User login
U.S. flu activity increases slightly
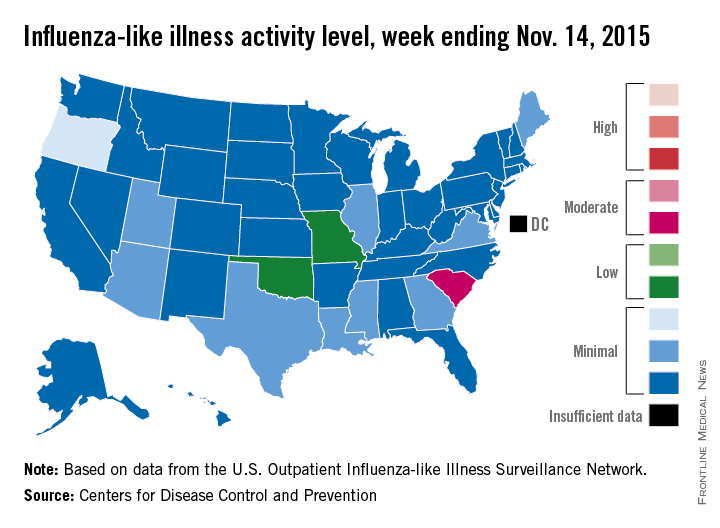
Activity of influenza-like illness (ILI) “increased slightly in the United States” during week 5 of the 2015-2016 influenza season, the Centers for Disease Control and Prevention reported Nov. 20.
Thirteen states were above level-1 activity as of Nov. 14, 2015, compared with seven the week before. South Carolina jumped all the way up to “moderate” activity (level 6) and Missouri and Oklahoma moved into the low-activity category (level 4). Oregon remained at a still-minimal level 3, while Arizona, Georgia, Illinois, Louisiana, Maine, Mississippi, Texas, Utah, and Virginia are at level 2, according to the CDC.
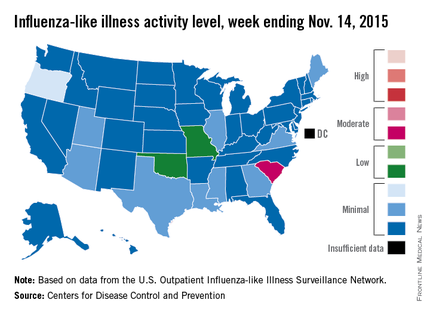
The first influenza-associated pediatric death was reported this week, although it actually occurred during week 4 (the week ending Nov. 7), the CDC said. There has been an average of 143 flu-associated pediatric deaths over the last three flu seasons.
ILI is defined as fever (temperature of 100° F or greater) and cough and/or sore throat. Activity level within a state is the proportion of outpatient visits to health care providers for influenza-like illness.
That proportion for the United States overall was 1.6%, which is up from last week’s 1.4% but still below the national baseline of 2.1%, the CDC said.
Activity of influenza-like illness (ILI) “increased slightly in the United States” during week 5 of the 2015-2016 influenza season, the Centers for Disease Control and Prevention reported Nov. 20.
Thirteen states were above level-1 activity as of Nov. 14, 2015, compared with seven the week before. South Carolina jumped all the way up to “moderate” activity (level 6) and Missouri and Oklahoma moved into the low-activity category (level 4). Oregon remained at a still-minimal level 3, while Arizona, Georgia, Illinois, Louisiana, Maine, Mississippi, Texas, Utah, and Virginia are at level 2, according to the CDC.

The first influenza-associated pediatric death was reported this week, although it actually occurred during week 4 (the week ending Nov. 7), the CDC said. There has been an average of 143 flu-associated pediatric deaths over the last three flu seasons.
ILI is defined as fever (temperature of 100° F or greater) and cough and/or sore throat. Activity level within a state is the proportion of outpatient visits to health care providers for influenza-like illness.
That proportion for the United States overall was 1.6%, which is up from last week’s 1.4% but still below the national baseline of 2.1%, the CDC said.
Activity of influenza-like illness (ILI) “increased slightly in the United States” during week 5 of the 2015-2016 influenza season, the Centers for Disease Control and Prevention reported Nov. 20.
Thirteen states were above level-1 activity as of Nov. 14, 2015, compared with seven the week before. South Carolina jumped all the way up to “moderate” activity (level 6) and Missouri and Oklahoma moved into the low-activity category (level 4). Oregon remained at a still-minimal level 3, while Arizona, Georgia, Illinois, Louisiana, Maine, Mississippi, Texas, Utah, and Virginia are at level 2, according to the CDC.

The first influenza-associated pediatric death was reported this week, although it actually occurred during week 4 (the week ending Nov. 7), the CDC said. There has been an average of 143 flu-associated pediatric deaths over the last three flu seasons.
ILI is defined as fever (temperature of 100° F or greater) and cough and/or sore throat. Activity level within a state is the proportion of outpatient visits to health care providers for influenza-like illness.
That proportion for the United States overall was 1.6%, which is up from last week’s 1.4% but still below the national baseline of 2.1%, the CDC said.
Malignant catatonia and aphasia follow multiple-drug overdose
CASE Improvement, then decline
Ms. M, age 37, is brought to the hospital after her husband found her at home, after an unknown duration of impaired consciousness. Her husband reports that Ms. M had normal cognitive functioning before this event, with no difficulty completing activities of daily living. Ms. M’s medical and psychiatric histories are notable for type 2 diabetes mellitus, unspecified bipolar disorder, and opioid, cocaine, and alcohol use disorders. Her medications include paroxetine, 40 mg/d, and gabapentin, 1,200 mg/d.
First admission. Poor inspiratory effort and oxygen saturation of 70% leads to emergent intubation. Serum laboratory studies reveal a white blood cell (WBC) count at 10,900/μL and creatinine phosphokinase level of 25,000 U/L. Urine drug screen is positive for tetrahydrocannabinol, cocaine, and opioids.
Ms. M is admitted to the ICU for management of rhabdomyolysis and multi-organ system failure, including acute hypoxic kidney injury.
By hospital Day 7, the tube is extubated with no recorded physical neurologic deficits. Mental status exam is normal, except for impaired memory of events surrounding the admission. Ms. M is discharged home with a recommendation for outpatient follow-up.
2 Weeks later. Ms. M is brought to the emergency department after a progressive decrease in social interaction, limited oral intake, decline in activities of daily living, and urinary incontinence. Results from laboratory studies are within normal limits; brain MRI is negative; EEG shows generalized moderate slowing.
During psychiatric evaluation, Ms. M is mute and staring continuously. Examination reveals oppositional paratonia (gegenhalten), catalepsy, prominent negativism, and waxy flexibility, all suggestive of catatonia. IV lorazepam is initiated at 1 mg every 8 hours, titrated to 2 mg, 3 times a day.
Ms. M is transferred to a psychiatric hospital for further treatment of catatonia.
Second admission. Evaluation with the Bush-Francis Catatonia Rating Scale supported a diagnosis of catatonia, with the presence of >3 features from the 14-item screen and a score of 16 on the 23-item rating scale.1 After titrating lorazepam to 9 mg/d with minimal therapeutic impact, the psychiatry team consults the electroconvulsive therapy (ECT) service, who deems Ms. M to be an appropriate candidate and petitions for court-ordered ECT.
On hospital Day 8, Ms. M has a fever of 104°F, tachycardia at 180 beats per minute, increased rigidity, and a WBC count of 17,800/μL. She is transferred to the ICU, with a presumptive diagnosis of malignant catatonia.
The medical evaluation, including general laboratory studies, EEG, and spinal fluid analysis, is unremarkable. Because of vital sign instability, 2 ECT treatments are completed in the general hospital before Ms. M resumes psychiatric inpatient care.
By the tenth ECT treatment, Ms. M is no longer febrile and experiences no further autonomic instability or psychomotor features of catatonia. Despite these improvements, she is noted to have persistent word-finding difficulty.
Which test would you order as the next step in your work up?
a) EEG
b) lumbar puncture
c) MRI
d) CT
The authors’ observations
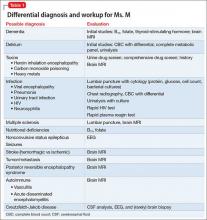
In approximately 25% of cases, catatonia is caused by a general medical condition2; as such, a comprehensive medical workup is vital for assessment and management of catatonic patients. In Ms. M’s case, we considered several medical causes, including nutritional deficiency, infection, a toxin, renal or hepatic impairment, hypothyroidism, seizure, and stroke. Evaluation included measurement of thyroid-stimulating hormone, vitamin B12, and folic acid levels; urinalysis and urine drug screen; chest radiography; lumbar puncture; neuroimaging; and EEG (Table 1).
Several conditions in the differential diagnosis were noteworthy. Ms. M’s severe and sudden neurologic decline, along with a positive urine drug screen for substances of abuse, raised concern about overdose leading to toxic encephalopathy or hypoxic brain injury. Ms. M’s oxygen saturation when she was found was moderately hypoxic at 70%, which is not a level associated with hypoxic brain damage.
We also considered posterior reversible encephalopathy syndrome (PRES), which presents variably with nausea, visual impairment, disturbance in consciousness, seizures, and focal neurologic signs.3 Although 67% to 80% of patients with PRES also have acute hypertension, blood pressure elevation is not necessary for the diagnosis.4 Similar to toxic leukoencephalopathy, PRES is diagnosed by brain MRI, with classic signs of posterior white-matter edema.
Case reports also describe an uncommon demyelinating syndrome, delayed post-hypoxic leukoencephalopathy (DPHL), which develops several weeks or months after a cerebral anoxic insult.5 In Ms. M’s case, brain MRI performed during her second medical hospitalization, 7 days after the initial neuropsychiatric decline, was unremarkable. Using this result to rule out DPHL would have been premature because pathognomonic abnormalities can appear as long as 40 days after the anoxic insult. Given our differential diagnosis, we ordered a repeat MRI.
Etiology and pathophysiology
First described in 1979, DPHL is rare, posing diagnostic challenges for clinical providers.6 Although the exact incidence of DPHL is unknown, the precipitating event typically involves cerebral anoxia, which can occur through carbon monoxide (CO) poisoning, strangulation, cardiac arrest, respiratory failure, and overdose from sedatives and narcotics (Table 2).7 DPHL was first observed in a small percentage (2.75%) of patients suffering from CO poisoning.8,9 Progression of the disease generally includes a period of unconsciousness, then a lucid interval that can last 2 to 40 days, followed by the abrupt onset of neuropsychiatric symptoms.10 The specific pathophysiologic mechanism is unknown, but has been hypothesized to involve inferior compensatory response to decreased oxygenation in the white matter.
Diagnosis and clinical features
DPHL can be divided into 2 clinical variations: parkinsonism and akinetic mutism. The former consists of conventional parkinsonian features along with agitation, apathy, hallucinations, dystonic posturing, and odd behaviors. The latter variant presents with apathy, minimal response to pain, functional bowel and bladder incontinence, mutism, and, at times, inappropriate laughter or tearfulness.5 Both variants share similar features with hypokinetic forms of catatonia.
DPHL is a diagnosis of exclusion. A careful history is critical to establish the possibility of a recent anoxic event. MRI findings, including hyperintensities in the cerebral white matter on T2-based sequencing, are suggestive of the disease. A choline peak on magnetic resonance spectroscopy also might be present in patients with DPHL, although it is not specific to the diagnosis.
Early reports of DPHL suggested an associated deficiency of arylsulfatase A, an enzyme required in the modulation of myelin; however, more recent case reports are conflicting.11 Familial mutations in the gene for arylsulfatase A also result in metachromatic leukodystrophy, and adult onset can present with psychiatric symptoms, including delusions and hallucinations.12
Treatment and prognosis
The treatment of DPHL consists primarily of supportive care and rehabilitation with physical, occupational, and speech therapy.11 With these measures, most patients improve after 3 to 6 months; however, a large percentage sustain some long-term cognitive deficit, the most prevalent symptom being frontal executive dysfunction.5
OUTCOME Supportive care
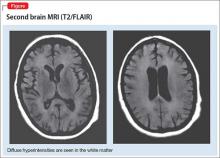
A second MRI shows diffuse hyperintensities in the white matter that spare the cerebellum and brainstem (Figure). This finding is pathognomonic for DPHL.
ECT is discontinued because there is no evidence to support ECT-associated improvement in DPHL. Moreover, ECT might worsen the clinical course through increased stress and metabolic demand on the brain.13
Because the primary treatment of DPHL is early rehabilitation, we consider that Ms. M would benefit most from increased supportive care and therapy. She is discharged to a brain injury rehabilitation facility, where metoprolol is prescribed for mild tachycardia, along with thiamine and vitamins B12 and D. Physical, occupational, and speech therapy are continued.
Approximately 3 weeks after admission to the rehabilitation program, Ms. M is discharged home. Although she improves in overall activities of daily living, she continues to experience moderate communication deficits and occasional external distractibility.
Bottom Line
Although delayed post-hypoxic leukoencephalopathy is considered rare, consider it in the differential diagnosis when a patient has a recent history of an anoxic event followed by the abrupt onset of neuropsychiatric symptoms. Keep in mind that the condition can be missed if an MRI is obtained too early, and the clinical signs can mimic hypokinetic catatonia.
Related Resources
• Meyer MA. Delayed post-hypoxic leukoencephalopathy: case report with a review of disease pathophysiology. Neurol Int. 2013;5(3):e13. doi: 10.4081/ni.2013.e13.
• Aljarallah S, Al-Hussain F. Acute fatal posthypoxic leukoencephalopathy following benzodiazepine overdose: a case report and review of the literature. BMC Neurol. 2015;15:69.
Drug Brand Names
Gabapentin • Neurontin
Lorazepam • Ativan
Metoprolol • Lopressor
Paroxetine • Paxil
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of com
1. Bush G, Fink M, Petrides G, et al. Catatonia. I. Rating scale and standardized examination. Acta Psychiatr Scand. 1996;93(2):129-136.
2. Azzam PN, Gopalan P. Prototypes of catatonia: diagnostic and therapeutic challenges in the general hospital. Psychosomatics. 2013;54(1):88-93.
3. Tormoehlen LM. Toxic leukoencephalopathies. Neurol Clin. 2011;29(3):591-605
4. Legriel S, Pico F, Azoulay E. Understanding posterior reversible encephalopathy syndrome. In: Vincent JL, ed. Annual update in intensive care and emergency medicine. Heidelberg, Germany: Springer Berlin Heidelberg; 2011:631-653.
5. Schprecher D, Mehta L. The syndrome of delayed post-hypoxic leukoencephalopathy. NeuroRehabilitation. 2010;26(1):65-72.
6. Wallace IR, Dynan C, Esmonde T. One confused patient, many confused physicians: a case of delayed post-hypoxic leucoencephalopathy. QJM. 2010;103(3):193-194.
7. Lou M, Jing CH, Selim MH, et al. Delayed substantia nigra damage and leukoencephalopathy after hypoxic-ischemic injury. J Neurol Sci. 2009;277(1-2):147-149.
8. Choi IS. Delayed neurologic sequelae in carbon monoxide intoxication. Arch Neurol. 1983;40(7):433-435.
9. Molloy S, Soh C, Williams TL. Reversible delayed posthypoxic leukoencephalopathy. AJNR Am J Neuroradiol. 2006;27(8):1763-1765.
10. Shprecher DR, Flanigan KM, Smith AG, et al. Clinical and diagnostic features of delayed hypoxic leukoencephalopathy. J Neuropsychiatry Clin Neurosci. 2008;20(4):473-477.
11. Lee BH, Lyketsos CG. Delayed post-hypoxic leukoencephalopathy. Psychosomatics. 2001;42(6):530-533.
12. Hyde TM, Ziegler JC, Weinberger DR. Psychiatric disturbances in metachromatic leukodystrophy. Insights into the neurobiology of psychosis. Arch Neurol. 1992;49(4):401-406.
13. Quinn DK, Abbott CC. Catatonia after cerebral hypoxia: do the usual treatments apply? Psychosomatics. 2014;55(6):525-535.
CASE Improvement, then decline
Ms. M, age 37, is brought to the hospital after her husband found her at home, after an unknown duration of impaired consciousness. Her husband reports that Ms. M had normal cognitive functioning before this event, with no difficulty completing activities of daily living. Ms. M’s medical and psychiatric histories are notable for type 2 diabetes mellitus, unspecified bipolar disorder, and opioid, cocaine, and alcohol use disorders. Her medications include paroxetine, 40 mg/d, and gabapentin, 1,200 mg/d.
First admission. Poor inspiratory effort and oxygen saturation of 70% leads to emergent intubation. Serum laboratory studies reveal a white blood cell (WBC) count at 10,900/μL and creatinine phosphokinase level of 25,000 U/L. Urine drug screen is positive for tetrahydrocannabinol, cocaine, and opioids.
Ms. M is admitted to the ICU for management of rhabdomyolysis and multi-organ system failure, including acute hypoxic kidney injury.
By hospital Day 7, the tube is extubated with no recorded physical neurologic deficits. Mental status exam is normal, except for impaired memory of events surrounding the admission. Ms. M is discharged home with a recommendation for outpatient follow-up.
2 Weeks later. Ms. M is brought to the emergency department after a progressive decrease in social interaction, limited oral intake, decline in activities of daily living, and urinary incontinence. Results from laboratory studies are within normal limits; brain MRI is negative; EEG shows generalized moderate slowing.
During psychiatric evaluation, Ms. M is mute and staring continuously. Examination reveals oppositional paratonia (gegenhalten), catalepsy, prominent negativism, and waxy flexibility, all suggestive of catatonia. IV lorazepam is initiated at 1 mg every 8 hours, titrated to 2 mg, 3 times a day.
Ms. M is transferred to a psychiatric hospital for further treatment of catatonia.
Second admission. Evaluation with the Bush-Francis Catatonia Rating Scale supported a diagnosis of catatonia, with the presence of >3 features from the 14-item screen and a score of 16 on the 23-item rating scale.1 After titrating lorazepam to 9 mg/d with minimal therapeutic impact, the psychiatry team consults the electroconvulsive therapy (ECT) service, who deems Ms. M to be an appropriate candidate and petitions for court-ordered ECT.
On hospital Day 8, Ms. M has a fever of 104°F, tachycardia at 180 beats per minute, increased rigidity, and a WBC count of 17,800/μL. She is transferred to the ICU, with a presumptive diagnosis of malignant catatonia.
The medical evaluation, including general laboratory studies, EEG, and spinal fluid analysis, is unremarkable. Because of vital sign instability, 2 ECT treatments are completed in the general hospital before Ms. M resumes psychiatric inpatient care.
By the tenth ECT treatment, Ms. M is no longer febrile and experiences no further autonomic instability or psychomotor features of catatonia. Despite these improvements, she is noted to have persistent word-finding difficulty.
Which test would you order as the next step in your work up?
a) EEG
b) lumbar puncture
c) MRI
d) CT
The authors’ observations
In approximately 25% of cases, catatonia is caused by a general medical condition2; as such, a comprehensive medical workup is vital for assessment and management of catatonic patients. In Ms. M’s case, we considered several medical causes, including nutritional deficiency, infection, a toxin, renal or hepatic impairment, hypothyroidism, seizure, and stroke. Evaluation included measurement of thyroid-stimulating hormone, vitamin B12, and folic acid levels; urinalysis and urine drug screen; chest radiography; lumbar puncture; neuroimaging; and EEG (Table 1).
Several conditions in the differential diagnosis were noteworthy. Ms. M’s severe and sudden neurologic decline, along with a positive urine drug screen for substances of abuse, raised concern about overdose leading to toxic encephalopathy or hypoxic brain injury. Ms. M’s oxygen saturation when she was found was moderately hypoxic at 70%, which is not a level associated with hypoxic brain damage.
We also considered posterior reversible encephalopathy syndrome (PRES), which presents variably with nausea, visual impairment, disturbance in consciousness, seizures, and focal neurologic signs.3 Although 67% to 80% of patients with PRES also have acute hypertension, blood pressure elevation is not necessary for the diagnosis.4 Similar to toxic leukoencephalopathy, PRES is diagnosed by brain MRI, with classic signs of posterior white-matter edema.
Case reports also describe an uncommon demyelinating syndrome, delayed post-hypoxic leukoencephalopathy (DPHL), which develops several weeks or months after a cerebral anoxic insult.5 In Ms. M’s case, brain MRI performed during her second medical hospitalization, 7 days after the initial neuropsychiatric decline, was unremarkable. Using this result to rule out DPHL would have been premature because pathognomonic abnormalities can appear as long as 40 days after the anoxic insult. Given our differential diagnosis, we ordered a repeat MRI.
Etiology and pathophysiology
First described in 1979, DPHL is rare, posing diagnostic challenges for clinical providers.6 Although the exact incidence of DPHL is unknown, the precipitating event typically involves cerebral anoxia, which can occur through carbon monoxide (CO) poisoning, strangulation, cardiac arrest, respiratory failure, and overdose from sedatives and narcotics (Table 2).7 DPHL was first observed in a small percentage (2.75%) of patients suffering from CO poisoning.8,9 Progression of the disease generally includes a period of unconsciousness, then a lucid interval that can last 2 to 40 days, followed by the abrupt onset of neuropsychiatric symptoms.10 The specific pathophysiologic mechanism is unknown, but has been hypothesized to involve inferior compensatory response to decreased oxygenation in the white matter.
Diagnosis and clinical features
DPHL can be divided into 2 clinical variations: parkinsonism and akinetic mutism. The former consists of conventional parkinsonian features along with agitation, apathy, hallucinations, dystonic posturing, and odd behaviors. The latter variant presents with apathy, minimal response to pain, functional bowel and bladder incontinence, mutism, and, at times, inappropriate laughter or tearfulness.5 Both variants share similar features with hypokinetic forms of catatonia.
DPHL is a diagnosis of exclusion. A careful history is critical to establish the possibility of a recent anoxic event. MRI findings, including hyperintensities in the cerebral white matter on T2-based sequencing, are suggestive of the disease. A choline peak on magnetic resonance spectroscopy also might be present in patients with DPHL, although it is not specific to the diagnosis.
Early reports of DPHL suggested an associated deficiency of arylsulfatase A, an enzyme required in the modulation of myelin; however, more recent case reports are conflicting.11 Familial mutations in the gene for arylsulfatase A also result in metachromatic leukodystrophy, and adult onset can present with psychiatric symptoms, including delusions and hallucinations.12
Treatment and prognosis
The treatment of DPHL consists primarily of supportive care and rehabilitation with physical, occupational, and speech therapy.11 With these measures, most patients improve after 3 to 6 months; however, a large percentage sustain some long-term cognitive deficit, the most prevalent symptom being frontal executive dysfunction.5
OUTCOME Supportive care
A second MRI shows diffuse hyperintensities in the white matter that spare the cerebellum and brainstem (Figure). This finding is pathognomonic for DPHL.
ECT is discontinued because there is no evidence to support ECT-associated improvement in DPHL. Moreover, ECT might worsen the clinical course through increased stress and metabolic demand on the brain.13
Because the primary treatment of DPHL is early rehabilitation, we consider that Ms. M would benefit most from increased supportive care and therapy. She is discharged to a brain injury rehabilitation facility, where metoprolol is prescribed for mild tachycardia, along with thiamine and vitamins B12 and D. Physical, occupational, and speech therapy are continued.
Approximately 3 weeks after admission to the rehabilitation program, Ms. M is discharged home. Although she improves in overall activities of daily living, she continues to experience moderate communication deficits and occasional external distractibility.
Bottom Line
Although delayed post-hypoxic leukoencephalopathy is considered rare, consider it in the differential diagnosis when a patient has a recent history of an anoxic event followed by the abrupt onset of neuropsychiatric symptoms. Keep in mind that the condition can be missed if an MRI is obtained too early, and the clinical signs can mimic hypokinetic catatonia.
Related Resources
• Meyer MA. Delayed post-hypoxic leukoencephalopathy: case report with a review of disease pathophysiology. Neurol Int. 2013;5(3):e13. doi: 10.4081/ni.2013.e13.
• Aljarallah S, Al-Hussain F. Acute fatal posthypoxic leukoencephalopathy following benzodiazepine overdose: a case report and review of the literature. BMC Neurol. 2015;15:69.
Drug Brand Names
Gabapentin • Neurontin
Lorazepam • Ativan
Metoprolol • Lopressor
Paroxetine • Paxil
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of com
CASE Improvement, then decline
Ms. M, age 37, is brought to the hospital after her husband found her at home, after an unknown duration of impaired consciousness. Her husband reports that Ms. M had normal cognitive functioning before this event, with no difficulty completing activities of daily living. Ms. M’s medical and psychiatric histories are notable for type 2 diabetes mellitus, unspecified bipolar disorder, and opioid, cocaine, and alcohol use disorders. Her medications include paroxetine, 40 mg/d, and gabapentin, 1,200 mg/d.
First admission. Poor inspiratory effort and oxygen saturation of 70% leads to emergent intubation. Serum laboratory studies reveal a white blood cell (WBC) count at 10,900/μL and creatinine phosphokinase level of 25,000 U/L. Urine drug screen is positive for tetrahydrocannabinol, cocaine, and opioids.
Ms. M is admitted to the ICU for management of rhabdomyolysis and multi-organ system failure, including acute hypoxic kidney injury.
By hospital Day 7, the tube is extubated with no recorded physical neurologic deficits. Mental status exam is normal, except for impaired memory of events surrounding the admission. Ms. M is discharged home with a recommendation for outpatient follow-up.
2 Weeks later. Ms. M is brought to the emergency department after a progressive decrease in social interaction, limited oral intake, decline in activities of daily living, and urinary incontinence. Results from laboratory studies are within normal limits; brain MRI is negative; EEG shows generalized moderate slowing.
During psychiatric evaluation, Ms. M is mute and staring continuously. Examination reveals oppositional paratonia (gegenhalten), catalepsy, prominent negativism, and waxy flexibility, all suggestive of catatonia. IV lorazepam is initiated at 1 mg every 8 hours, titrated to 2 mg, 3 times a day.
Ms. M is transferred to a psychiatric hospital for further treatment of catatonia.
Second admission. Evaluation with the Bush-Francis Catatonia Rating Scale supported a diagnosis of catatonia, with the presence of >3 features from the 14-item screen and a score of 16 on the 23-item rating scale.1 After titrating lorazepam to 9 mg/d with minimal therapeutic impact, the psychiatry team consults the electroconvulsive therapy (ECT) service, who deems Ms. M to be an appropriate candidate and petitions for court-ordered ECT.
On hospital Day 8, Ms. M has a fever of 104°F, tachycardia at 180 beats per minute, increased rigidity, and a WBC count of 17,800/μL. She is transferred to the ICU, with a presumptive diagnosis of malignant catatonia.
The medical evaluation, including general laboratory studies, EEG, and spinal fluid analysis, is unremarkable. Because of vital sign instability, 2 ECT treatments are completed in the general hospital before Ms. M resumes psychiatric inpatient care.
By the tenth ECT treatment, Ms. M is no longer febrile and experiences no further autonomic instability or psychomotor features of catatonia. Despite these improvements, she is noted to have persistent word-finding difficulty.
Which test would you order as the next step in your work up?
a) EEG
b) lumbar puncture
c) MRI
d) CT
The authors’ observations
In approximately 25% of cases, catatonia is caused by a general medical condition2; as such, a comprehensive medical workup is vital for assessment and management of catatonic patients. In Ms. M’s case, we considered several medical causes, including nutritional deficiency, infection, a toxin, renal or hepatic impairment, hypothyroidism, seizure, and stroke. Evaluation included measurement of thyroid-stimulating hormone, vitamin B12, and folic acid levels; urinalysis and urine drug screen; chest radiography; lumbar puncture; neuroimaging; and EEG (Table 1).
Several conditions in the differential diagnosis were noteworthy. Ms. M’s severe and sudden neurologic decline, along with a positive urine drug screen for substances of abuse, raised concern about overdose leading to toxic encephalopathy or hypoxic brain injury. Ms. M’s oxygen saturation when she was found was moderately hypoxic at 70%, which is not a level associated with hypoxic brain damage.
We also considered posterior reversible encephalopathy syndrome (PRES), which presents variably with nausea, visual impairment, disturbance in consciousness, seizures, and focal neurologic signs.3 Although 67% to 80% of patients with PRES also have acute hypertension, blood pressure elevation is not necessary for the diagnosis.4 Similar to toxic leukoencephalopathy, PRES is diagnosed by brain MRI, with classic signs of posterior white-matter edema.
Case reports also describe an uncommon demyelinating syndrome, delayed post-hypoxic leukoencephalopathy (DPHL), which develops several weeks or months after a cerebral anoxic insult.5 In Ms. M’s case, brain MRI performed during her second medical hospitalization, 7 days after the initial neuropsychiatric decline, was unremarkable. Using this result to rule out DPHL would have been premature because pathognomonic abnormalities can appear as long as 40 days after the anoxic insult. Given our differential diagnosis, we ordered a repeat MRI.
Etiology and pathophysiology
First described in 1979, DPHL is rare, posing diagnostic challenges for clinical providers.6 Although the exact incidence of DPHL is unknown, the precipitating event typically involves cerebral anoxia, which can occur through carbon monoxide (CO) poisoning, strangulation, cardiac arrest, respiratory failure, and overdose from sedatives and narcotics (Table 2).7 DPHL was first observed in a small percentage (2.75%) of patients suffering from CO poisoning.8,9 Progression of the disease generally includes a period of unconsciousness, then a lucid interval that can last 2 to 40 days, followed by the abrupt onset of neuropsychiatric symptoms.10 The specific pathophysiologic mechanism is unknown, but has been hypothesized to involve inferior compensatory response to decreased oxygenation in the white matter.
Diagnosis and clinical features
DPHL can be divided into 2 clinical variations: parkinsonism and akinetic mutism. The former consists of conventional parkinsonian features along with agitation, apathy, hallucinations, dystonic posturing, and odd behaviors. The latter variant presents with apathy, minimal response to pain, functional bowel and bladder incontinence, mutism, and, at times, inappropriate laughter or tearfulness.5 Both variants share similar features with hypokinetic forms of catatonia.
DPHL is a diagnosis of exclusion. A careful history is critical to establish the possibility of a recent anoxic event. MRI findings, including hyperintensities in the cerebral white matter on T2-based sequencing, are suggestive of the disease. A choline peak on magnetic resonance spectroscopy also might be present in patients with DPHL, although it is not specific to the diagnosis.
Early reports of DPHL suggested an associated deficiency of arylsulfatase A, an enzyme required in the modulation of myelin; however, more recent case reports are conflicting.11 Familial mutations in the gene for arylsulfatase A also result in metachromatic leukodystrophy, and adult onset can present with psychiatric symptoms, including delusions and hallucinations.12
Treatment and prognosis
The treatment of DPHL consists primarily of supportive care and rehabilitation with physical, occupational, and speech therapy.11 With these measures, most patients improve after 3 to 6 months; however, a large percentage sustain some long-term cognitive deficit, the most prevalent symptom being frontal executive dysfunction.5
OUTCOME Supportive care
A second MRI shows diffuse hyperintensities in the white matter that spare the cerebellum and brainstem (Figure). This finding is pathognomonic for DPHL.
ECT is discontinued because there is no evidence to support ECT-associated improvement in DPHL. Moreover, ECT might worsen the clinical course through increased stress and metabolic demand on the brain.13
Because the primary treatment of DPHL is early rehabilitation, we consider that Ms. M would benefit most from increased supportive care and therapy. She is discharged to a brain injury rehabilitation facility, where metoprolol is prescribed for mild tachycardia, along with thiamine and vitamins B12 and D. Physical, occupational, and speech therapy are continued.
Approximately 3 weeks after admission to the rehabilitation program, Ms. M is discharged home. Although she improves in overall activities of daily living, she continues to experience moderate communication deficits and occasional external distractibility.
Bottom Line
Although delayed post-hypoxic leukoencephalopathy is considered rare, consider it in the differential diagnosis when a patient has a recent history of an anoxic event followed by the abrupt onset of neuropsychiatric symptoms. Keep in mind that the condition can be missed if an MRI is obtained too early, and the clinical signs can mimic hypokinetic catatonia.
Related Resources
• Meyer MA. Delayed post-hypoxic leukoencephalopathy: case report with a review of disease pathophysiology. Neurol Int. 2013;5(3):e13. doi: 10.4081/ni.2013.e13.
• Aljarallah S, Al-Hussain F. Acute fatal posthypoxic leukoencephalopathy following benzodiazepine overdose: a case report and review of the literature. BMC Neurol. 2015;15:69.
Drug Brand Names
Gabapentin • Neurontin
Lorazepam • Ativan
Metoprolol • Lopressor
Paroxetine • Paxil
Disclosures
The authors report no financial relationships with any company whose products are mentioned in this article or with manufacturers of com
1. Bush G, Fink M, Petrides G, et al. Catatonia. I. Rating scale and standardized examination. Acta Psychiatr Scand. 1996;93(2):129-136.
2. Azzam PN, Gopalan P. Prototypes of catatonia: diagnostic and therapeutic challenges in the general hospital. Psychosomatics. 2013;54(1):88-93.
3. Tormoehlen LM. Toxic leukoencephalopathies. Neurol Clin. 2011;29(3):591-605
4. Legriel S, Pico F, Azoulay E. Understanding posterior reversible encephalopathy syndrome. In: Vincent JL, ed. Annual update in intensive care and emergency medicine. Heidelberg, Germany: Springer Berlin Heidelberg; 2011:631-653.
5. Schprecher D, Mehta L. The syndrome of delayed post-hypoxic leukoencephalopathy. NeuroRehabilitation. 2010;26(1):65-72.
6. Wallace IR, Dynan C, Esmonde T. One confused patient, many confused physicians: a case of delayed post-hypoxic leucoencephalopathy. QJM. 2010;103(3):193-194.
7. Lou M, Jing CH, Selim MH, et al. Delayed substantia nigra damage and leukoencephalopathy after hypoxic-ischemic injury. J Neurol Sci. 2009;277(1-2):147-149.
8. Choi IS. Delayed neurologic sequelae in carbon monoxide intoxication. Arch Neurol. 1983;40(7):433-435.
9. Molloy S, Soh C, Williams TL. Reversible delayed posthypoxic leukoencephalopathy. AJNR Am J Neuroradiol. 2006;27(8):1763-1765.
10. Shprecher DR, Flanigan KM, Smith AG, et al. Clinical and diagnostic features of delayed hypoxic leukoencephalopathy. J Neuropsychiatry Clin Neurosci. 2008;20(4):473-477.
11. Lee BH, Lyketsos CG. Delayed post-hypoxic leukoencephalopathy. Psychosomatics. 2001;42(6):530-533.
12. Hyde TM, Ziegler JC, Weinberger DR. Psychiatric disturbances in metachromatic leukodystrophy. Insights into the neurobiology of psychosis. Arch Neurol. 1992;49(4):401-406.
13. Quinn DK, Abbott CC. Catatonia after cerebral hypoxia: do the usual treatments apply? Psychosomatics. 2014;55(6):525-535.
1. Bush G, Fink M, Petrides G, et al. Catatonia. I. Rating scale and standardized examination. Acta Psychiatr Scand. 1996;93(2):129-136.
2. Azzam PN, Gopalan P. Prototypes of catatonia: diagnostic and therapeutic challenges in the general hospital. Psychosomatics. 2013;54(1):88-93.
3. Tormoehlen LM. Toxic leukoencephalopathies. Neurol Clin. 2011;29(3):591-605
4. Legriel S, Pico F, Azoulay E. Understanding posterior reversible encephalopathy syndrome. In: Vincent JL, ed. Annual update in intensive care and emergency medicine. Heidelberg, Germany: Springer Berlin Heidelberg; 2011:631-653.
5. Schprecher D, Mehta L. The syndrome of delayed post-hypoxic leukoencephalopathy. NeuroRehabilitation. 2010;26(1):65-72.
6. Wallace IR, Dynan C, Esmonde T. One confused patient, many confused physicians: a case of delayed post-hypoxic leucoencephalopathy. QJM. 2010;103(3):193-194.
7. Lou M, Jing CH, Selim MH, et al. Delayed substantia nigra damage and leukoencephalopathy after hypoxic-ischemic injury. J Neurol Sci. 2009;277(1-2):147-149.
8. Choi IS. Delayed neurologic sequelae in carbon monoxide intoxication. Arch Neurol. 1983;40(7):433-435.
9. Molloy S, Soh C, Williams TL. Reversible delayed posthypoxic leukoencephalopathy. AJNR Am J Neuroradiol. 2006;27(8):1763-1765.
10. Shprecher DR, Flanigan KM, Smith AG, et al. Clinical and diagnostic features of delayed hypoxic leukoencephalopathy. J Neuropsychiatry Clin Neurosci. 2008;20(4):473-477.
11. Lee BH, Lyketsos CG. Delayed post-hypoxic leukoencephalopathy. Psychosomatics. 2001;42(6):530-533.
12. Hyde TM, Ziegler JC, Weinberger DR. Psychiatric disturbances in metachromatic leukodystrophy. Insights into the neurobiology of psychosis. Arch Neurol. 1992;49(4):401-406.
13. Quinn DK, Abbott CC. Catatonia after cerebral hypoxia: do the usual treatments apply? Psychosomatics. 2014;55(6):525-535.
A decade after the CATIE study, the focus has shifted from effectiveness to neuroprotection
This past September, exactly 10 years after publication of the primary findings of the Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) study1—namely, that effectiveness (defined as all-cause discontinuation) was not different across first-generation antipsychotics (FGAs) and second generation antipsychotics (SGAs)— a new meta-analysis by Vita et al2 of differences in cortical gray-matter change between those 2 classes of antipsychotics offers a reminder: The clinical focus of the CATIE study overlooked important neurobiological and neuroprotective differences between FGAs and SGAs.
How drastically 1 decade can change the scientific perspective! Vita et al’s meta-analysis and meta-regression encompassed all 18 MRI studies of cortical gray matter in patients with schizophrenia.2 Earlier studies (published between 1983 and 2014) had lumped together patients who were receiving an FGA and those receiving an SGA, and authors reported overall reduction in cortical gray matter with prolonged antipsychotic treatment.
Remarkable findings emerge
When Vita et al2 analyzed FGA- and SGA-treated patients separately, however, they found a significant reduction in cortical gray matter in the FGA group but not in the SGA group. In fact, while higher daily dosages of FGAs were associated with greater reduction in cortical gray matter, higher dosages of SGAs were associated with lower cortical gray matter reduction and, in some samples, with an increase in volume of cortical gray matter.
The researchers hypothesized that the differential effects of FGAs and SGAs might be attributable to the neurotoxicity of typical FGAs and the neuroprotective effect of atypical SGAs.
Hindsight
The key neurobiological difference between FGAs and SGAs reported by Vita et al2 was not addressed in the CATIE study, leading, at that time, to a rush to judgment that all antipsychotics are the same. This conclusion emboldened managed-care organizations to mandate use of older (and cheaper) generic FGAs instead of newer (and more expensive) SGAs— most of which have become available as generic equivalents since the CATIE study was completed.
Investigators in the CATIE study— of which I was one—cannot be blamed for not focusing on neurotoxicity and neuroprotection; those data were not on the psychiatry’s radar when the CATIE study was designed in 1998. The major focus was on whether SGAs (new on the scene in the late 1990s) were more efficacious, safe, and tolerable (that is, more effective) than FGAs.
In fact, the first study reporting that SGAs stimulated neurogenesis (in animals) was published in 2002,3 when the CATIE study was more than half complete. Research into the neuroprotective properties of SGAs then grew rapidly. In fact, the principal investigator of the CATIE study conducted a head-to-head comparison of FGA haloperidol and SGA olanzapine in a sample of first-episode schizophrenia patients4; over 1 year of follow-up, it was determined that patients in the haloperidol-treated group exhibited significant brain volume loss on MRI but those in the olanzapine-treated group did not. This study was published in 2005—the same year the CATIE study was published!
SGAs offer neuroprotection
Over the past decade, the neuroprotective effects of SGAs5 and the neurotoxic effects of FGAs6 have been studied intensively, revealing that SGAs have multiple neuroprotective effects. These effects include:
• stimulation of the production of new brain cells (neurons and glia), known as neurogenesis5,7,8
• an increase in neurotrophic factors, such as nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF),9 which are found at a significantly low level in patients with psychosis10
• reversal of phencyclidine (PCP)-induced changes in gene expression11
• neuroprotection against ischemic stroke12-14
• reversal of PCP-induced loss of dendritic spines in the frontal cortex15
• prevention of oligodendrocyte damage caused by interferon gamma-stimulated microglia16,17
• reversal of loss of dendritic spines in the prefrontal cortex induced by dopamine depletion18
• an anti-inflammatory effect19,20
• protection against β-amyloid and hydrogen peroxide-induced cell death21
• protection against prefrontal cortical neuronal damage caused by dizocilpine (MK-801)22
• reversal of a PCP-induced decrease in the glutathione level and alteration of antioxidant defenses23
• protection of cortical neurons from glutamate neurotoxicity.24
One reason why SGAs are neuroprotective, but FGAs are not, can be attributed to their receptor profiles. FGAs block dopamine D2 receptors far more than serotonin 2A receptors, whereas SGAs do the opposite: They block 5-HT2A receptors 500% to 1,000% more than they block D2 receptors. This difference is associated in turn with a different neurobiological and neuroprotective profiles, such as a decrease or an increase in BDNF.25,26
Neither similar nor interchangeable
Since publication of the findings of the CATIE study, the primary investigator has proposed that neuroprotection can be a therapeutic strategy to prevent neurodegeneration and neurodeterioration associated with schizophrenia.27 Given the preponderance of data showing that SGAs have numerous neuroprotective properties but FGAs have many neurotoxic effects,6 the message to psychiatric practitioners, a decade after the CATIE study, is that the 2 generations of antipsychotic agents are not really similar or interchangeable. They might have similar clinical effectiveness but they exert very different neurobiological effects.
The proof of the pudding is in the eating: Despite the findings of the CATIE study, the vast majority of psychiatrists would prefer to treat their own family members with an SGA, not an FGA, if the need for antipsychotic medication arises.
1. Lieberman JA, Stroup TS, McEvoy JP, et al; Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) Investigators. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med. 2005;353(12):1209-1223.
2. Vita A, De Peri L, Deste G, et al. The effect of antipsychotic treatment on cortical gray matter changes in schizophrenia: does the class matter? A meta-analysis and meta-regression of longitudinal magnetic resonance imaging studies. Biol Psychiatry. 2015;78(6):403-412.
3. Wakade CG, Mahadik SP, Waller JL, et al. Atypical neuroleptics stimulate neurogenesis in adult rat brain. J Neurosci Res. 2002;69(1):72-79.
4. Lieberman JA, Tollefson GD, Charles C, et al; HGDH Study Group. Antipsychotic drug effects on brain morphology in first-episode psychosis. Arch Gen Psychiatry. 2005;62(4):361-370.
5. Nasrallah HA. Impaired neuroplasticity in schizophrenia and the neuro-regenerative effects of atypical antipsychotics. Medscape Psychiatry. http://www.medscape.org/viewarticle/569521. Published January 31, 2008. Accessed November 10, 2015.
6. Nasrallah HA. Haloperidol clearly is neurotoxic. Should it be banned? Current Psychiatry. 2012;12(7):7-8.
7. Nandra KS, Agius M. The differences between typical and atypical antipsychotics: the effects on neurogenesis. Psychiatr Danub. 2012;24(suppl 1):S95-S99.
8. Nasrallah HA, Hopkins T, Pixley SK, et al. Differential effects of antipsychotic and antidepressant drugs on neurogenic region in rats. Brain Res. 2010;354:23-29.
9. Pillai A, Tery AV, Mahadik SP. Differential effects of long-term treatment with typical and atypical antipsychotics on NGF and BNDF levels in rat striatum and hippocampus. Schizophr Res. 2006;82(1):95-106.
10. Buckley PF, Pillai A, Evans D, et al. Brain derived neurotropic factor in first-episode psychosis. Schizophr Res. 2007;91(1-3):1-5.
11. Martin MV, Mimics K, Nisenbaum LK, et al. Olanzapine reversed brain gene expression changes induced by phencyclidines treatment in non-human primates. Mol Neuropsychiatry. 2015;1(2):82-93.
12. Yan BC, Park JH, Ahn JH, et al. Neuroprotection of posttreatment with risperidone, an atypical antipsychotic drug, in rat and gerbil models of ischemic stroke and the maintenance of antioxidants in a gerbil model of ischemic stroke. J Neurosci Res. 2014;92(6):795-807.
13. Yulug B, Yildiz A, Güzel O, et al. Risperidone attenuates brain damage after focal cerebral ischemia in vivo. Brain Res Bull. 2006;69(6):656-659.
14. Yulug B, Yildiz A, Hüdaoglu O, et al. Olanzapine attenuates brain damage after focal cerebral ischemia in vivo. Brain Res Bull. 2006;71(1-3):296-300.
15. Elsworth JD, Morrow BA. Hajszan T, et al. Phencyclidine-induced loss of asymmetric spine synapses in rodent prefrontal cortex is reversed by acute and chronic treatment with olanzapine. Neuropsychopharmacology. 2001;36(10):2054-2061.
16. Seki Y, Kato TA, Monji A, et al. Pretreatment of aripiprazole and minocycline, but not haloperidol, suppresses oligodendrocyte damage from interferon-y-stimulated microglia in co-culture model. Schizophr Res. 2013;151(1-3):20-28.
17. Bian Q, Kato T, Monji A, et al. The effect of atypical antipsychotics, perospirone, ziprasidone and quetiapine on microglial activation induced by interferon-gamma. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(1):42-48.
18. Wang HD, Deutch AY. Dopamine depletion of the prefrontal cortex induces dendritic spine loss: reversal by atypical antipsychotic drug treatment. Neuropsychopharmacology. 2008;33(6):1276-1286.
19. Miller BJ, Buckley P, Seabolt W, et al. Meta-analysis of cytokine alternations in schizophrenia: clinical status and antipsychotic effects. Biol Psychiatry. 2011;70(7):663-671.
20. Nasrallah HA. Beyond dopamine: The ‘other’ effects of antipsychotics. Current Psychiatry. 2013;12(6):8-9.
21. Yang MC, Lung FW. Neuroprotection of paliperidone on SH-SY5Y cells against β-amyloid peptide(25-35), N-methyl-4-phenylpyridinium ion, and hydrogen peroxide-induced cell death. Psychopharmacology (Berl). 2011;217(3):397-410.
22. Peng L, Zhu D, Feng X, et al. Paliperidone protects prefrontal cortical neurons from damages caused by MK-801 via Akt1/GSK3β _signaling pathway. Schizophr Res. 2013;147(1):14-23.23.
Stojkovic´ T, Radonjic´ NV, Velimirovic´ M, et al. Risperidone reverses phencyclidine induced decrease in glutathione levels and alternations of antioxidant defense in rat brain. Prog Neuropsychopharmacol Biol Psychiatry. 2012;39(1):192-199.
24. Koprivica V, Regardie K, Wolff C, et al. Aripiprazole protects cortical neurons from glutamate toxicity. Eur J Pharmacol. 2011;651(1-3):73-76.
25. Vaidya VA, Marek GJ, Aghajanian GK, et al. 5-HT2A receptor-mediated regulation of brain-derived neurotrophic factor mRNA in the hippocampus and the neocortex. J Neurosci. 1997;17(8):2785-2795.
26. Meridith GE, Switzer RC 3rd, Napier TC. Short-term, D2 receptor blockade induces synaptic degeneration, reduces levels of tyrosine hydroxylase and brain-derived neurotrophic factor, and enhances D2-mediated firing in the ventral pallidum. Brain Res. 2004;995(1):14-22.
27. Lieberman JA, Perkins DO, Jarskog LF. Neuroprotection: a therapeutic strategy to prevent deterioration associated with schizophrenia. CNS Spectr. 2007;12(suppl 4):1-13; quiz 14.
This past September, exactly 10 years after publication of the primary findings of the Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) study1—namely, that effectiveness (defined as all-cause discontinuation) was not different across first-generation antipsychotics (FGAs) and second generation antipsychotics (SGAs)— a new meta-analysis by Vita et al2 of differences in cortical gray-matter change between those 2 classes of antipsychotics offers a reminder: The clinical focus of the CATIE study overlooked important neurobiological and neuroprotective differences between FGAs and SGAs.
How drastically 1 decade can change the scientific perspective! Vita et al’s meta-analysis and meta-regression encompassed all 18 MRI studies of cortical gray matter in patients with schizophrenia.2 Earlier studies (published between 1983 and 2014) had lumped together patients who were receiving an FGA and those receiving an SGA, and authors reported overall reduction in cortical gray matter with prolonged antipsychotic treatment.
Remarkable findings emerge
When Vita et al2 analyzed FGA- and SGA-treated patients separately, however, they found a significant reduction in cortical gray matter in the FGA group but not in the SGA group. In fact, while higher daily dosages of FGAs were associated with greater reduction in cortical gray matter, higher dosages of SGAs were associated with lower cortical gray matter reduction and, in some samples, with an increase in volume of cortical gray matter.
The researchers hypothesized that the differential effects of FGAs and SGAs might be attributable to the neurotoxicity of typical FGAs and the neuroprotective effect of atypical SGAs.
Hindsight
The key neurobiological difference between FGAs and SGAs reported by Vita et al2 was not addressed in the CATIE study, leading, at that time, to a rush to judgment that all antipsychotics are the same. This conclusion emboldened managed-care organizations to mandate use of older (and cheaper) generic FGAs instead of newer (and more expensive) SGAs— most of which have become available as generic equivalents since the CATIE study was completed.
Investigators in the CATIE study— of which I was one—cannot be blamed for not focusing on neurotoxicity and neuroprotection; those data were not on the psychiatry’s radar when the CATIE study was designed in 1998. The major focus was on whether SGAs (new on the scene in the late 1990s) were more efficacious, safe, and tolerable (that is, more effective) than FGAs.
In fact, the first study reporting that SGAs stimulated neurogenesis (in animals) was published in 2002,3 when the CATIE study was more than half complete. Research into the neuroprotective properties of SGAs then grew rapidly. In fact, the principal investigator of the CATIE study conducted a head-to-head comparison of FGA haloperidol and SGA olanzapine in a sample of first-episode schizophrenia patients4; over 1 year of follow-up, it was determined that patients in the haloperidol-treated group exhibited significant brain volume loss on MRI but those in the olanzapine-treated group did not. This study was published in 2005—the same year the CATIE study was published!
SGAs offer neuroprotection
Over the past decade, the neuroprotective effects of SGAs5 and the neurotoxic effects of FGAs6 have been studied intensively, revealing that SGAs have multiple neuroprotective effects. These effects include:
• stimulation of the production of new brain cells (neurons and glia), known as neurogenesis5,7,8
• an increase in neurotrophic factors, such as nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF),9 which are found at a significantly low level in patients with psychosis10
• reversal of phencyclidine (PCP)-induced changes in gene expression11
• neuroprotection against ischemic stroke12-14
• reversal of PCP-induced loss of dendritic spines in the frontal cortex15
• prevention of oligodendrocyte damage caused by interferon gamma-stimulated microglia16,17
• reversal of loss of dendritic spines in the prefrontal cortex induced by dopamine depletion18
• an anti-inflammatory effect19,20
• protection against β-amyloid and hydrogen peroxide-induced cell death21
• protection against prefrontal cortical neuronal damage caused by dizocilpine (MK-801)22
• reversal of a PCP-induced decrease in the glutathione level and alteration of antioxidant defenses23
• protection of cortical neurons from glutamate neurotoxicity.24
One reason why SGAs are neuroprotective, but FGAs are not, can be attributed to their receptor profiles. FGAs block dopamine D2 receptors far more than serotonin 2A receptors, whereas SGAs do the opposite: They block 5-HT2A receptors 500% to 1,000% more than they block D2 receptors. This difference is associated in turn with a different neurobiological and neuroprotective profiles, such as a decrease or an increase in BDNF.25,26
Neither similar nor interchangeable
Since publication of the findings of the CATIE study, the primary investigator has proposed that neuroprotection can be a therapeutic strategy to prevent neurodegeneration and neurodeterioration associated with schizophrenia.27 Given the preponderance of data showing that SGAs have numerous neuroprotective properties but FGAs have many neurotoxic effects,6 the message to psychiatric practitioners, a decade after the CATIE study, is that the 2 generations of antipsychotic agents are not really similar or interchangeable. They might have similar clinical effectiveness but they exert very different neurobiological effects.
The proof of the pudding is in the eating: Despite the findings of the CATIE study, the vast majority of psychiatrists would prefer to treat their own family members with an SGA, not an FGA, if the need for antipsychotic medication arises.
This past September, exactly 10 years after publication of the primary findings of the Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) study1—namely, that effectiveness (defined as all-cause discontinuation) was not different across first-generation antipsychotics (FGAs) and second generation antipsychotics (SGAs)— a new meta-analysis by Vita et al2 of differences in cortical gray-matter change between those 2 classes of antipsychotics offers a reminder: The clinical focus of the CATIE study overlooked important neurobiological and neuroprotective differences between FGAs and SGAs.
How drastically 1 decade can change the scientific perspective! Vita et al’s meta-analysis and meta-regression encompassed all 18 MRI studies of cortical gray matter in patients with schizophrenia.2 Earlier studies (published between 1983 and 2014) had lumped together patients who were receiving an FGA and those receiving an SGA, and authors reported overall reduction in cortical gray matter with prolonged antipsychotic treatment.
Remarkable findings emerge
When Vita et al2 analyzed FGA- and SGA-treated patients separately, however, they found a significant reduction in cortical gray matter in the FGA group but not in the SGA group. In fact, while higher daily dosages of FGAs were associated with greater reduction in cortical gray matter, higher dosages of SGAs were associated with lower cortical gray matter reduction and, in some samples, with an increase in volume of cortical gray matter.
The researchers hypothesized that the differential effects of FGAs and SGAs might be attributable to the neurotoxicity of typical FGAs and the neuroprotective effect of atypical SGAs.
Hindsight
The key neurobiological difference between FGAs and SGAs reported by Vita et al2 was not addressed in the CATIE study, leading, at that time, to a rush to judgment that all antipsychotics are the same. This conclusion emboldened managed-care organizations to mandate use of older (and cheaper) generic FGAs instead of newer (and more expensive) SGAs— most of which have become available as generic equivalents since the CATIE study was completed.
Investigators in the CATIE study— of which I was one—cannot be blamed for not focusing on neurotoxicity and neuroprotection; those data were not on the psychiatry’s radar when the CATIE study was designed in 1998. The major focus was on whether SGAs (new on the scene in the late 1990s) were more efficacious, safe, and tolerable (that is, more effective) than FGAs.
In fact, the first study reporting that SGAs stimulated neurogenesis (in animals) was published in 2002,3 when the CATIE study was more than half complete. Research into the neuroprotective properties of SGAs then grew rapidly. In fact, the principal investigator of the CATIE study conducted a head-to-head comparison of FGA haloperidol and SGA olanzapine in a sample of first-episode schizophrenia patients4; over 1 year of follow-up, it was determined that patients in the haloperidol-treated group exhibited significant brain volume loss on MRI but those in the olanzapine-treated group did not. This study was published in 2005—the same year the CATIE study was published!
SGAs offer neuroprotection
Over the past decade, the neuroprotective effects of SGAs5 and the neurotoxic effects of FGAs6 have been studied intensively, revealing that SGAs have multiple neuroprotective effects. These effects include:
• stimulation of the production of new brain cells (neurons and glia), known as neurogenesis5,7,8
• an increase in neurotrophic factors, such as nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF),9 which are found at a significantly low level in patients with psychosis10
• reversal of phencyclidine (PCP)-induced changes in gene expression11
• neuroprotection against ischemic stroke12-14
• reversal of PCP-induced loss of dendritic spines in the frontal cortex15
• prevention of oligodendrocyte damage caused by interferon gamma-stimulated microglia16,17
• reversal of loss of dendritic spines in the prefrontal cortex induced by dopamine depletion18
• an anti-inflammatory effect19,20
• protection against β-amyloid and hydrogen peroxide-induced cell death21
• protection against prefrontal cortical neuronal damage caused by dizocilpine (MK-801)22
• reversal of a PCP-induced decrease in the glutathione level and alteration of antioxidant defenses23
• protection of cortical neurons from glutamate neurotoxicity.24
One reason why SGAs are neuroprotective, but FGAs are not, can be attributed to their receptor profiles. FGAs block dopamine D2 receptors far more than serotonin 2A receptors, whereas SGAs do the opposite: They block 5-HT2A receptors 500% to 1,000% more than they block D2 receptors. This difference is associated in turn with a different neurobiological and neuroprotective profiles, such as a decrease or an increase in BDNF.25,26
Neither similar nor interchangeable
Since publication of the findings of the CATIE study, the primary investigator has proposed that neuroprotection can be a therapeutic strategy to prevent neurodegeneration and neurodeterioration associated with schizophrenia.27 Given the preponderance of data showing that SGAs have numerous neuroprotective properties but FGAs have many neurotoxic effects,6 the message to psychiatric practitioners, a decade after the CATIE study, is that the 2 generations of antipsychotic agents are not really similar or interchangeable. They might have similar clinical effectiveness but they exert very different neurobiological effects.
The proof of the pudding is in the eating: Despite the findings of the CATIE study, the vast majority of psychiatrists would prefer to treat their own family members with an SGA, not an FGA, if the need for antipsychotic medication arises.
1. Lieberman JA, Stroup TS, McEvoy JP, et al; Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) Investigators. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med. 2005;353(12):1209-1223.
2. Vita A, De Peri L, Deste G, et al. The effect of antipsychotic treatment on cortical gray matter changes in schizophrenia: does the class matter? A meta-analysis and meta-regression of longitudinal magnetic resonance imaging studies. Biol Psychiatry. 2015;78(6):403-412.
3. Wakade CG, Mahadik SP, Waller JL, et al. Atypical neuroleptics stimulate neurogenesis in adult rat brain. J Neurosci Res. 2002;69(1):72-79.
4. Lieberman JA, Tollefson GD, Charles C, et al; HGDH Study Group. Antipsychotic drug effects on brain morphology in first-episode psychosis. Arch Gen Psychiatry. 2005;62(4):361-370.
5. Nasrallah HA. Impaired neuroplasticity in schizophrenia and the neuro-regenerative effects of atypical antipsychotics. Medscape Psychiatry. http://www.medscape.org/viewarticle/569521. Published January 31, 2008. Accessed November 10, 2015.
6. Nasrallah HA. Haloperidol clearly is neurotoxic. Should it be banned? Current Psychiatry. 2012;12(7):7-8.
7. Nandra KS, Agius M. The differences between typical and atypical antipsychotics: the effects on neurogenesis. Psychiatr Danub. 2012;24(suppl 1):S95-S99.
8. Nasrallah HA, Hopkins T, Pixley SK, et al. Differential effects of antipsychotic and antidepressant drugs on neurogenic region in rats. Brain Res. 2010;354:23-29.
9. Pillai A, Tery AV, Mahadik SP. Differential effects of long-term treatment with typical and atypical antipsychotics on NGF and BNDF levels in rat striatum and hippocampus. Schizophr Res. 2006;82(1):95-106.
10. Buckley PF, Pillai A, Evans D, et al. Brain derived neurotropic factor in first-episode psychosis. Schizophr Res. 2007;91(1-3):1-5.
11. Martin MV, Mimics K, Nisenbaum LK, et al. Olanzapine reversed brain gene expression changes induced by phencyclidines treatment in non-human primates. Mol Neuropsychiatry. 2015;1(2):82-93.
12. Yan BC, Park JH, Ahn JH, et al. Neuroprotection of posttreatment with risperidone, an atypical antipsychotic drug, in rat and gerbil models of ischemic stroke and the maintenance of antioxidants in a gerbil model of ischemic stroke. J Neurosci Res. 2014;92(6):795-807.
13. Yulug B, Yildiz A, Güzel O, et al. Risperidone attenuates brain damage after focal cerebral ischemia in vivo. Brain Res Bull. 2006;69(6):656-659.
14. Yulug B, Yildiz A, Hüdaoglu O, et al. Olanzapine attenuates brain damage after focal cerebral ischemia in vivo. Brain Res Bull. 2006;71(1-3):296-300.
15. Elsworth JD, Morrow BA. Hajszan T, et al. Phencyclidine-induced loss of asymmetric spine synapses in rodent prefrontal cortex is reversed by acute and chronic treatment with olanzapine. Neuropsychopharmacology. 2001;36(10):2054-2061.
16. Seki Y, Kato TA, Monji A, et al. Pretreatment of aripiprazole and minocycline, but not haloperidol, suppresses oligodendrocyte damage from interferon-y-stimulated microglia in co-culture model. Schizophr Res. 2013;151(1-3):20-28.
17. Bian Q, Kato T, Monji A, et al. The effect of atypical antipsychotics, perospirone, ziprasidone and quetiapine on microglial activation induced by interferon-gamma. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(1):42-48.
18. Wang HD, Deutch AY. Dopamine depletion of the prefrontal cortex induces dendritic spine loss: reversal by atypical antipsychotic drug treatment. Neuropsychopharmacology. 2008;33(6):1276-1286.
19. Miller BJ, Buckley P, Seabolt W, et al. Meta-analysis of cytokine alternations in schizophrenia: clinical status and antipsychotic effects. Biol Psychiatry. 2011;70(7):663-671.
20. Nasrallah HA. Beyond dopamine: The ‘other’ effects of antipsychotics. Current Psychiatry. 2013;12(6):8-9.
21. Yang MC, Lung FW. Neuroprotection of paliperidone on SH-SY5Y cells against β-amyloid peptide(25-35), N-methyl-4-phenylpyridinium ion, and hydrogen peroxide-induced cell death. Psychopharmacology (Berl). 2011;217(3):397-410.
22. Peng L, Zhu D, Feng X, et al. Paliperidone protects prefrontal cortical neurons from damages caused by MK-801 via Akt1/GSK3β _signaling pathway. Schizophr Res. 2013;147(1):14-23.23.
Stojkovic´ T, Radonjic´ NV, Velimirovic´ M, et al. Risperidone reverses phencyclidine induced decrease in glutathione levels and alternations of antioxidant defense in rat brain. Prog Neuropsychopharmacol Biol Psychiatry. 2012;39(1):192-199.
24. Koprivica V, Regardie K, Wolff C, et al. Aripiprazole protects cortical neurons from glutamate toxicity. Eur J Pharmacol. 2011;651(1-3):73-76.
25. Vaidya VA, Marek GJ, Aghajanian GK, et al. 5-HT2A receptor-mediated regulation of brain-derived neurotrophic factor mRNA in the hippocampus and the neocortex. J Neurosci. 1997;17(8):2785-2795.
26. Meridith GE, Switzer RC 3rd, Napier TC. Short-term, D2 receptor blockade induces synaptic degeneration, reduces levels of tyrosine hydroxylase and brain-derived neurotrophic factor, and enhances D2-mediated firing in the ventral pallidum. Brain Res. 2004;995(1):14-22.
27. Lieberman JA, Perkins DO, Jarskog LF. Neuroprotection: a therapeutic strategy to prevent deterioration associated with schizophrenia. CNS Spectr. 2007;12(suppl 4):1-13; quiz 14.
1. Lieberman JA, Stroup TS, McEvoy JP, et al; Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) Investigators. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med. 2005;353(12):1209-1223.
2. Vita A, De Peri L, Deste G, et al. The effect of antipsychotic treatment on cortical gray matter changes in schizophrenia: does the class matter? A meta-analysis and meta-regression of longitudinal magnetic resonance imaging studies. Biol Psychiatry. 2015;78(6):403-412.
3. Wakade CG, Mahadik SP, Waller JL, et al. Atypical neuroleptics stimulate neurogenesis in adult rat brain. J Neurosci Res. 2002;69(1):72-79.
4. Lieberman JA, Tollefson GD, Charles C, et al; HGDH Study Group. Antipsychotic drug effects on brain morphology in first-episode psychosis. Arch Gen Psychiatry. 2005;62(4):361-370.
5. Nasrallah HA. Impaired neuroplasticity in schizophrenia and the neuro-regenerative effects of atypical antipsychotics. Medscape Psychiatry. http://www.medscape.org/viewarticle/569521. Published January 31, 2008. Accessed November 10, 2015.
6. Nasrallah HA. Haloperidol clearly is neurotoxic. Should it be banned? Current Psychiatry. 2012;12(7):7-8.
7. Nandra KS, Agius M. The differences between typical and atypical antipsychotics: the effects on neurogenesis. Psychiatr Danub. 2012;24(suppl 1):S95-S99.
8. Nasrallah HA, Hopkins T, Pixley SK, et al. Differential effects of antipsychotic and antidepressant drugs on neurogenic region in rats. Brain Res. 2010;354:23-29.
9. Pillai A, Tery AV, Mahadik SP. Differential effects of long-term treatment with typical and atypical antipsychotics on NGF and BNDF levels in rat striatum and hippocampus. Schizophr Res. 2006;82(1):95-106.
10. Buckley PF, Pillai A, Evans D, et al. Brain derived neurotropic factor in first-episode psychosis. Schizophr Res. 2007;91(1-3):1-5.
11. Martin MV, Mimics K, Nisenbaum LK, et al. Olanzapine reversed brain gene expression changes induced by phencyclidines treatment in non-human primates. Mol Neuropsychiatry. 2015;1(2):82-93.
12. Yan BC, Park JH, Ahn JH, et al. Neuroprotection of posttreatment with risperidone, an atypical antipsychotic drug, in rat and gerbil models of ischemic stroke and the maintenance of antioxidants in a gerbil model of ischemic stroke. J Neurosci Res. 2014;92(6):795-807.
13. Yulug B, Yildiz A, Güzel O, et al. Risperidone attenuates brain damage after focal cerebral ischemia in vivo. Brain Res Bull. 2006;69(6):656-659.
14. Yulug B, Yildiz A, Hüdaoglu O, et al. Olanzapine attenuates brain damage after focal cerebral ischemia in vivo. Brain Res Bull. 2006;71(1-3):296-300.
15. Elsworth JD, Morrow BA. Hajszan T, et al. Phencyclidine-induced loss of asymmetric spine synapses in rodent prefrontal cortex is reversed by acute and chronic treatment with olanzapine. Neuropsychopharmacology. 2001;36(10):2054-2061.
16. Seki Y, Kato TA, Monji A, et al. Pretreatment of aripiprazole and minocycline, but not haloperidol, suppresses oligodendrocyte damage from interferon-y-stimulated microglia in co-culture model. Schizophr Res. 2013;151(1-3):20-28.
17. Bian Q, Kato T, Monji A, et al. The effect of atypical antipsychotics, perospirone, ziprasidone and quetiapine on microglial activation induced by interferon-gamma. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(1):42-48.
18. Wang HD, Deutch AY. Dopamine depletion of the prefrontal cortex induces dendritic spine loss: reversal by atypical antipsychotic drug treatment. Neuropsychopharmacology. 2008;33(6):1276-1286.
19. Miller BJ, Buckley P, Seabolt W, et al. Meta-analysis of cytokine alternations in schizophrenia: clinical status and antipsychotic effects. Biol Psychiatry. 2011;70(7):663-671.
20. Nasrallah HA. Beyond dopamine: The ‘other’ effects of antipsychotics. Current Psychiatry. 2013;12(6):8-9.
21. Yang MC, Lung FW. Neuroprotection of paliperidone on SH-SY5Y cells against β-amyloid peptide(25-35), N-methyl-4-phenylpyridinium ion, and hydrogen peroxide-induced cell death. Psychopharmacology (Berl). 2011;217(3):397-410.
22. Peng L, Zhu D, Feng X, et al. Paliperidone protects prefrontal cortical neurons from damages caused by MK-801 via Akt1/GSK3β _signaling pathway. Schizophr Res. 2013;147(1):14-23.23.
Stojkovic´ T, Radonjic´ NV, Velimirovic´ M, et al. Risperidone reverses phencyclidine induced decrease in glutathione levels and alternations of antioxidant defense in rat brain. Prog Neuropsychopharmacol Biol Psychiatry. 2012;39(1):192-199.
24. Koprivica V, Regardie K, Wolff C, et al. Aripiprazole protects cortical neurons from glutamate toxicity. Eur J Pharmacol. 2011;651(1-3):73-76.
25. Vaidya VA, Marek GJ, Aghajanian GK, et al. 5-HT2A receptor-mediated regulation of brain-derived neurotrophic factor mRNA in the hippocampus and the neocortex. J Neurosci. 1997;17(8):2785-2795.
26. Meridith GE, Switzer RC 3rd, Napier TC. Short-term, D2 receptor blockade induces synaptic degeneration, reduces levels of tyrosine hydroxylase and brain-derived neurotrophic factor, and enhances D2-mediated firing in the ventral pallidum. Brain Res. 2004;995(1):14-22.
27. Lieberman JA, Perkins DO, Jarskog LF. Neuroprotection: a therapeutic strategy to prevent deterioration associated with schizophrenia. CNS Spectr. 2007;12(suppl 4):1-13; quiz 14.
Cardiothoracic Surgery Resident Poster Competition
Represent your institution with a scientific poster of your clinical/investigative research at the 2016 AATS Annual Meeting in Baltimore, Maryland, May 14-18, 2016. The Competition is open to senior cardiothoracic surgery residents and/or congenital heart surgery fellows worldwide
North American residents must (1) Be in their last year of either an ACGME-accredited or RCPSC-accredited US or Canadian cardiothoracic surgery residency program or congenital heart surgery fellowship, and (2) Upload a support letter from their Program Director as part of their application.
International residents must (1) Be in their last year of a cardiothoracic training program at an AATS Member’s institution, and (2) Upload a sponsor letter from an AATS member at their institution as part of their application.
Application — Residents/fellows must provide a brief abstract of the research on their posters. Research may include information previously presented and/or published.
Benefits — Participant/institutions will (1) Receive a $500 stipend to offset travel and hotel accommodation to the Annual Meeting, (2) Receive a complimentary Annual Meeting registration, and (3) Have access to a Skills Course (Saturday, May 14) and Postgraduate Course (Sunday, May 15).
The participant’s training program will be responsible for the cost of any additional resident/fellow travel and accommodations.
Deadline: January 22, 2016
Represent your institution with a scientific poster of your clinical/investigative research at the 2016 AATS Annual Meeting in Baltimore, Maryland, May 14-18, 2016. The Competition is open to senior cardiothoracic surgery residents and/or congenital heart surgery fellows worldwide
North American residents must (1) Be in their last year of either an ACGME-accredited or RCPSC-accredited US or Canadian cardiothoracic surgery residency program or congenital heart surgery fellowship, and (2) Upload a support letter from their Program Director as part of their application.
International residents must (1) Be in their last year of a cardiothoracic training program at an AATS Member’s institution, and (2) Upload a sponsor letter from an AATS member at their institution as part of their application.
Application — Residents/fellows must provide a brief abstract of the research on their posters. Research may include information previously presented and/or published.
Benefits — Participant/institutions will (1) Receive a $500 stipend to offset travel and hotel accommodation to the Annual Meeting, (2) Receive a complimentary Annual Meeting registration, and (3) Have access to a Skills Course (Saturday, May 14) and Postgraduate Course (Sunday, May 15).
The participant’s training program will be responsible for the cost of any additional resident/fellow travel and accommodations.
Deadline: January 22, 2016
Represent your institution with a scientific poster of your clinical/investigative research at the 2016 AATS Annual Meeting in Baltimore, Maryland, May 14-18, 2016. The Competition is open to senior cardiothoracic surgery residents and/or congenital heart surgery fellows worldwide
North American residents must (1) Be in their last year of either an ACGME-accredited or RCPSC-accredited US or Canadian cardiothoracic surgery residency program or congenital heart surgery fellowship, and (2) Upload a support letter from their Program Director as part of their application.
International residents must (1) Be in their last year of a cardiothoracic training program at an AATS Member’s institution, and (2) Upload a sponsor letter from an AATS member at their institution as part of their application.
Application — Residents/fellows must provide a brief abstract of the research on their posters. Research may include information previously presented and/or published.
Benefits — Participant/institutions will (1) Receive a $500 stipend to offset travel and hotel accommodation to the Annual Meeting, (2) Receive a complimentary Annual Meeting registration, and (3) Have access to a Skills Course (Saturday, May 14) and Postgraduate Course (Sunday, May 15).
The participant’s training program will be responsible for the cost of any additional resident/fellow travel and accommodations.
Deadline: January 22, 2016
Apply to the AATS Summer Internship Scholarship
Medical students: Submissions are open for the AATS Summer Internship Scholarship giving up to 40 successful candidates the opportunity to spend eight weeks during the summer working in the cardiothoracic surgery department of a North American AATS member.
Program Goal — The program aims to broaden medical students’ educational experiences and to provide insight into cardiothoracic surgery.
Eligibility — (1) Candidates must be a North American first- or second-year medical student (as of January 2, 2016), (2) Host sponsor (an AATS member) must provide a letter of support/approval, and (3) Internships must take place at the institution of the host sponsor.
Application — The application must include no more than a one-page outline that includes what the candidate hopes to accomplish during his/her eight-week scholarship and specifies the proposed intern’s exposure to CT surgery, including both laboratory and clinical experience
Selection — (1) No more than two students from a single institution may receive a scholarship, (2) If two students from the same institution are selected, each must have a different host sponsor, and (3) Award recipients must agree to submit a summary report and evaluation of their experience to AATS within 60 days of internship completion.
Deadline: Friday, January 15, 2016
Medical students: Submissions are open for the AATS Summer Internship Scholarship giving up to 40 successful candidates the opportunity to spend eight weeks during the summer working in the cardiothoracic surgery department of a North American AATS member.
Program Goal — The program aims to broaden medical students’ educational experiences and to provide insight into cardiothoracic surgery.
Eligibility — (1) Candidates must be a North American first- or second-year medical student (as of January 2, 2016), (2) Host sponsor (an AATS member) must provide a letter of support/approval, and (3) Internships must take place at the institution of the host sponsor.
Application — The application must include no more than a one-page outline that includes what the candidate hopes to accomplish during his/her eight-week scholarship and specifies the proposed intern’s exposure to CT surgery, including both laboratory and clinical experience
Selection — (1) No more than two students from a single institution may receive a scholarship, (2) If two students from the same institution are selected, each must have a different host sponsor, and (3) Award recipients must agree to submit a summary report and evaluation of their experience to AATS within 60 days of internship completion.
Deadline: Friday, January 15, 2016
Medical students: Submissions are open for the AATS Summer Internship Scholarship giving up to 40 successful candidates the opportunity to spend eight weeks during the summer working in the cardiothoracic surgery department of a North American AATS member.
Program Goal — The program aims to broaden medical students’ educational experiences and to provide insight into cardiothoracic surgery.
Eligibility — (1) Candidates must be a North American first- or second-year medical student (as of January 2, 2016), (2) Host sponsor (an AATS member) must provide a letter of support/approval, and (3) Internships must take place at the institution of the host sponsor.
Application — The application must include no more than a one-page outline that includes what the candidate hopes to accomplish during his/her eight-week scholarship and specifies the proposed intern’s exposure to CT surgery, including both laboratory and clinical experience
Selection — (1) No more than two students from a single institution may receive a scholarship, (2) If two students from the same institution are selected, each must have a different host sponsor, and (3) Award recipients must agree to submit a summary report and evaluation of their experience to AATS within 60 days of internship completion.
Deadline: Friday, January 15, 2016
Be an AATS Member for a Day
Medical Students, General Surgery Residents and I-6 CT Surgical Residents — Submissions are open for the AATS Member for a Day program. Up to 30 successful candidates will have the opportunity to accompany an AATS Member Mentor for portions of the 2016 AATS Annual Meeting.
Dates/Location: May 14-18, 2016, Baltimore, MD
Eligibility — Applicants must be North American medical students, general surgery residents or I-6 cardiothoracic residents (within their first three years).
Program Goals — Are to (1) offer insight into cardiothoracic surgery, and (2) provide an opportunity to network and build relationships within the cardiothoracic surgical community.
Benefits — (1) Complimentary hotel accommodation for a minimum of three and maximum of four nights at an AATS Annual Meeting hotel, (2) a $500 stipend to help offset travel costs, and (3) an additional $250 stipend to offset the cost of meals.
Deadline: Friday, January 15, 2016
Medical Students, General Surgery Residents and I-6 CT Surgical Residents — Submissions are open for the AATS Member for a Day program. Up to 30 successful candidates will have the opportunity to accompany an AATS Member Mentor for portions of the 2016 AATS Annual Meeting.
Dates/Location: May 14-18, 2016, Baltimore, MD
Eligibility — Applicants must be North American medical students, general surgery residents or I-6 cardiothoracic residents (within their first three years).
Program Goals — Are to (1) offer insight into cardiothoracic surgery, and (2) provide an opportunity to network and build relationships within the cardiothoracic surgical community.
Benefits — (1) Complimentary hotel accommodation for a minimum of three and maximum of four nights at an AATS Annual Meeting hotel, (2) a $500 stipend to help offset travel costs, and (3) an additional $250 stipend to offset the cost of meals.
Deadline: Friday, January 15, 2016
Medical Students, General Surgery Residents and I-6 CT Surgical Residents — Submissions are open for the AATS Member for a Day program. Up to 30 successful candidates will have the opportunity to accompany an AATS Member Mentor for portions of the 2016 AATS Annual Meeting.
Dates/Location: May 14-18, 2016, Baltimore, MD
Eligibility — Applicants must be North American medical students, general surgery residents or I-6 cardiothoracic residents (within their first three years).
Program Goals — Are to (1) offer insight into cardiothoracic surgery, and (2) provide an opportunity to network and build relationships within the cardiothoracic surgical community.
Benefits — (1) Complimentary hotel accommodation for a minimum of three and maximum of four nights at an AATS Annual Meeting hotel, (2) a $500 stipend to help offset travel costs, and (3) an additional $250 stipend to offset the cost of meals.
Deadline: Friday, January 15, 2016
ACR: Years of TNF blockers did not increase risk of lymphoma in RA
SAN FRANCISCO – Rheumatoid arthritis conferred a doubling of the risk of lymphoma when compared against the general population in a large Swedish registry study, regardless of the patients’ experience with biological disease-modifying antirheumatic drugs.
But patients who took biological disease-modifying antirheumatic drugs (bDMARDs) had a sixfold greater risk of natural killer or T-cell lymphoma than did the general population, and that association was about four times stronger than for patients who had never taken biologics, reported Dr. Karin Hellgren, who led the study at the Karolinska Institute in Stockholm. That finding in particular shows the need to keep studying the links between bDMARDs and specific lymphoma subtypes, he said.
Severe rheumatoid arthritis seems to strongly increase the risk of lymphoma (Arthritis Rheum. 2006;54[3]:692-701), but researchers have debated whether the reason relates to bDMARDs or RA itself. Clinical trials have produced conflicting results; observational studies have reported no overall link between bDMARDs and lymphoma, but have raised questions about long-term exposure, the effects of individual agents, and lymphoma subtypes, Dr. Hellgren said at the annual meeting of the American College of Rheumatology.
To delve deeper into these issues, he and his associates compared 15 years of data for 13,240 RA patients on bDMARDs from the Swedish Biologics (ARTIS), Patient, and Cancer Registers and a national cohort of 46,568 bDMARD-naive patients. The researchers also compared both groups with 458,846 age- and gender-matched adults from the Swedish Population Register, following individuals until the end of 2012 or until lymphoma diagnosis, death, emigration, or bDMARD initiation, in the case of the naive patients.
Overall, patients with RA averaged one diagnosis of lymphoma per 1,000 population, compared with 0.5 cases per 1,000 for the overall population of Sweden, Dr. Hellgren said. In terms of absolute numbers, there were 241 cases of lymphoma among bDMARD-naive patients, 1,413 cases in the general population, and 69 cases among patients on bDMARDs, including 68 who were taking TNF inhibitors. The average age of the latter group of patients was 57 years. They had been diagnosed with RA at about age 50, had a mean 28-joint Disease Activity Score score of 5.3, and averaged 5.9 years of exposure to TNF inhibitors. Their risk of any type of malignant lymphoma was about 20% higher than for bDMARD-naive patients, but the difference was insignificant overall and in subgroups stratified by gender, age, and year starting treatment. Likewise, although patients were at greater risk of lymphoma if they had been on bDMARDs for 5-15 years (hazard ratio, 1.9) than for less time (HRs, about 1.0), there was no significant difference in risk compared with bDMARD-naive patients.
“There also were no statistically significant differences between drugs,” including infliximab (Remicade), etanercept (Enbrel), and adalimumab (Humira), Dr. Hellgren said. “In terms of the newer TNF inhibitors and other biologics, data are still too scarce to evaluate,” he added. Both exposed and bDMARD-naive patients were at especially high risk of Hodgkin lymphoma and diffuse large B-cell lymphoma, compared with the general population, but the strongest association of all was between bDMARD exposure and natural killer or T-cell lymphoma (HR, 6.0; 95% CI, 2.7-13.3). “The distribution of lymphoma subtypes warrants further assessment,” Dr. Hellgren concluded.
The ARTIS registry is funded by AbbVie, Bristol-Myers Squibb, Roche, Merck, Pfizer, Sobi, Lilly, and UCB. Dr. Hellgren had no disclosures. One coauthor reported financial relationships with AstraZeneca, Pfizer, UCB, Roche, Merck, Bristol-Myers Squibb, and AbbVie.
SAN FRANCISCO – Rheumatoid arthritis conferred a doubling of the risk of lymphoma when compared against the general population in a large Swedish registry study, regardless of the patients’ experience with biological disease-modifying antirheumatic drugs.
But patients who took biological disease-modifying antirheumatic drugs (bDMARDs) had a sixfold greater risk of natural killer or T-cell lymphoma than did the general population, and that association was about four times stronger than for patients who had never taken biologics, reported Dr. Karin Hellgren, who led the study at the Karolinska Institute in Stockholm. That finding in particular shows the need to keep studying the links between bDMARDs and specific lymphoma subtypes, he said.
Severe rheumatoid arthritis seems to strongly increase the risk of lymphoma (Arthritis Rheum. 2006;54[3]:692-701), but researchers have debated whether the reason relates to bDMARDs or RA itself. Clinical trials have produced conflicting results; observational studies have reported no overall link between bDMARDs and lymphoma, but have raised questions about long-term exposure, the effects of individual agents, and lymphoma subtypes, Dr. Hellgren said at the annual meeting of the American College of Rheumatology.
To delve deeper into these issues, he and his associates compared 15 years of data for 13,240 RA patients on bDMARDs from the Swedish Biologics (ARTIS), Patient, and Cancer Registers and a national cohort of 46,568 bDMARD-naive patients. The researchers also compared both groups with 458,846 age- and gender-matched adults from the Swedish Population Register, following individuals until the end of 2012 or until lymphoma diagnosis, death, emigration, or bDMARD initiation, in the case of the naive patients.
Overall, patients with RA averaged one diagnosis of lymphoma per 1,000 population, compared with 0.5 cases per 1,000 for the overall population of Sweden, Dr. Hellgren said. In terms of absolute numbers, there were 241 cases of lymphoma among bDMARD-naive patients, 1,413 cases in the general population, and 69 cases among patients on bDMARDs, including 68 who were taking TNF inhibitors. The average age of the latter group of patients was 57 years. They had been diagnosed with RA at about age 50, had a mean 28-joint Disease Activity Score score of 5.3, and averaged 5.9 years of exposure to TNF inhibitors. Their risk of any type of malignant lymphoma was about 20% higher than for bDMARD-naive patients, but the difference was insignificant overall and in subgroups stratified by gender, age, and year starting treatment. Likewise, although patients were at greater risk of lymphoma if they had been on bDMARDs for 5-15 years (hazard ratio, 1.9) than for less time (HRs, about 1.0), there was no significant difference in risk compared with bDMARD-naive patients.
“There also were no statistically significant differences between drugs,” including infliximab (Remicade), etanercept (Enbrel), and adalimumab (Humira), Dr. Hellgren said. “In terms of the newer TNF inhibitors and other biologics, data are still too scarce to evaluate,” he added. Both exposed and bDMARD-naive patients were at especially high risk of Hodgkin lymphoma and diffuse large B-cell lymphoma, compared with the general population, but the strongest association of all was between bDMARD exposure and natural killer or T-cell lymphoma (HR, 6.0; 95% CI, 2.7-13.3). “The distribution of lymphoma subtypes warrants further assessment,” Dr. Hellgren concluded.
The ARTIS registry is funded by AbbVie, Bristol-Myers Squibb, Roche, Merck, Pfizer, Sobi, Lilly, and UCB. Dr. Hellgren had no disclosures. One coauthor reported financial relationships with AstraZeneca, Pfizer, UCB, Roche, Merck, Bristol-Myers Squibb, and AbbVie.
SAN FRANCISCO – Rheumatoid arthritis conferred a doubling of the risk of lymphoma when compared against the general population in a large Swedish registry study, regardless of the patients’ experience with biological disease-modifying antirheumatic drugs.
But patients who took biological disease-modifying antirheumatic drugs (bDMARDs) had a sixfold greater risk of natural killer or T-cell lymphoma than did the general population, and that association was about four times stronger than for patients who had never taken biologics, reported Dr. Karin Hellgren, who led the study at the Karolinska Institute in Stockholm. That finding in particular shows the need to keep studying the links between bDMARDs and specific lymphoma subtypes, he said.
Severe rheumatoid arthritis seems to strongly increase the risk of lymphoma (Arthritis Rheum. 2006;54[3]:692-701), but researchers have debated whether the reason relates to bDMARDs or RA itself. Clinical trials have produced conflicting results; observational studies have reported no overall link between bDMARDs and lymphoma, but have raised questions about long-term exposure, the effects of individual agents, and lymphoma subtypes, Dr. Hellgren said at the annual meeting of the American College of Rheumatology.
To delve deeper into these issues, he and his associates compared 15 years of data for 13,240 RA patients on bDMARDs from the Swedish Biologics (ARTIS), Patient, and Cancer Registers and a national cohort of 46,568 bDMARD-naive patients. The researchers also compared both groups with 458,846 age- and gender-matched adults from the Swedish Population Register, following individuals until the end of 2012 or until lymphoma diagnosis, death, emigration, or bDMARD initiation, in the case of the naive patients.
Overall, patients with RA averaged one diagnosis of lymphoma per 1,000 population, compared with 0.5 cases per 1,000 for the overall population of Sweden, Dr. Hellgren said. In terms of absolute numbers, there were 241 cases of lymphoma among bDMARD-naive patients, 1,413 cases in the general population, and 69 cases among patients on bDMARDs, including 68 who were taking TNF inhibitors. The average age of the latter group of patients was 57 years. They had been diagnosed with RA at about age 50, had a mean 28-joint Disease Activity Score score of 5.3, and averaged 5.9 years of exposure to TNF inhibitors. Their risk of any type of malignant lymphoma was about 20% higher than for bDMARD-naive patients, but the difference was insignificant overall and in subgroups stratified by gender, age, and year starting treatment. Likewise, although patients were at greater risk of lymphoma if they had been on bDMARDs for 5-15 years (hazard ratio, 1.9) than for less time (HRs, about 1.0), there was no significant difference in risk compared with bDMARD-naive patients.
“There also were no statistically significant differences between drugs,” including infliximab (Remicade), etanercept (Enbrel), and adalimumab (Humira), Dr. Hellgren said. “In terms of the newer TNF inhibitors and other biologics, data are still too scarce to evaluate,” he added. Both exposed and bDMARD-naive patients were at especially high risk of Hodgkin lymphoma and diffuse large B-cell lymphoma, compared with the general population, but the strongest association of all was between bDMARD exposure and natural killer or T-cell lymphoma (HR, 6.0; 95% CI, 2.7-13.3). “The distribution of lymphoma subtypes warrants further assessment,” Dr. Hellgren concluded.
The ARTIS registry is funded by AbbVie, Bristol-Myers Squibb, Roche, Merck, Pfizer, Sobi, Lilly, and UCB. Dr. Hellgren had no disclosures. One coauthor reported financial relationships with AstraZeneca, Pfizer, UCB, Roche, Merck, Bristol-Myers Squibb, and AbbVie.
AT THE ACR ANNUAL MEETING
Key clinical point: Exposure to biological disease-modifying antirheumatic drugs did not increase the overall risk of lymphoma among patients with rheumatoid arthritis.
Major finding: Patients were at greater risk of lymphoma if they had been on bDMARDs for 5-15 years (hazard ratio, 1.9) than for less time (HRs, about 1.0), but there was no significant difference in risk compared with bDMARD-naive patients. However, patients on bDMARDs had about a fourfold greater risk of NK/T-cell lymphoma compared with bDMARD-naive patients.
Data source: A matched registry analysis of 13,240 patients from the Swedish Biologics (ARTIS), Patient, and Cancer Registers; 46,568 bio-naive patients; and 458,846 members of the general population.
Disclosures: The ARTIS registry is funded by AbbVie, Bristol-Myers Squibb, Roche, Merck, Pfizer, Sobi, Lilly, and UCB. Dr. Hellgren had no disclosures. One coauthor reported financial relationships with AstraZeneca, Pfizer, UCB, Roche, Merck, Bristol-Myers Squibb, and AbbVie.
MDA may help prevent malaria resurgence

Plasmodium vivax
Image by Mae Melvin
Mass drug administration (MDA) may have helped prevent a resurgence of malaria in Greece, according to research published in PLOS Neglected Tropical Diseases.
Greece was declared malaria-free in 1974 and remained that way until 2011, when there was an outbreak of Plasmodium vivax malaria in Southern Greece.
The outbreak was linked to the presence of agricultural workers from malaria-endemic regions in malaria-receptive areas.
There were 21 P vivax cases from arriving immigrants reported in the Southern agricultural area of Evrotas, along with 36 local cases.
So Greece implemented an integrated control program, with house visits established to screen immigrants from malaria-endemic countries.
Screening included a rapid diagnostic test for those reporting symptoms associated with malaria, along with blood sampling for smear and molecular testing for malaria. Directly observed treatment was provided for all patients who tested positive.
A vector control program was also implemented, with indoor residual spraying and long-lasting insecticide nets provided in areas close to mosquito breeding sites.
Despite these interventions, 20 more cases of malaria were reported in 2012.
Due to fears that the malaria parasite may be re-establishing itself in the area, Greece implemented an MDA program. It consisted of a single course of chloroquine and primaquine, which are the first-line recommended antimalarials for P vivax.
The program was implemented prior to the onset of peak adult mosquito activity, and field teams remained in situ continuing the active case detection until the end of the mosquito season. They recorded and managed adverse events daily.
The researchers identified an immigrant population of 1270 individuals, mostly from Pakistan and Afghanistan. The MDA covered 87% of this population.
No malaria cases were reported for 2013 and 2014, when the MDA was ongoing.
Of the treated individuals, 13% reported gastrointestinal symptoms from primaquine, while 36% reported non-severe side effects from chloroquine, including headaches, dizziness, and gastrointestinal complaints.
One potentially serious adverse event was recorded. It was a case of primaquine-induced hemolysis due to a false-normal G6PD level obtained prior to enrollment. The patient was hospitalized and recovered fully.
The researchers said that, in this case, the MDA program was a suitable and effective response for a small and geographically confined population over a short seasonal transition period. And the combination of 2 drugs minimized the risk of drug resistance.
The team added that, although an observational study of this nature cannot assess the extent to which the MDA program was responsible for eliminating malaria, it indicates that MDA should be considered and can be effective in local settings alongside other malaria control measures. ![]()

Plasmodium vivax
Image by Mae Melvin
Mass drug administration (MDA) may have helped prevent a resurgence of malaria in Greece, according to research published in PLOS Neglected Tropical Diseases.
Greece was declared malaria-free in 1974 and remained that way until 2011, when there was an outbreak of Plasmodium vivax malaria in Southern Greece.
The outbreak was linked to the presence of agricultural workers from malaria-endemic regions in malaria-receptive areas.
There were 21 P vivax cases from arriving immigrants reported in the Southern agricultural area of Evrotas, along with 36 local cases.
So Greece implemented an integrated control program, with house visits established to screen immigrants from malaria-endemic countries.
Screening included a rapid diagnostic test for those reporting symptoms associated with malaria, along with blood sampling for smear and molecular testing for malaria. Directly observed treatment was provided for all patients who tested positive.
A vector control program was also implemented, with indoor residual spraying and long-lasting insecticide nets provided in areas close to mosquito breeding sites.
Despite these interventions, 20 more cases of malaria were reported in 2012.
Due to fears that the malaria parasite may be re-establishing itself in the area, Greece implemented an MDA program. It consisted of a single course of chloroquine and primaquine, which are the first-line recommended antimalarials for P vivax.
The program was implemented prior to the onset of peak adult mosquito activity, and field teams remained in situ continuing the active case detection until the end of the mosquito season. They recorded and managed adverse events daily.
The researchers identified an immigrant population of 1270 individuals, mostly from Pakistan and Afghanistan. The MDA covered 87% of this population.
No malaria cases were reported for 2013 and 2014, when the MDA was ongoing.
Of the treated individuals, 13% reported gastrointestinal symptoms from primaquine, while 36% reported non-severe side effects from chloroquine, including headaches, dizziness, and gastrointestinal complaints.
One potentially serious adverse event was recorded. It was a case of primaquine-induced hemolysis due to a false-normal G6PD level obtained prior to enrollment. The patient was hospitalized and recovered fully.
The researchers said that, in this case, the MDA program was a suitable and effective response for a small and geographically confined population over a short seasonal transition period. And the combination of 2 drugs minimized the risk of drug resistance.
The team added that, although an observational study of this nature cannot assess the extent to which the MDA program was responsible for eliminating malaria, it indicates that MDA should be considered and can be effective in local settings alongside other malaria control measures. ![]()

Plasmodium vivax
Image by Mae Melvin
Mass drug administration (MDA) may have helped prevent a resurgence of malaria in Greece, according to research published in PLOS Neglected Tropical Diseases.
Greece was declared malaria-free in 1974 and remained that way until 2011, when there was an outbreak of Plasmodium vivax malaria in Southern Greece.
The outbreak was linked to the presence of agricultural workers from malaria-endemic regions in malaria-receptive areas.
There were 21 P vivax cases from arriving immigrants reported in the Southern agricultural area of Evrotas, along with 36 local cases.
So Greece implemented an integrated control program, with house visits established to screen immigrants from malaria-endemic countries.
Screening included a rapid diagnostic test for those reporting symptoms associated with malaria, along with blood sampling for smear and molecular testing for malaria. Directly observed treatment was provided for all patients who tested positive.
A vector control program was also implemented, with indoor residual spraying and long-lasting insecticide nets provided in areas close to mosquito breeding sites.
Despite these interventions, 20 more cases of malaria were reported in 2012.
Due to fears that the malaria parasite may be re-establishing itself in the area, Greece implemented an MDA program. It consisted of a single course of chloroquine and primaquine, which are the first-line recommended antimalarials for P vivax.
The program was implemented prior to the onset of peak adult mosquito activity, and field teams remained in situ continuing the active case detection until the end of the mosquito season. They recorded and managed adverse events daily.
The researchers identified an immigrant population of 1270 individuals, mostly from Pakistan and Afghanistan. The MDA covered 87% of this population.
No malaria cases were reported for 2013 and 2014, when the MDA was ongoing.
Of the treated individuals, 13% reported gastrointestinal symptoms from primaquine, while 36% reported non-severe side effects from chloroquine, including headaches, dizziness, and gastrointestinal complaints.
One potentially serious adverse event was recorded. It was a case of primaquine-induced hemolysis due to a false-normal G6PD level obtained prior to enrollment. The patient was hospitalized and recovered fully.
The researchers said that, in this case, the MDA program was a suitable and effective response for a small and geographically confined population over a short seasonal transition period. And the combination of 2 drugs minimized the risk of drug resistance.
The team added that, although an observational study of this nature cannot assess the extent to which the MDA program was responsible for eliminating malaria, it indicates that MDA should be considered and can be effective in local settings alongside other malaria control measures. ![]()
CHMP recommends pegaspargase for ALL
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended marketing authorization for pegaspargase (Oncaspar) as part of combination antineoplastic therapy for pediatric and adult patients with acute lymphoblastic leukemia (ALL).
The CHMP’s opinion has been referred to the European Commission, which grants marketing authorization for drugs in the European Union.
If approved, pegaspargase will be marketed for the aforementioned indication in the 28 member countries of the European Union, as well as Iceland, Liechtenstein, and Norway.
Baxalta Incorporated, the company developing pegaspargase, expects a decision from the European Commission early next year.
First-line ALL
Researchers have evaluated the safety and effectiveness of pegaspargase in a study of 118 pediatric patients (ages 1 to 9) with newly diagnosed ALL. The patients were randomized 1:1 to pegaspargase or native E coli L-asparaginase, both as part of combination therapy.
Asparagine depletion (magnitude and duration) was similar between the 2 treatment arms. Event-free survival rates were also similar (about 80% in both arms), but the study was not designed to evaluate differences in event-free survival.
Grade 3/4 adverse events occurring in the pegaspargase and native E coli L-asparaginase arms, respectively, were abnormal liver tests (5% and 8%), elevated transaminases (3% and 7%), hyperbilirubinemia (2% and 2%), hyperglycemia (5% and 3%), central nervous system thrombosis (3% and 3%), coagulopathy (2% and 5%), pancreatitis (2% and 2%), and clinical allergic reactions to asparaginase (2% and 0%).
Previously treated ALL
Researchers have evaluated the effectiveness of pegaspargase in 4 open-label studies of patients with a history of prior clinical allergic reaction to asparaginase. The studies enrolled a total of 42 patients with multiply relapsed acute leukemia (39 with ALL).
Patients received pegaspargase as a single agent or in combination with multi-agent chemotherapy. The re-induction response rate was 50%—36% complete responses and 14% partial responses.
These results were similar to the overall response rates reported for patients with ALL receiving second-line, native E coli L-asparaginase-containing re-induction chemotherapy. However, antitumor activity was observed with single-agent pegaspargase as well (3 responses).
Adverse event information on pegaspargase in relapsed ALL has been compiled from 5 clinical trials. The studies enrolled a total of 174 patients with relapsed ALL who received pegaspargase as a single agent or in combination with multi-agent chemotherapy.
Sixty-two of the patients had prior hypersensitivity reactions to asparaginase, and 112 did not. Allergic reactions to pegaspargase occurred in 32% of previously hypersensitive patients and 10% of non-hypersensitive patients.
The most common adverse events observed in patients who received pegaspargase were clinical allergic reactions, elevated transaminases, hyperbilirubinemia, and coagulopathies. The most common serious adverse events due to pegaspargase were thrombosis (4%), hyperglycemia requiring insulin therapy (3%), and pancreatitis (1%).
For more details on these trials and pegaspargase, see the product information. ![]()

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended marketing authorization for pegaspargase (Oncaspar) as part of combination antineoplastic therapy for pediatric and adult patients with acute lymphoblastic leukemia (ALL).
The CHMP’s opinion has been referred to the European Commission, which grants marketing authorization for drugs in the European Union.
If approved, pegaspargase will be marketed for the aforementioned indication in the 28 member countries of the European Union, as well as Iceland, Liechtenstein, and Norway.
Baxalta Incorporated, the company developing pegaspargase, expects a decision from the European Commission early next year.
First-line ALL
Researchers have evaluated the safety and effectiveness of pegaspargase in a study of 118 pediatric patients (ages 1 to 9) with newly diagnosed ALL. The patients were randomized 1:1 to pegaspargase or native E coli L-asparaginase, both as part of combination therapy.
Asparagine depletion (magnitude and duration) was similar between the 2 treatment arms. Event-free survival rates were also similar (about 80% in both arms), but the study was not designed to evaluate differences in event-free survival.
Grade 3/4 adverse events occurring in the pegaspargase and native E coli L-asparaginase arms, respectively, were abnormal liver tests (5% and 8%), elevated transaminases (3% and 7%), hyperbilirubinemia (2% and 2%), hyperglycemia (5% and 3%), central nervous system thrombosis (3% and 3%), coagulopathy (2% and 5%), pancreatitis (2% and 2%), and clinical allergic reactions to asparaginase (2% and 0%).
Previously treated ALL
Researchers have evaluated the effectiveness of pegaspargase in 4 open-label studies of patients with a history of prior clinical allergic reaction to asparaginase. The studies enrolled a total of 42 patients with multiply relapsed acute leukemia (39 with ALL).
Patients received pegaspargase as a single agent or in combination with multi-agent chemotherapy. The re-induction response rate was 50%—36% complete responses and 14% partial responses.
These results were similar to the overall response rates reported for patients with ALL receiving second-line, native E coli L-asparaginase-containing re-induction chemotherapy. However, antitumor activity was observed with single-agent pegaspargase as well (3 responses).
Adverse event information on pegaspargase in relapsed ALL has been compiled from 5 clinical trials. The studies enrolled a total of 174 patients with relapsed ALL who received pegaspargase as a single agent or in combination with multi-agent chemotherapy.
Sixty-two of the patients had prior hypersensitivity reactions to asparaginase, and 112 did not. Allergic reactions to pegaspargase occurred in 32% of previously hypersensitive patients and 10% of non-hypersensitive patients.
The most common adverse events observed in patients who received pegaspargase were clinical allergic reactions, elevated transaminases, hyperbilirubinemia, and coagulopathies. The most common serious adverse events due to pegaspargase were thrombosis (4%), hyperglycemia requiring insulin therapy (3%), and pancreatitis (1%).
For more details on these trials and pegaspargase, see the product information. ![]()

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended marketing authorization for pegaspargase (Oncaspar) as part of combination antineoplastic therapy for pediatric and adult patients with acute lymphoblastic leukemia (ALL).
The CHMP’s opinion has been referred to the European Commission, which grants marketing authorization for drugs in the European Union.
If approved, pegaspargase will be marketed for the aforementioned indication in the 28 member countries of the European Union, as well as Iceland, Liechtenstein, and Norway.
Baxalta Incorporated, the company developing pegaspargase, expects a decision from the European Commission early next year.
First-line ALL
Researchers have evaluated the safety and effectiveness of pegaspargase in a study of 118 pediatric patients (ages 1 to 9) with newly diagnosed ALL. The patients were randomized 1:1 to pegaspargase or native E coli L-asparaginase, both as part of combination therapy.
Asparagine depletion (magnitude and duration) was similar between the 2 treatment arms. Event-free survival rates were also similar (about 80% in both arms), but the study was not designed to evaluate differences in event-free survival.
Grade 3/4 adverse events occurring in the pegaspargase and native E coli L-asparaginase arms, respectively, were abnormal liver tests (5% and 8%), elevated transaminases (3% and 7%), hyperbilirubinemia (2% and 2%), hyperglycemia (5% and 3%), central nervous system thrombosis (3% and 3%), coagulopathy (2% and 5%), pancreatitis (2% and 2%), and clinical allergic reactions to asparaginase (2% and 0%).
Previously treated ALL
Researchers have evaluated the effectiveness of pegaspargase in 4 open-label studies of patients with a history of prior clinical allergic reaction to asparaginase. The studies enrolled a total of 42 patients with multiply relapsed acute leukemia (39 with ALL).
Patients received pegaspargase as a single agent or in combination with multi-agent chemotherapy. The re-induction response rate was 50%—36% complete responses and 14% partial responses.
These results were similar to the overall response rates reported for patients with ALL receiving second-line, native E coli L-asparaginase-containing re-induction chemotherapy. However, antitumor activity was observed with single-agent pegaspargase as well (3 responses).
Adverse event information on pegaspargase in relapsed ALL has been compiled from 5 clinical trials. The studies enrolled a total of 174 patients with relapsed ALL who received pegaspargase as a single agent or in combination with multi-agent chemotherapy.
Sixty-two of the patients had prior hypersensitivity reactions to asparaginase, and 112 did not. Allergic reactions to pegaspargase occurred in 32% of previously hypersensitive patients and 10% of non-hypersensitive patients.
The most common adverse events observed in patients who received pegaspargase were clinical allergic reactions, elevated transaminases, hyperbilirubinemia, and coagulopathies. The most common serious adverse events due to pegaspargase were thrombosis (4%), hyperglycemia requiring insulin therapy (3%), and pancreatitis (1%).
For more details on these trials and pegaspargase, see the product information. ![]()
FDA approves new indication for dabigatran
Photo by ec-jpr
The US Food and Drug Administration (FDA) has approved the direct thrombin inhibitor dabigatran etexilate mesylate (Pradaxa) for the prophylaxis of deep venous thrombosis (DVT) and pulmonary embolism (PE) in patients who have undergone hip replacement surgery.
Dabigatran was initially approved by the FDA in 2010 to reduce the risk of stroke and systemic embolism in patients with non-valvular atrial fibrillation.
In 2014, dabigatran was approved to treat DVT and PE in patients who have been treated with a parenteral anticoagulant for 5 to 10 days and to reduce the risk of recurrent DVT and PE in patients who have been previously treated.
A reversal agent for dabigatran, known as idarucizumab (Praxbind), was approved by the FDA last month.
Trial data
The latest approval of dabigatran is based on results of 2 randomized, double-blind, phase 3 trials in patients undergoing total hip replacement, RE-NOVATE™ and RE-NOVATE II™.
In the RE-NOVATE trial, 3494 patients were randomized to 3 groups receiving prophylactic treatment with 1 of 2 doses of dabigatran (220 mg or 150 mg) once daily or enoxaparin at 40 mg once daily for 28 to 35 days.
The first dabigatran group was given a dose of 110 mg on the day of surgery and 220 mg daily thereafter. The second dabigatran group received a dose of 75 mg on the day of surgery and 150 mg daily thereafter.
Patients taking dabigatran at 220 mg had a lower composite total of venous thromboembolism (VTE) and all-cause death (6.0%) than patients on enoxaparin (6.7%). However, the rate of major bleeding was higher with dabigatran at 220 mg (2.0%) than with enoxaparin (1.6%).
In the RE-NOVATE II trial, 2055 patients were randomized to prophylactic treatment for 28 to 35 days with dabigatran at 220 mg once daily or enoxaparin at 40 mg once daily. Patients receiving dabigatran were treated with a dose of 110 mg on the day of surgery and 220 mg daily thereafter.
The composite total of VTE and all-cause death occurred in 7.7% of patients in the dabigatran group and 8.8% of patients in the enoxaparin group. Again, the rate of major bleeding was higher with dabigatran at 220 mg (1.4%) than with enoxaparin (0.9%).
In both studies, the rate of major gastrointestinal bleeds in patients receiving dabigatran and enoxaparin was the same (0.1%). The rate of any gastrointestinal bleeds was 1.4% for dabigatran and 0.9% for enoxaparin.
The most common adverse events in both studies were gastrointestinal disorders. The incidence was the same across the dabigatran and enoxaparin treatment groups (39.5%).
Dyspepsia (including abdominal pain upper, abdominal pain, abdominal discomfort, and epigastric discomfort) occurred more frequently in patients receiving dabigatran (4.1%) than enoxaparin (3.8%).
Gastritis-like symptoms (including gastritis, GERD, esophagitis, erosive gastritis, and gastric hemorrhage) were less common in patients receiving dabigatran (0.6%) than enoxaparin (1.0%). Clinical myocardial infarction was reported in 2 (0.1%) dabigatran patients and 6 (0.3%) enoxaparin patients.
Dabigatran is marketed as Pradaxa by Boehringer Ingelheim. For more details on the drug, see the prescribing information. ![]()

Photo by ec-jpr
The US Food and Drug Administration (FDA) has approved the direct thrombin inhibitor dabigatran etexilate mesylate (Pradaxa) for the prophylaxis of deep venous thrombosis (DVT) and pulmonary embolism (PE) in patients who have undergone hip replacement surgery.
Dabigatran was initially approved by the FDA in 2010 to reduce the risk of stroke and systemic embolism in patients with non-valvular atrial fibrillation.
In 2014, dabigatran was approved to treat DVT and PE in patients who have been treated with a parenteral anticoagulant for 5 to 10 days and to reduce the risk of recurrent DVT and PE in patients who have been previously treated.
A reversal agent for dabigatran, known as idarucizumab (Praxbind), was approved by the FDA last month.
Trial data
The latest approval of dabigatran is based on results of 2 randomized, double-blind, phase 3 trials in patients undergoing total hip replacement, RE-NOVATE™ and RE-NOVATE II™.
In the RE-NOVATE trial, 3494 patients were randomized to 3 groups receiving prophylactic treatment with 1 of 2 doses of dabigatran (220 mg or 150 mg) once daily or enoxaparin at 40 mg once daily for 28 to 35 days.
The first dabigatran group was given a dose of 110 mg on the day of surgery and 220 mg daily thereafter. The second dabigatran group received a dose of 75 mg on the day of surgery and 150 mg daily thereafter.
Patients taking dabigatran at 220 mg had a lower composite total of venous thromboembolism (VTE) and all-cause death (6.0%) than patients on enoxaparin (6.7%). However, the rate of major bleeding was higher with dabigatran at 220 mg (2.0%) than with enoxaparin (1.6%).
In the RE-NOVATE II trial, 2055 patients were randomized to prophylactic treatment for 28 to 35 days with dabigatran at 220 mg once daily or enoxaparin at 40 mg once daily. Patients receiving dabigatran were treated with a dose of 110 mg on the day of surgery and 220 mg daily thereafter.
The composite total of VTE and all-cause death occurred in 7.7% of patients in the dabigatran group and 8.8% of patients in the enoxaparin group. Again, the rate of major bleeding was higher with dabigatran at 220 mg (1.4%) than with enoxaparin (0.9%).
In both studies, the rate of major gastrointestinal bleeds in patients receiving dabigatran and enoxaparin was the same (0.1%). The rate of any gastrointestinal bleeds was 1.4% for dabigatran and 0.9% for enoxaparin.
The most common adverse events in both studies were gastrointestinal disorders. The incidence was the same across the dabigatran and enoxaparin treatment groups (39.5%).
Dyspepsia (including abdominal pain upper, abdominal pain, abdominal discomfort, and epigastric discomfort) occurred more frequently in patients receiving dabigatran (4.1%) than enoxaparin (3.8%).
Gastritis-like symptoms (including gastritis, GERD, esophagitis, erosive gastritis, and gastric hemorrhage) were less common in patients receiving dabigatran (0.6%) than enoxaparin (1.0%). Clinical myocardial infarction was reported in 2 (0.1%) dabigatran patients and 6 (0.3%) enoxaparin patients.
Dabigatran is marketed as Pradaxa by Boehringer Ingelheim. For more details on the drug, see the prescribing information. ![]()

Photo by ec-jpr
The US Food and Drug Administration (FDA) has approved the direct thrombin inhibitor dabigatran etexilate mesylate (Pradaxa) for the prophylaxis of deep venous thrombosis (DVT) and pulmonary embolism (PE) in patients who have undergone hip replacement surgery.
Dabigatran was initially approved by the FDA in 2010 to reduce the risk of stroke and systemic embolism in patients with non-valvular atrial fibrillation.
In 2014, dabigatran was approved to treat DVT and PE in patients who have been treated with a parenteral anticoagulant for 5 to 10 days and to reduce the risk of recurrent DVT and PE in patients who have been previously treated.
A reversal agent for dabigatran, known as idarucizumab (Praxbind), was approved by the FDA last month.
Trial data
The latest approval of dabigatran is based on results of 2 randomized, double-blind, phase 3 trials in patients undergoing total hip replacement, RE-NOVATE™ and RE-NOVATE II™.
In the RE-NOVATE trial, 3494 patients were randomized to 3 groups receiving prophylactic treatment with 1 of 2 doses of dabigatran (220 mg or 150 mg) once daily or enoxaparin at 40 mg once daily for 28 to 35 days.
The first dabigatran group was given a dose of 110 mg on the day of surgery and 220 mg daily thereafter. The second dabigatran group received a dose of 75 mg on the day of surgery and 150 mg daily thereafter.
Patients taking dabigatran at 220 mg had a lower composite total of venous thromboembolism (VTE) and all-cause death (6.0%) than patients on enoxaparin (6.7%). However, the rate of major bleeding was higher with dabigatran at 220 mg (2.0%) than with enoxaparin (1.6%).
In the RE-NOVATE II trial, 2055 patients were randomized to prophylactic treatment for 28 to 35 days with dabigatran at 220 mg once daily or enoxaparin at 40 mg once daily. Patients receiving dabigatran were treated with a dose of 110 mg on the day of surgery and 220 mg daily thereafter.
The composite total of VTE and all-cause death occurred in 7.7% of patients in the dabigatran group and 8.8% of patients in the enoxaparin group. Again, the rate of major bleeding was higher with dabigatran at 220 mg (1.4%) than with enoxaparin (0.9%).
In both studies, the rate of major gastrointestinal bleeds in patients receiving dabigatran and enoxaparin was the same (0.1%). The rate of any gastrointestinal bleeds was 1.4% for dabigatran and 0.9% for enoxaparin.
The most common adverse events in both studies were gastrointestinal disorders. The incidence was the same across the dabigatran and enoxaparin treatment groups (39.5%).
Dyspepsia (including abdominal pain upper, abdominal pain, abdominal discomfort, and epigastric discomfort) occurred more frequently in patients receiving dabigatran (4.1%) than enoxaparin (3.8%).
Gastritis-like symptoms (including gastritis, GERD, esophagitis, erosive gastritis, and gastric hemorrhage) were less common in patients receiving dabigatran (0.6%) than enoxaparin (1.0%). Clinical myocardial infarction was reported in 2 (0.1%) dabigatran patients and 6 (0.3%) enoxaparin patients.
Dabigatran is marketed as Pradaxa by Boehringer Ingelheim. For more details on the drug, see the prescribing information. ![]()