User login
Remove the ovaries at hysterectomy? Here’s the lowdown on risks and benefits
CASE: Hysterectomy candidate asks about her ovaries
A 51-year-old premenopausal woman complains of severe menorrhagia that often causes her to miss work. Although she is taking an iron supplement, her hemoglobin level often drops below 10 g/dL. She has already been identified as having fibroids, with a uterine size of 14 weeks. You order ultra-sonography, which reveals an enlarged uterus with multiple fibroids and normal endometrial thickness, but no intracavitary lesions.
After you describe the treatment options, including uterine artery embolization, the patient requests a hysterectomy as a reasonably low-risk means of cure. During informed consent, she asks whether she should have her ovaries removed during the surgery. Further discussion reveals that her father died of a myocardial infarction when he was 64 years old, but there is no family or personal history of ovarian or breast cancer.
How do you advise this patient, based on her history and recent findings from medical research?
Many gynecologists have been trained to recommend bilateral oophorectomy for women older than 45 or 50 years who request a hysterectomy for benign disease. In these women, oophorectomy is recommended to prevent ovarian cancer and avert the potential for other ovarian pathology that might require later surgery.
In the United States, 78% of women 45 to 64 years old and 55% of women overall undergo bilateral oophorectomy at the time of hysterectomy.1 These percentages mean that almost 300,000 women undergo bilateral oophorectomy each year.1
Hysterectomy alone can sometimes lead to early ovarian failure, but this phenomenon is infrequent. A prospective study of premenopausal women found that, after 5 years of follow-up, 20% of women who underwent simple hysterectomy reached menopause, compared with 7% of matched women who did not undergo hysterectomy.2
In this article, I explore the risks and benefits associated with bilateral oophorectomy and present an algorithm to aid in deciding whether the patient should keep her ovaries—and when oophorectomy might be a better option ( FIGURE ).
Among the hazards associated with bilateral oophorectomy are:
- an increased risk of death from coronary artery disease (CAD), lung cancer, all cancers (except ovarian), and all causes3,4
- an increased risk of osteoporosis and hip fracture5
- when performed before the onset of menopause, an increased risk of parkinsonism, cognitive impairment, dementia, anxiety, and depression.6-8
Benefits include a reduced risk of ovarian cancer, particularly among women who have a BRCA gene mutation or strong family history of ovarian or breast cancer.
Although ovarian cancer causes 15,000 deaths each year in the United States, that figure pales when compared with heart disease, which accounts for 350,000 deaths. In addition, hip fracture may cause approximately 66,000 deaths each year, and dementia attributable to bilateral oophorectomy may affect 100,000 to 200,000 women.9 Reoperation for adnexal pathology or pain after hysterectomy is rare, occurring in only 2.8% of women. Therefore, the benefits of oophorectomy are often outweighed by the risks of CAD, hip fracture, and neurologic conditions.
The assumption that medical treatment will ameliorate the risks associated with oophorectomy is unrealistic. Estrogen may mitigate some risks, but many women avoid hormone therapy. This avoidance can be especially problematic in young women.
In the Nurses’ Health Study, a separate analysis focused on women who had never used postmenopausal hormone therapy.4 In this analysis, all women who underwent bilateral oophorectomy had a greater risk of stroke (HR, 1.85; 95% CI, 1.09, 3.16) and lung cancer (HR, 2.09; 95% CI, 1.01, 4.33) than did women who retained their ovaries. Among women who underwent oophorectomy before age 50 and who did not take estrogen, the risk of coronary artery disease (CAD) was higher (HR, 1.98; 95% CI, 1.18, 3.32), as was the risk of death from all causes (HR, 1.40; 95% CI, 1.01, 1.96), compared with women who retained their ovaries.
Despite estrogen’s proven benefit among oophorectomized women, usage rates continue to decline. In the 6 months after publication of Women’s Health Initiative findings on estrogen–progestin therapy, the continuation rate of estrogen therapy decreased from 12.6% to 9.1%, and new starts also declined significantly.47
Among women who had a diagnosis of osteoporosis and who began treatment with estrogen, estrogen plus progestin, a bisphosphonate, or raloxifene, medication continuation rates were less than 25% at 12 months.48 Moreover, only 18% of women started on a statin to reduce the risk of cardiovascular disease were still taking the drug after 1 year.49
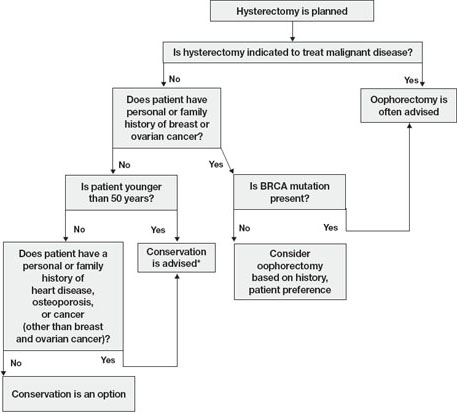
FIGURE Conservation vs oophorectomy: A guide to decision-making
* Estrogen replacement is recommended for women younger than 45 years who opt for oophorectomy
Ovarian cancer is a real, but relatively low, risk
In 2008, an estimated 21,650 new cases of ovarian cancer were diagnosed (age at diagnosis: mean, 63 years), and 15,520 women died from the disease.10 Because we lack a reliable screening test to detect early-stage ovarian cancer in the general population, most women are given a diagnosis when disease is advanced and the 5-year survival rate is 15% to 25%.
There is agreement that women who are known to have a BRCA mutation, which increases the risk of ovarian and breast cancer, should strongly consider oophorectomy once childbearing is complete.11 In the general population, however, the outlook is different.
In the United States, the lifetime risk of ovarian cancer is 1.4% overall. Among white women who have had three or more term pregnancies and who have used an oral contraceptive for at least 4 years, the lifetime risk of ovarian cancer drops to 0.3%.12
Need for reoperation is very low
The percentage of women who require reoperation after ovarian conservation—2.8%—may surprise you.13 That figure is lower than once thought because many studies were performed before asymptomatic, benign ovarian cysts were determined to be a fairly common phenomenon in postmenopausal women (prevalence, 6.6%). These cysts do not undergo transformation to cancer and, therefore, do not need to be removed.14
In addition, studies indicate that only 0.1% to 0.75% of women who retain their ovaries at the time of hysterectomy develop ovarian cancer.15,16 Therefore, the rationale of performing oophorectomy to avoid future surgery appears to be unfounded.
CAD risk rises sharply after oophorectomy
A recent systematic review found mixed evidence concerning the risk of CAD following bilateral salpingo-oophorectomy.17 In observational studies, however, earlier age of surgical or natural menopause has been associated with a higher risk of cardiovascular mortality.18-20 Early reports from the Nurses’ Health Study found that the risk of myocardial infarction doubled among women who underwent oophorectomy and never used estrogen, compared with age-matched premenopausal women (relative risk [RR], 2.2; 95% confidence interval [CI], 1.2, 4.2).3 Even after age 50, the risk of a first myocardial infarction is increased among oophorectomized women, compared with women who retain their ovaries (RR, 1.4; 95% CI, 1.0–2.0).21
A study by researchers from the Mayo Clinic, who examined all causes listed on the death certificate, found a significant association between bilateral oophorectomy before the age of 45 years and cardiovascular mortality (hazard ratio [HR], 1.44; 95% CI, 1.01–2.05).22 This risk was significantly increased among women who were not treated with estrogen through at least age 45, compared with estrogen-treated women.
Oophorectomy may impair bone health
After menopause, ovaries continue to produce significant amounts of the androgens testosterone and androstenedione, which are converted to estrone peripherally by skin, muscle, and fat cells.23,24 The levels of these hormones remain consistent and have been documented to age 80.25
Both estrogens and androgens inhibit bone resorption, and androgens also stimulate bone formation.26 Low levels of androgens and estrogens are linked to lower bone density and a higher risk of hip and vertebral fracture in postmenopausal women.27-29
Postmenopausal women who have been oophorectomized may have an even greater risk of osteoporosis. Over 16 years of follow-up, 340 women who had undergone oophorectomy at a median age of 62 years had 54% more osteoporotic fractures than women who had intact ovaries.5 Two other studies found no association between oophorectomy and bone loss or fracture risk, however.30,31
Hip fracture is a well-documented cause of increased morbidity and mortality in older women. One study found that, before hip fracture, 28% of patients were housebound; 1 year after hip fracture, the percentage was 46%.32 Women older than 60 who underwent oophorectomy had a doubled risk of mortality after low-trauma hip fracture, compared with women who had intact ovaries (odds ratio [OR], 2.18; 95% CI, 2.03–2.32).5
Loss of ovaries may affect mental health and sexuality
In a premenopausal woman, oophorectomy causes a sudden loss of estrogen and often triggers hot flashes, mood changes, sleep disturbances, headaches, and a decline in feelings of well-being.33,34 Over time, vaginal dryness, painful intercourse, loss of libido, bladder dysfunction, and depression may occur.35,36
Evidence suggests that, in women, sexual desire, sexual sensation, and orgasmic response are influenced by androgens. After elective oophorectomy, declines in sexual desire have been reported.37-39
Mental health and sexuality may rebound over time, however. One study found less improvement in mental health measures and body image 6 months after hysterectomy among women who were oophorectomized, compared with those who retained their ovaries. After 2 years, improvement levels were similar between groups.40
Cognitive function may suffer
Analysis of data from the Mayo Clinic Cohort Study of Oophorectomy and Aging found that bilateral oophorectomy before the onset of menopause increased the risk of parkinsonism, cognitive impairment or dementia, and anxiety or depression, particularly if estrogen was not replaced.6-8 These risks increased with younger age at oophorectomy.
The Women’s Health Initiative found an increased risk of dementia or mild cognitive impairment in women who were treated by estrogen alone or estrogen plus progestin after age 65.41-44
These disparate conclusions suggest that estrogen may have a protective effect on the brain if it is given right after natural menopause or premenopausal oophorectomy, but deleterious effects if it is started years later.45
Other studies of endogenous estrogen and cognitive function are few and yield inconsistent results.
Ovarian conservation boosts long-term survival
When there is no family history of ovarian cancer, ovarian conservation appears to maximize survival among healthy women 40 to 65 years old who undergo hysterectomy for benign disease.46 Among healthy women hysterectomized before the age of 55, calculations suggest that 8.6% more would be alive at age 80 if their ovaries were conserved than if they were removed.46
A study from the Mayo Clinic found that all-cause mortality was significantly higher among women who underwent prophylactic bilateral oophorectomy before the age of 45 than it was among women in the control group (HR, 1.67; 95% CI, 1.16–2.40); it was particularly high in women who did not receive estrogen treatment before age 45 (HR, 1.93; 95% CI, 1.25–2.96).22
In a recent study, investigators used the Nurses’ Health Study database to explore the long-term health outcomes of 29,380 women who underwent hysterectomy.4 Of these women, 13,035 (44.4%) had their ovaries conserved, and 16,345 (55.6%) underwent bilateral oophorectomy. Follow-up was 24 years. Oophorectomy was associated with an increased risk of nonfatal CAD among all women (HR, 1.17; 95% CI, 1.02, 1.35), especially those who underwent the procedure before age 45 (HR, 1.26; 95% CI, 1.04, 1.54). Oophorectomy was associated with a markedly reduced risk of ovarian cancer but an increased risk of lung cancer (HR, 1.26; 95% CI, 1.02–1.56).
In regard to fatal events, oophorectomy increased the risk of death from all causes (HR, 1.12; 95% CI, 1.03, 1.21). Specifically, there was an increased risk of death from CAD (HR, 1.28; 95% CI, 1.00, 1.64), lung cancer (HR, 1.31; 95% CI, 1.02, 1.68), and all cancers (HR, 1.17; 95% CI, 1.04, 1.32). There was no overall difference in the risk of death from stroke, breast cancer, and colorectal cancer between women who underwent oophorectomy and those who retained their ovaries.
During the 24 years of follow-up, 37 women died from ovarian cancer, accounting for 1.2% of all deaths. At no age did oophorectomy show a survival benefit.
How this evidence should inform your practice
It is unfortunate that the entire body of evidence on the risks and benefits of bilateral salpingo-oophorectomy consists of observational studies, which have significant inherent limitations. Although the Nurses’ Health Study was the largest prospective study to examine the effect of oophorectomy on women’s health, and involved the longest follow-up, the study was observational, and oophorectomy and ovarian conservation were self-selected. Nevertheless, recent data suggest that a more detailed informed-consent process is warranted than the process in place. Informed consent should cover the risks and benefits of both oophorectomy and ovarian conservation.
Prophylactic oophorectomy is recommended only if a preponderance of the evidence establishes that it clearly benefits the patient. The studies described in this article suggest that bilateral oophorectomy does harm more often than it does good. Therefore, a cautious approach to oophorectomy at the time of hysterectomy is advised.
CASE RESOLVED
After you describe the risks and benefits of oophorectomy, and address the patient’s concerns about her family history of heart disease, she decides to keep her ovaries.
1. Healthcare Cost and Utilization Project (HCUP), 1988–2001: A Federal–State–Industry Partnership in Health Data. Rockville, Md: Agency for Healthcare Research and Quality; July 2003.
2. Farquhar CM, Sadler L, Harvey SA, Stewart AW. The association of hysterectomy and menopause: a prospective cohort study. BJOG. 2005;112:956-962.
3. Colditz G, Willett W, Stampfer M, Rosner B, Speizer F, Hennekens C. Menopause and the risk of coronary heart disease in women. N Engl J Med. 1987;316:1105-1110.
4. Parker WH, Broder MS, Chang E, et al. Ovarian conservation at the time of hysterectomy and long-term health outcomes in the Nurses’ Health Study. Obstet Gynecol. 2009;113:1027-1037.
5. Melton LJ 3rd, Khosla S, Malkasian GD, Achenbach SJ, Oberg AL, Riggs BL. Fracture risk after bilateral oophorectomy in elderly women. J Bone Miner Res. 2003;18:900-905.
6. Rocca WA, Bower JH, Maraganore DM, et al. Increased risk of cognitive impairment or dementia in women who underwent oophorectomy before menopause. Neurology. 2007;69:1074.-
7. Rocca WA, Bower JH, Maraganore DM, et al. Increased risk of parkinsonism in women who underwent oophorectomy before menopause. Neurology. 2008;70:200-209.
8. Rocca WA, Grossardt BR, Geda YE, et al. Long-term risk of depressive and anxiety symptoms after early bilateral oophorectomy. Menopause. 2008;15:1050-1059.
9. Bennett DA. Editorial comment on “Prevalence of dementia in the United States: the aging, demographics, and memory study” by Plassman et al. Neuroepidemiology. 2007;29:133-135.
10. Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58:71-96.
11. Armstrong K, Schwartz J, Randall T, Rubin S, Weber B. Hormone replacement therapy and life expectancy after prophylactic oophorectomy in women with BRCA1/2 mutations: a decision analysis. J Clin Oncol. 2004;22:1045-1054.
12. Hartge P, Whittemore AS, Itnyre J, McGowan L, Cramer D. Rates and risks of ovarian cancer in subgroups of white women in the United States. The Collaborative Ovarian Cancer Group. Obstet Gynecol. 1994;84:760-764.
13. Dekel A, Efrat Z, Orvieto R, et al. The residual ovary syndrome: a 20-year experience. Eur J Obstet Gynecol Reprod Biol. 1996;68:159-164.
14. Bailey CL, Ueland FR, Land GL, et al. The malignant potential of small cystic ovarian tumors in women over 50 years of age. Gynecol Oncol. 1998;69:3-7.
15. Naylor AC. Hysterectomy—analysis of 2,901 personally performed procedures. S Afr Med J. 1984;65:242-245.
16. Christ JE, Lotze EC. The residual ovary syndrome. Obstet Gynecol. 1975;46:551-556.
17. Jacoby VL, Grady D, Sawaya GF. Oophorectomy as a risk factor for coronary heart disease. Am J Obstet Gynecol. 2009;200:140.e1-140.e9.
18. van der Schouw YT, van der Graaf Y, Steyerberg EW, Eijkemans JC, Banga JD. Age at menopause as a risk factor for cardiovascular mortality. Lancet. 1996;347:714-718.
19. Løkkegaard E, Jovanovic Z, Heitmann BL, et al. The association between early menopause and risk of ischaemic heart disease: influence of hormone therapy. Maturitas. 2006;53:226-233.
20. de Kleijn MJJ, van der Schouw YT, Verbeek ALM, et al. Endogenous estrogen exposure and cardiovascular mortality risk in postmenopausal women. Am J Epidemiol. 2002;155:339.-
21. Falkeborn M, Schairer C, Naessen T, Persson I. Risk of myocardial infarction after oophorectomy and hysterectomy. J Clin Epidemiol. 2000;53:832-837.
22. Rocca WA, Grossardt BR, de Andrade M, Malkasian GD, Melton LJ, 3rd. Survival patterns after oophorectomy in premenopausal women: a population-based cohort study. Lancet Oncol. 2006;7:821-828.
23. Judd H, Judd G, Lucas W, Yen S. Endocrine function of the postmenopausal ovary: concentration of androgens and estrogens in ovarian and peripheral vein blood. J Clin Endocrinol Metab. 1974;39:1020-1024.
24. Fogle R, Stanczyk F, Zhang X, Paulson R. Ovarian androgen production in postmenopausal women. J Clin Endocrinol Metab. 2007;92:3040-3043.
25. Meldrum D, Davidson B, Tataryn I, Judd H. Changes in circulating steroids with aging in postmenopausal women. Obstet Gynecol. 1981;57:624-628.
26. Raisz LG, Wilta B, Artis A, et al. Comparison of the effects of estrogen alone and estrogen plus androgen on biochemical markers of bone formation and resorption in postmenopausal women. J Clin Endocrinol Metab. 1996;81:37-43.
27. Johansson C, Mellstrom D, Milsom I. Reproductive factors as predictors of bone density and fractures in women at age 70. Maturitas. 1993;17:39-50.
28. Davidson BJ, Ross RK, Paganini-Hill A, et al. Total and free estrogens and androgens in postmenopausal women with hip fractures. J Clin Endocrinol Metab. 1982;54:115-120.
29. Cummings SR, Browner WS, Bauer D, et al. Endogenous hormones and the risk of hip and vertebral fractures among older women. Study of Osteoporotic Fractures Research Group. N Engl J Med. 1998;339:733-738.
30. Kritz-Silverstein D, von Mühlen D, Barrett-Connor E. Hysterectomy and oophorectomy are unrelated to bone loss in older women. Maturitas. 2004;47:61-69.
31. Antoniucci DM, Sellmeyer DE, Cauley JA, et al. Postmenopausal bilateral oophorectomy is not associated with increased fracture risk in older women. J Bone Miner Res. 2005;20:741-747.
32. Keene GS, Parker MJ, Pryor GA. Mortality and morbidity after hip fractures. BMJ. 1993;307:1248-1250.
33. Nathorst-Boos J, von Schoultz B, Carlstrom K. Elective ovarian removal and estrogen replacement therapy—effects on sexual life, psychological well-being and androgen status. J Psychosom Obstet Gynaecol. 1993;14:283-293.
34. Nieman L. Management of surgically hypogonadal patients unable to take sex hormone replacement therapy. Endocrinol Metab Clin North Am. 2003;32:325-336.
35. Shifren J. Androgen deficiency in the oophorectomized woman. Fertil Steril. 2002;77 Suppl 4:s60-s62.
36. Sherwin B, Gelfan M. The role of androgens in the maintenance of sexual functioning in oophorectomized women. Psychosom Med. 1978;49:397-409.
37. Shifren JL, Avis NE. Surgical menopause: effects on psychological well-being and sexuality. Menopause. 2007;14:586.-
38. Elit L, Esplen MJ, Butler K, Narod S. Quality of life and psychosexual adjustment after prophylactic oophorectomy for a family history of ovarian cancer. Fam Cancer. 2001;1:149-156.
39. Sherwin BB, Gelfan MM. The role of androgens in the maintenance of sexual functioning in oophorectomized women. Psychosom Med 1978;49:397-409.
40. Teplin V, Vittinghoff E, Lin F, Learman L, Richter H, Kuppermann M. Oophorectomy in premenopausal women: health-related quality of life and sexual functioning. Obstet Gynecol. 2007;109:347-354.
41. Shumaker SA, Legault C, Rapp SR, et al. WHIMS Investigators Estrogen plus progestin and the incidence of dementia and mild cognitive impairment in postmenopausal women: the Women’s Health Initiative Memory Study: a randomized controlled trial. JAMA. 2003;289:2651-2662.
42. Rapp SR, Espeland MA, Shumaker SA, et al. WHIMS Investigators. Effect of estrogen plus progestin on global cognitive function in postmenopausal women: the Women’s Health Initiative Memory Study: a randomized controlled trial. JAMA. 2003;289:2663-2672.
43. Espeland MA, Rapp SR, Shumaker SA, et al. Conjugated equine estrogens and global cognitive function in postmenopausal women: Women’s Health Initiative Memory Study. JAMA. 2004;291:2959-2968.
44. Shumaker SA, Legault C, Kuller L, et al. Conjugated equine estrogens and incidence of probable dementia and mild cognitive impairment in postmenopausal women: Women’s Health Initiative Memory Study. JAMA. 2004;291:2947-2958.
45. Siegfried T. Neuroscience: it’s all in the timing. Nature. 2007;445:359-361.
46. Parker W, Broder M, Liu Z, Shoupe D, Farquhar C, Berek J. Ovarian conservation at the time of hysterectomy for benign disease. Obstet Gynecol. 2005;106:219-226.
47. Buist DS, Newton KM, Miglioretti DL, et al. Hormone therapy prescribing patterns in the United States. Obstet Gynecol. 2004;104:1042-1050.
48. McCombs JS, Thiebaud P, McLaughlin-Miley C, Shi J. Compliance with drug therapies for the treatment and prevention of osteoporosis. Maturitas. 2004;48:271-287.
49. Huser MA, Evans TS, Berger V. Medication adherence trends with statins. Adv Ther. 2005;22:163-171.
CASE: Hysterectomy candidate asks about her ovaries
A 51-year-old premenopausal woman complains of severe menorrhagia that often causes her to miss work. Although she is taking an iron supplement, her hemoglobin level often drops below 10 g/dL. She has already been identified as having fibroids, with a uterine size of 14 weeks. You order ultra-sonography, which reveals an enlarged uterus with multiple fibroids and normal endometrial thickness, but no intracavitary lesions.
After you describe the treatment options, including uterine artery embolization, the patient requests a hysterectomy as a reasonably low-risk means of cure. During informed consent, she asks whether she should have her ovaries removed during the surgery. Further discussion reveals that her father died of a myocardial infarction when he was 64 years old, but there is no family or personal history of ovarian or breast cancer.
How do you advise this patient, based on her history and recent findings from medical research?
Many gynecologists have been trained to recommend bilateral oophorectomy for women older than 45 or 50 years who request a hysterectomy for benign disease. In these women, oophorectomy is recommended to prevent ovarian cancer and avert the potential for other ovarian pathology that might require later surgery.
In the United States, 78% of women 45 to 64 years old and 55% of women overall undergo bilateral oophorectomy at the time of hysterectomy.1 These percentages mean that almost 300,000 women undergo bilateral oophorectomy each year.1
Hysterectomy alone can sometimes lead to early ovarian failure, but this phenomenon is infrequent. A prospective study of premenopausal women found that, after 5 years of follow-up, 20% of women who underwent simple hysterectomy reached menopause, compared with 7% of matched women who did not undergo hysterectomy.2
In this article, I explore the risks and benefits associated with bilateral oophorectomy and present an algorithm to aid in deciding whether the patient should keep her ovaries—and when oophorectomy might be a better option ( FIGURE ).
Among the hazards associated with bilateral oophorectomy are:
- an increased risk of death from coronary artery disease (CAD), lung cancer, all cancers (except ovarian), and all causes3,4
- an increased risk of osteoporosis and hip fracture5
- when performed before the onset of menopause, an increased risk of parkinsonism, cognitive impairment, dementia, anxiety, and depression.6-8
Benefits include a reduced risk of ovarian cancer, particularly among women who have a BRCA gene mutation or strong family history of ovarian or breast cancer.
Although ovarian cancer causes 15,000 deaths each year in the United States, that figure pales when compared with heart disease, which accounts for 350,000 deaths. In addition, hip fracture may cause approximately 66,000 deaths each year, and dementia attributable to bilateral oophorectomy may affect 100,000 to 200,000 women.9 Reoperation for adnexal pathology or pain after hysterectomy is rare, occurring in only 2.8% of women. Therefore, the benefits of oophorectomy are often outweighed by the risks of CAD, hip fracture, and neurologic conditions.
The assumption that medical treatment will ameliorate the risks associated with oophorectomy is unrealistic. Estrogen may mitigate some risks, but many women avoid hormone therapy. This avoidance can be especially problematic in young women.
In the Nurses’ Health Study, a separate analysis focused on women who had never used postmenopausal hormone therapy.4 In this analysis, all women who underwent bilateral oophorectomy had a greater risk of stroke (HR, 1.85; 95% CI, 1.09, 3.16) and lung cancer (HR, 2.09; 95% CI, 1.01, 4.33) than did women who retained their ovaries. Among women who underwent oophorectomy before age 50 and who did not take estrogen, the risk of coronary artery disease (CAD) was higher (HR, 1.98; 95% CI, 1.18, 3.32), as was the risk of death from all causes (HR, 1.40; 95% CI, 1.01, 1.96), compared with women who retained their ovaries.
Despite estrogen’s proven benefit among oophorectomized women, usage rates continue to decline. In the 6 months after publication of Women’s Health Initiative findings on estrogen–progestin therapy, the continuation rate of estrogen therapy decreased from 12.6% to 9.1%, and new starts also declined significantly.47
Among women who had a diagnosis of osteoporosis and who began treatment with estrogen, estrogen plus progestin, a bisphosphonate, or raloxifene, medication continuation rates were less than 25% at 12 months.48 Moreover, only 18% of women started on a statin to reduce the risk of cardiovascular disease were still taking the drug after 1 year.49

FIGURE Conservation vs oophorectomy: A guide to decision-making
* Estrogen replacement is recommended for women younger than 45 years who opt for oophorectomy
Ovarian cancer is a real, but relatively low, risk
In 2008, an estimated 21,650 new cases of ovarian cancer were diagnosed (age at diagnosis: mean, 63 years), and 15,520 women died from the disease.10 Because we lack a reliable screening test to detect early-stage ovarian cancer in the general population, most women are given a diagnosis when disease is advanced and the 5-year survival rate is 15% to 25%.
There is agreement that women who are known to have a BRCA mutation, which increases the risk of ovarian and breast cancer, should strongly consider oophorectomy once childbearing is complete.11 In the general population, however, the outlook is different.
In the United States, the lifetime risk of ovarian cancer is 1.4% overall. Among white women who have had three or more term pregnancies and who have used an oral contraceptive for at least 4 years, the lifetime risk of ovarian cancer drops to 0.3%.12
Need for reoperation is very low
The percentage of women who require reoperation after ovarian conservation—2.8%—may surprise you.13 That figure is lower than once thought because many studies were performed before asymptomatic, benign ovarian cysts were determined to be a fairly common phenomenon in postmenopausal women (prevalence, 6.6%). These cysts do not undergo transformation to cancer and, therefore, do not need to be removed.14
In addition, studies indicate that only 0.1% to 0.75% of women who retain their ovaries at the time of hysterectomy develop ovarian cancer.15,16 Therefore, the rationale of performing oophorectomy to avoid future surgery appears to be unfounded.
CAD risk rises sharply after oophorectomy
A recent systematic review found mixed evidence concerning the risk of CAD following bilateral salpingo-oophorectomy.17 In observational studies, however, earlier age of surgical or natural menopause has been associated with a higher risk of cardiovascular mortality.18-20 Early reports from the Nurses’ Health Study found that the risk of myocardial infarction doubled among women who underwent oophorectomy and never used estrogen, compared with age-matched premenopausal women (relative risk [RR], 2.2; 95% confidence interval [CI], 1.2, 4.2).3 Even after age 50, the risk of a first myocardial infarction is increased among oophorectomized women, compared with women who retain their ovaries (RR, 1.4; 95% CI, 1.0–2.0).21
A study by researchers from the Mayo Clinic, who examined all causes listed on the death certificate, found a significant association between bilateral oophorectomy before the age of 45 years and cardiovascular mortality (hazard ratio [HR], 1.44; 95% CI, 1.01–2.05).22 This risk was significantly increased among women who were not treated with estrogen through at least age 45, compared with estrogen-treated women.
Oophorectomy may impair bone health
After menopause, ovaries continue to produce significant amounts of the androgens testosterone and androstenedione, which are converted to estrone peripherally by skin, muscle, and fat cells.23,24 The levels of these hormones remain consistent and have been documented to age 80.25
Both estrogens and androgens inhibit bone resorption, and androgens also stimulate bone formation.26 Low levels of androgens and estrogens are linked to lower bone density and a higher risk of hip and vertebral fracture in postmenopausal women.27-29
Postmenopausal women who have been oophorectomized may have an even greater risk of osteoporosis. Over 16 years of follow-up, 340 women who had undergone oophorectomy at a median age of 62 years had 54% more osteoporotic fractures than women who had intact ovaries.5 Two other studies found no association between oophorectomy and bone loss or fracture risk, however.30,31
Hip fracture is a well-documented cause of increased morbidity and mortality in older women. One study found that, before hip fracture, 28% of patients were housebound; 1 year after hip fracture, the percentage was 46%.32 Women older than 60 who underwent oophorectomy had a doubled risk of mortality after low-trauma hip fracture, compared with women who had intact ovaries (odds ratio [OR], 2.18; 95% CI, 2.03–2.32).5
Loss of ovaries may affect mental health and sexuality
In a premenopausal woman, oophorectomy causes a sudden loss of estrogen and often triggers hot flashes, mood changes, sleep disturbances, headaches, and a decline in feelings of well-being.33,34 Over time, vaginal dryness, painful intercourse, loss of libido, bladder dysfunction, and depression may occur.35,36
Evidence suggests that, in women, sexual desire, sexual sensation, and orgasmic response are influenced by androgens. After elective oophorectomy, declines in sexual desire have been reported.37-39
Mental health and sexuality may rebound over time, however. One study found less improvement in mental health measures and body image 6 months after hysterectomy among women who were oophorectomized, compared with those who retained their ovaries. After 2 years, improvement levels were similar between groups.40
Cognitive function may suffer
Analysis of data from the Mayo Clinic Cohort Study of Oophorectomy and Aging found that bilateral oophorectomy before the onset of menopause increased the risk of parkinsonism, cognitive impairment or dementia, and anxiety or depression, particularly if estrogen was not replaced.6-8 These risks increased with younger age at oophorectomy.
The Women’s Health Initiative found an increased risk of dementia or mild cognitive impairment in women who were treated by estrogen alone or estrogen plus progestin after age 65.41-44
These disparate conclusions suggest that estrogen may have a protective effect on the brain if it is given right after natural menopause or premenopausal oophorectomy, but deleterious effects if it is started years later.45
Other studies of endogenous estrogen and cognitive function are few and yield inconsistent results.
Ovarian conservation boosts long-term survival
When there is no family history of ovarian cancer, ovarian conservation appears to maximize survival among healthy women 40 to 65 years old who undergo hysterectomy for benign disease.46 Among healthy women hysterectomized before the age of 55, calculations suggest that 8.6% more would be alive at age 80 if their ovaries were conserved than if they were removed.46
A study from the Mayo Clinic found that all-cause mortality was significantly higher among women who underwent prophylactic bilateral oophorectomy before the age of 45 than it was among women in the control group (HR, 1.67; 95% CI, 1.16–2.40); it was particularly high in women who did not receive estrogen treatment before age 45 (HR, 1.93; 95% CI, 1.25–2.96).22
In a recent study, investigators used the Nurses’ Health Study database to explore the long-term health outcomes of 29,380 women who underwent hysterectomy.4 Of these women, 13,035 (44.4%) had their ovaries conserved, and 16,345 (55.6%) underwent bilateral oophorectomy. Follow-up was 24 years. Oophorectomy was associated with an increased risk of nonfatal CAD among all women (HR, 1.17; 95% CI, 1.02, 1.35), especially those who underwent the procedure before age 45 (HR, 1.26; 95% CI, 1.04, 1.54). Oophorectomy was associated with a markedly reduced risk of ovarian cancer but an increased risk of lung cancer (HR, 1.26; 95% CI, 1.02–1.56).
In regard to fatal events, oophorectomy increased the risk of death from all causes (HR, 1.12; 95% CI, 1.03, 1.21). Specifically, there was an increased risk of death from CAD (HR, 1.28; 95% CI, 1.00, 1.64), lung cancer (HR, 1.31; 95% CI, 1.02, 1.68), and all cancers (HR, 1.17; 95% CI, 1.04, 1.32). There was no overall difference in the risk of death from stroke, breast cancer, and colorectal cancer between women who underwent oophorectomy and those who retained their ovaries.
During the 24 years of follow-up, 37 women died from ovarian cancer, accounting for 1.2% of all deaths. At no age did oophorectomy show a survival benefit.
How this evidence should inform your practice
It is unfortunate that the entire body of evidence on the risks and benefits of bilateral salpingo-oophorectomy consists of observational studies, which have significant inherent limitations. Although the Nurses’ Health Study was the largest prospective study to examine the effect of oophorectomy on women’s health, and involved the longest follow-up, the study was observational, and oophorectomy and ovarian conservation were self-selected. Nevertheless, recent data suggest that a more detailed informed-consent process is warranted than the process in place. Informed consent should cover the risks and benefits of both oophorectomy and ovarian conservation.
Prophylactic oophorectomy is recommended only if a preponderance of the evidence establishes that it clearly benefits the patient. The studies described in this article suggest that bilateral oophorectomy does harm more often than it does good. Therefore, a cautious approach to oophorectomy at the time of hysterectomy is advised.
CASE RESOLVED
After you describe the risks and benefits of oophorectomy, and address the patient’s concerns about her family history of heart disease, she decides to keep her ovaries.
CASE: Hysterectomy candidate asks about her ovaries
A 51-year-old premenopausal woman complains of severe menorrhagia that often causes her to miss work. Although she is taking an iron supplement, her hemoglobin level often drops below 10 g/dL. She has already been identified as having fibroids, with a uterine size of 14 weeks. You order ultra-sonography, which reveals an enlarged uterus with multiple fibroids and normal endometrial thickness, but no intracavitary lesions.
After you describe the treatment options, including uterine artery embolization, the patient requests a hysterectomy as a reasonably low-risk means of cure. During informed consent, she asks whether she should have her ovaries removed during the surgery. Further discussion reveals that her father died of a myocardial infarction when he was 64 years old, but there is no family or personal history of ovarian or breast cancer.
How do you advise this patient, based on her history and recent findings from medical research?
Many gynecologists have been trained to recommend bilateral oophorectomy for women older than 45 or 50 years who request a hysterectomy for benign disease. In these women, oophorectomy is recommended to prevent ovarian cancer and avert the potential for other ovarian pathology that might require later surgery.
In the United States, 78% of women 45 to 64 years old and 55% of women overall undergo bilateral oophorectomy at the time of hysterectomy.1 These percentages mean that almost 300,000 women undergo bilateral oophorectomy each year.1
Hysterectomy alone can sometimes lead to early ovarian failure, but this phenomenon is infrequent. A prospective study of premenopausal women found that, after 5 years of follow-up, 20% of women who underwent simple hysterectomy reached menopause, compared with 7% of matched women who did not undergo hysterectomy.2
In this article, I explore the risks and benefits associated with bilateral oophorectomy and present an algorithm to aid in deciding whether the patient should keep her ovaries—and when oophorectomy might be a better option ( FIGURE ).
Among the hazards associated with bilateral oophorectomy are:
- an increased risk of death from coronary artery disease (CAD), lung cancer, all cancers (except ovarian), and all causes3,4
- an increased risk of osteoporosis and hip fracture5
- when performed before the onset of menopause, an increased risk of parkinsonism, cognitive impairment, dementia, anxiety, and depression.6-8
Benefits include a reduced risk of ovarian cancer, particularly among women who have a BRCA gene mutation or strong family history of ovarian or breast cancer.
Although ovarian cancer causes 15,000 deaths each year in the United States, that figure pales when compared with heart disease, which accounts for 350,000 deaths. In addition, hip fracture may cause approximately 66,000 deaths each year, and dementia attributable to bilateral oophorectomy may affect 100,000 to 200,000 women.9 Reoperation for adnexal pathology or pain after hysterectomy is rare, occurring in only 2.8% of women. Therefore, the benefits of oophorectomy are often outweighed by the risks of CAD, hip fracture, and neurologic conditions.
The assumption that medical treatment will ameliorate the risks associated with oophorectomy is unrealistic. Estrogen may mitigate some risks, but many women avoid hormone therapy. This avoidance can be especially problematic in young women.
In the Nurses’ Health Study, a separate analysis focused on women who had never used postmenopausal hormone therapy.4 In this analysis, all women who underwent bilateral oophorectomy had a greater risk of stroke (HR, 1.85; 95% CI, 1.09, 3.16) and lung cancer (HR, 2.09; 95% CI, 1.01, 4.33) than did women who retained their ovaries. Among women who underwent oophorectomy before age 50 and who did not take estrogen, the risk of coronary artery disease (CAD) was higher (HR, 1.98; 95% CI, 1.18, 3.32), as was the risk of death from all causes (HR, 1.40; 95% CI, 1.01, 1.96), compared with women who retained their ovaries.
Despite estrogen’s proven benefit among oophorectomized women, usage rates continue to decline. In the 6 months after publication of Women’s Health Initiative findings on estrogen–progestin therapy, the continuation rate of estrogen therapy decreased from 12.6% to 9.1%, and new starts also declined significantly.47
Among women who had a diagnosis of osteoporosis and who began treatment with estrogen, estrogen plus progestin, a bisphosphonate, or raloxifene, medication continuation rates were less than 25% at 12 months.48 Moreover, only 18% of women started on a statin to reduce the risk of cardiovascular disease were still taking the drug after 1 year.49

FIGURE Conservation vs oophorectomy: A guide to decision-making
* Estrogen replacement is recommended for women younger than 45 years who opt for oophorectomy
Ovarian cancer is a real, but relatively low, risk
In 2008, an estimated 21,650 new cases of ovarian cancer were diagnosed (age at diagnosis: mean, 63 years), and 15,520 women died from the disease.10 Because we lack a reliable screening test to detect early-stage ovarian cancer in the general population, most women are given a diagnosis when disease is advanced and the 5-year survival rate is 15% to 25%.
There is agreement that women who are known to have a BRCA mutation, which increases the risk of ovarian and breast cancer, should strongly consider oophorectomy once childbearing is complete.11 In the general population, however, the outlook is different.
In the United States, the lifetime risk of ovarian cancer is 1.4% overall. Among white women who have had three or more term pregnancies and who have used an oral contraceptive for at least 4 years, the lifetime risk of ovarian cancer drops to 0.3%.12
Need for reoperation is very low
The percentage of women who require reoperation after ovarian conservation—2.8%—may surprise you.13 That figure is lower than once thought because many studies were performed before asymptomatic, benign ovarian cysts were determined to be a fairly common phenomenon in postmenopausal women (prevalence, 6.6%). These cysts do not undergo transformation to cancer and, therefore, do not need to be removed.14
In addition, studies indicate that only 0.1% to 0.75% of women who retain their ovaries at the time of hysterectomy develop ovarian cancer.15,16 Therefore, the rationale of performing oophorectomy to avoid future surgery appears to be unfounded.
CAD risk rises sharply after oophorectomy
A recent systematic review found mixed evidence concerning the risk of CAD following bilateral salpingo-oophorectomy.17 In observational studies, however, earlier age of surgical or natural menopause has been associated with a higher risk of cardiovascular mortality.18-20 Early reports from the Nurses’ Health Study found that the risk of myocardial infarction doubled among women who underwent oophorectomy and never used estrogen, compared with age-matched premenopausal women (relative risk [RR], 2.2; 95% confidence interval [CI], 1.2, 4.2).3 Even after age 50, the risk of a first myocardial infarction is increased among oophorectomized women, compared with women who retain their ovaries (RR, 1.4; 95% CI, 1.0–2.0).21
A study by researchers from the Mayo Clinic, who examined all causes listed on the death certificate, found a significant association between bilateral oophorectomy before the age of 45 years and cardiovascular mortality (hazard ratio [HR], 1.44; 95% CI, 1.01–2.05).22 This risk was significantly increased among women who were not treated with estrogen through at least age 45, compared with estrogen-treated women.
Oophorectomy may impair bone health
After menopause, ovaries continue to produce significant amounts of the androgens testosterone and androstenedione, which are converted to estrone peripherally by skin, muscle, and fat cells.23,24 The levels of these hormones remain consistent and have been documented to age 80.25
Both estrogens and androgens inhibit bone resorption, and androgens also stimulate bone formation.26 Low levels of androgens and estrogens are linked to lower bone density and a higher risk of hip and vertebral fracture in postmenopausal women.27-29
Postmenopausal women who have been oophorectomized may have an even greater risk of osteoporosis. Over 16 years of follow-up, 340 women who had undergone oophorectomy at a median age of 62 years had 54% more osteoporotic fractures than women who had intact ovaries.5 Two other studies found no association between oophorectomy and bone loss or fracture risk, however.30,31
Hip fracture is a well-documented cause of increased morbidity and mortality in older women. One study found that, before hip fracture, 28% of patients were housebound; 1 year after hip fracture, the percentage was 46%.32 Women older than 60 who underwent oophorectomy had a doubled risk of mortality after low-trauma hip fracture, compared with women who had intact ovaries (odds ratio [OR], 2.18; 95% CI, 2.03–2.32).5
Loss of ovaries may affect mental health and sexuality
In a premenopausal woman, oophorectomy causes a sudden loss of estrogen and often triggers hot flashes, mood changes, sleep disturbances, headaches, and a decline in feelings of well-being.33,34 Over time, vaginal dryness, painful intercourse, loss of libido, bladder dysfunction, and depression may occur.35,36
Evidence suggests that, in women, sexual desire, sexual sensation, and orgasmic response are influenced by androgens. After elective oophorectomy, declines in sexual desire have been reported.37-39
Mental health and sexuality may rebound over time, however. One study found less improvement in mental health measures and body image 6 months after hysterectomy among women who were oophorectomized, compared with those who retained their ovaries. After 2 years, improvement levels were similar between groups.40
Cognitive function may suffer
Analysis of data from the Mayo Clinic Cohort Study of Oophorectomy and Aging found that bilateral oophorectomy before the onset of menopause increased the risk of parkinsonism, cognitive impairment or dementia, and anxiety or depression, particularly if estrogen was not replaced.6-8 These risks increased with younger age at oophorectomy.
The Women’s Health Initiative found an increased risk of dementia or mild cognitive impairment in women who were treated by estrogen alone or estrogen plus progestin after age 65.41-44
These disparate conclusions suggest that estrogen may have a protective effect on the brain if it is given right after natural menopause or premenopausal oophorectomy, but deleterious effects if it is started years later.45
Other studies of endogenous estrogen and cognitive function are few and yield inconsistent results.
Ovarian conservation boosts long-term survival
When there is no family history of ovarian cancer, ovarian conservation appears to maximize survival among healthy women 40 to 65 years old who undergo hysterectomy for benign disease.46 Among healthy women hysterectomized before the age of 55, calculations suggest that 8.6% more would be alive at age 80 if their ovaries were conserved than if they were removed.46
A study from the Mayo Clinic found that all-cause mortality was significantly higher among women who underwent prophylactic bilateral oophorectomy before the age of 45 than it was among women in the control group (HR, 1.67; 95% CI, 1.16–2.40); it was particularly high in women who did not receive estrogen treatment before age 45 (HR, 1.93; 95% CI, 1.25–2.96).22
In a recent study, investigators used the Nurses’ Health Study database to explore the long-term health outcomes of 29,380 women who underwent hysterectomy.4 Of these women, 13,035 (44.4%) had their ovaries conserved, and 16,345 (55.6%) underwent bilateral oophorectomy. Follow-up was 24 years. Oophorectomy was associated with an increased risk of nonfatal CAD among all women (HR, 1.17; 95% CI, 1.02, 1.35), especially those who underwent the procedure before age 45 (HR, 1.26; 95% CI, 1.04, 1.54). Oophorectomy was associated with a markedly reduced risk of ovarian cancer but an increased risk of lung cancer (HR, 1.26; 95% CI, 1.02–1.56).
In regard to fatal events, oophorectomy increased the risk of death from all causes (HR, 1.12; 95% CI, 1.03, 1.21). Specifically, there was an increased risk of death from CAD (HR, 1.28; 95% CI, 1.00, 1.64), lung cancer (HR, 1.31; 95% CI, 1.02, 1.68), and all cancers (HR, 1.17; 95% CI, 1.04, 1.32). There was no overall difference in the risk of death from stroke, breast cancer, and colorectal cancer between women who underwent oophorectomy and those who retained their ovaries.
During the 24 years of follow-up, 37 women died from ovarian cancer, accounting for 1.2% of all deaths. At no age did oophorectomy show a survival benefit.
How this evidence should inform your practice
It is unfortunate that the entire body of evidence on the risks and benefits of bilateral salpingo-oophorectomy consists of observational studies, which have significant inherent limitations. Although the Nurses’ Health Study was the largest prospective study to examine the effect of oophorectomy on women’s health, and involved the longest follow-up, the study was observational, and oophorectomy and ovarian conservation were self-selected. Nevertheless, recent data suggest that a more detailed informed-consent process is warranted than the process in place. Informed consent should cover the risks and benefits of both oophorectomy and ovarian conservation.
Prophylactic oophorectomy is recommended only if a preponderance of the evidence establishes that it clearly benefits the patient. The studies described in this article suggest that bilateral oophorectomy does harm more often than it does good. Therefore, a cautious approach to oophorectomy at the time of hysterectomy is advised.
CASE RESOLVED
After you describe the risks and benefits of oophorectomy, and address the patient’s concerns about her family history of heart disease, she decides to keep her ovaries.
1. Healthcare Cost and Utilization Project (HCUP), 1988–2001: A Federal–State–Industry Partnership in Health Data. Rockville, Md: Agency for Healthcare Research and Quality; July 2003.
2. Farquhar CM, Sadler L, Harvey SA, Stewart AW. The association of hysterectomy and menopause: a prospective cohort study. BJOG. 2005;112:956-962.
3. Colditz G, Willett W, Stampfer M, Rosner B, Speizer F, Hennekens C. Menopause and the risk of coronary heart disease in women. N Engl J Med. 1987;316:1105-1110.
4. Parker WH, Broder MS, Chang E, et al. Ovarian conservation at the time of hysterectomy and long-term health outcomes in the Nurses’ Health Study. Obstet Gynecol. 2009;113:1027-1037.
5. Melton LJ 3rd, Khosla S, Malkasian GD, Achenbach SJ, Oberg AL, Riggs BL. Fracture risk after bilateral oophorectomy in elderly women. J Bone Miner Res. 2003;18:900-905.
6. Rocca WA, Bower JH, Maraganore DM, et al. Increased risk of cognitive impairment or dementia in women who underwent oophorectomy before menopause. Neurology. 2007;69:1074.-
7. Rocca WA, Bower JH, Maraganore DM, et al. Increased risk of parkinsonism in women who underwent oophorectomy before menopause. Neurology. 2008;70:200-209.
8. Rocca WA, Grossardt BR, Geda YE, et al. Long-term risk of depressive and anxiety symptoms after early bilateral oophorectomy. Menopause. 2008;15:1050-1059.
9. Bennett DA. Editorial comment on “Prevalence of dementia in the United States: the aging, demographics, and memory study” by Plassman et al. Neuroepidemiology. 2007;29:133-135.
10. Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58:71-96.
11. Armstrong K, Schwartz J, Randall T, Rubin S, Weber B. Hormone replacement therapy and life expectancy after prophylactic oophorectomy in women with BRCA1/2 mutations: a decision analysis. J Clin Oncol. 2004;22:1045-1054.
12. Hartge P, Whittemore AS, Itnyre J, McGowan L, Cramer D. Rates and risks of ovarian cancer in subgroups of white women in the United States. The Collaborative Ovarian Cancer Group. Obstet Gynecol. 1994;84:760-764.
13. Dekel A, Efrat Z, Orvieto R, et al. The residual ovary syndrome: a 20-year experience. Eur J Obstet Gynecol Reprod Biol. 1996;68:159-164.
14. Bailey CL, Ueland FR, Land GL, et al. The malignant potential of small cystic ovarian tumors in women over 50 years of age. Gynecol Oncol. 1998;69:3-7.
15. Naylor AC. Hysterectomy—analysis of 2,901 personally performed procedures. S Afr Med J. 1984;65:242-245.
16. Christ JE, Lotze EC. The residual ovary syndrome. Obstet Gynecol. 1975;46:551-556.
17. Jacoby VL, Grady D, Sawaya GF. Oophorectomy as a risk factor for coronary heart disease. Am J Obstet Gynecol. 2009;200:140.e1-140.e9.
18. van der Schouw YT, van der Graaf Y, Steyerberg EW, Eijkemans JC, Banga JD. Age at menopause as a risk factor for cardiovascular mortality. Lancet. 1996;347:714-718.
19. Løkkegaard E, Jovanovic Z, Heitmann BL, et al. The association between early menopause and risk of ischaemic heart disease: influence of hormone therapy. Maturitas. 2006;53:226-233.
20. de Kleijn MJJ, van der Schouw YT, Verbeek ALM, et al. Endogenous estrogen exposure and cardiovascular mortality risk in postmenopausal women. Am J Epidemiol. 2002;155:339.-
21. Falkeborn M, Schairer C, Naessen T, Persson I. Risk of myocardial infarction after oophorectomy and hysterectomy. J Clin Epidemiol. 2000;53:832-837.
22. Rocca WA, Grossardt BR, de Andrade M, Malkasian GD, Melton LJ, 3rd. Survival patterns after oophorectomy in premenopausal women: a population-based cohort study. Lancet Oncol. 2006;7:821-828.
23. Judd H, Judd G, Lucas W, Yen S. Endocrine function of the postmenopausal ovary: concentration of androgens and estrogens in ovarian and peripheral vein blood. J Clin Endocrinol Metab. 1974;39:1020-1024.
24. Fogle R, Stanczyk F, Zhang X, Paulson R. Ovarian androgen production in postmenopausal women. J Clin Endocrinol Metab. 2007;92:3040-3043.
25. Meldrum D, Davidson B, Tataryn I, Judd H. Changes in circulating steroids with aging in postmenopausal women. Obstet Gynecol. 1981;57:624-628.
26. Raisz LG, Wilta B, Artis A, et al. Comparison of the effects of estrogen alone and estrogen plus androgen on biochemical markers of bone formation and resorption in postmenopausal women. J Clin Endocrinol Metab. 1996;81:37-43.
27. Johansson C, Mellstrom D, Milsom I. Reproductive factors as predictors of bone density and fractures in women at age 70. Maturitas. 1993;17:39-50.
28. Davidson BJ, Ross RK, Paganini-Hill A, et al. Total and free estrogens and androgens in postmenopausal women with hip fractures. J Clin Endocrinol Metab. 1982;54:115-120.
29. Cummings SR, Browner WS, Bauer D, et al. Endogenous hormones and the risk of hip and vertebral fractures among older women. Study of Osteoporotic Fractures Research Group. N Engl J Med. 1998;339:733-738.
30. Kritz-Silverstein D, von Mühlen D, Barrett-Connor E. Hysterectomy and oophorectomy are unrelated to bone loss in older women. Maturitas. 2004;47:61-69.
31. Antoniucci DM, Sellmeyer DE, Cauley JA, et al. Postmenopausal bilateral oophorectomy is not associated with increased fracture risk in older women. J Bone Miner Res. 2005;20:741-747.
32. Keene GS, Parker MJ, Pryor GA. Mortality and morbidity after hip fractures. BMJ. 1993;307:1248-1250.
33. Nathorst-Boos J, von Schoultz B, Carlstrom K. Elective ovarian removal and estrogen replacement therapy—effects on sexual life, psychological well-being and androgen status. J Psychosom Obstet Gynaecol. 1993;14:283-293.
34. Nieman L. Management of surgically hypogonadal patients unable to take sex hormone replacement therapy. Endocrinol Metab Clin North Am. 2003;32:325-336.
35. Shifren J. Androgen deficiency in the oophorectomized woman. Fertil Steril. 2002;77 Suppl 4:s60-s62.
36. Sherwin B, Gelfan M. The role of androgens in the maintenance of sexual functioning in oophorectomized women. Psychosom Med. 1978;49:397-409.
37. Shifren JL, Avis NE. Surgical menopause: effects on psychological well-being and sexuality. Menopause. 2007;14:586.-
38. Elit L, Esplen MJ, Butler K, Narod S. Quality of life and psychosexual adjustment after prophylactic oophorectomy for a family history of ovarian cancer. Fam Cancer. 2001;1:149-156.
39. Sherwin BB, Gelfan MM. The role of androgens in the maintenance of sexual functioning in oophorectomized women. Psychosom Med 1978;49:397-409.
40. Teplin V, Vittinghoff E, Lin F, Learman L, Richter H, Kuppermann M. Oophorectomy in premenopausal women: health-related quality of life and sexual functioning. Obstet Gynecol. 2007;109:347-354.
41. Shumaker SA, Legault C, Rapp SR, et al. WHIMS Investigators Estrogen plus progestin and the incidence of dementia and mild cognitive impairment in postmenopausal women: the Women’s Health Initiative Memory Study: a randomized controlled trial. JAMA. 2003;289:2651-2662.
42. Rapp SR, Espeland MA, Shumaker SA, et al. WHIMS Investigators. Effect of estrogen plus progestin on global cognitive function in postmenopausal women: the Women’s Health Initiative Memory Study: a randomized controlled trial. JAMA. 2003;289:2663-2672.
43. Espeland MA, Rapp SR, Shumaker SA, et al. Conjugated equine estrogens and global cognitive function in postmenopausal women: Women’s Health Initiative Memory Study. JAMA. 2004;291:2959-2968.
44. Shumaker SA, Legault C, Kuller L, et al. Conjugated equine estrogens and incidence of probable dementia and mild cognitive impairment in postmenopausal women: Women’s Health Initiative Memory Study. JAMA. 2004;291:2947-2958.
45. Siegfried T. Neuroscience: it’s all in the timing. Nature. 2007;445:359-361.
46. Parker W, Broder M, Liu Z, Shoupe D, Farquhar C, Berek J. Ovarian conservation at the time of hysterectomy for benign disease. Obstet Gynecol. 2005;106:219-226.
47. Buist DS, Newton KM, Miglioretti DL, et al. Hormone therapy prescribing patterns in the United States. Obstet Gynecol. 2004;104:1042-1050.
48. McCombs JS, Thiebaud P, McLaughlin-Miley C, Shi J. Compliance with drug therapies for the treatment and prevention of osteoporosis. Maturitas. 2004;48:271-287.
49. Huser MA, Evans TS, Berger V. Medication adherence trends with statins. Adv Ther. 2005;22:163-171.
1. Healthcare Cost and Utilization Project (HCUP), 1988–2001: A Federal–State–Industry Partnership in Health Data. Rockville, Md: Agency for Healthcare Research and Quality; July 2003.
2. Farquhar CM, Sadler L, Harvey SA, Stewart AW. The association of hysterectomy and menopause: a prospective cohort study. BJOG. 2005;112:956-962.
3. Colditz G, Willett W, Stampfer M, Rosner B, Speizer F, Hennekens C. Menopause and the risk of coronary heart disease in women. N Engl J Med. 1987;316:1105-1110.
4. Parker WH, Broder MS, Chang E, et al. Ovarian conservation at the time of hysterectomy and long-term health outcomes in the Nurses’ Health Study. Obstet Gynecol. 2009;113:1027-1037.
5. Melton LJ 3rd, Khosla S, Malkasian GD, Achenbach SJ, Oberg AL, Riggs BL. Fracture risk after bilateral oophorectomy in elderly women. J Bone Miner Res. 2003;18:900-905.
6. Rocca WA, Bower JH, Maraganore DM, et al. Increased risk of cognitive impairment or dementia in women who underwent oophorectomy before menopause. Neurology. 2007;69:1074.-
7. Rocca WA, Bower JH, Maraganore DM, et al. Increased risk of parkinsonism in women who underwent oophorectomy before menopause. Neurology. 2008;70:200-209.
8. Rocca WA, Grossardt BR, Geda YE, et al. Long-term risk of depressive and anxiety symptoms after early bilateral oophorectomy. Menopause. 2008;15:1050-1059.
9. Bennett DA. Editorial comment on “Prevalence of dementia in the United States: the aging, demographics, and memory study” by Plassman et al. Neuroepidemiology. 2007;29:133-135.
10. Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58:71-96.
11. Armstrong K, Schwartz J, Randall T, Rubin S, Weber B. Hormone replacement therapy and life expectancy after prophylactic oophorectomy in women with BRCA1/2 mutations: a decision analysis. J Clin Oncol. 2004;22:1045-1054.
12. Hartge P, Whittemore AS, Itnyre J, McGowan L, Cramer D. Rates and risks of ovarian cancer in subgroups of white women in the United States. The Collaborative Ovarian Cancer Group. Obstet Gynecol. 1994;84:760-764.
13. Dekel A, Efrat Z, Orvieto R, et al. The residual ovary syndrome: a 20-year experience. Eur J Obstet Gynecol Reprod Biol. 1996;68:159-164.
14. Bailey CL, Ueland FR, Land GL, et al. The malignant potential of small cystic ovarian tumors in women over 50 years of age. Gynecol Oncol. 1998;69:3-7.
15. Naylor AC. Hysterectomy—analysis of 2,901 personally performed procedures. S Afr Med J. 1984;65:242-245.
16. Christ JE, Lotze EC. The residual ovary syndrome. Obstet Gynecol. 1975;46:551-556.
17. Jacoby VL, Grady D, Sawaya GF. Oophorectomy as a risk factor for coronary heart disease. Am J Obstet Gynecol. 2009;200:140.e1-140.e9.
18. van der Schouw YT, van der Graaf Y, Steyerberg EW, Eijkemans JC, Banga JD. Age at menopause as a risk factor for cardiovascular mortality. Lancet. 1996;347:714-718.
19. Løkkegaard E, Jovanovic Z, Heitmann BL, et al. The association between early menopause and risk of ischaemic heart disease: influence of hormone therapy. Maturitas. 2006;53:226-233.
20. de Kleijn MJJ, van der Schouw YT, Verbeek ALM, et al. Endogenous estrogen exposure and cardiovascular mortality risk in postmenopausal women. Am J Epidemiol. 2002;155:339.-
21. Falkeborn M, Schairer C, Naessen T, Persson I. Risk of myocardial infarction after oophorectomy and hysterectomy. J Clin Epidemiol. 2000;53:832-837.
22. Rocca WA, Grossardt BR, de Andrade M, Malkasian GD, Melton LJ, 3rd. Survival patterns after oophorectomy in premenopausal women: a population-based cohort study. Lancet Oncol. 2006;7:821-828.
23. Judd H, Judd G, Lucas W, Yen S. Endocrine function of the postmenopausal ovary: concentration of androgens and estrogens in ovarian and peripheral vein blood. J Clin Endocrinol Metab. 1974;39:1020-1024.
24. Fogle R, Stanczyk F, Zhang X, Paulson R. Ovarian androgen production in postmenopausal women. J Clin Endocrinol Metab. 2007;92:3040-3043.
25. Meldrum D, Davidson B, Tataryn I, Judd H. Changes in circulating steroids with aging in postmenopausal women. Obstet Gynecol. 1981;57:624-628.
26. Raisz LG, Wilta B, Artis A, et al. Comparison of the effects of estrogen alone and estrogen plus androgen on biochemical markers of bone formation and resorption in postmenopausal women. J Clin Endocrinol Metab. 1996;81:37-43.
27. Johansson C, Mellstrom D, Milsom I. Reproductive factors as predictors of bone density and fractures in women at age 70. Maturitas. 1993;17:39-50.
28. Davidson BJ, Ross RK, Paganini-Hill A, et al. Total and free estrogens and androgens in postmenopausal women with hip fractures. J Clin Endocrinol Metab. 1982;54:115-120.
29. Cummings SR, Browner WS, Bauer D, et al. Endogenous hormones and the risk of hip and vertebral fractures among older women. Study of Osteoporotic Fractures Research Group. N Engl J Med. 1998;339:733-738.
30. Kritz-Silverstein D, von Mühlen D, Barrett-Connor E. Hysterectomy and oophorectomy are unrelated to bone loss in older women. Maturitas. 2004;47:61-69.
31. Antoniucci DM, Sellmeyer DE, Cauley JA, et al. Postmenopausal bilateral oophorectomy is not associated with increased fracture risk in older women. J Bone Miner Res. 2005;20:741-747.
32. Keene GS, Parker MJ, Pryor GA. Mortality and morbidity after hip fractures. BMJ. 1993;307:1248-1250.
33. Nathorst-Boos J, von Schoultz B, Carlstrom K. Elective ovarian removal and estrogen replacement therapy—effects on sexual life, psychological well-being and androgen status. J Psychosom Obstet Gynaecol. 1993;14:283-293.
34. Nieman L. Management of surgically hypogonadal patients unable to take sex hormone replacement therapy. Endocrinol Metab Clin North Am. 2003;32:325-336.
35. Shifren J. Androgen deficiency in the oophorectomized woman. Fertil Steril. 2002;77 Suppl 4:s60-s62.
36. Sherwin B, Gelfan M. The role of androgens in the maintenance of sexual functioning in oophorectomized women. Psychosom Med. 1978;49:397-409.
37. Shifren JL, Avis NE. Surgical menopause: effects on psychological well-being and sexuality. Menopause. 2007;14:586.-
38. Elit L, Esplen MJ, Butler K, Narod S. Quality of life and psychosexual adjustment after prophylactic oophorectomy for a family history of ovarian cancer. Fam Cancer. 2001;1:149-156.
39. Sherwin BB, Gelfan MM. The role of androgens in the maintenance of sexual functioning in oophorectomized women. Psychosom Med 1978;49:397-409.
40. Teplin V, Vittinghoff E, Lin F, Learman L, Richter H, Kuppermann M. Oophorectomy in premenopausal women: health-related quality of life and sexual functioning. Obstet Gynecol. 2007;109:347-354.
41. Shumaker SA, Legault C, Rapp SR, et al. WHIMS Investigators Estrogen plus progestin and the incidence of dementia and mild cognitive impairment in postmenopausal women: the Women’s Health Initiative Memory Study: a randomized controlled trial. JAMA. 2003;289:2651-2662.
42. Rapp SR, Espeland MA, Shumaker SA, et al. WHIMS Investigators. Effect of estrogen plus progestin on global cognitive function in postmenopausal women: the Women’s Health Initiative Memory Study: a randomized controlled trial. JAMA. 2003;289:2663-2672.
43. Espeland MA, Rapp SR, Shumaker SA, et al. Conjugated equine estrogens and global cognitive function in postmenopausal women: Women’s Health Initiative Memory Study. JAMA. 2004;291:2959-2968.
44. Shumaker SA, Legault C, Kuller L, et al. Conjugated equine estrogens and incidence of probable dementia and mild cognitive impairment in postmenopausal women: Women’s Health Initiative Memory Study. JAMA. 2004;291:2947-2958.
45. Siegfried T. Neuroscience: it’s all in the timing. Nature. 2007;445:359-361.
46. Parker W, Broder M, Liu Z, Shoupe D, Farquhar C, Berek J. Ovarian conservation at the time of hysterectomy for benign disease. Obstet Gynecol. 2005;106:219-226.
47. Buist DS, Newton KM, Miglioretti DL, et al. Hormone therapy prescribing patterns in the United States. Obstet Gynecol. 2004;104:1042-1050.
48. McCombs JS, Thiebaud P, McLaughlin-Miley C, Shi J. Compliance with drug therapies for the treatment and prevention of osteoporosis. Maturitas. 2004;48:271-287.
49. Huser MA, Evans TS, Berger V. Medication adherence trends with statins. Adv Ther. 2005;22:163-171.
UPDATE: FERTILITY
Infertility and its treatment can be a roller-coaster ride for patient and physician. Amid the emotional stress that arises, the goal of treatment can inadvertently shift from achievement of a successful singleton pregnancy to pregnancy at any cost—even high-order multiple gestation.
Here’s an essential question: Can the rate of multiple gestation be reduced without seriously compromising the pregnancy rate? Several developments of the past year suggest that it can be. In this article, we discuss:
- new guidelines that limit the number of embryos to be transferred at in vitro fertilization (IVF)
- strategies to reduce the risk of multiple gestation after controlled ovarian stimulation or ovulation induction
- the need to address the patient’s emotional status during treatment
- a new index that helps predict the pregnancy rate after surgical staging of endometriosis.
Multiple gestation is known to have adverse effects on infants, including a significantly elevated risk of prematurity and related physical and developmental problems. It also greatly increases the need for resources. And the high cost of caring for infants affected by prematurity further burdens an already overwhelmed health-care system.
Not only is it essential that we reduce the rate of high-order multiple gestation (i.e., more than two fetuses), but we should also attempt to lower the rate of twin pregnancy. A healthy singleton pregnancy, with its diminished risks and more reasonable health-care cost, should be our goal.
New guidelines limit the number of embryos to be transferred at IVF
Practice Committee of the American Society for Reproductive Medicine, Practice Committee of the Society for Assisted Reproductive Technology. Guidelines on number of embryos transferred. Fertil Steril. 2009;92:1518–1519.
Since the birth of Louise Brown in 1978, assisted reproductive technology (ART) has enjoyed dramatic technological advances. Intracytoplasmic sperm injection (FIGURE 1), preimplantation genetic diagnosis, and improvements in cryopreservation have broadened the application of ART and increased the live birth rate to 30% for every cycle that is initiated. The cumulative live birth rate from additional fresh and frozen-thawed cycles can reach 50% to 80%.
These gains have not come without cost, however. Multiple emotional, financial, and other variables affecting the practice of IVF have produced a higher-than-natural rate of multiple gestation.
In November 2009, the Society for Assisted Reproductive Technology (SART) and the American Society for Reproductive Medicine (ASRM) issued new guidelines limiting the number of embryos that should be transferred in one IVF cycle. IVF clinics are required to report outcomes, and approximately 93% of US cycles are reported to SART. High-order multiple-pregnancy rates are audited by SART, and outlier clinics must implement remediation programs to lower their high rate or risk expulsion from SART.
The increasing emphasis on single-embryo transfer in young women who have a good prognosis reflects the societies’ commitment to help patients achieve a healthy singleton pregnancy and good birth outcome.
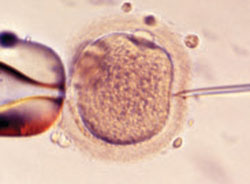
FIGURE 1 A wonder of technology
Intracytoplasmic sperm injection overcomes many barriers to fertilization, such as severe malefactor infertility. At some institutions, the technique yields a fertilization rate of 70% to 80%.
What makes a “good prognosis”?
Identification of patients who have a good prognosis is an essential component of these new guidelines. The patient is more likely to have a favorable outcome if one or more of the following is true:
- She is undergoing her first cycle of IVF
- The embryos have good morphology
- Excess embryos are available for cryopreservation
- She has had earlier success with IVF.
The TABLE details the recommended number of embryos to transfer, based on the age and prognosis of the patient. In cycles that involve a donor egg, base the number of embryos to be transferred on the age of the donor. In cycles that involve a frozen embryo, base the number of good-quality, thawed embryos to be transferred on the age of the patient at the time the embryos were created. One additional embryo may be transferred if the patient has a less favorable prognosis or a history of two failed, fresh IVF cycles.
Two important requisites: Careful counseling about the risk of high-order multiple gestation, and documentation of that counseling.
TABLE
SART and ASRM recommend limits on the number of embryos to be transferred at in vitro fertilization
| Prognosis | Age of patient (yr) | |||
|---|---|---|---|---|
| <35 | 35–37 | 38–40 | 41 and 42 | |
| CLEAVAGE-STAGE EMBRYOS* | ||||
| Favorable† | 1 or 2 | 2 | 3 | 5 |
| All others | 2 | 3 | 4 | 5 |
| BLASTOCYSTS* | ||||
| Favorable† | 1 | 2 | 2 | 3 |
| All others | 2 | 2 | 3 | 3 |
| * See text and guidelines for more complete explanations. Justification for transferring one additional embryo (above the recommended limit) should be clearly documented in the patient’s medical record. | ||||
| †Variables indicating favorable prognosis include first cycle of IVF, good embryo quality, availability of excess embryos for cryopreservation, and previous successful IVF cycle. | ||||
All IVF clinics must adhere to the new SART and ASRM guidelines limiting the number of embryos to transfer at in vitro fertilization. In addition, it is vital for you to counsel the patient about the risk of high-order multiple gestation, and to document that such counseling took place.
Judicious management can reduce the rate of multiple gestation in ovulation stimulation
Dickey RP. Strategies to reduce multiple pregnancies due to ovulation stimulation. Fertil Steril. 2009;91:1–17.
Efforts to reduce the rate of multiple gestation should focus not only on patients undergoing IVF but on those undergoing controlled ovarian stimulation (COS) or ovulation induction. In COS, pharmacologic treatment is used to stimulate the production of more than one oocyte. In ovulation induction, pharmacologic therapy is used to induce normal cycles in anovulatory or oligo-ovulatory women.1 A substantial majority of multiple gestations are conceived using ovarian stimulation and ovulation induction. These methods may be less difficult to manage than IVF because they are less dependent on technology. Like IVF, however, they carry a high risk of multiple gestation, especially high-order multiple gestation.2
Strategies to reduce multiple gestation
As Dickey points out in a comprehensive retrospective analysis, there are strategies that can help reduce multiple gestation during COS and ovulation induction. They include the following recommendations:
Be prepared to cancel a cycle. Initiate ovulation induction only if both patient and physician are prepared to cancel any cycle that involves an excessive number of preovulatory follicles. Singleton and twin births can be confidently expected only if the cycle is cancelled when there are more than two preovulatory follicles approximately 12 mm in diameter or larger. This may be psychologically difficult for some patients and doctors.
Preemptively identify risk factors for multiple gestation, including:
- seven or more preovulatory follicles
- an estradiol concentration of 1,000 pg/mL or higher
- early cycles of treatment (cycles 1–3)
- age younger than 32 years
- body mass index below 19 kg/m2
- use of donor sperm.
When any of these risk factors is present, consider starting the patient on a lower initial dosage of gonadotropin; perform more frequent monitoring; maintain a low threshold for cancellation; and consider performing IVF with single-embryo transfer rather than COS.
Use specific drugs. Increase the likelihood of monofollicular development and double-follicular recruitment and reduce the risk of high-order multiple gestation by using clomiphene citrate, a low dosage of gonadotropin, or pulsatile gonadotropin-releasing hormone (GnRH) in the initial three or four cycles.
Continue treatment for five or more cycles to achieve an overall pregnancy rate approaching 65% without high-order multiple gestation in patients younger than 38 years who develop one or two follicles in a cycle.
Don’t rely on multifetal pregnancy reduction
This strategy has been viewed by some as a way to control the outcome of multiple gestation. For example, this is a common approach in New York, New Jersey, and Connecticut. However, the procedure has pitfalls and should not be the primary means of reducing the rate of multiple gestation because:
- It is not an acceptable option for many patients
- All fetuses may be lost in some cases
- The risks associated with multiple gestation are not completely eliminated
- It may have adverse psychological consequences.3,4
A registry is needed
Although a registry exists for IVF cycles and their outcomes and complications, none exists for cycles involving COS or ovulation induction. Despite many challenges to its development, we support the creation of such a registry.
It is vital that you develop the expertise and adopt strategies to reduce the rate of multiple gestation associated with controlled ovarian stimulation and ovulation induction. If you chose not to do so, refer the patient to someone who has such expertise.
Consider the patient’s emotional status when determining treatment for infertility
Domar AD, Smith K, Conboy L, Iannone M, Alper M. A prospective investigation into the reasons why insured United States patients drop out of in vitro fertilization treatment. Fertil Steril. 2009 Jul 8 [Epub ahead of print].
Most physicians have been trained to concentrate on the physiologic diagnosis and management of disease. Many fertility specialists also pay attention to economic barriers to treatment, such as lack of insurance and high cost, and attempt to help their patients gain access to quality care. One aspect of infertility that might be overlooked, however, is the patient’s emotional health—but it may be as important to the success of treatment as physiologic and economic variables.
A recent prospective investigation into the reasons insured patients drop out of IVF in the United States clearly demonstrated the psychological toll infertility can take. The study found that emotional distress is the number one reason that patients discontinue treatment.
How to lower the patient’s stress level
It can be challenging to counsel the patient to set realistic expectations for success yet enable her to maintain a sense of optimism. Stress management may be a key to success.
Physicians who treat patients with fertility problems should consider offering an in-practice counseling service aimed at reducing stress and improving coping mechanisms. At the very least, physicians should refer patients to outside resources that may be able to provide these services in a way that is meaningful and accessible.
Caring for a patient’s emotional well-being takes both time and skill. Besides offering direct emotional support to your patients, you can be a bridge to mental health and support services.
Patients who participate in a stress-reduction program while undergoing fertility treatment are 1) less likely to experience harmful emotional side effects and 2) more likely to continue treatment. Physicians who make such “mind-body” programs available are likely to reduce treatment dropout, improve the pregnancy rate, and increase the number of patients who take home babies.
Pay attention to the patient’s emotional health during treatment for infertility. Offer her access to stress management and other resources.
New endometriosis fertility index predicts non-IVF success rate
Adamson GD, Pasta DJ. Endometriosis fertility index: the new, validated endometriosis staging system. Fertil Steril. 2009 Nov 18 [Epub ahead of print].
Endometriosis remains a frustrating disease for patients who have infertility, in part because no staging system has made it possible for physicians to predict the pregnancy rate with fertility treatment other than IVF. The new, validated endometriosis fertility index (EFI) changes that. This simple, robust clinical tool predicts the pregnancy rate after surgical staging of endometriosis. Using it can provide reassurance to patients who have a good prognosis and avoid cost and distress of treatment in patients who have a poor prognosis.
Among the variables the index utilizes to predict the likelihood of pregnancy are:
- age of the patient
- duration of infertility
- gravidity
- total revised American Fertility Society (R-AFS) score
- R-AFS lesion score
- the new “least function score” (capability of the tubes, fimbria, and ovaries to effect their reproductive function, as determined by the surgeon after operative treatment) (FIGURES 2 AND 3).
FIGURE 2 Estimated pregnancy rate, by EFI score
FIGURE 3 A look at the endometriosis fertility index (EFI)
The least-function (LF) score (A) is determined at the conclusion of surgery using this form. The endometriosis fertility index (EFI) (B) incorporates the LF score and other variables to determine the overall score.
If you manage endometriosis patients who have infertility, use the new endometriosis fertility index to develop a realistic treatment plan in women who have a good prognosis—or to avert the need for treatment in patients who are unlikely to conceive.
1. Zegers-Hochschild F, Adamson GD, de Mouzon J, et al. For ICMART and the World Health Organization. International Committee for Monitoring Assisted Reproductive Technology (ICMART) and the World Health Organization (WHO) revised glossary of ART terminology, 2009. Fertil Steril. 2009;92:1520-1524.
2. Reindollar RH, Regan MM, Neumann PJ, et al. A randomized clinical trial to evaluate optimal treatment for unexplained infertility: the fasttrack and standard treatment (FASTT) trial. Fertil Steril. 2009 Jun 16 [Epub ahead of print].
3. Practice Committee of the American Society for Reproductive Medicine. Multiple pregnancy associated with infertility therapy. Fertil Steril. 2006;86(5 Suppl 1):S106-S110.
4. Stone J, Eddleman K, Lynch L, Berkowitz KL. A single center experience with 1,000 consecutive cases of multifetal pregnancy reduction. Am J Obstet Gynecol. 2002;187:1163-1167.
Infertility and its treatment can be a roller-coaster ride for patient and physician. Amid the emotional stress that arises, the goal of treatment can inadvertently shift from achievement of a successful singleton pregnancy to pregnancy at any cost—even high-order multiple gestation.
Here’s an essential question: Can the rate of multiple gestation be reduced without seriously compromising the pregnancy rate? Several developments of the past year suggest that it can be. In this article, we discuss:
- new guidelines that limit the number of embryos to be transferred at in vitro fertilization (IVF)
- strategies to reduce the risk of multiple gestation after controlled ovarian stimulation or ovulation induction
- the need to address the patient’s emotional status during treatment
- a new index that helps predict the pregnancy rate after surgical staging of endometriosis.
Multiple gestation is known to have adverse effects on infants, including a significantly elevated risk of prematurity and related physical and developmental problems. It also greatly increases the need for resources. And the high cost of caring for infants affected by prematurity further burdens an already overwhelmed health-care system.
Not only is it essential that we reduce the rate of high-order multiple gestation (i.e., more than two fetuses), but we should also attempt to lower the rate of twin pregnancy. A healthy singleton pregnancy, with its diminished risks and more reasonable health-care cost, should be our goal.
New guidelines limit the number of embryos to be transferred at IVF
Practice Committee of the American Society for Reproductive Medicine, Practice Committee of the Society for Assisted Reproductive Technology. Guidelines on number of embryos transferred. Fertil Steril. 2009;92:1518–1519.
Since the birth of Louise Brown in 1978, assisted reproductive technology (ART) has enjoyed dramatic technological advances. Intracytoplasmic sperm injection (FIGURE 1), preimplantation genetic diagnosis, and improvements in cryopreservation have broadened the application of ART and increased the live birth rate to 30% for every cycle that is initiated. The cumulative live birth rate from additional fresh and frozen-thawed cycles can reach 50% to 80%.
These gains have not come without cost, however. Multiple emotional, financial, and other variables affecting the practice of IVF have produced a higher-than-natural rate of multiple gestation.
In November 2009, the Society for Assisted Reproductive Technology (SART) and the American Society for Reproductive Medicine (ASRM) issued new guidelines limiting the number of embryos that should be transferred in one IVF cycle. IVF clinics are required to report outcomes, and approximately 93% of US cycles are reported to SART. High-order multiple-pregnancy rates are audited by SART, and outlier clinics must implement remediation programs to lower their high rate or risk expulsion from SART.
The increasing emphasis on single-embryo transfer in young women who have a good prognosis reflects the societies’ commitment to help patients achieve a healthy singleton pregnancy and good birth outcome.

FIGURE 1 A wonder of technology
Intracytoplasmic sperm injection overcomes many barriers to fertilization, such as severe malefactor infertility. At some institutions, the technique yields a fertilization rate of 70% to 80%.
What makes a “good prognosis”?
Identification of patients who have a good prognosis is an essential component of these new guidelines. The patient is more likely to have a favorable outcome if one or more of the following is true:
- She is undergoing her first cycle of IVF
- The embryos have good morphology
- Excess embryos are available for cryopreservation
- She has had earlier success with IVF.
The TABLE details the recommended number of embryos to transfer, based on the age and prognosis of the patient. In cycles that involve a donor egg, base the number of embryos to be transferred on the age of the donor. In cycles that involve a frozen embryo, base the number of good-quality, thawed embryos to be transferred on the age of the patient at the time the embryos were created. One additional embryo may be transferred if the patient has a less favorable prognosis or a history of two failed, fresh IVF cycles.
Two important requisites: Careful counseling about the risk of high-order multiple gestation, and documentation of that counseling.
TABLE
SART and ASRM recommend limits on the number of embryos to be transferred at in vitro fertilization
| Prognosis | Age of patient (yr) | |||
|---|---|---|---|---|
| <35 | 35–37 | 38–40 | 41 and 42 | |
| CLEAVAGE-STAGE EMBRYOS* | ||||
| Favorable† | 1 or 2 | 2 | 3 | 5 |
| All others | 2 | 3 | 4 | 5 |
| BLASTOCYSTS* | ||||
| Favorable† | 1 | 2 | 2 | 3 |
| All others | 2 | 2 | 3 | 3 |
| * See text and guidelines for more complete explanations. Justification for transferring one additional embryo (above the recommended limit) should be clearly documented in the patient’s medical record. | ||||
| †Variables indicating favorable prognosis include first cycle of IVF, good embryo quality, availability of excess embryos for cryopreservation, and previous successful IVF cycle. | ||||
All IVF clinics must adhere to the new SART and ASRM guidelines limiting the number of embryos to transfer at in vitro fertilization. In addition, it is vital for you to counsel the patient about the risk of high-order multiple gestation, and to document that such counseling took place.
Judicious management can reduce the rate of multiple gestation in ovulation stimulation
Dickey RP. Strategies to reduce multiple pregnancies due to ovulation stimulation. Fertil Steril. 2009;91:1–17.
Efforts to reduce the rate of multiple gestation should focus not only on patients undergoing IVF but on those undergoing controlled ovarian stimulation (COS) or ovulation induction. In COS, pharmacologic treatment is used to stimulate the production of more than one oocyte. In ovulation induction, pharmacologic therapy is used to induce normal cycles in anovulatory or oligo-ovulatory women.1 A substantial majority of multiple gestations are conceived using ovarian stimulation and ovulation induction. These methods may be less difficult to manage than IVF because they are less dependent on technology. Like IVF, however, they carry a high risk of multiple gestation, especially high-order multiple gestation.2
Strategies to reduce multiple gestation
As Dickey points out in a comprehensive retrospective analysis, there are strategies that can help reduce multiple gestation during COS and ovulation induction. They include the following recommendations:
Be prepared to cancel a cycle. Initiate ovulation induction only if both patient and physician are prepared to cancel any cycle that involves an excessive number of preovulatory follicles. Singleton and twin births can be confidently expected only if the cycle is cancelled when there are more than two preovulatory follicles approximately 12 mm in diameter or larger. This may be psychologically difficult for some patients and doctors.
Preemptively identify risk factors for multiple gestation, including:
- seven or more preovulatory follicles
- an estradiol concentration of 1,000 pg/mL or higher
- early cycles of treatment (cycles 1–3)
- age younger than 32 years
- body mass index below 19 kg/m2
- use of donor sperm.
When any of these risk factors is present, consider starting the patient on a lower initial dosage of gonadotropin; perform more frequent monitoring; maintain a low threshold for cancellation; and consider performing IVF with single-embryo transfer rather than COS.
Use specific drugs. Increase the likelihood of monofollicular development and double-follicular recruitment and reduce the risk of high-order multiple gestation by using clomiphene citrate, a low dosage of gonadotropin, or pulsatile gonadotropin-releasing hormone (GnRH) in the initial three or four cycles.
Continue treatment for five or more cycles to achieve an overall pregnancy rate approaching 65% without high-order multiple gestation in patients younger than 38 years who develop one or two follicles in a cycle.
Don’t rely on multifetal pregnancy reduction
This strategy has been viewed by some as a way to control the outcome of multiple gestation. For example, this is a common approach in New York, New Jersey, and Connecticut. However, the procedure has pitfalls and should not be the primary means of reducing the rate of multiple gestation because:
- It is not an acceptable option for many patients
- All fetuses may be lost in some cases
- The risks associated with multiple gestation are not completely eliminated
- It may have adverse psychological consequences.3,4
A registry is needed
Although a registry exists for IVF cycles and their outcomes and complications, none exists for cycles involving COS or ovulation induction. Despite many challenges to its development, we support the creation of such a registry.
It is vital that you develop the expertise and adopt strategies to reduce the rate of multiple gestation associated with controlled ovarian stimulation and ovulation induction. If you chose not to do so, refer the patient to someone who has such expertise.
Consider the patient’s emotional status when determining treatment for infertility
Domar AD, Smith K, Conboy L, Iannone M, Alper M. A prospective investigation into the reasons why insured United States patients drop out of in vitro fertilization treatment. Fertil Steril. 2009 Jul 8 [Epub ahead of print].
Most physicians have been trained to concentrate on the physiologic diagnosis and management of disease. Many fertility specialists also pay attention to economic barriers to treatment, such as lack of insurance and high cost, and attempt to help their patients gain access to quality care. One aspect of infertility that might be overlooked, however, is the patient’s emotional health—but it may be as important to the success of treatment as physiologic and economic variables.
A recent prospective investigation into the reasons insured patients drop out of IVF in the United States clearly demonstrated the psychological toll infertility can take. The study found that emotional distress is the number one reason that patients discontinue treatment.
How to lower the patient’s stress level
It can be challenging to counsel the patient to set realistic expectations for success yet enable her to maintain a sense of optimism. Stress management may be a key to success.
Physicians who treat patients with fertility problems should consider offering an in-practice counseling service aimed at reducing stress and improving coping mechanisms. At the very least, physicians should refer patients to outside resources that may be able to provide these services in a way that is meaningful and accessible.
Caring for a patient’s emotional well-being takes both time and skill. Besides offering direct emotional support to your patients, you can be a bridge to mental health and support services.
Patients who participate in a stress-reduction program while undergoing fertility treatment are 1) less likely to experience harmful emotional side effects and 2) more likely to continue treatment. Physicians who make such “mind-body” programs available are likely to reduce treatment dropout, improve the pregnancy rate, and increase the number of patients who take home babies.
Pay attention to the patient’s emotional health during treatment for infertility. Offer her access to stress management and other resources.
New endometriosis fertility index predicts non-IVF success rate
Adamson GD, Pasta DJ. Endometriosis fertility index: the new, validated endometriosis staging system. Fertil Steril. 2009 Nov 18 [Epub ahead of print].
Endometriosis remains a frustrating disease for patients who have infertility, in part because no staging system has made it possible for physicians to predict the pregnancy rate with fertility treatment other than IVF. The new, validated endometriosis fertility index (EFI) changes that. This simple, robust clinical tool predicts the pregnancy rate after surgical staging of endometriosis. Using it can provide reassurance to patients who have a good prognosis and avoid cost and distress of treatment in patients who have a poor prognosis.
Among the variables the index utilizes to predict the likelihood of pregnancy are:
- age of the patient
- duration of infertility
- gravidity
- total revised American Fertility Society (R-AFS) score
- R-AFS lesion score
- the new “least function score” (capability of the tubes, fimbria, and ovaries to effect their reproductive function, as determined by the surgeon after operative treatment) (FIGURES 2 AND 3).

FIGURE 2 Estimated pregnancy rate, by EFI score
FIGURE 3 A look at the endometriosis fertility index (EFI)
The least-function (LF) score (A) is determined at the conclusion of surgery using this form. The endometriosis fertility index (EFI) (B) incorporates the LF score and other variables to determine the overall score.
If you manage endometriosis patients who have infertility, use the new endometriosis fertility index to develop a realistic treatment plan in women who have a good prognosis—or to avert the need for treatment in patients who are unlikely to conceive.
Infertility and its treatment can be a roller-coaster ride for patient and physician. Amid the emotional stress that arises, the goal of treatment can inadvertently shift from achievement of a successful singleton pregnancy to pregnancy at any cost—even high-order multiple gestation.
Here’s an essential question: Can the rate of multiple gestation be reduced without seriously compromising the pregnancy rate? Several developments of the past year suggest that it can be. In this article, we discuss:
- new guidelines that limit the number of embryos to be transferred at in vitro fertilization (IVF)
- strategies to reduce the risk of multiple gestation after controlled ovarian stimulation or ovulation induction
- the need to address the patient’s emotional status during treatment
- a new index that helps predict the pregnancy rate after surgical staging of endometriosis.
Multiple gestation is known to have adverse effects on infants, including a significantly elevated risk of prematurity and related physical and developmental problems. It also greatly increases the need for resources. And the high cost of caring for infants affected by prematurity further burdens an already overwhelmed health-care system.
Not only is it essential that we reduce the rate of high-order multiple gestation (i.e., more than two fetuses), but we should also attempt to lower the rate of twin pregnancy. A healthy singleton pregnancy, with its diminished risks and more reasonable health-care cost, should be our goal.
New guidelines limit the number of embryos to be transferred at IVF
Practice Committee of the American Society for Reproductive Medicine, Practice Committee of the Society for Assisted Reproductive Technology. Guidelines on number of embryos transferred. Fertil Steril. 2009;92:1518–1519.
Since the birth of Louise Brown in 1978, assisted reproductive technology (ART) has enjoyed dramatic technological advances. Intracytoplasmic sperm injection (FIGURE 1), preimplantation genetic diagnosis, and improvements in cryopreservation have broadened the application of ART and increased the live birth rate to 30% for every cycle that is initiated. The cumulative live birth rate from additional fresh and frozen-thawed cycles can reach 50% to 80%.
These gains have not come without cost, however. Multiple emotional, financial, and other variables affecting the practice of IVF have produced a higher-than-natural rate of multiple gestation.
In November 2009, the Society for Assisted Reproductive Technology (SART) and the American Society for Reproductive Medicine (ASRM) issued new guidelines limiting the number of embryos that should be transferred in one IVF cycle. IVF clinics are required to report outcomes, and approximately 93% of US cycles are reported to SART. High-order multiple-pregnancy rates are audited by SART, and outlier clinics must implement remediation programs to lower their high rate or risk expulsion from SART.
The increasing emphasis on single-embryo transfer in young women who have a good prognosis reflects the societies’ commitment to help patients achieve a healthy singleton pregnancy and good birth outcome.

FIGURE 1 A wonder of technology
Intracytoplasmic sperm injection overcomes many barriers to fertilization, such as severe malefactor infertility. At some institutions, the technique yields a fertilization rate of 70% to 80%.
What makes a “good prognosis”?
Identification of patients who have a good prognosis is an essential component of these new guidelines. The patient is more likely to have a favorable outcome if one or more of the following is true:
- She is undergoing her first cycle of IVF
- The embryos have good morphology
- Excess embryos are available for cryopreservation
- She has had earlier success with IVF.
The TABLE details the recommended number of embryos to transfer, based on the age and prognosis of the patient. In cycles that involve a donor egg, base the number of embryos to be transferred on the age of the donor. In cycles that involve a frozen embryo, base the number of good-quality, thawed embryos to be transferred on the age of the patient at the time the embryos were created. One additional embryo may be transferred if the patient has a less favorable prognosis or a history of two failed, fresh IVF cycles.
Two important requisites: Careful counseling about the risk of high-order multiple gestation, and documentation of that counseling.
TABLE
SART and ASRM recommend limits on the number of embryos to be transferred at in vitro fertilization
| Prognosis | Age of patient (yr) | |||
|---|---|---|---|---|
| <35 | 35–37 | 38–40 | 41 and 42 | |
| CLEAVAGE-STAGE EMBRYOS* | ||||
| Favorable† | 1 or 2 | 2 | 3 | 5 |
| All others | 2 | 3 | 4 | 5 |
| BLASTOCYSTS* | ||||
| Favorable† | 1 | 2 | 2 | 3 |
| All others | 2 | 2 | 3 | 3 |
| * See text and guidelines for more complete explanations. Justification for transferring one additional embryo (above the recommended limit) should be clearly documented in the patient’s medical record. | ||||
| †Variables indicating favorable prognosis include first cycle of IVF, good embryo quality, availability of excess embryos for cryopreservation, and previous successful IVF cycle. | ||||
All IVF clinics must adhere to the new SART and ASRM guidelines limiting the number of embryos to transfer at in vitro fertilization. In addition, it is vital for you to counsel the patient about the risk of high-order multiple gestation, and to document that such counseling took place.
Judicious management can reduce the rate of multiple gestation in ovulation stimulation
Dickey RP. Strategies to reduce multiple pregnancies due to ovulation stimulation. Fertil Steril. 2009;91:1–17.
Efforts to reduce the rate of multiple gestation should focus not only on patients undergoing IVF but on those undergoing controlled ovarian stimulation (COS) or ovulation induction. In COS, pharmacologic treatment is used to stimulate the production of more than one oocyte. In ovulation induction, pharmacologic therapy is used to induce normal cycles in anovulatory or oligo-ovulatory women.1 A substantial majority of multiple gestations are conceived using ovarian stimulation and ovulation induction. These methods may be less difficult to manage than IVF because they are less dependent on technology. Like IVF, however, they carry a high risk of multiple gestation, especially high-order multiple gestation.2
Strategies to reduce multiple gestation
As Dickey points out in a comprehensive retrospective analysis, there are strategies that can help reduce multiple gestation during COS and ovulation induction. They include the following recommendations:
Be prepared to cancel a cycle. Initiate ovulation induction only if both patient and physician are prepared to cancel any cycle that involves an excessive number of preovulatory follicles. Singleton and twin births can be confidently expected only if the cycle is cancelled when there are more than two preovulatory follicles approximately 12 mm in diameter or larger. This may be psychologically difficult for some patients and doctors.
Preemptively identify risk factors for multiple gestation, including:
- seven or more preovulatory follicles
- an estradiol concentration of 1,000 pg/mL or higher
- early cycles of treatment (cycles 1–3)
- age younger than 32 years
- body mass index below 19 kg/m2
- use of donor sperm.
When any of these risk factors is present, consider starting the patient on a lower initial dosage of gonadotropin; perform more frequent monitoring; maintain a low threshold for cancellation; and consider performing IVF with single-embryo transfer rather than COS.
Use specific drugs. Increase the likelihood of monofollicular development and double-follicular recruitment and reduce the risk of high-order multiple gestation by using clomiphene citrate, a low dosage of gonadotropin, or pulsatile gonadotropin-releasing hormone (GnRH) in the initial three or four cycles.
Continue treatment for five or more cycles to achieve an overall pregnancy rate approaching 65% without high-order multiple gestation in patients younger than 38 years who develop one or two follicles in a cycle.
Don’t rely on multifetal pregnancy reduction
This strategy has been viewed by some as a way to control the outcome of multiple gestation. For example, this is a common approach in New York, New Jersey, and Connecticut. However, the procedure has pitfalls and should not be the primary means of reducing the rate of multiple gestation because:
- It is not an acceptable option for many patients
- All fetuses may be lost in some cases
- The risks associated with multiple gestation are not completely eliminated
- It may have adverse psychological consequences.3,4
A registry is needed
Although a registry exists for IVF cycles and their outcomes and complications, none exists for cycles involving COS or ovulation induction. Despite many challenges to its development, we support the creation of such a registry.
It is vital that you develop the expertise and adopt strategies to reduce the rate of multiple gestation associated with controlled ovarian stimulation and ovulation induction. If you chose not to do so, refer the patient to someone who has such expertise.
Consider the patient’s emotional status when determining treatment for infertility
Domar AD, Smith K, Conboy L, Iannone M, Alper M. A prospective investigation into the reasons why insured United States patients drop out of in vitro fertilization treatment. Fertil Steril. 2009 Jul 8 [Epub ahead of print].
Most physicians have been trained to concentrate on the physiologic diagnosis and management of disease. Many fertility specialists also pay attention to economic barriers to treatment, such as lack of insurance and high cost, and attempt to help their patients gain access to quality care. One aspect of infertility that might be overlooked, however, is the patient’s emotional health—but it may be as important to the success of treatment as physiologic and economic variables.
A recent prospective investigation into the reasons insured patients drop out of IVF in the United States clearly demonstrated the psychological toll infertility can take. The study found that emotional distress is the number one reason that patients discontinue treatment.
How to lower the patient’s stress level
It can be challenging to counsel the patient to set realistic expectations for success yet enable her to maintain a sense of optimism. Stress management may be a key to success.
Physicians who treat patients with fertility problems should consider offering an in-practice counseling service aimed at reducing stress and improving coping mechanisms. At the very least, physicians should refer patients to outside resources that may be able to provide these services in a way that is meaningful and accessible.
Caring for a patient’s emotional well-being takes both time and skill. Besides offering direct emotional support to your patients, you can be a bridge to mental health and support services.
Patients who participate in a stress-reduction program while undergoing fertility treatment are 1) less likely to experience harmful emotional side effects and 2) more likely to continue treatment. Physicians who make such “mind-body” programs available are likely to reduce treatment dropout, improve the pregnancy rate, and increase the number of patients who take home babies.
Pay attention to the patient’s emotional health during treatment for infertility. Offer her access to stress management and other resources.
New endometriosis fertility index predicts non-IVF success rate
Adamson GD, Pasta DJ. Endometriosis fertility index: the new, validated endometriosis staging system. Fertil Steril. 2009 Nov 18 [Epub ahead of print].
Endometriosis remains a frustrating disease for patients who have infertility, in part because no staging system has made it possible for physicians to predict the pregnancy rate with fertility treatment other than IVF. The new, validated endometriosis fertility index (EFI) changes that. This simple, robust clinical tool predicts the pregnancy rate after surgical staging of endometriosis. Using it can provide reassurance to patients who have a good prognosis and avoid cost and distress of treatment in patients who have a poor prognosis.
Among the variables the index utilizes to predict the likelihood of pregnancy are:
- age of the patient
- duration of infertility
- gravidity
- total revised American Fertility Society (R-AFS) score
- R-AFS lesion score
- the new “least function score” (capability of the tubes, fimbria, and ovaries to effect their reproductive function, as determined by the surgeon after operative treatment) (FIGURES 2 AND 3).

FIGURE 2 Estimated pregnancy rate, by EFI score
FIGURE 3 A look at the endometriosis fertility index (EFI)
The least-function (LF) score (A) is determined at the conclusion of surgery using this form. The endometriosis fertility index (EFI) (B) incorporates the LF score and other variables to determine the overall score.
If you manage endometriosis patients who have infertility, use the new endometriosis fertility index to develop a realistic treatment plan in women who have a good prognosis—or to avert the need for treatment in patients who are unlikely to conceive.
1. Zegers-Hochschild F, Adamson GD, de Mouzon J, et al. For ICMART and the World Health Organization. International Committee for Monitoring Assisted Reproductive Technology (ICMART) and the World Health Organization (WHO) revised glossary of ART terminology, 2009. Fertil Steril. 2009;92:1520-1524.
2. Reindollar RH, Regan MM, Neumann PJ, et al. A randomized clinical trial to evaluate optimal treatment for unexplained infertility: the fasttrack and standard treatment (FASTT) trial. Fertil Steril. 2009 Jun 16 [Epub ahead of print].
3. Practice Committee of the American Society for Reproductive Medicine. Multiple pregnancy associated with infertility therapy. Fertil Steril. 2006;86(5 Suppl 1):S106-S110.
4. Stone J, Eddleman K, Lynch L, Berkowitz KL. A single center experience with 1,000 consecutive cases of multifetal pregnancy reduction. Am J Obstet Gynecol. 2002;187:1163-1167.
1. Zegers-Hochschild F, Adamson GD, de Mouzon J, et al. For ICMART and the World Health Organization. International Committee for Monitoring Assisted Reproductive Technology (ICMART) and the World Health Organization (WHO) revised glossary of ART terminology, 2009. Fertil Steril. 2009;92:1520-1524.
2. Reindollar RH, Regan MM, Neumann PJ, et al. A randomized clinical trial to evaluate optimal treatment for unexplained infertility: the fasttrack and standard treatment (FASTT) trial. Fertil Steril. 2009 Jun 16 [Epub ahead of print].
3. Practice Committee of the American Society for Reproductive Medicine. Multiple pregnancy associated with infertility therapy. Fertil Steril. 2006;86(5 Suppl 1):S106-S110.
4. Stone J, Eddleman K, Lynch L, Berkowitz KL. A single center experience with 1,000 consecutive cases of multifetal pregnancy reduction. Am J Obstet Gynecol. 2002;187:1163-1167.
What you can do to optimize blood conservation in ObGyn practice
The authors report no financial relationships relevant to this article.
Obstetric hemorrhage is responsible for approximately 17% to 25% of all pregnancy-related deaths.1 Excessive blood loss also is a risk during gynecologic surgery. Iron-deficiency anemia increases the risk of complication and the need for transfusion in both settings. By identifying and treating anemia before childbirth and elective surgery, you can optimize the patient’s condition and usually avert the need for emergency transfusion.
The Geisinger Health System has developed a unique Blood Conservation Program that focuses on the prevention of major blood loss by identifying and treating anemia in antepartum, postpartum, and gynecologic patients. The program’s protocols for treating anemia in antepartum and surgical patients are illustrated in FIGURES 1 AND 2. Geisinger practitioners have found that adherence to these protocols reduces the need for transfusion in many patients and improves their quality of life.
Here, we 1) look at the key data that will help you identify and then treat anemia in gynecologic, obstetric, and postpartum patients and 2) describe a variety of treatment options.
FIGURE 1 The gynecologic surgical patient: Preop diagnosis and treatment of anemia
* If anemia is refractory to iron therapy, consider erythropoietin therapy if benefits outweigh risks.
FIGURE 2 For the antepartum patient: Diagnosis and treatment of anemia
* If anemia is refractory to iron therapy, consider erythropoietin therapy if benefits outweigh risks.
Focus on the baseline hemoglobin level
The key to prevention of emergency transfusion—as well as postpartum anemia—is optimization of the patient’s hemoglobin level before delivery. It also is prudent when elective surgery is planned. In our institution, clinicians whose patients are at risk for hemorrhage or significant blood loss have the option of consulting with the Blood Conservation Program.
When the program began in November 2004, its primary purpose was to reduce the need for blood transfusion in elective surgery, including gynecologic procedures. It later expanded to include obstetric patients who have a hemoglobin level below 11 g/dL and patients who are considered to be at risk of major blood loss.
In addition to obstetric and surgical patients, the Geisinger Blood Conservation Program provides support for patients who will not accept blood or blood products for religious or personal reasons, even in life-threatening situations. The program has provided more than 8,000 consultations to date.
When to evaluate patients for anemia
Anemia in women is most often defined as a hemoglobin level below 12 g/dL or a hematocrit below 36%. In pregnant patients, the cutoff points are lower: 11 g/dL and 33%, respectively. During pregnancy, hemoglobin and hematocrit levels reach their nadir during the second trimester and then begin to rise until term.
Symptoms of anemia include fatigue, depression, shortness of breath, hypotension, and heart palpitations. However, some patients at risk of major blood loss during delivery or surgery do not display typical symptoms associated with anemia, and the condition can be confirmed only by laboratory testing.
At Geisinger, we recommend consultation with the Blood Conservation Program for any patient who exhibits symptoms of anemia or who is at risk of major blood loss. For example, the risk of blood loss during childbirth varies with the method of delivery.1 On average, obstetric patients lose 500 mL of blood during vaginal delivery; 1,000 mL during cesarean delivery; and 1,500 mL when cesarean delivery is followed by hysterectomy.1,2 Hemorrhage is classified as follows:
- Class 1 – Blood loss as high as 750 mL, or 15% of blood volume
- Class 2 – 750 to 1,500 mL, or 15% to 30% of blood volume
- Class 3 – 1,500 to 2,400 mL, or 30% to 40% of blood volume
- Class 4 – more than 2,400 mL, or more than 40% of blood volume.1
Abnormal placentation, such as placenta accreta, percreta, increta, and previa, which can often be diagnosed antepartum, may lead to significant blood loss during and after delivery. Obstetric emergencies, including abruption, trauma, and uterine rupture, may also be associated with major blood loss.
Iron deficiency lies at the root of most cases of anemia
Iron deficiency affects an estimated 2.15 billion people globally, with a prevalence of 12% to 43% worldwide.3,4 The daily iron requirement is 1 mg of elemental iron for nonobstetric patients, 2 mg for pregnant and lactating women. Latent iron deficiency is common in women who have had multiple pregnancies. These and other important facts about iron are described in the Box “Essential facts about iron”.
In iron-deficiency anemia, the following serum levels are reduced:
- Iron. A normal reading is 60 to 170 μg/dL.
- Hemoglobin, a measure of the production and turnover of red blood cells. A normal level is ≥12 g/dL (≥11 g/dL in pregnancy).
- Serum ferritin (a protein that stores iron). A normal reading is 12 to 150 ng/mL.
- Transferrin saturation. Transferrin is a transporting protein that shuttles iron to the bone marrow. The normal transferrin saturation level ranges from 20% to 50%.
Ferritin and hemoglobin levels tend to be the most efficient indicators of iron status.5
Some clinicians may also use:
- Mean corpuscular volume (MCV). Normal is 80 to 96 fL.
- Random distribution of red blood cell weight (RDW). A normal value is 11.5% to 15.5%.
- Reticulocyte count. Normal is 0.4% to 2.3%.
Laboratory tests for iron deficiency
When the Blood Conservation Program is initially consulted, the laboratory studies we recommend are based on the clinical presentation and condition of the patient. During pregnancy, we try to take account of the normal hemodynamic changes that occur during gestation. Therefore, we recommend:
- assessment of the serum ferritin level
- complete blood count (CBC) with differential. (If the hemoglobin/hematocrit is low, a peripheral smear is recommended to further evaluate microcytic anemia.)
Transferrin saturation and serum iron levels have not been shown to be useful markers in pregnant women because they are not specific for iron-restricted erythropoiesis and can be abnormally low during pregnancy.6
In nonpregnant patients, we recommend initial evaluation of:
- serum iron level
- total iron-binding capacity (TIBC). Normal levels are 240 to 450 μg/dL
- transferrin saturation.
A caveat about the ferritin level
Ferritin is both an iron-storage indicator and an acute-phase protein, so the clinician must be careful to exclude inflammatory processes that can elevate the ferritin level, giving a false indicator of iron stability in the maternal system. These inflammatory processes can include preeclampsia and neoplastic or infectious conditions.7 Transferrin saturation, however, is not affected by inflammatory processes and can be used as a confirmatory test for iron deficiency.4
Try oral iron supplementation first
When laboratory testing confirms the presence of iron-deficiency anemia, initial management is oral iron supplementation for 2 weeks, followed by repeat laboratory evaluation.
For patients scheduled for surgery, oral therapy includes a daily dosage of:
- 325 mg of ferrous sulfate
- 250 mg of vitamin C
- 800 μg of folic acid
- a multivitamin.
For perinatal patients, the daily oral regimen is:
- 325 mg of ferrous sulfate
- 250 mg of vitamin C
- a prenatal vitamin.
These medications are the least expensive alternatives for treating anemia.
Advise patients who are taking iron supplements not to ingest the medication with dairy products, coffee, tea, or foods that have a high content of phytic acid (e.g., grains, seeds, and legumes). Foods and prescription drugs that interact with iron supplements are listed in TABLE 1, along with recommendations on optimal timing of iron supplementation and other medications.
When you prescribe oral iron supplementation, bear in mind that some patients experience gastrointestinal side effects—constipation, nausea, diarrhea—so unpleasant that they stop taking their medication. In that scenario, you will need to find alternative formulations or delivery routes. One alternative you can suggest is a daily helping of blackstrap molasses, which supplies 27 mg of elemental iron per tablespoon.
Oral therapy should be continued even after hemoglobin and ferritin levels normalize. If laboratory values remain low after 2 weeks of oral therapy, parenteral therapy can be added to the oral regimen.
Therapy may be discontinued 2 months after delivery of the infant or surgery as long as the cause of the blood loss has been remedied. If the mother is breastfeeding, she should continue taking a prenatal vitamin until nursing has stopped.
TABLE 1
Some foods and drugs don’t mix well with iron
| Food or drug | Interaction | Recommendations |
|---|---|---|
| Foods high in phytic acid (grains, seeds, legumes) | Decreased absorption of iron | Do not take iron within 2 hours of eating foods high in phytic acid |
| Dairy products | Decreased bioavailability of iron | Do not take iron supplements within 1 hour of consuming dairy products, which can significantly decrease iron absorption |
| Levothyroxine | Iron reduces levothyroxine serum levels and efficacy | Take levothyroxine and iron at least 4 hours apart |
| Methyldopa | Oral iron reduces the efficacy of methyldopa | Consider IV iron or take oral iron and methyldopa as far apart as possible |
| Proton pump inhibitors (PPIs) | Absorption of oral iron is enhanced by gastric acid. PPIs decrease gastric acid production, thereby reducing the bioavailability of iron | Consider IV iron preparations |
| Ofloxacin | Iron reduces efficacy of ofloxacin | Administer ofloxacin and iron 2 hours apart |
| Cholestyramine | Decreased efficacy of iron | Administer iron and cholestyramine at least 4 hours apart |
| Calcium, aluminum, magnesium | Decreased absorption of iron | Iron should be taken at least 1 hour before or 2 hours after products that contain calcium, aluminum, or magnesium |
| Note: This table is not a comprehensive summary of all medications used in practice, but a list of those used commonly in obstetric and gynecologic populations | ||
IV iron isn’t as risky as you think
Historically, clinicians have avoided using parenteral iron sucrose (Venofer) because they have been taught that it can cause an anaphylactic reaction. In fact, although anaphylaxis may have been associated with older intravenous (IV) iron preparations, clinical trials have demonstrated the safety of IV iron sucrose and low-molecular-weight iron dextran. In a study involving 800 patients, Breymann and colleagues demonstrated that parenteral iron preparations containing dextran or iron dextrin could be safely given to pregnant women.4 Only 1.5% of the patients in the study experienced side effects from the therapy, and no anaphylactic reactions were observed.
In another study, 25 pregnant patients were given IV iron sucrose, and the only adverse reaction reported was a “not-unpleasant taste” during the injection.8
In an additional study, Breymann and colleagues found no adverse outcomes in 20 postpartum patients who received IV iron sucrose in addition to erythropoietin therapy.9
- The daily iron requirement is 2 mg of elemental iron in pregnancy and lactation, 1 mg at all other times
- The typical US diet contains about 18 mg of iron a day, of which only about 1 mg is absorbed
- Iron absorption occurs primarily in the second portion of the duodenum
- Iron absorption increases with iron deficiency
- One unit of blood contains 250 mg of iron
- Total body iron store is between 1,000 and 3,000 mg, depending on body size
- Each pregnancy depletes maternal iron stores by about 750 mg
- Latent iron deficiency is common in women who have had many pregnancies and in women who have menorrhagia.
Our preference for parenteral therapy is iron sucrose, classified by the Food and Drug Administration (FDA) as Pregnancy Category B. Iron sucrose is contraindicated in patients who have iron overload, hypersensitivity to inactive components of iron sucrose, or anemia that is not caused by iron deficiency. Adverse reactions to iron sucrose include, but are not limited to, anaphylaxis, hypotension, cramping, nausea, headache, vomiting, diarrhea, and chest pain. Adverse reactions are very rare, occurring in fewer than 1% of patients.
To determine whether the patient has an allergy to iron sucrose, give a test dosage of 25 mg via slow IV push and wait 20 minutes. If a reaction occurs, hold the remainder of the dose and consider alternative therapies. If no allergic reaction occurs, administer the remaining 275 mg in 50 mL to 100 mL of saline.
You may need to add erythropoietin to the regimen
Erythropoietin is a hormone made by the kidneys to promote formation of red blood cells in the bone marrow. A deficiency in this hormone causes anemia in patients who have renal disease, and nephrologists use a synthetic form of epoetin alfa (Epogen) to increase the hemoglobin level in dialysis patients.10 Epoetin alfa falls into FDA Pregnancy Category C.
In rare instances, erythropoietin-stimulating agents (ESAs), such as epoetin alfa, in addition to both IV and oral iron supplementation, are needed to increase the patient’s hemoglobin level and hematocrit before delivery or surgery. Before beginning ESA therapy, the patient’s platelet count and activity level need to be considered. ESAs have been linked to thrombolytic events and, therefore, should not be used in patients who have an elevated platelet count. The risk of thrombolytic events is a particular danger for antepartum patients on bed rest, and ESAs may be contraindicated for that reason.
Obstetric and surgical patients whose anemia has proven refractory to iron therapy may be considered for an ESA, as long as the benefits of this choice outweigh the risks. At an approximate cost of $400 for every 40,000 U, ESA therapy is by far the most expensive alternative to blood transfusion for patients who have iron-deficiency anemia. The patient typically receives one to two doses of an ESA.
Cost comparisons for alternative treatment modalities in iron-deficiency anemia can be found in TABLE 2.
TABLE 2
Estimated cost of treatment of anemia*
| Therapy | Dosage | Cost per dose |
|---|---|---|
| ORAL THERAPY | ||
| Ferrous sulfate | 325 mg | $0.05–$0.09 |
| Vitamin C | 500 mg | $0.04 |
| Vitron C | 1 tablet | $0.20 |
| Folic acid | 800 μg | $0.02 |
| INTRAVENOUS THERAPY | ||
| Iron sucrose | 100 mg | $80.00 |
| OTHER INTERVENTIONS | ||
| Transfusion | 1 U | $500.00–$600.00 |
| Erythropoietin | 40,000 U | $400.00 |
| * Local averages in central Pennsylvania | ||
TABLE 3
How safe are iron compounds in pregnancy and lactation?
| Compound | FDA pregnancy category | World Health Organization lactation recommendation | Thompson lactation rating |
|---|---|---|---|
| Parenteral iron dextran | C | Compatible with breastfeeding | Risk to infant cannot be ruled out |
| Parenteral iron sucrose | B | ||
| Oral iron | A | Unavailable | |
| Oral sodium ferric gluconate | A | Compatible with breastfeeding |
Erythropoietin-stimulating agents carry serious risks
The FDA issued the first of a series of letters to health-care professionals warning of adverse events associated with the use of ESAs in March 2007, after several randomized, controlled trials found an increased risk of stroke, blood clots, myocardial infarction, and death with high dosages. In November 2008, the FDA approved a black-box warning for the labels of Procrit and Aranesp, the two ESAs in general use in the United States. The new labels advise clinicians to modify dosages for patients who are in renal failure to maintain a target hemoglobin level between 11 and 12 g/dL, rather than the higher targets that had been in use.11,12
Transfusion is the last resort
Blood transfusion must also be considered as prophylaxis for blood loss in patients who have critically low hemoglobin levels, with due consideration of the procedure’s risks and benefits. Because the definition of “critically low” varies from patient to patient, other variables should be taken into consideration, including blood pressure; heart rate; urine output; tolerance for performing activities of daily living without dizziness, chest discomfort, or shortness of breath; and medical history. Potential drawbacks are considerable.
The multiple risks associated with transfusion include:
- immunosuppression
- fever
- chills
- urticaria
- hemolytic transfusion reaction
- septic transfusion reaction
- bacterial contamination
- anaphylaxis
- graft-versus-host reactions
- transfer of viral diseases, including hepatitis B and C and human immunodeficiency virus (HIV).
The risk of immunosuppression, in particular, should be weighed heavily for pregnant patients and those who are planning an elective surgical procedure. The possibility of viral transmission is also a deterrent. According to the Red Cross, the transmission rate is one in every 205,000 transfusions for hepatitis B, one in 2 million for hepatitis C, and one in 2,135,000 for HIV. These considerations, as well as the blood shortages that sometimes occur in practice, are sufficient reason to seek safer alternatives, when possible.
When a patient refuses transfusion
Caring for a patient who has an elevated risk of major blood loss can be particularly difficult when she is a member of a religious group such as Jehovah’s Witnesses. These patients generally decline the transfusion of plasma, packed red blood cells, white blood cells, platelets, and whole blood products.
In the Geisinger Health System, consultation with the Blood Conservation Program has been particularly helpful in these circumstances, offering clinicians alternative ways to correct anemia and prepare for the possibility of major blood loss. Patients who will not allow blood transfusion are often willing to accept plasma volume expanders that are not derived from blood, such as perfluorocarbon solutions, hydroxyethyl starch, crystalloid, or dextran.13 ESA therapy may be acceptable to some patients who refuse transfusion. Most are willing to go along with oral or IV iron supplementation to reduce their need for transfusion.
Postpartum patients may need special consideration
Iron supplementation is safe for breastfeeding mothers
Anemia in a breastfeeding woman is not uncommon and should be identified and treated. Iron supplementation with oral or IV compounds is considered safe for pregnant and breastfeeding women.
ESA therapy is a riskier strategy, whose benefits must clearly outweigh risks for all patients.
Anemia and postpartum depression
Studies have demonstrated a correlation between anemia and postpartum depression. Beard and colleagues showed a 25% improvement in cognition and improved scores on stress and depression scales in postpartum women who had iron-deficiency anemia when they were treated with daily iron and vitamin C.14 Other studies have addressed an increased risk of anemia in low-income postpartum women and the deleterious impact of iron-deficiency anemia on the quality of mother–child interactions and subsequent child development. Correcting maternal iron deficiency could prevent adverse outcomes in these mothers and their offspring.15,16
1. Gabbe SG, Niebyl JR, Simpson JL. Obstetrics: Normal and Problem Pregnancies. 5th ed. Philadelphia, Pa: Churchill Livingstone; 2007.
2. Creasy RK, Resnik R, Iams JD. Creasy and Resnik’s Maternal–Fetal Medicine: Principles and Practice. 6th ed. Philadelphia, Pa: Saunders; 2009.
3. Khusun H, Yip R, Schultink W, Dillon DHS. World Health Organization hemoglobin cutoff points for the detection of anemia are valid for an Indonesian population. J Nutr. 1999;129:1669-1674.
4. Breymann C. Intravenous iron in surgery and obstetrics. Transfus Altern Transfus Med. 2002;4(Suppl 2):22-23.
5. Mei Z, Cogswell ME, Parvanta I, et al. Hemoglobin and ferritin are currently the most efficient indicators of population response to iron interventions: an analysis of nine randomized controlled trials. J Nutr. 2005;135:1974-1980.
6. Gronowski AM, ed. Current Clinical Pathology: Handbook of Clinical Laboratory Testing During Pregnancy. Totowa, NJ: Humana Press; 2004:200.
7. Mani S, Duffy TP. Anemia of pregnancy. Clin Perinatol. 1995;22:593-607.
8. Bayoumeu F, Subiran-Buisset C, Baka NE, Legagneur H, Monnier-Barbarino P, Laxenaire MC. Iron therapy in iron deficiency anemia in pregnancy: intravenous route versus oral route. Am J Obstet Gynecol. 2002;186:518-522.
9. Breymann C, Richter C, Hüttner C, Huch R, Huch A. Effectiveness of recombinant erythropoietin and iron sucrose vs. iron therapy only, in patients with postpartum anaemia and blunted erythropoiesis. Eur J Clin Invest. 2000;30:154-161.
10. Bieber E. Erythropoietin, the biology of erythropoiesis and epoetin alfa. An overview. J Reprod Med. 2001;46(5 Suppl):521-530.
11. Patient Information for Procrit. Available at: www.procrit.com. Accessed December 1, 2009.
12. Patient Information for Aranesp. Available at: www.aranesp.com. Accessed December 1, 2009.
13. Baker BW. Blood conservation, obstetrics, and Jehovah’s Witnesses. Anesthesiol Clin North America. 1998;16:375-384.
14. Beard JL, Hendricks MK, Perez EM, et al. Maternal iron deficiency anemia affects postpartum emotions and cognition. J Nutr. 2005;135:267-272.
15. Bodnar LM, Cogswell ME, Scanlon KS. Low income postpartum women are at risk of iron deficiency. J Nutr. 2002;132:2298-2302.
16. Perez EM, Hendricks MK, Beard JL, et al. Mother–infant interactions and infant development are altered by maternal iron deficiency anemia. J Nutr. 2005;135:850-855.
The authors report no financial relationships relevant to this article.
Obstetric hemorrhage is responsible for approximately 17% to 25% of all pregnancy-related deaths.1 Excessive blood loss also is a risk during gynecologic surgery. Iron-deficiency anemia increases the risk of complication and the need for transfusion in both settings. By identifying and treating anemia before childbirth and elective surgery, you can optimize the patient’s condition and usually avert the need for emergency transfusion.
The Geisinger Health System has developed a unique Blood Conservation Program that focuses on the prevention of major blood loss by identifying and treating anemia in antepartum, postpartum, and gynecologic patients. The program’s protocols for treating anemia in antepartum and surgical patients are illustrated in FIGURES 1 AND 2. Geisinger practitioners have found that adherence to these protocols reduces the need for transfusion in many patients and improves their quality of life.
Here, we 1) look at the key data that will help you identify and then treat anemia in gynecologic, obstetric, and postpartum patients and 2) describe a variety of treatment options.
FIGURE 1 The gynecologic surgical patient: Preop diagnosis and treatment of anemia
* If anemia is refractory to iron therapy, consider erythropoietin therapy if benefits outweigh risks.
FIGURE 2 For the antepartum patient: Diagnosis and treatment of anemia
* If anemia is refractory to iron therapy, consider erythropoietin therapy if benefits outweigh risks.
Focus on the baseline hemoglobin level
The key to prevention of emergency transfusion—as well as postpartum anemia—is optimization of the patient’s hemoglobin level before delivery. It also is prudent when elective surgery is planned. In our institution, clinicians whose patients are at risk for hemorrhage or significant blood loss have the option of consulting with the Blood Conservation Program.
When the program began in November 2004, its primary purpose was to reduce the need for blood transfusion in elective surgery, including gynecologic procedures. It later expanded to include obstetric patients who have a hemoglobin level below 11 g/dL and patients who are considered to be at risk of major blood loss.
In addition to obstetric and surgical patients, the Geisinger Blood Conservation Program provides support for patients who will not accept blood or blood products for religious or personal reasons, even in life-threatening situations. The program has provided more than 8,000 consultations to date.
When to evaluate patients for anemia
Anemia in women is most often defined as a hemoglobin level below 12 g/dL or a hematocrit below 36%. In pregnant patients, the cutoff points are lower: 11 g/dL and 33%, respectively. During pregnancy, hemoglobin and hematocrit levels reach their nadir during the second trimester and then begin to rise until term.
Symptoms of anemia include fatigue, depression, shortness of breath, hypotension, and heart palpitations. However, some patients at risk of major blood loss during delivery or surgery do not display typical symptoms associated with anemia, and the condition can be confirmed only by laboratory testing.
At Geisinger, we recommend consultation with the Blood Conservation Program for any patient who exhibits symptoms of anemia or who is at risk of major blood loss. For example, the risk of blood loss during childbirth varies with the method of delivery.1 On average, obstetric patients lose 500 mL of blood during vaginal delivery; 1,000 mL during cesarean delivery; and 1,500 mL when cesarean delivery is followed by hysterectomy.1,2 Hemorrhage is classified as follows:
- Class 1 – Blood loss as high as 750 mL, or 15% of blood volume
- Class 2 – 750 to 1,500 mL, or 15% to 30% of blood volume
- Class 3 – 1,500 to 2,400 mL, or 30% to 40% of blood volume
- Class 4 – more than 2,400 mL, or more than 40% of blood volume.1
Abnormal placentation, such as placenta accreta, percreta, increta, and previa, which can often be diagnosed antepartum, may lead to significant blood loss during and after delivery. Obstetric emergencies, including abruption, trauma, and uterine rupture, may also be associated with major blood loss.
Iron deficiency lies at the root of most cases of anemia
Iron deficiency affects an estimated 2.15 billion people globally, with a prevalence of 12% to 43% worldwide.3,4 The daily iron requirement is 1 mg of elemental iron for nonobstetric patients, 2 mg for pregnant and lactating women. Latent iron deficiency is common in women who have had multiple pregnancies. These and other important facts about iron are described in the Box “Essential facts about iron”.
In iron-deficiency anemia, the following serum levels are reduced:
- Iron. A normal reading is 60 to 170 μg/dL.
- Hemoglobin, a measure of the production and turnover of red blood cells. A normal level is ≥12 g/dL (≥11 g/dL in pregnancy).
- Serum ferritin (a protein that stores iron). A normal reading is 12 to 150 ng/mL.
- Transferrin saturation. Transferrin is a transporting protein that shuttles iron to the bone marrow. The normal transferrin saturation level ranges from 20% to 50%.
Ferritin and hemoglobin levels tend to be the most efficient indicators of iron status.5
Some clinicians may also use:
- Mean corpuscular volume (MCV). Normal is 80 to 96 fL.
- Random distribution of red blood cell weight (RDW). A normal value is 11.5% to 15.5%.
- Reticulocyte count. Normal is 0.4% to 2.3%.
Laboratory tests for iron deficiency
When the Blood Conservation Program is initially consulted, the laboratory studies we recommend are based on the clinical presentation and condition of the patient. During pregnancy, we try to take account of the normal hemodynamic changes that occur during gestation. Therefore, we recommend:
- assessment of the serum ferritin level
- complete blood count (CBC) with differential. (If the hemoglobin/hematocrit is low, a peripheral smear is recommended to further evaluate microcytic anemia.)
Transferrin saturation and serum iron levels have not been shown to be useful markers in pregnant women because they are not specific for iron-restricted erythropoiesis and can be abnormally low during pregnancy.6
In nonpregnant patients, we recommend initial evaluation of:
- serum iron level
- total iron-binding capacity (TIBC). Normal levels are 240 to 450 μg/dL
- transferrin saturation.
A caveat about the ferritin level
Ferritin is both an iron-storage indicator and an acute-phase protein, so the clinician must be careful to exclude inflammatory processes that can elevate the ferritin level, giving a false indicator of iron stability in the maternal system. These inflammatory processes can include preeclampsia and neoplastic or infectious conditions.7 Transferrin saturation, however, is not affected by inflammatory processes and can be used as a confirmatory test for iron deficiency.4
Try oral iron supplementation first
When laboratory testing confirms the presence of iron-deficiency anemia, initial management is oral iron supplementation for 2 weeks, followed by repeat laboratory evaluation.
For patients scheduled for surgery, oral therapy includes a daily dosage of:
- 325 mg of ferrous sulfate
- 250 mg of vitamin C
- 800 μg of folic acid
- a multivitamin.
For perinatal patients, the daily oral regimen is:
- 325 mg of ferrous sulfate
- 250 mg of vitamin C
- a prenatal vitamin.
These medications are the least expensive alternatives for treating anemia.
Advise patients who are taking iron supplements not to ingest the medication with dairy products, coffee, tea, or foods that have a high content of phytic acid (e.g., grains, seeds, and legumes). Foods and prescription drugs that interact with iron supplements are listed in TABLE 1, along with recommendations on optimal timing of iron supplementation and other medications.
When you prescribe oral iron supplementation, bear in mind that some patients experience gastrointestinal side effects—constipation, nausea, diarrhea—so unpleasant that they stop taking their medication. In that scenario, you will need to find alternative formulations or delivery routes. One alternative you can suggest is a daily helping of blackstrap molasses, which supplies 27 mg of elemental iron per tablespoon.
Oral therapy should be continued even after hemoglobin and ferritin levels normalize. If laboratory values remain low after 2 weeks of oral therapy, parenteral therapy can be added to the oral regimen.
Therapy may be discontinued 2 months after delivery of the infant or surgery as long as the cause of the blood loss has been remedied. If the mother is breastfeeding, she should continue taking a prenatal vitamin until nursing has stopped.
TABLE 1
Some foods and drugs don’t mix well with iron
| Food or drug | Interaction | Recommendations |
|---|---|---|
| Foods high in phytic acid (grains, seeds, legumes) | Decreased absorption of iron | Do not take iron within 2 hours of eating foods high in phytic acid |
| Dairy products | Decreased bioavailability of iron | Do not take iron supplements within 1 hour of consuming dairy products, which can significantly decrease iron absorption |
| Levothyroxine | Iron reduces levothyroxine serum levels and efficacy | Take levothyroxine and iron at least 4 hours apart |
| Methyldopa | Oral iron reduces the efficacy of methyldopa | Consider IV iron or take oral iron and methyldopa as far apart as possible |
| Proton pump inhibitors (PPIs) | Absorption of oral iron is enhanced by gastric acid. PPIs decrease gastric acid production, thereby reducing the bioavailability of iron | Consider IV iron preparations |
| Ofloxacin | Iron reduces efficacy of ofloxacin | Administer ofloxacin and iron 2 hours apart |
| Cholestyramine | Decreased efficacy of iron | Administer iron and cholestyramine at least 4 hours apart |
| Calcium, aluminum, magnesium | Decreased absorption of iron | Iron should be taken at least 1 hour before or 2 hours after products that contain calcium, aluminum, or magnesium |
| Note: This table is not a comprehensive summary of all medications used in practice, but a list of those used commonly in obstetric and gynecologic populations | ||
IV iron isn’t as risky as you think
Historically, clinicians have avoided using parenteral iron sucrose (Venofer) because they have been taught that it can cause an anaphylactic reaction. In fact, although anaphylaxis may have been associated with older intravenous (IV) iron preparations, clinical trials have demonstrated the safety of IV iron sucrose and low-molecular-weight iron dextran. In a study involving 800 patients, Breymann and colleagues demonstrated that parenteral iron preparations containing dextran or iron dextrin could be safely given to pregnant women.4 Only 1.5% of the patients in the study experienced side effects from the therapy, and no anaphylactic reactions were observed.
In another study, 25 pregnant patients were given IV iron sucrose, and the only adverse reaction reported was a “not-unpleasant taste” during the injection.8
In an additional study, Breymann and colleagues found no adverse outcomes in 20 postpartum patients who received IV iron sucrose in addition to erythropoietin therapy.9
- The daily iron requirement is 2 mg of elemental iron in pregnancy and lactation, 1 mg at all other times
- The typical US diet contains about 18 mg of iron a day, of which only about 1 mg is absorbed
- Iron absorption occurs primarily in the second portion of the duodenum
- Iron absorption increases with iron deficiency
- One unit of blood contains 250 mg of iron
- Total body iron store is between 1,000 and 3,000 mg, depending on body size
- Each pregnancy depletes maternal iron stores by about 750 mg
- Latent iron deficiency is common in women who have had many pregnancies and in women who have menorrhagia.
Our preference for parenteral therapy is iron sucrose, classified by the Food and Drug Administration (FDA) as Pregnancy Category B. Iron sucrose is contraindicated in patients who have iron overload, hypersensitivity to inactive components of iron sucrose, or anemia that is not caused by iron deficiency. Adverse reactions to iron sucrose include, but are not limited to, anaphylaxis, hypotension, cramping, nausea, headache, vomiting, diarrhea, and chest pain. Adverse reactions are very rare, occurring in fewer than 1% of patients.
To determine whether the patient has an allergy to iron sucrose, give a test dosage of 25 mg via slow IV push and wait 20 minutes. If a reaction occurs, hold the remainder of the dose and consider alternative therapies. If no allergic reaction occurs, administer the remaining 275 mg in 50 mL to 100 mL of saline.
You may need to add erythropoietin to the regimen
Erythropoietin is a hormone made by the kidneys to promote formation of red blood cells in the bone marrow. A deficiency in this hormone causes anemia in patients who have renal disease, and nephrologists use a synthetic form of epoetin alfa (Epogen) to increase the hemoglobin level in dialysis patients.10 Epoetin alfa falls into FDA Pregnancy Category C.
In rare instances, erythropoietin-stimulating agents (ESAs), such as epoetin alfa, in addition to both IV and oral iron supplementation, are needed to increase the patient’s hemoglobin level and hematocrit before delivery or surgery. Before beginning ESA therapy, the patient’s platelet count and activity level need to be considered. ESAs have been linked to thrombolytic events and, therefore, should not be used in patients who have an elevated platelet count. The risk of thrombolytic events is a particular danger for antepartum patients on bed rest, and ESAs may be contraindicated for that reason.
Obstetric and surgical patients whose anemia has proven refractory to iron therapy may be considered for an ESA, as long as the benefits of this choice outweigh the risks. At an approximate cost of $400 for every 40,000 U, ESA therapy is by far the most expensive alternative to blood transfusion for patients who have iron-deficiency anemia. The patient typically receives one to two doses of an ESA.
Cost comparisons for alternative treatment modalities in iron-deficiency anemia can be found in TABLE 2.
TABLE 2
Estimated cost of treatment of anemia*
| Therapy | Dosage | Cost per dose |
|---|---|---|
| ORAL THERAPY | ||
| Ferrous sulfate | 325 mg | $0.05–$0.09 |
| Vitamin C | 500 mg | $0.04 |
| Vitron C | 1 tablet | $0.20 |
| Folic acid | 800 μg | $0.02 |
| INTRAVENOUS THERAPY | ||
| Iron sucrose | 100 mg | $80.00 |
| OTHER INTERVENTIONS | ||
| Transfusion | 1 U | $500.00–$600.00 |
| Erythropoietin | 40,000 U | $400.00 |
| * Local averages in central Pennsylvania | ||
TABLE 3
How safe are iron compounds in pregnancy and lactation?
| Compound | FDA pregnancy category | World Health Organization lactation recommendation | Thompson lactation rating |
|---|---|---|---|
| Parenteral iron dextran | C | Compatible with breastfeeding | Risk to infant cannot be ruled out |
| Parenteral iron sucrose | B | ||
| Oral iron | A | Unavailable | |
| Oral sodium ferric gluconate | A | Compatible with breastfeeding |
Erythropoietin-stimulating agents carry serious risks
The FDA issued the first of a series of letters to health-care professionals warning of adverse events associated with the use of ESAs in March 2007, after several randomized, controlled trials found an increased risk of stroke, blood clots, myocardial infarction, and death with high dosages. In November 2008, the FDA approved a black-box warning for the labels of Procrit and Aranesp, the two ESAs in general use in the United States. The new labels advise clinicians to modify dosages for patients who are in renal failure to maintain a target hemoglobin level between 11 and 12 g/dL, rather than the higher targets that had been in use.11,12
Transfusion is the last resort
Blood transfusion must also be considered as prophylaxis for blood loss in patients who have critically low hemoglobin levels, with due consideration of the procedure’s risks and benefits. Because the definition of “critically low” varies from patient to patient, other variables should be taken into consideration, including blood pressure; heart rate; urine output; tolerance for performing activities of daily living without dizziness, chest discomfort, or shortness of breath; and medical history. Potential drawbacks are considerable.
The multiple risks associated with transfusion include:
- immunosuppression
- fever
- chills
- urticaria
- hemolytic transfusion reaction
- septic transfusion reaction
- bacterial contamination
- anaphylaxis
- graft-versus-host reactions
- transfer of viral diseases, including hepatitis B and C and human immunodeficiency virus (HIV).
The risk of immunosuppression, in particular, should be weighed heavily for pregnant patients and those who are planning an elective surgical procedure. The possibility of viral transmission is also a deterrent. According to the Red Cross, the transmission rate is one in every 205,000 transfusions for hepatitis B, one in 2 million for hepatitis C, and one in 2,135,000 for HIV. These considerations, as well as the blood shortages that sometimes occur in practice, are sufficient reason to seek safer alternatives, when possible.
When a patient refuses transfusion
Caring for a patient who has an elevated risk of major blood loss can be particularly difficult when she is a member of a religious group such as Jehovah’s Witnesses. These patients generally decline the transfusion of plasma, packed red blood cells, white blood cells, platelets, and whole blood products.
In the Geisinger Health System, consultation with the Blood Conservation Program has been particularly helpful in these circumstances, offering clinicians alternative ways to correct anemia and prepare for the possibility of major blood loss. Patients who will not allow blood transfusion are often willing to accept plasma volume expanders that are not derived from blood, such as perfluorocarbon solutions, hydroxyethyl starch, crystalloid, or dextran.13 ESA therapy may be acceptable to some patients who refuse transfusion. Most are willing to go along with oral or IV iron supplementation to reduce their need for transfusion.
Postpartum patients may need special consideration
Iron supplementation is safe for breastfeeding mothers
Anemia in a breastfeeding woman is not uncommon and should be identified and treated. Iron supplementation with oral or IV compounds is considered safe for pregnant and breastfeeding women.
ESA therapy is a riskier strategy, whose benefits must clearly outweigh risks for all patients.
Anemia and postpartum depression
Studies have demonstrated a correlation between anemia and postpartum depression. Beard and colleagues showed a 25% improvement in cognition and improved scores on stress and depression scales in postpartum women who had iron-deficiency anemia when they were treated with daily iron and vitamin C.14 Other studies have addressed an increased risk of anemia in low-income postpartum women and the deleterious impact of iron-deficiency anemia on the quality of mother–child interactions and subsequent child development. Correcting maternal iron deficiency could prevent adverse outcomes in these mothers and their offspring.15,16
The authors report no financial relationships relevant to this article.
Obstetric hemorrhage is responsible for approximately 17% to 25% of all pregnancy-related deaths.1 Excessive blood loss also is a risk during gynecologic surgery. Iron-deficiency anemia increases the risk of complication and the need for transfusion in both settings. By identifying and treating anemia before childbirth and elective surgery, you can optimize the patient’s condition and usually avert the need for emergency transfusion.
The Geisinger Health System has developed a unique Blood Conservation Program that focuses on the prevention of major blood loss by identifying and treating anemia in antepartum, postpartum, and gynecologic patients. The program’s protocols for treating anemia in antepartum and surgical patients are illustrated in FIGURES 1 AND 2. Geisinger practitioners have found that adherence to these protocols reduces the need for transfusion in many patients and improves their quality of life.
Here, we 1) look at the key data that will help you identify and then treat anemia in gynecologic, obstetric, and postpartum patients and 2) describe a variety of treatment options.
FIGURE 1 The gynecologic surgical patient: Preop diagnosis and treatment of anemia
* If anemia is refractory to iron therapy, consider erythropoietin therapy if benefits outweigh risks.
FIGURE 2 For the antepartum patient: Diagnosis and treatment of anemia
* If anemia is refractory to iron therapy, consider erythropoietin therapy if benefits outweigh risks.
Focus on the baseline hemoglobin level
The key to prevention of emergency transfusion—as well as postpartum anemia—is optimization of the patient’s hemoglobin level before delivery. It also is prudent when elective surgery is planned. In our institution, clinicians whose patients are at risk for hemorrhage or significant blood loss have the option of consulting with the Blood Conservation Program.
When the program began in November 2004, its primary purpose was to reduce the need for blood transfusion in elective surgery, including gynecologic procedures. It later expanded to include obstetric patients who have a hemoglobin level below 11 g/dL and patients who are considered to be at risk of major blood loss.
In addition to obstetric and surgical patients, the Geisinger Blood Conservation Program provides support for patients who will not accept blood or blood products for religious or personal reasons, even in life-threatening situations. The program has provided more than 8,000 consultations to date.
When to evaluate patients for anemia
Anemia in women is most often defined as a hemoglobin level below 12 g/dL or a hematocrit below 36%. In pregnant patients, the cutoff points are lower: 11 g/dL and 33%, respectively. During pregnancy, hemoglobin and hematocrit levels reach their nadir during the second trimester and then begin to rise until term.
Symptoms of anemia include fatigue, depression, shortness of breath, hypotension, and heart palpitations. However, some patients at risk of major blood loss during delivery or surgery do not display typical symptoms associated with anemia, and the condition can be confirmed only by laboratory testing.
At Geisinger, we recommend consultation with the Blood Conservation Program for any patient who exhibits symptoms of anemia or who is at risk of major blood loss. For example, the risk of blood loss during childbirth varies with the method of delivery.1 On average, obstetric patients lose 500 mL of blood during vaginal delivery; 1,000 mL during cesarean delivery; and 1,500 mL when cesarean delivery is followed by hysterectomy.1,2 Hemorrhage is classified as follows:
- Class 1 – Blood loss as high as 750 mL, or 15% of blood volume
- Class 2 – 750 to 1,500 mL, or 15% to 30% of blood volume
- Class 3 – 1,500 to 2,400 mL, or 30% to 40% of blood volume
- Class 4 – more than 2,400 mL, or more than 40% of blood volume.1
Abnormal placentation, such as placenta accreta, percreta, increta, and previa, which can often be diagnosed antepartum, may lead to significant blood loss during and after delivery. Obstetric emergencies, including abruption, trauma, and uterine rupture, may also be associated with major blood loss.
Iron deficiency lies at the root of most cases of anemia
Iron deficiency affects an estimated 2.15 billion people globally, with a prevalence of 12% to 43% worldwide.3,4 The daily iron requirement is 1 mg of elemental iron for nonobstetric patients, 2 mg for pregnant and lactating women. Latent iron deficiency is common in women who have had multiple pregnancies. These and other important facts about iron are described in the Box “Essential facts about iron”.
In iron-deficiency anemia, the following serum levels are reduced:
- Iron. A normal reading is 60 to 170 μg/dL.
- Hemoglobin, a measure of the production and turnover of red blood cells. A normal level is ≥12 g/dL (≥11 g/dL in pregnancy).
- Serum ferritin (a protein that stores iron). A normal reading is 12 to 150 ng/mL.
- Transferrin saturation. Transferrin is a transporting protein that shuttles iron to the bone marrow. The normal transferrin saturation level ranges from 20% to 50%.
Ferritin and hemoglobin levels tend to be the most efficient indicators of iron status.5
Some clinicians may also use:
- Mean corpuscular volume (MCV). Normal is 80 to 96 fL.
- Random distribution of red blood cell weight (RDW). A normal value is 11.5% to 15.5%.
- Reticulocyte count. Normal is 0.4% to 2.3%.
Laboratory tests for iron deficiency
When the Blood Conservation Program is initially consulted, the laboratory studies we recommend are based on the clinical presentation and condition of the patient. During pregnancy, we try to take account of the normal hemodynamic changes that occur during gestation. Therefore, we recommend:
- assessment of the serum ferritin level
- complete blood count (CBC) with differential. (If the hemoglobin/hematocrit is low, a peripheral smear is recommended to further evaluate microcytic anemia.)
Transferrin saturation and serum iron levels have not been shown to be useful markers in pregnant women because they are not specific for iron-restricted erythropoiesis and can be abnormally low during pregnancy.6
In nonpregnant patients, we recommend initial evaluation of:
- serum iron level
- total iron-binding capacity (TIBC). Normal levels are 240 to 450 μg/dL
- transferrin saturation.
A caveat about the ferritin level
Ferritin is both an iron-storage indicator and an acute-phase protein, so the clinician must be careful to exclude inflammatory processes that can elevate the ferritin level, giving a false indicator of iron stability in the maternal system. These inflammatory processes can include preeclampsia and neoplastic or infectious conditions.7 Transferrin saturation, however, is not affected by inflammatory processes and can be used as a confirmatory test for iron deficiency.4
Try oral iron supplementation first
When laboratory testing confirms the presence of iron-deficiency anemia, initial management is oral iron supplementation for 2 weeks, followed by repeat laboratory evaluation.
For patients scheduled for surgery, oral therapy includes a daily dosage of:
- 325 mg of ferrous sulfate
- 250 mg of vitamin C
- 800 μg of folic acid
- a multivitamin.
For perinatal patients, the daily oral regimen is:
- 325 mg of ferrous sulfate
- 250 mg of vitamin C
- a prenatal vitamin.
These medications are the least expensive alternatives for treating anemia.
Advise patients who are taking iron supplements not to ingest the medication with dairy products, coffee, tea, or foods that have a high content of phytic acid (e.g., grains, seeds, and legumes). Foods and prescription drugs that interact with iron supplements are listed in TABLE 1, along with recommendations on optimal timing of iron supplementation and other medications.
When you prescribe oral iron supplementation, bear in mind that some patients experience gastrointestinal side effects—constipation, nausea, diarrhea—so unpleasant that they stop taking their medication. In that scenario, you will need to find alternative formulations or delivery routes. One alternative you can suggest is a daily helping of blackstrap molasses, which supplies 27 mg of elemental iron per tablespoon.
Oral therapy should be continued even after hemoglobin and ferritin levels normalize. If laboratory values remain low after 2 weeks of oral therapy, parenteral therapy can be added to the oral regimen.
Therapy may be discontinued 2 months after delivery of the infant or surgery as long as the cause of the blood loss has been remedied. If the mother is breastfeeding, she should continue taking a prenatal vitamin until nursing has stopped.
TABLE 1
Some foods and drugs don’t mix well with iron
| Food or drug | Interaction | Recommendations |
|---|---|---|
| Foods high in phytic acid (grains, seeds, legumes) | Decreased absorption of iron | Do not take iron within 2 hours of eating foods high in phytic acid |
| Dairy products | Decreased bioavailability of iron | Do not take iron supplements within 1 hour of consuming dairy products, which can significantly decrease iron absorption |
| Levothyroxine | Iron reduces levothyroxine serum levels and efficacy | Take levothyroxine and iron at least 4 hours apart |
| Methyldopa | Oral iron reduces the efficacy of methyldopa | Consider IV iron or take oral iron and methyldopa as far apart as possible |
| Proton pump inhibitors (PPIs) | Absorption of oral iron is enhanced by gastric acid. PPIs decrease gastric acid production, thereby reducing the bioavailability of iron | Consider IV iron preparations |
| Ofloxacin | Iron reduces efficacy of ofloxacin | Administer ofloxacin and iron 2 hours apart |
| Cholestyramine | Decreased efficacy of iron | Administer iron and cholestyramine at least 4 hours apart |
| Calcium, aluminum, magnesium | Decreased absorption of iron | Iron should be taken at least 1 hour before or 2 hours after products that contain calcium, aluminum, or magnesium |
| Note: This table is not a comprehensive summary of all medications used in practice, but a list of those used commonly in obstetric and gynecologic populations | ||
IV iron isn’t as risky as you think
Historically, clinicians have avoided using parenteral iron sucrose (Venofer) because they have been taught that it can cause an anaphylactic reaction. In fact, although anaphylaxis may have been associated with older intravenous (IV) iron preparations, clinical trials have demonstrated the safety of IV iron sucrose and low-molecular-weight iron dextran. In a study involving 800 patients, Breymann and colleagues demonstrated that parenteral iron preparations containing dextran or iron dextrin could be safely given to pregnant women.4 Only 1.5% of the patients in the study experienced side effects from the therapy, and no anaphylactic reactions were observed.
In another study, 25 pregnant patients were given IV iron sucrose, and the only adverse reaction reported was a “not-unpleasant taste” during the injection.8
In an additional study, Breymann and colleagues found no adverse outcomes in 20 postpartum patients who received IV iron sucrose in addition to erythropoietin therapy.9
- The daily iron requirement is 2 mg of elemental iron in pregnancy and lactation, 1 mg at all other times
- The typical US diet contains about 18 mg of iron a day, of which only about 1 mg is absorbed
- Iron absorption occurs primarily in the second portion of the duodenum
- Iron absorption increases with iron deficiency
- One unit of blood contains 250 mg of iron
- Total body iron store is between 1,000 and 3,000 mg, depending on body size
- Each pregnancy depletes maternal iron stores by about 750 mg
- Latent iron deficiency is common in women who have had many pregnancies and in women who have menorrhagia.
Our preference for parenteral therapy is iron sucrose, classified by the Food and Drug Administration (FDA) as Pregnancy Category B. Iron sucrose is contraindicated in patients who have iron overload, hypersensitivity to inactive components of iron sucrose, or anemia that is not caused by iron deficiency. Adverse reactions to iron sucrose include, but are not limited to, anaphylaxis, hypotension, cramping, nausea, headache, vomiting, diarrhea, and chest pain. Adverse reactions are very rare, occurring in fewer than 1% of patients.
To determine whether the patient has an allergy to iron sucrose, give a test dosage of 25 mg via slow IV push and wait 20 minutes. If a reaction occurs, hold the remainder of the dose and consider alternative therapies. If no allergic reaction occurs, administer the remaining 275 mg in 50 mL to 100 mL of saline.
You may need to add erythropoietin to the regimen
Erythropoietin is a hormone made by the kidneys to promote formation of red blood cells in the bone marrow. A deficiency in this hormone causes anemia in patients who have renal disease, and nephrologists use a synthetic form of epoetin alfa (Epogen) to increase the hemoglobin level in dialysis patients.10 Epoetin alfa falls into FDA Pregnancy Category C.
In rare instances, erythropoietin-stimulating agents (ESAs), such as epoetin alfa, in addition to both IV and oral iron supplementation, are needed to increase the patient’s hemoglobin level and hematocrit before delivery or surgery. Before beginning ESA therapy, the patient’s platelet count and activity level need to be considered. ESAs have been linked to thrombolytic events and, therefore, should not be used in patients who have an elevated platelet count. The risk of thrombolytic events is a particular danger for antepartum patients on bed rest, and ESAs may be contraindicated for that reason.
Obstetric and surgical patients whose anemia has proven refractory to iron therapy may be considered for an ESA, as long as the benefits of this choice outweigh the risks. At an approximate cost of $400 for every 40,000 U, ESA therapy is by far the most expensive alternative to blood transfusion for patients who have iron-deficiency anemia. The patient typically receives one to two doses of an ESA.
Cost comparisons for alternative treatment modalities in iron-deficiency anemia can be found in TABLE 2.
TABLE 2
Estimated cost of treatment of anemia*
| Therapy | Dosage | Cost per dose |
|---|---|---|
| ORAL THERAPY | ||
| Ferrous sulfate | 325 mg | $0.05–$0.09 |
| Vitamin C | 500 mg | $0.04 |
| Vitron C | 1 tablet | $0.20 |
| Folic acid | 800 μg | $0.02 |
| INTRAVENOUS THERAPY | ||
| Iron sucrose | 100 mg | $80.00 |
| OTHER INTERVENTIONS | ||
| Transfusion | 1 U | $500.00–$600.00 |
| Erythropoietin | 40,000 U | $400.00 |
| * Local averages in central Pennsylvania | ||
TABLE 3
How safe are iron compounds in pregnancy and lactation?
| Compound | FDA pregnancy category | World Health Organization lactation recommendation | Thompson lactation rating |
|---|---|---|---|
| Parenteral iron dextran | C | Compatible with breastfeeding | Risk to infant cannot be ruled out |
| Parenteral iron sucrose | B | ||
| Oral iron | A | Unavailable | |
| Oral sodium ferric gluconate | A | Compatible with breastfeeding |
Erythropoietin-stimulating agents carry serious risks
The FDA issued the first of a series of letters to health-care professionals warning of adverse events associated with the use of ESAs in March 2007, after several randomized, controlled trials found an increased risk of stroke, blood clots, myocardial infarction, and death with high dosages. In November 2008, the FDA approved a black-box warning for the labels of Procrit and Aranesp, the two ESAs in general use in the United States. The new labels advise clinicians to modify dosages for patients who are in renal failure to maintain a target hemoglobin level between 11 and 12 g/dL, rather than the higher targets that had been in use.11,12
Transfusion is the last resort
Blood transfusion must also be considered as prophylaxis for blood loss in patients who have critically low hemoglobin levels, with due consideration of the procedure’s risks and benefits. Because the definition of “critically low” varies from patient to patient, other variables should be taken into consideration, including blood pressure; heart rate; urine output; tolerance for performing activities of daily living without dizziness, chest discomfort, or shortness of breath; and medical history. Potential drawbacks are considerable.
The multiple risks associated with transfusion include:
- immunosuppression
- fever
- chills
- urticaria
- hemolytic transfusion reaction
- septic transfusion reaction
- bacterial contamination
- anaphylaxis
- graft-versus-host reactions
- transfer of viral diseases, including hepatitis B and C and human immunodeficiency virus (HIV).
The risk of immunosuppression, in particular, should be weighed heavily for pregnant patients and those who are planning an elective surgical procedure. The possibility of viral transmission is also a deterrent. According to the Red Cross, the transmission rate is one in every 205,000 transfusions for hepatitis B, one in 2 million for hepatitis C, and one in 2,135,000 for HIV. These considerations, as well as the blood shortages that sometimes occur in practice, are sufficient reason to seek safer alternatives, when possible.
When a patient refuses transfusion
Caring for a patient who has an elevated risk of major blood loss can be particularly difficult when she is a member of a religious group such as Jehovah’s Witnesses. These patients generally decline the transfusion of plasma, packed red blood cells, white blood cells, platelets, and whole blood products.
In the Geisinger Health System, consultation with the Blood Conservation Program has been particularly helpful in these circumstances, offering clinicians alternative ways to correct anemia and prepare for the possibility of major blood loss. Patients who will not allow blood transfusion are often willing to accept plasma volume expanders that are not derived from blood, such as perfluorocarbon solutions, hydroxyethyl starch, crystalloid, or dextran.13 ESA therapy may be acceptable to some patients who refuse transfusion. Most are willing to go along with oral or IV iron supplementation to reduce their need for transfusion.
Postpartum patients may need special consideration
Iron supplementation is safe for breastfeeding mothers
Anemia in a breastfeeding woman is not uncommon and should be identified and treated. Iron supplementation with oral or IV compounds is considered safe for pregnant and breastfeeding women.
ESA therapy is a riskier strategy, whose benefits must clearly outweigh risks for all patients.
Anemia and postpartum depression
Studies have demonstrated a correlation between anemia and postpartum depression. Beard and colleagues showed a 25% improvement in cognition and improved scores on stress and depression scales in postpartum women who had iron-deficiency anemia when they were treated with daily iron and vitamin C.14 Other studies have addressed an increased risk of anemia in low-income postpartum women and the deleterious impact of iron-deficiency anemia on the quality of mother–child interactions and subsequent child development. Correcting maternal iron deficiency could prevent adverse outcomes in these mothers and their offspring.15,16
1. Gabbe SG, Niebyl JR, Simpson JL. Obstetrics: Normal and Problem Pregnancies. 5th ed. Philadelphia, Pa: Churchill Livingstone; 2007.
2. Creasy RK, Resnik R, Iams JD. Creasy and Resnik’s Maternal–Fetal Medicine: Principles and Practice. 6th ed. Philadelphia, Pa: Saunders; 2009.
3. Khusun H, Yip R, Schultink W, Dillon DHS. World Health Organization hemoglobin cutoff points for the detection of anemia are valid for an Indonesian population. J Nutr. 1999;129:1669-1674.
4. Breymann C. Intravenous iron in surgery and obstetrics. Transfus Altern Transfus Med. 2002;4(Suppl 2):22-23.
5. Mei Z, Cogswell ME, Parvanta I, et al. Hemoglobin and ferritin are currently the most efficient indicators of population response to iron interventions: an analysis of nine randomized controlled trials. J Nutr. 2005;135:1974-1980.
6. Gronowski AM, ed. Current Clinical Pathology: Handbook of Clinical Laboratory Testing During Pregnancy. Totowa, NJ: Humana Press; 2004:200.
7. Mani S, Duffy TP. Anemia of pregnancy. Clin Perinatol. 1995;22:593-607.
8. Bayoumeu F, Subiran-Buisset C, Baka NE, Legagneur H, Monnier-Barbarino P, Laxenaire MC. Iron therapy in iron deficiency anemia in pregnancy: intravenous route versus oral route. Am J Obstet Gynecol. 2002;186:518-522.
9. Breymann C, Richter C, Hüttner C, Huch R, Huch A. Effectiveness of recombinant erythropoietin and iron sucrose vs. iron therapy only, in patients with postpartum anaemia and blunted erythropoiesis. Eur J Clin Invest. 2000;30:154-161.
10. Bieber E. Erythropoietin, the biology of erythropoiesis and epoetin alfa. An overview. J Reprod Med. 2001;46(5 Suppl):521-530.
11. Patient Information for Procrit. Available at: www.procrit.com. Accessed December 1, 2009.
12. Patient Information for Aranesp. Available at: www.aranesp.com. Accessed December 1, 2009.
13. Baker BW. Blood conservation, obstetrics, and Jehovah’s Witnesses. Anesthesiol Clin North America. 1998;16:375-384.
14. Beard JL, Hendricks MK, Perez EM, et al. Maternal iron deficiency anemia affects postpartum emotions and cognition. J Nutr. 2005;135:267-272.
15. Bodnar LM, Cogswell ME, Scanlon KS. Low income postpartum women are at risk of iron deficiency. J Nutr. 2002;132:2298-2302.
16. Perez EM, Hendricks MK, Beard JL, et al. Mother–infant interactions and infant development are altered by maternal iron deficiency anemia. J Nutr. 2005;135:850-855.
1. Gabbe SG, Niebyl JR, Simpson JL. Obstetrics: Normal and Problem Pregnancies. 5th ed. Philadelphia, Pa: Churchill Livingstone; 2007.
2. Creasy RK, Resnik R, Iams JD. Creasy and Resnik’s Maternal–Fetal Medicine: Principles and Practice. 6th ed. Philadelphia, Pa: Saunders; 2009.
3. Khusun H, Yip R, Schultink W, Dillon DHS. World Health Organization hemoglobin cutoff points for the detection of anemia are valid for an Indonesian population. J Nutr. 1999;129:1669-1674.
4. Breymann C. Intravenous iron in surgery and obstetrics. Transfus Altern Transfus Med. 2002;4(Suppl 2):22-23.
5. Mei Z, Cogswell ME, Parvanta I, et al. Hemoglobin and ferritin are currently the most efficient indicators of population response to iron interventions: an analysis of nine randomized controlled trials. J Nutr. 2005;135:1974-1980.
6. Gronowski AM, ed. Current Clinical Pathology: Handbook of Clinical Laboratory Testing During Pregnancy. Totowa, NJ: Humana Press; 2004:200.
7. Mani S, Duffy TP. Anemia of pregnancy. Clin Perinatol. 1995;22:593-607.
8. Bayoumeu F, Subiran-Buisset C, Baka NE, Legagneur H, Monnier-Barbarino P, Laxenaire MC. Iron therapy in iron deficiency anemia in pregnancy: intravenous route versus oral route. Am J Obstet Gynecol. 2002;186:518-522.
9. Breymann C, Richter C, Hüttner C, Huch R, Huch A. Effectiveness of recombinant erythropoietin and iron sucrose vs. iron therapy only, in patients with postpartum anaemia and blunted erythropoiesis. Eur J Clin Invest. 2000;30:154-161.
10. Bieber E. Erythropoietin, the biology of erythropoiesis and epoetin alfa. An overview. J Reprod Med. 2001;46(5 Suppl):521-530.
11. Patient Information for Procrit. Available at: www.procrit.com. Accessed December 1, 2009.
12. Patient Information for Aranesp. Available at: www.aranesp.com. Accessed December 1, 2009.
13. Baker BW. Blood conservation, obstetrics, and Jehovah’s Witnesses. Anesthesiol Clin North America. 1998;16:375-384.
14. Beard JL, Hendricks MK, Perez EM, et al. Maternal iron deficiency anemia affects postpartum emotions and cognition. J Nutr. 2005;135:267-272.
15. Bodnar LM, Cogswell ME, Scanlon KS. Low income postpartum women are at risk of iron deficiency. J Nutr. 2002;132:2298-2302.
16. Perez EM, Hendricks MK, Beard JL, et al. Mother–infant interactions and infant development are altered by maternal iron deficiency anemia. J Nutr. 2005;135:850-855.
Evaluation and Diagnosis
Intraocular Pressure Changes During Cardiopulmonary Bypass Pump and Off-Pump Cardiac Surgeries
Acute Obstructive Manifestation of a Jejunal Carcinoid
Dialysis and Elders' Functional Status
Does Blood Transfusion Benefit Patients with Acute Decompensated Heart Failure?
Management of Carotid Artery Disease
Carotid artery disease is a treatable cause of ischemic stroke, a potentially devastating event that affects approximately 700,000 Americans each year and results in more than 160,000 deaths.1,2 Stroke-related medical costs, including associated disability, now approach $60 billion per year. Despite advances in treatment, stroke remains the third leading cause of death in the United States.3
As the population ages, stroke prevention has become an increasing challenge for primary care providers. Guiding patients at risk toward the appropriate testing and treatment can offer lifelong benefits. This article will summarize current practice recommendations for screening asymptomatic individuals and for treatment of carotid artery disease using carotid endarterectomy or carotid angioplasty with stenting.
Scope and Screening
Carotid artery stenosis (CAS) is defined as atherosclerotic narrowing of the extracranial carotid arteries. Possibly 20% of ischemic strokes (which represent more than 85% of all strokes) result from CAS, a condition that may or may not be symptomatic.4Symptomatic CAS may be represented by a cerebrovascular accident, a transient ischemic attack, or one of an array of more subtle but enduring neurologic deficits.
The prime risk factor for CAS is prior history of cerebrovascular disease.4 Cardiovascular disease or cigarette smoking doubles a patient’s risk for developing CAS. Other risk factors include age greater than 65, male gender, hypertension, atrial fibrillation, and clotting disorders.
Population studies based on carotid ultrasonography estimate the prevalence of CAS at 0.5% to 8.0% in the general population.4-6 Clinically significant CAS (60% or higher) has been estimated at 1% in those older than 65.4
The degree of carotid occlusion correlates directly with the risk of ipsilateral stroke. The rate of stroke among asymptomatic patients with CAS of at least 80% is 3.5% to 5.0% per year.7 To date, there is no clinically useful risk model to identify those who have CAS or will develop it.
Screening the general population for asymptomatic CAS is not currently recommended.4,8 Guidelines published in 2007 by the Society for Vascular Surgery (SVS)3 advise ultrasound screening only for persons 55 and older who have cardiovascular risk factors, including diabetes, hypertension, hypercholesterolemia, a history of smoking, or known cerebrovascular disease. That same year, the American Society of Neuroimaging9 recommended screening of adults 65 or older who have three or more cardiovascular risk factors.
Ultrasound screening is approximately 94% sensitive and 92% specific for moderate to severe CAS (ie, 60% to 90% occlusion).4 Patients with positive ultrasound findings may next undergo computerized axial angiography, magnetic resonance angiography, or digital subtraction angiography.
Angiography can detect with good precision the degree and location of carotid occlusion, which in turn helps to select treatment options, in consideration of their inherent risks and benefits. These options are medical therapy alone, or medical therapy combined with carotid endarterectomy (CEA), or carotid angioplasty with stenting.
The Research
Stroke prevention, long since a medical priority, is most commonly sought by way of pharmacotherapy combined with lifestyle modification. Surgery, in the form of CEA, also plays an enduring and proven role. Randomized trials, including three landmark studies,10-12 have established CEA as standard treatment for symptomatic and high-grade occlusive carotid disease. The North American Symptomatic Carotid Endarterectomy Trial (NASCET)10 and the European Carotid Surgery Trial (ECST)11 provided the basis for stratifying symptomatic patients and determining whether surgery will produce a reasonable benefit. The Asymptomatic Carotid Atherosclerosis Study (ACAS)12 extended the research to asymptomatic patients with high-grade stenosis.8,13 The benefits of CEA for elderly patients (75 and older) with significant comorbidities were supported in the 2009 New York Carotid Artery Surgery Study (NYCAS).14
Researchers for ACAS,12 which compared medical therapy alone with CEA plus medical therapy in asymptomatic patients with CAS, reported a relative risk (RR) reduction of 0.53 in patients undergoing CEA, with a 5.1% five-year rate of stroke or death in the CEA group versus 11.0% among patients receiving medical therapy alone. The Asymptomatic Carotid Surgery Trial15 (ACST) yielded similar event rates (CEA, 6.4%; medical therapy alone, 11.8%). In both trials, the perioperative (30-day) risk of stroke or death associated with CEA ranged from 2.7% to 3.1%. In the long term (five to 10 years and beyond), RR reduction remains uncertain.
However, CEA remains the gold standard for the treatment of severe carotid artery disease. Currently, about 75% of patients who undergo CEA for significant CAS are asymptomatic.13
Complications associated with CEA occur at an ascending rate, commensurate with the patient’s preoperative stroke history. Researchers for the NYCAS14 reported a 30-day post-CEA rate of stroke or death of nearly 3% among asymptomatic patients with no history of stroke or TIA; nearly 8% among patients with previous stroke; and more than 13% in patients with crescendo TIA or evolving stroke. A significant increase in complications (including stroke or death) was reported among patients with coronary artery disease or with diabetes requiring insulin therapy.
The Case for Carotid Angioplasty with Stenting
Though broadly accepted and practiced, CEA carries significant risk for symptomatic patients and for those who face higher surgical risks, such as diabetes or cardiovascular disease, or anatomic issues such as contralateral occlusions (see Table 1).
Carotid angioplasty with stenting emerged in the 1990s as a less invasive alternative to CEA that could be performed under local anesthesia and with little or no sedation. In the Carotid and Vertebral Artery Transluminal Angioplasty Study (CAVATAS),17 no significant difference was found in three-year stroke risk between patients assigned randomly to CEA or to carotid stenting.
The Stenting and Angioplasty with Protection in Patients at High Risk for Endarterectomy (SAPPHIRE) trial,7,16,18 an industry-sponsored study, was the first multicenter study to compare CEA with stenting in patients considered at high surgical risk. All stenting procedures were performed using an intraoperative embolic protection device. Patients with stents had a 4.8% risk of stroke, MI, or death in the 30-day postoperative period, compared with 9.8% among patients who underwent CEA. Despite their potential clinical relevance, these results were not found to be statistically significant.
The risk of ipsilateral stroke at one year was similar between treatment groups. Follow-up data published in 2008 and 2009 showed comparable outcomes and no differences in repeat revascularization rates between CEA and stent groups.7,16 SAPPHIRE is now conducting a worldwide registry study in an effort to extend its results to a broader population. The Center for Medicare Services has approved carotid artery stenting with embolic protection for patients who meet the SAPPHIRE high-risk criteria.
Research on the effectiveness of distal protection devices in preventing intraoperative stroke is ongoing.19 In the interim, the SVS recommends embolic protection during all carotid stenting procedures.13 Perioperative medical management remains critical to the success of carotid stenting. This includes intraoperative heparin and clopidogrel for at least two to four weeks postoperatively.7,16
In elderly patients (80 and older), carotid artery stenting may present a particularly high risk.20,21 Investigators for the ongoing Carotid Revascularization Endarterectomy versus Stenting Trial (CREST) have found a periprocedural risk of death or stroke at 12.1% among older CAS patients, compared with 4.0% among their younger counterparts.21,22 This discrepancy has been attributed to age-related changes in vasculature that create a more hostile environment for endovascular devices.
Patients younger than 80 with significant but asymptomatic unilateral stenosis who are at average surgical risk are presently the focus of other trials. Now under way, the Carotid Angioplasty and Stenting versus Endarterectomy in Asymptomatic Subjects with Significant Extracranial Carotid Occlusive Disease Trial (ACT I) is the first major trial involving asymptomatic patients, and so far has shown a lower postintervention event rate than reported in smaller previous studies.23 However, these patients are less likely to experience postoperative events than their symptomatic counterparts. Thus, the role of stenting in asymptomatic patients will require long-term follow-up.
To date, CEA retains its gold standard status as the optimum surgical treatment for preventing stroke-associated morbidity and mortality. At the same time, carotid stenting is emerging as an effective and less invasive alternative, especially for patients younger than 80 who are at high perioperative risk for CEA.
Treatment Guidelines
In 2008, the SVS13 issued clinical practice guidelines based on an empirical analysis of the currently available research on carotid stenosis. These guidelines address both medical and anatomic risk and acknowledge the limitations of comparing massive data obtained from robust but independent clinical trials. The SVS authors acknowledge that some terms (eg, high perioperative risk) remain somewhat difficult to define and are subject to practitioners’ interpretation.
In an effort to achieve consensus, the SVS investigators employed the British-based GRADE (Grading of Recommendations, Assessment, Development and Evaluation) system24 to stratify the strength of its recommendations. This system takes into account factors other than the quality of the data, including the reviewers’ values and preferences, and their evaluation of the data as presented (see Table 213).
As the SVS authors note, there is no significant difference to date between outcomes for stenting versus CEA, including death or stroke within 30 days postprocedure and the need for revascularization within three years.13 They conclude, nevertheless, that CEA remains the treatment of choice for asymptomatic patients with moderate to severe stenosis. Symptomatic patients can be stratified based on age and medical and surgical risk when a choice is being made between CEA and stenting. In patients at high risk, lifelong pharmacologic therapy may be safer than either surgical or endovascular treatment.
The Role of Medical Management
Whether or not CEA or stenting is performed, medical therapy plays a crucial role in the management of carotid artery disease. Most patients are placed on aspirin therapy indefinitely unless its use is contraindicated (eg, by risk for gastrointestinal bleeding). The SVS practice guidelines13 incorporate medical therapy, citing joint recommendations issued in 2006 by the American Heart Association and the American Stroke Association (AHA/ASA)1,25 for tight control of hypertension, blood glucose, and elevated cholesterol.
The AHA/ASA researchers also recommend antiplatelet agents (aspirin, clopidogrel, and/or dipyridamole) for patients with a history of TIA or noncardioembolic ischemic stroke. The guidelines advise moderate alcohol consumption, weight reduction for obese patients, and increased physical activity. Smoking cessation remains the sine qua non of vascular disease management.1,25
Conclusion
Primary care providers play a pivotal role in identifying patients at risk for carotid artery disease and educating them about current treatment options. They have an opportunity to take a proactive role in screening patients (age 55 and over) who smoke or who have diabetes, high blood pressure, high cholesterol, or coronary artery disease.
Detection of moderate to severe CAS can lead to timely surgical intervention in asymptomatic individuals who may not realize they are at risk. Attentive medical and lifestyle management enhances the treatment of carotid disease and reduces the risk of stroke, its most devastating consequence.
1. Goldstein LB, Adams R, Alberts MJ, et al. Primary prevention of ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council: cosponsored by the Atherosclerotic Peripheral Vascular Disease Interdisciplinary Working Group; Cardiovascular Nursing Council; Clinical Cardiology Council; Nutrition, Physical Activity, and Metabolism Council; and the Quality of Care and Outcomes Research Interdisciplinary Working Group: the American Academy of Neurology affirms the value of this guideline. Stroke. 2006;37(6):1583–1633.
2. American Heart Association. Heart Disease and Stroke Statistics—2004 Update. www.americanheart.org/downloadable/heart/1079736729696HDSStats2004UpdateREV3-19-04.pdf. Accessed December 28, 2009.
3. Society for Vascular Surgery. SVS position statement on vascular screenings. www.vascularweb.org/patients/screenings/SVS_Position_Statement_on_Vascular_Screenings.html. Accessed December 28, 2009.
4. Wolff T, Guirguis-Blake J, Miller T, et al. Screening for carotid artery stenosis: an update of the evidence for the US Preventive Services Task Force. Ann Intern Med. 2007;147(12):860- 870.
5. Colgan MP, Strode GR, Sommer JD, et al. Prevalence of asymptomatic carotid disease: results of duplex scanning in 348 unselected volunteers. J Vasc Surg. 1988;8(6):674-678.
6. Pujia A, Rubba P, Spencer MP. Prevalence of extracranial carotid artery disease detectable by echo-Doppler in an elderly population. Stroke. 1992;23(6):818-822.
7. Gurm HS, Yadav JS, Fayad P, et al; SAPPHIRE Investigators. Long-term results of carotid stenting versus endarterectomy in high-risk patients. N Engl J Med. 2008;358(15):1572-1579.
8. US Preventive Services Task Force. Screening for carotid artery stenosis: US Preventive Services Task Force Recommendation Statement. Ann Intern Med. 2007;147(12):854-859.
9. Qureshi AI, Alexandrov AV, Tegeler CH, et al. Guidelines for screening of extracranial carotid artery disease: a statement for healthcare professionals from the multidisciplinary practice guidelines committee of the American Society of Neuroimaging; cosponsored by the Society of Vascular and Interventional Neurology. J Neuroimaging. 2007;17(1):19-47.
10. Ferguson GG, Eliasziw M, Barr HWK, et al. The North American Symptomatic Carotid Endarterectomy Trial. Stroke. 1999;30(9):1751-1758.
11. Rothwell PM, Gutnikov SA, Warlow CP; European Carotid Surgery Trial. Reanalysis of the final results of the European Carotid Surgery Trial. Stroke. 2003;34(2):514-23.
12. Executive Committee for the Asymptomatic Carotid Atherosclerosis Study. Endarterectomy for asymptomatic carotid artery stenosis. JAMA. 1995;273(18):1421-1428.
13. Hobson RW 2nd, Mackey WC, Ascher E, et al; Society for Vascular Surgery. Management of atherosclerotic carotid artery disease: clinical practice guidelines of the Society for Vascular Surgery. J Vasc Surg. 2008;48(2):480-486.
14. Halm EA, Tuhrim S, Wang JJ, et al. Risk factors for perioperative death and stroke after carotid endarterectomy: results of the New York Carotid Artery Surgery Study. Stroke. 2009;40(1):221-229.
15. Halliday A, Mansfield A, Marro J, et al; MRC Asymptomatic Carotid Surgery Trial (ACST). Prevention of disabling and fatal strokes by successful carotid endarterectomy in patients without recent neurological symptoms: randomised controlled trial. Lancet. 2004;363(9420):1491-1502.
16. Yadav JS, Wholey MH, Kuntz RE, et al; Stenting and Angioplasty with Protection in Patients at High Risk for Endarterectomy Investigators (SAPPHIRE). Protected carotid-artery stenting versus endarterectomy in high-risk patients. N Engl J Med. 2004;351(15):1493-1501.
17. Endovascular versus surgical treatment in patients with carotid stenosis in the Carotid and Vertebral Artery Transluminal Angioplasty Study (CAVATAS): a randomised trial. Lancet. 2001;357: 1729–1737.
18. Massop D, Dave R, Metzger C, et al. Stenting and angioplasty with protection in patients at high-risk for endarterectomy: SAPPHIRE Worldwide Registry first 2,001 patients. Catheter Cardiovasc Interv. 2009;73(2):129-136.
19. Barbato JE, Dillavou E, Horowitz MB, et al. A randomized trial of carotid artery stenting with and without cerebral protection. J Vasc Surg. 2008;47(4):760-765.
20. Suliman A, Greenberg J, Chandra A, et al. Carotid endarterectomy as the criterion standard in high-risk elderly patients. Arch Surg. 2008; 143(8):736-742.
21. Hobson RW 2nd, Howard VJ, Roubin GS, et al. Carotid artery stenting is associated with increased complications in octogenarians: 30-day stroke and death rates in the CREST lead-in phase. J Vasc Surg. 2004;40(6):1106-1111.
22. Lal BK, Brott TG. The Carotid Revascularization Endarterectomy vs. Stenting Trial completes randomization: lessons learned and anticipated results. J Vasc Surg. 2009;50(5):1224-1231.
23. Derdeyn CP. Carotid stenting for asymptomatic carotid stenosis: trial it. Stroke. 2007;38(2 suppl):715-720.
24. Guyatt GH, Oxman AD, Vist GE, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336(7650):924-926.
25. Sacco RL, Adams R, Albers G, et al. Guidelines for prevention of stroke in patients with ischemic stroke or transient ischemic attack: a statement for healthcare professionals from the American Heart Association/American Stroke Association Council on Stroke: co-sponsored by the Council on Cardiovascular Radiology and Intervention: the American Academy of Neurology affirms the value of this guideline. Stroke. 2006;37(2):577-617.
Carotid artery disease is a treatable cause of ischemic stroke, a potentially devastating event that affects approximately 700,000 Americans each year and results in more than 160,000 deaths.1,2 Stroke-related medical costs, including associated disability, now approach $60 billion per year. Despite advances in treatment, stroke remains the third leading cause of death in the United States.3
As the population ages, stroke prevention has become an increasing challenge for primary care providers. Guiding patients at risk toward the appropriate testing and treatment can offer lifelong benefits. This article will summarize current practice recommendations for screening asymptomatic individuals and for treatment of carotid artery disease using carotid endarterectomy or carotid angioplasty with stenting.
Scope and Screening
Carotid artery stenosis (CAS) is defined as atherosclerotic narrowing of the extracranial carotid arteries. Possibly 20% of ischemic strokes (which represent more than 85% of all strokes) result from CAS, a condition that may or may not be symptomatic.4Symptomatic CAS may be represented by a cerebrovascular accident, a transient ischemic attack, or one of an array of more subtle but enduring neurologic deficits.
The prime risk factor for CAS is prior history of cerebrovascular disease.4 Cardiovascular disease or cigarette smoking doubles a patient’s risk for developing CAS. Other risk factors include age greater than 65, male gender, hypertension, atrial fibrillation, and clotting disorders.
Population studies based on carotid ultrasonography estimate the prevalence of CAS at 0.5% to 8.0% in the general population.4-6 Clinically significant CAS (60% or higher) has been estimated at 1% in those older than 65.4
The degree of carotid occlusion correlates directly with the risk of ipsilateral stroke. The rate of stroke among asymptomatic patients with CAS of at least 80% is 3.5% to 5.0% per year.7 To date, there is no clinically useful risk model to identify those who have CAS or will develop it.
Screening the general population for asymptomatic CAS is not currently recommended.4,8 Guidelines published in 2007 by the Society for Vascular Surgery (SVS)3 advise ultrasound screening only for persons 55 and older who have cardiovascular risk factors, including diabetes, hypertension, hypercholesterolemia, a history of smoking, or known cerebrovascular disease. That same year, the American Society of Neuroimaging9 recommended screening of adults 65 or older who have three or more cardiovascular risk factors.
Ultrasound screening is approximately 94% sensitive and 92% specific for moderate to severe CAS (ie, 60% to 90% occlusion).4 Patients with positive ultrasound findings may next undergo computerized axial angiography, magnetic resonance angiography, or digital subtraction angiography.
Angiography can detect with good precision the degree and location of carotid occlusion, which in turn helps to select treatment options, in consideration of their inherent risks and benefits. These options are medical therapy alone, or medical therapy combined with carotid endarterectomy (CEA), or carotid angioplasty with stenting.
The Research
Stroke prevention, long since a medical priority, is most commonly sought by way of pharmacotherapy combined with lifestyle modification. Surgery, in the form of CEA, also plays an enduring and proven role. Randomized trials, including three landmark studies,10-12 have established CEA as standard treatment for symptomatic and high-grade occlusive carotid disease. The North American Symptomatic Carotid Endarterectomy Trial (NASCET)10 and the European Carotid Surgery Trial (ECST)11 provided the basis for stratifying symptomatic patients and determining whether surgery will produce a reasonable benefit. The Asymptomatic Carotid Atherosclerosis Study (ACAS)12 extended the research to asymptomatic patients with high-grade stenosis.8,13 The benefits of CEA for elderly patients (75 and older) with significant comorbidities were supported in the 2009 New York Carotid Artery Surgery Study (NYCAS).14
Researchers for ACAS,12 which compared medical therapy alone with CEA plus medical therapy in asymptomatic patients with CAS, reported a relative risk (RR) reduction of 0.53 in patients undergoing CEA, with a 5.1% five-year rate of stroke or death in the CEA group versus 11.0% among patients receiving medical therapy alone. The Asymptomatic Carotid Surgery Trial15 (ACST) yielded similar event rates (CEA, 6.4%; medical therapy alone, 11.8%). In both trials, the perioperative (30-day) risk of stroke or death associated with CEA ranged from 2.7% to 3.1%. In the long term (five to 10 years and beyond), RR reduction remains uncertain.
However, CEA remains the gold standard for the treatment of severe carotid artery disease. Currently, about 75% of patients who undergo CEA for significant CAS are asymptomatic.13
Complications associated with CEA occur at an ascending rate, commensurate with the patient’s preoperative stroke history. Researchers for the NYCAS14 reported a 30-day post-CEA rate of stroke or death of nearly 3% among asymptomatic patients with no history of stroke or TIA; nearly 8% among patients with previous stroke; and more than 13% in patients with crescendo TIA or evolving stroke. A significant increase in complications (including stroke or death) was reported among patients with coronary artery disease or with diabetes requiring insulin therapy.
The Case for Carotid Angioplasty with Stenting
Though broadly accepted and practiced, CEA carries significant risk for symptomatic patients and for those who face higher surgical risks, such as diabetes or cardiovascular disease, or anatomic issues such as contralateral occlusions (see Table 1).
Carotid angioplasty with stenting emerged in the 1990s as a less invasive alternative to CEA that could be performed under local anesthesia and with little or no sedation. In the Carotid and Vertebral Artery Transluminal Angioplasty Study (CAVATAS),17 no significant difference was found in three-year stroke risk between patients assigned randomly to CEA or to carotid stenting.
The Stenting and Angioplasty with Protection in Patients at High Risk for Endarterectomy (SAPPHIRE) trial,7,16,18 an industry-sponsored study, was the first multicenter study to compare CEA with stenting in patients considered at high surgical risk. All stenting procedures were performed using an intraoperative embolic protection device. Patients with stents had a 4.8% risk of stroke, MI, or death in the 30-day postoperative period, compared with 9.8% among patients who underwent CEA. Despite their potential clinical relevance, these results were not found to be statistically significant.
The risk of ipsilateral stroke at one year was similar between treatment groups. Follow-up data published in 2008 and 2009 showed comparable outcomes and no differences in repeat revascularization rates between CEA and stent groups.7,16 SAPPHIRE is now conducting a worldwide registry study in an effort to extend its results to a broader population. The Center for Medicare Services has approved carotid artery stenting with embolic protection for patients who meet the SAPPHIRE high-risk criteria.
Research on the effectiveness of distal protection devices in preventing intraoperative stroke is ongoing.19 In the interim, the SVS recommends embolic protection during all carotid stenting procedures.13 Perioperative medical management remains critical to the success of carotid stenting. This includes intraoperative heparin and clopidogrel for at least two to four weeks postoperatively.7,16
In elderly patients (80 and older), carotid artery stenting may present a particularly high risk.20,21 Investigators for the ongoing Carotid Revascularization Endarterectomy versus Stenting Trial (CREST) have found a periprocedural risk of death or stroke at 12.1% among older CAS patients, compared with 4.0% among their younger counterparts.21,22 This discrepancy has been attributed to age-related changes in vasculature that create a more hostile environment for endovascular devices.
Patients younger than 80 with significant but asymptomatic unilateral stenosis who are at average surgical risk are presently the focus of other trials. Now under way, the Carotid Angioplasty and Stenting versus Endarterectomy in Asymptomatic Subjects with Significant Extracranial Carotid Occlusive Disease Trial (ACT I) is the first major trial involving asymptomatic patients, and so far has shown a lower postintervention event rate than reported in smaller previous studies.23 However, these patients are less likely to experience postoperative events than their symptomatic counterparts. Thus, the role of stenting in asymptomatic patients will require long-term follow-up.
To date, CEA retains its gold standard status as the optimum surgical treatment for preventing stroke-associated morbidity and mortality. At the same time, carotid stenting is emerging as an effective and less invasive alternative, especially for patients younger than 80 who are at high perioperative risk for CEA.
Treatment Guidelines
In 2008, the SVS13 issued clinical practice guidelines based on an empirical analysis of the currently available research on carotid stenosis. These guidelines address both medical and anatomic risk and acknowledge the limitations of comparing massive data obtained from robust but independent clinical trials. The SVS authors acknowledge that some terms (eg, high perioperative risk) remain somewhat difficult to define and are subject to practitioners’ interpretation.
In an effort to achieve consensus, the SVS investigators employed the British-based GRADE (Grading of Recommendations, Assessment, Development and Evaluation) system24 to stratify the strength of its recommendations. This system takes into account factors other than the quality of the data, including the reviewers’ values and preferences, and their evaluation of the data as presented (see Table 213).
As the SVS authors note, there is no significant difference to date between outcomes for stenting versus CEA, including death or stroke within 30 days postprocedure and the need for revascularization within three years.13 They conclude, nevertheless, that CEA remains the treatment of choice for asymptomatic patients with moderate to severe stenosis. Symptomatic patients can be stratified based on age and medical and surgical risk when a choice is being made between CEA and stenting. In patients at high risk, lifelong pharmacologic therapy may be safer than either surgical or endovascular treatment.
The Role of Medical Management
Whether or not CEA or stenting is performed, medical therapy plays a crucial role in the management of carotid artery disease. Most patients are placed on aspirin therapy indefinitely unless its use is contraindicated (eg, by risk for gastrointestinal bleeding). The SVS practice guidelines13 incorporate medical therapy, citing joint recommendations issued in 2006 by the American Heart Association and the American Stroke Association (AHA/ASA)1,25 for tight control of hypertension, blood glucose, and elevated cholesterol.
The AHA/ASA researchers also recommend antiplatelet agents (aspirin, clopidogrel, and/or dipyridamole) for patients with a history of TIA or noncardioembolic ischemic stroke. The guidelines advise moderate alcohol consumption, weight reduction for obese patients, and increased physical activity. Smoking cessation remains the sine qua non of vascular disease management.1,25
Conclusion
Primary care providers play a pivotal role in identifying patients at risk for carotid artery disease and educating them about current treatment options. They have an opportunity to take a proactive role in screening patients (age 55 and over) who smoke or who have diabetes, high blood pressure, high cholesterol, or coronary artery disease.
Detection of moderate to severe CAS can lead to timely surgical intervention in asymptomatic individuals who may not realize they are at risk. Attentive medical and lifestyle management enhances the treatment of carotid disease and reduces the risk of stroke, its most devastating consequence.
Carotid artery disease is a treatable cause of ischemic stroke, a potentially devastating event that affects approximately 700,000 Americans each year and results in more than 160,000 deaths.1,2 Stroke-related medical costs, including associated disability, now approach $60 billion per year. Despite advances in treatment, stroke remains the third leading cause of death in the United States.3
As the population ages, stroke prevention has become an increasing challenge for primary care providers. Guiding patients at risk toward the appropriate testing and treatment can offer lifelong benefits. This article will summarize current practice recommendations for screening asymptomatic individuals and for treatment of carotid artery disease using carotid endarterectomy or carotid angioplasty with stenting.
Scope and Screening
Carotid artery stenosis (CAS) is defined as atherosclerotic narrowing of the extracranial carotid arteries. Possibly 20% of ischemic strokes (which represent more than 85% of all strokes) result from CAS, a condition that may or may not be symptomatic.4Symptomatic CAS may be represented by a cerebrovascular accident, a transient ischemic attack, or one of an array of more subtle but enduring neurologic deficits.
The prime risk factor for CAS is prior history of cerebrovascular disease.4 Cardiovascular disease or cigarette smoking doubles a patient’s risk for developing CAS. Other risk factors include age greater than 65, male gender, hypertension, atrial fibrillation, and clotting disorders.
Population studies based on carotid ultrasonography estimate the prevalence of CAS at 0.5% to 8.0% in the general population.4-6 Clinically significant CAS (60% or higher) has been estimated at 1% in those older than 65.4
The degree of carotid occlusion correlates directly with the risk of ipsilateral stroke. The rate of stroke among asymptomatic patients with CAS of at least 80% is 3.5% to 5.0% per year.7 To date, there is no clinically useful risk model to identify those who have CAS or will develop it.
Screening the general population for asymptomatic CAS is not currently recommended.4,8 Guidelines published in 2007 by the Society for Vascular Surgery (SVS)3 advise ultrasound screening only for persons 55 and older who have cardiovascular risk factors, including diabetes, hypertension, hypercholesterolemia, a history of smoking, or known cerebrovascular disease. That same year, the American Society of Neuroimaging9 recommended screening of adults 65 or older who have three or more cardiovascular risk factors.
Ultrasound screening is approximately 94% sensitive and 92% specific for moderate to severe CAS (ie, 60% to 90% occlusion).4 Patients with positive ultrasound findings may next undergo computerized axial angiography, magnetic resonance angiography, or digital subtraction angiography.
Angiography can detect with good precision the degree and location of carotid occlusion, which in turn helps to select treatment options, in consideration of their inherent risks and benefits. These options are medical therapy alone, or medical therapy combined with carotid endarterectomy (CEA), or carotid angioplasty with stenting.
The Research
Stroke prevention, long since a medical priority, is most commonly sought by way of pharmacotherapy combined with lifestyle modification. Surgery, in the form of CEA, also plays an enduring and proven role. Randomized trials, including three landmark studies,10-12 have established CEA as standard treatment for symptomatic and high-grade occlusive carotid disease. The North American Symptomatic Carotid Endarterectomy Trial (NASCET)10 and the European Carotid Surgery Trial (ECST)11 provided the basis for stratifying symptomatic patients and determining whether surgery will produce a reasonable benefit. The Asymptomatic Carotid Atherosclerosis Study (ACAS)12 extended the research to asymptomatic patients with high-grade stenosis.8,13 The benefits of CEA for elderly patients (75 and older) with significant comorbidities were supported in the 2009 New York Carotid Artery Surgery Study (NYCAS).14
Researchers for ACAS,12 which compared medical therapy alone with CEA plus medical therapy in asymptomatic patients with CAS, reported a relative risk (RR) reduction of 0.53 in patients undergoing CEA, with a 5.1% five-year rate of stroke or death in the CEA group versus 11.0% among patients receiving medical therapy alone. The Asymptomatic Carotid Surgery Trial15 (ACST) yielded similar event rates (CEA, 6.4%; medical therapy alone, 11.8%). In both trials, the perioperative (30-day) risk of stroke or death associated with CEA ranged from 2.7% to 3.1%. In the long term (five to 10 years and beyond), RR reduction remains uncertain.
However, CEA remains the gold standard for the treatment of severe carotid artery disease. Currently, about 75% of patients who undergo CEA for significant CAS are asymptomatic.13
Complications associated with CEA occur at an ascending rate, commensurate with the patient’s preoperative stroke history. Researchers for the NYCAS14 reported a 30-day post-CEA rate of stroke or death of nearly 3% among asymptomatic patients with no history of stroke or TIA; nearly 8% among patients with previous stroke; and more than 13% in patients with crescendo TIA or evolving stroke. A significant increase in complications (including stroke or death) was reported among patients with coronary artery disease or with diabetes requiring insulin therapy.
The Case for Carotid Angioplasty with Stenting
Though broadly accepted and practiced, CEA carries significant risk for symptomatic patients and for those who face higher surgical risks, such as diabetes or cardiovascular disease, or anatomic issues such as contralateral occlusions (see Table 1).
Carotid angioplasty with stenting emerged in the 1990s as a less invasive alternative to CEA that could be performed under local anesthesia and with little or no sedation. In the Carotid and Vertebral Artery Transluminal Angioplasty Study (CAVATAS),17 no significant difference was found in three-year stroke risk between patients assigned randomly to CEA or to carotid stenting.
The Stenting and Angioplasty with Protection in Patients at High Risk for Endarterectomy (SAPPHIRE) trial,7,16,18 an industry-sponsored study, was the first multicenter study to compare CEA with stenting in patients considered at high surgical risk. All stenting procedures were performed using an intraoperative embolic protection device. Patients with stents had a 4.8% risk of stroke, MI, or death in the 30-day postoperative period, compared with 9.8% among patients who underwent CEA. Despite their potential clinical relevance, these results were not found to be statistically significant.
The risk of ipsilateral stroke at one year was similar between treatment groups. Follow-up data published in 2008 and 2009 showed comparable outcomes and no differences in repeat revascularization rates between CEA and stent groups.7,16 SAPPHIRE is now conducting a worldwide registry study in an effort to extend its results to a broader population. The Center for Medicare Services has approved carotid artery stenting with embolic protection for patients who meet the SAPPHIRE high-risk criteria.
Research on the effectiveness of distal protection devices in preventing intraoperative stroke is ongoing.19 In the interim, the SVS recommends embolic protection during all carotid stenting procedures.13 Perioperative medical management remains critical to the success of carotid stenting. This includes intraoperative heparin and clopidogrel for at least two to four weeks postoperatively.7,16
In elderly patients (80 and older), carotid artery stenting may present a particularly high risk.20,21 Investigators for the ongoing Carotid Revascularization Endarterectomy versus Stenting Trial (CREST) have found a periprocedural risk of death or stroke at 12.1% among older CAS patients, compared with 4.0% among their younger counterparts.21,22 This discrepancy has been attributed to age-related changes in vasculature that create a more hostile environment for endovascular devices.
Patients younger than 80 with significant but asymptomatic unilateral stenosis who are at average surgical risk are presently the focus of other trials. Now under way, the Carotid Angioplasty and Stenting versus Endarterectomy in Asymptomatic Subjects with Significant Extracranial Carotid Occlusive Disease Trial (ACT I) is the first major trial involving asymptomatic patients, and so far has shown a lower postintervention event rate than reported in smaller previous studies.23 However, these patients are less likely to experience postoperative events than their symptomatic counterparts. Thus, the role of stenting in asymptomatic patients will require long-term follow-up.
To date, CEA retains its gold standard status as the optimum surgical treatment for preventing stroke-associated morbidity and mortality. At the same time, carotid stenting is emerging as an effective and less invasive alternative, especially for patients younger than 80 who are at high perioperative risk for CEA.
Treatment Guidelines
In 2008, the SVS13 issued clinical practice guidelines based on an empirical analysis of the currently available research on carotid stenosis. These guidelines address both medical and anatomic risk and acknowledge the limitations of comparing massive data obtained from robust but independent clinical trials. The SVS authors acknowledge that some terms (eg, high perioperative risk) remain somewhat difficult to define and are subject to practitioners’ interpretation.
In an effort to achieve consensus, the SVS investigators employed the British-based GRADE (Grading of Recommendations, Assessment, Development and Evaluation) system24 to stratify the strength of its recommendations. This system takes into account factors other than the quality of the data, including the reviewers’ values and preferences, and their evaluation of the data as presented (see Table 213).
As the SVS authors note, there is no significant difference to date between outcomes for stenting versus CEA, including death or stroke within 30 days postprocedure and the need for revascularization within three years.13 They conclude, nevertheless, that CEA remains the treatment of choice for asymptomatic patients with moderate to severe stenosis. Symptomatic patients can be stratified based on age and medical and surgical risk when a choice is being made between CEA and stenting. In patients at high risk, lifelong pharmacologic therapy may be safer than either surgical or endovascular treatment.
The Role of Medical Management
Whether or not CEA or stenting is performed, medical therapy plays a crucial role in the management of carotid artery disease. Most patients are placed on aspirin therapy indefinitely unless its use is contraindicated (eg, by risk for gastrointestinal bleeding). The SVS practice guidelines13 incorporate medical therapy, citing joint recommendations issued in 2006 by the American Heart Association and the American Stroke Association (AHA/ASA)1,25 for tight control of hypertension, blood glucose, and elevated cholesterol.
The AHA/ASA researchers also recommend antiplatelet agents (aspirin, clopidogrel, and/or dipyridamole) for patients with a history of TIA or noncardioembolic ischemic stroke. The guidelines advise moderate alcohol consumption, weight reduction for obese patients, and increased physical activity. Smoking cessation remains the sine qua non of vascular disease management.1,25
Conclusion
Primary care providers play a pivotal role in identifying patients at risk for carotid artery disease and educating them about current treatment options. They have an opportunity to take a proactive role in screening patients (age 55 and over) who smoke or who have diabetes, high blood pressure, high cholesterol, or coronary artery disease.
Detection of moderate to severe CAS can lead to timely surgical intervention in asymptomatic individuals who may not realize they are at risk. Attentive medical and lifestyle management enhances the treatment of carotid disease and reduces the risk of stroke, its most devastating consequence.
1. Goldstein LB, Adams R, Alberts MJ, et al. Primary prevention of ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council: cosponsored by the Atherosclerotic Peripheral Vascular Disease Interdisciplinary Working Group; Cardiovascular Nursing Council; Clinical Cardiology Council; Nutrition, Physical Activity, and Metabolism Council; and the Quality of Care and Outcomes Research Interdisciplinary Working Group: the American Academy of Neurology affirms the value of this guideline. Stroke. 2006;37(6):1583–1633.
2. American Heart Association. Heart Disease and Stroke Statistics—2004 Update. www.americanheart.org/downloadable/heart/1079736729696HDSStats2004UpdateREV3-19-04.pdf. Accessed December 28, 2009.
3. Society for Vascular Surgery. SVS position statement on vascular screenings. www.vascularweb.org/patients/screenings/SVS_Position_Statement_on_Vascular_Screenings.html. Accessed December 28, 2009.
4. Wolff T, Guirguis-Blake J, Miller T, et al. Screening for carotid artery stenosis: an update of the evidence for the US Preventive Services Task Force. Ann Intern Med. 2007;147(12):860- 870.
5. Colgan MP, Strode GR, Sommer JD, et al. Prevalence of asymptomatic carotid disease: results of duplex scanning in 348 unselected volunteers. J Vasc Surg. 1988;8(6):674-678.
6. Pujia A, Rubba P, Spencer MP. Prevalence of extracranial carotid artery disease detectable by echo-Doppler in an elderly population. Stroke. 1992;23(6):818-822.
7. Gurm HS, Yadav JS, Fayad P, et al; SAPPHIRE Investigators. Long-term results of carotid stenting versus endarterectomy in high-risk patients. N Engl J Med. 2008;358(15):1572-1579.
8. US Preventive Services Task Force. Screening for carotid artery stenosis: US Preventive Services Task Force Recommendation Statement. Ann Intern Med. 2007;147(12):854-859.
9. Qureshi AI, Alexandrov AV, Tegeler CH, et al. Guidelines for screening of extracranial carotid artery disease: a statement for healthcare professionals from the multidisciplinary practice guidelines committee of the American Society of Neuroimaging; cosponsored by the Society of Vascular and Interventional Neurology. J Neuroimaging. 2007;17(1):19-47.
10. Ferguson GG, Eliasziw M, Barr HWK, et al. The North American Symptomatic Carotid Endarterectomy Trial. Stroke. 1999;30(9):1751-1758.
11. Rothwell PM, Gutnikov SA, Warlow CP; European Carotid Surgery Trial. Reanalysis of the final results of the European Carotid Surgery Trial. Stroke. 2003;34(2):514-23.
12. Executive Committee for the Asymptomatic Carotid Atherosclerosis Study. Endarterectomy for asymptomatic carotid artery stenosis. JAMA. 1995;273(18):1421-1428.
13. Hobson RW 2nd, Mackey WC, Ascher E, et al; Society for Vascular Surgery. Management of atherosclerotic carotid artery disease: clinical practice guidelines of the Society for Vascular Surgery. J Vasc Surg. 2008;48(2):480-486.
14. Halm EA, Tuhrim S, Wang JJ, et al. Risk factors for perioperative death and stroke after carotid endarterectomy: results of the New York Carotid Artery Surgery Study. Stroke. 2009;40(1):221-229.
15. Halliday A, Mansfield A, Marro J, et al; MRC Asymptomatic Carotid Surgery Trial (ACST). Prevention of disabling and fatal strokes by successful carotid endarterectomy in patients without recent neurological symptoms: randomised controlled trial. Lancet. 2004;363(9420):1491-1502.
16. Yadav JS, Wholey MH, Kuntz RE, et al; Stenting and Angioplasty with Protection in Patients at High Risk for Endarterectomy Investigators (SAPPHIRE). Protected carotid-artery stenting versus endarterectomy in high-risk patients. N Engl J Med. 2004;351(15):1493-1501.
17. Endovascular versus surgical treatment in patients with carotid stenosis in the Carotid and Vertebral Artery Transluminal Angioplasty Study (CAVATAS): a randomised trial. Lancet. 2001;357: 1729–1737.
18. Massop D, Dave R, Metzger C, et al. Stenting and angioplasty with protection in patients at high-risk for endarterectomy: SAPPHIRE Worldwide Registry first 2,001 patients. Catheter Cardiovasc Interv. 2009;73(2):129-136.
19. Barbato JE, Dillavou E, Horowitz MB, et al. A randomized trial of carotid artery stenting with and without cerebral protection. J Vasc Surg. 2008;47(4):760-765.
20. Suliman A, Greenberg J, Chandra A, et al. Carotid endarterectomy as the criterion standard in high-risk elderly patients. Arch Surg. 2008; 143(8):736-742.
21. Hobson RW 2nd, Howard VJ, Roubin GS, et al. Carotid artery stenting is associated with increased complications in octogenarians: 30-day stroke and death rates in the CREST lead-in phase. J Vasc Surg. 2004;40(6):1106-1111.
22. Lal BK, Brott TG. The Carotid Revascularization Endarterectomy vs. Stenting Trial completes randomization: lessons learned and anticipated results. J Vasc Surg. 2009;50(5):1224-1231.
23. Derdeyn CP. Carotid stenting for asymptomatic carotid stenosis: trial it. Stroke. 2007;38(2 suppl):715-720.
24. Guyatt GH, Oxman AD, Vist GE, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336(7650):924-926.
25. Sacco RL, Adams R, Albers G, et al. Guidelines for prevention of stroke in patients with ischemic stroke or transient ischemic attack: a statement for healthcare professionals from the American Heart Association/American Stroke Association Council on Stroke: co-sponsored by the Council on Cardiovascular Radiology and Intervention: the American Academy of Neurology affirms the value of this guideline. Stroke. 2006;37(2):577-617.
1. Goldstein LB, Adams R, Alberts MJ, et al. Primary prevention of ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council: cosponsored by the Atherosclerotic Peripheral Vascular Disease Interdisciplinary Working Group; Cardiovascular Nursing Council; Clinical Cardiology Council; Nutrition, Physical Activity, and Metabolism Council; and the Quality of Care and Outcomes Research Interdisciplinary Working Group: the American Academy of Neurology affirms the value of this guideline. Stroke. 2006;37(6):1583–1633.
2. American Heart Association. Heart Disease and Stroke Statistics—2004 Update. www.americanheart.org/downloadable/heart/1079736729696HDSStats2004UpdateREV3-19-04.pdf. Accessed December 28, 2009.
3. Society for Vascular Surgery. SVS position statement on vascular screenings. www.vascularweb.org/patients/screenings/SVS_Position_Statement_on_Vascular_Screenings.html. Accessed December 28, 2009.
4. Wolff T, Guirguis-Blake J, Miller T, et al. Screening for carotid artery stenosis: an update of the evidence for the US Preventive Services Task Force. Ann Intern Med. 2007;147(12):860- 870.
5. Colgan MP, Strode GR, Sommer JD, et al. Prevalence of asymptomatic carotid disease: results of duplex scanning in 348 unselected volunteers. J Vasc Surg. 1988;8(6):674-678.
6. Pujia A, Rubba P, Spencer MP. Prevalence of extracranial carotid artery disease detectable by echo-Doppler in an elderly population. Stroke. 1992;23(6):818-822.
7. Gurm HS, Yadav JS, Fayad P, et al; SAPPHIRE Investigators. Long-term results of carotid stenting versus endarterectomy in high-risk patients. N Engl J Med. 2008;358(15):1572-1579.
8. US Preventive Services Task Force. Screening for carotid artery stenosis: US Preventive Services Task Force Recommendation Statement. Ann Intern Med. 2007;147(12):854-859.
9. Qureshi AI, Alexandrov AV, Tegeler CH, et al. Guidelines for screening of extracranial carotid artery disease: a statement for healthcare professionals from the multidisciplinary practice guidelines committee of the American Society of Neuroimaging; cosponsored by the Society of Vascular and Interventional Neurology. J Neuroimaging. 2007;17(1):19-47.
10. Ferguson GG, Eliasziw M, Barr HWK, et al. The North American Symptomatic Carotid Endarterectomy Trial. Stroke. 1999;30(9):1751-1758.
11. Rothwell PM, Gutnikov SA, Warlow CP; European Carotid Surgery Trial. Reanalysis of the final results of the European Carotid Surgery Trial. Stroke. 2003;34(2):514-23.
12. Executive Committee for the Asymptomatic Carotid Atherosclerosis Study. Endarterectomy for asymptomatic carotid artery stenosis. JAMA. 1995;273(18):1421-1428.
13. Hobson RW 2nd, Mackey WC, Ascher E, et al; Society for Vascular Surgery. Management of atherosclerotic carotid artery disease: clinical practice guidelines of the Society for Vascular Surgery. J Vasc Surg. 2008;48(2):480-486.
14. Halm EA, Tuhrim S, Wang JJ, et al. Risk factors for perioperative death and stroke after carotid endarterectomy: results of the New York Carotid Artery Surgery Study. Stroke. 2009;40(1):221-229.
15. Halliday A, Mansfield A, Marro J, et al; MRC Asymptomatic Carotid Surgery Trial (ACST). Prevention of disabling and fatal strokes by successful carotid endarterectomy in patients without recent neurological symptoms: randomised controlled trial. Lancet. 2004;363(9420):1491-1502.
16. Yadav JS, Wholey MH, Kuntz RE, et al; Stenting and Angioplasty with Protection in Patients at High Risk for Endarterectomy Investigators (SAPPHIRE). Protected carotid-artery stenting versus endarterectomy in high-risk patients. N Engl J Med. 2004;351(15):1493-1501.
17. Endovascular versus surgical treatment in patients with carotid stenosis in the Carotid and Vertebral Artery Transluminal Angioplasty Study (CAVATAS): a randomised trial. Lancet. 2001;357: 1729–1737.
18. Massop D, Dave R, Metzger C, et al. Stenting and angioplasty with protection in patients at high-risk for endarterectomy: SAPPHIRE Worldwide Registry first 2,001 patients. Catheter Cardiovasc Interv. 2009;73(2):129-136.
19. Barbato JE, Dillavou E, Horowitz MB, et al. A randomized trial of carotid artery stenting with and without cerebral protection. J Vasc Surg. 2008;47(4):760-765.
20. Suliman A, Greenberg J, Chandra A, et al. Carotid endarterectomy as the criterion standard in high-risk elderly patients. Arch Surg. 2008; 143(8):736-742.
21. Hobson RW 2nd, Howard VJ, Roubin GS, et al. Carotid artery stenting is associated with increased complications in octogenarians: 30-day stroke and death rates in the CREST lead-in phase. J Vasc Surg. 2004;40(6):1106-1111.
22. Lal BK, Brott TG. The Carotid Revascularization Endarterectomy vs. Stenting Trial completes randomization: lessons learned and anticipated results. J Vasc Surg. 2009;50(5):1224-1231.
23. Derdeyn CP. Carotid stenting for asymptomatic carotid stenosis: trial it. Stroke. 2007;38(2 suppl):715-720.
24. Guyatt GH, Oxman AD, Vist GE, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336(7650):924-926.
25. Sacco RL, Adams R, Albers G, et al. Guidelines for prevention of stroke in patients with ischemic stroke or transient ischemic attack: a statement for healthcare professionals from the American Heart Association/American Stroke Association Council on Stroke: co-sponsored by the Council on Cardiovascular Radiology and Intervention: the American Academy of Neurology affirms the value of this guideline. Stroke. 2006;37(2):577-617.
Lateral Epicondylitis: "But I Don't Play Tennis"
Lateral epicondylitis (LE), or tennis elbow, is an overuse syndrome that primary care providers commonly see. For affected patients, LE can represent an extensive problem, as noncompliance with simple conservative therapies commonly prolongs this condition. For most patients, surgical intervention is considered a last resort.
In patients who develop LE, repetitive wrist dorsiflexion with supination and pronation causes overuse of the extensor tendons of the forearm, resulting in subsequent microtears, collagen degeneration, and angiofibroblastic proliferation.1
LE affects men and women equally. It occurs in 1% to 3% of the population but primarily in those ages 40 and older who perform relevant repetitive motion. Considerable improvement or complete resolution of LE symptoms can be achieved with conservative treatment, although six to 24 months’ continuation of such a regimen may be required. Apparent remission of symptoms can be interrupted by recurrences.1
Once a diagnosis of tennis elbow is made, the patient’s response may be, “But I don’t play tennis.”
Patient Presentation and History
Patients with LE usually present with a history of several weeks’ elbow pain of an insidious onset, followed by worsening rather than improvement. Most patients deny any history of direct elbow trauma, although pain can be secondary to an acute event.2
The most commonly reported symptom is increased pain with overhand lifting and point tenderness over the lateral epicondyle or just distal to this area. Frequently patients report weakness or decreased grip strength.
The diagnosis of LE is based on the history and physical exam and may be supported by x-ray findings. Diagnosis may be prefaced by a routine patient history regarding onset of symptoms, aggravating or alleviating factors, hand dominance, occupation, and recreational activities. A more pressing history of recent repetitive motion activities, such as raking leaves, painting, or keyboard use, may illuminate the cause of symptoms.
The clinician should inquire about the effectiveness of any home self-treatments, such as NSAIDs or other pain medication, orthotics (ie, a brace or strap), or other supportive measures. An atypical presentation (eg, elbow pain just distal to or below the lateral epicondyle) might suggest a more complex diagnosis, such as radial tunnel syndrome. Such a case may warrant a more comprehensive exam; referral to an orthopedic specialist would be suggested. A differential diagnosis for LE is shown in the table2,3 below.
Physical Examination
A detailed history will usually direct the physical exam, enhancing its basic principles, and provide a preliminary diagnosis. The examining clinician should begin by observing for any noticeable deformity. Subtle or obvious swelling can be present over the lateral epicondyle, with localized erythema. Elbow joint effusion may indicate intra-articular disease.4
In the assessment for elbow range of motion, 0° (full extension) to 140° of flexion, and 50° of pronation (palm down) and supination (palm up) is required. Instability is checked with the patient’s arm fully extended. The examiner grasps the elbow with both hands and gently applies medial, then lateral pressure, observing for any ligament laxity.
Palpation of the bones should begin over the medial epicondyle and progress to the olecranon, then to the lateral epicondyle. Direct palpation over the lateral epicondyle increases the pressure over the origin of the extensor musculotendinous structures—specifically, the extensor carpi radialis brevis and extensor digitorum tendons. This pressure generally reproduces the pain associated with LE.
The most revealing diagnostic test in the physical exam is resisted extension of a dorsiflexed clenched fist on the affected side (see the figure, below). Other physical tests for assessing this pain are with resisted extension of the long finger and resisted supination of the affected extremity. These maneuvers will elicit distinct pain at the lateral epicondyle and guarding. Neurovascular status should be assessed distally.
A brief exam of the shoulder and wrist on the affected side is suggested for completeness and to rule out other etiologies, particularly in the event of a fall or other traumatic injury.
Radiographic Imaging
Plain film x-rays are obtained to rule out fracture, tumor, or degenerative changes. There is no clear evidence in the literature that plain film x-rays are helpful with the initial diagnosis, and repeat x-rays at subsequent visits are not required when no further trauma has occurred. In cases of extreme LE refractory to conservative treatment, further evaluation with MRI is required.5
Treatment
Treatment for LE is generally conservative. The use of NSAIDs, rest, ice, and a tennis elbow strap (ie, a nonarticular proximal forearm strap or brace) are considered first-line treatment options.1 The purpose of a tennis elbow strap is to relieve pressure over the lateral epicondyle by increasing pressure over the forearm muscles. Correct application of the strap is essential to alleviate pain.
Before turning to surgical intervention (of which a number of options exist for patients whose pain does not respond to conservative treatment6), the clinician may consider use of corticosteroid injections, which are relatively safe and usually have a short-term effect (ie, two to six weeks). Injection therapy for LE is usually considered appropriate for patients with chronic pain and disability that is not relieved by more conservative means, or who experience acute pain with functional impairment.7 Before performing corticosteroid soft-tissue injections, clinicians should consult the appropriate governing agency regarding this advanced practice privilege.
Patients who comply with orthotics and NSAID use and are able to avoid repetitive motion are most likely to see an enduring resolution of symptoms when steroid injections are administered as adjunct therapy. A review of the literature suggests no significant benefit from physical therapy or ultrasonography.8-10
Platelet-Rich Plasma Injections
Local injection of platelet-rich plasma is an alternative based on the understanding that platelets contain growth factors which aid in healing. This has been demonstrated as an effective treatment for LE.11,12 Whole blood obtained from the patient is centrifuged, with platelet-rich plasma then collected for local injection over the lateral epicondyle.
The cost of platelet-rich plasma therapy averages between $300 and $400 per injection (C. Whitney, personal communication, July 14, 2009). According to Mishra et al,12 one injection is ordinarily sufficient.
Patient Education
In-depth education prepares patients for long-term management of LE. Discussion explaining the causes, pathology, duration, and treatment may lead to better self-management for this chronic condition. Exacerbations are easily provoked by return to repetitive activities or direct trauma.
Clinicians who care for patients with LE are urged to emphasize the importance of complying with conservative therapies, avoiding repetitive activities, and adhering to ongoing conservative treatment measures. Follow-up is recommended six weeks after these treatment measures are begun; they should be continued if the patient’s symptoms are improving. Otherwise, further follow-up or referral to an orthopedic specialist can be made at the clinician’s discretion.
Conclusion
Lateral epicondylitis is a common elbow problem that can be diagnosed without difficulty, easily aggravated, and annoying for patients. Noncompliance with recommendations to use orthotics, avoid repetitive activities, and adhere to prescribed medication regimens is the most likely explanation for lack of improvement.
Whether or not your patient plays tennis, appropriate education and compliance with the agreed-on treatment support the optimal outcomes for this vexing condition. The ball is in their court.
1. Johnson GW, Cadwallader K, Scheffel SB, Epperly TD. Treatment of lateral epicondylitis. Am Fam Physician. 2007;76(6):843-848.
2. Murphy KP, Guiliani JR, Freedman BA. Management of lateral epicondylitis in the athlete. Operative Techniques Sports Med. 2006;14(2):67-74.
3. Kaminsky SB, Baker CL Jr. Lateral epicondylitis of the elbow. Tech Hand Up Extrem Surg. 2003;7(4):179-189.
4. Boyer MI, Hastings H 2nd. Lateral tennis elbow: “Is there any science out there?” J Shoulder Elbow Surg. 1999;8(5):481-491.
5. Pfahler M, Jessel C, Steinborn M, Refior HJ. Magnetic resonance imaging in lateral epicondylitis of the elbow. Arch Orthop Trauma Surg. 1998;118(3):121-125.
6. Lo MY, Safran MR. Surgical treatment of lateral epicondylitis: a systematic review. Clin Orthop Relat Res. 2007;463:98-106.
7. Cardone DA, Tallia AF. Diagnostic and therapeutic injection of the elbow region. Am Fam Physician. 2002;66(11):2097-2100.
8. Smidt N, Assendelft WJ, Arola H, et al. Effectiveness of physiotherapy for lateral epicondylitis: a systematic review. Ann Med. 2003;35(1):51-62.
9. Struijs PA, Kerkhoffs GM, Assendelft WJ, Van Dijk CN. Conservative treatment of lateral epicondylitis: brace versus physical therapy or a combination of both: a randomized clinical trial. Am J Sports Med. 2004;32(2):462-469.
10. D’Vaz AP, Ostor AJ, Speed CA, et al. Pulsed low-intensity ultrasound therapy for chronic lateral epicondylitis: a randomized controlled trial. Rheumatology (Oxford). 2006;45(5):566-570.
11. Sampson S, Gerhardt M, Mandelbaum B. Platelet rich plasma injection grafts for musculoskeletal injuries: a review. Curr Rev Musculoskelet Med. 2008;1(3-4):165-174.
12. Mishra A, Pavelko T. Treatment of chronic elbow tendinosis with buffered platelet-rich plasma. Am J Sports Med. 2006;34(11):1774-1778.
Lateral epicondylitis (LE), or tennis elbow, is an overuse syndrome that primary care providers commonly see. For affected patients, LE can represent an extensive problem, as noncompliance with simple conservative therapies commonly prolongs this condition. For most patients, surgical intervention is considered a last resort.
In patients who develop LE, repetitive wrist dorsiflexion with supination and pronation causes overuse of the extensor tendons of the forearm, resulting in subsequent microtears, collagen degeneration, and angiofibroblastic proliferation.1
LE affects men and women equally. It occurs in 1% to 3% of the population but primarily in those ages 40 and older who perform relevant repetitive motion. Considerable improvement or complete resolution of LE symptoms can be achieved with conservative treatment, although six to 24 months’ continuation of such a regimen may be required. Apparent remission of symptoms can be interrupted by recurrences.1
Once a diagnosis of tennis elbow is made, the patient’s response may be, “But I don’t play tennis.”
Patient Presentation and History
Patients with LE usually present with a history of several weeks’ elbow pain of an insidious onset, followed by worsening rather than improvement. Most patients deny any history of direct elbow trauma, although pain can be secondary to an acute event.2
The most commonly reported symptom is increased pain with overhand lifting and point tenderness over the lateral epicondyle or just distal to this area. Frequently patients report weakness or decreased grip strength.
The diagnosis of LE is based on the history and physical exam and may be supported by x-ray findings. Diagnosis may be prefaced by a routine patient history regarding onset of symptoms, aggravating or alleviating factors, hand dominance, occupation, and recreational activities. A more pressing history of recent repetitive motion activities, such as raking leaves, painting, or keyboard use, may illuminate the cause of symptoms.
The clinician should inquire about the effectiveness of any home self-treatments, such as NSAIDs or other pain medication, orthotics (ie, a brace or strap), or other supportive measures. An atypical presentation (eg, elbow pain just distal to or below the lateral epicondyle) might suggest a more complex diagnosis, such as radial tunnel syndrome. Such a case may warrant a more comprehensive exam; referral to an orthopedic specialist would be suggested. A differential diagnosis for LE is shown in the table2,3 below.
Physical Examination
A detailed history will usually direct the physical exam, enhancing its basic principles, and provide a preliminary diagnosis. The examining clinician should begin by observing for any noticeable deformity. Subtle or obvious swelling can be present over the lateral epicondyle, with localized erythema. Elbow joint effusion may indicate intra-articular disease.4
In the assessment for elbow range of motion, 0° (full extension) to 140° of flexion, and 50° of pronation (palm down) and supination (palm up) is required. Instability is checked with the patient’s arm fully extended. The examiner grasps the elbow with both hands and gently applies medial, then lateral pressure, observing for any ligament laxity.
Palpation of the bones should begin over the medial epicondyle and progress to the olecranon, then to the lateral epicondyle. Direct palpation over the lateral epicondyle increases the pressure over the origin of the extensor musculotendinous structures—specifically, the extensor carpi radialis brevis and extensor digitorum tendons. This pressure generally reproduces the pain associated with LE.
The most revealing diagnostic test in the physical exam is resisted extension of a dorsiflexed clenched fist on the affected side (see the figure, below). Other physical tests for assessing this pain are with resisted extension of the long finger and resisted supination of the affected extremity. These maneuvers will elicit distinct pain at the lateral epicondyle and guarding. Neurovascular status should be assessed distally.
A brief exam of the shoulder and wrist on the affected side is suggested for completeness and to rule out other etiologies, particularly in the event of a fall or other traumatic injury.
Radiographic Imaging
Plain film x-rays are obtained to rule out fracture, tumor, or degenerative changes. There is no clear evidence in the literature that plain film x-rays are helpful with the initial diagnosis, and repeat x-rays at subsequent visits are not required when no further trauma has occurred. In cases of extreme LE refractory to conservative treatment, further evaluation with MRI is required.5
Treatment
Treatment for LE is generally conservative. The use of NSAIDs, rest, ice, and a tennis elbow strap (ie, a nonarticular proximal forearm strap or brace) are considered first-line treatment options.1 The purpose of a tennis elbow strap is to relieve pressure over the lateral epicondyle by increasing pressure over the forearm muscles. Correct application of the strap is essential to alleviate pain.
Before turning to surgical intervention (of which a number of options exist for patients whose pain does not respond to conservative treatment6), the clinician may consider use of corticosteroid injections, which are relatively safe and usually have a short-term effect (ie, two to six weeks). Injection therapy for LE is usually considered appropriate for patients with chronic pain and disability that is not relieved by more conservative means, or who experience acute pain with functional impairment.7 Before performing corticosteroid soft-tissue injections, clinicians should consult the appropriate governing agency regarding this advanced practice privilege.
Patients who comply with orthotics and NSAID use and are able to avoid repetitive motion are most likely to see an enduring resolution of symptoms when steroid injections are administered as adjunct therapy. A review of the literature suggests no significant benefit from physical therapy or ultrasonography.8-10
Platelet-Rich Plasma Injections
Local injection of platelet-rich plasma is an alternative based on the understanding that platelets contain growth factors which aid in healing. This has been demonstrated as an effective treatment for LE.11,12 Whole blood obtained from the patient is centrifuged, with platelet-rich plasma then collected for local injection over the lateral epicondyle.
The cost of platelet-rich plasma therapy averages between $300 and $400 per injection (C. Whitney, personal communication, July 14, 2009). According to Mishra et al,12 one injection is ordinarily sufficient.
Patient Education
In-depth education prepares patients for long-term management of LE. Discussion explaining the causes, pathology, duration, and treatment may lead to better self-management for this chronic condition. Exacerbations are easily provoked by return to repetitive activities or direct trauma.
Clinicians who care for patients with LE are urged to emphasize the importance of complying with conservative therapies, avoiding repetitive activities, and adhering to ongoing conservative treatment measures. Follow-up is recommended six weeks after these treatment measures are begun; they should be continued if the patient’s symptoms are improving. Otherwise, further follow-up or referral to an orthopedic specialist can be made at the clinician’s discretion.
Conclusion
Lateral epicondylitis is a common elbow problem that can be diagnosed without difficulty, easily aggravated, and annoying for patients. Noncompliance with recommendations to use orthotics, avoid repetitive activities, and adhere to prescribed medication regimens is the most likely explanation for lack of improvement.
Whether or not your patient plays tennis, appropriate education and compliance with the agreed-on treatment support the optimal outcomes for this vexing condition. The ball is in their court.
Lateral epicondylitis (LE), or tennis elbow, is an overuse syndrome that primary care providers commonly see. For affected patients, LE can represent an extensive problem, as noncompliance with simple conservative therapies commonly prolongs this condition. For most patients, surgical intervention is considered a last resort.
In patients who develop LE, repetitive wrist dorsiflexion with supination and pronation causes overuse of the extensor tendons of the forearm, resulting in subsequent microtears, collagen degeneration, and angiofibroblastic proliferation.1
LE affects men and women equally. It occurs in 1% to 3% of the population but primarily in those ages 40 and older who perform relevant repetitive motion. Considerable improvement or complete resolution of LE symptoms can be achieved with conservative treatment, although six to 24 months’ continuation of such a regimen may be required. Apparent remission of symptoms can be interrupted by recurrences.1
Once a diagnosis of tennis elbow is made, the patient’s response may be, “But I don’t play tennis.”
Patient Presentation and History
Patients with LE usually present with a history of several weeks’ elbow pain of an insidious onset, followed by worsening rather than improvement. Most patients deny any history of direct elbow trauma, although pain can be secondary to an acute event.2
The most commonly reported symptom is increased pain with overhand lifting and point tenderness over the lateral epicondyle or just distal to this area. Frequently patients report weakness or decreased grip strength.
The diagnosis of LE is based on the history and physical exam and may be supported by x-ray findings. Diagnosis may be prefaced by a routine patient history regarding onset of symptoms, aggravating or alleviating factors, hand dominance, occupation, and recreational activities. A more pressing history of recent repetitive motion activities, such as raking leaves, painting, or keyboard use, may illuminate the cause of symptoms.
The clinician should inquire about the effectiveness of any home self-treatments, such as NSAIDs or other pain medication, orthotics (ie, a brace or strap), or other supportive measures. An atypical presentation (eg, elbow pain just distal to or below the lateral epicondyle) might suggest a more complex diagnosis, such as radial tunnel syndrome. Such a case may warrant a more comprehensive exam; referral to an orthopedic specialist would be suggested. A differential diagnosis for LE is shown in the table2,3 below.
Physical Examination
A detailed history will usually direct the physical exam, enhancing its basic principles, and provide a preliminary diagnosis. The examining clinician should begin by observing for any noticeable deformity. Subtle or obvious swelling can be present over the lateral epicondyle, with localized erythema. Elbow joint effusion may indicate intra-articular disease.4
In the assessment for elbow range of motion, 0° (full extension) to 140° of flexion, and 50° of pronation (palm down) and supination (palm up) is required. Instability is checked with the patient’s arm fully extended. The examiner grasps the elbow with both hands and gently applies medial, then lateral pressure, observing for any ligament laxity.
Palpation of the bones should begin over the medial epicondyle and progress to the olecranon, then to the lateral epicondyle. Direct palpation over the lateral epicondyle increases the pressure over the origin of the extensor musculotendinous structures—specifically, the extensor carpi radialis brevis and extensor digitorum tendons. This pressure generally reproduces the pain associated with LE.
The most revealing diagnostic test in the physical exam is resisted extension of a dorsiflexed clenched fist on the affected side (see the figure, below). Other physical tests for assessing this pain are with resisted extension of the long finger and resisted supination of the affected extremity. These maneuvers will elicit distinct pain at the lateral epicondyle and guarding. Neurovascular status should be assessed distally.
A brief exam of the shoulder and wrist on the affected side is suggested for completeness and to rule out other etiologies, particularly in the event of a fall or other traumatic injury.
Radiographic Imaging
Plain film x-rays are obtained to rule out fracture, tumor, or degenerative changes. There is no clear evidence in the literature that plain film x-rays are helpful with the initial diagnosis, and repeat x-rays at subsequent visits are not required when no further trauma has occurred. In cases of extreme LE refractory to conservative treatment, further evaluation with MRI is required.5
Treatment
Treatment for LE is generally conservative. The use of NSAIDs, rest, ice, and a tennis elbow strap (ie, a nonarticular proximal forearm strap or brace) are considered first-line treatment options.1 The purpose of a tennis elbow strap is to relieve pressure over the lateral epicondyle by increasing pressure over the forearm muscles. Correct application of the strap is essential to alleviate pain.
Before turning to surgical intervention (of which a number of options exist for patients whose pain does not respond to conservative treatment6), the clinician may consider use of corticosteroid injections, which are relatively safe and usually have a short-term effect (ie, two to six weeks). Injection therapy for LE is usually considered appropriate for patients with chronic pain and disability that is not relieved by more conservative means, or who experience acute pain with functional impairment.7 Before performing corticosteroid soft-tissue injections, clinicians should consult the appropriate governing agency regarding this advanced practice privilege.
Patients who comply with orthotics and NSAID use and are able to avoid repetitive motion are most likely to see an enduring resolution of symptoms when steroid injections are administered as adjunct therapy. A review of the literature suggests no significant benefit from physical therapy or ultrasonography.8-10
Platelet-Rich Plasma Injections
Local injection of platelet-rich plasma is an alternative based on the understanding that platelets contain growth factors which aid in healing. This has been demonstrated as an effective treatment for LE.11,12 Whole blood obtained from the patient is centrifuged, with platelet-rich plasma then collected for local injection over the lateral epicondyle.
The cost of platelet-rich plasma therapy averages between $300 and $400 per injection (C. Whitney, personal communication, July 14, 2009). According to Mishra et al,12 one injection is ordinarily sufficient.
Patient Education
In-depth education prepares patients for long-term management of LE. Discussion explaining the causes, pathology, duration, and treatment may lead to better self-management for this chronic condition. Exacerbations are easily provoked by return to repetitive activities or direct trauma.
Clinicians who care for patients with LE are urged to emphasize the importance of complying with conservative therapies, avoiding repetitive activities, and adhering to ongoing conservative treatment measures. Follow-up is recommended six weeks after these treatment measures are begun; they should be continued if the patient’s symptoms are improving. Otherwise, further follow-up or referral to an orthopedic specialist can be made at the clinician’s discretion.
Conclusion
Lateral epicondylitis is a common elbow problem that can be diagnosed without difficulty, easily aggravated, and annoying for patients. Noncompliance with recommendations to use orthotics, avoid repetitive activities, and adhere to prescribed medication regimens is the most likely explanation for lack of improvement.
Whether or not your patient plays tennis, appropriate education and compliance with the agreed-on treatment support the optimal outcomes for this vexing condition. The ball is in their court.
1. Johnson GW, Cadwallader K, Scheffel SB, Epperly TD. Treatment of lateral epicondylitis. Am Fam Physician. 2007;76(6):843-848.
2. Murphy KP, Guiliani JR, Freedman BA. Management of lateral epicondylitis in the athlete. Operative Techniques Sports Med. 2006;14(2):67-74.
3. Kaminsky SB, Baker CL Jr. Lateral epicondylitis of the elbow. Tech Hand Up Extrem Surg. 2003;7(4):179-189.
4. Boyer MI, Hastings H 2nd. Lateral tennis elbow: “Is there any science out there?” J Shoulder Elbow Surg. 1999;8(5):481-491.
5. Pfahler M, Jessel C, Steinborn M, Refior HJ. Magnetic resonance imaging in lateral epicondylitis of the elbow. Arch Orthop Trauma Surg. 1998;118(3):121-125.
6. Lo MY, Safran MR. Surgical treatment of lateral epicondylitis: a systematic review. Clin Orthop Relat Res. 2007;463:98-106.
7. Cardone DA, Tallia AF. Diagnostic and therapeutic injection of the elbow region. Am Fam Physician. 2002;66(11):2097-2100.
8. Smidt N, Assendelft WJ, Arola H, et al. Effectiveness of physiotherapy for lateral epicondylitis: a systematic review. Ann Med. 2003;35(1):51-62.
9. Struijs PA, Kerkhoffs GM, Assendelft WJ, Van Dijk CN. Conservative treatment of lateral epicondylitis: brace versus physical therapy or a combination of both: a randomized clinical trial. Am J Sports Med. 2004;32(2):462-469.
10. D’Vaz AP, Ostor AJ, Speed CA, et al. Pulsed low-intensity ultrasound therapy for chronic lateral epicondylitis: a randomized controlled trial. Rheumatology (Oxford). 2006;45(5):566-570.
11. Sampson S, Gerhardt M, Mandelbaum B. Platelet rich plasma injection grafts for musculoskeletal injuries: a review. Curr Rev Musculoskelet Med. 2008;1(3-4):165-174.
12. Mishra A, Pavelko T. Treatment of chronic elbow tendinosis with buffered platelet-rich plasma. Am J Sports Med. 2006;34(11):1774-1778.
1. Johnson GW, Cadwallader K, Scheffel SB, Epperly TD. Treatment of lateral epicondylitis. Am Fam Physician. 2007;76(6):843-848.
2. Murphy KP, Guiliani JR, Freedman BA. Management of lateral epicondylitis in the athlete. Operative Techniques Sports Med. 2006;14(2):67-74.
3. Kaminsky SB, Baker CL Jr. Lateral epicondylitis of the elbow. Tech Hand Up Extrem Surg. 2003;7(4):179-189.
4. Boyer MI, Hastings H 2nd. Lateral tennis elbow: “Is there any science out there?” J Shoulder Elbow Surg. 1999;8(5):481-491.
5. Pfahler M, Jessel C, Steinborn M, Refior HJ. Magnetic resonance imaging in lateral epicondylitis of the elbow. Arch Orthop Trauma Surg. 1998;118(3):121-125.
6. Lo MY, Safran MR. Surgical treatment of lateral epicondylitis: a systematic review. Clin Orthop Relat Res. 2007;463:98-106.
7. Cardone DA, Tallia AF. Diagnostic and therapeutic injection of the elbow region. Am Fam Physician. 2002;66(11):2097-2100.
8. Smidt N, Assendelft WJ, Arola H, et al. Effectiveness of physiotherapy for lateral epicondylitis: a systematic review. Ann Med. 2003;35(1):51-62.
9. Struijs PA, Kerkhoffs GM, Assendelft WJ, Van Dijk CN. Conservative treatment of lateral epicondylitis: brace versus physical therapy or a combination of both: a randomized clinical trial. Am J Sports Med. 2004;32(2):462-469.
10. D’Vaz AP, Ostor AJ, Speed CA, et al. Pulsed low-intensity ultrasound therapy for chronic lateral epicondylitis: a randomized controlled trial. Rheumatology (Oxford). 2006;45(5):566-570.
11. Sampson S, Gerhardt M, Mandelbaum B. Platelet rich plasma injection grafts for musculoskeletal injuries: a review. Curr Rev Musculoskelet Med. 2008;1(3-4):165-174.
12. Mishra A, Pavelko T. Treatment of chronic elbow tendinosis with buffered platelet-rich plasma. Am J Sports Med. 2006;34(11):1774-1778.