User login
Metastatic Brain Tumors
Series Editor: Arthur T. Skarin, MD, FACP, FCCP
Systemic cancer can affect the central nervous system in several different ways, including direct tumor metastasis and indirect remote effects. Intracranial metastasis can involve the skull, dura, and leptomeninges (arachnoid and pia mater), as well as the brain parenchyma. Of these, parenchymal brain metastases are the most common and have been found in as many as 24% of cancer patients in autopsy studies. It has been reported that metastatic brain tumors outnumber primary brain tumors 10 to 1.
To read the full article in PDF:
Series Editor: Arthur T. Skarin, MD, FACP, FCCP
Systemic cancer can affect the central nervous system in several different ways, including direct tumor metastasis and indirect remote effects. Intracranial metastasis can involve the skull, dura, and leptomeninges (arachnoid and pia mater), as well as the brain parenchyma. Of these, parenchymal brain metastases are the most common and have been found in as many as 24% of cancer patients in autopsy studies. It has been reported that metastatic brain tumors outnumber primary brain tumors 10 to 1.
To read the full article in PDF:
Series Editor: Arthur T. Skarin, MD, FACP, FCCP
Systemic cancer can affect the central nervous system in several different ways, including direct tumor metastasis and indirect remote effects. Intracranial metastasis can involve the skull, dura, and leptomeninges (arachnoid and pia mater), as well as the brain parenchyma. Of these, parenchymal brain metastases are the most common and have been found in as many as 24% of cancer patients in autopsy studies. It has been reported that metastatic brain tumors outnumber primary brain tumors 10 to 1.
To read the full article in PDF:
A summary of the new ACOG report on neonatal brachial plexus palsy. Part 2: Pathophysiology and causation
Obstetricians are often blamed for causing neonatal brachial plexus palsy (NBPP). For that reason, understanding the true pathophysiology and causation of this birth-related entity is of extreme importance.
In Part 1 of this two-part series, I summarized findings from the new report on NBPP from the American College of Obstetricians and Gynecologists (ACOG), focusing on whether the phenomenon of shoulder dystocia and NBPP can be predicted or prevented.1 Here, in Part 2, I focus on ACOG’s conclusions concerning pathophysiology and causation of NBPP, as well as the College’s recommendations for applying that knowledge to practice.
Some infants are more susceptible than others to the forces of labor and delivery
Babies emerge from the uterus and maternal pelvis by a combination of uterine contractions and maternal pushing (endogenous forces) aided by the traction forces applied by the birth attendant (exogenous forces). Research over the past 2 decades has shown that endogenous forces play a significant—if not dominant—role in the causation of NBPP.
Stretching and potential injury to the brachial plexus occur when the long axis of the fetus is pushed down the birth canal while either the maternal symphysis pubis or sacral promontory catches and holds either the anterior or posterior shoulder of the fetus, respectively. This conjunction of events generates a stretching force on the tissues that connect the fetal trunk and head—the neck—under which lies the brachial plexus. The same anatomic relationships and labor forces also vigorously compress the fetal neck against the maternal symphysis pubis or sacral promontory and may cause compression injury. Any traction applied by the clinician accentuates these stretching and pressure forces acting on the nerves of the brachial plexus.
How the neonate responds to these forces depends on the tensile strength of its tissues, the metabolic condition of the fetus after a potentially long labor (as measured by acid-base status), the degree of protective muscle tone around the fetal shoulder and neck, and other fluctuating conditions. In other words, because of the many variables involved, some fetuses are more or less susceptible to injury than others.
Maternal forces alone can cause NBPP
The ACOG report1 makes an important statement:
Some plaintiff attorneys and their expert witnesses have tried to make the case that, although endogenous forces can cause temporary brachial plexus injuries, they cannot cause permanent brachial plexus injuries. However, as the ACOG report goes on to state:
The report acknowledges that the clinician can increase brachial plexus stretch by applying downward lateral traction to the neonate’s head during delivery efforts. However, contrary to claims often made by the plaintiff bar, in the presence of shoulder dystocia, even properly applied axial traction will necessarily increase the stretching of the brachial plexus. The report also notes that traction applied in the plane of the fetal cervicothoracic spine typically is along a vector estimated to be 25° to 45° below the horizontal plane of a woman in lithotomy position, not in an exact straight line with the maternal trunk. This degree of delivery force below the horizon is defined as normal “axial traction.”
Exogenous forces have yet to be definitively measured
Multiple attempts have been made to quantify the amount of force applied by clinicians in various delivery scenarios. However, in the published studies in which this force has been “measured,” the accuracy of the findings has not been validated. The three studies in which delivery force was directly measured in a clinical setting “provide a limited assessment of exogenous forces” and “do not address the angle at which forces were applied.”3–5 All other studies used artificial models.
As a result, few conclusions from such studies are directly applicable to the clinical arena. Moreover, in other studies using simulated birth scenarios, there was no feedback to participating clinicians as to whether the force they applied would have been sufficient to deliver the “fetus.” It was therefore difficult for participants in such studies to “determine how the situation corresponds with the force they would apply clinically.”1
Cadaver studies have been inadequate to assess the in situ response of the brachial plexus
Many plaintiff claims regarding the cause of brachial plexus injury use cadaver studies as evidence. However, most such studies were conducted between 98 and 140 years ago. In these older studies, quantitative evaluation was rare. And in the few more recent studies, there are several reasons why the data obtained are problematic:
- the nerves being studied were dissected free from supporting tissues
- nerve tissue deteriorates quickly postmortem
- some studies used adult tissues; there may be significant differences between adult and newborn nerve tissue that obscure comparison.
The ACOG report concludes the section on cadaver studies by stating:
Physical models also fall short
The problem with the use of physical models in evaluating NBPP centers on the need to find materials that have the same or similar properties as the tissues of interest. These sorts of bioengineering limitations generally do not allow for findings that have direct clinical applicability.
Of interest, however, is the finding of at least two groups of investigators that less traction is required when simulating delivery of a model infant when rotational maneuvers (Rubin’s) are employed rather than after McRoberts repositioning.
Computer models have yielded data on the relative effects of endogenous and exogenous forces
Sophisticated computer analysis has been used to investigate both endogenous and exogenous delivery forces. Results of such studies have shown that maternal endogenous forces exert twice as much pressure on the base of the fetal neck against the maternal symphysis pubis as do deliverer-induced exogenous forces.
Is there a threshold of force?
Data that include measurement of the force applied to the brachial plexus nerves of a live infant during a real delivery are almost nonexistent. One group—on the basis of a single case of transient NBPP and potentially flawed pressure measurements—has suggested that the threshold for NBPP in the human is 100 Newtons.3 However, other studies have shown that physician-applied forces in routine deliveries commonly exceed this hypothesized cutoff—yet the rate of NBPP remains low. In measuring delivery forces it must be remembered that significant variation exists between individual neonates, both in terms of mechanical properties and anatomy. Because of this variation—and the nonlinear behavior of nerve tissues—the specific force needed to cause a nerve injury or rupture in a given neonate has not been established.
Chapter 3 of the ACOG report closes with a statement:
NBPP and shoulder dystocia
Shoulder dystocia is defined as a delivery that requires additional obstetric maneuvers after gentle downward traction on the fetal head fails to deliver the fetal shoulders. The ACOG report makes the important point that shoulder dystocia is not formally diagnosed until a trial of downward axial traction has been unsuccessful in delivering the anterior shoulder. This point is a refutation of the frequent plaintiff claim that, once a shoulder dystocia is thought to be present, no traction whatsoever should be applied by the clinician at any time during the remainder of the delivery.
Shoulder dystocia incidence is rising
The reported incidence of shoulder dystocia has increased over the past several decades. It is unclear whether this increase is related to maternal obesity, fetal macrosomia, or more widespread reporting. However, paradoxes exist in the relationship among risk factors, shoulder dystocia, and brachial plexus injury:
- although there is an increased incidence of shoulder dystocia with increased birth weight, the mean birth weight of neonates with recognized shoulder dystocia is not significantly higher than the mean birth weight of all term infants
- strategies to reduce NBPP by preventing shoulder dystocia—including early induction of labor and prophylactic use of McRoberts maneuver and suprapubic pressure—have not been effective in reducing the incidence of NBPP.
The ACOG report makes the statement: “Maternal and fetal factors associated with shoulder dystocia do not allow for reliable prediction of persistent NBPP.”1
What is optimal management of shoulder dystocia?
The last obstetric part of the ACOG report takes as its focus the management of shoulder dystocia. It discusses the importance of communication among members of the delivery team and with the mother whose neonate is experiencing a shoulder dystocia. The report states:
This statement contrasts with claims frequently made by plaintiff medical expert witnesses that the woman experiencing a shoulder dystocia should absolutely cease from pushing.
In a section on team training, the report describes the delivery team’s priorities:
- resolving the shoulder dystocia
- avoiding neonatal hypoxic-ischemic central nervous system injury
- minimizing strain on the neonatal brachial plexus.
Studies evaluating process standardization, the use of checklists, teamwork training, crew resource management, and evidence-based medicine have shown that these tools improve neonatal and maternal outcomes.
Simulation training also has been shown to help reduce transient NBPP (see the box below for more on simulation programs for shoulder dystocia). Whether it also can lower the rate of permanent NBPP is unclear.1
Can simulation training reduce the rate of neonatal brachial plexus injury after shoulder dystocia?
In the new ACOG report on neonatal brachial plexus injury, simulation training is discussed as one solution to the dilemma of how clinicians can gain experience in managing obstetric events that occur infrequently.1 Simulation training also has the potential to improve teamwork, communication, and the situational awareness of the health-care team as a whole. Several studies over the past few years have shown that, in some units, the implementation of simulation training actually has decreased the number of cases of neonatal brachial plexus palsy (NBPP), compared with no simulation training.
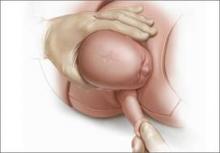
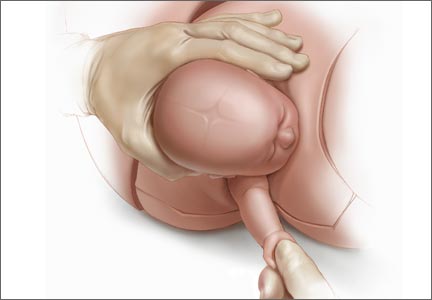
For example, Draycott and colleagues explored the rate of neonatal injury associated with shoulder dystocia before and after implementation of a mandatory 1-day simulation training program at Southmead Hospital in Bristol, United Kingdom.2 The program consisted of practice on a shoulder dystocia training mannequin and covered risk factors, recognition of shoulder dystocia, maneuvers, and documentation. The training used a stepwise approach, beginning with a call for help and continuing through McRoberts’ positioning, suprapubic pressure, and internal maneuvers such as delivery of the posterior arm (Figure).
There were 15,908 births in the pretraining period and 13,117 in the posttraining period, with shoulder dystocia rates comparable between the two periods. Not only did clinical management of shoulder dystocia improve after training, but there was a significant reduction in neonatal injury at birth after shoulder dystocia (30 injuries of 324 shoulder dystocia cases [9.3%] before training vs six injuries of 262 shoulder dystocia cases [2.3%] afterward).2
In another study of obstetric brachial plexus injury before and after implementation of simulation training for shoulder dystocia, Inglis and colleagues found a decline in the rate of such injury from 30% to 10.67% (P<.01).3 Shoulder dystocia training remained associated with reduced obstetric brachial plexus injury after logistic-regression analysis.3
Shoulder dystocia training is now recommended by the Joint Commission on Accreditation of Healthcare Organizations in the United States. However, in its report, ACOG concludes—despite studies from Draycott and colleagues and others—that, owing to “limited data,” “there remains no evidence that introduction of simulation can reduce the frequency of persistent NBPP.”1
References
- American College of Obstetricians and Gynecologists. Executive summary: neonatal brachial plexus palsy. Report of the American College of Obstetricians and Gynecologists’ Task Force on neonatal brachial plexus palsy. Obstet Gynecol. 2014;123(4):902–904.
- Draycott TJ, Crofts FJ, Ash JP, et al. Improving neonatal outcome through practical shoulder dystocia training. Obstet Gynecol. 2008;112(1):14–20.
- Inglis SR, Feier N, Chetiyaar JB, et al. Effects of shoulder dystocia training on the incidence of brachial plexus palsy. Am J Obstet Gynecol. 2011;204(4):322.e1–e6.
Delivery of the posterior arm
The report reaffirms the previous statement from the ACOG practice bulletin on shoulder dystocia, which asserts that no specific sequence of maneuvers for resolving shoulder dystocia has been shown to be superior to any other.6 It does note, however, that recent studies seem to demonstrate a benefit when delivery of the posterior arm is prioritized over the usual first-line maneuvers of McRoberts positioning and the application of suprapubic pressure. If confirmed, such findings may alter the standard of care for shoulder dystocia resolution and result in a change in ACOG recommendations.
Documentation may be enhanced by use of a checklist
The ACOG report stresses the importance of accurate, contemporaneous documentation of the management of shoulder dystocia, observing that checklists and documentation reminders help ensure the completeness and relevance of notes after shoulder dystocia deliveries and NBPP. ACOG has produced such a checklist, which can be found in the appendix of the report itself.1
How long before central neurologic injury occurs?
Another issue covered in the report is how long a clinician has to resolve a shoulder dystocia before central neurologic damage occurs. Studies have shown that permanent neurologic injury can occur as soon as 2 minutes after shoulder impaction, although the risk of acidosis or severe hypoxic-ischemic encephalopathy remains low until impaction has lasted at least 5 minutes.
Other issues covered in the report
The last chapters of the ACOG report focus on orthopedic aspects of brachial plexus injury, including diagnosis, treatment, and prognosis.
The report concludes with a glossary and three appendices:
- Royal College of Obstetricians and Gynecologists Green Top Guidebook #42 on shoulder dystocia
- ACOG Practice Bulletin #40 on shoulder dystocia
- ACOG Patient Safety Checklist #6 on the documentation of shoulder dystocia.
Why the ACOG report is foundational
The ACOG report on NBPP is an important and much-needed document. It includes a comprehensive review of the literature on brachial plexus injury and shoulder dystocia, written by nationally recognized experts in the field. Most important, it makes definitive statements that counteract false and dubious claims often made by the plaintiff bar in brachial plexus injury cases and provides evidence to back those statements.
The report:
- disproves the claim that “excessive” physician traction is the only etiology of brachial plexus injuries
- demonstrates that no differentiation can be made between the etiology of permanent versus temporary brachial plexus injuries
- describes how brachial plexus injuries can occur in the absence of physician traction or even of shoulder dystocia
- provides a summary of scientific information about brachial plexus injuries that will benefit obstetric clinicians
- provides a wealth of literature documentation that will enable physician defendants to counteract many of the claims plaintiffs and their expert witnesses make in brachial plexus injury cases.
The report is—and will remain—a foundational document in obstetrics for many years to come.
Share your thoughts on this article! Send your Letter to the Editor to [email protected].
1. American College of Obstetricians and Gynecologists. Executive summary: neonatal brachial plexus palsy. Report of the American College of Obstetricians and Gynecologists’ Task Force on neonatal brachial plexus palsy. Obstet Gynecol. 2014;123(4):902–904.
2. Lerner HM, Salamon E. Permanent brachial plexus injury following vaginal delivery without physician traction or shoulder dystocia. Am J Obstet Gynecol. 2008;198(3):e.7–e.8.
3. Allen R, Sorab J, Gonik B. Risk factors for shoulder dystocia: an engineering study of clinician-applied forces. Obstet Gynecol. 1991;77(3):352–355.
4. Poggi SH, Allen RH, Patel CR, Ghidini A, Pezzullo JC, Spong CY. Randomized trial of McRoberts versus lithotomy positioning to decrease the force that is applied to the fetus during delivery. Am J Obstet Gynecol. 2004;191(3):874–878.
5. Poggi SH, Allen RH, Patel C, et al. Effect of epidural anaesthesia on clinician-applied force during vaginal delivery. Am J Obstet Gynecol. 2004;191(3):903–906.
6. American College of Obstetricians and Gynecologists. Practice bulletin #40: shoulder dystocia. Obstet Gynecol. 2002;100(5 pt 1):1045–1050.
Obstetricians are often blamed for causing neonatal brachial plexus palsy (NBPP). For that reason, understanding the true pathophysiology and causation of this birth-related entity is of extreme importance.
In Part 1 of this two-part series, I summarized findings from the new report on NBPP from the American College of Obstetricians and Gynecologists (ACOG), focusing on whether the phenomenon of shoulder dystocia and NBPP can be predicted or prevented.1 Here, in Part 2, I focus on ACOG’s conclusions concerning pathophysiology and causation of NBPP, as well as the College’s recommendations for applying that knowledge to practice.
Some infants are more susceptible than others to the forces of labor and delivery
Babies emerge from the uterus and maternal pelvis by a combination of uterine contractions and maternal pushing (endogenous forces) aided by the traction forces applied by the birth attendant (exogenous forces). Research over the past 2 decades has shown that endogenous forces play a significant—if not dominant—role in the causation of NBPP.
Stretching and potential injury to the brachial plexus occur when the long axis of the fetus is pushed down the birth canal while either the maternal symphysis pubis or sacral promontory catches and holds either the anterior or posterior shoulder of the fetus, respectively. This conjunction of events generates a stretching force on the tissues that connect the fetal trunk and head—the neck—under which lies the brachial plexus. The same anatomic relationships and labor forces also vigorously compress the fetal neck against the maternal symphysis pubis or sacral promontory and may cause compression injury. Any traction applied by the clinician accentuates these stretching and pressure forces acting on the nerves of the brachial plexus.
How the neonate responds to these forces depends on the tensile strength of its tissues, the metabolic condition of the fetus after a potentially long labor (as measured by acid-base status), the degree of protective muscle tone around the fetal shoulder and neck, and other fluctuating conditions. In other words, because of the many variables involved, some fetuses are more or less susceptible to injury than others.
Maternal forces alone can cause NBPP
The ACOG report1 makes an important statement:
Some plaintiff attorneys and their expert witnesses have tried to make the case that, although endogenous forces can cause temporary brachial plexus injuries, they cannot cause permanent brachial plexus injuries. However, as the ACOG report goes on to state:
The report acknowledges that the clinician can increase brachial plexus stretch by applying downward lateral traction to the neonate’s head during delivery efforts. However, contrary to claims often made by the plaintiff bar, in the presence of shoulder dystocia, even properly applied axial traction will necessarily increase the stretching of the brachial plexus. The report also notes that traction applied in the plane of the fetal cervicothoracic spine typically is along a vector estimated to be 25° to 45° below the horizontal plane of a woman in lithotomy position, not in an exact straight line with the maternal trunk. This degree of delivery force below the horizon is defined as normal “axial traction.”
Exogenous forces have yet to be definitively measured
Multiple attempts have been made to quantify the amount of force applied by clinicians in various delivery scenarios. However, in the published studies in which this force has been “measured,” the accuracy of the findings has not been validated. The three studies in which delivery force was directly measured in a clinical setting “provide a limited assessment of exogenous forces” and “do not address the angle at which forces were applied.”3–5 All other studies used artificial models.
As a result, few conclusions from such studies are directly applicable to the clinical arena. Moreover, in other studies using simulated birth scenarios, there was no feedback to participating clinicians as to whether the force they applied would have been sufficient to deliver the “fetus.” It was therefore difficult for participants in such studies to “determine how the situation corresponds with the force they would apply clinically.”1
Cadaver studies have been inadequate to assess the in situ response of the brachial plexus
Many plaintiff claims regarding the cause of brachial plexus injury use cadaver studies as evidence. However, most such studies were conducted between 98 and 140 years ago. In these older studies, quantitative evaluation was rare. And in the few more recent studies, there are several reasons why the data obtained are problematic:
- the nerves being studied were dissected free from supporting tissues
- nerve tissue deteriorates quickly postmortem
- some studies used adult tissues; there may be significant differences between adult and newborn nerve tissue that obscure comparison.
The ACOG report concludes the section on cadaver studies by stating:
Physical models also fall short
The problem with the use of physical models in evaluating NBPP centers on the need to find materials that have the same or similar properties as the tissues of interest. These sorts of bioengineering limitations generally do not allow for findings that have direct clinical applicability.
Of interest, however, is the finding of at least two groups of investigators that less traction is required when simulating delivery of a model infant when rotational maneuvers (Rubin’s) are employed rather than after McRoberts repositioning.
Computer models have yielded data on the relative effects of endogenous and exogenous forces
Sophisticated computer analysis has been used to investigate both endogenous and exogenous delivery forces. Results of such studies have shown that maternal endogenous forces exert twice as much pressure on the base of the fetal neck against the maternal symphysis pubis as do deliverer-induced exogenous forces.
Is there a threshold of force?
Data that include measurement of the force applied to the brachial plexus nerves of a live infant during a real delivery are almost nonexistent. One group—on the basis of a single case of transient NBPP and potentially flawed pressure measurements—has suggested that the threshold for NBPP in the human is 100 Newtons.3 However, other studies have shown that physician-applied forces in routine deliveries commonly exceed this hypothesized cutoff—yet the rate of NBPP remains low. In measuring delivery forces it must be remembered that significant variation exists between individual neonates, both in terms of mechanical properties and anatomy. Because of this variation—and the nonlinear behavior of nerve tissues—the specific force needed to cause a nerve injury or rupture in a given neonate has not been established.
Chapter 3 of the ACOG report closes with a statement:
NBPP and shoulder dystocia
Shoulder dystocia is defined as a delivery that requires additional obstetric maneuvers after gentle downward traction on the fetal head fails to deliver the fetal shoulders. The ACOG report makes the important point that shoulder dystocia is not formally diagnosed until a trial of downward axial traction has been unsuccessful in delivering the anterior shoulder. This point is a refutation of the frequent plaintiff claim that, once a shoulder dystocia is thought to be present, no traction whatsoever should be applied by the clinician at any time during the remainder of the delivery.
Shoulder dystocia incidence is rising
The reported incidence of shoulder dystocia has increased over the past several decades. It is unclear whether this increase is related to maternal obesity, fetal macrosomia, or more widespread reporting. However, paradoxes exist in the relationship among risk factors, shoulder dystocia, and brachial plexus injury:
- although there is an increased incidence of shoulder dystocia with increased birth weight, the mean birth weight of neonates with recognized shoulder dystocia is not significantly higher than the mean birth weight of all term infants
- strategies to reduce NBPP by preventing shoulder dystocia—including early induction of labor and prophylactic use of McRoberts maneuver and suprapubic pressure—have not been effective in reducing the incidence of NBPP.
The ACOG report makes the statement: “Maternal and fetal factors associated with shoulder dystocia do not allow for reliable prediction of persistent NBPP.”1
What is optimal management of shoulder dystocia?
The last obstetric part of the ACOG report takes as its focus the management of shoulder dystocia. It discusses the importance of communication among members of the delivery team and with the mother whose neonate is experiencing a shoulder dystocia. The report states:
This statement contrasts with claims frequently made by plaintiff medical expert witnesses that the woman experiencing a shoulder dystocia should absolutely cease from pushing.
In a section on team training, the report describes the delivery team’s priorities:
- resolving the shoulder dystocia
- avoiding neonatal hypoxic-ischemic central nervous system injury
- minimizing strain on the neonatal brachial plexus.
Studies evaluating process standardization, the use of checklists, teamwork training, crew resource management, and evidence-based medicine have shown that these tools improve neonatal and maternal outcomes.
Simulation training also has been shown to help reduce transient NBPP (see the box below for more on simulation programs for shoulder dystocia). Whether it also can lower the rate of permanent NBPP is unclear.1
Can simulation training reduce the rate of neonatal brachial plexus injury after shoulder dystocia?
In the new ACOG report on neonatal brachial plexus injury, simulation training is discussed as one solution to the dilemma of how clinicians can gain experience in managing obstetric events that occur infrequently.1 Simulation training also has the potential to improve teamwork, communication, and the situational awareness of the health-care team as a whole. Several studies over the past few years have shown that, in some units, the implementation of simulation training actually has decreased the number of cases of neonatal brachial plexus palsy (NBPP), compared with no simulation training.
For example, Draycott and colleagues explored the rate of neonatal injury associated with shoulder dystocia before and after implementation of a mandatory 1-day simulation training program at Southmead Hospital in Bristol, United Kingdom.2 The program consisted of practice on a shoulder dystocia training mannequin and covered risk factors, recognition of shoulder dystocia, maneuvers, and documentation. The training used a stepwise approach, beginning with a call for help and continuing through McRoberts’ positioning, suprapubic pressure, and internal maneuvers such as delivery of the posterior arm (Figure).
There were 15,908 births in the pretraining period and 13,117 in the posttraining period, with shoulder dystocia rates comparable between the two periods. Not only did clinical management of shoulder dystocia improve after training, but there was a significant reduction in neonatal injury at birth after shoulder dystocia (30 injuries of 324 shoulder dystocia cases [9.3%] before training vs six injuries of 262 shoulder dystocia cases [2.3%] afterward).2
In another study of obstetric brachial plexus injury before and after implementation of simulation training for shoulder dystocia, Inglis and colleagues found a decline in the rate of such injury from 30% to 10.67% (P<.01).3 Shoulder dystocia training remained associated with reduced obstetric brachial plexus injury after logistic-regression analysis.3
Shoulder dystocia training is now recommended by the Joint Commission on Accreditation of Healthcare Organizations in the United States. However, in its report, ACOG concludes—despite studies from Draycott and colleagues and others—that, owing to “limited data,” “there remains no evidence that introduction of simulation can reduce the frequency of persistent NBPP.”1
References
- American College of Obstetricians and Gynecologists. Executive summary: neonatal brachial plexus palsy. Report of the American College of Obstetricians and Gynecologists’ Task Force on neonatal brachial plexus palsy. Obstet Gynecol. 2014;123(4):902–904.
- Draycott TJ, Crofts FJ, Ash JP, et al. Improving neonatal outcome through practical shoulder dystocia training. Obstet Gynecol. 2008;112(1):14–20.
- Inglis SR, Feier N, Chetiyaar JB, et al. Effects of shoulder dystocia training on the incidence of brachial plexus palsy. Am J Obstet Gynecol. 2011;204(4):322.e1–e6.
Delivery of the posterior arm
The report reaffirms the previous statement from the ACOG practice bulletin on shoulder dystocia, which asserts that no specific sequence of maneuvers for resolving shoulder dystocia has been shown to be superior to any other.6 It does note, however, that recent studies seem to demonstrate a benefit when delivery of the posterior arm is prioritized over the usual first-line maneuvers of McRoberts positioning and the application of suprapubic pressure. If confirmed, such findings may alter the standard of care for shoulder dystocia resolution and result in a change in ACOG recommendations.
Documentation may be enhanced by use of a checklist
The ACOG report stresses the importance of accurate, contemporaneous documentation of the management of shoulder dystocia, observing that checklists and documentation reminders help ensure the completeness and relevance of notes after shoulder dystocia deliveries and NBPP. ACOG has produced such a checklist, which can be found in the appendix of the report itself.1
How long before central neurologic injury occurs?
Another issue covered in the report is how long a clinician has to resolve a shoulder dystocia before central neurologic damage occurs. Studies have shown that permanent neurologic injury can occur as soon as 2 minutes after shoulder impaction, although the risk of acidosis or severe hypoxic-ischemic encephalopathy remains low until impaction has lasted at least 5 minutes.
Other issues covered in the report
The last chapters of the ACOG report focus on orthopedic aspects of brachial plexus injury, including diagnosis, treatment, and prognosis.
The report concludes with a glossary and three appendices:
- Royal College of Obstetricians and Gynecologists Green Top Guidebook #42 on shoulder dystocia
- ACOG Practice Bulletin #40 on shoulder dystocia
- ACOG Patient Safety Checklist #6 on the documentation of shoulder dystocia.
Why the ACOG report is foundational
The ACOG report on NBPP is an important and much-needed document. It includes a comprehensive review of the literature on brachial plexus injury and shoulder dystocia, written by nationally recognized experts in the field. Most important, it makes definitive statements that counteract false and dubious claims often made by the plaintiff bar in brachial plexus injury cases and provides evidence to back those statements.
The report:
- disproves the claim that “excessive” physician traction is the only etiology of brachial plexus injuries
- demonstrates that no differentiation can be made between the etiology of permanent versus temporary brachial plexus injuries
- describes how brachial plexus injuries can occur in the absence of physician traction or even of shoulder dystocia
- provides a summary of scientific information about brachial plexus injuries that will benefit obstetric clinicians
- provides a wealth of literature documentation that will enable physician defendants to counteract many of the claims plaintiffs and their expert witnesses make in brachial plexus injury cases.
The report is—and will remain—a foundational document in obstetrics for many years to come.
Share your thoughts on this article! Send your Letter to the Editor to [email protected].
Obstetricians are often blamed for causing neonatal brachial plexus palsy (NBPP). For that reason, understanding the true pathophysiology and causation of this birth-related entity is of extreme importance.
In Part 1 of this two-part series, I summarized findings from the new report on NBPP from the American College of Obstetricians and Gynecologists (ACOG), focusing on whether the phenomenon of shoulder dystocia and NBPP can be predicted or prevented.1 Here, in Part 2, I focus on ACOG’s conclusions concerning pathophysiology and causation of NBPP, as well as the College’s recommendations for applying that knowledge to practice.
Some infants are more susceptible than others to the forces of labor and delivery
Babies emerge from the uterus and maternal pelvis by a combination of uterine contractions and maternal pushing (endogenous forces) aided by the traction forces applied by the birth attendant (exogenous forces). Research over the past 2 decades has shown that endogenous forces play a significant—if not dominant—role in the causation of NBPP.
Stretching and potential injury to the brachial plexus occur when the long axis of the fetus is pushed down the birth canal while either the maternal symphysis pubis or sacral promontory catches and holds either the anterior or posterior shoulder of the fetus, respectively. This conjunction of events generates a stretching force on the tissues that connect the fetal trunk and head—the neck—under which lies the brachial plexus. The same anatomic relationships and labor forces also vigorously compress the fetal neck against the maternal symphysis pubis or sacral promontory and may cause compression injury. Any traction applied by the clinician accentuates these stretching and pressure forces acting on the nerves of the brachial plexus.
How the neonate responds to these forces depends on the tensile strength of its tissues, the metabolic condition of the fetus after a potentially long labor (as measured by acid-base status), the degree of protective muscle tone around the fetal shoulder and neck, and other fluctuating conditions. In other words, because of the many variables involved, some fetuses are more or less susceptible to injury than others.
Maternal forces alone can cause NBPP
The ACOG report1 makes an important statement:
Some plaintiff attorneys and their expert witnesses have tried to make the case that, although endogenous forces can cause temporary brachial plexus injuries, they cannot cause permanent brachial plexus injuries. However, as the ACOG report goes on to state:
The report acknowledges that the clinician can increase brachial plexus stretch by applying downward lateral traction to the neonate’s head during delivery efforts. However, contrary to claims often made by the plaintiff bar, in the presence of shoulder dystocia, even properly applied axial traction will necessarily increase the stretching of the brachial plexus. The report also notes that traction applied in the plane of the fetal cervicothoracic spine typically is along a vector estimated to be 25° to 45° below the horizontal plane of a woman in lithotomy position, not in an exact straight line with the maternal trunk. This degree of delivery force below the horizon is defined as normal “axial traction.”
Exogenous forces have yet to be definitively measured
Multiple attempts have been made to quantify the amount of force applied by clinicians in various delivery scenarios. However, in the published studies in which this force has been “measured,” the accuracy of the findings has not been validated. The three studies in which delivery force was directly measured in a clinical setting “provide a limited assessment of exogenous forces” and “do not address the angle at which forces were applied.”3–5 All other studies used artificial models.
As a result, few conclusions from such studies are directly applicable to the clinical arena. Moreover, in other studies using simulated birth scenarios, there was no feedback to participating clinicians as to whether the force they applied would have been sufficient to deliver the “fetus.” It was therefore difficult for participants in such studies to “determine how the situation corresponds with the force they would apply clinically.”1
Cadaver studies have been inadequate to assess the in situ response of the brachial plexus
Many plaintiff claims regarding the cause of brachial plexus injury use cadaver studies as evidence. However, most such studies were conducted between 98 and 140 years ago. In these older studies, quantitative evaluation was rare. And in the few more recent studies, there are several reasons why the data obtained are problematic:
- the nerves being studied were dissected free from supporting tissues
- nerve tissue deteriorates quickly postmortem
- some studies used adult tissues; there may be significant differences between adult and newborn nerve tissue that obscure comparison.
The ACOG report concludes the section on cadaver studies by stating:
Physical models also fall short
The problem with the use of physical models in evaluating NBPP centers on the need to find materials that have the same or similar properties as the tissues of interest. These sorts of bioengineering limitations generally do not allow for findings that have direct clinical applicability.
Of interest, however, is the finding of at least two groups of investigators that less traction is required when simulating delivery of a model infant when rotational maneuvers (Rubin’s) are employed rather than after McRoberts repositioning.
Computer models have yielded data on the relative effects of endogenous and exogenous forces
Sophisticated computer analysis has been used to investigate both endogenous and exogenous delivery forces. Results of such studies have shown that maternal endogenous forces exert twice as much pressure on the base of the fetal neck against the maternal symphysis pubis as do deliverer-induced exogenous forces.
Is there a threshold of force?
Data that include measurement of the force applied to the brachial plexus nerves of a live infant during a real delivery are almost nonexistent. One group—on the basis of a single case of transient NBPP and potentially flawed pressure measurements—has suggested that the threshold for NBPP in the human is 100 Newtons.3 However, other studies have shown that physician-applied forces in routine deliveries commonly exceed this hypothesized cutoff—yet the rate of NBPP remains low. In measuring delivery forces it must be remembered that significant variation exists between individual neonates, both in terms of mechanical properties and anatomy. Because of this variation—and the nonlinear behavior of nerve tissues—the specific force needed to cause a nerve injury or rupture in a given neonate has not been established.
Chapter 3 of the ACOG report closes with a statement:
NBPP and shoulder dystocia
Shoulder dystocia is defined as a delivery that requires additional obstetric maneuvers after gentle downward traction on the fetal head fails to deliver the fetal shoulders. The ACOG report makes the important point that shoulder dystocia is not formally diagnosed until a trial of downward axial traction has been unsuccessful in delivering the anterior shoulder. This point is a refutation of the frequent plaintiff claim that, once a shoulder dystocia is thought to be present, no traction whatsoever should be applied by the clinician at any time during the remainder of the delivery.
Shoulder dystocia incidence is rising
The reported incidence of shoulder dystocia has increased over the past several decades. It is unclear whether this increase is related to maternal obesity, fetal macrosomia, or more widespread reporting. However, paradoxes exist in the relationship among risk factors, shoulder dystocia, and brachial plexus injury:
- although there is an increased incidence of shoulder dystocia with increased birth weight, the mean birth weight of neonates with recognized shoulder dystocia is not significantly higher than the mean birth weight of all term infants
- strategies to reduce NBPP by preventing shoulder dystocia—including early induction of labor and prophylactic use of McRoberts maneuver and suprapubic pressure—have not been effective in reducing the incidence of NBPP.
The ACOG report makes the statement: “Maternal and fetal factors associated with shoulder dystocia do not allow for reliable prediction of persistent NBPP.”1
What is optimal management of shoulder dystocia?
The last obstetric part of the ACOG report takes as its focus the management of shoulder dystocia. It discusses the importance of communication among members of the delivery team and with the mother whose neonate is experiencing a shoulder dystocia. The report states:
This statement contrasts with claims frequently made by plaintiff medical expert witnesses that the woman experiencing a shoulder dystocia should absolutely cease from pushing.
In a section on team training, the report describes the delivery team’s priorities:
- resolving the shoulder dystocia
- avoiding neonatal hypoxic-ischemic central nervous system injury
- minimizing strain on the neonatal brachial plexus.
Studies evaluating process standardization, the use of checklists, teamwork training, crew resource management, and evidence-based medicine have shown that these tools improve neonatal and maternal outcomes.
Simulation training also has been shown to help reduce transient NBPP (see the box below for more on simulation programs for shoulder dystocia). Whether it also can lower the rate of permanent NBPP is unclear.1
Can simulation training reduce the rate of neonatal brachial plexus injury after shoulder dystocia?
In the new ACOG report on neonatal brachial plexus injury, simulation training is discussed as one solution to the dilemma of how clinicians can gain experience in managing obstetric events that occur infrequently.1 Simulation training also has the potential to improve teamwork, communication, and the situational awareness of the health-care team as a whole. Several studies over the past few years have shown that, in some units, the implementation of simulation training actually has decreased the number of cases of neonatal brachial plexus palsy (NBPP), compared with no simulation training.
For example, Draycott and colleagues explored the rate of neonatal injury associated with shoulder dystocia before and after implementation of a mandatory 1-day simulation training program at Southmead Hospital in Bristol, United Kingdom.2 The program consisted of practice on a shoulder dystocia training mannequin and covered risk factors, recognition of shoulder dystocia, maneuvers, and documentation. The training used a stepwise approach, beginning with a call for help and continuing through McRoberts’ positioning, suprapubic pressure, and internal maneuvers such as delivery of the posterior arm (Figure).
There were 15,908 births in the pretraining period and 13,117 in the posttraining period, with shoulder dystocia rates comparable between the two periods. Not only did clinical management of shoulder dystocia improve after training, but there was a significant reduction in neonatal injury at birth after shoulder dystocia (30 injuries of 324 shoulder dystocia cases [9.3%] before training vs six injuries of 262 shoulder dystocia cases [2.3%] afterward).2
In another study of obstetric brachial plexus injury before and after implementation of simulation training for shoulder dystocia, Inglis and colleagues found a decline in the rate of such injury from 30% to 10.67% (P<.01).3 Shoulder dystocia training remained associated with reduced obstetric brachial plexus injury after logistic-regression analysis.3
Shoulder dystocia training is now recommended by the Joint Commission on Accreditation of Healthcare Organizations in the United States. However, in its report, ACOG concludes—despite studies from Draycott and colleagues and others—that, owing to “limited data,” “there remains no evidence that introduction of simulation can reduce the frequency of persistent NBPP.”1
References
- American College of Obstetricians and Gynecologists. Executive summary: neonatal brachial plexus palsy. Report of the American College of Obstetricians and Gynecologists’ Task Force on neonatal brachial plexus palsy. Obstet Gynecol. 2014;123(4):902–904.
- Draycott TJ, Crofts FJ, Ash JP, et al. Improving neonatal outcome through practical shoulder dystocia training. Obstet Gynecol. 2008;112(1):14–20.
- Inglis SR, Feier N, Chetiyaar JB, et al. Effects of shoulder dystocia training on the incidence of brachial plexus palsy. Am J Obstet Gynecol. 2011;204(4):322.e1–e6.
Delivery of the posterior arm
The report reaffirms the previous statement from the ACOG practice bulletin on shoulder dystocia, which asserts that no specific sequence of maneuvers for resolving shoulder dystocia has been shown to be superior to any other.6 It does note, however, that recent studies seem to demonstrate a benefit when delivery of the posterior arm is prioritized over the usual first-line maneuvers of McRoberts positioning and the application of suprapubic pressure. If confirmed, such findings may alter the standard of care for shoulder dystocia resolution and result in a change in ACOG recommendations.
Documentation may be enhanced by use of a checklist
The ACOG report stresses the importance of accurate, contemporaneous documentation of the management of shoulder dystocia, observing that checklists and documentation reminders help ensure the completeness and relevance of notes after shoulder dystocia deliveries and NBPP. ACOG has produced such a checklist, which can be found in the appendix of the report itself.1
How long before central neurologic injury occurs?
Another issue covered in the report is how long a clinician has to resolve a shoulder dystocia before central neurologic damage occurs. Studies have shown that permanent neurologic injury can occur as soon as 2 minutes after shoulder impaction, although the risk of acidosis or severe hypoxic-ischemic encephalopathy remains low until impaction has lasted at least 5 minutes.
Other issues covered in the report
The last chapters of the ACOG report focus on orthopedic aspects of brachial plexus injury, including diagnosis, treatment, and prognosis.
The report concludes with a glossary and three appendices:
- Royal College of Obstetricians and Gynecologists Green Top Guidebook #42 on shoulder dystocia
- ACOG Practice Bulletin #40 on shoulder dystocia
- ACOG Patient Safety Checklist #6 on the documentation of shoulder dystocia.
Why the ACOG report is foundational
The ACOG report on NBPP is an important and much-needed document. It includes a comprehensive review of the literature on brachial plexus injury and shoulder dystocia, written by nationally recognized experts in the field. Most important, it makes definitive statements that counteract false and dubious claims often made by the plaintiff bar in brachial plexus injury cases and provides evidence to back those statements.
The report:
- disproves the claim that “excessive” physician traction is the only etiology of brachial plexus injuries
- demonstrates that no differentiation can be made between the etiology of permanent versus temporary brachial plexus injuries
- describes how brachial plexus injuries can occur in the absence of physician traction or even of shoulder dystocia
- provides a summary of scientific information about brachial plexus injuries that will benefit obstetric clinicians
- provides a wealth of literature documentation that will enable physician defendants to counteract many of the claims plaintiffs and their expert witnesses make in brachial plexus injury cases.
The report is—and will remain—a foundational document in obstetrics for many years to come.
Share your thoughts on this article! Send your Letter to the Editor to [email protected].
1. American College of Obstetricians and Gynecologists. Executive summary: neonatal brachial plexus palsy. Report of the American College of Obstetricians and Gynecologists’ Task Force on neonatal brachial plexus palsy. Obstet Gynecol. 2014;123(4):902–904.
2. Lerner HM, Salamon E. Permanent brachial plexus injury following vaginal delivery without physician traction or shoulder dystocia. Am J Obstet Gynecol. 2008;198(3):e.7–e.8.
3. Allen R, Sorab J, Gonik B. Risk factors for shoulder dystocia: an engineering study of clinician-applied forces. Obstet Gynecol. 1991;77(3):352–355.
4. Poggi SH, Allen RH, Patel CR, Ghidini A, Pezzullo JC, Spong CY. Randomized trial of McRoberts versus lithotomy positioning to decrease the force that is applied to the fetus during delivery. Am J Obstet Gynecol. 2004;191(3):874–878.
5. Poggi SH, Allen RH, Patel C, et al. Effect of epidural anaesthesia on clinician-applied force during vaginal delivery. Am J Obstet Gynecol. 2004;191(3):903–906.
6. American College of Obstetricians and Gynecologists. Practice bulletin #40: shoulder dystocia. Obstet Gynecol. 2002;100(5 pt 1):1045–1050.
1. American College of Obstetricians and Gynecologists. Executive summary: neonatal brachial plexus palsy. Report of the American College of Obstetricians and Gynecologists’ Task Force on neonatal brachial plexus palsy. Obstet Gynecol. 2014;123(4):902–904.
2. Lerner HM, Salamon E. Permanent brachial plexus injury following vaginal delivery without physician traction or shoulder dystocia. Am J Obstet Gynecol. 2008;198(3):e.7–e.8.
3. Allen R, Sorab J, Gonik B. Risk factors for shoulder dystocia: an engineering study of clinician-applied forces. Obstet Gynecol. 1991;77(3):352–355.
4. Poggi SH, Allen RH, Patel CR, Ghidini A, Pezzullo JC, Spong CY. Randomized trial of McRoberts versus lithotomy positioning to decrease the force that is applied to the fetus during delivery. Am J Obstet Gynecol. 2004;191(3):874–878.
5. Poggi SH, Allen RH, Patel C, et al. Effect of epidural anaesthesia on clinician-applied force during vaginal delivery. Am J Obstet Gynecol. 2004;191(3):903–906.
6. American College of Obstetricians and Gynecologists. Practice bulletin #40: shoulder dystocia. Obstet Gynecol. 2002;100(5 pt 1):1045–1050.
5 Points on Total Ankle Arthroplasty
Patellofemoral Crepitation and Clunk Complicating Posterior-Stabilized Total Knee Arthroplasty
Using Dashboard Technology to Monitor Overdose Risk
On October 10, 2013, a Congressional hearing was held to address the issue of opioid medication prescribing within VHA facilities and clinics (House Veteran Affairs Subcommittee hearing “Between Peril and Promise: Facing the Dangers of VA’s Skyrocketing Use of Prescription Painkillers to Treat Veterans”). Several individuals testified, including the widows of 2 veterans; both their husbands had overdosed on prescribed opioid medications. One husband had been taking as many as 15 pills a day and was additionally prescribed oxycodone/acetaminophen, which led to his death.1
Alongside the widows were 2 veterans who had been treated for chronic back pain injuries sustained before and during deployment in Iraq. Both had been prescribed several pain medications, including oxycodone/acetaminophen, methadone, and morphine. One reported that as his pain increased, his doctors continued to provide him additional prescriptions; at one point he had more than 13 prescriptions and could no longer work from being so “doped up.”1
In the past 2 decades, health care professionals (HCPs) have placed greater emphasis on chronic pain management. As a result, the rate of opioid medication prescribing has increased dramatically. Since 1994, the number of opioid medication prescriptions has nearly doubled; this change has been accompanied by an increase in opioid misuse, which has resulted in accidental or intentional overdose and death.2
Based on a recent National Institute on Drug Abuse (NIDA) report, the greatest impact has been on armed forces personnel.3 Prescriptions for pain relievers quadrupled between 2001 and 2009 to almost 3.8 million within the military population. Although civilian populations are more likely to abuse illicit drugs, military personnel are at particular risk of prescription abuse, including opioid medications.3 In 2008, 11% of armed forces service members reported misusing prescription drugs, with opioid medications being the most abused. This is an approximate 5- to 6-fold increase since 2002 (2% reported misuse in 2002).3 Particularly concerning is the associated rise in suicide rates among armed forces personnel, which surpassed civilian suicide rates in 2004. In 2009, one-third of suicides among armed forces personnel involved prescription drugs.3
Certain patient characteristics or factors are related to greater overdose risk. These risk factors include prescription dosage and frequency, history of suicide attempts or self-harm behavior, history of depression or posttraumatic stress disorder (PTSD) among other mental health-related diagnoses, a history of substance and/or alcohol abuse, and within the context of opioid medication use, the concurrent use of other central nervous system (CNS) depressants.4,5 Additionally, the stresses of deployment during wartime, physical injuries sustained in combat, and the unique military culture play a particularly important role in access to substances with high abuse potential and the subsequent development of substance abuse.3
Opioid Use and Risk Factors
More than 3% of adults in the U.S. are now receiving opioid medications for chronic noncancer pain.6 Substance abuse among patients with chronic pain ranges from 14% to 40%.5 Prescription opioid medications are the fastest growing drugs of abuse and the most common cause of unintentional overdose in the U.S.4 About 17,000 deaths occur each year as a result of prescription opioid medication overdose.7 Opioid medication-related overdose deaths began to increase in the early 2000s and continue to increase. Between 1999 and 2007, the rate of unintentional overdose-related deaths in the U.S. increased by 124%, largely due to the increase of prescription opioid medications.8
High-Dose Opioid Medication Use
A study by Dunn and colleagues found that patients receiving higher doses of prescribed opioid medications were at an increased risk of overdose.6 Patients receiving 50 mg to 99 mg morphine equivalent daily dose (MEDD) had a 3.7-fold increase in overdose risk (0.7% annual overdose rate) as compared with patients who received < 50 mg MEDD (0.2% annual overdose rate). Patients receiving ≥ 100 mg MEDD had a 1.8% annual overdose rate and a 9.8-fold increase in overdose risk as compared with patients who received < 50 mg MEDD. Overall, 51 patients experienced ≥ 1 overdose event, 40 of whom experienced fatal or serious overdoses and 6 of whom attempted suicide. Patients receiving the highest doses were male, current smokers, and had a history of depression and substance abuse.6 Similarly, a study by Bohnert and colleagues found that opioid medication overdose was most likely to occur in those patients with psychiatric and substance use disorders compared with patients who had no psychiatric illness history.8
Depression
Mood disorders are common in people with chronic pain.4,5,9,10 In particular, patients with a history of depression are more likely to receive chronic opioid medication prescriptions and are at a higher risk for opioid medication abuse. A substance abuse history is the most consistent predictor of both chronic opioid medication use and abuse. However, depression without substance abuse is significantly associated with 2 forms of opioid medication abuse: self-medication for stress or sleep and overmedication (using a higher dose than prescribed). More severe cases of depression show a stronger association for potential abuse.4
PTSD
Among Iraq and Afghanistan war veterans with ≥ 1 pain-related diagnosis, veterans with PTSD and veterans with a mental health disorder other than PTSD were significantly more likely to receive opioid medications for pain than were veterans without a mental health disorder (PTSD—17.8%, adjusted relative risk [RR] 2.58; other mental health disorder—11.7%, RR 1.74; no mental health disorder—6.5%).2 Although mental health disorders in general were related to a higher risk of opioid abuse, those with PTSD in particular were more likely to receive higher prescribed dosages; to continue taking opioids for a longer period; to receive concurrent prescriptions for opioid medications, sedative hypnotics, or both; to obtain early refills; and to have comorbid alcohol and substance use disorders. Based on these results, Seal and colleagues concluded that veterans with PTSD had the highest risk of alcohol, drug, and opioid-related accidents and overdose as well as self-inflicted injuries.2
Concurrent Use of Opioids and CNS Depressants
As mentioned earlier, studies suggest that people with PTSD are at a significantly higher risk for opioid medication overdose. One factor that may contribute to this higher risk is the concurrent use of CNS depressants/sedatives, particularly benzodiazepines and alcohol.
Benzodiazepines are often prescribed for people with PTSD. One study found that the concurrent use of benzodiazepines is significantly related to opioid overdose.5 Prescribing opioids for people already abusing or dependent on alcohol or other substances increases the risk of abuse and overdose. Furthermore, the concurrent use of multiple medications is associated with aberrant behaviors, cognitive impairment, and medication abuse, potentially leading to overdose. Overall, the combined administration of these medications is responsible for higher rates of adverse events, overdose, and death related to prescription opioid medication use.5,6,11
In summary, there are various risk factors that contribute to opioid medication overdose and more generally, risk of suicide, including (1) high-dose opioid medications; (2) history of psychiatric disorders, specifically depression and PTSD; (3) history of substance use disorders; and (4) concurrent use of opioid medications and prescription sedatives (specifically benzodiazepines) as well as alcohol and nonprescription drugs of abuse.
Suicide
Suicide is the tenth leading cause of death in the U.S., with 12.4 suicide deaths per 100,000 population.12 Suicide rates are even higher among veterans. According to the VHA, the age-adjusted rate of suicide for veterans using VHA facilities and clinics was 35.9 per 100,000 person-years for fiscal year 2009.13 Several risk factors for suicide attempts include depression and other mental health disorders, substance abuse, medical disorders, and prescription medications.
Prior suicide attempts or self-harm behavior is known to increase the risk of subsequent death by suicide. About 11 attempted suicides occur per suicide death where the medical severity of prior attempts increases the risk of future suicide, as does a history of multiple self-harm episodes.14,15 One study found that the single best predictor of suicide in a veteran population was an attempt in the previous 4 months.16
Among other risk factors, previous suicide attempts and violent behavior are major behavioral flags that warrant caution and require particular consideration when prescribing opioid medications. In a national survey on drug use and health, about 18% of prescription opioid users/abusers who experienced suicidal ideations actually attempted suicide. Only 11% of individuals who never used prescription opioid medications attempted suicide after reported suicidal ideations.17
Patient Data Aggregation
The early and widespread adoption of electronic medical records (EMRs) by the VHA allowed the aggregation of patient data for quality improvement. Initially, data were aggregated, and dashboards were designed retrospectively. However, the development of regional data warehouses that update patient information daily from the EMR allowed information to be aggregated prospectively, and dashboards were designed that provided real-time information.
The purpose of the current study is to demonstrate the efficacy and future potential of dashboard technology in assessing prospectively high-risk factors for opioid overdose. Dashboards are a user-friendly application that allows providers to isolate and calculate daily morphine equivalent opioid dosages and assess patients’ risk factors for overdose on an individual basis. By using this technology, providers who prescribe opioids can get a concise summary of opioid and other medications and adjust medications to decrease overdose risk on an individual basis.
What is the Dashboard?
The VISN 22 high-risk opioid dashboard is a business intelligence tool that serves as a report card, or progress report, to provide a global view of the number of veterans who are receiving opioid prescriptions totaling >120 mg MEDD and who have characteristics (history of depression, PTSD, substance abuse, or high-risk suicide flag) and prescriptions (concomitant CNS depressants) that may increase patient risk for overdose.
The VISN 22 dashboard allows the user to navigate to an individual HCP-level and patient-level report (Figures 1 and 2). Filter settings allow report users to select only high-risk patients; it serves as a single location for pertinent details to consider for safely prescribing opioids.
To calculate daily morphine equivalents, each patient’s opioid prescriptions were evaluated. The quantity was divided by the day’s supply to calculate an average daily quantity. From there, the drug strength was used to convert to MEDD. Health care providers were informed that these conversion factors were not recommendations for clinical opioid conversions.
Implementation and Design
In 2012, the VA Pharmacy Benefits Management (PBM) in VISN 21 created a dashboard that allowed users to identify patients on high-dose opioid prescriptions. Structured query language code was used to extract data from the regional data warehouse and calculate MEDD for all patients with active opioid prescriptions. In 2013, VISN 22 expanded that dashboard to incorporate factors that could indicate a high risk for overdose or other adverse outcomes, including a history of depression, PTSD, substance abuse or high-risk suicide flag, and concomitant use of CNS depressant medications.
The high-risk opioid dashboard (Figure 3) and accompanying patient-level report were first introduced to VISN 22 HCPs in January 2013. The business intelligence tools were introduced to each facility through the VISN 22 PBM group. Training on the use of the dashboard and the report was provided, with an initial target of decreasing MEDD of > 200 mg to < 5% of all veterans prescribed opioids at each VISN 22 facility. One month later (in February 2013), a second category of veterans (those with > 120 mg but < 199 mg MEDD) was added. Also the initial MEDD > 200 mg target of < 5% was decreased to < 3% to encourage additional progress.
Eight months after the VISN 22 dashboard technology was implemented there was a 17% decrease in patients with total daily morphine equivalents > 200 mg (January 2013; 1,137 patients vs August 2013; 940 patients—a decrease of 197 patients).
From March 2013 to August 2013, VISN 22 also saw a 12% decrease in the number of patients prescribed > 120 mg MEDD but < 199 MEDD (March 2013; 2,295 vs August 2013; 2,018—a decrease of 277 patients).
Figure 4 shows opioid use as of July 2014 for VISN 22 facilities. There were further reductions in the number of patients receiving > 120 mg but < 199 mg MEDD (August 2013; 2,018 patients vs July 2014; 1,189 patients) and patients receiving > 200 mg MEDD (August 2013; 940 patients vs July 2014; 836 patients).
Case Description
In January 2013, VISN 22 implemented dashboard technology to help providers assess and monitor opioid prescription levels in relation to high-risk variables. The benefits of this dashboard technology are illustrated in the case profile that follows.
A 67-year-old male veteran had a long history of chronic pain. Pain diagnoses included osteoarthritis with spine involvement, lumbar radiculopathy, arthralgia, and peripheral neuropathy. For the past 10 years, he was prescribed opioids with modest relief of his chronic pain symptoms despite recent prescriptions totaling 300 mg MEDD. This veteran had several risk factors for overdose, including a history of depression, suicide risk, PTSD, and concomitant use of the CNS depressants alprazolam and cyclobenzaprine.
More recently, in May 2013, the veteran exhibited aberrant behavior and requested early refills for alprazolam. In response, the pharmacist discussed the case with the HCP who prescribed the opioids, noting the concomitant overdose risk factors. As a result of this interaction, the veteran was referred for mental health services, and his prescriptions for opioids were gradually decreased. He is currently stable, now receiving 120 mg MEDD, and his pain is currently described as moderately controlled on the new lower dose.
In summary, this veteran was receiving > 200 mg MEDD with several known overdose risk factors. Once the HCP was made aware of these risk factors, necessary precautions were taken, and the veteran was safely tapered to a lower dose. Dashboard technology makes the list of risk factors readily available to HCPs who are prescribing (and the pharmacists reviewing the prescriptions), thus allowing a proactive discussion of risks and benefits before continuing, renewing, or initiating opioid prescriptions.
Discussion
As reported in 2013 by NIDA, the greater availability of opioid medications and the consequent increase in prescriptions may be contributing directly to their growing misuse by both civilians and military service personnel. A direct consequence has been an increase in both accidental and intentional overdose deaths.3 Several factors are related to the risk of overdose/death, including high-dose opioid medications, a history of psychiatric disorders (specifically depression and PTSD), a history of substance use disorders, concurrent use of opioid medications and prescription sedatives (specifically benzodiazepines) as well as alcohol and nonprescription drugs of abuse, and previous attempts of suicide.
The VISN 22 high-risk opioid dashboard was a rapid information technology response to the increasing risk faced by veterans who have chronic pain and comorbid psychiatric and substance use disorders and are prescribed opioids and CNS depressants. The purpose of implementing this dashboard technology was to assist HCPs in prescribing opioids safely, using a technology that allows for the monitoring and management of concomitant suicide risk factors. Following the national Opioid Safety Initiative, this dashboard tool is being used to identify veterans who are on high-dose opioids with the goal of reducing the number of veterans on > 200 mg MEDD. The dashboard allows data to be stratified, using the concomitant risk factors for suicide to assist facilities and their providers in the identification and prioritization of highest risk patients first.
An initial review of dashboard data in VISN 22 suggests that it is a useful tool for reducing high-dose opioid prescriptions (> 200 mg MEDD and > 120 mg but < 199 mg MEDD). Across the 5 VA locations in VISN 22, in the first 8 months of implementation, 4 locations were able to lower prescription opioid medication levels to the initial target of < 5%; 2 lowered rates even further (to < 3%). The VA Greater Los Angeles Healthcare System remains at a commendable 1%. Although the number of veterans with prescriptions totaling > 200 mg MEDD has decreased as a result, a greater reduction is expected with the coordinated education and systems improvement efforts associated with the national VHA Opioid Safety Initiative. As part of the process to lower the number of patients on high-dose opioids in the future, HCP and patient education will be provided in relation to the use of dashboard technology.
Limitations
There are several limitations that affect interpretation of the usefulness of the VISN 22 high-risk opioid dashboard. Prior to the implementation of the dashboard, 2 of 5 VISN sites already had efforts in place to reduce opioid overprescribing. The VA Greater Los Angeles Healthcare System had an opioid reduction program in place before the dashboard was implemented, so it is possible reductions in opioid prescribing were a result of their previous efforts and not related to the dashboard. Similarly the VA Long Beach Healthcare System had begun a quality improvement initiative to reduce high-dose opioid prescribing prior to dashboard implementation. However, it was difficult to pinpoint the direct effect the dashboard had on patient interventions due to lack of documentation of dashboard use in the clinical notes.
A direct relationship did exist between dashboard implementation and opioid dose reduction in patients with > 200 MEDD at the remaining 3 VISN 22 facilities. Overall, this suggests that the dashboard played a significant role across all sites. Implementation of the dashboard across VISN 22 was accompanied by an education effort that resulted in an increased awareness among HCPs to evaluate certain risks in patients on high-dose opioids and to evaluate the combination of opioid and CNS depressant use. Prior to dashboard implementation, there was no standardized monitoring system that cross-referenced high-dose opioid prescribing with psychiatric illness and suicide risk factors.
Conclusions
From 2000 to 2010, opioid prescriptions nearly doubled, yet this rate was not accompanied by a change/increase in the rate of nonopioid analgesic medication prescriptions.18 Health care providers need to account for veterans’ wishes for pain treatment and be aware of options other than opioids, particularly given the risk of opioid-related accidental or intentional overdose; it is imperative that treatment become more individualized and more closely monitored.19,20 It is recommended that opioids should be the treatment of last resort in managing chronic noncancer pain. The use of opioid prescription medications should be intended as a trial, supported by clear goals and an unequivocal understanding that doses will not be indiscriminately increased.20
Health care providers who prescribe opioids are ultimately responsible for monitoring risk factors that may increase overdose and death, and dashboard technology assists them in this effort. The VISN 22 high-risk opioid dashboard is a tool that allows providers to identify and prioritize veterans who are at high risk for overdose. Initial data collected suggest that the dashboard has decreased the risk of negative consequences associated with opioid medication use today. However, the authors wish to emphasize that this technology is only part of the solution; although it can be a tool to identify actions that may need to take place and can track progress of changes in care, there must be complementary efforts in provider and patient education, improved access to mental health care, and interdisciplinary models of care that expand current chronic pain treatment options. Future considerations of this technology may include incorporating other risk factors accounting for psychosocial variables specific to military personnel that may further increase the overall risk of overdose.
Acknowledgements
The authors wish to thank the leadership of VISN 22 for their support of this initiative. Dr. Kryskalla recognizes VA OI&T for making this work possible and her family for their support. Ms. Kern would like to thank Aaron, Leslie, and Rachel Kern for their continuous support. Dr. Hauser wishes to thank Cathy, Anika, Katia, Max, and Jirina Hauser for their unwavering support.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
[Published Online Ahead of Print: August 14, 2014.]
1. Brooks D. Hearing Spotlights painkiller overuse among soldiers. http://www.fayobserver.com/military/article_a6e4a2e9-827d-577c-a79a-87a6c07cf151.html. Fayobserver Website. Published October 10, 2013, Accessed June 9, 2014.
2. Seal KH, Shi Y, Cohen BE, Maguen S, Krebs EE, Neylan TC. Association of mental health disorders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012;307(9):940-947.
3. National Institute on Drug Abuse. DrugFacts: Substance Abuse in the Military. http://www.drugabuse.gov/publications/drugfacts/substance-abuse-in-military. National Institute on Drug Abuse Website. Revised March 2013. Accessed June 9, 2014.
4. Grattan A, Sullivan MD, Saunders KW, Campbell CI, Von Korff MR. Depression and prescription opioid misuse among chronic opioid therapy recipients with no history of substance abuse. Ann Fam Med. 2012;10(4):304-311.
5. Pergolizzi JV Jr, Gharibo C, Passik S, et al. Dynamic risk factors in the misuse of opioid analgesics. J Psychosom Res. 2012;72(6):443-451.
6. Dunn KM, Saunders KW, Rutter CM, et al. Opioid prescriptions for chronic pain and overdose: A cohort study. Ann Intern Med. 2010;152(2):85-92.
7. Substance Abuse and Mental Health Services Administration. SAMHSA Opioid Overdose Prevention Toolkit. HHS publication No. (SMA) 13-4742. Rockville, MD: Substance Abuse and Mental Health Service Administration; 2013.
8. Bohnert AS, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011;305(13):1315-1321.
9. Campbell LC, Clauw DJ, Keefe FJ. Persistent pain and depression: A biopsychosocial perspective. Biol Psychiatry. 2003;54(3):399-409.
10. Gatchel RJ, Peng YB, Peters ML, Fuchs PN, Turk DC. The biopsychosocial approach to chronic pain: Scientific advances and future directions. Psychol Bull. 2007;133(4):581-624.
11. Gudin JA, Mogali S, Jones JD, Comer SD. Risks, management, and monitoring of combination opioid, benzodiazepine, and/or alcohol use. Postgrad Med. 2013;125(4):115-130.
12. Centers for Disease Control and Prevention. FastStats: Deaths and mortality. Centers for Disease Control and Prevention Website. http://www.cdc.gov/nchs/fastats/deaths.htm. Updated February 13, 2014. Accessed June 9, 2014.
13. Kemp J, Bossarte R. Suicide Data Report, 2012. U.S. Department of Veterans Affairs Website. http://www.va.gov/opa/docs/Suicide-Data-Report-2012-final.pdf. Accessed July 1, 2014.
14. National Institute of Mental Health. Suicide in the U.S. Statistics. National Institute of Mental Health Website. http://www.nimh.nih.gov/statistics/index.shtml. Accessed June 27, 2014.
15. Miller M, Hempstead K, Nguyen T, Barber C, Rosenberg-Wohl S, Azrael D. Method choice in nonfatal self-harm as a predictor of subsequent episodes of self-harm and suicide: Implications for clinical practice. Am J Public Health. 2013;103(6):e61-e68.
16. Hartl TL, Rosen C, Drescher K, Lee TT, Gusman F. Predicting high-risk behaviors in Veterans with posttraumatic stress disorder. J Nerv Ment Dis. 2005;193(7):464-472.
17. Substance Abuse and Mental Health Services Administration. Results from the 2010 National Survey on Drug Use and Health: Summary of National Findings. NSDUH Series H-41, HHS Publication No. (SMA) 11-4658. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2011.
18. Daubresse M, Chang HY, Yu Y, et al. Ambulatory diagnosis and treatment of nonmalignant pain in the United States, 2000-2010. Med Care. 2013;51(10):870-878.
19. Bray RM, Pemberton MR, Lane ME, Hourani LL, Mattiko MJ, Babeu LA. Substance use and mental health trends among U.S. military active duty personnel. Key findings from the 2008 DoD Health Behavior Survey. Mil Med. 2010;175(6):390-399.
20. Cuevas-Trisan RL. The unfortunate turn of events in pain management. Fed Pract. 2013;30(3):8-9.
On October 10, 2013, a Congressional hearing was held to address the issue of opioid medication prescribing within VHA facilities and clinics (House Veteran Affairs Subcommittee hearing “Between Peril and Promise: Facing the Dangers of VA’s Skyrocketing Use of Prescription Painkillers to Treat Veterans”). Several individuals testified, including the widows of 2 veterans; both their husbands had overdosed on prescribed opioid medications. One husband had been taking as many as 15 pills a day and was additionally prescribed oxycodone/acetaminophen, which led to his death.1
Alongside the widows were 2 veterans who had been treated for chronic back pain injuries sustained before and during deployment in Iraq. Both had been prescribed several pain medications, including oxycodone/acetaminophen, methadone, and morphine. One reported that as his pain increased, his doctors continued to provide him additional prescriptions; at one point he had more than 13 prescriptions and could no longer work from being so “doped up.”1
In the past 2 decades, health care professionals (HCPs) have placed greater emphasis on chronic pain management. As a result, the rate of opioid medication prescribing has increased dramatically. Since 1994, the number of opioid medication prescriptions has nearly doubled; this change has been accompanied by an increase in opioid misuse, which has resulted in accidental or intentional overdose and death.2
Based on a recent National Institute on Drug Abuse (NIDA) report, the greatest impact has been on armed forces personnel.3 Prescriptions for pain relievers quadrupled between 2001 and 2009 to almost 3.8 million within the military population. Although civilian populations are more likely to abuse illicit drugs, military personnel are at particular risk of prescription abuse, including opioid medications.3 In 2008, 11% of armed forces service members reported misusing prescription drugs, with opioid medications being the most abused. This is an approximate 5- to 6-fold increase since 2002 (2% reported misuse in 2002).3 Particularly concerning is the associated rise in suicide rates among armed forces personnel, which surpassed civilian suicide rates in 2004. In 2009, one-third of suicides among armed forces personnel involved prescription drugs.3
Certain patient characteristics or factors are related to greater overdose risk. These risk factors include prescription dosage and frequency, history of suicide attempts or self-harm behavior, history of depression or posttraumatic stress disorder (PTSD) among other mental health-related diagnoses, a history of substance and/or alcohol abuse, and within the context of opioid medication use, the concurrent use of other central nervous system (CNS) depressants.4,5 Additionally, the stresses of deployment during wartime, physical injuries sustained in combat, and the unique military culture play a particularly important role in access to substances with high abuse potential and the subsequent development of substance abuse.3
Opioid Use and Risk Factors
More than 3% of adults in the U.S. are now receiving opioid medications for chronic noncancer pain.6 Substance abuse among patients with chronic pain ranges from 14% to 40%.5 Prescription opioid medications are the fastest growing drugs of abuse and the most common cause of unintentional overdose in the U.S.4 About 17,000 deaths occur each year as a result of prescription opioid medication overdose.7 Opioid medication-related overdose deaths began to increase in the early 2000s and continue to increase. Between 1999 and 2007, the rate of unintentional overdose-related deaths in the U.S. increased by 124%, largely due to the increase of prescription opioid medications.8
High-Dose Opioid Medication Use
A study by Dunn and colleagues found that patients receiving higher doses of prescribed opioid medications were at an increased risk of overdose.6 Patients receiving 50 mg to 99 mg morphine equivalent daily dose (MEDD) had a 3.7-fold increase in overdose risk (0.7% annual overdose rate) as compared with patients who received < 50 mg MEDD (0.2% annual overdose rate). Patients receiving ≥ 100 mg MEDD had a 1.8% annual overdose rate and a 9.8-fold increase in overdose risk as compared with patients who received < 50 mg MEDD. Overall, 51 patients experienced ≥ 1 overdose event, 40 of whom experienced fatal or serious overdoses and 6 of whom attempted suicide. Patients receiving the highest doses were male, current smokers, and had a history of depression and substance abuse.6 Similarly, a study by Bohnert and colleagues found that opioid medication overdose was most likely to occur in those patients with psychiatric and substance use disorders compared with patients who had no psychiatric illness history.8
Depression
Mood disorders are common in people with chronic pain.4,5,9,10 In particular, patients with a history of depression are more likely to receive chronic opioid medication prescriptions and are at a higher risk for opioid medication abuse. A substance abuse history is the most consistent predictor of both chronic opioid medication use and abuse. However, depression without substance abuse is significantly associated with 2 forms of opioid medication abuse: self-medication for stress or sleep and overmedication (using a higher dose than prescribed). More severe cases of depression show a stronger association for potential abuse.4
PTSD
Among Iraq and Afghanistan war veterans with ≥ 1 pain-related diagnosis, veterans with PTSD and veterans with a mental health disorder other than PTSD were significantly more likely to receive opioid medications for pain than were veterans without a mental health disorder (PTSD—17.8%, adjusted relative risk [RR] 2.58; other mental health disorder—11.7%, RR 1.74; no mental health disorder—6.5%).2 Although mental health disorders in general were related to a higher risk of opioid abuse, those with PTSD in particular were more likely to receive higher prescribed dosages; to continue taking opioids for a longer period; to receive concurrent prescriptions for opioid medications, sedative hypnotics, or both; to obtain early refills; and to have comorbid alcohol and substance use disorders. Based on these results, Seal and colleagues concluded that veterans with PTSD had the highest risk of alcohol, drug, and opioid-related accidents and overdose as well as self-inflicted injuries.2
Concurrent Use of Opioids and CNS Depressants
As mentioned earlier, studies suggest that people with PTSD are at a significantly higher risk for opioid medication overdose. One factor that may contribute to this higher risk is the concurrent use of CNS depressants/sedatives, particularly benzodiazepines and alcohol.
Benzodiazepines are often prescribed for people with PTSD. One study found that the concurrent use of benzodiazepines is significantly related to opioid overdose.5 Prescribing opioids for people already abusing or dependent on alcohol or other substances increases the risk of abuse and overdose. Furthermore, the concurrent use of multiple medications is associated with aberrant behaviors, cognitive impairment, and medication abuse, potentially leading to overdose. Overall, the combined administration of these medications is responsible for higher rates of adverse events, overdose, and death related to prescription opioid medication use.5,6,11
In summary, there are various risk factors that contribute to opioid medication overdose and more generally, risk of suicide, including (1) high-dose opioid medications; (2) history of psychiatric disorders, specifically depression and PTSD; (3) history of substance use disorders; and (4) concurrent use of opioid medications and prescription sedatives (specifically benzodiazepines) as well as alcohol and nonprescription drugs of abuse.
Suicide
Suicide is the tenth leading cause of death in the U.S., with 12.4 suicide deaths per 100,000 population.12 Suicide rates are even higher among veterans. According to the VHA, the age-adjusted rate of suicide for veterans using VHA facilities and clinics was 35.9 per 100,000 person-years for fiscal year 2009.13 Several risk factors for suicide attempts include depression and other mental health disorders, substance abuse, medical disorders, and prescription medications.
Prior suicide attempts or self-harm behavior is known to increase the risk of subsequent death by suicide. About 11 attempted suicides occur per suicide death where the medical severity of prior attempts increases the risk of future suicide, as does a history of multiple self-harm episodes.14,15 One study found that the single best predictor of suicide in a veteran population was an attempt in the previous 4 months.16
Among other risk factors, previous suicide attempts and violent behavior are major behavioral flags that warrant caution and require particular consideration when prescribing opioid medications. In a national survey on drug use and health, about 18% of prescription opioid users/abusers who experienced suicidal ideations actually attempted suicide. Only 11% of individuals who never used prescription opioid medications attempted suicide after reported suicidal ideations.17
Patient Data Aggregation
The early and widespread adoption of electronic medical records (EMRs) by the VHA allowed the aggregation of patient data for quality improvement. Initially, data were aggregated, and dashboards were designed retrospectively. However, the development of regional data warehouses that update patient information daily from the EMR allowed information to be aggregated prospectively, and dashboards were designed that provided real-time information.
The purpose of the current study is to demonstrate the efficacy and future potential of dashboard technology in assessing prospectively high-risk factors for opioid overdose. Dashboards are a user-friendly application that allows providers to isolate and calculate daily morphine equivalent opioid dosages and assess patients’ risk factors for overdose on an individual basis. By using this technology, providers who prescribe opioids can get a concise summary of opioid and other medications and adjust medications to decrease overdose risk on an individual basis.
What is the Dashboard?
The VISN 22 high-risk opioid dashboard is a business intelligence tool that serves as a report card, or progress report, to provide a global view of the number of veterans who are receiving opioid prescriptions totaling >120 mg MEDD and who have characteristics (history of depression, PTSD, substance abuse, or high-risk suicide flag) and prescriptions (concomitant CNS depressants) that may increase patient risk for overdose.
The VISN 22 dashboard allows the user to navigate to an individual HCP-level and patient-level report (Figures 1 and 2). Filter settings allow report users to select only high-risk patients; it serves as a single location for pertinent details to consider for safely prescribing opioids.
To calculate daily morphine equivalents, each patient’s opioid prescriptions were evaluated. The quantity was divided by the day’s supply to calculate an average daily quantity. From there, the drug strength was used to convert to MEDD. Health care providers were informed that these conversion factors were not recommendations for clinical opioid conversions.
Implementation and Design
In 2012, the VA Pharmacy Benefits Management (PBM) in VISN 21 created a dashboard that allowed users to identify patients on high-dose opioid prescriptions. Structured query language code was used to extract data from the regional data warehouse and calculate MEDD for all patients with active opioid prescriptions. In 2013, VISN 22 expanded that dashboard to incorporate factors that could indicate a high risk for overdose or other adverse outcomes, including a history of depression, PTSD, substance abuse or high-risk suicide flag, and concomitant use of CNS depressant medications.
The high-risk opioid dashboard (Figure 3) and accompanying patient-level report were first introduced to VISN 22 HCPs in January 2013. The business intelligence tools were introduced to each facility through the VISN 22 PBM group. Training on the use of the dashboard and the report was provided, with an initial target of decreasing MEDD of > 200 mg to < 5% of all veterans prescribed opioids at each VISN 22 facility. One month later (in February 2013), a second category of veterans (those with > 120 mg but < 199 mg MEDD) was added. Also the initial MEDD > 200 mg target of < 5% was decreased to < 3% to encourage additional progress.
Eight months after the VISN 22 dashboard technology was implemented there was a 17% decrease in patients with total daily morphine equivalents > 200 mg (January 2013; 1,137 patients vs August 2013; 940 patients—a decrease of 197 patients).
From March 2013 to August 2013, VISN 22 also saw a 12% decrease in the number of patients prescribed > 120 mg MEDD but < 199 MEDD (March 2013; 2,295 vs August 2013; 2,018—a decrease of 277 patients).
Figure 4 shows opioid use as of July 2014 for VISN 22 facilities. There were further reductions in the number of patients receiving > 120 mg but < 199 mg MEDD (August 2013; 2,018 patients vs July 2014; 1,189 patients) and patients receiving > 200 mg MEDD (August 2013; 940 patients vs July 2014; 836 patients).
Case Description
In January 2013, VISN 22 implemented dashboard technology to help providers assess and monitor opioid prescription levels in relation to high-risk variables. The benefits of this dashboard technology are illustrated in the case profile that follows.
A 67-year-old male veteran had a long history of chronic pain. Pain diagnoses included osteoarthritis with spine involvement, lumbar radiculopathy, arthralgia, and peripheral neuropathy. For the past 10 years, he was prescribed opioids with modest relief of his chronic pain symptoms despite recent prescriptions totaling 300 mg MEDD. This veteran had several risk factors for overdose, including a history of depression, suicide risk, PTSD, and concomitant use of the CNS depressants alprazolam and cyclobenzaprine.
More recently, in May 2013, the veteran exhibited aberrant behavior and requested early refills for alprazolam. In response, the pharmacist discussed the case with the HCP who prescribed the opioids, noting the concomitant overdose risk factors. As a result of this interaction, the veteran was referred for mental health services, and his prescriptions for opioids were gradually decreased. He is currently stable, now receiving 120 mg MEDD, and his pain is currently described as moderately controlled on the new lower dose.
In summary, this veteran was receiving > 200 mg MEDD with several known overdose risk factors. Once the HCP was made aware of these risk factors, necessary precautions were taken, and the veteran was safely tapered to a lower dose. Dashboard technology makes the list of risk factors readily available to HCPs who are prescribing (and the pharmacists reviewing the prescriptions), thus allowing a proactive discussion of risks and benefits before continuing, renewing, or initiating opioid prescriptions.
Discussion
As reported in 2013 by NIDA, the greater availability of opioid medications and the consequent increase in prescriptions may be contributing directly to their growing misuse by both civilians and military service personnel. A direct consequence has been an increase in both accidental and intentional overdose deaths.3 Several factors are related to the risk of overdose/death, including high-dose opioid medications, a history of psychiatric disorders (specifically depression and PTSD), a history of substance use disorders, concurrent use of opioid medications and prescription sedatives (specifically benzodiazepines) as well as alcohol and nonprescription drugs of abuse, and previous attempts of suicide.
The VISN 22 high-risk opioid dashboard was a rapid information technology response to the increasing risk faced by veterans who have chronic pain and comorbid psychiatric and substance use disorders and are prescribed opioids and CNS depressants. The purpose of implementing this dashboard technology was to assist HCPs in prescribing opioids safely, using a technology that allows for the monitoring and management of concomitant suicide risk factors. Following the national Opioid Safety Initiative, this dashboard tool is being used to identify veterans who are on high-dose opioids with the goal of reducing the number of veterans on > 200 mg MEDD. The dashboard allows data to be stratified, using the concomitant risk factors for suicide to assist facilities and their providers in the identification and prioritization of highest risk patients first.
An initial review of dashboard data in VISN 22 suggests that it is a useful tool for reducing high-dose opioid prescriptions (> 200 mg MEDD and > 120 mg but < 199 mg MEDD). Across the 5 VA locations in VISN 22, in the first 8 months of implementation, 4 locations were able to lower prescription opioid medication levels to the initial target of < 5%; 2 lowered rates even further (to < 3%). The VA Greater Los Angeles Healthcare System remains at a commendable 1%. Although the number of veterans with prescriptions totaling > 200 mg MEDD has decreased as a result, a greater reduction is expected with the coordinated education and systems improvement efforts associated with the national VHA Opioid Safety Initiative. As part of the process to lower the number of patients on high-dose opioids in the future, HCP and patient education will be provided in relation to the use of dashboard technology.
Limitations
There are several limitations that affect interpretation of the usefulness of the VISN 22 high-risk opioid dashboard. Prior to the implementation of the dashboard, 2 of 5 VISN sites already had efforts in place to reduce opioid overprescribing. The VA Greater Los Angeles Healthcare System had an opioid reduction program in place before the dashboard was implemented, so it is possible reductions in opioid prescribing were a result of their previous efforts and not related to the dashboard. Similarly the VA Long Beach Healthcare System had begun a quality improvement initiative to reduce high-dose opioid prescribing prior to dashboard implementation. However, it was difficult to pinpoint the direct effect the dashboard had on patient interventions due to lack of documentation of dashboard use in the clinical notes.
A direct relationship did exist between dashboard implementation and opioid dose reduction in patients with > 200 MEDD at the remaining 3 VISN 22 facilities. Overall, this suggests that the dashboard played a significant role across all sites. Implementation of the dashboard across VISN 22 was accompanied by an education effort that resulted in an increased awareness among HCPs to evaluate certain risks in patients on high-dose opioids and to evaluate the combination of opioid and CNS depressant use. Prior to dashboard implementation, there was no standardized monitoring system that cross-referenced high-dose opioid prescribing with psychiatric illness and suicide risk factors.
Conclusions
From 2000 to 2010, opioid prescriptions nearly doubled, yet this rate was not accompanied by a change/increase in the rate of nonopioid analgesic medication prescriptions.18 Health care providers need to account for veterans’ wishes for pain treatment and be aware of options other than opioids, particularly given the risk of opioid-related accidental or intentional overdose; it is imperative that treatment become more individualized and more closely monitored.19,20 It is recommended that opioids should be the treatment of last resort in managing chronic noncancer pain. The use of opioid prescription medications should be intended as a trial, supported by clear goals and an unequivocal understanding that doses will not be indiscriminately increased.20
Health care providers who prescribe opioids are ultimately responsible for monitoring risk factors that may increase overdose and death, and dashboard technology assists them in this effort. The VISN 22 high-risk opioid dashboard is a tool that allows providers to identify and prioritize veterans who are at high risk for overdose. Initial data collected suggest that the dashboard has decreased the risk of negative consequences associated with opioid medication use today. However, the authors wish to emphasize that this technology is only part of the solution; although it can be a tool to identify actions that may need to take place and can track progress of changes in care, there must be complementary efforts in provider and patient education, improved access to mental health care, and interdisciplinary models of care that expand current chronic pain treatment options. Future considerations of this technology may include incorporating other risk factors accounting for psychosocial variables specific to military personnel that may further increase the overall risk of overdose.
Acknowledgements
The authors wish to thank the leadership of VISN 22 for their support of this initiative. Dr. Kryskalla recognizes VA OI&T for making this work possible and her family for their support. Ms. Kern would like to thank Aaron, Leslie, and Rachel Kern for their continuous support. Dr. Hauser wishes to thank Cathy, Anika, Katia, Max, and Jirina Hauser for their unwavering support.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
[Published Online Ahead of Print: August 14, 2014.]
On October 10, 2013, a Congressional hearing was held to address the issue of opioid medication prescribing within VHA facilities and clinics (House Veteran Affairs Subcommittee hearing “Between Peril and Promise: Facing the Dangers of VA’s Skyrocketing Use of Prescription Painkillers to Treat Veterans”). Several individuals testified, including the widows of 2 veterans; both their husbands had overdosed on prescribed opioid medications. One husband had been taking as many as 15 pills a day and was additionally prescribed oxycodone/acetaminophen, which led to his death.1
Alongside the widows were 2 veterans who had been treated for chronic back pain injuries sustained before and during deployment in Iraq. Both had been prescribed several pain medications, including oxycodone/acetaminophen, methadone, and morphine. One reported that as his pain increased, his doctors continued to provide him additional prescriptions; at one point he had more than 13 prescriptions and could no longer work from being so “doped up.”1
In the past 2 decades, health care professionals (HCPs) have placed greater emphasis on chronic pain management. As a result, the rate of opioid medication prescribing has increased dramatically. Since 1994, the number of opioid medication prescriptions has nearly doubled; this change has been accompanied by an increase in opioid misuse, which has resulted in accidental or intentional overdose and death.2
Based on a recent National Institute on Drug Abuse (NIDA) report, the greatest impact has been on armed forces personnel.3 Prescriptions for pain relievers quadrupled between 2001 and 2009 to almost 3.8 million within the military population. Although civilian populations are more likely to abuse illicit drugs, military personnel are at particular risk of prescription abuse, including opioid medications.3 In 2008, 11% of armed forces service members reported misusing prescription drugs, with opioid medications being the most abused. This is an approximate 5- to 6-fold increase since 2002 (2% reported misuse in 2002).3 Particularly concerning is the associated rise in suicide rates among armed forces personnel, which surpassed civilian suicide rates in 2004. In 2009, one-third of suicides among armed forces personnel involved prescription drugs.3
Certain patient characteristics or factors are related to greater overdose risk. These risk factors include prescription dosage and frequency, history of suicide attempts or self-harm behavior, history of depression or posttraumatic stress disorder (PTSD) among other mental health-related diagnoses, a history of substance and/or alcohol abuse, and within the context of opioid medication use, the concurrent use of other central nervous system (CNS) depressants.4,5 Additionally, the stresses of deployment during wartime, physical injuries sustained in combat, and the unique military culture play a particularly important role in access to substances with high abuse potential and the subsequent development of substance abuse.3
Opioid Use and Risk Factors
More than 3% of adults in the U.S. are now receiving opioid medications for chronic noncancer pain.6 Substance abuse among patients with chronic pain ranges from 14% to 40%.5 Prescription opioid medications are the fastest growing drugs of abuse and the most common cause of unintentional overdose in the U.S.4 About 17,000 deaths occur each year as a result of prescription opioid medication overdose.7 Opioid medication-related overdose deaths began to increase in the early 2000s and continue to increase. Between 1999 and 2007, the rate of unintentional overdose-related deaths in the U.S. increased by 124%, largely due to the increase of prescription opioid medications.8
High-Dose Opioid Medication Use
A study by Dunn and colleagues found that patients receiving higher doses of prescribed opioid medications were at an increased risk of overdose.6 Patients receiving 50 mg to 99 mg morphine equivalent daily dose (MEDD) had a 3.7-fold increase in overdose risk (0.7% annual overdose rate) as compared with patients who received < 50 mg MEDD (0.2% annual overdose rate). Patients receiving ≥ 100 mg MEDD had a 1.8% annual overdose rate and a 9.8-fold increase in overdose risk as compared with patients who received < 50 mg MEDD. Overall, 51 patients experienced ≥ 1 overdose event, 40 of whom experienced fatal or serious overdoses and 6 of whom attempted suicide. Patients receiving the highest doses were male, current smokers, and had a history of depression and substance abuse.6 Similarly, a study by Bohnert and colleagues found that opioid medication overdose was most likely to occur in those patients with psychiatric and substance use disorders compared with patients who had no psychiatric illness history.8
Depression
Mood disorders are common in people with chronic pain.4,5,9,10 In particular, patients with a history of depression are more likely to receive chronic opioid medication prescriptions and are at a higher risk for opioid medication abuse. A substance abuse history is the most consistent predictor of both chronic opioid medication use and abuse. However, depression without substance abuse is significantly associated with 2 forms of opioid medication abuse: self-medication for stress or sleep and overmedication (using a higher dose than prescribed). More severe cases of depression show a stronger association for potential abuse.4
PTSD
Among Iraq and Afghanistan war veterans with ≥ 1 pain-related diagnosis, veterans with PTSD and veterans with a mental health disorder other than PTSD were significantly more likely to receive opioid medications for pain than were veterans without a mental health disorder (PTSD—17.8%, adjusted relative risk [RR] 2.58; other mental health disorder—11.7%, RR 1.74; no mental health disorder—6.5%).2 Although mental health disorders in general were related to a higher risk of opioid abuse, those with PTSD in particular were more likely to receive higher prescribed dosages; to continue taking opioids for a longer period; to receive concurrent prescriptions for opioid medications, sedative hypnotics, or both; to obtain early refills; and to have comorbid alcohol and substance use disorders. Based on these results, Seal and colleagues concluded that veterans with PTSD had the highest risk of alcohol, drug, and opioid-related accidents and overdose as well as self-inflicted injuries.2
Concurrent Use of Opioids and CNS Depressants
As mentioned earlier, studies suggest that people with PTSD are at a significantly higher risk for opioid medication overdose. One factor that may contribute to this higher risk is the concurrent use of CNS depressants/sedatives, particularly benzodiazepines and alcohol.
Benzodiazepines are often prescribed for people with PTSD. One study found that the concurrent use of benzodiazepines is significantly related to opioid overdose.5 Prescribing opioids for people already abusing or dependent on alcohol or other substances increases the risk of abuse and overdose. Furthermore, the concurrent use of multiple medications is associated with aberrant behaviors, cognitive impairment, and medication abuse, potentially leading to overdose. Overall, the combined administration of these medications is responsible for higher rates of adverse events, overdose, and death related to prescription opioid medication use.5,6,11
In summary, there are various risk factors that contribute to opioid medication overdose and more generally, risk of suicide, including (1) high-dose opioid medications; (2) history of psychiatric disorders, specifically depression and PTSD; (3) history of substance use disorders; and (4) concurrent use of opioid medications and prescription sedatives (specifically benzodiazepines) as well as alcohol and nonprescription drugs of abuse.
Suicide
Suicide is the tenth leading cause of death in the U.S., with 12.4 suicide deaths per 100,000 population.12 Suicide rates are even higher among veterans. According to the VHA, the age-adjusted rate of suicide for veterans using VHA facilities and clinics was 35.9 per 100,000 person-years for fiscal year 2009.13 Several risk factors for suicide attempts include depression and other mental health disorders, substance abuse, medical disorders, and prescription medications.
Prior suicide attempts or self-harm behavior is known to increase the risk of subsequent death by suicide. About 11 attempted suicides occur per suicide death where the medical severity of prior attempts increases the risk of future suicide, as does a history of multiple self-harm episodes.14,15 One study found that the single best predictor of suicide in a veteran population was an attempt in the previous 4 months.16
Among other risk factors, previous suicide attempts and violent behavior are major behavioral flags that warrant caution and require particular consideration when prescribing opioid medications. In a national survey on drug use and health, about 18% of prescription opioid users/abusers who experienced suicidal ideations actually attempted suicide. Only 11% of individuals who never used prescription opioid medications attempted suicide after reported suicidal ideations.17
Patient Data Aggregation
The early and widespread adoption of electronic medical records (EMRs) by the VHA allowed the aggregation of patient data for quality improvement. Initially, data were aggregated, and dashboards were designed retrospectively. However, the development of regional data warehouses that update patient information daily from the EMR allowed information to be aggregated prospectively, and dashboards were designed that provided real-time information.
The purpose of the current study is to demonstrate the efficacy and future potential of dashboard technology in assessing prospectively high-risk factors for opioid overdose. Dashboards are a user-friendly application that allows providers to isolate and calculate daily morphine equivalent opioid dosages and assess patients’ risk factors for overdose on an individual basis. By using this technology, providers who prescribe opioids can get a concise summary of opioid and other medications and adjust medications to decrease overdose risk on an individual basis.
What is the Dashboard?
The VISN 22 high-risk opioid dashboard is a business intelligence tool that serves as a report card, or progress report, to provide a global view of the number of veterans who are receiving opioid prescriptions totaling >120 mg MEDD and who have characteristics (history of depression, PTSD, substance abuse, or high-risk suicide flag) and prescriptions (concomitant CNS depressants) that may increase patient risk for overdose.
The VISN 22 dashboard allows the user to navigate to an individual HCP-level and patient-level report (Figures 1 and 2). Filter settings allow report users to select only high-risk patients; it serves as a single location for pertinent details to consider for safely prescribing opioids.
To calculate daily morphine equivalents, each patient’s opioid prescriptions were evaluated. The quantity was divided by the day’s supply to calculate an average daily quantity. From there, the drug strength was used to convert to MEDD. Health care providers were informed that these conversion factors were not recommendations for clinical opioid conversions.
Implementation and Design
In 2012, the VA Pharmacy Benefits Management (PBM) in VISN 21 created a dashboard that allowed users to identify patients on high-dose opioid prescriptions. Structured query language code was used to extract data from the regional data warehouse and calculate MEDD for all patients with active opioid prescriptions. In 2013, VISN 22 expanded that dashboard to incorporate factors that could indicate a high risk for overdose or other adverse outcomes, including a history of depression, PTSD, substance abuse or high-risk suicide flag, and concomitant use of CNS depressant medications.
The high-risk opioid dashboard (Figure 3) and accompanying patient-level report were first introduced to VISN 22 HCPs in January 2013. The business intelligence tools were introduced to each facility through the VISN 22 PBM group. Training on the use of the dashboard and the report was provided, with an initial target of decreasing MEDD of > 200 mg to < 5% of all veterans prescribed opioids at each VISN 22 facility. One month later (in February 2013), a second category of veterans (those with > 120 mg but < 199 mg MEDD) was added. Also the initial MEDD > 200 mg target of < 5% was decreased to < 3% to encourage additional progress.
Eight months after the VISN 22 dashboard technology was implemented there was a 17% decrease in patients with total daily morphine equivalents > 200 mg (January 2013; 1,137 patients vs August 2013; 940 patients—a decrease of 197 patients).
From March 2013 to August 2013, VISN 22 also saw a 12% decrease in the number of patients prescribed > 120 mg MEDD but < 199 MEDD (March 2013; 2,295 vs August 2013; 2,018—a decrease of 277 patients).
Figure 4 shows opioid use as of July 2014 for VISN 22 facilities. There were further reductions in the number of patients receiving > 120 mg but < 199 mg MEDD (August 2013; 2,018 patients vs July 2014; 1,189 patients) and patients receiving > 200 mg MEDD (August 2013; 940 patients vs July 2014; 836 patients).
Case Description
In January 2013, VISN 22 implemented dashboard technology to help providers assess and monitor opioid prescription levels in relation to high-risk variables. The benefits of this dashboard technology are illustrated in the case profile that follows.
A 67-year-old male veteran had a long history of chronic pain. Pain diagnoses included osteoarthritis with spine involvement, lumbar radiculopathy, arthralgia, and peripheral neuropathy. For the past 10 years, he was prescribed opioids with modest relief of his chronic pain symptoms despite recent prescriptions totaling 300 mg MEDD. This veteran had several risk factors for overdose, including a history of depression, suicide risk, PTSD, and concomitant use of the CNS depressants alprazolam and cyclobenzaprine.
More recently, in May 2013, the veteran exhibited aberrant behavior and requested early refills for alprazolam. In response, the pharmacist discussed the case with the HCP who prescribed the opioids, noting the concomitant overdose risk factors. As a result of this interaction, the veteran was referred for mental health services, and his prescriptions for opioids were gradually decreased. He is currently stable, now receiving 120 mg MEDD, and his pain is currently described as moderately controlled on the new lower dose.
In summary, this veteran was receiving > 200 mg MEDD with several known overdose risk factors. Once the HCP was made aware of these risk factors, necessary precautions were taken, and the veteran was safely tapered to a lower dose. Dashboard technology makes the list of risk factors readily available to HCPs who are prescribing (and the pharmacists reviewing the prescriptions), thus allowing a proactive discussion of risks and benefits before continuing, renewing, or initiating opioid prescriptions.
Discussion
As reported in 2013 by NIDA, the greater availability of opioid medications and the consequent increase in prescriptions may be contributing directly to their growing misuse by both civilians and military service personnel. A direct consequence has been an increase in both accidental and intentional overdose deaths.3 Several factors are related to the risk of overdose/death, including high-dose opioid medications, a history of psychiatric disorders (specifically depression and PTSD), a history of substance use disorders, concurrent use of opioid medications and prescription sedatives (specifically benzodiazepines) as well as alcohol and nonprescription drugs of abuse, and previous attempts of suicide.
The VISN 22 high-risk opioid dashboard was a rapid information technology response to the increasing risk faced by veterans who have chronic pain and comorbid psychiatric and substance use disorders and are prescribed opioids and CNS depressants. The purpose of implementing this dashboard technology was to assist HCPs in prescribing opioids safely, using a technology that allows for the monitoring and management of concomitant suicide risk factors. Following the national Opioid Safety Initiative, this dashboard tool is being used to identify veterans who are on high-dose opioids with the goal of reducing the number of veterans on > 200 mg MEDD. The dashboard allows data to be stratified, using the concomitant risk factors for suicide to assist facilities and their providers in the identification and prioritization of highest risk patients first.
An initial review of dashboard data in VISN 22 suggests that it is a useful tool for reducing high-dose opioid prescriptions (> 200 mg MEDD and > 120 mg but < 199 mg MEDD). Across the 5 VA locations in VISN 22, in the first 8 months of implementation, 4 locations were able to lower prescription opioid medication levels to the initial target of < 5%; 2 lowered rates even further (to < 3%). The VA Greater Los Angeles Healthcare System remains at a commendable 1%. Although the number of veterans with prescriptions totaling > 200 mg MEDD has decreased as a result, a greater reduction is expected with the coordinated education and systems improvement efforts associated with the national VHA Opioid Safety Initiative. As part of the process to lower the number of patients on high-dose opioids in the future, HCP and patient education will be provided in relation to the use of dashboard technology.
Limitations
There are several limitations that affect interpretation of the usefulness of the VISN 22 high-risk opioid dashboard. Prior to the implementation of the dashboard, 2 of 5 VISN sites already had efforts in place to reduce opioid overprescribing. The VA Greater Los Angeles Healthcare System had an opioid reduction program in place before the dashboard was implemented, so it is possible reductions in opioid prescribing were a result of their previous efforts and not related to the dashboard. Similarly the VA Long Beach Healthcare System had begun a quality improvement initiative to reduce high-dose opioid prescribing prior to dashboard implementation. However, it was difficult to pinpoint the direct effect the dashboard had on patient interventions due to lack of documentation of dashboard use in the clinical notes.
A direct relationship did exist between dashboard implementation and opioid dose reduction in patients with > 200 MEDD at the remaining 3 VISN 22 facilities. Overall, this suggests that the dashboard played a significant role across all sites. Implementation of the dashboard across VISN 22 was accompanied by an education effort that resulted in an increased awareness among HCPs to evaluate certain risks in patients on high-dose opioids and to evaluate the combination of opioid and CNS depressant use. Prior to dashboard implementation, there was no standardized monitoring system that cross-referenced high-dose opioid prescribing with psychiatric illness and suicide risk factors.
Conclusions
From 2000 to 2010, opioid prescriptions nearly doubled, yet this rate was not accompanied by a change/increase in the rate of nonopioid analgesic medication prescriptions.18 Health care providers need to account for veterans’ wishes for pain treatment and be aware of options other than opioids, particularly given the risk of opioid-related accidental or intentional overdose; it is imperative that treatment become more individualized and more closely monitored.19,20 It is recommended that opioids should be the treatment of last resort in managing chronic noncancer pain. The use of opioid prescription medications should be intended as a trial, supported by clear goals and an unequivocal understanding that doses will not be indiscriminately increased.20
Health care providers who prescribe opioids are ultimately responsible for monitoring risk factors that may increase overdose and death, and dashboard technology assists them in this effort. The VISN 22 high-risk opioid dashboard is a tool that allows providers to identify and prioritize veterans who are at high risk for overdose. Initial data collected suggest that the dashboard has decreased the risk of negative consequences associated with opioid medication use today. However, the authors wish to emphasize that this technology is only part of the solution; although it can be a tool to identify actions that may need to take place and can track progress of changes in care, there must be complementary efforts in provider and patient education, improved access to mental health care, and interdisciplinary models of care that expand current chronic pain treatment options. Future considerations of this technology may include incorporating other risk factors accounting for psychosocial variables specific to military personnel that may further increase the overall risk of overdose.
Acknowledgements
The authors wish to thank the leadership of VISN 22 for their support of this initiative. Dr. Kryskalla recognizes VA OI&T for making this work possible and her family for their support. Ms. Kern would like to thank Aaron, Leslie, and Rachel Kern for their continuous support. Dr. Hauser wishes to thank Cathy, Anika, Katia, Max, and Jirina Hauser for their unwavering support.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
[Published Online Ahead of Print: August 14, 2014.]
1. Brooks D. Hearing Spotlights painkiller overuse among soldiers. http://www.fayobserver.com/military/article_a6e4a2e9-827d-577c-a79a-87a6c07cf151.html. Fayobserver Website. Published October 10, 2013, Accessed June 9, 2014.
2. Seal KH, Shi Y, Cohen BE, Maguen S, Krebs EE, Neylan TC. Association of mental health disorders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012;307(9):940-947.
3. National Institute on Drug Abuse. DrugFacts: Substance Abuse in the Military. http://www.drugabuse.gov/publications/drugfacts/substance-abuse-in-military. National Institute on Drug Abuse Website. Revised March 2013. Accessed June 9, 2014.
4. Grattan A, Sullivan MD, Saunders KW, Campbell CI, Von Korff MR. Depression and prescription opioid misuse among chronic opioid therapy recipients with no history of substance abuse. Ann Fam Med. 2012;10(4):304-311.
5. Pergolizzi JV Jr, Gharibo C, Passik S, et al. Dynamic risk factors in the misuse of opioid analgesics. J Psychosom Res. 2012;72(6):443-451.
6. Dunn KM, Saunders KW, Rutter CM, et al. Opioid prescriptions for chronic pain and overdose: A cohort study. Ann Intern Med. 2010;152(2):85-92.
7. Substance Abuse and Mental Health Services Administration. SAMHSA Opioid Overdose Prevention Toolkit. HHS publication No. (SMA) 13-4742. Rockville, MD: Substance Abuse and Mental Health Service Administration; 2013.
8. Bohnert AS, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011;305(13):1315-1321.
9. Campbell LC, Clauw DJ, Keefe FJ. Persistent pain and depression: A biopsychosocial perspective. Biol Psychiatry. 2003;54(3):399-409.
10. Gatchel RJ, Peng YB, Peters ML, Fuchs PN, Turk DC. The biopsychosocial approach to chronic pain: Scientific advances and future directions. Psychol Bull. 2007;133(4):581-624.
11. Gudin JA, Mogali S, Jones JD, Comer SD. Risks, management, and monitoring of combination opioid, benzodiazepine, and/or alcohol use. Postgrad Med. 2013;125(4):115-130.
12. Centers for Disease Control and Prevention. FastStats: Deaths and mortality. Centers for Disease Control and Prevention Website. http://www.cdc.gov/nchs/fastats/deaths.htm. Updated February 13, 2014. Accessed June 9, 2014.
13. Kemp J, Bossarte R. Suicide Data Report, 2012. U.S. Department of Veterans Affairs Website. http://www.va.gov/opa/docs/Suicide-Data-Report-2012-final.pdf. Accessed July 1, 2014.
14. National Institute of Mental Health. Suicide in the U.S. Statistics. National Institute of Mental Health Website. http://www.nimh.nih.gov/statistics/index.shtml. Accessed June 27, 2014.
15. Miller M, Hempstead K, Nguyen T, Barber C, Rosenberg-Wohl S, Azrael D. Method choice in nonfatal self-harm as a predictor of subsequent episodes of self-harm and suicide: Implications for clinical practice. Am J Public Health. 2013;103(6):e61-e68.
16. Hartl TL, Rosen C, Drescher K, Lee TT, Gusman F. Predicting high-risk behaviors in Veterans with posttraumatic stress disorder. J Nerv Ment Dis. 2005;193(7):464-472.
17. Substance Abuse and Mental Health Services Administration. Results from the 2010 National Survey on Drug Use and Health: Summary of National Findings. NSDUH Series H-41, HHS Publication No. (SMA) 11-4658. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2011.
18. Daubresse M, Chang HY, Yu Y, et al. Ambulatory diagnosis and treatment of nonmalignant pain in the United States, 2000-2010. Med Care. 2013;51(10):870-878.
19. Bray RM, Pemberton MR, Lane ME, Hourani LL, Mattiko MJ, Babeu LA. Substance use and mental health trends among U.S. military active duty personnel. Key findings from the 2008 DoD Health Behavior Survey. Mil Med. 2010;175(6):390-399.
20. Cuevas-Trisan RL. The unfortunate turn of events in pain management. Fed Pract. 2013;30(3):8-9.
1. Brooks D. Hearing Spotlights painkiller overuse among soldiers. http://www.fayobserver.com/military/article_a6e4a2e9-827d-577c-a79a-87a6c07cf151.html. Fayobserver Website. Published October 10, 2013, Accessed June 9, 2014.
2. Seal KH, Shi Y, Cohen BE, Maguen S, Krebs EE, Neylan TC. Association of mental health disorders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012;307(9):940-947.
3. National Institute on Drug Abuse. DrugFacts: Substance Abuse in the Military. http://www.drugabuse.gov/publications/drugfacts/substance-abuse-in-military. National Institute on Drug Abuse Website. Revised March 2013. Accessed June 9, 2014.
4. Grattan A, Sullivan MD, Saunders KW, Campbell CI, Von Korff MR. Depression and prescription opioid misuse among chronic opioid therapy recipients with no history of substance abuse. Ann Fam Med. 2012;10(4):304-311.
5. Pergolizzi JV Jr, Gharibo C, Passik S, et al. Dynamic risk factors in the misuse of opioid analgesics. J Psychosom Res. 2012;72(6):443-451.
6. Dunn KM, Saunders KW, Rutter CM, et al. Opioid prescriptions for chronic pain and overdose: A cohort study. Ann Intern Med. 2010;152(2):85-92.
7. Substance Abuse and Mental Health Services Administration. SAMHSA Opioid Overdose Prevention Toolkit. HHS publication No. (SMA) 13-4742. Rockville, MD: Substance Abuse and Mental Health Service Administration; 2013.
8. Bohnert AS, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011;305(13):1315-1321.
9. Campbell LC, Clauw DJ, Keefe FJ. Persistent pain and depression: A biopsychosocial perspective. Biol Psychiatry. 2003;54(3):399-409.
10. Gatchel RJ, Peng YB, Peters ML, Fuchs PN, Turk DC. The biopsychosocial approach to chronic pain: Scientific advances and future directions. Psychol Bull. 2007;133(4):581-624.
11. Gudin JA, Mogali S, Jones JD, Comer SD. Risks, management, and monitoring of combination opioid, benzodiazepine, and/or alcohol use. Postgrad Med. 2013;125(4):115-130.
12. Centers for Disease Control and Prevention. FastStats: Deaths and mortality. Centers for Disease Control and Prevention Website. http://www.cdc.gov/nchs/fastats/deaths.htm. Updated February 13, 2014. Accessed June 9, 2014.
13. Kemp J, Bossarte R. Suicide Data Report, 2012. U.S. Department of Veterans Affairs Website. http://www.va.gov/opa/docs/Suicide-Data-Report-2012-final.pdf. Accessed July 1, 2014.
14. National Institute of Mental Health. Suicide in the U.S. Statistics. National Institute of Mental Health Website. http://www.nimh.nih.gov/statistics/index.shtml. Accessed June 27, 2014.
15. Miller M, Hempstead K, Nguyen T, Barber C, Rosenberg-Wohl S, Azrael D. Method choice in nonfatal self-harm as a predictor of subsequent episodes of self-harm and suicide: Implications for clinical practice. Am J Public Health. 2013;103(6):e61-e68.
16. Hartl TL, Rosen C, Drescher K, Lee TT, Gusman F. Predicting high-risk behaviors in Veterans with posttraumatic stress disorder. J Nerv Ment Dis. 2005;193(7):464-472.
17. Substance Abuse and Mental Health Services Administration. Results from the 2010 National Survey on Drug Use and Health: Summary of National Findings. NSDUH Series H-41, HHS Publication No. (SMA) 11-4658. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2011.
18. Daubresse M, Chang HY, Yu Y, et al. Ambulatory diagnosis and treatment of nonmalignant pain in the United States, 2000-2010. Med Care. 2013;51(10):870-878.
19. Bray RM, Pemberton MR, Lane ME, Hourani LL, Mattiko MJ, Babeu LA. Substance use and mental health trends among U.S. military active duty personnel. Key findings from the 2008 DoD Health Behavior Survey. Mil Med. 2010;175(6):390-399.
20. Cuevas-Trisan RL. The unfortunate turn of events in pain management. Fed Pract. 2013;30(3):8-9.
Another Reason Not to Smoke: Acute Eosinophilic Pneumonia
The following case was encountered and treated by Capt DellaVolpe during deployment with Operation Enduring Freedom-Trans Sahara on his tour as Special Operations Flight Surgeon with the Air Force Special Operations Command. The case highlights a rare but serious disease that can be particularly challenging to manage in military members serving overseas.
Case Presentation
After 2 days of worsening dyspnea on exertion, fever, and fatigue, a previously healthy 22-year-old man presented to a remote clinic established as part of a military deployment in central Africa. Despite having received azithromycin from a field medic, his condition continued to worsen. He had no cough, recent weight changes, or night sweats. The patient’s past medical history was unremarkable, including any prior history of pulmonary disorders.
The patient was a member of the U.S. Army Military Police Corps and had been deployed for 3 weeks. His job involved local patrols, and he had no history of airborne exposures, such as mold or chemical inhalants, or travel to sandy environments. Although he was previously a nonsmoker, he reported smoking local cigarettes to help him stay awake during night patrols over the past 2 weeks. The patient’s vaccination history included influenza, tetanus, measles/mumps/rubella, yellow fever, typhoid, hepatitis A/B, anthrax, meningococcus, and smallpox, all administered before deployment. At the time of evaluation, his temperature was 103.9°F, pulse 120 bpm, respiratory rate 32 breaths per minute, and blood pressure 110/70 mm Hg. His oxygen saturation was 80% on room air.
On examination, he was in significant distress and only able to speak in short sentences. There was no jugular venous distension or stridor. He was tachycardic, with a regular rhythm, without murmurs, rubs, or gallops. A pulmonary examination revealed decreased air movement bilaterally with bilateral inspiratory crackles at the bases. There was a tactile fremitus on the right side. He had no swelling or tenderness of the extremities, and no rashes were noted.
Laboratory capabilities were limited given the remote clinic location. Rapid malaria and rapid influenza were negative. A blood smear showed no organisms. A chest X-ray showed diffuse alveolar infiltrates and homogenous opacification of the right hemithorax.
The patient was placed on continuous oxygen by facemask and started on IV ceftriaxone and vancomycin. He was volume resuscitated with normal saline, with a modest effect on his heart rate. Attempts to wean his oxygen consumption were accompanied by an immediate oxygen desaturation to the low 80s. Because of the limited supply of oxygen available at the remote location as well as the patient’s poor response to broad-spectrum antibiotic coverage over the next 8 hours, he was evacuated by airborne casualty evacuation to the critical care team at Landstuhl Regional Medical Center in Germany.
Laboratory results revealed a peripheral leukocytosis with no eosinophilia. The patient underwent bronchoalveolar lavage (BAL), which showed 30% eosinophils. A diagnosis of acute eosinophilic pneumonia (AEP) was made, and the patient was started on IV methylprednisolone. He experienced a rapid resolution of symptoms and was completely weaned off oxygen 2 days later. The following week he was released from the hospital and able to return stateside to his unit.
Discussion
This case illustrates an uncommon but potentially life-threatening cause of respiratory failure, AEP. First described as a reversible, noninfectious cause of respiratory distress, AEP is now characterized as an uncommon yet severe febrile illness, which typically presents with hypoxia, pulmonary infiltrates, and increased eosinophilia on bronchoalveolar lavage in the absence of other causes.1-3 Untreated, AEP can progress to respiratory failure and death.
Acute eosinophilic pneumonia belongs to a heterogeneous group of disorders known as the eosinophilic lung syndromes. Although the pathophysiology has not been completely characterized, it is theorized that AEP is caused by a hypersensitivity reaction to an airborne antigen.2 Interleukin-5 has been implicated in the preferential activation of eosinophils and granule release and may be responsible for the eosinophilic alveolar exudates. In addition, interleukin-5 has been established as an eosinophil chemotactant.4-6
Acute eosinophilic pneumonia is relatively uncommon. Most epidemiologic studies have been limited to retrospective analyses and case series.7 However, the presentation of the disease suggests that it may be underdiagnosed.6,8 The most common presenting signs and symptoms are dyspnea, fever, cough, and crackles on inspiration. Acute eosinophilic pneumonia has been documented in men and women of all ages with a 2:1 male predominance, typically occurring in previously healthy individuals aged 18 to 40 years.9,10 Reports of AEP in infants also exist.11
Although idiopathic cases have been described in the literature, patients, on average, will have a history of airborne toxin exposure, including smoke, sand, dust, mold, or chemicals. However, a causal relationship has not been proven.12
Recent initiation of smoking has been associated with AEP, as was the case with this patient. This patient had no other history of airborne exposures, including burn pits, travel to sandy environments, mold, or chemical inhalants. Of all cases of AEP described to date, more than two-thirds have been associated with smoking.13,14 One Japanese case series further established the association through a “cigarette challenge test.”8,15,16 Typically, the patients described were in their mid-20s and rapidly developed symptoms consistent with AEP within 1 month of initiating smoking.
A high proportion of AEP has recently been described among newly deployed military personnel in Iraq.17 Out of 180,000 personnel deployed, 18 developed AEP with 14 having initiated smoking within 1 month of deployment. The authors concluded that compared with the controls, new-onset smokers had a significantly increased risk of developing AEP. Importantly, this case series describes 2 deaths associated with this disease, suggesting that this otherwise healthy population may have other environmental exposures that put them at a higher risk than that in the general population. Another case described a U.S. soldier in Korea with recent smoking history who was diagnosed with AEP on transbronchial lung biopsy.18
Given the nonspecific symptoms of the disease, early diagnosis relies on attention to clinical history, environmental exposures, and response to initial empiric treatment. Diagnosis is made based on pulmonary eosinophilia and exclusion of chronic causes of eosinophilic pulmonary disease, such as Churg-Strauss syndrome, chronic eosinophilic pneumonia, and tropical eosinophilia.9,19
Criteria for diagnosis has evolved since AEP was initially reported. Current criteria include acute onset febrile respiratory manifestations < 1-month duration, bilateral diffuse infiltrates on chest radiograph, arterial oxygen pressure of < 60 mm Hg or pulse oximetry < 90% on room air, and BAL > 25% eosinophils. Blood, sputum, and BAL cultures must be negative for bacteria, fungi, and parasitic causes.7 Exposure to toxins known to cause eosinophilia should also be ruled out. Therefore, timely BAL is of paramount importance in any suspected case of AEP. Peripheral blood count will usually show a nonspecific leukocytosis, with a normal distribution of eosinophils; however, it is not uncommon for peripheral eosinophilia to occur late in the disease.14,20 Pulmonary eosinophilia in the absence of a peripheral eosinophilia is likely due to pulmonary eosinophil sequestration.
In one case series, the mean duration of time from symptom onset until diagnosis was 3.5 days.13 Diagnosis usually occurs after the patient’s clinical status worsens despite broad-spectrum antibiotic therapy. Acute eosinophilic pneumonia typically appears on a chest radiograph as bilateral reticular nodules with interstitial patterning. Variability can exist between alveolar, interstitial, and mixed infiltrate patterns.19 Pleural effusions may also be seen. These nonspecific patterns allow AEP to be easily mistaken for a variety of other pathologies, such as acute respiratory distress syndrome and community acquired pneumonia.
Corticosteroid therapy is the mainstay of treatment for AEP. Intravenous methylprednisolone is typically administered at dosages of 60 mg to 125 mg every 6 hours, followed by an oral prednisone taper.21 Relapses of AEP are not typical, and the prognosis is typically excellent if identified rapidly and treated appropriately. In one study of 127 individuals treated with corticosteroids, all survived, and most were transferred out of the ICU after 3 days of treatment. Dyspnea improved on treatment day 3, and all symptoms disappeared with an average of 7 days of treatment.7
Conclusion
Acute eosinophilic pneumonia is likely to be underreported clinically. Presently, AEP is largely a diagnosis of exclusion; the current criteria for diagnosis are fairly rigid and rely on BAL while ruling out other identifiable causes (Table). Clinical suspicion should be raised in patients with a history of new-onset smoking or other airborne toxin exposure. The broad spectrum of clinical presentations and diagnostic findings leave important questions unanswered regarding the mechanisms of the disease.
This particular case illustrates the fundamental importance of taking a thorough history in any patient with a recent airborne exposure where AEP is suspected. Acute eosinophilic pneumonia should be considered in cases of pneumonia that continue to worsen despite the treatment of IV antibiotics; a BAL should be performed when appropriate.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Allen JN, Pacht ER, Gadek JE, Davis WB. Acute eosinophilic pneumonia as a reversible cause of noninfectious respiratory failure. N Engl J Med. 1989;321(9):569-574.
2. Badesch DB, King TE Jr, Schwarz MI. Acute eosinophilic pneumonia: A hypersensitivity phenomenon? Am Rev Respir Dis. 1989;139(1):249-252.
3. Buchheit J, Eid N, Rodgers G Jr, Feger T, Yakoub O. Acute eosinophilic pneumonia with respiratory failure: A new syndrome? Am Rev Respir Dis. 1992;145(3):716-718.
4. Okubo Y, Hossain M, Kai R, et al. Adhesion molecules on eosinophils in acute eosinophilic pneumonia. Am J Respir Crit Care Med. 1995;151(4):1259-1262.
5. Allen JN, Liao Z, Wewers MD, Altenberger EA, Moore SA, Allen ED. Detection of IL-5 and IL-1 receptor antagonist in bronchoalveolar lavage fluid in acute eosinophilic pneumonia. J Allergy Clin Immunol. 1996;97(6):1366-1374.
6. Faustino L, da Fonseca DM, Takenaka MC, et al. Regulatory T cells migrate to airways via CCR4 and attenuate the severity of airway allergic inflammation. J Immunol. 2013;190(6):2614-2621.
7. Rhee CK, Min KH, Yim NY, et al. Clinical characteristics and corticosteroid treatment of acute eosinophilic pneumonia. Eur Respir J. 2013;41(2):402-409.
8. Shiota Y, Kawai T, Matsumoto H, et al. Acute eosinophilic pneumonia following cigarette smoking. Intern Med. 2000;39(10):830-833.
9. Pope-Harman AL, Davis WB, Allen ED, Christoforidis AJ, Allen JN. Acute eosinophilic pneumonia. A summary of 15 cases and review of the literature. Medicine (Baltimore). 1996;75(6):334-342.
10. Rose DM, Hrncir DE. Primary eosinophilic lung diseases. Allergy Asthma Proc. 2013;34(1):19-25.
11. Park HN, Chung BH, Pyun JE, et al. Idiopathic acute eosinophilic pneumonia in a 14-month-old girl. Korean J Pediatr. 2013;56(1):37-41.
12. Kolb AG, Ives ST, Davies SF. Diagnosis in just over a minute: A case of chronic eosinophilic pneumonia. J Gen Intern Med. 2013;28(7):972-975.
13. Janz DR, O’Neal HR Jr, Ely EW. Acute eosinophilic pneumonia: A case report and review of the literature. Crit Care Med. 2009;37(4):1470-1474.
14. Philit F, Etienne-Mastroïanni B, Parrot A, Guérin C, Robert D, Cordier JF. Idiopathic acute eosinophilic pneumonia: A study of 22 patients. Am J Respir Crit Care Med. 2002;166(9):1235-1239.
15. Miki K, Miki M, Nakamura Y, et al. Early-phase neutrophilia in cigarette smoke-induced acute eosinophilic pneumonia. Intern Med. 2003;42(9):839-845.
16. Watanabe K, Fujimura M, Kasahara K, et al. Acute eosinophilic pneumonia following cigarette smoking: A case report including cigarette-smoking challenge test. Intern Med. 2002;41(11):1016-1020.
17. Shorr AF, Scoville SL, Cersovsky SB, et al. Acute eosinophilic pneumonia among US Military personnel deployed in or near Iraq. JAMA.2004;
292(24):2997-3005.
18. Lim SY, Suh GY, Jeon K. Acute eosinophilic pneumonia presenting as life-threatening hypoxaemia necessitating extracorporeal membrane oxygenation. Int J Tuberc Lung Dis. 2012;16(12):1711-1712.
19. Allen JN, Davis WB. Eosinophilic lung diseases. Am J Respir Crit Care Med. 1994;150(5, pt 1):1423-1438.
20. Hayakawa H, Sato A, Toyoshima M, Imokawa S, Taniguchi M. A clinical study of idiopathic eosinophilic pneumonia. Chest. 1994;105(5):1462-1466.
21. Jantz MA, Sahn SA. Corticosteroids in acute respiratory failure. Am J Respir Crit Care Med. 1999;160(4):1079-1100.
The following case was encountered and treated by Capt DellaVolpe during deployment with Operation Enduring Freedom-Trans Sahara on his tour as Special Operations Flight Surgeon with the Air Force Special Operations Command. The case highlights a rare but serious disease that can be particularly challenging to manage in military members serving overseas.
Case Presentation
After 2 days of worsening dyspnea on exertion, fever, and fatigue, a previously healthy 22-year-old man presented to a remote clinic established as part of a military deployment in central Africa. Despite having received azithromycin from a field medic, his condition continued to worsen. He had no cough, recent weight changes, or night sweats. The patient’s past medical history was unremarkable, including any prior history of pulmonary disorders.
The patient was a member of the U.S. Army Military Police Corps and had been deployed for 3 weeks. His job involved local patrols, and he had no history of airborne exposures, such as mold or chemical inhalants, or travel to sandy environments. Although he was previously a nonsmoker, he reported smoking local cigarettes to help him stay awake during night patrols over the past 2 weeks. The patient’s vaccination history included influenza, tetanus, measles/mumps/rubella, yellow fever, typhoid, hepatitis A/B, anthrax, meningococcus, and smallpox, all administered before deployment. At the time of evaluation, his temperature was 103.9°F, pulse 120 bpm, respiratory rate 32 breaths per minute, and blood pressure 110/70 mm Hg. His oxygen saturation was 80% on room air.
On examination, he was in significant distress and only able to speak in short sentences. There was no jugular venous distension or stridor. He was tachycardic, with a regular rhythm, without murmurs, rubs, or gallops. A pulmonary examination revealed decreased air movement bilaterally with bilateral inspiratory crackles at the bases. There was a tactile fremitus on the right side. He had no swelling or tenderness of the extremities, and no rashes were noted.
Laboratory capabilities were limited given the remote clinic location. Rapid malaria and rapid influenza were negative. A blood smear showed no organisms. A chest X-ray showed diffuse alveolar infiltrates and homogenous opacification of the right hemithorax.
The patient was placed on continuous oxygen by facemask and started on IV ceftriaxone and vancomycin. He was volume resuscitated with normal saline, with a modest effect on his heart rate. Attempts to wean his oxygen consumption were accompanied by an immediate oxygen desaturation to the low 80s. Because of the limited supply of oxygen available at the remote location as well as the patient’s poor response to broad-spectrum antibiotic coverage over the next 8 hours, he was evacuated by airborne casualty evacuation to the critical care team at Landstuhl Regional Medical Center in Germany.
Laboratory results revealed a peripheral leukocytosis with no eosinophilia. The patient underwent bronchoalveolar lavage (BAL), which showed 30% eosinophils. A diagnosis of acute eosinophilic pneumonia (AEP) was made, and the patient was started on IV methylprednisolone. He experienced a rapid resolution of symptoms and was completely weaned off oxygen 2 days later. The following week he was released from the hospital and able to return stateside to his unit.
Discussion
This case illustrates an uncommon but potentially life-threatening cause of respiratory failure, AEP. First described as a reversible, noninfectious cause of respiratory distress, AEP is now characterized as an uncommon yet severe febrile illness, which typically presents with hypoxia, pulmonary infiltrates, and increased eosinophilia on bronchoalveolar lavage in the absence of other causes.1-3 Untreated, AEP can progress to respiratory failure and death.
Acute eosinophilic pneumonia belongs to a heterogeneous group of disorders known as the eosinophilic lung syndromes. Although the pathophysiology has not been completely characterized, it is theorized that AEP is caused by a hypersensitivity reaction to an airborne antigen.2 Interleukin-5 has been implicated in the preferential activation of eosinophils and granule release and may be responsible for the eosinophilic alveolar exudates. In addition, interleukin-5 has been established as an eosinophil chemotactant.4-6
Acute eosinophilic pneumonia is relatively uncommon. Most epidemiologic studies have been limited to retrospective analyses and case series.7 However, the presentation of the disease suggests that it may be underdiagnosed.6,8 The most common presenting signs and symptoms are dyspnea, fever, cough, and crackles on inspiration. Acute eosinophilic pneumonia has been documented in men and women of all ages with a 2:1 male predominance, typically occurring in previously healthy individuals aged 18 to 40 years.9,10 Reports of AEP in infants also exist.11
Although idiopathic cases have been described in the literature, patients, on average, will have a history of airborne toxin exposure, including smoke, sand, dust, mold, or chemicals. However, a causal relationship has not been proven.12
Recent initiation of smoking has been associated with AEP, as was the case with this patient. This patient had no other history of airborne exposures, including burn pits, travel to sandy environments, mold, or chemical inhalants. Of all cases of AEP described to date, more than two-thirds have been associated with smoking.13,14 One Japanese case series further established the association through a “cigarette challenge test.”8,15,16 Typically, the patients described were in their mid-20s and rapidly developed symptoms consistent with AEP within 1 month of initiating smoking.
A high proportion of AEP has recently been described among newly deployed military personnel in Iraq.17 Out of 180,000 personnel deployed, 18 developed AEP with 14 having initiated smoking within 1 month of deployment. The authors concluded that compared with the controls, new-onset smokers had a significantly increased risk of developing AEP. Importantly, this case series describes 2 deaths associated with this disease, suggesting that this otherwise healthy population may have other environmental exposures that put them at a higher risk than that in the general population. Another case described a U.S. soldier in Korea with recent smoking history who was diagnosed with AEP on transbronchial lung biopsy.18
Given the nonspecific symptoms of the disease, early diagnosis relies on attention to clinical history, environmental exposures, and response to initial empiric treatment. Diagnosis is made based on pulmonary eosinophilia and exclusion of chronic causes of eosinophilic pulmonary disease, such as Churg-Strauss syndrome, chronic eosinophilic pneumonia, and tropical eosinophilia.9,19
Criteria for diagnosis has evolved since AEP was initially reported. Current criteria include acute onset febrile respiratory manifestations < 1-month duration, bilateral diffuse infiltrates on chest radiograph, arterial oxygen pressure of < 60 mm Hg or pulse oximetry < 90% on room air, and BAL > 25% eosinophils. Blood, sputum, and BAL cultures must be negative for bacteria, fungi, and parasitic causes.7 Exposure to toxins known to cause eosinophilia should also be ruled out. Therefore, timely BAL is of paramount importance in any suspected case of AEP. Peripheral blood count will usually show a nonspecific leukocytosis, with a normal distribution of eosinophils; however, it is not uncommon for peripheral eosinophilia to occur late in the disease.14,20 Pulmonary eosinophilia in the absence of a peripheral eosinophilia is likely due to pulmonary eosinophil sequestration.
In one case series, the mean duration of time from symptom onset until diagnosis was 3.5 days.13 Diagnosis usually occurs after the patient’s clinical status worsens despite broad-spectrum antibiotic therapy. Acute eosinophilic pneumonia typically appears on a chest radiograph as bilateral reticular nodules with interstitial patterning. Variability can exist between alveolar, interstitial, and mixed infiltrate patterns.19 Pleural effusions may also be seen. These nonspecific patterns allow AEP to be easily mistaken for a variety of other pathologies, such as acute respiratory distress syndrome and community acquired pneumonia.
Corticosteroid therapy is the mainstay of treatment for AEP. Intravenous methylprednisolone is typically administered at dosages of 60 mg to 125 mg every 6 hours, followed by an oral prednisone taper.21 Relapses of AEP are not typical, and the prognosis is typically excellent if identified rapidly and treated appropriately. In one study of 127 individuals treated with corticosteroids, all survived, and most were transferred out of the ICU after 3 days of treatment. Dyspnea improved on treatment day 3, and all symptoms disappeared with an average of 7 days of treatment.7
Conclusion
Acute eosinophilic pneumonia is likely to be underreported clinically. Presently, AEP is largely a diagnosis of exclusion; the current criteria for diagnosis are fairly rigid and rely on BAL while ruling out other identifiable causes (Table). Clinical suspicion should be raised in patients with a history of new-onset smoking or other airborne toxin exposure. The broad spectrum of clinical presentations and diagnostic findings leave important questions unanswered regarding the mechanisms of the disease.
This particular case illustrates the fundamental importance of taking a thorough history in any patient with a recent airborne exposure where AEP is suspected. Acute eosinophilic pneumonia should be considered in cases of pneumonia that continue to worsen despite the treatment of IV antibiotics; a BAL should be performed when appropriate.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
The following case was encountered and treated by Capt DellaVolpe during deployment with Operation Enduring Freedom-Trans Sahara on his tour as Special Operations Flight Surgeon with the Air Force Special Operations Command. The case highlights a rare but serious disease that can be particularly challenging to manage in military members serving overseas.
Case Presentation
After 2 days of worsening dyspnea on exertion, fever, and fatigue, a previously healthy 22-year-old man presented to a remote clinic established as part of a military deployment in central Africa. Despite having received azithromycin from a field medic, his condition continued to worsen. He had no cough, recent weight changes, or night sweats. The patient’s past medical history was unremarkable, including any prior history of pulmonary disorders.
The patient was a member of the U.S. Army Military Police Corps and had been deployed for 3 weeks. His job involved local patrols, and he had no history of airborne exposures, such as mold or chemical inhalants, or travel to sandy environments. Although he was previously a nonsmoker, he reported smoking local cigarettes to help him stay awake during night patrols over the past 2 weeks. The patient’s vaccination history included influenza, tetanus, measles/mumps/rubella, yellow fever, typhoid, hepatitis A/B, anthrax, meningococcus, and smallpox, all administered before deployment. At the time of evaluation, his temperature was 103.9°F, pulse 120 bpm, respiratory rate 32 breaths per minute, and blood pressure 110/70 mm Hg. His oxygen saturation was 80% on room air.
On examination, he was in significant distress and only able to speak in short sentences. There was no jugular venous distension or stridor. He was tachycardic, with a regular rhythm, without murmurs, rubs, or gallops. A pulmonary examination revealed decreased air movement bilaterally with bilateral inspiratory crackles at the bases. There was a tactile fremitus on the right side. He had no swelling or tenderness of the extremities, and no rashes were noted.
Laboratory capabilities were limited given the remote clinic location. Rapid malaria and rapid influenza were negative. A blood smear showed no organisms. A chest X-ray showed diffuse alveolar infiltrates and homogenous opacification of the right hemithorax.
The patient was placed on continuous oxygen by facemask and started on IV ceftriaxone and vancomycin. He was volume resuscitated with normal saline, with a modest effect on his heart rate. Attempts to wean his oxygen consumption were accompanied by an immediate oxygen desaturation to the low 80s. Because of the limited supply of oxygen available at the remote location as well as the patient’s poor response to broad-spectrum antibiotic coverage over the next 8 hours, he was evacuated by airborne casualty evacuation to the critical care team at Landstuhl Regional Medical Center in Germany.
Laboratory results revealed a peripheral leukocytosis with no eosinophilia. The patient underwent bronchoalveolar lavage (BAL), which showed 30% eosinophils. A diagnosis of acute eosinophilic pneumonia (AEP) was made, and the patient was started on IV methylprednisolone. He experienced a rapid resolution of symptoms and was completely weaned off oxygen 2 days later. The following week he was released from the hospital and able to return stateside to his unit.
Discussion
This case illustrates an uncommon but potentially life-threatening cause of respiratory failure, AEP. First described as a reversible, noninfectious cause of respiratory distress, AEP is now characterized as an uncommon yet severe febrile illness, which typically presents with hypoxia, pulmonary infiltrates, and increased eosinophilia on bronchoalveolar lavage in the absence of other causes.1-3 Untreated, AEP can progress to respiratory failure and death.
Acute eosinophilic pneumonia belongs to a heterogeneous group of disorders known as the eosinophilic lung syndromes. Although the pathophysiology has not been completely characterized, it is theorized that AEP is caused by a hypersensitivity reaction to an airborne antigen.2 Interleukin-5 has been implicated in the preferential activation of eosinophils and granule release and may be responsible for the eosinophilic alveolar exudates. In addition, interleukin-5 has been established as an eosinophil chemotactant.4-6
Acute eosinophilic pneumonia is relatively uncommon. Most epidemiologic studies have been limited to retrospective analyses and case series.7 However, the presentation of the disease suggests that it may be underdiagnosed.6,8 The most common presenting signs and symptoms are dyspnea, fever, cough, and crackles on inspiration. Acute eosinophilic pneumonia has been documented in men and women of all ages with a 2:1 male predominance, typically occurring in previously healthy individuals aged 18 to 40 years.9,10 Reports of AEP in infants also exist.11
Although idiopathic cases have been described in the literature, patients, on average, will have a history of airborne toxin exposure, including smoke, sand, dust, mold, or chemicals. However, a causal relationship has not been proven.12
Recent initiation of smoking has been associated with AEP, as was the case with this patient. This patient had no other history of airborne exposures, including burn pits, travel to sandy environments, mold, or chemical inhalants. Of all cases of AEP described to date, more than two-thirds have been associated with smoking.13,14 One Japanese case series further established the association through a “cigarette challenge test.”8,15,16 Typically, the patients described were in their mid-20s and rapidly developed symptoms consistent with AEP within 1 month of initiating smoking.
A high proportion of AEP has recently been described among newly deployed military personnel in Iraq.17 Out of 180,000 personnel deployed, 18 developed AEP with 14 having initiated smoking within 1 month of deployment. The authors concluded that compared with the controls, new-onset smokers had a significantly increased risk of developing AEP. Importantly, this case series describes 2 deaths associated with this disease, suggesting that this otherwise healthy population may have other environmental exposures that put them at a higher risk than that in the general population. Another case described a U.S. soldier in Korea with recent smoking history who was diagnosed with AEP on transbronchial lung biopsy.18
Given the nonspecific symptoms of the disease, early diagnosis relies on attention to clinical history, environmental exposures, and response to initial empiric treatment. Diagnosis is made based on pulmonary eosinophilia and exclusion of chronic causes of eosinophilic pulmonary disease, such as Churg-Strauss syndrome, chronic eosinophilic pneumonia, and tropical eosinophilia.9,19
Criteria for diagnosis has evolved since AEP was initially reported. Current criteria include acute onset febrile respiratory manifestations < 1-month duration, bilateral diffuse infiltrates on chest radiograph, arterial oxygen pressure of < 60 mm Hg or pulse oximetry < 90% on room air, and BAL > 25% eosinophils. Blood, sputum, and BAL cultures must be negative for bacteria, fungi, and parasitic causes.7 Exposure to toxins known to cause eosinophilia should also be ruled out. Therefore, timely BAL is of paramount importance in any suspected case of AEP. Peripheral blood count will usually show a nonspecific leukocytosis, with a normal distribution of eosinophils; however, it is not uncommon for peripheral eosinophilia to occur late in the disease.14,20 Pulmonary eosinophilia in the absence of a peripheral eosinophilia is likely due to pulmonary eosinophil sequestration.
In one case series, the mean duration of time from symptom onset until diagnosis was 3.5 days.13 Diagnosis usually occurs after the patient’s clinical status worsens despite broad-spectrum antibiotic therapy. Acute eosinophilic pneumonia typically appears on a chest radiograph as bilateral reticular nodules with interstitial patterning. Variability can exist between alveolar, interstitial, and mixed infiltrate patterns.19 Pleural effusions may also be seen. These nonspecific patterns allow AEP to be easily mistaken for a variety of other pathologies, such as acute respiratory distress syndrome and community acquired pneumonia.
Corticosteroid therapy is the mainstay of treatment for AEP. Intravenous methylprednisolone is typically administered at dosages of 60 mg to 125 mg every 6 hours, followed by an oral prednisone taper.21 Relapses of AEP are not typical, and the prognosis is typically excellent if identified rapidly and treated appropriately. In one study of 127 individuals treated with corticosteroids, all survived, and most were transferred out of the ICU after 3 days of treatment. Dyspnea improved on treatment day 3, and all symptoms disappeared with an average of 7 days of treatment.7
Conclusion
Acute eosinophilic pneumonia is likely to be underreported clinically. Presently, AEP is largely a diagnosis of exclusion; the current criteria for diagnosis are fairly rigid and rely on BAL while ruling out other identifiable causes (Table). Clinical suspicion should be raised in patients with a history of new-onset smoking or other airborne toxin exposure. The broad spectrum of clinical presentations and diagnostic findings leave important questions unanswered regarding the mechanisms of the disease.
This particular case illustrates the fundamental importance of taking a thorough history in any patient with a recent airborne exposure where AEP is suspected. Acute eosinophilic pneumonia should be considered in cases of pneumonia that continue to worsen despite the treatment of IV antibiotics; a BAL should be performed when appropriate.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Allen JN, Pacht ER, Gadek JE, Davis WB. Acute eosinophilic pneumonia as a reversible cause of noninfectious respiratory failure. N Engl J Med. 1989;321(9):569-574.
2. Badesch DB, King TE Jr, Schwarz MI. Acute eosinophilic pneumonia: A hypersensitivity phenomenon? Am Rev Respir Dis. 1989;139(1):249-252.
3. Buchheit J, Eid N, Rodgers G Jr, Feger T, Yakoub O. Acute eosinophilic pneumonia with respiratory failure: A new syndrome? Am Rev Respir Dis. 1992;145(3):716-718.
4. Okubo Y, Hossain M, Kai R, et al. Adhesion molecules on eosinophils in acute eosinophilic pneumonia. Am J Respir Crit Care Med. 1995;151(4):1259-1262.
5. Allen JN, Liao Z, Wewers MD, Altenberger EA, Moore SA, Allen ED. Detection of IL-5 and IL-1 receptor antagonist in bronchoalveolar lavage fluid in acute eosinophilic pneumonia. J Allergy Clin Immunol. 1996;97(6):1366-1374.
6. Faustino L, da Fonseca DM, Takenaka MC, et al. Regulatory T cells migrate to airways via CCR4 and attenuate the severity of airway allergic inflammation. J Immunol. 2013;190(6):2614-2621.
7. Rhee CK, Min KH, Yim NY, et al. Clinical characteristics and corticosteroid treatment of acute eosinophilic pneumonia. Eur Respir J. 2013;41(2):402-409.
8. Shiota Y, Kawai T, Matsumoto H, et al. Acute eosinophilic pneumonia following cigarette smoking. Intern Med. 2000;39(10):830-833.
9. Pope-Harman AL, Davis WB, Allen ED, Christoforidis AJ, Allen JN. Acute eosinophilic pneumonia. A summary of 15 cases and review of the literature. Medicine (Baltimore). 1996;75(6):334-342.
10. Rose DM, Hrncir DE. Primary eosinophilic lung diseases. Allergy Asthma Proc. 2013;34(1):19-25.
11. Park HN, Chung BH, Pyun JE, et al. Idiopathic acute eosinophilic pneumonia in a 14-month-old girl. Korean J Pediatr. 2013;56(1):37-41.
12. Kolb AG, Ives ST, Davies SF. Diagnosis in just over a minute: A case of chronic eosinophilic pneumonia. J Gen Intern Med. 2013;28(7):972-975.
13. Janz DR, O’Neal HR Jr, Ely EW. Acute eosinophilic pneumonia: A case report and review of the literature. Crit Care Med. 2009;37(4):1470-1474.
14. Philit F, Etienne-Mastroïanni B, Parrot A, Guérin C, Robert D, Cordier JF. Idiopathic acute eosinophilic pneumonia: A study of 22 patients. Am J Respir Crit Care Med. 2002;166(9):1235-1239.
15. Miki K, Miki M, Nakamura Y, et al. Early-phase neutrophilia in cigarette smoke-induced acute eosinophilic pneumonia. Intern Med. 2003;42(9):839-845.
16. Watanabe K, Fujimura M, Kasahara K, et al. Acute eosinophilic pneumonia following cigarette smoking: A case report including cigarette-smoking challenge test. Intern Med. 2002;41(11):1016-1020.
17. Shorr AF, Scoville SL, Cersovsky SB, et al. Acute eosinophilic pneumonia among US Military personnel deployed in or near Iraq. JAMA.2004;
292(24):2997-3005.
18. Lim SY, Suh GY, Jeon K. Acute eosinophilic pneumonia presenting as life-threatening hypoxaemia necessitating extracorporeal membrane oxygenation. Int J Tuberc Lung Dis. 2012;16(12):1711-1712.
19. Allen JN, Davis WB. Eosinophilic lung diseases. Am J Respir Crit Care Med. 1994;150(5, pt 1):1423-1438.
20. Hayakawa H, Sato A, Toyoshima M, Imokawa S, Taniguchi M. A clinical study of idiopathic eosinophilic pneumonia. Chest. 1994;105(5):1462-1466.
21. Jantz MA, Sahn SA. Corticosteroids in acute respiratory failure. Am J Respir Crit Care Med. 1999;160(4):1079-1100.
1. Allen JN, Pacht ER, Gadek JE, Davis WB. Acute eosinophilic pneumonia as a reversible cause of noninfectious respiratory failure. N Engl J Med. 1989;321(9):569-574.
2. Badesch DB, King TE Jr, Schwarz MI. Acute eosinophilic pneumonia: A hypersensitivity phenomenon? Am Rev Respir Dis. 1989;139(1):249-252.
3. Buchheit J, Eid N, Rodgers G Jr, Feger T, Yakoub O. Acute eosinophilic pneumonia with respiratory failure: A new syndrome? Am Rev Respir Dis. 1992;145(3):716-718.
4. Okubo Y, Hossain M, Kai R, et al. Adhesion molecules on eosinophils in acute eosinophilic pneumonia. Am J Respir Crit Care Med. 1995;151(4):1259-1262.
5. Allen JN, Liao Z, Wewers MD, Altenberger EA, Moore SA, Allen ED. Detection of IL-5 and IL-1 receptor antagonist in bronchoalveolar lavage fluid in acute eosinophilic pneumonia. J Allergy Clin Immunol. 1996;97(6):1366-1374.
6. Faustino L, da Fonseca DM, Takenaka MC, et al. Regulatory T cells migrate to airways via CCR4 and attenuate the severity of airway allergic inflammation. J Immunol. 2013;190(6):2614-2621.
7. Rhee CK, Min KH, Yim NY, et al. Clinical characteristics and corticosteroid treatment of acute eosinophilic pneumonia. Eur Respir J. 2013;41(2):402-409.
8. Shiota Y, Kawai T, Matsumoto H, et al. Acute eosinophilic pneumonia following cigarette smoking. Intern Med. 2000;39(10):830-833.
9. Pope-Harman AL, Davis WB, Allen ED, Christoforidis AJ, Allen JN. Acute eosinophilic pneumonia. A summary of 15 cases and review of the literature. Medicine (Baltimore). 1996;75(6):334-342.
10. Rose DM, Hrncir DE. Primary eosinophilic lung diseases. Allergy Asthma Proc. 2013;34(1):19-25.
11. Park HN, Chung BH, Pyun JE, et al. Idiopathic acute eosinophilic pneumonia in a 14-month-old girl. Korean J Pediatr. 2013;56(1):37-41.
12. Kolb AG, Ives ST, Davies SF. Diagnosis in just over a minute: A case of chronic eosinophilic pneumonia. J Gen Intern Med. 2013;28(7):972-975.
13. Janz DR, O’Neal HR Jr, Ely EW. Acute eosinophilic pneumonia: A case report and review of the literature. Crit Care Med. 2009;37(4):1470-1474.
14. Philit F, Etienne-Mastroïanni B, Parrot A, Guérin C, Robert D, Cordier JF. Idiopathic acute eosinophilic pneumonia: A study of 22 patients. Am J Respir Crit Care Med. 2002;166(9):1235-1239.
15. Miki K, Miki M, Nakamura Y, et al. Early-phase neutrophilia in cigarette smoke-induced acute eosinophilic pneumonia. Intern Med. 2003;42(9):839-845.
16. Watanabe K, Fujimura M, Kasahara K, et al. Acute eosinophilic pneumonia following cigarette smoking: A case report including cigarette-smoking challenge test. Intern Med. 2002;41(11):1016-1020.
17. Shorr AF, Scoville SL, Cersovsky SB, et al. Acute eosinophilic pneumonia among US Military personnel deployed in or near Iraq. JAMA.2004;
292(24):2997-3005.
18. Lim SY, Suh GY, Jeon K. Acute eosinophilic pneumonia presenting as life-threatening hypoxaemia necessitating extracorporeal membrane oxygenation. Int J Tuberc Lung Dis. 2012;16(12):1711-1712.
19. Allen JN, Davis WB. Eosinophilic lung diseases. Am J Respir Crit Care Med. 1994;150(5, pt 1):1423-1438.
20. Hayakawa H, Sato A, Toyoshima M, Imokawa S, Taniguchi M. A clinical study of idiopathic eosinophilic pneumonia. Chest. 1994;105(5):1462-1466.
21. Jantz MA, Sahn SA. Corticosteroids in acute respiratory failure. Am J Respir Crit Care Med. 1999;160(4):1079-1100.
Pharmacists in the Emergency Department: Feasibility and Cost
Clinical pharmacists have expanded their role over the past few decades in both outpatient and inpatient settings and are now members of an interdisciplinary health care team that includes nutritionists, physical therapists, physicians, and nurses.1,2 The emergency department (ED), however, has lagged behind in the inclusion of pharmacists.3 Despite well documented financial and ED operational benefits of pharmacists and the recommendation of their inclusion by the Institute of Medicine, only about 30% of academic EDs in a 2009 survey employed a pharmacist.4-8 A larger 2005 survey of 510 hospital pharmacy directors revealed that only 3.5% of hospitals sampled (academic and nonacademic) provided clinical pharmacy services in the ED.9
About 3.8 million annual preventable medical errors occur in the ED, giving the ED the highest rate of medication errors among all hospital departments.4 In 2000, Schenkel found that 3% of all inpatient medication errors were initiated in the ED.10 Similarly, Chin and colleagues found that 3.6% of elderly patients were administered an inappropriate medication in the ED with 5.6% receiving an inappropriate prescription at discharge.11
In a 2008 study conducted at the Durham VAMC in North Carolina, Hastings and colleagues found that suboptimal pharmacy was common among elderly veterans discharged from the ED (11%) and that potentially inappropriate medication use was associated with a 32% greater risk of repeat ED visits, hospitalization, or death (P = .10).12 In 2010, Rothschild and colleagues found 7.8 medication errors per 100 ED patients or 2.9 errors per 100 prescribed medications.13 Despite this unacceptably high rate of medication errors, most EDs do not employ pharmacy specialists or have a pharmacist easily available for consultation—options that could not only streamline ED operations, but also reduce patient risk.
The pharmacist role in the ED has changed considerably. In the 1970s, ED pharmacists were used mainly to dispense medicine, maintain inventories, and participate in cardiopulmonary resuscitation.3,14,15 Today, following the guidelines set by the American Society for Health-System Pharmacists, emergency pharmacists have an expanded, more direct role in patient care and evaluation and support of the physicians and other ED staff who work alongside them.4,14,16,17 Pharmacists gather accurate and complete medication histories, review and reconcile medication lists, and screen ED medication orders for errors or anticipated drug interactions.13,18-23 They adjust medication doses on a patient-by-patient basis, accounting for renal and hepatic clearance and closely monitor patients for treatment response. They also provide one-on-one patient education on medication dosing, administration, adverse drug events (ADEs), and interactions, increasing patients’ drug knowledge and adherence.17,24 Pharmacists provide information to patients on vaccinations and medication assistance programs, which is unlikely to be shared by other providers.3,19,20 Pharmacists in the ED reduce medication delays and omissions that occur in admitted patients staying in the ED.25,26
Aside from patient education, clinical pharmacists have an important role in providing education and consultation to ED physicians, midlevel providers, and house staff on topics that include availability of new medications and local antibiotic resistance patterns.14,27-29 Additionally, pharmacists monitor drug supplies and restock medications to avoid shortages during critical moments, offer the ED perspective in hospital formulary reviews, and increase efficiency and throughput in the ED while decreasing costs by evaluating and treating patients who present simply for prescription refills alongside a supervising physician.14
With this in mind, the ED of the Atlanta VAMC in Decatur, Georgia, conducted a pilot study to assess the financial and logistic feasibility of a full-time pharmacist in the ED setting with the hope that a pharmacist would integrate well into the health care team, reducing overall departmental expense and the risk of medication error associated with patient harm and simultaneously improving patient satisfaction and departmental efficiency.
Methodology
The ED of the Atlanta VAMC is part of a tertiary care teaching hospital affiliated with both the Emory and Morehouse schools of medicine. At the time of the pilot, the facility had 128 acute care medical/surgical beds, 12 inpatient palliative care beds, 40 acute care psychiatric beds, 24 medical surgical intensive care unit beds, and 60 inpatient nursing home beds. The ED provides care to > 37,000 veterans annually, and in December 2011 when this study was conducted, 3,195 veterans were seen in the ED.
The ED was divided into the main ED and the urgent care. Patient intake occurred through a centralized triage, and based on acuity, patients were sent to the appropriate setting for treatment. The ED used a 5-tier triage system. Patients with triage levels 1, 2, and 3 were sent to the main ED, and patients with triage levels 4 and 5 were sent to the urgent care.
Pharmacists
Pharmacy services were provided by 5 residency-trained doctors of pharmacy employed by the medical center working as clinical pharmacists with the inpatient medical teams at the time of the pilot. The pilot was conducted over a 2-week period in December 2011, Monday through Friday, for a total of 10 days. The clinical pharmacists divided the days among themselves. Each pharmacist provided services for a total of 2 days, 3 hours per day, from about 3 pm to 6 pm. The pharmacists were given a room previously used as a physician workroom in which to evaluate patients.
Patient Selection
Patients to be seen by the clinical pharmacist were chosen by the triage nurse, the charge nurse, the ED physician, the urgent care provider (physician or midlevel provider), or by the pharmacists. The triage nurse or charge nurse, based on chief medical problem and acuity, chose patients directly out of triage. Only patients with triage acuity level 4 or 5 were taken directly from triage without first seeing a physician or midlevel provider. These patients presented with the chief problem of medication refill or coumadin/International Normalized Ratio check. Once chosen as appropriate for the clinical pharmacist, the charge nurse helped with patient flow, and if the pharmacist was occupied with other patients, the nurse redirected the patient to urgent care.
Additional patients were chosen to see the clinical pharmacist after an evaluation of their initial problem was completed by a physician or midlevel practitioner in the urgent care or main ED. Patients whom the provider felt could benefit from any of the following services were directed to the clinical pharmacist: anticoagulation consult, diabetic education, pharmacokinetic consult, medication history, medication reconciliation, formulary management, medication refills, therapeutic interchange, screening for drug interactions, allergy review, and nonformulary or restricted medications requests. Additionally, the clinical pharmacist reviewed the charts of patients in the main ED whom they were not asked to see. They offered assistance when needed in all the aforementioned areas and for order clarification, assuring IV compatibility, reporting medication errors and ADEs, promotion of safe medical practices, and elimination of duplicate/redundant medications.
Data Collection
The pharmacists developed a log to record their activities. The log included the date and time of the intervention, number of minutes spent with the patient, the reason for intervention, and recommendations, if applicable. They categorized their interventions into 16 categories: anticoagulation, pharmacokinetics, drug information, order clarification, medication reconciliation, therapeutic interchange, formulary management, medication history, IV compatibility, screening for drug interactions, patient education, allergy documentation, promotion of safe medical practices, reporting of medication error/ADEs, nonformulary and restricted medication requests, and prescription refills. Patients could receive more than 1 intervention.
Though not a focus of this pilot, all patients seen by a pharmacist received a postencounter survey seeking their opinion on whether the pharmacist improved the value of their visit.
Review Process
At the conclusion of the pilot, 2 independent reviewers, both physicians, reviewed the logs, and each task was reassigned to 1 of 8 categories. These categories included either medication refills or 1 of 7 other areas that had established cost avoidance estimates from 2 other well accepted studies (Lee and colleagues and Ling and colleagues).30,31 These 7 categories included adjusting dose or frequency of medication, elimination of duplication of therapy, education/information inquiry, formulary management, prevention and management of ADEs, prevention or management of allergies, and therapeutic interchange. If the independent reviewers did not have initial concordance of classification of the intervention, they discussed the intervention and came to an agreement.
Cost Analysis
Cost avoidance estimates for 7 individual interventions were made, using data from Lee and colleagues and Ling and colleagues.30,31 Four of these came from the study by Lee and colleagues: prevent or manage drug allergy, adjust dosage or frequency, prevent or manage ADEs, and eliminate duplication of therapy.30 Lee and colleagues’ “drug interaction” group was not clearly defined, thus this was included with the “prevent or manage ADE” group. Ling and colleagues provided data for the 3 additional groups of interventions that pharmacists performed: education and information inquiry, formulary management, and therapeutic interchange.31
Financial estimates of cost avoidance were adjusted for inflation, using the consumer price index (CPI) of the U.S. Bureau of Labor Statistics.32 The Lee study was conducted in 2002, and estimates for cost avoidance using their model were adjusted to 2011 values using the CPI inflation rate of 25%. The Ling study was conducted in 2005, and estimates for cost avoidance using their model were adjusted for 2011 values using the CPI rate of inflation of 15.2%.32
For the remaining intervention, prescription refill, cost savings was determined by calculating the average times spent by the ED pharmacist on each intervention and then using the difference between hourly physician and pharmacist pay (about $50/h difference based on VA wage tables).
RESULTS
During the 30-hour total time in which a pharmacist was present in the ED, a total of 42 patients were assisted through 71 interventions (Table 1).
Pharmacists provided a diverse range of services to patients in the ED. The most common intervention was education and/or information inquiry. Tasks in this category included patient education about medication dosing, administration, AEs, interactions and warnings, as well as diabetes management. In several instances, education was provided to attending physicians or house staff, though it should be noted that this provider education was not counted as an intervention for this study unless it was associated with a patient (of which there were 3 total instances, eg, instruction on how to choose the proper insulin syringe).
Interventions, when a medication list was screened by the ED pharmacist for interactions or when drug choices were recommended to the physician or midlevel providers, were counted as prevention and management of ADEs. For example, the pharmacist noted a patient with a new diagnosis of gout who was prescribed hydrochlorothiazide; this was brought to the attention of the provider and alternative antihypertensives were suggested. In another instance, a patient was found to be on both ibuprofen and enoxaparin; the treating physician was alerted of this potential interaction. There were 15 such events in total.
Several other interventions arose from the screenings for ADEs, including adjusting dose or frequency of medication (11); therapeutic interchange (5); eliminating duplication of therapy (2); and prevention or management of allergies (1). Cases included hepatic and/or renal dose changes, substituting equivalent medications for better treatment outcome or adherence, or discontinuing 2 or more medications in a patient’s medication profile that were considered duplication.
During the pharmacist screening, one patient who had piperacillin/tazobactam ordered in the ED had a penicillin allergy. This intervention was categorized as prevention and management of an ADE as well as prevention and management of allergies. Interventions not accompanied by the “prevention of ADE” category included those in which the change did not provide a clear risk reduction. For example, one therapeutic interchange was from levofloxacin to moxifloxacin for a better-anticipated therapy. Another was a metformin dose increase, presumably for improved glycemic control.
Prescription refills occurred with the same frequency as prevention of ADEs.15 This intervention led in some cases to switching to pharmaceutical equivalents when a drug prescribed at another facility was not on the formulary. Other drugs that were not on the preferred list but available with nonformulary medication requests were ordered or approved with the assistance of the pharmacist. The pharmacist’s direct involvement significantly reduced the initial contact-to-approval time for these patients.
After tallying the total number of interventions, the potential financial cost savings to the ED were determined (Table 2). As mentioned previously, the Lee and Ling studies provided the categories for classification of 7 pharmacist interactions. The estimated cost avoidance for the 4 applicable groups from the Lee study had inflation-corrected values of $1,486 per adjusted dose or frequency of medication, $205 per elimination of duplication of therapy, $1,374 per prevention or management of ADEs, and $1,721 per prevention or management of allergies.30
The estimated cost avoidance for the 3 applicable groups from the Ling study had inflation-corrected values of $512.38 per education/information inquiry, $174.80 per formulary management, and $174.80 per therapeutic interchange.31 The eighth group, prescription refills, was valued at $12.50 each, using the difference between physician and pharmacist salary for an average of 15 minutes per interaction.
When multiplied by the number of interventions in each of these groups, the total potential cost avoidance in the study period was about $40,136.48. Extrapolated into a yearly amount, that is a $2,782,795.94 potential cost savings for the medical center.
Seventeen of the 42 (40.5%) postencounter surveys from the patients seen by the pharmacists were received. Of these veterans, 100% reported that they were “extremely satisfied” with the treatment they had received during their visit to the ED.
DISCUSSION
There is the potential for significant cost avoidance by adding a single full-time pharmacist to the ED: Annually, more than $2.7 million in potential savings for the medical center. Though surprising, this figure is actually in line with the much larger study by Lada and colleagues in which an estimated $3 million was avoided.15 At the same hospital 12 years earlier, Levy noted about $1 million in cost avoidance (not inflation-adjusted).33 The Ling study, however, did not have as high a figure, with annual cost avoidance estimated at $600,000.31 All these figures are based on estimates and, therefore, imprecise, but it is clear even using the most conservative model that the cost to employ a clinical pharmacist is justified.
The final value of cost savings is likely significantly underestimated relative to non-VA hospitals due to the decision to correct for inflation, using the total market inflation rate rather than the medical sector inflation rate over the same time period. The Lee study values were increased by 25.0% and the Ling study values by 15.2%, to bring them to 2011 amounts. Using the medical inflation rate instead (42.3% and 25.2%, respectively), an additional $378,000 in annual savings would have been realized. The lower CPI inflation rate rather than the higher rate in the medical sector was chosen to make the cost avoidance outcomes more conservative.
The true value of a clinical pharmacist comes from the services they provide to patients. In this pilot, as well as in several others, it has been shown that education is a commonly performed and highly valued task. Education was a service lacking in this ED prior to this intervention due to financial and logistical constraints. It is unclear how much instruction patients receive at the outpatient pharmacy while picking up medications after leaving the ED, but it is likely limited, given the large volumes and long lines often found at the in-house pharmacy. Education has a demonstrated effect on prevention and management of ADEs and was the most interactive of the interventions the pharmacist provided during this study. This type of intervention was most likely the source of increased patient satisfaction that was noted in the postencounter surveys.17,24
Prevention of ADEs, which was a frequent intervention in this pilot, has been noted by many sources to be the single most beneficial task performed by a clinical pharmacist both from financial and risk reduction standpoints.13,21-23 Although not able to assess patient outcomes after this limited pilot, the authors anticipate such an evaluation when a full-time ED pharmacist joins the department.
The Joint Commission recommends that a pharmacist review all medication orders before administration, though there is an exception for the emergency setting.34 The Joint Commission also recommends medication reconciliation at every visit, including those in the ED setting. The addition of a clinical pharmacist would increase compliance with this and other standards and bring ED operations up to the same benchmark as other practice settings.
LIMITATIONS
The most significant limitation of this study was sample size. The volunteered time of the pharmacists in the ED totaled only 30 hours over 2 weeks. In that limited time, however, the pharmacists had more patient interactions than were anticipated. Had the pilot been conducted over a longer period, it is unclear whether this would have been sustained or whether this was a coincidental overestimate of the effect that a full-time pharmacist would have on the department. Likely, it is an underestimate of their potential, as the availability of the pharmacist was novel and likely underused by other providers. Given more time with the ED staff, pharmacists would be more frequently called on for their expertise, because their skills and knowledge set would be better understood. During this pilot, the pharmacist was located in a separate room in the ED where not all ED staff knew they were available for consultation.
The other major limitation of the pilot was the inherent imprecision of cost avoidance estimates. The dollar amounts attributed to the duties fulfilled by the pharmacists relied on 2 studies. The first, by Lee and colleagues, provided cost avoidance estimates of certain pharmacist actions based on a combination of 4 to 5 clinicians’ estimates of risk reduction, combined with their individual location’s costs for hospitalization, laboratory tests, diagnostic procedures, medications, telephone care, clinic visits, and emergency department visits.30 The numbers are based not only on a small number of individual estimations of risk, but also on facility costs that are highly variable. Despite this, the authors believe the estimates are actually on the conservative side, since they do not account for costs of lost productivity and/or litigation.
The current pilot was performed in a different type of setting than the one by Lee. That study was conducted in a similar VAMC setting, but their study data were obtained from other areas of the medical center. Of 600 pharmacist interventions, 250 were in an outpatient clinic, 250 were in an inpatient setting, and 100 were in a nursing home.30 Despite this, the estimates are likely still relevant to this study, given that drugs used in the ED are often a mix of inpatient and outpatient ones, with the same risks to an individual regardless of where they are initiated, changed, or discontinued.
The study by Ling and colleagues was performed in an ED setting more closely matching this study’s setting and was a larger, well powered study. As with the Lee study, it was difficult if not impossible to obtain exact numbers on the expenses each pharmacist recommendation spared the hospital and/or patients.31 Not all drug interactions avoided would have led to symptoms, reevaluation, or hospitalization.35 Not all drug “allergies” avoided are true allergies (as seen dramatically by Raja and colleagues), and thus this action may not have spared any cost at all.36 In the end, however, the estimates provided by both studies are averaged over many patients and thus provided the best numbers available.
Unlike the Lee study, this pilot did not evaluate the medication cost differences between original treatment and the new recommended treatment. Given the small number of patients with whom significant changes were made in this study, evaluating the cost differences between the treatments would likely be insignificant. A larger study, such as Lee, was much more sufficiently powered to evaluate such a figure.30
Of note, in this pilot there were no cases seen in which there was any change in route of delivery, ie, IV to equivalent po treatments. This is typically a large source of cost savings secondary to reduction in equipment and nursing time. The Lada study found 66 such changes among 2,150 pharmacist interventions in the ED.15 The authors hypothesize that had their pilot been conducted over a longer period, significant cost savings would have resulted from similar interventions.
In this pilot, a significant number of patients presented for prescription refills. Veterans often prefer to fill medications at the VA pharmacy because of reduced cost and often bring prescriptions written by private sector physicians. These veterans are required to have a primary care physician assigned within the VA, but until they have their initial intake appointment, they use the ED for these prescriptions. Additionally, veterans from other VA locations presenting as visitors to the area or relocating to the city and not yet assigned to a primary care physician require their medication lists from other location(s) be accessed and reentered into intrafacility computerized ordering systems. Given these particulars of VA operation, the authors’ facility assuredly sees more patients presenting for prescription refill than nongovernment facilities. Thus our savings with this particular task may not be generalizable to settings outside the VA, at least in as high a number of encounters.
CONCLUSIONS
About 37,000 veterans received care at the ED of the Atlanta VAMC in 2011. Given these numbers and the evidence that EDs have some of the highest rates of preventable ADEs of any clinical environment, the presence of a clinical pharmacist in the ED is a necessary intervention, based on safety considerations alone. In addition to providing a needed layer of safety in the vulnerable ED environment, a clinical pharmacist likely provides a cost saving benefit to the ED, as demonstrated by this pilot and other studies. Further, the overwhelmingly positive response to this pilot by the veterans who participated shows that they want and need this service. Adding a clinical pharmacist to the ED is integral to the VA mission of providing patient-centered care. A larger study to obtain a more precise cost savings benefit within the VA system should be considered.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Schumock GT, Butler MG, Meek PD, et al. Evidence of the economic benefit of clinical pharmacy services: 1996-2000. Pharmacotherapy. 2003;23(1):113-132.
2. Pedersen CA, Schneider PJ, Scheckelhoff DJ. ASHP national survey of pharmacy practice in hospital settings: Prescribing and transcribing—2010. Am J Health Syst Pharm. 2011;68(8):669-688.
3. Rudis MI, Attwood RJ. Emergency medicine pharmacy practice. J Pharm Pract. 2011; 4(2):135-145.
4. Clancy CM. Evidence shows cost and patient safety benefits of emergency pharmacists. Am J Med Qual. 2008;23(3):231-233.
5. Fairbanks RJ, Hays DP, Webster DF, et al. Clinical pharmacy services in an emergency department. Am J Health Syst Pharm. 2004;61(9):934-937.
6. Abu-Ramaileh AM, Shane R, Churchill W, Steffenhagen A, Patka J, Rothschild JM. Evaluating and classifying pharmacists’ quality interventions in the emergency department. Am J Health Syst Pharm. 2011;68(23):2271-2275.
7. Committee on the Future of Emergency Care in the United States Health System. Hospital-Based Emergency Care: At the Breaking Point. Washington, DC: National Academies Press; 2006.
8. Szczesiul JM, Fairbanks RJ, Hildebrand JM, Hays DP, Shah MN. Survey of physicians regarding clinical pharmacy services in academic emergency departments. Am J Health Syst Pharm. 2009;66(6):576-579.
9. Pedersen CA, Schneider PJ, Scheckelhoff DJ. ASHP national survey of pharmacy practice in hospital settings: Dispensing and Administration—2005. Am J Health Syst Pharm. 2006;63(4):327-345.
10. Schenkel S. Promoting patient safety and preventing medical error in emergency departments. Acad Emerg Med. 2000;7(11):1204-1222.
11. Chin MH, Wang LC, Jin L, et al. Appropriateness of medication selection for older persons in an urban academic emergency department. Acad Emerg Med. 1999;6(12):1232-1242.
12. Hastings SN, Schmader KE, Sloane RJ, et al. Quality of pharmacotherapy and outcomes for older veterans discharged from the emergency department. J Am Geriatr Soc. 2008;56(5):875-880.
13. Rothschild JM, Churchill W, Erickson A, et al. Medication errors recovered by emergency department pharmacists. Ann Emerg Med. 2010;55(6):513-521.
14. Eppert HD, Reznek AJ; American Society of Health-System Pharmacists. ASHP guidelines on emergency medicine pharmacist services. Am J Health Syst Pharm. 2011;68(23):e81-95.
15. Lada P, Delgado G Jr. Documentation of pharmacists’ interventions in an emergency department and associated cost avoidance. Am J Health Syst Pharm. 2007;64(1):63-68.
16. Cohen V, Jellineck SP, Hatch A, Motov S. Effect of clinical pharmacists on care in the emergency department: A systematic review. Am J Health Syst Pharm. 2009;66(15):1353-1361.
17. Randolph TC. Expansion of pharmacists’ responsibilities in an emergency department. Am J Health Syst Pharm. 2009;66(16):1484-1487.
18. Hayes BD, Donovan JL, Smith BS, Hartman CA. Pharmacist-conducted medication reconciliation in an emergency department. Am J Health Syst Pharm. 2007;64(16):1720-1723.
19. DeWinter S, Spriet I, Indevuyst C, et al. Pharmacist-versus physician-acquired medication history: A prospective study at the emergency department. Qual Saf Health Care. 2010; 19(5):371-375.
20. American Society of Health-System Pharmacists. ASHP statement on pharmacy services to the emergency department. Am J Health Syst Pharm. 2008;65(24):2380-2383.
21. Ernst AA, Weiss SJ, Sullivan A IV, et al. On-site pharmacists in the ED improve medical errors. Am J Emerg Med. 2011;30(5):717-725.
22. Weant KA, Humphries RL, Hite K, Armitstead JA. Effect of emergency medicine pharmacists on medication-error reporting in an emergency department. Am J Health Syst Pharm. 2010;67(21):1851-1855.
23. Brown JN, Barnes CL, Beasley B, et al. Effect of pharmacists on medication errors in an emergency department. Am J Health Syst Pharm. 2008;65(4):330-333.
24. Holland R, Desborough J, Goodyer L, Hall S, Wright D, Loke YK. Does pharmacist-led medication review help to reduce hospital admissions and deaths in older people? A systematic review and meta-analysis. Br J Clin Pharmacol. 2008;65(3):303-316.
25. Marconi GP, Claudius I. Impact of an emergency department pharmacy on medication omission and delay. Pediatr Emerg Care. 2012;28(1):30-33.
26. Jellinek SP, Cohen V, Fancher LB, et al. Pharmacist improves timely administration of medications to boarded patients in the emergency department. J Emerg Nurs. 2010;36(2):105-110.
27. Pantanwala AE, Sanders AB, Thomas MC, et al. A prospective, multicenter study of pharmacist activities resulting in medication error interception in the emergency department. Ann Emerg Med. 2012;59(5)369-373.
28. Fairbanks RJ, Hildebrand JM, Kolstee KE, Schneider SM, Shah MN. Medical and nursing staff highly value clinical pharmacists in the emergency department. Emerg Med J. 2007;24(10):716-718.
29. Randolph TC, Parker A, Meyer L, Zeina R. Effect of pharmacist-managed culture review process on antimicrobial therapy in an emergency department. Am J Health Syst Pharm. 2011; 68(10):916-919.
30. Lee AJ, Boro MS, Knapp KK, Meier JL, Kirman NE. Clinical and economic outcomes of pharmacist recommendations in a veterans affairs medical center. Am J Health Syst Pharm. 2002;59(21):2070-2077.
31. Ling JM, Mike LA, Rubin J, et al. Documentation of pharmacist interventions in the emergency department. Am J Health Syst Pharm. 2005;62(17):1793-1797.
32. U.S. Bureau of Labor Statistics. Database, tools and calculators by subject. CPI inflation calculator. U.S. Bureau of Labor Statistics Website. http://www.bls.gov/data/inflation_calculator.htm. Accessed August 05, 2014.
33. Levy DB. Documentation of clinical and cost-saving pharmacy interventions in the emergency room. Hosp Pharm. 1993;28(7):624-627, 630-634, 653.
34. Uselton JP, Kienle P, Murdaugh LB, eds. Assuring Continuous Compliance With Joint Commission Standards: A Pharmacy Guide. 8th ed. Bethesda, MD: American Society of Health-System Pharmacists; 2010.
35. Pantanwala AE, Hays DP, Sanders AB, Erstad BL. Severity and probability of harm of medication errors intercepted by an emergency pharmacist. Int J Pharm Pract. 2011;19(5):358-362.
36. Raja AS, Lindsell CJ, Bernstein JA, Codispoti CD Moellman JJ. The use of penicillin skin testing to assess the prevalence of penicillin allergy in an emergency department setting. Ann Emerg Med. 2009;54(1):72-77.
Clinical pharmacists have expanded their role over the past few decades in both outpatient and inpatient settings and are now members of an interdisciplinary health care team that includes nutritionists, physical therapists, physicians, and nurses.1,2 The emergency department (ED), however, has lagged behind in the inclusion of pharmacists.3 Despite well documented financial and ED operational benefits of pharmacists and the recommendation of their inclusion by the Institute of Medicine, only about 30% of academic EDs in a 2009 survey employed a pharmacist.4-8 A larger 2005 survey of 510 hospital pharmacy directors revealed that only 3.5% of hospitals sampled (academic and nonacademic) provided clinical pharmacy services in the ED.9
About 3.8 million annual preventable medical errors occur in the ED, giving the ED the highest rate of medication errors among all hospital departments.4 In 2000, Schenkel found that 3% of all inpatient medication errors were initiated in the ED.10 Similarly, Chin and colleagues found that 3.6% of elderly patients were administered an inappropriate medication in the ED with 5.6% receiving an inappropriate prescription at discharge.11
In a 2008 study conducted at the Durham VAMC in North Carolina, Hastings and colleagues found that suboptimal pharmacy was common among elderly veterans discharged from the ED (11%) and that potentially inappropriate medication use was associated with a 32% greater risk of repeat ED visits, hospitalization, or death (P = .10).12 In 2010, Rothschild and colleagues found 7.8 medication errors per 100 ED patients or 2.9 errors per 100 prescribed medications.13 Despite this unacceptably high rate of medication errors, most EDs do not employ pharmacy specialists or have a pharmacist easily available for consultation—options that could not only streamline ED operations, but also reduce patient risk.
The pharmacist role in the ED has changed considerably. In the 1970s, ED pharmacists were used mainly to dispense medicine, maintain inventories, and participate in cardiopulmonary resuscitation.3,14,15 Today, following the guidelines set by the American Society for Health-System Pharmacists, emergency pharmacists have an expanded, more direct role in patient care and evaluation and support of the physicians and other ED staff who work alongside them.4,14,16,17 Pharmacists gather accurate and complete medication histories, review and reconcile medication lists, and screen ED medication orders for errors or anticipated drug interactions.13,18-23 They adjust medication doses on a patient-by-patient basis, accounting for renal and hepatic clearance and closely monitor patients for treatment response. They also provide one-on-one patient education on medication dosing, administration, adverse drug events (ADEs), and interactions, increasing patients’ drug knowledge and adherence.17,24 Pharmacists provide information to patients on vaccinations and medication assistance programs, which is unlikely to be shared by other providers.3,19,20 Pharmacists in the ED reduce medication delays and omissions that occur in admitted patients staying in the ED.25,26
Aside from patient education, clinical pharmacists have an important role in providing education and consultation to ED physicians, midlevel providers, and house staff on topics that include availability of new medications and local antibiotic resistance patterns.14,27-29 Additionally, pharmacists monitor drug supplies and restock medications to avoid shortages during critical moments, offer the ED perspective in hospital formulary reviews, and increase efficiency and throughput in the ED while decreasing costs by evaluating and treating patients who present simply for prescription refills alongside a supervising physician.14
With this in mind, the ED of the Atlanta VAMC in Decatur, Georgia, conducted a pilot study to assess the financial and logistic feasibility of a full-time pharmacist in the ED setting with the hope that a pharmacist would integrate well into the health care team, reducing overall departmental expense and the risk of medication error associated with patient harm and simultaneously improving patient satisfaction and departmental efficiency.
Methodology
The ED of the Atlanta VAMC is part of a tertiary care teaching hospital affiliated with both the Emory and Morehouse schools of medicine. At the time of the pilot, the facility had 128 acute care medical/surgical beds, 12 inpatient palliative care beds, 40 acute care psychiatric beds, 24 medical surgical intensive care unit beds, and 60 inpatient nursing home beds. The ED provides care to > 37,000 veterans annually, and in December 2011 when this study was conducted, 3,195 veterans were seen in the ED.
The ED was divided into the main ED and the urgent care. Patient intake occurred through a centralized triage, and based on acuity, patients were sent to the appropriate setting for treatment. The ED used a 5-tier triage system. Patients with triage levels 1, 2, and 3 were sent to the main ED, and patients with triage levels 4 and 5 were sent to the urgent care.
Pharmacists
Pharmacy services were provided by 5 residency-trained doctors of pharmacy employed by the medical center working as clinical pharmacists with the inpatient medical teams at the time of the pilot. The pilot was conducted over a 2-week period in December 2011, Monday through Friday, for a total of 10 days. The clinical pharmacists divided the days among themselves. Each pharmacist provided services for a total of 2 days, 3 hours per day, from about 3 pm to 6 pm. The pharmacists were given a room previously used as a physician workroom in which to evaluate patients.
Patient Selection
Patients to be seen by the clinical pharmacist were chosen by the triage nurse, the charge nurse, the ED physician, the urgent care provider (physician or midlevel provider), or by the pharmacists. The triage nurse or charge nurse, based on chief medical problem and acuity, chose patients directly out of triage. Only patients with triage acuity level 4 or 5 were taken directly from triage without first seeing a physician or midlevel provider. These patients presented with the chief problem of medication refill or coumadin/International Normalized Ratio check. Once chosen as appropriate for the clinical pharmacist, the charge nurse helped with patient flow, and if the pharmacist was occupied with other patients, the nurse redirected the patient to urgent care.
Additional patients were chosen to see the clinical pharmacist after an evaluation of their initial problem was completed by a physician or midlevel practitioner in the urgent care or main ED. Patients whom the provider felt could benefit from any of the following services were directed to the clinical pharmacist: anticoagulation consult, diabetic education, pharmacokinetic consult, medication history, medication reconciliation, formulary management, medication refills, therapeutic interchange, screening for drug interactions, allergy review, and nonformulary or restricted medications requests. Additionally, the clinical pharmacist reviewed the charts of patients in the main ED whom they were not asked to see. They offered assistance when needed in all the aforementioned areas and for order clarification, assuring IV compatibility, reporting medication errors and ADEs, promotion of safe medical practices, and elimination of duplicate/redundant medications.
Data Collection
The pharmacists developed a log to record their activities. The log included the date and time of the intervention, number of minutes spent with the patient, the reason for intervention, and recommendations, if applicable. They categorized their interventions into 16 categories: anticoagulation, pharmacokinetics, drug information, order clarification, medication reconciliation, therapeutic interchange, formulary management, medication history, IV compatibility, screening for drug interactions, patient education, allergy documentation, promotion of safe medical practices, reporting of medication error/ADEs, nonformulary and restricted medication requests, and prescription refills. Patients could receive more than 1 intervention.
Though not a focus of this pilot, all patients seen by a pharmacist received a postencounter survey seeking their opinion on whether the pharmacist improved the value of their visit.
Review Process
At the conclusion of the pilot, 2 independent reviewers, both physicians, reviewed the logs, and each task was reassigned to 1 of 8 categories. These categories included either medication refills or 1 of 7 other areas that had established cost avoidance estimates from 2 other well accepted studies (Lee and colleagues and Ling and colleagues).30,31 These 7 categories included adjusting dose or frequency of medication, elimination of duplication of therapy, education/information inquiry, formulary management, prevention and management of ADEs, prevention or management of allergies, and therapeutic interchange. If the independent reviewers did not have initial concordance of classification of the intervention, they discussed the intervention and came to an agreement.
Cost Analysis
Cost avoidance estimates for 7 individual interventions were made, using data from Lee and colleagues and Ling and colleagues.30,31 Four of these came from the study by Lee and colleagues: prevent or manage drug allergy, adjust dosage or frequency, prevent or manage ADEs, and eliminate duplication of therapy.30 Lee and colleagues’ “drug interaction” group was not clearly defined, thus this was included with the “prevent or manage ADE” group. Ling and colleagues provided data for the 3 additional groups of interventions that pharmacists performed: education and information inquiry, formulary management, and therapeutic interchange.31
Financial estimates of cost avoidance were adjusted for inflation, using the consumer price index (CPI) of the U.S. Bureau of Labor Statistics.32 The Lee study was conducted in 2002, and estimates for cost avoidance using their model were adjusted to 2011 values using the CPI inflation rate of 25%. The Ling study was conducted in 2005, and estimates for cost avoidance using their model were adjusted for 2011 values using the CPI rate of inflation of 15.2%.32
For the remaining intervention, prescription refill, cost savings was determined by calculating the average times spent by the ED pharmacist on each intervention and then using the difference between hourly physician and pharmacist pay (about $50/h difference based on VA wage tables).
RESULTS
During the 30-hour total time in which a pharmacist was present in the ED, a total of 42 patients were assisted through 71 interventions (Table 1).
Pharmacists provided a diverse range of services to patients in the ED. The most common intervention was education and/or information inquiry. Tasks in this category included patient education about medication dosing, administration, AEs, interactions and warnings, as well as diabetes management. In several instances, education was provided to attending physicians or house staff, though it should be noted that this provider education was not counted as an intervention for this study unless it was associated with a patient (of which there were 3 total instances, eg, instruction on how to choose the proper insulin syringe).
Interventions, when a medication list was screened by the ED pharmacist for interactions or when drug choices were recommended to the physician or midlevel providers, were counted as prevention and management of ADEs. For example, the pharmacist noted a patient with a new diagnosis of gout who was prescribed hydrochlorothiazide; this was brought to the attention of the provider and alternative antihypertensives were suggested. In another instance, a patient was found to be on both ibuprofen and enoxaparin; the treating physician was alerted of this potential interaction. There were 15 such events in total.
Several other interventions arose from the screenings for ADEs, including adjusting dose or frequency of medication (11); therapeutic interchange (5); eliminating duplication of therapy (2); and prevention or management of allergies (1). Cases included hepatic and/or renal dose changes, substituting equivalent medications for better treatment outcome or adherence, or discontinuing 2 or more medications in a patient’s medication profile that were considered duplication.
During the pharmacist screening, one patient who had piperacillin/tazobactam ordered in the ED had a penicillin allergy. This intervention was categorized as prevention and management of an ADE as well as prevention and management of allergies. Interventions not accompanied by the “prevention of ADE” category included those in which the change did not provide a clear risk reduction. For example, one therapeutic interchange was from levofloxacin to moxifloxacin for a better-anticipated therapy. Another was a metformin dose increase, presumably for improved glycemic control.
Prescription refills occurred with the same frequency as prevention of ADEs.15 This intervention led in some cases to switching to pharmaceutical equivalents when a drug prescribed at another facility was not on the formulary. Other drugs that were not on the preferred list but available with nonformulary medication requests were ordered or approved with the assistance of the pharmacist. The pharmacist’s direct involvement significantly reduced the initial contact-to-approval time for these patients.
After tallying the total number of interventions, the potential financial cost savings to the ED were determined (Table 2). As mentioned previously, the Lee and Ling studies provided the categories for classification of 7 pharmacist interactions. The estimated cost avoidance for the 4 applicable groups from the Lee study had inflation-corrected values of $1,486 per adjusted dose or frequency of medication, $205 per elimination of duplication of therapy, $1,374 per prevention or management of ADEs, and $1,721 per prevention or management of allergies.30
The estimated cost avoidance for the 3 applicable groups from the Ling study had inflation-corrected values of $512.38 per education/information inquiry, $174.80 per formulary management, and $174.80 per therapeutic interchange.31 The eighth group, prescription refills, was valued at $12.50 each, using the difference between physician and pharmacist salary for an average of 15 minutes per interaction.
When multiplied by the number of interventions in each of these groups, the total potential cost avoidance in the study period was about $40,136.48. Extrapolated into a yearly amount, that is a $2,782,795.94 potential cost savings for the medical center.
Seventeen of the 42 (40.5%) postencounter surveys from the patients seen by the pharmacists were received. Of these veterans, 100% reported that they were “extremely satisfied” with the treatment they had received during their visit to the ED.
DISCUSSION
There is the potential for significant cost avoidance by adding a single full-time pharmacist to the ED: Annually, more than $2.7 million in potential savings for the medical center. Though surprising, this figure is actually in line with the much larger study by Lada and colleagues in which an estimated $3 million was avoided.15 At the same hospital 12 years earlier, Levy noted about $1 million in cost avoidance (not inflation-adjusted).33 The Ling study, however, did not have as high a figure, with annual cost avoidance estimated at $600,000.31 All these figures are based on estimates and, therefore, imprecise, but it is clear even using the most conservative model that the cost to employ a clinical pharmacist is justified.
The final value of cost savings is likely significantly underestimated relative to non-VA hospitals due to the decision to correct for inflation, using the total market inflation rate rather than the medical sector inflation rate over the same time period. The Lee study values were increased by 25.0% and the Ling study values by 15.2%, to bring them to 2011 amounts. Using the medical inflation rate instead (42.3% and 25.2%, respectively), an additional $378,000 in annual savings would have been realized. The lower CPI inflation rate rather than the higher rate in the medical sector was chosen to make the cost avoidance outcomes more conservative.
The true value of a clinical pharmacist comes from the services they provide to patients. In this pilot, as well as in several others, it has been shown that education is a commonly performed and highly valued task. Education was a service lacking in this ED prior to this intervention due to financial and logistical constraints. It is unclear how much instruction patients receive at the outpatient pharmacy while picking up medications after leaving the ED, but it is likely limited, given the large volumes and long lines often found at the in-house pharmacy. Education has a demonstrated effect on prevention and management of ADEs and was the most interactive of the interventions the pharmacist provided during this study. This type of intervention was most likely the source of increased patient satisfaction that was noted in the postencounter surveys.17,24
Prevention of ADEs, which was a frequent intervention in this pilot, has been noted by many sources to be the single most beneficial task performed by a clinical pharmacist both from financial and risk reduction standpoints.13,21-23 Although not able to assess patient outcomes after this limited pilot, the authors anticipate such an evaluation when a full-time ED pharmacist joins the department.
The Joint Commission recommends that a pharmacist review all medication orders before administration, though there is an exception for the emergency setting.34 The Joint Commission also recommends medication reconciliation at every visit, including those in the ED setting. The addition of a clinical pharmacist would increase compliance with this and other standards and bring ED operations up to the same benchmark as other practice settings.
LIMITATIONS
The most significant limitation of this study was sample size. The volunteered time of the pharmacists in the ED totaled only 30 hours over 2 weeks. In that limited time, however, the pharmacists had more patient interactions than were anticipated. Had the pilot been conducted over a longer period, it is unclear whether this would have been sustained or whether this was a coincidental overestimate of the effect that a full-time pharmacist would have on the department. Likely, it is an underestimate of their potential, as the availability of the pharmacist was novel and likely underused by other providers. Given more time with the ED staff, pharmacists would be more frequently called on for their expertise, because their skills and knowledge set would be better understood. During this pilot, the pharmacist was located in a separate room in the ED where not all ED staff knew they were available for consultation.
The other major limitation of the pilot was the inherent imprecision of cost avoidance estimates. The dollar amounts attributed to the duties fulfilled by the pharmacists relied on 2 studies. The first, by Lee and colleagues, provided cost avoidance estimates of certain pharmacist actions based on a combination of 4 to 5 clinicians’ estimates of risk reduction, combined with their individual location’s costs for hospitalization, laboratory tests, diagnostic procedures, medications, telephone care, clinic visits, and emergency department visits.30 The numbers are based not only on a small number of individual estimations of risk, but also on facility costs that are highly variable. Despite this, the authors believe the estimates are actually on the conservative side, since they do not account for costs of lost productivity and/or litigation.
The current pilot was performed in a different type of setting than the one by Lee. That study was conducted in a similar VAMC setting, but their study data were obtained from other areas of the medical center. Of 600 pharmacist interventions, 250 were in an outpatient clinic, 250 were in an inpatient setting, and 100 were in a nursing home.30 Despite this, the estimates are likely still relevant to this study, given that drugs used in the ED are often a mix of inpatient and outpatient ones, with the same risks to an individual regardless of where they are initiated, changed, or discontinued.
The study by Ling and colleagues was performed in an ED setting more closely matching this study’s setting and was a larger, well powered study. As with the Lee study, it was difficult if not impossible to obtain exact numbers on the expenses each pharmacist recommendation spared the hospital and/or patients.31 Not all drug interactions avoided would have led to symptoms, reevaluation, or hospitalization.35 Not all drug “allergies” avoided are true allergies (as seen dramatically by Raja and colleagues), and thus this action may not have spared any cost at all.36 In the end, however, the estimates provided by both studies are averaged over many patients and thus provided the best numbers available.
Unlike the Lee study, this pilot did not evaluate the medication cost differences between original treatment and the new recommended treatment. Given the small number of patients with whom significant changes were made in this study, evaluating the cost differences between the treatments would likely be insignificant. A larger study, such as Lee, was much more sufficiently powered to evaluate such a figure.30
Of note, in this pilot there were no cases seen in which there was any change in route of delivery, ie, IV to equivalent po treatments. This is typically a large source of cost savings secondary to reduction in equipment and nursing time. The Lada study found 66 such changes among 2,150 pharmacist interventions in the ED.15 The authors hypothesize that had their pilot been conducted over a longer period, significant cost savings would have resulted from similar interventions.
In this pilot, a significant number of patients presented for prescription refills. Veterans often prefer to fill medications at the VA pharmacy because of reduced cost and often bring prescriptions written by private sector physicians. These veterans are required to have a primary care physician assigned within the VA, but until they have their initial intake appointment, they use the ED for these prescriptions. Additionally, veterans from other VA locations presenting as visitors to the area or relocating to the city and not yet assigned to a primary care physician require their medication lists from other location(s) be accessed and reentered into intrafacility computerized ordering systems. Given these particulars of VA operation, the authors’ facility assuredly sees more patients presenting for prescription refill than nongovernment facilities. Thus our savings with this particular task may not be generalizable to settings outside the VA, at least in as high a number of encounters.
CONCLUSIONS
About 37,000 veterans received care at the ED of the Atlanta VAMC in 2011. Given these numbers and the evidence that EDs have some of the highest rates of preventable ADEs of any clinical environment, the presence of a clinical pharmacist in the ED is a necessary intervention, based on safety considerations alone. In addition to providing a needed layer of safety in the vulnerable ED environment, a clinical pharmacist likely provides a cost saving benefit to the ED, as demonstrated by this pilot and other studies. Further, the overwhelmingly positive response to this pilot by the veterans who participated shows that they want and need this service. Adding a clinical pharmacist to the ED is integral to the VA mission of providing patient-centered care. A larger study to obtain a more precise cost savings benefit within the VA system should be considered.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Clinical pharmacists have expanded their role over the past few decades in both outpatient and inpatient settings and are now members of an interdisciplinary health care team that includes nutritionists, physical therapists, physicians, and nurses.1,2 The emergency department (ED), however, has lagged behind in the inclusion of pharmacists.3 Despite well documented financial and ED operational benefits of pharmacists and the recommendation of their inclusion by the Institute of Medicine, only about 30% of academic EDs in a 2009 survey employed a pharmacist.4-8 A larger 2005 survey of 510 hospital pharmacy directors revealed that only 3.5% of hospitals sampled (academic and nonacademic) provided clinical pharmacy services in the ED.9
About 3.8 million annual preventable medical errors occur in the ED, giving the ED the highest rate of medication errors among all hospital departments.4 In 2000, Schenkel found that 3% of all inpatient medication errors were initiated in the ED.10 Similarly, Chin and colleagues found that 3.6% of elderly patients were administered an inappropriate medication in the ED with 5.6% receiving an inappropriate prescription at discharge.11
In a 2008 study conducted at the Durham VAMC in North Carolina, Hastings and colleagues found that suboptimal pharmacy was common among elderly veterans discharged from the ED (11%) and that potentially inappropriate medication use was associated with a 32% greater risk of repeat ED visits, hospitalization, or death (P = .10).12 In 2010, Rothschild and colleagues found 7.8 medication errors per 100 ED patients or 2.9 errors per 100 prescribed medications.13 Despite this unacceptably high rate of medication errors, most EDs do not employ pharmacy specialists or have a pharmacist easily available for consultation—options that could not only streamline ED operations, but also reduce patient risk.
The pharmacist role in the ED has changed considerably. In the 1970s, ED pharmacists were used mainly to dispense medicine, maintain inventories, and participate in cardiopulmonary resuscitation.3,14,15 Today, following the guidelines set by the American Society for Health-System Pharmacists, emergency pharmacists have an expanded, more direct role in patient care and evaluation and support of the physicians and other ED staff who work alongside them.4,14,16,17 Pharmacists gather accurate and complete medication histories, review and reconcile medication lists, and screen ED medication orders for errors or anticipated drug interactions.13,18-23 They adjust medication doses on a patient-by-patient basis, accounting for renal and hepatic clearance and closely monitor patients for treatment response. They also provide one-on-one patient education on medication dosing, administration, adverse drug events (ADEs), and interactions, increasing patients’ drug knowledge and adherence.17,24 Pharmacists provide information to patients on vaccinations and medication assistance programs, which is unlikely to be shared by other providers.3,19,20 Pharmacists in the ED reduce medication delays and omissions that occur in admitted patients staying in the ED.25,26
Aside from patient education, clinical pharmacists have an important role in providing education and consultation to ED physicians, midlevel providers, and house staff on topics that include availability of new medications and local antibiotic resistance patterns.14,27-29 Additionally, pharmacists monitor drug supplies and restock medications to avoid shortages during critical moments, offer the ED perspective in hospital formulary reviews, and increase efficiency and throughput in the ED while decreasing costs by evaluating and treating patients who present simply for prescription refills alongside a supervising physician.14
With this in mind, the ED of the Atlanta VAMC in Decatur, Georgia, conducted a pilot study to assess the financial and logistic feasibility of a full-time pharmacist in the ED setting with the hope that a pharmacist would integrate well into the health care team, reducing overall departmental expense and the risk of medication error associated with patient harm and simultaneously improving patient satisfaction and departmental efficiency.
Methodology
The ED of the Atlanta VAMC is part of a tertiary care teaching hospital affiliated with both the Emory and Morehouse schools of medicine. At the time of the pilot, the facility had 128 acute care medical/surgical beds, 12 inpatient palliative care beds, 40 acute care psychiatric beds, 24 medical surgical intensive care unit beds, and 60 inpatient nursing home beds. The ED provides care to > 37,000 veterans annually, and in December 2011 when this study was conducted, 3,195 veterans were seen in the ED.
The ED was divided into the main ED and the urgent care. Patient intake occurred through a centralized triage, and based on acuity, patients were sent to the appropriate setting for treatment. The ED used a 5-tier triage system. Patients with triage levels 1, 2, and 3 were sent to the main ED, and patients with triage levels 4 and 5 were sent to the urgent care.
Pharmacists
Pharmacy services were provided by 5 residency-trained doctors of pharmacy employed by the medical center working as clinical pharmacists with the inpatient medical teams at the time of the pilot. The pilot was conducted over a 2-week period in December 2011, Monday through Friday, for a total of 10 days. The clinical pharmacists divided the days among themselves. Each pharmacist provided services for a total of 2 days, 3 hours per day, from about 3 pm to 6 pm. The pharmacists were given a room previously used as a physician workroom in which to evaluate patients.
Patient Selection
Patients to be seen by the clinical pharmacist were chosen by the triage nurse, the charge nurse, the ED physician, the urgent care provider (physician or midlevel provider), or by the pharmacists. The triage nurse or charge nurse, based on chief medical problem and acuity, chose patients directly out of triage. Only patients with triage acuity level 4 or 5 were taken directly from triage without first seeing a physician or midlevel provider. These patients presented with the chief problem of medication refill or coumadin/International Normalized Ratio check. Once chosen as appropriate for the clinical pharmacist, the charge nurse helped with patient flow, and if the pharmacist was occupied with other patients, the nurse redirected the patient to urgent care.
Additional patients were chosen to see the clinical pharmacist after an evaluation of their initial problem was completed by a physician or midlevel practitioner in the urgent care or main ED. Patients whom the provider felt could benefit from any of the following services were directed to the clinical pharmacist: anticoagulation consult, diabetic education, pharmacokinetic consult, medication history, medication reconciliation, formulary management, medication refills, therapeutic interchange, screening for drug interactions, allergy review, and nonformulary or restricted medications requests. Additionally, the clinical pharmacist reviewed the charts of patients in the main ED whom they were not asked to see. They offered assistance when needed in all the aforementioned areas and for order clarification, assuring IV compatibility, reporting medication errors and ADEs, promotion of safe medical practices, and elimination of duplicate/redundant medications.
Data Collection
The pharmacists developed a log to record their activities. The log included the date and time of the intervention, number of minutes spent with the patient, the reason for intervention, and recommendations, if applicable. They categorized their interventions into 16 categories: anticoagulation, pharmacokinetics, drug information, order clarification, medication reconciliation, therapeutic interchange, formulary management, medication history, IV compatibility, screening for drug interactions, patient education, allergy documentation, promotion of safe medical practices, reporting of medication error/ADEs, nonformulary and restricted medication requests, and prescription refills. Patients could receive more than 1 intervention.
Though not a focus of this pilot, all patients seen by a pharmacist received a postencounter survey seeking their opinion on whether the pharmacist improved the value of their visit.
Review Process
At the conclusion of the pilot, 2 independent reviewers, both physicians, reviewed the logs, and each task was reassigned to 1 of 8 categories. These categories included either medication refills or 1 of 7 other areas that had established cost avoidance estimates from 2 other well accepted studies (Lee and colleagues and Ling and colleagues).30,31 These 7 categories included adjusting dose or frequency of medication, elimination of duplication of therapy, education/information inquiry, formulary management, prevention and management of ADEs, prevention or management of allergies, and therapeutic interchange. If the independent reviewers did not have initial concordance of classification of the intervention, they discussed the intervention and came to an agreement.
Cost Analysis
Cost avoidance estimates for 7 individual interventions were made, using data from Lee and colleagues and Ling and colleagues.30,31 Four of these came from the study by Lee and colleagues: prevent or manage drug allergy, adjust dosage or frequency, prevent or manage ADEs, and eliminate duplication of therapy.30 Lee and colleagues’ “drug interaction” group was not clearly defined, thus this was included with the “prevent or manage ADE” group. Ling and colleagues provided data for the 3 additional groups of interventions that pharmacists performed: education and information inquiry, formulary management, and therapeutic interchange.31
Financial estimates of cost avoidance were adjusted for inflation, using the consumer price index (CPI) of the U.S. Bureau of Labor Statistics.32 The Lee study was conducted in 2002, and estimates for cost avoidance using their model were adjusted to 2011 values using the CPI inflation rate of 25%. The Ling study was conducted in 2005, and estimates for cost avoidance using their model were adjusted for 2011 values using the CPI rate of inflation of 15.2%.32
For the remaining intervention, prescription refill, cost savings was determined by calculating the average times spent by the ED pharmacist on each intervention and then using the difference between hourly physician and pharmacist pay (about $50/h difference based on VA wage tables).
RESULTS
During the 30-hour total time in which a pharmacist was present in the ED, a total of 42 patients were assisted through 71 interventions (Table 1).
Pharmacists provided a diverse range of services to patients in the ED. The most common intervention was education and/or information inquiry. Tasks in this category included patient education about medication dosing, administration, AEs, interactions and warnings, as well as diabetes management. In several instances, education was provided to attending physicians or house staff, though it should be noted that this provider education was not counted as an intervention for this study unless it was associated with a patient (of which there were 3 total instances, eg, instruction on how to choose the proper insulin syringe).
Interventions, when a medication list was screened by the ED pharmacist for interactions or when drug choices were recommended to the physician or midlevel providers, were counted as prevention and management of ADEs. For example, the pharmacist noted a patient with a new diagnosis of gout who was prescribed hydrochlorothiazide; this was brought to the attention of the provider and alternative antihypertensives were suggested. In another instance, a patient was found to be on both ibuprofen and enoxaparin; the treating physician was alerted of this potential interaction. There were 15 such events in total.
Several other interventions arose from the screenings for ADEs, including adjusting dose or frequency of medication (11); therapeutic interchange (5); eliminating duplication of therapy (2); and prevention or management of allergies (1). Cases included hepatic and/or renal dose changes, substituting equivalent medications for better treatment outcome or adherence, or discontinuing 2 or more medications in a patient’s medication profile that were considered duplication.
During the pharmacist screening, one patient who had piperacillin/tazobactam ordered in the ED had a penicillin allergy. This intervention was categorized as prevention and management of an ADE as well as prevention and management of allergies. Interventions not accompanied by the “prevention of ADE” category included those in which the change did not provide a clear risk reduction. For example, one therapeutic interchange was from levofloxacin to moxifloxacin for a better-anticipated therapy. Another was a metformin dose increase, presumably for improved glycemic control.
Prescription refills occurred with the same frequency as prevention of ADEs.15 This intervention led in some cases to switching to pharmaceutical equivalents when a drug prescribed at another facility was not on the formulary. Other drugs that were not on the preferred list but available with nonformulary medication requests were ordered or approved with the assistance of the pharmacist. The pharmacist’s direct involvement significantly reduced the initial contact-to-approval time for these patients.
After tallying the total number of interventions, the potential financial cost savings to the ED were determined (Table 2). As mentioned previously, the Lee and Ling studies provided the categories for classification of 7 pharmacist interactions. The estimated cost avoidance for the 4 applicable groups from the Lee study had inflation-corrected values of $1,486 per adjusted dose or frequency of medication, $205 per elimination of duplication of therapy, $1,374 per prevention or management of ADEs, and $1,721 per prevention or management of allergies.30
The estimated cost avoidance for the 3 applicable groups from the Ling study had inflation-corrected values of $512.38 per education/information inquiry, $174.80 per formulary management, and $174.80 per therapeutic interchange.31 The eighth group, prescription refills, was valued at $12.50 each, using the difference between physician and pharmacist salary for an average of 15 minutes per interaction.
When multiplied by the number of interventions in each of these groups, the total potential cost avoidance in the study period was about $40,136.48. Extrapolated into a yearly amount, that is a $2,782,795.94 potential cost savings for the medical center.
Seventeen of the 42 (40.5%) postencounter surveys from the patients seen by the pharmacists were received. Of these veterans, 100% reported that they were “extremely satisfied” with the treatment they had received during their visit to the ED.
DISCUSSION
There is the potential for significant cost avoidance by adding a single full-time pharmacist to the ED: Annually, more than $2.7 million in potential savings for the medical center. Though surprising, this figure is actually in line with the much larger study by Lada and colleagues in which an estimated $3 million was avoided.15 At the same hospital 12 years earlier, Levy noted about $1 million in cost avoidance (not inflation-adjusted).33 The Ling study, however, did not have as high a figure, with annual cost avoidance estimated at $600,000.31 All these figures are based on estimates and, therefore, imprecise, but it is clear even using the most conservative model that the cost to employ a clinical pharmacist is justified.
The final value of cost savings is likely significantly underestimated relative to non-VA hospitals due to the decision to correct for inflation, using the total market inflation rate rather than the medical sector inflation rate over the same time period. The Lee study values were increased by 25.0% and the Ling study values by 15.2%, to bring them to 2011 amounts. Using the medical inflation rate instead (42.3% and 25.2%, respectively), an additional $378,000 in annual savings would have been realized. The lower CPI inflation rate rather than the higher rate in the medical sector was chosen to make the cost avoidance outcomes more conservative.
The true value of a clinical pharmacist comes from the services they provide to patients. In this pilot, as well as in several others, it has been shown that education is a commonly performed and highly valued task. Education was a service lacking in this ED prior to this intervention due to financial and logistical constraints. It is unclear how much instruction patients receive at the outpatient pharmacy while picking up medications after leaving the ED, but it is likely limited, given the large volumes and long lines often found at the in-house pharmacy. Education has a demonstrated effect on prevention and management of ADEs and was the most interactive of the interventions the pharmacist provided during this study. This type of intervention was most likely the source of increased patient satisfaction that was noted in the postencounter surveys.17,24
Prevention of ADEs, which was a frequent intervention in this pilot, has been noted by many sources to be the single most beneficial task performed by a clinical pharmacist both from financial and risk reduction standpoints.13,21-23 Although not able to assess patient outcomes after this limited pilot, the authors anticipate such an evaluation when a full-time ED pharmacist joins the department.
The Joint Commission recommends that a pharmacist review all medication orders before administration, though there is an exception for the emergency setting.34 The Joint Commission also recommends medication reconciliation at every visit, including those in the ED setting. The addition of a clinical pharmacist would increase compliance with this and other standards and bring ED operations up to the same benchmark as other practice settings.
LIMITATIONS
The most significant limitation of this study was sample size. The volunteered time of the pharmacists in the ED totaled only 30 hours over 2 weeks. In that limited time, however, the pharmacists had more patient interactions than were anticipated. Had the pilot been conducted over a longer period, it is unclear whether this would have been sustained or whether this was a coincidental overestimate of the effect that a full-time pharmacist would have on the department. Likely, it is an underestimate of their potential, as the availability of the pharmacist was novel and likely underused by other providers. Given more time with the ED staff, pharmacists would be more frequently called on for their expertise, because their skills and knowledge set would be better understood. During this pilot, the pharmacist was located in a separate room in the ED where not all ED staff knew they were available for consultation.
The other major limitation of the pilot was the inherent imprecision of cost avoidance estimates. The dollar amounts attributed to the duties fulfilled by the pharmacists relied on 2 studies. The first, by Lee and colleagues, provided cost avoidance estimates of certain pharmacist actions based on a combination of 4 to 5 clinicians’ estimates of risk reduction, combined with their individual location’s costs for hospitalization, laboratory tests, diagnostic procedures, medications, telephone care, clinic visits, and emergency department visits.30 The numbers are based not only on a small number of individual estimations of risk, but also on facility costs that are highly variable. Despite this, the authors believe the estimates are actually on the conservative side, since they do not account for costs of lost productivity and/or litigation.
The current pilot was performed in a different type of setting than the one by Lee. That study was conducted in a similar VAMC setting, but their study data were obtained from other areas of the medical center. Of 600 pharmacist interventions, 250 were in an outpatient clinic, 250 were in an inpatient setting, and 100 were in a nursing home.30 Despite this, the estimates are likely still relevant to this study, given that drugs used in the ED are often a mix of inpatient and outpatient ones, with the same risks to an individual regardless of where they are initiated, changed, or discontinued.
The study by Ling and colleagues was performed in an ED setting more closely matching this study’s setting and was a larger, well powered study. As with the Lee study, it was difficult if not impossible to obtain exact numbers on the expenses each pharmacist recommendation spared the hospital and/or patients.31 Not all drug interactions avoided would have led to symptoms, reevaluation, or hospitalization.35 Not all drug “allergies” avoided are true allergies (as seen dramatically by Raja and colleagues), and thus this action may not have spared any cost at all.36 In the end, however, the estimates provided by both studies are averaged over many patients and thus provided the best numbers available.
Unlike the Lee study, this pilot did not evaluate the medication cost differences between original treatment and the new recommended treatment. Given the small number of patients with whom significant changes were made in this study, evaluating the cost differences between the treatments would likely be insignificant. A larger study, such as Lee, was much more sufficiently powered to evaluate such a figure.30
Of note, in this pilot there were no cases seen in which there was any change in route of delivery, ie, IV to equivalent po treatments. This is typically a large source of cost savings secondary to reduction in equipment and nursing time. The Lada study found 66 such changes among 2,150 pharmacist interventions in the ED.15 The authors hypothesize that had their pilot been conducted over a longer period, significant cost savings would have resulted from similar interventions.
In this pilot, a significant number of patients presented for prescription refills. Veterans often prefer to fill medications at the VA pharmacy because of reduced cost and often bring prescriptions written by private sector physicians. These veterans are required to have a primary care physician assigned within the VA, but until they have their initial intake appointment, they use the ED for these prescriptions. Additionally, veterans from other VA locations presenting as visitors to the area or relocating to the city and not yet assigned to a primary care physician require their medication lists from other location(s) be accessed and reentered into intrafacility computerized ordering systems. Given these particulars of VA operation, the authors’ facility assuredly sees more patients presenting for prescription refill than nongovernment facilities. Thus our savings with this particular task may not be generalizable to settings outside the VA, at least in as high a number of encounters.
CONCLUSIONS
About 37,000 veterans received care at the ED of the Atlanta VAMC in 2011. Given these numbers and the evidence that EDs have some of the highest rates of preventable ADEs of any clinical environment, the presence of a clinical pharmacist in the ED is a necessary intervention, based on safety considerations alone. In addition to providing a needed layer of safety in the vulnerable ED environment, a clinical pharmacist likely provides a cost saving benefit to the ED, as demonstrated by this pilot and other studies. Further, the overwhelmingly positive response to this pilot by the veterans who participated shows that they want and need this service. Adding a clinical pharmacist to the ED is integral to the VA mission of providing patient-centered care. A larger study to obtain a more precise cost savings benefit within the VA system should be considered.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Schumock GT, Butler MG, Meek PD, et al. Evidence of the economic benefit of clinical pharmacy services: 1996-2000. Pharmacotherapy. 2003;23(1):113-132.
2. Pedersen CA, Schneider PJ, Scheckelhoff DJ. ASHP national survey of pharmacy practice in hospital settings: Prescribing and transcribing—2010. Am J Health Syst Pharm. 2011;68(8):669-688.
3. Rudis MI, Attwood RJ. Emergency medicine pharmacy practice. J Pharm Pract. 2011; 4(2):135-145.
4. Clancy CM. Evidence shows cost and patient safety benefits of emergency pharmacists. Am J Med Qual. 2008;23(3):231-233.
5. Fairbanks RJ, Hays DP, Webster DF, et al. Clinical pharmacy services in an emergency department. Am J Health Syst Pharm. 2004;61(9):934-937.
6. Abu-Ramaileh AM, Shane R, Churchill W, Steffenhagen A, Patka J, Rothschild JM. Evaluating and classifying pharmacists’ quality interventions in the emergency department. Am J Health Syst Pharm. 2011;68(23):2271-2275.
7. Committee on the Future of Emergency Care in the United States Health System. Hospital-Based Emergency Care: At the Breaking Point. Washington, DC: National Academies Press; 2006.
8. Szczesiul JM, Fairbanks RJ, Hildebrand JM, Hays DP, Shah MN. Survey of physicians regarding clinical pharmacy services in academic emergency departments. Am J Health Syst Pharm. 2009;66(6):576-579.
9. Pedersen CA, Schneider PJ, Scheckelhoff DJ. ASHP national survey of pharmacy practice in hospital settings: Dispensing and Administration—2005. Am J Health Syst Pharm. 2006;63(4):327-345.
10. Schenkel S. Promoting patient safety and preventing medical error in emergency departments. Acad Emerg Med. 2000;7(11):1204-1222.
11. Chin MH, Wang LC, Jin L, et al. Appropriateness of medication selection for older persons in an urban academic emergency department. Acad Emerg Med. 1999;6(12):1232-1242.
12. Hastings SN, Schmader KE, Sloane RJ, et al. Quality of pharmacotherapy and outcomes for older veterans discharged from the emergency department. J Am Geriatr Soc. 2008;56(5):875-880.
13. Rothschild JM, Churchill W, Erickson A, et al. Medication errors recovered by emergency department pharmacists. Ann Emerg Med. 2010;55(6):513-521.
14. Eppert HD, Reznek AJ; American Society of Health-System Pharmacists. ASHP guidelines on emergency medicine pharmacist services. Am J Health Syst Pharm. 2011;68(23):e81-95.
15. Lada P, Delgado G Jr. Documentation of pharmacists’ interventions in an emergency department and associated cost avoidance. Am J Health Syst Pharm. 2007;64(1):63-68.
16. Cohen V, Jellineck SP, Hatch A, Motov S. Effect of clinical pharmacists on care in the emergency department: A systematic review. Am J Health Syst Pharm. 2009;66(15):1353-1361.
17. Randolph TC. Expansion of pharmacists’ responsibilities in an emergency department. Am J Health Syst Pharm. 2009;66(16):1484-1487.
18. Hayes BD, Donovan JL, Smith BS, Hartman CA. Pharmacist-conducted medication reconciliation in an emergency department. Am J Health Syst Pharm. 2007;64(16):1720-1723.
19. DeWinter S, Spriet I, Indevuyst C, et al. Pharmacist-versus physician-acquired medication history: A prospective study at the emergency department. Qual Saf Health Care. 2010; 19(5):371-375.
20. American Society of Health-System Pharmacists. ASHP statement on pharmacy services to the emergency department. Am J Health Syst Pharm. 2008;65(24):2380-2383.
21. Ernst AA, Weiss SJ, Sullivan A IV, et al. On-site pharmacists in the ED improve medical errors. Am J Emerg Med. 2011;30(5):717-725.
22. Weant KA, Humphries RL, Hite K, Armitstead JA. Effect of emergency medicine pharmacists on medication-error reporting in an emergency department. Am J Health Syst Pharm. 2010;67(21):1851-1855.
23. Brown JN, Barnes CL, Beasley B, et al. Effect of pharmacists on medication errors in an emergency department. Am J Health Syst Pharm. 2008;65(4):330-333.
24. Holland R, Desborough J, Goodyer L, Hall S, Wright D, Loke YK. Does pharmacist-led medication review help to reduce hospital admissions and deaths in older people? A systematic review and meta-analysis. Br J Clin Pharmacol. 2008;65(3):303-316.
25. Marconi GP, Claudius I. Impact of an emergency department pharmacy on medication omission and delay. Pediatr Emerg Care. 2012;28(1):30-33.
26. Jellinek SP, Cohen V, Fancher LB, et al. Pharmacist improves timely administration of medications to boarded patients in the emergency department. J Emerg Nurs. 2010;36(2):105-110.
27. Pantanwala AE, Sanders AB, Thomas MC, et al. A prospective, multicenter study of pharmacist activities resulting in medication error interception in the emergency department. Ann Emerg Med. 2012;59(5)369-373.
28. Fairbanks RJ, Hildebrand JM, Kolstee KE, Schneider SM, Shah MN. Medical and nursing staff highly value clinical pharmacists in the emergency department. Emerg Med J. 2007;24(10):716-718.
29. Randolph TC, Parker A, Meyer L, Zeina R. Effect of pharmacist-managed culture review process on antimicrobial therapy in an emergency department. Am J Health Syst Pharm. 2011; 68(10):916-919.
30. Lee AJ, Boro MS, Knapp KK, Meier JL, Kirman NE. Clinical and economic outcomes of pharmacist recommendations in a veterans affairs medical center. Am J Health Syst Pharm. 2002;59(21):2070-2077.
31. Ling JM, Mike LA, Rubin J, et al. Documentation of pharmacist interventions in the emergency department. Am J Health Syst Pharm. 2005;62(17):1793-1797.
32. U.S. Bureau of Labor Statistics. Database, tools and calculators by subject. CPI inflation calculator. U.S. Bureau of Labor Statistics Website. http://www.bls.gov/data/inflation_calculator.htm. Accessed August 05, 2014.
33. Levy DB. Documentation of clinical and cost-saving pharmacy interventions in the emergency room. Hosp Pharm. 1993;28(7):624-627, 630-634, 653.
34. Uselton JP, Kienle P, Murdaugh LB, eds. Assuring Continuous Compliance With Joint Commission Standards: A Pharmacy Guide. 8th ed. Bethesda, MD: American Society of Health-System Pharmacists; 2010.
35. Pantanwala AE, Hays DP, Sanders AB, Erstad BL. Severity and probability of harm of medication errors intercepted by an emergency pharmacist. Int J Pharm Pract. 2011;19(5):358-362.
36. Raja AS, Lindsell CJ, Bernstein JA, Codispoti CD Moellman JJ. The use of penicillin skin testing to assess the prevalence of penicillin allergy in an emergency department setting. Ann Emerg Med. 2009;54(1):72-77.
1. Schumock GT, Butler MG, Meek PD, et al. Evidence of the economic benefit of clinical pharmacy services: 1996-2000. Pharmacotherapy. 2003;23(1):113-132.
2. Pedersen CA, Schneider PJ, Scheckelhoff DJ. ASHP national survey of pharmacy practice in hospital settings: Prescribing and transcribing—2010. Am J Health Syst Pharm. 2011;68(8):669-688.
3. Rudis MI, Attwood RJ. Emergency medicine pharmacy practice. J Pharm Pract. 2011; 4(2):135-145.
4. Clancy CM. Evidence shows cost and patient safety benefits of emergency pharmacists. Am J Med Qual. 2008;23(3):231-233.
5. Fairbanks RJ, Hays DP, Webster DF, et al. Clinical pharmacy services in an emergency department. Am J Health Syst Pharm. 2004;61(9):934-937.
6. Abu-Ramaileh AM, Shane R, Churchill W, Steffenhagen A, Patka J, Rothschild JM. Evaluating and classifying pharmacists’ quality interventions in the emergency department. Am J Health Syst Pharm. 2011;68(23):2271-2275.
7. Committee on the Future of Emergency Care in the United States Health System. Hospital-Based Emergency Care: At the Breaking Point. Washington, DC: National Academies Press; 2006.
8. Szczesiul JM, Fairbanks RJ, Hildebrand JM, Hays DP, Shah MN. Survey of physicians regarding clinical pharmacy services in academic emergency departments. Am J Health Syst Pharm. 2009;66(6):576-579.
9. Pedersen CA, Schneider PJ, Scheckelhoff DJ. ASHP national survey of pharmacy practice in hospital settings: Dispensing and Administration—2005. Am J Health Syst Pharm. 2006;63(4):327-345.
10. Schenkel S. Promoting patient safety and preventing medical error in emergency departments. Acad Emerg Med. 2000;7(11):1204-1222.
11. Chin MH, Wang LC, Jin L, et al. Appropriateness of medication selection for older persons in an urban academic emergency department. Acad Emerg Med. 1999;6(12):1232-1242.
12. Hastings SN, Schmader KE, Sloane RJ, et al. Quality of pharmacotherapy and outcomes for older veterans discharged from the emergency department. J Am Geriatr Soc. 2008;56(5):875-880.
13. Rothschild JM, Churchill W, Erickson A, et al. Medication errors recovered by emergency department pharmacists. Ann Emerg Med. 2010;55(6):513-521.
14. Eppert HD, Reznek AJ; American Society of Health-System Pharmacists. ASHP guidelines on emergency medicine pharmacist services. Am J Health Syst Pharm. 2011;68(23):e81-95.
15. Lada P, Delgado G Jr. Documentation of pharmacists’ interventions in an emergency department and associated cost avoidance. Am J Health Syst Pharm. 2007;64(1):63-68.
16. Cohen V, Jellineck SP, Hatch A, Motov S. Effect of clinical pharmacists on care in the emergency department: A systematic review. Am J Health Syst Pharm. 2009;66(15):1353-1361.
17. Randolph TC. Expansion of pharmacists’ responsibilities in an emergency department. Am J Health Syst Pharm. 2009;66(16):1484-1487.
18. Hayes BD, Donovan JL, Smith BS, Hartman CA. Pharmacist-conducted medication reconciliation in an emergency department. Am J Health Syst Pharm. 2007;64(16):1720-1723.
19. DeWinter S, Spriet I, Indevuyst C, et al. Pharmacist-versus physician-acquired medication history: A prospective study at the emergency department. Qual Saf Health Care. 2010; 19(5):371-375.
20. American Society of Health-System Pharmacists. ASHP statement on pharmacy services to the emergency department. Am J Health Syst Pharm. 2008;65(24):2380-2383.
21. Ernst AA, Weiss SJ, Sullivan A IV, et al. On-site pharmacists in the ED improve medical errors. Am J Emerg Med. 2011;30(5):717-725.
22. Weant KA, Humphries RL, Hite K, Armitstead JA. Effect of emergency medicine pharmacists on medication-error reporting in an emergency department. Am J Health Syst Pharm. 2010;67(21):1851-1855.
23. Brown JN, Barnes CL, Beasley B, et al. Effect of pharmacists on medication errors in an emergency department. Am J Health Syst Pharm. 2008;65(4):330-333.
24. Holland R, Desborough J, Goodyer L, Hall S, Wright D, Loke YK. Does pharmacist-led medication review help to reduce hospital admissions and deaths in older people? A systematic review and meta-analysis. Br J Clin Pharmacol. 2008;65(3):303-316.
25. Marconi GP, Claudius I. Impact of an emergency department pharmacy on medication omission and delay. Pediatr Emerg Care. 2012;28(1):30-33.
26. Jellinek SP, Cohen V, Fancher LB, et al. Pharmacist improves timely administration of medications to boarded patients in the emergency department. J Emerg Nurs. 2010;36(2):105-110.
27. Pantanwala AE, Sanders AB, Thomas MC, et al. A prospective, multicenter study of pharmacist activities resulting in medication error interception in the emergency department. Ann Emerg Med. 2012;59(5)369-373.
28. Fairbanks RJ, Hildebrand JM, Kolstee KE, Schneider SM, Shah MN. Medical and nursing staff highly value clinical pharmacists in the emergency department. Emerg Med J. 2007;24(10):716-718.
29. Randolph TC, Parker A, Meyer L, Zeina R. Effect of pharmacist-managed culture review process on antimicrobial therapy in an emergency department. Am J Health Syst Pharm. 2011; 68(10):916-919.
30. Lee AJ, Boro MS, Knapp KK, Meier JL, Kirman NE. Clinical and economic outcomes of pharmacist recommendations in a veterans affairs medical center. Am J Health Syst Pharm. 2002;59(21):2070-2077.
31. Ling JM, Mike LA, Rubin J, et al. Documentation of pharmacist interventions in the emergency department. Am J Health Syst Pharm. 2005;62(17):1793-1797.
32. U.S. Bureau of Labor Statistics. Database, tools and calculators by subject. CPI inflation calculator. U.S. Bureau of Labor Statistics Website. http://www.bls.gov/data/inflation_calculator.htm. Accessed August 05, 2014.
33. Levy DB. Documentation of clinical and cost-saving pharmacy interventions in the emergency room. Hosp Pharm. 1993;28(7):624-627, 630-634, 653.
34. Uselton JP, Kienle P, Murdaugh LB, eds. Assuring Continuous Compliance With Joint Commission Standards: A Pharmacy Guide. 8th ed. Bethesda, MD: American Society of Health-System Pharmacists; 2010.
35. Pantanwala AE, Hays DP, Sanders AB, Erstad BL. Severity and probability of harm of medication errors intercepted by an emergency pharmacist. Int J Pharm Pract. 2011;19(5):358-362.
36. Raja AS, Lindsell CJ, Bernstein JA, Codispoti CD Moellman JJ. The use of penicillin skin testing to assess the prevalence of penicillin allergy in an emergency department setting. Ann Emerg Med. 2009;54(1):72-77.
Genetic Heart Failure in an Active-Duty Soldier
Left ventricular noncompaction (LVNC) is a rare disorder that is variably classified as a primary genetic cardiomyopathy (CM) by the American Heart Association.1 It is mostly believed to be a congenital abnormality, characterized by the arrest of the typical embryonic myocardial maturation process with the subsequent retention of the trabecular myocardial structure, which defines the early embryonic heart.2
During very early embryonic development, the left ventricular (LV)myocardium is composed of a loose network of fibers separated by deep recesses, which link it with the LV cavity. At 8 weeks of prenatal development, gradual compaction of these fibers occurs, and LVNC is thought to result from the arrest of this normal process.2,3 Significant variability in myocardial involvement exists, ranging from panventricular to isolated apical involvement, likely related to time of arrest of this maturation process.4 The decreased contractile capability and inadequate epicardial coronary system communication of this trabecular endocardium is thought to lead to the clinical manifestations of LVNC.1-7
This report describes the case of a 45-year-old male soldier who presented with a unique case of heart failure, diagnosed via cardiac magnetic resonance imaging (MRI).
Case Study
The patient presented to the San Antonio Military Medical Center emergency department in mid-2011 with increasing dyspnea for several weeks. He also reported significant lower-extremity and scrotal edema. Although the patient had been previously healthy, his recent medical history was remarkable for a severe combat injury suffered while on duty with the U.S. Army in Afghanistan: He was involved in an explosion from an improvised explosive device in August 2009. He was medically evacuated to the U.S., where he required multiple hospitalizations and surgeries. Prior to his current presentation, the patient had been briefly hospitalized for hospital-acquired pneumonia. During this hospitalization, he first noted abnormal swelling of his legs, a finding that was initially attributed to the large sodium load he had received with his IV antibiotics.
DIAGNOSIS
The patient’s vital signs on presentation were notable for 100/83 mm Hg blood pressure, 103 beats per minute (bpm) heart rate, and 18/min respiratory rate with a saturation of 100% on 4 liters of oxygen by nasal cannula. He was conversant but tachypneic and had to pause frequently to catch his breath. His neck veins were notably distended with jugular venous pulsations visible to the angle of the jaw with the patient at 30 degrees. His heart sounds were normal without an S3, but his lungs were notable for bilateral crackles over the lower- to mid-lung fields. He had profound bilateral upper and lower extremity and scrotal pitting edema. He had no lymphadenopathy or skin rashes.
On presentation, the patient’s laboratory results were remarkable for a 444 pg/mL brain natriuretic peptide. A chest X-ray revealed bilateral basilar opacities. An electrocardiogram showed normal sinus rhythm (70 bpm), with normal axis and poor R-wave progression across the precordium. An echocardiogram was performed and notable for a moderately dilated left ventricle with severely depressed systolic function of 10% to 15%, and elevated pulmonary artery pressures. Subsequently, the patient was referred for a coronary angiography, which showed no evidence of coronary atherosclerosis. A cardiac MRI was then performed to evaluate for nonischemic CM, which revealed prominent trabeculations in both ventricles, but most notably in the left ventricle, consistent with a diagnosis of LVNC.
The patient was treated with diuretics, beta-blockers, and an angiotensin-converting enzyme (ACE) inhibitor with improvement in his heart failure symptoms. He was started on systemic anticoagulation with warfarin for his severely depressed LV function. His hospital course was complicated by frequent, nonsustained ventricular tachycardia (VT), and he was referred to the electrophysiology service for implantation of an automated intracardiac cardioverter/defibrillator (AICD) for primary prevention of sudden cardiac death. His clinical course was otherwise unremarkable, and he was discharged after 8 days with complete resolution of his symptoms.
Discussion
The clinical presentation of LVNC is typically due to complications of ventricular dysfunction, including heart failure, arrhythmias, and cardioembolic events. Retrospective studies have shown much variability in the frequency of these complications, likely due to selection bias in earlier studies. These earlier studies had suggested a frequency of heart failure > 50%, but recent studies have shown a more modest frequency of 30% to 35% of affected patients.
Even greater variance has been found in the frequency of arrhythmias, but most studies have shown a frequency of at least 20% for VT. Poor blood flow in the deep intertrabecular recesses in patients with LVNC is additionally thought to lead to a predisposition for mural thrombus formation with an elevated frequency of systemic embolic events, ranging from 5% to 20% among previous studies.1-4,6,8
Much debate remains regarding the genetic association of this condition. The unique character of the resulting myocardium suggests a distinct CM, but the significant genetic heterogeneity with sarcomere protein gene mutations associated with several other CMs, including hypertrophic and dilated CM, suggests that LVNC may simply exist on a phenotypic continuum with these other conditions.4 Inheritance shows additional similarities to these other known CMs with autosomal-
dominant and X-linked modes of transmission shown with familial forms in about 25% of patients.5,7 This has led many to believe that screening of first-degree relatives of clinically affected patients may be appropriate.
The prevalence of LVNC in adults referred for echocardiography is about 0.014% to 1.3%.5 A recent increase in the rate of recognition has raised concerns of possible overdiagnosis, with attempts now made to develop specific imaging diagnostic criteria. Diagnosis of LVNC is most commonly suspected (but can be missed) on echocardiography using 2-D and color Doppler imaging modalities. Echocardiographic findings supporting the diagnosis of LVNC suggested by Oechslin and colleague include:
• Presence of multiple trabeculations, particularly in the LV apex and free wall;
• Multiple deep trabeculation recesses in communication with the LV cavity, usually seen on color Doppler imaging;
• A 2-layered structure of the endomyocardium with ratio of end systolic, noncompacted endocardial layer to compacted epicardial layer > 2 in adults; and
• Absence of other congenital or acquired heart disease, particularly those causing LV outflow obstruction.8
Another proposed standardized method for identifying LVNC via echocardiography by Chin and colleagues focuses on trabeculae at the LV apex on the parasternal short axis and apical views.2,3 LVNC is defined by a ratio of X/Y of ≤ 0.5, where X is the distance from the epicardial surface to the trough of the trabecular recess, and Y is the distance from the epicardial surface to the peak of the trabeculations.
Cardiac MRI is now a more common mode of imaging used for diagnosis of LVNC and often has better imaging characteristics than those of echocardiography. Using a ratio of noncompacted to compacted CM in diastole > 2.3 is suggestive of LVNC with sensitivity and specificity of 86% and 99%.9
The management of LVNC focuses primarily on treatment of complications, including heart failure, rhythm disturbances, and thromboembolic events. Treatment of heart failure is typically the same as for other CMs and includes medical therapy with salt restriction, diuretics, beta-blockers, and ACE inhibitors. In addition, exercise training, as tolerated, is beneficial to improve clinical status.3,10 Electrophysiology studies are often performed in these patients, and implantation of an AICD is typically done in cases of documented, sustained VT, presyncope with inducible VT or severally depressed ejection fraction of < 35%.4,10 Deep intertrabecular recesses and impaired blood flow increase the risk of thrombus formation. Hence, anticoagulation with warfarin (international normalized ratio target 2.3) for those with an impaired LV ejection fraction (< 40%) should be considered for the prevention of cardioembolic events.3,4,6,10
Summary
An active-duty solider with a history of battlefield trauma and multiple hospitalizations was admitted for symptomatic heart failure with cardiac MRI suggestive of LVNC. This condition is a phenotypic result of genetic heterogeneity with significant variability in clinical presentation and a predisposition for heart failure, ventricular arrhythmias, and systemic embolic events. The etiology of this patient’s clinical presentation remains unclear, and additional research is needed to understand whether his recent trauma and multiple hospitalizations played a role in the manifestation of his disease.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Maron BJ, Towbin JA, Thiene G, et al. Contemporary definitions and classification of the cardiomyopathies: An American Heart Association Scientific Statement from the Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups; and Council on Epidemiology and Prevention. Circulation. 2006;113(14):1807-1816.
2. Chin TK, Perloff JK, Williams RG, Jue K, Mohrmann R. Isolated noncompaction of left ventricular myocardium. A study of eight cases. Circulation. 1990;82(2):507-513.
3. Murphy RT, Thaman R, Blanes JG, et al. Natural history and familial characteristics of isolated left ventricular non-compaction. Eur Heart J. 2005;26(2):187-192.
4. Oechslin E, Jenni R. Left ventricular non-compaction revisited: A distinct phenotype with genetic heterogeneity? Eur Heart J. 2011;32(12):1446-1456.
5. Elliott P, Andersson B, Arbustini E, et al. Classification of the cardiomyopathies: A position statement from the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2008;29(2):270-276.
6. Oechslin EN, Attenhofer Jost CH, Rojas JR, Kaufmann PA, Jenni R. Longterm follow-up of 34 adults with isolated left ventricular noncompaction: A distinct cardiomyopathy with poor prognosis. J Am Coll Cardiol. 2000;36(2):493-500.
7. Spirito P, Autore C. Apical hypertrophic cardiomyopathy or left ventricular non-compaction? A difficult differential diagnosis [editorial]. Eur Heart J. 2007;28(16):1923-1924.
8. Oechslin E, Jenni R. Non-compaction of the left ventricular myocardium—From clinical observation to the discovery of a new disease. Eur Cardiol Review. 2005;1(1):23-24.
9. Petersen SE, Selvanayagam JB, Wiesmann F, et al. Left ventricular non-compaction: Insights from cardiovascular magnetic resonance imaging. J Am Coll Cardiol. 2005;46(1):101-105.
10. Hunt SA, Abraham WT, Chin MH, et al; American College of Cardiology Foundation; American Heart Association. 2009 Focused update incorporated into the ACC/AHA 2005 Guidelines for the Diagnosis and Management of Heart Failure in Adults: A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines Developed in Collaboration With the International Society for Heart and Lung Transplantation. Circulation. 2009;53(15):e1-e90.
Left ventricular noncompaction (LVNC) is a rare disorder that is variably classified as a primary genetic cardiomyopathy (CM) by the American Heart Association.1 It is mostly believed to be a congenital abnormality, characterized by the arrest of the typical embryonic myocardial maturation process with the subsequent retention of the trabecular myocardial structure, which defines the early embryonic heart.2
During very early embryonic development, the left ventricular (LV)myocardium is composed of a loose network of fibers separated by deep recesses, which link it with the LV cavity. At 8 weeks of prenatal development, gradual compaction of these fibers occurs, and LVNC is thought to result from the arrest of this normal process.2,3 Significant variability in myocardial involvement exists, ranging from panventricular to isolated apical involvement, likely related to time of arrest of this maturation process.4 The decreased contractile capability and inadequate epicardial coronary system communication of this trabecular endocardium is thought to lead to the clinical manifestations of LVNC.1-7
This report describes the case of a 45-year-old male soldier who presented with a unique case of heart failure, diagnosed via cardiac magnetic resonance imaging (MRI).
Case Study
The patient presented to the San Antonio Military Medical Center emergency department in mid-2011 with increasing dyspnea for several weeks. He also reported significant lower-extremity and scrotal edema. Although the patient had been previously healthy, his recent medical history was remarkable for a severe combat injury suffered while on duty with the U.S. Army in Afghanistan: He was involved in an explosion from an improvised explosive device in August 2009. He was medically evacuated to the U.S., where he required multiple hospitalizations and surgeries. Prior to his current presentation, the patient had been briefly hospitalized for hospital-acquired pneumonia. During this hospitalization, he first noted abnormal swelling of his legs, a finding that was initially attributed to the large sodium load he had received with his IV antibiotics.
DIAGNOSIS
The patient’s vital signs on presentation were notable for 100/83 mm Hg blood pressure, 103 beats per minute (bpm) heart rate, and 18/min respiratory rate with a saturation of 100% on 4 liters of oxygen by nasal cannula. He was conversant but tachypneic and had to pause frequently to catch his breath. His neck veins were notably distended with jugular venous pulsations visible to the angle of the jaw with the patient at 30 degrees. His heart sounds were normal without an S3, but his lungs were notable for bilateral crackles over the lower- to mid-lung fields. He had profound bilateral upper and lower extremity and scrotal pitting edema. He had no lymphadenopathy or skin rashes.
On presentation, the patient’s laboratory results were remarkable for a 444 pg/mL brain natriuretic peptide. A chest X-ray revealed bilateral basilar opacities. An electrocardiogram showed normal sinus rhythm (70 bpm), with normal axis and poor R-wave progression across the precordium. An echocardiogram was performed and notable for a moderately dilated left ventricle with severely depressed systolic function of 10% to 15%, and elevated pulmonary artery pressures. Subsequently, the patient was referred for a coronary angiography, which showed no evidence of coronary atherosclerosis. A cardiac MRI was then performed to evaluate for nonischemic CM, which revealed prominent trabeculations in both ventricles, but most notably in the left ventricle, consistent with a diagnosis of LVNC.
The patient was treated with diuretics, beta-blockers, and an angiotensin-converting enzyme (ACE) inhibitor with improvement in his heart failure symptoms. He was started on systemic anticoagulation with warfarin for his severely depressed LV function. His hospital course was complicated by frequent, nonsustained ventricular tachycardia (VT), and he was referred to the electrophysiology service for implantation of an automated intracardiac cardioverter/defibrillator (AICD) for primary prevention of sudden cardiac death. His clinical course was otherwise unremarkable, and he was discharged after 8 days with complete resolution of his symptoms.
Discussion
The clinical presentation of LVNC is typically due to complications of ventricular dysfunction, including heart failure, arrhythmias, and cardioembolic events. Retrospective studies have shown much variability in the frequency of these complications, likely due to selection bias in earlier studies. These earlier studies had suggested a frequency of heart failure > 50%, but recent studies have shown a more modest frequency of 30% to 35% of affected patients.
Even greater variance has been found in the frequency of arrhythmias, but most studies have shown a frequency of at least 20% for VT. Poor blood flow in the deep intertrabecular recesses in patients with LVNC is additionally thought to lead to a predisposition for mural thrombus formation with an elevated frequency of systemic embolic events, ranging from 5% to 20% among previous studies.1-4,6,8
Much debate remains regarding the genetic association of this condition. The unique character of the resulting myocardium suggests a distinct CM, but the significant genetic heterogeneity with sarcomere protein gene mutations associated with several other CMs, including hypertrophic and dilated CM, suggests that LVNC may simply exist on a phenotypic continuum with these other conditions.4 Inheritance shows additional similarities to these other known CMs with autosomal-
dominant and X-linked modes of transmission shown with familial forms in about 25% of patients.5,7 This has led many to believe that screening of first-degree relatives of clinically affected patients may be appropriate.
The prevalence of LVNC in adults referred for echocardiography is about 0.014% to 1.3%.5 A recent increase in the rate of recognition has raised concerns of possible overdiagnosis, with attempts now made to develop specific imaging diagnostic criteria. Diagnosis of LVNC is most commonly suspected (but can be missed) on echocardiography using 2-D and color Doppler imaging modalities. Echocardiographic findings supporting the diagnosis of LVNC suggested by Oechslin and colleague include:
• Presence of multiple trabeculations, particularly in the LV apex and free wall;
• Multiple deep trabeculation recesses in communication with the LV cavity, usually seen on color Doppler imaging;
• A 2-layered structure of the endomyocardium with ratio of end systolic, noncompacted endocardial layer to compacted epicardial layer > 2 in adults; and
• Absence of other congenital or acquired heart disease, particularly those causing LV outflow obstruction.8
Another proposed standardized method for identifying LVNC via echocardiography by Chin and colleagues focuses on trabeculae at the LV apex on the parasternal short axis and apical views.2,3 LVNC is defined by a ratio of X/Y of ≤ 0.5, where X is the distance from the epicardial surface to the trough of the trabecular recess, and Y is the distance from the epicardial surface to the peak of the trabeculations.
Cardiac MRI is now a more common mode of imaging used for diagnosis of LVNC and often has better imaging characteristics than those of echocardiography. Using a ratio of noncompacted to compacted CM in diastole > 2.3 is suggestive of LVNC with sensitivity and specificity of 86% and 99%.9
The management of LVNC focuses primarily on treatment of complications, including heart failure, rhythm disturbances, and thromboembolic events. Treatment of heart failure is typically the same as for other CMs and includes medical therapy with salt restriction, diuretics, beta-blockers, and ACE inhibitors. In addition, exercise training, as tolerated, is beneficial to improve clinical status.3,10 Electrophysiology studies are often performed in these patients, and implantation of an AICD is typically done in cases of documented, sustained VT, presyncope with inducible VT or severally depressed ejection fraction of < 35%.4,10 Deep intertrabecular recesses and impaired blood flow increase the risk of thrombus formation. Hence, anticoagulation with warfarin (international normalized ratio target 2.3) for those with an impaired LV ejection fraction (< 40%) should be considered for the prevention of cardioembolic events.3,4,6,10
Summary
An active-duty solider with a history of battlefield trauma and multiple hospitalizations was admitted for symptomatic heart failure with cardiac MRI suggestive of LVNC. This condition is a phenotypic result of genetic heterogeneity with significant variability in clinical presentation and a predisposition for heart failure, ventricular arrhythmias, and systemic embolic events. The etiology of this patient’s clinical presentation remains unclear, and additional research is needed to understand whether his recent trauma and multiple hospitalizations played a role in the manifestation of his disease.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Left ventricular noncompaction (LVNC) is a rare disorder that is variably classified as a primary genetic cardiomyopathy (CM) by the American Heart Association.1 It is mostly believed to be a congenital abnormality, characterized by the arrest of the typical embryonic myocardial maturation process with the subsequent retention of the trabecular myocardial structure, which defines the early embryonic heart.2
During very early embryonic development, the left ventricular (LV)myocardium is composed of a loose network of fibers separated by deep recesses, which link it with the LV cavity. At 8 weeks of prenatal development, gradual compaction of these fibers occurs, and LVNC is thought to result from the arrest of this normal process.2,3 Significant variability in myocardial involvement exists, ranging from panventricular to isolated apical involvement, likely related to time of arrest of this maturation process.4 The decreased contractile capability and inadequate epicardial coronary system communication of this trabecular endocardium is thought to lead to the clinical manifestations of LVNC.1-7
This report describes the case of a 45-year-old male soldier who presented with a unique case of heart failure, diagnosed via cardiac magnetic resonance imaging (MRI).
Case Study
The patient presented to the San Antonio Military Medical Center emergency department in mid-2011 with increasing dyspnea for several weeks. He also reported significant lower-extremity and scrotal edema. Although the patient had been previously healthy, his recent medical history was remarkable for a severe combat injury suffered while on duty with the U.S. Army in Afghanistan: He was involved in an explosion from an improvised explosive device in August 2009. He was medically evacuated to the U.S., where he required multiple hospitalizations and surgeries. Prior to his current presentation, the patient had been briefly hospitalized for hospital-acquired pneumonia. During this hospitalization, he first noted abnormal swelling of his legs, a finding that was initially attributed to the large sodium load he had received with his IV antibiotics.
DIAGNOSIS
The patient’s vital signs on presentation were notable for 100/83 mm Hg blood pressure, 103 beats per minute (bpm) heart rate, and 18/min respiratory rate with a saturation of 100% on 4 liters of oxygen by nasal cannula. He was conversant but tachypneic and had to pause frequently to catch his breath. His neck veins were notably distended with jugular venous pulsations visible to the angle of the jaw with the patient at 30 degrees. His heart sounds were normal without an S3, but his lungs were notable for bilateral crackles over the lower- to mid-lung fields. He had profound bilateral upper and lower extremity and scrotal pitting edema. He had no lymphadenopathy or skin rashes.
On presentation, the patient’s laboratory results were remarkable for a 444 pg/mL brain natriuretic peptide. A chest X-ray revealed bilateral basilar opacities. An electrocardiogram showed normal sinus rhythm (70 bpm), with normal axis and poor R-wave progression across the precordium. An echocardiogram was performed and notable for a moderately dilated left ventricle with severely depressed systolic function of 10% to 15%, and elevated pulmonary artery pressures. Subsequently, the patient was referred for a coronary angiography, which showed no evidence of coronary atherosclerosis. A cardiac MRI was then performed to evaluate for nonischemic CM, which revealed prominent trabeculations in both ventricles, but most notably in the left ventricle, consistent with a diagnosis of LVNC.
The patient was treated with diuretics, beta-blockers, and an angiotensin-converting enzyme (ACE) inhibitor with improvement in his heart failure symptoms. He was started on systemic anticoagulation with warfarin for his severely depressed LV function. His hospital course was complicated by frequent, nonsustained ventricular tachycardia (VT), and he was referred to the electrophysiology service for implantation of an automated intracardiac cardioverter/defibrillator (AICD) for primary prevention of sudden cardiac death. His clinical course was otherwise unremarkable, and he was discharged after 8 days with complete resolution of his symptoms.
Discussion
The clinical presentation of LVNC is typically due to complications of ventricular dysfunction, including heart failure, arrhythmias, and cardioembolic events. Retrospective studies have shown much variability in the frequency of these complications, likely due to selection bias in earlier studies. These earlier studies had suggested a frequency of heart failure > 50%, but recent studies have shown a more modest frequency of 30% to 35% of affected patients.
Even greater variance has been found in the frequency of arrhythmias, but most studies have shown a frequency of at least 20% for VT. Poor blood flow in the deep intertrabecular recesses in patients with LVNC is additionally thought to lead to a predisposition for mural thrombus formation with an elevated frequency of systemic embolic events, ranging from 5% to 20% among previous studies.1-4,6,8
Much debate remains regarding the genetic association of this condition. The unique character of the resulting myocardium suggests a distinct CM, but the significant genetic heterogeneity with sarcomere protein gene mutations associated with several other CMs, including hypertrophic and dilated CM, suggests that LVNC may simply exist on a phenotypic continuum with these other conditions.4 Inheritance shows additional similarities to these other known CMs with autosomal-
dominant and X-linked modes of transmission shown with familial forms in about 25% of patients.5,7 This has led many to believe that screening of first-degree relatives of clinically affected patients may be appropriate.
The prevalence of LVNC in adults referred for echocardiography is about 0.014% to 1.3%.5 A recent increase in the rate of recognition has raised concerns of possible overdiagnosis, with attempts now made to develop specific imaging diagnostic criteria. Diagnosis of LVNC is most commonly suspected (but can be missed) on echocardiography using 2-D and color Doppler imaging modalities. Echocardiographic findings supporting the diagnosis of LVNC suggested by Oechslin and colleague include:
• Presence of multiple trabeculations, particularly in the LV apex and free wall;
• Multiple deep trabeculation recesses in communication with the LV cavity, usually seen on color Doppler imaging;
• A 2-layered structure of the endomyocardium with ratio of end systolic, noncompacted endocardial layer to compacted epicardial layer > 2 in adults; and
• Absence of other congenital or acquired heart disease, particularly those causing LV outflow obstruction.8
Another proposed standardized method for identifying LVNC via echocardiography by Chin and colleagues focuses on trabeculae at the LV apex on the parasternal short axis and apical views.2,3 LVNC is defined by a ratio of X/Y of ≤ 0.5, where X is the distance from the epicardial surface to the trough of the trabecular recess, and Y is the distance from the epicardial surface to the peak of the trabeculations.
Cardiac MRI is now a more common mode of imaging used for diagnosis of LVNC and often has better imaging characteristics than those of echocardiography. Using a ratio of noncompacted to compacted CM in diastole > 2.3 is suggestive of LVNC with sensitivity and specificity of 86% and 99%.9
The management of LVNC focuses primarily on treatment of complications, including heart failure, rhythm disturbances, and thromboembolic events. Treatment of heart failure is typically the same as for other CMs and includes medical therapy with salt restriction, diuretics, beta-blockers, and ACE inhibitors. In addition, exercise training, as tolerated, is beneficial to improve clinical status.3,10 Electrophysiology studies are often performed in these patients, and implantation of an AICD is typically done in cases of documented, sustained VT, presyncope with inducible VT or severally depressed ejection fraction of < 35%.4,10 Deep intertrabecular recesses and impaired blood flow increase the risk of thrombus formation. Hence, anticoagulation with warfarin (international normalized ratio target 2.3) for those with an impaired LV ejection fraction (< 40%) should be considered for the prevention of cardioembolic events.3,4,6,10
Summary
An active-duty solider with a history of battlefield trauma and multiple hospitalizations was admitted for symptomatic heart failure with cardiac MRI suggestive of LVNC. This condition is a phenotypic result of genetic heterogeneity with significant variability in clinical presentation and a predisposition for heart failure, ventricular arrhythmias, and systemic embolic events. The etiology of this patient’s clinical presentation remains unclear, and additional research is needed to understand whether his recent trauma and multiple hospitalizations played a role in the manifestation of his disease.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Maron BJ, Towbin JA, Thiene G, et al. Contemporary definitions and classification of the cardiomyopathies: An American Heart Association Scientific Statement from the Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups; and Council on Epidemiology and Prevention. Circulation. 2006;113(14):1807-1816.
2. Chin TK, Perloff JK, Williams RG, Jue K, Mohrmann R. Isolated noncompaction of left ventricular myocardium. A study of eight cases. Circulation. 1990;82(2):507-513.
3. Murphy RT, Thaman R, Blanes JG, et al. Natural history and familial characteristics of isolated left ventricular non-compaction. Eur Heart J. 2005;26(2):187-192.
4. Oechslin E, Jenni R. Left ventricular non-compaction revisited: A distinct phenotype with genetic heterogeneity? Eur Heart J. 2011;32(12):1446-1456.
5. Elliott P, Andersson B, Arbustini E, et al. Classification of the cardiomyopathies: A position statement from the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2008;29(2):270-276.
6. Oechslin EN, Attenhofer Jost CH, Rojas JR, Kaufmann PA, Jenni R. Longterm follow-up of 34 adults with isolated left ventricular noncompaction: A distinct cardiomyopathy with poor prognosis. J Am Coll Cardiol. 2000;36(2):493-500.
7. Spirito P, Autore C. Apical hypertrophic cardiomyopathy or left ventricular non-compaction? A difficult differential diagnosis [editorial]. Eur Heart J. 2007;28(16):1923-1924.
8. Oechslin E, Jenni R. Non-compaction of the left ventricular myocardium—From clinical observation to the discovery of a new disease. Eur Cardiol Review. 2005;1(1):23-24.
9. Petersen SE, Selvanayagam JB, Wiesmann F, et al. Left ventricular non-compaction: Insights from cardiovascular magnetic resonance imaging. J Am Coll Cardiol. 2005;46(1):101-105.
10. Hunt SA, Abraham WT, Chin MH, et al; American College of Cardiology Foundation; American Heart Association. 2009 Focused update incorporated into the ACC/AHA 2005 Guidelines for the Diagnosis and Management of Heart Failure in Adults: A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines Developed in Collaboration With the International Society for Heart and Lung Transplantation. Circulation. 2009;53(15):e1-e90.
1. Maron BJ, Towbin JA, Thiene G, et al. Contemporary definitions and classification of the cardiomyopathies: An American Heart Association Scientific Statement from the Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups; and Council on Epidemiology and Prevention. Circulation. 2006;113(14):1807-1816.
2. Chin TK, Perloff JK, Williams RG, Jue K, Mohrmann R. Isolated noncompaction of left ventricular myocardium. A study of eight cases. Circulation. 1990;82(2):507-513.
3. Murphy RT, Thaman R, Blanes JG, et al. Natural history and familial characteristics of isolated left ventricular non-compaction. Eur Heart J. 2005;26(2):187-192.
4. Oechslin E, Jenni R. Left ventricular non-compaction revisited: A distinct phenotype with genetic heterogeneity? Eur Heart J. 2011;32(12):1446-1456.
5. Elliott P, Andersson B, Arbustini E, et al. Classification of the cardiomyopathies: A position statement from the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2008;29(2):270-276.
6. Oechslin EN, Attenhofer Jost CH, Rojas JR, Kaufmann PA, Jenni R. Longterm follow-up of 34 adults with isolated left ventricular noncompaction: A distinct cardiomyopathy with poor prognosis. J Am Coll Cardiol. 2000;36(2):493-500.
7. Spirito P, Autore C. Apical hypertrophic cardiomyopathy or left ventricular non-compaction? A difficult differential diagnosis [editorial]. Eur Heart J. 2007;28(16):1923-1924.
8. Oechslin E, Jenni R. Non-compaction of the left ventricular myocardium—From clinical observation to the discovery of a new disease. Eur Cardiol Review. 2005;1(1):23-24.
9. Petersen SE, Selvanayagam JB, Wiesmann F, et al. Left ventricular non-compaction: Insights from cardiovascular magnetic resonance imaging. J Am Coll Cardiol. 2005;46(1):101-105.
10. Hunt SA, Abraham WT, Chin MH, et al; American College of Cardiology Foundation; American Heart Association. 2009 Focused update incorporated into the ACC/AHA 2005 Guidelines for the Diagnosis and Management of Heart Failure in Adults: A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines Developed in Collaboration With the International Society for Heart and Lung Transplantation. Circulation. 2009;53(15):e1-e90.
Dexmedetomidine to Remove a Large Thyroid Mass
The following case report describes the use of dexmedetomidine as the primary sedative for an awake endotracheal intubation, as an adjuvant for general anesthesia, and for postoperative sedation for mechanical ventilation. This case illustrates problems that attracted the attention of federal institutions, specifically the management of difficult airways (with and without anatomic distortion), obesity, and obstructive sleep apnea (OSA). As such, it is of potential interest not only to anesthesiologists, but also other health care providers in the VA, especially those who might practice in intensive care settings.
Dexmedetomidine has useful pharmacologic properties that have potential use in a wide variety of clinical scenarios. Dexmedetomidine is currently indicated for sedation in nonintubated patients before and during surgical and other procedures and in intubated and mechanically ventilated patients during treatment in an intensive care setting.
Large neck masses can produce numerous problems that complicate the anesthetic management in the intraoperative and immediate postoperative arenas. The adjuvant use of dexmedetomidine, an alpha-2 agonist that has useful properties for both the anesthetic and intensive care situations, will be discussed. The problems involved with the management and resection of large neck masses include tracheal deviation, tracheal compression, airway edema, distorted anatomy, difficult mask ventilation, difficult intubation, postoperative recurrent laryngeal nerve dysfunction, and difficult exposure for tracheostomy.
Case Report
A 46-year-old man was referred for removal of a large thyroid mass. His past medical history included hypertension, obesity, and type 2 diabetes mellitus. Clinically, the patient seemed to be at risk for OSA, but he had not received a formal diagnosis. The patient met many of the criteria for screening OSA that are listed for a STOP-Bang Questionnaire.1 He was clinically and serologically euthyroid. Neck ultrasound revealed a very large thyroid mass with cystic and solid lesions throughout. Other than hoarseness, the patient reported no compressive symptoms, such as dysphagia or airway compromise. He was maintained on metoprolol, fosinopril, a thiazide for hypertension, and metformin and insulin for diabetes. A physical examination was remarkable for a Mallampati IV airway classification, a 61-cm neck circumference, 177 cm height, 142 kg weight, and a body mass index of 45. These preoperative assessments were predictive of a high probability of very difficult mask ventilation and intubation after the induction of a general anesthetic, or in any other situation requiring tracheal intubation, such as respiratory failure in the postoperative period.
Preoperative laboratory studies, chest radiograph, and electrocardiogram (ECG) were unremarkable. Computed tomography (CT) imaging of the neck revealed marked enlargement of the thyroid, which had a multinodular, heterogeneous appearance with scattered calcifications. The left lobe of the thyroid measured 13.0 cm craniocaudal by 9.47 cm transverse by 6.8 cm anteroposterior. The right lobe of the thyroid measured 12.0 cm craniocaudal by 7.6 cm transverse by 7.0 cm anteroposterior (Figure 1).
The first concern for this patient was a secure airway, which potentially could have been very difficult to procure with a standard IV induction of anesthesia followed by a direct laryngoscopy. This was further constrained by the surgical requirement that the patient be intubated with an electromyography (EMG) endotracheal tube for monitoring of the recurrent laryngeal nerves, as thyroid surgery carries the risk of injury to these nerves. The type of tube that was used had a larger diameter than that of a standard endotracheal tube (the EMG tube measured 10.2 mm outside diameter vs 9.6 mm outside diameter for a standard tube) but was also far more rigid, precluding nasal intubation and making navigation of the tip around corners and obstructions more difficult. A final laryngoscopy was also needed for confirmation of optimal electrode placement at the vocal cord level (Figure 2).
The anesthetic plan was to secure the airway with an awake oral fiberoptic intubation under sedation and topical local anesthetic to avoid the hypoxemia that would ensue if the patient lost spontaneous respiration. The patient was brought without preoperative sedation to the operating room, standard monitors (eg, ECG, noninvasive blood pressure, pulse oximetry) were applied and IV access was obtained. Blood pressure, heart rate, and oxygen saturation were within normal limits. He was placed on oxygen 2 L/min by nasal cannula and given a 1 μg/kg loading dose of dexmedetomidine over 10 minutes and thereafter maintained on a 0.4 μg/kg/h maintenance infusion during the entire airway intubation sequence. A topical anesthesia of 4% lidocaine spray was applied to the upper airway, and a transtracheal injection was performed with 2 mL of 4% lidocaine. The patient’s anatomy precluded the use of superior laryngeal nerve blocks. During the dexmedetomidine loading, he was given 1 mg midazolam and 100 μg fentanyl IV incrementally. No significant hemodynamic or respiratory changes occurred with this sedation regimen.
An attempt to place an oral intubation bite block failed, because the stiff EMG tube proved too difficult to pass through it. Therefore, the EMG tube and rolled gauze pads placed between the upper and lower teeth were used to protect the fiberoptic bronchoscope while it was guided past the base of the tongue. As was noted in the CT scan, the airway was deviated slightly to the left, and this information was useful for guiding the fiberscope. The location of the epiglottis was fairly difficult to ascertain due to redundant tissue in the hypopharyngeal area but was ultimately visible through the fiberscope.
The vocal cords were not visible, possibly due to the significant amount of airway edema and/or redundant tissue between the epiglottis and the vocal cords: Only the space beneath the epiglottis could be seen via the fiberscope. Passing the bronchoscope through the larynx also was problematic due to what may be described as altered spatial/angular relationships and due to the supraglottic edema/tissue leaving little room for the tip of the bronchoscope to be maneuvered. Figure 3 shows a CT scan image of the supraglottic area.
It took 45 minutes and multiple attempts to pass the bronchoscope into the trachea. The dexmedetomidine infusion was continued throughout this entire sequence. The patient tolerated this manipulation with little difficulty, despite the multiple airway maneuvers, and his hemodynamic and respiratory status remained clinically stable. Oxygen saturation was 95% to 100% during this sequence and the patient did not show evidence of significant upper airway collapse, desaturation, or apnea, which are sometimes encountered during sedation for airway manipulation.
The patient’s hemodynamic status remained near baseline values throughout the airway manipulation. The patient never lost his ability to cooperate. After manipulation of the fiberscope into the trachea, the tracheal rings and carina were visualized, and the tube was advanced over the scope. Minimal to mild coughing occurred once the tube passed through the vocal cords. The tube position in the trachea was verified with end-tidal CO2 and bronchoscopy and then the induction of anesthesia with propofol was completed. A laryngoscopy using a videolaryngoscope confirmed proper EMG electrode placement. Large-bore IV access and an arterial line were then secured.
The operation lasted about 15 hours. Maintenance of anesthesia was accomplished with the use of the volatile anesthetic desflurane, titrated to patient response to the surgical procedure. Additionally, 550 μg of IV fentanyl was used intermittently during the operation. Dexmedetomidine was infused at a rate of 0.2 to 0.4 μg/kg/h during the anesthetic, titrated to hemodynamic response. All hemodynamic parameters remained stable and within 20% of preoperative levels during the procedure. The blood loss during the procedure was minimal (< 100 mL), and acceptable readings from the EMG tube were confirmed throughout the surgical procedure.
The 686-gram thyroid mass was confirmed to be a multinodular goiter. Due to the difficulty with intubation, the length of the surgical procedure, and the likelihood of airway difficulties from edema possibly requiring reintubation, the patient was left intubated and mechanically ventilated overnight and sedated with a dexmedetomidine infusion of 0.3 μg/kg/h and propofol 35 mL/h. No further medications were required. He tolerated the ventilator without fighting, straining, coughing, or hypertensive responses and remained cooperative when aroused. He was successfully extubated the following day. Afterward, the patient maintained his airway and had only a mild right vocal cord paresis complicating his surgical management.
Discussion
The critical issues associated with this successful endotracheal intubation included the patient’s obesity, thyroid mass size, and deviation/compression of the trachea. Were this patient morbidly obese only, airway management would still be problematic; this was exacerbated by the concurrent pathologies. Dexmedetomidine possesses several advantageous properties for the perioperative period and was chosen as sedation for the awake intubation due to its sedative-analgesic effects, opioid sparing effects, lack of respiratory depression, maintenance of patient cooperation, and antisialagogue effect.2-5 Dexmedetomidine has previously been shown to be useful for awake intubation of difficult airway cases.4,6,7 Importantly, the dexmedetomidine sedation seemed to blunt the hypertensive responses often seen during airway maneuvers.8 It was also chosen as an intraoperative adjunct due to the above-noted opioid-sparing effects, given the importance of minimizing perioperative opioids needed for this morbidly obese individual with airway compromise.
In the literature, dexmedetomidine has been shown to greatly reduce the need for opioids, both intraoperatively and immediately postoperatively in many citations, for example, in postoperative mechanically ventilated coronary artery bypass graft patients.2,9 Opioid usage reduction is especially needed for the morbidly obese who are at increased risk of OSA and the attendant increased sensitivity to the respiratory depressant effects of narcotics. Postoperative opioids are being debated in the literature as potentially being a risk factor for cancer recurrence due to the effects on the immune system.5,10
Although the pathology report was benign for this patient, it was thought that prior to the surgery a reduction in opioid usage was important because he may have had a thyroid carcinoma in addition to the other respiratory considerations. Additionally, it was desired to decrease the amount of volatile anesthetic agents needed for this patient, because the surgical procedure was anticipated to be quite prolonged (it lasted 15 hours).
Conclusions
Recent research showed that a dexmedetomidine infusion combined with a low-dosage midazolam was superior to a higher dosage midazolam regimen for awake fiberoptic intubation in terms of stability, comfort, cooperation, and patient satisfaction.11 This is an example of the utility of dexmedetomidine. It is often insufficient when used alone, but as an adjunct will markedly reduce the dosage of other sedatives needed to achieve the desired Ramsey sedation scores and/or clinical benefit. Additionally, dexmedetomidine has been shown to facilitate weaning patients in a case series (who had previously failed weaning) from mechanical ventilation in surgical intensive care settings.12
The use of dexmedetomidine facilitated awake intubation for this patient and was helpful for postoperative sedation. The authors believe that dexmedetomidine has potential benefits in all phases of surgery and is a potentially valuable addition to the anesthesiologist’s and intensivist’s armamentarium.
Acknowledgements
This work was conducted at and supported by the G.V. (Sonny) Montgomery VA Medical Center in Jackson, Mississippi.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Hathaway B, Johnson JT. Safety of uvulopalatopharyngoplasty as outpatient surgery. Otolaryngol Head Neck Surg. 2006;134(4):542-544.
2. Gertler R, Brown HC, Mitchell DH, Silvius EN. Dexmedetomidine: A novel sedative-analgesic agent. Proc (Bayl Univ Med Cen). 2001;14(1):13-21.
3. Gurbet A, Basagan-Mogol E, Turker G, Ugun F, Kaya FN, Ozcan B. Intraoperative infusion of dexmedetomidine reduces perioperative analgesic requirements. Can J Anaesth. 2006;53(7):646-652.
4. Ebert TJ, Hall JE, Barney JA, Uhrich TD, Colinco MD. The effects of increasing plasma concentrations of dexmedetomidine in humans. Anesthesiology. 2000;93(2):382-394.
5. Venn RM, Bradshaw CJ, Spencer R, et al. Preliminary UK experience of dexmedetomidine, a novel agent for postoperative sedation in the intensive care unit. Anaesthesia. 1999;54(12):1136-1142.
6. Bergese SD, Khabiri B, Roberts WD, Howie MB, McSweeney TD, Gerhardt MA. Dexmedetomidine for conscious sedation in difficult awake fiberoptic intubation cases. J Clin Anesth. 2007;19(4):141-144.
7. Grant SA, Breslin DS, MacLeod DB, Gleason D, Martin G. Dexmedetomidine infusion for sedation during fiberoptic intubation: A report of three cases. J Clin Anesth. 2004;16(2):124-126.
8. Yildiz M, Tavlan A, Tuncer S, Reisli R, Yosunkaya A, Otelcioglu S. Effect of dexmedetomidine on haemodynamic responses to laryngoscopy and intubation: Perioperative haemodynamics and anaesthetic requirements. Drugs R D. 2006;7(1):43-52.
9. Herr DL, Sum-Ping ST, England M. ICU sedation after coronary artery bypass graft surgery: Dexmedetomidine-based versus propofol-based sedation regimens. J Cardiothorac Vasc Anesth. 2003;17(5):576-584.
10. Biki B, Mascha E, Moriarty DC, Fitzpatrick JM, Sessler DI, Buggy DJ. Anesthetic technique for radical prostatectomy surgery affects cancer recurrence: A retrospective analysis. Anesthesiology. 2008;109(2):180-187.
11. Bergese SD, Patrick Bender S, McSweeney TD, Fernandez S, Dzwonczyk R, Sage K. A comparative study of dexmedetomidine with midazolam and midazolam alone for sedation during elective awake fiberoptic intubation. J Clin Anesth. 2010;22(1):35-40.
12. Siobal MS, Kallet RH, Kivett VA, Tang JF. Use of dexmedetomidine to facilitate extubation in surgical intensive-care-unit patients who failed previous weaning attempts following prolonged mechanical ventilation: A pilot study. Respir Care. 2006;51(5):492-496.
The following case report describes the use of dexmedetomidine as the primary sedative for an awake endotracheal intubation, as an adjuvant for general anesthesia, and for postoperative sedation for mechanical ventilation. This case illustrates problems that attracted the attention of federal institutions, specifically the management of difficult airways (with and without anatomic distortion), obesity, and obstructive sleep apnea (OSA). As such, it is of potential interest not only to anesthesiologists, but also other health care providers in the VA, especially those who might practice in intensive care settings.
Dexmedetomidine has useful pharmacologic properties that have potential use in a wide variety of clinical scenarios. Dexmedetomidine is currently indicated for sedation in nonintubated patients before and during surgical and other procedures and in intubated and mechanically ventilated patients during treatment in an intensive care setting.
Large neck masses can produce numerous problems that complicate the anesthetic management in the intraoperative and immediate postoperative arenas. The adjuvant use of dexmedetomidine, an alpha-2 agonist that has useful properties for both the anesthetic and intensive care situations, will be discussed. The problems involved with the management and resection of large neck masses include tracheal deviation, tracheal compression, airway edema, distorted anatomy, difficult mask ventilation, difficult intubation, postoperative recurrent laryngeal nerve dysfunction, and difficult exposure for tracheostomy.
Case Report
A 46-year-old man was referred for removal of a large thyroid mass. His past medical history included hypertension, obesity, and type 2 diabetes mellitus. Clinically, the patient seemed to be at risk for OSA, but he had not received a formal diagnosis. The patient met many of the criteria for screening OSA that are listed for a STOP-Bang Questionnaire.1 He was clinically and serologically euthyroid. Neck ultrasound revealed a very large thyroid mass with cystic and solid lesions throughout. Other than hoarseness, the patient reported no compressive symptoms, such as dysphagia or airway compromise. He was maintained on metoprolol, fosinopril, a thiazide for hypertension, and metformin and insulin for diabetes. A physical examination was remarkable for a Mallampati IV airway classification, a 61-cm neck circumference, 177 cm height, 142 kg weight, and a body mass index of 45. These preoperative assessments were predictive of a high probability of very difficult mask ventilation and intubation after the induction of a general anesthetic, or in any other situation requiring tracheal intubation, such as respiratory failure in the postoperative period.
Preoperative laboratory studies, chest radiograph, and electrocardiogram (ECG) were unremarkable. Computed tomography (CT) imaging of the neck revealed marked enlargement of the thyroid, which had a multinodular, heterogeneous appearance with scattered calcifications. The left lobe of the thyroid measured 13.0 cm craniocaudal by 9.47 cm transverse by 6.8 cm anteroposterior. The right lobe of the thyroid measured 12.0 cm craniocaudal by 7.6 cm transverse by 7.0 cm anteroposterior (Figure 1).
The first concern for this patient was a secure airway, which potentially could have been very difficult to procure with a standard IV induction of anesthesia followed by a direct laryngoscopy. This was further constrained by the surgical requirement that the patient be intubated with an electromyography (EMG) endotracheal tube for monitoring of the recurrent laryngeal nerves, as thyroid surgery carries the risk of injury to these nerves. The type of tube that was used had a larger diameter than that of a standard endotracheal tube (the EMG tube measured 10.2 mm outside diameter vs 9.6 mm outside diameter for a standard tube) but was also far more rigid, precluding nasal intubation and making navigation of the tip around corners and obstructions more difficult. A final laryngoscopy was also needed for confirmation of optimal electrode placement at the vocal cord level (Figure 2).
The anesthetic plan was to secure the airway with an awake oral fiberoptic intubation under sedation and topical local anesthetic to avoid the hypoxemia that would ensue if the patient lost spontaneous respiration. The patient was brought without preoperative sedation to the operating room, standard monitors (eg, ECG, noninvasive blood pressure, pulse oximetry) were applied and IV access was obtained. Blood pressure, heart rate, and oxygen saturation were within normal limits. He was placed on oxygen 2 L/min by nasal cannula and given a 1 μg/kg loading dose of dexmedetomidine over 10 minutes and thereafter maintained on a 0.4 μg/kg/h maintenance infusion during the entire airway intubation sequence. A topical anesthesia of 4% lidocaine spray was applied to the upper airway, and a transtracheal injection was performed with 2 mL of 4% lidocaine. The patient’s anatomy precluded the use of superior laryngeal nerve blocks. During the dexmedetomidine loading, he was given 1 mg midazolam and 100 μg fentanyl IV incrementally. No significant hemodynamic or respiratory changes occurred with this sedation regimen.
An attempt to place an oral intubation bite block failed, because the stiff EMG tube proved too difficult to pass through it. Therefore, the EMG tube and rolled gauze pads placed between the upper and lower teeth were used to protect the fiberoptic bronchoscope while it was guided past the base of the tongue. As was noted in the CT scan, the airway was deviated slightly to the left, and this information was useful for guiding the fiberscope. The location of the epiglottis was fairly difficult to ascertain due to redundant tissue in the hypopharyngeal area but was ultimately visible through the fiberscope.
The vocal cords were not visible, possibly due to the significant amount of airway edema and/or redundant tissue between the epiglottis and the vocal cords: Only the space beneath the epiglottis could be seen via the fiberscope. Passing the bronchoscope through the larynx also was problematic due to what may be described as altered spatial/angular relationships and due to the supraglottic edema/tissue leaving little room for the tip of the bronchoscope to be maneuvered. Figure 3 shows a CT scan image of the supraglottic area.
It took 45 minutes and multiple attempts to pass the bronchoscope into the trachea. The dexmedetomidine infusion was continued throughout this entire sequence. The patient tolerated this manipulation with little difficulty, despite the multiple airway maneuvers, and his hemodynamic and respiratory status remained clinically stable. Oxygen saturation was 95% to 100% during this sequence and the patient did not show evidence of significant upper airway collapse, desaturation, or apnea, which are sometimes encountered during sedation for airway manipulation.
The patient’s hemodynamic status remained near baseline values throughout the airway manipulation. The patient never lost his ability to cooperate. After manipulation of the fiberscope into the trachea, the tracheal rings and carina were visualized, and the tube was advanced over the scope. Minimal to mild coughing occurred once the tube passed through the vocal cords. The tube position in the trachea was verified with end-tidal CO2 and bronchoscopy and then the induction of anesthesia with propofol was completed. A laryngoscopy using a videolaryngoscope confirmed proper EMG electrode placement. Large-bore IV access and an arterial line were then secured.
The operation lasted about 15 hours. Maintenance of anesthesia was accomplished with the use of the volatile anesthetic desflurane, titrated to patient response to the surgical procedure. Additionally, 550 μg of IV fentanyl was used intermittently during the operation. Dexmedetomidine was infused at a rate of 0.2 to 0.4 μg/kg/h during the anesthetic, titrated to hemodynamic response. All hemodynamic parameters remained stable and within 20% of preoperative levels during the procedure. The blood loss during the procedure was minimal (< 100 mL), and acceptable readings from the EMG tube were confirmed throughout the surgical procedure.
The 686-gram thyroid mass was confirmed to be a multinodular goiter. Due to the difficulty with intubation, the length of the surgical procedure, and the likelihood of airway difficulties from edema possibly requiring reintubation, the patient was left intubated and mechanically ventilated overnight and sedated with a dexmedetomidine infusion of 0.3 μg/kg/h and propofol 35 mL/h. No further medications were required. He tolerated the ventilator without fighting, straining, coughing, or hypertensive responses and remained cooperative when aroused. He was successfully extubated the following day. Afterward, the patient maintained his airway and had only a mild right vocal cord paresis complicating his surgical management.
Discussion
The critical issues associated with this successful endotracheal intubation included the patient’s obesity, thyroid mass size, and deviation/compression of the trachea. Were this patient morbidly obese only, airway management would still be problematic; this was exacerbated by the concurrent pathologies. Dexmedetomidine possesses several advantageous properties for the perioperative period and was chosen as sedation for the awake intubation due to its sedative-analgesic effects, opioid sparing effects, lack of respiratory depression, maintenance of patient cooperation, and antisialagogue effect.2-5 Dexmedetomidine has previously been shown to be useful for awake intubation of difficult airway cases.4,6,7 Importantly, the dexmedetomidine sedation seemed to blunt the hypertensive responses often seen during airway maneuvers.8 It was also chosen as an intraoperative adjunct due to the above-noted opioid-sparing effects, given the importance of minimizing perioperative opioids needed for this morbidly obese individual with airway compromise.
In the literature, dexmedetomidine has been shown to greatly reduce the need for opioids, both intraoperatively and immediately postoperatively in many citations, for example, in postoperative mechanically ventilated coronary artery bypass graft patients.2,9 Opioid usage reduction is especially needed for the morbidly obese who are at increased risk of OSA and the attendant increased sensitivity to the respiratory depressant effects of narcotics. Postoperative opioids are being debated in the literature as potentially being a risk factor for cancer recurrence due to the effects on the immune system.5,10
Although the pathology report was benign for this patient, it was thought that prior to the surgery a reduction in opioid usage was important because he may have had a thyroid carcinoma in addition to the other respiratory considerations. Additionally, it was desired to decrease the amount of volatile anesthetic agents needed for this patient, because the surgical procedure was anticipated to be quite prolonged (it lasted 15 hours).
Conclusions
Recent research showed that a dexmedetomidine infusion combined with a low-dosage midazolam was superior to a higher dosage midazolam regimen for awake fiberoptic intubation in terms of stability, comfort, cooperation, and patient satisfaction.11 This is an example of the utility of dexmedetomidine. It is often insufficient when used alone, but as an adjunct will markedly reduce the dosage of other sedatives needed to achieve the desired Ramsey sedation scores and/or clinical benefit. Additionally, dexmedetomidine has been shown to facilitate weaning patients in a case series (who had previously failed weaning) from mechanical ventilation in surgical intensive care settings.12
The use of dexmedetomidine facilitated awake intubation for this patient and was helpful for postoperative sedation. The authors believe that dexmedetomidine has potential benefits in all phases of surgery and is a potentially valuable addition to the anesthesiologist’s and intensivist’s armamentarium.
Acknowledgements
This work was conducted at and supported by the G.V. (Sonny) Montgomery VA Medical Center in Jackson, Mississippi.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
The following case report describes the use of dexmedetomidine as the primary sedative for an awake endotracheal intubation, as an adjuvant for general anesthesia, and for postoperative sedation for mechanical ventilation. This case illustrates problems that attracted the attention of federal institutions, specifically the management of difficult airways (with and without anatomic distortion), obesity, and obstructive sleep apnea (OSA). As such, it is of potential interest not only to anesthesiologists, but also other health care providers in the VA, especially those who might practice in intensive care settings.
Dexmedetomidine has useful pharmacologic properties that have potential use in a wide variety of clinical scenarios. Dexmedetomidine is currently indicated for sedation in nonintubated patients before and during surgical and other procedures and in intubated and mechanically ventilated patients during treatment in an intensive care setting.
Large neck masses can produce numerous problems that complicate the anesthetic management in the intraoperative and immediate postoperative arenas. The adjuvant use of dexmedetomidine, an alpha-2 agonist that has useful properties for both the anesthetic and intensive care situations, will be discussed. The problems involved with the management and resection of large neck masses include tracheal deviation, tracheal compression, airway edema, distorted anatomy, difficult mask ventilation, difficult intubation, postoperative recurrent laryngeal nerve dysfunction, and difficult exposure for tracheostomy.
Case Report
A 46-year-old man was referred for removal of a large thyroid mass. His past medical history included hypertension, obesity, and type 2 diabetes mellitus. Clinically, the patient seemed to be at risk for OSA, but he had not received a formal diagnosis. The patient met many of the criteria for screening OSA that are listed for a STOP-Bang Questionnaire.1 He was clinically and serologically euthyroid. Neck ultrasound revealed a very large thyroid mass with cystic and solid lesions throughout. Other than hoarseness, the patient reported no compressive symptoms, such as dysphagia or airway compromise. He was maintained on metoprolol, fosinopril, a thiazide for hypertension, and metformin and insulin for diabetes. A physical examination was remarkable for a Mallampati IV airway classification, a 61-cm neck circumference, 177 cm height, 142 kg weight, and a body mass index of 45. These preoperative assessments were predictive of a high probability of very difficult mask ventilation and intubation after the induction of a general anesthetic, or in any other situation requiring tracheal intubation, such as respiratory failure in the postoperative period.
Preoperative laboratory studies, chest radiograph, and electrocardiogram (ECG) were unremarkable. Computed tomography (CT) imaging of the neck revealed marked enlargement of the thyroid, which had a multinodular, heterogeneous appearance with scattered calcifications. The left lobe of the thyroid measured 13.0 cm craniocaudal by 9.47 cm transverse by 6.8 cm anteroposterior. The right lobe of the thyroid measured 12.0 cm craniocaudal by 7.6 cm transverse by 7.0 cm anteroposterior (Figure 1).
The first concern for this patient was a secure airway, which potentially could have been very difficult to procure with a standard IV induction of anesthesia followed by a direct laryngoscopy. This was further constrained by the surgical requirement that the patient be intubated with an electromyography (EMG) endotracheal tube for monitoring of the recurrent laryngeal nerves, as thyroid surgery carries the risk of injury to these nerves. The type of tube that was used had a larger diameter than that of a standard endotracheal tube (the EMG tube measured 10.2 mm outside diameter vs 9.6 mm outside diameter for a standard tube) but was also far more rigid, precluding nasal intubation and making navigation of the tip around corners and obstructions more difficult. A final laryngoscopy was also needed for confirmation of optimal electrode placement at the vocal cord level (Figure 2).
The anesthetic plan was to secure the airway with an awake oral fiberoptic intubation under sedation and topical local anesthetic to avoid the hypoxemia that would ensue if the patient lost spontaneous respiration. The patient was brought without preoperative sedation to the operating room, standard monitors (eg, ECG, noninvasive blood pressure, pulse oximetry) were applied and IV access was obtained. Blood pressure, heart rate, and oxygen saturation were within normal limits. He was placed on oxygen 2 L/min by nasal cannula and given a 1 μg/kg loading dose of dexmedetomidine over 10 minutes and thereafter maintained on a 0.4 μg/kg/h maintenance infusion during the entire airway intubation sequence. A topical anesthesia of 4% lidocaine spray was applied to the upper airway, and a transtracheal injection was performed with 2 mL of 4% lidocaine. The patient’s anatomy precluded the use of superior laryngeal nerve blocks. During the dexmedetomidine loading, he was given 1 mg midazolam and 100 μg fentanyl IV incrementally. No significant hemodynamic or respiratory changes occurred with this sedation regimen.
An attempt to place an oral intubation bite block failed, because the stiff EMG tube proved too difficult to pass through it. Therefore, the EMG tube and rolled gauze pads placed between the upper and lower teeth were used to protect the fiberoptic bronchoscope while it was guided past the base of the tongue. As was noted in the CT scan, the airway was deviated slightly to the left, and this information was useful for guiding the fiberscope. The location of the epiglottis was fairly difficult to ascertain due to redundant tissue in the hypopharyngeal area but was ultimately visible through the fiberscope.
The vocal cords were not visible, possibly due to the significant amount of airway edema and/or redundant tissue between the epiglottis and the vocal cords: Only the space beneath the epiglottis could be seen via the fiberscope. Passing the bronchoscope through the larynx also was problematic due to what may be described as altered spatial/angular relationships and due to the supraglottic edema/tissue leaving little room for the tip of the bronchoscope to be maneuvered. Figure 3 shows a CT scan image of the supraglottic area.
It took 45 minutes and multiple attempts to pass the bronchoscope into the trachea. The dexmedetomidine infusion was continued throughout this entire sequence. The patient tolerated this manipulation with little difficulty, despite the multiple airway maneuvers, and his hemodynamic and respiratory status remained clinically stable. Oxygen saturation was 95% to 100% during this sequence and the patient did not show evidence of significant upper airway collapse, desaturation, or apnea, which are sometimes encountered during sedation for airway manipulation.
The patient’s hemodynamic status remained near baseline values throughout the airway manipulation. The patient never lost his ability to cooperate. After manipulation of the fiberscope into the trachea, the tracheal rings and carina were visualized, and the tube was advanced over the scope. Minimal to mild coughing occurred once the tube passed through the vocal cords. The tube position in the trachea was verified with end-tidal CO2 and bronchoscopy and then the induction of anesthesia with propofol was completed. A laryngoscopy using a videolaryngoscope confirmed proper EMG electrode placement. Large-bore IV access and an arterial line were then secured.
The operation lasted about 15 hours. Maintenance of anesthesia was accomplished with the use of the volatile anesthetic desflurane, titrated to patient response to the surgical procedure. Additionally, 550 μg of IV fentanyl was used intermittently during the operation. Dexmedetomidine was infused at a rate of 0.2 to 0.4 μg/kg/h during the anesthetic, titrated to hemodynamic response. All hemodynamic parameters remained stable and within 20% of preoperative levels during the procedure. The blood loss during the procedure was minimal (< 100 mL), and acceptable readings from the EMG tube were confirmed throughout the surgical procedure.
The 686-gram thyroid mass was confirmed to be a multinodular goiter. Due to the difficulty with intubation, the length of the surgical procedure, and the likelihood of airway difficulties from edema possibly requiring reintubation, the patient was left intubated and mechanically ventilated overnight and sedated with a dexmedetomidine infusion of 0.3 μg/kg/h and propofol 35 mL/h. No further medications were required. He tolerated the ventilator without fighting, straining, coughing, or hypertensive responses and remained cooperative when aroused. He was successfully extubated the following day. Afterward, the patient maintained his airway and had only a mild right vocal cord paresis complicating his surgical management.
Discussion
The critical issues associated with this successful endotracheal intubation included the patient’s obesity, thyroid mass size, and deviation/compression of the trachea. Were this patient morbidly obese only, airway management would still be problematic; this was exacerbated by the concurrent pathologies. Dexmedetomidine possesses several advantageous properties for the perioperative period and was chosen as sedation for the awake intubation due to its sedative-analgesic effects, opioid sparing effects, lack of respiratory depression, maintenance of patient cooperation, and antisialagogue effect.2-5 Dexmedetomidine has previously been shown to be useful for awake intubation of difficult airway cases.4,6,7 Importantly, the dexmedetomidine sedation seemed to blunt the hypertensive responses often seen during airway maneuvers.8 It was also chosen as an intraoperative adjunct due to the above-noted opioid-sparing effects, given the importance of minimizing perioperative opioids needed for this morbidly obese individual with airway compromise.
In the literature, dexmedetomidine has been shown to greatly reduce the need for opioids, both intraoperatively and immediately postoperatively in many citations, for example, in postoperative mechanically ventilated coronary artery bypass graft patients.2,9 Opioid usage reduction is especially needed for the morbidly obese who are at increased risk of OSA and the attendant increased sensitivity to the respiratory depressant effects of narcotics. Postoperative opioids are being debated in the literature as potentially being a risk factor for cancer recurrence due to the effects on the immune system.5,10
Although the pathology report was benign for this patient, it was thought that prior to the surgery a reduction in opioid usage was important because he may have had a thyroid carcinoma in addition to the other respiratory considerations. Additionally, it was desired to decrease the amount of volatile anesthetic agents needed for this patient, because the surgical procedure was anticipated to be quite prolonged (it lasted 15 hours).
Conclusions
Recent research showed that a dexmedetomidine infusion combined with a low-dosage midazolam was superior to a higher dosage midazolam regimen for awake fiberoptic intubation in terms of stability, comfort, cooperation, and patient satisfaction.11 This is an example of the utility of dexmedetomidine. It is often insufficient when used alone, but as an adjunct will markedly reduce the dosage of other sedatives needed to achieve the desired Ramsey sedation scores and/or clinical benefit. Additionally, dexmedetomidine has been shown to facilitate weaning patients in a case series (who had previously failed weaning) from mechanical ventilation in surgical intensive care settings.12
The use of dexmedetomidine facilitated awake intubation for this patient and was helpful for postoperative sedation. The authors believe that dexmedetomidine has potential benefits in all phases of surgery and is a potentially valuable addition to the anesthesiologist’s and intensivist’s armamentarium.
Acknowledgements
This work was conducted at and supported by the G.V. (Sonny) Montgomery VA Medical Center in Jackson, Mississippi.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Hathaway B, Johnson JT. Safety of uvulopalatopharyngoplasty as outpatient surgery. Otolaryngol Head Neck Surg. 2006;134(4):542-544.
2. Gertler R, Brown HC, Mitchell DH, Silvius EN. Dexmedetomidine: A novel sedative-analgesic agent. Proc (Bayl Univ Med Cen). 2001;14(1):13-21.
3. Gurbet A, Basagan-Mogol E, Turker G, Ugun F, Kaya FN, Ozcan B. Intraoperative infusion of dexmedetomidine reduces perioperative analgesic requirements. Can J Anaesth. 2006;53(7):646-652.
4. Ebert TJ, Hall JE, Barney JA, Uhrich TD, Colinco MD. The effects of increasing plasma concentrations of dexmedetomidine in humans. Anesthesiology. 2000;93(2):382-394.
5. Venn RM, Bradshaw CJ, Spencer R, et al. Preliminary UK experience of dexmedetomidine, a novel agent for postoperative sedation in the intensive care unit. Anaesthesia. 1999;54(12):1136-1142.
6. Bergese SD, Khabiri B, Roberts WD, Howie MB, McSweeney TD, Gerhardt MA. Dexmedetomidine for conscious sedation in difficult awake fiberoptic intubation cases. J Clin Anesth. 2007;19(4):141-144.
7. Grant SA, Breslin DS, MacLeod DB, Gleason D, Martin G. Dexmedetomidine infusion for sedation during fiberoptic intubation: A report of three cases. J Clin Anesth. 2004;16(2):124-126.
8. Yildiz M, Tavlan A, Tuncer S, Reisli R, Yosunkaya A, Otelcioglu S. Effect of dexmedetomidine on haemodynamic responses to laryngoscopy and intubation: Perioperative haemodynamics and anaesthetic requirements. Drugs R D. 2006;7(1):43-52.
9. Herr DL, Sum-Ping ST, England M. ICU sedation after coronary artery bypass graft surgery: Dexmedetomidine-based versus propofol-based sedation regimens. J Cardiothorac Vasc Anesth. 2003;17(5):576-584.
10. Biki B, Mascha E, Moriarty DC, Fitzpatrick JM, Sessler DI, Buggy DJ. Anesthetic technique for radical prostatectomy surgery affects cancer recurrence: A retrospective analysis. Anesthesiology. 2008;109(2):180-187.
11. Bergese SD, Patrick Bender S, McSweeney TD, Fernandez S, Dzwonczyk R, Sage K. A comparative study of dexmedetomidine with midazolam and midazolam alone for sedation during elective awake fiberoptic intubation. J Clin Anesth. 2010;22(1):35-40.
12. Siobal MS, Kallet RH, Kivett VA, Tang JF. Use of dexmedetomidine to facilitate extubation in surgical intensive-care-unit patients who failed previous weaning attempts following prolonged mechanical ventilation: A pilot study. Respir Care. 2006;51(5):492-496.
1. Hathaway B, Johnson JT. Safety of uvulopalatopharyngoplasty as outpatient surgery. Otolaryngol Head Neck Surg. 2006;134(4):542-544.
2. Gertler R, Brown HC, Mitchell DH, Silvius EN. Dexmedetomidine: A novel sedative-analgesic agent. Proc (Bayl Univ Med Cen). 2001;14(1):13-21.
3. Gurbet A, Basagan-Mogol E, Turker G, Ugun F, Kaya FN, Ozcan B. Intraoperative infusion of dexmedetomidine reduces perioperative analgesic requirements. Can J Anaesth. 2006;53(7):646-652.
4. Ebert TJ, Hall JE, Barney JA, Uhrich TD, Colinco MD. The effects of increasing plasma concentrations of dexmedetomidine in humans. Anesthesiology. 2000;93(2):382-394.
5. Venn RM, Bradshaw CJ, Spencer R, et al. Preliminary UK experience of dexmedetomidine, a novel agent for postoperative sedation in the intensive care unit. Anaesthesia. 1999;54(12):1136-1142.
6. Bergese SD, Khabiri B, Roberts WD, Howie MB, McSweeney TD, Gerhardt MA. Dexmedetomidine for conscious sedation in difficult awake fiberoptic intubation cases. J Clin Anesth. 2007;19(4):141-144.
7. Grant SA, Breslin DS, MacLeod DB, Gleason D, Martin G. Dexmedetomidine infusion for sedation during fiberoptic intubation: A report of three cases. J Clin Anesth. 2004;16(2):124-126.
8. Yildiz M, Tavlan A, Tuncer S, Reisli R, Yosunkaya A, Otelcioglu S. Effect of dexmedetomidine on haemodynamic responses to laryngoscopy and intubation: Perioperative haemodynamics and anaesthetic requirements. Drugs R D. 2006;7(1):43-52.
9. Herr DL, Sum-Ping ST, England M. ICU sedation after coronary artery bypass graft surgery: Dexmedetomidine-based versus propofol-based sedation regimens. J Cardiothorac Vasc Anesth. 2003;17(5):576-584.
10. Biki B, Mascha E, Moriarty DC, Fitzpatrick JM, Sessler DI, Buggy DJ. Anesthetic technique for radical prostatectomy surgery affects cancer recurrence: A retrospective analysis. Anesthesiology. 2008;109(2):180-187.
11. Bergese SD, Patrick Bender S, McSweeney TD, Fernandez S, Dzwonczyk R, Sage K. A comparative study of dexmedetomidine with midazolam and midazolam alone for sedation during elective awake fiberoptic intubation. J Clin Anesth. 2010;22(1):35-40.
12. Siobal MS, Kallet RH, Kivett VA, Tang JF. Use of dexmedetomidine to facilitate extubation in surgical intensive-care-unit patients who failed previous weaning attempts following prolonged mechanical ventilation: A pilot study. Respir Care. 2006;51(5):492-496.
Education Pitfalls of Insulin Administration in Patients With Diabetes
Diabetes mellitus is a growing problem in the U.S., with the number of disease-related complications on the rise. It affects 29.1 million people of all ages; however, only 21 million people are diagnosed, leaving 8.1 million people undiagnosed.1 Heart disease death rates among adults with diabetes are 2 to 4 times higher than the rates for adults without diabetes.2 At least 68% of patients with diabetes aged > 65 years die of some form of heart disease; 16% die of stroke.2
Type 2 diabetes remains the leading cause for cardiovascular disorders, blindness, end-stage renal disease, amputations, and hospitalizations.3 Due to the long-term complications of diabetes, it is important to help patients control their disease. However, diabetes control in patients can be difficult because of the broad disease education needed and its medication administration.
Insulin requires the most extensive instruction when educating patients with diabetes. Specifically, patient counseling needs to incorporate the importance of proper insulin administration. If patients are not properly administering their insulin, controlling their diabetes will be very difficult. Many clinicians know to educate the patient about drawing insulin into a syringe and how to inject insulin properly. However, clinicians do not always think about other aspects of insulin administration education, such as the mixing of different insulins in 1 syringe. Patients and family members need to be taught about the types of insulins that can and cannot be mixed. The American Diabetes Association (ADA) provides recommendations on the appropriate time to mix insulin and the types of insulin that can and cannot be mixed (Table 1).4
CASE REPORT
A white male, aged 69 years, presented to a pharmacist-run pharmacotherapy clinic for a follow-up appointment for uncontrolled diabetes. The patient’s wife, who managed his medications, accompanied him. Significant past medical history included diabetes, nephropathy, retinopathy, degenerative joint disease, migraines, gastroesophageal reflux disease, depression, posttraumatic stress disorder, hyperlipidemia, hypertension, lumbago, panic attacks, medication noncompliance, status post cerebral vascular accident, and renal insufficiency.
The patient had a long history of type 2 diabetes, and his insulin had been titrated multiple times since he was established in this clinic in 2009. At his establishing visit, he was taken off his insulin pump due to noncompliance with blood glucose checks and placed on basal-bolus therapy with insulin glargine and insulin aspart. The patient then titrated his basal-bolus insulin for 6 weeks but stated his blood sugars were consistently elevated (reaching 600 mg/dL); therefore, he self-reinitiated the insulin pump. After restarting the insulin pump, the clinic made several attempts to follow-up with the patient, but none were successful. He was subsequently dismissed from the clinic following his admission to a local nursing home.
The patient was reestablished at the clinic in 2010 (about 1 year after dismissal). He reported discontinuing the insulin pump and using insulin glargine and insulin aspart injections but was self-adjusting insulin glargine based on readings. He was told not to self-adjust insulin glargine dose and was given a sliding scale for self-adjustment of his insulin aspart dose based on blood glucose readings. Since the reestablished visit, both insulin therapies were titrated without much success in controlling his blood glucose levels. He was also advised to check his fasting blood glucose (FBG) more often and was demonstrated correct insulin drawing technique.
At a follow-up visit in August 2012, the patient’s A1c was 10.7%, and FBG readings ranged from 108 mg/dL to 555 mg/dL. Goal A1c was between 8% and 8.5% per VA/DoD diabetes guidelines.5 After a discussion with the patient’s wife, it was discovered that the patient was improperly administrating his insulin. The patient had been administrating the insulin glargine and insulin aspart in the same syringe. Since the combined dose of insulin was greater than his syringe would allow, he adjusted the insulin glargine dose downward if more insulin aspart was needed per the sliding scale. He did this to avoid more injections than he thought were necessary. Based on his A1c and home blood glucose readings, it was also suspected that insulin doses were being missed. The patient and wife were instructed about the importance of adherence and not mixing these insulins in the same syringe.
At the most recent visit, the patient’s FBG readings (200 mg/dL-500 mg/dL) and A1c (10.7%) were still greatly elevated. He reported taking 40 units insulin glargine in the morning and 60 units at bedtime, along with 40 units insulin aspart plus sliding scale insulin (1:20 > 120 mg/dL) at breakfast and 40 to 70 units at lunch and supper. The patient reported compliance with insulin therapy; however, it was likely he was not dosing accurately, according to his sliding scale. He stated he was eating less and was worried about hypoglycemia. Due to the patient’s FBG and A1c still being elevated, insulin aspart was titrated again, which was closer to a 50% basal and 50% bolus regimen, and he was again educated about proper dosing.
DISCUSSION
Patients have many obstacles to managing their diseases. This is especially prevalent in patients with diabetes. These patients both experience the emotional stress of being diagnosed with diabetes and are given a wealth of information on diabetes, nutrition, therapy, and insulin-dosing technique at the same time. The information can be overwhelming for patients to hear and for the educator to present. Sometimes health care professionals (HCPs) overlook a patient’s hindrances due to the amount of information they have to give to the patient. For example, in this case, the patient was mixing insulin inappropriately, and it was overlooked by the HCP.
Insulin therapy has been used for several decades. It is obtained from either a pork pancreas or is chemically manufactured to be identical to human insulin. This can be achieved by recombinant DNA technology or chemical modification of pork insulin.4 Insulin is available as short-, intermediate-, or long-acting duration. The pharmacokinetics of available insulins is listed in Table 2.6
Some insulin can be mixed in the same syringe, but these mainly consist of the short- and intermediate-acting insulin. Insulin glargine, a long-acting formulation, should not be mixed with any other insulin due to its pharmacokinetic properties.7 Insulin glargine has been designed to have a low solubility at a neutral pH. After injection, the pH rises and leads to the formation of microprecipitates, causing a slow release of the insulin over 24 hours with no peak. If insulin glargine is mixed, it is likely the pH would be altered before entering the body. In addition, mixing insulin in the same syringe could likely contaminate the dose.
The maker of insulin glargine advises against mixing it with any other insulin.7 Several different studies have been done with admixtures of insulin glargine with short- or rapid-acting insulin. The studies revealed no differences in glycemic control, blunted and delayed rapid-acting insulin peak, the need for larger doses, or worsened glycemic control.8-12
Other education points about insulin administration that are often overlooked or sometimes ignored by patients and that require follow-up for compliance include the following:
• Manufacturers recommend discarding an open bottle of insulin at room temperature after 28 days.7
• Insulin should be kept in a temperature-controlled environment between 36°F and 86°F.4,7
• Rotation of injection sites is necessary to prevent lipodystrophy.4,7
• It is recommended that patients stick with 1 approved anatomical site for all insulin injections, such as the abdomen or leg, to maintain consistent pharmacokinetics.4,13,14
It is also important to know the constitution of the different insulins and whether they have been compromised. For example, if a clear solution insulin turns cloudy, it is considered compromised and should be thrown away.
CONCLUSION
Patients are diagnosed every day with diabetes, and many treatment regimens include insulin therapy. With the diagnosis of diabetes, patients are given extensive information on therapy, nutrition, preventative measures, and technique. Since controlling diabetes can call for intensive insulin therapy, medication administration instruction by HCPs is important. It is important to discuss in detail how the patient manages their insulin therapy at each visit so that issues will not be overlooked. Long-term, inappropriate use of insulin may lead to uncontrolled diabetes.
Diabetes is a complex disease to manage and takes a joint effort by both the HCP and patient to control. Patients need to understand the importance of compliance in all aspects of the disease, and the HCP needs to understand the importance of extensive counseling, including diet, exercise, and medication therapy.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Centers for Disease Control and Prevention. National Diabetes Statistics Report: Estimates of Diabetes and Its Burden in the United States, 2014. Atlanta, GA: U.S. Department of Health and Human Services; 2014. http://www.cdc.gov/diabetes/pubs/statsreport14.htm. Updated July 28, 2014. Accessed August 12, 2014.
2. Roger VL, Go AS, Lloyd-Jones DM. et al; American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2012 update: A report from the American Heart Association. Circulation. 2012;125(1):e2-e220.
3. Inzucchi SE, Bergenstal RM, Buse JB, et al; American Diabetes Association (ADA); European Association for the Study of Diabetes (EASD). Management of hyperglycemia in type 2 diabetes: A patient-centered approach: Position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2012;35(6):1364-1379.
4. American Diabetes Association. Insulin administration. Diabetes Care. 2004;27(suppl 1): S106-S109.
5. VA/DoD Clinical Practice Guidelines: Management of Diabetes Mellitus (DM). Version 4.0. Website: http://www.healthquality.va.gov/guidelines/CD/diabetes/DM2010_FUL-v4e.pdf. Updated August 2010. Accessed August 12, 2014.
6. McCulloch DK. General principles of isulin therapy in diabetes mellitus. UpToDate Website. http://www .uptodate.com /contents/general-principles-of-insulin-therapy-in-diabetes-mellitus. Accessed August 5, 2014.
7. Lantus [package insert]. Bridgewater, NJ: sanofi-aventis US; 2013.
8. Cengiz E, Tamborlane WV, Martin-Fredericksen M, Dziura J, Weinzimer SA. Early pharmacokinetic and pharmacodynamic effects of mixing lispro with glargine insulin: Results of glucose clamp studies in youth with type 1 diabetes. Diabetes Care. 2010;33(5):1009-1012.
9. Lucchesi MB, Komatsu WR, Gabbay MA, Dib SA. A 12-wk follow-up study to evaluate the effects of mixing insulin lispro and insulin glargine in young individuals with type 1 diabetes. Pediatr Diabetes. 2012;13(7):519-524.
10. Kaplan W, Rodriguez LM, Smith OE, Haymond MW, Heptulla RA. Effects of mixing glargine and short-acting insulin analogs on glucose control. Diabetes Care. 2004;27(11):2739-2740.
11. Fiallo-Scharer R, Horner B, McFann K, Walravens P, Chase HP. Mixing rapid-acting insulin analogs with insulin glargine in children with type 1 diabetes mellitus. J Pediatr. 2006;148(4):481-484.
12. Hassan K, Rodriguez LM, Johnson SE, Tadlock S, Heptulla RA. A randomized, controlled trial comparing twice-a-day insulin glargine mixed with rapid-acting insulin analogs versus standard neutral protamine Hagedorn (NPH) therapy in newly diagnosed type 1 diabetes. Pediatrics. 2008;121(3):e466 -e472.
13. Koivisto VA, Felig P. Alterations in insulin absorption and in blood glucose control associated with varying insulin injection sites in diabetic patients. Ann Intern Med. 1980;92(1):59-61.
14. Berger M, Cüppers HJ, Hegner H, Jörgens V, Berchtold P. Absorption kinetics and biologic effects of subcutaneously injected insulin preparations. Diabetes Care. 1982;5(2):77-91.
Diabetes mellitus is a growing problem in the U.S., with the number of disease-related complications on the rise. It affects 29.1 million people of all ages; however, only 21 million people are diagnosed, leaving 8.1 million people undiagnosed.1 Heart disease death rates among adults with diabetes are 2 to 4 times higher than the rates for adults without diabetes.2 At least 68% of patients with diabetes aged > 65 years die of some form of heart disease; 16% die of stroke.2
Type 2 diabetes remains the leading cause for cardiovascular disorders, blindness, end-stage renal disease, amputations, and hospitalizations.3 Due to the long-term complications of diabetes, it is important to help patients control their disease. However, diabetes control in patients can be difficult because of the broad disease education needed and its medication administration.
Insulin requires the most extensive instruction when educating patients with diabetes. Specifically, patient counseling needs to incorporate the importance of proper insulin administration. If patients are not properly administering their insulin, controlling their diabetes will be very difficult. Many clinicians know to educate the patient about drawing insulin into a syringe and how to inject insulin properly. However, clinicians do not always think about other aspects of insulin administration education, such as the mixing of different insulins in 1 syringe. Patients and family members need to be taught about the types of insulins that can and cannot be mixed. The American Diabetes Association (ADA) provides recommendations on the appropriate time to mix insulin and the types of insulin that can and cannot be mixed (Table 1).4
CASE REPORT
A white male, aged 69 years, presented to a pharmacist-run pharmacotherapy clinic for a follow-up appointment for uncontrolled diabetes. The patient’s wife, who managed his medications, accompanied him. Significant past medical history included diabetes, nephropathy, retinopathy, degenerative joint disease, migraines, gastroesophageal reflux disease, depression, posttraumatic stress disorder, hyperlipidemia, hypertension, lumbago, panic attacks, medication noncompliance, status post cerebral vascular accident, and renal insufficiency.
The patient had a long history of type 2 diabetes, and his insulin had been titrated multiple times since he was established in this clinic in 2009. At his establishing visit, he was taken off his insulin pump due to noncompliance with blood glucose checks and placed on basal-bolus therapy with insulin glargine and insulin aspart. The patient then titrated his basal-bolus insulin for 6 weeks but stated his blood sugars were consistently elevated (reaching 600 mg/dL); therefore, he self-reinitiated the insulin pump. After restarting the insulin pump, the clinic made several attempts to follow-up with the patient, but none were successful. He was subsequently dismissed from the clinic following his admission to a local nursing home.
The patient was reestablished at the clinic in 2010 (about 1 year after dismissal). He reported discontinuing the insulin pump and using insulin glargine and insulin aspart injections but was self-adjusting insulin glargine based on readings. He was told not to self-adjust insulin glargine dose and was given a sliding scale for self-adjustment of his insulin aspart dose based on blood glucose readings. Since the reestablished visit, both insulin therapies were titrated without much success in controlling his blood glucose levels. He was also advised to check his fasting blood glucose (FBG) more often and was demonstrated correct insulin drawing technique.
At a follow-up visit in August 2012, the patient’s A1c was 10.7%, and FBG readings ranged from 108 mg/dL to 555 mg/dL. Goal A1c was between 8% and 8.5% per VA/DoD diabetes guidelines.5 After a discussion with the patient’s wife, it was discovered that the patient was improperly administrating his insulin. The patient had been administrating the insulin glargine and insulin aspart in the same syringe. Since the combined dose of insulin was greater than his syringe would allow, he adjusted the insulin glargine dose downward if more insulin aspart was needed per the sliding scale. He did this to avoid more injections than he thought were necessary. Based on his A1c and home blood glucose readings, it was also suspected that insulin doses were being missed. The patient and wife were instructed about the importance of adherence and not mixing these insulins in the same syringe.
At the most recent visit, the patient’s FBG readings (200 mg/dL-500 mg/dL) and A1c (10.7%) were still greatly elevated. He reported taking 40 units insulin glargine in the morning and 60 units at bedtime, along with 40 units insulin aspart plus sliding scale insulin (1:20 > 120 mg/dL) at breakfast and 40 to 70 units at lunch and supper. The patient reported compliance with insulin therapy; however, it was likely he was not dosing accurately, according to his sliding scale. He stated he was eating less and was worried about hypoglycemia. Due to the patient’s FBG and A1c still being elevated, insulin aspart was titrated again, which was closer to a 50% basal and 50% bolus regimen, and he was again educated about proper dosing.
DISCUSSION
Patients have many obstacles to managing their diseases. This is especially prevalent in patients with diabetes. These patients both experience the emotional stress of being diagnosed with diabetes and are given a wealth of information on diabetes, nutrition, therapy, and insulin-dosing technique at the same time. The information can be overwhelming for patients to hear and for the educator to present. Sometimes health care professionals (HCPs) overlook a patient’s hindrances due to the amount of information they have to give to the patient. For example, in this case, the patient was mixing insulin inappropriately, and it was overlooked by the HCP.
Insulin therapy has been used for several decades. It is obtained from either a pork pancreas or is chemically manufactured to be identical to human insulin. This can be achieved by recombinant DNA technology or chemical modification of pork insulin.4 Insulin is available as short-, intermediate-, or long-acting duration. The pharmacokinetics of available insulins is listed in Table 2.6
Some insulin can be mixed in the same syringe, but these mainly consist of the short- and intermediate-acting insulin. Insulin glargine, a long-acting formulation, should not be mixed with any other insulin due to its pharmacokinetic properties.7 Insulin glargine has been designed to have a low solubility at a neutral pH. After injection, the pH rises and leads to the formation of microprecipitates, causing a slow release of the insulin over 24 hours with no peak. If insulin glargine is mixed, it is likely the pH would be altered before entering the body. In addition, mixing insulin in the same syringe could likely contaminate the dose.
The maker of insulin glargine advises against mixing it with any other insulin.7 Several different studies have been done with admixtures of insulin glargine with short- or rapid-acting insulin. The studies revealed no differences in glycemic control, blunted and delayed rapid-acting insulin peak, the need for larger doses, or worsened glycemic control.8-12
Other education points about insulin administration that are often overlooked or sometimes ignored by patients and that require follow-up for compliance include the following:
• Manufacturers recommend discarding an open bottle of insulin at room temperature after 28 days.7
• Insulin should be kept in a temperature-controlled environment between 36°F and 86°F.4,7
• Rotation of injection sites is necessary to prevent lipodystrophy.4,7
• It is recommended that patients stick with 1 approved anatomical site for all insulin injections, such as the abdomen or leg, to maintain consistent pharmacokinetics.4,13,14
It is also important to know the constitution of the different insulins and whether they have been compromised. For example, if a clear solution insulin turns cloudy, it is considered compromised and should be thrown away.
CONCLUSION
Patients are diagnosed every day with diabetes, and many treatment regimens include insulin therapy. With the diagnosis of diabetes, patients are given extensive information on therapy, nutrition, preventative measures, and technique. Since controlling diabetes can call for intensive insulin therapy, medication administration instruction by HCPs is important. It is important to discuss in detail how the patient manages their insulin therapy at each visit so that issues will not be overlooked. Long-term, inappropriate use of insulin may lead to uncontrolled diabetes.
Diabetes is a complex disease to manage and takes a joint effort by both the HCP and patient to control. Patients need to understand the importance of compliance in all aspects of the disease, and the HCP needs to understand the importance of extensive counseling, including diet, exercise, and medication therapy.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Diabetes mellitus is a growing problem in the U.S., with the number of disease-related complications on the rise. It affects 29.1 million people of all ages; however, only 21 million people are diagnosed, leaving 8.1 million people undiagnosed.1 Heart disease death rates among adults with diabetes are 2 to 4 times higher than the rates for adults without diabetes.2 At least 68% of patients with diabetes aged > 65 years die of some form of heart disease; 16% die of stroke.2
Type 2 diabetes remains the leading cause for cardiovascular disorders, blindness, end-stage renal disease, amputations, and hospitalizations.3 Due to the long-term complications of diabetes, it is important to help patients control their disease. However, diabetes control in patients can be difficult because of the broad disease education needed and its medication administration.
Insulin requires the most extensive instruction when educating patients with diabetes. Specifically, patient counseling needs to incorporate the importance of proper insulin administration. If patients are not properly administering their insulin, controlling their diabetes will be very difficult. Many clinicians know to educate the patient about drawing insulin into a syringe and how to inject insulin properly. However, clinicians do not always think about other aspects of insulin administration education, such as the mixing of different insulins in 1 syringe. Patients and family members need to be taught about the types of insulins that can and cannot be mixed. The American Diabetes Association (ADA) provides recommendations on the appropriate time to mix insulin and the types of insulin that can and cannot be mixed (Table 1).4
CASE REPORT
A white male, aged 69 years, presented to a pharmacist-run pharmacotherapy clinic for a follow-up appointment for uncontrolled diabetes. The patient’s wife, who managed his medications, accompanied him. Significant past medical history included diabetes, nephropathy, retinopathy, degenerative joint disease, migraines, gastroesophageal reflux disease, depression, posttraumatic stress disorder, hyperlipidemia, hypertension, lumbago, panic attacks, medication noncompliance, status post cerebral vascular accident, and renal insufficiency.
The patient had a long history of type 2 diabetes, and his insulin had been titrated multiple times since he was established in this clinic in 2009. At his establishing visit, he was taken off his insulin pump due to noncompliance with blood glucose checks and placed on basal-bolus therapy with insulin glargine and insulin aspart. The patient then titrated his basal-bolus insulin for 6 weeks but stated his blood sugars were consistently elevated (reaching 600 mg/dL); therefore, he self-reinitiated the insulin pump. After restarting the insulin pump, the clinic made several attempts to follow-up with the patient, but none were successful. He was subsequently dismissed from the clinic following his admission to a local nursing home.
The patient was reestablished at the clinic in 2010 (about 1 year after dismissal). He reported discontinuing the insulin pump and using insulin glargine and insulin aspart injections but was self-adjusting insulin glargine based on readings. He was told not to self-adjust insulin glargine dose and was given a sliding scale for self-adjustment of his insulin aspart dose based on blood glucose readings. Since the reestablished visit, both insulin therapies were titrated without much success in controlling his blood glucose levels. He was also advised to check his fasting blood glucose (FBG) more often and was demonstrated correct insulin drawing technique.
At a follow-up visit in August 2012, the patient’s A1c was 10.7%, and FBG readings ranged from 108 mg/dL to 555 mg/dL. Goal A1c was between 8% and 8.5% per VA/DoD diabetes guidelines.5 After a discussion with the patient’s wife, it was discovered that the patient was improperly administrating his insulin. The patient had been administrating the insulin glargine and insulin aspart in the same syringe. Since the combined dose of insulin was greater than his syringe would allow, he adjusted the insulin glargine dose downward if more insulin aspart was needed per the sliding scale. He did this to avoid more injections than he thought were necessary. Based on his A1c and home blood glucose readings, it was also suspected that insulin doses were being missed. The patient and wife were instructed about the importance of adherence and not mixing these insulins in the same syringe.
At the most recent visit, the patient’s FBG readings (200 mg/dL-500 mg/dL) and A1c (10.7%) were still greatly elevated. He reported taking 40 units insulin glargine in the morning and 60 units at bedtime, along with 40 units insulin aspart plus sliding scale insulin (1:20 > 120 mg/dL) at breakfast and 40 to 70 units at lunch and supper. The patient reported compliance with insulin therapy; however, it was likely he was not dosing accurately, according to his sliding scale. He stated he was eating less and was worried about hypoglycemia. Due to the patient’s FBG and A1c still being elevated, insulin aspart was titrated again, which was closer to a 50% basal and 50% bolus regimen, and he was again educated about proper dosing.
DISCUSSION
Patients have many obstacles to managing their diseases. This is especially prevalent in patients with diabetes. These patients both experience the emotional stress of being diagnosed with diabetes and are given a wealth of information on diabetes, nutrition, therapy, and insulin-dosing technique at the same time. The information can be overwhelming for patients to hear and for the educator to present. Sometimes health care professionals (HCPs) overlook a patient’s hindrances due to the amount of information they have to give to the patient. For example, in this case, the patient was mixing insulin inappropriately, and it was overlooked by the HCP.
Insulin therapy has been used for several decades. It is obtained from either a pork pancreas or is chemically manufactured to be identical to human insulin. This can be achieved by recombinant DNA technology or chemical modification of pork insulin.4 Insulin is available as short-, intermediate-, or long-acting duration. The pharmacokinetics of available insulins is listed in Table 2.6
Some insulin can be mixed in the same syringe, but these mainly consist of the short- and intermediate-acting insulin. Insulin glargine, a long-acting formulation, should not be mixed with any other insulin due to its pharmacokinetic properties.7 Insulin glargine has been designed to have a low solubility at a neutral pH. After injection, the pH rises and leads to the formation of microprecipitates, causing a slow release of the insulin over 24 hours with no peak. If insulin glargine is mixed, it is likely the pH would be altered before entering the body. In addition, mixing insulin in the same syringe could likely contaminate the dose.
The maker of insulin glargine advises against mixing it with any other insulin.7 Several different studies have been done with admixtures of insulin glargine with short- or rapid-acting insulin. The studies revealed no differences in glycemic control, blunted and delayed rapid-acting insulin peak, the need for larger doses, or worsened glycemic control.8-12
Other education points about insulin administration that are often overlooked or sometimes ignored by patients and that require follow-up for compliance include the following:
• Manufacturers recommend discarding an open bottle of insulin at room temperature after 28 days.7
• Insulin should be kept in a temperature-controlled environment between 36°F and 86°F.4,7
• Rotation of injection sites is necessary to prevent lipodystrophy.4,7
• It is recommended that patients stick with 1 approved anatomical site for all insulin injections, such as the abdomen or leg, to maintain consistent pharmacokinetics.4,13,14
It is also important to know the constitution of the different insulins and whether they have been compromised. For example, if a clear solution insulin turns cloudy, it is considered compromised and should be thrown away.
CONCLUSION
Patients are diagnosed every day with diabetes, and many treatment regimens include insulin therapy. With the diagnosis of diabetes, patients are given extensive information on therapy, nutrition, preventative measures, and technique. Since controlling diabetes can call for intensive insulin therapy, medication administration instruction by HCPs is important. It is important to discuss in detail how the patient manages their insulin therapy at each visit so that issues will not be overlooked. Long-term, inappropriate use of insulin may lead to uncontrolled diabetes.
Diabetes is a complex disease to manage and takes a joint effort by both the HCP and patient to control. Patients need to understand the importance of compliance in all aspects of the disease, and the HCP needs to understand the importance of extensive counseling, including diet, exercise, and medication therapy.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Centers for Disease Control and Prevention. National Diabetes Statistics Report: Estimates of Diabetes and Its Burden in the United States, 2014. Atlanta, GA: U.S. Department of Health and Human Services; 2014. http://www.cdc.gov/diabetes/pubs/statsreport14.htm. Updated July 28, 2014. Accessed August 12, 2014.
2. Roger VL, Go AS, Lloyd-Jones DM. et al; American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2012 update: A report from the American Heart Association. Circulation. 2012;125(1):e2-e220.
3. Inzucchi SE, Bergenstal RM, Buse JB, et al; American Diabetes Association (ADA); European Association for the Study of Diabetes (EASD). Management of hyperglycemia in type 2 diabetes: A patient-centered approach: Position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2012;35(6):1364-1379.
4. American Diabetes Association. Insulin administration. Diabetes Care. 2004;27(suppl 1): S106-S109.
5. VA/DoD Clinical Practice Guidelines: Management of Diabetes Mellitus (DM). Version 4.0. Website: http://www.healthquality.va.gov/guidelines/CD/diabetes/DM2010_FUL-v4e.pdf. Updated August 2010. Accessed August 12, 2014.
6. McCulloch DK. General principles of isulin therapy in diabetes mellitus. UpToDate Website. http://www .uptodate.com /contents/general-principles-of-insulin-therapy-in-diabetes-mellitus. Accessed August 5, 2014.
7. Lantus [package insert]. Bridgewater, NJ: sanofi-aventis US; 2013.
8. Cengiz E, Tamborlane WV, Martin-Fredericksen M, Dziura J, Weinzimer SA. Early pharmacokinetic and pharmacodynamic effects of mixing lispro with glargine insulin: Results of glucose clamp studies in youth with type 1 diabetes. Diabetes Care. 2010;33(5):1009-1012.
9. Lucchesi MB, Komatsu WR, Gabbay MA, Dib SA. A 12-wk follow-up study to evaluate the effects of mixing insulin lispro and insulin glargine in young individuals with type 1 diabetes. Pediatr Diabetes. 2012;13(7):519-524.
10. Kaplan W, Rodriguez LM, Smith OE, Haymond MW, Heptulla RA. Effects of mixing glargine and short-acting insulin analogs on glucose control. Diabetes Care. 2004;27(11):2739-2740.
11. Fiallo-Scharer R, Horner B, McFann K, Walravens P, Chase HP. Mixing rapid-acting insulin analogs with insulin glargine in children with type 1 diabetes mellitus. J Pediatr. 2006;148(4):481-484.
12. Hassan K, Rodriguez LM, Johnson SE, Tadlock S, Heptulla RA. A randomized, controlled trial comparing twice-a-day insulin glargine mixed with rapid-acting insulin analogs versus standard neutral protamine Hagedorn (NPH) therapy in newly diagnosed type 1 diabetes. Pediatrics. 2008;121(3):e466 -e472.
13. Koivisto VA, Felig P. Alterations in insulin absorption and in blood glucose control associated with varying insulin injection sites in diabetic patients. Ann Intern Med. 1980;92(1):59-61.
14. Berger M, Cüppers HJ, Hegner H, Jörgens V, Berchtold P. Absorption kinetics and biologic effects of subcutaneously injected insulin preparations. Diabetes Care. 1982;5(2):77-91.
1. Centers for Disease Control and Prevention. National Diabetes Statistics Report: Estimates of Diabetes and Its Burden in the United States, 2014. Atlanta, GA: U.S. Department of Health and Human Services; 2014. http://www.cdc.gov/diabetes/pubs/statsreport14.htm. Updated July 28, 2014. Accessed August 12, 2014.
2. Roger VL, Go AS, Lloyd-Jones DM. et al; American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2012 update: A report from the American Heart Association. Circulation. 2012;125(1):e2-e220.
3. Inzucchi SE, Bergenstal RM, Buse JB, et al; American Diabetes Association (ADA); European Association for the Study of Diabetes (EASD). Management of hyperglycemia in type 2 diabetes: A patient-centered approach: Position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2012;35(6):1364-1379.
4. American Diabetes Association. Insulin administration. Diabetes Care. 2004;27(suppl 1): S106-S109.
5. VA/DoD Clinical Practice Guidelines: Management of Diabetes Mellitus (DM). Version 4.0. Website: http://www.healthquality.va.gov/guidelines/CD/diabetes/DM2010_FUL-v4e.pdf. Updated August 2010. Accessed August 12, 2014.
6. McCulloch DK. General principles of isulin therapy in diabetes mellitus. UpToDate Website. http://www .uptodate.com /contents/general-principles-of-insulin-therapy-in-diabetes-mellitus. Accessed August 5, 2014.
7. Lantus [package insert]. Bridgewater, NJ: sanofi-aventis US; 2013.
8. Cengiz E, Tamborlane WV, Martin-Fredericksen M, Dziura J, Weinzimer SA. Early pharmacokinetic and pharmacodynamic effects of mixing lispro with glargine insulin: Results of glucose clamp studies in youth with type 1 diabetes. Diabetes Care. 2010;33(5):1009-1012.
9. Lucchesi MB, Komatsu WR, Gabbay MA, Dib SA. A 12-wk follow-up study to evaluate the effects of mixing insulin lispro and insulin glargine in young individuals with type 1 diabetes. Pediatr Diabetes. 2012;13(7):519-524.
10. Kaplan W, Rodriguez LM, Smith OE, Haymond MW, Heptulla RA. Effects of mixing glargine and short-acting insulin analogs on glucose control. Diabetes Care. 2004;27(11):2739-2740.
11. Fiallo-Scharer R, Horner B, McFann K, Walravens P, Chase HP. Mixing rapid-acting insulin analogs with insulin glargine in children with type 1 diabetes mellitus. J Pediatr. 2006;148(4):481-484.
12. Hassan K, Rodriguez LM, Johnson SE, Tadlock S, Heptulla RA. A randomized, controlled trial comparing twice-a-day insulin glargine mixed with rapid-acting insulin analogs versus standard neutral protamine Hagedorn (NPH) therapy in newly diagnosed type 1 diabetes. Pediatrics. 2008;121(3):e466 -e472.
13. Koivisto VA, Felig P. Alterations in insulin absorption and in blood glucose control associated with varying insulin injection sites in diabetic patients. Ann Intern Med. 1980;92(1):59-61.
14. Berger M, Cüppers HJ, Hegner H, Jörgens V, Berchtold P. Absorption kinetics and biologic effects of subcutaneously injected insulin preparations. Diabetes Care. 1982;5(2):77-91.