User login
News and Views that Matter to the Ob.Gyn.
gambling
compulsive behaviors
ammunition
assault rifle
black jack
Boko Haram
bondage
child abuse
cocaine
Daech
drug paraphernalia
explosion
gun
human trafficking
ISIL
ISIS
Islamic caliphate
Islamic state
mixed martial arts
MMA
molestation
national rifle association
NRA
nsfw
pedophile
pedophilia
poker
porn
pornography
psychedelic drug
recreational drug
sex slave rings
slot machine
terrorism
terrorist
Texas hold 'em
UFC
substance abuse
abuseed
abuseer
abusees
abuseing
abusely
abuses
aeolus
aeolused
aeoluser
aeoluses
aeolusing
aeolusly
aeoluss
ahole
aholeed
aholeer
aholees
aholeing
aholely
aholes
alcohol
alcoholed
alcoholer
alcoholes
alcoholing
alcoholly
alcohols
allman
allmaned
allmaner
allmanes
allmaning
allmanly
allmans
alted
altes
alting
altly
alts
analed
analer
anales
analing
anally
analprobe
analprobeed
analprobeer
analprobees
analprobeing
analprobely
analprobes
anals
anilingus
anilingused
anilinguser
anilinguses
anilingusing
anilingusly
anilinguss
anus
anused
anuser
anuses
anusing
anusly
anuss
areola
areolaed
areolaer
areolaes
areolaing
areolaly
areolas
areole
areoleed
areoleer
areolees
areoleing
areolely
areoles
arian
arianed
arianer
arianes
arianing
arianly
arians
aryan
aryaned
aryaner
aryanes
aryaning
aryanly
aryans
asiaed
asiaer
asiaes
asiaing
asialy
asias
ass
ass hole
ass lick
ass licked
ass licker
ass lickes
ass licking
ass lickly
ass licks
assbang
assbanged
assbangeded
assbangeder
assbangedes
assbangeding
assbangedly
assbangeds
assbanger
assbanges
assbanging
assbangly
assbangs
assbangsed
assbangser
assbangses
assbangsing
assbangsly
assbangss
assed
asser
asses
assesed
asseser
asseses
assesing
assesly
assess
assfuck
assfucked
assfucker
assfuckered
assfuckerer
assfuckeres
assfuckering
assfuckerly
assfuckers
assfuckes
assfucking
assfuckly
assfucks
asshat
asshated
asshater
asshates
asshating
asshatly
asshats
assholeed
assholeer
assholees
assholeing
assholely
assholes
assholesed
assholeser
assholeses
assholesing
assholesly
assholess
assing
assly
assmaster
assmastered
assmasterer
assmasteres
assmastering
assmasterly
assmasters
assmunch
assmunched
assmuncher
assmunches
assmunching
assmunchly
assmunchs
asss
asswipe
asswipeed
asswipeer
asswipees
asswipeing
asswipely
asswipes
asswipesed
asswipeser
asswipeses
asswipesing
asswipesly
asswipess
azz
azzed
azzer
azzes
azzing
azzly
azzs
babeed
babeer
babees
babeing
babely
babes
babesed
babeser
babeses
babesing
babesly
babess
ballsac
ballsaced
ballsacer
ballsaces
ballsacing
ballsack
ballsacked
ballsacker
ballsackes
ballsacking
ballsackly
ballsacks
ballsacly
ballsacs
ballsed
ballser
ballses
ballsing
ballsly
ballss
barf
barfed
barfer
barfes
barfing
barfly
barfs
bastard
bastarded
bastarder
bastardes
bastarding
bastardly
bastards
bastardsed
bastardser
bastardses
bastardsing
bastardsly
bastardss
bawdy
bawdyed
bawdyer
bawdyes
bawdying
bawdyly
bawdys
beaner
beanered
beanerer
beaneres
beanering
beanerly
beaners
beardedclam
beardedclamed
beardedclamer
beardedclames
beardedclaming
beardedclamly
beardedclams
beastiality
beastialityed
beastialityer
beastialityes
beastialitying
beastialityly
beastialitys
beatch
beatched
beatcher
beatches
beatching
beatchly
beatchs
beater
beatered
beaterer
beateres
beatering
beaterly
beaters
beered
beerer
beeres
beering
beerly
beeyotch
beeyotched
beeyotcher
beeyotches
beeyotching
beeyotchly
beeyotchs
beotch
beotched
beotcher
beotches
beotching
beotchly
beotchs
biatch
biatched
biatcher
biatches
biatching
biatchly
biatchs
big tits
big titsed
big titser
big titses
big titsing
big titsly
big titss
bigtits
bigtitsed
bigtitser
bigtitses
bigtitsing
bigtitsly
bigtitss
bimbo
bimboed
bimboer
bimboes
bimboing
bimboly
bimbos
bisexualed
bisexualer
bisexuales
bisexualing
bisexually
bisexuals
bitch
bitched
bitcheded
bitcheder
bitchedes
bitcheding
bitchedly
bitcheds
bitcher
bitches
bitchesed
bitcheser
bitcheses
bitchesing
bitchesly
bitchess
bitching
bitchly
bitchs
bitchy
bitchyed
bitchyer
bitchyes
bitchying
bitchyly
bitchys
bleached
bleacher
bleaches
bleaching
bleachly
bleachs
blow job
blow jobed
blow jober
blow jobes
blow jobing
blow jobly
blow jobs
blowed
blower
blowes
blowing
blowjob
blowjobed
blowjober
blowjobes
blowjobing
blowjobly
blowjobs
blowjobsed
blowjobser
blowjobses
blowjobsing
blowjobsly
blowjobss
blowly
blows
boink
boinked
boinker
boinkes
boinking
boinkly
boinks
bollock
bollocked
bollocker
bollockes
bollocking
bollockly
bollocks
bollocksed
bollockser
bollockses
bollocksing
bollocksly
bollockss
bollok
bolloked
bolloker
bollokes
bolloking
bollokly
bolloks
boner
bonered
bonerer
boneres
bonering
bonerly
boners
bonersed
bonerser
bonerses
bonersing
bonersly
bonerss
bong
bonged
bonger
bonges
bonging
bongly
bongs
boob
boobed
boober
boobes
boobies
boobiesed
boobieser
boobieses
boobiesing
boobiesly
boobiess
boobing
boobly
boobs
boobsed
boobser
boobses
boobsing
boobsly
boobss
booby
boobyed
boobyer
boobyes
boobying
boobyly
boobys
booger
boogered
boogerer
boogeres
boogering
boogerly
boogers
bookie
bookieed
bookieer
bookiees
bookieing
bookiely
bookies
bootee
booteeed
booteeer
booteees
booteeing
booteely
bootees
bootie
bootieed
bootieer
bootiees
bootieing
bootiely
booties
booty
bootyed
bootyer
bootyes
bootying
bootyly
bootys
boozeed
boozeer
boozees
boozeing
boozely
boozer
boozered
boozerer
boozeres
boozering
boozerly
boozers
boozes
boozy
boozyed
boozyer
boozyes
boozying
boozyly
boozys
bosomed
bosomer
bosomes
bosoming
bosomly
bosoms
bosomy
bosomyed
bosomyer
bosomyes
bosomying
bosomyly
bosomys
bugger
buggered
buggerer
buggeres
buggering
buggerly
buggers
bukkake
bukkakeed
bukkakeer
bukkakees
bukkakeing
bukkakely
bukkakes
bull shit
bull shited
bull shiter
bull shites
bull shiting
bull shitly
bull shits
bullshit
bullshited
bullshiter
bullshites
bullshiting
bullshitly
bullshits
bullshitsed
bullshitser
bullshitses
bullshitsing
bullshitsly
bullshitss
bullshitted
bullshitteded
bullshitteder
bullshittedes
bullshitteding
bullshittedly
bullshitteds
bullturds
bullturdsed
bullturdser
bullturdses
bullturdsing
bullturdsly
bullturdss
bung
bunged
bunger
bunges
bunging
bungly
bungs
busty
bustyed
bustyer
bustyes
bustying
bustyly
bustys
butt
butt fuck
butt fucked
butt fucker
butt fuckes
butt fucking
butt fuckly
butt fucks
butted
buttes
buttfuck
buttfucked
buttfucker
buttfuckered
buttfuckerer
buttfuckeres
buttfuckering
buttfuckerly
buttfuckers
buttfuckes
buttfucking
buttfuckly
buttfucks
butting
buttly
buttplug
buttpluged
buttpluger
buttpluges
buttpluging
buttplugly
buttplugs
butts
caca
cacaed
cacaer
cacaes
cacaing
cacaly
cacas
cahone
cahoneed
cahoneer
cahonees
cahoneing
cahonely
cahones
cameltoe
cameltoeed
cameltoeer
cameltoees
cameltoeing
cameltoely
cameltoes
carpetmuncher
carpetmunchered
carpetmuncherer
carpetmuncheres
carpetmunchering
carpetmuncherly
carpetmunchers
cawk
cawked
cawker
cawkes
cawking
cawkly
cawks
chinc
chinced
chincer
chinces
chincing
chincly
chincs
chincsed
chincser
chincses
chincsing
chincsly
chincss
chink
chinked
chinker
chinkes
chinking
chinkly
chinks
chode
chodeed
chodeer
chodees
chodeing
chodely
chodes
chodesed
chodeser
chodeses
chodesing
chodesly
chodess
clit
clited
cliter
clites
cliting
clitly
clitoris
clitorised
clitoriser
clitorises
clitorising
clitorisly
clitoriss
clitorus
clitorused
clitoruser
clitoruses
clitorusing
clitorusly
clitoruss
clits
clitsed
clitser
clitses
clitsing
clitsly
clitss
clitty
clittyed
clittyer
clittyes
clittying
clittyly
clittys
cocain
cocaine
cocained
cocaineed
cocaineer
cocainees
cocaineing
cocainely
cocainer
cocaines
cocaining
cocainly
cocains
cock
cock sucker
cock suckered
cock suckerer
cock suckeres
cock suckering
cock suckerly
cock suckers
cockblock
cockblocked
cockblocker
cockblockes
cockblocking
cockblockly
cockblocks
cocked
cocker
cockes
cockholster
cockholstered
cockholsterer
cockholsteres
cockholstering
cockholsterly
cockholsters
cocking
cockknocker
cockknockered
cockknockerer
cockknockeres
cockknockering
cockknockerly
cockknockers
cockly
cocks
cocksed
cockser
cockses
cocksing
cocksly
cocksmoker
cocksmokered
cocksmokerer
cocksmokeres
cocksmokering
cocksmokerly
cocksmokers
cockss
cocksucker
cocksuckered
cocksuckerer
cocksuckeres
cocksuckering
cocksuckerly
cocksuckers
coital
coitaled
coitaler
coitales
coitaling
coitally
coitals
commie
commieed
commieer
commiees
commieing
commiely
commies
condomed
condomer
condomes
condoming
condomly
condoms
coon
cooned
cooner
coones
cooning
coonly
coons
coonsed
coonser
coonses
coonsing
coonsly
coonss
corksucker
corksuckered
corksuckerer
corksuckeres
corksuckering
corksuckerly
corksuckers
cracked
crackwhore
crackwhoreed
crackwhoreer
crackwhorees
crackwhoreing
crackwhorely
crackwhores
crap
craped
craper
crapes
craping
craply
crappy
crappyed
crappyer
crappyes
crappying
crappyly
crappys
cum
cumed
cumer
cumes
cuming
cumly
cummin
cummined
cumminer
cummines
cumming
cumminged
cumminger
cumminges
cumminging
cummingly
cummings
cummining
cumminly
cummins
cums
cumshot
cumshoted
cumshoter
cumshotes
cumshoting
cumshotly
cumshots
cumshotsed
cumshotser
cumshotses
cumshotsing
cumshotsly
cumshotss
cumslut
cumsluted
cumsluter
cumslutes
cumsluting
cumslutly
cumsluts
cumstain
cumstained
cumstainer
cumstaines
cumstaining
cumstainly
cumstains
cunilingus
cunilingused
cunilinguser
cunilinguses
cunilingusing
cunilingusly
cunilinguss
cunnilingus
cunnilingused
cunnilinguser
cunnilinguses
cunnilingusing
cunnilingusly
cunnilinguss
cunny
cunnyed
cunnyer
cunnyes
cunnying
cunnyly
cunnys
cunt
cunted
cunter
cuntes
cuntface
cuntfaceed
cuntfaceer
cuntfacees
cuntfaceing
cuntfacely
cuntfaces
cunthunter
cunthuntered
cunthunterer
cunthunteres
cunthuntering
cunthunterly
cunthunters
cunting
cuntlick
cuntlicked
cuntlicker
cuntlickered
cuntlickerer
cuntlickeres
cuntlickering
cuntlickerly
cuntlickers
cuntlickes
cuntlicking
cuntlickly
cuntlicks
cuntly
cunts
cuntsed
cuntser
cuntses
cuntsing
cuntsly
cuntss
dago
dagoed
dagoer
dagoes
dagoing
dagoly
dagos
dagosed
dagoser
dagoses
dagosing
dagosly
dagoss
dammit
dammited
dammiter
dammites
dammiting
dammitly
dammits
damn
damned
damneded
damneder
damnedes
damneding
damnedly
damneds
damner
damnes
damning
damnit
damnited
damniter
damnites
damniting
damnitly
damnits
damnly
damns
dick
dickbag
dickbaged
dickbager
dickbages
dickbaging
dickbagly
dickbags
dickdipper
dickdippered
dickdipperer
dickdipperes
dickdippering
dickdipperly
dickdippers
dicked
dicker
dickes
dickface
dickfaceed
dickfaceer
dickfacees
dickfaceing
dickfacely
dickfaces
dickflipper
dickflippered
dickflipperer
dickflipperes
dickflippering
dickflipperly
dickflippers
dickhead
dickheaded
dickheader
dickheades
dickheading
dickheadly
dickheads
dickheadsed
dickheadser
dickheadses
dickheadsing
dickheadsly
dickheadss
dicking
dickish
dickished
dickisher
dickishes
dickishing
dickishly
dickishs
dickly
dickripper
dickrippered
dickripperer
dickripperes
dickrippering
dickripperly
dickrippers
dicks
dicksipper
dicksippered
dicksipperer
dicksipperes
dicksippering
dicksipperly
dicksippers
dickweed
dickweeded
dickweeder
dickweedes
dickweeding
dickweedly
dickweeds
dickwhipper
dickwhippered
dickwhipperer
dickwhipperes
dickwhippering
dickwhipperly
dickwhippers
dickzipper
dickzippered
dickzipperer
dickzipperes
dickzippering
dickzipperly
dickzippers
diddle
diddleed
diddleer
diddlees
diddleing
diddlely
diddles
dike
dikeed
dikeer
dikees
dikeing
dikely
dikes
dildo
dildoed
dildoer
dildoes
dildoing
dildoly
dildos
dildosed
dildoser
dildoses
dildosing
dildosly
dildoss
diligaf
diligafed
diligafer
diligafes
diligafing
diligafly
diligafs
dillweed
dillweeded
dillweeder
dillweedes
dillweeding
dillweedly
dillweeds
dimwit
dimwited
dimwiter
dimwites
dimwiting
dimwitly
dimwits
dingle
dingleed
dingleer
dinglees
dingleing
dinglely
dingles
dipship
dipshiped
dipshiper
dipshipes
dipshiping
dipshiply
dipships
dizzyed
dizzyer
dizzyes
dizzying
dizzyly
dizzys
doggiestyleed
doggiestyleer
doggiestylees
doggiestyleing
doggiestylely
doggiestyles
doggystyleed
doggystyleer
doggystylees
doggystyleing
doggystylely
doggystyles
dong
donged
donger
donges
donging
dongly
dongs
doofus
doofused
doofuser
doofuses
doofusing
doofusly
doofuss
doosh
dooshed
doosher
dooshes
dooshing
dooshly
dooshs
dopeyed
dopeyer
dopeyes
dopeying
dopeyly
dopeys
douchebag
douchebaged
douchebager
douchebages
douchebaging
douchebagly
douchebags
douchebagsed
douchebagser
douchebagses
douchebagsing
douchebagsly
douchebagss
doucheed
doucheer
douchees
doucheing
douchely
douches
douchey
doucheyed
doucheyer
doucheyes
doucheying
doucheyly
doucheys
drunk
drunked
drunker
drunkes
drunking
drunkly
drunks
dumass
dumassed
dumasser
dumasses
dumassing
dumassly
dumasss
dumbass
dumbassed
dumbasser
dumbasses
dumbassesed
dumbasseser
dumbasseses
dumbassesing
dumbassesly
dumbassess
dumbassing
dumbassly
dumbasss
dummy
dummyed
dummyer
dummyes
dummying
dummyly
dummys
dyke
dykeed
dykeer
dykees
dykeing
dykely
dykes
dykesed
dykeser
dykeses
dykesing
dykesly
dykess
erotic
eroticed
eroticer
erotices
eroticing
eroticly
erotics
extacy
extacyed
extacyer
extacyes
extacying
extacyly
extacys
extasy
extasyed
extasyer
extasyes
extasying
extasyly
extasys
fack
facked
facker
fackes
facking
fackly
facks
fag
faged
fager
fages
fagg
fagged
faggeded
faggeder
faggedes
faggeding
faggedly
faggeds
fagger
fagges
fagging
faggit
faggited
faggiter
faggites
faggiting
faggitly
faggits
faggly
faggot
faggoted
faggoter
faggotes
faggoting
faggotly
faggots
faggs
faging
fagly
fagot
fagoted
fagoter
fagotes
fagoting
fagotly
fagots
fags
fagsed
fagser
fagses
fagsing
fagsly
fagss
faig
faiged
faiger
faiges
faiging
faigly
faigs
faigt
faigted
faigter
faigtes
faigting
faigtly
faigts
fannybandit
fannybandited
fannybanditer
fannybandites
fannybanditing
fannybanditly
fannybandits
farted
farter
fartes
farting
fartknocker
fartknockered
fartknockerer
fartknockeres
fartknockering
fartknockerly
fartknockers
fartly
farts
felch
felched
felcher
felchered
felcherer
felcheres
felchering
felcherly
felchers
felches
felching
felchinged
felchinger
felchinges
felchinging
felchingly
felchings
felchly
felchs
fellate
fellateed
fellateer
fellatees
fellateing
fellately
fellates
fellatio
fellatioed
fellatioer
fellatioes
fellatioing
fellatioly
fellatios
feltch
feltched
feltcher
feltchered
feltcherer
feltcheres
feltchering
feltcherly
feltchers
feltches
feltching
feltchly
feltchs
feom
feomed
feomer
feomes
feoming
feomly
feoms
fisted
fisteded
fisteder
fistedes
fisteding
fistedly
fisteds
fisting
fistinged
fistinger
fistinges
fistinging
fistingly
fistings
fisty
fistyed
fistyer
fistyes
fistying
fistyly
fistys
floozy
floozyed
floozyer
floozyes
floozying
floozyly
floozys
foad
foaded
foader
foades
foading
foadly
foads
fondleed
fondleer
fondlees
fondleing
fondlely
fondles
foobar
foobared
foobarer
foobares
foobaring
foobarly
foobars
freex
freexed
freexer
freexes
freexing
freexly
freexs
frigg
frigga
friggaed
friggaer
friggaes
friggaing
friggaly
friggas
frigged
frigger
frigges
frigging
friggly
friggs
fubar
fubared
fubarer
fubares
fubaring
fubarly
fubars
fuck
fuckass
fuckassed
fuckasser
fuckasses
fuckassing
fuckassly
fuckasss
fucked
fuckeded
fuckeder
fuckedes
fuckeding
fuckedly
fuckeds
fucker
fuckered
fuckerer
fuckeres
fuckering
fuckerly
fuckers
fuckes
fuckface
fuckfaceed
fuckfaceer
fuckfacees
fuckfaceing
fuckfacely
fuckfaces
fuckin
fuckined
fuckiner
fuckines
fucking
fuckinged
fuckinger
fuckinges
fuckinging
fuckingly
fuckings
fuckining
fuckinly
fuckins
fuckly
fucknugget
fucknuggeted
fucknuggeter
fucknuggetes
fucknuggeting
fucknuggetly
fucknuggets
fucknut
fucknuted
fucknuter
fucknutes
fucknuting
fucknutly
fucknuts
fuckoff
fuckoffed
fuckoffer
fuckoffes
fuckoffing
fuckoffly
fuckoffs
fucks
fucksed
fuckser
fuckses
fucksing
fucksly
fuckss
fucktard
fucktarded
fucktarder
fucktardes
fucktarding
fucktardly
fucktards
fuckup
fuckuped
fuckuper
fuckupes
fuckuping
fuckuply
fuckups
fuckwad
fuckwaded
fuckwader
fuckwades
fuckwading
fuckwadly
fuckwads
fuckwit
fuckwited
fuckwiter
fuckwites
fuckwiting
fuckwitly
fuckwits
fudgepacker
fudgepackered
fudgepackerer
fudgepackeres
fudgepackering
fudgepackerly
fudgepackers
fuk
fuked
fuker
fukes
fuking
fukly
fuks
fvck
fvcked
fvcker
fvckes
fvcking
fvckly
fvcks
fxck
fxcked
fxcker
fxckes
fxcking
fxckly
fxcks
gae
gaeed
gaeer
gaees
gaeing
gaely
gaes
gai
gaied
gaier
gaies
gaiing
gaily
gais
ganja
ganjaed
ganjaer
ganjaes
ganjaing
ganjaly
ganjas
gayed
gayer
gayes
gaying
gayly
gays
gaysed
gayser
gayses
gaysing
gaysly
gayss
gey
geyed
geyer
geyes
geying
geyly
geys
gfc
gfced
gfcer
gfces
gfcing
gfcly
gfcs
gfy
gfyed
gfyer
gfyes
gfying
gfyly
gfys
ghay
ghayed
ghayer
ghayes
ghaying
ghayly
ghays
ghey
gheyed
gheyer
gheyes
gheying
gheyly
gheys
gigolo
gigoloed
gigoloer
gigoloes
gigoloing
gigololy
gigolos
goatse
goatseed
goatseer
goatsees
goatseing
goatsely
goatses
godamn
godamned
godamner
godamnes
godamning
godamnit
godamnited
godamniter
godamnites
godamniting
godamnitly
godamnits
godamnly
godamns
goddam
goddamed
goddamer
goddames
goddaming
goddamly
goddammit
goddammited
goddammiter
goddammites
goddammiting
goddammitly
goddammits
goddamn
goddamned
goddamner
goddamnes
goddamning
goddamnly
goddamns
goddams
goldenshower
goldenshowered
goldenshowerer
goldenshoweres
goldenshowering
goldenshowerly
goldenshowers
gonad
gonaded
gonader
gonades
gonading
gonadly
gonads
gonadsed
gonadser
gonadses
gonadsing
gonadsly
gonadss
gook
gooked
gooker
gookes
gooking
gookly
gooks
gooksed
gookser
gookses
gooksing
gooksly
gookss
gringo
gringoed
gringoer
gringoes
gringoing
gringoly
gringos
gspot
gspoted
gspoter
gspotes
gspoting
gspotly
gspots
gtfo
gtfoed
gtfoer
gtfoes
gtfoing
gtfoly
gtfos
guido
guidoed
guidoer
guidoes
guidoing
guidoly
guidos
handjob
handjobed
handjober
handjobes
handjobing
handjobly
handjobs
hard on
hard oned
hard oner
hard ones
hard oning
hard only
hard ons
hardknight
hardknighted
hardknighter
hardknightes
hardknighting
hardknightly
hardknights
hebe
hebeed
hebeer
hebees
hebeing
hebely
hebes
heeb
heebed
heeber
heebes
heebing
heebly
heebs
hell
helled
heller
helles
helling
hellly
hells
hemp
hemped
hemper
hempes
hemping
hemply
hemps
heroined
heroiner
heroines
heroining
heroinly
heroins
herp
herped
herper
herpes
herpesed
herpeser
herpeses
herpesing
herpesly
herpess
herping
herply
herps
herpy
herpyed
herpyer
herpyes
herpying
herpyly
herpys
hitler
hitlered
hitlerer
hitleres
hitlering
hitlerly
hitlers
hived
hiver
hives
hiving
hivly
hivs
hobag
hobaged
hobager
hobages
hobaging
hobagly
hobags
homey
homeyed
homeyer
homeyes
homeying
homeyly
homeys
homo
homoed
homoer
homoes
homoey
homoeyed
homoeyer
homoeyes
homoeying
homoeyly
homoeys
homoing
homoly
homos
honky
honkyed
honkyer
honkyes
honkying
honkyly
honkys
hooch
hooched
hoocher
hooches
hooching
hoochly
hoochs
hookah
hookahed
hookaher
hookahes
hookahing
hookahly
hookahs
hooker
hookered
hookerer
hookeres
hookering
hookerly
hookers
hoor
hoored
hoorer
hoores
hooring
hoorly
hoors
hootch
hootched
hootcher
hootches
hootching
hootchly
hootchs
hooter
hootered
hooterer
hooteres
hootering
hooterly
hooters
hootersed
hooterser
hooterses
hootersing
hootersly
hooterss
horny
hornyed
hornyer
hornyes
hornying
hornyly
hornys
houstoned
houstoner
houstones
houstoning
houstonly
houstons
hump
humped
humpeded
humpeder
humpedes
humpeding
humpedly
humpeds
humper
humpes
humping
humpinged
humpinger
humpinges
humpinging
humpingly
humpings
humply
humps
husbanded
husbander
husbandes
husbanding
husbandly
husbands
hussy
hussyed
hussyer
hussyes
hussying
hussyly
hussys
hymened
hymener
hymenes
hymening
hymenly
hymens
inbred
inbreded
inbreder
inbredes
inbreding
inbredly
inbreds
incest
incested
incester
incestes
incesting
incestly
incests
injun
injuned
injuner
injunes
injuning
injunly
injuns
jackass
jackassed
jackasser
jackasses
jackassing
jackassly
jackasss
jackhole
jackholeed
jackholeer
jackholees
jackholeing
jackholely
jackholes
jackoff
jackoffed
jackoffer
jackoffes
jackoffing
jackoffly
jackoffs
jap
japed
japer
japes
japing
japly
japs
japsed
japser
japses
japsing
japsly
japss
jerkoff
jerkoffed
jerkoffer
jerkoffes
jerkoffing
jerkoffly
jerkoffs
jerks
jism
jismed
jismer
jismes
jisming
jismly
jisms
jiz
jized
jizer
jizes
jizing
jizly
jizm
jizmed
jizmer
jizmes
jizming
jizmly
jizms
jizs
jizz
jizzed
jizzeded
jizzeder
jizzedes
jizzeding
jizzedly
jizzeds
jizzer
jizzes
jizzing
jizzly
jizzs
junkie
junkieed
junkieer
junkiees
junkieing
junkiely
junkies
junky
junkyed
junkyer
junkyes
junkying
junkyly
junkys
kike
kikeed
kikeer
kikees
kikeing
kikely
kikes
kikesed
kikeser
kikeses
kikesing
kikesly
kikess
killed
killer
killes
killing
killly
kills
kinky
kinkyed
kinkyer
kinkyes
kinkying
kinkyly
kinkys
kkk
kkked
kkker
kkkes
kkking
kkkly
kkks
klan
klaned
klaner
klanes
klaning
klanly
klans
knobend
knobended
knobender
knobendes
knobending
knobendly
knobends
kooch
kooched
koocher
kooches
koochesed
koocheser
koocheses
koochesing
koochesly
koochess
kooching
koochly
koochs
kootch
kootched
kootcher
kootches
kootching
kootchly
kootchs
kraut
krauted
krauter
krautes
krauting
krautly
krauts
kyke
kykeed
kykeer
kykees
kykeing
kykely
kykes
lech
leched
lecher
leches
leching
lechly
lechs
leper
lepered
leperer
leperes
lepering
leperly
lepers
lesbiansed
lesbianser
lesbianses
lesbiansing
lesbiansly
lesbianss
lesbo
lesboed
lesboer
lesboes
lesboing
lesboly
lesbos
lesbosed
lesboser
lesboses
lesbosing
lesbosly
lesboss
lez
lezbianed
lezbianer
lezbianes
lezbianing
lezbianly
lezbians
lezbiansed
lezbianser
lezbianses
lezbiansing
lezbiansly
lezbianss
lezbo
lezboed
lezboer
lezboes
lezboing
lezboly
lezbos
lezbosed
lezboser
lezboses
lezbosing
lezbosly
lezboss
lezed
lezer
lezes
lezing
lezly
lezs
lezzie
lezzieed
lezzieer
lezziees
lezzieing
lezziely
lezzies
lezziesed
lezzieser
lezzieses
lezziesing
lezziesly
lezziess
lezzy
lezzyed
lezzyer
lezzyes
lezzying
lezzyly
lezzys
lmaoed
lmaoer
lmaoes
lmaoing
lmaoly
lmaos
lmfao
lmfaoed
lmfaoer
lmfaoes
lmfaoing
lmfaoly
lmfaos
loined
loiner
loines
loining
loinly
loins
loinsed
loinser
loinses
loinsing
loinsly
loinss
lubeed
lubeer
lubees
lubeing
lubely
lubes
lusty
lustyed
lustyer
lustyes
lustying
lustyly
lustys
massa
massaed
massaer
massaes
massaing
massaly
massas
masterbate
masterbateed
masterbateer
masterbatees
masterbateing
masterbately
masterbates
masterbating
masterbatinged
masterbatinger
masterbatinges
masterbatinging
masterbatingly
masterbatings
masterbation
masterbationed
masterbationer
masterbationes
masterbationing
masterbationly
masterbations
masturbate
masturbateed
masturbateer
masturbatees
masturbateing
masturbately
masturbates
masturbating
masturbatinged
masturbatinger
masturbatinges
masturbatinging
masturbatingly
masturbatings
masturbation
masturbationed
masturbationer
masturbationes
masturbationing
masturbationly
masturbations
methed
mether
methes
mething
methly
meths
militaryed
militaryer
militaryes
militarying
militaryly
militarys
mofo
mofoed
mofoer
mofoes
mofoing
mofoly
mofos
molest
molested
molester
molestes
molesting
molestly
molests
moolie
moolieed
moolieer
mooliees
moolieing
mooliely
moolies
moron
moroned
moroner
morones
moroning
moronly
morons
motherfucka
motherfuckaed
motherfuckaer
motherfuckaes
motherfuckaing
motherfuckaly
motherfuckas
motherfucker
motherfuckered
motherfuckerer
motherfuckeres
motherfuckering
motherfuckerly
motherfuckers
motherfucking
motherfuckinged
motherfuckinger
motherfuckinges
motherfuckinging
motherfuckingly
motherfuckings
mtherfucker
mtherfuckered
mtherfuckerer
mtherfuckeres
mtherfuckering
mtherfuckerly
mtherfuckers
mthrfucker
mthrfuckered
mthrfuckerer
mthrfuckeres
mthrfuckering
mthrfuckerly
mthrfuckers
mthrfucking
mthrfuckinged
mthrfuckinger
mthrfuckinges
mthrfuckinging
mthrfuckingly
mthrfuckings
muff
muffdiver
muffdivered
muffdiverer
muffdiveres
muffdivering
muffdiverly
muffdivers
muffed
muffer
muffes
muffing
muffly
muffs
murdered
murderer
murderes
murdering
murderly
murders
muthafuckaz
muthafuckazed
muthafuckazer
muthafuckazes
muthafuckazing
muthafuckazly
muthafuckazs
muthafucker
muthafuckered
muthafuckerer
muthafuckeres
muthafuckering
muthafuckerly
muthafuckers
mutherfucker
mutherfuckered
mutherfuckerer
mutherfuckeres
mutherfuckering
mutherfuckerly
mutherfuckers
mutherfucking
mutherfuckinged
mutherfuckinger
mutherfuckinges
mutherfuckinging
mutherfuckingly
mutherfuckings
muthrfucking
muthrfuckinged
muthrfuckinger
muthrfuckinges
muthrfuckinging
muthrfuckingly
muthrfuckings
nad
naded
nader
nades
nading
nadly
nads
nadsed
nadser
nadses
nadsing
nadsly
nadss
nakeded
nakeder
nakedes
nakeding
nakedly
nakeds
napalm
napalmed
napalmer
napalmes
napalming
napalmly
napalms
nappy
nappyed
nappyer
nappyes
nappying
nappyly
nappys
nazi
nazied
nazier
nazies
naziing
nazily
nazis
nazism
nazismed
nazismer
nazismes
nazisming
nazismly
nazisms
negro
negroed
negroer
negroes
negroing
negroly
negros
nigga
niggaed
niggaer
niggaes
niggah
niggahed
niggaher
niggahes
niggahing
niggahly
niggahs
niggaing
niggaly
niggas
niggased
niggaser
niggases
niggasing
niggasly
niggass
niggaz
niggazed
niggazer
niggazes
niggazing
niggazly
niggazs
nigger
niggered
niggerer
niggeres
niggering
niggerly
niggers
niggersed
niggerser
niggerses
niggersing
niggersly
niggerss
niggle
niggleed
niggleer
nigglees
niggleing
nigglely
niggles
niglet
nigleted
nigleter
nigletes
nigleting
nigletly
niglets
nimrod
nimroded
nimroder
nimrodes
nimroding
nimrodly
nimrods
ninny
ninnyed
ninnyer
ninnyes
ninnying
ninnyly
ninnys
nooky
nookyed
nookyer
nookyes
nookying
nookyly
nookys
nuccitelli
nuccitellied
nuccitellier
nuccitellies
nuccitelliing
nuccitellily
nuccitellis
nympho
nymphoed
nymphoer
nymphoes
nymphoing
nympholy
nymphos
opium
opiumed
opiumer
opiumes
opiuming
opiumly
opiums
orgies
orgiesed
orgieser
orgieses
orgiesing
orgiesly
orgiess
orgy
orgyed
orgyer
orgyes
orgying
orgyly
orgys
paddy
paddyed
paddyer
paddyes
paddying
paddyly
paddys
paki
pakied
pakier
pakies
pakiing
pakily
pakis
pantie
pantieed
pantieer
pantiees
pantieing
pantiely
panties
pantiesed
pantieser
pantieses
pantiesing
pantiesly
pantiess
panty
pantyed
pantyer
pantyes
pantying
pantyly
pantys
pastie
pastieed
pastieer
pastiees
pastieing
pastiely
pasties
pasty
pastyed
pastyer
pastyes
pastying
pastyly
pastys
pecker
peckered
peckerer
peckeres
peckering
peckerly
peckers
pedo
pedoed
pedoer
pedoes
pedoing
pedoly
pedophile
pedophileed
pedophileer
pedophilees
pedophileing
pedophilely
pedophiles
pedophilia
pedophiliac
pedophiliaced
pedophiliacer
pedophiliaces
pedophiliacing
pedophiliacly
pedophiliacs
pedophiliaed
pedophiliaer
pedophiliaes
pedophiliaing
pedophilialy
pedophilias
pedos
penial
penialed
penialer
peniales
penialing
penially
penials
penile
penileed
penileer
penilees
penileing
penilely
peniles
penis
penised
peniser
penises
penising
penisly
peniss
perversion
perversioned
perversioner
perversiones
perversioning
perversionly
perversions
peyote
peyoteed
peyoteer
peyotees
peyoteing
peyotely
peyotes
phuck
phucked
phucker
phuckes
phucking
phuckly
phucks
pillowbiter
pillowbitered
pillowbiterer
pillowbiteres
pillowbitering
pillowbiterly
pillowbiters
pimp
pimped
pimper
pimpes
pimping
pimply
pimps
pinko
pinkoed
pinkoer
pinkoes
pinkoing
pinkoly
pinkos
pissed
pisseded
pisseder
pissedes
pisseding
pissedly
pisseds
pisser
pisses
pissing
pissly
pissoff
pissoffed
pissoffer
pissoffes
pissoffing
pissoffly
pissoffs
pisss
polack
polacked
polacker
polackes
polacking
polackly
polacks
pollock
pollocked
pollocker
pollockes
pollocking
pollockly
pollocks
poon
pooned
pooner
poones
pooning
poonly
poons
poontang
poontanged
poontanger
poontanges
poontanging
poontangly
poontangs
porn
porned
porner
pornes
porning
pornly
porno
pornoed
pornoer
pornoes
pornography
pornographyed
pornographyer
pornographyes
pornographying
pornographyly
pornographys
pornoing
pornoly
pornos
porns
prick
pricked
pricker
prickes
pricking
prickly
pricks
prig
priged
priger
priges
priging
prigly
prigs
prostitute
prostituteed
prostituteer
prostitutees
prostituteing
prostitutely
prostitutes
prude
prudeed
prudeer
prudees
prudeing
prudely
prudes
punkass
punkassed
punkasser
punkasses
punkassing
punkassly
punkasss
punky
punkyed
punkyer
punkyes
punkying
punkyly
punkys
puss
pussed
pusser
pusses
pussies
pussiesed
pussieser
pussieses
pussiesing
pussiesly
pussiess
pussing
pussly
pusss
pussy
pussyed
pussyer
pussyes
pussying
pussyly
pussypounder
pussypoundered
pussypounderer
pussypounderes
pussypoundering
pussypounderly
pussypounders
pussys
puto
putoed
putoer
putoes
putoing
putoly
putos
queaf
queafed
queafer
queafes
queafing
queafly
queafs
queef
queefed
queefer
queefes
queefing
queefly
queefs
queer
queered
queerer
queeres
queering
queerly
queero
queeroed
queeroer
queeroes
queeroing
queeroly
queeros
queers
queersed
queerser
queerses
queersing
queersly
queerss
quicky
quickyed
quickyer
quickyes
quickying
quickyly
quickys
quim
quimed
quimer
quimes
quiming
quimly
quims
racy
racyed
racyer
racyes
racying
racyly
racys
rape
raped
rapeded
rapeder
rapedes
rapeding
rapedly
rapeds
rapeed
rapeer
rapees
rapeing
rapely
raper
rapered
raperer
raperes
rapering
raperly
rapers
rapes
rapist
rapisted
rapister
rapistes
rapisting
rapistly
rapists
raunch
raunched
rauncher
raunches
raunching
raunchly
raunchs
rectus
rectused
rectuser
rectuses
rectusing
rectusly
rectuss
reefer
reefered
reeferer
reeferes
reefering
reeferly
reefers
reetard
reetarded
reetarder
reetardes
reetarding
reetardly
reetards
reich
reiched
reicher
reiches
reiching
reichly
reichs
retard
retarded
retardeded
retardeder
retardedes
retardeding
retardedly
retardeds
retarder
retardes
retarding
retardly
retards
rimjob
rimjobed
rimjober
rimjobes
rimjobing
rimjobly
rimjobs
ritard
ritarded
ritarder
ritardes
ritarding
ritardly
ritards
rtard
rtarded
rtarder
rtardes
rtarding
rtardly
rtards
rum
rumed
rumer
rumes
ruming
rumly
rump
rumped
rumper
rumpes
rumping
rumply
rumprammer
rumprammered
rumprammerer
rumprammeres
rumprammering
rumprammerly
rumprammers
rumps
rums
ruski
ruskied
ruskier
ruskies
ruskiing
ruskily
ruskis
sadism
sadismed
sadismer
sadismes
sadisming
sadismly
sadisms
sadist
sadisted
sadister
sadistes
sadisting
sadistly
sadists
scag
scaged
scager
scages
scaging
scagly
scags
scantily
scantilyed
scantilyer
scantilyes
scantilying
scantilyly
scantilys
schlong
schlonged
schlonger
schlonges
schlonging
schlongly
schlongs
scrog
scroged
scroger
scroges
scroging
scrogly
scrogs
scrot
scrote
scroted
scroteed
scroteer
scrotees
scroteing
scrotely
scroter
scrotes
scroting
scrotly
scrots
scrotum
scrotumed
scrotumer
scrotumes
scrotuming
scrotumly
scrotums
scrud
scruded
scruder
scrudes
scruding
scrudly
scruds
scum
scumed
scumer
scumes
scuming
scumly
scums
seaman
seamaned
seamaner
seamanes
seamaning
seamanly
seamans
seamen
seamened
seamener
seamenes
seamening
seamenly
seamens
seduceed
seduceer
seducees
seduceing
seducely
seduces
semen
semened
semener
semenes
semening
semenly
semens
shamedame
shamedameed
shamedameer
shamedamees
shamedameing
shamedamely
shamedames
shit
shite
shiteater
shiteatered
shiteaterer
shiteateres
shiteatering
shiteaterly
shiteaters
shited
shiteed
shiteer
shitees
shiteing
shitely
shiter
shites
shitface
shitfaceed
shitfaceer
shitfacees
shitfaceing
shitfacely
shitfaces
shithead
shitheaded
shitheader
shitheades
shitheading
shitheadly
shitheads
shithole
shitholeed
shitholeer
shitholees
shitholeing
shitholely
shitholes
shithouse
shithouseed
shithouseer
shithousees
shithouseing
shithousely
shithouses
shiting
shitly
shits
shitsed
shitser
shitses
shitsing
shitsly
shitss
shitt
shitted
shitteded
shitteder
shittedes
shitteding
shittedly
shitteds
shitter
shittered
shitterer
shitteres
shittering
shitterly
shitters
shittes
shitting
shittly
shitts
shitty
shittyed
shittyer
shittyes
shittying
shittyly
shittys
shiz
shized
shizer
shizes
shizing
shizly
shizs
shooted
shooter
shootes
shooting
shootly
shoots
sissy
sissyed
sissyer
sissyes
sissying
sissyly
sissys
skag
skaged
skager
skages
skaging
skagly
skags
skank
skanked
skanker
skankes
skanking
skankly
skanks
slave
slaveed
slaveer
slavees
slaveing
slavely
slaves
sleaze
sleazeed
sleazeer
sleazees
sleazeing
sleazely
sleazes
sleazy
sleazyed
sleazyer
sleazyes
sleazying
sleazyly
sleazys
slut
slutdumper
slutdumpered
slutdumperer
slutdumperes
slutdumpering
slutdumperly
slutdumpers
sluted
sluter
slutes
sluting
slutkiss
slutkissed
slutkisser
slutkisses
slutkissing
slutkissly
slutkisss
slutly
sluts
slutsed
slutser
slutses
slutsing
slutsly
slutss
smegma
smegmaed
smegmaer
smegmaes
smegmaing
smegmaly
smegmas
smut
smuted
smuter
smutes
smuting
smutly
smuts
smutty
smuttyed
smuttyer
smuttyes
smuttying
smuttyly
smuttys
snatch
snatched
snatcher
snatches
snatching
snatchly
snatchs
sniper
snipered
sniperer
sniperes
snipering
sniperly
snipers
snort
snorted
snorter
snortes
snorting
snortly
snorts
snuff
snuffed
snuffer
snuffes
snuffing
snuffly
snuffs
sodom
sodomed
sodomer
sodomes
sodoming
sodomly
sodoms
spic
spiced
spicer
spices
spicing
spick
spicked
spicker
spickes
spicking
spickly
spicks
spicly
spics
spik
spoof
spoofed
spoofer
spoofes
spoofing
spoofly
spoofs
spooge
spoogeed
spoogeer
spoogees
spoogeing
spoogely
spooges
spunk
spunked
spunker
spunkes
spunking
spunkly
spunks
steamyed
steamyer
steamyes
steamying
steamyly
steamys
stfu
stfued
stfuer
stfues
stfuing
stfuly
stfus
stiffy
stiffyed
stiffyer
stiffyes
stiffying
stiffyly
stiffys
stoneded
stoneder
stonedes
stoneding
stonedly
stoneds
stupided
stupider
stupides
stupiding
stupidly
stupids
suckeded
suckeder
suckedes
suckeding
suckedly
suckeds
sucker
suckes
sucking
suckinged
suckinger
suckinges
suckinging
suckingly
suckings
suckly
sucks
sumofabiatch
sumofabiatched
sumofabiatcher
sumofabiatches
sumofabiatching
sumofabiatchly
sumofabiatchs
tard
tarded
tarder
tardes
tarding
tardly
tards
tawdry
tawdryed
tawdryer
tawdryes
tawdrying
tawdryly
tawdrys
teabagging
teabagginged
teabagginger
teabagginges
teabagginging
teabaggingly
teabaggings
terd
terded
terder
terdes
terding
terdly
terds
teste
testee
testeed
testeeed
testeeer
testeees
testeeing
testeely
testeer
testees
testeing
testely
testes
testesed
testeser
testeses
testesing
testesly
testess
testicle
testicleed
testicleer
testiclees
testicleing
testiclely
testicles
testis
testised
testiser
testises
testising
testisly
testiss
thrusted
thruster
thrustes
thrusting
thrustly
thrusts
thug
thuged
thuger
thuges
thuging
thugly
thugs
tinkle
tinkleed
tinkleer
tinklees
tinkleing
tinklely
tinkles
tit
tited
titer
tites
titfuck
titfucked
titfucker
titfuckes
titfucking
titfuckly
titfucks
titi
titied
titier
tities
titiing
titily
titing
titis
titly
tits
titsed
titser
titses
titsing
titsly
titss
tittiefucker
tittiefuckered
tittiefuckerer
tittiefuckeres
tittiefuckering
tittiefuckerly
tittiefuckers
titties
tittiesed
tittieser
tittieses
tittiesing
tittiesly
tittiess
titty
tittyed
tittyer
tittyes
tittyfuck
tittyfucked
tittyfucker
tittyfuckered
tittyfuckerer
tittyfuckeres
tittyfuckering
tittyfuckerly
tittyfuckers
tittyfuckes
tittyfucking
tittyfuckly
tittyfucks
tittying
tittyly
tittys
toke
tokeed
tokeer
tokees
tokeing
tokely
tokes
toots
tootsed
tootser
tootses
tootsing
tootsly
tootss
tramp
tramped
tramper
trampes
tramping
tramply
tramps
transsexualed
transsexualer
transsexuales
transsexualing
transsexually
transsexuals
trashy
trashyed
trashyer
trashyes
trashying
trashyly
trashys
tubgirl
tubgirled
tubgirler
tubgirles
tubgirling
tubgirlly
tubgirls
turd
turded
turder
turdes
turding
turdly
turds
tush
tushed
tusher
tushes
tushing
tushly
tushs
twat
twated
twater
twates
twating
twatly
twats
twatsed
twatser
twatses
twatsing
twatsly
twatss
undies
undiesed
undieser
undieses
undiesing
undiesly
undiess
unweded
unweder
unwedes
unweding
unwedly
unweds
uzi
uzied
uzier
uzies
uziing
uzily
uzis
vag
vaged
vager
vages
vaging
vagly
vags
valium
valiumed
valiumer
valiumes
valiuming
valiumly
valiums
venous
virgined
virginer
virgines
virgining
virginly
virgins
vixen
vixened
vixener
vixenes
vixening
vixenly
vixens
vodkaed
vodkaer
vodkaes
vodkaing
vodkaly
vodkas
voyeur
voyeured
voyeurer
voyeures
voyeuring
voyeurly
voyeurs
vulgar
vulgared
vulgarer
vulgares
vulgaring
vulgarly
vulgars
wang
wanged
wanger
wanges
wanging
wangly
wangs
wank
wanked
wanker
wankered
wankerer
wankeres
wankering
wankerly
wankers
wankes
wanking
wankly
wanks
wazoo
wazooed
wazooer
wazooes
wazooing
wazooly
wazoos
wedgie
wedgieed
wedgieer
wedgiees
wedgieing
wedgiely
wedgies
weeded
weeder
weedes
weeding
weedly
weeds
weenie
weenieed
weenieer
weeniees
weenieing
weeniely
weenies
weewee
weeweeed
weeweeer
weeweees
weeweeing
weeweely
weewees
weiner
weinered
weinerer
weineres
weinering
weinerly
weiners
weirdo
weirdoed
weirdoer
weirdoes
weirdoing
weirdoly
weirdos
wench
wenched
wencher
wenches
wenching
wenchly
wenchs
wetback
wetbacked
wetbacker
wetbackes
wetbacking
wetbackly
wetbacks
whitey
whiteyed
whiteyer
whiteyes
whiteying
whiteyly
whiteys
whiz
whized
whizer
whizes
whizing
whizly
whizs
whoralicious
whoralicioused
whoraliciouser
whoraliciouses
whoraliciousing
whoraliciously
whoraliciouss
whore
whorealicious
whorealicioused
whorealiciouser
whorealiciouses
whorealiciousing
whorealiciously
whorealiciouss
whored
whoreded
whoreder
whoredes
whoreding
whoredly
whoreds
whoreed
whoreer
whorees
whoreface
whorefaceed
whorefaceer
whorefacees
whorefaceing
whorefacely
whorefaces
whorehopper
whorehoppered
whorehopperer
whorehopperes
whorehoppering
whorehopperly
whorehoppers
whorehouse
whorehouseed
whorehouseer
whorehousees
whorehouseing
whorehousely
whorehouses
whoreing
whorely
whores
whoresed
whoreser
whoreses
whoresing
whoresly
whoress
whoring
whoringed
whoringer
whoringes
whoringing
whoringly
whorings
wigger
wiggered
wiggerer
wiggeres
wiggering
wiggerly
wiggers
woody
woodyed
woodyer
woodyes
woodying
woodyly
woodys
wop
woped
woper
wopes
woping
woply
wops
wtf
wtfed
wtfer
wtfes
wtfing
wtfly
wtfs
xxx
xxxed
xxxer
xxxes
xxxing
xxxly
xxxs
yeasty
yeastyed
yeastyer
yeastyes
yeastying
yeastyly
yeastys
yobbo
yobboed
yobboer
yobboes
yobboing
yobboly
yobbos
zoophile
zoophileed
zoophileer
zoophilees
zoophileing
zoophilely
zoophiles
anal
ass
ass lick
balls
ballsac
bisexual
bleach
causas
cheap
cost of miracles
cunt
display network stats
fart
fda and death
fda AND warn
fda AND warning
fda AND warns
feom
fuck
gfc
humira AND expensive
illegal
madvocate
masturbation
nuccitelli
overdose
porn
shit
snort
texarkana
Biologics may protect psoriasis patients against severe COVID-19
presented at the virtual annual congress of the European Academy of Dermatology and Venereology.
“Biologics seem to be very protective against severe, poor-prognosis COVID-19, but they do not prevent infection with the virus,” reported Giovanni Damiani, MD, a dermatologist at the University of Milan.
This apparent protective effect of biologic agents against severe and even fatal COVID-19 is all the more impressive because the psoriasis patients included in the Italian study – as is true of those elsewhere throughout the world – had relatively high rates of obesity, smoking, and chronic obstructive pulmonary disease, known risk factors for severe COVID-19, he added.
He presented a case-control study including 1,193 adult psoriasis patients on biologics or apremilast (Otezla) at Milan’s San Donato Hospital during the period from Feb. 21 to April 9, 2020. The control group comprised more than 10 million individuals, the entire adult population of the Lombardy region, of which Milan is the capital. This was the hardest-hit area in all of Italy during the first wave of COVID-19.
Twenty-two of the 1,193 psoriasis patients experienced confirmed COVID-19 during the study period. Seventeen were quarantined at home because their disease was mild. Five were hospitalized. But no psoriasis patients were placed in intensive care, and none died.
Psoriasis patients on biologics were significantly more likely than the general Lombardian population to test positive for COVID-19, with an unadjusted odds ratio of 3.43. They were at 9.05-fold increased risk of home quarantine for mild disease, and at 3.59-fold greater risk than controls for hospitalization for COVID-19. However, they were not at significantly increased risk of ICU admission. And while they actually had a 59% relative risk reduction for death, this didn’t achieve statistical significance.
Forty-five percent of the psoriasis patients were on an interleukin-17 (IL-17) inhibitor, 22% were on a tumor necrosis factor–alpha inhibitor, and 20% were taking an IL-12/23 inhibitor. Of note, none of 77 patients on apremilast developed COVID-19, even though it is widely considered a less potent psoriasis therapy than the injectable monoclonal antibody biologics.
The French experience
Anne-Claire Fougerousse, MD, and her French coinvestigators conducted a study designed to address a different question: Is it safe to start psoriasis patients on biologics or older conventional systemic agents such as methotrexate during the pandemic?
She presented a French national cross-sectional study of 1,418 adult psoriasis patients on a biologic or standard systemic therapy during a snapshot in time near the peak of the first wave of the pandemic in France: the period from April 27 to May 7, 2020. The group included 1,188 psoriasis patients on maintenance therapy and 230 who had initiated systemic treatment within the past 4 months. More than one-third of the patients had at least one risk factor for severe COVID-19.
Although testing wasn’t available to confirm all cases, 54 patients developed probable COVID-19 during the study period. Only five required hospitalization. None died. The two hospitalized psoriasis patients admitted to an ICU had obesity as a risk factor for severe COVID-19, as did another of the five hospitalized patients, reported Dr. Fougerousse, a dermatologist at the Bégin Military Teaching Hospital in Saint-Mandé, France. Hospitalization for COVID-19 was required in 0.43% of the French treatment initiators, not significantly different from the 0.34% rate in patients on maintenance systemic therapy. A study limitation was the lack of a control group.
Nonetheless, the data did answer the investigators’ main question: “This is the first data showing no increased incidence of severe COVID-19 in psoriasis patients receiving systemic therapy in the treatment initiation period compared to those on maintenance therapy. This may now allow physicians to initiate conventional systemic or biologic therapy in patients with severe psoriasis on a case-by-case basis in the context of the persistent COVID-19 pandemic,” Dr. Fougerousse concluded.
Proposed mechanism of benefit
The Italian study findings that biologics boost the risk of infection with the SARS-CoV-2 virus in psoriasis patients while potentially protecting them against ICU admission and death are backed by a biologically plausible albeit as yet unproven mechanism of action, Dr. Damiani asserted.
He elaborated: A vast body of high-quality clinical trials data demonstrates that these targeted immunosuppressive agents are associated with modestly increased risk of viral infections, including both skin and respiratory tract infections. So there is no reason to suppose these agents would offer protection against the first phase of COVID-19, involving SARS-CoV-2 infection, nor protect against the second (pulmonary phase), whose hallmarks are dyspnea with or without hypoxia. But progression to the third phase, involving hyperinflammation and hypercoagulation – dubbed the cytokine storm – could be a different matter.
“Of particular interest was that our patients on IL-17 inhibitors displayed a really great outcome. Interleukin-17 has procoagulant and prothrombotic effects, organizes bronchoalveolar remodeling, has a profibrotic effect, induces mitochondrial dysfunction, and encourages dendritic cell migration in peribronchial lymph nodes. Therefore, by antagonizing this interleukin, we may have a better prognosis, although further studies are needed to be certain,” Dr. Damiani commented.
Publication of his preliminary findings drew the attention of a group of highly respected thought leaders in psoriasis, including James G. Krueger, MD, head of the laboratory for investigative dermatology and codirector of the center for clinical and investigative science at Rockefeller University, New York.
The Italian report prompted them to analyze data from the phase 4, double-blind, randomized ObePso-S study investigating the effects of the IL-17 inhibitor secukinumab (Cosentyx) on systemic inflammatory markers and gene expression in psoriasis patients. The investigators demonstrated that IL-17–mediated inflammation in psoriasis patients was associated with increased expression of the angiotensin-converting enzyme 2 (ACE2) receptor in lesional skin, and that treatment with secukinumab dropped ACE2 expression to levels seen in nonlesional skin. Given that ACE2 is the chief portal of entry for SARS-CoV-2 and that IL-17 exerts systemic proinflammatory effects, it’s plausible that inhibition of IL-17–mediated inflammation via dampening of ACE2 expression in noncutaneous epithelia “could prove to be advantageous in patients with psoriasis who are at risk for SARS-CoV-2 infection,” according to Dr. Krueger and his coinvestigators in the Journal of Allergy and Clinical Immunology.
Dr. Damiani and Dr. Fougerousse reported having no financial conflicts regarding their studies. The secukinumab/ACE2 receptor study was funded by Novartis.
presented at the virtual annual congress of the European Academy of Dermatology and Venereology.
“Biologics seem to be very protective against severe, poor-prognosis COVID-19, but they do not prevent infection with the virus,” reported Giovanni Damiani, MD, a dermatologist at the University of Milan.
This apparent protective effect of biologic agents against severe and even fatal COVID-19 is all the more impressive because the psoriasis patients included in the Italian study – as is true of those elsewhere throughout the world – had relatively high rates of obesity, smoking, and chronic obstructive pulmonary disease, known risk factors for severe COVID-19, he added.
He presented a case-control study including 1,193 adult psoriasis patients on biologics or apremilast (Otezla) at Milan’s San Donato Hospital during the period from Feb. 21 to April 9, 2020. The control group comprised more than 10 million individuals, the entire adult population of the Lombardy region, of which Milan is the capital. This was the hardest-hit area in all of Italy during the first wave of COVID-19.
Twenty-two of the 1,193 psoriasis patients experienced confirmed COVID-19 during the study period. Seventeen were quarantined at home because their disease was mild. Five were hospitalized. But no psoriasis patients were placed in intensive care, and none died.
Psoriasis patients on biologics were significantly more likely than the general Lombardian population to test positive for COVID-19, with an unadjusted odds ratio of 3.43. They were at 9.05-fold increased risk of home quarantine for mild disease, and at 3.59-fold greater risk than controls for hospitalization for COVID-19. However, they were not at significantly increased risk of ICU admission. And while they actually had a 59% relative risk reduction for death, this didn’t achieve statistical significance.
Forty-five percent of the psoriasis patients were on an interleukin-17 (IL-17) inhibitor, 22% were on a tumor necrosis factor–alpha inhibitor, and 20% were taking an IL-12/23 inhibitor. Of note, none of 77 patients on apremilast developed COVID-19, even though it is widely considered a less potent psoriasis therapy than the injectable monoclonal antibody biologics.
The French experience
Anne-Claire Fougerousse, MD, and her French coinvestigators conducted a study designed to address a different question: Is it safe to start psoriasis patients on biologics or older conventional systemic agents such as methotrexate during the pandemic?
She presented a French national cross-sectional study of 1,418 adult psoriasis patients on a biologic or standard systemic therapy during a snapshot in time near the peak of the first wave of the pandemic in France: the period from April 27 to May 7, 2020. The group included 1,188 psoriasis patients on maintenance therapy and 230 who had initiated systemic treatment within the past 4 months. More than one-third of the patients had at least one risk factor for severe COVID-19.
Although testing wasn’t available to confirm all cases, 54 patients developed probable COVID-19 during the study period. Only five required hospitalization. None died. The two hospitalized psoriasis patients admitted to an ICU had obesity as a risk factor for severe COVID-19, as did another of the five hospitalized patients, reported Dr. Fougerousse, a dermatologist at the Bégin Military Teaching Hospital in Saint-Mandé, France. Hospitalization for COVID-19 was required in 0.43% of the French treatment initiators, not significantly different from the 0.34% rate in patients on maintenance systemic therapy. A study limitation was the lack of a control group.
Nonetheless, the data did answer the investigators’ main question: “This is the first data showing no increased incidence of severe COVID-19 in psoriasis patients receiving systemic therapy in the treatment initiation period compared to those on maintenance therapy. This may now allow physicians to initiate conventional systemic or biologic therapy in patients with severe psoriasis on a case-by-case basis in the context of the persistent COVID-19 pandemic,” Dr. Fougerousse concluded.
Proposed mechanism of benefit
The Italian study findings that biologics boost the risk of infection with the SARS-CoV-2 virus in psoriasis patients while potentially protecting them against ICU admission and death are backed by a biologically plausible albeit as yet unproven mechanism of action, Dr. Damiani asserted.
He elaborated: A vast body of high-quality clinical trials data demonstrates that these targeted immunosuppressive agents are associated with modestly increased risk of viral infections, including both skin and respiratory tract infections. So there is no reason to suppose these agents would offer protection against the first phase of COVID-19, involving SARS-CoV-2 infection, nor protect against the second (pulmonary phase), whose hallmarks are dyspnea with or without hypoxia. But progression to the third phase, involving hyperinflammation and hypercoagulation – dubbed the cytokine storm – could be a different matter.
“Of particular interest was that our patients on IL-17 inhibitors displayed a really great outcome. Interleukin-17 has procoagulant and prothrombotic effects, organizes bronchoalveolar remodeling, has a profibrotic effect, induces mitochondrial dysfunction, and encourages dendritic cell migration in peribronchial lymph nodes. Therefore, by antagonizing this interleukin, we may have a better prognosis, although further studies are needed to be certain,” Dr. Damiani commented.
Publication of his preliminary findings drew the attention of a group of highly respected thought leaders in psoriasis, including James G. Krueger, MD, head of the laboratory for investigative dermatology and codirector of the center for clinical and investigative science at Rockefeller University, New York.
The Italian report prompted them to analyze data from the phase 4, double-blind, randomized ObePso-S study investigating the effects of the IL-17 inhibitor secukinumab (Cosentyx) on systemic inflammatory markers and gene expression in psoriasis patients. The investigators demonstrated that IL-17–mediated inflammation in psoriasis patients was associated with increased expression of the angiotensin-converting enzyme 2 (ACE2) receptor in lesional skin, and that treatment with secukinumab dropped ACE2 expression to levels seen in nonlesional skin. Given that ACE2 is the chief portal of entry for SARS-CoV-2 and that IL-17 exerts systemic proinflammatory effects, it’s plausible that inhibition of IL-17–mediated inflammation via dampening of ACE2 expression in noncutaneous epithelia “could prove to be advantageous in patients with psoriasis who are at risk for SARS-CoV-2 infection,” according to Dr. Krueger and his coinvestigators in the Journal of Allergy and Clinical Immunology.
Dr. Damiani and Dr. Fougerousse reported having no financial conflicts regarding their studies. The secukinumab/ACE2 receptor study was funded by Novartis.
presented at the virtual annual congress of the European Academy of Dermatology and Venereology.
“Biologics seem to be very protective against severe, poor-prognosis COVID-19, but they do not prevent infection with the virus,” reported Giovanni Damiani, MD, a dermatologist at the University of Milan.
This apparent protective effect of biologic agents against severe and even fatal COVID-19 is all the more impressive because the psoriasis patients included in the Italian study – as is true of those elsewhere throughout the world – had relatively high rates of obesity, smoking, and chronic obstructive pulmonary disease, known risk factors for severe COVID-19, he added.
He presented a case-control study including 1,193 adult psoriasis patients on biologics or apremilast (Otezla) at Milan’s San Donato Hospital during the period from Feb. 21 to April 9, 2020. The control group comprised more than 10 million individuals, the entire adult population of the Lombardy region, of which Milan is the capital. This was the hardest-hit area in all of Italy during the first wave of COVID-19.
Twenty-two of the 1,193 psoriasis patients experienced confirmed COVID-19 during the study period. Seventeen were quarantined at home because their disease was mild. Five were hospitalized. But no psoriasis patients were placed in intensive care, and none died.
Psoriasis patients on biologics were significantly more likely than the general Lombardian population to test positive for COVID-19, with an unadjusted odds ratio of 3.43. They were at 9.05-fold increased risk of home quarantine for mild disease, and at 3.59-fold greater risk than controls for hospitalization for COVID-19. However, they were not at significantly increased risk of ICU admission. And while they actually had a 59% relative risk reduction for death, this didn’t achieve statistical significance.
Forty-five percent of the psoriasis patients were on an interleukin-17 (IL-17) inhibitor, 22% were on a tumor necrosis factor–alpha inhibitor, and 20% were taking an IL-12/23 inhibitor. Of note, none of 77 patients on apremilast developed COVID-19, even though it is widely considered a less potent psoriasis therapy than the injectable monoclonal antibody biologics.
The French experience
Anne-Claire Fougerousse, MD, and her French coinvestigators conducted a study designed to address a different question: Is it safe to start psoriasis patients on biologics or older conventional systemic agents such as methotrexate during the pandemic?
She presented a French national cross-sectional study of 1,418 adult psoriasis patients on a biologic or standard systemic therapy during a snapshot in time near the peak of the first wave of the pandemic in France: the period from April 27 to May 7, 2020. The group included 1,188 psoriasis patients on maintenance therapy and 230 who had initiated systemic treatment within the past 4 months. More than one-third of the patients had at least one risk factor for severe COVID-19.
Although testing wasn’t available to confirm all cases, 54 patients developed probable COVID-19 during the study period. Only five required hospitalization. None died. The two hospitalized psoriasis patients admitted to an ICU had obesity as a risk factor for severe COVID-19, as did another of the five hospitalized patients, reported Dr. Fougerousse, a dermatologist at the Bégin Military Teaching Hospital in Saint-Mandé, France. Hospitalization for COVID-19 was required in 0.43% of the French treatment initiators, not significantly different from the 0.34% rate in patients on maintenance systemic therapy. A study limitation was the lack of a control group.
Nonetheless, the data did answer the investigators’ main question: “This is the first data showing no increased incidence of severe COVID-19 in psoriasis patients receiving systemic therapy in the treatment initiation period compared to those on maintenance therapy. This may now allow physicians to initiate conventional systemic or biologic therapy in patients with severe psoriasis on a case-by-case basis in the context of the persistent COVID-19 pandemic,” Dr. Fougerousse concluded.
Proposed mechanism of benefit
The Italian study findings that biologics boost the risk of infection with the SARS-CoV-2 virus in psoriasis patients while potentially protecting them against ICU admission and death are backed by a biologically plausible albeit as yet unproven mechanism of action, Dr. Damiani asserted.
He elaborated: A vast body of high-quality clinical trials data demonstrates that these targeted immunosuppressive agents are associated with modestly increased risk of viral infections, including both skin and respiratory tract infections. So there is no reason to suppose these agents would offer protection against the first phase of COVID-19, involving SARS-CoV-2 infection, nor protect against the second (pulmonary phase), whose hallmarks are dyspnea with or without hypoxia. But progression to the third phase, involving hyperinflammation and hypercoagulation – dubbed the cytokine storm – could be a different matter.
“Of particular interest was that our patients on IL-17 inhibitors displayed a really great outcome. Interleukin-17 has procoagulant and prothrombotic effects, organizes bronchoalveolar remodeling, has a profibrotic effect, induces mitochondrial dysfunction, and encourages dendritic cell migration in peribronchial lymph nodes. Therefore, by antagonizing this interleukin, we may have a better prognosis, although further studies are needed to be certain,” Dr. Damiani commented.
Publication of his preliminary findings drew the attention of a group of highly respected thought leaders in psoriasis, including James G. Krueger, MD, head of the laboratory for investigative dermatology and codirector of the center for clinical and investigative science at Rockefeller University, New York.
The Italian report prompted them to analyze data from the phase 4, double-blind, randomized ObePso-S study investigating the effects of the IL-17 inhibitor secukinumab (Cosentyx) on systemic inflammatory markers and gene expression in psoriasis patients. The investigators demonstrated that IL-17–mediated inflammation in psoriasis patients was associated with increased expression of the angiotensin-converting enzyme 2 (ACE2) receptor in lesional skin, and that treatment with secukinumab dropped ACE2 expression to levels seen in nonlesional skin. Given that ACE2 is the chief portal of entry for SARS-CoV-2 and that IL-17 exerts systemic proinflammatory effects, it’s plausible that inhibition of IL-17–mediated inflammation via dampening of ACE2 expression in noncutaneous epithelia “could prove to be advantageous in patients with psoriasis who are at risk for SARS-CoV-2 infection,” according to Dr. Krueger and his coinvestigators in the Journal of Allergy and Clinical Immunology.
Dr. Damiani and Dr. Fougerousse reported having no financial conflicts regarding their studies. The secukinumab/ACE2 receptor study was funded by Novartis.
FROM THE EADV CONGRESS
Health sector has spent $464 million on lobbying in 2020
, according to the Center for Responsive Politics.
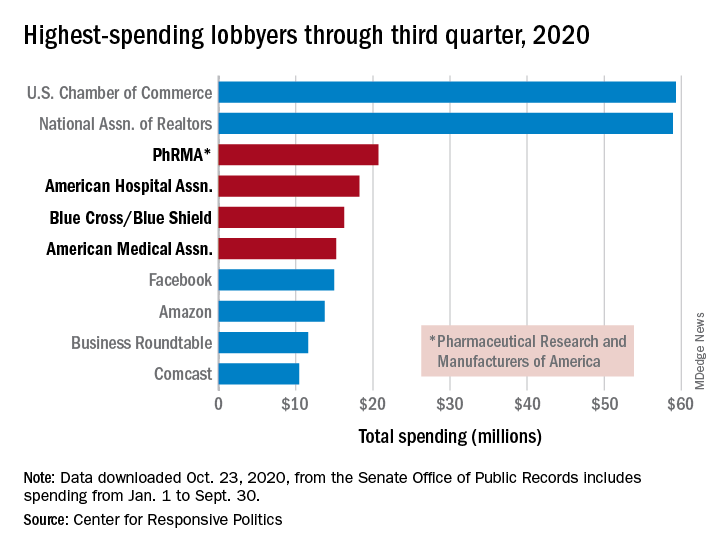
PhRMA spent $20.7 million on lobbying through the end of September, good enough for third on the overall list of U.S. companies and organizations. Three other members of the health sector made the top 10: the American Hospital Association ($18.3 million), BlueCross/BlueShield ($16.3 million), and the American Medical Association ($15.2 million), the center reported.
Total spending by the health sector was $464 million from Jan. 1 to Sept. 30, topping the finance/insurance/real estate sector at $403 million, and miscellaneous business at $371 million. Miscellaneous business is the home of the U.S. Chamber of Commerce, the annual leader in such spending for the last 20 years, based on data from the Senate Office of Public Records.
The largest share of health sector spending came from pharmaceuticals/health products, with a total of almost $233 million, just slightly more than the sector’s four other constituents combined: hospitals/nursing homes ($80 million), health services/HMOs ($75 million), health professionals ($67 million), and miscellaneous health ($9.5 million), the center said on OpenSecrets.org.
Taking one step down from the sector level, that $233 million made pharmaceuticals/health products the highest spending of about 100 industries in 2020, nearly doubling the efforts of electronics manufacturing and equipment ($118 million), which came a distant second. Hospitals/nursing homes was eighth on the industry list, the center noted.
, according to the Center for Responsive Politics.

PhRMA spent $20.7 million on lobbying through the end of September, good enough for third on the overall list of U.S. companies and organizations. Three other members of the health sector made the top 10: the American Hospital Association ($18.3 million), BlueCross/BlueShield ($16.3 million), and the American Medical Association ($15.2 million), the center reported.
Total spending by the health sector was $464 million from Jan. 1 to Sept. 30, topping the finance/insurance/real estate sector at $403 million, and miscellaneous business at $371 million. Miscellaneous business is the home of the U.S. Chamber of Commerce, the annual leader in such spending for the last 20 years, based on data from the Senate Office of Public Records.
The largest share of health sector spending came from pharmaceuticals/health products, with a total of almost $233 million, just slightly more than the sector’s four other constituents combined: hospitals/nursing homes ($80 million), health services/HMOs ($75 million), health professionals ($67 million), and miscellaneous health ($9.5 million), the center said on OpenSecrets.org.
Taking one step down from the sector level, that $233 million made pharmaceuticals/health products the highest spending of about 100 industries in 2020, nearly doubling the efforts of electronics manufacturing and equipment ($118 million), which came a distant second. Hospitals/nursing homes was eighth on the industry list, the center noted.
, according to the Center for Responsive Politics.

PhRMA spent $20.7 million on lobbying through the end of September, good enough for third on the overall list of U.S. companies and organizations. Three other members of the health sector made the top 10: the American Hospital Association ($18.3 million), BlueCross/BlueShield ($16.3 million), and the American Medical Association ($15.2 million), the center reported.
Total spending by the health sector was $464 million from Jan. 1 to Sept. 30, topping the finance/insurance/real estate sector at $403 million, and miscellaneous business at $371 million. Miscellaneous business is the home of the U.S. Chamber of Commerce, the annual leader in such spending for the last 20 years, based on data from the Senate Office of Public Records.
The largest share of health sector spending came from pharmaceuticals/health products, with a total of almost $233 million, just slightly more than the sector’s four other constituents combined: hospitals/nursing homes ($80 million), health services/HMOs ($75 million), health professionals ($67 million), and miscellaneous health ($9.5 million), the center said on OpenSecrets.org.
Taking one step down from the sector level, that $233 million made pharmaceuticals/health products the highest spending of about 100 industries in 2020, nearly doubling the efforts of electronics manufacturing and equipment ($118 million), which came a distant second. Hospitals/nursing homes was eighth on the industry list, the center noted.
New technologies aim to improve ovarian cancer detection
Encouraging trends abound in the management of ovarian cancer. As rates of ovarian disease continue to decline, there has also been a notable increase in tools for detecting it earlier in its course.
To better understand these developments, this news organization reached out to Rebecca Stone, MD, an ovarian cancer expert and associate professor of gynecologic oncology at Johns Hopkins University, in Baltimore, Maryland. This interview has been edited for length and clarity.
There has been a decline in the rates of ovarian cancer in recent years. What are the possible causes of this?Dr. Stone: The number of new cases in the United States has actually been declining over the past 2 decades. This is thought to be attributable to the increased prescribing of oral contraceptive pills in the late 1990s and the uptake of preventive measures, such as risk-reducing gynecologic surgery for women with genetic predisposition to ovarian cancer, as well as opportunistic salpingectomy in the general population. Opportunistic salpingectomy was introduced about 10 years ago. It is a surgical means for primary prevention of tubo-ovarian cancer by removing both fallopian tubes at the time of elective surgery for women who have completed childbearing or in lieu of “tying the tubes” for women who desire permanent surgical sterility.
What can you tell us about a recent study suggesting that high-grade serous epithelial ovarian cancer may be detected earlier in the course of the disease by testing for TP53 clonal variants in DNA from Papanicolaou (Pap) tests performed during cervical cancer screening?
The idea here is that early mutational events that ultimately result in the development of epithelial ovarian cancer can be detected by performing gene sequencing on genetic material collected at the time of routine Pap smear screening done for cervical cancer. Pap tests are known to contain cells and genetic material shed from the fallopian tubes, where the precancerous lesions thought to give rise to epithelial ovarian cancer, predominantly serous epithelial ovarian cancers, start.
p53 gene mutations are thought to occur early in the evolution of ovarian cancer. There are data indicating that these mutations actually occur in cells lining the fallopian tubes. Polymerase chain reaction–based DNA/gene sequencing performed on cervical fluid collected by Pap smears could detect these p53-mutated cells shed from the fallopian tubes.
A strength of this study is that it included healthy controls. None of their Pap smears screened positive for the p53 mutations, unlike the Pap smears of women predating their diagnosis of ovarian cancer. Limitations of the study include the fact that it had a small sample size. Findings will need to be confirmed in a larger patient population.
Also, the study only looked for p53 gene mutations. Ovarian cancers, like other cancers, are largely thought to occur when there is a buildup of mutations in critical genes that result in uncontrolled cell growth and division. These genetic changes/mutations are acquired during a person’s lifetime. Thus, there are likely early genetic changes/mutations that occur in addition to p53 mutations that ultimately lead to the development of ovarian cancer. Detecting these along with p53 mutations could improve the sensitivity/detection rate of the screening strategy that the authors are investigating.
Finally, this screening strategy may not prove effective for the early detection of all histologic subtypes of epithelial ovarian cancer or for nonepithelial ovarian cancers.
What other recent developments in the diagnosis of ovarian cancer should clinicians be aware of?
Liquid biopsies using circulating tumor DNA (ctDNA) have shown promising results for cancer detection and management, including ovarian cancer. However, further clarification is needed to define the minimum tumor size/burden detectable using ctDNA-based approaches. Moreover, large prospective studies are needed to determine the clinical utility of ctDNA detection for early diagnosis of ovarian cancer and its impact on patient outcomes.
DNA methylation is an early event in carcinogenesis and can be detected in blood plasma samples from cancer patients. Data related to the discovery and validation of discriminated methylated DNA marker candidates extracted from ovarian cancer tissues were presented at the American Society of Clinical Oncology meeting this year. Findings were subsequently evaluated in plasma from women with and without ovarian cancer.
In addition to blood, peritoneal fluid and uterine lavage have been used to obtain cell pellets that are used for the identification of common mutant genes – TP53, BRCA1, and BRCA2. These body fluids have also been shown as the source of tumor-derived material that can be used to differentiate between ovarian cancer patients and healthy individuals.
Further studies are needed to determine the sensitivity and specificity of other noninvasive tests for the diagnosis of ovarian cancer.
The American Cancer Society issued a statement that the human papillomavirus (HPV) test is the preferred cervical cancer screening tool. Why do they prefer the HPV test over the Pap test?
The American Cancer Society recommends that cervical cancer testing (screening) begin at age 25 years. Women aged 25-65 years should have a primary HPV test every 5 years. If primary HPV testing is not available, screening may be done with either a co-test that combines an HPV test with a Pap test every 5 years or a Pap test alone every 3 years.
The HPV test is widely available. The cost of an HPV test is approximately $44 (unit cost, 2014 USD). The cost of a Pap test is approximately $30 (unit cost, 2014 USD).
The HPV test is preferred over cytologic testing (Pap) for several reasons.
Firstly, in well-designed studies, the sensitivity of a single Pap smear for detecting high-grade precancer of the cervix is around 50%, which is less than optimal for a cancer screening test. Sensitivity means the chance that, if you have the disease (in this case, high-grade precancer of the cervix), the test will detect it. In particular, cytology is known to have an even more limited ability to detect glandular precancers, which arise in the endocervical canal rather than on or in close proximity to the exterior surface of the cervix (ectocervix). Thus, HPV-based screening programs hold the promise of improving detection of cervical adenocarcinoma.
Secondly, to function reliably, cytology programs require substantial infrastructure, highly qualified human resources, and a well‐defined quality-control system, which have proved to be costly and difficult to implement. This results in global disparities in cytology-based cervical cancer screening programs.
Thirdly, although co‐testing with both cytology and HPV tests is an option for screening programs, studies have confirmed that there is limited benefit from adding cytology to HPV screening. Long‐term studies from Kaiser Permanente that included over 1 million women found that HPV testing has a very high negative predictive value for precancerous lesions. Women with negative HPV tests were very unlikely to develop precancerous lesions in the following 5 years. The 5‐year risk of high-grade precancer or cancer of the cervix following a negative HPV test was 0.14%, whereas for women with a negative cytology, it was 0.31%. The screening benefit of co‐testing is largely driven by HPV testing and not cytology.
So, in summary, HPV testing is preferred over cytologic screening for cervical cancer, given its improved sensitivity and quality assurance, the opportunity to automate testing, and ultimately, its prospect of reducing the overall number of lifetime screenings for women.
Dr. Stone has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Encouraging trends abound in the management of ovarian cancer. As rates of ovarian disease continue to decline, there has also been a notable increase in tools for detecting it earlier in its course.
To better understand these developments, this news organization reached out to Rebecca Stone, MD, an ovarian cancer expert and associate professor of gynecologic oncology at Johns Hopkins University, in Baltimore, Maryland. This interview has been edited for length and clarity.
There has been a decline in the rates of ovarian cancer in recent years. What are the possible causes of this?Dr. Stone: The number of new cases in the United States has actually been declining over the past 2 decades. This is thought to be attributable to the increased prescribing of oral contraceptive pills in the late 1990s and the uptake of preventive measures, such as risk-reducing gynecologic surgery for women with genetic predisposition to ovarian cancer, as well as opportunistic salpingectomy in the general population. Opportunistic salpingectomy was introduced about 10 years ago. It is a surgical means for primary prevention of tubo-ovarian cancer by removing both fallopian tubes at the time of elective surgery for women who have completed childbearing or in lieu of “tying the tubes” for women who desire permanent surgical sterility.
What can you tell us about a recent study suggesting that high-grade serous epithelial ovarian cancer may be detected earlier in the course of the disease by testing for TP53 clonal variants in DNA from Papanicolaou (Pap) tests performed during cervical cancer screening?
The idea here is that early mutational events that ultimately result in the development of epithelial ovarian cancer can be detected by performing gene sequencing on genetic material collected at the time of routine Pap smear screening done for cervical cancer. Pap tests are known to contain cells and genetic material shed from the fallopian tubes, where the precancerous lesions thought to give rise to epithelial ovarian cancer, predominantly serous epithelial ovarian cancers, start.
p53 gene mutations are thought to occur early in the evolution of ovarian cancer. There are data indicating that these mutations actually occur in cells lining the fallopian tubes. Polymerase chain reaction–based DNA/gene sequencing performed on cervical fluid collected by Pap smears could detect these p53-mutated cells shed from the fallopian tubes.
A strength of this study is that it included healthy controls. None of their Pap smears screened positive for the p53 mutations, unlike the Pap smears of women predating their diagnosis of ovarian cancer. Limitations of the study include the fact that it had a small sample size. Findings will need to be confirmed in a larger patient population.
Also, the study only looked for p53 gene mutations. Ovarian cancers, like other cancers, are largely thought to occur when there is a buildup of mutations in critical genes that result in uncontrolled cell growth and division. These genetic changes/mutations are acquired during a person’s lifetime. Thus, there are likely early genetic changes/mutations that occur in addition to p53 mutations that ultimately lead to the development of ovarian cancer. Detecting these along with p53 mutations could improve the sensitivity/detection rate of the screening strategy that the authors are investigating.
Finally, this screening strategy may not prove effective for the early detection of all histologic subtypes of epithelial ovarian cancer or for nonepithelial ovarian cancers.
What other recent developments in the diagnosis of ovarian cancer should clinicians be aware of?
Liquid biopsies using circulating tumor DNA (ctDNA) have shown promising results for cancer detection and management, including ovarian cancer. However, further clarification is needed to define the minimum tumor size/burden detectable using ctDNA-based approaches. Moreover, large prospective studies are needed to determine the clinical utility of ctDNA detection for early diagnosis of ovarian cancer and its impact on patient outcomes.
DNA methylation is an early event in carcinogenesis and can be detected in blood plasma samples from cancer patients. Data related to the discovery and validation of discriminated methylated DNA marker candidates extracted from ovarian cancer tissues were presented at the American Society of Clinical Oncology meeting this year. Findings were subsequently evaluated in plasma from women with and without ovarian cancer.
In addition to blood, peritoneal fluid and uterine lavage have been used to obtain cell pellets that are used for the identification of common mutant genes – TP53, BRCA1, and BRCA2. These body fluids have also been shown as the source of tumor-derived material that can be used to differentiate between ovarian cancer patients and healthy individuals.
Further studies are needed to determine the sensitivity and specificity of other noninvasive tests for the diagnosis of ovarian cancer.
The American Cancer Society issued a statement that the human papillomavirus (HPV) test is the preferred cervical cancer screening tool. Why do they prefer the HPV test over the Pap test?
The American Cancer Society recommends that cervical cancer testing (screening) begin at age 25 years. Women aged 25-65 years should have a primary HPV test every 5 years. If primary HPV testing is not available, screening may be done with either a co-test that combines an HPV test with a Pap test every 5 years or a Pap test alone every 3 years.
The HPV test is widely available. The cost of an HPV test is approximately $44 (unit cost, 2014 USD). The cost of a Pap test is approximately $30 (unit cost, 2014 USD).
The HPV test is preferred over cytologic testing (Pap) for several reasons.
Firstly, in well-designed studies, the sensitivity of a single Pap smear for detecting high-grade precancer of the cervix is around 50%, which is less than optimal for a cancer screening test. Sensitivity means the chance that, if you have the disease (in this case, high-grade precancer of the cervix), the test will detect it. In particular, cytology is known to have an even more limited ability to detect glandular precancers, which arise in the endocervical canal rather than on or in close proximity to the exterior surface of the cervix (ectocervix). Thus, HPV-based screening programs hold the promise of improving detection of cervical adenocarcinoma.
Secondly, to function reliably, cytology programs require substantial infrastructure, highly qualified human resources, and a well‐defined quality-control system, which have proved to be costly and difficult to implement. This results in global disparities in cytology-based cervical cancer screening programs.
Thirdly, although co‐testing with both cytology and HPV tests is an option for screening programs, studies have confirmed that there is limited benefit from adding cytology to HPV screening. Long‐term studies from Kaiser Permanente that included over 1 million women found that HPV testing has a very high negative predictive value for precancerous lesions. Women with negative HPV tests were very unlikely to develop precancerous lesions in the following 5 years. The 5‐year risk of high-grade precancer or cancer of the cervix following a negative HPV test was 0.14%, whereas for women with a negative cytology, it was 0.31%. The screening benefit of co‐testing is largely driven by HPV testing and not cytology.
So, in summary, HPV testing is preferred over cytologic screening for cervical cancer, given its improved sensitivity and quality assurance, the opportunity to automate testing, and ultimately, its prospect of reducing the overall number of lifetime screenings for women.
Dr. Stone has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Encouraging trends abound in the management of ovarian cancer. As rates of ovarian disease continue to decline, there has also been a notable increase in tools for detecting it earlier in its course.
To better understand these developments, this news organization reached out to Rebecca Stone, MD, an ovarian cancer expert and associate professor of gynecologic oncology at Johns Hopkins University, in Baltimore, Maryland. This interview has been edited for length and clarity.
There has been a decline in the rates of ovarian cancer in recent years. What are the possible causes of this?Dr. Stone: The number of new cases in the United States has actually been declining over the past 2 decades. This is thought to be attributable to the increased prescribing of oral contraceptive pills in the late 1990s and the uptake of preventive measures, such as risk-reducing gynecologic surgery for women with genetic predisposition to ovarian cancer, as well as opportunistic salpingectomy in the general population. Opportunistic salpingectomy was introduced about 10 years ago. It is a surgical means for primary prevention of tubo-ovarian cancer by removing both fallopian tubes at the time of elective surgery for women who have completed childbearing or in lieu of “tying the tubes” for women who desire permanent surgical sterility.
What can you tell us about a recent study suggesting that high-grade serous epithelial ovarian cancer may be detected earlier in the course of the disease by testing for TP53 clonal variants in DNA from Papanicolaou (Pap) tests performed during cervical cancer screening?
The idea here is that early mutational events that ultimately result in the development of epithelial ovarian cancer can be detected by performing gene sequencing on genetic material collected at the time of routine Pap smear screening done for cervical cancer. Pap tests are known to contain cells and genetic material shed from the fallopian tubes, where the precancerous lesions thought to give rise to epithelial ovarian cancer, predominantly serous epithelial ovarian cancers, start.
p53 gene mutations are thought to occur early in the evolution of ovarian cancer. There are data indicating that these mutations actually occur in cells lining the fallopian tubes. Polymerase chain reaction–based DNA/gene sequencing performed on cervical fluid collected by Pap smears could detect these p53-mutated cells shed from the fallopian tubes.
A strength of this study is that it included healthy controls. None of their Pap smears screened positive for the p53 mutations, unlike the Pap smears of women predating their diagnosis of ovarian cancer. Limitations of the study include the fact that it had a small sample size. Findings will need to be confirmed in a larger patient population.
Also, the study only looked for p53 gene mutations. Ovarian cancers, like other cancers, are largely thought to occur when there is a buildup of mutations in critical genes that result in uncontrolled cell growth and division. These genetic changes/mutations are acquired during a person’s lifetime. Thus, there are likely early genetic changes/mutations that occur in addition to p53 mutations that ultimately lead to the development of ovarian cancer. Detecting these along with p53 mutations could improve the sensitivity/detection rate of the screening strategy that the authors are investigating.
Finally, this screening strategy may not prove effective for the early detection of all histologic subtypes of epithelial ovarian cancer or for nonepithelial ovarian cancers.
What other recent developments in the diagnosis of ovarian cancer should clinicians be aware of?
Liquid biopsies using circulating tumor DNA (ctDNA) have shown promising results for cancer detection and management, including ovarian cancer. However, further clarification is needed to define the minimum tumor size/burden detectable using ctDNA-based approaches. Moreover, large prospective studies are needed to determine the clinical utility of ctDNA detection for early diagnosis of ovarian cancer and its impact on patient outcomes.
DNA methylation is an early event in carcinogenesis and can be detected in blood plasma samples from cancer patients. Data related to the discovery and validation of discriminated methylated DNA marker candidates extracted from ovarian cancer tissues were presented at the American Society of Clinical Oncology meeting this year. Findings were subsequently evaluated in plasma from women with and without ovarian cancer.
In addition to blood, peritoneal fluid and uterine lavage have been used to obtain cell pellets that are used for the identification of common mutant genes – TP53, BRCA1, and BRCA2. These body fluids have also been shown as the source of tumor-derived material that can be used to differentiate between ovarian cancer patients and healthy individuals.
Further studies are needed to determine the sensitivity and specificity of other noninvasive tests for the diagnosis of ovarian cancer.
The American Cancer Society issued a statement that the human papillomavirus (HPV) test is the preferred cervical cancer screening tool. Why do they prefer the HPV test over the Pap test?
The American Cancer Society recommends that cervical cancer testing (screening) begin at age 25 years. Women aged 25-65 years should have a primary HPV test every 5 years. If primary HPV testing is not available, screening may be done with either a co-test that combines an HPV test with a Pap test every 5 years or a Pap test alone every 3 years.
The HPV test is widely available. The cost of an HPV test is approximately $44 (unit cost, 2014 USD). The cost of a Pap test is approximately $30 (unit cost, 2014 USD).
The HPV test is preferred over cytologic testing (Pap) for several reasons.
Firstly, in well-designed studies, the sensitivity of a single Pap smear for detecting high-grade precancer of the cervix is around 50%, which is less than optimal for a cancer screening test. Sensitivity means the chance that, if you have the disease (in this case, high-grade precancer of the cervix), the test will detect it. In particular, cytology is known to have an even more limited ability to detect glandular precancers, which arise in the endocervical canal rather than on or in close proximity to the exterior surface of the cervix (ectocervix). Thus, HPV-based screening programs hold the promise of improving detection of cervical adenocarcinoma.
Secondly, to function reliably, cytology programs require substantial infrastructure, highly qualified human resources, and a well‐defined quality-control system, which have proved to be costly and difficult to implement. This results in global disparities in cytology-based cervical cancer screening programs.
Thirdly, although co‐testing with both cytology and HPV tests is an option for screening programs, studies have confirmed that there is limited benefit from adding cytology to HPV screening. Long‐term studies from Kaiser Permanente that included over 1 million women found that HPV testing has a very high negative predictive value for precancerous lesions. Women with negative HPV tests were very unlikely to develop precancerous lesions in the following 5 years. The 5‐year risk of high-grade precancer or cancer of the cervix following a negative HPV test was 0.14%, whereas for women with a negative cytology, it was 0.31%. The screening benefit of co‐testing is largely driven by HPV testing and not cytology.
So, in summary, HPV testing is preferred over cytologic screening for cervical cancer, given its improved sensitivity and quality assurance, the opportunity to automate testing, and ultimately, its prospect of reducing the overall number of lifetime screenings for women.
Dr. Stone has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Med student’s cardiac crisis a COVID-era medical mystery
Within minutes of her arrival at Community North Hospital in Indianapolis, Ramya Yeleti’s vital signs plummeted; her pulse was at 45 beats per minute and her ejection fraction was hovering near 10%. “I definitely thought there was a chance I would close my eyes and never open them again, but I only had a few seconds to process that,” she recalled. Then everything went black. Ramya fell unconscious as shock pads were positioned and a swarm of clinicians prepared to insert an Impella heart pump through a catheter into her aorta.
The third-year medical student and aspiring psychiatrist had been doing in-person neurology rotations in July when she began to experience fever and uncontrolled vomiting. Her initial thought was that she must have caught the flu from a patient.
After all, Ramya, along with her father Ram Yeleti, MD, mother Indira, and twin sister Divya, had all weathered COVID-19 in previous months and later tested positive for SARS-CoV-2 antibodies. The only family member who had been spared was her younger brother Rohith.
Indira suffered a severe case, requiring ICU care for 2 days but no ventilator; the others experienced mostly mild symptoms. Ramya — who was studying for her third-year board exams after classes at Marian University College of Osteopathic Medicine in Indianapolis went virtual in March — was left with lingering fatigue; however, her cough and muscle aches abated and her sense of taste and smell returned. When she started rotations, she thought her life was getting back to normal.
Ramya’s flu symptoms did not improve. A university-mandated rapid COVID test came back negative, but 2 more days of vomiting started to worry both her and her father, who is a cardiologist and chief physician executive at Community Health Network in Indianapolis. After Ramya felt some chest pain, she asked her father to listen to her heart. All sounded normal, and Ram prescribed ondansetron for her nausea.
But the antiemetic didn’t work, and by the next morning both father and daughter were convinced that they needed to head to the emergency department.
“I wanted to double-check if I was missing something about her being dehydrated,” Ram told Medscape Medical News. “Several things can cause protracted nausea, like hepatitis, appendicitis, or another infection. I feel terribly guilty I didn’t realize she had a heart condition.”
A surprising turn for the worst
Ramya’s subtle symptoms quickly gave way to the dramatic cardiac crisis that unfolded just after her arrival at Community North. “Her EKG looked absolutely horrendous, like a 75-year-old having a heart attack,” Ram said.
As a cardiologist, he knew his daughter’s situation was growing dire when he heard physicians shouting that the Impella wasn’t working and she needed extracorporeal membrane oxygenation (ECMO).
“At that point, I didn’t think she’d survive,” her father recalled. “We had 10 physicians in the room who worked on her for 5 hours to get her stabilized.”
“It was especially traumatic because, obviously, I knew exactly what was happening,” he added. “You can’t sugarcoat anything.”
After being connected to the heart–lung equipment, Ramya was transferred to IU Health Methodist Hospital, also in Indianapolis, where she was tested again for COVID-19. Unlike the rapid test administered just days earlier, the PCR assay came back positive.
“I knew she had acute myocarditis, but coronavirus never crossed my mind,” said Ram.
“As we were dealing with her heart, we were also dealing with this challenge: she was coming back positive for COVID-19 again,” said Roopa Rao, MD, the heart failure transplant cardiologist at IU Health who treated Ramya.
“We weren’t sure whether we were dealing with an active infection or dead virus” from her previous infection, Rao said, “so we started treating her like she had active COVID-19 and gave her remdesivir, convalescent plasma, and steroids, which was the protocol in our hospital.”
A biopsy of Ramya’s heart tissue, along with blood tests, indicated a past parvovirus infection. It’s possible that Ramya’s previous coronavirus infection made her susceptible to heart damage from a newer parvovirus infection, said Rao. Either virus, or both together, could have been responsible for the calamity.
Although it was unheard of during Ramya’s cardiac crisis in early August, evolving evidence now raises the possibility that she is one of a handful of people in the world to be reinfected with SARS-CoV-2. Also emerging are cases of COVID-related myocarditis and other extreme heart complications, particularly in young people.
“At the time, it wasn’t really clear if people could have another infection so quickly,” Rao told Medscape Medical News. “It is possible she is one of these rare individuals to have COVID-19 twice. I’m hoping at some point we will have some clarity.”
“I would favor a coinfection as probably the triggering factor for her sickness,” she said. “It may take some time, but like any other disease — and it doesn’t look like COVID will go away magically — I hope we’ll have some answers down the road.”
Another wrinkle
The next 48 hours brought astonishing news: Ramya’s heart function had rebounded to nearly normal, and her ejection fraction increased to about 45%. Heart transplantation wouldn’t be necessary, although Rao stood poised to follow through if ECMO only sustained, rather than improved, Ramya’s prognosis.
“Ramya was so sick that if she didn’t recover, the only option would be a heart transplant,” said Rao. “But we wanted to do everything to keep that heart.”
After steroid and COVID treatment, Ramya’s heart started to come back. “It didn’t make sense to me,” said Rao. “I don’t know what helped. If we hadn’t done ECMO, her heart probably wouldn’t have recovered, so I would say we have to support these patients and give them time for the heart to recover, even to the point of ECMO.”
Despite the good news, Ramya’s survival still hung in the balance. When she was disconnected from ECMO, clinicians discovered that the Impella device had caused a rare complication, damaging her mitral valve. The valve could be repaired surgically, but both Rao and Ram felt great trepidation at the prospect of cardiopulmonary bypass during the open-heart procedure.
“They would need to stop her heart and restart it, and I was concerned it would not restart,” Ram explained. “I didn’t like the idea of open-heart surgery, but my biggest fear was she was not going to survive it because of a really fresh, sick heart.”
The cardiologists’ fears did, in fact, come to pass: it took an hour to coax Ramya’s heart back at the end of surgery. But, just as the surgeon was preparing to reconnect Ramya to ECMO in desperation, “her heart recovered again,” Rao reported.
“Some things you never forget in life,” she said. “I can’t describe how everyone in the OR felt, all taking care of her. I told Ramya, ‘you are a fighter’.”
New strength
Six days would pass before Ramya woke up and learned of the astounding series of events that saved her. She knew “something was really wrong” because of the incision at the center of her chest, but learning she’d been on ECMO and the heart transplant list drove home how close to death she’d actually come.
“Most people don’t get off ECMO; they die on it,” she said. “And the chances of dying on the heart transplant list are very high. It was very strange to me that this was my story all of a sudden, when a week and a half earlier I was on rotation.”
Ongoing physical therapy over the past 3 months has transformed Ramya from a state of profound physical weakness to a place of relative strength. The now-fourth-year med student is turning 26 in November and is hungry to restart in-person rotations. Her downtime has been filled in part with researching myocarditis and collaborating with Rao on her own case study for journal publication.
But the mental trauma from her experience has girded her in ways she knows will make her stronger personally and professionally in the years ahead.
“It’s still very hard. I’m still recovering,” she acknowledged. “I described it to my therapist as an invisible wound on my brain.”
“When I came out of the hospital, I still had ECMO wounds, deep gashes on my legs that affected how fast and how long I could walk,” she said. “I felt like the same thing was going on my brain — a huge cut no one could see.”
Her intention to specialize in psychiatry has become more pressing now that Ramya has realized the impact of trauma on mental health.
“My body failing me was awful, but I could handle it,” she said. “Losing any part of my mind would have been way worse. I want to take care of that in my patients.”
This article first appeared on Medscape.com.
Within minutes of her arrival at Community North Hospital in Indianapolis, Ramya Yeleti’s vital signs plummeted; her pulse was at 45 beats per minute and her ejection fraction was hovering near 10%. “I definitely thought there was a chance I would close my eyes and never open them again, but I only had a few seconds to process that,” she recalled. Then everything went black. Ramya fell unconscious as shock pads were positioned and a swarm of clinicians prepared to insert an Impella heart pump through a catheter into her aorta.
The third-year medical student and aspiring psychiatrist had been doing in-person neurology rotations in July when she began to experience fever and uncontrolled vomiting. Her initial thought was that she must have caught the flu from a patient.
After all, Ramya, along with her father Ram Yeleti, MD, mother Indira, and twin sister Divya, had all weathered COVID-19 in previous months and later tested positive for SARS-CoV-2 antibodies. The only family member who had been spared was her younger brother Rohith.
Indira suffered a severe case, requiring ICU care for 2 days but no ventilator; the others experienced mostly mild symptoms. Ramya — who was studying for her third-year board exams after classes at Marian University College of Osteopathic Medicine in Indianapolis went virtual in March — was left with lingering fatigue; however, her cough and muscle aches abated and her sense of taste and smell returned. When she started rotations, she thought her life was getting back to normal.
Ramya’s flu symptoms did not improve. A university-mandated rapid COVID test came back negative, but 2 more days of vomiting started to worry both her and her father, who is a cardiologist and chief physician executive at Community Health Network in Indianapolis. After Ramya felt some chest pain, she asked her father to listen to her heart. All sounded normal, and Ram prescribed ondansetron for her nausea.
But the antiemetic didn’t work, and by the next morning both father and daughter were convinced that they needed to head to the emergency department.
“I wanted to double-check if I was missing something about her being dehydrated,” Ram told Medscape Medical News. “Several things can cause protracted nausea, like hepatitis, appendicitis, or another infection. I feel terribly guilty I didn’t realize she had a heart condition.”
A surprising turn for the worst
Ramya’s subtle symptoms quickly gave way to the dramatic cardiac crisis that unfolded just after her arrival at Community North. “Her EKG looked absolutely horrendous, like a 75-year-old having a heart attack,” Ram said.
As a cardiologist, he knew his daughter’s situation was growing dire when he heard physicians shouting that the Impella wasn’t working and she needed extracorporeal membrane oxygenation (ECMO).
“At that point, I didn’t think she’d survive,” her father recalled. “We had 10 physicians in the room who worked on her for 5 hours to get her stabilized.”
“It was especially traumatic because, obviously, I knew exactly what was happening,” he added. “You can’t sugarcoat anything.”
After being connected to the heart–lung equipment, Ramya was transferred to IU Health Methodist Hospital, also in Indianapolis, where she was tested again for COVID-19. Unlike the rapid test administered just days earlier, the PCR assay came back positive.
“I knew she had acute myocarditis, but coronavirus never crossed my mind,” said Ram.
“As we were dealing with her heart, we were also dealing with this challenge: she was coming back positive for COVID-19 again,” said Roopa Rao, MD, the heart failure transplant cardiologist at IU Health who treated Ramya.
“We weren’t sure whether we were dealing with an active infection or dead virus” from her previous infection, Rao said, “so we started treating her like she had active COVID-19 and gave her remdesivir, convalescent plasma, and steroids, which was the protocol in our hospital.”
A biopsy of Ramya’s heart tissue, along with blood tests, indicated a past parvovirus infection. It’s possible that Ramya’s previous coronavirus infection made her susceptible to heart damage from a newer parvovirus infection, said Rao. Either virus, or both together, could have been responsible for the calamity.
Although it was unheard of during Ramya’s cardiac crisis in early August, evolving evidence now raises the possibility that she is one of a handful of people in the world to be reinfected with SARS-CoV-2. Also emerging are cases of COVID-related myocarditis and other extreme heart complications, particularly in young people.
“At the time, it wasn’t really clear if people could have another infection so quickly,” Rao told Medscape Medical News. “It is possible she is one of these rare individuals to have COVID-19 twice. I’m hoping at some point we will have some clarity.”
“I would favor a coinfection as probably the triggering factor for her sickness,” she said. “It may take some time, but like any other disease — and it doesn’t look like COVID will go away magically — I hope we’ll have some answers down the road.”
Another wrinkle
The next 48 hours brought astonishing news: Ramya’s heart function had rebounded to nearly normal, and her ejection fraction increased to about 45%. Heart transplantation wouldn’t be necessary, although Rao stood poised to follow through if ECMO only sustained, rather than improved, Ramya’s prognosis.
“Ramya was so sick that if she didn’t recover, the only option would be a heart transplant,” said Rao. “But we wanted to do everything to keep that heart.”
After steroid and COVID treatment, Ramya’s heart started to come back. “It didn’t make sense to me,” said Rao. “I don’t know what helped. If we hadn’t done ECMO, her heart probably wouldn’t have recovered, so I would say we have to support these patients and give them time for the heart to recover, even to the point of ECMO.”
Despite the good news, Ramya’s survival still hung in the balance. When she was disconnected from ECMO, clinicians discovered that the Impella device had caused a rare complication, damaging her mitral valve. The valve could be repaired surgically, but both Rao and Ram felt great trepidation at the prospect of cardiopulmonary bypass during the open-heart procedure.
“They would need to stop her heart and restart it, and I was concerned it would not restart,” Ram explained. “I didn’t like the idea of open-heart surgery, but my biggest fear was she was not going to survive it because of a really fresh, sick heart.”
The cardiologists’ fears did, in fact, come to pass: it took an hour to coax Ramya’s heart back at the end of surgery. But, just as the surgeon was preparing to reconnect Ramya to ECMO in desperation, “her heart recovered again,” Rao reported.
“Some things you never forget in life,” she said. “I can’t describe how everyone in the OR felt, all taking care of her. I told Ramya, ‘you are a fighter’.”
New strength
Six days would pass before Ramya woke up and learned of the astounding series of events that saved her. She knew “something was really wrong” because of the incision at the center of her chest, but learning she’d been on ECMO and the heart transplant list drove home how close to death she’d actually come.
“Most people don’t get off ECMO; they die on it,” she said. “And the chances of dying on the heart transplant list are very high. It was very strange to me that this was my story all of a sudden, when a week and a half earlier I was on rotation.”
Ongoing physical therapy over the past 3 months has transformed Ramya from a state of profound physical weakness to a place of relative strength. The now-fourth-year med student is turning 26 in November and is hungry to restart in-person rotations. Her downtime has been filled in part with researching myocarditis and collaborating with Rao on her own case study for journal publication.
But the mental trauma from her experience has girded her in ways she knows will make her stronger personally and professionally in the years ahead.
“It’s still very hard. I’m still recovering,” she acknowledged. “I described it to my therapist as an invisible wound on my brain.”
“When I came out of the hospital, I still had ECMO wounds, deep gashes on my legs that affected how fast and how long I could walk,” she said. “I felt like the same thing was going on my brain — a huge cut no one could see.”
Her intention to specialize in psychiatry has become more pressing now that Ramya has realized the impact of trauma on mental health.
“My body failing me was awful, but I could handle it,” she said. “Losing any part of my mind would have been way worse. I want to take care of that in my patients.”
This article first appeared on Medscape.com.
Within minutes of her arrival at Community North Hospital in Indianapolis, Ramya Yeleti’s vital signs plummeted; her pulse was at 45 beats per minute and her ejection fraction was hovering near 10%. “I definitely thought there was a chance I would close my eyes and never open them again, but I only had a few seconds to process that,” she recalled. Then everything went black. Ramya fell unconscious as shock pads were positioned and a swarm of clinicians prepared to insert an Impella heart pump through a catheter into her aorta.
The third-year medical student and aspiring psychiatrist had been doing in-person neurology rotations in July when she began to experience fever and uncontrolled vomiting. Her initial thought was that she must have caught the flu from a patient.
After all, Ramya, along with her father Ram Yeleti, MD, mother Indira, and twin sister Divya, had all weathered COVID-19 in previous months and later tested positive for SARS-CoV-2 antibodies. The only family member who had been spared was her younger brother Rohith.
Indira suffered a severe case, requiring ICU care for 2 days but no ventilator; the others experienced mostly mild symptoms. Ramya — who was studying for her third-year board exams after classes at Marian University College of Osteopathic Medicine in Indianapolis went virtual in March — was left with lingering fatigue; however, her cough and muscle aches abated and her sense of taste and smell returned. When she started rotations, she thought her life was getting back to normal.
Ramya’s flu symptoms did not improve. A university-mandated rapid COVID test came back negative, but 2 more days of vomiting started to worry both her and her father, who is a cardiologist and chief physician executive at Community Health Network in Indianapolis. After Ramya felt some chest pain, she asked her father to listen to her heart. All sounded normal, and Ram prescribed ondansetron for her nausea.
But the antiemetic didn’t work, and by the next morning both father and daughter were convinced that they needed to head to the emergency department.
“I wanted to double-check if I was missing something about her being dehydrated,” Ram told Medscape Medical News. “Several things can cause protracted nausea, like hepatitis, appendicitis, or another infection. I feel terribly guilty I didn’t realize she had a heart condition.”
A surprising turn for the worst
Ramya’s subtle symptoms quickly gave way to the dramatic cardiac crisis that unfolded just after her arrival at Community North. “Her EKG looked absolutely horrendous, like a 75-year-old having a heart attack,” Ram said.
As a cardiologist, he knew his daughter’s situation was growing dire when he heard physicians shouting that the Impella wasn’t working and she needed extracorporeal membrane oxygenation (ECMO).
“At that point, I didn’t think she’d survive,” her father recalled. “We had 10 physicians in the room who worked on her for 5 hours to get her stabilized.”
“It was especially traumatic because, obviously, I knew exactly what was happening,” he added. “You can’t sugarcoat anything.”
After being connected to the heart–lung equipment, Ramya was transferred to IU Health Methodist Hospital, also in Indianapolis, where she was tested again for COVID-19. Unlike the rapid test administered just days earlier, the PCR assay came back positive.
“I knew she had acute myocarditis, but coronavirus never crossed my mind,” said Ram.
“As we were dealing with her heart, we were also dealing with this challenge: she was coming back positive for COVID-19 again,” said Roopa Rao, MD, the heart failure transplant cardiologist at IU Health who treated Ramya.
“We weren’t sure whether we were dealing with an active infection or dead virus” from her previous infection, Rao said, “so we started treating her like she had active COVID-19 and gave her remdesivir, convalescent plasma, and steroids, which was the protocol in our hospital.”
A biopsy of Ramya’s heart tissue, along with blood tests, indicated a past parvovirus infection. It’s possible that Ramya’s previous coronavirus infection made her susceptible to heart damage from a newer parvovirus infection, said Rao. Either virus, or both together, could have been responsible for the calamity.
Although it was unheard of during Ramya’s cardiac crisis in early August, evolving evidence now raises the possibility that she is one of a handful of people in the world to be reinfected with SARS-CoV-2. Also emerging are cases of COVID-related myocarditis and other extreme heart complications, particularly in young people.
“At the time, it wasn’t really clear if people could have another infection so quickly,” Rao told Medscape Medical News. “It is possible she is one of these rare individuals to have COVID-19 twice. I’m hoping at some point we will have some clarity.”
“I would favor a coinfection as probably the triggering factor for her sickness,” she said. “It may take some time, but like any other disease — and it doesn’t look like COVID will go away magically — I hope we’ll have some answers down the road.”
Another wrinkle
The next 48 hours brought astonishing news: Ramya’s heart function had rebounded to nearly normal, and her ejection fraction increased to about 45%. Heart transplantation wouldn’t be necessary, although Rao stood poised to follow through if ECMO only sustained, rather than improved, Ramya’s prognosis.
“Ramya was so sick that if she didn’t recover, the only option would be a heart transplant,” said Rao. “But we wanted to do everything to keep that heart.”
After steroid and COVID treatment, Ramya’s heart started to come back. “It didn’t make sense to me,” said Rao. “I don’t know what helped. If we hadn’t done ECMO, her heart probably wouldn’t have recovered, so I would say we have to support these patients and give them time for the heart to recover, even to the point of ECMO.”
Despite the good news, Ramya’s survival still hung in the balance. When she was disconnected from ECMO, clinicians discovered that the Impella device had caused a rare complication, damaging her mitral valve. The valve could be repaired surgically, but both Rao and Ram felt great trepidation at the prospect of cardiopulmonary bypass during the open-heart procedure.
“They would need to stop her heart and restart it, and I was concerned it would not restart,” Ram explained. “I didn’t like the idea of open-heart surgery, but my biggest fear was she was not going to survive it because of a really fresh, sick heart.”
The cardiologists’ fears did, in fact, come to pass: it took an hour to coax Ramya’s heart back at the end of surgery. But, just as the surgeon was preparing to reconnect Ramya to ECMO in desperation, “her heart recovered again,” Rao reported.
“Some things you never forget in life,” she said. “I can’t describe how everyone in the OR felt, all taking care of her. I told Ramya, ‘you are a fighter’.”
New strength
Six days would pass before Ramya woke up and learned of the astounding series of events that saved her. She knew “something was really wrong” because of the incision at the center of her chest, but learning she’d been on ECMO and the heart transplant list drove home how close to death she’d actually come.
“Most people don’t get off ECMO; they die on it,” she said. “And the chances of dying on the heart transplant list are very high. It was very strange to me that this was my story all of a sudden, when a week and a half earlier I was on rotation.”
Ongoing physical therapy over the past 3 months has transformed Ramya from a state of profound physical weakness to a place of relative strength. The now-fourth-year med student is turning 26 in November and is hungry to restart in-person rotations. Her downtime has been filled in part with researching myocarditis and collaborating with Rao on her own case study for journal publication.
But the mental trauma from her experience has girded her in ways she knows will make her stronger personally and professionally in the years ahead.
“It’s still very hard. I’m still recovering,” she acknowledged. “I described it to my therapist as an invisible wound on my brain.”
“When I came out of the hospital, I still had ECMO wounds, deep gashes on my legs that affected how fast and how long I could walk,” she said. “I felt like the same thing was going on my brain — a huge cut no one could see.”
Her intention to specialize in psychiatry has become more pressing now that Ramya has realized the impact of trauma on mental health.
“My body failing me was awful, but I could handle it,” she said. “Losing any part of my mind would have been way worse. I want to take care of that in my patients.”
This article first appeared on Medscape.com.
About 17% of COVID-19 survivors retest positive in follow-up study
For reasons unknown, about one in six people who recovered from COVID-19 subsequently retested positive at least 2 weeks later, researchers reported in a study in Italy.
Sore throat and rhinitis were the only symptoms associated with a positive result. “Patients who continued to have respiratory symptoms, especially, were more likely to have a new positive test result,” lead author Francesco Landi, MD, PhD, said in an interview.
“This suggests the persistence of respiratory symptoms should not be underestimated and should be adequately assessed in all patients considered recovered from COVID-19,” he said.
“The study results are interesting,” Akiko Iwasaki, PhD, an immunobiologist at Yale University and the Howard Hughes Medical Institute, both in New Haven, Conn.,, said in an interview. “There are other reports of RNA detection postdischarge, but this study ... found that only two symptoms out of many – sore throat and rhinitis – were higher in those with PCR [polymerase chain reaction]-positive status.”
The study was published online Sept. 18 in the American Journal of Preventive Medicine.
The findings could carry important implications for people who continue to be symptomatic. “It is reasonable to be cautious and avoid close contact with others, wear a face mask, and possibly undergo an additional nasopharyngeal swab,” said Dr. Landi, associate professor of internal medicine at Catholic University of the Sacred Heart in Rome.
“One of most interesting findings is that persistent symptoms do not correlate with PCR positivity, suggesting that symptoms are in many cases not due to ongoing viral replication,” Jonathan Karn, PhD, professor and chair of the department of molecular biology and microbiology at Case Western Reserve University, Cleveland, said in an interview.
“The key technical problem, which they have discussed, is that a viral RNA signal in the PCR assay does not necessarily mean that infectious virus is present,” Dr. Karn said. He added that new comprehensive viral RNA analyses would be needed to answer this question.
Official COVID-19 recovery
To identify risk factors and COVID-19 survivors more likely to retest positive, Dr. Landi and members of the Gemelli Against COVID-19 Post-Acute Care Study Group evaluated 131 people after hospital discharge.
All participants met World Health Organization criteria for release from isolation, including two negative test results at least 24 hours apart, and were studied between April 21 and May 21. Mean age was 56 and 39% were women. Only a slightly higher mean body mass index of 27.6 kg/m2 in the positive group versus 25.9 in the negative group, was significant.
Although 51% of survivors reported fatigue, 44% had dyspnea, and 17% were coughing, the rates did not differ significantly between groups. In contrast, 18% of positive survivors and 4% of negative survivors had a sore throat (P = .04), and 27% versus 12%, respectively, reported rhinitis (P = .05).
People returned for follow-up visits a mean 17 days after the second negative swab test.
Asymptomatic COVID-19 carriers
“These findings indicate that a noteworthy rate of recovered patients with COVID-19 could still be asymptomatic carriers of the virus,” the researchers noted in the paper. “Even in the absence of specific guidelines, the 22 patients who tested positive for COVID-19 again were suggested to quarantine for a second time.”
No family member or close contact of the positive survivors reported SARS-CoV-2 infection. All patients continued to wear masks and observe social distancing recommendations, which makes it “very difficult to affirm whether these patients were really contagious,” the researchers noted.
Next steps
Evaluating all COVID-19 survivors to identify any who retest positive “will be a crucial contribution to a better understanding of both the natural history of COVID-19 as well as the public health implications of viral shedding,” the authors wrote.
One study limitation is that the reverse transcriptase–PCR test reveals genetic sequences specific to COVID-19. “It is important to underline that this is not a viral culture and cannot determine whether the virus is viable and transmissible,” the researchers noted.
“In this respect, we are trying to better understand if the persistence of long-time positive [reverse transcriptase]–PCR test for COVID-19 is really correlated to a potential contagiousness,” they added.
Dr. Landi and colleagues said their findings should be considered preliminary, and larger data samples are warranted to validate the results.
Dr. Landi and Dr. Karn disclosed no relevant financial relationships. Dr. Iwasaki disclosed a research grant from Condair, a 5% or greater equity interest in RIGImmune, and income of $250 or more from PureTec.
A version of this article originally appeared on Medscape.com.
For reasons unknown, about one in six people who recovered from COVID-19 subsequently retested positive at least 2 weeks later, researchers reported in a study in Italy.
Sore throat and rhinitis were the only symptoms associated with a positive result. “Patients who continued to have respiratory symptoms, especially, were more likely to have a new positive test result,” lead author Francesco Landi, MD, PhD, said in an interview.
“This suggests the persistence of respiratory symptoms should not be underestimated and should be adequately assessed in all patients considered recovered from COVID-19,” he said.
“The study results are interesting,” Akiko Iwasaki, PhD, an immunobiologist at Yale University and the Howard Hughes Medical Institute, both in New Haven, Conn.,, said in an interview. “There are other reports of RNA detection postdischarge, but this study ... found that only two symptoms out of many – sore throat and rhinitis – were higher in those with PCR [polymerase chain reaction]-positive status.”
The study was published online Sept. 18 in the American Journal of Preventive Medicine.
The findings could carry important implications for people who continue to be symptomatic. “It is reasonable to be cautious and avoid close contact with others, wear a face mask, and possibly undergo an additional nasopharyngeal swab,” said Dr. Landi, associate professor of internal medicine at Catholic University of the Sacred Heart in Rome.
“One of most interesting findings is that persistent symptoms do not correlate with PCR positivity, suggesting that symptoms are in many cases not due to ongoing viral replication,” Jonathan Karn, PhD, professor and chair of the department of molecular biology and microbiology at Case Western Reserve University, Cleveland, said in an interview.
“The key technical problem, which they have discussed, is that a viral RNA signal in the PCR assay does not necessarily mean that infectious virus is present,” Dr. Karn said. He added that new comprehensive viral RNA analyses would be needed to answer this question.
Official COVID-19 recovery
To identify risk factors and COVID-19 survivors more likely to retest positive, Dr. Landi and members of the Gemelli Against COVID-19 Post-Acute Care Study Group evaluated 131 people after hospital discharge.
All participants met World Health Organization criteria for release from isolation, including two negative test results at least 24 hours apart, and were studied between April 21 and May 21. Mean age was 56 and 39% were women. Only a slightly higher mean body mass index of 27.6 kg/m2 in the positive group versus 25.9 in the negative group, was significant.
Although 51% of survivors reported fatigue, 44% had dyspnea, and 17% were coughing, the rates did not differ significantly between groups. In contrast, 18% of positive survivors and 4% of negative survivors had a sore throat (P = .04), and 27% versus 12%, respectively, reported rhinitis (P = .05).
People returned for follow-up visits a mean 17 days after the second negative swab test.
Asymptomatic COVID-19 carriers
“These findings indicate that a noteworthy rate of recovered patients with COVID-19 could still be asymptomatic carriers of the virus,” the researchers noted in the paper. “Even in the absence of specific guidelines, the 22 patients who tested positive for COVID-19 again were suggested to quarantine for a second time.”
No family member or close contact of the positive survivors reported SARS-CoV-2 infection. All patients continued to wear masks and observe social distancing recommendations, which makes it “very difficult to affirm whether these patients were really contagious,” the researchers noted.
Next steps
Evaluating all COVID-19 survivors to identify any who retest positive “will be a crucial contribution to a better understanding of both the natural history of COVID-19 as well as the public health implications of viral shedding,” the authors wrote.
One study limitation is that the reverse transcriptase–PCR test reveals genetic sequences specific to COVID-19. “It is important to underline that this is not a viral culture and cannot determine whether the virus is viable and transmissible,” the researchers noted.
“In this respect, we are trying to better understand if the persistence of long-time positive [reverse transcriptase]–PCR test for COVID-19 is really correlated to a potential contagiousness,” they added.
Dr. Landi and colleagues said their findings should be considered preliminary, and larger data samples are warranted to validate the results.
Dr. Landi and Dr. Karn disclosed no relevant financial relationships. Dr. Iwasaki disclosed a research grant from Condair, a 5% or greater equity interest in RIGImmune, and income of $250 or more from PureTec.
A version of this article originally appeared on Medscape.com.
For reasons unknown, about one in six people who recovered from COVID-19 subsequently retested positive at least 2 weeks later, researchers reported in a study in Italy.
Sore throat and rhinitis were the only symptoms associated with a positive result. “Patients who continued to have respiratory symptoms, especially, were more likely to have a new positive test result,” lead author Francesco Landi, MD, PhD, said in an interview.
“This suggests the persistence of respiratory symptoms should not be underestimated and should be adequately assessed in all patients considered recovered from COVID-19,” he said.
“The study results are interesting,” Akiko Iwasaki, PhD, an immunobiologist at Yale University and the Howard Hughes Medical Institute, both in New Haven, Conn.,, said in an interview. “There are other reports of RNA detection postdischarge, but this study ... found that only two symptoms out of many – sore throat and rhinitis – were higher in those with PCR [polymerase chain reaction]-positive status.”
The study was published online Sept. 18 in the American Journal of Preventive Medicine.
The findings could carry important implications for people who continue to be symptomatic. “It is reasonable to be cautious and avoid close contact with others, wear a face mask, and possibly undergo an additional nasopharyngeal swab,” said Dr. Landi, associate professor of internal medicine at Catholic University of the Sacred Heart in Rome.
“One of most interesting findings is that persistent symptoms do not correlate with PCR positivity, suggesting that symptoms are in many cases not due to ongoing viral replication,” Jonathan Karn, PhD, professor and chair of the department of molecular biology and microbiology at Case Western Reserve University, Cleveland, said in an interview.
“The key technical problem, which they have discussed, is that a viral RNA signal in the PCR assay does not necessarily mean that infectious virus is present,” Dr. Karn said. He added that new comprehensive viral RNA analyses would be needed to answer this question.
Official COVID-19 recovery
To identify risk factors and COVID-19 survivors more likely to retest positive, Dr. Landi and members of the Gemelli Against COVID-19 Post-Acute Care Study Group evaluated 131 people after hospital discharge.
All participants met World Health Organization criteria for release from isolation, including two negative test results at least 24 hours apart, and were studied between April 21 and May 21. Mean age was 56 and 39% were women. Only a slightly higher mean body mass index of 27.6 kg/m2 in the positive group versus 25.9 in the negative group, was significant.
Although 51% of survivors reported fatigue, 44% had dyspnea, and 17% were coughing, the rates did not differ significantly between groups. In contrast, 18% of positive survivors and 4% of negative survivors had a sore throat (P = .04), and 27% versus 12%, respectively, reported rhinitis (P = .05).
People returned for follow-up visits a mean 17 days after the second negative swab test.
Asymptomatic COVID-19 carriers
“These findings indicate that a noteworthy rate of recovered patients with COVID-19 could still be asymptomatic carriers of the virus,” the researchers noted in the paper. “Even in the absence of specific guidelines, the 22 patients who tested positive for COVID-19 again were suggested to quarantine for a second time.”
No family member or close contact of the positive survivors reported SARS-CoV-2 infection. All patients continued to wear masks and observe social distancing recommendations, which makes it “very difficult to affirm whether these patients were really contagious,” the researchers noted.
Next steps
Evaluating all COVID-19 survivors to identify any who retest positive “will be a crucial contribution to a better understanding of both the natural history of COVID-19 as well as the public health implications of viral shedding,” the authors wrote.
One study limitation is that the reverse transcriptase–PCR test reveals genetic sequences specific to COVID-19. “It is important to underline that this is not a viral culture and cannot determine whether the virus is viable and transmissible,” the researchers noted.
“In this respect, we are trying to better understand if the persistence of long-time positive [reverse transcriptase]–PCR test for COVID-19 is really correlated to a potential contagiousness,” they added.
Dr. Landi and colleagues said their findings should be considered preliminary, and larger data samples are warranted to validate the results.
Dr. Landi and Dr. Karn disclosed no relevant financial relationships. Dr. Iwasaki disclosed a research grant from Condair, a 5% or greater equity interest in RIGImmune, and income of $250 or more from PureTec.
A version of this article originally appeared on Medscape.com.
CDC panel takes on COVID vaccine rollout, risks, and side effects
Federal advisers who will help determine which Americans get the first COVID vaccines took an in-depth look Oct. 30 at the challenges they face in selecting priority groups.
The Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention (CDC) will face two key decisions once a COVID vaccine wins clearance from the US Food and Drug Administration (FDA).
ACIP will need to decide whether to recommend its use in adults (the age group in which vaccines are currently being tested). The group will also need to offer direction on which groups should get priority in vaccine allocation, inasmuch as early supplies will not be sufficient to vaccinate everyone.
At the Oct. 30 meeting, CDC’s Kathleen Dooling, MD, MPH, suggested that ACIP plan on tackling these issues as two separate questions when it comes time to weigh in on an approved vaccine. Although there was no formal vote among ACIP members at the meeting, Dooling’s proposal for tackling a future recommendation in a two-part fashion drew positive feedback.
ACIP member Katherine A. Poehling, MD, MPH, suggested that the panel and CDC be ready to reexamine the situation frequently regarding COVID vaccination. “Perhaps we could think about reviewing data on a monthly basis and updating the recommendation, so that we can account for the concerns and balance both the benefits and the [potential] harm,” Poehling said.
Dooling agreed. “Both the vaccine recommendation and allocation will be revisited in what is a very dynamic situation,” Dooling replied to Poehling. “So all new evidence will be brought to ACIP, and certainly the allocation as vaccine distribution proceeds will need to be adjusted accordingly.”
Ethics and limited evidence
During the meeting, ACIP members repeatedly expressed discomfort with the prospect of having to weigh in on widespread use of COVID vaccines on the basis of limited evidence.
Within months, FDA may opt for a special clearance, known as an emergency use authorization (EUA), for one or more of the experimental COVID vaccines now in advanced testing. Many of FDA’s past EUA clearances were granted for test kits. For those EUA approvals, the agency considered risks of false results but not longer-term, direct harm to patients from these products.
With a COVID vaccine, there will be strong pressure to distribute doses as quickly as possible with the hope of curbing the pandemic, which has already led to more than 229,000 deaths in the United States alone and has disrupted lives and economies around the world. But questions will persist about the possibility of serious complications from these vaccines, ACIP members noted.
“My personal struggle is the ethical side and how to balance these two,” said ACIP member Robert L. Atmar, MD, of Baylor College of Medicine, Houston, Texas, who noted that he expects his fellow panelists to share this concern.
Currently, four experimental COVID vaccines likely to be used in the United States have advanced to phase 3 testing. Pfizer Inc and BioNtech have enrolled more than 42,000 participants in a test of their candidate, BNT162b2 vaccine, and rival Moderna has enrolled about 30,000 participants in a test of its mRNA-1273 vaccine, CDC staff said.
The other two advanced COVID vaccine candidates have overcome recent hurdles. AstraZeneca Plc on Oct. 23 announced that FDA had removed a hold on the testing of its AZD1222 vaccine candidate; the trial will enroll approximately 30,000 people. Johnson & Johnson’s Janssen unit also announced that day the lifting of a safety pause for its Ad26.COV2.S vaccine; the phase 3 trial for that vaccine will enroll approximately 60,000 volunteers. Federal agencies, states, and territories have developed plans for future distribution of COVID vaccines, CDC staff said in briefing materials for today’s ACIP meeting.
Several ACIP members raised many of the same concerns that members of an FDA advisory committee raised at a meeting earlier in October. ACIP and FDA advisers honed in on the FDA’s decision to set a median follow-up duration of 2 months in phase 3 trials in connection with expected EUA applications for COVID-19 vaccines.
“I struggle with following people for 2 months after their second vaccination as a time point to start making final decisions about safety,” said ACIP member Sharon E. Frey, MD, a professor at St. Louis University School of Medicine, St. Louis, Missouri. “I just want to put that out there.”
Medical front line, then who?
There is consensus that healthcare workers be in the first stage ― Phase 1 ― of distribution. That recommendation was made in a report from the National Academies of Sciences, Engineering, and Medicine (NASEM). Phase 1A would include first responders; Phase 1B might include people of all ages who have two or more comorbidities that put them at significantly higher risk for COVID-19 or death, as well as older adults living in congregate or overcrowded settings, the NASEM report said.
A presentation from the CDC’s Matthew Biggerstaff, ScD, MPH, underscored challenges in distributing what are expected to be limited initial supplies of COVID vaccines.
Biggerstaff showed several scenarios the CDC’s Data, Analytics, and Modeling Task Force had studied. The initial allocation of vaccines would be for healthcare workers, followed by what the CDC called Phase 1B.
Choices for a rollout may include next giving COVID vaccines to people at high risk, such as persons who have one or more chronic medical conditions, including heart disease, diabetes, kidney disease, or obesity. Other options for the rollout could be to vaccinate people aged 65 years and older or essential workers whose employment puts them in contact with the public, thus raising the risk of contracting the virus.
The CDC’s research found that the greatest impact in preventing death was to initially vaccinate adults aged 65 and older in Phase 1B. The agency staff described this approach as likely to result in an about “1 to 11% increase in averted deaths across the scenarios.”
Initially vaccinating essential workers or high-risk adults in Phase 1B would avert the most infections. The agency staff described this approach as yielding about “1 to 5% increase in averted infections across the scenarios,” Biggerstaff said during his presentation.
The following are other findings of the CDC staff:
The earlier the vaccine rollout relative to increasing transmission, the greater the averted percentage and differences between the strategies.
Differences were not substantial in some scenarios.
The need to continue efforts to slow the spread of COVID-19 should be emphasized.
Adverse effects
ACIP members also heard about strategies for tracking potential side effects of future vaccines. A presentation by Tom Shimabukuro, MD, MPH, MBA, from the CDC’s COVID-19 Vaccine Task Force/Vaccine Safety Team, included details about a new smartphone-based active surveillance program for COVID-19 vaccine safety.
Known as v-safe, this system would use Web-based survey monitoring and incorporate text messaging. It would conduct electronic health checks on vaccine recipients, which would occur daily during the first week post vaccination and weekly thereafter for 6 weeks from the time of vaccination.
Clinicians “can play an important role in helping CDC enroll patients in v-safe at the time of vaccination,” Shimabukuro noted in his presentation. This would add another task, though, for clinicians, the CDC staff noted.
Pregnancy and breastfeeding are special concerns
Of special concern with the rollout of a COVID vaccine are recommendations regarding pregnancy and breastfeeding. Women constitute about 75% of the healthcare workforce, CDC staff noted.
At the time the initial ACIP COVID vaccination recommendations are made, there could be approximately 330,000 healthcare personnel who are pregnant or who have recently given birth. Available data indicate potentially increased risks for severe maternal illness and preterm birth associated with SARS-CoV-2 infection, said CDC’s Megan Wallace, DrPH, MPH, in a presentation for the Friday meeting.
In an Oct. 27 letter to ACIP, Chair Jose Romero, the American College of Obstetricians and Gynecologists (ACOG), urged the panel to ensure that pregnant women and new mothers in the healthcare workforce have priority access to a COVID vaccine. Pregnant and lactating women were “noticeably and alarmingly absent from the NASEM vaccine allocation plan for COVID-19,” wrote Christopher M. Zahn, MD, vice president for practice activities at ACOG, in the letter to Romero.
“ACOG urges ACIP to incorporate pregnant and lactating women clearly and explicitly into its COVID-19 vaccine allocation and prioritization framework,” Zahn wrote. “Should an Emergency Use Authorization be executed for one or more COVID-19 vaccines and provide a permissive recommendation for pregnant and lactating women, pregnant health care workers, pregnant first responders, and pregnant individuals with underlying conditions should be prioritized for vaccination alongside their non-pregnant peers.”
This article first appeared on Medscape.com.
Federal advisers who will help determine which Americans get the first COVID vaccines took an in-depth look Oct. 30 at the challenges they face in selecting priority groups.
The Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention (CDC) will face two key decisions once a COVID vaccine wins clearance from the US Food and Drug Administration (FDA).
ACIP will need to decide whether to recommend its use in adults (the age group in which vaccines are currently being tested). The group will also need to offer direction on which groups should get priority in vaccine allocation, inasmuch as early supplies will not be sufficient to vaccinate everyone.
At the Oct. 30 meeting, CDC’s Kathleen Dooling, MD, MPH, suggested that ACIP plan on tackling these issues as two separate questions when it comes time to weigh in on an approved vaccine. Although there was no formal vote among ACIP members at the meeting, Dooling’s proposal for tackling a future recommendation in a two-part fashion drew positive feedback.
ACIP member Katherine A. Poehling, MD, MPH, suggested that the panel and CDC be ready to reexamine the situation frequently regarding COVID vaccination. “Perhaps we could think about reviewing data on a monthly basis and updating the recommendation, so that we can account for the concerns and balance both the benefits and the [potential] harm,” Poehling said.
Dooling agreed. “Both the vaccine recommendation and allocation will be revisited in what is a very dynamic situation,” Dooling replied to Poehling. “So all new evidence will be brought to ACIP, and certainly the allocation as vaccine distribution proceeds will need to be adjusted accordingly.”
Ethics and limited evidence
During the meeting, ACIP members repeatedly expressed discomfort with the prospect of having to weigh in on widespread use of COVID vaccines on the basis of limited evidence.
Within months, FDA may opt for a special clearance, known as an emergency use authorization (EUA), for one or more of the experimental COVID vaccines now in advanced testing. Many of FDA’s past EUA clearances were granted for test kits. For those EUA approvals, the agency considered risks of false results but not longer-term, direct harm to patients from these products.
With a COVID vaccine, there will be strong pressure to distribute doses as quickly as possible with the hope of curbing the pandemic, which has already led to more than 229,000 deaths in the United States alone and has disrupted lives and economies around the world. But questions will persist about the possibility of serious complications from these vaccines, ACIP members noted.
“My personal struggle is the ethical side and how to balance these two,” said ACIP member Robert L. Atmar, MD, of Baylor College of Medicine, Houston, Texas, who noted that he expects his fellow panelists to share this concern.
Currently, four experimental COVID vaccines likely to be used in the United States have advanced to phase 3 testing. Pfizer Inc and BioNtech have enrolled more than 42,000 participants in a test of their candidate, BNT162b2 vaccine, and rival Moderna has enrolled about 30,000 participants in a test of its mRNA-1273 vaccine, CDC staff said.
The other two advanced COVID vaccine candidates have overcome recent hurdles. AstraZeneca Plc on Oct. 23 announced that FDA had removed a hold on the testing of its AZD1222 vaccine candidate; the trial will enroll approximately 30,000 people. Johnson & Johnson’s Janssen unit also announced that day the lifting of a safety pause for its Ad26.COV2.S vaccine; the phase 3 trial for that vaccine will enroll approximately 60,000 volunteers. Federal agencies, states, and territories have developed plans for future distribution of COVID vaccines, CDC staff said in briefing materials for today’s ACIP meeting.
Several ACIP members raised many of the same concerns that members of an FDA advisory committee raised at a meeting earlier in October. ACIP and FDA advisers honed in on the FDA’s decision to set a median follow-up duration of 2 months in phase 3 trials in connection with expected EUA applications for COVID-19 vaccines.
“I struggle with following people for 2 months after their second vaccination as a time point to start making final decisions about safety,” said ACIP member Sharon E. Frey, MD, a professor at St. Louis University School of Medicine, St. Louis, Missouri. “I just want to put that out there.”
Medical front line, then who?
There is consensus that healthcare workers be in the first stage ― Phase 1 ― of distribution. That recommendation was made in a report from the National Academies of Sciences, Engineering, and Medicine (NASEM). Phase 1A would include first responders; Phase 1B might include people of all ages who have two or more comorbidities that put them at significantly higher risk for COVID-19 or death, as well as older adults living in congregate or overcrowded settings, the NASEM report said.
A presentation from the CDC’s Matthew Biggerstaff, ScD, MPH, underscored challenges in distributing what are expected to be limited initial supplies of COVID vaccines.
Biggerstaff showed several scenarios the CDC’s Data, Analytics, and Modeling Task Force had studied. The initial allocation of vaccines would be for healthcare workers, followed by what the CDC called Phase 1B.
Choices for a rollout may include next giving COVID vaccines to people at high risk, such as persons who have one or more chronic medical conditions, including heart disease, diabetes, kidney disease, or obesity. Other options for the rollout could be to vaccinate people aged 65 years and older or essential workers whose employment puts them in contact with the public, thus raising the risk of contracting the virus.
The CDC’s research found that the greatest impact in preventing death was to initially vaccinate adults aged 65 and older in Phase 1B. The agency staff described this approach as likely to result in an about “1 to 11% increase in averted deaths across the scenarios.”
Initially vaccinating essential workers or high-risk adults in Phase 1B would avert the most infections. The agency staff described this approach as yielding about “1 to 5% increase in averted infections across the scenarios,” Biggerstaff said during his presentation.
The following are other findings of the CDC staff:
The earlier the vaccine rollout relative to increasing transmission, the greater the averted percentage and differences between the strategies.
Differences were not substantial in some scenarios.
The need to continue efforts to slow the spread of COVID-19 should be emphasized.
Adverse effects
ACIP members also heard about strategies for tracking potential side effects of future vaccines. A presentation by Tom Shimabukuro, MD, MPH, MBA, from the CDC’s COVID-19 Vaccine Task Force/Vaccine Safety Team, included details about a new smartphone-based active surveillance program for COVID-19 vaccine safety.
Known as v-safe, this system would use Web-based survey monitoring and incorporate text messaging. It would conduct electronic health checks on vaccine recipients, which would occur daily during the first week post vaccination and weekly thereafter for 6 weeks from the time of vaccination.
Clinicians “can play an important role in helping CDC enroll patients in v-safe at the time of vaccination,” Shimabukuro noted in his presentation. This would add another task, though, for clinicians, the CDC staff noted.
Pregnancy and breastfeeding are special concerns
Of special concern with the rollout of a COVID vaccine are recommendations regarding pregnancy and breastfeeding. Women constitute about 75% of the healthcare workforce, CDC staff noted.
At the time the initial ACIP COVID vaccination recommendations are made, there could be approximately 330,000 healthcare personnel who are pregnant or who have recently given birth. Available data indicate potentially increased risks for severe maternal illness and preterm birth associated with SARS-CoV-2 infection, said CDC’s Megan Wallace, DrPH, MPH, in a presentation for the Friday meeting.
In an Oct. 27 letter to ACIP, Chair Jose Romero, the American College of Obstetricians and Gynecologists (ACOG), urged the panel to ensure that pregnant women and new mothers in the healthcare workforce have priority access to a COVID vaccine. Pregnant and lactating women were “noticeably and alarmingly absent from the NASEM vaccine allocation plan for COVID-19,” wrote Christopher M. Zahn, MD, vice president for practice activities at ACOG, in the letter to Romero.
“ACOG urges ACIP to incorporate pregnant and lactating women clearly and explicitly into its COVID-19 vaccine allocation and prioritization framework,” Zahn wrote. “Should an Emergency Use Authorization be executed for one or more COVID-19 vaccines and provide a permissive recommendation for pregnant and lactating women, pregnant health care workers, pregnant first responders, and pregnant individuals with underlying conditions should be prioritized for vaccination alongside their non-pregnant peers.”
This article first appeared on Medscape.com.
Federal advisers who will help determine which Americans get the first COVID vaccines took an in-depth look Oct. 30 at the challenges they face in selecting priority groups.
The Advisory Committee on Immunization Practices (ACIP) of the Centers for Disease Control and Prevention (CDC) will face two key decisions once a COVID vaccine wins clearance from the US Food and Drug Administration (FDA).
ACIP will need to decide whether to recommend its use in adults (the age group in which vaccines are currently being tested). The group will also need to offer direction on which groups should get priority in vaccine allocation, inasmuch as early supplies will not be sufficient to vaccinate everyone.
At the Oct. 30 meeting, CDC’s Kathleen Dooling, MD, MPH, suggested that ACIP plan on tackling these issues as two separate questions when it comes time to weigh in on an approved vaccine. Although there was no formal vote among ACIP members at the meeting, Dooling’s proposal for tackling a future recommendation in a two-part fashion drew positive feedback.
ACIP member Katherine A. Poehling, MD, MPH, suggested that the panel and CDC be ready to reexamine the situation frequently regarding COVID vaccination. “Perhaps we could think about reviewing data on a monthly basis and updating the recommendation, so that we can account for the concerns and balance both the benefits and the [potential] harm,” Poehling said.
Dooling agreed. “Both the vaccine recommendation and allocation will be revisited in what is a very dynamic situation,” Dooling replied to Poehling. “So all new evidence will be brought to ACIP, and certainly the allocation as vaccine distribution proceeds will need to be adjusted accordingly.”
Ethics and limited evidence
During the meeting, ACIP members repeatedly expressed discomfort with the prospect of having to weigh in on widespread use of COVID vaccines on the basis of limited evidence.
Within months, FDA may opt for a special clearance, known as an emergency use authorization (EUA), for one or more of the experimental COVID vaccines now in advanced testing. Many of FDA’s past EUA clearances were granted for test kits. For those EUA approvals, the agency considered risks of false results but not longer-term, direct harm to patients from these products.
With a COVID vaccine, there will be strong pressure to distribute doses as quickly as possible with the hope of curbing the pandemic, which has already led to more than 229,000 deaths in the United States alone and has disrupted lives and economies around the world. But questions will persist about the possibility of serious complications from these vaccines, ACIP members noted.
“My personal struggle is the ethical side and how to balance these two,” said ACIP member Robert L. Atmar, MD, of Baylor College of Medicine, Houston, Texas, who noted that he expects his fellow panelists to share this concern.
Currently, four experimental COVID vaccines likely to be used in the United States have advanced to phase 3 testing. Pfizer Inc and BioNtech have enrolled more than 42,000 participants in a test of their candidate, BNT162b2 vaccine, and rival Moderna has enrolled about 30,000 participants in a test of its mRNA-1273 vaccine, CDC staff said.
The other two advanced COVID vaccine candidates have overcome recent hurdles. AstraZeneca Plc on Oct. 23 announced that FDA had removed a hold on the testing of its AZD1222 vaccine candidate; the trial will enroll approximately 30,000 people. Johnson & Johnson’s Janssen unit also announced that day the lifting of a safety pause for its Ad26.COV2.S vaccine; the phase 3 trial for that vaccine will enroll approximately 60,000 volunteers. Federal agencies, states, and territories have developed plans for future distribution of COVID vaccines, CDC staff said in briefing materials for today’s ACIP meeting.
Several ACIP members raised many of the same concerns that members of an FDA advisory committee raised at a meeting earlier in October. ACIP and FDA advisers honed in on the FDA’s decision to set a median follow-up duration of 2 months in phase 3 trials in connection with expected EUA applications for COVID-19 vaccines.
“I struggle with following people for 2 months after their second vaccination as a time point to start making final decisions about safety,” said ACIP member Sharon E. Frey, MD, a professor at St. Louis University School of Medicine, St. Louis, Missouri. “I just want to put that out there.”
Medical front line, then who?
There is consensus that healthcare workers be in the first stage ― Phase 1 ― of distribution. That recommendation was made in a report from the National Academies of Sciences, Engineering, and Medicine (NASEM). Phase 1A would include first responders; Phase 1B might include people of all ages who have two or more comorbidities that put them at significantly higher risk for COVID-19 or death, as well as older adults living in congregate or overcrowded settings, the NASEM report said.
A presentation from the CDC’s Matthew Biggerstaff, ScD, MPH, underscored challenges in distributing what are expected to be limited initial supplies of COVID vaccines.
Biggerstaff showed several scenarios the CDC’s Data, Analytics, and Modeling Task Force had studied. The initial allocation of vaccines would be for healthcare workers, followed by what the CDC called Phase 1B.
Choices for a rollout may include next giving COVID vaccines to people at high risk, such as persons who have one or more chronic medical conditions, including heart disease, diabetes, kidney disease, or obesity. Other options for the rollout could be to vaccinate people aged 65 years and older or essential workers whose employment puts them in contact with the public, thus raising the risk of contracting the virus.
The CDC’s research found that the greatest impact in preventing death was to initially vaccinate adults aged 65 and older in Phase 1B. The agency staff described this approach as likely to result in an about “1 to 11% increase in averted deaths across the scenarios.”
Initially vaccinating essential workers or high-risk adults in Phase 1B would avert the most infections. The agency staff described this approach as yielding about “1 to 5% increase in averted infections across the scenarios,” Biggerstaff said during his presentation.
The following are other findings of the CDC staff:
The earlier the vaccine rollout relative to increasing transmission, the greater the averted percentage and differences between the strategies.
Differences were not substantial in some scenarios.
The need to continue efforts to slow the spread of COVID-19 should be emphasized.
Adverse effects
ACIP members also heard about strategies for tracking potential side effects of future vaccines. A presentation by Tom Shimabukuro, MD, MPH, MBA, from the CDC’s COVID-19 Vaccine Task Force/Vaccine Safety Team, included details about a new smartphone-based active surveillance program for COVID-19 vaccine safety.
Known as v-safe, this system would use Web-based survey monitoring and incorporate text messaging. It would conduct electronic health checks on vaccine recipients, which would occur daily during the first week post vaccination and weekly thereafter for 6 weeks from the time of vaccination.
Clinicians “can play an important role in helping CDC enroll patients in v-safe at the time of vaccination,” Shimabukuro noted in his presentation. This would add another task, though, for clinicians, the CDC staff noted.
Pregnancy and breastfeeding are special concerns
Of special concern with the rollout of a COVID vaccine are recommendations regarding pregnancy and breastfeeding. Women constitute about 75% of the healthcare workforce, CDC staff noted.
At the time the initial ACIP COVID vaccination recommendations are made, there could be approximately 330,000 healthcare personnel who are pregnant or who have recently given birth. Available data indicate potentially increased risks for severe maternal illness and preterm birth associated with SARS-CoV-2 infection, said CDC’s Megan Wallace, DrPH, MPH, in a presentation for the Friday meeting.
In an Oct. 27 letter to ACIP, Chair Jose Romero, the American College of Obstetricians and Gynecologists (ACOG), urged the panel to ensure that pregnant women and new mothers in the healthcare workforce have priority access to a COVID vaccine. Pregnant and lactating women were “noticeably and alarmingly absent from the NASEM vaccine allocation plan for COVID-19,” wrote Christopher M. Zahn, MD, vice president for practice activities at ACOG, in the letter to Romero.
“ACOG urges ACIP to incorporate pregnant and lactating women clearly and explicitly into its COVID-19 vaccine allocation and prioritization framework,” Zahn wrote. “Should an Emergency Use Authorization be executed for one or more COVID-19 vaccines and provide a permissive recommendation for pregnant and lactating women, pregnant health care workers, pregnant first responders, and pregnant individuals with underlying conditions should be prioritized for vaccination alongside their non-pregnant peers.”
This article first appeared on Medscape.com.
Frivolous lawsuits: Still a big threat to doctors?
Dr. G, a New York surgeon, was only a couple years into practice when he faced his first lawsuit.
After undergoing liposuction surgery on the area of her calf and ankle, a patient claimed she had developed a severe allergic reaction, characterized by small areas of necrosis on the lower extremities, said Dr. G, who asked to remain anonymous. However, the alleged injury seemed suspicious, said Dr. G, considering that 3 weeks after the surgery, the area had shown a successful result with minimal swelling.
Six months into the suit, Dr. G received a shocking phone call. It was the patient’s estranged husband, who revealed that his wife was having an affair with another man, a physician. In recorded phone calls, the patient and her paramour had discussed causing an injury near the patient’s calf in an attempt to sue and get rich, the husband relayed. Dr. G immediately contacted his insurance carrier with the news, but his attorney said the information would not be admissible in court. Instead, the insurer settled with the patient, who received about $125,000.
At the time, Dr. G did not have a consent-to-settle clause in his contract, so the insurer was able to settle without his approval.
In legal practice, a frivolous claim is defined as one that lacks a supporting legal argument or any factual basis. A claim issued with the intent of disturbing, annoying, or harassing the opposing party can also be described as legally frivolous, said Michael Stinson, vice president of government relations and public policy for the Medical Professional Liability Association (MPL Association), a trade association for medical liability insurers.
However, when most physicians refer to “frivolous claims,” they often mean a claim in which there is no attributable negligence. Such suits represent a second category of claims – nonmeritorious lawsuits.
“I think people intermix nonmeritorious and frivolous all the time,” Mr. Stinson said. “In the vast majority of nonmeritorious claims, the patient has suffered an adverse outcome, it’s just that it wasn’t the result of negligence, whereas with a frivolous lawsuit, they really haven’t suffered any damage, so they’ve got no business filing a lawsuit on any level.”
A third type of so-called frivolous suit is that of a fraudulent or fake claim, in which, as Dr. G experienced, a patient causes a self-injury or lies about a condition to craft a false claim against a physician.
If a patient files a claim that the patient knows is false, the patient commits fraud and may be subject to counterclaims for malicious prosecution or abuse of process, said Jeffrey Segal, MD, JD, a neurosurgeon and health law attorney. Further, the patient would be testifying under oath, and such testimony can be considered perjury, a criminal offense with criminal penalties.
Sadly, Dr. G was the target of another frivolous lawsuit years later. In that suit, a patient claimed the surgeon had left a piece of sponge in her breast cavity during surgery. The case was dismissed when medical records proved the patient knew that the foreign body resulted from an unrelated procedure she had undergone years earlier.
“There is so much abuse in the court system,” Dr. G said. “You really don’t think stuff like that will happen to you, especially if you honor the profession. It’s unfortunate. It’s left a very bitter taste in my mouth.”
Frivolous claims have long been a subject of debate. Tort reform advocates often contend that such claims are pervasive. They cite them as key reasons for high health care costs and say that they have led to the rise of defensive medicine. Plaintiffs’ attorneys counter that the rate of frivolous claims is widely exaggerated and argue that the pursuit of frivolous claims would be “bad business” for legal firms.
“I have never seen a frivolous malpractice claim,” says Malcolm P. McConnell III, JD, a Richmond, Va., medical malpractice attorney and chair of the Medical Malpractice Legislative Subcommittee for the Virginia Trial Lawyers Association. “I cannot say that such things never happen, but any lawyer bringing such a thing is foolish, because there is no reward for it.”
Are shotgun lawsuits frivolous?
To many physicians, being dragged into a lawsuit over a complaint or medical outcome in which they were not involved is frivolous, said Stanislaw Stawicki, MD, a trauma surgeon and researcher based in Bethlehem, Pa. Dr. Stawicki was named in a lawsuit along with a long list of medical staff who interacted in some way with the plaintiff. Dr. Stawicki himself saw the patient once and made a note in the chart but had nothing to do with the patient’s surgery or with any critical decisions regarding his care, he said.
“Nothing really prepares you for seeing your name on a legal complaint,” Dr. Stawicki said. “It’s traumatic. I had to block out entire days to give depositions, which were really kind of pointless. Questions like, ‘Is this really your name? Where did you train? Were you there that morning?’ Stuff that was really not consequential to the fact that someone had surgery a month earlier and had some sort of complication.”
Dr. Stawicki was eventually dropped from the claim, but not before a nearly year-long ordeal of legal proceedings, meetings, and paperwork.
It is common practice for plaintiffs’ attorneys to add codefendants in the early stages of a claim, said David M. Studdert, ScD, a leading health law researcher and a professor of law at Stanford (Calif.) Law School. Defendants are gradually dismissed as the case moves forward and details of the incident become clearer, he said.
“Plaintiffs’ attorneys have strong incentives to try and choose claims that will be successful,” Dr. Studdert said. “However, in the early point in the process, neither the patient nor the attorney may have a good idea what has actually happened with care. So sometimes, filing a lawsuit may be the only way to begin the process of opening up that information.”
A study by Dr. Studdert in which medical malpractice claims, errors, and compensation payments were analyzed found that, out of 1,452 claims, about one-third (37%) did not involve errors.
“Many physicians might call those frivolous lawsuits, but in fact, most of those don’t go on to receive compensation,” he said. “We suspect that in many instances, those claims are simply dropped once it becomes apparent that there wasn’t error involved.
“They can still be burdensome, anxiety provoking, and time consuming for physicians who are named in those suits, so I don’t want to suggest that claims that don’t involve errors are not a problem,” said Dr. Studdert. “However, I think it’s wrong to assume, as many people do when they use the term ‘frivolous lawsuit,’ that this is really an extortionary effort by a plaintiffs’ attorney to try to get money out of a hospital or a physician for care that was really unproblematic.”
Certain ‘frivolous’ cases more common than others
Nonmeritorious claims still occur relatively frequently today, according to data from the Medical Professional Liability Association’s Data Sharing Project. Of about 18,000 liability claims reported from 2016 to 2018, 65% were dropped, withdrawn, or dismissed. Of the 6% of claims that went before a jury, more than 85% resulted in a verdict for the defendant, the researchers found.
“Basically, any claim that does not result in a payment because the underlying claim of negligence on the part of a health professional had been demonstrated, proven, or adjudicated false is one we would describe as nonmeritorious,” Mr. Stinson said.
The MPL Association does not track cases that meet the legal definition of frivolous, said Mr. Stinson, and they “don’t see truly frivolous lawsuits very often.”
Malpractice claims are risky, expensive, and aggressively defended, says Mr. McConnell, the plaintiffs’ attorney. Mr. McConnell, who has been practicing for 30 years, said his own claim selection process is very rigorous and that he cannot afford to pursue claims that aren’t well supported by science and medicine.
“Pursuing frivolous cases would bankrupt me and ruin my reputation,” he said. “A lawyer I know once said he would write a check for $10,000 to anyone who could show him a lawyer who makes a living pursuing frivolous medical malpractice cases. It’s a fair challenge. The economics and the practices of liability carriers and defense lawyers make frivolous cases a dead end for plaintiff lawyers.”
Most medical malpractice cases are taken on a contingency fee basis, Mr. McConnell noted, meaning that the plaintiff’s lawyer is not paid unless the claim is successful.
“This means that the plaintiff’s lawyer is risking 2 years of intensive labor on a case which may yield no fee at all,” he said. “Obviously, any reasonable lawyer is going to want to minimize that risk. The only way to minimize that risk is for the case to be solid, not weak, and certainly not frivolous.”
But Dr. Segal, the health law attorney, says that plenty of frivolous liability claims are levied each year, with attorneys willing to pursue them.
It’s true that seasoned plaintiffs’ attorneys generally screen for merit and damages, Dr. Segal said, but in some instances, attorneys who are not trained in malpractice law accept frivolous claims and take them forward. In some cases, they are slip-and-fall accident attorneys accustomed to receiving modest amounts from insurance companies quickly, said Dr. Segal, founder of Medical Justice, a company that helps deter frivolous lawsuits against physicians.
“If we lived in a perfectly rational universe where plaintiffs’ attorneys screened cases well and only took the meritorious cases forward, we would see less frivolous cases filed, but that’s not the universe I live in,” Dr. Segal said. “There are well over a million attorneys in this country, and some are hungrier than others. The attorneys may frequently get burned in the end, and maybe that attorney won’t move another malpractice case forward, but there’s always someone else willing to take their place.”
Medical Justice has twice run a Most Frivolous Lawsuit Contest on its website, one in 2008 and one in late 2018. The first contest drew 30 entries, and the second garnered nearly 40 submissions, primarily from physicians who were defendants in the cases, according to Dr. Segal. (Dr. G’s lawsuit was highlighted in the most recent contest.)
In one case, an emergency physician was drawn into litigation by the family of a deceased patient. The patient experienced sudden cardiac arrythmia at home, and paramedics were unable to intubate her or establish IV access. She was transferred to the hospital, where resuscitation efforts continued, but she remained in asystole and was pronounced dead after 15 minutes.
At the hospital, blood tests were conducted. They showed that her serum potassium concentration was elevated to about 12 mEq/L, Dr. Segal said. The family initiated a claim in which they accused the emergency physician of failure to diagnose hyperkalemia. They alleged that had the hyperkalemia been discovered sooner, the patient’s death could have been prevented.
“If you had no other facts about this, you would wonder how a person with potassium that high would even be alive,” Dr. Segal said. “But what they were looking at was the body decomposing and all the potassium in the cells being released into the bloodstream. It wasn’t the cause of the problem, it was an effect of the problem. She really was dead on arrival, and she was probably dead at home.”
The case was eventually dropped.
Although the outcome for the patient was tragic, says Dr. Segal, the case is one of many types of frivolous claims that exist today.
“Yes, frivolous cases are out there,” he said.
Fraudulent claims uncommon
As for fraudulent medical liability claims, legal experts say they’re rare. J. Richard Moore, JD, an Indianapolis-based medical liability defense attorney, said he’s never personally encountered a medical malpractice claim in which he believed a plaintiff caused an injury or an illness and attempted to blame it on a physician.
However, Mr. Moore has defended many claims in which the illness or condition the plaintiff claimed was caused or was made worse through medical negligence was actually a preexisting condition or a preexisting condition that worsened and was not related to any medical negligence, Mr. Moore said.
“Although I have often felt in such cases that the plaintiff really knew that the condition was not affected by any alleged medical negligence, I would not put that in the ‘fraudulent claim’ category because it can be very difficult to establish a person’s subjective state of mind,” he said. “Usually in those cases, the plaintiff just denies memory of previous medical records or claims that the previous doctor who treated him or her for the same condition ‘got it wrong.’ In those cases, it is generally left to the jury whether to believe the plaintiff or not.”
Mr. Stinson also says he has not come across a truly fraudulent medical liability case. He noted that such a claim might be similar to a person falsely claiming a soft-tissue injury following an alleged slip-and-fall accident.
“Clearly, a fraudulent claim could be viewed as riskier from the plaintiff’s perspective because they could face criminal prosecution for insurance fraud, whereas if a claim is merely frivolous, they probably only run the risk of court-issued fine, if even that. That may be why we don’t often see fraudulent MPL claims.”
Ways to prevent or fight frivolous lawsuits
Since Dr. Stawicki’s legal nightmare as a resident, rules have tightened in Pennsylvania, and it is now more difficult to file frivolous claims, he said.
Pennsylvania is one of at least 28 states that require a certificate of merit in order for a medical liability claim to move forward. The provisions generally state that an appropriately licensed professional must supply a written statement attesting that the care the patient received failed to meet acceptable professional standards and that such conduct was a cause in the alleged harm.
“There is now a much greater burden of proof regarding what can proceed,” Dr. Stawicki said. “I’ve been involved in a couple cases that did not proceed because there was no certificate of merit.”
Although these reforms may help, not all merit rules are created equal. Some states require that the expert who signs the affidavit be knowledgeable in the relevant issues involved in the action. Other states have looser requirements. In one of the cases featured in Medical Justice’s Most Frivolous Lawsuit Contest, a podiatrist signed a supporting declaration for a claim related to obstetric care.
For physicians facing a frivolous claim, fighting it out in court depends on a number of factors. Without a consent-to-settle clause in the contract, an insurer can make the final decision on whether to defend or settle a case.
Resolving a malpractice claim is generally a business decision for the insurer, Dr. Studdert said.
“When the claim is for a relatively low amount of money, the costs of moving forward to defend that claim may be much more than the costs of simply settling it would be,” he said. “On the other hand, liability insurers and their lawyers are repeat players here, as are the plaintiffs’ attorneys. They don’t want to incentivize plaintiffs’ attorneys to bring questionable claims, and if they settle quickly, that may do so.”
Mr. Stinson, of the MPL Association, said a truly frivolous claim – one with no legal basis – is highly unlikely to be settled, “especially by MPL Association members who go beyond having a purely financial interest in their insureds to also focus on their professional reputation/integrity.” MPL Association members insure nearly 2 million health care professionals globally, including 2,500 hospitals and more than two-thirds of America’s physicians who are in private practice.
Physicians should make sure they know what is and what is not included in their policy, Dr. Segal said.
“The broker should sit down with the doctor, ideally before initial purchase or renewal, and explain in clear terms what the carrier’s obligations are and what the physician’s obligations are,” he said. “Know what type of protection is being purchased and what conditions might trigger a surprising and unhappy outcome.”
Should I countersue?
For truly frivolous claims, physicians have the legal right to sue for damages caused by the unfounded complaint.
Perhaps the most well-known case of a successful malpractice countersuit is that of Louisville neurosurgeon John Guarnaschelli, MD, who in 2000 won $72,000 in damages against a plaintiffs’ attorney for malicious prosecution.
The physician’s countersuit followed the dismissal of a negligence claim against Dr. Guarnaschelli by a patient who contracted meningitis. The plaintiffs’ attorney had made little effort to gather evidence to connect Dr. Guarnaschelli to the patient’s injuries and had consulted only one other physician, a client of his, before filing the lawsuit, according to a summary of the case in the American Bar Association Journal.
Malicious prosecution is the most common legal theory of recovery for physicians in countersuits, according to a review of successful countersuits by doctors. Dr. Stawicki is a coauthor of that review. Other legal theories that physicians can raise include abuse of process, negligence, defamation, invasion of privacy, and infliction of emotional distress. Of the 13 cases evaluated in the article by Dr. Stawicki and colleagues, damages awarded to physicians ranged from about $13,000 to $125,000.
Although some doctors have success, pursuing a counterclaim can be a difficult feat, said Benjamin Braslow, MD, a trauma surgeon and professor of clinical surgery at the University of Pennsylvania in Philadelphia.
“The main takeaways were it’s an uphill battle often met with not only resistance but diminishing returns to countersue,” said Dr. Braslow, a coauthor of the countersuits analysis. “You have to meet very specific criteria regarding leveling the suit, and it may end up being a costly, time-consuming battle.”
To prove malicious prosecution, for example, a physician must show that a claim was instituted without probable cause, that the suing party acted maliciously in instituting the action, and that the doctor was damaged by the action, among other essential elements.
As for Dr. G, the surgeon, he now has a contract with a consent-to-settle clause and has taken other legal precautions since his lawsuits. He requires that his patients sign an agreement that any negligence claims they levy go to arbitration. If an arbitrator finds in the patient’s favor, the case may proceed to court, he said. However, he requires another agreement such that if patients lose in court, they are responsible for his legal fees.
“I’m just more careful,” he said. “I ask all my staff in the office to use their judgment, however superficial, if they feel something is wrong with an individual to tell me so. I’d rather send them away than operate on them and have it result in a lawsuit.”
A version of this article originally appeared on Medscape.com.
Dr. G, a New York surgeon, was only a couple years into practice when he faced his first lawsuit.
After undergoing liposuction surgery on the area of her calf and ankle, a patient claimed she had developed a severe allergic reaction, characterized by small areas of necrosis on the lower extremities, said Dr. G, who asked to remain anonymous. However, the alleged injury seemed suspicious, said Dr. G, considering that 3 weeks after the surgery, the area had shown a successful result with minimal swelling.
Six months into the suit, Dr. G received a shocking phone call. It was the patient’s estranged husband, who revealed that his wife was having an affair with another man, a physician. In recorded phone calls, the patient and her paramour had discussed causing an injury near the patient’s calf in an attempt to sue and get rich, the husband relayed. Dr. G immediately contacted his insurance carrier with the news, but his attorney said the information would not be admissible in court. Instead, the insurer settled with the patient, who received about $125,000.
At the time, Dr. G did not have a consent-to-settle clause in his contract, so the insurer was able to settle without his approval.
In legal practice, a frivolous claim is defined as one that lacks a supporting legal argument or any factual basis. A claim issued with the intent of disturbing, annoying, or harassing the opposing party can also be described as legally frivolous, said Michael Stinson, vice president of government relations and public policy for the Medical Professional Liability Association (MPL Association), a trade association for medical liability insurers.
However, when most physicians refer to “frivolous claims,” they often mean a claim in which there is no attributable negligence. Such suits represent a second category of claims – nonmeritorious lawsuits.
“I think people intermix nonmeritorious and frivolous all the time,” Mr. Stinson said. “In the vast majority of nonmeritorious claims, the patient has suffered an adverse outcome, it’s just that it wasn’t the result of negligence, whereas with a frivolous lawsuit, they really haven’t suffered any damage, so they’ve got no business filing a lawsuit on any level.”
A third type of so-called frivolous suit is that of a fraudulent or fake claim, in which, as Dr. G experienced, a patient causes a self-injury or lies about a condition to craft a false claim against a physician.
If a patient files a claim that the patient knows is false, the patient commits fraud and may be subject to counterclaims for malicious prosecution or abuse of process, said Jeffrey Segal, MD, JD, a neurosurgeon and health law attorney. Further, the patient would be testifying under oath, and such testimony can be considered perjury, a criminal offense with criminal penalties.
Sadly, Dr. G was the target of another frivolous lawsuit years later. In that suit, a patient claimed the surgeon had left a piece of sponge in her breast cavity during surgery. The case was dismissed when medical records proved the patient knew that the foreign body resulted from an unrelated procedure she had undergone years earlier.
“There is so much abuse in the court system,” Dr. G said. “You really don’t think stuff like that will happen to you, especially if you honor the profession. It’s unfortunate. It’s left a very bitter taste in my mouth.”
Frivolous claims have long been a subject of debate. Tort reform advocates often contend that such claims are pervasive. They cite them as key reasons for high health care costs and say that they have led to the rise of defensive medicine. Plaintiffs’ attorneys counter that the rate of frivolous claims is widely exaggerated and argue that the pursuit of frivolous claims would be “bad business” for legal firms.
“I have never seen a frivolous malpractice claim,” says Malcolm P. McConnell III, JD, a Richmond, Va., medical malpractice attorney and chair of the Medical Malpractice Legislative Subcommittee for the Virginia Trial Lawyers Association. “I cannot say that such things never happen, but any lawyer bringing such a thing is foolish, because there is no reward for it.”
Are shotgun lawsuits frivolous?
To many physicians, being dragged into a lawsuit over a complaint or medical outcome in which they were not involved is frivolous, said Stanislaw Stawicki, MD, a trauma surgeon and researcher based in Bethlehem, Pa. Dr. Stawicki was named in a lawsuit along with a long list of medical staff who interacted in some way with the plaintiff. Dr. Stawicki himself saw the patient once and made a note in the chart but had nothing to do with the patient’s surgery or with any critical decisions regarding his care, he said.
“Nothing really prepares you for seeing your name on a legal complaint,” Dr. Stawicki said. “It’s traumatic. I had to block out entire days to give depositions, which were really kind of pointless. Questions like, ‘Is this really your name? Where did you train? Were you there that morning?’ Stuff that was really not consequential to the fact that someone had surgery a month earlier and had some sort of complication.”
Dr. Stawicki was eventually dropped from the claim, but not before a nearly year-long ordeal of legal proceedings, meetings, and paperwork.
It is common practice for plaintiffs’ attorneys to add codefendants in the early stages of a claim, said David M. Studdert, ScD, a leading health law researcher and a professor of law at Stanford (Calif.) Law School. Defendants are gradually dismissed as the case moves forward and details of the incident become clearer, he said.
“Plaintiffs’ attorneys have strong incentives to try and choose claims that will be successful,” Dr. Studdert said. “However, in the early point in the process, neither the patient nor the attorney may have a good idea what has actually happened with care. So sometimes, filing a lawsuit may be the only way to begin the process of opening up that information.”
A study by Dr. Studdert in which medical malpractice claims, errors, and compensation payments were analyzed found that, out of 1,452 claims, about one-third (37%) did not involve errors.
“Many physicians might call those frivolous lawsuits, but in fact, most of those don’t go on to receive compensation,” he said. “We suspect that in many instances, those claims are simply dropped once it becomes apparent that there wasn’t error involved.
“They can still be burdensome, anxiety provoking, and time consuming for physicians who are named in those suits, so I don’t want to suggest that claims that don’t involve errors are not a problem,” said Dr. Studdert. “However, I think it’s wrong to assume, as many people do when they use the term ‘frivolous lawsuit,’ that this is really an extortionary effort by a plaintiffs’ attorney to try to get money out of a hospital or a physician for care that was really unproblematic.”
Certain ‘frivolous’ cases more common than others
Nonmeritorious claims still occur relatively frequently today, according to data from the Medical Professional Liability Association’s Data Sharing Project. Of about 18,000 liability claims reported from 2016 to 2018, 65% were dropped, withdrawn, or dismissed. Of the 6% of claims that went before a jury, more than 85% resulted in a verdict for the defendant, the researchers found.
“Basically, any claim that does not result in a payment because the underlying claim of negligence on the part of a health professional had been demonstrated, proven, or adjudicated false is one we would describe as nonmeritorious,” Mr. Stinson said.
The MPL Association does not track cases that meet the legal definition of frivolous, said Mr. Stinson, and they “don’t see truly frivolous lawsuits very often.”
Malpractice claims are risky, expensive, and aggressively defended, says Mr. McConnell, the plaintiffs’ attorney. Mr. McConnell, who has been practicing for 30 years, said his own claim selection process is very rigorous and that he cannot afford to pursue claims that aren’t well supported by science and medicine.
“Pursuing frivolous cases would bankrupt me and ruin my reputation,” he said. “A lawyer I know once said he would write a check for $10,000 to anyone who could show him a lawyer who makes a living pursuing frivolous medical malpractice cases. It’s a fair challenge. The economics and the practices of liability carriers and defense lawyers make frivolous cases a dead end for plaintiff lawyers.”
Most medical malpractice cases are taken on a contingency fee basis, Mr. McConnell noted, meaning that the plaintiff’s lawyer is not paid unless the claim is successful.
“This means that the plaintiff’s lawyer is risking 2 years of intensive labor on a case which may yield no fee at all,” he said. “Obviously, any reasonable lawyer is going to want to minimize that risk. The only way to minimize that risk is for the case to be solid, not weak, and certainly not frivolous.”
But Dr. Segal, the health law attorney, says that plenty of frivolous liability claims are levied each year, with attorneys willing to pursue them.
It’s true that seasoned plaintiffs’ attorneys generally screen for merit and damages, Dr. Segal said, but in some instances, attorneys who are not trained in malpractice law accept frivolous claims and take them forward. In some cases, they are slip-and-fall accident attorneys accustomed to receiving modest amounts from insurance companies quickly, said Dr. Segal, founder of Medical Justice, a company that helps deter frivolous lawsuits against physicians.
“If we lived in a perfectly rational universe where plaintiffs’ attorneys screened cases well and only took the meritorious cases forward, we would see less frivolous cases filed, but that’s not the universe I live in,” Dr. Segal said. “There are well over a million attorneys in this country, and some are hungrier than others. The attorneys may frequently get burned in the end, and maybe that attorney won’t move another malpractice case forward, but there’s always someone else willing to take their place.”
Medical Justice has twice run a Most Frivolous Lawsuit Contest on its website, one in 2008 and one in late 2018. The first contest drew 30 entries, and the second garnered nearly 40 submissions, primarily from physicians who were defendants in the cases, according to Dr. Segal. (Dr. G’s lawsuit was highlighted in the most recent contest.)
In one case, an emergency physician was drawn into litigation by the family of a deceased patient. The patient experienced sudden cardiac arrythmia at home, and paramedics were unable to intubate her or establish IV access. She was transferred to the hospital, where resuscitation efforts continued, but she remained in asystole and was pronounced dead after 15 minutes.
At the hospital, blood tests were conducted. They showed that her serum potassium concentration was elevated to about 12 mEq/L, Dr. Segal said. The family initiated a claim in which they accused the emergency physician of failure to diagnose hyperkalemia. They alleged that had the hyperkalemia been discovered sooner, the patient’s death could have been prevented.
“If you had no other facts about this, you would wonder how a person with potassium that high would even be alive,” Dr. Segal said. “But what they were looking at was the body decomposing and all the potassium in the cells being released into the bloodstream. It wasn’t the cause of the problem, it was an effect of the problem. She really was dead on arrival, and she was probably dead at home.”
The case was eventually dropped.
Although the outcome for the patient was tragic, says Dr. Segal, the case is one of many types of frivolous claims that exist today.
“Yes, frivolous cases are out there,” he said.
Fraudulent claims uncommon
As for fraudulent medical liability claims, legal experts say they’re rare. J. Richard Moore, JD, an Indianapolis-based medical liability defense attorney, said he’s never personally encountered a medical malpractice claim in which he believed a plaintiff caused an injury or an illness and attempted to blame it on a physician.
However, Mr. Moore has defended many claims in which the illness or condition the plaintiff claimed was caused or was made worse through medical negligence was actually a preexisting condition or a preexisting condition that worsened and was not related to any medical negligence, Mr. Moore said.
“Although I have often felt in such cases that the plaintiff really knew that the condition was not affected by any alleged medical negligence, I would not put that in the ‘fraudulent claim’ category because it can be very difficult to establish a person’s subjective state of mind,” he said. “Usually in those cases, the plaintiff just denies memory of previous medical records or claims that the previous doctor who treated him or her for the same condition ‘got it wrong.’ In those cases, it is generally left to the jury whether to believe the plaintiff or not.”
Mr. Stinson also says he has not come across a truly fraudulent medical liability case. He noted that such a claim might be similar to a person falsely claiming a soft-tissue injury following an alleged slip-and-fall accident.
“Clearly, a fraudulent claim could be viewed as riskier from the plaintiff’s perspective because they could face criminal prosecution for insurance fraud, whereas if a claim is merely frivolous, they probably only run the risk of court-issued fine, if even that. That may be why we don’t often see fraudulent MPL claims.”
Ways to prevent or fight frivolous lawsuits
Since Dr. Stawicki’s legal nightmare as a resident, rules have tightened in Pennsylvania, and it is now more difficult to file frivolous claims, he said.
Pennsylvania is one of at least 28 states that require a certificate of merit in order for a medical liability claim to move forward. The provisions generally state that an appropriately licensed professional must supply a written statement attesting that the care the patient received failed to meet acceptable professional standards and that such conduct was a cause in the alleged harm.
“There is now a much greater burden of proof regarding what can proceed,” Dr. Stawicki said. “I’ve been involved in a couple cases that did not proceed because there was no certificate of merit.”
Although these reforms may help, not all merit rules are created equal. Some states require that the expert who signs the affidavit be knowledgeable in the relevant issues involved in the action. Other states have looser requirements. In one of the cases featured in Medical Justice’s Most Frivolous Lawsuit Contest, a podiatrist signed a supporting declaration for a claim related to obstetric care.
For physicians facing a frivolous claim, fighting it out in court depends on a number of factors. Without a consent-to-settle clause in the contract, an insurer can make the final decision on whether to defend or settle a case.
Resolving a malpractice claim is generally a business decision for the insurer, Dr. Studdert said.
“When the claim is for a relatively low amount of money, the costs of moving forward to defend that claim may be much more than the costs of simply settling it would be,” he said. “On the other hand, liability insurers and their lawyers are repeat players here, as are the plaintiffs’ attorneys. They don’t want to incentivize plaintiffs’ attorneys to bring questionable claims, and if they settle quickly, that may do so.”
Mr. Stinson, of the MPL Association, said a truly frivolous claim – one with no legal basis – is highly unlikely to be settled, “especially by MPL Association members who go beyond having a purely financial interest in their insureds to also focus on their professional reputation/integrity.” MPL Association members insure nearly 2 million health care professionals globally, including 2,500 hospitals and more than two-thirds of America’s physicians who are in private practice.
Physicians should make sure they know what is and what is not included in their policy, Dr. Segal said.
“The broker should sit down with the doctor, ideally before initial purchase or renewal, and explain in clear terms what the carrier’s obligations are and what the physician’s obligations are,” he said. “Know what type of protection is being purchased and what conditions might trigger a surprising and unhappy outcome.”
Should I countersue?
For truly frivolous claims, physicians have the legal right to sue for damages caused by the unfounded complaint.
Perhaps the most well-known case of a successful malpractice countersuit is that of Louisville neurosurgeon John Guarnaschelli, MD, who in 2000 won $72,000 in damages against a plaintiffs’ attorney for malicious prosecution.
The physician’s countersuit followed the dismissal of a negligence claim against Dr. Guarnaschelli by a patient who contracted meningitis. The plaintiffs’ attorney had made little effort to gather evidence to connect Dr. Guarnaschelli to the patient’s injuries and had consulted only one other physician, a client of his, before filing the lawsuit, according to a summary of the case in the American Bar Association Journal.
Malicious prosecution is the most common legal theory of recovery for physicians in countersuits, according to a review of successful countersuits by doctors. Dr. Stawicki is a coauthor of that review. Other legal theories that physicians can raise include abuse of process, negligence, defamation, invasion of privacy, and infliction of emotional distress. Of the 13 cases evaluated in the article by Dr. Stawicki and colleagues, damages awarded to physicians ranged from about $13,000 to $125,000.
Although some doctors have success, pursuing a counterclaim can be a difficult feat, said Benjamin Braslow, MD, a trauma surgeon and professor of clinical surgery at the University of Pennsylvania in Philadelphia.
“The main takeaways were it’s an uphill battle often met with not only resistance but diminishing returns to countersue,” said Dr. Braslow, a coauthor of the countersuits analysis. “You have to meet very specific criteria regarding leveling the suit, and it may end up being a costly, time-consuming battle.”
To prove malicious prosecution, for example, a physician must show that a claim was instituted without probable cause, that the suing party acted maliciously in instituting the action, and that the doctor was damaged by the action, among other essential elements.
As for Dr. G, the surgeon, he now has a contract with a consent-to-settle clause and has taken other legal precautions since his lawsuits. He requires that his patients sign an agreement that any negligence claims they levy go to arbitration. If an arbitrator finds in the patient’s favor, the case may proceed to court, he said. However, he requires another agreement such that if patients lose in court, they are responsible for his legal fees.
“I’m just more careful,” he said. “I ask all my staff in the office to use their judgment, however superficial, if they feel something is wrong with an individual to tell me so. I’d rather send them away than operate on them and have it result in a lawsuit.”
A version of this article originally appeared on Medscape.com.
Dr. G, a New York surgeon, was only a couple years into practice when he faced his first lawsuit.
After undergoing liposuction surgery on the area of her calf and ankle, a patient claimed she had developed a severe allergic reaction, characterized by small areas of necrosis on the lower extremities, said Dr. G, who asked to remain anonymous. However, the alleged injury seemed suspicious, said Dr. G, considering that 3 weeks after the surgery, the area had shown a successful result with minimal swelling.
Six months into the suit, Dr. G received a shocking phone call. It was the patient’s estranged husband, who revealed that his wife was having an affair with another man, a physician. In recorded phone calls, the patient and her paramour had discussed causing an injury near the patient’s calf in an attempt to sue and get rich, the husband relayed. Dr. G immediately contacted his insurance carrier with the news, but his attorney said the information would not be admissible in court. Instead, the insurer settled with the patient, who received about $125,000.
At the time, Dr. G did not have a consent-to-settle clause in his contract, so the insurer was able to settle without his approval.
In legal practice, a frivolous claim is defined as one that lacks a supporting legal argument or any factual basis. A claim issued with the intent of disturbing, annoying, or harassing the opposing party can also be described as legally frivolous, said Michael Stinson, vice president of government relations and public policy for the Medical Professional Liability Association (MPL Association), a trade association for medical liability insurers.
However, when most physicians refer to “frivolous claims,” they often mean a claim in which there is no attributable negligence. Such suits represent a second category of claims – nonmeritorious lawsuits.
“I think people intermix nonmeritorious and frivolous all the time,” Mr. Stinson said. “In the vast majority of nonmeritorious claims, the patient has suffered an adverse outcome, it’s just that it wasn’t the result of negligence, whereas with a frivolous lawsuit, they really haven’t suffered any damage, so they’ve got no business filing a lawsuit on any level.”
A third type of so-called frivolous suit is that of a fraudulent or fake claim, in which, as Dr. G experienced, a patient causes a self-injury or lies about a condition to craft a false claim against a physician.
If a patient files a claim that the patient knows is false, the patient commits fraud and may be subject to counterclaims for malicious prosecution or abuse of process, said Jeffrey Segal, MD, JD, a neurosurgeon and health law attorney. Further, the patient would be testifying under oath, and such testimony can be considered perjury, a criminal offense with criminal penalties.
Sadly, Dr. G was the target of another frivolous lawsuit years later. In that suit, a patient claimed the surgeon had left a piece of sponge in her breast cavity during surgery. The case was dismissed when medical records proved the patient knew that the foreign body resulted from an unrelated procedure she had undergone years earlier.
“There is so much abuse in the court system,” Dr. G said. “You really don’t think stuff like that will happen to you, especially if you honor the profession. It’s unfortunate. It’s left a very bitter taste in my mouth.”
Frivolous claims have long been a subject of debate. Tort reform advocates often contend that such claims are pervasive. They cite them as key reasons for high health care costs and say that they have led to the rise of defensive medicine. Plaintiffs’ attorneys counter that the rate of frivolous claims is widely exaggerated and argue that the pursuit of frivolous claims would be “bad business” for legal firms.
“I have never seen a frivolous malpractice claim,” says Malcolm P. McConnell III, JD, a Richmond, Va., medical malpractice attorney and chair of the Medical Malpractice Legislative Subcommittee for the Virginia Trial Lawyers Association. “I cannot say that such things never happen, but any lawyer bringing such a thing is foolish, because there is no reward for it.”
Are shotgun lawsuits frivolous?
To many physicians, being dragged into a lawsuit over a complaint or medical outcome in which they were not involved is frivolous, said Stanislaw Stawicki, MD, a trauma surgeon and researcher based in Bethlehem, Pa. Dr. Stawicki was named in a lawsuit along with a long list of medical staff who interacted in some way with the plaintiff. Dr. Stawicki himself saw the patient once and made a note in the chart but had nothing to do with the patient’s surgery or with any critical decisions regarding his care, he said.
“Nothing really prepares you for seeing your name on a legal complaint,” Dr. Stawicki said. “It’s traumatic. I had to block out entire days to give depositions, which were really kind of pointless. Questions like, ‘Is this really your name? Where did you train? Were you there that morning?’ Stuff that was really not consequential to the fact that someone had surgery a month earlier and had some sort of complication.”
Dr. Stawicki was eventually dropped from the claim, but not before a nearly year-long ordeal of legal proceedings, meetings, and paperwork.
It is common practice for plaintiffs’ attorneys to add codefendants in the early stages of a claim, said David M. Studdert, ScD, a leading health law researcher and a professor of law at Stanford (Calif.) Law School. Defendants are gradually dismissed as the case moves forward and details of the incident become clearer, he said.
“Plaintiffs’ attorneys have strong incentives to try and choose claims that will be successful,” Dr. Studdert said. “However, in the early point in the process, neither the patient nor the attorney may have a good idea what has actually happened with care. So sometimes, filing a lawsuit may be the only way to begin the process of opening up that information.”
A study by Dr. Studdert in which medical malpractice claims, errors, and compensation payments were analyzed found that, out of 1,452 claims, about one-third (37%) did not involve errors.
“Many physicians might call those frivolous lawsuits, but in fact, most of those don’t go on to receive compensation,” he said. “We suspect that in many instances, those claims are simply dropped once it becomes apparent that there wasn’t error involved.
“They can still be burdensome, anxiety provoking, and time consuming for physicians who are named in those suits, so I don’t want to suggest that claims that don’t involve errors are not a problem,” said Dr. Studdert. “However, I think it’s wrong to assume, as many people do when they use the term ‘frivolous lawsuit,’ that this is really an extortionary effort by a plaintiffs’ attorney to try to get money out of a hospital or a physician for care that was really unproblematic.”
Certain ‘frivolous’ cases more common than others
Nonmeritorious claims still occur relatively frequently today, according to data from the Medical Professional Liability Association’s Data Sharing Project. Of about 18,000 liability claims reported from 2016 to 2018, 65% were dropped, withdrawn, or dismissed. Of the 6% of claims that went before a jury, more than 85% resulted in a verdict for the defendant, the researchers found.
“Basically, any claim that does not result in a payment because the underlying claim of negligence on the part of a health professional had been demonstrated, proven, or adjudicated false is one we would describe as nonmeritorious,” Mr. Stinson said.
The MPL Association does not track cases that meet the legal definition of frivolous, said Mr. Stinson, and they “don’t see truly frivolous lawsuits very often.”
Malpractice claims are risky, expensive, and aggressively defended, says Mr. McConnell, the plaintiffs’ attorney. Mr. McConnell, who has been practicing for 30 years, said his own claim selection process is very rigorous and that he cannot afford to pursue claims that aren’t well supported by science and medicine.
“Pursuing frivolous cases would bankrupt me and ruin my reputation,” he said. “A lawyer I know once said he would write a check for $10,000 to anyone who could show him a lawyer who makes a living pursuing frivolous medical malpractice cases. It’s a fair challenge. The economics and the practices of liability carriers and defense lawyers make frivolous cases a dead end for plaintiff lawyers.”
Most medical malpractice cases are taken on a contingency fee basis, Mr. McConnell noted, meaning that the plaintiff’s lawyer is not paid unless the claim is successful.
“This means that the plaintiff’s lawyer is risking 2 years of intensive labor on a case which may yield no fee at all,” he said. “Obviously, any reasonable lawyer is going to want to minimize that risk. The only way to minimize that risk is for the case to be solid, not weak, and certainly not frivolous.”
But Dr. Segal, the health law attorney, says that plenty of frivolous liability claims are levied each year, with attorneys willing to pursue them.
It’s true that seasoned plaintiffs’ attorneys generally screen for merit and damages, Dr. Segal said, but in some instances, attorneys who are not trained in malpractice law accept frivolous claims and take them forward. In some cases, they are slip-and-fall accident attorneys accustomed to receiving modest amounts from insurance companies quickly, said Dr. Segal, founder of Medical Justice, a company that helps deter frivolous lawsuits against physicians.
“If we lived in a perfectly rational universe where plaintiffs’ attorneys screened cases well and only took the meritorious cases forward, we would see less frivolous cases filed, but that’s not the universe I live in,” Dr. Segal said. “There are well over a million attorneys in this country, and some are hungrier than others. The attorneys may frequently get burned in the end, and maybe that attorney won’t move another malpractice case forward, but there’s always someone else willing to take their place.”
Medical Justice has twice run a Most Frivolous Lawsuit Contest on its website, one in 2008 and one in late 2018. The first contest drew 30 entries, and the second garnered nearly 40 submissions, primarily from physicians who were defendants in the cases, according to Dr. Segal. (Dr. G’s lawsuit was highlighted in the most recent contest.)
In one case, an emergency physician was drawn into litigation by the family of a deceased patient. The patient experienced sudden cardiac arrythmia at home, and paramedics were unable to intubate her or establish IV access. She was transferred to the hospital, where resuscitation efforts continued, but she remained in asystole and was pronounced dead after 15 minutes.
At the hospital, blood tests were conducted. They showed that her serum potassium concentration was elevated to about 12 mEq/L, Dr. Segal said. The family initiated a claim in which they accused the emergency physician of failure to diagnose hyperkalemia. They alleged that had the hyperkalemia been discovered sooner, the patient’s death could have been prevented.
“If you had no other facts about this, you would wonder how a person with potassium that high would even be alive,” Dr. Segal said. “But what they were looking at was the body decomposing and all the potassium in the cells being released into the bloodstream. It wasn’t the cause of the problem, it was an effect of the problem. She really was dead on arrival, and she was probably dead at home.”
The case was eventually dropped.
Although the outcome for the patient was tragic, says Dr. Segal, the case is one of many types of frivolous claims that exist today.
“Yes, frivolous cases are out there,” he said.
Fraudulent claims uncommon
As for fraudulent medical liability claims, legal experts say they’re rare. J. Richard Moore, JD, an Indianapolis-based medical liability defense attorney, said he’s never personally encountered a medical malpractice claim in which he believed a plaintiff caused an injury or an illness and attempted to blame it on a physician.
However, Mr. Moore has defended many claims in which the illness or condition the plaintiff claimed was caused or was made worse through medical negligence was actually a preexisting condition or a preexisting condition that worsened and was not related to any medical negligence, Mr. Moore said.
“Although I have often felt in such cases that the plaintiff really knew that the condition was not affected by any alleged medical negligence, I would not put that in the ‘fraudulent claim’ category because it can be very difficult to establish a person’s subjective state of mind,” he said. “Usually in those cases, the plaintiff just denies memory of previous medical records or claims that the previous doctor who treated him or her for the same condition ‘got it wrong.’ In those cases, it is generally left to the jury whether to believe the plaintiff or not.”
Mr. Stinson also says he has not come across a truly fraudulent medical liability case. He noted that such a claim might be similar to a person falsely claiming a soft-tissue injury following an alleged slip-and-fall accident.
“Clearly, a fraudulent claim could be viewed as riskier from the plaintiff’s perspective because they could face criminal prosecution for insurance fraud, whereas if a claim is merely frivolous, they probably only run the risk of court-issued fine, if even that. That may be why we don’t often see fraudulent MPL claims.”
Ways to prevent or fight frivolous lawsuits
Since Dr. Stawicki’s legal nightmare as a resident, rules have tightened in Pennsylvania, and it is now more difficult to file frivolous claims, he said.
Pennsylvania is one of at least 28 states that require a certificate of merit in order for a medical liability claim to move forward. The provisions generally state that an appropriately licensed professional must supply a written statement attesting that the care the patient received failed to meet acceptable professional standards and that such conduct was a cause in the alleged harm.
“There is now a much greater burden of proof regarding what can proceed,” Dr. Stawicki said. “I’ve been involved in a couple cases that did not proceed because there was no certificate of merit.”
Although these reforms may help, not all merit rules are created equal. Some states require that the expert who signs the affidavit be knowledgeable in the relevant issues involved in the action. Other states have looser requirements. In one of the cases featured in Medical Justice’s Most Frivolous Lawsuit Contest, a podiatrist signed a supporting declaration for a claim related to obstetric care.
For physicians facing a frivolous claim, fighting it out in court depends on a number of factors. Without a consent-to-settle clause in the contract, an insurer can make the final decision on whether to defend or settle a case.
Resolving a malpractice claim is generally a business decision for the insurer, Dr. Studdert said.
“When the claim is for a relatively low amount of money, the costs of moving forward to defend that claim may be much more than the costs of simply settling it would be,” he said. “On the other hand, liability insurers and their lawyers are repeat players here, as are the plaintiffs’ attorneys. They don’t want to incentivize plaintiffs’ attorneys to bring questionable claims, and if they settle quickly, that may do so.”
Mr. Stinson, of the MPL Association, said a truly frivolous claim – one with no legal basis – is highly unlikely to be settled, “especially by MPL Association members who go beyond having a purely financial interest in their insureds to also focus on their professional reputation/integrity.” MPL Association members insure nearly 2 million health care professionals globally, including 2,500 hospitals and more than two-thirds of America’s physicians who are in private practice.
Physicians should make sure they know what is and what is not included in their policy, Dr. Segal said.
“The broker should sit down with the doctor, ideally before initial purchase or renewal, and explain in clear terms what the carrier’s obligations are and what the physician’s obligations are,” he said. “Know what type of protection is being purchased and what conditions might trigger a surprising and unhappy outcome.”
Should I countersue?
For truly frivolous claims, physicians have the legal right to sue for damages caused by the unfounded complaint.
Perhaps the most well-known case of a successful malpractice countersuit is that of Louisville neurosurgeon John Guarnaschelli, MD, who in 2000 won $72,000 in damages against a plaintiffs’ attorney for malicious prosecution.
The physician’s countersuit followed the dismissal of a negligence claim against Dr. Guarnaschelli by a patient who contracted meningitis. The plaintiffs’ attorney had made little effort to gather evidence to connect Dr. Guarnaschelli to the patient’s injuries and had consulted only one other physician, a client of his, before filing the lawsuit, according to a summary of the case in the American Bar Association Journal.
Malicious prosecution is the most common legal theory of recovery for physicians in countersuits, according to a review of successful countersuits by doctors. Dr. Stawicki is a coauthor of that review. Other legal theories that physicians can raise include abuse of process, negligence, defamation, invasion of privacy, and infliction of emotional distress. Of the 13 cases evaluated in the article by Dr. Stawicki and colleagues, damages awarded to physicians ranged from about $13,000 to $125,000.
Although some doctors have success, pursuing a counterclaim can be a difficult feat, said Benjamin Braslow, MD, a trauma surgeon and professor of clinical surgery at the University of Pennsylvania in Philadelphia.
“The main takeaways were it’s an uphill battle often met with not only resistance but diminishing returns to countersue,” said Dr. Braslow, a coauthor of the countersuits analysis. “You have to meet very specific criteria regarding leveling the suit, and it may end up being a costly, time-consuming battle.”
To prove malicious prosecution, for example, a physician must show that a claim was instituted without probable cause, that the suing party acted maliciously in instituting the action, and that the doctor was damaged by the action, among other essential elements.
As for Dr. G, the surgeon, he now has a contract with a consent-to-settle clause and has taken other legal precautions since his lawsuits. He requires that his patients sign an agreement that any negligence claims they levy go to arbitration. If an arbitrator finds in the patient’s favor, the case may proceed to court, he said. However, he requires another agreement such that if patients lose in court, they are responsible for his legal fees.
“I’m just more careful,” he said. “I ask all my staff in the office to use their judgment, however superficial, if they feel something is wrong with an individual to tell me so. I’d rather send them away than operate on them and have it result in a lawsuit.”
A version of this article originally appeared on Medscape.com.
Hospitalists are natural leaders in the COVID-19 battle
Christopher Pribula, MD, a hospitalist at Sanford Broadway Medical Center in Fargo, N.D., didn’t anticipate becoming his hospital’s resident expert on COVID-19. Having just returned from vacation in March, he agreed to cover for a colleague on what would become the special care unit. “When our hospital medicine group decided that it would be the COVID unit, I just ran with it,” he said. Dr. Pribula spent the next 18 days doing 8- to 14-hour shifts and learning as much as he could as the hospital – and the nation – wrestled with the pandemic.
“Because I was the first hospitalist, along with our infectious disease specialist, Dr. Avish Nagpal, to really engage with the virus, people came to me with their questions,” Dr. Pribula said. Working to establish protocols for the care of COVID-19 patients involved a lot of planning, from nursing protocols to discharge planning.
Dr. Pribula was part of the hospital’s incident command structure, thought about how the system could scale up for a potential surge, and worked with the North Dakota Medical Association to reach out to outlying medical centers on safety and infection control. He even drew on his prior work experience as a medical technologist doing negative-pressure containment in a cell-processing facility to help create the hospital’s negative-pressure unit in an old ICU.
“We did a lot of communication from the start. To a certain extent we were making it up as we went along, but we sat down and huddled as a team every day at 9 and 4,” he explained. “We started out with observation and retrospective research, and learned piece by piece. But that’s how science works.”
Hospitalists across the country have played leading roles in their hospitals’ and health systems’ response to the pandemic, and not just because they are on the front lines providing patient care. Their job as doctors who work full-time in the hospital makes them natural leaders in improving clinical quality and hospital administrative protocols as well as studying the latest information and educating their colleagues. Responding to the pandemic has required lots of planning, careful attention to schedules and assignments and staff stress, and working with other departments in the hospital and groups in the community, including public health authorities.
Where is hospital treatment for COVID-19 at today?
As knowledge has grown, Dr. Pribula said, COVID-19 treatment in the hospital has come to incorporate remdesivir, a broad-spectrum antiviral; dexamethasone, a common steroid medication; and convalescent plasma, blood products from people who have recovered from the illness. “We went from no steroids to giving steroids. We went from putting patients on ventilators to avoid acute respiratory distress syndrome (ARDS) initially to now working to avoid intubation at all costs,” he said.
“What we found is that we need to pressure-support these patients. We do proning and CPAP while we let the lungs heal. By the time they arrive at the hospital, more often than not they’re on the backside of the viral load. But now we’re dealing with the body’s inflammatory response.”
Navneet Attri, MD, a hospitalist at Sutter Santa Rosa Regional Hospital in Santa Rosa, Calif., 50 miles north of San Francisco, experienced fears and uncertainties working at a hospital that treated early COVID patients from the Grand Princess cruise ship. Early on, she wrote a post describing her experience for The Hospitalist Leader, the Society of Hospital Medicine’s blog page.
Dr. Attri said she has gone through the gamut of emotions while caring for COVID-19 patients, addressing their fears and trying to support family members who aren’t allowed to enter the hospital to be at their loved one’s side. Sometimes, patient after patient with COVID-19 becomes almost too much. But seeing a lot of them in the intervening 6 months has increased her confidence level.
Understanding of how the disease is spread has continued to evolve, with a recent return to focusing on airborne transmission, she said. Frontline workers need N95 masks and eye shields, even if all of that PPE feels like a burden. Dr. Attri said she hardly notices the PPE anymore. “Putting it on is just a habit.”
She sits on Sonoma County’s COVID-19 surge planning group, which has representatives from the three local hospitals, the public health department, and other community agencies. “I report back to my hospitalist group about the situation in the community. Because our facilities were well prepared, our hospitals have not been overwhelmed,” she said.
The importance of teamwork
Sunil Shah, MD, a hospitalist with Northwell Health’s Southside Hospital in Bay Shore, N.Y., is part of the massive hospital medicine team, including reassigned specialists and volunteers from across the country, deployed at Northwell hospitals in Greater New York City and Long Island during the COVID-19 surge. Northwell probably has cared for more COVID-19 patients than any other health system in the country, and at the height of the surge the intensity of hospital care was like nothing he’s ever seen. But he also expressed gratitude that doctors from other parts of the country were willing to come and help out.
Southside Hospital went almost overnight from a 200-bed acute facility to a full, 350-bed, regional COVID-19–only hospital. “On busy days, our entire hospital was like a floating ICU,” he said. “You’d hear ‘rapid response’ or ‘code blue’ over the intercom every few seconds. Normally we’d have a designated rapid response person for the day, but with COVID, everybody stepped in to help – whoever was closest,” he said.
Majid Sheikh, MD, a hospitalist at Emory University Hospital in Atlanta, also became a go-to COVID-19 expert for his group. “I didn’t specifically volunteer, but my partner and I had the first cases, and the leadership group was happy to have us there,” he explained.
“One interesting thing I learned was the concept of the ‘happy’ hypoxemic patient, who is having a significant drop in oxygen saturation without developing any obvious signs of respiratory distress,” he said. “We’d be checking the accuracy of the reading and trying to figure out if it was real.” Emory was also one of the leaders in studying anticoagulant treatments for COVID-19 patients.
“Six months later I would say we’re definitely getting better outcomes on the floor, and our COVID patients aren’t landing in the ICU as easily,” Dr. Sheikh said. “It was scary at first, and doubly scary when doctors sometimes don’t feel they can say, ‘Hey, I’m scared too,’ or ‘By the way, I really don’t know what I’m doing.’ So, we’d be trying to reassure the patients when the information was coming to us in fragments.”
But he also believes that the pandemic has afforded hospitalists the opportunity to be the clinical detectives they were trained to be, sifting through clues. “I had to think more and really pay attention clinically in a much different way. You could say it was exciting and scary at the same time,” he said.
A human fix in the hospital
Dr. Pribula agreed that the pandemic has been both a difficult experience and a rewarding one. “I think of the people I first admitted. If they had shown up even a month later, would they still be with us?” He believes that his group and his field are going to get to a place where they have solid treatment plans for how to provide optimal care and how to protect providers from exposure.
One of the first COVID-19 patients in Fargo had dementia and was very distressed. “She had no idea why nobody was visiting or why we wouldn’t let her out of her room,” Dr. Pribula said. “Instead of reaching for sedatives, one of our nurses went into the room and talked with her, prayed a rosary, and played two hands of cards with her and didn’t have to sedate her. That’s what people need when they’re alone and scared. It wasn’t a medical fix but a human fix.”
A version of this article originally appeared on Medscape.com.
Christopher Pribula, MD, a hospitalist at Sanford Broadway Medical Center in Fargo, N.D., didn’t anticipate becoming his hospital’s resident expert on COVID-19. Having just returned from vacation in March, he agreed to cover for a colleague on what would become the special care unit. “When our hospital medicine group decided that it would be the COVID unit, I just ran with it,” he said. Dr. Pribula spent the next 18 days doing 8- to 14-hour shifts and learning as much as he could as the hospital – and the nation – wrestled with the pandemic.
“Because I was the first hospitalist, along with our infectious disease specialist, Dr. Avish Nagpal, to really engage with the virus, people came to me with their questions,” Dr. Pribula said. Working to establish protocols for the care of COVID-19 patients involved a lot of planning, from nursing protocols to discharge planning.
Dr. Pribula was part of the hospital’s incident command structure, thought about how the system could scale up for a potential surge, and worked with the North Dakota Medical Association to reach out to outlying medical centers on safety and infection control. He even drew on his prior work experience as a medical technologist doing negative-pressure containment in a cell-processing facility to help create the hospital’s negative-pressure unit in an old ICU.
“We did a lot of communication from the start. To a certain extent we were making it up as we went along, but we sat down and huddled as a team every day at 9 and 4,” he explained. “We started out with observation and retrospective research, and learned piece by piece. But that’s how science works.”
Hospitalists across the country have played leading roles in their hospitals’ and health systems’ response to the pandemic, and not just because they are on the front lines providing patient care. Their job as doctors who work full-time in the hospital makes them natural leaders in improving clinical quality and hospital administrative protocols as well as studying the latest information and educating their colleagues. Responding to the pandemic has required lots of planning, careful attention to schedules and assignments and staff stress, and working with other departments in the hospital and groups in the community, including public health authorities.
Where is hospital treatment for COVID-19 at today?
As knowledge has grown, Dr. Pribula said, COVID-19 treatment in the hospital has come to incorporate remdesivir, a broad-spectrum antiviral; dexamethasone, a common steroid medication; and convalescent plasma, blood products from people who have recovered from the illness. “We went from no steroids to giving steroids. We went from putting patients on ventilators to avoid acute respiratory distress syndrome (ARDS) initially to now working to avoid intubation at all costs,” he said.
“What we found is that we need to pressure-support these patients. We do proning and CPAP while we let the lungs heal. By the time they arrive at the hospital, more often than not they’re on the backside of the viral load. But now we’re dealing with the body’s inflammatory response.”
Navneet Attri, MD, a hospitalist at Sutter Santa Rosa Regional Hospital in Santa Rosa, Calif., 50 miles north of San Francisco, experienced fears and uncertainties working at a hospital that treated early COVID patients from the Grand Princess cruise ship. Early on, she wrote a post describing her experience for The Hospitalist Leader, the Society of Hospital Medicine’s blog page.
Dr. Attri said she has gone through the gamut of emotions while caring for COVID-19 patients, addressing their fears and trying to support family members who aren’t allowed to enter the hospital to be at their loved one’s side. Sometimes, patient after patient with COVID-19 becomes almost too much. But seeing a lot of them in the intervening 6 months has increased her confidence level.
Understanding of how the disease is spread has continued to evolve, with a recent return to focusing on airborne transmission, she said. Frontline workers need N95 masks and eye shields, even if all of that PPE feels like a burden. Dr. Attri said she hardly notices the PPE anymore. “Putting it on is just a habit.”
She sits on Sonoma County’s COVID-19 surge planning group, which has representatives from the three local hospitals, the public health department, and other community agencies. “I report back to my hospitalist group about the situation in the community. Because our facilities were well prepared, our hospitals have not been overwhelmed,” she said.
The importance of teamwork
Sunil Shah, MD, a hospitalist with Northwell Health’s Southside Hospital in Bay Shore, N.Y., is part of the massive hospital medicine team, including reassigned specialists and volunteers from across the country, deployed at Northwell hospitals in Greater New York City and Long Island during the COVID-19 surge. Northwell probably has cared for more COVID-19 patients than any other health system in the country, and at the height of the surge the intensity of hospital care was like nothing he’s ever seen. But he also expressed gratitude that doctors from other parts of the country were willing to come and help out.
Southside Hospital went almost overnight from a 200-bed acute facility to a full, 350-bed, regional COVID-19–only hospital. “On busy days, our entire hospital was like a floating ICU,” he said. “You’d hear ‘rapid response’ or ‘code blue’ over the intercom every few seconds. Normally we’d have a designated rapid response person for the day, but with COVID, everybody stepped in to help – whoever was closest,” he said.
Majid Sheikh, MD, a hospitalist at Emory University Hospital in Atlanta, also became a go-to COVID-19 expert for his group. “I didn’t specifically volunteer, but my partner and I had the first cases, and the leadership group was happy to have us there,” he explained.
“One interesting thing I learned was the concept of the ‘happy’ hypoxemic patient, who is having a significant drop in oxygen saturation without developing any obvious signs of respiratory distress,” he said. “We’d be checking the accuracy of the reading and trying to figure out if it was real.” Emory was also one of the leaders in studying anticoagulant treatments for COVID-19 patients.
“Six months later I would say we’re definitely getting better outcomes on the floor, and our COVID patients aren’t landing in the ICU as easily,” Dr. Sheikh said. “It was scary at first, and doubly scary when doctors sometimes don’t feel they can say, ‘Hey, I’m scared too,’ or ‘By the way, I really don’t know what I’m doing.’ So, we’d be trying to reassure the patients when the information was coming to us in fragments.”
But he also believes that the pandemic has afforded hospitalists the opportunity to be the clinical detectives they were trained to be, sifting through clues. “I had to think more and really pay attention clinically in a much different way. You could say it was exciting and scary at the same time,” he said.
A human fix in the hospital
Dr. Pribula agreed that the pandemic has been both a difficult experience and a rewarding one. “I think of the people I first admitted. If they had shown up even a month later, would they still be with us?” He believes that his group and his field are going to get to a place where they have solid treatment plans for how to provide optimal care and how to protect providers from exposure.
One of the first COVID-19 patients in Fargo had dementia and was very distressed. “She had no idea why nobody was visiting or why we wouldn’t let her out of her room,” Dr. Pribula said. “Instead of reaching for sedatives, one of our nurses went into the room and talked with her, prayed a rosary, and played two hands of cards with her and didn’t have to sedate her. That’s what people need when they’re alone and scared. It wasn’t a medical fix but a human fix.”
A version of this article originally appeared on Medscape.com.
Christopher Pribula, MD, a hospitalist at Sanford Broadway Medical Center in Fargo, N.D., didn’t anticipate becoming his hospital’s resident expert on COVID-19. Having just returned from vacation in March, he agreed to cover for a colleague on what would become the special care unit. “When our hospital medicine group decided that it would be the COVID unit, I just ran with it,” he said. Dr. Pribula spent the next 18 days doing 8- to 14-hour shifts and learning as much as he could as the hospital – and the nation – wrestled with the pandemic.
“Because I was the first hospitalist, along with our infectious disease specialist, Dr. Avish Nagpal, to really engage with the virus, people came to me with their questions,” Dr. Pribula said. Working to establish protocols for the care of COVID-19 patients involved a lot of planning, from nursing protocols to discharge planning.
Dr. Pribula was part of the hospital’s incident command structure, thought about how the system could scale up for a potential surge, and worked with the North Dakota Medical Association to reach out to outlying medical centers on safety and infection control. He even drew on his prior work experience as a medical technologist doing negative-pressure containment in a cell-processing facility to help create the hospital’s negative-pressure unit in an old ICU.
“We did a lot of communication from the start. To a certain extent we were making it up as we went along, but we sat down and huddled as a team every day at 9 and 4,” he explained. “We started out with observation and retrospective research, and learned piece by piece. But that’s how science works.”
Hospitalists across the country have played leading roles in their hospitals’ and health systems’ response to the pandemic, and not just because they are on the front lines providing patient care. Their job as doctors who work full-time in the hospital makes them natural leaders in improving clinical quality and hospital administrative protocols as well as studying the latest information and educating their colleagues. Responding to the pandemic has required lots of planning, careful attention to schedules and assignments and staff stress, and working with other departments in the hospital and groups in the community, including public health authorities.
Where is hospital treatment for COVID-19 at today?
As knowledge has grown, Dr. Pribula said, COVID-19 treatment in the hospital has come to incorporate remdesivir, a broad-spectrum antiviral; dexamethasone, a common steroid medication; and convalescent plasma, blood products from people who have recovered from the illness. “We went from no steroids to giving steroids. We went from putting patients on ventilators to avoid acute respiratory distress syndrome (ARDS) initially to now working to avoid intubation at all costs,” he said.
“What we found is that we need to pressure-support these patients. We do proning and CPAP while we let the lungs heal. By the time they arrive at the hospital, more often than not they’re on the backside of the viral load. But now we’re dealing with the body’s inflammatory response.”
Navneet Attri, MD, a hospitalist at Sutter Santa Rosa Regional Hospital in Santa Rosa, Calif., 50 miles north of San Francisco, experienced fears and uncertainties working at a hospital that treated early COVID patients from the Grand Princess cruise ship. Early on, she wrote a post describing her experience for The Hospitalist Leader, the Society of Hospital Medicine’s blog page.
Dr. Attri said she has gone through the gamut of emotions while caring for COVID-19 patients, addressing their fears and trying to support family members who aren’t allowed to enter the hospital to be at their loved one’s side. Sometimes, patient after patient with COVID-19 becomes almost too much. But seeing a lot of them in the intervening 6 months has increased her confidence level.
Understanding of how the disease is spread has continued to evolve, with a recent return to focusing on airborne transmission, she said. Frontline workers need N95 masks and eye shields, even if all of that PPE feels like a burden. Dr. Attri said she hardly notices the PPE anymore. “Putting it on is just a habit.”
She sits on Sonoma County’s COVID-19 surge planning group, which has representatives from the three local hospitals, the public health department, and other community agencies. “I report back to my hospitalist group about the situation in the community. Because our facilities were well prepared, our hospitals have not been overwhelmed,” she said.
The importance of teamwork
Sunil Shah, MD, a hospitalist with Northwell Health’s Southside Hospital in Bay Shore, N.Y., is part of the massive hospital medicine team, including reassigned specialists and volunteers from across the country, deployed at Northwell hospitals in Greater New York City and Long Island during the COVID-19 surge. Northwell probably has cared for more COVID-19 patients than any other health system in the country, and at the height of the surge the intensity of hospital care was like nothing he’s ever seen. But he also expressed gratitude that doctors from other parts of the country were willing to come and help out.
Southside Hospital went almost overnight from a 200-bed acute facility to a full, 350-bed, regional COVID-19–only hospital. “On busy days, our entire hospital was like a floating ICU,” he said. “You’d hear ‘rapid response’ or ‘code blue’ over the intercom every few seconds. Normally we’d have a designated rapid response person for the day, but with COVID, everybody stepped in to help – whoever was closest,” he said.
Majid Sheikh, MD, a hospitalist at Emory University Hospital in Atlanta, also became a go-to COVID-19 expert for his group. “I didn’t specifically volunteer, but my partner and I had the first cases, and the leadership group was happy to have us there,” he explained.
“One interesting thing I learned was the concept of the ‘happy’ hypoxemic patient, who is having a significant drop in oxygen saturation without developing any obvious signs of respiratory distress,” he said. “We’d be checking the accuracy of the reading and trying to figure out if it was real.” Emory was also one of the leaders in studying anticoagulant treatments for COVID-19 patients.
“Six months later I would say we’re definitely getting better outcomes on the floor, and our COVID patients aren’t landing in the ICU as easily,” Dr. Sheikh said. “It was scary at first, and doubly scary when doctors sometimes don’t feel they can say, ‘Hey, I’m scared too,’ or ‘By the way, I really don’t know what I’m doing.’ So, we’d be trying to reassure the patients when the information was coming to us in fragments.”
But he also believes that the pandemic has afforded hospitalists the opportunity to be the clinical detectives they were trained to be, sifting through clues. “I had to think more and really pay attention clinically in a much different way. You could say it was exciting and scary at the same time,” he said.
A human fix in the hospital
Dr. Pribula agreed that the pandemic has been both a difficult experience and a rewarding one. “I think of the people I first admitted. If they had shown up even a month later, would they still be with us?” He believes that his group and his field are going to get to a place where they have solid treatment plans for how to provide optimal care and how to protect providers from exposure.
One of the first COVID-19 patients in Fargo had dementia and was very distressed. “She had no idea why nobody was visiting or why we wouldn’t let her out of her room,” Dr. Pribula said. “Instead of reaching for sedatives, one of our nurses went into the room and talked with her, prayed a rosary, and played two hands of cards with her and didn’t have to sedate her. That’s what people need when they’re alone and scared. It wasn’t a medical fix but a human fix.”
A version of this article originally appeared on Medscape.com.
COVID and med ed cost: Are future docs paying more for less?
Like most medical students, Kaitlyn Thomas’s education was abruptly interrupted by the pandemic. Her school, an osteopathic medicine institution in the Midwest, followed guidelines issued by the American Association of Medical Colleges in March, shifting lectures online and suspending activities in which students interacted with patients. But even as Ms. Thomas’s learning opportunities dwindled for the sake of safety, the costs kept piling up.
Instead of going home to live with her family, she stayed in her apartment near school – and kept paying rent – so she could be nearby for the two licensing exams she was scheduled to take 3 months later. Both tests were canceled 9 days before she was scheduled to take them, one without any notification. This meant she had to travel to two different testing sites in two different states. All told, she said, the whole thing cost her around $2,000.
Ms. Thomas’s experience isn’t rare. Across the country, medical students find themselves paying substantial costs for a medical education now greatly altered by the pandemic. Despite restrictions on time spent in hospitals, hands-on learning, social events, and access to libraries, gyms, study spaces, and instructors, the price of tuition hasn’t dropped but has remained the same or has even risen.
In response, students have become vocal about the return on their pricey investment. “Am I just going to end up doing most of my year online, and what does that look like for my future patients?” Ms. Thomas asked. “It really doesn’t feel like a time to be limiting education.”
Medical schools and administrators are scrambling to find creative solutions for safely educating students. No matter what those solutions may be, experts say, the pandemic has drawn fresh attention to enduring questions about how the cost of medical education compares to its value. Although many are frustrated, some see the potential for COVID to open new opportunities for lasting innovation. At the very least, the pandemic has sparked conversations about what matters most in terms of producing qualified physicians.
“While this is a challenging time, we will get through it, and we will continue to educate doctors, and we will get them through to practice,” says Robert Cain, president and CEO of the American Association of Colleges of Osteopathic Medicine. Many in the midst of training still have one lingering question: Is the price future doctors are now paying still worth it?
COVID’s “hidden costs” for students
Tom is a third-year student at an allopathic medicine institution in the Caribbean. He asked not to be fully identified here, owing to concern about possible backlash. In March, Tom was doing clinical rotations in New York City when his training was put on hold. He returned home to Connecticut and resumed working 60-80 hours a week as a paramedic. As much as 75% of that income went to pay for the New York City apartment he was no longer living in – an apartment that cost more than $2,000 a month – and for student loans that suddenly came due when his enrollment status changed.
Tom has been able to take some online courses through his school. But he still doesn’t know whether state licensing boards will accept them, how residency programs will view them, or whether he will eventually have to retake those online classes in person. At the end of September, he was allowed to return to the hospital but was relocated to Chicago and was forced to move on short notice.
Like many students, Tom has worried that the pandemic may prevent him from acquiring crucial elements for his residency applications, things like letters of recommendation or key experiences. That could delay his next stage of training, which would mean lost future income, increasing student loan interest, and lost work experience. “This could also mean the difference between getting a residency and being able to practice medicine and not being able to practice my intended specialty,” he said. “This is the real hidden cost we may have to deal with.”
International medical students hoping to practice in the United States face additional costs. Michelle Warncke earned her bachelor’s degree in America but went to the United Kingdom for her master’s and her medical degree, which she completed in 2019. She then moved to North Carolina with her husband and saved money to take the exams she needed for residency in the states. But her scheduled Step 2 CS exam was canceled because of the pandemic. Now, like hundreds or even thousands of other students, she said she is unable to apply for residency, even as her student loans collect interest. An active Facebook group of international medical graduates includes about 1,500 people with comparable dilemmas.
The path to becoming a physician carries a well-known price tag, one that is already quite high. Now, for many, that price is substantially increasing. “The only way I can actually keep my medical credentials up to date and passable, to be able to ever get a shot at a residency in the following years,” she said, “is to move to another country and work for less pay, pay for a visa, pay for my exams, pay for my language test, and wait and hope that I might be able to as an older graduate then be able to apply for residency.”
Scaling back the price of med school?
Questions about the economics of medical education aren’t new, says David Asch, MD, MBA, an internal medicine physician and executive director of the Center for Health Care Innovation at the University of Pennsylvania, Philadelphia. But the changes forced by COVID could lead to innovations that may finally better balance the financial scales.
Such innovations are necessary, many say, given how medical education costs have skyrocketed over the past half century. In the 1960s, 4 years of medical school cost about $40,000 in today’s dollars, Dr. Asch and colleagues wrote in a 2020 analysis, which they conducted before the pandemic began. By 2018, the price of a medical education in the United States had ballooned to about $300,000. About 75% of students were taking out loans. Upon graduating, the average debt was $200,000.
Medical school is expensive for many tangible reasons, Dr. Asch said. Schools must pay for curriculum, faculty, technology, textbooks, lab materials, facilities, administrators, and more. But policy changes could decrease those costs.
He says one idea would be for medical schools to join forces and give students access to the same basic lectures in the early years, delivered online by top-notch instructors. Students could then participate in on-campus programs that might only require 3 years to complete instead of 4. By demonstrating what can be done via online platforms, he said, the pandemic might pave the way to permanent changes that could reduce costs.
“I’m not trying to pick on biochemistry professors and medical schools, but how many do we need in the country?” Dr. Asch asked. “We’re all watching the same episode of Seinfeld. Why can’t we all watch the same episode of the Krebs cycle?” If all 190 or so medical schools in the United States shared such preclinical courses, he says, each would require a fraction of the current cost to produce. “We could save 99.5% of the cost. So why don’t we do that?”
Pandemic as opportunity
Although the price of medical education has yet to decrease, schools are working to leverage the pandemic to provide increased educational value.
This generation of physicians will not only have to cope with the fallout of this pandemic, they will be the ones responsible for confronting the next pandemic as well, says Donald Brady, MD, senior associate dean for health sciences education at Vanderbilt University, Nashville, Tenn. “They will be the leaders in the future who will better be able to know how to handle it [a pandemic] because they were able to watch it and be part of it safely in the current circumstance.”
As much as possible, Vanderbilt is using the pandemic as an opportunity. As soon as it became clear that students couldn’t be involved in certain hands-on training, instructors developed a course about pandemics that included lectures on ethics, global health, systemic racism, and other topics. It also included experiential components of pandemic management, such as opportunities to work with patients through telehealth.
Students say they feel that they are getting less for their money and that they are paying for experiences that are no longer available, such as hands-on patient contact and community events. However, Dr. Brady said, schools have had to account for new expenses, including various now-required technologies and transitioning to courses online.
Some challenges can’t be solved with money alone. Medical schools across the country are working together to ensure that they are still adequately preparing students. Vanderbilt participates in an AAMC group that meets regularly and is also one of 37 institutions involved in an American Medical Association Consortium (AACOM). These groups discuss challenges, strategies, and opportunities for optimizing medical education during the pandemic.
Some institutions have come up with creative solutions. Ohio University’s Heritage College of Osteopathic Medicine, in Athens, Ohio, in collaboration with the Ohio Department of Health, launched a 4-week rotation for third-year students that focuses on public health. Harvard Medical School, Boston, was one of several schools that allowed students to graduate early in the spring. “We’re constantly talking to our colleagues and friends,” Dr. Brady said. “We learn from each other. There’s a lot of sharing going on.”
Other organizations are also working to make sure students ultimately get what they are paying for: a high-quality education. As soon as the pandemic began, the AACOM organized four working groups to address how schools could better use technology to deliver curricula and how students could participate in public health efforts, among other topics. “For the students, the part they don’t see and can’t really be aware of is all the things that happen behind the scenes,” Mr. Cain said. “People were working really hard to make sure that their education was still delivered, and delivered in a way that was going to assure a good product at the end.”
Ultimately, that product will be held to a rigid standard, said Geoffrey Young, the AAMC’s senior director for student affairs and programs. Medical schools must still meet standards of competency set by the liaison committee on medical education. Mr. Young says that even now those standards remain rigorous enough to ensure that medical students are learning what they need to know. “The core elements for competency may be slightly altered to address the realities that we’re experiencing because of COVID, but the core tenants of competencies will not change,” he said.
Even as conversations continue about what a medical education is worth, the pandemic is drawing new attention to the profession. No signs suggest that the value of tuition or a shift to more virtual offerings are scaring students away. Applications for medical schools were up 17% for the fall of 2021.
Brady expects the surge in interest to continue. “The increased focus and emphasis on public health, the increased focus and emphasis on health equity, the increased focus on the need for a more diverse physician workforce, the interest in basic science research around viruses, the interest in COVID itself – there are a lot of different elements that are setting us up for a potential boom in applications to medical school,” he said.
Beyond increasing interest, the pandemic may also finally force a reckoning on the disconnection between how schools think about costs and how students think about value, Dr. Asch said. “When students say: ‘I’m not getting as much from this,’ they’re saying, ‘you should price this according to its lower value.’ And when the medical schools are saying: ‘Oh, but it’s costing us so much more,’ they’re talking about pricing according to the cost. It’s like one group is speaking Latin and the other group is speaking Greek.” Perhaps, he said, COVID-related changes will finally get them speaking the same language.
This article first appeared on Medscape.com.
Like most medical students, Kaitlyn Thomas’s education was abruptly interrupted by the pandemic. Her school, an osteopathic medicine institution in the Midwest, followed guidelines issued by the American Association of Medical Colleges in March, shifting lectures online and suspending activities in which students interacted with patients. But even as Ms. Thomas’s learning opportunities dwindled for the sake of safety, the costs kept piling up.
Instead of going home to live with her family, she stayed in her apartment near school – and kept paying rent – so she could be nearby for the two licensing exams she was scheduled to take 3 months later. Both tests were canceled 9 days before she was scheduled to take them, one without any notification. This meant she had to travel to two different testing sites in two different states. All told, she said, the whole thing cost her around $2,000.
Ms. Thomas’s experience isn’t rare. Across the country, medical students find themselves paying substantial costs for a medical education now greatly altered by the pandemic. Despite restrictions on time spent in hospitals, hands-on learning, social events, and access to libraries, gyms, study spaces, and instructors, the price of tuition hasn’t dropped but has remained the same or has even risen.
In response, students have become vocal about the return on their pricey investment. “Am I just going to end up doing most of my year online, and what does that look like for my future patients?” Ms. Thomas asked. “It really doesn’t feel like a time to be limiting education.”
Medical schools and administrators are scrambling to find creative solutions for safely educating students. No matter what those solutions may be, experts say, the pandemic has drawn fresh attention to enduring questions about how the cost of medical education compares to its value. Although many are frustrated, some see the potential for COVID to open new opportunities for lasting innovation. At the very least, the pandemic has sparked conversations about what matters most in terms of producing qualified physicians.
“While this is a challenging time, we will get through it, and we will continue to educate doctors, and we will get them through to practice,” says Robert Cain, president and CEO of the American Association of Colleges of Osteopathic Medicine. Many in the midst of training still have one lingering question: Is the price future doctors are now paying still worth it?
COVID’s “hidden costs” for students
Tom is a third-year student at an allopathic medicine institution in the Caribbean. He asked not to be fully identified here, owing to concern about possible backlash. In March, Tom was doing clinical rotations in New York City when his training was put on hold. He returned home to Connecticut and resumed working 60-80 hours a week as a paramedic. As much as 75% of that income went to pay for the New York City apartment he was no longer living in – an apartment that cost more than $2,000 a month – and for student loans that suddenly came due when his enrollment status changed.
Tom has been able to take some online courses through his school. But he still doesn’t know whether state licensing boards will accept them, how residency programs will view them, or whether he will eventually have to retake those online classes in person. At the end of September, he was allowed to return to the hospital but was relocated to Chicago and was forced to move on short notice.
Like many students, Tom has worried that the pandemic may prevent him from acquiring crucial elements for his residency applications, things like letters of recommendation or key experiences. That could delay his next stage of training, which would mean lost future income, increasing student loan interest, and lost work experience. “This could also mean the difference between getting a residency and being able to practice medicine and not being able to practice my intended specialty,” he said. “This is the real hidden cost we may have to deal with.”
International medical students hoping to practice in the United States face additional costs. Michelle Warncke earned her bachelor’s degree in America but went to the United Kingdom for her master’s and her medical degree, which she completed in 2019. She then moved to North Carolina with her husband and saved money to take the exams she needed for residency in the states. But her scheduled Step 2 CS exam was canceled because of the pandemic. Now, like hundreds or even thousands of other students, she said she is unable to apply for residency, even as her student loans collect interest. An active Facebook group of international medical graduates includes about 1,500 people with comparable dilemmas.
The path to becoming a physician carries a well-known price tag, one that is already quite high. Now, for many, that price is substantially increasing. “The only way I can actually keep my medical credentials up to date and passable, to be able to ever get a shot at a residency in the following years,” she said, “is to move to another country and work for less pay, pay for a visa, pay for my exams, pay for my language test, and wait and hope that I might be able to as an older graduate then be able to apply for residency.”
Scaling back the price of med school?
Questions about the economics of medical education aren’t new, says David Asch, MD, MBA, an internal medicine physician and executive director of the Center for Health Care Innovation at the University of Pennsylvania, Philadelphia. But the changes forced by COVID could lead to innovations that may finally better balance the financial scales.
Such innovations are necessary, many say, given how medical education costs have skyrocketed over the past half century. In the 1960s, 4 years of medical school cost about $40,000 in today’s dollars, Dr. Asch and colleagues wrote in a 2020 analysis, which they conducted before the pandemic began. By 2018, the price of a medical education in the United States had ballooned to about $300,000. About 75% of students were taking out loans. Upon graduating, the average debt was $200,000.
Medical school is expensive for many tangible reasons, Dr. Asch said. Schools must pay for curriculum, faculty, technology, textbooks, lab materials, facilities, administrators, and more. But policy changes could decrease those costs.
He says one idea would be for medical schools to join forces and give students access to the same basic lectures in the early years, delivered online by top-notch instructors. Students could then participate in on-campus programs that might only require 3 years to complete instead of 4. By demonstrating what can be done via online platforms, he said, the pandemic might pave the way to permanent changes that could reduce costs.
“I’m not trying to pick on biochemistry professors and medical schools, but how many do we need in the country?” Dr. Asch asked. “We’re all watching the same episode of Seinfeld. Why can’t we all watch the same episode of the Krebs cycle?” If all 190 or so medical schools in the United States shared such preclinical courses, he says, each would require a fraction of the current cost to produce. “We could save 99.5% of the cost. So why don’t we do that?”
Pandemic as opportunity
Although the price of medical education has yet to decrease, schools are working to leverage the pandemic to provide increased educational value.
This generation of physicians will not only have to cope with the fallout of this pandemic, they will be the ones responsible for confronting the next pandemic as well, says Donald Brady, MD, senior associate dean for health sciences education at Vanderbilt University, Nashville, Tenn. “They will be the leaders in the future who will better be able to know how to handle it [a pandemic] because they were able to watch it and be part of it safely in the current circumstance.”
As much as possible, Vanderbilt is using the pandemic as an opportunity. As soon as it became clear that students couldn’t be involved in certain hands-on training, instructors developed a course about pandemics that included lectures on ethics, global health, systemic racism, and other topics. It also included experiential components of pandemic management, such as opportunities to work with patients through telehealth.
Students say they feel that they are getting less for their money and that they are paying for experiences that are no longer available, such as hands-on patient contact and community events. However, Dr. Brady said, schools have had to account for new expenses, including various now-required technologies and transitioning to courses online.
Some challenges can’t be solved with money alone. Medical schools across the country are working together to ensure that they are still adequately preparing students. Vanderbilt participates in an AAMC group that meets regularly and is also one of 37 institutions involved in an American Medical Association Consortium (AACOM). These groups discuss challenges, strategies, and opportunities for optimizing medical education during the pandemic.
Some institutions have come up with creative solutions. Ohio University’s Heritage College of Osteopathic Medicine, in Athens, Ohio, in collaboration with the Ohio Department of Health, launched a 4-week rotation for third-year students that focuses on public health. Harvard Medical School, Boston, was one of several schools that allowed students to graduate early in the spring. “We’re constantly talking to our colleagues and friends,” Dr. Brady said. “We learn from each other. There’s a lot of sharing going on.”
Other organizations are also working to make sure students ultimately get what they are paying for: a high-quality education. As soon as the pandemic began, the AACOM organized four working groups to address how schools could better use technology to deliver curricula and how students could participate in public health efforts, among other topics. “For the students, the part they don’t see and can’t really be aware of is all the things that happen behind the scenes,” Mr. Cain said. “People were working really hard to make sure that their education was still delivered, and delivered in a way that was going to assure a good product at the end.”
Ultimately, that product will be held to a rigid standard, said Geoffrey Young, the AAMC’s senior director for student affairs and programs. Medical schools must still meet standards of competency set by the liaison committee on medical education. Mr. Young says that even now those standards remain rigorous enough to ensure that medical students are learning what they need to know. “The core elements for competency may be slightly altered to address the realities that we’re experiencing because of COVID, but the core tenants of competencies will not change,” he said.
Even as conversations continue about what a medical education is worth, the pandemic is drawing new attention to the profession. No signs suggest that the value of tuition or a shift to more virtual offerings are scaring students away. Applications for medical schools were up 17% for the fall of 2021.
Brady expects the surge in interest to continue. “The increased focus and emphasis on public health, the increased focus and emphasis on health equity, the increased focus on the need for a more diverse physician workforce, the interest in basic science research around viruses, the interest in COVID itself – there are a lot of different elements that are setting us up for a potential boom in applications to medical school,” he said.
Beyond increasing interest, the pandemic may also finally force a reckoning on the disconnection between how schools think about costs and how students think about value, Dr. Asch said. “When students say: ‘I’m not getting as much from this,’ they’re saying, ‘you should price this according to its lower value.’ And when the medical schools are saying: ‘Oh, but it’s costing us so much more,’ they’re talking about pricing according to the cost. It’s like one group is speaking Latin and the other group is speaking Greek.” Perhaps, he said, COVID-related changes will finally get them speaking the same language.
This article first appeared on Medscape.com.
Like most medical students, Kaitlyn Thomas’s education was abruptly interrupted by the pandemic. Her school, an osteopathic medicine institution in the Midwest, followed guidelines issued by the American Association of Medical Colleges in March, shifting lectures online and suspending activities in which students interacted with patients. But even as Ms. Thomas’s learning opportunities dwindled for the sake of safety, the costs kept piling up.
Instead of going home to live with her family, she stayed in her apartment near school – and kept paying rent – so she could be nearby for the two licensing exams she was scheduled to take 3 months later. Both tests were canceled 9 days before she was scheduled to take them, one without any notification. This meant she had to travel to two different testing sites in two different states. All told, she said, the whole thing cost her around $2,000.
Ms. Thomas’s experience isn’t rare. Across the country, medical students find themselves paying substantial costs for a medical education now greatly altered by the pandemic. Despite restrictions on time spent in hospitals, hands-on learning, social events, and access to libraries, gyms, study spaces, and instructors, the price of tuition hasn’t dropped but has remained the same or has even risen.
In response, students have become vocal about the return on their pricey investment. “Am I just going to end up doing most of my year online, and what does that look like for my future patients?” Ms. Thomas asked. “It really doesn’t feel like a time to be limiting education.”
Medical schools and administrators are scrambling to find creative solutions for safely educating students. No matter what those solutions may be, experts say, the pandemic has drawn fresh attention to enduring questions about how the cost of medical education compares to its value. Although many are frustrated, some see the potential for COVID to open new opportunities for lasting innovation. At the very least, the pandemic has sparked conversations about what matters most in terms of producing qualified physicians.
“While this is a challenging time, we will get through it, and we will continue to educate doctors, and we will get them through to practice,” says Robert Cain, president and CEO of the American Association of Colleges of Osteopathic Medicine. Many in the midst of training still have one lingering question: Is the price future doctors are now paying still worth it?
COVID’s “hidden costs” for students
Tom is a third-year student at an allopathic medicine institution in the Caribbean. He asked not to be fully identified here, owing to concern about possible backlash. In March, Tom was doing clinical rotations in New York City when his training was put on hold. He returned home to Connecticut and resumed working 60-80 hours a week as a paramedic. As much as 75% of that income went to pay for the New York City apartment he was no longer living in – an apartment that cost more than $2,000 a month – and for student loans that suddenly came due when his enrollment status changed.
Tom has been able to take some online courses through his school. But he still doesn’t know whether state licensing boards will accept them, how residency programs will view them, or whether he will eventually have to retake those online classes in person. At the end of September, he was allowed to return to the hospital but was relocated to Chicago and was forced to move on short notice.
Like many students, Tom has worried that the pandemic may prevent him from acquiring crucial elements for his residency applications, things like letters of recommendation or key experiences. That could delay his next stage of training, which would mean lost future income, increasing student loan interest, and lost work experience. “This could also mean the difference between getting a residency and being able to practice medicine and not being able to practice my intended specialty,” he said. “This is the real hidden cost we may have to deal with.”
International medical students hoping to practice in the United States face additional costs. Michelle Warncke earned her bachelor’s degree in America but went to the United Kingdom for her master’s and her medical degree, which she completed in 2019. She then moved to North Carolina with her husband and saved money to take the exams she needed for residency in the states. But her scheduled Step 2 CS exam was canceled because of the pandemic. Now, like hundreds or even thousands of other students, she said she is unable to apply for residency, even as her student loans collect interest. An active Facebook group of international medical graduates includes about 1,500 people with comparable dilemmas.
The path to becoming a physician carries a well-known price tag, one that is already quite high. Now, for many, that price is substantially increasing. “The only way I can actually keep my medical credentials up to date and passable, to be able to ever get a shot at a residency in the following years,” she said, “is to move to another country and work for less pay, pay for a visa, pay for my exams, pay for my language test, and wait and hope that I might be able to as an older graduate then be able to apply for residency.”
Scaling back the price of med school?
Questions about the economics of medical education aren’t new, says David Asch, MD, MBA, an internal medicine physician and executive director of the Center for Health Care Innovation at the University of Pennsylvania, Philadelphia. But the changes forced by COVID could lead to innovations that may finally better balance the financial scales.
Such innovations are necessary, many say, given how medical education costs have skyrocketed over the past half century. In the 1960s, 4 years of medical school cost about $40,000 in today’s dollars, Dr. Asch and colleagues wrote in a 2020 analysis, which they conducted before the pandemic began. By 2018, the price of a medical education in the United States had ballooned to about $300,000. About 75% of students were taking out loans. Upon graduating, the average debt was $200,000.
Medical school is expensive for many tangible reasons, Dr. Asch said. Schools must pay for curriculum, faculty, technology, textbooks, lab materials, facilities, administrators, and more. But policy changes could decrease those costs.
He says one idea would be for medical schools to join forces and give students access to the same basic lectures in the early years, delivered online by top-notch instructors. Students could then participate in on-campus programs that might only require 3 years to complete instead of 4. By demonstrating what can be done via online platforms, he said, the pandemic might pave the way to permanent changes that could reduce costs.
“I’m not trying to pick on biochemistry professors and medical schools, but how many do we need in the country?” Dr. Asch asked. “We’re all watching the same episode of Seinfeld. Why can’t we all watch the same episode of the Krebs cycle?” If all 190 or so medical schools in the United States shared such preclinical courses, he says, each would require a fraction of the current cost to produce. “We could save 99.5% of the cost. So why don’t we do that?”
Pandemic as opportunity
Although the price of medical education has yet to decrease, schools are working to leverage the pandemic to provide increased educational value.
This generation of physicians will not only have to cope with the fallout of this pandemic, they will be the ones responsible for confronting the next pandemic as well, says Donald Brady, MD, senior associate dean for health sciences education at Vanderbilt University, Nashville, Tenn. “They will be the leaders in the future who will better be able to know how to handle it [a pandemic] because they were able to watch it and be part of it safely in the current circumstance.”
As much as possible, Vanderbilt is using the pandemic as an opportunity. As soon as it became clear that students couldn’t be involved in certain hands-on training, instructors developed a course about pandemics that included lectures on ethics, global health, systemic racism, and other topics. It also included experiential components of pandemic management, such as opportunities to work with patients through telehealth.
Students say they feel that they are getting less for their money and that they are paying for experiences that are no longer available, such as hands-on patient contact and community events. However, Dr. Brady said, schools have had to account for new expenses, including various now-required technologies and transitioning to courses online.
Some challenges can’t be solved with money alone. Medical schools across the country are working together to ensure that they are still adequately preparing students. Vanderbilt participates in an AAMC group that meets regularly and is also one of 37 institutions involved in an American Medical Association Consortium (AACOM). These groups discuss challenges, strategies, and opportunities for optimizing medical education during the pandemic.
Some institutions have come up with creative solutions. Ohio University’s Heritage College of Osteopathic Medicine, in Athens, Ohio, in collaboration with the Ohio Department of Health, launched a 4-week rotation for third-year students that focuses on public health. Harvard Medical School, Boston, was one of several schools that allowed students to graduate early in the spring. “We’re constantly talking to our colleagues and friends,” Dr. Brady said. “We learn from each other. There’s a lot of sharing going on.”
Other organizations are also working to make sure students ultimately get what they are paying for: a high-quality education. As soon as the pandemic began, the AACOM organized four working groups to address how schools could better use technology to deliver curricula and how students could participate in public health efforts, among other topics. “For the students, the part they don’t see and can’t really be aware of is all the things that happen behind the scenes,” Mr. Cain said. “People were working really hard to make sure that their education was still delivered, and delivered in a way that was going to assure a good product at the end.”
Ultimately, that product will be held to a rigid standard, said Geoffrey Young, the AAMC’s senior director for student affairs and programs. Medical schools must still meet standards of competency set by the liaison committee on medical education. Mr. Young says that even now those standards remain rigorous enough to ensure that medical students are learning what they need to know. “The core elements for competency may be slightly altered to address the realities that we’re experiencing because of COVID, but the core tenants of competencies will not change,” he said.
Even as conversations continue about what a medical education is worth, the pandemic is drawing new attention to the profession. No signs suggest that the value of tuition or a shift to more virtual offerings are scaring students away. Applications for medical schools were up 17% for the fall of 2021.
Brady expects the surge in interest to continue. “The increased focus and emphasis on public health, the increased focus and emphasis on health equity, the increased focus on the need for a more diverse physician workforce, the interest in basic science research around viruses, the interest in COVID itself – there are a lot of different elements that are setting us up for a potential boom in applications to medical school,” he said.
Beyond increasing interest, the pandemic may also finally force a reckoning on the disconnection between how schools think about costs and how students think about value, Dr. Asch said. “When students say: ‘I’m not getting as much from this,’ they’re saying, ‘you should price this according to its lower value.’ And when the medical schools are saying: ‘Oh, but it’s costing us so much more,’ they’re talking about pricing according to the cost. It’s like one group is speaking Latin and the other group is speaking Greek.” Perhaps, he said, COVID-related changes will finally get them speaking the same language.
This article first appeared on Medscape.com.
Two COVID-19 outpatient antibody drugs show encouraging results
Two COVID-19 antibody treatments, one developed by Regeneron and the other by Eli Lilly, show promise in the outpatient setting in results released on Oct. 28.
Regeneron, in a randomized, double-blind trial, is assessing the effect of adding its investigational antibody cocktail REGN-COV2 to usual standard of care in comparison with adding placebo to standard of care. A descriptive analysis from the first 275 patients was previously reported. The data described on Oct. 28, which involve an additional 524 patients, show that the trial met all of the first nine endpoints.
Regeneron announced prospective results from its phase 2/3 trial showing REGN-COV2 significantly reduced viral load and patient medical visits, which included hospitalizations, visits to an emergency department, visits for urgent care, and/or physician office/telemedicine visits.
Interest in the cocktail spiked after President Donald Trump extolled its benefits after it was used in his own COVID-19 treatment earlier in October.
Trump received the highest dose of the drug, 8 g, but, according to a Regeneron news release announcing the latest findings, “results showed no significant difference in virologic or clinical efficacy between the REGN-COV2 high dose (8 grams) and low dose (2.4 grams).”
The company described further results of the industry-funded study in the release: “On the primary endpoint, the average daily change in viral load through day 7 (mean time-weighted average change from baseline) in patients with high viral load (defined as greater than107 copies/mL) was a 0.68 log10 copies/mL greater reduction with REGN-COV2 compared to placebo (combined dose groups; P < .0001). There was a 1.08 log greater reduction with REGN-COV2 treatment by day 5, which corresponds to REGN-COV2 patients having, on average, a greater than 10-fold reduction in viral load, compared to placebo.”
The treatment appears to be most effective in patients most at risk, whether because of high viral load, ineffective baseline antibody immune response, or preexisting conditions, according to the researchers.
According to the press release, these results have not been peer reviewed but have been submitted to the US Food and Drug Administration, which is reviewing a potential emergency use authorization for the treatment in high-risk adults with mild to moderate COVID-19.
Operation Warp Speed, the Trump administration’s treatment and vaccine program, contracted in July with Regeneron for up to 300,000 doses of its antibody cocktail.
Lilly treatment shows drop in hospitalizations, symptoms
Another treatment, also given in the outpatient setting, shows promise as well.
Patients recently diagnosed with mild to moderate COVID-19 who received Eli Lilly’s antibody treatment LY-CoV555 had fewer hospitalizations and symptoms compared with a group that received placebo, an interim analysis of a phase 2 trial indicates.
Peter Chen, MD, with the Department of Medicine, Women’s Guild Lung Institute at Cedars-Sinai Medical Center, Los Angeles, California, and colleagues found that the most profound effects were in the high-risk groups.
The interim findings of the BLAZE-1 study, which was funded by Eli Lilly, were published online October 28 in The New England Journal of Medicine.
Researchers randomly assigned 452 patients to receive an intravenous infusion of LY-CoV555 in one of three doses (700 mg, 2800 mg, or 7000 mg) or placebo.
In the interim analysis, the researchers found that for the entire population, more than 99.97% of viral RNA was eliminated.
For patients who received the 2800-mg dose, the difference from placebo in the decrease from baseline was −0.53 (95% CI, −0.98 to −0.08; P = .02), for a log viral load that was lower by a factor of 3.4. Benefit over placebo was not significant with the other doses.
At day 29, according to the investigators, the percentage of patients hospitalized with COVID-19 was 1.6% (5 of 309 patients) in the treatment group compared with 6.3% (9 of 143 patients) in the placebo group.
Data indicate that the safety profile was similar whether patients received the active treatment or placebo.
“If these results are confirmed in additional analyses in this trial, LY-CoV555 could become a useful treatment for emergency use in patients with recently diagnosed Covid-19,” the authors write.
Deborah Fuller, PhD, professor in the Department of Microbiology at the University of Washington School of Medicine in Seattle, told Medscape Medical News the findings are «exciting» but only part of the treatment solution.
“What’s remarkable about these two studies and others I’ve seen,” she said, “is how consistent they are in terms of the window of time they will be effective, and that’s because they are just targeting the virus itself. They do not have an effect on the inflammation unless they stop the replication early enough.”
The treatments are effective when they are given near the time of diagnosis, she pointed out.
“Once the virus has started that inflammatory cascade in your body, then that train has left the station and you have to deal with the inflammation,” Fuller said.
She says future treatments will likely have to include both the antiviral and anti-inflammatory properties, and physicians will have to assess what’s best, given the stage of the the patient’s disease.
The trial of REGN-COV2 is funded by Regeneron. The BLAZE-1 study is funded by Eli Lilly. Many of the authors have financial ties to Eli Lilly. Fuller has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Two COVID-19 antibody treatments, one developed by Regeneron and the other by Eli Lilly, show promise in the outpatient setting in results released on Oct. 28.
Regeneron, in a randomized, double-blind trial, is assessing the effect of adding its investigational antibody cocktail REGN-COV2 to usual standard of care in comparison with adding placebo to standard of care. A descriptive analysis from the first 275 patients was previously reported. The data described on Oct. 28, which involve an additional 524 patients, show that the trial met all of the first nine endpoints.
Regeneron announced prospective results from its phase 2/3 trial showing REGN-COV2 significantly reduced viral load and patient medical visits, which included hospitalizations, visits to an emergency department, visits for urgent care, and/or physician office/telemedicine visits.
Interest in the cocktail spiked after President Donald Trump extolled its benefits after it was used in his own COVID-19 treatment earlier in October.
Trump received the highest dose of the drug, 8 g, but, according to a Regeneron news release announcing the latest findings, “results showed no significant difference in virologic or clinical efficacy between the REGN-COV2 high dose (8 grams) and low dose (2.4 grams).”
The company described further results of the industry-funded study in the release: “On the primary endpoint, the average daily change in viral load through day 7 (mean time-weighted average change from baseline) in patients with high viral load (defined as greater than107 copies/mL) was a 0.68 log10 copies/mL greater reduction with REGN-COV2 compared to placebo (combined dose groups; P < .0001). There was a 1.08 log greater reduction with REGN-COV2 treatment by day 5, which corresponds to REGN-COV2 patients having, on average, a greater than 10-fold reduction in viral load, compared to placebo.”
The treatment appears to be most effective in patients most at risk, whether because of high viral load, ineffective baseline antibody immune response, or preexisting conditions, according to the researchers.
According to the press release, these results have not been peer reviewed but have been submitted to the US Food and Drug Administration, which is reviewing a potential emergency use authorization for the treatment in high-risk adults with mild to moderate COVID-19.
Operation Warp Speed, the Trump administration’s treatment and vaccine program, contracted in July with Regeneron for up to 300,000 doses of its antibody cocktail.
Lilly treatment shows drop in hospitalizations, symptoms
Another treatment, also given in the outpatient setting, shows promise as well.
Patients recently diagnosed with mild to moderate COVID-19 who received Eli Lilly’s antibody treatment LY-CoV555 had fewer hospitalizations and symptoms compared with a group that received placebo, an interim analysis of a phase 2 trial indicates.
Peter Chen, MD, with the Department of Medicine, Women’s Guild Lung Institute at Cedars-Sinai Medical Center, Los Angeles, California, and colleagues found that the most profound effects were in the high-risk groups.
The interim findings of the BLAZE-1 study, which was funded by Eli Lilly, were published online October 28 in The New England Journal of Medicine.
Researchers randomly assigned 452 patients to receive an intravenous infusion of LY-CoV555 in one of three doses (700 mg, 2800 mg, or 7000 mg) or placebo.
In the interim analysis, the researchers found that for the entire population, more than 99.97% of viral RNA was eliminated.
For patients who received the 2800-mg dose, the difference from placebo in the decrease from baseline was −0.53 (95% CI, −0.98 to −0.08; P = .02), for a log viral load that was lower by a factor of 3.4. Benefit over placebo was not significant with the other doses.
At day 29, according to the investigators, the percentage of patients hospitalized with COVID-19 was 1.6% (5 of 309 patients) in the treatment group compared with 6.3% (9 of 143 patients) in the placebo group.
Data indicate that the safety profile was similar whether patients received the active treatment or placebo.
“If these results are confirmed in additional analyses in this trial, LY-CoV555 could become a useful treatment for emergency use in patients with recently diagnosed Covid-19,” the authors write.
Deborah Fuller, PhD, professor in the Department of Microbiology at the University of Washington School of Medicine in Seattle, told Medscape Medical News the findings are «exciting» but only part of the treatment solution.
“What’s remarkable about these two studies and others I’ve seen,” she said, “is how consistent they are in terms of the window of time they will be effective, and that’s because they are just targeting the virus itself. They do not have an effect on the inflammation unless they stop the replication early enough.”
The treatments are effective when they are given near the time of diagnosis, she pointed out.
“Once the virus has started that inflammatory cascade in your body, then that train has left the station and you have to deal with the inflammation,” Fuller said.
She says future treatments will likely have to include both the antiviral and anti-inflammatory properties, and physicians will have to assess what’s best, given the stage of the the patient’s disease.
The trial of REGN-COV2 is funded by Regeneron. The BLAZE-1 study is funded by Eli Lilly. Many of the authors have financial ties to Eli Lilly. Fuller has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Two COVID-19 antibody treatments, one developed by Regeneron and the other by Eli Lilly, show promise in the outpatient setting in results released on Oct. 28.
Regeneron, in a randomized, double-blind trial, is assessing the effect of adding its investigational antibody cocktail REGN-COV2 to usual standard of care in comparison with adding placebo to standard of care. A descriptive analysis from the first 275 patients was previously reported. The data described on Oct. 28, which involve an additional 524 patients, show that the trial met all of the first nine endpoints.
Regeneron announced prospective results from its phase 2/3 trial showing REGN-COV2 significantly reduced viral load and patient medical visits, which included hospitalizations, visits to an emergency department, visits for urgent care, and/or physician office/telemedicine visits.
Interest in the cocktail spiked after President Donald Trump extolled its benefits after it was used in his own COVID-19 treatment earlier in October.
Trump received the highest dose of the drug, 8 g, but, according to a Regeneron news release announcing the latest findings, “results showed no significant difference in virologic or clinical efficacy between the REGN-COV2 high dose (8 grams) and low dose (2.4 grams).”
The company described further results of the industry-funded study in the release: “On the primary endpoint, the average daily change in viral load through day 7 (mean time-weighted average change from baseline) in patients with high viral load (defined as greater than107 copies/mL) was a 0.68 log10 copies/mL greater reduction with REGN-COV2 compared to placebo (combined dose groups; P < .0001). There was a 1.08 log greater reduction with REGN-COV2 treatment by day 5, which corresponds to REGN-COV2 patients having, on average, a greater than 10-fold reduction in viral load, compared to placebo.”
The treatment appears to be most effective in patients most at risk, whether because of high viral load, ineffective baseline antibody immune response, or preexisting conditions, according to the researchers.
According to the press release, these results have not been peer reviewed but have been submitted to the US Food and Drug Administration, which is reviewing a potential emergency use authorization for the treatment in high-risk adults with mild to moderate COVID-19.
Operation Warp Speed, the Trump administration’s treatment and vaccine program, contracted in July with Regeneron for up to 300,000 doses of its antibody cocktail.
Lilly treatment shows drop in hospitalizations, symptoms
Another treatment, also given in the outpatient setting, shows promise as well.
Patients recently diagnosed with mild to moderate COVID-19 who received Eli Lilly’s antibody treatment LY-CoV555 had fewer hospitalizations and symptoms compared with a group that received placebo, an interim analysis of a phase 2 trial indicates.
Peter Chen, MD, with the Department of Medicine, Women’s Guild Lung Institute at Cedars-Sinai Medical Center, Los Angeles, California, and colleagues found that the most profound effects were in the high-risk groups.
The interim findings of the BLAZE-1 study, which was funded by Eli Lilly, were published online October 28 in The New England Journal of Medicine.
Researchers randomly assigned 452 patients to receive an intravenous infusion of LY-CoV555 in one of three doses (700 mg, 2800 mg, or 7000 mg) or placebo.
In the interim analysis, the researchers found that for the entire population, more than 99.97% of viral RNA was eliminated.
For patients who received the 2800-mg dose, the difference from placebo in the decrease from baseline was −0.53 (95% CI, −0.98 to −0.08; P = .02), for a log viral load that was lower by a factor of 3.4. Benefit over placebo was not significant with the other doses.
At day 29, according to the investigators, the percentage of patients hospitalized with COVID-19 was 1.6% (5 of 309 patients) in the treatment group compared with 6.3% (9 of 143 patients) in the placebo group.
Data indicate that the safety profile was similar whether patients received the active treatment or placebo.
“If these results are confirmed in additional analyses in this trial, LY-CoV555 could become a useful treatment for emergency use in patients with recently diagnosed Covid-19,” the authors write.
Deborah Fuller, PhD, professor in the Department of Microbiology at the University of Washington School of Medicine in Seattle, told Medscape Medical News the findings are «exciting» but only part of the treatment solution.
“What’s remarkable about these two studies and others I’ve seen,” she said, “is how consistent they are in terms of the window of time they will be effective, and that’s because they are just targeting the virus itself. They do not have an effect on the inflammation unless they stop the replication early enough.”
The treatments are effective when they are given near the time of diagnosis, she pointed out.
“Once the virus has started that inflammatory cascade in your body, then that train has left the station and you have to deal with the inflammation,” Fuller said.
She says future treatments will likely have to include both the antiviral and anti-inflammatory properties, and physicians will have to assess what’s best, given the stage of the the patient’s disease.
The trial of REGN-COV2 is funded by Regeneron. The BLAZE-1 study is funded by Eli Lilly. Many of the authors have financial ties to Eli Lilly. Fuller has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.