diabetes (30%), chronic lung disease (18%), and renal disease (7.6%), and there were no significant differences between males and females, Ms. Stokes and associates said.
The pandemic “is an ongoing public health crisis in the United States that continues to affect all populations and result in severe outcomes including death,” they said, emphasizing “the continued need for community mitigation strategies, especially for vulnerable populations, to slow COVID-19 transmission.”
SOURCE: Stokes EK et al. MMWR. 2020 Jun 15;69(early release):1-7.
, compared with those who have no such condition, according to the Centers for Disease Control and Prevention.

Among those with underlying conditions such as cardiovascular disease or diabetes, 45.4% of patients with COVID-19 were hospitalized, versus 7.6% of patients without an underlying condition, said Erin K. Stokes, MPH, and associates of the CDC COVID-19 Emergency Response team.
The difference in deaths was even greater over the study period of Jan. 22–May 30, 2020: 19.5% of COVID-19 patients with underlying conditions died, compared with 1.6% of those with no underlying condition. The gap narrowed, however, for ICU admissions, with corresponding rates of 8.5% and 1.5%, the investigators reported June 15 in the Morbidity and Mortality Weekly Report.
“The COVID-19 pandemic continues to be severe, particularly in certain population groups,” they said.
The cumulative incidence of laboratory-confirmed cases up to May 30, for instance, was nearly twice as high for those aged 80 years and over (902 per 100,000 population) than for those aged 70-79 years (464.2 per 100,000). Those aged 50-59 years had the second-highest incidence, 550.5 per 100,000, Ms. Stokes and associates said.
“Among cases with known race and ethnicity, 33% of persons were Hispanic, 22% were black, and 1.3% were [American Indian/Alaska Native]. These findings suggest that persons in these groups, who account for 18%, 13%, and 0.7% of the U.S. population, respectively, are disproportionately affected by the COVID-19 pandemic,” they wrote.
Another source of disparity: “Incidence among males and females was similar overall, [but] severe outcomes were more commonly reported among males,” the investigators noted. Cumulative incidence was 401.1 per 100,000 for males and 406.0 for females, but 6.0% of male patients died, compared with 4.8% of females.
As of May 30, a total of 1,761,503 cases and 103,700 deaths had been reported to the CDC. Of those cases, approximately 1.3 million were included in the analysis, with data on individual underlying health conditions available for 287,320 (22%). The split on those cases was 198,879 with health conditions and 88,411 without, the CDC data show.
The most frequently reported underlying conditions were cardiovascular disease (32%), diabetes (30%), chronic lung disease (18%), and renal disease (7.6%), and there were no significant differences between males and females, Ms. Stokes and associates said.
The pandemic “is an ongoing public health crisis in the United States that continues to affect all populations and result in severe outcomes including death,” they said, emphasizing “the continued need for community mitigation strategies, especially for vulnerable populations, to slow COVID-19 transmission.”
SOURCE: Stokes EK et al. MMWR. 2020 Jun 15;69(early release):1-7.
FROM MMWR
Health experts link rise in Arizona COVID cases to end of stay-at-home order
With new daily coronavirus cases rising in at least two dozen states,
Arizona has emerged as one of the country’s newest coronavirus hot spots, with the weekly average of daily cases more than doubling from 2 weeks ago. The total number of people hospitalized is climbing, too.
Over the past week, Arizona has seen an average of more than 1,300 new COVID-19 cases each day.
After the state’s largest hospital system warned about a shortage of ICU beds, Arizona Gov. Doug Ducey, a Republican, pushed back on claims that the health care system could soon be overwhelmed.
“The entire time we’ve been focused on a possible worst-case scenario with surge capacity for hospital beds, ICU beds and ventilators,” Ducey told reporters on Thursday. “Those are not needed or necessary right now.”
While he acknowledged a spike in positive cases, Ducey said a second stay-at-home order was “not under discussion.”
“We put the stay-at-home order there so we could prepare for what we are going through,” he said.
Some states have reopened more slowly with a set of specific benchmarks for different regions, but Arizona took a more aggressive approach.
The state began easing restrictions on businesses in early May and lifted its statewide lockdown order after May 15. Under Arizona’s reopening plan, businesses are advised to follow federal guidance on social distancing.
There is also no requirement for everyone to wear masks in public.
Public health experts agree: The timing of this spike reflects the state’s reopening.
“Perhaps, Arizona will be a warning sign to other areas,” said Katherine Ellingson, an epidemiologist at the University of Arizona. “We never had that consistent downward trend that would signal it’s time to reopen and we have everything in place to do it safely.”
Before Arizona lifted its stay-at-home order, only about 5% of COVID-19 tests registered as positive. On Monday, that number was around 16%.
A slower reopening gives public health agencies time to identify whether cases are rising and then respond with contact tracing and isolating those who are infected.
“With a fast, rapid reopening, we don’t have the time to mobilize those resources,” said Ellingson.
Maricopa County, home to about 60% of the state’s population, has ramped up contact tracing in recent weeks, but it may not have enough capacity if the surge in cases continues.
Dr. Peter Hotez said the spike in Arizona, as well as in parts of Texas such as Houston, Dallas and Austin, is the consequence of removing restrictions too quickly and without a public health system that can keep pace.
“It was just ‘open it up’ and then more or less business as usual, with a little bit of window dressing,” said Hotez, the dean for the National School of Tropical Medicine at Baylor College of Medicine in Houston. “This is not an abstract number of cases. We’re seeing people pile into intensive care units.”
Arizona’s governor has also faced criticism from the mayors of Arizona’s two biggest cities for not putting in place more stringent requirements.
“There is a pandemic and it’s spreading uncontrollably,” said Tucson Mayor Regina Romero, a Democrat. Ducey, she said, “is just putting up his hands and saying ‘the spread is happening and we just have to go about our business.’”
And the governor’s executive order forbids local governments from implementing their own extra measures, which adds to Romero’s frustration. Texas has a similar measure.
“What he did was pretty much tie the hands of mayors and public health officials,” Romero said.
Arizona’s hospital industry has tried to tamp down fears that it’s on the verge of a crisis. Hospitals are still performing elective surgeries.
“It’s very unfortunate because hospitals right now in Arizona are quite busy with elective procedures,” said Saskia Popescu, a Phoenix-based epidemiologist with George Mason University. “You throw in increasing cases of COVID, and that’s going to very much stress your hospital systems.”
Phoenix’s triple-digit summer temperatures actually may fuel the spread of the virus. People forgo outdoor activities and retreat to air-conditioned indoor spaces, where the risk of transmitting the virus goes up significantly.
“My concern is we’re going to see a lot more people in close quarters for prolonged periods of time,” Popescu said.
Since the stay-at-home order was lifted, Popescu and others say they’ve seen people returning to a pre-pandemic mindset, neglecting to wear masks or maintain social distance. Videos of crowded bars have only propelled these fears.
On Thursday, however, Arizona’s top doctor stressed there were also dangers to keeping the state on lockdown, including the mental health effects of loneliness and isolation.
“We know that it’s in the community. We are not going to be able to stop the spread. And so we can’t stop living as well,” said Dr. Cara Christ, health director for the Arizona Department of Health Services.
But Dr. Quinn Snyder, an emergency medicine physician in Mesa, Arizona, said there needs to be more consistent messaging on public health measures like wearing masks.
“Frankly, I just think a wholesale reevaluation of where we’re at is critical right now, but I can tell you that we’re not doing nearly enough,” said Snyder, who has seen the uptick in seriously ill COVID-19 patients firsthand.
“If we continue to head down this path, the virus will press our health care facilities beyond capacity, where we’re going to have to be making tough decisions like who gets a ventilator and who doesn’t.”
A version of this article originally appeared on Kaiser Health News, which is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
With new daily coronavirus cases rising in at least two dozen states,
Arizona has emerged as one of the country’s newest coronavirus hot spots, with the weekly average of daily cases more than doubling from 2 weeks ago. The total number of people hospitalized is climbing, too.
Over the past week, Arizona has seen an average of more than 1,300 new COVID-19 cases each day.
After the state’s largest hospital system warned about a shortage of ICU beds, Arizona Gov. Doug Ducey, a Republican, pushed back on claims that the health care system could soon be overwhelmed.
“The entire time we’ve been focused on a possible worst-case scenario with surge capacity for hospital beds, ICU beds and ventilators,” Ducey told reporters on Thursday. “Those are not needed or necessary right now.”
While he acknowledged a spike in positive cases, Ducey said a second stay-at-home order was “not under discussion.”
“We put the stay-at-home order there so we could prepare for what we are going through,” he said.
Some states have reopened more slowly with a set of specific benchmarks for different regions, but Arizona took a more aggressive approach.
The state began easing restrictions on businesses in early May and lifted its statewide lockdown order after May 15. Under Arizona’s reopening plan, businesses are advised to follow federal guidance on social distancing.
There is also no requirement for everyone to wear masks in public.
Public health experts agree: The timing of this spike reflects the state’s reopening.
“Perhaps, Arizona will be a warning sign to other areas,” said Katherine Ellingson, an epidemiologist at the University of Arizona. “We never had that consistent downward trend that would signal it’s time to reopen and we have everything in place to do it safely.”
Before Arizona lifted its stay-at-home order, only about 5% of COVID-19 tests registered as positive. On Monday, that number was around 16%.
A slower reopening gives public health agencies time to identify whether cases are rising and then respond with contact tracing and isolating those who are infected.
“With a fast, rapid reopening, we don’t have the time to mobilize those resources,” said Ellingson.
Maricopa County, home to about 60% of the state’s population, has ramped up contact tracing in recent weeks, but it may not have enough capacity if the surge in cases continues.
Dr. Peter Hotez said the spike in Arizona, as well as in parts of Texas such as Houston, Dallas and Austin, is the consequence of removing restrictions too quickly and without a public health system that can keep pace.
“It was just ‘open it up’ and then more or less business as usual, with a little bit of window dressing,” said Hotez, the dean for the National School of Tropical Medicine at Baylor College of Medicine in Houston. “This is not an abstract number of cases. We’re seeing people pile into intensive care units.”
Arizona’s governor has also faced criticism from the mayors of Arizona’s two biggest cities for not putting in place more stringent requirements.
“There is a pandemic and it’s spreading uncontrollably,” said Tucson Mayor Regina Romero, a Democrat. Ducey, she said, “is just putting up his hands and saying ‘the spread is happening and we just have to go about our business.’”
And the governor’s executive order forbids local governments from implementing their own extra measures, which adds to Romero’s frustration. Texas has a similar measure.
“What he did was pretty much tie the hands of mayors and public health officials,” Romero said.
Arizona’s hospital industry has tried to tamp down fears that it’s on the verge of a crisis. Hospitals are still performing elective surgeries.
“It’s very unfortunate because hospitals right now in Arizona are quite busy with elective procedures,” said Saskia Popescu, a Phoenix-based epidemiologist with George Mason University. “You throw in increasing cases of COVID, and that’s going to very much stress your hospital systems.”
Phoenix’s triple-digit summer temperatures actually may fuel the spread of the virus. People forgo outdoor activities and retreat to air-conditioned indoor spaces, where the risk of transmitting the virus goes up significantly.
“My concern is we’re going to see a lot more people in close quarters for prolonged periods of time,” Popescu said.
Since the stay-at-home order was lifted, Popescu and others say they’ve seen people returning to a pre-pandemic mindset, neglecting to wear masks or maintain social distance. Videos of crowded bars have only propelled these fears.
On Thursday, however, Arizona’s top doctor stressed there were also dangers to keeping the state on lockdown, including the mental health effects of loneliness and isolation.
“We know that it’s in the community. We are not going to be able to stop the spread. And so we can’t stop living as well,” said Dr. Cara Christ, health director for the Arizona Department of Health Services.
But Dr. Quinn Snyder, an emergency medicine physician in Mesa, Arizona, said there needs to be more consistent messaging on public health measures like wearing masks.
“Frankly, I just think a wholesale reevaluation of where we’re at is critical right now, but I can tell you that we’re not doing nearly enough,” said Snyder, who has seen the uptick in seriously ill COVID-19 patients firsthand.
“If we continue to head down this path, the virus will press our health care facilities beyond capacity, where we’re going to have to be making tough decisions like who gets a ventilator and who doesn’t.”
A version of this article originally appeared on Kaiser Health News, which is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
With new daily coronavirus cases rising in at least two dozen states,
Arizona has emerged as one of the country’s newest coronavirus hot spots, with the weekly average of daily cases more than doubling from 2 weeks ago. The total number of people hospitalized is climbing, too.
Over the past week, Arizona has seen an average of more than 1,300 new COVID-19 cases each day.
After the state’s largest hospital system warned about a shortage of ICU beds, Arizona Gov. Doug Ducey, a Republican, pushed back on claims that the health care system could soon be overwhelmed.
“The entire time we’ve been focused on a possible worst-case scenario with surge capacity for hospital beds, ICU beds and ventilators,” Ducey told reporters on Thursday. “Those are not needed or necessary right now.”
While he acknowledged a spike in positive cases, Ducey said a second stay-at-home order was “not under discussion.”
“We put the stay-at-home order there so we could prepare for what we are going through,” he said.
Some states have reopened more slowly with a set of specific benchmarks for different regions, but Arizona took a more aggressive approach.
The state began easing restrictions on businesses in early May and lifted its statewide lockdown order after May 15. Under Arizona’s reopening plan, businesses are advised to follow federal guidance on social distancing.
There is also no requirement for everyone to wear masks in public.
Public health experts agree: The timing of this spike reflects the state’s reopening.
“Perhaps, Arizona will be a warning sign to other areas,” said Katherine Ellingson, an epidemiologist at the University of Arizona. “We never had that consistent downward trend that would signal it’s time to reopen and we have everything in place to do it safely.”
Before Arizona lifted its stay-at-home order, only about 5% of COVID-19 tests registered as positive. On Monday, that number was around 16%.
A slower reopening gives public health agencies time to identify whether cases are rising and then respond with contact tracing and isolating those who are infected.
“With a fast, rapid reopening, we don’t have the time to mobilize those resources,” said Ellingson.
Maricopa County, home to about 60% of the state’s population, has ramped up contact tracing in recent weeks, but it may not have enough capacity if the surge in cases continues.
Dr. Peter Hotez said the spike in Arizona, as well as in parts of Texas such as Houston, Dallas and Austin, is the consequence of removing restrictions too quickly and without a public health system that can keep pace.
“It was just ‘open it up’ and then more or less business as usual, with a little bit of window dressing,” said Hotez, the dean for the National School of Tropical Medicine at Baylor College of Medicine in Houston. “This is not an abstract number of cases. We’re seeing people pile into intensive care units.”
Arizona’s governor has also faced criticism from the mayors of Arizona’s two biggest cities for not putting in place more stringent requirements.
“There is a pandemic and it’s spreading uncontrollably,” said Tucson Mayor Regina Romero, a Democrat. Ducey, she said, “is just putting up his hands and saying ‘the spread is happening and we just have to go about our business.’”
And the governor’s executive order forbids local governments from implementing their own extra measures, which adds to Romero’s frustration. Texas has a similar measure.
“What he did was pretty much tie the hands of mayors and public health officials,” Romero said.
Arizona’s hospital industry has tried to tamp down fears that it’s on the verge of a crisis. Hospitals are still performing elective surgeries.
“It’s very unfortunate because hospitals right now in Arizona are quite busy with elective procedures,” said Saskia Popescu, a Phoenix-based epidemiologist with George Mason University. “You throw in increasing cases of COVID, and that’s going to very much stress your hospital systems.”
Phoenix’s triple-digit summer temperatures actually may fuel the spread of the virus. People forgo outdoor activities and retreat to air-conditioned indoor spaces, where the risk of transmitting the virus goes up significantly.
“My concern is we’re going to see a lot more people in close quarters for prolonged periods of time,” Popescu said.
Since the stay-at-home order was lifted, Popescu and others say they’ve seen people returning to a pre-pandemic mindset, neglecting to wear masks or maintain social distance. Videos of crowded bars have only propelled these fears.
On Thursday, however, Arizona’s top doctor stressed there were also dangers to keeping the state on lockdown, including the mental health effects of loneliness and isolation.
“We know that it’s in the community. We are not going to be able to stop the spread. And so we can’t stop living as well,” said Dr. Cara Christ, health director for the Arizona Department of Health Services.
But Dr. Quinn Snyder, an emergency medicine physician in Mesa, Arizona, said there needs to be more consistent messaging on public health measures like wearing masks.
“Frankly, I just think a wholesale reevaluation of where we’re at is critical right now, but I can tell you that we’re not doing nearly enough,” said Snyder, who has seen the uptick in seriously ill COVID-19 patients firsthand.
“If we continue to head down this path, the virus will press our health care facilities beyond capacity, where we’re going to have to be making tough decisions like who gets a ventilator and who doesn’t.”
A version of this article originally appeared on Kaiser Health News, which is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
Survey: 26% of parents hesitant about influenza vaccine
according to a nationally representative survey.
Influenza vaccination hesitancy may be driven by concerns about vaccine effectiveness, researchers wrote in Pediatrics. These findings “underscore the importance of better communicating to providers and parents the effectiveness of influenza vaccines in reducing severity and morbidity from influenza, even in years when the vaccine has relatively low effectiveness,” noted Allison Kempe, MD, MPH, professor of pediatrics and director of the Adult and Child Consortium for Health Outcomes Research and Delivery Science at the University of Colorado at Denver, Aurora, and colleagues.
The World Health Organization considers vaccine hesitancy a leading threat to global health, but national data about vaccine hesitancy in the United States are limited. To assess hesitancy about routine childhood and influenza vaccinations and related factors, Dr. Kempe and colleagues surveyed more than 2,000 parents in February 2019.
The investigators used an online panel to survey a nationally representative sample of families with children aged between 6 months and 18 years. Parents completed a modified version of the Vaccine Hesitancy Scale, which measures confidence in and concerns about vaccines. Parents with an average score greater than 3 on the scale were considered hesitant.
Factors associated with vaccine hesitancy
Of 4,445 parents sampled, 2,176 completed the survey and 2,052 were eligible respondents. For routine childhood vaccines, the average score on the modified Vaccine Hesitancy Scale was 2 and the percentage of hesitant parents was 6%. For influenza vaccine, the average score was 2 and the percentage of hesitant parents was 26%.
Among hesitant parents, 68% had deferred or refused routine childhood vaccination, compared with 9% of nonhesitant parents (risk ratio, 8.0). For the influenza vaccine, 70% of hesitant parents had deferred or refused influenza vaccination for their child versus 10% of nonhesitant parents (RR, 7.0). Parents were more likely to strongly agree that routine childhood vaccines are effective, compared with the influenza vaccine (70% vs. 26%). “Hesitancy about influenza vaccination is largely driven by concerns about low vaccine effectiveness,” Dr. Kempe and associates wrote.
Although concern about serious side effects was the factor most associated with hesitancy, the percentage of parents who were strongly (12%) or somewhat (27%) concerned about serious side effects was the same for routine childhood vaccines and influenza vaccines. Other factors associated with hesitancy for both routine childhood vaccines and influenza vaccines included lower educational level and household income less than 400% of the federal poverty level.
The survey data may be subject to reporting bias based on social desirability, the authors noted. In addition, the exclusion of infants younger than 6 months may have resulted in an underestimate of hesitancy.
“Although influenza vaccine could be included as a ‘routine’ vaccine, in that it is recommended yearly, we hypothesized that parents view it differently from other childhood vaccines because each year it needs to be given again, its content and effectiveness vary, and it addresses a disease that is often perceived as minor, compared with other childhood diseases,” Dr. Kempe and colleagues wrote. Interventions to counter hesitancy have “a surprising lack of evidence,” and “more work needs to be done to develop methods that are practical and effective for convincing vaccine-hesitant parents to vaccinate.”
Logical next step
“From the pragmatic standpoint of improving immunization rates and disease control, determining the correct evidence-based messaging to counter these perceptions is the next logical step,” Annabelle de St. Maurice, MD, MPH, an assistant professor of pediatrics in the division of infectious diseases at University of California, Los Angeles, and Kathryn Edwards, MD, a professor of pediatrics and director of the vaccine research program at Vanderbilt University, Nashville, wrote in an accompanying editorial.
“Communications should be focused on the burden of influenza in children, rebranding influenza vaccine as a ‘routine’ childhood immunization, reassurance on influenza vaccine safety, and discussion of the efficacy of influenza vaccine in preventing severe disease,” they wrote. “Even in the years when there is a poor match, the vaccine is impactful.”
The research was supported by the National Institutes of Health. Two study authors disclosed financial ties to Sanofi Pasteur, with one also disclosing financial ties to Merck, for work related to vaccinations. The remaining investigators had no relevant financial disclosures. Dr. de St. Maurice indicated that she had no relevant financial disclosures. Dr. Edwards disclosed grants from the Centers for Disease Control and Prevention and the NIH; consulting for Merck, Bionet, and IBM; and serving on data safety and monitoring boards for Sanofi, X4 Pharmaceuticals, Seqirus, Moderna, and Pfizer.
SOURCE: Kempe A et al. Pediatrics. 2020 Jun 15. doi: 10.1542/peds.2019-3852.
according to a nationally representative survey.
Influenza vaccination hesitancy may be driven by concerns about vaccine effectiveness, researchers wrote in Pediatrics. These findings “underscore the importance of better communicating to providers and parents the effectiveness of influenza vaccines in reducing severity and morbidity from influenza, even in years when the vaccine has relatively low effectiveness,” noted Allison Kempe, MD, MPH, professor of pediatrics and director of the Adult and Child Consortium for Health Outcomes Research and Delivery Science at the University of Colorado at Denver, Aurora, and colleagues.
The World Health Organization considers vaccine hesitancy a leading threat to global health, but national data about vaccine hesitancy in the United States are limited. To assess hesitancy about routine childhood and influenza vaccinations and related factors, Dr. Kempe and colleagues surveyed more than 2,000 parents in February 2019.
The investigators used an online panel to survey a nationally representative sample of families with children aged between 6 months and 18 years. Parents completed a modified version of the Vaccine Hesitancy Scale, which measures confidence in and concerns about vaccines. Parents with an average score greater than 3 on the scale were considered hesitant.
Factors associated with vaccine hesitancy
Of 4,445 parents sampled, 2,176 completed the survey and 2,052 were eligible respondents. For routine childhood vaccines, the average score on the modified Vaccine Hesitancy Scale was 2 and the percentage of hesitant parents was 6%. For influenza vaccine, the average score was 2 and the percentage of hesitant parents was 26%.
Among hesitant parents, 68% had deferred or refused routine childhood vaccination, compared with 9% of nonhesitant parents (risk ratio, 8.0). For the influenza vaccine, 70% of hesitant parents had deferred or refused influenza vaccination for their child versus 10% of nonhesitant parents (RR, 7.0). Parents were more likely to strongly agree that routine childhood vaccines are effective, compared with the influenza vaccine (70% vs. 26%). “Hesitancy about influenza vaccination is largely driven by concerns about low vaccine effectiveness,” Dr. Kempe and associates wrote.
Although concern about serious side effects was the factor most associated with hesitancy, the percentage of parents who were strongly (12%) or somewhat (27%) concerned about serious side effects was the same for routine childhood vaccines and influenza vaccines. Other factors associated with hesitancy for both routine childhood vaccines and influenza vaccines included lower educational level and household income less than 400% of the federal poverty level.
The survey data may be subject to reporting bias based on social desirability, the authors noted. In addition, the exclusion of infants younger than 6 months may have resulted in an underestimate of hesitancy.
“Although influenza vaccine could be included as a ‘routine’ vaccine, in that it is recommended yearly, we hypothesized that parents view it differently from other childhood vaccines because each year it needs to be given again, its content and effectiveness vary, and it addresses a disease that is often perceived as minor, compared with other childhood diseases,” Dr. Kempe and colleagues wrote. Interventions to counter hesitancy have “a surprising lack of evidence,” and “more work needs to be done to develop methods that are practical and effective for convincing vaccine-hesitant parents to vaccinate.”
Logical next step
“From the pragmatic standpoint of improving immunization rates and disease control, determining the correct evidence-based messaging to counter these perceptions is the next logical step,” Annabelle de St. Maurice, MD, MPH, an assistant professor of pediatrics in the division of infectious diseases at University of California, Los Angeles, and Kathryn Edwards, MD, a professor of pediatrics and director of the vaccine research program at Vanderbilt University, Nashville, wrote in an accompanying editorial.
“Communications should be focused on the burden of influenza in children, rebranding influenza vaccine as a ‘routine’ childhood immunization, reassurance on influenza vaccine safety, and discussion of the efficacy of influenza vaccine in preventing severe disease,” they wrote. “Even in the years when there is a poor match, the vaccine is impactful.”
The research was supported by the National Institutes of Health. Two study authors disclosed financial ties to Sanofi Pasteur, with one also disclosing financial ties to Merck, for work related to vaccinations. The remaining investigators had no relevant financial disclosures. Dr. de St. Maurice indicated that she had no relevant financial disclosures. Dr. Edwards disclosed grants from the Centers for Disease Control and Prevention and the NIH; consulting for Merck, Bionet, and IBM; and serving on data safety and monitoring boards for Sanofi, X4 Pharmaceuticals, Seqirus, Moderna, and Pfizer.
SOURCE: Kempe A et al. Pediatrics. 2020 Jun 15. doi: 10.1542/peds.2019-3852.
according to a nationally representative survey.
Influenza vaccination hesitancy may be driven by concerns about vaccine effectiveness, researchers wrote in Pediatrics. These findings “underscore the importance of better communicating to providers and parents the effectiveness of influenza vaccines in reducing severity and morbidity from influenza, even in years when the vaccine has relatively low effectiveness,” noted Allison Kempe, MD, MPH, professor of pediatrics and director of the Adult and Child Consortium for Health Outcomes Research and Delivery Science at the University of Colorado at Denver, Aurora, and colleagues.
The World Health Organization considers vaccine hesitancy a leading threat to global health, but national data about vaccine hesitancy in the United States are limited. To assess hesitancy about routine childhood and influenza vaccinations and related factors, Dr. Kempe and colleagues surveyed more than 2,000 parents in February 2019.
The investigators used an online panel to survey a nationally representative sample of families with children aged between 6 months and 18 years. Parents completed a modified version of the Vaccine Hesitancy Scale, which measures confidence in and concerns about vaccines. Parents with an average score greater than 3 on the scale were considered hesitant.
Factors associated with vaccine hesitancy
Of 4,445 parents sampled, 2,176 completed the survey and 2,052 were eligible respondents. For routine childhood vaccines, the average score on the modified Vaccine Hesitancy Scale was 2 and the percentage of hesitant parents was 6%. For influenza vaccine, the average score was 2 and the percentage of hesitant parents was 26%.
Among hesitant parents, 68% had deferred or refused routine childhood vaccination, compared with 9% of nonhesitant parents (risk ratio, 8.0). For the influenza vaccine, 70% of hesitant parents had deferred or refused influenza vaccination for their child versus 10% of nonhesitant parents (RR, 7.0). Parents were more likely to strongly agree that routine childhood vaccines are effective, compared with the influenza vaccine (70% vs. 26%). “Hesitancy about influenza vaccination is largely driven by concerns about low vaccine effectiveness,” Dr. Kempe and associates wrote.
Although concern about serious side effects was the factor most associated with hesitancy, the percentage of parents who were strongly (12%) or somewhat (27%) concerned about serious side effects was the same for routine childhood vaccines and influenza vaccines. Other factors associated with hesitancy for both routine childhood vaccines and influenza vaccines included lower educational level and household income less than 400% of the federal poverty level.
The survey data may be subject to reporting bias based on social desirability, the authors noted. In addition, the exclusion of infants younger than 6 months may have resulted in an underestimate of hesitancy.
“Although influenza vaccine could be included as a ‘routine’ vaccine, in that it is recommended yearly, we hypothesized that parents view it differently from other childhood vaccines because each year it needs to be given again, its content and effectiveness vary, and it addresses a disease that is often perceived as minor, compared with other childhood diseases,” Dr. Kempe and colleagues wrote. Interventions to counter hesitancy have “a surprising lack of evidence,” and “more work needs to be done to develop methods that are practical and effective for convincing vaccine-hesitant parents to vaccinate.”
Logical next step
“From the pragmatic standpoint of improving immunization rates and disease control, determining the correct evidence-based messaging to counter these perceptions is the next logical step,” Annabelle de St. Maurice, MD, MPH, an assistant professor of pediatrics in the division of infectious diseases at University of California, Los Angeles, and Kathryn Edwards, MD, a professor of pediatrics and director of the vaccine research program at Vanderbilt University, Nashville, wrote in an accompanying editorial.
“Communications should be focused on the burden of influenza in children, rebranding influenza vaccine as a ‘routine’ childhood immunization, reassurance on influenza vaccine safety, and discussion of the efficacy of influenza vaccine in preventing severe disease,” they wrote. “Even in the years when there is a poor match, the vaccine is impactful.”
The research was supported by the National Institutes of Health. Two study authors disclosed financial ties to Sanofi Pasteur, with one also disclosing financial ties to Merck, for work related to vaccinations. The remaining investigators had no relevant financial disclosures. Dr. de St. Maurice indicated that she had no relevant financial disclosures. Dr. Edwards disclosed grants from the Centers for Disease Control and Prevention and the NIH; consulting for Merck, Bionet, and IBM; and serving on data safety and monitoring boards for Sanofi, X4 Pharmaceuticals, Seqirus, Moderna, and Pfizer.
SOURCE: Kempe A et al. Pediatrics. 2020 Jun 15. doi: 10.1542/peds.2019-3852.
FROM PEDIATRICS
FDA revokes emergency use of hydroxychloroquine
The U.S. Food and Drug Administration revoked its decision from March 28 allowing use of hydroxychloroquine and chloroquine to treat people hospitalized with COVID-19 under an emergency use authorization (EUA).
“Based on its ongoing analysis of the EUA and emerging scientific data, the FDA determined that chloroquine and hydroxychloroquine are unlikely to be effective in treating COVID-19 for the authorized uses in the EUA,” the agency announced in a June 15 statement.
The FDA also warned today that the use of hydroxychloroquine or chloroquine may have a potential drug interaction with the investigational antiviral drug remdesivir that limits its effectiveness against COVID-19.
Remdesivir was granted emergency use authorization by the FDA on May 1.
“Based on a recently completed nonclinical laboratory study, the FDA is revising the fact sheet for healthcare providers that accompanies the drug to state that coadministration of remdesivir and chloroquine phosphate or hydroxychloroquine sulfate is not recommended as it may result in reduced antiviral activity of remdesivir. The agency is not aware of instances of this reduced activity occurring in the clinical setting but is continuing to evaluate all data related to remdesivir,” the FDA said in a news release.
Controversy over hydroxychloroquine
Even with such federal permission, since late March the use of these two agents has been mired in controversy.
President Donald J. Trump promoted the use of hydroxychloroquine and chloroquine to treat Americans with COVID-19, while scientific studies raised questions about their safety and effectiveness. Recent research, for example, pointed to elevated cardiovascular risks, as reported by Medscape Medical News.
The FDA acknowledged this recent evidence. “Additionally, in light of ongoing serious cardiac adverse events and other potential serious side effects, the known and potential benefits of chloroquine and hydroxychloroquine no longer outweigh the known and potential risks for the authorized use.”
The full suspension of the EUA follows a warning the agency issued on April 24. The FDA’s Safety Communication cautioned against use of the two agents outside of a hospital setting, citing an increase in outpatient prescriptions and “reports of serious heart rhythm problems.”
“While additional clinical trials continue to evaluate the potential benefit of these drugs in treating or preventing COVID-19, we determined the emergency use authorization was no longer appropriate,” based on a rigorous assessment by scientists in our Center for Drug Evaluation and Research,” Patrizia Cavazzoni, MD, acting director of CDER, noted in the FDA statement.
This article first appeared on Medscape.com.
The U.S. Food and Drug Administration revoked its decision from March 28 allowing use of hydroxychloroquine and chloroquine to treat people hospitalized with COVID-19 under an emergency use authorization (EUA).
“Based on its ongoing analysis of the EUA and emerging scientific data, the FDA determined that chloroquine and hydroxychloroquine are unlikely to be effective in treating COVID-19 for the authorized uses in the EUA,” the agency announced in a June 15 statement.
The FDA also warned today that the use of hydroxychloroquine or chloroquine may have a potential drug interaction with the investigational antiviral drug remdesivir that limits its effectiveness against COVID-19.
Remdesivir was granted emergency use authorization by the FDA on May 1.
“Based on a recently completed nonclinical laboratory study, the FDA is revising the fact sheet for healthcare providers that accompanies the drug to state that coadministration of remdesivir and chloroquine phosphate or hydroxychloroquine sulfate is not recommended as it may result in reduced antiviral activity of remdesivir. The agency is not aware of instances of this reduced activity occurring in the clinical setting but is continuing to evaluate all data related to remdesivir,” the FDA said in a news release.
Controversy over hydroxychloroquine
Even with such federal permission, since late March the use of these two agents has been mired in controversy.
President Donald J. Trump promoted the use of hydroxychloroquine and chloroquine to treat Americans with COVID-19, while scientific studies raised questions about their safety and effectiveness. Recent research, for example, pointed to elevated cardiovascular risks, as reported by Medscape Medical News.
The FDA acknowledged this recent evidence. “Additionally, in light of ongoing serious cardiac adverse events and other potential serious side effects, the known and potential benefits of chloroquine and hydroxychloroquine no longer outweigh the known and potential risks for the authorized use.”
The full suspension of the EUA follows a warning the agency issued on April 24. The FDA’s Safety Communication cautioned against use of the two agents outside of a hospital setting, citing an increase in outpatient prescriptions and “reports of serious heart rhythm problems.”
“While additional clinical trials continue to evaluate the potential benefit of these drugs in treating or preventing COVID-19, we determined the emergency use authorization was no longer appropriate,” based on a rigorous assessment by scientists in our Center for Drug Evaluation and Research,” Patrizia Cavazzoni, MD, acting director of CDER, noted in the FDA statement.
This article first appeared on Medscape.com.
The U.S. Food and Drug Administration revoked its decision from March 28 allowing use of hydroxychloroquine and chloroquine to treat people hospitalized with COVID-19 under an emergency use authorization (EUA).
“Based on its ongoing analysis of the EUA and emerging scientific data, the FDA determined that chloroquine and hydroxychloroquine are unlikely to be effective in treating COVID-19 for the authorized uses in the EUA,” the agency announced in a June 15 statement.
The FDA also warned today that the use of hydroxychloroquine or chloroquine may have a potential drug interaction with the investigational antiviral drug remdesivir that limits its effectiveness against COVID-19.
Remdesivir was granted emergency use authorization by the FDA on May 1.
“Based on a recently completed nonclinical laboratory study, the FDA is revising the fact sheet for healthcare providers that accompanies the drug to state that coadministration of remdesivir and chloroquine phosphate or hydroxychloroquine sulfate is not recommended as it may result in reduced antiviral activity of remdesivir. The agency is not aware of instances of this reduced activity occurring in the clinical setting but is continuing to evaluate all data related to remdesivir,” the FDA said in a news release.
Controversy over hydroxychloroquine
Even with such federal permission, since late March the use of these two agents has been mired in controversy.
President Donald J. Trump promoted the use of hydroxychloroquine and chloroquine to treat Americans with COVID-19, while scientific studies raised questions about their safety and effectiveness. Recent research, for example, pointed to elevated cardiovascular risks, as reported by Medscape Medical News.
The FDA acknowledged this recent evidence. “Additionally, in light of ongoing serious cardiac adverse events and other potential serious side effects, the known and potential benefits of chloroquine and hydroxychloroquine no longer outweigh the known and potential risks for the authorized use.”
The full suspension of the EUA follows a warning the agency issued on April 24. The FDA’s Safety Communication cautioned against use of the two agents outside of a hospital setting, citing an increase in outpatient prescriptions and “reports of serious heart rhythm problems.”
“While additional clinical trials continue to evaluate the potential benefit of these drugs in treating or preventing COVID-19, we determined the emergency use authorization was no longer appropriate,” based on a rigorous assessment by scientists in our Center for Drug Evaluation and Research,” Patrizia Cavazzoni, MD, acting director of CDER, noted in the FDA statement.
This article first appeared on Medscape.com.
Perfect storm of SARS-CoV-2 during flu season
COVID-19 now. The urban phase of the U.S. pandemic is leveling somewhat, while the rural phase is accelerating – in part because of food processing and handling industries. The pediatric burden has been surprisingly small, with the multisystem inflammatory disease (MIS-c) in children noted in several hundred cases now being seen across the country.
Next wave? Given ongoing COVID-19 disease, controversy rages about when and how to re-open the country. Regardless how more reopening occurs over the next months, we should expect a next or ongoing COVID-19 wave, particularly given loss of social distancing during social justice protests. A sawtooth disease prevalence pattern is predicted by many experts: a drop in prevalence leading to reopening, leading to scattered prevalence increases and regional if not local restriction tightening, followed by another drop in prevalence. Then “rinse and repeat” until 70% of the population is immune either by disease experience or vaccine-induced immunity, likely sometime in 2021.
Influenza too. A COVID-19 up-cycle is likely during influenza season, although influenza season’s onset could be altered because of whatever social distancing rules are in place in November and December. That said, we need to consider the worst. We have seen what happens if we fail to prepare and then react only after a prevalent respiratory infection has surged into the overall population. Best estimates are that at most 20% of the U.S. population is currently immune to SARS-CoV-2. Given that at least some of that 20% of individuals currently immune to SARS-CoV-2 will lose their neutralizing antibody over the next 4-6 months, we can still expect 70%-80% of the U.S. population to be susceptible to SARS-CoV-2 infection in the fall of 2020.
Pediatric preparedness. As pediatric providers, we have struggled with lower patient loads and dramatic income losses/declines. Many clinics/offices’ attendance remain less than 50% of pre–COVID-19 levels, with necessary furloughs of personnel and spotty office hours. But influenza is coming, and SARS-CoV-2 will not be gone yet. How do we prepare for concurrent influenza and COVID-19?
The annual purchase/administration of influenza vaccine in summer/fall is expensive, time consuming, and logistically difficult even in the best times. Given the loss of income, likely reluctance of patients to come to clinics/offices if COVID-19 is still circulating, and likely need for some form of social distancing during late summer and early fall, how will providers, health departments, and hospitals implement influenza vaccine administration this year?
Minimize double whammy infections. It is easy to understand why we should maximize influenza protection in SARS-CoV-2 vulnerables (elderly or persons with existing comorbidities). But is it as critical for otherwise healthy children? My answer is yes.
Children are not currently known as SARS-CoV-2 vectors, but children are excellent influenza vectors, shedding higher titers for longer than other age groups. As with SARS-CoV-2, influenza exposure is cumulative, i.e., the more intense and more frequently a person is exposed, the more likely that infection/disease will result. So, the fewer who get and can transmit influenza during the COVID-19 pandemic, the fewer people are likely to get a double whammy of SARS-CoV-2 concurrent or in tandem with influenza. Double whammy infections likely would further increase the medical care burden and return us to March-April crisis mode.
One alarming new question is whether recent influenza could make children vulnerable to SARS-CoV-2 and trigger hospitalizations. A surge in pediatric plus adult COVID-19 disease plus a surge in all-ages influenza disease would likely break the medical care system, at least in some areas.
Staggering COVID-19 burden. As of June 8, we have had approximately 2 million SARS-CoV-2 cases with 500,000 hospitalizations and 120,000 deaths. Over the past 10 years, total annual U.S. influenza hospitalizations ranged from 180,000 (2011-2012) to 825,000 (2017-2018). The interquartile range for hospitalization length of stay for influenza is 4-6 days1 vs. 15-23 days2 for SARS-CoV-2. One COVID-19 hospitalization uses hospital resources roughly equal to four influenza hospitalizations. To date COVID-19 hospitalizations have used resources equal to an estimated 1.9 million influenza hospitalizations – over twice the worst influenza season in this century – and we are still on the rise. We are likely not even halfway to truly controlling the U.S. pandemic, so expect another 500,000 hospitalizations – equal to another 1.9 million influenza hospitalizations. Further, pneumonia deaths have skyrocketed this year when COVID-19 was superimposed on the last third of influenza season. One hope is that widespread use of antivirals (for example, new antivirals, convalescent plasma, or other interventions) can reduce length of stay by 30% for COVID-19 hospitalizations, yet even with that the numbers remain grim.
Less influenza disease can free up medical resources. Planning ahead could prevent a bad influenza season (for example, up to 850,000 hospitalizations just for influenza). Can we preemptively use vaccine to reduce influenza hospitalizations below 2011-2012 levels – less than 150,000 hospitalizations? Perhaps, if we start by reducing pediatric influenza.
1. Aim to exceed 75% influenza vaccine uptake in your patients.
a. It is ambitious, but if there was ever a year that needed influenza herd immunity, it is 2020-2021.
2. Review practice/group/institution plans for vaccine purchase and ensure adequate personnel to administer vaccine.
3. Plan safe and efficient processes to vaccinate large numbers in August through November.
a. Consider that routine and influenza vaccines can be given concurrently with the annual uptick in school and sports physical examinations.
b. What social distancing and masking rules will be needed?
i. Will patients need to bring their own masks, or will you supply them?
c. What extra supplies and efforts are needed, e.g. hand sanitizer, new signage, 6-foot interval markings on floors or sidewalks, families calling from parking lot to announce their arrivals, etc.?
d. Remember younger patients need two doses before Dec 1, 2020.
e. Be creative, for example, are parking-lot tents for influenza vaccination feasible?
f. Can we partner with other providers to implement influenza vaccine–specific mass clinics?
Ramping up to give seasonal influenza vaccine in 2020 is daunting. But if we do not prepare, it will be even more difficult. Let’s make this the mildest influenza season in memory by vaccinating more than any time in memory – and by doing so, we can hope to blunt medical care burdens despite ongoing COVID-19 disease.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Kansas City (Mo.). Children’s Mercy receives funding from GlaxoSmithKline, Merck, and Pfizer for vaccine research studies on which Dr. Harrison is an investigator. Email him at [email protected].
References
1.. HCUP Statistical Brief #253. 2019 Oct.
2. medrxiv. 2020 Apr 10. doi: 10.1101/2020.04.07.20057299.
COVID-19 now. The urban phase of the U.S. pandemic is leveling somewhat, while the rural phase is accelerating – in part because of food processing and handling industries. The pediatric burden has been surprisingly small, with the multisystem inflammatory disease (MIS-c) in children noted in several hundred cases now being seen across the country.

Next wave? Given ongoing COVID-19 disease, controversy rages about when and how to re-open the country. Regardless how more reopening occurs over the next months, we should expect a next or ongoing COVID-19 wave, particularly given loss of social distancing during social justice protests. A sawtooth disease prevalence pattern is predicted by many experts: a drop in prevalence leading to reopening, leading to scattered prevalence increases and regional if not local restriction tightening, followed by another drop in prevalence. Then “rinse and repeat” until 70% of the population is immune either by disease experience or vaccine-induced immunity, likely sometime in 2021.
Influenza too. A COVID-19 up-cycle is likely during influenza season, although influenza season’s onset could be altered because of whatever social distancing rules are in place in November and December. That said, we need to consider the worst. We have seen what happens if we fail to prepare and then react only after a prevalent respiratory infection has surged into the overall population. Best estimates are that at most 20% of the U.S. population is currently immune to SARS-CoV-2. Given that at least some of that 20% of individuals currently immune to SARS-CoV-2 will lose their neutralizing antibody over the next 4-6 months, we can still expect 70%-80% of the U.S. population to be susceptible to SARS-CoV-2 infection in the fall of 2020.
Pediatric preparedness. As pediatric providers, we have struggled with lower patient loads and dramatic income losses/declines. Many clinics/offices’ attendance remain less than 50% of pre–COVID-19 levels, with necessary furloughs of personnel and spotty office hours. But influenza is coming, and SARS-CoV-2 will not be gone yet. How do we prepare for concurrent influenza and COVID-19?
The annual purchase/administration of influenza vaccine in summer/fall is expensive, time consuming, and logistically difficult even in the best times. Given the loss of income, likely reluctance of patients to come to clinics/offices if COVID-19 is still circulating, and likely need for some form of social distancing during late summer and early fall, how will providers, health departments, and hospitals implement influenza vaccine administration this year?
Minimize double whammy infections. It is easy to understand why we should maximize influenza protection in SARS-CoV-2 vulnerables (elderly or persons with existing comorbidities). But is it as critical for otherwise healthy children? My answer is yes.
Children are not currently known as SARS-CoV-2 vectors, but children are excellent influenza vectors, shedding higher titers for longer than other age groups. As with SARS-CoV-2, influenza exposure is cumulative, i.e., the more intense and more frequently a person is exposed, the more likely that infection/disease will result. So, the fewer who get and can transmit influenza during the COVID-19 pandemic, the fewer people are likely to get a double whammy of SARS-CoV-2 concurrent or in tandem with influenza. Double whammy infections likely would further increase the medical care burden and return us to March-April crisis mode.
One alarming new question is whether recent influenza could make children vulnerable to SARS-CoV-2 and trigger hospitalizations. A surge in pediatric plus adult COVID-19 disease plus a surge in all-ages influenza disease would likely break the medical care system, at least in some areas.
Staggering COVID-19 burden. As of June 8, we have had approximately 2 million SARS-CoV-2 cases with 500,000 hospitalizations and 120,000 deaths. Over the past 10 years, total annual U.S. influenza hospitalizations ranged from 180,000 (2011-2012) to 825,000 (2017-2018). The interquartile range for hospitalization length of stay for influenza is 4-6 days1 vs. 15-23 days2 for SARS-CoV-2. One COVID-19 hospitalization uses hospital resources roughly equal to four influenza hospitalizations. To date COVID-19 hospitalizations have used resources equal to an estimated 1.9 million influenza hospitalizations – over twice the worst influenza season in this century – and we are still on the rise. We are likely not even halfway to truly controlling the U.S. pandemic, so expect another 500,000 hospitalizations – equal to another 1.9 million influenza hospitalizations. Further, pneumonia deaths have skyrocketed this year when COVID-19 was superimposed on the last third of influenza season. One hope is that widespread use of antivirals (for example, new antivirals, convalescent plasma, or other interventions) can reduce length of stay by 30% for COVID-19 hospitalizations, yet even with that the numbers remain grim.
Less influenza disease can free up medical resources. Planning ahead could prevent a bad influenza season (for example, up to 850,000 hospitalizations just for influenza). Can we preemptively use vaccine to reduce influenza hospitalizations below 2011-2012 levels – less than 150,000 hospitalizations? Perhaps, if we start by reducing pediatric influenza.
1. Aim to exceed 75% influenza vaccine uptake in your patients.
a. It is ambitious, but if there was ever a year that needed influenza herd immunity, it is 2020-2021.
2. Review practice/group/institution plans for vaccine purchase and ensure adequate personnel to administer vaccine.
3. Plan safe and efficient processes to vaccinate large numbers in August through November.
a. Consider that routine and influenza vaccines can be given concurrently with the annual uptick in school and sports physical examinations.
b. What social distancing and masking rules will be needed?
i. Will patients need to bring their own masks, or will you supply them?
c. What extra supplies and efforts are needed, e.g. hand sanitizer, new signage, 6-foot interval markings on floors or sidewalks, families calling from parking lot to announce their arrivals, etc.?
d. Remember younger patients need two doses before Dec 1, 2020.
e. Be creative, for example, are parking-lot tents for influenza vaccination feasible?
f. Can we partner with other providers to implement influenza vaccine–specific mass clinics?
Ramping up to give seasonal influenza vaccine in 2020 is daunting. But if we do not prepare, it will be even more difficult. Let’s make this the mildest influenza season in memory by vaccinating more than any time in memory – and by doing so, we can hope to blunt medical care burdens despite ongoing COVID-19 disease.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Kansas City (Mo.). Children’s Mercy receives funding from GlaxoSmithKline, Merck, and Pfizer for vaccine research studies on which Dr. Harrison is an investigator. Email him at [email protected].
References
1.. HCUP Statistical Brief #253. 2019 Oct.
2. medrxiv. 2020 Apr 10. doi: 10.1101/2020.04.07.20057299.
COVID-19 now. The urban phase of the U.S. pandemic is leveling somewhat, while the rural phase is accelerating – in part because of food processing and handling industries. The pediatric burden has been surprisingly small, with the multisystem inflammatory disease (MIS-c) in children noted in several hundred cases now being seen across the country.

Next wave? Given ongoing COVID-19 disease, controversy rages about when and how to re-open the country. Regardless how more reopening occurs over the next months, we should expect a next or ongoing COVID-19 wave, particularly given loss of social distancing during social justice protests. A sawtooth disease prevalence pattern is predicted by many experts: a drop in prevalence leading to reopening, leading to scattered prevalence increases and regional if not local restriction tightening, followed by another drop in prevalence. Then “rinse and repeat” until 70% of the population is immune either by disease experience or vaccine-induced immunity, likely sometime in 2021.
Influenza too. A COVID-19 up-cycle is likely during influenza season, although influenza season’s onset could be altered because of whatever social distancing rules are in place in November and December. That said, we need to consider the worst. We have seen what happens if we fail to prepare and then react only after a prevalent respiratory infection has surged into the overall population. Best estimates are that at most 20% of the U.S. population is currently immune to SARS-CoV-2. Given that at least some of that 20% of individuals currently immune to SARS-CoV-2 will lose their neutralizing antibody over the next 4-6 months, we can still expect 70%-80% of the U.S. population to be susceptible to SARS-CoV-2 infection in the fall of 2020.
Pediatric preparedness. As pediatric providers, we have struggled with lower patient loads and dramatic income losses/declines. Many clinics/offices’ attendance remain less than 50% of pre–COVID-19 levels, with necessary furloughs of personnel and spotty office hours. But influenza is coming, and SARS-CoV-2 will not be gone yet. How do we prepare for concurrent influenza and COVID-19?
The annual purchase/administration of influenza vaccine in summer/fall is expensive, time consuming, and logistically difficult even in the best times. Given the loss of income, likely reluctance of patients to come to clinics/offices if COVID-19 is still circulating, and likely need for some form of social distancing during late summer and early fall, how will providers, health departments, and hospitals implement influenza vaccine administration this year?
Minimize double whammy infections. It is easy to understand why we should maximize influenza protection in SARS-CoV-2 vulnerables (elderly or persons with existing comorbidities). But is it as critical for otherwise healthy children? My answer is yes.
Children are not currently known as SARS-CoV-2 vectors, but children are excellent influenza vectors, shedding higher titers for longer than other age groups. As with SARS-CoV-2, influenza exposure is cumulative, i.e., the more intense and more frequently a person is exposed, the more likely that infection/disease will result. So, the fewer who get and can transmit influenza during the COVID-19 pandemic, the fewer people are likely to get a double whammy of SARS-CoV-2 concurrent or in tandem with influenza. Double whammy infections likely would further increase the medical care burden and return us to March-April crisis mode.
One alarming new question is whether recent influenza could make children vulnerable to SARS-CoV-2 and trigger hospitalizations. A surge in pediatric plus adult COVID-19 disease plus a surge in all-ages influenza disease would likely break the medical care system, at least in some areas.
Staggering COVID-19 burden. As of June 8, we have had approximately 2 million SARS-CoV-2 cases with 500,000 hospitalizations and 120,000 deaths. Over the past 10 years, total annual U.S. influenza hospitalizations ranged from 180,000 (2011-2012) to 825,000 (2017-2018). The interquartile range for hospitalization length of stay for influenza is 4-6 days1 vs. 15-23 days2 for SARS-CoV-2. One COVID-19 hospitalization uses hospital resources roughly equal to four influenza hospitalizations. To date COVID-19 hospitalizations have used resources equal to an estimated 1.9 million influenza hospitalizations – over twice the worst influenza season in this century – and we are still on the rise. We are likely not even halfway to truly controlling the U.S. pandemic, so expect another 500,000 hospitalizations – equal to another 1.9 million influenza hospitalizations. Further, pneumonia deaths have skyrocketed this year when COVID-19 was superimposed on the last third of influenza season. One hope is that widespread use of antivirals (for example, new antivirals, convalescent plasma, or other interventions) can reduce length of stay by 30% for COVID-19 hospitalizations, yet even with that the numbers remain grim.
Less influenza disease can free up medical resources. Planning ahead could prevent a bad influenza season (for example, up to 850,000 hospitalizations just for influenza). Can we preemptively use vaccine to reduce influenza hospitalizations below 2011-2012 levels – less than 150,000 hospitalizations? Perhaps, if we start by reducing pediatric influenza.
1. Aim to exceed 75% influenza vaccine uptake in your patients.
a. It is ambitious, but if there was ever a year that needed influenza herd immunity, it is 2020-2021.
2. Review practice/group/institution plans for vaccine purchase and ensure adequate personnel to administer vaccine.
3. Plan safe and efficient processes to vaccinate large numbers in August through November.
a. Consider that routine and influenza vaccines can be given concurrently with the annual uptick in school and sports physical examinations.
b. What social distancing and masking rules will be needed?
i. Will patients need to bring their own masks, or will you supply them?
c. What extra supplies and efforts are needed, e.g. hand sanitizer, new signage, 6-foot interval markings on floors or sidewalks, families calling from parking lot to announce their arrivals, etc.?
d. Remember younger patients need two doses before Dec 1, 2020.
e. Be creative, for example, are parking-lot tents for influenza vaccination feasible?
f. Can we partner with other providers to implement influenza vaccine–specific mass clinics?
Ramping up to give seasonal influenza vaccine in 2020 is daunting. But if we do not prepare, it will be even more difficult. Let’s make this the mildest influenza season in memory by vaccinating more than any time in memory – and by doing so, we can hope to blunt medical care burdens despite ongoing COVID-19 disease.
Dr. Harrison is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Kansas City (Mo.). Children’s Mercy receives funding from GlaxoSmithKline, Merck, and Pfizer for vaccine research studies on which Dr. Harrison is an investigator. Email him at [email protected].
References
1.. HCUP Statistical Brief #253. 2019 Oct.
2. medrxiv. 2020 Apr 10. doi: 10.1101/2020.04.07.20057299.
Learning the ICU
Although deployment of hospitalists into ICUs during the COVID-19 crisis varies widely, in that sense it reflects the pre-COVID hospital landscape of variable involvement, in which many hospitalists pressed into this role expressed discomfort practicing critical care beyond their scope of training, according to a survey published in the Journal of Hospital Medicine in 2018.1 “Hospitalists frequently deliver critical care services without adequate training or support, most prevalently in rural hospitals,” the authors concluded.
A Critical Care for the Hospitalist Series of resources and lectures developed by Eric Siegal, MD, a pulmonologist in Milwaukee, Wisc., and David Aymond, MD, a hospitalist in Alexandria, La., is available on the SHM website. They recommend that hospitalists trying to get oriented to working in the ICU start with the online courses on fluid resuscitation, mechanical ventilation, and noninvasive ventilation.
“Ninety-five percent of management of COVID-19 patients is nothing other than practicing sound critical care medicine,” Dr. Siegal said. “If you want to take effective care of sick COVID patients, you need to develop good foundational critical care skills and knowledge. Without them, you’re doing stuff without understand it.”
Dr. Aymond also encourages hospitalists to develop a stronger understanding of key physiological concepts by reviewing the critical care clinical topics compiled at SHM’s website.
References
1. Sweigart JR et al. Characterizing hospitalist practice and perceptions of critical care delivery. J Hosp Med. 2018 Jan;13(1):6-12.
Although deployment of hospitalists into ICUs during the COVID-19 crisis varies widely, in that sense it reflects the pre-COVID hospital landscape of variable involvement, in which many hospitalists pressed into this role expressed discomfort practicing critical care beyond their scope of training, according to a survey published in the Journal of Hospital Medicine in 2018.1 “Hospitalists frequently deliver critical care services without adequate training or support, most prevalently in rural hospitals,” the authors concluded.
A Critical Care for the Hospitalist Series of resources and lectures developed by Eric Siegal, MD, a pulmonologist in Milwaukee, Wisc., and David Aymond, MD, a hospitalist in Alexandria, La., is available on the SHM website. They recommend that hospitalists trying to get oriented to working in the ICU start with the online courses on fluid resuscitation, mechanical ventilation, and noninvasive ventilation.
“Ninety-five percent of management of COVID-19 patients is nothing other than practicing sound critical care medicine,” Dr. Siegal said. “If you want to take effective care of sick COVID patients, you need to develop good foundational critical care skills and knowledge. Without them, you’re doing stuff without understand it.”
Dr. Aymond also encourages hospitalists to develop a stronger understanding of key physiological concepts by reviewing the critical care clinical topics compiled at SHM’s website.
References
1. Sweigart JR et al. Characterizing hospitalist practice and perceptions of critical care delivery. J Hosp Med. 2018 Jan;13(1):6-12.
Although deployment of hospitalists into ICUs during the COVID-19 crisis varies widely, in that sense it reflects the pre-COVID hospital landscape of variable involvement, in which many hospitalists pressed into this role expressed discomfort practicing critical care beyond their scope of training, according to a survey published in the Journal of Hospital Medicine in 2018.1 “Hospitalists frequently deliver critical care services without adequate training or support, most prevalently in rural hospitals,” the authors concluded.
A Critical Care for the Hospitalist Series of resources and lectures developed by Eric Siegal, MD, a pulmonologist in Milwaukee, Wisc., and David Aymond, MD, a hospitalist in Alexandria, La., is available on the SHM website. They recommend that hospitalists trying to get oriented to working in the ICU start with the online courses on fluid resuscitation, mechanical ventilation, and noninvasive ventilation.
“Ninety-five percent of management of COVID-19 patients is nothing other than practicing sound critical care medicine,” Dr. Siegal said. “If you want to take effective care of sick COVID patients, you need to develop good foundational critical care skills and knowledge. Without them, you’re doing stuff without understand it.”
Dr. Aymond also encourages hospitalists to develop a stronger understanding of key physiological concepts by reviewing the critical care clinical topics compiled at SHM’s website.
References
1. Sweigart JR et al. Characterizing hospitalist practice and perceptions of critical care delivery. J Hosp Med. 2018 Jan;13(1):6-12.
For COVID-19 plus diabetes, glycemic control tops treatment list
Optimizing glycemic control “is the key to overall treatment in people with diabetes and COVID-19,” said Antonio Ceriello, MD, during a June 5 webinar sponsored by Harvard Medical School, Boston.
Dr. Ceriello, a research consultant with the Italian Ministry of Health, IRCCS Multi-Medica, Milan, highlighted a recent study that examined the association of blood glucose control and outcomes in COVID-19 patients with preexisting type 2 diabetes.
Among 7,000 cases of COVID-19, type 2 diabetes correlated with a higher death rate. However, those with well-controlled blood glucose (upper limit ≤10 mmol/L) had a survival rate of 98.9%, compared with just 11% among those with poorly controlled blood glucose (upper limit >10 mmol/L), a reduction in risk of 86% (adjusted hazard ratio, 0.14; Cell Metab. 2020 May 1. doi: 10.1016/j.cmet.2020.04.021).
Clinicians should also consider the possible side effects of hypoglycemic agents in the evolution of this disease. This is true of all patients, not just diabetes patients, Dr. Ceriello said. “We have data showing that ... hyperglycemia contributes directly to worsening the prognosis of COVID-19 independent of the presence of diabetes.”
One study found that the glycosylation of ACE-2 played an important role in allowing cellular entry of the virus (Am J Physiol Endocrinol Metab. 2020 Mar 31;318:E736-41). “This is something that could be related to hyperglycemia,” he added.
Another risk factor is thrombosis, a clear contributor to death rates in COVID-19. Research on thrombosis incidence in COVID-19 patients with diabetes reported higher levels of D-dimer levels in people with diabetes, especially among those who couldn’t manage their disease.
Tying all of these factors together, Dr. Ceriello discussed how ACE-2 glycosylation, in combination with other factors in SARS-CoV-2 infection, could lead to hyperglycemia, thrombosis, and subsequently multiorgan damage in diabetes patients.
Other research has associated higher HbA1c levels (mean HbA1c, 7.5%) with higher mortality risk in COVID-19 patients, said another speaker, Linong Ji, MD, director for endocrinology and metabolism at Peking University People’s Hospital, Beijing, and director of Peking University’s Diabetes Center. Proper guidance is key to ensuring early detection of hyperglycemic crisis in people with diabetes, advised Dr. Ji.
Global management of diabetes in SARS-CoV-2 patients is “quite challenging,” given that most patients don’t have their diabetes under control, said host and moderator A. Enrique Caballero, MD, an endocrinologist/investigator in the division of endocrinology, diabetes, and hypertension and division of global health equity at Brigham and Women’s Hospital, Boston. “They are not meeting treatment targets for cholesterol or glucose control. So we’re not managing optimal care. And now on top of this, we have COVID-19.”
Optimizing glycemic control “is the key to overall treatment in people with diabetes and COVID-19,” said Antonio Ceriello, MD, during a June 5 webinar sponsored by Harvard Medical School, Boston.
Dr. Ceriello, a research consultant with the Italian Ministry of Health, IRCCS Multi-Medica, Milan, highlighted a recent study that examined the association of blood glucose control and outcomes in COVID-19 patients with preexisting type 2 diabetes.
Among 7,000 cases of COVID-19, type 2 diabetes correlated with a higher death rate. However, those with well-controlled blood glucose (upper limit ≤10 mmol/L) had a survival rate of 98.9%, compared with just 11% among those with poorly controlled blood glucose (upper limit >10 mmol/L), a reduction in risk of 86% (adjusted hazard ratio, 0.14; Cell Metab. 2020 May 1. doi: 10.1016/j.cmet.2020.04.021).
Clinicians should also consider the possible side effects of hypoglycemic agents in the evolution of this disease. This is true of all patients, not just diabetes patients, Dr. Ceriello said. “We have data showing that ... hyperglycemia contributes directly to worsening the prognosis of COVID-19 independent of the presence of diabetes.”
One study found that the glycosylation of ACE-2 played an important role in allowing cellular entry of the virus (Am J Physiol Endocrinol Metab. 2020 Mar 31;318:E736-41). “This is something that could be related to hyperglycemia,” he added.
Another risk factor is thrombosis, a clear contributor to death rates in COVID-19. Research on thrombosis incidence in COVID-19 patients with diabetes reported higher levels of D-dimer levels in people with diabetes, especially among those who couldn’t manage their disease.
Tying all of these factors together, Dr. Ceriello discussed how ACE-2 glycosylation, in combination with other factors in SARS-CoV-2 infection, could lead to hyperglycemia, thrombosis, and subsequently multiorgan damage in diabetes patients.
Other research has associated higher HbA1c levels (mean HbA1c, 7.5%) with higher mortality risk in COVID-19 patients, said another speaker, Linong Ji, MD, director for endocrinology and metabolism at Peking University People’s Hospital, Beijing, and director of Peking University’s Diabetes Center. Proper guidance is key to ensuring early detection of hyperglycemic crisis in people with diabetes, advised Dr. Ji.
Global management of diabetes in SARS-CoV-2 patients is “quite challenging,” given that most patients don’t have their diabetes under control, said host and moderator A. Enrique Caballero, MD, an endocrinologist/investigator in the division of endocrinology, diabetes, and hypertension and division of global health equity at Brigham and Women’s Hospital, Boston. “They are not meeting treatment targets for cholesterol or glucose control. So we’re not managing optimal care. And now on top of this, we have COVID-19.”
Optimizing glycemic control “is the key to overall treatment in people with diabetes and COVID-19,” said Antonio Ceriello, MD, during a June 5 webinar sponsored by Harvard Medical School, Boston.
Dr. Ceriello, a research consultant with the Italian Ministry of Health, IRCCS Multi-Medica, Milan, highlighted a recent study that examined the association of blood glucose control and outcomes in COVID-19 patients with preexisting type 2 diabetes.
Among 7,000 cases of COVID-19, type 2 diabetes correlated with a higher death rate. However, those with well-controlled blood glucose (upper limit ≤10 mmol/L) had a survival rate of 98.9%, compared with just 11% among those with poorly controlled blood glucose (upper limit >10 mmol/L), a reduction in risk of 86% (adjusted hazard ratio, 0.14; Cell Metab. 2020 May 1. doi: 10.1016/j.cmet.2020.04.021).
Clinicians should also consider the possible side effects of hypoglycemic agents in the evolution of this disease. This is true of all patients, not just diabetes patients, Dr. Ceriello said. “We have data showing that ... hyperglycemia contributes directly to worsening the prognosis of COVID-19 independent of the presence of diabetes.”
One study found that the glycosylation of ACE-2 played an important role in allowing cellular entry of the virus (Am J Physiol Endocrinol Metab. 2020 Mar 31;318:E736-41). “This is something that could be related to hyperglycemia,” he added.
Another risk factor is thrombosis, a clear contributor to death rates in COVID-19. Research on thrombosis incidence in COVID-19 patients with diabetes reported higher levels of D-dimer levels in people with diabetes, especially among those who couldn’t manage their disease.
Tying all of these factors together, Dr. Ceriello discussed how ACE-2 glycosylation, in combination with other factors in SARS-CoV-2 infection, could lead to hyperglycemia, thrombosis, and subsequently multiorgan damage in diabetes patients.
Other research has associated higher HbA1c levels (mean HbA1c, 7.5%) with higher mortality risk in COVID-19 patients, said another speaker, Linong Ji, MD, director for endocrinology and metabolism at Peking University People’s Hospital, Beijing, and director of Peking University’s Diabetes Center. Proper guidance is key to ensuring early detection of hyperglycemic crisis in people with diabetes, advised Dr. Ji.
Global management of diabetes in SARS-CoV-2 patients is “quite challenging,” given that most patients don’t have their diabetes under control, said host and moderator A. Enrique Caballero, MD, an endocrinologist/investigator in the division of endocrinology, diabetes, and hypertension and division of global health equity at Brigham and Women’s Hospital, Boston. “They are not meeting treatment targets for cholesterol or glucose control. So we’re not managing optimal care. And now on top of this, we have COVID-19.”
Secondary infections common in COVID-19, implications unclear
but at this point, most pulmonologists aren’t sure what to make of this understudied phenomenon.
“We really do not understand the implications of secondary infections on outcomes in COVID-19 patients,” David L. Bowton, MD, FCCP, said in an interview. “In most early reports the incidence of secondary infections was much higher in patients dying from COVID-19, compared to survivors, but it isn’t clear whether this indicates that the secondary infection itself led to excess mortality or was more a marker of the severity of the COVID-19 infection.
“Further, details of the diagnostic criteria used, the microbiology, and the appropriateness of treatment of these secondary infections has not generally been included in these reports,” added Dr. Bowton, a pulmonologist and professor emeritus of critical care anesthesiology at Wake Forest University, Winston-Salem, N.C.
One such early retrospective cohort study included 191 COVID-19 patients in Wuhan, China. Of the 54 who died in hospital, half had secondary bacterial lung infections (Lancet. 2020 Mar 28;395[10229]:1054-62). That comes as no surprise to U.S. pulmonologists, who learned back in their training that many deaths during the so-called Spanish influenza epidemic of 1918-1920 were actually caused by secondary pneumonia involving Staphylococcus aureus, commented Daniel L. Ouellette, MD, FCCP, associate director of medical critical care at Henry Ford Hospital, Detroit.
“Critically ill patients are highly susceptible to secondary infections regardless of the cause of the patient’s critical illness,” he noted in an interview. “Recent reports of secondary infections in patients critically ill from COVID-19 are interesting but should be considered in this context. To confirm that COVID-19 patients have a different, or increased, risk of infection at specific sites or from specific agents will require careful study.”
That will be no easy matter given the challenges of obtaining bronchoalveolar lavage samples in mechanically ventilated patients with COVID-19, according to Eric J. Gartman, MD, FCCP, a pulmonologist at Brown University, Providence, R.I., and director of the pulmonary function laboratory at the Providence Veterans Affairs Medical Center.
“Unfortunately, many of the invasive modalities that are typically employed to help diagnose secondary infections in critically ill patients are being severely limited or even prohibited in COVID-19 patients due to infection control measures,” he said. As a result, Dr. Gartman noted, intensivists are often resorting to empiric broad-spectrum antimicrobial therapy in patients with severe COVID-19 and are without ready access to the bacterial cultures which might otherwise permit later treatment de-escalation or retargeting.
Among the myriad areas of uncertainty regarding COVID-19 is the proportion of bacterial coinfections that are hospital acquired. Given the lengthy duration of invasive mechanical ventilation in patients with severe COVID-19 – a mean of 9.1 days in the United Kingdom – the chances of hospital-acquired infection are likely substantial. Moreover, a recent single-center U.K. study involving microbiologic testing in 195 consecutive patients newly hospitalized for COVID-19 reported that community-acquired bacterial infection was uncommon: Just 4% of patients had pneumococcal coinfection at hospital admission, and S. aureus wasn’t detected in anyone (Lancet. 2020;1:362. doi:10.1016/S2666-5247[20]30036-7). French investigators have reported detecting putative invasive pulmonary aspergillosis in nearly one-third of a small series of 27 consecutive mechanically ventilated COVID-19 patients (Lancet Resp Med. 2020; 8[6]:e48-9). Dr. Gartman said the diagnostic testing methods utilized in this and similar reports haven’t been prospectively validated in COVID-19. The testing methods may not indicate invasive Aspergillus infection in this population with a high degree of certainty, since they have previously been performed mainly in patients with hematologic malignancies.
“Although there is nothing definitive regarding this research, as a practicing critical care doctor one should respect these findings and consider this secondary diagnosis if the supporting clinical data is positive, especially given that the mortality risk in this population is high,” he advised.
Dr. Bowton said that he and his fellow intensivists at Wake Forest Baptist Health don’t routinely screen COVID-19 patients for secondary bacterial or fungal infections. And in talking with colleagues around the country, it’s his impression that most have similarly elected not to do so.
“However, our clinical index of suspicion for secondary infections is heightened and, if triggered, will initiate a search for and treatment of these secondary infections,” Dr. Bowton said.
but at this point, most pulmonologists aren’t sure what to make of this understudied phenomenon.
“We really do not understand the implications of secondary infections on outcomes in COVID-19 patients,” David L. Bowton, MD, FCCP, said in an interview. “In most early reports the incidence of secondary infections was much higher in patients dying from COVID-19, compared to survivors, but it isn’t clear whether this indicates that the secondary infection itself led to excess mortality or was more a marker of the severity of the COVID-19 infection.
“Further, details of the diagnostic criteria used, the microbiology, and the appropriateness of treatment of these secondary infections has not generally been included in these reports,” added Dr. Bowton, a pulmonologist and professor emeritus of critical care anesthesiology at Wake Forest University, Winston-Salem, N.C.
One such early retrospective cohort study included 191 COVID-19 patients in Wuhan, China. Of the 54 who died in hospital, half had secondary bacterial lung infections (Lancet. 2020 Mar 28;395[10229]:1054-62). That comes as no surprise to U.S. pulmonologists, who learned back in their training that many deaths during the so-called Spanish influenza epidemic of 1918-1920 were actually caused by secondary pneumonia involving Staphylococcus aureus, commented Daniel L. Ouellette, MD, FCCP, associate director of medical critical care at Henry Ford Hospital, Detroit.
“Critically ill patients are highly susceptible to secondary infections regardless of the cause of the patient’s critical illness,” he noted in an interview. “Recent reports of secondary infections in patients critically ill from COVID-19 are interesting but should be considered in this context. To confirm that COVID-19 patients have a different, or increased, risk of infection at specific sites or from specific agents will require careful study.”
That will be no easy matter given the challenges of obtaining bronchoalveolar lavage samples in mechanically ventilated patients with COVID-19, according to Eric J. Gartman, MD, FCCP, a pulmonologist at Brown University, Providence, R.I., and director of the pulmonary function laboratory at the Providence Veterans Affairs Medical Center.
“Unfortunately, many of the invasive modalities that are typically employed to help diagnose secondary infections in critically ill patients are being severely limited or even prohibited in COVID-19 patients due to infection control measures,” he said. As a result, Dr. Gartman noted, intensivists are often resorting to empiric broad-spectrum antimicrobial therapy in patients with severe COVID-19 and are without ready access to the bacterial cultures which might otherwise permit later treatment de-escalation or retargeting.
Among the myriad areas of uncertainty regarding COVID-19 is the proportion of bacterial coinfections that are hospital acquired. Given the lengthy duration of invasive mechanical ventilation in patients with severe COVID-19 – a mean of 9.1 days in the United Kingdom – the chances of hospital-acquired infection are likely substantial. Moreover, a recent single-center U.K. study involving microbiologic testing in 195 consecutive patients newly hospitalized for COVID-19 reported that community-acquired bacterial infection was uncommon: Just 4% of patients had pneumococcal coinfection at hospital admission, and S. aureus wasn’t detected in anyone (Lancet. 2020;1:362. doi:10.1016/S2666-5247[20]30036-7). French investigators have reported detecting putative invasive pulmonary aspergillosis in nearly one-third of a small series of 27 consecutive mechanically ventilated COVID-19 patients (Lancet Resp Med. 2020; 8[6]:e48-9). Dr. Gartman said the diagnostic testing methods utilized in this and similar reports haven’t been prospectively validated in COVID-19. The testing methods may not indicate invasive Aspergillus infection in this population with a high degree of certainty, since they have previously been performed mainly in patients with hematologic malignancies.
“Although there is nothing definitive regarding this research, as a practicing critical care doctor one should respect these findings and consider this secondary diagnosis if the supporting clinical data is positive, especially given that the mortality risk in this population is high,” he advised.
Dr. Bowton said that he and his fellow intensivists at Wake Forest Baptist Health don’t routinely screen COVID-19 patients for secondary bacterial or fungal infections. And in talking with colleagues around the country, it’s his impression that most have similarly elected not to do so.
“However, our clinical index of suspicion for secondary infections is heightened and, if triggered, will initiate a search for and treatment of these secondary infections,” Dr. Bowton said.
but at this point, most pulmonologists aren’t sure what to make of this understudied phenomenon.
“We really do not understand the implications of secondary infections on outcomes in COVID-19 patients,” David L. Bowton, MD, FCCP, said in an interview. “In most early reports the incidence of secondary infections was much higher in patients dying from COVID-19, compared to survivors, but it isn’t clear whether this indicates that the secondary infection itself led to excess mortality or was more a marker of the severity of the COVID-19 infection.
“Further, details of the diagnostic criteria used, the microbiology, and the appropriateness of treatment of these secondary infections has not generally been included in these reports,” added Dr. Bowton, a pulmonologist and professor emeritus of critical care anesthesiology at Wake Forest University, Winston-Salem, N.C.
One such early retrospective cohort study included 191 COVID-19 patients in Wuhan, China. Of the 54 who died in hospital, half had secondary bacterial lung infections (Lancet. 2020 Mar 28;395[10229]:1054-62). That comes as no surprise to U.S. pulmonologists, who learned back in their training that many deaths during the so-called Spanish influenza epidemic of 1918-1920 were actually caused by secondary pneumonia involving Staphylococcus aureus, commented Daniel L. Ouellette, MD, FCCP, associate director of medical critical care at Henry Ford Hospital, Detroit.
“Critically ill patients are highly susceptible to secondary infections regardless of the cause of the patient’s critical illness,” he noted in an interview. “Recent reports of secondary infections in patients critically ill from COVID-19 are interesting but should be considered in this context. To confirm that COVID-19 patients have a different, or increased, risk of infection at specific sites or from specific agents will require careful study.”
That will be no easy matter given the challenges of obtaining bronchoalveolar lavage samples in mechanically ventilated patients with COVID-19, according to Eric J. Gartman, MD, FCCP, a pulmonologist at Brown University, Providence, R.I., and director of the pulmonary function laboratory at the Providence Veterans Affairs Medical Center.
“Unfortunately, many of the invasive modalities that are typically employed to help diagnose secondary infections in critically ill patients are being severely limited or even prohibited in COVID-19 patients due to infection control measures,” he said. As a result, Dr. Gartman noted, intensivists are often resorting to empiric broad-spectrum antimicrobial therapy in patients with severe COVID-19 and are without ready access to the bacterial cultures which might otherwise permit later treatment de-escalation or retargeting.
Among the myriad areas of uncertainty regarding COVID-19 is the proportion of bacterial coinfections that are hospital acquired. Given the lengthy duration of invasive mechanical ventilation in patients with severe COVID-19 – a mean of 9.1 days in the United Kingdom – the chances of hospital-acquired infection are likely substantial. Moreover, a recent single-center U.K. study involving microbiologic testing in 195 consecutive patients newly hospitalized for COVID-19 reported that community-acquired bacterial infection was uncommon: Just 4% of patients had pneumococcal coinfection at hospital admission, and S. aureus wasn’t detected in anyone (Lancet. 2020;1:362. doi:10.1016/S2666-5247[20]30036-7). French investigators have reported detecting putative invasive pulmonary aspergillosis in nearly one-third of a small series of 27 consecutive mechanically ventilated COVID-19 patients (Lancet Resp Med. 2020; 8[6]:e48-9). Dr. Gartman said the diagnostic testing methods utilized in this and similar reports haven’t been prospectively validated in COVID-19. The testing methods may not indicate invasive Aspergillus infection in this population with a high degree of certainty, since they have previously been performed mainly in patients with hematologic malignancies.
“Although there is nothing definitive regarding this research, as a practicing critical care doctor one should respect these findings and consider this secondary diagnosis if the supporting clinical data is positive, especially given that the mortality risk in this population is high,” he advised.
Dr. Bowton said that he and his fellow intensivists at Wake Forest Baptist Health don’t routinely screen COVID-19 patients for secondary bacterial or fungal infections. And in talking with colleagues around the country, it’s his impression that most have similarly elected not to do so.
“However, our clinical index of suspicion for secondary infections is heightened and, if triggered, will initiate a search for and treatment of these secondary infections,” Dr. Bowton said.
Organoid model unveils response to Shiga toxin
The study explored new territory in Shiga toxin research, enabled by the use of human intestinal organoids (HIOs), reported lead author Suman Pradhan, PhD, of the University of Cincinnati, and colleagues.
Each year, Shiga toxin–producing Escherichia coli infections cause approximately 3 million cases of bloody diarrheal disease, with about 4,000 of those patients developing the life-threatening complication of hemolytic uremic syndrome (HUS), the investigators wrote in Cellular and Molecular Gastroenterology and Hepatology.
But little is known about the underlying biological processes driving Shiga-induced disease.
“Developing effective interventions for disease resulting from Shiga toxin is exacerbated by a lack of tractable model systems,” the investigators wrote. “Mice do not develop the symptoms characteristic of HUS, and the murine intestinal tract is resistant to Shiga toxin.”
To overcome this obstacle, Dr. Pradhan and colleagues turned to HIOs, which are grown in culture by directing differentiation of pluripotent stem cells. HIOs represent the small bowel, complete with a lumen surrounded by epithelial and mesenchymal layers that include typical cell types, such as goblet cells and myofibroblasts. The model is made more realistic by transplantation into mice, where it grows under the kidney capsule to form crypts, structured villi, and proliferating progenitor zones. And HIOs grown with neuronal precursors develop an enteric nervous system, complete with functional peristalsis.
For the present study, the investigators evaluated the effect of Shiga toxin on HIOs both in culture and after transplantation into mice.
First, they demonstrated that HIOs in culture expressed glycolipid Gb3, the Shiga toxin receptor. “Reports regarding expression of glycolipid Gb3 ... on human intestine have been inconsistent,” the investigators noted. “For negative reports, the inability to detect Gb3 could be owing to technical limitations.”
Next, Dr. Pradhan and colleagues showed that HIOs were susceptible to Shiga toxin whether it be delivered lumenally or basolaterally, which respectively represent intestinal exposure and exposure via circulating toxin or after breakdown of the epithelial barrier. Leakage from the lumen was observed with both Shiga toxin 1 (Stx1) and 2 (Stx2). Subsequent testing involved only Stx2, as this form is more relevant to human disease.
In addition to lumenal leakage, Stx2 exposure caused significant transcriptional up-regulation of multiple gene families, including those involved in cellular transport and metabolic processes. Increased expression also was observed for epithelial structural proteins, lineage-specific proteins, factors involved in mucus layer formation and stabilization, and cytokines interleukin-18 and CCL15.
In both epithelial and mesenchymal layers, transcriptional changes were accompanied by cellular necrosis and apoptosis, and, to a greater degree with interstitial exposure, cellular proliferation.
With lumenal exposure, mesenchymal necrosis was observed before loss of epithelial barrier function, indicating toxin access to mesenchymal cells. This phenomenon was explained by transcytosis, which the investigators observed in two-dimensional monolayers of enteroids grown in Transwells.
“[Shiga toxin] was transferred from the apical to the basolateral surface in the absence of loss of epithelial barrier function,” the investigators wrote, noting that this finding explains how Shiga toxin can quickly access the circulatory system, and from there damage the kidneys and brain, as seen in cases of HUS.
Mice with transplanted HIOs, and those receiving HIOs with an enteric nervous system (HIO + ENS), lost weight when organoids were injected with 10 ng of Stx2. Mice with HIO + ENS transplants developed more severe responses, prompting closer analysis.
Postmortem histologic examination of HIO + ENS transplants revealed epithelial damage and blood accumulation in the mesenchyme and villi. Additional staining showed signs of apoptosis and mesenchymal-epithelial transition.
Dr. Pradham and colleagues suggested that their findings could inform therapeutic research.
“If preventing cellular death is to be an effective intervention, it is likely that both necrosis and apoptosis need to be targeted,” the investigators wrote.
More generally, the study supports the use of HIOs as a disease model for future investigations.
“The advent of stem cell–derived human tissue models, both in vitro and in vivo, has a tremendous potential to increase our understanding of Shiga toxin disease and lead to development of therapeutic interventions,” the investigators concluded.
The study was funded by the National Institutes of Health, the Center for Clinical and Translational Science, the National Institute of Diabetes and Digestive and Kidney Diseases, and others. The investigators disclosed no conflicts of interest.
SOURCE: Pradhan S et al. Cell Mol Gastroenterol Hepatol. 2020 Mar 5. doi: 10.1016/j.jcmgh.2020.02.006.
Limited therapies exist to mitigate the life-threatening sequelae of Shiga toxin (Stx)–producing Escherichia coli (STEC) infections. Stx continues to be a leading cause of hemolytic uremic syndrome and can devastate the kidneys, central nervous system, and other vital organs. Conflicting results from animal models and cell lines have left important questions unanswered, slowing therapy development. This study by Pradhan et al. takes advantage of the human intestinal organoid system to provide insight to questions pertinent to understanding Stx mechanism of action. Importantly, the authors find that intestinal epithelial cells (IECs) are a direct target of Stx and express the Stx receptor, Gb3, a point that had not been previously well established. They further confirm that IECs efficiently transport Stx from the apical to basolateral surface, before barrier integrity is compromised. This likely allows Stx to rapidly access circulation and other affected organs to cause disease.
Nicole Maloney Belle, MD, PhD, is an instructor of medicine, division of gastroenterology and hepatology, at the University of Pennsylvania, Philadelphia. She has no conflicts.
Limited therapies exist to mitigate the life-threatening sequelae of Shiga toxin (Stx)–producing Escherichia coli (STEC) infections. Stx continues to be a leading cause of hemolytic uremic syndrome and can devastate the kidneys, central nervous system, and other vital organs. Conflicting results from animal models and cell lines have left important questions unanswered, slowing therapy development. This study by Pradhan et al. takes advantage of the human intestinal organoid system to provide insight to questions pertinent to understanding Stx mechanism of action. Importantly, the authors find that intestinal epithelial cells (IECs) are a direct target of Stx and express the Stx receptor, Gb3, a point that had not been previously well established. They further confirm that IECs efficiently transport Stx from the apical to basolateral surface, before barrier integrity is compromised. This likely allows Stx to rapidly access circulation and other affected organs to cause disease.
Nicole Maloney Belle, MD, PhD, is an instructor of medicine, division of gastroenterology and hepatology, at the University of Pennsylvania, Philadelphia. She has no conflicts.
Limited therapies exist to mitigate the life-threatening sequelae of Shiga toxin (Stx)–producing Escherichia coli (STEC) infections. Stx continues to be a leading cause of hemolytic uremic syndrome and can devastate the kidneys, central nervous system, and other vital organs. Conflicting results from animal models and cell lines have left important questions unanswered, slowing therapy development. This study by Pradhan et al. takes advantage of the human intestinal organoid system to provide insight to questions pertinent to understanding Stx mechanism of action. Importantly, the authors find that intestinal epithelial cells (IECs) are a direct target of Stx and express the Stx receptor, Gb3, a point that had not been previously well established. They further confirm that IECs efficiently transport Stx from the apical to basolateral surface, before barrier integrity is compromised. This likely allows Stx to rapidly access circulation and other affected organs to cause disease.
Nicole Maloney Belle, MD, PhD, is an instructor of medicine, division of gastroenterology and hepatology, at the University of Pennsylvania, Philadelphia. She has no conflicts.
The study explored new territory in Shiga toxin research, enabled by the use of human intestinal organoids (HIOs), reported lead author Suman Pradhan, PhD, of the University of Cincinnati, and colleagues.
Each year, Shiga toxin–producing Escherichia coli infections cause approximately 3 million cases of bloody diarrheal disease, with about 4,000 of those patients developing the life-threatening complication of hemolytic uremic syndrome (HUS), the investigators wrote in Cellular and Molecular Gastroenterology and Hepatology.
But little is known about the underlying biological processes driving Shiga-induced disease.
“Developing effective interventions for disease resulting from Shiga toxin is exacerbated by a lack of tractable model systems,” the investigators wrote. “Mice do not develop the symptoms characteristic of HUS, and the murine intestinal tract is resistant to Shiga toxin.”
To overcome this obstacle, Dr. Pradhan and colleagues turned to HIOs, which are grown in culture by directing differentiation of pluripotent stem cells. HIOs represent the small bowel, complete with a lumen surrounded by epithelial and mesenchymal layers that include typical cell types, such as goblet cells and myofibroblasts. The model is made more realistic by transplantation into mice, where it grows under the kidney capsule to form crypts, structured villi, and proliferating progenitor zones. And HIOs grown with neuronal precursors develop an enteric nervous system, complete with functional peristalsis.
For the present study, the investigators evaluated the effect of Shiga toxin on HIOs both in culture and after transplantation into mice.
First, they demonstrated that HIOs in culture expressed glycolipid Gb3, the Shiga toxin receptor. “Reports regarding expression of glycolipid Gb3 ... on human intestine have been inconsistent,” the investigators noted. “For negative reports, the inability to detect Gb3 could be owing to technical limitations.”
Next, Dr. Pradhan and colleagues showed that HIOs were susceptible to Shiga toxin whether it be delivered lumenally or basolaterally, which respectively represent intestinal exposure and exposure via circulating toxin or after breakdown of the epithelial barrier. Leakage from the lumen was observed with both Shiga toxin 1 (Stx1) and 2 (Stx2). Subsequent testing involved only Stx2, as this form is more relevant to human disease.
In addition to lumenal leakage, Stx2 exposure caused significant transcriptional up-regulation of multiple gene families, including those involved in cellular transport and metabolic processes. Increased expression also was observed for epithelial structural proteins, lineage-specific proteins, factors involved in mucus layer formation and stabilization, and cytokines interleukin-18 and CCL15.
In both epithelial and mesenchymal layers, transcriptional changes were accompanied by cellular necrosis and apoptosis, and, to a greater degree with interstitial exposure, cellular proliferation.
With lumenal exposure, mesenchymal necrosis was observed before loss of epithelial barrier function, indicating toxin access to mesenchymal cells. This phenomenon was explained by transcytosis, which the investigators observed in two-dimensional monolayers of enteroids grown in Transwells.
“[Shiga toxin] was transferred from the apical to the basolateral surface in the absence of loss of epithelial barrier function,” the investigators wrote, noting that this finding explains how Shiga toxin can quickly access the circulatory system, and from there damage the kidneys and brain, as seen in cases of HUS.
Mice with transplanted HIOs, and those receiving HIOs with an enteric nervous system (HIO + ENS), lost weight when organoids were injected with 10 ng of Stx2. Mice with HIO + ENS transplants developed more severe responses, prompting closer analysis.
Postmortem histologic examination of HIO + ENS transplants revealed epithelial damage and blood accumulation in the mesenchyme and villi. Additional staining showed signs of apoptosis and mesenchymal-epithelial transition.
Dr. Pradham and colleagues suggested that their findings could inform therapeutic research.
“If preventing cellular death is to be an effective intervention, it is likely that both necrosis and apoptosis need to be targeted,” the investigators wrote.
More generally, the study supports the use of HIOs as a disease model for future investigations.
“The advent of stem cell–derived human tissue models, both in vitro and in vivo, has a tremendous potential to increase our understanding of Shiga toxin disease and lead to development of therapeutic interventions,” the investigators concluded.
The study was funded by the National Institutes of Health, the Center for Clinical and Translational Science, the National Institute of Diabetes and Digestive and Kidney Diseases, and others. The investigators disclosed no conflicts of interest.
SOURCE: Pradhan S et al. Cell Mol Gastroenterol Hepatol. 2020 Mar 5. doi: 10.1016/j.jcmgh.2020.02.006.
The study explored new territory in Shiga toxin research, enabled by the use of human intestinal organoids (HIOs), reported lead author Suman Pradhan, PhD, of the University of Cincinnati, and colleagues.
Each year, Shiga toxin–producing Escherichia coli infections cause approximately 3 million cases of bloody diarrheal disease, with about 4,000 of those patients developing the life-threatening complication of hemolytic uremic syndrome (HUS), the investigators wrote in Cellular and Molecular Gastroenterology and Hepatology.
But little is known about the underlying biological processes driving Shiga-induced disease.
“Developing effective interventions for disease resulting from Shiga toxin is exacerbated by a lack of tractable model systems,” the investigators wrote. “Mice do not develop the symptoms characteristic of HUS, and the murine intestinal tract is resistant to Shiga toxin.”
To overcome this obstacle, Dr. Pradhan and colleagues turned to HIOs, which are grown in culture by directing differentiation of pluripotent stem cells. HIOs represent the small bowel, complete with a lumen surrounded by epithelial and mesenchymal layers that include typical cell types, such as goblet cells and myofibroblasts. The model is made more realistic by transplantation into mice, where it grows under the kidney capsule to form crypts, structured villi, and proliferating progenitor zones. And HIOs grown with neuronal precursors develop an enteric nervous system, complete with functional peristalsis.
For the present study, the investigators evaluated the effect of Shiga toxin on HIOs both in culture and after transplantation into mice.
First, they demonstrated that HIOs in culture expressed glycolipid Gb3, the Shiga toxin receptor. “Reports regarding expression of glycolipid Gb3 ... on human intestine have been inconsistent,” the investigators noted. “For negative reports, the inability to detect Gb3 could be owing to technical limitations.”
Next, Dr. Pradhan and colleagues showed that HIOs were susceptible to Shiga toxin whether it be delivered lumenally or basolaterally, which respectively represent intestinal exposure and exposure via circulating toxin or after breakdown of the epithelial barrier. Leakage from the lumen was observed with both Shiga toxin 1 (Stx1) and 2 (Stx2). Subsequent testing involved only Stx2, as this form is more relevant to human disease.
In addition to lumenal leakage, Stx2 exposure caused significant transcriptional up-regulation of multiple gene families, including those involved in cellular transport and metabolic processes. Increased expression also was observed for epithelial structural proteins, lineage-specific proteins, factors involved in mucus layer formation and stabilization, and cytokines interleukin-18 and CCL15.
In both epithelial and mesenchymal layers, transcriptional changes were accompanied by cellular necrosis and apoptosis, and, to a greater degree with interstitial exposure, cellular proliferation.
With lumenal exposure, mesenchymal necrosis was observed before loss of epithelial barrier function, indicating toxin access to mesenchymal cells. This phenomenon was explained by transcytosis, which the investigators observed in two-dimensional monolayers of enteroids grown in Transwells.
“[Shiga toxin] was transferred from the apical to the basolateral surface in the absence of loss of epithelial barrier function,” the investigators wrote, noting that this finding explains how Shiga toxin can quickly access the circulatory system, and from there damage the kidneys and brain, as seen in cases of HUS.
Mice with transplanted HIOs, and those receiving HIOs with an enteric nervous system (HIO + ENS), lost weight when organoids were injected with 10 ng of Stx2. Mice with HIO + ENS transplants developed more severe responses, prompting closer analysis.
Postmortem histologic examination of HIO + ENS transplants revealed epithelial damage and blood accumulation in the mesenchyme and villi. Additional staining showed signs of apoptosis and mesenchymal-epithelial transition.
Dr. Pradham and colleagues suggested that their findings could inform therapeutic research.
“If preventing cellular death is to be an effective intervention, it is likely that both necrosis and apoptosis need to be targeted,” the investigators wrote.
More generally, the study supports the use of HIOs as a disease model for future investigations.
“The advent of stem cell–derived human tissue models, both in vitro and in vivo, has a tremendous potential to increase our understanding of Shiga toxin disease and lead to development of therapeutic interventions,” the investigators concluded.
The study was funded by the National Institutes of Health, the Center for Clinical and Translational Science, the National Institute of Diabetes and Digestive and Kidney Diseases, and others. The investigators disclosed no conflicts of interest.
SOURCE: Pradhan S et al. Cell Mol Gastroenterol Hepatol. 2020 Mar 5. doi: 10.1016/j.jcmgh.2020.02.006.
FROM CELLULAR AND MOLECULAR GASTROENTEROLOGY AND HEPATOLOGY


















