User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'medstat-accordion-set article-series')]
Five ways to avert a malpractice lawsuit with better EHR techniques
Although most physicians have gotten used to working with EHRs, despite their irritations, the use of EHRs has contributed to a growing number of malpractice lawsuits. Defense attorneys say that
According to a study in the Journal of Patient Safety, more than 30% of all EHR-related malpractice cases are associated with medication errors; 28% with diagnosis; and 31% with a complication of treatment, such as entering wrong information, entering information in the wrong place, and overlooking EHR flags and warnings for interactions or contraindications.
The study gave these examples of EHR-related errors that led to patient harm and ultimately to malpractice lawsuits:
- A discharge order omitted a patient’s medication that prevented strokes; the patient had a stroke days later.
- An electronic order for morphine failed to state the upper dose limit; the patient died.
- A physician meant to click on “discontinue” for an anticoagulant but mistakenly clicked on “continue” for home use.
Catching potential issues such as drug interactions or critical medical history that should inform treatment is more important than ever. “We know from safety engineering principles that just relying on vigilance is not a long-term safety strategy,” says Aaron Zach Hettinger, MD, chief research information officer at MedStar Health Research Institute, Washington, D.C. “So, it’s critical that we design these safe systems and leverage the data that’s in them.”
Here are five smart EHR practices to help protect your patients’ health and your own liability.
1. Double-check dropdown boxes
When it comes to user error, it’s easy to click the wrong choice from a drop-down menu. Better to take the time to explain your answer in a box, even if it takes a few more minutes. Or if you are choosing from a menu, proofread any information it auto-fills in the chart.
Dr. Hettinger says you can strike a balance between these templated approaches to diagnosis and long-term care by working with third-party systems and your organization or vendor IT department to help with follow-up questions to keep populated data in check.
“Make sure you have a back-end system that can help monitor that structured data,” says Dr. Hettinger. Structured data are the patient’s demographic information, like name, address, age, height, weight, vital signs, and data elements like diagnosis, medications, and lab results. “Wherever you can leverage the underlying tools that are part of the electronic health record to make sure that we’re constantly checking the right results, that helps reduce the workload so that clinicians can focus on taking care of the patients and doing the right thing and not be as focused on entering data into the system.”
2. Supplement EHR notes with direct communication
The failure to diagnose cancer because one physician doesn’t know what another physician saw in an imaging report is one of the most common claims in the cases he tries, says Aaron Boeder, a plaintiff’s medical negligence lawyer in Chicago.
Physicians often assume that if they put a note in the electronic chart, others will look for it, but Mr. Boeder says it’s far more prudent to communicate directly.
“Let’s say a radiologist interprets a scan and sees what might be cancer,” he says. “If the ordering doctor is an orthopedist who’s ordered a CT scan for DVT, there’s going to be a report for that scan. It’s going to get auto-populated back into that physician’s note,” says Mr. Boeder.
The physician may or may not look at it, but it will be in their note, and they’re supposed to follow up on it because they ordered the scan. “But they may not follow up on it, and they may not get a call from the radiologist,” he says.
“Next thing you know, 2 or 3 years later, that patient is diagnosed with very advanced cancer.”
3. Tailor auto-fill information to your common practices
Suppose, as a physician, you find that you need to change a default setting time and time again. Dr. Hettinger says it’s worth your time to take an extra couple of minutes to work with your vendor or your health system to try and make changes to auto-population settings that align with your practices.
“Let’s say a default dose of 20 milligrams of a medication is what automatically pops up, but in reality, your practice is to use a smaller dose because it’s safer, even though they’re all within the acceptable realm of what you would order,” he says. “Rather than have the default to the higher dose, see if you can change the default to a lower dose. And that way, you don’t have to catch yourself every time.”
If your auto-fills are amounts that constantly need changing, an interruption could easily knock you off course before you make that correction.
“If there are ways to have the system defaults be safer or more in line with your clinical practice, and especially across a group, then you’re designing a safer system and not relying on vigilance or memory prone to interruptions,” says Dr. Hettinger.
4. Curb the copy and paste
It’s tempting to copy a note from a previous patient visit and make only minimal changes as needed, but you risk including outdated information if you do. Even if you’re repeating questions asked by the intake nurse, it is safer to not to rely on that information, says Beth Kanik, a defense medical malpractice attorney in Atlanta.
“If it later goes into litigation, the argument then becomes that it looks like you didn’t do your job,” says Ms. Kanik. “Instead, try to ask questions in a way that would elicit responses that may be a little different than what the nurse got, so that it’s clear you asked the questions and didn’t just simply rely upon someone else’s information.”
5. Separate typing from listening
While EHR may be an excellent tool for data collection and safety checking, it’s not a stand-in for doctor-patient interaction. As technology practices push medicine toward more and more efficiency, Mr. Boeder says it’s most often listening over all else that makes the difference in the quality of care. And good listening requires full attention.
“A real concern for physicians is the number of visits they’re expected to accomplish in a set amount of time,” says Mr. Boeder. “Often this translates into a doctor talking to a patient while typing notes or while reading a note from the last time the patient was in.”
Taking the time to pause after entering data and briefly reviewing your understanding of what your patient has told you can be invaluable and may save you – and your patient – problems later.
“In so many cases, it comes down to people not being heard,” says Mr. Boeder. “So listen to what your patients are saying.”
A version of this article first appeared on Medscape.com.
Although most physicians have gotten used to working with EHRs, despite their irritations, the use of EHRs has contributed to a growing number of malpractice lawsuits. Defense attorneys say that
According to a study in the Journal of Patient Safety, more than 30% of all EHR-related malpractice cases are associated with medication errors; 28% with diagnosis; and 31% with a complication of treatment, such as entering wrong information, entering information in the wrong place, and overlooking EHR flags and warnings for interactions or contraindications.
The study gave these examples of EHR-related errors that led to patient harm and ultimately to malpractice lawsuits:
- A discharge order omitted a patient’s medication that prevented strokes; the patient had a stroke days later.
- An electronic order for morphine failed to state the upper dose limit; the patient died.
- A physician meant to click on “discontinue” for an anticoagulant but mistakenly clicked on “continue” for home use.
Catching potential issues such as drug interactions or critical medical history that should inform treatment is more important than ever. “We know from safety engineering principles that just relying on vigilance is not a long-term safety strategy,” says Aaron Zach Hettinger, MD, chief research information officer at MedStar Health Research Institute, Washington, D.C. “So, it’s critical that we design these safe systems and leverage the data that’s in them.”
Here are five smart EHR practices to help protect your patients’ health and your own liability.
1. Double-check dropdown boxes
When it comes to user error, it’s easy to click the wrong choice from a drop-down menu. Better to take the time to explain your answer in a box, even if it takes a few more minutes. Or if you are choosing from a menu, proofread any information it auto-fills in the chart.
Dr. Hettinger says you can strike a balance between these templated approaches to diagnosis and long-term care by working with third-party systems and your organization or vendor IT department to help with follow-up questions to keep populated data in check.
“Make sure you have a back-end system that can help monitor that structured data,” says Dr. Hettinger. Structured data are the patient’s demographic information, like name, address, age, height, weight, vital signs, and data elements like diagnosis, medications, and lab results. “Wherever you can leverage the underlying tools that are part of the electronic health record to make sure that we’re constantly checking the right results, that helps reduce the workload so that clinicians can focus on taking care of the patients and doing the right thing and not be as focused on entering data into the system.”
2. Supplement EHR notes with direct communication
The failure to diagnose cancer because one physician doesn’t know what another physician saw in an imaging report is one of the most common claims in the cases he tries, says Aaron Boeder, a plaintiff’s medical negligence lawyer in Chicago.
Physicians often assume that if they put a note in the electronic chart, others will look for it, but Mr. Boeder says it’s far more prudent to communicate directly.
“Let’s say a radiologist interprets a scan and sees what might be cancer,” he says. “If the ordering doctor is an orthopedist who’s ordered a CT scan for DVT, there’s going to be a report for that scan. It’s going to get auto-populated back into that physician’s note,” says Mr. Boeder.
The physician may or may not look at it, but it will be in their note, and they’re supposed to follow up on it because they ordered the scan. “But they may not follow up on it, and they may not get a call from the radiologist,” he says.
“Next thing you know, 2 or 3 years later, that patient is diagnosed with very advanced cancer.”
3. Tailor auto-fill information to your common practices
Suppose, as a physician, you find that you need to change a default setting time and time again. Dr. Hettinger says it’s worth your time to take an extra couple of minutes to work with your vendor or your health system to try and make changes to auto-population settings that align with your practices.
“Let’s say a default dose of 20 milligrams of a medication is what automatically pops up, but in reality, your practice is to use a smaller dose because it’s safer, even though they’re all within the acceptable realm of what you would order,” he says. “Rather than have the default to the higher dose, see if you can change the default to a lower dose. And that way, you don’t have to catch yourself every time.”
If your auto-fills are amounts that constantly need changing, an interruption could easily knock you off course before you make that correction.
“If there are ways to have the system defaults be safer or more in line with your clinical practice, and especially across a group, then you’re designing a safer system and not relying on vigilance or memory prone to interruptions,” says Dr. Hettinger.
4. Curb the copy and paste
It’s tempting to copy a note from a previous patient visit and make only minimal changes as needed, but you risk including outdated information if you do. Even if you’re repeating questions asked by the intake nurse, it is safer to not to rely on that information, says Beth Kanik, a defense medical malpractice attorney in Atlanta.
“If it later goes into litigation, the argument then becomes that it looks like you didn’t do your job,” says Ms. Kanik. “Instead, try to ask questions in a way that would elicit responses that may be a little different than what the nurse got, so that it’s clear you asked the questions and didn’t just simply rely upon someone else’s information.”
5. Separate typing from listening
While EHR may be an excellent tool for data collection and safety checking, it’s not a stand-in for doctor-patient interaction. As technology practices push medicine toward more and more efficiency, Mr. Boeder says it’s most often listening over all else that makes the difference in the quality of care. And good listening requires full attention.
“A real concern for physicians is the number of visits they’re expected to accomplish in a set amount of time,” says Mr. Boeder. “Often this translates into a doctor talking to a patient while typing notes or while reading a note from the last time the patient was in.”
Taking the time to pause after entering data and briefly reviewing your understanding of what your patient has told you can be invaluable and may save you – and your patient – problems later.
“In so many cases, it comes down to people not being heard,” says Mr. Boeder. “So listen to what your patients are saying.”
A version of this article first appeared on Medscape.com.
Although most physicians have gotten used to working with EHRs, despite their irritations, the use of EHRs has contributed to a growing number of malpractice lawsuits. Defense attorneys say that
According to a study in the Journal of Patient Safety, more than 30% of all EHR-related malpractice cases are associated with medication errors; 28% with diagnosis; and 31% with a complication of treatment, such as entering wrong information, entering information in the wrong place, and overlooking EHR flags and warnings for interactions or contraindications.
The study gave these examples of EHR-related errors that led to patient harm and ultimately to malpractice lawsuits:
- A discharge order omitted a patient’s medication that prevented strokes; the patient had a stroke days later.
- An electronic order for morphine failed to state the upper dose limit; the patient died.
- A physician meant to click on “discontinue” for an anticoagulant but mistakenly clicked on “continue” for home use.
Catching potential issues such as drug interactions or critical medical history that should inform treatment is more important than ever. “We know from safety engineering principles that just relying on vigilance is not a long-term safety strategy,” says Aaron Zach Hettinger, MD, chief research information officer at MedStar Health Research Institute, Washington, D.C. “So, it’s critical that we design these safe systems and leverage the data that’s in them.”
Here are five smart EHR practices to help protect your patients’ health and your own liability.
1. Double-check dropdown boxes
When it comes to user error, it’s easy to click the wrong choice from a drop-down menu. Better to take the time to explain your answer in a box, even if it takes a few more minutes. Or if you are choosing from a menu, proofread any information it auto-fills in the chart.
Dr. Hettinger says you can strike a balance between these templated approaches to diagnosis and long-term care by working with third-party systems and your organization or vendor IT department to help with follow-up questions to keep populated data in check.
“Make sure you have a back-end system that can help monitor that structured data,” says Dr. Hettinger. Structured data are the patient’s demographic information, like name, address, age, height, weight, vital signs, and data elements like diagnosis, medications, and lab results. “Wherever you can leverage the underlying tools that are part of the electronic health record to make sure that we’re constantly checking the right results, that helps reduce the workload so that clinicians can focus on taking care of the patients and doing the right thing and not be as focused on entering data into the system.”
2. Supplement EHR notes with direct communication
The failure to diagnose cancer because one physician doesn’t know what another physician saw in an imaging report is one of the most common claims in the cases he tries, says Aaron Boeder, a plaintiff’s medical negligence lawyer in Chicago.
Physicians often assume that if they put a note in the electronic chart, others will look for it, but Mr. Boeder says it’s far more prudent to communicate directly.
“Let’s say a radiologist interprets a scan and sees what might be cancer,” he says. “If the ordering doctor is an orthopedist who’s ordered a CT scan for DVT, there’s going to be a report for that scan. It’s going to get auto-populated back into that physician’s note,” says Mr. Boeder.
The physician may or may not look at it, but it will be in their note, and they’re supposed to follow up on it because they ordered the scan. “But they may not follow up on it, and they may not get a call from the radiologist,” he says.
“Next thing you know, 2 or 3 years later, that patient is diagnosed with very advanced cancer.”
3. Tailor auto-fill information to your common practices
Suppose, as a physician, you find that you need to change a default setting time and time again. Dr. Hettinger says it’s worth your time to take an extra couple of minutes to work with your vendor or your health system to try and make changes to auto-population settings that align with your practices.
“Let’s say a default dose of 20 milligrams of a medication is what automatically pops up, but in reality, your practice is to use a smaller dose because it’s safer, even though they’re all within the acceptable realm of what you would order,” he says. “Rather than have the default to the higher dose, see if you can change the default to a lower dose. And that way, you don’t have to catch yourself every time.”
If your auto-fills are amounts that constantly need changing, an interruption could easily knock you off course before you make that correction.
“If there are ways to have the system defaults be safer or more in line with your clinical practice, and especially across a group, then you’re designing a safer system and not relying on vigilance or memory prone to interruptions,” says Dr. Hettinger.
4. Curb the copy and paste
It’s tempting to copy a note from a previous patient visit and make only minimal changes as needed, but you risk including outdated information if you do. Even if you’re repeating questions asked by the intake nurse, it is safer to not to rely on that information, says Beth Kanik, a defense medical malpractice attorney in Atlanta.
“If it later goes into litigation, the argument then becomes that it looks like you didn’t do your job,” says Ms. Kanik. “Instead, try to ask questions in a way that would elicit responses that may be a little different than what the nurse got, so that it’s clear you asked the questions and didn’t just simply rely upon someone else’s information.”
5. Separate typing from listening
While EHR may be an excellent tool for data collection and safety checking, it’s not a stand-in for doctor-patient interaction. As technology practices push medicine toward more and more efficiency, Mr. Boeder says it’s most often listening over all else that makes the difference in the quality of care. And good listening requires full attention.
“A real concern for physicians is the number of visits they’re expected to accomplish in a set amount of time,” says Mr. Boeder. “Often this translates into a doctor talking to a patient while typing notes or while reading a note from the last time the patient was in.”
Taking the time to pause after entering data and briefly reviewing your understanding of what your patient has told you can be invaluable and may save you – and your patient – problems later.
“In so many cases, it comes down to people not being heard,” says Mr. Boeder. “So listen to what your patients are saying.”
A version of this article first appeared on Medscape.com.
Domestic violence in health care is real and underreported
To protect survivors’ identities, some names have been changed or shortened.
Natasha Abadilla, MD, met the man who would become her abuser while working abroad for a public health nonprofit. When he began emotionally and physically abusing her, she did everything she could to hide it.
“My coworkers knew nothing of the abuse. I became an expert in applying makeup to hide the bruises,” recalls Dr. Abadilla, now a second-year resident and pediatric neurologist at Lucile Packard Children’s Hospital at Stanford.
Dr. Abadilla says she strongly identifies as a hard worker and – to this day – hopes her work did not falter despite her partner’s constant drain on her. But the impact of the abuse continued to affect her for years. Like many survivors of domestic violence, she struggled with PTSD and depression.
Health care workers are often the first point of contact for survivors of domestic violence. Experts and advocates continue to push for more training for clinicians to identify and respond to signs among their patients. Often missing from this conversation is the reality that those tasked with screening can also be victims of intimate partner violence themselves.
What’s more: The very strengths that medical professionals often pride themselves on – perfectionism, empathy, grit – can make it harder for them to identify abuse in their own relationships and push through humiliation and shame to seek help.
Dr. Abadilla is exceptional among survivors in the medical field. Rather than keep her experience quiet, she has shared it publicly.
Awareness, she believes, can save lives.
An understudied problem in an underserved group
The majority of research on health care workers in this area has focused on workplace violence, which 62% experience worldwide. But intimate partner violence remains understudied and underdiscussed. Some medical professionals are even saddled with a “double burden,” facing trauma at work and at home, note the authors of a 2022 meta-analysis published in the journal Trauma, Violence, & Abuse.
The problem has had dire consequences. In recent years, many health care workers have been killed by their abusers:
- In 2016, Casey M. Drawert, MD, a Texas-based critical care anesthesiologist, was fatally shot by her husband in a murder-suicide.
- In 2018, Tamara O’Neal, MD, an ER physician, and Dayna Less, a first-year pharmacy resident, were killed by Dr. O’Neal’s ex-fiancé at Mercy Hospital in Chicago.
- In 2019, Sarah Hawley, MD, a first-year University of Utah resident, was fatally shot by her boyfriend in a murder-suicide.
- In 2021, Moria Kinsey, a nurse practitioner in Tahlequah, Okla., was murdered by a physician.
- In July of 2023, Gwendolyn Lavonne Riddick, DO, an ob.gyn. in North Carolina, was fatally shot by the father of her 3-year-old son.
There are others.
In the wake of these tragedies, calls for health care workers to screen each other as well as patients have grown. But for an untold number of survivors, breaking the silence is still not possible due to concerns about their reputation, professional consequences, the threat of harassment from abusers who are often in the same field, a medical culture of selfless endurance, and a lack of appropriate resources.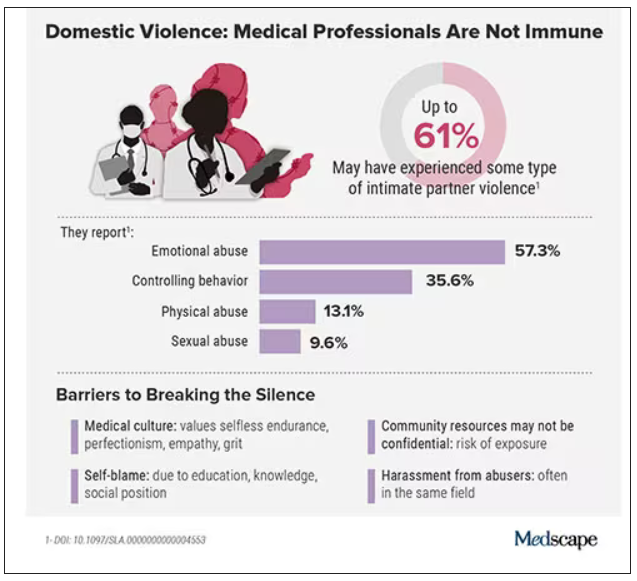
While the vast majority have stayed silent, those who have spoken out say there’s a need for targeted interventions to educate medical professionals as well as more supportive policies throughout the health care system.
Are health care workers more at risk?
Although more studies are needed, research indicates health care workers experience domestic violence at rates comparable to those of other populations, whereas some data suggest rates may be higher.
In the United States, more than one in three women and one in four men experience some form of intimate partner violence in their lifetime. Similarly, a 2020 study found that 24% of 400 physicians responding to a survey reported a history of domestic violence, with 15% reporting verbal abuse, 8% reporting physical violence, 4% reporting sexual abuse, and 4% reporting stalking.
Meanwhile, in an anonymous survey completed by 882 practicing surgeons and trainees in the United States from late 2018 to early 2019, more than 60% reported experiencing some type of intimate partner violence, most commonly emotional abuse.
Recent studies in the United Kingdom, Australia, and elsewhere show that significant numbers of medical professionals are fighting this battle. A 2019 study of more than 2,000 nurses, midwives, and health care assistants in the United Kingdom found that nurses were three times more likely to experience domestic violence than the average person.
What would help solve this problem: More study of health care worker-survivors as a unique group with unique risk factors. In general, domestic violence is most prevalent among women and people in marginalized groups. But young adults, such as medical students and trainees, can face an increased risk due to economic strain. Major life changes, such as relocating for residency, can also drive up stress and fray social connections, further isolating victims.
Why it’s so much harder for medical professionals to reveal abuse
For medical professionals accustomed to being strong and forging on, identifying as a victim of abuse can seem like a personal contradiction. It can feel easier to separate their personal and professional lives rather than face a complex reality.
In a personal essay on KevinMD.com, medical student Chloe N. L. Lee describes this emotional turmoil. “As an aspiring psychiatrist, I questioned my character judgment (how did I end up with a misogynistic abuser?) and wondered if I ought to have known better. I worried that my colleagues would deem me unfit to care for patients. And I thought that this was not supposed to happen to women like me,” Ms. Lee writes.
Kimberly, a licensed therapist, experienced a similar pattern of self-blame when her partner began exhibiting violent behavior. “For a long time, I felt guilty because I said to myself, You’re a therapist. You’re supposed to know this,” she recalls. At the same time, she felt driven to help him and sought couples therapy as his violence escalated.
Whitney, a pharmacist, recognized the “hallmarks” of abuse in her relationship, but she coped by compartmentalizing. Whitney says she was vulnerable to her abuser as a young college student who struggled financially. As he showered her with gifts, she found herself waving away red flags like aggressiveness or overprotectiveness.
After Whitney graduated, her partner’s emotional manipulation escalated into frequent physical assaults. When he gave her a black eye, she could not bring herself to go into work. She quit her job without notice. Despite a spotless record, none of her coworkers ever reached out to investigate her sudden departure.
It would take 8 years for Whitney to acknowledge the abuse and seize a moment to escape. She fled with just her purse and started over in a new city, rebuilding her life in the midst of harassment and threats from her ex. She says she’s grateful to be alive.
An imperfect system doesn’t help
Health care workers rarely ask for support or disclose abuse at work. Some have cited stigma, a lack of confidentiality (especially when the abuser is also in health care), fears about colleagues’ judgment, and a culture that doesn’t prioritize self-care.
Sometimes policies get in the way: In a 2021 qualitative study of interviews with 21 female physician-survivors in the United Kingdom, many said that despite the intense stress of abuse and recovery, they were unable to take any time off.
Of 180 UK-based midwife-survivors interviewed in a 2018 study, only 60 sought support at work and 30 received it. Many said their supervisors pressured them to report the abuse and get back to work, called social services behind their back, or reported them to their professional regulator. “I was treated like the perpetrator,” one said. Barbara Hernandez, PhD, a researcher who studies physician-survivors and director of physician vitality at Loma Linda University in southern California, says workplace violence and mistreatment from patients or colleagues – and a poor institutional response – can make those in health care feel like they have to “shut up and put up,” priming them to also tolerate abuse at home.
When survivors do reach out, there can be a disconnect between the resources they need and those they’re offered, Dr. Hernandez adds. In a recent survey of 400 physicians she conducted, respondents typically said they would advise a physician-survivor to “get to a shelter quickly.” But when roles were reversed, they admitted going to a shelter was the least feasible option. Support groups can also be problematic in smaller communities where physicians might be recognized or see their own patients.
Complicating matters further, the violence often comes from within the medical community. This can lead to particularly malicious abuse tactics like sending false accusations to a victim’s regulatory college or board; prolonged court and custody battles to drain them of all resources and their ability to hold a job; or even sabotage, harassment, or violence at work. The sheen of the abuser’s public persona, on the other hand, can guard them from any accountability.
For example, one physician-survivor said her ex-partner, a psychiatrist, coerced her into believing she was mentally ill, claimed she was “psychotic” in order to take back their children after she left, and had numerous colleagues serve as character witnesses in court for him, “saying he couldn’t have done any of these things, how great he is, and what a wonderful father he is.”
Slow progress is still progress
After Sherilyn M. Gordon-Burroughs, MD, a Texas-based transplant surgeon, mother, and educator, was killed by her husband in a murder-suicide in 2017, her friends Barbara Lee Bass, MD, president of the American College of Surgeons, and Patricia L. Turner, MD, were spurred into action. Together, they founded the ACS Intimate Partner Violence Task Force. Their mission is to educate surgeons to identify the signs of intimate partner violence (IPV) in themselves and their colleagues and connect them with resources.
“There is a concerted effort to close that gap,” says D’Andrea K. Joseph, MD, cochair of the task force and chief of trauma and acute care surgery at NYU Langone in New York. In the future, Dr. Joseph predicts, “making this a part of the curriculum, that it’s standardized for residents and trainees, that there is a safe place for victims ... and that we can band together and really recognize and assist our colleagues who are in trouble.”
Resources created by the ACS IPV task force, such as the toolkit and curriculum, provide a model for other health care leaders. But there have been few similar initiatives aimed at increasing IPV intervention within the medical system.
What you can do in your workplace
In her essay, Ms. Lee explains that a major turning point came when a physician friend explicitly asked if she was experiencing abuse. He then gently confirmed she was, and asked without judgment how he could support her, an approach that mirrors advice from the National Domestic Violence Hotline.
“Having a physician validate that this was, indeed, an abusive situation helped enormously ... I believe it may have saved my life,” she writes.
That validation can be crucial, and Dr. Abadilla urges other physicians to regularly check in with colleagues, especially those who seem particularly positive with a go-getter attitude and yet may not seem themselves. That was how she presented when she was struggling the most.
Supporting systemic changes within your organization and beyond is also important. The authors of the 2022 meta-analysis stress the need for domestic violence training, legislative changes, paid leave, and union support.
Finding strength in recovery
Over a decade after escaping her abuser, Whitney says she’s only just begun to share her experience, but what she’s learned has made her a better pharmacist. She says she’s more attuned to subtle signs something could be off with patients and coworkers. When someone makes comments about feeling anxious or that they can’t do anything right, it’s important to ask why, she says.
Recently, Kimberly has opened up to her mentor and other therapists, many of whom have shared that they’re also survivors.
“The last thing I said to [my abuser] is you think you’ve won and you’re hurting me, but what you’ve done to me – I’m going to utilize this and I’m going to help other people,” Kimberly says. “This pain that I have will go away, and I’m going to save the lives of others.”
A version of this article first appeared on Medscape.com.
To protect survivors’ identities, some names have been changed or shortened.
Natasha Abadilla, MD, met the man who would become her abuser while working abroad for a public health nonprofit. When he began emotionally and physically abusing her, she did everything she could to hide it.
“My coworkers knew nothing of the abuse. I became an expert in applying makeup to hide the bruises,” recalls Dr. Abadilla, now a second-year resident and pediatric neurologist at Lucile Packard Children’s Hospital at Stanford.
Dr. Abadilla says she strongly identifies as a hard worker and – to this day – hopes her work did not falter despite her partner’s constant drain on her. But the impact of the abuse continued to affect her for years. Like many survivors of domestic violence, she struggled with PTSD and depression.
Health care workers are often the first point of contact for survivors of domestic violence. Experts and advocates continue to push for more training for clinicians to identify and respond to signs among their patients. Often missing from this conversation is the reality that those tasked with screening can also be victims of intimate partner violence themselves.
What’s more: The very strengths that medical professionals often pride themselves on – perfectionism, empathy, grit – can make it harder for them to identify abuse in their own relationships and push through humiliation and shame to seek help.
Dr. Abadilla is exceptional among survivors in the medical field. Rather than keep her experience quiet, she has shared it publicly.
Awareness, she believes, can save lives.
An understudied problem in an underserved group
The majority of research on health care workers in this area has focused on workplace violence, which 62% experience worldwide. But intimate partner violence remains understudied and underdiscussed. Some medical professionals are even saddled with a “double burden,” facing trauma at work and at home, note the authors of a 2022 meta-analysis published in the journal Trauma, Violence, & Abuse.
The problem has had dire consequences. In recent years, many health care workers have been killed by their abusers:
- In 2016, Casey M. Drawert, MD, a Texas-based critical care anesthesiologist, was fatally shot by her husband in a murder-suicide.
- In 2018, Tamara O’Neal, MD, an ER physician, and Dayna Less, a first-year pharmacy resident, were killed by Dr. O’Neal’s ex-fiancé at Mercy Hospital in Chicago.
- In 2019, Sarah Hawley, MD, a first-year University of Utah resident, was fatally shot by her boyfriend in a murder-suicide.
- In 2021, Moria Kinsey, a nurse practitioner in Tahlequah, Okla., was murdered by a physician.
- In July of 2023, Gwendolyn Lavonne Riddick, DO, an ob.gyn. in North Carolina, was fatally shot by the father of her 3-year-old son.
There are others.
In the wake of these tragedies, calls for health care workers to screen each other as well as patients have grown. But for an untold number of survivors, breaking the silence is still not possible due to concerns about their reputation, professional consequences, the threat of harassment from abusers who are often in the same field, a medical culture of selfless endurance, and a lack of appropriate resources.
While the vast majority have stayed silent, those who have spoken out say there’s a need for targeted interventions to educate medical professionals as well as more supportive policies throughout the health care system.
Are health care workers more at risk?
Although more studies are needed, research indicates health care workers experience domestic violence at rates comparable to those of other populations, whereas some data suggest rates may be higher.
In the United States, more than one in three women and one in four men experience some form of intimate partner violence in their lifetime. Similarly, a 2020 study found that 24% of 400 physicians responding to a survey reported a history of domestic violence, with 15% reporting verbal abuse, 8% reporting physical violence, 4% reporting sexual abuse, and 4% reporting stalking.
Meanwhile, in an anonymous survey completed by 882 practicing surgeons and trainees in the United States from late 2018 to early 2019, more than 60% reported experiencing some type of intimate partner violence, most commonly emotional abuse.
Recent studies in the United Kingdom, Australia, and elsewhere show that significant numbers of medical professionals are fighting this battle. A 2019 study of more than 2,000 nurses, midwives, and health care assistants in the United Kingdom found that nurses were three times more likely to experience domestic violence than the average person.
What would help solve this problem: More study of health care worker-survivors as a unique group with unique risk factors. In general, domestic violence is most prevalent among women and people in marginalized groups. But young adults, such as medical students and trainees, can face an increased risk due to economic strain. Major life changes, such as relocating for residency, can also drive up stress and fray social connections, further isolating victims.
Why it’s so much harder for medical professionals to reveal abuse
For medical professionals accustomed to being strong and forging on, identifying as a victim of abuse can seem like a personal contradiction. It can feel easier to separate their personal and professional lives rather than face a complex reality.
In a personal essay on KevinMD.com, medical student Chloe N. L. Lee describes this emotional turmoil. “As an aspiring psychiatrist, I questioned my character judgment (how did I end up with a misogynistic abuser?) and wondered if I ought to have known better. I worried that my colleagues would deem me unfit to care for patients. And I thought that this was not supposed to happen to women like me,” Ms. Lee writes.
Kimberly, a licensed therapist, experienced a similar pattern of self-blame when her partner began exhibiting violent behavior. “For a long time, I felt guilty because I said to myself, You’re a therapist. You’re supposed to know this,” she recalls. At the same time, she felt driven to help him and sought couples therapy as his violence escalated.
Whitney, a pharmacist, recognized the “hallmarks” of abuse in her relationship, but she coped by compartmentalizing. Whitney says she was vulnerable to her abuser as a young college student who struggled financially. As he showered her with gifts, she found herself waving away red flags like aggressiveness or overprotectiveness.
After Whitney graduated, her partner’s emotional manipulation escalated into frequent physical assaults. When he gave her a black eye, she could not bring herself to go into work. She quit her job without notice. Despite a spotless record, none of her coworkers ever reached out to investigate her sudden departure.
It would take 8 years for Whitney to acknowledge the abuse and seize a moment to escape. She fled with just her purse and started over in a new city, rebuilding her life in the midst of harassment and threats from her ex. She says she’s grateful to be alive.
An imperfect system doesn’t help
Health care workers rarely ask for support or disclose abuse at work. Some have cited stigma, a lack of confidentiality (especially when the abuser is also in health care), fears about colleagues’ judgment, and a culture that doesn’t prioritize self-care.
Sometimes policies get in the way: In a 2021 qualitative study of interviews with 21 female physician-survivors in the United Kingdom, many said that despite the intense stress of abuse and recovery, they were unable to take any time off.
Of 180 UK-based midwife-survivors interviewed in a 2018 study, only 60 sought support at work and 30 received it. Many said their supervisors pressured them to report the abuse and get back to work, called social services behind their back, or reported them to their professional regulator. “I was treated like the perpetrator,” one said. Barbara Hernandez, PhD, a researcher who studies physician-survivors and director of physician vitality at Loma Linda University in southern California, says workplace violence and mistreatment from patients or colleagues – and a poor institutional response – can make those in health care feel like they have to “shut up and put up,” priming them to also tolerate abuse at home.
When survivors do reach out, there can be a disconnect between the resources they need and those they’re offered, Dr. Hernandez adds. In a recent survey of 400 physicians she conducted, respondents typically said they would advise a physician-survivor to “get to a shelter quickly.” But when roles were reversed, they admitted going to a shelter was the least feasible option. Support groups can also be problematic in smaller communities where physicians might be recognized or see their own patients.
Complicating matters further, the violence often comes from within the medical community. This can lead to particularly malicious abuse tactics like sending false accusations to a victim’s regulatory college or board; prolonged court and custody battles to drain them of all resources and their ability to hold a job; or even sabotage, harassment, or violence at work. The sheen of the abuser’s public persona, on the other hand, can guard them from any accountability.
For example, one physician-survivor said her ex-partner, a psychiatrist, coerced her into believing she was mentally ill, claimed she was “psychotic” in order to take back their children after she left, and had numerous colleagues serve as character witnesses in court for him, “saying he couldn’t have done any of these things, how great he is, and what a wonderful father he is.”
Slow progress is still progress
After Sherilyn M. Gordon-Burroughs, MD, a Texas-based transplant surgeon, mother, and educator, was killed by her husband in a murder-suicide in 2017, her friends Barbara Lee Bass, MD, president of the American College of Surgeons, and Patricia L. Turner, MD, were spurred into action. Together, they founded the ACS Intimate Partner Violence Task Force. Their mission is to educate surgeons to identify the signs of intimate partner violence (IPV) in themselves and their colleagues and connect them with resources.
“There is a concerted effort to close that gap,” says D’Andrea K. Joseph, MD, cochair of the task force and chief of trauma and acute care surgery at NYU Langone in New York. In the future, Dr. Joseph predicts, “making this a part of the curriculum, that it’s standardized for residents and trainees, that there is a safe place for victims ... and that we can band together and really recognize and assist our colleagues who are in trouble.”
Resources created by the ACS IPV task force, such as the toolkit and curriculum, provide a model for other health care leaders. But there have been few similar initiatives aimed at increasing IPV intervention within the medical system.
What you can do in your workplace
In her essay, Ms. Lee explains that a major turning point came when a physician friend explicitly asked if she was experiencing abuse. He then gently confirmed she was, and asked without judgment how he could support her, an approach that mirrors advice from the National Domestic Violence Hotline.
“Having a physician validate that this was, indeed, an abusive situation helped enormously ... I believe it may have saved my life,” she writes.
That validation can be crucial, and Dr. Abadilla urges other physicians to regularly check in with colleagues, especially those who seem particularly positive with a go-getter attitude and yet may not seem themselves. That was how she presented when she was struggling the most.
Supporting systemic changes within your organization and beyond is also important. The authors of the 2022 meta-analysis stress the need for domestic violence training, legislative changes, paid leave, and union support.
Finding strength in recovery
Over a decade after escaping her abuser, Whitney says she’s only just begun to share her experience, but what she’s learned has made her a better pharmacist. She says she’s more attuned to subtle signs something could be off with patients and coworkers. When someone makes comments about feeling anxious or that they can’t do anything right, it’s important to ask why, she says.
Recently, Kimberly has opened up to her mentor and other therapists, many of whom have shared that they’re also survivors.
“The last thing I said to [my abuser] is you think you’ve won and you’re hurting me, but what you’ve done to me – I’m going to utilize this and I’m going to help other people,” Kimberly says. “This pain that I have will go away, and I’m going to save the lives of others.”
A version of this article first appeared on Medscape.com.
To protect survivors’ identities, some names have been changed or shortened.
Natasha Abadilla, MD, met the man who would become her abuser while working abroad for a public health nonprofit. When he began emotionally and physically abusing her, she did everything she could to hide it.
“My coworkers knew nothing of the abuse. I became an expert in applying makeup to hide the bruises,” recalls Dr. Abadilla, now a second-year resident and pediatric neurologist at Lucile Packard Children’s Hospital at Stanford.
Dr. Abadilla says she strongly identifies as a hard worker and – to this day – hopes her work did not falter despite her partner’s constant drain on her. But the impact of the abuse continued to affect her for years. Like many survivors of domestic violence, she struggled with PTSD and depression.
Health care workers are often the first point of contact for survivors of domestic violence. Experts and advocates continue to push for more training for clinicians to identify and respond to signs among their patients. Often missing from this conversation is the reality that those tasked with screening can also be victims of intimate partner violence themselves.
What’s more: The very strengths that medical professionals often pride themselves on – perfectionism, empathy, grit – can make it harder for them to identify abuse in their own relationships and push through humiliation and shame to seek help.
Dr. Abadilla is exceptional among survivors in the medical field. Rather than keep her experience quiet, she has shared it publicly.
Awareness, she believes, can save lives.
An understudied problem in an underserved group
The majority of research on health care workers in this area has focused on workplace violence, which 62% experience worldwide. But intimate partner violence remains understudied and underdiscussed. Some medical professionals are even saddled with a “double burden,” facing trauma at work and at home, note the authors of a 2022 meta-analysis published in the journal Trauma, Violence, & Abuse.
The problem has had dire consequences. In recent years, many health care workers have been killed by their abusers:
- In 2016, Casey M. Drawert, MD, a Texas-based critical care anesthesiologist, was fatally shot by her husband in a murder-suicide.
- In 2018, Tamara O’Neal, MD, an ER physician, and Dayna Less, a first-year pharmacy resident, were killed by Dr. O’Neal’s ex-fiancé at Mercy Hospital in Chicago.
- In 2019, Sarah Hawley, MD, a first-year University of Utah resident, was fatally shot by her boyfriend in a murder-suicide.
- In 2021, Moria Kinsey, a nurse practitioner in Tahlequah, Okla., was murdered by a physician.
- In July of 2023, Gwendolyn Lavonne Riddick, DO, an ob.gyn. in North Carolina, was fatally shot by the father of her 3-year-old son.
There are others.
In the wake of these tragedies, calls for health care workers to screen each other as well as patients have grown. But for an untold number of survivors, breaking the silence is still not possible due to concerns about their reputation, professional consequences, the threat of harassment from abusers who are often in the same field, a medical culture of selfless endurance, and a lack of appropriate resources.
While the vast majority have stayed silent, those who have spoken out say there’s a need for targeted interventions to educate medical professionals as well as more supportive policies throughout the health care system.
Are health care workers more at risk?
Although more studies are needed, research indicates health care workers experience domestic violence at rates comparable to those of other populations, whereas some data suggest rates may be higher.
In the United States, more than one in three women and one in four men experience some form of intimate partner violence in their lifetime. Similarly, a 2020 study found that 24% of 400 physicians responding to a survey reported a history of domestic violence, with 15% reporting verbal abuse, 8% reporting physical violence, 4% reporting sexual abuse, and 4% reporting stalking.
Meanwhile, in an anonymous survey completed by 882 practicing surgeons and trainees in the United States from late 2018 to early 2019, more than 60% reported experiencing some type of intimate partner violence, most commonly emotional abuse.
Recent studies in the United Kingdom, Australia, and elsewhere show that significant numbers of medical professionals are fighting this battle. A 2019 study of more than 2,000 nurses, midwives, and health care assistants in the United Kingdom found that nurses were three times more likely to experience domestic violence than the average person.
What would help solve this problem: More study of health care worker-survivors as a unique group with unique risk factors. In general, domestic violence is most prevalent among women and people in marginalized groups. But young adults, such as medical students and trainees, can face an increased risk due to economic strain. Major life changes, such as relocating for residency, can also drive up stress and fray social connections, further isolating victims.
Why it’s so much harder for medical professionals to reveal abuse
For medical professionals accustomed to being strong and forging on, identifying as a victim of abuse can seem like a personal contradiction. It can feel easier to separate their personal and professional lives rather than face a complex reality.
In a personal essay on KevinMD.com, medical student Chloe N. L. Lee describes this emotional turmoil. “As an aspiring psychiatrist, I questioned my character judgment (how did I end up with a misogynistic abuser?) and wondered if I ought to have known better. I worried that my colleagues would deem me unfit to care for patients. And I thought that this was not supposed to happen to women like me,” Ms. Lee writes.
Kimberly, a licensed therapist, experienced a similar pattern of self-blame when her partner began exhibiting violent behavior. “For a long time, I felt guilty because I said to myself, You’re a therapist. You’re supposed to know this,” she recalls. At the same time, she felt driven to help him and sought couples therapy as his violence escalated.
Whitney, a pharmacist, recognized the “hallmarks” of abuse in her relationship, but she coped by compartmentalizing. Whitney says she was vulnerable to her abuser as a young college student who struggled financially. As he showered her with gifts, she found herself waving away red flags like aggressiveness or overprotectiveness.
After Whitney graduated, her partner’s emotional manipulation escalated into frequent physical assaults. When he gave her a black eye, she could not bring herself to go into work. She quit her job without notice. Despite a spotless record, none of her coworkers ever reached out to investigate her sudden departure.
It would take 8 years for Whitney to acknowledge the abuse and seize a moment to escape. She fled with just her purse and started over in a new city, rebuilding her life in the midst of harassment and threats from her ex. She says she’s grateful to be alive.
An imperfect system doesn’t help
Health care workers rarely ask for support or disclose abuse at work. Some have cited stigma, a lack of confidentiality (especially when the abuser is also in health care), fears about colleagues’ judgment, and a culture that doesn’t prioritize self-care.
Sometimes policies get in the way: In a 2021 qualitative study of interviews with 21 female physician-survivors in the United Kingdom, many said that despite the intense stress of abuse and recovery, they were unable to take any time off.
Of 180 UK-based midwife-survivors interviewed in a 2018 study, only 60 sought support at work and 30 received it. Many said their supervisors pressured them to report the abuse and get back to work, called social services behind their back, or reported them to their professional regulator. “I was treated like the perpetrator,” one said. Barbara Hernandez, PhD, a researcher who studies physician-survivors and director of physician vitality at Loma Linda University in southern California, says workplace violence and mistreatment from patients or colleagues – and a poor institutional response – can make those in health care feel like they have to “shut up and put up,” priming them to also tolerate abuse at home.
When survivors do reach out, there can be a disconnect between the resources they need and those they’re offered, Dr. Hernandez adds. In a recent survey of 400 physicians she conducted, respondents typically said they would advise a physician-survivor to “get to a shelter quickly.” But when roles were reversed, they admitted going to a shelter was the least feasible option. Support groups can also be problematic in smaller communities where physicians might be recognized or see their own patients.
Complicating matters further, the violence often comes from within the medical community. This can lead to particularly malicious abuse tactics like sending false accusations to a victim’s regulatory college or board; prolonged court and custody battles to drain them of all resources and their ability to hold a job; or even sabotage, harassment, or violence at work. The sheen of the abuser’s public persona, on the other hand, can guard them from any accountability.
For example, one physician-survivor said her ex-partner, a psychiatrist, coerced her into believing she was mentally ill, claimed she was “psychotic” in order to take back their children after she left, and had numerous colleagues serve as character witnesses in court for him, “saying he couldn’t have done any of these things, how great he is, and what a wonderful father he is.”
Slow progress is still progress
After Sherilyn M. Gordon-Burroughs, MD, a Texas-based transplant surgeon, mother, and educator, was killed by her husband in a murder-suicide in 2017, her friends Barbara Lee Bass, MD, president of the American College of Surgeons, and Patricia L. Turner, MD, were spurred into action. Together, they founded the ACS Intimate Partner Violence Task Force. Their mission is to educate surgeons to identify the signs of intimate partner violence (IPV) in themselves and their colleagues and connect them with resources.
“There is a concerted effort to close that gap,” says D’Andrea K. Joseph, MD, cochair of the task force and chief of trauma and acute care surgery at NYU Langone in New York. In the future, Dr. Joseph predicts, “making this a part of the curriculum, that it’s standardized for residents and trainees, that there is a safe place for victims ... and that we can band together and really recognize and assist our colleagues who are in trouble.”
Resources created by the ACS IPV task force, such as the toolkit and curriculum, provide a model for other health care leaders. But there have been few similar initiatives aimed at increasing IPV intervention within the medical system.
What you can do in your workplace
In her essay, Ms. Lee explains that a major turning point came when a physician friend explicitly asked if she was experiencing abuse. He then gently confirmed she was, and asked without judgment how he could support her, an approach that mirrors advice from the National Domestic Violence Hotline.
“Having a physician validate that this was, indeed, an abusive situation helped enormously ... I believe it may have saved my life,” she writes.
That validation can be crucial, and Dr. Abadilla urges other physicians to regularly check in with colleagues, especially those who seem particularly positive with a go-getter attitude and yet may not seem themselves. That was how she presented when she was struggling the most.
Supporting systemic changes within your organization and beyond is also important. The authors of the 2022 meta-analysis stress the need for domestic violence training, legislative changes, paid leave, and union support.
Finding strength in recovery
Over a decade after escaping her abuser, Whitney says she’s only just begun to share her experience, but what she’s learned has made her a better pharmacist. She says she’s more attuned to subtle signs something could be off with patients and coworkers. When someone makes comments about feeling anxious or that they can’t do anything right, it’s important to ask why, she says.
Recently, Kimberly has opened up to her mentor and other therapists, many of whom have shared that they’re also survivors.
“The last thing I said to [my abuser] is you think you’ve won and you’re hurting me, but what you’ve done to me – I’m going to utilize this and I’m going to help other people,” Kimberly says. “This pain that I have will go away, and I’m going to save the lives of others.”
A version of this article first appeared on Medscape.com.
One in five doctors with long COVID can no longer work: Survey
Crippling symptoms, lost careers, and eroded incomes: This is the harsh reality for doctors suffering with long COVID, according to the first major survey of physicians with the condition.
The survey, conducted by the British Medical Association and the Long COVID Doctors for Action support group, sheds light on the lingering effects of long COVID on more than 600 chronically ill and disabled doctors with the condition. It also spotlights what they describe as a lack of medical and financial support from their government and employers at the National Health Service.
“We feel betrayed and abandoned,” said Kelly Fearnley, MBChB, chair and cofounder of Long COVID Doctors for Action. “At a time of national crisis, when health care workers were asked to step up, we did. When the nation needed us, we stepped up. We put our lives on the line. We put our families’ lives on the line. And now that we are injured after knowingly being unprotected and deliberately and repeatedly exposed to a level 3 biohazard, we now find ourselves in this position.”
Dr. Fearnley fell ill while working in a hospital’s COVID ward in November 2020. She is one of an estimated 2 million people in the United Kingdom – including thousands of NHS employees – with long COVID. She hasn’t been able to return to work in nearly 3 years.
Long COVID affects more than 65 million people worldwide. It is estimated that 1 in 10 people infected with the virus develop long-term symptoms. In the United Kingdom, health care and social care workers are seven times more likely to have had severe COVID-19 than other types of employees.
Doctors responding to the BMA survey reported a wide range of long COVID symptoms, including fatigue, headaches, muscular pain, nerve damage, joint pain, and respiratory problems.
Among the survey’s key findings, 60% of doctors said long COVID has affected their ability to carry out day-to-day tasks on a regular basis. Almost one in five (18%) said they were no longer able to work, while fewer than one in three (31%) were working full time. This compares with more than half (57%) of respondents working full time before the onset of their COVID illness – a decline of 46%.
Nearly half (48%) of respondents said they have experienced some form of loss of earnings as a result of long COVID, and almost half of the doctors were never referred to an NHS long COVID clinic. The survey included the following first-person accounts from doctors living with the condition.
- One doctor said: “I nearly lost my life, my home, my partner and my career. I have received little support to help keep these. The impact on my mental health nearly cost [me] my life again.”
- A senior consulting physician commented: “Life is absolutely miserable. Every day is a struggle. I wake up exhausted, the insomnia and night terrors are horrendous as I live through my worst fears every night. Any activity such as eating meals, washing, etc., will mean I have to go to bed for a few hours. I am unable to look after myself or my child, exercise or maintain social relationships. I have no financial security. Long COVID has totally destroyed my life.”
- A salaried general practitioner said: “I can no longer work, finances are ruined. I didn’t have employment protection so am now unemployed and penniless.”
Calls for action from the BMA include the following:
- Financial support for doctors and health care staff with long COVID.
- The recognition of long COVID as an occupational disease among health care workers, along with a definition of the condition that covers all of the debilitating disease’s symptoms.
- Improved access to physical and mental health services to help comprehensive assessment, investigations, and treatment.
- Greater workplace protection for health care staff who risk their lives for others.
- Better support for long COVID sufferers to return to work safely if they can, including a flexible approach to the use of workplace adjustments.
“One would think, given the circumstances under which we fell ill and current workforce shortages, NHS employers would be eager to do everything to facilitate the return to work of people with long COVID,” said Dr. Fearnley. “However, NHS employers are legally required to implement only ‘reasonable adjustments,’ and so things such as extended phased return or adjustments to shift patterns are not always being facilitated. Instead, an increasing number of employers are choosing to terminate contracts.”
Raymond Agius, the BMA’s occupational medicine committee cochair, also put the blame on inadequate safety measures for doctors. Those inadequate measures persist to this day, inasmuch as U.K. hospitals have dropped masking requirements.
“During the COVID-19 pandemic, doctors were left exposed and unprotected at work,” he said in a BMA press release. “They often did not have access to the right PPE. ... Too many risk assessments of workplaces and especially of vulnerable doctors were not undertaken.”
A small minority of doctors who were surveyed said they had access to respiratory protective equipment about the time they contracted COVID-19. Only 11% had access to an FFP2 respirator (the equivalent of an N95 mask); 16% had an FFP3 respirator (the equivalent of an N99 mask).
To date, the British government hasn’t issued much of a response to the survey, saying only that it has invested more than ₤50 million to better understand long COVID.
A version of this article first appeared on Medscape.com.
Crippling symptoms, lost careers, and eroded incomes: This is the harsh reality for doctors suffering with long COVID, according to the first major survey of physicians with the condition.
The survey, conducted by the British Medical Association and the Long COVID Doctors for Action support group, sheds light on the lingering effects of long COVID on more than 600 chronically ill and disabled doctors with the condition. It also spotlights what they describe as a lack of medical and financial support from their government and employers at the National Health Service.
“We feel betrayed and abandoned,” said Kelly Fearnley, MBChB, chair and cofounder of Long COVID Doctors for Action. “At a time of national crisis, when health care workers were asked to step up, we did. When the nation needed us, we stepped up. We put our lives on the line. We put our families’ lives on the line. And now that we are injured after knowingly being unprotected and deliberately and repeatedly exposed to a level 3 biohazard, we now find ourselves in this position.”
Dr. Fearnley fell ill while working in a hospital’s COVID ward in November 2020. She is one of an estimated 2 million people in the United Kingdom – including thousands of NHS employees – with long COVID. She hasn’t been able to return to work in nearly 3 years.
Long COVID affects more than 65 million people worldwide. It is estimated that 1 in 10 people infected with the virus develop long-term symptoms. In the United Kingdom, health care and social care workers are seven times more likely to have had severe COVID-19 than other types of employees.
Doctors responding to the BMA survey reported a wide range of long COVID symptoms, including fatigue, headaches, muscular pain, nerve damage, joint pain, and respiratory problems.
Among the survey’s key findings, 60% of doctors said long COVID has affected their ability to carry out day-to-day tasks on a regular basis. Almost one in five (18%) said they were no longer able to work, while fewer than one in three (31%) were working full time. This compares with more than half (57%) of respondents working full time before the onset of their COVID illness – a decline of 46%.
Nearly half (48%) of respondents said they have experienced some form of loss of earnings as a result of long COVID, and almost half of the doctors were never referred to an NHS long COVID clinic. The survey included the following first-person accounts from doctors living with the condition.
- One doctor said: “I nearly lost my life, my home, my partner and my career. I have received little support to help keep these. The impact on my mental health nearly cost [me] my life again.”
- A senior consulting physician commented: “Life is absolutely miserable. Every day is a struggle. I wake up exhausted, the insomnia and night terrors are horrendous as I live through my worst fears every night. Any activity such as eating meals, washing, etc., will mean I have to go to bed for a few hours. I am unable to look after myself or my child, exercise or maintain social relationships. I have no financial security. Long COVID has totally destroyed my life.”
- A salaried general practitioner said: “I can no longer work, finances are ruined. I didn’t have employment protection so am now unemployed and penniless.”
Calls for action from the BMA include the following:
- Financial support for doctors and health care staff with long COVID.
- The recognition of long COVID as an occupational disease among health care workers, along with a definition of the condition that covers all of the debilitating disease’s symptoms.
- Improved access to physical and mental health services to help comprehensive assessment, investigations, and treatment.
- Greater workplace protection for health care staff who risk their lives for others.
- Better support for long COVID sufferers to return to work safely if they can, including a flexible approach to the use of workplace adjustments.
“One would think, given the circumstances under which we fell ill and current workforce shortages, NHS employers would be eager to do everything to facilitate the return to work of people with long COVID,” said Dr. Fearnley. “However, NHS employers are legally required to implement only ‘reasonable adjustments,’ and so things such as extended phased return or adjustments to shift patterns are not always being facilitated. Instead, an increasing number of employers are choosing to terminate contracts.”
Raymond Agius, the BMA’s occupational medicine committee cochair, also put the blame on inadequate safety measures for doctors. Those inadequate measures persist to this day, inasmuch as U.K. hospitals have dropped masking requirements.
“During the COVID-19 pandemic, doctors were left exposed and unprotected at work,” he said in a BMA press release. “They often did not have access to the right PPE. ... Too many risk assessments of workplaces and especially of vulnerable doctors were not undertaken.”
A small minority of doctors who were surveyed said they had access to respiratory protective equipment about the time they contracted COVID-19. Only 11% had access to an FFP2 respirator (the equivalent of an N95 mask); 16% had an FFP3 respirator (the equivalent of an N99 mask).
To date, the British government hasn’t issued much of a response to the survey, saying only that it has invested more than ₤50 million to better understand long COVID.
A version of this article first appeared on Medscape.com.
Crippling symptoms, lost careers, and eroded incomes: This is the harsh reality for doctors suffering with long COVID, according to the first major survey of physicians with the condition.
The survey, conducted by the British Medical Association and the Long COVID Doctors for Action support group, sheds light on the lingering effects of long COVID on more than 600 chronically ill and disabled doctors with the condition. It also spotlights what they describe as a lack of medical and financial support from their government and employers at the National Health Service.
“We feel betrayed and abandoned,” said Kelly Fearnley, MBChB, chair and cofounder of Long COVID Doctors for Action. “At a time of national crisis, when health care workers were asked to step up, we did. When the nation needed us, we stepped up. We put our lives on the line. We put our families’ lives on the line. And now that we are injured after knowingly being unprotected and deliberately and repeatedly exposed to a level 3 biohazard, we now find ourselves in this position.”
Dr. Fearnley fell ill while working in a hospital’s COVID ward in November 2020. She is one of an estimated 2 million people in the United Kingdom – including thousands of NHS employees – with long COVID. She hasn’t been able to return to work in nearly 3 years.
Long COVID affects more than 65 million people worldwide. It is estimated that 1 in 10 people infected with the virus develop long-term symptoms. In the United Kingdom, health care and social care workers are seven times more likely to have had severe COVID-19 than other types of employees.
Doctors responding to the BMA survey reported a wide range of long COVID symptoms, including fatigue, headaches, muscular pain, nerve damage, joint pain, and respiratory problems.
Among the survey’s key findings, 60% of doctors said long COVID has affected their ability to carry out day-to-day tasks on a regular basis. Almost one in five (18%) said they were no longer able to work, while fewer than one in three (31%) were working full time. This compares with more than half (57%) of respondents working full time before the onset of their COVID illness – a decline of 46%.
Nearly half (48%) of respondents said they have experienced some form of loss of earnings as a result of long COVID, and almost half of the doctors were never referred to an NHS long COVID clinic. The survey included the following first-person accounts from doctors living with the condition.
- One doctor said: “I nearly lost my life, my home, my partner and my career. I have received little support to help keep these. The impact on my mental health nearly cost [me] my life again.”
- A senior consulting physician commented: “Life is absolutely miserable. Every day is a struggle. I wake up exhausted, the insomnia and night terrors are horrendous as I live through my worst fears every night. Any activity such as eating meals, washing, etc., will mean I have to go to bed for a few hours. I am unable to look after myself or my child, exercise or maintain social relationships. I have no financial security. Long COVID has totally destroyed my life.”
- A salaried general practitioner said: “I can no longer work, finances are ruined. I didn’t have employment protection so am now unemployed and penniless.”
Calls for action from the BMA include the following:
- Financial support for doctors and health care staff with long COVID.
- The recognition of long COVID as an occupational disease among health care workers, along with a definition of the condition that covers all of the debilitating disease’s symptoms.
- Improved access to physical and mental health services to help comprehensive assessment, investigations, and treatment.
- Greater workplace protection for health care staff who risk their lives for others.
- Better support for long COVID sufferers to return to work safely if they can, including a flexible approach to the use of workplace adjustments.
“One would think, given the circumstances under which we fell ill and current workforce shortages, NHS employers would be eager to do everything to facilitate the return to work of people with long COVID,” said Dr. Fearnley. “However, NHS employers are legally required to implement only ‘reasonable adjustments,’ and so things such as extended phased return or adjustments to shift patterns are not always being facilitated. Instead, an increasing number of employers are choosing to terminate contracts.”
Raymond Agius, the BMA’s occupational medicine committee cochair, also put the blame on inadequate safety measures for doctors. Those inadequate measures persist to this day, inasmuch as U.K. hospitals have dropped masking requirements.
“During the COVID-19 pandemic, doctors were left exposed and unprotected at work,” he said in a BMA press release. “They often did not have access to the right PPE. ... Too many risk assessments of workplaces and especially of vulnerable doctors were not undertaken.”
A small minority of doctors who were surveyed said they had access to respiratory protective equipment about the time they contracted COVID-19. Only 11% had access to an FFP2 respirator (the equivalent of an N95 mask); 16% had an FFP3 respirator (the equivalent of an N99 mask).
To date, the British government hasn’t issued much of a response to the survey, saying only that it has invested more than ₤50 million to better understand long COVID.
A version of this article first appeared on Medscape.com.
Resident creates AI alternative to U.S. News med school ranking
For decades, pre-med students depended on the annual medical school rankings by U.S. News and World Report to decide where to apply for physician education. But after several prominent med schools pulled out of the rankings, one resident began experimenting with artificial intelligence (AI) to create an alternative.
Brandon Turner MD, MSc, a radiation oncology resident at Massachusetts General Hospital in Boston, developed a free do-it-yourself tool using AI that allows prospective students to rank medical schools based on considerations that are most important to them. His research was published online in JAMA Network Open.
“One of the flaws with conventional ranking systems is that the metrics used in these tools are weighted based on the preferences and views of the people who developed these rankings, but those may not work for everyone,” Dr. Turner told this news organization.
He explained that there are different types of metrics used in the U.S. News ranking: one for research and the other for primary care. “The research rankings carry the most prestige and are the ones that most people know about,” he explained. These metrics take into account factors such as how many grant dollars the medical school receives and the average size of those grants per faculty member, Dr. Turner said.
Admission metrics are also included – for example, the median grade point average or MCAT scores of students who have been accepted. “These don’t tell you anything about the research output of the school, only about how selective the school is,” he said.
Primary care metrics might focus on how many graduates of a given school go into primary care, or how other schools rate the quality of primary care training at a given school – a process called peer assessment, Dr. Turner said.
But even though these might be helpful, students may be more interested in the cost of attendance, average debt, representation of minorities, and how many graduates pass their boards, he said. “U.S. News metrics don’t capture these things, but I included them in my algorithm.”
A U.S. News spokesperson said that the publication continues to help students and their families make decisions about their future education. The spokesperson cited U.S. News’ explanation of how it calculates its rankings. “A school’s overall Best Medical Schools rank should be one consideration and not the lone determinant in where a student applies and accepts,” the article states.
Dr. Turner agreed ranking systems are a good starting point when researching med schools, “but the values reflected in the ranking may not reflect an individual’s goals.”
Tyra-Lee Brett, a premed student at the University of South Florida, Tampa, believes an additional tool for students to evaluate medical schools is needed – and she could potentially see herself using Dr. Turner’s creation.
Still, Ms. Brett, a premed trustee of the American Medical Student Association, doesn’t regard any ranking tool as the “be all and end all.” Rather, she feels that the most effective tool would be based on students’ lived experiences. The AMSA is developing a scorecard in which students grade schools based on their opinions about such issues as housing, family planning, and environmental health, she said.
No prior judgments
To develop his algorithm, Dr. Turner used a branch of AI called “unsupervised learning.” It doesn’t make a prior judgment about what the data should look like, Dr. Turner explained.
“You’re just analyzing natural trends within the data.”
The algorithm tries to find and discover clusters or patterns within the data. “It’s like saying to the algorithm: ‘I want you to tell me what schools you think should be grouped together based on the data I feed you,’ which is the data that the user selects based on his or her personal preferences.”
U.S. News has been transparent about the metrics it uses, Dr. Turner notes. “When I started looking into how rankings are developed, I saw that there was transparency, and the reasoning for choosing the metrics used to develop the ranking was pretty sound,” he said.
“But I didn’t see any justification as to why they chose the particular metrics and weighted them in the way that they did.”
Dr. Turner extracted data from the 2023 U.S. News report, which ranked 109 allopathic medical schools, and applied several scenarios to the results to create his alternative ranking system.
In one scenario, he used the same research metrics used by U.S. News, such as a peer research assessment, median federal research activity per full-time faculty member, median GPA, median MCAT, acceptance rate, and faculty-student ratio.
In another scenario, he included four additional metrics: debt, in-state cost of attendance, USMLE Step 1 passing rate, and percentage of underrepresented students with minority race or ethnicity at the school.
For example, a user can rank the importance of the diversity of the class, amount of debt students expect to incur, and amount of research funding the medical school receives. After selecting those factors, the tool generates tiered results displayed in a circle, a shape chosen to avoid the appearance of the hierarchy associated with traditional rankings, Dr. Turner said.
“A prospective student might not care about acceptance rates and MCAT scores, and instead cares about diversity and debt,” Dr. Turner said. He looks forward to extending this approach to the ranking of colleges as well.
‘Imperfect measures’
“The model and interesting online tool that Dr. Turner created allows a premed [student] to generate custom rankings that are in line with their own priorities,” said Christopher Worsham, MD, MPH, a critical care physician in Mass General’s division of pulmonary and critical care medicine.
But Dr. Worsham, also a teaching associate at Harvard Medical School’s department of health care policy, expressed concern that factors figuring into the rankings by U.S. News and Dr. Turner’s alternative “are imperfect measures of medical school quality.”
For example, a student interested in research might favor federal research funding in their customized rankings with Dr. Turner’s model. “But higher research funding doesn’t necessarily translate into a better education for students, particularly when differentiating between two major research systems,” Dr. Worsham noted.
Dr. Worsham added that neither ranking system accurately predicts the quality of doctors graduating from the schools. Instead, he’d like to see ranking systems based on which schools’ graduates deliver the best patient outcomes, whether that’s through direct patient care, impactful research, or leadership within the health care system.
Michael Sauder, PhD, professor of sociology at the University of Iowa, Iowa City, said the model could offer a valuable alternative to the U.S. News ranking system. It might help users develop their own criteria for determining the ranking of medical schools, which is a big improvement over a “one-size-fits-all” approach, Dr. Sauder said.
And Hanna Stotland, an admission consultant based in Chicago, noted that most students rely on rankings because they “don’t have the luxury of advisers who know the ins and outs of different medical schools.” Given the role that rankings play, Ms. Stotland expects that every new ranking tool will have some influence on students.
This tool in particular “has the potential to be useful for students who have identified values they want their medical school to share.” For example, students who care about racial diversity “could use it to easily identify schools that are successful on that metric,” Ms. Stotland said.
Sujay Ratna, a 2nd-year med student at Icahn School of Medicine at Mount Sinai in New York, said he considered the U.S. News ranking his “go-to tool” when he was applying to med school.
But after reading Dr. Turner’s article, the AMSA membership vice president tried the algorithm. “I definitely would have used it had it existed when I was thinking of what schools to apply to and what [schools] to attend.”
The study had no specific funding. Dr. Turner, Dr. Worsham, Dr. Sauder, Ms. Stotland, Ms. Brett, and Mr. Ratna report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For decades, pre-med students depended on the annual medical school rankings by U.S. News and World Report to decide where to apply for physician education. But after several prominent med schools pulled out of the rankings, one resident began experimenting with artificial intelligence (AI) to create an alternative.
Brandon Turner MD, MSc, a radiation oncology resident at Massachusetts General Hospital in Boston, developed a free do-it-yourself tool using AI that allows prospective students to rank medical schools based on considerations that are most important to them. His research was published online in JAMA Network Open.
“One of the flaws with conventional ranking systems is that the metrics used in these tools are weighted based on the preferences and views of the people who developed these rankings, but those may not work for everyone,” Dr. Turner told this news organization.
He explained that there are different types of metrics used in the U.S. News ranking: one for research and the other for primary care. “The research rankings carry the most prestige and are the ones that most people know about,” he explained. These metrics take into account factors such as how many grant dollars the medical school receives and the average size of those grants per faculty member, Dr. Turner said.
Admission metrics are also included – for example, the median grade point average or MCAT scores of students who have been accepted. “These don’t tell you anything about the research output of the school, only about how selective the school is,” he said.
Primary care metrics might focus on how many graduates of a given school go into primary care, or how other schools rate the quality of primary care training at a given school – a process called peer assessment, Dr. Turner said.
But even though these might be helpful, students may be more interested in the cost of attendance, average debt, representation of minorities, and how many graduates pass their boards, he said. “U.S. News metrics don’t capture these things, but I included them in my algorithm.”
A U.S. News spokesperson said that the publication continues to help students and their families make decisions about their future education. The spokesperson cited U.S. News’ explanation of how it calculates its rankings. “A school’s overall Best Medical Schools rank should be one consideration and not the lone determinant in where a student applies and accepts,” the article states.
Dr. Turner agreed ranking systems are a good starting point when researching med schools, “but the values reflected in the ranking may not reflect an individual’s goals.”
Tyra-Lee Brett, a premed student at the University of South Florida, Tampa, believes an additional tool for students to evaluate medical schools is needed – and she could potentially see herself using Dr. Turner’s creation.
Still, Ms. Brett, a premed trustee of the American Medical Student Association, doesn’t regard any ranking tool as the “be all and end all.” Rather, she feels that the most effective tool would be based on students’ lived experiences. The AMSA is developing a scorecard in which students grade schools based on their opinions about such issues as housing, family planning, and environmental health, she said.
No prior judgments
To develop his algorithm, Dr. Turner used a branch of AI called “unsupervised learning.” It doesn’t make a prior judgment about what the data should look like, Dr. Turner explained.
“You’re just analyzing natural trends within the data.”
The algorithm tries to find and discover clusters or patterns within the data. “It’s like saying to the algorithm: ‘I want you to tell me what schools you think should be grouped together based on the data I feed you,’ which is the data that the user selects based on his or her personal preferences.”
U.S. News has been transparent about the metrics it uses, Dr. Turner notes. “When I started looking into how rankings are developed, I saw that there was transparency, and the reasoning for choosing the metrics used to develop the ranking was pretty sound,” he said.
“But I didn’t see any justification as to why they chose the particular metrics and weighted them in the way that they did.”
Dr. Turner extracted data from the 2023 U.S. News report, which ranked 109 allopathic medical schools, and applied several scenarios to the results to create his alternative ranking system.
In one scenario, he used the same research metrics used by U.S. News, such as a peer research assessment, median federal research activity per full-time faculty member, median GPA, median MCAT, acceptance rate, and faculty-student ratio.
In another scenario, he included four additional metrics: debt, in-state cost of attendance, USMLE Step 1 passing rate, and percentage of underrepresented students with minority race or ethnicity at the school.
For example, a user can rank the importance of the diversity of the class, amount of debt students expect to incur, and amount of research funding the medical school receives. After selecting those factors, the tool generates tiered results displayed in a circle, a shape chosen to avoid the appearance of the hierarchy associated with traditional rankings, Dr. Turner said.
“A prospective student might not care about acceptance rates and MCAT scores, and instead cares about diversity and debt,” Dr. Turner said. He looks forward to extending this approach to the ranking of colleges as well.
‘Imperfect measures’
“The model and interesting online tool that Dr. Turner created allows a premed [student] to generate custom rankings that are in line with their own priorities,” said Christopher Worsham, MD, MPH, a critical care physician in Mass General’s division of pulmonary and critical care medicine.
But Dr. Worsham, also a teaching associate at Harvard Medical School’s department of health care policy, expressed concern that factors figuring into the rankings by U.S. News and Dr. Turner’s alternative “are imperfect measures of medical school quality.”
For example, a student interested in research might favor federal research funding in their customized rankings with Dr. Turner’s model. “But higher research funding doesn’t necessarily translate into a better education for students, particularly when differentiating between two major research systems,” Dr. Worsham noted.
Dr. Worsham added that neither ranking system accurately predicts the quality of doctors graduating from the schools. Instead, he’d like to see ranking systems based on which schools’ graduates deliver the best patient outcomes, whether that’s through direct patient care, impactful research, or leadership within the health care system.
Michael Sauder, PhD, professor of sociology at the University of Iowa, Iowa City, said the model could offer a valuable alternative to the U.S. News ranking system. It might help users develop their own criteria for determining the ranking of medical schools, which is a big improvement over a “one-size-fits-all” approach, Dr. Sauder said.
And Hanna Stotland, an admission consultant based in Chicago, noted that most students rely on rankings because they “don’t have the luxury of advisers who know the ins and outs of different medical schools.” Given the role that rankings play, Ms. Stotland expects that every new ranking tool will have some influence on students.
This tool in particular “has the potential to be useful for students who have identified values they want their medical school to share.” For example, students who care about racial diversity “could use it to easily identify schools that are successful on that metric,” Ms. Stotland said.
Sujay Ratna, a 2nd-year med student at Icahn School of Medicine at Mount Sinai in New York, said he considered the U.S. News ranking his “go-to tool” when he was applying to med school.
But after reading Dr. Turner’s article, the AMSA membership vice president tried the algorithm. “I definitely would have used it had it existed when I was thinking of what schools to apply to and what [schools] to attend.”
The study had no specific funding. Dr. Turner, Dr. Worsham, Dr. Sauder, Ms. Stotland, Ms. Brett, and Mr. Ratna report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For decades, pre-med students depended on the annual medical school rankings by U.S. News and World Report to decide where to apply for physician education. But after several prominent med schools pulled out of the rankings, one resident began experimenting with artificial intelligence (AI) to create an alternative.
Brandon Turner MD, MSc, a radiation oncology resident at Massachusetts General Hospital in Boston, developed a free do-it-yourself tool using AI that allows prospective students to rank medical schools based on considerations that are most important to them. His research was published online in JAMA Network Open.
“One of the flaws with conventional ranking systems is that the metrics used in these tools are weighted based on the preferences and views of the people who developed these rankings, but those may not work for everyone,” Dr. Turner told this news organization.
He explained that there are different types of metrics used in the U.S. News ranking: one for research and the other for primary care. “The research rankings carry the most prestige and are the ones that most people know about,” he explained. These metrics take into account factors such as how many grant dollars the medical school receives and the average size of those grants per faculty member, Dr. Turner said.
Admission metrics are also included – for example, the median grade point average or MCAT scores of students who have been accepted. “These don’t tell you anything about the research output of the school, only about how selective the school is,” he said.
Primary care metrics might focus on how many graduates of a given school go into primary care, or how other schools rate the quality of primary care training at a given school – a process called peer assessment, Dr. Turner said.
But even though these might be helpful, students may be more interested in the cost of attendance, average debt, representation of minorities, and how many graduates pass their boards, he said. “U.S. News metrics don’t capture these things, but I included them in my algorithm.”
A U.S. News spokesperson said that the publication continues to help students and their families make decisions about their future education. The spokesperson cited U.S. News’ explanation of how it calculates its rankings. “A school’s overall Best Medical Schools rank should be one consideration and not the lone determinant in where a student applies and accepts,” the article states.
Dr. Turner agreed ranking systems are a good starting point when researching med schools, “but the values reflected in the ranking may not reflect an individual’s goals.”
Tyra-Lee Brett, a premed student at the University of South Florida, Tampa, believes an additional tool for students to evaluate medical schools is needed – and she could potentially see herself using Dr. Turner’s creation.
Still, Ms. Brett, a premed trustee of the American Medical Student Association, doesn’t regard any ranking tool as the “be all and end all.” Rather, she feels that the most effective tool would be based on students’ lived experiences. The AMSA is developing a scorecard in which students grade schools based on their opinions about such issues as housing, family planning, and environmental health, she said.
No prior judgments
To develop his algorithm, Dr. Turner used a branch of AI called “unsupervised learning.” It doesn’t make a prior judgment about what the data should look like, Dr. Turner explained.
“You’re just analyzing natural trends within the data.”
The algorithm tries to find and discover clusters or patterns within the data. “It’s like saying to the algorithm: ‘I want you to tell me what schools you think should be grouped together based on the data I feed you,’ which is the data that the user selects based on his or her personal preferences.”
U.S. News has been transparent about the metrics it uses, Dr. Turner notes. “When I started looking into how rankings are developed, I saw that there was transparency, and the reasoning for choosing the metrics used to develop the ranking was pretty sound,” he said.
“But I didn’t see any justification as to why they chose the particular metrics and weighted them in the way that they did.”
Dr. Turner extracted data from the 2023 U.S. News report, which ranked 109 allopathic medical schools, and applied several scenarios to the results to create his alternative ranking system.
In one scenario, he used the same research metrics used by U.S. News, such as a peer research assessment, median federal research activity per full-time faculty member, median GPA, median MCAT, acceptance rate, and faculty-student ratio.
In another scenario, he included four additional metrics: debt, in-state cost of attendance, USMLE Step 1 passing rate, and percentage of underrepresented students with minority race or ethnicity at the school.
For example, a user can rank the importance of the diversity of the class, amount of debt students expect to incur, and amount of research funding the medical school receives. After selecting those factors, the tool generates tiered results displayed in a circle, a shape chosen to avoid the appearance of the hierarchy associated with traditional rankings, Dr. Turner said.
“A prospective student might not care about acceptance rates and MCAT scores, and instead cares about diversity and debt,” Dr. Turner said. He looks forward to extending this approach to the ranking of colleges as well.
‘Imperfect measures’
“The model and interesting online tool that Dr. Turner created allows a premed [student] to generate custom rankings that are in line with their own priorities,” said Christopher Worsham, MD, MPH, a critical care physician in Mass General’s division of pulmonary and critical care medicine.
But Dr. Worsham, also a teaching associate at Harvard Medical School’s department of health care policy, expressed concern that factors figuring into the rankings by U.S. News and Dr. Turner’s alternative “are imperfect measures of medical school quality.”
For example, a student interested in research might favor federal research funding in their customized rankings with Dr. Turner’s model. “But higher research funding doesn’t necessarily translate into a better education for students, particularly when differentiating between two major research systems,” Dr. Worsham noted.
Dr. Worsham added that neither ranking system accurately predicts the quality of doctors graduating from the schools. Instead, he’d like to see ranking systems based on which schools’ graduates deliver the best patient outcomes, whether that’s through direct patient care, impactful research, or leadership within the health care system.
Michael Sauder, PhD, professor of sociology at the University of Iowa, Iowa City, said the model could offer a valuable alternative to the U.S. News ranking system. It might help users develop their own criteria for determining the ranking of medical schools, which is a big improvement over a “one-size-fits-all” approach, Dr. Sauder said.
And Hanna Stotland, an admission consultant based in Chicago, noted that most students rely on rankings because they “don’t have the luxury of advisers who know the ins and outs of different medical schools.” Given the role that rankings play, Ms. Stotland expects that every new ranking tool will have some influence on students.
This tool in particular “has the potential to be useful for students who have identified values they want their medical school to share.” For example, students who care about racial diversity “could use it to easily identify schools that are successful on that metric,” Ms. Stotland said.
Sujay Ratna, a 2nd-year med student at Icahn School of Medicine at Mount Sinai in New York, said he considered the U.S. News ranking his “go-to tool” when he was applying to med school.
But after reading Dr. Turner’s article, the AMSA membership vice president tried the algorithm. “I definitely would have used it had it existed when I was thinking of what schools to apply to and what [schools] to attend.”
The study had no specific funding. Dr. Turner, Dr. Worsham, Dr. Sauder, Ms. Stotland, Ms. Brett, and Mr. Ratna report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Q&A: What to know about the new BA 2.86 COVID variant
The Centers for Disease Control and Prevention and the World Health Organization have dubbed the BA 2.86 variant of COVID-19 as a variant to watch.
So far, only 26 cases of “Pirola,” as the new variant is being called, have been identified: 10 in Denmark, four each in Sweden and the United States, three in South Africa, two in Portugal, and one each the United Kingdom, Israel, and Canada. BA 2.86 is a subvariant of Omicron, but according to reports from the CDC, the strain has many more mutations than the ones that came before it.
With so many facts still unknown about this new variant, this news organization asked experts what people need to be aware of as it continues to spread.
What is unique about the BA 2.86 variant?
“It is unique in that it has more than three mutations on the spike protein,” said Purvi S. Parikh, MD, an infectious disease expert at New York University’s Langone Health. The virus uses the spike proteins to enter our cells.
This “may mean it will be more transmissible, cause more severe disease, and/or our vaccines and treatments may not work as well, as compared to other variants,” she said.
What do we need to watch with BA 2.86 going forward?
“We don’t know if this variant will be associated with a change in the disease severity. We currently see increased numbers of cases in general, even though we don’t yet see the BA.2.86 in our system,” said Heba Mostafa, PhD, director of the molecular virology laboratory at Johns Hopkins Hospital in Baltimore.
“It is important to monitor BA.2.86 (and other variants) and understand how its evolution impacts the number of cases and disease outcomes,” she said. “We should all be aware of the current increase in cases, though, and try to get tested and be treated as soon as possible, as antivirals should be effective against the circulating variants.”
What should doctors know?
Dr. Parikh said doctors should generally expect more COVID cases in their clinics and make sure to screen patients even if their symptoms are mild.
“We have tools that can be used – antivirals like Paxlovid are still efficacious with current dominant strains such as EG.5,” she said. “And encourage your patients to get their boosters, mask, wash hands, and social distance.”
How well can our vaccines fight BA 2.86?
“Vaccine coverage for the BA.2.86 is an area of uncertainty right now,” said Dr. Mostafa.
In its report, the CDC said scientists are still figuring out how well the updated COVID vaccine works. It’s expected to be available in the fall, and for now, they believe the new shot will still make infections less severe, new variants and all.
Prior vaccinations and infections have created antibodies in many people, and that will likely provide some protection, Dr. Mostafa said. “When we experienced the Omicron wave in December 2021, even though the variant was distant from what circulated before its emergence and was associated with a very large increase in the number of cases, vaccinations were still protective against severe disease.”
What is the most important thing to keep track of when it comes to this variant?
According to Dr. Parikh, “it’s most important to monitor how transmissible [BA 2.86] is, how severe it is, and if our current treatments and vaccines work.”
Dr. Mostafa said how well the new variants escape existing antibody protection should also be studied and watched closely.
What does this stage of the virus mutation tell us about where we are in the pandemic?
The history of the coronavirus over the past few years shows that variants with many changes evolve and can spread very quickly, Dr. Mostafa said. “Now that the virus is endemic, it is essential to monitor, update vaccinations if necessary, diagnose, treat, and implement infection control measures when necessary.”
With the limited data we have so far, experts seem to agree that while the variant’s makeup raises some red flags, it is too soon to jump to any conclusions about how easy it is to catch it and the ways it may change how the virus impacts those who contract it.
A version of this article first appeared on WebMD.com.
The Centers for Disease Control and Prevention and the World Health Organization have dubbed the BA 2.86 variant of COVID-19 as a variant to watch.
So far, only 26 cases of “Pirola,” as the new variant is being called, have been identified: 10 in Denmark, four each in Sweden and the United States, three in South Africa, two in Portugal, and one each the United Kingdom, Israel, and Canada. BA 2.86 is a subvariant of Omicron, but according to reports from the CDC, the strain has many more mutations than the ones that came before it.
With so many facts still unknown about this new variant, this news organization asked experts what people need to be aware of as it continues to spread.
What is unique about the BA 2.86 variant?
“It is unique in that it has more than three mutations on the spike protein,” said Purvi S. Parikh, MD, an infectious disease expert at New York University’s Langone Health. The virus uses the spike proteins to enter our cells.
This “may mean it will be more transmissible, cause more severe disease, and/or our vaccines and treatments may not work as well, as compared to other variants,” she said.
What do we need to watch with BA 2.86 going forward?
“We don’t know if this variant will be associated with a change in the disease severity. We currently see increased numbers of cases in general, even though we don’t yet see the BA.2.86 in our system,” said Heba Mostafa, PhD, director of the molecular virology laboratory at Johns Hopkins Hospital in Baltimore.
“It is important to monitor BA.2.86 (and other variants) and understand how its evolution impacts the number of cases and disease outcomes,” she said. “We should all be aware of the current increase in cases, though, and try to get tested and be treated as soon as possible, as antivirals should be effective against the circulating variants.”
What should doctors know?
Dr. Parikh said doctors should generally expect more COVID cases in their clinics and make sure to screen patients even if their symptoms are mild.
“We have tools that can be used – antivirals like Paxlovid are still efficacious with current dominant strains such as EG.5,” she said. “And encourage your patients to get their boosters, mask, wash hands, and social distance.”
How well can our vaccines fight BA 2.86?
“Vaccine coverage for the BA.2.86 is an area of uncertainty right now,” said Dr. Mostafa.
In its report, the CDC said scientists are still figuring out how well the updated COVID vaccine works. It’s expected to be available in the fall, and for now, they believe the new shot will still make infections less severe, new variants and all.
Prior vaccinations and infections have created antibodies in many people, and that will likely provide some protection, Dr. Mostafa said. “When we experienced the Omicron wave in December 2021, even though the variant was distant from what circulated before its emergence and was associated with a very large increase in the number of cases, vaccinations were still protective against severe disease.”
What is the most important thing to keep track of when it comes to this variant?
According to Dr. Parikh, “it’s most important to monitor how transmissible [BA 2.86] is, how severe it is, and if our current treatments and vaccines work.”
Dr. Mostafa said how well the new variants escape existing antibody protection should also be studied and watched closely.
What does this stage of the virus mutation tell us about where we are in the pandemic?
The history of the coronavirus over the past few years shows that variants with many changes evolve and can spread very quickly, Dr. Mostafa said. “Now that the virus is endemic, it is essential to monitor, update vaccinations if necessary, diagnose, treat, and implement infection control measures when necessary.”
With the limited data we have so far, experts seem to agree that while the variant’s makeup raises some red flags, it is too soon to jump to any conclusions about how easy it is to catch it and the ways it may change how the virus impacts those who contract it.
A version of this article first appeared on WebMD.com.
The Centers for Disease Control and Prevention and the World Health Organization have dubbed the BA 2.86 variant of COVID-19 as a variant to watch.
So far, only 26 cases of “Pirola,” as the new variant is being called, have been identified: 10 in Denmark, four each in Sweden and the United States, three in South Africa, two in Portugal, and one each the United Kingdom, Israel, and Canada. BA 2.86 is a subvariant of Omicron, but according to reports from the CDC, the strain has many more mutations than the ones that came before it.
With so many facts still unknown about this new variant, this news organization asked experts what people need to be aware of as it continues to spread.
What is unique about the BA 2.86 variant?
“It is unique in that it has more than three mutations on the spike protein,” said Purvi S. Parikh, MD, an infectious disease expert at New York University’s Langone Health. The virus uses the spike proteins to enter our cells.
This “may mean it will be more transmissible, cause more severe disease, and/or our vaccines and treatments may not work as well, as compared to other variants,” she said.
What do we need to watch with BA 2.86 going forward?
“We don’t know if this variant will be associated with a change in the disease severity. We currently see increased numbers of cases in general, even though we don’t yet see the BA.2.86 in our system,” said Heba Mostafa, PhD, director of the molecular virology laboratory at Johns Hopkins Hospital in Baltimore.
“It is important to monitor BA.2.86 (and other variants) and understand how its evolution impacts the number of cases and disease outcomes,” she said. “We should all be aware of the current increase in cases, though, and try to get tested and be treated as soon as possible, as antivirals should be effective against the circulating variants.”
What should doctors know?
Dr. Parikh said doctors should generally expect more COVID cases in their clinics and make sure to screen patients even if their symptoms are mild.
“We have tools that can be used – antivirals like Paxlovid are still efficacious with current dominant strains such as EG.5,” she said. “And encourage your patients to get their boosters, mask, wash hands, and social distance.”
How well can our vaccines fight BA 2.86?
“Vaccine coverage for the BA.2.86 is an area of uncertainty right now,” said Dr. Mostafa.
In its report, the CDC said scientists are still figuring out how well the updated COVID vaccine works. It’s expected to be available in the fall, and for now, they believe the new shot will still make infections less severe, new variants and all.
Prior vaccinations and infections have created antibodies in many people, and that will likely provide some protection, Dr. Mostafa said. “When we experienced the Omicron wave in December 2021, even though the variant was distant from what circulated before its emergence and was associated with a very large increase in the number of cases, vaccinations were still protective against severe disease.”
What is the most important thing to keep track of when it comes to this variant?
According to Dr. Parikh, “it’s most important to monitor how transmissible [BA 2.86] is, how severe it is, and if our current treatments and vaccines work.”
Dr. Mostafa said how well the new variants escape existing antibody protection should also be studied and watched closely.
What does this stage of the virus mutation tell us about where we are in the pandemic?
The history of the coronavirus over the past few years shows that variants with many changes evolve and can spread very quickly, Dr. Mostafa said. “Now that the virus is endemic, it is essential to monitor, update vaccinations if necessary, diagnose, treat, and implement infection control measures when necessary.”
With the limited data we have so far, experts seem to agree that while the variant’s makeup raises some red flags, it is too soon to jump to any conclusions about how easy it is to catch it and the ways it may change how the virus impacts those who contract it.
A version of this article first appeared on WebMD.com.
Commentary: Cardiovascular risk, anti-drug antibodies, and prednisolone in RA, September 2023
Anti-drug antibody (ADA) testing for biologics, particularly anti–tumor necrosis factor (TNF) agents, has been commercially available for several years, though its clinical use in rheumatoid arthritis (RA) is not known owing to lack of prospective data. Bitoun and colleagues analyzed data from the European ABI-RA registry to evaluate the association between ADA and the anti–TNF monoclonal antibodies (mAb) etanercept, tocilizumab, and rituximab, and clinical response (as measured by disease activity scores, inflammatory markers, and European Alliance of Associations for Rheumatology [EULAR] response rate). Higher rates of ADA positivity were seen in patients treated with rituximab (50%), anti-TNF mAb (38%), and tocilizumab (20%) compared with etanercept (6%). Patients who had a positive ADA test were less likely to have a EULAR response. In addition, patients treated with methotrexate were less likely to have persistent ADA. Though the study was not powered enough to detect differences between the drug classes, the evidence presented is compelling and suggests a role for measuring ADA in patients with RA who do not respond to treatment.
RA is well-known to be associated with cardiovascular disease, particularly atherosclerotic disease and heart failure, but its association with valvular heart disease and its progression has not been well-explored in the literature. Johnson and colleagues performed a cohort study of over 73,000 patients with RA in the Veterans Health Administration (VHA) system compared with 640,000 patients without RA to evaluate the incidence of aortic stenosis, need for intervention, and risk for death. Though the overall incidence rate was low (about 3%), patients with RA had a higher risk for aortic stenosis, with a hazard ratio of 1.48 compared with those without RA, as well as a higher risk for aortic valve replacement and aortic stenosis–related death. The risk for aortic stenosis was associated with hypertension, stroke, and other cardiovascular disease, as well as a body mass index > 30 kg/m2, although not with a history of smoking or diabetes. Because the study was performed using data from the VHA — that is, from predominantly male patients — this finding may not be generalizable. In addition, the diagnosis of aortic stenosis is generally reliant on echocardiography and may be detected while searching for other conditions not evaluated here (such as pericarditis). As such, these findings would not support routine screening in patients with RA without other reasons for suspicion of valvular heart disease.
In particular, the increase in cardiovascular risk associated with glucocorticoid therapy in patients with RA has received increased scrutiny, along with other side effects of systemic glucocorticoids. In a recent retrospective study, So and colleagues examined the clinical data of over 12,000 patients with RA treated in public hospitals in Hong Kong with a mean of 9 years of follow-up. Consistent with prior studies, systemic glucocorticoid use (prednisolone equivalent > 5 mg daily) was associated with an increased risk for adverse cardiovascular events, whereas lower doses did not increase cardiovascular risk. Because the data on some disease activity measures and traditional cardiovascular risk factors (such as smoking or obesity) were not available in the database, the study supports, but does not expand on, prior evidence regarding cardiovascular risk.
Almayali and colleagues also looked at glucocorticoid therapy in RA in a follow-up study to the previously published pragmatic randomized double-blinded placebo-controlled GLORIA study, which evaluated the effects of 5 mg/d prednisolone added to standard care for 2 years in patients with active RA who were age 65 years or older. In the current study, 191 patients out of the initial 451 were followed for 3 months and prednisolone tapered off. Patients who tapered off prednisolone had, as expected, an increased risk for flare but no evidence of adrenal insufficiency. Although, again, this is not likely to change practice, it does suggest that glucocorticoid tapering is a reasonable goal in RA therapeutic trials.
Anti-drug antibody (ADA) testing for biologics, particularly anti–tumor necrosis factor (TNF) agents, has been commercially available for several years, though its clinical use in rheumatoid arthritis (RA) is not known owing to lack of prospective data. Bitoun and colleagues analyzed data from the European ABI-RA registry to evaluate the association between ADA and the anti–TNF monoclonal antibodies (mAb) etanercept, tocilizumab, and rituximab, and clinical response (as measured by disease activity scores, inflammatory markers, and European Alliance of Associations for Rheumatology [EULAR] response rate). Higher rates of ADA positivity were seen in patients treated with rituximab (50%), anti-TNF mAb (38%), and tocilizumab (20%) compared with etanercept (6%). Patients who had a positive ADA test were less likely to have a EULAR response. In addition, patients treated with methotrexate were less likely to have persistent ADA. Though the study was not powered enough to detect differences between the drug classes, the evidence presented is compelling and suggests a role for measuring ADA in patients with RA who do not respond to treatment.
RA is well-known to be associated with cardiovascular disease, particularly atherosclerotic disease and heart failure, but its association with valvular heart disease and its progression has not been well-explored in the literature. Johnson and colleagues performed a cohort study of over 73,000 patients with RA in the Veterans Health Administration (VHA) system compared with 640,000 patients without RA to evaluate the incidence of aortic stenosis, need for intervention, and risk for death. Though the overall incidence rate was low (about 3%), patients with RA had a higher risk for aortic stenosis, with a hazard ratio of 1.48 compared with those without RA, as well as a higher risk for aortic valve replacement and aortic stenosis–related death. The risk for aortic stenosis was associated with hypertension, stroke, and other cardiovascular disease, as well as a body mass index > 30 kg/m2, although not with a history of smoking or diabetes. Because the study was performed using data from the VHA — that is, from predominantly male patients — this finding may not be generalizable. In addition, the diagnosis of aortic stenosis is generally reliant on echocardiography and may be detected while searching for other conditions not evaluated here (such as pericarditis). As such, these findings would not support routine screening in patients with RA without other reasons for suspicion of valvular heart disease.
In particular, the increase in cardiovascular risk associated with glucocorticoid therapy in patients with RA has received increased scrutiny, along with other side effects of systemic glucocorticoids. In a recent retrospective study, So and colleagues examined the clinical data of over 12,000 patients with RA treated in public hospitals in Hong Kong with a mean of 9 years of follow-up. Consistent with prior studies, systemic glucocorticoid use (prednisolone equivalent > 5 mg daily) was associated with an increased risk for adverse cardiovascular events, whereas lower doses did not increase cardiovascular risk. Because the data on some disease activity measures and traditional cardiovascular risk factors (such as smoking or obesity) were not available in the database, the study supports, but does not expand on, prior evidence regarding cardiovascular risk.
Almayali and colleagues also looked at glucocorticoid therapy in RA in a follow-up study to the previously published pragmatic randomized double-blinded placebo-controlled GLORIA study, which evaluated the effects of 5 mg/d prednisolone added to standard care for 2 years in patients with active RA who were age 65 years or older. In the current study, 191 patients out of the initial 451 were followed for 3 months and prednisolone tapered off. Patients who tapered off prednisolone had, as expected, an increased risk for flare but no evidence of adrenal insufficiency. Although, again, this is not likely to change practice, it does suggest that glucocorticoid tapering is a reasonable goal in RA therapeutic trials.
Anti-drug antibody (ADA) testing for biologics, particularly anti–tumor necrosis factor (TNF) agents, has been commercially available for several years, though its clinical use in rheumatoid arthritis (RA) is not known owing to lack of prospective data. Bitoun and colleagues analyzed data from the European ABI-RA registry to evaluate the association between ADA and the anti–TNF monoclonal antibodies (mAb) etanercept, tocilizumab, and rituximab, and clinical response (as measured by disease activity scores, inflammatory markers, and European Alliance of Associations for Rheumatology [EULAR] response rate). Higher rates of ADA positivity were seen in patients treated with rituximab (50%), anti-TNF mAb (38%), and tocilizumab (20%) compared with etanercept (6%). Patients who had a positive ADA test were less likely to have a EULAR response. In addition, patients treated with methotrexate were less likely to have persistent ADA. Though the study was not powered enough to detect differences between the drug classes, the evidence presented is compelling and suggests a role for measuring ADA in patients with RA who do not respond to treatment.
RA is well-known to be associated with cardiovascular disease, particularly atherosclerotic disease and heart failure, but its association with valvular heart disease and its progression has not been well-explored in the literature. Johnson and colleagues performed a cohort study of over 73,000 patients with RA in the Veterans Health Administration (VHA) system compared with 640,000 patients without RA to evaluate the incidence of aortic stenosis, need for intervention, and risk for death. Though the overall incidence rate was low (about 3%), patients with RA had a higher risk for aortic stenosis, with a hazard ratio of 1.48 compared with those without RA, as well as a higher risk for aortic valve replacement and aortic stenosis–related death. The risk for aortic stenosis was associated with hypertension, stroke, and other cardiovascular disease, as well as a body mass index > 30 kg/m2, although not with a history of smoking or diabetes. Because the study was performed using data from the VHA — that is, from predominantly male patients — this finding may not be generalizable. In addition, the diagnosis of aortic stenosis is generally reliant on echocardiography and may be detected while searching for other conditions not evaluated here (such as pericarditis). As such, these findings would not support routine screening in patients with RA without other reasons for suspicion of valvular heart disease.
In particular, the increase in cardiovascular risk associated with glucocorticoid therapy in patients with RA has received increased scrutiny, along with other side effects of systemic glucocorticoids. In a recent retrospective study, So and colleagues examined the clinical data of over 12,000 patients with RA treated in public hospitals in Hong Kong with a mean of 9 years of follow-up. Consistent with prior studies, systemic glucocorticoid use (prednisolone equivalent > 5 mg daily) was associated with an increased risk for adverse cardiovascular events, whereas lower doses did not increase cardiovascular risk. Because the data on some disease activity measures and traditional cardiovascular risk factors (such as smoking or obesity) were not available in the database, the study supports, but does not expand on, prior evidence regarding cardiovascular risk.
Almayali and colleagues also looked at glucocorticoid therapy in RA in a follow-up study to the previously published pragmatic randomized double-blinded placebo-controlled GLORIA study, which evaluated the effects of 5 mg/d prednisolone added to standard care for 2 years in patients with active RA who were age 65 years or older. In the current study, 191 patients out of the initial 451 were followed for 3 months and prednisolone tapered off. Patients who tapered off prednisolone had, as expected, an increased risk for flare but no evidence of adrenal insufficiency. Although, again, this is not likely to change practice, it does suggest that glucocorticoid tapering is a reasonable goal in RA therapeutic trials.
ACR: Rheumatologists help reduce ED, hospitalization costs
Rheumatology care can save health systems more than $2,700 per patient per year, according to a new report from the American College of Rheumatology.
In a white paper and corresponding position statement, the organization outlined how rheumatology care delivers financial benefits for health systems.
The work also highlighted prior research on the positive outcomes associated with rheumatology care, including a decline in hip and knee replacements for patients with rheumatoid arthritis after the introduction of biologics, while the total number of hip and knee replacements for patients with osteoarthritis increased, as well as lower 30-day readmission rates among patients with systemic lupus erythematosus with access to a rheumatology clinic post discharge.
“Many rheumatologists can attest to the value they bring to the care team at a health care system,” said Christina Downey, MD, an assistant professor of medicine at Loma Linda (Calif.) University, in a press release. She is the lead author of the white paper and chair of the ACR’s Government Affairs Committee. “Our goal with the paper and position statement is to emphasize what that value looks like from a preventive and financial perspective. A rheumatologist on the care team benefits patients, practices, and the economy.”
The analysis used adjusted claims insurance data to compare markets with a high vs. low supply of rheumatologists. A high supply was defined as at least 1.5 rheumatologists per 100,000 population, whereas a low supply was less than this amount. On average, markets with a high supply of rheumatologists had lower emergency department (ED) and hospitalization costs per patient per year.
Added together, high-supply rheumatology markets save on average $2,762 in ED visit and hospitalization costs per patient per year.
Dr. Downey and colleagues also tallied the direct and downstream billings associated with rheumatologists, including office visits, consultations, lab testing, and radiology services. The average revenue generated per rheumatologist was $3.5 million per year.
“Emphasizing the impact rheumatologists have on the entire medical community is more important than ever, especially as we contend with an impending rheumatology workforce shortage coupled with an expected increase in patient demand for rheumatologic care,” Dr. Downey said. “This paper supports our recruitment and sustainability efforts for the specialty by spotlighting the significant contributions we make every day and every year to patient outcomes, hospitals, and other health care practices.”
A version of this article first appeared on Medscape.com.
Rheumatology care can save health systems more than $2,700 per patient per year, according to a new report from the American College of Rheumatology.
In a white paper and corresponding position statement, the organization outlined how rheumatology care delivers financial benefits for health systems.
The work also highlighted prior research on the positive outcomes associated with rheumatology care, including a decline in hip and knee replacements for patients with rheumatoid arthritis after the introduction of biologics, while the total number of hip and knee replacements for patients with osteoarthritis increased, as well as lower 30-day readmission rates among patients with systemic lupus erythematosus with access to a rheumatology clinic post discharge.
“Many rheumatologists can attest to the value they bring to the care team at a health care system,” said Christina Downey, MD, an assistant professor of medicine at Loma Linda (Calif.) University, in a press release. She is the lead author of the white paper and chair of the ACR’s Government Affairs Committee. “Our goal with the paper and position statement is to emphasize what that value looks like from a preventive and financial perspective. A rheumatologist on the care team benefits patients, practices, and the economy.”
The analysis used adjusted claims insurance data to compare markets with a high vs. low supply of rheumatologists. A high supply was defined as at least 1.5 rheumatologists per 100,000 population, whereas a low supply was less than this amount. On average, markets with a high supply of rheumatologists had lower emergency department (ED) and hospitalization costs per patient per year.
Added together, high-supply rheumatology markets save on average $2,762 in ED visit and hospitalization costs per patient per year.
Dr. Downey and colleagues also tallied the direct and downstream billings associated with rheumatologists, including office visits, consultations, lab testing, and radiology services. The average revenue generated per rheumatologist was $3.5 million per year.
“Emphasizing the impact rheumatologists have on the entire medical community is more important than ever, especially as we contend with an impending rheumatology workforce shortage coupled with an expected increase in patient demand for rheumatologic care,” Dr. Downey said. “This paper supports our recruitment and sustainability efforts for the specialty by spotlighting the significant contributions we make every day and every year to patient outcomes, hospitals, and other health care practices.”
A version of this article first appeared on Medscape.com.
Rheumatology care can save health systems more than $2,700 per patient per year, according to a new report from the American College of Rheumatology.
In a white paper and corresponding position statement, the organization outlined how rheumatology care delivers financial benefits for health systems.
The work also highlighted prior research on the positive outcomes associated with rheumatology care, including a decline in hip and knee replacements for patients with rheumatoid arthritis after the introduction of biologics, while the total number of hip and knee replacements for patients with osteoarthritis increased, as well as lower 30-day readmission rates among patients with systemic lupus erythematosus with access to a rheumatology clinic post discharge.
“Many rheumatologists can attest to the value they bring to the care team at a health care system,” said Christina Downey, MD, an assistant professor of medicine at Loma Linda (Calif.) University, in a press release. She is the lead author of the white paper and chair of the ACR’s Government Affairs Committee. “Our goal with the paper and position statement is to emphasize what that value looks like from a preventive and financial perspective. A rheumatologist on the care team benefits patients, practices, and the economy.”
The analysis used adjusted claims insurance data to compare markets with a high vs. low supply of rheumatologists. A high supply was defined as at least 1.5 rheumatologists per 100,000 population, whereas a low supply was less than this amount. On average, markets with a high supply of rheumatologists had lower emergency department (ED) and hospitalization costs per patient per year.
Added together, high-supply rheumatology markets save on average $2,762 in ED visit and hospitalization costs per patient per year.
Dr. Downey and colleagues also tallied the direct and downstream billings associated with rheumatologists, including office visits, consultations, lab testing, and radiology services. The average revenue generated per rheumatologist was $3.5 million per year.
“Emphasizing the impact rheumatologists have on the entire medical community is more important than ever, especially as we contend with an impending rheumatology workforce shortage coupled with an expected increase in patient demand for rheumatologic care,” Dr. Downey said. “This paper supports our recruitment and sustainability efforts for the specialty by spotlighting the significant contributions we make every day and every year to patient outcomes, hospitals, and other health care practices.”
A version of this article first appeared on Medscape.com.
Diabetes drug class appears to reduce recurrent gout flares
The glucose-lowering drug class sodium-glucose cotransporter 2 (SGLT2) inhibitors appear to reduce the risk for recurrent gout flares in people with gout and type 2 diabetes, and to lessen excess mortality in those individuals, compared with those who initiated other types of glucose-lowering medications, new data suggest.
Among nearly 6,000 adults with both type 2 diabetes and gout from a U.K. primary care database, initiation of SGLT2 inhibitor treatment was associated with 19% fewer recurrent gout flares and 29% lower mortality.
Moreover, unlike other urate-lowering therapies, there were no apparent transient increases in the risk of gout flares after initiating therapy, Jie Wei, PhD, of Health Management Center, Xiangya Hospital, Central South University, Changsha, China, and colleagues reported in JAMA Network Open.
These results are important because current management of gout is suboptimal. Many patients either don’t receive adequate urate-lowering therapies such as allopurinol or stop taking them, Dr. Wei and colleagues said.
In addition to lowering glucose, SGLT2 inhibitors also reduce the risk for major adverse cardiovascular events and all-cause mortality in people regardless of their diabetes status. Previous studies have also found that SGLT2 inhibitors reduce the risk for developing gout and of gout flares.
Asked to comment, gout specialist John D. FitzGerald, MD, PhD, clinical chief of rheumatology at the University of California, Los Angeles, said in an interview: “I think it’s a well-done paper, with a large dataset. I think it just reinforces the findings from the other papers. Mostly anything that lowers uric acid levels is going to lower recurrent gout attacks, so it all makes sense.”
However, while Dr. FitzGerald thinks the drug class is a good option for people with diabetes or cardiorenal indications for them who also have gout, he doesn’t envision it as first-line for most other patients with gout. “The current treatments are very effective. Allopurinol brings down uric acid levels by 5-7 points. There are patients who fail allopurinol, but those are less than 5%.”
The most common reason patients stop taking allopurinol is the frequent initial gout flare. But that’s preventable, Dr. FitzGerald said, either by titrating up slowly, or by adding colchicine along with it. “By going slowly, you can avoid that flare risk. I think that’s what’s going on with the SGLT2 inhibitor. It’s not a dramatic urate-lowering drug, but it is clinically meaningful. I think that’s what this paper is showing.”
But, he noted, “I think there are so many reasons to start the SGLT2 inhibitors that if somebody also has gout, all the better. And, if somebody is on the margin with diabetes and gout control and can’t go with allopurinol, it would be great to add for both conditions.”
Less gout recurrence, lower mortality
The retrospective study was conducted from Jan. 1, 2013, to March 31, 2022. Among 5,931 patients with both type 2 diabetes and gout, 1,548 (26.1%) initiated an SGLT2 inhibitor (dapagliflozin, empagliflozin, or canagliflozin), while 4,383 (73.9%) initiated treatment with other active comparators, mostly (92.6%) dipeptidyl peptidase–4 inhibitors.
Gout flares were identified in the charts for a total of 86% of the participants. The weighted incidence rates for the first recurrent flare were 32.4 versus 41.2 per 1,000 person-years in the SGLT2 inhibitor versus comparator groups, with a weighted absolute rate difference of –8.8/1,000 and weighted hazard ratio of 0.81, a significant difference.
All-cause mortality was 18.8 versus 24.9 per 1,000 person-years, respectively, giving an HR of 0.71 at 5-year follow-up.
Dr. FitzGerald, who chaired the American College of Rheumatology’s 2020 gout guidelines, said he anticipates that the SGLT2 inhibitors will be mentioned in the next update to the ACR’s now “living” guidelines, although he was not speaking on the organization’s behalf.
“We talk about losartan in the current [ACR guidelines], about its specific uric acid–lowering effect. Drugs can make uric acid worse or better. For example, thiazides make it higher. I think the SGLT2 [inhibitors] are important, but I don’t think they’re huge. The study is great, and I think the drugs are great, but I don’t think they will change the way gout is managed.”
This work was supported by grants from the National Key Research and Development Plan, the National Natural Science Foundation of China, the Project Program of National Clinical Research Center for Geriatric Disorders, and from the Natural Science Foundation of Hunan Province. Dr. Wei reported receiving grant funding from Xiangya Hospital Central South University Project Program of National Clinical Research Center for Geriatric Disorders and the Science and Technology Department of Hunan Province, the Natural Science Foundation of Hunan Province, during the conduct of the study. Dr. FitzGerald reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The glucose-lowering drug class sodium-glucose cotransporter 2 (SGLT2) inhibitors appear to reduce the risk for recurrent gout flares in people with gout and type 2 diabetes, and to lessen excess mortality in those individuals, compared with those who initiated other types of glucose-lowering medications, new data suggest.
Among nearly 6,000 adults with both type 2 diabetes and gout from a U.K. primary care database, initiation of SGLT2 inhibitor treatment was associated with 19% fewer recurrent gout flares and 29% lower mortality.
Moreover, unlike other urate-lowering therapies, there were no apparent transient increases in the risk of gout flares after initiating therapy, Jie Wei, PhD, of Health Management Center, Xiangya Hospital, Central South University, Changsha, China, and colleagues reported in JAMA Network Open.
These results are important because current management of gout is suboptimal. Many patients either don’t receive adequate urate-lowering therapies such as allopurinol or stop taking them, Dr. Wei and colleagues said.
In addition to lowering glucose, SGLT2 inhibitors also reduce the risk for major adverse cardiovascular events and all-cause mortality in people regardless of their diabetes status. Previous studies have also found that SGLT2 inhibitors reduce the risk for developing gout and of gout flares.
Asked to comment, gout specialist John D. FitzGerald, MD, PhD, clinical chief of rheumatology at the University of California, Los Angeles, said in an interview: “I think it’s a well-done paper, with a large dataset. I think it just reinforces the findings from the other papers. Mostly anything that lowers uric acid levels is going to lower recurrent gout attacks, so it all makes sense.”
However, while Dr. FitzGerald thinks the drug class is a good option for people with diabetes or cardiorenal indications for them who also have gout, he doesn’t envision it as first-line for most other patients with gout. “The current treatments are very effective. Allopurinol brings down uric acid levels by 5-7 points. There are patients who fail allopurinol, but those are less than 5%.”
The most common reason patients stop taking allopurinol is the frequent initial gout flare. But that’s preventable, Dr. FitzGerald said, either by titrating up slowly, or by adding colchicine along with it. “By going slowly, you can avoid that flare risk. I think that’s what’s going on with the SGLT2 inhibitor. It’s not a dramatic urate-lowering drug, but it is clinically meaningful. I think that’s what this paper is showing.”
But, he noted, “I think there are so many reasons to start the SGLT2 inhibitors that if somebody also has gout, all the better. And, if somebody is on the margin with diabetes and gout control and can’t go with allopurinol, it would be great to add for both conditions.”
Less gout recurrence, lower mortality
The retrospective study was conducted from Jan. 1, 2013, to March 31, 2022. Among 5,931 patients with both type 2 diabetes and gout, 1,548 (26.1%) initiated an SGLT2 inhibitor (dapagliflozin, empagliflozin, or canagliflozin), while 4,383 (73.9%) initiated treatment with other active comparators, mostly (92.6%) dipeptidyl peptidase–4 inhibitors.
Gout flares were identified in the charts for a total of 86% of the participants. The weighted incidence rates for the first recurrent flare were 32.4 versus 41.2 per 1,000 person-years in the SGLT2 inhibitor versus comparator groups, with a weighted absolute rate difference of –8.8/1,000 and weighted hazard ratio of 0.81, a significant difference.
All-cause mortality was 18.8 versus 24.9 per 1,000 person-years, respectively, giving an HR of 0.71 at 5-year follow-up.
Dr. FitzGerald, who chaired the American College of Rheumatology’s 2020 gout guidelines, said he anticipates that the SGLT2 inhibitors will be mentioned in the next update to the ACR’s now “living” guidelines, although he was not speaking on the organization’s behalf.
“We talk about losartan in the current [ACR guidelines], about its specific uric acid–lowering effect. Drugs can make uric acid worse or better. For example, thiazides make it higher. I think the SGLT2 [inhibitors] are important, but I don’t think they’re huge. The study is great, and I think the drugs are great, but I don’t think they will change the way gout is managed.”
This work was supported by grants from the National Key Research and Development Plan, the National Natural Science Foundation of China, the Project Program of National Clinical Research Center for Geriatric Disorders, and from the Natural Science Foundation of Hunan Province. Dr. Wei reported receiving grant funding from Xiangya Hospital Central South University Project Program of National Clinical Research Center for Geriatric Disorders and the Science and Technology Department of Hunan Province, the Natural Science Foundation of Hunan Province, during the conduct of the study. Dr. FitzGerald reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The glucose-lowering drug class sodium-glucose cotransporter 2 (SGLT2) inhibitors appear to reduce the risk for recurrent gout flares in people with gout and type 2 diabetes, and to lessen excess mortality in those individuals, compared with those who initiated other types of glucose-lowering medications, new data suggest.
Among nearly 6,000 adults with both type 2 diabetes and gout from a U.K. primary care database, initiation of SGLT2 inhibitor treatment was associated with 19% fewer recurrent gout flares and 29% lower mortality.
Moreover, unlike other urate-lowering therapies, there were no apparent transient increases in the risk of gout flares after initiating therapy, Jie Wei, PhD, of Health Management Center, Xiangya Hospital, Central South University, Changsha, China, and colleagues reported in JAMA Network Open.
These results are important because current management of gout is suboptimal. Many patients either don’t receive adequate urate-lowering therapies such as allopurinol or stop taking them, Dr. Wei and colleagues said.
In addition to lowering glucose, SGLT2 inhibitors also reduce the risk for major adverse cardiovascular events and all-cause mortality in people regardless of their diabetes status. Previous studies have also found that SGLT2 inhibitors reduce the risk for developing gout and of gout flares.
Asked to comment, gout specialist John D. FitzGerald, MD, PhD, clinical chief of rheumatology at the University of California, Los Angeles, said in an interview: “I think it’s a well-done paper, with a large dataset. I think it just reinforces the findings from the other papers. Mostly anything that lowers uric acid levels is going to lower recurrent gout attacks, so it all makes sense.”
However, while Dr. FitzGerald thinks the drug class is a good option for people with diabetes or cardiorenal indications for them who also have gout, he doesn’t envision it as first-line for most other patients with gout. “The current treatments are very effective. Allopurinol brings down uric acid levels by 5-7 points. There are patients who fail allopurinol, but those are less than 5%.”
The most common reason patients stop taking allopurinol is the frequent initial gout flare. But that’s preventable, Dr. FitzGerald said, either by titrating up slowly, or by adding colchicine along with it. “By going slowly, you can avoid that flare risk. I think that’s what’s going on with the SGLT2 inhibitor. It’s not a dramatic urate-lowering drug, but it is clinically meaningful. I think that’s what this paper is showing.”
But, he noted, “I think there are so many reasons to start the SGLT2 inhibitors that if somebody also has gout, all the better. And, if somebody is on the margin with diabetes and gout control and can’t go with allopurinol, it would be great to add for both conditions.”
Less gout recurrence, lower mortality
The retrospective study was conducted from Jan. 1, 2013, to March 31, 2022. Among 5,931 patients with both type 2 diabetes and gout, 1,548 (26.1%) initiated an SGLT2 inhibitor (dapagliflozin, empagliflozin, or canagliflozin), while 4,383 (73.9%) initiated treatment with other active comparators, mostly (92.6%) dipeptidyl peptidase–4 inhibitors.
Gout flares were identified in the charts for a total of 86% of the participants. The weighted incidence rates for the first recurrent flare were 32.4 versus 41.2 per 1,000 person-years in the SGLT2 inhibitor versus comparator groups, with a weighted absolute rate difference of –8.8/1,000 and weighted hazard ratio of 0.81, a significant difference.
All-cause mortality was 18.8 versus 24.9 per 1,000 person-years, respectively, giving an HR of 0.71 at 5-year follow-up.
Dr. FitzGerald, who chaired the American College of Rheumatology’s 2020 gout guidelines, said he anticipates that the SGLT2 inhibitors will be mentioned in the next update to the ACR’s now “living” guidelines, although he was not speaking on the organization’s behalf.
“We talk about losartan in the current [ACR guidelines], about its specific uric acid–lowering effect. Drugs can make uric acid worse or better. For example, thiazides make it higher. I think the SGLT2 [inhibitors] are important, but I don’t think they’re huge. The study is great, and I think the drugs are great, but I don’t think they will change the way gout is managed.”
This work was supported by grants from the National Key Research and Development Plan, the National Natural Science Foundation of China, the Project Program of National Clinical Research Center for Geriatric Disorders, and from the Natural Science Foundation of Hunan Province. Dr. Wei reported receiving grant funding from Xiangya Hospital Central South University Project Program of National Clinical Research Center for Geriatric Disorders and the Science and Technology Department of Hunan Province, the Natural Science Foundation of Hunan Province, during the conduct of the study. Dr. FitzGerald reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
NPs, PAs, and physicians hope to join doctors’ union in rare alliance
Advanced practice providers (APPs) such as nurse practitioners (NPs) and physician assistants (PAs) have long been at odds with doctor groups over scope of practice issues. But in a rare alliance, in late September. If successful, the Allina group will join the Doctors Council SEIU, Local 10MD.
The Allina health care providers share concerns about their working conditions, such as understaffing and inadequate resources, limited decision-making authority, and health systems valuing productivity and profit over patient care.
Although doctors and APPs have said that they generally work well together, the relationship has been strained in recent years as APPs argue for greater scope of practice. Meanwhile, physician groups, such as the American Medical Association, believe that APPs need more oversight.
An Allina union organizer, Britta V. Kasmarik, CNP, acknowledges the tension between physicians and APPs. But she said in an interview that the union effort helped bond this group of health care providers. “We share common goals of providing high-quality care for patients in a safe way, and we see the same things day in and day out with our patients.”
Matt Hoffman, MD, a primary care physician at Allina, told this news organization that APPs in his specialty perform the same job as doctors “and the working conditions are really identical. In our view, that means we should be unionizing together.”
The decision to hold a union vote follows similar action by nearly 150 Allina Mercy Hospital physicians in March. Allina Health appealed the vote.
In response to a New York Times investigation, the Minnesota Attorney General’s office began reviewing reports of aggressive billing practices and denied care at Allina Health.
The Allina Health system, which reports $4 billion in annual revenue, cut off nonemergency services to patients, including children, if their medical debt exceeded $4,500, according to the New York Times article. For Allina’s physicians and APPs, that meant leaving patients’ illnesses untreated.
Less than a week after the attorney general announced its investigation, the health system ended this practice.
In a prepared statement to this news organization, Allina Health said that its providers are “critical members of our teams. … We deeply value and share their commitment to providing high-quality care to our patients.”
The health system said it planned to make operational improvements, implement new communication tools, and provide additional well-being resources and enhanced employee benefits “to improve the provider experience.” In addition, it hoped to continue to “foster a culture of collaboration with all our employees.”
Having a union will allow health care providers to advocate for their patients and give health care providers more decision-making power instead of corporate leaders maintaining full authority, Ms. Kasmarik told this news organization.
Union organizers are also concerned with changes to the daily practice of medicine. “We don’t want to be spending our time doing paperwork and calling insurance companies and filling out forms,” said Dr. Hoffman. “We want to be in the exam room with a patient.”
The Allina providers organized after multiple requests to corporate managers failed to address their concerns. Their demands include increased staffing and help with nonclinical work so that clinicians can spend more time with their patients.
“What I’m really excited about is that we will be able to work with the other unionized groups to make change ... by being involved in health care policy at a state or national level,” Dr. Hoffman said. For example, that involvement might include challenging insurance company decisions.
Doctors Council bills itself as the largest union for attending physicians in the country, with 3,500 members, according to Joe Crane, national organizing director.
Despite an increase in union efforts since the pandemic, health care workers – particularly doctors – have been slow to join unions. Mr. Crane estimated that only about 3% of U.S. physicians are currently union members. He cited union campaigns in Massachusetts, New York, and Washington, DC. For comparison, a minority of advanced practice registered nurses (APRNs) (9%) report union membership, according to Medscape’s APRN compensation report last year.
Dr. Hoffman is confident the Allina health care providers will have enough votes to win the election to join the union. “We should have done this years ago.”
A version of this article appeared on Medscape.com.
Advanced practice providers (APPs) such as nurse practitioners (NPs) and physician assistants (PAs) have long been at odds with doctor groups over scope of practice issues. But in a rare alliance, in late September. If successful, the Allina group will join the Doctors Council SEIU, Local 10MD.
The Allina health care providers share concerns about their working conditions, such as understaffing and inadequate resources, limited decision-making authority, and health systems valuing productivity and profit over patient care.
Although doctors and APPs have said that they generally work well together, the relationship has been strained in recent years as APPs argue for greater scope of practice. Meanwhile, physician groups, such as the American Medical Association, believe that APPs need more oversight.
An Allina union organizer, Britta V. Kasmarik, CNP, acknowledges the tension between physicians and APPs. But she said in an interview that the union effort helped bond this group of health care providers. “We share common goals of providing high-quality care for patients in a safe way, and we see the same things day in and day out with our patients.”
Matt Hoffman, MD, a primary care physician at Allina, told this news organization that APPs in his specialty perform the same job as doctors “and the working conditions are really identical. In our view, that means we should be unionizing together.”
The decision to hold a union vote follows similar action by nearly 150 Allina Mercy Hospital physicians in March. Allina Health appealed the vote.
In response to a New York Times investigation, the Minnesota Attorney General’s office began reviewing reports of aggressive billing practices and denied care at Allina Health.
The Allina Health system, which reports $4 billion in annual revenue, cut off nonemergency services to patients, including children, if their medical debt exceeded $4,500, according to the New York Times article. For Allina’s physicians and APPs, that meant leaving patients’ illnesses untreated.
Less than a week after the attorney general announced its investigation, the health system ended this practice.
In a prepared statement to this news organization, Allina Health said that its providers are “critical members of our teams. … We deeply value and share their commitment to providing high-quality care to our patients.”
The health system said it planned to make operational improvements, implement new communication tools, and provide additional well-being resources and enhanced employee benefits “to improve the provider experience.” In addition, it hoped to continue to “foster a culture of collaboration with all our employees.”
Having a union will allow health care providers to advocate for their patients and give health care providers more decision-making power instead of corporate leaders maintaining full authority, Ms. Kasmarik told this news organization.
Union organizers are also concerned with changes to the daily practice of medicine. “We don’t want to be spending our time doing paperwork and calling insurance companies and filling out forms,” said Dr. Hoffman. “We want to be in the exam room with a patient.”
The Allina providers organized after multiple requests to corporate managers failed to address their concerns. Their demands include increased staffing and help with nonclinical work so that clinicians can spend more time with their patients.
“What I’m really excited about is that we will be able to work with the other unionized groups to make change ... by being involved in health care policy at a state or national level,” Dr. Hoffman said. For example, that involvement might include challenging insurance company decisions.
Doctors Council bills itself as the largest union for attending physicians in the country, with 3,500 members, according to Joe Crane, national organizing director.
Despite an increase in union efforts since the pandemic, health care workers – particularly doctors – have been slow to join unions. Mr. Crane estimated that only about 3% of U.S. physicians are currently union members. He cited union campaigns in Massachusetts, New York, and Washington, DC. For comparison, a minority of advanced practice registered nurses (APRNs) (9%) report union membership, according to Medscape’s APRN compensation report last year.
Dr. Hoffman is confident the Allina health care providers will have enough votes to win the election to join the union. “We should have done this years ago.”
A version of this article appeared on Medscape.com.
Advanced practice providers (APPs) such as nurse practitioners (NPs) and physician assistants (PAs) have long been at odds with doctor groups over scope of practice issues. But in a rare alliance, in late September. If successful, the Allina group will join the Doctors Council SEIU, Local 10MD.
The Allina health care providers share concerns about their working conditions, such as understaffing and inadequate resources, limited decision-making authority, and health systems valuing productivity and profit over patient care.
Although doctors and APPs have said that they generally work well together, the relationship has been strained in recent years as APPs argue for greater scope of practice. Meanwhile, physician groups, such as the American Medical Association, believe that APPs need more oversight.
An Allina union organizer, Britta V. Kasmarik, CNP, acknowledges the tension between physicians and APPs. But she said in an interview that the union effort helped bond this group of health care providers. “We share common goals of providing high-quality care for patients in a safe way, and we see the same things day in and day out with our patients.”
Matt Hoffman, MD, a primary care physician at Allina, told this news organization that APPs in his specialty perform the same job as doctors “and the working conditions are really identical. In our view, that means we should be unionizing together.”
The decision to hold a union vote follows similar action by nearly 150 Allina Mercy Hospital physicians in March. Allina Health appealed the vote.
In response to a New York Times investigation, the Minnesota Attorney General’s office began reviewing reports of aggressive billing practices and denied care at Allina Health.
The Allina Health system, which reports $4 billion in annual revenue, cut off nonemergency services to patients, including children, if their medical debt exceeded $4,500, according to the New York Times article. For Allina’s physicians and APPs, that meant leaving patients’ illnesses untreated.
Less than a week after the attorney general announced its investigation, the health system ended this practice.
In a prepared statement to this news organization, Allina Health said that its providers are “critical members of our teams. … We deeply value and share their commitment to providing high-quality care to our patients.”
The health system said it planned to make operational improvements, implement new communication tools, and provide additional well-being resources and enhanced employee benefits “to improve the provider experience.” In addition, it hoped to continue to “foster a culture of collaboration with all our employees.”
Having a union will allow health care providers to advocate for their patients and give health care providers more decision-making power instead of corporate leaders maintaining full authority, Ms. Kasmarik told this news organization.
Union organizers are also concerned with changes to the daily practice of medicine. “We don’t want to be spending our time doing paperwork and calling insurance companies and filling out forms,” said Dr. Hoffman. “We want to be in the exam room with a patient.”
The Allina providers organized after multiple requests to corporate managers failed to address their concerns. Their demands include increased staffing and help with nonclinical work so that clinicians can spend more time with their patients.
“What I’m really excited about is that we will be able to work with the other unionized groups to make change ... by being involved in health care policy at a state or national level,” Dr. Hoffman said. For example, that involvement might include challenging insurance company decisions.
Doctors Council bills itself as the largest union for attending physicians in the country, with 3,500 members, according to Joe Crane, national organizing director.
Despite an increase in union efforts since the pandemic, health care workers – particularly doctors – have been slow to join unions. Mr. Crane estimated that only about 3% of U.S. physicians are currently union members. He cited union campaigns in Massachusetts, New York, and Washington, DC. For comparison, a minority of advanced practice registered nurses (APRNs) (9%) report union membership, according to Medscape’s APRN compensation report last year.
Dr. Hoffman is confident the Allina health care providers will have enough votes to win the election to join the union. “We should have done this years ago.”
A version of this article appeared on Medscape.com.
Five questions for COVID experts: How concerned should we be?
COVID-19 hospitalizations have been on the rise for weeks as summer nears its end, but how concerned should you be? SARS-CoV-2, the virus behind COVID, continues to evolve and surprise us. So COVID transmission, hospitalization, and death rates can be difficult to predict.
, especially now that testing and vaccinations are no longer free of charge.
Question 1: Are you expecting an end-of-summer COVID wave to be substantial?
Eric Topol, MD: “This wave won’t likely be substantial and could be more of a ‘wavelet.’ I’m not thinking that physicians are too concerned,” said Dr. Topol, founder and director of Scripps Research Translational Institute in La Jolla, Calif.
Thomas Gut, DO: “It’s always impossible to predict the severity of COVID waves. Although the virus has generally mutated in ways that favor easier transmission and milder illness, there have been a handful of surprising mutations that were more dangerous and deadly than the preceding strain,” said Dr. Gut, associate chair of medicine at Staten Island University Hospital/Northwell Health in New York.
Robert Atmar, MD: “I’ll start with the caveat that prognosticating for SARS-CoV-2 is a bit hazardous as we remain in unknown territory for some aspects of its epidemiology and evolution,” said Dr. Atmar, a professor of infectious diseases at Baylor College of Medicine in Houston. “It depends on your definition of substantial. We, at least in Houston, are already in the midst of a substantial surge in the burden of infection, at least as monitored through wastewater surveillance. The amount of virus in the wastewater already exceeds the peak level we saw last winter. That said, the increased infection burden has not translated into large increases in hospitalizations for COVID-19. Most persons hospitalized in our hospital are admitted with infection, not for the consequences of infection.”
Stuart Campbell Ray, MD: “It looks like there is a rise in infections, but the proportional rise in hospitalizations from severe cases is lower than in the past, suggesting that folks are protected by the immunity we’ve gained over the past few years through vaccination and prior infections. Of course, we should be thinking about how that applies to each of us – how recently we had a vaccine or COVID-19, and whether we might see more severe infections as immunity wanes,” said Dr. Ray, who is a professor of medicine in the division of infectious diseases at Johns Hopkins University in Baltimore.
Question 2: Is a return to masks or mask mandates coming this fall or winter?
Dr. Topol: “Mandating masks doesn’t work very well, but we may see wide use again if a descendant of [variant] BA.2.86 takes off.”
Dr. Gut: “It’s difficult to predict if there are any mask mandates returning at any point. Ever since the Omicron strains emerged, COVID has been relatively mild, compared to previous strains, so there probably won’t be any plan to start masking in public unless a more deadly strain appears.”
Dr. Atmar: “I do not think we will see a return to mask mandates this fall or winter for a variety of reasons. The primary one is that I don’t think the public will accept mask mandates. However, I think masking can continue to be an adjunctive measure to enhance protection from infection, along with booster vaccination.”
Dr. Ray: “Some people will choose to wear masks during a surge, particularly in situations like commuting where they don’t interfere with what they’re doing. They will wear masks particularly if they want to avoid infection due to concerns about others they care about, disruption of work or travel plans, or concerns about long-term consequences of repeated COVID-19.”
Question 3: Now that COVID testing and vaccinations are no longer free of charge, how might that affect their use?
Dr. Topol: “It was already low, and this will undoubtedly further compromise their uptake.”
Dr. Gut: “I do expect that testing will become less common now that tests are no longer free. I’m sure there will be a lower amount of detection in patients with milder or asymptomatic disease compared to what we had previously.”
Dr. Atmar: “If there are out-of-pocket costs for the SARS-CoV-2 vaccine, or if the administrative paperwork attached to getting a vaccine is increased, the uptake of SARS-CoV-2 vaccines will likely decrease. It will be important to communicate to the populations targeted for vaccination the potential benefits of such vaccination.”
Dr. Ray: “A challenge with COVID-19, all along, has been disparities in access to care, and this will be worse without public support for prevention and testing. This applies to everyone but is especially burdensome for those who are often marginalized in our health care system and society in general. I hope that we’ll find ways to ensure that people who need tests and vaccinations are able to access them, as good health is in everyone’s interest.”
Question 4: Will the new vaccines against COVID work for the currently circulating variants?
Dr. Topol: “The XBB.1.5 boosters will be out Sept. 14. They should help versus EG.5.1 and FL.1.5.1. The FL.1.5.1 variant is gaining now.”
Dr. Gut: “In the next several weeks, we expect the newer monovalent XBB-based vaccines to be offered that offer good protection against current circulating COVID variants along with the new Eris variant.”
Dr. Atmar: “The vaccines are expected to induce immune responses to the currently circulating variants, most of which are strains that evolved from the vaccine strain. The vaccine is expected to be most effective in preventing severe illness and will likely be less effective in preventing infection and mild illness.”
Dr. Ray: “Yes, the updated vaccine design has a spike antigen (XBB.1.5) nearly identical to the current dominant variant (EG.5). Even as variants change, the boosters stimulate B cells and T cells to help protect in a way that is safer than getting COVID-19 infection.”
Question 5: Is there anything we should watch out for regarding the BA.2.86 variant in particular?
Dr. Topol: “The scenario could change if there are new functional mutations added to it.”
Dr. Gut: “BA.2.86 is still fairly uncommon and does not have much data to directly make any informed guesses. However, in general, people that have been exposed to more recent mutations of the COVID virus have been shown to have more protection from newer upcoming mutations. It’s fair to guess that people that have not had recent infection from COVID, or have not had a recent booster, are at higher risk for being infected by any XBB- or BA.2-based strains.”
Dr. Atmar: BA.2.86 has been designated as a variant under monitoring. We will want to see whether it becomes more common and if there are any unexpected characteristics associated with infection by this variant.”
Dr. Ray: “It’s still rare, but it’s been seen in geographically dispersed places, so it’s got legs. The question is how effectively it will bypass some of the immunity we’ve gained. T cells are likely to remain protective, because they target so many parts of the virus that change more slowly, but antibodies from B cells to spike protein may have more trouble recognizing BA.2.86, whether those antibodies were made to a vaccine or a prior variant.”
A version of this article first appeared on WebMD.com.
COVID-19 hospitalizations have been on the rise for weeks as summer nears its end, but how concerned should you be? SARS-CoV-2, the virus behind COVID, continues to evolve and surprise us. So COVID transmission, hospitalization, and death rates can be difficult to predict.
, especially now that testing and vaccinations are no longer free of charge.
Question 1: Are you expecting an end-of-summer COVID wave to be substantial?
Eric Topol, MD: “This wave won’t likely be substantial and could be more of a ‘wavelet.’ I’m not thinking that physicians are too concerned,” said Dr. Topol, founder and director of Scripps Research Translational Institute in La Jolla, Calif.
Thomas Gut, DO: “It’s always impossible to predict the severity of COVID waves. Although the virus has generally mutated in ways that favor easier transmission and milder illness, there have been a handful of surprising mutations that were more dangerous and deadly than the preceding strain,” said Dr. Gut, associate chair of medicine at Staten Island University Hospital/Northwell Health in New York.
Robert Atmar, MD: “I’ll start with the caveat that prognosticating for SARS-CoV-2 is a bit hazardous as we remain in unknown territory for some aspects of its epidemiology and evolution,” said Dr. Atmar, a professor of infectious diseases at Baylor College of Medicine in Houston. “It depends on your definition of substantial. We, at least in Houston, are already in the midst of a substantial surge in the burden of infection, at least as monitored through wastewater surveillance. The amount of virus in the wastewater already exceeds the peak level we saw last winter. That said, the increased infection burden has not translated into large increases in hospitalizations for COVID-19. Most persons hospitalized in our hospital are admitted with infection, not for the consequences of infection.”
Stuart Campbell Ray, MD: “It looks like there is a rise in infections, but the proportional rise in hospitalizations from severe cases is lower than in the past, suggesting that folks are protected by the immunity we’ve gained over the past few years through vaccination and prior infections. Of course, we should be thinking about how that applies to each of us – how recently we had a vaccine or COVID-19, and whether we might see more severe infections as immunity wanes,” said Dr. Ray, who is a professor of medicine in the division of infectious diseases at Johns Hopkins University in Baltimore.
Question 2: Is a return to masks or mask mandates coming this fall or winter?
Dr. Topol: “Mandating masks doesn’t work very well, but we may see wide use again if a descendant of [variant] BA.2.86 takes off.”
Dr. Gut: “It’s difficult to predict if there are any mask mandates returning at any point. Ever since the Omicron strains emerged, COVID has been relatively mild, compared to previous strains, so there probably won’t be any plan to start masking in public unless a more deadly strain appears.”
Dr. Atmar: “I do not think we will see a return to mask mandates this fall or winter for a variety of reasons. The primary one is that I don’t think the public will accept mask mandates. However, I think masking can continue to be an adjunctive measure to enhance protection from infection, along with booster vaccination.”
Dr. Ray: “Some people will choose to wear masks during a surge, particularly in situations like commuting where they don’t interfere with what they’re doing. They will wear masks particularly if they want to avoid infection due to concerns about others they care about, disruption of work or travel plans, or concerns about long-term consequences of repeated COVID-19.”
Question 3: Now that COVID testing and vaccinations are no longer free of charge, how might that affect their use?
Dr. Topol: “It was already low, and this will undoubtedly further compromise their uptake.”
Dr. Gut: “I do expect that testing will become less common now that tests are no longer free. I’m sure there will be a lower amount of detection in patients with milder or asymptomatic disease compared to what we had previously.”
Dr. Atmar: “If there are out-of-pocket costs for the SARS-CoV-2 vaccine, or if the administrative paperwork attached to getting a vaccine is increased, the uptake of SARS-CoV-2 vaccines will likely decrease. It will be important to communicate to the populations targeted for vaccination the potential benefits of such vaccination.”
Dr. Ray: “A challenge with COVID-19, all along, has been disparities in access to care, and this will be worse without public support for prevention and testing. This applies to everyone but is especially burdensome for those who are often marginalized in our health care system and society in general. I hope that we’ll find ways to ensure that people who need tests and vaccinations are able to access them, as good health is in everyone’s interest.”
Question 4: Will the new vaccines against COVID work for the currently circulating variants?
Dr. Topol: “The XBB.1.5 boosters will be out Sept. 14. They should help versus EG.5.1 and FL.1.5.1. The FL.1.5.1 variant is gaining now.”
Dr. Gut: “In the next several weeks, we expect the newer monovalent XBB-based vaccines to be offered that offer good protection against current circulating COVID variants along with the new Eris variant.”
Dr. Atmar: “The vaccines are expected to induce immune responses to the currently circulating variants, most of which are strains that evolved from the vaccine strain. The vaccine is expected to be most effective in preventing severe illness and will likely be less effective in preventing infection and mild illness.”
Dr. Ray: “Yes, the updated vaccine design has a spike antigen (XBB.1.5) nearly identical to the current dominant variant (EG.5). Even as variants change, the boosters stimulate B cells and T cells to help protect in a way that is safer than getting COVID-19 infection.”
Question 5: Is there anything we should watch out for regarding the BA.2.86 variant in particular?
Dr. Topol: “The scenario could change if there are new functional mutations added to it.”
Dr. Gut: “BA.2.86 is still fairly uncommon and does not have much data to directly make any informed guesses. However, in general, people that have been exposed to more recent mutations of the COVID virus have been shown to have more protection from newer upcoming mutations. It’s fair to guess that people that have not had recent infection from COVID, or have not had a recent booster, are at higher risk for being infected by any XBB- or BA.2-based strains.”
Dr. Atmar: BA.2.86 has been designated as a variant under monitoring. We will want to see whether it becomes more common and if there are any unexpected characteristics associated with infection by this variant.”
Dr. Ray: “It’s still rare, but it’s been seen in geographically dispersed places, so it’s got legs. The question is how effectively it will bypass some of the immunity we’ve gained. T cells are likely to remain protective, because they target so many parts of the virus that change more slowly, but antibodies from B cells to spike protein may have more trouble recognizing BA.2.86, whether those antibodies were made to a vaccine or a prior variant.”
A version of this article first appeared on WebMD.com.
COVID-19 hospitalizations have been on the rise for weeks as summer nears its end, but how concerned should you be? SARS-CoV-2, the virus behind COVID, continues to evolve and surprise us. So COVID transmission, hospitalization, and death rates can be difficult to predict.
, especially now that testing and vaccinations are no longer free of charge.
Question 1: Are you expecting an end-of-summer COVID wave to be substantial?
Eric Topol, MD: “This wave won’t likely be substantial and could be more of a ‘wavelet.’ I’m not thinking that physicians are too concerned,” said Dr. Topol, founder and director of Scripps Research Translational Institute in La Jolla, Calif.
Thomas Gut, DO: “It’s always impossible to predict the severity of COVID waves. Although the virus has generally mutated in ways that favor easier transmission and milder illness, there have been a handful of surprising mutations that were more dangerous and deadly than the preceding strain,” said Dr. Gut, associate chair of medicine at Staten Island University Hospital/Northwell Health in New York.
Robert Atmar, MD: “I’ll start with the caveat that prognosticating for SARS-CoV-2 is a bit hazardous as we remain in unknown territory for some aspects of its epidemiology and evolution,” said Dr. Atmar, a professor of infectious diseases at Baylor College of Medicine in Houston. “It depends on your definition of substantial. We, at least in Houston, are already in the midst of a substantial surge in the burden of infection, at least as monitored through wastewater surveillance. The amount of virus in the wastewater already exceeds the peak level we saw last winter. That said, the increased infection burden has not translated into large increases in hospitalizations for COVID-19. Most persons hospitalized in our hospital are admitted with infection, not for the consequences of infection.”
Stuart Campbell Ray, MD: “It looks like there is a rise in infections, but the proportional rise in hospitalizations from severe cases is lower than in the past, suggesting that folks are protected by the immunity we’ve gained over the past few years through vaccination and prior infections. Of course, we should be thinking about how that applies to each of us – how recently we had a vaccine or COVID-19, and whether we might see more severe infections as immunity wanes,” said Dr. Ray, who is a professor of medicine in the division of infectious diseases at Johns Hopkins University in Baltimore.
Question 2: Is a return to masks or mask mandates coming this fall or winter?
Dr. Topol: “Mandating masks doesn’t work very well, but we may see wide use again if a descendant of [variant] BA.2.86 takes off.”
Dr. Gut: “It’s difficult to predict if there are any mask mandates returning at any point. Ever since the Omicron strains emerged, COVID has been relatively mild, compared to previous strains, so there probably won’t be any plan to start masking in public unless a more deadly strain appears.”
Dr. Atmar: “I do not think we will see a return to mask mandates this fall or winter for a variety of reasons. The primary one is that I don’t think the public will accept mask mandates. However, I think masking can continue to be an adjunctive measure to enhance protection from infection, along with booster vaccination.”
Dr. Ray: “Some people will choose to wear masks during a surge, particularly in situations like commuting where they don’t interfere with what they’re doing. They will wear masks particularly if they want to avoid infection due to concerns about others they care about, disruption of work or travel plans, or concerns about long-term consequences of repeated COVID-19.”
Question 3: Now that COVID testing and vaccinations are no longer free of charge, how might that affect their use?
Dr. Topol: “It was already low, and this will undoubtedly further compromise their uptake.”
Dr. Gut: “I do expect that testing will become less common now that tests are no longer free. I’m sure there will be a lower amount of detection in patients with milder or asymptomatic disease compared to what we had previously.”
Dr. Atmar: “If there are out-of-pocket costs for the SARS-CoV-2 vaccine, or if the administrative paperwork attached to getting a vaccine is increased, the uptake of SARS-CoV-2 vaccines will likely decrease. It will be important to communicate to the populations targeted for vaccination the potential benefits of such vaccination.”
Dr. Ray: “A challenge with COVID-19, all along, has been disparities in access to care, and this will be worse without public support for prevention and testing. This applies to everyone but is especially burdensome for those who are often marginalized in our health care system and society in general. I hope that we’ll find ways to ensure that people who need tests and vaccinations are able to access them, as good health is in everyone’s interest.”
Question 4: Will the new vaccines against COVID work for the currently circulating variants?
Dr. Topol: “The XBB.1.5 boosters will be out Sept. 14. They should help versus EG.5.1 and FL.1.5.1. The FL.1.5.1 variant is gaining now.”
Dr. Gut: “In the next several weeks, we expect the newer monovalent XBB-based vaccines to be offered that offer good protection against current circulating COVID variants along with the new Eris variant.”
Dr. Atmar: “The vaccines are expected to induce immune responses to the currently circulating variants, most of which are strains that evolved from the vaccine strain. The vaccine is expected to be most effective in preventing severe illness and will likely be less effective in preventing infection and mild illness.”
Dr. Ray: “Yes, the updated vaccine design has a spike antigen (XBB.1.5) nearly identical to the current dominant variant (EG.5). Even as variants change, the boosters stimulate B cells and T cells to help protect in a way that is safer than getting COVID-19 infection.”
Question 5: Is there anything we should watch out for regarding the BA.2.86 variant in particular?
Dr. Topol: “The scenario could change if there are new functional mutations added to it.”
Dr. Gut: “BA.2.86 is still fairly uncommon and does not have much data to directly make any informed guesses. However, in general, people that have been exposed to more recent mutations of the COVID virus have been shown to have more protection from newer upcoming mutations. It’s fair to guess that people that have not had recent infection from COVID, or have not had a recent booster, are at higher risk for being infected by any XBB- or BA.2-based strains.”
Dr. Atmar: BA.2.86 has been designated as a variant under monitoring. We will want to see whether it becomes more common and if there are any unexpected characteristics associated with infection by this variant.”
Dr. Ray: “It’s still rare, but it’s been seen in geographically dispersed places, so it’s got legs. The question is how effectively it will bypass some of the immunity we’ve gained. T cells are likely to remain protective, because they target so many parts of the virus that change more slowly, but antibodies from B cells to spike protein may have more trouble recognizing BA.2.86, whether those antibodies were made to a vaccine or a prior variant.”
A version of this article first appeared on WebMD.com.

