User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
Low-dose naltrexone falls short for fibromyalgia
TOPLINE:
Women with fibromyalgia who received low-dose naltrexone showed no significant improvement in pain at 12 weeks, compared with those who received placebo in a randomized trial.
METHODOLOGY:
- The researchers randomly assigned 99 women with fibromyalgia at a single center in Denmark to a daily dose of 6 mg of naltrexone or a placebo for 12 weeks.
- The primary outcome was within-group change in pain intensity from baseline to 12 weeks, measured using an 11-point numeric rating scale (NRS) and the Fibromyalgia Impact Questionnaire-Revised (FIQR); outcomes were measured at 4, 8, and 12 weeks.
- Secondary outcomes included the global impact of FIQR total scores, as well as FIQR scores for tenderness, fatigue, , , anxiety, memory, stiffness, and physical function.
TAKEAWAY:
- The patients ranged in age from 18 to 64 years, with a mean age of 50.6 years, and all but one was White.
- At 12 weeks, the mean change in pain intensity was greater in the naltrexone group compared with the placebo group (−1.3 points vs −0.9 points, respectively), but the difference was not statistically significant.
- Of the secondary outcomes, only memory problems related to fibromyalgia showed significant improvement with naltrexone compared with placebo (−0.93 vs −0.30; P = .004), although the significance was lost after adjusting for multiplicity.
- Adverse events were infrequent and similar between the groups; four of 49 patients in the naltrexone group and three of 50 in the placebo group discontinued their assigned treatments because of side effects, and no safety concerns appeared related to the 6-mg dose.
IN PRACTICE:
“At this time we recommend that off-label treatment of patients who have responded to low-dose naltrexone should not be terminated, but we recommend against initiating low-dose naltrexone for low-dose naltrexone-naive patients with fibromyalgia pending the results of additional adequately powered studies with distinctive inflammatory and autoantibody patient profiles,” Winfried Häuser, MD, of the Center for Pain Medicine and Mental Health, Saarbrücken, Germany, and Mary-Ann Fitzcharles, MD, of McGill University Health Centre, Montreal, Canada, wrote in an accompanying editorial.
SOURCE:
The lead author of the study was Karin Due Bruun, MD, of Odense University Hospital, Denmark. The full study and accompanying editorial were published online in The Lancet Rheumatology.
LIMITATIONS:
The study was only powered to detect a difference of 1.0 NRS points for pain intensity; other limitations include the homogenous population that prevents generalizability to other groups and the relatively short follow-up period.
DISCLOSURES:
The study was supported by the Danish Rheumatism Association, Odense University Hospital, Danielsen’s Foundation, and the Oak Foundation. The researchers had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
TOPLINE:
Women with fibromyalgia who received low-dose naltrexone showed no significant improvement in pain at 12 weeks, compared with those who received placebo in a randomized trial.
METHODOLOGY:
- The researchers randomly assigned 99 women with fibromyalgia at a single center in Denmark to a daily dose of 6 mg of naltrexone or a placebo for 12 weeks.
- The primary outcome was within-group change in pain intensity from baseline to 12 weeks, measured using an 11-point numeric rating scale (NRS) and the Fibromyalgia Impact Questionnaire-Revised (FIQR); outcomes were measured at 4, 8, and 12 weeks.
- Secondary outcomes included the global impact of FIQR total scores, as well as FIQR scores for tenderness, fatigue, , , anxiety, memory, stiffness, and physical function.
TAKEAWAY:
- The patients ranged in age from 18 to 64 years, with a mean age of 50.6 years, and all but one was White.
- At 12 weeks, the mean change in pain intensity was greater in the naltrexone group compared with the placebo group (−1.3 points vs −0.9 points, respectively), but the difference was not statistically significant.
- Of the secondary outcomes, only memory problems related to fibromyalgia showed significant improvement with naltrexone compared with placebo (−0.93 vs −0.30; P = .004), although the significance was lost after adjusting for multiplicity.
- Adverse events were infrequent and similar between the groups; four of 49 patients in the naltrexone group and three of 50 in the placebo group discontinued their assigned treatments because of side effects, and no safety concerns appeared related to the 6-mg dose.
IN PRACTICE:
“At this time we recommend that off-label treatment of patients who have responded to low-dose naltrexone should not be terminated, but we recommend against initiating low-dose naltrexone for low-dose naltrexone-naive patients with fibromyalgia pending the results of additional adequately powered studies with distinctive inflammatory and autoantibody patient profiles,” Winfried Häuser, MD, of the Center for Pain Medicine and Mental Health, Saarbrücken, Germany, and Mary-Ann Fitzcharles, MD, of McGill University Health Centre, Montreal, Canada, wrote in an accompanying editorial.
SOURCE:
The lead author of the study was Karin Due Bruun, MD, of Odense University Hospital, Denmark. The full study and accompanying editorial were published online in The Lancet Rheumatology.
LIMITATIONS:
The study was only powered to detect a difference of 1.0 NRS points for pain intensity; other limitations include the homogenous population that prevents generalizability to other groups and the relatively short follow-up period.
DISCLOSURES:
The study was supported by the Danish Rheumatism Association, Odense University Hospital, Danielsen’s Foundation, and the Oak Foundation. The researchers had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
TOPLINE:
Women with fibromyalgia who received low-dose naltrexone showed no significant improvement in pain at 12 weeks, compared with those who received placebo in a randomized trial.
METHODOLOGY:
- The researchers randomly assigned 99 women with fibromyalgia at a single center in Denmark to a daily dose of 6 mg of naltrexone or a placebo for 12 weeks.
- The primary outcome was within-group change in pain intensity from baseline to 12 weeks, measured using an 11-point numeric rating scale (NRS) and the Fibromyalgia Impact Questionnaire-Revised (FIQR); outcomes were measured at 4, 8, and 12 weeks.
- Secondary outcomes included the global impact of FIQR total scores, as well as FIQR scores for tenderness, fatigue, , , anxiety, memory, stiffness, and physical function.
TAKEAWAY:
- The patients ranged in age from 18 to 64 years, with a mean age of 50.6 years, and all but one was White.
- At 12 weeks, the mean change in pain intensity was greater in the naltrexone group compared with the placebo group (−1.3 points vs −0.9 points, respectively), but the difference was not statistically significant.
- Of the secondary outcomes, only memory problems related to fibromyalgia showed significant improvement with naltrexone compared with placebo (−0.93 vs −0.30; P = .004), although the significance was lost after adjusting for multiplicity.
- Adverse events were infrequent and similar between the groups; four of 49 patients in the naltrexone group and three of 50 in the placebo group discontinued their assigned treatments because of side effects, and no safety concerns appeared related to the 6-mg dose.
IN PRACTICE:
“At this time we recommend that off-label treatment of patients who have responded to low-dose naltrexone should not be terminated, but we recommend against initiating low-dose naltrexone for low-dose naltrexone-naive patients with fibromyalgia pending the results of additional adequately powered studies with distinctive inflammatory and autoantibody patient profiles,” Winfried Häuser, MD, of the Center for Pain Medicine and Mental Health, Saarbrücken, Germany, and Mary-Ann Fitzcharles, MD, of McGill University Health Centre, Montreal, Canada, wrote in an accompanying editorial.
SOURCE:
The lead author of the study was Karin Due Bruun, MD, of Odense University Hospital, Denmark. The full study and accompanying editorial were published online in The Lancet Rheumatology.
LIMITATIONS:
The study was only powered to detect a difference of 1.0 NRS points for pain intensity; other limitations include the homogenous population that prevents generalizability to other groups and the relatively short follow-up period.
DISCLOSURES:
The study was supported by the Danish Rheumatism Association, Odense University Hospital, Danielsen’s Foundation, and the Oak Foundation. The researchers had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
Specific personality traits may influence dementia risk
TOPLINE:
, whereas those who score highly for neuroticism and have a negative outlook may be at increased risk, new research suggests.
METHODOLOGY:
- Researchers examined the link between the “big five” personality traits (conscientiousness, extraversion, openness to experience, neuroticism, and agreeableness) and subjective well-being (positive and negative affect and life satisfaction) and clinical symptoms of dementia (cognitive test performance) and neuropathology at autopsy.
- Data for the meta-analysis came from eight longitudinal studies with 44,531 adults (aged 49-81 years at baseline; 26%-61% women) followed for up to 21 years, during which 1703 incident cases of dementia occurred.
- Bayesian multilevel models tested whether personality traits and subjective well-being differentially predicted neuropsychological and neuropathologic characteristics of dementia.
TAKEAWAY:
- High neuroticism, negative affect, and low conscientiousness were risk factors for dementia, whereas conscientiousness, extraversion, and positive affect were protective.
- Across all analyses, there was directional consistency in estimates across samples, which is noteworthy given between-study differences in sociodemographic and design characteristics.
- No consistent associations were found between psychological factors and neuropathology.
- However, individuals higher in conscientiousness who did not receive a clinical diagnosis tended to have a lower Braak stage at autopsy, suggesting the possibility that conscientiousness is related to cognitive resilience.
IN PRACTICE:
“These results replicate and extend evidence that personality traits may assist in early identification and dementia-care planning strategies, as well as risk stratification for dementia diagnosis. Moreover, our findings provide further support for recommendations to incorporate psychological trait measures into clinical screening or diagnosis criteria,” the authors write. SOURCE:
The study, with first author Emorie Beck, PhD, Department of Psychology, University of California, Davis, was published online on November 29, 2023, in Alzheimer’s & Dementia.
LIMITATIONS:
Access to autopsy data was limited. The findings may not generalize across racial groups. The analysis did not examine dynamic associations between changing personality and cognition and neuropathology over time.
DISCLOSURES:
The study was supported by grants from the National Institute on Aging. The authors have declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
, whereas those who score highly for neuroticism and have a negative outlook may be at increased risk, new research suggests.
METHODOLOGY:
- Researchers examined the link between the “big five” personality traits (conscientiousness, extraversion, openness to experience, neuroticism, and agreeableness) and subjective well-being (positive and negative affect and life satisfaction) and clinical symptoms of dementia (cognitive test performance) and neuropathology at autopsy.
- Data for the meta-analysis came from eight longitudinal studies with 44,531 adults (aged 49-81 years at baseline; 26%-61% women) followed for up to 21 years, during which 1703 incident cases of dementia occurred.
- Bayesian multilevel models tested whether personality traits and subjective well-being differentially predicted neuropsychological and neuropathologic characteristics of dementia.
TAKEAWAY:
- High neuroticism, negative affect, and low conscientiousness were risk factors for dementia, whereas conscientiousness, extraversion, and positive affect were protective.
- Across all analyses, there was directional consistency in estimates across samples, which is noteworthy given between-study differences in sociodemographic and design characteristics.
- No consistent associations were found between psychological factors and neuropathology.
- However, individuals higher in conscientiousness who did not receive a clinical diagnosis tended to have a lower Braak stage at autopsy, suggesting the possibility that conscientiousness is related to cognitive resilience.
IN PRACTICE:
“These results replicate and extend evidence that personality traits may assist in early identification and dementia-care planning strategies, as well as risk stratification for dementia diagnosis. Moreover, our findings provide further support for recommendations to incorporate psychological trait measures into clinical screening or diagnosis criteria,” the authors write. SOURCE:
The study, with first author Emorie Beck, PhD, Department of Psychology, University of California, Davis, was published online on November 29, 2023, in Alzheimer’s & Dementia.
LIMITATIONS:
Access to autopsy data was limited. The findings may not generalize across racial groups. The analysis did not examine dynamic associations between changing personality and cognition and neuropathology over time.
DISCLOSURES:
The study was supported by grants from the National Institute on Aging. The authors have declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
, whereas those who score highly for neuroticism and have a negative outlook may be at increased risk, new research suggests.
METHODOLOGY:
- Researchers examined the link between the “big five” personality traits (conscientiousness, extraversion, openness to experience, neuroticism, and agreeableness) and subjective well-being (positive and negative affect and life satisfaction) and clinical symptoms of dementia (cognitive test performance) and neuropathology at autopsy.
- Data for the meta-analysis came from eight longitudinal studies with 44,531 adults (aged 49-81 years at baseline; 26%-61% women) followed for up to 21 years, during which 1703 incident cases of dementia occurred.
- Bayesian multilevel models tested whether personality traits and subjective well-being differentially predicted neuropsychological and neuropathologic characteristics of dementia.
TAKEAWAY:
- High neuroticism, negative affect, and low conscientiousness were risk factors for dementia, whereas conscientiousness, extraversion, and positive affect were protective.
- Across all analyses, there was directional consistency in estimates across samples, which is noteworthy given between-study differences in sociodemographic and design characteristics.
- No consistent associations were found between psychological factors and neuropathology.
- However, individuals higher in conscientiousness who did not receive a clinical diagnosis tended to have a lower Braak stage at autopsy, suggesting the possibility that conscientiousness is related to cognitive resilience.
IN PRACTICE:
“These results replicate and extend evidence that personality traits may assist in early identification and dementia-care planning strategies, as well as risk stratification for dementia diagnosis. Moreover, our findings provide further support for recommendations to incorporate psychological trait measures into clinical screening or diagnosis criteria,” the authors write. SOURCE:
The study, with first author Emorie Beck, PhD, Department of Psychology, University of California, Davis, was published online on November 29, 2023, in Alzheimer’s & Dementia.
LIMITATIONS:
Access to autopsy data was limited. The findings may not generalize across racial groups. The analysis did not examine dynamic associations between changing personality and cognition and neuropathology over time.
DISCLOSURES:
The study was supported by grants from the National Institute on Aging. The authors have declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
CAR T-Cell Therapy: Cure for Systemic Autoimmune Diseases?
A single infusion of autologous CD19-directed CAR T-cell therapy led to persistent, drug-free remission in 15 patients with life-threatening systemic lupus erythematosus, idiopathic inflammatory myositis, or systemic sclerosis, according to research presented at the American Society of Hematology annual meeting.
The responses persisted at 15 months median follow-up, with all patients achieving complete remission, reported Fabian Mueller, MD, of the Bavarian Cancer Research Center and Friedrich-Alexander University of Erlangen-Nuremberg, Bavaria, Germany.
The CAR T-cell treatment appears to provide an “entire reset of B cells,” possibly even a cure, for these 15 patients who had run out of treatment options and had short life expectancies, Dr. Mueller said. “It’s impressive that we have treated these patients.”
Some of the cases have been described previously — including in Annals of the Rheumatic Diseases earlier this year, Nature Medicine in 2022, and the New England Journal of Medicine in 2021.
Now with substantially longer follow-up, the investigators have gained a greater understanding of “the B-cell biology behind our treatment,” Dr. Mueller said. However, “we need longer follow-up to establish how effective the treatment is going to be in the long run.”
All 15 patients included in the analysis were heavily pretreated and had multi-organ involvement. Prior to CAR T-cell therapy, patients had a median disease duration of 3 years, ranging from 1 to as many as 20 years, and had failed a median of five previous treatments. Patients were young — a median age of 36 years — which is much younger than most oncology patients who undergo CAR T-cell therapy, Dr. Mueller said.
The 15 patients underwent typical lymphodepletion and were apheresed and treated with a single infusion of 1 x 106 CD19 CAR T cells per kg of body weight — an established safe dose used in a phase 1 trial of B cell malignancies.
The CAR T cells, manufactured in-house, expanded rapidly, peaking around day 9. B cells disappeared within 7 days and began to reoccur in peripheral blood in all patients between 60 and 180 days. However, no disease flares occurred, Dr. Mueller said.
After 3 months, eight patients with systemic lupus erythematosus showed no sign of disease activity and dramatic improvement in symptoms. Three patients with idiopathic inflammatory myositis experienced major improvements in symptoms and normalization of creatinine kinase levels, the most clinically relevant marker for muscle inflammation. And three of four patients with systemic sclerosis demonstrated major improvements in symptoms and no new disease activity. These responses lasted for a median of 15 months, and all patients stopped taking immunosuppressive drugs.
Patients also tolerated the CAR T-cell treatment well, especially compared with the adverse event profile in oncology patients. Only low-grade inflammatory CAR T-related side effects occurred, and few patients required support for B-cell-derived immune deficiency.
However, infectious complications occurred in 14 patients, including urinary tract and respiratory infections, over the 12-month follow-up. One patient was hospitalized for severe pneumonia a few weeks after CAR T therapy, and two patients experienced herpes zoster reactivations, including one at 6 months and one at 12 months following treatment.
During a press briefing at the ASH conference, Dr. Mueller addressed the “critical question” of patient selection for CAR T-cell therapy, especially in light of the recently announced US Food and Drug Administration investigation exploring whether CAR T cells can cause secondary blood cancers.
Although the T-cell malignancy risk complicates matters, CAR T cells appear to behave differently in patients with autoimmune diseases than those with cancer, he said.
“We don’t understand the biology” related to the malignancy risk yet, Dr. Mueller said, but the benefit for end-of-life patients with no other treatment option likely outweighs the risk. That risk-benefit assessment, however, is more uncertain for those with less severe autoimmune diseases.
For now, it’s important to conduct individual assessments and inform patients about the risk, Dr. Mueller said.
Dr. Mueller disclosed relationships with BMS, AstraZeneca, Gilead, Janssen, Miltenyi Biomedicine, Novartis, Incyte, Abbvie, Sobi, and BeiGene.
A version of this article appeared on Medscape.com.
A single infusion of autologous CD19-directed CAR T-cell therapy led to persistent, drug-free remission in 15 patients with life-threatening systemic lupus erythematosus, idiopathic inflammatory myositis, or systemic sclerosis, according to research presented at the American Society of Hematology annual meeting.
The responses persisted at 15 months median follow-up, with all patients achieving complete remission, reported Fabian Mueller, MD, of the Bavarian Cancer Research Center and Friedrich-Alexander University of Erlangen-Nuremberg, Bavaria, Germany.
The CAR T-cell treatment appears to provide an “entire reset of B cells,” possibly even a cure, for these 15 patients who had run out of treatment options and had short life expectancies, Dr. Mueller said. “It’s impressive that we have treated these patients.”
Some of the cases have been described previously — including in Annals of the Rheumatic Diseases earlier this year, Nature Medicine in 2022, and the New England Journal of Medicine in 2021.
Now with substantially longer follow-up, the investigators have gained a greater understanding of “the B-cell biology behind our treatment,” Dr. Mueller said. However, “we need longer follow-up to establish how effective the treatment is going to be in the long run.”
All 15 patients included in the analysis were heavily pretreated and had multi-organ involvement. Prior to CAR T-cell therapy, patients had a median disease duration of 3 years, ranging from 1 to as many as 20 years, and had failed a median of five previous treatments. Patients were young — a median age of 36 years — which is much younger than most oncology patients who undergo CAR T-cell therapy, Dr. Mueller said.
The 15 patients underwent typical lymphodepletion and were apheresed and treated with a single infusion of 1 x 106 CD19 CAR T cells per kg of body weight — an established safe dose used in a phase 1 trial of B cell malignancies.
The CAR T cells, manufactured in-house, expanded rapidly, peaking around day 9. B cells disappeared within 7 days and began to reoccur in peripheral blood in all patients between 60 and 180 days. However, no disease flares occurred, Dr. Mueller said.
After 3 months, eight patients with systemic lupus erythematosus showed no sign of disease activity and dramatic improvement in symptoms. Three patients with idiopathic inflammatory myositis experienced major improvements in symptoms and normalization of creatinine kinase levels, the most clinically relevant marker for muscle inflammation. And three of four patients with systemic sclerosis demonstrated major improvements in symptoms and no new disease activity. These responses lasted for a median of 15 months, and all patients stopped taking immunosuppressive drugs.
Patients also tolerated the CAR T-cell treatment well, especially compared with the adverse event profile in oncology patients. Only low-grade inflammatory CAR T-related side effects occurred, and few patients required support for B-cell-derived immune deficiency.
However, infectious complications occurred in 14 patients, including urinary tract and respiratory infections, over the 12-month follow-up. One patient was hospitalized for severe pneumonia a few weeks after CAR T therapy, and two patients experienced herpes zoster reactivations, including one at 6 months and one at 12 months following treatment.
During a press briefing at the ASH conference, Dr. Mueller addressed the “critical question” of patient selection for CAR T-cell therapy, especially in light of the recently announced US Food and Drug Administration investigation exploring whether CAR T cells can cause secondary blood cancers.
Although the T-cell malignancy risk complicates matters, CAR T cells appear to behave differently in patients with autoimmune diseases than those with cancer, he said.
“We don’t understand the biology” related to the malignancy risk yet, Dr. Mueller said, but the benefit for end-of-life patients with no other treatment option likely outweighs the risk. That risk-benefit assessment, however, is more uncertain for those with less severe autoimmune diseases.
For now, it’s important to conduct individual assessments and inform patients about the risk, Dr. Mueller said.
Dr. Mueller disclosed relationships with BMS, AstraZeneca, Gilead, Janssen, Miltenyi Biomedicine, Novartis, Incyte, Abbvie, Sobi, and BeiGene.
A version of this article appeared on Medscape.com.
A single infusion of autologous CD19-directed CAR T-cell therapy led to persistent, drug-free remission in 15 patients with life-threatening systemic lupus erythematosus, idiopathic inflammatory myositis, or systemic sclerosis, according to research presented at the American Society of Hematology annual meeting.
The responses persisted at 15 months median follow-up, with all patients achieving complete remission, reported Fabian Mueller, MD, of the Bavarian Cancer Research Center and Friedrich-Alexander University of Erlangen-Nuremberg, Bavaria, Germany.
The CAR T-cell treatment appears to provide an “entire reset of B cells,” possibly even a cure, for these 15 patients who had run out of treatment options and had short life expectancies, Dr. Mueller said. “It’s impressive that we have treated these patients.”
Some of the cases have been described previously — including in Annals of the Rheumatic Diseases earlier this year, Nature Medicine in 2022, and the New England Journal of Medicine in 2021.
Now with substantially longer follow-up, the investigators have gained a greater understanding of “the B-cell biology behind our treatment,” Dr. Mueller said. However, “we need longer follow-up to establish how effective the treatment is going to be in the long run.”
All 15 patients included in the analysis were heavily pretreated and had multi-organ involvement. Prior to CAR T-cell therapy, patients had a median disease duration of 3 years, ranging from 1 to as many as 20 years, and had failed a median of five previous treatments. Patients were young — a median age of 36 years — which is much younger than most oncology patients who undergo CAR T-cell therapy, Dr. Mueller said.
The 15 patients underwent typical lymphodepletion and were apheresed and treated with a single infusion of 1 x 106 CD19 CAR T cells per kg of body weight — an established safe dose used in a phase 1 trial of B cell malignancies.
The CAR T cells, manufactured in-house, expanded rapidly, peaking around day 9. B cells disappeared within 7 days and began to reoccur in peripheral blood in all patients between 60 and 180 days. However, no disease flares occurred, Dr. Mueller said.
After 3 months, eight patients with systemic lupus erythematosus showed no sign of disease activity and dramatic improvement in symptoms. Three patients with idiopathic inflammatory myositis experienced major improvements in symptoms and normalization of creatinine kinase levels, the most clinically relevant marker for muscle inflammation. And three of four patients with systemic sclerosis demonstrated major improvements in symptoms and no new disease activity. These responses lasted for a median of 15 months, and all patients stopped taking immunosuppressive drugs.
Patients also tolerated the CAR T-cell treatment well, especially compared with the adverse event profile in oncology patients. Only low-grade inflammatory CAR T-related side effects occurred, and few patients required support for B-cell-derived immune deficiency.
However, infectious complications occurred in 14 patients, including urinary tract and respiratory infections, over the 12-month follow-up. One patient was hospitalized for severe pneumonia a few weeks after CAR T therapy, and two patients experienced herpes zoster reactivations, including one at 6 months and one at 12 months following treatment.
During a press briefing at the ASH conference, Dr. Mueller addressed the “critical question” of patient selection for CAR T-cell therapy, especially in light of the recently announced US Food and Drug Administration investigation exploring whether CAR T cells can cause secondary blood cancers.
Although the T-cell malignancy risk complicates matters, CAR T cells appear to behave differently in patients with autoimmune diseases than those with cancer, he said.
“We don’t understand the biology” related to the malignancy risk yet, Dr. Mueller said, but the benefit for end-of-life patients with no other treatment option likely outweighs the risk. That risk-benefit assessment, however, is more uncertain for those with less severe autoimmune diseases.
For now, it’s important to conduct individual assessments and inform patients about the risk, Dr. Mueller said.
Dr. Mueller disclosed relationships with BMS, AstraZeneca, Gilead, Janssen, Miltenyi Biomedicine, Novartis, Incyte, Abbvie, Sobi, and BeiGene.
A version of this article appeared on Medscape.com.
FROM ASH 2023
Bimekizumab shows promise for palmoplantar pustular psoriasis
.
PPP is a type of pustular psoriasis that remains a treatment challenge, and available treatments for palmoplantar plaque psoriasis with pustules also “remain unsatisfactory,” according to Thierry Passeron, MD, PhD, of the dermatology service at Centre Hospitalier Universitaire de Nice (France), and colleagues. Bimekizumab, an anti-interleukin (IL)-17A and anti-IL-17F antibody therapy, has been used for psoriasis and psoriatic arthritis (PsA), but its effectiveness for PPP has not been studied, they said. In the United States, bimekizumab (Bimzelx), administered subcutaneously, was recently approved for treating moderate to severe plaque psoriasis in adults; in the European Union, it is approved for treating psoriasis, in addition to psoriatic arthritis, axial spondyloarthritis and ankylosing spondylitis.
In the case series published in JAMA Dermatology, Dr. Passeron and coinvestigators identified 11 adults with PPP and 10 with palmoplantar plaque psoriasis with pustules who were treated at one of seven tertiary dermatology centers in France from September 2022 through June 2023. PPP also has been associated with bone and joint inflammation in SAPHO (synovitis, acne, pustulosis, hyperostosis, osteitis) syndrome.
All patients received bimekizumab for at least 3 months. The patients — 19 women and 2 men — ranged in age from 24 to 68 years (mean age, 46 years). The primary outcome was complete clearance, defined as an Investigator Global Assessment (IGA) score of 0.
A total of 17 patients achieved an IGA score of zero in 1-4 months. Over 3-6 months, three patients achieved an IGA score of 1 (almost clear), and one patient achieved an IGA score of 2 (mild).
Three patients with PPP also had acrodermatitis continua of Hallopeau; in these patients, nail involvement improved by 50%-70% after 4-6 months of bimekizumab use. Two patients with SAPHO experienced complete clearance of skin lesions associated with improvement in joint pain.
Four patients developed oral and genital candidiasis during treatment, but all were treated successfully with antifungals. None of the patients discontinued bimekizumab because of adverse events. “All patients are still receiving treatment, and their psoriatic lesions remain controlled,” the authors wrote.
“The rapid and consistent improvement observed in the present case series supports the effectiveness of bimekizumab therapy in managing PPP, palmoplantar plaque psoriasis with pustules, and SAPHO syndrome,” they said in their discussion.
The study findings were limited by several factors including the small sample size and short follow-up period, and by the inclusion of only patients with severe disease; and prospective, placebo-controlled studies are needed to confirm the results, the researchers noted.
However, the results suggest that bimekizumab could be a treatment approach for PPP, palmoplantar plaque psoriasis with pustules, and SAPHO syndrome, and warrant a prospective, randomized, placebo-controlled, randomized clinical trial to confirm the findings, they concluded.
Dr. Passeron disclosed fees from AbbVie, ACM Pharma, Almirall, Boehringer Ingelheim, Bristol Myers Squibb, Calypso, Celgene, Galderma, Genzyme/Sanofi, GlaxoSmithKline, Incyte, Janssen, LEO Pharma, Eli Lilly, Novartis, Roivant Sciences, Sun Pharmaceuticals, and VYNE Therapeutics outside the current study; he is a cofounder of Yukin Therapeutics. Three authors disclosed receiving personal fees from UCB, manufacturer of bimekizumab, outside of the submitted work, another author disclosed receiving personal fees from UCB during the conduct of the study, and another reported receiving grants from UCB and several other companies, outside the submitted work.
The study findings were also presented at a meeting, Les Journées Dermatologiques de Paris 2023, on December 6, in Paris.
.
PPP is a type of pustular psoriasis that remains a treatment challenge, and available treatments for palmoplantar plaque psoriasis with pustules also “remain unsatisfactory,” according to Thierry Passeron, MD, PhD, of the dermatology service at Centre Hospitalier Universitaire de Nice (France), and colleagues. Bimekizumab, an anti-interleukin (IL)-17A and anti-IL-17F antibody therapy, has been used for psoriasis and psoriatic arthritis (PsA), but its effectiveness for PPP has not been studied, they said. In the United States, bimekizumab (Bimzelx), administered subcutaneously, was recently approved for treating moderate to severe plaque psoriasis in adults; in the European Union, it is approved for treating psoriasis, in addition to psoriatic arthritis, axial spondyloarthritis and ankylosing spondylitis.
In the case series published in JAMA Dermatology, Dr. Passeron and coinvestigators identified 11 adults with PPP and 10 with palmoplantar plaque psoriasis with pustules who were treated at one of seven tertiary dermatology centers in France from September 2022 through June 2023. PPP also has been associated with bone and joint inflammation in SAPHO (synovitis, acne, pustulosis, hyperostosis, osteitis) syndrome.
All patients received bimekizumab for at least 3 months. The patients — 19 women and 2 men — ranged in age from 24 to 68 years (mean age, 46 years). The primary outcome was complete clearance, defined as an Investigator Global Assessment (IGA) score of 0.
A total of 17 patients achieved an IGA score of zero in 1-4 months. Over 3-6 months, three patients achieved an IGA score of 1 (almost clear), and one patient achieved an IGA score of 2 (mild).
Three patients with PPP also had acrodermatitis continua of Hallopeau; in these patients, nail involvement improved by 50%-70% after 4-6 months of bimekizumab use. Two patients with SAPHO experienced complete clearance of skin lesions associated with improvement in joint pain.
Four patients developed oral and genital candidiasis during treatment, but all were treated successfully with antifungals. None of the patients discontinued bimekizumab because of adverse events. “All patients are still receiving treatment, and their psoriatic lesions remain controlled,” the authors wrote.
“The rapid and consistent improvement observed in the present case series supports the effectiveness of bimekizumab therapy in managing PPP, palmoplantar plaque psoriasis with pustules, and SAPHO syndrome,” they said in their discussion.
The study findings were limited by several factors including the small sample size and short follow-up period, and by the inclusion of only patients with severe disease; and prospective, placebo-controlled studies are needed to confirm the results, the researchers noted.
However, the results suggest that bimekizumab could be a treatment approach for PPP, palmoplantar plaque psoriasis with pustules, and SAPHO syndrome, and warrant a prospective, randomized, placebo-controlled, randomized clinical trial to confirm the findings, they concluded.
Dr. Passeron disclosed fees from AbbVie, ACM Pharma, Almirall, Boehringer Ingelheim, Bristol Myers Squibb, Calypso, Celgene, Galderma, Genzyme/Sanofi, GlaxoSmithKline, Incyte, Janssen, LEO Pharma, Eli Lilly, Novartis, Roivant Sciences, Sun Pharmaceuticals, and VYNE Therapeutics outside the current study; he is a cofounder of Yukin Therapeutics. Three authors disclosed receiving personal fees from UCB, manufacturer of bimekizumab, outside of the submitted work, another author disclosed receiving personal fees from UCB during the conduct of the study, and another reported receiving grants from UCB and several other companies, outside the submitted work.
The study findings were also presented at a meeting, Les Journées Dermatologiques de Paris 2023, on December 6, in Paris.
.
PPP is a type of pustular psoriasis that remains a treatment challenge, and available treatments for palmoplantar plaque psoriasis with pustules also “remain unsatisfactory,” according to Thierry Passeron, MD, PhD, of the dermatology service at Centre Hospitalier Universitaire de Nice (France), and colleagues. Bimekizumab, an anti-interleukin (IL)-17A and anti-IL-17F antibody therapy, has been used for psoriasis and psoriatic arthritis (PsA), but its effectiveness for PPP has not been studied, they said. In the United States, bimekizumab (Bimzelx), administered subcutaneously, was recently approved for treating moderate to severe plaque psoriasis in adults; in the European Union, it is approved for treating psoriasis, in addition to psoriatic arthritis, axial spondyloarthritis and ankylosing spondylitis.
In the case series published in JAMA Dermatology, Dr. Passeron and coinvestigators identified 11 adults with PPP and 10 with palmoplantar plaque psoriasis with pustules who were treated at one of seven tertiary dermatology centers in France from September 2022 through June 2023. PPP also has been associated with bone and joint inflammation in SAPHO (synovitis, acne, pustulosis, hyperostosis, osteitis) syndrome.
All patients received bimekizumab for at least 3 months. The patients — 19 women and 2 men — ranged in age from 24 to 68 years (mean age, 46 years). The primary outcome was complete clearance, defined as an Investigator Global Assessment (IGA) score of 0.
A total of 17 patients achieved an IGA score of zero in 1-4 months. Over 3-6 months, three patients achieved an IGA score of 1 (almost clear), and one patient achieved an IGA score of 2 (mild).
Three patients with PPP also had acrodermatitis continua of Hallopeau; in these patients, nail involvement improved by 50%-70% after 4-6 months of bimekizumab use. Two patients with SAPHO experienced complete clearance of skin lesions associated with improvement in joint pain.
Four patients developed oral and genital candidiasis during treatment, but all were treated successfully with antifungals. None of the patients discontinued bimekizumab because of adverse events. “All patients are still receiving treatment, and their psoriatic lesions remain controlled,” the authors wrote.
“The rapid and consistent improvement observed in the present case series supports the effectiveness of bimekizumab therapy in managing PPP, palmoplantar plaque psoriasis with pustules, and SAPHO syndrome,” they said in their discussion.
The study findings were limited by several factors including the small sample size and short follow-up period, and by the inclusion of only patients with severe disease; and prospective, placebo-controlled studies are needed to confirm the results, the researchers noted.
However, the results suggest that bimekizumab could be a treatment approach for PPP, palmoplantar plaque psoriasis with pustules, and SAPHO syndrome, and warrant a prospective, randomized, placebo-controlled, randomized clinical trial to confirm the findings, they concluded.
Dr. Passeron disclosed fees from AbbVie, ACM Pharma, Almirall, Boehringer Ingelheim, Bristol Myers Squibb, Calypso, Celgene, Galderma, Genzyme/Sanofi, GlaxoSmithKline, Incyte, Janssen, LEO Pharma, Eli Lilly, Novartis, Roivant Sciences, Sun Pharmaceuticals, and VYNE Therapeutics outside the current study; he is a cofounder of Yukin Therapeutics. Three authors disclosed receiving personal fees from UCB, manufacturer of bimekizumab, outside of the submitted work, another author disclosed receiving personal fees from UCB during the conduct of the study, and another reported receiving grants from UCB and several other companies, outside the submitted work.
The study findings were also presented at a meeting, Les Journées Dermatologiques de Paris 2023, on December 6, in Paris.
FROM JAMA DERMATOLOGY
Some reasons to get off the fence about COVID booster
Though many people remain on the fence about getting the latest COVID vaccine booster, new research suggests a strong argument for getting the shot this winter: It sharply reduces the risk for COVID.
The risk reduction was 37% for those who received two doses. Experts say the research provides a strong argument for getting the vaccine, noting that about 10% of people infected with COVID go on to have long COVID, which can be debilitating for one quarter of those with long-lasting symptoms.
The data come from a systematic literature review and meta-analysis published in October in Antimicrobial Stewardship & Epidemiology. Researchers examined 32 studies published between December 2019 and June 2023, involving 775,931 adults. Twenty-four studies, encompassing 620,221 individuals, were included in the meta-analysis.
“The body of evidence from all these different studies converge on one single reality — that vaccines reduce the risk of long COVID, and people who keep up to date on their vaccinations also fared better than people who got it once or twice and didn’t follow up,” said Ziyad Al-Aly, MD, a clinical epidemiologist at Washington University in St Louis.
Researchers have reported similar results for children. The National Institutes of Health RECOVER Initiative team found that vaccines are up to 42% effective in preventing long COVID in children, said Dr. Carlos Oliveira, MD, a pediatric infectious diseases specialist and Yale researcher who contributed to the study, which is in preprint.
Vaccines also protect children from multisystem inflammatory syndrome, a condition that can happen after COVID, as well as protect against other COVID-related problems, such as missed school days, Oliveira said. “Even if the vaccine doesn’t completely stop long COVID, it’s still good for kids to get vaccinated for all these other reasons.”
However, uptake for the latest boosters has been slow: the Centers for Disease Control and Prevention reported that by mid-November, less than 16% of people aged 18 years or older had received a shot. For children, the number was closer to 6%. A recent Kaiser Family Foundation survey found that booster rates for adults are similar to what it was 1 year ago.
The survey results suggest that people are no longer as worried about COVID, which is why there is less concerned about keeping up with boosters. Though the current mutation of the virus is not as debilitating as its predecessors, long COVID continues to be a problem: as of January 2023, 28% of people who had contracted the virus had experienced long-COVID symptoms. And though the mechanisms are still not fully understood, and researchers have yet to agree on a definition of long COVID, they are certain about this much: The best way to avoid it is to avoid getting infected to begin with.
The lack of a diagnostic test for long COVID and the fact that the symptoms mimic those of other diseases lead to inconsistency that can make studies hard to replicate. In the papers reviewed for the Antimicrobial Stewardship & Epidemiology study, long COVID was defined as having symptoms lasting from more than 4 weeks to more than 6 months. Alexandre Marra, MD, the lead author and a researcher at the Hospital Israelita Albert Einstein, in São Paulo, Brazil, and at the University of Iowa, said that a clear standard definition is needed to better understand the actual prevalence and evaluate vaccine effectiveness.
Al-Aly noted that there is a logical explanation for one finding in the paper: The percentage of individuals who had COVID and reported that long-COVID symptoms declined from 19% in June 2022 to 11% in January 2023.
Because a pandemic is a dynamic event, constantly producing different variants with different phenotypes, the prevalence of disease is naturally going to be affected. “People who got infected early in the pandemic may have a different long COVID profile and long COVID risk than people who got infected in the second or third year of the pandemic,” Al-Aly said.
Most of the studies reported data from before the Omicron-variant era. Only eight reported data during that era. Omicron was not as lethal as previous variants, and consequently, fewer patients developed long COVID during that time.
One of those who did is Yeng Chang, age 40 years, a family doctor who lives in Sherwood Park, Alberta, Canada. Chang developed long COVID during fall 2022 after getting the virus in June. By then, she’d been vaccinated three times, but she isn’t surprised that she got sick because each vaccine she had was developed before Omicron.
“When I had COVID I was really sick, but I was well enough to stay home,” she said. “I think if I didn’t have my immunizations, I might have been hospitalized, and I don’t know what would have happened.”
Long COVID has left Chang with brain fog, fatigue, and a lack of physical stamina that forced her to pause her medical practice. For the past year and a half, she’s spent more time as a patient than a physician.
Chang had her fifth COVID vaccination in the fall and recommends that others do the same. “The booster you got however many years ago was effective for the COVID of that time but there is a new COVID now. You can’t just say, ‘I had one and I’m fine forever.’”
A version of this article appeared on Medscape.com.
Though many people remain on the fence about getting the latest COVID vaccine booster, new research suggests a strong argument for getting the shot this winter: It sharply reduces the risk for COVID.
The risk reduction was 37% for those who received two doses. Experts say the research provides a strong argument for getting the vaccine, noting that about 10% of people infected with COVID go on to have long COVID, which can be debilitating for one quarter of those with long-lasting symptoms.
The data come from a systematic literature review and meta-analysis published in October in Antimicrobial Stewardship & Epidemiology. Researchers examined 32 studies published between December 2019 and June 2023, involving 775,931 adults. Twenty-four studies, encompassing 620,221 individuals, were included in the meta-analysis.
“The body of evidence from all these different studies converge on one single reality — that vaccines reduce the risk of long COVID, and people who keep up to date on their vaccinations also fared better than people who got it once or twice and didn’t follow up,” said Ziyad Al-Aly, MD, a clinical epidemiologist at Washington University in St Louis.
Researchers have reported similar results for children. The National Institutes of Health RECOVER Initiative team found that vaccines are up to 42% effective in preventing long COVID in children, said Dr. Carlos Oliveira, MD, a pediatric infectious diseases specialist and Yale researcher who contributed to the study, which is in preprint.
Vaccines also protect children from multisystem inflammatory syndrome, a condition that can happen after COVID, as well as protect against other COVID-related problems, such as missed school days, Oliveira said. “Even if the vaccine doesn’t completely stop long COVID, it’s still good for kids to get vaccinated for all these other reasons.”
However, uptake for the latest boosters has been slow: the Centers for Disease Control and Prevention reported that by mid-November, less than 16% of people aged 18 years or older had received a shot. For children, the number was closer to 6%. A recent Kaiser Family Foundation survey found that booster rates for adults are similar to what it was 1 year ago.
The survey results suggest that people are no longer as worried about COVID, which is why there is less concerned about keeping up with boosters. Though the current mutation of the virus is not as debilitating as its predecessors, long COVID continues to be a problem: as of January 2023, 28% of people who had contracted the virus had experienced long-COVID symptoms. And though the mechanisms are still not fully understood, and researchers have yet to agree on a definition of long COVID, they are certain about this much: The best way to avoid it is to avoid getting infected to begin with.
The lack of a diagnostic test for long COVID and the fact that the symptoms mimic those of other diseases lead to inconsistency that can make studies hard to replicate. In the papers reviewed for the Antimicrobial Stewardship & Epidemiology study, long COVID was defined as having symptoms lasting from more than 4 weeks to more than 6 months. Alexandre Marra, MD, the lead author and a researcher at the Hospital Israelita Albert Einstein, in São Paulo, Brazil, and at the University of Iowa, said that a clear standard definition is needed to better understand the actual prevalence and evaluate vaccine effectiveness.
Al-Aly noted that there is a logical explanation for one finding in the paper: The percentage of individuals who had COVID and reported that long-COVID symptoms declined from 19% in June 2022 to 11% in January 2023.
Because a pandemic is a dynamic event, constantly producing different variants with different phenotypes, the prevalence of disease is naturally going to be affected. “People who got infected early in the pandemic may have a different long COVID profile and long COVID risk than people who got infected in the second or third year of the pandemic,” Al-Aly said.
Most of the studies reported data from before the Omicron-variant era. Only eight reported data during that era. Omicron was not as lethal as previous variants, and consequently, fewer patients developed long COVID during that time.
One of those who did is Yeng Chang, age 40 years, a family doctor who lives in Sherwood Park, Alberta, Canada. Chang developed long COVID during fall 2022 after getting the virus in June. By then, she’d been vaccinated three times, but she isn’t surprised that she got sick because each vaccine she had was developed before Omicron.
“When I had COVID I was really sick, but I was well enough to stay home,” she said. “I think if I didn’t have my immunizations, I might have been hospitalized, and I don’t know what would have happened.”
Long COVID has left Chang with brain fog, fatigue, and a lack of physical stamina that forced her to pause her medical practice. For the past year and a half, she’s spent more time as a patient than a physician.
Chang had her fifth COVID vaccination in the fall and recommends that others do the same. “The booster you got however many years ago was effective for the COVID of that time but there is a new COVID now. You can’t just say, ‘I had one and I’m fine forever.’”
A version of this article appeared on Medscape.com.
Though many people remain on the fence about getting the latest COVID vaccine booster, new research suggests a strong argument for getting the shot this winter: It sharply reduces the risk for COVID.
The risk reduction was 37% for those who received two doses. Experts say the research provides a strong argument for getting the vaccine, noting that about 10% of people infected with COVID go on to have long COVID, which can be debilitating for one quarter of those with long-lasting symptoms.
The data come from a systematic literature review and meta-analysis published in October in Antimicrobial Stewardship & Epidemiology. Researchers examined 32 studies published between December 2019 and June 2023, involving 775,931 adults. Twenty-four studies, encompassing 620,221 individuals, were included in the meta-analysis.
“The body of evidence from all these different studies converge on one single reality — that vaccines reduce the risk of long COVID, and people who keep up to date on their vaccinations also fared better than people who got it once or twice and didn’t follow up,” said Ziyad Al-Aly, MD, a clinical epidemiologist at Washington University in St Louis.
Researchers have reported similar results for children. The National Institutes of Health RECOVER Initiative team found that vaccines are up to 42% effective in preventing long COVID in children, said Dr. Carlos Oliveira, MD, a pediatric infectious diseases specialist and Yale researcher who contributed to the study, which is in preprint.
Vaccines also protect children from multisystem inflammatory syndrome, a condition that can happen after COVID, as well as protect against other COVID-related problems, such as missed school days, Oliveira said. “Even if the vaccine doesn’t completely stop long COVID, it’s still good for kids to get vaccinated for all these other reasons.”
However, uptake for the latest boosters has been slow: the Centers for Disease Control and Prevention reported that by mid-November, less than 16% of people aged 18 years or older had received a shot. For children, the number was closer to 6%. A recent Kaiser Family Foundation survey found that booster rates for adults are similar to what it was 1 year ago.
The survey results suggest that people are no longer as worried about COVID, which is why there is less concerned about keeping up with boosters. Though the current mutation of the virus is not as debilitating as its predecessors, long COVID continues to be a problem: as of January 2023, 28% of people who had contracted the virus had experienced long-COVID symptoms. And though the mechanisms are still not fully understood, and researchers have yet to agree on a definition of long COVID, they are certain about this much: The best way to avoid it is to avoid getting infected to begin with.
The lack of a diagnostic test for long COVID and the fact that the symptoms mimic those of other diseases lead to inconsistency that can make studies hard to replicate. In the papers reviewed for the Antimicrobial Stewardship & Epidemiology study, long COVID was defined as having symptoms lasting from more than 4 weeks to more than 6 months. Alexandre Marra, MD, the lead author and a researcher at the Hospital Israelita Albert Einstein, in São Paulo, Brazil, and at the University of Iowa, said that a clear standard definition is needed to better understand the actual prevalence and evaluate vaccine effectiveness.
Al-Aly noted that there is a logical explanation for one finding in the paper: The percentage of individuals who had COVID and reported that long-COVID symptoms declined from 19% in June 2022 to 11% in January 2023.
Because a pandemic is a dynamic event, constantly producing different variants with different phenotypes, the prevalence of disease is naturally going to be affected. “People who got infected early in the pandemic may have a different long COVID profile and long COVID risk than people who got infected in the second or third year of the pandemic,” Al-Aly said.
Most of the studies reported data from before the Omicron-variant era. Only eight reported data during that era. Omicron was not as lethal as previous variants, and consequently, fewer patients developed long COVID during that time.
One of those who did is Yeng Chang, age 40 years, a family doctor who lives in Sherwood Park, Alberta, Canada. Chang developed long COVID during fall 2022 after getting the virus in June. By then, she’d been vaccinated three times, but she isn’t surprised that she got sick because each vaccine she had was developed before Omicron.
“When I had COVID I was really sick, but I was well enough to stay home,” she said. “I think if I didn’t have my immunizations, I might have been hospitalized, and I don’t know what would have happened.”
Long COVID has left Chang with brain fog, fatigue, and a lack of physical stamina that forced her to pause her medical practice. For the past year and a half, she’s spent more time as a patient than a physician.
Chang had her fifth COVID vaccination in the fall and recommends that others do the same. “The booster you got however many years ago was effective for the COVID of that time but there is a new COVID now. You can’t just say, ‘I had one and I’m fine forever.’”
A version of this article appeared on Medscape.com.
Vegan diet confers cardiometabolic benefits
TOPLINE:
Compared with a healthy omnivore diet, low-density lipoprotein cholesterol (LDL-C) as well as fasting insulin and weight loss in a randomized controlled trial of identical twins.
METHODOLOGY:
- Researchers randomly assigned 22 pairs of healthy adult identical twins (34 women, mean age 39 years, mean body mass index 25.9) to a healthy vegan or omnivore diet (1 twin per pair) for 8 weeks.
- For the first 4 weeks, diet-specific meals were provided via a meal delivery service. For the final 4 weeks, participants prepared their own diet-appropriate meals/snacks.
- The primary outcome was change in LDL-C; secondary outcomes included changes in body weight and fasting insulin.
TAKEAWAY:
- After 8 weeks, twins eating a vegan diet showed a significant mean decrease of 13.9 mg/dL in LDL-C compared with twins eating an omnivorous diet.
- The vegan diet also led to a significant mean decrease of 2.9 Times New RomanμIU/mL in fasting insulin and 1.9 kg in body weight after 8 weeks compared with the omnivore diet, although weight loss was observed in both diet groups.
- The vegan diet group also had a larger but nonsignificant absolute median decrease in fasting HDL-C triglycerides, vitamin B12, glucose, and trimethylamine N-oxide levels at 8 weeks.
IN PRACTICE:
“Our results corroborate a previous finding showing that eating a vegan diet can improve cardiovascular health. Clinicians may consider recommending plant-based diets to reduce cardiometabolic risk factors, as well as aligning with environmental benefits,” the researchers concluded.
SOURCE:
The study, with first author Matthew J. Landry, PhD, RDN, Stanford Prevention Research Center, Stanford University School of Medicine, California, was published online November 30 in JAMA Network Open.
LIMITATIONS:
The adult twin population was generally healthy and findings may not be generalizable to other populations. The sample size was small, and the duration of intervention was short and there was no follow-up period, which limits insights on stability and sustainability of the diets.
DISCLOSURES:
Funding was provided by the Vogt Foundation, and grants from Stanford University and the National Heart, Lung, and Blood Institute. Dr. Landry has no relevant disclosures. One author reported receiving funding from Beyond Meat outside of this study.
A version of this article appeared on Medscape.com.
TOPLINE:
Compared with a healthy omnivore diet, low-density lipoprotein cholesterol (LDL-C) as well as fasting insulin and weight loss in a randomized controlled trial of identical twins.
METHODOLOGY:
- Researchers randomly assigned 22 pairs of healthy adult identical twins (34 women, mean age 39 years, mean body mass index 25.9) to a healthy vegan or omnivore diet (1 twin per pair) for 8 weeks.
- For the first 4 weeks, diet-specific meals were provided via a meal delivery service. For the final 4 weeks, participants prepared their own diet-appropriate meals/snacks.
- The primary outcome was change in LDL-C; secondary outcomes included changes in body weight and fasting insulin.
TAKEAWAY:
- After 8 weeks, twins eating a vegan diet showed a significant mean decrease of 13.9 mg/dL in LDL-C compared with twins eating an omnivorous diet.
- The vegan diet also led to a significant mean decrease of 2.9 Times New RomanμIU/mL in fasting insulin and 1.9 kg in body weight after 8 weeks compared with the omnivore diet, although weight loss was observed in both diet groups.
- The vegan diet group also had a larger but nonsignificant absolute median decrease in fasting HDL-C triglycerides, vitamin B12, glucose, and trimethylamine N-oxide levels at 8 weeks.
IN PRACTICE:
“Our results corroborate a previous finding showing that eating a vegan diet can improve cardiovascular health. Clinicians may consider recommending plant-based diets to reduce cardiometabolic risk factors, as well as aligning with environmental benefits,” the researchers concluded.
SOURCE:
The study, with first author Matthew J. Landry, PhD, RDN, Stanford Prevention Research Center, Stanford University School of Medicine, California, was published online November 30 in JAMA Network Open.
LIMITATIONS:
The adult twin population was generally healthy and findings may not be generalizable to other populations. The sample size was small, and the duration of intervention was short and there was no follow-up period, which limits insights on stability and sustainability of the diets.
DISCLOSURES:
Funding was provided by the Vogt Foundation, and grants from Stanford University and the National Heart, Lung, and Blood Institute. Dr. Landry has no relevant disclosures. One author reported receiving funding from Beyond Meat outside of this study.
A version of this article appeared on Medscape.com.
TOPLINE:
Compared with a healthy omnivore diet, low-density lipoprotein cholesterol (LDL-C) as well as fasting insulin and weight loss in a randomized controlled trial of identical twins.
METHODOLOGY:
- Researchers randomly assigned 22 pairs of healthy adult identical twins (34 women, mean age 39 years, mean body mass index 25.9) to a healthy vegan or omnivore diet (1 twin per pair) for 8 weeks.
- For the first 4 weeks, diet-specific meals were provided via a meal delivery service. For the final 4 weeks, participants prepared their own diet-appropriate meals/snacks.
- The primary outcome was change in LDL-C; secondary outcomes included changes in body weight and fasting insulin.
TAKEAWAY:
- After 8 weeks, twins eating a vegan diet showed a significant mean decrease of 13.9 mg/dL in LDL-C compared with twins eating an omnivorous diet.
- The vegan diet also led to a significant mean decrease of 2.9 Times New RomanμIU/mL in fasting insulin and 1.9 kg in body weight after 8 weeks compared with the omnivore diet, although weight loss was observed in both diet groups.
- The vegan diet group also had a larger but nonsignificant absolute median decrease in fasting HDL-C triglycerides, vitamin B12, glucose, and trimethylamine N-oxide levels at 8 weeks.
IN PRACTICE:
“Our results corroborate a previous finding showing that eating a vegan diet can improve cardiovascular health. Clinicians may consider recommending plant-based diets to reduce cardiometabolic risk factors, as well as aligning with environmental benefits,” the researchers concluded.
SOURCE:
The study, with first author Matthew J. Landry, PhD, RDN, Stanford Prevention Research Center, Stanford University School of Medicine, California, was published online November 30 in JAMA Network Open.
LIMITATIONS:
The adult twin population was generally healthy and findings may not be generalizable to other populations. The sample size was small, and the duration of intervention was short and there was no follow-up period, which limits insights on stability and sustainability of the diets.
DISCLOSURES:
Funding was provided by the Vogt Foundation, and grants from Stanford University and the National Heart, Lung, and Blood Institute. Dr. Landry has no relevant disclosures. One author reported receiving funding from Beyond Meat outside of this study.
A version of this article appeared on Medscape.com.
Are you sure your patient is alive?
This transcript has been edited for clarity.
Much of my research focuses on what is known as clinical decision support — prompts and messages to providers to help them make good decisions for their patients. I know that these things can be annoying, which is exactly why I study them — to figure out which ones actually help.
When I got started on this about 10 years ago, we were learning a lot about how best to message providers about their patients. My team had developed a simple alert for acute kidney injury (AKI). We knew that providers often missed the diagnosis, so maybe letting them know would improve patient outcomes.
As we tested the alert, we got feedback, and I have kept an email from an ICU doctor from those early days. It read:
Dear Dr. Wilson: Thank you for the automated alert informing me that my patient had AKI. Regrettably, the alert fired about an hour after the patient had died. I feel that the information is less than actionable at this time.
Our early system had neglected to add a conditional flag ensuring that the patient was still alive at the time it sent the alert message. A small oversight, but one that had very large implications. Future studies would show that “false positive” alerts like this seriously degrade physician confidence in the system. And why wouldn’t they?
Not knowing the vital status of a patient can have major consequences.
Health systems send messages to their patients all the time: reminders of appointments, reminders for preventive care, reminders for vaccinations, and so on.
But what if the patient being reminded has died? It’s a waste of resources, of course, but more than that, it can be painful for their families and reflects poorly on the health care system. Of all the people who should know whether someone is alive or dead, shouldn’t their doctor be at the top of the list?
A new study in JAMA Internal Medicine quantifies this very phenomenon.
Researchers examined 11,658 primary care patients in their health system who met the criteria of being “seriously ill” and followed them for 2 years. During that period of time, 25% were recorded as deceased in the electronic health record. But 30.8% had died. That left 676 patients who had died, but were not known to have died, left in the system.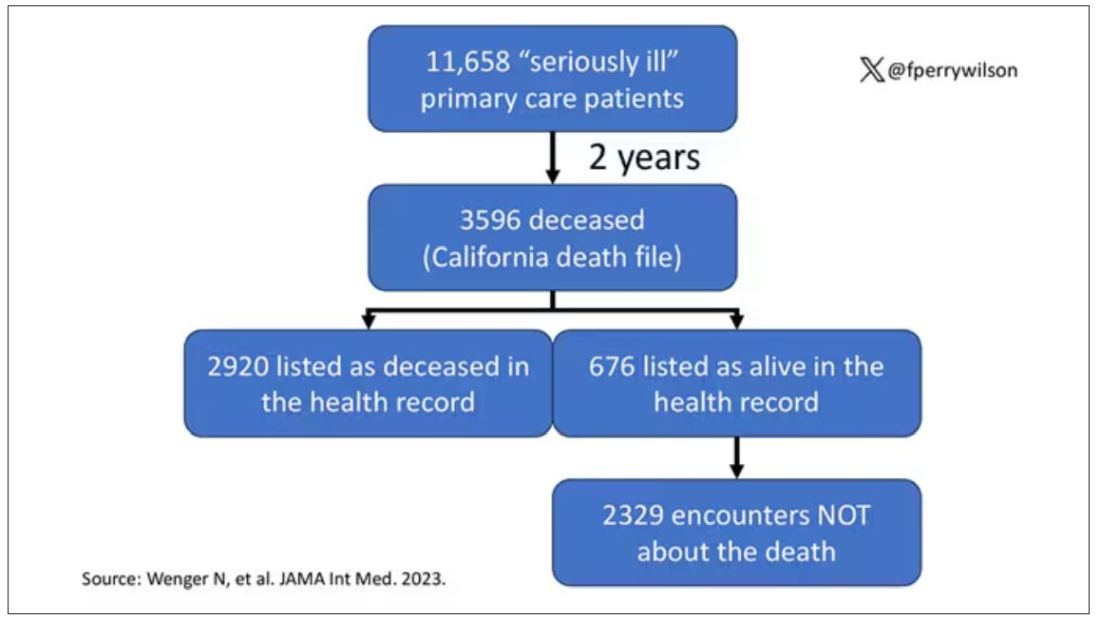
And those 676 were not left to rest in peace. They received 221 telephone and 338 health portal messages not related to death, and 920 letters reminding them about unmet primary care metrics like flu shots and cancer screening. Orders were entered into the health record for things like vaccines and routine screenings for 158 patients, and 310 future appointments — destined to be no-shows — were still on the books. One can only imagine the frustration of families checking their mail and finding yet another letter reminding their deceased loved one to get a mammogram.
How did the researchers figure out who had died? It turns out it’s not that hard. California keeps a record of all deaths in the state; they simply had to search it. Like all state death records, they tend to lag a bit so it’s not clinically terribly useful, but it works. California and most other states also have a very accurate and up-to-date death file which can only be used by law enforcement to investigate criminal activity and fraud; health care is left in the lurch.
Nationwide, there is the real-time fact of death service, supported by the National Association for Public Health Statistics and Information Systems. This allows employers to verify, in real time, whether the person applying for a job is alive. Healthcare systems are not allowed to use it.
Let’s also remember that very few people die in this country without some health care agency knowing about it and recording it. But sharing of medical information is so poor in the United States that your patient could die in a hospital one city away from you and you might not find out until you’re calling them to see why they missed a scheduled follow-up appointment.
These events — the embarrassing lack of knowledge about the very vital status of our patients — highlight a huge problem with health care in our country. The fragmented health care system is terrible at data sharing, in part because of poor protocols, in part because of unfounded concerns about patient privacy, and in part because of a tendency to hoard data that might be valuable in the future. It has to stop. We need to know how our patients are doing even when they are not sitting in front of us. When it comes to life and death, the knowledge is out there; we just can’t access it. Seems like a pretty easy fix.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com .
This transcript has been edited for clarity.
Much of my research focuses on what is known as clinical decision support — prompts and messages to providers to help them make good decisions for their patients. I know that these things can be annoying, which is exactly why I study them — to figure out which ones actually help.
When I got started on this about 10 years ago, we were learning a lot about how best to message providers about their patients. My team had developed a simple alert for acute kidney injury (AKI). We knew that providers often missed the diagnosis, so maybe letting them know would improve patient outcomes.
As we tested the alert, we got feedback, and I have kept an email from an ICU doctor from those early days. It read:
Dear Dr. Wilson: Thank you for the automated alert informing me that my patient had AKI. Regrettably, the alert fired about an hour after the patient had died. I feel that the information is less than actionable at this time.
Our early system had neglected to add a conditional flag ensuring that the patient was still alive at the time it sent the alert message. A small oversight, but one that had very large implications. Future studies would show that “false positive” alerts like this seriously degrade physician confidence in the system. And why wouldn’t they?
Not knowing the vital status of a patient can have major consequences.
Health systems send messages to their patients all the time: reminders of appointments, reminders for preventive care, reminders for vaccinations, and so on.
But what if the patient being reminded has died? It’s a waste of resources, of course, but more than that, it can be painful for their families and reflects poorly on the health care system. Of all the people who should know whether someone is alive or dead, shouldn’t their doctor be at the top of the list?
A new study in JAMA Internal Medicine quantifies this very phenomenon.
Researchers examined 11,658 primary care patients in their health system who met the criteria of being “seriously ill” and followed them for 2 years. During that period of time, 25% were recorded as deceased in the electronic health record. But 30.8% had died. That left 676 patients who had died, but were not known to have died, left in the system.
And those 676 were not left to rest in peace. They received 221 telephone and 338 health portal messages not related to death, and 920 letters reminding them about unmet primary care metrics like flu shots and cancer screening. Orders were entered into the health record for things like vaccines and routine screenings for 158 patients, and 310 future appointments — destined to be no-shows — were still on the books. One can only imagine the frustration of families checking their mail and finding yet another letter reminding their deceased loved one to get a mammogram.
How did the researchers figure out who had died? It turns out it’s not that hard. California keeps a record of all deaths in the state; they simply had to search it. Like all state death records, they tend to lag a bit so it’s not clinically terribly useful, but it works. California and most other states also have a very accurate and up-to-date death file which can only be used by law enforcement to investigate criminal activity and fraud; health care is left in the lurch.
Nationwide, there is the real-time fact of death service, supported by the National Association for Public Health Statistics and Information Systems. This allows employers to verify, in real time, whether the person applying for a job is alive. Healthcare systems are not allowed to use it.
Let’s also remember that very few people die in this country without some health care agency knowing about it and recording it. But sharing of medical information is so poor in the United States that your patient could die in a hospital one city away from you and you might not find out until you’re calling them to see why they missed a scheduled follow-up appointment.
These events — the embarrassing lack of knowledge about the very vital status of our patients — highlight a huge problem with health care in our country. The fragmented health care system is terrible at data sharing, in part because of poor protocols, in part because of unfounded concerns about patient privacy, and in part because of a tendency to hoard data that might be valuable in the future. It has to stop. We need to know how our patients are doing even when they are not sitting in front of us. When it comes to life and death, the knowledge is out there; we just can’t access it. Seems like a pretty easy fix.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com .
This transcript has been edited for clarity.
Much of my research focuses on what is known as clinical decision support — prompts and messages to providers to help them make good decisions for their patients. I know that these things can be annoying, which is exactly why I study them — to figure out which ones actually help.
When I got started on this about 10 years ago, we were learning a lot about how best to message providers about their patients. My team had developed a simple alert for acute kidney injury (AKI). We knew that providers often missed the diagnosis, so maybe letting them know would improve patient outcomes.
As we tested the alert, we got feedback, and I have kept an email from an ICU doctor from those early days. It read:
Dear Dr. Wilson: Thank you for the automated alert informing me that my patient had AKI. Regrettably, the alert fired about an hour after the patient had died. I feel that the information is less than actionable at this time.
Our early system had neglected to add a conditional flag ensuring that the patient was still alive at the time it sent the alert message. A small oversight, but one that had very large implications. Future studies would show that “false positive” alerts like this seriously degrade physician confidence in the system. And why wouldn’t they?
Not knowing the vital status of a patient can have major consequences.
Health systems send messages to their patients all the time: reminders of appointments, reminders for preventive care, reminders for vaccinations, and so on.
But what if the patient being reminded has died? It’s a waste of resources, of course, but more than that, it can be painful for their families and reflects poorly on the health care system. Of all the people who should know whether someone is alive or dead, shouldn’t their doctor be at the top of the list?
A new study in JAMA Internal Medicine quantifies this very phenomenon.
Researchers examined 11,658 primary care patients in their health system who met the criteria of being “seriously ill” and followed them for 2 years. During that period of time, 25% were recorded as deceased in the electronic health record. But 30.8% had died. That left 676 patients who had died, but were not known to have died, left in the system.
And those 676 were not left to rest in peace. They received 221 telephone and 338 health portal messages not related to death, and 920 letters reminding them about unmet primary care metrics like flu shots and cancer screening. Orders were entered into the health record for things like vaccines and routine screenings for 158 patients, and 310 future appointments — destined to be no-shows — were still on the books. One can only imagine the frustration of families checking their mail and finding yet another letter reminding their deceased loved one to get a mammogram.
How did the researchers figure out who had died? It turns out it’s not that hard. California keeps a record of all deaths in the state; they simply had to search it. Like all state death records, they tend to lag a bit so it’s not clinically terribly useful, but it works. California and most other states also have a very accurate and up-to-date death file which can only be used by law enforcement to investigate criminal activity and fraud; health care is left in the lurch.
Nationwide, there is the real-time fact of death service, supported by the National Association for Public Health Statistics and Information Systems. This allows employers to verify, in real time, whether the person applying for a job is alive. Healthcare systems are not allowed to use it.
Let’s also remember that very few people die in this country without some health care agency knowing about it and recording it. But sharing of medical information is so poor in the United States that your patient could die in a hospital one city away from you and you might not find out until you’re calling them to see why they missed a scheduled follow-up appointment.
These events — the embarrassing lack of knowledge about the very vital status of our patients — highlight a huge problem with health care in our country. The fragmented health care system is terrible at data sharing, in part because of poor protocols, in part because of unfounded concerns about patient privacy, and in part because of a tendency to hoard data that might be valuable in the future. It has to stop. We need to know how our patients are doing even when they are not sitting in front of us. When it comes to life and death, the knowledge is out there; we just can’t access it. Seems like a pretty easy fix.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com .
The prospect of a medication to treat OSA is getting closer
For researchers involved with sleep disorders, developing a pharmacologic treatment for obstructive sleep apnea (OSA) is a bit like searching for the holy grail. P K Schweitzer and colleagues have published the results of the randomized MARIPOSA study assessing a combination of two medicinal products known as AD109, one of the products having an antimuscarinic effect (aroxybutynin), and the other a noradrenergic effect (atomoxetine), in treating this condition.
MARIPOSA Methodology
The trial included 209 patients, 176 of whom completed the 4-week protocol. The trial was double-blinded according to four parallel arms: participants in the first and second arms received AD109 containing doses of 2.5 mg/75 mg and 5 mg/75 mg of aroxybutynin and atomoxetine, respectively. The third arm received atomoxetine alone (75 mg), and the fourth arm was given a placebo.
Two polysomnograms (PSGs) were carried out at the start and end of the trial, allowing researchers to calculate the apnea-hypopnea index (AHI) and to quantify nocturnal desaturation. The impact of these variables are now being deemed as the primary marker of the risk for cardiovascular complications secondary to OSA. Finally, questionnaires that evaluated excessive daytime sleepiness, fatigue, and sleep quality were completed.
The median age varied from 5 to 57 years, depending on the arm of the study, and body mass index varied between 31.2 and 34.5. Inclusion criteria comprised an AHI between 10 and 45 events per hour, of which, at least 75% were described as obstructive. Where continuous positive airway pressure (CPAP) was used (21%-30% of cases), it was abandoned during the trial (in a time frame that is perhaps too short to consider these patients as treatment naive).
Combination Brought Improvements
After the 4 weeks of treatment, the AHI measured via follow-up PSG went from a median of 20.5 to 10.8 (in arm one and from 19.4 to 9.5 in arm two (P < .0001 vs placebo in these two arms). For participants in arm three, AHI went from 19.0 to 11.8 (P < .01 vs placebo).
The rate of nocturnal desaturation (in percentage per hour) declined from -12.7 in arm one (P = .03), from -16.6 in arm two (P = .005), and from -5.2 in arm three (P = .003) compared with the placebo. The fatigue score was significantly improved by AD109 2.5 mg/75 mg. The use of atomoxetine alone slightly worsened the sleep disturbance score.
The main side effects were dry mouth sensation (which was markedly more common with AD109 5 mg/75 mg), difficulty passing urine in 7%-22% of cases, tachycardia in all trial arms, and increased diastolic blood pressure at the 2.5-mg/75-mg dose. The authors concluded that AD109, a combination of noradrenergic and antimuscarinic molecules, is effective in correcting mild to severe OSA.
The 2.5-mg/75-mg dose was as effective as the 5-mg/75-mg dose. Atomoxetine alone is less effective, has more side effects, and is associated with lower quality sleep. Finally, it is reported that compliance with oral treatment was not checked, yet the argument of patient noncompliance with CPAP is largely used by the authors in their presentation of their study. A phase 3 trial is underway.
Nevertheless, these results herald important scientific benefits if we consider that Colin Sullivan’s original 1981 research paper, which ushered in the CPAP era, presented the results of just five participants.
This article was translated from JIM, which is part of the Medscape professional network.
A version of this article appeared on Medscape.com.
For researchers involved with sleep disorders, developing a pharmacologic treatment for obstructive sleep apnea (OSA) is a bit like searching for the holy grail. P K Schweitzer and colleagues have published the results of the randomized MARIPOSA study assessing a combination of two medicinal products known as AD109, one of the products having an antimuscarinic effect (aroxybutynin), and the other a noradrenergic effect (atomoxetine), in treating this condition.
MARIPOSA Methodology
The trial included 209 patients, 176 of whom completed the 4-week protocol. The trial was double-blinded according to four parallel arms: participants in the first and second arms received AD109 containing doses of 2.5 mg/75 mg and 5 mg/75 mg of aroxybutynin and atomoxetine, respectively. The third arm received atomoxetine alone (75 mg), and the fourth arm was given a placebo.
Two polysomnograms (PSGs) were carried out at the start and end of the trial, allowing researchers to calculate the apnea-hypopnea index (AHI) and to quantify nocturnal desaturation. The impact of these variables are now being deemed as the primary marker of the risk for cardiovascular complications secondary to OSA. Finally, questionnaires that evaluated excessive daytime sleepiness, fatigue, and sleep quality were completed.
The median age varied from 5 to 57 years, depending on the arm of the study, and body mass index varied between 31.2 and 34.5. Inclusion criteria comprised an AHI between 10 and 45 events per hour, of which, at least 75% were described as obstructive. Where continuous positive airway pressure (CPAP) was used (21%-30% of cases), it was abandoned during the trial (in a time frame that is perhaps too short to consider these patients as treatment naive).
Combination Brought Improvements
After the 4 weeks of treatment, the AHI measured via follow-up PSG went from a median of 20.5 to 10.8 (in arm one and from 19.4 to 9.5 in arm two (P < .0001 vs placebo in these two arms). For participants in arm three, AHI went from 19.0 to 11.8 (P < .01 vs placebo).
The rate of nocturnal desaturation (in percentage per hour) declined from -12.7 in arm one (P = .03), from -16.6 in arm two (P = .005), and from -5.2 in arm three (P = .003) compared with the placebo. The fatigue score was significantly improved by AD109 2.5 mg/75 mg. The use of atomoxetine alone slightly worsened the sleep disturbance score.
The main side effects were dry mouth sensation (which was markedly more common with AD109 5 mg/75 mg), difficulty passing urine in 7%-22% of cases, tachycardia in all trial arms, and increased diastolic blood pressure at the 2.5-mg/75-mg dose. The authors concluded that AD109, a combination of noradrenergic and antimuscarinic molecules, is effective in correcting mild to severe OSA.
The 2.5-mg/75-mg dose was as effective as the 5-mg/75-mg dose. Atomoxetine alone is less effective, has more side effects, and is associated with lower quality sleep. Finally, it is reported that compliance with oral treatment was not checked, yet the argument of patient noncompliance with CPAP is largely used by the authors in their presentation of their study. A phase 3 trial is underway.
Nevertheless, these results herald important scientific benefits if we consider that Colin Sullivan’s original 1981 research paper, which ushered in the CPAP era, presented the results of just five participants.
This article was translated from JIM, which is part of the Medscape professional network.
A version of this article appeared on Medscape.com.
For researchers involved with sleep disorders, developing a pharmacologic treatment for obstructive sleep apnea (OSA) is a bit like searching for the holy grail. P K Schweitzer and colleagues have published the results of the randomized MARIPOSA study assessing a combination of two medicinal products known as AD109, one of the products having an antimuscarinic effect (aroxybutynin), and the other a noradrenergic effect (atomoxetine), in treating this condition.
MARIPOSA Methodology
The trial included 209 patients, 176 of whom completed the 4-week protocol. The trial was double-blinded according to four parallel arms: participants in the first and second arms received AD109 containing doses of 2.5 mg/75 mg and 5 mg/75 mg of aroxybutynin and atomoxetine, respectively. The third arm received atomoxetine alone (75 mg), and the fourth arm was given a placebo.
Two polysomnograms (PSGs) were carried out at the start and end of the trial, allowing researchers to calculate the apnea-hypopnea index (AHI) and to quantify nocturnal desaturation. The impact of these variables are now being deemed as the primary marker of the risk for cardiovascular complications secondary to OSA. Finally, questionnaires that evaluated excessive daytime sleepiness, fatigue, and sleep quality were completed.
The median age varied from 5 to 57 years, depending on the arm of the study, and body mass index varied between 31.2 and 34.5. Inclusion criteria comprised an AHI between 10 and 45 events per hour, of which, at least 75% were described as obstructive. Where continuous positive airway pressure (CPAP) was used (21%-30% of cases), it was abandoned during the trial (in a time frame that is perhaps too short to consider these patients as treatment naive).
Combination Brought Improvements
After the 4 weeks of treatment, the AHI measured via follow-up PSG went from a median of 20.5 to 10.8 (in arm one and from 19.4 to 9.5 in arm two (P < .0001 vs placebo in these two arms). For participants in arm three, AHI went from 19.0 to 11.8 (P < .01 vs placebo).
The rate of nocturnal desaturation (in percentage per hour) declined from -12.7 in arm one (P = .03), from -16.6 in arm two (P = .005), and from -5.2 in arm three (P = .003) compared with the placebo. The fatigue score was significantly improved by AD109 2.5 mg/75 mg. The use of atomoxetine alone slightly worsened the sleep disturbance score.
The main side effects were dry mouth sensation (which was markedly more common with AD109 5 mg/75 mg), difficulty passing urine in 7%-22% of cases, tachycardia in all trial arms, and increased diastolic blood pressure at the 2.5-mg/75-mg dose. The authors concluded that AD109, a combination of noradrenergic and antimuscarinic molecules, is effective in correcting mild to severe OSA.
The 2.5-mg/75-mg dose was as effective as the 5-mg/75-mg dose. Atomoxetine alone is less effective, has more side effects, and is associated with lower quality sleep. Finally, it is reported that compliance with oral treatment was not checked, yet the argument of patient noncompliance with CPAP is largely used by the authors in their presentation of their study. A phase 3 trial is underway.
Nevertheless, these results herald important scientific benefits if we consider that Colin Sullivan’s original 1981 research paper, which ushered in the CPAP era, presented the results of just five participants.
This article was translated from JIM, which is part of the Medscape professional network.
A version of this article appeared on Medscape.com.
‘Hidden hearing loss’ may cause tinnitus: Study
Scientists know that tinnitus, or ringing in the ears, affects 10% of adults worldwide. But they’re not exactly sure what causes the condition.
The traditional belief is that tinnitus happens in people who had already lost hearing. But some people who have tinnitus are still able to perform well on standard hearing tests, according to researchers at the Massachusetts Eye and Ear Infirmary. That happens because the tests don’t pick up auditory nerve loss, sometimes called “hidden hearing loss.”
Stéphane F. Maison, PhD, the lead author of a new study on tinnitus, said in a news release about the study.
Tinnitus is sometimes compared to phantom limb syndrome, in which people feel pain in limbs they no longer have. While the study published in Scientific Reports doesn’t refer to phantom limb syndrome, it does talk about “phantom sound.”
“In other words, the brain tries to compensate for the loss of hearing by increasing its activity, resulting in the perception of a phantom sound, tinnitus. Until recently though, this idea was disputed as some tinnitus sufferers have normal hearing tests,” the researchers explained in the news release.
Annoyed by the ringing in your ears? What causes tinnitus, and how can you get the sound to buzz off?
The study included 294 adults — 201 who had never reported having tinnitus, 64 who had reported having temporary tinnitus, and 29 who had reported having constant tinnitus for 6 months or more.
All 294 had performed normally on a pure tone test, in which subjects raise their hands when they hear beeps to measure the quietest sounds they can detect.
In a different kind of test, electrodes measured responses to clicking sounds in the inner ear, the auditory nerve, and the brain. The second test found reduced response in the auditory nerves and increased activity in the brainstem activity among those who had tinnitus.
Dr Maison, a principal investigator at Eaton-Peabody Laboratories at Mass Eye and Ear/Harvard Medical School, called the study “a first step toward our ultimate goal of silencing tinnitus.”
“Beyond the nuisance of having persistent ringing or other sounds in the ears, tinnitus symptoms are debilitating in many patients, causing sleep deprivation, social isolation, anxiety and depression, adversely affecting work performance, and reducing significantly their quality of life,” he said in the news release. “We won’t be able to cure tinnitus until we fully understand the mechanisms underlying its genesis.”
A version of this article appeared on WebMD.com.
Scientists know that tinnitus, or ringing in the ears, affects 10% of adults worldwide. But they’re not exactly sure what causes the condition.
The traditional belief is that tinnitus happens in people who had already lost hearing. But some people who have tinnitus are still able to perform well on standard hearing tests, according to researchers at the Massachusetts Eye and Ear Infirmary. That happens because the tests don’t pick up auditory nerve loss, sometimes called “hidden hearing loss.”
Stéphane F. Maison, PhD, the lead author of a new study on tinnitus, said in a news release about the study.
Tinnitus is sometimes compared to phantom limb syndrome, in which people feel pain in limbs they no longer have. While the study published in Scientific Reports doesn’t refer to phantom limb syndrome, it does talk about “phantom sound.”
“In other words, the brain tries to compensate for the loss of hearing by increasing its activity, resulting in the perception of a phantom sound, tinnitus. Until recently though, this idea was disputed as some tinnitus sufferers have normal hearing tests,” the researchers explained in the news release.
Annoyed by the ringing in your ears? What causes tinnitus, and how can you get the sound to buzz off?
The study included 294 adults — 201 who had never reported having tinnitus, 64 who had reported having temporary tinnitus, and 29 who had reported having constant tinnitus for 6 months or more.
All 294 had performed normally on a pure tone test, in which subjects raise their hands when they hear beeps to measure the quietest sounds they can detect.
In a different kind of test, electrodes measured responses to clicking sounds in the inner ear, the auditory nerve, and the brain. The second test found reduced response in the auditory nerves and increased activity in the brainstem activity among those who had tinnitus.
Dr Maison, a principal investigator at Eaton-Peabody Laboratories at Mass Eye and Ear/Harvard Medical School, called the study “a first step toward our ultimate goal of silencing tinnitus.”
“Beyond the nuisance of having persistent ringing or other sounds in the ears, tinnitus symptoms are debilitating in many patients, causing sleep deprivation, social isolation, anxiety and depression, adversely affecting work performance, and reducing significantly their quality of life,” he said in the news release. “We won’t be able to cure tinnitus until we fully understand the mechanisms underlying its genesis.”
A version of this article appeared on WebMD.com.
Scientists know that tinnitus, or ringing in the ears, affects 10% of adults worldwide. But they’re not exactly sure what causes the condition.
The traditional belief is that tinnitus happens in people who had already lost hearing. But some people who have tinnitus are still able to perform well on standard hearing tests, according to researchers at the Massachusetts Eye and Ear Infirmary. That happens because the tests don’t pick up auditory nerve loss, sometimes called “hidden hearing loss.”
Stéphane F. Maison, PhD, the lead author of a new study on tinnitus, said in a news release about the study.
Tinnitus is sometimes compared to phantom limb syndrome, in which people feel pain in limbs they no longer have. While the study published in Scientific Reports doesn’t refer to phantom limb syndrome, it does talk about “phantom sound.”
“In other words, the brain tries to compensate for the loss of hearing by increasing its activity, resulting in the perception of a phantom sound, tinnitus. Until recently though, this idea was disputed as some tinnitus sufferers have normal hearing tests,” the researchers explained in the news release.
Annoyed by the ringing in your ears? What causes tinnitus, and how can you get the sound to buzz off?
The study included 294 adults — 201 who had never reported having tinnitus, 64 who had reported having temporary tinnitus, and 29 who had reported having constant tinnitus for 6 months or more.
All 294 had performed normally on a pure tone test, in which subjects raise their hands when they hear beeps to measure the quietest sounds they can detect.
In a different kind of test, electrodes measured responses to clicking sounds in the inner ear, the auditory nerve, and the brain. The second test found reduced response in the auditory nerves and increased activity in the brainstem activity among those who had tinnitus.
Dr Maison, a principal investigator at Eaton-Peabody Laboratories at Mass Eye and Ear/Harvard Medical School, called the study “a first step toward our ultimate goal of silencing tinnitus.”
“Beyond the nuisance of having persistent ringing or other sounds in the ears, tinnitus symptoms are debilitating in many patients, causing sleep deprivation, social isolation, anxiety and depression, adversely affecting work performance, and reducing significantly their quality of life,” he said in the news release. “We won’t be able to cure tinnitus until we fully understand the mechanisms underlying its genesis.”
A version of this article appeared on WebMD.com.
FROM SCIENTIFIC REPORTS
Eight wealth tips just for doctors
The average physician makes $352,000, and some earn well into the $500,000s. So, doctors don’t have to worry about money, right?
You know the answer to that.
One thing all physicians have in common about money, says James M. Dahle, MD, FACEP, founder of The White Coat Investor, is that they don’t receive any training in business, personal finance, or investing throughout their schooling or careers unless they seek it out. This leaves many unprepared to make the best investing and money-saving decisions, while others get too frustrated about their lack of knowledge to even dip their toe into the investing pool.
Exhibit A: Four out of 10 physicians have a net worth below $1 million, according to the Medscape Physician Wealth & Debt Report 2023. Elizabeth Chiang, MD, PhD, an oculoplastic surgeon and a physician money coach at Grow Your Wealthy Mindset, notes that many of those doctors are over age 65, “which means they essentially can’t retire.”
And that’s just one pain point.
Physicians have money concerns specific to their profession and background. Luckily, some fellow doctors also serve as financial and wealth advisors just for other doctors.
Blind Spot #1
The early lean years skew doctors’ money outlook. “We have an extended training period, which commonly consists of taking on a large amount of debt, followed by 3 to 8 years of being paid a modest salary, and then finally a large boost in income,” explains Dr. Chiang. This can lay a shaky foundation for the earning years to come, and as a result, a lot of doctors just don’t think about money in healthy ways. Once their incomes increase, physicians may be surprised, for example, that making a multiple six-figure salary means paying six figures in taxes.
The Fix
Treat financial health like physical health. That means money cannot be a taboo subject. “The misguided mindset is that we didn’t become physicians to make money, we did it to help people,” explains Jordan Frey, MD, creator of the blog, The Prudent Plastic Surgeon.
Dr. Frey acknowledges that the desire to help is certainly true. But the result is a false idea that “to think about our personal finances makes us a worse doctor.”
Blind Spot #2
Because doctors know a lot about one thing (medicine), they might assume they know a lot about everything (such as investing). “Totally different fields with a different language and different way to think about it,” Dahle explains. This overconfidence could lead to some negligent or risky financial decisions.
The Fix
Educate yourself. There are several books on personal finance and investing written by physicians for physicians. Dr. Chiang recommends The Physician Philosopher’s Guide to Personal Finance, by James Turner, MD; Financial Freedom Rx, by Chirag Shah, MD, and Jayanth Sridhar, MD; and The Physician’s Guide to Finance, by Nicholas Christian and Amanda Christian, MD. There are also podcasts, blogs, and courses to help educate doctors on finance, such as the Fire Your Financial Advisor course by The White Coat Investor.
Blind Spot #3
Undersaving. Retirement saving is one thing, but 24% of doctors say they don’t even put money away in a taxable savings account, according to the Wealth & Debt Report.
Cobin Soelberg, MD, JD, a board-certified anesthesiologist and founder and principal advisor with Greeley Wealth Management, is the treasurer of his anesthesiology group. “I get to see every month how much people are saving, and even on an anesthesiologist salary, where everyone’s making about $400,000 a year, a lot of people are not saving anything, which is crazy.”
Undersaving can be both a time issue and a mindset one.
Time: Doctors often start investing in their retirement accounts later than the average professional, says Dr. Chiang. “A lot of physicians will max out their 401k or 403b,” she explains. “But if you’re putting in $20,000 a year and only starting when you’re in your early 30s, that’s not enough to get you to retirement.”
Mindset: Doctors also see people of all ages who are sick, dying, and injured. “They all know someone who worked hard and saved and then dropped dead at 55,” explains Dr. Dahle. This, he says, can lead to a bit of a “you only live once” attitude that prioritizes spending over saving.
The Fix
Shoot for 20%. If you can’t save 20% of your gross now, strive to get to that point. Think of it as telling a patient they have to change their behavior or trouble will come - not if, but when. “Develop a written investing plan and then stick with it through thick and thin,” says Dr. Dahle. “Once you have a reasonable plan, all you have to do is fund it adequately by saving 20% of your gross income, and a doctor will easily retire as a multimillionaire.”
Blind Spot #4
Bad investment strategies. Thirty-six percent of doctors experience their largest financial losses from lousy investments, according to the Wealth & Debt Report. Meanwhile, 17% of PCPs and 12% of specialists say they haven’t made any investments at all. That’s a terrible mix of doing the wrong thing and doing a worse thing.
The Fix
Don’t overthink investing, but don’t underthink it either. “As high-income earners, doctors just don’t need to take this high level of risk to reach their financial goals,” Dr. Frey says. A good investment plan doesn’t require you to time the stock market or predict individual stock winners. Consider what Vanguard founder Jack Bogle once said about investing: “Be bored by the process but elated by the outcome.”
Dr. Frey suggests going super-simple: index funds. Ignore investing strategies with actively managed mutual funds or individual stocks, as well as risky alternative investments such as cryptocurrency and angel investments. Everyone assumes doctors have money to burn, and they will push sketchy investment ideas at them. Avoid.
Blind Spot #5
Not taking debt seriously enough. The average medical student debt is $250,000 and can exceed $500,000, says Dr. Soelberg. Many doctors spend the first 10 to 20 years of their careers paying this off. Today’s graduates are paying more than 7% on their loans.
And it’s not just student debt: 39% of physicians carry five or more credit cards, and 34% have mortgages larger than $300,000 (with half of those are more than than $500K), per the Wealth & Debt Report.
The Fix
Treat debt like cancer. It’s a lethal enemy you can’t get rid of right away, but a steady, aggressive, long-term attack will have the best results. Dr. Soelberg suggests allocating the most you can afford per month, whether that’s $1000 or $5000, toward debt. Raise the amount as your income grows. Do the same with your 401k or retirement plan. Whatever is left, you can spend. Five to 10 years later, you will realize, “Wow. I’m debt free.”
Blind Spot #6
Not putting in the work to improve your situation. Seventy-one percent of doctors admit they haven’t done anything to reduce major expenses, according to the Wealth & Debt Report. Are you leaving major money on the table?
The Fix
Audit yourself in major areas like housing and taxes. While the average professional may need to put 10% to 20% down on a home, physicians can qualify for physician mortgage loans and can often put down 3% or less, says Dr. Chiang. If you can afford the higher mortgage payment, excess savings earmarked for a larger down payment can be put toward debt or invested.
Another trick, if you’re able, is to seek an area that is less in demand at a higher salary. “Physicians in places like New York City or San Francisco tend to make less than physicians in the Midwest or the South,” Dr. Chiang explains. A colleague of hers moved to rural Pennsylvania, where he made a high salary and had a low cost of living for 3½ years, paid off his student debt, and then relocated to an area where he wanted to live long term.
As for taxes, become familiar with tax law. Research things like, “What is considered a business expense for doctors?” says Brett Mollard, MD, a diagnostic radiologist who provides financial advice to younger physicians. “What will your estimated total tax burden be at the end of the year? Will you need to make extra payments to prevent owing a large sum of money from underpaying or to avoid tax penalties?”
Blind Spot #7
Living like a rock star on a doctor’s income. Getting caught up in trying to live the same lifestyle as your colleagues is a classic bear trap. “Sitting in the doctor’s lounge, it’s so crazy,” Dr. Soelberg says. He describes conversations like, “‘Where did you go on your trip?’ ‘What new toys are you buying?’” There’s pressure to live up to an image of what a doctor’s life is supposed to look like before you’ve sorted the basic things like paying off debt.
The Fix
Live like a resident even if you haven’t been one for years, at least until you’re in a better financial position. “You’re already used to living a life of lower means, and you’re an expert when it comes to delaying gratification,” says Dr. Mollard. “Do it a little longer.” Live frugally and spend only on things that bring you joy. “A lot of physicians are trying to be really rich in all areas of their life instead of the ones that actually matter to them,” Dr. Soelberg says. Identify what’s important to you and only splurge on that.
Blind Spot #8
Never asking for help. The right financial planner can provide expert help. Emphasis on right. “Doctors can be very trusting of other professionals, even when they should not be,” says Dr. Dahle. He notes that in financial services, many people masquerade as knowledgeable advisors who are really just salespeople. While legitimate financial advisors strive to make their clients money, they are also ultimately out to line their pockets and love to work with physician salaries. Thus, doctors can end up working with financial planners that don’t specifically understand their situations or end up taking too much from their clients.
The Fix
Find a planner who specializes in, or at least understands, physicians. Ask them how they make money, says Dr. Chiang. If someone hesitates to tell you about their fee structure or if it sounds like a lot, shop around and ask colleagues for recommendations.
“Ultimately, the path to wealth is to create and grow the margin between what you make and what you spend,” says Dr. Frey. Throw some investing into the mix and physicians can set themselves up on a path for a stress-free financial life.
A version of this article appeared on Medscape.com.
The average physician makes $352,000, and some earn well into the $500,000s. So, doctors don’t have to worry about money, right?
You know the answer to that.
One thing all physicians have in common about money, says James M. Dahle, MD, FACEP, founder of The White Coat Investor, is that they don’t receive any training in business, personal finance, or investing throughout their schooling or careers unless they seek it out. This leaves many unprepared to make the best investing and money-saving decisions, while others get too frustrated about their lack of knowledge to even dip their toe into the investing pool.
Exhibit A: Four out of 10 physicians have a net worth below $1 million, according to the Medscape Physician Wealth & Debt Report 2023. Elizabeth Chiang, MD, PhD, an oculoplastic surgeon and a physician money coach at Grow Your Wealthy Mindset, notes that many of those doctors are over age 65, “which means they essentially can’t retire.”
And that’s just one pain point.
Physicians have money concerns specific to their profession and background. Luckily, some fellow doctors also serve as financial and wealth advisors just for other doctors.
Blind Spot #1
The early lean years skew doctors’ money outlook. “We have an extended training period, which commonly consists of taking on a large amount of debt, followed by 3 to 8 years of being paid a modest salary, and then finally a large boost in income,” explains Dr. Chiang. This can lay a shaky foundation for the earning years to come, and as a result, a lot of doctors just don’t think about money in healthy ways. Once their incomes increase, physicians may be surprised, for example, that making a multiple six-figure salary means paying six figures in taxes.
The Fix
Treat financial health like physical health. That means money cannot be a taboo subject. “The misguided mindset is that we didn’t become physicians to make money, we did it to help people,” explains Jordan Frey, MD, creator of the blog, The Prudent Plastic Surgeon.
Dr. Frey acknowledges that the desire to help is certainly true. But the result is a false idea that “to think about our personal finances makes us a worse doctor.”
Blind Spot #2
Because doctors know a lot about one thing (medicine), they might assume they know a lot about everything (such as investing). “Totally different fields with a different language and different way to think about it,” Dahle explains. This overconfidence could lead to some negligent or risky financial decisions.
The Fix
Educate yourself. There are several books on personal finance and investing written by physicians for physicians. Dr. Chiang recommends The Physician Philosopher’s Guide to Personal Finance, by James Turner, MD; Financial Freedom Rx, by Chirag Shah, MD, and Jayanth Sridhar, MD; and The Physician’s Guide to Finance, by Nicholas Christian and Amanda Christian, MD. There are also podcasts, blogs, and courses to help educate doctors on finance, such as the Fire Your Financial Advisor course by The White Coat Investor.
Blind Spot #3
Undersaving. Retirement saving is one thing, but 24% of doctors say they don’t even put money away in a taxable savings account, according to the Wealth & Debt Report.
Cobin Soelberg, MD, JD, a board-certified anesthesiologist and founder and principal advisor with Greeley Wealth Management, is the treasurer of his anesthesiology group. “I get to see every month how much people are saving, and even on an anesthesiologist salary, where everyone’s making about $400,000 a year, a lot of people are not saving anything, which is crazy.”
Undersaving can be both a time issue and a mindset one.
Time: Doctors often start investing in their retirement accounts later than the average professional, says Dr. Chiang. “A lot of physicians will max out their 401k or 403b,” she explains. “But if you’re putting in $20,000 a year and only starting when you’re in your early 30s, that’s not enough to get you to retirement.”
Mindset: Doctors also see people of all ages who are sick, dying, and injured. “They all know someone who worked hard and saved and then dropped dead at 55,” explains Dr. Dahle. This, he says, can lead to a bit of a “you only live once” attitude that prioritizes spending over saving.
The Fix
Shoot for 20%. If you can’t save 20% of your gross now, strive to get to that point. Think of it as telling a patient they have to change their behavior or trouble will come - not if, but when. “Develop a written investing plan and then stick with it through thick and thin,” says Dr. Dahle. “Once you have a reasonable plan, all you have to do is fund it adequately by saving 20% of your gross income, and a doctor will easily retire as a multimillionaire.”
Blind Spot #4
Bad investment strategies. Thirty-six percent of doctors experience their largest financial losses from lousy investments, according to the Wealth & Debt Report. Meanwhile, 17% of PCPs and 12% of specialists say they haven’t made any investments at all. That’s a terrible mix of doing the wrong thing and doing a worse thing.
The Fix
Don’t overthink investing, but don’t underthink it either. “As high-income earners, doctors just don’t need to take this high level of risk to reach their financial goals,” Dr. Frey says. A good investment plan doesn’t require you to time the stock market or predict individual stock winners. Consider what Vanguard founder Jack Bogle once said about investing: “Be bored by the process but elated by the outcome.”
Dr. Frey suggests going super-simple: index funds. Ignore investing strategies with actively managed mutual funds or individual stocks, as well as risky alternative investments such as cryptocurrency and angel investments. Everyone assumes doctors have money to burn, and they will push sketchy investment ideas at them. Avoid.
Blind Spot #5
Not taking debt seriously enough. The average medical student debt is $250,000 and can exceed $500,000, says Dr. Soelberg. Many doctors spend the first 10 to 20 years of their careers paying this off. Today’s graduates are paying more than 7% on their loans.
And it’s not just student debt: 39% of physicians carry five or more credit cards, and 34% have mortgages larger than $300,000 (with half of those are more than than $500K), per the Wealth & Debt Report.
The Fix
Treat debt like cancer. It’s a lethal enemy you can’t get rid of right away, but a steady, aggressive, long-term attack will have the best results. Dr. Soelberg suggests allocating the most you can afford per month, whether that’s $1000 or $5000, toward debt. Raise the amount as your income grows. Do the same with your 401k or retirement plan. Whatever is left, you can spend. Five to 10 years later, you will realize, “Wow. I’m debt free.”
Blind Spot #6
Not putting in the work to improve your situation. Seventy-one percent of doctors admit they haven’t done anything to reduce major expenses, according to the Wealth & Debt Report. Are you leaving major money on the table?
The Fix
Audit yourself in major areas like housing and taxes. While the average professional may need to put 10% to 20% down on a home, physicians can qualify for physician mortgage loans and can often put down 3% or less, says Dr. Chiang. If you can afford the higher mortgage payment, excess savings earmarked for a larger down payment can be put toward debt or invested.
Another trick, if you’re able, is to seek an area that is less in demand at a higher salary. “Physicians in places like New York City or San Francisco tend to make less than physicians in the Midwest or the South,” Dr. Chiang explains. A colleague of hers moved to rural Pennsylvania, where he made a high salary and had a low cost of living for 3½ years, paid off his student debt, and then relocated to an area where he wanted to live long term.
As for taxes, become familiar with tax law. Research things like, “What is considered a business expense for doctors?” says Brett Mollard, MD, a diagnostic radiologist who provides financial advice to younger physicians. “What will your estimated total tax burden be at the end of the year? Will you need to make extra payments to prevent owing a large sum of money from underpaying or to avoid tax penalties?”
Blind Spot #7
Living like a rock star on a doctor’s income. Getting caught up in trying to live the same lifestyle as your colleagues is a classic bear trap. “Sitting in the doctor’s lounge, it’s so crazy,” Dr. Soelberg says. He describes conversations like, “‘Where did you go on your trip?’ ‘What new toys are you buying?’” There’s pressure to live up to an image of what a doctor’s life is supposed to look like before you’ve sorted the basic things like paying off debt.
The Fix
Live like a resident even if you haven’t been one for years, at least until you’re in a better financial position. “You’re already used to living a life of lower means, and you’re an expert when it comes to delaying gratification,” says Dr. Mollard. “Do it a little longer.” Live frugally and spend only on things that bring you joy. “A lot of physicians are trying to be really rich in all areas of their life instead of the ones that actually matter to them,” Dr. Soelberg says. Identify what’s important to you and only splurge on that.
Blind Spot #8
Never asking for help. The right financial planner can provide expert help. Emphasis on right. “Doctors can be very trusting of other professionals, even when they should not be,” says Dr. Dahle. He notes that in financial services, many people masquerade as knowledgeable advisors who are really just salespeople. While legitimate financial advisors strive to make their clients money, they are also ultimately out to line their pockets and love to work with physician salaries. Thus, doctors can end up working with financial planners that don’t specifically understand their situations or end up taking too much from their clients.
The Fix
Find a planner who specializes in, or at least understands, physicians. Ask them how they make money, says Dr. Chiang. If someone hesitates to tell you about their fee structure or if it sounds like a lot, shop around and ask colleagues for recommendations.
“Ultimately, the path to wealth is to create and grow the margin between what you make and what you spend,” says Dr. Frey. Throw some investing into the mix and physicians can set themselves up on a path for a stress-free financial life.
A version of this article appeared on Medscape.com.
The average physician makes $352,000, and some earn well into the $500,000s. So, doctors don’t have to worry about money, right?
You know the answer to that.
One thing all physicians have in common about money, says James M. Dahle, MD, FACEP, founder of The White Coat Investor, is that they don’t receive any training in business, personal finance, or investing throughout their schooling or careers unless they seek it out. This leaves many unprepared to make the best investing and money-saving decisions, while others get too frustrated about their lack of knowledge to even dip their toe into the investing pool.
Exhibit A: Four out of 10 physicians have a net worth below $1 million, according to the Medscape Physician Wealth & Debt Report 2023. Elizabeth Chiang, MD, PhD, an oculoplastic surgeon and a physician money coach at Grow Your Wealthy Mindset, notes that many of those doctors are over age 65, “which means they essentially can’t retire.”
And that’s just one pain point.
Physicians have money concerns specific to their profession and background. Luckily, some fellow doctors also serve as financial and wealth advisors just for other doctors.
Blind Spot #1
The early lean years skew doctors’ money outlook. “We have an extended training period, which commonly consists of taking on a large amount of debt, followed by 3 to 8 years of being paid a modest salary, and then finally a large boost in income,” explains Dr. Chiang. This can lay a shaky foundation for the earning years to come, and as a result, a lot of doctors just don’t think about money in healthy ways. Once their incomes increase, physicians may be surprised, for example, that making a multiple six-figure salary means paying six figures in taxes.
The Fix
Treat financial health like physical health. That means money cannot be a taboo subject. “The misguided mindset is that we didn’t become physicians to make money, we did it to help people,” explains Jordan Frey, MD, creator of the blog, The Prudent Plastic Surgeon.
Dr. Frey acknowledges that the desire to help is certainly true. But the result is a false idea that “to think about our personal finances makes us a worse doctor.”
Blind Spot #2
Because doctors know a lot about one thing (medicine), they might assume they know a lot about everything (such as investing). “Totally different fields with a different language and different way to think about it,” Dahle explains. This overconfidence could lead to some negligent or risky financial decisions.
The Fix
Educate yourself. There are several books on personal finance and investing written by physicians for physicians. Dr. Chiang recommends The Physician Philosopher’s Guide to Personal Finance, by James Turner, MD; Financial Freedom Rx, by Chirag Shah, MD, and Jayanth Sridhar, MD; and The Physician’s Guide to Finance, by Nicholas Christian and Amanda Christian, MD. There are also podcasts, blogs, and courses to help educate doctors on finance, such as the Fire Your Financial Advisor course by The White Coat Investor.
Blind Spot #3
Undersaving. Retirement saving is one thing, but 24% of doctors say they don’t even put money away in a taxable savings account, according to the Wealth & Debt Report.
Cobin Soelberg, MD, JD, a board-certified anesthesiologist and founder and principal advisor with Greeley Wealth Management, is the treasurer of his anesthesiology group. “I get to see every month how much people are saving, and even on an anesthesiologist salary, where everyone’s making about $400,000 a year, a lot of people are not saving anything, which is crazy.”
Undersaving can be both a time issue and a mindset one.
Time: Doctors often start investing in their retirement accounts later than the average professional, says Dr. Chiang. “A lot of physicians will max out their 401k or 403b,” she explains. “But if you’re putting in $20,000 a year and only starting when you’re in your early 30s, that’s not enough to get you to retirement.”
Mindset: Doctors also see people of all ages who are sick, dying, and injured. “They all know someone who worked hard and saved and then dropped dead at 55,” explains Dr. Dahle. This, he says, can lead to a bit of a “you only live once” attitude that prioritizes spending over saving.
The Fix
Shoot for 20%. If you can’t save 20% of your gross now, strive to get to that point. Think of it as telling a patient they have to change their behavior or trouble will come - not if, but when. “Develop a written investing plan and then stick with it through thick and thin,” says Dr. Dahle. “Once you have a reasonable plan, all you have to do is fund it adequately by saving 20% of your gross income, and a doctor will easily retire as a multimillionaire.”
Blind Spot #4
Bad investment strategies. Thirty-six percent of doctors experience their largest financial losses from lousy investments, according to the Wealth & Debt Report. Meanwhile, 17% of PCPs and 12% of specialists say they haven’t made any investments at all. That’s a terrible mix of doing the wrong thing and doing a worse thing.
The Fix
Don’t overthink investing, but don’t underthink it either. “As high-income earners, doctors just don’t need to take this high level of risk to reach their financial goals,” Dr. Frey says. A good investment plan doesn’t require you to time the stock market or predict individual stock winners. Consider what Vanguard founder Jack Bogle once said about investing: “Be bored by the process but elated by the outcome.”
Dr. Frey suggests going super-simple: index funds. Ignore investing strategies with actively managed mutual funds or individual stocks, as well as risky alternative investments such as cryptocurrency and angel investments. Everyone assumes doctors have money to burn, and they will push sketchy investment ideas at them. Avoid.
Blind Spot #5
Not taking debt seriously enough. The average medical student debt is $250,000 and can exceed $500,000, says Dr. Soelberg. Many doctors spend the first 10 to 20 years of their careers paying this off. Today’s graduates are paying more than 7% on their loans.
And it’s not just student debt: 39% of physicians carry five or more credit cards, and 34% have mortgages larger than $300,000 (with half of those are more than than $500K), per the Wealth & Debt Report.
The Fix
Treat debt like cancer. It’s a lethal enemy you can’t get rid of right away, but a steady, aggressive, long-term attack will have the best results. Dr. Soelberg suggests allocating the most you can afford per month, whether that’s $1000 or $5000, toward debt. Raise the amount as your income grows. Do the same with your 401k or retirement plan. Whatever is left, you can spend. Five to 10 years later, you will realize, “Wow. I’m debt free.”
Blind Spot #6
Not putting in the work to improve your situation. Seventy-one percent of doctors admit they haven’t done anything to reduce major expenses, according to the Wealth & Debt Report. Are you leaving major money on the table?
The Fix
Audit yourself in major areas like housing and taxes. While the average professional may need to put 10% to 20% down on a home, physicians can qualify for physician mortgage loans and can often put down 3% or less, says Dr. Chiang. If you can afford the higher mortgage payment, excess savings earmarked for a larger down payment can be put toward debt or invested.
Another trick, if you’re able, is to seek an area that is less in demand at a higher salary. “Physicians in places like New York City or San Francisco tend to make less than physicians in the Midwest or the South,” Dr. Chiang explains. A colleague of hers moved to rural Pennsylvania, where he made a high salary and had a low cost of living for 3½ years, paid off his student debt, and then relocated to an area where he wanted to live long term.
As for taxes, become familiar with tax law. Research things like, “What is considered a business expense for doctors?” says Brett Mollard, MD, a diagnostic radiologist who provides financial advice to younger physicians. “What will your estimated total tax burden be at the end of the year? Will you need to make extra payments to prevent owing a large sum of money from underpaying or to avoid tax penalties?”
Blind Spot #7
Living like a rock star on a doctor’s income. Getting caught up in trying to live the same lifestyle as your colleagues is a classic bear trap. “Sitting in the doctor’s lounge, it’s so crazy,” Dr. Soelberg says. He describes conversations like, “‘Where did you go on your trip?’ ‘What new toys are you buying?’” There’s pressure to live up to an image of what a doctor’s life is supposed to look like before you’ve sorted the basic things like paying off debt.
The Fix
Live like a resident even if you haven’t been one for years, at least until you’re in a better financial position. “You’re already used to living a life of lower means, and you’re an expert when it comes to delaying gratification,” says Dr. Mollard. “Do it a little longer.” Live frugally and spend only on things that bring you joy. “A lot of physicians are trying to be really rich in all areas of their life instead of the ones that actually matter to them,” Dr. Soelberg says. Identify what’s important to you and only splurge on that.
Blind Spot #8
Never asking for help. The right financial planner can provide expert help. Emphasis on right. “Doctors can be very trusting of other professionals, even when they should not be,” says Dr. Dahle. He notes that in financial services, many people masquerade as knowledgeable advisors who are really just salespeople. While legitimate financial advisors strive to make their clients money, they are also ultimately out to line their pockets and love to work with physician salaries. Thus, doctors can end up working with financial planners that don’t specifically understand their situations or end up taking too much from their clients.
The Fix
Find a planner who specializes in, or at least understands, physicians. Ask them how they make money, says Dr. Chiang. If someone hesitates to tell you about their fee structure or if it sounds like a lot, shop around and ask colleagues for recommendations.
“Ultimately, the path to wealth is to create and grow the margin between what you make and what you spend,” says Dr. Frey. Throw some investing into the mix and physicians can set themselves up on a path for a stress-free financial life.
A version of this article appeared on Medscape.com.