User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
How PCPs are penalized for positive outcomes from lifestyle change
The Centers for Medicare & Medicaid Services 2022 National Quality Strategy is described as an “ambitious long-term initiative that aims to promote the highest quality outcomes and safest care for all individuals.” It is a commendable goal for an overburdened U.S. health care system that spends more than other high-income counties yet experiences poorer outcomes. But whole-person, person-centered care cannot be achieved under current misaligned quality measures that fail to measure what we purport to value: the quintuple aim of improved health outcomes, cost savings, patient satisfaction, clinician well-being, and health equity.
Lifestyle first
Clinical practice guidelines for many chronic diseases recommend lifestyle intervention as the first and optimal treatment. A growing body of evidence supports lifestyle behavior interventions to treat and, when used intensively, even reverse common chronic conditions such as cardiovascular disease, obesity, and type 2 diabetes, while also providing effective prevention for those conditions. However, no current quality measures consider lifestyle interventions. In fact, some quality measures unintentionally penalize physicians for successfully treating or reversing disease through lifestyle behavior interventions while rewarding clinicians for meeting process measures – usually adherence to medication – regardless of whether health outcomes improved.
Rewarding medication adherence for the treatment of diseases in which lifestyle is a primary therapy (such as hypertension), combined with other health care constraints (lack of lifestyle education, time to spend with patients, and infrastructure support) incentivizes physicians to skip the conversation about lifestyle changes and go straight to medication prescription. Meanwhile, the clinician who takes the extra time to guide a patient toward lifestyle interventions that could treat their current disease and prevent future diseases – without side effects – is penalized.
Misaligned quality measures like these can stifle clinical judgment and risk reducing the practice of medicine to mindless box-checking. In many cases, patients are not even informed that lifestyle behavior change may be a treatment option (much less the first recommended option) for their conditions. This delivery of care is not person-centered and, in fact, may raise questions about the adequacy of informed treatment consent.
Reimbursement barriers
Lifestyle medicine is a growing medical specialty that uses therapeutic lifestyle interventions as a primary modality to treat chronic conditions. Since certification began in 2017, almost 2500 US physicians and 1000 nonphysician health professionals have earned certification. Health systems, including the U.S. military, are increasingly integrating lifestyle medicine. There have been advancements since one survey found that more than half of lifestyle medicine clinicians reported receiving no reimbursement for lifestyle behavior interventions. However, barriers, especially in fee-for-service systems, still inhibit many patients from receiving insurance coverage for comprehensive, interdisciplinary, and whole-person treatments called intensive therapeutic lifestyle change (ITLC) programs.
Existing comprehensive lifestyle programs that patients are eligible for (ie, the Diabetes Prevention Program and intensive behavioral therapy) are often so poorly reimbursed that clinicians and health systems decline to offer them. An example of a well-reimbursed ITLC program is intensive cardiac rehabilitation (ICR), which remains underutilized and limited to a narrow segment of patients, despite ICR›s proven benefits for managing comorbid risk factors such as hemoglobin A1c and weight. Even when lifestyle intervention programs are available and patients are eligible to participate (often through shared medical appointments), patient copays for the frequent visits required to achieve and sustain behavior change – or the lack of reimbursement for interdisciplinary team members – discourage engagement.
Penalizing successful outcomes
Despite the fact that lifestyle behaviors are top contributors to health and, conversely, contribute to up to 80% of chronic diseases, few quality measures focus on screening for lifestyle factors or treating diseases with lifestyle interventions. An example of an existing quality measure is screening or treatment for harmful substance use.
Specific quality measures that penalize lifestyle medicine approaches include pharmacotherapy for type 2 diabetes, dyslipidemia, osteoporosis, and gout as well as approaches to rheumatoid arthritis.
Statins offer a useful example of the conundrum faced by clinicians who want to offer lifestyle interventions. A lifestyle medicine primary care physician had a patient covered by Medicare Advantage who was diagnosed with hyperlipidemia. The patient had total cholesterol of 226 and a triglycerides level of 132. Instead of prescribing the routine statin, the physician prescribed lifestyle behavior modifications. Within 3 weeks, the patient›s total cholesterol improved to 171 and triglycerides to 75. This was a great success for the delighted patient. However, the CMS 5-Star Rating System assigned the primary care physician a grade of C rather than A, which put the physician›s 5-star rating at risk. Why? Because the system bases its score largely on medication compliance. The physician was penalized despite achieving the optimal health outcome, and at a lower cost than with medication. This misalignment does not incentivize patient-centered care because it disregards patient preference, shared decision-making, and evidence-based practice.
Risk adjustment
Rather than automatically managing disease with ever-increasing quantities of costly medications and procedures, lifestyle medicine clinicians first pursue a goal of health restoration when appropriate. But Medicare risk adjustment incentivizes physicians to manage rather than reverse disease. How much Medicare pays health plans is determined in part by how sick the patients are; the sicker the patient, the more Medicare pays, because those patients› costs are expected to be higher. This ensures that health plans are not penalized for enrolling sicker patients. But a physician utilizing diet alone to achieve remission in a patient with type 2 diabetes is penalized financially because, when the risk is adjusted, diabetes is no longer listed among the patient›s conditions. So, Medicare pays the physician less money. That misalignment incentivizes clinicians to manage the symptoms of type 2 diabetes rather than achieve remission, despite remission being the ideal clinical outcome.
Realigning quality measures
Quality measures were developed to quantify health care processes and outcomes, and to ensure the delivery of safe care to all patients. However, over time the number of quality measures has swelled to 2500, evolving into a confusing, time-consuming, and even soul-crushing responsibility for the physician.
Instead of relying heavily on process measures, we must incentivize outcome measures that honor patient autonomy and allow clinicians to offer lifestyle intervention as the first line of treatment. Risk-score calculations should be adjusted so that we stop incentivizing disease management and penalizing disease reversal.
CMS’s proposed development of “a universal foundation” of quality measures is an opportunity to begin the realignment of quality measures and values. This foundation is intended to establish more consistent and meaningful measures, reduce clinician burnout by streamlining the reporting process, and advance health equity. For this change to be successful, it is vital that lifestyle behavior interventions – optimal nutrition, physical activity, restorative sleep, social connections, stress management, and avoidance of harmful substances – become the foundation of universal quality measures. This will ensure that every clinician is incentivized to discuss lifestyle behaviors with patients and pursue the first clinical step recommended by clinical practice guidelines for most chronic diseases. Only then can we truly deliver high-value, whole-person, person-centered care and achieve the quintuple aim.
Dr. Patel is president-elect, American College of Lifestyle Medicine; Lifestyle Medicine Medical Director, Wellvana Health, Midland, Tex. She has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The Centers for Medicare & Medicaid Services 2022 National Quality Strategy is described as an “ambitious long-term initiative that aims to promote the highest quality outcomes and safest care for all individuals.” It is a commendable goal for an overburdened U.S. health care system that spends more than other high-income counties yet experiences poorer outcomes. But whole-person, person-centered care cannot be achieved under current misaligned quality measures that fail to measure what we purport to value: the quintuple aim of improved health outcomes, cost savings, patient satisfaction, clinician well-being, and health equity.
Lifestyle first
Clinical practice guidelines for many chronic diseases recommend lifestyle intervention as the first and optimal treatment. A growing body of evidence supports lifestyle behavior interventions to treat and, when used intensively, even reverse common chronic conditions such as cardiovascular disease, obesity, and type 2 diabetes, while also providing effective prevention for those conditions. However, no current quality measures consider lifestyle interventions. In fact, some quality measures unintentionally penalize physicians for successfully treating or reversing disease through lifestyle behavior interventions while rewarding clinicians for meeting process measures – usually adherence to medication – regardless of whether health outcomes improved.
Rewarding medication adherence for the treatment of diseases in which lifestyle is a primary therapy (such as hypertension), combined with other health care constraints (lack of lifestyle education, time to spend with patients, and infrastructure support) incentivizes physicians to skip the conversation about lifestyle changes and go straight to medication prescription. Meanwhile, the clinician who takes the extra time to guide a patient toward lifestyle interventions that could treat their current disease and prevent future diseases – without side effects – is penalized.
Misaligned quality measures like these can stifle clinical judgment and risk reducing the practice of medicine to mindless box-checking. In many cases, patients are not even informed that lifestyle behavior change may be a treatment option (much less the first recommended option) for their conditions. This delivery of care is not person-centered and, in fact, may raise questions about the adequacy of informed treatment consent.
Reimbursement barriers
Lifestyle medicine is a growing medical specialty that uses therapeutic lifestyle interventions as a primary modality to treat chronic conditions. Since certification began in 2017, almost 2500 US physicians and 1000 nonphysician health professionals have earned certification. Health systems, including the U.S. military, are increasingly integrating lifestyle medicine. There have been advancements since one survey found that more than half of lifestyle medicine clinicians reported receiving no reimbursement for lifestyle behavior interventions. However, barriers, especially in fee-for-service systems, still inhibit many patients from receiving insurance coverage for comprehensive, interdisciplinary, and whole-person treatments called intensive therapeutic lifestyle change (ITLC) programs.
Existing comprehensive lifestyle programs that patients are eligible for (ie, the Diabetes Prevention Program and intensive behavioral therapy) are often so poorly reimbursed that clinicians and health systems decline to offer them. An example of a well-reimbursed ITLC program is intensive cardiac rehabilitation (ICR), which remains underutilized and limited to a narrow segment of patients, despite ICR›s proven benefits for managing comorbid risk factors such as hemoglobin A1c and weight. Even when lifestyle intervention programs are available and patients are eligible to participate (often through shared medical appointments), patient copays for the frequent visits required to achieve and sustain behavior change – or the lack of reimbursement for interdisciplinary team members – discourage engagement.
Penalizing successful outcomes
Despite the fact that lifestyle behaviors are top contributors to health and, conversely, contribute to up to 80% of chronic diseases, few quality measures focus on screening for lifestyle factors or treating diseases with lifestyle interventions. An example of an existing quality measure is screening or treatment for harmful substance use.
Specific quality measures that penalize lifestyle medicine approaches include pharmacotherapy for type 2 diabetes, dyslipidemia, osteoporosis, and gout as well as approaches to rheumatoid arthritis.
Statins offer a useful example of the conundrum faced by clinicians who want to offer lifestyle interventions. A lifestyle medicine primary care physician had a patient covered by Medicare Advantage who was diagnosed with hyperlipidemia. The patient had total cholesterol of 226 and a triglycerides level of 132. Instead of prescribing the routine statin, the physician prescribed lifestyle behavior modifications. Within 3 weeks, the patient›s total cholesterol improved to 171 and triglycerides to 75. This was a great success for the delighted patient. However, the CMS 5-Star Rating System assigned the primary care physician a grade of C rather than A, which put the physician›s 5-star rating at risk. Why? Because the system bases its score largely on medication compliance. The physician was penalized despite achieving the optimal health outcome, and at a lower cost than with medication. This misalignment does not incentivize patient-centered care because it disregards patient preference, shared decision-making, and evidence-based practice.
Risk adjustment
Rather than automatically managing disease with ever-increasing quantities of costly medications and procedures, lifestyle medicine clinicians first pursue a goal of health restoration when appropriate. But Medicare risk adjustment incentivizes physicians to manage rather than reverse disease. How much Medicare pays health plans is determined in part by how sick the patients are; the sicker the patient, the more Medicare pays, because those patients› costs are expected to be higher. This ensures that health plans are not penalized for enrolling sicker patients. But a physician utilizing diet alone to achieve remission in a patient with type 2 diabetes is penalized financially because, when the risk is adjusted, diabetes is no longer listed among the patient›s conditions. So, Medicare pays the physician less money. That misalignment incentivizes clinicians to manage the symptoms of type 2 diabetes rather than achieve remission, despite remission being the ideal clinical outcome.
Realigning quality measures
Quality measures were developed to quantify health care processes and outcomes, and to ensure the delivery of safe care to all patients. However, over time the number of quality measures has swelled to 2500, evolving into a confusing, time-consuming, and even soul-crushing responsibility for the physician.
Instead of relying heavily on process measures, we must incentivize outcome measures that honor patient autonomy and allow clinicians to offer lifestyle intervention as the first line of treatment. Risk-score calculations should be adjusted so that we stop incentivizing disease management and penalizing disease reversal.
CMS’s proposed development of “a universal foundation” of quality measures is an opportunity to begin the realignment of quality measures and values. This foundation is intended to establish more consistent and meaningful measures, reduce clinician burnout by streamlining the reporting process, and advance health equity. For this change to be successful, it is vital that lifestyle behavior interventions – optimal nutrition, physical activity, restorative sleep, social connections, stress management, and avoidance of harmful substances – become the foundation of universal quality measures. This will ensure that every clinician is incentivized to discuss lifestyle behaviors with patients and pursue the first clinical step recommended by clinical practice guidelines for most chronic diseases. Only then can we truly deliver high-value, whole-person, person-centered care and achieve the quintuple aim.
Dr. Patel is president-elect, American College of Lifestyle Medicine; Lifestyle Medicine Medical Director, Wellvana Health, Midland, Tex. She has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The Centers for Medicare & Medicaid Services 2022 National Quality Strategy is described as an “ambitious long-term initiative that aims to promote the highest quality outcomes and safest care for all individuals.” It is a commendable goal for an overburdened U.S. health care system that spends more than other high-income counties yet experiences poorer outcomes. But whole-person, person-centered care cannot be achieved under current misaligned quality measures that fail to measure what we purport to value: the quintuple aim of improved health outcomes, cost savings, patient satisfaction, clinician well-being, and health equity.
Lifestyle first
Clinical practice guidelines for many chronic diseases recommend lifestyle intervention as the first and optimal treatment. A growing body of evidence supports lifestyle behavior interventions to treat and, when used intensively, even reverse common chronic conditions such as cardiovascular disease, obesity, and type 2 diabetes, while also providing effective prevention for those conditions. However, no current quality measures consider lifestyle interventions. In fact, some quality measures unintentionally penalize physicians for successfully treating or reversing disease through lifestyle behavior interventions while rewarding clinicians for meeting process measures – usually adherence to medication – regardless of whether health outcomes improved.
Rewarding medication adherence for the treatment of diseases in which lifestyle is a primary therapy (such as hypertension), combined with other health care constraints (lack of lifestyle education, time to spend with patients, and infrastructure support) incentivizes physicians to skip the conversation about lifestyle changes and go straight to medication prescription. Meanwhile, the clinician who takes the extra time to guide a patient toward lifestyle interventions that could treat their current disease and prevent future diseases – without side effects – is penalized.
Misaligned quality measures like these can stifle clinical judgment and risk reducing the practice of medicine to mindless box-checking. In many cases, patients are not even informed that lifestyle behavior change may be a treatment option (much less the first recommended option) for their conditions. This delivery of care is not person-centered and, in fact, may raise questions about the adequacy of informed treatment consent.
Reimbursement barriers
Lifestyle medicine is a growing medical specialty that uses therapeutic lifestyle interventions as a primary modality to treat chronic conditions. Since certification began in 2017, almost 2500 US physicians and 1000 nonphysician health professionals have earned certification. Health systems, including the U.S. military, are increasingly integrating lifestyle medicine. There have been advancements since one survey found that more than half of lifestyle medicine clinicians reported receiving no reimbursement for lifestyle behavior interventions. However, barriers, especially in fee-for-service systems, still inhibit many patients from receiving insurance coverage for comprehensive, interdisciplinary, and whole-person treatments called intensive therapeutic lifestyle change (ITLC) programs.
Existing comprehensive lifestyle programs that patients are eligible for (ie, the Diabetes Prevention Program and intensive behavioral therapy) are often so poorly reimbursed that clinicians and health systems decline to offer them. An example of a well-reimbursed ITLC program is intensive cardiac rehabilitation (ICR), which remains underutilized and limited to a narrow segment of patients, despite ICR›s proven benefits for managing comorbid risk factors such as hemoglobin A1c and weight. Even when lifestyle intervention programs are available and patients are eligible to participate (often through shared medical appointments), patient copays for the frequent visits required to achieve and sustain behavior change – or the lack of reimbursement for interdisciplinary team members – discourage engagement.
Penalizing successful outcomes
Despite the fact that lifestyle behaviors are top contributors to health and, conversely, contribute to up to 80% of chronic diseases, few quality measures focus on screening for lifestyle factors or treating diseases with lifestyle interventions. An example of an existing quality measure is screening or treatment for harmful substance use.
Specific quality measures that penalize lifestyle medicine approaches include pharmacotherapy for type 2 diabetes, dyslipidemia, osteoporosis, and gout as well as approaches to rheumatoid arthritis.
Statins offer a useful example of the conundrum faced by clinicians who want to offer lifestyle interventions. A lifestyle medicine primary care physician had a patient covered by Medicare Advantage who was diagnosed with hyperlipidemia. The patient had total cholesterol of 226 and a triglycerides level of 132. Instead of prescribing the routine statin, the physician prescribed lifestyle behavior modifications. Within 3 weeks, the patient›s total cholesterol improved to 171 and triglycerides to 75. This was a great success for the delighted patient. However, the CMS 5-Star Rating System assigned the primary care physician a grade of C rather than A, which put the physician›s 5-star rating at risk. Why? Because the system bases its score largely on medication compliance. The physician was penalized despite achieving the optimal health outcome, and at a lower cost than with medication. This misalignment does not incentivize patient-centered care because it disregards patient preference, shared decision-making, and evidence-based practice.
Risk adjustment
Rather than automatically managing disease with ever-increasing quantities of costly medications and procedures, lifestyle medicine clinicians first pursue a goal of health restoration when appropriate. But Medicare risk adjustment incentivizes physicians to manage rather than reverse disease. How much Medicare pays health plans is determined in part by how sick the patients are; the sicker the patient, the more Medicare pays, because those patients› costs are expected to be higher. This ensures that health plans are not penalized for enrolling sicker patients. But a physician utilizing diet alone to achieve remission in a patient with type 2 diabetes is penalized financially because, when the risk is adjusted, diabetes is no longer listed among the patient›s conditions. So, Medicare pays the physician less money. That misalignment incentivizes clinicians to manage the symptoms of type 2 diabetes rather than achieve remission, despite remission being the ideal clinical outcome.
Realigning quality measures
Quality measures were developed to quantify health care processes and outcomes, and to ensure the delivery of safe care to all patients. However, over time the number of quality measures has swelled to 2500, evolving into a confusing, time-consuming, and even soul-crushing responsibility for the physician.
Instead of relying heavily on process measures, we must incentivize outcome measures that honor patient autonomy and allow clinicians to offer lifestyle intervention as the first line of treatment. Risk-score calculations should be adjusted so that we stop incentivizing disease management and penalizing disease reversal.
CMS’s proposed development of “a universal foundation” of quality measures is an opportunity to begin the realignment of quality measures and values. This foundation is intended to establish more consistent and meaningful measures, reduce clinician burnout by streamlining the reporting process, and advance health equity. For this change to be successful, it is vital that lifestyle behavior interventions – optimal nutrition, physical activity, restorative sleep, social connections, stress management, and avoidance of harmful substances – become the foundation of universal quality measures. This will ensure that every clinician is incentivized to discuss lifestyle behaviors with patients and pursue the first clinical step recommended by clinical practice guidelines for most chronic diseases. Only then can we truly deliver high-value, whole-person, person-centered care and achieve the quintuple aim.
Dr. Patel is president-elect, American College of Lifestyle Medicine; Lifestyle Medicine Medical Director, Wellvana Health, Midland, Tex. She has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Bone degradation measure can sway osteoporosis diagnosis
Assessing a key aspect of bone architecture, for which clinicians can now be reimbursed under Medicare, can significantly improve the ability to predict a patient’s risk for bone fracture.
Although bone mineral density (BMD) is traditionally used to identify patients with osteoporosis or low bone mass, some physicians have begun incorporating the trabecular bone score (TBS) into their exams.
At the Cleveland Clinic Center for Specialized Women’s Health, factoring in the TBS changed the diagnosis for 16% of 432 patients, according to Holly Thacker, MD, the center’s director.
“Importantly, 11% got worse diagnoses, and I use that in terms of prioritizing treatment,” Dr. Thacker said in an interview. The ability to determine how degraded the bone microarchitecture is through a software system “is a huge advance.”
Dr. Thacker described her center’s experience with the technology at the annual meeting of the Menopause Society (formerly The North American Menopause Society).
While BMD captures the amount of minerals like calcium in the skeleton, TBS assesses the underlying microarchitecture by looking at the distribution of shades of gray on dual-energy x-ray absorptiometry (DXA) scans.
Based on the TBS, patients’ bones are classified as normal, partially degraded, or degraded. Among the 432 patients who received a TBS analysis in 2022, 3% shifted from a normal diagnosis to osteopenia, 8% worsened from osteopenia to osteoporosis, 4% went from osteopenia to normal, and 1.6% downgraded from osteoporosis to osteopenia, Dr. Thacker reported.
The new test may also provide some reassurance for female patients who have thinner bones, which may raise alarms based on BMD. TBS, however, may show that the structure of the bone looks normal.
“When you know that the microarchitecture is normal, you’re a lot less concerned that they actually have a bone disease of osteoporosis,” Dr. Thacker said.
Conversely, unexpectedly degraded bone raises questions.
“That makes you go back and say [to the patient]: ‘Have you been on steroids? Were you malnourished? Is there some other metabolic problem? Have you had some calcium disorder?’ ” Dr. Thacker said.
Dr. Thacker leverages the TBS to help patients obtain effective therapy, typically an anabolic agent followed by antiresorptive medication.
“When I see a patient who not only has osteoporosis on bone density but has completely degraded bone architecture, it’s a lot easier for me to make the argument to the insurance company that this patient is at grave risk for a low trauma fracture and bad outcome without the best treatment,” Dr. Thacker said.
10-year-old tech, recently covered
The Food and Drug Administration approved TBS software in 2012, but Medicare only recently started paying for it.
Medimaps Group, a company that markets imaging software to calculate TBS, announced in 2022 that reimbursement from the Centers for Medicare & Medicaid Services was available, at $41.53 on the Physician Fee Schedule and $82.61 on the Hospital Outpatient Prospective Payment Schedule.
“Reimbursement through CMS is an important endorsement of the clinical value of TBS for clinicians and their patients,” Didier Hans, PhD, MBA, the CEO of Medimaps, said in a statement at the time. He noted that more than 600,000 TBS procedures were being performed in the United States each year.
Nevertheless, the initial investment in purchasing the software may be a barrier for health systems.
“We are the first and only site in our health system to offer TBS, as this is an extra expense and not uniformly reimbursed by insurers,” Dr. Thacker reported at the meeting.
Potential drawbacks
The TBS software used in Dr. Thacker’s study has been validated only in Asian and White patients between certain ages and weights, meaning the system is not designed to be used for other populations. Other researchers have highlighted a need for trabecular bone scoring to be validated more broadly. The authors of a recent analysis, however, suggest that TBS can be used the same way no matter a patient’s race.
TBS “is going to be most helpful in those with osteopenia who are right near the threshold for treatment,” said Marcella Donovan Walker, MD, MS, in a presentation on bone quality at the meeting.
Many studies have shown that TBS “provides additive information to bone density,” said Dr. Walker, a professor of medicine in the division of endocrinology at Columbia University, New York. For example, a large study of women in Manitoba found that, regardless of whether their bone density was normal, osteopenic, or osteoporotic, those with a low TBS had a much higher risk for fracture.
‘Opportunistic screening’ with CT?
TBS relies on the same DXA scans that are used to calculate bone mineral density, so obtaining the score does not add time or radiation to the scanning process, Dr. Thacker said.
But many patients who should receive DXA scans do not, which adds to the promise of “opportunistic screening” for osteoporosis, Dr. Walker said. With this approach, physicians would analyze a CT scan that a patient received for another purpose, such as to investigate abdominal pain or chest pain.
“In these images is information about the bone,” Dr. Walker said.
Researchers have used high-resolution peripheral quantitative CT to perform finite element analysis, where a computer program simulates compression of the bone to create a measure of bone stiffness and determine the load required for a break.
One study found that including those elements predicted fractures better than bone mineral density or the Fracture Risk Assessment Tool alone, Dr. Walker noted.
Other aspects of bone quality include how many cracks are in the bone, the amount of adipose in the marrow space, and the rate at which bone is broken down and rebuilt. But Dr. Walker suggested that the longstanding focus on bone mineral density in clinical practice makes sense.
“By far, bone mass is the most important bone quality,” Dr. Walker said.
Dr. Thacker is the executive director of the nonprofit Speaking of Women’s Health. Dr. Walker reported receiving funding from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and Amgen.
A version of this article first appeared on Medscape.com.
Assessing a key aspect of bone architecture, for which clinicians can now be reimbursed under Medicare, can significantly improve the ability to predict a patient’s risk for bone fracture.
Although bone mineral density (BMD) is traditionally used to identify patients with osteoporosis or low bone mass, some physicians have begun incorporating the trabecular bone score (TBS) into their exams.
At the Cleveland Clinic Center for Specialized Women’s Health, factoring in the TBS changed the diagnosis for 16% of 432 patients, according to Holly Thacker, MD, the center’s director.
“Importantly, 11% got worse diagnoses, and I use that in terms of prioritizing treatment,” Dr. Thacker said in an interview. The ability to determine how degraded the bone microarchitecture is through a software system “is a huge advance.”
Dr. Thacker described her center’s experience with the technology at the annual meeting of the Menopause Society (formerly The North American Menopause Society).
While BMD captures the amount of minerals like calcium in the skeleton, TBS assesses the underlying microarchitecture by looking at the distribution of shades of gray on dual-energy x-ray absorptiometry (DXA) scans.
Based on the TBS, patients’ bones are classified as normal, partially degraded, or degraded. Among the 432 patients who received a TBS analysis in 2022, 3% shifted from a normal diagnosis to osteopenia, 8% worsened from osteopenia to osteoporosis, 4% went from osteopenia to normal, and 1.6% downgraded from osteoporosis to osteopenia, Dr. Thacker reported.
The new test may also provide some reassurance for female patients who have thinner bones, which may raise alarms based on BMD. TBS, however, may show that the structure of the bone looks normal.
“When you know that the microarchitecture is normal, you’re a lot less concerned that they actually have a bone disease of osteoporosis,” Dr. Thacker said.
Conversely, unexpectedly degraded bone raises questions.
“That makes you go back and say [to the patient]: ‘Have you been on steroids? Were you malnourished? Is there some other metabolic problem? Have you had some calcium disorder?’ ” Dr. Thacker said.
Dr. Thacker leverages the TBS to help patients obtain effective therapy, typically an anabolic agent followed by antiresorptive medication.
“When I see a patient who not only has osteoporosis on bone density but has completely degraded bone architecture, it’s a lot easier for me to make the argument to the insurance company that this patient is at grave risk for a low trauma fracture and bad outcome without the best treatment,” Dr. Thacker said.
10-year-old tech, recently covered
The Food and Drug Administration approved TBS software in 2012, but Medicare only recently started paying for it.
Medimaps Group, a company that markets imaging software to calculate TBS, announced in 2022 that reimbursement from the Centers for Medicare & Medicaid Services was available, at $41.53 on the Physician Fee Schedule and $82.61 on the Hospital Outpatient Prospective Payment Schedule.
“Reimbursement through CMS is an important endorsement of the clinical value of TBS for clinicians and their patients,” Didier Hans, PhD, MBA, the CEO of Medimaps, said in a statement at the time. He noted that more than 600,000 TBS procedures were being performed in the United States each year.
Nevertheless, the initial investment in purchasing the software may be a barrier for health systems.
“We are the first and only site in our health system to offer TBS, as this is an extra expense and not uniformly reimbursed by insurers,” Dr. Thacker reported at the meeting.
Potential drawbacks
The TBS software used in Dr. Thacker’s study has been validated only in Asian and White patients between certain ages and weights, meaning the system is not designed to be used for other populations. Other researchers have highlighted a need for trabecular bone scoring to be validated more broadly. The authors of a recent analysis, however, suggest that TBS can be used the same way no matter a patient’s race.
TBS “is going to be most helpful in those with osteopenia who are right near the threshold for treatment,” said Marcella Donovan Walker, MD, MS, in a presentation on bone quality at the meeting.
Many studies have shown that TBS “provides additive information to bone density,” said Dr. Walker, a professor of medicine in the division of endocrinology at Columbia University, New York. For example, a large study of women in Manitoba found that, regardless of whether their bone density was normal, osteopenic, or osteoporotic, those with a low TBS had a much higher risk for fracture.
‘Opportunistic screening’ with CT?
TBS relies on the same DXA scans that are used to calculate bone mineral density, so obtaining the score does not add time or radiation to the scanning process, Dr. Thacker said.
But many patients who should receive DXA scans do not, which adds to the promise of “opportunistic screening” for osteoporosis, Dr. Walker said. With this approach, physicians would analyze a CT scan that a patient received for another purpose, such as to investigate abdominal pain or chest pain.
“In these images is information about the bone,” Dr. Walker said.
Researchers have used high-resolution peripheral quantitative CT to perform finite element analysis, where a computer program simulates compression of the bone to create a measure of bone stiffness and determine the load required for a break.
One study found that including those elements predicted fractures better than bone mineral density or the Fracture Risk Assessment Tool alone, Dr. Walker noted.
Other aspects of bone quality include how many cracks are in the bone, the amount of adipose in the marrow space, and the rate at which bone is broken down and rebuilt. But Dr. Walker suggested that the longstanding focus on bone mineral density in clinical practice makes sense.
“By far, bone mass is the most important bone quality,” Dr. Walker said.
Dr. Thacker is the executive director of the nonprofit Speaking of Women’s Health. Dr. Walker reported receiving funding from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and Amgen.
A version of this article first appeared on Medscape.com.
Assessing a key aspect of bone architecture, for which clinicians can now be reimbursed under Medicare, can significantly improve the ability to predict a patient’s risk for bone fracture.
Although bone mineral density (BMD) is traditionally used to identify patients with osteoporosis or low bone mass, some physicians have begun incorporating the trabecular bone score (TBS) into their exams.
At the Cleveland Clinic Center for Specialized Women’s Health, factoring in the TBS changed the diagnosis for 16% of 432 patients, according to Holly Thacker, MD, the center’s director.
“Importantly, 11% got worse diagnoses, and I use that in terms of prioritizing treatment,” Dr. Thacker said in an interview. The ability to determine how degraded the bone microarchitecture is through a software system “is a huge advance.”
Dr. Thacker described her center’s experience with the technology at the annual meeting of the Menopause Society (formerly The North American Menopause Society).
While BMD captures the amount of minerals like calcium in the skeleton, TBS assesses the underlying microarchitecture by looking at the distribution of shades of gray on dual-energy x-ray absorptiometry (DXA) scans.
Based on the TBS, patients’ bones are classified as normal, partially degraded, or degraded. Among the 432 patients who received a TBS analysis in 2022, 3% shifted from a normal diagnosis to osteopenia, 8% worsened from osteopenia to osteoporosis, 4% went from osteopenia to normal, and 1.6% downgraded from osteoporosis to osteopenia, Dr. Thacker reported.
The new test may also provide some reassurance for female patients who have thinner bones, which may raise alarms based on BMD. TBS, however, may show that the structure of the bone looks normal.
“When you know that the microarchitecture is normal, you’re a lot less concerned that they actually have a bone disease of osteoporosis,” Dr. Thacker said.
Conversely, unexpectedly degraded bone raises questions.
“That makes you go back and say [to the patient]: ‘Have you been on steroids? Were you malnourished? Is there some other metabolic problem? Have you had some calcium disorder?’ ” Dr. Thacker said.
Dr. Thacker leverages the TBS to help patients obtain effective therapy, typically an anabolic agent followed by antiresorptive medication.
“When I see a patient who not only has osteoporosis on bone density but has completely degraded bone architecture, it’s a lot easier for me to make the argument to the insurance company that this patient is at grave risk for a low trauma fracture and bad outcome without the best treatment,” Dr. Thacker said.
10-year-old tech, recently covered
The Food and Drug Administration approved TBS software in 2012, but Medicare only recently started paying for it.
Medimaps Group, a company that markets imaging software to calculate TBS, announced in 2022 that reimbursement from the Centers for Medicare & Medicaid Services was available, at $41.53 on the Physician Fee Schedule and $82.61 on the Hospital Outpatient Prospective Payment Schedule.
“Reimbursement through CMS is an important endorsement of the clinical value of TBS for clinicians and their patients,” Didier Hans, PhD, MBA, the CEO of Medimaps, said in a statement at the time. He noted that more than 600,000 TBS procedures were being performed in the United States each year.
Nevertheless, the initial investment in purchasing the software may be a barrier for health systems.
“We are the first and only site in our health system to offer TBS, as this is an extra expense and not uniformly reimbursed by insurers,” Dr. Thacker reported at the meeting.
Potential drawbacks
The TBS software used in Dr. Thacker’s study has been validated only in Asian and White patients between certain ages and weights, meaning the system is not designed to be used for other populations. Other researchers have highlighted a need for trabecular bone scoring to be validated more broadly. The authors of a recent analysis, however, suggest that TBS can be used the same way no matter a patient’s race.
TBS “is going to be most helpful in those with osteopenia who are right near the threshold for treatment,” said Marcella Donovan Walker, MD, MS, in a presentation on bone quality at the meeting.
Many studies have shown that TBS “provides additive information to bone density,” said Dr. Walker, a professor of medicine in the division of endocrinology at Columbia University, New York. For example, a large study of women in Manitoba found that, regardless of whether their bone density was normal, osteopenic, or osteoporotic, those with a low TBS had a much higher risk for fracture.
‘Opportunistic screening’ with CT?
TBS relies on the same DXA scans that are used to calculate bone mineral density, so obtaining the score does not add time or radiation to the scanning process, Dr. Thacker said.
But many patients who should receive DXA scans do not, which adds to the promise of “opportunistic screening” for osteoporosis, Dr. Walker said. With this approach, physicians would analyze a CT scan that a patient received for another purpose, such as to investigate abdominal pain or chest pain.
“In these images is information about the bone,” Dr. Walker said.
Researchers have used high-resolution peripheral quantitative CT to perform finite element analysis, where a computer program simulates compression of the bone to create a measure of bone stiffness and determine the load required for a break.
One study found that including those elements predicted fractures better than bone mineral density or the Fracture Risk Assessment Tool alone, Dr. Walker noted.
Other aspects of bone quality include how many cracks are in the bone, the amount of adipose in the marrow space, and the rate at which bone is broken down and rebuilt. But Dr. Walker suggested that the longstanding focus on bone mineral density in clinical practice makes sense.
“By far, bone mass is the most important bone quality,” Dr. Walker said.
Dr. Thacker is the executive director of the nonprofit Speaking of Women’s Health. Dr. Walker reported receiving funding from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and Amgen.
A version of this article first appeared on Medscape.com.
FROM NAMS 2023
EMA warns that omega-3-acid ethyl esters may cause AFib
In its September meeting, the Should atrial fibrillation develop, intake of the medication must be stopped permanently.
Omega-3-acid ethyl esters are used to treat hypertriglyceridemia if lifestyle changes, particularly those related to nutrition, have not been sufficient to lower the blood triglyceride level. Hypertriglyceridemia is a risk factor for coronary heart disease.
During a Periodic Safety Update Single Assessment Procedure, the EMA safety committee analyzed systematic overviews and meta-analyses of randomized, controlled clinical studies. Experts found a dose-dependent increase in the risk for atrial fibrillation in patients with cardiovascular diseases or cardiovascular risk factors who were being treated with omega-3-acid ethyl esters, compared with those treated with placebo. The observed risk was at its highest at a dose of 4 g/d.
The PRAC will recommend an update to the Summary of Product Characteristics for preparations that contain omega-3-acid ethyl esters. The aim is to inform physicians, pharmacists, and patients of the risk for atrial fibrillation. A notification will be sent to health care professionals soon to inform them of further details.
This article was translated from the Medscape German Edition. A version appeared on Medscape.com.
In its September meeting, the Should atrial fibrillation develop, intake of the medication must be stopped permanently.
Omega-3-acid ethyl esters are used to treat hypertriglyceridemia if lifestyle changes, particularly those related to nutrition, have not been sufficient to lower the blood triglyceride level. Hypertriglyceridemia is a risk factor for coronary heart disease.
During a Periodic Safety Update Single Assessment Procedure, the EMA safety committee analyzed systematic overviews and meta-analyses of randomized, controlled clinical studies. Experts found a dose-dependent increase in the risk for atrial fibrillation in patients with cardiovascular diseases or cardiovascular risk factors who were being treated with omega-3-acid ethyl esters, compared with those treated with placebo. The observed risk was at its highest at a dose of 4 g/d.
The PRAC will recommend an update to the Summary of Product Characteristics for preparations that contain omega-3-acid ethyl esters. The aim is to inform physicians, pharmacists, and patients of the risk for atrial fibrillation. A notification will be sent to health care professionals soon to inform them of further details.
This article was translated from the Medscape German Edition. A version appeared on Medscape.com.
In its September meeting, the Should atrial fibrillation develop, intake of the medication must be stopped permanently.
Omega-3-acid ethyl esters are used to treat hypertriglyceridemia if lifestyle changes, particularly those related to nutrition, have not been sufficient to lower the blood triglyceride level. Hypertriglyceridemia is a risk factor for coronary heart disease.
During a Periodic Safety Update Single Assessment Procedure, the EMA safety committee analyzed systematic overviews and meta-analyses of randomized, controlled clinical studies. Experts found a dose-dependent increase in the risk for atrial fibrillation in patients with cardiovascular diseases or cardiovascular risk factors who were being treated with omega-3-acid ethyl esters, compared with those treated with placebo. The observed risk was at its highest at a dose of 4 g/d.
The PRAC will recommend an update to the Summary of Product Characteristics for preparations that contain omega-3-acid ethyl esters. The aim is to inform physicians, pharmacists, and patients of the risk for atrial fibrillation. A notification will be sent to health care professionals soon to inform them of further details.
This article was translated from the Medscape German Edition. A version appeared on Medscape.com.
Tool in development aims to steer telehealth visit appropriateness in rheumatology
Researchers have created a model to suggest which rheumatology visits can be effectively carried out via telehealth and which should remain in-person visits. The model not only could help to alleviate the decision burden on providers but also help to navigate how to incorporate telehealth into daily rheumatology practice as the COVID-19 pandemic subsides, experts say.
The beginning of the pandemic quickly drove rheumatology practices to adopt telehealth – which in the past has been studied only in selective groups of relatively stable patients. In Duke University’s department of rheumatology, nearly 90% of visits were conducted via telehealth for several weeks in April and May 2020, said David L. Leverenz, MD, an assistant professor of medicine at Duke University in Durham, N.C.
Since then, the practice has continued providing telehealth for a wider variety of patients: patients with high disease activity, those with low disease activity, people living 3 hours away or just 5 minutes from the medical center.
“Although the pandemic has really improved, and certainly we feel very safe providing in-person care, we’ve realized that it’s actually really possible to provide telehealth care to a lot of patients,” he said.
The model was then piloted with four Duke University rheumatologists for visits that took place from March through July 2022. A month prior to visits, researchers provided the rheumatologists a list of in-patient visits that the model suggested could be changed to telehealth visits. The list also included false changes, in which randomly selected visits were presented as being recommended for telehealth, although the model had not actually recommended the changes.
Dr. Leverenz and colleagues measured how frequently providers agreed with changes recommended by the model and the false changes. If a provider accepted a model-suggested change, the patient was contacted to either accept or reject the proposed switch. If a provider agreed with a false change, no additional action was taken.
Of the 201 patient encounters, there were 101 model-recommended changes and 100 artificial changes. Nearly all (99%) model-recommended changes suggested moving from in-person visits to telehealth visits. There was one recommendation to change a scheduled telehealth encounter to an in-person encounter. Of the false recommendations, 90 were for scheduled in-person visits, and 10 were for scheduled telehealth appointments.
Providers agreed with 71% of the model-recommended changes and with 21% of the false changes. Dr. Leverenz and colleagues reported “moderate agreement” between providers and model recommendations, reflected by a Cohen kappa coefficient of 0.45.
“This lined up pretty well with what providers thought was appropriate for the patient when presented with the information, and it also showed that, on top of current practice, that the model really does have the ability to expand appropriate telehealth care, which is really what we’re looking to do,” Dr. Leverenz said.
The article describing these results was published in Arthritis Care and Research.
Perhaps unsurprisingly, clinician preference for and experience with telemedicine were the biggest predictors of whether changes suggested by the model were accepted.
Focus model to specific diagnoses or many?
Currently, clinicians who are already juggling many other responsibilities throughout the day must use their own judgment to determine whether telemedicine may be appropriate. A model such as this could help alleviate that decision burden, said Kathleen Fear, PhD, the director of data and analytics at the University of Rochester Medical Center Health Lab, in New York.
“A model that can help with scheduling or prompt a provider or patient for when a visit is appropriate for telemedicine seems like a really effective way to make the most of telemedicine while reducing potential burden on providers,” she said.
Dr. Leverenz imagines that this model could be embedded into electronic health records as a “decision support tool” to aid discussions between patients and providers on whether telehealth might be appropriate for upcoming visits.
But developing a model that can generate predictions for the wide variety of conditions seen in daily rheumatology practice can be a challenge, said Christine Peoples, MD, a clinical associate professor of medicine and director of the tele-rheumatology program at the University of Pittsburgh.
“If you focus the model to certain diagnoses, at least in the beginning, that’s very helpful, because it’s too difficult to have one model for every single reason that folks see a rheumatologist,” she said.
Daniel A. Albert, MD, a professor of medicine and pediatrics at the Geisel School of Medicine at Dartmouth in Hanover, N,H., agreed. The model is “a good start,” he said, and highlights that tele-rheumatology continues to be underutilized in practice. But he argued that the moderate agreement found with the model was relatively low.
A more focused algorithm that targets a single or several more common conditions may be more accurate, he said. “You probably want to break it down,” Dr. Albert said.
But Dr. Leverenz argued that the novelty of this model is that it incorporates the many different conditions seen in daily rheumatology practice, whereas previous programs utilizing telehealth focused on specific conditions and patients with low disease activity.
In addition, the model is currently provider centric and does not take patient preference into account, Dr. Albert added. Dr. Leverenz said that that is the next step in further developing this model. He is currently conducting qualitative analyses with patients to better understand what patients think and how often their views on telehealth differ from that of their care providers.
“Hopefully, we can expand appropriate telehealth visits by teaching providers not just to do what they think is right for the patient but also meet the patient’s expectations and needs, based on what we learn,” he said.
The study was funded by a grant from Pfizer. Dr. Leverenz has received grants from Pfizer and has served as a consultant for Sanofi. None of the study’s other authors report relevant financial relationships. Dr. Peoples is an educational consultant on telehealth for Pfizer. Dr. Alberts has previously received grant funding from Pfizer. Dr. Fear has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Researchers have created a model to suggest which rheumatology visits can be effectively carried out via telehealth and which should remain in-person visits. The model not only could help to alleviate the decision burden on providers but also help to navigate how to incorporate telehealth into daily rheumatology practice as the COVID-19 pandemic subsides, experts say.
The beginning of the pandemic quickly drove rheumatology practices to adopt telehealth – which in the past has been studied only in selective groups of relatively stable patients. In Duke University’s department of rheumatology, nearly 90% of visits were conducted via telehealth for several weeks in April and May 2020, said David L. Leverenz, MD, an assistant professor of medicine at Duke University in Durham, N.C.
Since then, the practice has continued providing telehealth for a wider variety of patients: patients with high disease activity, those with low disease activity, people living 3 hours away or just 5 minutes from the medical center.
“Although the pandemic has really improved, and certainly we feel very safe providing in-person care, we’ve realized that it’s actually really possible to provide telehealth care to a lot of patients,” he said.
The model was then piloted with four Duke University rheumatologists for visits that took place from March through July 2022. A month prior to visits, researchers provided the rheumatologists a list of in-patient visits that the model suggested could be changed to telehealth visits. The list also included false changes, in which randomly selected visits were presented as being recommended for telehealth, although the model had not actually recommended the changes.
Dr. Leverenz and colleagues measured how frequently providers agreed with changes recommended by the model and the false changes. If a provider accepted a model-suggested change, the patient was contacted to either accept or reject the proposed switch. If a provider agreed with a false change, no additional action was taken.
Of the 201 patient encounters, there were 101 model-recommended changes and 100 artificial changes. Nearly all (99%) model-recommended changes suggested moving from in-person visits to telehealth visits. There was one recommendation to change a scheduled telehealth encounter to an in-person encounter. Of the false recommendations, 90 were for scheduled in-person visits, and 10 were for scheduled telehealth appointments.
Providers agreed with 71% of the model-recommended changes and with 21% of the false changes. Dr. Leverenz and colleagues reported “moderate agreement” between providers and model recommendations, reflected by a Cohen kappa coefficient of 0.45.
“This lined up pretty well with what providers thought was appropriate for the patient when presented with the information, and it also showed that, on top of current practice, that the model really does have the ability to expand appropriate telehealth care, which is really what we’re looking to do,” Dr. Leverenz said.
The article describing these results was published in Arthritis Care and Research.
Perhaps unsurprisingly, clinician preference for and experience with telemedicine were the biggest predictors of whether changes suggested by the model were accepted.
Focus model to specific diagnoses or many?
Currently, clinicians who are already juggling many other responsibilities throughout the day must use their own judgment to determine whether telemedicine may be appropriate. A model such as this could help alleviate that decision burden, said Kathleen Fear, PhD, the director of data and analytics at the University of Rochester Medical Center Health Lab, in New York.
“A model that can help with scheduling or prompt a provider or patient for when a visit is appropriate for telemedicine seems like a really effective way to make the most of telemedicine while reducing potential burden on providers,” she said.
Dr. Leverenz imagines that this model could be embedded into electronic health records as a “decision support tool” to aid discussions between patients and providers on whether telehealth might be appropriate for upcoming visits.
But developing a model that can generate predictions for the wide variety of conditions seen in daily rheumatology practice can be a challenge, said Christine Peoples, MD, a clinical associate professor of medicine and director of the tele-rheumatology program at the University of Pittsburgh.
“If you focus the model to certain diagnoses, at least in the beginning, that’s very helpful, because it’s too difficult to have one model for every single reason that folks see a rheumatologist,” she said.
Daniel A. Albert, MD, a professor of medicine and pediatrics at the Geisel School of Medicine at Dartmouth in Hanover, N,H., agreed. The model is “a good start,” he said, and highlights that tele-rheumatology continues to be underutilized in practice. But he argued that the moderate agreement found with the model was relatively low.
A more focused algorithm that targets a single or several more common conditions may be more accurate, he said. “You probably want to break it down,” Dr. Albert said.
But Dr. Leverenz argued that the novelty of this model is that it incorporates the many different conditions seen in daily rheumatology practice, whereas previous programs utilizing telehealth focused on specific conditions and patients with low disease activity.
In addition, the model is currently provider centric and does not take patient preference into account, Dr. Albert added. Dr. Leverenz said that that is the next step in further developing this model. He is currently conducting qualitative analyses with patients to better understand what patients think and how often their views on telehealth differ from that of their care providers.
“Hopefully, we can expand appropriate telehealth visits by teaching providers not just to do what they think is right for the patient but also meet the patient’s expectations and needs, based on what we learn,” he said.
The study was funded by a grant from Pfizer. Dr. Leverenz has received grants from Pfizer and has served as a consultant for Sanofi. None of the study’s other authors report relevant financial relationships. Dr. Peoples is an educational consultant on telehealth for Pfizer. Dr. Alberts has previously received grant funding from Pfizer. Dr. Fear has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Researchers have created a model to suggest which rheumatology visits can be effectively carried out via telehealth and which should remain in-person visits. The model not only could help to alleviate the decision burden on providers but also help to navigate how to incorporate telehealth into daily rheumatology practice as the COVID-19 pandemic subsides, experts say.
The beginning of the pandemic quickly drove rheumatology practices to adopt telehealth – which in the past has been studied only in selective groups of relatively stable patients. In Duke University’s department of rheumatology, nearly 90% of visits were conducted via telehealth for several weeks in April and May 2020, said David L. Leverenz, MD, an assistant professor of medicine at Duke University in Durham, N.C.
Since then, the practice has continued providing telehealth for a wider variety of patients: patients with high disease activity, those with low disease activity, people living 3 hours away or just 5 minutes from the medical center.
“Although the pandemic has really improved, and certainly we feel very safe providing in-person care, we’ve realized that it’s actually really possible to provide telehealth care to a lot of patients,” he said.
The model was then piloted with four Duke University rheumatologists for visits that took place from March through July 2022. A month prior to visits, researchers provided the rheumatologists a list of in-patient visits that the model suggested could be changed to telehealth visits. The list also included false changes, in which randomly selected visits were presented as being recommended for telehealth, although the model had not actually recommended the changes.
Dr. Leverenz and colleagues measured how frequently providers agreed with changes recommended by the model and the false changes. If a provider accepted a model-suggested change, the patient was contacted to either accept or reject the proposed switch. If a provider agreed with a false change, no additional action was taken.
Of the 201 patient encounters, there were 101 model-recommended changes and 100 artificial changes. Nearly all (99%) model-recommended changes suggested moving from in-person visits to telehealth visits. There was one recommendation to change a scheduled telehealth encounter to an in-person encounter. Of the false recommendations, 90 were for scheduled in-person visits, and 10 were for scheduled telehealth appointments.
Providers agreed with 71% of the model-recommended changes and with 21% of the false changes. Dr. Leverenz and colleagues reported “moderate agreement” between providers and model recommendations, reflected by a Cohen kappa coefficient of 0.45.
“This lined up pretty well with what providers thought was appropriate for the patient when presented with the information, and it also showed that, on top of current practice, that the model really does have the ability to expand appropriate telehealth care, which is really what we’re looking to do,” Dr. Leverenz said.
The article describing these results was published in Arthritis Care and Research.
Perhaps unsurprisingly, clinician preference for and experience with telemedicine were the biggest predictors of whether changes suggested by the model were accepted.
Focus model to specific diagnoses or many?
Currently, clinicians who are already juggling many other responsibilities throughout the day must use their own judgment to determine whether telemedicine may be appropriate. A model such as this could help alleviate that decision burden, said Kathleen Fear, PhD, the director of data and analytics at the University of Rochester Medical Center Health Lab, in New York.
“A model that can help with scheduling or prompt a provider or patient for when a visit is appropriate for telemedicine seems like a really effective way to make the most of telemedicine while reducing potential burden on providers,” she said.
Dr. Leverenz imagines that this model could be embedded into electronic health records as a “decision support tool” to aid discussions between patients and providers on whether telehealth might be appropriate for upcoming visits.
But developing a model that can generate predictions for the wide variety of conditions seen in daily rheumatology practice can be a challenge, said Christine Peoples, MD, a clinical associate professor of medicine and director of the tele-rheumatology program at the University of Pittsburgh.
“If you focus the model to certain diagnoses, at least in the beginning, that’s very helpful, because it’s too difficult to have one model for every single reason that folks see a rheumatologist,” she said.
Daniel A. Albert, MD, a professor of medicine and pediatrics at the Geisel School of Medicine at Dartmouth in Hanover, N,H., agreed. The model is “a good start,” he said, and highlights that tele-rheumatology continues to be underutilized in practice. But he argued that the moderate agreement found with the model was relatively low.
A more focused algorithm that targets a single or several more common conditions may be more accurate, he said. “You probably want to break it down,” Dr. Albert said.
But Dr. Leverenz argued that the novelty of this model is that it incorporates the many different conditions seen in daily rheumatology practice, whereas previous programs utilizing telehealth focused on specific conditions and patients with low disease activity.
In addition, the model is currently provider centric and does not take patient preference into account, Dr. Albert added. Dr. Leverenz said that that is the next step in further developing this model. He is currently conducting qualitative analyses with patients to better understand what patients think and how often their views on telehealth differ from that of their care providers.
“Hopefully, we can expand appropriate telehealth visits by teaching providers not just to do what they think is right for the patient but also meet the patient’s expectations and needs, based on what we learn,” he said.
The study was funded by a grant from Pfizer. Dr. Leverenz has received grants from Pfizer and has served as a consultant for Sanofi. None of the study’s other authors report relevant financial relationships. Dr. Peoples is an educational consultant on telehealth for Pfizer. Dr. Alberts has previously received grant funding from Pfizer. Dr. Fear has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ARTHRITIS CARE AND RESEARCH
3-D stereophotogrammetry helps detect progression of craniofacial morphea
TOPLINE:
over time.
METHODOLOGY:
- Existing tools that detect disease progression in patients with CM are limited.
- In a prospective cohort study, researchers evaluated the use of 3-D stereophotogrammetry, a noninvasive, radiation-free imaging modality, to detect disease progression in 27 consecutive patients with CM seen at Boston Children’s Hospital and Brigham and Women’s Hospital from April 1, 2019, to March 1, 2023.
- After clinical and 3-D stereophotogrammetry assessments were performed at 2- to 12-month intervals, the 3-D images were rated by an expert (a board-certified plastic craniofacial surgeon) and a nonexpert (a board-certified dermatologist) as demonstrating progression or no progression.
- Kappa coefficients were used to calculate inter-rater reliability.
TAKEAWAY:
- Most of the study participants (73%) were female, their median age was 14 years (range, 5-40 years), and each underwent 3-D stereophotogrammetry imaging at least two times spaced a median of 3 months apart.
- On the basis of clinical assessments during the 48-month study period, 10 patients (37%) experienced progression of their disease.
- 3-D stereophotogrammetry not only corroborated clinical impressions of disease progression with strong inter-rater reliability (kappa = 0.80; 95% confidence interval, 0.61-0.99), but it also detected occult progression of asymmetry not noted on clinical examination in three additional patients.
- In subgroup analyses, assessment of 3-D images demonstrated substantial to near-perfect inter-rater reliability in patients with Fitzpatrick skin types IV-VI.
IN PRACTICE:
“Further work is necessary to validate this measure in a larger cohort and to guide its incorporation into medical decision-making for patients with CM,” the researchers wrote.
SOURCE:
Katharina S. Shaw, MD, of the department of dermatology at the University of Pennsylvania, Philadelphia, led the research. The study was published online in JAMA Dermatology.
LIMITATIONS:
The sample was small, and a criterion standard for assessing CM was lacking.
DISCLOSURES:
The researchers reported having no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
over time.
METHODOLOGY:
- Existing tools that detect disease progression in patients with CM are limited.
- In a prospective cohort study, researchers evaluated the use of 3-D stereophotogrammetry, a noninvasive, radiation-free imaging modality, to detect disease progression in 27 consecutive patients with CM seen at Boston Children’s Hospital and Brigham and Women’s Hospital from April 1, 2019, to March 1, 2023.
- After clinical and 3-D stereophotogrammetry assessments were performed at 2- to 12-month intervals, the 3-D images were rated by an expert (a board-certified plastic craniofacial surgeon) and a nonexpert (a board-certified dermatologist) as demonstrating progression or no progression.
- Kappa coefficients were used to calculate inter-rater reliability.
TAKEAWAY:
- Most of the study participants (73%) were female, their median age was 14 years (range, 5-40 years), and each underwent 3-D stereophotogrammetry imaging at least two times spaced a median of 3 months apart.
- On the basis of clinical assessments during the 48-month study period, 10 patients (37%) experienced progression of their disease.
- 3-D stereophotogrammetry not only corroborated clinical impressions of disease progression with strong inter-rater reliability (kappa = 0.80; 95% confidence interval, 0.61-0.99), but it also detected occult progression of asymmetry not noted on clinical examination in three additional patients.
- In subgroup analyses, assessment of 3-D images demonstrated substantial to near-perfect inter-rater reliability in patients with Fitzpatrick skin types IV-VI.
IN PRACTICE:
“Further work is necessary to validate this measure in a larger cohort and to guide its incorporation into medical decision-making for patients with CM,” the researchers wrote.
SOURCE:
Katharina S. Shaw, MD, of the department of dermatology at the University of Pennsylvania, Philadelphia, led the research. The study was published online in JAMA Dermatology.
LIMITATIONS:
The sample was small, and a criterion standard for assessing CM was lacking.
DISCLOSURES:
The researchers reported having no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
over time.
METHODOLOGY:
- Existing tools that detect disease progression in patients with CM are limited.
- In a prospective cohort study, researchers evaluated the use of 3-D stereophotogrammetry, a noninvasive, radiation-free imaging modality, to detect disease progression in 27 consecutive patients with CM seen at Boston Children’s Hospital and Brigham and Women’s Hospital from April 1, 2019, to March 1, 2023.
- After clinical and 3-D stereophotogrammetry assessments were performed at 2- to 12-month intervals, the 3-D images were rated by an expert (a board-certified plastic craniofacial surgeon) and a nonexpert (a board-certified dermatologist) as demonstrating progression or no progression.
- Kappa coefficients were used to calculate inter-rater reliability.
TAKEAWAY:
- Most of the study participants (73%) were female, their median age was 14 years (range, 5-40 years), and each underwent 3-D stereophotogrammetry imaging at least two times spaced a median of 3 months apart.
- On the basis of clinical assessments during the 48-month study period, 10 patients (37%) experienced progression of their disease.
- 3-D stereophotogrammetry not only corroborated clinical impressions of disease progression with strong inter-rater reliability (kappa = 0.80; 95% confidence interval, 0.61-0.99), but it also detected occult progression of asymmetry not noted on clinical examination in three additional patients.
- In subgroup analyses, assessment of 3-D images demonstrated substantial to near-perfect inter-rater reliability in patients with Fitzpatrick skin types IV-VI.
IN PRACTICE:
“Further work is necessary to validate this measure in a larger cohort and to guide its incorporation into medical decision-making for patients with CM,” the researchers wrote.
SOURCE:
Katharina S. Shaw, MD, of the department of dermatology at the University of Pennsylvania, Philadelphia, led the research. The study was published online in JAMA Dermatology.
LIMITATIONS:
The sample was small, and a criterion standard for assessing CM was lacking.
DISCLOSURES:
The researchers reported having no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Pediatric psoriasis: Black children, males more likely to have palmoplantar subtype, study finds
TOPLINE:
.
METHODOLOGY:
- Researchers reviewed data on 330 children and youths aged 0-18 years who had received a primary psoriasis diagnosis and who were seen at an academic pediatric dermatology clinic from 2012 to 2022. Among these patients, 50 cases of palmoplantar psoriasis (PP) were identified by pediatric dermatologists.
- The study population was stratified by race/ethnicity on the basis of self-identification. The cohort included White, Black, and Hispanic/Latino patients, as well as patients who identified as other; 71.5% were White persons, 59.1% were female patients.
- The researchers used a regression analysis to investigate the association between race/ethnicity and PP after controlling for multiple confounding variables, including age and gender.
TAKEAWAY:
- Black children were significantly more likely to have PP than White children (adjusted odds ratio, 6.386; P < .0001). PP was diagnosed in 41.9%, 11.5%, and 8.9% of Black, Hispanic/Latino, and White children, respectively.
- Male gender was also identified as an independent risk factor for PP (aOR, 2.241).
- Nail involvement occurred in significantly more Black and Hispanic/Latino patients than in White patients (53.2%, 50.0%, and 33.9%, respectively).
- Black patients had significantly more palm and sole involvement, compared with the other groups (P < .0001 for both); however, White children had significantly more scalp involvement, compared with the other groups (P = .04).
IN PRACTICE:
“Further research is warranted to better understand the degree to which these associations are affected by racial disparities and environmental factors,” as well as potential genetic associations, the researchers noted.
SOURCE:
The corresponding author on the study was Amy Theos, MD, of the department of dermatology at the University of Alabama, Birmingham. The study was published online in Pediatric Dermatology.
LIMITATIONS:
The findings were limited by the small sample size and incomplete data for some patients.
DISCLOSURES:
The study received no outside funding. The researchers had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
TOPLINE:
.
METHODOLOGY:
- Researchers reviewed data on 330 children and youths aged 0-18 years who had received a primary psoriasis diagnosis and who were seen at an academic pediatric dermatology clinic from 2012 to 2022. Among these patients, 50 cases of palmoplantar psoriasis (PP) were identified by pediatric dermatologists.
- The study population was stratified by race/ethnicity on the basis of self-identification. The cohort included White, Black, and Hispanic/Latino patients, as well as patients who identified as other; 71.5% were White persons, 59.1% were female patients.
- The researchers used a regression analysis to investigate the association between race/ethnicity and PP after controlling for multiple confounding variables, including age and gender.
TAKEAWAY:
- Black children were significantly more likely to have PP than White children (adjusted odds ratio, 6.386; P < .0001). PP was diagnosed in 41.9%, 11.5%, and 8.9% of Black, Hispanic/Latino, and White children, respectively.
- Male gender was also identified as an independent risk factor for PP (aOR, 2.241).
- Nail involvement occurred in significantly more Black and Hispanic/Latino patients than in White patients (53.2%, 50.0%, and 33.9%, respectively).
- Black patients had significantly more palm and sole involvement, compared with the other groups (P < .0001 for both); however, White children had significantly more scalp involvement, compared with the other groups (P = .04).
IN PRACTICE:
“Further research is warranted to better understand the degree to which these associations are affected by racial disparities and environmental factors,” as well as potential genetic associations, the researchers noted.
SOURCE:
The corresponding author on the study was Amy Theos, MD, of the department of dermatology at the University of Alabama, Birmingham. The study was published online in Pediatric Dermatology.
LIMITATIONS:
The findings were limited by the small sample size and incomplete data for some patients.
DISCLOSURES:
The study received no outside funding. The researchers had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
TOPLINE:
.
METHODOLOGY:
- Researchers reviewed data on 330 children and youths aged 0-18 years who had received a primary psoriasis diagnosis and who were seen at an academic pediatric dermatology clinic from 2012 to 2022. Among these patients, 50 cases of palmoplantar psoriasis (PP) were identified by pediatric dermatologists.
- The study population was stratified by race/ethnicity on the basis of self-identification. The cohort included White, Black, and Hispanic/Latino patients, as well as patients who identified as other; 71.5% were White persons, 59.1% were female patients.
- The researchers used a regression analysis to investigate the association between race/ethnicity and PP after controlling for multiple confounding variables, including age and gender.
TAKEAWAY:
- Black children were significantly more likely to have PP than White children (adjusted odds ratio, 6.386; P < .0001). PP was diagnosed in 41.9%, 11.5%, and 8.9% of Black, Hispanic/Latino, and White children, respectively.
- Male gender was also identified as an independent risk factor for PP (aOR, 2.241).
- Nail involvement occurred in significantly more Black and Hispanic/Latino patients than in White patients (53.2%, 50.0%, and 33.9%, respectively).
- Black patients had significantly more palm and sole involvement, compared with the other groups (P < .0001 for both); however, White children had significantly more scalp involvement, compared with the other groups (P = .04).
IN PRACTICE:
“Further research is warranted to better understand the degree to which these associations are affected by racial disparities and environmental factors,” as well as potential genetic associations, the researchers noted.
SOURCE:
The corresponding author on the study was Amy Theos, MD, of the department of dermatology at the University of Alabama, Birmingham. The study was published online in Pediatric Dermatology.
LIMITATIONS:
The findings were limited by the small sample size and incomplete data for some patients.
DISCLOSURES:
The study received no outside funding. The researchers had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Anemia, iron deficit common in rheumatic disease pregnancy
TOPLINE:
, according to findings from a longitudinal cohort study.
METHODOLOGY:
- Researchers analyzed data from 368 pregnancies in women with rheumatic diseases during the period 2014-2022; nearly two-thirds (62%) had a connective tissue disease, 16% had rheumatoid arthritis or juvenile idiopathic arthritis, 14% had spondyloarthritis, 3% had vasculitis, and 7% had other diseases.
- Patients were aged 17-44 years, with a median age of 32 years at the time of birth.
- Researchers examined the frequency of anemia and iron deficiency and the impact of anemia on adverse maternal and child outcomes.
TAKEAWAY:
- The prevalence of iron deficiency was 28%, 51%, and 62% in the first, second, and third trimesters, respectively.
- The prevalence of anemia was 18%, 27%, and 33% in the first, second, and third trimesters, respectively.
- There was an increased risk for fetal complications such as malformation, infections, small for gestational age, neonatal lupus, preterm birth, and abortion or stillbirth in association with maternal connective tissue disease (odds ratio, 2.14) and also with low maternal hemoglobin levels and maternal iron deficiency (ORs, 0.52 and 0.86, respectively).
- Lower maternal hemoglobin levels were associated with an increased risk for maternal complications (OR, 1.47) such as flare with adaption of rheumatic medication and pregnancy-related adverse events (preeclampsia, gestational diabetes, bleeding complications, and thromboembolism), but patients with connective tissue disease had a lower risk for maternal complications (OR, 0.51); mean serum ferritin had no significant impact on maternal complications (OR, 1.02).
IN PRACTICE:
“Patients with rheumatic diseases suffer more often and already in early pregnancy from iron deficiency,” the researchers write. Therefore, early identification of anemia and iron deficiency in this population could inform prepregnancy counseling.
SOURCE:
The lead author on the study was Ann-Christin Pecher, MD, of University Hospital Tübingen, Germany. The study was published online in Joint Bone Spine.
LIMITATIONS:
The findings were limited by the use of a single dataset that might not be representative of all pregnant patients with rheumatic diseases. Other limitations included the lack of a standardized approach to iron supplementation.
DISCLOSURES:
The study was supported by a grant from the Medical Faculty of Tübingen Clinician-Scientist to the lead author. The researchers report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
TOPLINE:
, according to findings from a longitudinal cohort study.
METHODOLOGY:
- Researchers analyzed data from 368 pregnancies in women with rheumatic diseases during the period 2014-2022; nearly two-thirds (62%) had a connective tissue disease, 16% had rheumatoid arthritis or juvenile idiopathic arthritis, 14% had spondyloarthritis, 3% had vasculitis, and 7% had other diseases.
- Patients were aged 17-44 years, with a median age of 32 years at the time of birth.
- Researchers examined the frequency of anemia and iron deficiency and the impact of anemia on adverse maternal and child outcomes.
TAKEAWAY:
- The prevalence of iron deficiency was 28%, 51%, and 62% in the first, second, and third trimesters, respectively.
- The prevalence of anemia was 18%, 27%, and 33% in the first, second, and third trimesters, respectively.
- There was an increased risk for fetal complications such as malformation, infections, small for gestational age, neonatal lupus, preterm birth, and abortion or stillbirth in association with maternal connective tissue disease (odds ratio, 2.14) and also with low maternal hemoglobin levels and maternal iron deficiency (ORs, 0.52 and 0.86, respectively).
- Lower maternal hemoglobin levels were associated with an increased risk for maternal complications (OR, 1.47) such as flare with adaption of rheumatic medication and pregnancy-related adverse events (preeclampsia, gestational diabetes, bleeding complications, and thromboembolism), but patients with connective tissue disease had a lower risk for maternal complications (OR, 0.51); mean serum ferritin had no significant impact on maternal complications (OR, 1.02).
IN PRACTICE:
“Patients with rheumatic diseases suffer more often and already in early pregnancy from iron deficiency,” the researchers write. Therefore, early identification of anemia and iron deficiency in this population could inform prepregnancy counseling.
SOURCE:
The lead author on the study was Ann-Christin Pecher, MD, of University Hospital Tübingen, Germany. The study was published online in Joint Bone Spine.
LIMITATIONS:
The findings were limited by the use of a single dataset that might not be representative of all pregnant patients with rheumatic diseases. Other limitations included the lack of a standardized approach to iron supplementation.
DISCLOSURES:
The study was supported by a grant from the Medical Faculty of Tübingen Clinician-Scientist to the lead author. The researchers report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
TOPLINE:
, according to findings from a longitudinal cohort study.
METHODOLOGY:
- Researchers analyzed data from 368 pregnancies in women with rheumatic diseases during the period 2014-2022; nearly two-thirds (62%) had a connective tissue disease, 16% had rheumatoid arthritis or juvenile idiopathic arthritis, 14% had spondyloarthritis, 3% had vasculitis, and 7% had other diseases.
- Patients were aged 17-44 years, with a median age of 32 years at the time of birth.
- Researchers examined the frequency of anemia and iron deficiency and the impact of anemia on adverse maternal and child outcomes.
TAKEAWAY:
- The prevalence of iron deficiency was 28%, 51%, and 62% in the first, second, and third trimesters, respectively.
- The prevalence of anemia was 18%, 27%, and 33% in the first, second, and third trimesters, respectively.
- There was an increased risk for fetal complications such as malformation, infections, small for gestational age, neonatal lupus, preterm birth, and abortion or stillbirth in association with maternal connective tissue disease (odds ratio, 2.14) and also with low maternal hemoglobin levels and maternal iron deficiency (ORs, 0.52 and 0.86, respectively).
- Lower maternal hemoglobin levels were associated with an increased risk for maternal complications (OR, 1.47) such as flare with adaption of rheumatic medication and pregnancy-related adverse events (preeclampsia, gestational diabetes, bleeding complications, and thromboembolism), but patients with connective tissue disease had a lower risk for maternal complications (OR, 0.51); mean serum ferritin had no significant impact on maternal complications (OR, 1.02).
IN PRACTICE:
“Patients with rheumatic diseases suffer more often and already in early pregnancy from iron deficiency,” the researchers write. Therefore, early identification of anemia and iron deficiency in this population could inform prepregnancy counseling.
SOURCE:
The lead author on the study was Ann-Christin Pecher, MD, of University Hospital Tübingen, Germany. The study was published online in Joint Bone Spine.
LIMITATIONS:
The findings were limited by the use of a single dataset that might not be representative of all pregnant patients with rheumatic diseases. Other limitations included the lack of a standardized approach to iron supplementation.
DISCLOSURES:
The study was supported by a grant from the Medical Faculty of Tübingen Clinician-Scientist to the lead author. The researchers report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Anti-acid meds lower strength of systemic sclerosis drug
TOPLINE:
Anti-acid drugs used by patients with systemic sclerosis reduce the bioavailability of mycophenolate mofetil (MMF).
METHODOLOGY:
- Researchers conducted an open-label, pragmatic crossover study of 20 patients (all female) with systemic sclerosis at a single center who were on a stable MMF dose (1.5-2 g/day) for the last 3 months or more.
- Participants sequentially took MMF alone for 1 month, then with the H2 receptor blocker (HRB) ranitidine 300 mg/day in the second month, then with the proton pump inhibitor (PPI) esomeprazole 40 mg/day in the third month.
- Researchers measured the bioavailability of MMF in the patients during treatment with ranitidine or esomeprazole and the impact of the drugs on the total GI score of the UCLA Scleroderma Clinical Trial Consortium Gastrointestinal Tract 2.0 instrument.
- Patients were excluded if they were receiving co-prescription of cholestyramine, magnesium- or aluminum-containing antacids, and rifampicin; taking prednisolone-equivalent dose > 5 mg/day; taking MMF plus a PPI or an HRB at baseline; living with chronic kidney disease with a glomerular filtration rate < 30 mL/min; positive for HIV, HCV, or HBV; or living with end-stage lung disease or gastroduodenal ulcers.
TAKEAWAY:
- Mean estimated 12-hour area under curve levels of mycophenolic acid dropped by 32.7% (mean difference = 22.28 mcg h mL–1) when patients added esomeprazole, and they dipped by 21.97% (mean difference = 14.93 mcg h mL–1) when they added ranitidine vs. MMF alone.
- The pharmacokinetic parameter T-max did not differ significantly between MMF alone vs. MMF plus ranitidine but was significantly different with esomeprazole. C-max significantly declined with administration of ranitidine or esomeprazole vs. MMF alone.
- Total GI scores dipped when patients added esomeprazole or ranitidine.
IN PRACTICE:
In patients with significant gastroesophageal reflux disease symptoms who need to take MMF, management options may include monitoring MMF drug levels, switching to enteric-coated mycophenolate sodium, and spacing doses with anti-acid drugs.
SOURCE:
Glaxon Alex, MD, and colleagues from the Center for Arthritis and Rheumatism Excellence in Kochi, India, conducted the study, which was published online in Seminars in Arthritis & Rheumatism.
LIMITATIONS:
The sample size is small, and the optimum dose of MMF is unknown.
DISCLOSURES:
The study had no outside funding. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
TOPLINE:
Anti-acid drugs used by patients with systemic sclerosis reduce the bioavailability of mycophenolate mofetil (MMF).
METHODOLOGY:
- Researchers conducted an open-label, pragmatic crossover study of 20 patients (all female) with systemic sclerosis at a single center who were on a stable MMF dose (1.5-2 g/day) for the last 3 months or more.
- Participants sequentially took MMF alone for 1 month, then with the H2 receptor blocker (HRB) ranitidine 300 mg/day in the second month, then with the proton pump inhibitor (PPI) esomeprazole 40 mg/day in the third month.
- Researchers measured the bioavailability of MMF in the patients during treatment with ranitidine or esomeprazole and the impact of the drugs on the total GI score of the UCLA Scleroderma Clinical Trial Consortium Gastrointestinal Tract 2.0 instrument.
- Patients were excluded if they were receiving co-prescription of cholestyramine, magnesium- or aluminum-containing antacids, and rifampicin; taking prednisolone-equivalent dose > 5 mg/day; taking MMF plus a PPI or an HRB at baseline; living with chronic kidney disease with a glomerular filtration rate < 30 mL/min; positive for HIV, HCV, or HBV; or living with end-stage lung disease or gastroduodenal ulcers.
TAKEAWAY:
- Mean estimated 12-hour area under curve levels of mycophenolic acid dropped by 32.7% (mean difference = 22.28 mcg h mL–1) when patients added esomeprazole, and they dipped by 21.97% (mean difference = 14.93 mcg h mL–1) when they added ranitidine vs. MMF alone.
- The pharmacokinetic parameter T-max did not differ significantly between MMF alone vs. MMF plus ranitidine but was significantly different with esomeprazole. C-max significantly declined with administration of ranitidine or esomeprazole vs. MMF alone.
- Total GI scores dipped when patients added esomeprazole or ranitidine.
IN PRACTICE:
In patients with significant gastroesophageal reflux disease symptoms who need to take MMF, management options may include monitoring MMF drug levels, switching to enteric-coated mycophenolate sodium, and spacing doses with anti-acid drugs.
SOURCE:
Glaxon Alex, MD, and colleagues from the Center for Arthritis and Rheumatism Excellence in Kochi, India, conducted the study, which was published online in Seminars in Arthritis & Rheumatism.
LIMITATIONS:
The sample size is small, and the optimum dose of MMF is unknown.
DISCLOSURES:
The study had no outside funding. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
TOPLINE:
Anti-acid drugs used by patients with systemic sclerosis reduce the bioavailability of mycophenolate mofetil (MMF).
METHODOLOGY:
- Researchers conducted an open-label, pragmatic crossover study of 20 patients (all female) with systemic sclerosis at a single center who were on a stable MMF dose (1.5-2 g/day) for the last 3 months or more.
- Participants sequentially took MMF alone for 1 month, then with the H2 receptor blocker (HRB) ranitidine 300 mg/day in the second month, then with the proton pump inhibitor (PPI) esomeprazole 40 mg/day in the third month.
- Researchers measured the bioavailability of MMF in the patients during treatment with ranitidine or esomeprazole and the impact of the drugs on the total GI score of the UCLA Scleroderma Clinical Trial Consortium Gastrointestinal Tract 2.0 instrument.
- Patients were excluded if they were receiving co-prescription of cholestyramine, magnesium- or aluminum-containing antacids, and rifampicin; taking prednisolone-equivalent dose > 5 mg/day; taking MMF plus a PPI or an HRB at baseline; living with chronic kidney disease with a glomerular filtration rate < 30 mL/min; positive for HIV, HCV, or HBV; or living with end-stage lung disease or gastroduodenal ulcers.
TAKEAWAY:
- Mean estimated 12-hour area under curve levels of mycophenolic acid dropped by 32.7% (mean difference = 22.28 mcg h mL–1) when patients added esomeprazole, and they dipped by 21.97% (mean difference = 14.93 mcg h mL–1) when they added ranitidine vs. MMF alone.
- The pharmacokinetic parameter T-max did not differ significantly between MMF alone vs. MMF plus ranitidine but was significantly different with esomeprazole. C-max significantly declined with administration of ranitidine or esomeprazole vs. MMF alone.
- Total GI scores dipped when patients added esomeprazole or ranitidine.
IN PRACTICE:
In patients with significant gastroesophageal reflux disease symptoms who need to take MMF, management options may include monitoring MMF drug levels, switching to enteric-coated mycophenolate sodium, and spacing doses with anti-acid drugs.
SOURCE:
Glaxon Alex, MD, and colleagues from the Center for Arthritis and Rheumatism Excellence in Kochi, India, conducted the study, which was published online in Seminars in Arthritis & Rheumatism.
LIMITATIONS:
The sample size is small, and the optimum dose of MMF is unknown.
DISCLOSURES:
The study had no outside funding. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Every click you make, the EHR is watching you
This transcript has been edited for clarity.
When I close my eyes and imagine what it is I do for a living, I see a computer screen.
I’m primarily a clinical researcher, so much of what I do is looking at statistical software, or, more recently, writing grant applications. But even when I think of my clinical duties, I see that computer screen.
The reason? The electronic health record (EHR) – the hot, beating heart of medical care in the modern era. Our most powerful tool and our greatest enemy.
The EHR records everything – not just the vital signs and lab values of our patients, not just our notes and billing codes. Everything. Every interaction we have is tracked and can be analyzed. The EHR is basically Sting in the song “Every Breath You Take.” Every click you make, it is watching you.
Researchers are leveraging that panopticon to give insight into something we don’t talk about frequently: the issue of racial bias in medicine. Is our true nature revealed by our interactions with the EHR?
We’re talking about this study in JAMA Network Open.
Researchers leveraged huge amounts of EHR data from two big academic medical centers, Vanderbilt University Medical Center and Northwestern University Medical Center. All told, there are data from nearly 250,000 hospitalizations here.
The researchers created a metric for EHR engagement. Basically, they summed the amount of clicks and other EHR interactions that occurred during the hospitalization, divided by the length of stay in days, to create a sort of average “engagement per day” metric. This number was categorized into four groups: low engagement, medium engagement, high engagement, and very high engagement.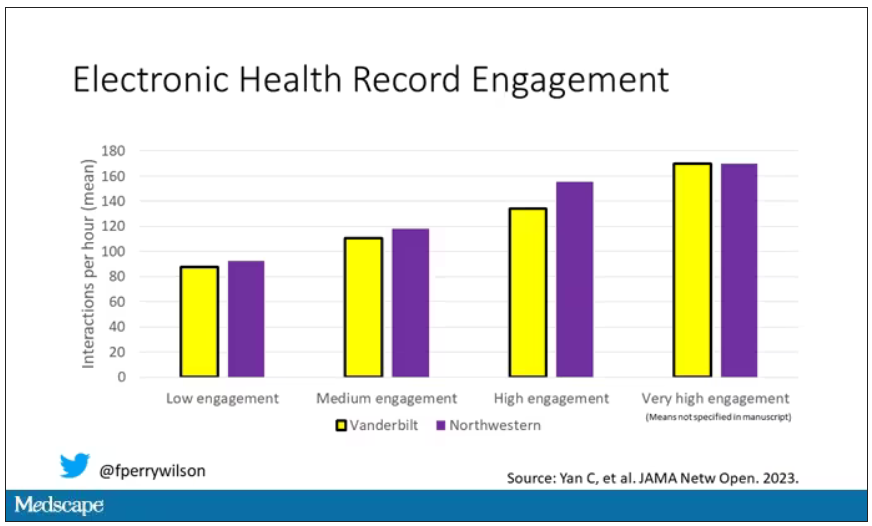
What factors would predict higher engagement? Well, , except among Black patients who actually got a bit more engagement.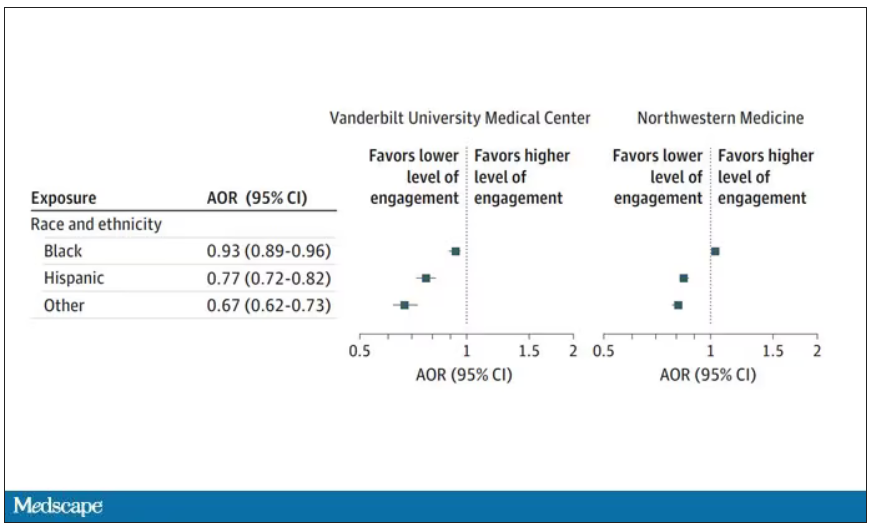
So, right away we need to be concerned about the obvious implications. Less engagement with the EHR may mean lower-quality care, right? Less attention to medical issues. And if that differs systematically by race, that’s a problem.
But we need to be careful here, because engagement in the health record is not random. Many factors would lead you to spend more time in one patient’s chart vs. another. Medical complexity is the most obvious one. The authors did their best to account for this, adjusting for patients’ age, sex, insurance status, comorbidity score, and social deprivation index based on their ZIP code. But notably, they did not account for the acuity of illness during the hospitalization. If individuals identifying as a minority were, all else being equal, less likely to be severely ill by the time they were hospitalized, you might see results like this.
The authors also restrict their analysis to individuals who were discharged alive. I’m not entirely clear why they made this choice. Most people don’t die in the hospital; the inpatient mortality rate at most centers is 1%-1.5%. But excluding those patients could potentially bias these results, especially if race is, all else being equal, a predictor of inpatient mortality, as some studies have shown.
But the truth is, these data aren’t coming out of nowhere; they don’t exist in a vacuum. Numerous studies demonstrate different intensity of care among minority vs. nonminority individuals. There is this study, which shows that minority populations are less likely to be placed on the liver transplant waitlist.
There is this study, which found that minority kids with type 1 diabetes were less likely to get insulin pumps than were their White counterparts. And this one, which showed that kids with acute appendicitis were less likely to get pain-control medications if they were Black.
This study shows that although life expectancy decreased across all races during the pandemic, it decreased the most among minority populations.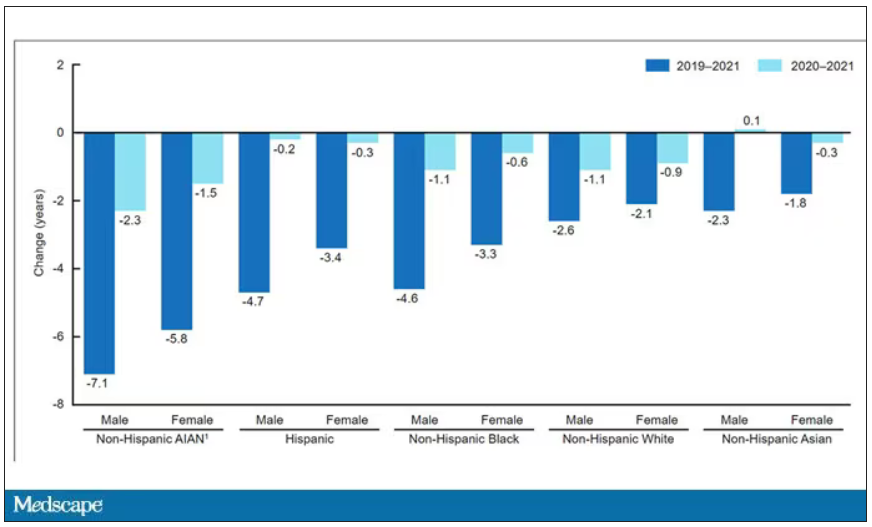
This list goes on. It’s why the CDC has called racism a “fundamental cause of ... disease.”
So, yes, it is clear that there are racial disparities in health care outcomes. It is clear that there are racial disparities in treatments. It is also clear that virtually every physician believes they deliver equitable care. Somewhere, this disconnect arises. Could the actions we take in the EHR reveal the unconscious biases we have? Does the all-seeing eye of the EHR see not only into our brains but into our hearts? And if it can, are we ready to confront what it sees?
F. Perry Wilson, MD, MSCE, is associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator in New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
When I close my eyes and imagine what it is I do for a living, I see a computer screen.
I’m primarily a clinical researcher, so much of what I do is looking at statistical software, or, more recently, writing grant applications. But even when I think of my clinical duties, I see that computer screen.
The reason? The electronic health record (EHR) – the hot, beating heart of medical care in the modern era. Our most powerful tool and our greatest enemy.
The EHR records everything – not just the vital signs and lab values of our patients, not just our notes and billing codes. Everything. Every interaction we have is tracked and can be analyzed. The EHR is basically Sting in the song “Every Breath You Take.” Every click you make, it is watching you.
Researchers are leveraging that panopticon to give insight into something we don’t talk about frequently: the issue of racial bias in medicine. Is our true nature revealed by our interactions with the EHR?
We’re talking about this study in JAMA Network Open.
Researchers leveraged huge amounts of EHR data from two big academic medical centers, Vanderbilt University Medical Center and Northwestern University Medical Center. All told, there are data from nearly 250,000 hospitalizations here.
The researchers created a metric for EHR engagement. Basically, they summed the amount of clicks and other EHR interactions that occurred during the hospitalization, divided by the length of stay in days, to create a sort of average “engagement per day” metric. This number was categorized into four groups: low engagement, medium engagement, high engagement, and very high engagement.
What factors would predict higher engagement? Well, , except among Black patients who actually got a bit more engagement.
So, right away we need to be concerned about the obvious implications. Less engagement with the EHR may mean lower-quality care, right? Less attention to medical issues. And if that differs systematically by race, that’s a problem.
But we need to be careful here, because engagement in the health record is not random. Many factors would lead you to spend more time in one patient’s chart vs. another. Medical complexity is the most obvious one. The authors did their best to account for this, adjusting for patients’ age, sex, insurance status, comorbidity score, and social deprivation index based on their ZIP code. But notably, they did not account for the acuity of illness during the hospitalization. If individuals identifying as a minority were, all else being equal, less likely to be severely ill by the time they were hospitalized, you might see results like this.
The authors also restrict their analysis to individuals who were discharged alive. I’m not entirely clear why they made this choice. Most people don’t die in the hospital; the inpatient mortality rate at most centers is 1%-1.5%. But excluding those patients could potentially bias these results, especially if race is, all else being equal, a predictor of inpatient mortality, as some studies have shown.
But the truth is, these data aren’t coming out of nowhere; they don’t exist in a vacuum. Numerous studies demonstrate different intensity of care among minority vs. nonminority individuals. There is this study, which shows that minority populations are less likely to be placed on the liver transplant waitlist.
There is this study, which found that minority kids with type 1 diabetes were less likely to get insulin pumps than were their White counterparts. And this one, which showed that kids with acute appendicitis were less likely to get pain-control medications if they were Black.
This study shows that although life expectancy decreased across all races during the pandemic, it decreased the most among minority populations.
This list goes on. It’s why the CDC has called racism a “fundamental cause of ... disease.”
So, yes, it is clear that there are racial disparities in health care outcomes. It is clear that there are racial disparities in treatments. It is also clear that virtually every physician believes they deliver equitable care. Somewhere, this disconnect arises. Could the actions we take in the EHR reveal the unconscious biases we have? Does the all-seeing eye of the EHR see not only into our brains but into our hearts? And if it can, are we ready to confront what it sees?
F. Perry Wilson, MD, MSCE, is associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator in New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
When I close my eyes and imagine what it is I do for a living, I see a computer screen.
I’m primarily a clinical researcher, so much of what I do is looking at statistical software, or, more recently, writing grant applications. But even when I think of my clinical duties, I see that computer screen.
The reason? The electronic health record (EHR) – the hot, beating heart of medical care in the modern era. Our most powerful tool and our greatest enemy.
The EHR records everything – not just the vital signs and lab values of our patients, not just our notes and billing codes. Everything. Every interaction we have is tracked and can be analyzed. The EHR is basically Sting in the song “Every Breath You Take.” Every click you make, it is watching you.
Researchers are leveraging that panopticon to give insight into something we don’t talk about frequently: the issue of racial bias in medicine. Is our true nature revealed by our interactions with the EHR?
We’re talking about this study in JAMA Network Open.
Researchers leveraged huge amounts of EHR data from two big academic medical centers, Vanderbilt University Medical Center and Northwestern University Medical Center. All told, there are data from nearly 250,000 hospitalizations here.
The researchers created a metric for EHR engagement. Basically, they summed the amount of clicks and other EHR interactions that occurred during the hospitalization, divided by the length of stay in days, to create a sort of average “engagement per day” metric. This number was categorized into four groups: low engagement, medium engagement, high engagement, and very high engagement.
What factors would predict higher engagement? Well, , except among Black patients who actually got a bit more engagement.
So, right away we need to be concerned about the obvious implications. Less engagement with the EHR may mean lower-quality care, right? Less attention to medical issues. And if that differs systematically by race, that’s a problem.
But we need to be careful here, because engagement in the health record is not random. Many factors would lead you to spend more time in one patient’s chart vs. another. Medical complexity is the most obvious one. The authors did their best to account for this, adjusting for patients’ age, sex, insurance status, comorbidity score, and social deprivation index based on their ZIP code. But notably, they did not account for the acuity of illness during the hospitalization. If individuals identifying as a minority were, all else being equal, less likely to be severely ill by the time they were hospitalized, you might see results like this.
The authors also restrict their analysis to individuals who were discharged alive. I’m not entirely clear why they made this choice. Most people don’t die in the hospital; the inpatient mortality rate at most centers is 1%-1.5%. But excluding those patients could potentially bias these results, especially if race is, all else being equal, a predictor of inpatient mortality, as some studies have shown.
But the truth is, these data aren’t coming out of nowhere; they don’t exist in a vacuum. Numerous studies demonstrate different intensity of care among minority vs. nonminority individuals. There is this study, which shows that minority populations are less likely to be placed on the liver transplant waitlist.
There is this study, which found that minority kids with type 1 diabetes were less likely to get insulin pumps than were their White counterparts. And this one, which showed that kids with acute appendicitis were less likely to get pain-control medications if they were Black.
This study shows that although life expectancy decreased across all races during the pandemic, it decreased the most among minority populations.
This list goes on. It’s why the CDC has called racism a “fundamental cause of ... disease.”
So, yes, it is clear that there are racial disparities in health care outcomes. It is clear that there are racial disparities in treatments. It is also clear that virtually every physician believes they deliver equitable care. Somewhere, this disconnect arises. Could the actions we take in the EHR reveal the unconscious biases we have? Does the all-seeing eye of the EHR see not only into our brains but into our hearts? And if it can, are we ready to confront what it sees?
F. Perry Wilson, MD, MSCE, is associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator in New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Are women and men with rheumatism treated equally?
LEIPZIG, GERMANY – Women eat more healthily, visit their physician more often, and accept offers of prophylactic treatment more frequently than their male counterparts. Nevertheless, they are generally diagnosed with a rheumatic disease much later. “With systemic sclerosis for example, diagnosis occurs a whole year later than for male patients,” said Uta Kiltz, MD, senior physician at the Ruhrgebiet Rheumatism Center in Bochum, Germany, at a press conference for the annual congress of the German Society for Rheumatology.
In addition, certain markers and antibodies can be detected earlier in men’s blood – for example in systemic sclerosis. “What’s more, women exhibit a more diverse array of symptoms, which can make an unequivocal diagnosis difficult,” Dr. Kiltz explained.
Differences between the sexes in terms of disease progression and clinical presentation have been described for most rheumatic diseases. Roughly speaking, women often exhibit a much wider range of symptoms and report a higher disease burden, whereas men tend to experience a more severe progression of the disease.
Comorbidities also occur at different rates between the sexes. Whereas women with rheumatoid arthritis suffer more frequently from osteoporosis and depression, men are more likely to develop cardiovascular diseases and diabetes.
Gender-sensitive approach
Like Dr. Kiltz, Susanna Späthling-Mestekemper, MD, PhD, of the Munich-Pasing (Germany) Rheumatology Practice, also advocates a gender-sensitive approach to diagnosis and therapy. Dr. Späthling-Mestekemper referred to this during the conference, stating that women are still treated more poorly than men. The difference in treatment quality results from gaps in knowledge in the following areas:
- Sex-specific differences in the diagnosis and therapy of rheumatic diseases and in basic and clinical research
- Sex-specific differences in communication between male and female patients and between male and female physicians.
Dr. Späthling-Mestekemper used axial spondyloarthritis (axSpA) as a “prominent example” of false diagnoses. “Men more commonly fulfill the modified New York criteria – involvement of the axial skeleton, the lumbar spine, and increasing radiological progression.”
In contrast, women with axSpA exhibit the following differences:
- It is more likely for the cervical spine to be affected.
- Women are more likely to suffer from peripheral joint involvement.
- They suffer more from whole body pain.
- They have fatigue and exhaustion.
- They exhibit fewer humoral signs of inflammation (lower C-reactive protein).
- They are rarely HLA-B27 positive.
“We also have to completely rethink how we make the diagnosis in women,” said Dr. Späthling-Mestekemper. The current approach leads to women with axSpA being diagnosed much later than men. “Depending on the study, the difference can range from 7 months to 2 years,” according to Dr. Späthling-Mestekemper.
A 2018 Spanish study reported that the most common incorrect diagnoses in women with axSpA were sciatica, osteoarthritis, and fibromyalgia.
However, it is not just in axSpA that there are significant differences between men and women. There is evidence that women with systemic lupus erythematosus suffer more from musculoskeletal symptoms, while men with lupus exhibit more severe organ involvement (especially more serositis and nephritis).
For systemic sclerosis, women have the higher survival rate. They also exhibit skin involvement more frequently. Men, however, are more likely to have organ involvement, especially with the lungs.
TNF blockers
Using the example of axSpA, Dr. Späthling-Mestekemper also showed that men and women respond differently to tumor necrosis factor (TNF) blocker therapy. “The duration of therapy with TNF blockers is shorter for women: 33.4 months versus 44.9 months. They respond less to this therapy; they stop and change more frequently.”
Data from March 2023 show that, in contrast, there is no evidence of a difference in response to Janus kinase inhibitor treatment.
The presence of enthesitis has been discussed as one reason for the worse response to TNF blockers in women, since they have it more often than men do. “In fact, a better response to TNF blockers is associated with HLA-B27 positivity, with the absence of enthesitis and with TNF blocker naivety. In women, higher fat-mass index could also play a part, or even abdominal weight gain, which also increases in women after menopause,” said Dr. Späthling-Mestekemper.
She mentioned the following other potential reasons for a delayed therapy response to biological drugs in women:
- Genetic, physical, or hormonal causes
- Widespread pain or fibromyalgia
- Late diagnosis or late application of therapy, which lowers the chances of remission.
Even the science itself has shown the following sex-specific shortcomings:
- Disregarding sex-specific differences in animal-experimental studies (which, until recently, were only conducted in male mice to avoid hormone fluctuations)
- Women in clinical studies are still underrepresented: only 37% of the populations in phase 3 studies are women; 64% of studies do not describe any sex-specific differences
- Most of the data come from epidemiological analyses (not from basic research)
- Gaps in medical textbooks
Communication differences
Female patients are looking for explanations, whereas male patients describe specific symptoms. Female physicians talk, while male physicians treat. They sound like stereotypes, but they have been substantiated in multiple studies, said Dr. Späthling-Mestekemper. In general, the study results show that male patients behave in the following ways:
- Describe their symptoms in terms of specifics
- Do not like to admit having mental health issues
- Are three to five times more likely to commit suicide because of depression than women
On the other hand, female patients behave in the following ways:
- Look for an explanation for their symptoms
- Often do not have their physical symptoms taken seriously
- Are often pushed in a psychosomatic direction.
Female physicians focus on the following questions:
- Prevention, communication, shared decision-making, open-ended questions, “positive” discussions, patient self-management (chronic diseases such as diabetes: female physicians are better at reaching the therapy goals set by the ADA guidelines than male physicians)
- Psychosocial situations: consultations last 1 minute longer (10%).
Male physicians focus on the following questions:
- Medical history
- Physical examination (cardiac catheterizations after a heart attack are arranged much more commonly by male rather than female physicians)
- Diagnostics
Recognition and training
A large-scale surgical study in 2021 made a few waves. The study analyzed whether it makes a difference if women are operated on by men or by women. The results showed that women who had been operated on by men exhibited a higher level of risk after the surgery, compared with men who had been operated on by men or by women. The risk took the following forms:
- 15% higher risk for a worse surgery result
- 16% higher risk for complications
- 11% higher risk for repeat hospitalization
- 20% higher risk for a longer period of hospitalization
- 32% higher risk for mortality
The study authors provided the following potential reasons for these differences:
- Male physicians underestimate the severity of symptoms in their female patients
- Women are less comfortable indicating their postoperative pain to a male physician
- Different working style and treatment decisions between female and male physicians
- Unconsciously incorporated role patterns and preconceptions
“Our potential solutions are recognition and training. We need a personalized style of medicine; we need to have a closer look. We owe our male and female patients as much,” said Dr. Späthling-Mestekemper.
This article was translated from the Medscape German Edition and a version appeared on Medscape.com.
LEIPZIG, GERMANY – Women eat more healthily, visit their physician more often, and accept offers of prophylactic treatment more frequently than their male counterparts. Nevertheless, they are generally diagnosed with a rheumatic disease much later. “With systemic sclerosis for example, diagnosis occurs a whole year later than for male patients,” said Uta Kiltz, MD, senior physician at the Ruhrgebiet Rheumatism Center in Bochum, Germany, at a press conference for the annual congress of the German Society for Rheumatology.
In addition, certain markers and antibodies can be detected earlier in men’s blood – for example in systemic sclerosis. “What’s more, women exhibit a more diverse array of symptoms, which can make an unequivocal diagnosis difficult,” Dr. Kiltz explained.
Differences between the sexes in terms of disease progression and clinical presentation have been described for most rheumatic diseases. Roughly speaking, women often exhibit a much wider range of symptoms and report a higher disease burden, whereas men tend to experience a more severe progression of the disease.
Comorbidities also occur at different rates between the sexes. Whereas women with rheumatoid arthritis suffer more frequently from osteoporosis and depression, men are more likely to develop cardiovascular diseases and diabetes.
Gender-sensitive approach
Like Dr. Kiltz, Susanna Späthling-Mestekemper, MD, PhD, of the Munich-Pasing (Germany) Rheumatology Practice, also advocates a gender-sensitive approach to diagnosis and therapy. Dr. Späthling-Mestekemper referred to this during the conference, stating that women are still treated more poorly than men. The difference in treatment quality results from gaps in knowledge in the following areas:
- Sex-specific differences in the diagnosis and therapy of rheumatic diseases and in basic and clinical research
- Sex-specific differences in communication between male and female patients and between male and female physicians.
Dr. Späthling-Mestekemper used axial spondyloarthritis (axSpA) as a “prominent example” of false diagnoses. “Men more commonly fulfill the modified New York criteria – involvement of the axial skeleton, the lumbar spine, and increasing radiological progression.”
In contrast, women with axSpA exhibit the following differences:
- It is more likely for the cervical spine to be affected.
- Women are more likely to suffer from peripheral joint involvement.
- They suffer more from whole body pain.
- They have fatigue and exhaustion.
- They exhibit fewer humoral signs of inflammation (lower C-reactive protein).
- They are rarely HLA-B27 positive.
“We also have to completely rethink how we make the diagnosis in women,” said Dr. Späthling-Mestekemper. The current approach leads to women with axSpA being diagnosed much later than men. “Depending on the study, the difference can range from 7 months to 2 years,” according to Dr. Späthling-Mestekemper.
A 2018 Spanish study reported that the most common incorrect diagnoses in women with axSpA were sciatica, osteoarthritis, and fibromyalgia.
However, it is not just in axSpA that there are significant differences between men and women. There is evidence that women with systemic lupus erythematosus suffer more from musculoskeletal symptoms, while men with lupus exhibit more severe organ involvement (especially more serositis and nephritis).
For systemic sclerosis, women have the higher survival rate. They also exhibit skin involvement more frequently. Men, however, are more likely to have organ involvement, especially with the lungs.
TNF blockers
Using the example of axSpA, Dr. Späthling-Mestekemper also showed that men and women respond differently to tumor necrosis factor (TNF) blocker therapy. “The duration of therapy with TNF blockers is shorter for women: 33.4 months versus 44.9 months. They respond less to this therapy; they stop and change more frequently.”
Data from March 2023 show that, in contrast, there is no evidence of a difference in response to Janus kinase inhibitor treatment.
The presence of enthesitis has been discussed as one reason for the worse response to TNF blockers in women, since they have it more often than men do. “In fact, a better response to TNF blockers is associated with HLA-B27 positivity, with the absence of enthesitis and with TNF blocker naivety. In women, higher fat-mass index could also play a part, or even abdominal weight gain, which also increases in women after menopause,” said Dr. Späthling-Mestekemper.
She mentioned the following other potential reasons for a delayed therapy response to biological drugs in women:
- Genetic, physical, or hormonal causes
- Widespread pain or fibromyalgia
- Late diagnosis or late application of therapy, which lowers the chances of remission.
Even the science itself has shown the following sex-specific shortcomings:
- Disregarding sex-specific differences in animal-experimental studies (which, until recently, were only conducted in male mice to avoid hormone fluctuations)
- Women in clinical studies are still underrepresented: only 37% of the populations in phase 3 studies are women; 64% of studies do not describe any sex-specific differences
- Most of the data come from epidemiological analyses (not from basic research)
- Gaps in medical textbooks
Communication differences
Female patients are looking for explanations, whereas male patients describe specific symptoms. Female physicians talk, while male physicians treat. They sound like stereotypes, but they have been substantiated in multiple studies, said Dr. Späthling-Mestekemper. In general, the study results show that male patients behave in the following ways:
- Describe their symptoms in terms of specifics
- Do not like to admit having mental health issues
- Are three to five times more likely to commit suicide because of depression than women
On the other hand, female patients behave in the following ways:
- Look for an explanation for their symptoms
- Often do not have their physical symptoms taken seriously
- Are often pushed in a psychosomatic direction.
Female physicians focus on the following questions:
- Prevention, communication, shared decision-making, open-ended questions, “positive” discussions, patient self-management (chronic diseases such as diabetes: female physicians are better at reaching the therapy goals set by the ADA guidelines than male physicians)
- Psychosocial situations: consultations last 1 minute longer (10%).
Male physicians focus on the following questions:
- Medical history
- Physical examination (cardiac catheterizations after a heart attack are arranged much more commonly by male rather than female physicians)
- Diagnostics
Recognition and training
A large-scale surgical study in 2021 made a few waves. The study analyzed whether it makes a difference if women are operated on by men or by women. The results showed that women who had been operated on by men exhibited a higher level of risk after the surgery, compared with men who had been operated on by men or by women. The risk took the following forms:
- 15% higher risk for a worse surgery result
- 16% higher risk for complications
- 11% higher risk for repeat hospitalization
- 20% higher risk for a longer period of hospitalization
- 32% higher risk for mortality
The study authors provided the following potential reasons for these differences:
- Male physicians underestimate the severity of symptoms in their female patients
- Women are less comfortable indicating their postoperative pain to a male physician
- Different working style and treatment decisions between female and male physicians
- Unconsciously incorporated role patterns and preconceptions
“Our potential solutions are recognition and training. We need a personalized style of medicine; we need to have a closer look. We owe our male and female patients as much,” said Dr. Späthling-Mestekemper.
This article was translated from the Medscape German Edition and a version appeared on Medscape.com.
LEIPZIG, GERMANY – Women eat more healthily, visit their physician more often, and accept offers of prophylactic treatment more frequently than their male counterparts. Nevertheless, they are generally diagnosed with a rheumatic disease much later. “With systemic sclerosis for example, diagnosis occurs a whole year later than for male patients,” said Uta Kiltz, MD, senior physician at the Ruhrgebiet Rheumatism Center in Bochum, Germany, at a press conference for the annual congress of the German Society for Rheumatology.
In addition, certain markers and antibodies can be detected earlier in men’s blood – for example in systemic sclerosis. “What’s more, women exhibit a more diverse array of symptoms, which can make an unequivocal diagnosis difficult,” Dr. Kiltz explained.
Differences between the sexes in terms of disease progression and clinical presentation have been described for most rheumatic diseases. Roughly speaking, women often exhibit a much wider range of symptoms and report a higher disease burden, whereas men tend to experience a more severe progression of the disease.
Comorbidities also occur at different rates between the sexes. Whereas women with rheumatoid arthritis suffer more frequently from osteoporosis and depression, men are more likely to develop cardiovascular diseases and diabetes.
Gender-sensitive approach
Like Dr. Kiltz, Susanna Späthling-Mestekemper, MD, PhD, of the Munich-Pasing (Germany) Rheumatology Practice, also advocates a gender-sensitive approach to diagnosis and therapy. Dr. Späthling-Mestekemper referred to this during the conference, stating that women are still treated more poorly than men. The difference in treatment quality results from gaps in knowledge in the following areas:
- Sex-specific differences in the diagnosis and therapy of rheumatic diseases and in basic and clinical research
- Sex-specific differences in communication between male and female patients and between male and female physicians.
Dr. Späthling-Mestekemper used axial spondyloarthritis (axSpA) as a “prominent example” of false diagnoses. “Men more commonly fulfill the modified New York criteria – involvement of the axial skeleton, the lumbar spine, and increasing radiological progression.”
In contrast, women with axSpA exhibit the following differences:
- It is more likely for the cervical spine to be affected.
- Women are more likely to suffer from peripheral joint involvement.
- They suffer more from whole body pain.
- They have fatigue and exhaustion.
- They exhibit fewer humoral signs of inflammation (lower C-reactive protein).
- They are rarely HLA-B27 positive.
“We also have to completely rethink how we make the diagnosis in women,” said Dr. Späthling-Mestekemper. The current approach leads to women with axSpA being diagnosed much later than men. “Depending on the study, the difference can range from 7 months to 2 years,” according to Dr. Späthling-Mestekemper.
A 2018 Spanish study reported that the most common incorrect diagnoses in women with axSpA were sciatica, osteoarthritis, and fibromyalgia.
However, it is not just in axSpA that there are significant differences between men and women. There is evidence that women with systemic lupus erythematosus suffer more from musculoskeletal symptoms, while men with lupus exhibit more severe organ involvement (especially more serositis and nephritis).
For systemic sclerosis, women have the higher survival rate. They also exhibit skin involvement more frequently. Men, however, are more likely to have organ involvement, especially with the lungs.
TNF blockers
Using the example of axSpA, Dr. Späthling-Mestekemper also showed that men and women respond differently to tumor necrosis factor (TNF) blocker therapy. “The duration of therapy with TNF blockers is shorter for women: 33.4 months versus 44.9 months. They respond less to this therapy; they stop and change more frequently.”
Data from March 2023 show that, in contrast, there is no evidence of a difference in response to Janus kinase inhibitor treatment.
The presence of enthesitis has been discussed as one reason for the worse response to TNF blockers in women, since they have it more often than men do. “In fact, a better response to TNF blockers is associated with HLA-B27 positivity, with the absence of enthesitis and with TNF blocker naivety. In women, higher fat-mass index could also play a part, or even abdominal weight gain, which also increases in women after menopause,” said Dr. Späthling-Mestekemper.
She mentioned the following other potential reasons for a delayed therapy response to biological drugs in women:
- Genetic, physical, or hormonal causes
- Widespread pain or fibromyalgia
- Late diagnosis or late application of therapy, which lowers the chances of remission.
Even the science itself has shown the following sex-specific shortcomings:
- Disregarding sex-specific differences in animal-experimental studies (which, until recently, were only conducted in male mice to avoid hormone fluctuations)
- Women in clinical studies are still underrepresented: only 37% of the populations in phase 3 studies are women; 64% of studies do not describe any sex-specific differences
- Most of the data come from epidemiological analyses (not from basic research)
- Gaps in medical textbooks
Communication differences
Female patients are looking for explanations, whereas male patients describe specific symptoms. Female physicians talk, while male physicians treat. They sound like stereotypes, but they have been substantiated in multiple studies, said Dr. Späthling-Mestekemper. In general, the study results show that male patients behave in the following ways:
- Describe their symptoms in terms of specifics
- Do not like to admit having mental health issues
- Are three to five times more likely to commit suicide because of depression than women
On the other hand, female patients behave in the following ways:
- Look for an explanation for their symptoms
- Often do not have their physical symptoms taken seriously
- Are often pushed in a psychosomatic direction.
Female physicians focus on the following questions:
- Prevention, communication, shared decision-making, open-ended questions, “positive” discussions, patient self-management (chronic diseases such as diabetes: female physicians are better at reaching the therapy goals set by the ADA guidelines than male physicians)
- Psychosocial situations: consultations last 1 minute longer (10%).
Male physicians focus on the following questions:
- Medical history
- Physical examination (cardiac catheterizations after a heart attack are arranged much more commonly by male rather than female physicians)
- Diagnostics
Recognition and training
A large-scale surgical study in 2021 made a few waves. The study analyzed whether it makes a difference if women are operated on by men or by women. The results showed that women who had been operated on by men exhibited a higher level of risk after the surgery, compared with men who had been operated on by men or by women. The risk took the following forms:
- 15% higher risk for a worse surgery result
- 16% higher risk for complications
- 11% higher risk for repeat hospitalization
- 20% higher risk for a longer period of hospitalization
- 32% higher risk for mortality
The study authors provided the following potential reasons for these differences:
- Male physicians underestimate the severity of symptoms in their female patients
- Women are less comfortable indicating their postoperative pain to a male physician
- Different working style and treatment decisions between female and male physicians
- Unconsciously incorporated role patterns and preconceptions
“Our potential solutions are recognition and training. We need a personalized style of medicine; we need to have a closer look. We owe our male and female patients as much,” said Dr. Späthling-Mestekemper.
This article was translated from the Medscape German Edition and a version appeared on Medscape.com.
AT THE GERMAN RHEUMATOLOGY CONGRESS 2023




