User login
HM19: Interprofessional rounds
Better process, outcomes
Session presenters
Surekha Bhamidipati, MD, FACP; Preetham Talari, MD, FACP, SFHM; Mark V. Williams, MD, FACP, MHM
Session title
Interprofessional rounds: What’s the right way?
Session summary
Interprofessional or multidisciplinary rounds involve all members of the care delivery team, including physicians, nurses, case managers, social workers, pharmacists, nurse facilitators, and of course, patients. The primary goal for these rounds is patient-centered care, and to improve communication among the health care team members, as well as with patients and their families.
At HM19, Dr. Preetham Talari and Dr. Mark Williams of the University of Kentucky, and Dr. Surekha Bhamidipati of Christiana Care Health System in Newark, Del., discussed their system-based efforts to try to implement interprofessional rounds, and the role of these rounds in improving patient outcome measures.
The presenters noted that the purpose of these rounds is effective communication and efficient patient care. As shown by multiple studies, there is significant impact in team member satisfaction, decrease in length of stay,1 reduction in adverse events2 and improvement in patient experience.3 They emphasized the importance of implementing these rounds at the bedside, so that patients and families can be engaged in the patient’s care, thereby improving closed communication among the team and the patient. These rounds always offer an opportunity for the patient to ask questions of multiple health care team members as they are gathered together at the same time.
The University of Kentucky named these rounds the “Interprofessional Teamwork Innovation Model (ITIM),” to promote communication and patient-centered coordinated care. Their model showed a significant reduction in readmission rates, and no increase in costs despite adding pharmacy and case managers to the rounds.
Dr. Bhamidipati described how Christiana Care Health System designed multidisciplinary rounds based on the application of Team STEPPS 2.0, a teamwork system developed by the Department of Defense and the Agency for Healthcare Research and Quality to improve the institutional collaboration and communication relating to patient safety.
The Christiana Care model is based on a few principles of team structure, communication, leadership, situation monitoring, and mutual support. The interprofessional team was trained and observed, and a short video recording was made. This video was used as an educational tool in coaching the rest of the team. Dr. Bhamidipati described the importance of interprofessional leaders as coaches to train other team members, and highlighted the engagement of unit leaders in successfully implementing these rounds. The Christiana Care team used its informational technology system to collect real-time data, which was then used for team review.
In summary, the presenters from both the University of Kentucky and Christiana Care highlighted the importance of interprofessional rounds, as well as the need for continued measurement of process and outcome metrics.
Dr. Jonnalagadda is a physician advisor, and Dr. Medarametla is medical director, Hospital Medicine, at Baystate Medical Center, Springfield, Mass.
References
1. Yoo JW et al. Effects of an internal medicine floor interdisciplinary team on hospital and clinical outcomes of seniors with acute medical illness. Geriatr Gerontol Int. 2013 Oct;13(4):942-8. doi: 10.1111/ggi.12035. Epub 2013 Feb 26.
2. O’Leary KJ et al. Structured interdisciplinary rounds in a medical teaching unit: improving patient safety. Arch Intern Med. 2011 Apr 11;171(7):678-84. doi: 10.1001/archinternmed.2011.128.
3. Ratelle JT et al. Implementing bedside rounds to improve patient-centred outcomes: a systematic review. BMJ Qual Saf. 2019;28:317-326.
Better process, outcomes
Better process, outcomes
Session presenters
Surekha Bhamidipati, MD, FACP; Preetham Talari, MD, FACP, SFHM; Mark V. Williams, MD, FACP, MHM
Session title
Interprofessional rounds: What’s the right way?
Session summary
Interprofessional or multidisciplinary rounds involve all members of the care delivery team, including physicians, nurses, case managers, social workers, pharmacists, nurse facilitators, and of course, patients. The primary goal for these rounds is patient-centered care, and to improve communication among the health care team members, as well as with patients and their families.
At HM19, Dr. Preetham Talari and Dr. Mark Williams of the University of Kentucky, and Dr. Surekha Bhamidipati of Christiana Care Health System in Newark, Del., discussed their system-based efforts to try to implement interprofessional rounds, and the role of these rounds in improving patient outcome measures.
The presenters noted that the purpose of these rounds is effective communication and efficient patient care. As shown by multiple studies, there is significant impact in team member satisfaction, decrease in length of stay,1 reduction in adverse events2 and improvement in patient experience.3 They emphasized the importance of implementing these rounds at the bedside, so that patients and families can be engaged in the patient’s care, thereby improving closed communication among the team and the patient. These rounds always offer an opportunity for the patient to ask questions of multiple health care team members as they are gathered together at the same time.
The University of Kentucky named these rounds the “Interprofessional Teamwork Innovation Model (ITIM),” to promote communication and patient-centered coordinated care. Their model showed a significant reduction in readmission rates, and no increase in costs despite adding pharmacy and case managers to the rounds.
Dr. Bhamidipati described how Christiana Care Health System designed multidisciplinary rounds based on the application of Team STEPPS 2.0, a teamwork system developed by the Department of Defense and the Agency for Healthcare Research and Quality to improve the institutional collaboration and communication relating to patient safety.
The Christiana Care model is based on a few principles of team structure, communication, leadership, situation monitoring, and mutual support. The interprofessional team was trained and observed, and a short video recording was made. This video was used as an educational tool in coaching the rest of the team. Dr. Bhamidipati described the importance of interprofessional leaders as coaches to train other team members, and highlighted the engagement of unit leaders in successfully implementing these rounds. The Christiana Care team used its informational technology system to collect real-time data, which was then used for team review.
In summary, the presenters from both the University of Kentucky and Christiana Care highlighted the importance of interprofessional rounds, as well as the need for continued measurement of process and outcome metrics.
Dr. Jonnalagadda is a physician advisor, and Dr. Medarametla is medical director, Hospital Medicine, at Baystate Medical Center, Springfield, Mass.
References
1. Yoo JW et al. Effects of an internal medicine floor interdisciplinary team on hospital and clinical outcomes of seniors with acute medical illness. Geriatr Gerontol Int. 2013 Oct;13(4):942-8. doi: 10.1111/ggi.12035. Epub 2013 Feb 26.
2. O’Leary KJ et al. Structured interdisciplinary rounds in a medical teaching unit: improving patient safety. Arch Intern Med. 2011 Apr 11;171(7):678-84. doi: 10.1001/archinternmed.2011.128.
3. Ratelle JT et al. Implementing bedside rounds to improve patient-centred outcomes: a systematic review. BMJ Qual Saf. 2019;28:317-326.
Session presenters
Surekha Bhamidipati, MD, FACP; Preetham Talari, MD, FACP, SFHM; Mark V. Williams, MD, FACP, MHM
Session title
Interprofessional rounds: What’s the right way?
Session summary
Interprofessional or multidisciplinary rounds involve all members of the care delivery team, including physicians, nurses, case managers, social workers, pharmacists, nurse facilitators, and of course, patients. The primary goal for these rounds is patient-centered care, and to improve communication among the health care team members, as well as with patients and their families.
At HM19, Dr. Preetham Talari and Dr. Mark Williams of the University of Kentucky, and Dr. Surekha Bhamidipati of Christiana Care Health System in Newark, Del., discussed their system-based efforts to try to implement interprofessional rounds, and the role of these rounds in improving patient outcome measures.
The presenters noted that the purpose of these rounds is effective communication and efficient patient care. As shown by multiple studies, there is significant impact in team member satisfaction, decrease in length of stay,1 reduction in adverse events2 and improvement in patient experience.3 They emphasized the importance of implementing these rounds at the bedside, so that patients and families can be engaged in the patient’s care, thereby improving closed communication among the team and the patient. These rounds always offer an opportunity for the patient to ask questions of multiple health care team members as they are gathered together at the same time.
The University of Kentucky named these rounds the “Interprofessional Teamwork Innovation Model (ITIM),” to promote communication and patient-centered coordinated care. Their model showed a significant reduction in readmission rates, and no increase in costs despite adding pharmacy and case managers to the rounds.
Dr. Bhamidipati described how Christiana Care Health System designed multidisciplinary rounds based on the application of Team STEPPS 2.0, a teamwork system developed by the Department of Defense and the Agency for Healthcare Research and Quality to improve the institutional collaboration and communication relating to patient safety.
The Christiana Care model is based on a few principles of team structure, communication, leadership, situation monitoring, and mutual support. The interprofessional team was trained and observed, and a short video recording was made. This video was used as an educational tool in coaching the rest of the team. Dr. Bhamidipati described the importance of interprofessional leaders as coaches to train other team members, and highlighted the engagement of unit leaders in successfully implementing these rounds. The Christiana Care team used its informational technology system to collect real-time data, which was then used for team review.
In summary, the presenters from both the University of Kentucky and Christiana Care highlighted the importance of interprofessional rounds, as well as the need for continued measurement of process and outcome metrics.
Dr. Jonnalagadda is a physician advisor, and Dr. Medarametla is medical director, Hospital Medicine, at Baystate Medical Center, Springfield, Mass.
References
1. Yoo JW et al. Effects of an internal medicine floor interdisciplinary team on hospital and clinical outcomes of seniors with acute medical illness. Geriatr Gerontol Int. 2013 Oct;13(4):942-8. doi: 10.1111/ggi.12035. Epub 2013 Feb 26.
2. O’Leary KJ et al. Structured interdisciplinary rounds in a medical teaching unit: improving patient safety. Arch Intern Med. 2011 Apr 11;171(7):678-84. doi: 10.1001/archinternmed.2011.128.
3. Ratelle JT et al. Implementing bedside rounds to improve patient-centred outcomes: a systematic review. BMJ Qual Saf. 2019;28:317-326.
Living into your legacy
What I learned from women of impact
The word legacy has been synonymous with death to me. When so and so dies, we discuss their legacy. I had a powerful experience that changed my mind on this word that is befitting for this Legacies column.
Seven years ago, I was sitting in a room of powerful women and I was the youngest one there. I wasn’t sure how I got there, but I was glad I did because it changed my life. At the time, I was panicked. The exercise was called “Craft your legacy statement.”
But, this exercise was different. The ask was to “live into your legacy.” Craft a legacy statement in THREE minutes that summarizes what you want your legacy to be … and then decide the three things you need to do now to get there. So, here is my exact legacy 3-minute statement: I am an innovator pushing teaching hospitals to optimize training and patient care delivery through novel technologies and systems science. Clearly, I did not aim high enough. One of the other attendees stated her legacy simply as “Unleash the impossible!” So clearly, I was not able to think big at that moment, but I trudged on.
Next, I had to write the three things I was going to do to enact my legacy today. Things went from bad to worse quickly since I knew this was not going to be easy. The #1 thing had to be something I was going to stop doing because it did not fit with my legacy; #2 was what I was going to start doing to enact this legacy now; and, #3 was something I was going to do to get me closer to what I wanted to be doing. So, my #1, resign my current leadership role that I had had for 8 years; #2, start joining national committees that bridge education and quality; and #3, meet with senior leadership to pitch this new role as a bridging leader, aligning education and quality.
Like all conferences, I went home and forgot what I had done and learned. I settled back into my old life and routines. A few weeks later, a plain looking envelope with awful penmanship showed up at my doorstep addressed to me. It wasn’t until after I opened it and read what was inside that I realized I was the one with horrible penmanship! I completely forgot that I wrote this letter to myself even though they told me it would come and I would forget I wrote it! So, how did I do? Let’s just say if the letter did not arrive, I am not sure where I would be. Fortunately, it did come, and I followed my own orders. Fast forward to present day and I recently stepped into a new role – associate chief medical officer: clinical learning environment – a bridging leader who aligns education and clinical care missions for our health system. Let’s just say again, had that letter not arrived, I am not sure where I would be now.
I have been fortunate to do many things in hospital medicine – clinician, researcher, educator, and my latest role as a leader. Through it all, I would say that there are some lessons that I have picked up along the way that helped me advance, in ways I did not realize:
- Be bold. Years ago, when I was asked by my chair who they should pick to be chief resident, I thought “This must be a trick question – I should definitely tell him why I should be chosen – and then pick the next best person who I want to work with.” Apparently, I was the only person who did that, and that is why my chair chose me. Everyone else picked two other people. So the take-home point here is do not sell yourself short … ever.
- Look for the hidden gateways. A few years ago, I was asked if I wanted to be an institutional leader by the person who currently had that role. I was kind of thrown for a loop, since of course I would not want to appear like I wanted to take his job. I said everything was fine and I felt pretty good about my current positions. It was only a few weeks later that I realized that he was ascertaining my interest in his job since he was leaving. They gave the job to someone else and the word on the street was I was not interested. I totally missed the gate! While it wasn’t necessarily the job I missed out on, it was the opportunity to consider the job because I was afraid. So, don’t miss the gate. It’s the wormhole to a different life that may be the right one for you, but you need to “see it” to seize it.
- Work hard for the money and for the fun. There are many things Gwyneth Paltrow does that I do not agree with, but I will give her credit for one important lesson: she divides her movie roles into those she does for love (for example, The Royal Tenenbaums) and those she does for money (for example, Shallow Hal). It made me realize that even a Hollywood starlet has to do the stuff she may not want to do for the money. So, as a young person, you have to work hard for the money, but ideally it will help you take on a project you love, whatever it is. You’ve won the game when you’re mostly paid to work for the fun ... but that may take some time.
- Always optimize what is best for you personally AND professionally. While I was on maternity leave, the job of my dreams presented itself – or so I thought it did. It was at the intersection of policy, quality, and education, with a national stage, and I would not need to move. But, I knew I could not accept the travel commitment with a young child. While I wondered if I would have regrets, it turns out the right decision professionally also has to work personally. Likewise, there are professional obligations that I take on because it works personally.
- Figure out who your tea house pals are. A few years ago, I was in San Francisco with two close friends having an epic moment about what to do with our lives as adults. We were all on the cusp of changing our directions. Not surprisingly, we could see what the other needed to do, but we could not see it for ourselves. We still text each other sometimes about the need to go back to the Tea House. Sometimes your “tea house pals” are not necessarily those around you every day. They know you, but not everyone in your work place. This “arm’s length” or distance gives them the rational, unbiased perspective to advise you, that you or your colleagues will never have.
- Look for ways to enjoy the journey. Medicine is a very long road. I routinely think about this working with all the trainees and junior faculty I encounter. You can’t be in this solely for the end of the journey. The key is to find the joy in the journey. For me, that has always come from seeking out like-minded fellow travelers to share my highs and lows. While I tweet for many reasons, a big reason is that I take pleasure in watching others on the journey and also sharing my own journey.
Here’s to your journey and living your legacy!
Dr. Arora is associate chief medical officer, clinical learning environment, at University of Chicago Medicine, and assistant dean for scholarship and discovery at the University of Chicago Pritzker School of Medicine. You can follow her journey on Twitter.
What I learned from women of impact
What I learned from women of impact
The word legacy has been synonymous with death to me. When so and so dies, we discuss their legacy. I had a powerful experience that changed my mind on this word that is befitting for this Legacies column.
Seven years ago, I was sitting in a room of powerful women and I was the youngest one there. I wasn’t sure how I got there, but I was glad I did because it changed my life. At the time, I was panicked. The exercise was called “Craft your legacy statement.”
But, this exercise was different. The ask was to “live into your legacy.” Craft a legacy statement in THREE minutes that summarizes what you want your legacy to be … and then decide the three things you need to do now to get there. So, here is my exact legacy 3-minute statement: I am an innovator pushing teaching hospitals to optimize training and patient care delivery through novel technologies and systems science. Clearly, I did not aim high enough. One of the other attendees stated her legacy simply as “Unleash the impossible!” So clearly, I was not able to think big at that moment, but I trudged on.
Next, I had to write the three things I was going to do to enact my legacy today. Things went from bad to worse quickly since I knew this was not going to be easy. The #1 thing had to be something I was going to stop doing because it did not fit with my legacy; #2 was what I was going to start doing to enact this legacy now; and, #3 was something I was going to do to get me closer to what I wanted to be doing. So, my #1, resign my current leadership role that I had had for 8 years; #2, start joining national committees that bridge education and quality; and #3, meet with senior leadership to pitch this new role as a bridging leader, aligning education and quality.
Like all conferences, I went home and forgot what I had done and learned. I settled back into my old life and routines. A few weeks later, a plain looking envelope with awful penmanship showed up at my doorstep addressed to me. It wasn’t until after I opened it and read what was inside that I realized I was the one with horrible penmanship! I completely forgot that I wrote this letter to myself even though they told me it would come and I would forget I wrote it! So, how did I do? Let’s just say if the letter did not arrive, I am not sure where I would be. Fortunately, it did come, and I followed my own orders. Fast forward to present day and I recently stepped into a new role – associate chief medical officer: clinical learning environment – a bridging leader who aligns education and clinical care missions for our health system. Let’s just say again, had that letter not arrived, I am not sure where I would be now.
I have been fortunate to do many things in hospital medicine – clinician, researcher, educator, and my latest role as a leader. Through it all, I would say that there are some lessons that I have picked up along the way that helped me advance, in ways I did not realize:
- Be bold. Years ago, when I was asked by my chair who they should pick to be chief resident, I thought “This must be a trick question – I should definitely tell him why I should be chosen – and then pick the next best person who I want to work with.” Apparently, I was the only person who did that, and that is why my chair chose me. Everyone else picked two other people. So the take-home point here is do not sell yourself short … ever.
- Look for the hidden gateways. A few years ago, I was asked if I wanted to be an institutional leader by the person who currently had that role. I was kind of thrown for a loop, since of course I would not want to appear like I wanted to take his job. I said everything was fine and I felt pretty good about my current positions. It was only a few weeks later that I realized that he was ascertaining my interest in his job since he was leaving. They gave the job to someone else and the word on the street was I was not interested. I totally missed the gate! While it wasn’t necessarily the job I missed out on, it was the opportunity to consider the job because I was afraid. So, don’t miss the gate. It’s the wormhole to a different life that may be the right one for you, but you need to “see it” to seize it.
- Work hard for the money and for the fun. There are many things Gwyneth Paltrow does that I do not agree with, but I will give her credit for one important lesson: she divides her movie roles into those she does for love (for example, The Royal Tenenbaums) and those she does for money (for example, Shallow Hal). It made me realize that even a Hollywood starlet has to do the stuff she may not want to do for the money. So, as a young person, you have to work hard for the money, but ideally it will help you take on a project you love, whatever it is. You’ve won the game when you’re mostly paid to work for the fun ... but that may take some time.
- Always optimize what is best for you personally AND professionally. While I was on maternity leave, the job of my dreams presented itself – or so I thought it did. It was at the intersection of policy, quality, and education, with a national stage, and I would not need to move. But, I knew I could not accept the travel commitment with a young child. While I wondered if I would have regrets, it turns out the right decision professionally also has to work personally. Likewise, there are professional obligations that I take on because it works personally.
- Figure out who your tea house pals are. A few years ago, I was in San Francisco with two close friends having an epic moment about what to do with our lives as adults. We were all on the cusp of changing our directions. Not surprisingly, we could see what the other needed to do, but we could not see it for ourselves. We still text each other sometimes about the need to go back to the Tea House. Sometimes your “tea house pals” are not necessarily those around you every day. They know you, but not everyone in your work place. This “arm’s length” or distance gives them the rational, unbiased perspective to advise you, that you or your colleagues will never have.
- Look for ways to enjoy the journey. Medicine is a very long road. I routinely think about this working with all the trainees and junior faculty I encounter. You can’t be in this solely for the end of the journey. The key is to find the joy in the journey. For me, that has always come from seeking out like-minded fellow travelers to share my highs and lows. While I tweet for many reasons, a big reason is that I take pleasure in watching others on the journey and also sharing my own journey.
Here’s to your journey and living your legacy!
Dr. Arora is associate chief medical officer, clinical learning environment, at University of Chicago Medicine, and assistant dean for scholarship and discovery at the University of Chicago Pritzker School of Medicine. You can follow her journey on Twitter.
The word legacy has been synonymous with death to me. When so and so dies, we discuss their legacy. I had a powerful experience that changed my mind on this word that is befitting for this Legacies column.
Seven years ago, I was sitting in a room of powerful women and I was the youngest one there. I wasn’t sure how I got there, but I was glad I did because it changed my life. At the time, I was panicked. The exercise was called “Craft your legacy statement.”
But, this exercise was different. The ask was to “live into your legacy.” Craft a legacy statement in THREE minutes that summarizes what you want your legacy to be … and then decide the three things you need to do now to get there. So, here is my exact legacy 3-minute statement: I am an innovator pushing teaching hospitals to optimize training and patient care delivery through novel technologies and systems science. Clearly, I did not aim high enough. One of the other attendees stated her legacy simply as “Unleash the impossible!” So clearly, I was not able to think big at that moment, but I trudged on.
Next, I had to write the three things I was going to do to enact my legacy today. Things went from bad to worse quickly since I knew this was not going to be easy. The #1 thing had to be something I was going to stop doing because it did not fit with my legacy; #2 was what I was going to start doing to enact this legacy now; and, #3 was something I was going to do to get me closer to what I wanted to be doing. So, my #1, resign my current leadership role that I had had for 8 years; #2, start joining national committees that bridge education and quality; and #3, meet with senior leadership to pitch this new role as a bridging leader, aligning education and quality.
Like all conferences, I went home and forgot what I had done and learned. I settled back into my old life and routines. A few weeks later, a plain looking envelope with awful penmanship showed up at my doorstep addressed to me. It wasn’t until after I opened it and read what was inside that I realized I was the one with horrible penmanship! I completely forgot that I wrote this letter to myself even though they told me it would come and I would forget I wrote it! So, how did I do? Let’s just say if the letter did not arrive, I am not sure where I would be. Fortunately, it did come, and I followed my own orders. Fast forward to present day and I recently stepped into a new role – associate chief medical officer: clinical learning environment – a bridging leader who aligns education and clinical care missions for our health system. Let’s just say again, had that letter not arrived, I am not sure where I would be now.
I have been fortunate to do many things in hospital medicine – clinician, researcher, educator, and my latest role as a leader. Through it all, I would say that there are some lessons that I have picked up along the way that helped me advance, in ways I did not realize:
- Be bold. Years ago, when I was asked by my chair who they should pick to be chief resident, I thought “This must be a trick question – I should definitely tell him why I should be chosen – and then pick the next best person who I want to work with.” Apparently, I was the only person who did that, and that is why my chair chose me. Everyone else picked two other people. So the take-home point here is do not sell yourself short … ever.
- Look for the hidden gateways. A few years ago, I was asked if I wanted to be an institutional leader by the person who currently had that role. I was kind of thrown for a loop, since of course I would not want to appear like I wanted to take his job. I said everything was fine and I felt pretty good about my current positions. It was only a few weeks later that I realized that he was ascertaining my interest in his job since he was leaving. They gave the job to someone else and the word on the street was I was not interested. I totally missed the gate! While it wasn’t necessarily the job I missed out on, it was the opportunity to consider the job because I was afraid. So, don’t miss the gate. It’s the wormhole to a different life that may be the right one for you, but you need to “see it” to seize it.
- Work hard for the money and for the fun. There are many things Gwyneth Paltrow does that I do not agree with, but I will give her credit for one important lesson: she divides her movie roles into those she does for love (for example, The Royal Tenenbaums) and those she does for money (for example, Shallow Hal). It made me realize that even a Hollywood starlet has to do the stuff she may not want to do for the money. So, as a young person, you have to work hard for the money, but ideally it will help you take on a project you love, whatever it is. You’ve won the game when you’re mostly paid to work for the fun ... but that may take some time.
- Always optimize what is best for you personally AND professionally. While I was on maternity leave, the job of my dreams presented itself – or so I thought it did. It was at the intersection of policy, quality, and education, with a national stage, and I would not need to move. But, I knew I could not accept the travel commitment with a young child. While I wondered if I would have regrets, it turns out the right decision professionally also has to work personally. Likewise, there are professional obligations that I take on because it works personally.
- Figure out who your tea house pals are. A few years ago, I was in San Francisco with two close friends having an epic moment about what to do with our lives as adults. We were all on the cusp of changing our directions. Not surprisingly, we could see what the other needed to do, but we could not see it for ourselves. We still text each other sometimes about the need to go back to the Tea House. Sometimes your “tea house pals” are not necessarily those around you every day. They know you, but not everyone in your work place. This “arm’s length” or distance gives them the rational, unbiased perspective to advise you, that you or your colleagues will never have.
- Look for ways to enjoy the journey. Medicine is a very long road. I routinely think about this working with all the trainees and junior faculty I encounter. You can’t be in this solely for the end of the journey. The key is to find the joy in the journey. For me, that has always come from seeking out like-minded fellow travelers to share my highs and lows. While I tweet for many reasons, a big reason is that I take pleasure in watching others on the journey and also sharing my own journey.
Here’s to your journey and living your legacy!
Dr. Arora is associate chief medical officer, clinical learning environment, at University of Chicago Medicine, and assistant dean for scholarship and discovery at the University of Chicago Pritzker School of Medicine. You can follow her journey on Twitter.
Severe OSA increases cardiovascular risk after surgery
Unrecognized severe obstructive sleep apnea is a risk factor for cardiovascular complications after major noncardiac surgery, according to a study published in JAMA.
The researchers state that perioperative mismanagement of obstructive sleep apnea can lead to serious medical consequences. “General anesthetics, sedatives, and postoperative analgesics are potent respiratory depressants that relax the upper airway dilator muscles and impair ventilatory response to hypoxemia and hypercapnia. Each of these events exacerbates [obstructive sleep apnea] and may predispose patients to postoperative cardiovascular complications,” said researchers who conducted the The Postoperative vascular complications in unrecognised Obstructive Sleep apnoea (POSA) study (NCT01494181).
They undertook a prospective observational cohort study involving 1,218 patients undergoing major noncardiac surgery, who were already considered at high risk of postoperative cardiovascular events – having, for example, a history of coronary artery disease, stroke, diabetes, or renal impairment. However, none had a prior diagnosis of obstructive sleep apnea.
Preoperative sleep monitoring revealed that two-thirds of the cohort had unrecognized and untreated obstructive sleep apnea, including 11.2% with severe obstructive sleep apnea.
At 30 days after surgery, patients with obstructive sleep apnea had a 49% higher risk of the primary outcome of myocardial injury, cardiac death, heart failure, thromboembolism, atrial fibrillation, or stroke, compared with those without obstructive sleep apnea.
However, this association was largely due to a significant 2.23-fold higher risk among patients with severe obstructive sleep apnea, while those with only moderate or mild sleep apnea did not show a significant increased risk of cardiovascular complications.
Patients in this study with severe obstructive sleep apnea had a 13-fold higher risk of cardiac death, 80% higher risk of myocardial injury, more than sixfold higher risk of heart failure, and nearly fourfold higher risk of atrial fibrillation.
Researchers also saw an association between obstructive sleep apnea and increased risk of infective outcomes, unplanned tracheal intubation, postoperative lung ventilation, and readmission to the ICU.
The majority of patients received nocturnal oximetry monitoring during their first 3 nights after surgery. This revealed that patients without obstructive sleep apnea had significant increases in oxygen desaturation index during their first night after surgery, while those with sleep apnea did not return to their baseline oxygen desaturation index until the third night after surgery.
“Despite a substantial decrease in ODI [oxygen desaturation index] with oxygen therapy in patients with OSA during the first 3 postoperative nights, supplemental oxygen did not modify the association between OSA and postoperative cardiovascular event,” wrote Matthew T.V. Chan, MD, of Chinese University of Hong Kong, Prince of Wales Hospital, and coauthors.
Given that the events were associated with longer durations of severe oxyhemoglobin desaturation, more aggressive interventions such as positive airway pressure or oral appliances may be required, they noted.
“However, high-level evidence demonstrating the effect of these measures on perioperative outcomes is lacking [and] further clinical trials are now required to test if additional monitoring or alternative interventions would reduce the risk,” they wrote.
The study was supported by the Health and Medical Research Fund (Hong Kong), National Healthcare Group–Khoo Teck Puat Hospital, University Health Network Foundation, University of Malaya, Malaysian Society of Anaesthesiologists, Auckland Medical Research Foundation, and ResMed. One author declared grants from private industry and a patent pending on an obstructive sleep apnea risk questionnaire used in the study.
SOURCE: Chan M et al. JAMA 2019;321[18]:1788-98. doi: 10.1001/jama.2019.4783.
This study is large, prospective, and rigorous and adds important new information to the puzzle of the impact of sleep apnea on postoperative risk, Dennis Auckley, MD, and Stavros Memtsoudis, MD, wrote in an editorial accompanying this study. The study focused on predetermined clinically significant and measurable events, used standardized and objective sleep apnea testing, and attempted to control for many of the confounders that might have influenced outcomes.
The results suggest that obstructive sleep apnea should be recognized as a major perioperative risk factor, and it should receive the same attention and optimization efforts as comorbidities such as diabetes.
Dr. Auckley is from the division of pulmonary, critical care and sleep medicine at MetroHealth Medical Center, Case Western Reserve University, Cleveland, and Dr. Memtsoudis is clinical professor of anesthesiology at Cornell University, New York. These comments are adapted from an editorial (JAMA 2019;231[18]:1775-6). Both declared board and executive positions with the Society of Anesthesia and Sleep Medicine. Dr. Auckley declared research funding from Medtronic, and Dr. Memtsoudis declared personal fees from Teikoku and Sandoz.
This study is large, prospective, and rigorous and adds important new information to the puzzle of the impact of sleep apnea on postoperative risk, Dennis Auckley, MD, and Stavros Memtsoudis, MD, wrote in an editorial accompanying this study. The study focused on predetermined clinically significant and measurable events, used standardized and objective sleep apnea testing, and attempted to control for many of the confounders that might have influenced outcomes.
The results suggest that obstructive sleep apnea should be recognized as a major perioperative risk factor, and it should receive the same attention and optimization efforts as comorbidities such as diabetes.
Dr. Auckley is from the division of pulmonary, critical care and sleep medicine at MetroHealth Medical Center, Case Western Reserve University, Cleveland, and Dr. Memtsoudis is clinical professor of anesthesiology at Cornell University, New York. These comments are adapted from an editorial (JAMA 2019;231[18]:1775-6). Both declared board and executive positions with the Society of Anesthesia and Sleep Medicine. Dr. Auckley declared research funding from Medtronic, and Dr. Memtsoudis declared personal fees from Teikoku and Sandoz.
This study is large, prospective, and rigorous and adds important new information to the puzzle of the impact of sleep apnea on postoperative risk, Dennis Auckley, MD, and Stavros Memtsoudis, MD, wrote in an editorial accompanying this study. The study focused on predetermined clinically significant and measurable events, used standardized and objective sleep apnea testing, and attempted to control for many of the confounders that might have influenced outcomes.
The results suggest that obstructive sleep apnea should be recognized as a major perioperative risk factor, and it should receive the same attention and optimization efforts as comorbidities such as diabetes.
Dr. Auckley is from the division of pulmonary, critical care and sleep medicine at MetroHealth Medical Center, Case Western Reserve University, Cleveland, and Dr. Memtsoudis is clinical professor of anesthesiology at Cornell University, New York. These comments are adapted from an editorial (JAMA 2019;231[18]:1775-6). Both declared board and executive positions with the Society of Anesthesia and Sleep Medicine. Dr. Auckley declared research funding from Medtronic, and Dr. Memtsoudis declared personal fees from Teikoku and Sandoz.
Unrecognized severe obstructive sleep apnea is a risk factor for cardiovascular complications after major noncardiac surgery, according to a study published in JAMA.
The researchers state that perioperative mismanagement of obstructive sleep apnea can lead to serious medical consequences. “General anesthetics, sedatives, and postoperative analgesics are potent respiratory depressants that relax the upper airway dilator muscles and impair ventilatory response to hypoxemia and hypercapnia. Each of these events exacerbates [obstructive sleep apnea] and may predispose patients to postoperative cardiovascular complications,” said researchers who conducted the The Postoperative vascular complications in unrecognised Obstructive Sleep apnoea (POSA) study (NCT01494181).
They undertook a prospective observational cohort study involving 1,218 patients undergoing major noncardiac surgery, who were already considered at high risk of postoperative cardiovascular events – having, for example, a history of coronary artery disease, stroke, diabetes, or renal impairment. However, none had a prior diagnosis of obstructive sleep apnea.
Preoperative sleep monitoring revealed that two-thirds of the cohort had unrecognized and untreated obstructive sleep apnea, including 11.2% with severe obstructive sleep apnea.
At 30 days after surgery, patients with obstructive sleep apnea had a 49% higher risk of the primary outcome of myocardial injury, cardiac death, heart failure, thromboembolism, atrial fibrillation, or stroke, compared with those without obstructive sleep apnea.
However, this association was largely due to a significant 2.23-fold higher risk among patients with severe obstructive sleep apnea, while those with only moderate or mild sleep apnea did not show a significant increased risk of cardiovascular complications.
Patients in this study with severe obstructive sleep apnea had a 13-fold higher risk of cardiac death, 80% higher risk of myocardial injury, more than sixfold higher risk of heart failure, and nearly fourfold higher risk of atrial fibrillation.
Researchers also saw an association between obstructive sleep apnea and increased risk of infective outcomes, unplanned tracheal intubation, postoperative lung ventilation, and readmission to the ICU.
The majority of patients received nocturnal oximetry monitoring during their first 3 nights after surgery. This revealed that patients without obstructive sleep apnea had significant increases in oxygen desaturation index during their first night after surgery, while those with sleep apnea did not return to their baseline oxygen desaturation index until the third night after surgery.
“Despite a substantial decrease in ODI [oxygen desaturation index] with oxygen therapy in patients with OSA during the first 3 postoperative nights, supplemental oxygen did not modify the association between OSA and postoperative cardiovascular event,” wrote Matthew T.V. Chan, MD, of Chinese University of Hong Kong, Prince of Wales Hospital, and coauthors.
Given that the events were associated with longer durations of severe oxyhemoglobin desaturation, more aggressive interventions such as positive airway pressure or oral appliances may be required, they noted.
“However, high-level evidence demonstrating the effect of these measures on perioperative outcomes is lacking [and] further clinical trials are now required to test if additional monitoring or alternative interventions would reduce the risk,” they wrote.
The study was supported by the Health and Medical Research Fund (Hong Kong), National Healthcare Group–Khoo Teck Puat Hospital, University Health Network Foundation, University of Malaya, Malaysian Society of Anaesthesiologists, Auckland Medical Research Foundation, and ResMed. One author declared grants from private industry and a patent pending on an obstructive sleep apnea risk questionnaire used in the study.
SOURCE: Chan M et al. JAMA 2019;321[18]:1788-98. doi: 10.1001/jama.2019.4783.
Unrecognized severe obstructive sleep apnea is a risk factor for cardiovascular complications after major noncardiac surgery, according to a study published in JAMA.
The researchers state that perioperative mismanagement of obstructive sleep apnea can lead to serious medical consequences. “General anesthetics, sedatives, and postoperative analgesics are potent respiratory depressants that relax the upper airway dilator muscles and impair ventilatory response to hypoxemia and hypercapnia. Each of these events exacerbates [obstructive sleep apnea] and may predispose patients to postoperative cardiovascular complications,” said researchers who conducted the The Postoperative vascular complications in unrecognised Obstructive Sleep apnoea (POSA) study (NCT01494181).
They undertook a prospective observational cohort study involving 1,218 patients undergoing major noncardiac surgery, who were already considered at high risk of postoperative cardiovascular events – having, for example, a history of coronary artery disease, stroke, diabetes, or renal impairment. However, none had a prior diagnosis of obstructive sleep apnea.
Preoperative sleep monitoring revealed that two-thirds of the cohort had unrecognized and untreated obstructive sleep apnea, including 11.2% with severe obstructive sleep apnea.
At 30 days after surgery, patients with obstructive sleep apnea had a 49% higher risk of the primary outcome of myocardial injury, cardiac death, heart failure, thromboembolism, atrial fibrillation, or stroke, compared with those without obstructive sleep apnea.
However, this association was largely due to a significant 2.23-fold higher risk among patients with severe obstructive sleep apnea, while those with only moderate or mild sleep apnea did not show a significant increased risk of cardiovascular complications.
Patients in this study with severe obstructive sleep apnea had a 13-fold higher risk of cardiac death, 80% higher risk of myocardial injury, more than sixfold higher risk of heart failure, and nearly fourfold higher risk of atrial fibrillation.
Researchers also saw an association between obstructive sleep apnea and increased risk of infective outcomes, unplanned tracheal intubation, postoperative lung ventilation, and readmission to the ICU.
The majority of patients received nocturnal oximetry monitoring during their first 3 nights after surgery. This revealed that patients without obstructive sleep apnea had significant increases in oxygen desaturation index during their first night after surgery, while those with sleep apnea did not return to their baseline oxygen desaturation index until the third night after surgery.
“Despite a substantial decrease in ODI [oxygen desaturation index] with oxygen therapy in patients with OSA during the first 3 postoperative nights, supplemental oxygen did not modify the association between OSA and postoperative cardiovascular event,” wrote Matthew T.V. Chan, MD, of Chinese University of Hong Kong, Prince of Wales Hospital, and coauthors.
Given that the events were associated with longer durations of severe oxyhemoglobin desaturation, more aggressive interventions such as positive airway pressure or oral appliances may be required, they noted.
“However, high-level evidence demonstrating the effect of these measures on perioperative outcomes is lacking [and] further clinical trials are now required to test if additional monitoring or alternative interventions would reduce the risk,” they wrote.
The study was supported by the Health and Medical Research Fund (Hong Kong), National Healthcare Group–Khoo Teck Puat Hospital, University Health Network Foundation, University of Malaya, Malaysian Society of Anaesthesiologists, Auckland Medical Research Foundation, and ResMed. One author declared grants from private industry and a patent pending on an obstructive sleep apnea risk questionnaire used in the study.
SOURCE: Chan M et al. JAMA 2019;321[18]:1788-98. doi: 10.1001/jama.2019.4783.
FROM JAMA
Pilot program trains residents in telemedicine
WASHINGTON – while still providing the same quality of care during clinic visits, according to the results of a new pilot program.
While telephone visits have been shown to be an effective alternative to face-to-face visits for physicians, residents are not trained to perform telemedicine.
“We have no structured training program on how to teach these residents these skills. We assume that the same skills needed in the clinic are applicable through telephone medicine and we’re not sure about that yet,” Jenna L. Laughlin, DO, said at the annual meeting of the Society of General Internal Medicine.
The pilot study took place at Christiana Care Health System, Wilmington, Del., from October 2016 through December 2018. Nine residents were enrolled in the study, but only six participated.
The objective of the pilot was to demonstrate the feasibility of adding telephone visits into the internal medicine residency practice. This feasibility was measured by templating, scheduling, and supervision, noted Dr. Laughlin, an internist at Christiana Care Health System in Newark, Del. Templating was a term used to describe the designing of the program. For example, the researcher templated 1-hour telephone visit blocks, meaning they designed this structure to integrate into the resident’s training program.
Each resident had 1 hour within the weekly schedule to conduct telephone visits. First-year residents were allowed to do one telephone visit during that hour, second-year residents could do two telephone visits within the hour, and third-year residents could do three telephone visits in that hour.
“There were some challenges associated with scheduling and templating, such as initial schedule templates and a high utilization learning curve,” said Dr. Laughlin, lead author of the study and the program’s leader.
Some didn’t schedule enough telemedicine visits; those residents were assigned to acute clinic visits during the times that they should have been practicing telemedicine.
The program’s second objective was to evaluate the resident experience of scheduled telemedicine visits. This included provider-patient continuity and patient ownership.
The six residents who were surveyed about their experiences said these telephone visits increased their patient ownership and allowed them to build rapport with patients. They said the level of supervision they received was appropriate.
In terms of continuity of care, out the 273 telephone visits scheduled, 179 (65%) were with the patient’s primary care provider and 71 (26.0%) were within the primary care provider’s firm.
“As we build primary care practices in the future, there will be many alternative methods of working with our patients, compared to the traditional face-to-face encounters. I think these are increasingly going to be something that our medical residents need for their future,” Dr. Laughlin said.
The authors reported no conflicts of interest.
WASHINGTON – while still providing the same quality of care during clinic visits, according to the results of a new pilot program.
While telephone visits have been shown to be an effective alternative to face-to-face visits for physicians, residents are not trained to perform telemedicine.
“We have no structured training program on how to teach these residents these skills. We assume that the same skills needed in the clinic are applicable through telephone medicine and we’re not sure about that yet,” Jenna L. Laughlin, DO, said at the annual meeting of the Society of General Internal Medicine.
The pilot study took place at Christiana Care Health System, Wilmington, Del., from October 2016 through December 2018. Nine residents were enrolled in the study, but only six participated.
The objective of the pilot was to demonstrate the feasibility of adding telephone visits into the internal medicine residency practice. This feasibility was measured by templating, scheduling, and supervision, noted Dr. Laughlin, an internist at Christiana Care Health System in Newark, Del. Templating was a term used to describe the designing of the program. For example, the researcher templated 1-hour telephone visit blocks, meaning they designed this structure to integrate into the resident’s training program.
Each resident had 1 hour within the weekly schedule to conduct telephone visits. First-year residents were allowed to do one telephone visit during that hour, second-year residents could do two telephone visits within the hour, and third-year residents could do three telephone visits in that hour.
“There were some challenges associated with scheduling and templating, such as initial schedule templates and a high utilization learning curve,” said Dr. Laughlin, lead author of the study and the program’s leader.
Some didn’t schedule enough telemedicine visits; those residents were assigned to acute clinic visits during the times that they should have been practicing telemedicine.
The program’s second objective was to evaluate the resident experience of scheduled telemedicine visits. This included provider-patient continuity and patient ownership.
The six residents who were surveyed about their experiences said these telephone visits increased their patient ownership and allowed them to build rapport with patients. They said the level of supervision they received was appropriate.
In terms of continuity of care, out the 273 telephone visits scheduled, 179 (65%) were with the patient’s primary care provider and 71 (26.0%) were within the primary care provider’s firm.
“As we build primary care practices in the future, there will be many alternative methods of working with our patients, compared to the traditional face-to-face encounters. I think these are increasingly going to be something that our medical residents need for their future,” Dr. Laughlin said.
The authors reported no conflicts of interest.
WASHINGTON – while still providing the same quality of care during clinic visits, according to the results of a new pilot program.
While telephone visits have been shown to be an effective alternative to face-to-face visits for physicians, residents are not trained to perform telemedicine.
“We have no structured training program on how to teach these residents these skills. We assume that the same skills needed in the clinic are applicable through telephone medicine and we’re not sure about that yet,” Jenna L. Laughlin, DO, said at the annual meeting of the Society of General Internal Medicine.
The pilot study took place at Christiana Care Health System, Wilmington, Del., from October 2016 through December 2018. Nine residents were enrolled in the study, but only six participated.
The objective of the pilot was to demonstrate the feasibility of adding telephone visits into the internal medicine residency practice. This feasibility was measured by templating, scheduling, and supervision, noted Dr. Laughlin, an internist at Christiana Care Health System in Newark, Del. Templating was a term used to describe the designing of the program. For example, the researcher templated 1-hour telephone visit blocks, meaning they designed this structure to integrate into the resident’s training program.
Each resident had 1 hour within the weekly schedule to conduct telephone visits. First-year residents were allowed to do one telephone visit during that hour, second-year residents could do two telephone visits within the hour, and third-year residents could do three telephone visits in that hour.
“There were some challenges associated with scheduling and templating, such as initial schedule templates and a high utilization learning curve,” said Dr. Laughlin, lead author of the study and the program’s leader.
Some didn’t schedule enough telemedicine visits; those residents were assigned to acute clinic visits during the times that they should have been practicing telemedicine.
The program’s second objective was to evaluate the resident experience of scheduled telemedicine visits. This included provider-patient continuity and patient ownership.
The six residents who were surveyed about their experiences said these telephone visits increased their patient ownership and allowed them to build rapport with patients. They said the level of supervision they received was appropriate.
In terms of continuity of care, out the 273 telephone visits scheduled, 179 (65%) were with the patient’s primary care provider and 71 (26.0%) were within the primary care provider’s firm.
“As we build primary care practices in the future, there will be many alternative methods of working with our patients, compared to the traditional face-to-face encounters. I think these are increasingly going to be something that our medical residents need for their future,” Dr. Laughlin said.
The authors reported no conflicts of interest.
REPORTING FROM SGIM 2019
N.Y. hospitals report near-universal CMV screening when newborns fail hearing tests
BALTIMORE – Over the past 2 years, Northwell Health, a large medical system in the metropolitan New York area, increased cytomegalovirus screening for infants who fail hearing tests from 6.6% to 95% at five of its birth hospitals, according to a presentation at the Pediatric Academic Societies annual meeting.
Three cases of congenital cytomegalovirus (CMV) have been picked up so far. The plan is to roll the program out to all 10 of the system’s birth hospitals, where over 40,000 children are born each year.
“We feel very satisfied and proud” of the progress that’s been made at Northwell in such a short time, said Alia Chauhan, MD, a Northwell pediatrician who presented the findings.
Northwell launched its “Hearing Plus” program in 2017 to catch the infection before infants leave the hospital. Several other health systems around the country have launched similar programs, and a handful of states – including New York – now require CMV screening for infants who fail mandated hearing tests.
The issue is gaining traction because hearing loss is often the only sign of congenital CMV, so it’s a bellwether for infection. Screening children with hearing loss is an easy way to pick it up early, so steps can be taken to prevent problems down the road. As it is, congenital CMV is the leading nongenetic cause of hearing loss in infants, accounting for at least 10% of cases.
The Northwell program kicked off with an education campaign to build consensus among pediatricians, hospitalists, and nurses. A flyer was made about CMV screening for moms whose infants fail hearing tests, printed in both English and Spanish.
Initially, the program used urine PCR [polymerase chain reaction] to screen for CMV, but waiting for infants to produce a sample often delayed discharge, so a switch was soon made to saliva swab PCRs, which take seconds, with urine PCR held in reserve to confirm positive swabs.
To streamline the process, a standing order was added to the electronic records system so nurses could order saliva PCRs without having to get physician approval. “I think [that] was one of the biggest things that’s helped us,” Dr. Chauhan said.
Children who test positive must have urine confirmation within 21 days of birth; most are long gone from the hospital by then and have to be called back in. “We haven’t lost anyone to follow-up, but it can be stressful trying to get someone to come back,” she said.
Six of 449 infants have screened positive on saliva – three were false positives with negative urine screens. Of the three confirmed cases, two infants later turned out to have normal hearing on repeat testing and were otherwise asymptomatic.
These days, Dr. Chauhan said, if children have a positive saliva PCR but later turn out to have normal hearing, and are otherwise free of symptoms with no CMV risk factors, “we are not confirming with urine.”
Dr. Chauhan did not have any disclosures. No funding source was mentioned.
SOURCE: Chauhan A et al. PAS 2019. Abstract 306
BALTIMORE – Over the past 2 years, Northwell Health, a large medical system in the metropolitan New York area, increased cytomegalovirus screening for infants who fail hearing tests from 6.6% to 95% at five of its birth hospitals, according to a presentation at the Pediatric Academic Societies annual meeting.
Three cases of congenital cytomegalovirus (CMV) have been picked up so far. The plan is to roll the program out to all 10 of the system’s birth hospitals, where over 40,000 children are born each year.
“We feel very satisfied and proud” of the progress that’s been made at Northwell in such a short time, said Alia Chauhan, MD, a Northwell pediatrician who presented the findings.
Northwell launched its “Hearing Plus” program in 2017 to catch the infection before infants leave the hospital. Several other health systems around the country have launched similar programs, and a handful of states – including New York – now require CMV screening for infants who fail mandated hearing tests.
The issue is gaining traction because hearing loss is often the only sign of congenital CMV, so it’s a bellwether for infection. Screening children with hearing loss is an easy way to pick it up early, so steps can be taken to prevent problems down the road. As it is, congenital CMV is the leading nongenetic cause of hearing loss in infants, accounting for at least 10% of cases.
The Northwell program kicked off with an education campaign to build consensus among pediatricians, hospitalists, and nurses. A flyer was made about CMV screening for moms whose infants fail hearing tests, printed in both English and Spanish.
Initially, the program used urine PCR [polymerase chain reaction] to screen for CMV, but waiting for infants to produce a sample often delayed discharge, so a switch was soon made to saliva swab PCRs, which take seconds, with urine PCR held in reserve to confirm positive swabs.
To streamline the process, a standing order was added to the electronic records system so nurses could order saliva PCRs without having to get physician approval. “I think [that] was one of the biggest things that’s helped us,” Dr. Chauhan said.
Children who test positive must have urine confirmation within 21 days of birth; most are long gone from the hospital by then and have to be called back in. “We haven’t lost anyone to follow-up, but it can be stressful trying to get someone to come back,” she said.
Six of 449 infants have screened positive on saliva – three were false positives with negative urine screens. Of the three confirmed cases, two infants later turned out to have normal hearing on repeat testing and were otherwise asymptomatic.
These days, Dr. Chauhan said, if children have a positive saliva PCR but later turn out to have normal hearing, and are otherwise free of symptoms with no CMV risk factors, “we are not confirming with urine.”
Dr. Chauhan did not have any disclosures. No funding source was mentioned.
SOURCE: Chauhan A et al. PAS 2019. Abstract 306
BALTIMORE – Over the past 2 years, Northwell Health, a large medical system in the metropolitan New York area, increased cytomegalovirus screening for infants who fail hearing tests from 6.6% to 95% at five of its birth hospitals, according to a presentation at the Pediatric Academic Societies annual meeting.
Three cases of congenital cytomegalovirus (CMV) have been picked up so far. The plan is to roll the program out to all 10 of the system’s birth hospitals, where over 40,000 children are born each year.
“We feel very satisfied and proud” of the progress that’s been made at Northwell in such a short time, said Alia Chauhan, MD, a Northwell pediatrician who presented the findings.
Northwell launched its “Hearing Plus” program in 2017 to catch the infection before infants leave the hospital. Several other health systems around the country have launched similar programs, and a handful of states – including New York – now require CMV screening for infants who fail mandated hearing tests.
The issue is gaining traction because hearing loss is often the only sign of congenital CMV, so it’s a bellwether for infection. Screening children with hearing loss is an easy way to pick it up early, so steps can be taken to prevent problems down the road. As it is, congenital CMV is the leading nongenetic cause of hearing loss in infants, accounting for at least 10% of cases.
The Northwell program kicked off with an education campaign to build consensus among pediatricians, hospitalists, and nurses. A flyer was made about CMV screening for moms whose infants fail hearing tests, printed in both English and Spanish.
Initially, the program used urine PCR [polymerase chain reaction] to screen for CMV, but waiting for infants to produce a sample often delayed discharge, so a switch was soon made to saliva swab PCRs, which take seconds, with urine PCR held in reserve to confirm positive swabs.
To streamline the process, a standing order was added to the electronic records system so nurses could order saliva PCRs without having to get physician approval. “I think [that] was one of the biggest things that’s helped us,” Dr. Chauhan said.
Children who test positive must have urine confirmation within 21 days of birth; most are long gone from the hospital by then and have to be called back in. “We haven’t lost anyone to follow-up, but it can be stressful trying to get someone to come back,” she said.
Six of 449 infants have screened positive on saliva – three were false positives with negative urine screens. Of the three confirmed cases, two infants later turned out to have normal hearing on repeat testing and were otherwise asymptomatic.
These days, Dr. Chauhan said, if children have a positive saliva PCR but later turn out to have normal hearing, and are otherwise free of symptoms with no CMV risk factors, “we are not confirming with urine.”
Dr. Chauhan did not have any disclosures. No funding source was mentioned.
SOURCE: Chauhan A et al. PAS 2019. Abstract 306
REPORTING FROM PAS 2019
Key clinical point: A metropolitan N.Y. health system provides a model for how to implement cytomegalovirus screening for infants who fail hearing tests.
Major finding: .
Study details: Pre-post quality improvement project.
Disclosures: The lead investigator had no disclosures. No funding source was mentioned.
Source: Chauhan A et al. PAS 2019. Abstract 306.
AFib screening cuts hospitalizations and ED visits
NEW ORLEANS – People diagnosed with atrial fibrillation by screening with a wearable ECG patch had significantly fewer emergency department visits or hospital admissions, compared with similar people diagnosed with atrial fibrillation by usual-care surveillance in an observational study with 5,109 total participants.
People diagnosed with atrial fibrillation (AFib) through screening had a statistically significant 80% relative cut in hospitalizations and a 65% cut in emergency department visits during 12 months of follow-up, compared with controls in the study who had their AFib identified and diagnosed as part of routine practice, Steven R. Steinhubl, MD, said at the annual meeting of the American College of Cardiology.
The data also showed no difference between the screened and control patients identified with AFib in the average number of cardiologist consultations during a year of follow-up, and a trend that missed statistical significance for 16% fewer primary care physician visits in Afib patients diagnosed by screening rather than by routine surveillance.
These findings provided some insight into the potential clinical impact of AFib screening in at-risk people. Dr. Steinhubl and his associates plan to report on the incidence of strokes and MIs in the two study subgroups after 3 years of follow-up, but he noted that preliminary findings for these two outcomes after 1 year indicated that active screening for AFib also had reduced these rates, compared with waiting for the arrhythmia to become apparent by emergence of symptoms.
The data came from the mSToPS (mHealth Screening to Prevent Strokes) study, which randomized 2,659 U.S. residents enrolled in a large health plan who had risk factors for AFib to either immediate or delayed arrhythmia assessment by ECG patches. Half the participants used a patch for about 14 days immediately and then a second time 3 months later, while the other half waited 4 months and then wore an ECG patch for 2 weeks and again 3 months later. The primary endpoint, first reported at the ACC annual meeting a year before and subsequently published, was the incidence of newly diagnosed AFib during the first 4 months in the actively monitored cohort, compared with a cohort followed by usual care. The results showed that screening identified AFib in 3.9% of people, while no screening and usual-practice follow-up identified a 0.9% incidence of AFib, showing that screening worked better for AFib case identification (JAMA. 2018 Jul 10;320[2]:146-55).
To examine the clinical impact of screening and an increased incidence of diagnosed AFib cases, Dr. Steinhubl and his associates focused on 1,725 of the original 2,659 patients who underwent ECG patch assessment, either immediate or delayed, and continued through 12 months of follow-up, and compared them with 3,384 matched controls who never underwent ECG patch screening but were also followed for 12 months for incident AFib identified during routine care and surveillance. This resulted in a cumulative incidence of newly diagnosed AFib of 6.3% in those who had worn two ECG patches and 2.3% among the matched controls.
During follow-up, use of various interventions was more common among the screened people than the controls. Initiation of anticoagulation treatment started in 4.0% of the entire screened group, compared with 1.9% of the controls, The screened people also had a 0.9% rate of receiving a pacemaker or defibrillator, a 0.8% rate of starting on treatment with an antiarrhythmic drug, and a 0.3% rate of undergoing catheter ablation, compared with none, 0.3%, and one of the controls, respectively, said Dr. Steinhubl, director of digital medicine at the Scripps Research Translational Institute in La Jolla, Calif.
The mSToPS study was funded by Janssen. Dr. Steinhubl has received research funding from DynoSense, EasyG, Janssen, the Qualcomm Foundation, and Striv.
SOURCE: Steinhubl SR et al. J Am Coll Cardiol. 2019 Mar 12;73(9)suppl 1:296.
NEW ORLEANS – People diagnosed with atrial fibrillation by screening with a wearable ECG patch had significantly fewer emergency department visits or hospital admissions, compared with similar people diagnosed with atrial fibrillation by usual-care surveillance in an observational study with 5,109 total participants.
People diagnosed with atrial fibrillation (AFib) through screening had a statistically significant 80% relative cut in hospitalizations and a 65% cut in emergency department visits during 12 months of follow-up, compared with controls in the study who had their AFib identified and diagnosed as part of routine practice, Steven R. Steinhubl, MD, said at the annual meeting of the American College of Cardiology.
The data also showed no difference between the screened and control patients identified with AFib in the average number of cardiologist consultations during a year of follow-up, and a trend that missed statistical significance for 16% fewer primary care physician visits in Afib patients diagnosed by screening rather than by routine surveillance.
These findings provided some insight into the potential clinical impact of AFib screening in at-risk people. Dr. Steinhubl and his associates plan to report on the incidence of strokes and MIs in the two study subgroups after 3 years of follow-up, but he noted that preliminary findings for these two outcomes after 1 year indicated that active screening for AFib also had reduced these rates, compared with waiting for the arrhythmia to become apparent by emergence of symptoms.
The data came from the mSToPS (mHealth Screening to Prevent Strokes) study, which randomized 2,659 U.S. residents enrolled in a large health plan who had risk factors for AFib to either immediate or delayed arrhythmia assessment by ECG patches. Half the participants used a patch for about 14 days immediately and then a second time 3 months later, while the other half waited 4 months and then wore an ECG patch for 2 weeks and again 3 months later. The primary endpoint, first reported at the ACC annual meeting a year before and subsequently published, was the incidence of newly diagnosed AFib during the first 4 months in the actively monitored cohort, compared with a cohort followed by usual care. The results showed that screening identified AFib in 3.9% of people, while no screening and usual-practice follow-up identified a 0.9% incidence of AFib, showing that screening worked better for AFib case identification (JAMA. 2018 Jul 10;320[2]:146-55).
To examine the clinical impact of screening and an increased incidence of diagnosed AFib cases, Dr. Steinhubl and his associates focused on 1,725 of the original 2,659 patients who underwent ECG patch assessment, either immediate or delayed, and continued through 12 months of follow-up, and compared them with 3,384 matched controls who never underwent ECG patch screening but were also followed for 12 months for incident AFib identified during routine care and surveillance. This resulted in a cumulative incidence of newly diagnosed AFib of 6.3% in those who had worn two ECG patches and 2.3% among the matched controls.
During follow-up, use of various interventions was more common among the screened people than the controls. Initiation of anticoagulation treatment started in 4.0% of the entire screened group, compared with 1.9% of the controls, The screened people also had a 0.9% rate of receiving a pacemaker or defibrillator, a 0.8% rate of starting on treatment with an antiarrhythmic drug, and a 0.3% rate of undergoing catheter ablation, compared with none, 0.3%, and one of the controls, respectively, said Dr. Steinhubl, director of digital medicine at the Scripps Research Translational Institute in La Jolla, Calif.
The mSToPS study was funded by Janssen. Dr. Steinhubl has received research funding from DynoSense, EasyG, Janssen, the Qualcomm Foundation, and Striv.
SOURCE: Steinhubl SR et al. J Am Coll Cardiol. 2019 Mar 12;73(9)suppl 1:296.
NEW ORLEANS – People diagnosed with atrial fibrillation by screening with a wearable ECG patch had significantly fewer emergency department visits or hospital admissions, compared with similar people diagnosed with atrial fibrillation by usual-care surveillance in an observational study with 5,109 total participants.
People diagnosed with atrial fibrillation (AFib) through screening had a statistically significant 80% relative cut in hospitalizations and a 65% cut in emergency department visits during 12 months of follow-up, compared with controls in the study who had their AFib identified and diagnosed as part of routine practice, Steven R. Steinhubl, MD, said at the annual meeting of the American College of Cardiology.
The data also showed no difference between the screened and control patients identified with AFib in the average number of cardiologist consultations during a year of follow-up, and a trend that missed statistical significance for 16% fewer primary care physician visits in Afib patients diagnosed by screening rather than by routine surveillance.
These findings provided some insight into the potential clinical impact of AFib screening in at-risk people. Dr. Steinhubl and his associates plan to report on the incidence of strokes and MIs in the two study subgroups after 3 years of follow-up, but he noted that preliminary findings for these two outcomes after 1 year indicated that active screening for AFib also had reduced these rates, compared with waiting for the arrhythmia to become apparent by emergence of symptoms.
The data came from the mSToPS (mHealth Screening to Prevent Strokes) study, which randomized 2,659 U.S. residents enrolled in a large health plan who had risk factors for AFib to either immediate or delayed arrhythmia assessment by ECG patches. Half the participants used a patch for about 14 days immediately and then a second time 3 months later, while the other half waited 4 months and then wore an ECG patch for 2 weeks and again 3 months later. The primary endpoint, first reported at the ACC annual meeting a year before and subsequently published, was the incidence of newly diagnosed AFib during the first 4 months in the actively monitored cohort, compared with a cohort followed by usual care. The results showed that screening identified AFib in 3.9% of people, while no screening and usual-practice follow-up identified a 0.9% incidence of AFib, showing that screening worked better for AFib case identification (JAMA. 2018 Jul 10;320[2]:146-55).
To examine the clinical impact of screening and an increased incidence of diagnosed AFib cases, Dr. Steinhubl and his associates focused on 1,725 of the original 2,659 patients who underwent ECG patch assessment, either immediate or delayed, and continued through 12 months of follow-up, and compared them with 3,384 matched controls who never underwent ECG patch screening but were also followed for 12 months for incident AFib identified during routine care and surveillance. This resulted in a cumulative incidence of newly diagnosed AFib of 6.3% in those who had worn two ECG patches and 2.3% among the matched controls.
During follow-up, use of various interventions was more common among the screened people than the controls. Initiation of anticoagulation treatment started in 4.0% of the entire screened group, compared with 1.9% of the controls, The screened people also had a 0.9% rate of receiving a pacemaker or defibrillator, a 0.8% rate of starting on treatment with an antiarrhythmic drug, and a 0.3% rate of undergoing catheter ablation, compared with none, 0.3%, and one of the controls, respectively, said Dr. Steinhubl, director of digital medicine at the Scripps Research Translational Institute in La Jolla, Calif.
The mSToPS study was funded by Janssen. Dr. Steinhubl has received research funding from DynoSense, EasyG, Janssen, the Qualcomm Foundation, and Striv.
SOURCE: Steinhubl SR et al. J Am Coll Cardiol. 2019 Mar 12;73(9)suppl 1:296.
REPORTING FROM ACC 2019
Biomarker-based score predicts poor outcomes after acute ischemic stroke
PHILADELPHIA – A prognostic score for acute ischemic stroke that incorporates copeptin levels, age, recanalization, and National Institutes of Health Stroke Scale score has been externally validated and accurately predicts unfavorable outcome, according to research presented at the annual meeting of the American Academy of Neurology.
Although the four-item score could not be validated for mortality prediction, it had reasonable accuracy for predicting unfavorable functional outcome, defined as disability or mortality 3 months after ischemic stroke, Gian Marco De Marchis, MD, of the department of neurology and the stroke center at University Hospital Basel (Switzerland), said in a presentation.
“The use of a biomarker increases prognostic accuracy, allowing us to personalize prognosis in the frame of individualized, precision medicine,” Dr. De Marchis said.
Copeptin has been linked to disability and mortality at 3 months in two independent, large cohort studies of patients with ischemic stroke, he said.
The four-item prognostic score devised by Dr. De Marchis and his coinvestigators, which they call the CoRisk score, was developed based on a derivation cohort of 319 acute ischemic stroke patients and a validation cohort including another 783 patients in the Copeptin for Risk Stratification in Acute Stroke Patients (CoRisk) Study.
Diagnostic accuracy was 82% for the endpoint of unfavorable functional outcome at 3 months, according to Dr. De Marchis.
“The observed outcomes matched well with the expected outcomes,” he said in his presentation.
Further analyses demonstrated that the addition of copeptin indeed contributed to the diagnostic accuracy of the score, improving the classification for 46%; in other words, about half of the patients were reclassified based on addition of the biomarker data.
By contrast, the score is not well suited to predict mortality alone at 3 months, the results of the analyses showed.
The algorithm used to calculate the score based on its four variables is somewhat complex, but available as a free app and online calculator, Dr. De Marchis said.
Dr. De Marchis and his coauthors had nothing to disclose related to their study. A full report on the study was published ahead of print on March 1 in Neurology.
SOURCE: De Marchis GM et al. AAN 2019, Abstract S47.001.
PHILADELPHIA – A prognostic score for acute ischemic stroke that incorporates copeptin levels, age, recanalization, and National Institutes of Health Stroke Scale score has been externally validated and accurately predicts unfavorable outcome, according to research presented at the annual meeting of the American Academy of Neurology.
Although the four-item score could not be validated for mortality prediction, it had reasonable accuracy for predicting unfavorable functional outcome, defined as disability or mortality 3 months after ischemic stroke, Gian Marco De Marchis, MD, of the department of neurology and the stroke center at University Hospital Basel (Switzerland), said in a presentation.
“The use of a biomarker increases prognostic accuracy, allowing us to personalize prognosis in the frame of individualized, precision medicine,” Dr. De Marchis said.
Copeptin has been linked to disability and mortality at 3 months in two independent, large cohort studies of patients with ischemic stroke, he said.
The four-item prognostic score devised by Dr. De Marchis and his coinvestigators, which they call the CoRisk score, was developed based on a derivation cohort of 319 acute ischemic stroke patients and a validation cohort including another 783 patients in the Copeptin for Risk Stratification in Acute Stroke Patients (CoRisk) Study.
Diagnostic accuracy was 82% for the endpoint of unfavorable functional outcome at 3 months, according to Dr. De Marchis.
“The observed outcomes matched well with the expected outcomes,” he said in his presentation.
Further analyses demonstrated that the addition of copeptin indeed contributed to the diagnostic accuracy of the score, improving the classification for 46%; in other words, about half of the patients were reclassified based on addition of the biomarker data.
By contrast, the score is not well suited to predict mortality alone at 3 months, the results of the analyses showed.
The algorithm used to calculate the score based on its four variables is somewhat complex, but available as a free app and online calculator, Dr. De Marchis said.
Dr. De Marchis and his coauthors had nothing to disclose related to their study. A full report on the study was published ahead of print on March 1 in Neurology.
SOURCE: De Marchis GM et al. AAN 2019, Abstract S47.001.
PHILADELPHIA – A prognostic score for acute ischemic stroke that incorporates copeptin levels, age, recanalization, and National Institutes of Health Stroke Scale score has been externally validated and accurately predicts unfavorable outcome, according to research presented at the annual meeting of the American Academy of Neurology.
Although the four-item score could not be validated for mortality prediction, it had reasonable accuracy for predicting unfavorable functional outcome, defined as disability or mortality 3 months after ischemic stroke, Gian Marco De Marchis, MD, of the department of neurology and the stroke center at University Hospital Basel (Switzerland), said in a presentation.
“The use of a biomarker increases prognostic accuracy, allowing us to personalize prognosis in the frame of individualized, precision medicine,” Dr. De Marchis said.
Copeptin has been linked to disability and mortality at 3 months in two independent, large cohort studies of patients with ischemic stroke, he said.
The four-item prognostic score devised by Dr. De Marchis and his coinvestigators, which they call the CoRisk score, was developed based on a derivation cohort of 319 acute ischemic stroke patients and a validation cohort including another 783 patients in the Copeptin for Risk Stratification in Acute Stroke Patients (CoRisk) Study.
Diagnostic accuracy was 82% for the endpoint of unfavorable functional outcome at 3 months, according to Dr. De Marchis.
“The observed outcomes matched well with the expected outcomes,” he said in his presentation.
Further analyses demonstrated that the addition of copeptin indeed contributed to the diagnostic accuracy of the score, improving the classification for 46%; in other words, about half of the patients were reclassified based on addition of the biomarker data.
By contrast, the score is not well suited to predict mortality alone at 3 months, the results of the analyses showed.
The algorithm used to calculate the score based on its four variables is somewhat complex, but available as a free app and online calculator, Dr. De Marchis said.
Dr. De Marchis and his coauthors had nothing to disclose related to their study. A full report on the study was published ahead of print on March 1 in Neurology.
SOURCE: De Marchis GM et al. AAN 2019, Abstract S47.001.
REPORTING FROM AAN 2019
Unit-based models of care
A tool for ensuring patient safety
“To me, teamwork is the beauty of our sport, where you have five acting as one. You become selfless.” – Mike Krzyzewski
High-performing teams plan, communicate, reflect, and take action together. Teamwork can transform seemingly impossible tasks into opportunities for people to come together and create value.
The increasing complexity of health care makes team-based care necessary to achieve successful health outcomes for patients. At the Brooklyn (N.Y.) Hospital Center, a 464-bed care center, we transformed the model of care on the medical wards into a geographic, unit-based team model. Here we describe our journey – the successes, the challenges, and the opportunities for growth.
Previous model
In the previous care model on our medical wards, no set structures were in place. Teams would travel to multiple wards throughout the hospital to see the patients they were rounding on. Each floor had its own set of social workers and case managers, therefore a hospital medicine team routinely dealt with more than eight social workers and case managers to address their patients’ needs in a single day.
Multidisciplinary rounds for all medical patients were held at 11 a.m. in a room located a significant distance away from the medical wards. All case managers and social workers would sit in this room from 11 a.m. until noon, and teams would travel to that room to discuss their patients.
Many challenges were identified in this model, including a lack of communication, a de-emphasis on teamwork, and a design that did not take physician workflows into account resulting in low efficiency. Thus, these challenges sparked a desire to create a more effective and team-based methodology of accomplishing excellence in delivery of clinical care. Dr. Pendharkar, having worked primarily in centers with unit-based care, determined that a geographic, unit-based model of care could transform care delivery at the Brooklyn Hospital Center.
Looking ahead
The efforts for transforming the vision of geographic, unit-based teams into a reality started by gathering all stakeholders together to unite for a common mission. Initial meetings were held with all parties including social workers, case managers, residents, nursing staff, bed board and attending physicians in internal medicine, and the emergency department.
The vision of a geographic, unit-based team was shared and explained to all team members. Exercises in LEAN methodology were conducted, including one-piece flow exercises, to highlight the possibilities of what could be accomplished through teamwork. Once support for the vision was in place from all parties, the logistics were addressed.
The biggest challenge to overcome was how to place all of one team’s patients on a singular medical ward. In our hospital, a medical ward holds anywhere from 30 to 33 patients. Each hospital medicine team, of which there are many, typically carries 20-23 patients. We created a blueprint to map out the floor to which each team and attending would be assigned. Next, we partnered with both IT and bed board to design an admission order set that specified the particular geographic location that a team and attending were associated with so that patients could be placed accordingly from the ED.
It was important for the ED doctors, bed board, and the internal medicine residents to understand these changes because all of these parties were involved in the initial admitting process. Dr. Pendharkar and Dr. Malieckal provided all groups with in-person training on how the logistics of the system would unfold. Noon conference lectures were also held to explain the vision to residents.
Over 3 weeks, the first ward we chose to implement our model on slowly accumulated the patients of one team – this was the gradual trickle phase. We then selected a “re-set” date. On the re-set date, it was determined that all patients would go to the team that was assigned to that floor, with the exception of any private attendings’ patients.
On the day before the re-set date, time was spent ensuring that all hand-offs were safe. Dr. Pendharkar and Dr. Malieckal spoke with every intern and team that would be handing off and/or receiving patients as a result of the re-set policy. The goal was to ensure that on that date a ward had close to 100% of its patients belonging to the team/attending that was assigned to that area.
The good
Once we began our geographic, unit-based model, our rounding process was transformed.
Now, our morning rounds were joined by the bedside nurse, case manager, social worker, clinical pharmacy, and nutrition in addition to the core team. The entire team went from room to room on one ward rounding on all 20 to 25 patients back to back, which created an unparalleled level of efficiency and a forum for effective communication lasting throughout the day.
We also added workstations on wheels (WOWS) to the rounding process so that labs, radiology, and more could be reviewed on rounds with the entire team. A standard script was developed so that each patient was introduced to all members of the team, and the care plan was disclosed and highlighted. One patient noted, “I feel so cared for, knowing I have this entire team taking care of me.” We also rounded in the afternoon with the case managers and social workers to follow up tasks that were to be completed that day.
Our first few weeks utilizing the geographic, unit-based model of rounding was largely successful. The residents, now able to round on all of their patients in one location with one case manager and one social worker, noted, “This model of rounding makes my life so much easier, I feel like I can focus on the patient rather than running around. … and I know the social worker and case manager will help me.”
Provider satisfaction had improved, from residents to physicians to nurses, case managers, social workers, and more. Our case manager also noted her satisfaction with the new model, stating that her communication with the medical team was much easier. As the attending, I witnessed firsthand how working together with the team moved care forward much more quickly, compared with the previous model, because of the simple factor of increased ease of communication.
Now all team members were together in the patient room and discussion was much easier. There was less confusion, fewer delays, and better communication – I think unit-based teams can even be described as a lifesaving measure that reduces harm to patients. An additional benefit is the relationship that now developed between doctors, social workers, and case managers – they spent more time together and really got to know one another, creating a feeling of shared success and a deeper drive to help one another succeed.
In our model, 87% of surveyed residents said they felt less burned out in the new geographic, unit-based model of care, and 91% of physicians surveyed said it was easier to talk with team members to coordinate care. Additionally, our HCAHPS (Hospital Consumer Assessment of Healthcare Providers and Systems) scores saw a drastic increase in many domains. Nursing communication improved by more than 42% on domain 7B; doctor communication improved by more than 31%. Additionally, all other domains saw at least 10% improvement. We are now 5 months out from our initial rollout of the model and continue to see sustained improvements in quality measures.
The bad
The biggest challenges that we are working through with this model are hand-offs and transfer of patients from one team to another. Sometimes, it happens that one team’s patient will wind up on a floor that is the designated floor of another team because of bed availability. We continue to work with bed board to address this issue. We want to minimize transfers and hand-offs to promote continuity and have to balance that with the need for geographic location. With clear communication, hospital collaboration from bed board and safe hand-off methods, this problem can be safely addressed.
Conclusions
The experience of implementing the unit-based team model has been an eye-opening journey. One thing that stands out is that, in an increasingly complex health care system, design thinking is critical.
Design thinking takes into consideration the needs of those who are using a system. In this case, patients and health care workers including doctors, nurses, case managers, and social workers are the end users of the health care system. All parties are utilizing the health care system to optimize patient health. Therefore, we must create systems that are easy to navigate and use by patients and health care workers so that they can ensure the success of patients.
Unit-based teams offer a basic framework to optimize the inpatient system to facilitate better workflow. In our system, it allowed us to optimize communications between health care workers and also between health care workers and patients. It allowed team members to work in close proximity to better share ideas with each other.
We spent a significant amount of time upfront earning the support of all of the disciplines for this effort. We had support from all leaders within the organization and continue to make our case for this model by sharing metrics and holding forums to discuss the process.
Initial data show a marked improvement in many domains of HCAHPS scores. Our frontline staff, including attendings, residents, nursing, case managers, and social workers, also continue to support this effort since it has a positive impact on their workflow and improves their workday quality. One nurse mentioned specifically, “in my 30 years at this hospital I have never seen people work together so well.”
To sustain this effort, we continue to have regular meetings, and there are new features that we would like to add to the program. For example, we are working with our IT group to ensure that each unit-based team will have dashboards available to incorporate real time, actionable data into daily workflows.
We are excited by the potential of our high-performing teams to highlight the patient experience, placing the patient at the center for care, decision making, and rounding. Health care is a team sport, and anytime you build something where all teams are playing together and approaching the finish line as a unit, you will never go wrong!
Dr. Pendharkar is division chief of hospital medicine at the Brooklyn (N.Y.) Hospital Center, medical director of inpatient services and director of quality for the department of medicine at the Brooklyn Hospital Center and assistant professor of medicine, Icahn School of Medicine at Mount Sinai, New York. Dr. Malieckal is chief resident, internal medicine, at the Brooklyn Hospital Center. Dr. Gasperino is chair, department of medicine; vice president for critical care, perioperative, and hospital medicine; and associate chief medical officer at the Brooklyn Hospital Center.
A tool for ensuring patient safety
A tool for ensuring patient safety
“To me, teamwork is the beauty of our sport, where you have five acting as one. You become selfless.” – Mike Krzyzewski
High-performing teams plan, communicate, reflect, and take action together. Teamwork can transform seemingly impossible tasks into opportunities for people to come together and create value.
The increasing complexity of health care makes team-based care necessary to achieve successful health outcomes for patients. At the Brooklyn (N.Y.) Hospital Center, a 464-bed care center, we transformed the model of care on the medical wards into a geographic, unit-based team model. Here we describe our journey – the successes, the challenges, and the opportunities for growth.
Previous model
In the previous care model on our medical wards, no set structures were in place. Teams would travel to multiple wards throughout the hospital to see the patients they were rounding on. Each floor had its own set of social workers and case managers, therefore a hospital medicine team routinely dealt with more than eight social workers and case managers to address their patients’ needs in a single day.
Multidisciplinary rounds for all medical patients were held at 11 a.m. in a room located a significant distance away from the medical wards. All case managers and social workers would sit in this room from 11 a.m. until noon, and teams would travel to that room to discuss their patients.
Many challenges were identified in this model, including a lack of communication, a de-emphasis on teamwork, and a design that did not take physician workflows into account resulting in low efficiency. Thus, these challenges sparked a desire to create a more effective and team-based methodology of accomplishing excellence in delivery of clinical care. Dr. Pendharkar, having worked primarily in centers with unit-based care, determined that a geographic, unit-based model of care could transform care delivery at the Brooklyn Hospital Center.
Looking ahead
The efforts for transforming the vision of geographic, unit-based teams into a reality started by gathering all stakeholders together to unite for a common mission. Initial meetings were held with all parties including social workers, case managers, residents, nursing staff, bed board and attending physicians in internal medicine, and the emergency department.
The vision of a geographic, unit-based team was shared and explained to all team members. Exercises in LEAN methodology were conducted, including one-piece flow exercises, to highlight the possibilities of what could be accomplished through teamwork. Once support for the vision was in place from all parties, the logistics were addressed.
The biggest challenge to overcome was how to place all of one team’s patients on a singular medical ward. In our hospital, a medical ward holds anywhere from 30 to 33 patients. Each hospital medicine team, of which there are many, typically carries 20-23 patients. We created a blueprint to map out the floor to which each team and attending would be assigned. Next, we partnered with both IT and bed board to design an admission order set that specified the particular geographic location that a team and attending were associated with so that patients could be placed accordingly from the ED.
It was important for the ED doctors, bed board, and the internal medicine residents to understand these changes because all of these parties were involved in the initial admitting process. Dr. Pendharkar and Dr. Malieckal provided all groups with in-person training on how the logistics of the system would unfold. Noon conference lectures were also held to explain the vision to residents.
Over 3 weeks, the first ward we chose to implement our model on slowly accumulated the patients of one team – this was the gradual trickle phase. We then selected a “re-set” date. On the re-set date, it was determined that all patients would go to the team that was assigned to that floor, with the exception of any private attendings’ patients.
On the day before the re-set date, time was spent ensuring that all hand-offs were safe. Dr. Pendharkar and Dr. Malieckal spoke with every intern and team that would be handing off and/or receiving patients as a result of the re-set policy. The goal was to ensure that on that date a ward had close to 100% of its patients belonging to the team/attending that was assigned to that area.
The good
Once we began our geographic, unit-based model, our rounding process was transformed.
Now, our morning rounds were joined by the bedside nurse, case manager, social worker, clinical pharmacy, and nutrition in addition to the core team. The entire team went from room to room on one ward rounding on all 20 to 25 patients back to back, which created an unparalleled level of efficiency and a forum for effective communication lasting throughout the day.
We also added workstations on wheels (WOWS) to the rounding process so that labs, radiology, and more could be reviewed on rounds with the entire team. A standard script was developed so that each patient was introduced to all members of the team, and the care plan was disclosed and highlighted. One patient noted, “I feel so cared for, knowing I have this entire team taking care of me.” We also rounded in the afternoon with the case managers and social workers to follow up tasks that were to be completed that day.
Our first few weeks utilizing the geographic, unit-based model of rounding was largely successful. The residents, now able to round on all of their patients in one location with one case manager and one social worker, noted, “This model of rounding makes my life so much easier, I feel like I can focus on the patient rather than running around. … and I know the social worker and case manager will help me.”
Provider satisfaction had improved, from residents to physicians to nurses, case managers, social workers, and more. Our case manager also noted her satisfaction with the new model, stating that her communication with the medical team was much easier. As the attending, I witnessed firsthand how working together with the team moved care forward much more quickly, compared with the previous model, because of the simple factor of increased ease of communication.
Now all team members were together in the patient room and discussion was much easier. There was less confusion, fewer delays, and better communication – I think unit-based teams can even be described as a lifesaving measure that reduces harm to patients. An additional benefit is the relationship that now developed between doctors, social workers, and case managers – they spent more time together and really got to know one another, creating a feeling of shared success and a deeper drive to help one another succeed.
In our model, 87% of surveyed residents said they felt less burned out in the new geographic, unit-based model of care, and 91% of physicians surveyed said it was easier to talk with team members to coordinate care. Additionally, our HCAHPS (Hospital Consumer Assessment of Healthcare Providers and Systems) scores saw a drastic increase in many domains. Nursing communication improved by more than 42% on domain 7B; doctor communication improved by more than 31%. Additionally, all other domains saw at least 10% improvement. We are now 5 months out from our initial rollout of the model and continue to see sustained improvements in quality measures.
The bad
The biggest challenges that we are working through with this model are hand-offs and transfer of patients from one team to another. Sometimes, it happens that one team’s patient will wind up on a floor that is the designated floor of another team because of bed availability. We continue to work with bed board to address this issue. We want to minimize transfers and hand-offs to promote continuity and have to balance that with the need for geographic location. With clear communication, hospital collaboration from bed board and safe hand-off methods, this problem can be safely addressed.
Conclusions
The experience of implementing the unit-based team model has been an eye-opening journey. One thing that stands out is that, in an increasingly complex health care system, design thinking is critical.
Design thinking takes into consideration the needs of those who are using a system. In this case, patients and health care workers including doctors, nurses, case managers, and social workers are the end users of the health care system. All parties are utilizing the health care system to optimize patient health. Therefore, we must create systems that are easy to navigate and use by patients and health care workers so that they can ensure the success of patients.
Unit-based teams offer a basic framework to optimize the inpatient system to facilitate better workflow. In our system, it allowed us to optimize communications between health care workers and also between health care workers and patients. It allowed team members to work in close proximity to better share ideas with each other.
We spent a significant amount of time upfront earning the support of all of the disciplines for this effort. We had support from all leaders within the organization and continue to make our case for this model by sharing metrics and holding forums to discuss the process.
Initial data show a marked improvement in many domains of HCAHPS scores. Our frontline staff, including attendings, residents, nursing, case managers, and social workers, also continue to support this effort since it has a positive impact on their workflow and improves their workday quality. One nurse mentioned specifically, “in my 30 years at this hospital I have never seen people work together so well.”
To sustain this effort, we continue to have regular meetings, and there are new features that we would like to add to the program. For example, we are working with our IT group to ensure that each unit-based team will have dashboards available to incorporate real time, actionable data into daily workflows.
We are excited by the potential of our high-performing teams to highlight the patient experience, placing the patient at the center for care, decision making, and rounding. Health care is a team sport, and anytime you build something where all teams are playing together and approaching the finish line as a unit, you will never go wrong!
Dr. Pendharkar is division chief of hospital medicine at the Brooklyn (N.Y.) Hospital Center, medical director of inpatient services and director of quality for the department of medicine at the Brooklyn Hospital Center and assistant professor of medicine, Icahn School of Medicine at Mount Sinai, New York. Dr. Malieckal is chief resident, internal medicine, at the Brooklyn Hospital Center. Dr. Gasperino is chair, department of medicine; vice president for critical care, perioperative, and hospital medicine; and associate chief medical officer at the Brooklyn Hospital Center.
“To me, teamwork is the beauty of our sport, where you have five acting as one. You become selfless.” – Mike Krzyzewski
High-performing teams plan, communicate, reflect, and take action together. Teamwork can transform seemingly impossible tasks into opportunities for people to come together and create value.
The increasing complexity of health care makes team-based care necessary to achieve successful health outcomes for patients. At the Brooklyn (N.Y.) Hospital Center, a 464-bed care center, we transformed the model of care on the medical wards into a geographic, unit-based team model. Here we describe our journey – the successes, the challenges, and the opportunities for growth.
Previous model
In the previous care model on our medical wards, no set structures were in place. Teams would travel to multiple wards throughout the hospital to see the patients they were rounding on. Each floor had its own set of social workers and case managers, therefore a hospital medicine team routinely dealt with more than eight social workers and case managers to address their patients’ needs in a single day.
Multidisciplinary rounds for all medical patients were held at 11 a.m. in a room located a significant distance away from the medical wards. All case managers and social workers would sit in this room from 11 a.m. until noon, and teams would travel to that room to discuss their patients.
Many challenges were identified in this model, including a lack of communication, a de-emphasis on teamwork, and a design that did not take physician workflows into account resulting in low efficiency. Thus, these challenges sparked a desire to create a more effective and team-based methodology of accomplishing excellence in delivery of clinical care. Dr. Pendharkar, having worked primarily in centers with unit-based care, determined that a geographic, unit-based model of care could transform care delivery at the Brooklyn Hospital Center.
Looking ahead
The efforts for transforming the vision of geographic, unit-based teams into a reality started by gathering all stakeholders together to unite for a common mission. Initial meetings were held with all parties including social workers, case managers, residents, nursing staff, bed board and attending physicians in internal medicine, and the emergency department.
The vision of a geographic, unit-based team was shared and explained to all team members. Exercises in LEAN methodology were conducted, including one-piece flow exercises, to highlight the possibilities of what could be accomplished through teamwork. Once support for the vision was in place from all parties, the logistics were addressed.
The biggest challenge to overcome was how to place all of one team’s patients on a singular medical ward. In our hospital, a medical ward holds anywhere from 30 to 33 patients. Each hospital medicine team, of which there are many, typically carries 20-23 patients. We created a blueprint to map out the floor to which each team and attending would be assigned. Next, we partnered with both IT and bed board to design an admission order set that specified the particular geographic location that a team and attending were associated with so that patients could be placed accordingly from the ED.
It was important for the ED doctors, bed board, and the internal medicine residents to understand these changes because all of these parties were involved in the initial admitting process. Dr. Pendharkar and Dr. Malieckal provided all groups with in-person training on how the logistics of the system would unfold. Noon conference lectures were also held to explain the vision to residents.
Over 3 weeks, the first ward we chose to implement our model on slowly accumulated the patients of one team – this was the gradual trickle phase. We then selected a “re-set” date. On the re-set date, it was determined that all patients would go to the team that was assigned to that floor, with the exception of any private attendings’ patients.
On the day before the re-set date, time was spent ensuring that all hand-offs were safe. Dr. Pendharkar and Dr. Malieckal spoke with every intern and team that would be handing off and/or receiving patients as a result of the re-set policy. The goal was to ensure that on that date a ward had close to 100% of its patients belonging to the team/attending that was assigned to that area.
The good
Once we began our geographic, unit-based model, our rounding process was transformed.
Now, our morning rounds were joined by the bedside nurse, case manager, social worker, clinical pharmacy, and nutrition in addition to the core team. The entire team went from room to room on one ward rounding on all 20 to 25 patients back to back, which created an unparalleled level of efficiency and a forum for effective communication lasting throughout the day.
We also added workstations on wheels (WOWS) to the rounding process so that labs, radiology, and more could be reviewed on rounds with the entire team. A standard script was developed so that each patient was introduced to all members of the team, and the care plan was disclosed and highlighted. One patient noted, “I feel so cared for, knowing I have this entire team taking care of me.” We also rounded in the afternoon with the case managers and social workers to follow up tasks that were to be completed that day.
Our first few weeks utilizing the geographic, unit-based model of rounding was largely successful. The residents, now able to round on all of their patients in one location with one case manager and one social worker, noted, “This model of rounding makes my life so much easier, I feel like I can focus on the patient rather than running around. … and I know the social worker and case manager will help me.”
Provider satisfaction had improved, from residents to physicians to nurses, case managers, social workers, and more. Our case manager also noted her satisfaction with the new model, stating that her communication with the medical team was much easier. As the attending, I witnessed firsthand how working together with the team moved care forward much more quickly, compared with the previous model, because of the simple factor of increased ease of communication.
Now all team members were together in the patient room and discussion was much easier. There was less confusion, fewer delays, and better communication – I think unit-based teams can even be described as a lifesaving measure that reduces harm to patients. An additional benefit is the relationship that now developed between doctors, social workers, and case managers – they spent more time together and really got to know one another, creating a feeling of shared success and a deeper drive to help one another succeed.
In our model, 87% of surveyed residents said they felt less burned out in the new geographic, unit-based model of care, and 91% of physicians surveyed said it was easier to talk with team members to coordinate care. Additionally, our HCAHPS (Hospital Consumer Assessment of Healthcare Providers and Systems) scores saw a drastic increase in many domains. Nursing communication improved by more than 42% on domain 7B; doctor communication improved by more than 31%. Additionally, all other domains saw at least 10% improvement. We are now 5 months out from our initial rollout of the model and continue to see sustained improvements in quality measures.
The bad
The biggest challenges that we are working through with this model are hand-offs and transfer of patients from one team to another. Sometimes, it happens that one team’s patient will wind up on a floor that is the designated floor of another team because of bed availability. We continue to work with bed board to address this issue. We want to minimize transfers and hand-offs to promote continuity and have to balance that with the need for geographic location. With clear communication, hospital collaboration from bed board and safe hand-off methods, this problem can be safely addressed.
Conclusions
The experience of implementing the unit-based team model has been an eye-opening journey. One thing that stands out is that, in an increasingly complex health care system, design thinking is critical.
Design thinking takes into consideration the needs of those who are using a system. In this case, patients and health care workers including doctors, nurses, case managers, and social workers are the end users of the health care system. All parties are utilizing the health care system to optimize patient health. Therefore, we must create systems that are easy to navigate and use by patients and health care workers so that they can ensure the success of patients.
Unit-based teams offer a basic framework to optimize the inpatient system to facilitate better workflow. In our system, it allowed us to optimize communications between health care workers and also between health care workers and patients. It allowed team members to work in close proximity to better share ideas with each other.
We spent a significant amount of time upfront earning the support of all of the disciplines for this effort. We had support from all leaders within the organization and continue to make our case for this model by sharing metrics and holding forums to discuss the process.
Initial data show a marked improvement in many domains of HCAHPS scores. Our frontline staff, including attendings, residents, nursing, case managers, and social workers, also continue to support this effort since it has a positive impact on their workflow and improves their workday quality. One nurse mentioned specifically, “in my 30 years at this hospital I have never seen people work together so well.”
To sustain this effort, we continue to have regular meetings, and there are new features that we would like to add to the program. For example, we are working with our IT group to ensure that each unit-based team will have dashboards available to incorporate real time, actionable data into daily workflows.
We are excited by the potential of our high-performing teams to highlight the patient experience, placing the patient at the center for care, decision making, and rounding. Health care is a team sport, and anytime you build something where all teams are playing together and approaching the finish line as a unit, you will never go wrong!
Dr. Pendharkar is division chief of hospital medicine at the Brooklyn (N.Y.) Hospital Center, medical director of inpatient services and director of quality for the department of medicine at the Brooklyn Hospital Center and assistant professor of medicine, Icahn School of Medicine at Mount Sinai, New York. Dr. Malieckal is chief resident, internal medicine, at the Brooklyn Hospital Center. Dr. Gasperino is chair, department of medicine; vice president for critical care, perioperative, and hospital medicine; and associate chief medical officer at the Brooklyn Hospital Center.
Survey: High costs lead to skipped or postponed health care
Half of all Americans with employer-sponsored health benefits say that they or someone in their family has skipped or postponed care because of the cost, according to a survey by the Kaiser Family Foundation and the Los Angeles Times.
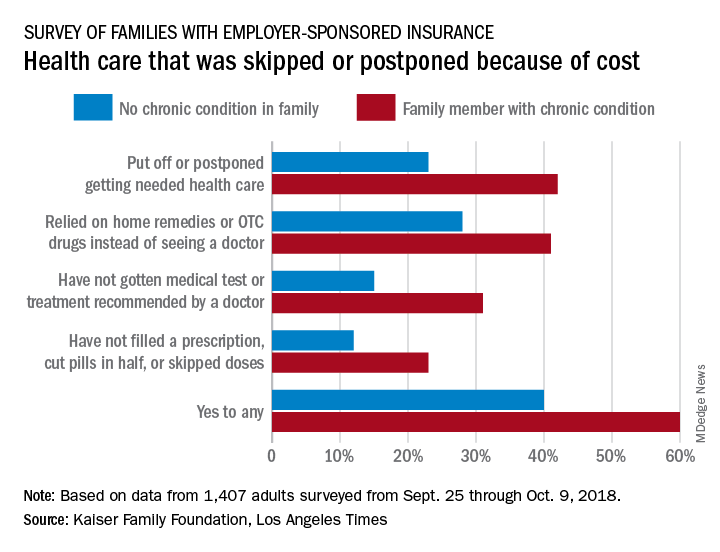
That number changes, however, when chronic conditions are considered. In the survey of Americans covered by employer-sponsored health insurance, 60% of those with a family member who had a chronic condition said that cost had altered the care of someone in the family over the previous 12 months, compared with 40% of those who had no chronic condition in their family, the KFF and L.A. Times noted in their report.
More specifically, families with an individual who had a chronic condition were more likely to put off or postpone needed care (42% vs. 23%) and to rely on home remedies or OTC drugs instead of visiting a physician (41% vs. 28%) than were families without chronic conditions, the report’s authors said.
When asked about the affordability of their health care, 49% of those in families with a chronic health condition said they had a problem paying for their coverage in the past year, compared with 29% of respondents in families with no chronic condition.
“Drilling down into the consequences of these affordability problems reveals more about the financial burden of health care on families with chronic conditions,” compared with those without chronic conditions: cut back spending on food, clothes, household items (35% vs. 16%); used up all or most of their savings (26% vs. 11%); and borrowed money from friends or family (14% vs. 6%), according to the researchers.
Although respondents felt “that the cost of health care for people like them is too high, more say the current U.S. health insurance system works well for people with employer coverage than say it works well for people on Medicare or Medicaid or those who purchase their own insurance. Asked who is to blame for high costs, majorities point the finger at pharmaceutical and insurance companies, while fewer see hospitals, doctors, or employers as deserving of blame,” the KFF and L.A. Times investigators wrote.
The survey involved a sample of 1,407 adults aged 18-64 years and was conducted from Sept. 25 through Oct. 9, 2018. The margin of the sampling error is ±3 percentage points.
Half of all Americans with employer-sponsored health benefits say that they or someone in their family has skipped or postponed care because of the cost, according to a survey by the Kaiser Family Foundation and the Los Angeles Times.

That number changes, however, when chronic conditions are considered. In the survey of Americans covered by employer-sponsored health insurance, 60% of those with a family member who had a chronic condition said that cost had altered the care of someone in the family over the previous 12 months, compared with 40% of those who had no chronic condition in their family, the KFF and L.A. Times noted in their report.
More specifically, families with an individual who had a chronic condition were more likely to put off or postpone needed care (42% vs. 23%) and to rely on home remedies or OTC drugs instead of visiting a physician (41% vs. 28%) than were families without chronic conditions, the report’s authors said.
When asked about the affordability of their health care, 49% of those in families with a chronic health condition said they had a problem paying for their coverage in the past year, compared with 29% of respondents in families with no chronic condition.
“Drilling down into the consequences of these affordability problems reveals more about the financial burden of health care on families with chronic conditions,” compared with those without chronic conditions: cut back spending on food, clothes, household items (35% vs. 16%); used up all or most of their savings (26% vs. 11%); and borrowed money from friends or family (14% vs. 6%), according to the researchers.
Although respondents felt “that the cost of health care for people like them is too high, more say the current U.S. health insurance system works well for people with employer coverage than say it works well for people on Medicare or Medicaid or those who purchase their own insurance. Asked who is to blame for high costs, majorities point the finger at pharmaceutical and insurance companies, while fewer see hospitals, doctors, or employers as deserving of blame,” the KFF and L.A. Times investigators wrote.
The survey involved a sample of 1,407 adults aged 18-64 years and was conducted from Sept. 25 through Oct. 9, 2018. The margin of the sampling error is ±3 percentage points.
Half of all Americans with employer-sponsored health benefits say that they or someone in their family has skipped or postponed care because of the cost, according to a survey by the Kaiser Family Foundation and the Los Angeles Times.

That number changes, however, when chronic conditions are considered. In the survey of Americans covered by employer-sponsored health insurance, 60% of those with a family member who had a chronic condition said that cost had altered the care of someone in the family over the previous 12 months, compared with 40% of those who had no chronic condition in their family, the KFF and L.A. Times noted in their report.
More specifically, families with an individual who had a chronic condition were more likely to put off or postpone needed care (42% vs. 23%) and to rely on home remedies or OTC drugs instead of visiting a physician (41% vs. 28%) than were families without chronic conditions, the report’s authors said.
When asked about the affordability of their health care, 49% of those in families with a chronic health condition said they had a problem paying for their coverage in the past year, compared with 29% of respondents in families with no chronic condition.
“Drilling down into the consequences of these affordability problems reveals more about the financial burden of health care on families with chronic conditions,” compared with those without chronic conditions: cut back spending on food, clothes, household items (35% vs. 16%); used up all or most of their savings (26% vs. 11%); and borrowed money from friends or family (14% vs. 6%), according to the researchers.
Although respondents felt “that the cost of health care for people like them is too high, more say the current U.S. health insurance system works well for people with employer coverage than say it works well for people on Medicare or Medicaid or those who purchase their own insurance. Asked who is to blame for high costs, majorities point the finger at pharmaceutical and insurance companies, while fewer see hospitals, doctors, or employers as deserving of blame,” the KFF and L.A. Times investigators wrote.
The survey involved a sample of 1,407 adults aged 18-64 years and was conducted from Sept. 25 through Oct. 9, 2018. The margin of the sampling error is ±3 percentage points.
CBO predicts more Medicare spending with drug rebate proposal
Medicare spending on pharmaceuticals is projected to increase if the Centers for Medicare & Medicaid Services finalizes changes to drug rebates in the Medicare program.
The Congressional Budget Office is estimating that Medicare spending would increase by $170 billion from 2020-2029 if the rebate rule goes into effect, according to a report released May 2.
The proposed rule, issued Jan. 31, would make it illegal for drug manufacturers to pay rebates to health plans and pharmacy benefit managers in return for better formulary placement. Instead of rebates, manufacturers could offer discounts directly to beneficiaries by lowering list prices or making a payment to the pharmacy for the full amount of the negotiated discount – a chargeback. Under the proposal, a beneficiary’s cost sharing would be based on the lower list price or the price after the chargeback.
The CBO’s projected spending increases are based on the assumption that manufacturers will withhold 15% of current-law rebates, as well as increases in federal subsidies for premiums, changes in annual thresholds to beneficiary cost sharing, and the cost of implementing the chargeback system.
The agency expects premiums to rise, as many plans currently use the rebates they receive from drug companies to lower premiums across the board.
However, some beneficiaries “would pay lower prices on their prescription drugs, and for some beneficiaries, those reductions would be greater than their premium increases,” the CBO stated in its report. For beneficiaries who use few drugs or who use drugs that have no significant rebates, “the premium increase would outweigh the price reduction.”
Another reason federal spending would increase under this proposal is an expected increase in utilization that would come with the lowering of prices.
“In CBO’s estimate, the additional Part D utilization stemming from implementing the proposed rule would increase federal spending for beneficiaries who are not enrolled in the low-income subsidy program over the 2020-2029 period by a total of about 2% or $10 billion,” the report noted.
But the increase in utilization would have a net positive effect on Medicare spending for this population, as more beneficiaries followed their drug regimens resulting in lower spending for physician and hospital services under Medicare Part A and Part B by an estimated $20 billion over the same period, according to the CBO.
“On net, those effects are projected to reduce Medicare spending by $10 billion over the 2020-2029 period,” according to the report.
Medicare spending on pharmaceuticals is projected to increase if the Centers for Medicare & Medicaid Services finalizes changes to drug rebates in the Medicare program.
The Congressional Budget Office is estimating that Medicare spending would increase by $170 billion from 2020-2029 if the rebate rule goes into effect, according to a report released May 2.
The proposed rule, issued Jan. 31, would make it illegal for drug manufacturers to pay rebates to health plans and pharmacy benefit managers in return for better formulary placement. Instead of rebates, manufacturers could offer discounts directly to beneficiaries by lowering list prices or making a payment to the pharmacy for the full amount of the negotiated discount – a chargeback. Under the proposal, a beneficiary’s cost sharing would be based on the lower list price or the price after the chargeback.
The CBO’s projected spending increases are based on the assumption that manufacturers will withhold 15% of current-law rebates, as well as increases in federal subsidies for premiums, changes in annual thresholds to beneficiary cost sharing, and the cost of implementing the chargeback system.
The agency expects premiums to rise, as many plans currently use the rebates they receive from drug companies to lower premiums across the board.
However, some beneficiaries “would pay lower prices on their prescription drugs, and for some beneficiaries, those reductions would be greater than their premium increases,” the CBO stated in its report. For beneficiaries who use few drugs or who use drugs that have no significant rebates, “the premium increase would outweigh the price reduction.”
Another reason federal spending would increase under this proposal is an expected increase in utilization that would come with the lowering of prices.
“In CBO’s estimate, the additional Part D utilization stemming from implementing the proposed rule would increase federal spending for beneficiaries who are not enrolled in the low-income subsidy program over the 2020-2029 period by a total of about 2% or $10 billion,” the report noted.
But the increase in utilization would have a net positive effect on Medicare spending for this population, as more beneficiaries followed their drug regimens resulting in lower spending for physician and hospital services under Medicare Part A and Part B by an estimated $20 billion over the same period, according to the CBO.
“On net, those effects are projected to reduce Medicare spending by $10 billion over the 2020-2029 period,” according to the report.
Medicare spending on pharmaceuticals is projected to increase if the Centers for Medicare & Medicaid Services finalizes changes to drug rebates in the Medicare program.
The Congressional Budget Office is estimating that Medicare spending would increase by $170 billion from 2020-2029 if the rebate rule goes into effect, according to a report released May 2.
The proposed rule, issued Jan. 31, would make it illegal for drug manufacturers to pay rebates to health plans and pharmacy benefit managers in return for better formulary placement. Instead of rebates, manufacturers could offer discounts directly to beneficiaries by lowering list prices or making a payment to the pharmacy for the full amount of the negotiated discount – a chargeback. Under the proposal, a beneficiary’s cost sharing would be based on the lower list price or the price after the chargeback.
The CBO’s projected spending increases are based on the assumption that manufacturers will withhold 15% of current-law rebates, as well as increases in federal subsidies for premiums, changes in annual thresholds to beneficiary cost sharing, and the cost of implementing the chargeback system.
The agency expects premiums to rise, as many plans currently use the rebates they receive from drug companies to lower premiums across the board.
However, some beneficiaries “would pay lower prices on their prescription drugs, and for some beneficiaries, those reductions would be greater than their premium increases,” the CBO stated in its report. For beneficiaries who use few drugs or who use drugs that have no significant rebates, “the premium increase would outweigh the price reduction.”
Another reason federal spending would increase under this proposal is an expected increase in utilization that would come with the lowering of prices.
“In CBO’s estimate, the additional Part D utilization stemming from implementing the proposed rule would increase federal spending for beneficiaries who are not enrolled in the low-income subsidy program over the 2020-2029 period by a total of about 2% or $10 billion,” the report noted.
But the increase in utilization would have a net positive effect on Medicare spending for this population, as more beneficiaries followed their drug regimens resulting in lower spending for physician and hospital services under Medicare Part A and Part B by an estimated $20 billion over the same period, according to the CBO.
“On net, those effects are projected to reduce Medicare spending by $10 billion over the 2020-2029 period,” according to the report.