User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Could these old drugs help fight COVID-19 and save lives?
Early in the COVID-19 pandemic, entrepreneur and philanthropist Steve Kirsch realized that until we have a vaccine against SARS-CoV-2, we would be at the mercy of this virus. He realized that the fastest and most effective way to reduce COVID-19 fatalities would be to leverage existing drugs to treat patients at the onset of infection — before they become sick.
Medscape spoke with CETF’s chief medical advisor, Lisa Danzig, MD, about the organization’s aim to fund promising research on repurposed drugs to treat COVID-19.
What is CETF trying to do?
Two things: save lives, and get control of this pandemic.
We are facing perhaps the greatest crisis of our lifetime. Doctors who have taken care of patients with COVID are really frustrated about not having anything to offer; they just watch patients die. We want to change that. CETF was founded to find treatments that, when given early, could improve outcomes and avoid catastrophic complications in patients suffering from COVID-19. That means reducing hospitalizations, which can reduce mortality, but it also can mean reducing viral load, and that can have a profound impact on transmission within communities. We are a funding organization — a Band-Aid. We shouldn’t exist, but we do, aiming to close gaps until a coordinated response can get set up.
Tell us about drug repurposing and why you think existing drugs might have a role in mitigating COVID-19 or slowing its transmission.
This disease has two components — the viral infection, and the immunopathology. So the two promising categories of drugs are classical antivirals (or repurposed drugs with antiviral activity), and the immunomodulators. We are mechanism-agnostic. It doesn’t matter what kind of drug it is if it keeps people out of the hospital and prevents chronic morbidity and mortality.
Repurposed drugs are sort of the low-hanging fruit of clinical drugs. The QBI Coronavirus Research Group identified 69 compounds that have theoretical activity against SARS-CoV-2, 29 of which are already FDA-approved drugs. We thought, why don’t we start testing them?
Some people might call this a long shot. Does drug repurposing really work?
Drugmakers don’t test their drugs on every disease they might be effective for. Drug repurposing can work, but if we don’t look, we definitely won’t find anything. The classic repurposed drug is Viagra, a failed hypertension drug. When the studies ended because it didn’t work, the drug company asked patients to send back the unused drugs. The women all returned the drugs, but the men didn’t. And the rest is history.
There’s a long list of potential drugs that can be repurposed, but few are being tested. The famous poster child of a repurposed drug — hydroxychloroquine — has been the subject of more than 250 clinical trials, but the others weren’t getting much attention.
The beauty of a repurposed drug is that if you can get funding and start enrolling patients, you could potentially find out fairly quickly, as early as a few months, if that drug has an antiviral effect or not. These data would help prioritize drugs to be tested in larger confirmatory studies.
Your focus is on early treatment. What’s the rationale for that?
We are focusing on early treatment because it has been overlooked. The attention has been on vaccines and therapeutics for hospitalized patients. But if you are spending $20 billion on potential vaccines and billions more on diagnostics, we need to give proportional resources toward drugs that might actually work, when given early, in preventing severe disease and death.
Early treatment, if successful, would allow us to avoid the severe complications that we are seeing now. If we can find an early treatment with an existing drug, it would be the fastest, most clinically- and cost-effective way to mitigate the impact of COVID-19 and get us on the road to recovery.
How do you get from a potential repurposed drug for COVID-19 to having a therapeutic agent that will save lives?
Most of the studies we are funding are smaller outpatient studies with virologic endpoints. We are looking for a signal that the drug has antiviral activity. We want to know whether a drug works before we spend the money on questions that take a much larger sample size to answer, for example, a big postexposure prophylaxis study. We’d like to see a meaningful signal in proof-of-concept studies, so we can look at a small group of patients with positive tests and see whether their viral load dropped by more than half if they got the drug compared with those who took the placebo. If the drug had an impact on the viral load and shortened the period of infectivity and was safe, these findings would provide justification to spend a lot of money on a large clinical trial. That would probably encourage the NIH and ACTIV [Accelerating COVID-19 Therapeutic Interventions and Vaccines] collaboration to prioritize the drug for one of their big platform trials. That›s what we are aiming for.
CETF isn’t a drug developer — we are a funder for a good proposal to study a repurposed drug. We want to help move the dial — can we get an early yes or an early no? In drug development, we say, “fail fast and fail early.” It’s a numbers game. Only 10% of early candidates will become approved drugs. The value is in the data, whether they are positive or negative — it doesn’t matter. If the study is a definitive “no,” that is just as helpful as a definitive “yes.” Of course, we all want the definitive “yes,” but there are so many things to look at, the “no’s” will help us redirect resources toward what may really help.
You first announced these funding opportunities in April. How is it going so far?
As soon as the website went up, we got 40 applications. Our scientific advisory board, which has expertise from medicinal chemistry and coronavirology to translational and clinical trial expertise, reviewed the applications and prioritized 11 fundable proposals. We are using milestone-based funding; in other words, funding those who are ready to go.
Which drugs are being tested in the funded studies?
One of the earliest grants we supported was Dr David Boulaware’s randomized controlled trial of hydroxychloroquine (NCT 04308668) in 821 asymptomatic patients within 4 days after a high-risk or moderate-risk exposure. That trial did not show any benefit of hydroxychloroquine as postexposure prophylaxis against COVID-19. This trial was important for another reason. It proved the feasibility of a no-contact trial design in the setting of COVID-19, and participants enrolled themselves through a secure Internet-based survey using the Research Electronic Data Capture (REDCap) system.
Camostat, a transmembrane serine protease (TMPRSS2) inhibitor licensed for use in Japan to treat pancreatitis and esophagitis, combined with the antiandrogen bicalutamide, is being explored for early COVID-19 treatment. TMPRSS2 primes the SARS-CoV-2 spike protein to bind to the ACE2 receptor and gain entry to the cell, and has been shown to have antiviral activity. CETF has provided funding support to ongoing trials of Camostat at Yale University and Aarhus University in Denmark.
Another outpatient trial for fluvoxamine, a drug approved in the United States and routinely prescribed for depression, was also partially funded by a CETF grant to Washington University in St. Louis. Fluvoxamine is a serotonin regulator but also activates the sigma-1 receptor, which reduces the body’s immune response to prevent an overactive immune response or cytokine storm, a major cause of clinical deterioration, serious organ damage, and even death from COVID. This trial was recently completed, and the results have been submitted for publication.
Other promising drugs include niclosamide, doxazosin, favipiravir, leronlimab, interferon beta, interferon lambda, and other monoclonal antibodies. New compounds considered to have potential against COVID include a flu drug (MK-4482/EIDD-2801) and GS-441524, a metabolite of the antiviral drug, remdesivir.
Why not just put all of our resources into vaccine development?
We absolutely need a vaccine to control the outbreak and stop the pandemic. However, it’s a long road to finding an effective vaccine, and in the meantime, we need tools to keep people alive. If we can find an antiviral drug that acts early, we can reduce transmission and contribute to outbreak control. All these tools help us get back to normal while we are waiting for a vaccine. The vaccine is only good if we can give it to every susceptible person in the world — which will take longer than 3 years. And there are no guarantees. Remember, we are still waiting for an HIV vaccine.
You are calling on Americans to help. What do you want them to do?
Everyone must participate in the behavioral changes designed to control the outbreak — physical distancing, face-covering, and paying attention to case counts in local areas to enable them to take appropriate precautions. I know people are bored of that message, but we are going to repeat it until we have a vaccine or herd immunity.
This organism is ripping like wildfire through our unimmunized population. Personal behaviors might slow it down, but finding a drug that can be given to people after they’ve been exposed and test positive will have a meaningful impact on helping us get back to normal.
There’s a great spirit of volunteerism — people are constantly asking how they can help. Through us at CETF, we offer three ways that people can help. They can participate as subjects in clinical trials, many of which are ongoing, including clinical trials, surveillance studies, and follow-up studies. They can donate to our fund and help support the research needed to find an effective early treatment. We have a link on our website, TreatEarly.org. And finally, researchers can apply for funding. We think everybody can help in one of these ways by participating in trials, donating, or applying for funding. It’s an all-hands-on-deck moment for our country.
Danzig is the chief medical advisor of the COVID-19 Early Treatment Fund. She has spent more than 20 years in the pharmaceutical industry developing vaccines, diagnostics, and drugs and is currently advising companies and investors.
This article first appeared on Medscape.com.
Early in the COVID-19 pandemic, entrepreneur and philanthropist Steve Kirsch realized that until we have a vaccine against SARS-CoV-2, we would be at the mercy of this virus. He realized that the fastest and most effective way to reduce COVID-19 fatalities would be to leverage existing drugs to treat patients at the onset of infection — before they become sick.
Medscape spoke with CETF’s chief medical advisor, Lisa Danzig, MD, about the organization’s aim to fund promising research on repurposed drugs to treat COVID-19.
What is CETF trying to do?
Two things: save lives, and get control of this pandemic.
We are facing perhaps the greatest crisis of our lifetime. Doctors who have taken care of patients with COVID are really frustrated about not having anything to offer; they just watch patients die. We want to change that. CETF was founded to find treatments that, when given early, could improve outcomes and avoid catastrophic complications in patients suffering from COVID-19. That means reducing hospitalizations, which can reduce mortality, but it also can mean reducing viral load, and that can have a profound impact on transmission within communities. We are a funding organization — a Band-Aid. We shouldn’t exist, but we do, aiming to close gaps until a coordinated response can get set up.
Tell us about drug repurposing and why you think existing drugs might have a role in mitigating COVID-19 or slowing its transmission.
This disease has two components — the viral infection, and the immunopathology. So the two promising categories of drugs are classical antivirals (or repurposed drugs with antiviral activity), and the immunomodulators. We are mechanism-agnostic. It doesn’t matter what kind of drug it is if it keeps people out of the hospital and prevents chronic morbidity and mortality.
Repurposed drugs are sort of the low-hanging fruit of clinical drugs. The QBI Coronavirus Research Group identified 69 compounds that have theoretical activity against SARS-CoV-2, 29 of which are already FDA-approved drugs. We thought, why don’t we start testing them?
Some people might call this a long shot. Does drug repurposing really work?
Drugmakers don’t test their drugs on every disease they might be effective for. Drug repurposing can work, but if we don’t look, we definitely won’t find anything. The classic repurposed drug is Viagra, a failed hypertension drug. When the studies ended because it didn’t work, the drug company asked patients to send back the unused drugs. The women all returned the drugs, but the men didn’t. And the rest is history.
There’s a long list of potential drugs that can be repurposed, but few are being tested. The famous poster child of a repurposed drug — hydroxychloroquine — has been the subject of more than 250 clinical trials, but the others weren’t getting much attention.
The beauty of a repurposed drug is that if you can get funding and start enrolling patients, you could potentially find out fairly quickly, as early as a few months, if that drug has an antiviral effect or not. These data would help prioritize drugs to be tested in larger confirmatory studies.
Your focus is on early treatment. What’s the rationale for that?
We are focusing on early treatment because it has been overlooked. The attention has been on vaccines and therapeutics for hospitalized patients. But if you are spending $20 billion on potential vaccines and billions more on diagnostics, we need to give proportional resources toward drugs that might actually work, when given early, in preventing severe disease and death.
Early treatment, if successful, would allow us to avoid the severe complications that we are seeing now. If we can find an early treatment with an existing drug, it would be the fastest, most clinically- and cost-effective way to mitigate the impact of COVID-19 and get us on the road to recovery.
How do you get from a potential repurposed drug for COVID-19 to having a therapeutic agent that will save lives?
Most of the studies we are funding are smaller outpatient studies with virologic endpoints. We are looking for a signal that the drug has antiviral activity. We want to know whether a drug works before we spend the money on questions that take a much larger sample size to answer, for example, a big postexposure prophylaxis study. We’d like to see a meaningful signal in proof-of-concept studies, so we can look at a small group of patients with positive tests and see whether their viral load dropped by more than half if they got the drug compared with those who took the placebo. If the drug had an impact on the viral load and shortened the period of infectivity and was safe, these findings would provide justification to spend a lot of money on a large clinical trial. That would probably encourage the NIH and ACTIV [Accelerating COVID-19 Therapeutic Interventions and Vaccines] collaboration to prioritize the drug for one of their big platform trials. That›s what we are aiming for.
CETF isn’t a drug developer — we are a funder for a good proposal to study a repurposed drug. We want to help move the dial — can we get an early yes or an early no? In drug development, we say, “fail fast and fail early.” It’s a numbers game. Only 10% of early candidates will become approved drugs. The value is in the data, whether they are positive or negative — it doesn’t matter. If the study is a definitive “no,” that is just as helpful as a definitive “yes.” Of course, we all want the definitive “yes,” but there are so many things to look at, the “no’s” will help us redirect resources toward what may really help.
You first announced these funding opportunities in April. How is it going so far?
As soon as the website went up, we got 40 applications. Our scientific advisory board, which has expertise from medicinal chemistry and coronavirology to translational and clinical trial expertise, reviewed the applications and prioritized 11 fundable proposals. We are using milestone-based funding; in other words, funding those who are ready to go.
Which drugs are being tested in the funded studies?
One of the earliest grants we supported was Dr David Boulaware’s randomized controlled trial of hydroxychloroquine (NCT 04308668) in 821 asymptomatic patients within 4 days after a high-risk or moderate-risk exposure. That trial did not show any benefit of hydroxychloroquine as postexposure prophylaxis against COVID-19. This trial was important for another reason. It proved the feasibility of a no-contact trial design in the setting of COVID-19, and participants enrolled themselves through a secure Internet-based survey using the Research Electronic Data Capture (REDCap) system.
Camostat, a transmembrane serine protease (TMPRSS2) inhibitor licensed for use in Japan to treat pancreatitis and esophagitis, combined with the antiandrogen bicalutamide, is being explored for early COVID-19 treatment. TMPRSS2 primes the SARS-CoV-2 spike protein to bind to the ACE2 receptor and gain entry to the cell, and has been shown to have antiviral activity. CETF has provided funding support to ongoing trials of Camostat at Yale University and Aarhus University in Denmark.
Another outpatient trial for fluvoxamine, a drug approved in the United States and routinely prescribed for depression, was also partially funded by a CETF grant to Washington University in St. Louis. Fluvoxamine is a serotonin regulator but also activates the sigma-1 receptor, which reduces the body’s immune response to prevent an overactive immune response or cytokine storm, a major cause of clinical deterioration, serious organ damage, and even death from COVID. This trial was recently completed, and the results have been submitted for publication.
Other promising drugs include niclosamide, doxazosin, favipiravir, leronlimab, interferon beta, interferon lambda, and other monoclonal antibodies. New compounds considered to have potential against COVID include a flu drug (MK-4482/EIDD-2801) and GS-441524, a metabolite of the antiviral drug, remdesivir.
Why not just put all of our resources into vaccine development?
We absolutely need a vaccine to control the outbreak and stop the pandemic. However, it’s a long road to finding an effective vaccine, and in the meantime, we need tools to keep people alive. If we can find an antiviral drug that acts early, we can reduce transmission and contribute to outbreak control. All these tools help us get back to normal while we are waiting for a vaccine. The vaccine is only good if we can give it to every susceptible person in the world — which will take longer than 3 years. And there are no guarantees. Remember, we are still waiting for an HIV vaccine.
You are calling on Americans to help. What do you want them to do?
Everyone must participate in the behavioral changes designed to control the outbreak — physical distancing, face-covering, and paying attention to case counts in local areas to enable them to take appropriate precautions. I know people are bored of that message, but we are going to repeat it until we have a vaccine or herd immunity.
This organism is ripping like wildfire through our unimmunized population. Personal behaviors might slow it down, but finding a drug that can be given to people after they’ve been exposed and test positive will have a meaningful impact on helping us get back to normal.
There’s a great spirit of volunteerism — people are constantly asking how they can help. Through us at CETF, we offer three ways that people can help. They can participate as subjects in clinical trials, many of which are ongoing, including clinical trials, surveillance studies, and follow-up studies. They can donate to our fund and help support the research needed to find an effective early treatment. We have a link on our website, TreatEarly.org. And finally, researchers can apply for funding. We think everybody can help in one of these ways by participating in trials, donating, or applying for funding. It’s an all-hands-on-deck moment for our country.
Danzig is the chief medical advisor of the COVID-19 Early Treatment Fund. She has spent more than 20 years in the pharmaceutical industry developing vaccines, diagnostics, and drugs and is currently advising companies and investors.
This article first appeared on Medscape.com.
Early in the COVID-19 pandemic, entrepreneur and philanthropist Steve Kirsch realized that until we have a vaccine against SARS-CoV-2, we would be at the mercy of this virus. He realized that the fastest and most effective way to reduce COVID-19 fatalities would be to leverage existing drugs to treat patients at the onset of infection — before they become sick.
Medscape spoke with CETF’s chief medical advisor, Lisa Danzig, MD, about the organization’s aim to fund promising research on repurposed drugs to treat COVID-19.
What is CETF trying to do?
Two things: save lives, and get control of this pandemic.
We are facing perhaps the greatest crisis of our lifetime. Doctors who have taken care of patients with COVID are really frustrated about not having anything to offer; they just watch patients die. We want to change that. CETF was founded to find treatments that, when given early, could improve outcomes and avoid catastrophic complications in patients suffering from COVID-19. That means reducing hospitalizations, which can reduce mortality, but it also can mean reducing viral load, and that can have a profound impact on transmission within communities. We are a funding organization — a Band-Aid. We shouldn’t exist, but we do, aiming to close gaps until a coordinated response can get set up.
Tell us about drug repurposing and why you think existing drugs might have a role in mitigating COVID-19 or slowing its transmission.
This disease has two components — the viral infection, and the immunopathology. So the two promising categories of drugs are classical antivirals (or repurposed drugs with antiviral activity), and the immunomodulators. We are mechanism-agnostic. It doesn’t matter what kind of drug it is if it keeps people out of the hospital and prevents chronic morbidity and mortality.
Repurposed drugs are sort of the low-hanging fruit of clinical drugs. The QBI Coronavirus Research Group identified 69 compounds that have theoretical activity against SARS-CoV-2, 29 of which are already FDA-approved drugs. We thought, why don’t we start testing them?
Some people might call this a long shot. Does drug repurposing really work?
Drugmakers don’t test their drugs on every disease they might be effective for. Drug repurposing can work, but if we don’t look, we definitely won’t find anything. The classic repurposed drug is Viagra, a failed hypertension drug. When the studies ended because it didn’t work, the drug company asked patients to send back the unused drugs. The women all returned the drugs, but the men didn’t. And the rest is history.
There’s a long list of potential drugs that can be repurposed, but few are being tested. The famous poster child of a repurposed drug — hydroxychloroquine — has been the subject of more than 250 clinical trials, but the others weren’t getting much attention.
The beauty of a repurposed drug is that if you can get funding and start enrolling patients, you could potentially find out fairly quickly, as early as a few months, if that drug has an antiviral effect or not. These data would help prioritize drugs to be tested in larger confirmatory studies.
Your focus is on early treatment. What’s the rationale for that?
We are focusing on early treatment because it has been overlooked. The attention has been on vaccines and therapeutics for hospitalized patients. But if you are spending $20 billion on potential vaccines and billions more on diagnostics, we need to give proportional resources toward drugs that might actually work, when given early, in preventing severe disease and death.
Early treatment, if successful, would allow us to avoid the severe complications that we are seeing now. If we can find an early treatment with an existing drug, it would be the fastest, most clinically- and cost-effective way to mitigate the impact of COVID-19 and get us on the road to recovery.
How do you get from a potential repurposed drug for COVID-19 to having a therapeutic agent that will save lives?
Most of the studies we are funding are smaller outpatient studies with virologic endpoints. We are looking for a signal that the drug has antiviral activity. We want to know whether a drug works before we spend the money on questions that take a much larger sample size to answer, for example, a big postexposure prophylaxis study. We’d like to see a meaningful signal in proof-of-concept studies, so we can look at a small group of patients with positive tests and see whether their viral load dropped by more than half if they got the drug compared with those who took the placebo. If the drug had an impact on the viral load and shortened the period of infectivity and was safe, these findings would provide justification to spend a lot of money on a large clinical trial. That would probably encourage the NIH and ACTIV [Accelerating COVID-19 Therapeutic Interventions and Vaccines] collaboration to prioritize the drug for one of their big platform trials. That›s what we are aiming for.
CETF isn’t a drug developer — we are a funder for a good proposal to study a repurposed drug. We want to help move the dial — can we get an early yes or an early no? In drug development, we say, “fail fast and fail early.” It’s a numbers game. Only 10% of early candidates will become approved drugs. The value is in the data, whether they are positive or negative — it doesn’t matter. If the study is a definitive “no,” that is just as helpful as a definitive “yes.” Of course, we all want the definitive “yes,” but there are so many things to look at, the “no’s” will help us redirect resources toward what may really help.
You first announced these funding opportunities in April. How is it going so far?
As soon as the website went up, we got 40 applications. Our scientific advisory board, which has expertise from medicinal chemistry and coronavirology to translational and clinical trial expertise, reviewed the applications and prioritized 11 fundable proposals. We are using milestone-based funding; in other words, funding those who are ready to go.
Which drugs are being tested in the funded studies?
One of the earliest grants we supported was Dr David Boulaware’s randomized controlled trial of hydroxychloroquine (NCT 04308668) in 821 asymptomatic patients within 4 days after a high-risk or moderate-risk exposure. That trial did not show any benefit of hydroxychloroquine as postexposure prophylaxis against COVID-19. This trial was important for another reason. It proved the feasibility of a no-contact trial design in the setting of COVID-19, and participants enrolled themselves through a secure Internet-based survey using the Research Electronic Data Capture (REDCap) system.
Camostat, a transmembrane serine protease (TMPRSS2) inhibitor licensed for use in Japan to treat pancreatitis and esophagitis, combined with the antiandrogen bicalutamide, is being explored for early COVID-19 treatment. TMPRSS2 primes the SARS-CoV-2 spike protein to bind to the ACE2 receptor and gain entry to the cell, and has been shown to have antiviral activity. CETF has provided funding support to ongoing trials of Camostat at Yale University and Aarhus University in Denmark.
Another outpatient trial for fluvoxamine, a drug approved in the United States and routinely prescribed for depression, was also partially funded by a CETF grant to Washington University in St. Louis. Fluvoxamine is a serotonin regulator but also activates the sigma-1 receptor, which reduces the body’s immune response to prevent an overactive immune response or cytokine storm, a major cause of clinical deterioration, serious organ damage, and even death from COVID. This trial was recently completed, and the results have been submitted for publication.
Other promising drugs include niclosamide, doxazosin, favipiravir, leronlimab, interferon beta, interferon lambda, and other monoclonal antibodies. New compounds considered to have potential against COVID include a flu drug (MK-4482/EIDD-2801) and GS-441524, a metabolite of the antiviral drug, remdesivir.
Why not just put all of our resources into vaccine development?
We absolutely need a vaccine to control the outbreak and stop the pandemic. However, it’s a long road to finding an effective vaccine, and in the meantime, we need tools to keep people alive. If we can find an antiviral drug that acts early, we can reduce transmission and contribute to outbreak control. All these tools help us get back to normal while we are waiting for a vaccine. The vaccine is only good if we can give it to every susceptible person in the world — which will take longer than 3 years. And there are no guarantees. Remember, we are still waiting for an HIV vaccine.
You are calling on Americans to help. What do you want them to do?
Everyone must participate in the behavioral changes designed to control the outbreak — physical distancing, face-covering, and paying attention to case counts in local areas to enable them to take appropriate precautions. I know people are bored of that message, but we are going to repeat it until we have a vaccine or herd immunity.
This organism is ripping like wildfire through our unimmunized population. Personal behaviors might slow it down, but finding a drug that can be given to people after they’ve been exposed and test positive will have a meaningful impact on helping us get back to normal.
There’s a great spirit of volunteerism — people are constantly asking how they can help. Through us at CETF, we offer three ways that people can help. They can participate as subjects in clinical trials, many of which are ongoing, including clinical trials, surveillance studies, and follow-up studies. They can donate to our fund and help support the research needed to find an effective early treatment. We have a link on our website, TreatEarly.org. And finally, researchers can apply for funding. We think everybody can help in one of these ways by participating in trials, donating, or applying for funding. It’s an all-hands-on-deck moment for our country.
Danzig is the chief medical advisor of the COVID-19 Early Treatment Fund. She has spent more than 20 years in the pharmaceutical industry developing vaccines, diagnostics, and drugs and is currently advising companies and investors.
This article first appeared on Medscape.com.
Unexpected results in new COVID-19 ‘cytokine storm’ data
The immune system overactivation known as a “cytokine storm” does not play a major role in more severe COVID-19 outcomes, according to unexpected findings in new research. The findings stand in direct contrast to many previous reports.
“We were indeed surprised by the results of our study,” senior study author Peter Pickkers, MD, PhD, said in an interview.
In a unique approach, Dr. Pickkers and colleagues compared cytokine levels in critically ill people with COVID-19 with those in patients with bacterial sepsis, trauma, and after cardiac arrest.
“For the first time, we measured the cytokines in different diseases using the same methods. Our results convincingly show that the circulating cytokine concentrations are not higher, but lower, compared to other diseases,” said Dr. Pickkers, who is affiliated with the department of intensive care medicine at Radboud University Medical Center in Nijmegen, the Netherlands.
The team’s research was published online on Sept. 3 in a letter in JAMA.
Cytokines lower than expected
Normally, cytokines trigger inflammation and promote healing after trauma, infection, or other conditions.
Although a cytokine storm remains ill defined, the authors noted, many researchers have implicated a hyperinflammatory response involving these small proteins in the pathophysiology of COVID-19.
The question remains, however, whether all cytokine storms strike people with different conditions the same way.
Dr. Pickkers, lead author Matthijs Kox, PhD, and colleagues studied 46 people with COVID-19 and acute respiratory distress syndrome (ARDS) who were admitted to the ICU at Radboud University Medical Center. All participants underwent mechanical ventilation and were treated between March 11 and April 27, 2020.
The investigators measured plasma levels of cytokines, including tumor necrosis factor (TNF), interleukin-6, and IL-8. They compared results in this group with those in 51 patients who experienced septic shock and ARDS, 15 patients with septic shock without ARDS, 30 people with out-of-hospital cardiac arrest, and 62 people who experienced multiple traumas. They used historical data for the non–COVID-19 cohorts.
Conditional findings
Compared with patients with septic shock and ARDS, the COVID-19 cohort had lower levels of TNF, IL-6, and IL-8. The differences were statistically significant for TNF (P < .01), as well as for IL-6 and IL-8 concentrations (for both, P < .001).
In addition, the COVID-19 group had significantly lower IL-6 and IL-8 concentrations compared with the patients who had septic shock without ARDS.
The researchers likewise found lower concentrations of IL-8 in patients with COVID-19, compared with the out-of-hospital cardiac arrest patients. IL-8 levels did not differ between the COVID-19 and trauma groups.
Furthermore, the researchers found no differences in IL-6 concentrations between patients with COVID-19 and those who experienced out-of-hospital cardiac arrest or trauma.
However, levels of TNF in people with COVID-19 were higher than in trauma patients.
The small sample sizes and single-center study design are limitations.
“The findings of this preliminary analysis suggest COVID-19 may not be characterized by cytokine storm,” the researchers noted. However, they added, “whether anticytokine therapies will benefit patients with COVID-19 remains to be determined.”
Going forward, Dr. Pickkers and colleagues are investigating the effectiveness of different treatments to lower cytokine levels. They are treating people with COVID-19, for example, with the IL-1 cytokine inhibitor anakinra and steroids.
They also plan to assess the long-term effects of COVID-19 on the immune system. “Following an infection, it is known that the immune system may be suppressed for a longer period of time, and we are determining to what extent this is also present in COVID-19 patients,” Dr. Pickkers said.
Enough to cause a storm?
The study “is quite interesting, and data in this paper are consistent with our data,” Tadamitsu Kishimoto, MD, PhD, of the department of immune regulation at the Immunology Frontier Research Center at Osaka (Japan) University, said in an interview.
His study, published online August 21 in PNAS, also revealed lower serum IL-6 levels among people with COVID-19, compared with patients with bacterial ARDS or sepsis.
Dr. Kishimoto drew a distinction, however: COVID-19 patients can develop severe respiratory failure, suggesting a distinct immune reaction, compared with patients with bacterial sepsis. SARS-CoV-2 directly infects and activates endothelial cells rather than macrophages, as occurs in sepsis.
For this reason, Dr. Kishimoto said, “SARS-CoV-2 infection causes critical illness and severe dysfunction in respiratory organs and induces a cytokine storm,” even in the setting of lower but still elevated serum IL-6 levels.
Dr. Pickkers and Dr. Kishimoto reported no relevant financial relationships.
This story first appeared on Medscape.com.
The immune system overactivation known as a “cytokine storm” does not play a major role in more severe COVID-19 outcomes, according to unexpected findings in new research. The findings stand in direct contrast to many previous reports.
“We were indeed surprised by the results of our study,” senior study author Peter Pickkers, MD, PhD, said in an interview.
In a unique approach, Dr. Pickkers and colleagues compared cytokine levels in critically ill people with COVID-19 with those in patients with bacterial sepsis, trauma, and after cardiac arrest.
“For the first time, we measured the cytokines in different diseases using the same methods. Our results convincingly show that the circulating cytokine concentrations are not higher, but lower, compared to other diseases,” said Dr. Pickkers, who is affiliated with the department of intensive care medicine at Radboud University Medical Center in Nijmegen, the Netherlands.
The team’s research was published online on Sept. 3 in a letter in JAMA.
Cytokines lower than expected
Normally, cytokines trigger inflammation and promote healing after trauma, infection, or other conditions.
Although a cytokine storm remains ill defined, the authors noted, many researchers have implicated a hyperinflammatory response involving these small proteins in the pathophysiology of COVID-19.
The question remains, however, whether all cytokine storms strike people with different conditions the same way.
Dr. Pickkers, lead author Matthijs Kox, PhD, and colleagues studied 46 people with COVID-19 and acute respiratory distress syndrome (ARDS) who were admitted to the ICU at Radboud University Medical Center. All participants underwent mechanical ventilation and were treated between March 11 and April 27, 2020.
The investigators measured plasma levels of cytokines, including tumor necrosis factor (TNF), interleukin-6, and IL-8. They compared results in this group with those in 51 patients who experienced septic shock and ARDS, 15 patients with septic shock without ARDS, 30 people with out-of-hospital cardiac arrest, and 62 people who experienced multiple traumas. They used historical data for the non–COVID-19 cohorts.
Conditional findings
Compared with patients with septic shock and ARDS, the COVID-19 cohort had lower levels of TNF, IL-6, and IL-8. The differences were statistically significant for TNF (P < .01), as well as for IL-6 and IL-8 concentrations (for both, P < .001).
In addition, the COVID-19 group had significantly lower IL-6 and IL-8 concentrations compared with the patients who had septic shock without ARDS.
The researchers likewise found lower concentrations of IL-8 in patients with COVID-19, compared with the out-of-hospital cardiac arrest patients. IL-8 levels did not differ between the COVID-19 and trauma groups.
Furthermore, the researchers found no differences in IL-6 concentrations between patients with COVID-19 and those who experienced out-of-hospital cardiac arrest or trauma.
However, levels of TNF in people with COVID-19 were higher than in trauma patients.
The small sample sizes and single-center study design are limitations.
“The findings of this preliminary analysis suggest COVID-19 may not be characterized by cytokine storm,” the researchers noted. However, they added, “whether anticytokine therapies will benefit patients with COVID-19 remains to be determined.”
Going forward, Dr. Pickkers and colleagues are investigating the effectiveness of different treatments to lower cytokine levels. They are treating people with COVID-19, for example, with the IL-1 cytokine inhibitor anakinra and steroids.
They also plan to assess the long-term effects of COVID-19 on the immune system. “Following an infection, it is known that the immune system may be suppressed for a longer period of time, and we are determining to what extent this is also present in COVID-19 patients,” Dr. Pickkers said.
Enough to cause a storm?
The study “is quite interesting, and data in this paper are consistent with our data,” Tadamitsu Kishimoto, MD, PhD, of the department of immune regulation at the Immunology Frontier Research Center at Osaka (Japan) University, said in an interview.
His study, published online August 21 in PNAS, also revealed lower serum IL-6 levels among people with COVID-19, compared with patients with bacterial ARDS or sepsis.
Dr. Kishimoto drew a distinction, however: COVID-19 patients can develop severe respiratory failure, suggesting a distinct immune reaction, compared with patients with bacterial sepsis. SARS-CoV-2 directly infects and activates endothelial cells rather than macrophages, as occurs in sepsis.
For this reason, Dr. Kishimoto said, “SARS-CoV-2 infection causes critical illness and severe dysfunction in respiratory organs and induces a cytokine storm,” even in the setting of lower but still elevated serum IL-6 levels.
Dr. Pickkers and Dr. Kishimoto reported no relevant financial relationships.
This story first appeared on Medscape.com.
The immune system overactivation known as a “cytokine storm” does not play a major role in more severe COVID-19 outcomes, according to unexpected findings in new research. The findings stand in direct contrast to many previous reports.
“We were indeed surprised by the results of our study,” senior study author Peter Pickkers, MD, PhD, said in an interview.
In a unique approach, Dr. Pickkers and colleagues compared cytokine levels in critically ill people with COVID-19 with those in patients with bacterial sepsis, trauma, and after cardiac arrest.
“For the first time, we measured the cytokines in different diseases using the same methods. Our results convincingly show that the circulating cytokine concentrations are not higher, but lower, compared to other diseases,” said Dr. Pickkers, who is affiliated with the department of intensive care medicine at Radboud University Medical Center in Nijmegen, the Netherlands.
The team’s research was published online on Sept. 3 in a letter in JAMA.
Cytokines lower than expected
Normally, cytokines trigger inflammation and promote healing after trauma, infection, or other conditions.
Although a cytokine storm remains ill defined, the authors noted, many researchers have implicated a hyperinflammatory response involving these small proteins in the pathophysiology of COVID-19.
The question remains, however, whether all cytokine storms strike people with different conditions the same way.
Dr. Pickkers, lead author Matthijs Kox, PhD, and colleagues studied 46 people with COVID-19 and acute respiratory distress syndrome (ARDS) who were admitted to the ICU at Radboud University Medical Center. All participants underwent mechanical ventilation and were treated between March 11 and April 27, 2020.
The investigators measured plasma levels of cytokines, including tumor necrosis factor (TNF), interleukin-6, and IL-8. They compared results in this group with those in 51 patients who experienced septic shock and ARDS, 15 patients with septic shock without ARDS, 30 people with out-of-hospital cardiac arrest, and 62 people who experienced multiple traumas. They used historical data for the non–COVID-19 cohorts.
Conditional findings
Compared with patients with septic shock and ARDS, the COVID-19 cohort had lower levels of TNF, IL-6, and IL-8. The differences were statistically significant for TNF (P < .01), as well as for IL-6 and IL-8 concentrations (for both, P < .001).
In addition, the COVID-19 group had significantly lower IL-6 and IL-8 concentrations compared with the patients who had septic shock without ARDS.
The researchers likewise found lower concentrations of IL-8 in patients with COVID-19, compared with the out-of-hospital cardiac arrest patients. IL-8 levels did not differ between the COVID-19 and trauma groups.
Furthermore, the researchers found no differences in IL-6 concentrations between patients with COVID-19 and those who experienced out-of-hospital cardiac arrest or trauma.
However, levels of TNF in people with COVID-19 were higher than in trauma patients.
The small sample sizes and single-center study design are limitations.
“The findings of this preliminary analysis suggest COVID-19 may not be characterized by cytokine storm,” the researchers noted. However, they added, “whether anticytokine therapies will benefit patients with COVID-19 remains to be determined.”
Going forward, Dr. Pickkers and colleagues are investigating the effectiveness of different treatments to lower cytokine levels. They are treating people with COVID-19, for example, with the IL-1 cytokine inhibitor anakinra and steroids.
They also plan to assess the long-term effects of COVID-19 on the immune system. “Following an infection, it is known that the immune system may be suppressed for a longer period of time, and we are determining to what extent this is also present in COVID-19 patients,” Dr. Pickkers said.
Enough to cause a storm?
The study “is quite interesting, and data in this paper are consistent with our data,” Tadamitsu Kishimoto, MD, PhD, of the department of immune regulation at the Immunology Frontier Research Center at Osaka (Japan) University, said in an interview.
His study, published online August 21 in PNAS, also revealed lower serum IL-6 levels among people with COVID-19, compared with patients with bacterial ARDS or sepsis.
Dr. Kishimoto drew a distinction, however: COVID-19 patients can develop severe respiratory failure, suggesting a distinct immune reaction, compared with patients with bacterial sepsis. SARS-CoV-2 directly infects and activates endothelial cells rather than macrophages, as occurs in sepsis.
For this reason, Dr. Kishimoto said, “SARS-CoV-2 infection causes critical illness and severe dysfunction in respiratory organs and induces a cytokine storm,” even in the setting of lower but still elevated serum IL-6 levels.
Dr. Pickkers and Dr. Kishimoto reported no relevant financial relationships.
This story first appeared on Medscape.com.
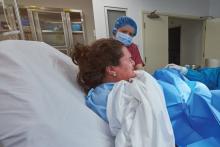
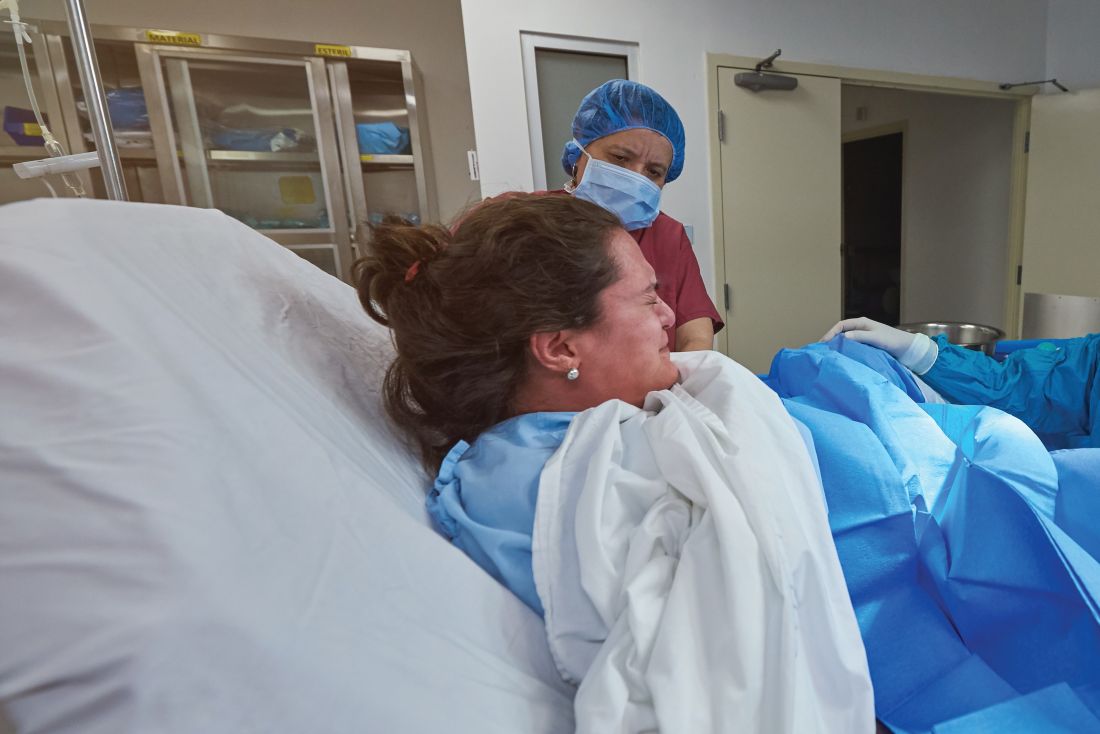
Study: 10% of pregnant women test positive for COVID-19, with most asymptomatic
according to a living systematic review from the PregCOV-19 Living Systematic Review Consortium.
The study, published in BMJ, shows an increased risk of preterm delivery, as well as the need for invasive ventilation in these women, wrote John Allotey, PhD, of the University of Birmingham (England) and colleagues. The findings “will produce a strong evidence base for living guidelines on COVID-19 and pregnancy,” they noted.
The systematic review included 77 studies, one-third each from the United States and China, with the remaining studies from Belgium, Brazil, Denmark, France, Israel, Italy, Japan, Mexico, the Netherlands Portugal, Spain, and the United Kingdom.
The studies included women with COVID-19, of whom 13,118 were either pregnant or in the postpartum or postabortion period and 83,486 were of reproductive age but not pregnant. Some studies also included healthy pregnant women for comparison.
In the pregnant and recently pregnant women, the most common COVID-19 symptoms were fever (40%) and cough (39%), with lymphopenia (35%) and raised C reactive protein levels (49%) being the most common laboratory findings. Pregnant and recently pregnant women with COVID-19 were less likely to have fever (odds ratio, 0.43) and myalgia (OR, 0.48), compared with nonpregnant women of reproductive age with COVID-19, reported the authors.
The overall preterm and spontaneous preterm birth rates in the COVID-19–positive women were 17% and 6% respectively. Dr. Allotey and authors noted that “these preterm births could be medically indicated, as the overall rates of spontaneous preterm births in pregnant women with COVID-19 was broadly similar to those observed in the pre-pandemic period.” There were 18 stillbirths and 6 neonatal deaths in the COVID-19 cohort.
Overall, 73 (0.1%) of pregnant women with confirmed COVID-19 died from any cause, and severe COVID-19 infection was diagnosed in 13%. Maternal risk factors associated with severe infection included older age (OR, 1.78), high body mass index (OR, 2.3), chronic hypertension (OR, 2.0), and preexisting diabetes (OR, 2.51). Compared with nonpregnant women with COVID-19, pregnant or recently pregnant women with the infection were at increased risk of admission to intensive care (OR, 1.62) and needing invasive ventilation (OR, 1.88).
The report included studies published between December 1, 2019, and June 26, 2020, but the living systematic review will involve weekly search updates, with analysis performed every 2-4 weeks and reported through a dedicated website.
The value of a living meta-analysis
Asked to comment on the findings, Torri Metz, MD, a maternal-fetal medicine subspecialist at the University of Utah, Salt Lake City, expressed surprise at the 10% rate of infection in the pregnant or recently pregnant population. “This is higher than currently observed at many hospitals in the United States,” she said in an interview. “This may overestimate the actual risk as many of these studies were published early in the pandemic and did not universally sample women who were pregnant for SARS-CoV-2.”
She noted the value of a living meta-analysis in that it will be updated on a regular basis as new evidence emerges. “During this time of rapidly accumulating publications about COVID-19 infection, clinicians will find it useful to have a resource in which the available data can be combined in one source.”
And there are still some outstanding questions that new studies hopefully will shed light on, she added. “The authors found that many of the risk factors for severe disease, like diabetes, obesity and high blood pressure, in nonpregnant adults are the same in the pregnant population. What remains unknown is if pregnant patients with COVID-19 infection are at higher risk than those who are not pregnant. The authors note that this information is still limited and largely influenced in this published analysis by a CDC [Centers for Disease Control and Prevention] study in which the majority of patients had unknown pregnancy status. We also do not know if COVID-19 infection is associated with any birth defects since the majority of women with COVID-19 infection in the first trimester have not yet delivered.”
Malavika Prabhu, MD, an obstetetrician/gyneologist at Weill Cornell Medicine in New York City added that “this systematic review and meta analysis, which is a compilation of other studies done around the globe, confirms that pregnant women with preexisting medical conditions such as diabetes, hypertension, and obesity, are at increased risk of severe COVID-19 and that pregnant women with COVID-19 are at increased risk of invasive ventilation, compared to nonpregnant women with COVID-19, particularly if they have a preexisting medical condition.”
She said the preterm delivery rate of COVID-positive women is “challenging to interpret given that the total preterm birth rate potentially included many medically indicated preterm deliveries – which is to be expected – and there is no comparison group for spontaneous preterm birth presented”.
Other outstanding questions about COVID-19 pregnancies include whether they are associated with preeclampsia or smaller/growth restricted infants and why the cesarean delivery rate is high, she said. “But some of these questions are tough to answer with this data because it primarily reflects a COVID infection close to the delivery, not one that occurred several months prior to a delivery.”
Deborah Money, MD, professor of obstetrics and gynecology, medicine, and the school of population and public health, University of British Columbia, Vancouver, commented that “this is a group that have been doing ongoing living systematic reviews of the literature scanning for pregnancy outcomes. They post their information in real time on their website, so many of us in this area follow these postings as their methodology is robust and they work hard to only include high-quality literature and avoid duplication of cases in multiple papers. There has been a problem of re-reporting the same severe cases of COVID-19 in the literature.”
This “amplifies the importance of collecting Canadian-specific data to ensure that we understand if these kind of outcomes will also be found in Canada. The data presented in this paper represent outcomes from a broad range of countries with different methods of collecting information on pregnancy and highly variable prenatal care systems. This makes our pan-Canadian study of outcomes of COVID-19 for pregnant women and their infants, CANCOVID-Preg, even more important,” she said.
“Globally, we all must continue to monitor outcomes of COVID-19 in pregnancy to minimize adverse impact on women and their infants,” said Dr. Money, who was not involved in the study.
The study was partially funded by the World Health Organization and supported by Katie’s Team, a dedicated patient and public involvement group in Women’s Health. Dr. Metz is principal investigator for the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) Maternal-Fetal Medicine Units Network COVID-19 study; the study is funded by NICHD and enrollment is ongoing. Dr. Prabhu had no relevant financial disclosures. Dr. Money received funding from the Canadian Institutes for Health Research and the Public Health Agency of Canada and received a small grant from theBC Women’s Foundation for COVID-19 in pregnancy research.
SOURCE: Allotey J et al. BMJ. 2020;370:m3320.
according to a living systematic review from the PregCOV-19 Living Systematic Review Consortium.
The study, published in BMJ, shows an increased risk of preterm delivery, as well as the need for invasive ventilation in these women, wrote John Allotey, PhD, of the University of Birmingham (England) and colleagues. The findings “will produce a strong evidence base for living guidelines on COVID-19 and pregnancy,” they noted.
The systematic review included 77 studies, one-third each from the United States and China, with the remaining studies from Belgium, Brazil, Denmark, France, Israel, Italy, Japan, Mexico, the Netherlands Portugal, Spain, and the United Kingdom.
The studies included women with COVID-19, of whom 13,118 were either pregnant or in the postpartum or postabortion period and 83,486 were of reproductive age but not pregnant. Some studies also included healthy pregnant women for comparison.
In the pregnant and recently pregnant women, the most common COVID-19 symptoms were fever (40%) and cough (39%), with lymphopenia (35%) and raised C reactive protein levels (49%) being the most common laboratory findings. Pregnant and recently pregnant women with COVID-19 were less likely to have fever (odds ratio, 0.43) and myalgia (OR, 0.48), compared with nonpregnant women of reproductive age with COVID-19, reported the authors.
The overall preterm and spontaneous preterm birth rates in the COVID-19–positive women were 17% and 6% respectively. Dr. Allotey and authors noted that “these preterm births could be medically indicated, as the overall rates of spontaneous preterm births in pregnant women with COVID-19 was broadly similar to those observed in the pre-pandemic period.” There were 18 stillbirths and 6 neonatal deaths in the COVID-19 cohort.
Overall, 73 (0.1%) of pregnant women with confirmed COVID-19 died from any cause, and severe COVID-19 infection was diagnosed in 13%. Maternal risk factors associated with severe infection included older age (OR, 1.78), high body mass index (OR, 2.3), chronic hypertension (OR, 2.0), and preexisting diabetes (OR, 2.51). Compared with nonpregnant women with COVID-19, pregnant or recently pregnant women with the infection were at increased risk of admission to intensive care (OR, 1.62) and needing invasive ventilation (OR, 1.88).
The report included studies published between December 1, 2019, and June 26, 2020, but the living systematic review will involve weekly search updates, with analysis performed every 2-4 weeks and reported through a dedicated website.
The value of a living meta-analysis
Asked to comment on the findings, Torri Metz, MD, a maternal-fetal medicine subspecialist at the University of Utah, Salt Lake City, expressed surprise at the 10% rate of infection in the pregnant or recently pregnant population. “This is higher than currently observed at many hospitals in the United States,” she said in an interview. “This may overestimate the actual risk as many of these studies were published early in the pandemic and did not universally sample women who were pregnant for SARS-CoV-2.”
She noted the value of a living meta-analysis in that it will be updated on a regular basis as new evidence emerges. “During this time of rapidly accumulating publications about COVID-19 infection, clinicians will find it useful to have a resource in which the available data can be combined in one source.”
And there are still some outstanding questions that new studies hopefully will shed light on, she added. “The authors found that many of the risk factors for severe disease, like diabetes, obesity and high blood pressure, in nonpregnant adults are the same in the pregnant population. What remains unknown is if pregnant patients with COVID-19 infection are at higher risk than those who are not pregnant. The authors note that this information is still limited and largely influenced in this published analysis by a CDC [Centers for Disease Control and Prevention] study in which the majority of patients had unknown pregnancy status. We also do not know if COVID-19 infection is associated with any birth defects since the majority of women with COVID-19 infection in the first trimester have not yet delivered.”
Malavika Prabhu, MD, an obstetetrician/gyneologist at Weill Cornell Medicine in New York City added that “this systematic review and meta analysis, which is a compilation of other studies done around the globe, confirms that pregnant women with preexisting medical conditions such as diabetes, hypertension, and obesity, are at increased risk of severe COVID-19 and that pregnant women with COVID-19 are at increased risk of invasive ventilation, compared to nonpregnant women with COVID-19, particularly if they have a preexisting medical condition.”
She said the preterm delivery rate of COVID-positive women is “challenging to interpret given that the total preterm birth rate potentially included many medically indicated preterm deliveries – which is to be expected – and there is no comparison group for spontaneous preterm birth presented”.
Other outstanding questions about COVID-19 pregnancies include whether they are associated with preeclampsia or smaller/growth restricted infants and why the cesarean delivery rate is high, she said. “But some of these questions are tough to answer with this data because it primarily reflects a COVID infection close to the delivery, not one that occurred several months prior to a delivery.”
Deborah Money, MD, professor of obstetrics and gynecology, medicine, and the school of population and public health, University of British Columbia, Vancouver, commented that “this is a group that have been doing ongoing living systematic reviews of the literature scanning for pregnancy outcomes. They post their information in real time on their website, so many of us in this area follow these postings as their methodology is robust and they work hard to only include high-quality literature and avoid duplication of cases in multiple papers. There has been a problem of re-reporting the same severe cases of COVID-19 in the literature.”
This “amplifies the importance of collecting Canadian-specific data to ensure that we understand if these kind of outcomes will also be found in Canada. The data presented in this paper represent outcomes from a broad range of countries with different methods of collecting information on pregnancy and highly variable prenatal care systems. This makes our pan-Canadian study of outcomes of COVID-19 for pregnant women and their infants, CANCOVID-Preg, even more important,” she said.
“Globally, we all must continue to monitor outcomes of COVID-19 in pregnancy to minimize adverse impact on women and their infants,” said Dr. Money, who was not involved in the study.
The study was partially funded by the World Health Organization and supported by Katie’s Team, a dedicated patient and public involvement group in Women’s Health. Dr. Metz is principal investigator for the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) Maternal-Fetal Medicine Units Network COVID-19 study; the study is funded by NICHD and enrollment is ongoing. Dr. Prabhu had no relevant financial disclosures. Dr. Money received funding from the Canadian Institutes for Health Research and the Public Health Agency of Canada and received a small grant from theBC Women’s Foundation for COVID-19 in pregnancy research.
SOURCE: Allotey J et al. BMJ. 2020;370:m3320.
according to a living systematic review from the PregCOV-19 Living Systematic Review Consortium.
The study, published in BMJ, shows an increased risk of preterm delivery, as well as the need for invasive ventilation in these women, wrote John Allotey, PhD, of the University of Birmingham (England) and colleagues. The findings “will produce a strong evidence base for living guidelines on COVID-19 and pregnancy,” they noted.
The systematic review included 77 studies, one-third each from the United States and China, with the remaining studies from Belgium, Brazil, Denmark, France, Israel, Italy, Japan, Mexico, the Netherlands Portugal, Spain, and the United Kingdom.
The studies included women with COVID-19, of whom 13,118 were either pregnant or in the postpartum or postabortion period and 83,486 were of reproductive age but not pregnant. Some studies also included healthy pregnant women for comparison.
In the pregnant and recently pregnant women, the most common COVID-19 symptoms were fever (40%) and cough (39%), with lymphopenia (35%) and raised C reactive protein levels (49%) being the most common laboratory findings. Pregnant and recently pregnant women with COVID-19 were less likely to have fever (odds ratio, 0.43) and myalgia (OR, 0.48), compared with nonpregnant women of reproductive age with COVID-19, reported the authors.
The overall preterm and spontaneous preterm birth rates in the COVID-19–positive women were 17% and 6% respectively. Dr. Allotey and authors noted that “these preterm births could be medically indicated, as the overall rates of spontaneous preterm births in pregnant women with COVID-19 was broadly similar to those observed in the pre-pandemic period.” There were 18 stillbirths and 6 neonatal deaths in the COVID-19 cohort.
Overall, 73 (0.1%) of pregnant women with confirmed COVID-19 died from any cause, and severe COVID-19 infection was diagnosed in 13%. Maternal risk factors associated with severe infection included older age (OR, 1.78), high body mass index (OR, 2.3), chronic hypertension (OR, 2.0), and preexisting diabetes (OR, 2.51). Compared with nonpregnant women with COVID-19, pregnant or recently pregnant women with the infection were at increased risk of admission to intensive care (OR, 1.62) and needing invasive ventilation (OR, 1.88).
The report included studies published between December 1, 2019, and June 26, 2020, but the living systematic review will involve weekly search updates, with analysis performed every 2-4 weeks and reported through a dedicated website.
The value of a living meta-analysis
Asked to comment on the findings, Torri Metz, MD, a maternal-fetal medicine subspecialist at the University of Utah, Salt Lake City, expressed surprise at the 10% rate of infection in the pregnant or recently pregnant population. “This is higher than currently observed at many hospitals in the United States,” she said in an interview. “This may overestimate the actual risk as many of these studies were published early in the pandemic and did not universally sample women who were pregnant for SARS-CoV-2.”
She noted the value of a living meta-analysis in that it will be updated on a regular basis as new evidence emerges. “During this time of rapidly accumulating publications about COVID-19 infection, clinicians will find it useful to have a resource in which the available data can be combined in one source.”
And there are still some outstanding questions that new studies hopefully will shed light on, she added. “The authors found that many of the risk factors for severe disease, like diabetes, obesity and high blood pressure, in nonpregnant adults are the same in the pregnant population. What remains unknown is if pregnant patients with COVID-19 infection are at higher risk than those who are not pregnant. The authors note that this information is still limited and largely influenced in this published analysis by a CDC [Centers for Disease Control and Prevention] study in which the majority of patients had unknown pregnancy status. We also do not know if COVID-19 infection is associated with any birth defects since the majority of women with COVID-19 infection in the first trimester have not yet delivered.”
Malavika Prabhu, MD, an obstetetrician/gyneologist at Weill Cornell Medicine in New York City added that “this systematic review and meta analysis, which is a compilation of other studies done around the globe, confirms that pregnant women with preexisting medical conditions such as diabetes, hypertension, and obesity, are at increased risk of severe COVID-19 and that pregnant women with COVID-19 are at increased risk of invasive ventilation, compared to nonpregnant women with COVID-19, particularly if they have a preexisting medical condition.”
She said the preterm delivery rate of COVID-positive women is “challenging to interpret given that the total preterm birth rate potentially included many medically indicated preterm deliveries – which is to be expected – and there is no comparison group for spontaneous preterm birth presented”.
Other outstanding questions about COVID-19 pregnancies include whether they are associated with preeclampsia or smaller/growth restricted infants and why the cesarean delivery rate is high, she said. “But some of these questions are tough to answer with this data because it primarily reflects a COVID infection close to the delivery, not one that occurred several months prior to a delivery.”
Deborah Money, MD, professor of obstetrics and gynecology, medicine, and the school of population and public health, University of British Columbia, Vancouver, commented that “this is a group that have been doing ongoing living systematic reviews of the literature scanning for pregnancy outcomes. They post their information in real time on their website, so many of us in this area follow these postings as their methodology is robust and they work hard to only include high-quality literature and avoid duplication of cases in multiple papers. There has been a problem of re-reporting the same severe cases of COVID-19 in the literature.”
This “amplifies the importance of collecting Canadian-specific data to ensure that we understand if these kind of outcomes will also be found in Canada. The data presented in this paper represent outcomes from a broad range of countries with different methods of collecting information on pregnancy and highly variable prenatal care systems. This makes our pan-Canadian study of outcomes of COVID-19 for pregnant women and their infants, CANCOVID-Preg, even more important,” she said.
“Globally, we all must continue to monitor outcomes of COVID-19 in pregnancy to minimize adverse impact on women and their infants,” said Dr. Money, who was not involved in the study.
The study was partially funded by the World Health Organization and supported by Katie’s Team, a dedicated patient and public involvement group in Women’s Health. Dr. Metz is principal investigator for the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) Maternal-Fetal Medicine Units Network COVID-19 study; the study is funded by NICHD and enrollment is ongoing. Dr. Prabhu had no relevant financial disclosures. Dr. Money received funding from the Canadian Institutes for Health Research and the Public Health Agency of Canada and received a small grant from theBC Women’s Foundation for COVID-19 in pregnancy research.
SOURCE: Allotey J et al. BMJ. 2020;370:m3320.
FROM BMJ
Drug combo slows functional decline in ALS
, according to results of the phase 2/3 CENTAUR study.
Patients with a fast-progressing form of ALS who were treated with AMX0035 “retained higher levels of physical function over 6 months compared with those who received placebo,” reported principal investigator Sabrina Paganoni, MD, PhD, of the Sean M. Healey and AMG Center for ALS at Massachusetts General Hospital, Boston.
“This is very hopeful news for people affected by ALS, especially because we were able to see a treatment effect in a relatively short period of time,” Dr. Paganoni said.
The study was published online Sept. 3 in the New England Journal of Medicine.
In this study, AMX0035 demonstrated a “clinically meaningful benefit and a favorable safety profile for people living with ALS,” Josh Cohen, co-CEO, chairman, and cofounder at Amylyx, said in a news release. The company is “working collaboratively and expeditiously with agencies worldwide to bring this potential new treatment option forward.”
“The data ... makes a clear and compelling case that AMX0035 should be made available to people with ALS as soon as possible,” Calaneet Balas, president and CEO of The ALS Association, said in the release.
The CENTAUR trial
Sodium phenylbutyrate and taurursodiol have been found to reduce neuronal death in experimental models. AMX0035 combines 3 g sodium phenylbutyrate and 1 g of taurursodiol.
The CENTAUR trial tested AMX0035 against placebo in 137 ALS patients with symptom onset within the prior 18 months, with 89 patients in the AMX0035 group and 48 in the placebo group. AMX0035 or matching placebo were administered once daily for 3 weeks and then twice daily for a planned duration of 24 weeks.
In a modified intention-to-treat analysis, the mean rate of change in the Amyotrophic Lateral Sclerosis Functional Rating Scale–Revised (ALSFRS-R) score was −1.24 points per month with AMX0035 and −1.66 points per month with placebo (difference, 0.42 points per month; 95% CI, 0.03 - 0.81; P = .03). After 24 weeks, patients treated with AMX0035 scored on average 2.32 points higher on the ALSFRS-R than their peers on placebo group (P = .03).
“The score, consisting of four subdomains, showed a change that was most prominent for the fine-motor subscale and less apparent for the other subscales,” the investigators said.
Treatment with AMX0035 led to slowing of disease progression in a population in which many participants were receiving riluzole (Tiglutik), edaravone (Radicava) or both, they pointed out.
The secondary outcomes were rate of decline in isometric muscle strength and breathing function; change in plasma phosphorylated axonal neurofilament H subunit (pNF-H) levels; and the time to composite events of death, tracheostomy, permanent ventilation, and hospitalization. These outcomes did not differ significantly between the two groups.
Open-label extension ongoing
AMX0035 was generally well tolerated. Nearly all patients in both groups had one or more adverse events. Events occurring at 2% or greater frequency in the AMX0035 group were primarily gastrointestinal (diarrhea, nausea, salivary hypersecretion, and abdominal discomfort). Serious adverse events were more common in the placebo group (19% vs. 12%). The incidence of respiratory serious adverse events was 8% in the placebo group and 3% in the AMX0035 group.
More patients on active treatment than placebo (19% vs. 8%) stopped the trial regimen early owing to adverse events. The most common adverse events leading to discontinuation of the trial regimen were diarrhea and respiratory failure.
The trial was “too short for us to detect an effect on survival,” Dr. Paganoni said in an interview. Most of the participants who completed the trial elected to enroll in an open-label extension study and receive AMX0035 long-term. “This is important because it will teach us about the impact of AMX0035 on survival,” said Dr. Paganoni.
Interim data from the ongoing open-label extension study are being submitted to a peer-reviewed journal shortly and will be published in the coming months.
A cause for hope
“There has been understandable frustration with the slow pace of development of therapy for ALS,” Michael Benatar, MD, PhD, University of Miami, and Michael McDermott, PhD, University of Rochester (N.Y.), said in an accompanying editorial.
“Despite dozens of trials, few pharmacologic agents have emerged that affect functional decline or survival – and all only modestly so. Although the effects of sodium phenylbutyrate–taurursodiol are similarly modest, the incremental gains that they provide in the battle against ALS are a cause for hope,” they wrote.
They caution, however, that this study was enriched for patients with more rapidly progressive disease, which “raises questions about generalizability to the broader population of patients with ALS.
“Although the patients who were enrolled in the trial may not be biologically different from the broader population of patients with ALS, the magnitude of therapeutic effect may be smaller in the latter,” Dr. Benatar and Dr. McDermott noted.
They said that in light of “residual questions about efficacy and the ability of patients to continue taking the drug,” they agree with the authors’ conclusion that “longer and larger trials are needed to evaluate the efficacy and safety of sodium phenylbutyrate–taurursodiol in persons with ALS.”
Given these “tantalizing preliminary data,” Dr. Benatar and Dr. McDermott said they look forward to “a confirmatory phase 3 trial.”
The study was supported by Amylyx Pharmaceuticals, the ALS Finding a Cure Foundation, and the ALS Association. Dr. Paganoni has received grants from Revalesio, Ra Pharma, Biohaven, Clene, and Prilenia. A complete list of disclosures for authors and editorialists is available with the original article.
A version of this article originally appeared on Medscape.com.
, according to results of the phase 2/3 CENTAUR study.
Patients with a fast-progressing form of ALS who were treated with AMX0035 “retained higher levels of physical function over 6 months compared with those who received placebo,” reported principal investigator Sabrina Paganoni, MD, PhD, of the Sean M. Healey and AMG Center for ALS at Massachusetts General Hospital, Boston.
“This is very hopeful news for people affected by ALS, especially because we were able to see a treatment effect in a relatively short period of time,” Dr. Paganoni said.
The study was published online Sept. 3 in the New England Journal of Medicine.
In this study, AMX0035 demonstrated a “clinically meaningful benefit and a favorable safety profile for people living with ALS,” Josh Cohen, co-CEO, chairman, and cofounder at Amylyx, said in a news release. The company is “working collaboratively and expeditiously with agencies worldwide to bring this potential new treatment option forward.”
“The data ... makes a clear and compelling case that AMX0035 should be made available to people with ALS as soon as possible,” Calaneet Balas, president and CEO of The ALS Association, said in the release.
The CENTAUR trial
Sodium phenylbutyrate and taurursodiol have been found to reduce neuronal death in experimental models. AMX0035 combines 3 g sodium phenylbutyrate and 1 g of taurursodiol.
The CENTAUR trial tested AMX0035 against placebo in 137 ALS patients with symptom onset within the prior 18 months, with 89 patients in the AMX0035 group and 48 in the placebo group. AMX0035 or matching placebo were administered once daily for 3 weeks and then twice daily for a planned duration of 24 weeks.
In a modified intention-to-treat analysis, the mean rate of change in the Amyotrophic Lateral Sclerosis Functional Rating Scale–Revised (ALSFRS-R) score was −1.24 points per month with AMX0035 and −1.66 points per month with placebo (difference, 0.42 points per month; 95% CI, 0.03 - 0.81; P = .03). After 24 weeks, patients treated with AMX0035 scored on average 2.32 points higher on the ALSFRS-R than their peers on placebo group (P = .03).
“The score, consisting of four subdomains, showed a change that was most prominent for the fine-motor subscale and less apparent for the other subscales,” the investigators said.
Treatment with AMX0035 led to slowing of disease progression in a population in which many participants were receiving riluzole (Tiglutik), edaravone (Radicava) or both, they pointed out.
The secondary outcomes were rate of decline in isometric muscle strength and breathing function; change in plasma phosphorylated axonal neurofilament H subunit (pNF-H) levels; and the time to composite events of death, tracheostomy, permanent ventilation, and hospitalization. These outcomes did not differ significantly between the two groups.
Open-label extension ongoing
AMX0035 was generally well tolerated. Nearly all patients in both groups had one or more adverse events. Events occurring at 2% or greater frequency in the AMX0035 group were primarily gastrointestinal (diarrhea, nausea, salivary hypersecretion, and abdominal discomfort). Serious adverse events were more common in the placebo group (19% vs. 12%). The incidence of respiratory serious adverse events was 8% in the placebo group and 3% in the AMX0035 group.
More patients on active treatment than placebo (19% vs. 8%) stopped the trial regimen early owing to adverse events. The most common adverse events leading to discontinuation of the trial regimen were diarrhea and respiratory failure.
The trial was “too short for us to detect an effect on survival,” Dr. Paganoni said in an interview. Most of the participants who completed the trial elected to enroll in an open-label extension study and receive AMX0035 long-term. “This is important because it will teach us about the impact of AMX0035 on survival,” said Dr. Paganoni.
Interim data from the ongoing open-label extension study are being submitted to a peer-reviewed journal shortly and will be published in the coming months.
A cause for hope
“There has been understandable frustration with the slow pace of development of therapy for ALS,” Michael Benatar, MD, PhD, University of Miami, and Michael McDermott, PhD, University of Rochester (N.Y.), said in an accompanying editorial.
“Despite dozens of trials, few pharmacologic agents have emerged that affect functional decline or survival – and all only modestly so. Although the effects of sodium phenylbutyrate–taurursodiol are similarly modest, the incremental gains that they provide in the battle against ALS are a cause for hope,” they wrote.
They caution, however, that this study was enriched for patients with more rapidly progressive disease, which “raises questions about generalizability to the broader population of patients with ALS.
“Although the patients who were enrolled in the trial may not be biologically different from the broader population of patients with ALS, the magnitude of therapeutic effect may be smaller in the latter,” Dr. Benatar and Dr. McDermott noted.
They said that in light of “residual questions about efficacy and the ability of patients to continue taking the drug,” they agree with the authors’ conclusion that “longer and larger trials are needed to evaluate the efficacy and safety of sodium phenylbutyrate–taurursodiol in persons with ALS.”
Given these “tantalizing preliminary data,” Dr. Benatar and Dr. McDermott said they look forward to “a confirmatory phase 3 trial.”
The study was supported by Amylyx Pharmaceuticals, the ALS Finding a Cure Foundation, and the ALS Association. Dr. Paganoni has received grants from Revalesio, Ra Pharma, Biohaven, Clene, and Prilenia. A complete list of disclosures for authors and editorialists is available with the original article.
A version of this article originally appeared on Medscape.com.
, according to results of the phase 2/3 CENTAUR study.
Patients with a fast-progressing form of ALS who were treated with AMX0035 “retained higher levels of physical function over 6 months compared with those who received placebo,” reported principal investigator Sabrina Paganoni, MD, PhD, of the Sean M. Healey and AMG Center for ALS at Massachusetts General Hospital, Boston.
“This is very hopeful news for people affected by ALS, especially because we were able to see a treatment effect in a relatively short period of time,” Dr. Paganoni said.
The study was published online Sept. 3 in the New England Journal of Medicine.
In this study, AMX0035 demonstrated a “clinically meaningful benefit and a favorable safety profile for people living with ALS,” Josh Cohen, co-CEO, chairman, and cofounder at Amylyx, said in a news release. The company is “working collaboratively and expeditiously with agencies worldwide to bring this potential new treatment option forward.”
“The data ... makes a clear and compelling case that AMX0035 should be made available to people with ALS as soon as possible,” Calaneet Balas, president and CEO of The ALS Association, said in the release.
The CENTAUR trial
Sodium phenylbutyrate and taurursodiol have been found to reduce neuronal death in experimental models. AMX0035 combines 3 g sodium phenylbutyrate and 1 g of taurursodiol.
The CENTAUR trial tested AMX0035 against placebo in 137 ALS patients with symptom onset within the prior 18 months, with 89 patients in the AMX0035 group and 48 in the placebo group. AMX0035 or matching placebo were administered once daily for 3 weeks and then twice daily for a planned duration of 24 weeks.
In a modified intention-to-treat analysis, the mean rate of change in the Amyotrophic Lateral Sclerosis Functional Rating Scale–Revised (ALSFRS-R) score was −1.24 points per month with AMX0035 and −1.66 points per month with placebo (difference, 0.42 points per month; 95% CI, 0.03 - 0.81; P = .03). After 24 weeks, patients treated with AMX0035 scored on average 2.32 points higher on the ALSFRS-R than their peers on placebo group (P = .03).
“The score, consisting of four subdomains, showed a change that was most prominent for the fine-motor subscale and less apparent for the other subscales,” the investigators said.
Treatment with AMX0035 led to slowing of disease progression in a population in which many participants were receiving riluzole (Tiglutik), edaravone (Radicava) or both, they pointed out.
The secondary outcomes were rate of decline in isometric muscle strength and breathing function; change in plasma phosphorylated axonal neurofilament H subunit (pNF-H) levels; and the time to composite events of death, tracheostomy, permanent ventilation, and hospitalization. These outcomes did not differ significantly between the two groups.
Open-label extension ongoing
AMX0035 was generally well tolerated. Nearly all patients in both groups had one or more adverse events. Events occurring at 2% or greater frequency in the AMX0035 group were primarily gastrointestinal (diarrhea, nausea, salivary hypersecretion, and abdominal discomfort). Serious adverse events were more common in the placebo group (19% vs. 12%). The incidence of respiratory serious adverse events was 8% in the placebo group and 3% in the AMX0035 group.
More patients on active treatment than placebo (19% vs. 8%) stopped the trial regimen early owing to adverse events. The most common adverse events leading to discontinuation of the trial regimen were diarrhea and respiratory failure.
The trial was “too short for us to detect an effect on survival,” Dr. Paganoni said in an interview. Most of the participants who completed the trial elected to enroll in an open-label extension study and receive AMX0035 long-term. “This is important because it will teach us about the impact of AMX0035 on survival,” said Dr. Paganoni.
Interim data from the ongoing open-label extension study are being submitted to a peer-reviewed journal shortly and will be published in the coming months.
A cause for hope
“There has been understandable frustration with the slow pace of development of therapy for ALS,” Michael Benatar, MD, PhD, University of Miami, and Michael McDermott, PhD, University of Rochester (N.Y.), said in an accompanying editorial.
“Despite dozens of trials, few pharmacologic agents have emerged that affect functional decline or survival – and all only modestly so. Although the effects of sodium phenylbutyrate–taurursodiol are similarly modest, the incremental gains that they provide in the battle against ALS are a cause for hope,” they wrote.
They caution, however, that this study was enriched for patients with more rapidly progressive disease, which “raises questions about generalizability to the broader population of patients with ALS.
“Although the patients who were enrolled in the trial may not be biologically different from the broader population of patients with ALS, the magnitude of therapeutic effect may be smaller in the latter,” Dr. Benatar and Dr. McDermott noted.
They said that in light of “residual questions about efficacy and the ability of patients to continue taking the drug,” they agree with the authors’ conclusion that “longer and larger trials are needed to evaluate the efficacy and safety of sodium phenylbutyrate–taurursodiol in persons with ALS.”
Given these “tantalizing preliminary data,” Dr. Benatar and Dr. McDermott said they look forward to “a confirmatory phase 3 trial.”
The study was supported by Amylyx Pharmaceuticals, the ALS Finding a Cure Foundation, and the ALS Association. Dr. Paganoni has received grants from Revalesio, Ra Pharma, Biohaven, Clene, and Prilenia. A complete list of disclosures for authors and editorialists is available with the original article.
A version of this article originally appeared on Medscape.com.
From New England Journal of Medicine
‘No mobile phone’ phobia tied to sleep problems in college students
In a study of more than 300 college students, nearly 9 in 10 (89%) were classified as having moderate to severe nomophobia. Greater levels of nomophobia were significantly linked to daytime sleepiness and more behaviors associated with poor sleep hygiene.
“My undergraduate research team came up with the idea for this study,” said study investigator Jennifer Peszka, PhD, professor of psychology at Hendrix College, Conway, Ark. She explained that her students had been looking at the impact of technology use in the 2 hours before bed, and hypothesized that ‘cell phone addiction’ might play a role in sleep problems.
Incidentally, “that group of students were all pretty high on nomophobia themselves so they were really interested in the outcome,” Dr. Peszka said.
The study findings were presented at the virtual annual meeting of the Associated Professional Sleep Societies.
A likely suspect
The study involved 327 undergraduates (mean age, 19.7 years) recruited from introductory psychology courses and campus newsletters. They completed several questionnaires, including the Nomophobia Questionnaire, the Epworth Sleepiness Scale, and the Sleep Hygiene Index.
Nomophobia was prevalent, with mild, moderate, and severe nomophobia reported by 10%, 83%, and 7% of students, respectively. Only one student reported no nomophobia at all. Dr. Peszka said the fact that 89% of students had moderate or severe nomophobia is “concerning,” given a 2012 study suggesting that 77% of 18- to 24-year-olds had nomophobia. This phobia “very well may be on a rapid rise,” she lamented.
Greater severity of nomophobia was significantly correlated with greater sleepiness measured by both the Epworth Sleepiness Scale (P < .05) and the Associated Features of Poor Sleep Hygiene daytime sleepiness item (P < .05). More severe nomophobia was also related to decreased motivation (a commonly reported symptom of insufficient sleep) and with more maladaptive sleep hygiene behaviors (including using technology during sleep time, long daytime naps, inconsistent wake and bed times, using bed for nonsleep purposes, uncomfortable bed, and bedtime cognitive rumination).
Prior research has shown that smartphones may lead to compulsive “checking” habits, compulsive usage, increased distress, and potentially addictive behaviors. Active phone use at bedtime has also been implicated in disrupted sleep. Nomophobia is likely to be an important consideration when treating sleep disorders and/or making any sleep hygiene recommendations, Dr. Peszka said.
Proliferation of ‘night owls’
Reached for comment, Rajkumar (Raj) Dasgupta, MD, University of Southern California, Los Angeles, said this is a “very timely study with COVID-19. Right now, more than ever, technology is a double-edged sword. I’m a father of three kids and, for now, technology is the only way some kids are going to be socializing and learning.”
Yet a foundation of good sleep hygiene is keeping a nightly sleep routine, said Dr. Dasgupta, who was not involved in the study. “Right now, it seems like all my sleep patients are becoming night owls and sleep time is becoming more and more delayed because there is so much news to keep up with. Also, you may be stressed at night and you may not have the motivation to wake up early in the morning.”
He said it is important to counsel patients to “put technology away at night. That goes for kids and adults.”
Support for the study was provided by Hendrix College Charles Brewer Fund for Psychology. Dr. Peszka and Dr. Dasgupta disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In a study of more than 300 college students, nearly 9 in 10 (89%) were classified as having moderate to severe nomophobia. Greater levels of nomophobia were significantly linked to daytime sleepiness and more behaviors associated with poor sleep hygiene.
“My undergraduate research team came up with the idea for this study,” said study investigator Jennifer Peszka, PhD, professor of psychology at Hendrix College, Conway, Ark. She explained that her students had been looking at the impact of technology use in the 2 hours before bed, and hypothesized that ‘cell phone addiction’ might play a role in sleep problems.
Incidentally, “that group of students were all pretty high on nomophobia themselves so they were really interested in the outcome,” Dr. Peszka said.
The study findings were presented at the virtual annual meeting of the Associated Professional Sleep Societies.
A likely suspect
The study involved 327 undergraduates (mean age, 19.7 years) recruited from introductory psychology courses and campus newsletters. They completed several questionnaires, including the Nomophobia Questionnaire, the Epworth Sleepiness Scale, and the Sleep Hygiene Index.
Nomophobia was prevalent, with mild, moderate, and severe nomophobia reported by 10%, 83%, and 7% of students, respectively. Only one student reported no nomophobia at all. Dr. Peszka said the fact that 89% of students had moderate or severe nomophobia is “concerning,” given a 2012 study suggesting that 77% of 18- to 24-year-olds had nomophobia. This phobia “very well may be on a rapid rise,” she lamented.
Greater severity of nomophobia was significantly correlated with greater sleepiness measured by both the Epworth Sleepiness Scale (P < .05) and the Associated Features of Poor Sleep Hygiene daytime sleepiness item (P < .05). More severe nomophobia was also related to decreased motivation (a commonly reported symptom of insufficient sleep) and with more maladaptive sleep hygiene behaviors (including using technology during sleep time, long daytime naps, inconsistent wake and bed times, using bed for nonsleep purposes, uncomfortable bed, and bedtime cognitive rumination).
Prior research has shown that smartphones may lead to compulsive “checking” habits, compulsive usage, increased distress, and potentially addictive behaviors. Active phone use at bedtime has also been implicated in disrupted sleep. Nomophobia is likely to be an important consideration when treating sleep disorders and/or making any sleep hygiene recommendations, Dr. Peszka said.
Proliferation of ‘night owls’
Reached for comment, Rajkumar (Raj) Dasgupta, MD, University of Southern California, Los Angeles, said this is a “very timely study with COVID-19. Right now, more than ever, technology is a double-edged sword. I’m a father of three kids and, for now, technology is the only way some kids are going to be socializing and learning.”
Yet a foundation of good sleep hygiene is keeping a nightly sleep routine, said Dr. Dasgupta, who was not involved in the study. “Right now, it seems like all my sleep patients are becoming night owls and sleep time is becoming more and more delayed because there is so much news to keep up with. Also, you may be stressed at night and you may not have the motivation to wake up early in the morning.”
He said it is important to counsel patients to “put technology away at night. That goes for kids and adults.”
Support for the study was provided by Hendrix College Charles Brewer Fund for Psychology. Dr. Peszka and Dr. Dasgupta disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In a study of more than 300 college students, nearly 9 in 10 (89%) were classified as having moderate to severe nomophobia. Greater levels of nomophobia were significantly linked to daytime sleepiness and more behaviors associated with poor sleep hygiene.
“My undergraduate research team came up with the idea for this study,” said study investigator Jennifer Peszka, PhD, professor of psychology at Hendrix College, Conway, Ark. She explained that her students had been looking at the impact of technology use in the 2 hours before bed, and hypothesized that ‘cell phone addiction’ might play a role in sleep problems.
Incidentally, “that group of students were all pretty high on nomophobia themselves so they were really interested in the outcome,” Dr. Peszka said.
The study findings were presented at the virtual annual meeting of the Associated Professional Sleep Societies.
A likely suspect
The study involved 327 undergraduates (mean age, 19.7 years) recruited from introductory psychology courses and campus newsletters. They completed several questionnaires, including the Nomophobia Questionnaire, the Epworth Sleepiness Scale, and the Sleep Hygiene Index.
Nomophobia was prevalent, with mild, moderate, and severe nomophobia reported by 10%, 83%, and 7% of students, respectively. Only one student reported no nomophobia at all. Dr. Peszka said the fact that 89% of students had moderate or severe nomophobia is “concerning,” given a 2012 study suggesting that 77% of 18- to 24-year-olds had nomophobia. This phobia “very well may be on a rapid rise,” she lamented.
Greater severity of nomophobia was significantly correlated with greater sleepiness measured by both the Epworth Sleepiness Scale (P < .05) and the Associated Features of Poor Sleep Hygiene daytime sleepiness item (P < .05). More severe nomophobia was also related to decreased motivation (a commonly reported symptom of insufficient sleep) and with more maladaptive sleep hygiene behaviors (including using technology during sleep time, long daytime naps, inconsistent wake and bed times, using bed for nonsleep purposes, uncomfortable bed, and bedtime cognitive rumination).
Prior research has shown that smartphones may lead to compulsive “checking” habits, compulsive usage, increased distress, and potentially addictive behaviors. Active phone use at bedtime has also been implicated in disrupted sleep. Nomophobia is likely to be an important consideration when treating sleep disorders and/or making any sleep hygiene recommendations, Dr. Peszka said.
Proliferation of ‘night owls’
Reached for comment, Rajkumar (Raj) Dasgupta, MD, University of Southern California, Los Angeles, said this is a “very timely study with COVID-19. Right now, more than ever, technology is a double-edged sword. I’m a father of three kids and, for now, technology is the only way some kids are going to be socializing and learning.”
Yet a foundation of good sleep hygiene is keeping a nightly sleep routine, said Dr. Dasgupta, who was not involved in the study. “Right now, it seems like all my sleep patients are becoming night owls and sleep time is becoming more and more delayed because there is so much news to keep up with. Also, you may be stressed at night and you may not have the motivation to wake up early in the morning.”
He said it is important to counsel patients to “put technology away at night. That goes for kids and adults.”
Support for the study was provided by Hendrix College Charles Brewer Fund for Psychology. Dr. Peszka and Dr. Dasgupta disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM SLEEP 2020
Experts advocate for the elimination of daylight savings time
In the interest of public health and safety, – a recommendation that has garnered strong support from multiple medical and other high-profile organizations.
“Permanent, year-round standard time is the best choice to most closely match our circadian sleep-wake cycle,” M. Adeel Rishi, MD, lead author of the AASM position statement, said in a news release. “Daylight saving time results in more darkness in the morning and more light in the evening, disrupting the body’s natural rhythm,” said Dr. Rishi, of the department of pulmonology, critical care, and sleep medicine, Mayo Clinic, Eau Claire, Wis., and vice chair of the AASM Public Safety Committee.
The position statement was published Aug. 26 in the Journal of Clinical Sleep Medicine to coincide with the virtual annual meeting of the Associated Professional Sleep Societies .
Significant health risks
In the United States, the annual “spring forward” to daylight saving time and “fall back” to standard time is required by law, although under the statute some exceptions are permitted.
There has been intense debate over the last several years about transitioning between standard and daylight saving time. The AASM says there is “an abundance of evidence” to indicate that quick transition from standard time to daylight saving time incurs significant public health and safety risks, including increased risk of heart attack, stroke, mood disorders, and car crashes.
“Although chronic effects of remaining in daylight saving time year-round have not been well-studied, daylight saving time is less aligned with human circadian biology – which, because of the impacts of the delayed natural light/dark cycle on human activity, could result in circadian misalignment, which has been associated in some studies with increased cardiovascular disease risk, metabolic syndrome and other health risks,” the authors wrote.
A recent study also showed an increase in medical errors in the week after switching to daylight saving time.
“Because the adoption of permanent standard time would be beneficial for public health and safety, the AASM will be advocating at the federal level for this legislative change,” said AASM President Kannan Ramar, MBBS, MD, with the Mayo Clinic in Rochester, Minn.
It seems that many Americans are in favor of the change. In July, an AASM survey of roughly 2,000 U.S. adults showed that two-thirds support doing away with the seasonal time change. Only 11% opposed it. In addition, the academy’s 2019 survey showed more than half of adults feel extremely, or somewhat, tired after the springing ahead to daylight saving time.
Strong support
The position statement has been endorsed by 19 organizations, including the American Academy of Cardiovascular Sleep Medicine, American College of Chest Physicians (CHEST), American College of Occupational and Environmental Medicine, National PTA, National Safety Council, Society of Anesthesia and Sleep Medicine, and the Society of Behavioral Sleep Medicine.
Weighing in on the issue, Saul Rothenberg, PhD, from the Sleep Center at Greenwich Hospital, Conn., said the literature on daylight saving time has grown over the past 20 years. He said he was ”humbled” by the research that shows that a “relatively small” misalignment of biological and social clocks has a measurable impact on human health and behavior.
“Because misalignment is associated with negative health and performance outcomes, keeping one set of hours year-round is promoted to minimize misalignment and associated consequences,” he added.
In light of this research, the recommendation to dispense with daylight saving time seems “quite reasonable” from a public health perspective. “I am left with a strengthened view on the importance of regular adequate sleep as a way to enhance health, performance, and quality of life,” he added.
This research had no commercial funding. Dr. Rishi and Dr. Rothenberg have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In the interest of public health and safety, – a recommendation that has garnered strong support from multiple medical and other high-profile organizations.
“Permanent, year-round standard time is the best choice to most closely match our circadian sleep-wake cycle,” M. Adeel Rishi, MD, lead author of the AASM position statement, said in a news release. “Daylight saving time results in more darkness in the morning and more light in the evening, disrupting the body’s natural rhythm,” said Dr. Rishi, of the department of pulmonology, critical care, and sleep medicine, Mayo Clinic, Eau Claire, Wis., and vice chair of the AASM Public Safety Committee.
The position statement was published Aug. 26 in the Journal of Clinical Sleep Medicine to coincide with the virtual annual meeting of the Associated Professional Sleep Societies .
Significant health risks
In the United States, the annual “spring forward” to daylight saving time and “fall back” to standard time is required by law, although under the statute some exceptions are permitted.
There has been intense debate over the last several years about transitioning between standard and daylight saving time. The AASM says there is “an abundance of evidence” to indicate that quick transition from standard time to daylight saving time incurs significant public health and safety risks, including increased risk of heart attack, stroke, mood disorders, and car crashes.
“Although chronic effects of remaining in daylight saving time year-round have not been well-studied, daylight saving time is less aligned with human circadian biology – which, because of the impacts of the delayed natural light/dark cycle on human activity, could result in circadian misalignment, which has been associated in some studies with increased cardiovascular disease risk, metabolic syndrome and other health risks,” the authors wrote.
A recent study also showed an increase in medical errors in the week after switching to daylight saving time.
“Because the adoption of permanent standard time would be beneficial for public health and safety, the AASM will be advocating at the federal level for this legislative change,” said AASM President Kannan Ramar, MBBS, MD, with the Mayo Clinic in Rochester, Minn.
It seems that many Americans are in favor of the change. In July, an AASM survey of roughly 2,000 U.S. adults showed that two-thirds support doing away with the seasonal time change. Only 11% opposed it. In addition, the academy’s 2019 survey showed more than half of adults feel extremely, or somewhat, tired after the springing ahead to daylight saving time.
Strong support
The position statement has been endorsed by 19 organizations, including the American Academy of Cardiovascular Sleep Medicine, American College of Chest Physicians (CHEST), American College of Occupational and Environmental Medicine, National PTA, National Safety Council, Society of Anesthesia and Sleep Medicine, and the Society of Behavioral Sleep Medicine.
Weighing in on the issue, Saul Rothenberg, PhD, from the Sleep Center at Greenwich Hospital, Conn., said the literature on daylight saving time has grown over the past 20 years. He said he was ”humbled” by the research that shows that a “relatively small” misalignment of biological and social clocks has a measurable impact on human health and behavior.
“Because misalignment is associated with negative health and performance outcomes, keeping one set of hours year-round is promoted to minimize misalignment and associated consequences,” he added.
In light of this research, the recommendation to dispense with daylight saving time seems “quite reasonable” from a public health perspective. “I am left with a strengthened view on the importance of regular adequate sleep as a way to enhance health, performance, and quality of life,” he added.
This research had no commercial funding. Dr. Rishi and Dr. Rothenberg have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In the interest of public health and safety, – a recommendation that has garnered strong support from multiple medical and other high-profile organizations.
“Permanent, year-round standard time is the best choice to most closely match our circadian sleep-wake cycle,” M. Adeel Rishi, MD, lead author of the AASM position statement, said in a news release. “Daylight saving time results in more darkness in the morning and more light in the evening, disrupting the body’s natural rhythm,” said Dr. Rishi, of the department of pulmonology, critical care, and sleep medicine, Mayo Clinic, Eau Claire, Wis., and vice chair of the AASM Public Safety Committee.
The position statement was published Aug. 26 in the Journal of Clinical Sleep Medicine to coincide with the virtual annual meeting of the Associated Professional Sleep Societies .
Significant health risks
In the United States, the annual “spring forward” to daylight saving time and “fall back” to standard time is required by law, although under the statute some exceptions are permitted.
There has been intense debate over the last several years about transitioning between standard and daylight saving time. The AASM says there is “an abundance of evidence” to indicate that quick transition from standard time to daylight saving time incurs significant public health and safety risks, including increased risk of heart attack, stroke, mood disorders, and car crashes.
“Although chronic effects of remaining in daylight saving time year-round have not been well-studied, daylight saving time is less aligned with human circadian biology – which, because of the impacts of the delayed natural light/dark cycle on human activity, could result in circadian misalignment, which has been associated in some studies with increased cardiovascular disease risk, metabolic syndrome and other health risks,” the authors wrote.
A recent study also showed an increase in medical errors in the week after switching to daylight saving time.
“Because the adoption of permanent standard time would be beneficial for public health and safety, the AASM will be advocating at the federal level for this legislative change,” said AASM President Kannan Ramar, MBBS, MD, with the Mayo Clinic in Rochester, Minn.
It seems that many Americans are in favor of the change. In July, an AASM survey of roughly 2,000 U.S. adults showed that two-thirds support doing away with the seasonal time change. Only 11% opposed it. In addition, the academy’s 2019 survey showed more than half of adults feel extremely, or somewhat, tired after the springing ahead to daylight saving time.
Strong support
The position statement has been endorsed by 19 organizations, including the American Academy of Cardiovascular Sleep Medicine, American College of Chest Physicians (CHEST), American College of Occupational and Environmental Medicine, National PTA, National Safety Council, Society of Anesthesia and Sleep Medicine, and the Society of Behavioral Sleep Medicine.
Weighing in on the issue, Saul Rothenberg, PhD, from the Sleep Center at Greenwich Hospital, Conn., said the literature on daylight saving time has grown over the past 20 years. He said he was ”humbled” by the research that shows that a “relatively small” misalignment of biological and social clocks has a measurable impact on human health and behavior.
“Because misalignment is associated with negative health and performance outcomes, keeping one set of hours year-round is promoted to minimize misalignment and associated consequences,” he added.
In light of this research, the recommendation to dispense with daylight saving time seems “quite reasonable” from a public health perspective. “I am left with a strengthened view on the importance of regular adequate sleep as a way to enhance health, performance, and quality of life,” he added.
This research had no commercial funding. Dr. Rishi and Dr. Rothenberg have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM SLEEP 2020
Ten ways docs are cutting costs and saving money
“Some of our physician clients have seen their income decrease by as much as 50%,” says Joel Greenwald, MD, CEO of Greenwald Wealth Management in St. Louis Park, Minnesota. “Many physicians had previously figured that whatever financial obligations they had wouldn’t be a problem because whatever amount they were making would continue, and if there were a decline it would be gradual.” However, assumption is now creating financial strain for many doctors.
Vikram Tarugu, MD, a gastroenterologist and CEO of Detox of South Florida in Okeechobee, Florida, says he has watched his budget for years, but has become even more careful with his spending in the past few months.
“It has helped me a lot to adjust to the new normal when it comes to the financial side of things,” Dr. Tarugu said. “Patients aren’t coming in as much as they used to, so my income has really been affected.”
Primary care physicians have seen a 55% decrease in revenue and a 20%-30% decrease in patient volume as a result of COVID-19. The impact has been even more severe for specialists. Even for physicians whose practices remain busy and whose family members haven’t lost their jobs or income, broader concerns about the economy may be reason enough for physicians to adopt cost-cutting measures.
In Medscape’s Physician Compensation Report 2020, we asked physicians to share their best cost-cutting tips. Many illustrate the lengths to which physicians are going to conserve money.
Here’s a look at some of the advice they shared, along with guidance from experts on how to make it work for you:
1. Create a written budget, even if you think it’s pointless.
Physicians said their most important piece of advice includes the following: “Use a formal budget to track progress,” “write out a budget,” “plan intermittent/large expenses in advance,” “Make sure all expenses are paid before you spend on leisure.”
Nearly 7 in 10 physicians say they have a budget for personal expenses, yet only one-quarter of those who do have a formal, written budget. Writing out a spending plan is key to being intentional about your spending, making sure that you’re living within your means, and identifying areas in which you may be able to cut back.
“Financial planning is all about cash flow, and everybody should know the amount of money coming in, how much is going out, and the difference between the two,” says Amy Guerich, a partner with Stepp & Rothwell, a Kansas City–based financial planning firm. “That’s important in good times, but it’s even more important now when we see physicians taking pay cuts.”
Many physicians have found that budget apps or software programs are easier to work with than anticipated; some even walk you through the process of creating a budget. To get the most out of the apps, you’ll need to check them regularly and make changes based on their data.
“Sometimes there’s this false belief that just by signing up, you are automatically going to be better at budgeting,” says Scott Snider, CFP, a certified financial planner and partner with Mellen Money Management in Ponte Vedra Beach, Florida. “Basically, these apps are a great way to identify problem areas of spending. We have a tendency as humans to underestimate how much we spend on things like Starbucks, dining out, and Amazon shopping.”
One of the doctors’ tips that requires the most willpower is to “pay all expenses before spending on leisure.” That’s because we live in an instant gratification world, and want everything right away, Ms. Guerich said.
“I also think there’s an element of ‘keeping up with the Joneses’ and pressure associated with this profession,” she said. “The stereotype is that physicians are high-income earners so ‘We should be able to do that’ or ‘Mom and dad are doctors, so they can afford it.’ “
Creating and then revisiting your budget progress on a monthly or quarterly basis can give you a feeling of accomplishment and keep you motivated to stay with it.
Keep in mind that budgeting is a continual process rather than a singular event, and you’ll likely adjust it over time as your income and goals change.
2. Save more as you earn more.
Respondents to our Physician Compensation Report gave the following recommendations: “Pay yourself first,” “I put half of my bonus into an investment account no matter how much it is,” “I allocate extra money and put it into a savings account.”
Dr. Greenwald said, “I have a rule that every client needs to be saving 20% of their gross income toward retirement, including whatever the employer is putting in.”
Putting a portion of every paycheck into savings is key to making progress toward financial goals. Start by building an emergency fund with at least 3-6 months’ worth of expenses in it and making sure you’re saving at least enough for retirement to get any potential employer match.
Mr. Snider suggests increasing the percentage you save every time you get a raise.
“The thought behind that strategy is that when a doctor receives a pay raise – even if it’s just a cost-of-living raise of 3% – an extra 1% saved doesn’t reduce their take-home pay year-over-year,” he says. “In fact, they still take home more money, and they save more money. Win-win.”
3. Focus on paying down your debt.
Physicians told us how they were working to pay down debt with the following recommendations: “Accelerate debt reduction,” “I make additional principal payment to our home mortgage,” “We are aggressively attacking our remaining student loans.”
Reducing or eliminating debt is key to increasing cash flow, which can make it easier to meet all of your other financial goals. One-quarter of physicians have credit card debt, which typically carries interest rates higher than other types of debt, making it far more expensive. Focus on paying off such high-interest debt first, before moving on to other types of debt such as auto loans, student loans, or a mortgage.
“Credit card debt and any unsecured debt should be paid before anything else,” Mr. Snider says. “Getting rid of those high interest rates should be a priority. And that type of debt has less flexible terms than student debt.”
4. Great opportunity to take advantage of record-low interest rates.
Physicians said that, to save money, they are recommending the following: “Consolidating student debt into our mortgage,” “Accelerating payments of the principle on our mortgage,” “Making sure we have an affordable mortgage.”
With interest rates at an all-time low, even those who’ve recently refinanced might see significant savings by refinancing again. Given the associated fees, it typically makes sense to refinance if you can reduce your mortgage rate by at least a point, and you’re planning to stay in the home for at least 5 years.
“Depending on how much lower your rate is, refinancing can make a big difference in your monthly payments,” Ms. Guerich said. “For physicians who might need an emergency reserve but don’t have cash on hand, a HELOC [Home Equity Line of Credit] is a great way to accomplish that.”
5. Be wary of credit cards dangers; use cards wisely.
Physician respondents recommended the following: “Use 0% interest offers on credit cards,” “Only have one card and pay it off every month,” “Never carry over balance.”
Nearly 80% of physicians have three or more credit cards, with 18% reporting that they have seven or more. When used wisely, credit cards can be an important tool for managing finances. Many credit cards come with tools that can help with budgeting, and credit cards rewards and perks can offer real value to users. That said, rewards typically are not valuable enough to offset the cost of interest on balances (or the associated damage to your credit score) that aren’t paid off each month.
“If you’re paying a high rate on credit card balances that carry over every month, regardless of your income, that could be a symptom that you may be spending more than you should,” says Dan Keady, a CFP and chief financial planning strategist at financial services firm TIAA.
6. Give less to Uncle Sam: Keep it for yourself.
Physicians said that they do the following: “Maximize tax-free/deferred savings (401k, HSA, etc.),” “Give to charity to reduce tax,” “Use pre-tax dollars for childcare and healthcare.”
Not only does saving in workplace retirement accounts help you build your nest egg, but it also reduces the amount that you have to pay in taxes in a given year. Physicians should also take advantage of other ways to reduce their income for tax purposes, such as saving money in a health savings account or flexible savings account.
The 401(k) or 403(b) contribution limit for this year is $19,500 ($26,000) for those age 50 years and older. Self-employed physicians can save even more money via a Simplified Employee Pension (SEP) IRA, says Ms. Guerich said. They can save up to 25% of compensation, up to $57,000 in 2020.
7. Automate everything and spare yourself the headache.
Physicians said the following: “Designate money from your paycheck directly to tax deferred and taxable accounts automatically,” “Use automatic payment for credit card balance monthly,” “Automate your savings.”
You probably already automate your 401(k) contributions, but you can also automate bill payments, emergency savings contributions and other financial tasks. For busy physicians, this can make it easier to stick to your financial plan and achieve your goals.
“The older you get, the busier you get, said Mr. Snider says. “Automation can definitely help with that. But make sure you are checking in quarterly to make sure that everything is still in line with your plan. The problem with automation is when you forget about it completely and just let everything sit there.”
8. Save separately for big purchases.
Sometimes it’s the big major expenses that can start to derail a budget. Physicians told us the following tactics for large purchases: “We buy affordable cars and take budget vacations,” “I buy used cars,” “We save in advance for new cars and only buy cars with cash.”
The decision of which car to purchase or where to go on a family vacation is a personal one, and some physicians take great enjoyment and pride in driving a luxury vehicle or traveling to exotic locales. The key, experts say, is to factor the cost of that car into the rest of your budget, and make sure that it’s not preventing you from achieving other financial goals.
“I don’t like to judge or tell clients how they should spend their money,” said Andrew Musbach, a certified financial planner and cofounder of MD Wealth Management in Chelsea, Mich. “Some people like cars, we have clients that have two planes, others want a second house or like to travel. Each person has their own interest where they may spend more relative to other people, but as long as they are meeting their savings targets, I encourage them to spend their money and enjoy what they enjoy most, guilt free.”
Mr. Snider suggests setting up a savings account separate from emergency or retirement accounts to set aside money if you have a goal for a large future purchase, such as a boat or a second home.
“That way, the funds don’t get commingled, and it’s explicitly clear whether or not the doctor is on target,” he says. “It also prevents them from treating their emergency savings account as an ATM machine.”
9. Start saving for college when the kids are little.
Respondents said the following: “We are buying less to save for the kids’ college education,” “We set up direct deposit into college and retirement savings plans,” “We have a 529 account for college savings.”
Helping pay for their children’s college education is an important financial goal for many physicians. The earlier that you start saving, the less you’ll have to save overall, thanks to compound interest. State 529 accounts are often a good place to start, especially if your state offers a tax incentive for doing so.
Mr. Snider recommends that physicians start small, with an initial investment of $1,000 per month and $100 per month contributions. Assuming a 7% rate of return and 17 years’ worth of savings, this would generate just over $42,000. (Note, current typical rates of return are less than 7%).
“Ideally, as other goals are accomplished and personal debt gets paid off, the doctor is ramping up their savings to have at least 50% of college expenses covered from their 529 college savings,” he says.
10. Watch out for the temptation of impulse purchases.
Physicians said the following: “Avoid impulse purchases,” “Avoid impulse shopping, make a list for the store and stick to it,” “Wait to buy things on sale.”
Nothing wrecks a budget like an impulse buy. More than half (54%) of U.S. shoppers have admitted to spending $100 or more on an impulse purchase. And 20% of shoppers have spent at least $1,000 on an impulse buy. Avoid buyers’ remorse by waiting a few days to make large purchase decisions or by limiting your unplanned spending to a certain dollar amount within your budget.
Online shopping may be a particular temptation. Dr. Tarugu, the Florida gastroenterologist, has focused on reducing those impulse buys as well, deleting all online shopping apps from his and his family’s phones.
“You won’t notice how much you have ordered online until it arrives at your doorstep,” he said. “It’s really important to keep it at bay.”
Mr. Keady, the TIAA chief planning strategist, recommended this tactic: Calculate the number of patients (or hours) you’d need to see in order to earn the cash required to make the purchase.
“Then, in a mindful way, figure out the amount of value derived from the purchase,” he said.
A version of this article originally appeared on Medscape.com.
“Some of our physician clients have seen their income decrease by as much as 50%,” says Joel Greenwald, MD, CEO of Greenwald Wealth Management in St. Louis Park, Minnesota. “Many physicians had previously figured that whatever financial obligations they had wouldn’t be a problem because whatever amount they were making would continue, and if there were a decline it would be gradual.” However, assumption is now creating financial strain for many doctors.
Vikram Tarugu, MD, a gastroenterologist and CEO of Detox of South Florida in Okeechobee, Florida, says he has watched his budget for years, but has become even more careful with his spending in the past few months.
“It has helped me a lot to adjust to the new normal when it comes to the financial side of things,” Dr. Tarugu said. “Patients aren’t coming in as much as they used to, so my income has really been affected.”
Primary care physicians have seen a 55% decrease in revenue and a 20%-30% decrease in patient volume as a result of COVID-19. The impact has been even more severe for specialists. Even for physicians whose practices remain busy and whose family members haven’t lost their jobs or income, broader concerns about the economy may be reason enough for physicians to adopt cost-cutting measures.
In Medscape’s Physician Compensation Report 2020, we asked physicians to share their best cost-cutting tips. Many illustrate the lengths to which physicians are going to conserve money.
Here’s a look at some of the advice they shared, along with guidance from experts on how to make it work for you:
1. Create a written budget, even if you think it’s pointless.
Physicians said their most important piece of advice includes the following: “Use a formal budget to track progress,” “write out a budget,” “plan intermittent/large expenses in advance,” “Make sure all expenses are paid before you spend on leisure.”
Nearly 7 in 10 physicians say they have a budget for personal expenses, yet only one-quarter of those who do have a formal, written budget. Writing out a spending plan is key to being intentional about your spending, making sure that you’re living within your means, and identifying areas in which you may be able to cut back.
“Financial planning is all about cash flow, and everybody should know the amount of money coming in, how much is going out, and the difference between the two,” says Amy Guerich, a partner with Stepp & Rothwell, a Kansas City–based financial planning firm. “That’s important in good times, but it’s even more important now when we see physicians taking pay cuts.”
Many physicians have found that budget apps or software programs are easier to work with than anticipated; some even walk you through the process of creating a budget. To get the most out of the apps, you’ll need to check them regularly and make changes based on their data.
“Sometimes there’s this false belief that just by signing up, you are automatically going to be better at budgeting,” says Scott Snider, CFP, a certified financial planner and partner with Mellen Money Management in Ponte Vedra Beach, Florida. “Basically, these apps are a great way to identify problem areas of spending. We have a tendency as humans to underestimate how much we spend on things like Starbucks, dining out, and Amazon shopping.”
One of the doctors’ tips that requires the most willpower is to “pay all expenses before spending on leisure.” That’s because we live in an instant gratification world, and want everything right away, Ms. Guerich said.
“I also think there’s an element of ‘keeping up with the Joneses’ and pressure associated with this profession,” she said. “The stereotype is that physicians are high-income earners so ‘We should be able to do that’ or ‘Mom and dad are doctors, so they can afford it.’ “
Creating and then revisiting your budget progress on a monthly or quarterly basis can give you a feeling of accomplishment and keep you motivated to stay with it.
Keep in mind that budgeting is a continual process rather than a singular event, and you’ll likely adjust it over time as your income and goals change.
2. Save more as you earn more.
Respondents to our Physician Compensation Report gave the following recommendations: “Pay yourself first,” “I put half of my bonus into an investment account no matter how much it is,” “I allocate extra money and put it into a savings account.”
Dr. Greenwald said, “I have a rule that every client needs to be saving 20% of their gross income toward retirement, including whatever the employer is putting in.”
Putting a portion of every paycheck into savings is key to making progress toward financial goals. Start by building an emergency fund with at least 3-6 months’ worth of expenses in it and making sure you’re saving at least enough for retirement to get any potential employer match.
Mr. Snider suggests increasing the percentage you save every time you get a raise.
“The thought behind that strategy is that when a doctor receives a pay raise – even if it’s just a cost-of-living raise of 3% – an extra 1% saved doesn’t reduce their take-home pay year-over-year,” he says. “In fact, they still take home more money, and they save more money. Win-win.”
3. Focus on paying down your debt.
Physicians told us how they were working to pay down debt with the following recommendations: “Accelerate debt reduction,” “I make additional principal payment to our home mortgage,” “We are aggressively attacking our remaining student loans.”
Reducing or eliminating debt is key to increasing cash flow, which can make it easier to meet all of your other financial goals. One-quarter of physicians have credit card debt, which typically carries interest rates higher than other types of debt, making it far more expensive. Focus on paying off such high-interest debt first, before moving on to other types of debt such as auto loans, student loans, or a mortgage.
“Credit card debt and any unsecured debt should be paid before anything else,” Mr. Snider says. “Getting rid of those high interest rates should be a priority. And that type of debt has less flexible terms than student debt.”
4. Great opportunity to take advantage of record-low interest rates.
Physicians said that, to save money, they are recommending the following: “Consolidating student debt into our mortgage,” “Accelerating payments of the principle on our mortgage,” “Making sure we have an affordable mortgage.”
With interest rates at an all-time low, even those who’ve recently refinanced might see significant savings by refinancing again. Given the associated fees, it typically makes sense to refinance if you can reduce your mortgage rate by at least a point, and you’re planning to stay in the home for at least 5 years.
“Depending on how much lower your rate is, refinancing can make a big difference in your monthly payments,” Ms. Guerich said. “For physicians who might need an emergency reserve but don’t have cash on hand, a HELOC [Home Equity Line of Credit] is a great way to accomplish that.”
5. Be wary of credit cards dangers; use cards wisely.
Physician respondents recommended the following: “Use 0% interest offers on credit cards,” “Only have one card and pay it off every month,” “Never carry over balance.”
Nearly 80% of physicians have three or more credit cards, with 18% reporting that they have seven or more. When used wisely, credit cards can be an important tool for managing finances. Many credit cards come with tools that can help with budgeting, and credit cards rewards and perks can offer real value to users. That said, rewards typically are not valuable enough to offset the cost of interest on balances (or the associated damage to your credit score) that aren’t paid off each month.
“If you’re paying a high rate on credit card balances that carry over every month, regardless of your income, that could be a symptom that you may be spending more than you should,” says Dan Keady, a CFP and chief financial planning strategist at financial services firm TIAA.
6. Give less to Uncle Sam: Keep it for yourself.
Physicians said that they do the following: “Maximize tax-free/deferred savings (401k, HSA, etc.),” “Give to charity to reduce tax,” “Use pre-tax dollars for childcare and healthcare.”
Not only does saving in workplace retirement accounts help you build your nest egg, but it also reduces the amount that you have to pay in taxes in a given year. Physicians should also take advantage of other ways to reduce their income for tax purposes, such as saving money in a health savings account or flexible savings account.
The 401(k) or 403(b) contribution limit for this year is $19,500 ($26,000) for those age 50 years and older. Self-employed physicians can save even more money via a Simplified Employee Pension (SEP) IRA, says Ms. Guerich said. They can save up to 25% of compensation, up to $57,000 in 2020.
7. Automate everything and spare yourself the headache.
Physicians said the following: “Designate money from your paycheck directly to tax deferred and taxable accounts automatically,” “Use automatic payment for credit card balance monthly,” “Automate your savings.”
You probably already automate your 401(k) contributions, but you can also automate bill payments, emergency savings contributions and other financial tasks. For busy physicians, this can make it easier to stick to your financial plan and achieve your goals.
“The older you get, the busier you get, said Mr. Snider says. “Automation can definitely help with that. But make sure you are checking in quarterly to make sure that everything is still in line with your plan. The problem with automation is when you forget about it completely and just let everything sit there.”
8. Save separately for big purchases.
Sometimes it’s the big major expenses that can start to derail a budget. Physicians told us the following tactics for large purchases: “We buy affordable cars and take budget vacations,” “I buy used cars,” “We save in advance for new cars and only buy cars with cash.”
The decision of which car to purchase or where to go on a family vacation is a personal one, and some physicians take great enjoyment and pride in driving a luxury vehicle or traveling to exotic locales. The key, experts say, is to factor the cost of that car into the rest of your budget, and make sure that it’s not preventing you from achieving other financial goals.
“I don’t like to judge or tell clients how they should spend their money,” said Andrew Musbach, a certified financial planner and cofounder of MD Wealth Management in Chelsea, Mich. “Some people like cars, we have clients that have two planes, others want a second house or like to travel. Each person has their own interest where they may spend more relative to other people, but as long as they are meeting their savings targets, I encourage them to spend their money and enjoy what they enjoy most, guilt free.”
Mr. Snider suggests setting up a savings account separate from emergency or retirement accounts to set aside money if you have a goal for a large future purchase, such as a boat or a second home.
“That way, the funds don’t get commingled, and it’s explicitly clear whether or not the doctor is on target,” he says. “It also prevents them from treating their emergency savings account as an ATM machine.”
9. Start saving for college when the kids are little.
Respondents said the following: “We are buying less to save for the kids’ college education,” “We set up direct deposit into college and retirement savings plans,” “We have a 529 account for college savings.”
Helping pay for their children’s college education is an important financial goal for many physicians. The earlier that you start saving, the less you’ll have to save overall, thanks to compound interest. State 529 accounts are often a good place to start, especially if your state offers a tax incentive for doing so.
Mr. Snider recommends that physicians start small, with an initial investment of $1,000 per month and $100 per month contributions. Assuming a 7% rate of return and 17 years’ worth of savings, this would generate just over $42,000. (Note, current typical rates of return are less than 7%).
“Ideally, as other goals are accomplished and personal debt gets paid off, the doctor is ramping up their savings to have at least 50% of college expenses covered from their 529 college savings,” he says.
10. Watch out for the temptation of impulse purchases.
Physicians said the following: “Avoid impulse purchases,” “Avoid impulse shopping, make a list for the store and stick to it,” “Wait to buy things on sale.”
Nothing wrecks a budget like an impulse buy. More than half (54%) of U.S. shoppers have admitted to spending $100 or more on an impulse purchase. And 20% of shoppers have spent at least $1,000 on an impulse buy. Avoid buyers’ remorse by waiting a few days to make large purchase decisions or by limiting your unplanned spending to a certain dollar amount within your budget.
Online shopping may be a particular temptation. Dr. Tarugu, the Florida gastroenterologist, has focused on reducing those impulse buys as well, deleting all online shopping apps from his and his family’s phones.
“You won’t notice how much you have ordered online until it arrives at your doorstep,” he said. “It’s really important to keep it at bay.”
Mr. Keady, the TIAA chief planning strategist, recommended this tactic: Calculate the number of patients (or hours) you’d need to see in order to earn the cash required to make the purchase.
“Then, in a mindful way, figure out the amount of value derived from the purchase,” he said.
A version of this article originally appeared on Medscape.com.
“Some of our physician clients have seen their income decrease by as much as 50%,” says Joel Greenwald, MD, CEO of Greenwald Wealth Management in St. Louis Park, Minnesota. “Many physicians had previously figured that whatever financial obligations they had wouldn’t be a problem because whatever amount they were making would continue, and if there were a decline it would be gradual.” However, assumption is now creating financial strain for many doctors.
Vikram Tarugu, MD, a gastroenterologist and CEO of Detox of South Florida in Okeechobee, Florida, says he has watched his budget for years, but has become even more careful with his spending in the past few months.
“It has helped me a lot to adjust to the new normal when it comes to the financial side of things,” Dr. Tarugu said. “Patients aren’t coming in as much as they used to, so my income has really been affected.”
Primary care physicians have seen a 55% decrease in revenue and a 20%-30% decrease in patient volume as a result of COVID-19. The impact has been even more severe for specialists. Even for physicians whose practices remain busy and whose family members haven’t lost their jobs or income, broader concerns about the economy may be reason enough for physicians to adopt cost-cutting measures.
In Medscape’s Physician Compensation Report 2020, we asked physicians to share their best cost-cutting tips. Many illustrate the lengths to which physicians are going to conserve money.
Here’s a look at some of the advice they shared, along with guidance from experts on how to make it work for you:
1. Create a written budget, even if you think it’s pointless.
Physicians said their most important piece of advice includes the following: “Use a formal budget to track progress,” “write out a budget,” “plan intermittent/large expenses in advance,” “Make sure all expenses are paid before you spend on leisure.”
Nearly 7 in 10 physicians say they have a budget for personal expenses, yet only one-quarter of those who do have a formal, written budget. Writing out a spending plan is key to being intentional about your spending, making sure that you’re living within your means, and identifying areas in which you may be able to cut back.
“Financial planning is all about cash flow, and everybody should know the amount of money coming in, how much is going out, and the difference between the two,” says Amy Guerich, a partner with Stepp & Rothwell, a Kansas City–based financial planning firm. “That’s important in good times, but it’s even more important now when we see physicians taking pay cuts.”
Many physicians have found that budget apps or software programs are easier to work with than anticipated; some even walk you through the process of creating a budget. To get the most out of the apps, you’ll need to check them regularly and make changes based on their data.
“Sometimes there’s this false belief that just by signing up, you are automatically going to be better at budgeting,” says Scott Snider, CFP, a certified financial planner and partner with Mellen Money Management in Ponte Vedra Beach, Florida. “Basically, these apps are a great way to identify problem areas of spending. We have a tendency as humans to underestimate how much we spend on things like Starbucks, dining out, and Amazon shopping.”
One of the doctors’ tips that requires the most willpower is to “pay all expenses before spending on leisure.” That’s because we live in an instant gratification world, and want everything right away, Ms. Guerich said.
“I also think there’s an element of ‘keeping up with the Joneses’ and pressure associated with this profession,” she said. “The stereotype is that physicians are high-income earners so ‘We should be able to do that’ or ‘Mom and dad are doctors, so they can afford it.’ “
Creating and then revisiting your budget progress on a monthly or quarterly basis can give you a feeling of accomplishment and keep you motivated to stay with it.
Keep in mind that budgeting is a continual process rather than a singular event, and you’ll likely adjust it over time as your income and goals change.
2. Save more as you earn more.
Respondents to our Physician Compensation Report gave the following recommendations: “Pay yourself first,” “I put half of my bonus into an investment account no matter how much it is,” “I allocate extra money and put it into a savings account.”
Dr. Greenwald said, “I have a rule that every client needs to be saving 20% of their gross income toward retirement, including whatever the employer is putting in.”
Putting a portion of every paycheck into savings is key to making progress toward financial goals. Start by building an emergency fund with at least 3-6 months’ worth of expenses in it and making sure you’re saving at least enough for retirement to get any potential employer match.
Mr. Snider suggests increasing the percentage you save every time you get a raise.
“The thought behind that strategy is that when a doctor receives a pay raise – even if it’s just a cost-of-living raise of 3% – an extra 1% saved doesn’t reduce their take-home pay year-over-year,” he says. “In fact, they still take home more money, and they save more money. Win-win.”
3. Focus on paying down your debt.
Physicians told us how they were working to pay down debt with the following recommendations: “Accelerate debt reduction,” “I make additional principal payment to our home mortgage,” “We are aggressively attacking our remaining student loans.”
Reducing or eliminating debt is key to increasing cash flow, which can make it easier to meet all of your other financial goals. One-quarter of physicians have credit card debt, which typically carries interest rates higher than other types of debt, making it far more expensive. Focus on paying off such high-interest debt first, before moving on to other types of debt such as auto loans, student loans, or a mortgage.
“Credit card debt and any unsecured debt should be paid before anything else,” Mr. Snider says. “Getting rid of those high interest rates should be a priority. And that type of debt has less flexible terms than student debt.”
4. Great opportunity to take advantage of record-low interest rates.
Physicians said that, to save money, they are recommending the following: “Consolidating student debt into our mortgage,” “Accelerating payments of the principle on our mortgage,” “Making sure we have an affordable mortgage.”
With interest rates at an all-time low, even those who’ve recently refinanced might see significant savings by refinancing again. Given the associated fees, it typically makes sense to refinance if you can reduce your mortgage rate by at least a point, and you’re planning to stay in the home for at least 5 years.
“Depending on how much lower your rate is, refinancing can make a big difference in your monthly payments,” Ms. Guerich said. “For physicians who might need an emergency reserve but don’t have cash on hand, a HELOC [Home Equity Line of Credit] is a great way to accomplish that.”
5. Be wary of credit cards dangers; use cards wisely.
Physician respondents recommended the following: “Use 0% interest offers on credit cards,” “Only have one card and pay it off every month,” “Never carry over balance.”
Nearly 80% of physicians have three or more credit cards, with 18% reporting that they have seven or more. When used wisely, credit cards can be an important tool for managing finances. Many credit cards come with tools that can help with budgeting, and credit cards rewards and perks can offer real value to users. That said, rewards typically are not valuable enough to offset the cost of interest on balances (or the associated damage to your credit score) that aren’t paid off each month.
“If you’re paying a high rate on credit card balances that carry over every month, regardless of your income, that could be a symptom that you may be spending more than you should,” says Dan Keady, a CFP and chief financial planning strategist at financial services firm TIAA.
6. Give less to Uncle Sam: Keep it for yourself.
Physicians said that they do the following: “Maximize tax-free/deferred savings (401k, HSA, etc.),” “Give to charity to reduce tax,” “Use pre-tax dollars for childcare and healthcare.”
Not only does saving in workplace retirement accounts help you build your nest egg, but it also reduces the amount that you have to pay in taxes in a given year. Physicians should also take advantage of other ways to reduce their income for tax purposes, such as saving money in a health savings account or flexible savings account.
The 401(k) or 403(b) contribution limit for this year is $19,500 ($26,000) for those age 50 years and older. Self-employed physicians can save even more money via a Simplified Employee Pension (SEP) IRA, says Ms. Guerich said. They can save up to 25% of compensation, up to $57,000 in 2020.
7. Automate everything and spare yourself the headache.
Physicians said the following: “Designate money from your paycheck directly to tax deferred and taxable accounts automatically,” “Use automatic payment for credit card balance monthly,” “Automate your savings.”
You probably already automate your 401(k) contributions, but you can also automate bill payments, emergency savings contributions and other financial tasks. For busy physicians, this can make it easier to stick to your financial plan and achieve your goals.
“The older you get, the busier you get, said Mr. Snider says. “Automation can definitely help with that. But make sure you are checking in quarterly to make sure that everything is still in line with your plan. The problem with automation is when you forget about it completely and just let everything sit there.”
8. Save separately for big purchases.
Sometimes it’s the big major expenses that can start to derail a budget. Physicians told us the following tactics for large purchases: “We buy affordable cars and take budget vacations,” “I buy used cars,” “We save in advance for new cars and only buy cars with cash.”
The decision of which car to purchase or where to go on a family vacation is a personal one, and some physicians take great enjoyment and pride in driving a luxury vehicle or traveling to exotic locales. The key, experts say, is to factor the cost of that car into the rest of your budget, and make sure that it’s not preventing you from achieving other financial goals.
“I don’t like to judge or tell clients how they should spend their money,” said Andrew Musbach, a certified financial planner and cofounder of MD Wealth Management in Chelsea, Mich. “Some people like cars, we have clients that have two planes, others want a second house or like to travel. Each person has their own interest where they may spend more relative to other people, but as long as they are meeting their savings targets, I encourage them to spend their money and enjoy what they enjoy most, guilt free.”
Mr. Snider suggests setting up a savings account separate from emergency or retirement accounts to set aside money if you have a goal for a large future purchase, such as a boat or a second home.
“That way, the funds don’t get commingled, and it’s explicitly clear whether or not the doctor is on target,” he says. “It also prevents them from treating their emergency savings account as an ATM machine.”
9. Start saving for college when the kids are little.
Respondents said the following: “We are buying less to save for the kids’ college education,” “We set up direct deposit into college and retirement savings plans,” “We have a 529 account for college savings.”
Helping pay for their children’s college education is an important financial goal for many physicians. The earlier that you start saving, the less you’ll have to save overall, thanks to compound interest. State 529 accounts are often a good place to start, especially if your state offers a tax incentive for doing so.
Mr. Snider recommends that physicians start small, with an initial investment of $1,000 per month and $100 per month contributions. Assuming a 7% rate of return and 17 years’ worth of savings, this would generate just over $42,000. (Note, current typical rates of return are less than 7%).
“Ideally, as other goals are accomplished and personal debt gets paid off, the doctor is ramping up their savings to have at least 50% of college expenses covered from their 529 college savings,” he says.
10. Watch out for the temptation of impulse purchases.
Physicians said the following: “Avoid impulse purchases,” “Avoid impulse shopping, make a list for the store and stick to it,” “Wait to buy things on sale.”
Nothing wrecks a budget like an impulse buy. More than half (54%) of U.S. shoppers have admitted to spending $100 or more on an impulse purchase. And 20% of shoppers have spent at least $1,000 on an impulse buy. Avoid buyers’ remorse by waiting a few days to make large purchase decisions or by limiting your unplanned spending to a certain dollar amount within your budget.
Online shopping may be a particular temptation. Dr. Tarugu, the Florida gastroenterologist, has focused on reducing those impulse buys as well, deleting all online shopping apps from his and his family’s phones.
“You won’t notice how much you have ordered online until it arrives at your doorstep,” he said. “It’s really important to keep it at bay.”
Mr. Keady, the TIAA chief planning strategist, recommended this tactic: Calculate the number of patients (or hours) you’d need to see in order to earn the cash required to make the purchase.
“Then, in a mindful way, figure out the amount of value derived from the purchase,” he said.
A version of this article originally appeared on Medscape.com.
Blood biomarkers could help predict when athletes recover from concussions
, according to a new study of collegiate athletes and recovery time. “Although preliminary, the current results highlight the potential role of biomarkers in tracking neuronal recovery, which may be associated with duration of [return to sport],” wrote Cassandra L. Pattinson, PhD, of the University of Queensland, Brisbane, Australia, and the National Institutes of Health, Bethesda, Md., along with coauthors. The study was published in JAMA Network Open.
To determine if three specific blood biomarkers – total tau protein, glial fibrillary acidic protein (GFAP), and neurofilament light chain protein (NfL) – can help predict when athletes should return from sports-related concussions, a multicenter, prospective diagnostic study was launched and led by the Advanced Research Core (ARC) of the Concussion Assessment, Research, and Education (CARE) Consortium. The consortium is a joint effort of the National Collegiate Athletics Association (NCAA) and the U.S. Department of Defense.
From among the CARE ARC database, researchers evaluated 127 eligible student athletes who had experienced a sports-related concussion, underwent clinical testing and blood collection before and after their injuries, and returned to their sports. Their average age was 18.9 years old, 76% were men, and 65% were White. Biomarker levels were measured from nonfasting blood samples via ultrasensitive single molecule array technology. As current NCAA guidelines indicate that most athletes will be asymptomatic roughly 2 weeks after a concussion, the study used 14 days as a cutoff period.
Among the 127 athletes, the median return-to-sport time was 14 days; 65 returned to their sports in less than 14 days while 62 returned to their sports in 14 days or more. According to the study’s linear mixed models, athletes with a return-to-sport time of 14 days or longer had significantly higher total tau levels at 24-48 hours post injury (mean difference –0.51 pg/mL, 95% confidence interval, –0.88 to –0.14; P = .008) and when symptoms had resolved (mean difference –0.71 pg/mL, 95% CI, –1.09 to –0.34; P < .001) compared with athletes with a return-to-sport time of less than 14 days. Athletes who returned in 14 days or more also had comparatively lower levels of GFAP postinjury than did those who returned in under 14 days (4.39 pg/mL versus 4.72 pg/mL; P = .04).
Preliminary steps toward an appropriate point-of-care test
“This particular study is one of several emerging studies on what these biomarkers look like,” Brian W. Hainline, MD, chief medical officer of the NCAA, said in an interview. “It’s all still very preliminary – you couldn’t make policy changes based on what we have – but the data is accumulating. Ultimately, we should be able to perform a multivariate analysis of all the different objective biomarkers, looking at repetitive head impact exposure, looking at imaging, looking at these blood-based biomarkers. Then you can say, ‘OK, what can we do? Can we actually predict recovery, who is likely or less likely to do well?’ ”
“It’s not realistic to be taking blood samples all the time,” said Dr. Hainline, who was not involved in the study. “Another goal, once we know which biomarkers are valuable, is to convert to a point-of-care test. You get a finger prick or even a salivary test and we get the result immediately; that’s the direction that all of this is heading. But first, we have to lay out the groundwork. We envision a day, in the not too distant future, where we can get this information much more quickly.”
The authors acknowledged their study’s limitations, including an inability to standardize the time of biomarker collection and the fact that they analyzed a “relatively small number of athletes” who met their specific criteria. That said, they emphasized that their work is based on “the largest prospective sample of sports-related concussions in athletes to date” and that they “anticipate that we will be able to continue to gather a more representative sample” in the future to better generalize to the larger collegiate community.
The study was supported by the Grand Alliance Concussion Assessment, Research, and Education Consortium, which was funded in part by the NCAA and the Department of Defense. The authors disclosed receiving grants and travel reimbursements from – or working as advisers or consultants for – various organizations, college programs, and sports leagues.
SOURCE: Pattinson CL, et al. JAMA Netw Open. 2020 Aug 27. doi: 10.1001/jamanetworkopen.2020.13191.
, according to a new study of collegiate athletes and recovery time. “Although preliminary, the current results highlight the potential role of biomarkers in tracking neuronal recovery, which may be associated with duration of [return to sport],” wrote Cassandra L. Pattinson, PhD, of the University of Queensland, Brisbane, Australia, and the National Institutes of Health, Bethesda, Md., along with coauthors. The study was published in JAMA Network Open.
To determine if three specific blood biomarkers – total tau protein, glial fibrillary acidic protein (GFAP), and neurofilament light chain protein (NfL) – can help predict when athletes should return from sports-related concussions, a multicenter, prospective diagnostic study was launched and led by the Advanced Research Core (ARC) of the Concussion Assessment, Research, and Education (CARE) Consortium. The consortium is a joint effort of the National Collegiate Athletics Association (NCAA) and the U.S. Department of Defense.
From among the CARE ARC database, researchers evaluated 127 eligible student athletes who had experienced a sports-related concussion, underwent clinical testing and blood collection before and after their injuries, and returned to their sports. Their average age was 18.9 years old, 76% were men, and 65% were White. Biomarker levels were measured from nonfasting blood samples via ultrasensitive single molecule array technology. As current NCAA guidelines indicate that most athletes will be asymptomatic roughly 2 weeks after a concussion, the study used 14 days as a cutoff period.
Among the 127 athletes, the median return-to-sport time was 14 days; 65 returned to their sports in less than 14 days while 62 returned to their sports in 14 days or more. According to the study’s linear mixed models, athletes with a return-to-sport time of 14 days or longer had significantly higher total tau levels at 24-48 hours post injury (mean difference –0.51 pg/mL, 95% confidence interval, –0.88 to –0.14; P = .008) and when symptoms had resolved (mean difference –0.71 pg/mL, 95% CI, –1.09 to –0.34; P < .001) compared with athletes with a return-to-sport time of less than 14 days. Athletes who returned in 14 days or more also had comparatively lower levels of GFAP postinjury than did those who returned in under 14 days (4.39 pg/mL versus 4.72 pg/mL; P = .04).
Preliminary steps toward an appropriate point-of-care test
“This particular study is one of several emerging studies on what these biomarkers look like,” Brian W. Hainline, MD, chief medical officer of the NCAA, said in an interview. “It’s all still very preliminary – you couldn’t make policy changes based on what we have – but the data is accumulating. Ultimately, we should be able to perform a multivariate analysis of all the different objective biomarkers, looking at repetitive head impact exposure, looking at imaging, looking at these blood-based biomarkers. Then you can say, ‘OK, what can we do? Can we actually predict recovery, who is likely or less likely to do well?’ ”
“It’s not realistic to be taking blood samples all the time,” said Dr. Hainline, who was not involved in the study. “Another goal, once we know which biomarkers are valuable, is to convert to a point-of-care test. You get a finger prick or even a salivary test and we get the result immediately; that’s the direction that all of this is heading. But first, we have to lay out the groundwork. We envision a day, in the not too distant future, where we can get this information much more quickly.”
The authors acknowledged their study’s limitations, including an inability to standardize the time of biomarker collection and the fact that they analyzed a “relatively small number of athletes” who met their specific criteria. That said, they emphasized that their work is based on “the largest prospective sample of sports-related concussions in athletes to date” and that they “anticipate that we will be able to continue to gather a more representative sample” in the future to better generalize to the larger collegiate community.
The study was supported by the Grand Alliance Concussion Assessment, Research, and Education Consortium, which was funded in part by the NCAA and the Department of Defense. The authors disclosed receiving grants and travel reimbursements from – or working as advisers or consultants for – various organizations, college programs, and sports leagues.
SOURCE: Pattinson CL, et al. JAMA Netw Open. 2020 Aug 27. doi: 10.1001/jamanetworkopen.2020.13191.
, according to a new study of collegiate athletes and recovery time. “Although preliminary, the current results highlight the potential role of biomarkers in tracking neuronal recovery, which may be associated with duration of [return to sport],” wrote Cassandra L. Pattinson, PhD, of the University of Queensland, Brisbane, Australia, and the National Institutes of Health, Bethesda, Md., along with coauthors. The study was published in JAMA Network Open.
To determine if three specific blood biomarkers – total tau protein, glial fibrillary acidic protein (GFAP), and neurofilament light chain protein (NfL) – can help predict when athletes should return from sports-related concussions, a multicenter, prospective diagnostic study was launched and led by the Advanced Research Core (ARC) of the Concussion Assessment, Research, and Education (CARE) Consortium. The consortium is a joint effort of the National Collegiate Athletics Association (NCAA) and the U.S. Department of Defense.
From among the CARE ARC database, researchers evaluated 127 eligible student athletes who had experienced a sports-related concussion, underwent clinical testing and blood collection before and after their injuries, and returned to their sports. Their average age was 18.9 years old, 76% were men, and 65% were White. Biomarker levels were measured from nonfasting blood samples via ultrasensitive single molecule array technology. As current NCAA guidelines indicate that most athletes will be asymptomatic roughly 2 weeks after a concussion, the study used 14 days as a cutoff period.
Among the 127 athletes, the median return-to-sport time was 14 days; 65 returned to their sports in less than 14 days while 62 returned to their sports in 14 days or more. According to the study’s linear mixed models, athletes with a return-to-sport time of 14 days or longer had significantly higher total tau levels at 24-48 hours post injury (mean difference –0.51 pg/mL, 95% confidence interval, –0.88 to –0.14; P = .008) and when symptoms had resolved (mean difference –0.71 pg/mL, 95% CI, –1.09 to –0.34; P < .001) compared with athletes with a return-to-sport time of less than 14 days. Athletes who returned in 14 days or more also had comparatively lower levels of GFAP postinjury than did those who returned in under 14 days (4.39 pg/mL versus 4.72 pg/mL; P = .04).
Preliminary steps toward an appropriate point-of-care test
“This particular study is one of several emerging studies on what these biomarkers look like,” Brian W. Hainline, MD, chief medical officer of the NCAA, said in an interview. “It’s all still very preliminary – you couldn’t make policy changes based on what we have – but the data is accumulating. Ultimately, we should be able to perform a multivariate analysis of all the different objective biomarkers, looking at repetitive head impact exposure, looking at imaging, looking at these blood-based biomarkers. Then you can say, ‘OK, what can we do? Can we actually predict recovery, who is likely or less likely to do well?’ ”
“It’s not realistic to be taking blood samples all the time,” said Dr. Hainline, who was not involved in the study. “Another goal, once we know which biomarkers are valuable, is to convert to a point-of-care test. You get a finger prick or even a salivary test and we get the result immediately; that’s the direction that all of this is heading. But first, we have to lay out the groundwork. We envision a day, in the not too distant future, where we can get this information much more quickly.”
The authors acknowledged their study’s limitations, including an inability to standardize the time of biomarker collection and the fact that they analyzed a “relatively small number of athletes” who met their specific criteria. That said, they emphasized that their work is based on “the largest prospective sample of sports-related concussions in athletes to date” and that they “anticipate that we will be able to continue to gather a more representative sample” in the future to better generalize to the larger collegiate community.
The study was supported by the Grand Alliance Concussion Assessment, Research, and Education Consortium, which was funded in part by the NCAA and the Department of Defense. The authors disclosed receiving grants and travel reimbursements from – or working as advisers or consultants for – various organizations, college programs, and sports leagues.
SOURCE: Pattinson CL, et al. JAMA Netw Open. 2020 Aug 27. doi: 10.1001/jamanetworkopen.2020.13191.
FROM JAMA NETWORK OPEN
Asymptomatic children may transmit COVID-19 in communities
About 22% of children with COVID-19 infections were asymptomatic, and 66% of the symptomatic children had unrecognized symptoms at the time of diagnosis, based on data from a case series of 91 confirmed cases.
Although recent reports suggest that COVID-19 infections in children are generally mild, data on the full spectrum of illness and duration of viral RNA in children are limited, wrote Mi Seon Han, MD, PhD, of Seoul (South Korea) Metropolitan Government–Seoul National University Boramae Medical Center, and colleagues.
To examine the full clinical course and duration of COVID-19 RNA detectability in children with confirmed infections, the researchers reviewed data from 91 individuals with confirmed infections. The children ranged in age from 27 days to 18 years, and 58% were male. The children were monitored at 20 hospitals and 2 isolation facilities for a mean 21.9 days. The findings were published in JAMA Pediatrics.
Overall, COVID-19 viral RNA was present in the study population for a mean 17.6 days, with testing done at a median interval of 3 days. A total of 20 children (22%) were asymptomatic throughout the study period. In these children, viral RNA was detected for a mean 14 days.
“The major hurdle implicated in this study in diagnosing and treating children with COVID-19 is that the researchers noted.
Of the 71 symptomatic children, 47 (66%) had unrecognized symptoms prior to diagnosis, 18 (25%) developed symptoms after diagnosis, and 6 (9%) were diagnosed at the time of symptom onset. The symptomatic children were symptomatic for a median of 11 days; 43 (61%) remained symptomatic at 7 days’ follow-up after the study period, 27 (38%) were symptomatic at 14 days, and 7 (10%) were symptomatic at 21 days.
A total of 41 children had upper respiratory infections (58%) and 22 children (24%) had lower respiratory tract infections. No difference in the duration of virus RNA was detected between children with upper respiratory tract infections and lower respiratory tract infections (average, 18.7 days vs. 19.9 days).
Among the symptomatic children, 46 (65%) had mild cases and 20 (28%) had moderate cases.
For treatment, 14 children (15%) received lopinavir-ritonavir and/or hydroxychloroquine. Two patients had severe illness and received oxygen via nasal prong, without the need for mechanical ventilation. All the children in the case series recovered from their infections with no fatalities.
The study’s main limitation was the inability to analyze the transmission potential of the children because of the quarantine and isolation policies in Korea, the researchers noted. In addition, the researchers did not perform follow-up testing at consistent intervals, so the duration of COVID-19 RNA detection may be inexact.
However, the results suggest “that suspecting and diagnosing COVID-19 in children based on their symptoms without epidemiologic information and virus testing is very challenging,” the researchers emphasized.
“Most of the children with COVID-19 have silent disease, but SARS-CoV-2 RNA can still be detected in the respiratory tract for a prolonged period,” they wrote. More research is needed to explore the potential for disease transmission by children in the community, and increased surveillance with laboratory screening can help identify children with unrecognized infections.
The study is the first known to focus on the frequency of asymptomatic infection in children and the duration of symptoms in both asymptomatic and symptomatic children, Roberta L. DeBiasi, MD, and Meghan Delaney, DO, both affiliated with Children’s National Hospital and Research Institute, Washington, and George Washington University, Washington, wrote in an accompanying editorial. The structure of the Korean public health system “allowed for the sequential observation, testing (median testing interval of every 3 days), and comparison of 91 asymptomatic, presymptomatic, and symptomatic children with mild to moderate upper and lower respiratory tract infection, identified primarily by contact tracing from laboratory-proven cases.”
Two take-home points from the study are that not all infected children are symptomatic, and the duration of symptoms in those who are varies widely, they noted. “Interestingly, this study aligns with adult data in which up to 40% of adults may remain asymptomatic in the face of infection.”
However, “The third and most important take-home point from this study relates to the duration of viral shedding in infected pediatric patients,” Dr. DeBiasi and Dr. Delaney said (JAMA Pediatr. 2020 Aug 28. doi: 10.1001/jamapediatrics.2020.3996).
“Fully half of symptomatic children with both upper and lower tract disease were still shedding virus at 21 days. These are striking data, particularly since 86 of 88 diagnosed children (98%) either had no symptoms or mild or moderate disease,” they explained. The results highlight the need for improvements in qualitative molecular testing and formal studies to identify differences in results from different testing scenarios, such as hospital entry, preprocedure screening, and symptomatic testing. In addition, “these findings are highly relevant to the development of public health strategies to mitigate and contain spread within communities, particularly as affected communities begin their recovery phases.”
The study is important because “schools are opening, and we don’t know what is going to happen,” Michael E. Pichichero, MD, of Rochester General Hospital, N.Y., said in an interview.
“Clinicians, parents, students, school administrators and politicians are worried,” he said. “This study adds to others recently published, bringing into focus the challenges to several suppositions that existed when the COVID-19 pandemic began and over the summer.”
“This study of 91 Korean children tells us that taking a child’s temperature as a screening tool to decide if they may enter school will not be a highly successful strategy,” he said. “Many children are without fever and asymptomatic when infected and contagious. The notion that children shed less virus or shed it for shorter lengths of time we keep learning from this type of research is not true. In another recent study the authors found that children shed as much of the SARS-CoV-2 virus as an adult in the ICU on a ventilator.”
Dr. Pichichero said he was not surprised by the study findings. “A similar paper was published last week in the Journal of Pediatrics from Massachusetts General Hospital, so the findings in the JAMA paper are similar to what has been reported in the United States.”
“Availability of testing will continue to be a challenge in some communities,” said Dr. Pichichero. “Here in the Rochester, New York, area we will use a screening questionnaire based on the CDC [Centers for Disease Control and Prevention] symptom criteria of SARS-CoV-2 infections to decide whom to test.”
As for additional research, “We have so much more to learn about SARS-CoV-2 in children,” he emphasized. “The focus has been on adults because the morbidity and mortality has been greatest in adults, especially the elderly and those with compromised health.”
“The National Institutes of Health has issued a call for more research in children to characterize the spectrum of SARS-CoV-2 illness, including the multisystem inflammatory syndrome in children [MIS-C] and try to identify biomarkers and/or biosignatures for a prognostic algorithm to predict the longitudinal risk of disease severity after a child is exposed to and may be infected with SARS-CoV-2,” said Dr. Pichichero. “NIH has asked researchers to answer the following questions.”
- Why do children have milder illness?
- Are there differences in childhood biology (e.g., gender, puberty, etc.) that contribute to illness severity?
- Are there genetic host differences associated with different disease severity phenotypes, including MIS-C?
- Are there innate mucosal, humoral, cellular and other adaptive immune profiles that are associated with reduced or increased risk of progressive disease, including previous coronavirus infections?
- Will SARS-CoV-2 reinfection cause worse disease as seen with antibody-dependent enhancement (ADE) in other viral infections (e.g., dengue)? Will future vaccines carry a risk of the ADE phenomenon?
- Does substance use (e.g., nicotine, marijuana) exacerbate or trigger MIS-C through immune activation?
“We have no knowledge yet about SARS-CoV-2 vaccination of children, especially young children,” Dr. Pichichero emphasized. “There are different types of vaccines – messenger RNA, adenovirus vector and purified spike proteins of the virus – among others, but questions remain: Will the vaccines work in children? What about side effects? Will the antibodies and cellular immunity protect partially or completely?”
The researchers and editorialists had no financial conflicts to disclose. Dr. Pichichero had no financial conflicts to disclose.
SOURCE: Han MS et al. JAMA Pediatr. 2020 Aug 28. doi:10.1001/jamapediatrics.2020.3988.
About 22% of children with COVID-19 infections were asymptomatic, and 66% of the symptomatic children had unrecognized symptoms at the time of diagnosis, based on data from a case series of 91 confirmed cases.
Although recent reports suggest that COVID-19 infections in children are generally mild, data on the full spectrum of illness and duration of viral RNA in children are limited, wrote Mi Seon Han, MD, PhD, of Seoul (South Korea) Metropolitan Government–Seoul National University Boramae Medical Center, and colleagues.
To examine the full clinical course and duration of COVID-19 RNA detectability in children with confirmed infections, the researchers reviewed data from 91 individuals with confirmed infections. The children ranged in age from 27 days to 18 years, and 58% were male. The children were monitored at 20 hospitals and 2 isolation facilities for a mean 21.9 days. The findings were published in JAMA Pediatrics.
Overall, COVID-19 viral RNA was present in the study population for a mean 17.6 days, with testing done at a median interval of 3 days. A total of 20 children (22%) were asymptomatic throughout the study period. In these children, viral RNA was detected for a mean 14 days.
“The major hurdle implicated in this study in diagnosing and treating children with COVID-19 is that the researchers noted.
Of the 71 symptomatic children, 47 (66%) had unrecognized symptoms prior to diagnosis, 18 (25%) developed symptoms after diagnosis, and 6 (9%) were diagnosed at the time of symptom onset. The symptomatic children were symptomatic for a median of 11 days; 43 (61%) remained symptomatic at 7 days’ follow-up after the study period, 27 (38%) were symptomatic at 14 days, and 7 (10%) were symptomatic at 21 days.
A total of 41 children had upper respiratory infections (58%) and 22 children (24%) had lower respiratory tract infections. No difference in the duration of virus RNA was detected between children with upper respiratory tract infections and lower respiratory tract infections (average, 18.7 days vs. 19.9 days).
Among the symptomatic children, 46 (65%) had mild cases and 20 (28%) had moderate cases.
For treatment, 14 children (15%) received lopinavir-ritonavir and/or hydroxychloroquine. Two patients had severe illness and received oxygen via nasal prong, without the need for mechanical ventilation. All the children in the case series recovered from their infections with no fatalities.
The study’s main limitation was the inability to analyze the transmission potential of the children because of the quarantine and isolation policies in Korea, the researchers noted. In addition, the researchers did not perform follow-up testing at consistent intervals, so the duration of COVID-19 RNA detection may be inexact.
However, the results suggest “that suspecting and diagnosing COVID-19 in children based on their symptoms without epidemiologic information and virus testing is very challenging,” the researchers emphasized.
“Most of the children with COVID-19 have silent disease, but SARS-CoV-2 RNA can still be detected in the respiratory tract for a prolonged period,” they wrote. More research is needed to explore the potential for disease transmission by children in the community, and increased surveillance with laboratory screening can help identify children with unrecognized infections.
The study is the first known to focus on the frequency of asymptomatic infection in children and the duration of symptoms in both asymptomatic and symptomatic children, Roberta L. DeBiasi, MD, and Meghan Delaney, DO, both affiliated with Children’s National Hospital and Research Institute, Washington, and George Washington University, Washington, wrote in an accompanying editorial. The structure of the Korean public health system “allowed for the sequential observation, testing (median testing interval of every 3 days), and comparison of 91 asymptomatic, presymptomatic, and symptomatic children with mild to moderate upper and lower respiratory tract infection, identified primarily by contact tracing from laboratory-proven cases.”
Two take-home points from the study are that not all infected children are symptomatic, and the duration of symptoms in those who are varies widely, they noted. “Interestingly, this study aligns with adult data in which up to 40% of adults may remain asymptomatic in the face of infection.”
However, “The third and most important take-home point from this study relates to the duration of viral shedding in infected pediatric patients,” Dr. DeBiasi and Dr. Delaney said (JAMA Pediatr. 2020 Aug 28. doi: 10.1001/jamapediatrics.2020.3996).
“Fully half of symptomatic children with both upper and lower tract disease were still shedding virus at 21 days. These are striking data, particularly since 86 of 88 diagnosed children (98%) either had no symptoms or mild or moderate disease,” they explained. The results highlight the need for improvements in qualitative molecular testing and formal studies to identify differences in results from different testing scenarios, such as hospital entry, preprocedure screening, and symptomatic testing. In addition, “these findings are highly relevant to the development of public health strategies to mitigate and contain spread within communities, particularly as affected communities begin their recovery phases.”
The study is important because “schools are opening, and we don’t know what is going to happen,” Michael E. Pichichero, MD, of Rochester General Hospital, N.Y., said in an interview.
“Clinicians, parents, students, school administrators and politicians are worried,” he said. “This study adds to others recently published, bringing into focus the challenges to several suppositions that existed when the COVID-19 pandemic began and over the summer.”
“This study of 91 Korean children tells us that taking a child’s temperature as a screening tool to decide if they may enter school will not be a highly successful strategy,” he said. “Many children are without fever and asymptomatic when infected and contagious. The notion that children shed less virus or shed it for shorter lengths of time we keep learning from this type of research is not true. In another recent study the authors found that children shed as much of the SARS-CoV-2 virus as an adult in the ICU on a ventilator.”
Dr. Pichichero said he was not surprised by the study findings. “A similar paper was published last week in the Journal of Pediatrics from Massachusetts General Hospital, so the findings in the JAMA paper are similar to what has been reported in the United States.”
“Availability of testing will continue to be a challenge in some communities,” said Dr. Pichichero. “Here in the Rochester, New York, area we will use a screening questionnaire based on the CDC [Centers for Disease Control and Prevention] symptom criteria of SARS-CoV-2 infections to decide whom to test.”
As for additional research, “We have so much more to learn about SARS-CoV-2 in children,” he emphasized. “The focus has been on adults because the morbidity and mortality has been greatest in adults, especially the elderly and those with compromised health.”
“The National Institutes of Health has issued a call for more research in children to characterize the spectrum of SARS-CoV-2 illness, including the multisystem inflammatory syndrome in children [MIS-C] and try to identify biomarkers and/or biosignatures for a prognostic algorithm to predict the longitudinal risk of disease severity after a child is exposed to and may be infected with SARS-CoV-2,” said Dr. Pichichero. “NIH has asked researchers to answer the following questions.”
- Why do children have milder illness?
- Are there differences in childhood biology (e.g., gender, puberty, etc.) that contribute to illness severity?
- Are there genetic host differences associated with different disease severity phenotypes, including MIS-C?
- Are there innate mucosal, humoral, cellular and other adaptive immune profiles that are associated with reduced or increased risk of progressive disease, including previous coronavirus infections?
- Will SARS-CoV-2 reinfection cause worse disease as seen with antibody-dependent enhancement (ADE) in other viral infections (e.g., dengue)? Will future vaccines carry a risk of the ADE phenomenon?
- Does substance use (e.g., nicotine, marijuana) exacerbate or trigger MIS-C through immune activation?
“We have no knowledge yet about SARS-CoV-2 vaccination of children, especially young children,” Dr. Pichichero emphasized. “There are different types of vaccines – messenger RNA, adenovirus vector and purified spike proteins of the virus – among others, but questions remain: Will the vaccines work in children? What about side effects? Will the antibodies and cellular immunity protect partially or completely?”
The researchers and editorialists had no financial conflicts to disclose. Dr. Pichichero had no financial conflicts to disclose.
SOURCE: Han MS et al. JAMA Pediatr. 2020 Aug 28. doi:10.1001/jamapediatrics.2020.3988.
About 22% of children with COVID-19 infections were asymptomatic, and 66% of the symptomatic children had unrecognized symptoms at the time of diagnosis, based on data from a case series of 91 confirmed cases.
Although recent reports suggest that COVID-19 infections in children are generally mild, data on the full spectrum of illness and duration of viral RNA in children are limited, wrote Mi Seon Han, MD, PhD, of Seoul (South Korea) Metropolitan Government–Seoul National University Boramae Medical Center, and colleagues.
To examine the full clinical course and duration of COVID-19 RNA detectability in children with confirmed infections, the researchers reviewed data from 91 individuals with confirmed infections. The children ranged in age from 27 days to 18 years, and 58% were male. The children were monitored at 20 hospitals and 2 isolation facilities for a mean 21.9 days. The findings were published in JAMA Pediatrics.
Overall, COVID-19 viral RNA was present in the study population for a mean 17.6 days, with testing done at a median interval of 3 days. A total of 20 children (22%) were asymptomatic throughout the study period. In these children, viral RNA was detected for a mean 14 days.
“The major hurdle implicated in this study in diagnosing and treating children with COVID-19 is that the researchers noted.
Of the 71 symptomatic children, 47 (66%) had unrecognized symptoms prior to diagnosis, 18 (25%) developed symptoms after diagnosis, and 6 (9%) were diagnosed at the time of symptom onset. The symptomatic children were symptomatic for a median of 11 days; 43 (61%) remained symptomatic at 7 days’ follow-up after the study period, 27 (38%) were symptomatic at 14 days, and 7 (10%) were symptomatic at 21 days.
A total of 41 children had upper respiratory infections (58%) and 22 children (24%) had lower respiratory tract infections. No difference in the duration of virus RNA was detected between children with upper respiratory tract infections and lower respiratory tract infections (average, 18.7 days vs. 19.9 days).
Among the symptomatic children, 46 (65%) had mild cases and 20 (28%) had moderate cases.
For treatment, 14 children (15%) received lopinavir-ritonavir and/or hydroxychloroquine. Two patients had severe illness and received oxygen via nasal prong, without the need for mechanical ventilation. All the children in the case series recovered from their infections with no fatalities.
The study’s main limitation was the inability to analyze the transmission potential of the children because of the quarantine and isolation policies in Korea, the researchers noted. In addition, the researchers did not perform follow-up testing at consistent intervals, so the duration of COVID-19 RNA detection may be inexact.
However, the results suggest “that suspecting and diagnosing COVID-19 in children based on their symptoms without epidemiologic information and virus testing is very challenging,” the researchers emphasized.
“Most of the children with COVID-19 have silent disease, but SARS-CoV-2 RNA can still be detected in the respiratory tract for a prolonged period,” they wrote. More research is needed to explore the potential for disease transmission by children in the community, and increased surveillance with laboratory screening can help identify children with unrecognized infections.
The study is the first known to focus on the frequency of asymptomatic infection in children and the duration of symptoms in both asymptomatic and symptomatic children, Roberta L. DeBiasi, MD, and Meghan Delaney, DO, both affiliated with Children’s National Hospital and Research Institute, Washington, and George Washington University, Washington, wrote in an accompanying editorial. The structure of the Korean public health system “allowed for the sequential observation, testing (median testing interval of every 3 days), and comparison of 91 asymptomatic, presymptomatic, and symptomatic children with mild to moderate upper and lower respiratory tract infection, identified primarily by contact tracing from laboratory-proven cases.”
Two take-home points from the study are that not all infected children are symptomatic, and the duration of symptoms in those who are varies widely, they noted. “Interestingly, this study aligns with adult data in which up to 40% of adults may remain asymptomatic in the face of infection.”
However, “The third and most important take-home point from this study relates to the duration of viral shedding in infected pediatric patients,” Dr. DeBiasi and Dr. Delaney said (JAMA Pediatr. 2020 Aug 28. doi: 10.1001/jamapediatrics.2020.3996).
“Fully half of symptomatic children with both upper and lower tract disease were still shedding virus at 21 days. These are striking data, particularly since 86 of 88 diagnosed children (98%) either had no symptoms or mild or moderate disease,” they explained. The results highlight the need for improvements in qualitative molecular testing and formal studies to identify differences in results from different testing scenarios, such as hospital entry, preprocedure screening, and symptomatic testing. In addition, “these findings are highly relevant to the development of public health strategies to mitigate and contain spread within communities, particularly as affected communities begin their recovery phases.”
The study is important because “schools are opening, and we don’t know what is going to happen,” Michael E. Pichichero, MD, of Rochester General Hospital, N.Y., said in an interview.
“Clinicians, parents, students, school administrators and politicians are worried,” he said. “This study adds to others recently published, bringing into focus the challenges to several suppositions that existed when the COVID-19 pandemic began and over the summer.”
“This study of 91 Korean children tells us that taking a child’s temperature as a screening tool to decide if they may enter school will not be a highly successful strategy,” he said. “Many children are without fever and asymptomatic when infected and contagious. The notion that children shed less virus or shed it for shorter lengths of time we keep learning from this type of research is not true. In another recent study the authors found that children shed as much of the SARS-CoV-2 virus as an adult in the ICU on a ventilator.”
Dr. Pichichero said he was not surprised by the study findings. “A similar paper was published last week in the Journal of Pediatrics from Massachusetts General Hospital, so the findings in the JAMA paper are similar to what has been reported in the United States.”
“Availability of testing will continue to be a challenge in some communities,” said Dr. Pichichero. “Here in the Rochester, New York, area we will use a screening questionnaire based on the CDC [Centers for Disease Control and Prevention] symptom criteria of SARS-CoV-2 infections to decide whom to test.”
As for additional research, “We have so much more to learn about SARS-CoV-2 in children,” he emphasized. “The focus has been on adults because the morbidity and mortality has been greatest in adults, especially the elderly and those with compromised health.”
“The National Institutes of Health has issued a call for more research in children to characterize the spectrum of SARS-CoV-2 illness, including the multisystem inflammatory syndrome in children [MIS-C] and try to identify biomarkers and/or biosignatures for a prognostic algorithm to predict the longitudinal risk of disease severity after a child is exposed to and may be infected with SARS-CoV-2,” said Dr. Pichichero. “NIH has asked researchers to answer the following questions.”
- Why do children have milder illness?
- Are there differences in childhood biology (e.g., gender, puberty, etc.) that contribute to illness severity?
- Are there genetic host differences associated with different disease severity phenotypes, including MIS-C?
- Are there innate mucosal, humoral, cellular and other adaptive immune profiles that are associated with reduced or increased risk of progressive disease, including previous coronavirus infections?
- Will SARS-CoV-2 reinfection cause worse disease as seen with antibody-dependent enhancement (ADE) in other viral infections (e.g., dengue)? Will future vaccines carry a risk of the ADE phenomenon?
- Does substance use (e.g., nicotine, marijuana) exacerbate or trigger MIS-C through immune activation?
“We have no knowledge yet about SARS-CoV-2 vaccination of children, especially young children,” Dr. Pichichero emphasized. “There are different types of vaccines – messenger RNA, adenovirus vector and purified spike proteins of the virus – among others, but questions remain: Will the vaccines work in children? What about side effects? Will the antibodies and cellular immunity protect partially or completely?”
The researchers and editorialists had no financial conflicts to disclose. Dr. Pichichero had no financial conflicts to disclose.
SOURCE: Han MS et al. JAMA Pediatr. 2020 Aug 28. doi:10.1001/jamapediatrics.2020.3988.
FROM JAMA PEDIATRICS
Sleep disorders may be undetected precursors for cardiometabolic disease in U.S. Latinos
Sleep disorders may be silent precursors of cardiometabolic disease among U.S. Latinos, said authors of a newly published study.
Xiaoyu Li, ScD, and Susan Redline, MD, MPH, of Harvard Medical School and Brigham and Women’s Hospital, Boston, and coauthors conducted a study of people who self-identified as Latino, who had baseline sleeping disorders, and who developed hypertension and diabetes over time. The study was published in the American Journal of Respiratory and Critical Care Medicine.
The findings suggested that sleep disorders preceded the development of hypertension and diabetes. Examining records from a major multiyear federal project, the Hispanic Community Health Study/Study of Latinos, Dr. Li, Dr. Redline, and coauthors found sleep-disordered breathing (SDB) was associated with a 1.54 higher adjusted odds of incident hypertension (95% confidence interval [CI], 1.18-2.00) and 1.33 higher odds of incident diabetes (95% CI, 1.05-1.67), compared with no SDB. Insomnia was associated with incident hypertension (odds ratio, 1.37; 95% CI, 1.11-1.69), but not diabetes. The association between insomnia and incident hypertension was stronger among men than women, they reported.
“We now need large-scale rigorous trials to evaluate the impact of early treatment of sleep disordered breathing and insomnia on preventing the development of hypertension and diabetes,” Dr. Redline said in an interview. “Clinicians should consider screening their patients at risk for hypertension and diabetes for both sleep-disordered breathing and insomnia.”
Implications for public health strategies
The study results may have implications for health strategies and policies aimed at addressing health differentials among ethnic and economic groups in the United States.
Suboptimal sleep health may be an important fundamental but understudied contributor to health disparities, Chandra L. Jackson, PhD, MS, of the National Institute of Environmental Health Sciences, Research Triangle, N.C., said in an interview. Dr. Jackson is the lead author for a report published in August on a 2018 National Institutes of Health workshop regarding the importance of studying sleep health disparities (Sleep 2020 Mar 10. doi: 10.1093/sleep/zsaa037). The NIH workshop emphasized how little research has been done on the prevalence, incidence, morbidity, or mortality of sleep deficiencies of racial and ethnic minority populations, even though members of these groups are generally more likely to experience sleep disorders. The report urged “a nuanced integration between health disparity causal pathways and sleep and circadian-related mechanisms” tailored for these groups, with attention paid to sociocultural context.
Dr. Jackson said the study by Dr. Li and colleagues fits nicely with the strategies recommended in this report. She added: “Prospective design is particularly important for establishing temporality or that the SDB and insomnia symptoms occurred before the outcomes of hypertension and diabetes.”
In commenting on the Xi/Redline paper, Krishna M. Sundar, MD, FCCP, medical director of the Sleep-Wake Center at the University of Utah, Salt Lake City, commended the study and noted that one of the challenges in sleep research is the long time period over which the effects of disordered breathing become clear, he said.
“Things don’t happen immediately. It takes months, years for the effects to develop,” Dr. Sundar said. “To try to piece together the relationships, you need very well planned studies.”
Study design: Participants and exclusions
Latinos currently make up 17.8%, or 57.5 million, of the U.S. population, and this group is expected to double within the next 4 decades, the investigators wrote. A few prior studies on the roles of sleep disorders in the cardiometabolic health of Latinos, though suggestive, were limited by cross-sectional designs, relatively small samples, and underrepresentation of various Latino heritage groups.
The investigators on this new study worked with data from the federal Hispanic Community Health Study/Study of Latinos (HCHS/SOL) in which more than 16,000 people participated.
This multiyear federal study drew people who self-identified with different heritage groups, including Cuban, Dominican, Mexican, Central American, South American, and Puerto Rican. Participants initially aged 18-74 years underwent a first round of exams and assessments between 2008 and 2011 to determine what risk factors they had at the start of the study. In the second phase, which took place from 2013 to 2018, participants had a second set of exams. The National Heart, Lung, and Blood Institute and the National Institute of Diabetes and Digestive and Kidney Diseases funded the HCHS/SOL.
The investigators initially had a potential data pool of 11,623 participants in the HCHS/SOL. About 1 of 8 in this group, or 1,424 participants (12.3%), did not undergo a sleep study or did not have sufficient sleep data for analyses. Another 93 (0.8%) participants were excluded for missing data on covariates.
For incident hypertension analyses, participants who had prevalent hypertension at the first screening in the HCHS/SOL (n = 3,139) or had missing data on hypertension (n = 2) were excluded. That resulted in an analytic sample of 6,965 for hypertension questions.
For incident diabetes analyses, participants who had prevalent diabetes at the first screening (n = 2,062) or had missing data on diabetes (n = 21) were excluded, yielding an analytic sample of 8,023.
Incident hypertension was defined as participants not having hypertension at baseline and having hypertension, defined as a systolic blood pressure 140 mm Hg or greater, diastolic blood pressure 90 mm Hg or greater, or receiving antihypertensive medication within 4 weeks, at the second round of screening.
Cardiometabolic disease definitions
The researchers did not discriminate between type 1 and type 2 diabetes. They used the American Diabetes Association definition as a fasting plasma glucose 126 mg/dL or greater, 2-hour, postload plasma glucose 200 mg/dL or greater, or hemoglobin A1c 6.5% or greater, with an additional criterion on self-reported use of antidiabetic medication within 4 weeks before the visit.
In line with previous research, the investigators controlled for potential confounders measured at baseline including sociodemographic factors, health behaviors, and adiposity, which are considered important risk factors for both sleep disorders and incident metabolic diseases. These factors include education level, age, gender, and body mass index and whether participants had ever been smokers or users of alcohol.
Study limitations
Limitations of the study include use of a home sleep apnea test device that did not allow evaluation of arousal or sleep architecture. The researchers said this may have led to an underestimation of disease severity both due to overestimation of sleep time and underrecognition of hypopneas unassociated with desaturation. In addition, prior research has suggested that minority populations might underreport sleep disturbances, possibly “due to social desirability (a tendency not to encode a negative event), stress, stereotype threat, acculturation, attitudes, etc.” The participants were recruited mostly from urban areas, and the results might not be generalized to rural populations. In addition, 41% of study participants were of Mexican heritage, compared with 63% of the Hispanic population being of Mexican heritage in the United States.
Another researcher in the field of health disparities, Julia Roncoroni, PhD, assistant professor of psychology at the University of Denver, also noted this slight underrepresentation of Hispanics of Mexican origin and an overrepresentation of urban individuals in the HCHS/SOL.
“However, using data from HCHS/SOL, which is the largest multicenter epidemiological study of cardiovascular risk factors and sleep traits in U.S. Hispanics/Latinx, allows researchers to answer a high-impact question that would otherwise be prohibitively expensive and time consuming,” wrote Dr. Roncoroni.
Conclusions
The study offers “the first prospective evidence on the associations between SDB and insomnia with incident hypertension and diabetes in US Hispanics/Latinos.” The investigators concluded: “Given the fact that sleep disorders are largely undiagnosed and undertreated, they might represent modifiable targets for disease prevention and reduction among US Hispanics/Latinos. Culturally informed interventions focusing on detecting and treating sleep disorders might improve cardiometabolic health among US Hispanics/Latinos.”
Dr. Redline was partially supported by NIH grant R35 HL135818. This study drew from the Hispanic Community Health Study/Study of Latinos, which has been supported by contracts from the National Heart, Lung, and Blood Institute.
SOURCE: Li X et al. Am J Respir Crit Care Med. 2020 Aug 6. doi: 10.1164/rccm.201912-2330OC.
Sleep disorders may be silent precursors of cardiometabolic disease among U.S. Latinos, said authors of a newly published study.
Xiaoyu Li, ScD, and Susan Redline, MD, MPH, of Harvard Medical School and Brigham and Women’s Hospital, Boston, and coauthors conducted a study of people who self-identified as Latino, who had baseline sleeping disorders, and who developed hypertension and diabetes over time. The study was published in the American Journal of Respiratory and Critical Care Medicine.
The findings suggested that sleep disorders preceded the development of hypertension and diabetes. Examining records from a major multiyear federal project, the Hispanic Community Health Study/Study of Latinos, Dr. Li, Dr. Redline, and coauthors found sleep-disordered breathing (SDB) was associated with a 1.54 higher adjusted odds of incident hypertension (95% confidence interval [CI], 1.18-2.00) and 1.33 higher odds of incident diabetes (95% CI, 1.05-1.67), compared with no SDB. Insomnia was associated with incident hypertension (odds ratio, 1.37; 95% CI, 1.11-1.69), but not diabetes. The association between insomnia and incident hypertension was stronger among men than women, they reported.
“We now need large-scale rigorous trials to evaluate the impact of early treatment of sleep disordered breathing and insomnia on preventing the development of hypertension and diabetes,” Dr. Redline said in an interview. “Clinicians should consider screening their patients at risk for hypertension and diabetes for both sleep-disordered breathing and insomnia.”
Implications for public health strategies
The study results may have implications for health strategies and policies aimed at addressing health differentials among ethnic and economic groups in the United States.
Suboptimal sleep health may be an important fundamental but understudied contributor to health disparities, Chandra L. Jackson, PhD, MS, of the National Institute of Environmental Health Sciences, Research Triangle, N.C., said in an interview. Dr. Jackson is the lead author for a report published in August on a 2018 National Institutes of Health workshop regarding the importance of studying sleep health disparities (Sleep 2020 Mar 10. doi: 10.1093/sleep/zsaa037). The NIH workshop emphasized how little research has been done on the prevalence, incidence, morbidity, or mortality of sleep deficiencies of racial and ethnic minority populations, even though members of these groups are generally more likely to experience sleep disorders. The report urged “a nuanced integration between health disparity causal pathways and sleep and circadian-related mechanisms” tailored for these groups, with attention paid to sociocultural context.
Dr. Jackson said the study by Dr. Li and colleagues fits nicely with the strategies recommended in this report. She added: “Prospective design is particularly important for establishing temporality or that the SDB and insomnia symptoms occurred before the outcomes of hypertension and diabetes.”
In commenting on the Xi/Redline paper, Krishna M. Sundar, MD, FCCP, medical director of the Sleep-Wake Center at the University of Utah, Salt Lake City, commended the study and noted that one of the challenges in sleep research is the long time period over which the effects of disordered breathing become clear, he said.
“Things don’t happen immediately. It takes months, years for the effects to develop,” Dr. Sundar said. “To try to piece together the relationships, you need very well planned studies.”
Study design: Participants and exclusions
Latinos currently make up 17.8%, or 57.5 million, of the U.S. population, and this group is expected to double within the next 4 decades, the investigators wrote. A few prior studies on the roles of sleep disorders in the cardiometabolic health of Latinos, though suggestive, were limited by cross-sectional designs, relatively small samples, and underrepresentation of various Latino heritage groups.
The investigators on this new study worked with data from the federal Hispanic Community Health Study/Study of Latinos (HCHS/SOL) in which more than 16,000 people participated.
This multiyear federal study drew people who self-identified with different heritage groups, including Cuban, Dominican, Mexican, Central American, South American, and Puerto Rican. Participants initially aged 18-74 years underwent a first round of exams and assessments between 2008 and 2011 to determine what risk factors they had at the start of the study. In the second phase, which took place from 2013 to 2018, participants had a second set of exams. The National Heart, Lung, and Blood Institute and the National Institute of Diabetes and Digestive and Kidney Diseases funded the HCHS/SOL.
The investigators initially had a potential data pool of 11,623 participants in the HCHS/SOL. About 1 of 8 in this group, or 1,424 participants (12.3%), did not undergo a sleep study or did not have sufficient sleep data for analyses. Another 93 (0.8%) participants were excluded for missing data on covariates.
For incident hypertension analyses, participants who had prevalent hypertension at the first screening in the HCHS/SOL (n = 3,139) or had missing data on hypertension (n = 2) were excluded. That resulted in an analytic sample of 6,965 for hypertension questions.
For incident diabetes analyses, participants who had prevalent diabetes at the first screening (n = 2,062) or had missing data on diabetes (n = 21) were excluded, yielding an analytic sample of 8,023.
Incident hypertension was defined as participants not having hypertension at baseline and having hypertension, defined as a systolic blood pressure 140 mm Hg or greater, diastolic blood pressure 90 mm Hg or greater, or receiving antihypertensive medication within 4 weeks, at the second round of screening.
Cardiometabolic disease definitions
The researchers did not discriminate between type 1 and type 2 diabetes. They used the American Diabetes Association definition as a fasting plasma glucose 126 mg/dL or greater, 2-hour, postload plasma glucose 200 mg/dL or greater, or hemoglobin A1c 6.5% or greater, with an additional criterion on self-reported use of antidiabetic medication within 4 weeks before the visit.
In line with previous research, the investigators controlled for potential confounders measured at baseline including sociodemographic factors, health behaviors, and adiposity, which are considered important risk factors for both sleep disorders and incident metabolic diseases. These factors include education level, age, gender, and body mass index and whether participants had ever been smokers or users of alcohol.
Study limitations
Limitations of the study include use of a home sleep apnea test device that did not allow evaluation of arousal or sleep architecture. The researchers said this may have led to an underestimation of disease severity both due to overestimation of sleep time and underrecognition of hypopneas unassociated with desaturation. In addition, prior research has suggested that minority populations might underreport sleep disturbances, possibly “due to social desirability (a tendency not to encode a negative event), stress, stereotype threat, acculturation, attitudes, etc.” The participants were recruited mostly from urban areas, and the results might not be generalized to rural populations. In addition, 41% of study participants were of Mexican heritage, compared with 63% of the Hispanic population being of Mexican heritage in the United States.
Another researcher in the field of health disparities, Julia Roncoroni, PhD, assistant professor of psychology at the University of Denver, also noted this slight underrepresentation of Hispanics of Mexican origin and an overrepresentation of urban individuals in the HCHS/SOL.
“However, using data from HCHS/SOL, which is the largest multicenter epidemiological study of cardiovascular risk factors and sleep traits in U.S. Hispanics/Latinx, allows researchers to answer a high-impact question that would otherwise be prohibitively expensive and time consuming,” wrote Dr. Roncoroni.
Conclusions
The study offers “the first prospective evidence on the associations between SDB and insomnia with incident hypertension and diabetes in US Hispanics/Latinos.” The investigators concluded: “Given the fact that sleep disorders are largely undiagnosed and undertreated, they might represent modifiable targets for disease prevention and reduction among US Hispanics/Latinos. Culturally informed interventions focusing on detecting and treating sleep disorders might improve cardiometabolic health among US Hispanics/Latinos.”
Dr. Redline was partially supported by NIH grant R35 HL135818. This study drew from the Hispanic Community Health Study/Study of Latinos, which has been supported by contracts from the National Heart, Lung, and Blood Institute.
SOURCE: Li X et al. Am J Respir Crit Care Med. 2020 Aug 6. doi: 10.1164/rccm.201912-2330OC.
Sleep disorders may be silent precursors of cardiometabolic disease among U.S. Latinos, said authors of a newly published study.
Xiaoyu Li, ScD, and Susan Redline, MD, MPH, of Harvard Medical School and Brigham and Women’s Hospital, Boston, and coauthors conducted a study of people who self-identified as Latino, who had baseline sleeping disorders, and who developed hypertension and diabetes over time. The study was published in the American Journal of Respiratory and Critical Care Medicine.
The findings suggested that sleep disorders preceded the development of hypertension and diabetes. Examining records from a major multiyear federal project, the Hispanic Community Health Study/Study of Latinos, Dr. Li, Dr. Redline, and coauthors found sleep-disordered breathing (SDB) was associated with a 1.54 higher adjusted odds of incident hypertension (95% confidence interval [CI], 1.18-2.00) and 1.33 higher odds of incident diabetes (95% CI, 1.05-1.67), compared with no SDB. Insomnia was associated with incident hypertension (odds ratio, 1.37; 95% CI, 1.11-1.69), but not diabetes. The association between insomnia and incident hypertension was stronger among men than women, they reported.
“We now need large-scale rigorous trials to evaluate the impact of early treatment of sleep disordered breathing and insomnia on preventing the development of hypertension and diabetes,” Dr. Redline said in an interview. “Clinicians should consider screening their patients at risk for hypertension and diabetes for both sleep-disordered breathing and insomnia.”
Implications for public health strategies
The study results may have implications for health strategies and policies aimed at addressing health differentials among ethnic and economic groups in the United States.
Suboptimal sleep health may be an important fundamental but understudied contributor to health disparities, Chandra L. Jackson, PhD, MS, of the National Institute of Environmental Health Sciences, Research Triangle, N.C., said in an interview. Dr. Jackson is the lead author for a report published in August on a 2018 National Institutes of Health workshop regarding the importance of studying sleep health disparities (Sleep 2020 Mar 10. doi: 10.1093/sleep/zsaa037). The NIH workshop emphasized how little research has been done on the prevalence, incidence, morbidity, or mortality of sleep deficiencies of racial and ethnic minority populations, even though members of these groups are generally more likely to experience sleep disorders. The report urged “a nuanced integration between health disparity causal pathways and sleep and circadian-related mechanisms” tailored for these groups, with attention paid to sociocultural context.
Dr. Jackson said the study by Dr. Li and colleagues fits nicely with the strategies recommended in this report. She added: “Prospective design is particularly important for establishing temporality or that the SDB and insomnia symptoms occurred before the outcomes of hypertension and diabetes.”
In commenting on the Xi/Redline paper, Krishna M. Sundar, MD, FCCP, medical director of the Sleep-Wake Center at the University of Utah, Salt Lake City, commended the study and noted that one of the challenges in sleep research is the long time period over which the effects of disordered breathing become clear, he said.
“Things don’t happen immediately. It takes months, years for the effects to develop,” Dr. Sundar said. “To try to piece together the relationships, you need very well planned studies.”
Study design: Participants and exclusions
Latinos currently make up 17.8%, or 57.5 million, of the U.S. population, and this group is expected to double within the next 4 decades, the investigators wrote. A few prior studies on the roles of sleep disorders in the cardiometabolic health of Latinos, though suggestive, were limited by cross-sectional designs, relatively small samples, and underrepresentation of various Latino heritage groups.
The investigators on this new study worked with data from the federal Hispanic Community Health Study/Study of Latinos (HCHS/SOL) in which more than 16,000 people participated.
This multiyear federal study drew people who self-identified with different heritage groups, including Cuban, Dominican, Mexican, Central American, South American, and Puerto Rican. Participants initially aged 18-74 years underwent a first round of exams and assessments between 2008 and 2011 to determine what risk factors they had at the start of the study. In the second phase, which took place from 2013 to 2018, participants had a second set of exams. The National Heart, Lung, and Blood Institute and the National Institute of Diabetes and Digestive and Kidney Diseases funded the HCHS/SOL.
The investigators initially had a potential data pool of 11,623 participants in the HCHS/SOL. About 1 of 8 in this group, or 1,424 participants (12.3%), did not undergo a sleep study or did not have sufficient sleep data for analyses. Another 93 (0.8%) participants were excluded for missing data on covariates.
For incident hypertension analyses, participants who had prevalent hypertension at the first screening in the HCHS/SOL (n = 3,139) or had missing data on hypertension (n = 2) were excluded. That resulted in an analytic sample of 6,965 for hypertension questions.
For incident diabetes analyses, participants who had prevalent diabetes at the first screening (n = 2,062) or had missing data on diabetes (n = 21) were excluded, yielding an analytic sample of 8,023.
Incident hypertension was defined as participants not having hypertension at baseline and having hypertension, defined as a systolic blood pressure 140 mm Hg or greater, diastolic blood pressure 90 mm Hg or greater, or receiving antihypertensive medication within 4 weeks, at the second round of screening.
Cardiometabolic disease definitions
The researchers did not discriminate between type 1 and type 2 diabetes. They used the American Diabetes Association definition as a fasting plasma glucose 126 mg/dL or greater, 2-hour, postload plasma glucose 200 mg/dL or greater, or hemoglobin A1c 6.5% or greater, with an additional criterion on self-reported use of antidiabetic medication within 4 weeks before the visit.
In line with previous research, the investigators controlled for potential confounders measured at baseline including sociodemographic factors, health behaviors, and adiposity, which are considered important risk factors for both sleep disorders and incident metabolic diseases. These factors include education level, age, gender, and body mass index and whether participants had ever been smokers or users of alcohol.
Study limitations
Limitations of the study include use of a home sleep apnea test device that did not allow evaluation of arousal or sleep architecture. The researchers said this may have led to an underestimation of disease severity both due to overestimation of sleep time and underrecognition of hypopneas unassociated with desaturation. In addition, prior research has suggested that minority populations might underreport sleep disturbances, possibly “due to social desirability (a tendency not to encode a negative event), stress, stereotype threat, acculturation, attitudes, etc.” The participants were recruited mostly from urban areas, and the results might not be generalized to rural populations. In addition, 41% of study participants were of Mexican heritage, compared with 63% of the Hispanic population being of Mexican heritage in the United States.
Another researcher in the field of health disparities, Julia Roncoroni, PhD, assistant professor of psychology at the University of Denver, also noted this slight underrepresentation of Hispanics of Mexican origin and an overrepresentation of urban individuals in the HCHS/SOL.
“However, using data from HCHS/SOL, which is the largest multicenter epidemiological study of cardiovascular risk factors and sleep traits in U.S. Hispanics/Latinx, allows researchers to answer a high-impact question that would otherwise be prohibitively expensive and time consuming,” wrote Dr. Roncoroni.
Conclusions
The study offers “the first prospective evidence on the associations between SDB and insomnia with incident hypertension and diabetes in US Hispanics/Latinos.” The investigators concluded: “Given the fact that sleep disorders are largely undiagnosed and undertreated, they might represent modifiable targets for disease prevention and reduction among US Hispanics/Latinos. Culturally informed interventions focusing on detecting and treating sleep disorders might improve cardiometabolic health among US Hispanics/Latinos.”
Dr. Redline was partially supported by NIH grant R35 HL135818. This study drew from the Hispanic Community Health Study/Study of Latinos, which has been supported by contracts from the National Heart, Lung, and Blood Institute.
SOURCE: Li X et al. Am J Respir Crit Care Med. 2020 Aug 6. doi: 10.1164/rccm.201912-2330OC.
FROM THE AMERICAN JOURNAL OF RESPIRATORY AND CRITICAL CARE MEDICINE