User login
Is COVID-19 accelerating progress toward high-value care?
As Rachna Rawal, MD, was donning her personal protective equipment (PPE), a process that has become deeply ingrained into her muscle memory, a nurse approached her to ask, “Hey, for Mr. Smith, any chance we can time these labs to be done together with his medication administration? We’ve been in and out of that room a few times already.”
As someone who embraces high-value care, this simple suggestion surprised her. What an easy strategy to minimize room entry with full PPE, lab testing, and patient interruptions. That same day, someone else asked, “Do we need overnight vitals?”
COVID-19 has forced hospitalists to reconsider almost every aspect of care. It feels like every decision we make including things we do routinely – labs, vital signs, imaging – needs to be reassessed to determine the actual benefit to the patient balanced against concerns about staff safety, dwindling PPE supplies, and medication reserves. We are all faced with frequently answering the question, “How will this intervention help the patient?” This question lies at the heart of delivering high-value care.
High-value care is providing the best care possible through efficient use of resources, achieving optimal results for each patient. While high-value care has become a prominent focus over the past decade, COVID-19’s high transmissibility without a cure – and associated scarcity of health care resources – have sparked additional discussions on the front lines about promoting patient outcomes while avoiding waste. Clinicians may not have realized that these were high-value care conversations.
The United States’ health care quality and cost crises, worsened in the face of the current pandemic, have been glaringly apparent for years. Our country is spending more money on health care than anywhere else in the world without desired improvements in patient outcomes. A 2019 JAMA study found that 25% of all health care spending, an estimated $760 to $935 billion, is considered waste, and a significant proportion of this waste is due to repetitive care, overuse and unnecessary care in the U.S.1
Examples of low-value care tests include ordering daily labs in stable medicine inpatients, routine urine electrolytes in acute kidney injury, and folate testing in anemia. The Choosing Wisely® national campaign, Journal of Hospital Medicine’s “Things We Do For No Reason,” and JAMA Internal Medicine’s “Teachable Moment” series have provided guidance on areas where common testing or interventions may not benefit patient outcomes.
The COVID-19 pandemic has raised questions related to other widely-utilized practices: Can medication times be readjusted to allow only one entry into the room? Will these labs or imaging studies actually change management? Are vital checks every 4 hours needed?
Why did it take the COVID-19 threat to our medical system to force many of us to have these discussions? Despite prior efforts to integrate high-value care into hospital practices, long-standing habits and deep-seeded culture are challenging to overcome. Once clinicians develop practice habits, these behaviors tend to persist throughout their careers.2 In many ways, COVID-19 was like hitting a “reset button” as health care professionals were forced to rapidly confront their deeply-ingrained hospital practices and habits. From new protocols for patient rounding to universal masking and social distancing to ground-breaking strategies like awake proning, the response to COVID-19 has represented an unprecedented rapid shift in practice. Previously, consequences of overuse were too downstream or too abstract for clinicians to see in real-time. However, now the ramifications of these choices hit closer to home with obvious potential consequences – like spreading a terrifying virus.
There are three interventions that hospitalists should consider implementing immediately in the COVID-19 era that accelerate us toward high-value care. Routine lab tests, imaging, and overnight vitals represent opportunities to provide patient-centered care while also remaining cognizant of resource utilization.
One area in hospital medicine that has proven challenging to significantly change practice has been routine daily labs. Patients on a general medical inpatient service who are clinically stable generally do not benefit from routine lab work.3 Avoiding these tests does not increase mortality or length of stay in clinically stable patients.3 However, despite this evidence, many patients with COVID-19 and other conditions experience lab draws that are not timed together and are done each morning out of “routine.” Choosing Wisely® recommendations from the Society of Hospital Medicine encourage clinicians to question routine lab work for COVID-19 patients and to consider batching them, if possible.3,4 In COVID-19 patients, the risks of not batching tests are magnified, both in terms of the patient-centered experience and for clinician safety. In essence, COVID-19 has pushed us to consider the elements of safety, PPE conservation and other factors, rather than making decisions based solely on their own comfort, convenience, or historical practice.
Clinicians are also reconsidering the necessity of imaging during the pandemic. The “Things We Do For No Reason” article on “Choosing Wisely® in the COVID-19 era” highlights this well.4 It is more important now than ever to decide whether the timing and type of imaging will change management for your patient. Questions to ask include: Can a portable x-ray be used to avoid patient travel and will that CT scan help your patient? A posterior-anterior/lateral x-ray can potentially provide more information depending on the clinical scenario. However, we now need to assess if that extra information is going to impact patient management. Downstream consequences of these decisions include not only risks to the patient but also infectious exposures for staff and others during patient travel.
Lastly, overnight vital sign checks are another intervention we should analyze through this high-value care lens. The Journal of Hospital Medicine released a “Things We Do For No Reason” article about minimizing overnight vitals to promote uninterrupted sleep at night.5 Deleterious effects of interrupting the sleep of our patients include delirium and patient dissatisfaction.5 Studies have shown the benefits of this approach, yet the shift away from routine overnight vitals has not yet widely occurred.
COVID-19 has pressed us to save PPE and minimize exposure risk; hence, some centers are coordinating the timing of vitals with medication administration times, when feasible. In the stable patient recovering from COVID-19, overnight vitals may not be necessary, particularly if remote monitoring is available. This accomplishes multiple goals: Providing high quality patient care, reducing resource utilization, and minimizing patient nighttime interruptions – all culminating in high-value care.
Even though the COVID-19 pandemic has brought unforeseen emotional, physical, and financial challenges for the health care system and its workers, there may be a silver lining. The pandemic has sparked high-value care discussions, and the urgency of the crisis may be instilling new practices in our daily work. This virus has indeed left a terrible wake of destruction, but may also be a nudge to permanently change our culture of overuse to help us shape the habits of all trainees during this tumultuous time. This experience will hopefully culminate in a culture in which clinicians routinely ask, “How will this intervention help the patient?”
Dr. Rawal is clinical assistant professor of medicine, University of Pittsburgh. Dr. Linker is assistant professor of medicine, Mount Sinai Hospital, Icahn School of Medicine at Mount Sinai, New York. Dr. Moriates is associate professor of internal medicine, Dell Medical School at the University of Texas at Austin.
References
1. Shrank W et al. Waste in The US healthcare system. JAMA. 2019;322(15):1501-9.
2. Chen C et al. Spending patterns in region of residency training and subsequent expenditures for care provided by practicing physicians for Medicare beneficiaries. JAMA. 2014;312(22):2385-93.
3. Eaton KP et al. Evidence-based guidelines to eliminate repetitive laboratory testing. JAMA Intern Med. 2017;177(12):1833-9.
4. Cho H et al. Choosing Wisely in the COVID-19 Era: Preventing harm to healthcare workers. J Hosp Med. 2020;15(6):360-2.
5. Orlov N and Arora V. Things we do for no reason: Routine overnight vital sign checks. J Hosp Med. 2020;15(5):272-27.
As Rachna Rawal, MD, was donning her personal protective equipment (PPE), a process that has become deeply ingrained into her muscle memory, a nurse approached her to ask, “Hey, for Mr. Smith, any chance we can time these labs to be done together with his medication administration? We’ve been in and out of that room a few times already.”
As someone who embraces high-value care, this simple suggestion surprised her. What an easy strategy to minimize room entry with full PPE, lab testing, and patient interruptions. That same day, someone else asked, “Do we need overnight vitals?”
COVID-19 has forced hospitalists to reconsider almost every aspect of care. It feels like every decision we make including things we do routinely – labs, vital signs, imaging – needs to be reassessed to determine the actual benefit to the patient balanced against concerns about staff safety, dwindling PPE supplies, and medication reserves. We are all faced with frequently answering the question, “How will this intervention help the patient?” This question lies at the heart of delivering high-value care.
High-value care is providing the best care possible through efficient use of resources, achieving optimal results for each patient. While high-value care has become a prominent focus over the past decade, COVID-19’s high transmissibility without a cure – and associated scarcity of health care resources – have sparked additional discussions on the front lines about promoting patient outcomes while avoiding waste. Clinicians may not have realized that these were high-value care conversations.
The United States’ health care quality and cost crises, worsened in the face of the current pandemic, have been glaringly apparent for years. Our country is spending more money on health care than anywhere else in the world without desired improvements in patient outcomes. A 2019 JAMA study found that 25% of all health care spending, an estimated $760 to $935 billion, is considered waste, and a significant proportion of this waste is due to repetitive care, overuse and unnecessary care in the U.S.1
Examples of low-value care tests include ordering daily labs in stable medicine inpatients, routine urine electrolytes in acute kidney injury, and folate testing in anemia. The Choosing Wisely® national campaign, Journal of Hospital Medicine’s “Things We Do For No Reason,” and JAMA Internal Medicine’s “Teachable Moment” series have provided guidance on areas where common testing or interventions may not benefit patient outcomes.
The COVID-19 pandemic has raised questions related to other widely-utilized practices: Can medication times be readjusted to allow only one entry into the room? Will these labs or imaging studies actually change management? Are vital checks every 4 hours needed?
Why did it take the COVID-19 threat to our medical system to force many of us to have these discussions? Despite prior efforts to integrate high-value care into hospital practices, long-standing habits and deep-seeded culture are challenging to overcome. Once clinicians develop practice habits, these behaviors tend to persist throughout their careers.2 In many ways, COVID-19 was like hitting a “reset button” as health care professionals were forced to rapidly confront their deeply-ingrained hospital practices and habits. From new protocols for patient rounding to universal masking and social distancing to ground-breaking strategies like awake proning, the response to COVID-19 has represented an unprecedented rapid shift in practice. Previously, consequences of overuse were too downstream or too abstract for clinicians to see in real-time. However, now the ramifications of these choices hit closer to home with obvious potential consequences – like spreading a terrifying virus.
There are three interventions that hospitalists should consider implementing immediately in the COVID-19 era that accelerate us toward high-value care. Routine lab tests, imaging, and overnight vitals represent opportunities to provide patient-centered care while also remaining cognizant of resource utilization.
One area in hospital medicine that has proven challenging to significantly change practice has been routine daily labs. Patients on a general medical inpatient service who are clinically stable generally do not benefit from routine lab work.3 Avoiding these tests does not increase mortality or length of stay in clinically stable patients.3 However, despite this evidence, many patients with COVID-19 and other conditions experience lab draws that are not timed together and are done each morning out of “routine.” Choosing Wisely® recommendations from the Society of Hospital Medicine encourage clinicians to question routine lab work for COVID-19 patients and to consider batching them, if possible.3,4 In COVID-19 patients, the risks of not batching tests are magnified, both in terms of the patient-centered experience and for clinician safety. In essence, COVID-19 has pushed us to consider the elements of safety, PPE conservation and other factors, rather than making decisions based solely on their own comfort, convenience, or historical practice.
Clinicians are also reconsidering the necessity of imaging during the pandemic. The “Things We Do For No Reason” article on “Choosing Wisely® in the COVID-19 era” highlights this well.4 It is more important now than ever to decide whether the timing and type of imaging will change management for your patient. Questions to ask include: Can a portable x-ray be used to avoid patient travel and will that CT scan help your patient? A posterior-anterior/lateral x-ray can potentially provide more information depending on the clinical scenario. However, we now need to assess if that extra information is going to impact patient management. Downstream consequences of these decisions include not only risks to the patient but also infectious exposures for staff and others during patient travel.
Lastly, overnight vital sign checks are another intervention we should analyze through this high-value care lens. The Journal of Hospital Medicine released a “Things We Do For No Reason” article about minimizing overnight vitals to promote uninterrupted sleep at night.5 Deleterious effects of interrupting the sleep of our patients include delirium and patient dissatisfaction.5 Studies have shown the benefits of this approach, yet the shift away from routine overnight vitals has not yet widely occurred.
COVID-19 has pressed us to save PPE and minimize exposure risk; hence, some centers are coordinating the timing of vitals with medication administration times, when feasible. In the stable patient recovering from COVID-19, overnight vitals may not be necessary, particularly if remote monitoring is available. This accomplishes multiple goals: Providing high quality patient care, reducing resource utilization, and minimizing patient nighttime interruptions – all culminating in high-value care.
Even though the COVID-19 pandemic has brought unforeseen emotional, physical, and financial challenges for the health care system and its workers, there may be a silver lining. The pandemic has sparked high-value care discussions, and the urgency of the crisis may be instilling new practices in our daily work. This virus has indeed left a terrible wake of destruction, but may also be a nudge to permanently change our culture of overuse to help us shape the habits of all trainees during this tumultuous time. This experience will hopefully culminate in a culture in which clinicians routinely ask, “How will this intervention help the patient?”
Dr. Rawal is clinical assistant professor of medicine, University of Pittsburgh. Dr. Linker is assistant professor of medicine, Mount Sinai Hospital, Icahn School of Medicine at Mount Sinai, New York. Dr. Moriates is associate professor of internal medicine, Dell Medical School at the University of Texas at Austin.
References
1. Shrank W et al. Waste in The US healthcare system. JAMA. 2019;322(15):1501-9.
2. Chen C et al. Spending patterns in region of residency training and subsequent expenditures for care provided by practicing physicians for Medicare beneficiaries. JAMA. 2014;312(22):2385-93.
3. Eaton KP et al. Evidence-based guidelines to eliminate repetitive laboratory testing. JAMA Intern Med. 2017;177(12):1833-9.
4. Cho H et al. Choosing Wisely in the COVID-19 Era: Preventing harm to healthcare workers. J Hosp Med. 2020;15(6):360-2.
5. Orlov N and Arora V. Things we do for no reason: Routine overnight vital sign checks. J Hosp Med. 2020;15(5):272-27.
As Rachna Rawal, MD, was donning her personal protective equipment (PPE), a process that has become deeply ingrained into her muscle memory, a nurse approached her to ask, “Hey, for Mr. Smith, any chance we can time these labs to be done together with his medication administration? We’ve been in and out of that room a few times already.”
As someone who embraces high-value care, this simple suggestion surprised her. What an easy strategy to minimize room entry with full PPE, lab testing, and patient interruptions. That same day, someone else asked, “Do we need overnight vitals?”
COVID-19 has forced hospitalists to reconsider almost every aspect of care. It feels like every decision we make including things we do routinely – labs, vital signs, imaging – needs to be reassessed to determine the actual benefit to the patient balanced against concerns about staff safety, dwindling PPE supplies, and medication reserves. We are all faced with frequently answering the question, “How will this intervention help the patient?” This question lies at the heart of delivering high-value care.
High-value care is providing the best care possible through efficient use of resources, achieving optimal results for each patient. While high-value care has become a prominent focus over the past decade, COVID-19’s high transmissibility without a cure – and associated scarcity of health care resources – have sparked additional discussions on the front lines about promoting patient outcomes while avoiding waste. Clinicians may not have realized that these were high-value care conversations.
The United States’ health care quality and cost crises, worsened in the face of the current pandemic, have been glaringly apparent for years. Our country is spending more money on health care than anywhere else in the world without desired improvements in patient outcomes. A 2019 JAMA study found that 25% of all health care spending, an estimated $760 to $935 billion, is considered waste, and a significant proportion of this waste is due to repetitive care, overuse and unnecessary care in the U.S.1
Examples of low-value care tests include ordering daily labs in stable medicine inpatients, routine urine electrolytes in acute kidney injury, and folate testing in anemia. The Choosing Wisely® national campaign, Journal of Hospital Medicine’s “Things We Do For No Reason,” and JAMA Internal Medicine’s “Teachable Moment” series have provided guidance on areas where common testing or interventions may not benefit patient outcomes.
The COVID-19 pandemic has raised questions related to other widely-utilized practices: Can medication times be readjusted to allow only one entry into the room? Will these labs or imaging studies actually change management? Are vital checks every 4 hours needed?
Why did it take the COVID-19 threat to our medical system to force many of us to have these discussions? Despite prior efforts to integrate high-value care into hospital practices, long-standing habits and deep-seeded culture are challenging to overcome. Once clinicians develop practice habits, these behaviors tend to persist throughout their careers.2 In many ways, COVID-19 was like hitting a “reset button” as health care professionals were forced to rapidly confront their deeply-ingrained hospital practices and habits. From new protocols for patient rounding to universal masking and social distancing to ground-breaking strategies like awake proning, the response to COVID-19 has represented an unprecedented rapid shift in practice. Previously, consequences of overuse were too downstream or too abstract for clinicians to see in real-time. However, now the ramifications of these choices hit closer to home with obvious potential consequences – like spreading a terrifying virus.
There are three interventions that hospitalists should consider implementing immediately in the COVID-19 era that accelerate us toward high-value care. Routine lab tests, imaging, and overnight vitals represent opportunities to provide patient-centered care while also remaining cognizant of resource utilization.
One area in hospital medicine that has proven challenging to significantly change practice has been routine daily labs. Patients on a general medical inpatient service who are clinically stable generally do not benefit from routine lab work.3 Avoiding these tests does not increase mortality or length of stay in clinically stable patients.3 However, despite this evidence, many patients with COVID-19 and other conditions experience lab draws that are not timed together and are done each morning out of “routine.” Choosing Wisely® recommendations from the Society of Hospital Medicine encourage clinicians to question routine lab work for COVID-19 patients and to consider batching them, if possible.3,4 In COVID-19 patients, the risks of not batching tests are magnified, both in terms of the patient-centered experience and for clinician safety. In essence, COVID-19 has pushed us to consider the elements of safety, PPE conservation and other factors, rather than making decisions based solely on their own comfort, convenience, or historical practice.
Clinicians are also reconsidering the necessity of imaging during the pandemic. The “Things We Do For No Reason” article on “Choosing Wisely® in the COVID-19 era” highlights this well.4 It is more important now than ever to decide whether the timing and type of imaging will change management for your patient. Questions to ask include: Can a portable x-ray be used to avoid patient travel and will that CT scan help your patient? A posterior-anterior/lateral x-ray can potentially provide more information depending on the clinical scenario. However, we now need to assess if that extra information is going to impact patient management. Downstream consequences of these decisions include not only risks to the patient but also infectious exposures for staff and others during patient travel.
Lastly, overnight vital sign checks are another intervention we should analyze through this high-value care lens. The Journal of Hospital Medicine released a “Things We Do For No Reason” article about minimizing overnight vitals to promote uninterrupted sleep at night.5 Deleterious effects of interrupting the sleep of our patients include delirium and patient dissatisfaction.5 Studies have shown the benefits of this approach, yet the shift away from routine overnight vitals has not yet widely occurred.
COVID-19 has pressed us to save PPE and minimize exposure risk; hence, some centers are coordinating the timing of vitals with medication administration times, when feasible. In the stable patient recovering from COVID-19, overnight vitals may not be necessary, particularly if remote monitoring is available. This accomplishes multiple goals: Providing high quality patient care, reducing resource utilization, and minimizing patient nighttime interruptions – all culminating in high-value care.
Even though the COVID-19 pandemic has brought unforeseen emotional, physical, and financial challenges for the health care system and its workers, there may be a silver lining. The pandemic has sparked high-value care discussions, and the urgency of the crisis may be instilling new practices in our daily work. This virus has indeed left a terrible wake of destruction, but may also be a nudge to permanently change our culture of overuse to help us shape the habits of all trainees during this tumultuous time. This experience will hopefully culminate in a culture in which clinicians routinely ask, “How will this intervention help the patient?”
Dr. Rawal is clinical assistant professor of medicine, University of Pittsburgh. Dr. Linker is assistant professor of medicine, Mount Sinai Hospital, Icahn School of Medicine at Mount Sinai, New York. Dr. Moriates is associate professor of internal medicine, Dell Medical School at the University of Texas at Austin.
References
1. Shrank W et al. Waste in The US healthcare system. JAMA. 2019;322(15):1501-9.
2. Chen C et al. Spending patterns in region of residency training and subsequent expenditures for care provided by practicing physicians for Medicare beneficiaries. JAMA. 2014;312(22):2385-93.
3. Eaton KP et al. Evidence-based guidelines to eliminate repetitive laboratory testing. JAMA Intern Med. 2017;177(12):1833-9.
4. Cho H et al. Choosing Wisely in the COVID-19 Era: Preventing harm to healthcare workers. J Hosp Med. 2020;15(6):360-2.
5. Orlov N and Arora V. Things we do for no reason: Routine overnight vital sign checks. J Hosp Med. 2020;15(5):272-27.
Some COVID-19 vaccine reactions could be pseudoallergic, experts say
On Jan. 13, 2 days after a drive-through vaccination “superstation” opened in San Diego, six people were treated for anaphylaxis after they received the Moderna vaccine, leading the California state epidemiologist to recommend pausing the administration of that particular lot.
A group of allergy and immunology experts and public health officials reviewed the cases, as well as an incident that occurred the day before, and concluded that at least some of the responses were angioedema, or swelling — a serious allergic reaction — but none were actually anaphylaxis. No similar clusters had occurred with the same vaccine lot in other states, and California resumed using the doses.
Yet questions remain about the reactions and the mechanisms for them. Some might have been triggered by an allergy to a vaccine component, most likely the polyethylene glycol (PEG) that stabilizes the lipid surrounding the mRNA, the key vaccine component in both the Moderna and Pfizer vaccines. Another possible explanation is that some could be pseudoallergic reactions to a blood protein known as complement, a little-understood process that resembles an antigen-based reaction but doesn’t leave an immune memory and might not recur.
Cases of complement-activation-related pseudoallergy look like a severe allergic reaction but occur through a different mechanism and don’t require previous exposure to an allergen.
“It has the same signs and symptoms and is treated the same way, but it occurs through a different pathway,” explained Neal Halsey, MD, director emeritus of the Institute for Vaccine Safety and emeritus professor at the Johns Hopkins Bloomberg School of Public Health in Baltimore.
Pseudoallergies are not well understood, but they have been associated with reactions to the contrast media used in imaging, such as with MRI. “If people have had an anaphylaxis-type reaction following the injection of contrast-dye material, that is a strong signal that it might be a complement-activation-related pseudoallergy,” said Dr. Halsey, a member of the Clinical Immunization Safety Assessment Network. “Those are the people who definitely need to consider seeing an allergist before getting the COVID vaccines.”
When Aleena Banerji, MD, clinical director of the allergy and clinical immunology unit at Massachusetts General Hospital in Boston, talks to patients about vaccine reactions, she addresses the risk for COVID-19 infection. All of the people who developed allergies after the Pfizer and Moderna vaccines recovered, but more than 445,000 Americans have died from COVID-19.
Most people with common allergies, such as to food or oral medications, don’t need to worry about reactions, said Dr. Banerji, lead author of a review that assessed the risk for allergic reactions to the Pfizer and Moderna vaccines.
Investigating reactions
As investigators search for the answers to what causes reactions, transparency is crucial to trust, said Kathryn Edwards, MD, principal investigator of the Clinical Immunization Safety Assessment Project, a vaccine safety network funded by the Centers for Disease Control and Prevention.
“Unless the public knows that we’re really investigating and we’re taking this seriously, then I think the vaccine hesitancy is going to increase,” said Dr. Edwards, professor of pediatrics at Vanderbilt University Medical Center and scientific director of the Vanderbilt Vaccine Research Program in Nashville, Tenn.
First reports of anaphylaxis came quickly after COVID-19 vaccinations began. In the 2 weeks before the holidays, almost 2 million health care workers received the Pfizer vaccine, and 21 of them developed anaphylaxis, according to CDC researchers who reviewed case reports from the Vaccine Adverse Event Reporting System (VAERS). That rate of about 1 in 100,000 is 10 times higher than the occurrence with other vaccines. No deaths from anaphylaxis were reported.
As the vaccinations ramped up, the rate declined. As of Jan. 18, 50 cases of anaphylaxis were reported to VAERS after the administration of 9,943,247 Pfizer doses, for a rate of 5.0 per million, according to data presented at the Jan. 27 meeting of the CDC Advisory Committee on Immunization Practices. And 21 cases of anaphylaxis were reported to VAERS after the administration of 7,581,429 Moderna doses, for a rate of 2.8 per million.
The anaphylaxis occurred almost exclusively in women; only three of the VAERS anaphylaxis reports were from men. Only 24% had a history of anaphylaxis.
The earlier CDC report explored the potential link to allergies. One person with anaphylaxis had a history of allergy to iodinated contrast media, and others had allergies to various medications, vaccines, foods, and animals. The researchers reported 86 nonanaphylaxis allergic reactions and 61 nonallergic adverse events among the 175 case reports they reviewed as possible cases of severe allergic reaction.
Of 1,266 reports that VAERS received from Dec. 21 to Jan. 10, the CDC identified 108 possible cases of severe allergic reaction after the Moderna vaccine. Only 10 met the case definition of anaphylaxis put forward by the Brighton Collaboration, a vaccine safety organization. All but one case involved a history of allergies or allergic reactions; only five had a previously experienced anaphylaxis.
There were 47 nonanaphylaxis allergic reactions.
The San Diego cluster also met the Brighton case definition for anaphylaxis, Dr. Edwards reported. This discrepancy highlights the difficulties in characterizing vaccine reactions.
Measuring a pseudoallergic reaction is a challenge. It requires that a blood sample be drawn soon after the incident and then frozen to protect heat-sensitive blood markers, Dr. Edwards explained.
And as vaccinations rise, so do adverse-event reports. But unlike in clinical trials, there is no control group for comparison. That is why vaccine safety experts urge caution when evaluating events and, where possible, advise looking at background rates.
“A major way to determine whether the adverse event is causally related is to assess the incidence of the adverse event in vaccines versus nonvaccines,” said Walter Orenstein, MD, who directed the U.S. Immunization Program from 1988 to 2004 and is now associate director of the Emory Vaccine Center and professor of infectious diseases at Emory University in Atlanta. Public health officials could then identify vaccine risk factors, he said.
When a reaction occurs almost immediately after vaccination, vaccine safety investigators look for probable triggers. If allergy to PEG is the culprit in anaphylactic reactions, then the individuals would have had a previous exposure, perhaps from injectable medications, Dr. Edwards said.
It might be feasible to perform a skin test for allergy to PEG. “If the skin testing is negative, that doesn’t completely rule out allergy, but it can be used in the decision-making about giving the first or second vaccine dose,” Dr. Banerji said.
Other vaccines, such as childhood vaccines, contain polysorbate as a stabilizer, which has a similar chemical structure, and it’s not clear why someone would react to PEG but not to polysorbate, Dr. Edwards said.
Meanwhile, other illnesses and even deaths sometimes occur in the days after vaccination, but that doesn’t mean the vaccine caused them, cautioned Steve Black, MD, emeritus professor of pediatrics at Cincinnati Children’s Hospital and cofounder of the Global Vaccine Data Network, an international vaccine safety collaboration.
“Different events and clusters of events will occur by chance alone, as these events can occur without vaccines. We need to not immediately assume that they’re due to the vaccine,” he said. “You don’t want to undermine the whole vaccine program every time something comes up and assume that it’s associated with the vaccine.”
The CDC only has three contraindications for the vaccines:
- Severe allergic reaction (such as anaphylaxis) after a previous dose of an mRNA COVID-19 vaccine or any of its components.
- Immediate allergic reaction of any severity to a previous dose of an mRNA COVID-19 vaccine or any of its components (including PEG).
- Immediate allergic reaction of any severity to polysorbate (due to potential cross-reactive hypersensitivity with PEG).
People who have had an immediate allergic reaction to other vaccines or injectable therapies should consider consulting with an allergist or immunologist before getting the Pfizer or Moderna vaccines, the CDC advises.
The CDC also says that people with a history of anaphylaxis from any cause should be observed for 30 minutes after vaccination. Vaccination protocol calls for everyone else to wait on site for 15 minutes after vaccination.
A version of this article first appeared on Medscape.com.
On Jan. 13, 2 days after a drive-through vaccination “superstation” opened in San Diego, six people were treated for anaphylaxis after they received the Moderna vaccine, leading the California state epidemiologist to recommend pausing the administration of that particular lot.
A group of allergy and immunology experts and public health officials reviewed the cases, as well as an incident that occurred the day before, and concluded that at least some of the responses were angioedema, or swelling — a serious allergic reaction — but none were actually anaphylaxis. No similar clusters had occurred with the same vaccine lot in other states, and California resumed using the doses.
Yet questions remain about the reactions and the mechanisms for them. Some might have been triggered by an allergy to a vaccine component, most likely the polyethylene glycol (PEG) that stabilizes the lipid surrounding the mRNA, the key vaccine component in both the Moderna and Pfizer vaccines. Another possible explanation is that some could be pseudoallergic reactions to a blood protein known as complement, a little-understood process that resembles an antigen-based reaction but doesn’t leave an immune memory and might not recur.
Cases of complement-activation-related pseudoallergy look like a severe allergic reaction but occur through a different mechanism and don’t require previous exposure to an allergen.
“It has the same signs and symptoms and is treated the same way, but it occurs through a different pathway,” explained Neal Halsey, MD, director emeritus of the Institute for Vaccine Safety and emeritus professor at the Johns Hopkins Bloomberg School of Public Health in Baltimore.
Pseudoallergies are not well understood, but they have been associated with reactions to the contrast media used in imaging, such as with MRI. “If people have had an anaphylaxis-type reaction following the injection of contrast-dye material, that is a strong signal that it might be a complement-activation-related pseudoallergy,” said Dr. Halsey, a member of the Clinical Immunization Safety Assessment Network. “Those are the people who definitely need to consider seeing an allergist before getting the COVID vaccines.”
When Aleena Banerji, MD, clinical director of the allergy and clinical immunology unit at Massachusetts General Hospital in Boston, talks to patients about vaccine reactions, she addresses the risk for COVID-19 infection. All of the people who developed allergies after the Pfizer and Moderna vaccines recovered, but more than 445,000 Americans have died from COVID-19.
Most people with common allergies, such as to food or oral medications, don’t need to worry about reactions, said Dr. Banerji, lead author of a review that assessed the risk for allergic reactions to the Pfizer and Moderna vaccines.
Investigating reactions
As investigators search for the answers to what causes reactions, transparency is crucial to trust, said Kathryn Edwards, MD, principal investigator of the Clinical Immunization Safety Assessment Project, a vaccine safety network funded by the Centers for Disease Control and Prevention.
“Unless the public knows that we’re really investigating and we’re taking this seriously, then I think the vaccine hesitancy is going to increase,” said Dr. Edwards, professor of pediatrics at Vanderbilt University Medical Center and scientific director of the Vanderbilt Vaccine Research Program in Nashville, Tenn.
First reports of anaphylaxis came quickly after COVID-19 vaccinations began. In the 2 weeks before the holidays, almost 2 million health care workers received the Pfizer vaccine, and 21 of them developed anaphylaxis, according to CDC researchers who reviewed case reports from the Vaccine Adverse Event Reporting System (VAERS). That rate of about 1 in 100,000 is 10 times higher than the occurrence with other vaccines. No deaths from anaphylaxis were reported.
As the vaccinations ramped up, the rate declined. As of Jan. 18, 50 cases of anaphylaxis were reported to VAERS after the administration of 9,943,247 Pfizer doses, for a rate of 5.0 per million, according to data presented at the Jan. 27 meeting of the CDC Advisory Committee on Immunization Practices. And 21 cases of anaphylaxis were reported to VAERS after the administration of 7,581,429 Moderna doses, for a rate of 2.8 per million.
The anaphylaxis occurred almost exclusively in women; only three of the VAERS anaphylaxis reports were from men. Only 24% had a history of anaphylaxis.
The earlier CDC report explored the potential link to allergies. One person with anaphylaxis had a history of allergy to iodinated contrast media, and others had allergies to various medications, vaccines, foods, and animals. The researchers reported 86 nonanaphylaxis allergic reactions and 61 nonallergic adverse events among the 175 case reports they reviewed as possible cases of severe allergic reaction.
Of 1,266 reports that VAERS received from Dec. 21 to Jan. 10, the CDC identified 108 possible cases of severe allergic reaction after the Moderna vaccine. Only 10 met the case definition of anaphylaxis put forward by the Brighton Collaboration, a vaccine safety organization. All but one case involved a history of allergies or allergic reactions; only five had a previously experienced anaphylaxis.
There were 47 nonanaphylaxis allergic reactions.
The San Diego cluster also met the Brighton case definition for anaphylaxis, Dr. Edwards reported. This discrepancy highlights the difficulties in characterizing vaccine reactions.
Measuring a pseudoallergic reaction is a challenge. It requires that a blood sample be drawn soon after the incident and then frozen to protect heat-sensitive blood markers, Dr. Edwards explained.
And as vaccinations rise, so do adverse-event reports. But unlike in clinical trials, there is no control group for comparison. That is why vaccine safety experts urge caution when evaluating events and, where possible, advise looking at background rates.
“A major way to determine whether the adverse event is causally related is to assess the incidence of the adverse event in vaccines versus nonvaccines,” said Walter Orenstein, MD, who directed the U.S. Immunization Program from 1988 to 2004 and is now associate director of the Emory Vaccine Center and professor of infectious diseases at Emory University in Atlanta. Public health officials could then identify vaccine risk factors, he said.
When a reaction occurs almost immediately after vaccination, vaccine safety investigators look for probable triggers. If allergy to PEG is the culprit in anaphylactic reactions, then the individuals would have had a previous exposure, perhaps from injectable medications, Dr. Edwards said.
It might be feasible to perform a skin test for allergy to PEG. “If the skin testing is negative, that doesn’t completely rule out allergy, but it can be used in the decision-making about giving the first or second vaccine dose,” Dr. Banerji said.
Other vaccines, such as childhood vaccines, contain polysorbate as a stabilizer, which has a similar chemical structure, and it’s not clear why someone would react to PEG but not to polysorbate, Dr. Edwards said.
Meanwhile, other illnesses and even deaths sometimes occur in the days after vaccination, but that doesn’t mean the vaccine caused them, cautioned Steve Black, MD, emeritus professor of pediatrics at Cincinnati Children’s Hospital and cofounder of the Global Vaccine Data Network, an international vaccine safety collaboration.
“Different events and clusters of events will occur by chance alone, as these events can occur without vaccines. We need to not immediately assume that they’re due to the vaccine,” he said. “You don’t want to undermine the whole vaccine program every time something comes up and assume that it’s associated with the vaccine.”
The CDC only has three contraindications for the vaccines:
- Severe allergic reaction (such as anaphylaxis) after a previous dose of an mRNA COVID-19 vaccine or any of its components.
- Immediate allergic reaction of any severity to a previous dose of an mRNA COVID-19 vaccine or any of its components (including PEG).
- Immediate allergic reaction of any severity to polysorbate (due to potential cross-reactive hypersensitivity with PEG).
People who have had an immediate allergic reaction to other vaccines or injectable therapies should consider consulting with an allergist or immunologist before getting the Pfizer or Moderna vaccines, the CDC advises.
The CDC also says that people with a history of anaphylaxis from any cause should be observed for 30 minutes after vaccination. Vaccination protocol calls for everyone else to wait on site for 15 minutes after vaccination.
A version of this article first appeared on Medscape.com.
On Jan. 13, 2 days after a drive-through vaccination “superstation” opened in San Diego, six people were treated for anaphylaxis after they received the Moderna vaccine, leading the California state epidemiologist to recommend pausing the administration of that particular lot.
A group of allergy and immunology experts and public health officials reviewed the cases, as well as an incident that occurred the day before, and concluded that at least some of the responses were angioedema, or swelling — a serious allergic reaction — but none were actually anaphylaxis. No similar clusters had occurred with the same vaccine lot in other states, and California resumed using the doses.
Yet questions remain about the reactions and the mechanisms for them. Some might have been triggered by an allergy to a vaccine component, most likely the polyethylene glycol (PEG) that stabilizes the lipid surrounding the mRNA, the key vaccine component in both the Moderna and Pfizer vaccines. Another possible explanation is that some could be pseudoallergic reactions to a blood protein known as complement, a little-understood process that resembles an antigen-based reaction but doesn’t leave an immune memory and might not recur.
Cases of complement-activation-related pseudoallergy look like a severe allergic reaction but occur through a different mechanism and don’t require previous exposure to an allergen.
“It has the same signs and symptoms and is treated the same way, but it occurs through a different pathway,” explained Neal Halsey, MD, director emeritus of the Institute for Vaccine Safety and emeritus professor at the Johns Hopkins Bloomberg School of Public Health in Baltimore.
Pseudoallergies are not well understood, but they have been associated with reactions to the contrast media used in imaging, such as with MRI. “If people have had an anaphylaxis-type reaction following the injection of contrast-dye material, that is a strong signal that it might be a complement-activation-related pseudoallergy,” said Dr. Halsey, a member of the Clinical Immunization Safety Assessment Network. “Those are the people who definitely need to consider seeing an allergist before getting the COVID vaccines.”
When Aleena Banerji, MD, clinical director of the allergy and clinical immunology unit at Massachusetts General Hospital in Boston, talks to patients about vaccine reactions, she addresses the risk for COVID-19 infection. All of the people who developed allergies after the Pfizer and Moderna vaccines recovered, but more than 445,000 Americans have died from COVID-19.
Most people with common allergies, such as to food or oral medications, don’t need to worry about reactions, said Dr. Banerji, lead author of a review that assessed the risk for allergic reactions to the Pfizer and Moderna vaccines.
Investigating reactions
As investigators search for the answers to what causes reactions, transparency is crucial to trust, said Kathryn Edwards, MD, principal investigator of the Clinical Immunization Safety Assessment Project, a vaccine safety network funded by the Centers for Disease Control and Prevention.
“Unless the public knows that we’re really investigating and we’re taking this seriously, then I think the vaccine hesitancy is going to increase,” said Dr. Edwards, professor of pediatrics at Vanderbilt University Medical Center and scientific director of the Vanderbilt Vaccine Research Program in Nashville, Tenn.
First reports of anaphylaxis came quickly after COVID-19 vaccinations began. In the 2 weeks before the holidays, almost 2 million health care workers received the Pfizer vaccine, and 21 of them developed anaphylaxis, according to CDC researchers who reviewed case reports from the Vaccine Adverse Event Reporting System (VAERS). That rate of about 1 in 100,000 is 10 times higher than the occurrence with other vaccines. No deaths from anaphylaxis were reported.
As the vaccinations ramped up, the rate declined. As of Jan. 18, 50 cases of anaphylaxis were reported to VAERS after the administration of 9,943,247 Pfizer doses, for a rate of 5.0 per million, according to data presented at the Jan. 27 meeting of the CDC Advisory Committee on Immunization Practices. And 21 cases of anaphylaxis were reported to VAERS after the administration of 7,581,429 Moderna doses, for a rate of 2.8 per million.
The anaphylaxis occurred almost exclusively in women; only three of the VAERS anaphylaxis reports were from men. Only 24% had a history of anaphylaxis.
The earlier CDC report explored the potential link to allergies. One person with anaphylaxis had a history of allergy to iodinated contrast media, and others had allergies to various medications, vaccines, foods, and animals. The researchers reported 86 nonanaphylaxis allergic reactions and 61 nonallergic adverse events among the 175 case reports they reviewed as possible cases of severe allergic reaction.
Of 1,266 reports that VAERS received from Dec. 21 to Jan. 10, the CDC identified 108 possible cases of severe allergic reaction after the Moderna vaccine. Only 10 met the case definition of anaphylaxis put forward by the Brighton Collaboration, a vaccine safety organization. All but one case involved a history of allergies or allergic reactions; only five had a previously experienced anaphylaxis.
There were 47 nonanaphylaxis allergic reactions.
The San Diego cluster also met the Brighton case definition for anaphylaxis, Dr. Edwards reported. This discrepancy highlights the difficulties in characterizing vaccine reactions.
Measuring a pseudoallergic reaction is a challenge. It requires that a blood sample be drawn soon after the incident and then frozen to protect heat-sensitive blood markers, Dr. Edwards explained.
And as vaccinations rise, so do adverse-event reports. But unlike in clinical trials, there is no control group for comparison. That is why vaccine safety experts urge caution when evaluating events and, where possible, advise looking at background rates.
“A major way to determine whether the adverse event is causally related is to assess the incidence of the adverse event in vaccines versus nonvaccines,” said Walter Orenstein, MD, who directed the U.S. Immunization Program from 1988 to 2004 and is now associate director of the Emory Vaccine Center and professor of infectious diseases at Emory University in Atlanta. Public health officials could then identify vaccine risk factors, he said.
When a reaction occurs almost immediately after vaccination, vaccine safety investigators look for probable triggers. If allergy to PEG is the culprit in anaphylactic reactions, then the individuals would have had a previous exposure, perhaps from injectable medications, Dr. Edwards said.
It might be feasible to perform a skin test for allergy to PEG. “If the skin testing is negative, that doesn’t completely rule out allergy, but it can be used in the decision-making about giving the first or second vaccine dose,” Dr. Banerji said.
Other vaccines, such as childhood vaccines, contain polysorbate as a stabilizer, which has a similar chemical structure, and it’s not clear why someone would react to PEG but not to polysorbate, Dr. Edwards said.
Meanwhile, other illnesses and even deaths sometimes occur in the days after vaccination, but that doesn’t mean the vaccine caused them, cautioned Steve Black, MD, emeritus professor of pediatrics at Cincinnati Children’s Hospital and cofounder of the Global Vaccine Data Network, an international vaccine safety collaboration.
“Different events and clusters of events will occur by chance alone, as these events can occur without vaccines. We need to not immediately assume that they’re due to the vaccine,” he said. “You don’t want to undermine the whole vaccine program every time something comes up and assume that it’s associated with the vaccine.”
The CDC only has three contraindications for the vaccines:
- Severe allergic reaction (such as anaphylaxis) after a previous dose of an mRNA COVID-19 vaccine or any of its components.
- Immediate allergic reaction of any severity to a previous dose of an mRNA COVID-19 vaccine or any of its components (including PEG).
- Immediate allergic reaction of any severity to polysorbate (due to potential cross-reactive hypersensitivity with PEG).
People who have had an immediate allergic reaction to other vaccines or injectable therapies should consider consulting with an allergist or immunologist before getting the Pfizer or Moderna vaccines, the CDC advises.
The CDC also says that people with a history of anaphylaxis from any cause should be observed for 30 minutes after vaccination. Vaccination protocol calls for everyone else to wait on site for 15 minutes after vaccination.
A version of this article first appeared on Medscape.com.
COVID-19 in children: New cases down for third straight week
New COVID-19 cases in children dropped for the third consecutive week, even as children continue to make up a larger share of all cases, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
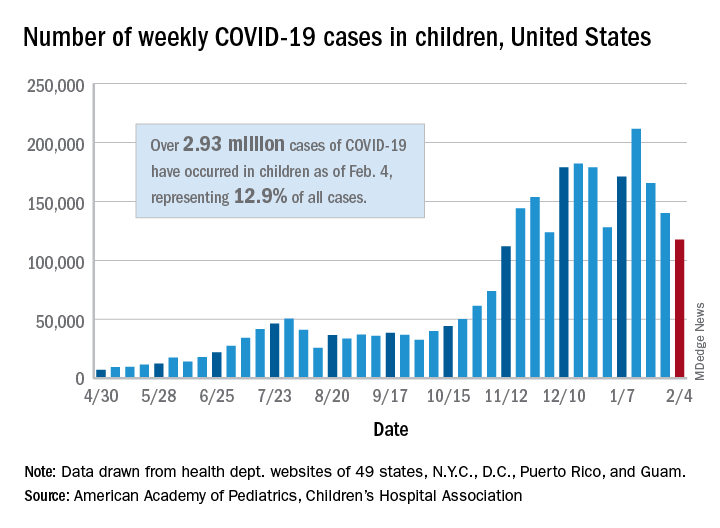
New child cases totaled almost 118,000 for the week of Jan. 29-Feb. 4, continuing the decline that began right after the United States topped 200,000 cases for the only time Jan. 8-14, the AAP and the CHA said in their weekly COVID-19 report.
For the latest week, however, children represented 16.0% of all new COVID-19 cases, continuing a 5-week increase that began in early December 2020, after the proportion had dropped to 12.6%, based on data collected from the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. During the week of Sept. 11-17, children made up 16.9% of all cases, the highest level seen during the pandemic.
The 2.93 million cases that have been reported in children make up 12.9% of all cases since the pandemic began, and the overall rate of pediatric coronavirus infection is 3,899 cases per 100,000 children in the population. Taking a step down from the national level, 30 states are above that rate and 18 are below it, along with D.C., New York City, Puerto Rico, and Guam (New York and Texas are excluded), the AAP and CHA reported.
There were 12 new COVID-19–related child deaths in the 43 states, along with New York City and Guam, that are reporting such data, bringing the total to 227. Nationally, 0.06% of all deaths have occurred in children, with rates ranging from 0.00% (11 states) to 0.26% (Nebraska) in the 45 jurisdictions, the AAP/CHA report shows.
Child hospitalizations rose to 1.9% of all hospitalizations after holding at 1.8% since mid-November in 25 reporting jurisdictions (24 states and New York City), but the hospitalization rate among children with COVID held at 0.8%, where it has been for the last 4 weeks. Hospitalization rates as high as 3.8% were recorded early in the pandemic, the AAP and CHA noted.
New COVID-19 cases in children dropped for the third consecutive week, even as children continue to make up a larger share of all cases, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

New child cases totaled almost 118,000 for the week of Jan. 29-Feb. 4, continuing the decline that began right after the United States topped 200,000 cases for the only time Jan. 8-14, the AAP and the CHA said in their weekly COVID-19 report.
For the latest week, however, children represented 16.0% of all new COVID-19 cases, continuing a 5-week increase that began in early December 2020, after the proportion had dropped to 12.6%, based on data collected from the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. During the week of Sept. 11-17, children made up 16.9% of all cases, the highest level seen during the pandemic.
The 2.93 million cases that have been reported in children make up 12.9% of all cases since the pandemic began, and the overall rate of pediatric coronavirus infection is 3,899 cases per 100,000 children in the population. Taking a step down from the national level, 30 states are above that rate and 18 are below it, along with D.C., New York City, Puerto Rico, and Guam (New York and Texas are excluded), the AAP and CHA reported.
There were 12 new COVID-19–related child deaths in the 43 states, along with New York City and Guam, that are reporting such data, bringing the total to 227. Nationally, 0.06% of all deaths have occurred in children, with rates ranging from 0.00% (11 states) to 0.26% (Nebraska) in the 45 jurisdictions, the AAP/CHA report shows.
Child hospitalizations rose to 1.9% of all hospitalizations after holding at 1.8% since mid-November in 25 reporting jurisdictions (24 states and New York City), but the hospitalization rate among children with COVID held at 0.8%, where it has been for the last 4 weeks. Hospitalization rates as high as 3.8% were recorded early in the pandemic, the AAP and CHA noted.
New COVID-19 cases in children dropped for the third consecutive week, even as children continue to make up a larger share of all cases, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

New child cases totaled almost 118,000 for the week of Jan. 29-Feb. 4, continuing the decline that began right after the United States topped 200,000 cases for the only time Jan. 8-14, the AAP and the CHA said in their weekly COVID-19 report.
For the latest week, however, children represented 16.0% of all new COVID-19 cases, continuing a 5-week increase that began in early December 2020, after the proportion had dropped to 12.6%, based on data collected from the health departments of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. During the week of Sept. 11-17, children made up 16.9% of all cases, the highest level seen during the pandemic.
The 2.93 million cases that have been reported in children make up 12.9% of all cases since the pandemic began, and the overall rate of pediatric coronavirus infection is 3,899 cases per 100,000 children in the population. Taking a step down from the national level, 30 states are above that rate and 18 are below it, along with D.C., New York City, Puerto Rico, and Guam (New York and Texas are excluded), the AAP and CHA reported.
There were 12 new COVID-19–related child deaths in the 43 states, along with New York City and Guam, that are reporting such data, bringing the total to 227. Nationally, 0.06% of all deaths have occurred in children, with rates ranging from 0.00% (11 states) to 0.26% (Nebraska) in the 45 jurisdictions, the AAP/CHA report shows.
Child hospitalizations rose to 1.9% of all hospitalizations after holding at 1.8% since mid-November in 25 reporting jurisdictions (24 states and New York City), but the hospitalization rate among children with COVID held at 0.8%, where it has been for the last 4 weeks. Hospitalization rates as high as 3.8% were recorded early in the pandemic, the AAP and CHA noted.
Are diagnosticians chasing COVID-linked zebras and missing horses?
The emergence of multiple inflammatory syndrome in children (MIS-C) in association with COVID-19 may be complicating the investigation and diagnosis of more common viral and bacterial infections, potentially delaying treatment and prolonging hospital stays.
Two recent articles published online in Hospital Pediatrics provide evidence of this phenomenon. The articles outlined case studies of children who underwent extensive investigation for MIS-C when in fact they had less severe and more common infections. MIS-C is a severe but rare syndrome that involves systemic hyperinflammation with fever and multisystem organ dysfunction similar to that of Kawasaki disease (KD).
In one of the articles, Matthew Molloy, MD, MPH, of the division of pediatric hospital medicine at Cincinnati Children’s Hospital Medical Center, and colleagues aptly asked: “What are we missing in our search for MIS-C?”
E. coli, not SARS-CoV-2
That question arose from a case involving a 3-year-old boy who had a 6-day history of fever and fatigue. Three days earlier, he had tested negative for strep antigen and COVID-19. He had a persistent, high fever, reduced appetite, and reduced urine output and was taken to the ED. On physical examination, there was no rash, skin peeling, redness of the eye or oral mucosa, congestion, rhinorrhea, cough, shortness of breath, chest pain, abdominal pain, nausea, vomiting, or diarrhea.
Urinalysis results and exam findings were suspicious for pyelonephritis. Other findings from an extensive laboratory workup raised the alarm that the boy was suffering from MIS-C as opposed to incomplete KD. After admission to hospital medicine, the cardiology, rheumatology, and infectious disease teams were called in to consult.
Repeat labs were planned for the following day before initiating therapy. On day 2, the child’s urine culture was positive for gram-negative rods, later identified as Escherichia coli. The boy was started on ceftriaxone. Left renal scarring was apparent on ultrasound. The patient’s condition resolved after 36 hours, and he was discharged home with antibiotics.
‘Diagnosis derailed’
Calling this a case of “diagnosis derailed,” the authors noted that, in the pre-COVID era, this child’s signs and symptoms would likely have triggered a more targeted and less costly evaluation for more common infectious and noninfectious causes, including pyelonephritis, absent any physical exam findings consistent with KD.
“However, the patient presented in the midst of the COVID-19 pandemic with growing awareness of a new clinical entity,” Dr. Molloy and colleagues wrote. “Anchored to the patient’s persistent fever, the medical team initiated an extensive, costly, and ultimately unnecessary workup to avoid missing the diagnosis of MIS-C; a not yet well-described diagnosis with potentially severe morbidity.”
Confirmation bias and diagnostic momentum likely contributed to the early focus on MIS-C rather than more common alternatives, the authors acknowledged. The addition of mildly abnormal laboratory data not typically obtained in the evaluation of fever led the team astray. “The diagnosis and definitive treatment may have been made earlier had the focus on concern for MIS-C not been present,” Dr. Molloy said in an interview.
Keeping value in care
The authors recognized that their initial approach to evaluating for MIS-C provided low-value care. “In our desire to not ‘miss’ MIS-C, we were performing costly evaluations that at times produced mildly abnormal, nonspecific results,” they wrote. That triggered a cascade of specialty consultations, follow-up testing, and an unwarranted diagnostic preoccupation with MIS-C.
Determining the extra price tag for the child’s workup would be complex and difficult because there is a difference in the cost to the hospital and the cost to the family, Dr. Molloy said. “However, there are potential cost savings that would be related to making a correct diagnosis in a timely manner in terms of preventing downstream effects from delayed diagnoses.”
Even as clinicians struggle with the challenging SARS-CoV-2 learning curve, Dr. Molloy and associates urged them to continue to strive for high-value care, with an unwavering focus on using only necessary resources, a stewardship the pandemic has shown to be critical.
“The COVID-19 pandemic has been an incredibly stressful time for physicians and for families,” Dr. Molloy said. “COVID-19 and related conditions like MIS-C are new, and we are learning more and more about them every week. These diagnoses are understandably on the minds of physicians and families when children present with fever.” Notwithstanding, the boy’s case underscores the need for clinicians to consider alternate diagnoses and the value of the care provided.
Impact of bias
Dr. Molloy’s group brings home the cognitive biases practitioners often suffer from, including anchoring and confirmation bias and diagnostic momentum, according to J. Howard Smart, MD, chief of pediatrics at Sharp Mary Birch Hospital for Women and Newborns, San Diego, and an assistant clinical professor of pediatrics at University of California, San Diego.
“But it is one thing to recognize these in retrospect and quite another to consider whether they may be happening to you yourself in real time,” he said in an interview. “It is almost as if we need to have a ‘time out,’ where we stop and ask ourselves whether there is something else that could be explaining our patient’s presentation, something that would be more common and more likely to be occurring.”
According to Dr. Smart, who was not involved in Dr. Molloy’s study, the team’s premature diagnostic focus on MIS-C was almost the inverse of what typically happens with KD. “It is usually the case that Kawasaki disease does not enter the differential diagnosis until late in the course of the fever, typically on day 5 or later, when it may have been better to think of it earlier,” he said.
In the second article, Andrea Dean, MD, of the department of pediatrics at Baylor College of Medicine and Texas Children’s Hospital, both in Houston, and colleagues outlined the cases of five patients aged 8-17 years who were hospitalized in May 2020 for suspected MIS-C. They exhibited inflammatory and other concerning indicators but were eventually discharged with a diagnosis of murine typhus.
This flea-borne infection, most commonly reported in the United States in the southeastern Gulf Coast region, Hawaii, and California, is often associated with a triad of fever, rash, and headache.
Cases have been rising in southern Texas, and Dr. Dean and colleagues postulated that school closures and social distancing may have increased exposure as a result of children spending more time outdoors or with pets. “Alternatively, parental concern for SARS-CoV-2 infection could mean children with symptoms are presenting to care and being referred or admitted to the hospital more frequently due to provider concern for MIS-C,” they wrote.
Cardiac involvement
The most concerning of the five cases in terms of possible MIS-C, Dr. Dean said in an interview, was that of a 12-year-old boy who had fever for 6 days in association with headache, eczematous rash, dry lips, and conjunctivitis. Laboratory tests showed a mildly elevated C-reactive protein level, hyponatremia, and thrombocytopenia, as well as sterile pyuria and mildly elevated prothrombin time. He was treated empirically with doxycycline, and his fever resolved over the next 24 hours.
An echocardiogram at initial evaluation, however, revealed mild dilation of the left anterior descending and right coronary arteries, which led to the administration of intravenous immunoglobulin and aspirin for atypical KD, in contrast to MIS-C. The authors postulated that mild cardiac involvement in disorders other than MIS-C and KD may be underrecognized.
The lesson from these cases, Dr. Dean and associates concluded, is that hospitalists must maintain a wide differential diagnosis when assessing a child with prolonged fever and evidence of systemic inflammation. The CDC stipulates that a diagnosis of MIS-C requires the absence of a plausible alternative diagnosis.
In addition to common viral, bacterial, and noninfectious disorders, a range of regional endemic rickettsial and parasitic infections must be considered as alternative diagnoses to MIS-C. “Many of these diseases cannot be reliably differentiated from MIS-C on presentation, and as community exposure to SARS-CoV-2 grows, hospitalists should be prepared to admit febrile children with evidence of systemic inflammation for brief observation periods to evaluate for MIS-C,” Dr. Dean’s group wrote. In this context, however, empiric treatment for common or even uncommon infectious diseases may avoid overdiagnosis and overtreatment of MIS-C as well as improve patient outcomes.
“We do have specific MIS-C guidelines at our institution,” Dr. Dean said, “but like all institutions, we are dealing with the broad definition of MIS-C according to the World Health Organization and the CDC, which is really the takeaway from this paper.”
More difficult differentiation
Both groups of authors pointed out that, as SARS-CoV-2 spreads throughout a community, a higher percentage of the population will have positive results on antibody testing, and such results will become less useful for differentiating between MIS-C and other conditions.
Despite these series’ cautionary lessons, other experts point to the critical importance of including MIS-C early on in the interest of efficient diagnosis and therapy. “In the cases cited, other pathologies were evaluated for and treated accordingly,” said Kara Gross Margolis, MD, AGAF, an associate professor of pediatrics in the division of pediatric gastroenterology, hepatology, and nutrition at Morgan Stanley Children’s Hospital,New York. “These papers stress the need for a balance that is important, and all potential diagnoses need to be considered, but MIS-C, due to its potential severe consequences, also needs to be on our differential now.”
In her view, as this new high-morbidity entity becomes more widespread during the pandemic, it will be increasingly important to keep this condition on the diagnostic radar.
Interestingly, in a converse example of diagnostic clouding, Dr. Gross Margolis’s group reported (Gastroenterology. 2020 Oct;159[4]:1571-4.e2) last year on a pediatric case series in which the presence of gastrointestinal symptoms in children with COVID-19–related MIS-C muddied the diagnosis by confusing this potentially severe syndrome with more common and less toxic gastrointestinal infections.
According to Dr. Smart, although the two reports don’t offer evidence for a particular diagnostic practice, they can inform the decision-making process. “It may be that we will have enough evidence shortly to say what the best practice is regarding diagnostic evaluation of possible MIS-C cases,” he said. “Until then, we must remember that common things occur commonly, even during a global pandemic.”
Neither of the two reports received any specific funding. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The emergence of multiple inflammatory syndrome in children (MIS-C) in association with COVID-19 may be complicating the investigation and diagnosis of more common viral and bacterial infections, potentially delaying treatment and prolonging hospital stays.
Two recent articles published online in Hospital Pediatrics provide evidence of this phenomenon. The articles outlined case studies of children who underwent extensive investigation for MIS-C when in fact they had less severe and more common infections. MIS-C is a severe but rare syndrome that involves systemic hyperinflammation with fever and multisystem organ dysfunction similar to that of Kawasaki disease (KD).
In one of the articles, Matthew Molloy, MD, MPH, of the division of pediatric hospital medicine at Cincinnati Children’s Hospital Medical Center, and colleagues aptly asked: “What are we missing in our search for MIS-C?”
E. coli, not SARS-CoV-2
That question arose from a case involving a 3-year-old boy who had a 6-day history of fever and fatigue. Three days earlier, he had tested negative for strep antigen and COVID-19. He had a persistent, high fever, reduced appetite, and reduced urine output and was taken to the ED. On physical examination, there was no rash, skin peeling, redness of the eye or oral mucosa, congestion, rhinorrhea, cough, shortness of breath, chest pain, abdominal pain, nausea, vomiting, or diarrhea.
Urinalysis results and exam findings were suspicious for pyelonephritis. Other findings from an extensive laboratory workup raised the alarm that the boy was suffering from MIS-C as opposed to incomplete KD. After admission to hospital medicine, the cardiology, rheumatology, and infectious disease teams were called in to consult.
Repeat labs were planned for the following day before initiating therapy. On day 2, the child’s urine culture was positive for gram-negative rods, later identified as Escherichia coli. The boy was started on ceftriaxone. Left renal scarring was apparent on ultrasound. The patient’s condition resolved after 36 hours, and he was discharged home with antibiotics.
‘Diagnosis derailed’
Calling this a case of “diagnosis derailed,” the authors noted that, in the pre-COVID era, this child’s signs and symptoms would likely have triggered a more targeted and less costly evaluation for more common infectious and noninfectious causes, including pyelonephritis, absent any physical exam findings consistent with KD.
“However, the patient presented in the midst of the COVID-19 pandemic with growing awareness of a new clinical entity,” Dr. Molloy and colleagues wrote. “Anchored to the patient’s persistent fever, the medical team initiated an extensive, costly, and ultimately unnecessary workup to avoid missing the diagnosis of MIS-C; a not yet well-described diagnosis with potentially severe morbidity.”
Confirmation bias and diagnostic momentum likely contributed to the early focus on MIS-C rather than more common alternatives, the authors acknowledged. The addition of mildly abnormal laboratory data not typically obtained in the evaluation of fever led the team astray. “The diagnosis and definitive treatment may have been made earlier had the focus on concern for MIS-C not been present,” Dr. Molloy said in an interview.
Keeping value in care
The authors recognized that their initial approach to evaluating for MIS-C provided low-value care. “In our desire to not ‘miss’ MIS-C, we were performing costly evaluations that at times produced mildly abnormal, nonspecific results,” they wrote. That triggered a cascade of specialty consultations, follow-up testing, and an unwarranted diagnostic preoccupation with MIS-C.
Determining the extra price tag for the child’s workup would be complex and difficult because there is a difference in the cost to the hospital and the cost to the family, Dr. Molloy said. “However, there are potential cost savings that would be related to making a correct diagnosis in a timely manner in terms of preventing downstream effects from delayed diagnoses.”
Even as clinicians struggle with the challenging SARS-CoV-2 learning curve, Dr. Molloy and associates urged them to continue to strive for high-value care, with an unwavering focus on using only necessary resources, a stewardship the pandemic has shown to be critical.
“The COVID-19 pandemic has been an incredibly stressful time for physicians and for families,” Dr. Molloy said. “COVID-19 and related conditions like MIS-C are new, and we are learning more and more about them every week. These diagnoses are understandably on the minds of physicians and families when children present with fever.” Notwithstanding, the boy’s case underscores the need for clinicians to consider alternate diagnoses and the value of the care provided.
Impact of bias
Dr. Molloy’s group brings home the cognitive biases practitioners often suffer from, including anchoring and confirmation bias and diagnostic momentum, according to J. Howard Smart, MD, chief of pediatrics at Sharp Mary Birch Hospital for Women and Newborns, San Diego, and an assistant clinical professor of pediatrics at University of California, San Diego.
“But it is one thing to recognize these in retrospect and quite another to consider whether they may be happening to you yourself in real time,” he said in an interview. “It is almost as if we need to have a ‘time out,’ where we stop and ask ourselves whether there is something else that could be explaining our patient’s presentation, something that would be more common and more likely to be occurring.”
According to Dr. Smart, who was not involved in Dr. Molloy’s study, the team’s premature diagnostic focus on MIS-C was almost the inverse of what typically happens with KD. “It is usually the case that Kawasaki disease does not enter the differential diagnosis until late in the course of the fever, typically on day 5 or later, when it may have been better to think of it earlier,” he said.
In the second article, Andrea Dean, MD, of the department of pediatrics at Baylor College of Medicine and Texas Children’s Hospital, both in Houston, and colleagues outlined the cases of five patients aged 8-17 years who were hospitalized in May 2020 for suspected MIS-C. They exhibited inflammatory and other concerning indicators but were eventually discharged with a diagnosis of murine typhus.
This flea-borne infection, most commonly reported in the United States in the southeastern Gulf Coast region, Hawaii, and California, is often associated with a triad of fever, rash, and headache.
Cases have been rising in southern Texas, and Dr. Dean and colleagues postulated that school closures and social distancing may have increased exposure as a result of children spending more time outdoors or with pets. “Alternatively, parental concern for SARS-CoV-2 infection could mean children with symptoms are presenting to care and being referred or admitted to the hospital more frequently due to provider concern for MIS-C,” they wrote.
Cardiac involvement
The most concerning of the five cases in terms of possible MIS-C, Dr. Dean said in an interview, was that of a 12-year-old boy who had fever for 6 days in association with headache, eczematous rash, dry lips, and conjunctivitis. Laboratory tests showed a mildly elevated C-reactive protein level, hyponatremia, and thrombocytopenia, as well as sterile pyuria and mildly elevated prothrombin time. He was treated empirically with doxycycline, and his fever resolved over the next 24 hours.
An echocardiogram at initial evaluation, however, revealed mild dilation of the left anterior descending and right coronary arteries, which led to the administration of intravenous immunoglobulin and aspirin for atypical KD, in contrast to MIS-C. The authors postulated that mild cardiac involvement in disorders other than MIS-C and KD may be underrecognized.
The lesson from these cases, Dr. Dean and associates concluded, is that hospitalists must maintain a wide differential diagnosis when assessing a child with prolonged fever and evidence of systemic inflammation. The CDC stipulates that a diagnosis of MIS-C requires the absence of a plausible alternative diagnosis.
In addition to common viral, bacterial, and noninfectious disorders, a range of regional endemic rickettsial and parasitic infections must be considered as alternative diagnoses to MIS-C. “Many of these diseases cannot be reliably differentiated from MIS-C on presentation, and as community exposure to SARS-CoV-2 grows, hospitalists should be prepared to admit febrile children with evidence of systemic inflammation for brief observation periods to evaluate for MIS-C,” Dr. Dean’s group wrote. In this context, however, empiric treatment for common or even uncommon infectious diseases may avoid overdiagnosis and overtreatment of MIS-C as well as improve patient outcomes.
“We do have specific MIS-C guidelines at our institution,” Dr. Dean said, “but like all institutions, we are dealing with the broad definition of MIS-C according to the World Health Organization and the CDC, which is really the takeaway from this paper.”
More difficult differentiation
Both groups of authors pointed out that, as SARS-CoV-2 spreads throughout a community, a higher percentage of the population will have positive results on antibody testing, and such results will become less useful for differentiating between MIS-C and other conditions.
Despite these series’ cautionary lessons, other experts point to the critical importance of including MIS-C early on in the interest of efficient diagnosis and therapy. “In the cases cited, other pathologies were evaluated for and treated accordingly,” said Kara Gross Margolis, MD, AGAF, an associate professor of pediatrics in the division of pediatric gastroenterology, hepatology, and nutrition at Morgan Stanley Children’s Hospital,New York. “These papers stress the need for a balance that is important, and all potential diagnoses need to be considered, but MIS-C, due to its potential severe consequences, also needs to be on our differential now.”
In her view, as this new high-morbidity entity becomes more widespread during the pandemic, it will be increasingly important to keep this condition on the diagnostic radar.
Interestingly, in a converse example of diagnostic clouding, Dr. Gross Margolis’s group reported (Gastroenterology. 2020 Oct;159[4]:1571-4.e2) last year on a pediatric case series in which the presence of gastrointestinal symptoms in children with COVID-19–related MIS-C muddied the diagnosis by confusing this potentially severe syndrome with more common and less toxic gastrointestinal infections.
According to Dr. Smart, although the two reports don’t offer evidence for a particular diagnostic practice, they can inform the decision-making process. “It may be that we will have enough evidence shortly to say what the best practice is regarding diagnostic evaluation of possible MIS-C cases,” he said. “Until then, we must remember that common things occur commonly, even during a global pandemic.”
Neither of the two reports received any specific funding. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The emergence of multiple inflammatory syndrome in children (MIS-C) in association with COVID-19 may be complicating the investigation and diagnosis of more common viral and bacterial infections, potentially delaying treatment and prolonging hospital stays.
Two recent articles published online in Hospital Pediatrics provide evidence of this phenomenon. The articles outlined case studies of children who underwent extensive investigation for MIS-C when in fact they had less severe and more common infections. MIS-C is a severe but rare syndrome that involves systemic hyperinflammation with fever and multisystem organ dysfunction similar to that of Kawasaki disease (KD).
In one of the articles, Matthew Molloy, MD, MPH, of the division of pediatric hospital medicine at Cincinnati Children’s Hospital Medical Center, and colleagues aptly asked: “What are we missing in our search for MIS-C?”
E. coli, not SARS-CoV-2
That question arose from a case involving a 3-year-old boy who had a 6-day history of fever and fatigue. Three days earlier, he had tested negative for strep antigen and COVID-19. He had a persistent, high fever, reduced appetite, and reduced urine output and was taken to the ED. On physical examination, there was no rash, skin peeling, redness of the eye or oral mucosa, congestion, rhinorrhea, cough, shortness of breath, chest pain, abdominal pain, nausea, vomiting, or diarrhea.
Urinalysis results and exam findings were suspicious for pyelonephritis. Other findings from an extensive laboratory workup raised the alarm that the boy was suffering from MIS-C as opposed to incomplete KD. After admission to hospital medicine, the cardiology, rheumatology, and infectious disease teams were called in to consult.
Repeat labs were planned for the following day before initiating therapy. On day 2, the child’s urine culture was positive for gram-negative rods, later identified as Escherichia coli. The boy was started on ceftriaxone. Left renal scarring was apparent on ultrasound. The patient’s condition resolved after 36 hours, and he was discharged home with antibiotics.
‘Diagnosis derailed’
Calling this a case of “diagnosis derailed,” the authors noted that, in the pre-COVID era, this child’s signs and symptoms would likely have triggered a more targeted and less costly evaluation for more common infectious and noninfectious causes, including pyelonephritis, absent any physical exam findings consistent with KD.
“However, the patient presented in the midst of the COVID-19 pandemic with growing awareness of a new clinical entity,” Dr. Molloy and colleagues wrote. “Anchored to the patient’s persistent fever, the medical team initiated an extensive, costly, and ultimately unnecessary workup to avoid missing the diagnosis of MIS-C; a not yet well-described diagnosis with potentially severe morbidity.”
Confirmation bias and diagnostic momentum likely contributed to the early focus on MIS-C rather than more common alternatives, the authors acknowledged. The addition of mildly abnormal laboratory data not typically obtained in the evaluation of fever led the team astray. “The diagnosis and definitive treatment may have been made earlier had the focus on concern for MIS-C not been present,” Dr. Molloy said in an interview.
Keeping value in care
The authors recognized that their initial approach to evaluating for MIS-C provided low-value care. “In our desire to not ‘miss’ MIS-C, we were performing costly evaluations that at times produced mildly abnormal, nonspecific results,” they wrote. That triggered a cascade of specialty consultations, follow-up testing, and an unwarranted diagnostic preoccupation with MIS-C.
Determining the extra price tag for the child’s workup would be complex and difficult because there is a difference in the cost to the hospital and the cost to the family, Dr. Molloy said. “However, there are potential cost savings that would be related to making a correct diagnosis in a timely manner in terms of preventing downstream effects from delayed diagnoses.”
Even as clinicians struggle with the challenging SARS-CoV-2 learning curve, Dr. Molloy and associates urged them to continue to strive for high-value care, with an unwavering focus on using only necessary resources, a stewardship the pandemic has shown to be critical.
“The COVID-19 pandemic has been an incredibly stressful time for physicians and for families,” Dr. Molloy said. “COVID-19 and related conditions like MIS-C are new, and we are learning more and more about them every week. These diagnoses are understandably on the minds of physicians and families when children present with fever.” Notwithstanding, the boy’s case underscores the need for clinicians to consider alternate diagnoses and the value of the care provided.
Impact of bias
Dr. Molloy’s group brings home the cognitive biases practitioners often suffer from, including anchoring and confirmation bias and diagnostic momentum, according to J. Howard Smart, MD, chief of pediatrics at Sharp Mary Birch Hospital for Women and Newborns, San Diego, and an assistant clinical professor of pediatrics at University of California, San Diego.
“But it is one thing to recognize these in retrospect and quite another to consider whether they may be happening to you yourself in real time,” he said in an interview. “It is almost as if we need to have a ‘time out,’ where we stop and ask ourselves whether there is something else that could be explaining our patient’s presentation, something that would be more common and more likely to be occurring.”
According to Dr. Smart, who was not involved in Dr. Molloy’s study, the team’s premature diagnostic focus on MIS-C was almost the inverse of what typically happens with KD. “It is usually the case that Kawasaki disease does not enter the differential diagnosis until late in the course of the fever, typically on day 5 or later, when it may have been better to think of it earlier,” he said.
In the second article, Andrea Dean, MD, of the department of pediatrics at Baylor College of Medicine and Texas Children’s Hospital, both in Houston, and colleagues outlined the cases of five patients aged 8-17 years who were hospitalized in May 2020 for suspected MIS-C. They exhibited inflammatory and other concerning indicators but were eventually discharged with a diagnosis of murine typhus.
This flea-borne infection, most commonly reported in the United States in the southeastern Gulf Coast region, Hawaii, and California, is often associated with a triad of fever, rash, and headache.
Cases have been rising in southern Texas, and Dr. Dean and colleagues postulated that school closures and social distancing may have increased exposure as a result of children spending more time outdoors or with pets. “Alternatively, parental concern for SARS-CoV-2 infection could mean children with symptoms are presenting to care and being referred or admitted to the hospital more frequently due to provider concern for MIS-C,” they wrote.
Cardiac involvement
The most concerning of the five cases in terms of possible MIS-C, Dr. Dean said in an interview, was that of a 12-year-old boy who had fever for 6 days in association with headache, eczematous rash, dry lips, and conjunctivitis. Laboratory tests showed a mildly elevated C-reactive protein level, hyponatremia, and thrombocytopenia, as well as sterile pyuria and mildly elevated prothrombin time. He was treated empirically with doxycycline, and his fever resolved over the next 24 hours.
An echocardiogram at initial evaluation, however, revealed mild dilation of the left anterior descending and right coronary arteries, which led to the administration of intravenous immunoglobulin and aspirin for atypical KD, in contrast to MIS-C. The authors postulated that mild cardiac involvement in disorders other than MIS-C and KD may be underrecognized.
The lesson from these cases, Dr. Dean and associates concluded, is that hospitalists must maintain a wide differential diagnosis when assessing a child with prolonged fever and evidence of systemic inflammation. The CDC stipulates that a diagnosis of MIS-C requires the absence of a plausible alternative diagnosis.
In addition to common viral, bacterial, and noninfectious disorders, a range of regional endemic rickettsial and parasitic infections must be considered as alternative diagnoses to MIS-C. “Many of these diseases cannot be reliably differentiated from MIS-C on presentation, and as community exposure to SARS-CoV-2 grows, hospitalists should be prepared to admit febrile children with evidence of systemic inflammation for brief observation periods to evaluate for MIS-C,” Dr. Dean’s group wrote. In this context, however, empiric treatment for common or even uncommon infectious diseases may avoid overdiagnosis and overtreatment of MIS-C as well as improve patient outcomes.
“We do have specific MIS-C guidelines at our institution,” Dr. Dean said, “but like all institutions, we are dealing with the broad definition of MIS-C according to the World Health Organization and the CDC, which is really the takeaway from this paper.”
More difficult differentiation
Both groups of authors pointed out that, as SARS-CoV-2 spreads throughout a community, a higher percentage of the population will have positive results on antibody testing, and such results will become less useful for differentiating between MIS-C and other conditions.
Despite these series’ cautionary lessons, other experts point to the critical importance of including MIS-C early on in the interest of efficient diagnosis and therapy. “In the cases cited, other pathologies were evaluated for and treated accordingly,” said Kara Gross Margolis, MD, AGAF, an associate professor of pediatrics in the division of pediatric gastroenterology, hepatology, and nutrition at Morgan Stanley Children’s Hospital,New York. “These papers stress the need for a balance that is important, and all potential diagnoses need to be considered, but MIS-C, due to its potential severe consequences, also needs to be on our differential now.”
In her view, as this new high-morbidity entity becomes more widespread during the pandemic, it will be increasingly important to keep this condition on the diagnostic radar.
Interestingly, in a converse example of diagnostic clouding, Dr. Gross Margolis’s group reported (Gastroenterology. 2020 Oct;159[4]:1571-4.e2) last year on a pediatric case series in which the presence of gastrointestinal symptoms in children with COVID-19–related MIS-C muddied the diagnosis by confusing this potentially severe syndrome with more common and less toxic gastrointestinal infections.
According to Dr. Smart, although the two reports don’t offer evidence for a particular diagnostic practice, they can inform the decision-making process. “It may be that we will have enough evidence shortly to say what the best practice is regarding diagnostic evaluation of possible MIS-C cases,” he said. “Until then, we must remember that common things occur commonly, even during a global pandemic.”
Neither of the two reports received any specific funding. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
SARS-CoV-2 in hospitalized children and youth
Clinical syndromes and predictors of disease severity
Clinical questions: What are the demographics and clinical features of pediatric severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) syndromes, and which admitting demographics and clinical features are predictive of disease severity?
Background: In children, SARS-CoV-2 causes respiratory disease and multisystem inflammatory syndrome in children (MIS-C) as well as other clinical manifestations. The authors of this study chose to address the gap of identifying characteristics for severe disease caused by SARS-CoV-2, including respiratory disease, MIS-C and other manifestations.
Study design: Retrospective and prospective cohort analysis of hospitalized children
Setting: Participating hospitals in Tri-State Pediatric COVID-19 Consortium, including hospitals in New York, New Jersey, and Connecticut.
Synopsis: The authors identified hospitalized patients 22 years old or younger who had a positive SARS-CoV-2 test or met the U.S. Centers for Disease Control and Preventions’ MIS-C case definition. For comparative analysis, patients were divided into a respiratory disease group (based on the World Health Organization’s criteria for COVID-19), MIS-C group or other group (based on the primary reason for hospitalization).
The authors included 281 patients in the study. 51% of the patients presented with respiratory disease, 25% with MIS-C and 25% with other symptoms, including gastrointestinal, or fever. 51% of all patients were Hispanic and 23% were non-Black Hispanic. The most common pre-existing comorbidities amongst all groups were obesity (34%) and asthma (14%).
Patients with respiratory disease had a median age of 14 years while those with MIS-C had a median age of 7 years. Patients more commonly identified as non-Hispanic Black in the MIS-C group vs the respiratory group (35% vs. 18%). Obesity and medical complexity were more prevalent in the respiratory group relative to the MIS-C group. 75% of patients with MIS-C had gastrointestinal symptoms. 44% of respiratory patients had a chest radiograph with bilateral infiltrates on admission, and 18% or respiratory patients required invasive mechanical ventilation. The most common complications in the respiratory group were acute respiratory distress syndrome (17%) and acute kidney injury (11%), whereas shock (35%) and cardiac dysfunction (25%) were the most common complications in the MIS-C group. The median length of stay for all patients was 4 days (IQR 2-8 days).
Patients with MIS-C were more likely to be admitted to the intensive care unit (ICU) but all deaths (7 patients) occurred in the respiratory group. 40% of patients with respiratory disease, 56% of patients with MIS-C, and 6% of other patients met the authors’ definition of severe disease (ICU admission > 48 hours). For the respiratory group, younger age, obesity, increasing white blood cell count, hypoxia, and bilateral infiltrates on chest radiograph were independent predictors of severe disease based on multivariate analyses. For the MIS-C group, lower absolute lymphocyte count and increasing CRP at admission were independent predictors of severity.
Bottom line: Mortality in pediatric patients is low. Ethnicity and race were not predictive of disease severity in this model, even though 51% of the patients studied were Hispanic and 23% were non-Hispanic Black. Severity of illness for patients with respiratory disease was found to be associated with younger age, obesity, increasing white blood cell count, hypoxia, and bilateral infiltrates on chest radiograph. Severity of illness in patients with MIS-C was associated with lower absolute lymphocyte count and increasing CRP.
Citation: Fernandes DM, et al. Severe acute respiratory syndrome coronavirus 2 clinical syndromes and predictors of disease severity in hospitalized children and youth. J Pediatr. 2020 Nov 14;S0022-3476(20):31393-7. DOI: 10.1016/j.jpeds.2020.11.016.
Dr. Kumar is an assistant professor of pediatrics at the Cleveland Clinic Lerner College of Medicine of Case Western Reserve University and a pediatric hospitalist at Cleveland Clinic Children’s. She is the pediatric editor of The Hospitalist.
Clinical syndromes and predictors of disease severity
Clinical syndromes and predictors of disease severity
Clinical questions: What are the demographics and clinical features of pediatric severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) syndromes, and which admitting demographics and clinical features are predictive of disease severity?
Background: In children, SARS-CoV-2 causes respiratory disease and multisystem inflammatory syndrome in children (MIS-C) as well as other clinical manifestations. The authors of this study chose to address the gap of identifying characteristics for severe disease caused by SARS-CoV-2, including respiratory disease, MIS-C and other manifestations.
Study design: Retrospective and prospective cohort analysis of hospitalized children
Setting: Participating hospitals in Tri-State Pediatric COVID-19 Consortium, including hospitals in New York, New Jersey, and Connecticut.
Synopsis: The authors identified hospitalized patients 22 years old or younger who had a positive SARS-CoV-2 test or met the U.S. Centers for Disease Control and Preventions’ MIS-C case definition. For comparative analysis, patients were divided into a respiratory disease group (based on the World Health Organization’s criteria for COVID-19), MIS-C group or other group (based on the primary reason for hospitalization).
The authors included 281 patients in the study. 51% of the patients presented with respiratory disease, 25% with MIS-C and 25% with other symptoms, including gastrointestinal, or fever. 51% of all patients were Hispanic and 23% were non-Black Hispanic. The most common pre-existing comorbidities amongst all groups were obesity (34%) and asthma (14%).
Patients with respiratory disease had a median age of 14 years while those with MIS-C had a median age of 7 years. Patients more commonly identified as non-Hispanic Black in the MIS-C group vs the respiratory group (35% vs. 18%). Obesity and medical complexity were more prevalent in the respiratory group relative to the MIS-C group. 75% of patients with MIS-C had gastrointestinal symptoms. 44% of respiratory patients had a chest radiograph with bilateral infiltrates on admission, and 18% or respiratory patients required invasive mechanical ventilation. The most common complications in the respiratory group were acute respiratory distress syndrome (17%) and acute kidney injury (11%), whereas shock (35%) and cardiac dysfunction (25%) were the most common complications in the MIS-C group. The median length of stay for all patients was 4 days (IQR 2-8 days).
Patients with MIS-C were more likely to be admitted to the intensive care unit (ICU) but all deaths (7 patients) occurred in the respiratory group. 40% of patients with respiratory disease, 56% of patients with MIS-C, and 6% of other patients met the authors’ definition of severe disease (ICU admission > 48 hours). For the respiratory group, younger age, obesity, increasing white blood cell count, hypoxia, and bilateral infiltrates on chest radiograph were independent predictors of severe disease based on multivariate analyses. For the MIS-C group, lower absolute lymphocyte count and increasing CRP at admission were independent predictors of severity.
Bottom line: Mortality in pediatric patients is low. Ethnicity and race were not predictive of disease severity in this model, even though 51% of the patients studied were Hispanic and 23% were non-Hispanic Black. Severity of illness for patients with respiratory disease was found to be associated with younger age, obesity, increasing white blood cell count, hypoxia, and bilateral infiltrates on chest radiograph. Severity of illness in patients with MIS-C was associated with lower absolute lymphocyte count and increasing CRP.
Citation: Fernandes DM, et al. Severe acute respiratory syndrome coronavirus 2 clinical syndromes and predictors of disease severity in hospitalized children and youth. J Pediatr. 2020 Nov 14;S0022-3476(20):31393-7. DOI: 10.1016/j.jpeds.2020.11.016.
Dr. Kumar is an assistant professor of pediatrics at the Cleveland Clinic Lerner College of Medicine of Case Western Reserve University and a pediatric hospitalist at Cleveland Clinic Children’s. She is the pediatric editor of The Hospitalist.
Clinical questions: What are the demographics and clinical features of pediatric severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) syndromes, and which admitting demographics and clinical features are predictive of disease severity?
Background: In children, SARS-CoV-2 causes respiratory disease and multisystem inflammatory syndrome in children (MIS-C) as well as other clinical manifestations. The authors of this study chose to address the gap of identifying characteristics for severe disease caused by SARS-CoV-2, including respiratory disease, MIS-C and other manifestations.
Study design: Retrospective and prospective cohort analysis of hospitalized children
Setting: Participating hospitals in Tri-State Pediatric COVID-19 Consortium, including hospitals in New York, New Jersey, and Connecticut.
Synopsis: The authors identified hospitalized patients 22 years old or younger who had a positive SARS-CoV-2 test or met the U.S. Centers for Disease Control and Preventions’ MIS-C case definition. For comparative analysis, patients were divided into a respiratory disease group (based on the World Health Organization’s criteria for COVID-19), MIS-C group or other group (based on the primary reason for hospitalization).
The authors included 281 patients in the study. 51% of the patients presented with respiratory disease, 25% with MIS-C and 25% with other symptoms, including gastrointestinal, or fever. 51% of all patients were Hispanic and 23% were non-Black Hispanic. The most common pre-existing comorbidities amongst all groups were obesity (34%) and asthma (14%).
Patients with respiratory disease had a median age of 14 years while those with MIS-C had a median age of 7 years. Patients more commonly identified as non-Hispanic Black in the MIS-C group vs the respiratory group (35% vs. 18%). Obesity and medical complexity were more prevalent in the respiratory group relative to the MIS-C group. 75% of patients with MIS-C had gastrointestinal symptoms. 44% of respiratory patients had a chest radiograph with bilateral infiltrates on admission, and 18% or respiratory patients required invasive mechanical ventilation. The most common complications in the respiratory group were acute respiratory distress syndrome (17%) and acute kidney injury (11%), whereas shock (35%) and cardiac dysfunction (25%) were the most common complications in the MIS-C group. The median length of stay for all patients was 4 days (IQR 2-8 days).
Patients with MIS-C were more likely to be admitted to the intensive care unit (ICU) but all deaths (7 patients) occurred in the respiratory group. 40% of patients with respiratory disease, 56% of patients with MIS-C, and 6% of other patients met the authors’ definition of severe disease (ICU admission > 48 hours). For the respiratory group, younger age, obesity, increasing white blood cell count, hypoxia, and bilateral infiltrates on chest radiograph were independent predictors of severe disease based on multivariate analyses. For the MIS-C group, lower absolute lymphocyte count and increasing CRP at admission were independent predictors of severity.
Bottom line: Mortality in pediatric patients is low. Ethnicity and race were not predictive of disease severity in this model, even though 51% of the patients studied were Hispanic and 23% were non-Hispanic Black. Severity of illness for patients with respiratory disease was found to be associated with younger age, obesity, increasing white blood cell count, hypoxia, and bilateral infiltrates on chest radiograph. Severity of illness in patients with MIS-C was associated with lower absolute lymphocyte count and increasing CRP.
Citation: Fernandes DM, et al. Severe acute respiratory syndrome coronavirus 2 clinical syndromes and predictors of disease severity in hospitalized children and youth. J Pediatr. 2020 Nov 14;S0022-3476(20):31393-7. DOI: 10.1016/j.jpeds.2020.11.016.
Dr. Kumar is an assistant professor of pediatrics at the Cleveland Clinic Lerner College of Medicine of Case Western Reserve University and a pediatric hospitalist at Cleveland Clinic Children’s. She is the pediatric editor of The Hospitalist.
FROM THE JOURNAL OF PEDIATRICS
The journey from burnout to wellbeing
A check-in for you and your peers
COVID-19 did not discriminate when it came to the impact it imposed on our hospitalist community. As the nomenclature moves away from the negative connotations of ‘burnout’ to ‘wellbeing,’ the pandemic has taught us something important about being intentional about our personal health: we must secure our own oxygen masks before helping others.
In February 2020, the Society of Hospital Medicine’s Wellbeing Taskforce efforts quickly changed focus from addressing general wellbeing, to wellbeing during COVID-19. Our Taskforce was commissioned by SHM’s Board with a new charge: Address immediate and ongoing needs of well-being and resiliency support for hospitalists during the COVID-19 pandemic. In this essay, I will discuss how our SHM Wellbeing Taskforce approached the overall topic of wellbeing for hospitalists during the COVID-19 pandemic, including some of our Taskforce group experiences.
The Taskforce started with a framework to aide in cultivating open and authentic conversations within hospital medicine groups. Creating spaces for honest sharing around how providers are doing is a crucial first step to reducing stigma, building mutual support within a group, and elevating issues of wellbeing to the level where structural change can take place. The Taskforce established two objectives for normalizing and mitigating stressors we face as hospitalists during the COVID-19 pandemic:
- Provide a framework for hospitalists to take their own emotional pulse
- Provide an approach to reduce stigma of hospitalists who are suffering from pandemic stress
While a more typical approach to fix stress and burnout is using formal institutional interventions, we used the value and insight provided by SHM’s 7 Drivers of Burnout in Hospital Medicine to help guide the creation of SHM resources in addressing the severe emotional strain being felt across the country by hospitalists. The 7 Drivers support the idea that the social role peers and hospital leaders can make a crucial difference in mitigating stress and burnout. Two examples of social support come to mind from the Wellbeing Taskforce experience:
- Participate in your meetings. One example comes from a member of our group who had underestimated the “healing power” that our group meetings had provided to his psyche. The simple act of participating in our Taskforce meeting and being in the presence of our group had provided such a positive impact that he was better able to face the “death and misery” in his unit with a smile on his face.
- Share what is stressful. The second example of social support comes from an hour of Zoom-based facilitation meetings between the SHM’s Wellbeing Taskforce members and Chapter Leaders in late October. During our Taskforce debrief after the meeting, we came to realize the enormous burden of grief our peers were carrying as one hospitalist had lost a group colleague the previous week due to suicide. Our member who led this meeting was moved – as were we – at how this had impacted his small team, and he was reminded he was not alone.
To form meaningful relationships that foster support, there needs to be a space where people can safely come together at times that initially might feel awkward. After taking steps toward your peers, these conversations can become normalized and contribute to meaningful relationships, providing the opportunity for healthy exchanges on vulnerable topics like emotional and psychological wellbeing. A printable guide for this specific purpose (“HM COVID-19 Check-In Guide for Self and Peers”) was designed to help hospitalists move into safe and supportive conversations with each other. While it is difficult to place a value on the importance these types of conversations have on individual wellbeing, it is known that the quality of a positive work environment where people feel supported can moderate stress, morale, and depression. In other words, hospitalist groups can positively contribute to their social environment during stressful times by sharing meaningful and difficult experiences with one another.
Second, the Taskforce created a social media campaign to provide a public social space for sharing hospitalists’ COVID-19 experiences. We believed that sharing collective experiences with the theme of #YouAreNotAlone and a complementary social media campaign, SHM Cares, on SHM’s social media channels, would further connect the national hospitalist community and provide a different communication pathway to decrease a sense of isolation. This idea came from the second social support idea mentioned earlier to share what is stressful with others in a safe space. We understood that some hospitalists would be more comfortable sharing publicly their comments, photos, and videos in achieving a sense of hospitalist unity.
Using our shared experiences, we identified three pillars for the final structure of the HM COVID-19 Check-In Guide for Self and Peers:
- Pillar 1. Recognize your issues. Recall our oxygen mask metaphor and this is what we mean by recognizing symptoms of new stressors (e.g., sleeplessness, irritability, forgetfulness).
- Pillar 2. Know what to say. A simple open-ended question about how the other person is working through the pandemic is an easy way to start a connection. We learned from a mental health perspective that it is unlikely that you could say anything to make a situation worse by offering a listening ear.
- Pillar 3. Check in with others. Listen to others without trying to fix the person or the situation. When appropriate, offer humorous reflections without diminishing the problem. Be a partner and commit to check in regularly with the other person.
Cultivating human connections outside of your immediate peer group can be valuable and offer additional perspective to stressful situations. For instance, one of my roles as a hospitalist administrator has been offering support by regularly listening as my physicians ‘talk out’ their day confidentially for as long as they needed. Offering open conversation in a safe and confidential way can have a healing effect. As one of my former hospitalists used to say, if issues are not addressed, they will “ooze out somewhere else.”
The HM COVID-19 Check-In Guide for Self and Peers and the SHM Cares social media campaign was the result of the Taskforce’s collective observations to help others normalize the feeling that ‘it’s OK not to be OK.’ Using the pandemic as context, the 7 Drivers of Hospitalist Burnout reminded us that the increased burnout issues we face will require continued attention past the pandemic. The value in cultivating human connections has never been more important. The SHM Wellbeing Taskforce is committed to provide continued resources. Checking in with others and listening to peers are all part of a personal wellbeing and resilience strategy. On behalf of the SHM Wellbeing Taskforce, we hope the information in this article will highlight the importance of continued attention to personal wellbeing during and after the pandemic.
Dr. Robinson received her PhD in organizational learning, performance and change from Colorado State University in 2019. Her dissertation topic was exploring hospitalist burnout, engagement, and social support. She is administrative director of inpatient medicine at St. Mary’s Medical Center in Grand Junction, Colo., a part of SCL Health. She has volunteered in numerous SHM committees, and currently serves on the SHM Wellbeing Taskforce.
A check-in for you and your peers
A check-in for you and your peers
COVID-19 did not discriminate when it came to the impact it imposed on our hospitalist community. As the nomenclature moves away from the negative connotations of ‘burnout’ to ‘wellbeing,’ the pandemic has taught us something important about being intentional about our personal health: we must secure our own oxygen masks before helping others.
In February 2020, the Society of Hospital Medicine’s Wellbeing Taskforce efforts quickly changed focus from addressing general wellbeing, to wellbeing during COVID-19. Our Taskforce was commissioned by SHM’s Board with a new charge: Address immediate and ongoing needs of well-being and resiliency support for hospitalists during the COVID-19 pandemic. In this essay, I will discuss how our SHM Wellbeing Taskforce approached the overall topic of wellbeing for hospitalists during the COVID-19 pandemic, including some of our Taskforce group experiences.
The Taskforce started with a framework to aide in cultivating open and authentic conversations within hospital medicine groups. Creating spaces for honest sharing around how providers are doing is a crucial first step to reducing stigma, building mutual support within a group, and elevating issues of wellbeing to the level where structural change can take place. The Taskforce established two objectives for normalizing and mitigating stressors we face as hospitalists during the COVID-19 pandemic:
- Provide a framework for hospitalists to take their own emotional pulse
- Provide an approach to reduce stigma of hospitalists who are suffering from pandemic stress
While a more typical approach to fix stress and burnout is using formal institutional interventions, we used the value and insight provided by SHM’s 7 Drivers of Burnout in Hospital Medicine to help guide the creation of SHM resources in addressing the severe emotional strain being felt across the country by hospitalists. The 7 Drivers support the idea that the social role peers and hospital leaders can make a crucial difference in mitigating stress and burnout. Two examples of social support come to mind from the Wellbeing Taskforce experience:
- Participate in your meetings. One example comes from a member of our group who had underestimated the “healing power” that our group meetings had provided to his psyche. The simple act of participating in our Taskforce meeting and being in the presence of our group had provided such a positive impact that he was better able to face the “death and misery” in his unit with a smile on his face.
- Share what is stressful. The second example of social support comes from an hour of Zoom-based facilitation meetings between the SHM’s Wellbeing Taskforce members and Chapter Leaders in late October. During our Taskforce debrief after the meeting, we came to realize the enormous burden of grief our peers were carrying as one hospitalist had lost a group colleague the previous week due to suicide. Our member who led this meeting was moved – as were we – at how this had impacted his small team, and he was reminded he was not alone.
To form meaningful relationships that foster support, there needs to be a space where people can safely come together at times that initially might feel awkward. After taking steps toward your peers, these conversations can become normalized and contribute to meaningful relationships, providing the opportunity for healthy exchanges on vulnerable topics like emotional and psychological wellbeing. A printable guide for this specific purpose (“HM COVID-19 Check-In Guide for Self and Peers”) was designed to help hospitalists move into safe and supportive conversations with each other. While it is difficult to place a value on the importance these types of conversations have on individual wellbeing, it is known that the quality of a positive work environment where people feel supported can moderate stress, morale, and depression. In other words, hospitalist groups can positively contribute to their social environment during stressful times by sharing meaningful and difficult experiences with one another.
Second, the Taskforce created a social media campaign to provide a public social space for sharing hospitalists’ COVID-19 experiences. We believed that sharing collective experiences with the theme of #YouAreNotAlone and a complementary social media campaign, SHM Cares, on SHM’s social media channels, would further connect the national hospitalist community and provide a different communication pathway to decrease a sense of isolation. This idea came from the second social support idea mentioned earlier to share what is stressful with others in a safe space. We understood that some hospitalists would be more comfortable sharing publicly their comments, photos, and videos in achieving a sense of hospitalist unity.
Using our shared experiences, we identified three pillars for the final structure of the HM COVID-19 Check-In Guide for Self and Peers:
- Pillar 1. Recognize your issues. Recall our oxygen mask metaphor and this is what we mean by recognizing symptoms of new stressors (e.g., sleeplessness, irritability, forgetfulness).
- Pillar 2. Know what to say. A simple open-ended question about how the other person is working through the pandemic is an easy way to start a connection. We learned from a mental health perspective that it is unlikely that you could say anything to make a situation worse by offering a listening ear.
- Pillar 3. Check in with others. Listen to others without trying to fix the person or the situation. When appropriate, offer humorous reflections without diminishing the problem. Be a partner and commit to check in regularly with the other person.
Cultivating human connections outside of your immediate peer group can be valuable and offer additional perspective to stressful situations. For instance, one of my roles as a hospitalist administrator has been offering support by regularly listening as my physicians ‘talk out’ their day confidentially for as long as they needed. Offering open conversation in a safe and confidential way can have a healing effect. As one of my former hospitalists used to say, if issues are not addressed, they will “ooze out somewhere else.”
The HM COVID-19 Check-In Guide for Self and Peers and the SHM Cares social media campaign was the result of the Taskforce’s collective observations to help others normalize the feeling that ‘it’s OK not to be OK.’ Using the pandemic as context, the 7 Drivers of Hospitalist Burnout reminded us that the increased burnout issues we face will require continued attention past the pandemic. The value in cultivating human connections has never been more important. The SHM Wellbeing Taskforce is committed to provide continued resources. Checking in with others and listening to peers are all part of a personal wellbeing and resilience strategy. On behalf of the SHM Wellbeing Taskforce, we hope the information in this article will highlight the importance of continued attention to personal wellbeing during and after the pandemic.
Dr. Robinson received her PhD in organizational learning, performance and change from Colorado State University in 2019. Her dissertation topic was exploring hospitalist burnout, engagement, and social support. She is administrative director of inpatient medicine at St. Mary’s Medical Center in Grand Junction, Colo., a part of SCL Health. She has volunteered in numerous SHM committees, and currently serves on the SHM Wellbeing Taskforce.
COVID-19 did not discriminate when it came to the impact it imposed on our hospitalist community. As the nomenclature moves away from the negative connotations of ‘burnout’ to ‘wellbeing,’ the pandemic has taught us something important about being intentional about our personal health: we must secure our own oxygen masks before helping others.
In February 2020, the Society of Hospital Medicine’s Wellbeing Taskforce efforts quickly changed focus from addressing general wellbeing, to wellbeing during COVID-19. Our Taskforce was commissioned by SHM’s Board with a new charge: Address immediate and ongoing needs of well-being and resiliency support for hospitalists during the COVID-19 pandemic. In this essay, I will discuss how our SHM Wellbeing Taskforce approached the overall topic of wellbeing for hospitalists during the COVID-19 pandemic, including some of our Taskforce group experiences.
The Taskforce started with a framework to aide in cultivating open and authentic conversations within hospital medicine groups. Creating spaces for honest sharing around how providers are doing is a crucial first step to reducing stigma, building mutual support within a group, and elevating issues of wellbeing to the level where structural change can take place. The Taskforce established two objectives for normalizing and mitigating stressors we face as hospitalists during the COVID-19 pandemic:
- Provide a framework for hospitalists to take their own emotional pulse
- Provide an approach to reduce stigma of hospitalists who are suffering from pandemic stress
While a more typical approach to fix stress and burnout is using formal institutional interventions, we used the value and insight provided by SHM’s 7 Drivers of Burnout in Hospital Medicine to help guide the creation of SHM resources in addressing the severe emotional strain being felt across the country by hospitalists. The 7 Drivers support the idea that the social role peers and hospital leaders can make a crucial difference in mitigating stress and burnout. Two examples of social support come to mind from the Wellbeing Taskforce experience:
- Participate in your meetings. One example comes from a member of our group who had underestimated the “healing power” that our group meetings had provided to his psyche. The simple act of participating in our Taskforce meeting and being in the presence of our group had provided such a positive impact that he was better able to face the “death and misery” in his unit with a smile on his face.
- Share what is stressful. The second example of social support comes from an hour of Zoom-based facilitation meetings between the SHM’s Wellbeing Taskforce members and Chapter Leaders in late October. During our Taskforce debrief after the meeting, we came to realize the enormous burden of grief our peers were carrying as one hospitalist had lost a group colleague the previous week due to suicide. Our member who led this meeting was moved – as were we – at how this had impacted his small team, and he was reminded he was not alone.
To form meaningful relationships that foster support, there needs to be a space where people can safely come together at times that initially might feel awkward. After taking steps toward your peers, these conversations can become normalized and contribute to meaningful relationships, providing the opportunity for healthy exchanges on vulnerable topics like emotional and psychological wellbeing. A printable guide for this specific purpose (“HM COVID-19 Check-In Guide for Self and Peers”) was designed to help hospitalists move into safe and supportive conversations with each other. While it is difficult to place a value on the importance these types of conversations have on individual wellbeing, it is known that the quality of a positive work environment where people feel supported can moderate stress, morale, and depression. In other words, hospitalist groups can positively contribute to their social environment during stressful times by sharing meaningful and difficult experiences with one another.
Second, the Taskforce created a social media campaign to provide a public social space for sharing hospitalists’ COVID-19 experiences. We believed that sharing collective experiences with the theme of #YouAreNotAlone and a complementary social media campaign, SHM Cares, on SHM’s social media channels, would further connect the national hospitalist community and provide a different communication pathway to decrease a sense of isolation. This idea came from the second social support idea mentioned earlier to share what is stressful with others in a safe space. We understood that some hospitalists would be more comfortable sharing publicly their comments, photos, and videos in achieving a sense of hospitalist unity.
Using our shared experiences, we identified three pillars for the final structure of the HM COVID-19 Check-In Guide for Self and Peers:
- Pillar 1. Recognize your issues. Recall our oxygen mask metaphor and this is what we mean by recognizing symptoms of new stressors (e.g., sleeplessness, irritability, forgetfulness).
- Pillar 2. Know what to say. A simple open-ended question about how the other person is working through the pandemic is an easy way to start a connection. We learned from a mental health perspective that it is unlikely that you could say anything to make a situation worse by offering a listening ear.
- Pillar 3. Check in with others. Listen to others without trying to fix the person or the situation. When appropriate, offer humorous reflections without diminishing the problem. Be a partner and commit to check in regularly with the other person.
Cultivating human connections outside of your immediate peer group can be valuable and offer additional perspective to stressful situations. For instance, one of my roles as a hospitalist administrator has been offering support by regularly listening as my physicians ‘talk out’ their day confidentially for as long as they needed. Offering open conversation in a safe and confidential way can have a healing effect. As one of my former hospitalists used to say, if issues are not addressed, they will “ooze out somewhere else.”
The HM COVID-19 Check-In Guide for Self and Peers and the SHM Cares social media campaign was the result of the Taskforce’s collective observations to help others normalize the feeling that ‘it’s OK not to be OK.’ Using the pandemic as context, the 7 Drivers of Hospitalist Burnout reminded us that the increased burnout issues we face will require continued attention past the pandemic. The value in cultivating human connections has never been more important. The SHM Wellbeing Taskforce is committed to provide continued resources. Checking in with others and listening to peers are all part of a personal wellbeing and resilience strategy. On behalf of the SHM Wellbeing Taskforce, we hope the information in this article will highlight the importance of continued attention to personal wellbeing during and after the pandemic.
Dr. Robinson received her PhD in organizational learning, performance and change from Colorado State University in 2019. Her dissertation topic was exploring hospitalist burnout, engagement, and social support. She is administrative director of inpatient medicine at St. Mary’s Medical Center in Grand Junction, Colo., a part of SCL Health. She has volunteered in numerous SHM committees, and currently serves on the SHM Wellbeing Taskforce.
Rollout of COVID-19 monoclonal antibodies lacked unified plan: expert panel
Monoclonal antibodies (mAbs) to treat COVID-19 are in ample supply, but scant evidence on their effectiveness, paltry reimbursement, and a lack of a planned infrastructure to administer them has led to major underutilization of these potentially useful therapies, according to a new report from The National Academies of Sciences, Engineering, and Medicine.
The 35-page report described missed opportunities to work with states and hospitals to establish trust with clinicians and patients and to set up an infusion infrastructure to funnel patients to sites. Though the therapies still need more study, they should be an option for the right patient at the right time, said the National Academies experts in their report, Rapid Expert Consultation on Allocating COVID-19 Monoclonal Antibody Therapies and Other Novel Therapeutics.
“No potentially eligible patient should be left uninformed, and no eligible patient should be denied access, if there are doses available and the patient and doctor agree it is a reasonable course,” they concluded. The report also noted that underuse, and in particular underuse by members of vulnerable and underserved communities “raises concerns about exacerbating already dramatic health disparities.”
The federal government has spent $375 million on Eli Lilly’s bamlanivimab and $450 million on Regeneron’s casirivimab plus imdevimab cocktail, and agreed last month to spend as much as $2.6 billion more on up to 1.25 million additional doses.
Some 785,000 doses of the two therapeutics have been produced and about a half million have been distributed to states. But about three quarters have gone unused. The U.S. Department of Health & Human Services has launched an online treatment locater to try to spur interest in the therapies.
But the federal government hasn’t addressed some of the basic barriers to use of the monoclonals, said the National Academies experts.
“Lack of awareness, interest, and confidence in COVID-19 mAb therapies among patients and providers are major issues,” they said in the report. Patients who have tested positive might not want to travel to an infusion site, while others might not have access to health care or only seek such treatments when it’s too late. Some who are eligible might not have the time, resources, or transportation to go to a site and sit through a 2-hour treatment.
In addition, “the supply and availability of infusion centers and personnel was identified as a greater constraint than the supply of COVID-19 mAbs,” said the report.
Cost a big impediment
While the federal government has covered the cost of the therapies, hospitals and patients inevitably incur related costs.
“The fragmented payment system in the United States has not provided adequate support to cover the spectrum of costs associated with COVID-19 mAb therapies,” said the report. That is compounded by chronic underfunding and restrictions on federally qualified health centers for community health, the report said.
Patients may have to pay for testing, office visits, follow-up appointments, transportation to and from the infusion site, and potentially a copay for the administration of the drug.
While Medicare pays hospitals $309 per infusion, that might not be enough, especially if a hospital or other site had to build out a new infusion center, the report shows. For clinicians, the administrative payment under Medicare Part B does “not cover the total practice cost to furnish infusion services, resulting in a substantial cost-reimbursement disparity,” the report states.
In addition, there are no specific codes for observing patients during the 2-hour procedure.
“The established Medicare payment rate for furnishing COVID-19 mAb therapies does not cover the cost associated with coordinating care for those patients, nor does it justify the risk and opportunity costs associated with investing in infrastructure modifications to safely integrate COVID-19 patients into existing facilities or building temporary infusion capacity,” the report concluded.
More data needed
The U.S. Food and Drug Administration issued emergency-use authorizations (EUAs) for the two monoclonal therapies based on phase 2 trial data, and that leaves a lot of uncertainty, noted the National Academies.
In trials, both therapies reduced COVID-19-related hospitalizations and emergency room visits within 28 days after treatment among patients at high risk of progression, compared with those who received placebo.
But clinicians aren’t certain about who should use the monoclonals, said the report. The underuse has in turn led to trouble collecting data – either through ongoing trials or in starting new trials.
The National Academies recommended allocating the monoclonal antibodies in a way that would give rise to better data collection to inform clinicians. Payers could support the development of a core data platform or registry, or Medicare could develop pilot trials, said the report.
Lilly and UnitedHealth Group are collaborating on a study in high-risk Medicare patients, according to Reuters. Patients who test positive will be given bamlanivimab at home.
“Building infusion capacity and developing the evidence base about the impact of COVID-19 mAbs on clinical outcomes other than hospitalization, including mortality, are the most promising strategies for increasing effective utilization moving forward,” stated the National Academies report.
A version of this article first appeared on Medscape.com.
Monoclonal antibodies (mAbs) to treat COVID-19 are in ample supply, but scant evidence on their effectiveness, paltry reimbursement, and a lack of a planned infrastructure to administer them has led to major underutilization of these potentially useful therapies, according to a new report from The National Academies of Sciences, Engineering, and Medicine.
The 35-page report described missed opportunities to work with states and hospitals to establish trust with clinicians and patients and to set up an infusion infrastructure to funnel patients to sites. Though the therapies still need more study, they should be an option for the right patient at the right time, said the National Academies experts in their report, Rapid Expert Consultation on Allocating COVID-19 Monoclonal Antibody Therapies and Other Novel Therapeutics.
“No potentially eligible patient should be left uninformed, and no eligible patient should be denied access, if there are doses available and the patient and doctor agree it is a reasonable course,” they concluded. The report also noted that underuse, and in particular underuse by members of vulnerable and underserved communities “raises concerns about exacerbating already dramatic health disparities.”
The federal government has spent $375 million on Eli Lilly’s bamlanivimab and $450 million on Regeneron’s casirivimab plus imdevimab cocktail, and agreed last month to spend as much as $2.6 billion more on up to 1.25 million additional doses.
Some 785,000 doses of the two therapeutics have been produced and about a half million have been distributed to states. But about three quarters have gone unused. The U.S. Department of Health & Human Services has launched an online treatment locater to try to spur interest in the therapies.
But the federal government hasn’t addressed some of the basic barriers to use of the monoclonals, said the National Academies experts.
“Lack of awareness, interest, and confidence in COVID-19 mAb therapies among patients and providers are major issues,” they said in the report. Patients who have tested positive might not want to travel to an infusion site, while others might not have access to health care or only seek such treatments when it’s too late. Some who are eligible might not have the time, resources, or transportation to go to a site and sit through a 2-hour treatment.
In addition, “the supply and availability of infusion centers and personnel was identified as a greater constraint than the supply of COVID-19 mAbs,” said the report.
Cost a big impediment
While the federal government has covered the cost of the therapies, hospitals and patients inevitably incur related costs.
“The fragmented payment system in the United States has not provided adequate support to cover the spectrum of costs associated with COVID-19 mAb therapies,” said the report. That is compounded by chronic underfunding and restrictions on federally qualified health centers for community health, the report said.
Patients may have to pay for testing, office visits, follow-up appointments, transportation to and from the infusion site, and potentially a copay for the administration of the drug.
While Medicare pays hospitals $309 per infusion, that might not be enough, especially if a hospital or other site had to build out a new infusion center, the report shows. For clinicians, the administrative payment under Medicare Part B does “not cover the total practice cost to furnish infusion services, resulting in a substantial cost-reimbursement disparity,” the report states.
In addition, there are no specific codes for observing patients during the 2-hour procedure.
“The established Medicare payment rate for furnishing COVID-19 mAb therapies does not cover the cost associated with coordinating care for those patients, nor does it justify the risk and opportunity costs associated with investing in infrastructure modifications to safely integrate COVID-19 patients into existing facilities or building temporary infusion capacity,” the report concluded.
More data needed
The U.S. Food and Drug Administration issued emergency-use authorizations (EUAs) for the two monoclonal therapies based on phase 2 trial data, and that leaves a lot of uncertainty, noted the National Academies.
In trials, both therapies reduced COVID-19-related hospitalizations and emergency room visits within 28 days after treatment among patients at high risk of progression, compared with those who received placebo.
But clinicians aren’t certain about who should use the monoclonals, said the report. The underuse has in turn led to trouble collecting data – either through ongoing trials or in starting new trials.
The National Academies recommended allocating the monoclonal antibodies in a way that would give rise to better data collection to inform clinicians. Payers could support the development of a core data platform or registry, or Medicare could develop pilot trials, said the report.
Lilly and UnitedHealth Group are collaborating on a study in high-risk Medicare patients, according to Reuters. Patients who test positive will be given bamlanivimab at home.
“Building infusion capacity and developing the evidence base about the impact of COVID-19 mAbs on clinical outcomes other than hospitalization, including mortality, are the most promising strategies for increasing effective utilization moving forward,” stated the National Academies report.
A version of this article first appeared on Medscape.com.
Monoclonal antibodies (mAbs) to treat COVID-19 are in ample supply, but scant evidence on their effectiveness, paltry reimbursement, and a lack of a planned infrastructure to administer them has led to major underutilization of these potentially useful therapies, according to a new report from The National Academies of Sciences, Engineering, and Medicine.
The 35-page report described missed opportunities to work with states and hospitals to establish trust with clinicians and patients and to set up an infusion infrastructure to funnel patients to sites. Though the therapies still need more study, they should be an option for the right patient at the right time, said the National Academies experts in their report, Rapid Expert Consultation on Allocating COVID-19 Monoclonal Antibody Therapies and Other Novel Therapeutics.
“No potentially eligible patient should be left uninformed, and no eligible patient should be denied access, if there are doses available and the patient and doctor agree it is a reasonable course,” they concluded. The report also noted that underuse, and in particular underuse by members of vulnerable and underserved communities “raises concerns about exacerbating already dramatic health disparities.”
The federal government has spent $375 million on Eli Lilly’s bamlanivimab and $450 million on Regeneron’s casirivimab plus imdevimab cocktail, and agreed last month to spend as much as $2.6 billion more on up to 1.25 million additional doses.
Some 785,000 doses of the two therapeutics have been produced and about a half million have been distributed to states. But about three quarters have gone unused. The U.S. Department of Health & Human Services has launched an online treatment locater to try to spur interest in the therapies.
But the federal government hasn’t addressed some of the basic barriers to use of the monoclonals, said the National Academies experts.
“Lack of awareness, interest, and confidence in COVID-19 mAb therapies among patients and providers are major issues,” they said in the report. Patients who have tested positive might not want to travel to an infusion site, while others might not have access to health care or only seek such treatments when it’s too late. Some who are eligible might not have the time, resources, or transportation to go to a site and sit through a 2-hour treatment.
In addition, “the supply and availability of infusion centers and personnel was identified as a greater constraint than the supply of COVID-19 mAbs,” said the report.
Cost a big impediment
While the federal government has covered the cost of the therapies, hospitals and patients inevitably incur related costs.
“The fragmented payment system in the United States has not provided adequate support to cover the spectrum of costs associated with COVID-19 mAb therapies,” said the report. That is compounded by chronic underfunding and restrictions on federally qualified health centers for community health, the report said.
Patients may have to pay for testing, office visits, follow-up appointments, transportation to and from the infusion site, and potentially a copay for the administration of the drug.
While Medicare pays hospitals $309 per infusion, that might not be enough, especially if a hospital or other site had to build out a new infusion center, the report shows. For clinicians, the administrative payment under Medicare Part B does “not cover the total practice cost to furnish infusion services, resulting in a substantial cost-reimbursement disparity,” the report states.
In addition, there are no specific codes for observing patients during the 2-hour procedure.
“The established Medicare payment rate for furnishing COVID-19 mAb therapies does not cover the cost associated with coordinating care for those patients, nor does it justify the risk and opportunity costs associated with investing in infrastructure modifications to safely integrate COVID-19 patients into existing facilities or building temporary infusion capacity,” the report concluded.
More data needed
The U.S. Food and Drug Administration issued emergency-use authorizations (EUAs) for the two monoclonal therapies based on phase 2 trial data, and that leaves a lot of uncertainty, noted the National Academies.
In trials, both therapies reduced COVID-19-related hospitalizations and emergency room visits within 28 days after treatment among patients at high risk of progression, compared with those who received placebo.
But clinicians aren’t certain about who should use the monoclonals, said the report. The underuse has in turn led to trouble collecting data – either through ongoing trials or in starting new trials.
The National Academies recommended allocating the monoclonal antibodies in a way that would give rise to better data collection to inform clinicians. Payers could support the development of a core data platform or registry, or Medicare could develop pilot trials, said the report.
Lilly and UnitedHealth Group are collaborating on a study in high-risk Medicare patients, according to Reuters. Patients who test positive will be given bamlanivimab at home.
“Building infusion capacity and developing the evidence base about the impact of COVID-19 mAbs on clinical outcomes other than hospitalization, including mortality, are the most promising strategies for increasing effective utilization moving forward,” stated the National Academies report.
A version of this article first appeared on Medscape.com.
Cardiac activity not uncommon after lifesaving measures stop
Among critically ill patients pulseless after planned withdrawal of life-sustaining therapies, cardiac activity restarted in 14% of cases, research shows.
Reassuringly, most resumption of heart activity happened in the first 1-2 minutes and most lasted 1 or 2 seconds.
“The reason we wanted to look at death determination specifically is we know that the stories persist about people coming back to life following death, and that’s not just in the public, it’s in the medical community as well,” lead author Sonny Dhanani, MD, of Children’s Hospital of Eastern Ontario, Ottawa, said in an interview.
“We thought that if we provided scientific evidence of whether this happened or not, we might dispel some myths and misunderstanding, which would hopefully promote organ donation.”
About 70% of organ donations occur after brain death, but an increasing number follow circulatory determination of death, he noted. Most protocols recommend 5 minutes of apnea and pulselessness by arterial catheter monitor before declaring death. But practices vary from 10 minutes in some European countries to 75 seconds in infant heart donors at one Colorado hospital.
Reports of patients recovering 10 minutes after pulselessness have raised concerns about the Lazarus phenomenon, or autoresuscitation, but are based in patients after cardiopulmonary resuscitation was terminated.
The present study, known as Death Prediction and Physiology after Removal of Therapy (DePParRT), enrolled patients at 20 intensive care sites in Canada, the Czech Republic, and the Netherlands, only if surrogate decision-makers agreed on withdrawal of life-sustaining measures without CPR and imminent death was anticipated.
As reported Jan. 28 in the New England Journal of Medicine, physicians observed resumption of circulation or cardiac activity prospectively in 1% of 631 patients based on bedside ECG, arterial pressure catheter monitors, palpated arterial pulse, breaths, or physical movements.
A retrospective review of data from 480 patients with complete ECG and arterial waveforms and at least 5 minutes of continuous waveform monitoring after pulselessness showed resumption of cardiac activity in 14% of patients.
The longest period of pulselessness before the heart showed signs of activity again was 4 minutes and 20 seconds. “So that was a reassuring number, because that’s within our 5-minute window that we currently use,” Dr. Dhanani said.
Importantly, “nobody woke up, nobody ended up being resuscitated, and all of these individuals died. And I think that’s going to be very helpful in this context,” he added.
In all, there were 77 cessations and resumptions in 67 of the 480 patients. The median duration of resumed cardiac activity was 3.9 seconds but, notably, ranged from 1 second to 13 minutes and 14 seconds.
“Though surprising, I think maybe not unreasonable,” observed Dr. Dhanani. “The heart is a very robust organ, and we maybe should anticipate these things happening, where at the end of life the heart may restart for minutes.”
In this situation, it’s important to wait the 13 minutes for the heart to stop again and then “wait another 5 minutes to make sure it doesn’t restart before determining death,” he said. “I think that’s where this study is going to now inform policy makers and guidelines, especially in the context of donations.”
The findings will be taken as strong support for the 5-minute window, said Robert Truog, MD, director of the Harvard Medical School Center for Bioethics and the Frances Glessner Lee Professor of Medical Ethics, Anaesthesia, and Pediatrics, Boston.
“I think it’s a safe point, I think people will refer to it, and it will be used to support the 5-minute window, and that’s probably reasonable,” he told this news organization. “Certainly, if it’s read in Europe it will cut the time from 10 minutes to 5 minutes, and that’s a good thing because 10 minutes is a very long time to wait.”
He noted that the 5-minute window provides reasonable assurance to the public and, with new technologies, permits most organs to be usable for donation after cardiac death. That said, there’s nothing magical about the number.
“In some ways I see this paper as providing interesting data but not actually providing an answer, because from the patient’s perspective and from the recipient’s perspective, waiting until the heart has made its last squeeze may not be the most relevant ethical question,” Dr. Truog said. “It may be, once we know this patient is not going to have return of cardiorespiratory function, is not going to wake up, that’s the point at which we ought to focus on organ preservation and organ retrieval, and that can be much sooner than 5 minutes.”
Dr. Dhanani and colleagues note that the generalizability of the results might be limited because patients without arterial pressure catheters were excluded, and 24% of enrolled patients could not be included in the retrospective waveform analysis owing to incomplete data.
“Our study definition of cardiac activity used an arbitrary threshold of pulse pressure (less than 5 mm Hg) that does not imply meaningful circulation,” they add. “This conservative consensus definition may have been partially responsible for the ostensibly high incidence (14%) of transient resumptions of cardiac activity identified through waveform adjudication.”
The study was supported by the Canadian Institutes for Health Research as part of the Canadian Donation and Transplantation Research Program, CHEO Research Institute, and Karel Pavlík Foundation. Dr. Dhanani has consulted for Canadian Blood Services. Dr. Truog reports no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Among critically ill patients pulseless after planned withdrawal of life-sustaining therapies, cardiac activity restarted in 14% of cases, research shows.
Reassuringly, most resumption of heart activity happened in the first 1-2 minutes and most lasted 1 or 2 seconds.
“The reason we wanted to look at death determination specifically is we know that the stories persist about people coming back to life following death, and that’s not just in the public, it’s in the medical community as well,” lead author Sonny Dhanani, MD, of Children’s Hospital of Eastern Ontario, Ottawa, said in an interview.
“We thought that if we provided scientific evidence of whether this happened or not, we might dispel some myths and misunderstanding, which would hopefully promote organ donation.”
About 70% of organ donations occur after brain death, but an increasing number follow circulatory determination of death, he noted. Most protocols recommend 5 minutes of apnea and pulselessness by arterial catheter monitor before declaring death. But practices vary from 10 minutes in some European countries to 75 seconds in infant heart donors at one Colorado hospital.
Reports of patients recovering 10 minutes after pulselessness have raised concerns about the Lazarus phenomenon, or autoresuscitation, but are based in patients after cardiopulmonary resuscitation was terminated.
The present study, known as Death Prediction and Physiology after Removal of Therapy (DePParRT), enrolled patients at 20 intensive care sites in Canada, the Czech Republic, and the Netherlands, only if surrogate decision-makers agreed on withdrawal of life-sustaining measures without CPR and imminent death was anticipated.
As reported Jan. 28 in the New England Journal of Medicine, physicians observed resumption of circulation or cardiac activity prospectively in 1% of 631 patients based on bedside ECG, arterial pressure catheter monitors, palpated arterial pulse, breaths, or physical movements.
A retrospective review of data from 480 patients with complete ECG and arterial waveforms and at least 5 minutes of continuous waveform monitoring after pulselessness showed resumption of cardiac activity in 14% of patients.
The longest period of pulselessness before the heart showed signs of activity again was 4 minutes and 20 seconds. “So that was a reassuring number, because that’s within our 5-minute window that we currently use,” Dr. Dhanani said.
Importantly, “nobody woke up, nobody ended up being resuscitated, and all of these individuals died. And I think that’s going to be very helpful in this context,” he added.
In all, there were 77 cessations and resumptions in 67 of the 480 patients. The median duration of resumed cardiac activity was 3.9 seconds but, notably, ranged from 1 second to 13 minutes and 14 seconds.
“Though surprising, I think maybe not unreasonable,” observed Dr. Dhanani. “The heart is a very robust organ, and we maybe should anticipate these things happening, where at the end of life the heart may restart for minutes.”
In this situation, it’s important to wait the 13 minutes for the heart to stop again and then “wait another 5 minutes to make sure it doesn’t restart before determining death,” he said. “I think that’s where this study is going to now inform policy makers and guidelines, especially in the context of donations.”
The findings will be taken as strong support for the 5-minute window, said Robert Truog, MD, director of the Harvard Medical School Center for Bioethics and the Frances Glessner Lee Professor of Medical Ethics, Anaesthesia, and Pediatrics, Boston.
“I think it’s a safe point, I think people will refer to it, and it will be used to support the 5-minute window, and that’s probably reasonable,” he told this news organization. “Certainly, if it’s read in Europe it will cut the time from 10 minutes to 5 minutes, and that’s a good thing because 10 minutes is a very long time to wait.”
He noted that the 5-minute window provides reasonable assurance to the public and, with new technologies, permits most organs to be usable for donation after cardiac death. That said, there’s nothing magical about the number.
“In some ways I see this paper as providing interesting data but not actually providing an answer, because from the patient’s perspective and from the recipient’s perspective, waiting until the heart has made its last squeeze may not be the most relevant ethical question,” Dr. Truog said. “It may be, once we know this patient is not going to have return of cardiorespiratory function, is not going to wake up, that’s the point at which we ought to focus on organ preservation and organ retrieval, and that can be much sooner than 5 minutes.”
Dr. Dhanani and colleagues note that the generalizability of the results might be limited because patients without arterial pressure catheters were excluded, and 24% of enrolled patients could not be included in the retrospective waveform analysis owing to incomplete data.
“Our study definition of cardiac activity used an arbitrary threshold of pulse pressure (less than 5 mm Hg) that does not imply meaningful circulation,” they add. “This conservative consensus definition may have been partially responsible for the ostensibly high incidence (14%) of transient resumptions of cardiac activity identified through waveform adjudication.”
The study was supported by the Canadian Institutes for Health Research as part of the Canadian Donation and Transplantation Research Program, CHEO Research Institute, and Karel Pavlík Foundation. Dr. Dhanani has consulted for Canadian Blood Services. Dr. Truog reports no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Among critically ill patients pulseless after planned withdrawal of life-sustaining therapies, cardiac activity restarted in 14% of cases, research shows.
Reassuringly, most resumption of heart activity happened in the first 1-2 minutes and most lasted 1 or 2 seconds.
“The reason we wanted to look at death determination specifically is we know that the stories persist about people coming back to life following death, and that’s not just in the public, it’s in the medical community as well,” lead author Sonny Dhanani, MD, of Children’s Hospital of Eastern Ontario, Ottawa, said in an interview.
“We thought that if we provided scientific evidence of whether this happened or not, we might dispel some myths and misunderstanding, which would hopefully promote organ donation.”
About 70% of organ donations occur after brain death, but an increasing number follow circulatory determination of death, he noted. Most protocols recommend 5 minutes of apnea and pulselessness by arterial catheter monitor before declaring death. But practices vary from 10 minutes in some European countries to 75 seconds in infant heart donors at one Colorado hospital.
Reports of patients recovering 10 minutes after pulselessness have raised concerns about the Lazarus phenomenon, or autoresuscitation, but are based in patients after cardiopulmonary resuscitation was terminated.
The present study, known as Death Prediction and Physiology after Removal of Therapy (DePParRT), enrolled patients at 20 intensive care sites in Canada, the Czech Republic, and the Netherlands, only if surrogate decision-makers agreed on withdrawal of life-sustaining measures without CPR and imminent death was anticipated.
As reported Jan. 28 in the New England Journal of Medicine, physicians observed resumption of circulation or cardiac activity prospectively in 1% of 631 patients based on bedside ECG, arterial pressure catheter monitors, palpated arterial pulse, breaths, or physical movements.
A retrospective review of data from 480 patients with complete ECG and arterial waveforms and at least 5 minutes of continuous waveform monitoring after pulselessness showed resumption of cardiac activity in 14% of patients.
The longest period of pulselessness before the heart showed signs of activity again was 4 minutes and 20 seconds. “So that was a reassuring number, because that’s within our 5-minute window that we currently use,” Dr. Dhanani said.
Importantly, “nobody woke up, nobody ended up being resuscitated, and all of these individuals died. And I think that’s going to be very helpful in this context,” he added.
In all, there were 77 cessations and resumptions in 67 of the 480 patients. The median duration of resumed cardiac activity was 3.9 seconds but, notably, ranged from 1 second to 13 minutes and 14 seconds.
“Though surprising, I think maybe not unreasonable,” observed Dr. Dhanani. “The heart is a very robust organ, and we maybe should anticipate these things happening, where at the end of life the heart may restart for minutes.”
In this situation, it’s important to wait the 13 minutes for the heart to stop again and then “wait another 5 minutes to make sure it doesn’t restart before determining death,” he said. “I think that’s where this study is going to now inform policy makers and guidelines, especially in the context of donations.”
The findings will be taken as strong support for the 5-minute window, said Robert Truog, MD, director of the Harvard Medical School Center for Bioethics and the Frances Glessner Lee Professor of Medical Ethics, Anaesthesia, and Pediatrics, Boston.
“I think it’s a safe point, I think people will refer to it, and it will be used to support the 5-minute window, and that’s probably reasonable,” he told this news organization. “Certainly, if it’s read in Europe it will cut the time from 10 minutes to 5 minutes, and that’s a good thing because 10 minutes is a very long time to wait.”
He noted that the 5-minute window provides reasonable assurance to the public and, with new technologies, permits most organs to be usable for donation after cardiac death. That said, there’s nothing magical about the number.
“In some ways I see this paper as providing interesting data but not actually providing an answer, because from the patient’s perspective and from the recipient’s perspective, waiting until the heart has made its last squeeze may not be the most relevant ethical question,” Dr. Truog said. “It may be, once we know this patient is not going to have return of cardiorespiratory function, is not going to wake up, that’s the point at which we ought to focus on organ preservation and organ retrieval, and that can be much sooner than 5 minutes.”
Dr. Dhanani and colleagues note that the generalizability of the results might be limited because patients without arterial pressure catheters were excluded, and 24% of enrolled patients could not be included in the retrospective waveform analysis owing to incomplete data.
“Our study definition of cardiac activity used an arbitrary threshold of pulse pressure (less than 5 mm Hg) that does not imply meaningful circulation,” they add. “This conservative consensus definition may have been partially responsible for the ostensibly high incidence (14%) of transient resumptions of cardiac activity identified through waveform adjudication.”
The study was supported by the Canadian Institutes for Health Research as part of the Canadian Donation and Transplantation Research Program, CHEO Research Institute, and Karel Pavlík Foundation. Dr. Dhanani has consulted for Canadian Blood Services. Dr. Truog reports no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Poor sensitivity for blood cultures drawn after antibiotics
Background: Early antibiotic administration reduces mortality in patients with severe sepsis. Administering antibiotics before blood cultures could potentially decrease time to treatment and improve outcomes, but the diagnostic yield of blood cultures drawn shortly after antibiotics is unknown.
Study design: Prospective, patient-level, pre- and post-study.
Setting: Multicenter study in USA & Canada.
Synopsis: During 2013-2018, 330 adult patients were recruited from seven urban EDs. Patients with severe manifestations of sepsis (spontaneous bacterial peritonitis [SBP] less than 90 mm Hg and lactic acid of 4 or more) had blood cultures drawn before and after empiric antibiotic administration. Blood cultures were positive for one or more microbial pathogens in 31.4% of patients when drawn before antibiotics and in 19.4% of patients when drawn after antibiotics (absolute difference of 12.0% (95% confidence interval, 5.4%-18.6%; P less than .001). The sensitivity of blood cultures after antibiotic administration was 52.9% (95% CI, 43%-63%).
There were several study limitations including: lack of sequential recruitment, lower than expected proportion of bacteremic patients, and variation in blood culture collection. Despite this, the magnitude of the findings are convincing and support current practice.
Bottom line: Continue to obtain blood cultures before antibiotics.
Citation: Cheng MP et al. Blood culture results before and after antimicrobial administration in patients with severe manifestations of sepsis. Ann Intern Med. 2019 Oct 15;171(8):547-54.
Dr. Waner is clinical instructor of medicine, hospital medicine, at the Rocky Mountain Veterans Affairs Regional Medical Center, Aurora, Colo.
Background: Early antibiotic administration reduces mortality in patients with severe sepsis. Administering antibiotics before blood cultures could potentially decrease time to treatment and improve outcomes, but the diagnostic yield of blood cultures drawn shortly after antibiotics is unknown.
Study design: Prospective, patient-level, pre- and post-study.
Setting: Multicenter study in USA & Canada.
Synopsis: During 2013-2018, 330 adult patients were recruited from seven urban EDs. Patients with severe manifestations of sepsis (spontaneous bacterial peritonitis [SBP] less than 90 mm Hg and lactic acid of 4 or more) had blood cultures drawn before and after empiric antibiotic administration. Blood cultures were positive for one or more microbial pathogens in 31.4% of patients when drawn before antibiotics and in 19.4% of patients when drawn after antibiotics (absolute difference of 12.0% (95% confidence interval, 5.4%-18.6%; P less than .001). The sensitivity of blood cultures after antibiotic administration was 52.9% (95% CI, 43%-63%).
There were several study limitations including: lack of sequential recruitment, lower than expected proportion of bacteremic patients, and variation in blood culture collection. Despite this, the magnitude of the findings are convincing and support current practice.
Bottom line: Continue to obtain blood cultures before antibiotics.
Citation: Cheng MP et al. Blood culture results before and after antimicrobial administration in patients with severe manifestations of sepsis. Ann Intern Med. 2019 Oct 15;171(8):547-54.
Dr. Waner is clinical instructor of medicine, hospital medicine, at the Rocky Mountain Veterans Affairs Regional Medical Center, Aurora, Colo.
Background: Early antibiotic administration reduces mortality in patients with severe sepsis. Administering antibiotics before blood cultures could potentially decrease time to treatment and improve outcomes, but the diagnostic yield of blood cultures drawn shortly after antibiotics is unknown.
Study design: Prospective, patient-level, pre- and post-study.
Setting: Multicenter study in USA & Canada.
Synopsis: During 2013-2018, 330 adult patients were recruited from seven urban EDs. Patients with severe manifestations of sepsis (spontaneous bacterial peritonitis [SBP] less than 90 mm Hg and lactic acid of 4 or more) had blood cultures drawn before and after empiric antibiotic administration. Blood cultures were positive for one or more microbial pathogens in 31.4% of patients when drawn before antibiotics and in 19.4% of patients when drawn after antibiotics (absolute difference of 12.0% (95% confidence interval, 5.4%-18.6%; P less than .001). The sensitivity of blood cultures after antibiotic administration was 52.9% (95% CI, 43%-63%).
There were several study limitations including: lack of sequential recruitment, lower than expected proportion of bacteremic patients, and variation in blood culture collection. Despite this, the magnitude of the findings are convincing and support current practice.
Bottom line: Continue to obtain blood cultures before antibiotics.
Citation: Cheng MP et al. Blood culture results before and after antimicrobial administration in patients with severe manifestations of sepsis. Ann Intern Med. 2019 Oct 15;171(8):547-54.
Dr. Waner is clinical instructor of medicine, hospital medicine, at the Rocky Mountain Veterans Affairs Regional Medical Center, Aurora, Colo.
Rheumatologic disease activity an important influencer of COVID-19 death risk
People with rheumatic and musculoskeletal diseases (RMDs) who contract the SARS-CoV-2 virus appear more likely to die from COVID-19 if their rheumatologic condition is not being well controlled at the time of their infection.
New data from the COVID-19 Global Rheumatology Alliance (GRA) physician registry reported in Annals of the Rheumatic Diseases have found that the odds of dying from COVID-19 were 87% higher in individuals recorded as having moderate to high disease activity versus those reported to be in remission or having low disease activity.
“I think this really highlights the importance of continuing to appropriately, and actively, treat our patients, and the importance of controlling their disease,” Pedro Machado, MD, PhD, said in an interview. Dr. Machado, an associate professor in rheumatology and muscle diseases at University College London and a consultant rheumatologist at several U.K. hospitals, has been involved in the GRA physician registry from the start, and sits on the GRA steering committee.
Alongside higher disease activity, several other important factors were found to be associated with increased odds of dying from COVID-19 – older age, male gender, and the presence of one or more comorbidities, such as hypertension combined with cardiovascular disease or chronic lung disease.
These demographic and disease-based factors have been linked to an increased risk for COVID-19–related hospitalization before, both in people with RMDs and in the general population, but the latest GRA physician registry data now take that a step further, and link them also to an increased risk for death, together with several other factors more specific to RMDs.
Logging COVID-19 rheumatologic cases
Since the start of the global pandemic, the potential effects that SARS-CoV-2 infection might have on people with RMDs in particular has concerned the rheumatology community. The main worries being that, either because of the underlying RMD itself or to its treatment, there may be immunoregulatory deficits or other risk factors that would make individuals more susceptible to not only infection but also to developing more severe COVID-19 than the general population.
These concerns led to the rapid formation of the GRA and the COVID-19 GRA physician registry in March 2020 to collect and analyze data on adults with rheumatic disease and confirmed or presumptive COVID-19. Entries into the registry are made by or under the direction of rheumatologists, and this is a voluntary process.
“This population cannot ever be entirely representative of the population of patients with rheumatic diseases,” Dr. Machado acknowledged. There will be selection and other biases that affect the reported data. That said, it’s the largest database of reported COVID-19 cases in adult rheumatology patients across the world, with more than 9,000 cases so far included from multiple registries, including those based in Europe and North and South America. Data from one of these – the French RMD cohort – have also recently been published in Annals of the Rheumatic Diseases, showing much the same findings but on a national level.
Hospitalization was the focus of a previous report because “you need large sample sizes” to look at endpoints that occur less frequently. When the first analysis was done, there were around 600 cases from 40 countries in the registry with sufficient data that could be used. Now, with a greater number of recorded cases, factors influencing the risk for death could be examined.
Death rate and risk factors found
Data on 3,729 COVID-19 cases in people with RMDs were included in the current analysis, all recorded in the first few months of the registry being open and up until July 1, 2020. In all, 390 (10.5%) of people died. While this is “clearly higher” than reported in the general population in most countries, the analysis was not designed to calculate a precise estimate.
“It should not be taken as an estimate of the overall death rate among patients with rheumatic diseases and COVID-19,” Dr. Machado and coauthors have been keen to point out.
“Age is always the biggest risk factor,” Dr. Machado explained. “There’s always a gradient: the older the patient, the worse the outcome.”
Indeed, there was a threefold increased risk for death among those aged 66-75 years versus those who were 65 years or younger (odds ratio, 3.00), and a sixfold increased risk for patients older than 75, compared with the younger age group (OR, 6.18).
Having both hypertension and cardiovascular disease was associated with an OR of 1.89, and coexisting chronic lung disease also significantly increased the chances of dying from COVID-19 (OR, 1.68).
Being of male sex was associated with a 46% increased risk for death from COVID-19 versus being of female sex.
The risk for COVID-19 death also rose with the use of corticosteroids. Compared with no steroid use, there was a 69% increased risk for with death at doses of 10 mg or more prednisolone equivalent per day.
“The finding about moderate to high doses of steroids being associated with a worse outcome is consistent with the first report; it was the same for hospitalization,” Dr. Machado observed.
The general consensus on steroid use in the COVID-19 setting is that they should be continued as needed, but at the lowest possible dose, as outlined in provisional recommendations set out by the recently renamed European Alliance of Associations for Rheumatology.
The GRA physician registry findings provide further support for this, suggesting that disease control should be optimized with disease-modifying antirheumatic drugs, ideally without increasing the dose of steroids.
Surprise over sulfasalazine risk
“Taking all medications into account – such as methotrexate, leflunomide, hydroxychloroquine, [tumor necrosis factor] blockers, interleukin-6 blockers, and [Janus kinase] inhibitors – it is quite reassuring because we did not see an association with worse outcome with those drugs overall,” Dr. Machado said.
However, treatment with rituximab (OR, 4.0), sulfasalazine (OR, 3.6), and immunosuppressive agents such as azathioprine, cyclophosphamide, cyclosporine, mycophenolate, or tacrolimus (OR, 2.2), were associated with higher odds of dying from COVID-19 when compared with treatment with methotrexate alone.
The findings for rituximab and immunosuppressant use were perhaps not unexpected, but the possible association between sulfasalazine and COVID-19 death was “a bit intriguing,” Dr. Machado observed. “Sulfasalazine is believed to have low immunosuppressive effect.”
This warrants further investigation, but there are likely a range of confounding factors at play. One could be that people considered to be at higher risk may have been more often prescribed sulfasalazine because it was thought to be less immunosuppressive. Another might be because people taking sulfasalazine were more likely to be smokers, and they were also not advised to protect themselves from exposure to the virus (shielding) during the first wave of the pandemic, at least not in the United Kingdom.
Rituximab caution and vaccination
“Rituximab is a concern,” Dr. Machado acknowledged. “It is a concern that rheumatologists are now aware of and they are addressing, but then it’s a concern for a very specific subgroup of patients.”
While rheumatologists are, and will continue to prescribe it, there will be even more careful consideration over when, in whom, and how to use it during, and possibly even after, the pandemic.
“COVID is here to stay, it will become endemic, and it’s going to be part of our lives like the flu virus is,” Dr. Machado predicted.
Then there is the issue on vaccinating people against COVID-19, should those on rituximab still receive it? The answer is a yes, but, as with other vaccinations it’s all about the timing of when the vaccination is given.
Societies such as the British Society for Rheumatology have already begun to include guidance on this, recommending one of the available COVID-19 vaccines is given at least a month before the next or first dose of rituximab is due. As rituximab is given every few months, with doses sometimes spaced as much as 9 months or even a year apart, this should not be too much of a problem, but it is “better to have the vaccine first,” Dr. Machado said.
Has COVID-19 care improved in RMDs?
In separate research published in The Lancet Rheumatology, April Jorge, MD, of Massachusetts General Hospital and Harvard Medical School, both in Boston, and associates found that the risks of severe COVID-19 outcomes have improved over time, although they still “remain substantial.”
Dr. Jorge and colleagues looked at temporal trends in COVID-19 outcomes in patients with RMDs over the course of the first 6 months of the pandemic in 2020, using data from a large, multicenter, electronic health record network (TriNetX).
They formed two patient cohorts – a late (diagnosed from April 20 to July 20) and an early (diagnosed from January 20 to April 20) cohort – to see if outcomes had improved and discovered lower relative risks among patients in the late cohort for hospitalization (0.67), admission to the ICU (0.56), mechanical ventilation (0.39), acute kidney injury (0.66), renal replacement (0.53), and death (0.39).
“These results are encouraging,” but it’s difficult to match these different populations of patients, Dr. Machado said. “There are always factors that you cannot match for” and were not included in the U.S. analysis.
While there are important caveats in how the analysis was performed and thus in interpreting these data, they do “suggest that one of the reasons why outcomes have improved is because we have become better at treating these patients,” Dr. Machado added.
“Our treatment has improved, and our capacity to treat the complications has improved. We understand better how the disease behaves – we know that they can have thromboembolic complications that we can manage, and we are now able to manage ventilation issues better.”
Moreover, Dr. Machado said that, not only were clinicians more aware of what they should or should not do, there were treatments that were being used routinely or in some cases based on recent clinical trial results. “I think we are indeed treating these patients better.”
The COVID-19 GRA physician registry is financially supported by the American College of Rheumatology and EULAR. Dr. Machado had no relevant conflicts of interest.
People with rheumatic and musculoskeletal diseases (RMDs) who contract the SARS-CoV-2 virus appear more likely to die from COVID-19 if their rheumatologic condition is not being well controlled at the time of their infection.
New data from the COVID-19 Global Rheumatology Alliance (GRA) physician registry reported in Annals of the Rheumatic Diseases have found that the odds of dying from COVID-19 were 87% higher in individuals recorded as having moderate to high disease activity versus those reported to be in remission or having low disease activity.
“I think this really highlights the importance of continuing to appropriately, and actively, treat our patients, and the importance of controlling their disease,” Pedro Machado, MD, PhD, said in an interview. Dr. Machado, an associate professor in rheumatology and muscle diseases at University College London and a consultant rheumatologist at several U.K. hospitals, has been involved in the GRA physician registry from the start, and sits on the GRA steering committee.
Alongside higher disease activity, several other important factors were found to be associated with increased odds of dying from COVID-19 – older age, male gender, and the presence of one or more comorbidities, such as hypertension combined with cardiovascular disease or chronic lung disease.
These demographic and disease-based factors have been linked to an increased risk for COVID-19–related hospitalization before, both in people with RMDs and in the general population, but the latest GRA physician registry data now take that a step further, and link them also to an increased risk for death, together with several other factors more specific to RMDs.
Logging COVID-19 rheumatologic cases
Since the start of the global pandemic, the potential effects that SARS-CoV-2 infection might have on people with RMDs in particular has concerned the rheumatology community. The main worries being that, either because of the underlying RMD itself or to its treatment, there may be immunoregulatory deficits or other risk factors that would make individuals more susceptible to not only infection but also to developing more severe COVID-19 than the general population.
These concerns led to the rapid formation of the GRA and the COVID-19 GRA physician registry in March 2020 to collect and analyze data on adults with rheumatic disease and confirmed or presumptive COVID-19. Entries into the registry are made by or under the direction of rheumatologists, and this is a voluntary process.
“This population cannot ever be entirely representative of the population of patients with rheumatic diseases,” Dr. Machado acknowledged. There will be selection and other biases that affect the reported data. That said, it’s the largest database of reported COVID-19 cases in adult rheumatology patients across the world, with more than 9,000 cases so far included from multiple registries, including those based in Europe and North and South America. Data from one of these – the French RMD cohort – have also recently been published in Annals of the Rheumatic Diseases, showing much the same findings but on a national level.
Hospitalization was the focus of a previous report because “you need large sample sizes” to look at endpoints that occur less frequently. When the first analysis was done, there were around 600 cases from 40 countries in the registry with sufficient data that could be used. Now, with a greater number of recorded cases, factors influencing the risk for death could be examined.
Death rate and risk factors found
Data on 3,729 COVID-19 cases in people with RMDs were included in the current analysis, all recorded in the first few months of the registry being open and up until July 1, 2020. In all, 390 (10.5%) of people died. While this is “clearly higher” than reported in the general population in most countries, the analysis was not designed to calculate a precise estimate.
“It should not be taken as an estimate of the overall death rate among patients with rheumatic diseases and COVID-19,” Dr. Machado and coauthors have been keen to point out.
“Age is always the biggest risk factor,” Dr. Machado explained. “There’s always a gradient: the older the patient, the worse the outcome.”
Indeed, there was a threefold increased risk for death among those aged 66-75 years versus those who were 65 years or younger (odds ratio, 3.00), and a sixfold increased risk for patients older than 75, compared with the younger age group (OR, 6.18).
Having both hypertension and cardiovascular disease was associated with an OR of 1.89, and coexisting chronic lung disease also significantly increased the chances of dying from COVID-19 (OR, 1.68).
Being of male sex was associated with a 46% increased risk for death from COVID-19 versus being of female sex.
The risk for COVID-19 death also rose with the use of corticosteroids. Compared with no steroid use, there was a 69% increased risk for with death at doses of 10 mg or more prednisolone equivalent per day.
“The finding about moderate to high doses of steroids being associated with a worse outcome is consistent with the first report; it was the same for hospitalization,” Dr. Machado observed.
The general consensus on steroid use in the COVID-19 setting is that they should be continued as needed, but at the lowest possible dose, as outlined in provisional recommendations set out by the recently renamed European Alliance of Associations for Rheumatology.
The GRA physician registry findings provide further support for this, suggesting that disease control should be optimized with disease-modifying antirheumatic drugs, ideally without increasing the dose of steroids.
Surprise over sulfasalazine risk
“Taking all medications into account – such as methotrexate, leflunomide, hydroxychloroquine, [tumor necrosis factor] blockers, interleukin-6 blockers, and [Janus kinase] inhibitors – it is quite reassuring because we did not see an association with worse outcome with those drugs overall,” Dr. Machado said.
However, treatment with rituximab (OR, 4.0), sulfasalazine (OR, 3.6), and immunosuppressive agents such as azathioprine, cyclophosphamide, cyclosporine, mycophenolate, or tacrolimus (OR, 2.2), were associated with higher odds of dying from COVID-19 when compared with treatment with methotrexate alone.
The findings for rituximab and immunosuppressant use were perhaps not unexpected, but the possible association between sulfasalazine and COVID-19 death was “a bit intriguing,” Dr. Machado observed. “Sulfasalazine is believed to have low immunosuppressive effect.”
This warrants further investigation, but there are likely a range of confounding factors at play. One could be that people considered to be at higher risk may have been more often prescribed sulfasalazine because it was thought to be less immunosuppressive. Another might be because people taking sulfasalazine were more likely to be smokers, and they were also not advised to protect themselves from exposure to the virus (shielding) during the first wave of the pandemic, at least not in the United Kingdom.
Rituximab caution and vaccination
“Rituximab is a concern,” Dr. Machado acknowledged. “It is a concern that rheumatologists are now aware of and they are addressing, but then it’s a concern for a very specific subgroup of patients.”
While rheumatologists are, and will continue to prescribe it, there will be even more careful consideration over when, in whom, and how to use it during, and possibly even after, the pandemic.
“COVID is here to stay, it will become endemic, and it’s going to be part of our lives like the flu virus is,” Dr. Machado predicted.
Then there is the issue on vaccinating people against COVID-19, should those on rituximab still receive it? The answer is a yes, but, as with other vaccinations it’s all about the timing of when the vaccination is given.
Societies such as the British Society for Rheumatology have already begun to include guidance on this, recommending one of the available COVID-19 vaccines is given at least a month before the next or first dose of rituximab is due. As rituximab is given every few months, with doses sometimes spaced as much as 9 months or even a year apart, this should not be too much of a problem, but it is “better to have the vaccine first,” Dr. Machado said.
Has COVID-19 care improved in RMDs?
In separate research published in The Lancet Rheumatology, April Jorge, MD, of Massachusetts General Hospital and Harvard Medical School, both in Boston, and associates found that the risks of severe COVID-19 outcomes have improved over time, although they still “remain substantial.”
Dr. Jorge and colleagues looked at temporal trends in COVID-19 outcomes in patients with RMDs over the course of the first 6 months of the pandemic in 2020, using data from a large, multicenter, electronic health record network (TriNetX).
They formed two patient cohorts – a late (diagnosed from April 20 to July 20) and an early (diagnosed from January 20 to April 20) cohort – to see if outcomes had improved and discovered lower relative risks among patients in the late cohort for hospitalization (0.67), admission to the ICU (0.56), mechanical ventilation (0.39), acute kidney injury (0.66), renal replacement (0.53), and death (0.39).
“These results are encouraging,” but it’s difficult to match these different populations of patients, Dr. Machado said. “There are always factors that you cannot match for” and were not included in the U.S. analysis.
While there are important caveats in how the analysis was performed and thus in interpreting these data, they do “suggest that one of the reasons why outcomes have improved is because we have become better at treating these patients,” Dr. Machado added.
“Our treatment has improved, and our capacity to treat the complications has improved. We understand better how the disease behaves – we know that they can have thromboembolic complications that we can manage, and we are now able to manage ventilation issues better.”
Moreover, Dr. Machado said that, not only were clinicians more aware of what they should or should not do, there were treatments that were being used routinely or in some cases based on recent clinical trial results. “I think we are indeed treating these patients better.”
The COVID-19 GRA physician registry is financially supported by the American College of Rheumatology and EULAR. Dr. Machado had no relevant conflicts of interest.
People with rheumatic and musculoskeletal diseases (RMDs) who contract the SARS-CoV-2 virus appear more likely to die from COVID-19 if their rheumatologic condition is not being well controlled at the time of their infection.
New data from the COVID-19 Global Rheumatology Alliance (GRA) physician registry reported in Annals of the Rheumatic Diseases have found that the odds of dying from COVID-19 were 87% higher in individuals recorded as having moderate to high disease activity versus those reported to be in remission or having low disease activity.
“I think this really highlights the importance of continuing to appropriately, and actively, treat our patients, and the importance of controlling their disease,” Pedro Machado, MD, PhD, said in an interview. Dr. Machado, an associate professor in rheumatology and muscle diseases at University College London and a consultant rheumatologist at several U.K. hospitals, has been involved in the GRA physician registry from the start, and sits on the GRA steering committee.
Alongside higher disease activity, several other important factors were found to be associated with increased odds of dying from COVID-19 – older age, male gender, and the presence of one or more comorbidities, such as hypertension combined with cardiovascular disease or chronic lung disease.
These demographic and disease-based factors have been linked to an increased risk for COVID-19–related hospitalization before, both in people with RMDs and in the general population, but the latest GRA physician registry data now take that a step further, and link them also to an increased risk for death, together with several other factors more specific to RMDs.
Logging COVID-19 rheumatologic cases
Since the start of the global pandemic, the potential effects that SARS-CoV-2 infection might have on people with RMDs in particular has concerned the rheumatology community. The main worries being that, either because of the underlying RMD itself or to its treatment, there may be immunoregulatory deficits or other risk factors that would make individuals more susceptible to not only infection but also to developing more severe COVID-19 than the general population.
These concerns led to the rapid formation of the GRA and the COVID-19 GRA physician registry in March 2020 to collect and analyze data on adults with rheumatic disease and confirmed or presumptive COVID-19. Entries into the registry are made by or under the direction of rheumatologists, and this is a voluntary process.
“This population cannot ever be entirely representative of the population of patients with rheumatic diseases,” Dr. Machado acknowledged. There will be selection and other biases that affect the reported data. That said, it’s the largest database of reported COVID-19 cases in adult rheumatology patients across the world, with more than 9,000 cases so far included from multiple registries, including those based in Europe and North and South America. Data from one of these – the French RMD cohort – have also recently been published in Annals of the Rheumatic Diseases, showing much the same findings but on a national level.
Hospitalization was the focus of a previous report because “you need large sample sizes” to look at endpoints that occur less frequently. When the first analysis was done, there were around 600 cases from 40 countries in the registry with sufficient data that could be used. Now, with a greater number of recorded cases, factors influencing the risk for death could be examined.
Death rate and risk factors found
Data on 3,729 COVID-19 cases in people with RMDs were included in the current analysis, all recorded in the first few months of the registry being open and up until July 1, 2020. In all, 390 (10.5%) of people died. While this is “clearly higher” than reported in the general population in most countries, the analysis was not designed to calculate a precise estimate.
“It should not be taken as an estimate of the overall death rate among patients with rheumatic diseases and COVID-19,” Dr. Machado and coauthors have been keen to point out.
“Age is always the biggest risk factor,” Dr. Machado explained. “There’s always a gradient: the older the patient, the worse the outcome.”
Indeed, there was a threefold increased risk for death among those aged 66-75 years versus those who were 65 years or younger (odds ratio, 3.00), and a sixfold increased risk for patients older than 75, compared with the younger age group (OR, 6.18).
Having both hypertension and cardiovascular disease was associated with an OR of 1.89, and coexisting chronic lung disease also significantly increased the chances of dying from COVID-19 (OR, 1.68).
Being of male sex was associated with a 46% increased risk for death from COVID-19 versus being of female sex.
The risk for COVID-19 death also rose with the use of corticosteroids. Compared with no steroid use, there was a 69% increased risk for with death at doses of 10 mg or more prednisolone equivalent per day.
“The finding about moderate to high doses of steroids being associated with a worse outcome is consistent with the first report; it was the same for hospitalization,” Dr. Machado observed.
The general consensus on steroid use in the COVID-19 setting is that they should be continued as needed, but at the lowest possible dose, as outlined in provisional recommendations set out by the recently renamed European Alliance of Associations for Rheumatology.
The GRA physician registry findings provide further support for this, suggesting that disease control should be optimized with disease-modifying antirheumatic drugs, ideally without increasing the dose of steroids.
Surprise over sulfasalazine risk
“Taking all medications into account – such as methotrexate, leflunomide, hydroxychloroquine, [tumor necrosis factor] blockers, interleukin-6 blockers, and [Janus kinase] inhibitors – it is quite reassuring because we did not see an association with worse outcome with those drugs overall,” Dr. Machado said.
However, treatment with rituximab (OR, 4.0), sulfasalazine (OR, 3.6), and immunosuppressive agents such as azathioprine, cyclophosphamide, cyclosporine, mycophenolate, or tacrolimus (OR, 2.2), were associated with higher odds of dying from COVID-19 when compared with treatment with methotrexate alone.
The findings for rituximab and immunosuppressant use were perhaps not unexpected, but the possible association between sulfasalazine and COVID-19 death was “a bit intriguing,” Dr. Machado observed. “Sulfasalazine is believed to have low immunosuppressive effect.”
This warrants further investigation, but there are likely a range of confounding factors at play. One could be that people considered to be at higher risk may have been more often prescribed sulfasalazine because it was thought to be less immunosuppressive. Another might be because people taking sulfasalazine were more likely to be smokers, and they were also not advised to protect themselves from exposure to the virus (shielding) during the first wave of the pandemic, at least not in the United Kingdom.
Rituximab caution and vaccination
“Rituximab is a concern,” Dr. Machado acknowledged. “It is a concern that rheumatologists are now aware of and they are addressing, but then it’s a concern for a very specific subgroup of patients.”
While rheumatologists are, and will continue to prescribe it, there will be even more careful consideration over when, in whom, and how to use it during, and possibly even after, the pandemic.
“COVID is here to stay, it will become endemic, and it’s going to be part of our lives like the flu virus is,” Dr. Machado predicted.
Then there is the issue on vaccinating people against COVID-19, should those on rituximab still receive it? The answer is a yes, but, as with other vaccinations it’s all about the timing of when the vaccination is given.
Societies such as the British Society for Rheumatology have already begun to include guidance on this, recommending one of the available COVID-19 vaccines is given at least a month before the next or first dose of rituximab is due. As rituximab is given every few months, with doses sometimes spaced as much as 9 months or even a year apart, this should not be too much of a problem, but it is “better to have the vaccine first,” Dr. Machado said.
Has COVID-19 care improved in RMDs?
In separate research published in The Lancet Rheumatology, April Jorge, MD, of Massachusetts General Hospital and Harvard Medical School, both in Boston, and associates found that the risks of severe COVID-19 outcomes have improved over time, although they still “remain substantial.”
Dr. Jorge and colleagues looked at temporal trends in COVID-19 outcomes in patients with RMDs over the course of the first 6 months of the pandemic in 2020, using data from a large, multicenter, electronic health record network (TriNetX).
They formed two patient cohorts – a late (diagnosed from April 20 to July 20) and an early (diagnosed from January 20 to April 20) cohort – to see if outcomes had improved and discovered lower relative risks among patients in the late cohort for hospitalization (0.67), admission to the ICU (0.56), mechanical ventilation (0.39), acute kidney injury (0.66), renal replacement (0.53), and death (0.39).
“These results are encouraging,” but it’s difficult to match these different populations of patients, Dr. Machado said. “There are always factors that you cannot match for” and were not included in the U.S. analysis.
While there are important caveats in how the analysis was performed and thus in interpreting these data, they do “suggest that one of the reasons why outcomes have improved is because we have become better at treating these patients,” Dr. Machado added.
“Our treatment has improved, and our capacity to treat the complications has improved. We understand better how the disease behaves – we know that they can have thromboembolic complications that we can manage, and we are now able to manage ventilation issues better.”
Moreover, Dr. Machado said that, not only were clinicians more aware of what they should or should not do, there were treatments that were being used routinely or in some cases based on recent clinical trial results. “I think we are indeed treating these patients better.”
The COVID-19 GRA physician registry is financially supported by the American College of Rheumatology and EULAR. Dr. Machado had no relevant conflicts of interest.
FROM ANNALS OF THE RHEUMATIC DISEASES











