User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'main-prefix')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
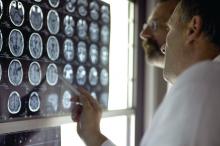
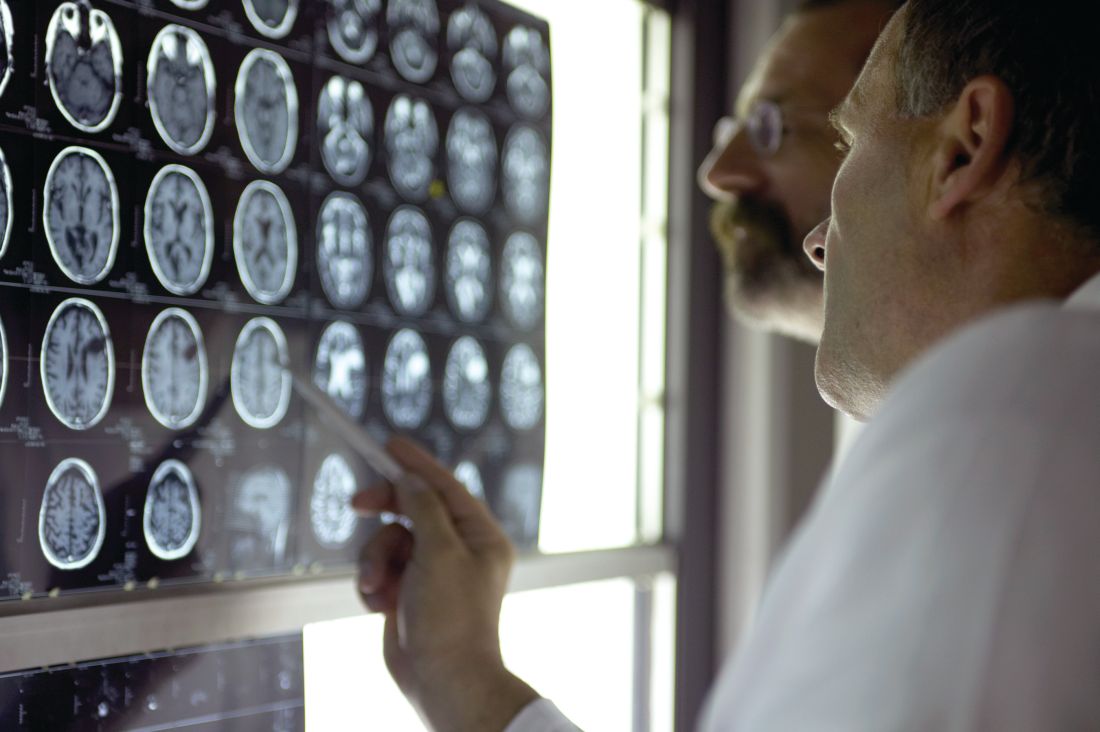
Tocilizumab stumbles as COVID-19 treatment, narrow role possible
Tocilizumab (Actemra/RoActemra) was not found to have any clear role as a treatment for COVID-19 in four new studies.
Three randomized controlled trials showed that the drug either had no benefit or only a modest one, contradicting a large retrospective study that had hinted at a more robust effect.
“This is not a blockbuster,” said David Cennimo, MD, an infectious disease expert at Rutgers New Jersey Medical School, Newark, New Jersey. “This is not something that’s going to revolutionize our treatment of COVID-19.”
But some researchers still regard these studies as showing evidence that the drug benefits certain patients with severe inflammation.
The immune response to SARS-CoV-2 includes elevated levels of the cytokine interleukin-6 (IL-6). In some patients, this response becomes a nonspecific inflammation, a “cytokine storm,” involving edema and inflammatory cell infiltration in the lungs. These cases are among the most severe.
Dexamethasone has proved effective in controlling this inflammation in some patients. Researchers have theorized that a more targeted suppression of IL-6 could be even more effective or work in cases that don’t respond to dexamethasone.
A recombinant monoclonal antibody, tocilizumab blocks IL-6 receptors. It is approved by the US Food and Drug Administration for use in patients with rheumatologic disorders and cytokine release syndrome induced by chimeric antigen receptor T-cell therapy.
Current National Institutes of Health (NIH) guidelines recommend against the use of tocilizumab as a treatment for COVID-19, despite earlier observational studies that suggested the drug might help patients with moderate to severe disease. Controlled trials were lacking until now.
The most hopeful results in this batch came from the CORIMUNO-19 platform of open-label, randomized controlled trials of immune modulatory treatments for moderate or severe COVID-19 in France.
Published in JAMA Internal Medicine , the trial recruited patients from nine French hospitals. Patients were eligible if they required at least 3 L/min of oxygen without ventilation or admission to the intensive care unit.
The investigators randomly assigned 64 patients to receive tocilizumab 8 mg/kg body weight intravenously plus usual care and 67 patients to usual care alone. Usual care included antibiotic agents, antiviral agents, corticosteroids, vasopressor support, and anticoagulants.
After 4 days, the investigators scored patients on the World Health Organization 10-point Clinical Progression Scale. Twelve of the patients who received tocilizumab scored higher than 5 vs 19 of the patients in the usual care group, with higher scores indicating clinical deterioration.
After 14 days, 24% of the patients taking tocilizumab required either noninvasive ventilation or mechanical ventilation or had died, vs 36% in the usual care group (median posterior hazard ratio [HR], 0.58; 90% credible interval, 0.33 – 1.00).
“We reduced the risk of dying or requiring mechanical ventilation, so for me, the study was positive,” said Olivier Hermine, MD, PhD, a professor of hematology at Paris Descartes University in Paris, France.
However, there was no difference in mortality at 28 days. Hermine hopes to have longer-term outcomes soon, he told Medscape Medical News.
A second randomized controlled trial, also published in JAMA Internal Medicine , provided less hope. In this RCT-TCZ-COVID-19 Study Group trial, conducted at 24 Italian centers, patients were enrolled if their partial pressure of arterial oxygen to fraction of inspired oxygen (PaO2/FiO2) ratios were between 200 and 300 mm Hg and if their inflammatory phenotypes were defined by fever and elevated C-reactive protein level.
The investigators randomly assigned 60 patients to receive tocilizumab 8 mg/kg up to a maximum of 800 mg within 8 hours of randomization, followed by a second dose after 12 hours. They assigned 66 patients to a control group that received supportive care until clinical worsening, at which point patients could receive tocilizumab as a rescue therapy.
Of the patients who received tocilizumab, 28.3% showed clinical worsening within 14 days, compared to 27.0% in the control group (rate ratio, 1.05; 95% CI, 0.59 – 1.86). There was no significant difference between the groups in terms of the proportion admitted to intensive care. The researchers stopped the trial prematurely because tocilizumab did not seem to be making a difference.
The BACC Bay Tocilizumab Trial was conducted at seven Boston hospitals. The results, which were published in The New England Journal of Medicine, were also discouraging.
In that trial, enrolled patients met two sets of parameters. First, the patients had at least one of the following signs: C-reactive protein level higher than 50 mg/L, ferritin level higher than 500 ng/mL, D-dimer level higher than 1000 ng/mL, or a lactate dehydrogenase level higher than 250 U/L. Second, the patients had to have at least two of the following signs: body temperature >38° C, pulmonary infiltrates, or the need for supplemental oxygen to maintain an oxygen saturation greater than 92%.
The investigators randomly assigned 161 patients to receive intravenous tocilizumab 8 mg/kg up to 800 mg and 81 to receive a placebo.
They didn’t find a statistically significant difference between the groups. The hazard ratio for intubation or death in the tocilizumab group as compared with the placebo group was 0.83 (95% CI, 0.38 – 1.81; P = .64). The hazard ratio for disease worsening was 1.11 (95% CI, 0.59 – 2.10; P = .73). At 14 days, the conditions of 18.0% of the patients who received tocilizumab and 14.9% of the patients who received the placebo worsened.
In contrast to these randomized trials, STOP-COVID, a retrospective analysis of 3924 patients, also published in JAMA Internal Medicine, found that the risk for death was lower for patients treated with tocilizumab compared with those not treated with tocilizumab (HR, 0.71; 95% CI, 0.56 – 0.92) over a median follow-up period of 27 days.
Also on the bright side, none of the new studies showed significant adverse reactions to tocilizumab.
More randomized clinical trials are underway. In press releases announcing topline data, Roche reported mostly negative results in its phase 3 COVACTA trial but noted a 44% reduction in the risk for progression to death or ventilation in its phase 3 IMPACTA trial. Roche did not comment on the ethnicity of its COVACTA patients; it said IMPACTA enrolled a majority of Hispanic patients and included large representations of Native American and Black patients.
Results don’t support routine use
Commenting on the new studies, editorialists in both JAMA Internal Medicine and The New England Journal of Medicine concluded that the tocilizumab results were not strong enough to support routine use.
“My take-home point from looking at all of these together is that, even if it does help, it’s most likely in a small subset of the population and/or a small effect,” Cennimo told Medscape Medical News.
But the NIH recommendation against tocilizumab goes too far, argued Cristina Mussini, MD, a professor of infectious diseases at the University of Modena and Reggio Emilia in Italy, who is a coauthor of a cohort study of tocilizumab and served on the CORIMUNO-19 Data Safety and Monitoring Board.
“I really think it’s too early to recommend against it because at least two clinical trials showed protection against mechanical ventilation and death,” she said.
She prescribes tocilizumab for patients who have not been helped by dexamethasone. “It’s just a rescue drug,” she told Medscape Medical News. “It’s not something you use for everybody, but it’s the only weapon we have now when the patient is really going to the intensive care unit.”
The BACC Bay Tocilizumab Trial was funded by Genentech/Roche. Genentech/Roche provided the drug for the CORIMUNO and RCT-TCZ-COVID-19 trials. The STOP-COVID study was supported by grants from the NIH and by the Frankel Cardiovascular Center COVID-19: Impact Research Ignitor. Cennimo, Hermine, and Mussini have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Tocilizumab (Actemra/RoActemra) was not found to have any clear role as a treatment for COVID-19 in four new studies.
Three randomized controlled trials showed that the drug either had no benefit or only a modest one, contradicting a large retrospective study that had hinted at a more robust effect.
“This is not a blockbuster,” said David Cennimo, MD, an infectious disease expert at Rutgers New Jersey Medical School, Newark, New Jersey. “This is not something that’s going to revolutionize our treatment of COVID-19.”
But some researchers still regard these studies as showing evidence that the drug benefits certain patients with severe inflammation.
The immune response to SARS-CoV-2 includes elevated levels of the cytokine interleukin-6 (IL-6). In some patients, this response becomes a nonspecific inflammation, a “cytokine storm,” involving edema and inflammatory cell infiltration in the lungs. These cases are among the most severe.
Dexamethasone has proved effective in controlling this inflammation in some patients. Researchers have theorized that a more targeted suppression of IL-6 could be even more effective or work in cases that don’t respond to dexamethasone.
A recombinant monoclonal antibody, tocilizumab blocks IL-6 receptors. It is approved by the US Food and Drug Administration for use in patients with rheumatologic disorders and cytokine release syndrome induced by chimeric antigen receptor T-cell therapy.
Current National Institutes of Health (NIH) guidelines recommend against the use of tocilizumab as a treatment for COVID-19, despite earlier observational studies that suggested the drug might help patients with moderate to severe disease. Controlled trials were lacking until now.
The most hopeful results in this batch came from the CORIMUNO-19 platform of open-label, randomized controlled trials of immune modulatory treatments for moderate or severe COVID-19 in France.
Published in JAMA Internal Medicine , the trial recruited patients from nine French hospitals. Patients were eligible if they required at least 3 L/min of oxygen without ventilation or admission to the intensive care unit.
The investigators randomly assigned 64 patients to receive tocilizumab 8 mg/kg body weight intravenously plus usual care and 67 patients to usual care alone. Usual care included antibiotic agents, antiviral agents, corticosteroids, vasopressor support, and anticoagulants.
After 4 days, the investigators scored patients on the World Health Organization 10-point Clinical Progression Scale. Twelve of the patients who received tocilizumab scored higher than 5 vs 19 of the patients in the usual care group, with higher scores indicating clinical deterioration.
After 14 days, 24% of the patients taking tocilizumab required either noninvasive ventilation or mechanical ventilation or had died, vs 36% in the usual care group (median posterior hazard ratio [HR], 0.58; 90% credible interval, 0.33 – 1.00).
“We reduced the risk of dying or requiring mechanical ventilation, so for me, the study was positive,” said Olivier Hermine, MD, PhD, a professor of hematology at Paris Descartes University in Paris, France.
However, there was no difference in mortality at 28 days. Hermine hopes to have longer-term outcomes soon, he told Medscape Medical News.
A second randomized controlled trial, also published in JAMA Internal Medicine , provided less hope. In this RCT-TCZ-COVID-19 Study Group trial, conducted at 24 Italian centers, patients were enrolled if their partial pressure of arterial oxygen to fraction of inspired oxygen (PaO2/FiO2) ratios were between 200 and 300 mm Hg and if their inflammatory phenotypes were defined by fever and elevated C-reactive protein level.
The investigators randomly assigned 60 patients to receive tocilizumab 8 mg/kg up to a maximum of 800 mg within 8 hours of randomization, followed by a second dose after 12 hours. They assigned 66 patients to a control group that received supportive care until clinical worsening, at which point patients could receive tocilizumab as a rescue therapy.
Of the patients who received tocilizumab, 28.3% showed clinical worsening within 14 days, compared to 27.0% in the control group (rate ratio, 1.05; 95% CI, 0.59 – 1.86). There was no significant difference between the groups in terms of the proportion admitted to intensive care. The researchers stopped the trial prematurely because tocilizumab did not seem to be making a difference.
The BACC Bay Tocilizumab Trial was conducted at seven Boston hospitals. The results, which were published in The New England Journal of Medicine, were also discouraging.
In that trial, enrolled patients met two sets of parameters. First, the patients had at least one of the following signs: C-reactive protein level higher than 50 mg/L, ferritin level higher than 500 ng/mL, D-dimer level higher than 1000 ng/mL, or a lactate dehydrogenase level higher than 250 U/L. Second, the patients had to have at least two of the following signs: body temperature >38° C, pulmonary infiltrates, or the need for supplemental oxygen to maintain an oxygen saturation greater than 92%.
The investigators randomly assigned 161 patients to receive intravenous tocilizumab 8 mg/kg up to 800 mg and 81 to receive a placebo.
They didn’t find a statistically significant difference between the groups. The hazard ratio for intubation or death in the tocilizumab group as compared with the placebo group was 0.83 (95% CI, 0.38 – 1.81; P = .64). The hazard ratio for disease worsening was 1.11 (95% CI, 0.59 – 2.10; P = .73). At 14 days, the conditions of 18.0% of the patients who received tocilizumab and 14.9% of the patients who received the placebo worsened.
In contrast to these randomized trials, STOP-COVID, a retrospective analysis of 3924 patients, also published in JAMA Internal Medicine, found that the risk for death was lower for patients treated with tocilizumab compared with those not treated with tocilizumab (HR, 0.71; 95% CI, 0.56 – 0.92) over a median follow-up period of 27 days.
Also on the bright side, none of the new studies showed significant adverse reactions to tocilizumab.
More randomized clinical trials are underway. In press releases announcing topline data, Roche reported mostly negative results in its phase 3 COVACTA trial but noted a 44% reduction in the risk for progression to death or ventilation in its phase 3 IMPACTA trial. Roche did not comment on the ethnicity of its COVACTA patients; it said IMPACTA enrolled a majority of Hispanic patients and included large representations of Native American and Black patients.
Results don’t support routine use
Commenting on the new studies, editorialists in both JAMA Internal Medicine and The New England Journal of Medicine concluded that the tocilizumab results were not strong enough to support routine use.
“My take-home point from looking at all of these together is that, even if it does help, it’s most likely in a small subset of the population and/or a small effect,” Cennimo told Medscape Medical News.
But the NIH recommendation against tocilizumab goes too far, argued Cristina Mussini, MD, a professor of infectious diseases at the University of Modena and Reggio Emilia in Italy, who is a coauthor of a cohort study of tocilizumab and served on the CORIMUNO-19 Data Safety and Monitoring Board.
“I really think it’s too early to recommend against it because at least two clinical trials showed protection against mechanical ventilation and death,” she said.
She prescribes tocilizumab for patients who have not been helped by dexamethasone. “It’s just a rescue drug,” she told Medscape Medical News. “It’s not something you use for everybody, but it’s the only weapon we have now when the patient is really going to the intensive care unit.”
The BACC Bay Tocilizumab Trial was funded by Genentech/Roche. Genentech/Roche provided the drug for the CORIMUNO and RCT-TCZ-COVID-19 trials. The STOP-COVID study was supported by grants from the NIH and by the Frankel Cardiovascular Center COVID-19: Impact Research Ignitor. Cennimo, Hermine, and Mussini have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Tocilizumab (Actemra/RoActemra) was not found to have any clear role as a treatment for COVID-19 in four new studies.
Three randomized controlled trials showed that the drug either had no benefit or only a modest one, contradicting a large retrospective study that had hinted at a more robust effect.
“This is not a blockbuster,” said David Cennimo, MD, an infectious disease expert at Rutgers New Jersey Medical School, Newark, New Jersey. “This is not something that’s going to revolutionize our treatment of COVID-19.”
But some researchers still regard these studies as showing evidence that the drug benefits certain patients with severe inflammation.
The immune response to SARS-CoV-2 includes elevated levels of the cytokine interleukin-6 (IL-6). In some patients, this response becomes a nonspecific inflammation, a “cytokine storm,” involving edema and inflammatory cell infiltration in the lungs. These cases are among the most severe.
Dexamethasone has proved effective in controlling this inflammation in some patients. Researchers have theorized that a more targeted suppression of IL-6 could be even more effective or work in cases that don’t respond to dexamethasone.
A recombinant monoclonal antibody, tocilizumab blocks IL-6 receptors. It is approved by the US Food and Drug Administration for use in patients with rheumatologic disorders and cytokine release syndrome induced by chimeric antigen receptor T-cell therapy.
Current National Institutes of Health (NIH) guidelines recommend against the use of tocilizumab as a treatment for COVID-19, despite earlier observational studies that suggested the drug might help patients with moderate to severe disease. Controlled trials were lacking until now.
The most hopeful results in this batch came from the CORIMUNO-19 platform of open-label, randomized controlled trials of immune modulatory treatments for moderate or severe COVID-19 in France.
Published in JAMA Internal Medicine , the trial recruited patients from nine French hospitals. Patients were eligible if they required at least 3 L/min of oxygen without ventilation or admission to the intensive care unit.
The investigators randomly assigned 64 patients to receive tocilizumab 8 mg/kg body weight intravenously plus usual care and 67 patients to usual care alone. Usual care included antibiotic agents, antiviral agents, corticosteroids, vasopressor support, and anticoagulants.
After 4 days, the investigators scored patients on the World Health Organization 10-point Clinical Progression Scale. Twelve of the patients who received tocilizumab scored higher than 5 vs 19 of the patients in the usual care group, with higher scores indicating clinical deterioration.
After 14 days, 24% of the patients taking tocilizumab required either noninvasive ventilation or mechanical ventilation or had died, vs 36% in the usual care group (median posterior hazard ratio [HR], 0.58; 90% credible interval, 0.33 – 1.00).
“We reduced the risk of dying or requiring mechanical ventilation, so for me, the study was positive,” said Olivier Hermine, MD, PhD, a professor of hematology at Paris Descartes University in Paris, France.
However, there was no difference in mortality at 28 days. Hermine hopes to have longer-term outcomes soon, he told Medscape Medical News.
A second randomized controlled trial, also published in JAMA Internal Medicine , provided less hope. In this RCT-TCZ-COVID-19 Study Group trial, conducted at 24 Italian centers, patients were enrolled if their partial pressure of arterial oxygen to fraction of inspired oxygen (PaO2/FiO2) ratios were between 200 and 300 mm Hg and if their inflammatory phenotypes were defined by fever and elevated C-reactive protein level.
The investigators randomly assigned 60 patients to receive tocilizumab 8 mg/kg up to a maximum of 800 mg within 8 hours of randomization, followed by a second dose after 12 hours. They assigned 66 patients to a control group that received supportive care until clinical worsening, at which point patients could receive tocilizumab as a rescue therapy.
Of the patients who received tocilizumab, 28.3% showed clinical worsening within 14 days, compared to 27.0% in the control group (rate ratio, 1.05; 95% CI, 0.59 – 1.86). There was no significant difference between the groups in terms of the proportion admitted to intensive care. The researchers stopped the trial prematurely because tocilizumab did not seem to be making a difference.
The BACC Bay Tocilizumab Trial was conducted at seven Boston hospitals. The results, which were published in The New England Journal of Medicine, were also discouraging.
In that trial, enrolled patients met two sets of parameters. First, the patients had at least one of the following signs: C-reactive protein level higher than 50 mg/L, ferritin level higher than 500 ng/mL, D-dimer level higher than 1000 ng/mL, or a lactate dehydrogenase level higher than 250 U/L. Second, the patients had to have at least two of the following signs: body temperature >38° C, pulmonary infiltrates, or the need for supplemental oxygen to maintain an oxygen saturation greater than 92%.
The investigators randomly assigned 161 patients to receive intravenous tocilizumab 8 mg/kg up to 800 mg and 81 to receive a placebo.
They didn’t find a statistically significant difference between the groups. The hazard ratio for intubation or death in the tocilizumab group as compared with the placebo group was 0.83 (95% CI, 0.38 – 1.81; P = .64). The hazard ratio for disease worsening was 1.11 (95% CI, 0.59 – 2.10; P = .73). At 14 days, the conditions of 18.0% of the patients who received tocilizumab and 14.9% of the patients who received the placebo worsened.
In contrast to these randomized trials, STOP-COVID, a retrospective analysis of 3924 patients, also published in JAMA Internal Medicine, found that the risk for death was lower for patients treated with tocilizumab compared with those not treated with tocilizumab (HR, 0.71; 95% CI, 0.56 – 0.92) over a median follow-up period of 27 days.
Also on the bright side, none of the new studies showed significant adverse reactions to tocilizumab.
More randomized clinical trials are underway. In press releases announcing topline data, Roche reported mostly negative results in its phase 3 COVACTA trial but noted a 44% reduction in the risk for progression to death or ventilation in its phase 3 IMPACTA trial. Roche did not comment on the ethnicity of its COVACTA patients; it said IMPACTA enrolled a majority of Hispanic patients and included large representations of Native American and Black patients.
Results don’t support routine use
Commenting on the new studies, editorialists in both JAMA Internal Medicine and The New England Journal of Medicine concluded that the tocilizumab results were not strong enough to support routine use.
“My take-home point from looking at all of these together is that, even if it does help, it’s most likely in a small subset of the population and/or a small effect,” Cennimo told Medscape Medical News.
But the NIH recommendation against tocilizumab goes too far, argued Cristina Mussini, MD, a professor of infectious diseases at the University of Modena and Reggio Emilia in Italy, who is a coauthor of a cohort study of tocilizumab and served on the CORIMUNO-19 Data Safety and Monitoring Board.
“I really think it’s too early to recommend against it because at least two clinical trials showed protection against mechanical ventilation and death,” she said.
She prescribes tocilizumab for patients who have not been helped by dexamethasone. “It’s just a rescue drug,” she told Medscape Medical News. “It’s not something you use for everybody, but it’s the only weapon we have now when the patient is really going to the intensive care unit.”
The BACC Bay Tocilizumab Trial was funded by Genentech/Roche. Genentech/Roche provided the drug for the CORIMUNO and RCT-TCZ-COVID-19 trials. The STOP-COVID study was supported by grants from the NIH and by the Frankel Cardiovascular Center COVID-19: Impact Research Ignitor. Cennimo, Hermine, and Mussini have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Lilly stops antibody trial in hospitalized COVID-19 patients, other trials continue
Eli Lilly announced it will halt its ACTIV-3 trial evaluating the antibody bamlanivimab in combination with remdesivir for people hospitalized with COVID-19, after new evidence regarding efficacy emerged.
The new data from the National Institutes of Health suggest that the experimental neutralizing antibody therapy does not offer significant clinical benefit for people with more advanced COVID-19 illness, according to a company statement.
Eli Lilly also announced it plans to continue its other trials evaluating the antibody, including those assessing a potential role in treating people in the earlier stages of COVID-19.
“While there was insufficient evidence that bamlanivimab improved clinical outcomes when added to other treatments in hospitalized patients with COVID-19, we remain confident based on data from Lilly’s BLAZE-1 study that bamlanivimab monotherapy may prevent progression of disease for those earlier in the course of COVID-19,” the statement reads.
The ACTIV-3 trial was paused on October 13 after a data and safety monitoring board cited safety concerns.
The most recent data update that triggered an end to the trial did not reveal any significant differences in safety, though.
This article first appeared on Medscape.com.
Eli Lilly announced it will halt its ACTIV-3 trial evaluating the antibody bamlanivimab in combination with remdesivir for people hospitalized with COVID-19, after new evidence regarding efficacy emerged.
The new data from the National Institutes of Health suggest that the experimental neutralizing antibody therapy does not offer significant clinical benefit for people with more advanced COVID-19 illness, according to a company statement.
Eli Lilly also announced it plans to continue its other trials evaluating the antibody, including those assessing a potential role in treating people in the earlier stages of COVID-19.
“While there was insufficient evidence that bamlanivimab improved clinical outcomes when added to other treatments in hospitalized patients with COVID-19, we remain confident based on data from Lilly’s BLAZE-1 study that bamlanivimab monotherapy may prevent progression of disease for those earlier in the course of COVID-19,” the statement reads.
The ACTIV-3 trial was paused on October 13 after a data and safety monitoring board cited safety concerns.
The most recent data update that triggered an end to the trial did not reveal any significant differences in safety, though.
This article first appeared on Medscape.com.
Eli Lilly announced it will halt its ACTIV-3 trial evaluating the antibody bamlanivimab in combination with remdesivir for people hospitalized with COVID-19, after new evidence regarding efficacy emerged.
The new data from the National Institutes of Health suggest that the experimental neutralizing antibody therapy does not offer significant clinical benefit for people with more advanced COVID-19 illness, according to a company statement.
Eli Lilly also announced it plans to continue its other trials evaluating the antibody, including those assessing a potential role in treating people in the earlier stages of COVID-19.
“While there was insufficient evidence that bamlanivimab improved clinical outcomes when added to other treatments in hospitalized patients with COVID-19, we remain confident based on data from Lilly’s BLAZE-1 study that bamlanivimab monotherapy may prevent progression of disease for those earlier in the course of COVID-19,” the statement reads.
The ACTIV-3 trial was paused on October 13 after a data and safety monitoring board cited safety concerns.
The most recent data update that triggered an end to the trial did not reveal any significant differences in safety, though.
This article first appeared on Medscape.com.
Security breach in Finland leads to psychiatric patient blackmail
Hackers have accessed patient records at Finland’s largest private psychotherapy system, emailing some patients to pay up or face having their private medical records released online.
Vastaamo treats about 40,000 patients and runs 25 centers across the country. Hackers emailed some of the centers’ patients asking for a blackmail payment of 200 euro in bitcoin, The Guardian reports.
Agencies such as the country’s National Bureau of Investigation are urging victims not to comply with the blackmailers’ demand and instead requesting that patients report these incidents to authorities and turn over incriminating emails. However, some data from patient records have already been released online.
“We deeply regret what happened and on behalf of our [patients] who have been compromised, we apologize for the shortcoming in data security, the consequences and human cost of which have been extremely heavy,” the center said in a statement. They added that the investigation into the situation is ongoing.
‘Sobering reminder’
In a comment, John Torous, MD, director of digital psychiatry at Beth Israel Deaconess Medical Center, Boston, Massachusetts, said this is “a sobering reminder that any digital data is subject to hacking.”
Torous is also chair of the American Psychiatric Association’s Health and Technology Committee.
“This is not the first time psychotherapy notes have been targeted and it actually happened, on a smaller scale, in the US in 2017,” he said.
In April of that year, confidential patient record information from a mental health center in Maine, including evaluations, session notes, and names of sex-abuse victims, was listed on the dark web.
Also in April, computer hackers released the WannaCry virus into the operating system of the United Kingdom’s National Health Service, which subsequently locked clinicians out of patient records and other digital tools for 3 days.
In addition, in 2016 hackers took Hollywood Presbyterian Medical Center in Los Angeles offline for more than a week after demanding a ransom of $3.6 million.
Criminal investigation
For Vastaamo, three of its employees were approached by the blackmailer via email at the end of September, the company reports. These incidents were immediately disclosed and the Central Criminal Police launched a criminal investigation.
In addition, several agencies were contacted, including the Finnish Cyber Security Center, the Data Protection Commission, and a cyber security company.
Investigators believe the breach, which led to the customer database theft, occurred back in November 2018. In addition, security “deficiencies” remained until March 2019.
“We do not know that the database was stolen after November 2018, but it is possible that individual data [have been] viewed or copied,” Vastaamo said in a press release. No additional “vulnerabilities were identified after March 2019.”
The center’s CEO, Ville Tapio, who did not disclose any of these incidents to the parent company and its board of directors, was subsequently fired.
Once the police investigation began, Vastaamo said it was not granted permission by the authorities to communicate the occurrence to its patients. However, after the blackmailer released some patient information online early on Oct. 21, permission to inform patients was granted.
The company noted that the blackmailer has started emailing victims, informing of the data breach, and demanding ransom. So far, the emails have not contained harmful digital content or “malware,” but authorities warn that any attachments should not be opened. The police have requested that such emails be kept so they can be used as evidence.
In a Q&A section on its website, Vastaamo noted that videos are never recorded during its centers’ telehealth sessions and patients should not be concerned about the possibility of leaked videos.
In addition, the cybercrime has not interrupted Vastaamo’s operations.
“The authorities and the response office will do their utmost to find out what happened, to prevent the dissemination of information, and to bring the perpetrators to justice,” the center said.
“The most important task ... is to support customers in the midst of an exceptionally serious and difficult situation,” it added.
“Worst-case scenario”
In a comment, Ipsit Vahia, MD, medical director at the Institute for Technology and Psychiatry at McLean Hospital, Belmont, Mass., said Vastaamo’s data breach “represents the worst-case scenario for digital health.”
He added that more information is needed about the specifics of the case, including exactly what happened, how the system was hacked, and what information was compromised.
Still, “it raises fundamental questions that healthcare systems, clinicians, and patients everywhere should be asking about what measures are in place to protect electronic medical records and other personal digital information,” said Dr. Vahia.
“This incident also serves as another reminder that the issue of data security and privacy is foundational to digital mental health. Ultimately, without a commitment from all stakeholders to maintaining the strictest levels of security, as well as transparency around how data are handled there will be little to no trust from clinicians or patients,” he said. All of that could prevent digital healthcare from achieving its full potential, he added.
In addition, Dr. Vahia noted that the rapid uptick of telemedicine because of the pandemic has accelerated the use of other forms of digital information in mental healthcare.
“This unfortunate incident should serve as a wake-up call and bring the issue of data protection back firmly into the spotlight,” said Dr. Vahia.
Now that telehealth has become a larger part of clinical practice, said Torous, it’s important for clinicians to be vigilant regarding security procedures.
“Telehealth and digital data are here to stay, and with them new benefits as well as risks. We can continue to work to minimize the risks and protect privacy while ensuring the benefits to patients expand,” he added.
This article first appeared on Medscape.com.
Hackers have accessed patient records at Finland’s largest private psychotherapy system, emailing some patients to pay up or face having their private medical records released online.
Vastaamo treats about 40,000 patients and runs 25 centers across the country. Hackers emailed some of the centers’ patients asking for a blackmail payment of 200 euro in bitcoin, The Guardian reports.
Agencies such as the country’s National Bureau of Investigation are urging victims not to comply with the blackmailers’ demand and instead requesting that patients report these incidents to authorities and turn over incriminating emails. However, some data from patient records have already been released online.
“We deeply regret what happened and on behalf of our [patients] who have been compromised, we apologize for the shortcoming in data security, the consequences and human cost of which have been extremely heavy,” the center said in a statement. They added that the investigation into the situation is ongoing.
‘Sobering reminder’
In a comment, John Torous, MD, director of digital psychiatry at Beth Israel Deaconess Medical Center, Boston, Massachusetts, said this is “a sobering reminder that any digital data is subject to hacking.”
Torous is also chair of the American Psychiatric Association’s Health and Technology Committee.
“This is not the first time psychotherapy notes have been targeted and it actually happened, on a smaller scale, in the US in 2017,” he said.
In April of that year, confidential patient record information from a mental health center in Maine, including evaluations, session notes, and names of sex-abuse victims, was listed on the dark web.
Also in April, computer hackers released the WannaCry virus into the operating system of the United Kingdom’s National Health Service, which subsequently locked clinicians out of patient records and other digital tools for 3 days.
In addition, in 2016 hackers took Hollywood Presbyterian Medical Center in Los Angeles offline for more than a week after demanding a ransom of $3.6 million.
Criminal investigation
For Vastaamo, three of its employees were approached by the blackmailer via email at the end of September, the company reports. These incidents were immediately disclosed and the Central Criminal Police launched a criminal investigation.
In addition, several agencies were contacted, including the Finnish Cyber Security Center, the Data Protection Commission, and a cyber security company.
Investigators believe the breach, which led to the customer database theft, occurred back in November 2018. In addition, security “deficiencies” remained until March 2019.
“We do not know that the database was stolen after November 2018, but it is possible that individual data [have been] viewed or copied,” Vastaamo said in a press release. No additional “vulnerabilities were identified after March 2019.”
The center’s CEO, Ville Tapio, who did not disclose any of these incidents to the parent company and its board of directors, was subsequently fired.
Once the police investigation began, Vastaamo said it was not granted permission by the authorities to communicate the occurrence to its patients. However, after the blackmailer released some patient information online early on Oct. 21, permission to inform patients was granted.
The company noted that the blackmailer has started emailing victims, informing of the data breach, and demanding ransom. So far, the emails have not contained harmful digital content or “malware,” but authorities warn that any attachments should not be opened. The police have requested that such emails be kept so they can be used as evidence.
In a Q&A section on its website, Vastaamo noted that videos are never recorded during its centers’ telehealth sessions and patients should not be concerned about the possibility of leaked videos.
In addition, the cybercrime has not interrupted Vastaamo’s operations.
“The authorities and the response office will do their utmost to find out what happened, to prevent the dissemination of information, and to bring the perpetrators to justice,” the center said.
“The most important task ... is to support customers in the midst of an exceptionally serious and difficult situation,” it added.
“Worst-case scenario”
In a comment, Ipsit Vahia, MD, medical director at the Institute for Technology and Psychiatry at McLean Hospital, Belmont, Mass., said Vastaamo’s data breach “represents the worst-case scenario for digital health.”
He added that more information is needed about the specifics of the case, including exactly what happened, how the system was hacked, and what information was compromised.
Still, “it raises fundamental questions that healthcare systems, clinicians, and patients everywhere should be asking about what measures are in place to protect electronic medical records and other personal digital information,” said Dr. Vahia.
“This incident also serves as another reminder that the issue of data security and privacy is foundational to digital mental health. Ultimately, without a commitment from all stakeholders to maintaining the strictest levels of security, as well as transparency around how data are handled there will be little to no trust from clinicians or patients,” he said. All of that could prevent digital healthcare from achieving its full potential, he added.
In addition, Dr. Vahia noted that the rapid uptick of telemedicine because of the pandemic has accelerated the use of other forms of digital information in mental healthcare.
“This unfortunate incident should serve as a wake-up call and bring the issue of data protection back firmly into the spotlight,” said Dr. Vahia.
Now that telehealth has become a larger part of clinical practice, said Torous, it’s important for clinicians to be vigilant regarding security procedures.
“Telehealth and digital data are here to stay, and with them new benefits as well as risks. We can continue to work to minimize the risks and protect privacy while ensuring the benefits to patients expand,” he added.
This article first appeared on Medscape.com.
Hackers have accessed patient records at Finland’s largest private psychotherapy system, emailing some patients to pay up or face having their private medical records released online.
Vastaamo treats about 40,000 patients and runs 25 centers across the country. Hackers emailed some of the centers’ patients asking for a blackmail payment of 200 euro in bitcoin, The Guardian reports.
Agencies such as the country’s National Bureau of Investigation are urging victims not to comply with the blackmailers’ demand and instead requesting that patients report these incidents to authorities and turn over incriminating emails. However, some data from patient records have already been released online.
“We deeply regret what happened and on behalf of our [patients] who have been compromised, we apologize for the shortcoming in data security, the consequences and human cost of which have been extremely heavy,” the center said in a statement. They added that the investigation into the situation is ongoing.
‘Sobering reminder’
In a comment, John Torous, MD, director of digital psychiatry at Beth Israel Deaconess Medical Center, Boston, Massachusetts, said this is “a sobering reminder that any digital data is subject to hacking.”
Torous is also chair of the American Psychiatric Association’s Health and Technology Committee.
“This is not the first time psychotherapy notes have been targeted and it actually happened, on a smaller scale, in the US in 2017,” he said.
In April of that year, confidential patient record information from a mental health center in Maine, including evaluations, session notes, and names of sex-abuse victims, was listed on the dark web.
Also in April, computer hackers released the WannaCry virus into the operating system of the United Kingdom’s National Health Service, which subsequently locked clinicians out of patient records and other digital tools for 3 days.
In addition, in 2016 hackers took Hollywood Presbyterian Medical Center in Los Angeles offline for more than a week after demanding a ransom of $3.6 million.
Criminal investigation
For Vastaamo, three of its employees were approached by the blackmailer via email at the end of September, the company reports. These incidents were immediately disclosed and the Central Criminal Police launched a criminal investigation.
In addition, several agencies were contacted, including the Finnish Cyber Security Center, the Data Protection Commission, and a cyber security company.
Investigators believe the breach, which led to the customer database theft, occurred back in November 2018. In addition, security “deficiencies” remained until March 2019.
“We do not know that the database was stolen after November 2018, but it is possible that individual data [have been] viewed or copied,” Vastaamo said in a press release. No additional “vulnerabilities were identified after March 2019.”
The center’s CEO, Ville Tapio, who did not disclose any of these incidents to the parent company and its board of directors, was subsequently fired.
Once the police investigation began, Vastaamo said it was not granted permission by the authorities to communicate the occurrence to its patients. However, after the blackmailer released some patient information online early on Oct. 21, permission to inform patients was granted.
The company noted that the blackmailer has started emailing victims, informing of the data breach, and demanding ransom. So far, the emails have not contained harmful digital content or “malware,” but authorities warn that any attachments should not be opened. The police have requested that such emails be kept so they can be used as evidence.
In a Q&A section on its website, Vastaamo noted that videos are never recorded during its centers’ telehealth sessions and patients should not be concerned about the possibility of leaked videos.
In addition, the cybercrime has not interrupted Vastaamo’s operations.
“The authorities and the response office will do their utmost to find out what happened, to prevent the dissemination of information, and to bring the perpetrators to justice,” the center said.
“The most important task ... is to support customers in the midst of an exceptionally serious and difficult situation,” it added.
“Worst-case scenario”
In a comment, Ipsit Vahia, MD, medical director at the Institute for Technology and Psychiatry at McLean Hospital, Belmont, Mass., said Vastaamo’s data breach “represents the worst-case scenario for digital health.”
He added that more information is needed about the specifics of the case, including exactly what happened, how the system was hacked, and what information was compromised.
Still, “it raises fundamental questions that healthcare systems, clinicians, and patients everywhere should be asking about what measures are in place to protect electronic medical records and other personal digital information,” said Dr. Vahia.
“This incident also serves as another reminder that the issue of data security and privacy is foundational to digital mental health. Ultimately, without a commitment from all stakeholders to maintaining the strictest levels of security, as well as transparency around how data are handled there will be little to no trust from clinicians or patients,” he said. All of that could prevent digital healthcare from achieving its full potential, he added.
In addition, Dr. Vahia noted that the rapid uptick of telemedicine because of the pandemic has accelerated the use of other forms of digital information in mental healthcare.
“This unfortunate incident should serve as a wake-up call and bring the issue of data protection back firmly into the spotlight,” said Dr. Vahia.
Now that telehealth has become a larger part of clinical practice, said Torous, it’s important for clinicians to be vigilant regarding security procedures.
“Telehealth and digital data are here to stay, and with them new benefits as well as risks. We can continue to work to minimize the risks and protect privacy while ensuring the benefits to patients expand,” he added.
This article first appeared on Medscape.com.
Decide ADHD pharmacotherapy based on medication onset, duration of action
Clinicians have numerous pharmacotherapy options available to treat ADHD in their toolbox. How do you know which formulation or combination of therapies is right for your patient with ADHD?
According to Jeffrey R. Strawn, MD, the answer depends on onset and duration of the medication and how that fits in to the patient’s current needs.
The most common treatment for ADHD, stimulants, are amphetamine-based and methylphenidate-based compounds known for improving core symptoms of inattention, impulsivity, and hyperactivity and are “probably associated with the most efficacy relative to the other interventions,” Dr. Strawn, associate professor of psychiatry, pediatrics, and clinical pharmacology at Cincinnati Children’s Hospital Medical Center, said at Psychopharmacology Update presented by Current Psychiatry and Global Academy for Medical Education. “But what I think is also really important for us to remember as clinicians is that they improve adherence, social interactions, [and] academic efficiency as well as accuracy.”
Other ADHD pharmacotherapy options include nonstimulant norepinephrine reuptake inhibitors (NRIs) like atomoxetine, and alpha-2 agonists like the extended-release forms of guanfacine and clonidine. All are Food and Drug Administration–approved for the treatment of ADHD, and the FDA has approved some combination alpha-2 agonists and stimulants treatments for ADHD as well.
When making decisions about formulations for ADHD pharmacotherapy, clinicians should think about whether the patient has issues swallowing tablets or capsules. Tablets, capsules, and chewable tablets may be appropriate for patients who can easily take these medications, while patients who have problems with swallowing pills may benefit from dissolvable tablets, solutions, and transdermal applications. Each of these options “have differences in terms of absorption, also differences in terms of intestinal transit time in younger children, as well as patients perhaps with irritable bowel, as well as other conditions that may affect absorption,” Dr. Strawn said. Different formulations have unique considerations: liquid formulations have the benefit of making precise adjustments, sublingual formulations may have quick absorption and onset, and oral dissolvable tablets can improve treatment adherence and reduce misuse of medication.
Formulations can be available as a delayed release, extended release, pulsatile release, targeted release, or a combination of immediate, delayed, and/or extended release. “Ultimately, what this gives rise to is differences in onset of action and duration, as well as differences in the elimination profile of the medication,” he said.
Transdermal formulations “avoid the first-pass metabolism, which may reduce side effects or increase efficacy,” but patients converting from an oral formulation may require reducing the dose. “It’s always important to remember, for example, with something like Daytrana, the transdermal methylphenidate formulation, if we’re converting a patient from an oral methylphenidate, we roughly need to use half the dose for the transdermal formulation,” Dr. Strawn explained. Transdermal formulations can carry benefits of steady plasma concentrations and longer duration of action but may cause skin irritation or accidentally be removed. “It’s really important they’re properly disposed of because oftentimes they do contain some active medication within the residual matrix.”
Methylphenidate, mixed amphetamine salt–based preparations
Modified-release formulations include matrix- or reservoir-based formulations and are most importantly differentiated from other formulations by their gastrointestinal (GI) transit time and the permeation through the GI membrane. When considering what formulation to choose, “it’s important to consider that, even with an ‘extended release formulation,’ all of these medications have some percentage that is immediately released, and that percentage varies considerably from formulation to formulation,” Dr. Strawn said.
He noted that brand names are sometimes used for formulations “because it’s often very difficult for us as clinicians and even for pharmacists to distinguish between these various formulations of the medication, which often have the same ‘extended’ or ‘delayed release’ modifying term within the name of the medication.”
Examples of medications that have greater immediate release include Metadate CD (30%), Aptensio XR (37%), long-acting methylphenidate (50%), dexmethylphenidate extended-release (50%), and Mixed Salts amphetamine extended release (50%). Formulations with a less immediate release include Quillivant solution or Quillichew chewable tablets (20%), Dyanaval XR solution (20%), OROS methylphenidate (22%), Daytrana that begins within 1 or 2 hours and lasts for 9 hours, or lisdexamfetamine that begins within 1 hour and lasts for 9 hours.
Depending on a patient’s needs, one particular formulation may work better than another. Dexmethylphenidate (Focalin XR) has a 50% immediate release and 50% extended release formulation, which “may be really important for a high school student who has first period precalculus followed by second period human geography,” Dr. Strawn said, while “a patient who may have first period study hall and second period art” may benefit from OROS methylphenidate.
Clinicians should also consider the effect of counterclockwise hysteresis when adding a short-acting stimulant later in the day. “There seems to be something really magic about having that ascending concentration time curve that, when we’re on the descending loop of that concentration time curve, we really seem to get a dramatic waning of the effect of the medication, even though technically the concentration is within the ‘therapeutic range,’ ” Dr. Strawn said. “With counterclockwise hysteresis, we see that the effect increases with time for a given concentration of the medication.”
Combining ADHD pharmacotherapies
For children and adolescents with ADHD, atomoxetine is a nonstimulant, FDA-approved treatment option. “It seems to be effective not just in terms of total ADHD symptoms, but also in terms of hyperactive and impulsive symptoms as well as the inattentive symptoms,” Dr. Strawn said.
Pharmacogenetics can be a guide for selecting an atomoxetine for a patient with ADHD, he noted. “What I think is most relevant here is the way in which pharmacogenetics can actually help guide our dosing, which then optimizes tolerability, potentially efficacy of atomoxetine,” he said. “Atomoxetine is pretty extensively metabolized by [CYP]2D6, and it’s one of about 300 medications that actually has specific labeling from the FDA on dosing based on genotype. It recommends a slower titration, as well as a lower target dose of atomoxetine in individuals who are P450 2D6 poor metabolizers relative to those patients who are ultra-rapid or normal metabolizers.”
Atomoxetine is most often combined with methylphenidate and has some evidence of benefit in children or adolescents who do not have an adequate response to stimulants alone. When combining stimulants with the alpha-2 agonists guanfacine or clonidine, “there are some improvements in terms of the combination treatment relative to the monotherapy,” Dr. Strawn said. He also emphasized that patients taking guanfacine immediate release tend to have better absorption and faster onset, compared with the extended release formulation. “This is something that potentially is very important when we think beyond steady state and we think about the practical use of this medication,” he said.
Baseline history is important
Overall, taking a baseline history of a patient with ADHD is “critically important” before starting them on stimulants, Dr. Strawn said. “Specifically, I would recommend documenting a negative history of syncope, family history of sudden cardiac death, as well as the lack of any known history of structural cardiac abnormalities,” he said. “Without a consultation with the cardiologist specifically around this question, I’m very, very, very hesitant – as in I don’t – use stimulants in patients who have histories of aortic stenosis, Wolff-Parkinson-White, as well as arrhythmogenic right ventricular dysplasia.”
Although patients with ADHD were typically followed with routine hemodynamic monitoring every 3 months prior to the COVID-19 pandemic, some clinicians see their patients with ADHD less frequently if they have been stabilized on a stimulant. , particularly in young children. In adults, it may also be very helpful to talk with spouses,” Dr. Strawn said.
Dr. Strawn also called attention to a recommendation to perform a routine electrocardiogram (EKG) in patients with ADHD who might receive stimulants. “At present, there is no recommendation to obtain a routine screening EKG in these patients, provided that we have an absence of those other red flags on the history,” he said. “Certainly, I would consider it in situations where I do have persistent tachycardia or hypertension, or there are other treatment-emergent symptoms, although really in many of these situations, I’m actually speaking on the phone with my pediatric or adult cardiology colleagues.”
Global Academy and this news organization are owned by the same parent company. Dr. Strawn reported receiving research support from Allergan, the FDA, the National Institutes of Health, Neuronetics, and Otsuka; serving as a consultant and receiving material support from Myriad; receiving royalties from Springer Publishing; and serving as a consultant for Intra-Cellular Therapies. In addition, he has been on the speaker’s bureau for the Neuroscience Education Institute and CMEology, and Medscape.
Clinicians have numerous pharmacotherapy options available to treat ADHD in their toolbox. How do you know which formulation or combination of therapies is right for your patient with ADHD?
According to Jeffrey R. Strawn, MD, the answer depends on onset and duration of the medication and how that fits in to the patient’s current needs.
The most common treatment for ADHD, stimulants, are amphetamine-based and methylphenidate-based compounds known for improving core symptoms of inattention, impulsivity, and hyperactivity and are “probably associated with the most efficacy relative to the other interventions,” Dr. Strawn, associate professor of psychiatry, pediatrics, and clinical pharmacology at Cincinnati Children’s Hospital Medical Center, said at Psychopharmacology Update presented by Current Psychiatry and Global Academy for Medical Education. “But what I think is also really important for us to remember as clinicians is that they improve adherence, social interactions, [and] academic efficiency as well as accuracy.”
Other ADHD pharmacotherapy options include nonstimulant norepinephrine reuptake inhibitors (NRIs) like atomoxetine, and alpha-2 agonists like the extended-release forms of guanfacine and clonidine. All are Food and Drug Administration–approved for the treatment of ADHD, and the FDA has approved some combination alpha-2 agonists and stimulants treatments for ADHD as well.
When making decisions about formulations for ADHD pharmacotherapy, clinicians should think about whether the patient has issues swallowing tablets or capsules. Tablets, capsules, and chewable tablets may be appropriate for patients who can easily take these medications, while patients who have problems with swallowing pills may benefit from dissolvable tablets, solutions, and transdermal applications. Each of these options “have differences in terms of absorption, also differences in terms of intestinal transit time in younger children, as well as patients perhaps with irritable bowel, as well as other conditions that may affect absorption,” Dr. Strawn said. Different formulations have unique considerations: liquid formulations have the benefit of making precise adjustments, sublingual formulations may have quick absorption and onset, and oral dissolvable tablets can improve treatment adherence and reduce misuse of medication.
Formulations can be available as a delayed release, extended release, pulsatile release, targeted release, or a combination of immediate, delayed, and/or extended release. “Ultimately, what this gives rise to is differences in onset of action and duration, as well as differences in the elimination profile of the medication,” he said.
Transdermal formulations “avoid the first-pass metabolism, which may reduce side effects or increase efficacy,” but patients converting from an oral formulation may require reducing the dose. “It’s always important to remember, for example, with something like Daytrana, the transdermal methylphenidate formulation, if we’re converting a patient from an oral methylphenidate, we roughly need to use half the dose for the transdermal formulation,” Dr. Strawn explained. Transdermal formulations can carry benefits of steady plasma concentrations and longer duration of action but may cause skin irritation or accidentally be removed. “It’s really important they’re properly disposed of because oftentimes they do contain some active medication within the residual matrix.”
Methylphenidate, mixed amphetamine salt–based preparations
Modified-release formulations include matrix- or reservoir-based formulations and are most importantly differentiated from other formulations by their gastrointestinal (GI) transit time and the permeation through the GI membrane. When considering what formulation to choose, “it’s important to consider that, even with an ‘extended release formulation,’ all of these medications have some percentage that is immediately released, and that percentage varies considerably from formulation to formulation,” Dr. Strawn said.
He noted that brand names are sometimes used for formulations “because it’s often very difficult for us as clinicians and even for pharmacists to distinguish between these various formulations of the medication, which often have the same ‘extended’ or ‘delayed release’ modifying term within the name of the medication.”
Examples of medications that have greater immediate release include Metadate CD (30%), Aptensio XR (37%), long-acting methylphenidate (50%), dexmethylphenidate extended-release (50%), and Mixed Salts amphetamine extended release (50%). Formulations with a less immediate release include Quillivant solution or Quillichew chewable tablets (20%), Dyanaval XR solution (20%), OROS methylphenidate (22%), Daytrana that begins within 1 or 2 hours and lasts for 9 hours, or lisdexamfetamine that begins within 1 hour and lasts for 9 hours.
Depending on a patient’s needs, one particular formulation may work better than another. Dexmethylphenidate (Focalin XR) has a 50% immediate release and 50% extended release formulation, which “may be really important for a high school student who has first period precalculus followed by second period human geography,” Dr. Strawn said, while “a patient who may have first period study hall and second period art” may benefit from OROS methylphenidate.
Clinicians should also consider the effect of counterclockwise hysteresis when adding a short-acting stimulant later in the day. “There seems to be something really magic about having that ascending concentration time curve that, when we’re on the descending loop of that concentration time curve, we really seem to get a dramatic waning of the effect of the medication, even though technically the concentration is within the ‘therapeutic range,’ ” Dr. Strawn said. “With counterclockwise hysteresis, we see that the effect increases with time for a given concentration of the medication.”
Combining ADHD pharmacotherapies
For children and adolescents with ADHD, atomoxetine is a nonstimulant, FDA-approved treatment option. “It seems to be effective not just in terms of total ADHD symptoms, but also in terms of hyperactive and impulsive symptoms as well as the inattentive symptoms,” Dr. Strawn said.
Pharmacogenetics can be a guide for selecting an atomoxetine for a patient with ADHD, he noted. “What I think is most relevant here is the way in which pharmacogenetics can actually help guide our dosing, which then optimizes tolerability, potentially efficacy of atomoxetine,” he said. “Atomoxetine is pretty extensively metabolized by [CYP]2D6, and it’s one of about 300 medications that actually has specific labeling from the FDA on dosing based on genotype. It recommends a slower titration, as well as a lower target dose of atomoxetine in individuals who are P450 2D6 poor metabolizers relative to those patients who are ultra-rapid or normal metabolizers.”
Atomoxetine is most often combined with methylphenidate and has some evidence of benefit in children or adolescents who do not have an adequate response to stimulants alone. When combining stimulants with the alpha-2 agonists guanfacine or clonidine, “there are some improvements in terms of the combination treatment relative to the monotherapy,” Dr. Strawn said. He also emphasized that patients taking guanfacine immediate release tend to have better absorption and faster onset, compared with the extended release formulation. “This is something that potentially is very important when we think beyond steady state and we think about the practical use of this medication,” he said.
Baseline history is important
Overall, taking a baseline history of a patient with ADHD is “critically important” before starting them on stimulants, Dr. Strawn said. “Specifically, I would recommend documenting a negative history of syncope, family history of sudden cardiac death, as well as the lack of any known history of structural cardiac abnormalities,” he said. “Without a consultation with the cardiologist specifically around this question, I’m very, very, very hesitant – as in I don’t – use stimulants in patients who have histories of aortic stenosis, Wolff-Parkinson-White, as well as arrhythmogenic right ventricular dysplasia.”
Although patients with ADHD were typically followed with routine hemodynamic monitoring every 3 months prior to the COVID-19 pandemic, some clinicians see their patients with ADHD less frequently if they have been stabilized on a stimulant. , particularly in young children. In adults, it may also be very helpful to talk with spouses,” Dr. Strawn said.
Dr. Strawn also called attention to a recommendation to perform a routine electrocardiogram (EKG) in patients with ADHD who might receive stimulants. “At present, there is no recommendation to obtain a routine screening EKG in these patients, provided that we have an absence of those other red flags on the history,” he said. “Certainly, I would consider it in situations where I do have persistent tachycardia or hypertension, or there are other treatment-emergent symptoms, although really in many of these situations, I’m actually speaking on the phone with my pediatric or adult cardiology colleagues.”
Global Academy and this news organization are owned by the same parent company. Dr. Strawn reported receiving research support from Allergan, the FDA, the National Institutes of Health, Neuronetics, and Otsuka; serving as a consultant and receiving material support from Myriad; receiving royalties from Springer Publishing; and serving as a consultant for Intra-Cellular Therapies. In addition, he has been on the speaker’s bureau for the Neuroscience Education Institute and CMEology, and Medscape.
Clinicians have numerous pharmacotherapy options available to treat ADHD in their toolbox. How do you know which formulation or combination of therapies is right for your patient with ADHD?
According to Jeffrey R. Strawn, MD, the answer depends on onset and duration of the medication and how that fits in to the patient’s current needs.
The most common treatment for ADHD, stimulants, are amphetamine-based and methylphenidate-based compounds known for improving core symptoms of inattention, impulsivity, and hyperactivity and are “probably associated with the most efficacy relative to the other interventions,” Dr. Strawn, associate professor of psychiatry, pediatrics, and clinical pharmacology at Cincinnati Children’s Hospital Medical Center, said at Psychopharmacology Update presented by Current Psychiatry and Global Academy for Medical Education. “But what I think is also really important for us to remember as clinicians is that they improve adherence, social interactions, [and] academic efficiency as well as accuracy.”
Other ADHD pharmacotherapy options include nonstimulant norepinephrine reuptake inhibitors (NRIs) like atomoxetine, and alpha-2 agonists like the extended-release forms of guanfacine and clonidine. All are Food and Drug Administration–approved for the treatment of ADHD, and the FDA has approved some combination alpha-2 agonists and stimulants treatments for ADHD as well.
When making decisions about formulations for ADHD pharmacotherapy, clinicians should think about whether the patient has issues swallowing tablets or capsules. Tablets, capsules, and chewable tablets may be appropriate for patients who can easily take these medications, while patients who have problems with swallowing pills may benefit from dissolvable tablets, solutions, and transdermal applications. Each of these options “have differences in terms of absorption, also differences in terms of intestinal transit time in younger children, as well as patients perhaps with irritable bowel, as well as other conditions that may affect absorption,” Dr. Strawn said. Different formulations have unique considerations: liquid formulations have the benefit of making precise adjustments, sublingual formulations may have quick absorption and onset, and oral dissolvable tablets can improve treatment adherence and reduce misuse of medication.
Formulations can be available as a delayed release, extended release, pulsatile release, targeted release, or a combination of immediate, delayed, and/or extended release. “Ultimately, what this gives rise to is differences in onset of action and duration, as well as differences in the elimination profile of the medication,” he said.
Transdermal formulations “avoid the first-pass metabolism, which may reduce side effects or increase efficacy,” but patients converting from an oral formulation may require reducing the dose. “It’s always important to remember, for example, with something like Daytrana, the transdermal methylphenidate formulation, if we’re converting a patient from an oral methylphenidate, we roughly need to use half the dose for the transdermal formulation,” Dr. Strawn explained. Transdermal formulations can carry benefits of steady plasma concentrations and longer duration of action but may cause skin irritation or accidentally be removed. “It’s really important they’re properly disposed of because oftentimes they do contain some active medication within the residual matrix.”
Methylphenidate, mixed amphetamine salt–based preparations
Modified-release formulations include matrix- or reservoir-based formulations and are most importantly differentiated from other formulations by their gastrointestinal (GI) transit time and the permeation through the GI membrane. When considering what formulation to choose, “it’s important to consider that, even with an ‘extended release formulation,’ all of these medications have some percentage that is immediately released, and that percentage varies considerably from formulation to formulation,” Dr. Strawn said.
He noted that brand names are sometimes used for formulations “because it’s often very difficult for us as clinicians and even for pharmacists to distinguish between these various formulations of the medication, which often have the same ‘extended’ or ‘delayed release’ modifying term within the name of the medication.”
Examples of medications that have greater immediate release include Metadate CD (30%), Aptensio XR (37%), long-acting methylphenidate (50%), dexmethylphenidate extended-release (50%), and Mixed Salts amphetamine extended release (50%). Formulations with a less immediate release include Quillivant solution or Quillichew chewable tablets (20%), Dyanaval XR solution (20%), OROS methylphenidate (22%), Daytrana that begins within 1 or 2 hours and lasts for 9 hours, or lisdexamfetamine that begins within 1 hour and lasts for 9 hours.
Depending on a patient’s needs, one particular formulation may work better than another. Dexmethylphenidate (Focalin XR) has a 50% immediate release and 50% extended release formulation, which “may be really important for a high school student who has first period precalculus followed by second period human geography,” Dr. Strawn said, while “a patient who may have first period study hall and second period art” may benefit from OROS methylphenidate.
Clinicians should also consider the effect of counterclockwise hysteresis when adding a short-acting stimulant later in the day. “There seems to be something really magic about having that ascending concentration time curve that, when we’re on the descending loop of that concentration time curve, we really seem to get a dramatic waning of the effect of the medication, even though technically the concentration is within the ‘therapeutic range,’ ” Dr. Strawn said. “With counterclockwise hysteresis, we see that the effect increases with time for a given concentration of the medication.”
Combining ADHD pharmacotherapies
For children and adolescents with ADHD, atomoxetine is a nonstimulant, FDA-approved treatment option. “It seems to be effective not just in terms of total ADHD symptoms, but also in terms of hyperactive and impulsive symptoms as well as the inattentive symptoms,” Dr. Strawn said.
Pharmacogenetics can be a guide for selecting an atomoxetine for a patient with ADHD, he noted. “What I think is most relevant here is the way in which pharmacogenetics can actually help guide our dosing, which then optimizes tolerability, potentially efficacy of atomoxetine,” he said. “Atomoxetine is pretty extensively metabolized by [CYP]2D6, and it’s one of about 300 medications that actually has specific labeling from the FDA on dosing based on genotype. It recommends a slower titration, as well as a lower target dose of atomoxetine in individuals who are P450 2D6 poor metabolizers relative to those patients who are ultra-rapid or normal metabolizers.”
Atomoxetine is most often combined with methylphenidate and has some evidence of benefit in children or adolescents who do not have an adequate response to stimulants alone. When combining stimulants with the alpha-2 agonists guanfacine or clonidine, “there are some improvements in terms of the combination treatment relative to the monotherapy,” Dr. Strawn said. He also emphasized that patients taking guanfacine immediate release tend to have better absorption and faster onset, compared with the extended release formulation. “This is something that potentially is very important when we think beyond steady state and we think about the practical use of this medication,” he said.
Baseline history is important
Overall, taking a baseline history of a patient with ADHD is “critically important” before starting them on stimulants, Dr. Strawn said. “Specifically, I would recommend documenting a negative history of syncope, family history of sudden cardiac death, as well as the lack of any known history of structural cardiac abnormalities,” he said. “Without a consultation with the cardiologist specifically around this question, I’m very, very, very hesitant – as in I don’t – use stimulants in patients who have histories of aortic stenosis, Wolff-Parkinson-White, as well as arrhythmogenic right ventricular dysplasia.”
Although patients with ADHD were typically followed with routine hemodynamic monitoring every 3 months prior to the COVID-19 pandemic, some clinicians see their patients with ADHD less frequently if they have been stabilized on a stimulant. , particularly in young children. In adults, it may also be very helpful to talk with spouses,” Dr. Strawn said.
Dr. Strawn also called attention to a recommendation to perform a routine electrocardiogram (EKG) in patients with ADHD who might receive stimulants. “At present, there is no recommendation to obtain a routine screening EKG in these patients, provided that we have an absence of those other red flags on the history,” he said. “Certainly, I would consider it in situations where I do have persistent tachycardia or hypertension, or there are other treatment-emergent symptoms, although really in many of these situations, I’m actually speaking on the phone with my pediatric or adult cardiology colleagues.”
Global Academy and this news organization are owned by the same parent company. Dr. Strawn reported receiving research support from Allergan, the FDA, the National Institutes of Health, Neuronetics, and Otsuka; serving as a consultant and receiving material support from Myriad; receiving royalties from Springer Publishing; and serving as a consultant for Intra-Cellular Therapies. In addition, he has been on the speaker’s bureau for the Neuroscience Education Institute and CMEology, and Medscape.
FROM PSYCHOPHARMACOLOGY UPDATE
Vertebral fractures in COVID-19 linked to mortality
Vertebral fractures appear to be common in people with severe COVID-19, and also raise the mortality risk, findings from a retrospective cohort suggest.
Among 114 patients with COVID-19 who underwent lateral chest x-rays at the San Raffaele Hospital ED in Milan, more than a third were found to have thoracic vertebral fractures. And, those individuals were more than twice as likely to die as were those without vertebral fractures.
“Morphometric vertebral fractures are one of the most common comorbidities among adults hospitalized with COVID-19, and the presence of such fractures may predict the severity of disease outcomes,” lead investigator Andrea Giustina, MD, said in an interview.
This is the first study to examine vertebral fracture prevalence in any coronavirus disease, but such fractures have been linked to an increased risk of pneumonia and impaired respiratory function, including restrictive pulmonary dysfunction. One possible mechanism may be that they cause anatomical changes, such as kyphosis, which negatively impact respiratory function by decreasing vital capacity, forced expiratory volume in 1 second, and inspiratory time, explained Dr. Giustina, professor of endocrinology, San Raffaele Vita Salute University, Milan, and president of the European Society of Endocrinology. The results were published in the Journal of Clinical Endocrinology and Metabolism.
Clinically, the findings suggest that all patients with COVID-19 who are undergoing chest x-rays should have morphometric vertebral x-ray evaluation, said Dr. Giustina.
“One interesting aspect of the study is that without morphometry, approximatively two thirds of vertebral fractures [would have been] missed. Therefore, they are largely underestimated in clinical practice,” he noted.
Thoracic vertebral fractures assessed via lateral chest x-rays
The 114 study subjects included were those whose lateral chest x-rays allowed for a high-quality assessment and in which all the thoracic tract of T4-T12 were viewable and assessable. None had been using glucocorticoids and only 3% had a prior diagnosis of osteoporosis.
The majority (75%) were male, and median age was 57 years. Most (79%) were hospitalized after evaluation in the ED. Of those, 12% (13) were admitted to the ICU and 15% (16) died.
Thoracic vertebral fractures were detected on the lateral chest x-rays in 36% (41) of the patients. In contrast, in studies of women aged 50 years and older from the general European population, morphometric vertebral fracture prevalence ranged from 18% to 26%, the investigators noted.
Of the total 65 vertebral fractures detected, 60% were classified as mild (height ratio decrease <25%), 33.3% as moderate (25%-40% decrease) and 7.7% as severe (>40%). Patients with more than one vertebral fracture were classified by their most severe one.
Those with versus without vertebral fractures didn’t differ by sex, body mass index, or clinical or biological parameters evaluated in the ED. But, compared with those without vertebral fractures, those with them were significantly older (68 vs. 54 years) and were more likely to have arterial hypertension (56% vs. 30%) and coronary artery disease (22% vs. 7%).
In multivariate analysis, age was the only statistically significant predictor of vertebral fractures (odds ratio, 1.04; P < .001).
Mortality doubled, though not significantly
Those with vertebral fractures were more likely to be hospitalized, although not significantly (88% vs. 74%). There was no significant difference in ICU admission (11% vs. 12.5%).
However, those with vertebral fractures required noninvasive mechanical ventilation significantly more often (48.8% vs. 27.4%; P = .02), and were more than twice as likely to die (22% vs. 10%; P = .07). While the difference in overall mortality wasn’t quite statistically significant, those with severe vertebral fractures were significantly more likely to die, compared with those with mild or moderate fractures (60%, 7%, 24%, respectively, for severe, moderate, and mild; P = .04), despite no significant differences in clinical or laboratory parameters.
“Our data from the field reinforce the need of implementing previously published recommendations concerning the importance of bone fragility care during the COVID pandemic with at least those patients already treated with antiosteoporotic drugs maintaining their adherence to treatments including vitamin D, which have also been suggested very recently to have no relevant predisposing effect on COVID-19,” Dr. Giustina and colleagues wrote.
Moreover, they added, “continuity of care should also include bone density monitoring despite very restricted access to clinical facilities, during the COVID-19 pandemic. Finally, all patients with fractures should start antiresorptive treatment right away, even during hospital stay.”
The authors reported having no disclosures.
SOURCE: Giustina A et al. J Clin Endocrinol Metab. 2020 Oct 21. doi: 10.1210/clinem/dgaa738.
Vertebral fractures appear to be common in people with severe COVID-19, and also raise the mortality risk, findings from a retrospective cohort suggest.
Among 114 patients with COVID-19 who underwent lateral chest x-rays at the San Raffaele Hospital ED in Milan, more than a third were found to have thoracic vertebral fractures. And, those individuals were more than twice as likely to die as were those without vertebral fractures.
“Morphometric vertebral fractures are one of the most common comorbidities among adults hospitalized with COVID-19, and the presence of such fractures may predict the severity of disease outcomes,” lead investigator Andrea Giustina, MD, said in an interview.
This is the first study to examine vertebral fracture prevalence in any coronavirus disease, but such fractures have been linked to an increased risk of pneumonia and impaired respiratory function, including restrictive pulmonary dysfunction. One possible mechanism may be that they cause anatomical changes, such as kyphosis, which negatively impact respiratory function by decreasing vital capacity, forced expiratory volume in 1 second, and inspiratory time, explained Dr. Giustina, professor of endocrinology, San Raffaele Vita Salute University, Milan, and president of the European Society of Endocrinology. The results were published in the Journal of Clinical Endocrinology and Metabolism.
Clinically, the findings suggest that all patients with COVID-19 who are undergoing chest x-rays should have morphometric vertebral x-ray evaluation, said Dr. Giustina.
“One interesting aspect of the study is that without morphometry, approximatively two thirds of vertebral fractures [would have been] missed. Therefore, they are largely underestimated in clinical practice,” he noted.
Thoracic vertebral fractures assessed via lateral chest x-rays
The 114 study subjects included were those whose lateral chest x-rays allowed for a high-quality assessment and in which all the thoracic tract of T4-T12 were viewable and assessable. None had been using glucocorticoids and only 3% had a prior diagnosis of osteoporosis.
The majority (75%) were male, and median age was 57 years. Most (79%) were hospitalized after evaluation in the ED. Of those, 12% (13) were admitted to the ICU and 15% (16) died.
Thoracic vertebral fractures were detected on the lateral chest x-rays in 36% (41) of the patients. In contrast, in studies of women aged 50 years and older from the general European population, morphometric vertebral fracture prevalence ranged from 18% to 26%, the investigators noted.
Of the total 65 vertebral fractures detected, 60% were classified as mild (height ratio decrease <25%), 33.3% as moderate (25%-40% decrease) and 7.7% as severe (>40%). Patients with more than one vertebral fracture were classified by their most severe one.
Those with versus without vertebral fractures didn’t differ by sex, body mass index, or clinical or biological parameters evaluated in the ED. But, compared with those without vertebral fractures, those with them were significantly older (68 vs. 54 years) and were more likely to have arterial hypertension (56% vs. 30%) and coronary artery disease (22% vs. 7%).
In multivariate analysis, age was the only statistically significant predictor of vertebral fractures (odds ratio, 1.04; P < .001).
Mortality doubled, though not significantly
Those with vertebral fractures were more likely to be hospitalized, although not significantly (88% vs. 74%). There was no significant difference in ICU admission (11% vs. 12.5%).
However, those with vertebral fractures required noninvasive mechanical ventilation significantly more often (48.8% vs. 27.4%; P = .02), and were more than twice as likely to die (22% vs. 10%; P = .07). While the difference in overall mortality wasn’t quite statistically significant, those with severe vertebral fractures were significantly more likely to die, compared with those with mild or moderate fractures (60%, 7%, 24%, respectively, for severe, moderate, and mild; P = .04), despite no significant differences in clinical or laboratory parameters.
“Our data from the field reinforce the need of implementing previously published recommendations concerning the importance of bone fragility care during the COVID pandemic with at least those patients already treated with antiosteoporotic drugs maintaining their adherence to treatments including vitamin D, which have also been suggested very recently to have no relevant predisposing effect on COVID-19,” Dr. Giustina and colleagues wrote.
Moreover, they added, “continuity of care should also include bone density monitoring despite very restricted access to clinical facilities, during the COVID-19 pandemic. Finally, all patients with fractures should start antiresorptive treatment right away, even during hospital stay.”
The authors reported having no disclosures.
SOURCE: Giustina A et al. J Clin Endocrinol Metab. 2020 Oct 21. doi: 10.1210/clinem/dgaa738.
Vertebral fractures appear to be common in people with severe COVID-19, and also raise the mortality risk, findings from a retrospective cohort suggest.
Among 114 patients with COVID-19 who underwent lateral chest x-rays at the San Raffaele Hospital ED in Milan, more than a third were found to have thoracic vertebral fractures. And, those individuals were more than twice as likely to die as were those without vertebral fractures.
“Morphometric vertebral fractures are one of the most common comorbidities among adults hospitalized with COVID-19, and the presence of such fractures may predict the severity of disease outcomes,” lead investigator Andrea Giustina, MD, said in an interview.
This is the first study to examine vertebral fracture prevalence in any coronavirus disease, but such fractures have been linked to an increased risk of pneumonia and impaired respiratory function, including restrictive pulmonary dysfunction. One possible mechanism may be that they cause anatomical changes, such as kyphosis, which negatively impact respiratory function by decreasing vital capacity, forced expiratory volume in 1 second, and inspiratory time, explained Dr. Giustina, professor of endocrinology, San Raffaele Vita Salute University, Milan, and president of the European Society of Endocrinology. The results were published in the Journal of Clinical Endocrinology and Metabolism.
Clinically, the findings suggest that all patients with COVID-19 who are undergoing chest x-rays should have morphometric vertebral x-ray evaluation, said Dr. Giustina.
“One interesting aspect of the study is that without morphometry, approximatively two thirds of vertebral fractures [would have been] missed. Therefore, they are largely underestimated in clinical practice,” he noted.
Thoracic vertebral fractures assessed via lateral chest x-rays
The 114 study subjects included were those whose lateral chest x-rays allowed for a high-quality assessment and in which all the thoracic tract of T4-T12 were viewable and assessable. None had been using glucocorticoids and only 3% had a prior diagnosis of osteoporosis.
The majority (75%) were male, and median age was 57 years. Most (79%) were hospitalized after evaluation in the ED. Of those, 12% (13) were admitted to the ICU and 15% (16) died.
Thoracic vertebral fractures were detected on the lateral chest x-rays in 36% (41) of the patients. In contrast, in studies of women aged 50 years and older from the general European population, morphometric vertebral fracture prevalence ranged from 18% to 26%, the investigators noted.
Of the total 65 vertebral fractures detected, 60% were classified as mild (height ratio decrease <25%), 33.3% as moderate (25%-40% decrease) and 7.7% as severe (>40%). Patients with more than one vertebral fracture were classified by their most severe one.
Those with versus without vertebral fractures didn’t differ by sex, body mass index, or clinical or biological parameters evaluated in the ED. But, compared with those without vertebral fractures, those with them were significantly older (68 vs. 54 years) and were more likely to have arterial hypertension (56% vs. 30%) and coronary artery disease (22% vs. 7%).
In multivariate analysis, age was the only statistically significant predictor of vertebral fractures (odds ratio, 1.04; P < .001).
Mortality doubled, though not significantly
Those with vertebral fractures were more likely to be hospitalized, although not significantly (88% vs. 74%). There was no significant difference in ICU admission (11% vs. 12.5%).
However, those with vertebral fractures required noninvasive mechanical ventilation significantly more often (48.8% vs. 27.4%; P = .02), and were more than twice as likely to die (22% vs. 10%; P = .07). While the difference in overall mortality wasn’t quite statistically significant, those with severe vertebral fractures were significantly more likely to die, compared with those with mild or moderate fractures (60%, 7%, 24%, respectively, for severe, moderate, and mild; P = .04), despite no significant differences in clinical or laboratory parameters.
“Our data from the field reinforce the need of implementing previously published recommendations concerning the importance of bone fragility care during the COVID pandemic with at least those patients already treated with antiosteoporotic drugs maintaining their adherence to treatments including vitamin D, which have also been suggested very recently to have no relevant predisposing effect on COVID-19,” Dr. Giustina and colleagues wrote.
Moreover, they added, “continuity of care should also include bone density monitoring despite very restricted access to clinical facilities, during the COVID-19 pandemic. Finally, all patients with fractures should start antiresorptive treatment right away, even during hospital stay.”
The authors reported having no disclosures.
SOURCE: Giustina A et al. J Clin Endocrinol Metab. 2020 Oct 21. doi: 10.1210/clinem/dgaa738.
FROM THE JOURNAL OF CLINICAL ENDOCRINOLOGY AND METABOLISM
Brain imaging reveals a neural basis for partisan politics
The differences between politically left- and right-leaning individuals may have neural underpinnings, results from a new brain imaging study suggest.
Investigators found that despite watching the same videos related to immigration policy, neural responses differed between liberals and conservatives.
“This divergence was strongest when the videos used language that highlighted threat, morality, and emotions, suggesting that certain words are more likely to drive polarized response,” lead researcher Yuan Chang Leong, PhD, a postdoctoral scholar in cognitive neuroscience at the University of California, Berkeley, told Medscape Medical News.
“The results suggest a neural basis for partisan biases in interpreting political messages, the effects these biases have on attitude change, and the type of language most likely to drive biased interpretations,” Leong added.
The study was published online Oct. 20 in Proceedings of the National Academy of Sciences.
Hardwired to disagree?
The researchers combined fMRI with semantic content analysis to investigate neural mechanisms that underlie the biased processing of political content.
They scanned 38 middle-aged men and women with liberal- or conservative-leaning immigration attitudes while the participants watched short news clips, campaign ads, and public speeches related to various immigration policies.
These policies included those that led to the United States–Mexico border wall, Deferred Action for Childhood Arrivals (DACA) protections for undocumented immigrants, the ban on refugees from majority-Muslim countries coming to the United States, and the cutting of federal funding to sanctuary cities.
After each video, participants rated on a scale of 1 to 5 how much they agreed with the general message of the video, the credibility of the information presented, and the extent to which the video made them likely to change their position and to support the policy in question.
The study revealed evidence of “neural polarization” – activity in the brain that diverges between people who hold liberal vs. conservative political views, the researchers reported.
Neural polarization was observed in the dorsomedial prefrontal cortex (DMPFC), a brain region associated with the interpretation of narrative content.
Neural polarization in this region intensified during moments in the videos that included risk-related and moral-emotional language, highlighting content features most likely to drive divergent interpretations between conservatives and liberals, they noted.
For a given individual, the closer that brain activity resembled that of the “average conservative” or “average liberal,” the more likely the person was to adopt that group’s position after watching the videos.
“We know that partisans respond differently to the same information. So in that sense, it’s not surprising to find that their brains respond differently as well,” Leong told Medscape Medical News.
“What we weren’t sure about was where in the brain we would find these differences, how neural differences were related to attitude change, and what type of content would be most likely to be associated with these differences,” he said.
Importantly, said Leong, these differences do not imply that people are hardwired to disagree. Rather, individual experiences and the media that is consumed likely contribute to neural polarization.
“ – for example, by framing messages to appeal to the core values of the respective voter,” he said.
Brain stimulation to alter political perception?
Reached for comment, Shaheen Lakhan, MD, PhD, neurologist in Newton, Mass., and executive director, Global Neuroscience Initiative Foundation, said the research “puts us one step closer to identifying how our brains interpret political information.”
The study, Lakhan noted, implicates a specific brain structure, the DMPFC, which is the “lens” in which information that gets into our brain is “viewed and acted on.”
“I trust that there will be plenty more work using a similar fMRI approach to tease out scenarios outside of immigration policy, as used in this study. Down the line, brain signatures through fMRI may be able to tell an individual’s political bent, and perhaps technologies like transcranial magnetic stimulation may be able to modulate our perceptions of political content, Shaheen said.
The research was supported by a grant from the Army Research Office. Leong and Lakhan have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The differences between politically left- and right-leaning individuals may have neural underpinnings, results from a new brain imaging study suggest.
Investigators found that despite watching the same videos related to immigration policy, neural responses differed between liberals and conservatives.
“This divergence was strongest when the videos used language that highlighted threat, morality, and emotions, suggesting that certain words are more likely to drive polarized response,” lead researcher Yuan Chang Leong, PhD, a postdoctoral scholar in cognitive neuroscience at the University of California, Berkeley, told Medscape Medical News.
“The results suggest a neural basis for partisan biases in interpreting political messages, the effects these biases have on attitude change, and the type of language most likely to drive biased interpretations,” Leong added.
The study was published online Oct. 20 in Proceedings of the National Academy of Sciences.
Hardwired to disagree?
The researchers combined fMRI with semantic content analysis to investigate neural mechanisms that underlie the biased processing of political content.
They scanned 38 middle-aged men and women with liberal- or conservative-leaning immigration attitudes while the participants watched short news clips, campaign ads, and public speeches related to various immigration policies.
These policies included those that led to the United States–Mexico border wall, Deferred Action for Childhood Arrivals (DACA) protections for undocumented immigrants, the ban on refugees from majority-Muslim countries coming to the United States, and the cutting of federal funding to sanctuary cities.
After each video, participants rated on a scale of 1 to 5 how much they agreed with the general message of the video, the credibility of the information presented, and the extent to which the video made them likely to change their position and to support the policy in question.
The study revealed evidence of “neural polarization” – activity in the brain that diverges between people who hold liberal vs. conservative political views, the researchers reported.
Neural polarization was observed in the dorsomedial prefrontal cortex (DMPFC), a brain region associated with the interpretation of narrative content.
Neural polarization in this region intensified during moments in the videos that included risk-related and moral-emotional language, highlighting content features most likely to drive divergent interpretations between conservatives and liberals, they noted.
For a given individual, the closer that brain activity resembled that of the “average conservative” or “average liberal,” the more likely the person was to adopt that group’s position after watching the videos.
“We know that partisans respond differently to the same information. So in that sense, it’s not surprising to find that their brains respond differently as well,” Leong told Medscape Medical News.
“What we weren’t sure about was where in the brain we would find these differences, how neural differences were related to attitude change, and what type of content would be most likely to be associated with these differences,” he said.
Importantly, said Leong, these differences do not imply that people are hardwired to disagree. Rather, individual experiences and the media that is consumed likely contribute to neural polarization.
“ – for example, by framing messages to appeal to the core values of the respective voter,” he said.
Brain stimulation to alter political perception?
Reached for comment, Shaheen Lakhan, MD, PhD, neurologist in Newton, Mass., and executive director, Global Neuroscience Initiative Foundation, said the research “puts us one step closer to identifying how our brains interpret political information.”
The study, Lakhan noted, implicates a specific brain structure, the DMPFC, which is the “lens” in which information that gets into our brain is “viewed and acted on.”
“I trust that there will be plenty more work using a similar fMRI approach to tease out scenarios outside of immigration policy, as used in this study. Down the line, brain signatures through fMRI may be able to tell an individual’s political bent, and perhaps technologies like transcranial magnetic stimulation may be able to modulate our perceptions of political content, Shaheen said.
The research was supported by a grant from the Army Research Office. Leong and Lakhan have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The differences between politically left- and right-leaning individuals may have neural underpinnings, results from a new brain imaging study suggest.
Investigators found that despite watching the same videos related to immigration policy, neural responses differed between liberals and conservatives.
“This divergence was strongest when the videos used language that highlighted threat, morality, and emotions, suggesting that certain words are more likely to drive polarized response,” lead researcher Yuan Chang Leong, PhD, a postdoctoral scholar in cognitive neuroscience at the University of California, Berkeley, told Medscape Medical News.
“The results suggest a neural basis for partisan biases in interpreting political messages, the effects these biases have on attitude change, and the type of language most likely to drive biased interpretations,” Leong added.
The study was published online Oct. 20 in Proceedings of the National Academy of Sciences.
Hardwired to disagree?
The researchers combined fMRI with semantic content analysis to investigate neural mechanisms that underlie the biased processing of political content.
They scanned 38 middle-aged men and women with liberal- or conservative-leaning immigration attitudes while the participants watched short news clips, campaign ads, and public speeches related to various immigration policies.
These policies included those that led to the United States–Mexico border wall, Deferred Action for Childhood Arrivals (DACA) protections for undocumented immigrants, the ban on refugees from majority-Muslim countries coming to the United States, and the cutting of federal funding to sanctuary cities.
After each video, participants rated on a scale of 1 to 5 how much they agreed with the general message of the video, the credibility of the information presented, and the extent to which the video made them likely to change their position and to support the policy in question.
The study revealed evidence of “neural polarization” – activity in the brain that diverges between people who hold liberal vs. conservative political views, the researchers reported.
Neural polarization was observed in the dorsomedial prefrontal cortex (DMPFC), a brain region associated with the interpretation of narrative content.
Neural polarization in this region intensified during moments in the videos that included risk-related and moral-emotional language, highlighting content features most likely to drive divergent interpretations between conservatives and liberals, they noted.
For a given individual, the closer that brain activity resembled that of the “average conservative” or “average liberal,” the more likely the person was to adopt that group’s position after watching the videos.
“We know that partisans respond differently to the same information. So in that sense, it’s not surprising to find that their brains respond differently as well,” Leong told Medscape Medical News.
“What we weren’t sure about was where in the brain we would find these differences, how neural differences were related to attitude change, and what type of content would be most likely to be associated with these differences,” he said.
Importantly, said Leong, these differences do not imply that people are hardwired to disagree. Rather, individual experiences and the media that is consumed likely contribute to neural polarization.
“ – for example, by framing messages to appeal to the core values of the respective voter,” he said.
Brain stimulation to alter political perception?
Reached for comment, Shaheen Lakhan, MD, PhD, neurologist in Newton, Mass., and executive director, Global Neuroscience Initiative Foundation, said the research “puts us one step closer to identifying how our brains interpret political information.”
The study, Lakhan noted, implicates a specific brain structure, the DMPFC, which is the “lens” in which information that gets into our brain is “viewed and acted on.”
“I trust that there will be plenty more work using a similar fMRI approach to tease out scenarios outside of immigration policy, as used in this study. Down the line, brain signatures through fMRI may be able to tell an individual’s political bent, and perhaps technologies like transcranial magnetic stimulation may be able to modulate our perceptions of political content, Shaheen said.
The research was supported by a grant from the Army Research Office. Leong and Lakhan have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
COVID-19: Immunity from antibodies may decline rapidly
Antibody response to the SARS-CoV-2 virus wanes over time, latest research has suggested.
An ongoing study led by Imperial College London (ICL) found that the proportion of people testing positive for COVID-19 antibodies dropped by 26.5% over a 3-month period between June and September.
The findings from a non–peer reviewed preprint suggested that infection with SARS-CoV-2 confers only limited protection against reinfection.
Professor Paul Elliott, director of the REACT-2 programme at ICL, said: “Testing positive for antibodies does not mean you are immune to COVID-19.
“It remains unclear what level of immunity antibodies provide, or for how long this immunity lasts.”
Experts said that, while the findings suggested that immunity might fade over time, the severity of illness from further infections could be reduced.
Antibody prevalence declined in all adults
Results from cross-sectional studies over the 3-month period involved 365,104 adults who self-administered a lateral flow immunoassay test.
There were 17,576 positive tests over the three rounds.
Antibody prevalence, adjusted for test characteristics and weighted to the adult population of England, declined from 6.0% to 4.4%, a reduction of 26.5% over the 3 months.
The decline was seen in all age groups. However, the lowest prevalence of a positive test, and the largest fall, was seen in those aged 75 years and older.
No change was seen in positive antibody tests in health care workers over the 3 months.
The results suggested that people who did not show symptoms of COVID-19 were more likely to lose detectable antibodies sooner than those who did show symptoms.
Prof Helen Ward, one of the lead authors of the report said that, while it was clear that the proportion of people with antibodies was falling over time, “We don’t yet know whether this will leave these people at risk of reinfection with the virus that causes COVID-19, but it is essential that everyone continues to follow guidance to reduce the risk to themselves and others.”
Results ‘weaken argument for herd immunity’
Commenting on the results to the Science Media Centre, Rowland Kao, professor of veterinary epidemiology and data science at the University of Edinburgh, warned that, if the results were correct, “any strategy that relies on ‘herd immunity’ lacks credibility.”
However, he added that, “while the decline is substantial, nevertheless substantial proportions of the population do retain some immune response, over 4 months after the peak of the epidemic”.
Eleanor Riley, professor of immunology and infectious disease, also from the University of Edinburgh, said it was too early to assume that immunity to SARS-CoV-2 did not last because “the study does not look at antibody concentrations, antibody function, or other aspects of immunity such as T-cell immunity and does not look at the trajectory of antibody levels in the same individuals over time”.
However, she said the findings did not mean that a vaccine would be ineffective because vaccines contained adjuvants that could induce durable immune responses, particularly with multiple immunizations.
“What is not clear is how quickly antibody levels would rise again if a person encounters the SARS-CoV-2 virus a second time. It is possible they will still rapidly respond, and either have a milder illness, or remain protected through immune memory,” commented Dr. Alexander Edwards, associate professor in biomedical technology at the University of Reading.
Health Minister Lord Bethell said: “Regardless of the result of an antibody test, everyone must continue to comply with government guidelines including social distancing, self-isolating, and getting a test if you have symptoms, and always remember: hands, face, space.”
This article first appeared on Medscape.com.
Antibody response to the SARS-CoV-2 virus wanes over time, latest research has suggested.
An ongoing study led by Imperial College London (ICL) found that the proportion of people testing positive for COVID-19 antibodies dropped by 26.5% over a 3-month period between June and September.
The findings from a non–peer reviewed preprint suggested that infection with SARS-CoV-2 confers only limited protection against reinfection.
Professor Paul Elliott, director of the REACT-2 programme at ICL, said: “Testing positive for antibodies does not mean you are immune to COVID-19.
“It remains unclear what level of immunity antibodies provide, or for how long this immunity lasts.”
Experts said that, while the findings suggested that immunity might fade over time, the severity of illness from further infections could be reduced.
Antibody prevalence declined in all adults
Results from cross-sectional studies over the 3-month period involved 365,104 adults who self-administered a lateral flow immunoassay test.
There were 17,576 positive tests over the three rounds.
Antibody prevalence, adjusted for test characteristics and weighted to the adult population of England, declined from 6.0% to 4.4%, a reduction of 26.5% over the 3 months.
The decline was seen in all age groups. However, the lowest prevalence of a positive test, and the largest fall, was seen in those aged 75 years and older.
No change was seen in positive antibody tests in health care workers over the 3 months.
The results suggested that people who did not show symptoms of COVID-19 were more likely to lose detectable antibodies sooner than those who did show symptoms.
Prof Helen Ward, one of the lead authors of the report said that, while it was clear that the proportion of people with antibodies was falling over time, “We don’t yet know whether this will leave these people at risk of reinfection with the virus that causes COVID-19, but it is essential that everyone continues to follow guidance to reduce the risk to themselves and others.”
Results ‘weaken argument for herd immunity’
Commenting on the results to the Science Media Centre, Rowland Kao, professor of veterinary epidemiology and data science at the University of Edinburgh, warned that, if the results were correct, “any strategy that relies on ‘herd immunity’ lacks credibility.”
However, he added that, “while the decline is substantial, nevertheless substantial proportions of the population do retain some immune response, over 4 months after the peak of the epidemic”.
Eleanor Riley, professor of immunology and infectious disease, also from the University of Edinburgh, said it was too early to assume that immunity to SARS-CoV-2 did not last because “the study does not look at antibody concentrations, antibody function, or other aspects of immunity such as T-cell immunity and does not look at the trajectory of antibody levels in the same individuals over time”.
However, she said the findings did not mean that a vaccine would be ineffective because vaccines contained adjuvants that could induce durable immune responses, particularly with multiple immunizations.
“What is not clear is how quickly antibody levels would rise again if a person encounters the SARS-CoV-2 virus a second time. It is possible they will still rapidly respond, and either have a milder illness, or remain protected through immune memory,” commented Dr. Alexander Edwards, associate professor in biomedical technology at the University of Reading.
Health Minister Lord Bethell said: “Regardless of the result of an antibody test, everyone must continue to comply with government guidelines including social distancing, self-isolating, and getting a test if you have symptoms, and always remember: hands, face, space.”
This article first appeared on Medscape.com.
Antibody response to the SARS-CoV-2 virus wanes over time, latest research has suggested.
An ongoing study led by Imperial College London (ICL) found that the proportion of people testing positive for COVID-19 antibodies dropped by 26.5% over a 3-month period between June and September.
The findings from a non–peer reviewed preprint suggested that infection with SARS-CoV-2 confers only limited protection against reinfection.
Professor Paul Elliott, director of the REACT-2 programme at ICL, said: “Testing positive for antibodies does not mean you are immune to COVID-19.
“It remains unclear what level of immunity antibodies provide, or for how long this immunity lasts.”
Experts said that, while the findings suggested that immunity might fade over time, the severity of illness from further infections could be reduced.
Antibody prevalence declined in all adults
Results from cross-sectional studies over the 3-month period involved 365,104 adults who self-administered a lateral flow immunoassay test.
There were 17,576 positive tests over the three rounds.
Antibody prevalence, adjusted for test characteristics and weighted to the adult population of England, declined from 6.0% to 4.4%, a reduction of 26.5% over the 3 months.
The decline was seen in all age groups. However, the lowest prevalence of a positive test, and the largest fall, was seen in those aged 75 years and older.
No change was seen in positive antibody tests in health care workers over the 3 months.
The results suggested that people who did not show symptoms of COVID-19 were more likely to lose detectable antibodies sooner than those who did show symptoms.
Prof Helen Ward, one of the lead authors of the report said that, while it was clear that the proportion of people with antibodies was falling over time, “We don’t yet know whether this will leave these people at risk of reinfection with the virus that causes COVID-19, but it is essential that everyone continues to follow guidance to reduce the risk to themselves and others.”
Results ‘weaken argument for herd immunity’
Commenting on the results to the Science Media Centre, Rowland Kao, professor of veterinary epidemiology and data science at the University of Edinburgh, warned that, if the results were correct, “any strategy that relies on ‘herd immunity’ lacks credibility.”
However, he added that, “while the decline is substantial, nevertheless substantial proportions of the population do retain some immune response, over 4 months after the peak of the epidemic”.
Eleanor Riley, professor of immunology and infectious disease, also from the University of Edinburgh, said it was too early to assume that immunity to SARS-CoV-2 did not last because “the study does not look at antibody concentrations, antibody function, or other aspects of immunity such as T-cell immunity and does not look at the trajectory of antibody levels in the same individuals over time”.
However, she said the findings did not mean that a vaccine would be ineffective because vaccines contained adjuvants that could induce durable immune responses, particularly with multiple immunizations.
“What is not clear is how quickly antibody levels would rise again if a person encounters the SARS-CoV-2 virus a second time. It is possible they will still rapidly respond, and either have a milder illness, or remain protected through immune memory,” commented Dr. Alexander Edwards, associate professor in biomedical technology at the University of Reading.
Health Minister Lord Bethell said: “Regardless of the result of an antibody test, everyone must continue to comply with government guidelines including social distancing, self-isolating, and getting a test if you have symptoms, and always remember: hands, face, space.”
This article first appeared on Medscape.com.
Valvular disease and COVID-19 are a deadly mix; don’t delay intervention
Danny Dvir, MD, has a message for physicians who have patients with severe valvular heart disease who are deferring valve replacement or repair until after the COVID-19 pandemic: Urge them not to wait.
Data from the Multicenter International Valve Disease Registry vividly demonstrate that clinical outcomes are poor in patients with uncorrected valve disease who become hospitalized with COVID-19. Indeed, the mortality rate within 30 days after hospital admission in 136 such patients enrolled in the registry from centers in Europe, North America, and Israel was 42%, Dr. Dvir reported at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
“That’s dramatically higher than for an age-matched population infected with COVID-19 without valvular heart disease, which is 10%-15%,” he noted at the meeting sponsored by the Cardiovascular Research Foundation.
The bright spot was that, in the small subgroup of 15 registry participants who underwent transcatheter or, much less frequently, surgical treatment of their failing valve while COVID-19 infected, 30-day mortality was far lower. In fact, it was comparable with the background rate in hospitalized COVID-19 patients without valve disease, according to Dr. Dvir, an interventional cardiologist at Shaare Zedek Medical Center, Hebrew University, Jerusalem.
He personally did several of the transcatheter aortic valve replacements.
“It’s doable. I truly believe that when you get a severe aortic stenosis patient who’s infected with the coronavirus, they get very unstable, but we can treat them. We can treat them even during the infection,” Dr. Dvir said.
The majority of patients in the registry had severe aortic stenosis. In the 42 such patients aged 80 years or more who didn’t undergo transcatheter aortic valve replacement (TAVR) or surgical valve replacement, 30-day mortality was 60%. In contrast, only one of the six patients in this advanced-age category who underwent valve replacement while infected died. Similarly, 30-day mortality was 24% among those younger than age 80 who valve remained untreated, but it dropped to 11% in those who received a prosthetic valve.
“We try our best to protect our patients through social distancing, but we have a treatment that can potentially reduce their mortality risk if they get infected later on. So I say to my patients: ‘Don’t wait at home. Do not wait! If you get infected when you have severe aortic stenosis, the clinical outcome is bad.’ But it seems reasonable that if they get infected when they’ve already been treated for their aortic stenosis or mitral regurgitation, they will do better.”
Dr. Dvir noted that, although the case numbers in the registry series were small and subject to potential bias, the data suggest this treatment approach may be lifesaving.
Session comoderator Timothy D. Henry, MD, commented that this registry study contains a great take-home point: “This is really consistent with what see in a lot of the other areas of COVID, that what we know to be best clinical care, we should do it, with or without the COVID.”
He asked Dr. Dvir about any special measures he takes while doing TAVR in this extreme setting. In the United States, for example, interventionalists are increasingly using transesophageal echocardiography to guide their procedures using conscious sedation, without intubation, noted Dr. Henry, medical director of the Carl and Edyth Lindner Center for Research at the Christ Hospital, Cincinnati.
“We try to minimize the procedure time; that’s one of the important things,” Dr. Dvir replied. “And you need to be protected during the procedure in a very cautious and meticulous way. You need many fans in the room because you sweat a lot.”
Discussant Renu Virmani, MD, president of the CVPath Institute in Gaithersburg, Md., commented: “The main thing I get from this presentation is the need for patients to be educated that if you’ve got valve disease, you’re better off getting it treated before you’ve got COVID. Obviously, try to prevent getting COVID – that’s the best thing you can do – but you can’t always control that.”
Discussant Mamas Mamas, MD, professor of cardiology at Keele University, Staffordshire, England, said deferred treatment of severe valvular heart disease during the pandemic has created a looming public health crisis in the United Kingdom.
“We’ve analyzed the U.K. management of aortic stenosis, and what we’ve found is that during the COVID pandemic there have been 2,500 fewer cases of aortic stenosis that have been treated. We’ve got 2,500 patients on the waiting list, and we’ve got to work out how we’re going to treat them. We estimate with simulations that about 300 of them are going to die before we can get them treated for their aortic stenosis,” according to Dr. Mamas.
Dr. Henry commented that deferral of valve procedures is “really challenging” for a couple of reasons: Not only are patients scared to come into the hospital because they fear getting COVID, but they don’t want to be hospitalized during the pandemic because their family can’t visit them there.
“These patients are mostly over 80 years old. No one wants to come in the hospital when the family won’t be around, especially when you’re 90 years old,” the interventional cardiologist said.
Dr. Dvir reported serving as a consultant to Medtronic, Edwards Lifesciences, Abbott, and Jena.
Danny Dvir, MD, has a message for physicians who have patients with severe valvular heart disease who are deferring valve replacement or repair until after the COVID-19 pandemic: Urge them not to wait.
Data from the Multicenter International Valve Disease Registry vividly demonstrate that clinical outcomes are poor in patients with uncorrected valve disease who become hospitalized with COVID-19. Indeed, the mortality rate within 30 days after hospital admission in 136 such patients enrolled in the registry from centers in Europe, North America, and Israel was 42%, Dr. Dvir reported at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
“That’s dramatically higher than for an age-matched population infected with COVID-19 without valvular heart disease, which is 10%-15%,” he noted at the meeting sponsored by the Cardiovascular Research Foundation.
The bright spot was that, in the small subgroup of 15 registry participants who underwent transcatheter or, much less frequently, surgical treatment of their failing valve while COVID-19 infected, 30-day mortality was far lower. In fact, it was comparable with the background rate in hospitalized COVID-19 patients without valve disease, according to Dr. Dvir, an interventional cardiologist at Shaare Zedek Medical Center, Hebrew University, Jerusalem.
He personally did several of the transcatheter aortic valve replacements.
“It’s doable. I truly believe that when you get a severe aortic stenosis patient who’s infected with the coronavirus, they get very unstable, but we can treat them. We can treat them even during the infection,” Dr. Dvir said.
The majority of patients in the registry had severe aortic stenosis. In the 42 such patients aged 80 years or more who didn’t undergo transcatheter aortic valve replacement (TAVR) or surgical valve replacement, 30-day mortality was 60%. In contrast, only one of the six patients in this advanced-age category who underwent valve replacement while infected died. Similarly, 30-day mortality was 24% among those younger than age 80 who valve remained untreated, but it dropped to 11% in those who received a prosthetic valve.
“We try our best to protect our patients through social distancing, but we have a treatment that can potentially reduce their mortality risk if they get infected later on. So I say to my patients: ‘Don’t wait at home. Do not wait! If you get infected when you have severe aortic stenosis, the clinical outcome is bad.’ But it seems reasonable that if they get infected when they’ve already been treated for their aortic stenosis or mitral regurgitation, they will do better.”
Dr. Dvir noted that, although the case numbers in the registry series were small and subject to potential bias, the data suggest this treatment approach may be lifesaving.
Session comoderator Timothy D. Henry, MD, commented that this registry study contains a great take-home point: “This is really consistent with what see in a lot of the other areas of COVID, that what we know to be best clinical care, we should do it, with or without the COVID.”
He asked Dr. Dvir about any special measures he takes while doing TAVR in this extreme setting. In the United States, for example, interventionalists are increasingly using transesophageal echocardiography to guide their procedures using conscious sedation, without intubation, noted Dr. Henry, medical director of the Carl and Edyth Lindner Center for Research at the Christ Hospital, Cincinnati.
“We try to minimize the procedure time; that’s one of the important things,” Dr. Dvir replied. “And you need to be protected during the procedure in a very cautious and meticulous way. You need many fans in the room because you sweat a lot.”
Discussant Renu Virmani, MD, president of the CVPath Institute in Gaithersburg, Md., commented: “The main thing I get from this presentation is the need for patients to be educated that if you’ve got valve disease, you’re better off getting it treated before you’ve got COVID. Obviously, try to prevent getting COVID – that’s the best thing you can do – but you can’t always control that.”
Discussant Mamas Mamas, MD, professor of cardiology at Keele University, Staffordshire, England, said deferred treatment of severe valvular heart disease during the pandemic has created a looming public health crisis in the United Kingdom.
“We’ve analyzed the U.K. management of aortic stenosis, and what we’ve found is that during the COVID pandemic there have been 2,500 fewer cases of aortic stenosis that have been treated. We’ve got 2,500 patients on the waiting list, and we’ve got to work out how we’re going to treat them. We estimate with simulations that about 300 of them are going to die before we can get them treated for their aortic stenosis,” according to Dr. Mamas.
Dr. Henry commented that deferral of valve procedures is “really challenging” for a couple of reasons: Not only are patients scared to come into the hospital because they fear getting COVID, but they don’t want to be hospitalized during the pandemic because their family can’t visit them there.
“These patients are mostly over 80 years old. No one wants to come in the hospital when the family won’t be around, especially when you’re 90 years old,” the interventional cardiologist said.
Dr. Dvir reported serving as a consultant to Medtronic, Edwards Lifesciences, Abbott, and Jena.
Danny Dvir, MD, has a message for physicians who have patients with severe valvular heart disease who are deferring valve replacement or repair until after the COVID-19 pandemic: Urge them not to wait.
Data from the Multicenter International Valve Disease Registry vividly demonstrate that clinical outcomes are poor in patients with uncorrected valve disease who become hospitalized with COVID-19. Indeed, the mortality rate within 30 days after hospital admission in 136 such patients enrolled in the registry from centers in Europe, North America, and Israel was 42%, Dr. Dvir reported at the Transcatheter Cardiovascular Research Therapeutics virtual annual meeting.
“That’s dramatically higher than for an age-matched population infected with COVID-19 without valvular heart disease, which is 10%-15%,” he noted at the meeting sponsored by the Cardiovascular Research Foundation.
The bright spot was that, in the small subgroup of 15 registry participants who underwent transcatheter or, much less frequently, surgical treatment of their failing valve while COVID-19 infected, 30-day mortality was far lower. In fact, it was comparable with the background rate in hospitalized COVID-19 patients without valve disease, according to Dr. Dvir, an interventional cardiologist at Shaare Zedek Medical Center, Hebrew University, Jerusalem.
He personally did several of the transcatheter aortic valve replacements.
“It’s doable. I truly believe that when you get a severe aortic stenosis patient who’s infected with the coronavirus, they get very unstable, but we can treat them. We can treat them even during the infection,” Dr. Dvir said.
The majority of patients in the registry had severe aortic stenosis. In the 42 such patients aged 80 years or more who didn’t undergo transcatheter aortic valve replacement (TAVR) or surgical valve replacement, 30-day mortality was 60%. In contrast, only one of the six patients in this advanced-age category who underwent valve replacement while infected died. Similarly, 30-day mortality was 24% among those younger than age 80 who valve remained untreated, but it dropped to 11% in those who received a prosthetic valve.
“We try our best to protect our patients through social distancing, but we have a treatment that can potentially reduce their mortality risk if they get infected later on. So I say to my patients: ‘Don’t wait at home. Do not wait! If you get infected when you have severe aortic stenosis, the clinical outcome is bad.’ But it seems reasonable that if they get infected when they’ve already been treated for their aortic stenosis or mitral regurgitation, they will do better.”
Dr. Dvir noted that, although the case numbers in the registry series were small and subject to potential bias, the data suggest this treatment approach may be lifesaving.
Session comoderator Timothy D. Henry, MD, commented that this registry study contains a great take-home point: “This is really consistent with what see in a lot of the other areas of COVID, that what we know to be best clinical care, we should do it, with or without the COVID.”
He asked Dr. Dvir about any special measures he takes while doing TAVR in this extreme setting. In the United States, for example, interventionalists are increasingly using transesophageal echocardiography to guide their procedures using conscious sedation, without intubation, noted Dr. Henry, medical director of the Carl and Edyth Lindner Center for Research at the Christ Hospital, Cincinnati.
“We try to minimize the procedure time; that’s one of the important things,” Dr. Dvir replied. “And you need to be protected during the procedure in a very cautious and meticulous way. You need many fans in the room because you sweat a lot.”
Discussant Renu Virmani, MD, president of the CVPath Institute in Gaithersburg, Md., commented: “The main thing I get from this presentation is the need for patients to be educated that if you’ve got valve disease, you’re better off getting it treated before you’ve got COVID. Obviously, try to prevent getting COVID – that’s the best thing you can do – but you can’t always control that.”
Discussant Mamas Mamas, MD, professor of cardiology at Keele University, Staffordshire, England, said deferred treatment of severe valvular heart disease during the pandemic has created a looming public health crisis in the United Kingdom.
“We’ve analyzed the U.K. management of aortic stenosis, and what we’ve found is that during the COVID pandemic there have been 2,500 fewer cases of aortic stenosis that have been treated. We’ve got 2,500 patients on the waiting list, and we’ve got to work out how we’re going to treat them. We estimate with simulations that about 300 of them are going to die before we can get them treated for their aortic stenosis,” according to Dr. Mamas.
Dr. Henry commented that deferral of valve procedures is “really challenging” for a couple of reasons: Not only are patients scared to come into the hospital because they fear getting COVID, but they don’t want to be hospitalized during the pandemic because their family can’t visit them there.
“These patients are mostly over 80 years old. No one wants to come in the hospital when the family won’t be around, especially when you’re 90 years old,” the interventional cardiologist said.
Dr. Dvir reported serving as a consultant to Medtronic, Edwards Lifesciences, Abbott, and Jena.
FROM TCT 2020
COVID-19: Thromboembolic events high despite prophylaxis
in a new large observational U.S. study.
“Despite very high rate of antithrombotic prophylaxis there were a high rate of thromboembolic events suggesting that we are probably not providing enough thromboprophylaxis,” lead author Gregory Piazza, MD, Brigham and Women’s Hospital, Boston, said in an interview.
“Standard prophylaxis as recommended in the guidelines is a low dose of low-molecular-weight heparin once daily, but these results suggest [patients] probably need higher doses,” he added.
However, Dr. Piazza cautioned that this is an observational study and randomized trials are needed to make changes in treatment strategies. Several such trials are currently underway.
The current study was published online ahead of print in the Nov. 3 issue of the Journal of the American College of Cardiology.
Rates similar to other very sick patients
The study showed that while thromboembolic complications were high, they were not as high as seen in some of the earlier studies from Asia and Europe, Dr. Piazza noted.
“The numbers we were seeing in early reports were so high we couldn’t figure out how that was possible,” he said. “Our study suggests that, in a U.S. population receiving thromboprophylaxis, the rate of thromboembolic complications [are] more in line with what we would expect to see in other very sick patients who end up in ICU.”
He suggested that the very high rates of thromboembolic complications in the early studies from Asia may have been because of the lack of thromboprophylaxis, which is not routine in hospitalized patients there. “Some of the earlier studies also used routine ultrasound and so picked up asymptomatic thrombotic events, which was not the case in our study. So our results are more representative of the U.S. population.”
Dr. Piazza attributed the high rate of thromboembolic complications being reported with COVID-19 to the sheer number of very sick patients being admitted to the hospital.
“We are accustomed to seeing a rare case of thrombosis despite prophylaxis in hospitalized patients, but we are seeing more in COVID patients. This is probably just because we have more critically ill patients,” he said.
“We are seeing an incredible influx of patients to the ICU that we have never experienced before, so the increase in thromboembolic complications is more obvious. In prior years we probably haven’t had enough critically ill patients at any one time to raise the flag about thromboprophylaxis,” he commented.
The study also found a high rate of cardiovascular complications. They are seeing an increase in the risk of MI, which is to be expected in such sick patients, but they also see quite a bit of new atrial fibrillation, myocarditis, and heart failure in patients who don’t always have underlying cardiovascular disease, he said.
“So this virus does appear to have a predilection to causing cardiovascular complications, but this is probably because it is making patients so sick,” Dr. Piazza said. “If flu was this virulent and resulted in such high rates of acute respiratory distress syndrome (ARDS), we would probably see similar cardiovascular complication rates.”
For the current report, the researchers analyzed a retrospective cohort of 1,114 patients with COVID-19 diagnosed through the Mass General Brigham integrated health network. Of these, 170 had been admitted to the ICU, 229 had been hospitalized but not treated in ICU, and 715 were outpatients. In terms of ethnicity, 22% were Hispanic/Latino and 44% were non-White.
Cardiovascular risk factors were common, with 36% of patients having hypertension, 29% hyperlipidemia, and 18% diabetes. Prophylactic anticoagulation was prescribed in 89% of patients with COVID-19 in the intensive care cohort and 85% of those in the hospitalized non–intensive care setting.
Results showed that major arterial or venous thromboembolism (VTE) occurred in 35% of the intensive care cohort, 2.6% of those hospitalized but not treated in ICU, and 0% of outpatients.
Major adverse cardiovascular events occurred in 46% of the intensive care cohort, 6.1% of those hospitalized but non-ICU, and 0% of outpatients.
Symptomatic VTE occurred in 27% of those admitted to ICU, 2.2% of those hospitalized but non-ICU, and 0% of outpatients.
“We found that outpatients had a very low rate of thromboembolic complications, with the vast majority of the risk being in hospitalized patients, especially those in ICU,” Dr. Piazza said.
“These results suggest that we don’t need routine thromboprophylaxis for all outpatients with COVID-19, but there will probably be some patients who need it – those with risk factors for thromboembolism.”
Catheter- and device-associated deep vein thrombosis accounted for 76.9% of the DVTs observed in the study.
“Our finding of high frequency of catheter-associated DVT supports the judicious use of central venous catheters that have been widely implemented, especially in the ICU, to minimize recurrent health care team exposure and facilitate monitoring,” the researchers wrote.
ARDS biggest risk factor
Of all the markers of disease severity, the presence of ARDS had the strongest association with adverse outcomes, including major arterial or VTE, major adverse cardiovascular events, symptomatic VTE, and death.
“The severe inflammatory state associated with ARDS and other complications of COVID-19 and its resultant hypercoagulability may explain, at least in part, the high frequency of thromboembolic events. Improved risk stratification, utilizing biochemical markers of inflammation and activated coagulation as well as clinical indicators, such as ARDS, may play an important role in the early identification of patients with an increased likelihood of developing symptomatic VTE or arterial thrombosis,” the researchers wrote. “They may benefit from full- or intermediate-intensity antithrombotic therapy rather than prophylactic anticoagulation.”
They point out that this study provides a cross-sectional view of the cardiovascular complications of COVID-19 in a large health care network, consisting of two academic medical centers serving the greater Boston area, several community hospitals, and numerous outpatient care sites.
“The study incorporates a wide scope of clinically meaningful cardiovascular endpoints and utilizes a rigorous process of event adjudication. Although data on patients with COVID-19 in the ICU have been the subject of most reports, our study provides insights into the broad spectrum of all hospitalized and outpatient populations,” the authors noted.
“The high frequency of arterial or venous thromboembolism in hospitalized patients despite routine thromboprophylaxis suggests the need for improved risk stratification and enhanced preventive efforts,” they concluded.
The study is continuing, and the researchers expect to have data on 10,000 patients by the end of winter.
Wait for randomized trials
In an accompanying editorial, Robert McBane, MD, Mayo Clinic, Rochester, Minn., said that these data provide important real-world arterial and venous thrombotic event rates across a large, integrated health care network and an experienced roster of clinician-scientists devoted to thrombosis research.
Noting that whether to interpret these results as alarming or reassuring requires a comparison of expected thromboembolic event rates separate from the pandemic, he pointed out that, while the overall VTE rate among ICU patients was high, the vast majority of these events were attributable to central venous lines, and apart from these, the event rates do not appear inflated relative to prior published incidence rates from the pre–COVID-19 era.
“It is therefore important to resist the urge to overprevent or overtreat patients and expose them to the serious risks of major bleeding,” Dr. McBane wrote, adding that “the systematized approach to delivery of guideline-driven VTE prophylaxis across this large, integrated health network likely contributed to the relatively low rates of serious thrombotic outcomes reported.”
He further noted that, as the majority of VTE events were related to central venous lines in ICU patients, “this underscores the importance of a bundled care approach to central venous line management with daily assessment of the continued necessity of central access.
“A number of important clinical trials aimed at optimizing thromboprophylaxis during hospitalization, following hospital dismissal, and in ambulatory settings are underway. Until available, the lessons of thoughtful anticoagulant prophylaxis and treatment guidelines harvested from years of clinical research appear to apply,” he concluded.
This study was funded, in part, by a research grant from Janssen Pharmaceuticals. Dr. Piazza has received research grant support from EKOS Corporation, Bayer, Bristol-Myers Squibb/Pfizer, Portola Pharmaceuticals, and Janssen Pharmaceuticals; and has received consulting fees from Amgen, Pfizer, Boston Scientific, Agile, and Thrombolex. Dr. McBane reported no relevant disclosures.
A version of this article originally appeared on Medscape.com.
in a new large observational U.S. study.
“Despite very high rate of antithrombotic prophylaxis there were a high rate of thromboembolic events suggesting that we are probably not providing enough thromboprophylaxis,” lead author Gregory Piazza, MD, Brigham and Women’s Hospital, Boston, said in an interview.
“Standard prophylaxis as recommended in the guidelines is a low dose of low-molecular-weight heparin once daily, but these results suggest [patients] probably need higher doses,” he added.
However, Dr. Piazza cautioned that this is an observational study and randomized trials are needed to make changes in treatment strategies. Several such trials are currently underway.
The current study was published online ahead of print in the Nov. 3 issue of the Journal of the American College of Cardiology.
Rates similar to other very sick patients
The study showed that while thromboembolic complications were high, they were not as high as seen in some of the earlier studies from Asia and Europe, Dr. Piazza noted.
“The numbers we were seeing in early reports were so high we couldn’t figure out how that was possible,” he said. “Our study suggests that, in a U.S. population receiving thromboprophylaxis, the rate of thromboembolic complications [are] more in line with what we would expect to see in other very sick patients who end up in ICU.”
He suggested that the very high rates of thromboembolic complications in the early studies from Asia may have been because of the lack of thromboprophylaxis, which is not routine in hospitalized patients there. “Some of the earlier studies also used routine ultrasound and so picked up asymptomatic thrombotic events, which was not the case in our study. So our results are more representative of the U.S. population.”
Dr. Piazza attributed the high rate of thromboembolic complications being reported with COVID-19 to the sheer number of very sick patients being admitted to the hospital.
“We are accustomed to seeing a rare case of thrombosis despite prophylaxis in hospitalized patients, but we are seeing more in COVID patients. This is probably just because we have more critically ill patients,” he said.
“We are seeing an incredible influx of patients to the ICU that we have never experienced before, so the increase in thromboembolic complications is more obvious. In prior years we probably haven’t had enough critically ill patients at any one time to raise the flag about thromboprophylaxis,” he commented.
The study also found a high rate of cardiovascular complications. They are seeing an increase in the risk of MI, which is to be expected in such sick patients, but they also see quite a bit of new atrial fibrillation, myocarditis, and heart failure in patients who don’t always have underlying cardiovascular disease, he said.
“So this virus does appear to have a predilection to causing cardiovascular complications, but this is probably because it is making patients so sick,” Dr. Piazza said. “If flu was this virulent and resulted in such high rates of acute respiratory distress syndrome (ARDS), we would probably see similar cardiovascular complication rates.”
For the current report, the researchers analyzed a retrospective cohort of 1,114 patients with COVID-19 diagnosed through the Mass General Brigham integrated health network. Of these, 170 had been admitted to the ICU, 229 had been hospitalized but not treated in ICU, and 715 were outpatients. In terms of ethnicity, 22% were Hispanic/Latino and 44% were non-White.
Cardiovascular risk factors were common, with 36% of patients having hypertension, 29% hyperlipidemia, and 18% diabetes. Prophylactic anticoagulation was prescribed in 89% of patients with COVID-19 in the intensive care cohort and 85% of those in the hospitalized non–intensive care setting.
Results showed that major arterial or venous thromboembolism (VTE) occurred in 35% of the intensive care cohort, 2.6% of those hospitalized but not treated in ICU, and 0% of outpatients.
Major adverse cardiovascular events occurred in 46% of the intensive care cohort, 6.1% of those hospitalized but non-ICU, and 0% of outpatients.
Symptomatic VTE occurred in 27% of those admitted to ICU, 2.2% of those hospitalized but non-ICU, and 0% of outpatients.
“We found that outpatients had a very low rate of thromboembolic complications, with the vast majority of the risk being in hospitalized patients, especially those in ICU,” Dr. Piazza said.
“These results suggest that we don’t need routine thromboprophylaxis for all outpatients with COVID-19, but there will probably be some patients who need it – those with risk factors for thromboembolism.”
Catheter- and device-associated deep vein thrombosis accounted for 76.9% of the DVTs observed in the study.
“Our finding of high frequency of catheter-associated DVT supports the judicious use of central venous catheters that have been widely implemented, especially in the ICU, to minimize recurrent health care team exposure and facilitate monitoring,” the researchers wrote.
ARDS biggest risk factor
Of all the markers of disease severity, the presence of ARDS had the strongest association with adverse outcomes, including major arterial or VTE, major adverse cardiovascular events, symptomatic VTE, and death.
“The severe inflammatory state associated with ARDS and other complications of COVID-19 and its resultant hypercoagulability may explain, at least in part, the high frequency of thromboembolic events. Improved risk stratification, utilizing biochemical markers of inflammation and activated coagulation as well as clinical indicators, such as ARDS, may play an important role in the early identification of patients with an increased likelihood of developing symptomatic VTE or arterial thrombosis,” the researchers wrote. “They may benefit from full- or intermediate-intensity antithrombotic therapy rather than prophylactic anticoagulation.”
They point out that this study provides a cross-sectional view of the cardiovascular complications of COVID-19 in a large health care network, consisting of two academic medical centers serving the greater Boston area, several community hospitals, and numerous outpatient care sites.
“The study incorporates a wide scope of clinically meaningful cardiovascular endpoints and utilizes a rigorous process of event adjudication. Although data on patients with COVID-19 in the ICU have been the subject of most reports, our study provides insights into the broad spectrum of all hospitalized and outpatient populations,” the authors noted.
“The high frequency of arterial or venous thromboembolism in hospitalized patients despite routine thromboprophylaxis suggests the need for improved risk stratification and enhanced preventive efforts,” they concluded.
The study is continuing, and the researchers expect to have data on 10,000 patients by the end of winter.
Wait for randomized trials
In an accompanying editorial, Robert McBane, MD, Mayo Clinic, Rochester, Minn., said that these data provide important real-world arterial and venous thrombotic event rates across a large, integrated health care network and an experienced roster of clinician-scientists devoted to thrombosis research.
Noting that whether to interpret these results as alarming or reassuring requires a comparison of expected thromboembolic event rates separate from the pandemic, he pointed out that, while the overall VTE rate among ICU patients was high, the vast majority of these events were attributable to central venous lines, and apart from these, the event rates do not appear inflated relative to prior published incidence rates from the pre–COVID-19 era.
“It is therefore important to resist the urge to overprevent or overtreat patients and expose them to the serious risks of major bleeding,” Dr. McBane wrote, adding that “the systematized approach to delivery of guideline-driven VTE prophylaxis across this large, integrated health network likely contributed to the relatively low rates of serious thrombotic outcomes reported.”
He further noted that, as the majority of VTE events were related to central venous lines in ICU patients, “this underscores the importance of a bundled care approach to central venous line management with daily assessment of the continued necessity of central access.
“A number of important clinical trials aimed at optimizing thromboprophylaxis during hospitalization, following hospital dismissal, and in ambulatory settings are underway. Until available, the lessons of thoughtful anticoagulant prophylaxis and treatment guidelines harvested from years of clinical research appear to apply,” he concluded.
This study was funded, in part, by a research grant from Janssen Pharmaceuticals. Dr. Piazza has received research grant support from EKOS Corporation, Bayer, Bristol-Myers Squibb/Pfizer, Portola Pharmaceuticals, and Janssen Pharmaceuticals; and has received consulting fees from Amgen, Pfizer, Boston Scientific, Agile, and Thrombolex. Dr. McBane reported no relevant disclosures.
A version of this article originally appeared on Medscape.com.
in a new large observational U.S. study.
“Despite very high rate of antithrombotic prophylaxis there were a high rate of thromboembolic events suggesting that we are probably not providing enough thromboprophylaxis,” lead author Gregory Piazza, MD, Brigham and Women’s Hospital, Boston, said in an interview.
“Standard prophylaxis as recommended in the guidelines is a low dose of low-molecular-weight heparin once daily, but these results suggest [patients] probably need higher doses,” he added.
However, Dr. Piazza cautioned that this is an observational study and randomized trials are needed to make changes in treatment strategies. Several such trials are currently underway.
The current study was published online ahead of print in the Nov. 3 issue of the Journal of the American College of Cardiology.
Rates similar to other very sick patients
The study showed that while thromboembolic complications were high, they were not as high as seen in some of the earlier studies from Asia and Europe, Dr. Piazza noted.
“The numbers we were seeing in early reports were so high we couldn’t figure out how that was possible,” he said. “Our study suggests that, in a U.S. population receiving thromboprophylaxis, the rate of thromboembolic complications [are] more in line with what we would expect to see in other very sick patients who end up in ICU.”
He suggested that the very high rates of thromboembolic complications in the early studies from Asia may have been because of the lack of thromboprophylaxis, which is not routine in hospitalized patients there. “Some of the earlier studies also used routine ultrasound and so picked up asymptomatic thrombotic events, which was not the case in our study. So our results are more representative of the U.S. population.”
Dr. Piazza attributed the high rate of thromboembolic complications being reported with COVID-19 to the sheer number of very sick patients being admitted to the hospital.
“We are accustomed to seeing a rare case of thrombosis despite prophylaxis in hospitalized patients, but we are seeing more in COVID patients. This is probably just because we have more critically ill patients,” he said.
“We are seeing an incredible influx of patients to the ICU that we have never experienced before, so the increase in thromboembolic complications is more obvious. In prior years we probably haven’t had enough critically ill patients at any one time to raise the flag about thromboprophylaxis,” he commented.
The study also found a high rate of cardiovascular complications. They are seeing an increase in the risk of MI, which is to be expected in such sick patients, but they also see quite a bit of new atrial fibrillation, myocarditis, and heart failure in patients who don’t always have underlying cardiovascular disease, he said.
“So this virus does appear to have a predilection to causing cardiovascular complications, but this is probably because it is making patients so sick,” Dr. Piazza said. “If flu was this virulent and resulted in such high rates of acute respiratory distress syndrome (ARDS), we would probably see similar cardiovascular complication rates.”
For the current report, the researchers analyzed a retrospective cohort of 1,114 patients with COVID-19 diagnosed through the Mass General Brigham integrated health network. Of these, 170 had been admitted to the ICU, 229 had been hospitalized but not treated in ICU, and 715 were outpatients. In terms of ethnicity, 22% were Hispanic/Latino and 44% were non-White.
Cardiovascular risk factors were common, with 36% of patients having hypertension, 29% hyperlipidemia, and 18% diabetes. Prophylactic anticoagulation was prescribed in 89% of patients with COVID-19 in the intensive care cohort and 85% of those in the hospitalized non–intensive care setting.
Results showed that major arterial or venous thromboembolism (VTE) occurred in 35% of the intensive care cohort, 2.6% of those hospitalized but not treated in ICU, and 0% of outpatients.
Major adverse cardiovascular events occurred in 46% of the intensive care cohort, 6.1% of those hospitalized but non-ICU, and 0% of outpatients.
Symptomatic VTE occurred in 27% of those admitted to ICU, 2.2% of those hospitalized but non-ICU, and 0% of outpatients.
“We found that outpatients had a very low rate of thromboembolic complications, with the vast majority of the risk being in hospitalized patients, especially those in ICU,” Dr. Piazza said.
“These results suggest that we don’t need routine thromboprophylaxis for all outpatients with COVID-19, but there will probably be some patients who need it – those with risk factors for thromboembolism.”
Catheter- and device-associated deep vein thrombosis accounted for 76.9% of the DVTs observed in the study.
“Our finding of high frequency of catheter-associated DVT supports the judicious use of central venous catheters that have been widely implemented, especially in the ICU, to minimize recurrent health care team exposure and facilitate monitoring,” the researchers wrote.
ARDS biggest risk factor
Of all the markers of disease severity, the presence of ARDS had the strongest association with adverse outcomes, including major arterial or VTE, major adverse cardiovascular events, symptomatic VTE, and death.
“The severe inflammatory state associated with ARDS and other complications of COVID-19 and its resultant hypercoagulability may explain, at least in part, the high frequency of thromboembolic events. Improved risk stratification, utilizing biochemical markers of inflammation and activated coagulation as well as clinical indicators, such as ARDS, may play an important role in the early identification of patients with an increased likelihood of developing symptomatic VTE or arterial thrombosis,” the researchers wrote. “They may benefit from full- or intermediate-intensity antithrombotic therapy rather than prophylactic anticoagulation.”
They point out that this study provides a cross-sectional view of the cardiovascular complications of COVID-19 in a large health care network, consisting of two academic medical centers serving the greater Boston area, several community hospitals, and numerous outpatient care sites.
“The study incorporates a wide scope of clinically meaningful cardiovascular endpoints and utilizes a rigorous process of event adjudication. Although data on patients with COVID-19 in the ICU have been the subject of most reports, our study provides insights into the broad spectrum of all hospitalized and outpatient populations,” the authors noted.
“The high frequency of arterial or venous thromboembolism in hospitalized patients despite routine thromboprophylaxis suggests the need for improved risk stratification and enhanced preventive efforts,” they concluded.
The study is continuing, and the researchers expect to have data on 10,000 patients by the end of winter.
Wait for randomized trials
In an accompanying editorial, Robert McBane, MD, Mayo Clinic, Rochester, Minn., said that these data provide important real-world arterial and venous thrombotic event rates across a large, integrated health care network and an experienced roster of clinician-scientists devoted to thrombosis research.
Noting that whether to interpret these results as alarming or reassuring requires a comparison of expected thromboembolic event rates separate from the pandemic, he pointed out that, while the overall VTE rate among ICU patients was high, the vast majority of these events were attributable to central venous lines, and apart from these, the event rates do not appear inflated relative to prior published incidence rates from the pre–COVID-19 era.
“It is therefore important to resist the urge to overprevent or overtreat patients and expose them to the serious risks of major bleeding,” Dr. McBane wrote, adding that “the systematized approach to delivery of guideline-driven VTE prophylaxis across this large, integrated health network likely contributed to the relatively low rates of serious thrombotic outcomes reported.”
He further noted that, as the majority of VTE events were related to central venous lines in ICU patients, “this underscores the importance of a bundled care approach to central venous line management with daily assessment of the continued necessity of central access.
“A number of important clinical trials aimed at optimizing thromboprophylaxis during hospitalization, following hospital dismissal, and in ambulatory settings are underway. Until available, the lessons of thoughtful anticoagulant prophylaxis and treatment guidelines harvested from years of clinical research appear to apply,” he concluded.
This study was funded, in part, by a research grant from Janssen Pharmaceuticals. Dr. Piazza has received research grant support from EKOS Corporation, Bayer, Bristol-Myers Squibb/Pfizer, Portola Pharmaceuticals, and Janssen Pharmaceuticals; and has received consulting fees from Amgen, Pfizer, Boston Scientific, Agile, and Thrombolex. Dr. McBane reported no relevant disclosures.
A version of this article originally appeared on Medscape.com.
Health care workers implore OSHA for more oversight on COVID-19 safety
Last spring, when Cliff Willmeng, RN, was working at United Hospital in St. Paul, Minnesota, he’d take off his personal protective equipment (PPE) in the same hallway where children were transported from ambulances to the neighboring Children’s Hospital emergency department. Stretchers would roll across red tape on the floor that designated the area as a “hot zone.” The door from a break room was about 10 feet away.
Willmeng has been a union activist all his life, but he’d never filed a complaint with the Occupational Safety and Health Administration (OSHA) until the COVID-19 pandemic hit.
Concerned about the inadequate space for doffing PPE and other situations in which the spread of SARS-CoV-2 seemed possible, Willmeng and other colleagues filed multiple OSHA complaints with the Minnesota Department of Labor in March and April. Willmeng was also worried about bringing SARS-CoV-2 on his scrubs home to his wife and kids, and he started wearing hospital-supplied scrubs that were meant for doctors and that were washed on site, which was against hospital policy. The hospital fired Willmeng on May 8, citing code of conduct and respectful workplace violations arising from the uniform dispute.
In August, the state agency issued Willmeng’s hospital a $2,100 fine for failure to comply with guidance regarding “respiratory protection” in response to worker complaints over the fact that they were instructed to restaple elastic bands on N95 masks early in the pandemic. In a statement, United Hospital said it contested the citation, and it is in discussions with Minnesota OSHA. “We have and continue to instruct employees not to alter N95 respirators or reuse damaged or soiled N95 respirators,” such as when the straps are broken, the statement says.
Minnesota OSHA has received three times as many emails and phone calls from workers and employers requesting information and assistance during the pandemic, compared with last year, said spokesperson James Honerman. “If Minnesota OSHA is made aware of a workplace safety or health issue, it assesses the situation and determines how best to respond, including conducting a workplace investigation.”
But Willmeng, who has been out of work since he was fired, says that without a receipt or confirmation from OSHA, he has no way of knowing whether there has been any follow-up regarding his complaints. Minnesota OSHA said workers should receive a letter once a case is resolved.
Like Willmeng’s case, none of the more than 10,000 COVID-related complaints the federal OSHA office has received from across the country have resulted in meaningful sanctions. Unions have picketed local OSHA offices and publicized complaints on behalf of their members to protest what they see as a lack of oversight. Legislators have called on US Department of Labor Secretary Eugene Scalia to step up enforcement.
For many health care workers, complaining to OSHA is a last resort after failing to get satisfactory responses from supervisors and appealing to unions for help. But with such minimal oversight from OSHA, some union leaders and legislators say it’s actually more dangerous than not having workplace safety enforcement at all. Lack of directives from the Trump administration has left the agency without the teeth it has cut under previous administrations, and recent changes to the agency’s rules raise questions about whether companies are ever required to report workers’ hospitalizations due to COVID-19.
“It’s so ineffective that it’s more dangerous to workers,” said Kim Cordova, president of United Food and Commercial Workers (UFCW) Local 7, which represents 22,000 health care and other workers in Colorado and Wyoming. “Employers only do what they’re forced to do.” Instead of deterring a multi-billion-dollar company, she said, such low fines signal that a company doesn’t need to worry about COVID-related safety.
“OSHA is doing a lamentably poor job protecting workers during the pandemic,” said James Brudney, JD, a professor at Fordham Law School, in New York, and former chief counsel of the U.S. Senate Subcommittee on Labor. “I’m not alone in saying that the agency has performed so badly.”
Former government officials writing in JAMA were similarly critical: “In the face of the greatest worker health crisis in recent history, OSHA, the lead government agency responsible for worker health and safety, has not fulfilled its responsibilities.”
What could have been
There were early signs that the agency wouldn’t be heavy-handed about COVID-19 safety concerns, Brudney said.
The agency could have issued Emergency Temporary Standards, rules it can put in place during pandemics that address specific short-term concerns. These rules could have required employers to take infection-control measures to protect workers, including mask wearing, providing proper PPE, and screening for COVID-19 symptoms. “That’s what the agency is supposed to do. They’re supposed to respond to an emergency with emergency measures,” Brudney said.
But despite legislative pressure and a court case, Secretary of Labor Eugene Scalia has declined to do so, saying that the agency would instead rely on its regular general duty clause, which is always in place to keep workplaces free from hazards that “cause death or serious physical harm.” The agency invoked the general duty clause for COVID-19–related violations for the first time in September to levy modest fines.
In response to a request for an interview, a Department of Labor spokesperson said that preexisting OSHA requirements apply to workers during the pandemic, including providing PPE for workers and assessing sanitation and cleanliness standards. The agency has issued specific guidance to companies on pandemic preparedness, she said, and that it responds to all complaints. Additionally, she cited whistleblower laws that make it illegal for employers to retaliate against employees for making safety and health complaints.
The federal OSHA office received 10,868 COVID-related complaints from Feb. 1 through Oct. 20, citing issues ranging from failure to provide proper PPE to not informing workers about exposures. As of Oct. 22, a total of 2,349 of the complaints involved healthcare workers. This count doesn’t include the untold number of “informal” complaints handled by state OSHA offices.
In a recent JAMA opinion piece, two former government officials agreed that “the federal government has not fully utilized OSHA’s public safety authority” and called the issuing of an Emergency Temporary Standard that would require employers to develop and implement infection control plans “the most important action the federal government could take” to protect workers.
“Employers are more likely to implement these controls if they are mandated by a government agency that has adequate enforcement tools to ensure compliance,” wrote former Assistant Secretary of Labor David Michaels, PhD, MPH, now at the Milken Institute School of Public Health of the George Washington University, Washington, and Gregory Wagner, MD, a former senior adviser at the National Institute for Occupational Safety and Health at the Centers for Disease Control and Prevention, now at the Harvard T.H. Chan School of Public Health, Boston.
They cited the success of a standard that OSHA issued in 1991 in response to the HIV/AIDS crisis. “The bloodborne pathogens standard has contributed to a substantial decline in health care worker risk for bloodborne diseases like HIV and hepatitis B and C,” they wrote. In a new report for the Century Foundation, the pair offered recommendations to the federal government for controlling the spread of the disease by ramping up OSHA’s role.
OSHA did issue a response plan that requires employers to report in regard to employees who experienced workplace exposures to SARS-CoV-2 and who were hospitalized with COVID-19 or died of the disease within certain time frames, but recent changes to these rules make experts question whether companies are in fact required to report hospitalizations.
In its second revision of guidelines, added to its FAQ page on Sept. 30, the agency said that, in order to be reportable, “an in-patient hospitalization due to COVID-19 must occur within 24 hours of an exposure to SARS-CoV-2 at work” and that the employer must report the hospitalization within 24 hours of learning both that the employee has been hospitalized and that the reason for the hospitalization was a work-related case of COVID-19. Previously, the 24-hour hospitalization window started at the time of diagnosis of the disease, rather than the work-related exposure.
The agency subsequently dropped the first citation it had issued for a COVID-related violation, even though the company, a nursing home, had already agreed to pay $3,904 for reporting employee hospitalizations late.
“It’s a step backwards from an important workplace and public health function that OSHA should be doing,” said Wagner, coauthor of the JAMA opinion piece.
Even without issuing Emergency Temporary Standards, critics say OSHA could have acted much earlier. OSHA issued its first COVID-related federal citation, the one against the nursing home that was dropped, in May for events that occurred in mid-April. The second COVID-related federal citation came in July.
The agency could also charge much more substantial fines for the citations it has issued. If a medical facility was cited for a PPE violation, such as the Minnesota hospital where workers were told to restaple the elastic bands on N95s, the agency could have cited the hospital for one violation per employee. Such fines based on multiple violations could add up to the hundreds of thousands to millions of dollars.
“It would send a signal to the highest-risk employers that these are violations that need to be addressed immediately,” Brudney said.
Many of the 22 state OSHA offices appear to be more responsive to COVID-related complaints than the federal agency, creating a system in which health care workers have substantially different rights from one state to the next. The governor of California, for example, recently authorized California’s OSHA division to consider COVID-19 an imminent hazard, to prohibit workers from entering areas where the hazard exists, and to require employers to disclose exposures. The state also recently issued large fines for COVID safety issues: $222,075 to frozen food manufacturer Overhill Farms and $214,080 to employment agency Jobsource North America.
Elsewhere, state laws such as New Jersey’s Conscientious Employee Protection Act give workers the right to refuse to work in unsafe situations, Brudney said. “A lot more action is going on at the state level because so little is being done at the federal level,” he said. “Some of it is governors committed to protecting essential workers and their families.”
Unions call for sanctions
Unions are both decrying the lack of enforcement thus far and seeking more oversight going forward.
In August, the National Nurses’ United (NNU) union filed a complaint to implore OSHA to investigate the country’s biggest hospital systems, HCA Healthcare, which operates 184 hospitals and about 2,000 other care sites in 21 states and the United Kingdom. The union describes how, throughout HCA hospitals, there is an environment conducive to the spread of coronavirus. Nurses share space and equipment, such as computers, desks, phones, bathrooms, and break rooms, where staff take off masks to eat and drink. The complaint also describes how there is resistance to testing nurses and a lack of communication about infections among colleagues.
“When they have total disregard for safety, they should be punished to the utmost,” said Markowitz, noting that HCA Healthcare is worth $40 billion. “They can penalize them, but if it’s unsafe conditions for RNs and healthcare workers, we know it’s unsafe for the patients. There needs to be drastic measures to prevent hospital corporations from behaving that way.”
In a statement, HCA spokesman Harlow Sumerford said the company has followed CDC guidance for protecting frontline caregivers. “We’re proud of our response and the significant resources we’ve deployed to help protect our colleagues. Meanwhile, the NNU has chosen to use this pandemic as an opportunity to gain publicity by attacking hospitals across the country,” Sumerford said.
Members of the union recently protested in front of the federal OSHA offices in Denver.
After several months, OSHA finally penalized a meat packing plant where eight workers (six union members) had died of COVID-19 last spring. But the amount – $15,615 – was so low that Cordova worries it will actually have a worse impact than no fine.
“It’s more dangerous to workers because now employers know [they won’t be punished meaningfully],” she said. “During the pandemic, OSHA has been absolutely absent.”
Thus, the recent picketing outside the offices in Denver. But, Cordova noted, it’s unlikely OSHA employees saw them. Their own offices were deemed too risky to stay open during the pandemic. They were vacant.
A version of this article originally appeared on Medscape.com.
Last spring, when Cliff Willmeng, RN, was working at United Hospital in St. Paul, Minnesota, he’d take off his personal protective equipment (PPE) in the same hallway where children were transported from ambulances to the neighboring Children’s Hospital emergency department. Stretchers would roll across red tape on the floor that designated the area as a “hot zone.” The door from a break room was about 10 feet away.
Willmeng has been a union activist all his life, but he’d never filed a complaint with the Occupational Safety and Health Administration (OSHA) until the COVID-19 pandemic hit.
Concerned about the inadequate space for doffing PPE and other situations in which the spread of SARS-CoV-2 seemed possible, Willmeng and other colleagues filed multiple OSHA complaints with the Minnesota Department of Labor in March and April. Willmeng was also worried about bringing SARS-CoV-2 on his scrubs home to his wife and kids, and he started wearing hospital-supplied scrubs that were meant for doctors and that were washed on site, which was against hospital policy. The hospital fired Willmeng on May 8, citing code of conduct and respectful workplace violations arising from the uniform dispute.
In August, the state agency issued Willmeng’s hospital a $2,100 fine for failure to comply with guidance regarding “respiratory protection” in response to worker complaints over the fact that they were instructed to restaple elastic bands on N95 masks early in the pandemic. In a statement, United Hospital said it contested the citation, and it is in discussions with Minnesota OSHA. “We have and continue to instruct employees not to alter N95 respirators or reuse damaged or soiled N95 respirators,” such as when the straps are broken, the statement says.
Minnesota OSHA has received three times as many emails and phone calls from workers and employers requesting information and assistance during the pandemic, compared with last year, said spokesperson James Honerman. “If Minnesota OSHA is made aware of a workplace safety or health issue, it assesses the situation and determines how best to respond, including conducting a workplace investigation.”
But Willmeng, who has been out of work since he was fired, says that without a receipt or confirmation from OSHA, he has no way of knowing whether there has been any follow-up regarding his complaints. Minnesota OSHA said workers should receive a letter once a case is resolved.
Like Willmeng’s case, none of the more than 10,000 COVID-related complaints the federal OSHA office has received from across the country have resulted in meaningful sanctions. Unions have picketed local OSHA offices and publicized complaints on behalf of their members to protest what they see as a lack of oversight. Legislators have called on US Department of Labor Secretary Eugene Scalia to step up enforcement.
For many health care workers, complaining to OSHA is a last resort after failing to get satisfactory responses from supervisors and appealing to unions for help. But with such minimal oversight from OSHA, some union leaders and legislators say it’s actually more dangerous than not having workplace safety enforcement at all. Lack of directives from the Trump administration has left the agency without the teeth it has cut under previous administrations, and recent changes to the agency’s rules raise questions about whether companies are ever required to report workers’ hospitalizations due to COVID-19.
“It’s so ineffective that it’s more dangerous to workers,” said Kim Cordova, president of United Food and Commercial Workers (UFCW) Local 7, which represents 22,000 health care and other workers in Colorado and Wyoming. “Employers only do what they’re forced to do.” Instead of deterring a multi-billion-dollar company, she said, such low fines signal that a company doesn’t need to worry about COVID-related safety.
“OSHA is doing a lamentably poor job protecting workers during the pandemic,” said James Brudney, JD, a professor at Fordham Law School, in New York, and former chief counsel of the U.S. Senate Subcommittee on Labor. “I’m not alone in saying that the agency has performed so badly.”
Former government officials writing in JAMA were similarly critical: “In the face of the greatest worker health crisis in recent history, OSHA, the lead government agency responsible for worker health and safety, has not fulfilled its responsibilities.”
What could have been
There were early signs that the agency wouldn’t be heavy-handed about COVID-19 safety concerns, Brudney said.
The agency could have issued Emergency Temporary Standards, rules it can put in place during pandemics that address specific short-term concerns. These rules could have required employers to take infection-control measures to protect workers, including mask wearing, providing proper PPE, and screening for COVID-19 symptoms. “That’s what the agency is supposed to do. They’re supposed to respond to an emergency with emergency measures,” Brudney said.
But despite legislative pressure and a court case, Secretary of Labor Eugene Scalia has declined to do so, saying that the agency would instead rely on its regular general duty clause, which is always in place to keep workplaces free from hazards that “cause death or serious physical harm.” The agency invoked the general duty clause for COVID-19–related violations for the first time in September to levy modest fines.
In response to a request for an interview, a Department of Labor spokesperson said that preexisting OSHA requirements apply to workers during the pandemic, including providing PPE for workers and assessing sanitation and cleanliness standards. The agency has issued specific guidance to companies on pandemic preparedness, she said, and that it responds to all complaints. Additionally, she cited whistleblower laws that make it illegal for employers to retaliate against employees for making safety and health complaints.
The federal OSHA office received 10,868 COVID-related complaints from Feb. 1 through Oct. 20, citing issues ranging from failure to provide proper PPE to not informing workers about exposures. As of Oct. 22, a total of 2,349 of the complaints involved healthcare workers. This count doesn’t include the untold number of “informal” complaints handled by state OSHA offices.
In a recent JAMA opinion piece, two former government officials agreed that “the federal government has not fully utilized OSHA’s public safety authority” and called the issuing of an Emergency Temporary Standard that would require employers to develop and implement infection control plans “the most important action the federal government could take” to protect workers.
“Employers are more likely to implement these controls if they are mandated by a government agency that has adequate enforcement tools to ensure compliance,” wrote former Assistant Secretary of Labor David Michaels, PhD, MPH, now at the Milken Institute School of Public Health of the George Washington University, Washington, and Gregory Wagner, MD, a former senior adviser at the National Institute for Occupational Safety and Health at the Centers for Disease Control and Prevention, now at the Harvard T.H. Chan School of Public Health, Boston.
They cited the success of a standard that OSHA issued in 1991 in response to the HIV/AIDS crisis. “The bloodborne pathogens standard has contributed to a substantial decline in health care worker risk for bloodborne diseases like HIV and hepatitis B and C,” they wrote. In a new report for the Century Foundation, the pair offered recommendations to the federal government for controlling the spread of the disease by ramping up OSHA’s role.
OSHA did issue a response plan that requires employers to report in regard to employees who experienced workplace exposures to SARS-CoV-2 and who were hospitalized with COVID-19 or died of the disease within certain time frames, but recent changes to these rules make experts question whether companies are in fact required to report hospitalizations.
In its second revision of guidelines, added to its FAQ page on Sept. 30, the agency said that, in order to be reportable, “an in-patient hospitalization due to COVID-19 must occur within 24 hours of an exposure to SARS-CoV-2 at work” and that the employer must report the hospitalization within 24 hours of learning both that the employee has been hospitalized and that the reason for the hospitalization was a work-related case of COVID-19. Previously, the 24-hour hospitalization window started at the time of diagnosis of the disease, rather than the work-related exposure.
The agency subsequently dropped the first citation it had issued for a COVID-related violation, even though the company, a nursing home, had already agreed to pay $3,904 for reporting employee hospitalizations late.
“It’s a step backwards from an important workplace and public health function that OSHA should be doing,” said Wagner, coauthor of the JAMA opinion piece.
Even without issuing Emergency Temporary Standards, critics say OSHA could have acted much earlier. OSHA issued its first COVID-related federal citation, the one against the nursing home that was dropped, in May for events that occurred in mid-April. The second COVID-related federal citation came in July.
The agency could also charge much more substantial fines for the citations it has issued. If a medical facility was cited for a PPE violation, such as the Minnesota hospital where workers were told to restaple the elastic bands on N95s, the agency could have cited the hospital for one violation per employee. Such fines based on multiple violations could add up to the hundreds of thousands to millions of dollars.
“It would send a signal to the highest-risk employers that these are violations that need to be addressed immediately,” Brudney said.
Many of the 22 state OSHA offices appear to be more responsive to COVID-related complaints than the federal agency, creating a system in which health care workers have substantially different rights from one state to the next. The governor of California, for example, recently authorized California’s OSHA division to consider COVID-19 an imminent hazard, to prohibit workers from entering areas where the hazard exists, and to require employers to disclose exposures. The state also recently issued large fines for COVID safety issues: $222,075 to frozen food manufacturer Overhill Farms and $214,080 to employment agency Jobsource North America.
Elsewhere, state laws such as New Jersey’s Conscientious Employee Protection Act give workers the right to refuse to work in unsafe situations, Brudney said. “A lot more action is going on at the state level because so little is being done at the federal level,” he said. “Some of it is governors committed to protecting essential workers and their families.”
Unions call for sanctions
Unions are both decrying the lack of enforcement thus far and seeking more oversight going forward.
In August, the National Nurses’ United (NNU) union filed a complaint to implore OSHA to investigate the country’s biggest hospital systems, HCA Healthcare, which operates 184 hospitals and about 2,000 other care sites in 21 states and the United Kingdom. The union describes how, throughout HCA hospitals, there is an environment conducive to the spread of coronavirus. Nurses share space and equipment, such as computers, desks, phones, bathrooms, and break rooms, where staff take off masks to eat and drink. The complaint also describes how there is resistance to testing nurses and a lack of communication about infections among colleagues.
“When they have total disregard for safety, they should be punished to the utmost,” said Markowitz, noting that HCA Healthcare is worth $40 billion. “They can penalize them, but if it’s unsafe conditions for RNs and healthcare workers, we know it’s unsafe for the patients. There needs to be drastic measures to prevent hospital corporations from behaving that way.”
In a statement, HCA spokesman Harlow Sumerford said the company has followed CDC guidance for protecting frontline caregivers. “We’re proud of our response and the significant resources we’ve deployed to help protect our colleagues. Meanwhile, the NNU has chosen to use this pandemic as an opportunity to gain publicity by attacking hospitals across the country,” Sumerford said.
Members of the union recently protested in front of the federal OSHA offices in Denver.
After several months, OSHA finally penalized a meat packing plant where eight workers (six union members) had died of COVID-19 last spring. But the amount – $15,615 – was so low that Cordova worries it will actually have a worse impact than no fine.
“It’s more dangerous to workers because now employers know [they won’t be punished meaningfully],” she said. “During the pandemic, OSHA has been absolutely absent.”
Thus, the recent picketing outside the offices in Denver. But, Cordova noted, it’s unlikely OSHA employees saw them. Their own offices were deemed too risky to stay open during the pandemic. They were vacant.
A version of this article originally appeared on Medscape.com.
Last spring, when Cliff Willmeng, RN, was working at United Hospital in St. Paul, Minnesota, he’d take off his personal protective equipment (PPE) in the same hallway where children were transported from ambulances to the neighboring Children’s Hospital emergency department. Stretchers would roll across red tape on the floor that designated the area as a “hot zone.” The door from a break room was about 10 feet away.
Willmeng has been a union activist all his life, but he’d never filed a complaint with the Occupational Safety and Health Administration (OSHA) until the COVID-19 pandemic hit.
Concerned about the inadequate space for doffing PPE and other situations in which the spread of SARS-CoV-2 seemed possible, Willmeng and other colleagues filed multiple OSHA complaints with the Minnesota Department of Labor in March and April. Willmeng was also worried about bringing SARS-CoV-2 on his scrubs home to his wife and kids, and he started wearing hospital-supplied scrubs that were meant for doctors and that were washed on site, which was against hospital policy. The hospital fired Willmeng on May 8, citing code of conduct and respectful workplace violations arising from the uniform dispute.
In August, the state agency issued Willmeng’s hospital a $2,100 fine for failure to comply with guidance regarding “respiratory protection” in response to worker complaints over the fact that they were instructed to restaple elastic bands on N95 masks early in the pandemic. In a statement, United Hospital said it contested the citation, and it is in discussions with Minnesota OSHA. “We have and continue to instruct employees not to alter N95 respirators or reuse damaged or soiled N95 respirators,” such as when the straps are broken, the statement says.
Minnesota OSHA has received three times as many emails and phone calls from workers and employers requesting information and assistance during the pandemic, compared with last year, said spokesperson James Honerman. “If Minnesota OSHA is made aware of a workplace safety or health issue, it assesses the situation and determines how best to respond, including conducting a workplace investigation.”
But Willmeng, who has been out of work since he was fired, says that without a receipt or confirmation from OSHA, he has no way of knowing whether there has been any follow-up regarding his complaints. Minnesota OSHA said workers should receive a letter once a case is resolved.
Like Willmeng’s case, none of the more than 10,000 COVID-related complaints the federal OSHA office has received from across the country have resulted in meaningful sanctions. Unions have picketed local OSHA offices and publicized complaints on behalf of their members to protest what they see as a lack of oversight. Legislators have called on US Department of Labor Secretary Eugene Scalia to step up enforcement.
For many health care workers, complaining to OSHA is a last resort after failing to get satisfactory responses from supervisors and appealing to unions for help. But with such minimal oversight from OSHA, some union leaders and legislators say it’s actually more dangerous than not having workplace safety enforcement at all. Lack of directives from the Trump administration has left the agency without the teeth it has cut under previous administrations, and recent changes to the agency’s rules raise questions about whether companies are ever required to report workers’ hospitalizations due to COVID-19.
“It’s so ineffective that it’s more dangerous to workers,” said Kim Cordova, president of United Food and Commercial Workers (UFCW) Local 7, which represents 22,000 health care and other workers in Colorado and Wyoming. “Employers only do what they’re forced to do.” Instead of deterring a multi-billion-dollar company, she said, such low fines signal that a company doesn’t need to worry about COVID-related safety.
“OSHA is doing a lamentably poor job protecting workers during the pandemic,” said James Brudney, JD, a professor at Fordham Law School, in New York, and former chief counsel of the U.S. Senate Subcommittee on Labor. “I’m not alone in saying that the agency has performed so badly.”
Former government officials writing in JAMA were similarly critical: “In the face of the greatest worker health crisis in recent history, OSHA, the lead government agency responsible for worker health and safety, has not fulfilled its responsibilities.”
What could have been
There were early signs that the agency wouldn’t be heavy-handed about COVID-19 safety concerns, Brudney said.
The agency could have issued Emergency Temporary Standards, rules it can put in place during pandemics that address specific short-term concerns. These rules could have required employers to take infection-control measures to protect workers, including mask wearing, providing proper PPE, and screening for COVID-19 symptoms. “That’s what the agency is supposed to do. They’re supposed to respond to an emergency with emergency measures,” Brudney said.
But despite legislative pressure and a court case, Secretary of Labor Eugene Scalia has declined to do so, saying that the agency would instead rely on its regular general duty clause, which is always in place to keep workplaces free from hazards that “cause death or serious physical harm.” The agency invoked the general duty clause for COVID-19–related violations for the first time in September to levy modest fines.
In response to a request for an interview, a Department of Labor spokesperson said that preexisting OSHA requirements apply to workers during the pandemic, including providing PPE for workers and assessing sanitation and cleanliness standards. The agency has issued specific guidance to companies on pandemic preparedness, she said, and that it responds to all complaints. Additionally, she cited whistleblower laws that make it illegal for employers to retaliate against employees for making safety and health complaints.
The federal OSHA office received 10,868 COVID-related complaints from Feb. 1 through Oct. 20, citing issues ranging from failure to provide proper PPE to not informing workers about exposures. As of Oct. 22, a total of 2,349 of the complaints involved healthcare workers. This count doesn’t include the untold number of “informal” complaints handled by state OSHA offices.
In a recent JAMA opinion piece, two former government officials agreed that “the federal government has not fully utilized OSHA’s public safety authority” and called the issuing of an Emergency Temporary Standard that would require employers to develop and implement infection control plans “the most important action the federal government could take” to protect workers.
“Employers are more likely to implement these controls if they are mandated by a government agency that has adequate enforcement tools to ensure compliance,” wrote former Assistant Secretary of Labor David Michaels, PhD, MPH, now at the Milken Institute School of Public Health of the George Washington University, Washington, and Gregory Wagner, MD, a former senior adviser at the National Institute for Occupational Safety and Health at the Centers for Disease Control and Prevention, now at the Harvard T.H. Chan School of Public Health, Boston.
They cited the success of a standard that OSHA issued in 1991 in response to the HIV/AIDS crisis. “The bloodborne pathogens standard has contributed to a substantial decline in health care worker risk for bloodborne diseases like HIV and hepatitis B and C,” they wrote. In a new report for the Century Foundation, the pair offered recommendations to the federal government for controlling the spread of the disease by ramping up OSHA’s role.
OSHA did issue a response plan that requires employers to report in regard to employees who experienced workplace exposures to SARS-CoV-2 and who were hospitalized with COVID-19 or died of the disease within certain time frames, but recent changes to these rules make experts question whether companies are in fact required to report hospitalizations.
In its second revision of guidelines, added to its FAQ page on Sept. 30, the agency said that, in order to be reportable, “an in-patient hospitalization due to COVID-19 must occur within 24 hours of an exposure to SARS-CoV-2 at work” and that the employer must report the hospitalization within 24 hours of learning both that the employee has been hospitalized and that the reason for the hospitalization was a work-related case of COVID-19. Previously, the 24-hour hospitalization window started at the time of diagnosis of the disease, rather than the work-related exposure.
The agency subsequently dropped the first citation it had issued for a COVID-related violation, even though the company, a nursing home, had already agreed to pay $3,904 for reporting employee hospitalizations late.
“It’s a step backwards from an important workplace and public health function that OSHA should be doing,” said Wagner, coauthor of the JAMA opinion piece.
Even without issuing Emergency Temporary Standards, critics say OSHA could have acted much earlier. OSHA issued its first COVID-related federal citation, the one against the nursing home that was dropped, in May for events that occurred in mid-April. The second COVID-related federal citation came in July.
The agency could also charge much more substantial fines for the citations it has issued. If a medical facility was cited for a PPE violation, such as the Minnesota hospital where workers were told to restaple the elastic bands on N95s, the agency could have cited the hospital for one violation per employee. Such fines based on multiple violations could add up to the hundreds of thousands to millions of dollars.
“It would send a signal to the highest-risk employers that these are violations that need to be addressed immediately,” Brudney said.
Many of the 22 state OSHA offices appear to be more responsive to COVID-related complaints than the federal agency, creating a system in which health care workers have substantially different rights from one state to the next. The governor of California, for example, recently authorized California’s OSHA division to consider COVID-19 an imminent hazard, to prohibit workers from entering areas where the hazard exists, and to require employers to disclose exposures. The state also recently issued large fines for COVID safety issues: $222,075 to frozen food manufacturer Overhill Farms and $214,080 to employment agency Jobsource North America.
Elsewhere, state laws such as New Jersey’s Conscientious Employee Protection Act give workers the right to refuse to work in unsafe situations, Brudney said. “A lot more action is going on at the state level because so little is being done at the federal level,” he said. “Some of it is governors committed to protecting essential workers and their families.”
Unions call for sanctions
Unions are both decrying the lack of enforcement thus far and seeking more oversight going forward.
In August, the National Nurses’ United (NNU) union filed a complaint to implore OSHA to investigate the country’s biggest hospital systems, HCA Healthcare, which operates 184 hospitals and about 2,000 other care sites in 21 states and the United Kingdom. The union describes how, throughout HCA hospitals, there is an environment conducive to the spread of coronavirus. Nurses share space and equipment, such as computers, desks, phones, bathrooms, and break rooms, where staff take off masks to eat and drink. The complaint also describes how there is resistance to testing nurses and a lack of communication about infections among colleagues.
“When they have total disregard for safety, they should be punished to the utmost,” said Markowitz, noting that HCA Healthcare is worth $40 billion. “They can penalize them, but if it’s unsafe conditions for RNs and healthcare workers, we know it’s unsafe for the patients. There needs to be drastic measures to prevent hospital corporations from behaving that way.”
In a statement, HCA spokesman Harlow Sumerford said the company has followed CDC guidance for protecting frontline caregivers. “We’re proud of our response and the significant resources we’ve deployed to help protect our colleagues. Meanwhile, the NNU has chosen to use this pandemic as an opportunity to gain publicity by attacking hospitals across the country,” Sumerford said.
Members of the union recently protested in front of the federal OSHA offices in Denver.
After several months, OSHA finally penalized a meat packing plant where eight workers (six union members) had died of COVID-19 last spring. But the amount – $15,615 – was so low that Cordova worries it will actually have a worse impact than no fine.
“It’s more dangerous to workers because now employers know [they won’t be punished meaningfully],” she said. “During the pandemic, OSHA has been absolutely absent.”
Thus, the recent picketing outside the offices in Denver. But, Cordova noted, it’s unlikely OSHA employees saw them. Their own offices were deemed too risky to stay open during the pandemic. They were vacant.
A version of this article originally appeared on Medscape.com.